US20220047506A1 - Praziquantel Formulations - Google Patents
Praziquantel Formulations Download PDFInfo
- Publication number
- US20220047506A1 US20220047506A1 US16/991,397 US202016991397A US2022047506A1 US 20220047506 A1 US20220047506 A1 US 20220047506A1 US 202016991397 A US202016991397 A US 202016991397A US 2022047506 A1 US2022047506 A1 US 2022047506A1
- Authority
- US
- United States
- Prior art keywords
- praziquantel
- peg
- formulation
- patient
- powdered
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Abandoned
Links
- FSVJFNAIGNNGKK-UHFFFAOYSA-N O=C(C1CCCCC1)N1CC(=O)N2CCC3=C(C=CC=C3)C2C1 Chemical compound O=C(C1CCCCC1)N1CC(=O)N2CCC3=C(C=CC=C3)C2C1 FSVJFNAIGNNGKK-UHFFFAOYSA-N 0.000 description 2
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/08—Solutions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/4985—Pyrazines or piperazines ortho- or peri-condensed with heterocyclic ring systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7028—Compounds having saccharide radicals attached to non-saccharide compounds by glycosidic linkages
- A61K31/7034—Compounds having saccharide radicals attached to non-saccharide compounds by glycosidic linkages attached to a carbocyclic compound, e.g. phloridzin
- A61K31/704—Compounds having saccharide radicals attached to non-saccharide compounds by glycosidic linkages attached to a carbocyclic compound, e.g. phloridzin attached to a condensed carbocyclic ring system, e.g. sennosides, thiocolchicosides, escin, daunorubicin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/10—Alcohols; Phenols; Salts thereof, e.g. glycerol; Polyethylene glycols [PEG]; Poloxamers; PEG/POE alkyl ethers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/12—Carboxylic acids; Salts or anhydrides thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/20—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing sulfur, e.g. dimethyl sulfoxide [DMSO], docusate, sodium lauryl sulfate or aminosulfonic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/26—Carbohydrates, e.g. sugar alcohols, amino sugars, nucleic acids, mono-, di- or oligo-saccharides; Derivatives thereof, e.g. polysorbates, sorbitan fatty acid esters or glycyrrhizin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/30—Macromolecular organic or inorganic compounds, e.g. inorganic polyphosphates
- A61K47/32—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds, e.g. carbomers, poly(meth)acrylates, or polyvinyl pyrrolidone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/30—Macromolecular organic or inorganic compounds, e.g. inorganic polyphosphates
- A61K47/36—Polysaccharides; Derivatives thereof, e.g. gums, starch, alginate, dextrin, hyaluronic acid, chitosan, inulin, agar or pectin
- A61K47/38—Cellulose; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0087—Galenical forms not covered by A61K9/02 - A61K9/7023
- A61K9/0095—Drinks; Beverages; Syrups; Compositions for reconstitution thereof, e.g. powders or tablets to be dispersed in a glass of water; Veterinary drenches
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2009—Inorganic compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2013—Organic compounds, e.g. phospholipids, fats
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/2027—Organic macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyvinyl pyrrolidone, poly(meth)acrylates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/205—Polysaccharides, e.g. alginate, gums; Cyclodextrin
- A61K9/2054—Cellulose; Cellulose derivatives, e.g. hydroxypropyl methylcellulose
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/205—Polysaccharides, e.g. alginate, gums; Cyclodextrin
- A61K9/2059—Starch, including chemically or physically modified derivatives; Amylose; Amylopectin; Dextrin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P33/00—Antiparasitic agents
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02A—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE
- Y02A50/00—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE in human health protection, e.g. against extreme weather
- Y02A50/30—Against vector-borne diseases, e.g. mosquito-borne, fly-borne, tick-borne or waterborne diseases whose impact is exacerbated by climate change
Definitions
- This invention is related to the area of formulations and treatments for parasite infections.
- it relates to praziquantel formulations.
- Praziquantel is a medication used to treat a number of types of parasitic worm infections. Specifically it is used for schistosomiasis, clonorchiasis, opisthorchiasis, tapeworm infections, cysticercosis, hydatid disease, and other fluke infections. Often the treatment with Praziquantel leads to unwanted side effects, such as gastrointestinal discomfort attributed to build up of immobilized or killed parasites. Reported side effects include: headache, dizziness, stomach pain, nausea, tiredness, weakness, joint/muscle pain, loss of appetite, vomiting, and sweating. These side effects can harm the patient and makes the experience of using the drug unpleasant and may discourage patient compliance with prescribed medicine.
