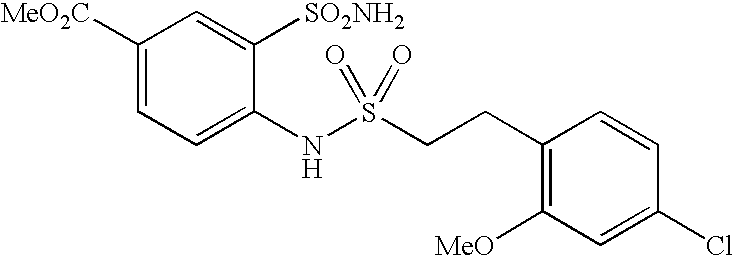
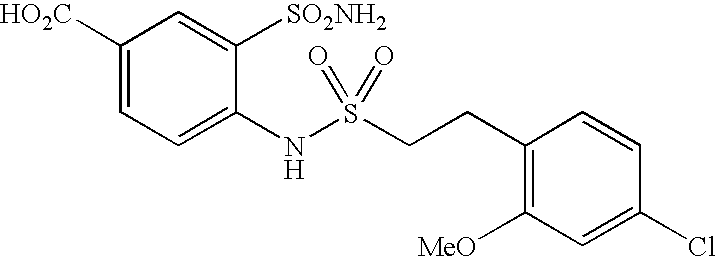
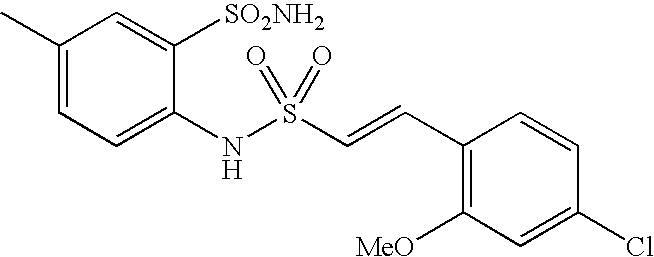
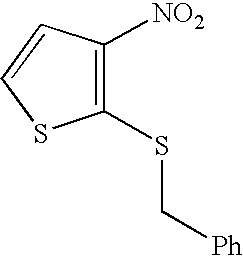
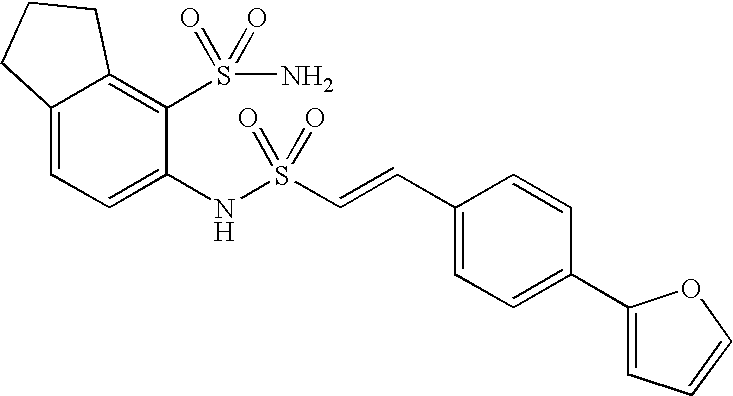
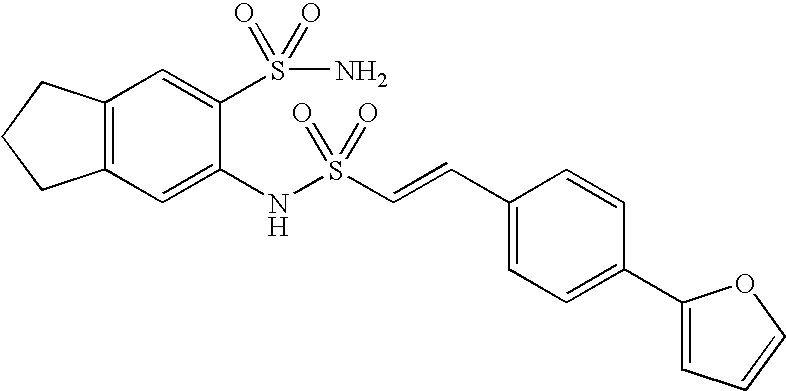
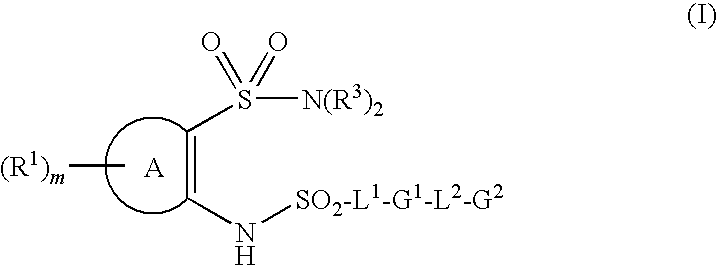US20090163586A1 - Bis-(Sulfonylamino) Derivatives in Therapy 205 - Google Patents
Bis-(Sulfonylamino) Derivatives in Therapy 205 Download PDFInfo
- Publication number
- US20090163586A1 US20090163586A1 US12/337,125 US33712508A US2009163586A1 US 20090163586 A1 US20090163586 A1 US 20090163586A1 US 33712508 A US33712508 A US 33712508A US 2009163586 A1 US2009163586 A1 US 2009163586A1
- Authority
- US
- United States
- Prior art keywords
- benzenesulfonamide
- phenyl
- sulfonylamino
- fluoro
- ethenyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Abandoned
Links
- 238000002560 therapeutic procedure Methods 0.000 title abstract description 11
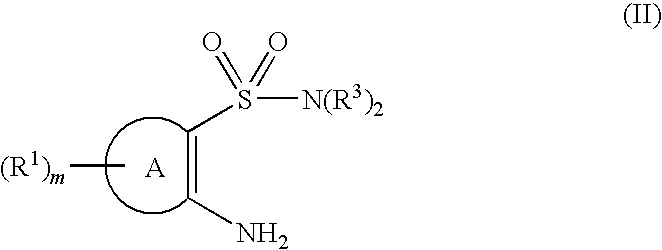
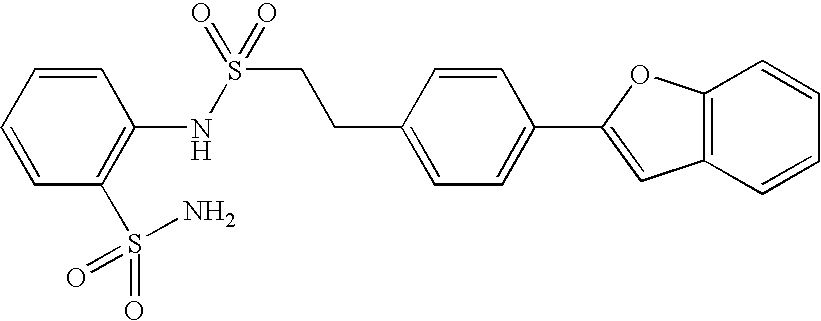
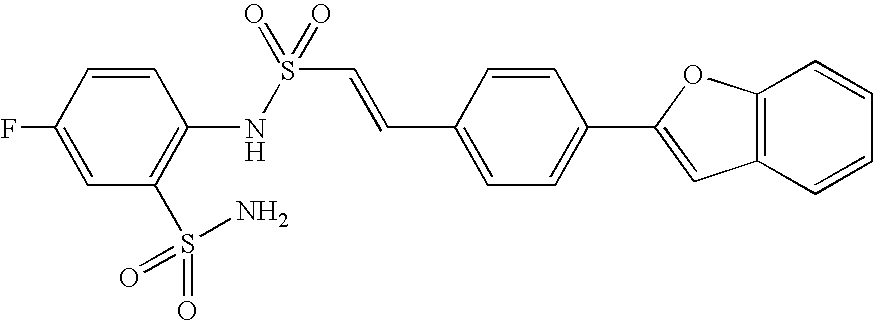
- 150000001875 compounds Chemical class 0.000 claims abstract description 370
- 238000000034 method Methods 0.000 claims abstract description 85
- 150000003839 salts Chemical class 0.000 claims abstract description 55
- 102100033076 Prostaglandin E synthase Human genes 0.000 claims abstract description 25
- 101710096361 Prostaglandin E synthase Proteins 0.000 claims abstract description 25
- 238000002360 preparation method Methods 0.000 claims abstract description 24
- 230000008569 process Effects 0.000 claims abstract description 16
- 239000008194 pharmaceutical composition Substances 0.000 claims abstract description 14
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 179
- -1 2,3-dihydro-indenyl Chemical group 0.000 claims description 109
- 125000006570 (C5-C6) heteroaryl group Chemical group 0.000 claims description 99
- 125000004178 (C1-C4) alkyl group Chemical group 0.000 claims description 91
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 claims description 65
- 229910052736 halogen Inorganic materials 0.000 claims description 59
- 150000002367 halogens Chemical class 0.000 claims description 59
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 55
- 229910052739 hydrogen Inorganic materials 0.000 claims description 50
- 239000001257 hydrogen Substances 0.000 claims description 49
- 125000004076 pyridyl group Chemical group 0.000 claims description 44
- 125000001153 fluoro group Chemical group F* 0.000 claims description 41
- 125000000229 (C1-C4)alkoxy group Chemical group 0.000 claims description 38
- 125000001424 substituent group Chemical group 0.000 claims description 38
- 125000004191 (C1-C6) alkoxy group Chemical group 0.000 claims description 37
- 125000000623 heterocyclic group Chemical group 0.000 claims description 37
- 125000001544 thienyl group Chemical group 0.000 claims description 31
- 125000001072 heteroaryl group Chemical group 0.000 claims description 30
- 125000004452 carbocyclyl group Chemical group 0.000 claims description 28
- 208000002193 Pain Diseases 0.000 claims description 26
- 125000004435 hydrogen atom Chemical class [H]* 0.000 claims description 23
- 206010028980 Neoplasm Diseases 0.000 claims description 18
- 230000000694 effects Effects 0.000 claims description 16
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 claims description 16
- 125000001041 indolyl group Chemical group 0.000 claims description 13
- 125000000335 thiazolyl group Chemical group 0.000 claims description 13
- 230000001154 acute effect Effects 0.000 claims description 12
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 claims description 12
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 claims description 12
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims description 12
- 125000002541 furyl group Chemical group 0.000 claims description 12
- 125000003226 pyrazolyl group Chemical group 0.000 claims description 12
- 125000000714 pyrimidinyl group Chemical group 0.000 claims description 12
- 229910052717 sulfur Inorganic materials 0.000 claims description 12
- 125000003718 tetrahydrofuranyl group Chemical group 0.000 claims description 12
- 201000001320 Atherosclerosis Diseases 0.000 claims description 11
- 208000000094 Chronic Pain Diseases 0.000 claims description 11
- 208000005298 acute pain Diseases 0.000 claims description 11
- 125000002433 cyclopentenyl group Chemical group C1(=CCCC1)* 0.000 claims description 11
- 201000010099 disease Diseases 0.000 claims description 11
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 claims description 11
- 201000008482 osteoarthritis Diseases 0.000 claims description 11
- 206010039073 rheumatoid arthritis Diseases 0.000 claims description 11
- ZMSIFDIKIXVLDF-UHFFFAOYSA-N 2-methyl-1,3,4-oxadiazole Chemical compound CC1=NN=CO1 ZMSIFDIKIXVLDF-UHFFFAOYSA-N 0.000 claims description 10
- 208000034972 Sudden Infant Death Diseases 0.000 claims description 10
- 206010042440 Sudden infant death syndrome Diseases 0.000 claims description 10
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 10
- 125000001624 naphthyl group Chemical group 0.000 claims description 10
- HCMJWOGOISXSDL-UHFFFAOYSA-N (2-isothiocyanato-1-phenylethyl)benzene Chemical compound C=1C=CC=CC=1C(CN=C=S)C1=CC=CC=C1 HCMJWOGOISXSDL-UHFFFAOYSA-N 0.000 claims description 9
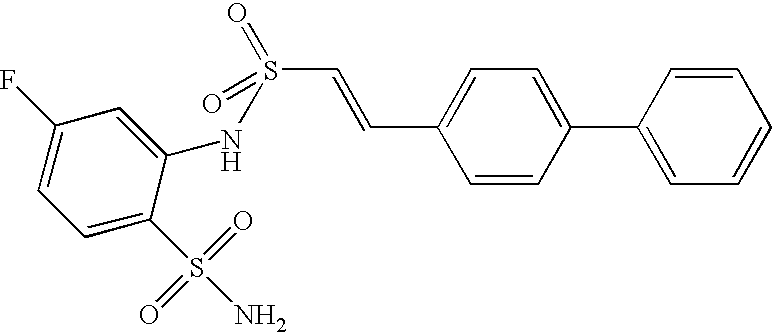
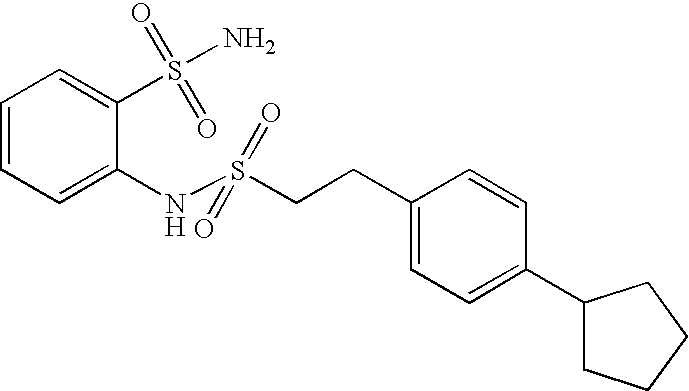
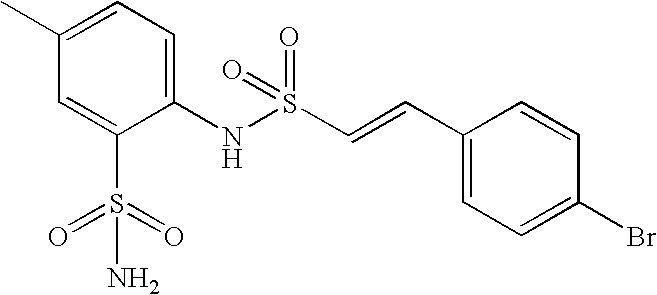
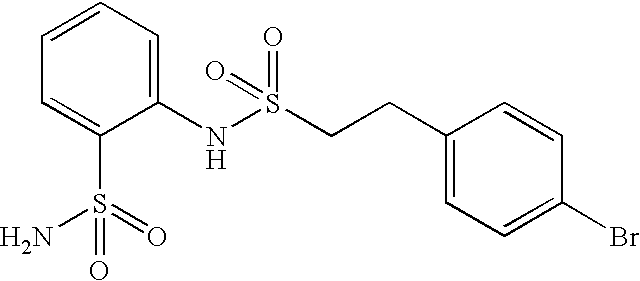
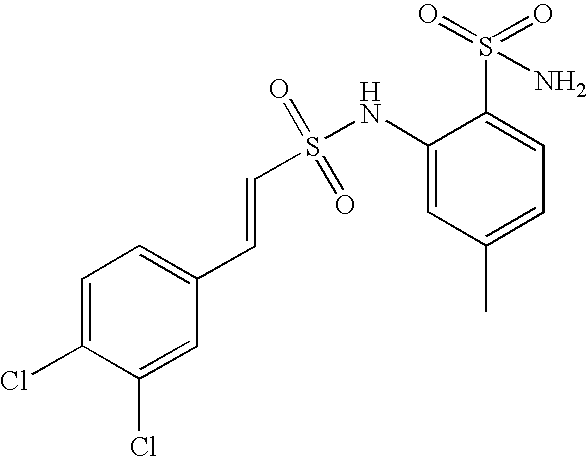
- HWNKAJDTOOPJAM-CMDGGOBGSA-N 2-[[(e)-2-(4-bromophenyl)ethenyl]sulfonylamino]-5-methylbenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(C)=CC=C1NS(=O)(=O)\C=C\C1=CC=C(Br)C=C1 HWNKAJDTOOPJAM-CMDGGOBGSA-N 0.000 claims description 9
- 201000011510 cancer Diseases 0.000 claims description 9
- 230000003211 malignant effect Effects 0.000 claims description 9
- 230000009826 neoplastic cell growth Effects 0.000 claims description 9
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 9
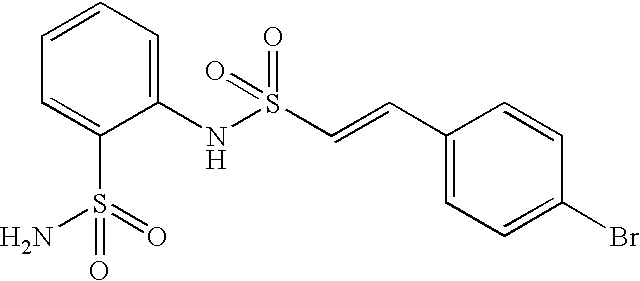
- ZNCMOBUIHAAYQH-MDZDMXLPSA-N 2-[[(e)-2-(4-bromophenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)\C=C\C1=CC=C(Br)C=C1 ZNCMOBUIHAAYQH-MDZDMXLPSA-N 0.000 claims description 8
- NDGUTZUJOOVPOQ-ACCUITESSA-N 2-[[(e)-2-[4-(furan-2-yl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)\C=C\C1=CC=C(C=2OC=CC=2)C=C1 NDGUTZUJOOVPOQ-ACCUITESSA-N 0.000 claims description 8
- KHBQMWCZKVMBLN-UHFFFAOYSA-N Benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1 KHBQMWCZKVMBLN-UHFFFAOYSA-N 0.000 claims description 8
- 239000002671 adjuvant Substances 0.000 claims description 8
- 208000004296 neuralgia Diseases 0.000 claims description 8
- 208000021722 neuropathic pain Diseases 0.000 claims description 8
- DSRGLCZJLTWWFA-UHFFFAOYSA-N 2,3-dichloro-n-(4-methyl-2-sulfamoylphenyl)benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(C)=CC=C1NS(=O)(=O)C1=CC=CC(Cl)=C1Cl DSRGLCZJLTWWFA-UHFFFAOYSA-N 0.000 claims description 7
- LGEWRKDNMAFFRB-UHFFFAOYSA-N 2-[2-(4-bromophenyl)ethylsulfonylamino]-5-methylbenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(C)=CC=C1NS(=O)(=O)CCC1=CC=C(Br)C=C1 LGEWRKDNMAFFRB-UHFFFAOYSA-N 0.000 claims description 7
- MZYSHZXYUOUIBX-UHFFFAOYSA-N 2-[2-(4-chlorophenyl)ethylsulfonylamino]-5-methylbenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(C)=CC=C1NS(=O)(=O)CCC1=CC=C(Cl)C=C1 MZYSHZXYUOUIBX-UHFFFAOYSA-N 0.000 claims description 7
- RWIPRVONYAMWLE-UHFFFAOYSA-N 2-[[3-(3,4-dichlorophenyl)phenyl]sulfonylamino]-5-methylbenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(C)=CC=C1NS(=O)(=O)C1=CC=CC(C=2C=C(Cl)C(Cl)=CC=2)=C1 RWIPRVONYAMWLE-UHFFFAOYSA-N 0.000 claims description 7
- 208000024827 Alzheimer disease Diseases 0.000 claims description 7
- 125000003118 aryl group Chemical group 0.000 claims description 7
- 239000003085 diluting agent Substances 0.000 claims description 7
- 125000004972 1-butynyl group Chemical group [H]C([H])([H])C([H])([H])C#C* 0.000 claims description 6
- VQTKZMCTWPHJHD-UHFFFAOYSA-N 2-(1-phenylpropan-2-ylsulfonylamino)benzenesulfonamide Chemical compound C=1C=CC=C(S(N)(=O)=O)C=1NS(=O)(=O)C(C)CC1=CC=CC=C1 VQTKZMCTWPHJHD-UHFFFAOYSA-N 0.000 claims description 6
- BWRGXJUAIRMKBR-UHFFFAOYSA-N 2-[(2-nitrophenyl)sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC=C1[N+]([O-])=O BWRGXJUAIRMKBR-UHFFFAOYSA-N 0.000 claims description 6
- MTYAYRDXCSXATC-UHFFFAOYSA-N 2-[(4-aminophenyl)sulfonylamino]benzenesulfonamide Chemical compound C1=CC(N)=CC=C1S(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O MTYAYRDXCSXATC-UHFFFAOYSA-N 0.000 claims description 6
- PCWVGEPALSDKRK-UHFFFAOYSA-N 2-[(4-nitrophenyl)sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=C([N+]([O-])=O)C=C1 PCWVGEPALSDKRK-UHFFFAOYSA-N 0.000 claims description 6
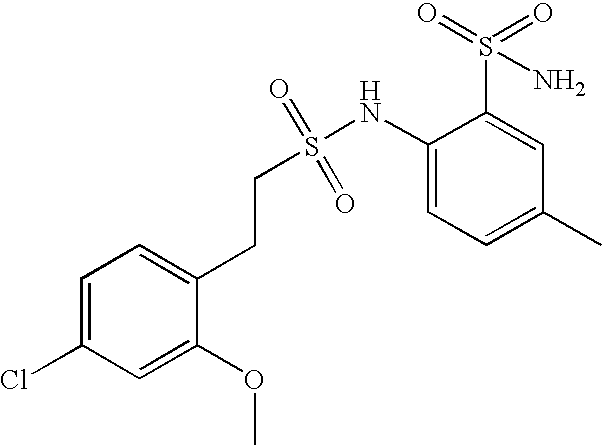
- OVELXBOBYSFKET-UHFFFAOYSA-N 2-[2-(4-chloro-2-methoxyphenyl)ethylsulfonylamino]-5-methylbenzenesulfonamide Chemical compound COC1=CC(Cl)=CC=C1CCS(=O)(=O)NC1=CC=C(C)C=C1S(N)(=O)=O OVELXBOBYSFKET-UHFFFAOYSA-N 0.000 claims description 6
- XJWAJGXHQJXRMR-UHFFFAOYSA-N 2-[2-(4-chlorophenyl)prop-2-enylsulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)CC(=C)C1=CC=C(Cl)C=C1 XJWAJGXHQJXRMR-UHFFFAOYSA-N 0.000 claims description 6
- TWKSLVDUWUDLGR-CSKARUKUSA-N 2-[[(e)-1-(4-chlorophenyl)prop-1-en-2-yl]sulfonylamino]-4-fluorobenzenesulfonamide Chemical compound C=1C(F)=CC=C(S(N)(=O)=O)C=1NS(=O)(=O)C(/C)=C/C1=CC=C(Cl)C=C1 TWKSLVDUWUDLGR-CSKARUKUSA-N 0.000 claims description 6
- RLJRHWYXJQHCIL-ZHACJKMWSA-N 2-[[(e)-1-(4-chlorophenyl)prop-1-en-2-yl]sulfonylamino]benzenesulfonamide Chemical compound C=1C=CC=C(S(N)(=O)=O)C=1NS(=O)(=O)C(/C)=C/C1=CC=C(Cl)C=C1 RLJRHWYXJQHCIL-ZHACJKMWSA-N 0.000 claims description 6
- APMRRWAAFOTXJP-VOTSOKGWSA-N 2-[[(e)-2-(3,4-dichlorophenyl)ethenyl]sulfonylamino]-4-methylbenzenesulfonamide Chemical compound CC1=CC=C(S(N)(=O)=O)C(NS(=O)(=O)\C=C\C=2C=C(Cl)C(Cl)=CC=2)=C1 APMRRWAAFOTXJP-VOTSOKGWSA-N 0.000 claims description 6
- TVWUTLDKFWRVKN-ZHACJKMWSA-N 2-[[(e)-2-(4-chlorophenyl)prop-1-enyl]sulfonylamino]benzenesulfonamide Chemical compound C=1C=C(Cl)C=CC=1C(/C)=C/S(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O TVWUTLDKFWRVKN-ZHACJKMWSA-N 0.000 claims description 6
- SYRHWHGERYMDIA-CMDGGOBGSA-N 2-[[(e)-2-(4-cyanophenyl)ethenyl]sulfonylamino]-6-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=C(F)C=CC=C1NS(=O)(=O)\C=C\C1=CC=C(C#N)C=C1 SYRHWHGERYMDIA-CMDGGOBGSA-N 0.000 claims description 6
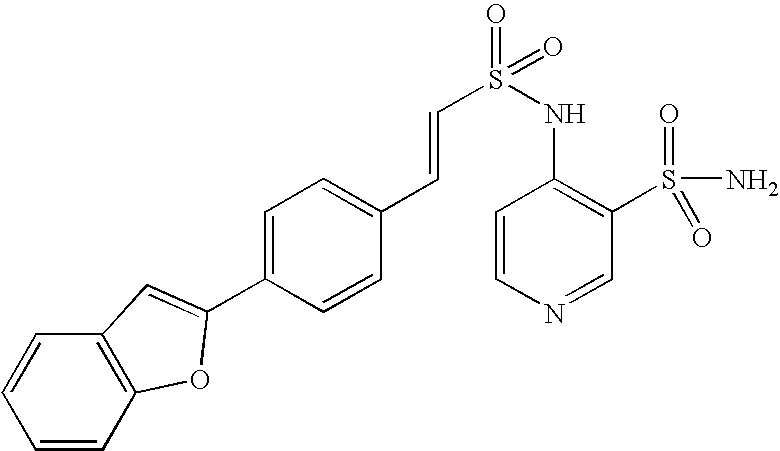
- QSLCIVOOBRTCPR-BUHFOSPRSA-N 2-[[(e)-2-[4-(1-benzofuran-2-yl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)\C=C\C1=CC=C(C=2OC3=CC=CC=C3C=2)C=C1 QSLCIVOOBRTCPR-BUHFOSPRSA-N 0.000 claims description 6
- UYGDVXHADRKHEX-UHFFFAOYSA-N 2-amino-n-(2-sulfamoylphenyl)benzenesulfonamide Chemical compound NC1=CC=CC=C1S(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O UYGDVXHADRKHEX-UHFFFAOYSA-N 0.000 claims description 6
- SDYMTGFQIZOEHK-PKNBQFBNSA-N 5-fluoro-2-[[(e)-2-[4-(furan-2-yl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(F)=CC=C1NS(=O)(=O)\C=C\C1=CC=C(C=2OC=CC=2)C=C1 SDYMTGFQIZOEHK-PKNBQFBNSA-N 0.000 claims description 6
- 201000002481 Myositis Diseases 0.000 claims description 6
- 230000009286 beneficial effect Effects 0.000 claims description 6
- 125000001246 bromo group Chemical group Br* 0.000 claims description 6
- 125000001309 chloro group Chemical group Cl* 0.000 claims description 6
- 208000027866 inflammatory disease Diseases 0.000 claims description 6
- URCXBBNYLDJMDK-UHFFFAOYSA-N n-(2-sulfamoylphenyl)quinoline-8-sulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC2=CC=CN=C12 URCXBBNYLDJMDK-UHFFFAOYSA-N 0.000 claims description 6
- MPRDETHCLKAVEA-UHFFFAOYSA-N n-[2-(dimethylsulfamoyl)phenyl]-5,7-dimethyl-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide Chemical compound CN(C)S(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=NN2C(C)=CC(C)=NC2=N1 MPRDETHCLKAVEA-UHFFFAOYSA-N 0.000 claims description 6
- RPISIWMSRIPHRF-UHFFFAOYSA-N 2-[1-(4-chlorophenyl)propan-2-ylsulfonylamino]-4-fluorobenzenesulfonamide Chemical compound C=1C(F)=CC=C(S(N)(=O)=O)C=1NS(=O)(=O)C(C)CC1=CC=C(Cl)C=C1 RPISIWMSRIPHRF-UHFFFAOYSA-N 0.000 claims description 5
- BCKYAJYAYZILII-UHFFFAOYSA-N 2-[1-(4-chlorophenyl)propan-2-ylsulfonylamino]benzenesulfonamide Chemical compound C=1C=CC=C(S(N)(=O)=O)C=1NS(=O)(=O)C(C)CC1=CC=C(Cl)C=C1 BCKYAJYAYZILII-UHFFFAOYSA-N 0.000 claims description 5
- MZDQAHVWNXCFEO-UHFFFAOYSA-N 2-[2-(4-chloro-2-methoxyphenyl)ethylsulfonylamino]benzenesulfonamide Chemical compound COC1=CC(Cl)=CC=C1CCS(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O MZDQAHVWNXCFEO-UHFFFAOYSA-N 0.000 claims description 5
- JQIOOEILFNIXIL-UHFFFAOYSA-N 2-[2-(4-chlorophenyl)ethylsulfonylamino]-5-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(F)=CC=C1NS(=O)(=O)CCC1=CC=C(Cl)C=C1 JQIOOEILFNIXIL-UHFFFAOYSA-N 0.000 claims description 5
- ZGQJLSDJMZCDEC-UHFFFAOYSA-N 2-[2-[4-(1-benzofuran-2-yl)phenyl]ethylsulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)CCC1=CC=C(C=2OC3=CC=CC=C3C=2)C=C1 ZGQJLSDJMZCDEC-UHFFFAOYSA-N 0.000 claims description 5
- YGBRXEGHBVZLBL-UHFFFAOYSA-N 2-[2-[4-(2,3-dihydro-1-benzofuran-2-yl)phenyl]ethylsulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)CCC1=CC=C(C2OC3=CC=CC=C3C2)C=C1 YGBRXEGHBVZLBL-UHFFFAOYSA-N 0.000 claims description 5
- PJTFTBFWKUUNMQ-UHFFFAOYSA-N 2-[2-[4-(difluoromethoxy)phenyl]ethylsulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)CCC1=CC=C(OC(F)F)C=C1 PJTFTBFWKUUNMQ-UHFFFAOYSA-N 0.000 claims description 5
- IKMYIGLPUUEJTB-UHFFFAOYSA-N 2-[2-[4-(furan-2-yl)phenyl]ethylsulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)CCC1=CC=C(C=2OC=CC=2)C=C1 IKMYIGLPUUEJTB-UHFFFAOYSA-N 0.000 claims description 5
- YSHYBMWKTMZFDP-UHFFFAOYSA-N 2-[2-[4-(oxolan-2-yl)phenyl]ethylsulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)CCC1=CC=C(C2OCCC2)C=C1 YSHYBMWKTMZFDP-UHFFFAOYSA-N 0.000 claims description 5
- SWPNMXLELMPZFB-MDZDMXLPSA-N 2-[[(e)-1-(4-chlorophenyl)prop-1-en-2-yl]sulfonylamino]-3-fluorobenzenesulfonamide Chemical compound FC=1C=CC=C(S(N)(=O)=O)C=1NS(=O)(=O)C(/C)=C/C1=CC=C(Cl)C=C1 SWPNMXLELMPZFB-MDZDMXLPSA-N 0.000 claims description 5
- KTMTUGUPALZGRR-CSKARUKUSA-N 2-[[(e)-1-(4-chlorophenyl)prop-1-en-2-yl]sulfonylamino]-5-fluorobenzenesulfonamide Chemical compound C=1C=C(F)C=C(S(N)(=O)=O)C=1NS(=O)(=O)C(/C)=C/C1=CC=C(Cl)C=C1 KTMTUGUPALZGRR-CSKARUKUSA-N 0.000 claims description 5
- CPKNRIHSAARGAO-ZRDIBKRKSA-N 2-[[(e)-1-(4-chlorophenyl)prop-1-en-2-yl]sulfonylamino]-5-methylbenzenesulfonamide Chemical compound C=1C=C(C)C=C(S(N)(=O)=O)C=1NS(=O)(=O)C(/C)=C/C1=CC=C(Cl)C=C1 CPKNRIHSAARGAO-ZRDIBKRKSA-N 0.000 claims description 5
- HLWQMRNUHFSKJH-MDZDMXLPSA-N 2-[[(e)-1-(4-chlorophenyl)prop-1-en-2-yl]sulfonylamino]-6-fluorobenzenesulfonamide Chemical compound C=1C=CC(F)=C(S(N)(=O)=O)C=1NS(=O)(=O)C(/C)=C/C1=CC=C(Cl)C=C1 HLWQMRNUHFSKJH-MDZDMXLPSA-N 0.000 claims description 5
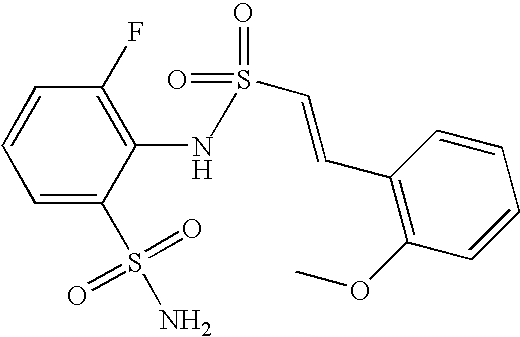
- KXXOPFAJQXWSPI-ZHACJKMWSA-N 2-[[(e)-2-(2-methoxyphenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound COC1=CC=CC=C1\C=C\S(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O KXXOPFAJQXWSPI-ZHACJKMWSA-N 0.000 claims description 5
- CECLTTVHKRBDMR-AATRIKPKSA-N 2-[[(e)-2-(3,4-dichlorophenyl)ethenyl]sulfonylamino]-4-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=C(F)C=C1NS(=O)(=O)\C=C\C1=CC=C(Cl)C(Cl)=C1 CECLTTVHKRBDMR-AATRIKPKSA-N 0.000 claims description 5
- NVQRQHLCEOOCFV-AATRIKPKSA-N 2-[[(e)-2-(3,4-dichlorophenyl)ethenyl]sulfonylamino]-5-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(F)=CC=C1NS(=O)(=O)\C=C\C1=CC=C(Cl)C(Cl)=C1 NVQRQHLCEOOCFV-AATRIKPKSA-N 0.000 claims description 5
- JCBKUIKVKXUYRF-VOTSOKGWSA-N 2-[[(e)-2-(3,4-dichlorophenyl)ethenyl]sulfonylamino]-6-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=C(F)C=CC=C1NS(=O)(=O)\C=C\C1=CC=C(Cl)C(Cl)=C1 JCBKUIKVKXUYRF-VOTSOKGWSA-N 0.000 claims description 5
- MVRMEOTUXAYJKH-BQYQJAHWSA-N 2-[[(e)-2-(4-chlorophenyl)ethenyl]sulfonylamino]-4-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=C(F)C=C1NS(=O)(=O)\C=C\C1=CC=C(Cl)C=C1 MVRMEOTUXAYJKH-BQYQJAHWSA-N 0.000 claims description 5
- BUEDELQFGJYNOH-BQYQJAHWSA-N 2-[[(e)-2-(4-chlorophenyl)ethenyl]sulfonylamino]-5-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(F)=CC=C1NS(=O)(=O)\C=C\C1=CC=C(Cl)C=C1 BUEDELQFGJYNOH-BQYQJAHWSA-N 0.000 claims description 5
- GHUJQXDFIFXCND-BQYQJAHWSA-N 2-[[(e)-2-(4-cyanophenyl)ethenyl]sulfonylamino]-4-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=C(F)C=C1NS(=O)(=O)\C=C\C1=CC=C(C#N)C=C1 GHUJQXDFIFXCND-BQYQJAHWSA-N 0.000 claims description 5
- HLPZWFZIFXKYNH-BQYQJAHWSA-N 2-[[(e)-2-(4-cyanophenyl)ethenyl]sulfonylamino]-5-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(F)=CC=C1NS(=O)(=O)\C=C\C1=CC=C(C#N)C=C1 HLPZWFZIFXKYNH-BQYQJAHWSA-N 0.000 claims description 5
- XNXMPQWOZHEZAU-MDZDMXLPSA-N 2-[[(e)-2-(4-cyanophenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)\C=C\C1=CC=C(C#N)C=C1 XNXMPQWOZHEZAU-MDZDMXLPSA-N 0.000 claims description 5
- FTEWRPXFKJSQBT-VOTSOKGWSA-N 2-[[(e)-2-(5-bromo-2-methoxyphenyl)ethenyl]sulfonylamino]-4-fluorobenzenesulfonamide Chemical compound COC1=CC=C(Br)C=C1\C=C\S(=O)(=O)NC1=CC(F)=CC=C1S(N)(=O)=O FTEWRPXFKJSQBT-VOTSOKGWSA-N 0.000 claims description 5
- IKAGTRGAUYEFEB-VOTSOKGWSA-N 2-[[(e)-2-(5-bromo-2-methoxyphenyl)ethenyl]sulfonylamino]-5-fluorobenzenesulfonamide Chemical compound COC1=CC=C(Br)C=C1\C=C\S(=O)(=O)NC1=CC=C(F)C=C1S(N)(=O)=O IKAGTRGAUYEFEB-VOTSOKGWSA-N 0.000 claims description 5
- RXXNOVINMXGGHJ-BQYQJAHWSA-N 2-[[(e)-2-(5-bromo-2-methoxyphenyl)ethenyl]sulfonylamino]-6-fluorobenzenesulfonamide Chemical compound COC1=CC=C(Br)C=C1\C=C\S(=O)(=O)NC1=CC=CC(F)=C1S(N)(=O)=O RXXNOVINMXGGHJ-BQYQJAHWSA-N 0.000 claims description 5
- CDMLAJTVRJZPNT-CMDGGOBGSA-N 2-[[(e)-2-(5-bromo-2-methoxyphenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound COC1=CC=C(Br)C=C1\C=C\S(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O CDMLAJTVRJZPNT-CMDGGOBGSA-N 0.000 claims description 5
- RMYDADGWCAXZGR-VAWYXSNFSA-N 2-[[(e)-2-[4-(1-benzofuran-2-yl)phenyl]ethenyl]sulfonylamino]-4-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=C(F)C=C1NS(=O)(=O)\C=C\C1=CC=C(C=2OC3=CC=CC=C3C=2)C=C1 RMYDADGWCAXZGR-VAWYXSNFSA-N 0.000 claims description 5
- AHOUWQUZYZKABN-VAWYXSNFSA-N 2-[[(e)-2-[4-(1-benzofuran-2-yl)phenyl]ethenyl]sulfonylamino]-5-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(F)=CC=C1NS(=O)(=O)\C=C\C1=CC=C(C=2OC3=CC=CC=C3C=2)C=C1 AHOUWQUZYZKABN-VAWYXSNFSA-N 0.000 claims description 5
- VJTALGROLDPLIQ-OUKQBFOZSA-N 2-[[(e)-2-[4-(1-benzofuran-2-yl)phenyl]ethenyl]sulfonylamino]-6-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=C(F)C=CC=C1NS(=O)(=O)\C=C\C1=CC=C(C=2OC3=CC=CC=C3C=2)C=C1 VJTALGROLDPLIQ-OUKQBFOZSA-N 0.000 claims description 5
- ZKOSCZAJBSCADS-BUHFOSPRSA-N 2-[[(e)-2-[4-(cyclopenten-1-yl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)\C=C\C1=CC=C(C=2CCCC=2)C=C1 ZKOSCZAJBSCADS-BUHFOSPRSA-N 0.000 claims description 5
- UQPDTOOCKMJAMI-MDZDMXLPSA-N 2-[[(e)-2-[4-(difluoromethoxy)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)\C=C\C1=CC=C(OC(F)F)C=C1 UQPDTOOCKMJAMI-MDZDMXLPSA-N 0.000 claims description 5
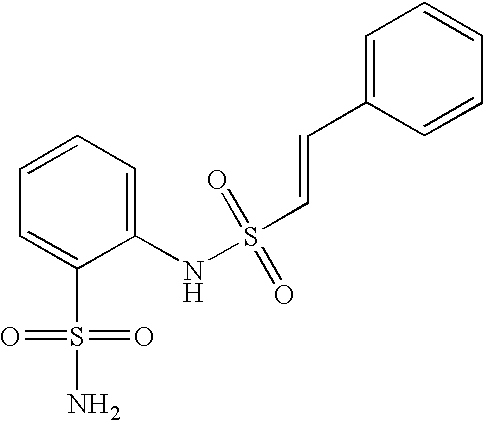
- KUXYLAJDYAHOEG-ZHACJKMWSA-N 2-[[(e)-2-phenylethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)\C=C\C1=CC=CC=C1 KUXYLAJDYAHOEG-ZHACJKMWSA-N 0.000 claims description 5
- KDOXJJPEZQAITK-ZRDIBKRKSA-N 2-fluoro-6-[[(e)-2-[4-(furan-2-yl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=C(F)C=CC=C1NS(=O)(=O)\C=C\C1=CC=C(C=2OC=CC=2)C=C1 KDOXJJPEZQAITK-ZRDIBKRKSA-N 0.000 claims description 5
- RDFJCOMPENGOJW-CMDGGOBGSA-N 4-fluoro-2-[[(e)-2-(4-methylsulfanylphenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound C1=CC(SC)=CC=C1\C=C\S(=O)(=O)NC1=CC(F)=CC=C1S(N)(=O)=O RDFJCOMPENGOJW-CMDGGOBGSA-N 0.000 claims description 5
- DJIDAAFNARCKGM-PKNBQFBNSA-N 4-fluoro-2-[[(e)-2-[4-(furan-2-yl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=C(F)C=C1NS(=O)(=O)\C=C\C1=CC=C(C=2OC=CC=2)C=C1 DJIDAAFNARCKGM-PKNBQFBNSA-N 0.000 claims description 5
- AGIXEQRJWSUMFM-UHFFFAOYSA-N 5-bromo-2-[[3-(3,4-dichlorophenyl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(Br)=CC=C1NS(=O)(=O)C1=CC=CC(C=2C=C(Cl)C(Cl)=CC=2)=C1 AGIXEQRJWSUMFM-UHFFFAOYSA-N 0.000 claims description 5
- BEVKWSHWIMEENI-CMDGGOBGSA-N 5-fluoro-2-[[(e)-2-(4-methylsulfanylphenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound C1=CC(SC)=CC=C1\C=C\S(=O)(=O)NC1=CC=C(F)C=C1S(N)(=O)=O BEVKWSHWIMEENI-CMDGGOBGSA-N 0.000 claims description 5
- FALFQDQFHAAZRE-UHFFFAOYSA-N methyl 4-[(5-fluoro-3-methyl-1-benzothiophen-2-yl)sulfonylamino]-3-sulfamoylbenzoate Chemical compound NS(=O)(=O)C1=CC(C(=O)OC)=CC=C1NS(=O)(=O)C1=C(C)C2=CC(F)=CC=C2S1 FALFQDQFHAAZRE-UHFFFAOYSA-N 0.000 claims description 5
- LAUBMMIAHPHZIQ-OAHLLOKOSA-N (2r)-n-[2-chloro-4-[(2-sulfamoylphenyl)sulfamoyl]phenyl]-3,3,3-trifluoro-2-hydroxy-2-methylpropanamide Chemical compound C1=C(Cl)C(NC(=O)[C@](O)(C)C(F)(F)F)=CC=C1S(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O LAUBMMIAHPHZIQ-OAHLLOKOSA-N 0.000 claims description 4
- QSCZKUJMKKBSPL-UHFFFAOYSA-N 1-(4-chlorophenyl)-3-[3-methyl-4-[(2-sulfamoylphenyl)sulfamoyl]phenyl]urea Chemical compound C=1C=C(S(=O)(=O)NC=2C(=CC=CC=2)S(N)(=O)=O)C(C)=CC=1NC(=O)NC1=CC=C(Cl)C=C1 QSCZKUJMKKBSPL-UHFFFAOYSA-N 0.000 claims description 4
- ZVAMPIMQFAFMJN-UHFFFAOYSA-N 2,3-dichloro-n-(2-sulfamoylphenyl)benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC(Cl)=C1Cl ZVAMPIMQFAFMJN-UHFFFAOYSA-N 0.000 claims description 4
- QWADIGJMBYJPNK-UHFFFAOYSA-N 2,3-dichloro-n-(4-chloro-2-sulfamoylphenyl)benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(Cl)=CC=C1NS(=O)(=O)C1=CC=CC(Cl)=C1Cl QWADIGJMBYJPNK-UHFFFAOYSA-N 0.000 claims description 4
- HKTDZGHSWIAQRP-UHFFFAOYSA-N 2,3-dichloro-n-(4-hydroxy-2-sulfamoylphenyl)benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(O)=CC=C1NS(=O)(=O)C1=CC=CC(Cl)=C1Cl HKTDZGHSWIAQRP-UHFFFAOYSA-N 0.000 claims description 4
- OUQFVCUSXVIXBK-UHFFFAOYSA-N 2,3-dichloro-n-[4-(hydroxymethyl)-2-sulfamoylphenyl]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(CO)=CC=C1NS(=O)(=O)C1=CC=CC(Cl)=C1Cl OUQFVCUSXVIXBK-UHFFFAOYSA-N 0.000 claims description 4
- VQLPLQBGYSGBOQ-UHFFFAOYSA-N 2,3-difluoro-n-(2-sulfamoylphenyl)benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC(F)=C1F VQLPLQBGYSGBOQ-UHFFFAOYSA-N 0.000 claims description 4
- HZBSGVBSJGIEOJ-UHFFFAOYSA-N 2,3-difluoro-n-(4-fluoro-2-sulfamoylphenyl)benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(F)=CC=C1NS(=O)(=O)C1=CC=CC(F)=C1F HZBSGVBSJGIEOJ-UHFFFAOYSA-N 0.000 claims description 4
- ZALQZIYUAXUPTH-UHFFFAOYSA-N 2-(2-naphthalen-2-ylethylsulfonylamino)benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)CCC1=CC=C(C=CC=C2)C2=C1 ZALQZIYUAXUPTH-UHFFFAOYSA-N 0.000 claims description 4
- TWZMYIKVQSABDX-UHFFFAOYSA-N 2-(2-phenylethylsulfonylamino)benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)CCC1=CC=CC=C1 TWZMYIKVQSABDX-UHFFFAOYSA-N 0.000 claims description 4
- CALLBRUBPRHBMJ-UHFFFAOYSA-N 2-(2-phenylpropylsulfonylamino)benzenesulfonamide Chemical compound C=1C=CC=CC=1C(C)CS(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O CALLBRUBPRHBMJ-UHFFFAOYSA-N 0.000 claims description 4
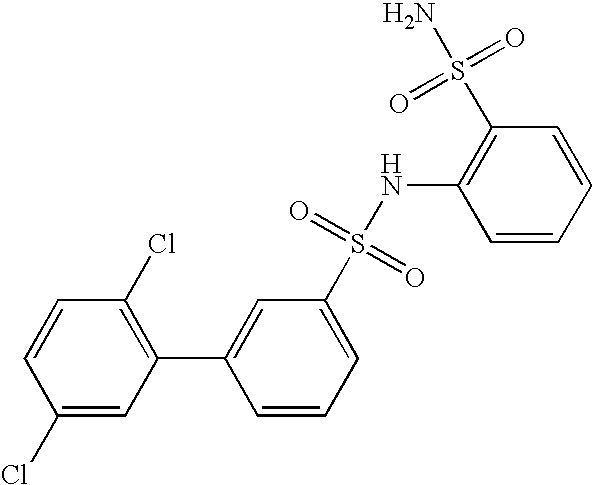
- JGYVPMJDYWSSHY-UHFFFAOYSA-N 2-(3,4-dichlorophenyl)-n-(2-sulfamoylphenyl)benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC=C1C1=CC=C(Cl)C(Cl)=C1 JGYVPMJDYWSSHY-UHFFFAOYSA-N 0.000 claims description 4
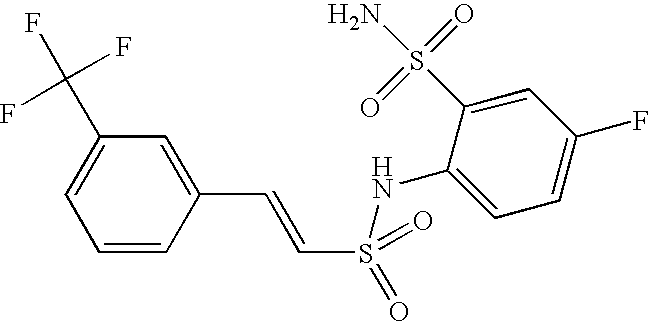
- DYZSPSGBULFYAN-UHFFFAOYSA-N 2-(benzenesulfonamido)-4-(trifluoromethyl)benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=C(C(F)(F)F)C=C1NS(=O)(=O)C1=CC=CC=C1 DYZSPSGBULFYAN-UHFFFAOYSA-N 0.000 claims description 4
- VJFLFBSXBAGBAO-UHFFFAOYSA-N 2-[(2,3-dichlorophenyl)sulfonylamino]-4,5-difluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(F)=C(F)C=C1NS(=O)(=O)C1=CC=CC(Cl)=C1Cl VJFLFBSXBAGBAO-UHFFFAOYSA-N 0.000 claims description 4
- DMPJMEGOWQUUQO-UHFFFAOYSA-N 2-[(2,3-dichlorophenyl)sulfonylamino]-4,6-difluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=C(F)C=C(F)C=C1NS(=O)(=O)C1=CC=CC(Cl)=C1Cl DMPJMEGOWQUUQO-UHFFFAOYSA-N 0.000 claims description 4
- KGGGSYTXBBEDQA-UHFFFAOYSA-N 2-[(2,3-dichlorophenyl)sulfonylamino]-5-fluoro-4-methoxybenzenesulfonamide Chemical compound C1=C(F)C(OC)=CC(NS(=O)(=O)C=2C(=C(Cl)C=CC=2)Cl)=C1S(N)(=O)=O KGGGSYTXBBEDQA-UHFFFAOYSA-N 0.000 claims description 4
- FDCUNYZCSYGCPF-UHFFFAOYSA-N 2-[(3-dibenzofuran-4-ylphenyl)sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC(C=2C3=C(C4=CC=CC=C4O3)C=CC=2)=C1 FDCUNYZCSYGCPF-UHFFFAOYSA-N 0.000 claims description 4
- YKZMUKGPTJVFES-UHFFFAOYSA-N 2-[(3-phenylphenyl)sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC(C=2C=CC=CC=2)=C1 YKZMUKGPTJVFES-UHFFFAOYSA-N 0.000 claims description 4
- BXOGJRNRDNEHCB-UHFFFAOYSA-N 2-[(4-bromophenyl)sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=C(Br)C=C1 BXOGJRNRDNEHCB-UHFFFAOYSA-N 0.000 claims description 4
- VZAQAQHIWYRXDR-UHFFFAOYSA-N 2-[(4-chlorophenyl)methylsulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)CC1=CC=C(Cl)C=C1 VZAQAQHIWYRXDR-UHFFFAOYSA-N 0.000 claims description 4
- PEJJOTNQSJUPDC-UHFFFAOYSA-N 2-[(4-cyclohexylphenyl)sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=C(C2CCCCC2)C=C1 PEJJOTNQSJUPDC-UHFFFAOYSA-N 0.000 claims description 4
- VXOPRHVIYKPQSP-UHFFFAOYSA-N 2-[(4-fluorophenyl)sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=C(F)C=C1 VXOPRHVIYKPQSP-UHFFFAOYSA-N 0.000 claims description 4
- LNBYNKPUMVARRU-UHFFFAOYSA-N 2-[(4-methylphenyl)sulfonylamino]-4-(trifluoromethyl)benzenesulfonamide Chemical compound C1=CC(C)=CC=C1S(=O)(=O)NC1=CC(C(F)(F)F)=CC=C1S(N)(=O)=O LNBYNKPUMVARRU-UHFFFAOYSA-N 0.000 claims description 4
- PHYZEXLCOJVVFD-UHFFFAOYSA-N 2-[(4-methylphenyl)sulfonylamino]benzenesulfonamide Chemical compound C1=CC(C)=CC=C1S(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O PHYZEXLCOJVVFD-UHFFFAOYSA-N 0.000 claims description 4
- WYHXZBBIYLDTCH-UHFFFAOYSA-N 2-[(4-phenylmethoxyphenyl)sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C(C=C1)=CC=C1OCC1=CC=CC=C1 WYHXZBBIYLDTCH-UHFFFAOYSA-N 0.000 claims description 4
- KYRLTUNGHJLEJE-UHFFFAOYSA-N 2-[(4-tert-butylphenyl)sulfonylamino]benzenesulfonamide Chemical compound C1=CC(C(C)(C)C)=CC=C1S(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O KYRLTUNGHJLEJE-UHFFFAOYSA-N 0.000 claims description 4
- PZVACPHKBWKVIP-UHFFFAOYSA-N 2-[2-(2-chlorophenyl)ethenylsulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C=CC1=CC=CC=C1Cl PZVACPHKBWKVIP-UHFFFAOYSA-N 0.000 claims description 4
- VXXQJLMUZWBWNG-UHFFFAOYSA-N 2-[2-(2-chlorophenyl)ethylsulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)CCC1=CC=CC=C1Cl VXXQJLMUZWBWNG-UHFFFAOYSA-N 0.000 claims description 4
- HYWXZJNZWDXMHH-UHFFFAOYSA-N 2-[2-(2-hydroxyphenyl)ethenylsulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C=CC1=CC=CC=C1O HYWXZJNZWDXMHH-UHFFFAOYSA-N 0.000 claims description 4
- WNZQKXLBUZMELB-UHFFFAOYSA-N 2-[2-(2-methoxyphenyl)ethylsulfonylamino]benzenesulfonamide Chemical compound COC1=CC=CC=C1CCS(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O WNZQKXLBUZMELB-UHFFFAOYSA-N 0.000 claims description 4
- BQARJCVSHFPJNW-UHFFFAOYSA-N 2-[2-(3,4-dichlorophenyl)ethylsulfonylamino]-3,5-difluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(F)=CC(F)=C1NS(=O)(=O)CCC1=CC=C(Cl)C(Cl)=C1 BQARJCVSHFPJNW-UHFFFAOYSA-N 0.000 claims description 4
- FOHOXSZPMMGVJP-UHFFFAOYSA-N 2-[2-(3,4-dichlorophenyl)ethylsulfonylamino]-4,6-difluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=C(F)C=C(F)C=C1NS(=O)(=O)CCC1=CC=C(Cl)C(Cl)=C1 FOHOXSZPMMGVJP-UHFFFAOYSA-N 0.000 claims description 4
- NEIGAPREMXPKID-UHFFFAOYSA-N 2-[2-(4-chloro-2-methoxyphenyl)ethenylsulfonylamino]benzenesulfonamide Chemical compound COC1=CC(Cl)=CC=C1C=CS(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O NEIGAPREMXPKID-UHFFFAOYSA-N 0.000 claims description 4
- BUGCZQAKLDHJRU-UHFFFAOYSA-N 2-[2-(4-chloro-2-methoxyphenyl)ethylsulfonylamino]-4,6-difluorobenzenesulfonamide Chemical compound COC1=CC(Cl)=CC=C1CCS(=O)(=O)NC1=CC(F)=CC(F)=C1S(N)(=O)=O BUGCZQAKLDHJRU-UHFFFAOYSA-N 0.000 claims description 4
- BSXXISGHGICHCP-UHFFFAOYSA-N 2-[2-(4-chloro-2-methoxyphenyl)ethylsulfonylamino]-4-cyanobenzenesulfonamide Chemical compound COC1=CC(Cl)=CC=C1CCS(=O)(=O)NC1=CC(C#N)=CC=C1S(N)(=O)=O BSXXISGHGICHCP-UHFFFAOYSA-N 0.000 claims description 4
- ISZGJPAGBUPEKN-UHFFFAOYSA-N 2-[2-(4-chloro-2-methoxyphenyl)ethylsulfonylamino]-4-methylbenzenesulfonamide Chemical compound COC1=CC(Cl)=CC=C1CCS(=O)(=O)NC1=CC(C)=CC=C1S(N)(=O)=O ISZGJPAGBUPEKN-UHFFFAOYSA-N 0.000 claims description 4
- YIOUAQVZUCGGRD-UHFFFAOYSA-N 2-[2-(4-chloro-2-methoxyphenyl)ethylsulfonylamino]-5-(hydroxymethyl)benzenesulfonamide Chemical compound COC1=CC(Cl)=CC=C1CCS(=O)(=O)NC1=CC=C(CO)C=C1S(N)(=O)=O YIOUAQVZUCGGRD-UHFFFAOYSA-N 0.000 claims description 4
- IHNUIVJXKRGXEV-UHFFFAOYSA-N 2-[2-(4-chloro-2-methoxyphenyl)ethylsulfonylamino]-5-cyanobenzenesulfonamide Chemical compound COC1=CC(Cl)=CC=C1CCS(=O)(=O)NC1=CC=C(C#N)C=C1S(N)(=O)=O IHNUIVJXKRGXEV-UHFFFAOYSA-N 0.000 claims description 4
- OPKXYGSFCHBLHJ-UHFFFAOYSA-N 2-[2-(4-chlorophenyl)ethylsulfonylamino]-4-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=C(F)C=C1NS(=O)(=O)CCC1=CC=C(Cl)C=C1 OPKXYGSFCHBLHJ-UHFFFAOYSA-N 0.000 claims description 4
- JTWRNOBJVPSQRX-UHFFFAOYSA-N 2-[2-(4-chlorophenyl)ethylsulfonylamino]-5-(hydroxymethyl)benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(CO)=CC=C1NS(=O)(=O)CCC1=CC=C(Cl)C=C1 JTWRNOBJVPSQRX-UHFFFAOYSA-N 0.000 claims description 4
- VMWSNBVSSUDCBY-UHFFFAOYSA-N 2-[2-(4-chlorophenyl)ethylsulfonylamino]-5-(pyridin-2-ylmethoxy)benzenesulfonamide Chemical compound C=1C=C(NS(=O)(=O)CCC=2C=CC(Cl)=CC=2)C(S(=O)(=O)N)=CC=1OCC1=CC=CC=N1 VMWSNBVSSUDCBY-UHFFFAOYSA-N 0.000 claims description 4
- MKFMGQFLXZONSO-UHFFFAOYSA-N 2-[2-(4-chlorophenyl)ethylsulfonylamino]-5-[(5-methyl-1,3,4-oxadiazol-2-yl)methoxy]benzenesulfonamide Chemical compound O1C(C)=NN=C1COC(C=C1S(N)(=O)=O)=CC=C1NS(=O)(=O)CCC1=CC=C(Cl)C=C1 MKFMGQFLXZONSO-UHFFFAOYSA-N 0.000 claims description 4
- FCMFNLCGOJKUJL-UHFFFAOYSA-N 2-[2-(4-chlorophenyl)ethylsulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)CCC1=CC=C(Cl)C=C1 FCMFNLCGOJKUJL-UHFFFAOYSA-N 0.000 claims description 4
- SVCVCZULARUJMJ-UHFFFAOYSA-N 2-[2-(4-chlorophenyl)propylsulfonylamino]benzenesulfonamide Chemical compound C=1C=C(Cl)C=CC=1C(C)CS(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O SVCVCZULARUJMJ-UHFFFAOYSA-N 0.000 claims description 4
- DFWTWVNQOUUFKR-UHFFFAOYSA-N 2-[2-(4-cyclopentylphenyl)ethylsulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)CCC1=CC=C(C2CCCC2)C=C1 DFWTWVNQOUUFKR-UHFFFAOYSA-N 0.000 claims description 4
- YFWHPOMMWDMSFG-UHFFFAOYSA-N 2-[2-(4-tert-butylphenyl)ethylsulfonylamino]-4-fluorobenzenesulfonamide Chemical compound C1=CC(C(C)(C)C)=CC=C1CCS(=O)(=O)NC1=CC(F)=CC=C1S(N)(=O)=O YFWHPOMMWDMSFG-UHFFFAOYSA-N 0.000 claims description 4
- XDKRKRKYJNVUJW-UHFFFAOYSA-N 2-[2-(4-tert-butylphenyl)ethylsulfonylamino]benzenesulfonamide Chemical compound C1=CC(C(C)(C)C)=CC=C1CCS(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O XDKRKRKYJNVUJW-UHFFFAOYSA-N 0.000 claims description 4
- RTLWKJXLJUKQCS-UHFFFAOYSA-N 2-[2-[4-(2-cyclopentylethynyl)phenyl]ethylsulfonylamino]-5-(hydroxymethyl)benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(CO)=CC=C1NS(=O)(=O)CCC1=CC=C(C#CC2CCCC2)C=C1 RTLWKJXLJUKQCS-UHFFFAOYSA-N 0.000 claims description 4
- RDRNGNJZJAJZEF-UHFFFAOYSA-N 2-[2-[4-(3,3-dimethylbut-1-ynyl)phenyl]ethylsulfonylamino]-5-(hydroxymethyl)benzenesulfonamide Chemical compound C1=CC(C#CC(C)(C)C)=CC=C1CCS(=O)(=O)NC1=CC=C(CO)C=C1S(N)(=O)=O RDRNGNJZJAJZEF-UHFFFAOYSA-N 0.000 claims description 4
- VGBHKDOSWIFJDR-UHFFFAOYSA-N 2-[2-[4-(3,3-dimethylbut-1-ynyl)phenyl]ethylsulfonylamino]-5-methylbenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(C)=CC=C1NS(=O)(=O)CCC1=CC=C(C#CC(C)(C)C)C=C1 VGBHKDOSWIFJDR-UHFFFAOYSA-N 0.000 claims description 4
- FOEXABOANPSPJK-UHFFFAOYSA-N 2-[2-[4-(3,3-dimethylbut-1-ynyl)phenyl]ethylsulfonylamino]benzenesulfonamide Chemical compound C1=CC(C#CC(C)(C)C)=CC=C1CCS(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O FOEXABOANPSPJK-UHFFFAOYSA-N 0.000 claims description 4
- GMMAYPWTUNISFR-UHFFFAOYSA-N 2-[2-[4-(3-hydroxy-3-methylbut-1-ynyl)phenyl]ethylsulfonylamino]benzenesulfonamide Chemical compound C1=CC(C#CC(C)(O)C)=CC=C1CCS(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O GMMAYPWTUNISFR-UHFFFAOYSA-N 0.000 claims description 4
- YKYLTLDRANIQSC-UHFFFAOYSA-N 2-[2-[6-(2-cyclohexylethynyl)pyridin-3-yl]ethylsulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)CCC1=CC=C(C#CC2CCCCC2)N=C1 YKYLTLDRANIQSC-UHFFFAOYSA-N 0.000 claims description 4
- NRYOGTRHDDJCKP-UHFFFAOYSA-N 2-[2-[6-(3,3-dimethylbut-1-ynyl)pyridin-3-yl]ethylsulfonylamino]benzenesulfonamide Chemical compound C1=NC(C#CC(C)(C)C)=CC=C1CCS(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O NRYOGTRHDDJCKP-UHFFFAOYSA-N 0.000 claims description 4
- BIDKABNCGXRVPK-UHFFFAOYSA-N 2-[2-[6-[2-[4-(trifluoromethyl)phenyl]ethynyl]pyridin-3-yl]ethylsulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)CCC1=CC=C(C#CC=2C=CC(=CC=2)C(F)(F)F)N=C1 BIDKABNCGXRVPK-UHFFFAOYSA-N 0.000 claims description 4
- ZKSXEUCSXZHYEO-CMDGGOBGSA-N 2-[[(e)-2-(2,6-dichlorophenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)\C=C\C1=C(Cl)C=CC=C1Cl ZKSXEUCSXZHYEO-CMDGGOBGSA-N 0.000 claims description 4
- PFCRYMZMFRABLG-CMDGGOBGSA-N 2-[[(e)-2-(2,6-difluorophenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)\C=C\C1=C(F)C=CC=C1F PFCRYMZMFRABLG-CMDGGOBGSA-N 0.000 claims description 4
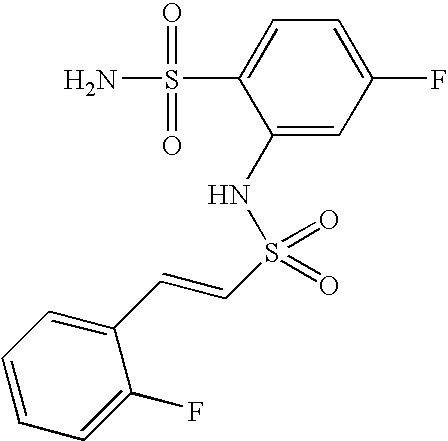
- FSIRJPPWFLKZKJ-MDZDMXLPSA-N 2-[[(e)-2-(2-fluorophenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)\C=C\C1=CC=CC=C1F FSIRJPPWFLKZKJ-MDZDMXLPSA-N 0.000 claims description 4
- LRFVDTORUXWCPF-CSKARUKUSA-N 2-[[(e)-2-(3,4-dichlorophenyl)ethenyl]sulfonylamino]-5-(3-hydroxy-3-methylbut-1-ynyl)benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(C#CC(C)(O)C)=CC=C1NS(=O)(=O)\C=C\C1=CC=C(Cl)C(Cl)=C1 LRFVDTORUXWCPF-CSKARUKUSA-N 0.000 claims description 4
- PRXPZTYRWJJIBS-BQYQJAHWSA-N 2-[[(e)-2-(3-chlorophenyl)ethenyl]sulfonylamino]-3-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC(F)=C1NS(=O)(=O)\C=C\C1=CC=CC(Cl)=C1 PRXPZTYRWJJIBS-BQYQJAHWSA-N 0.000 claims description 4
- LXXXODWLGXFFAC-VOTSOKGWSA-N 2-[[(e)-2-(3-chlorophenyl)ethenyl]sulfonylamino]-5-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(F)=CC=C1NS(=O)(=O)\C=C\C1=CC=CC(Cl)=C1 LXXXODWLGXFFAC-VOTSOKGWSA-N 0.000 claims description 4
- KSXCBPVRMHGLIE-BQYQJAHWSA-N 2-[[(e)-2-(3-chlorophenyl)ethenyl]sulfonylamino]-5-methylbenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(C)=CC=C1NS(=O)(=O)\C=C\C1=CC=CC(Cl)=C1 KSXCBPVRMHGLIE-BQYQJAHWSA-N 0.000 claims description 4
- PFTARWRKFGFEIG-BQYQJAHWSA-N 2-[[(e)-2-(3-chlorophenyl)ethenyl]sulfonylamino]-6-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=C(F)C=CC=C1NS(=O)(=O)\C=C\C1=CC=CC(Cl)=C1 PFTARWRKFGFEIG-BQYQJAHWSA-N 0.000 claims description 4
- HOYVJKRBYPBAIU-BQYQJAHWSA-N 2-[[(e)-2-(4-bromophenyl)ethenyl]sulfonylamino]-4-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=C(F)C=C1NS(=O)(=O)\C=C\C1=CC=C(Br)C=C1 HOYVJKRBYPBAIU-BQYQJAHWSA-N 0.000 claims description 4
- MTNSKMIFAMGTCD-BQYQJAHWSA-N 2-[[(e)-2-(4-bromophenyl)ethenyl]sulfonylamino]-5-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(F)=CC=C1NS(=O)(=O)\C=C\C1=CC=C(Br)C=C1 MTNSKMIFAMGTCD-BQYQJAHWSA-N 0.000 claims description 4
- AOBLVYNWAWKBSO-CMDGGOBGSA-N 2-[[(e)-2-(4-bromophenyl)ethenyl]sulfonylamino]-6-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=C(F)C=CC=C1NS(=O)(=O)\C=C\C1=CC=C(Br)C=C1 AOBLVYNWAWKBSO-CMDGGOBGSA-N 0.000 claims description 4
- JHULIZMVYMKKNQ-CMDGGOBGSA-N 2-[[(e)-2-(4-chlorophenyl)ethenyl]sulfonylamino]-6-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=C(F)C=CC=C1NS(=O)(=O)\C=C\C1=CC=C(Cl)C=C1 JHULIZMVYMKKNQ-CMDGGOBGSA-N 0.000 claims description 4
- KVNSYLGFLWBRFJ-MDZDMXLPSA-N 2-[[(e)-2-(4-chlorophenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)\C=C\C1=CC=C(Cl)C=C1 KVNSYLGFLWBRFJ-MDZDMXLPSA-N 0.000 claims description 4
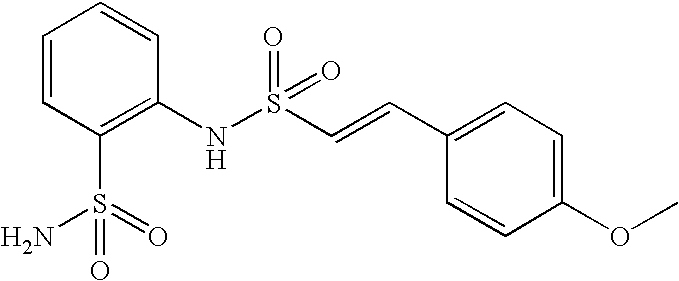
- MAYHYHDIWBDOHI-ZHACJKMWSA-N 2-[[(e)-2-(4-methoxyphenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound C1=CC(OC)=CC=C1\C=C\S(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O MAYHYHDIWBDOHI-ZHACJKMWSA-N 0.000 claims description 4
- LGYHVJIVFAHPIR-ZHACJKMWSA-N 2-[[(e)-2-(4-methylphenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound C1=CC(C)=CC=C1\C=C\S(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O LGYHVJIVFAHPIR-ZHACJKMWSA-N 0.000 claims description 4
- BKBFCBFQMAJTHR-ZHACJKMWSA-N 2-[[(e)-2-(4-methylsulfanylphenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound C1=CC(SC)=CC=C1\C=C\S(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O BKBFCBFQMAJTHR-ZHACJKMWSA-N 0.000 claims description 4
- JROIATIKNCJWJM-MDZDMXLPSA-N 2-[[(e)-2-[2-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)\C=C\C1=CC=CC=C1C(F)(F)F JROIATIKNCJWJM-MDZDMXLPSA-N 0.000 claims description 4
- NUHQMUHSNWIXEV-CMDGGOBGSA-N 2-[[(e)-2-[3-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)\C=C\C1=CC=CC(C(F)(F)F)=C1 NUHQMUHSNWIXEV-CMDGGOBGSA-N 0.000 claims description 4
- CYYTUJYPPFBVLL-FYWRMAATSA-N 2-[[(e)-2-[4-(3,3-dimethylbut-1-ynyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound C1=CC(C#CC(C)(C)C)=CC=C1\C=C\S(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O CYYTUJYPPFBVLL-FYWRMAATSA-N 0.000 claims description 4
- RSBIMJNRILCOKF-WYMLVPIESA-N 2-[[(e)-2-[4-(3-hydroxy-3-methylbut-1-ynyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound C1=CC(C#CC(C)(O)C)=CC=C1\C=C\S(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O RSBIMJNRILCOKF-WYMLVPIESA-N 0.000 claims description 4
- DEGMFLXOCOECJS-BQYQJAHWSA-N 2-[[(e)-2-[4-(difluoromethoxy)phenyl]ethenyl]sulfonylamino]-4-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=C(F)C=C1NS(=O)(=O)\C=C\C1=CC=C(OC(F)F)C=C1 DEGMFLXOCOECJS-BQYQJAHWSA-N 0.000 claims description 4
- RHEJXPRFCTZRMK-BQYQJAHWSA-N 2-[[(e)-2-[4-(difluoromethoxy)phenyl]ethenyl]sulfonylamino]-5-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(F)=CC=C1NS(=O)(=O)\C=C\C1=CC=C(OC(F)F)C=C1 RHEJXPRFCTZRMK-BQYQJAHWSA-N 0.000 claims description 4
- MHIKZSBFPQHBEK-CMDGGOBGSA-N 2-[[(e)-2-[4-(difluoromethoxy)phenyl]ethenyl]sulfonylamino]-6-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=C(F)C=CC=C1NS(=O)(=O)\C=C\C1=CC=C(OC(F)F)C=C1 MHIKZSBFPQHBEK-CMDGGOBGSA-N 0.000 claims description 4
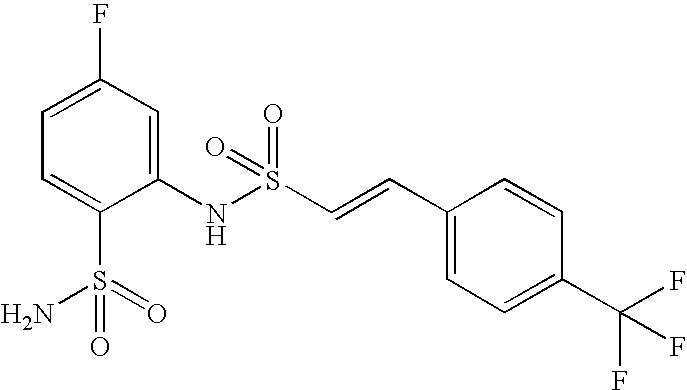
- WHLZXLDTKDAPOF-MDZDMXLPSA-N 2-[[(e)-2-[4-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)\C=C\C1=CC=C(C(F)(F)F)C=C1 WHLZXLDTKDAPOF-MDZDMXLPSA-N 0.000 claims description 4
- HYCPOXDKDLGPGQ-VAWYXSNFSA-N 2-[[(e)-2-naphthalen-2-ylethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)\C=C\C1=CC=C(C=CC=C2)C2=C1 HYCPOXDKDLGPGQ-VAWYXSNFSA-N 0.000 claims description 4
- VPKBTAOMRLZVLN-UHFFFAOYSA-N 2-[[3-(1-benzofuran-2-yl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC(C=2OC3=CC=CC=C3C=2)=C1 VPKBTAOMRLZVLN-UHFFFAOYSA-N 0.000 claims description 4
- AXEQNLURGBKDBD-UHFFFAOYSA-N 2-[[3-(1-methylindol-2-yl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound C=1C2=CC=CC=C2N(C)C=1C(C=1)=CC=CC=1S(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O AXEQNLURGBKDBD-UHFFFAOYSA-N 0.000 claims description 4
- HOTXPWMEDFTQKG-UHFFFAOYSA-N 2-[[3-(1h-indol-5-yl)phenyl]sulfonylamino]benzenesulfonimidic acid Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC(C=2C=C3C=CNC3=CC=2)=C1 HOTXPWMEDFTQKG-UHFFFAOYSA-N 0.000 claims description 4
- MAUILWZKUXYPMS-UHFFFAOYSA-N 2-[[3-(2,3-dichlorophenyl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC(C=2C(=C(Cl)C=CC=2)Cl)=C1 MAUILWZKUXYPMS-UHFFFAOYSA-N 0.000 claims description 4
- XNTVYGTVNNKSPT-UHFFFAOYSA-N 2-[[3-(2,3-dihydro-1-benzofuran-5-yl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC(C=2C=C3CCOC3=CC=2)=C1 XNTVYGTVNNKSPT-UHFFFAOYSA-N 0.000 claims description 4
- BRUXMZBCOXWVKZ-UHFFFAOYSA-N 2-[[3-(2,4-dichlorophenyl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC(C=2C(=CC(Cl)=CC=2)Cl)=C1 BRUXMZBCOXWVKZ-UHFFFAOYSA-N 0.000 claims description 4
- IGOZPIQTCQRCEI-UHFFFAOYSA-N 2-[[3-(2,4-difluorophenyl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC(C=2C(=CC(F)=CC=2)F)=C1 IGOZPIQTCQRCEI-UHFFFAOYSA-N 0.000 claims description 4
- FFJLIRSJLDZHKY-UHFFFAOYSA-N 2-[[3-(2,4-dimethoxypyrimidin-5-yl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound COC1=NC(OC)=NC=C1C1=CC=CC(S(=O)(=O)NC=2C(=CC=CC=2)S(N)(=O)=O)=C1 FFJLIRSJLDZHKY-UHFFFAOYSA-N 0.000 claims description 4
- NDOPMVYGXSPZQU-UHFFFAOYSA-N 2-[[3-(2,5-dichlorophenyl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC(C=2C(=CC=C(Cl)C=2)Cl)=C1 NDOPMVYGXSPZQU-UHFFFAOYSA-N 0.000 claims description 4
- FKEBLYCNBLWJQH-UHFFFAOYSA-N 2-[[3-(2-methoxypyridin-3-yl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound COC1=NC=CC=C1C1=CC=CC(S(=O)(=O)NC=2C(=CC=CC=2)S(N)(=O)=O)=C1 FKEBLYCNBLWJQH-UHFFFAOYSA-N 0.000 claims description 4
- TVAJCUPDVAXVLP-UHFFFAOYSA-N 2-[[3-(2-methoxypyrimidin-5-yl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound C1=NC(OC)=NC=C1C1=CC=CC(S(=O)(=O)NC=2C(=CC=CC=2)S(N)(=O)=O)=C1 TVAJCUPDVAXVLP-UHFFFAOYSA-N 0.000 claims description 4
- NLEIGGGHAMBEGX-UHFFFAOYSA-N 2-[[3-(2-methyl-1,3-thiazol-4-yl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound S1C(C)=NC(C=2C=C(C=CC=2)S(=O)(=O)NC=2C(=CC=CC=2)S(N)(=O)=O)=C1 NLEIGGGHAMBEGX-UHFFFAOYSA-N 0.000 claims description 4
- UYMBNGIJDCRCQM-UHFFFAOYSA-N 2-[[3-(3,3-dimethylbut-1-ynyl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound CC(C)(C)C#CC1=CC=CC(S(=O)(=O)NC=2C(=CC=CC=2)S(N)(=O)=O)=C1 UYMBNGIJDCRCQM-UHFFFAOYSA-N 0.000 claims description 4
- AZNAGQOXVYSLGD-UHFFFAOYSA-N 2-[[3-(3,4-dichlorophenyl)phenyl]sulfonylamino]-5-phenoxybenzenesulfonamide Chemical compound C=1C=C(NS(=O)(=O)C=2C=C(C=CC=2)C=2C=C(Cl)C(Cl)=CC=2)C(S(=O)(=O)N)=CC=1OC1=CC=CC=C1 AZNAGQOXVYSLGD-UHFFFAOYSA-N 0.000 claims description 4
- IFLNYTQBXFZJKX-UHFFFAOYSA-N 2-[[3-(3,4-dichlorophenyl)phenyl]sulfonylamino]-5-phenylbenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(C=2C=CC=CC=2)=CC=C1NS(=O)(=O)C(C=1)=CC=CC=1C1=CC=C(Cl)C(Cl)=C1 IFLNYTQBXFZJKX-UHFFFAOYSA-N 0.000 claims description 4
- YIIIGFOFCJCKBI-UHFFFAOYSA-N 2-[[3-(3,4-dichlorophenyl)phenyl]sulfonylamino]-n-methylbenzenesulfonamide Chemical compound CNS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC(C=2C=C(Cl)C(Cl)=CC=2)=C1 YIIIGFOFCJCKBI-UHFFFAOYSA-N 0.000 claims description 4
- NWFQUOGEVIWGPG-UHFFFAOYSA-N 2-[[3-(3,4-dichlorophenyl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC(C=2C=C(Cl)C(Cl)=CC=2)=C1 NWFQUOGEVIWGPG-UHFFFAOYSA-N 0.000 claims description 4
- IPBDOFHBXTVDPD-UHFFFAOYSA-N 2-[[3-(3,5-difluorophenyl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC(C=2C=C(F)C=C(F)C=2)=C1 IPBDOFHBXTVDPD-UHFFFAOYSA-N 0.000 claims description 4
- UNZUEWWVCCSYKB-UHFFFAOYSA-N 2-[[3-(3-cyanophenyl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC(C=2C=C(C=CC=2)C#N)=C1 UNZUEWWVCCSYKB-UHFFFAOYSA-N 0.000 claims description 4
- ASTRFLZRWGTIAX-UHFFFAOYSA-N 2-[[3-(3-methoxyphenyl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound COC1=CC=CC(C=2C=C(C=CC=2)S(=O)(=O)NC=2C(=CC=CC=2)S(N)(=O)=O)=C1 ASTRFLZRWGTIAX-UHFFFAOYSA-N 0.000 claims description 4
- QCPPYETXSAOFNW-UHFFFAOYSA-N 2-[[3-(4-cyanophenyl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC(C=2C=CC(=CC=2)C#N)=C1 QCPPYETXSAOFNW-UHFFFAOYSA-N 0.000 claims description 4
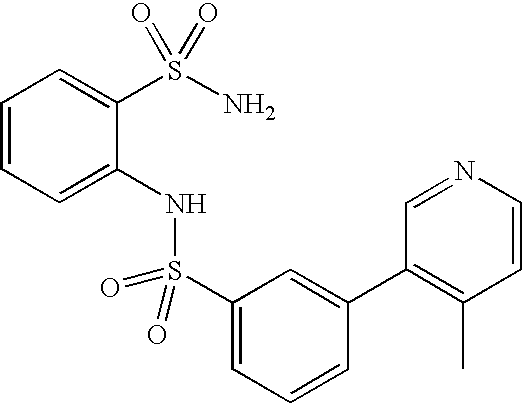
- GDNJVCUHTQDFMS-UHFFFAOYSA-N 2-[[3-(4-methylpyridin-3-yl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound CC1=CC=NC=C1C1=CC=CC(S(=O)(=O)NC=2C(=CC=CC=2)S(N)(=O)=O)=C1 GDNJVCUHTQDFMS-UHFFFAOYSA-N 0.000 claims description 4
- ZIBUOVUBBSDTAG-UHFFFAOYSA-N 2-[[3-(5-chloro-2-methoxyphenyl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound COC1=CC=C(Cl)C=C1C1=CC=CC(S(=O)(=O)NC=2C(=CC=CC=2)S(N)(=O)=O)=C1 ZIBUOVUBBSDTAG-UHFFFAOYSA-N 0.000 claims description 4
- WVXMZKAIOMLXRH-UHFFFAOYSA-N 2-[[3-(5-chloro-6-methoxypyridin-3-yl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound C1=C(Cl)C(OC)=NC=C1C1=CC=CC(S(=O)(=O)NC=2C(=CC=CC=2)S(N)(=O)=O)=C1 WVXMZKAIOMLXRH-UHFFFAOYSA-N 0.000 claims description 4
- IUVJDPUEHUKSQZ-UHFFFAOYSA-N 2-[[3-(5-fluoro-6-methoxypyridin-3-yl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound C1=C(F)C(OC)=NC=C1C1=CC=CC(S(=O)(=O)NC=2C(=CC=CC=2)S(N)(=O)=O)=C1 IUVJDPUEHUKSQZ-UHFFFAOYSA-N 0.000 claims description 4
- ZBDYJHVPWQTBTB-UHFFFAOYSA-N 2-[[3-(6-methoxypyridin-2-yl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound COC1=CC=CC(C=2C=C(C=CC=2)S(=O)(=O)NC=2C(=CC=CC=2)S(N)(=O)=O)=N1 ZBDYJHVPWQTBTB-UHFFFAOYSA-N 0.000 claims description 4
- GPLVRZXLCTYXQU-UHFFFAOYSA-N 2-[[3-(6-methoxypyridin-3-yl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound C1=NC(OC)=CC=C1C1=CC=CC(S(=O)(=O)NC=2C(=CC=CC=2)S(N)(=O)=O)=C1 GPLVRZXLCTYXQU-UHFFFAOYSA-N 0.000 claims description 4
- NSLHRYXEGZHKLF-UHFFFAOYSA-N 2-[[3-(furan-3-yl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC(C2=COC=C2)=C1 NSLHRYXEGZHKLF-UHFFFAOYSA-N 0.000 claims description 4
- FFSWWBCBRBSNIN-UHFFFAOYSA-N 2-[[3-[3-(hydroxymethyl)phenyl]phenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC(C=2C=C(CO)C=CC=2)=C1 FFSWWBCBRBSNIN-UHFFFAOYSA-N 0.000 claims description 4
- KEHHVWLUUPMVKF-UHFFFAOYSA-N 2-[[3-[3-(trifluoromethoxy)phenyl]phenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC(C=2C=C(OC(F)(F)F)C=CC=2)=C1 KEHHVWLUUPMVKF-UHFFFAOYSA-N 0.000 claims description 4
- HIHARMTZACXORE-UHFFFAOYSA-N 2-[[3-[3-(trifluoromethyl)phenyl]phenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC(C=2C=C(C=CC=2)C(F)(F)F)=C1 HIHARMTZACXORE-UHFFFAOYSA-N 0.000 claims description 4
- NXTVGFGFUQUUQJ-UHFFFAOYSA-N 2-[[3-[4-(hydroxymethyl)phenyl]phenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC(C=2C=CC(CO)=CC=2)=C1 NXTVGFGFUQUUQJ-UHFFFAOYSA-N 0.000 claims description 4
- BFGIWJCXRSZXEX-UHFFFAOYSA-N 2-[[3-[4-(trifluoromethyl)phenyl]phenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC(C=2C=CC(=CC=2)C(F)(F)F)=C1 BFGIWJCXRSZXEX-UHFFFAOYSA-N 0.000 claims description 4
- YFKPHJOWQCHRTC-UHFFFAOYSA-N 2-[[3-[6-(dimethylamino)pyridin-3-yl]phenyl]sulfonylamino]benzenesulfonamide Chemical compound C1=NC(N(C)C)=CC=C1C1=CC=CC(S(=O)(=O)NC=2C(=CC=CC=2)S(N)(=O)=O)=C1 YFKPHJOWQCHRTC-UHFFFAOYSA-N 0.000 claims description 4
- NJWIHJVBNIAIOH-UHFFFAOYSA-N 2-[[4-(1-benzofuran-2-yl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=C(C=2OC3=CC=CC=C3C=2)C=C1 NJWIHJVBNIAIOH-UHFFFAOYSA-N 0.000 claims description 4
- OTGMCQXZMNGHCS-UHFFFAOYSA-N 2-[[4-(3,4-dichlorophenyl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=C(C=2C=C(Cl)C(Cl)=CC=2)C=C1 OTGMCQXZMNGHCS-UHFFFAOYSA-N 0.000 claims description 4
- ABBGTBRNVJKHDD-UHFFFAOYSA-N 2-[[4-(4-chlorophenoxy)phenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C(C=C1)=CC=C1OC1=CC=C(Cl)C=C1 ABBGTBRNVJKHDD-UHFFFAOYSA-N 0.000 claims description 4
- HLDWKHZNPUNPCQ-UHFFFAOYSA-N 2-[[4-(trifluoromethyl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=C(C(F)(F)F)C=C1 HLDWKHZNPUNPCQ-UHFFFAOYSA-N 0.000 claims description 4
- BAFDLWVPOWXPTR-UHFFFAOYSA-N 2-[[4-[3-chloro-5-(trifluoromethyl)pyridin-2-yl]oxyphenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C(C=C1)=CC=C1OC1=NC=C(C(F)(F)F)C=C1Cl BAFDLWVPOWXPTR-UHFFFAOYSA-N 0.000 claims description 4
- HVOQTXHUIVBSHG-CMDGGOBGSA-N 2-fluoro-6-[[(e)-2-(2-fluorophenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=C(F)C=CC=C1NS(=O)(=O)\C=C\C1=CC=CC=C1F HVOQTXHUIVBSHG-CMDGGOBGSA-N 0.000 claims description 4
- SXOSYTOXUGKPMP-MDZDMXLPSA-N 2-fluoro-6-[[(e)-2-(2-methoxyphenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound COC1=CC=CC=C1\C=C\S(=O)(=O)NC1=CC=CC(F)=C1S(N)(=O)=O SXOSYTOXUGKPMP-MDZDMXLPSA-N 0.000 claims description 4
- JPFVUNRUSHOLFE-MDZDMXLPSA-N 2-fluoro-6-[[(e)-2-(4-methylsulfanylphenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound C1=CC(SC)=CC=C1\C=C\S(=O)(=O)NC1=CC=CC(F)=C1S(N)(=O)=O JPFVUNRUSHOLFE-MDZDMXLPSA-N 0.000 claims description 4
- SCCYHNSRBRDKHV-BUHFOSPRSA-N 2-fluoro-6-[[(e)-2-(4-phenylphenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=C(F)C=CC=C1NS(=O)(=O)\C=C\C1=CC=C(C=2C=CC=CC=2)C=C1 SCCYHNSRBRDKHV-BUHFOSPRSA-N 0.000 claims description 4
- ZPKKJTVYIGNFSV-CMDGGOBGSA-N 2-fluoro-6-[[(e)-2-[2-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=C(F)C=CC=C1NS(=O)(=O)\C=C\C1=CC=CC=C1C(F)(F)F ZPKKJTVYIGNFSV-CMDGGOBGSA-N 0.000 claims description 4
- VIFIWJGHCRMAKZ-BQYQJAHWSA-N 2-fluoro-6-[[(e)-2-[3-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=C(F)C=CC=C1NS(=O)(=O)\C=C\C1=CC=CC(C(F)(F)F)=C1 VIFIWJGHCRMAKZ-BQYQJAHWSA-N 0.000 claims description 4
- MHXHQGAFZMTQCQ-CMDGGOBGSA-N 2-fluoro-6-[[(e)-2-[4-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=C(F)C=CC=C1NS(=O)(=O)\C=C\C1=CC=C(C(F)(F)F)C=C1 MHXHQGAFZMTQCQ-CMDGGOBGSA-N 0.000 claims description 4
- WQGBYKFFCVZDJA-ZHACJKMWSA-N 2-fluoro-6-[[(e)-2-naphthalen-2-ylethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=C(F)C=CC=C1NS(=O)(=O)\C=C\C1=CC=C(C=CC=C2)C2=C1 WQGBYKFFCVZDJA-ZHACJKMWSA-N 0.000 claims description 4
- UKVZXVOXIYNYEM-UHFFFAOYSA-N 2-n-methyl-5,5-dioxo-8-n-(2-sulfamoylphenyl)dibenzothiophene-2,8-disulfonamide Chemical compound C=1C(S(=O)(=O)NC)=CC=C(S(C2=CC=3)(=O)=O)C=1C2=CC=3S(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O UKVZXVOXIYNYEM-UHFFFAOYSA-N 0.000 claims description 4
- MONJCWNWWACJNL-UHFFFAOYSA-N 3,4-dichloro-n-(2-sulfamoylphenyl)benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=C(Cl)C(Cl)=C1 MONJCWNWWACJNL-UHFFFAOYSA-N 0.000 claims description 4
- PATDHLLXCTVXSL-UHFFFAOYSA-N 3-(benzenesulfonamido)pyridine-2-sulfonamide Chemical compound NS(=O)(=O)C1=NC=CC=C1NS(=O)(=O)C1=CC=CC=C1 PATDHLLXCTVXSL-UHFFFAOYSA-N 0.000 claims description 4
- XRHUALFDDSZNQB-UHFFFAOYSA-N 3-[2-[4-(3,3-dimethylbut-1-ynyl)phenyl]ethylsulfonylamino]thiophene-2-sulfonamide Chemical compound C1=CC(C#CC(C)(C)C)=CC=C1CCS(=O)(=O)NC1=C(S(N)(=O)=O)SC=C1 XRHUALFDDSZNQB-UHFFFAOYSA-N 0.000 claims description 4
- HWRCYKDMQBYLOC-UHFFFAOYSA-N 3-[[3-(5-chloro-6-methoxypyridin-3-yl)phenyl]sulfonylamino]thiophene-2-sulfonamide Chemical compound C1=C(Cl)C(OC)=NC=C1C1=CC=CC(S(=O)(=O)NC2=C(SC=C2)S(N)(=O)=O)=C1 HWRCYKDMQBYLOC-UHFFFAOYSA-N 0.000 claims description 4
- RKRXADYMGOLJCR-UHFFFAOYSA-N 3-amino-4-hydroxy-n-(2-sulfamoylphenyl)benzenesulfonamide Chemical compound C1=C(O)C(N)=CC(S(=O)(=O)NC=2C(=CC=CC=2)S(N)(=O)=O)=C1 RKRXADYMGOLJCR-UHFFFAOYSA-N 0.000 claims description 4
- OSYGCQSMZXIRMT-UHFFFAOYSA-N 3-chloro-2-fluoro-n-(2-sulfamoylphenyl)benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC(Cl)=C1F OSYGCQSMZXIRMT-UHFFFAOYSA-N 0.000 claims description 4
- LWCOQNYUXXLDIE-MDZDMXLPSA-N 3-fluoro-2-[[(e)-2-(2-methoxyphenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound COC1=CC=CC=C1\C=C\S(=O)(=O)NC1=C(F)C=CC=C1S(N)(=O)=O LWCOQNYUXXLDIE-MDZDMXLPSA-N 0.000 claims description 4
- UGQNFDPSCUJAND-BUHFOSPRSA-N 3-fluoro-2-[[(e)-2-(4-phenylphenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC(F)=C1NS(=O)(=O)\C=C\C1=CC=C(C=2C=CC=CC=2)C=C1 UGQNFDPSCUJAND-BUHFOSPRSA-N 0.000 claims description 4
- MEPCNOKPFQVUEG-BQYQJAHWSA-N 3-fluoro-2-[[(e)-2-[3-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC(F)=C1NS(=O)(=O)\C=C\C1=CC=CC(C(F)(F)F)=C1 MEPCNOKPFQVUEG-BQYQJAHWSA-N 0.000 claims description 4
- GHHIWTKGUAFFNW-UHFFFAOYSA-N 4,5-dichloro-n-(2-sulfamoylphenyl)thiophene-2-sulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC(Cl)=C(Cl)S1 GHHIWTKGUAFFNW-UHFFFAOYSA-N 0.000 claims description 4
- IRODHXJPBUOFOH-UHFFFAOYSA-N 4-[2-(4-chloro-2-methoxyphenyl)ethylsulfonylamino]pyridine-3-sulfonamide Chemical compound COC1=CC(Cl)=CC=C1CCS(=O)(=O)NC1=CC=NC=C1S(N)(=O)=O IRODHXJPBUOFOH-UHFFFAOYSA-N 0.000 claims description 4
- BHWVTEQYWFKUFJ-UHFFFAOYSA-N 4-[2-[4-(1-benzofuran-2-yl)phenyl]ethylsulfonylamino]pyridine-3-sulfonamide Chemical compound NS(=O)(=O)C1=CN=CC=C1NS(=O)(=O)CCC1=CC=C(C=2OC3=CC=CC=C3C=2)C=C1 BHWVTEQYWFKUFJ-UHFFFAOYSA-N 0.000 claims description 4
- AJYPNMPWEAMMQR-UHFFFAOYSA-N 4-[[3-(3,4-dichlorophenyl)phenyl]sulfonylamino]-3-sulfamoylbenzamide Chemical compound NS(=O)(=O)C1=CC(C(=O)N)=CC=C1NS(=O)(=O)C1=CC=CC(C=2C=C(Cl)C(Cl)=CC=2)=C1 AJYPNMPWEAMMQR-UHFFFAOYSA-N 0.000 claims description 4
- VJNVCJXRBPUNAM-UHFFFAOYSA-N 4-[[3-(5-chloro-6-methoxypyridin-3-yl)phenyl]sulfonylamino]pyridine-3-sulfonamide Chemical compound C1=C(Cl)C(OC)=NC=C1C1=CC=CC(S(=O)(=O)NC=2C(=CN=CC=2)S(N)(=O)=O)=C1 VJNVCJXRBPUNAM-UHFFFAOYSA-N 0.000 claims description 4
- QQFDTOOHEWWDFO-UHFFFAOYSA-N 4-chloro-2-[2-(2-methoxyphenyl)ethylsulfonylamino]benzenesulfonamide Chemical compound COC1=CC=CC=C1CCS(=O)(=O)NC1=CC(Cl)=CC=C1S(N)(=O)=O QQFDTOOHEWWDFO-UHFFFAOYSA-N 0.000 claims description 4
- KERGBMOKJBJGJS-UHFFFAOYSA-N 4-chloro-2-[2-(3,4-dichlorophenyl)ethylsulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=C(Cl)C=C1NS(=O)(=O)CCC1=CC=C(Cl)C(Cl)=C1 KERGBMOKJBJGJS-UHFFFAOYSA-N 0.000 claims description 4
- CYTHDUWTVSBZDS-UHFFFAOYSA-N 4-chloro-2-[2-(4-chloro-2-methoxyphenyl)ethylsulfonylamino]benzenesulfonamide Chemical compound COC1=CC(Cl)=CC=C1CCS(=O)(=O)NC1=CC(Cl)=CC=C1S(N)(=O)=O CYTHDUWTVSBZDS-UHFFFAOYSA-N 0.000 claims description 4
- JOSHCJKFUNAZAS-UHFFFAOYSA-N 4-chloro-2-[2-[4-(difluoromethoxy)phenyl]ethylsulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=C(Cl)C=C1NS(=O)(=O)CCC1=CC=C(OC(F)F)C=C1 JOSHCJKFUNAZAS-UHFFFAOYSA-N 0.000 claims description 4
- VYUVWHYOLSBKIV-UHFFFAOYSA-N 4-cyano-2-[2-(3,4-dichlorophenyl)ethylsulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=C(C#N)C=C1NS(=O)(=O)CCC1=CC=C(Cl)C(Cl)=C1 VYUVWHYOLSBKIV-UHFFFAOYSA-N 0.000 claims description 4
- JNOQAKBQORYZSW-UHFFFAOYSA-N 4-fluoro-2-(2-naphthalen-2-ylethylsulfonylamino)benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=C(F)C=C1NS(=O)(=O)CCC1=CC=C(C=CC=C2)C2=C1 JNOQAKBQORYZSW-UHFFFAOYSA-N 0.000 claims description 4
- WHWNKOZGHXSTIK-BQYQJAHWSA-N 4-fluoro-2-[[(e)-2-(2-fluorophenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=C(F)C=C1NS(=O)(=O)\C=C\C1=CC=CC=C1F WHWNKOZGHXSTIK-BQYQJAHWSA-N 0.000 claims description 4
- IPXVRODNSDMLRN-CMDGGOBGSA-N 4-fluoro-2-[[(e)-2-(2-methoxyphenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound COC1=CC=CC=C1\C=C\S(=O)(=O)NC1=CC(F)=CC=C1S(N)(=O)=O IPXVRODNSDMLRN-CMDGGOBGSA-N 0.000 claims description 4
- MPLJFUGYPPMSSD-OUKQBFOZSA-N 4-fluoro-2-[[(e)-2-(4-phenylphenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=C(F)C=C1NS(=O)(=O)\C=C\C1=CC=C(C=2C=CC=CC=2)C=C1 MPLJFUGYPPMSSD-OUKQBFOZSA-N 0.000 claims description 4
- XQUCGVHWRTZAFB-BQYQJAHWSA-N 4-fluoro-2-[[(e)-2-[2-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=C(F)C=C1NS(=O)(=O)\C=C\C1=CC=CC=C1C(F)(F)F XQUCGVHWRTZAFB-BQYQJAHWSA-N 0.000 claims description 4
- IRVLPZGJFTWTCD-VOTSOKGWSA-N 4-fluoro-2-[[(e)-2-[3-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=C(F)C=C1NS(=O)(=O)\C=C\C1=CC=CC(C(F)(F)F)=C1 IRVLPZGJFTWTCD-VOTSOKGWSA-N 0.000 claims description 4
- JOORWSAPATXFFR-BQYQJAHWSA-N 4-fluoro-2-[[(e)-2-[4-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=C(F)C=C1NS(=O)(=O)\C=C\C1=CC=C(C(F)(F)F)C=C1 JOORWSAPATXFFR-BQYQJAHWSA-N 0.000 claims description 4
- LZFJNDKMICVCEA-MDZDMXLPSA-N 4-fluoro-2-[[(e)-2-naphthalen-2-ylethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=C(F)C=C1NS(=O)(=O)\C=C\C1=CC=C(C=CC=C2)C2=C1 LZFJNDKMICVCEA-MDZDMXLPSA-N 0.000 claims description 4
- BHBGPLKXJMQYSM-UHFFFAOYSA-N 4-methyl-n-(2-sulfamoylphenyl)-2,3-dihydro-1,4-benzoxazine-7-sulfonamide Chemical compound C=1C=C2N(C)CCOC2=CC=1S(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O BHBGPLKXJMQYSM-UHFFFAOYSA-N 0.000 claims description 4
- LIEOFMOIVIYZGG-UHFFFAOYSA-N 5-[1-methyl-5-(trifluoromethyl)pyrazol-3-yl]-n-(2-sulfamoylphenyl)thiophene-2-sulfonamide Chemical compound C1=C(C(F)(F)F)N(C)N=C1C1=CC=C(S(=O)(=O)NC=2C(=CC=CC=2)S(N)(=O)=O)S1 LIEOFMOIVIYZGG-UHFFFAOYSA-N 0.000 claims description 4
- UVTNMELHHFGQIB-UHFFFAOYSA-N 5-[2-(3,4-dichlorophenyl)ethylsulfonylamino]-2,3-dihydro-1h-indene-4-sulfonamide Chemical compound NS(=O)(=O)C1=C2CCCC2=CC=C1NS(=O)(=O)CCC1=CC=C(Cl)C(Cl)=C1 UVTNMELHHFGQIB-UHFFFAOYSA-N 0.000 claims description 4
- NGTLUSPFNXFAIH-UHFFFAOYSA-N 5-[2-[4-(furan-2-yl)phenyl]ethylsulfonylamino]-2,3-dihydro-1h-indene-4-sulfonamide Chemical compound NS(=O)(=O)C1=C2CCCC2=CC=C1NS(=O)(=O)CCC(C=C1)=CC=C1C1=CC=CO1 NGTLUSPFNXFAIH-UHFFFAOYSA-N 0.000 claims description 4
- WESBVZHTGWTWDV-ZHACJKMWSA-N 5-[[(e)-2-(4-chlorophenyl)ethenyl]sulfonylamino]-2,3-dihydro-1h-indene-4-sulfonamide Chemical compound NS(=O)(=O)C1=C2CCCC2=CC=C1NS(=O)(=O)\C=C\C1=CC=C(Cl)C=C1 WESBVZHTGWTWDV-ZHACJKMWSA-N 0.000 claims description 4
- GPEPULYVRCYIHI-UHFFFAOYSA-N 5-chloro-2-[2-(3,4-dichlorophenyl)ethylsulfonylamino]-4-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(Cl)=C(F)C=C1NS(=O)(=O)CCC1=CC=C(Cl)C(Cl)=C1 GPEPULYVRCYIHI-UHFFFAOYSA-N 0.000 claims description 4
