US20100301315A1 - Organic electroluminescence element - Google Patents
Organic electroluminescence element Download PDFInfo
- Publication number
- US20100301315A1 US20100301315A1 US12/780,581 US78058110A US2010301315A1 US 20100301315 A1 US20100301315 A1 US 20100301315A1 US 78058110 A US78058110 A US 78058110A US 2010301315 A1 US2010301315 A1 US 2010301315A1
- Authority
- US
- United States
- Prior art keywords
- group
- light
- organic electroluminescence
- layer
- carbon atoms
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Abandoned
Links
- 238000005401 electroluminescence Methods 0.000 title claims abstract description 67
- 239000010410 layer Substances 0.000 claims abstract description 139
- 239000000463 material Substances 0.000 claims abstract description 85
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 claims abstract description 70
- 150000001875 compounds Chemical class 0.000 claims abstract description 35
- 229910052697 platinum Inorganic materials 0.000 claims abstract description 35
- 239000003446 ligand Substances 0.000 claims abstract description 34
- 125000001424 substituent group Chemical group 0.000 claims abstract description 33
- 239000012044 organic layer Substances 0.000 claims abstract description 15
- 125000004093 cyano group Chemical group *C#N 0.000 claims abstract description 7
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims abstract description 6
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims abstract description 5
- 229910052705 radium Inorganic materials 0.000 claims abstract description 5
- 229910052701 rubidium Inorganic materials 0.000 claims abstract description 5
- 229910052757 nitrogen Inorganic materials 0.000 claims description 17
- 125000004433 nitrogen atom Chemical group N* 0.000 claims description 17
- 229910052717 sulfur Inorganic materials 0.000 claims description 17
- 125000004434 sulfur atom Chemical group 0.000 claims description 17
- 125000004430 oxygen atom Chemical group O* 0.000 claims description 15
- 238000004020 luminiscence type Methods 0.000 claims description 3
- 125000004432 carbon atom Chemical group C* 0.000 description 82
- -1 dimethylene group Chemical group 0.000 description 54
- 238000000034 method Methods 0.000 description 35
- 238000002347 injection Methods 0.000 description 26
- 239000007924 injection Substances 0.000 description 26
- 239000000758 substrate Substances 0.000 description 22
- 230000000052 comparative effect Effects 0.000 description 19
- 230000005525 hole transport Effects 0.000 description 18
- 0 CC.CC.[1*]C1=C([2*])C([3*])=C([4*])C2=C1CC1=N3C(=CC=C1)CC1=N4C(=CC=C1)CC1=C(C([8*])=C([7*])C([6*])=C1[5*])[Pt]243 Chemical compound CC.CC.[1*]C1=C([2*])C([3*])=C([4*])C2=C1CC1=N3C(=CC=C1)CC1=N4C(=CC=C1)CC1=C(C([8*])=C([7*])C([6*])=C1[5*])[Pt]243 0.000 description 16
- 229910052751 metal Inorganic materials 0.000 description 15
- 239000002184 metal Substances 0.000 description 15
- 229920005989 resin Polymers 0.000 description 11
- 239000011347 resin Substances 0.000 description 11
- 238000004544 sputter deposition Methods 0.000 description 11
- 238000007740 vapor deposition Methods 0.000 description 9
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 8
- 239000003822 epoxy resin Substances 0.000 description 8
- LNEPOXFFQSENCJ-UHFFFAOYSA-N haloperidol Chemical compound C1CC(O)(C=2C=CC(Cl)=CC=2)CCN1CCCC(=O)C1=CC=C(F)C=C1 LNEPOXFFQSENCJ-UHFFFAOYSA-N 0.000 description 8
- 229920000647 polyepoxide Polymers 0.000 description 8
- 239000012945 sealing adhesive Substances 0.000 description 8
- 229910052782 aluminium Inorganic materials 0.000 description 7
- 229910010272 inorganic material Inorganic materials 0.000 description 7
- 150000004706 metal oxides Chemical class 0.000 description 7
- 239000000203 mixture Substances 0.000 description 7
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 6
- UFVXQDWNSAGPHN-UHFFFAOYSA-K bis[(2-methylquinolin-8-yl)oxy]-(4-phenylphenoxy)alumane Chemical compound [Al+3].C1=CC=C([O-])C2=NC(C)=CC=C21.C1=CC=C([O-])C2=NC(C)=CC=C21.C1=CC([O-])=CC=C1C1=CC=CC=C1 UFVXQDWNSAGPHN-UHFFFAOYSA-K 0.000 description 6
- 239000002274 desiccant Substances 0.000 description 6
- 239000002019 doping agent Substances 0.000 description 6
- 239000011521 glass Substances 0.000 description 6
- 229910044991 metal oxide Inorganic materials 0.000 description 6
- 150000002739 metals Chemical class 0.000 description 6
- 238000002360 preparation method Methods 0.000 description 6
- 238000007639 printing Methods 0.000 description 6
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 5
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 5
- 125000003118 aryl group Chemical group 0.000 description 5
- QVQLCTNNEUAWMS-UHFFFAOYSA-N barium oxide Inorganic materials [Ba]=O QVQLCTNNEUAWMS-UHFFFAOYSA-N 0.000 description 5
- 229910052731 fluorine Inorganic materials 0.000 description 5
- 239000007789 gas Substances 0.000 description 5
- 150000002484 inorganic compounds Chemical class 0.000 description 5
- 150000002894 organic compounds Chemical class 0.000 description 5
- 229910052814 silicon oxide Inorganic materials 0.000 description 5
- 239000010409 thin film Substances 0.000 description 5
- 238000001771 vacuum deposition Methods 0.000 description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 5
- ODINCKMPIJJUCX-UHFFFAOYSA-N Calcium oxide Chemical compound [Ca]=O ODINCKMPIJJUCX-UHFFFAOYSA-N 0.000 description 4
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 4
- 150000007945 N-acyl ureas Chemical group 0.000 description 4
- 150000001342 alkaline earth metals Chemical class 0.000 description 4
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical group [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 4
- 239000011248 coating agent Substances 0.000 description 4
- 238000000576 coating method Methods 0.000 description 4
- 229920001577 copolymer Polymers 0.000 description 4
- 239000010408 film Substances 0.000 description 4
- 239000011737 fluorine Substances 0.000 description 4
- 238000007733 ion plating Methods 0.000 description 4
- PQXKHYXIUOZZFA-UHFFFAOYSA-M lithium fluoride Chemical compound [Li+].[F-] PQXKHYXIUOZZFA-UHFFFAOYSA-M 0.000 description 4
- 238000005259 measurement Methods 0.000 description 4
- UHOVQNZJYSORNB-UHFFFAOYSA-N monobenzene Natural products C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 4
- 239000001301 oxygen Substances 0.000 description 4
- 229910052760 oxygen Inorganic materials 0.000 description 4
- 239000011241 protective layer Substances 0.000 description 4
- 239000002356 single layer Substances 0.000 description 4
- 150000003624 transition metals Chemical group 0.000 description 4
- BPMFPOGUJAAYHL-UHFFFAOYSA-N 9H-Pyrido[2,3-b]indole Chemical compound C1=CC=C2C3=CC=CC=C3NC2=N1 BPMFPOGUJAAYHL-UHFFFAOYSA-N 0.000 description 3
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- 229910000838 Al alloy Inorganic materials 0.000 description 3
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 3
- CPLXHLVBOLITMK-UHFFFAOYSA-N Magnesium oxide Chemical compound [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 description 3
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 3
- 229910052581 Si3N4 Inorganic materials 0.000 description 3
- 229910052783 alkali metal Inorganic materials 0.000 description 3
- 150000001340 alkali metals Chemical class 0.000 description 3
- 125000003277 amino group Chemical group 0.000 description 3
- 230000004888 barrier function Effects 0.000 description 3
- 239000000292 calcium oxide Substances 0.000 description 3
- 235000012255 calcium oxide Nutrition 0.000 description 3
- 229910052799 carbon Inorganic materials 0.000 description 3
- 239000000945 filler Substances 0.000 description 3
- 229910052737 gold Inorganic materials 0.000 description 3
- 239000010931 gold Substances 0.000 description 3
- 125000005843 halogen group Chemical group 0.000 description 3
- RAXXELZNTBOGNW-UHFFFAOYSA-N imidazole Natural products C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 description 3
- 238000007641 inkjet printing Methods 0.000 description 3
- 229910052741 iridium Inorganic materials 0.000 description 3
- GKOZUEZYRPOHIO-UHFFFAOYSA-N iridium atom Chemical compound [Ir] GKOZUEZYRPOHIO-UHFFFAOYSA-N 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 230000003287 optical effect Effects 0.000 description 3
- IEQIEDJGQAUEQZ-UHFFFAOYSA-N phthalocyanine Chemical class N1C(N=C2C3=CC=CC=C3C(N=C3C4=CC=CC=C4C(=N4)N3)=N2)=C(C=CC=C2)C2=C1N=C1C2=CC=CC=C2C4=N1 IEQIEDJGQAUEQZ-UHFFFAOYSA-N 0.000 description 3
- 238000007789 sealing Methods 0.000 description 3
- HQVNEWCFYHHQES-UHFFFAOYSA-N silicon nitride Chemical compound N12[Si]34N5[Si]62N3[Si]51N64 HQVNEWCFYHHQES-UHFFFAOYSA-N 0.000 description 3
- 229910052709 silver Inorganic materials 0.000 description 3
- 238000001228 spectrum Methods 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- 229910052723 transition metal Inorganic materials 0.000 description 3
- VERMWGQSKPXSPZ-BUHFOSPRSA-N 1-[(e)-2-phenylethenyl]anthracene Chemical compound C=1C=CC2=CC3=CC=CC=C3C=C2C=1\C=C\C1=CC=CC=C1 VERMWGQSKPXSPZ-BUHFOSPRSA-N 0.000 description 2
- VQGHOUODWALEFC-UHFFFAOYSA-N 2-phenylpyridine Chemical compound C1=CC=CC=C1C1=CC=CC=N1 VQGHOUODWALEFC-UHFFFAOYSA-N 0.000 description 2
- UJOBWOGCFQCDNV-UHFFFAOYSA-N 9H-carbazole Chemical compound C1=CC=C2C3=CC=CC=C3NC2=C1 UJOBWOGCFQCDNV-UHFFFAOYSA-N 0.000 description 2
- 229910001148 Al-Li alloy Inorganic materials 0.000 description 2
- KLZUFWVZNOTSEM-UHFFFAOYSA-K Aluminium flouride Chemical compound F[Al](F)F KLZUFWVZNOTSEM-UHFFFAOYSA-K 0.000 description 2
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical group [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 2
- 229910052693 Europium Inorganic materials 0.000 description 2
- 229910052688 Gadolinium Inorganic materials 0.000 description 2
- SIKJAQJRHWYJAI-UHFFFAOYSA-N Indole Chemical compound C1=CC=C2NC=CC2=C1 SIKJAQJRHWYJAI-UHFFFAOYSA-N 0.000 description 2
- TWRXJAOTZQYOKJ-UHFFFAOYSA-L Magnesium chloride Chemical compound [Mg+2].[Cl-].[Cl-] TWRXJAOTZQYOKJ-UHFFFAOYSA-L 0.000 description 2
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 2
- UFWIBTONFRDIAS-UHFFFAOYSA-N Naphthalene Chemical compound C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 2
- 229910052779 Neodymium Inorganic materials 0.000 description 2
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 2
- 239000004642 Polyimide Substances 0.000 description 2
- KAESVJOAVNADME-UHFFFAOYSA-N Pyrrole Chemical compound C=1C=CNC=1 KAESVJOAVNADME-UHFFFAOYSA-N 0.000 description 2
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical group [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 2
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Thiophene Chemical compound C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 description 2
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 2
- 229910052769 Ytterbium Inorganic materials 0.000 description 2
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 description 2
- JFBZPFYRPYOZCQ-UHFFFAOYSA-N [Li].[Al] Chemical compound [Li].[Al] JFBZPFYRPYOZCQ-UHFFFAOYSA-N 0.000 description 2
- 239000002250 absorbent Substances 0.000 description 2
- 230000002745 absorbent Effects 0.000 description 2
- 238000010521 absorption reaction Methods 0.000 description 2
- YRKCREAYFQTBPV-UHFFFAOYSA-N acetylacetone Chemical compound CC(=O)CC(C)=O YRKCREAYFQTBPV-UHFFFAOYSA-N 0.000 description 2
- 125000003545 alkoxy group Chemical group 0.000 description 2
- 125000000217 alkyl group Chemical group 0.000 description 2
- 229910045601 alloy Inorganic materials 0.000 description 2
- 239000000956 alloy Substances 0.000 description 2
- VSCWAEJMTAWNJL-UHFFFAOYSA-K aluminium trichloride Chemical compound Cl[Al](Cl)Cl VSCWAEJMTAWNJL-UHFFFAOYSA-K 0.000 description 2
- 125000004397 aminosulfonyl group Chemical group NS(=O)(=O)* 0.000 description 2
- 150000004982 aromatic amines Chemical class 0.000 description 2
- 125000004104 aryloxy group Chemical group 0.000 description 2
- 125000005334 azaindolyl group Chemical class N1N=C(C2=CC=CC=C12)* 0.000 description 2
- WZJYKHNJTSNBHV-UHFFFAOYSA-N benzo[h]quinoline Chemical compound C1=CN=C2C3=CC=CC=C3C=CC2=C1 WZJYKHNJTSNBHV-UHFFFAOYSA-N 0.000 description 2
- IOJUPLGTWVMSFF-UHFFFAOYSA-N benzothiazole Chemical compound C1=CC=C2SC=NC2=C1 IOJUPLGTWVMSFF-UHFFFAOYSA-N 0.000 description 2
- XJHCXCQVJFPJIK-UHFFFAOYSA-M caesium fluoride Chemical compound [F-].[Cs+] XJHCXCQVJFPJIK-UHFFFAOYSA-M 0.000 description 2
- BRPQOXSCLDDYGP-UHFFFAOYSA-N calcium oxide Chemical compound [O-2].[Ca+2] BRPQOXSCLDDYGP-UHFFFAOYSA-N 0.000 description 2
- OSGAYBCDTDRGGQ-UHFFFAOYSA-L calcium sulfate Chemical compound [Ca+2].[O-]S([O-])(=O)=O OSGAYBCDTDRGGQ-UHFFFAOYSA-L 0.000 description 2
- 238000004364 calculation method Methods 0.000 description 2
- 125000003917 carbamoyl group Chemical group [H]N([H])C(*)=O 0.000 description 2
- 150000001716 carbazoles Chemical class 0.000 description 2
- 125000000609 carbazolyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3NC12)* 0.000 description 2
- 230000015556 catabolic process Effects 0.000 description 2
- 238000006243 chemical reaction Methods 0.000 description 2
- 229910052801 chlorine Inorganic materials 0.000 description 2
- 229910052681 coesite Inorganic materials 0.000 description 2
- 239000003086 colorant Substances 0.000 description 2
- 238000004891 communication Methods 0.000 description 2
- 229940052810 complex b Drugs 0.000 description 2
- 239000004020 conductor Substances 0.000 description 2
- 150000004696 coordination complex Chemical class 0.000 description 2
- 229910052906 cristobalite Inorganic materials 0.000 description 2
- 239000013078 crystal Substances 0.000 description 2
- 238000006731 degradation reaction Methods 0.000 description 2
- 238000000151 deposition Methods 0.000 description 2
- 230000008021 deposition Effects 0.000 description 2
- 238000010586 diagram Methods 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 238000005530 etching Methods 0.000 description 2
- OGPBJKLSAFTDLK-UHFFFAOYSA-N europium atom Chemical compound [Eu] OGPBJKLSAFTDLK-UHFFFAOYSA-N 0.000 description 2
- 238000000605 extraction Methods 0.000 description 2
- 150000008376 fluorenones Chemical class 0.000 description 2
- UIWYJDYFSGRHKR-UHFFFAOYSA-N gadolinium atom Chemical compound [Gd] UIWYJDYFSGRHKR-UHFFFAOYSA-N 0.000 description 2
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 2
- 125000000623 heterocyclic group Chemical group 0.000 description 2
- 150000007857 hydrazones Chemical class 0.000 description 2
- 229910052738 indium Inorganic materials 0.000 description 2
- AMGQUBHHOARCQH-UHFFFAOYSA-N indium;oxotin Chemical compound [In].[Sn]=O AMGQUBHHOARCQH-UHFFFAOYSA-N 0.000 description 2
- 239000011147 inorganic material Substances 0.000 description 2
- 229910052747 lanthanoid Inorganic materials 0.000 description 2
- 150000002602 lanthanoids Chemical group 0.000 description 2
- 239000011133 lead Substances 0.000 description 2
- 239000000395 magnesium oxide Substances 0.000 description 2
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 2
- 125000000250 methylamino group Chemical group [H]N(*)C([H])([H])[H] 0.000 description 2
- 125000004170 methylsulfonyl group Chemical group [H]C([H])([H])S(*)(=O)=O 0.000 description 2
- JKQOBWVOAYFWKG-UHFFFAOYSA-N molybdenum trioxide Chemical compound O=[Mo](=O)=O JKQOBWVOAYFWKG-UHFFFAOYSA-N 0.000 description 2
- QEFYFXOXNSNQGX-UHFFFAOYSA-N neodymium atom Chemical compound [Nd] QEFYFXOXNSNQGX-UHFFFAOYSA-N 0.000 description 2
- 229910052759 nickel Inorganic materials 0.000 description 2
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 2
- 150000004866 oxadiazoles Chemical class 0.000 description 2
- 150000007978 oxazole derivatives Chemical class 0.000 description 2
- 229920001721 polyimide Polymers 0.000 description 2
- 229920001296 polysiloxane Polymers 0.000 description 2
- 229920000123 polythiophene Polymers 0.000 description 2
- JEXVQSWXXUJEMA-UHFFFAOYSA-N pyrazol-3-one Chemical compound O=C1C=CN=N1 JEXVQSWXXUJEMA-UHFFFAOYSA-N 0.000 description 2
- 230000009467 reduction Effects 0.000 description 2
- 229910052702 rhenium Inorganic materials 0.000 description 2
- WUAPFZMCVAUBPE-UHFFFAOYSA-N rhenium atom Chemical compound [Re] WUAPFZMCVAUBPE-UHFFFAOYSA-N 0.000 description 2
- 239000004065 semiconductor Substances 0.000 description 2
- 229910052710 silicon Inorganic materials 0.000 description 2
- 239000010703 silicon Substances 0.000 description 2
- 239000000377 silicon dioxide Substances 0.000 description 2
- 239000004332 silver Substances 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- PJANXHGTPQOBST-UHFFFAOYSA-N stilbene Chemical compound C=1C=CC=CC=1C=CC1=CC=CC=C1 PJANXHGTPQOBST-UHFFFAOYSA-N 0.000 description 2
- 229910052682 stishovite Inorganic materials 0.000 description 2
- IATRAKWUXMZMIY-UHFFFAOYSA-N strontium oxide Chemical compound [O-2].[Sr+2] IATRAKWUXMZMIY-UHFFFAOYSA-N 0.000 description 2
- 229940042055 systemic antimycotics triazole derivative Drugs 0.000 description 2
- 229920001187 thermosetting polymer Polymers 0.000 description 2