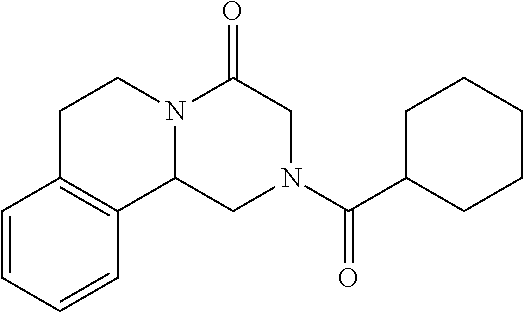
- Praziquantel ((RS)-2-(Cyclohexylcarbonyl)-1,2,3,6,7,11b-hexahydro-4H-pyrazino[2,1-a]isoquinolin-4-one) (C 19 H 24 N 2 O 2 ) is represented as:
- a liquid pharmaceutical formulation comprises: polyethylene glycol (PEG); rubusoside; and praziquantel.
- PEG polyethylene glycol
- rubusoside rubusoside
- praziquantel praziquantel
- Another embodiment is a method of treating an infection by a blood fluke or tapeworm in a patient.
- a liquid pharmaceutical formulation is administered to the patient.
- the formulation comprises: polyethylene glycol (PEG); rubusoside; and praziquantel.
- Yet another embodiment is a powdered formulation of praziquantel for reconstitution in water and subsequent administration to a patient as a liquid formulation.
- the powdered formulation comprises: polyethylene glycol (PEG); rubusoside; and praziquantel.
- a powdered formulation of praziquantel comprises: rubusoside; and praziquantel.
- the inventor has developed a method of formulating praziquantel so that it is easily, accurately, and pleasantly administered and reduces one or more side effects associated with its use. Additional benefits to pharmacokinetic properties may also accrue.
- Praziquantel is only moderately soluble in water. According the Merck Index, its solubility is 400 mg/l. However, the combination of elements in the formulations as disclosed here are able to achieve a higher degree of solubility, permitting liquid dosing in a palatable volume.
- the liquid or powdered formulation comprises: polyethylene glycol (PEG); rubusoside; and praziquantel.
- PEG polyethylene glycol
- rubusoside rubusoside
- praziquantel The pharmacokinetic properties such as absorption may also be altered by this combination.
- the ratio of rubusoside to praziquantel in the formulation may range from about 2:1 to about 10:1. This may be adjusted to achieve a suitable solubility level, gastrointestinal absorption, and agreeable taste profile.
- the combination may be used to form a liquid formulation. In other instances it may be desirable to use it to form a tablet.
- Polyethylene glycol as used in the liquid and powdered formulations has an average molecular weight large enough for the polymer to serve as an osmotic laxative. Typically this is between 2000 and 6000 daltons, between 3000 and 4500, or between 3200 and 3700. Popular commercially available versions are 3350, 4000 and 6000.
- the preparation of PEG may be polydisperse or monodisperse, for example. If polydisperse, then the molecular weight describes the weighted average molecular weight of the preparation.
- the ratio of PEG to praziquantel may range between and including 5:1 and 10:1. In one embodiment the ratio is about 8:1.
- the powdered form may be reconstituted in a liquid vehicle, either at the point of manufacture, at the dispensing pharmacy, or by the patient.
- the liquid vehicle may be water, a buffered aqueous solution, or an aqueous beverage, such as an energy drink or electrolyte rich drink.
- the powdered preparation of rubusoside and praziquantel may be constituted in a tablet or pill—with or without PEG.
- Suitable ingredients for a tablet or pill may include any or all of corn starch, magnesium stearate, microcrystalline cellulose, povidone, sodium lauryl sulfate, polyethylene glycol, titanium dioxide and hypromellose.
- a variety of parasites and the diseases they cause may be treated using the formulations disclosed here. These include schistosomiasis (bilharzia, bilharziasis, or snail fever) caused by schistosomes, fluke infections caused by the trematode Clonorchis sinensis (Chinese or oriental liver fluke), trematodes Opisthorchis viverrini (Southeast Asian liver fluke) and Opisthorchis felineus (cat liver fluke), taeniasis or cysticercosis caused by the tapeworm species Taenia saginata (beef tapeworm), Taenia solium (pork tapeworm), and Taenia asiatica (Asian tapeworm), tiny tapeworms of the genus Echinocococcus causing either cystic echinococcosis (hydatid disease) or alveolar echinococcosis.
- schistosomiasis bilharzia, bil
- Patients which are treated can be either human or veterinary.
- Commonly infected veterinary animals include horse, dog, cat, poultry, cattle, pigs, and ruminants. Any of these can be treated using the formulations disclosed here.