- SNHJGTNTGPDKQS-UHFFFAOYSA-N 5-chloro-2-[2-(3,4-dichlorophenyl)ethylsulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(Cl)=CC=C1NS(=O)(=O)CCC1=CC=C(Cl)C(Cl)=C1 SNHJGTNTGPDKQS-UHFFFAOYSA-N 0.000 claims description 4
- QFWRUMUZPWOHPZ-UHFFFAOYSA-N 5-chloro-2-[2-(4-chloro-2-methoxyphenyl)ethylsulfonylamino]benzenesulfonamide Chemical compound COC1=CC(Cl)=CC=C1CCS(=O)(=O)NC1=CC=C(Cl)C=C1S(N)(=O)=O QFWRUMUZPWOHPZ-UHFFFAOYSA-N 0.000 claims description 4
- ACRPAVGELMUKKQ-UHFFFAOYSA-N 5-chloro-2-[2-(4-chlorophenyl)ethylsulfonylamino]-4-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(Cl)=C(F)C=C1NS(=O)(=O)CCC1=CC=C(Cl)C=C1 ACRPAVGELMUKKQ-UHFFFAOYSA-N 0.000 claims description 4
- RZFHXJOYEOFMNS-UHFFFAOYSA-N 5-chloro-n-(2-sulfamoylphenyl)thiophene-2-sulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=C(Cl)S1 RZFHXJOYEOFMNS-UHFFFAOYSA-N 0.000 claims description 4
- PRQQCUZLGGHJFA-UHFFFAOYSA-N 5-cyano-2-[2-(3,4-dichlorophenyl)ethylsulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(C#N)=CC=C1NS(=O)(=O)CCC1=CC=C(Cl)C(Cl)=C1 PRQQCUZLGGHJFA-UHFFFAOYSA-N 0.000 claims description 4
- LCZFMSKPYIDKBC-CMDGGOBGSA-N 5-fluoro-2-[[(e)-2-(2-methoxyphenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound COC1=CC=CC=C1\C=C\S(=O)(=O)NC1=CC=C(F)C=C1S(N)(=O)=O LCZFMSKPYIDKBC-CMDGGOBGSA-N 0.000 claims description 4
- OJWFMDJRUVBVRR-OUKQBFOZSA-N 5-fluoro-2-[[(e)-2-(4-phenylphenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(F)=CC=C1NS(=O)(=O)\C=C\C1=CC=C(C=2C=CC=CC=2)C=C1 OJWFMDJRUVBVRR-OUKQBFOZSA-N 0.000 claims description 4
- HMPCUALYVXNBHH-BQYQJAHWSA-N 5-fluoro-2-[[(e)-2-[2-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(F)=CC=C1NS(=O)(=O)\C=C\C1=CC=CC=C1C(F)(F)F HMPCUALYVXNBHH-BQYQJAHWSA-N 0.000 claims description 4
- HTMXSWLTLMOZSR-VOTSOKGWSA-N 5-fluoro-2-[[(e)-2-[3-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(F)=CC=C1NS(=O)(=O)\C=C\C1=CC=CC(C(F)(F)F)=C1 HTMXSWLTLMOZSR-VOTSOKGWSA-N 0.000 claims description 4
- NBSJYIDCWGJSJH-BQYQJAHWSA-N 5-fluoro-2-[[(e)-2-[4-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(F)=CC=C1NS(=O)(=O)\C=C\C1=CC=C(C(F)(F)F)C=C1 NBSJYIDCWGJSJH-BQYQJAHWSA-N 0.000 claims description 4
- NQEKXVMPNFXWRW-MDZDMXLPSA-N 5-fluoro-2-[[(e)-2-naphthalen-2-ylethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(F)=CC=C1NS(=O)(=O)\C=C\C1=CC=C(C=CC=C2)C2=C1 NQEKXVMPNFXWRW-MDZDMXLPSA-N 0.000 claims description 4
- JUTNZTKVJBJSPG-BQYQJAHWSA-N 5-methyl-2-[[(e)-2-[3-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(C)=CC=C1NS(=O)(=O)\C=C\C1=CC=CC(C(F)(F)F)=C1 JUTNZTKVJBJSPG-BQYQJAHWSA-N 0.000 claims description 4
- RUICTWDYCKTARZ-CMDGGOBGSA-N 5-methyl-2-[[(e)-2-[4-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(C)=CC=C1NS(=O)(=O)\C=C\C1=CC=C(C(F)(F)F)C=C1 RUICTWDYCKTARZ-CMDGGOBGSA-N 0.000 claims description 4
- GDDJPJCLUOQVHW-ZHACJKMWSA-N 5-methyl-2-[[(e)-2-naphthalen-2-ylethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(C)=CC=C1NS(=O)(=O)\C=C\C1=CC=C(C=CC=C2)C2=C1 GDDJPJCLUOQVHW-ZHACJKMWSA-N 0.000 claims description 4
- SMWQWMWWLRBXBX-UHFFFAOYSA-N 5-methyl-2-[[3-[3-(trifluoromethoxy)phenyl]phenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(C)=CC=C1NS(=O)(=O)C1=CC=CC(C=2C=C(OC(F)(F)F)C=CC=2)=C1 SMWQWMWWLRBXBX-UHFFFAOYSA-N 0.000 claims description 4
- VSPPINFJJYISGF-UHFFFAOYSA-N 5-phenoxy-2-[[3-[3-(trifluoromethoxy)phenyl]phenyl]sulfonylamino]benzenesulfonamide Chemical compound C=1C=C(NS(=O)(=O)C=2C=C(C=CC=2)C=2C=C(OC(F)(F)F)C=CC=2)C(S(=O)(=O)N)=CC=1OC1=CC=CC=C1 VSPPINFJJYISGF-UHFFFAOYSA-N 0.000 claims description 4
- TULPKMPSRGSYIM-UHFFFAOYSA-N 6-[2-[4-(furan-2-yl)phenyl]ethylsulfonylamino]-2,3-dihydro-1h-indene-5-sulfonamide Chemical compound NS(=O)(=O)C1=CC=2CCCC=2C=C1NS(=O)(=O)CCC(C=C1)=CC=C1C1=CC=CO1 TULPKMPSRGSYIM-UHFFFAOYSA-N 0.000 claims description 4
- SYEPGKTXIPHAIJ-CMDGGOBGSA-N 6-[[(e)-2-(4-chlorophenyl)ethenyl]sulfonylamino]-2,3-dihydro-1h-indene-5-sulfonamide Chemical compound NS(=O)(=O)C1=CC=2CCCC=2C=C1NS(=O)(=O)\C=C\C1=CC=C(Cl)C=C1 SYEPGKTXIPHAIJ-CMDGGOBGSA-N 0.000 claims description 4
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 claims description 4
- 125000002816 methylsulfanyl group Chemical group [H]C([H])([H])S[*] 0.000 claims description 4
- HXSKKQWQFMFQST-UHFFFAOYSA-N n-(2-sulfamoylphenyl)-1,2,3,4-tetrahydronaphthalene-2-sulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1CC2=CC=CC=C2CC1 HXSKKQWQFMFQST-UHFFFAOYSA-N 0.000 claims description 4
- FQJQZYXRBONFSE-UHFFFAOYSA-N n-(2-sulfamoylphenyl)-2,3-dihydro-1h-indene-5-sulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=C(CCC2)C2=C1 FQJQZYXRBONFSE-UHFFFAOYSA-N 0.000 claims description 4
- 125000006296 sulfonyl amino group Chemical group [H]N(*)S(*)(=O)=O 0.000 claims description 4
- FMVDEWWWQXDCSW-UHFFFAOYSA-N 2,3,4,5,6-pentafluoro-n-(2-sulfamoylphenyl)benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=C(F)C(F)=C(F)C(F)=C1F FMVDEWWWQXDCSW-UHFFFAOYSA-N 0.000 claims description 3
- DYNYBEKITWMKLF-UHFFFAOYSA-N 2,5-dibromo-n-[2-(ethylsulfamoyl)phenyl]benzenesulfonamide Chemical compound CCNS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC(Br)=CC=C1Br DYNYBEKITWMKLF-UHFFFAOYSA-N 0.000 claims description 3
- SZRJALUZWCATBB-UHFFFAOYSA-N 2-[2-[4-(1,1,2,2-tetrafluoroethoxy)phenyl]ethylsulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)CCC1=CC=C(OC(F)(F)C(F)F)C=C1 SZRJALUZWCATBB-UHFFFAOYSA-N 0.000 claims description 3
- ISNPAGGMEVRVGU-UHFFFAOYSA-N 2-[2-[4-[4-(trifluoromethyl)phenyl]phenyl]ethylsulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)CCC1=CC=C(C=2C=CC(=CC=2)C(F)(F)F)C=C1 ISNPAGGMEVRVGU-UHFFFAOYSA-N 0.000 claims description 3
- CMBFXOIUIXAOQN-VAWYXSNFSA-N 2-[[(e)-2-(2-ethoxyphenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound CCOC1=CC=CC=C1\C=C\S(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O CMBFXOIUIXAOQN-VAWYXSNFSA-N 0.000 claims description 3
- VSFZNUXMYZEATD-ZHACJKMWSA-N 2-[[(e)-2-(2-methylsulfanylphenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound CSC1=CC=CC=C1\C=C\S(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O VSFZNUXMYZEATD-ZHACJKMWSA-N 0.000 claims description 3
- UQMUMQWNKWOKTG-VAWYXSNFSA-N 2-[[(e)-2-(2-propan-2-yloxyphenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound CC(C)OC1=CC=CC=C1\C=C\S(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O UQMUMQWNKWOKTG-VAWYXSNFSA-N 0.000 claims description 3
- CQCPXFXCGALQJM-MDZDMXLPSA-N 2-[[(e)-2-[2-(difluoromethoxy)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)\C=C\C1=CC=CC=C1OC(F)F CQCPXFXCGALQJM-MDZDMXLPSA-N 0.000 claims description 3
- IZQFFBFULSOYNB-MDZDMXLPSA-N 2-[[(e)-2-[2-(trifluoromethoxy)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)\C=C\C1=CC=CC=C1OC(F)(F)F IZQFFBFULSOYNB-MDZDMXLPSA-N 0.000 claims description 3
- BKZLGDOHABYLAO-MDZDMXLPSA-N 2-[[(e)-2-[4-(1,1,2,2-tetrafluoroethoxy)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)\C=C\C1=CC=C(OC(F)(F)C(F)F)C=C1 BKZLGDOHABYLAO-MDZDMXLPSA-N 0.000 claims description 3
- CBXRXUPDVFENDR-BUHFOSPRSA-N 2-[[(e)-2-[4-[4-(trifluoromethyl)phenyl]phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)\C=C\C1=CC=C(C=2C=CC(=CC=2)C(F)(F)F)C=C1 CBXRXUPDVFENDR-BUHFOSPRSA-N 0.000 claims description 3
- OCWFNUPEEXDUNG-UHFFFAOYSA-N 2-[[4-[(2-chloro-1,3-thiazol-5-yl)methoxy]phenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C(C=C1)=CC=C1OCC1=CN=C(Cl)S1 OCWFNUPEEXDUNG-UHFFFAOYSA-N 0.000 claims description 3
- XQVVQJGSNAKPKT-UHFFFAOYSA-N 4-tert-butyl-n-[2-(ethylsulfamoyl)phenyl]-2,6-dimethylbenzenesulfonamide Chemical compound CCNS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=C(C)C=C(C(C)(C)C)C=C1C XQVVQJGSNAKPKT-UHFFFAOYSA-N 0.000 claims description 3
- LYIVUQKRDHMEKB-UHFFFAOYSA-N 6-chloro-n-[2-(dimethylsulfamoyl)phenyl]-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide Chemical compound CN(C)S(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=NN2C=C(Cl)C=NC2=N1 LYIVUQKRDHMEKB-UHFFFAOYSA-N 0.000 claims description 3
- ZWRUSMDJKMOBDX-UHFFFAOYSA-N 6-chloro-n-[2-[ethyl(methyl)sulfamoyl]phenyl]-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide Chemical compound CCN(C)S(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=NN2C=C(Cl)C=NC2=N1 ZWRUSMDJKMOBDX-UHFFFAOYSA-N 0.000 claims description 3
- UNQGEDQDLGFLPV-UHFFFAOYSA-N 6-methyl-n-(2-sulfamoylphenyl)-[1,3]thiazolo[3,2-b][1,2,4]triazole-2-sulfonamide Chemical compound N=1N2C(C)=CSC2=NC=1S(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O UNQGEDQDLGFLPV-UHFFFAOYSA-N 0.000 claims description 3
- 206010002329 Aneurysm Diseases 0.000 claims description 3
- 206010020843 Hyperthermia Diseases 0.000 claims description 3
- FLTQXIWFKWZPEV-UHFFFAOYSA-N N-[2-(dimethylsulfamoyl)phenyl]-7-methyl-1,8,10,12-tetrazatricyclo[7.3.0.02,6]dodeca-2(6),7,9,11-tetraene-11-sulfonamide Chemical compound CN(C)S(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=NN2C(CCC3)=C3C(C)=NC2=N1 FLTQXIWFKWZPEV-UHFFFAOYSA-N 0.000 claims description 3
- 208000008784 apnea Diseases 0.000 claims description 3
- 206010003246 arthritis Diseases 0.000 claims description 3
- 125000005620 boronic acid group Chemical group 0.000 claims description 3
- 125000005843 halogen group Chemical group 0.000 claims description 3
- 230000036031 hyperthermia Effects 0.000 claims description 3
- 238000002156 mixing Methods 0.000 claims description 3
- QXZKFPTZFKPJQH-UHFFFAOYSA-N n-[2-(dimethylsulfamoyl)-5-(trifluoromethyl)phenyl]-5,7-dimethyl-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide Chemical compound CN(C)S(=O)(=O)C1=CC=C(C(F)(F)F)C=C1NS(=O)(=O)C1=NN2C(C)=CC(C)=NC2=N1 QXZKFPTZFKPJQH-UHFFFAOYSA-N 0.000 claims description 3
- FZGHIEJEIQWSBQ-UHFFFAOYSA-N n-[2-(tert-butylsulfamoyl)phenyl]-5,7-dimethyl-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide Chemical compound N1=C2N=C(C)C=C(C)N2N=C1S(=O)(=O)NC1=CC=CC=C1S(=O)(=O)NC(C)(C)C FZGHIEJEIQWSBQ-UHFFFAOYSA-N 0.000 claims description 3
- STOVHGAJJXISHN-UHFFFAOYSA-N n-[2-[ethyl(methyl)sulfamoyl]phenyl]-5,7-dimethyl-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide Chemical compound CCN(C)S(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=NN2C(C)=CC(C)=NC2=N1 STOVHGAJJXISHN-UHFFFAOYSA-N 0.000 claims description 3
- YQDNLUGYZDRJJZ-UHFFFAOYSA-N n-[2-[methoxy(methyl)sulfamoyl]phenyl]-5,7-dimethyl-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide Chemical compound CON(C)S(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=NN2C(C)=CC(C)=NC2=N1 YQDNLUGYZDRJJZ-UHFFFAOYSA-N 0.000 claims description 3
- 239000012038 nucleophile Substances 0.000 claims description 3
- 125000000962 organic group Chemical group 0.000 claims description 3
- PTIBMWDNNRHISW-UHFFFAOYSA-N 5,7-dimethyl-n-(2-sulfamoylphenyl)-[1,2,4]triazolo[1,5-c]pyrimidine-2-sulfonamide Chemical compound N=1N2C(C)=NC(C)=CC2=NC=1S(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O PTIBMWDNNRHISW-UHFFFAOYSA-N 0.000 claims description 2
- 239000003112 inhibitor Substances 0.000 abstract description 2
- 230000003287 optical effect Effects 0.000 abstract description 2
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 325
- IAZDPXIOMUYVGZ-WFGJKAKNSA-N Dimethyl sulfoxide Chemical compound [2H]C([2H])([2H])S(=O)C([2H])([2H])[2H] IAZDPXIOMUYVGZ-WFGJKAKNSA-N 0.000 description 312
- 238000005160 1H NMR spectroscopy Methods 0.000 description 306
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 163
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 131
- OKKJLVBELUTLKV-MZCSYVLQSA-N Deuterated methanol Chemical compound [2H]OC([2H])([2H])[2H] OKKJLVBELUTLKV-MZCSYVLQSA-N 0.000 description 130
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 122
- 239000000243 solution Substances 0.000 description 119
- 235000019439 ethyl acetate Nutrition 0.000 description 112
- 239000000203 mixture Substances 0.000 description 107
- 239000011541 reaction mixture Substances 0.000 description 107
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical class CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 102
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 98
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 86
- 238000002330 electrospray ionisation mass spectrometry Methods 0.000 description 83
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 81
- 238000000746 purification Methods 0.000 description 80
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 76
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 67
- 238000006243 chemical reaction Methods 0.000 description 63
- 238000002953 preparative HPLC Methods 0.000 description 63
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 60
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 57
- 239000002904 solvent Substances 0.000 description 56
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 51
- 239000003480 eluent Substances 0.000 description 51
- 239000012074 organic phase Substances 0.000 description 47
- 239000007787 solid Substances 0.000 description 47
- 238000004440 column chromatography Methods 0.000 description 45
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 43
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 42
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 42
- 239000003643 water by type Substances 0.000 description 40
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 38
- USFZMSVCRYTOJT-UHFFFAOYSA-N Ammonium acetate Chemical compound N.CC(O)=O USFZMSVCRYTOJT-UHFFFAOYSA-N 0.000 description 34
- 239000000047 product Substances 0.000 description 33
- 239000012267 brine Substances 0.000 description 32
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical compound O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 32
- 239000012043 crude product Substances 0.000 description 29
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 27
- IXCSERBJSXMMFS-UHFFFAOYSA-N hydrogen chloride Substances Cl.Cl IXCSERBJSXMMFS-UHFFFAOYSA-N 0.000 description 27
- LSNNMFCWUKXFEE-UHFFFAOYSA-M Bisulfite Chemical compound OS([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-M 0.000 description 25
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 description 25
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 25
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 25
- 235000019341 magnesium sulphate Nutrition 0.000 description 25
- 239000005695 Ammonium acetate Substances 0.000 description 24
- 235000019257 ammonium acetate Nutrition 0.000 description 24
- 229940043376 ammonium acetate Drugs 0.000 description 24
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 23
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 23
- 229910052938 sodium sulfate Inorganic materials 0.000 description 23
- 239000000872 buffer Substances 0.000 description 22
- 229910052786 argon Inorganic materials 0.000 description 21
- 230000015572 biosynthetic process Effects 0.000 description 21
- XEYBRNLFEZDVAW-ARSRFYASSA-N dinoprostone Chemical compound CCCCC[C@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1C\C=C/CCCC(O)=O XEYBRNLFEZDVAW-ARSRFYASSA-N 0.000 description 21
- 235000011152 sodium sulphate Nutrition 0.000 description 21
- YAZSBRQTAHVVGE-UHFFFAOYSA-N 2-aminobenzenesulfonamide Chemical compound NC1=CC=CC=C1S(N)(=O)=O YAZSBRQTAHVVGE-UHFFFAOYSA-N 0.000 description 20
- 229960002986 dinoprostone Drugs 0.000 description 20
- XEYBRNLFEZDVAW-UHFFFAOYSA-N prostaglandin E2 Natural products CCCCCC(O)C=CC1C(O)CC(=O)C1CC=CCCCC(O)=O XEYBRNLFEZDVAW-UHFFFAOYSA-N 0.000 description 20
- RAHZWNYVWXNFOC-UHFFFAOYSA-N sulfur dioxide Inorganic materials O=S=O RAHZWNYVWXNFOC-UHFFFAOYSA-N 0.000 description 20
- 238000003786 synthesis reaction Methods 0.000 description 20
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 19
- 239000000725 suspension Substances 0.000 description 19
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 18
- 238000007429 general method Methods 0.000 description 18
- 238000003818 flash chromatography Methods 0.000 description 17
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 17
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 16
- 239000002244 precipitate Substances 0.000 description 16
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 15
- MZRVEZGGRBJDDB-UHFFFAOYSA-N N-Butyllithium Chemical compound [Li]CCCC MZRVEZGGRBJDDB-UHFFFAOYSA-N 0.000 description 15
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 15
- WEVYAHXRMPXWCK-FIBGUPNXSA-N acetonitrile-d3 Chemical compound [2H]C([2H])([2H])C#N WEVYAHXRMPXWCK-FIBGUPNXSA-N 0.000 description 15
- 239000012298 atmosphere Substances 0.000 description 15
- 150000002431 hydrogen Chemical class 0.000 description 15
- NFHFRUOZVGFOOS-UHFFFAOYSA-N Pd(PPh3)4 Substances [Pd].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 NFHFRUOZVGFOOS-UHFFFAOYSA-N 0.000 description 14
- 238000001914 filtration Methods 0.000 description 14
- 238000011282 treatment Methods 0.000 description 14
- 239000003814 drug Substances 0.000 description 13
- 238000003756 stirring Methods 0.000 description 13
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 12
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 12
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 12
- 229910052796 boron Inorganic materials 0.000 description 12
- 239000000460 chlorine Substances 0.000 description 12
- 238000004519 manufacturing process Methods 0.000 description 12
- FYSNRJHAOHDILO-UHFFFAOYSA-N thionyl chloride Chemical compound ClS(Cl)=O FYSNRJHAOHDILO-UHFFFAOYSA-N 0.000 description 12
- 239000000706 filtrate Substances 0.000 description 11
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 11
- YBBRCQOCSYXUOC-UHFFFAOYSA-N sulfuryl dichloride Chemical compound ClS(Cl)(=O)=O YBBRCQOCSYXUOC-UHFFFAOYSA-N 0.000 description 11
- 239000000284 extract Substances 0.000 description 10
- YIBNHAJFJUQSRA-YNNPMVKQSA-N prostaglandin H2 Chemical compound C1[C@@H]2OO[C@H]1[C@H](/C=C/[C@@H](O)CCCCC)[C@H]2C\C=C/CCCC(O)=O YIBNHAJFJUQSRA-YNNPMVKQSA-N 0.000 description 10
- DPKBAXPHAYBPRL-UHFFFAOYSA-M tetrabutylazanium;iodide Chemical compound [I-].CCCC[N+](CCCC)(CCCC)CCCC DPKBAXPHAYBPRL-UHFFFAOYSA-M 0.000 description 10
- GKTVIFGJASUBGA-UHFFFAOYSA-N 2-amino-5-methylbenzenesulfonamide Chemical compound CC1=CC=C(N)C(S(N)(=O)=O)=C1 GKTVIFGJASUBGA-UHFFFAOYSA-N 0.000 description 9
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 9
- 238000005481 NMR spectroscopy Methods 0.000 description 9
- OFBQJSOFQDEBGM-UHFFFAOYSA-N Pentane Chemical compound CCCCC OFBQJSOFQDEBGM-UHFFFAOYSA-N 0.000 description 9
- 108090000748 Prostaglandin-E Synthases Proteins 0.000 description 9
- 102000004226 Prostaglandin-E Synthases Human genes 0.000 description 9
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 9
- 238000010992 reflux Methods 0.000 description 9
- 238000000926 separation method Methods 0.000 description 9
- JVGPHYSDQTWWNG-ONEGZZNKSA-N (e)-2-(3,4-dichlorophenyl)ethenesulfonyl chloride Chemical compound ClC1=CC=C(\C=C\S(Cl)(=O)=O)C=C1Cl JVGPHYSDQTWWNG-ONEGZZNKSA-N 0.000 description 8
- BDAGIHXWWSANSR-UHFFFAOYSA-N Formic acid Chemical compound OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 8
- 102000004005 Prostaglandin-endoperoxide synthases Human genes 0.000 description 8
- 108090000459 Prostaglandin-endoperoxide synthases Proteins 0.000 description 8
- CSCPPACGZOOCGX-WFGJKAKNSA-N acetone d6 Chemical compound [2H]C([2H])([2H])C(=O)C([2H])([2H])[2H] CSCPPACGZOOCGX-WFGJKAKNSA-N 0.000 description 8
- VSCWAEJMTAWNJL-UHFFFAOYSA-K aluminium trichloride Chemical compound Cl[Al](Cl)Cl VSCWAEJMTAWNJL-UHFFFAOYSA-K 0.000 description 8
- 125000002485 formyl group Chemical group [H]C(*)=O 0.000 description 8
- 239000000741 silica gel Substances 0.000 description 8
- 229910002027 silica gel Inorganic materials 0.000 description 8
- 229910000029 sodium carbonate Inorganic materials 0.000 description 8
- RIOQSEWOXXDEQQ-UHFFFAOYSA-N triphenylphosphine Chemical compound C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 RIOQSEWOXXDEQQ-UHFFFAOYSA-N 0.000 description 8
- LWIHDJKSTIGBAC-UHFFFAOYSA-K tripotassium phosphate Chemical compound [K+].[K+].[K+].[O-]P([O-])([O-])=O LWIHDJKSTIGBAC-UHFFFAOYSA-K 0.000 description 8
- OOGHHXOBEGKELD-LNKPDPKZSA-N *.CC.CN/C=C\S(C)(=O)=O Chemical compound *.CC.CN/C=C\S(C)(=O)=O OOGHHXOBEGKELD-LNKPDPKZSA-N 0.000 description 7
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 7
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 7
- 239000002253 acid Substances 0.000 description 7
- 125000000217 alkyl group Chemical group 0.000 description 7
- 125000004432 carbon atom Chemical group C* 0.000 description 7
- 230000004054 inflammatory process Effects 0.000 description 7
- 150000002500 ions Chemical class 0.000 description 7
- 239000003921 oil Substances 0.000 description 7
- 235000019198 oils Nutrition 0.000 description 7
- UCYNBMBCQKXUHI-SNAWJCMRSA-N (e)-2-(4-chloro-2-methoxyphenyl)ethenesulfonyl chloride Chemical compound COC1=CC(Cl)=CC=C1\C=C\S(Cl)(=O)=O UCYNBMBCQKXUHI-SNAWJCMRSA-N 0.000 description 6
- PQODWTNHDKDHIW-UHFFFAOYSA-N 2,3-dichlorobenzenesulfonyl chloride Chemical compound ClC1=CC=CC(S(Cl)(=O)=O)=C1Cl PQODWTNHDKDHIW-UHFFFAOYSA-N 0.000 description 6
- YCUSKOREEWVGMS-UHFFFAOYSA-N 2-[(3-bromophenyl)sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC(Br)=C1 YCUSKOREEWVGMS-UHFFFAOYSA-N 0.000 description 6
- PNBAVBCBCPRSMK-UHFFFAOYSA-N 2-[2-(4-chloro-2-methoxyphenyl)ethenylsulfonylamino]-5-methylbenzenesulfonamide Chemical compound COC1=CC(Cl)=CC=C1C=CS(=O)(=O)NC1=CC=C(C)C=C1S(N)(=O)=O PNBAVBCBCPRSMK-UHFFFAOYSA-N 0.000 description 6
- PPWNCLVNXGCGAF-UHFFFAOYSA-N 3,3-dimethylbut-1-yne Chemical compound CC(C)(C)C#C PPWNCLVNXGCGAF-UHFFFAOYSA-N 0.000 description 6
- PJGOLCXVWIYXRQ-UHFFFAOYSA-N 3-bromobenzenesulfonyl chloride Chemical compound ClS(=O)(=O)C1=CC=CC(Br)=C1 PJGOLCXVWIYXRQ-UHFFFAOYSA-N 0.000 description 6
- HWIOYTMAUQHHGR-UHFFFAOYSA-N 6-amino-2,3-dihydro-1h-indene-5-sulfonamide Chemical compound C1=C(S(N)(=O)=O)C(N)=CC2=C1CCC2 HWIOYTMAUQHHGR-UHFFFAOYSA-N 0.000 description 6
- KZBUYRJDOAKODT-UHFFFAOYSA-N Chlorine Chemical compound ClCl KZBUYRJDOAKODT-UHFFFAOYSA-N 0.000 description 6
- XTHFKEDIFFGKHM-UHFFFAOYSA-N Dimethoxyethane Chemical compound COCCOC XTHFKEDIFFGKHM-UHFFFAOYSA-N 0.000 description 6
- 206010061218 Inflammation Diseases 0.000 description 6
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 6
- 125000002947 alkylene group Chemical group 0.000 description 6
- 238000004458 analytical method Methods 0.000 description 6
- YZXBAPSDXZZRGB-DOFZRALJSA-N arachidonic acid Chemical compound CCCCC\C=C/C\C=C/C\C=C/C\C=C/CCCC(O)=O YZXBAPSDXZZRGB-DOFZRALJSA-N 0.000 description 6
- 238000003556 assay Methods 0.000 description 6
- 239000008280 blood Substances 0.000 description 6
- 210000004369 blood Anatomy 0.000 description 6
- ILAHWRKJUDSMFH-UHFFFAOYSA-N boron tribromide Chemical compound BrB(Br)Br ILAHWRKJUDSMFH-UHFFFAOYSA-N 0.000 description 6
- 239000013058 crude material Substances 0.000 description 6
- NXQGGXCHGDYOHB-UHFFFAOYSA-L cyclopenta-1,4-dien-1-yl(diphenyl)phosphane;dichloropalladium;iron(2+) Chemical compound [Fe+2].Cl[Pd]Cl.[CH-]1C=CC(P(C=2C=CC=CC=2)C=2C=CC=CC=2)=C1.[CH-]1C=CC(P(C=2C=CC=CC=2)C=2C=CC=CC=2)=C1 NXQGGXCHGDYOHB-UHFFFAOYSA-L 0.000 description 6
- UAOMVDZJSHZZME-UHFFFAOYSA-N diisopropylamine Chemical compound CC(C)NC(C)C UAOMVDZJSHZZME-UHFFFAOYSA-N 0.000 description 6
- RWSXRVCMGQZWBV-WDSKDSINSA-N glutathione Chemical compound OC(=O)[C@@H](N)CCC(=O)N[C@@H](CS)C(=O)NCC(O)=O RWSXRVCMGQZWBV-WDSKDSINSA-N 0.000 description 6
- 238000004128 high performance liquid chromatography Methods 0.000 description 6
- LYGJENNIWJXYER-UHFFFAOYSA-N nitromethane Chemical compound C[N+]([O-])=O LYGJENNIWJXYER-UHFFFAOYSA-N 0.000 description 6
- 239000012044 organic layer Substances 0.000 description 6
- 239000007858 starting material Substances 0.000 description 6
- 238000012360 testing method Methods 0.000 description 6
- 239000003039 volatile agent Substances 0.000 description 6
- UCYNBMBCQKXUHI-UHFFFAOYSA-N 2-(4-chloro-2-methoxyphenyl)ethenesulfonyl chloride Chemical compound COC1=CC(Cl)=CC=C1C=CS(Cl)(=O)=O UCYNBMBCQKXUHI-UHFFFAOYSA-N 0.000 description 5
- DTOAHLYOVKHINE-UHFFFAOYSA-N 2-[2-(4-bromophenyl)ethylsulfonylamino]-5-(hydroxymethyl)benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(CO)=CC=C1NS(=O)(=O)CCC1=CC=C(Br)C=C1 DTOAHLYOVKHINE-UHFFFAOYSA-N 0.000 description 5
- HWQDHHCUOZZFIG-UHFFFAOYSA-N 2-amino-4-chlorobenzenesulfonamide Chemical compound NC1=CC(Cl)=CC=C1S(N)(=O)=O HWQDHHCUOZZFIG-UHFFFAOYSA-N 0.000 description 5
- 102000004190 Enzymes Human genes 0.000 description 5
- 108090000790 Enzymes Proteins 0.000 description 5
- 239000001828 Gelatine Substances 0.000 description 5
- 229910021529 ammonia Inorganic materials 0.000 description 5
- 239000002585 base Substances 0.000 description 5
- FJDQFPXHSGXQBY-UHFFFAOYSA-L caesium carbonate Chemical compound [Cs+].[Cs+].[O-]C([O-])=O FJDQFPXHSGXQBY-UHFFFAOYSA-L 0.000 description 5
- 229910000024 caesium carbonate Inorganic materials 0.000 description 5
- 239000011203 carbon fibre reinforced carbon Substances 0.000 description 5
- 210000004027 cell Anatomy 0.000 description 5
- 239000003610 charcoal Substances 0.000 description 5
- 229910052801 chlorine Inorganic materials 0.000 description 5
- 125000004122 cyclic group Chemical group 0.000 description 5
- 239000002158 endotoxin Substances 0.000 description 5
- 235000019253 formic acid Nutrition 0.000 description 5
- 229920000159 gelatin Polymers 0.000 description 5
- 235000019322 gelatine Nutrition 0.000 description 5
- 239000000543 intermediate Substances 0.000 description 5
- 229920006008 lipopolysaccharide Polymers 0.000 description 5
- 239000000463 material Substances 0.000 description 5
- 230000003228 microsomal effect Effects 0.000 description 5
- 229910052757 nitrogen Inorganic materials 0.000 description 5
- YJVFFLUZDVXJQI-UHFFFAOYSA-L palladium(ii) acetate Chemical compound [Pd+2].CC([O-])=O.CC([O-])=O YJVFFLUZDVXJQI-UHFFFAOYSA-L 0.000 description 5
- 239000008057 potassium phosphate buffer Substances 0.000 description 5
- 229940127293 prostanoid Drugs 0.000 description 5
- 150000003814 prostanoids Chemical class 0.000 description 5
- 239000000523 sample Substances 0.000 description 5
- 229920006395 saturated elastomer Polymers 0.000 description 5
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 5
- 235000017557 sodium bicarbonate Nutrition 0.000 description 5
- 239000011550 stock solution Substances 0.000 description 5
- 239000003826 tablet Substances 0.000 description 5
- 0 *.CC.CCCO(O)SN/C=C\S(C)(=O)=O Chemical compound *.CC.CCCO(O)SN/C=C\S(C)(=O)=O 0.000 description 4
- KLVVIEOBVCVMFK-UHFFFAOYSA-N 2-[(3-bromophenyl)sulfonylamino]-5-methylbenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(C)=CC=C1NS(=O)(=O)C1=CC=CC(Br)=C1 KLVVIEOBVCVMFK-UHFFFAOYSA-N 0.000 description 4
- UMMATWMLLMKWAX-UHFFFAOYSA-N 2-[(3-bromophenyl)sulfonylamino]-5-phenoxybenzenesulfonamide Chemical compound C=1C=C(NS(=O)(=O)C=2C=C(Br)C=CC=2)C(S(=O)(=O)N)=CC=1OC1=CC=CC=C1 UMMATWMLLMKWAX-UHFFFAOYSA-N 0.000 description 4
- RQYNKOPXZBLCIC-UHFFFAOYSA-N 2-[2-(6-bromopyridin-3-yl)ethylsulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)CCC1=CC=C(Br)N=C1 RQYNKOPXZBLCIC-UHFFFAOYSA-N 0.000 description 4
- VHYFNPMBLIVWCW-UHFFFAOYSA-N 4-Dimethylaminopyridine Chemical compound CN(C)C1=CC=NC=C1 VHYFNPMBLIVWCW-UHFFFAOYSA-N 0.000 description 4
- MJRHGUXKSAWFOC-UHFFFAOYSA-N 4-amino-n,n-bis(prop-2-enyl)pyridine-3-sulfonamide Chemical compound NC1=CC=NC=C1S(=O)(=O)N(CC=C)CC=C MJRHGUXKSAWFOC-UHFFFAOYSA-N 0.000 description 4
- ICGLPKIVTVWCFT-UHFFFAOYSA-N 4-methylbenzenesulfonohydrazide Chemical compound CC1=CC=C(S(=O)(=O)NN)C=C1 ICGLPKIVTVWCFT-UHFFFAOYSA-N 0.000 description 4
- PAYRUJLWNCNPSJ-UHFFFAOYSA-N Aniline Chemical compound NC1=CC=CC=C1 PAYRUJLWNCNPSJ-UHFFFAOYSA-N 0.000 description 4
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 4
- 229910021595 Copper(I) iodide Inorganic materials 0.000 description 4
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 4
- VMHLLURERBWHNL-UHFFFAOYSA-M Sodium acetate Chemical compound [Na+].CC([O-])=O VMHLLURERBWHNL-UHFFFAOYSA-M 0.000 description 4
- 208000006011 Stroke Diseases 0.000 description 4
- 125000000499 benzofuranyl group Chemical group O1C(=CC2=C1C=CC=C2)* 0.000 description 4
- 239000002775 capsule Substances 0.000 description 4
- WRJWRGBVPUUDLA-UHFFFAOYSA-N chlorosulfonyl isocyanate Chemical compound ClS(=O)(=O)N=C=O WRJWRGBVPUUDLA-UHFFFAOYSA-N 0.000 description 4
- LSXDOTMGLUJQCM-UHFFFAOYSA-M copper(i) iodide Chemical compound I[Cu] LSXDOTMGLUJQCM-UHFFFAOYSA-M 0.000 description 4
- LCGLNKUTAGEVQW-WFGJKAKNSA-N dimethyl ether-d6 Chemical compound [2H]C([2H])([2H])OC([2H])([2H])[2H] LCGLNKUTAGEVQW-WFGJKAKNSA-N 0.000 description 4
- 150000002148 esters Chemical class 0.000 description 4
- 125000004494 ethyl ester group Chemical group 0.000 description 4
- 238000002868 homogeneous time resolved fluorescence Methods 0.000 description 4
- 239000007788 liquid Substances 0.000 description 4
- 239000012280 lithium aluminium hydride Substances 0.000 description 4
- 238000001819 mass spectrum Methods 0.000 description 4
- 210000001589 microsome Anatomy 0.000 description 4
- 239000013642 negative control Substances 0.000 description 4
- 239000003960 organic solvent Substances 0.000 description 4
- 239000012071 phase Substances 0.000 description 4
- 239000013641 positive control Substances 0.000 description 4
- 239000012286 potassium permanganate Substances 0.000 description 4
- 229910000160 potassium phosphate Inorganic materials 0.000 description 4
- 235000011009 potassium phosphates Nutrition 0.000 description 4
- 238000004237 preparative chromatography Methods 0.000 description 4
- 230000000770 proinflammatory effect Effects 0.000 description 4
- 125000006239 protecting group Chemical group 0.000 description 4
- 238000001953 recrystallisation Methods 0.000 description 4
- 230000009467 reduction Effects 0.000 description 4
- 239000001632 sodium acetate Substances 0.000 description 4
- 235000017281 sodium acetate Nutrition 0.000 description 4
- DYHSDKLCOJIUFX-UHFFFAOYSA-N tert-butoxycarbonyl anhydride Chemical compound CC(C)(C)OC(=O)OC(=O)OC(C)(C)C DYHSDKLCOJIUFX-UHFFFAOYSA-N 0.000 description 4
- GFLQVHMQXCOBLB-UHFFFAOYSA-N tert-butyl n-[2-[2-(4-chlorophenyl)ethylsulfonylamino]-5-hydroxyphenyl]sulfonylcarbamate Chemical compound CC(C)(C)OC(=O)NS(=O)(=O)C1=CC(O)=CC=C1NS(=O)(=O)CCC1=CC=C(Cl)C=C1 GFLQVHMQXCOBLB-UHFFFAOYSA-N 0.000 description 4
- WLPUWLXVBWGYMZ-UHFFFAOYSA-N tricyclohexylphosphine Chemical compound C1CCCCC1P(C1CCCCC1)C1CCCCC1 WLPUWLXVBWGYMZ-UHFFFAOYSA-N 0.000 description 4
- 230000029663 wound healing Effects 0.000 description 4
- OEVVUIFFYVWIJW-MDZDMXLPSA-N (e)-2-[4-(1-benzofuran-2-yl)phenyl]ethenesulfonyl chloride Chemical compound C1=CC(/C=C/S(=O)(=O)Cl)=CC=C1C1=CC2=CC=CC=C2O1 OEVVUIFFYVWIJW-MDZDMXLPSA-N 0.000 description 3
- 238000004293 19F NMR spectroscopy Methods 0.000 description 3
- WGUYMLVRJKFPHI-UHFFFAOYSA-N 2-(4-bromophenyl)ethanesulfonyl chloride Chemical compound ClS(=O)(=O)CCC1=CC=C(Br)C=C1 WGUYMLVRJKFPHI-UHFFFAOYSA-N 0.000 description 3
- RDLOYHZHKDCZHO-UHFFFAOYSA-N 2-(4-chlorophenyl)ethanesulfonyl chloride Chemical compound ClC1=CC=C(CCS(Cl)(=O)=O)C=C1 RDLOYHZHKDCZHO-UHFFFAOYSA-N 0.000 description 3
- MIRLMGWYXXOHNG-UHFFFAOYSA-N 2-amino-4-cyanobenzenesulfonamide Chemical compound NC1=CC(C#N)=CC=C1S(N)(=O)=O MIRLMGWYXXOHNG-UHFFFAOYSA-N 0.000 description 3
- QDRZCTURWDQVCY-UHFFFAOYSA-N 2-amino-5-cyanobenzenesulfonamide Chemical compound NC1=CC=C(C#N)C=C1S(N)(=O)=O QDRZCTURWDQVCY-UHFFFAOYSA-N 0.000 description 3
- CEBKHWWANWSNTI-UHFFFAOYSA-N 2-methylbut-3-yn-2-ol Chemical compound CC(C)(O)C#C CEBKHWWANWSNTI-UHFFFAOYSA-N 0.000 description 3
- ZDEOFXOTIZSVOG-UHFFFAOYSA-N 3-aminothiophene-2-sulfonamide Chemical compound NC=1C=CSC=1S(N)(=O)=O ZDEOFXOTIZSVOG-UHFFFAOYSA-N 0.000 description 3
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 3
- KHMOPAFSRWCTQL-ZRDIBKRKSA-N 6-[[(e)-2-[4-(furan-2-yl)phenyl]ethenyl]sulfonylamino]-2,3-dihydro-1h-indene-5-sulfonamide Chemical compound NS(=O)(=O)C1=CC=2CCCC=2C=C1NS(=O)(=O)\C=C\C(C=C1)=CC=C1C1=CC=CO1 KHMOPAFSRWCTQL-ZRDIBKRKSA-N 0.000 description 3
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 3
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 3
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 3
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 3
- 101001135391 Homo sapiens Prostaglandin E synthase Proteins 0.000 description 3
- CMWTZPSULFXXJA-UHFFFAOYSA-N Naproxen Natural products C1=C(C(C)C(O)=O)C=CC2=CC(OC)=CC=C21 CMWTZPSULFXXJA-UHFFFAOYSA-N 0.000 description 3
- 208000001294 Nociceptive Pain Diseases 0.000 description 3
- YNHIGQDRGKUECZ-UHFFFAOYSA-L PdCl2(PPh3)2 Substances [Cl-].[Cl-].[Pd+2].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 YNHIGQDRGKUECZ-UHFFFAOYSA-L 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- 102100038280 Prostaglandin G/H synthase 2 Human genes 0.000 description 3
- 108050003267 Prostaglandin G/H synthase 2 Proteins 0.000 description 3
- DWAQJAXMDSEUJJ-UHFFFAOYSA-M Sodium bisulfite Chemical compound [Na+].OS([O-])=O DWAQJAXMDSEUJJ-UHFFFAOYSA-M 0.000 description 3
- 235000011114 ammonium hydroxide Nutrition 0.000 description 3
- 239000007864 aqueous solution Substances 0.000 description 3
- 229940114079 arachidonic acid Drugs 0.000 description 3
- 235000021342 arachidonic acid Nutrition 0.000 description 3
- 125000001164 benzothiazolyl group Chemical group S1C(=NC2=C1C=CC=C2)* 0.000 description 3
- 125000004196 benzothienyl group Chemical group S1C(=CC2=C1C=CC=C2)* 0.000 description 3
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 3
- 229910052794 bromium Inorganic materials 0.000 description 3
- 239000003054 catalyst Substances 0.000 description 3
- 150000001805 chlorine compounds Chemical class 0.000 description 3
- 238000004587 chromatography analysis Methods 0.000 description 3
- 238000001816 cooling Methods 0.000 description 3
- LGTLXDJOAJDFLR-UHFFFAOYSA-N diethyl chlorophosphate Chemical compound CCOP(Cl)(=O)OCC LGTLXDJOAJDFLR-UHFFFAOYSA-N 0.000 description 3
- 125000000723 dihydrobenzofuranyl group Chemical group O1C(CC2=C1C=CC=C2)* 0.000 description 3
- CXPXCCAOKXGQQZ-UHFFFAOYSA-N ethyl diethoxyphosphorylmethanesulfonate Chemical compound CCOP(=O)(OCC)CS(=O)(=O)OCC CXPXCCAOKXGQQZ-UHFFFAOYSA-N 0.000 description 3
- 239000011737 fluorine Substances 0.000 description 3
- 229910052731 fluorine Inorganic materials 0.000 description 3
- 230000002068 genetic effect Effects 0.000 description 3
- 229960003180 glutathione Drugs 0.000 description 3
- 102000047789 human PTGES Human genes 0.000 description 3
- 125000003392 indanyl group Chemical group C1(CCC2=CC=CC=C12)* 0.000 description 3
- 230000002401 inhibitory effect Effects 0.000 description 3
- 230000005764 inhibitory process Effects 0.000 description 3
- 239000002207 metabolite Substances 0.000 description 3
- 125000002950 monocyclic group Chemical group 0.000 description 3
- 229960002009 naproxen Drugs 0.000 description 3
- CMWTZPSULFXXJA-VIFPVBQESA-N naproxen Chemical compound C1=C([C@H](C)C(O)=O)C=CC2=CC(OC)=CC=C21 CMWTZPSULFXXJA-VIFPVBQESA-N 0.000 description 3
- 229940021182 non-steroidal anti-inflammatory drug Drugs 0.000 description 3
- 229910052760 oxygen Inorganic materials 0.000 description 3
- 238000012746 preparative thin layer chromatography Methods 0.000 description 3
- 238000011321 prophylaxis Methods 0.000 description 3
- 239000000377 silicon dioxide Substances 0.000 description 3
- 235000010267 sodium hydrogen sulphite Nutrition 0.000 description 3
- 238000010561 standard procedure Methods 0.000 description 3
- 229940124530 sulfonamide Drugs 0.000 description 3
- NHGXDBSUJJNIRV-UHFFFAOYSA-M tetrabutylammonium chloride Chemical class [Cl-].CCCC[N+](CCCC)(CCCC)CCCC NHGXDBSUJJNIRV-UHFFFAOYSA-M 0.000 description 3
- 230000001225 therapeutic effect Effects 0.000 description 3
- JKIGHOARKAIPJI-UHFFFAOYSA-N (3,4-dichlorophenyl)boronic acid Chemical compound OB(O)C1=CC=C(Cl)C(Cl)=C1 JKIGHOARKAIPJI-UHFFFAOYSA-N 0.000 description 2
- LOLPLMDQLOJSDH-UHFFFAOYSA-N (5-chloro-6-methoxypyridin-3-yl)boronic acid Chemical compound COC1=NC=C(B(O)O)C=C1Cl LOLPLMDQLOJSDH-UHFFFAOYSA-N 0.000 description 2
- QXZIBRSHYJDVHF-SCBDLNNBSA-M (E)-2-(6-bromopyridin-3-yl)ethenesulfonate tetrabutylazanium Chemical compound CCCC[N+](CCCC)(CCCC)CCCC.C1=CC(=NC=C1/C=C/S(=O)(=O)[O-])Br QXZIBRSHYJDVHF-SCBDLNNBSA-M 0.000 description 2
- QAAQAKSRVIIUEV-AATRIKPKSA-N (e)-2-(4-bromophenyl)ethenesulfonyl chloride Chemical compound ClS(=O)(=O)\C=C\C1=CC=C(Br)C=C1 QAAQAKSRVIIUEV-AATRIKPKSA-N 0.000 description 2
- SMXDMHCNNWGZJY-CMDGGOBGSA-N (e)-2-(4-tert-butylphenyl)ethenesulfonyl chloride Chemical compound CC(C)(C)C1=CC=C(\C=C\S(Cl)(=O)=O)C=C1 SMXDMHCNNWGZJY-CMDGGOBGSA-N 0.000 description 2
- NEIUMZBEBUYYEQ-ONEGZZNKSA-N (e)-2-(6-bromopyridin-3-yl)ethenesulfonyl chloride Chemical compound ClS(=O)(=O)\C=C\C1=CC=C(Br)N=C1 NEIUMZBEBUYYEQ-ONEGZZNKSA-N 0.000 description 2
- NAJBCCLGBGBJLI-MDZDMXLPSA-N (e)-2-[4-(1-benzofuran-2-yl)phenyl]ethenesulfonic acid Chemical compound C1=CC(/C=C/S(=O)(=O)O)=CC=C1C1=CC2=CC=CC=C2O1 NAJBCCLGBGBJLI-MDZDMXLPSA-N 0.000 description 2
- SCRLCACWJGTYQU-OLGQORCHSA-N *.CC.CS(=O)(=O)/C=C\N Chemical compound *.CC.CS(=O)(=O)/C=C\N SCRLCACWJGTYQU-OLGQORCHSA-N 0.000 description 2
- 125000005871 1,3-benzodioxolyl group Chemical group 0.000 description 2
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 2
- YBYIRNPNPLQARY-UHFFFAOYSA-N 1H-indene Natural products C1=CC=C2CC=CC2=C1 YBYIRNPNPLQARY-UHFFFAOYSA-N 0.000 description 2
- BPAYQKBSSNYTSP-UHFFFAOYSA-N 2,3-dichloro-n-(4-methoxy-2-sulfamoylphenyl)benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(OC)=CC=C1NS(=O)(=O)C1=CC=CC(Cl)=C1Cl BPAYQKBSSNYTSP-UHFFFAOYSA-N 0.000 description 2
- VHCVYGNNCGAWBP-UHFFFAOYSA-N 2,3-difluorobenzenesulfonyl chloride Chemical compound FC1=CC=CC(S(Cl)(=O)=O)=C1F VHCVYGNNCGAWBP-UHFFFAOYSA-N 0.000 description 2
- ACJBOCNAUWWCMN-UHFFFAOYSA-N 2-(4-chloro-2-methoxyphenyl)ethenesulfonic acid Chemical compound COC1=CC(Cl)=CC=C1C=CS(O)(=O)=O ACJBOCNAUWWCMN-UHFFFAOYSA-N 0.000 description 2
- AFABGHUZZDYHJO-UHFFFAOYSA-N 2-Methylpentane Chemical compound CCCC(C)C AFABGHUZZDYHJO-UHFFFAOYSA-N 0.000 description 2
- CMBFXOIUIXAOQN-UHFFFAOYSA-N 2-[2-(2-ethoxyphenyl)ethenylsulfonylamino]benzenesulfonamide Chemical compound CCOC1=CC=CC=C1C=CS(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O CMBFXOIUIXAOQN-UHFFFAOYSA-N 0.000 description 2
- WVDCPCYQNCWPIN-UHFFFAOYSA-N 2-[2-(4-bromophenyl)ethylsulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)CCC1=CC=C(Br)C=C1 WVDCPCYQNCWPIN-UHFFFAOYSA-N 0.000 description 2
- KEYSMNWVLJLQAC-UHFFFAOYSA-N 2-[2-(4-chlorophenyl)ethylsulfonylamino]-5-hydroxybenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(O)=CC=C1NS(=O)(=O)CCC1=CC=C(Cl)C=C1 KEYSMNWVLJLQAC-UHFFFAOYSA-N 0.000 description 2
- AMILJNWKRZQXLD-UHFFFAOYSA-N 2-[2-(4-chlorophenyl)ethylsulfonylamino]-5-methoxybenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(OC)=CC=C1NS(=O)(=O)CCC1=CC=C(Cl)C=C1 AMILJNWKRZQXLD-UHFFFAOYSA-N 0.000 description 2
- NAGPYCOEEXJFOJ-ONEGZZNKSA-N 2-[[(e)-2-(3,4-dichlorophenyl)ethenyl]sulfonylamino]-4,6-difluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=C(F)C=C(F)C=C1NS(=O)(=O)\C=C\C1=CC=C(Cl)C(Cl)=C1 NAGPYCOEEXJFOJ-ONEGZZNKSA-N 0.000 description 2
- BQLXYHWCGKBXNN-SNAWJCMRSA-N 2-[[(e)-2-(4-chloro-2-methoxyphenyl)ethenyl]sulfonylamino]-4,6-difluorobenzenesulfonamide Chemical compound COC1=CC(Cl)=CC=C1\C=C\S(=O)(=O)NC1=CC(F)=CC(F)=C1S(N)(=O)=O BQLXYHWCGKBXNN-SNAWJCMRSA-N 0.000 description 2
- OYIXHXOHQKULEB-VOTSOKGWSA-N 2-[[(e)-2-(4-chloro-2-methoxyphenyl)ethenyl]sulfonylamino]-4-cyanobenzenesulfonamide Chemical compound COC1=CC(Cl)=CC=C1\C=C\S(=O)(=O)NC1=CC(C#N)=CC=C1S(N)(=O)=O OYIXHXOHQKULEB-VOTSOKGWSA-N 0.000 description 2
- KBAHWXSODBZIBT-BQYQJAHWSA-N 2-[[(e)-2-(4-chloro-2-methoxyphenyl)ethenyl]sulfonylamino]-4-methylbenzenesulfonamide Chemical compound COC1=CC(Cl)=CC=C1\C=C\S(=O)(=O)NC1=CC(C)=CC=C1S(N)(=O)=O KBAHWXSODBZIBT-BQYQJAHWSA-N 0.000 description 2
- UPOJMVSUPUKODZ-VOTSOKGWSA-N 2-[[(e)-2-(4-chloro-2-methoxyphenyl)ethenyl]sulfonylamino]-5-cyanobenzenesulfonamide Chemical compound COC1=CC(Cl)=CC=C1\C=C\S(=O)(=O)NC1=CC=C(C#N)C=C1S(N)(=O)=O UPOJMVSUPUKODZ-VOTSOKGWSA-N 0.000 description 2
- INELXZOACWZODF-BQYQJAHWSA-N 2-[[(e)-2-(6-bromopyridin-3-yl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)\C=C\C1=CC=C(Br)N=C1 INELXZOACWZODF-BQYQJAHWSA-N 0.000 description 2
- OMMLWHXDRUANRD-UHFFFAOYSA-N 2-amino-4,6-difluorobenzenesulfonamide Chemical compound NC1=CC(F)=CC(F)=C1S(N)(=O)=O OMMLWHXDRUANRD-UHFFFAOYSA-N 0.000 description 2
- NECSZWFYSDDJSW-UHFFFAOYSA-N 2-amino-4-methylbenzenesulfonamide Chemical compound CC1=CC=C(S(N)(=O)=O)C(N)=C1 NECSZWFYSDDJSW-UHFFFAOYSA-N 0.000 description 2
- YXTRCGAYRIYIPJ-UHFFFAOYSA-N 2-amino-5-(3-hydroxy-3-methylbut-1-ynyl)benzenesulfonamide Chemical compound CC(C)(O)C#CC1=CC=C(N)C(S(N)(=O)=O)=C1 YXTRCGAYRIYIPJ-UHFFFAOYSA-N 0.000 description 2
- BGILQOXCWMHWOM-UHFFFAOYSA-N 2-amino-5-bromobenzenesulfonamide Chemical compound NC1=CC=C(Br)C=C1S(N)(=O)=O BGILQOXCWMHWOM-UHFFFAOYSA-N 0.000 description 2
- OVGLLRGKUCZOAG-UHFFFAOYSA-N 2-amino-5-chloro-4-fluorobenzenesulfonamide Chemical compound NC1=CC(F)=C(Cl)C=C1S(N)(=O)=O OVGLLRGKUCZOAG-UHFFFAOYSA-N 0.000 description 2
- RBCLOSOKWLMKMB-UHFFFAOYSA-N 2-amino-5-chlorobenzenesulfonamide Chemical compound NC1=CC=C(Cl)C=C1S(N)(=O)=O RBCLOSOKWLMKMB-UHFFFAOYSA-N 0.000 description 2
- DSUQOTGCKDGCEZ-UHFFFAOYSA-N 2-amino-5-methoxybenzenesulfonamide Chemical compound COC1=CC=C(N)C(S(N)(=O)=O)=C1 DSUQOTGCKDGCEZ-UHFFFAOYSA-N 0.000 description 2
- VSADXYMYSLJIOY-UHFFFAOYSA-N 2-amino-5-phenoxybenzenesulfonamide Chemical compound C1=C(S(N)(=O)=O)C(N)=CC=C1OC1=CC=CC=C1 VSADXYMYSLJIOY-UHFFFAOYSA-N 0.000 description 2
- ZFSITVKIPCUGFK-UHFFFAOYSA-N 2-benzylsulfanyl-3-nitrothiophene Chemical compound C1=CSC(SCC=2C=CC=CC=2)=C1[N+](=O)[O-] ZFSITVKIPCUGFK-UHFFFAOYSA-N 0.000 description 2
- ICTGMBLTEWEKHJ-UHFFFAOYSA-N 2-n-methyl-8-n-(2-sulfamoylphenyl)dibenzothiophene-2,8-disulfonamide Chemical compound C1=C2C3=CC(S(=O)(=O)NC)=CC=C3SC2=CC=C1S(=O)(=O)NC1=CC=CC=C1S(N)(=O)=O ICTGMBLTEWEKHJ-UHFFFAOYSA-N 0.000 description 2
- IGFBHZODFKDWRW-UHFFFAOYSA-N 3-(3,4-dichlorophenyl)benzenesulfonyl chloride Chemical compound C1=C(Cl)C(Cl)=CC=C1C1=CC=CC(S(Cl)(=O)=O)=C1 IGFBHZODFKDWRW-UHFFFAOYSA-N 0.000 description 2
- KSXZQDSMFSILAA-UHFFFAOYSA-N 3-[(3-bromophenyl)sulfonylamino]thiophene-2-sulfonamide Chemical compound S1C=CC(NS(=O)(=O)C=2C=C(Br)C=CC=2)=C1S(=O)(=O)N KSXZQDSMFSILAA-UHFFFAOYSA-N 0.000 description 2
- UKQUKMDLPWATJO-UHFFFAOYSA-N 3-[2-(4-bromophenyl)ethylsulfonylamino]thiophene-2-sulfonamide Chemical compound S1C=CC(NS(=O)(=O)CCC=2C=CC(Br)=CC=2)=C1S(=O)(=O)N UKQUKMDLPWATJO-UHFFFAOYSA-N 0.000 description 2
- QYVPJSVSONXMFU-UHFFFAOYSA-N 3-nitrothiophene-2-sulfonamide Chemical compound NS(=O)(=O)C=1SC=CC=1[N+]([O-])=O QYVPJSVSONXMFU-UHFFFAOYSA-N 0.000 description 2
- IHMXBKUVDMZNTB-UHFFFAOYSA-N 3-nitrothiophene-2-sulfonyl chloride Chemical compound [O-][N+](=O)C=1C=CSC=1S(Cl)(=O)=O IHMXBKUVDMZNTB-UHFFFAOYSA-N 0.000 description 2
- NZFNRSKNXSUNBO-UHFFFAOYSA-N 4-[(2,3-dichlorophenyl)sulfonylamino]-3-sulfamoylbenzoic acid Chemical compound NS(=O)(=O)C1=CC(C(O)=O)=CC=C1NS(=O)(=O)C1=CC=CC(Cl)=C1Cl NZFNRSKNXSUNBO-UHFFFAOYSA-N 0.000 description 2
- KQGBJWGGEABNBJ-UHFFFAOYSA-N 4-[(3-bromophenyl)sulfonylamino]-n,n-bis(prop-2-enyl)pyridine-3-sulfonamide Chemical compound BrC1=CC=CC(S(=O)(=O)NC=2C(=CN=CC=2)S(=O)(=O)N(CC=C)CC=C)=C1 KQGBJWGGEABNBJ-UHFFFAOYSA-N 0.000 description 2
- BJVZALHFAIKTEM-UHFFFAOYSA-N 4-[2-(4-bromophenyl)ethylsulfonylamino]-3-sulfamoylbenzoic acid Chemical compound NS(=O)(=O)C1=CC(C(O)=O)=CC=C1NS(=O)(=O)CCC1=CC=C(Br)C=C1 BJVZALHFAIKTEM-UHFFFAOYSA-N 0.000 description 2
- RDYFIHZKILVGHG-UHFFFAOYSA-N 4-[2-(4-chloro-2-methoxyphenyl)ethylsulfonylamino]-3-sulfamoylbenzoic acid Chemical compound COC1=CC(Cl)=CC=C1CCS(=O)(=O)NC1=CC=C(C(O)=O)C=C1S(N)(=O)=O RDYFIHZKILVGHG-UHFFFAOYSA-N 0.000 description 2
- IFUWMUANXROVIL-UHFFFAOYSA-N 4-[2-(4-chlorophenyl)ethylsulfonylamino]-3-sulfamoylbenzoic acid Chemical compound NS(=O)(=O)C1=CC(C(O)=O)=CC=C1NS(=O)(=O)CCC1=CC=C(Cl)C=C1 IFUWMUANXROVIL-UHFFFAOYSA-N 0.000 description 2
- PGYPHEBXNGPCQY-GXDHUFHOSA-N 4-[[(e)-2-(4-bromophenyl)ethenyl]sulfonylamino]-n,n-bis(prop-2-enyl)pyridine-3-sulfonamide Chemical compound C1=CC(Br)=CC=C1\C=C\S(=O)(=O)NC1=CC=NC=C1S(=O)(=O)N(CC=C)CC=C PGYPHEBXNGPCQY-GXDHUFHOSA-N 0.000 description 2
- QWBCSMOBULXBQY-UKTHLTGXSA-N 4-[[(e)-2-(4-chloro-2-methoxyphenyl)ethenyl]sulfonylamino]-n,n-bis(prop-2-enyl)pyridine-3-sulfonamide Chemical compound COC1=CC(Cl)=CC=C1\C=C\S(=O)(=O)NC1=CC=NC=C1S(=O)(=O)N(CC=C)CC=C QWBCSMOBULXBQY-UKTHLTGXSA-N 0.000 description 2
- YUHMJWZFTUQDDA-FNORWQNLSA-N 4-[[(e)-2-(4-chloro-2-methoxyphenyl)ethenyl]sulfonylamino]pyridine-3-sulfonamide Chemical compound COC1=CC(Cl)=CC=C1\C=C\S(=O)(=O)NC1=CC=NC=C1S(N)(=O)=O YUHMJWZFTUQDDA-FNORWQNLSA-N 0.000 description 2
- BESNZTZUWXCXMV-NBVRZTHBSA-N 4-[[(e)-2-[4-(1-benzofuran-2-yl)phenyl]ethenyl]sulfonylamino]-n,n-bis(prop-2-enyl)pyridine-3-sulfonamide Chemical compound C=CCN(CC=C)S(=O)(=O)C1=CN=CC=C1NS(=O)(=O)\C=C\C1=CC=C(C=2OC3=CC=CC=C3C=2)C=C1 BESNZTZUWXCXMV-NBVRZTHBSA-N 0.000 description 2
- XOCKOFZQQFAHFG-ZRDIBKRKSA-N 4-[[(e)-2-[4-(1-benzofuran-2-yl)phenyl]ethenyl]sulfonylamino]pyridine-3-sulfonamide Chemical compound NS(=O)(=O)C1=CN=CC=C1NS(=O)(=O)\C=C\C1=CC=C(C=2OC3=CC=CC=C3C=2)C=C1 XOCKOFZQQFAHFG-ZRDIBKRKSA-N 0.000 description 2
- ACMQFQJUMVRBPC-UHFFFAOYSA-N 4-[[3-(5-chloro-6-methoxypyridin-3-yl)phenyl]sulfonylamino]-n,n-bis(prop-2-enyl)pyridine-3-sulfonamide Chemical compound C1=C(Cl)C(OC)=NC=C1C1=CC=CC(S(=O)(=O)NC=2C(=CN=CC=2)S(=O)(=O)N(CC=C)CC=C)=C1 ACMQFQJUMVRBPC-UHFFFAOYSA-N 0.000 description 2
- JMAVLPHROCRAJF-CMDGGOBGSA-N 4-chloro-2-[[(e)-2-(2-methoxyphenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound COC1=CC=CC=C1\C=C\S(=O)(=O)NC1=CC(Cl)=CC=C1S(N)(=O)=O JMAVLPHROCRAJF-CMDGGOBGSA-N 0.000 description 2
- GNVINIBMTOOYJC-AATRIKPKSA-N 4-chloro-2-[[(e)-2-(3,4-dichlorophenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=C(Cl)C=C1NS(=O)(=O)\C=C\C1=CC=C(Cl)C(Cl)=C1 GNVINIBMTOOYJC-AATRIKPKSA-N 0.000 description 2
- NRNYVFNYESEQDS-VOTSOKGWSA-N 4-chloro-2-[[(e)-2-(4-chloro-2-methoxyphenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound COC1=CC(Cl)=CC=C1\C=C\S(=O)(=O)NC1=CC(Cl)=CC=C1S(N)(=O)=O NRNYVFNYESEQDS-VOTSOKGWSA-N 0.000 description 2
- SHYNMFKFTLOZMY-BQYQJAHWSA-N 4-chloro-2-[[(e)-2-[4-(difluoromethoxy)phenyl]ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=C(Cl)C=C1NS(=O)(=O)\C=C\C1=CC=C(OC(F)F)C=C1 SHYNMFKFTLOZMY-BQYQJAHWSA-N 0.000 description 2
- YVDNQKAAXFHMIJ-UHFFFAOYSA-N 4-chloro-n,n-bis(prop-2-enyl)pyridine-3-sulfonamide Chemical compound ClC1=CC=NC=C1S(=O)(=O)N(CC=C)CC=C YVDNQKAAXFHMIJ-UHFFFAOYSA-N 0.000 description 2
- XPDBIVWSRQWVGO-AATRIKPKSA-N 4-cyano-2-[[(e)-2-(3,4-dichlorophenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=C(C#N)C=C1NS(=O)(=O)\C=C\C1=CC=C(Cl)C(Cl)=C1 XPDBIVWSRQWVGO-AATRIKPKSA-N 0.000 description 2
- 229960000549 4-dimethylaminophenol Drugs 0.000 description 2
- YYROPELSRYBVMQ-UHFFFAOYSA-N 4-toluenesulfonyl chloride Chemical compound CC1=CC=C(S(Cl)(=O)=O)C=C1 YYROPELSRYBVMQ-UHFFFAOYSA-N 0.000 description 2
- MLIQQYBDEIIAKW-ONEGZZNKSA-N 5-chloro-2-[[(e)-2-(3,4-dichlorophenyl)ethenyl]sulfonylamino]-4-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(Cl)=C(F)C=C1NS(=O)(=O)\C=C\C1=CC=C(Cl)C(Cl)=C1 MLIQQYBDEIIAKW-ONEGZZNKSA-N 0.000 description 2
- BNKGCFNGRLCJST-AATRIKPKSA-N 5-chloro-2-[[(e)-2-(3,4-dichlorophenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(Cl)=CC=C1NS(=O)(=O)\C=C\C1=CC=C(Cl)C(Cl)=C1 BNKGCFNGRLCJST-AATRIKPKSA-N 0.000 description 2
- MXCPXUGVBVEOPY-VOTSOKGWSA-N 5-chloro-2-[[(e)-2-(4-chloro-2-methoxyphenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound COC1=CC(Cl)=CC=C1\C=C\S(=O)(=O)NC1=CC=C(Cl)C=C1S(N)(=O)=O MXCPXUGVBVEOPY-VOTSOKGWSA-N 0.000 description 2
- NLXVQNFULGKRCQ-AATRIKPKSA-N 5-chloro-2-[[(e)-2-(4-chlorophenyl)ethenyl]sulfonylamino]-4-fluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(Cl)=C(F)C=C1NS(=O)(=O)\C=C\C1=CC=C(Cl)C=C1 NLXVQNFULGKRCQ-AATRIKPKSA-N 0.000 description 2
- CUTAPVVLZBKAEC-AATRIKPKSA-N 5-cyano-2-[[(e)-2-(3,4-dichlorophenyl)ethenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(C#N)=CC=C1NS(=O)(=O)\C=C\C1=CC=C(Cl)C(Cl)=C1 CUTAPVVLZBKAEC-AATRIKPKSA-N 0.000 description 2
- BSYNRYMUTXBXSQ-FOQJRBATSA-N 59096-14-9 Chemical compound CC(=O)OC1=CC=CC=C1[14C](O)=O BSYNRYMUTXBXSQ-FOQJRBATSA-N 0.000 description 2
- JFVHGFVNWMMYLD-VOTSOKGWSA-N 6-[[(e)-2-(3,4-dichlorophenyl)ethenyl]sulfonylamino]-2,3-dihydro-1h-indene-5-sulfonamide Chemical compound NS(=O)(=O)C1=CC=2CCCC=2C=C1NS(=O)(=O)\C=C\C1=CC=C(Cl)C(Cl)=C1 JFVHGFVNWMMYLD-VOTSOKGWSA-N 0.000 description 2
- ZZPWPCXHSCJIEN-UHFFFAOYSA-N 7-methyl-1,1-dioxo-4h-1$l^{6},2,4-benzothiadiazin-3-one Chemical compound N1C(=O)NS(=O)(=O)C2=CC(C)=CC=C21 ZZPWPCXHSCJIEN-UHFFFAOYSA-N 0.000 description 2
- PVJOFMIXZYIIIL-UHFFFAOYSA-N 8-(methylsulfamoyl)dibenzothiophene-2-sulfonyl chloride Chemical compound C1=C(S(Cl)(=O)=O)C=C2C3=CC(S(=O)(=O)NC)=CC=C3SC2=C1 PVJOFMIXZYIIIL-UHFFFAOYSA-N 0.000 description 2
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonia chloride Chemical class [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 2
- 229910015845 BBr3 Inorganic materials 0.000 description 2
- HHVIBTZHLRERCL-UHFFFAOYSA-N CS(C)(=O)=O Chemical compound CS(C)(=O)=O HHVIBTZHLRERCL-UHFFFAOYSA-N 0.000 description 2
- 208000005623 Carcinogenesis Diseases 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 2
- 229910020323 ClF3 Inorganic materials 0.000 description 2
- PLUBXMRUUVWRLT-UHFFFAOYSA-N Ethyl methanesulfonate Chemical compound CCOS(C)(=O)=O PLUBXMRUUVWRLT-UHFFFAOYSA-N 0.000 description 2
- UGJMXCAKCUNAIE-UHFFFAOYSA-N Gabapentin Chemical compound OC(=O)CC1(CN)CCCCC1 UGJMXCAKCUNAIE-UHFFFAOYSA-N 0.000 description 2
- MHAJPDPJQMAIIY-UHFFFAOYSA-N Hydrogen peroxide Chemical compound OO MHAJPDPJQMAIIY-UHFFFAOYSA-N 0.000 description 2
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 2
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 description 2
- BAVYZALUXZFZLV-UHFFFAOYSA-N Methylamine Chemical compound NC BAVYZALUXZFZLV-UHFFFAOYSA-N 0.000 description 2
- 241000699670 Mus sp. Species 0.000 description 2
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 2
- 239000007832 Na2SO4 Substances 0.000 description 2
- 102000015439 Phospholipases Human genes 0.000 description 2
- 108010064785 Phospholipases Proteins 0.000 description 2
- 239000002202 Polyethylene glycol Substances 0.000 description 2
- 229940124639 Selective inhibitor Drugs 0.000 description 2
- UIIMBOGNXHQVGW-DEQYMQKBSA-M Sodium bicarbonate-14C Chemical class [Na+].O[14C]([O-])=O UIIMBOGNXHQVGW-DEQYMQKBSA-M 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical class [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 2
- UWDFWVLAHRQSKK-UHFFFAOYSA-N [3-(trifluoromethoxy)phenyl]boronic acid Chemical compound OB(O)C1=CC=CC(OC(F)(F)F)=C1 UWDFWVLAHRQSKK-UHFFFAOYSA-N 0.000 description 2
- 230000002378 acidificating effect Effects 0.000 description 2
- 239000004480 active ingredient Substances 0.000 description 2
- 125000005073 adamantyl group Chemical group C12(CC3CC(CC(C1)C3)C2)* 0.000 description 2
- 150000001299 aldehydes Chemical class 0.000 description 2
- 150000003973 alkyl amines Chemical class 0.000 description 2
- 239000000908 ammonium hydroxide Substances 0.000 description 2
- 150000001448 anilines Chemical class 0.000 description 2
- 238000010171 animal model Methods 0.000 description 2
- 239000008346 aqueous phase Substances 0.000 description 2
- 125000003785 benzimidazolyl group Chemical group N1=C(NC2=C1C=CC=C2)* 0.000 description 2
- 238000010256 biochemical assay Methods 0.000 description 2
- XJHCXCQVJFPJIK-UHFFFAOYSA-M caesium fluoride Chemical compound [F-].[Cs+] XJHCXCQVJFPJIK-UHFFFAOYSA-M 0.000 description 2
- 230000036952 cancer formation Effects 0.000 description 2
- 231100000504 carcinogenesis Toxicity 0.000 description 2
- OSVXSBDYLRYLIG-UHFFFAOYSA-N chlorine dioxide Inorganic materials O=Cl=O OSVXSBDYLRYLIG-UHFFFAOYSA-N 0.000 description 2
- 125000000753 cycloalkyl group Chemical group 0.000 description 2
- ARUKYTASOALXFG-UHFFFAOYSA-N cycloheptylcycloheptane Chemical group C1CCCCCC1C1CCCCCC1 ARUKYTASOALXFG-UHFFFAOYSA-N 0.000 description 2
- 238000010511 deprotection reaction Methods 0.000 description 2
- 238000004807 desolvation Methods 0.000 description 2
- JPPNISPSWXBWTP-UHFFFAOYSA-N dibenzothiophene-2-sulfonyl chloride Chemical compound C1=CC=C2C3=CC(S(=O)(=O)Cl)=CC=C3SC2=C1 JPPNISPSWXBWTP-UHFFFAOYSA-N 0.000 description 2
- IJKVHSBPTUYDLN-UHFFFAOYSA-N dihydroxy(oxo)silane Chemical compound O[Si](O)=O IJKVHSBPTUYDLN-UHFFFAOYSA-N 0.000 description 2
- 229940043279 diisopropylamine Drugs 0.000 description 2
- 230000009977 dual effect Effects 0.000 description 2
- 238000006911 enzymatic reaction Methods 0.000 description 2
- OJFBGMHFDXQGBP-CMDGGOBGSA-N ethyl (e)-1-(4-chlorophenyl)prop-1-ene-2-sulfonate Chemical compound CCOS(=O)(=O)C(\C)=C\C1=CC=C(Cl)C=C1 OJFBGMHFDXQGBP-CMDGGOBGSA-N 0.000 description 2
- PTSWCZGMYVMSAO-BQYQJAHWSA-N ethyl (e)-2-(4-bromophenyl)ethenesulfonate Chemical compound CCOS(=O)(=O)\C=C\C1=CC=C(Br)C=C1 PTSWCZGMYVMSAO-BQYQJAHWSA-N 0.000 description 2
- IZFTWQOESBJAGC-AATRIKPKSA-N ethyl (e)-2-(6-bromopyridin-3-yl)ethenesulfonate Chemical compound CCOS(=O)(=O)\C=C\C1=CC=C(Br)N=C1 IZFTWQOESBJAGC-AATRIKPKSA-N 0.000 description 2
- ZOPBOKJARHTXBX-UHFFFAOYSA-N ethyl 2-(4-chloro-2-methoxyphenyl)ethenesulfonate Chemical compound CCOS(=O)(=O)C=CC1=CC=C(Cl)C=C1OC ZOPBOKJARHTXBX-UHFFFAOYSA-N 0.000 description 2
- GSQVONWMKFXCKQ-UHFFFAOYSA-N ethyl 4-[(2,3-dichlorophenyl)sulfonylamino]-3-sulfamoylbenzoate Chemical compound NS(=O)(=O)C1=CC(C(=O)OCC)=CC=C1NS(=O)(=O)C1=CC=CC(Cl)=C1Cl GSQVONWMKFXCKQ-UHFFFAOYSA-N 0.000 description 2
- 238000009472 formulation Methods 0.000 description 2
- 125000000524 functional group Chemical group 0.000 description 2
- 239000007789 gas Substances 0.000 description 2
- 239000008187 granular material Substances 0.000 description 2
- 125000005842 heteroatom Chemical group 0.000 description 2
- 238000005984 hydrogenation reaction Methods 0.000 description 2
- 125000003453 indazolyl group Chemical group N1N=C(C2=C1C=CC=C2)* 0.000 description 2
- 125000003454 indenyl group Chemical group C1(C=CC2=CC=CC=C12)* 0.000 description 2
- 238000005342 ion exchange Methods 0.000 description 2
- 125000000842 isoxazolyl group Chemical group 0.000 description 2
- 239000010410 layer Substances 0.000 description 2
- 239000003446 ligand Substances 0.000 description 2
- HSZCZNFXUDYRKD-UHFFFAOYSA-M lithium iodide Chemical compound [Li+].[I-] HSZCZNFXUDYRKD-UHFFFAOYSA-M 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- IZQCHVGTQUPMMI-UHFFFAOYSA-N methyl 4-[2-(4-chloro-2-methoxyphenyl)ethylsulfonylamino]-3-sulfamoylbenzoate Chemical compound NS(=O)(=O)C1=CC(C(=O)OC)=CC=C1NS(=O)(=O)CCC1=CC=C(Cl)C=C1OC IZQCHVGTQUPMMI-UHFFFAOYSA-N 0.000 description 2
- 125000002757 morpholinyl group Chemical group 0.000 description 2
- RNFXNHPYRVCWLZ-UHFFFAOYSA-N n-methyldibenzothiophene-2-sulfonamide Chemical compound C1=CC=C2C3=CC(S(=O)(=O)NC)=CC=C3SC2=C1 RNFXNHPYRVCWLZ-UHFFFAOYSA-N 0.000 description 2
- MCSAJNNLRCFZED-UHFFFAOYSA-N nitroethane Chemical compound CC[N+]([O-])=O MCSAJNNLRCFZED-UHFFFAOYSA-N 0.000 description 2
- 239000000041 non-steroidal anti-inflammatory agent Substances 0.000 description 2
- 125000002971 oxazolyl group Chemical group 0.000 description 2
- RZXMPPFPUUCRFN-UHFFFAOYSA-N p-toluidine Chemical compound CC1=CC=C(N)C=C1 RZXMPPFPUUCRFN-UHFFFAOYSA-N 0.000 description 2
- 229910052763 palladium Inorganic materials 0.000 description 2
- 239000000546 pharmaceutical excipient Substances 0.000 description 2
- HXITXNWTGFUOAU-UHFFFAOYSA-N phenylboronic acid Chemical compound OB(O)C1=CC=CC=C1 HXITXNWTGFUOAU-UHFFFAOYSA-N 0.000 description 2
- 229920001223 polyethylene glycol Polymers 0.000 description 2
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 2
- 150000003180 prostaglandins Chemical class 0.000 description 2
- 238000000425 proton nuclear magnetic resonance spectrum Methods 0.000 description 2
- YCWRZEKTBDOZSQ-UHFFFAOYSA-N pyridin-2-ylmethyl 4-methylbenzenesulfonate Chemical compound C1=CC(C)=CC=C1S(=O)(=O)OCC1=CC=CC=N1 YCWRZEKTBDOZSQ-UHFFFAOYSA-N 0.000 description 2
- 125000000168 pyrrolyl group Chemical group 0.000 description 2
- 125000005493 quinolyl group Chemical group 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- XIIOFHFUYBLOLW-UHFFFAOYSA-N selpercatinib Chemical compound OC(COC=1C=C(C=2N(C=1)N=CC=2C#N)C=1C=NC(=CC=1)N1CC2N(C(C1)C2)CC=1C=NC(=CC=1)OC)(C)C XIIOFHFUYBLOLW-UHFFFAOYSA-N 0.000 description 2
- XGVXKJKTISMIOW-ZDUSSCGKSA-N simurosertib Chemical compound N1N=CC(C=2SC=3C(=O)NC(=NC=3C=2)[C@H]2N3CCC(CC3)C2)=C1C XGVXKJKTISMIOW-ZDUSSCGKSA-N 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 229910052708 sodium Inorganic materials 0.000 description 2
- 229910000104 sodium hydride Inorganic materials 0.000 description 2
- 239000004289 sodium hydrogen sulphite Substances 0.000 description 2