- 150000003577 thiophenes Chemical class 0.000 description 2
- 229910052718 tin Inorganic materials 0.000 description 2
- 239000011135 tin Substances 0.000 description 2
- 229910001887 tin oxide Inorganic materials 0.000 description 2
- 229910052905 tridymite Inorganic materials 0.000 description 2
- 238000005406 washing Methods 0.000 description 2
- NAWDYIZEMPQZHO-UHFFFAOYSA-N ytterbium Chemical compound [Yb] NAWDYIZEMPQZHO-UHFFFAOYSA-N 0.000 description 2
- 229910001233 yttria-stabilized zirconia Inorganic materials 0.000 description 2
- UWRZIZXBOLBCON-VOTSOKGWSA-N (e)-2-phenylethenamine Chemical class N\C=C\C1=CC=CC=C1 UWRZIZXBOLBCON-VOTSOKGWSA-N 0.000 description 1
- POILWHVDKZOXJZ-ARJAWSKDSA-M (z)-4-oxopent-2-en-2-olate Chemical compound C\C([O-])=C\C(C)=O POILWHVDKZOXJZ-ARJAWSKDSA-M 0.000 description 1
- UPVJEODAZWTJKZ-UHFFFAOYSA-N 1,2-dichloro-1,2-difluoroethene Chemical class FC(Cl)=C(F)Cl UPVJEODAZWTJKZ-UHFFFAOYSA-N 0.000 description 1
- GEYOCULIXLDCMW-UHFFFAOYSA-N 1,2-phenylenediamine Chemical compound NC1=CC=CC=C1N GEYOCULIXLDCMW-UHFFFAOYSA-N 0.000 description 1
- BCMCBBGGLRIHSE-UHFFFAOYSA-N 1,3-benzoxazole Chemical compound C1=CC=C2OC=NC2=C1 BCMCBBGGLRIHSE-UHFFFAOYSA-N 0.000 description 1
- 125000005978 1-naphthyloxy group Chemical group 0.000 description 1
- BAXOFTOLAUCFNW-UHFFFAOYSA-N 1H-indazole Chemical compound C1=CC=C2C=NNC2=C1 BAXOFTOLAUCFNW-UHFFFAOYSA-N 0.000 description 1
- FZKCAHQKNJXICB-UHFFFAOYSA-N 2,1-benzoxazole Chemical group C1=CC=CC2=CON=C21 FZKCAHQKNJXICB-UHFFFAOYSA-N 0.000 description 1
- YQTCQNIPQMJNTI-UHFFFAOYSA-N 2,2-dimethylpropan-1-one Chemical group CC(C)(C)[C]=O YQTCQNIPQMJNTI-UHFFFAOYSA-N 0.000 description 1
- WGLLSSPDPJPLOR-UHFFFAOYSA-N 2,3-dimethylbut-2-ene Chemical group CC(C)=C(C)C WGLLSSPDPJPLOR-UHFFFAOYSA-N 0.000 description 1
- MVWPVABZQQJTPL-UHFFFAOYSA-N 2,3-diphenylcyclohexa-2,5-diene-1,4-dione Chemical class O=C1C=CC(=O)C(C=2C=CC=CC=2)=C1C1=CC=CC=C1 MVWPVABZQQJTPL-UHFFFAOYSA-N 0.000 description 1
- 125000004974 2-butenyl group Chemical group C(C=CC)* 0.000 description 1
- XNMQEEKYCVKGBD-UHFFFAOYSA-N 2-butyne Chemical group CC#CC XNMQEEKYCVKGBD-UHFFFAOYSA-N 0.000 description 1
- NEAQRZUHTPSBBM-UHFFFAOYSA-N 2-hydroxy-3,3-dimethyl-7-nitro-4h-isoquinolin-1-one Chemical compound C1=C([N+]([O-])=O)C=C2C(=O)N(O)C(C)(C)CC2=C1 NEAQRZUHTPSBBM-UHFFFAOYSA-N 0.000 description 1
- 125000005979 2-naphthyloxy group Chemical group 0.000 description 1
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 1
- 125000001494 2-propynyl group Chemical group [H]C#CC([H])([H])* 0.000 description 1
- 125000000590 4-methylphenyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)C([H])([H])[H] 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical group [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- ZYASLTYCYTYKFC-UHFFFAOYSA-N 9-methylidenefluorene Chemical class C1=CC=C2C(=C)C3=CC=CC=C3C2=C1 ZYASLTYCYTYKFC-UHFFFAOYSA-N 0.000 description 1
- 239000004925 Acrylic resin Substances 0.000 description 1
- 229920000178 Acrylic resin Polymers 0.000 description 1
- 229910001316 Ag alloy Inorganic materials 0.000 description 1
- ROFVEXUMMXZLPA-UHFFFAOYSA-N Bipyridyl Chemical group N1=CC=CC=C1C1=CC=CC=N1 ROFVEXUMMXZLPA-UHFFFAOYSA-N 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical group [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- NSXJEEMTGWMJPY-UHFFFAOYSA-N C1=CC(C2=CC(N3C4=C(C=CC=C4)C4=C3C=CC=C4)=CC=C2)=CC(N2C3=C(C=CC=C3)C3=C2C=CC=C3)=C1 Chemical compound C1=CC(C2=CC(N3C4=C(C=CC=C4)C4=C3C=CC=C4)=CC=C2)=CC(N2C3=C(C=CC=C3)C3=C2C=CC=C3)=C1 NSXJEEMTGWMJPY-UHFFFAOYSA-N 0.000 description 1
- PQAONLRZGHWORA-QXDODRCKSA-N C1=CC(C2=CC(N3C4=C(C=CC=C4)C4=C3C=CC=C4)=CC=C2)=CC(N2C3=C(C=CC=C3)C3=C2C=CC=C3)=C1.[2H]C1=C([2H])C2=C(C([2H])=C1[2H])N(C1=C([2H])C(N3C4=C(C([2H])=C([2H])C([2H])=C4[2H])C(C(C)(C)C)C3[2H])=C([2H])C([2H])=C1[2H])/C([2H])=C\2C(C)(C)C.[2H]C1=C([2H])C2=C(C([2H])=C1[2H])N(C1=C([2H])C([2H])=C([2H])C(N3C4=C(C([2H])=C([2H])C([2H])=C4[2H])C4=C3C([2H])=C([2H])C([2H])=C4[2H])=C1[2H])C1=C2C([2H])=C([2H])C([2H])=C1[2H].[2H]C1=C([2H])C2=C(C([2H])=C1[2H])N(C1=CC=CC(C3=CC=CC(N4C5=C(C([2H])=C([2H])C([2H])=C5[2H])C5=C4C([2H])=C([2H])C([2H])=C5[2H])=C3)=C1)C1=C2C([2H])=C([2H])C([2H])=C1[2H] Chemical compound C1=CC(C2=CC(N3C4=C(C=CC=C4)C4=C3C=CC=C4)=CC=C2)=CC(N2C3=C(C=CC=C3)C3=C2C=CC=C3)=C1.[2H]C1=C([2H])C2=C(C([2H])=C1[2H])N(C1=C([2H])C(N3C4=C(C([2H])=C([2H])C([2H])=C4[2H])C(C(C)(C)C)C3[2H])=C([2H])C([2H])=C1[2H])/C([2H])=C\2C(C)(C)C.[2H]C1=C([2H])C2=C(C([2H])=C1[2H])N(C1=C([2H])C([2H])=C([2H])C(N3C4=C(C([2H])=C([2H])C([2H])=C4[2H])C4=C3C([2H])=C([2H])C([2H])=C4[2H])=C1[2H])C1=C2C([2H])=C([2H])C([2H])=C1[2H].[2H]C1=C([2H])C2=C(C([2H])=C1[2H])N(C1=CC=CC(C3=CC=CC(N4C5=C(C([2H])=C([2H])C([2H])=C5[2H])C5=C4C([2H])=C([2H])C([2H])=C5[2H])=C3)=C1)C1=C2C([2H])=C([2H])C([2H])=C1[2H] PQAONLRZGHWORA-QXDODRCKSA-N 0.000 description 1
- LKYSJNVRNPXXPR-UHFFFAOYSA-N C1=CC(N2C3=C(C=CC=C3)C3=C(C=CC=C3)C3=C2C=CC=C3)=CC(N2C3=C(C=CC=C3)C3=C(C=CC=C3)C3=C2C=CC=C3)=C1.C1=CC2=C(C=C1)C1=C(C=CC=C1)N(C1=CC(N3C4=C(C=CC=C4)C4=C(C=CC=C4)C4=C3C=CC=C4)=CC(N3C4=C(C=CC=C4)C4=C(C=CC=C4)C4=C3C=CC=C4)=C1)C1=C2C=CC=C1.C1=CC2=C(C=C1)C1=C(C=CC=C1)N(C1=CC=C(C3=CC(C4=CC=C(N5C6=C(C=CC=C6)C6=C(C=CC=C6)C6=C5C=CC=C6)C=C4)=CC(C4=CC=C(N5C6=C(C=CC=C6)C6=C(C=CC=C6)C6=C5C=CC=C6)C=C4)=C3)C=C1)C1=C2C=CC=C1.C1=CC2=C(C=C1)C1=C(C=CC=C1)N(C1=CC=C(N(C3=CC=C(N4C5=C(C=CC=C5)C5=C(C=CC=C5)C5=C4C=CC=C5)C=C3)C3=CC=C(N4C5=C(C=CC=C5)C5=C(C=CC=C5)C5=C4C=CC=C5)C=C3)C=C1)C1=C2C=CC=C1 Chemical compound C1=CC(N2C3=C(C=CC=C3)C3=C(C=CC=C3)C3=C2C=CC=C3)=CC(N2C3=C(C=CC=C3)C3=C(C=CC=C3)C3=C2C=CC=C3)=C1.C1=CC2=C(C=C1)C1=C(C=CC=C1)N(C1=CC(N3C4=C(C=CC=C4)C4=C(C=CC=C4)C4=C3C=CC=C4)=CC(N3C4=C(C=CC=C4)C4=C(C=CC=C4)C4=C3C=CC=C4)=C1)C1=C2C=CC=C1.C1=CC2=C(C=C1)C1=C(C=CC=C1)N(C1=CC=C(C3=CC(C4=CC=C(N5C6=C(C=CC=C6)C6=C(C=CC=C6)C6=C5C=CC=C6)C=C4)=CC(C4=CC=C(N5C6=C(C=CC=C6)C6=C(C=CC=C6)C6=C5C=CC=C6)C=C4)=C3)C=C1)C1=C2C=CC=C1.C1=CC2=C(C=C1)C1=C(C=CC=C1)N(C1=CC=C(N(C3=CC=C(N4C5=C(C=CC=C5)C5=C(C=CC=C5)C5=C4C=CC=C5)C=C3)C3=CC=C(N4C5=C(C=CC=C5)C5=C(C=CC=C5)C5=C4C=CC=C5)C=C3)C=C1)C1=C2C=CC=C1 LKYSJNVRNPXXPR-UHFFFAOYSA-N 0.000 description 1
- CGBJKYUUERKFEN-UHFFFAOYSA-N C1=CC(N2C3=C(C=CC=C3)C3=C2C=CC=C3)=CC(N2C3=C(C=CC=C3)C3=C2C=CC=C3)=C1.C1=CC2=C(C=C1)N(C1=CC(N3C4=C(C=CC=C4)C4=C3C=CC=C4)=CC(N3C4=C(C=CC=C4)C4=C3C=CC=C4)=C1)C1=C2C=CC=C1.C1=CC2=C(C=C1)N(C1=CC=C(C3=CC=C(N4C5=C(C=CC=C5)C5=C4C=CC=C5)C=C3)C=C1)C1=C2C=CC=C1.C1=CC=C(C2=CC(N3C4=C(C=CC=C4)C4=C3C=CC=C4)=CC=C2C2=CC=C(N3C4=C(C=CC=C4)C4=C3C=CC=C4)C=C2C2=CC=CC=C2)C=C1.CC1=CC(N2C3=C(C=CC=C3)C3=C2C=CC=C3)=CC=C1C1=CC=C(N2C3=C(C=CC=C3)C3=C2C=CC=C3)C=C1C Chemical compound C1=CC(N2C3=C(C=CC=C3)C3=C2C=CC=C3)=CC(N2C3=C(C=CC=C3)C3=C2C=CC=C3)=C1.C1=CC2=C(C=C1)N(C1=CC(N3C4=C(C=CC=C4)C4=C3C=CC=C4)=CC(N3C4=C(C=CC=C4)C4=C3C=CC=C4)=C1)C1=C2C=CC=C1.C1=CC2=C(C=C1)N(C1=CC=C(C3=CC=C(N4C5=C(C=CC=C5)C5=C4C=CC=C5)C=C3)C=C1)C1=C2C=CC=C1.C1=CC=C(C2=CC(N3C4=C(C=CC=C4)C4=C3C=CC=C4)=CC=C2C2=CC=C(N3C4=C(C=CC=C4)C4=C3C=CC=C4)C=C2C2=CC=CC=C2)C=C1.CC1=CC(N2C3=C(C=CC=C3)C3=C2C=CC=C3)=CC=C1C1=CC=C(N2C3=C(C=CC=C3)C3=C2C=CC=C3)C=C1C CGBJKYUUERKFEN-UHFFFAOYSA-N 0.000 description 1
- SARBNIMTLZAYRF-UHFFFAOYSA-N C1=CC2=C(C=C1)C1=C(C=CC=C1)N(C1=CC=C(C3=CC=C(N4C5=C(C=CC=C5)C5=C(C=CC=C5)C5=C4C=CC=C5)C=C3)C=C1)C1=C2C=CC=C1.C1=CC=C(N(C2=CC=C(N(C3=CC=C(N(C4=CC=CC=C4)C4=CC=CC5=C4C=CC=C5)C=C3)C3=CC=C(N(C4=CC=CC=C4)C4=CC=CC5=C4C=CC=C5)C=C3)C=C2)C2=CC=CC3=C2C=CC=C3)C=C1.CC1=C(C2=C(C)C=C(N3C4=C(C=CC=C4)C4=C(C=CC=C4)C4=C3C=CC=C4)C=C2)C=CC(N2C3=C(C=CC=C3)C3=C(C=CC=C3)C3=C2C=CC=C3)=C1.CC1=CC=CC(N(C2=CC=CC=C2)C2=CC=C(N(C3=CC=C(N(C4=CC=CC=C4)C4=CC(C)=CC=C4)C=C3)C3=CC=C(N(C4=CC=CC=C4)C4=CC(C)=CC=C4)C=C3)C=C2)=C1 Chemical compound C1=CC2=C(C=C1)C1=C(C=CC=C1)N(C1=CC=C(C3=CC=C(N4C5=C(C=CC=C5)C5=C(C=CC=C5)C5=C4C=CC=C5)C=C3)C=C1)C1=C2C=CC=C1.C1=CC=C(N(C2=CC=C(N(C3=CC=C(N(C4=CC=CC=C4)C4=CC=CC5=C4C=CC=C5)C=C3)C3=CC=C(N(C4=CC=CC=C4)C4=CC=CC5=C4C=CC=C5)C=C3)C=C2)C2=CC=CC3=C2C=CC=C3)C=C1.CC1=C(C2=C(C)C=C(N3C4=C(C=CC=C4)C4=C(C=CC=C4)C4=C3C=CC=C4)C=C2)C=CC(N2C3=C(C=CC=C3)C3=C(C=CC=C3)C3=C2C=CC=C3)=C1.CC1=CC=CC(N(C2=CC=CC=C2)C2=CC=C(N(C3=CC=C(N(C4=CC=CC=C4)C4=CC(C)=CC=C4)C=C3)C3=CC=C(N(C4=CC=CC=C4)C4=CC(C)=CC=C4)C=C3)C=C2)=C1 SARBNIMTLZAYRF-UHFFFAOYSA-N 0.000 description 1
- ZXCRIXHGJSYRMZ-UHFFFAOYSA-N C1=CC2=C(C=C1)C1=C(S2)C2=CC=CC3=N2[Pt]12C1=C(SC4=C1C=CC=C4)C1=N2C(=CC=C1)C3.C1=CC2=CC3=N(C=C2C=C1)[Pt@]12C4=C(C=C5/C=C\C=C/C5=C4CC4=C1C3=CC1=C4C=CC=C1)C1=CC3=C(C=CC=C3)C=N12.C1=CC=C2C(=C1)C1=N3C(=CC=C1)CC1=N4C(=CC=C1)C1=C(C=CC=C1)[Pt]243.C1=CC=C2C(=C1)C1=N3C4=C(C=CC5=C4C4=N6C(=CC=C4/C=C\5)C4=C(C=CC=C4)[Pt]236)C=C1.C1=CC=C2C(=C1)C1=N3C4=C(C=CC=C4C=C1)C1=N4C(=CC=C1)C1=C(C=CC=C1)[Pt]243.C1=CC=N2C(=C1)C1=C3C(=CC=C1)CC1=C4C(=CC=C1)C1=N(C=CC=C1)[Pt@@]432.O=C1C2=CC=CC3=C2[Pt@@]2(C4=C1C=CC=C4C1=N2C=CC2=C1C=CC=C2)N1=CC=C2C=CC=CC2=C31.O=C1C2=CC=CC3=N2[Pt]2(C4=CC=NC=C43)C3=C(C=NC=C3)C3=CC=CC1=N32 Chemical compound C1=CC2=C(C=C1)C1=C(S2)C2=CC=CC3=N2[Pt]12C1=C(SC4=C1C=CC=C4)C1=N2C(=CC=C1)C3.C1=CC2=CC3=N(C=C2C=C1)[Pt@]12C4=C(C=C5/C=C\C=C/C5=C4CC4=C1C3=CC1=C4C=CC=C1)C1=CC3=C(C=CC=C3)C=N12.C1=CC=C2C(=C1)C1=N3C(=CC=C1)CC1=N4C(=CC=C1)C1=C(C=CC=C1)[Pt]243.C1=CC=C2C(=C1)C1=N3C4=C(C=CC5=C4C4=N6C(=CC=C4/C=C\5)C4=C(C=CC=C4)[Pt]236)C=C1.C1=CC=C2C(=C1)C1=N3C4=C(C=CC=C4C=C1)C1=N4C(=CC=C1)C1=C(C=CC=C1)[Pt]243.C1=CC=N2C(=C1)C1=C3C(=CC=C1)CC1=C4C(=CC=C1)C1=N(C=CC=C1)[Pt@@]432.O=C1C2=CC=CC3=C2[Pt@@]2(C4=C1C=CC=C4C1=N2C=CC2=C1C=CC=C2)N1=CC=C2C=CC=CC2=C31.O=C1C2=CC=CC3=N2[Pt]2(C4=CC=NC=C43)C3=C(C=NC=C3)C3=CC=CC1=N32 ZXCRIXHGJSYRMZ-UHFFFAOYSA-N 0.000 description 1
- IXRKNBPWHRYFNR-UHFFFAOYSA-N C1=CC2=C(C=C1)N(C1=CC=C(C3=CC(C4=CC=C(N5C6=C(C=CC=C6)C6=C5C=CC=C6)C=C4)=CC(C4=CC=C(N5C6=C(C=CC=C6)C6=C5C=CC=C6)C=C4)=C3)C=C1)C1=C2C=CC=C1.C1=CC2=C(C=C1)N(C1=CC=C(N(C3=CC=C(N4C5=C(C=CC=C5)C5=C4C=CC=C5)C=C3)C3=CC=C(N4C5=C(C=CC=C5)C5=C4C=CC=C5)C=C3)C=C1)C1=C2C=CC=C1.C1=CC=C([Si](C2=CC=CC=C2)(C2=CC=CC=C2)C2=CC(N3C4=C(C=CC=C4)C4=C3C=CC=C4)=CC(N3C4=C(C=CC=C4)C4=C3C=CC=C4)=C2)C=C1 Chemical compound C1=CC2=C(C=C1)N(C1=CC=C(C3=CC(C4=CC=C(N5C6=C(C=CC=C6)C6=C5C=CC=C6)C=C4)=CC(C4=CC=C(N5C6=C(C=CC=C6)C6=C5C=CC=C6)C=C4)=C3)C=C1)C1=C2C=CC=C1.C1=CC2=C(C=C1)N(C1=CC=C(N(C3=CC=C(N4C5=C(C=CC=C5)C5=C4C=CC=C5)C=C3)C3=CC=C(N4C5=C(C=CC=C5)C5=C4C=CC=C5)C=C3)C=C1)C1=C2C=CC=C1.C1=CC=C([Si](C2=CC=CC=C2)(C2=CC=CC=C2)C2=CC(N3C4=C(C=CC=C4)C4=C3C=CC=C4)=CC(N3C4=C(C=CC=C4)C4=C3C=CC=C4)=C2)C=C1 IXRKNBPWHRYFNR-UHFFFAOYSA-N 0.000 description 1
- GMFOPVMWPBUCGT-DVPJIXMTSA-L C1=CC2=N3C(=C1)C1=CC=C/C4=C/C5=CC=CC6=N5[Pt]3(N14)N1C6=CC=C/C1=C/2.C1=CC=C(C2=CC3=N4C5=C2/C=C\C2=C(C6=CC=CC=C6)C=C6C7=C(C=CC=C7)O[Pt]4(OC4=CC=CC=C43)N6=C25)C=C1.CC1(C)C2=CC3=C(C=CC=C3)C3=C2[Pt]24C5=C(C=C6C=CC=CC6=C5C(C)(C)C5=N2C3=CC2=C5C=CC=C2)C2=C3C=CC=CC3=CC1=N24.CC1(C)C2=CC=CC3=C2[Pt]24C5=C(C=CC=C5C(C)(C)C5=N2C3=C2C=CC=CC2=C5)C2=C3C=CC=CC3=CC1=N24.CC1(C)C2=CC=CC3=C2[Pt]24C5=C(C=CC=C5C(C)(C)C5=N2C3=CC2=C5C=CC=C2)C2=CC3=C(C=CC=C3)C1=N24.CC1(C)C2=CC=CC3=C2[Pt]24C5=C(C=CC=C5C(C)(C)C5=N2C3=CC=C5)C2=CC=CC1=N24 Chemical compound C1=CC2=N3C(=C1)C1=CC=C/C4=C/C5=CC=CC6=N5[Pt]3(N14)N1C6=CC=C/C1=C/2.C1=CC=C(C2=CC3=N4C5=C2/C=C\C2=C(C6=CC=CC=C6)C=C6C7=C(C=CC=C7)O[Pt]4(OC4=CC=CC=C43)N6=C25)C=C1.CC1(C)C2=CC3=C(C=CC=C3)C3=C2[Pt]24C5=C(C=C6C=CC=CC6=C5C(C)(C)C5=N2C3=CC2=C5C=CC=C2)C2=C3C=CC=CC3=CC1=N24.CC1(C)C2=CC=CC3=C2[Pt]24C5=C(C=CC=C5C(C)(C)C5=N2C3=C2C=CC=CC2=C5)C2=C3C=CC=CC3=CC1=N24.CC1(C)C2=CC=CC3=C2[Pt]24C5=C(C=CC=C5C(C)(C)C5=N2C3=CC2=C5C=CC=C2)C2=CC3=C(C=CC=C3)C1=N24.CC1(C)C2=CC=CC3=C2[Pt]24C5=C(C=CC=C5C(C)(C)C5=N2C3=CC=C5)C2=CC=CC1=N24 GMFOPVMWPBUCGT-DVPJIXMTSA-L 0.000 description 1
- WLHZMSUDUIKWSY-UHFFFAOYSA-N C1=CC=C(C(C2=CC=CC=C2)(C2=CC=C(N3C4=C(C=CC=C4)C4=C3C=CC=C4)C=C2)C2=CC=C(N3C4=C(C=CC=C4)C4=C3C=CC=C4)C=C2)C=C1.C1=CC=C(C2=C(C3=CC(N4C5=C(C=CC=C5)C5=C4C=CC=C5)=CC=C3)C=CC=C2)C(C2=CC(N3C4=C(C=CC=C4)C4=C3C=CC=C4)=CC=C2)=C1.C1=CC=C(C2=C(N3C4=C(C=CC=C4)C4=C3C=CC=C4)C=CC=C2)C(C2=C(C3=C(N4C5=C(C=CC=C5)C5=C4C=CC=C5)C=CC=C3)C=CC=C2)=C1.C1=CC=C([Si](C2=CC=CC=C2)(C2=CC=C(N3C4=C(C=CC=C4)C4=C3C=CC=C4)C=C2)C2=CC=C(N3C4=C(C=CC=C4)C4=C3C=CC=C4)C=C2)C=C1 Chemical compound C1=CC=C(C(C2=CC=CC=C2)(C2=CC=C(N3C4=C(C=CC=C4)C4=C3C=CC=C4)C=C2)C2=CC=C(N3C4=C(C=CC=C4)C4=C3C=CC=C4)C=C2)C=C1.C1=CC=C(C2=C(C3=CC(N4C5=C(C=CC=C5)C5=C4C=CC=C5)=CC=C3)C=CC=C2)C(C2=CC(N3C4=C(C=CC=C4)C4=C3C=CC=C4)=CC=C2)=C1.C1=CC=C(C2=C(N3C4=C(C=CC=C4)C4=C3C=CC=C4)C=CC=C2)C(C2=C(C3=C(N4C5=C(C=CC=C5)C5=C4C=CC=C5)C=CC=C3)C=CC=C2)=C1.C1=CC=C([Si](C2=CC=CC=C2)(C2=CC=C(N3C4=C(C=CC=C4)C4=C3C=CC=C4)C=C2)C2=CC=C(N3C4=C(C=CC=C4)C4=C3C=CC=C4)C=C2)C=C1 WLHZMSUDUIKWSY-UHFFFAOYSA-N 0.000 description 1
- ZCNGYDFBOFFVNO-UHFFFAOYSA-N C1=CC=C(C2=C(C3=CC=C(N4C5=C(C=CC=C5)C5=C4C=CC=C5)C=C3)C=CC=C2)C(C2=CC=C(N3C4=C(C=CC=C4)C4=C3C=CC=C4)C=C2)=C1.C1=CC=C(C2=C(C3=CC=CC=C3)C(C3=CC=C(N4C5=C(C=CC=C5)C5=C4C=CC=C5)C=C3)=C(C3=CC=C(N4C5=C(C=CC=C5)C5=C4C=CC=C5)C=C3)C(C3=CC=CC=C3)=C2C2=CC=CC=C2)C=C1.C1=CC=C(C2=C(C3=CC=CC=C3)C(C3=CC=C(N4C5=C(C=CC=C5)C5=C4C=CC=C5)C=C3)=C(C3=CC=CC=C3)C(C3=CC=CC=C3)=C2C2=CC=C(N3C4=C(C=CC=C4)C4=C3C=CC=C4)C=C2)C=C1 Chemical compound C1=CC=C(C2=C(C3=CC=C(N4C5=C(C=CC=C5)C5=C4C=CC=C5)C=C3)C=CC=C2)C(C2=CC=C(N3C4=C(C=CC=C4)C4=C3C=CC=C4)C=C2)=C1.C1=CC=C(C2=C(C3=CC=CC=C3)C(C3=CC=C(N4C5=C(C=CC=C5)C5=C4C=CC=C5)C=C3)=C(C3=CC=C(N4C5=C(C=CC=C5)C5=C4C=CC=C5)C=C3)C(C3=CC=CC=C3)=C2C2=CC=CC=C2)C=C1.C1=CC=C(C2=C(C3=CC=CC=C3)C(C3=CC=C(N4C5=C(C=CC=C5)C5=C4C=CC=C5)C=C3)=C(C3=CC=CC=C3)C(C3=CC=CC=C3)=C2C2=CC=C(N3C4=C(C=CC=C4)C4=C3C=CC=C4)C=C2)C=C1 ZCNGYDFBOFFVNO-UHFFFAOYSA-N 0.000 description 1
- ZFNQNWKGUZUPPO-OYPNLFBBSA-H C1=CC=C(C2=CC3=N4C(=CC=C3)C3=N5C(=CC=C3)/C=C(/C3=CC=CC=C3)O[Pt]45O2)C=C1.C1=CC=C2C(=C1)C1=N3C(=CC4=C1C=CC=C4)CC1=N4C(=C5C=CC=CC5=C1)C1=C(C=CC=C1)[Pt]243.C1=CC=C2C(=C1)O[Pt]13OC4=C(C=CC=C4)C4=CC=CC(=N41)C1=CC=CC2=N13.C1=CC=C2O[Pt]34OC5=C(C=CC=C5)C=N3C3=CC=CC=C3/N4=C/C2=C1.CN1C2=C(C=CC=C2)C2=C1C1=CC=CC3=N1[Pt]21C2=C(C4=N1C(=CC=C4)C3)N(C)C1=C2C=CC=C1.COC1=CC=C2C(=C1)C1=N3C(=C4/C=C\C=C/C4=C1)CC1=N4C(=CC5=C1C=CC=C5)C1=C(C=CC(OC)=C1)[Pt]234 Chemical compound C1=CC=C(C2=CC3=N4C(=CC=C3)C3=N5C(=CC=C3)/C=C(/C3=CC=CC=C3)O[Pt]45O2)C=C1.C1=CC=C2C(=C1)C1=N3C(=CC4=C1C=CC=C4)CC1=N4C(=C5C=CC=CC5=C1)C1=C(C=CC=C1)[Pt]243.C1=CC=C2C(=C1)O[Pt]13OC4=C(C=CC=C4)C4=CC=CC(=N41)C1=CC=CC2=N13.C1=CC=C2O[Pt]34OC5=C(C=CC=C5)C=N3C3=CC=CC=C3/N4=C/C2=C1.CN1C2=C(C=CC=C2)C2=C1C1=CC=CC3=N1[Pt]21C2=C(C4=N1C(=CC=C4)C3)N(C)C1=C2C=CC=C1.COC1=CC=C2C(=C1)C1=N3C(=C4/C=C\C=C/C4=C1)CC1=N4C(=CC5=C1C=CC=C5)C1=C(C=CC(OC)=C1)[Pt]234 ZFNQNWKGUZUPPO-OYPNLFBBSA-H 0.000 description 1
- RIUXAMSMGVCBGW-UHFFFAOYSA-N C1=CC=C(N(C2=CC=C(C3=CC=C(N(C4=CC=CC=C4)C4=C5C=CC=CC5=CC=C4)C=C3)C=C2)C2=C3C=CC=CC3=CC=C2)C=C1.C1=CC=C(N(C2=CC=C(N(C3=CC=C(N(C4=CC=CC=C4)C4=CC5=C(C=CC=C5)C=C4)C=C3)C3=CC=C(N(C4=CC=CC=C4)C4=CC5=C(C=CC=C5)C=C4)C=C3)C=C2)C2=CC3=C(C=CC=C3)C=C2)C=C1.CC1=CC=CC(N(C2=CC=C(N(C3=CC=CC=C3)C3=CC=C(C4=CC=C(N(C5=CC=CC=C5)C5=CC=C(N(C6=CC=CC(C)=C6)C6=CC(C)=CC=C6)C=C5)C=C4)C=C3)C=C2)C2=CC=CC(C)=C2)=C1 Chemical compound C1=CC=C(N(C2=CC=C(C3=CC=C(N(C4=CC=CC=C4)C4=C5C=CC=CC5=CC=C4)C=C3)C=C2)C2=C3C=CC=CC3=CC=C2)C=C1.C1=CC=C(N(C2=CC=C(N(C3=CC=C(N(C4=CC=CC=C4)C4=CC5=C(C=CC=C5)C=C4)C=C3)C3=CC=C(N(C4=CC=CC=C4)C4=CC5=C(C=CC=C5)C=C4)C=C3)C=C2)C2=CC3=C(C=CC=C3)C=C2)C=C1.CC1=CC=CC(N(C2=CC=C(N(C3=CC=CC=C3)C3=CC=C(C4=CC=C(N(C5=CC=CC=C5)C5=CC=C(N(C6=CC=CC(C)=C6)C6=CC(C)=CC=C6)C=C5)C=C4)C=C3)C=C2)C2=CC=CC(C)=C2)=C1 RIUXAMSMGVCBGW-UHFFFAOYSA-N 0.000 description 1
- FTZJCFJUIGHHMZ-UHFFFAOYSA-N C1=CC=C(N2C3=C4C=CC=CC4=CC4=N3[Pt]3(C5=CC=CC=C54)C4=C(C=CC=C4)C4=CC5=C(/C=C\C=C/5)C2=N43)C=C1.C1=CC=C(N2C3=CC=CC4=C3[Pt]3(C5=C2C=CC=C5C2=CC=CC=N23)N2=C4C=CC=C2)C=C1.C1=CC=C(N2C3=CC=CC4=N3[Pt]3(C5=C4SC=C5)C4C=C[SH]=C4C4=CC=CC2=N43)C=C1.C1=CC=C(N2C3=CC=CC4=N3[Pt]3(C5=CC=C6C=CC=CC6=C54)C4=C(C5=CC=CC2=N53)C2=C(C=CC=C2)C=C4)C=C1.C=C1C2=CC=CC3=N2[Pt]2(C4=CC=CC=C4C4=CC=CC1=N42)C1=C3C=CC=C1.O=C1C2=CC=CC3=N2[Pt]2(C4=CC=CC=C4C4=CC=CC1=N42)C1=C3C=CC=C1 Chemical compound C1=CC=C(N2C3=C4C=CC=CC4=CC4=N3[Pt]3(C5=CC=CC=C54)C4=C(C=CC=C4)C4=CC5=C(/C=C\C=C/5)C2=N43)C=C1.C1=CC=C(N2C3=CC=CC4=C3[Pt]3(C5=C2C=CC=C5C2=CC=CC=N23)N2=C4C=CC=C2)C=C1.C1=CC=C(N2C3=CC=CC4=N3[Pt]3(C5=C4SC=C5)C4C=C[SH]=C4C4=CC=CC2=N43)C=C1.C1=CC=C(N2C3=CC=CC4=N3[Pt]3(C5=CC=C6C=CC=CC6=C54)C4=C(C5=CC=CC2=N53)C2=C(C=CC=C2)C=C4)C=C1.C=C1C2=CC=CC3=N2[Pt]2(C4=CC=CC=C4C4=CC=CC1=N42)C1=C3C=CC=C1.O=C1C2=CC=CC3=N2[Pt]2(C4=CC=CC=C4C4=CC=CC1=N42)C1=C3C=CC=C1 FTZJCFJUIGHHMZ-UHFFFAOYSA-N 0.000 description 1
- ATCJKDSZGULBON-UHFFFAOYSA-N C1=CC=C(N2C3=CC4=C(C=CC=C4)C4=N3[Pt]3(C5=CC=CC=C54)C4=C(C=CC=C4)C4=C5C=CC=CC5=CC2=N43)C=C1.C1=CC=C(N2C3=N4C(=CC=C3)C3=CC=CC=C3[Pt]43C4=C(C=CC=C4)C4=CC=CC2=N43)C=C1.C1=CC=C2C(=C1)C1=CC=CC3=N1[Pt]14N2C2=C(C=CC=C2)N1C1=C(C=CC=C1)C1=CC=CC3=N14.CC1(C)C2=CC=CC3=C2[Pt]24C5=C(C=CC6=C5C5=C(C=CC7=CC=C3N2=C75)C=C6)C2=CC=CC1=N24.CC1(C)C2=CC=CC3=C2[Pt]24C5=C1C=CC=C5C1=N2C(=CN=C1)C(C)(C)C1=N4C3=CN=C1.CC1(C)C2=CC=CC3=N2[Pt]2(C4=CC=C5C=CC=CC5=C43)C3=C(C=CC=C3)C3=CC=CC1=N32.CC1(C)C2=CC=CC3=N2[Pt]2(C4=CC=CC=C43)N3C(=NC4=C3C=CC=C4)C3=CC=CC1=N32 Chemical compound C1=CC=C(N2C3=CC4=C(C=CC=C4)C4=N3[Pt]3(C5=CC=CC=C54)C4=C(C=CC=C4)C4=C5C=CC=CC5=CC2=N43)C=C1.C1=CC=C(N2C3=N4C(=CC=C3)C3=CC=CC=C3[Pt]43C4=C(C=CC=C4)C4=CC=CC2=N43)C=C1.C1=CC=C2C(=C1)C1=CC=CC3=N1[Pt]14N2C2=C(C=CC=C2)N1C1=C(C=CC=C1)C1=CC=CC3=N14.CC1(C)C2=CC=CC3=C2[Pt]24C5=C(C=CC6=C5C5=C(C=CC7=CC=C3N2=C75)C=C6)C2=CC=CC1=N24.CC1(C)C2=CC=CC3=C2[Pt]24C5=C1C=CC=C5C1=N2C(=CN=C1)C(C)(C)C1=N4C3=CN=C1.CC1(C)C2=CC=CC3=N2[Pt]2(C4=CC=C5C=CC=CC5=C43)C3=C(C=CC=C3)C3=CC=CC1=N32.CC1(C)C2=CC=CC3=N2[Pt]2(C4=CC=CC=C43)N3C(=NC4=C3C=CC=C4)C3=CC=CC1=N32 ATCJKDSZGULBON-UHFFFAOYSA-N 0.000 description 1