- ingredients are deposited into a sealed container with ethanol and vortex-mixed to form a solution.
- the solution is subjected to centrifugation.
- the ethanol is evaporated off and dried mixture is dissolved in water.
- This mixture is centrifuged again and the supernatant is filtered though a membrane.
- the flow-through is dried the forming the compounded praziquantel.
- the liquid praziquantel it is mixed with Rubusoside to create a water soluble chemical.
- the formulation ratio (10/1) being 100 mg of Rubusoside to 10 mg praziquantel is prepared.
- Ingredients are deposited into a sealed container with 1 ml of ethanol and vortex for 15 minutes to form a solution. Then the mixture is subjected to centrifugation at 12,000 rpm for 10 min. Next, the ethanol is evaporated off and mixture is then dissolved in 1 ml of water. This mixture is centrifuged again at 12,000 rpm for 10 minutes and filtered though a 0.20 ⁇ m membrane and dried. The resulting mixture can be used to make a liquid formulation of praziquantel when reconstituted in water. This gives 110 mg solids in the formula at a 10/1 ratio.
- this formula can vary from 2/1 ratio-10/1 ratio depending on conditions.
- Inactive ingredients and active ingredients are mixed to homogeneity.
- the mixture is then reconstituted with 2.667 oz of purified water and a flavor enhancer such as FLAVORx.
- the dose for an adult human is 20 mg of praziquantel per kg 3 ⁇ daily, e.g., every 5 hours during wakeful hours.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Epidemiology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Engineering & Computer Science (AREA)
- Molecular Biology (AREA)
- Inorganic Chemistry (AREA)
- Biochemistry (AREA)
- Biophysics (AREA)
- Tropical Medicine & Parasitology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
Abstract
Praziquantel may be formulated to enhance its pharmacokinetic, toxicity, and palatability properties. It can be stored and/or dispensed as a liquid, powder, or tablet. Reduction in the most common side effects improves patient compliance and satisfaction. Altered taste profile improves patient compliance and satisfaction. Once formulated it can be used to treat a variety of blood flukes and worms in human and veterinary subjects.
Description
- This invention is related to the area of formulations and treatments for parasite infections. In particular, it relates to praziquantel formulations.
- Praziquantel is a medication used to treat a number of types of parasitic worm infections. Specifically it is used for schistosomiasis, clonorchiasis, opisthorchiasis, tapeworm infections, cysticercosis, hydatid disease, and other fluke infections. Often the treatment with Praziquantel leads to unwanted side effects, such as gastrointestinal discomfort attributed to build up of immobilized or killed parasites. Reported side effects include: headache, dizziness, stomach pain, nausea, tiredness, weakness, joint/muscle pain, loss of appetite, vomiting, and sweating. These side effects can harm the patient and makes the experience of using the drug unpleasant and may discourage patient compliance with prescribed medicine.
- Praziquantel ((RS)-2-(Cyclohexylcarbonyl)-1,2,3,6,7,11b-hexahydro-4H-pyrazino[2,1-a]isoquinolin-4-one) (C19H24N2O2) is represented as:
- There is a continuing need in the art to treat parasites with reduced side effects.
- According to one embodiment of the invention a liquid pharmaceutical formulation is provided. The formulation comprises: polyethylene glycol (PEG); rubusoside; and praziquantel.
- Another embodiment is a method of treating an infection by a blood fluke or tapeworm in a patient. A liquid pharmaceutical formulation is administered to the patient. The formulation comprises: polyethylene glycol (PEG); rubusoside; and praziquantel.
- Yet another embodiment is a powdered formulation of praziquantel for reconstitution in water and subsequent administration to a patient as a liquid formulation. The powdered formulation comprises: polyethylene glycol (PEG); rubusoside; and praziquantel.
- In still another embodiment a powdered formulation of praziquantel is provided. The powdered formulation comprises: rubusoside; and praziquantel.
- These and other embodiments which will be apparent to those of skill in the art upon reading the specification provide the art with improvements in patient compliance, satisfaction, comfort, and overall treatment experience.
- The inventor has developed a method of formulating praziquantel so that it is easily, accurately, and pleasantly administered and reduces one or more side effects associated with its use. Additional benefits to pharmacokinetic properties may also accrue.
- The current practice in the art is to dispense praziquantel as a large tablet that must be split—sometimes into multiple segment—to achieve a proper dose for a patient. By using a powdered or liquid formulation, dispensing proper doses is easier, reducing errors, variations, and waste based on variations in pill splitting technique.