- GEHJYWRUCIMESM-UHFFFAOYSA-L sodium sulfite Chemical compound [Na+].[Na+].[O-]S([O-])=O GEHJYWRUCIMESM-UHFFFAOYSA-L 0.000 description 2
- BVAHWQAYONTDDL-UHFFFAOYSA-M sodium;2-(4-bromophenyl)ethanesulfonate Chemical compound [Na+].[O-]S(=O)(=O)CCC1=CC=C(Br)C=C1 BVAHWQAYONTDDL-UHFFFAOYSA-M 0.000 description 2
- 239000000758 substrate Substances 0.000 description 2
- 235000000346 sugar Nutrition 0.000 description 2
- NVBFHJWHLNUMCV-UHFFFAOYSA-N sulfamide Chemical compound NS(N)(=O)=O NVBFHJWHLNUMCV-UHFFFAOYSA-N 0.000 description 2
- HXJUTPCZVOIRIF-UHFFFAOYSA-N sulfolane Chemical compound O=S1(=O)CCCC1 HXJUTPCZVOIRIF-UHFFFAOYSA-N 0.000 description 2
- 239000006188 syrup Substances 0.000 description 2
- 235000020357 syrup Nutrition 0.000 description 2
- 125000001712 tetrahydronaphthyl group Chemical group C1(CCCC2=CC=CC=C12)* 0.000 description 2
- 238000004809 thin layer chromatography Methods 0.000 description 2
- RZWIIPASKMUIAC-VQTJNVASSA-N thromboxane Chemical compound CCCCCCCC[C@H]1OCCC[C@@H]1CCCCCCC RZWIIPASKMUIAC-VQTJNVASSA-N 0.000 description 2
- 238000013389 whole blood assay Methods 0.000 description 2
- 238000010626 work up procedure Methods 0.000 description 2
- VCGRFBXVSFAGGA-UHFFFAOYSA-N (1,1-dioxo-1,4-thiazinan-4-yl)-[6-[[3-(4-fluorophenyl)-5-methyl-1,2-oxazol-4-yl]methoxy]pyridin-3-yl]methanone Chemical compound CC=1ON=C(C=2C=CC(F)=CC=2)C=1COC(N=C1)=CC=C1C(=O)N1CCS(=O)(=O)CC1 VCGRFBXVSFAGGA-UHFFFAOYSA-N 0.000 description 1
- TYIKXPOMOYDGCS-UHFFFAOYSA-N (2,3-dichlorophenyl)boronic acid Chemical compound OB(O)C1=CC=CC(Cl)=C1Cl TYIKXPOMOYDGCS-UHFFFAOYSA-N 0.000 description 1
- NNTFPBXQPOQRBT-UHFFFAOYSA-N (2,5-dichlorophenyl)boronic acid Chemical compound OB(O)C1=CC(Cl)=CC=C1Cl NNTFPBXQPOQRBT-UHFFFAOYSA-N 0.000 description 1
- LOGFVTREOLYCPF-KXNHARMFSA-N (2s,3r)-2-[[(2r)-1-[(2s)-2,6-diaminohexanoyl]pyrrolidine-2-carbonyl]amino]-3-hydroxybutanoic acid Chemical compound C[C@@H](O)[C@@H](C(O)=O)NC(=O)[C@H]1CCCN1C(=O)[C@@H](N)CCCCN LOGFVTREOLYCPF-KXNHARMFSA-N 0.000 description 1
- MAYZWDRUFKUGGP-VIFPVBQESA-N (3s)-1-[5-tert-butyl-3-[(1-methyltetrazol-5-yl)methyl]triazolo[4,5-d]pyrimidin-7-yl]pyrrolidin-3-ol Chemical compound CN1N=NN=C1CN1C2=NC(C(C)(C)C)=NC(N3C[C@@H](O)CC3)=C2N=N1 MAYZWDRUFKUGGP-VIFPVBQESA-N 0.000 description 1
- 125000006656 (C2-C4) alkenyl group Chemical group 0.000 description 1
- 125000006650 (C2-C4) alkynyl group Chemical group 0.000 description 1
- PXGPLTODNUVGFL-BRIYLRKRSA-N (E,Z)-(1R,2R,3R,5S)-7-(3,5-Dihydroxy-2-((3S)-(3-hydroxy-1-octenyl))cyclopentyl)-5-heptenoic acid Chemical compound CCCCC[C@H](O)C=C[C@H]1[C@H](O)C[C@H](O)[C@@H]1CC=CCCCC(O)=O PXGPLTODNUVGFL-BRIYLRKRSA-N 0.000 description 1
- FJHXODLLSVBQQO-VOTSOKGWSA-N (e)-1-(4-chlorophenyl)prop-1-ene-2-sulfonic acid Chemical class OS(=O)(=O)C(/C)=C/C1=CC=C(Cl)C=C1 FJHXODLLSVBQQO-VOTSOKGWSA-N 0.000 description 1
- DODCULSWGAULOG-VOTSOKGWSA-N (e)-1-(4-chlorophenyl)prop-1-ene-2-sulfonyl chloride Chemical compound ClS(=O)(=O)C(/C)=C/C1=CC=C(Cl)C=C1 DODCULSWGAULOG-VOTSOKGWSA-N 0.000 description 1
- CIJAPQWSEHDXFM-AATRIKPKSA-N (e)-2-(2-fluorophenyl)ethenesulfonyl chloride Chemical compound FC1=CC=CC=C1\C=C\S(Cl)(=O)=O CIJAPQWSEHDXFM-AATRIKPKSA-N 0.000 description 1
- DEDFXMJWTVBWDG-VOTSOKGWSA-N (e)-2-(2-methoxyphenyl)ethenesulfonyl chloride Chemical compound COC1=CC=CC=C1\C=C\S(Cl)(=O)=O DEDFXMJWTVBWDG-VOTSOKGWSA-N 0.000 description 1
- HVBRPCXLQIUQLT-AATRIKPKSA-N (e)-2-(4-chlorophenyl)ethenesulfonyl chloride Chemical compound ClC1=CC=C(\C=C\S(Cl)(=O)=O)C=C1 HVBRPCXLQIUQLT-AATRIKPKSA-N 0.000 description 1
- QLUZUUFPDCUXCT-ZHACJKMWSA-N (e)-2-(4-phenylphenyl)ethenesulfonyl chloride Chemical compound C1=CC(/C=C/S(=O)(=O)Cl)=CC=C1C1=CC=CC=C1 QLUZUUFPDCUXCT-ZHACJKMWSA-N 0.000 description 1
- CJYYDBBOPIFULK-SNAWJCMRSA-N (e)-2-(5-bromo-2-methoxyphenyl)ethenesulfonyl chloride Chemical compound COC1=CC=C(Br)C=C1\C=C\S(Cl)(=O)=O CJYYDBBOPIFULK-SNAWJCMRSA-N 0.000 description 1
- UXWAUMKUSFSCTH-SNAWJCMRSA-N (e)-2-[3-(trifluoromethyl)phenyl]ethenesulfonyl chloride Chemical compound FC(F)(F)C1=CC=CC(\C=C\S(Cl)(=O)=O)=C1 UXWAUMKUSFSCTH-SNAWJCMRSA-N 0.000 description 1
- BWOMTUUVBIZVTD-AATRIKPKSA-N (e)-2-[4-(difluoromethoxy)phenyl]ethenesulfonyl chloride Chemical compound FC(F)OC1=CC=C(\C=C\S(Cl)(=O)=O)C=C1 BWOMTUUVBIZVTD-AATRIKPKSA-N 0.000 description 1
- WWOUUMYKLNEGCG-VQHVLOKHSA-N (e)-2-[4-(furan-2-yl)phenyl]ethenesulfonyl chloride Chemical compound C1=CC(/C=C/S(=O)(=O)Cl)=CC=C1C1=CC=CO1 WWOUUMYKLNEGCG-VQHVLOKHSA-N 0.000 description 1
- UKGJZDSUJSPAJL-YPUOHESYSA-N (e)-n-[(1r)-1-[3,5-difluoro-4-(methanesulfonamido)phenyl]ethyl]-3-[2-propyl-6-(trifluoromethyl)pyridin-3-yl]prop-2-enamide Chemical compound CCCC1=NC(C(F)(F)F)=CC=C1\C=C\C(=O)N[C@H](C)C1=CC(F)=C(NS(C)(=O)=O)C(F)=C1 UKGJZDSUJSPAJL-YPUOHESYSA-N 0.000 description 1
- ZGYIXVSQHOKQRZ-COIATFDQSA-N (e)-n-[4-[3-chloro-4-(pyridin-2-ylmethoxy)anilino]-3-cyano-7-[(3s)-oxolan-3-yl]oxyquinolin-6-yl]-4-(dimethylamino)but-2-enamide Chemical compound N#CC1=CN=C2C=C(O[C@@H]3COCC3)C(NC(=O)/C=C/CN(C)C)=CC2=C1NC(C=C1Cl)=CC=C1OCC1=CC=CC=N1 ZGYIXVSQHOKQRZ-COIATFDQSA-N 0.000 description 1
- MOWXJLUYGFNTAL-DEOSSOPVSA-N (s)-[2-chloro-4-fluoro-5-(7-morpholin-4-ylquinazolin-4-yl)phenyl]-(6-methoxypyridazin-3-yl)methanol Chemical compound N1=NC(OC)=CC=C1[C@@H](O)C1=CC(C=2C3=CC=C(C=C3N=CN=2)N2CCOCC2)=C(F)C=C1Cl MOWXJLUYGFNTAL-DEOSSOPVSA-N 0.000 description 1
- KZPYGQFFRCFCPP-UHFFFAOYSA-N 1,1'-bis(diphenylphosphino)ferrocene Chemical compound [Fe+2].C1=CC=C[C-]1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=C[C-]1P(C=1C=CC=CC=1)C1=CC=CC=C1 KZPYGQFFRCFCPP-UHFFFAOYSA-N 0.000 description 1
- 150000008349 1,2,4-benzothiadiazines Chemical class 0.000 description 1
- QFMZQPDHXULLKC-UHFFFAOYSA-N 1,2-bis(diphenylphosphino)ethane Chemical compound C=1C=CC=CC=1P(C=1C=CC=CC=1)CCP(C=1C=CC=CC=1)C1=CC=CC=C1 QFMZQPDHXULLKC-UHFFFAOYSA-N 0.000 description 1
- KEQGZUUPPQEDPF-UHFFFAOYSA-N 1,3-dichloro-5,5-dimethylimidazolidine-2,4-dione Chemical compound CC1(C)N(Cl)C(=O)N(Cl)C1=O KEQGZUUPPQEDPF-UHFFFAOYSA-N 0.000 description 1
- VVSASNKOFCZVES-UHFFFAOYSA-N 1,3-dimethyl-1,3-diazinane-2,4,6-trione Chemical compound CN1C(=O)CC(=O)N(C)C1=O VVSASNKOFCZVES-UHFFFAOYSA-N 0.000 description 1
- JPRPJUMQRZTTED-UHFFFAOYSA-N 1,3-dioxolanyl Chemical group [CH]1OCCO1 JPRPJUMQRZTTED-UHFFFAOYSA-N 0.000 description 1
- 125000005940 1,4-dioxanyl group Chemical group 0.000 description 1
- APWRZPQBPCAXFP-UHFFFAOYSA-N 1-(1-oxo-2H-isoquinolin-5-yl)-5-(trifluoromethyl)-N-[2-(trifluoromethyl)pyridin-4-yl]pyrazole-4-carboxamide Chemical compound O=C1NC=CC2=C(C=CC=C12)N1N=CC(=C1C(F)(F)F)C(=O)NC1=CC(=NC=C1)C(F)(F)F APWRZPQBPCAXFP-UHFFFAOYSA-N 0.000 description 1
- VXNZUUAINFGPBY-UHFFFAOYSA-N 1-Butene Chemical group CCC=C VXNZUUAINFGPBY-UHFFFAOYSA-N 0.000 description 1
- ABDDQTDRAHXHOC-QMMMGPOBSA-N 1-[(7s)-5,7-dihydro-4h-thieno[2,3-c]pyran-7-yl]-n-methylmethanamine Chemical compound CNC[C@@H]1OCCC2=C1SC=C2 ABDDQTDRAHXHOC-QMMMGPOBSA-N 0.000 description 1
- PKRRNTJIHGOMRC-UHFFFAOYSA-N 1-benzofuran-2-ylboronic acid Chemical compound C1=CC=C2OC(B(O)O)=CC2=C1 PKRRNTJIHGOMRC-UHFFFAOYSA-N 0.000 description 1
- APTDRDYSJZQPPI-UHFFFAOYSA-N 1-bromo-4-(2-bromoethyl)benzene Chemical compound BrCCC1=CC=C(Br)C=C1 APTDRDYSJZQPPI-UHFFFAOYSA-N 0.000 description 1
- WQDGTJOEMPEHHL-UHFFFAOYSA-N 1-chloro-4-prop-1-en-2-ylbenzene Chemical compound CC(=C)C1=CC=C(Cl)C=C1 WQDGTJOEMPEHHL-UHFFFAOYSA-N 0.000 description 1
- XTKBMZQCDBHHKY-UHFFFAOYSA-N 1-ethynyl-4-(trifluoromethyl)benzene Chemical compound FC(F)(F)C1=CC=C(C#C)C=C1 XTKBMZQCDBHHKY-UHFFFAOYSA-N 0.000 description 1
- LEWZOBYWGWKNCK-UHFFFAOYSA-N 2,3-dihydro-1h-inden-5-amine Chemical compound NC1=CC=C2CCCC2=C1 LEWZOBYWGWKNCK-UHFFFAOYSA-N 0.000 description 1
- SWLIXMXSCZYVTQ-UHFFFAOYSA-N 2,3-dihydro-1h-indene-5-sulfonyl chloride Chemical compound ClS(=O)(=O)C1=CC=C2CCCC2=C1 SWLIXMXSCZYVTQ-UHFFFAOYSA-N 0.000 description 1
- AUJUFTDORPTZBS-UHFFFAOYSA-N 2-(2-chlorophenyl)ethenesulfonyl chloride Chemical compound ClC1=CC=CC=C1C=CS(Cl)(=O)=O AUJUFTDORPTZBS-UHFFFAOYSA-N 0.000 description 1
- XEJVDLVXIOIKQT-UHFFFAOYSA-N 2-(3,4-dichlorophenyl)ethanesulfonyl chloride Chemical compound ClC1=CC=C(CCS(Cl)(=O)=O)C=C1Cl XEJVDLVXIOIKQT-UHFFFAOYSA-N 0.000 description 1
- XVYPJQUTCSNGMH-UHFFFAOYSA-N 2-(4-chlorophenyl)prop-1-ene-1-sulfonyl chloride Chemical compound ClS(=O)(=O)C=C(C)C1=CC=C(Cl)C=C1 XVYPJQUTCSNGMH-UHFFFAOYSA-N 0.000 description 1
- AGLPAQXTQLTNFW-UHFFFAOYSA-N 2-(4-chlorophenyl)prop-2-ene-1-sulfonyl chloride Chemical compound ClC1=CC=C(C(=C)CS(Cl)(=O)=O)C=C1 AGLPAQXTQLTNFW-UHFFFAOYSA-N 0.000 description 1
- KCZWMHSIBCZXSN-UHFFFAOYSA-N 2-(5,7-dimethyl-2h-[1,2,4]triazolo[1,5-a]pyrimidin-1-yl)-n-methoxy-n-methylbenzenesulfonamide Chemical compound CON(C)S(=O)(=O)C1=CC=CC=C1N1N2C(C)=CC(C)=NC2=NC1 KCZWMHSIBCZXSN-UHFFFAOYSA-N 0.000 description 1
- GHXVJWOAPMXUAY-ONEGZZNKSA-N 2-[[(e)-2-(3,4-dichlorophenyl)ethenyl]sulfonylamino]-3,5-difluorobenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC(F)=CC(F)=C1NS(=O)(=O)\C=C\C1=CC=C(Cl)C(Cl)=C1 GHXVJWOAPMXUAY-ONEGZZNKSA-N 0.000 description 1
- PNBAVBCBCPRSMK-BQYQJAHWSA-N 2-[[(e)-2-(4-chloro-2-methoxyphenyl)ethenyl]sulfonylamino]-5-methylbenzenesulfonamide Chemical compound COC1=CC(Cl)=CC=C1\C=C\S(=O)(=O)NC1=CC=C(C)C=C1S(N)(=O)=O PNBAVBCBCPRSMK-BQYQJAHWSA-N 0.000 description 1
- GSQKPIUDORXKER-UHFFFAOYSA-N 2-[[3-(5-chloropyridin-3-yl)phenyl]sulfonylamino]benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1NS(=O)(=O)C1=CC=CC(C=2C=C(Cl)C=NC=2)=C1 GSQKPIUDORXKER-UHFFFAOYSA-N 0.000 description 1
- PQSIISBSDXDVLD-UHFFFAOYSA-N 2-amino-4,5-difluorobenzenesulfonamide Chemical compound NC1=CC(F)=C(F)C=C1S(N)(=O)=O PQSIISBSDXDVLD-UHFFFAOYSA-N 0.000 description 1
- BFRSTNOZWFCLRI-UHFFFAOYSA-N 2-amino-4-iodobenzenesulfonamide Chemical compound NC1=CC(I)=CC=C1S(N)(=O)=O BFRSTNOZWFCLRI-UHFFFAOYSA-N 0.000 description 1
- MFZOWSMDIXQZDO-UHFFFAOYSA-N 2-amino-5-fluoro-4-methoxybenzenesulfonamide Chemical compound COC1=CC(N)=C(S(N)(=O)=O)C=C1F MFZOWSMDIXQZDO-UHFFFAOYSA-N 0.000 description 1
- YKQPZGMHAXHTJQ-UHFFFAOYSA-N 2-amino-5-fluorobenzenesulfonamide Chemical compound NC1=CC=C(F)C=C1S(N)(=O)=O YKQPZGMHAXHTJQ-UHFFFAOYSA-N 0.000 description 1
- TZPOFKNGJJYOPZ-UHFFFAOYSA-N 2-amino-5-iodobenzenesulfonamide Chemical compound NC1=CC=C(I)C=C1S(N)(=O)=O TZPOFKNGJJYOPZ-UHFFFAOYSA-N 0.000 description 1
- XZFGOCSYBRXBPU-UHFFFAOYSA-N 2-amino-6-fluorobenzenesulfonamide Chemical compound NC1=CC=CC(F)=C1S(N)(=O)=O XZFGOCSYBRXBPU-UHFFFAOYSA-N 0.000 description 1
- IJICRIUYZZESMW-UHFFFAOYSA-N 2-bromodibenzothiophene Chemical compound C1=CC=C2C3=CC(Br)=CC=C3SC2=C1 IJICRIUYZZESMW-UHFFFAOYSA-N 0.000 description 1
- OFPRGOSJWUNETN-UHFFFAOYSA-N 2-chloro-3-nitrothiophene Chemical compound [O-][N+](=O)C=1C=CSC=1Cl OFPRGOSJWUNETN-UHFFFAOYSA-N 0.000 description 1
- QZBOWJXHXLIMGD-UHFFFAOYSA-N 2-cyanobenzenesulfonamide Chemical class NS(=O)(=O)C1=CC=CC=C1C#N QZBOWJXHXLIMGD-UHFFFAOYSA-N 0.000 description 1
- JKHHRSIUFVAEOY-UHFFFAOYSA-N 2-iodobenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1I JKHHRSIUFVAEOY-UHFFFAOYSA-N 0.000 description 1
- VYWCRVNLPOVYJH-UHFFFAOYSA-N 2-methoxy-6-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)pyridine Chemical compound COC1=CC=CC(B2OC(C)(C)C(C)(C)O2)=N1 VYWCRVNLPOVYJH-UHFFFAOYSA-N 0.000 description 1
- QRKDOAWSBBGNLE-UHFFFAOYSA-N 2h-1,2,4-benzothiadiazine Chemical class C1=CC=C2N=CNSC2=C1 QRKDOAWSBBGNLE-UHFFFAOYSA-N 0.000 description 1
- FLHZJMZNIOCRSX-UHFFFAOYSA-N 3,3-dimethylbut-1-ynyl-di(propan-2-yloxy)borane Chemical compound CC(C)OB(OC(C)C)C#CC(C)(C)C FLHZJMZNIOCRSX-UHFFFAOYSA-N 0.000 description 1
- NYIBPWGZGSXURD-UHFFFAOYSA-N 3,4-dichlorobenzenesulfonyl chloride Chemical compound ClC1=CC=C(S(Cl)(=O)=O)C=C1Cl NYIBPWGZGSXURD-UHFFFAOYSA-N 0.000 description 1
- HCDMJFOHIXMBOV-UHFFFAOYSA-N 3-(2,6-difluoro-3,5-dimethoxyphenyl)-1-ethyl-8-(morpholin-4-ylmethyl)-4,7-dihydropyrrolo[4,5]pyrido[1,2-d]pyrimidin-2-one Chemical compound C=1C2=C3N(CC)C(=O)N(C=4C(=C(OC)C=C(OC)C=4F)F)CC3=CN=C2NC=1CN1CCOCC1 HCDMJFOHIXMBOV-UHFFFAOYSA-N 0.000 description 1
- ZZGWJBAJNLBBRB-UHFFFAOYSA-N 3-(2-methyl-1,3-thiazol-4-yl)benzenesulfonyl chloride Chemical compound S1C(C)=NC(C=2C=C(C=CC=2)S(Cl)(=O)=O)=C1 ZZGWJBAJNLBBRB-UHFFFAOYSA-N 0.000 description 1
- BYHQTRFJOGIQAO-GOSISDBHSA-N 3-(4-bromophenyl)-8-[(2R)-2-hydroxypropyl]-1-[(3-methoxyphenyl)methyl]-1,3,8-triazaspiro[4.5]decan-2-one Chemical compound C[C@H](CN1CCC2(CC1)CN(C(=O)N2CC3=CC(=CC=C3)OC)C4=CC=C(C=C4)Br)O BYHQTRFJOGIQAO-GOSISDBHSA-N 0.000 description 1
- YGYGASJNJTYNOL-CQSZACIVSA-N 3-[(4r)-2,2-dimethyl-1,1-dioxothian-4-yl]-5-(4-fluorophenyl)-1h-indole-7-carboxamide Chemical compound C1CS(=O)(=O)C(C)(C)C[C@@H]1C1=CNC2=C(C(N)=O)C=C(C=3C=CC(F)=CC=3)C=C12 YGYGASJNJTYNOL-CQSZACIVSA-N 0.000 description 1
- WNEODWDFDXWOLU-QHCPKHFHSA-N 3-[3-(hydroxymethyl)-4-[1-methyl-5-[[5-[(2s)-2-methyl-4-(oxetan-3-yl)piperazin-1-yl]pyridin-2-yl]amino]-6-oxopyridin-3-yl]pyridin-2-yl]-7,7-dimethyl-1,2,6,8-tetrahydrocyclopenta[3,4]pyrrolo[3,5-b]pyrazin-4-one Chemical compound C([C@@H](N(CC1)C=2C=NC(NC=3C(N(C)C=C(C=3)C=3C(=C(N4C(C5=CC=6CC(C)(C)CC=6N5CC4)=O)N=CC=3)CO)=O)=CC=2)C)N1C1COC1 WNEODWDFDXWOLU-QHCPKHFHSA-N 0.000 description 1
- SRVXSISGYBMIHR-UHFFFAOYSA-N 3-[3-[3-(2-amino-2-oxoethyl)phenyl]-5-chlorophenyl]-3-(5-methyl-1,3-thiazol-2-yl)propanoic acid Chemical compound S1C(C)=CN=C1C(CC(O)=O)C1=CC(Cl)=CC(C=2C=C(CC(N)=O)C=CC=2)=C1 SRVXSISGYBMIHR-UHFFFAOYSA-N 0.000 description 1
- BMYNFMYTOJXKLE-UHFFFAOYSA-N 3-azaniumyl-2-hydroxypropanoate Chemical compound NCC(O)C(O)=O BMYNFMYTOJXKLE-UHFFFAOYSA-N 0.000 description 1
- LIRQNYQIFFLGIE-UHFFFAOYSA-N 3-chloro-2-fluorobenzenesulfonyl chloride Chemical compound FC1=C(Cl)C=CC=C1S(Cl)(=O)=O LIRQNYQIFFLGIE-UHFFFAOYSA-N 0.000 description 1
- YYVAMBNPSIGVGI-SECBINFHSA-N 3-chloro-4-[[(2r)-3,3,3-trifluoro-2-hydroxy-2-methylpropanoyl]amino]benzenesulfonyl chloride Chemical compound FC(F)(F)[C@@](O)(C)C(=O)NC1=CC=C(S(Cl)(=O)=O)C=C1Cl YYVAMBNPSIGVGI-SECBINFHSA-N 0.000 description 1
- HRXULFWGEYSULN-UHFFFAOYSA-N 3-chloro-n-hydroxy-1-benzothiophene-2-carboxamide Chemical compound C1=CC=C2C(Cl)=C(C(=O)NO)SC2=C1 HRXULFWGEYSULN-UHFFFAOYSA-N 0.000 description 1
- IVTWLTRKVRJPNG-UHFFFAOYSA-N 4,5-dichlorothiophene-2-sulfonyl chloride Chemical compound ClC=1C=C(S(Cl)(=O)=O)SC=1Cl IVTWLTRKVRJPNG-UHFFFAOYSA-N 0.000 description 1
- OIZXQBONLHTKDY-UHFFFAOYSA-N 4-[(2-chloro-1,3-thiazol-5-yl)methoxy]benzenesulfonyl chloride Chemical compound S1C(Cl)=NC=C1COC1=CC=C(S(Cl)(=O)=O)C=C1 OIZXQBONLHTKDY-UHFFFAOYSA-N 0.000 description 1
- YFCIFWOJYYFDQP-PTWZRHHISA-N 4-[3-amino-6-[(1S,3S,4S)-3-fluoro-4-hydroxycyclohexyl]pyrazin-2-yl]-N-[(1S)-1-(3-bromo-5-fluorophenyl)-2-(methylamino)ethyl]-2-fluorobenzamide Chemical compound CNC[C@@H](NC(=O)c1ccc(cc1F)-c1nc(cnc1N)[C@H]1CC[C@H](O)[C@@H](F)C1)c1cc(F)cc(Br)c1 YFCIFWOJYYFDQP-PTWZRHHISA-N 0.000 description 1
- XYWIPYBIIRTJMM-IBGZPJMESA-N 4-[[(2S)-2-[4-[5-chloro-2-[4-(trifluoromethyl)triazol-1-yl]phenyl]-5-methoxy-2-oxopyridin-1-yl]butanoyl]amino]-2-fluorobenzamide Chemical compound CC[C@H](N1C=C(OC)C(=CC1=O)C1=C(C=CC(Cl)=C1)N1C=C(N=N1)C(F)(F)F)C(=O)NC1=CC(F)=C(C=C1)C(N)=O XYWIPYBIIRTJMM-IBGZPJMESA-N 0.000 description 1
- GFFHQNZNTXHXOE-UHFFFAOYSA-N 4-[[3-(3,4-dichlorophenyl)phenyl]sulfonylamino]-3-sulfamoylbenzoic acid Chemical compound NS(=O)(=O)C1=CC(C(O)=O)=CC=C1NS(=O)(=O)C1=CC=CC(C=2C=C(Cl)C(Cl)=CC=2)=C1 GFFHQNZNTXHXOE-UHFFFAOYSA-N 0.000 description 1
- KVCQTKNUUQOELD-UHFFFAOYSA-N 4-amino-n-[1-(3-chloro-2-fluoroanilino)-6-methylisoquinolin-5-yl]thieno[3,2-d]pyrimidine-7-carboxamide Chemical compound N=1C=CC2=C(NC(=O)C=3C4=NC=NC(N)=C4SC=3)C(C)=CC=C2C=1NC1=CC=CC(Cl)=C1F KVCQTKNUUQOELD-UHFFFAOYSA-N 0.000 description 1
- ZRYZBQLXDKPBDU-UHFFFAOYSA-N 4-bromobenzaldehyde Chemical compound BrC1=CC=C(C=O)C=C1 ZRYZBQLXDKPBDU-UHFFFAOYSA-N 0.000 description 1
- IVBZHFWJBYTRNG-UHFFFAOYSA-N 4-chloro-2-methoxybenzaldehyde Chemical compound COC1=CC(Cl)=CC=C1C=O IVBZHFWJBYTRNG-UHFFFAOYSA-N 0.000 description 1
- AVPYQKSLYISFPO-UHFFFAOYSA-N 4-chlorobenzaldehyde Chemical compound ClC1=CC=C(C=O)C=C1 AVPYQKSLYISFPO-UHFFFAOYSA-N 0.000 description 1
- DGIINIBYHCODIH-UHFFFAOYSA-N 4-chloropyridine-3-sulfonamide Chemical compound NS(=O)(=O)C1=CN=CC=C1Cl DGIINIBYHCODIH-UHFFFAOYSA-N 0.000 description 1
- WOYZXEVUWXQVNV-UHFFFAOYSA-N 4-phenoxyaniline Chemical compound C1=CC(N)=CC=C1OC1=CC=CC=C1 WOYZXEVUWXQVNV-UHFFFAOYSA-N 0.000 description 1
- ALBQXDHCMLLQMB-UHFFFAOYSA-N 4-phenylbenzenesulfonyl chloride Chemical compound C1=CC(S(=O)(=O)Cl)=CC=C1C1=CC=CC=C1 ALBQXDHCMLLQMB-UHFFFAOYSA-N 0.000 description 1
- IRPVABHDSJVBNZ-RTHVDDQRSA-N 5-[1-(cyclopropylmethyl)-5-[(1R,5S)-3-(oxetan-3-yl)-3-azabicyclo[3.1.0]hexan-6-yl]pyrazol-3-yl]-3-(trifluoromethyl)pyridin-2-amine Chemical compound C1=C(C(F)(F)F)C(N)=NC=C1C1=NN(CC2CC2)C(C2[C@@H]3CN(C[C@@H]32)C2COC2)=C1 IRPVABHDSJVBNZ-RTHVDDQRSA-N 0.000 description 1
- BWAKFBCMBNHGPF-CMDGGOBGSA-N 5-[[(e)-2-(3,4-dichlorophenyl)ethenyl]sulfonylamino]-2,3-dihydro-1h-indene-4-sulfonamide Chemical compound NS(=O)(=O)C1=C2CCCC2=CC=C1NS(=O)(=O)\C=C\C1=CC=C(Cl)C(Cl)=C1 BWAKFBCMBNHGPF-CMDGGOBGSA-N 0.000 description 1
- UPCXCMHPWULQQB-WYMLVPIESA-N 5-[[(e)-2-[4-(furan-2-yl)phenyl]ethenyl]sulfonylamino]-2,3-dihydro-1h-indene-4-sulfonamide Chemical compound NS(=O)(=O)C1=C2CCCC2=CC=C1NS(=O)(=O)\C=C\C(C=C1)=CC=C1C1=CC=CO1 UPCXCMHPWULQQB-WYMLVPIESA-N 0.000 description 1
- SORSTNOXGOXWAO-UHFFFAOYSA-N 5-chlorothiophene-2-sulfonyl chloride Chemical compound ClC1=CC=C(S(Cl)(=O)=O)S1 SORSTNOXGOXWAO-UHFFFAOYSA-N 0.000 description 1
- VJOWMORERYNYON-UHFFFAOYSA-N 5-ethenyl-2-methylpyridine Chemical compound CC1=CC=C(C=C)C=N1 VJOWMORERYNYON-UHFFFAOYSA-N 0.000 description 1
- KCBWAFJCKVKYHO-UHFFFAOYSA-N 6-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-1-[[4-[1-propan-2-yl-4-(trifluoromethyl)imidazol-2-yl]phenyl]methyl]pyrazolo[3,4-d]pyrimidine Chemical compound C1(CC1)C1=NC=NC(=C1C1=NC=C2C(=N1)N(N=C2)CC1=CC=C(C=C1)C=1N(C=C(N=1)C(F)(F)F)C(C)C)OC KCBWAFJCKVKYHO-UHFFFAOYSA-N 0.000 description 1
- PVUKGNBRJFTFNJ-UHFFFAOYSA-N 6-bromopyridine-3-carbaldehyde Chemical compound BrC1=CC=C(C=O)C=N1 PVUKGNBRJFTFNJ-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- CYJRNFFLTBEQSQ-UHFFFAOYSA-N 8-(3-methyl-1-benzothiophen-5-yl)-N-(4-methylsulfonylpyridin-3-yl)quinoxalin-6-amine Chemical compound CS(=O)(=O)C1=C(C=NC=C1)NC=1C=C2N=CC=NC2=C(C=1)C=1C=CC2=C(C(=CS2)C)C=1 CYJRNFFLTBEQSQ-UHFFFAOYSA-N 0.000 description 1
- ZRPZPNYZFSJUPA-UHFFFAOYSA-N ARS-1620 Chemical compound Oc1cccc(F)c1-c1c(Cl)cc2c(ncnc2c1F)N1CCN(CC1)C(=O)C=C ZRPZPNYZFSJUPA-UHFFFAOYSA-N 0.000 description 1
- 244000215068 Acacia senegal Species 0.000 description 1
- RZVAJINKPMORJF-UHFFFAOYSA-N Acetaminophen Chemical compound CC(=O)NC1=CC=C(O)C=C1 RZVAJINKPMORJF-UHFFFAOYSA-N 0.000 description 1
- WKEMJKQOLOHJLZ-UHFFFAOYSA-N Almogran Chemical compound C1=C2C(CCN(C)C)=CNC2=CC=C1CS(=O)(=O)N1CCCC1 WKEMJKQOLOHJLZ-UHFFFAOYSA-N 0.000 description 1
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 1
- 229920000945 Amylopectin Polymers 0.000 description 1
- BSYNRYMUTXBXSQ-UHFFFAOYSA-N Aspirin Chemical compound CC(=O)OC1=CC=CC=C1C(O)=O BSYNRYMUTXBXSQ-UHFFFAOYSA-N 0.000 description 1
- KORNTPPJEAJQIU-KJXAQDMKSA-N Cabaser Chemical compound C1=CC([C@H]2C[C@H](CN(CC=C)[C@@H]2C2)C(=O)N(CCCN(C)C)C(=O)NCC)=C3C2=CNC3=C1 KORNTPPJEAJQIU-KJXAQDMKSA-N 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- OKTJSMMVPCPJKN-OUBTZVSYSA-N Carbon-13 Chemical compound [13C] OKTJSMMVPCPJKN-OUBTZVSYSA-N 0.000 description 1
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 1
- 208000014882 Carotid artery disease Diseases 0.000 description 1
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 1
- 101000862089 Clarkia lewisii Glucose-6-phosphate isomerase, cytosolic 1A Proteins 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- 229920002261 Corn starch Polymers 0.000 description 1
- 102000004127 Cytokines Human genes 0.000 description 1
- 108090000695 Cytokines Proteins 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- 206010061818 Disease progression Diseases 0.000 description 1
- 239000004150 EU approved colour Substances 0.000 description 1
- GISRWBROCYNDME-PELMWDNLSA-N F[C@H]1[C@H]([C@H](NC1=O)COC1=NC=CC2=CC(=C(C=C12)OC)C(=O)N)C Chemical compound F[C@H]1[C@H]([C@H](NC1=O)COC1=NC=CC2=CC(=C(C=C12)OC)C(=O)N)C GISRWBROCYNDME-PELMWDNLSA-N 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- YCKRFDGAMUMZLT-IGMARMGPSA-N Fluorine-19 Chemical compound [19F] YCKRFDGAMUMZLT-IGMARMGPSA-N 0.000 description 1
- 108010024636 Glutathione Proteins 0.000 description 1
- 229920000084 Gum arabic Polymers 0.000 description 1
- 102100029100 Hematopoietic prostaglandin D synthase Human genes 0.000 description 1
- 101000988802 Homo sapiens Hematopoietic prostaglandin D synthase Proteins 0.000 description 1
- 206010062016 Immunosuppression Diseases 0.000 description 1
- 206010061216 Infarction Diseases 0.000 description 1
- 206010065390 Inflammatory pain Diseases 0.000 description 1
- 102000003777 Interleukin-1 beta Human genes 0.000 description 1
- 108090000193 Interleukin-1 beta Proteins 0.000 description 1
- 229910021578 Iron(III) chloride Inorganic materials 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- 229910010084 LiAlH4 Inorganic materials 0.000 description 1
- NNJVILVZKWQKPM-UHFFFAOYSA-N Lidocaine Chemical compound CCN(CC)CC(=O)NC1=C(C)C=CC=C1C NNJVILVZKWQKPM-UHFFFAOYSA-N 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- 229930195725 Mannitol Natural products 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- 208000019695 Migraine disease Diseases 0.000 description 1
- 208000010428 Muscle Weakness Diseases 0.000 description 1
- 208000029578 Muscle disease Diseases 0.000 description 1
- 206010049565 Muscle fatigue Diseases 0.000 description 1
- 208000021642 Muscular disease Diseases 0.000 description 1
- 206010028372 Muscular weakness Diseases 0.000 description 1
- AYCPARAPKDAOEN-LJQANCHMSA-N N-[(1S)-2-(dimethylamino)-1-phenylethyl]-6,6-dimethyl-3-[(2-methyl-4-thieno[3,2-d]pyrimidinyl)amino]-1,4-dihydropyrrolo[3,4-c]pyrazole-5-carboxamide Chemical compound C1([C@H](NC(=O)N2C(C=3NN=C(NC=4C=5SC=CC=5N=C(C)N=4)C=3C2)(C)C)CN(C)C)=CC=CC=C1 AYCPARAPKDAOEN-LJQANCHMSA-N 0.000 description 1
- AFBPFSWMIHJQDM-UHFFFAOYSA-N N-methyl-N-phenylamine Natural products CNC1=CC=CC=C1 AFBPFSWMIHJQDM-UHFFFAOYSA-N 0.000 description 1
- 206010061309 Neoplasm progression Diseases 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- JCXJVPUVTGWSNB-UHFFFAOYSA-N Nitrogen dioxide Chemical compound O=[N]=O JCXJVPUVTGWSNB-UHFFFAOYSA-N 0.000 description 1
- IDRGFNPZDVBSSE-UHFFFAOYSA-N OCCN1CCN(CC1)c1ccc(Nc2ncc3cccc(-c4cccc(NC(=O)C=C)c4)c3n2)c(F)c1F Chemical compound OCCN1CCN(CC1)c1ccc(Nc2ncc3cccc(-c4cccc(NC(=O)C=C)c4)c3n2)c(F)c1F IDRGFNPZDVBSSE-UHFFFAOYSA-N 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 206010037660 Pyrexia Diseases 0.000 description 1
- BKRGVLQUQGGVSM-KBXCAEBGSA-N Revanil Chemical compound C1=CC(C=2[C@H](N(C)C[C@H](C=2)NC(=O)N(CC)CC)C2)=C3C2=CNC3=C1 BKRGVLQUQGGVSM-KBXCAEBGSA-N 0.000 description 1
- 208000025747 Rheumatic disease Diseases 0.000 description 1
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical compound [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical group [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- 208000025865 Ulcer Diseases 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- LXRZVMYMQHNYJB-UNXOBOICSA-N [(1R,2S,4R)-4-[[5-[4-[(1R)-7-chloro-1,2,3,4-tetrahydroisoquinolin-1-yl]-5-methylthiophene-2-carbonyl]pyrimidin-4-yl]amino]-2-hydroxycyclopentyl]methyl sulfamate Chemical compound CC1=C(C=C(S1)C(=O)C1=C(N[C@H]2C[C@H](O)[C@@H](COS(N)(=O)=O)C2)N=CN=C1)[C@@H]1NCCC2=C1C=C(Cl)C=C2 LXRZVMYMQHNYJB-UNXOBOICSA-N 0.000 description 1
- XFQAUDMUZLSXTG-UHFFFAOYSA-N [2-methyl-4-[(2-sulfamoylphenyl)sulfamoyl]phenyl] 2,2-dimethylpropanoate Chemical compound C1=C(OC(=O)C(C)(C)C)C(C)=CC(S(=O)(=O)NC=2C(=CC=CC=2)S(N)(=O)=O)=C1 XFQAUDMUZLSXTG-UHFFFAOYSA-N 0.000 description 1
- 230000003187 abdominal effect Effects 0.000 description 1
- 238000002835 absorbance Methods 0.000 description 1
- 239000000205 acacia gum Substances 0.000 description 1
- 235000010489 acacia gum Nutrition 0.000 description 1
- RBYGDVHOECIAFC-UHFFFAOYSA-L acetonitrile;palladium(2+);dichloride Chemical compound [Cl-].[Cl-].[Pd+2].CC#N.CC#N RBYGDVHOECIAFC-UHFFFAOYSA-L 0.000 description 1
- 229960001138 acetylsalicylic acid Drugs 0.000 description 1
- 239000003929 acidic solution Substances 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 125000000641 acridinyl group Chemical group C1(=CC=CC2=NC3=CC=CC=C3C=C12)* 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 150000001340 alkali metals Chemical class 0.000 description 1
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 1
- 125000004450 alkenylene group Chemical group 0.000 description 1
- 150000001345 alkine derivatives Chemical class 0.000 description 1
- 230000029936 alkylation Effects 0.000 description 1
- 238000005804 alkylation reaction Methods 0.000 description 1
- 125000004419 alkynylene group Chemical group 0.000 description 1
- IYABWNGZIDDRAK-UHFFFAOYSA-N allene Chemical group C=C=C IYABWNGZIDDRAK-UHFFFAOYSA-N 0.000 description 1
- BHELZAPQIKSEDF-UHFFFAOYSA-N allyl bromide Chemical compound BrCC=C BHELZAPQIKSEDF-UHFFFAOYSA-N 0.000 description 1
- 229960002133 almotriptan Drugs 0.000 description 1
- 229960003805 amantadine Drugs 0.000 description 1
- DKNWSYNQZKUICI-UHFFFAOYSA-N amantadine Chemical compound C1C(C2)CC3CC2CC1(N)C3 DKNWSYNQZKUICI-UHFFFAOYSA-N 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- 230000033115 angiogenesis Effects 0.000 description 1
- 125000002178 anthracenyl group Chemical group C1(=CC=CC2=CC3=CC=CC=C3C=C12)* 0.000 description 1
- 230000003110 anti-inflammatory effect Effects 0.000 description 1
- 238000011861 anti-inflammatory therapy Methods 0.000 description 1
- 230000006907 apoptotic process Effects 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 150000003974 aralkylamines Chemical class 0.000 description 1
- 150000001543 aryl boronic acids Chemical class 0.000 description 1
- 150000001499 aryl bromides Chemical class 0.000 description 1
- 150000001502 aryl halides Chemical class 0.000 description 1
- 230000036523 atherogenesis Effects 0.000 description 1
- 230000036778 atheroma formation Effects 0.000 description 1
- 125000002393 azetidinyl group Chemical group 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid group Chemical group C(C1=CC=CC=C1)(=O)O WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 1
- UENWRTRMUIOCKN-UHFFFAOYSA-N benzyl thiol Chemical compound SCC1=CC=CC=C1 UENWRTRMUIOCKN-UHFFFAOYSA-N 0.000 description 1
- 125000002619 bicyclic group Chemical group 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 230000004071 biological effect Effects 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- 238000007664 blowing Methods 0.000 description 1
- 238000009835 boiling Methods 0.000 description 1
- 150000001639 boron compounds Chemical class 0.000 description 1
- ZADPBFCGQRWHPN-UHFFFAOYSA-N boronic acid Chemical compound OBO ZADPBFCGQRWHPN-UHFFFAOYSA-N 0.000 description 1
- OZVBMTJYIDMWIL-AYFBDAFISA-N bromocriptine Chemical compound C1=CC(C=2[C@H](N(C)C[C@@H](C=2)C(=O)N[C@]2(C(=O)N3[C@H](C(N4CCC[C@H]4[C@]3(O)O2)=O)CC(C)C)C(C)C)C2)=C3C2=C(Br)NC3=C1 OZVBMTJYIDMWIL-AYFBDAFISA-N 0.000 description 1
- 229960002802 bromocriptine Drugs 0.000 description 1
- UZVHFVZFNXBMQJ-UHFFFAOYSA-N butalbital Chemical compound CC(C)CC1(CC=C)C(=O)NC(=O)NC1=O UZVHFVZFNXBMQJ-UHFFFAOYSA-N 0.000 description 1
- 229960002546 butalbital Drugs 0.000 description 1
- 229960004596 cabergoline Drugs 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- CJZGTCYPCWQAJB-UHFFFAOYSA-L calcium stearate Chemical compound [Ca+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O CJZGTCYPCWQAJB-UHFFFAOYSA-L 0.000 description 1
- 235000013539 calcium stearate Nutrition 0.000 description 1
- 239000008116 calcium stearate Substances 0.000 description 1
- 125000000609 carbazolyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3NC12)* 0.000 description 1
- 125000002837 carbocyclic group Chemical group 0.000 description 1
- 238000001460 carbon-13 nuclear magnetic resonance spectrum Methods 0.000 description 1
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 1
- PFKFTWBEEFSNDU-UHFFFAOYSA-N carbonyldiimidazole Chemical compound C1=CN=CN1C(=O)N1C=CN=C1 PFKFTWBEEFSNDU-UHFFFAOYSA-N 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 1
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 1
- 208000037876 carotid Atherosclerosis Diseases 0.000 description 1
- 230000003197 catalytic effect Effects 0.000 description 1
- 229960000590 celecoxib Drugs 0.000 description 1
- RZEKVGVHFLEQIL-UHFFFAOYSA-N celecoxib Chemical compound C1=CC(C)=CC=C1C1=CC(C(F)(F)F)=NN1C1=CC=C(S(N)(=O)=O)C=C1 RZEKVGVHFLEQIL-UHFFFAOYSA-N 0.000 description 1
- 230000004663 cell proliferation Effects 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 238000005119 centrifugation Methods 0.000 description 1
- DGLFSNZWRYADFC-UHFFFAOYSA-N chembl2334586 Chemical compound C1CCC2=CN=C(N)N=C2C2=C1NC1=CC=C(C#CC(C)(O)C)C=C12 DGLFSNZWRYADFC-UHFFFAOYSA-N 0.000 description 1
- 238000000451 chemical ionisation Methods 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- XTHPWXDJESJLNJ-UHFFFAOYSA-N chlorosulfonic acid Substances OS(Cl)(=O)=O XTHPWXDJESJLNJ-UHFFFAOYSA-N 0.000 description 1
- 125000003016 chromanyl group Chemical group O1C(CCC2=CC=CC=C12)* 0.000 description 1
- 230000001684 chronic effect Effects 0.000 description 1
- 208000037976 chronic inflammation Diseases 0.000 description 1
- 208000037893 chronic inflammatory disorder Diseases 0.000 description 1
- 238000002648 combination therapy Methods 0.000 description 1
- 238000002967 competitive immunoassay Methods 0.000 description 1
- 239000002299 complementary DNA Substances 0.000 description 1
- 210000002808 connective tissue Anatomy 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- DOBRDRYODQBAMW-UHFFFAOYSA-N copper(i) cyanide Chemical compound [Cu+].N#[C-] DOBRDRYODQBAMW-UHFFFAOYSA-N 0.000 description 1
- 239000008120 corn starch Substances 0.000 description 1
- 229940111134 coxibs Drugs 0.000 description 1
- 239000006071 cream Substances 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 239000003255 cyclooxygenase 2 inhibitor Substances 0.000 description 1
- UZBHNSVUMGIKLU-UHFFFAOYSA-N cyclopenten-1-ylboronic acid Chemical compound OB(O)C1=CCCC1 UZBHNSVUMGIKLU-UHFFFAOYSA-N 0.000 description 1
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- DEZRYPDIMOWBDS-UHFFFAOYSA-N dcm dichloromethane Chemical compound ClCCl.ClCCl DEZRYPDIMOWBDS-UHFFFAOYSA-N 0.000 description 1
- 238000012217 deletion Methods 0.000 description 1
- 230000037430 deletion Effects 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- 125000004988 dibenzothienyl group Chemical group C1(=CC=CC=2SC3=C(C21)C=CC=C3)* 0.000 description 1
- ATKXDQOHNICLQW-UHFFFAOYSA-N dichloralphenazone Chemical compound OC(O)C(Cl)(Cl)Cl.OC(O)C(Cl)(Cl)Cl.CN1C(C)=CC(=O)N1C1=CC=CC=C1 ATKXDQOHNICLQW-UHFFFAOYSA-N 0.000 description 1
- 229960005422 dichloralphenazone Drugs 0.000 description 1
- SAUMVKNLVQDHMJ-UHFFFAOYSA-N dichlorine trioxide Inorganic materials ClOCl(=O)=O SAUMVKNLVQDHMJ-UHFFFAOYSA-N 0.000 description 1
- DCOPUUMXTXDBNB-UHFFFAOYSA-N diclofenac Chemical compound OC(=O)CC1=CC=CC=C1NC1=C(Cl)C=CC=C1Cl DCOPUUMXTXDBNB-UHFFFAOYSA-N 0.000 description 1
- 229960001259 diclofenac Drugs 0.000 description 1
- LCSNDSFWVKMJCT-UHFFFAOYSA-N dicyclohexyl-(2-phenylphenyl)phosphane Chemical group C1CCCCC1P(C=1C(=CC=CC=1)C=1C=CC=CC=1)C1CCCCC1 LCSNDSFWVKMJCT-UHFFFAOYSA-N 0.000 description 1
- 235000013681 dietary sucrose Nutrition 0.000 description 1
- 230000010339 dilation Effects 0.000 description 1
- 238000007865 diluting Methods 0.000 description 1
- 238000010790 dilution Methods 0.000 description 1
- 239000012895 dilution Substances 0.000 description 1
- UXGNZZKBCMGWAZ-UHFFFAOYSA-N dimethylformamide dmf Chemical compound CN(C)C=O.CN(C)C=O UXGNZZKBCMGWAZ-UHFFFAOYSA-N 0.000 description 1
- 229910001873 dinitrogen Inorganic materials 0.000 description 1
- XPPKVPWEQAFLFU-UHFFFAOYSA-N diphosphoric acid Chemical compound OP(O)(=O)OP(O)(O)=O XPPKVPWEQAFLFU-UHFFFAOYSA-N 0.000 description 1
- 230000005750 disease progression Effects 0.000 description 1
- 208000035475 disorder Diseases 0.000 description 1
- 125000005411 dithiolanyl group Chemical group S1SC(CC1)* 0.000 description 1
- CETRZFQIITUQQL-UHFFFAOYSA-N dmso dimethylsulfoxide Chemical compound CS(C)=O.CS(C)=O CETRZFQIITUQQL-UHFFFAOYSA-N 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 229960002472 eletriptan Drugs 0.000 description 1
- OTLDLQZJRFYOJR-LJQANCHMSA-N eletriptan Chemical compound CN1CCC[C@@H]1CC1=CN=C2[C]1C=C(CCS(=O)(=O)C=1C=CC=CC=1)C=C2 OTLDLQZJRFYOJR-LJQANCHMSA-N 0.000 description 1
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 description 1
- DMJDTKJDDQHHKM-ZHACJKMWSA-N ethyl (e)-2-(4-tert-butylphenyl)ethenesulfonate Chemical compound CCOS(=O)(=O)\C=C\C1=CC=C(C(C)(C)C)C=C1 DMJDTKJDDQHHKM-ZHACJKMWSA-N 0.000 description 1
- PPCXFTKZPBHXIW-UHFFFAOYSA-N ethyl ethanesulfonate Chemical compound CCOS(=O)(=O)CC PPCXFTKZPBHXIW-UHFFFAOYSA-N 0.000 description 1
- 125000004705 ethylthio group Chemical group C(C)S* 0.000 description 1
- SSDZYLQUYMOSAK-UHFFFAOYSA-N ethynylcyclohexane Chemical group C#CC1CCCCC1 SSDZYLQUYMOSAK-UHFFFAOYSA-N 0.000 description 1
- TXVJSWLZYQMWPC-UHFFFAOYSA-N ethynylcyclopentane Chemical compound C#CC1CCCC1 TXVJSWLZYQMWPC-UHFFFAOYSA-N 0.000 description 1
- 229960004945 etoricoxib Drugs 0.000 description 1
- MNJVRJDLRVPLFE-UHFFFAOYSA-N etoricoxib Chemical compound C1=NC(C)=CC=C1C1=NC=C(Cl)C=C1C1=CC=C(S(C)(=O)=O)C=C1 MNJVRJDLRVPLFE-UHFFFAOYSA-N 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 229960002413 ferric citrate Drugs 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 238000004401 flow injection analysis Methods 0.000 description 1
- 125000003983 fluorenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3CC12)* 0.000 description 1
- 238000000806 fluorine-19 nuclear magnetic resonance spectrum Methods 0.000 description 1
- 235000013355 food flavoring agent Nutrition 0.000 description 1
- 229960002284 frovatriptan Drugs 0.000 description 1
- SIBNYOSJIXCDRI-SECBINFHSA-N frovatriptan Chemical compound C1=C(C(N)=O)[CH]C2=C(C[C@H](NC)CC3)C3=NC2=C1 SIBNYOSJIXCDRI-SECBINFHSA-N 0.000 description 1
- 229960002870 gabapentin Drugs 0.000 description 1
- 230000002496 gastric effect Effects 0.000 description 1
- 231100000414 gastrointestinal toxicity Toxicity 0.000 description 1
- 210000001035 gastrointestinal tract Anatomy 0.000 description 1
- FVIZARNDLVOMSU-UHFFFAOYSA-N ginsenoside K Natural products C1CC(C2(CCC3C(C)(C)C(O)CCC3(C)C2CC2O)C)(C)C2C1C(C)(CCC=C(C)C)OC1OC(CO)C(O)C(O)C1O FVIZARNDLVOMSU-UHFFFAOYSA-N 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 239000008241 heterogeneous mixture Substances 0.000 description 1
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 150000002430 hydrocarbons Chemical group 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- 150000004679 hydroxides Chemical class 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 125000002632 imidazolidinyl group Chemical group 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- 230000001506 immunosuppresive effect Effects 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 238000011065 in-situ storage Methods 0.000 description 1
- 125000003387 indolinyl group Chemical group N1(CCC2=CC=CC=C12)* 0.000 description 1
- 230000001939 inductive effect Effects 0.000 description 1
- 230000007574 infarction Effects 0.000 description 1
- 230000002757 inflammatory effect Effects 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 230000000968 intestinal effect Effects 0.000 description 1
- PNDPGZBMCMUPRI-UHFFFAOYSA-N iodine Chemical compound II PNDPGZBMCMUPRI-UHFFFAOYSA-N 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- RBTARNINKXHZNM-UHFFFAOYSA-K iron trichloride Chemical compound Cl[Fe](Cl)Cl RBTARNINKXHZNM-UHFFFAOYSA-K 0.000 description 1
- 125000000904 isoindolyl group Chemical group C=1(NC=C2C=CC=CC12)* 0.000 description 1
- 125000005956 isoquinolyl group Chemical group 0.000 description 1
- 125000001786 isothiazolyl group Chemical group 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 229940060977 lidoderm Drugs 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 229960003587 lisuride Drugs 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- 229910003002 lithium salt Inorganic materials 0.000 description 1
- 159000000002 lithium salts Chemical class 0.000 description 1
- 229960002373 loxoprofen Drugs 0.000 description 1
- BAZQYVYVKYOAGO-UHFFFAOYSA-M loxoprofen sodium hydrate Chemical compound O.O.[Na+].C1=CC(C(C([O-])=O)C)=CC=C1CC1C(=O)CCC1 BAZQYVYVKYOAGO-UHFFFAOYSA-M 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 229960000994 lumiracoxib Drugs 0.000 description 1
- KHPKQFYUPIUARC-UHFFFAOYSA-N lumiracoxib Chemical compound OC(=O)CC1=CC(C)=CC=C1NC1=C(F)C=CC=C1Cl KHPKQFYUPIUARC-UHFFFAOYSA-N 0.000 description 1
- 210000000191 macrophage derived foam cell Anatomy 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 239000000594 mannitol Substances 0.000 description 1
- 235000010355 mannitol Nutrition 0.000 description 1
- QSHDDOUJBYECFT-UHFFFAOYSA-N mercury Chemical compound [Hg] QSHDDOUJBYECFT-UHFFFAOYSA-N 0.000 description 1
- 229910052753 mercury Inorganic materials 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 150000004702 methyl esters Chemical class 0.000 description 1
- 206010027599 migraine Diseases 0.000 description 1
- 239000002480 mineral oil Substances 0.000 description 1
- 235000010446 mineral oil Nutrition 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- PEECTLLHENGOKU-UHFFFAOYSA-N n,n-dimethylpyridin-4-amine Chemical compound CN(C)C1=CC=NC=C1.CN(C)C1=CC=NC=C1 PEECTLLHENGOKU-UHFFFAOYSA-N 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 229960005254 naratriptan Drugs 0.000 description 1
- AMKVXSZCKVJAGH-UHFFFAOYSA-N naratriptan Chemical compound C12=CC(CCS(=O)(=O)NC)=CC=C2NC=C1C1CCN(C)CC1 AMKVXSZCKVJAGH-UHFFFAOYSA-N 0.000 description 1
- 231100000417 nephrotoxicity Toxicity 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 230000003472 neutralizing effect Effects 0.000 description 1
- 229910052759 nickel Inorganic materials 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- 125000004433 nitrogen atom Chemical group N* 0.000 description 1
- 229910000069 nitrogen hydride Inorganic materials 0.000 description 1
- 238000000655 nuclear magnetic resonance spectrum Methods 0.000 description 1
- 229940006093 opthalmologic coloring agent diagnostic Drugs 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 150000007530 organic bases Chemical class 0.000 description 1
- 125000001715 oxadiazolyl group Chemical group 0.000 description 1
- 125000000160 oxazolidinyl group Chemical group 0.000 description 1
- 125000004430 oxygen atom Chemical group O* 0.000 description 1
- 229940094443 oxytocics prostaglandins Drugs 0.000 description 1
- QJPQVXSHYBGQGM-UHFFFAOYSA-N palladium;triphenylphosphane Chemical compound [Pd].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 QJPQVXSHYBGQGM-UHFFFAOYSA-N 0.000 description 1
- 229960005489 paracetamol Drugs 0.000 description 1
- 239000012188 paraffin wax Substances 0.000 description 1
- 238000007911 parenteral administration Methods 0.000 description 1
- 230000037361 pathway Effects 0.000 description 1
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 1
- 229960004851 pergolide Drugs 0.000 description 1
- YEHCICAEULNIGD-MZMPZRCHSA-N pergolide Chemical compound C1=CC([C@H]2C[C@@H](CSC)CN([C@@H]2C2)CCC)=C3C2=CNC3=C1 YEHCICAEULNIGD-MZMPZRCHSA-N 0.000 description 1
- 230000000144 pharmacologic effect Effects 0.000 description 1
- 125000001792 phenanthrenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3C=CC12)* 0.000 description 1
- 125000001484 phenothiazinyl group Chemical group C1(=CC=CC=2SC3=CC=CC=C3NC12)* 0.000 description 1
- 125000001644 phenoxazinyl group Chemical group C1(=CC=CC=2OC3=CC=CC=C3NC12)* 0.000 description 1
- 125000004193 piperazinyl group Chemical group 0.000 description 1
- 125000005936 piperidyl group Chemical group 0.000 description 1
- 239000013612 plasmid Substances 0.000 description 1
- 125000003367 polycyclic group Chemical group 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- LZMJNVRJMFMYQS-UHFFFAOYSA-N poseltinib Chemical compound C1CN(C)CCN1C(C=C1)=CC=C1NC1=NC(OC=2C=C(NC(=O)C=C)C=CC=2)=C(OC=C2)C2=N1 LZMJNVRJMFMYQS-UHFFFAOYSA-N 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910000027 potassium carbonate Inorganic materials 0.000 description 1
- 229920001592 potato starch Polymers 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 229960003089 pramipexole Drugs 0.000 description 1
- FASDKYOPVNHBLU-ZETCQYMHSA-N pramipexole Chemical compound C1[C@@H](NCCC)CCC2=C1SC(N)=N2 FASDKYOPVNHBLU-ZETCQYMHSA-N 0.000 description 1
- AYXYPKUFHZROOJ-ZETCQYMHSA-N pregabalin Chemical compound CC(C)C[C@H](CN)CC(O)=O AYXYPKUFHZROOJ-ZETCQYMHSA-N 0.000 description 1
- BHMBVRSPMRCCGG-OUTUXVNYSA-N prostaglandin D2 Chemical compound CCCCC[C@H](O)\C=C\[C@@H]1[C@@H](C\C=C/CCCC(O)=O)[C@@H](O)CC1=O BHMBVRSPMRCCGG-OUTUXVNYSA-N 0.000 description 1
- KAQKFAOMNZTLHT-OZUDYXHBSA-N prostaglandin I2 Chemical compound O1\C(=C/CCCC(O)=O)C[C@@H]2[C@@H](/C=C/[C@@H](O)CCCCC)[C@H](O)C[C@@H]21 KAQKFAOMNZTLHT-OZUDYXHBSA-N 0.000 description 1
- 239000002599 prostaglandin synthase inhibitor Substances 0.000 description 1
- BHMBVRSPMRCCGG-UHFFFAOYSA-N prostaglandine D2 Natural products CCCCCC(O)C=CC1C(CC=CCCCC(O)=O)C(O)CC1=O BHMBVRSPMRCCGG-UHFFFAOYSA-N 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 125000003373 pyrazinyl group Chemical group 0.000 description 1
- 125000003072 pyrazolidinyl group Chemical group 0.000 description 1
- 125000002755 pyrazolinyl group Chemical group 0.000 description 1
- 125000002098 pyridazinyl group Chemical group 0.000 description 1
- SHNUBALDGXWUJI-UHFFFAOYSA-N pyridin-2-ylmethanol Chemical compound OCC1=CC=CC=N1 SHNUBALDGXWUJI-UHFFFAOYSA-N 0.000 description 1
- 125000000719 pyrrolidinyl group Chemical group 0.000 description 1
- 125000002294 quinazolinyl group Chemical group N1=C(N=CC2=CC=CC=C12)* 0.000 description 1
- 125000001567 quinoxalinyl group Chemical group N1=C(C=NC2=CC=CC=C12)* 0.000 description 1
- 102000005962 receptors Human genes 0.000 description 1
- 230000008929 regeneration Effects 0.000 description 1
- 238000011069 regeneration method Methods 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- QEVHRUUCFGRFIF-MDEJGZGSSA-N reserpine Chemical compound O([C@H]1[C@@H]([C@H]([C@H]2C[C@@H]3C4=C(C5=CC=C(OC)C=C5N4)CCN3C[C@H]2C1)C(=O)OC)OC)C(=O)C1=CC(OC)=C(OC)C(OC)=C1 QEVHRUUCFGRFIF-MDEJGZGSSA-N 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 125000006413 ring segment Chemical group 0.000 description 1
- 229960000425 rizatriptan Drugs 0.000 description 1
- TXHZXHICDBAVJW-UHFFFAOYSA-N rizatriptan Chemical compound C=1[C]2C(CCN(C)C)=CN=C2C=CC=1CN1C=NC=N1 TXHZXHICDBAVJW-UHFFFAOYSA-N 0.000 description 1
- 229960000371 rofecoxib Drugs 0.000 description 1
- RZJQGNCSTQAWON-UHFFFAOYSA-N rofecoxib Chemical compound C1=CC(S(=O)(=O)C)=CC=C1C1=C(C=2C=CC=CC=2)C(=O)OC1 RZJQGNCSTQAWON-UHFFFAOYSA-N 0.000 description 1
- 229960001879 ropinirole Drugs 0.000 description 1
- UHSKFQJFRQCDBE-UHFFFAOYSA-N ropinirole Chemical compound CCCN(CCC)CCC1=CC=CC2=C1CC(=O)N2 UHSKFQJFRQCDBE-UHFFFAOYSA-N 0.000 description 1
- CVHZOJJKTDOEJC-UHFFFAOYSA-N saccharin Chemical compound C1=CC=C2C(=O)NS(=O)(=O)C2=C1 CVHZOJJKTDOEJC-UHFFFAOYSA-N 0.000 description 1
- 229930195734 saturated hydrocarbon Natural products 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 238000011894 semi-preparative HPLC Methods 0.000 description 1
- 238000010898 silica gel chromatography Methods 0.000 description 1
- 210000002027 skeletal muscle Anatomy 0.000 description 1
- 210000003491 skin Anatomy 0.000 description 1
- 210000002460 smooth muscle Anatomy 0.000 description 1
- 239000012312 sodium hydride Substances 0.000 description 1
- NESLWCLHZZISNB-UHFFFAOYSA-M sodium phenolate Chemical compound [Na+].[O-]C1=CC=CC=C1 NESLWCLHZZISNB-UHFFFAOYSA-M 0.000 description 1
- 159000000000 sodium salts Chemical class 0.000 description 1
- 235000010265 sodium sulphite Nutrition 0.000 description 1
- AKHNMLFCWUSKQB-UHFFFAOYSA-L sodium thiosulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=S AKHNMLFCWUSKQB-UHFFFAOYSA-L 0.000 description 1
- 235000019345 sodium thiosulphate Nutrition 0.000 description 1
- ZWCDUDOOPJLODP-UHFFFAOYSA-M sodium;2-(3,4-dichlorophenyl)ethanesulfonate Chemical compound [Na+].[O-]S(=O)(=O)CCC1=CC=C(Cl)C(Cl)=C1 ZWCDUDOOPJLODP-UHFFFAOYSA-M 0.000 description 1
- 239000011877 solvent mixture Substances 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 235000010356 sorbitol Nutrition 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 230000003595 spectral effect Effects 0.000 description 1
- 238000001228 spectrum Methods 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 230000000638 stimulation Effects 0.000 description 1
- 239000012258 stirred mixture Substances 0.000 description 1
- 150000003440 styrenes Chemical class 0.000 description 1
- 238000007920 subcutaneous administration Methods 0.000 description 1
- 229960004793 sucrose Drugs 0.000 description 1
- 125000000565 sulfonamide group Chemical group 0.000 description 1
- 150000003456 sulfonamides Chemical class 0.000 description 1
- 229960003708 sumatriptan Drugs 0.000 description 1
- KQKPFRSPSRPDEB-UHFFFAOYSA-N sumatriptan Chemical compound CNS(=O)(=O)CC1=CC=C2NC=C(CCN(C)C)C2=C1 KQKPFRSPSRPDEB-UHFFFAOYSA-N 0.000 description 1
- 239000000829 suppository Substances 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 235000012222 talc Nutrition 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- WHRNULOCNSKMGB-UHFFFAOYSA-N tetrahydrofuran thf Chemical compound C1CCOC1.C1CCOC1 WHRNULOCNSKMGB-UHFFFAOYSA-N 0.000 description 1
- 125000001412 tetrahydropyranyl group Chemical group 0.000 description 1
- 125000005958 tetrahydrothienyl group Chemical group 0.000 description 1
- WROMPOXWARCANT-UHFFFAOYSA-N tfa trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F.OC(=O)C(F)(F)F WROMPOXWARCANT-UHFFFAOYSA-N 0.000 description 1
- 125000001113 thiadiazolyl group Chemical group 0.000 description 1
- 125000001984 thiazolidinyl group Chemical group 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- 125000005309 thioalkoxy group Chemical group 0.000 description 1
- 125000004568 thiomorpholinyl group Chemical group 0.000 description 1
- 210000001519 tissue Anatomy 0.000 description 1
- 230000008354 tissue degradation Effects 0.000 description 1
- 239000004408 titanium dioxide Substances 0.000 description 1
- 235000010215 titanium dioxide Nutrition 0.000 description 1
- 230000001131 transforming effect Effects 0.000 description 1
- 125000001425 triazolyl group Chemical group 0.000 description 1
- LGQXXHMEBUOXRP-UHFFFAOYSA-N tributyl borate Chemical compound CCCCOB(OCCCC)OCCCC LGQXXHMEBUOXRP-UHFFFAOYSA-N 0.000 description 1
- WRECIMRULFAWHA-UHFFFAOYSA-N trimethyl borate Chemical compound COB(OC)OC WRECIMRULFAWHA-UHFFFAOYSA-N 0.000 description 1
- NHDIQVFFNDKAQU-UHFFFAOYSA-N tripropan-2-yl borate Chemical compound CC(C)OB(OC(C)C)OC(C)C NHDIQVFFNDKAQU-UHFFFAOYSA-N 0.000 description 1
- 230000036269 ulceration Effects 0.000 description 1
- 229930195735 unsaturated hydrocarbon Natural products 0.000 description 1
- 229960002004 valdecoxib Drugs 0.000 description 1
- LNPDTQAFDNKSHK-UHFFFAOYSA-N valdecoxib Chemical compound CC=1ON=C(C=2C=CC=CC=2)C=1C1=CC=C(S(N)(=O)=O)C=C1 LNPDTQAFDNKSHK-UHFFFAOYSA-N 0.000 description 1
- 210000004509 vascular smooth muscle cell Anatomy 0.000 description 1
- 210000005166 vasculature Anatomy 0.000 description 1
- 235000015112 vegetable and seed oil Nutrition 0.000 description 1
- 239000008158 vegetable oil Substances 0.000 description 1
- 239000001993 wax Substances 0.000 description 1
- 125000001834 xanthenyl group Chemical group C1=CC=CC=2OC3=CC=CC=C3C(C12)* 0.000 description 1
- UGOMMVLRQDMAQQ-UHFFFAOYSA-N xphos Chemical compound CC(C)C1=CC(C(C)C)=CC(C(C)C)=C1C1=CC=CC=C1P(C1CCCCC1)C1CCCCC1 UGOMMVLRQDMAQQ-UHFFFAOYSA-N 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 229960001360 zolmitriptan Drugs 0.000 description 1
- ULSDMUVEXKOYBU-ZDUSSCGKSA-N zolmitriptan Chemical compound C1=C2C(CCN(C)C)=CNC2=CC=C1C[C@H]1COC(=O)N1 ULSDMUVEXKOYBU-ZDUSSCGKSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D307/00—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom
- C07D307/77—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom ortho- or peri-condensed with carbocyclic rings or ring systems
- C07D307/78—Benzo [b] furans; Hydrogenated benzo [b] furans
- C07D307/79—Benzo [b] furans; Hydrogenated benzo [b] furans with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to carbon atoms of the hetero ring
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P21/00—Drugs for disorders of the muscular or neuromuscular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/04—Centrally acting analgesics, e.g. opioids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C311/00—Amides of sulfonic acids, i.e. compounds having singly-bound oxygen atoms of sulfo groups replaced by nitrogen atoms, not being part of nitro or nitroso groups
- C07C311/30—Sulfonamides, the carbon skeleton of the acid part being further substituted by singly-bound nitrogen atoms, not being part of nitro or nitroso groups
- C07C311/37—Sulfonamides, the carbon skeleton of the acid part being further substituted by singly-bound nitrogen atoms, not being part of nitro or nitroso groups having the sulfur atom of at least one of the sulfonamide groups bound to a carbon atom of a six-membered aromatic ring
- C07C311/38—Sulfonamides, the carbon skeleton of the acid part being further substituted by singly-bound nitrogen atoms, not being part of nitro or nitroso groups having the sulfur atom of at least one of the sulfonamide groups bound to a carbon atom of a six-membered aromatic ring having sulfur atoms of sulfonamide groups and amino groups bound to carbon atoms of six-membered rings of the same carbon skeleton
- C07C311/39—Sulfonamides, the carbon skeleton of the acid part being further substituted by singly-bound nitrogen atoms, not being part of nitro or nitroso groups having the sulfur atom of at least one of the sulfonamide groups bound to a carbon atom of a six-membered aromatic ring having sulfur atoms of sulfonamide groups and amino groups bound to carbon atoms of six-membered rings of the same carbon skeleton having the nitrogen atom of at least one of the sulfonamide groups bound to hydrogen atoms or to an acyclic carbon atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C323/00—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups
- C07C323/64—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and sulfur atoms, not being part of thio groups, bound to the same carbon skeleton
- C07C323/67—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and sulfur atoms, not being part of thio groups, bound to the same carbon skeleton containing sulfur atoms of sulfonamide groups, bound to the carbon skeleton
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D209/00—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D209/02—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom condensed with one carbocyclic ring
- C07D209/04—Indoles; Hydrogenated indoles
- C07D209/08—Indoles; Hydrogenated indoles with only hydrogen atoms or radicals containing only hydrogen and carbon atoms, directly attached to carbon atoms of the hetero ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D209/00—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D209/02—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom condensed with one carbocyclic ring
- C07D209/04—Indoles; Hydrogenated indoles
- C07D209/10—Indoles; Hydrogenated indoles with substituted hydrocarbon radicals attached to carbon atoms of the hetero ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/24—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with substituted hydrocarbon radicals attached to ring carbon atoms
- C07D213/28—Radicals substituted by singly-bound oxygen or sulphur atoms
- C07D213/30—Oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/60—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D213/61—Halogen atoms or nitro radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/60—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D213/62—Oxygen or sulfur atoms
- C07D213/63—One oxygen atom
- C07D213/64—One oxygen atom attached in position 2 or 6
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D239/00—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings
- C07D239/02—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings
- C07D239/24—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members
- C07D239/28—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, directly attached to ring carbon atoms
- C07D239/32—One oxygen, sulfur or nitrogen atom
- C07D239/34—One oxygen atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D265/00—Heterocyclic compounds containing six-membered rings having one nitrogen atom and one oxygen atom as the only ring hetero atoms
- C07D265/28—1,4-Oxazines; Hydrogenated 1,4-oxazines
- C07D265/34—1,4-Oxazines; Hydrogenated 1,4-oxazines condensed with carbocyclic rings
- C07D265/36—1,4-Oxazines; Hydrogenated 1,4-oxazines condensed with carbocyclic rings condensed with one six-membered ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D271/00—Heterocyclic compounds containing five-membered rings having two nitrogen atoms and one oxygen atom as the only ring hetero atoms
- C07D271/02—Heterocyclic compounds containing five-membered rings having two nitrogen atoms and one oxygen atom as the only ring hetero atoms not condensed with other rings
- C07D271/10—1,3,4-Oxadiazoles; Hydrogenated 1,3,4-oxadiazoles
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D277/00—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings
- C07D277/02—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings
- C07D277/20—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D277/32—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D307/00—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom
- C07D307/02—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings
- C07D307/34—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D307/38—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with substituted hydrocarbon radicals attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D307/00—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom
- C07D307/77—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom ortho- or peri-condensed with carbocyclic rings or ring systems
- C07D307/91—Dibenzofurans; Hydrogenated dibenzofurans
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D333/00—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom
- C07D333/02—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings
- C07D333/04—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings not substituted on the ring sulphur atom
- C07D333/26—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings not substituted on the ring sulphur atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D333/30—Hetero atoms other than halogen
- C07D333/34—Sulfur atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings
- C07D409/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2601/00—Systems containing only non-condensed rings
- C07C2601/06—Systems containing only non-condensed rings with a five-membered ring
- C07C2601/08—Systems containing only non-condensed rings with a five-membered ring the ring being saturated
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2601/00—Systems containing only non-condensed rings
- C07C2601/06—Systems containing only non-condensed rings with a five-membered ring
- C07C2601/10—Systems containing only non-condensed rings with a five-membered ring the ring being unsaturated
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2602/00—Systems containing two condensed rings
- C07C2602/02—Systems containing two condensed rings the rings having only two atoms in common
- C07C2602/04—One of the condensed rings being a six-membered aromatic ring
- C07C2602/08—One of the condensed rings being a six-membered aromatic ring the other ring being five-membered, e.g. indane
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2602/00—Systems containing two condensed rings
- C07C2602/02—Systems containing two condensed rings the rings having only two atoms in common
- C07C2602/04—One of the condensed rings being a six-membered aromatic ring
- C07C2602/10—One of the condensed rings being a six-membered aromatic ring the other ring being six-membered, e.g. tetraline
Definitions
- the present invention relates to bis-(sulfonylamino) derivatives, processes for their preparation, pharmaceutical compositions containing them and their use in therapy.