- XMWRBQBLMFGWIX-UHFFFAOYSA-N C60 fullerene Chemical class C12=C3C(C4=C56)=C7C8=C5C5=C9C%10=C6C6=C4C1=C1C4=C6C6=C%10C%10=C9C9=C%11C5=C8C5=C8C7=C3C3=C7C2=C1C1=C2C4=C6C4=C%10C6=C9C9=C%11C5=C5C8=C3C3=C7C1=C1C2=C4C6=C2C9=C5C3=C12 XMWRBQBLMFGWIX-UHFFFAOYSA-N 0.000 description 1
- LMNSYBZTYATVDL-UHFFFAOYSA-N CC1(C)C2=CC=CC3=N2[Pt@@]2(C4=CC(C(F)(F)F)=NN4C4=CC=CC1=N42)C1=CC(C(F)(F)F)=NN13 Chemical compound CC1(C)C2=CC=CC3=N2[Pt@@]2(C4=CC(C(F)(F)F)=NN4C4=CC=CC1=N42)C1=CC(C(F)(F)F)=NN13 LMNSYBZTYATVDL-UHFFFAOYSA-N 0.000 description 1
- KZSMGVWCAXVUNW-UHFFFAOYSA-N CC1(C)C2=CC=CC3=N2[Pt]2(C4=CC(F)=C(C5=CC=CC=C5)C(F)=C43)C3=C(C4=CC=CC1=N42)C(F)=C(C1=CCCC=C1)C(F)=C3.CC1(C)C2=CC=CC3=N2[Pt]2(C4=CC=C(C5=CC=CC=C5)C=C43)C3=C(C=C(C4=CC=CC=C4)C=C3)C3=CC=CC1=N32.CC1=CC2=N3C(=C1)C(C)(C)C1=N4C(=CC(C)=C1)C1=C(C=CC(C5=CC=CC=C5)=C1)[Pt]34C1=CC=C(C3=CC=CC=C3)C=C12.[C-]#[N+]C1=C(C(F)(F)F)C=C2C(=C1C(F)(F)F)C1=N3C(=CC=C1)C(C)(C)C1=N4C(=CC=C1)C1=C(C=C(C(F)(F)F)C(C#N)=C1C(F)(F)F)[Pt]234.[C-]#[N+]C1=C2C(=CC(C#N)=C1C(F)(F)F)[Pt]13C4=C(C5=CC=CC(=N51)C(C)(C)C1=CC=CC2=N13)C(C#N)=C(C(F)(F)F)C(C#N)=C4 Chemical compound CC1(C)C2=CC=CC3=N2[Pt]2(C4=CC(F)=C(C5=CC=CC=C5)C(F)=C43)C3=C(C4=CC=CC1=N42)C(F)=C(C1=CCCC=C1)C(F)=C3.CC1(C)C2=CC=CC3=N2[Pt]2(C4=CC=C(C5=CC=CC=C5)C=C43)C3=C(C=C(C4=CC=CC=C4)C=C3)C3=CC=CC1=N32.CC1=CC2=N3C(=C1)C(C)(C)C1=N4C(=CC(C)=C1)C1=C(C=CC(C5=CC=CC=C5)=C1)[Pt]34C1=CC=C(C3=CC=CC=C3)C=C12.[C-]#[N+]C1=C(C(F)(F)F)C=C2C(=C1C(F)(F)F)C1=N3C(=CC=C1)C(C)(C)C1=N4C(=CC=C1)C1=C(C=C(C(F)(F)F)C(C#N)=C1C(F)(F)F)[Pt]234.[C-]#[N+]C1=C2C(=CC(C#N)=C1C(F)(F)F)[Pt]13C4=C(C5=CC=CC(=N51)C(C)(C)C1=CC=CC2=N13)C(C#N)=C(C(F)(F)F)C(C#N)=C4 KZSMGVWCAXVUNW-UHFFFAOYSA-N 0.000 description 1
- XXJXGUPMPLQAFD-UHFFFAOYSA-N CC1(C)C2=CC=CC3=N2[Pt]2(C4=CC=C(C5=CC=CC=C5)C=C43)C3=C(C=C(C4=CC=CC=C4)C=C3)C3=CC=CC1=N32 Chemical compound CC1(C)C2=CC=CC3=N2[Pt]2(C4=CC=C(C5=CC=CC=C5)C=C43)C3=C(C=C(C4=CC=CC=C4)C=C3)C3=CC=CC1=N32 XXJXGUPMPLQAFD-UHFFFAOYSA-N 0.000 description 1
- NZYLGULEFFCNHA-UHFFFAOYSA-N CC1=CC(C)=C(C2=C(F)C=C3C(=C2F)C2=N4C(=CC=C2)C(C)(C)C2=N5C(=CC=C2)C2=C(C=C(F)C(C6=C(C)C=C(C)C=C6C)=C2F)[Pt]345)C(C)=C1.CC1=CC(C)=C(C2=CC=C3C(=C2)C2=N4C(=CC=C2)C(C)(C)C2=N5C(=CC=C2)C2=C(C=CC(C6=C(C)C=C(C)C=C6C)=C2)[Pt]345)C(C)=C1 Chemical compound CC1=CC(C)=C(C2=C(F)C=C3C(=C2F)C2=N4C(=CC=C2)C(C)(C)C2=N5C(=CC=C2)C2=C(C=C(F)C(C6=C(C)C=C(C)C=C6C)=C2F)[Pt]345)C(C)=C1.CC1=CC(C)=C(C2=CC=C3C(=C2)C2=N4C(=CC=C2)C(C)(C)C2=N5C(=CC=C2)C2=C(C=CC(C6=C(C)C=C(C)C=C6C)=C2)[Pt]345)C(C)=C1 NZYLGULEFFCNHA-UHFFFAOYSA-N 0.000 description 1
- PTOAJBINSLHRSJ-UHFFFAOYSA-N CC1=CC2=N3C(=C1)C(C)(C)C1=N4C(=CC(C)=C1)C1=C(C=CC(C5=CC=CC=C5)=C1)[Pt]34C1=CC=C(C3=CC=CC=C3)C=C12 Chemical compound CC1=CC2=N3C(=C1)C(C)(C)C1=N4C(=CC(C)=C1)C1=C(C=CC(C5=CC=CC=C5)=C1)[Pt]34C1=CC=C(C3=CC=CC=C3)C=C12 PTOAJBINSLHRSJ-UHFFFAOYSA-N 0.000 description 1
- UXVMQQNJUSDDNG-UHFFFAOYSA-L Calcium chloride Chemical compound [Cl-].[Cl-].[Ca+2] UXVMQQNJUSDDNG-UHFFFAOYSA-L 0.000 description 1
- UGFAIRIUMAVXCW-UHFFFAOYSA-N Carbon monoxide Chemical compound [O+]#[C-] UGFAIRIUMAVXCW-UHFFFAOYSA-N 0.000 description 1
- 229910052684 Cerium Inorganic materials 0.000 description 1
- DQFBYFPFKXHELB-UHFFFAOYSA-N Chalcone Natural products C=1C=CC=CC=1C(=O)C=CC1=CC=CC=C1 DQFBYFPFKXHELB-UHFFFAOYSA-N 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 description 1
- YZCKVEUIGOORGS-OUBTZVSYSA-N Deuterium Chemical group [2H] YZCKVEUIGOORGS-OUBTZVSYSA-N 0.000 description 1
- 229910052692 Dysprosium Inorganic materials 0.000 description 1
- 229910052691 Erbium Inorganic materials 0.000 description 1
- 229910052689 Holmium Inorganic materials 0.000 description 1
- 229910021578 Iron(III) chloride Inorganic materials 0.000 description 1
- 229910000799 K alloy Inorganic materials 0.000 description 1
- PAYRUJLWNCNPSJ-UHFFFAOYSA-N N-phenyl amine Natural products NC1=CC=CC=C1 PAYRUJLWNCNPSJ-UHFFFAOYSA-N 0.000 description 1
- 229920001774 Perfluoroether Polymers 0.000 description 1
- 239000004695 Polyether sulfone Substances 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- 239000004743 Polypropylene Substances 0.000 description 1
- 239000004793 Polystyrene Substances 0.000 description 1
- 229920002396 Polyurea Polymers 0.000 description 1
- 229910052777 Praseodymium Inorganic materials 0.000 description 1
- WTKZEGDFNFYCGP-UHFFFAOYSA-N Pyrazole Chemical compound C=1C=NNC=1 WTKZEGDFNFYCGP-UHFFFAOYSA-N 0.000 description 1
- KJTLSVCANCCWHF-UHFFFAOYSA-N Ruthenium Chemical compound [Ru] KJTLSVCANCCWHF-UHFFFAOYSA-N 0.000 description 1
- 229910052772 Samarium Inorganic materials 0.000 description 1
- 229910004205 SiNX Inorganic materials 0.000 description 1
- 229910004286 SiNxOy Inorganic materials 0.000 description 1
- BLRPTPMANUNPDV-UHFFFAOYSA-N Silane Chemical compound [SiH4] BLRPTPMANUNPDV-UHFFFAOYSA-N 0.000 description 1
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 1
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 1
- PJANXHGTPQOBST-VAWYXSNFSA-N Stilbene Natural products C=1C=CC=CC=1/C=C/C1=CC=CC=C1 PJANXHGTPQOBST-VAWYXSNFSA-N 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical group [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- 229910052775 Thulium Inorganic materials 0.000 description 1
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 1
- 229910021536 Zeolite Inorganic materials 0.000 description 1
- DGEZNRSVGBDHLK-UHFFFAOYSA-N [1,10]phenanthroline Chemical compound C1=CN=C2C3=NC=CC=C3C=CC2=C1 DGEZNRSVGBDHLK-UHFFFAOYSA-N 0.000 description 1
- HHAWXHPCNOWKBX-UHFFFAOYSA-N [C-]#[N+]C1=C(C#N)C=C2C(=C1[N+]#[C-])C1=N3C(=CC=C1)C(C)(C)C1=N4C(=CC=C1)C1=C(C=C(C#N)C(C#N)=C1C#N)[Pt]234.[C-]#[N+]C1=C(F)C=C2C(=C1F)C1=N3C(=CC=C1)C(C)(C)C1=N4C(=CC=C1)C1=C(C=C(F)C(C#N)=C1F)[Pt]234.[C-]#[N+]C1=C(F)C=C2C(=C1F)C1=N3C(=CC=C1)C(C)(C)C1=N4C(=CC=C1)C1=C(C=CC=C1)[Pt]234.[C-]#[N+]C1=C2C(=CC(C#N)=C1[N+](=O)[O-])[Pt]13C4=C(C5=CC=CC(=N51)C(C)(C)C1=CC=CC2=N13)C(C#N)=C([N+](=O)[O-])C(C#N)=C4.[C-]#[N+]C1=CC=C2C(=C1)C1=N3C(=CC=C1)C(C)(C)C1=N4C(=CC=C1)C1=C(C=CC(C#N)=C1)[Pt]234 Chemical compound [C-]#[N+]C1=C(C#N)C=C2C(=C1[N+]#[C-])C1=N3C(=CC=C1)C(C)(C)C1=N4C(=CC=C1)C1=C(C=C(C#N)C(C#N)=C1C#N)[Pt]234.[C-]#[N+]C1=C(F)C=C2C(=C1F)C1=N3C(=CC=C1)C(C)(C)C1=N4C(=CC=C1)C1=C(C=C(F)C(C#N)=C1F)[Pt]234.[C-]#[N+]C1=C(F)C=C2C(=C1F)C1=N3C(=CC=C1)C(C)(C)C1=N4C(=CC=C1)C1=C(C=CC=C1)[Pt]234.[C-]#[N+]C1=C2C(=CC(C#N)=C1[N+](=O)[O-])[Pt]13C4=C(C5=CC=CC(=N51)C(C)(C)C1=CC=CC2=N13)C(C#N)=C([N+](=O)[O-])C(C#N)=C4.[C-]#[N+]C1=CC=C2C(=C1)C1=N3C(=CC=C1)C(C)(C)C1=N4C(=CC=C1)C1=C(C=CC(C#N)=C1)[Pt]234 HHAWXHPCNOWKBX-UHFFFAOYSA-N 0.000 description 1
- GPIKSKRAQGFEHR-UHFFFAOYSA-N [C-]#[N+]C1=C(F)C=C2C(=C1F)C1=N3C(=CC=C1)C(C)(C)C1=N4C(=CC=C1)C1=C(C=C(F)C(C#N)=C1F)[Pt]234 Chemical compound [C-]#[N+]C1=C(F)C=C2C(=C1F)C1=N3C(=CC=C1)C(C)(C)C1=N4C(=CC=C1)C1=C(C=C(F)C(C#N)=C1F)[Pt]234 GPIKSKRAQGFEHR-UHFFFAOYSA-N 0.000 description 1
- UVMGNYVBKJYFOM-UHFFFAOYSA-N [C-]#[N+]C1=C(F)C=C2C(=C1F)C1=N3C(=CC=C1)C1(C4=CC=CC5=N4[Pt]4(C6=CC(F)=C(C#N)C(F)=C65)C5=C(C6=CC=CC1=N64)C(F)=C(C#N)C(F)=C5)C1=N4C(=CC=C1)C1=C(C=C(F)C([N+]#[C-])=C1F)[Pt]234 Chemical compound [C-]#[N+]C1=C(F)C=C2C(=C1F)C1=N3C(=CC=C1)C1(C4=CC=CC5=N4[Pt]4(C6=CC(F)=C(C#N)C(F)=C65)C5=C(C6=CC=CC1=N64)C(F)=C(C#N)C(F)=C5)C1=N4C(=CC=C1)C1=C(C=C(F)C([N+]#[C-])=C1F)[Pt]234 UVMGNYVBKJYFOM-UHFFFAOYSA-N 0.000 description 1
- AOIIREDZWFTKQH-UHFFFAOYSA-N [C-]#[N+]C1=CC=C2C(=C1)C1=N3C(=CC=C1)C(C)(C)C1=N4C(=CC=C1)C1=C(C=CC(C#N)=C1)[Pt]234 Chemical compound [C-]#[N+]C1=CC=C2C(=C1)C1=N3C(=CC=C1)C(C)(C)C1=N4C(=CC=C1)C1=C(C=CC(C#N)=C1)[Pt]234 AOIIREDZWFTKQH-UHFFFAOYSA-N 0.000 description 1
- XHCLAFWTIXFWPH-UHFFFAOYSA-N [O-2].[O-2].[O-2].[O-2].[O-2].[V+5].[V+5] Chemical compound [O-2].[O-2].[O-2].[O-2].[O-2].[V+5].[V+5] XHCLAFWTIXFWPH-UHFFFAOYSA-N 0.000 description 1
- 239000011358 absorbing material Substances 0.000 description 1
- 125000000738 acetamido group Chemical group [H]C([H])([H])C(=O)N([H])[*] 0.000 description 1
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 1
- CUJRVFIICFDLGR-UHFFFAOYSA-N acetylacetonate Chemical compound CC(=O)[CH-]C(C)=O CUJRVFIICFDLGR-UHFFFAOYSA-N 0.000 description 1
- 125000003668 acetyloxy group Chemical group [H]C([H])([H])C(=O)O[*] 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 125000002252 acyl group Chemical group 0.000 description 1
- 125000004442 acylamino group Chemical group 0.000 description 1
- 125000004423 acyloxy group Chemical group 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 238000004220 aggregation Methods 0.000 description 1
- 150000001335 aliphatic alkanes Chemical class 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 229910000287 alkaline earth metal oxide Inorganic materials 0.000 description 1
- 125000003342 alkenyl group Chemical group 0.000 description 1
- 125000004450 alkenylene group Chemical group 0.000 description 1
- 125000004453 alkoxycarbonyl group Chemical group 0.000 description 1
- 125000004466 alkoxycarbonylamino group Chemical group 0.000 description 1
- 125000004414 alkyl thio group Chemical group 0.000 description 1
- 125000002947 alkylene group Chemical group 0.000 description 1
- 125000000304 alkynyl group Chemical group 0.000 description 1
- 125000004419 alkynylene group Chemical group 0.000 description 1
- AZDRQVAHHNSJOQ-UHFFFAOYSA-N alumane Chemical class [AlH3] AZDRQVAHHNSJOQ-UHFFFAOYSA-N 0.000 description 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- SNAAJJQQZSMGQD-UHFFFAOYSA-N aluminum magnesium Chemical compound [Mg].[Al] SNAAJJQQZSMGQD-UHFFFAOYSA-N 0.000 description 1
- 125000002490 anilino group Chemical group [H]N(*)C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- MHDLAWFYLQAULB-UHFFFAOYSA-N anilinophosphonic acid Chemical group OP(O)(=O)NC1=CC=CC=C1 MHDLAWFYLQAULB-UHFFFAOYSA-N 0.000 description 1
- 150000008425 anthrones Chemical class 0.000 description 1
- 229910052787 antimony Inorganic materials 0.000 description 1
- WATWJIUSRGPENY-UHFFFAOYSA-N antimony atom Chemical compound [Sb] WATWJIUSRGPENY-UHFFFAOYSA-N 0.000 description 1
- VMPVEPPRYRXYNP-UHFFFAOYSA-I antimony(5+);pentachloride Chemical compound Cl[Sb](Cl)(Cl)(Cl)Cl VMPVEPPRYRXYNP-UHFFFAOYSA-I 0.000 description 1
- 229940054051 antipsychotic indole derivative Drugs 0.000 description 1
- 229910052786 argon Inorganic materials 0.000 description 1
- 125000005162 aryl oxy carbonyl amino group Chemical group 0.000 description 1
- 125000005161 aryl oxy carbonyl group Chemical group 0.000 description 1
- 125000005110 aryl thio group Chemical group 0.000 description 1
- 125000000732 arylene group Chemical group 0.000 description 1
- 125000004429 atom Chemical group 0.000 description 1
- 125000000043 benzamido group Chemical group [H]N([*])C(=O)C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- HFACYLZERDEVSX-UHFFFAOYSA-N benzidine Chemical compound C1=CC(N)=CC=C1C1=CC=C(N)C=C1 HFACYLZERDEVSX-UHFFFAOYSA-N 0.000 description 1
- 125000003785 benzimidazolyl group Chemical group N1=C(NC2=C1C=CC=C2)* 0.000 description 1
- 125000001164 benzothiazolyl group Chemical group S1C(=NC2=C1C=CC=C2)* 0.000 description 1
- 125000004541 benzoxazolyl group Chemical group O1C(=NC2=C1C=CC=C2)* 0.000 description 1
- 125000003236 benzoyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C(*)=O 0.000 description 1
- 125000001231 benzoyloxy group Chemical group C(C1=CC=CC=C1)(=O)O* 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 125000004106 butoxy group Chemical group [*]OC([H])([H])C([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 1
- 229910052792 caesium Inorganic materials 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910001622 calcium bromide Inorganic materials 0.000 description 1
- 239000001110 calcium chloride Substances 0.000 description 1
- 229910001628 calcium chloride Inorganic materials 0.000 description 1
- WGEFECGEFUFIQW-UHFFFAOYSA-L calcium dibromide Chemical compound [Ca+2].[Br-].[Br-] WGEFECGEFUFIQW-UHFFFAOYSA-L 0.000 description 1
- WUKWITHWXAAZEY-UHFFFAOYSA-L calcium difluoride Chemical compound [F-].[F-].[Ca+2] WUKWITHWXAAZEY-UHFFFAOYSA-L 0.000 description 1
- 229910001634 calcium fluoride Inorganic materials 0.000 description 1
- 235000011132 calcium sulphate Nutrition 0.000 description 1
- 150000001718 carbodiimides Chemical class 0.000 description 1
- 229910002091 carbon monoxide Inorganic materials 0.000 description 1
- 239000002041 carbon nanotube Substances 0.000 description 1
- 229910021393 carbon nanotube Inorganic materials 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 150000001735 carboxylic acids Chemical class 0.000 description 1
- ZMIGMASIKSOYAM-UHFFFAOYSA-N cerium Chemical compound [Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce] ZMIGMASIKSOYAM-UHFFFAOYSA-N 0.000 description 1
- 235000005513 chalcones Nutrition 0.000 description 1
- 238000003486 chemical etching Methods 0.000 description 1
- 239000000460 chlorine Substances 0.000 description 1
- 125000001309 chloro group Chemical group Cl* 0.000 description 1
- 229910052804 chromium Inorganic materials 0.000 description 1
- 239000011651 chromium Substances 0.000 description 1
- 238000007334 copolymerization reaction Methods 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- ORTQZVOHEJQUHG-UHFFFAOYSA-L copper(II) chloride Chemical compound Cl[Cu]Cl ORTQZVOHEJQUHG-UHFFFAOYSA-L 0.000 description 1
- OMZSGWSJDCOLKM-UHFFFAOYSA-N copper(II) sulfide Chemical compound [S-2].[Cu+2] OMZSGWSJDCOLKM-UHFFFAOYSA-N 0.000 description 1
- GBRBMTNGQBKBQE-UHFFFAOYSA-L copper;diiodide Chemical compound I[Cu]I GBRBMTNGQBKBQE-UHFFFAOYSA-L 0.000 description 1
- 229910052593 corundum Inorganic materials 0.000 description 1
- 125000000753 cycloalkyl group Chemical group 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- 230000007423 decrease Effects 0.000 description 1
- 229910052805 deuterium Inorganic materials 0.000 description 1
- 125000001664 diethylamino group Chemical group [H]C([H])([H])C([H])([H])N(*)C([H])([H])C([H])([H])[H] 0.000 description 1
- NBAUUSKPFGFBQZ-UHFFFAOYSA-N diethylaminophosphonic acid Chemical group CCN(CC)P(O)(O)=O NBAUUSKPFGFBQZ-UHFFFAOYSA-N 0.000 description 1
- 125000005594 diketone group Chemical group 0.000 description 1
- 125000002147 dimethylamino group Chemical group [H]C([H])([H])N(*)C([H])([H])[H] 0.000 description 1
- HNPSIPDUKPIQMN-UHFFFAOYSA-N dioxosilane;oxo(oxoalumanyloxy)alumane Chemical compound O=[Si]=O.O=[Al]O[Al]=O HNPSIPDUKPIQMN-UHFFFAOYSA-N 0.000 description 1
- KBQHZAAAGSGFKK-UHFFFAOYSA-N dysprosium atom Chemical compound [Dy] KBQHZAAAGSGFKK-UHFFFAOYSA-N 0.000 description 1
- 229920001971 elastomer Polymers 0.000 description 1
- 230000005684 electric field Effects 0.000 description 1
- 239000007772 electrode material Substances 0.000 description 1
- 238000000295 emission spectrum Methods 0.000 description 1
- ZSWFCLXCOIISFI-UHFFFAOYSA-N endo-cyclopentadiene Natural products C1C=CC=C1 ZSWFCLXCOIISFI-UHFFFAOYSA-N 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- UYAHIZSMUZPPFV-UHFFFAOYSA-N erbium Chemical compound [Er] UYAHIZSMUZPPFV-UHFFFAOYSA-N 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 125000005678 ethenylene group Chemical group [H]C([*:1])=C([H])[*:2] 0.000 description 1
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 description 1
- 125000003754 ethoxycarbonyl group Chemical group C(=O)(OCC)* 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 125000000816 ethylene group Chemical group [H]C([H])([*:1])C([H])([H])[*:2] 0.000 description 1
- 125000004705 ethylthio group Chemical group C(C)S* 0.000 description 1
- 230000005284 excitation Effects 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- YLQWCDOCJODRMT-UHFFFAOYSA-N fluoren-9-one Chemical compound C1=CC=C2C(=O)C3=CC=CC=C3C2=C1 YLQWCDOCJODRMT-UHFFFAOYSA-N 0.000 description 1
- 239000007850 fluorescent dye Substances 0.000 description 1
- 125000001153 fluoro group Chemical group F* 0.000 description 1
- 125000002485 formyl group Chemical group [H]C(*)=O 0.000 description 1
- 229910003472 fullerene Inorganic materials 0.000 description 1
- 125000002541 furyl group Chemical group 0.000 description 1
- UPWPDUACHOATKO-UHFFFAOYSA-K gallium trichloride Chemical compound Cl[Ga](Cl)Cl UPWPDUACHOATKO-UHFFFAOYSA-K 0.000 description 1
- JVZRCNQLWOELDU-UHFFFAOYSA-N gamma-Phenylpyridine Natural products C1=CC=CC=C1C1=CC=NC=C1 JVZRCNQLWOELDU-UHFFFAOYSA-N 0.000 description 1
- PVADDRMAFCOOPC-UHFFFAOYSA-N germanium monoxide Inorganic materials [Ge]=O PVADDRMAFCOOPC-UHFFFAOYSA-N 0.000 description 1
- 229910052736 halogen Inorganic materials 0.000 description 1
- 150000002367 halogens Chemical class 0.000 description 1
- 125000005549 heteroarylene group Chemical group 0.000 description 1
- 125000005842 heteroatom Chemical group 0.000 description 1
- 229940083761 high-ceiling diuretics pyrazolone derivative Drugs 0.000 description 1
- KJZYNXUDTRRSPN-UHFFFAOYSA-N holmium atom Chemical compound [Ho] KJZYNXUDTRRSPN-UHFFFAOYSA-N 0.000 description 1