- Praziquantel is only moderately soluble in water. According the Merck Index, its solubility is 400 mg/l. However, the combination of elements in the formulations as disclosed here are able to achieve a higher degree of solubility, permitting liquid dosing in a palatable volume. The liquid or powdered formulation comprises: polyethylene glycol (PEG); rubusoside; and praziquantel. The pharmacokinetic properties such as absorption may also be altered by this combination.
- The ratio of rubusoside to praziquantel in the formulation may range from about 2:1 to about 10:1. This may be adjusted to achieve a suitable solubility level, gastrointestinal absorption, and agreeable taste profile.
- In some cases the combination may be used to form a liquid formulation. In other instances it may be desirable to use it to form a tablet.
- Polyethylene glycol as used in the liquid and powdered formulations has an average molecular weight large enough for the polymer to serve as an osmotic laxative. Typically this is between 2000 and 6000 daltons, between 3000 and 4500, or between 3200 and 3700. Popular commercially available versions are 3350, 4000 and 6000. The preparation of PEG may be polydisperse or monodisperse, for example. If polydisperse, then the molecular weight describes the weighted average molecular weight of the preparation. According to the formulations in powder or liquid form, the ratio of PEG to praziquantel may range between and including 5:1 and 10:1. In one embodiment the ratio is about 8:1.
- For administration, the powdered form may be reconstituted in a liquid vehicle, either at the point of manufacture, at the dispensing pharmacy, or by the patient. The liquid vehicle may be water, a buffered aqueous solution, or an aqueous beverage, such as an energy drink or electrolyte rich drink. Alternatively, the powdered preparation of rubusoside and praziquantel may be constituted in a tablet or pill—with or without PEG. Suitable ingredients for a tablet or pill may include any or all of corn starch, magnesium stearate, microcrystalline cellulose, povidone, sodium lauryl sulfate, polyethylene glycol, titanium dioxide and hypromellose.
- A variety of parasites and the diseases they cause may be treated using the formulations disclosed here. These include schistosomiasis (bilharzia, bilharziasis, or snail fever) caused by schistosomes, fluke infections caused by the trematode Clonorchis sinensis (Chinese or oriental liver fluke), trematodes Opisthorchis viverrini (Southeast Asian liver fluke) and Opisthorchis felineus (cat liver fluke), taeniasis or cysticercosis caused by the tapeworm species Taenia saginata (beef tapeworm), Taenia solium (pork tapeworm), and Taenia asiatica (Asian tapeworm), tiny tapeworms of the genus Echinocococcus causing either cystic echinococcosis (hydatid disease) or alveolar echinococcosis.
- Patients which are treated can be either human or veterinary. Commonly infected veterinary animals include horse, dog, cat, poultry, cattle, pigs, and ruminants. Any of these can be treated using the formulations disclosed here.
- The above disclosure generally describes the present invention. All references disclosed herein are expressly incorporated by reference. A more complete understanding can be obtained by reference to the following specific examples which are provided herein for purposes of illustration only, and are not intended to limit the scope of the invention.
- Compounding of Praziquantel with Rubusoside
- Generally, ingredients are deposited into a sealed container with ethanol and vortex-mixed to form a solution. The solution is subjected to centrifugation. The ethanol is evaporated off and dried mixture is dissolved in water. This mixture is centrifuged again and the supernatant is filtered though a membrane. The flow-through is dried the forming the compounded praziquantel.
- In a specific example of making the liquid praziquantel, it is mixed with Rubusoside to create a water soluble chemical. The formulation ratio (10/1) being 100 mg of Rubusoside to 10 mg praziquantel is prepared. Ingredients are deposited into a sealed container with 1 ml of ethanol and vortex for 15 minutes to form a solution. Then the mixture is subjected to centrifugation at 12,000 rpm for 10 min. Next, the ethanol is evaporated off and mixture is then dissolved in 1 ml of water. This mixture is centrifuged again at 12,000 rpm for 10 minutes and filtered though a 0.20 μm membrane and dried. The resulting mixture can be used to make a liquid formulation of praziquantel when reconstituted in water. This gives 110 mg solids in the formula at a 10/1 ratio.
- To adjust sweetness and solubility this formula can vary from 2/1 ratio-10/1 ratio depending on conditions.
-
-
Praziquantel (liquid recipe 10/1) 7,333.37 mg Glycol 3350 5,666.67 mg Croscarmellose sodium 43.17 mg Providone 43.17 mg Sodium Laurel Sulfate 43.17 mg Magnesium Stearate 43.17 mg Brilliant Blue FCF (Blue1) 43.17 Total 13,216.22 mg - Inactive ingredients and active ingredients (praziquantel liquid recipe 10/1 and Glycol 3350; see Example 1) are mixed to homogeneity. The mixture is then reconstituted with 2.667 oz of purified water and a flavor enhancer such as FLAVORx. The dose for an adult human is 20 mg of praziquantel per kg 3× daily, e.g., every 5 hours during wakeful hours.