- G 3 is phenyl, pyridyl or 2-methyl-1,3,4-oxadiazole
- G 2 represents H, phenyl or 5- or 6-membered heteroaryl; optionally fused to one further ring independently selected from phenyl, a 5- or 6-membered heteroaryl, C 5-6 carbocyclyl or C 5-6 heterocyclyl ring; and any phenyl or heteroaryl moieties in G 1 and G 2 are optionally substituted by one or more substituents independently selected from halogen, C 1-6 alkyl and C 1-6 alkoxy; said C 1-6 alkyl being optionally substituted by OH or by one or more F atoms.
- n an integer 0, 1, 2, 3 or 4;
- neuropathic pain therapies including for example gabapentin, lidoderm, pregablin and equivalents and pharmaceutically active isomer(s) and metabolite(s) thereof.
- R 3 , R 5 , R 6 and R 10 represents hydrogen
- G 3 represents phenyl or 5- or 6-membered heteroaryl
- n-Butyllithium (59.0 mL, 1.6M solution in hexanes) was added dropwise during 25 min to a solution of ethylmethanesulfonate (9.1 mL, 86 mmol) in THF (200 mL) at ⁇ 78° C.
- the reaction mixture was stirred for 15 min then diethyl chlorophosphate (7.2 ml, 50 mmol) was added dropwise.
- the solution was stirred at ⁇ 78° C. for 30 min, allowed to warm to 0° C. and then cooled to ⁇ 78° C. again.
Landscapes
- Organic Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Engineering & Computer Science (AREA)
- Pharmacology & Pharmacy (AREA)
- Veterinary Medicine (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Neurology (AREA)
- Physical Education & Sports Medicine (AREA)
- Neurosurgery (AREA)
- Diabetes (AREA)
- Biomedical Technology (AREA)
- Heart & Thoracic Surgery (AREA)
- Cardiology (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Rheumatology (AREA)
- Pain & Pain Management (AREA)
- Vascular Medicine (AREA)
- Immunology (AREA)
- Urology & Nephrology (AREA)
- Psychiatry (AREA)
- Obesity (AREA)
- Hematology (AREA)
- Pulmonology (AREA)
- Endocrinology (AREA)
- Emergency Medicine (AREA)
- Hospice & Palliative Care (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Thiazole And Isothizaole Compounds (AREA)
- Furan Compounds (AREA)
Abstract
The invention provides compounds of formula (I)
wherein R1, R3, L1, L2, G1, G2, A and m are as defined in the specification and optical isomers, racemates and tautomers thereof, and pharmaceutically acceptable salts thereof; together with processes for their preparation, pharmaceutical compositions containing them and their use in therapy. The compounds are inhibitors of microsomal prostaglandin E synthase-1.
Description
- This application claims benefit under 35 U.S.C. §119(e) of U.S. Provisional Application 61/015,287, filed Dec. 20, 2007, which is incorporated herein by reference in its entirety.
- The present invention relates to bis-(sulfonylamino) derivatives, processes for their preparation, pharmaceutical compositions containing them and their use in therapy.
- Modulation of prostaglandin metabolism is at the center of current anti-inflammatory therapies. NSAIDs and COX-2 inhibitors block the activity of cyclooxygenases and their ability to convert arachidonic acid into prostaglandin H2 (PGH2). PGH2 can be subsequently metabolized by terminal prostaglandin synthases to the corresponding biologically active PGs, namely, PGI2, thromboxane (Tx) A2, PGD2, PGF2α, and PGE2. A combination of pharmacological, genetic and neutralizing antibody approaches demonstrates the importance of PGE2 in inflammation. The conversion of PGH2 to PGE2 by prostaglandin E synthases (PGES) may therefore represent a pivotal step in the propagation of inflammatory stimuli.
- Microsomal prostaglandin E synthase-1 (mPGES-1) is an inducible PGES after exposure to pro-inflammatory stimuli. mPGES-1 is induced in the periphery and in the CNS by inflammation and represents therefore a target for acute and chronic inflammatory disorders.
- PGE2 is a major prostanoid driving inflammatory processes. The Prostanoid is produced from arachidonic acid liberated by Phospholipases (PLAs). Arachidonic acid is tranformed by the action of Prostaglandin H Synthase (PGH Synthase, cycloxygenase) into PGH2 which is a substrate for mPGES-1, that is the terminal enzyme transforming PGH2 to the pro-inflammatory PGE2.
- NSAIDs reduce PGE2 by inhibiting cyclooxygenase, but at the same time reducing other prostanoids, giving side effects such as ulcerations in the GI tract. mPGES-1 inhibition gives a similar effect on PGE2 production without affecting the formation of other prostanoids, and hence a more favourable profile. By blocking the formation of PGE2 in animal models of inflammatory pain a reduced inflammation, pain and fever response has been demonstrated (see e.g., Kojima et al., The Journal of Immunology 2008, 180, 8361-6, Xu et al., The Journal of Pharmacology and Experimental Therapeutics 2008, 326, 754-63).
- In abdominal aortic aneurism, inflammation leads to connective tissue degradation and smooth muscle apoptosis ultimately leading to aortic dilation and rupture. In animals lacking mPGES-1 a slower disease progression and disease severity has been demonstrated (see e.g., Wang et al. Circulation, 2008, 117, 1302-1309).
- Several lines of evidence indicate that PGE2 is involved in malignant growth. PGE2 facilitates tumour progression by stimulation of cellular proliferation and angiogenesis and by modulation of immunosuppression. In support of a role for PGE2 in carcinogenesis genetic deletion of mPGES-1 in mice suppress the intestinal tumorigenesis (Nakanishi et al. Cancer Research 2008, 68(9), 3251-9). In man, mPGES-1 is also upregulated in cancers such as clorectal cancer (see e.g., Schroder Journal of Lipid Research 2006, 47, 1071-80).
- Myositis is chronic muscle disorder characterized by muscle weakness and fatigue. Proinflammatory cytokines and prostanoids have been implicated in the development of myositis. In skeletal muscle tissue from patients suffering from myositis an increase in cyclooxygenases and mPGES-1 has been demonstrated, implicating mPGES-1 as a target for treating this condition (see e.g., Korotkova Annals of the Rheumatic Diseases 2008, 67, 1596-1602).
- In atherosclerosis inflammation of the vasculature leads to atheroma formation that eventually may progress into infarction. In patients with carotid atherosclerosis an increase in mPGES-1 in plaque regions have been found (Gómez-Hernández Atherosclerosis 2006, 187, 139-49). In an animal model of atherosclerosis, mice lacking the mPGES-1 receptor were found to show a retarded atherogenesis and a concomitant reduction in macrophage—derived foam cells together with an increase in vascular smooth muscle cells (see e.g., Wang Proceedings of National Academy of Sciences 2006, 103(39), 14507-12).
- The present invention is directed to novel compounds that are selective inhibitors of the microsomal prostaglandin E synthase-1 enzyme and would therefore be useful for the treatment of pain and inflammation in a variety of diseases or conditions.
- In one aspect we disclose a compound of formula (I) or a pharmaceutically acceptable salt thereof
- wherein:
- A is selected from phenyl or a 5- or 6-membered heteroaryl or a 5- or 6-membered heterocyclenyl moiety; said phenyl or 5- or 6-membered heteroaryl or 5- or 6-membered heterocyclenyl moiety in group A being optionally fused to a phenyl, a 5- or 6-membered heteroaryl, C5-6carbocyclyl or C5-6heterocyclyl ring;
- each R1 is independently selected from halogen, nitro, SF5, CN, NR5R6, OH, CHO, CO2C1-4alkyl, CONR5R6, C1-4alkyl, C2-4alkenyl, C2-4alkynyl, C1-4alkoxy, G3, OG3 or OCH2G3; said C1-4alkyl, C2-4alkenyl, C2-4alkynyl or C1-4alkoxy being optionally substituted by one or more substituents selected from OH, NR5R6, C1-4alkyl or F;
- m represents an integer 0, 1, 2, 3 or 4;
- each R3 is independently selected from hydrogen, CN and C1-4alkyl; said C1-4alkyl being optionally substituted with OH, CN, C1-4alkoxy, NR7R8, or one or more F atoms;
- L1 represents a direct bond, C1-4alkylene, C2-4alkenylene or C2-4alkynylene;
- L2 represents a direct bond, —O—, —OCH2—, C1-2alkylene, —C≡C— or —NHCONH—;
- G1 represents phenyl, 5- or 6-membered heteroaryl, C3-10carbocyclyl or C5-8heterocyclyl;
- G2 represents H, C1-6alkyl, phenyl, 5- or 6-membered heteroaryl, C3-10carbocyclyl or C5-8heterocyclyl; said C1-6alkyl being optionally further substituted by one or more groups selected from OH, C1-6alkoxy and halogen;
- the phenyl, heteroaryl, carbocyclyl or heterocyclyl moieties in G1 and G2 being optionally fused to one or two further rings independently selected from phenyl, a 5- or 6-membered heteroaryl, C5-6carbocyclyl or C5-6heterocyclyl ring;
- any phenyl, heteroaryl, carbocyclyl or heterocyclyl moieties in G1 and G2 being optionally substituted by one or more substituents independently selected from halogen, OH, CN, NO2, CO2R9, C1-6alkyl, C1-6alkoxy, C1-4thioalkoxy, SO2NR10R11, NR12R13, —NHCOC(OH)(CH3)CF3 or —CH2OCH2CF2CHF2; said C1-6alkyl or C1-6alkoxy being optionally substituted by OH or by one or more F atoms;
- R9 represents C1-6alkyl;
- G3 represents phenyl or 5- or 6-membered heteroaryl; and
- each R4, R5, R6, R7, R8, R10, R11, R12 and R13 is independently selected from H or C1-4alkyl;
- with the proviso that the following compounds are excluded:
- 3-[(phenylsulfonyl)amino]pyridine-2-sulfonamide;
- 2-amino-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 4-tert-butyl-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 3-amino-4-hydroxy-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 4-bromo-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 4-fluoro-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 4-methyl-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 4-nitro-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-nitro-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 4-amino-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[(phenylsulfonyl)amino]-4-(trifluoromethyl)benzenesulfonamide;
- 2-{[(4-methylphenyl)sulfonyl]amino}-4-(trifluoromethyl)benzenesulfonamide;
- N-(2-sulfamoylphenyl)quinoline-8-sulfonamide;
- 4-tert-butyl-N-(2-(N-ethylsulfamoyl)phenyl)-2,6-dimethylbenzenesulfonamide;
- 2,5-dibromo-N-(2-( -ethylsulfamoyl)phenyl)benzenesulfonamide;
- methyl 4-(5-fluoro-3-methylbenzo[b]thiophene-2-sulfonamido)-3-sulfamoylbenzoate;
- 2,3,4,5,6-pentafluoro-N-(2-sulfamoylphenyl)benzenesulfonamide;
- N-(2-(N,N-dimethylsulfamoyl)phenyl)-5,7-dimethyl-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide;
- N-(2-(N-tert-butylsulfamoyl)phenyl)-5,7-dimethyl-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide;
- N-(2-(N-methoxy-N-methylsulfamoyl)phenyl)-5,7-dimethyl-[1,2,4]triazolo[1,5-a]pyrimidine-
- N-(2-(N-methoxy-N-methylsulfamoyl)phenyl)-5,7-dimethyl-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide;
- 6-methyl-N-(2-sulfamoylphenyl)thiazolo[3,2-b][1,2,4]triazole-2-sulfonamide;
- 6-chloro-N-(2-(N,N-dimethylsulfamoyl)phenyl)-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide;
- 6-chloro-N-(2-(N-ethyl-N-methylsulfamoyl)phenyl)-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide;
- N-(2-(N,N-dimethylsulfamoyl)phenyl)-5-methyl-7,8-dihydro-6H-cyclopenta[e][1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide;
- N-(2-(N,N-dimethylsulfamoyl)-5-(trifluoromethyl)phenyl)-5,7-dimethyl-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide;
- N-(2-(N-ethyl-N-methylsulfamoyl)phenyl)-5,7-dimethyl-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide; and
- N-(2-(N,N-dimethylsulfamoyl)phenyl)-5,7-dimethyl-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide.
- As used herein, a C1-C6 alkyl moiety is a linear or branched alkyl moiety containing from 1 to 6 carbon atoms, such as a C1-C4 or C1-C2 alkyl moiety. Examples of C1-C6 alkyl moieties include methyl, ethyl, n-propyl, i-propyl, n-butyl, s-butyl and t-butyl, pentyl and hexyl. For the avoidance of doubt, where two alkyl moieties are present in a substituent, the alkyl moieties may be the same or different.
- As used herein, a C1-C4 alkylene or C1-C2 alkylene group is any divalent linear or branched C1-C4 or C1-C2 alkyl moiety. Linear C1-C4 alkylene groups are methylene, ethylene, n-propylene and n-butylene groups. Branched C1-C4 alkylene groups include —CH(CH3)—, —CH(CH3)—CH2— and —CH2—CH(CH3)—.
- As used herein, a C2-C4 alkenyl moiety is a linear or branched alkyl moiety containing from 2 to 4 carbon atoms that includes a carbon-carbon double bond. As used herein, a C2-C4 alkenylene group is any divalent linear or branched C2-C4 alkylene moiety that includes a carbon-carbon double bond.
- As used herein, a C2-C4 alkynyl moiety is a linear or branched alkyl moiety containing from 2 to 4 carbon atoms that includes a carbon-carbon triple bond. As used herein, a C2-C4 alkynylene group is any divalent linear or branched C2-C4 alkylene moiety that includes a carbon-carbon triple bond.
- As used herein, a halogen is chlorine, fluorine, bromine or iodine. A halogen is typically fluorine, chlorine or bromine.
- As used herein, a C1-C6 alkoxy moiety is a said C1-C6 alkyl moiety attached to an oxygen atom. Examples include methoxy and ethoxy.
- As used herein, a C1-C4 thioalkoxy moiety is a said C1-C4 alkyl moiety attached to a sulphur atom. Examples include methylthio and ethylthio.
- As used herein, a 5- or 6-membered heteroaryl moiety is a monocyclic 5- or 6-membered aromatic ring, containing at least one heteroatom, for example 1, 2 or 3 heteroatoms, selected from O, S and N. Examples include imidazolyl, isoxazolyl, pyrrolyl, thienyl, thiazolyl, furanyl, pyridyl, pyrazinyl, pyrimidinyl, pyridazinyl, oxadiazolyl, oxazolyl, isoxazolyl, thiazolyl, isothiazolyl, thiadiazolyl, pyrazolyl and triazolyl moieties.
- In one embodiment, a 5- or 6-membered heteroaryl moiety is pyrrolyl, thienyl, furanyl, pyridyl, pyrimidinyl, oxazolyl, thiazolyl or pyrazolyl moiety.
- As used herein, a 5- to 8-membered heterocyclyl moiety is a monocyclic non-aromatic, saturated or unsaturated C5-C8 carbocyclic ring, in which at least one, for example, 1, 2 or 3, carbon atoms in the ring are replaced with a moiety selected independently from O, S, SO, SO2 and N and optionally incorporating one or more carbonyl (C═O) groups. When such a ring contains a carbon-carbon double bond, it may alternatively be referred to as a heterocyclenyl moiety. Typically, a heterocyclyl moiety is a saturated C5-C8 ring such as a C5-C6 ring in which 1, 2 or 3 of the carbon atoms in the ring are replaced with a moiety selected from O, S, SO2 and NH and optionally incorporating one or two CO moieties. Examples include azetidinyl, pyrazolidinyl, piperidyl, piperidin-2,6-dionyl, piperidin-2-onyl, perhydroazepinyl (hexamethylene iminyl), piperazinyl, morpholinyl, thiomorpholinyl, S-oxothiomorpholinyl, S,S-dioxothiomorpholinyl, 1,3-dioxolanyl, 1,4-dioxanyl, pyrrolidinyl, imidazolidinyl, imidazol-2-onyl, pyrrolidin-2-onyl, tetrahydrofuranyl, tetrahydrothienyl, S,S-dioxotetrahydrothienyl(tetramethylenesulfonyl), dithiolanyl, thiazolidinyl, oxazolidinyl, tetrahydropyranyl and pyrazolinyl moieties.
- In one embodiment, a 5- to 8-membered heterocyclyl moiety is morpholinyl, tetrahydrofuranyl or S,S-dioxotetrahydrothienyl.
- For the avoidance of doubt, although the above definitions of heteroaryl and heterocyclyl groups refer to an “N” moiety which can be present in the ring, as will be evident to a skilled chemist the N atom will carry a hydrogen atom (or will carry a substituent as defined above) if it is attached to each of the adjacent ring atoms via a single bond.
- As used herein, a C3-C10 carbocyclyl moiety is a monocyclic or polycyclic non-aromatic saturated or unsaturated hydrocarbon ring having from 3 to 10 carbon atoms.
- In one embodiment, it is a saturated ring system (i.e. a cycloalkyl moiety) having from 3 to 7 carbon atoms. Examples include adamantyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclopentenyl, cyclohexyl and cycloheptyl and bicycloheptyl.
- In one embodiment, a C3-C10 carbocyclyl moiety is adamantyl, cyclopentyl, cyclohexyl or bicycloheptyl moiety.
- In another embodiment, it is a C5-C6 cycloalkyl moiety.
- Examples of bicyclic ring systems in which the two rings are fused together include naphthyl, indanyl, quinolyl, tetrahydroquinolyl, benzofuranyl, indolyl, isoindolyl, indolinyl, benzofuranyl, 2,3-dihydro-bensofuranyl, benzothienyl, indazolyl, benzimidazolyl, benzthiazolyl, benzmorpholinyl, isoquinolyl, chromanyl, indenyl, 2,3-dihydro-indenyl, quinazolyl, quinoxalyl, isocromanyl, tetrahydronaphthyl, pyrido-oxazolyl, pyridothiazolyl, dihydrobenzofuranyl, 1,3-benzodioxolyl, 2,3-dihydro-1,4-benzodioxinyl, 1,3-benzodioxinyl and 3,4-dihydro-isochromenyl.
- In one embodiment, a bicyclic fused ring system is a naphthyl, indanyl, indolyl, benzofuranyl, benzothienyl, benzthiazolyl, benzmorpholinyl, pyrido-oxazolyl, pyridothiazolyl or dihydrobenzofuranyl moiety.
- Examples of tricyclic ring systems in which the three rings are fused together include xanthenyl, carbazolyl, acridinyl, phenothiazinyl, phenoxazinyl, dibenzofuranyl, dibenzothienyl, S,S,-dioxodibenzothienyl, fluorenyl, phenanthrenyl and anthracenyl. In one embodiment, a tricyclic fused ring system is a dibenzofuranyl or S,S,-dioxodibenzothienyl moiety.
- For the avoidance of doubt, when the phenyl, heteroaryl, carbocyclyl or heterocyclyl moieties in G1 and G2 are fused to one or two further rings, said fused rings may be substituted at one or more ring positions with such substituents as described above.
- In one embodiment, A is selected from phenyl, pyridyl or thienyl; said phenyl or pyridyl being optionally fused to a phenyl, a 5- or 6-membered heteroaryl, C5-6carbocyclyl or C5-6heterocyclyl ring. Examples of fused ring systems for A include naphthyl, indanyl, quinolyl, tetrahydroquinolyl, benzofuranyl, indolyl, benzothienyl, indazolyl, benzimidazolyl, benzthiazolyl, indenyl, 2,3-dihydro-indenyl, tetrahydronaphthyl, pyrido-oxazolyl, pyridothiazolyl, dihydrobenzofuranyl, 1,3-benzodioxolyl and 2,3-dihydro-1,4-benzodioxinyl.
- In one embodiment, A represents phenyl, pyridyl, thienyl or 2,3-dihydro-indenyl.
- In another embodiment, A is phenyl or pyridyl.
- In another embodiment, A is phenyl.
- In another embodiment, A is pyridyl.
- In another embodiment, A is thienyl.
- In another embodiment, A is 2,3-dihydro-indenyl.
- In one embodiment, R1 is independently selected from halogen, nitro, SF5, OH, CHO, C1-4alkyl or C1-4alkoxy; said C1-4alkyl or C1-4alkoxy being optionally substituted by OH or by one or more F atoms.
- In one embodiment, R1 is independently selected from halogen, CN, OH, CONR5R6, NR5R6, C1-4alkyl, C2-4alkynyl, C1-4alkoxy, G3, OG3 or OCH2G3; said C1-4alkyl or C2-4alkynyl being optionally substituted by OH or by one or more F atoms.
- In one embodiment, R1 is independently selected from halogen, CN, OH, CONR5R6, NR5R6, C1-4alkyl, C2-4alkynyl, C1-4alkoxy, G3, OG3 or OCH2G3; said C1-4alkyl or C2-4alkynyl being optionally substituted by OH.
- In one embodiment, G is phenyl, pyridyl or 2-methyl-1,3,4-oxadiazole.
- In one embodiment, R5 and R6 is hydrogen.
- In another embodiment, R1 is independently selected from halogen, C1-4alkyl or C1-4alkoxy; said C1-4alkyl or C1-4alkoxy being optionally substituted by OH or by one or more F atoms.
- In one embodiment, m represents an integer 0, 1 or 2.
- In one embodiment, m represents an integer 0 or 1. In another embodiment, m represents an integer 0.
- In one embodiment, each R3 is independently selected from hydrogen, CN and C1-4alkyl. In another embodiment, each R3 represents hydrogen.
- In one embodiment, L1 represents a direct bond, C1-3alkylene or C2-3alkenylene.
- In one embodiment, L1 represents a direct bond, C1-2alkylene or C2alkenylene. In another embodiment, L1 represents a direct bond.
- In one embodiment, L2 represents a direct bond, —O—, —OCH2—, —NHCONH— or —C≡C—.
- In one embodiment, L2 represents a direct bond, —O—, —OCH2—, C1-2alkylene or —C≡C—. In another embodiment, L2 represents a direct bond or —C≡C—. In another embodiment, L2 represents a direct bond. In another embodiment, L2 represents —C≡C—.
- In one embodiment, G1 represents phenyl or 5- or 6-membered heteroaryl, said phenyl or 5- or 6-membered heteroaryl being optionally fused to one or two further rings independently selected from phenyl, C5-6carbocyclyl or C5-6heterocyclyl ring.
- In one embodiment, G1 represents phenyl or 5- or 6-membered heteroaryl; optionally fused to one further ring independently selected from phenyl and 5- or 6-membered heteroaryl. In another embodiment, G1 represents phenyl; optionally fused to one further ring independently selected from phenyl and 5- or 6-membered heteroaryl. In another embodiment, G1 represents phenyl.
- In one embodiment, G2 represents H, C1-6alkyl, phenyl or 5- or 6-membered heteroaryl; said phenyl or 5- or 6-membered heteroaryl being optionally fused to one further ring independently selected from phenyl, a 5- or 6-membered heteroaryl, C5-6carbocyclyl or C5-6heterocyclyl ring.
- In one embodiment, G1 represents phenyl, naphtyl, pyridyl, thienyl, 2,3-dihydro-indenyl tetrahydronaphtyl, 2,3-dihydro-1,4-benzodioxinyl or S,S,-dioxodibenzothienyl.
- In one embodiment, G2 represents hydrogen, methyl, ethyl, tert-butyl, iso-propyl, phenyl, pyridyl, pyrimidyl, cyclopentyl, cyclopentenyl, cyclohexyl, furanyl, tetrahydrofuranyl, thienyl, thiazolyl, pyrazolyl, bensofuranyl, 2,3-dihydro-bensofuranyl, indolyl, tetrahydronaphtyl or dibenzofuranyl.
- In one embodiment, any phenyl, heteroaryl, carbocyclyl or heterocyclyl moieties in G1 and G2 are optionally substituted by one or more substituents independently selected from halogen, CN, NO2, C1-6alkyl and C1-6alkoxy; said C1-6alkyl or C1-6alkoxy being optionally substituted by OH or by one or more F atoms.
- In one embodiment, any phenyl, heteroaryl, carbocyclyl or heterocyclyl moieties in G1 and G2 are optionally substituted by one or more substituents independently selected from halogen, CN, OH, C1-4alkyl, C1thioalkoxy, NR12R13, —NHCOC(OH)(CH3)CF3, SO2NR10R11, and C1alkoxy; said C1-4alkyl or C1alkoxy being optionally substituted by OH or by one or more F atoms.
- In one embodiment, R10 is hydrogen.
- In one embodiment, R11, R12 and R13 is methyl.
- In another embodiment, any phenyl, heteroaryl, carbocyclyl or heterocyclyl moieties in G1 and G2 are optionally substituted by one or more substituents independently selected from halogen, C1-6alkyl and C1-6alkoxy; said C1-6alkyl being optionally substituted by OH or by one or more F atoms.
- In one embodiment, A is phenyl, pyridyl, thienyl or 2,3-dihydro-indenyl;
- R1 is independently selected from halogen, CN, OH, CONR5R6, NR5R6, C1-4alkyl, C2-4alkynyl, C1-4alkoxy, G3, OG3 and OCH2G3; said C1-4alkyl or C2-4alkynyl being optionally substituted by one or more substituents selected from OH and C1-4alkyl;
- m represents an integer 0, 1 or 2;
- G3 is phenyl, pyridyl or 2-methyl-1,3,4-oxadiazole;
- R3, R5, R6 and R10 represents hydrogen;
- L1 represents a direct bond, C1-3alkylene or C2-3alkenylene;
- L2 represents a direct bond, —O—, —OCH2—, —NHCONH— or —C≡C—;
- G1 represents phenyl or 5- or 6-membered heteroaryl; said phenyl or 5- or 6-membered heteroaryl being optionally fused to one or two further rings independently selected from phenyl, C5-6carbocyclyl or C5-6heterocyclyl ring;
- G2 represents H, C1-6alkyl, phenyl, 5- or 6-membered heteroaryl, C3-10carbocyclyl or C5-8heterocyclyl;
- said phenyl or 5- or 6-membered heteroaryl being optionally fused to one further ring independently selected from phenyl, 5- or 6-membered heteroaryl, C5-6carbocyclyl and C5-6heterocyclyl ring;
- said C1-6alkyl being optionally further substituted by one or more groups selected from OH, C1-6alkoxy or halogen.
- any phenyl, heteroaryl, carbocyclyl or heterocyclyl moieties in G1 and G2 being optionally substituted by one or more substituents independently selected from halogen, CN, OH, C1-4alkyl, C1thioalkoxy, NR12R13, —NHCOC(OH)(CH3)CF3, SO2NR10R11, and C1alkoxy;
- said C1-4alkyl or C1alkoxy being optionally substituted by OH or by one or more F atoms;
- R11 is methyl; R12 and R13 is methyl.
- In one embodiment, A is phenyl, pyridyl, thienyl or 2,3-dihydro-indenyl;
- R1 is independently selected from halogen, CN, OH, CONR5R6, NR5R6, C1-4alkyl, C2-4alkynyl, C1-4alkoxy, G3, OG3 and OCH2G3; said C1-4alkyl or C2-4alkynyl being optionally substituted by one or more substituents selected from OH and C1-4alkyl;
- m represents an integer 0, 1 or 2;
- G3 is phenyl, pyridyl or 2-methyl-1,3,4-oxadiazole;
- R3, R5, R6 and R10 represents hydrogen;
- L1 represents a direct bond, C1-3alkylene or C2-3alkenylene;
- L2 represents a direct bond, —O—, —OCH2—, —NHCONH— or —C≡C—;
- G1 represents phenyl, naphtyl, pyridyl, thienyl, 2,3-dihydro-indenyl tetrahydronaphtyl, 2,3-dihydro-1,4-benzodioxinyl or S,S,-dioxodibenzothienyl;
- G2 represents hydrogen, C1-4alkyl, phenyl, pyridyl, pyrimidyl, cyclopentyl, cyclopentenyl, cyclohexyl, furanyl, tetrahydrofuranyl, thienyl, thiazolyl, pyrazolyl, bensofuranyl, 2,3-dihydro-bensofuranyl, indolyl, tetrahydronaphtyl and dibenzofuranyl;
- any phenyl, heteroaryl, carbocyclyl or heterocyclyl moieties in G1 and G2 are optionally substituted by one or more substituents independently selected from halogen, CN, OH, C1-4alkyl, C1thioalkoxy, NR12R13, —NHCOC(OH)(CH3)CF3, SO2NR10R11, and C1alkoxy;
- said C1-4alkyl or C1alkoxy being optionally substituted by OH or by one or more F atoms;
- R11, R12 and R13 is methyl.
- In one embodiment A is phenyl, pyridyl or thienyl or 2,3-dihydro-indenyl;
- R1 is independently selected from chloro, fluoro, bromo, CN, OH, CONR5R6, methyl, 1-butynyl, methoxy, G3, OG3 or OCH2G3; said methyl and 1-butynyl being optionally substituted by OH or methyl;
- m represents an integer 0, 1 or 2;
- G3 is phenyl, pyridyl or 2-methyl-1,3,4-oxadiazole;
- R3, R5, R6 and R10 represents hydrogen;
- L1 represents a direct bond, ethylene, 1-methylethylene, 2-methylethylene, ethenylene, —C(CH3)CH—, —CHC(CH3)— or —CH2C(CH2)—;
- L2 represents a direct bond, —O—, —OCH2—, —NHCONH— or —C≡C—;
- G1 represents phenyl, naphtyl, pyridyl, thienyl, 2,3-dihydro-indenyl tetrahydronaphtyl, 2,3-dihydro-1,4-benzodioxinyl or S,S,-dioxodibenzothienyl;
- G2 represents hydrogen, methyl, ethyl, tert-butyl, iso-propyl phenyl, pyridyl, pyrimidyl, cyclopentyl, cyclopentenyl, cyclohexyl, furanyl, tetrahydrofuranyl, thienyl, thiazolyl, pyrazolyl, bensofuranyl, 2,3-dihydro-bensofuranyl, indolyl, tetrahydronaphtyl or dibenzofuranyl;
- any methyl, ethyl, tert-butyl, iso-propyl phenyl, pyridyl, pyrimidyl, cyclopentyl, cyclopentenyl, cyclohexyl, furanyl, tetrahydrofuranyl, thienyl, thiazolyl, pyrazolyl, bensofuranyl, 2,3-dihydro-bensofuranyl, indolyl, tetrahydronaphtyl or dibenzofuranyl in G1 and G2 are optionally substituted by one or more substituents independently selected from bromo, chloro, fluoro, CN, OH, methyl, tert-butyl, methylthio, NR12R13, —NHCOC(OH)(CH3)CF3, SO2NR10R11, and methoxy; said methyl, tert-butyl or methoxy being optionally substituted by OH or by one or more F atoms;
- R11, R12 and R13 is methyl
- In one embodiment, A is phenyl or pyridyl;
- R1 is independently selected from halogen, C1-4alkyl or C1-4alkoxy; said C1-4alkyl or C1-4alkoxy being optionally substituted by OH or by one or more F atoms; m represents an integer 0 or 1;
- each R3 represents hydrogen;
- L1 represents a direct bond;
- L2 represents a direct bond;
- G1 represents phenyl; optionally fused to one further ring independently selected from phenyl and 5- or 6-membered heteroaryl;
- G2 represents H, phenyl or 5- or 6-membered heteroaryl; optionally fused to one further ring independently selected from phenyl, a 5- or 6-membered heteroaryl, C5-6carbocyclyl or C5-6heterocyclyl ring; and any phenyl or heteroaryl moieties in G1 and G2 are optionally substituted by one or more substituents independently selected from halogen, C1-6alkyl and C1-6alkoxy; said C1-6alkyl being optionally substituted by OH or by one or more F atoms.
- In one embodiment, A is phenyl;
- m represents an integer 0;
- each R3 represents hydrogen;
- L1 represents a direct bond;
-
- L2 represents a direct bond;
- G1 represents phenyl; optionally fused to one further ring independently selected from phenyl and 5- or 6-membered heteroaryl;
- G2 represents H, phenyl or 5- or 6-membered heteroaryl; optionally fused to one further ring independently selected from phenyl, a 5- or 6-membered heteroaryl, C5-6carbocyclyl or C5-6heterocyclyl ring; and any phenyl or heteroaryl moieties in G1 and G2 are optionally substituted by one or more substituents independently selected from halogen, C1-6alkyl and C1-6alkoxy; said C1-6alkyl being optionally substituted by OH or by one or more F atoms.
- In one embodiment, A is phenyl;
- m represents an integer 0;
- each R3 represents hydrogen;
- L1 represents a direct bond;
- L2 represents —C≡C—;
- G1 represents phenyl; optionally fused to one further ring independently selected from phenyl and 5- or 6-membered heteroaryl;
- G2 represents C1-6alkyl optionally substituted by one or more groups selected from OH, C1-6alkoxy and halogen; and any phenyl or heteroaryl moieties in G1 is optionally substituted by one or more substituents independently selected from halogen, C1-6alkyl and C1-6alkoxy; said C1-6alkyl being optionally substituted by OH or by one or more F atoms.
- In one aspect we disclose a compound of formula (I) or a pharmaceutically acceptable salt thereof
- wherein:
- A is selected from phenyl or a 5- or 6-membered heteroaryl or a 5- or 6-membered heterocyclenyl moiety; said phenyl or 5- or 6-membered heteroaryl or 5- or 6-membered heterocyclenyl moiety in group A being optionally fused to a phenyl, a 5- or 6-membered heteroaryl, C5-6carbocyclyl or C5-6heterocyclyl ring;
- each R1 is independently selected from halogen, nitro, SF5, CN, NR5R6, OH, CHO, CO2C1-4alkyl, CONR5R6, C1-4alkyl, C2-4alkenyl, C2-4alkynyl, C1-4alkoxy, G3, OG3 or OCH2G3; said C1-4alkyl, C2-4alkenyl, C2-4alkynyl or C1-4alkoxy being optionally substituted by one or more substituents selected from OH, NR5R6, C1-4alkyl or F;
- m represents an integer 0, 1, 2, 3 or 4;
- each R3 is independently selected from hydrogen, CN and C1-4alkyl; said C1-4alkyl being optionally substituted with OH, CN, C1-4alkoxy, NR7R8, or one or more F atoms;
- L1 represents a direct bond, C1-4alkylene, C2-4alkenylene or C2-4alkynylene;
- L2 represents a direct bond, —O—, —OCH2—, C1-2alkylene, —C≡C— or —NHCONH—;
- G1 represents phenyl, 5- or 6-membered heteroaryl, C3-10carbocyclyl or C5-8heterocyclyl;
- G2 represents H, C1-6alkyl, phenyl, 5- or 6-membered heteroaryl, C3-10carbocyclyl or C5-8 heterocyclyl; said C1-6alkyl being optionally further substituted by one or more groups selected from OH, C1-6alkoxy and halogen;
- the phenyl, heteroaryl, carbocyclyl or heterocyclyl moieties in G1 and G2 being optionally fused to one or two further rings independently selected from phenyl, a 5- or 6-membered heteroaryl, C5-6carbocyclyl or C5-6heterocyclyl ring;
- any phenyl, heteroaryl, carbocyclyl or heterocyclyl moieties in G1 and G2 being optionally substituted by one or more substituents independently selected from halogen, OH, CN, NO2, CO2R9, C1-6alkyl, C1-6alkoxy, C1-4thioalkoxy, SO2NR10R11, NR12R13, —NHCOC(OH)(CH3)CF3 or —CH2OCH2CF2CHF2; said C1-6alkyl or C1-6alkoxy being optionally substituted by OH or by one or more F atoms;
- R9 represents C1-6alkyl;
- G3 represents phenyl or 5- or 6-membered heteroaryl; and
- each R4, R5, R6, R7, R8, R10, R11, R12 and R13 is independently selected from H or C1-4alkyl;
- with the proviso that the following compounds are excluded:
- 3-[(phenylsulfonyl)amino]pyridine-2-sulfonamide;
- 2-amino-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 4-tert-butyl-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 3-amino-4-hydroxy-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 4-bromo-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 4-fluoro-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 4-methyl-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 4-nitro-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-nitro-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 4-amino-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-methyl-4-[(2-sulfamoylphenyl)sulfamoyl]phenyl 2,2-dimethylpropanoate;
- 2-[(phenylsulfonyl) amino]-4-(trifluoromethyl)benzenesulfonamide;
- 2-{[(4-methylphenyl)sulfonyl]amino}-4-(trifluoromethyl)benzenesulfonamide; and
- N-(2-sulfamoylphenyl)quinoline-8-sulfonamide.
- In one aspect we disclose a compound of formula (I) or a pharmaceutically acceptable salt thereof
- wherein:
- A is selected from phenyl or a 5- or 6-membered heteroaryl or a 5- or 6-membered heterocyclenyl moiety; said phenyl or 5- or 6-membered heteroaryl or 5- or 6-membered heterocyclenyl moiety in group A being optionally fused to a phenyl, a 5- or 6-membered heteroaryl, C5-6carbocyclyl or C5-6heterocyclyl ring;
- each R1 is independently selected from halogen, nitro, SF5, CN, NR5R6, OH, CHO, CO2C1-4alkyl, CONR5R6, C1-4alkyl, C2-4alkenyl, C2-4alkynyl, C1-4alkoxy, G3, OG3 or OCH2G3; said C1-4alkyl, C2-4alkenyl, C2-4alkynyl or C1-4alkoxy being optionally substituted by one or more substituents selected from OH, NR5R6, C1-4alkyl or F;
- m represents an integer 0, 1, 2, 3 or 4;
- each R3 is independently selected from hydrogen, CN and C1-4alkyl; said C1-4alkyl being optionally substituted with OH, CN, C1-4alkoxy, NR7R8, or one or more F atoms;
- L1 represents a direct bond, C1-4alkylene, C2-4alkenylene or C2-4alkynylene;
- L2 represents a direct bond, —O—, —OCH2—, C1-2alkylene, —C≡C— or —NHCONH—;
- G1 represents phenyl, 5- or 6-membered heteroaryl, C3-10carbocyclyl or C5-8heterocyclyl;
- G2 represents H, C1-6alkyl, phenyl, 5- or 6-membered heteroaryl, C3-10carbocyclyl or C5-8heterocyclyl; said C1-6alkyl being optionally further substituted by one or more groups selected from OH, C1-6alkoxy and halogen;
- the phenyl, heteroaryl, carbocyclyl or heterocyclyl moieties in G1 and G2 being optionally fused to one or two further rings independently selected from phenyl, a 5- or 6-membered heteroaryl, C5-6carbocyclyl or C5-6heterocyclyl ring;
- any phenyl, heteroaryl, carbocyclyl or heterocyclyl moieties in G1 and G2 being optionally substituted by one or more substituents independently selected from halogen, OH, CN, NO2, CO2R9, C1-6alkyl, C1-6alkoxy, C1-4thioalkoxy, SO2NR11R12, NR12R13, —NHCOC(OH)(CH3)CF3 or —CH2OCH2CF2CHF2; said C1-6alkyl or C1-6alkoxy being optionally substituted by OH or by one or more F atoms;
- R9 represents C1-6alkyl;
- G3 represents phenyl or 5- or 6-membered heteroaryl; and
- each R4, R5, R6, R7, R8, R10, R11, R12 and R13 is independently selected from H or C1-4alkyl;
- with the proviso that the following compounds are excluded:
- 2-amino-N-(2-sulfamoylphenyl)benzenesulfonamide;
- methyl 4-(5-fluoro-3-methylbenzo[b]thiophene-2-sulfonamido)-3-sulfamoylbenzoate;
- 4-nitro-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-nitro-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 4-amino-N-(2-sulfamoylphenyl)benzenesulfonamide;
- N-(2-sulfamoylphenyl)quinoline-8-sulfonamide;
- for the use in therapy.
- In one aspect we disclose a compound of formula (I) or a pharmaceutically acceptable salt thereof
- wherein:
- A is selected from phenyl or a 5- or 6-membered heteroaryl or a 5- or 6-membered heterocyclenyl moiety; said phenyl or 5- or 6-membered heteroaryl or 5- or 6-membered heterocyclenyl moiety in group A being optionally fused to a phenyl, a 5- or 6-membered heteroaryl, C5-6carbocyclyl or C5-6heterocyclyl ring;
- each R1 is independently selected from halogen, nitro, SF5, CN, NR5R6, OH, CHO, CO2C1-4alkyl, CONR5R6, C1-4alkyl, C2-4alkenyl, C2-4alkynyl, C1-4alkoxy, G3, OG3 or OCH2G3; said C1-4alkyl, C2-4alkenyl, C2-4alkynyl or C1-4alkoxy being optionally substituted by one or more substituents selected from OH, NR5R6, C1-4alkyl or F;
- m represents an integer 0, 1, 2, 3 or 4;
- each R3 is independently selected from hydrogen, CN and C1-4alkyl; said C1-4alkyl being optionally substituted with OH, CN, C1-4alkoxy, NR7R8, or one or more F atoms;
- L1 represents a direct bond, C1-4alkylene, C2-4alkenylene or C2-4alkynylene;
- L2 represents a direct bond, —O—, —OCH2—, C1-2alkylene, —C≡C— or —NHCONH—;
- G1 represents phenyl, 5- or 6-membered heteroaryl, C3-10carbocyclyl or C5-8heterocyclyl;
- G2 represents H, C1-6alkyl, phenyl, 5- or 6-membered heteroaryl, C3-10carbocyclyl or C5-8heterocyclyl; said C1-6alkyl being optionally further substituted by one or more groups selected from OH, C1-6alkoxy and halogen;
- the phenyl, heteroaryl, carbocyclyl or heterocyclyl moieties in G1 and G2 being optionally fused to one or two further rings independently selected from phenyl, a 5- or 6-membered heteroaryl, C5-6carbocyclyl or C5-6heterocyclyl ring; any phenyl, heteroaryl, carbocyclyl or heterocyclyl moieties in G1 and G2 being optionally substituted by one or more substituents independently selected from halogen, OH, CN, NO2, CO2R9, C1-6alkyl, C1-6alkoxy, C1-4thioalkoxy, SO2NR10R11, NR12R13, —NHCOC(OH)(CH3)CF3 or —CH2OCH2CF2CHF2; said C1-6alkyl or C1-6alkoxy being optionally substituted by OH or by one or more F atoms;
- R9 represents C1-6alkyl;
- G3 represents phenyl or 5- or 6-membered heteroaryl; and
- each R4, R5, R6, R7, R8, R10, R11, R12 and R13 is independently selected from H or C1-4alkyl;
- in the manufacture of a medicament for use in treating acute or chronic pain, nociceptive pain or neuropathic pain.
- Examples of compounds of the invention include:
- 2-[2-(4-Benzofuran-2-ylphenyl)ethylsulfonylamino]benzenesulfonamide;
- 2-[2-[4-(2,3-Dihydrobenzofuran-2-yl)phenyl]ethylsulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-(4-Benzofuran-2-ylphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-(4-Benzofuran-2-ylphenyl)ethenyl]sulfonylamino]-5-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(4-Benzofuran-2-ylphenyl)ethenyl]sulfonylamino]-4-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(4-Benzofuran-2-ylphenyl)ethenyl]sulfonylamino]-6-fluoro-benzenesulfonamide;
- 2-[1-(4-Chlorophenyl)propan-2-ylsulfonylamino]benzenesulfonamide;
- 2-(1-Phenylpropan-2-ylsulfonylamino)benzenesulfonamide;
- 2-[[(E)-1-(4-Chlorophenyl)prop-1-en-2-yl]sulfonylamino]benzenesulfonamide;
- 2-[1-(4-Chlorophenyl)propan-2-ylsulfonylamino]-4-fluoro-benzenesulfonamide;
- 2-[[(E)-1-(4-chlorophenyl)prop-1-en-2-yl]sulfonylamino]-4-fluoro-benzenesulfonamide;
- 2-[[(E)-1-(4-Chlorophenyl)prop-1-en-2-yl]sulfonylamino]-5-fluoro-benzenesulfonamide;
- 2-[[(E)-1-(4-Chlorophenyl)prop-1-en-2-yl]sulfonylamino]-6-fluoro-benzenesulfonamide;
- 2-[[(E)-1-(4-Chlorophenyl)prop-1-en-2-yl]sulfonylamino]-3-fluoro-benzenesulfonamide;
- 2-[[(E)-1-(4-Chlorophenyl)prop-1-en-2-yl]sulfonylamino]-5-methyl-benzenesulfonamide;
- 2-[2-[4-(2-Furyl)phenyl]ethylsulfonylamino]benzenesulfonamide;
- 2-[2-[4-(Oxolan-2-yl)phenyl]ethylsulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-[4-(2-Furyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[2-[4-(Difluoromethoxy)phenyl]ethylsulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-[4-(Difluoromethoxy)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
- 2-[2-(4-Chlorophenyl)ethylsulfonylamino]-5-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(4-Chlorophenyl)ethenyl]sulfonylamino]-5-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(3,4-Dichlorophenyl)ethenyl]sulfonylamino]-4-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(3,4-Dichlorophenyl)ethenyl]sulfonylamino]-5-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(3,4-Dichlorophenyl)ethenyl]sulfonylamino]-6-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(5-Bromo-2-methoxy-phenyl)ethenyl]sulfonylamino]-4-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(5-Bromo-2-methoxy-phenyl)ethenyl]sulfonylamino]-5-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(5-Bromo-2-methoxy-phenyl)ethenyl]sulfonylamino]-6-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(5-Bromo-2-methoxy-phenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-(4-Cyanophenyl)ethenyl]sulfonylamino]-4-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(4-Cyanophenyl)ethenyl]sulfonylamino]-5-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(4-Cyanophenyl)ethenyl]sulfonylamino]-6-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(4-Cyanophenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 4-Fluoro-2-[[(E)-2-[4-(2-furyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide;
- 5-Fluoro-2-[[(E)-2-[4-(2-furyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide;
- 2-Fluoro-6-[[(E)-2-[4-(2-furyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide;
- 4-Fluoro-2-[[(E)-2-(4-methylsulfanylphenyl)ethenyl]sulfonylamino]-benzenesulfonamide;
- 5-Fluoro-2-[[(E)-2-(4-methylsulfanylphenyl)ethenyl]sulfonylamino]-benzenesulfonamide;
- 2-Fluoro-6-[[(E)-2-(4-methylsulfanylphenyl)ethenyl]sulfonylamino]-benzenesulfonamide;
- 2-[[(E)-2-(4-Methylsulfanylphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-[4-(Difluoromethoxy)phenyl]ethenyl]sulfonylamino]-4-fluoro-benzenesulfonamide;
- 2-[[(E)-2-[4-(Difluoromethoxy)phenyl]ethenyl]sulfonylamino]-5-fluoro-benzenesulfonamide;
- 2-[[(E)-2-[4-(Difluoromethoxy)phenyl]ethenyl]sulfonylamino]-6-fluoro-benzenesulfonamide;
- 2-[2-(4-Chlorophenyl)ethylsulfonylamino]-5-(hydroxymethyl)-benzenesulfonamide;
- 2-[2-(4-Chloro-phenyl)-ethanesulfonylamino]-5-methylbenzenesulfonamide;
- 2-[2-(4-Chlorophenyl)ethylsulfonylamino]-5-(pyridin-2-ylmethoxy)benzenesulfonamide;
- 2-[2-(4-Chlorophenyl)ethylsulfonylamino]-5-[(5-methyl-1,3,4-oxadiazol-2-yl)methoxy]benzenesulfonamide;
- 2-[2-(2-Chloro-phenyl)-ethenesulfonylamino]-benzenesulfonamide;
- 2-[2-(4-Chloro-2-methoxy-phenyl)-ethenesulfonylamino]-benzenesulfonamide;
- 2-Fluoro-6-[[(E)-2-(4-phenylphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-(3-Chlorophenyl)ethenyl]sulfonylamino]-5-fluoro-benzenesulfonamide;
- 5-Fluoro-2-[[(E)-2-(4-phenylphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 4-Fluoro-2-[[(E)-2-(4-phenylphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 5-Fluoro-2-[[(E)-2-[3-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
- 5-Fluoro-2-[[(E)-2-(2-methoxyphenyl)ethenyl]sulfonylamino]-benzenesulfonamide;
- 2-[[(E)-2-(3-Chlorophenyl)ethenyl]sulfonylamino]-6-fluoro-benzenesulfonamide;
- 2-Fluoro-6-[[(E)-2-(2-methoxyphenyl)ethenyl]sulfonylamino]-benzenesulfonamide;
- 4-Fluoro-2-[[(E)-2-(2-methoxyphenyl)ethenyl]sulfonylamino]-benzenesulfonamide;
- 3-Fluoro-2-[[(E)-2-(4-phenylphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 3-Fluoro-2-[[(E)-2-(2-methoxyphenyl)ethenyl]sulfonylamino]-benzenesulfonamide;
- 4-Fluoro-2-[[(E)-2-[3-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
- 2-Fluoro-6-[[(E)-2-(2-fluorophenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-(3-Chlorophenyl)ethenyl]sulfonylamino]-5-methyl-benzenesulfonamide;
- 2-[[(E)-2-(3-Chlorophenyl)ethenyl]sulfonylamino]-3-fluoro-benzenesulfonamide;
- 3-Fluoro-2-[[(E)-2-[3-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
- 2-Fluoro-6-[[(E)-2-[3-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
- 4-Fluoro-2-[[(E)-2-(2-fluorophenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 5-Methyl-2-[[(E)-2-[3-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
- 2-[2-(4-Cyclopentylphenyl)ethylsulfonylamino]benzenesulfonamide;
- 2-[2-(4-Tert-butylphenyl)ethylsulfonylamino]benzenesulfonamide;
- 4-Fluoro-2-[2-(4-tert-butylphenyl)ethylsulfonylamino]benzenesulfonamide;
- 2-[2-(4-Chlorophenyl)ethylsulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-(4-Chlorophenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 2-(Phenethylsulfonylamino)benzenesulfonamide;
- 2-[[(E)-2-Phenylethenyl]sulfonylamino]benzenesulfonamide;
- 2-[2-(2-Chlorophenyl)ethylsulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-(4-Methoxyphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-(4-Chlorophenyl)ethenyl]sulfonylamino]-4-fluoro-benzenesulfonamide;
- 2-(2-Phenylpropylsulfonylamino)benzenesulfonamide;
- 2-[[(E)-2-(4-Chlorophenyl)ethenyl]sulfonylamino]-6-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(2,6-Dichlorophenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-(4-Methylphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-(2-Methoxyphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-Naphthalen-2-ylethenyl]sulfonylamino]benzenesulfonamide;
- 2-[2-(4-Chlorophenyl)ethylsulfonylamino]-4-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(4-Chlorophenyl)prop-1-enyl]sulfonylamino]benzenesulfonamide;
- 2-[2-(4-Chlorophenyl)prop-2-enylsulfonylamino]benzenesulfonamide;
- 2-Fluoro-6-[[(E)-2-naphthalen-2-ylethenyl]sulfonylamino]benzenesulfonamide;
- 2-Fluoro-6-[[(E)-2-[2-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
- 2-Fluoro-6-[[(E)-2-[4-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
- 2-[[(E)-2-(4-Bromophenyl)ethenyl]sulfonylamino]-6-fluoro-benzenesulfonamide;
- 4-Fluoro-2-[[(E)-2-naphthalen-2-ylethenyl]sulfonylamino]benzenesulfonamide;
- 4-Fluoro-2-[[(E)-2-[2-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
- 4-Fluoro-2-[[(E)-2-[4-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
- 2-[[(E)-2-(4-Bromophenyl)ethenyl]sulfonylamino]-4-fluoro-benzenesulfonamide;
- 5-Methyl-2-[[(E)-2-naphthalen-2-ylethenyl]sulfonylamino]benzenesulfonamide;
- 5-Methyl-2-[[(E)-2-[4-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
- 2-[[(E)-2-(4-Bromophenyl)ethenyl]sulfonylamino]-5-methyl-benzenesulfonamide;
- 5-Fluoro-2-[[(E)-2-naphthalen-2-ylethenyl]sulfonylamino]benzenesulfonamide;
- 5-Fluoro-2-[[(E)-2-[2-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
- 5-Fluoro-2-[[(E)-2-[4-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
- 2-[[(E)-2-(4-Bromophenyl)ethenyl]sulfonylamino]-5-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(2,6-Difluorophenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-[2-(Trifluoromethyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-[4-(Trifluoromethyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-(4-Bromophenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[(E)-2-(4-Bromophenyl) ethylsulfonylamino]benzenesulfonamide;
- 2-[2-(4-Chlorophenyl)propylsulfonylamino]benzenesulfonamide;
- 2-[2-(2-Methoxyphenyl)ethylsulfonylamino]benzenesulfonamide;
- 2-(2-Naphthalen-2-ylethylsulfonylamino)benzenesulfonamide;
- 4-Fluoro-2-(2-naphthalen-2-ylethylsulfonylamino)benzenesulfonamide;
- 2-[(4-Chlorophenyl)methylsulfonylamino]benzenesulfonamide;
- 2-[[3-(2,3-Dichlorophenyl)phenyl]sulfonylamino]benzenesulfonamide;
- 2-[[3-(3,5-difluorophenyl)phenyl]sulfonylamino]benzenesulfonamide;
- 3-(5-Chloro-2-methoxy-phenyl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[[3-(3-Cyanophenyl)phenyl]sulfonylamino]benzenesulfonamide;
- 2-[[3-(4-Cyanophenyl)phenyl]sulfonylamino]benzenesulfonamide;
- 3-(2,4-Dimethoxypyrimidin-5-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[[3-(3-Furyl)phenyl]sulfonylamino]benzenesulfonamide;
- 2-[[3-(1H-Indol-5-yl)phenyl]sulfonylamino]benzenesulfonamide;
- 2-[[3-[3-(Trifluoromethoxy)phenyl]phenyl]sulfonylamino]benzenesulfonamide;
- N-(2-Sulfamoylphenyl)-2,3-dihydro-1H-indene-5-sulfonamide;
- 7-Methyl-N-(2-sulfamoylphenyl)-10-oxa-7-azabicyclo[4.4.0]deca-1,3,5-triene-3-sulfonamide;
- 2-(3,4-Dichlorophenyl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[[4-[3-Chloro-5-(trifluoromethyl)pyridin-2-yl]oxyphenyl]sulfonylamino]benzenesulfonamide;
- N-(2-Sulfamoylphenyl)tetralin-2-sulfonamide;
- 4-Phenylmethoxy-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[(4-Cyclohexylphenyl)sulfonylamino]benzenesulfonamide;
- 3-Phenyl-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 5-(1-Methyl-5-trifluoromethyl-1H-pyrazol-3-yl)-thiophene-2-sulfonic acid (2-sulfamoyl-phenyl)-amide;
- N-(2-Sulfamoylphenyl)-4-(trifluoromethyl)benzenesulfonamide;
- 2-[[3-(2,5-Dichlorophenyl)phenyl]sulfonylamino]benzenesulfonamide;
- 3-Dibenzofuran-4-yl-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[[3-[4-(Trifluoromethyl)phenyl]phenyl]sulfonylamino]benzenesulfonamide;
- 3-(3-Methoxyphenyl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 3-Benzofuran-2-yl-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[[3-(2,3-Dihydrobenzofuran-5-yl)phenyl]sulfonylamino]benzenesulfonamide;
- 3-(6-Methoxypyridin-3-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[[3-(2,4-Dichlorophenyl)phenyl]sulfonylamino]benzenesulfonamide;
- 3-(1-Methylindol-2-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[[3-[3-(Trifluoromethyl)phenyl]phenyl]sulfonylamino]benzenesulfonamide;
- 4-Benzofuran-2-yl-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[[3-(2,4-Difluorophenyl)phenyl]sulfonylamino]benzenesulfonamide;
- 5-Bromo-2-[[3-(3,4-dichlorophenyl)phenyl]sulfonylamino]benzenesulfonamide;
- 2-[[3-(3,4-Dichlorophenyl)phenyl]sulfonylamino]-5-phenyl-benzenesulfonamide;
- 2,3-Difluoro-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2,3-Difluoro-N-(4-fluoro-2-sulfamoyl-phenyl)benzenesulfonamide;
- 3-Chloro-2-fluoro-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2,3-Dichloro-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[[3-(3,4-Dichlorophenyl)phenyl]sulfonylamino]-5-methyl-benzenesulfonamide;
- 4-[[3-(3,4-Dichlorophenyl)phenyl]sulfonylamino]-3-sulfamoyl-benzamide;
- 5-Methyl-2-[[3-[3-(trifluoromethoxy)phenyl]phenyl]sulfonylamino]-benzenesulfonamide;
- 2-(2,3-Dichloro-benzenesulfonylamino)-5-hydroxymethyl-benzenesulfonamide;
- 2-(2,3-Dichloro-benzenesulfonylamino)-5-methyl-benzenesulfonamide;
- 2-[[3-(3,4-Dichlorophenyl)phenyl]sulfonylamino]-5-phenoxy-benzenesulfonamide;
- 5-Phenoxy-2-[[3-[3-(trifluoromethoxy)phenyl]phenyl]sulfonylamino]-benzenesulfonamide;
- 2-(2,3-Dichloro-benzenesulfonylamino)-5-hydroxy-benzenesulfonamide;
- (2R)—N-[2-Chloro-4-[(2-sulfamoylphenyl)sulfamoyl]phenyl]-3,3,3-trifluoro-2-hydroxy-2-methyl-propanamide;
- 5-Chloro-N-(2-sulfamoylphenyl)thiophene-2-sulfonamide;
- 4,5-Dichloro-N-(2-sulfamoylphenyl)thiophene-2-sulfonamide;
- 3,4-Dichloro-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 3-(6-Methoxypyridin-2-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[[4-[(2-Chloro 1,3-thiazol-5-yl)methoxy]phenyl]sulfonylamino]-benzenesulfonamide;
- 3-(5-Fluoro-6-methoxy-pyridin-3-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 3-(2-Methoxypyrimidin-5-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 3-(4-Methylpyridin-3-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 3-(2-Methoxypyridin-3-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[[3-(5-Mhloropyridin-3-yl)phenyl]sulfonylamino]benzenesulfonamide;
- 3-(5-Chloro-6-methoxy-pyridin-3-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 3-(6-Dimethylaminopyridin-3-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[[3-[3-(Hydroxymethyl)phenyl]phenyl]sulfonylamino]benzenesulfonamide;
- 2-[[3-[4-(Hydroxymethyl)phenyl]phenyl]sulfonylamino]benzenesulfonamide;
- 2-[[4-(3,4-Dichlorophenyl)phenyl]sulfonylamino]benzenesulfonamide;
- 2-[[3-(3,4-Dichlorophenyl)phenyl]sulfonylamino]benzenesulfonamide;
- 2-[[4-(4-Chlorophenoxy)phenyl]sulfonylamino]benzenesulfonamide;
- 3-(4-Chlorophenyl)-1-[3-methyl-4-[(2-sulfamoylphenyl)sulfamoyl]phenyl]urea;
- N-Methyl-N′-(2-sulfamoylphenyl)dibenzo[b,d]thiophene-2,8-disulfonamide 5,5-dioxide;
- 2,3-Dichloro-N-(4,5-difluoro-2-sulfamoylphenyl)benzenesulfonamide;
- (E)-2-(2-(3,4-Dichlorophenyl)vinylsulfonamido)-5-(3-hydroxy-3-methylbut-1-ynyl)benzenesulfonamide;
- 4-Cyano-2-(2-(3,4-dichlorophenyl)ethylsulfonamido)benzenesulfonamide;
- 2-(2-(4-Chloro-2-methoxyphenyl)ethylsulfonamido)-4-cyanobenzenesulfonamide;
- 2-(2-(4-Chloro-2-methoxyphenyl)ethylsulfonamido)-5-cyanobenzenesulfonamide;
- 2-(2-(4-(3,3-dimethylbut-1-ynyl)phenyl)ethylsulfonamido)benzenesulfonamide;
- 2-(2-(4-(3-Hydroxy-3-methylbut-1-ynyl)phenyl)ethylsulfonamido)benzenesulfonamide;
- 3-(3,3-Dimethylbut-1-ynyl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-(2-(4-Chloro-2-methoxyphenyl)ethylsulfonamido)benzenesulfonamide;
- 2-(2-(6-(3,3-Dimethylbut-1-ynyl)pyridin-3-yl)ethylsulfonamido)benzenesulfonamide;
- 2-(2-(6-(cyclohexylethynyl)pyridin-3-yl)ethylsulfonamido)benzenesulfonamide;
- 2-(2-(4-Bromophenyl)ethylsulfonamido)-5-methylbenzenesulfonamide;
- (E)-2-(2-(4-Bromophenyl)vinylsulfonamido)-5-methylbenzenesulfonamide;
- 3-(2-Methylthiazol-4-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- (E)-2-(2-(3-(Trifluoromethyl)phenyl)vinylsulfonamido)benzenesulfonamide;
- (E)-2-(2-(2-Fluorophenyl)vinylsulfonamido)benzenesulfonamide;
- 5-Chloro-2-(2-(4-chloro-2-methoxyphenyl)ethylsulfonamido)benzenesulfonamide;
- 2,3-Dichloro-N-(4-chloro-2-sulfamoylphenyl)benzenesulfonamide;
- 2,3-Dichloro-N-(3,5-difluoro-2-sulfamoylphenyl)benzenesulfonamide;
- 2-(2-(3,4-Dichlorophenyl)ethylsulfonamido)-4,6-difluorobenzenesulfonamide;
- 2-(2-(4-Chloro-2-methoxyphenyl)ethylsulfonamido)-4,6-difluorobenzenesulfonamide;
- 5-Cyano-2-(2-(3,4-dichlorophenyl)ethylsulfonamido)benzenesulfonamide;
- (E)-2-(2-(3,4-Dichlorophenyl)vinylsulfonamido)-4-methylbenzenesulfonamide;
- 2-(2-(4-Chloro-2-methoxyphenyl)ethylsulfonamido)-4-methylbenzenesulfonamide;
- 5-Chloro-2-(2-(3,4-dichlorophenyl)ethylsulfonamido)benzenesulfonamide;
- 4-(3-(5-Chloro-6-methoxypyridin-3-yl)phenylsulfonamido)pyridine-3-sulfonamide;
- 4-(2-(4-(Benzofuran-2-yl)phenyl)ethylsulfonamido)pyridine-3-sulfonamide;
- 2-(2-(4-Chloro-2-methoxyphenyl)ethylsulfonamido)-5-methylbenzenesulfonamide;
- 4-(2-(4-Chloro-2-methoxyphenyl)ethylsulfonamido)pyridine-3-sulfonamide;
- (E)-2-(2-(4-Cyclopentenylphenyl)vinylsulfonamido)benzenesulfonamide;
- (E)-2-(2-(4-(3,3-Dimethylbut-1-ynyl)phenyl)vinylsulfonamido)benzenesulfonamide;
- (E)-2-(2-(4-(3-Hydroxy-3-methylbut-1-ynyl)phenyl)vinylsulfonamido)-benzenesulfonamide;
- 2-{2-[4-(3,3-Dimethyl-but-1-ynyl)-phenyl]-ethanesulfonylamino}-5-methyl-benzenesulfonamide;
- 2-{2-[4-(3,3-Dimethyl-but-1-ynyl)-phenyl]-ethanesulfonylamino}-5-hydroxymethyl-benzenesulfonamide;
- 2-[2-(4-Cyclopentylethynyl-phenyl)-ethanesulfonylamino]-5-hydroxymethyl-benzenesulfonamide;
- 2-[2-(4-Chloro-2-methoxy-phenyl)-ethanesulfonylamino]-5-hydroxymethyl-benzenesulfonamide;
- 3-{2-[4-(3,3-Dimethyl-but-1-ynyl)-phenyl]-ethanesulfonylamino}-thiophene-2-sulfonic acid amide;
- 3-[3-(5-Chloro-6-methoxy-pyridin-3-yl)-benzenesulfonylamino]-thiophene-2-sulfonic acid amide;
- (E)-2-(2-(4-(1,1,2,2-tetrafluoroethoxy)phenyl)vinylsulfonamido)benzenesulfonamide;
- (E)-2-(2-(2-(difluoromethoxy)phenyl)vinylsulfonamido)benzenesulfonamide;
- (E)-2-(2-(4′-(trifluoromethyl)biphenyl-4-yl)vinylsulfonamido)benzenesulfonamide;
- (E)-2-(2-(2-isopropoxyphenyl)vinylsulfonamido)benzenesulfonamide;
- (E)-2-(2-(2-(methylthio)phenyl)vinylsulfonamido)benzenesulfonamide;
- (E)-2-(2-(2-(trifluoromethoxy)phenyl)vinylsulfonamido)benzenesulfonamide;
- (E)-2-(2-(2-ethoxyphenyl)vinylsulfonamido)benzenesulfonamide;
- 2-(2-(4-(1,1,2,2-tetrafluoroethoxy)phenyl)ethylsulfonamido)benzenesulfonamide;
- 2-(2-(4′-(trifluoromethyl)biphenyl-4-yl)ethylsulfonamido)benzenesulfonamide;
- 2-[2-(2-Hydroxy-phenyl)-ethenesulfonylamino]-benzenesulfonamide;
- 4-Chloro-2-(2-(4-(difluoromethoxy)phenyl)ethylsulfonamido)benzenesulfonamide;
- 4-Chloro-2-(2-(2-methoxyphenyl)ethylsulfonamido)benzenesulfonamide;
- 4-Chloro-2-(2-(4-chloro-2-methoxyphenyl)ethylsulfonamido)benzenesulfonamide;
- 2-(2-(3,4-Dichlorophenyl)ethylsulfonamido)-3,5-difluorobenzenesulfonamide;
- 2,3-Dichloro-N-(4-fluoro-5-methoxy-2-sulfamoylphenyl)benzenesulfonamide;
- 4-Chloro-2-(2-(3,4-dichlorophenyl)ethylsulfonamido)benzenesulfonamid;
- 5-Chloro-2-(2-(4-chlorophenyl)ethylsulfonamido)-4-fluorobenzenesulfonamide;
- 5-Chloro-2-(2-(3,4-dichlorophenyl)ethylsulfonamido)-4-fluorobenzenesulfonamide;
- 5-(2-(4-(Furan-2-yl)phenyl)ethylsulfonamido)-2,3-dihydro-1H-indene-4-sulfonamide;
- 6-(2-(4-(Furan-2-yl)phenyl)ethylsulfonamido)-2,3-dihydro-1H-indene-5-sulfonamid;
- 5-(2-(3,4-Dichlorophenyl)ethylsulfonamido)-2,3-dihydro-1H-indene-4-sulfonamide;
- (E)-5-(2-(4-Chlorophenyl)vinylsulfonamido)-2,3-dihydro-1H-indene-4-sulfonamide;
- (E)-6-(2-(4-Chlorophenyl)vinylsulfonamido)-2,3-dihydro-1H-indene-5-sulfonamide;
- 2-[[3-(3,4-Dichlorophenyl)phenyl]sulfonylamino]-N-methyl-benzenesulfonamide; and
- 2-(2-(6-((4-(Trifluoromethyl)phenyl)ethynyl)pyridin-3-yl)ethylsulfonamido)-benzenesulfonamide
- and pharmaceutically acceptable salts of any one thereof.
- The present invention further provides a process for the preparation of a compound of formula (I) or a pharmaceutically acceptable salt thereof as defined above which comprises,
- (a) reacting a compound of formula (II)
- wherein R1, R3, A and m are as defined in formula (I),
- with a compound of formula (III)
- wherein L1, L2, G1 and G2 are as defined in formula (I) and X represents a leaving group such as a halogen; or
- (b) when L2 represents a direct bond and G1 and G2 are both aromatic moieties, reacting a compound of formula (IV)
- wherein Hal represents a halogen atom and R1, R3, A, m and L1 are as defined in formula (I), with a nucleophile G2-M wherein M represents an organo-tin or organo boronic acid group; and optionally after (a) or (b) carrying out one or more of the following:
-
- converting the compound obtained to a further compound of the invention
- forming a pharmaceutically acceptable salt of the compound.
- In process (a), the reaction may conveniently be carried out in an organic solvent such as acetonitrile, dichloromethane, N,N-dimethylformamide, pyridine or N-methylpyrrolidinone at a temperature, for example, in the range from -20 ° C. to the boiling point of the solvent. The leaving group is a halogen such as fluorine, chlorine, bromine or iodine, preferrably chlorine. If necessary or desired, a base may be added. In one embodiment, the solvent is pyridine and the aniline is reacted with a sulfonyl chloride at room temperature.
- In process (b), the reaction may conveniently be carried out by reaction with an appropriate aryl boronic acid or an aryl boronic ester. The reaction may be carried out using a suitable palladium catalyst such as Pd(PPh3)4, Pd(dppf)Cl2, or Pd(OAc)2 or Pd2(dba)3 together with a suitable ligand such as P(tert-butyl)3, 2-(dicyclohexylphosphino)biphenyl, or 2-(2′,6′-dimethoxybiphenyl)-dicyclohexylphosphine, or a nickel catalyst such as nickel on charcoal or Ni(dppe)Cl2 together with zinc and sodium triphenylphosphinetrimetasulfonate. A suitable base such as an alkyl amine, e.g., triethylamine, or potassium carbonate, sodium carbonate, cesium carbonate, sodium hydroxide or cesium fluoride may be used in the reaction, which can be performed in the temperature range of +20° C. to +160° C., using an oil bath or a microwave oven, in a suitable solvent or solvent mixture such as toluene, tetrahydrofuran, ethanol, dimethoxyethane/water, N,N-dimethylformamide or dioxane. The boronic acid or boronic ester may be formed in situ, by reaction of the corresponding aryl halide (e.g., the aryl bromide) with an alkyllithium reagent such as butyllithium to form an intermediate aryl lithium species, which then is reacted with a suitable boron compound, e.g., trimethyl borate, tributyl borate or triisopropyl borate.
- Alternatively, the reaction may be carried out by reaction with an appropriate alkyne. The reaction may be carried out using a suitable palladium catalyst such as Pd(PPh3)4, PdCl2(PPh3)2, [PdCl2(CH3CN)2]or Pd(PPh3)2(OAc)2. The reaction may be preformed in the presence of a suitable ligand such as Xphos. The reaction may also be preformed in the presence of a suitable copper catalyst such as copper(I) iodide. A suitable base such as triethylamine, buthylamine, diisopropylamine, N,N-diisopropylethylamine or cesium carbonate may be used in the reaction, which can be performed in the temperature range of +20° C. to +160° C., using an oil bath or a microwave oven, in a suitable solvent or a mixture of solvents such as N,N-dimethylformamide, dimethyl sulfoxide, acetonitrile, toluene, tetrahydrofuran, dimethoxyethane/water or dioxane.
- Specific processes for the preparation of compounds of Formula (I) are disclosed within the Examples section of the present specification. Such processes form an aspect of the present invention.
- The necessary starting materials are either commercially available, are known in the literature or may be prepared using known techniques. Specific processes for the preparation of certain key starting materials are disclosed within the Examples section of the present specification and such processes form an aspect of the present invention.
- Certain intermediates are novel. Such novel intermediates form another aspect of the invention.
- Compounds of formula (I) can be converted into further compounds of formula (I) using standard procedures.
- It will be appreciated by those skilled in the art that in the processes of the present invention certain functional groups such as hydroxyl or amino groups may need to be protected by protecting groups. Thus, the preparation of the compounds of formula (I) may involve, at an appropriate stage, the addition and/or removal of one or more protecting groups.
- The protection and deprotection of functional groups is described in ‘Protective Groups in Organic Chemistry’, edited by J. W. F. McOmie, Plenum Press (1973) and ‘Protective Groups in Organic Synthesis’, 3rd edition, T. W. Greene and P. G. M. Wuts, Wiley-Interscience (1999).
- As used herein, a pharmaceutically acceptable salt is a salt with a pharmaceutically acceptable acid or base. Pharmaceutically acceptable acids include both inorganic acids such as hydrochloric, sulphuric, phosphoric, diphosphoric, hydrobromic or nitric acid and organic acids such as citric, fumaric, maleic, malic, ascorbic, succinic, tartaric, benzoic, acetic, methanesulphonic, ethanesulphonic, benzenesulphonic orp-toluenesulphonic acid. Pharmaceutically acceptable bases include alkali metal (e.g., sodium or potassium) and alkali earth metal (e.g., calcium or magnesium) hydroxides and organic bases such as alkyl amines, aralkyl amines and heterocyclic amines.
- Compounds of formula (I) are capable of existing in stereoisomeric forms. It will be understood that the invention encompasses the use of all geometric and optical isomers (including atropisomers) of the compounds of formula (I) and mixtures thereof including racemates. The use of tautomers and mixtures thereof also form an aspect of the present invention. Enantiomerically pure forms are particularly desired.
- The compounds of formula (I) and their pharmaceutically acceptable salts have activity as pharmaceuticals, in particular as selective inhibitors of the microsomal prostaglandin E synthase-1 enzyme, and may therefore be beneficial in the treatment or prophylaxis of pain and of inflammatory diseases and conditions. Furthermore, by selectively inhibiting the pro-inflammatory PGE2, it is believed the compounds of the invention would have a reduced potential for side effects associated with the inhibition of other prostaglandins by conventional non-steroidal anti-inflammatory drugs, such as gastrointestinal and renal toxicity.
- More particularly, the compounds of formula (I) and their pharmaceutically acceptable salts may be used in the treatment of osteoarthritis, rheumatoid arthritis, acute or chronic pain, neuropathic pain, apneal/SID, wound healing, cancer, benign or malignant neoplasias, stroke, atherosclerosis and Alzheimer's disease.
- Even more particularly, the compounds of formula (I) and their pharmaceutically acceptable salts may be used in the treatment of osteoarthritis, rheumatoid arthritis, benign or malignant neoplasias or acute or chronic pain.
- Thus, the present invention provides a compound of formula (I) or a pharmaceutically-acceptable salt thereof as hereinbefore defined for use in therapy.
- In a further aspect, the present invention provides the use of a compound of formula (I) or a pharmaceutically acceptable salt thereof as hereinbefore defined in the manufacture of a medicament for use in therapy.
- In a further aspect, the present invention provides the use of a compound of formula (I) or a pharmaceutically acceptable salt thereof as hereinbefore defined in the manufacture of a medicament for the treatment of human diseases or conditions in which modulation of microsomal prostaglandin E synthase-1 activity is beneficial.
- In a further aspect, the present invention provides the use of a compound of formula (I) or a pharmaceutically acceptable salt thereof as hereinbefore defined in the manufacture of a medicament for use in the treatment of an inflammatory disease or condition.
- In a further aspect, the present invention provides the use of a compound of formula (I) or a pharmaceutically acceptable salt thereof as hereinbefore defined in the manufacture of a medicament for use in treating osteoarthritis, rheumatoid arthritis, acute or chronic pain, neuropathic pain, apneal/SID, wound healing, cancer, benign or malignant neoplasias, stroke, atherosclerosis or Alzheimer's disease.
- In a further aspect, the present invention provides the use of a compound of formula (I) or a pharmaceutically acceptable salt thereof as hereinbefore defined in the manufacture of a medicament for use in treating osteoarthritis, rheumatoid arthritis, benign or malignant neoplasias or acute or chronic pain.
- In another aspect, the invention provides a compound of formula (I) or a pharmaceutically acceptable salt thereof as hereinbefore defined for use as a medicament.
- In another aspect, the invention provides a compound of formula (I) or a pharmaceutically acceptable salt thereof as hereinbefore defined for the treatment of diseases or conditions in which modulation of microsomal prostaglandin E synthase-1 activity is beneficial.
- In another aspect, the invention provides a compound of formula (I) or a pharmaceutically acceptable salt thereof as hereinbefore defined for the treatment of an inflammatory disease or condition.
- In another aspect, the invention provides a compound of formula (I) or a pharmaceutically acceptable salt thereof as hereinbefore defined for the treatment of osteoarthritis, rheumatoid arthritis, acute or chronic pain, neuropathic pain, apneal/SID, wound healing, cancer, benign or malignant neoplasias, stroke, atherosclerosis or Alzheimer's disease.
- In another aspect, the invention provides a compound of formula (I) or a pharmaceutically acceptable salt thereof as hereinbefore defined for the treatment of osteoarthritis, rheumatoid arthritis, benign or malignant neoplasias, apnea, sudden infant death (SID), atherosclerosis, cancer, aneurysm, hyperthermia, myositis, Alzheimer's disease or arthritis.
- In another aspect, the invention provides a compound of formula (I) or a pharmaceutically acceptable salt thereof as hereinbefore defined for the treatment of osteoarthritis, rheumatoid arthritis, benign or malignant neoplasias or acute or chronic pain.
- In the context of the present specification, the term “therapy” also includes “prophylaxis” unless there are specific indications to the contrary. The terms “therapeutic” and “therapeutically” should be construed accordingly.
- Prophylaxis is expected to be particularly relevant to the treatment of persons who have suffered a previous episode of, or are otherwise considered to be at increased risk of, the disease or condition in question. Persons at risk of developing a particular disease or condition generally include those having a family history of the disease or condition, or those who have been identified by genetic testing or screening to be particularly susceptible to developing the disease or condition.
- The invention also provides a method of treating, or reducing the risk of, a disease or condition in which modulation of microsomal prostaglandin E synthase-1 activity is beneficial which comprises administering to a patient in need thereof a therapeutically effective amount of a compound of formula (I) or a pharmaceutically acceptable salt thereof as hereinbefore defined.
- The invention still further provides a method of treating, or reducing the risk of, an inflammatory disease or condition which comprises administering to a patient in need thereof a therapeutically effective amount of a compound of formula (I) or a pharmaceutically acceptable salt thereof as hereinbefore defined.
- The invention still further provides a method of treating, or reducing the risk of, osteoarthritis, rheumatoid arthritis, acute or chronic pain, neuropathic pain, apneal/SID, wound healing, cancer, stroke, atherosclerosis or Alzheimer's disease which comprises administering to a patient in need thereof a therapeutically effective amount of a compound of formula (I) or a pharmaceutically acceptable salt thereof as hereinbefore defined. The invention still further provides a method of treating, or reducing the risk of, osteoarthritis, rheumatoid arthritis or acute or chronic pain which comprises administering to a patient in need thereof a therapeutically effective amount of a compound of formula (I) or a pharmaceutically acceptable salt thereof as hereinbefore defined.
- For the above-mentioned therapeutic uses the dosage administered will, of course, vary with the compound employed, the mode of administration, the treatment desired and the disorder indicated. The daily dosage of the compound of the invention may be in the range from 0.05 mg/kg to 100 mg/kg.
- The compounds of formula (I) and pharmaceutically acceptable salts thereof may be used on their own but will generally be administered in the form of a pharmaceutical composition in which the formula (I) compound/salt (active ingredient) is in association with a pharmaceutically acceptable adjuvant, diluent or carrier. Conventional procedures for the selection and preparation of suitable pharmaceutical formulations are described in, for example, “Pharmaceuticals—The Science of Dosage Form Designs”, M. E. Aulton, Churchill Livingstone, 1988.
- Depending on the mode of administration, the pharmaceutical composition will preferably comprise from 0.05 to 99%w (per cent by weight), more preferably from 0.05 to 80%w, still more preferably from 0.10 to 70%w, and even more preferably from 0.10 to 50%w, of active ingredient, all percentages by weight being based on total composition.
- The present invention also provides a pharmaceutical composition comprising a compound of formula (I) or a pharmaceutically acceptable salt thereof as hereinbefore defined, in association with a pharmaceutically acceptable adjuvant, diluent or carrier.
- The invention further provides a process for the preparation of a pharmaceutical composition of the invention which comprises mixing a compound of formula (I) or a pharmaceutically acceptable salt thereof as hereinbefore defined with a pharmaceutically acceptable adjuvant, diluent or carrier.
- The pharmaceutical compositions may be administered topically (e.g., to the skin) in the form, e.g., of creams, solutions or suspensions; or systemically, e.g., by oral administration in the form of tablets, capsules, syrups, powders or granules; or by parenteral administration in the form of solutions or suspensions; or by subcutaneous administration; or by rectal administration in the form of suppositories; or transdermally.
- For oral administration the compound of the invention may be admixed with an adjuvant or a carrier, for example, lactose, saccharose, sorbitol, mannitol; a starch, for example, potato starch, corn starch or amylopectin; a cellulose derivative; a binder, for example, gelatine or polyvinylpyrrolidone; and/or a lubricant, for example, magnesium stearate, calcium stearate, polyethylene glycol, a wax, paraffin, and the like, and then compressed into tablets. If coated tablets are required, the cores, prepared as described above, may be coated with a concentrated sugar solution which may contain, for example, gum arabic, gelatine, talcum and titanium dioxide. Alternatively, the tablet may be coated with a suitable polymer dissolved in a readily volatile organic solvent.
- For the preparation of soft gelatine capsules, the compound of the invention may be admixed with, for example, a vegetable oil or polyethylene glycol. Hard gelatine capsules may contain granules of the compound using either the above-mentioned excipients for tablets. Also liquid or semisolid formulations of the compound of the invention may be filled into hard gelatine capsules.
- Liquid preparations for oral application may be in the form of syrups or suspensions, for example, solutions containing the compound of the invention, the balance being sugar and a mixture of ethanol, water, glycerol and propylene glycol. Optionally such liquid preparations may contain colouring agents, flavouring agents, saccharine and/or carboxymethylcellulose as a thickening agent or other excipients known to those skilled in art.
- The compounds of the invention may also be administered in conjunction with other compounds used for the treatment of the above conditions.
- Thus, the invention further relates to combination therapies wherein a compound of formula (I) or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition or formulation comprising a compound of formula (I) is administered concurrently, simultaneously, sequentially or separately with another pharmaceutically active compound or compounds selected from the following:
- (i) neuropathic pain therapies including for example gabapentin, lidoderm, pregablin and equivalents and pharmaceutically active isomer(s) and metabolite(s) thereof.
- (ii) nociceptive pain therapies such as celecoxib, etoricoxib, lumiracoxib, rofecoxib, valdecoxib, diclofenac, loxoprofen, naproxen, paracetamol and equivalents and pharmaceutically active isomer(s) and metabolite(s) thereof.
- (iii) migraine therapies including for example almotriptan, amantadine, bromocriptine, butalbital, cabergoline, dichloralphenazone, eletriptan, frovatriptan, lisuride, naratriptan, pergolide, pramipexole, rizatriptan, ropinirole, sumatriptan, zolmitriptan, zomitriptan, and equivalents and pharmaceutically active isomer(s) and metabolite(s) thereof.
- Such combination products employ the compounds of this invention within the dosage range described herein and the other pharmaceutically active compound or compounds within approved dosage ranges and/or the dosage described in their respective publication reference(s).