- 125000000717 hydrazino group Chemical group [H]N([*])N([H])[H] 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 150000002460 imidazoles Chemical class 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- 125000001841 imino group Chemical group [H]N=* 0.000 description 1
- APFVFJFRJDLVQX-UHFFFAOYSA-N indium atom Chemical compound [In] APFVFJFRJDLVQX-UHFFFAOYSA-N 0.000 description 1
- 229910003437 indium oxide Inorganic materials 0.000 description 1
- PSCMQHVBLHHWTO-UHFFFAOYSA-K indium(iii) chloride Chemical compound Cl[In](Cl)Cl PSCMQHVBLHHWTO-UHFFFAOYSA-K 0.000 description 1
- PJXISJQVUVHSOJ-UHFFFAOYSA-N indium(iii) oxide Chemical compound [O-2].[O-2].[O-2].[In+3].[In+3] PJXISJQVUVHSOJ-UHFFFAOYSA-N 0.000 description 1
- PZOUSPYUWWUPPK-UHFFFAOYSA-N indole Natural products CC1=CC=CC2=C1C=CN2 PZOUSPYUWWUPPK-UHFFFAOYSA-N 0.000 description 1
- RKJUIXBNRJVNHR-UHFFFAOYSA-N indolenine Natural products C1=CC=C2CC=NC2=C1 RKJUIXBNRJVNHR-UHFFFAOYSA-N 0.000 description 1
- 150000002475 indoles Chemical class 0.000 description 1
- 229940079865 intestinal antiinfectives imidazole derivative Drugs 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 238000010884 ion-beam technique Methods 0.000 description 1
- RBTARNINKXHZNM-UHFFFAOYSA-K iron trichloride Chemical compound Cl[Fe](Cl)Cl RBTARNINKXHZNM-UHFFFAOYSA-K 0.000 description 1
- JEIPFZHSYJVQDO-UHFFFAOYSA-N iron(III) oxide Inorganic materials O=[Fe]O[Fe]=O JEIPFZHSYJVQDO-UHFFFAOYSA-N 0.000 description 1
- 150000002527 isonitriles Chemical class 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 229910052746 lanthanum Inorganic materials 0.000 description 1
- FZLIPJUXYLNCLC-UHFFFAOYSA-N lanthanum atom Chemical compound [La] FZLIPJUXYLNCLC-UHFFFAOYSA-N 0.000 description 1
- 150000002605 large molecules Chemical class 0.000 description 1
- 238000001182 laser chemical vapour deposition Methods 0.000 description 1
- 229910052745 lead Inorganic materials 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910001629 magnesium chloride Inorganic materials 0.000 description 1
- 229910001635 magnesium fluoride Inorganic materials 0.000 description 1
- SJCKRGFTWFGHGZ-UHFFFAOYSA-N magnesium silver Chemical compound [Mg].[Ag] SJCKRGFTWFGHGZ-UHFFFAOYSA-N 0.000 description 1
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 1
- 235000019341 magnesium sulphate Nutrition 0.000 description 1
- AXZKOIWUVFPNLO-UHFFFAOYSA-N magnesium;oxygen(2-) Chemical compound [O-2].[Mg+2] AXZKOIWUVFPNLO-UHFFFAOYSA-N 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 229910001512 metal fluoride Inorganic materials 0.000 description 1
- 229910001507 metal halide Inorganic materials 0.000 description 1
- 150000005309 metal halides Chemical class 0.000 description 1
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Natural products C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 1
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 1
- 125000001160 methoxycarbonyl group Chemical group [H]C([H])([H])OC(*)=O 0.000 description 1
- 125000006626 methoxycarbonylamino group Chemical group 0.000 description 1
- 125000001570 methylene group Chemical group [H]C([H])([*:1])[*:2] 0.000 description 1
- 125000002816 methylsulfanyl group Chemical group [H]C([H])([H])S[*] 0.000 description 1
- 239000002808 molecular sieve Substances 0.000 description 1
- 239000000178 monomer Substances 0.000 description 1
- 125000004573 morpholin-4-yl group Chemical group N1(CCOCC1)* 0.000 description 1
- IBHBKWKFFTZAHE-UHFFFAOYSA-N n-[4-[4-(n-naphthalen-1-ylanilino)phenyl]phenyl]-n-phenylnaphthalen-1-amine Chemical compound C1=CC=CC=C1N(C=1C2=CC=CC=C2C=CC=1)C1=CC=C(C=2C=CC(=CC=2)N(C=2C=CC=CC=2)C=2C3=CC=CC=C3C=CC=2)C=C1 IBHBKWKFFTZAHE-UHFFFAOYSA-N 0.000 description 1
- 125000001624 naphthyl group Chemical group 0.000 description 1
- 125000004957 naphthylene group Chemical group 0.000 description 1
- GNRSAWUEBMWBQH-UHFFFAOYSA-N nickel(II) oxide Inorganic materials [Ni]=O GNRSAWUEBMWBQH-UHFFFAOYSA-N 0.000 description 1
- 150000004767 nitrides Chemical class 0.000 description 1
- IJGRMHOSHXDMSA-UHFFFAOYSA-N nitrogen Substances N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 1
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 1
- GVGCUCJTUSOZKP-UHFFFAOYSA-N nitrogen trifluoride Chemical class FN(F)F GVGCUCJTUSOZKP-UHFFFAOYSA-N 0.000 description 1
- JFNLZVQOOSMTJK-KNVOCYPGSA-N norbornene Chemical compound C1[C@@H]2CC[C@H]1C=C2 JFNLZVQOOSMTJK-KNVOCYPGSA-N 0.000 description 1
- 239000011368 organic material Substances 0.000 description 1
- 150000001282 organosilanes Chemical class 0.000 description 1
- 229910052762 osmium Inorganic materials 0.000 description 1
- SYQBFIAQOQZEGI-UHFFFAOYSA-N osmium atom Chemical compound [Os] SYQBFIAQOQZEGI-UHFFFAOYSA-N 0.000 description 1
- 230000001590 oxidative effect Effects 0.000 description 1
- 125000001820 oxy group Chemical group [*:1]O[*:2] 0.000 description 1
- UJMWVICAENGCRF-UHFFFAOYSA-N oxygen difluoride Chemical class FOF UJMWVICAENGCRF-UHFFFAOYSA-N 0.000 description 1
- 229910052763 palladium Inorganic materials 0.000 description 1
- 238000000059 patterning Methods 0.000 description 1
- AOLPZAHRYHXPLR-UHFFFAOYSA-I pentafluoroniobium Chemical compound F[Nb](F)(F)(F)F AOLPZAHRYHXPLR-UHFFFAOYSA-I 0.000 description 1
- 125000002080 perylenyl group Chemical group C1(=CC=C2C=CC=C3C4=CC=CC5=CC=CC(C1=C23)=C45)* 0.000 description 1
- CSHWQDPOILHKBI-UHFFFAOYSA-N peryrene Natural products C1=CC(C2=CC=CC=3C2=C2C=CC=3)=C3C2=CC=CC3=C1 CSHWQDPOILHKBI-UHFFFAOYSA-N 0.000 description 1
- 150000005041 phenanthrolines Chemical class 0.000 description 1
- ISWSIDIOOBJBQZ-UHFFFAOYSA-M phenolate Chemical compound [O-]C1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-M 0.000 description 1
- 229940031826 phenolate Drugs 0.000 description 1
- 125000000843 phenylene group Chemical group C1(=C(C=CC=C1)*)* 0.000 description 1
- 150000004986 phenylenediamines Chemical class 0.000 description 1
- 125000003356 phenylsulfanyl group Chemical group [*]SC1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- 125000003170 phenylsulfonyl group Chemical group C1(=CC=CC=C1)S(=O)(=O)* 0.000 description 1
- PTMHPRAIXMAOOB-UHFFFAOYSA-N phosphoramidic acid Chemical group NP(O)(O)=O PTMHPRAIXMAOOB-UHFFFAOYSA-N 0.000 description 1
- 238000000206 photolithography Methods 0.000 description 1
- 238000000053 physical method Methods 0.000 description 1
- SIOXPEMLGUPBBT-UHFFFAOYSA-M picolinate Chemical compound [O-]C(=O)C1=CC=CC=N1 SIOXPEMLGUPBBT-UHFFFAOYSA-M 0.000 description 1
- 125000005936 piperidyl group Chemical group 0.000 description 1
- 238000005268 plasma chemical vapour deposition Methods 0.000 description 1
- 229920003227 poly(N-vinyl carbazole) Polymers 0.000 description 1
- 229920002493 poly(chlorotrifluoroethylene) Polymers 0.000 description 1
- 229920003207 poly(ethylene-2,6-naphthalate) Polymers 0.000 description 1
- 229920003229 poly(methyl methacrylate) Polymers 0.000 description 1
- 229920000548 poly(silane) polymer Polymers 0.000 description 1
- 229920003050 poly-cycloolefin Polymers 0.000 description 1
- 229920000767 polyaniline Polymers 0.000 description 1
- 229920001230 polyarylate Polymers 0.000 description 1
- 239000004417 polycarbonate Substances 0.000 description 1
- 229920000515 polycarbonate Polymers 0.000 description 1
- 229920000728 polyester Polymers 0.000 description 1
- 229920006393 polyether sulfone Polymers 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 239000011112 polyethylene naphthalate Substances 0.000 description 1
- 229920000139 polyethylene terephthalate Polymers 0.000 description 1
- 239000005020 polyethylene terephthalate Substances 0.000 description 1
- 238000006116 polymerization reaction Methods 0.000 description 1
- 230000000379 polymerizing effect Effects 0.000 description 1
- 229920001155 polypropylene Polymers 0.000 description 1
- 229920000128 polypyrrole Polymers 0.000 description 1
- 229920002223 polystyrene Polymers 0.000 description 1
- 229920001343 polytetrafluoroethylene Polymers 0.000 description 1
- BITYAPCSNKJESK-UHFFFAOYSA-N potassiosodium Chemical compound [Na].[K] BITYAPCSNKJESK-UHFFFAOYSA-N 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- CHWRSCGUEQEHOH-UHFFFAOYSA-N potassium oxide Chemical compound [O-2].[K+].[K+] CHWRSCGUEQEHOH-UHFFFAOYSA-N 0.000 description 1
- 229910001950 potassium oxide Inorganic materials 0.000 description 1
- PUDIUYLPXJFUGB-UHFFFAOYSA-N praseodymium atom Chemical compound [Pr] PUDIUYLPXJFUGB-UHFFFAOYSA-N 0.000 description 1
- 230000003449 preventive effect Effects 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- DNXIASIHZYFFRO-UHFFFAOYSA-N pyrazoline Chemical compound C1CN=NC1 DNXIASIHZYFFRO-UHFFFAOYSA-N 0.000 description 1
- 150000003219 pyrazolines Chemical class 0.000 description 1
- 125000004076 pyridyl group Chemical group 0.000 description 1
- 125000005554 pyridyloxy group Chemical group 0.000 description 1
- 125000005030 pyridylthio group Chemical group N1=C(C=CC=C1)S* 0.000 description 1
- 150000003233 pyrroles Chemical class 0.000 description 1
- LISFMEBWQUVKPJ-UHFFFAOYSA-N quinolin-2-ol Chemical compound C1=CC=C2NC(=O)C=CC2=C1 LISFMEBWQUVKPJ-UHFFFAOYSA-N 0.000 description 1
- MCJGNVYPOGVAJF-UHFFFAOYSA-N quinolin-8-ol Chemical class C1=CN=C2C(O)=CC=CC2=C1 MCJGNVYPOGVAJF-UHFFFAOYSA-N 0.000 description 1
- 125000005493 quinolyl group Chemical group 0.000 description 1
- 150000004053 quinones Chemical class 0.000 description 1
- 229910052761 rare earth metal Inorganic materials 0.000 description 1
- 150000002910 rare earth metals Chemical class 0.000 description 1
- 238000005546 reactive sputtering Methods 0.000 description 1
- 238000005215 recombination Methods 0.000 description 1
- 230000006798 recombination Effects 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 229910052703 rhodium Inorganic materials 0.000 description 1
- 239000010948 rhodium Substances 0.000 description 1
- MHOVAHRLVXNVSD-UHFFFAOYSA-N rhodium atom Chemical compound [Rh] MHOVAHRLVXNVSD-UHFFFAOYSA-N 0.000 description 1
- 229910052707 ruthenium Inorganic materials 0.000 description 1
- KZUNJOHGWZRPMI-UHFFFAOYSA-N samarium atom Chemical compound [Sm] KZUNJOHGWZRPMI-UHFFFAOYSA-N 0.000 description 1
- 229910000077 silane Inorganic materials 0.000 description 1
- 229920002545 silicone oil Polymers 0.000 description 1
- 229920002050 silicone resin Polymers 0.000 description 1
- 125000004469 siloxy group Chemical group [SiH3]O* 0.000 description 1
- 239000010944 silver (metal) Substances 0.000 description 1
- 125000003808 silyl group Chemical group [H][Si]([H])([H])[*] 0.000 description 1
- 150000003384 small molecules Chemical class 0.000 description 1
- 239000005361 soda-lime glass Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- URGAHOPLAPQHLN-UHFFFAOYSA-N sodium aluminosilicate Chemical compound [Na+].[Al+3].[O-][Si]([O-])=O.[O-][Si]([O-])=O URGAHOPLAPQHLN-UHFFFAOYSA-N 0.000 description 1
- KKCBUQHMOMHUOY-UHFFFAOYSA-N sodium oxide Chemical compound [O-2].[Na+].[Na+] KKCBUQHMOMHUOY-UHFFFAOYSA-N 0.000 description 1
- 229910001948 sodium oxide Inorganic materials 0.000 description 1
- 229910052938 sodium sulfate Inorganic materials 0.000 description 1
- 235000011152 sodium sulphate Nutrition 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 239000010935 stainless steel Substances 0.000 description 1
- 229910001220 stainless steel Inorganic materials 0.000 description 1
- 235000021286 stilbenes Nutrition 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 125000000213 sulfino group Chemical group [H]OS(*)=O 0.000 description 1
- 125000000475 sulfinyl group Chemical group [*:2]S([*:1])=O 0.000 description 1
- 125000000020 sulfo group Chemical group O=S(=O)([*])O[H] 0.000 description 1
- 125000006296 sulfonyl amino group Chemical group [H]N(*)S(*)(=O)=O 0.000 description 1
- 125000000472 sulfonyl group Chemical group *S(*)(=O)=O 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 150000003512 tertiary amines Chemical group 0.000 description 1
- BFKJFAAPBSQJPD-UHFFFAOYSA-N tetrafluoroethene Chemical group FC(F)=C(F)F BFKJFAAPBSQJPD-UHFFFAOYSA-N 0.000 description 1
- DLYUQMMRRRQYAE-UHFFFAOYSA-N tetraphosphorus decaoxide Chemical compound O1P(O2)(=O)OP3(=O)OP1(=O)OP2(=O)O3 DLYUQMMRRRQYAE-UHFFFAOYSA-N 0.000 description 1
- 238000002230 thermal chemical vapour deposition Methods 0.000 description 1
- 125000001544 thienyl group Chemical group 0.000 description 1
- 125000003396 thiol group Chemical group [H]S* 0.000 description 1
- 229930192474 thiophene Natural products 0.000 description 1
- XOLBLPGZBRYERU-UHFFFAOYSA-N tin dioxide Chemical compound O=[Sn]=O XOLBLPGZBRYERU-UHFFFAOYSA-N 0.000 description 1
- QHGNHLZPVBIIPX-UHFFFAOYSA-N tin(ii) oxide Chemical class [Sn]=O QHGNHLZPVBIIPX-UHFFFAOYSA-N 0.000 description 1
- 229910052719 titanium Inorganic materials 0.000 description 1
- 125000002088 tosyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1C([H])([H])[H])S(*)(=O)=O 0.000 description 1
- 150000003852 triazoles Chemical class 0.000 description 1
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 description 1
- 125000000026 trimethylsilyl group Chemical group [H]C([H])([H])[Si]([*])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- WFKWXMTUELFFGS-UHFFFAOYSA-N tungsten Chemical compound [W] WFKWXMTUELFFGS-UHFFFAOYSA-N 0.000 description 1
- 229910052721 tungsten Inorganic materials 0.000 description 1
- 239000010937 tungsten Substances 0.000 description 1
- ZOYIPGHJSALYPY-UHFFFAOYSA-K vanadium(iii) bromide Chemical compound [V+3].[Br-].[Br-].[Br-] ZOYIPGHJSALYPY-UHFFFAOYSA-K 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 229910001845 yogo sapphire Inorganic materials 0.000 description 1
- RUDFQVOCFDJEEF-UHFFFAOYSA-N yttrium(III) oxide Inorganic materials [O-2].[O-2].[O-2].[Y+3].[Y+3] RUDFQVOCFDJEEF-UHFFFAOYSA-N 0.000 description 1
- 239000010457 zeolite Substances 0.000 description 1
- YVTHLONGBIQYBO-UHFFFAOYSA-N zinc indium(3+) oxygen(2-) Chemical compound [O--].[Zn++].[In+3] YVTHLONGBIQYBO-UHFFFAOYSA-N 0.000 description 1
- 239000011787 zinc oxide Substances 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05B—ELECTRIC HEATING; ELECTRIC LIGHT SOURCES NOT OTHERWISE PROVIDED FOR; CIRCUIT ARRANGEMENTS FOR ELECTRIC LIGHT SOURCES, IN GENERAL
- H05B33/00—Electroluminescent light sources
- H05B33/12—Light sources with substantially two-dimensional radiating surfaces
- H05B33/14—Light sources with substantially two-dimensional radiating surfaces characterised by the chemical or physical composition or the arrangement of the electroluminescent material, or by the simultaneous addition of the electroluminescent material in or onto the light source
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/30—Coordination compounds
- H10K85/341—Transition metal complexes, e.g. Ru(II)polypyridine complexes
- H10K85/346—Transition metal complexes, e.g. Ru(II)polypyridine complexes comprising platinum
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1029—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/18—Metal complexes
- C09K2211/185—Metal complexes of the platinum group, i.e. Os, Ir, Pt, Ru, Rh or Pd
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K2101/00—Properties of the organic materials covered by group H10K85/00
- H10K2101/10—Triplet emission
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6572—Polycyclic condensed heteroaromatic hydrocarbons comprising only nitrogen in the heteroaromatic polycondensed ring system, e.g. phenanthroline or carbazole
Definitions
- the present invention relates to an organic electroluminescence element (may also referred to as an organic EL element, hereinafter).
- Organic electroluminescence elements have characteristics such as self-luminescence, high-speed response, and the like, and thus application thereof in flat panel displays has been expected. Especially after information had been made public regarding 2-layer (laminate) elements in which a hole-transport organic thin film (hole-transport layer) and an electron-transport organic thin film (electron-transport layer) are laminated, organic electroluminescence elements have attracted attention as large-scale light-emitting elements capable of emitting at low voltages of 10 V or less.
- the laminate organic EL element has a basic structure as follows: an anode, a hole-transport layer, a light-emitting layer, an electron-transport layer, and a cathode, in this order.
- the organic EL element has realized energy efficiency (i.e. by using lower voltages for emitting light) and high emission efficiency due to the aforementioned structure.
- JP-A No. 2006-332622 it is possible to improve the energy saving and emission efficiency to a certain degree.
- the current situation is that further improvements in energy efficiency and emission efficiency are desired.
- the present invention aims at providing an organic electroluminescence element that can maintain high emission efficiency while lowering a driving voltage thereof.
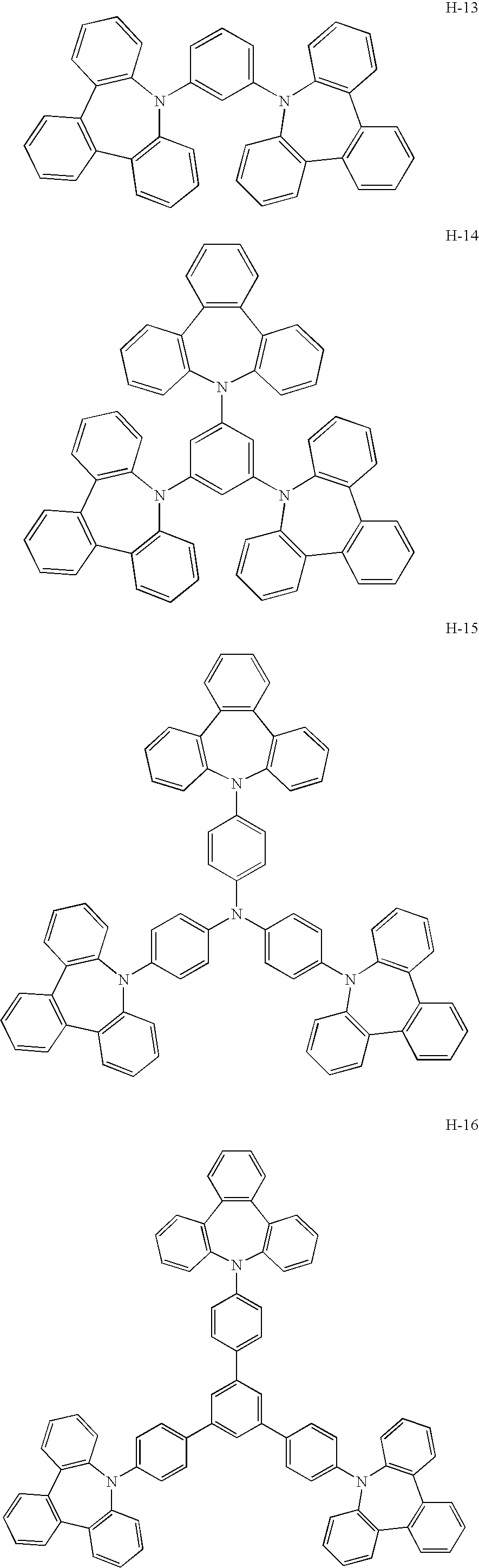
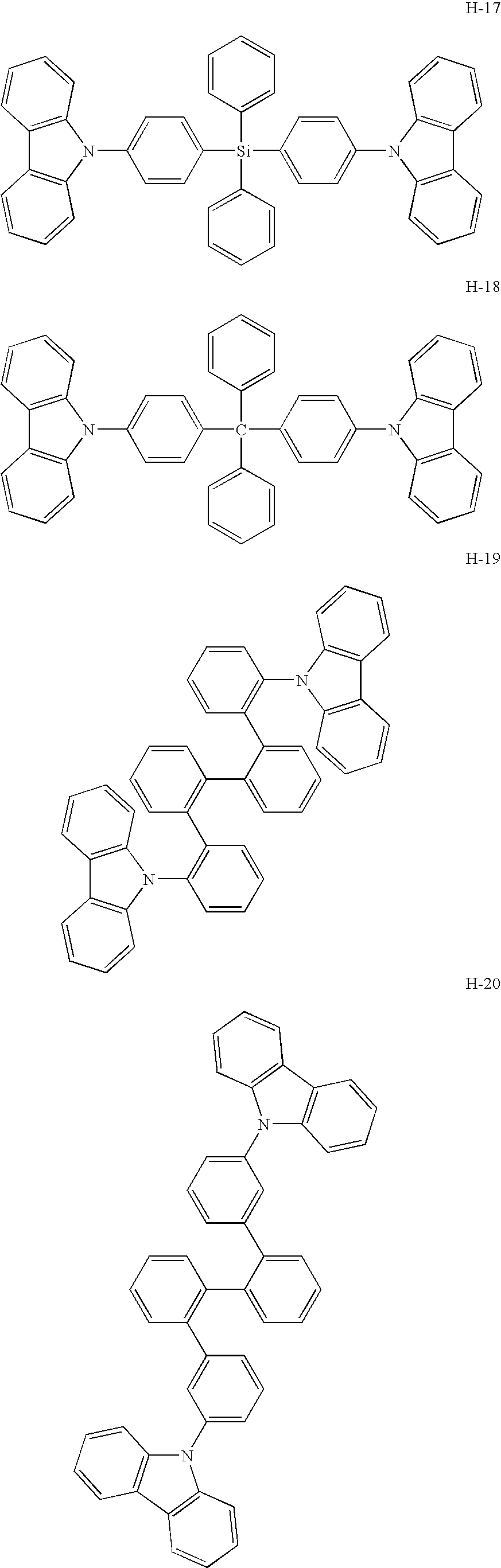
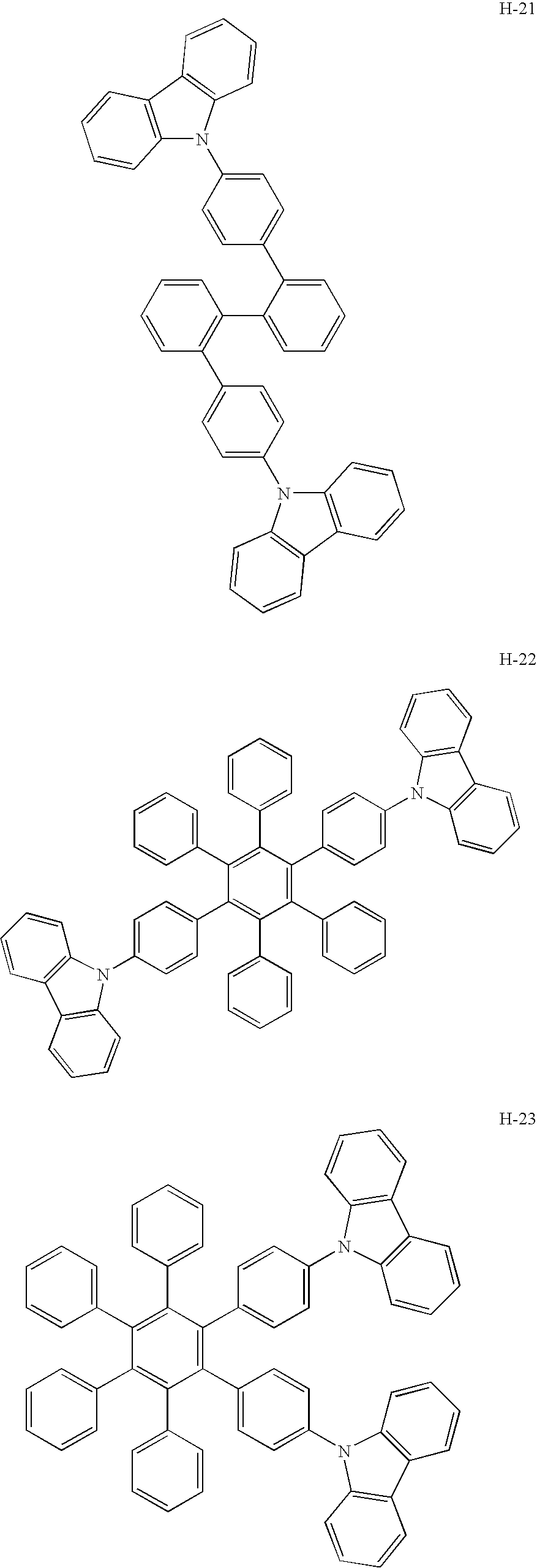
- a platinum complex compound containing a tetradentate ligand for use in the present invention has high electron-transporting performance.