Claims (36)
1. A liquid pharmaceutical formulation in a vehicle consisting of an aqueous vehicle, said formulation comprising:
(a) polyethylene glycol (PEG);
(b) rubusoside; and
(c) praziquantel.
2. The liquid pharmaceutical formulation of claim 1 wherein the PEG is polydisperse.
3. The liquid pharmaceutical formulation of claim 1 wherein the PEG has an average molecular weight of 2000-6000.
4. The liquid pharmaceutical formulation of claim 1 wherein the PEG is PEG 3350.
5. The liquid pharmaceutical formulation of claim 1 wherein the weight ratio of PEG to praziquantel is between 5:1 and 10:1.
6. The liquid pharmaceutical formulation of claim 1 wherein the weight ratio of rubusoside to praziquantel in the liquid pharmaceutical formulation is between 2:1 and 10:1.
7. A method of treating an infection by a blood fluke or tapeworm in a patient, comprising:
administering the liquid pharmaceutical formulation of claim 1 to the patient.
8. The method of claim 7 wherein the patient is a human.
9. The method of claim 7 wherein the patient is a veterinary patient.
10. The method of claim 7 wherein the infection is schistosomiasis.
11. The method of claim 7 wherein the blood fluke is Clonorchis sinensis.
12. The method of claim 7 wherein the blood fluke is Opisthorchis viverrini.
13. The method of claim 7 wherein the blood fluke is Opisthorchis felineus.
14. The method of claim 7 wherein the tapeworm is Taenia saginata.
15. The method of claim 7 wherein the tapeworm is Taenia solium.
16. The method of claim 7 wherein the tapeworm is Taenia asiatica.
17. The method of claim 7 wherein the infection is Echinococcosis.
18. The method of claim 7 wherein the patient is selected from the group consisting of a horse, dog, cat, poultry, and ruminant.
19. The method of claim 7 wherein the dose of praziquantel is between 10 and 40 mg per kg of patient weight.
20. The method of claim 7 wherein the patient is human and is administered 3 doses daily of between 10 and 40 mg per kg of patient weight.
21. The method of claim 7 wherein the dose of praziquantel is between 10 and 40 mg per kg of patient weight.
22. The method of claim 7 wherein the patient is human and is administered 3 doses daily of between 10 and 40 mg per kg of patient weight.
23. A powdered formulation of praziquantel and a vehicle consisting of an aqueous vehicle for reconstitution of said powdered formulation and subsequent administration to a patient as an aqueous liquid formulation, said powdered formulation comprising:
(a) polyethylene glycol (PEG);
(b) rubusoside; and
(c) praziquantel.
24. The powdered formulation of claim 23 wherein the PEG is polydisperse.
25. The powdered formulation of claim 23 wherein the PEG has an average molecular weight of 2000-6000.
26. The powdered formulation of claim 23 wherein the PEG is PEG 3350.
27. The powdered formulation of claim 23 wherein the weight ratio of PEG to praziquantel is between 5:1 and 10:1.
28. The powdered formulation of claim 23 wherein the weight ratio of rubusoside to praziquantel in the powdered formulation is between 2:1 and 10:1.
29. (canceled)
30. (canceled)
31. The liquid pharmaceutical formulation of claim 1 which consists of the aqueous vehicle and:
(a) polyethylene glycol (PEG);
(b) rubusoside; and
(c) praziquantel.
32. The powdered formulation and the vehicle of claim 23 wherein the powdered formulation consists of:
(a) polyethylene glycol (PEG);
(b) rubusoside; and
(c) praziquantel.
33. The liquid pharmaceutical formulation of claim 1 which contains one or more components selected from the group consisting of: buffered aqueous solution, an aqueous beverage, croscarmellose sodium, povidone, brilliant blue FCF, and a flavor enhancer.
34. The liquid pharmaceutical formulation of claim 1 which consists of the aqueous vehicle and
(a) polyethylene glycol (PEG);
(b) rubusoside; and
(c) praziquantel, and optionally one or more components selected from the group consisting of buffered aqueous solution, an aqueous beverage, croscarmellose sodium, povidone, brilliant blue FCF, and a flavor enhancer.