- 1. A compound of formula (I) or a pharmaceutically acceptable salt thereof
- wherein:
- A is selected from phenyl or a 5- or 6-membered heteroaryl or a 5- or 6-membered heterocyclenyl moiety; said phenyl or 5- or 6-membered heteroaryl or 5- or 6-membered heterocyclenyl moiety in group A being optionally fused to a phenyl, a 5- or 6-membered heteroaryl, C5-6carbocyclyl or C5-6heterocyclyl ring;
- each R1 is independently selected from halogen, nitro, SF5, CN, NR5R6, OH, CHO, CO2C1-4alkyl, CONR5R6, C1-4alkyl, C2-4alkenyl, C2-4alkynyl, C1-4alkoxy, G3, OG3 or OCH2G3; said C1-4alkyl, C2-4alkenyl, C2-4alkynyl or C1-4alkoxy being optionally substituted by one or more substituents selected from OH, NR5R6, C1-4alkyl or F;
- m represents an integer 0,1, 2, 3 or 4;
- each R3 is independently selected from hydrogen, CN and C1-4alkyl; said C1-4alkyl being optionally substituted with OH, CN, C1-4alkoxy, NR7R8, or one or more F atoms;
- L1 represents a direct bond, C1-4alkylene, C2-4alkenylene or C2-4alkynylene;
- L2 represents a direct bond, —O—, —OCH2—, C1-2alkylene, —C≡C— or —NHCONH—;
- G1 represents phenyl, 5- or 6-membered heteroaryl, C3-10carbocyclyl or C5-8heterocyclyl;
- G2 represents H, C1-6alkyl, phenyl, 5- or 6-membered heteroaryl, C3-10carbocyclyl or C5-8heterocyclyl; said C1-6alkyl being optionally further substituted by one or more groups selected from OH, C1-6alkoxy and halogen;
- the phenyl, heteroaryl, carbocyclyl or heterocyclyl moieties in G1 and G2 being optionally fused to one or two further rings independently selected from phenyl, a 5- or 6-membered heteroaryl, C5-6carbocyclyl or C5-6heterocyclyl ring;
- any phenyl, heteroaryl, carbocyclyl or heterocyclyl moieties in G1 and G2 being optionally substituted by one or more substituents independently selected from halogen, OH, CN, NO2, CO2R9, C1-6alkyl, C1-6alkoxy, C1-4thioalkoxy, SO2NR10R11, NR12R13, —NHCOC(OH)(CH3)CF3 or —CH2OCH2CF2CHF2; said C1-6alkyl or C1-6alkoxy being optionally substituted by OH or by one or more F atoms;
- R9 represents C1-6alkyl;
- G3 represents phenyl or 5- or 6-membered heteroaryl; and
- each R4, R5, R6, R7, R8, R10, R11, R12 and R13 is independently selected from H or C1-4alkyl;
- with the proviso that the following compounds are excluded:
- 3-[(phenylsulfonyl)amino]pyridine-2-sulfonamide;
- 2-amino-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 4-tert-butyl-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 3-amino-4-hydroxy-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 4-bromo-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 4-fluoro-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 4-methyl-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 4-nitro-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-nitro-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 4-amino-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[(phenylsulfonyl) amino]-4-(trifluoromethyl)benzenesulfonamide;
- 2-{[(4-methylphenyl)sulfonyl]amino }-4-(trifluoromethyl)benzenesulfonamide; N-(2-sulfamoylphenyl)quinoline-8-sulfonamide;
- 4-tert-butyl-N-(2-(N-ethylsulfamoyl)phenyl)-2,6-dimethylbenzenesulfonamide;
- 2,5-dibromo-N-(2-(N-ethylsulfamoyl)phenyl)benzenesulfonamide;
- methyl 4-(5-fluoro-3-methylbenzo[b]thiophene-2-sulfonamido)-3-sulfamoylbenzoate;
- 2,3,4,5,6-pentafluoro-N-(2-sulfamoylphenyl)benzenesulfonamide;
- N-(2-(N,N-dimethylsulfamoyl)phenyl)-5,7-dimethyl-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide;
- N-(2-(N-tert-butylsulfamoyl)phenyl)-5,7-dimethyl-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide;
- N-(2-(N-methoxy-N-methylsulfamoyl)phenyl)-5,7-dimethyl-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide;
- 5,7-dimethyl-N-(2-sulfamoylphenyl)-[1,2,4]triazolo[1,5-c]pyrimidine-2-sulfonamide;
- 6-methyl-N-(2-sulfamoylphenyl)thiazolo[3,2-b][1,2,4]triazole-2-sulfonamide;
- 6-chloro-N-(2-(N,N-dimethylsulfamoyl)phenyl)-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide;
- 6-chloro-N-(2-(N-ethyl-N-methylsulfamoyl)phenyl)-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide;
- N-(2-(N,N-dimethylsulfamoyl)phenyl)-5-methyl-7,8-dihydro-6H-cyclopenta[e][1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide;
- N-(2-(N,N-dimethylsulfamoyl)-5-(trifluoromethyl)phenyl)-5,7-dimethyl-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide;
- N-(2-(N-ethyl-N-methylsulfamoyl)phenyl)-5,7-dimethyl-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide; and
- N-(2-(N,N-dimethylsulfamoyl)phenyl)-5,7-dimethyl-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide.
- 2. A compound according to embodiment 1 wherein each R3 represents hydrogen.
- 3. A compound according to embodiment 1 or embodiment 2 wherein A represents phenyl, pyridyl, thienyl or 2,3-dihydro-indenyl.
- 4. A compound according to any one of embodiments 1-3 wherein m represents an integer 0, 1 or 2.
- 5. A compound according to any one of embodiments 1-4 wherein L1 represents a direct bond, C1-3alkylene or C2-3alkenylene.
- 6. A compound according to any one of embodiments 1-5 wherein L2 represents a direct bond, —O—, —OCH2—, —NHCONH— or —C≡C—.
- 7. A compound according to any one of embodiments 1-6 wherein G1 represents phenyl or 5- or 6-membered heteroaryl, said phenyl or 5- or 6-membered heteroaryl being optionally fused to one or two further rings independently selected from phenyl, C5-6carbocyclyl or C5-6heterocyclyl ring.
- 8. A compound according to embodiment 7 wherein G1 represents phenyl, naphtyl, pyridyl, thienyl, 2,3-dihydro-indenyl tetrahydronaphtyl, 2,3-dihydro-1,4-benzodioxinyl or S,S,-dioxodibenzothienyl.
- 9. A compound according to any one of embodiments 1-8 wherein G2 represents H, C1-6alkyl, phenyl, 5- or 6-membered heteroaryl, C3-10carbocyclyl or C5-8heterocyclyl; said phenyl or 5- or 6-membered heteroaryl being optionally fused to one further ring independently selected from phenyl, a 5- or 6-membered heteroaryl, C5-6carbocyclyl and C5-6heterocyclyl ring;
- said C1-6alkyl being optionally further substituted by one or more groups selected from OH, C1-6alkoxy and halogen.
- 10. A compound according to any embodiment 9 wherein G2 represents hydrogen, methyl, ethyl, tert-butyl, iso-propyl, phenyl, pyridyl, pyrimidyl, cyclopentyl, cyclopentenyl, cyclohexyl, furanyl, tetrahydrofuranyl, thienyl, thiazolyl, pyrazolyl, bensofuranyl, 2,3-dihydro-bensofuranyl, indolyl, tetrahydronaphtyl or dibenzofuranyl.
- 11. A compound according to any one of embodiments 1-10 wherein R5 and R6 is hydrogen.
- 12. A compound according to any one of embodiments 1-11 wherein R10 is hydrogen.
- 13. A compound according to any one of embodiments 1-12 wherein R11, R12 and R13 is methyl.
- 14. A compound according to any one of embodiments 1-13 wherein R1 is independently selected from halogen, CN, OH, CONR5R6, NR5R6, C1-4alkyl, C2-4alkynyl, C1-4alkoxy, G3, OG3 and OCH2G3; said C1-4alkyl or C2-4alkynyl being optionally substituted by OH.
- 15. A compound according to any one of embodiments 1-14 wherein G3 is phenyl, pyridyl or 2-methyl-1,3,4-oxadiazole.
- 16. A compound according to any one of embodiments 1-15 wherein any phenyl, heteroaryl, carbocyclyl or heterocyclyl moieties in G1 and G2 2 are optionally substituted by one or more substituents independently selected from halogen, CN, OH, C1-4alkyl, C1thioalkoxy, NR12R13, —NHCOC(OH)(CH3)CF3, SO2NR10R11, and C1alkoxy; said C1-4alkyl or C1alkoxy being optionally substituted by OH or by one or more F atoms.
- 17. A compound according to embodiment 1 wherein A is phenyl, pyridyl, thienyl or 2,3-dihydro-indenyl;
- R1 is independently selected from halogen, CN, OH, CONR5R6, NR5R6, C1-4alkyl, C2-4alkynyl, C1-4alkoxy, G3, OG3 and OCH2G3; said C1-4alkyl or C2-4alkynyl being optionally substituted by one or more substituents selected from OH and C1-4alkyl;
- m represents an integer 0,1 or 2;
- G3 is phenyl, pyridyl or 2-methyl-1,3,4-oxadiazole;
- R3, R5, R6 and R10 represents hydrogen;
- L1 represents a direct bond, C1-3alkylene or C2-3alkenylene;
- L2 represents a direct bond, —O—, —OCH2—, —NHCONH— or —C≡C—;
- G1 represents phenyl or 5- or 6-membered heteroaryl; said phenyl or 5- or 6-membered heteroaryl being optionally fused to one or two further rings independently selected from phenyl, C5-6carbocyclyl or C5-6heterocyclyl ring;
- G2 represents H, C1-6alkyl, phenyl, 5- or 6-membered heteroaryl, C3-10carbocyclyl or C5-8 heterocyclyl; said phenyl or 5- or 6-membered heteroaryl being optionally fused to one further ring independently selected from phenyl, a 5- or 6-membered heteroaryl, C5-6carbocyclyl or C5-6heterocyclyl ring; said C1-6alkyl being optionally further substituted by one or more groups selected from OH, C1-6alkoxy and halogen;
- any phenyl, heteroaryl, carbocyclyl or heterocyclyl moieties in G1 and G2 being optionally substituted by one or more substituents independently selected from halogen, CN, OH, C1-4alkyl, C1thioalkoxy, NR12R13, —NHCOC(OH)(CH3)CF3, SO2NR10R11, and C1alkoxy;
- said C1-4alkyl or C1alkoxy being optionally substituted by OH or by one or more F atoms; and R11, R12 and R13 are methyl.
- 18. A compound according to embodiment 1 wherein A is phenyl, pyridyl, thienyl or 2,3-dihydro-indenyl;
- R1 is independently selected from chloro, fluoro, bromo, CN, OH, CONR5R6, methyl, 1-butynyl, methoxy, G3, OG3 or OCH2G3; said methyl and 1-butynyl being optionally substituted by OH or methyl;
- m represents an integer 0, 1 or 2;
- G3 is phenyl, pyridyl or 2-methyl-1,3,4-oxadiazole;
- R3, R5, R6 and R10 represents hydrogen;
- L2 represents a direct bond, ethylene, 1-methylethylene, 2-methylethylene, ethenylene, —C(CH3)CH—, —CHC(CH3)— or —CH2C(CH2)—;
- L2 represents a direct bond, —O—, —OCH2—, —NHCONH— or —C≡C—;
- G1 represents phenyl, naphtyl, pyridyl, thienyl, 2,3-dihydro-indenyl tetrahydronaphtyl, 2,3-dihydro-1,4-benzodioxinyl or S,S,-dioxodibenzothienyl;
- G2 represents hydrogen, methyl, ethyl, tert-butyl, iso-propyl phenyl, pyridyl, pyrimidyl, cyclopentyl, cyclopentenyl, cyclohexyl, furanyl, tetrahydrofuranyl, thienyl, thiazolyl, pyrazolyl, bensofuranyl, 2,3-dihydro-bensofuranyl, indolyl, tetrahydronaphtyl or dibenzofuranyl;
- any methyl, ethyl, tert-butyl, iso-propyl phenyl, pyridyl, pyrimidyl, cyclopentyl, cyclopentenyl, cyclohexyl, furanyl, tetrahydrofuranyl, thienyl, thiazolyl, pyrazolyl, bensofuranyl, 2,3-dihydro-bensofuranyl, indolyl, tetrahydronaphtyl or dibenzofuranyl in G1 and G2 being optionally substituted by one or more substituents independently selected from bromo, chloro, fluoro, CN, OH, methyl, tert-butyl, methylthio, NR12R13, —
- NHCOC(OH)(CH3)CF3, SO2NR10R11, and methoxy; said methyl, tert-butyl or methoxy being optionally substituted by OH or by one or more F atoms; and
- R11, R12 and R13 are methyl.
- 19. A compound according to any one of embodiments 1-18 being an entity selected from:
- 2-[2-(4-Benzofuran-2-ylphenyl)ethylsulfonylamino]benzenesulfonamide;
- 2-[2-[4-(2,3-Dihydrobenzofuran-2-yl)phenyl]ethylsulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-(4-Benzofuran-2-ylphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-(4-Benzofuran-2-ylphenyl)ethenyl]sulfonylamino]-5-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(4-Benzofuran-2-ylphenyl)ethenyl]sulfonylamino]-4-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(4-Benzofuran-2-ylphenyl)ethenyl]sulfonylamino]-6-fluoro-benzenesulfonamide;
- 2-[1-(4-Chlorophenyl)propan-2-ylsulfonylamino]benzenesulfonamide;
- 2-(1-Phenylpropan-2-ylsulfonylamino)benzenesulfonamide;
- 2-[[(E)-1-(4-Chlorophenyl)prop-1-en-2-yl]sulfonylamino]benzenesulfonamide;
- 2-[1-(4-Chlorophenyl)propan-2-ylsulfonylamino]-4-fluoro-benzenesulfonamide;
- 2-[[(E)-1-(4-chlorophenyl)prop-1-en-2-yl]sulfonylamino]-4-fluoro-benzenesulfonamide;
- 2-[[(E)-1-(4-Chlorophenyl)prop-1-en-2-yl]sulfonylamino]-5-fluoro-benzenesulfonamide;
- 2-[[(E)-1-(4-Chlorophenyl)prop-1-en-2-yl]sulfonylamino]-6-fluoro-benzenesulfonamide;
- 2-[[(E)-1-(4-Chlorophenyl)prop-1-en-2-yl]sulfonylamino]-3-fluoro-benzenesulfonamide;
- 2-[[(E)-1-(4-Chlorophenyl)prop-1-en-2-yl]sulfonylamino]-5-methyl-benzenesulfonamide;
- 2-[2-[4-(2-Furyl)phenyl]ethylsulfonylamino]benzenesulfonamide;
- 2-[2-[4-(Oxolan-2-yl)phenyl]ethylsulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-[4-(2-Furyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[2-[4-(Difluoromethoxy)phenyl]ethylsulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-[4-(Difluoromethoxy)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
- 2-[2-(4-Chlorophenyl)ethylsulfonylamino]-5-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(4-Chlorophenyl)ethenyl]sulfonylamino]-5-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(3,4-Dichlorophenyl)ethenyl]sulfonylamino]-4-fluoro-benzenesulfonamide; 2-[[(E)-2-(3,4-Dichlorophenyl)ethenyl]sulfonylamino]-5-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(3,4-Dichlorophenyl)ethenyl]sulfonylamino]-6-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(5-Bromo-2-methoxy-phenyl)ethenyl]sulfonylamino]-4-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(5-Bromo-2-methoxy-phenyl)ethenyl]sulfonylamino]-5-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(5-Bromo-2-methoxy-phenyl)ethenyl]sulfonylamino]-6-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(5-Bromo-2-methoxy-phenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-(4-Cyanophenyl)ethenyl]sulfonylamino]-4-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(4-Cyanophenyl)ethenyl]sulfonylamino]-5-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(4-Cyanophenyl)ethenyl]sulfonylamino]-6-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(4-Cyanophenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 4-Fluoro-2-[[(E)-2-[4-(2-furyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide;
- 5-Fluoro-2-[[(E)-2-[4-(2-furyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide;
- 2-Fluoro-6-[[(E)-2-[4-(2-furyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide;
- 4-Fluoro-2-[[(E)-2-(4-methylsulfanylphenyl)ethenyl]sulfonylamino]-benzenesulfonamide;
- 5-Fluoro-2-[[(E)-2-(4-methylsulfanylphenyl)ethenyl]sulfonylamino]-benzenesulfonamide;
- 2-Fluoro-6-[[(E)-2-(4-methylsulfanylphenyl)ethenyl]sulfonylamino]-benzenesulfonamide;
- 2-[[(E)-2-(4-Methylsulfanylphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-[4-(Difluoromethoxy)phenyl]ethenyl]sulfonylamino]-4-fluoro-benzenesulfonamide;
- 2-[[(E)-2-[4-(Difluoromethoxy)phenyl]ethenyl]sulfonylamino]-5-fluoro-benzenesulfonamide;
- 2-[[(E)-2-[4-(Difluoromethoxy)phenyl]ethenyl]sulfonylamino]-6-fluoro-benzenesulfonamide;
- 2-[2-(4-Chlorophenyl)ethylsulfonylamino]-5-(hydroxymethyl)-benzenesulfonamide;
- 2-[2-(4-Chloro-phenyl)-ethanesulfonylamino]-5-methylbenzenesulfonamide;
- 2-[2-(4-Chlorophenyl)ethylsulfonylamino]-5-(pyridin-2-ylmethoxy)benzenesulfonamide;
- 2-[2-(4-Chlorophenyl)ethylsulfonylamino]-5-[(5-methyl-1,3,4-oxadiazol-2-yl)methoxy]benzenesulfonamide;
- 2-[2-(2-Chloro-phenyl)-ethenesulfonylamino]-benzenesulfonamide;
- 2-[2-(4-Chloro-2-methoxy-phenyl)-ethenesulfonylamino]-benzenesulfonamide;
- 2-Fluoro-6-[[(E)-2-(4-phenylphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-(3-Chlorophenyl)ethenyl]sulfonylamino]-5-fluoro-benzenesulfonamide;
- 5-Fluoro-2-[[(E)-2-(4-phenylphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 4-Fluoro-2-[[(E)-2-(4-phenylphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 5-Fluoro-2-[[(E)-2-[3-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
- 5-Fluoro-2-[[(E)-2-(2-methoxyphenyl)ethenyl]sulfonylamino]-benzenesulfonamide;
- 2-[[(E)-2-(3-Chlorophenyl)ethenyl]sulfonylamino]-6-fluoro-benzenesulfonamide;
- 2-Fluoro-6-[[(E)-2-(2-methoxyphenyl)ethenyl]sulfonylamino]-benzenesulfonamide;
- 4-Fluoro-2-[[(E)-2-(2-methoxyphenyl)ethenyl]sulfonylamino]-benzenesulfonamide;
- 3-Fluoro-2-[[(E)-2-(4-phenylphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 3-Fluoro-2-[[(E)-2-(2-methoxyphenyl)ethenyl]sulfonylamino]-benzenesulfonamide;
- 4-Fluoro-2-[[(E)-2-[3-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
- 2-Fluoro-6-[[(E)-2-(2-fluorophenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-(3-Chlorophenyl)ethenyl]sulfonylamino]-5-methyl-benzenesulfonamide;
- 2-[[(E)-2-(3-Chlorophenyl)ethenyl]sulfonylamino]-3-fluoro-benzenesulfonamide;
- 3-Fluoro-2-[[(E)-2-[3-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
- 2-Fluoro-6-[[(E)-2-[3-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
- 4-Fluoro-2-[[(E)-2-(2-fluorophenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 5-Methyl-2-[[(E)-2-[3-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
- 2-[2-(4-Cyclopentylphenyl)ethylsulfonylamino]benzenesulfonamide;
- 2-[2-(4-Tert-butylphenyl)ethylsulfonylamino]benzenesulfonamide;
- 4-Fluoro-2-[2-(4-tert-butylphenyl)ethylsulfonylamino]benzenesulfonamide;
- 2-[2-(4-Chlorophenyl)ethylsulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-(4-Chlorophenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 2-(Phenethylsulfonylamino)benzenesulfonamide;
- 2-[[(E)-2-Phenylethenyl]sulfonylamino]benzenesulfonamide;
- 2-[2-(2-Chlorophenyl)ethylsulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-(4-Methoxyphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-(4-Chlorophenyl)ethenyl]sulfonylamino]-4-fluoro-benzenesulfonamide;
- 2-(2-Phenylpropylsulfonylamino)benzenesulfonamide;
- 2-[[(E)-2-(4-Chlorophenyl)ethenyl]sulfonylamino]-6-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(2,6-Dichlorophenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-(4-Methylphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-(2-Methoxyphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-Naphthalen-2-ylethenyl]sulfonylamino]benzenesulfonamide;
- 2-[2-(4-Chlorophenyl)ethylsulfonylamino]-4-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(4-Chlorophenyl)prop-1-enyl]sulfonylamino]benzenesulfonamide;
- 2-[2-(4-Chlorophenyl)prop-2-enylsulfonylamino]benzenesulfonamide;
- 2-Fluoro-6-[[(E)-2-naphthalen-2-ylethenyl]sulfonylamino]benzenesulfonamide;
- 2-Fluoro-6-[[(E)-2-[2-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
- 2-Fluoro-6-[[(E)-2-[4-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
- 2-[[(E)-2-(4-Bromophenyl)ethenyl]sulfonylamino]-6-fluoro-benzenesulfonamide;
- 4-Fluoro-2-[[(E)-2-naphthalen-2-ylethenyl]sulfonylamino]benzenesulfonamide;
- 4-Fluoro-2-[[(E)-2-[2-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
- 4-Fluoro-2-[[(E)-2-[4-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
- 2-[[(E)-2-(4-Bromophenyl)ethenyl]sulfonylamino]-4-fluoro-benzenesulfonamide;
- 5-Methyl-2-[[(E)-2-naphthalen-2-ylethenyl]sulfonylamino]benzenesulfonamide;
- 5-Methyl-2-[[(E)-2-[4-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
- 2-[[(E)-2-(4-Bromophenyl)ethenyl]sulfonylamino]-5-methyl-benzenesulfonamide;
- 5-Fluoro-2-[[(E)-2-naphthalen-2-ylethenyl]sulfonylamino]benzenesulfonamide;
- 5-Fluoro-2-[[(E)-2-[2-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
- 5-Fluoro-2-[[(E)-2-[4-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
- 2-[[(E)-2-(4-Bromophenyl)ethenyl]sulfonylamino]-5-fluoro-benzenesulfonamide;
- 2-[[(E)-2-(2,6-Difluorophenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-[2-(Trifluoromethyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-[4-(Trifluoromethyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[[(E)-2-(4-Bromophenyl)ethenyl]sulfonylamino]benzenesulfonamide;
- 2-[(E)-2-(4-Bromophenyl) ethylsulfonylamino]benzenesulfonamide;
- 2-[2-(4-Chlorophenyl)propylsulfonylamino]benzenesulfonamide;
- 2-[2-(2-Methoxyphenyl)ethylsulfonylamino]benzenesulfonamide;
- 2-(2-Naphthalen-2-ylethylsulfonylamino)benzenesulfonamide;
- 4-Fluoro-2-(2-naphthalen-2-ylethylsulfonylamino)benzenesulfonamide;
- 2-[(4-Chlorophenyl)methylsulfonylamino]benzenesulfonamide;
- 2-[[3-(2,3-Dichlorophenyl)phenyl]sulfonylamino]benzenesulfonamide;
- 2-[[3-(3,5-difluorophenyl)phenyl]sulfonylamino]benzenesulfonamide;
- 3-(5-Chloro-2-methoxy-phenyl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[[3-(3-Cyanophenyl)phenyl]sulfonylamino]benzenesulfonamide;
- 2-[[3-(4-Cyanophenyl)phenyl]sulfonylamino]benzenesulfonamide;
- 3-(2,4-Dimethoxypyrimidin-5-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[[3-(3-Furyl)phenyl]sulfonylamino]benzenesulfonamide;
- 2-[[3-(1H-Indol-5-yl)phenyl]sulfonylamino]benzenesulfonamide;
- 2-[[3-[3-(Trifluoromethoxy)phenyl]phenyl]sulfonylamino]benzenesulfonamide;
- N-(2-Sulfamoylphenyl)-2,3-dihydro-1H-indene-5-sulfonamide;
- 7-Methyl-N-(2-sulfamoylphenyl)-10-oxa-7-azabicyclo[4.4.0]deca-1,3,5-triene-3-sulfonamide;
- 2-(3,4-Dichlorophenyl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[[4-[3-Chloro-5-(trifluoromethyl)pyridin-2-yl]oxyphenyl]sulfonylamino]benzenesulfonamide;
- N-(2-Sulfamoylphenyl)tetralin-2-sulfonamide;
- 4-Phenylmethoxy-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[(4-Cyclohexylphenyl)sulfonylamino]benzenesulfonamide;
- 3-Phenyl-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 5-(1-Methyl-5-trifluoromethyl-1H-pyrazol-3-yl)-thiophene-2-sulfonic acid (2-sulfamoyl-phenyl)-amide;
- N-(2-Sulfamoylphenyl)-4-(trifluoromethyl)benzenesulfonamide;
- 2-[[3-(2,5-Dichlorophenyl)phenyl]sulfonylamino]benzenesulfonamide;
- 3-Dibenzofuran-4-yl-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[[3-[4-(Trifluoromethyl)phenyl]phenyl]sulfonylamino]benzenesulfonamide;
- 3-(3-Methoxyphenyl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 3-Benzofuran-2-yl-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[[3-(2,3-Dihydrobenzofuran-5-yl)phenyl]sulfonylamino]benzenesulfonamide;
- 3-(6-Methoxypyridin-3-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[[3-(2,4-Dichlorophenyl)phenyl]sulfonylamino]benzenesulfonamide;
- 3-(1-Methylindol-2-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[[3-[3-(Trifluoromethyl)phenyl]phenyl]sulfonylamino]benzenesulfonamide;
- 4-Benzofuran-2-yl-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[[3-(2,4-Difluorophenyl)phenyl]sulfonylamino]benzenesulfonamide;
- 5-Bromo-2-[[3-(3,4-dichlorophenyl)phenyl]sulfonylamino]benzenesulfonamide;
- 2-[[3-(3,4-Dichlorophenyl)phenyl]sulfonylamino]-5-phenyl-benzenesulfonamide;
- 2,3-Difluoro-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2,3-Difluoro-N-(4-fluoro-2-sulfamoyl-phenyl)benzenesulfonamide;
- 3-Chloro-2-fluoro-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2,3-Dichloro-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[[3-(3,4-Dichlorophenyl)phenyl]sulfonylamino]-5-methyl-benzenesulfonamide;
- 4-[[3-(3,4-Dichlorophenyl)phenyl]sulfonylamino]-3-sulfamoyl-benzamide;
- 5-Methyl-2-[[3-[3-(trifluoromethoxy)phenyl]phenyl]sulfonylamino]-benzenesulfonamide;
- 2-(2,3-Dichloro-benzenesulfonylamino)-5-hydroxymethyl-benzenesulfonamide;
- 2-(2,3-Dichloro-benzenesulfonylamino)-5-methyl-benzenesulfonamide;
- 2-[[3-(3,4-Dichlorophenyl)phenyl]sulfonylamino]-5-phenoxy-benzenesulfonamide;
- 5-Phenoxy-2-[[3-[3-(trifluoromethoxy)phenyl]phenyl]sulfonylamino]-benzenesulfonamide;
- 2-(2,3-Dichloro-benzenesulfonylamino)-5-hydroxy-benzenesulfonamide;
- (2R)—N-[2-Chloro-4-[(2-sulfamoylphenyl)sulfamoyl]phenyl]-3,3,3-trifluoro-2-hydroxy-2-methyl-propanamide;
- 5-Chloro-N-(2-sulfamoylphenyl)thiophene-2-sulfonamide;
- 4,5-Dichloro-N-(2-sulfamoylphenyl)thiophene-2-sulfonamide;
- 3,4-Dichloro-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 3-(6-Methoxypyridin-2-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[[4-[(2-Chloro 1,3-thiazol-5-yl)methoxy]phenyl]sulfonylamino]-benzenesulfonamide;
- 3-(5-Fluoro-6-methoxy-pyridin-3-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 3-(2-Methoxypyrimidin-5-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 3-(4-Methylpyridin-3-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 3-(2-Methoxypyridin-3-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[[3-(5-Mhloropyridin-3-yl)phenyl]sulfonylamino]benzenesulfonamide;
- 3-(5-Chloro-6-methoxy-pyridin-3-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 3-(6-Dimethylaminopyridin-3-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-[[3-[3-(Hydroxymethyl)phenyl]phenyl]sulfonylamino]benzenesulfonamide;
- 2-[[3-[4-(Hydroxymethyl)phenyl]phenyl]sulfonylamino]benzenesulfonamide;
- 2-[[4-(3,4-Dichlorophenyl)phenyl]sulfonylamino]benzenesulfonamide;
- 2-[[3-(3,4-Dichlorophenyl)phenyl]sulfonylamino]benzenesulfonamide;
- 2-[[4-(4-Chlorophenoxy)phenyl]sulfonylamino]benzenesulfonamide;
- 3-(4-Chlorophenyl)-1-[3-methyl-4-[(2-sulfamoylphenyl)sulfamoyl]phenyl]urea;
- N-Methyl-N′-(2-sulfamoylphenyl)dibenzo[b,d]thiophene-2,8-disulfonamide 5,5-dioxide;
- 2,3-Dichloro-N-(4,5-difluoro-2-sulfamoylphenyl)benzenesulfonamide;
- (E)-2-(2-(3,4-Dichlorophenyl)vinylsulfonamido)-5-(3-hydroxy-3-methylbut-1-ynyl)benzenesulfonamide;
- 4-Cyano-2-(2-(3,4-dichlorophenyl)ethylsulfonamido)benzenesulfonamide;
- 2-(2-(4-Chloro-2-methoxyphenyl)ethylsulfonamido)-4-cyanobenzenesulfonamide;
- 2-(2-(4-Chloro-2-methoxyphenyl)ethylsulfonamido)-5-cyanobenzenesulfonamide;
- 2-(2-(4-(3,3-dimethylbut-1-ynyl)phenyl)ethylsulfonamido)benzenesulfonamide;
- 2-(2-(4-(3-Hydroxy-3-methylbut-1-ynyl)phenyl)ethylsulfonamido)benzenesulfonamide;
- 3-(3,3-Dimethylbut-1-ynyl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-(2-(4-Chloro-2-methoxyphenyl)ethylsulfonamido)benzenesulfonamide;
- 2-(2-(6-(3,3-Dimethylbut-1-ynyl)pyridin-3-yl)ethylsulfonamido)benzenesulfonamide;
- 2-(2-(6-(cyclohexylethynyl)pyridin-3-yl)ethylsulfonamido)benzenesulfonamide;
- 2-(2-(4-Bromophenyl)ethylsulfonamido)-5-methylbenzenesulfonamide;
- (E)-2-(2-(4-Bromophenyl)vinylsulfonamido)-5-methylbenzenesulfonamide;
- 3-(2-Methylthiazol-4-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
- (E)-2-(2-(3-(Trifluoromethyl)phenyl)vinylsulfonamido)benzenesulfonamide;
- (E)-2-(2-(2-Fluorophenyl)vinylsulfonamido)benzenesulfonamide;
- 5-Chloro-2-(2-(4-chloro-2-methoxyphenyl)ethylsulfonamido)benzenesulfonamide;
- 2,3-Dichloro-N-(4-chloro-2-sulfamoylphenyl)benzenesulfonamide;
- 2,3-Dichloro-N-(3,5-difluoro-2-sulfamoylphenyl)benzenesulfonamide;
- 2-(2-(3,4-Dichlorophenyl)ethylsulfonamido)-4,6-difluorobenzenesulfonamide;
- 2-(2-(4-Chloro-2-methoxyphenyl)ethylsulfonamido)-4,6-difluorobenzenesulfonamide;
- 5-Cyano-2-(2-(3,4-dichlorophenyl)ethylsulfonamido)benzenesulfonamide;
- (E)-2-(2-(3,4-Dichlorophenyl)vinylsulfonamido)-4-methylbenzenesulfonamide;
- 2-(2-(4-Chloro-2-methoxyphenyl)ethylsulfonamido)-4-methylbenzenesulfonamide;
- 5-Chloro-2-(2-(3,4-dichlorophenyl)ethylsulfonamido)benzenesulfonamide;
- 4-(3-(5-Chloro-6-methoxypyridin-3-yl)phenylsulfonamido)pyridine-3-sulfonamide;
- 4-(2-(4-(Benzofuran-2-yl)phenyl)ethylsulfonamido)pyridine-3-sulfonamide;
- 2-(2-(4-Chloro-2-methoxyphenyl)ethylsulfonamido)-5-methylbenzenesulfonamide;
- 4-(2-(4-Chloro-2-methoxyphenyl)ethylsulfonamido)pyridine-3-sulfonamide;
- (E)-2-(2-(4-Cyclopentenylphenyl)vinylsulfonamido)benzenesulfonamide;
- (E)-2-(2-(4-(3,3-Dimethylbut-1-ynyl)phenyl)vinylsulfonamido)benzenesulfonamide;
- (E)-2-(2-(4-(3-Hydroxy-3-methylbut-1-ynyl)phenyl)vinylsulfonamido)-benzenesulfonamide;
- 2-{2-[4-(3,3-Dimethyl-but-1-ynyl)-phenyl]-ethanesulfonylamino}-5-methyl-benzenesulfonamide;
- 2-{2-[4-(3,3-Dimethyl-but-1-ynyl)-phenyl]-ethanesulfonylamino}-5-hydroxymethyl-benzenesulfonamide;
- 2-[2-(4-Cyclopentylethynyl-phenyl)-ethanesulfonylamino]-5-hydroxymethyl-benzenesulfonamide;
- 2-[2-(4-Chloro-2-methoxy-phenyl)-ethanesulfonylamino]-5-hydroxymethyl-benzenesulfonamide;
- 3-{2-[4-(3,3-Dimethyl-but-1-ynyl)-phenyl]-ethanesulfonylamino}-thiophene-2-sulfonic acid amide;
- 3-[3-(5-Chloro-6-methoxy-pyridin-3-yl)-benzenesulfonylamino]-thiophene-2-sulfonic acid amide;
- (E)-2-(2-(4-(1,1,2,2-tetrafluoroethoxy)phenyl)vinylsulfonamido)benzenesulfonamide;
- (E)-2-(2-(2-(difluoromethoxy)phenyl)vinylsulfonamido)benzenesulfonamide;
- (E)-2-(2-(4′-(trifluoromethyl)biphenyl-4-yl)vinylsulfonamido)benzenesulfonamide;
- (E)-2-(2-(2-isopropoxyphenyl)vinylsulfonamido)benzenesulfonamide;
- (E)-2-(2-(2-(methylthio)phenyl)vinylsulfonamido)benzenesulfonamide;
- (E)-2-(2-(2-(trifluoromethoxy)phenyl)vinylsulfonamido)benzenesulfonamide;
- (E)-2-(2-(2-ethoxyphenyl)vinylsulfonamido)benzenesulfonamide;
- 2-(2-(4-(1,1,2,2-tetrafluoroethoxy)phenyl)ethylsulfonamido)benzenesulfonamide;
- 2-(2-(4′-(trifluoromethyl)biphenyl-4-yl)ethylsulfonamido)benzenesulfonamide;
- 2-[2-(2-Hydroxy-phenyl)-ethenesulfonylamino]-benzenesulfonamide;
- 4-Chloro-2-(2-(4-(difluoromethoxy)phenyl)ethylsulfonamido)benzenesulfonamide;
- 4-Chloro-2-(2-(2-methoxyphenyl)ethylsulfonamido)benzenesulfonamide;
- 4-Chloro-2-(2-(4-chloro-2-methoxyphenyl)ethylsulfonamido)benzenesulfonamide;
- 2-(2-(3,4-Dichlorophenyl)ethylsulfonamido)-3,5-difluorobenzenesulfonamide;
- 2,3-Dichloro-N-(4-fluoro-5-methoxy-2-sulfamoylphenyl)benzenesulfonamide;
- 4-Chloro-2-(2-(3,4-dichlorophenyl)ethylsulfonamido)benzenesulfonamid;
- 5-Chloro-2-(2-(4-chlorophenyl)ethylsulfonamido)-4-fluorobenzenesulfonamide;
- 5-Chloro-2-(2-(3,4-dichlorophenyl)ethylsulfonamido)-4-fluorobenzenesulfonamide;
- 5-(2-(4-(Furan-2-yl)phenyl)ethylsulfonamido)-2,3-dihydro-1H-indene-4-sulfonamide;
- 6-(2-(4-(Furan-2-yl)phenyl)ethylsulfonamido)-2,3-dihydro-1H-indene-5-sulfonamid;
- 5-(2-(3,4-Dichlorophenyl)ethylsulfonamido)-2,3-dihydro-1H-indene-4-sulfonamide;
- (E)-5-(2-(4-Chlorophenyl)vinylsulfonamido)-2,3-dihydro-1H-indene-4-sulfonamide;
- (E)-6-(2-(4-Chlorophenyl)vinylsulfonamido)-2,3-dihydro-1H-indene-5-sulfonamide;
- 2-[[3-(3,4-Dichlorophenyl)phenyl]sulfonylamino]-N-methyl-benzenesulfonamide; and
- 2-(2-(6-((4-(Trifluoromethyl)phenyl)ethynyl)pyridin-3-yl)ethylsulfonamido)-benzenesulfonamide;
- or a pharmaceutically acceptable salt thereof.
- 20. A process for the preparation of a compound of formula (I) or a pharmaceutically acceptable salt thereof as defined in embodiment 1 which comprises,
- (a) reacting a compound of formula (II)
- wherein R1, R3, A and m are as defined in formula (I),
- with a compound of formula (III)
- wherein L1, L2, G1 and G2 are as defined in formula (I) and X represents a leaving group such as a halogen; or
- (b) when L2 represents a direct bond and G1 and G2 are both aromatic moieties, reacting a compound of formula (IV)
- wherein Hal represents a halogen atom and R1, R3, A, m and L1 are as defined in formula (I), with a nucleophile G2-M wherein M represents an organo-tin or organo boronic acid group; and optionally after (a) or (b) carrying out one or more of the following:
-
- converting the compound obtained to a further compound of the invention
- forming a pharmaceutically acceptable salt of the compound.
- 21. A pharmaceutical composition comprising a compound of formula (I) or a pharmaceutically acceptable salt thereof as in any one of embodiments 1 to 19 in association with a pharmaceutically acceptable adjuvant, diluent or carrier.
- 22. A process for the preparation of a pharmaceutical composition as in embodiment 21 which comprises mixing a compound of formula (I) or a pharmaceutically acceptable salt thereof as in any one of embodiments 1 to 19 with a pharmaceutically acceptable adjuvant, diluent or carrier.
- 23. A compound of formula (I) or a pharmaceutically acceptable salt thereof
- wherein:
- A is selected from phenyl or a 5- or 6-membered heteroaryl or a 5- or 6-membered heterocyclenyl moiety; said phenyl or 5- or 6-membered heteroaryl or 5- or 6-membered heterocyclenyl moiety in group A being optionally fused to a phenyl, a 5- or 6-membered heteroaryl, C5-6carbocyclyl or C5-6heterocyclyl ring;
- each R1 is independently selected from halogen, nitro, SF5, CN, NR5R6, OH, CHO, CO2C1-4alkyl, CONR5R6, C1-4alkyl, C2-4alkenyl, C2-4alkynyl, C1-4alkoxy, G3, OG3 or OCH2G3; said C1-4alkyl, C2-4alkenyl, C2-4alkynyl or C1-4alkoxy being optionally substituted by one or more substituents selected from OH, NR5R6, C1-4alkyl or F;
- m represents an integer 0,1, 2, 3 or 4;
- each R3 is independently selected from hydrogen, CN and C1-4alkyl; said C1-4alkyl being optionally substituted with OH, CN, C1-4alkoxy, NR7R8, or one or more F atoms;
- L1 represents a direct bond, C1-4alkylene, C2-4alkenylene or C2-4alkynylene;
- L2 represents a direct bond, —O—, —OCH2—, C1-2alkylene, —C≡C— or —NHCONH—;
- G1 represents phenyl, 5- or 6-membered heteroaryl, C3-10carbocyclyl or C5-8heterocyclyl;
- G2 represents H, C1-6alkyl, phenyl, 5- or 6-membered heteroaryl, C3-10carbocyclyl or C5-8heterocyclyl; said C1-6alkyl being optionally further substituted by one or more groups selected from OH, C1-6alkoxy and halogen;
- the phenyl, heteroaryl, carbocyclyl or heterocyclyl moieties in G1 and G2 being optionally fused to one or two further rings independently selected from phenyl, a 5- or 6-membered heteroaryl, C5-6carbocyclyl or C5-6heterocyclyl ring;
- any phenyl, heteroaryl, carbocyclyl or heterocyclyl moieties in G1 and G2 being optionally substituted by one or more substituents independently selected from halogen, OH, CN, NO2, CO2R9, C1-6alkyl, C1-6alkoxy, C1-4thioalkoxy, SO2NR10R11, NR12R13, —NHCOC(OH)(CH3)CF3 or —CH2OCH2CF2CHF2; said C1-6alkyl or C1-6alkoxy being optionally substituted by OH or by one or more F atoms;
- R9 represents C1-6alkyl;
- G3 represents phenyl or 5- or 6-membered heteroaryl; and
- each R4, R5, R6, R7, R8, R10, R11, R12 and R13 is independently selected from H or C1-4alkyl;
- with the proviso that the following compounds are excluded:
- 2-amino-N-(2-sulfamoylphenyl)benzenesulfonamide;
- methyl 4-(5-fluoro-3-methylbenzo[b]thiophene-2-sulfonamido)-3-sulfamoylbenzoate;
- 4-nitro-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 2-nitro-N-(2-sulfamoylphenyl)benzenesulfonamide;
- 4-amino-N-(2-sulfamoylphenyl)benzenesulfonamide;
- N-(2-sulfamoylphenyl)quinoline-8-sulfonamide;
- for the use in therapy.
- 24. Use of a compound of formula (I) or a pharmaceutically acceptable salt thereof
- wherein:
- A is selected from phenyl or a 5- or 6-membered heteroaryl or a 5- or 6-membered heterocyclenyl moiety; said phenyl or 5- or 6-membered heteroaryl or 5- or 6-membered heterocyclenyl moiety in group A being optionally fused to a phenyl, a 5- or 6-membered heteroaryl, C5-6carbocyclyl or C5-6heterocyclyl ring;
- each R1 is independently selected from halogen, nitro, SF5, CN, NR5R6, OH, CHO, CO2C1-4alkyl, CONR5R6, C1-4alkyl, C2-4alkenyl, C2-4alkynyl, C1-4alkoxy, G3, OG3 or OCH2G3; said C1-4alkyl, C2-4alkenyl, C2-4alkynyl or C1-4alkoxy being optionally substituted by one or more substituents selected from OH, NR5, R6, C1-4alkyl or F;
- m represents an integer 0, 1, 2, 3 or 4;
- each R3 is independently selected from hydrogen, CN and C1-4alkyl; said C1-4alkyl being optionally substituted with OH, CN, C1-4alkoxy, NR7R8, or one or more F atoms;
- L1 represents a direct bond, C1-4alkylene, C2-4alkenylene or C2-4alkynylene;
- L2 represents a direct bond, —O—, —OCH2—, C1-2alkylene, —C≡C— or —NHCONH—;
- G1 represents phenyl, 5- or 6-membered heteroaryl, C3-10carbocyclyl or C5-8heterocyclyl;
- G2 represents H, C1-6alkyl, phenyl, 5- or 6-membered heteroaryl, C3-10carbocyclyl or C5-8heterocyclyl; said C1-6alkyl being optionally further substituted by one or more groups selected from OH, C1-6alkoxy and halogen;
- the phenyl, heteroaryl, carbocyclyl or heterocyclyl moieties in G1 and G2 being optionally fused to one or two further rings independently selected from phenyl, a 5- or 6-membered heteroaryl, C5-6carbocyclyl or C5-6heterocyclyl ring;
- any phenyl, heteroaryl, carbocyclyl or heterocyclyl moieties in G1 and G2 being optionally substituted by one or more substituents independently selected from halogen, OH, CN, NO2, CO2R9, C1-6alkyl, C1-6alkoxy, C1-4thioalkoxy, SO2NR10R11, NR12R13, —NHCOC(OH)(CH3)CF3 or —CH2OCH2CF2CHF2; said C1-6alkyl or C1-6alkoxy being optionally substituted by OH or by one or more F atoms;
- R9 represents C1-6alkyl;
- G3 represents phenyl or 5- or 6-membered heteroaryl; and
- each R4, R5, R6, R7, R8, R10, R11, R12 and R13 is independently selected from H or C1-4alkyl;
- in the manufacture of a medicament for use in treating acute or chronic pain, nociceptive pain or neuropathic pain.
- 25. Use of a compound of formula (I) or a pharmaceutically acceptable salt thereof as in embodiment 24 in the manufacture of a medicament for use in treating osteoarthritis, rheumatoid arthritis, benign or malignant neoplasias, apnea, sudden infant death (SID), atherosclerosis, cancer, aneurysm, hyperthermia, myositis, Alzheimer's disease or arthritis.
- 26. A method of treating, or reducing the risk of, an inflammatory disease or condition which comprises administering to a patient in need thereof a therapeutically effective amount of a compound of formula (I) or a pharmaceutically acceptable salt thereof as in embodiment 23.
- 27. A method of treating, or reducing the risk of, human diseases or conditions in which modulation of microsomal prostaglandin E synthase-1 activity is beneficial which comprises administering to a patient in need thereof a therapeutically effective amount of a compound of formula (I) or a pharmaceutically acceptable salt thereof as in embodiment 24.
- The present invention will now be further explained by reference to the following illustrative examples.
- General Methods
- All solvents used were analytical grade and commercially available anhydrous solvents were routinely used for reactions. Reactions were typically run under an inert atmosphere of nitrogen or argon.
- 1H, 19F and 13C NMR spectra were recorded on a Varian Unity+400 NMR Spectrometer equipped with a 5 mm BBO probehead with Z-gradients, or a Varian Gemini 300 NMR spectrometer equipped with a 5 mm BBI probehead, or a Bruker Avance 400 NMR spectrometer equipped with a 60 μl dual inverse flow probehead with Z-gradients, or a
- Varian Mercury Plus 400 NMR Spectrometer equipped with a Varian 400 ATB PFG probe, or a Bruker DPX400 NMR spectrometer equipped with a 4-nucleus probehead equipped with Z-gradients, or a Bruker Avance 600 NMR spectrometer equipped with a 5 mm BBI probehead with Z-gradients, or a Bruker av400 NMR spectrometer operating at 400 MHz 1H and 100 MHz for 13C equipped with a 3 mm flow injection SEI 1H/D-13C probehead with Z-gradients, using a BEST 215 liquid handler for sample injection, or a Bruker DRX600 NMR spectrometer, operating at 600 MHz for 1H, 150 MHz for 13C, and 60 MHz for 15N equipped with a 5 mm TXI probehead with Z-gradients, or equipped with a 5 mm BBO probehead with Z-gradients, or equipped with a 2.5 mm BBI probehead with Z-gradients, or a Bruker 500 MHz Avance III NMR spectrometer, operating at 500 MHz for 1H, 125 MHz for 13C, and 50 MHz for 15N equipped with a 5 mm TXI probehead with Z-gradients or equipped with a 5 mm TCI cryogenically cooled probehead with Z-gradients. Unless specifically noted in the examples, spectra were recorded at 400 MHz for proton, 376 MHz for fluorine-19 and 100 MHz for carbon-13.
- The following reference signals were used: the middle line of DMSO-d6 δ 2.50 (1H), δ 39.51 (13C); the middle line of CD3OD δ 3.31 (1H) or δ 49.15 (13C); CDCl3 δ 7.26 (1H) and the middle line of CDCl3 δ 77.16 (13C) (unless otherwise indicated). NMR spectra are either reported from high to low field or from low to high field.
- Mass spectra were recorded on a Waters LCMS consisting of an Alliance 2795 (LC), Waters PDA 2996 and a ZQ single quadrupole mass spectrometer. The mass spectrometer was equipped with an electrospray ion source (ESI) operated in a positive or negative ion mode. The capillary voltage was 3 kV and cone voltage was 30 V. The mass spectrometer was scanned between m/z 100-700 with a scan time of 0.3s. Separations were performed on either Waters X-Terra MS C8 (3.5 μm, 50 or 100 mm×2.1 mm i.d.) or an ACE 3 AQ (100 mm×2.1 mm i.d.) obtained from ScantecLab. Flow rates were regulated to 1.0 or 0.3 mL/min, respectively. The column temperature was set to 40° C. A linear gradient was applied using a neutral or acidic mobile phase system, starting at 100% A (A: 95:5 10 mM NH4OAc:MeCN, or 95:5 8 mM HCOOH:MeCN) ending at 100% B (MeCN).
- Alternatively, mass spectra were recorded on a Waters LCMS consisting of an Alliance 2690 Separations Module, Waters 2487 Dual 1 Absorbance Detector (220 and 254 nm) and a Waters ZQ single quadrupole mass spectrometer. The mass spectrometer was equipped with an electrospray ion source (ESI) operated in a positive or negative ion mode. The capillary voltage was 3 kV and cone voltage was 30 V. The mass spectrometer was scanned between m/z 97-800 with a scan time of 0.3 or 0.8 s. Separations were performed on a Chromolith Performance RP-18e (100×4.6 mm). A linear gradient was applied starting at 95% A (A: 0.1% HCOOH (aq.)) ending at 100% B (MeCN) in 5 minutes. Flow rate: 2.0 mL/min.
- Alternatively, LC-MS analyses were performed on a LC-MS system consisting of a Waters 5 Alliance 2795HPLC, a Waters PDA 2996 diode array detector, a Sedex 85 ELS detector and a ZQ single quadrupole mass spectrometer. The mass spectrometer was equipped with an electrospray ion source (ES) operated in positive and negative ion mode. The capillary voltage was set to 3.3 kV and the cone voltage to 28 V, respectively. The mass spectrometer scanned between m/z 100-800 with a scan time of 0.3 s. The diode array 10 detector scanned from 200-400 nm. The temperature of the ELS detector was adjusted to 40° C. and the pressure was set to 1.9 bar. Separation was performed on an Gemini C18, 3.0 mm×50 mm, 3 μm, (Phenomenex) run at a flow rate of 1 mL/min. A linear gradient was applied starting at 100% A (A: 10 mM NH4OAc in 5% CH3CN) ending at 100% B (B: CH3CN) in 4.0 min followed by 100 % B until 5.5 min. The column oven temperature was 15 set to 40° C.
- Alternatively, LC-MS analyses were performed on a LC-MS consisting of a Waters sample manager 2777C, a Waters 1525μ binary pump, a Waters 1500 column oven, a Waters ZQ single quadrupole mass spectrometer, a Waters PDA2996 diode array detector and a Sedex 85 ELS detector. The mass spectrometer was configured with an atmospheric pressure 20 chemical ionisation (APCI) ion source which was further equipped with atmospheric pressure photo ionisation (APPI) device. The mass spectrometer scanned in the positive mode, switching between APCI and APPI mode. The mass range was set to m/z 100-800 using a scan time of 0.1 s. The APPI repeller and the APCI corona were set to 0.58 kV and 0.70 μA, respectively. In addition, the desolvation temperature (350° C.), desolvation gas 25 (450 L/Hr) and cone gas (0 L/Hr) were constant for both APCI and APPI mode. Separation was performed using a Gemini column C18, 3.0 mm×50 mm, 3 μm, (Phenomenex) and run at a flow rate of 0.8 mL/min. A linear gradient was used starting at 100% A (A: 10 mM NH4OAc in 5% MeOH) and ending at 100% B (MeOH) in 4.0 min followed by 100% B until 5.5 min. The column oven temperature was set to 55° C.
- Microwave irradiation was performed in a Creator™, Initiator™ or Smith Synthesizer™ Single-mode microwave cavity producing continuous irradiation at 2450 MHz.
- HPLC analyses were performed on an Agilent HP1000 system consisting of G1379A Micro Vacuum Degasser, G1312A Binary Pump, G1367A Well plate auto-sampler, G1316A Thermostatted Column Compartment and G1315B Diode Array Detector. Column: X-Terra MS, Waters, 3.0×100 mm, 3.5 μm. The column temperature was set to 40° C. and the flow rate to 1.0 ml/min. The Diode Array Detector was scanned from 210-300 nm, step and peak width were set to 2 nm and 0.05 min, respectively. A linear gradient was applied, starting at 100% A (A: 95:5 10 mM NH4OAc:MeCN) and ending at 100% B (B:MeCN), in 4 min. Alternatively, HPLC analyses were performed on a Gynkotek P580 HPG consisting of gradient pump with a Gynkotek UVD 170S UV-vis.-detector equipped with a Chromolith Performance RP column (C18, 100 mm×4.6 mm). The column temperature was set to 25° C. A linear gradient was applied using MeCN/0.1 trifluoroacetic acid in MilliQ water, run from 10% to 100% MeCN in 5 minutes. Flow rate: 3 ml/min.
- Thin layer chromatography (TLC) was performed on Merck TLC-plates (Silica gel 60 F254) and UV visualized the spots. Column chromatography was performed on a Combi Flash® Companion™ using RediSep™ normal-phase columns or using Merck Silica gel 60 (0.040-0.063 mm). Typical solvents used for column chromatography were mixtures of chloroform/methanol, dichloromethane/methanol, heptane/ethyl acetate, chloroform/methanol/ammonia (aq.) and dichlorormethane/methanol/NH3 (aq.). SCX ion exchange columns were performed on Isolute® columns. Chromatography through ion exchange columns were typically performed in solvents such a methanol.
- Preparative chromatography was run on a Waters autopurification HPLC with a diode array detector. Column: XTerra MS C8, 19×300 mm, 10 μm. Narrow gradients with MeCN/(95:5 0.1M NH4OAc:MeCN) were used at a flow rate of 20 ml/min. Alternatively, purification was achieved on a semi preparative Shimadzu LC-8A HPLC with a Shimadzu SPD-10A UV-vis.-detector equipped with a Waters Symmetry® column (C18, 5 μm, 100 mm×19 mm). Narrow gradients with MeCN/0.1% trifluoroacetic acid in MilliQ Water were used at a flow rate of 10 ml/min.
- Alternatively, preparative chromatography was run on a Waters FractionLynx system with a Autosampler combined Automated Fraction Collector (Waters 2767), Gradient Pump (Waters 2525), Column Switch (Waters CFO) and PDA (Waters 2996). Column; XTerra® Prep MS C8 10 μm OBD™ 19×300 mm, with guard column; XTerra® Prep MS C8 10 μm 19×10 mm Cartridge. A gradient from 100% A (95% 0.1M NH4OAc in MilliQ water and 5% MeCN) to 100% B (100% MeCN) was applied for LC-separation at flow rate 20 mL/min. The PDA was scanned from 210-350 nm. UV triggering determined the fraction collection. Alternatively, preparative chromatography was run on a Waters FractionLynx system with a Autosampler combined Automated Fraction Collector (Waters 2767), Gradient Pump (Waters 2425), Make Up Pump (Waters 515), Waters Passive Splitter, Column Switch (Waters SFO), PDA (Waters 2996) and Waters ZQ mass spectrometer. Column; XBridge™ Prep C8 5 μm OBD™ 19×250 mm, with guard column; XTerra® Prep MS C8 10 μm 19×10 mm Cartridge. A gradient from within 100% A (95% 0.1 M NH4OAc in MilliQ water and 5% MeCN) to 100% B (100% MeCN) was applied for LC-separation at flow rate 20 mL/min. The PDA was scanned from 210-350 nm. The ZQ mass spectrometer was run with ESI in positive or negative mode. The Capillary Voltage was 3 kV and the Cone Voltage was 30V. Mixed triggering, UV and MS signal, determined the fraction collection.
- H-cube (http://thalesnano.com/H-Cube) is a system for hydrogenation under continuous flow conditions (typical settings used were full hydrogen, flow 1 ml/min, ethyl acetate/methanol 1:1 as eluent, 50° C.).
- Naming was done with CambridgeSoft MedChem ELN v2.1, ChemDraw Ultra 7.0 or ACDName
- Abbreviations:
- DCM dichloromethane
- DMAP 4-(dimethylamino)pyridine
- DMF N,N-dimethylformamide
- DMSO dimethyl sulfoxide
- TFA trifluoroacetic acid
- THF tetrahydrofuran
- RT room temperature
- Rf retention time
-
- 2-[[(E)-2-(4-Benzofuran-2-ylphenyl)ethenyl]sulfonylamino]benzenesulfonamide (Example 3, 43 mg, 95 μmol) was dissolved in ethyl acetate (5 ml) and subjected to the H-cube (1 ml/min, full hydrogen. 10% Pd/C). The material obtained was purified by preparative HPLC (XTerra MS C8 column, acetonitrile/ammonium acetate buffer) to give the title compound (12 mg, 28%) as a white solid. The compound of Example 2 was also obtained.
- 1H NMR (400 MHz, CD3OD) δ ppm 7.94 (dd, 1H), 7.75-7.82 (m, 3H), 7.53-7.60 (m, 2H), 7.49 (d, 1H), 7.29 (d, 2H), 7.18-7.29 (m, 3H), 7.13 (s, 1H), 3.50-3.58 (m, 2H), 3.09-3.17 (m, 2H);
- ESMS: m/z [M+1]457.0, [M−1]455.0.
-
- The title compound was obtained from the synthesis of Example 1 as a white solid (12 mg, 28%).
- 1H NMR (400 MHz, CD3OD) δ ppm 7.93 (dd, 1H), 7.76 (dd, 1H), 7.55 (td, 1H), 7.28 (d, 2H), 7.24 (t, 1H), 7.14-7.20 (m, 3H), 7.11 (t, 1H), 6.84 (t, 1H), 6.77 (d, 1H), 5.68 (dd, 1H), 3.60 (dd, 1H), 3.46-3.53 (m, 2H), 3.03-3.12 (m, 3H);
- ESMS: m/z [M+1]458.9, [M−1]456.9.
-
- 2-Aminobenzenesulfonamide (38 mg, 0.22 mmol) was dissolved in pyridine (2 ml), (E)-2-(4-benzofuran-2-ylphenyl)ethenesulfonyl chloride (70 mg, 0.22 mmol) was added and the mixture stirred for 2 h. The solvent was removed in vacuo and the residue was purified by preparative HPLC (XTerra MS C8 column, acetonitrile/ammonium acetate buffer) to give the title compound (46 mg, 46%).
- 1H NMR (400 MHz, CD3OD) δ ppm 7.87-7.93 (m, 3H), 7.75 (d, 1H), 7.63 (d, 2H), 7.61 (d, 1H), 7.50-7.58 (m, 3H), 7.31 (td, 1H), 7.29 (s, 1H), 7.21-7.26 (m, 2H), 7.18 (d, 1H);
- ESMS: m/z [M+1]454.7, [M−1]452.8.
- Sulfurylchloride (0.21 mL, 2.6 mmol) was added to a solution of triphenylphosphine (0.68 g, 2.5 mmol) in CH2Cl2 (10 mL) at 0° C. The ice-bath was then removed and (E)-2-(4-benzofuran-2-ylphenyl)ethenesulfonic acid (0.38 g, 1.18 mmol) was added and the resulting mixture was stirred for 6 h at RT. A catalytic amount of tetrabutyl ammonium iodide was added and the reaction was stirred for an additional 12 h. The mixture was then filtered and the filtrate was concentrated in vacuo and the product was purified by column chromatography eluting with 20% EtOAc/hexanes to give the product as a yellow solid (0.27 g, 73%).
- 1H NMR (400 MHz, CDCl3) δ ppm 7.95-8.01 (m, 2H), 7.78 (d, 1H), 7.63-7.70 (m, 3H), 7.56-7.59 (m, 1H), 7.37 (ddd, 1H), 7.30 (d, 1H), 7.27-7.30 (m, 1H), 7.21 (d, 1H).
- 1-Bromo-4-[(E)-2-ethoxysulfonylethenyl]benzene (1.0 g, 3.4 mmol) and benzofuran-2-boronic acid (0.61 g, 3.7 mmol) were dissolved in DMF (20 mL). Sodium carbonate (2M, 10 mL) and tetrakis(triphenylphosphine) palladium(0) (200 mg, 0.17 mmol) were added and the resulting mixture was stirred under argon at 90° C. for 12 h. Water was added to the mixture and the resulting precipitate was collected, washed with water and dried to give the product as a sodium salt which was used in the next step without further purification (0.38 g, 35%).
- 1H NMR (400 MHz, DMSO-d6) δ ppm 7.89 (d, 2H), 7.59-7.69 (m, 4H), 7.48 (s, 1H), 7.33 (td, 1H), 7.27 (td, 1H), 6.95 (s, 2H);
- ESMS: m/z [M−1]298.9.
- n-Butyllithium (59.0 mL, 1.6M solution in hexanes) was added dropwise during 25 min to a solution of ethylmethanesulfonate (9.1 mL, 86 mmol) in THF (200 mL) at −78° C. The reaction mixture was stirred for 15 min then diethyl chlorophosphate (7.2 ml, 50 mmol) was added dropwise. The solution was stirred at −78° C. for 30 min, allowed to warm to 0° C. and then cooled to −78° C. again. 4-Bromobenzaldehyde (9.2 g, 50 mmol) in THF (50 mL) was then added dropwise during 15 minutes and the resulting mixture was then allowed to slowly reach RT and stirred overnight. Water (20 mL) was added to the mixture and the reaction was concentrated in vacuo. The residue was extracted with diethyl ether and the combined organic extracts were washed with aqueous saturated sodium chloride, dried over magnesium sulfate, filtered and the solvent was evaporated in vacuo. Purification by column chromatography, using 15% ethyl acetate/hexanes as the eluent, afforded the title compound (9.3 g, 64%).
- 1H NMR (600 MHz, CDCl3) δ ppm 7.58 (d, 2H), 7.55 (d, 1H), 7.39 (d, 2H), 6.75 (d, 1H), 4.25 (q, 2H), 1.42 (t, 3H).
-
- The compound was synthesized using an analogous procedure to that described for Example 3 to give 23 mg, 44%.
- 1H NMR (400 MHz, CD3OD) δ ppm 7.91 (d, 2H), 7.76 (dd, 1H), 7.58-7.66 (m, 4H), 7.52 (d, 1H), 7.48 (d, 1H), 7.27-7.36 (m, 3H), 7.24 (t, 1H), 7.16 (d, 1H);
- ESMS: m/z [M−1]470.6.
-
- The compound was synthesized using an analogous procedure to that described for Example 3 to give 31 mg, 16%.
- 1H NMR (400 MHz, CD3OD) δ ppm 7.88-7.96 (m, 3H), 7.67 (d, 2H), 7.62 (d, 1H), 7.61 (d, 1H), 7.53 (d, 1H), 7.48 (dd, 1H), 7.29-7.34 (m, 2H), 7.21-7.27 (m, 1H), 7.21 (d, 1H), 6.88-6.99 (m, 1H);
- ESMS: m/z [M−1]471.1.
-
- The compound was synthesized using an analogous procedure to that described for Example 3 to give 39 mg, 19%.
- 1H NMR (400 MHz, CD3OD) δ ppm 7.92 (d, 2H), 7.67 (d, 2H), 7.62 (d, 1H), 7.60 (d, 1H), 7.49-7.56 (m, 3H), 7.32 (td, 1H), 7.29 (s, 1H), 7.24 (td, 1H), 7.15 (d, 1H), 6.94-7.04 (m, 1H);
- ESMS: m/z [M+1]473.0, [M−1]471.0.
-
- 2-[[(E)-1-(4-Chlorophenyl)prop-1-en-2-yl]sulfonylamino]benzenesulfonamide (100 mg, 0.26 mmol) was dissolved in ethyl acetate (3 ml) and subjected to the H-cube (1 ml/min, full hydrogen. 10% Pd/C, 3 runs). The material obtained was purified by preparative HPLC (XTerra MS C8 column, acetonitrile/ammonium acetate buffer) to give the title compound (38 mg, 38%). The compound of Example 8 was also obtained.
- 1H NMR (400 MHz, CD3OD) δ ppm 7.94 (dd, 1H), 7.83 (dd, 1H), 7.56 (ddd, 1H), 7.19-7.30 (m, 3H), 7.14 (d, 2H), 3.50-3.63 (m, 1H), 3.38 (dd, 1H), 2.72 (dd, 1H), 1.24 (d, 3H);
- ESMS: m/z [M−1]385.1, 387.0.
-
- The title compound (12 mg, 12%) was obtained from the synthesis of Example 7.
- 1H NMR (400 MHz, CD3OD) δ ppm 7.94 (dd, 1H), 7.84 (dd, 1H), 7.55 (ddd, 1H), 7.16-7.30 (m, 4H), 7.13 (d, 2H), 3.49-3.61 (m, 1H), 3.42 (dd, 1H), 2.68 (dd, 1H), 1.23 (d, 3H);
- ESMS: m/z [M−1]353.
-
- 2-Aminobenzenesulfonamide (61 mg, 0.35 mmol) was dissolved in pyridine (2 ml) and (E)-2-(4-benzofuran-2-ylphenyl)ethenesulfonyl chloride (70 mg, 0.22 mmol) was added and the reaction stirred for 2 h. The solvent was removed in vacuo and the residue was purified by preparative HPLC (XTerra MS C8 column, acetonitrile/ammonium acetate buffer) to give the title compound (46 mg, 46%).
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.14 (s, 1H), 7.88 (s, 2H), 7.85 (d, 1H), 7.54-7.63 (m, 3H), 7.49 (s, 4H), 7.21-7.31 (m, 1H), 2.13 (d, 3H);
- ESMS: m/z [M+1]386.9, 388.9, [M−1]385.0, 387.0.
- Tetrabutylammonium iodide (7.0 g, 19.0 mmol) was added to a solution of 1-chloro-4-[(E)-2-ethoxysulfonylprop-1-enyl]benzene (4.5 g, 17.3 mmol) in acetone (40 mL) at RT. The mixture was heated to reflux and stirred overnight, then cooled to RT and concentrated in vacuo. The residue was taken up in DCM then washed with water, dried over magnesium sulfate, filtered and concentrated in vacuo to give the title compound as the tetrabutylammonium salt which was used in the next step without further purification. Sulfuryl chloride (3.1 mL, 38.0 mmol) was added to a solution of triphenylphosphine (9.73 g, 37.2 mmol) in DCM (100 mL) at 0° C. The ice-bath was then removed and the reaction was stirred for 15 min. The crude (E)-1-(4-chlorophenyl)prop-1-ene-2-sulfonic acid salt (17.3 mmol) dissolved in DCM (40 mL) was then added during 15 min and the resulting mixture was stirred for 2 h at RT. The reaction was concentrated in vacuo and the product was purified by column chromatography using 10% EtOAc/hexanes as the eluent to give the product (3.3 g, 77%) as a yellow solid.
- 1H NMR (400 MHz, CDCl3) δ ppm 7.71 (br. s., 1H), 7.45-7.49 (m, 2H), 7.38-7.43 (m, 2H), 2.48 (d, 3H).
- n-Butyllithium (35.0 mL, 1.6M solution in hexanes) was added dropwise during 25 min to a solution of ethyl ethanesulfonate (6.9 g, 50 mmol) (Synth. Comm. 1985, 15, 1057) in THF (100 mL) at −78° C. The reaction mixture was stirred for 15 min then diethyl chlorophosphate (4.35 ml, 30 mmol) was added dropwise. The solution was stirred at −78° C. for 30 min, allowed to warm to 0° C. and then cooled to −78° C. again. 4-Chlorobenzaldehyde (4.2 g, 30 mmol) in THF (30 mL) was then added dropwise during 15 minutes and the resulting mixture was stirred for 1 h at −78° C. and then allowed to slowly reach RT. Water (10 mL) was added to the mixture and the mixture was concentrated in vacuo. The residue was extracted with diethyl ether and the combined organic extracts were washed with aqueous saturated sodium chloride, dried over magnesium sulfate, filtered and the solvent was evaporated in vacuo. Purification by column chromatography, using a gradient of 10% ethyl acetate/hexanes to 15% ethyl acetate/hexanes as eluent, afforded the title compound (4.5 g, 58%).
- 1H NMR (400 MHz, CDCl3) δ ppm 7.56 (br. s., 1H), 7.40-7.45 (m, 2H), 7.32-7.38 (m, 2H), 4.21 (q, 2H), 2.27 (d, 3H), 1.41 (t, 3H).
-
- 2-[[(E)-1-(4-Chlorophenyl)prop-1-en-2-yl]sulfonylamino]-4-fluoro-benzenesulfonamide (60 mg, 0.15 mmol) was dissolved in ethyl acetate (3 mL) and mixture was subjected to reduction using the H-cube instrument. The reaction mixture was eluted through the Pd/C (10%) cartridge using full hydrogen (1 atm) at 1 ml/min. The mixture was subjected to the H-cube once again using the same conditions. The crude product was purified by preparative HPLC (XTerra MS C8 column, acetonitrile/ammonium acetate buffer) to give the title compound (25 mg, 38%).
- 1H NMR (400 MHz, CD3OD) δ ppm 7.87 (dd, 1H), 7.52 (dd, 1H), 7.26 (d, 2H), 7.17 (dd, 2H), 6.74 (ddd, 1H), 3.37-3.48 (m, 2H), 2.70 (dd, 1H), 1.22 (d, 3H);
- ESMS: m/z [M−1]470.0, 472.1.
-
- The compound was synthesized using an analogous procedure to that described for Example 9 to give 78 mg, 55%.
- 1H NMR (400 MHz, CD3OD) δ ppm 7.95 (dd, 1H), 7.63 (s, 1H), 7.35-7.49 (m, 5H), 6.97 (t, 1H), 2.19 (d, 3H);
- ESMS: m/z [M−1]402.9, 404.8.
-
- The compound was synthesized using an analogous procedure to that described for Example 9 to give 35 mg, 71%.
- 1H NMR (400 MHz, CD3OD) δ ppm 7.74 (dd, 1H), 7.65 (dd, 1H), 7.54 (br. s., 1H), 7.39-7.44 (m, 2H), 7.35-7.38 (m, 2H), 7.32 (ddd, 1H), 2.19 (d, 3H);
- ESMS: m/z [M−1]402.9, 404.8.
-
- The compound was synthesized using an analogous procedure to that described for Example 9 to give 33 mg, 69%.
- 1H NMR (400 MHz, CD3OD) δ ppm 7.56 (br s, 1H), 7.47-7.54 (m, 2H), 7.38-7.44 (m, 2H), 7.31-7.37 (m, 2H), 6.95-7.04 (m, 1H), 2.18 (d, 3H);
- ESMS: m/z [M+1]404.9, 406.9, [M−1]402.9, 404.8.
-
- The compound was synthesized using an analogous procedure to that described for Example 9 to give 10 mg, 20%.
- 1H NMR (400 MHz, CD3OD) δ ppm 7.74-7.87 (m, 1H), 7.34-7.49 (m, 7H), 2.37 (d, 3H);
- ESMS: m/z [M−1]402.5, 404.7.
-
- The compound was synthesized using an analogous procedure to that described for Example 9 to give 35 mg, 73%.
- 1H NMR (400 MHz, CD3OD) δ ppm 7.72 (d, 1H), 7.59 (d, 1H), 7.53 (s, 1H), 7.37-7.43 (m, 2H), 7.31-7.37 (m, 3H), 2.33 (s, 3H), 2.17 (d, 3H);
- ESMS: m/z [M+1]398.8, 400.8, [M−1]400.9, 402.8.
-
- 2-[[(E)-2-[4-(2-Furyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide (Example 18) (140 mg, 0.35 mmol) was dissolved in ethyl acetate (1.5 mL) and methanol (1.5 mL) and the mixture was subjected to reduction using the H-cube instrument. The reaction mixture was eluted through the Pd/C (10%) cartridge using full hydrogen (1 atm) at 0.5 ml/min. The solvent was then removed and the crude material was purified on semi-preparative HPLC using an X-Terra column to give the title compound (18 mg, 13%) as a white solid.
- The compound of Example 17 was also obtained.
- 1H NMR (400 MHz, CD3OD) δ ppm 7.95 (dd, 1H), 7.78 (dd, 1H), 7.53-7.61 (m, 3H), 7.51 (dd, 1H), 7.27 (t, 1H), 7.20 (d, 2H), 6.69 (d, 1H), 6.48 (dd, 1H), 3.46-3.56 (m, 2H), 3.03-3.12 (m, 2H);
- ESMS: m/z [M−1]404.8.
-
- The title compound was obtained from the synthesis of Example 17 as a white solid (7 mg, 5%).
- 1H NMR (400 MHz, CD3OD) δ ppm 7.93 (dd, 1H), 7.75 (dd, 1H), 7.55 (td, 1H), 7.18-7.27 (m, 3H), 7.13 (d, 2H), 4.80 (t, 1H), 4.05 (dt, 1H), 3.85-3.92 (m, 1H), 3.44-3.51 (m, 2H), 3.03-3.09 (m, 2H), 2.24-2.33 (m, 1H), 1.96-2.05 (m, 2H), 1.62-1.80 (m, 1H);
- ESMS: m/z [M−1]409.0.
-
- 2-Aminobenzenesulfonamide (61 mg, 0.35 mmol) was dissolved in pyridine (2.5 mL) and (E)-2-[4-(2-furyl)phenyl]ethenesulfonyl chloride ) (see General procedure for synthesis of styrenyl sulfonyl chlorides) (95 mg, 0.35 mmol) was added. The reaction was stirred at RT overnight. The solvent was removed, the residue was partitioned between water and ethyl acetate, made acidic (HCl) and the mixture was extracted twice with ethyl acetate. The combined organic phases were washed with water, treated with brine, dried (MgSO4), and evaporated to give the title compound (140 mg, 98%).
- MS m/z M−H 403, M+H 405; Rf 3.12 min (System: Gemini column 3μ C18, 50×3.0 mm, 10 mM ammonium acetate with 5% acetonitrile/acetonitrile 0-100% during 6 min, 1 mL/min).
-
- The title compound (57 mg, 42%) was synthesized by a method analogous to that described for the preparation of Example 16.
- 1H NMR (400 MHz, CD3OD) δ ppm 7.95 (dd, 1H), 7.77 (dd, 1H), 7.57 (dd, 1H), 7.24-7.32 (m, 1H), 7.20 (d, 2H), 7.02 (d, 2H), 6.74 (t, 1H), 3.46-3.55 (m, 2H), 3.02-3.12 (m, 2H);
- ESMS: m/z [M−1]404.8.
-
- The title compound (135 mg, 94%) was synthesized by a method analogous to that described for the preparation of Example 18.
- MS m/z M−H 403, M+H 405; Rf 3.12 min (System: Gemini column 3μ C18, 50×3.0 mm, 10 mM ammonium acetate with 5% acetonitrile/acetonitrile 0-100% during 6 min, 1 mL/min).
-
- The title compound (74 mg, 36%) was synthesized by a method analogous to that described for the preparation of Example 16.
- 1H NMR (400 MHz, CD3OD) δ ppm 7.77 (dd, 1H), 7.69 (dd, 1H), 7.35 (ddd, 1H), 7.21-7.27 (m, 2H), 7.15-7.22 (m, 2H), 3.45-3.54 (m, 2H), 3.00-3.10 (m, 2H);
- ESMS: m/z [M−1]391.1, 393.1
-
- The title compound (210 mg, 90%) was synthesized by a method analogous to that described for the preparation of Example 18.
- 1H NMR (400 MHz, CD3CN-d3) δ ppm 7.66 (dd, 1H), 7.58 (dd, 1H), 7.51-7.55 (m, 2H), 7.51 (d, 1H), 7.40-7.44 (m, 2H), 7.31 (ddd, 1H), 7.04 (d, 1H);
-
- ESMS: m/z [M−1]389.0.
- Stock solutions:
- The anilines (800 μmol) were dissolved in pyridine (2 ml).
- The sufonyl chlorides (prepared according to the General procedure for synthesis of styrenyl sulfonyl chlorides) (1.2 mmol) were dissolved in pyridine (6 ml).
- Reaction: (24 reactions)
- The stock solution of the anilines (500 μl, 200 μmol) were mixed with the stock solutions of the sulfonyl chlorides (1.0 ml, 200 μl) in 24 wells and the reaction was put on a shaker overnight. The solvent was removed (centrifuge) and preparative chromatography was run on a Waters FractionLynx system with a Autosampler combined Automated Fraction Collector (Waters 2767), Gradient Pump (Waters 2525), Regeneration Pump (Waters 600), Make Up Pump (Waters 515), Waters Active Splitter, Column Switch (Waters CFO), PDA (Waters 2996) and Waters ZQ mass spectrometer. Column; XBridge Prep C8 5 μm OBD 19×100 mm, with guard column; XTerra® Prep MS C8 10 mm 19×10 mm Cartridge. A gradient from 100% A (95% 0.1M NH4OAc in MilliQ water and 5% MeCN) to 100% B (100% MeCN) was applied for LC-separation at flow rate 25 ml/min. The PDA was scanned from 210-350 nm. The ZQ mass spectrometer was run with ESI in positive mode. The Capillary Voltage was 3 kV and the Cone Voltage was 30V. Mixed triggering, UV and MS signal, determined the fraction collection.
- Purity analysis was run on a Water Acquity system with PDA (Waters 2996) and Waters ZQ mass spectrometer. Column; Acquity UPLC™ BEH C8 1.7 μm 2.1×50 mm. The column temperature was set to 65° C. A linear 2 min 15 sec gradient from 100% A (A: 95% 0.01M NH4OAc in MilliQ water and 5% MeCN) to 100% B (5% 0.01M NH4OAc in MilliQ water and 95% MeCN) was applied for LC-separation at flow rate 1.0 ml/min. The PDA was scanned from 210-350 nm and 254 nm was extracted for purity determination. The ZQ mass spectrometer was run with ESI in pos/neg switching mode. The Capillary Voltage was 3 kV and the Cone Voltage was 30V.
-
- 11 mg, 13%. ESMS: m/z [M+1]424.8, 426.8, Rf 1.00 min.
-
- 27 mg, 32%. ESMS: m/z [M+1]424.8, 426.8, Rf 1.01 min.
-
- 32 mg, 37%. ESMS: m/z [M+1]424.8, 426.8, Rf 1.03 min.
-
- 18 mg, 19%. ESMS: m/z [M+1]464.8, 466.8, Rf 0.95 min.
-
- 23 mg, 24%. ESMS: m/z [M+1]464.8, 466.8, Rf 1.00 min.
-
- 25 mg, 27%. ESMS: m/z [M+1]464.8, 466.8, Rf 1.03 min
-
- 32 mg, 36%. ESMS: m/z [M+1]446.8, 448.8, Rf 0.99 min.
-
- 22 mg, 29%. ESMS: m/z [M+1]399.0 (ammonia adduct), Rf 0.77 min.
-
- 32 mg, 42%. ESMS: m/z [M+1]398.9 (ammonia adduct), Rf 0.77 min.
-
- 32 mg, 42%. ESMS: m/z [M+1]398.9 (ammonia adduct), Rf 0.77 min.
-
- 27 mg, 37%. ESMS: m/z [M+1]363.9, Rf 0.74 min.
-
- 14 mg, 17%. ESMS: m/z [M+1]422.9, Rf 0.96 min.
-
- 19 mg, 22%. ESMS: m/z [M+1]422.9, Rf 1.01 min.
-
- 25 mg, 30%. ESMS: m/z [M+1]422.9, Rf 1.04 min.
-
- 17 mg, 21%. ESMS: m/z [M+1]402.9, Rf 0.90 min.
-
- 23 mg, 28%. ESMS: m/z [M+1]402.9, Rf 0.95 min.
-
- 28 mg, 34%. ESMS: m/z [M+1]402.9, Rf 0.99 min.
-
- 20 mg, 26%. ESMS: m/z [M+1]384.9, Rf 0.94 min.
-
- 20 mg, 23%. ESMS: m/z [M+1]422.9, Rf 0.90 min.
-
- 20 mg, 23%. ESMS: m/z [M+1]422.9, Rf 0.93 min.
-
- 28 mg, 33%. ESMS: m/z [M+1]422.8, Rf 0.96 min.
-
- To a solution of 4-[2-(4-chloro-phenyl)-ethanesulfonylamino]-3-sulfamoyl-benzoic acid (190 mg, 0.45 mmol) in dry THF (15 mL) was added LiAlH4 (1M in THF, 2 mL, 2 mmol). The reaction mixture was heated at 45° C. overnight, then cooled to 0° C. and quenched by addition of water, followed by 1N HCl. The product was extracted with ethyl acetate, the combined organic phases were washed with water and brine, and dried over sodium sulfate. Volatiles were removed under reduced pressure and the crude product was purified by preparative HPLC to title compound as a white solid (51 mg, 28%).
- 1H NMR (400 MHz, acetone-d6): δ ppm 8.78 (br. s., 1H), 7.97 (d, 1H), 7.75 (d, 1H), 7.58 (dd, 1H), 7.21-7.35 (m, 4H), 7.15 (br. s., 2H), 4.66 (s, 2H), 3.49-3.66 (m, 2H), 3.03-3.16 (m, 2H);
- ESMS: m/z [M−1]402.93 and 404.82 (Cl isotopes).
- A mixture of 2-[2-(4-chloro-phenyl)-ethanesulfonylamino]-5-methylbenzenesulfonamide (Example 45) (2.0 g, 5.14 mmol), 2.5N NaOH (27 mL, 6.75 mmol), water (100 mL) and KMnO4 (3.35 g, 21.19 mmol) was stirred at 50° C. overnight. The reaction mixture was then filtered through Celite and the filtrate was acidified using 2N HCl. The product was extracted with ethyl acetate and the organic phase was concentrated under reduced pressure. The crude product was purified by flash column chromatography using a gradient of 5 to 10% MeOH in DCM to afford the title compound (490 mg, 23%).
- 1H NMR (400 MHz, CD3OD): δ ppm 8.59 (s, 1H), 8.16 (d, J=8.61 Hz, 1H), 7.84 (d, J=8.61 Hz, 1H), 7.23 (m, 2H), 7.17 (m, 2H), 4.87 (br. S, 4H), 3.66-3.54 (m, 2H), 3.10 (d, J=8.22 Hz, 2H);
- ESMS: m/z [M−1]416.92.
-
- A mixture of 2-amino-5-methyl-benzenesulfonamide (5.0 g, 26.84 mmol), 2-(4-chloro-phenyl)-ethanesulfonyl chloride (8.0 g, 33.56 mmol) and pyridine (100 mL) was stirred at RT overnight. The reaction mixture was concentrated under reduced pressure and the residue was partitioned between ethyl acetate and 1N HCl. The organic phase was concentrated in vacuo and the crude product was purified by flash column chromatography to afford the title compound (8.0 g, 51%).
- 1H NMR (400 MHz, CDCl3): δ ppm 8.13 (br. s, 1H), 7.79 (br. s, 1H), 7.56-7.44 (m, 1H), 7.40 (d, 1H), 7.32-7.19 (m, 3H), 7.15 (d, 2H), 5.26 (br., s, 2H), 3.56-3.43 (m, 2H), 3.19 (dd, 2H), 2.40 (br, s, 3H);
- ESMS: [M−1]386.86 and 388.82 (Cl isotopes).
- A mixture of 7-methyl-1,1-dioxo-1,4-dihydro-2-H-benzo[1,2,4]thiadiazin-3-one (7.20 g, 36.36 mmol) in aqueous H2SO4 (50% v/v, 200 mL) was heated at 105° C. for 16 h. The clear solution was then cooled to 0° C. and neutralized using 5N NaOH. The product was extracted with ethyl acetate. The combined extracts were washed with water and concentrated under reduced pressure. The crude product was purified by flash column chromatography using ethyl acetate/hexane (1:1) to afford 4.5 g (85%) of the title compound.
- 1H NMR (400 MHz, DMSO-d6): δ ppm 7.33 (s, 1H), 7.16 (s, 2H), 7.05 (d, 1H), 6.70 (d, 1H), 5.65 (s, 2H), 2.19 (s, 3H).
- p-Toluidine (11.00 g, 102.65 mmol) dissolved in nitromethane (50 mL) was added slowly over 20 minutes to a solution of chlorosulfonyl isocyanate (16.89 g, 119.40 mmol) in nitromethane (160 mL) at −5° C. The suspension was stirred at −5° C. for an additional 15 minutes and AlCl3 (17.00 g, 129.00 mmol) was added portionwise. The reaction mixture was then heated at 105° C. for 30 minutes. The hot solution was poured onto ice (1L) and the resulting precipitate was filtered off and washed with water (300 mL). The solid was then dissolved in a hot aqueous sodium bicarbonate solution (10 g/200 mL), treated with charcoal, and filtered. The cooled solution was neutralized using 6N HCl and the resulting white precipitate was filtered off, washed with water and dried to afford the title compound (14 g, 64%).
- 1H NMR (400 MHz, DMSO-d6): δ ppm 11.19 (s, 1H), 7.60 (s, 1H), 7.42 (d, 1H), 7.09 (d, 1H), 2.38 (s, 3H).
-
- To a solution of NaH (57%, 56 mg, 1.33 mmol) in dry DMSO (10 mL) was added N-Boc-2-[2-(4-chloro-phenyl)-ethanesulfonylamino]-5-hydroxy-benzenesulfonamide (Example 46b) (220 mg, 0.44 mmol). The reaction mixture was stirred at RT for 2 h (solution turned brown). Toluene-4-sulfonic acid pyridine-2-ylmethyl ester (141 mg, 0.53 mmol) was then added and stirring was continued for 4 h at RT. The reaction mixture was quenched by addition of saturated NH4Cl and the product was extracted with EtOAc (3×30 mL). The combined extracts were dried over anhydrous MgSO4 and concentrated in vacuo to give a yellow oil (182 mg) which was then dissolved in DCM (2 mL). The resulting solution was cooled to 0° C., TFA (1 mL) was added and the reaction mixture was allowed to warm to RT over 3 h. Volatiles were removed under reduced pressure and the residue was purified by preparative HPLC to give the title compound as a white solid (TFA salt, 90 mg, 60%).
- 1H NMR (400 MHz, CD3OD): δ ppm 8.63 (d, 1H), 8.09 (t, 1H), 7.76( d, 1H) 7.70( (d, 1H), 7.63 (d, 1H), 7.57(t, 1H), 7.29 (m, 1H), 7.24(d, 2H), 7.18 (d, 2H), 5.30 (s, 2H), 3.47 (m, 2H), 3.05( m, 2H); 19F NMR (400 MHz, CDCl3): δ(ppm) −77.57;
- ESMS: m/z [M++1]481.89.
- Powdered KOH (1.74 g, 30.65 mmol) was added to a vigorously stirred solution of pyridin-2-yl-methanol (2 mL, 20.72 mmol) in THF (100 mL) at 0° C. The reaction mixture was stirred for 15 minutes and tosyl chloride (5.13 g, 26.90 mmol) was then added. The reaction mixture was allowed to warm to RT and stirring was continued overnight. The reaction mixture was quenched with saturated NaHCO3 and the product was extracted with EtOAc (3×50 mL). The combined extracts were dried over MgSO4 and concentrated in vacuo to give a yellow oil. The residue was purified by flash column chromatography using EtOAc:hexane (1:4) to yield the title compound (4.6 g, 85%) as a yellowish solid.