- the use thereof as a host material enables significantly low driving voltage.
- the use of the platinum complex compound (containing the tetradentate ligand) together with a hole-transporting host material as a mixed host realizes lowered driving voltage as well as high emission efficiency.
- the present invention has been made based upon the aforementioned insight of the present inventors, and means for solving the problems are as follows.
- An organic electroluminescence element containing:
- the organic layer disposed between the anode and the cathode, the organic layer including a light-emitting layer,
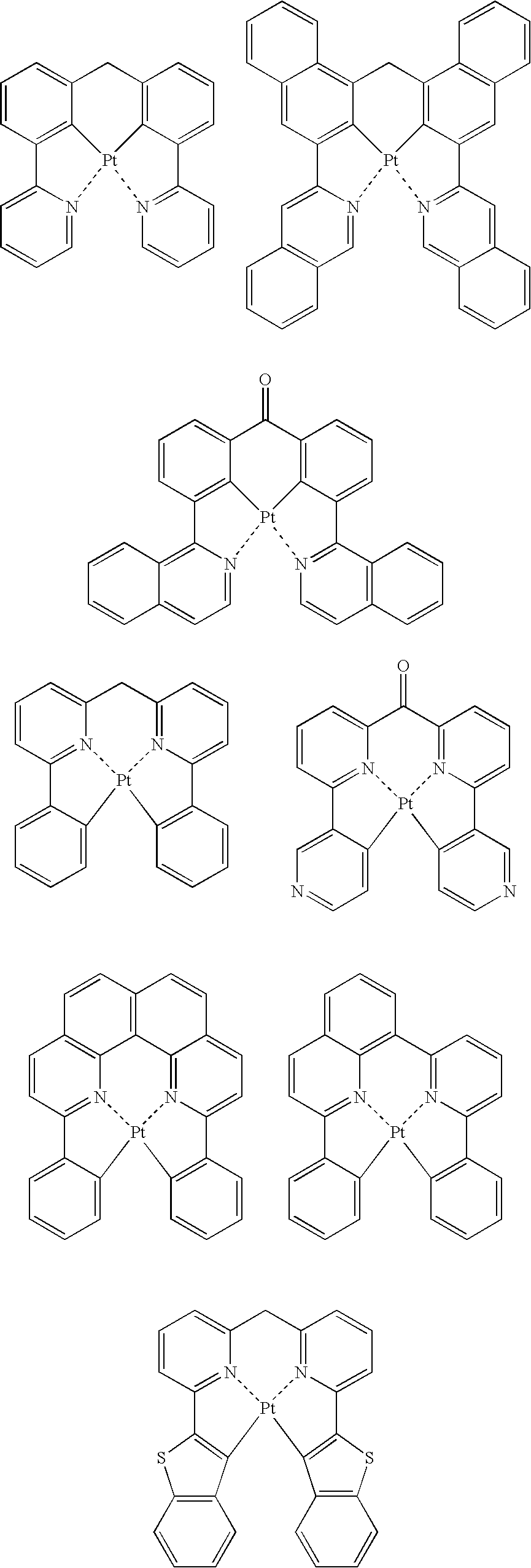
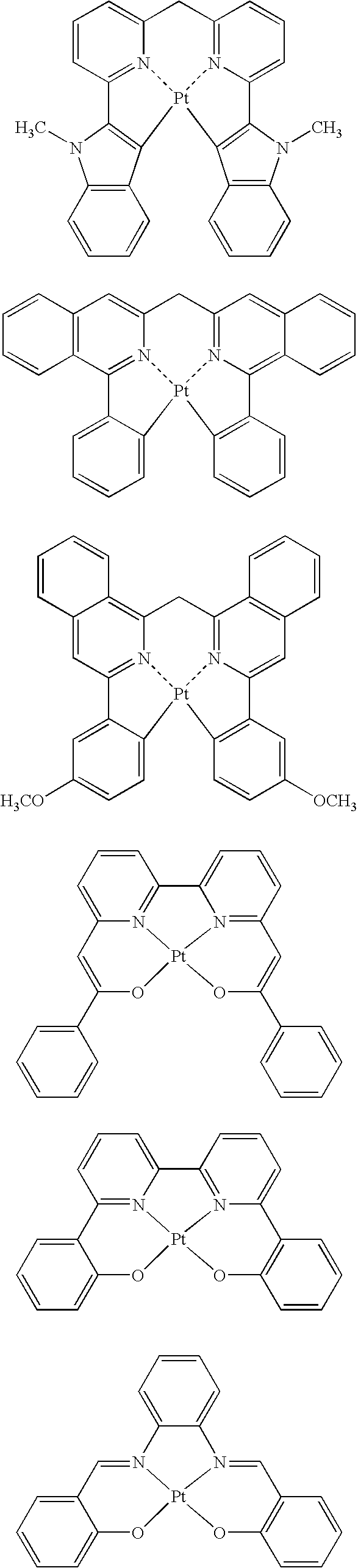
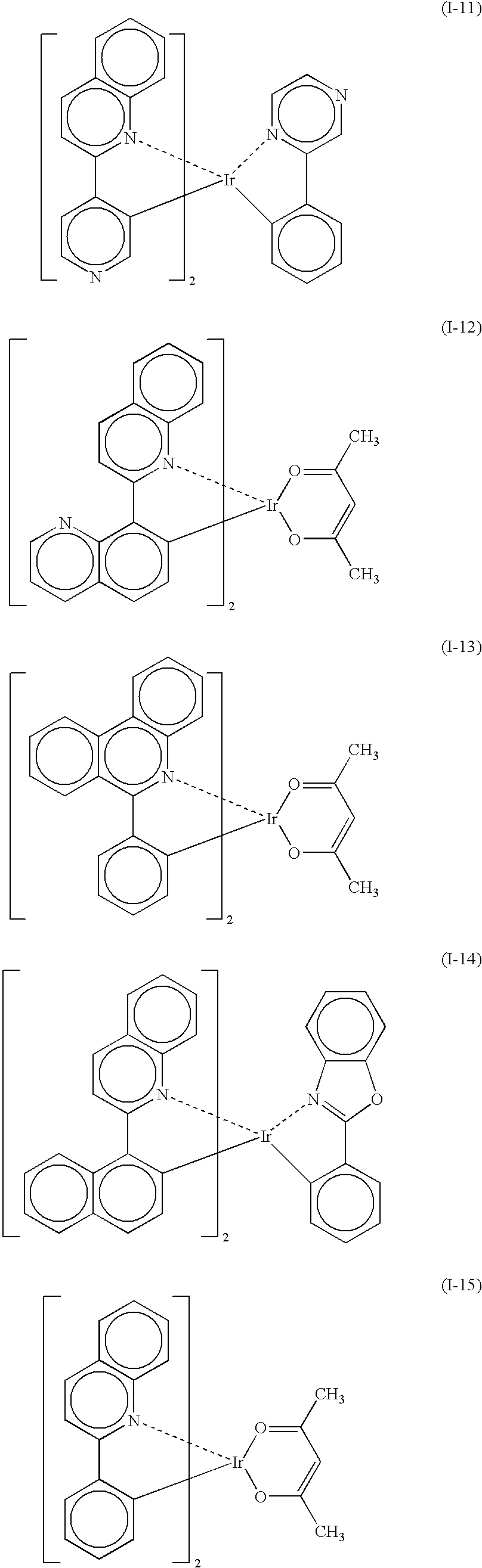
- the light-emitting layer contains a host material and a phosphorescent light-emitting material, and the host material contains at least one platinum complex compound containing a tetradentade ligand, expressed by the following general formula 1;
- L 1 , L 2 , and L 3 are each a single bond or a bridging group
- R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , and R 8 are each a hydrogen atom or a substituent, and at least one of R 1 to R 8 is a phenyl group or a cyano group
- R a and R b are each a substituent
- n and m are each an integer of 0 to 3.
- ⁇ 2> The organic electroluminescence element according to ⁇ 1>, wherein the organic electroluminescence element exhibits a luminescence peak at 550 nm or more.
- ⁇ 3> The organic electroluminescence element according to any of ⁇ 1> or ⁇ 2>, wherein the host material contains at least one hole transporting host material.
- ⁇ 4> The organic electroluminescence element according to any one of ⁇ 1> to ⁇ 3>, wherein the phosphorescent light-emitting material is a compound expressed by any of the following general formulae 2 to 4;
- n is an integer of 1 to 3;
- X-Y represents a bidentate ligand;
- a ring A is a ring structure which may contain at least one selected from the group consisting of a nitrogen atom, a sulfur atom, and an oxygen atom;
- R 11 is a substituent, m1 is an integer of 0 to 6, and in the case where m1 is 2 or more, a plurality of R 11 s adjacent to each other may bond to form a ring, which may contain at least one selected from the group consisting of a nitrogen atom, a sulfur atom, and an oxygen atom, and may have further one or more substituents;
- R 12 is a substituent, m2 is an integer of 0 to 4, and in the case where m2 is 2 or more, a plurality of R 12 s adjacent to each other may bond to form a ring, which may contain at least one selected from the group consisting of a nitrogen atom, a sulfur atom, and an oxygen atom
- the present invention can solve the problems in the art and provide an organic electroluminescence element that, can maintain high emission efficiency while lowering driving voltage.
- FIG. 1 is a schematic diagram showing an example of a layer structure of the organic electroluminescence element of the invention.
- the organic electroluminescence element of the present invention contains an anode, a cathode, and at least one organic layer containing a light-emitting layer, disposed between the anode and the cathode, and may further contain other layers, if necessary.
- the light-emitting layer contains a host material and a phosphorescent light-emitting material, and may further contain other substances, if necessary.
- a host material contain at least one platinum complex compound containing a tetradentate ligand expressed by the following general formula 1, and further contain at least one hole-transporting host material.
- L 1 , L 2 , and L 3 are each a single bond or a bridging group;
- R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , and R 8 are each a hydrogen atom or a substituent, and at least one of R 1 to R 8 is a phenyl group or a cyano group;
- R a and R b are each a substituent; and
- n and m are each an integer of 0 to 3.
- Bridging groups denoted as L 1 , L 2 , and L 3 may be suitably selected depending on the intended purpose without any restriction. Examples thereof include: an alkylene group such as a methylene group, a dimethylene group, a diisopropylmethylene group, a diphenylmethylene group, an ethylene group, and a tetramethylethylene group; an alkenylene group such as a vinylene group, and a dimethylvinylene group; an alkynylene group such as an ethnylene group; an arylene group such as a phenylene group and a naphthylene group; a heteroarylene group such as a pyridilene group, a pyradilene group, and a quinolilene group; an oxygen bridging group; a sulfur bridging group; a nitrogen bridging group such as a methyl amino bridging group, a phenyl amino bridging group, and a t-butyl amino
- R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 8 , R a , and R b may be suitably selected depending on the intended purpose without any restriction.
- Examples thereof include: an alkyl group, preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, even more preferably 1 to 10 carbon atoms, such as a methyl group, an ethyl group, an isopropyl group, a tert-butyl group, an n-octyl group, an n-decyl group, an n-hexadecyl group, a cyclopropyl group, a cyclopentyl group, and a cyclohexyl group; an alkenyl group, preferably having 2 to 30 carbon atoms, more preferably 2 to 20 carbon atoms, yet more preferably 2 to 10 carbon atoms, such as a vinyl group, an allyl group, a 2-butenyl group, and a 3-pentenyl group; an alkynyl group, preferably having 2 to 30 carbon atoms, more preferably 2 to 20 carbon atoms, even more preferably 2 to
- platinum complex compound containing the tetradentate ligand expressed by the general formula 1 include the following compounds, but the examples are not limited to the followings.
- the amount of the platinum complex compound containing the tetradentade ligand, expressed by the general formula 1, is preferably 5% by mass to 99.5% by mass, more preferably 10% by mass to 99.5% by mass, even more preferably 10% by mass to 50% by mass, relative to the total amount of all the compounds contained in the light emitting layer.
- the hole-transporting host material may be suitably selected depending on the intended purpose without any restriction.
- Examples thereof include pyrrole, indole, carbazole, azaindole, azacarbazole, pyrazole, imidazole, polyaryl alkane, pyrazoline, pyrazolone, phenylenediamine, arylamine, amino-substituted chalcone, styrylanthracene, fluorenone, hydrazone, stilbene, silazane, atomatic tertiary amine compound, styrylamine compound, aromatic dimethylidine compound, porphyrin compound, polysilane compound, poly(N-vinyl carbazole), aniline copolymer, high-molecular weight conductive oligomer, such as thiophene oilomer and polythiophene, organic silane, carbon film, and derivatives thereof.
- indole derivatives carbazole derivatives, azaindole derivatives, azacarbazole derivatives, aromatic tertiary amine compounds and thiophene derivatives are preferable, and those containing a plurality of insole structures, carbazole structures, azaindole structures, azacarbazole structures, or atomatic tertiary amine structures per molecule are particularly preferable.
- a host material for use in the present invention a host material in which part of or all of the hydrogen atoms contained therein are substituted with deuterium may be used (see JP-A Nos. 2009-277790, and 2004-515506).
- hole-transporting host material examples include the following compounds, but not limited thereto.
- the amount of the hole-transporting host material is preferably 10% by mass to 99% by mass, more preferably 10% by mass to 90% by mass, even more preferably 20% by mass to 90% by mass, relative to the total amount of all the compounds contained in the light-emitting layer.
- a complex containing a transition metal atom or a lanthanoid atom is generally used.
- the transition metal include ruthenium, rhodium, palladium, tungsten, rhenium, osmium, iridium, and platinum. Among them, rhenium, iridium, and platinum are preferable, iridium and platinum being more preferable.
- Examples of the lanthanoid atom include lanthanum, cerium, praseodymium, neodymium, samarium, europium, gadolinium, teibium, dysprosium, holmium, erbium, thulium, ytterbium, and lutetecium. Among them, neodymium, europium, and gadolinium are particularly preferable.
- ligand of the complex examples include ligands disclosed in G. Wilkinson et al., Comprehensive Coordination Chemistry , Pergamon Press, 1987; H. Yersin, Photochemistry and Photophysics of Coordination Compounds , Springer-Verlag, 1987; and Akio Yamamoto, Organic Metal Chemistry—Foundation and Application , Shokado Publishing Co., Ltd., 1982.
- the ligand include: a halogen ligand, preferably a chlorine ligand; a aromatic carbon ring ligand, such as a cyclopentadienyl anion, a benzene anion, and a naphthyl anion; a nitrogen-containing heterocyclic ligand, such as a phenyl pyridine, a benzoquinoline, a quinolinol, bipyridyl, and a phenanthroline; a diketone ligand, such as a acetyl acetone; a carboxylic acid ligand, such as an acetic acid ligand; an alkolate ligand, such as a phenolate ligand; a carbon monoxide ligand; an isonitrile ligand; and a cyano ligand.
- the nitrogen-containing heterocyclic ligand is preferable.
- the complex may contain one transition metal in the compound.
- the complex may be a binuclear complex containing two or more transition metals.
- different types of the metal atoms may be contained at the same time.
- the following are listed as specific examples of the light-emitting material, but the examples of the light-emitting material are not limited to the following.
- the phosphorescent light-emitting material may be suitably selected depending on the intended purpose without any restriction, but is preferably a compound expressed by any one of the following general formulae 2 to 4:
- n is an integer of 1 to 3;
- X-Y represents a bidentate ligand;
- a ring A is a ring structure which may contain at least one selected from the group consisting of a nitrogen atom, a sulfur atom, and an oxygen atom;
- R 11 is a substituent, m1 is an integer of 0 to 6, and in the case where m1 is 2 or more, a plurality of R 11 s adjacent to each other may bond to form a ring, which may contain at least one selected from the group consisting of a nitrogen atom, a sulfur atom, and an oxygen atom, and may further contain one or more substituents;
- R 12 is a substituent, m2 is an integer of 0 to 4, and in the case where m2 is 2 or more, a plurality of R 12 s adjacent to each other may bond to form a ring, which may contain at least one selected from the group consisting of a nitrogen atom, a sulfur atom, and an oxygen atom
- the ring A denotes a ring structure, which may contain at least one selected from the group consisting of a nitrogen atom, a sulfur atom, and an oxygen atom, and is preferably a five-member ring, a six-member ring, or the like.
- the ring may contain one or more substituents.
- X-Y denotes a bidentate ligand, and preferable examples thereof include bidentate monoanion ligands, and the like.
- bidentate monoanion ligand examples include picolinate (pic), acetylacetonate (acac), and dipicaloylmethanate (t-butyl acac).
- ligands examples include, other than listed above, ligands disclosed in International Publication No. WO 02/15645 (Lamansky et al., pp. 89-91).
- Substituents denoted as R 11 and R 12 may be suitably selected depending on the intended purpose without any restriction. Examples thereof include halogen atoms, alkoxy groups, amino groups, alkyl groups, cycloalkyl groups, aryl groups which may contain a nitrogen atom or sulfur atom, and aryloxy groups which may contain a nitrogen atom or sulfur atom. These substituents may further contain one or more substituents therein.
- Adjacent substituents denoted as R 11 and R 12 may bond to each other to form a ring that may contain a nitrogen atom, a sulfur atom, or an oxygen atom, and suitable examples of such the ring include a five-member ring and a six-member ring. In addition, such the ring may further contain one or more substituents.
- the amount of phosphorescent light-emitting material is preferably 0.5% by mass to 30% by mass, more preferably 0.5% by mass to 20% by mass, more preferably 3% by mass to 10% by mass relative to the total amount of all the compounds contained in the light-emitting layer.
- the emission efficiency When the amount thereof is less than 0.5% by mass, the emission efficiency may be undesirably low. When the amount thereof is more than 30% by mass, the emission efficiency may be undesirably low due to the aggregations between the phosphorescent light-emitting materials.
- the light-emitting layer has the function of receiving holes from the anode, the hole-injection layer, or the hole-transport layer, receiving electrons from the cathode, the electron-injection layer, or the electron-transport layer, and providing a field for recombination of the holes with the electrons for light emission, when an electric field is applied.
- the light-emitting layer can be formed by known methods in the art, without any restriction.
- Preferable examples of the forming method thereof include: dry film-forming methods such as vapor deposition, and sputtering; wet coating; transferring; printing; and inkjet printing.
- the thickness of the light-emitting layer may be suitably adjusted depending on the intended purpose without any restriction.
- the thickness thereof is preferably 2 nm to 500 nm, more preferably 3 nm to 200 nm, even more preferably 10 nm to 200 nm, in view of the emission efficiency.
- the light-emitting layer may be formed of a single layer, or, two or more layers.
- the organic electroluminescence element contains an organic layer including the light-emitting layer between an anode and a cathode, and may further contain other layers, if necessary.
- the organic layer contains at least the light-emitting layer, an electron-transport layer, and an electron-injection layer, and optionally further contains a hole-injection layer, a hole-transport layer, a hole-blocking layer, an electron-blocking layer, and the like.
- the electron-injection layer and the electron-transport layer both have a function of receiving electrons from the anode side and transport to the cathode side.
- the electron-injection layer and the electron-transport layer may be of a monolayer structure, or a laminate structure containing a plurality of layers each formed of identical or different compositions.
- the electron-injection layer and the electron-transport layer may be suitably selected depending on the intended purpose without any restriction.
- the material for forming the electron-injection layer and/or the electron-transport layer include: triazole derivative; oxazole derivative; oxadiazole derivative; fluorenone derivative; anthraquinodimehane derivative; anthrone derivative; diphenylquinone derivative; thiopyrandioxide derivative; carbodiimide derivative; fluorenylidenemethane derivative; distyrylpyradine derivative; heterocyclic carboxylic acid anhydride such as naphthalene and perylene; phthalocyanine derivative; metal complex such as 8-quinolinol derivative; metal phthalocyanine; and metal complex containing benzoxazole or benzothiazole as a ligand.
- the electron-injection layer and the electron-transport layer may contain a hole-accepting dopant, respectively.
- the hole-accepting dopant may be an inorganic compound or an organic compound, provided that it accepts holes, and has a function of reducing organic compounds.
- the inorganic compound may be suitably selected depending on the intended purpose without any restriction. Examples thereof include alkali metals, alkaline earth metals, and metal oxides thereof.
- the electron-injection layer and the electron-transport layer each preferably have a thickness of 1 nm to 5 ⁇ m, more preferably 5 nm to 1 ⁇ m, even more preferably 10 nm to 500 nm.
- the hole-injection layer and the hole-transport layer each have a function of receiving holes from the anode or the anode side and transporting to the cathode side.
- the hole-injection layer and the hole-transport layer may be of a monolayer structure, or a laminate structure containing a plurality of layers each formed of identical or different compositions.
- the hole-injection material or hole-transport material used for these layers may be a low-molecular-weight compound or a high-molecular-weight compound.
- the hole-injection material or the hole-transport material may be suitably selected depending on the intended purpose without any restriction.
- Examples thereof include pyrrole derivatives, carbazole derivatives, triazole derivatives, oxazole derivatives, oxadiazole derivatives, imidazole derivatives, polyarylalkane derivatives, pyrazoline derivatives, pyrazolone derivatives, phenylenediamine derivatives, arylamine derivatives, amino-substituted chalcone derivatives, styrylanthracene derivatives, fluorenone derivatives, hydrazone derivatives, stilbene derivatives, silazane derivatives, aromatic tertiary amine compounds, styrylamine compounds, aromatic dimethylidine compounds, phthalocyanine compounds, porphyrin compounds, thiophene derivatives, organosilane derivatives and carbon. These may be used independently or in combination.
- the hole-injection layer and the hole-transport layer, respectively, may contain an electron-accepting dopant.
- the electron-accepting dopant may be an inorganic compound or an organic compound, provided that it accepts electrons, and has a function of oxidizing organic compounds.
- the inorganic compound is suitably selected depending on the intended purpose without any restriction.
- examples thereof include: metal halides, such as ferric chloride, aluminum chloride, gallium chloride, indium chloride and antimony pentachloride; and metal oxides such as vanadium pentaoxide and molybdenum trioxide.
- the organic compound may be suitably selected depending on the intended purpose without any restriction.
- examples thereof include: those having a substituent such as a nitro group, a halogen atom, a cyano group and a trifluoromethyl group; quinone compounds; acid anhydride compounds; and fullerenes.
- These electron-accepting dopants may be used independently, or in combination.
- the amount of the electron-accepting dopant is not restricted, though it may vary depending on the types of the materials thereof.
- the amount thereof is preferably 0.01% by mass to 50% by mass, more preferably 0.05% by mass to 30% by mass, even more preferably 0.1% by mass to 30% by mass relative to the amount of the hole-transport material or hole-injection material.
- the hole-injection layer and the hole-transport layer may be formed by known methods in the art, without any restriction.
- forming methods thereof include: dry film-forming methods such as vapor deposition and sputtering; wet film-forming methods; transferring; printing; and ink-jet printing.
- the hole-injection layer and the hole-transport layer preferably each have a thickness of 1 nm to 500 nm, more preferably 5 nm to 250 nm, yet more preferably 10 nm to 200 nm.
- the hole-blocking layer has a function of preventing holes transported from the anode side to the light-emitting layer from passing through to the cathode side, and is generally provided as an organic layer adjacent to the light-emitting layer on the cathode side.
- the electron-blocking layer has the function to prevent the electrons transported from the cathode side to the light-emitting layer from passing through to the anode side, and is generally provided as an organic layer adjacent to the light-emitting layer on the anode side.
- Examples of the compound for forming the hole-blocking layer include aluminum complexes such as BAlq; triazole derivatives; and phenanthroline derivatives such as BCP.
- Examples of the compound for forming the electron-blocking layer are those listed above as the hole-transport material.
- the electron-blocking layer and the hole-blocking layer may be formed by known methods in the art, without any restriction.
- forming methods thereof include: dry film-forming methods such as vapor deposition and sputtering; wet film-forming methods; transferring; printing; and ink-jet printing.