35. The powdered formulation and the vehicle of claim 23 wherein the powdered formulation consists of:
(a) polyethylene glycol (PEG);
(b) rubsoside; and
(c) praziquantel; and optionally one or more components selected from the group consisting of: croscarmellose sodium, povidone, brilliant blue FCF, and a flavor enhancer.
36. A method of formulating a powdered formulation of praziquantel for administration to a patient as an aqueous liquid formulation, said method comprising mixing a powdered formulation comprising:
(a) polyethylene glycol (PEG);
(b) rubusoside; and
(c) praziquantel;
with a vehicle consisting of an aqueous vehicle.
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US16/991,397 US20220047506A1 (en) | 2020-08-12 | 2020-08-12 | Praziquantel Formulations |
| PCT/US2021/044665 WO2022035674A1 (en) | 2020-08-12 | 2021-08-05 | Praziquantel formulations |
| EP21856462.3A EP4196225A4 (en) | 2020-08-12 | 2021-08-05 | Praziquantel formulations |
| CN202180055408.7A CN116056704A (en) | 2020-08-12 | 2021-08-05 | Praziquantel preparations |
| US17/583,738 US12390417B2 (en) | 2020-08-12 | 2022-01-25 | Praziquantel formulations |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US16/991,397 US20220047506A1 (en) | 2020-08-12 | 2020-08-12 | Praziquantel Formulations |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US17/583,738 Continuation US12390417B2 (en) | 2020-08-12 | 2022-01-25 | Praziquantel formulations |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| US20220047506A1 true US20220047506A1 (en) | 2022-02-17 |
Family
ID=80223677
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US16/991,397 Abandoned US20220047506A1 (en) | 2020-08-12 | 2020-08-12 | Praziquantel Formulations |
| US17/583,738 Active US12390417B2 (en) | 2020-08-12 | 2022-01-25 | Praziquantel formulations |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US17/583,738 Active US12390417B2 (en) | 2020-08-12 | 2022-01-25 | Praziquantel formulations |
Country Status (4)
| Country | Link |
|---|---|
| US (2) | US20220047506A1 (en) |
| EP (1) | EP4196225A4 (en) |
| CN (1) | CN116056704A (en) |
| WO (1) | WO2022035674A1 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2024158694A1 (en) * | 2023-01-23 | 2024-08-02 | Villya LLC | Compositions and methods for improving the solubility of erectile dysfunction therapeutics |
| WO2024158704A1 (en) * | 2023-01-23 | 2024-08-02 | Villya LLC | Compositions and methods for improving the solubility of drugs and compounds |
Family Cites Families (45)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE3619030A1 (en) | 1986-06-06 | 1987-12-10 | Bayer Ag | Compositions for topical use |
| DE19520275A1 (en) | 1995-06-02 | 1996-12-05 | Bayer Ag | Endoparasiticidal agents |
| US6416779B1 (en) | 1997-06-11 | 2002-07-09 | Umd, Inc. | Device and method for intravaginal or transvaginal treatment of fungal, bacterial, viral or parasitic infections |
| WO1999041233A1 (en) | 1998-02-17 | 1999-08-19 | Micio Pharma Chemical Aktiengesellschaft | A watersoluble nimesulide adduct also for injectable use |
| US6224573B1 (en) | 1999-01-15 | 2001-05-01 | Nexmed Holdings, Inc. | Medicament dispenser |
| WO2000078149A1 (en) * | 1999-06-18 | 2000-12-28 | Bayer Aktiengesellschaft | Anthelmintic compositions |
| WO2001049268A1 (en) | 2000-01-05 | 2001-07-12 | Imarx Therapeutics, Inc. | Pharmaceutical formulations for the delivery of drugs having low aqueous solubility |
| DK1197215T3 (en) | 2000-10-10 | 2006-07-10 | Wyeth Corp | Anthelmintic compositions |
| WO2003087335A2 (en) | 2002-04-11 | 2003-10-23 | Medimmune Vaccines, Inc. | Preservation of bioactive materials by spray drying |
| EP1556009B2 (en) | 2002-10-25 | 2021-07-21 | Foamix Pharmaceuticals Ltd. | Cosmetic and pharmaceutical foam |
| GB0304636D0 (en) | 2003-02-28 | 2003-04-02 | Britannia Pharmaceuticals Ltd | Pharmaceutical composition for nasal delivery |
| MXPA05010659A (en) | 2003-04-04 | 2005-12-12 | Merial Ltd | Topical anthelmintic veterinary formulations. |