- 1H NMR (400 MHz, CDCl3): δ ppm 8.53 (m, 1H), 7.85 (d, 2H), 7.73 (t, 1H), 7.44 (d, 1H), 7.35(d, 2H), 7.25 (m, 1H), 5.14 (s, 2H), 2.44 (s, 3H);
- ESMS: m/z [M++1]263.31.
- To a solution of 2-[2-(4-chloro-phenyl)-ethanesulfonylamino]-5-hydroxy-benzenesulfonamide (2.50 g, 6.41 mmol) in DCM (150 mL) at 0° C. was added Et3N (4.46 mL, 14.07 mmol) followed by Boc2O (3.07 g, 14.07 mmol). The resulting mixture was stirred for 15 minutes at 0° C. and then DMAP (0.78 g, 6.41 mmol) was added in one portion (reaction became exothermic). The reaction mixture was stirred at 0° C. for another hour, allowed to warm to RT and stirred overnight. The organic phase was washed with 2N HCl (3×50 mL), water (3×30 mL) and dried over MgSO4. Removal of solvent under reduced pressure afforded 3.5 g (94%) of the di-Boc intermediate which was used in the next step without purification. In the 1H NMR spectrum, two singlets were observed at 1.42 ppm and 1.34 ppm, which correspond to O-Boc and N-Boc groups respectively.
- To a solution of the di-Boc intermediate (1.20 g, 2.03 mmol) in EtOH (150 mL) was added LiOH (10% aqueous solution, 40 mL) at RT. The reaction mixture was stirred at 55° C. for 14 h. The progress of the reaction was monitored by 1H NMR. The singlet at 1.42 ppm for the O-Boc group was not observed upon completion of the reaction. The reaction mixture was concentrated to approximately half volume and acidified by the addition of 5N HCl to a pH of 4-5. The product was extracted with EtOAc (3×50 mL) and the combined extracts were dried over MgSO4 and concentrated in vacuo. The residue was purified by column chromatography using 1% MeOH/DCM to afford the product as a yellow solid (747 mg, 75%).
- 1H NMR (400 MHz, CD3OD): δ ppm 7.64 (d, 1H), 7.37 (s, 1H), 7.26(d, 2H), 7.18 (d, 2H), 7.09 (m, 1H), 3.44 (m, 2H), 3.05(m, 2H), 1.34 (s, 9H);
- ESMS: m/z [M−1]489.25.
- To a solution of 2-[2-(4-chloro-phenyl)-ethanesulfonylamino]-5-methoxy-benzenesulfonamide (4.0 g, 9.90 mmol) in DCM (250 mL) at −78° C., BBr3 (1M in DCM, 70 mL, 70 mmol) was added dropwise over 20 minutes. The reaction mixture was allowed to warm to RT and the stirring was continued overnight. The reaction mixture was then cooled to −78° C., quenched by addition of methanol (20 mL) and water (50 mL), and the product was extracted with ethyl acetate. The organic extracts were washed with water, dried over sodium sulfate and concentrated under reduced pressure. The crude product was recrystallized from methanol to the title compound (3.45 g, 92%).
- 1H NMR (400 MHz, DMSO-D6): δ ppm 10.04 (s, 1H), 8.66 (s, 1H), 7.65 (s, 2H), 7.45 (d, 1H), 7.35 (m, 3H), 7.27 (d, 2H), 6.99(dd, 1H), 3.53 (m, 2H), 2.97 (m, 2H);
- ESMS: m/z [M−1]389.
- To a solution of 2-amino-5-methoxy-benzenesulfonamide (4.0 g, 19.80 mmol) in pyridine (60 mL), 2-(4-chloro-phenyl)-ethanesulfonyl chloride (4.73 g, 19.79 mmol) was added slowly at 0° C. The reaction mixture was stirred at RT for 16h and then concentrated under reduced pressure at 35° C. The residue was then partitioned between ethyl acetate and 1N HCl. The organic layer was separated, dried over sodium sulfate and concentrated in vacuo. The crude product was purified by flash column chromatography using ethyl acetate/hexane (1:1) to give the title compound (7.1 g, 88%).
- 1H NMR (400 MHz, CD3OD) δ ppm 7.65 (d, 1H), 7.49 (d, 1H), 7.21-7.27 (m, 2H), 7.15-7.19 (m, 2H), 7.12-7.15 (m, 1H), 3.83 (s, 3H), 3.42-3.47 (m, 2H), 3.02 (dd, 2H).
-
- Following the general procedure as described in Example 46, sodium phenolate generated from N-Boc-2-[2-(4-chloro-phenyl)-ethanesulfonylamino]-5-hydroxy-benzenesulfonamide (210 mg, 0.43 mmol) was reacted with toluene-4-sulfonic acid 5-methyl-[1,3,4]oxadiazol-2-ylmethyl ester to afford 178 mg of the crude alkylation product. After deprotection of the sulfonamide group, the crude product was purified by preparative HPLC to give the title compound as a white solid (TFA salt, 95 mg, 65%).
- 1H NMR (400 MHz, CD3OD) δ ppm 7.71 (d, 1H), 7.61 (d, 1H), 7.30(m, 1H), 7.25 (d, 2H), 7.18(d, 2H), 5.35(s, 2H), 3.48 (m, 2H), 3.05 (m, 2H), 2.54 (s, 3H);
- ESMS: m/z [M−1]484.85.
- Following the general procedure as described in Example 46a, the title compound was prepared from commercially available 5-methyl-[1,3,4]oxadiazol-2-yl)-methanol (1.0 g, 80%).
- 1H NMR (400 MHz, CD3OD): δ ppm 7.80 (d, 2H), 7.41 (d, 2H), 5.30 (s, 2H), 2.50 (s, 3H), 2.30 (s, 3H);
- ESMS: m/z [M++1]268.89.
-
- 2-Aminobenzene sulfonamide (153 mg, 0.89 mmol) was added in one portion to a solution of 2-(2-chloro-phenyl)-ethenesulfonyl chloride (150 mg, 0.63 mmol) in pyridine (2 mL) at RT. After 30 minutes of stirring, no more starting material was detected by tlc. The reaction mixture was concentrated under reduced pressure and the residue was taken up in ethyl acetate and a small amount of methanol. The organic phase was washed with 1N HCl, saturated sodium bicarbonate, dried over MgSO4, and concentrated under reduced pressure. Purification by preparative tlc afforded the target compound, which was then recrystallized from ethyl acetate/pentane/diethyl ether to afford the title compound (85 mg, 36%) as a white solid.
- 1H NMR (400 MHz, DMSO-d6): δ ppm 9.19 (s, 1H), 7.89-7.83 (m, 2H), 7.81 (d, 1H), 7.71 (d, 1H), 7.66-7.59 (m, 2H), 7.57-7.50 (m, 1H), 7.50-7.43 (m, 1H), 7.43-7.36 (m, 1H), 7.35-7.27 (m, 1H);
- ESMS: m/z [M−1]371.09;
- CHN for C14H13ClN2O4S2. Calc.: C, 45.10; H, 3.51; N, 7.51. Found: C, 44.89; H, 3.71; N, 7.54.
-
- 2-Aminobenzene sulfonamide (194 mg, 1.13 mmol) was added in one portion to a solution of 2-(4-chloro-2-methoxyphenyl)-ethenesulfonyl chloride (300 mg, 1.13 mmol) in pyridine (3 mL) at RT. After 1 h stirring, no more starting material was detected by tlc. The reaction mixture was concentrated and the residue was taken up in ethyl acetate with a small amount of methanol. The organic phase was washed with 1N HCl, saturated sodium bicarbonate, dried over MgSO4 and concentrated under reduced pressure. The crude product was purified on silica gel using DCM/methanol, 99:1 to afford material which was further purified by recrystallization from methanol to give the title compound (184 mg, 41%) as a white solid.
- 1H NMR (400 MHz, DMSO-d6): δ ppm 9.05 (s, 1H), 7.86-7.78 (m, 3H), 7.66-7.55 (m, 4H), 7.40 (d, 1H), 7.31-7.23 (m, 1H), 7.18 (d, 1H), 7.05 (dd, 1H), 3.88 (s, 3H);
- ESMS: m/z [M−1]401.12;
- CHN for C15H15ClN2O5S2, 0.6MeOH. Calc.: C, 44.39; H, 4.15; N, 6.64. Found: C, 44.42; H, 4.29; N, 6.72.
- Thionyl chloride (8.1 mL, 111.19 mmol) was added dropwise at 0° C. to a solution of 2-(4-chloro-2-methoxy-phenyl)-ethenesulfonic acid (5.53 g, 22.24 mmol) in DMF (10 mL). The reaction mixture was stirred at RT for 3 h and then was poured onto crushed ice. Ethyl acetate (50 mL) was added to the mixture. The organic phase was separated, washed with brine (3×20 mL), dried over MgSO4 and concentrated under reduced pressure. The oily residue was purified on silica gel using hexane/ethyl acetate, 4:1 to give the target compound which was then recrystallized from pentane/diethyl ether to afford the title compound (2.30 g, 61% over 3 steps) as a white solid.
- 1H NMR (400 MHz, CDCl3): δ ppm 7.78 (d, 1H), 7.52 (d, 1H), 7.38 (d, 1H), 7.04 (dd, 1H), 6.99 (d, 1H), 3.97 (s, 3H);
- ESMS: m/z [M−1]247.05 (corresponding sulfonic acid M=248);
- CHN for C9H8Cl2O3S. Calc.: C, 40.47; H, 3.02. Found: C, 40.67; H, 3.11.
- Tetrabutyl-ammonium iodide (6.25 g, 16.91 mmol) was added to a solution of 2-(4-chloro-2-methoxy-phenyl)-ethenesulfonic acid ethyl ester (3.90 g, 14.09 mmol) in acetone (300 mL). The reaction mixture was heated at reflux overnight, then was cooled to RT and concentrated under reduced pressure. The residue was purified on silica gel using hexane/ethyl acetate, 1:1 to elute the impurities, then DCM/methanol, 95:5 to afford crude material which contained tetrabutyl-ammonium iodide. The oily residue was triturated with diethyl ether to afford a beige solid (9.15 g) which was used in the next step without further purification. To a stirred solution of the solid (9.15 g crude from preceding step) in water (100 mL) and methanol (20 mL) was added dropwise 3N HCl until the pH of the solution was approximately 1. The reaction mixture was concentrated under reduced pressure to afford an oily residue which was purified on silica gel using DCM/methanol, 95:5 to 90:10 to afford the title compound (5.53 g) contaminated with some tetrabutyl-ammonium chloride and which was used in the next step without further purification.
- 1H NMR (400 MHz, CDCl3): δ ppm 7.52 (d, 1H), 7.13-7.08 (m, 2H), 6.97 (dd, 1H), 6.83 (d, 1H), 3.86 (s, 3H).
- A 2.5M solution of n-butyllithium in hexanes (6.15 mL, 15.37 mmol) was added dropwise to a stirred solution of (diethoxy-phosphoryl)-methanesulfonic acid ethyl ester (4.0 g, 15.37 mmol) in THF (200 mL) at −78° C. The reaction mixture was stirred at −78° C. for 15 minutes. A solution of 4-chloro-2-methoxy-benzaldehyde (2.41 g, 14.10 mmol) in THF (20 mL) was then added dropwise. After the addition was complete the reaction mixture was warmed to RT, stirred for 1 h and quenched with brine. The reaction mixture was concentrated under reduced pressure and DCM (200 mL) was added. The organic layer was separated, washed with brine (3×100 mL) and dried over MgSO4. Removal of solvent under reduced pressure, followed by purification on silica gel using hexane/ethyl acetate, 4:1 to 2:1 afforded the title compound (3.90 g, quantitative) as a white solid.
- 1H NMR (400 MHz, CDCl3): δ ppm 7.70 (d, 1H), 7.36 (d, 1H), 6.99 (d, 1H), 6.97-6.92 (m, 2H), 4.22 (q, 2H), 3.92 (s, 3H), 1.40 (t, 3H); ESMS: m/z [M++1]277.04.
-
- 2-Amino-6-fluoro-benzenesulfonamide (38 mg, 200 μmol) and (E)-2-(4-phenylphenyl)ethenesulfonyl chloride (56 mg, 200 μmol) were dissolved in pyridine (1.5 mL) and the reaction mixture was shaken for 12 h. The solvent was removed in vacuum and the residues were purified by HPLC to give the product (16.6 mg, 19%).
- MS m/z M−H 431, Rf 1.1 min (see above system in General procedure for Examples 23 to 43).
- The compounds of Examples 51 to 68 were prepared using the general procedure described for Example 50.
-
- 35 mg, 45%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.06 (br. s., 1H), 7.90 (br.s., 2H), 7.80 (br. s., 1H), 7.56-7.72 (m, 3H), 7.38-7.56 (m, 5H); MS m/z M−H 389.
-
- 30 mg, 35%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.03 (br. s., 1H), 7.92 (br. s., 2H), 7.44-7.76 (m, 12H), 7.35-7.44 (m, 2H); MS m/z M−H 431.
-
- 10 mg, 12%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.23 (br. s., 1H), 7.64-7.95 (m, 10H), 7.35-7.57 (m, 5H), 7.14 (br. s., 1H); MS m/z M−H 431.
-
- 10 mg, 11%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.09 (br. s., 1H), 8.09 (s, 1H), 7.97 (d, 1H), 7.89 (br. s., 2H), 7.78 (d, 1H), 7.44-7.71 (m, 6H); MS m/z M−H 423.
-
- 13 mg, 16%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 8.95 (s, 1H), 7.91 (br. s., 2H), 7.55-7.68 (m, 4H), 7.47-7.55 (m, 1H), 7.43 (t, 1H), 7.32 (d, 1H), 7.08 (d, 1H), 6.97 (t, 1H), 3.84 (s, 3H);
- MS m/z M−H 385.
-
- 25 mg, 32%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.86 (s, 1H), 8.15 (br. s., 2H), 7.88 (br. s., 1H), 7.34-7.77 (m, 7H), 7.12 (t, 1H); MS m/z M+H 391, M−H 389.
-
- 11 mg, 14%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.78 (s, 1H), 8.15 (s, 2H), 7.75 (d, 1H), 7.66 (d, 1H), 7.56-7.64 (m, 1H), 7.42-7.49 (m, 1H), 7.31-7.40 (m, 2H), 7.05-7.16 (m, 2H), 6.98 (t, 1H), 3.87 (s, 3H); MS m/z M+H 387, M−H 385.
-
- 12 mg, 16%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.15 (br. s., 1H), 6.91-7.96 (m, 11H), 3.86 (s, 3H);
- MS m/z M+H 387, M−H 385.
-
- 22 mg, 25%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.25 (br. s., 1H), 7.67-7.82 (m, 7H), 7.34-7.63 (m, 9H); MS m/z M−H 431.
-
- 31 mg, 40%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.17 (br. s., 1H), 7.74 (d, 1H), 7.63 (d, 1H), 7.41-7.61 (m, 6H), 7.37 (d, 1H), 7.11 (d, 1H), 7.02 (t, 1H), 3.87 (s, 3H); MS m/z M+H 387, M−H 385.
-
- 9 mg, 10%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.28 (br. s., 1H), 8.14 (br. s., 1H), 8.01 (d, 1H), 7.61-7.93 (m, 7H), 7.39 (d, 1H), 7.15 (br. s., 1H); MS m/z M−H 423.
-
- 36 mg, 48%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.83 (s, 1H), 8.14 (br. s., 2H), 7.83 (t, 1H), 7.57-7.68 (m, 2H), 7.44-7.57 (m, 2H), 7.39 (d, 1H), 7.23-7.35 (m, 2H), 7.14 (t, 1H); MS m/z
- M+H 375, M−H 373.
-
- 31 mg, 41%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.04 (s, 1H), 7.80 (br. s., 1H), 7.74 (br. s., 2H), 7.59-7.66 (m, 2H), 7.37-7.55 (m, 6H), 2.29 (s, 3H); MS m/z M+H 387, M−H 385.
-
- 40 mg, 51%. MS m/z M+H 391, M−H 389, Rf 0.8 min (see above system in General procedure for Examples 23-43).
-
- 14 mg, 16%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.32 (br. s., 1H), 8.09 (s, 1H), 8.00 (d, 1H), 7.79 (d, 1H), 7.74 (d, 1H), 7.68 (t, 1H), 7.45-7.63 (m, 6H); MS m/z M+H 425, M−H 423.
-
- 9 mg, 10%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.87 (s, 1H), 8.11-8.21 (m, 3H), 8.03 (d, 1H), 7.55-7.84 (m, 5H), 7.41 (d, 1H), 7.12 (t, 1H); MS m/z M+H 425, M−H 423.
-
- 27 mg, 37%. MS m/z M+H 375, M−H 373, 0.8 min (see above system in General procedure for Examples 23-43).
-
- 9 mg, 10%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.06 (s, 1H), 8.09 (s, 1H), 7.96 (d, 1H), 7.71-7.82 (m, 3H), 7.49-7.69 (m, 5H), 7.40 (d, 1H), 2.29 (s, 3H); MS m/z M−H 419.
-
- (E)-2-(2-(4-Cyclopentenylphenyl)vinylsulfonamido)benzenesulfonamide (25.00 mg, 0.06 mmol) was dissolved in EtOAc (1.6 mL) and MeOH (1.6 mL) and hydrogenated under continuous flow hydrogenation conditions (full hydrogen, flow 1 ml/min, ethyl acetate/methanol 1:1 as eluent) at 50° C. using an H-Cube system. The solvent was removed in vacuo and the residue was purified by column chromatography on silica using heptane/ethyl acetate gradient mixtures as eluent (0-100% ethyl acetate) to give the product as a solid (14 mg, 55%).
- 1H NMR (400 MHz, DMSO-d6) δ ppm 8.93 (s, 1H), 7.81-7.91 (m, 3H), 7.68 (d, 1H), 7.61 (t, 1H), 7.33 (t, 1H), 7.06-7.16 (m, 4H), 3.51-3.60 (m, 2H), 2.83-2.99 (m, 3H), 1.90-2.00 (m, 2H), 1.67-1.79 (m, 2H), 1.55-1.68 (m, 2H), 1.39-1.52 (m, 2H);
- MS m/z M−H 407.
-
- 2-Aminobenzenesulfonamide (0.057 g, 0.33 mmol) was dissolved in pyridine (2.5 mL) followed by addition of (E)-2-(4-tert-butylphenyl)ethenesulfonyl chloride (0.078 g, 0.30 mmol) and the reaction was stirred at RT overnight. The solvent was then removed under redused pressure and the pale yellow oily residue was used in the next step without further purification. The crude material (0.118 g, 0.3 mmol) was dissolved in MeOH (5 mL) with a few drops of EtOAc and was subjected to reduction using the H-cube instrument. The reaction mixture was eluted through the Pd/C (10%) cartridge using full hydrogen (1 atm) at 1 ml/min. The mixture was subjected to the H-cube once again using the same conditions for full conversion of the starting material and purified by preparative HPLC (XTerra MS C8 column, acetonitrile/ammonium acetate buffer) to give the title compound (56 mg, 47%) as a white solid.
- 1H NMR (400 MHz, CDCl3) δ ppm 7.97 (dd, 1H), 7.56-7.66 (m, 2H), 7.27-7.35 (m, 3H), 7.13 (d, 2H), 3.52-3.58 (m, 2H), 3.17-3.23 (m, 2H), 1.30 (s, 9H). MS m/z M−H 395.
- The title compound (0.65 g, 16%) was synthesized by an analogous method to that described for the preparation of Example 9a.
- 1H NMR (400 MHz, CDCl3) δ ppm 7.73 (d, 1H), 7.50 (s, 4H), 7.21 (d, 1H), 1.35 (s, 9H); (GC)MS m/z M+H 259.
- The title compound (4.12 g, 62%) was synthesized by an analogous method to that described for the preparation of Example 9b.
- 1H NMR (400 MHz, CDCl3) δ ppm 7.60 (d, 1H), 7.46 (s, 4H), 6.71 (d, 1H), 4.22 (q, 2H), 1.40 (t, 3H), 1.34 (s, 9H). (GC)MS m/z M+H 269.
-
- The title compound (31 mg, 25%) was synthesized by the analogous method to that described for Example 70.
- 1H NMR (400 MHz, CDCl3) δ ppm 8.42 (s, 1H), 7.97 (dd, 1H), 7.41 (dd, 1H), 7.30-7.36 (m, 2H), 7.11-7.16 (m, 2H), 6.93-6.99 (m, 1H), 5.07 (s, 2H), 3.49-3.58 (m, 2H), 3.15-3.22 (m, 2H), 1.30 (s, 9H). MS m/z M−H 413.
-
- 2-Aminobenzenesulfonamide (0.52 g, 3 mmol) was dissolved in pyridine (20 mL) followed by addition of 2-(4-chlorophenyl)ethanesulfonyl chloride (0.80 g, 3.3 mmol) and the reaction was stirred at RT overnight. The solvent was then removed under reduced pressure and the crude material was purified by preparative HPLC (XTerra MS C8 column, acetonitrile/ammonium acetate buffer) to give the title compound (87 mg, 8%) as a white solid.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 8.95 (br. s., 1H), 7.81-7.90 (m, 3H), 7.67 (d, 1H), 7.60 (t, 1H), 7.28-7.35 (m, 3H), 7.21-7.27 (m, 2H), 3.55-3.63 (m, 2H), 2.95-3.02 (m, 2H); MS m/z M−H 373.
- The compounds of Examples 73, 75 to 78, 80 to 84 and 88 to 106 were prepared using the general method described for Example 72.
-
- 283 mg, 76%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.13 (s, 1H), 7.84 (s, 2H), 7.79-7.85 (m, 1H), 7.70 (d, 2H), 7.54-7.64 (m, 3H), 7.49 (d, 2H), 7.46 (d, 1H), 7.28 (t, 1H). MS m/z M−H 372.
-
- 2-[[(E)-2-Phenylethenyl]sulfonylamino]benzenesulfonamide (0.052 g, 0.15 mmol) was dissolved in MeOH (5 mL) followed by the addition of 10% Pd/C (30 mg). The solution was subjected to hydrogen overnight at 40° C. After filtration, the crude product was purified by preparative HPLC (XTerra MS C8 column, acetonitrile/ammonium acetate buffer) to give the title compound (4 mg, 8%) as a white solid.
- 1H NMR (400 MHz, CDCl3) δ ppm 8.26 (br. s, 1H), 7.98 (dd, 1H), 7.56-7.68 (m, 2H), 7.24-7.35 (m, 4H), 7.16-7.24 (m, 2H), 5.14 (br. s., 2H), 3.52-3.60 (m, 2H), 3.19-3.27 (m, 2H).
- MS m/z M−H 339.
-
- 90 mg, 42%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.12 (s, 1H), 7.85 (s, 2H), 7.82 (d, 1H), 7.52-7.70 (m, 5H), 7.36-7.47 (m, 4H), 7.26 (t, 1H). LC-MS, m/z, M−H 337, M+H, 339, Rf 2.99 min.
-
- 104 mg, 23%.
- 1H NMR (400 MHz, CD3CN) δ ppm 7.89 (dd, 1H), 7.69-7.73 (m, 1H), 7.56-7.61 (m, 1H), 7.29-7.37 (m, 2H), 7.19-7.28 (m, 3H), 3.49-3.56 (m, 2H), 3.17-3.24 (m, 2H). MS m/z M+H 375.
-
- 19 mg, 30%.
- 1H NMR (400 MHz, CDCl3) δ ppm 8.35 (s, 1H), 7.89-8.02 (m, 1H), 7.52-7.69 (m, 3H), 7.41-7.52 (m, 2H), 6.89-6.98 (m, 2H), 6.73-6.86 (m, 2H), 5.10 (s, 2H), 3.79-3.94 (m, 3H). MS m/z M−H 367.
-
- 164 mg, 45%.
- 1H NMR (400 MHz, CD3CN) δ ppm 7.79 (dd, 1H), 7.52-7.56 (m, 2H), 7.49 (d, 1H), 7.37-7.42 (m, 2H), 7.32 (dd, 1H), 7.03 (d, 1H), 6.70 (ddd, 1H). MS m/z M−H 389.
-
- A mixture of 2-[[(E)-2-(4-chlorophenyl)prop-1-enyl]sulfonylamino]benzenesulfonamide and 2-[2-(4-chlorophenyl)prop-2-enylsulfonylamino]benzenesulfonamide (40 mg, 0.11 mmol) was dissolved in methanol (3 ml) and subjected to the H-cube (1 ml/min, full hydrogen. 10% Pd/C, 2 runs). The product obtained was purified by preparative HPLC (XTerra MS C8 column, acetonitrile/ammonium acetate buffer) to give the title compound (13 mg, 32%) as a white solid.
- 1H NMR (400 MHz, CDCl3) δ ppm 7.93 (ddd, 1H), 7.51-7.60 (m, 2H), 7.18-7.34 (m, 6H), 3.58-3.68 (m, 1H), 3.45-3.57 (m, 2H), 1.51 (d, 3H). MS m/z M−H 353.
-
- 147 mg, 89%.
- 1H NMR (400 MHz, CD3CN) δ ppm 7.53-7.59 (m, 3H), 7.48-7.52 (m, 1H), 7.44-7.47 (m, 1H), 7.40-7.44 (m, 2H), 7.04 (d, 1H), 6.91-6.98 (m, 1H). MS m/z M−H 389.
-
- 140 mg, 69%.
- 1H NMR (400 MHz, CD3CN) δ ppm 7.88 (dd, 1H), 7.74 (dd, 1H), 7.64 (d, 1H), 7.60 (ddd, 1H), 7.40-7.45 (m, 2H), 7.32 (ddd, 1H), 7.27 (ddd, 1H), 7.11 (d, 1H).). MS m/z M−H 405.
-
- 143 mg, 81%.
- 1H NMR (400 MHz, CD3CN) δ ppm 7.85 (dd, 1H), 7.67 (dd, 1H), 7.58 (d, 1H), 7.56 (ddd, 1H), 7.41-7.47 (m, 2H), 7.18-7.26 (m, 3H), 7.00 (d, 1H), 2.34 (s, 3H). MS m/z M−H 351.
-
- 158 mg, 86%.
- 1H NMR (400 MHz, CD3CN) δ ppm 7.86 (dd, 1H), 7.73 (d, 1H), 7.68 (dd, 1H), 7.57 (ddd, 1H), 7.48 (dd, 1H), 7.42 (ddd, 1H), 7.23 (ddd, 1H), 7.21 (d, 1H), 7.03 (d, 1H), 6.97 (td, 1H), 3.87 (s, 3H). MS m/z M−H 367.
-
- 111 mg, 57%.
- 1H NMR (400 MHz, CD3CN) δ ppm 8.05-8.09 (m, 1H), 7.87-7.93 (m, 3H), 7.86 (dd, 1H), 7.77 (d, 1H), 7.72 (dd, 1H), 7.66 (dd, 1H), 7.53-7.61 (m, 3H), 7.22 (ddd, 1H), 7.18 (d, 1H).
- MS m/z M−H 387.
-
- 2-[[(E)-2-(4-Chlorophenyl)ethenyl]sulfonylamino]-4-fluoro-benzenesulfonamide (120 mg, 0.31 mmol) was dissolved in ethyl acetate (5 ml) and subjected to the H-cube (1 ml/min, full hydrogen. 10% Pd/C). The product obtained was purified by preparative HPLC (XTerra MS C8 column, acetonitrile/ammonium acetate buffer) to give the title compound (60 mg, 49%) as a white solid.
- 1H NMR (400 MHz, CD3CN) δ ppm 7.92 (dd, 1H), 7.44 (dd, 1H), 7.25-7.31 (m, 2H), 7.16-7.24 (m, 2H), 6.98 (ddd, 1H), 3.54-3.61 (m, 2H), 3.07-3.13 (m, 2H). MS m/z M−H 391.
-
- Sulfuryl chloride (60 mmol) was added dropwise to stirred anhydrous DMF (4.6 mL) cooled to 0° C. under nitrogen. After the addition was completed, the mixture was warmed to RT and stirred further for 0.5 h. 4-Chloro-α-methylstyrene (0.25 mol) was then slowly added and the reaction mixture was run overnight at 45° C. The reaction mixture was cooled and then poured onto crushed ice and then extracted with Et2O, washed with brine and dried over Na2SO4. After solvent removal, the residue was purified by chromatography (silica gel, eluents: ethyl acetate/heptane) to furnish a mixture of 2-(4-chlorophenyl)prop-2-ene-1-sulfonyl chloride and 2-(4-chlorophenyl)prop-1-ene-1-sulfonyl chloride (2.17 g, 29%) as a white solid.
- The title compound (72 mg, 14%) and the compound of Example 87 were then prepared using the general method described for Example 72.
- 1H NMR (400 MHz, CD3CN) δ ppm 7.87 (ddd, 1H), 7.65 (ddd, 1H), 7.56 (ddd, 1H), 7.36-7.45 (m, 4H), 7.23 (ddd, 1H), 6.73 (q, 1H), 2.45 (d, 3H). MS m/z M−H 385.
-
- The title compound (26 mg, 5%) was obtained from the synthesis in Example 86.
- 1H NMR (400 MHz, CD3CN) δ ppm 7.84 (ddd, 1H), 7.66 (dd, 1H), 7.58 (ddd, 1H), 7.39-7.43 (m, 2H), 7.27-7.31 (m, 2H), 7.24 (ddd, 1H), 5.70 (s, 1H), 5.39 (s, 1H), 4.50 (d, 2H).
- MS m/z M−H 385.
-
- 31 mg, 38%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.88 (s, 1H), 8.27 (s, 1H), 8.16 (s, 2H), 7.90-7.98 (m, 3H), 7.6 (d, 1H), 7.81 (d, 1H), 7.53-7.65 (m, 4H), 7.44 (d, 1H), 7.10 (t, 1H). MS m/z M−H 405.
-
- 35 mg, 41%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.85 (br. s., 1H), 8.14 (br. s., 2H), 7.95 (d, 1H), 7.82 (d, 1H), 7.53-7.77 (m, 5H), 7.37 (d, 1H), 7.16 (t, 1H). MS m/z M−H 423.
-
- 34 mg, 40%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.85 (br. s., 1H), 8.15 (s, 2H), 7.95 (d, 2H), 7.79 (d, 2H), 7.76 (d, 1H), 7.52-7.69 (m, 2H), 7.41 (d, 1H), 7.13 (t, 1H). MS m/z M−H 423.
-
- 31 mg, 36%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.83 (s, 1H), 8.15 (s, 2H), 7.55-7.71 (m, 6H), 7.51 (d, 1H), 7.39 (d, 1H), 7.11 (t, 1H). MS m/z M−H 433/435.
-
- 30 mg, 37%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.26 (br. s., 1H), 8.25 (s, 1H), 7.75-8.00 (m, 8H), 7.52-7.67 (m, 3H), 7.42 (dd, 1H), 7.02-7.20 (m, 1H). MS m/z M−H 405.
-
- 16 mg, 19%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.33 (br. s., 1H), 7.93 (d, 1H), 7.82-7.92 (m, 1H), 7.81 (d, 2H), 7.62-7.77 (m, 4H), 7.49-7.59 (m, 1H), 7.34 (d, 1H), 7.10-7.22 (m, 1H). MS m/z M−H 423.
-
- 23 mg, 27%.
- ESMS: m/z [M−1]423, [M+1]425, Rf 0.96 min according to the general MS procedure described for Examples 23-43.
-
- 26 mg, 30%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.23 (br. s., 1H), 7.77-7.94 (m, 3H), 7.59-7.73 (m, 5H), 7.52 (d, 1H), 7.37 (d, 1H), 7.01-7.22 (m, 1H). MS m/z M−H 433/435.
-
- 40 mg, 49%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.04 (s, 1H), 8.19 (s, 1H), 7.89-7.96 (m, 3H), 7.77 (d, 1H), 7.76 (s, 2H), 7.67 (d, 1H), 7.63 (s, 1H), 7.53-7.61 (m, 3H), 7.48 (d, 1H), 7.40 (d, 1H), 2.27 (s, 3H). MS m/z M−H 401.
-
- 42 mg, 50%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.06 (s, 1H), 7.87 (d, 2H), 7.77 (d, 2H), 7.73 (s, 2H), 7.65 (s, 1H), 7.60 (d, 1H), 7.48-7.56 (m, 2H), 7.40 (d, 1H), 2.28 (s, 3H). MS m/z M−H 419.
-
- 40 mg, 46%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.01 (s, 1H), 7.74 (s, 2H), 7.64 (s, 1H), 7.57-7.63 (m, 4H), 7.51 (d, 1H), 7.50 (d, 1H), 7.39 (d, 1H), 7.40 (d, 1H), 2.29 (s, 3H). MS m/z M−H 429/431.
-
- 34 mg, 41%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.06 (s, 1H), 8.19 (s, 1H), 7.88-7.99 (m, 5H), 7.78 (d, 1H), 7.43-7.72 (m, 7H). MS m/z M−H 405.
-
- 31 mg, 36%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.15 (br. s., 1H), 7.90 (d, 1H), 7.86 (br. s., 2H), 7.80 (d, 1H), 7.73 (t, 1H), 7.58-7.68 (m, 3H), 7.49-7.57 (m, 2H), 7.42 (d, 1H). MS m/z M−H 423.
-
- 31 mg, 37%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.11 (br. s., 1H), 7.83-7.95 (m, 4H), 7.74-7.81 (m, 2H), 7.45-7.69 (m, 5H). MS m/z M−H 423.
-
- 32 mg, 36%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.04 (s, 1H), 7.89 (br. s., 2H), 7.58-7.68 (m, 6H), 7.48 (d, 1H), 7.44-7.53 (m, 1H), 7.40 (d, 1H). MS m/z M−H 433/435.
-
- 18 mg, 24%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.11 (s, 1H), 7.84 (d, 1H), 7.78 (br. s., 2H), 7.59-7.64 (m, 2H), 7.51-7.59 (m, 1H), 7.42 (d, 1H), 7.39 (d, 1H), 7.28-7.35 (m, 1H), 7.18-7.26 (m, 2H). MS m/z M−H 373.
-
- 31 mg, 38%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.19 (br. s., 1H), 7.89 (d, 1H), 7.83 (d, 1H), 7.76-7.82 (m, 3H), 7.73 (t, 1H), 7.56-7.67 (m, 4H), 7.49 (d, 1H), 7.28-7.35 (m, 1H). MS m/z M−H 405.
-
- 35 mg, 43%.
- ESMS: m/z [M−1]405, [M+1]407, Rf 1.00 min. according to the general MS procedure for Examples 23-43.
-
- 36 mg, 43%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.12 (s, 1H), 7.83 (d, 1H), 7.81 (s, 2H),, 7.53-7.64 (m, 7H), 7.45 (d, 1H), 7.27 (t, 1H). MS m/z M−H 415/417.
-
- To a solution of (E)-2-(2-(4-bromophenyl)vinylsulfonamido)benzenesulfonamide (0.063 g, 0.15 mmol) in THF (10 mL) were added 4-methylbenzenesulfonohydrazide (1.676 g, 9.00 mmol) and sodium acetate (0.738 g, 9.00 mmol). (J. Org. Chem. 2006, 5870). The mixture was heated at reflux under argon overnight, quenched with water (5 mL), and extracted three times with ethyl acetate. The combined organic phases were extracted with 1M HCl, washed with water, brine and followed by organic solvent removal. The brine phase was then extracted three times with ethyl acetate. The combined organic solvent was then removed under reduced pressure and the crude product was purified by preparative HPLC (XTerra MS C8 column, acetonitrile/ammonium acetate buffer) to give the title compound (25 mg, 40%) as a white solid.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 8.94 (s, 1H), 7.79-7.94 (m, 3H), 7.56-7.72 (m, 2H), 7.39-7.50 (m, 2H), 7.26-7.38 (m, 1H), 7.15-7.24 (m, 2H), 3.53-3.67 (m, 2H), 2.90-3.02 (m, 2H). MS m/z M−H 417/419.
-
- A mixture of (E)-2-(2-(4-chlorophenyl)prop-1-enylsulfonamido)benzenesulfonamide and 2-[2-(4-chlorophenyl)prop-2-enylsulfonylamino]benzenesulfonamide (100 mg, 0.26 mmol) was dissolved in EtOAc in a 5 mL syringe. The solution was filtered and injected into the H-cube (1 ml/min, full hydrogen. 10% Pd/C). The solvent was then removed under reduced pressure and the crude material was purified by preparative HPLC (XTerra MS C8 column, acetonitrile/ammonium acetate buffer) to give the title compound (56 mg, 56%).
- 1H NMR (400 MHz, CDCl3) δ ppm 8.20 (s, 1H), 7.94 (dd, 1H), 7.48-7.62 (m, 2H), 7.21-7.32 (m, 3H), 7.08-7.18 (m, 2H), 5.07 (s, 2H), 3.53-3.64 (m, 1H), 3.42-3.54 (m, 2H), 1.48 (d, 3H). MS m/z M−H 387.
-
- (E)-2-(2-(2-Methoxyphenyl)vinylsulfonamido)benzenesulfonamide (94 mg, 0.26 mmol) was dissolved in methanol (5 mL) in a 5 mL syringe. The solution was injected to the H-cube (1 ml/min, full hydrogen; 10% Pd/C). The solvent was then removed under reduced pressure and the crude material was purified by preparative HPLC (XTerra MS C8 column, acetonitrile/ammonium acetate buffer) to give the title compound (69 mg, 73%).
- 1H NMR (400 MHz, CDCl3) δ ppm 8.28 (s, 1H), 7.98 (dd, 1H), 7.66 (dd, 1H), 7.56-7.62 (m, 1H), 7.27-7.32 (m, 1H), 7.21-7.27 (m, 1H), 7.16 (dd, 1H), 6.90 (td, 1H), 6.84 (dd, 1H), 5.23 (s, 2H), 3.75 (s, 3H), 3.53-3.63 (m, 2H), 3.15-3.25 (m, 2H). MS m/z M−H 369.
-
- The title compound (45 mg, 50%) was synthesized using the general method described for Example 108.
- 1H NMR (400 MHz, CD3CN) δ ppm 7.85 (dd, 1H), 7.66-7.76 (m, 4H), 7.63 (s, 1H), 7.50 (ddd, 1H), 7.30-7.39 (m, 2H), 7.28 (dd, 1H), 7.17 (ddd, 1H), 3.54-3.64 (m, 2H), 3.12-3.23 (m, 2H). MS m/z M−H 389.
-
- The title compound (13 mg, 27%) was synthesized using the general method described for Example 70.
- 1H NMR (400 MHz, CD3CN) δ ppm 7.89 (dd, 1H), 7.67-7.77 (m, 3H), 7.66 (s, 1H), 7.48 (dd, 1H), 7.27-7.40 (m, 3H), 6.92 (ddd, 1H), 3.64-3.73 (m, 2H), 3.14-3.27 (m, 2H). MS m/z M−H 407.
-
- The title compound (89 mg, 49%) was synthesized using the general procedure described for Example 72.
- 1H NMR (400 MHz, CD3CN) δ ppm 7.86-7.90 (m, 1H), 7.51-7.56 (m, 2H), 7.29-7.36 (m, 4H), 7.24-7.28 (m, 1H), 4.57 (s, 2H). MS m/z M−H 359.
-
- Tetrakis(triphenylphosphine)palladium(0) (14 mg, 0.012 mmol), was added to a solution of 2,3-dichlorophenylboronic acid (57 mg, 0.30 mmol) and 2-[(3-bromophenyl)-sulfonylamino]benzenesulfonamide (97 mg, 0.25 mmol) in DMF (1 mL). The reaction mixture was stirred at RT under argon for 15 minutes before addition of 2M Na2CO3 (0.5 mL). The reaction was stirred overnight at 90° C., filtered and purified by preparative HPLC (XTerra MS C8 column, acetonitrile/ammonium acetate buffer) to give the title compound (61 mg, 53%) as a white solid.
- 1H NMR (400 MHz, Acetone-d6) δ ppm 7.97-8.01 (m, 2H), 7.80-7.89 (m, 1H), 7.60-7.74 (m, 4H), 7.47-7.52 (m, 2H), 7.42 (d, 1H), 7.15 (t, 1H). MS m/z M−H 455/457/459.
- 3-Bromo-benzenesulfonyl chloride (8.34 mL, 57.9 mmol) was added to a solution of 2-amino-benzenesulfonamide (9.965 g, 57.9 mmol) dissolved in pyridine (90 mL). The reaction was stirred for 4 h followed by solvent removal under vacuo. The reaction mixture was added to water and the product was extracted three times with EtOAc. The combined organic extracts were washed with brine, dried (MgSO4), filtered and evaporated to furnish a dark yellow oil. The oil was dissolved in EtOAc and precipitated by addition of heptane. The solid was finally recrystallized from EtOH to give the title compound (10.784 g, 48%) as a white solid.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.39 (br. s., 1H), 8.04 (t, 1H), 7.84-7.91 (m, 2H), 7.80 (dd, 1H), 7.76 (s, 2H), 7.51-7.58 (m, 2H), 7.49 (dd, 1H), 7.27 (t, 1H). MS m/z M−H 389/391.
- The compounds of Examples 114 to 121 were prepared using the general method described for Example 113.
-
- 33 mg, 31%.
- 1H NMR (400 MHz, CD3CN) δ ppm 8.07-8.11 (m, 1H), 7.82-7.89 (m, 2H), 7.73 (dd, 1H), 7.65-7.68 (m, 1H), 7.55-7.61 (m, 3H), 7.48-7.53 (m, 1H), 7.40-7.46 (m, 3H), 7.22-7.28 (m, 1H), 7.04-7.11 (m, 1H). MS m/z M−H 423.
-
- 36 mg, 32%.
- 1H NMR (400 MHz, CD3CN) δ ppm 7.96-8.00 (m, 1H), 7.81 (ddd, 1H), 7.74 (dd, 1H), 7.68 (ddd, 1H), 7.65 (dd, 1H), 7.51 (dt, 1H), 7.45 (ddd, 1H), 7.28 (dd, 1H), 7.20 (d, 1H), 7.06-7.12 (m, 1H), 7.04 (d, 1H), 1.85 (s, 3H). MS m/z M−H 451.
-
- 48 mg, 46%.
- 1H NMR (400 MHz, Acetone-d6) δ ppm 8.25 (t, 1H), 8.07-8.09 (m, 1H), 8.03 (ddd, 1H), 7.97-8.01 (m, 2H), 7.80-7.85 (m, 2H), 7.77 (dd, 1H), 7.65-7.74 (m, 2H), 7.53 (ddd, 1H), 7.16 (ddd, 1H). MS m/z M−H 412.
-
- 43 mg, 42%.
- 1H NMR (400 MHz, Acetone-d6) δ ppm 8.24-8.25 (m, 1H), 7.96-8.03 (m, 2H), 7.86-7.92 (m, 4H), 7.83 (dd, 1H), 7.77 (dd, 1H), 7.70 (t, 1H), 7.53 (ddd, 1H), 7.13-7.20 (m, 1H). MS m/z M−H 412.
-
- 16 mg, 17%.
- 1H NMR (400 MHz, CD3OD) δ ppm 8.25 (s, 1H), 8.02 (s, 1H), 7.85 (d, 1H), 7.82 (d, 1H), 7.68-7.77 (m, 2H), 7.55 (t, 1H), 7.49 (t, 1H), 7.18 (t, 1H), 4.04 (s, 3H), 4.03 (s, 3H). MS m/z M−H 449.
-
- 68 mg, 72%.
- 1H NMR (400 MHz, Acetone-d6) δ ppm 8.09-8.17 (m, 2H), 7.78-7.89 (m, 3H), 7.72 (dd, 1H), 7.67 (t, 1H), 7.57 (dt, 1H), 7.52 (ddd, 1H), 7.16 (ddd, 1H), 6.94 (dd, 1H). ). MS m/z M−H 377.
-
- 78 mg, 73%.
- 1H NMR (400 MHz, CD3OD) δ ppm 8.10 (t, 1H), 7.77-7.88 (m, 3H), 7.76 (d, 1H), 7.71 (d, 1H), 7.53 (t, 1H), 7.47 (d, 1H), 7.45 (d, 1H), 7.33 (dd, 1H), 7.27 (d, 1H), 7.14 (t, 1H), 6.52 (dd, 1H). MS m/z M−H 426.
-
- 65 mg, 55%.
- 1H NMR (400 MHz, CD3OD) δ ppm 8.06 (t, 1H), 7.89 (dd, 1H), 7.79-7.85 (m, 2H), 7.74 (d, 1H), 7.42-7.62 (m, 5H), 7.31 (d, 1H), 7.15 (t, 1H). MS m/z M−H 471.
-
- Indan-5-sulfonyl chloride (43 mg, 0.2 mmol) was added to a pyridine stock solution of 1.34M 2-amino-benzenesulfonamide (0.150 mL, 0.2 mmol). The reaction mixture was shaken overnight followed by solvent removal under vacuo. The crude mixture was dissolved in DMSO and purified by preparative HPLC (XTerra MS C8 column, acetonitrile/ammonium acetate buffer) to give the title compound (22 mg, 32%) as a white solid. MS m/z M−H 351. Rf 0.91 min (see above system in General procedure for Examples 23-43).
- The compounds of Examples 123 to 131 were prepared using the general method described for Example 122.
-
- 31 mg, 41%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.14 (s, 1H), 7.82 (s, 2H), 7.77 (d, 1H), 7.45-7.56 (m, 2H), 7.18 (ddd, 1H), 7.14 (dd, 1H), 7.10 (d, 1H), 6.77 (d, 1H), 4.20-4.28 (m, 4H), 2.82 (s, 3H). MS m/z M−H 382.
-
- 27 mg, 30%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.40 (br. s., 1H), 7.90-8.05 (m, 5H), 7.76-7.86 (m, 3H), 7.74 (s, 2H), 7.54 (br. s., 2H), 7.16-7.28 (m, 1H). MS m/z M−H 455.
-
- 67 mg, 66%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.40 (br. s., 1H), 8.62 (d, 1H), 8.53-8.55 (m, 1H), 8.02 (d, 2H), 7.83 (br. s, 3H), 7.53-7.60 (m, 2H), 7.48 (d, 2H), 7.22-7.32 (m, 1H). MS m/z M−H 506.
-
- 48 mg, 65%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.28 (s, 1H), 7.82 (s, 2H), 7.77 (d, 1H), 7.65 (s, 1H), 7.60 (dd, 1H), 7.47-7.52 (m, 2H), 7.24 (d, 1H), 7.16-7.22 (m, 1H), 2.72-2.77 (m, 4H), 1.68-1.73 (m, 4H). MS m/z M−H 365.
-
- 45 mg, 54%.
- MS m/z M−H 417. Rf 1.1 min (see above system in General procedure for Examples 23 to 43).
-
- 45 mg, 57%.
- MS m/z M−H 393. RF 1.12 min (see above system in General procedure for Examples 23 to 43).
-
- 35 mg, 46%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.36 (br. s., 1H), 8.12 (s, 1H), 7.95 (d, 1H), 7.89 (d, 1H), 7.81 (br. s., 2H), 7.78 (d, 1H), 7.63-7.70 (m, 3H), 7.47-7.60 (m, 4H), 7.39-7.45 (m, 1H), 7.19-7.27 (m, 1H). MS m/z M−H 387.
-
- 50 mg, 53%.
- MS m/z M−H 465. Rf 0.41 min (see above system in General procedure for Examples 23 to 43).
-
- 16 mg, 22%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.48 (br. s., 1H), 8.07 (d, 2H), 7.92-8.02 (m, 2H), 7.67-7.85 (m, 3H), 7.47-7.60 (m, 2H), 7.22-7.35 (m, 1H). MS m/z M−H 379.
-
- Tetrakis(triphenylphosphine)palladium(0) (14 mg, 0.012 mmol) was added to 2,5-dichlorophenylboronic acid (57 mg, 0.30 mmol) in DMF (0.25 mL) and a DMF stock solution of 0.5M 2-[(3-bromophenyl)sulfonylamino]benzenesulfonamide (0.5 mL, 0.25 mmol). The reaction mixture was stirred at RT under argon for 15 minutes before addition of 2M Na2CO3 (0.5 mL). The reaction was stirred overnight at 90° C., filtered and purified by preparative HPLC (XTerra MS C8 column, acetonitrile/ammonium acetate buffer) to give the title compound (15 mg, 13%) as a white solid.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.37 (br. s., 1H), 7.95 (d, 1H), 7.91 (s, 1H), 7.65-7.86 (m, 5H), 7.62 (d, 1H), 7.50-7.59 (m, 3H), 7.48 (d, 1H), 7.25 (t, 1H). MS m/z M−H 455.
- The compounds of Examples 133 to 143 were prepared using the general method described for Example 132.
-
- 38 mg, 32%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.45 (br. s., 1H), 8.43 (s, 1H), 8.20-8.27 (m, 3H), 8.00 (d, 1H), 7.76-7.87(m, 4H), 7.70-7.75 (m, 2H), 7.64 (d, 1H), 7.52-7.60 (m, 3H), 7.46 (t, 1H), 7.24 (t, 1H) MS m/z M−H 477.
-
- 36 mg, 32%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.37 (br. s., 1H), 8.20 (s, 1H), 8.03 (d, 1H), 7.95 (d, 1H), 7.90 (d, 2H), 7.85 (d, 2H), 7.75-7.82 (m, 3H), 7.71 (t, 1H), 7.50-7.62 (m, 2H), 7.24 (t, 1H). MS m/z M−H 455.
-
- 36 mg, 34%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.35 (s, 1H), 8.12 (s, 1H), 7.96 (d, 1H), 7.89 (d, 1H), 7.81 (s, 2H), 7.78 (d, 1H), 7.51-7.69 (m, 3H), 7.40 (t, 1H), 7.16-7.26 (m, 3H), 6.99 (dd, 1H), 3.83 (s, 3H). MS m/z M−H 417.
-
- 25 mg, 24%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.42 (br. s., 1H), 8.38 (s, 1H), 8.19 (d, 1H), 7.89 (d, 1H), 7.75-7.84 (m, 3H), 7.65-7.74 (m, 3H), 7.51-7.65 (m, 3H), 7.37 (t, 1H), 7.29 (t, 1H), 7.20-7.27 (m, 1H). MS m/z M−H 427.
-
- 42 mg, 39%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.35 (s, 1H), 8.06 (s, 1H), 7.87 (d, 1H), 7.81 (br. s., 2H), 7.78 (d, 1H), 7.52-7.66 (m, 5H), 7.39 (dd, 1H), 7.23 (t, 1H), 6.86 (d, 1H), 4.58 (t, 2H), 3.23 (t, 2H). MS m/z M−H 429.
-
- 32 mg, 30%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.35 (br. s., 1H), 8.48 (d, 1H), 8.13 (br. s., 1H), 8.01 (dd, 1H), 7.95 (d, 1H), 7.88 (d, 1H), 7.82 (br. s., 2H), 7.77 (d, 1H), 7.66 (t, 1H), 7.50-7.61 (m, 2H), 7.23 (t, 1H), 6.94 (d, 1H), 3.91 (s, 3H). MS m/z M−H 418.
-
- 22 mg, 19%.
- MS m/z M−H 455. Rf 0.55 min (see above system in General procedure for Examples 23 to 43).
-
- 18 mg, 16%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.38 (br. s., 1H), 7.99 (s, 1H), 7.92 (d, 1H), 7.88 (d, 1H), 7.77-7.83 (m, 3H), 7.71 (t, 1H), 7.58 (d, 1H), 7.54-7.65 (m, 2H), 7.50 (d, 1H), 7.26 (t, 1H), 7.21 (t, 1H), 7.08 (t, 1H), 6.62 (s, 1H), 3.64 (s, 3H). MS m/z M−H 440.
-
- 40 mg, 36%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.35 (br. s., 1H), 8.19 (s, 1H), 8.04 (d, 1H), 7.90-8.00 (m, 3H), 7.65-7.83 (m, 6H), 7.57-7.63 (m, 1H), 7.53 (t, 1H), 7.23 (t, 1H). MS m/z M−H 455.
-
- 13 mg, 12%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.39 (br. s., 1H), 8.09 (d, 2H), 8.00 (d, 2H), 7.75-7.84 (m, 3H), 7.71 (d, 1H), 7.62-7.67 (m, 2H), 7.55 (br. s., 2H), 7.37 (t, 1H), 7.29 (t, 1H), 7.21-7.26 (m, 1H). MS m/z M−H 427.
-
- 12 mg, 11%.
- MS m/z M−H 423. Rf 0.59 min (see above system in General procedure for Examples 23 to 43).
-
- 2-Amino-5-bromobenzenesulfonamide (0.60 g, 2.39 mmol) was dissolved in pyridine (6 ml) and 3-(3,4-dichlorophenyl)-phenylsulfonyl chloride (0.82 g, 2.51 mmol) was added and the reaction stirred overnight. The solvent was removed in vacuo and the residue was purified by preparative HPLC (XTerra MS C8 column, acetonitrile/ammonium acetate buffer) to give the title compound (0.70 g, 55% yield).
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.35 (br. s., 1H), 8.19 (t, 1H), 8.03 (d, 1H), 7.97 (d, 1H), 7.92 (s, 2H), 7.85-7.91 (m, 2H), 7.77 (d, 1H), 7.65-7.76 (m, 3H), 7.54 (d, 1H);
- MS m/z M−H 533, 535, 537.
-
- 5-Bromo-2-[[3-(3,4-dichlorophenyl)phenyl]sulfonylamino]benzenesulfonamide (70 mg, 0.13 mmol) was dissolved in DMF (700 μl), then phenylboronic acid (20 mg, 0.16 mmol) was added followed by the addition of 2M sodium carbonate solution (260 μl). The mixture was subjected to vacuum/argon (×3), tetrakis(triphenylphosphine)palladium (8 mg, 0.05 mol %) was added and the reaction was allowed to stir at 80° C. overnight.
- The cooled reaction mixture was filtered and was then purified by preparative HPLC (XTerra MS C8 column, acetonitrile/ammonium acetate buffer) to give the title compound (23 mg, 32%).
- 1H NMR (400 MHz, CD3OD) δ ppm 8.07 (d, 1H), 8.04 (t, 1H), 7.91 (ddd, 1H), 7.85 (d, 1H), 7.81 (ddd, 1H), 7.76-7.79 (m, 1H), 7.72 (d, 1H), 7.59 (d, 1H), 7.53-7.58 (m, 3H), 7.48 (dd, 1H), 7.38-7.44 (m, 2H), 7.31-7.36 (m, 1H); MS m/z M−H 531, 533.
-
- 2-Aminobenzenesulfonamide (26 mg, 0. 15 mmol) was dissolved in pyridine (1 ml) and 2,3-difluorophenylsulfonyl chloride (19 μl, 0.15 mmol) was added and the reaction stirred overnight. The solvent was removed in vacuo and the residue was purified by preparative HPLC (XTerra MS C8 column, acetonitrile/ammonium acetate buffer) to give the title compound (30 mg, 58%).
- 1H NMR (400 MHz, CD3OD) δ ppm 6.77-6.85 (m, 1H) 7.12-7.27 (m, 2H) 7.31-7.45 (m, 2H) 7.64-7.78 (m, 2H); MS m/z M−H 347.
-
- 2-Amino-5-fluorobenzenesulfonamide (29 mg, 0.15 mmol) was dissolved in pyridine (1 ml) and 2,3-difluorophenylsulfonyl chloride (19 μl, 0.15 mmol) was added and the reaction stirred overnight. The solvent was removed in vacuo and the residue was purified by preparative HPLC (XTerra MS C8 column, acetonitrile/ammonium acetate buffer) to give the title compound (25 mg, 46%).
- 1H NMR (400 MHz, CD3OD) δ ppm 6.98-7.07 (m, 1H) 7.12-7.22 (m, 1H) 7.30-7.41 (m, 1H) 7.42-7.49 (m, 2H) 7.63-7.71 (m, 1H); MS m/z M−H 364.
-
- 2-Aminobenzenesulfonamide (54 mg, 0.31 mmol) and 3-chloro-2-fluoro-benzenesulfonyl chloride (47 mg, 0.21 mmol) were dissolved in pyridine (1 ml) and the reaction mixture was stirred for 12 h. The solvent was removed in vacuo and the residue was purified by HPLC to give the product (15 mg, 19%).
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.72 (br. s., 1H), 7.90 (t, 1H), 7.78-7.86 (m, 2H), 7.73 (br. s., 2H), 7.51-7.58 (m, 1H), 7.46-7.51 (m, 1H), 7.39 (t, 1H), 7.26-7.34 (m, 1H);
- MS m/z M−H 363.
-
- The title compound (56%) was synthesized using the general method described for Example 148.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.88 (br. s., 1H), 8.19 (d, 1H), 7.99 (d, 1H), 7.80-7.92 (m, 3H), 7.63 (t, 1H), 7.48 (t, 1H), 7.31 (d, 1H), 7.24 (t, 1H); MS m/z M−H 379, 381.
-
- To a solution of 2-(3-bromobenzenesulfonylamino)-5-methyl-benzensulfonamide (1.50 g, 3.70 mmol), 3,4-dichlorobenzeneboronic acid (0.84 g, 4.44 mmol), tricyclohexyl phosphine (0.45 g, 1.60 mmol) and potassium phosphate (3 g, 14.15 mmol) in DMF (10 mL), was added Pd(OAc)2 (0.17 g, 0.74 mmol). The reaction mixture was stirred at 80° C. for 10 h, diluted with 1N HCl and the product was extracted with ethyl acetate. The organic phase was separated, dried over sodium sulfate and concentrated in vacuo. The crude product was purified by flash column chromatography using ethyl acetate/hexane (1:3) to afford the title compound (0.9 g, 51%).
- 1H NMR (400 MHz, DMSO-d6): δ ppm 9.23 (s, 1H), 8.15 (s, 1H), 8.02 (d, 1H), 7.95 (d, 1H), 7.89 (d, 1H), 7.78-7.72 (m, 3H), 7.70-7.65 (m, 2H), 7.60 (d, 1H), 7.50 (d, 1H), 7.36 (d, 1H), 2.22 (s, 3H);
- ESMS: m/z [M−1]468.91 and 470.86 (Cl isotopes).
- To a solution of 2-amino-5-methyl-benzenesulfonamide (Example 45a) (6.0 g, 32.3 mmol) in pyridine (20 mL), 3-bromobenzenesulphonyl chloride (8.24 g, 32.3 mmol) was added portionwise at 0° C. The reaction was stirred at RT for 10 h. The pyridine was removed in vacuo at 35° C. and the residue was dissolved in ethyl acetate. The organic phase was washed with aqueous 1N HCl, water and brine, dried over sodium sulfate, and concentrated under reduced pressure. The crude product was purified by flash column chromatography using ethyl acetate/hexane (1:1) as eluent to give the title compound (8 g, 63%).
- 1H NMR (400 MHz, DMSO-d6); δ ppm 9.31 (s, 1H), 8.01 (s, 1H), 7.89-7.81 (m, 2H), 7.70 (s, 2H), 7.61 (s, 1H), 7.58-7.50 (m, 1H), 7.41-7.34 (m, 2H), 2.22(s, 3H);
- ESMS: m/z [M−1]402.93 and 404.95 (Br isotopes).
-
- 2-[[3-(3,4-Dichlorophenyl)phenyl]sulfonylamino]-5-methylbenzene sulfonamide (Example 150) (0.5 g, 1.06 mmol) was dissolved in 1N NaOH (50 mL). KMnO4 (1 g, 6.33 mmol) was added and the reaction mixture was stirred at 80° C. for 4 h, cooled to RT and filtered through Celite. The filtrate was treated with sodium bisulphite (3 g) until a colourless solution was obtained. The reaction mixture was then acidified to pH 3 by addition of 3N HCl. The resulting white precipitate was filtered off and washed with water to give a crude product which was used in the next step without further purification.
- To a solution of 4-[[3-(3,4-dichlorophenyl)phenyl]sulfonylamino]-3-sulfamoylbenzoic acid (0.20 g, 0.39 mmol) in DMF (5 mL) was added carbonyldiimidazole (0.06 g, 0.39 mmol). The reaction mixture was heated at 80° C. for 2h, then aqueous ammonia solution (30%, 2 mL) was added and the resulting solution was stirred at RT for 2 h. The pH of the reaction mixture was then adjusted to pH 5 using 6N HCl and the resulting white precipitate was collected by filtration. The crude product was purified by preparative HPLC to afford the title compound (13 mg, 7%).
- 1H NMR (400 MHz, acetone-D6): δ ppm 9.47 (br, 1H), 8.38 (d, 1H), 8.27 (s, 1H), 8.08-8.00 (m, 3H), 7.93 (s, 1H), 7.82 (d, 1H), 7.74-7.70 (m, 3H), 7.65 (s, 1H), 7.26 (s, 2H), 6.79 (s, 1H);
- ESMS: m/z [M−1]499.72.
-
- Pd(OAc)2 (44 mg, 0.19 mmol) was added to a solution of 2-(3-bromobenzenesulfonylamino)-5-methyl-benzensulfonamide (Example 150 a) (0.40 g, 0.98 mmol), 3-(trifluoromethoxy)benzeneboronic acid (0.61 g, 2.96 mmol), tricyclohexyl phosphine (82 mg, 0.29 mmol) and potassium phosphate (0.42 g, 1.19 mmol) in toluene (10 mL). A few drops of water were added and the reaction mixture was stirred at 110° C. overnight. The reaction mixture was then diluted with 1N HCl and the product was extracted with ethyl acetate. The organic phase was separated, dried over sodium sulfate and concentrated in vacuo. The crude product was purified by flash column chromatography using ethyl acetate /hexane (1:3) to afford the title compound (140 mg, 30%).
- 1H NMR (400 MHz, DMSO-d6): δ ppm 9.25 (s, 1H), 8. 10 (s, 1H), 7.98 (d, 1H), 7.87 (d, 1H), 7.54-7.81 (m, 7H), 7.49 (d, 1H) 7.42 (d, 1H), 7.34 (d, 1H), 2.23 (s, 3H);
- 19F NMR (400 MHz, DMSO-d6): δ ppm −57.12;
- ESMS: m/z 484.79 [M−1].
-
- Lithium aluminum hydride (1M in THF, 6.6 mL, 6.6 mmol) was added slowly to a stirred solution of 4-(2,3-dichloro-benzenesulfonylamino)-3-sulfamoyl-benzoic acid ethyl ester (600 mg, 1.32 mmol) in dry THF (15 mL) at −10° C. The reaction mixture was allowed to warm to RT and stirring was continued for 4 h. The reaction mixture was then cooled to 0° C. and quenched by addition of water, followed by 1N HCl. The product was extracted with ethyl acetate, the combined organic phases were washed with water and brine, and dried over sodium sulfate. Volatiles were removed under reduced pressure and the crude product was purified by preparative HPLC to afford the title compound (60 mg, 11%).
- 1H NMR (400 MHz, DMSO-D6): δ ppm 9.86 (br. s., 1H), 8.10-8.30 (m, 1H), 7.99 (d, 1H), 7.84 (m, 3H), 7.62 (dd, 1H), 7.39 (d, 1H), 7.28 (d, 1H), 4.44 (s, 2H), 1.84-2.22 (m, 1H);
- ESMS: m/z [M−1]408.79 and 410.75 (Cl isotopes).
- A solution of 4-(2,3-dichloro-benzenesulfonylamino)-3-sulfamoyl-benzoic acid (1.3 g, 3.05 mmol) in ethanol (20 mL) was stirred at RT and a few drops of thionyl chloride were added. The reaction mixture was heated at reflux for 7 h, cooled to RT and concentrated under reduced pressure. The residue was purified by flash column chromatography using ethyl acetate/hexane (1:1) as eluent to afford the title compound (1.4 g, 100%).
- 1H NMR (400 MHz, DMSO-d6); δ ppm 8.41 (d, 1H), 8.19-8.31 (m, 1H), 8.00-8.17 (m, 4H), 7.66 (dd, 1H), 7.48 (d, 1H), 4.30 (q, 2H), 1.29 (t, 3H).
- 2-(2,3-Dichloro-benzenesulfonylamino)-5-methyl-benzenesulfonamide (Example 154) (186.1 mg, 0.47 mmol) was dissolved in 1N NaOH (2 mL) and water (10 mL). KMnO4 (0.24 g, 1.52 mmol) was added and the reaction mixture was stirred at 70° C. for 3 h, cooled to RT and filtered through Celite. The filtrate was treated with sodium bisulphite until a colourless solution was obtained and the mixture was then acidified to pH 3 by addition of 3N HCl. The product was extracted with ethyl acetate and the organic phase was concentrated under reduced pressure. The crude product was purified by preparative HPLC to afford the title compound (124 mg, 62%).
- 1H NMR (400 MHz, DMSO-D6): δ ppm 8.41 (d, 1H), 8.24 (d, 1H), 7.93-8.11 (m, 4H), 7.66 (dd, 1H), 7.45 (d, 1H);
- ESMS: m/z [M−1]: 422.72 and 424.67.
-
- To a solution of 2-amino-5-methyl-benzenesulfonamide (1.86 g, 1 mmol) in pyridine (20 mL), 2,3-dichlorobenzenesulphonyl chloride (3.0 g, 1.25 mmol) was added at 0° C. The reaction was stirred at RT for 17 h. The pyridine was removed in vacuo and the residue was dissolved in ethyl acetate. The organic phase was washed with aqueous 1N HCl, water and brine, dried over sodium sulfate, and concentrated under reduced pressure. The crude product was purified by flash column chromatography using ethyl acetate/hexane (1:1) as eluent to give the title compound (2.4 g, 60%).
- 1H NMR (400 MHz, DMSO-D6): δ ppm 9.77 (br. s., 1H), 8.02-8.25 (m, 1H), 7.95 (dd, 1H), 7.77 (s, 2H), 7.47-7.73 (m, 2H), 7.04-7.36 (m, 2H), 2.23 (s, 3H);
- ESMS: m/z [M−1]: 392.85 and 394.67 (Cl isotopes).
-
- To a solution of 2-(3-bromophenylsulfonamido)-5-phenoxybenzenesulfonamide (241.5 mg, 0.5 mmol), 3,4-dichlorobenzeneboronic acid (190.8 mg, 1.0 mmol), tricyclohexyl phosphine (50 mg, 0.18 mmol) and potassium phosphate (340 mg, 1.6 mmol) in DMF (10 mL), was added Pd(OAc)2 (20 mg, 0.09 mmol). The reaction mixture was stirred at 80° C. for 7 h, diluted with 1N HCl and the product was extracted with ethyl acetate. The organic phase was separated, dried over sodium sulfate and concentrated in vacuo. The crude product was purified by flash column chromatography using ethyl acetate/hexane (1:1) to afford the title compound (42 mg, 15%).
- 1H NMR (400 MHz, DMSO-d6): δ ppm 9.26 (s, 1H), 8.14 (s, 1H), 8.02 (s, 1H), 7.97 (d, 1H), 7.88 (d, 1H), 7.73-7.81 (m, 3H), 7.64-7.73 (m, 2H), 7.56 (d, 1H), 7.39 (dd, 2H), 7.34 (d, 1H), 7.13-7.26 (m, 2H), 6.99 (d, 2H);
- ESMS: m/z [M−1]546.80 and 548.69 (Cl isotopes).
- To a solution of 2-amino-5-phenoxybenzenesulfonamide (528 mg, 2 mmol) in pyridine (15 mL), 3-bromobenzenesulphonyl chloride (669.3 mg, 2.3 mmol) was added portionwise at 0 ° C. The reaction was stirred at RT overnight. The pyridine was removed in vacuo and the residue was dissolved in ethyl acetate. The organic phase was washed with aqueous 1N HCl, water and brine, dried over sodium sulfate, and concentrated under reduced pressure. The crude product was purified by flash column chromatography using ethyl acetate/hexane (1:3) as eluent to give the title compound (700 mg, 72%).
- 1H NMR (400 MHz, DMSO-d6); δ (ppm 9.30 (br s, 1H), 7.94 (s, 1H), 7.76-7.91 (m, 2H), 7.71 (s, 2H), 7.52 (dd, 1H), 7.48 (d, 1H), 7.41 (dd, 2H), 7.35 (d, 1H), 7.13-7.27 (m, 2H), 7.01 (d, 2H); ESMS: m/z [M−1]480.88 and 482.83 (Br isotopes).
- A mixture of 7-phenoxy-1,1-dioxo-1,4-dihydro-2-H-benzo[1,2,4]thiadiazin-3-one (18.0 g, 61.8 mmol) in aqueous H2SO4 (50% v/v, 1 L) was heated at 105° C. for 7 h. The clear solution was then cooled to 0° C. and neutralized using 5N NaOH. The product was extracted with ethyl acetate. The combined extracts were washed with water and concentrated under reduced pressure. The crude product was purified by flash column chromatography using ethyl acetate/hexane (1:1) to afford the title compound (4.0 g, 24%).
- 1H NMR (400 MHz, DMSO-d6): δ ppm 7.27-7.41 (m, 4H), 7.21 (d, 1H), 7.07 (m, 2H), 6.91 (d, 2H), 6.85 (d, 1H), 5.76 (s, 2H).
- 4-Phenoxybenzenamine (18.4 g, 101.1 mmol) dissolved in nitromethane (30 mL) was added slowly over 25 minutes to a solution of chlorosulfonyl isocyanate (16.4 g, 115.8 mmol) in nitromethane (100 mL) at −5° C. The suspension was stirred at −5° C. for an additional 15 minutes and AlCl3 (16.8 g, 125.9 mmol) was added portionwise. The reaction mixture was then heated at 105° C. for 30 minutes. The hot solution was poured onto ice (1L) and the resulting precipitate was filtered off and washed with water (300 mL). The solid was then dissolved in hot aqueous sodium bicarbonate solution (10 g/200 mL), treated with charcoal, and filtered. The cooled solution was neutralized using 6N HCl and the resulting white precipitate was filtered off, washed with water and dried to afford the title compound (18 g, 62%).
- 1H NMR (400 MHz, DMSO-d6): δ ppm 11.21 (s, 1H), 7.40-7.47 (m, 2H), 7.38 (dd, 1H), 7.26-7.31 (m, 2H), 7.17-7.25 (m, 2H), 7.07 (d, 2H).
-
- To a solution of 2-(3-bromophenylsulfonamido)-5-phenoxybenzenesulfonamide (Example 155a) (150.0 mg, 0.31 mmol), 3-(trifluoromethoxy)benzeneboronic acid (192 mg, 0.93 mmol), tricyclohexyl phosphine (25 mg, 0.09 mmol) and potassium phosphate (131 mg, 0.62 mmol) in DMF (10 mL), was added Pd(OAc)2 (14 mg, 0.06 mmol). The reaction mixture was stirred at 80° C. overnight, diluted with 1N HCl and the product was extracted with ethyl acetate. The organic phase was separated, dried over sodium sulfate and concentrated in vacuo. The crude product was purified by flash column chromatography using methanol/DCM (1:99) to afford the title compound (92 mg, 53%).
- 1H NMR (400 MHz, CD3OD) δ ppm 8.00 (s, 1H), 7.82-7.88 (m, 2H), 7.75 (d, 1H), 7.52-7.64 (m, 3H), 7.49 (s, 1H), 7.41 (d, 1H), 7.33 (dd, 3H), 7.09-7.18 (m, 2H), 6.93 (d, 2H); 19F NMR (400 MHz, CD3OD) δ ppm −59.72; ESMS: m/z [M−1]562.92.
-
- To a solution of 2-(2,3-dichloro-benzenesulfonylamino)-5-methoxy-benzenesulfonamide (4.8 g, 11.68 mmol) in DCM (200 mL) at −20° C., BBr3 (1M in DCM, 60 mL, 60 mmol) was added dropwise over 30 minutes. The reaction mixture was allowed to warm to RT and the stirring was continued overnight. The reaction mixture was then cooled to −20° C., quenched by addition of methanol (20 mL) and volatiles were removed under reduced pressure. The residue was partitioned between water (50 mL) and ethyl acetate (100 mL). The organic phase was washed with water, dried over sodium sulfate and concentrated under reduced pressure. The crude product was recrystallized from methanol to afford the title compound (4.6 g, 65%).
- 1H NMR (400 MHz, DMSO-d6): δ ppm 10.04 (s, 1H), 9.52 (s, 1H), 7.86-8.12 (m, 2H), 7.41-7.70 (m, 3H), 7.25 (d, 1H), 7.08 (d, 1H), 6.88 (dd, 1H);
- ESMS: m/z [M−1]394.80 and 396.76 (Cl isotopes).
- To a solution of 2-amino-5-methoxy-benzenesulfonamide (2.73 g, 13.43 mmol) in pyridine (25 mL), 2,3-dichlorobenzenesulfonyl chloride (3.68 g, 15.0 mmol) was added slowly at 0° C. The reaction mixture was stirred at RT overnight and then concentrated under reduced pressure. The residue was then partitioned between ethyl acetate and 1N HCl. The organic layer was separated, dried over sodium sulfate and concentrated in vacuo. The crude product was purified by flash column chromatography using ethyl acetate/hexane (1:1) to afford the title compound (4.8 g, 87%).
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.64 (br. s., 1H), 8.03 (d, 1H), 7.94 (d, 1H), 7.71 (s, 2H) 7.55 (dd, 1H), 7.35 (d, 1H), 7.21 (d, 1H), 7.01-7.17 (m, 1H), 3.73 (s, 3H).
-
- To a solution of 3-chloro-4-[[(2R)-3,3,3-trifluoro-2-hydroxy-2-methyl-propanoyl]amino]benzenesulfonyl chloride (110 mg, 0.3 mmol) in DCM (5 ml) and pyridine (30 μl, 0.33 mmol) was added DMAP (˜2 mg) followed by 2-aminobenzenesulfonamide (52 mg, 0.3 mmol). The resulting mixture was stirred at RT for 18 h. The solution was concentrated and partitioned between ethyl acetate (6 ml) and 1M HCl (5 ml). The organic layer was separated, washed with saturated NaHCO3, dried (MgSO4) and evaporated to give a gum which was dissolved in a small amount of ethyl acetate and then iso-hexane was added to precipitate the title compound as a solid.
- LCMS 502.2 (M+H), Rf 7.64 min. [Column: HYPERSIL ODS; Flow Rate: 1.5 ml /min; Detector Wavelength: 254 nm; Solvent A: 0.1% Formic Acid in Water; Solvent B: 0.1% Formic Acid in Acetonitrile; Gradient: 5-95% B]
-
- To a solution of 2-aminobenzenesulfonamide (172 mg) in pyridine (3 ml) was added 5-chlorothiophene-2-sulfonyl chloride (217 mg) and the resulting mixture was heated at 60° C. for 18 h. The mixture was concentrated and then partitioned between DCM (20 ml) and 2N HCl (10 ml). The organics were separated, dried (MgSO4) and evaporated to a gum which was purified by silica chromatography eluting with a gradient of ethyl acetate/hexane to give the title compound as a solid (201 mg).
- LCMS 351, 353 (M−H, Cl pattern); 1H NMR (400 MHz, DMSO-d6) δ 7.35 (d, 1H), 7.35 (m, 2H), 7.6 (m, 2H), 7.75 (s, 2H), 7.85 (d, 1H, 9.5 (bs, 1H).
-
- The title compound was prepared using the general method described in Example 159 but using 4,5-dichlorothiophene-2-sulfonyl chloride.
- LCMS 386, 388, 390 (M−H, Cl2 pattern); 1H NMR (400 MHz, DMSO-d6) δ 7.35 (m, 1H), 7.55-7.9 (m, 6H), 9.6 (bs, 1H).
-
- The title compound was prepared using the general method described in Example 159 but using 3,4-dichlorophenylsulfonyl chloride.
- LCMS 380, 382, 384 (M−H, Cl2 pattern); 1H NMR (400 MHz, DMSO-d6) δ 7.3 (t, 1H), 7.55 (m, 2H), 7.70 (s, 2H), 7.82 (m, 3H), 8.09 (s, 1H), 9.5 (bs, 1H).
-
- 1,1′-Bis(diphenylphosphino)ferrocene palladium dichloride methylene chloride adduct (14 mg, 0.02 mmol) was added to a solution of 6-methoxy pyridine-2-boronic acid pinacol ester (135 mg, 0.57 mmol), 2-[(3-bromophenyl)sulfonylamino]benzenesulfonamide (150 mg, 0.38 mmol) (see Example 113a) and Na2CO3 (0.18 mg, 1.72 mmol) in THF:water, 10:1 (4 mL). The reaction mixture was heated in a microwave reactor at 120° C. for 20 min. Some DCM was added followed by brine and the aqueous layer was extracted twice with DCM. The combined organic layers were dried with Na2SO4 and after filtration and evaporation the crude was purified by preparative HPLC (XTerra MS C8 column, acetonitrile/ammonium acetate buffer) to give the title compound (30 mg, 19%) as a yellow oil.
- 1H NMR (400 MHz, CD3OD) δ ppm 8.57 (t, 1H), 8.22 (ddd, 1H), 7.92 (ddd, 1H), 7.79 (dd, 1H), 7.69 (dd, 1H), 7.67 (dd, 1H), 7.56 (t, 1H), 7.43 (dd, 1H), 7.40 (ddd, 1H), 7.00-7.10 (m, 1H), 6.73 (d, 1H), 4.95 (br. s., 2H), 1.94 (s, 3H). MS m/z M+H 420, M−H 418.
-
- To 2-aminobenzenesulsulfonamide (53 mg, 0.308 mmol) in pyridine (1 mL) was added 4-(2-Chloro-thiazol-5-ylmethoxy)-benzenesulfonyl chloride (100 mg, 0.308 mmol). The reaction mixture was stirred for 40 h. Some conversion was observed but much solid was formed. More 2-aminobenzenesulsulfonamide (53 mg, 0.308 mmol) and DCM (1 mL) were added. The reaction mixture was stirred overnight. After removal of the pyridine by blowing with nitrogen gas, the residue was purified by preparative HPLC (XTerra MS C8 column, acetonitrile/ammonium acetate buffer) to give the title compound (30 mg, 21%) as an off-white solid.
- 1H NMR (400 MHz, CD3OD) δ ppm 7.81-7.87 (m, 2H), 7.80 (dd, 1H), 7.60-7.67 (m, 2H), 7.43 (ddd, 1H), 7.12 (t, 1H), 7.05-7.09 (m, 2H), 5.30 (s, 2H).
- MS m/z M+H 460/462, M−H 458/460.
- The compounds of Examples 164 to 172 were prepared using the general method described for Example 113.
-
- 33 mg, 31%.
- 1H NMR (400 MHz, CD3OD) δ ppm 8.15 (d, 1H), 8.04 (t, 1H), 7.85 (ddd, 1H), 7.79-7.83 (m, 2H), 7.76 (d, 1H), 7.75 (dd, 1H), 7.57 (t, 1H), 7.46-7.53 (m, 1H), 7.18 (t, 1H), 4.04 (s, 3H). MS m/z M+H 438, M−H 436.
-
- 35.1 mg, 83%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.37 (br s, 1H), 8.93 (s, 2H), 8.23 (br s, 1H), 7.45-8.08 (m, 8H), 7.23 (br s, 1H), 3.98 (s, 3H). MS m/z M+H 421, M−H 419.
-
- 32.8 mg, 43%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.40 (br s, 1H), 8.50 (br s, 1H), 8.35 (br s, 1H), 7.92 (ddd, 1H), 7.72-7.83 (m, 4H), 7.65-7.71 (m, 2H), 7.59 (dd, 1H), 7.53 (ddd, 1H), 7.35 (d, 1H), 7.23 (t, 1H), 2.10 (s, 3H). MS m/z M+H 404, M−H 402.
-
- 7 mg, 9%.
- 1H NMR (400 MHz, CD3OD) δ ppm 8.15 (dd, 1H), 8.06 (t, 1H), 7.85 (ddd, 1H), 7.83 (dd, 1H), 7.77 (ddd, 1H), 7.66-7.73 (m, 2H), 7.54 (t, 1H), 7.48 (ddd, 1H), 7.13-7.19 (m, 1H), 7.05 (dd, 1H), 3.91 (s, 3H). MS m/z M+H 420, M−H 418.
-
- 30 mg, 37%.
- 1H NMR (400 MHz, CD3OD) δ ppm 8.70 (s, 1H), 8.57 (s, 1H), 8.06-8.16 (m, 2H), 7.93 (d, 1H), 7.86 (d, 1H), 7.80 (dd, 1H), 7.73 (d, 1H), 7.60 (t, 1H), 7.46 (dt, 1H), 7.12 (t, 1H). MS m/z M+H 423/425, M−H 421/423.
-
- 39 mg, 45%.
- 1H NMR (600 MHz, DMSO-d6) δ ppm 9.35 (br. s., 1H), 8.46 (d, 1H), 8.25 (d, 1H), 8.19 (br. s., 1H), 7.99 (dd, 1H), 7.91 (d, 1H), 7.83 (br. s., 2H), 7.77 (d, 1H), 7.66 (t, 1H), 7.57-7.60 (m, 1H), 7.51-7.56 (m, 1H), 7.15-7.28 (m, 1H), 3.98 (s, 3H).
- MS m/z M+H 454/456, M−H 452/454.
-
- 33 mg, 40%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.35 (br. s., 1H), 8.42 (d, 1H), 8.06 (s, 1H), 7.87 (d, 1H), 7.82 (dd, 1H), 7.72-7.82 (m, 4H), 7.46-7.63 (m, 3H), 7.09-7.28 (m, 1H), 6.73 (d, 1H), 3.07 (s, 6H). MS m/z M+H 433, M−H 431.
-
- 35 mg, 44%.
- 1H NMR (400 MHz, CD3OD) δ ppm 8.09 (t, 1H), 7.86 (d, 1H), 7.78-7.83 (m, 2H), 7.71 (d, 1H), 7.51-7.58 (m, 2H), 7.35-7.50 (m, 4H), 7.12 (t, 1H), 4.67 (s, 2H).
- MS m/z M−H 417.
-
- 28 mg, 35%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.38 (br. s, 1H), 8.13 (br. s., 1H), 7.96 (d, 1H), 7.89 (d, 1H), 7.83 (br. s., 2H), 7.79 (d, 1H), 7.63 (d, 2H), 7.51-7.69 (m, 3H), 7.44 (d, 2H), 7.24 (t, 1H), 5.27 (t, 1H), 4.56 (d, 2H). MS m/z M−H 417.
- The compounds of Examples 173 to 176 were prepared using the general method described for Example 163.
-
- 117 mg, 64%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.41 (s, 1H), 7.97-8.05 (m, 3H), 7.91-7.97 (m, 2H), 7.75-7.86 (m, 3H), 7.74 (s, 2H), 7.48-7.59 (m, 2H), 7.18-7.28 (m, 1H).
- MS m/z M−H 455/457/459.
-
- 135 mg, 74%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.35 (s, 1H), 8.19 (t, 1H), 8.01 (d, 1H), 7.96 (d, 1H), 7.93 (d, 1H), 7.81 (s, 2H), 7.77 (d, 1H), 7.76 (d, 1H), 7.64-7.70 (m, 2H), 7.50-7.60 (m, 2H), 7.24 (t, 1H). MS m/z M−H 455/457/459.
-
- 154 mg, 88%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.29 (s, 1H), 7.90 (d, 2H), 7.80 (s, 2H), 7.79 (d, 1H), 7.45-7.57 (m, 4H), 7.24 (ddd, 1H), 7.13-7.19 (m, 2H), 7.10 (d, 2H). M−H 437/439.
-
- 131 mg, 66%.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.44 (s, 1H), 9.12 (s, 1H), 8.96 (s, 1H), 7.96 (d, 1H), 7.90 (s, 2H), 7.80 (dd, 1H), 7.52 (dd, 1H), 7.44-7.50 (m, 3H), 7.37-7.43 (m, 2H), 7.30-7.36 (m, 2H), 7.17 (ddd, 1H), 2.55 (s, 3H). M+H 495/497 M−H 493/495.
-
- Hydrogen peroxide (35%, 1.2 ml) was added to a suspension of N-methyl-N′-(2-sulfamoylphenyl)dibenzo[b,d]thiophene-2,8-disulfonamide (160 mg) in acetic acid (1.8 ml) and the mixture was heated at 100° C. for 1 h. The mixture was diluted with water (5 ml), the solid was recovered by filtration, washed with water and dried to give the title compound (86 mg, 51%).
- 1H NMR (400 MHz), DMSO-d6) δ 2.47 (s, 3H), 7.27 (brs, 1H), 7.53 (m, 2H), 7.73 (m, 4H), 8.03 (d, 1H), 8.12 (d, 1H), 8.29 (m, 2H), 8.7 (s, 1H), 8.93 (s, 1H), 9.5 (brs, 1H).
- Mass spectrum: EI 542.8.
- 8-(Methylsulfamoyl)dibenzo[b,d]thiophene-2-sulfonyl chloride (200 mg) was added to a solution of 2-aminobenzenesulfonamide (191 mg) in sulfolane (2 ml). The mixture was stirred at RT for 20 h then diluted with 1N hydrochloric acid. The solid was recovered, washed with 1N hydrochloric acid and dried to give the title compound (196 mg, 72%).
- Mass spectrum: EI 511.1.
- N-Methyldibenzo[b,d]thiophene-2-sulfonamide (1.56 g) was added to chlorosulfonic acid (3.27 g) and the mixture was stirred at RT for 5 h. The mixture was then treated with ice cold water, the precipitate was filtered off, washed with cold water and dried to give the title compound (1.6 g, 76%).
- 1HNMR (400 MHz, DMSO-d6) δ 2.49 (s, 3H), 7.53 (brs, 1H), 7.84 (d, 1H), 7.9 (d, 2H), 8.1 (d, 1H), 8.3 (d, 1H), 8.59 (s, 1H), 8.77 (s, 1H).