- the hole-blocking layer and the electron-blocking layer each preferably have a thickness of 1 nm to 200 nm, more preferably 1 nm to 50 nm, yet more preferably 3 nm to 10 nm.
- the hole-blocking layer and the electron-blocking layer may be of a monolayer structure forming of one or more materials mentioned above, or of a laminate structure having a plurality of layers each formed of identical or different compositions.
- the organic electroluminescence element contains a pair of electrodes; i.e., an anode and a cathode.
- a pair of electrodes i.e., an anode and a cathode.
- at least one of the anode and the cathode is preferably transparent.
- the anode may serve as an electrode which supplies holes to an organic layer
- the cathode may serve as an electrode which injects electrons into an organic layer.
- the electrode may be suitably selected from the electrode materials known in the art depending on the use of the organic electroluminescence element, without any restriction.
- the material for forming the electrode include metals, alloys, metal oxides, conductive compounds, and mixtures thereof.
- Examples of the material for forming the anode include; conductive metal oxides such as tin oxides doped with, for example, antimony and fluorine (ATO and FTO); tin oxide, zinc oxide, indium oxide, indium tin oxide (ITO) and indium zinc oxide (IZO); metals such as gold, silver, chromium and nickel; mixtures or laminates of these metals and the conductive metal oxides; inorganic conductive materials such as copper iodide and copper sulfide; organic conductive materials such as polyaniline, polythiophene and polypyrrole; and laminates of these materials and ITO.
- conductive metal oxides are preferable.
- ITO is preferable for its productivity, high conductivity, and transparency.
- Examples of the material for forming the cathode include alkali metals (e.g., Li, Na, K and Cs), alkaline earth metals (e.g., Mg and Ca), gold, silver, lead, aluminum, sodium-potassium alloys, lithium-aluminum alloys, magnesium-silver alloys and rare earth metals (e.g., indium and ytterbium). These may be used independently, but preferably in combination for the purpose of favorable stability and electron-injection properties.
- alkali metals e.g., Li, Na, K and Cs
- alkaline earth metals e.g., Mg and Ca
- gold, silver, lead, aluminum, sodium-potassium alloys, lithium-aluminum alloys, magnesium-silver alloys and rare earth metals e.g., indium and ytterbium.
- alkali metals and alkaline earth metals are preferable in terms of the electron-injection properties, and the material containing aluminum as a main component is preferable in terms of excellent storage stability.
- material containing aluminum as a main component refers to a material composed of aluminum alone; alloys containing aluminum and 0.01% by mass to 10% by mass of an alkali or alkaline earth metal; or the mixtures thereof (e.g., lithium-aluminum alloys and magnesium-aluminum alloys).
- the electrode can be formed by known methods in the art, without any restriction. Examples of forming methods of the electrode include: wet methods such as printing and coating; physical methods such as vacuum deposition, sputtering, and ion plating; and chemical methods such as CVD, and plasma CDV. According to a method appropriately selected from these methods in consideration of suitability for the material constituting the anode, the anode can be formed on a substrate. For example, when ITO is used as the material for the anode, the anode may be formed with DC or high-frequency sputtering methods, vacuum deposition methods, or ion plating methods. Notably, when metals are used as a material for the below-described cathode, the cathode can be formed by, for example, sputtering one type of metal, or, two or more types of metals simultaneously or sequentially.
- Patterning for forming the anode may be performed by a chemical etching method such as photolithography; a physical etching method such as etching by laser; a method of vacuum deposition or sputtering using a mask; a lift-off method; or a printing method.
- the organic electroluminescence element is preferably provided on a substrate.
- the organic electroluminescence element may be provided so that the electrode thereof is in direct contact with the substrate, or so that the electrode is in contact with the substrate via an intermediate layer.
- the material for the substrate may be suitably selected depending on the intended purpose without any restriction.
- examples thereof include inorganic materials such as yttria-stabilized zirconia (YSZ) and glass (alkali-free glass and soda-lime glass); and organic materials such as polyesters (e.g., polyethylene terephthalate, polybutylene phthalate and polyethylene naphthalate), polystyrene, polycarbonate, polyether sulfone, polyarylate, polyimide, polycycloolefin, norbornene resins and poly(chlorotrifluoroethylene).
- YSZ yttria-stabilized zirconia
- glass alkali-free glass and soda-lime glass
- organic materials such as polyesters (e.g., polyethylene terephthalate, polybutylene phthalate and polyethylene naphthalate), polystyrene, polycarbonate, polyether sulfone, polyarylate, polyimide, polycyclo
- the substrate may be suitably adjusted depending on the use, purpose, and the like of the electroluminescence element, without any restriction. In general, it is preferable to provide the substrate in the form of a sheet.
- the substrate may have a single- or multi-layered structure, and may be a single member or a combination of two or more members.
- the substrate may be opaque, colorless transparent, or colored transparent.
- the substrate may be provided with a moisture permeation-preventing layer (gas barrier layer) on the front and/or back surface thereof.
- a moisture permeation-preventing layer gas barrier layer
- the moisture permeation-preventing layer (gas barrier layer) is preferably made from an inorganic compound such as silicon nitride and silicon oxide.
- the moisture permeation-preventing layer (gas barrier layer) can be formed through, for example, high-frequency sputtering.
- the entire organic electroluminescence element may be protected with a protective layer.
- the materials contained in the protective layer may be suitably selected depending on the intended purpose without any restriction, provided that the resulted protective layer will have the function to prevent water, oxygen, and the like, which promote the degradation of the element, from entering into the element.
- examples thereof include: metals such as In, Sn, Pb, Au, Cu, Ag, Al, Ti and Ni; metal oxides such as MgO, SiO, SiO 2 , Al 2 O 3 , GeO, NiO, CaO, BaO, Fe 2 O 3 , Y 2 O 3 and TiO 2 ; metal nitrides such as SiN x and SiN x O y ; metal fluorides such as MgF 2 , LiF, AlF 3 and CaF 2 ; polyethylenes, polypropylenes, polymethyl methacrylates, polyimides, polyureas, polytetrafluoroethylenes, polychlorotrifluoroethylens, polydichlorodifluoroethylenes, copolymers of
- the formation method of the protective layer is suitably selected depending on the intended purpose without any restriction. Examples thereof include vacuum deposition, sputtering, reactive sputtering, molecular beam epitaxial (MBE), cluster ion beam, ion plating, plasma polymerization (high-frequency excitation ion plating), plasma CVD, laser CVD, thermal CVD, gas source CVD, coating, printing and transferring.
- vacuum deposition sputtering, reactive sputtering, molecular beam epitaxial (MBE), cluster ion beam, ion plating, plasma polymerization (high-frequency excitation ion plating), plasma CVD, laser CVD, thermal CVD, gas source CVD, coating, printing and transferring.
- the organic electroluminescence element may be entirely sealed with a seal container. Further, a moisture absorbent or an inert liquid may be contained in the space between the seal container and the organic electroluminescence element.
- the moisture absorbent is not particularly limited and may be appropriately selected depending on the purpose. Examples thereof include barium oxide, sodium oxide, potassium oxide, calcium oxide, sodium sulfate, calcium sulfate, magnesium sulfate, phosphorus pentaoxide, calcium chloride, magnesium chloride, copper chloride, cesium fluoride, niobium fluoride, calcium bromide, vanadium bromide, molecular sieve, zeolite and magnesium oxide.
- the inert liquid is not particularly limited and may be appropriately selected depending on the purpose.
- examples thereof include paraffins; liquid paraffins; fluorine-containing solvents such as perfluoroalkanes, perfluoroamines and perfluoroethers; chlorinated solvents; and silicone oils.
- the organic electroluminescence element is preferably sealed with a resin seal layer so as to prevent degradation thereof due to oxygen and/or moisture contained in the air.
- the resin material for the resin seal layer may be suitably selected depending on the intended purpose without any restriction.
- examples thereof include acrylic resins, epoxy resins, fluorine-containing resins, silicone resins, rubber resins and ester resins.
- epoxy resins are preferred from the standpoint of its excellent properties in water impermeability.
- thermosetting epoxy resins and photo-curable epoxy resins are preferred.
- the forming method for the resin seal layer is not particularly limited and may be appropriately selected depending on the purpose. Examples thereof include a method by coating a resin solution, a method by press-bonding or hot press-bonding a resin sheet, and a method by polymerizing under dry conditions (e.g., vapor deposition and sputtering).
- the organic electroluminescence element contains a sealing adhesive having the function of preventing permeation of moisture or oxygen from the edges thereof.
- the material for the sealing adhesive may be those used in the resin seal layer.
- epoxy resins are preferred from the viewpoint of preventing water permeation, with photo-curable epoxy resins and thermosetting epoxy resins being more preferred.
- a filler is preferably added to the sealing adhesive.
- the filler is preferably inorganic materials such as SiO 2 , SiO (silicon oxide), SiON (silicon oxynitride) and SiN (silicon nitride). The filler increases the viscosity of the sealing adhesive to improve processability and humidity resistance.
- the sealing adhesive may also contain a desiccant.
- the desiccant include barium oxide, calcium oxide or strontium oxide.
- the amount of the desiccant added to the sealing adhesive is preferably 0.01% by mass to 20% by mass, more preferably 0.05% by mass to 15% by mass. When the amount is less than 0.01% by mass, the desiccant exhibits reduced effects. Whereas when the amount is more than 20% by mass, it may be difficult to homogeneously disperse the desiccant in the sealing adhesive.
- the sealing adhesive containing the desiccant is applied in a predetermined amount using, for example, a dispenser. Thereafter, a second substrate is overlaid, followed by curing for sealing.
- FIG. 1 is a schematic diagram showing one example of the layer structure of the organic electroluminescence element of the invention.
- the organic EL element 10 has a layer structure in which an anode 2 (e.g., ITO electrode) formed on a glass substrate 1 , a hole-injection layer 3 , a hole-transport layer 4 , a light-emitting layer 5 , an electron-transport layer 6 , an electron-injection layer 7 , and a cathode 8 (e.g., Al—Li electrode) are laminated in this order.
- the anode 2 (e.g., ITO electrode) and the cathode 8 e.g., Al—Li electrode
- the cathode 8 e.g., Al—Li electrode
- the organic electroluminescence element can emit light when a DC voltage (which, if necessary, may contain AC components) (generally 2 volts to 15 volts) or a DC is applied to between the anode and the cathode.
- a DC voltage which, if necessary, may contain AC components
- AC components generally 2 volts to 15 volts
- the organic electroluminescence element can be applied to an active matrix by a thin film transistor (TFT).
- TFT thin film transistor
- An active layer of the thin film transistor can be formed from, for example, amorphous silicone, high-temperature polysilicone, low-temperature polysilicone, microcrystalline silicone, oxide semiconductor, organic semiconductor or carbon nanotube.
- Examples of the thin film transistor applicable in the organic electroluminescence element include those described in, for example, WO2005/088726, JP-A No. 2006-165529, and U.S. Pat. Application Publication No. 2008/0237598 A1.
- the light-extraction efficiency of the organic electroluminescence element of the present invention can be improved by various known methods, without any restriction. It is possible to increase the light-extraction efficiency to improve the external quantum efficiency, for example, by processing the surface shape of the substrate (for example, by forming a fine concavo-convex pattern), by controlling the refractive index of the substrate, the ITO layer and/or the organic layer, or by controlling the thickness of the substrate, the ITO layer and/or the organic layer.
- the organic electroluminescence element may be used in a top-emission configuration or a bottom-emission configuration, in order for light to be extracted.
- the organic electroluminescence element may have a resonator structure.
- a multi-layered film mirror composed of a plurality of laminated films having different reflective indices, a transparent or semi-transparent electrode, a light-emitting layer and a metal electrode.
- the light generated in the light-emitting layer is repeatedly reflected between the multi-layered film mirror and the metal electrode (which serve as reflection plates). Therefore, in this matter the light is resonated.
- a transparent or semi-transparent electrode and a metal electrode are stacked on a transparent substrate.
- the light generated in the light-emitting layer is repeatedly reflected between the transparent or semi-transparent electrode and the metal electrode (which serve as reflection plates); i.e., is resonated.
- an optical path length determined based on the effective refractive index of two reflection plates, and on the refractive index and the thickness of each of the layers between the reflection plates is adjusted to be an optimal value for obtaining a desired resonance wavelength.
- the calculation formula applied in the case of the first embodiment is described in JP-A No. 09-180883.
- the calculation formula in the case of the second embodiment is described in JP-A No. 2004-127795.
- the organic electroluminescence element of the present invention is suitably selected depending on the intended purpose without any restriction.
- the organic electroluminescence element of the present invention can be suitable used in display elements, displays, backlights, electrophotography, illuminating light sources, recording light sources, exposing light sources, reading light sources, markers, signs, interior accessories and optical communication.
- a tricolor light emission method by arranging, on a substrate, organic EL elements emitting lights corresponding to three primary colors (blue color (B), green color (G) and red color (R)); a white color method by separating white light emitted from an organic electroluminescence element for white color emission into three primary colors through a color filter; and a color conversion method by converting a blue light emitted from an organic electroluminescence element for blue light emission into red color (R) and green color (G) through a fluorescent dye layer.
- a glass substrate having a thickness of 0.5 mm, and a size of 2.5 cm ⁇ 2.5 cm was placed in a washing container, subjected to ultrasonic washing in 2-propanol, followed by subjected to a UV-ozone treatment for 30 minutes.
- the following layers were deposited by vacuum deposition. Note that, in Examples and Comparative Examples, the deposition rate was 0.2 nm/sec., unless otherwise stated. The deposition rate was measured by a crystal resonator. In addition, the thickness of each layer described below was also measured by a crystal resonator.
- ITO Indium Tin Oxide
- ⁇ -NPD bis[N-(1-naphthyl)-N-pheny]benzidine
- a light-emitting layer was deposited in the thickness of 30 nm by vapor deposition.
- the light-emitting layer was formed of the platinum complex A expressed by the following structural formula, which served as a host material 1, and the compound (I-15) expressed by the following structural formula, which served as a phosphorescent light-emitting material and provided in an amount of 5% by mass relative to the amount of the host material 1.
- BAlq bis-(2-methyl-8-quinolinolato)-4-(phenyl-phenolate)-aluminium (III)
- BAlq bis-(2-methyl-8-quinolinolato)-4-(phenyl-phenolate)-aluminium (III)
- LiF was deposited in the thickness of 1 nm by vapor deposition, as an electron injection layer.
- a patterned mask (the mask to give a pattern having a light-emitting region of 2 mm ⁇ 2 mm) was placed on the electron injection layer, and aluminum was deposited thereon in the thickness of 100 nm by vapor deposition as a cathode.
- the obtained laminate was placed in a glove compartment in which the atmosphere had been replaced with argon gas, and was sealed by using a sealing tin formed of stainless steel and a UV-curable adhesive (XNR5516HV, manufactured by Nagase ChemteX Corporation).
- a sealing tin formed of stainless steel and a UV-curable adhesive XNR5516HV, manufactured by Nagase ChemteX Corporation.
- the organic electroluminescence element of Comparative Example 2 was prepared in the same manner as Comparative Example 1, provided that the platinum complex A used as the host material 1 in the light-emitting layer was replaced with the platinum complex B expressed by the following structural formula.
- the organic electroluminescence layer of Example 1 was prepared in the same manner as in Comparative Example 1, provided that the platinum complex A used as the host material 1 in the light-emitting layer was replaced with the platinum complex E expressed by the following structural formula.
- the organic electroluminescence layer of Example 2 was prepared in the same manner as in Comparative Example 1, provided that the platinum complex A used as the host material 1 in the light-emitting layer was replaced with the platinum complex F expressed by the following structural formula.
- the organic electroluminescence layer of Example 3 was prepared in the same manner as in Comparative Example 1, provided that the platinum complex A used as the host material 1 in the light-emitting layer was replaced with the platinum complex J expressed by the following structural formula.
- the organic electroluminescence layer of Example 4 was prepared in the same manner as in Comparative Example 1, provided that the platinum complex A used as the host material 1 in the light-emitting layer was replaced with the platinum complex K expressed by the following structural formula.
- the organic electroluminescence element of Comparative Example 3 was prepared in the same manner as Comparative Example 1, provided that the light-emitting layer was replaced with a light-emitting layer containing 20% by mass of BAlq as a host material 1, 75% by mass of the compound (H-24) expressed by the following structural formula as a host material 2, and 5% by mass of the compound (I-15) expressed by the structural formula presented earlier as the phosphorescent light-emitting material.
- the organic electroluminescence element of Comparative Example 4 was prepared in the same manner as Comparative Example 1, provided that the light-emitting layer was replaced with a light-emitting layer containing 20% by mass of the aforementioned platinum complex A as a host material 1, 75% by mass of the compound (H-24) expressed by the structural formula presented earlier as a host material 2, and 5% by mass of the compound (I-15) expressed by the structural formula presented earlier as the phosphorescent light-emitting material.
- the organic electroluminescence element of Example 5 was prepared in the same manner as Comparative Example 1, provided that the light-emitting layer was replaced with alight-emitting layer containing 20% by mass of the aforementioned platinum complex E as a host material 1, 75% by mass of the compound (H-24) expressed by the structural formula presented earlier as a host material 2, and 5% by mass of the compound (I-15) expressed by the structural formula presented earlier as the phosphorescent light-emitting material.
- the organic electroluminescence element of Example 6 was prepared in the same manner as Comparative Example 1, provided that the light-emitting layer was replaced with a light-emitting layer containing 20% by mass of the aforementioned platinum complex F as a host material 1, 75% by mass of the compound (H-24) expressed by the structural formula presented earlier as a host material 2, and 5% by mass of the compound (I-15) expressed by the structural formula presented earlier as the phosphorescent light-emitting material.
- a direct voltage was applied to the organic electroluminescence element by a source measure unit 2400 (manufactured by Keithley Instruments Inc.) to allow the organic electroluminescence element to emit, and the voltage at which the current density was 2.5 mA/cm 2 was measured.
- a direct voltage was applied to the organic electroluminescence element by a source measure unit 2400, manufactured by Keithley Instruments Inc. to allow the organic electroluminescence element to emit.
- the luminance of the emission was measured by a luminance meter (BM-8, manufactured by Topcon Corporation).
- the emission spectrum and the emission wavelength were measured by a spectrum analyzer PMA-11, manufactured by Hamamatsu Photonics K.K. Based on these values, the emission efficiency at the current density of 2.5 mA/cm 2 was calculated as external quantum efficiency in accordance with the luminance conversion method.
- the peak wavelength was determined from the spectrum obtained by a spectrum analyzer PMA-11, manufactured by Hamamatsu Photonics K.K.
- the organic electroluminescence element of the present invention can realize both reduction in voltage for use and high efficiency, it can be suitably applied in display elements, displays, backlights, electrophotography, illuminating light sources, recording light sources, exposing light sources, reading light sources, markers, signs, interior accessories and optical communication.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Organic Chemistry (AREA)
- Crystallography & Structural Chemistry (AREA)
- Inorganic Chemistry (AREA)
- Electroluminescent Light Sources (AREA)
Abstract
To provide an organic electroluminescence element, containing: an anode; a cathode; and at least one organic layer disposed between and the anode and the cathode, the organic layer containing a light-emitting layer, wherein the light-emitting layer contains a host material and a phosphorescent light-emitting material, and the host material contains at least one platinum complex compound containing a tetradentade ligand, expressed by the following general formula 1:
where L1 to L3 are each a single bond or a bridging group; R1 to R8 are each a hydrogen atom or a substituent, and at least one of R1 to R8 is a phenyl group or a cyano group; Ra and Rb are each a substituent; and n and m are each an integer of 0 to 3.
Description
- 1. Field of the Invention
- The present invention relates to an organic electroluminescence element (may also referred to as an organic EL element, hereinafter).
- 2. Description of the Related Art
- Organic electroluminescence elements have characteristics such as self-luminescence, high-speed response, and the like, and thus application thereof in flat panel displays has been expected. Especially after information had been made public regarding 2-layer (laminate) elements in which a hole-transport organic thin film (hole-transport layer) and an electron-transport organic thin film (electron-transport layer) are laminated, organic electroluminescence elements have attracted attention as large-scale light-emitting elements capable of emitting at low voltages of 10 V or less. The laminate organic EL element has a basic structure as follows: an anode, a hole-transport layer, a light-emitting layer, an electron-transport layer, and a cathode, in this order. The organic EL element has realized energy efficiency (i.e. by using lower voltages for emitting light) and high emission efficiency due to the aforementioned structure.
- In the technology of organic electroluminescence elements, various studies have been conducted to realize higher energy efficiency and higher emission efficiency. For example, there has been proposed an organic electroluminescence element in which a host material and a light-emitting material are contained in a light-emitting layer, and the host material contains a certain Pt complex (see Japanese Patent Application Laid-Open (JP-A) No. 2006-332622).
- According to the technique disclosed in JP-A No. 2006-332622, it is possible to improve the energy saving and emission efficiency to a certain degree. However, the current situation is that further improvements in energy efficiency and emission efficiency are desired.
- The present invention aims at providing an organic electroluminescence element that can maintain high emission efficiency while lowering a driving voltage thereof.
- As a result of the diligent researches and studies conducted by the present inventors for solving the problems in the art, the present inventors have come to the insights that a platinum complex compound containing a tetradentate ligand for use in the present invention has high electron-transporting performance. The use thereof as a host material enables significantly low driving voltage. Additionally, the use of the platinum complex compound (containing the tetradentate ligand) together with a hole-transporting host material as a mixed host realizes lowered driving voltage as well as high emission efficiency.
- The present invention has been made based upon the aforementioned insight of the present inventors, and means for solving the problems are as follows.
- <1> An organic electroluminescence element, containing:
- an anode;
- a cathode; and
- at least one organic layer disposed between the anode and the cathode, the organic layer including a light-emitting layer,
- wherein the light-emitting layer contains a host material and a phosphorescent light-emitting material, and the host material contains at least one platinum complex compound containing a tetradentade ligand, expressed by the following
general formula 1; - where L1, L2, and L3 are each a single bond or a bridging group; R1, R2, R3, R4, R5, R6, R7, and R8 are each a hydrogen atom or a substituent, and at least one of R1 to R8 is a phenyl group or a cyano group; Ra and Rb are each a substituent; and n and m are each an integer of 0 to 3.
- <2> The organic electroluminescence element according to <1>, wherein the organic electroluminescence element exhibits a luminescence peak at 550 nm or more.
<3> The organic electroluminescence element according to any of <1> or <2>, wherein the host material contains at least one hole transporting host material.
<4> The organic electroluminescence element according to any one of <1> to <3>, wherein the phosphorescent light-emitting material is a compound expressed by any of the followinggeneral formulae 2 to 4; - where n is an integer of 1 to 3; X-Y represents a bidentate ligand; a ring A is a ring structure which may contain at least one selected from the group consisting of a nitrogen atom, a sulfur atom, and an oxygen atom; R11 is a substituent, m1 is an integer of 0 to 6, and in the case where m1 is 2 or more, a plurality of R11s adjacent to each other may bond to form a ring, which may contain at least one selected from the group consisting of a nitrogen atom, a sulfur atom, and an oxygen atom, and may have further one or more substituents; R12 is a substituent, m2 is an integer of 0 to 4, and in the case where m2 is 2 or more, a plurality of R12s adjacent to each other may bond to form a ring, which may contain at least one selected from the group consisting of a nitrogen atom, a sulfur atom, and an oxygen atom, and may further contain one or more substituents; R11 and R12 may bond to each other to form a ring, which may contain at least one selected from the group consisting of a nitrogen atom, a sulfur atom, and an oxygen atom, and may further contain one or more substituents.
- The present invention can solve the problems in the art and provide an organic electroluminescence element that, can maintain high emission efficiency while lowering driving voltage.
-
FIG. 1 is a schematic diagram showing an example of a layer structure of the organic electroluminescence element of the invention. - The organic electroluminescence element of the present invention contains an anode, a cathode, and at least one organic layer containing a light-emitting layer, disposed between the anode and the cathode, and may further contain other layers, if necessary.
- The light-emitting layer contains a host material and a phosphorescent light-emitting material, and may further contain other substances, if necessary.
- It is preferred that a host material contain at least one platinum complex compound containing a tetradentate ligand expressed by the following
general formula 1, and further contain at least one hole-transporting host material. - In the
general formula 1, L1, L2, and L3 are each a single bond or a bridging group; R1, R2, R3, R4, R5, R6, R7, and R8 are each a hydrogen atom or a substituent, and at least one of R1 to R8 is a phenyl group or a cyano group; Ra and Rb are each a substituent; and n and m are each an integer of 0 to 3. - Bridging groups denoted as L1, L2, and L3 may be suitably selected depending on the intended purpose without any restriction. Examples thereof include: an alkylene group such as a methylene group, a dimethylene group, a diisopropylmethylene group, a diphenylmethylene group, an ethylene group, and a tetramethylethylene group; an alkenylene group such as a vinylene group, and a dimethylvinylene group; an alkynylene group such as an ethnylene group; an arylene group such as a phenylene group and a naphthylene group; a heteroarylene group such as a pyridilene group, a pyradilene group, and a quinolilene group; an oxygen bridging group; a sulfur bridging group; a nitrogen bridging group such as a methyl amino bridging group, a phenyl amino bridging group, and a t-butyl amino bridging group; a silicon bridging group; and a bridging group combined thereof, such as an oxylenemethylene group.