| AR045142A1 (en) * | 2003-07-30 | 2005-10-19 | Novartis Ag | BUEN SABOR DUCTILE MASTICABLE VETERINARY COMPOSITION |
| US20060292225A1 (en) | 2005-06-24 | 2006-12-28 | Felix Arthur M | Water soluble analgesic formulations and methods for production |
| WO2007011349A1 (en) | 2005-07-15 | 2007-01-25 | Teva Pharmaceutical Industries Ltd. | Novel granulation process and granulate produced therefrom |
| BRPI0615292A8 (en) | 2005-08-31 | 2018-03-06 | Abraxis Bioscience Llc | compositions and methods for preparing poorly soluble water drugs with increased stability |
| EP2004214B1 (en) | 2006-03-16 | 2012-11-07 | STELLARIS PHARMACEUTICALS Aps | Local treatment with factor vii |
| WO2008077130A2 (en) | 2006-12-19 | 2008-06-26 | Merial Limited | Homogeneous paste and gel formulations |
| RU2009141539A (en) | 2007-04-25 | 2011-05-27 | Тева Фармасьютикал Индастриес Лтд. (Il) | COMPLEX OF PHARMACEUTICAL FILLER |
| JP2010535231A (en) | 2007-07-31 | 2010-11-18 | ワイス・エルエルシー | Internal parasite control topical composition |
| US9333329B2 (en) | 2008-03-04 | 2016-05-10 | Elan Ziv | Vaginal carrier for the controlled release of substances |
| US20110033525A1 (en) | 2008-04-11 | 2011-02-10 | Zhijun Liu | Diterpene Glycosides as Natural Solubilizers |
| US8551507B2 (en) | 2009-06-24 | 2013-10-08 | Board Of Supervisors Of Louisiana State University And Agricultural And Mechanical College | Terpene glycosides and their combinations as solubilizing agents |
| WO2011047227A2 (en) * | 2009-10-15 | 2011-04-21 | Board Of Supervisors Of Louisiana State University And Agricultural And Mechanical College | Water soluble drug-solubilizer powders and their uses |
| GB2477914B (en) | 2010-02-12 | 2012-01-04 | Univ Newcastle | Compounds and methods for biofilm disruption and prevention |
| US8889687B2 (en) * | 2011-03-04 | 2014-11-18 | Concert Pharmaceuticals, Inc. | Deuterated pyrazinoisoquinoline compounds |
| US10201576B2 (en) | 2012-05-18 | 2019-02-12 | Ultratech India Limited | Herbal composition for vaginal treatment |
| ZA201400898B (en) | 2013-02-06 | 2015-12-23 | Cipla Ltd | Pemetrexed complexes and pharmaceutical compositions containing pemetrexed complexes |
| AR095937A1 (en) | 2013-04-05 | 2015-11-25 | Acraf | POTENTIAL OF SOLUBILITY IN GLUCOGEN BASED WATER |
| CN104327077B (en) * | 2013-10-17 | 2015-08-26 | 苏州同力生物医药有限公司 | Levo-praziquantel crystal formation and its preparation method and application |
| ZA201408333B (en) * | 2013-11-14 | 2015-12-23 | Cipla Ltd | Pharmaceutical compositions |
| EP2937092A1 (en) | 2014-04-24 | 2015-10-28 | PharmAlp SA | Anti-candida compositions and uses thereof |
| US10350042B2 (en) | 2014-04-25 | 2019-07-16 | Kimberly-Clark Worldwide, Inc. | Vaginal insert |
| WO2015171319A1 (en) | 2014-05-05 | 2015-11-12 | Repros Therapeutics Inc. | Formulations and methods for vaginal delivery of antiprogestins |
| PT3212169T (en) | 2014-10-31 | 2021-05-06 | Bend Res Inc | Process for forming active domains dispersed in a matrix |
| US20170360791A1 (en) | 2014-12-04 | 2017-12-21 | Astex Pharmaceuticals, Inc. | Pharmaceutical compositions for increasing the bioavailability of poorly soluble drugs |
| WO2016143939A1 (en) * | 2015-03-09 | 2016-09-15 | 서울대학교산학협력단 | Production method for complex of steviol glycoside and sparingly soluble material having improved solubility, and complex of steviol glycoside and sparingly soluble material having improved solubility produced thereby |
| CA3009697C (en) | 2016-01-04 | 2024-05-28 | Jurox Pty Ltd | Drug release device and use |
| CN105816421B (en) | 2016-05-03 | 2019-01-11 | 中国农业科学院兰州畜牧与兽药研究所 | A kind of praziquantel nano-emulsion in-situ gel and its preparation method and application for prevention and cure of schistosomiasis |
| WO2018023037A1 (en) | 2016-07-29 | 2018-02-01 | Renovia Inc. | Devices, systems, and methods for training pelvic floor muscles |
| US20200268027A1 (en) * | 2017-10-06 | 2020-08-27 | Cargill, Incorporated | Stabilized steviol glycoside compositions and uses thereof |