- A large excess of aqueous methylamine was added to a suspension of dibenzo[b,d]thiophene-2-sulfonyl chloride (2.83 g) in sulfolane (15 ml) and the mixture was stirred at RT for 3 h. The reaction mixture was diluted with water, and the precipitate was recovered, washed with water, dried and purified by silica gel chromatography eluting with a gradient of ethyl acetate:DCM of 0:100 to 10:90 to give the title compound (1.79 g, 64%).
- 1H NMR (400 MHz, DMSO-d6) δ 2.48 (s, 3H), 7.52 (brs, 1H), 7.61 (m, 2H), 7.89 (d, 1H), 8.13 (d, 1H), 8.31 (d, 1H), 8.52 (d, 1H), 8.75 (s, 1H).
- BuLi (2.5M, 10 ml) was added dropwise to a suspension of 2-bromodibenzo[b,d]thiophene (6.31 g) (N. M. Cullinane, J. Chem. Soc. 1936, 1435) in dry ether (80 ml) under argon at −5° C., and the mixture was stirred at this temperature for 15 minutes. This solution was added to a stirred solution of sulfur dioxide (80 ml) in ether (80 ml) at −50° C. over 5 minutes, the mixture was stirred for 15 minutes at −50° C. and the temperature of the reaction mixture was then allowed to reach RT over 1 h. The solvent was evaporated and the residue was dried under vacuum overnight. The lithium salt was suspended in ether (150 ml), sulfuryl chloride (3.24 g) was added at 0° C. and the mixture was stirred at RT for 1 h. The mixture was concentrated to dryness to give the title compound (6.8 g, 100%).
- 1HNMR (400 MHz, DMSO-d6) 7.53 (m, 2H), 7.78 (m, 1H), 8.0 (d, 1H), 8.04 (m, 1H), 8.36 (m, 1H), 8.52 (s, 1H).
- Method A
- Sulfuryl chloride (360 mmol) was added dropwise to vigorously stirred anhydrous DMF (30 mL) at 0° C. while maintaining the temperature below 10° C. The reaction mixture was allowed to warm to RT and stirring was continued for 30 minutes. The appropriate styrene derivative (36 mmol) was then added in one portion and the resulting solution was heated at 90° C. for 2 h. The reaction mixture was cooled, poured onto crushed ice and the product was extracted with ethyl acetate. The organic phase was washed with cold water, dried over sodium sulfate and concentrated under reduced pressure. The crude product was purified by flash column chromatography using 2% ethyl acetate/hexane as eluent, followed by recrystallization from ether/pentane to afford the pure sample.
- The compounds listed in Table 1 were synthesized following this method.
-
TABLE 1 Structure Yield Analytical data 5% 1H NMR (400 MHz, CDCl3) δ ppm 7.88 (d, 1H), 7.56 (d, 1H), 7.52-7.45 (m, 2H), 7.05-6.98 (m, 2H), 3.98 (s, 3H). ESMS m/z [M − 1] 213 for corresponding sulfonic acid (M = 214). CHN for C9H9ClO3S Calc.: C, 46.46; H, 3.90. Found: C, 46.91; H, 4.01. 17% 1H NMR (400 MHz, CDCl3) δ ppm 8.05 (s, 1H), 7.95-7.85 (m, 4H), 7.66-7.57 (m, 3H), 7.37 (d, 1H). ESMS m/z [M − 1] 233 for corresponding sulfonic acid (M = 234). CHN for C12H9ClO2S Calc.: C, 57.03; H, 3.59. Found: C, 57.20; H, 3.64. 40% 1H NMR (400 MHz, CDCl3) δ ppm 7.90 (d, 1H), 7.59 (d, 1H), 7.55-7.48 (m, 1H), 7.06-7.01 (m, 2H). ESMS m/z [M − 1] 219 for corresponding sulfonic acid (M = 220). CHN for C8H5ClF2O2S Calc.: C, 40.26; H, 2.11. Found: C, 40.27; H, 2.28. 39% 1H NMR (400 MHz, CDCl3) δ ppm 8.13 (d, 1H), 8.10 (m, 1H), 7.82-7.62 (m, 3H), 7.23 (d, 1H). ESMS m/z [M − 1] 251 for corresponding sulfonic acid (M = 252). CHN for C9H6ClF3O2S Calc.: C, 39.94; H, 2.23. Found: C, 40.30; H, 2.44. 15% 1H NMR (400 MHz, CDCl3) δ ppm 7.78 (d, 1H), 7.72 (m, 2H), 7.62 (m, 4H), 7.55-7.42 (m, 3H), 7.27 (d, 1H). ESMS m/z [M − 1] 259 for corresponding sulfonic acid (M = 260). CHN for C14H11ClO2S Calc.: C, 60.32; H, 3.98 Found: C, 60.79; H, 3.60. 54% 1H NMR (400 MHz, CDCl3) δ ppm 7.85-7.72 (m, 4H) 7.64 (t, J = 7.83 Hz, 1H) 7.32 (d, J = 15.26 Hz, 1H). ESMS m/z [M − 1] 250 for corresponding sulfonic acid, (M = 252.21) CHN for C9H6ClF3O2S, Calc: C, 39.94; H, 2.23; Found: C, 40.12; H, 2.39; 7% 1H NMR (400 MHz, CDCl3) δ ppm 7.78 (d, J = 15.26 Hz, 1H) 7.57-7.47 (m, 2H) 7.41 (d, J = 15.26 Hz, 1H) 7.26 (d, 1H) 7.23-7.15 (m, 1H). ESMS m/z [M − 1] 200.89 for corresponding sulfonic acid, (M = 202.20) CHN for C8H6ClFO2S, Calc: C, 43.55; H, 2.74; Found: C, 43.72; H, 2.82; - Method B
- Method B.1 Synthesis of Arylethenesulfonic Acid Ethyl Esters
- A 2.5M solution of n-butyllithium in hexanes (15.40 mmol) was added dropwise to a stirred solution of (diethoxyphosphoryl)methanesulfonic acid ethyl ester A (15.40 mmol) in THF (160 mL) at −78° C. The reaction mixture was stirred at −78° C. for 15 minutes, then a solution of the appropriate aldehyde (14.10 mmol) in THF (5 mL) was added dropwise. After the addition was complete, the reaction mixture was warmed to RT, stirred for 1 h, and quenched with brine. Most of the THF was then removed in vacuo and DCM (200 mL) was added. The organic layer was separated, washed with brine (3×100 mL), dried over MgSO4 and concentrated under reduced pressure. The crude product was purified on silica gel using hexane/ethyl acetate (2:1) to afford the ester B.
- The compounds listed in Table 2 were synthesized following this procedure.
-
TABLE 2 Structure Yield Analytical data 99% 1H NMR (400 MHz, CDCl3) δ ppm 7.71 (d, 1H), 7.58-7.48 (m, 2H), 6.95 (d, 1H), 6.82 (d, 1H), 4.21 (q, 2H), 3.92 (s, 3H), 1.40 (t, 3H). ESMS m/z [M + 1] 320 and 322 (Br isotopes). 91% 1H NMR (400 MHz, CDCl3) δ ppm 7.54 (d, J = 15.60 Hz, 1H) 7.42 (d, J = 8.58 Hz, 2H) 7.24 (s, 2H) 6.68 (d, J = 15.60 Hz, 1H) 4.22 (q, J = 7.15 Hz, 2H) 2.51 (s, 3H) 1.40 (t, J = 7.22 Hz, 3H). 77% 1H NMR (400 MHz, CDCl3) δ ppm 7.74 (d, J = 8.19 Hz, 2H) 7.66-7.56 (m, 3H) 6.87 (d, J = 15.60 Hz, 1H) 4.28 (q, J = 7.02 Hz, 2H) 1.43 (t, J = 7.22 Hz, 3H). 99% 1H NMR (400 MHz, CDCl3) δ ppm 7.620- 7.50 (m, 3H) 7.18 (d, J = 8.22 Hz, 2H) 6.77- 6.63 (m, 1H) 6.57 (t, 1H) 4.24 (q, J = 7.04 Hz, 2H) 1.41 (t, J = 7.24 Hz, 3H). 81% 1H NMR (400 MHz, CDCl3) δ ppm 7.72 (d, J = 8.61 Hz, 2H) 7.59 (d, J = 15.65 Hz, 1H) 7.53 (d, J = 8.61 Hz, 3H) 6.77 (d, J = 3.52 Hz, 1H) 6.73 (d, J = 15.26 Hz, 1H) 6.52 (dd, J = 3.52, 1.96 Hz, 1H) 4.24 (q, J = 7.04 Hz, 2H) 1.41 (t, J = 7.04 Hz, 3H). - Method B.2 Synthesis of Arylethenesulfonyl Chloride D
- Step i) Arylethenesulfonatetetrabutyl-ammonium C
- Tetrabutyl-ammonium iodide (18.10 mmol) was added to a solution of arylethenesulfonic acid ethyl ester B (15.08 mmol) in acetone (500 mL) and the reaction mixture was heated at reflux overnight. The reaction mixture was then cooled to RT and concentrated under reduced pressure. The residue was purified on silica gel using DCM/methanol (95:5) to afford a mixture of C and tetrabutyl-ammonium iodide. The oily residue was then triturated with diethyl ether to afford C (contaminated with tetrabutylammonium iodide) which was used in the next step without further purification.
- Step ii) Arylethenesulfonyl chloride D
- Sulfuryl chloride (1.72 g, 12.7 mmol) was added to a solution of triphenylphosphine (3.09 g, 11.8 mmol) in dry DCM (50 mL) at 0° C. The mixture was stirred at 0° C. for 15 minutes and the crude ammonium salt (5.8 mmol, prepared as described in step i) dissolved in DCM (20 mL) was added. The resulting solution was allowed to warm to RT and stirring was continued overnight. The reaction mixture was then concentrated under reduced pressure and the crude product was purified by flash column chromatography using 1-5% ethyl acetate/hexane to give sulfonyl chloride D.
- The compounds listed in Table 3 were synthesized following this method.
-
TABLE 3 Structure Yielda) Analytical data 44% 1H NMR (400 MHz, CDCl3) δ ppm 7.78 (d, 1H), 7.58-7.54 (m, 3H), 7.51 (d, 1H), 6.88 (d, 1H), 3.95 (s, 3H). ESMS m/z [M − 1] 290 and 292 (Br isotopes) for corresponding sulfonic acid (M = 293). CHN for C9H8BrClO3S Calc.: C, 34.69; H, 2.59. Found: C, 34.39; H, 2.70. 56% 1H NMR (400 MHz, CDCl3) δ ppm 7.77 (t, J = 8.22 Hz, 2H) 7.74-7.63 (m, 3H) 7.33 (d, J = 14.87 Hz, 1H). ESMS m/z [M − 1] 207.88 for corresponding sulfonic acid, (M = 209.22). CHN for C9H6ClNO2S, Calc: C, 47.48; H, 2.66; N, 6.15 Found: C, 47.48; H, 2.76; N, 6.20 34% 1H NMR (400 MHz, CDCl3) δ ppm 7.68 (d, J = 15.26 Hz, 1H) 7.45 (d, J = 8.22 Hz, 2H) 7.30- 7.24 (m, 2H) 7.17 (d, J = 15.26 Hz, 1H) 2.53 (s, 3H). ESMS m/z [M − 1] 228.93 for corresponding sulfonic acid, (M = 230.30). CHN for C9H9ClO2S2, Calc: C, 43.46; H, 3.65; Found: C, 43.71; H, 3.96. 49.5% 1H NMR (400 MHz, CDCl3) δ ppm 7.71 (d, J = 14.87 Hz, 1H) 7.58 (d, J = 8.61 Hz, 2H) 7.28- 7.14 (m, 3H) 6.80-6.39 (m, 1H). ESMS m/z [M − 1] 248.98 for corresponding sulfonic acid, (M = 250.22). CHN for C9H7ClF2O3S, Calc: C, 40.23; H, 2.63; Found: C, 40.60; H, 2.79. 33% 1H NMR (400 MHz, CDCl3) δ ppm 7.80-7.60 (m, 3H) 7.61-7.51 (m, 3H) 7.22 (d, J = 15.26 Hz, 1H) 6.81 (d, J = 3.52 Hz, 1H) 6.53 (d, J = 1.56 Hz, 1H). ESMS m/z [M − 1] 248.91 for corresponding sulfonic acid, (M = 250.27). CHN for C9H7ClF2O3S, Calc: C, 53.64; H, 3.38; Found: C, 53.58; H, 3.49. a)yield calculated over 2 steps -
- 2-Amino-4,5-difluorobenzenesulfonamide (99 mg, 0.48 mmol) and dichlorobenzenesulfonyl chloride (152 mg, 0.62 mmol) were dissolved in anhydrous pyridine (2 mL). The mixture was stirred at ambient temperature under an atmosphere of argon for 24 h. The solvent was evaporated in vacuo, water was added and the product was extracted with ethyl acetate. The combined organic phases were washed with brine, dried over magnesium sulfate and concentrated. Purification by preparative HPLC gave 107 mg (54% yield) of the title compound.
- 1H NMR (400 MHz, (CD3)2CO) δ ppm 8.30 (dd, 1H) 7.88 (dd, 1H) 7.76 (dd, 1H) 7.62 (t, 1H) 7.42 (dd, 6.95 Hz, 1H); MS (ESI) m/z 415, 417 [M−1]−.
-
- The title compound was synthesized as described for Example 178 in 16% yield, starting from 2-amino-5-(3-hydroxy-3-methylbut-1-ynyl)benzenesulfonamide and (E)-2-(5-bromo-2-methoxyphenyl)ethenesulfonyl chloride (93 mg, 0.30 mmol). Purification by preparative HPLC.
- MS (ESI) m/z 527,529 [M−1]−.
-
- 2-Amino-5-bromobenzenesulfonamide (2 g, 7.96 mmol), copper(I) iodide (0.076 g, 0.40 mmol) and bis(triphenylphosphine)palladium(II) chloride (0.280 g, 0.40 mmol) were slurried in anhydrous N,N-dimethylformamide (6 mL) in a microwave vial. 2-Methyl-3-butyn-2-ol (2.316 mL, 23.89 mmol) followed by diisopropylamine (3.37 mL, 23.89 mmol) were added, the vial was capped, purged with argon and the mixture was heated in a microwave at 100° C. for 30 min. The mixture was filtered and the solvent was evaporated in vacuo. Purification by column chromatography, using a gradient of 60-70% ethyl acetate in heptane as the eluent, gave 0.92 g (46% yield) of the title compound.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 7.55 (d, 1H) 7.30 (s, 2H) 7.20 (dd, 1H) 6.74 (d, 1H) 6.13 (s, 2H) 5.33 (s, 1H) 1.43 (s, 6H); MS (ESI) m/z 253 [M−1]−.
-
- (E)-4-Cyano-2-(2-(3,4-dichlorophenyl)vinylsulfonamido)benzenesulfonamide (123 mg, 0.28 mmol), sodium acetate (93 mg, 1.14 mmol) and para-toluenesulfonhydrazide (212 mg, 1.14 mmol) were dissolved in anhydrous tetrahydrofuran (3 mL) in a microwave vial. The vial was capped and evacuated two times with argon. The mixture was heated in a microwave at 180° C. for 15 min. The mixture was filtered and the solvent was evaporated in vacuo. Purification twice by preparative HPLC gave 4.8 mg (4% yield) of the title compound.
- 1H NMR (400 MHz, CD3OD) δ ppm 8.02-8.06 (m, 2H) 7.54 (d, 1H) 7.42 (d, 1H) 7.39 (d, 1H) 7.17 (dd, 1H) 3.56-3.62 (m, 2H) 3.07-3.13 (m, 2H); MS (ESI) m/z432, 434 [M−1]−.
-
- The title compound was synthesized as described for Example 178 in 57% yield, starting from 2-amino-4-cyanobenzenesulfonamide and (E)-2-(3,4-dichlorophenyl)ethenesulfonyl chloride.
- MS (ESI) m/z 430, 432 [M−1]−.
-
- The title compound was synthesized as described for Example 180 in 6% yield, starting from (E)-2-(2-(4-chloro-2-methoxyphenyl)vinylsulfonamido)-4-cyanobenzenesulfonamide.
- 1H NMR (400 MHz, CDCl3) δ ppm 8.05 (d, 1H) 7.86 (d, 1H) 7.49 (dd, 1H) 7.09 (d, 1H) 6.88 (dd, 1H) 6.82 (d, 1H) 3.78 (s, 3H) 3.53-3.58 (m, 2H) 3.12-3.18 (m, 2H); MS (ESI) m/z 428, 430 [M−1]−.
-
- The title compound was synthesized as described for Example 178 in 36% yield, starting from 2-amino-4-cyanobenzenesulfonamide and 2-(4-chloro-2-methoxy-phenyl)-ethenesulfonyl chloride.
- MS (ESI) m/z 426, 428 [M−1]−.
-
- The title compound was synthesized as described for Example 180 in 88% yield, starting from (E)-2-(2-(4-chloro-2-methoxyphenyl)vinylsulfonamido)-5-cyanobenzenesulfonamide (26 mg, 0.06 mmol). The reaction was run at 120° C.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 7.80 (d, 1H) 7.51 (dd, 1H) 7.44 (d, 1H) 7.15 (d, 1H) 6.98 (d, 1H) 6.89 (dd, 1H) 3.76 (s, 3H) 2.97-3.03 (m, 2H) 2.90-2.96 (m, 2H); MS (ESI) m/z 428, 430 [M−1]−.
-
- The title compound was synthesized as described for Example 178 in 27% yield, starting from 2-amino-5-cyanobenzenesulfonamide and 2-(4-chloro-2-methoxy-phenyl)-ethenesulfonyl chloride.
- MS (ESI) m/z 426, 428 [M−1]−.
-
- The title compound was synthesized as described for Example 179 a) in 16% yield, starting from 2-(2-(4-bromophenyl)ethylsulfonamido)benzenesulfonamide and 3,3-dimethyl-1-butyne.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 8.94 (s, 1H) 7.80-7.91 (m, 3H) 7.65-7.71 (m, 1H) 7.61 (t, 1H) 7.33 (t, 1H) 7.20-7.27 (m, 2H) 7.14-7.20 (m, 2H) 3.54-3.63 (m, 2H) 2.95-3.03 (m, 2H) 1.27 (s, 9H); MS (ESI) m/z 419 [M−1]−.
-
- The title compound was synthesized as described for Example 179 a) in 42% yield, starting from 2-[2-(4-bromophenyl)ethylsulfonylamino]benzenesulfonamide and 2-methyl-3-butyn-2-ol.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 8.94 (s, 1H) 7.81-7.90 (m, 3H) 7.66-7.71 (m, 1H) 7.61 (t, 1H) 7.33 (t, 1H) 7.25-7.30 (m, 2H) 7.18-7.23 (m, 2H) 5.43 (s, 1H) 3.57-3.64 (m, 2H) 2.97-3.04 (m, 2H) 1.44 (s, 6H); MS (ESI) m/z 421 [M−1]−.
-
- The title compound was synthesized as described for Example 179 a) in 27% yield, starting from 3-bromo-N-(2-sulfamoylphenyl)benzenesulfonamide and 3,3-dimethyl-1-butyne.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.37 (br. s., 1H) 7.75-7.86 (m, 5H) 7.61 (d, 1H) 7.54 (t, 2H) 7.45 (d, 1H) 7.22-7.30 (m, 1H) 1.29 (s, 9H); MS (ESI) m/z 391 [M−1]−
-
- The title compound was synthesized as described for Example 178 in 48% yield, starting from 2-aminobenzenesulfonamide and 3-bromobenzenesulfonyl chloride. The product solidified after the extractive work up. Diethyl ether (25 mL) was added and the crystals were crushed with a glass stick. The mixture was filtered and the solid was rinsed with diethyl ether and dried in vacuo at ambient temperature.
- MS (ESI) m/z 389, 391 [M−1]−.
-
- The title compound was synthesized as described for Example 180 in 47% yield, starting from 2-[2-(4-chloro-2-methoxy-phenyl)-ethanesulfonylamino]-benzenesulfonamide. The starting material was divided into two microwave vials and the reactions were run at 130° C. The reaction mixtures were pooled and then filtered. The solvent was evaporated in vacuo, water was added and the product was extracted with ethyl acetate. The organic phase was washed with brine, dried over magnesium sulfate and concentrated. Purification by recrystallization in methanol.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 8.95 (s, 1H) 7.88 (dd, 1H) 7.83 (s, 2H) 7.59-7.68 (m, 2H) 7.30-7.36 (m, 1H) 7.17 (d, 1H) 6.98 (d, 1H) 6.90 (dd, 1H) 3.66 (s, 3H) 3.45-3.52 (m, 2H) 2.90-2.97 (m, 2H); MS (ESI) m/z 403, 405 [M−1]−.
-
- The title compound was synthesized as described for Example 179 a) in 34% yield, starting from 2-(2-(6-bromopyridin-3-yl)ethylsulfonamido)benzenesulfonamide and 3,3-dimethyl-1-butyne. Purification by column chromatography, using 10 to 60% ethyl acetate in heptane as the eluent.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 8.96 (s, 1H) 8.36 (d, 1H) 7.78-7.92 (m, 3H) 7.57-7.70 (m, 3H) 7.29-7.38 (m, 2H) 3.59-3.71 (m, 2H) 2.99-3.07 (m, 2H) 1.29 (s, 9H); MS (ESI) m/z 420 [M−1]−
-
- The title compound was synthesized as described for Example 180 in 62% yield, starting from (E)-2-(2-(6-bromopyridin-3-yl)vinylsulfonamido)benzenesulfonamide. The reaction was run at 110° C. for 30 min. Purification by column chromatography using 5% methanol in chloroform as the eluent.
-
- The title compound was synthesized as described for Example 178 in 69% yield, starting from 2-aminobenzenesulfonamide and (E)-2-(6-bromopyridin-3-yl)ethenesulfonyl chloride. Purification by column chromatography, using ammonium hydroxide/methanol/chloroform (1:10:89) as the eluent.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 8.60 (d, 1H) 8.12 (dd, 1H) 7.57 (d, 1H) 7.50 (d, 1H) 7.40 (d, 1H) 6.98-7.33 (m, 6H) 6.67 (br. s., 1H).
-
- Triphenylphosphine (2.74 g, 10.44 mmol) was dissolved in anhydrous dichloromethane (30 mL) under an atmosphere of argon and the mixture was cooled to 0° C. with an ice bath. Sulfuryl chloride (0.87 mL, 10.68 mmol) was added dropwise and the cooling bath was removed. The solution was stirred at ambient temperature for 15 min. A solution of (E)-2-(6-bromopyridin-3-yl)ethenesulfonic acid tetrabutylammonium (2.46 g, 4.86 mmol) in anhydrous dichloromethane (10 mL) was added dropwise over 10 min. The reaction was stirred at ambient temperature for 1.5 h and the mixture was concentrated. Purification by column chromatography, using a gradient of 5-30% ethyl acetate in heptane as the eluent, gave 0.51 g of the title compound.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 8.54 (d, 1H) 8.07 (dd, 1H) 7.47 (d, 1H) 7.09 (d, 1H) 6.92 (d, 1H).
-
- (E)-Ethyl 2-(6-bromopyridin-3-yl)ethenesulfonate (1.42 g, 4.86 mmol) and tetrabutylammonium iodide (1.98 g, 5.35 mmol) were dissolved in anhydrous acetone and the mixture was heated at 85° C. over night. The mixture was filtered and the solvent was evaporated to give 2.99 g of the title compound.
- MS (ESI) m/z 262, 264 [M−1]−.
-
- Ethyl methanesulfonate (1.00 mL, 9.34 mmol) was dissolved in anhydrous tetrahydrofuran (20 mL) in an oven dried round bottle under an atmosphere of argon. The solution was cooled to −78° C. and 2.5 M n-butyllithium in hexane (4.09 mL, 10.23 mmol) was added dropwise over 15 min. The mixture was stirred at −78° C. for 15 min and diethyl chlorophosphate (0.78 mL, 5.38 mmol) was added dropwise. The reaction was stirred at −78° C. for 1 h and then the temperature was allowed to reach 0° C. before recooling to −78° C. A solution of 2-bromo-5-formylpyridine (1.0 g, 5.38 mmol) in anhydrous tetrahydrofuran (5 mL) was added dropwise over 15 min and the resulting mixture was stirred for 15 min and then allowed to reach ambient temperature. Water (3 mL) was added and the solvent was evaporated in vacuo. The residue was partitioned between diethyl ether and water. The aqueous phase was extracted with diethyl ether and the formed precipitate was filtered off and rinsed with diethyl ether. This was pure product which was pooled with the rest of the material after purification. The aqueous phase was extracted once more with diethyl ether and the combined organic phases were dried over magnesium sulfate and concentrated. Purification by column chromatography, using a gradient of 10-60% ethyl acetate in heptane as the eluent, gave 0.77 g pooled with the material from the extractive work up to give 1.42 g (90% yield) of the title compound.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 8.77 (d, 1H) 8.19 (dd, 1H) 7.79 (d, 1H) 7.67 (d, 2H) 4.22 (q, 2H) 1.30 (t, 3H); MS (ESI) m/z 262, 264 [M−1]− (M-ethyl).
-
- The title compound was synthesized as described for Example 179 a) in 25% yield, starting from 2-(2-(6-bromopyridin-3-yl)ethylsulfonamido)benzenesulfonamide and cyclohexylacetylene. Purification by column chromatography using a gradient of 10-60% ethyl acetate in heptane as the eluent.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 8.96 (s, 1H) 8.37 (d, 1H) 7.78-7.92 (m, 3H) 7.57-7.70 (m, 3H) 7.29-7.39 (m, 2H) 3.65 (t, 2H) 2.98-3.07 (m, 2H) 2.60-2.70 (m, 1H) 1.77-1.87 (m, 2H) 1.61-1.73 (m, 2H) 1.41-1.55 (m, 3H) 1.29-1.40 (m, 3H); MS (ESI) m/z 446 [M−1]−.
-
- The title compound was synthesized as described for Example 180 in 62% yield, starting from (E)-2-(2-(4-bromophenyl)vinylsulfonamido)-5-methylbenzenesulfonamide. The reaction was run at 120° C. for 30 min. Purification by column chromatography using 10 to 50% ethyl acetate in heptane as the eluent.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 8.84 (br.s, 1H) 7.76 (br.s, 2H) 7.69 (br. s, 1H) 7.55 (d, 1H) 7.39-7.48 (m, 3H) 7.15-7.21 (m, 2H) 3.50-3.58 (m, 2H) 2.92-2.99 (m, 2H) 2.33 (s, 3H); MS (ESI) m/z431, 433 [M−1]−.
-
- The title compound was synthesized as described for Example 178 in 96% yield, starting from 2-amino-5-methylbenzenesulfonamide and (E)-2-(4-bromophenyl)ethenesulfonyl chloride (US20040186134 A1). Purification by column chromatography using a gradient of 10-50% ethyl acetate in heptane as the eluent.
- MS (ESI) m/z 429, 431 [M−1]−.
-
- The title compound was synthesized as described for Example 178 in 5% yield, starting from 2-aminobenzenesulfonamide and 3-(2-methylthiazol-4-yl)benzene-1-sulfonyl chloride.
- 1H NMR (600 MHz, DMSO-d6) δ ppm 9.40 (s, 1H) 8.47 (s, 1H) 8.20 (d, 1H) 8.11 (s, 1H) 7.74-7.87 (m, 4H) 7.64 (t, 1H) 7.52 (br. s, 2H) 7.19-7.26 (m, 1H) 2.73 (s, 3H); MS
- (ESI) m/z 408 [M−1]−.
-
- The title compound was synthesized as described for Example 178 in 37% yield, starting from 2-aminobenzenesulfonamide and (E)-2-(3-(trifluoromethyl)phenyl)ethenesulfonyl chloride.
- 1H NMR (600 MHz, DMSO-d6) δ ppm 9.17 (s, 1H) 8.12 (s, 1H) 7.99 (d, 1H) 7.81-7.85 (m, 3H) 7.79 (d, 1H) 7.61-7.73 (m, 4H) 7.59 (t, 1H) 7.28 (t, 1H); MS (ESI) m/z 405 [M−1]−1.
-
- The title compound was synthesized as described for Example 178 in 31% yield, starting from 2-aminobenzenesulfonamide and (E)-2-(2-fluorophenyl)ethenesulfonyl chloride.
- 1H NMR (600 MHz, DMSO-d6) δ ppm 9.12 (s, 1H) 7.79-7.87 (m, 3H) 7.77 (t, 1H) 7.41-7.65 (m, 5H) 7.23-7.33 (m, 3H); MS (ESI) m/z 355 [M−1]−.
-
- Sodium acetate (64 mg, 0.78 mmol) and para-toluenesulfonhydrazide (145 mg, 0.78 mmol) was added to a solution of (E)-5-chloro-2-(2-(4-chloro-2-methoxyphenyl)vinylsulfonamido)benzenesulfonamide (85 mg, 0.19 mmol) in tetrahydrofuran (3 mL). The reaction mixture was heated at 180° C. for 15 min in a microwave reactor. The solids were removed by filtration, washed with tetrahydrofuran and the combined filtrates were evaporated. Purification by preparative HPLC gave 0.037 g (43% yield) of the title compound.
- 1H NMR (DMSO-d6) δ ppm 8.99 (br s, 1H) 8.15-7.75 (m, 3H) 7.62 (br s, 2H) 7.17 (d, 1H) 7.00 (d, 1H) 6.91 (dd, 1H) 3.70 (s, 3H) 3.46 (br s., 2H) 2.88-3.03 (m, 2H); MS (ESI) m/z 437 [M−1]−.
-
- A solution of 2-amino-5-chlorobenzenesulfonamide (0.041 g, 0.20 mmol) and (E)-2-(4-chloro-2-methoxyphenyl)ethenesulfonyl chloride (0.053 g, 0.20 mmol) in pyridine (2 mL) was stirred at room temperature over night. The reaction mixture was concentrated, 1 M hydrochloric acid was added and the mixture was extracted with dichloromethane. The organic phase was dried over magnesium sulfate and the solvent was evaporated to give 0.087 g (99% yield) of the title compound.
- MS (ESI) m/z 435 [M−1]−.
-
- The title compound was synthesized as described for Example 194 a) in 21% yield, using 2,3-dichlorobenzene-1-sulfonyl chloride (1.5 equiv.). Purification by preparative HPLC.
- 1H NMR (DMSO-d6) δ ppm 8.15 (dd, 1H) 7.88-8.07 (m, 3H) 7.84 (d, 1H) 7.54-7.67 (m, 2H) 7.36 (d, 1H); MS (ESI) m/z 413, 415 [M−1]−.
-
- The title compound was synthesized as described for Example 194 a) in 7% yield, starting from 2-amino-4,6-difluorobenzenesulfonamide and 2,3-dichlorobenzene-1-sulfonyl chloride (1.5 equiv.). Purification by preparative HPLC.
- 1H NMR (DMSO-d6) δ ppm 8.27 (d, 2H) 8.03 (d, 1H) 7.65 (t, 1H) 6.96 (d, 1H); MS (ESI) m/z 415, 417 [M−1]−.
-
- The title compound was synthesized as described for Example 194 in 28% yield, starting from (E)-2-(2-(3,4-dichlorophenyl)vinylsulfonamido)-4,6-difluorobenzenesulfonamide. The reaction was run at 140° C. Purification by preparative HPLC.
- 1H NMR (DMSO-d6) δ ppm 9.60 (br s, 1H) 8.05 (br s, 2H) 7.43 (d, 1H) 7.36 (d, 1H) 7.02-7.18 (m, 3H) 3.67 (t, 2H) 2.80-2.94 (m, 2H); MS (ESI) m/z 443, 445 [M−1]−.
-
- The title compound was synthesized as described for Example 194 a) in 49% yield, starting from 2-amino-4,6-difluorobenzenesulfonamide and (E)-2-(3,4-dichlorophenyl)ethenesulfonyl chloride (1.5 equiv.). Purification by column chromatography, using chloroform/methanol (95:5) as the eluent.
- 1H NMR (DMSO-d6) δ ppm 10.01 (br s, 1H) 8.25 (br s., 2H) 8.14 (s, 1H) 7.66-7.83 (m, 4H) 7.12-7.39 (m, 2H); MS (ESI) m/z 441, 443 [M−1]−.
-
- The title compound was synthesized as described for Example 197 in 44% yield, starting from (E)-2-(2-(4-chloro-2-methoxyphenyl)vinylsulfonamido)-4,6-difluorobenzenesulfonamide. Purification by preparative HPLC.
- 1H NMR (DMSO-d6) δ ppm 9.82 (br s, 1H) 8.24 (s, 2H) 7.19-7.35 (m, 3H) 7.00 (d, 1H) 6.92 (dd, 1H) 3.58-3.74 (m, 5H) 2.92-3.01 (m, 2H); MS (ESI) m/z 439 [M−1]−.
-
- The title compound was synthesized as described for Example 197 a) in 48% yield, using (E)-2-(4-chloro-2-methoxyphenyl)ethenesulfonyl chloride (1.5 equiv.). Purification by column chromatography, using chloroform/methanol (95:5) as the eluent.
- 1H NMR (DMSO-d6) δ ppm 9.95 (br s, 1H) 8.24 (br s, 2H) 7.68-7.80 (m, 2H) 7.51 (d, 1H) 7.04-7.34 (m, 4H) 3.91 (s, 3H); MS (ESI) m/z 437 [M−1]−.
-
- The title compound was synthesized as described for Example 194 in 26% yield, starting from (E)-5-cyano-2-(2-(3,4-dichlorophenyl)vinylsulfonamido)benzenesulfonamide. The reaction was run at 120° C. Purification by preparative HPLC.
- 1H NMR (DMSO-d6) δ ppm 9.00 (br s, 1H) 7.99 (br s, 1H) 7.74-7.93 (m, 3H) 7.58 (d, 1H) 7.29-7.40 (m, 2H) 7.06 (dd, 1H) 3.62 (br s, 2H) 2.82-2.91 (m, 2H); MS (ESI) m/z 432, 434 [M−1]−.
-
- The title compound was synthesized as described for Example 194 a), starting from 2-amino-5-cyanobenzenesulfonamide and (E)-2-(3,4-dichlorophenyl)ethenesulfonyl chloride (1.5 equiv.). Purification by column chromatography, using chloroform/methanol (95:5-9:1) as the eluent.
- MS (ESI) m/z 430, 432 [M−1]−.
-
- The title compound was synthesized as described for Example 197 in 32% yield, starting from (E)-2-(2-(3,4-dichlorophenyl)vinylsulfonamido)-4-methylbenzenesulfonamide.
- Purification by preparative HPLC.
- 1H NMR (DMSO-d6) δ ppm 8.81 (s, 1H) 7.58-7.75 (m, 3H) 7.40-7.49 (m, 2H) 7.34 (s, 1H) 7.14 (dd, 1H) 7.04 (d, 1H) 3.48-3.65 (m, 2H) 2.87-3.00 (m, 2H) 2.27 (s, 3H); MS (ESI) m/z 421, 423 [M−1]−.
-
- The title compound was synthesized as described for Example 194 a) in 54% yield, starting from 2-amino-4-methylbenzenesulfonamide and (E)-2-(3,4-dichlorophenyl)ethenesulfonyl chloride (1.5 equiv.). Purification by column chromatography, using chloroform/methanol (95:5) as the eluent.
- 1H NMR (DMSO-d6) δ ppm 9.13 (s, 1H) 8.06 (s, 1H) 7.76 (s, 2H) 7.67-7.74 (m, 3H) 7.52-7.66 (m, 2H) 7.41 (s, 1H) 7.11 (d, 1H) 2.35 (s, 3H); MS (ESI) m/z 419, 421 [M−1]−.
-
- The title compound was synthesized as described for Example 197 in 66% yield, starting from (E)-2-(2-(4-chloro-2-methoxyphenyl)vinylsulfonamido)-4-methylbenzenesulfonamide.
- Purification by preparative HPLC.
- 1H NMR (DMSO-d6) δ ppm 8.91 (s, 1H) 7.72-7.82 (m, 3H) 7.44 (d, 1H) 7.10-7.21 (m, 2H) 7.00 (d, 1H) 6.91 (dd, 1H) 3.68 (s, 3H) 3.45-3.55 (m, 2H) 2.91-2.99 (m, 2H) 2.37 (s, 3H); MS (ESI) m/z 417 [M−1]−.
-
- The title compound was synthesized as described for Example 194 a) in 69% yield, starting from 2-amino-4-methylbenzenesulfonamide and (E)-2-(4-chloro-2-methoxyphenyl)ethenesulfonyl chloride (1.5 equiv.). Purification by column chromatography, using chloroform/methanol (95:5) as the eluent.
- 1H NMR (DMSO-d6) δ ppm 9.03 (s, 1H) 7.75 (s, 2H) 7.59-7.73 (m, 3H) 7.35-7.46 (m, 2H) 7.20 (d, 1H) 7.08 (td, 2H) 3.89 (s, 3H) 2.34 (s, 3H); MS (ESI) m/z 415 [M−1]−.
-
- The title compound was synthesized as described for Example 197 in 59% yield, starting from (E)-5-chloro-2-(2-(3,4-dichlorophenyl)vinylsulfonamido)benzenesulfonamide.
- Purification by preparative HPLC.
- 1H NMR (DMSO-d6) δ ppm 8.97 (s, 1H) 7.96 (br s, 2H) 7.86 (s, 1H) 7.64-7.74 (m, 2H) 7.49-7.59 (m, 2H) 7.25 (dd, 1H) 3.62-3.74 (m, 2H) 2.97-3.09 (m, 2H); MS (ESI) m/z 441, 443 [M−1]−.
-
- The title compound was synthesized as described for Example 194 a) in 49% yield, starting from 2-amino-5-chlorobenzenesulfonamide and (E)-2-(3,4-dichlorophenyl)ethenesulfonyl chloride (1.5 equiv.). Purification by column chromatography, using chloroform/methanol (95:5) as the eluent.
- 1H NMR (DMSO-d6) δ ppm 9.18 (br s, 1H) 8.06 (d, 1H) 7.96 (br s, 2H) 7.83 (s, 1H) 7.51-7.77 (m, 6H); MS (ESI) m/z 439, 441 [M−1]−.
-
- A solution of N,N-diallyl-4-(3-(5-chloro-6-methoxypyridin-3-yl)phenylsulfonamido)pyridine-3-sulfonamide (0.19 g, 0.36 mmol) in dichloromethane (5 mL) was added to tetrakis(triphenylphosphine)palladium(0) (0.021 g, 0.022 mmol) and 1,3-dimethylbarbituric acid (0.336 g, 2.15 mmol) under an atmosphere of argon. The reaction was heated at 100° C. for 15 min in a microwave reactor and the reaction mixture was concentrated. Purification by column chromatography, using chloroform/methanol (95:5-9:1) as the eluent, gave the title compound in 49% yield.
- 1H NMR (DMSO-d6) δ ppm 13.01 (br s, 1H) 8.39 (dd, 2H) 8.18 (d, 1H) 8.09 (t, 1H) 7.94 (dd, 1H) 7.73-7.79 (m, 2H) 7.48 (t, 1H) 7.33 (d, 1H) 6.89 (s, 2H) 3.88 (s, 3H); MS (ESI) m/z453 [M−1]−.
-
- A mixture of N,N-diallyl-4-(3-bromophenylsulfonamido)pyridine-3-sulfonamide (0.29 g, 0.62 mmol), 5-chloro-6-methoxypyridin-3-ylboronic acid (0.15 g, 0.81 mmol), (1,1′-bis(diphenylphosphino)ferrocene)-dichloropalladium(II) (0.026 g, 0.030 mmol), cesium carbonate (0.30 g, 0.93 mmol), 1,2-dimethoxyethane (4.5 mL) and water (0.5 mL) under an atmosphere of argon was heated at 100° C. for 15 min in a microwave reactor. The reaction mixture was partitioned between ethyl acetate and diluted hydrochloric acid, the organic phase was dried over magnesium sulfate and the solvent was evaporated. Purification by column chromatography, using chloroform/methanol (98:2) as the eluent, gave the title compound in 58% yield.
- MS (ESI) m/z 533 [M−1]−.
-
- A mixture of N,N-diallyl-4-aminopyridine-3-sulfonamide (0.25 g, 1.0 mmol) and cesium carbonate (0.82 g, 2.5 mmol) in tetrahydrofuran (10 mL) was stirred for 5 min, 3-bromobenzenesulfonyl chloride (0.17 mL, 1.2 mmol) was added and the reaction was stirred at room temperature over night. Diluted hydrochloric acid was added and the mixture was extracted with ethyl acetate. The organic phase was dried over magnesium sulfate and the solvent was evaporated. Purification by column chromatography, using chloroform/methanol (95:5) as the eluent, gave the title compound in 72% yield.
- 1H NMR (DMSO-d6) δ ppm 13.19 (br s, 1H) 8.54 (d, 1H) 8.07 (dd, 1H) 7.95 (t, 1H) 7.82-7.88 (m, 1H) 7.79 (ddd, 1H) 7.52 (t, 1H) 7.44 (d, 1H) 5.47-5.60 (m, 2H) 5.02-5.11 (m, 4H) 3.74 (d, 4H); MS (ESI) m/z 470, 472 [M−1]−.
-
- A mixture of N,N-diallyl-4-chloropyridine-3-sulfonamide (1.5 g, 5.5 mmol), lithium iodide (0.74 g, 5.5 mmol) and ammonia (7 M in methanol, 14 mL, 5.5 mmol) under an amosphere of argon was heated at 120° C. for 2 h in a microwave reactor. Silica was added and the solvent was evaporated. Purification by column chromatography, using chloroform/methanol (95:5) as the eluent, gave the title compound in 59% yield.
- 1H NMR (DMSO-d6) δ ppm 8.43 (s, 1H) 8.12 (d, 1H) 6.67-6.97 (m, 3H) 5.57-5.69 (m, 2H) 5.12-5.23 (m, 4H) 3.80 (d, 4H); MS (ESI) m/z 254 [M−1]−.
-
- 4-Chloropyridine-3-sulfonamide (1.93 g, 10.0 mmol) was added in portions to a solution of sodium hydride (60% in mineral oil, 0.88 g, 22.0 mmol) in N,N-dimethylformamide (50 mL). The reaction mixture was stirred at room temperature for 1 h, cooled to 0° C. and allyl bromide (1.90 mL, 22.0 mmol) was added. After 3 h at room temperature methanol was added and the reaction was partitioned between chloroform and water, the organic phase was dried over magnesium sulfate and the solvent was evaporated. Purification by column chromatography, using chloroform/methanol (99:1) as the eluent, gave the title compound in 87% yield.
- 1H NMR (DMSO-d6) δ ppm 9.06 (s, 1H) 8.79 (d, 1H) 7.83 (d, 1H) 5.59-5.73 (m, 2H) 5.12-5.24 (m, 4H) 3.93 (d, 4H).
-
- The title compound was synthesized as described for Example 197 in 36% yield, starting from (E)-4-(2-(4-(benzofuran-2-yl)phenyl)vinylsulfonamido)pyridine-3-sulfonamide.
- Purification by preparative HPLC.
- 1H NMR (DMSO-d6) δ ppm 12.82 (br s, 1H) 8.48 (s, 1H) 8.02 (d, 1H) 7.84-7.90 (m, 2H) 7.59-7.68 (m, 2H) 7.37-7.48 (m, 4H) 7.23-7.35 (m, 2H) 6.99 (br s, 2H) 3.42-3.50 (m, 2H) 3.07-3.20 (m, 2H); MS (ESI) m/z 456 [M−1]−.
-
- The title compound was synthesized as described for Example 203 in 12% yield, starting from (E)-N,N-diallyl-4-(2-(4-(benzofuran-2-yl)phenyl)vinylsulfonamido)pyridine-3-sulfonamide and heated at 120° C. for 1 h 15 min. Purification by column chromatography, using chloroform/methanol (97:3-9:1) as the eluent.
- 1H NMR (DMSO-d6) δ ppm 13.01 (br s, 1H) 8.51 (s, 1H) 8.09 (d, 1H) 7.96 (d, 2H) 7.80 (d, 2H) 7.67 (ddd, 2H) 7.55 (d, 1H) 7.43 (dd, 2H) 7.23-7.40 (m, 3H) 6.98 (br s, 2H); MS (ESI) m/z 454 [M−1]−.
-
- A mixture of (E)-N,N-diallyl-4-(2-(4-bromophenyl)vinylsulfonamido)pyridine-3-sulfonamide (0.47 g, 0.94 mmol), 2-benzofuranboronic acid (0.20 g, 1.23 mmol), (1,1′-bis(diphenylphosphino)ferrocene)-dichloropalladium(II) (0.039 g, 0.050 mmol), cesium carbonate (0.46 g, 1.41 mmol), 1,2-dimethoxyethane (9 mL) and water (1 mL) under an atmosphere of argon was heated at 120° C. for 15 min in a microwave. The reaction mixture was partitioned between ethyl acetate and diluted hydrochloric acid, the organic phase was dried over magnesium sulfate and the solvent was evaporated. Purification by column chromatography, using chloroform/methanol (98:2) as the eluent, gave the title compound in 38% yield.
- 1H NMR (DMSO-d6) δ ppm 13.03 (br s, 1H) 8.52 (d, 1H) 8.04 (dd, 1H) 7.96 (d, 2H) 7.83 (d, 2H) 7.67 (ddd, 2H) 7.57 (d, 1H) 7.43-7.51 (m, 2H) 7.25-7.41 (m, 3H) 5.60-5.73 (m, 2H) 5.00-5.15 (m, 4H) 3.87-3.96 (m, 4H); MS (ESI) m/z 534 [M−1]−.
-
- Triethylamine (0.440 mL, 3.16 mmol) added was to a solution of N,N-diallyl-4-aminopyridine-3-sulfonamide (0.40 g, 1.58 mmol) in pyridine (10 mL), the mixture was stirred for 5 min and (E)-2-(4-bromophenyl)ethenesulfonyl chloride (0.53 g, 1.89 mmol) was added. After stirring over night the pyridine was evaporated, diluted hydrochloric acid was added and the mixture was extracted with ethyl acetate. The organic phase was dried over magnesium sulfate and the solvent was evaporated. Purification by column chromatography, using chloroform/methanol (97:3) as the eluent, gave the title compound in 60% yield.
- 1H NMR (DMSO-d6) δ ppm 13.03 (br s, 1H) 8.52 (d, 1H) 8.03 (dd, 1H) 7.58-7.72 (m, 4H) 7.30-7.47 (m, 3H) 5.57-5.74 (m, 2H) 4.99-5.15 (m, 4H) 3.89 (d, 4H).
-
- The title compound was synthesized as described for Example 194 in 37% yield, starting from (E)-2-(2-(4-chloro-2-methoxyphenyl)vinylsulfonamido)-5-methylbenzenesulfonamide. The reaction was run at 180° C. Purification by preparative HPLC.
- 1H NMR (DMSO-d6) δ ppm 8.85 (s, 1H) 7.75 (s, 2H) 7.69 (d, 1H) 7.54 (d, 1H) 7.44 (dd, 1H) 7.16 (d, 1H) 6.99 (d, 1H) 6.90 (dd, 1H) 3.68 (s, 3H) 3.39-3.47 (m, 2H) 2.88-2.96 (m, 2H) 2.33 (s, 3H); MS (ESI) m/z 417 [M−1]−.
-
- The title compound was synthesized as described for Example 194 a) in 81% yield, starting from 2-amino-5-methylbenzenesulfonamide and (E)-2-(4-chloro-2-methoxyphenyl)ethenesulfonyl chloride (1 equiv.).
- 1H NMR (DMSO-d6) δ ppm 8.95 (s, 1H) 7.76 (s, 2H) 7.60-7.66 (m, 2H) 7.48-7.59 (m, 2H) 7.34-7.44 (m, 2H) 7.19 (d, 1H) 7.06 (dd, 1H) 3.88 (s, 3H) 2.29 (s, 3H); MS (ESI) m/z 415 [M−1]−.
-
- The title compound was synthesized as described for Example 194 in 38% yield, starting from (E)-4-(2-(4-chloro-2-methoxyphenyl)vinylsulfonamido)pyridine-3-sulfonamide. The reaction was run at 180° C. Purification by preparative HPLC.
- 1H NMR (DMSO-d6) δ ppm 12.83 (br s, 1H) 8.48 (s, 1H) 8.01 (d, 1H) 7.36 (d, 1H) 7.22 (d, 1H) 7.04 (d, 1H) 6.87-7.01 (m, 3H) 3.82 (s, 3H) 3.23-3.30 (m, 2H) 2.94-3.04 (m, 2H);
- MS (ESI) m/z 404 [M−1]−.
-
- The title compound was synthesized as described for Example 203 in 65% yield, starting from (E)-N,N-diallyl-4-(2-(4-chloro-2-methoxyphenyl)vinylsulfonamido)pyridine-3-sulfonamide and heating at 100° C. for 1 h. The reaction mixture was evaporated and subsequent purification by column chromatography, using chloroform/methanol (98:2-9:1) as the eluent.
- 1H NMR (DMSO-d6) δ ppm 13.04 (br s, 1H) 8.52 (s, 1H) 8.09 (d, 1H) 7.68 (d, 1H) 7.51-7.59 (m, 1H) 7.37-7.47 (m, 2H) 7.19 (d, 1H) 7.06 (dd, 1H) 6.96 (br s, 2H) 3.90 (s, 3H);
- MS (ESI) m/z 402 [M−1]−.
-
- The title compound was synthesized as described for Example 204 c) in 50% yield, starting from N,N-diallyl-4-aminopyridine-3-sulfonamide and (E)-2-(4-chloro-2-methoxyphenyl)ethenesulfonyl chloride. Purification by column chromatography, using chloroform/methanol (98:2) as the eluent.
- 1H NMR (CDCl3) δ ppm 9.27 (br s, 1H) 8.88 (s, 1H) 8.56 (d, 1H) 7.82 (d, 1H) 7.54 (d, 1H) 7.37 (d, 1H) 7.10 (d, 1H) 7.01 (dd, 1H) 6.96 (d, 1H) 5.59-5.73 (m, 2H) 5.18-5.29 (m, 4H) 3.88-3.97 (m, 7H); MS (ESI) m/z 482[M−1]−.
-
- 2-[[(E)-2-(4-Bromophenyl)ethenylsulfonylamino]benzenesulfonamide (75 mg, 0.18 mmol), cyclopenten-1-ylboronic acid (40.2 mg, 0.36 mmol), (1,1′-bis(diphenylphosphino)ferrocene)-dichloropalladium(II) (14.79 mg, 0.02 mmol) and sodium carbonate (38.1 mg, 0.36 mmol) were suspended in N,N-dimethylformamide (2 mL) and the reaction mixture was stirred under an atmosphere of argon at 90° C. for 16 hours. The reaction mixture was diluted with tetrahydrofuran and filtered through Celite. The solvent was removed in vacuo and the residue was purified by preparative HPLC. The residue was purified a second time by column chromatography, using gradient mixtures of ethyl acetate/heptane as eluent (0% -100% ethyl acetate) to give 50 mg (69% yield) of the title compound.
- 1H NMR (DMSO-d6) δ ppm 7.84 (m, 3H), 7.62 (m, 2H), 7.56 (m, 2H), 7.49 (m, 2H), 7.39 (m, 1H), 7.25 (m, 1H), 2.65 (m, 2H), 2.49 (m, 2H), 1.49 (m, 2H); MS (ESI) m/z 403 [M−1]−.
-
- 2-[[(E)-2-(4-Bromophenyl)ethenylsulfonylamino]benzenesulfonamide (79 mg, 0.19 mmol), (2-tert-butyl-1-ethynyl)diisopropoxyborane (80 mg, 0.38 mmol), (1,1′-bis(diphenylphosphino)ferrocene)-dichloropalladium(II) (7.79 mg, 9.47 μmol) and sodium carbonate (40.1 mg, 0.38 mmol) were suspended in N,N-dimethylformamide (2.5 mL) and water (0.2 mL) and the reaction mixture was stirred vigorously under an atmosphere of argon at 90° C. for 3 hours. The mixture was filtered through a pad of Celite, the solvent was removed in vacuo and the residue was purified by preparative HPLC to give 10 mg (23% yield) of the title compound.
- 1H NMR (CD3OD) δ ppm 7.88 (m, 1H), 7.72 (m, 1H), 7.50 (m, 4H), 7.33 (m, 2H), 7.20 (br. s., 1H), 7.10 (m, 1H), 1.30 (s, 9H); MS (ESI) m/z417 [M−1]−.
-
- 2-[[(E)-2-(4-Bromophenyl)ethenylsulfonylamino]benzenesulfonamide (79 mg, 0.19 mmol), 2-methyl-3-butyn-2-ol (18 μL, 0.19 mmol), copper(I) iodide (9.00 mg, 0.05 mmol), tetrakis(triphenylphosphine)palladium(0) (30.0 mg, 0.03 mmol) and triethylamine (79 μL, 0.57 mmol) were dissolved in tetrahydrofuran (2 mL), the reaction mixture was stirred at 50° C. under an atmosphere of argon for 4 hours and subsequently stirred at room temperature for 10 hours. The reaction mixture was filtered, the solvent was removed in vacuo and the residue was purified by preparative HPLC to give 4 mg (5% yield) of the title compound.
- 1H NMR (CD3OD) δ ppm 7.78 (m, 1H), 7.73 (m, 1H), 7.5 (m, 4H), 7.38 (m, 2H), 7.23 (m, 1H), 7.15 (m, 1H), 1.54 (s, 6H); MS (ESI) m/z419 [M−1]−.
-
- A mixture of 2-[2-(4-bromo-phenyl)-ethanesulfonylamino]-5-methyl-benzenesulfonamide (0.08 g, 0.18 mmol), N,N-diisopropylethylamine (0.1 mL, 0.55 mmol), and copper(I) iodide (7 mg, 0.04 mmol) in anhydrous N,N-dimethylformamide (2 mL) was degassed with nitrogen for 30 min. (1,1′-Bis(diphenylphosphino)ferrocene)-dichloropalladium(II) (14 mg, 0.02 mmol) and 3,3-dimethyl-1-butyne (0.045 mL, 0.37 mmol) were added and the reaction mixture was heated in a sealed tube at 80° C. overnight. The reaction mixture was diluted with ethyl acetate (20 mL), washed with water and brine, dried over sodium sulfate and concentrated under reduced pressure. The residue was purified by preparative TLC, using 5% methanol in dichloromethane as the eluent, to give 20 mg (25% yield) of the title compound.
- 1H NMR (400 MHz, CD3OD) δ ppm 7.77 (s, 1H), 7.65 (d, 1H), 7.40 (d, 1H), 7.22 (d, 2H), 7.20 (d, 2H), 3.48 (m, 2H), 3.45 (m, 2H), 2.37 (m, 3H), 1.28 (s, 9H); MS (ESI) m/z 433 [M−1]−.
-
- A mixture of 2-amino-5-methyl-benzenesulfonamide (0.1 g, 0.53 mmol) and 2-(4-bromo-phenyl)-ethanesulfonyl chloride (0.14 g, 0.49 mmol) in anhydrous pyridine (4 mL) was stirred at room temperature overnight. The reaction mixture was diluted with ethyl acetate (20 mL) and the organic phase was washed with aqueous hydrochloric acid (1 M, 20 mL), saturated sodium bicarbonate (20 mL) and brine, dried over magnesium sulfate and concentrated under reduced pressure. The residue was purified by preparative TLC, using 5% methanol in dichloromethane as the eluent, to give 80 mg (38% yield) of title compound.
- 1H NMR (400 MHz, CD3OD) δ ppm 7.77 (s, 1H), 7.64 (d, 1H), 7.40-7.37 (m, 3H), 7.11 (d, 2H), 3.49-3.45 (m, 2H), 3.04-3.02 (m, 2H), 2.37 (s, 3H)
-
- Thionyl chloride (1.51 mL, 20.8 mmol) was added dropwise at 0° C. to a suspension of sodium 2-(4-bromo-phenyl)-ethanesulfonate (4.33 g, 15.08 mmol) in a mixture of anhydrous benzene (50 mL) and N,N-dimethylformamide (1 mL). The reaction mixture was allowed to warm to room temperature and was then heated at 80° C. for 18 hours. The reaction mixture was cooled to room temperature and filtered through a pad of Celite. The solids were washed with benzene and the combined filtrates were concentrated in vacuo. The residue was taken up in hexane (30 mL) and the resulting suspension was heated at 50° C. for 30 min. After cooling to room temperature, the formed precipitate was collected by filtration to give 4.06 g (95% yield) of the title compound.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 7.42 (d, 2H), 7.17 (d, 2H), 2.89-2.79 (m, 2H), 2.80-2.68 (m, 2H); MS (ESI) m/z 263 [M−1]− (corresponding sulfonic acid MW=265)
-
- Sodium sulfite (4.75 g, 37.68 mmol) was added to a suspension of 1-bromo-4-(2-bromo-ethyl)-benzene (5.00 g, 18.94 mmol) in water (60 mL). The reaction mixture was heated at 105-107° C. for 18 hours and then cooled to −5° C. The formed precipitate was removed by filtration, washed with water (50 mL) and dried under vacuum to give 4.33 g (86% yield) of the title compound.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 7.44 (d, 2H), 7.18 (d, 2H), 2.88-2.80 (m, 2H), 2.68-2.61 (m, 2H); MS (ESI) m/z 263 [M−1]−(corresponding sulfonic acid, MW=265).
-
- The title compound was synthesized as described for Example 210 in 5% yield, starting from 2-[2-(4-bromo-phenyl)-ethanesulfonylamino]-5-hydroxymethyl-benzenesulfonamide and 3,3-dimethyl-1-butyne. Purification by preparative HPLC.
- 1HNMR (400 MHz, CD3OD) δ ppm 7.97 (s, 1H), 7.74 (d, 1H), 7.54 (d, 1H), 7.20 (d, 2H), 7.09 (d, 2H), 4.60 (s, 2H), 3.46 (m, 2H), 3.02 (m, 2H), 1.28 (s, 9H); MS (ESI) m/z 449 [M−1]−.
-
- Lithium aluminum hydride (1 M in tetrahydrofuran, 1.9 mL, 1.9 mmol) was added dropwise to a solution of 4-[2-(4-bromo-phenyl)-ethanesulfonylamino]-3-sulfamoyl-benzoic acid (0.22 g, 0.47 mmol) in anhydrous tetrahydrofuran (20 mL) at 0° C. The reaction mixture was stirred at room temperature for 5 hours, cooled to 0° C. and quenched by addition of saturated sodium sulfate solution (20 mL). The precipitate was removed by filtration and the filtrate was extracted with ethyl acetate. The organic phase was washed with brine, dried over sodium sulfate and concentrated under reduced pressure to give 0.15 g (71% yield) of the title compound.
- 1H NMR (400 MHz, CD3OD) δ ppm 7.82 (s, 1H), 7.49 (d, 2H), 7.39 (d, 1H), 7.28 (d, 1H), 7.13 (d, 2H), 4.56 (s, 2H), 3.45 (m, 2H), 3.06 (m, 2H); MS (ESI) m/z 447, 449 [M−1]−.
-
- To a suspension of 2-[2-(4-bromo-phenyl)-ethanesulfonylamino]-5-methyl-benzenesulfonamide (0.3 g, 0.69 mmol) in water (12 mL) was added 2.5 M sodium hydroxide until a clear solution was obtained (˜12 mL). Potassium permanganate (0.55 g, 3.46 mmol) was added and the resulting reaction mixture was heated at 70° C. for 3 hours. The hot solution was filtered through a pad of Celite and the solids were washed with water. Solid sodium bisulfite was added to the filtrate until the pink color disappeared. The aqueous solution was treated with charcoal, filtered, and the pH was adjusted to pH˜1 using 6 M hydrochloric acid. The formed precipitate was collected by filtration, washed with water, and dried to give 0.22 g (69% yield) of the title compound.
- 1H NMR (400 MHz, CD3OD) δ ppm 8.58 (s, 1H), 8.18 (d, 1H), 7.85 (d, 1H), 7.39 (d, 2H), 7.13 (d, 2H), 3.63-3.59 (m, 2H), 3.10-3.06 (m, 2H); MS (ESI) m/z 461, 463 [M−1]−.
-
- The title compound was synthesized as described for Example 210 in 11% yield, starting from 2-[2-(4-bromo-phenyl)-ethanesulfonylamino]-5-hydroxymethyl-benzenesulfonamide and ethynyl-cyclopentane. Purification by preparative HPLC.
- 1H NMR (400 MHz, CD3OD) δ ppm 7.96 (s, 1H), 7.75 (d, 1H), 7.55 (d, 1H), 7.21 (d, 2H), 7.09 (d, 2H), 4.62 (s, 2H), 3.40-3.53 (m, 2H), 2.98-3.10 (m, 2H), 2.68-2.85 (m, 1H), 1.89-2.06 (m, 2H), 1.54-1.83 (m, 6H); MS (ESI) m/z 461 [M−1]−.
-
- To a solution of methyl ester 4-[2-(4-chloro-2-methoxy-phenyl)-ethanesulfonylamino]-3-sulfamoyl-benzoic acid methyl ester (0.18 g, 0.39 mmol) in anhydrous tetrahydrofuran (30 mL) lithium aluminum hydride (0.20 g, 5.27 mmol) was added in small portions at −10° C. The reaction mixture was allowed to warm to room temperature and stirred for 14 hours. The mixture was cooled to −10° C., quenched by the addition of saturated aqueous sodium sulfate, acidified using 2 M hydrochloric acid and extracted with ethyl acetate. The combined extracts were washed with water and brine, dried over sodium sulfate and concentrated under reduced pressure. Purification by preparative HPLC gave 12.8 mg (7.5% yield) of the title compound.
- 1H NMR (400 MHz, (CD3)2O) δ ppm 8.84 (s, 1H), 7.98 (s, 1H), 7.74 (d, 1H), 7.56-7.64 (m, 1H), 7.10-7.22 (m, 3H), 6.83-6.96 (m, 3H), 4.67 (s, 2H), 4.67 (s, 3H), 3.47-3.56 (m, 2H), 3.02-3.09 (m, 2H); MS (ESI) m/z 433, 435 [M−1]−.
-
- To a solution of 4-[2-(4-chloro-2-methoxy-phenyl)-ethanesulfonylamino]-3-sulfamoyl-benzoic acid (0.18 g, 0.40 mmol) in anhydrous methanol (15 mL), thionyl chloride (1 mL) was added and the mixture was heated at reflux for 18 hours. The volatiles were removed under reduced pressure to give 0.18 g (97% yield) of the title compound.
- 1H NMR (400 MHz, CD3OD) δ ppm 8.56 (br. s, 1H), 8.15 (m, 1H), 7.82 (m, 1H), 7.06-7.12 (m, 1H), 6.86 (dd, 1H), 6.82 (dd, 1H), 3.92 (s, 3H), 3.67 (s, 3H), 3.38-3.46 (m, 2H), 3.00-3.10 (m, 2H); MS (ESI) m/z 461, 463 [M−1]−.
-
- The title compound was synthesized as described for Example 211 b) in 34% yield, starting from 2-[2-(4-chloro-2-methoxy-phenyl)-ethanesulfonylamino]-5-methyl-benzenesulfonamide.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.16 (br s., 1H), 8.82 (s., 1H), 8.43 (d, 1H), 8.12 (dd, 1H), 8.08 (br s., 2H), 7.17 (d, 2H), 6.98 (dd, 1H), 6.90 (dd, 1H), 3.67 (s, 3H), 3.47-3.41 (m, 2H), 3.00-2.85 (m, 2H); MS (ESI) m/z 447, 449 [M−1]−.
-
- Palladium on carbon (5 wt. %, 0.4 g) was added to a solution of 2-[2-(4-chloro-2-methoxy-phenyl)-ethenesulfonylamino]-5-methyl-benzenesulfonamide (0.55 g, 1.32 mmol) in ethyl acetate (20 mL) and the reaction mixture was shaken under a hydrogen atmosphere (30 psi) for 4 hours. The solution was filtered through a pad of Celite and concentrated under reduced pressure to give 0.31 g (57% yield) of the title compound.
- 1H NMR (400 MHz, CD3OD) δ ppm 7.77 (d, 1H), 7.63 (d, 1H), 7.41 (d, 1H), 7.08 (d, 1H), 6.88 (d, 1H), 6.83 (dd, 1H), 3.64 (s, 3H), 3.36-3.46 (m, 2H), 2.92-3.05 (m, 2H), 2.37 (s, 3H); MS (ESI) m/z 417, 419 [M−1]−.
-
- 2-(4-Chloro-2-methoxy-phenyl)-ethenesulfonyl chloride (0.50 g, 1.87 mmol) was added slowly to a solution of 2-amino-5-methyl-benzenesulfonamide (0.38 g, 2.05 mmol) in anhydrous pyridine (10 mL) and the reaction mixture was stirred for 18 hours at room temperature. The solvent was removed in vacuo and the residue was dissolved in ethyl acetate (30 mL). The organic phase was washed with 1 M hydrochloric acid and brine, dried over anhydrous sodium sulfate and concentrated under reduced pressure. Purification by column chromatography gave 0.42 g (54% yield) of the title compound.
- 1H NMR (400 MHz, CD3OD) δ ppm 7.70 (s, 1H), 7.52-7.61 (m, 2H), 7.42 (d, 1H), 7.36 (d, 1H), 7.14 (d, 1H), 7.06 (s, 1H), 6.94 (d, 1H), 3.72 (s, 3H).
-
- Thionyl chloride (1.15 mL, 15.83 mmol) was added dropwise to a suspension of sodium 2-(3,4-dichloro-phenyl)-ethanesulfonate (3.17 g, 11.47 mmol) in a mixture of anhydrous benzene (50 mL) and anhydrous N,N-dimethylformamide (1 mL) at 0° C. The reaction mixture was allowed to warm slowly to room temperature and was then heated at 80° C. for 18 hours. The reaction mixture was cooled to room temperature and filtered through a pad of Celite. The solids were washed with benzene and the combined filtrates were concentrated in vacuo. The residue was taken up in hexane and the resulting suspension was heated at 50° C. for 30 min. After cooling to room temperature, a solid was collected by filtration to give 2.20 g (70% yield) of the title compound.
- 1H NMR (400 MHz, CDCl3) δ ppm 7.44 (d, 1H), 7.36 (d, 1H), 7.10 (d, 1H), 3.84-3.93 (m, 2H), 3.27-3.34 (m, 2H); MS (ESI) m/z 253 [M−1]− (corresponding sulfonic acid Mw=255).
-
- The title compound was synthesized as described for Example 210 in 18% yield, starting from 3-[2-(4-bromo-phenyl)-ethanesulfonylamino]-thiophene-2-sulfonic acid amide and 3,3-dimethyl-1-butyne. Purification by column chromatography, using 50% ethyl acetate in hexanes as the eluent, followed by purification by preparative HPLC.
- 1H NMR (400 MHz, CD3OD) δ ppm 7.66 (d, 1H), 7.30 (d, 1H), 7.22 (d, 2H), 7.12 (d, 2H), 3.40 (m, 2H), 3.04 (m, 2H), 1.29 (s, 9H); MS (ESI) m/z 425 [M−1]+.
-
- A mixture of 3-amino-thiophene-2-sulfonamide (0.10 g, 0.56 mmol) and 2-(4-bromo-phenyl)-ethanesulfonyl chloride (0.18 g, 0.62 mmol) in anhydrous pyridine (3 mL) was stirred at room temperature overnight. The reaction mixture was diluted with ethyl acetate (25 mL) and the organic phase was washed with 1 M hydrochloric acid (25 mL), saturated sodium bicarbonate (25 mL) and brine, dried over magnesium sulfate and concentrated under reduced pressure to give 0.16 g (67% yield) of the title compound.
- 1H NMR (400 MHz, CD3OD) δ ppm 7.65 (d, 1H), 7.41 (d, 2H), 7.32 (d, 1H), 7.12 (d, 2H), 3.43 (m, 2H), 3.04 (m, 2H)
-
- A 57% aqueous solution of hydriodic acid (15 mL) was added to neat 3-nitro-thiophene-2-sulfonamide (1.02 g, 4.90 mmol) at room temperature. The reaction mixture was heated at 90° C. for 1 hour, cooled to room temperature and diluted with ethyl acetate (50 mL). The mixture was washed with a solution of sodium thiosulfate and the pH was adjusted to pH˜8 with sodium bicarbonate. The mixture was extracted with 10% methanol in ethyl acetate and the organic phase was dried over magnesium sulfate and concentrated in vacuo. Purification by column chromatography, using a gradient of 30-70% ethyl acetate in hexanes as the eluent, gave 65% yield of the title compound.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 7.54 (d, 1H), 7.29 (s, 2H), 6.62 (d, 1H), 5.85 (s, 2H); MS (ESI) m/z 179 [M+1]+.
-
- Ammonium hydroxide (28% aqueous, 2.4 mL, 17.57 mmol) was added to a stirred solution of 3-nitro-thiophene-2-sulfonyl chloride (1.00 g, 4.39 mmol) in tetrahydrofuran (50 mL) at room temperature. After 15 min the reaction mixture was concentrated under reduced pressure. The residue was triturated in methanol and dichloromethane. The solid was collected by filtration and dried to give 1.02 g (quantitative yield) of the title compound.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 7.93 (d, 1H) 7.74 (d, 1H) 7.58 (br. s, 2H); MS (ESI) m/z 207 [M−1]−.
-
- Water (35 mL) and formic acid (35 mL) were added to a solution of 2-benzylsulfanyl-3-nitro-thiophene (1.00 g, 3.98 mmol) in dichloromethane (70 mL). The reaction mixture was cooled to −5° C. and chlorine gas was bubbled through the heterogeneous mixture with vigorous stirring for 15 min. The reaction mixture was warmed to room temperature and stirred for 30 min under a nitrogen flow to remove excess chlorine gas. Dichloromethane (50 mL) was added and the phases were separated. The organic phase was washed with brine, dried over magnesium sulfate and concentrated in vacou. Purification by column chromatography, using a gradient of 15-30% ethyl acetate in hexanes as the eluent, gave 59% yield of the title compound.
- 1H NMR (400 MHz, CDCl3) δ ppm 7.78 (q, 2H); MS (ESI) m/z 208 [M−1]− (corresponding sulfonic acid, MW=209)
-
- A mixture of 2-chloro-3-nitrothiophene (1.00 g, 6.11 mmol), N,N-diisopropylethylamine (2.1 mL, 12.22 mmol) and benzyl mercaptan (0.8 mL, 6.72 mmol) in dimethyl sulfoxide (5 mL) was μlaced in a sealed tube. The reaction mixture was heated at 80° C. for 1 hour, cooled to room temperature and water (10 mL) was added. The mixture was diluted with ethyl acetate (50 mL) and the organic phase was washed with brine, dried over magnesium sulfate, concentrated in vacuo and dried in vacuo to give 97% yield of the title compound.
- 1H NMR (400 MHz, CDCl3) δ ppm 7.61 (d, 1H), 7.40-7.48 (m, 2H), 7.29-7.41 (m, 5H), 7.07 (d, 1H), 4.27 (s, 2H); MS (ESI) m/z 252 [M+1]+.
-
- Bis(triphenylphosphine)palladium(II) chloride (4.65 mg, 6.62 μmol) was added to a degassed mixture of 3-chloro-2-methoxy-pyridine-5-boronic acid (62.02 mg, 0.33 mmol), sulfonamide 3-(3-bromo-benzenesulfonylamino)-thiophene-2-sulfonic acid amide (146.10 mg, 0.37 mmol), triethylamine (0.10 mL, 0.73 mmol ) in anhydrous ethanol (5 mL). The resulting mixture was heated in a microwave reactor at 140° C. for 10 min, cooled to room temperature, diluted with ethyl acetate (10 mL), filtered through a pad of Celite and concentrated in vacuo. Purification by preparative HPLC, using 0.1% aqueous trifluoroacetic acid/acetonitrile as the eluent, gave 83.3 mg (55% yield) of the title compound.
- 1H NMR (400 MHz, CD3OD) δ ppm 8.31 (d, 1H), 7.96-8.06 (m, 2H), 7.79-7.86 (m, 2H), 7.56-7.64 (m, 2H), 7.33 (d, 1H), 4.04 (s, 3H); MS (ESI) m/z 460, 462 [M+1]+.
-
- A mixture of 3-amino-thiophene-2-sulfonic acid amide (100 mg, 0.56 mmol) and 3-bromo-benzenesulfonyl chloride (97 μL, 0.67 mmol) in anhydrous pyridine (3 mL) was stirred at room temperature for 17 hours. The solvent was removed in vacuo and the residue was taken up in dichloromethane (25 mL). The organic phase was washed with 1 M hydrochloric acid, saturated sodium bicarbonate and brine, dried over sodium sulfate and concentrated under reduced pressure. Purification by column chromatography, using 4-6% methanol in dichloromethane as the eluent, gave 147.6 mg (66% yield) of the title compound.
- 1H NMR (400 MHz, CD3OD) δ ppm 7.87 (d, 1H), 7.68 (m, 2H), 7.53 (d, 1H), 7.34 (t, 1H), 7.16 (d, 1H); MS (ESI) m/z 395, 397 [M−1]− (corresponding sulfonic acid Mw=397).
- Method C
- Method C.1 Synthesis of Arylethenesulfonic Acid Ethyl Esters (F)
- n-Butyllithium (11 mmol, 2.5 M solution in hexanes) was added dropwise to a stirred solution of diethoxy(phosphoryl) methanesulfonic acid ethyl ester E (11 mmol) in 150 mL of tetrahydrofurane at −78° C. The reaction mixture was stirred at −78° C. for 15 min and a solution of the corresponding aldehyde (10 mmol) in 5 mL tetrahydrofuran was added dropwise. After the addition was complete the reaction mixture was warmed to room temperature, stirred for 1 hour and quenched with brine. Most of the tetrahydrofuran was removed in vacuo and dichloromethane (200 mL) was added. The organic phase was separated, washed with brine, dried over magnesium sulfate and the solvent was evaporated. Purification by column chromatography, using hexane/ethyl acetate (2:1) as the eluent, gave ester F.
- Compounds listed in Table 4 were synthesized following this procedure.
-
TABLE 4 Structure Yield Analytical data 50% 1H NMR (400 MHz, CDCl3) δ ppm 7.48-7.65 (m, 3H), 7.22-7.36 (m, 2H), 6.73 (d, 1H), 5.94 (t, 1H), 4.25 (q, 2 H), 1.41 (t, 3H) 90% 1H NMR (400 MHz, CDCl3) δ ppm 7.79 (dd, 1H), 7.40- 7.48 (m, 1H), 7.35 (d, 1H), 7.24-7.31 (m, 1H), 7.16 (d, 1H), 6.56 (t, 1H), 6.48 (d, 1H), 4.15 (q, 2H), 1.25 (t, 3H) 90% 1H NMR (400 MHz, CDCl3) δ ppm 7.77 (d, 1H), 7.34- 7.46 (m, 2H), 6.91-7.01 (m, 3H), 4.67 (m, 1H), 4.22 (q, 2H), 1.37-1.46 (m, 9H) 90% 1H NMR (400 MHz, CDCl3) δ ppm 7.61-7.73 (m, 9H), 6.81 (d, 1H), 4.26 (q, 2H), 1.42 (t, 3H) 65% 1H NMR not available 71% 1H NMR (400 MHz, CDCl3) δ ppm 7.81 (d, 1H), 7.62 (d, 1H), 7.45-7.56 (m, 1H), 7.32-7.43 (m, 2H), 6.85 (d, 1H), 4.25 (q, 2H), 1.42 (t, 3H) 79% 1H NMR (400 MHz, CDCl3) □ ppm 7.77 (d, 1H), 7.33- 7.46 (m, 2H), 6.88-7.07 (m, 3H), 4.19-4.30 (m, 2H), 4.06-4.19 (m, 2H), 1.45-1.55 (m, 3H), 1.32-1.44 (m, 3H) - Method C.2 Synthesis of Arylethenesulfonyl Chlorides (I)
- a) Arylethenesulfonatetetrabutyl-ammonium (H)
- Tetrabutyl-ammonium iodide (12 mmol) was added to a solution of arylethenesulfonic acid ethyl ester F in acetone (500 mL) and the reaction mixture was heated at reflux overnight. The reaction mixture was cooled to room temperature and concentrated under reduced pressure. Purification by column chromatography, using dichloromethane/methanol (95:5) as the eluent. The residue was triturated with diethyl ether to afford H (contaminated with tetrabutylammonium iodide) used in the next step without further purification.
- b) Arylethenesulfonyl chlorides (I)
- Thionyl chloride (45 mmol) was added dropwise at 0° C. to a solution of H (15 mmol) in N,N-dimethylformamide (30 mL). The reaction mixture was stirred at 10-15° C. for 15-45 min, poured onto crushed ice and ethyl acetate (100 mL) was added. The organic phase was separated, washed with brine, dried over magnesium sulfate, and concentrated under reduced pressure. Purification by column chromatography, using 10% ethyl acetate in hexanes as the eluent, followed by recrystallization from pentane/diethyl ether gave the desired sulfonyl chloride I.
- Compounds listed in Table 5 were synthesized following this method.
-
TABLE 5 Yield (over two Structure steps) Analytical data 19% 1H NMR (400 MHz, CDCl3) δ ppm 7.72 (d, 1H), 7.61 (m, 2H), 7.34 (m, 2H), 7.22 (d, 1H), 5.94 (t, 1H) MS (ESI) m/z 299 [M − 1]− (corresponding sulfonic acid MW = 300) CHN Calc.: C, 38.25; H, 2.39. Found: C, 38.18; H, 2.59 (1/22 hexane) 43% 1H NMR (400 MHz, CDCl3) δ ppm 7.86 (d, 1H), 7.54 (d, 1H), 7.38-7.49 (m, 2H), 6.93-7.05 (m, 2H), 4.63-4.78 (m, 1H), 1.44 (d, 6H). MS (ESI) m/z 241 [M − 1]− (corresponding sulfonic acid MW = 242). CHN Calc.: C, 50.67; H, 5.03. Found: C, 50.84; H, 4.43. 53% 1H NMR (400 MHz, CDCl3) δ ppm 7.93 (d, 1H), 7.49-7.64 (m, 2H), 7.27-7.43 (m, 2H), 7.24 (s, 1H), 6.64 (t, 1H). MS (ESI) m/z 249 [M − 1]− (corresponding sulfonic acid MW = 250). CHN Calc.: C, 40.23; H, 2.63. Found: C, 40.55; H, 2.82. 57% 1H NMR (400 MHz, CDCl3) δ ppm 7.65-7.82 (m, 9H), 7.29 (d, 1H) MS (ESI) m/z 327 [M − 1]− (corresponding sulfonic acid MW = 328). CHN Calc.: C, 51.96; H, 2.91. Found: C, 51.99; H, 3.02. 68% 1H NMR (400 MHz, CDCl3) δ ppm 8.26 (d, 1H), 7.52 (d, 1H), 7.38-7.49 (m, 2H) 7.22-7.29 (m, 2H), 2.53 (s, 3H) MS (ESI) m/z 229 [M − 1]− (corresponding sulfonic acid MW = 230). CHN Calc.: C, 43.46; H, 3.65. Found: C, 43.44; H, 4.04 31% 1H NMR (400 MHz, CDCl3) δ ppm 7.93 (d, 1H), 7.64 (dd, 1H), 7.54-7.61 (m, 1H), 7.37-7.45 (m, 2H), 7.34 (d, 1H) MS (ESI) m/z 267 [M − 1]− (corresponding sulfonic acid MW = 268) CHN Calc.: C, 37.71; H, 2.11. Found: C, 37.78; H, 2.21 24% 1H NMR (400 MHz, CDCl3) δ ppm 7.86 (d, 1H), 7.57 (d, 1H), 7.40-7.52 (m, 2H), 6.91-7.11 (m, 2H), 4.19 (q, 2H), 1.42-1.63 (m, 4H) MS (ESI) m/z 227 [M − 1]− (corresponding sulfonic acid MW = 228). CHN Calc.: C, 48.68; H, 4.49. Found: C, 48.84; H, 4.63 - Method D
- A solution of 2-amino-benzenesulfonamide (1 mmol) and styrenyl sulfonyl chloride J (1 mmol) in anhydrous pyridine (2 mL) was stirred at room temperature for 18 hours. The solvent was removed under reduced pressure and the residue was dissolved in dichloromethane. The organic phase was washed with 1 M hydrochloric acid, saturated aqueous sodium bicarbonate and brine, dried over sodium sulfate and concentrated in vacuo. Purification by column chromatography, using 5% methanol in dichloromethane as the eluent, or by preparative HPLC gave the title compound J.
- Compounds listed in Table 6 were synthesized following this procedure.
-
TABLE 6 Example Structure Yield Analytical data 216 77% 1H NMR (400 MHz, DMSO-d6) δ ppm 9.12 (s, 1H), 7.72-7.89 (m, 5H), 7.52-7.66 (m, 3H), 7.39-7.48 (m, 1H), 7.31 (d, 2H), 7.26 (t, 1H), 6.80 (t, 1H) MS (ESI) m/z 455 [M + 1]+ 217 55% 1H NMR (400 MHz, DMSO-d6) δ ppm 9.12 (s, 1H), 7.75-7.85 (m, 4H), 7.38-7.67 (m, 6H), 7.24-7.31 (m, 3H) MS (ESI) m/z 403 [M − 1]− 218 25% 1H NMR (400 MHz, DMSO-d6) δ ppm 9.15 (s, 1H), 7.94 (d, 2H), 7.78-7.88 (m, 9H), 7.56-7.69 (m, 3H), 7.51 (d, 1H), 7.28 (t, 1H) MS (ESI) m/z 481 [M − 1]− 219 42% 1H NMR (400 MHz, CDCl3) δ ppm 8.43 (s, 1H), 7.91-7.97 (m, 1H), 7.78 (m, 1H), 7.68 (d, 1H), 7.50-7.57 (m, 1H), 7.32- 7.42 (m, 2H), 7.14-7.25 (m, 2H), 6.89- 6.97 (m, 2H), 5.26 (s, 2H), 4.64 (m, 1H), 1.37 (d, 6H) MS (ESI) m/z 395 [M − 1]− 220 30% MS (ESI) m/z 383 [M − 1]− CHN Calc.: C, 48.86; H, 4.19. Found: C, 47.02; H, 4.21 221 25% 1H NMR (400 MHz, CD3OD) δ ppm 7.90 (dd, 1H), 7.69-7.76 (m, 2H), 7.63 (d, 1H), 7.48-7.57 (m, 2H), 7.32-7.41 (m, 2H), 7.18-7.28 (m, 2H) MS (ESI) m/z 421 [M − 1] 222 64% 1H NMR (400 MHz, CD3OD) δ ppm 7.85- 7.95 (m, 1H), 7.63-7.77 (m, 2H), 7.48- 7.59 (m, 1H), 7.44 (dd, 1H) 7.29-7.40 (m, 1H), 7.17-7.26 (m, 2H), 6.99 (d, 1H), 6.92 (t, 1H), 4.09 (q, 2H), 1.41 (t, 3H) MS (ESI) m/z 381 [M − 1]− - Method E
- Palladium on carbon (10 wt. %) was added to a solution of styrenyl sulfonamides J (0.2 mmol) in absolute alcohol (10 mL) or in a mixture of absolute alcohol (10 mL) and ethyl acetate (5 mL) and the resulting mixture was shaken on a Parr Shaker under a hydrogen atmosphere (50 psi) for 4 to 16 hours. The reaction mixture was filtered through a pad of Celite and the filtrate was concentrated under reduced pressure. Purification by preparative HPLC gave title compound K.
- Compounds listed in Table 7 were synthesized following this procedure.
-
TABLE 7 Example Structure Yield Analytical data 223 27% 1H NMR (400 MHz, (CD3)2O) δ ppm 7.97 (dd, 1H), 7.81 (d, 1H), 7.63 (m, 1H), 7.34- 7.40 (m, 2H), 7.31 (t, 1H), 7.19 (d, 2H), 6.47 (m, 1H), 3.59-3.70 (m, 2H), 3.09-3.23 (m, 2H) ESMS m/z [M − 1] 455 224 26% 1H NMR (400 MHz, (CD3)2O) δ ppm 7.97 (d, 1H), 7.75-7.89 (m, 4H), 7.61-7.68 (m, 4 H), 7.40 (d, 2H), 7.30 (t, 1H), 3.63-3.73 (m, 2H), 3.15-3.24 (m, 2H) ESMS m/z [M − 1] 483 -
- Boron tribromide (1 M in dichloromethane, 5.4 mL, 5.44 mmol) was added dropwise over 5 min to a suspension of 2-[2-(2-ethoxy-phenyl)-ethenesulfonylamino]-benzenesulfonamide (example 222) (0.21 g, 0.54 mmol) in dichloromethane (10 mL) at −78° C. The reaction mixture was allowed to warm to room temperature and stirred overnight. Cooled to −20° C. and methanol (5 mL) was added slowly. The resulting solution was warmed up to room temperature and heated at 50° C. for 1 hour. The volatiles were removed under reduced pressure and the residue was taken up in methanol (5 mL). The mixture was concentrated in vacuo and the process was repeated once more. The residue was taken up in water (5 mL) and ethyl acetate (15 mL). The organic phase was separated, dried over magnesium sulfate and concentrated in vacuo. Purification by preparative HPLC gave 31 mg (16% yield) of the title compound.
- 1H NMR (400 MHz, CD3OD) δ ppm 7.87 (dd, 1H) 7.65-7.71 (m, 2H) 7.46-7.54 (m, 1H) 7.37 (d, 1H) 7.09-7.24 (m, 3H) 6.79-6.85 (m, 2H); MS (ESI) m/z 355 [M+1]+.