- Substituents denoted as R1, R2, R3, R4, R5, R6, R7, R8, Ra, and Rb may be suitably selected depending on the intended purpose without any restriction. Examples thereof include: an alkyl group, preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, even more preferably 1 to 10 carbon atoms, such as a methyl group, an ethyl group, an isopropyl group, a tert-butyl group, an n-octyl group, an n-decyl group, an n-hexadecyl group, a cyclopropyl group, a cyclopentyl group, and a cyclohexyl group; an alkenyl group, preferably having 2 to 30 carbon atoms, more preferably 2 to 20 carbon atoms, yet more preferably 2 to 10 carbon atoms, such as a vinyl group, an allyl group, a 2-butenyl group, and a 3-pentenyl group; an alkynyl group, preferably having 2 to 30 carbon atoms, more preferably 2 to 20 carbon atoms, even more preferably 2 to 10 carbon atoms, such as a propargyl group, and a 3-pentynyl group; an aryl group, preferably having 6 to 30 carbon atoms, more preferably 6 to 20 carbon atoms, even more preferably 6 to 12 carbon atoms, such as a phenyl group, a p-methylphenyl group, a naphthyl group, and an anthranil group; an amino group, preferably having 0 to 30 carbon atoms, more preferably 0 to 20 carbon atoms, even more preferably 0 to 10 carbon atoms, such as an amino group, a methylamino group, a dimethylamino group, a diethylamino group, a dibenzyl amino group, a diphenylamino group, and a ditolylamino group; an alkoxy group, preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, even more preferably 1 to 10 carbon atoms, such as a methoxy group, an ethoxy group, a butoxy group, and a 2-ethylhexysiloxy group; an aryloxy group, preferably having 6 to 30 carbon atoms, more preferably 6 to 20 carbon atoms, even more preferably 6 to 12 carbon atoms, such as a phenyloxy group, a 1-naphthyloxy group, and a 2-naphthyloxy group; a heterocyclic oxy group, preferably having 1 to 30 carbon atoms, preferably 1 to 20 carbon atoms, even more preferably 1 to 12 carbon atoms, such as a pyridyloxy group, a pyradyloxy group, a pyrimidyloxy group, and a quinolyloxy group; an acyl group, preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, even more preferably 1 to 12 carbon atoms, such as an acetyl group, a benzoyl group, a formyl group, and a pivaloyl group; an alkoxy carbonyl group, preferably having 2 to 30 carbon atoms, more preferably 2 to 20 carbon atoms, even more preferably 2 to 12 carbon atoms, such as a methoxy carbonyl group, and a ethoxy carbonyl group; an aryloxy carbonyl group, preferably having 7 to 30 carbon atoms, more preferably 7 to 20 carbon atoms, even more preferably 7 to 12 carbon atoms, such as a phenyloxy carbonyl group; an acyloxy group, preferably having 2 to 30 carbon atoms, more preferably 2 to 20 carbon atoms, even more preferably 2 to 10 carbon atoms, such as an acetoxy group and a benzoyloxy group; an acylamino group, preferably having 2 to 30 carbon atoms, more preferably 2 to 20 carbon atoms, even more preferably 2 to 10 carbon atoms, such as an acetylamino group, and a benzoylamino group; an alkoxycarbonyl amino group, preferably having 2 to 30 carbon atoms, more preferably 2 to 20 carbon atoms, even more preferably 2 to 12 carbon atoms, such as a methoxycarbonyl amino group; an aryloxycarbonyl amino group, preferably having 7 to 30 carbon atoms, more preferably 7 to 20 carbon atoms, even more preferably 7 to 12 carbon toms, such as a phenyloxycarbonyl amino group; a sulfonyl amino group, preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, even more preferably 1 to 12 carbon atoms, such as a methane sulfonyl amino group, and a benzene sulfonyl amino group; a sulfamoyl group, preferably having 0 to 30 carbon atoms, more preferably 0 to 20 carbon atoms, even more preferably 0 to 12 carbon atoms, such as a sulfamoyl group, a methylsulfamoyl group, a dimethylsulfamoyl group, and a phenylsulfamoyl group; a carbamoyl group, preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, even more preferably 1 to 12 carbon atoms, such as a carbamoyl group, a methylcarbamoyl group, a diethylcarbamoyl group, and a phenylcarbamoyl group; an alkyl thio group, preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, even more preferably 1 to 12 carbon atoms, such as a methylthio group, and an ethyl thio group; an aryl thio group, preferably having 6 to 30 carbon atoms, more preferably 6 to 20 carbon atoms, even more preferably 6 to 12 carbon atoms, such as a phenyl thio group; a heterocyclic-thio group, preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, even more preferably 1 to 12 carbon atoms, such as a pyridyl thio group, a 2-benzimidazolyl thio group, and a 2-benzoxazolyl thio group, a 2-benzothiazolyl thio; a sulfonyl group, preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, even more preferably 1 to 12 carbon atoms, such as a mesyl group, and a tosyl group; a sulfinyl group, preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, even more preferably 1 to 12 carbon atoms, such as a methane sulfinyl group, and a benzene sulfinyl group; a ureide group, preferably having 1 to 30 carbon atoms, more preferably having 1 to 20 carbon atoms, even more preferably 1 to 12 carbon atoms, such as a ureide group, a methyl ureide group, and a phenyl ureide group; a phosphoric amide group, preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, even more preferably 1 to 12 carbon atoms, such as a diethylphosphoric amide group, and a phenylphosphoric amide group; a hydroxyl group; a mercapto group; a halogen atom, such as a fluorine atom, a chlorine atom, a bromine atom, and a iodine atom; a cyano group; a sulfo group; a carboxyl group; a nitro group; a hydroxamic acid group; a sulfino group; a hydrazino group; an imino group; a heterocyclic group, preferably having 1 to 30 carbon atoms, more preferably 1 to 12 carbon atoms, including nitrogen atoms, oxygen atoms, or sulfur atoms as heteroatoms, such as a imidazolyl group, a pyridyl group, a quinolyl group, a furyl group, a thienyl group, a piperidyl group, a morpholino group, a benzoxazolyl group, a benzimidazolyl group, a benzthiazolyl group, a carbazolyl group, and an azepynyl group; a silyl group, preferably having 3 to 40 carbon atoms, more preferably 3 to 30 carbon atoms, even more preferably 3 to 24 carbon atoms, such as a trimethylsilyl group, and a triphenylsilyl group; and a silyloxy group, preferably having 3 to 40 carbon atoms, more preferably 3 to 30 carbon atoms, even more preferably 3 to 24 carbon atoms, such as a trimethylsilyloxy group, and a triphenylsilyloxy group. These substituents may contain another substituent therein.
- Specific examples of the platinum complex compound containing the tetradentate ligand expressed by the
general formula 1 include the following compounds, but the examples are not limited to the followings. - The amount of the platinum complex compound containing the tetradentade ligand, expressed by the
general formula 1, is preferably 5% by mass to 99.5% by mass, more preferably 10% by mass to 99.5% by mass, even more preferably 10% by mass to 50% by mass, relative to the total amount of all the compounds contained in the light emitting layer. - When the amount thereof is less than 5% by mass, the effect of voltage reduction may decline.
- The hole-transporting host material may be suitably selected depending on the intended purpose without any restriction. Examples thereof include pyrrole, indole, carbazole, azaindole, azacarbazole, pyrazole, imidazole, polyaryl alkane, pyrazoline, pyrazolone, phenylenediamine, arylamine, amino-substituted chalcone, styrylanthracene, fluorenone, hydrazone, stilbene, silazane, atomatic tertiary amine compound, styrylamine compound, aromatic dimethylidine compound, porphyrin compound, polysilane compound, poly(N-vinyl carbazole), aniline copolymer, high-molecular weight conductive oligomer, such as thiophene oilomer and polythiophene, organic silane, carbon film, and derivatives thereof.
- Among them, indole derivatives, carbazole derivatives, azaindole derivatives, azacarbazole derivatives, aromatic tertiary amine compounds and thiophene derivatives are preferable, and those containing a plurality of insole structures, carbazole structures, azaindole structures, azacarbazole structures, or atomatic tertiary amine structures per molecule are particularly preferable.
- Moreover, as the host material for use in the present invention, a host material in which part of or all of the hydrogen atoms contained therein are substituted with deuterium may be used (see JP-A Nos. 2009-277790, and 2004-515506).
- Specific examples of such hole-transporting host material include the following compounds, but not limited thereto.
- The amount of the hole-transporting host material is preferably 10% by mass to 99% by mass, more preferably 10% by mass to 90% by mass, even more preferably 20% by mass to 90% by mass, relative to the total amount of all the compounds contained in the light-emitting layer.
- As the phosphorescent light-emitting material, a complex containing a transition metal atom or a lanthanoid atom is generally used. Examples of the transition metal include ruthenium, rhodium, palladium, tungsten, rhenium, osmium, iridium, and platinum. Among them, rhenium, iridium, and platinum are preferable, iridium and platinum being more preferable.
- Examples of the lanthanoid atom include lanthanum, cerium, praseodymium, neodymium, samarium, europium, gadolinium, teibium, dysprosium, holmium, erbium, thulium, ytterbium, and lutetecium. Among them, neodymium, europium, and gadolinium are particularly preferable.
- Examples of the ligand of the complex include ligands disclosed in G. Wilkinson et al., Comprehensive Coordination Chemistry, Pergamon Press, 1987; H. Yersin, Photochemistry and Photophysics of Coordination Compounds, Springer-Verlag, 1987; and Akio Yamamoto, Organic Metal Chemistry—Foundation and Application, Shokado Publishing Co., Ltd., 1982.
- Specific example of the ligand include: a halogen ligand, preferably a chlorine ligand; a aromatic carbon ring ligand, such as a cyclopentadienyl anion, a benzene anion, and a naphthyl anion; a nitrogen-containing heterocyclic ligand, such as a phenyl pyridine, a benzoquinoline, a quinolinol, bipyridyl, and a phenanthroline; a diketone ligand, such as a acetyl acetone; a carboxylic acid ligand, such as an acetic acid ligand; an alkolate ligand, such as a phenolate ligand; a carbon monoxide ligand; an isonitrile ligand; and a cyano ligand. Among them, the nitrogen-containing heterocyclic ligand is preferable.
- The complex may contain one transition metal in the compound. Alternatively, the complex may be a binuclear complex containing two or more transition metals. In this case, different types of the metal atoms may be contained at the same time. Among them, the following are listed as specific examples of the light-emitting material, but the examples of the light-emitting material are not limited to the following.
- The phosphorescent light-emitting material may be suitably selected depending on the intended purpose without any restriction, but is preferably a compound expressed by any one of the following
general formulae 2 to 4: - In the
general formulae 2 to 4, n is an integer of 1 to 3; X-Y represents a bidentate ligand; a ring A is a ring structure which may contain at least one selected from the group consisting of a nitrogen atom, a sulfur atom, and an oxygen atom; R11 is a substituent, m1 is an integer of 0 to 6, and in the case where m1 is 2 or more, a plurality of R11s adjacent to each other may bond to form a ring, which may contain at least one selected from the group consisting of a nitrogen atom, a sulfur atom, and an oxygen atom, and may further contain one or more substituents; R12 is a substituent, m2 is an integer of 0 to 4, and in the case where m2 is 2 or more, a plurality of R12s adjacent to each other may bond to form a ring, which may contain at least one selected from the group consisting of a nitrogen atom, a sulfur atom, and an oxygen atom, the ring may contain a substituent; R11 and R12 may bond to each other to form a ring, which may contain at least one selected from the group consisting of a nitrogen atom, a sulfur atom, and an oxygen atom, and may further contain one or more substituents. - The ring A denotes a ring structure, which may contain at least one selected from the group consisting of a nitrogen atom, a sulfur atom, and an oxygen atom, and is preferably a five-member ring, a six-member ring, or the like. The ring may contain one or more substituents.
- X-Y denotes a bidentate ligand, and preferable examples thereof include bidentate monoanion ligands, and the like.
- Examples of the bidentate monoanion ligand include picolinate (pic), acetylacetonate (acac), and dipicaloylmethanate (t-butyl acac).
- Examples of the ligand include, other than listed above, ligands disclosed in International Publication No. WO 02/15645 (Lamansky et al., pp. 89-91).
- Substituents denoted as R11 and R12 may be suitably selected depending on the intended purpose without any restriction. Examples thereof include halogen atoms, alkoxy groups, amino groups, alkyl groups, cycloalkyl groups, aryl groups which may contain a nitrogen atom or sulfur atom, and aryloxy groups which may contain a nitrogen atom or sulfur atom. These substituents may further contain one or more substituents therein.
- Adjacent substituents denoted as R11 and R12 may bond to each other to form a ring that may contain a nitrogen atom, a sulfur atom, or an oxygen atom, and suitable examples of such the ring include a five-member ring and a six-member ring. In addition, such the ring may further contain one or more substituents.
- Specific examples of the compound expressed by any of the
general formulae 2 to 4 include the following, but are not limited thereto. - In general, the amount of phosphorescent light-emitting material is preferably 0.5% by mass to 30% by mass, more preferably 0.5% by mass to 20% by mass, more preferably 3% by mass to 10% by mass relative to the total amount of all the compounds contained in the light-emitting layer.
- When the amount thereof is less than 0.5% by mass, the emission efficiency may be undesirably low. When the amount thereof is more than 30% by mass, the emission efficiency may be undesirably low due to the aggregations between the phosphorescent light-emitting materials.
- The light-emitting layer has the function of receiving holes from the anode, the hole-injection layer, or the hole-transport layer, receiving electrons from the cathode, the electron-injection layer, or the electron-transport layer, and providing a field for recombination of the holes with the electrons for light emission, when an electric field is applied.
- The light-emitting layer can be formed by known methods in the art, without any restriction. Preferable examples of the forming method thereof include: dry film-forming methods such as vapor deposition, and sputtering; wet coating; transferring; printing; and inkjet printing.
- The thickness of the light-emitting layer may be suitably adjusted depending on the intended purpose without any restriction. The thickness thereof is preferably 2 nm to 500 nm, more preferably 3 nm to 200 nm, even more preferably 10 nm to 200 nm, in view of the emission efficiency. Moreover, the light-emitting layer may be formed of a single layer, or, two or more layers.
- The organic electroluminescence element contains an organic layer including the light-emitting layer between an anode and a cathode, and may further contain other layers, if necessary.
- The organic layer contains at least the light-emitting layer, an electron-transport layer, and an electron-injection layer, and optionally further contains a hole-injection layer, a hole-transport layer, a hole-blocking layer, an electron-blocking layer, and the like.
- The electron-injection layer and the electron-transport layer both have a function of receiving electrons from the anode side and transport to the cathode side. The electron-injection layer and the electron-transport layer may be of a monolayer structure, or a laminate structure containing a plurality of layers each formed of identical or different compositions.
- The electron-injection layer and the electron-transport layer, respectively, may be suitably selected depending on the intended purpose without any restriction. Examples of the material for forming the electron-injection layer and/or the electron-transport layer include: triazole derivative; oxazole derivative; oxadiazole derivative; fluorenone derivative; anthraquinodimehane derivative; anthrone derivative; diphenylquinone derivative; thiopyrandioxide derivative; carbodiimide derivative; fluorenylidenemethane derivative; distyrylpyradine derivative; heterocyclic carboxylic acid anhydride such as naphthalene and perylene; phthalocyanine derivative; metal complex such as 8-quinolinol derivative; metal phthalocyanine; and metal complex containing benzoxazole or benzothiazole as a ligand.
- The electron-injection layer and the electron-transport layer may contain a hole-accepting dopant, respectively.
- The hole-accepting dopant may be an inorganic compound or an organic compound, provided that it accepts holes, and has a function of reducing organic compounds.
- The inorganic compound may be suitably selected depending on the intended purpose without any restriction. Examples thereof include alkali metals, alkaline earth metals, and metal oxides thereof.
- The electron-injection layer and the electron-transport layer each preferably have a thickness of 1 nm to 5 μm, more preferably 5 nm to 1 μm, even more preferably 10 nm to 500 nm.
- The hole-injection layer and the hole-transport layer each have a function of receiving holes from the anode or the anode side and transporting to the cathode side. The hole-injection layer and the hole-transport layer may be of a monolayer structure, or a laminate structure containing a plurality of layers each formed of identical or different compositions.
- The hole-injection material or hole-transport material used for these layers may be a low-molecular-weight compound or a high-molecular-weight compound.
- The hole-injection material or the hole-transport material may be suitably selected depending on the intended purpose without any restriction. Examples thereof include pyrrole derivatives, carbazole derivatives, triazole derivatives, oxazole derivatives, oxadiazole derivatives, imidazole derivatives, polyarylalkane derivatives, pyrazoline derivatives, pyrazolone derivatives, phenylenediamine derivatives, arylamine derivatives, amino-substituted chalcone derivatives, styrylanthracene derivatives, fluorenone derivatives, hydrazone derivatives, stilbene derivatives, silazane derivatives, aromatic tertiary amine compounds, styrylamine compounds, aromatic dimethylidine compounds, phthalocyanine compounds, porphyrin compounds, thiophene derivatives, organosilane derivatives and carbon. These may be used independently or in combination.
- The hole-injection layer and the hole-transport layer, respectively, may contain an electron-accepting dopant.
- The electron-accepting dopant may be an inorganic compound or an organic compound, provided that it accepts electrons, and has a function of oxidizing organic compounds.
- The inorganic compound is suitably selected depending on the intended purpose without any restriction. Examples thereof include: metal halides, such as ferric chloride, aluminum chloride, gallium chloride, indium chloride and antimony pentachloride; and metal oxides such as vanadium pentaoxide and molybdenum trioxide.
- The organic compound may be suitably selected depending on the intended purpose without any restriction. Examples thereof include: those having a substituent such as a nitro group, a halogen atom, a cyano group and a trifluoromethyl group; quinone compounds; acid anhydride compounds; and fullerenes.
- These electron-accepting dopants may be used independently, or in combination.
- The amount of the electron-accepting dopant is not restricted, though it may vary depending on the types of the materials thereof. The amount thereof is preferably 0.01% by mass to 50% by mass, more preferably 0.05% by mass to 30% by mass, even more preferably 0.1% by mass to 30% by mass relative to the amount of the hole-transport material or hole-injection material.
- The hole-injection layer and the hole-transport layer may be formed by known methods in the art, without any restriction. Preferable examples of forming methods thereof include: dry film-forming methods such as vapor deposition and sputtering; wet film-forming methods; transferring; printing; and ink-jet printing.
- The hole-injection layer and the hole-transport layer preferably each have a thickness of 1 nm to 500 nm, more preferably 5 nm to 250 nm, yet more preferably 10 nm to 200 nm.
- The hole-blocking layer has a function of preventing holes transported from the anode side to the light-emitting layer from passing through to the cathode side, and is generally provided as an organic layer adjacent to the light-emitting layer on the cathode side.
- The electron-blocking layer has the function to prevent the electrons transported from the cathode side to the light-emitting layer from passing through to the anode side, and is generally provided as an organic layer adjacent to the light-emitting layer on the anode side.
- Examples of the compound for forming the hole-blocking layer include aluminum complexes such as BAlq; triazole derivatives; and phenanthroline derivatives such as BCP.
- Examples of the compound for forming the electron-blocking layer are those listed above as the hole-transport material.
- The electron-blocking layer and the hole-blocking layer may be formed by known methods in the art, without any restriction. Preferable examples of forming methods thereof include: dry film-forming methods such as vapor deposition and sputtering; wet film-forming methods; transferring; printing; and ink-jet printing.
- The hole-blocking layer and the electron-blocking layer each preferably have a thickness of 1 nm to 200 nm, more preferably 1 nm to 50 nm, yet more preferably 3 nm to 10 nm. The hole-blocking layer and the electron-blocking layer may be of a monolayer structure forming of one or more materials mentioned above, or of a laminate structure having a plurality of layers each formed of identical or different compositions.
- The organic electroluminescence element contains a pair of electrodes; i.e., an anode and a cathode. In consideration of the characteristics of the organic electroluminescence element, at least one of the anode and the cathode is preferably transparent. In general, the anode may serve as an electrode which supplies holes to an organic layer, and the cathode may serve as an electrode which injects electrons into an organic layer.
- In terms of the shape, structure, size and the like, the electrode may be suitably selected from the electrode materials known in the art depending on the use of the organic electroluminescence element, without any restriction.
- Suitably examples of the material for forming the electrode include metals, alloys, metal oxides, conductive compounds, and mixtures thereof.
- Examples of the material for forming the anode include; conductive metal oxides such as tin oxides doped with, for example, antimony and fluorine (ATO and FTO); tin oxide, zinc oxide, indium oxide, indium tin oxide (ITO) and indium zinc oxide (IZO); metals such as gold, silver, chromium and nickel; mixtures or laminates of these metals and the conductive metal oxides; inorganic conductive materials such as copper iodide and copper sulfide; organic conductive materials such as polyaniline, polythiophene and polypyrrole; and laminates of these materials and ITO. Among them, conductive metal oxides are preferable. In particular, ITO is preferable for its productivity, high conductivity, and transparency.
- Examples of the material for forming the cathode include alkali metals (e.g., Li, Na, K and Cs), alkaline earth metals (e.g., Mg and Ca), gold, silver, lead, aluminum, sodium-potassium alloys, lithium-aluminum alloys, magnesium-silver alloys and rare earth metals (e.g., indium and ytterbium). These may be used independently, but preferably in combination for the purpose of favorable stability and electron-injection properties.
- Among them, alkali metals and alkaline earth metals are preferable in terms of the electron-injection properties, and the material containing aluminum as a main component is preferable in terms of excellent storage stability.
- The phrase “material containing aluminum as a main component” refers to a material composed of aluminum alone; alloys containing aluminum and 0.01% by mass to 10% by mass of an alkali or alkaline earth metal; or the mixtures thereof (e.g., lithium-aluminum alloys and magnesium-aluminum alloys).
- The electrode can be formed by known methods in the art, without any restriction. Examples of forming methods of the electrode include: wet methods such as printing and coating; physical methods such as vacuum deposition, sputtering, and ion plating; and chemical methods such as CVD, and plasma CDV. According to a method appropriately selected from these methods in consideration of suitability for the material constituting the anode, the anode can be formed on a substrate. For example, when ITO is used as the material for the anode, the anode may be formed with DC or high-frequency sputtering methods, vacuum deposition methods, or ion plating methods. Notably, when metals are used as a material for the below-described cathode, the cathode can be formed by, for example, sputtering one type of metal, or, two or more types of metals simultaneously or sequentially.
- Patterning for forming the anode may be performed by a chemical etching method such as photolithography; a physical etching method such as etching by laser; a method of vacuum deposition or sputtering using a mask; a lift-off method; or a printing method.
- The organic electroluminescence element is preferably provided on a substrate. The organic electroluminescence element may be provided so that the electrode thereof is in direct contact with the substrate, or so that the electrode is in contact with the substrate via an intermediate layer.
- The material for the substrate may be suitably selected depending on the intended purpose without any restriction. Examples thereof include inorganic materials such as yttria-stabilized zirconia (YSZ) and glass (alkali-free glass and soda-lime glass); and organic materials such as polyesters (e.g., polyethylene terephthalate, polybutylene phthalate and polyethylene naphthalate), polystyrene, polycarbonate, polyether sulfone, polyarylate, polyimide, polycycloolefin, norbornene resins and poly(chlorotrifluoroethylene).
- In terms of the shape, structure, size and the like, the substrate may be suitably adjusted depending on the use, purpose, and the like of the electroluminescence element, without any restriction. In general, it is preferable to provide the substrate in the form of a sheet. The substrate may have a single- or multi-layered structure, and may be a single member or a combination of two or more members. The substrate may be opaque, colorless transparent, or colored transparent.
- The substrate may be provided with a moisture permeation-preventing layer (gas barrier layer) on the front and/or back surface thereof.
- The moisture permeation-preventing layer (gas barrier layer) is preferably made from an inorganic compound such as silicon nitride and silicon oxide.
- The moisture permeation-preventing layer (gas barrier layer) can be formed through, for example, high-frequency sputtering.
- The entire organic electroluminescence element may be protected with a protective layer.
- The materials contained in the protective layer may be suitably selected depending on the intended purpose without any restriction, provided that the resulted protective layer will have the function to prevent water, oxygen, and the like, which promote the degradation of the element, from entering into the element. Examples thereof include: metals such as In, Sn, Pb, Au, Cu, Ag, Al, Ti and Ni; metal oxides such as MgO, SiO, SiO2, Al2O3, GeO, NiO, CaO, BaO, Fe2O3, Y2O3 and TiO2; metal nitrides such as SiNx and SiNxOy; metal fluorides such as MgF2, LiF, AlF3 and CaF2; polyethylenes, polypropylenes, polymethyl methacrylates, polyimides, polyureas, polytetrafluoroethylenes, polychlorotrifluoroethylens, polydichlorodifluoroethylenes, copolymers of chlorotrifluoroethylens and dichlorodifluoroethylenes, copolymers produced through compolymerization of a monomer mixture containing tetrafluoroethylene and at least one comonomer, fluorine-containing copolymers containing a ring structure in the copolymerization main chain, water-absorbing materials each having a water absorption rate of 1% or more, and moisture permeation preventive substances each having a water absorption rate of 0.1% or less.
- The formation method of the protective layer is suitably selected depending on the intended purpose without any restriction. Examples thereof include vacuum deposition, sputtering, reactive sputtering, molecular beam epitaxial (MBE), cluster ion beam, ion plating, plasma polymerization (high-frequency excitation ion plating), plasma CVD, laser CVD, thermal CVD, gas source CVD, coating, printing and transferring.
- The organic electroluminescence element may be entirely sealed with a seal container. Further, a moisture absorbent or an inert liquid may be contained in the space between the seal container and the organic electroluminescence element.
- The moisture absorbent is not particularly limited and may be appropriately selected depending on the purpose. Examples thereof include barium oxide, sodium oxide, potassium oxide, calcium oxide, sodium sulfate, calcium sulfate, magnesium sulfate, phosphorus pentaoxide, calcium chloride, magnesium chloride, copper chloride, cesium fluoride, niobium fluoride, calcium bromide, vanadium bromide, molecular sieve, zeolite and magnesium oxide.
- The inert liquid is not particularly limited and may be appropriately selected depending on the purpose. Examples thereof include paraffins; liquid paraffins; fluorine-containing solvents such as perfluoroalkanes, perfluoroamines and perfluoroethers; chlorinated solvents; and silicone oils.
- The organic electroluminescence element is preferably sealed with a resin seal layer so as to prevent degradation thereof due to oxygen and/or moisture contained in the air.
- The resin material for the resin seal layer may be suitably selected depending on the intended purpose without any restriction. Examples thereof include acrylic resins, epoxy resins, fluorine-containing resins, silicone resins, rubber resins and ester resins. Among them, epoxy resins are preferred from the standpoint of its excellent properties in water impermeability. Among the epoxy resins, thermosetting epoxy resins and photo-curable epoxy resins are preferred.
- The forming method for the resin seal layer is not particularly limited and may be appropriately selected depending on the purpose. Examples thereof include a method by coating a resin solution, a method by press-bonding or hot press-bonding a resin sheet, and a method by polymerizing under dry conditions (e.g., vapor deposition and sputtering).
- The organic electroluminescence element contains a sealing adhesive having the function of preventing permeation of moisture or oxygen from the edges thereof.
- The material for the sealing adhesive may be those used in the resin seal layer. Among them, epoxy resins are preferred from the viewpoint of preventing water permeation, with photo-curable epoxy resins and thermosetting epoxy resins being more preferred.
- Also, a filler is preferably added to the sealing adhesive. The filler is preferably inorganic materials such as SiO2, SiO (silicon oxide), SiON (silicon oxynitride) and SiN (silicon nitride). The filler increases the viscosity of the sealing adhesive to improve processability and humidity resistance.