| RU2681214C1 (en) | 2017-10-12 | 2019-03-05 | Федеральное государственное бюджетное научное учреждение "Федеральный научный центр - Всероссийский научно-исследовательский институт экспериментальной ветеринарии имени К.И. Скрябина и Я.Р. Коваленко Российской академии наук | Method of obtaining agent for the treatment of solid-hoofed animals with parasitosis |
| WO2020061584A1 (en) | 2018-09-21 | 2020-03-26 | Msb Holdings, Inc. | Taste-masked dosage forms |
| CA3150301A1 (en) | 2019-09-06 | 2021-03-11 | Stacy ROSS | Palatable granular veterinary compositions |
| US10857151B1 (en) | 2020-02-21 | 2020-12-08 | Villya LLC | Treatment of female genital schistosomiasis |
-
2020
- 2020-08-12 US US16/991,397 patent/US20220047506A1/en not_active Abandoned
-
2021
- 2021-08-05 EP EP21856462.3A patent/EP4196225A4/en not_active Withdrawn
- 2021-08-05 CN CN202180055408.7A patent/CN116056704A/en active Pending
- 2021-08-05 WO PCT/US2021/044665 patent/WO2022035674A1/en not_active Ceased
-
2022
- 2022-01-25 US US17/583,738 patent/US12390417B2/en active Active
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2024158694A1 (en) * | 2023-01-23 | 2024-08-02 | Villya LLC | Compositions and methods for improving the solubility of erectile dysfunction therapeutics |
| WO2024158704A1 (en) * | 2023-01-23 | 2024-08-02 | Villya LLC | Compositions and methods for improving the solubility of drugs and compounds |
| US12115222B2 (en) | 2023-01-23 | 2024-10-15 | Villya LLC | Compositions and methods for improving the solubility of erectile dysfunction therapeutics |
Also Published As
| Publication number | Publication date |
|---|---|
| EP4196225A1 (en) | 2023-06-21 |
| CN116056704A (en) | 2023-05-02 |
| US12390417B2 (en) | 2025-08-19 |
| EP4196225A4 (en) | 2024-08-07 |
| US20220142920A1 (en) | 2022-05-12 |
| WO2022035674A1 (en) | 2022-02-17 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR960011772B1 (en) | Oral dosing formulations of dideoxy purine nucleosides | |
| JP7213882B2 (en) | Fenfluramine preparations adapted to the ketogenic diet | |
| ES2539414T3 (en) | Liquid formulation for deferiprone with appetizing flavor | |
| US12390417B2 (en) | Praziquantel formulations | |
| US8093408B2 (en) | Antidepressant oral pharmaceutical compositions | |
| US20250000984A1 (en) | Diphenhydramine syrup formulation or suspension | |
| AU2002350081B2 (en) | Ribavirin syrup formulations | |
| US8153824B2 (en) | Antidepressant oral liquid compositions | |
| TW201542240A (en) | Liquid pharmaceutical composition for oral administration comprising FEXOFENADINE | |
| EP4543414A1 (en) | An orodispersible pharmaceutical composition of baclofen and its process of preparation | |
| ES2763321T3 (en) | Premix and pharmaceutical composition for oral administration of memantine as a permanent suspension or preparation prior to administration to the patient, optionally by enteral feeding tube and corresponding procedures | |
| US11096890B2 (en) | Chewable dosage forms containing sitagliptin and metformin | |
| RU2778848C2 (en) | The composition of fenfluramine, compatible with a ketogenic diet | |
| EP1894557B1 (en) | Liquid composition for prevention and/or treatment of different bone metabolic diseases, uses thereof, and preparation process therefore | |
| NL2024161B1 (en) | Pharmaceutical liquid composition, kit of parts comprising the pharmaceutical liquid composition, and method for preparing the pharmaceutical liquid composition | |
| NL2024160B1 (en) | Pharmaceutical liquid composition, kit of parts comprising the pharmaceutical liquid composition, and method for preparing the pharmaceutical liquid composition | |
| US20230321248A1 (en) | Syrup formulation or suspension | |
| HK1066719B (en) | Ribavirin syrup formulations |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: VILLYA LLC, FLORIDA Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNOR:MILLER, WILLIAM;REEL/FRAME:053584/0372 Effective date: 20200814 |
|
| STCB | Information on status: application discontinuation |
Free format text: ABANDONED -- FAILURE TO RESPOND TO AN OFFICE ACTION |