- The sealing adhesive may also contain a desiccant. Examples of the desiccant include barium oxide, calcium oxide or strontium oxide. The amount of the desiccant added to the sealing adhesive is preferably 0.01% by mass to 20% by mass, more preferably 0.05% by mass to 15% by mass. When the amount is less than 0.01% by mass, the desiccant exhibits reduced effects. Whereas when the amount is more than 20% by mass, it may be difficult to homogeneously disperse the desiccant in the sealing adhesive.
- In the present invention, the sealing adhesive containing the desiccant is applied in a predetermined amount using, for example, a dispenser. Thereafter, a second substrate is overlaid, followed by curing for sealing.
-
FIG. 1 is a schematic diagram showing one example of the layer structure of the organic electroluminescence element of the invention. Theorganic EL element 10 has a layer structure in which an anode 2 (e.g., ITO electrode) formed on aglass substrate 1, a hole-injection layer 3, a hole-transport layer 4, a light-emitting layer 5, an electron-transport layer 6, an electron-injection layer 7, and a cathode 8 (e.g., Al—Li electrode) are laminated in this order. Note that, the anode 2 (e.g., ITO electrode) and the cathode 8 (e.g., Al—Li electrode) are connected to each other via a power source. - The organic electroluminescence element can emit light when a DC voltage (which, if necessary, may contain AC components) (generally 2 volts to 15 volts) or a DC is applied to between the anode and the cathode.
- The organic electroluminescence element can be applied to an active matrix by a thin film transistor (TFT). An active layer of the thin film transistor can be formed from, for example, amorphous silicone, high-temperature polysilicone, low-temperature polysilicone, microcrystalline silicone, oxide semiconductor, organic semiconductor or carbon nanotube.
- Examples of the thin film transistor applicable in the organic electroluminescence element include those described in, for example, WO2005/088726, JP-A No. 2006-165529, and U.S. Pat. Application Publication No. 2008/0237598 A1.
- The light-extraction efficiency of the organic electroluminescence element of the present invention can be improved by various known methods, without any restriction. It is possible to increase the light-extraction efficiency to improve the external quantum efficiency, for example, by processing the surface shape of the substrate (for example, by forming a fine concavo-convex pattern), by controlling the refractive index of the substrate, the ITO layer and/or the organic layer, or by controlling the thickness of the substrate, the ITO layer and/or the organic layer.
- The organic electroluminescence element may be used in a top-emission configuration or a bottom-emission configuration, in order for light to be extracted.
- The organic electroluminescence element may have a resonator structure. For example, on a transparent substrate are stacked a multi-layered film mirror composed of a plurality of laminated films having different reflective indices, a transparent or semi-transparent electrode, a light-emitting layer and a metal electrode. The light generated in the light-emitting layer is repeatedly reflected between the multi-layered film mirror and the metal electrode (which serve as reflection plates). Therefore, in this matter the light is resonated.
- In another preferred embodiment, a transparent or semi-transparent electrode and a metal electrode are stacked on a transparent substrate. In this structure, the light generated in the light-emitting layer is repeatedly reflected between the transparent or semi-transparent electrode and the metal electrode (which serve as reflection plates); i.e., is resonated.
- For forming the resonance structure, an optical path length determined based on the effective refractive index of two reflection plates, and on the refractive index and the thickness of each of the layers between the reflection plates is adjusted to be an optimal value for obtaining a desired resonance wavelength. The calculation formula applied in the case of the first embodiment is described in JP-A No. 09-180883. The calculation formula in the case of the second embodiment is described in JP-A No. 2004-127795.
- The use of the organic electroluminescence element of the present invention is suitably selected depending on the intended purpose without any restriction. For example, the organic electroluminescence element of the present invention can be suitable used in display elements, displays, backlights, electrophotography, illuminating light sources, recording light sources, exposing light sources, reading light sources, markers, signs, interior accessories and optical communication.
- As methods for forming a full color-type organic EL display, there are known, for example (as described in “Monthly Display,” September 2000, pp. 33 to 37), a tricolor light emission method by arranging, on a substrate, organic EL elements emitting lights corresponding to three primary colors (blue color (B), green color (G) and red color (R)); a white color method by separating white light emitted from an organic electroluminescence element for white color emission into three primary colors through a color filter; and a color conversion method by converting a blue light emitted from an organic electroluminescence element for blue light emission into red color (R) and green color (G) through a fluorescent dye layer.
- Examples of the present invention will be explained hereinafter, but these examples shall not be construed as to limit the scope of the present invention.
- A glass substrate having a thickness of 0.5 mm, and a size of 2.5 cm×2.5 cm was placed in a washing container, subjected to ultrasonic washing in 2-propanol, followed by subjected to a UV-ozone treatment for 30 minutes. Onto this glass substrate, the following layers were deposited by vacuum deposition. Note that, in Examples and Comparative Examples, the deposition rate was 0.2 nm/sec., unless otherwise stated. The deposition rate was measured by a crystal resonator. In addition, the thickness of each layer described below was also measured by a crystal resonator.
- At first, on the glass substrate, Indium Tin Oxide (ITO) was deposited in the thickness of 100 nm by sputtering as an anode.
- Then, on the anode (ITO), α-NPD (bis[N-(1-naphthyl)-N-pheny]benzidine) was deposited in the thickness of 40 nm by vapor deposition, as a hole-transport layer.
- On the hole-transport layer, a light-emitting layer was deposited in the thickness of 30 nm by vapor deposition. The light-emitting layer was formed of the platinum complex A expressed by the following structural formula, which served as a
host material 1, and the compound (I-15) expressed by the following structural formula, which served as a phosphorescent light-emitting material and provided in an amount of 5% by mass relative to the amount of thehost material 1. - On the light-emitting layer, BAlq (bis-(2-methyl-8-quinolinolato)-4-(phenyl-phenolate)-aluminium (III)) was deposited in the thickness of 55 nm by vapor deposition as an electron-transport layer.
- Then, on the electron-transport layer, LiF was deposited in the thickness of 1 nm by vapor deposition, as an electron injection layer.
- Next, a patterned mask (the mask to give a pattern having a light-emitting region of 2 mm×2 mm) was placed on the electron injection layer, and aluminum was deposited thereon in the thickness of 100 nm by vapor deposition as a cathode.
- The obtained laminate was placed in a glove compartment in which the atmosphere had been replaced with argon gas, and was sealed by using a sealing tin formed of stainless steel and a UV-curable adhesive (XNR5516HV, manufactured by Nagase ChemteX Corporation). In the manner as described above, the organic electroluminescence element of Comparative Example 1 was prepared.
- The organic electroluminescence element of Comparative Example 2 was prepared in the same manner as Comparative Example 1, provided that the platinum complex A used as the
host material 1 in the light-emitting layer was replaced with the platinum complex B expressed by the following structural formula. - The organic electroluminescence layer of Example 1 was prepared in the same manner as in Comparative Example 1, provided that the platinum complex A used as the
host material 1 in the light-emitting layer was replaced with the platinum complex E expressed by the following structural formula. - The organic electroluminescence layer of Example 2 was prepared in the same manner as in Comparative Example 1, provided that the platinum complex A used as the
host material 1 in the light-emitting layer was replaced with the platinum complex F expressed by the following structural formula. - The organic electroluminescence layer of Example 3 was prepared in the same manner as in Comparative Example 1, provided that the platinum complex A used as the
host material 1 in the light-emitting layer was replaced with the platinum complex J expressed by the following structural formula. - The organic electroluminescence layer of Example 4 was prepared in the same manner as in Comparative Example 1, provided that the platinum complex A used as the
host material 1 in the light-emitting layer was replaced with the platinum complex K expressed by the following structural formula. - The organic electroluminescence element of Comparative Example 3 was prepared in the same manner as Comparative Example 1, provided that the light-emitting layer was replaced with a light-emitting layer containing 20% by mass of BAlq as a
host material 1, 75% by mass of the compound (H-24) expressed by the following structural formula as ahost material 2, and 5% by mass of the compound (I-15) expressed by the structural formula presented earlier as the phosphorescent light-emitting material. - The organic electroluminescence element of Comparative Example 4 was prepared in the same manner as Comparative Example 1, provided that the light-emitting layer was replaced with a light-emitting layer containing 20% by mass of the aforementioned platinum complex A as a
host material 1, 75% by mass of the compound (H-24) expressed by the structural formula presented earlier as ahost material 2, and 5% by mass of the compound (I-15) expressed by the structural formula presented earlier as the phosphorescent light-emitting material. - The organic electroluminescence element of Example 5 was prepared in the same manner as Comparative Example 1, provided that the light-emitting layer was replaced with alight-emitting layer containing 20% by mass of the aforementioned platinum complex E as a
host material 1, 75% by mass of the compound (H-24) expressed by the structural formula presented earlier as ahost material 2, and 5% by mass of the compound (I-15) expressed by the structural formula presented earlier as the phosphorescent light-emitting material. - The organic electroluminescence element of Example 6 was prepared in the same manner as Comparative Example 1, provided that the light-emitting layer was replaced with a light-emitting layer containing 20% by mass of the aforementioned platinum complex F as a
host material 1, 75% by mass of the compound (H-24) expressed by the structural formula presented earlier as ahost material 2, and 5% by mass of the compound (I-15) expressed by the structural formula presented earlier as the phosphorescent light-emitting material. - The prepared organic electroluminescence elements of Examples 1 to 6 and Comparative Examples 1 to 4 were respectively subjected to the measurements of the driving voltage, external quantum efficiency, and peak wavelength. The results are shown in Tables 1-1 and 1-2.
- A direct voltage was applied to the organic electroluminescence element by a source measure unit 2400 (manufactured by Keithley Instruments Inc.) to allow the organic electroluminescence element to emit, and the voltage at which the current density was 2.5 mA/cm2 was measured.
- A direct voltage was applied to the organic electroluminescence element by a source measure unit 2400, manufactured by Keithley Instruments Inc. to allow the organic electroluminescence element to emit. The luminance of the emission was measured by a luminance meter (BM-8, manufactured by Topcon Corporation). The emission spectrum and the emission wavelength were measured by a spectrum analyzer PMA-11, manufactured by Hamamatsu Photonics K.K. Based on these values, the emission efficiency at the current density of 2.5 mA/cm2 was calculated as external quantum efficiency in accordance with the luminance conversion method.
- The peak wavelength was determined from the spectrum obtained by a spectrum analyzer PMA-11, manufactured by Hamamatsu Photonics K.K.
-
TABLE 1-1 Phosphorescent External light-emitting Driving voltage quantum Peak Host material material [V] efficiency [%] wavelength (% by mass) (% by mass) (at 2.5 mA/cm2) (at 2.5 mA/cm2) (nm) Comp. Platinum I-15 (5) 7.2 7.6 595 Ex. 1 complex A (95) Comp. Platinum I-15 (5) 7.5 6.5 594 Ex. 2 complex B (95) Ex. 1 Platinum I-15 (5) 6.0 9.6 596 complex E (95) Ex. 2 Platinum I-15 (5) 6.2 9.4 595 complex F (95) Ex. 3 Platinum I-15 (5) 6.4 9.8 594 complex J (95) Ex. 4 Platinum I-15 (5) 6.8 9.2 596 complex K (95) -
TABLE 1-2 Phosphorescent Driving External Host Host light-emitting voltage quantum Peak material 1 material 2material [V] efficiency [%] wavelength (% by mass) (% by mass) (% by mass) (at 2.5 mA/cm2) (at 2.5 mA/cm2) (nm) Comp. BAlq (20) H-24 (75) I-15 (5) 8.5 10.5 595 Ex. 3 Comp. Platinum H-24 (75) I-15 (5) 7.5 12.1 595 Ex. 4 complex A (20) Ex. 5 Platinum H-24 (75) I-15 (5) 6.5 13.5 596 complex E (20) Ex. 6 Platinum H-24 (75) I-15 (5) 6.7 13.2 595 complex F (20) - Since the organic electroluminescence element of the present invention can realize both reduction in voltage for use and high efficiency, it can be suitably applied in display elements, displays, backlights, electrophotography, illuminating light sources, recording light sources, exposing light sources, reading light sources, markers, signs, interior accessories and optical communication.
Claims (4)
1. An organic electroluminescence element, comprising:
an anode;
a cathode; and
at least one organic layer disposed between the cathode and the anode, the organic layer comprising a light-emitting layer,
wherein the light-emitting layer contains a host material and a phosphorescent light-emitting material, and the host material contains at least one platinum complex compound containing a tetradentade ligand, expressed by the following general formula 1:
2. The organic electroluminescence element according to claim 1 , wherein the organic electroluminescence element exhibits a luminescence peak at 550 nm or more.
3. The organic electroluminescence element according to claim 1 , wherein the host material contains at least one hole-transporting host material.
4. The organic electroluminescence element according to claim 1 , wherein the phosphorescent light-emitting material is a compound expressed by any of the following general formulae 2 to 4:
where n is an integer of 1 to 3; X-Y represents a bidentate ligand; a ring A is a ring structure which may contain at least one selected from the group consisting of a nitrogen atom, a sulfur atom, and an oxygen atom; R11 is a substituent, m1 is an integer of 0 to 6, and in the case where m1 is 2 or more, a plurality of R11s adjacent to each other may bond to form a ring, which may contain at least one selected from the group consisting of a nitrogen atom, a sulfur atom, and an oxygen atom, and may have further one or more substituents; R12 is a substituent, m2 is an integer of 0 to 4, and in the case where m2 is 2 or more, a plurality of R12s adjacent to each other may bond to form a ring, which may contain at least one selected from the group consisting of a nitrogen atom, a sulfur atom, and an oxygen atom, and may further contain one or more substituents; R11 and R12 may bond to each other to form a ring, which may contain at least one selected from the group consisting of a nitrogen atom, a sulfur atom, and an oxygen atom, and may further contain one or more substituents.
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2009-132033 | 2009-06-01 | ||
| JP2009132033 | 2009-06-01 | ||
| JP2010-098624 | 2010-04-22 | ||
| JP2010098624A JP5456554B2 (en) | 2009-06-01 | 2010-04-22 | Organic electroluminescence device |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| US20100301315A1 true US20100301315A1 (en) | 2010-12-02 |
Family
ID=43219204
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/780,581 Abandoned US20100301315A1 (en) | 2009-06-01 | 2010-05-14 | Organic electroluminescence element |
Country Status (2)
| Country | Link |
|---|---|
| US (1) | US20100301315A1 (en) |
| JP (1) | JP5456554B2 (en) |
Cited By (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB2508191A (en) * | 2012-11-23 | 2014-05-28 | Cambridge Display Tech Ltd | Organic light emissive device |
| US20180208615A1 (en) * | 2017-01-20 | 2018-07-26 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US20210193936A1 (en) * | 2017-10-17 | 2021-06-24 | Arizona Board Of Regents On Behalf Of Arizona State University | Phosphorescent excimers with preferred molecular orientation as monochromatic emitters for display and lighting applications |
| US11647643B2 (en) | 2017-10-17 | 2023-05-09 | Arizona Board Of Regents On Behalf Of Arizona State University | Hole-blocking materials for organic light emitting diodes |
| US11653560B2 (en) | 2014-11-10 | 2023-05-16 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate metal complexes with carbon group bridging ligands |
| US11708385B2 (en) | 2017-01-27 | 2023-07-25 | Arizona Board Of Regents On Behalf Of Arizona State University | Metal-assisted delayed fluorescent emitters employing pyrido-pyrrolo-acridine and analogues |
| US11785838B2 (en) | 2019-10-02 | 2023-10-10 | Arizona Board Of Regents On Behalf Of Arizona State University | Green and red organic light-emitting diodes employing excimer emitters |
| US11839144B2 (en) | 2014-06-02 | 2023-12-05 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate cyclometalated platinum complexes containing 9,10-dihydroacridine and its analogues |
| US11878988B2 (en) | 2019-01-24 | 2024-01-23 | Arizona Board Of Regents On Behalf Of Arizona State University | Blue phosphorescent emitters employing functionalized imidazophenthridine and analogues |
| US11930698B2 (en) | 2014-01-07 | 2024-03-12 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate platinum and palladium complex emitters containing phenyl-pyrazole and its analogues |
| US11945985B2 (en) | 2020-05-19 | 2024-04-02 | Arizona Board Of Regents On Behalf Of Arizona State University | Metal assisted delayed fluorescent emitters for organic light-emitting diodes |
| US11974495B2 (en) | 2017-05-19 | 2024-04-30 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate platinum and palladium complexes based on biscarbazole and analogues |
| US12010908B2 (en) | 2017-05-19 | 2024-06-11 | Arizona Board Of Regents On Behalf Of Arizona State University | Metal-assisted delayed fluorescent emitters employing benzo-imidazo-phenanthridine and analogues |
| US12037348B2 (en) | 2018-03-09 | 2024-07-16 | Arizona Board Of Regents On Behalf Of Arizona State University | Blue and narrow band green and red emitting metal complexes |
| US12043611B2 (en) | 2014-08-15 | 2024-07-23 | Arizona Board Of Regents On Behalf Of Arizona State University | Non-platinum metal complexes for excimer based single dopant white organic light emitting diodes |
| US12043633B2 (en) | 2012-10-26 | 2024-07-23 | Arizona Board Of Regents On Behalf Of Arizona State University | Metal complexes, methods, and uses thereof |
| US12082486B2 (en) | 2014-07-24 | 2024-09-03 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate platinum (II) complexes cyclometalated with functionalized phenyl carbene ligands and their analogues |
| US12167676B2 (en) | 2013-10-14 | 2024-12-10 | Arizona Board Of Regents On Behalf Of Arizona State University | Platinum complexes and devices |
| US12232411B2 (en) | 2012-09-24 | 2025-02-18 | Arizona Board Of Regents On Behalf Of Arizona State University | Metal compounds, methods, and uses thereof |
| US12312366B2 (en) | 2015-06-03 | 2025-05-27 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate and octahedral metal complexes containing naphthyridinocarbazole and its analogues |
| US12503644B2 (en) | 2020-05-19 | 2025-12-23 | Arizona Board Of Regents On Behalf Of Arizona State University | Metal assisted delayed fluorescent emitters for organic light-emitting diodes |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20040127710A1 (en) * | 2002-12-28 | 2004-07-01 | Soo-Jin Park | Red luminescent compound and organic electroluminescent device using the same |
| US20090026936A1 (en) * | 2007-07-27 | 2009-01-29 | Tasuku Satou | Organic electroluminescence element |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2008108617A (en) * | 2006-10-26 | 2008-05-08 | Fujifilm Corp | Organic electroluminescence device |
| JP2010278390A (en) * | 2009-06-01 | 2010-12-09 | Fujifilm Corp | Organic electroluminescence device |
-
2010
- 2010-04-22 JP JP2010098624A patent/JP5456554B2/en active Active
- 2010-05-14 US US12/780,581 patent/US20100301315A1/en not_active Abandoned
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20040127710A1 (en) * | 2002-12-28 | 2004-07-01 | Soo-Jin Park | Red luminescent compound and organic electroluminescent device using the same |
| US20090026936A1 (en) * | 2007-07-27 | 2009-01-29 | Tasuku Satou | Organic electroluminescence element |
Cited By (31)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US12232411B2 (en) | 2012-09-24 | 2025-02-18 | Arizona Board Of Regents On Behalf Of Arizona State University | Metal compounds, methods, and uses thereof |
| US12043633B2 (en) | 2012-10-26 | 2024-07-23 | Arizona Board Of Regents On Behalf Of Arizona State University | Metal complexes, methods, and uses thereof |
| CN103840083A (en) * | 2012-11-23 | 2014-06-04 | 剑桥显示技术有限公司 | Organic light emissive device |
| GB2508191B (en) * | 2012-11-23 | 2015-10-07 | Cambridge Display Tech Ltd | Organic light emissive device |
| GB2508191A (en) * | 2012-11-23 | 2014-05-28 | Cambridge Display Tech Ltd | Organic light emissive device |
| US12167676B2 (en) | 2013-10-14 | 2024-12-10 | Arizona Board Of Regents On Behalf Of Arizona State University | Platinum complexes and devices |
| US11930698B2 (en) | 2014-01-07 | 2024-03-12 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate platinum and palladium complex emitters containing phenyl-pyrazole and its analogues |
| US11839144B2 (en) | 2014-06-02 | 2023-12-05 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate cyclometalated platinum complexes containing 9,10-dihydroacridine and its analogues |
| US12082486B2 (en) | 2014-07-24 | 2024-09-03 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate platinum (II) complexes cyclometalated with functionalized phenyl carbene ligands and their analogues |
| US12043611B2 (en) | 2014-08-15 | 2024-07-23 | Arizona Board Of Regents On Behalf Of Arizona State University | Non-platinum metal complexes for excimer based single dopant white organic light emitting diodes |
| US12302745B2 (en) | 2014-11-10 | 2025-05-13 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate metal complexes with carbon group bridging ligands |
| US11653560B2 (en) | 2014-11-10 | 2023-05-16 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate metal complexes with carbon group bridging ligands |
| US12312366B2 (en) | 2015-06-03 | 2025-05-27 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate and octahedral metal complexes containing naphthyridinocarbazole and its analogues |
| US11566034B2 (en) | 2017-01-20 | 2023-01-31 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11053268B2 (en) * | 2017-01-20 | 2021-07-06 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US20180208615A1 (en) * | 2017-01-20 | 2018-07-26 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11708385B2 (en) | 2017-01-27 | 2023-07-25 | Arizona Board Of Regents On Behalf Of Arizona State University | Metal-assisted delayed fluorescent emitters employing pyrido-pyrrolo-acridine and analogues |
| US11974495B2 (en) | 2017-05-19 | 2024-04-30 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate platinum and palladium complexes based on biscarbazole and analogues |
| US12010908B2 (en) | 2017-05-19 | 2024-06-11 | Arizona Board Of Regents On Behalf Of Arizona State University | Metal-assisted delayed fluorescent emitters employing benzo-imidazo-phenanthridine and analogues |
| US11594688B2 (en) * | 2017-10-17 | 2023-02-28 | Arizona Board Of Regents On Behalf Of Arizona State University | Display and lighting devices comprising phosphorescent excimers with preferred molecular orientation as monochromatic emitters |
| US12120945B2 (en) * | 2017-10-17 | 2024-10-15 | Arizona Board Of Regents On Behalf Of Arizona State University | Display and lighting devices comprising phosphorescent excimers with preferred molecular orientation as monochromatic emitters |
| US20230247897A1 (en) * | 2017-10-17 | 2023-08-03 | Arizona Board Of Regents On Behalf Of Arizona State University | Phosphorescent Excimers with Preferred Molecular Orientation as Monochromatic Emitters for Display And Lighting Applications |
| US11647643B2 (en) | 2017-10-17 | 2023-05-09 | Arizona Board Of Regents On Behalf Of Arizona State University | Hole-blocking materials for organic light emitting diodes |
| US20210193936A1 (en) * | 2017-10-17 | 2021-06-24 | Arizona Board Of Regents On Behalf Of Arizona State University | Phosphorescent excimers with preferred molecular orientation as monochromatic emitters for display and lighting applications |
| US12037348B2 (en) | 2018-03-09 | 2024-07-16 | Arizona Board Of Regents On Behalf Of Arizona State University | Blue and narrow band green and red emitting metal complexes |
| US11878988B2 (en) | 2019-01-24 | 2024-01-23 | Arizona Board Of Regents On Behalf Of Arizona State University | Blue phosphorescent emitters employing functionalized imidazophenthridine and analogues |
| US12448405B2 (en) | 2019-01-24 | 2025-10-21 | Arizona Board Of Regents On Behalf Of Arizona State University | Blue phosphorescent emitters employing functionalized imidazophenthridine and analogues |
| US12120946B2 (en) | 2019-10-02 | 2024-10-15 | Arizona Board Of Regents On Behalf Of Arizona State University | Green and red organic light-emitting diodes employing excimer emitters |
| US11785838B2 (en) | 2019-10-02 | 2023-10-10 | Arizona Board Of Regents On Behalf Of Arizona State University | Green and red organic light-emitting diodes employing excimer emitters |
| US11945985B2 (en) | 2020-05-19 | 2024-04-02 | Arizona Board Of Regents On Behalf Of Arizona State University | Metal assisted delayed fluorescent emitters for organic light-emitting diodes |
| US12503644B2 (en) | 2020-05-19 | 2025-12-23 | Arizona Board Of Regents On Behalf Of Arizona State University | Metal assisted delayed fluorescent emitters for organic light-emitting diodes |
Also Published As
| Publication number | Publication date |
|---|---|
| JP5456554B2 (en) | 2014-04-02 |
| JP2011014873A (en) | 2011-01-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20100301315A1 (en) | Organic electroluminescence element | |
| JP5210187B2 (en) | Organic electroluminescence device | |
| JP4620802B1 (en) | Organic electroluminescence device | |
| US20080241518A1 (en) | Organic electroluminescence element | |
| US20120068165A1 (en) | Organic electroluminescence element | |
| US9935275B2 (en) | Organic electroluminescence device and method for producing the same | |
| JP2009032990A (en) | Organic electroluminescence device | |
| WO2011021433A1 (en) | Organic electroluminescent element | |
| JP5497510B2 (en) | Organic electroluminescence device | |
| JP2007287652A (en) | Light emitting element | |
| JP5610848B2 (en) | Organic electroluminescence device | |
| JP2007221097A (en) | Organic electroluminescence device | |
| JP2010278390A (en) | Organic electroluminescence device | |
| JP2011171279A (en) | Organic electric field light-emitting element | |
| JP5761962B2 (en) | Organic electroluminescence device | |
| JP2011228569A (en) | Organic electroluminescence element | |
| JP4554721B1 (en) | Organic electroluminescence device and evaluation method thereof | |
| JP2007221113A (en) | Organic electroluminescence device | |
| JP5912224B2 (en) | White organic electroluminescence device | |
| JP5511454B2 (en) | Organic electroluminescence device | |
| JP5833614B2 (en) | Organic electroluminescence device | |
| JP2010171204A (en) | Organic electric field light-emitting element |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: FUJIFILM CORPORATION, JAPAN Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNOR:MASUI, KENSUKE;REEL/FRAME:024392/0079 Effective date: 20100427 |
|
| STCB | Information on status: application discontinuation |
Free format text: ABANDONED -- FAILURE TO RESPOND TO AN OFFICE ACTION |
|
| AS | Assignment |
Owner name: UDC IRELAND LIMITED, IRELAND Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNOR:FUJIFILM CORPORATION;REEL/FRAME:028889/0759 Effective date: 20120726 |