- Method F
- a) General Method for Synthesis of 1,2,4-benzothiadiazine derivatives
- A solution of aniline L (50 mmol) in anhydrous nitromethane (50 mL) was added slowly to a vigorously stirred mixture of chlorosulfonyl isocyanate (58 mmol) in anhydrous nitromethane (150 mL) at −5° C. Anhydrous aluminium chloride (62.5 mmol) was added in small portions to the resulting suspension and the mixture was heated at reflux for 30 min. The hot solution was poured onto ice (800 g) and stirred for 30 min. The precipitate was collected by filtration, washed with water (75 mL) and suspended in aqueous sodium bicarbonate (10 g/200 mL of water). The suspension was heated until most of the precipitate dissolved, treated with charcoal and filtered. The pH of the filtrate was adjusted to pH˜1 using 6 M hydrochloric acid and the solid was collected by filtration, washed with water, air-dried and used in the next step without further purification.
- b) General Procedure for the Synthesis of Amino Sulfonamide Building Blocks
- A suspension of 1,2,4-benzothiadiazine derivative M (20.64 mmol) in aqueous sulfuric acid (50% w/v) was heated at reflux for 17-24 hours until a clear solution was obtained. The reaction mixture was then cooled to 0° C. and the pH was adjusted to pH˜7 using aqueous sodium hydroxide (30% w/v). The crude product was collected by filtration, washed with water, air-dried and purified by column chromatography, using 20-50% ethyl acetate in hexanes as the eluent, or by preparative HPLC.
- Compounds listed in Table 8 were synthesized following this method.
-
TABLE 8 Structure Yield Spectral data 19% 1H NMR (400 MHz, DMSO-d6) δ ppm 7.60 (br. s., 2H), 6.64 (br. s., 2H), 6.34-6.44 (m, 2H) MS (ESI) m/z 207 [M − 1]− 21% 1H NMR (400 MHz, DMSO-d6) δ ppm 7.61 (br. s., 2H), 7.42 (ddd, 1H), 7.17-7.26 (m, 1H), 5.69 (br. s., 2H) MS (ESI) m/z 209 [M + 1]+ 29% 1H NMR (400 MHz, DMSO-d6) ppm 7.49 (dd, 1H), 7.40 (br. s., 2H), 6.77 (dd, 1H), 5.97 (br. s., 2H) MS (ESI) m/z 209 [M + 1]+ 34% 1H NMR (400 MHz, DMSO-d6) δ ppm 7.26 (d, 1H), 7.19 (br.s., 2H), 6.51 (d, 1 H), 5.76 (br.s., 2H), 3.80 (s, 3H) MS (ESI) m/z 221 [M + 1]+ 23% 1H NMR (400 MHz, DMSO-d6) δ ppm 7.45 (s, 2H), 7.40 (d, 1H), 7.19-7.30 (m, 1H), 6.62 (m, 1H), 5.79 (s, 2H) MS (ESI) m/z 198 [M − 1]− 27% 1H NMR (400 MHz, CD3OD) δ ppm 7.34 (d, 1H), 7.24 (d, 1H), 6.99 (dd, 1H) MS (ESI) m/z 299 [M + 1]+ 22% 1H NMR (400 MHz, DMSO-d6) ppm 7.79 (d, 1H), 7.48 (dd, 1H), 7.34 (br. s., 2H), 6.64 (d, 1H), 6.02 (br. s., 2H) MS (ESI) m/z 299 [M + 1]+ - Method G
- Copper(I) cyanide (10 mmol) was added to a solution of iodo-benzenesulfonamide O (5 mmol) in anhydrous pyridine (15 mL) at room temperature and the resulting mixture was heated in a sealed tube at 160° C. for 4 hours. Most of the pyridine was removed in vacuo and the residue was taken up in ethyl acetate (100 mL). The organic phase was washed with water and brine, dried over sodium sulfate and concentrated under reduced pressure. Purification by column chromatography, using 50% ethyl acetate in pentane as the eluent, gave the title compound P.
-
- The title compound was synthesized as described for method G in 31% yield, starting from 2-amino-4-iodo-benzenesulfonamide.
- 1H NMR (400 MHz, (CD3)2O)) δ ppm 7.80 (d, 1H), 7.26 (d, 1H), 7.03 (dd, 1H), 6.77 (br. s, 2H), 6.01 (br. s, 2H); MS (ESI) m/z 196 [M−1]−.
-
- The title compound was synthesized as described for method G in 42% yield, starting from 2-amino-5-iodo-benzenesulfonamide.
- 1H NMR (400 MHz, CD3OD) δ ppm 7.96 (d, 1H), 7.50 (dd, 1H), 6.88 (d, 1H); MS (ESI) m/z 196 [M−1]−
-
- (E)-4-Chloro-2-(2-(4-(difluoromethoxy)phenyl)vinylsulfonamido)benzenesulfonamide (0.426 g, 0.97 mmol) was dissolved in anhydrous tetrahydrofuran (2 mL). Sodium acetate (0.318 g, 3.88 mmol) and p-toluenesulfonic acid hydrazide (0.723 g, 3.88 mmol) were added and the reaction was heated in a microwave at 180° C. for 60 min. The mixture was filtered and the solvent was evaporated under vacuum. Purification by preparative HPLC, gave 41 mg (10% yield) of the title compound.
- 1H NMR (DMSO-d6) δ ppm 9.03 (br. s, 1H) 7.93 (br. s, 2H) 7.86 (d, 1H) 7.67 (d, 1H) 7.41 (d, 1H) 7.33-7.26 (m, 2H) 7.07 (d, 2H) 3.73-3.60 (m, 2H) 3.07-2.91 (m, 2H); MS (ESI) m/z 439 [M−1]−
-
- 2-Amino-4-chlorobenzenesulfonamide (200 mg, 0.97 mmol) and (E)-2-(4-(difluoromethoxy)phenyl)ethenesulfonyl chloride (260 mg, 0.97 mmol) were dissolved in anhydrous dichloromethane (5 mL) and flushed with argon. Anhydrous pyridine (0.391 mL, 4.84 mmol) was added and the reaction was stirred at room temperature under an inert atmosphere over night. The reaction was quenched using 2 M hydrochloric acid (20 mL) and extracted with dichloromethane. The combined organic phase was washed with water, dried over magnesium sulfate and concentrated under vacuum to give 426 mg (100% yield) of the title compound.
- MS (ESI) m/z 437 [M−1]−.
-
- The title compound was synthesized as described for Example 226 in 21% yield, starting from (E)-4-chloro-2-(2-(2-methoxyphenyl)vinylsulfonamido)benzenesulfonamide.
- 1H NMR (DMSO-d6) δ ppm 9.03 (s, 1H) 7.94 (br. s, 2H) 7.88 (d, 1H) 7.66 (d, 1H) 7.42 (d, 1H) 7.23-7.13 (m, 2H) 6.92 (d, 1H) 6.84 (t, 1H) 3.59-3.52 (m, 2H) 3.66 (s, 3H) 3.00-2.93 (m, 2H); MS (ESI) m/z 403 [M−1]−
-
- The title compound was synthesized as described for Example 226 a) starting from 2-amino-4-chlorobenzenesulfonamide and (E)-2-(2-methoxyphenyl)ethenesulfonyl chloride.
- MS (ESI) m/z 401 [M−1]−
-
- The title compound was synthesized as described for Example 226 in 41% yield, starting from (E)-4-chloro-2-(2-(4-chloro-2-methoxyphenyl)vinylsulfonamido)benzenesulfonamide.
- 1H NMR (DMSO-d6) δ ppm 9.03 (br. s, 1H) 7.92 (br. s, 2H) 7.86 (d, 1H) 7.63 (d, 1H) 7.41 (d, 1H) 7.19 (d, 1H) 6.99 (d, 1H) 6.90 (dd, 1H) 3.68 (s, 3H) 3.60-3.51 (m, 2H) 2.98-2.90 (m, 2H); MS (ESI) m/z 437 [M−1]−
-
- The title compound was synthesized as described for Example 226 a) in 100% yield, starting from 2-amino-4-chlorobenzenesulfonamide and (E)-2-(4-chloro-2-methoxyphenyl)ethenesulfonyl chloride.
- MS (ESI) m/z 435 [M−1]−
-
- The title compound was synthesized as described for Example 226 in 3% yield, starting from (E)-2-(2-(3,4-dichlorophenyl)vinylsulfonamido)-3,5-difluorobenzenesulfonamide.
- 1H NMR (DMSO-d6) δ ppm 9.28 (br. s, 1H) 7.81-7.67 (m, 3H) 7.64-7.49 (m, 3H) 7.32 (dd, 1H) 3.62-3.52 (m, 2H) 3.20-3.07 (m, 2H); MS (ESI) m/z 443 [M−1]−
-
- The title compound was synthesized as described for Example 226 a) in 100% yield, starting from 2-amino-4-chlorobenzenesulfonamide and (E)-2-(3,4-dichlorophenyl)ethenesulfonyl chloride.
- MS (ESI) m/z 442 [M−1]−
-
- 2-Amino-5-fluoro-4-methoxybenzenesulfonamide (200 mg, 0.91 mmol) and 2,3-dichlorobenzene-1-sulfonyl chloride (290 mg, 1.18 mmol) were dissolved in anhydrous dichloromethane( 5 mL). Anhydrous triethylamine (0.290 mL, 2.09 mmol) was added and the reaction was stirred at room temperature for 48 hours under an inert atmosphere. The reaction was quenched using 2 M hydrochloric acid (20 mL) and was extracted with dichloromethane. The combined organic phase were washed with water, dried over magnesium sulfate and concentrated under vacuum. Purification by preparative HPLC gave 23 mg (6% yield) of the title compound.
- 1H NMR (CD3OD) δ ppm 8.05 (dd, 1H) 7.53 (dd, 1H) 7.35-7.21 (m, 2H) 6.95 (d, 1H) 3.61 (s, 3H); MS (ESI) m/z 427 [M−1]−
-
- The title compound was synthesized as described for Example 226 in 3% yield, starting from (E)-4-chloro-2-(2-(3,4-dichlorophenyl)vinylsulfonamido)benzenesulfonamide.
- 1H NMR (CD3OD) δ ppm 7.80 (d, 1H) 7.65 (d, 1H) 7.32-7.27 (m, 2H) 7.16 (dd, 1H) 7.05 (dd, 1H) 3.51-3.42 (m, 2H) 3.02-2.95 (m, 2H); MS (ESI) m/z 443 [M−1]−
-
- 2-Amino-4-chlorobenzenesulfonamide (100 mg, 0.48 mmol) and (E)-2-(3,4-dichlorophenyl)ethenesulfonyl chloride (171 mg, 0.63 mmol) were dissolved in anhydrous dichloromethane (5 mL) and were flushed with argon. Anhydrous triethylamine (0.155 mL, 1.11 mmol) was added and the reaction was stirred at room temperature under an inert atmosphere over night. The reaction was quenched using 2 M hydrochloric acid (20 mL) and extracted with dichloromethane. The combined organic phases were washed with water, dried over magnesium sulfate and concentrated under vacuum to give 194 mg (91% yield) of the title compound.
- MS (ESI) m/z 441 [M−1]−
-
- The title compound was synthesized as described for Example 226 in 24% yield, starting from (E)-5-chloro-2-(2-(4-chlorophenyl)vinylsulfonamido)-4-fluorobenzenesulfonamide.
- 1H NMR (CD3OD) δ ppm 7.92 (d, 1H) 7.54 (d, 1H) 7.16-7.04 (m, 4H) 3.51-3.42 (m, 2H) 3.03-2.90 (m, 2H); MS (ESI) m/z 425 [M−1]−
-
- The title compound was synthesized as described for 226 a) in 61% yield, starting from 2-amino-5-chloro-4-fluorobenzenesulfonamide and (E)-2-(4-chlorophenyl)ethenesulfonyl chloride.
- MS (ESI) m/z 423 [M−1]−
-
- The title compound was synthesized as described for Example 226 in 15% yield, starting from (E)-5-chloro-2-(2-(3,4-dichlorophenyl)vinylsulfonamido)-4-fluorobenzenesulfonamide.
- 1H NMR (CD3OD) δ ppm 7.90 (d, 1H) 7.52 (d, 1H) 7.33-7.26 (m, 2H) 7.06 (dd, 1H) 3.51-3.44 (m, 2H) 3.02-2.96 (m, 2H); MS (ESI) m/z 461 [M−1]−
-
- The title compound was synthesized as described for Example 226 a) in 40% yield, starting from 2-amino-5-chloro-4-fluorobenzenesulfonamide and (E)-2-(3,4-dichlorophenyl)ethenesulfonyl chloride.
- MS (ESI) m/z 459 [M−1]−
-
- The title compound was synthesized as described for Example 226 in 40% yield, starting from (E)-5-(2-(4-(Furan-2-yl)phenyl)vinylsulfonamido)-2,3-dihydro-1H-indene-4-sulfonamide.
- 1H NMR (CDCl3) δ ppm 7.61 (d, 2H) 7.46 (dd, 1H) 7.42 (d, 2H) 7.22 (d, 2H) 6.63 (dd, 1H) 6.47 (dd,, 1H) 3.53-3.47 (m, 2H) 3.31-3.19 (m, 4H) 2.94 (t, 2H) 2.19-2.09 (m, 2H); MS (ESI) m/z 445 [M−1]−
-
- The title compound was synthesized as described for Example 226 a) in 18% yield, starting from a mixture of 5-amino-2,3-dihydro-1H-indene-4-sulfonamide compound and 6-amino-2,3-dihydro-1H-indene-5-sulfonamide (1:1). The two isomers were separated by preparative HPLC.
- MS (ESI) m/z 443 [M−1]−
-
- Sulfurisocyanatidic chloride (0.752 mL, 8.63 mmol) and anhydrous nitroethane (10 mL) were mixed, cooled to −5° C. (ice/salt-bath) and a solution of 2,3-dihydro-1H-inden-5-amine (1 g, 7.51 mmol) in anhydrous nitroethane (10 mL) was added dropwise under vigorous stirring. When the addition was completed, anhydrous aluminum chloride (1.251 g, 9.39 mmol) was added and the suspension was heated at 110° C. for 60 min. The solution was cooled to room temperature and diluted with water (50 mL) and ethyl acetate (50 mL). The mixture was filtered and the layers were separated. The organic phase was dried over magnesium sulfate and concentrated under vacuum. The residue was dissolved in sulfuric acid and water (1:1 30 mL) and heated at 140° C. for 90 min before it was allowed to reach room temperature and stirred over night. The mixture was neutralized using 15% sodium hydroxide and extracted with ethyl acetate. The combined organic phases were dried over magnesium sulfate and concentrated under vacuum to give 0.664 g (21% yield) of the title mixture.
- MS (ESI) m/z 211 [M−1]−
-
- The title compound was synthesized as described for Example 226 in 47% yield, starting from (E)-6-(2-(4-(furan-2-yl)phenyl)vinylsulfonamido)-2,3-dihydro-1H-indene-5-sulfonamide. Heated in a microwave at 180° C. for 15 min. The mixture was filtered and the solvent was evaporated under vacuum.
- 1H NMR (CDCl3) δ ppm 7.80 (s, 1H) 7.61 (d, 2H) 7.49-7.43 (m, 2H) 7.23 (d, 2H) 6.63 (dd, 1H) 6.47 (dd, 1H) 3.61-3.47 (m, 2H) 3.27-3.18 (m, 2H) 3.02-2.89 (m, 4H) 2.23-2.04 (m, 2H); MS (ESI) m/z 445 [M−1]−
-
- The title compound was synthesized as described for Example 226 a) in 18% yield, starting from a mixture of 5-amino-2,3-dihydro-1H-indene-4-sulfonamide compound and 6-amino-2,3-dihydro-1H-indene-5-sulfonamide (1:1) The two isomers were separated by preparative HPLC.
- MS (ESI) m/z 443 [M−1]−
-
- The title compound was synthesized as described for Example 226 in 20% yield, starting from a mixture of (E)-5-(2-(3,4-dichlorophenyl)vinylsulfonamido)-2,3-dihydro-1H-indene-4-sulfonamide compound and (E)-6-(2-(3,4-dichlorophenyl)vinylsulfonamido)-2,3-dihydro-1H-indene-5-sulfonamide (1:1). Purification by preparative HPLC.
- 1H NMR (CD3OD) δ ppm 7.50 (d, 1H) 7.39-7.29 (m, 3H) 7.08 (dd, 1H) 3.51-3.42 (m, 2H) 3.30-3.21 (m, 2H) 3.05-2.99 (m, 2H) 2.90 (t, 2H) 2.02-2.16 (m, 2H); MS (ESI) m/z 447 [M−1]−
-
- The title compound was synthesized as described for Example 226 a) in 38% yield, starting from a mixture of 5-amino-2,3-dihydro-1H-indene-4-sulfonamide compound and 6-amino-2,3-dihydro- 1H-indene-5-sulfonamide (1:1).
- MS (ESI) m/z 445 [M−1]−
-
- The title compound was synthesized as described for Example 226 a) in 41% yield, starting from a mixture of 5-amino-2,3-dihydro-1H-indene-4-sulfonamide compound with 6-amino-2,3-dihydro-1H-indene-5-sulfonamide (1:1). Purification by preparative HPLC.
- 1H NMR (CDCl3) δ ppm 7.79 (s, 1H) 7.55 (d, 1H) 7.48 (s, 1H) 7.46-7.37 (m, 3H) 6.94 (d, 1H) 5.05 (s, 1H) 2.94 (ddd, 4H) 2.22-2.07 (m, 2H); MS (ESI) m/z 411 [M−1]−
- (E)-6-(2-(4-Chlorophenyl)vinylsulfonamido)-2,3-dihydro-1H-indene-5-sulfonamide
- The title compound was synthesized as described for Example 226 a) in 60% yield, starting from a mixture of 5-amino-2,3-dihydro-1H-indene-4-sulfonamide compound with 6-amino-2,3-dihydro-1H-indene-5-sulfonamide (1:1). Purification by preparative HPLC.
- 1H NMR (CDCl3) δ ppm 8.72 (s, 1H) 7.43 (d, 1H) 7.37-7.27 (m, 4H) 6.82 (d, 1H) 5.00 (s, 1H) 3.19 (t, 2H) 2.84 (t, 2H) 2.09-2.00 (m, 2H); MS (ESI) m/z411 [M−1]−
-
- The title compound was synthesized by the analogous preparation of Example 148 in 47% yield, starting from 3-(3,4-dichlorophenyl)benzenesulfonyl chloride.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 9.30 (br. s, 1H), 8.19 (br. s, 1H), 8.03 (d, 1H), 7.90-7.98 (m, 3H), 7.77 (d, 1H), 7.66-7.74 (m, 3H), 7.58-7.66 (m, 2H), 7.29 (t, 1H), 2.30 (d, 3H); MS m/z 469, 471 [M−1]−
-
- The title compound was synthesized as described for Example 179 a) in 28% yield, starting from 2-(2-(6-bromopyridin-3-yl)ethylsulfonamido)benzenesulfonamide and 4-ethynyl-alpha,alpha,alpha-trifluorotoluene. Purification by preparative HPLC.
- 1H NMR (400 MHz, DMSO-d6) δ ppm 8.98 (s, 1H) 8.51 (d, 1H) 7.89 (dd, 1H) 7.81-7.86 (m, 6H) 7.77 (dd, 1H) 7.59-7.69 (m, 3H) 7.32-7.37 (m, 1H) 3.67-3.74 (m, 2H) 3.06-3.12 (m, 2H); MS (ESI) m/z 508 [M−1]−.
- Assays for Determining Biological Activity
- Inhibition of Prostaglandin E Synthase Activity
- Compounds were tested as inhibitors of microsomal prostaglandin E synthase activity in microsomal prostaglandin E synthase assays and whole cell assays. These assays measure prostaglandin E2 (PGE2) synthesis which is taken as a measure of prostaglandin E synthase activity. Microsomal prostaglandin E synthase biochemical assays used microsomal prostaglandin E synthase-1 in microsomal preparations. The source of the microsomes can be for example interleukin-1β-stimulated human A549 cells (which express human mPGES-1) or Sf9 cells transfected with plasmids encoding human mPGES-1 cDNA. The whole blood assay [described by Patrignani, P. et al., Journal of Pharmacology and Experimental Therapeutics, 1994, vol. 271, pp 1705-1712] was used as the whole cell assay for testing the compounds. Whole blood provides a protein and cell rich milieu for the study of biochemical efficacy of anti-inflammatory compounds such as prostaglandin synthase inhibitors. To study the inhibitory activities of these compounds, human blood was stimulated with lipopolysaccharide (LPS) for typically 16 hours to induce mPGES-1 expression, after which the concentration of produced PGE2 was measured by competitive-immuno assay (homogeneous time-resolved fluorescence, HTRF) as read out for effectiveness against mPGES-1-dependent PGE2 production.
- Microsomal Prostaglandin E Synthase Biochemical Assay
- A solution of test compound was added to a diluted microsome preparation containing human mPGES-1 and pre-incubated for 15 minutes in potassium phosphate buffer pH 6.8 with cofactor glutathione (GSH). Corresponding solutions without test compound were used as positive controls, and corresponding solutions without test compound and without microsomes were used as negative controls. The enzymatic reaction was then started by addition of the substrate PGH2 in an organic solution (dry acetonitrile).
- The typical reaction conditions of the enzymatic reaction were thus: Test compound: ranging from 60 μM to 0.002 μM, or zero in positive and negative controls; potassium phosphate buffer pH 6.8: 50 mM; GSH: 2.5 mM; mPGES-1-containing microsomes: 2 μg/mL (sample and positive controls) or 0 μg/mL (negative control); PGH2: 10.8 μM; Acetonitrile: 7.7% (v/v); DMSO: 0.6% (v/v). The reaction was stopped after one minute by adding an acidic solution (pH 1.9) of ferric chloride and citrate (final concentrations 7 mM and 47 mM respectively), by which the PGH2 was sequestered (the PGH2 is reduced to mainly 12-hydroxy heptadecatrineoic acid (12-HHT) which is not detected by the subsequent PGE2 detection step). The resulting solution was then pH neutralized by addition of potassium phosphate buffer, prior to diluting an aliquot of the resulting solution in a weak potassium phosphate buffer (50 mM, pH 6.8) containing 0.2% BSA (w/v). [Adapted from Jacobsson et al., Proc. Natl. Acad. Sci. USA, 1999, vol. 96, pp. 7220-7225]The PGE2 formed was quantified by use of a commercial HTRF based kit (catalogue #62PG2PEC or #62P2APEC from Cisbio International). 100% activity was defined as the PGE2 production in positive controls subtracted by the PGE2 production in the negative controls. IC50 values were then determined using standard procedures.
- Data from this assay for representative compounds is shown in the Table below. The potency is expressed as IC50 and the value indicated is an average of at least n=2. The data indicate that the compounds of the invention are expected to possess useful therapeutic properties.
-
Example No. IC50 (μM) 1 0.13 2 0.27 3 0.14 4 0.079 5 0.04 6 0.074 7 0.28 8 2.2 9 0.61 10 0.45 11 0.59 12 1 13 0.52 14 3.1 15 7.6 16 0.19 17 5.3 18 0.044 19 1.4 20 0.7 21 0.32 22 0.43 23 0.27 24 0.3 25 0.077 26 0.42 27 0.27 28 0.29 29 0.28 30 5.9 31 5.7 32 5.1 33 1.1 34 0.12 35 0.17 36 0.057 37 0.16 38 0.22 39 1.4 40 1.2 41 1.1 42 1.6 43 0.11 44 0.75 45 1.3 46 1.1 47 1.6 48 0.59 49 0.036 50 0.13 51 4.9 52 0.084 53 0.16 54 0.27 55 0.36 56 0.53 57 0.62 58 0.3 59 2.3 60 2.4 61 2.4 62 2.5 63 3 64 3.1 65 3.8 66 6.2 67 6.4 68 9.1 69 0.27 70 0.22 71 0.3 72 0.49 73 0.29 74 9.8 75 2.6 76 1.2 77 3.1 78 0.35 79 9.9 80 0.57 81 2.9 82 7.3 83 0.43 84 0.19 85 0.8 86 0.7 87 2.4 88 1.3 89 2.8 90 0.87 91 0.31 92 0.091 93 0.65 94 0.65 95 0.31 96 4.8 97 3.4 98 0.68 99 0.29 100 4 101 1.6 102 0.85 103 9.2 104 4.6 105 0.67 106 0.074 107 0.14 108 4.9 109 6.1 110 0.49 111 0.76 112 6.1 113 0.48 114 0.83 115 2.6 116 1.7 117 1.3 118 4.7 119 4.5 120 3.3 121 0.22 122 6.7 123 9.3 124 1.3 125 7.2 126 1.2 127 5.1 128 1.7 129 2.5 130 5.2 131 6.6 132 4.5 133 2.2 134 3.7 135 8.3 136 3.8 137 2.1 138 0.57 139 1.8 140 2.9 141 3.1 142 4.9 143 1.1 144 1.1 145 4.1 146 4.7 147 3.9 148 0.38 149 0.65 150 4.2 151 3.7 152 3.9 153 3.2 154 9.3 155 9.8 156 5.4 157 1.1 158 8.7 159 3.5 160 1.2 161 1.3 162 5.7 163 0.46 164 0.19 165 2.3 166 4.8 167 3.8 168 0.63 169 0.022 170 0.29 171 1.7 172 7.3 173 1.5 174 0.22 175 3.7 176 0.9 177 2.2 178 2.7 179 6.2 180 1.4 181 0.37 182 0.52 183 0.081 184 9.6 185 2.5 186 0.25 187 0.68 188 0.27 189 1.4 190 0.68 191 8.3 192 3.2 193 7.9 194 1 195 5.5 196 1.9 197 0.64 198 0.29 199 2.5 200 1.4 201 0.72 202 2.9 203 2.1 204 0.62 205 0.56 206 9.1 207 0.097 208 0.055 209 0.22 210 0.36 211 0.075 212 0.041 213 0.33 214 1.4 215 0.74 216 0.13 217 0.11 218 0.18 219 0.95 220 0.27 221 0.42 222 0.38 223 0.42 224 0.1 225 6.3 226 1.3 227 1.9 228 0.13 229 5.5 230 6.1 231 0.13 232 0.87 233 0.15 234 6.2 235 6.2 236 4.6 237 8.9 238 2.1 239 6.06 240 0.031 - Whole blood assay
- Human blood collected from human volunteers in heparinized tubes was incubated with 100 μM acetyl salicylic acid, in order to inhibit the constitutively expressed cyclooxygenase (COX)-1/COX-2 enzymes, and then stimulated with 0.1 μg/ml LPS to induce the expression of enzymes along the COX-2 pathway, e.g., COX-2 and mPGES-1. 100 μL of this blood was added to the wells of a 384-well μlate containing 1 μL DMSO solutions of compounds typically in the final concentration range 316 μM to 0.01 μM. Naproxen was used as reference compound. The mix was incubated at 37° C. for 16 hours. Plasma was harvested by centrifugation and stored at −70° C. until further analysis of PGE2 levels. For the calculations, the 0%-activity value was represented by blood treated with acetyl salicylic acid, LPS and the reference compound (1 mM Naproxen). The 100%-activity value was represented by blood treated with aspirin, LPS and DMSO. [Reference: Patrignani, P. et al., Journal of Pharmacology and Experimental Therapeutics, 1994, vol. 271, pp 1705-1712]. The PGE2 formed was quantified, after dilution in a weak potassium phosphate buffer (50 mM, pH 6.8) containing 0.2% BSA (w/v), by use of a commercial HTRF based kit (catalogue #62PG2PEC or #62P2APEC from Cisbio International). IC50 values were then determined using standard procedures.
Claims (26)
1. A compound of formula (I) or a pharmaceutically acceptable salt thereof
wherein:
A is selected from phenyl or a 5- or 6-membered heteroaryl or a 5- or 6-membered heterocyclenyl moiety; said phenyl or 5- or 6-membered heteroaryl or 5- or 6-membered heterocyclenyl moiety in group A being optionally fused to a phenyl, a 5- or 6-membered heteroaryl, C5-6carbocyclyl or C5-6heterocyclyl ring;
each R1 is independently selected from halogen, nitro, SF5, CN, NR5R6, OH, CHO, CO2C1-4alkyl, CONR5R6, C1-4alkyl, C2-4alkenyl, C2-4alkynyl, C1-4alkoxy, G3, OG3 or OCH2G3; said C1-4alkyl, C2-4alkenyl, C2-4alkynyl or C1-4alkoxy being optionally substituted by one or more substituents selected from OH, NR5R6, C1-4alkyl or F;
m represents an integer 0, 1, 2, 3 or 4;
each R3is independently selected from hydrogen, CN and C1-4alkyl; said C1-4alkyl being optionally substituted with OH, CN, C1-4alkoxy, NR7R8, or one or more F atoms;
L1 represents a direct bond, C1-4alkylene, C2-4alkenylene or C2-4alkynylene;
L2 represents a direct bond, —O—, —OCH2—, C1-2alkylene, —C≡C— or —NHCONH—;
G1 represents phenyl, 5- or 6-membered heteroaryl, C3-10carbocyclyl or C5-8heterocyclyl;
G2 represents H, C1-6alkyl, phenyl, 5- or 6-membered heteroaryl, C3-10carbocyclyl or C5-8heterocyclyl; said C1-6alkyl being optionally further substituted by one or more groups selected from OH, C1-6alkoxy and halogen;
the phenyl, heteroaryl, carbocyclyl or heterocyclyl moieties in G1 and G2 being optionally fused to one or two further rings independently selected from phenyl, a 5- or 6-membered heteroaryl, C5-6carbocyclyl or C5-6 heterocyclyl ring;
any phenyl, heteroaryl, carbocyclyl or heterocyclyl moieties in G1 and G2 being optionally substituted by one or more substituents independently selected from halogen, OH, CN, NO2, CO2R9, C1-6alkyl, C1-6alkoxy, C1-4thioalkoxy, SO2NR10R11, NR12R13, —NHCOC(OH)(CH3)CF3 or —CH2OCH2CF2CHF2; said C1-6alkyl or C1-6alkoxy being optionally substituted by OH or by one or more F atoms;
R9 represents C1-6alkyl;
G3 represents phenyl or 5- or 6-membered heteroaryl; and
each, R4, R5, R6, R7, R8, R10, R11, R12 and R13 is independently selected from H or C1-4alkyl;
with the proviso that the following compounds are excluded:
3-[(phenylsulfonyl)amino]pyridine-2-sulfonamide;
2-amino-N-(2-sulfamoylphenyl)benzenesulfonamide;
4-tert-butyl-N-(2-sulfamoylphenyl)benzenesulfonamide;
3-amino-4-hydroxy-N-(2-sulfamoylphenyl)benzenesulfonamide;
4-bromo-N-(2-sulfamoylphenyl)benzenesulfonamide;
4-fluoro-N-(2-sulfamoylphenyl)benzenesulfonamide;
4-methyl-N-(2-sulfamoylphenyl)benzenesulfonamide;
4-nitro-N-(2-sulfamoylphenyl)benzenesulfonamide;
2-nitro-N-(2-sulfamoylphenyl)benzenesulfonamide;
4-amino-N-(2-sulfamoylphenyl)benzenesulfonamide;
2-[(phenylsulfonyl)amino]-4-(trifluoromethyl)benzenesulfonamide;
2-{[(4-methylphenyl)sulfonyl]amino }-4-(trifluoromethyl)benzenesulfonamide; N-(2-sulfamoylphenyl)quinoline-8-sulfonamide;
4-tert-butyl-N-(2-(N-ethylsulfamoyl)phenyl)-2,6-dimethylbenzenesulfonamide;
2,5-dibromo-N-(2-(N-ethylsulfamoyl)phenyl)benzenesulfonamide;
methyl 4-(5-fluoro-3-methylbenzo[b]thiophene-2-sulfonamido)-3-sulfamoylbenzoate;
2,3,4,5,6-pentafluoro-N-(2-sulfamoylphenyl)benzenesulfonamide;
N-(2-(N,N-dimethylsulfamoyl)phenyl)-5,7-dimethyl-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide;
N-(2-(N-tert-butylsulfamoyl)phenyl)-5,7-dimethyl-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide;
N-(2-(N-methoxy-N-methylsulfamoyl)phenyl)-5,7-dimethyl-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide;
5,7-dimethyl-N-(2-sulfamoylphenyl)-[1,2,4]triazolo[1,5-c]pyrimidine-2-sulfonamide;
6-methyl-N-(2-sulfamoylphenyl)thiazolo[3,2-b][1,2,4]triazole-2-sulfonamide;
6-chloro-N-(2-(N,N-dimethylsulfamoyl)phenyl)-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide;
6-chloro-N-(2-(N-ethyl-N-methylsulfamoyl)phenyl)-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide;
N-(2-(N,N-dimethylsulfamoyl)phenyl)-5-methyl-7,8-dihydro-6H-cyclopenta[e][1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide;
N-(2-(N,N-dimethylsulfamoyl)-5-(trifluoromethyl)phenyl)-5,7-dimethyl-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide;
N-(2-(N-ethyl-N-methylsulfamoyl)phenyl)-5,7-dimethyl-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide; and
N-(2-(N,N-dimethylsulfamoyl)phenyl)-5,7-dimethyl-[1,2,4]triazolo[1,5-a]pyrimidine-2-sulfonamide.
2. A compound according to claim 1 wherein each R3 represents hydrogen.
3. A compound according to claim 1 wherein A represents phenyl, pyridyl, thienyl or 2,3-dihydro-indenyl.
4. A compound according to claim 1 wherein m represents an integer 0, 1 or 2.
5. A compound according to claim 1 wherein L1 represents a direct bond, C1-3alkylene or C2-3alkenylene.
6. A compound according to claim 1 wherein L2 represents a direct bond, —O—, —OCH2—, —NHCONH— or —C≡C—.
7. A compound according to claim 1 wherein G1 represents phenyl or 5- or 6-membered heteroaryl, said phenyl or 5- or 6-membered heteroaryl being optionally fused to one or two further rings independently selected from phenyl, C5-6carbocyclyl or C5-6 heterocyclyl ring.
8. A compound according to claim 7 wherein G1 represents phenyl, naphtyl, pyridyl, thienyl, 2,3-dihydro-indenyl tetrahydronaphtyl, 2,3-dihydro-1,4-benzodioxinyl or S,S, -dioxodibenzothienyl.
9. A compound according to claim 1 wherein G2 represents H, C1-6alkyl, phenyl, 5- or 6-membered heteroaryl, C3-10carbocyclyl or C5-8heterocyclyl;
said phenyl or 5- or 6-membered heteroaryl being optionally fused to one further ring independently selected from phenyl, a 5- or 6-membered heteroaryl, C5-6carbocyclyl and C5-6 heterocyclyl ring;
said C1-6alkyl being optionally further substituted by one or more groups selected from OH, C1-6alkoxy and halogen.
10. A compound according to claim 9 wherein G2 represents hydrogen, methyl, ethyl, tert-butyl, iso-propyl, phenyl, pyridyl, pyrimidyl, cyclopentyl, cyclopentenyl, cyclohexyl, furanyl, tetrahydrofuranyl, thienyl, thiazolyl, pyrazolyl, bensofuranyl, 2,3-dihydro-bensofuranyl, indolyl, tetrahydronaphtyl or dibenzofuranyl.
11. A compound according to claim 1 wherein R5 and R6 is hydrogen.
12. A compound according to claim 1 wherein R10 is hydrogen.
13. A compound according to claim 1 wherein R11, R12 and R13 is methyl.
14. A compound according to claim 1 wherein R1 is independently selected from halogen, CN, OH, CONR5R6, NR5R6, C1-4alkyl, C2-4alkynyl, C1-4alkoxy, G3, OG3 and OCH2G ; said C1-4alkyl or C2-4alkynyl being optionally substituted by OH.
15. A compound according to claim 1 wherein G3 is phenyl, pyridyl or 2-methyl-1,3,4-oxadiazole.
16. A compound according to claim 1 wherein any phenyl, heteroaryl, carbocyclyl or heterocyclyl moieties in G1 and G2 are optionally substituted by one or more substituents independently selected from halogen, CN, OH, C1-4alkyl, C1thioalkoxy, NR12R13, —NHCOC(OH)(CH3)CF3, SO2NR10R11l , and C 1alkoxy; said C1-4alkyl or C1alkoxy being optionally substituted by OH or by one or more F atoms.
17. A compound according to claim 1 wherein A is phenyl, pyridyl, thienyl or 2,3-dihydro-indenyl;
R1 is independently selected from halogen, CN, OH, CONR5R6, NR5R6, C1-4alkyl, C2-4alkynyl, C1-4alkoxy, G3, OG3 and OCH2G3; said C1-4alkyl or C2-4alkynyl being optionally substituted by one or more substituents selected from OH and C1-4alkyl;
m represents an integer 0, 1 or 2;
G3 is phenyl, pyridyl or 2-methyl-1,3,4-oxadiazole;
R3, R5, R6 and R10 represents hydrogen;
L1 represents a direct bond, C1-3alkylene or C2-3alkenylene;
L3 represents a direct bond, —O—, —OCH2—, —NHCONH— or —C≡C—;
G1 represents phenyl or 5- or 6-membered heteroaryl; said phenyl or 5- or 6-membered heteroaryl being optionally fused to one or two further rings independently selected from phenyl, C5-6carbocyclyl or C5-6 heterocyclyl ring;
G2 represents H, C1-6alkyl, phenyl, 5- or 6-membered heteroaryl, C3-10carbocyclyl or C5-8heterocyclyl; said phenyl or 5- or 6-membered heteroaryl being optionally fused to one further ring independently selected from phenyl, a 5- or 6-membered heteroaryl, C5-6carbocyclyl or C5-6 heterocyclyl ring; said C1-6alkyl being optionally further substituted by one or more groups selected from OH, C1-6alkoxy and halogen;
any phenyl, heteroaryl, carbocyclyl or heterocyclyl moieties in G1 and G2 being optionally substituted by one or more substituents independently selected from halogen, CN, OH, C1-4alkyl, C1thioalkoxy, NR12R13, —NHCOC(OH)(CH3)CF3, SO2NR10R11, and C1alkoxy; said C1-4alkyl or C1alkoxy being optionally substituted by OH or by one or more F atoms; and
R11, R12 and R13 are methyl.
18. A compound according to claim 1 wherein A is phenyl, pyridyl, thienyl or 2,3-dihydro-indenyl;
R1 is independently selected from chloro, fluoro, bromo, CN, OH, CONR5R6, methyl, 1-butynyl, methoxy, G3, OG3 or OCH2G3; said methyl and 1-butynyl being optionally substituted by OH or methyl;
m represents an integer 0, 1 or 2;
G3 is phenyl, pyridyl or 2-methyl-1,3,4-oxadiazole;
R3, R5, R6 and R10 represents hydrogen;
L1 represents a direct bond, ethylene, 1-methylethylene, 2-methylethylene, ethenylene, —C(CH3)CH —, —CHC(CH3)— or —CH2C(CH2)—;
L2 represents a direct bond, —O—, —OCH2—, —NHCONH— or —C≡C—;
G1 represents phenyl, naphtyl, pyridyl, thienyl, 2,3-dihydro-indenyl tetrahydronaphtyl, 2,3-dihydro-1,4-benzodioxinyl or S,S,-dioxodibenzothienyl;
G2 represents hydrogen, methyl, ethyl, tert-butyl, iso-propyl phenyl, pyridyl, pyrimidyl, cyclopentyl, cyclopentenyl, cyclohexyl, furanyl, tetrahydrofuranyl, thienyl, thiazolyl, pyrazolyl, bensofuranyl, 2,3-dihydro-bensofuranyl, indolyl, tetrahydronaphtyl or dibenzofuranyl;
any methyl, ethyl, tert-butyl, iso-propyl phenyl, pyridyl, pyrimidyl, cyclopentyl, cyclopentenyl, cyclohexyl, furanyl, tetrahydrofuranyl, thienyl, thiazolyl, pyrazolyl, bensofuranyl, 2,3-dihydro-bensofuranyl, indolyl, tetrahydronaphtyl or dibenzofuranyl in G1 and G2 being optionally substituted by one or more substituents independently selected from bromo, chloro, fluoro, CN, OH, methyl, tert-butyl, methylthio, NR12R13, —NHCOC(OH)(CH3)CF3, SO2NR10R11, and methoxy; said methyl, tert-butyl or methoxy being optionally substituted by OH or by one or more F atoms; and
R11, R12 and R13 methyl.
19. A compound selected from the group consisting of:
2-[2-(4-Benzofuran-2-ylphenyl)ethylsulfonylamino]benzenesulfonamide;
2-[2-[4-(2,3-Dihydrobenzofuran-2-yl)phenyl]ethylsulfonylamino]benzenesulfonamide;
2-[[(E)-2-(4-Benzofuran-2-ylphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
2-[[(E)-2-(4-Benzofuran-2-ylphenyl)ethenyl]sulfonylamino]-5-fluoro-benzenesulfonamide;
2-[[(E)-2-(4-Benzofuran-2-ylphenyl)ethenyl]sulfonylamino]-4-fluoro-benzenesulfonamide;
2-[[(E)-2-(4-Benzofuran-2-ylphenyl)ethenyl]sulfonylamino]-6-fluoro-benzenesulfonamide;
2-[1-(4-Chlorophenyl)propan-2-ylsulfonylamino]benzenesulfonamide;
2-(1-Phenylpropan-2-ylsulfonylamino)benzenesulfonamide;
2-[[(E)-1-(4-Chlorophenyl)prop-1-en-2-yl]sulfonylamino]benzenesulfonamide;
2-[1-(4-Chlorophenyl)propan-2-ylsulfonylamino]-4-fluoro-benzenesulfonamide;
2-[[(E)-1-(4-chlorophenyl)prop-1-en-2-yl]sulfonylamino]-4-fluoro-benzenesulfonamide;
2-[[(E)-1-(4-Chlorophenyl)prop-1-en-2-yl]sulfonylamino]-5-fluoro-benzenesulfonamide;
2-[[(E)-1-(4-Chlorophenyl)prop-1-en-2-yl]sulfonylamino]-6-fluoro-benzenesulfonamide;
2-[[(E)-1-(4-Chlorophenyl)prop-1-en-2-yl]sulfonylamino]-3-fluoro-benzenesulfonamide;
2-[[(E)-1-(4-Chlorophenyl)prop-1-en-2-yl]sulfonylamino]-5-methyl-benzenesulfonamide;
2-[2-[4-(2-Furyl)phenyl]ethylsulfonylamino]benzenesulfonamide;
2-[2-[4-(Oxolan-2-yl)phenyl]ethylsulfonylamino]benzenesulfonamide;
2-[[(E)-2-[4-(2-Furyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide;
2-[2-[4-(Difluoromethoxy)phenyl]ethylsulfonylamino]benzenesulfonamide;
2-[[(E)-2-[4-(Difluoromethoxy)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
2-[2-(4-Chlorophenyl)ethylsulfonylamino]-5-fluoro-benzenesulfonamide;
2-[[(E)-2-(4-Chlorophenyl)ethenyl]sulfonylamino]-5-fluoro-benzenesulfonamide;
2-[[(E)-2-(3,4-Dichlorophenyl)ethenyl]sulfonylamino]-4-fluoro-benzenesulfonamide;
2-[[(E)-2-(3,4-Dichlorophenyl)ethenyl]sulfonylamino]-5-fluoro-benzenesulfonamide;
2-[[(E)-2-(3,4-Dichlorophenyl)ethenyl]sulfonylamino]-6-fluoro-benzenesulfonamide;
2-[[(E)-2-(5-Bromo-2-methoxy-phenyl)ethenyl]sulfonylamino]-4-fluoro-benzenesulfonamide;
2-[[(E)-2-(5-Bromo-2-methoxy-phenyl)ethenyl]sulfonylamino]-5-fluoro-benzenesulfonamide;
2-[[(E)-2-(5-Bromo-2-methoxy-phenyl)ethenyl]sulfonylamino]-6-fluoro-benzenesulfonamide;
2-[[(E)-2-(5-Bromo-2-methoxy-phenyl)ethenyl]sulfonylamino]benzenesulfonamide;
2-[[(E)-2-(4-Cyanophenyl)ethenyl]sulfonylamino]-4-fluoro-benzenesulfonamide;
2-[[(E)-2-(4-Cyanophenyl)ethenyl]sulfonylamino]-5-fluoro-benzenesulfonamide;
2-[[(E)-2-(4-Cyanophenyl)ethenyl]sulfonylamino]-6-fluoro-benzenesulfonamide;
2-[[(E)-2-(4-Cyanophenyl)ethenyl]sulfonylamino]benzenesulfonamide;
4-Fluoro-2-[[(E)-2-[4-(2-furyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide;
5-Fluoro-2-[[(E)-2-[4-(2-furyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide;
2-Fluoro-6-[[(E)-2-[4-(2-furyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide;
4-Fluoro-2-[[(E)-2-(4-methylsulfanylphenyl)ethenyl]sulfonylamino]-benzenesulfonamide;
5-Fluoro-2-[[(E)-2-(4-methylsulfanylphenyl)ethenyl]sulfonylamino]-benzenesulfonamide;
2-Fluoro-6-[[(E)-2-(4-methylsulfanylphenyl)ethenyl]sulfonylamino]-benzenesulfonamide;
2-[[(E)-2-(4-Methylsulfanylphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
2-[[(E)-2-[4-(Difluoromethoxy)phenyl]ethenyl]sulfonylamino]-4-fluoro-benzenesulfonamide;
2-[[(E)-2-[4-(Difluoromethoxy)phenyl]ethenyl]sulfonylamino]-5-fluoro-benzenesulfonamide;
2-[[(E)-2-[4-(Difluoromethoxy)phenyl]ethenyl]sulfonylamino]-6-fluoro-benzenesulfonamide;
2-[2-(4-Chlorophenyl)ethylsulfonylamino]-5-(hydroxymethyl)-benzenesulfonamide;
2-[2-(4-Chloro-phenyl)-ethanesulfonylamino]-5-methylbenzenesulfonamide;
2-[2-(4-Chlorophenyl)ethylsulfonylamino]-5-(pyridin-2-ylmethoxy)benzenesulfonamide;
2-[2-(4-Chlorophenyl)ethylsulfonylamino]-5-[(5-methyl-1,3,4-oxadiazol-2-yl)methoxy]benzenesulfonamide;
2-[2-(2-Chloro-phenyl)-ethenesulfonylamino]-benzenesulfonamide;
2-[2-(4-Chloro-2-methoxy-phenyl)-ethenesulfonylamino]-benzenesulfonamide;
2-Fluoro-6-[[(E)-2-(4-phenylphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
2-[[(E)-2-(3-Chlorophenyl)ethenyl]sulfonylamino]-5-fluoro-benzenesulfonamide;
5-Fluoro-2-[[(E)-2-(4-phenylphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
4-Fluoro-2-[[(E)-2-(4-phenylphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
5-Fluoro-2-[[(E)-2-[3-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
5-Fluoro-2-[[(E)-2-(2-methoxyphenyl)ethenyl]sulfonylamino]-benzenesulfonamide;
2-[[(E)-2-(3-Chlorophenyl)ethenyl]sulfonylamino]-6-fluoro-benzenesulfonamide;
2-Fluoro-6-[[(E)-2-(2-methoxyphenyl)ethenyl]sulfonylamino]-benzenesulfonamide;
4-Fluoro-2-[[(E)-2-(2-methoxyphenyl)ethenyl]sulfonylamino]-benzenesulfonamide;
3-Fluoro-2-[[(E)-2-(4-phenylphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
3-Fluoro-2-[[(E)-2-(2-methoxyphenyl)ethenyl]sulfonylamino]-benzenesulfonamide;
4-Fluoro-2-[[(E)-2-[3-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
2-Fluoro-6-[[(E)-2-(2-fluorophenyl)ethenyl]sulfonylamino]benzenesulfonamide;
2-[[(E)-2-(3-Chlorophenyl)ethenyl]sulfonylamino]-5-methyl-benzenesulfonamide;
2-[[(E)-2-(3-Chlorophenyl)ethenyl]sulfonylamino]-3-fluoro-benzenesulfonamide;
3-Fluoro-2-[[(E)-2-[3-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
2-Fluoro-6-[[(E)-2-[3-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
4-Fluoro-2-[[(E)-2-(2-fluorophenyl)ethenyl]sulfonylamino]benzenesulfonamide;
5-Methyl-2-[[(E)-2-[3-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
2-[2-(4-Cyclopentylphenyl)ethylsulfonylamino]benzenesulfonamide;
2-[2-(4-Tert-butylphenyl)ethylsulfonylamino]benzenesulfonamide;
4-Fluoro-2-[2-(4-tert-butylphenyl)ethylsulfonylamino]benzenesulfonamide;
2-[2-(4-Chlorophenyl)ethylsulfonylamino]benzenesulfonamide;
2-[[(E)-2-(4-Chlorophenyl)ethenyl]sulfonylamino]benzenesulfonamide;
2-(Phenethylsulfonylamino)benzenesulfonamide;
2-[[(E)-2-Phenylethenyl]sulfonylamino]benzenesulfonamide;
2-[2-(2-Chlorophenyl)ethylsulfonylamino]benzenesulfonamide;
2-[[(E)-2-(4-Methoxyphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
2-[[(E)-2-(4-Chlorophenyl)ethenyl]sulfonylamino]-4-fluoro-benzenesulfonamide;
2-(2-Phenylpropylsulfonylamino)benzenesulfonamide;
2-[[(E)-2-(4-Chlorophenyl)ethenyl]sulfonylamino]-6-fluoro-benzenesulfonamide;
2-[[(E)-2-(2,6-Dichlorophenyl)ethenyl]sulfonylamino]benzenesulfonamide;
2-[[(E)-2-(4-Methylphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
2-[[(E)-2-(2-Methoxyphenyl)ethenyl]sulfonylamino]benzenesulfonamide;
2-[[(E)-2-Naphthalen-2-ylethenyl]sulfonylamino]benzenesulfonamide;
2-[2-(4-Chlorophenyl)ethylsulfonylamino]-4-fluoro-benzenesulfonamide;
2-[[(E)-2-(4-Chlorophenyl)prop-1-enyl]sulfonylamino]benzenesulfonamide;
2-[2-(4-Chlorophenyl)prop-2-enylsulfonylamino]benzenesulfonamide;
2-Fluoro-6-[[(E)-2-naphthalen-2-ylethenyl]sulfonylamino]benzenesulfonamide;
2-Fluoro-6-[[(E)-2-[2-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
2-Fluoro-6-[[(E)-2-[4-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
2-[[(E)-2-(4-Bromophenyl)ethenyl]sulfonylamino]-6-fluoro-benzenesulfonamide;
4-Fluoro-2-[[(E)-2-naphthalen-2-ylethenyl]sulfonylamino]benzenesulfonamide;
4-Fluoro-2-[[(E)-2-[2-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
4-Fluoro-2-[[(E)-2-[4-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
2-[[(E)-2-(4-Bromophenyl)ethenyl]sulfonylamino]-4-fluoro-benzenesulfonamide;
5-Methyl-2-[[(E)-2-naphthalen-2-ylethenyl]sulfonylamino]benzenesulfonamide;
5-Methyl-2-[[(E)-2-[4-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
2-[[(E)-2-(4-Bromophenyl)ethenyl]sulfonylamino]-5-methyl-benzenesulfonamide;
5-Fluoro-2-[[(E)-2-naphthalen-2-ylethenyl]sulfonylamino]benzenesulfonamide;
5-Fluoro-2-[[(E)-2-[2-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
5-Fluoro-2-[[(E)-2-[4-(trifluoromethyl)phenyl]ethenyl]sulfonylamino]-benzenesulfonamide;
2-[[(E)-2-(4-Bromophenyl)ethenyl]sulfonylamino]-5-fluoro-benzenesulfonamide;
2-[[(E)-2-(2,6-Difluorophenyl)ethenyl]sulfonylamino]benzenesulfonamide;
2-[[(E)-2-[2-(Trifluoromethyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide;
2-[[(E)-2-[4-(Trifluoromethyl)phenyl]ethenyl]sulfonylamino]benzenesulfonamide;
2-[[(E)-2-(4-Bromophenyl)ethenyl]sulfonylamino]benzenesulfonamide;
2-[(E)-2-(4-Bromophenyl) ethylsulfonylamino]benzenesulfonamide;
2-[2-(4-Chlorophenyl)propylsulfonylamino]benzenesulfonamide;
2-[2-(2-Methoxyphenyl)ethylsulfonylamino]benzenesulfonamide;
2-(2-Naphthalen-2-ylethylsulfonylamino)benzenesulfonamide;
4-Fluoro-2-(2-naphthalen-2-ylethylsulfonylamino)benzenesulfonamide;
2-[(4-Chlorophenyl)methylsulfonylamino]benzenesulfonamide;
2-[[3-(2,3-Dichlorophenyl)phenyl]sulfonylamino]benzenesulfonamide;
2-[[3-(3,5-difluorophenyl)phenyl]sulfonylamino]benzenesulfonamide;
3-(5-Chloro-2-methoxy-phenyl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
2-[[3-(3-Cyanophenyl)phenyl]sulfonylamino]benzenesulfonamide;
2-[[3-(4-Cyanophenyl)phenyl]sulfonylamino]benzenesulfonamide;
3-(2,4-Dimethoxypyrimidin-5-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
2-[[3-(3-Furyl)phenyl]sulfonylamino]benzenesulfonamide;
2-[[3-(1H-Indol-5-yl)phenyl]sulfonylamino]benzenesulfonamide;
2-[[3-[3-(Trifluoromethoxy)phenyl]phenyl]sulfonylamino]benzenesulfonamide;
N-(2-Sulfamoylphenyl)-2,3-dihydro-1H-indene-5-sulfonamide;
7-Methyl-N-(2-sulfamoylphenyl)-10-oxa-7-azabicyclo[4.4.0]deca-1,3,5-triene-3-sulfonamide;
2-(3,4-Dichlorophenyl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
2-[[4-[3-Chloro-5-(trifluoromethyl)pyridin-2-yl]oxyphenyl]sulfonylamino]benzenesulfonamide;
N-(2-Sulfamoylphenyl)tetralin-2-sulfonamide;
4-Phenylmethoxy-N-(2-sulfamoylphenyl)benzenesulfonamide;
2-[(4-Cyclohexylphenyl)sulfonylamino]benzenesulfonamide;
3-Phenyl-N-(2-sulfamoylphenyl)benzenesulfonamide;
5-(1-Methyl-5-trifluoromethyl-1H-pyrazol-3-yl)-thiophene-2-sulfonic acid (2-sulfamoyl-phenyl)-amide;
N-(2-Sulfamoylphenyl)-4-(trifluoromethyl)benzenesulfonamide;
2-[[3-(2,5-Dichlorophenyl)phenyl]sulfonylamino]benzenesulfonamide;
3-Dibenzofuran-4-yl-N-(2-sulfamoylphenyl)benzenesulfonamide;
2-[[3-[4-(Trifluoromethyl)phenyl]phenyl]sulfonylamino]benzenesulfonamide;
3-(3-Methoxyphenyl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
3-Benzofuran-2-yl-N-(2-sulfamoylphenyl)benzenesulfonamide;
2-[[3-(2,3-Dihydrobenzofuran-5-yl)phenyl]sulfonylamino]benzenesulfonamide;
3-(6-Methoxypyridin-3-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
2-[[3-(2,4-Dichlorophenyl)phenyl]sulfonylamino]benzenesulfonamide;
3-(1-Methylindol-2-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
2-[[3-[3-(Trifluoromethyl)phenyl]phenyl]sulfonylamino]benzenesulfonamide;
4-Benzofuran-2-yl-N-(2-sulfamoylphenyl)benzenesulfonamide;
2-[[3-(2,4-Difluorophenyl)phenyl]sulfonylamino]benzenesulfonamide;
5-Bromo-2-[[3-(3,4-dichlorophenyl)phenyl]sulfonylamino]benzenesulfonamide;
2-[[3-(3,4-Dichlorophenyl)phenyl]sulfonylamino]-5-phenyl-benzenesulfonamide;
2,3-Difluoro-N-(2-sulfamoylphenyl)benzenesulfonamide;
2,3-Difluoro-N-(4-fluoro-2-sulfamoyl-phenyl)benzenesulfonamide;
3-Chloro-2-fluoro-N-(2-sulfamoylphenyl)benzenesulfonamide;
2,3-Dichloro-N-(2-sulfamoylphenyl)benzenesulfonamide;
2-[[3-(3,4-Dichlorophenyl)phenyl]sulfonylamino]-5-methyl-benzenesulfonamide;
4-[[3-(3,4-Dichlorophenyl)phenyl]sulfonylamino]-3-sulfamoyl-benzamide;
5-Methyl-2-[[3-[3-(trifluoromethoxy)phenyl]phenyl]sulfonylamino]-benzenesulfonamide;
2-(2,3-Dichloro-benzenesulfonylamino)-5-hydroxymethyl-benzenesulfonamide;
2-(2,3-Dichloro-benzenesulfonylamino)-5-methyl-benzenesulfonamide;
2-[[3-(3,4-Dichlorophenyl)phenyl]sulfonylamino]-5-phenoxy-benzenesulfonamide;
5-Phenoxy-2-[[3-[3-(trifluoromethoxy)phenyl]phenyl]sulfonylamino]-benzenesulfonamide;
2-(2,3-Dichloro-benzenesulfonylamino)-5-hydroxy-benzenesulfonamide;
(2R)—N-[2-Chloro-4-[(2-sulfamoylphenyl)sulfamoyl]phenyl]-3,3,3-trifluoro-2-hydroxy-2-methyl-propanamide;
5-Chloro-N-(2-sulfamoylphenyl)thiophene-2-sulfonamide;
4,5-Dichloro-N-(2-sulfamoylphenyl)thiophene-2-sulfonamide;
3,4-Dichloro-N-(2-sulfamoylphenyl)benzenesulfonamide;
3-(6-Methoxypyridin-2-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
2-[[4-[(2-Chloro 1,3-thiazol-5-yl)methoxy]phenyl]sulfonylamino]-benzenesulfonamide;
3-(5-Fluoro-6-methoxy-pyridin-3-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
3-(2-Methoxypyrimidin-5-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
3-(4-Methylpyridin-3-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
3-(2-Methoxypyridin-3-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
2-[[3-(5-Mhloropyridin-3-yl)phenyl]sulfonylamino]benzenesulfonamide;
3-(5-Chloro-6-methoxy-pyridin-3-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
3-(6-Dimethylaminopyridin-3-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
2-[[3-[3-(Hydroxymethyl)phenyl]phenyl]sulfonylamino]benzenesulfonamide;
2-[[3-[4-(Hydroxymethyl)phenyl]phenyl]sulfonylamino]benzenesulfonamide;
2-[[4-(3,4-Dichlorophenyl)phenyl]sulfonylamino]benzenesulfonamide;
2-[[3-(3,4-Dichlorophenyl)phenyl]sulfonylamino]benzenesulfonamide;
2-[[4-(4-Chlorophenoxy)phenyl]sulfonylamino]benzenesulfonamide;
3-(4-Chlorophenyl)-1-[3-methyl-4-[(2-sulfamoylphenyl)sulfamoyl]phenyl]urea;
N-Methyl-N′-(2-sulfamoylphenyl)dibenzo[b,d]thiophene-2,8-disulfonamide 5,5-dioxide;
2,3-Dichloro-N-(4,5-difluoro-2-sulfamoylphenyl)benzenesulfonamide;
(E)-2-(2-(3,4-Dichlorophenyl)vinylsulfonamido)-5-(3-hydroxy-3-methylbut-1-ynyl)benzenesulfonamide;
4-Cyano-2-(2-(3,4-dichlorophenyl)ethylsulfonamido)benzenesulfonamide;
2-(2-(4-Chloro-2-methoxyphenyl)ethylsulfonamido)-4-cyanobenzenesulfonamide;
2-(2-(4-Chloro-2-methoxyphenyl)ethylsulfonamido)-5-cyanobenzenesulfonamide;
2-(2-(4-(3,3-dimethylbut-1-ynyl)phenyl)ethylsulfonamido)benzenesulfonamide;
2-(2-(4-(3-Hydroxy-3-methylbut-1-ynyl)phenyl)ethylsulfonamido)benzenesulfonamide;
3-(3,3-Dimethylbut-1-ynyl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
2-(2-(4-Chloro-2-methoxyphenyl)ethylsulfonamido)benzenesulfonamide;
2-(2-(6-(3,3-Dimethylbut-1-ynyl)pyridin-3-yl)ethylsulfonamido)benzenesulfonamide;
2-(2-(6-(cyclohexylethynyl)pyridin-3-yl)ethylsulfonamido)benzenesulfonamide;
2-(2-(4-Bromophenyl)ethylsulfonamido)-5-methylbenzenesulfonamide;
(E)-2-(2-(4-Bromophenyl)vinylsulfonamido)-5-methylbenzenesulfonamide;
3-(2-Methylthiazol-4-yl)-N-(2-sulfamoylphenyl)benzenesulfonamide;
(E)-2-(2-(3-(Trifluoromethyl)phenyl)vinylsulfonamido)benzenesulfonamide;
(E)-2-(2-(2-Fluorophenyl)vinylsulfonamido)benzenesulfonamide;
5-Chloro-2-(2-(4-chloro-2-methoxyphenyl)ethylsulfonamido)benzenesulfonamide;
2,3-Dichloro-N-(4-chloro-2-sulfamoylphenyl)benzenesulfonamide;
2,3-Dichloro-N-(3,5-difluoro-2-sulfamoylphenyl)benzenesulfonamide;
2-(2-(3,4-Dichlorophenyl)ethylsulfonamido)-4,6-difluorobenzenesulfonamide;
2-(2-(4-Chloro-2-methoxyphenyl)ethylsulfonamido)-4,6-difluorobenzenesulfonamide;
5-Cyano-2-(2-(3,4-dichlorophenyl)ethylsulfonamido)benzenesulfonamide; (E)-2-(2-(3,4-Dichlorophenyl)vinylsulfonamido)-4-methylbenzenesulfonamide;
2-(2-(4-Chloro-2-methoxyphenyl)ethylsulfonamido)-4-methylbenzenesulfonamide;
5-Chloro-2-(2-(3,4-dichlorophenyl)ethylsulfonamido)benzenesulfonamide;
4-(3-(5-Chloro-6-methoxypyridin-3-yl)phenylsulfonamido)pyridine-3-sulfonamide;
4-(2-(4-(Benzofuran-2-yl)phenyl)ethylsulfonamido)pyridine-3-sulfonamide;
2-(2-(4-Chloro-2-methoxyphenyl)ethylsulfonamido)-5-methylbenzenesulfonamide;
4-(2-(4-Chloro-2-methoxyphenyl)ethylsulfonamido)pyridine-3-sulfonamide;
(E)-2-(2-(4-Cyclopentenylphenyl)vinylsulfonamido)benzenesulfonamide;
(E)-2-(2-(4-(3,3-Dimethylbut-1-ynyl)phenyl)vinylsulfonamido)benzenesulfonamide;
(E)-2-(2-(4-(3-Hydroxy-3-methylbut-1-ynyl)phenyl)vinylsulfonamido)- benzenesulfonamide;
2-{2-[4-(3,3-Dimethyl-but-1-ynyl)-phenyl]-ethanesulfonylamino}-5-methyl-benzenesulfonamide;
2-{2-[4-(3,3-Dimethyl-but-1-ynyl)-phenyl]-ethanesulfonylamino}-5-hydroxymethyl-benzenesulfonamide;
2-[2-(4-Cyclopentylethynyl-phenyl)-ethanesulfonylamino]-5-hydroxymethyl-benzenesulfonamide;
2-[2-(4-Chloro-2-methoxy-phenyl)-ethanesulfonylamino]-5-hydroxymethyl-benzenesulfonamide;
3-{2-[4-(3,3-Dimethyl-but-1-ynyl)-phenyl]-ethanesulfonylamino}-thiophene-2-sulfonic acid amide;
3-[3-(5-Chloro-6-methoxy-pyridin-3-yl)-benzenesulfonylamino]-thiophene-2-sulfonic acid amide;
(E)-2-(2-(4-(1,1,2,2-tetrafluoroethoxy)phenyl)vinylsulfonamido)benzenesulfonamide;
(E)-2-(2-(2-(difluoromethoxy)phenyl)vinylsulfonamido)benzenesulfonamide;
(E)-2-(2-(4′-(trifluoromethyl)biphenyl-4-yl)vinylsulfonamido)benzenesulfonamide;
(E)-2-(2-(2-isopropoxyphenyl)vinylsulfonamido)benzenesulfonamide;
(E)-2-(2-(2-(methylthio)phenyl)vinylsulfonamido)benzenesulfonamide;
(E)-2-(2-(2-(trifluoromethoxy)phenyl)vinylsulfonamido)benzenesulfonamide;
(E)-2-(2-(2-ethoxyphenyl)vinylsulfonamido)benzenesulfonamide;
2-(2-(4-(1,1,2,2-tetrafluoroethoxy)phenyl)ethylsulfonamido)benzenesulfonamide;
2-(2-(4′-(trifluoromethyl)biphenyl-4-yl)ethylsulfonamido)benzenesulfonamide;
2-[2-(2-Hydroxy-phenyl)-ethenesulfonylamino]-benzenesulfonamide;
4-Chloro-2-(2-(4-(difluoromethoxy)phenyl)ethylsulfonamido)benzenesulfonamide;
4-Chloro-2-(2-(2-methoxyphenyl)ethylsulfonamido)benzenesulfonamide;
4-Chloro-2-(2-(4-chloro-2-methoxyphenyl)ethylsulfonamido)benzenesulfonamide;
2-(2-(3,4-Dichlorophenyl)ethylsulfonamido)-3,5-difluorobenzenesulfonamide;
2,3-Dichloro-N-(4-fluoro-5-methoxy-2-sulfamoylphenyl)benzenesulfonamide;
4-Chloro-2-(2-(3,4-dichlorophenyl)ethylsulfonamido)benzenesulfonamid;
5-Chloro-2-(2-(4-chlorophenyl)ethylsulfonamido)-4-fluorobenzenesulfonamide;
5-Chloro-2-(2-(3,4-dichlorophenyl)ethylsulfonamido)-4-fluorobenzenesulfonamide;
5-(2-(4-(Furan-2-yl)phenyl)ethylsulfonamido)-2,3-dihydro-1H-indene-4-sulfonamide;
6-(2-(4-(Furan-2-yl)phenyl)ethylsulfonamido)-2,3-dihydro-1H-indene-5-sulfonamid;
5-(2-(3,4-Dichlorophenyl)ethylsulfonamido)-2,3-dihydro-1H-indene-4-sulfonamide;
(E)-5-(2-(4-Chlorophenyl)vinylsulfonamido)-2,3-dihydro-1H-indene-4-sulfonamide;
(E)-6-(2-(4-Chlorophenyl)vinylsulfonamido)-2,3-dihydro-1H-indene-5-sulfonamide;
2-[[3-(3,4-Dichlorophenyl)phenyl]sulfonylamino]-N-methyl-benzenesulfonamide; and
2-(2-(6-((4-(Trifluoromethyl)phenyl)ethynyl)pyridin-3-yl)ethylsulfonamido)-benzenesulfonamide; or a pharmaceutically acceptable salt thereof.
20. A process for the preparation of a compound of formula (I) or a pharmaceutically acceptable salt thereof as defined in claim 1 which comprises,
(a) reacting a compound of formula (II)
wherein L1, L2, G1 and G2 are as defined in formula (I) and X represents a leaving group such as a halogen; or
(b) when L2 represents a direct bond and G1 and G2 are both aromatic moieties, reacting a compound of formula (IV)
wherein Hal represents a halogen atom and R1, R3, A, m and L1 are as defined in formula (I), with a nucleophile G2-M wherein M represents an organo-tin or organo boronic acid group; and optionally after (a) or (b) carrying out one or more of the following:
converting the compound obtained to a further compound of the invention
forming a pharmaceutically acceptable salt of the compound.
21. A pharmaceutical composition comprising a compound of formula (I) or a pharmaceutically acceptable salt thereof as claimed in claim 1 in association with a pharmaceutically acceptable adjuvant, diluent or carrier.
22. A process for the preparation of a pharmaceutical composition comprising mixing the compound of claim 1 or a pharmaceutically acceptable salt thereof with a pharmaceutically acceptable adjuvant, diluent or carrier.
23. A method for treating acute or chronic pain, moniceptive pain or neuropathic pain, comprising administering to a patient in need thereof a therapeutically effective amount of the compound of claim 1 or a pharmaceutically acceptable salt thereof.
24. A method for treating osteoarthritis, rheumatoid arthritis, benign or malignant neoplasias, apnea, sudden infant death (SID), atherosclerosis, cancer, aneurysm, hyperthermia, myositis, Alzheimer's disease or arthritis, comprising administering to a patient in need thereof a therapeutically effective amount of the compound of claim 1 or a pharmaceutically acceptable salt thereof.
25. A method of treating, or reducing the risk of, an inflammatory disease or condition comprising administering to a patient in need thereof a therapeutically effective amount of the compound of claim 1 or a pharmaceutically acceptable salt thereof.
26. A method of treating, or reducing the risk of, human diseases or conditions in which modulation of microsomal prostaglandin E synthase-1 activity is beneficial comprising administering to a patient in need thereof a therapeutically effective amount of the compound of claim 1 or a pharmaceutically acceptable salt thereof.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US12/337,125 US20090163586A1 (en) | 2007-12-20 | 2008-12-17 | Bis-(Sulfonylamino) Derivatives in Therapy 205 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US1528707P | 2007-12-20 | 2007-12-20 | |
| US12/337,125 US20090163586A1 (en) | 2007-12-20 | 2008-12-17 | Bis-(Sulfonylamino) Derivatives in Therapy 205 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| US20090163586A1 true US20090163586A1 (en) | 2009-06-25 |
Family
ID=40789387
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/337,125 Abandoned US20090163586A1 (en) | 2007-12-20 | 2008-12-17 | Bis-(Sulfonylamino) Derivatives in Therapy 205 |
| US12/747,049 Expired - Fee Related US9145380B2 (en) | 2007-12-20 | 2008-12-18 | Bis-(sulfonylamino) derivatives for use in therapy |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/747,049 Expired - Fee Related US9145380B2 (en) | 2007-12-20 | 2008-12-18 | Bis-(sulfonylamino) derivatives for use in therapy |
Country Status (23)
| Country | Link |
|---|---|
| US (2) | US20090163586A1 (en) |
| EP (1) | EP2234993B1 (en) |
| JP (1) | JP5555175B2 (en) |
| KR (1) | KR20100098711A (en) |
| CN (1) | CN101945859B (en) |
| AR (1) | AR069919A1 (en) |
| AU (1) | AU2008341173B2 (en) |
| BR (1) | BRPI0821299A2 (en) |
| CA (1) | CA2709510A1 (en) |
| CL (1) | CL2008003848A1 (en) |
| CO (1) | CO6270307A2 (en) |
| CR (1) | CR11517A (en) |
| DO (1) | DOP2010000191A (en) |
| EA (1) | EA017940B1 (en) |
| EC (1) | ECSP10010272A (en) |
| ES (1) | ES2397502T3 (en) |
| IL (1) | IL205758A0 (en) |
| NI (1) | NI201000109A (en) |
| PE (1) | PE20091175A1 (en) |
| TW (1) | TW200932210A (en) |
| UY (1) | UY31546A1 (en) |
| WO (1) | WO2009082347A1 (en) |
| ZA (1) | ZA201004226B (en) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20100292279A1 (en) * | 2009-05-14 | 2010-11-18 | Astrazeneca Ab | Bis-(Sulfonylamino) Derivatives in Therapy |
| US20110021540A1 (en) * | 2007-11-15 | 2011-01-27 | Astrazeneca Ab | Bis-(Sulfonylamino) Derivatives in Therapy 066 |
| WO2014062044A1 (en) | 2012-10-15 | 2014-04-24 | Vilnius University | Fluorinated benzenesulfonamides as inhibitors of carbonic anhydrase |
| WO2016085392A1 (en) * | 2014-11-27 | 2016-06-02 | Acturum Life Science AB | Bis(sulfonamide) derivatives and their use as mpges inhibitors |
| WO2016085391A1 (en) * | 2014-11-27 | 2016-06-02 | Acturum Life Science AB | Bis(sulfonamide) derivatives and their use as mpges inhibitors |
Families Citing this family (44)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU2007271734B2 (en) | 2006-07-05 | 2012-03-29 | Certa Therapeutics Pty. Ltd. | Therapeutic compounds |
| EP3045170B1 (en) | 2007-12-21 | 2018-07-04 | Fibrotech Therapeutics PTY LTD | Halogenated analogues of anti-fibrotic agents |
| WO2010144959A1 (en) * | 2009-06-18 | 2010-12-23 | Fibrotech Therapeutics Pty Ltd | Analogues of anti-fibrotic agents |
| CN105153188B (en) | 2009-10-22 | 2018-06-01 | 法博太科制药有限公司 | The fused ring analogs of antifibrotic agents |
| CN102838567B (en) * | 2011-06-23 | 2014-12-31 | 深圳市湘雅生物医药研究院 | Benzenesulfonyl piperazine compounds or benzoyl piperazine compounds, preparation methods and uses thereof |
| CA2904489C (en) | 2013-03-18 | 2017-02-14 | Nippon Soda Co., Ltd. | Method for producing disulfonylamine alkali metal salt |
| US10590100B2 (en) * | 2015-11-11 | 2020-03-17 | Ambient Photonics, Inc. | Benzofuran derivatives for the treatment of CNS and other disorders |
| EP3577103A1 (en) | 2017-02-03 | 2019-12-11 | Certa Therapeutics Pty Ltd. | Anti-fibrotic compounds |
| WO2020263830A1 (en) | 2019-06-25 | 2020-12-30 | Gilead Sciences, Inc. | Flt3l-fc fusion proteins and methods of use |
| CN118178645A (en) | 2019-10-18 | 2024-06-14 | 四十七公司 | Combination therapy for the treatment of myelodysplastic syndrome and acute myeloid leukemia |
| KR20220091576A (en) | 2019-10-31 | 2022-06-30 | 포티 세븐, 인코포레이티드 | Anti-CD47 and Anti-CD20 Based Treatment of Hematological Cancers |
| TWI778443B (en) | 2019-11-12 | 2022-09-21 | 美商基利科學股份有限公司 | Mcl1 inhibitors |
| HRP20241518T1 (en) | 2019-12-24 | 2025-01-03 | Carna Biosciences, Inc. | Diacylglycerol kinase modulating compounds |
| PH12022552122A1 (en) | 2020-02-14 | 2024-01-29 | Jounce Therapeutics Inc | Antibodies and fusion proteins that bind to ccr8 and uses thereof |
| US12110294B2 (en) | 2020-05-01 | 2024-10-08 | Gilead Sciences, Inc. | CD73 compounds |
| TW202302145A (en) | 2021-04-14 | 2023-01-16 | 美商基利科學股份有限公司 | Co-inhibition of cd47/sirpα binding and nedd8-activating enzyme e1 regulatory subunit for the treatment of cancer |
| WO2022245671A1 (en) | 2021-05-18 | 2022-11-24 | Gilead Sciences, Inc. | Methods of using flt3l-fc fusion proteins |
| WO2022271659A1 (en) | 2021-06-23 | 2022-12-29 | Gilead Sciences, Inc. | Diacylglyercol kinase modulating compounds |
| US11976072B2 (en) | 2021-06-23 | 2024-05-07 | Gilead Sciences, Inc. | Diacylglycerol kinase modulating compounds |
| EP4359413A1 (en) | 2021-06-23 | 2024-05-01 | Gilead Sciences, Inc. | Diacylglyercol kinase modulating compounds |
| US11932634B2 (en) | 2021-06-23 | 2024-03-19 | Gilead Sciences, Inc. | Diacylglycerol kinase modulating compounds |
| US20230183216A1 (en) | 2021-10-28 | 2023-06-15 | Gilead Sciences, Inc. | Pyridizin-3(2h)-one derivatives |
| CR20240173A (en) | 2021-10-29 | 2024-06-20 | Gilead Sciences Inc | CD73 COMPOUNDS |
| JP2024546851A (en) | 2021-12-22 | 2024-12-26 | ギリアード サイエンシーズ, インコーポレイテッド | IKAROS ZINC FINGER FAMILY DEGRADANT AND USES THEREOF |
| CN118488948A (en) | 2021-12-22 | 2024-08-13 | 吉利德科学公司 | IKAROS zinc finger family degraders and their uses |
| TW202340168A (en) | 2022-01-28 | 2023-10-16 | 美商基利科學股份有限公司 | Parp7 inhibitors |
| JP2025509610A (en) | 2022-03-17 | 2025-04-11 | ギリアード サイエンシーズ, インコーポレイテッド | IKAROS ZINC FINGER FAMILY DEGRADANT AND USES THEREOF |
| WO2023183817A1 (en) | 2022-03-24 | 2023-09-28 | Gilead Sciences, Inc. | Combination therapy for treating trop-2 expressing cancers |
| TWI876305B (en) | 2022-04-05 | 2025-03-11 | 美商基利科學股份有限公司 | Combination therapy for treating colorectal cancer |
| JP2025513258A (en) | 2022-04-21 | 2025-04-24 | ギリアード サイエンシーズ, インコーポレイテッド | KRA G12D modulating compounds |
| CN117285484A (en) * | 2022-06-15 | 2023-12-26 | 上海医药工业研究院有限公司 | Benzothiadiazine 1, 1-dioxide compounds inhibiting HDAC6 enzyme, preparation method and application thereof |
| IL317958A (en) | 2022-07-01 | 2025-02-01 | Gilead Sciences Inc | Cd73 compounds |
| US20240091351A1 (en) | 2022-09-21 | 2024-03-21 | Gilead Sciences, Inc. | FOCAL IONIZING RADIATION AND CD47/SIRPa DISRUPTION ANTICANCER COMBINATION THERAPY |
| KR20250122479A (en) | 2022-12-22 | 2025-08-13 | 길리애드 사이언시즈, 인코포레이티드 | PRMT5 inhibitors and uses thereof |
| CN120882725A (en) | 2023-04-11 | 2025-10-31 | 吉利德科学公司 | KRAS-regulated compounds |
| CN121079300A (en) | 2023-04-21 | 2025-12-05 | 吉利德科学公司 | PRMT5 inhibitors and their uses |
| WO2025006720A1 (en) | 2023-06-30 | 2025-01-02 | Gilead Sciences, Inc. | Kras modulating compounds |
| WO2025024811A1 (en) | 2023-07-26 | 2025-01-30 | Gilead Sciences, Inc. | Parp7 inhibitors |
| WO2025024663A1 (en) | 2023-07-26 | 2025-01-30 | Gilead Sciences, Inc. | Parp7 inhibitors |
| US20250101042A1 (en) | 2023-09-08 | 2025-03-27 | Gilead Sciences, Inc. | Kras g12d modulating compounds |
| US20250109147A1 (en) | 2023-09-08 | 2025-04-03 | Gilead Sciences, Inc. | Kras g12d modulating compounds |
| WO2025096589A1 (en) | 2023-11-03 | 2025-05-08 | Gilead Sciences, Inc. | Prmt5 inhibitors and uses thereof |
| WO2025137640A1 (en) | 2023-12-22 | 2025-06-26 | Gilead Sciences, Inc. | Azaspiro wrn inhibitors |
| US20250376484A1 (en) | 2024-05-21 | 2025-12-11 | Gilead Sciences, Inc. | Prmt5 inhibitors and uses thereof |
Family Cites Families (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5529976A (en) * | 1990-01-10 | 1996-06-25 | Hoechst Aktiengesellschaft | Pyridyl sulphonyl ureas as herbicides and plant growth regulators |
| DE19629144A1 (en) | 1996-07-19 | 1998-01-22 | Bayer Ag | Substituted triazoloazine sulfonamides |
| US5886191A (en) | 1997-08-18 | 1999-03-23 | Dupont Pharmaceuticals Company | Amidinoindoles, amidinoazoles, and analogs thereof |
| DE19941559A1 (en) | 1999-09-01 | 2001-03-15 | Aventis Pharma Gmbh | Use of bissulfonamides for the preparation of medicaments for the prophylaxis or treatment of hyperlipidemia |
| JP2003531194A (en) | 2000-04-24 | 2003-10-21 | メルク フロスト カナダ アンド カンパニー | Therapeutic methods using phenyl and biaryl derivatives as prostaglandin E inhibitors and compounds useful in the methods |
| JP3847711B2 (en) | 2000-09-18 | 2006-11-22 | トーアエイヨー株式会社 | N-substituted benzothiophenesulfonamide derivatives |
| US7071220B2 (en) | 2000-09-18 | 2006-07-04 | Toa Eiyo Ltd. | N-substituted benzothiophenesulfonamide derivatives |
| AUPR738301A0 (en) * | 2001-08-30 | 2001-09-20 | Starpharma Limited | Chemotherapeutic agents |
| IL164212A0 (en) | 2002-03-28 | 2005-12-18 | Applied Research Systems | Thiazolidine carboxamide derivatives, their preparation and pharmaceutical compositions containing them |
| WO2004026823A1 (en) | 2002-09-20 | 2004-04-01 | Pfizer Products Inc. | Amide and sulfonamide ligands for the estrogen receptor |
| CA2583710A1 (en) | 2004-10-12 | 2006-04-27 | Decode Genetics, Inc. | Aryl sulfonamide peri-substituted bicyclics for occlusive artery disease |
| US20090234014A1 (en) * | 2005-10-13 | 2009-09-17 | Benjamin Pelcman | Naphthalene-Disulfonamides Useful for the Treatment of Inflammation |
| WO2008129276A1 (en) * | 2007-04-19 | 2008-10-30 | Boehringer Ingelheim International Gmbh | Disulfonamides useful in the treatment of inflammation |
| WO2008129288A2 (en) * | 2007-04-19 | 2008-10-30 | Boehringer Ingelheim International Gmbh | Disulfonamides useful in the treatment of inflammation |
| TW200930368A (en) | 2007-11-15 | 2009-07-16 | Astrazeneca Ab | Bis-(sulfonylamino) derivatives in therapy |
| TW200930369A (en) | 2007-11-15 | 2009-07-16 | Astrazeneca Ab | Bis-(sulfonylamino) derivatives in therapy |
| US20100292279A1 (en) | 2009-05-14 | 2010-11-18 | Astrazeneca Ab | Bis-(Sulfonylamino) Derivatives in Therapy |
-
2008
- 2008-12-17 US US12/337,125 patent/US20090163586A1/en not_active Abandoned
- 2008-12-18 BR BRPI0821299-6A patent/BRPI0821299A2/en not_active IP Right Cessation
- 2008-12-18 EA EA201001013A patent/EA017940B1/en not_active IP Right Cessation
- 2008-12-18 EP EP08863493A patent/EP2234993B1/en active Active
- 2008-12-18 KR KR1020107016075A patent/KR20100098711A/en not_active Ceased
- 2008-12-18 PE PE2008002126A patent/PE20091175A1/en not_active Application Discontinuation
- 2008-12-18 AU AU2008341173A patent/AU2008341173B2/en not_active Ceased
- 2008-12-18 ES ES08863493T patent/ES2397502T3/en active Active
- 2008-12-18 UY UY31546A patent/UY31546A1/en unknown
- 2008-12-18 JP JP2010539383A patent/JP5555175B2/en not_active Expired - Fee Related
- 2008-12-18 US US12/747,049 patent/US9145380B2/en not_active Expired - Fee Related
- 2008-12-18 CA CA2709510A patent/CA2709510A1/en not_active Abandoned
- 2008-12-18 TW TW097149496A patent/TW200932210A/en unknown
- 2008-12-18 WO PCT/SE2008/051500 patent/WO2009082347A1/en not_active Ceased
- 2008-12-18 CN CN200880127180.2A patent/CN101945859B/en not_active Expired - Fee Related
- 2008-12-19 CL CL2008003848A patent/CL2008003848A1/en unknown
- 2008-12-19 AR ARP080105595A patent/AR069919A1/en unknown
-
2010
- 2010-05-13 CO CO10057535A patent/CO6270307A2/en not_active Application Discontinuation
- 2010-05-13 IL IL205758A patent/IL205758A0/en unknown
- 2010-06-14 ZA ZA2010/04226A patent/ZA201004226B/en unknown
- 2010-06-18 EC EC2010010272A patent/ECSP10010272A/en unknown
- 2010-06-18 CR CR11517A patent/CR11517A/en not_active Application Discontinuation
- 2010-06-18 DO DO2010000191A patent/DOP2010000191A/en unknown
- 2010-06-18 NI NI201000109A patent/NI201000109A/en unknown
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20110021540A1 (en) * | 2007-11-15 | 2011-01-27 | Astrazeneca Ab | Bis-(Sulfonylamino) Derivatives in Therapy 066 |
| US20100292279A1 (en) * | 2009-05-14 | 2010-11-18 | Astrazeneca Ab | Bis-(Sulfonylamino) Derivatives in Therapy |
| WO2014062044A1 (en) | 2012-10-15 | 2014-04-24 | Vilnius University | Fluorinated benzenesulfonamides as inhibitors of carbonic anhydrase |
| US9725467B2 (en) | 2012-10-15 | 2017-08-08 | Vilnius University | Fluorinated benzenesulfonamides as inhibitors of carbonic anhydrase |
| WO2016085392A1 (en) * | 2014-11-27 | 2016-06-02 | Acturum Life Science AB | Bis(sulfonamide) derivatives and their use as mpges inhibitors |
| WO2016085391A1 (en) * | 2014-11-27 | 2016-06-02 | Acturum Life Science AB | Bis(sulfonamide) derivatives and their use as mpges inhibitors |
| US10081614B2 (en) | 2014-11-27 | 2018-09-25 | Acturum Real Estate Ab | Bis(sulfonamide) derivatives and their use as mPGES inhibitors |
| US10227296B2 (en) | 2014-11-27 | 2019-03-12 | Arcturum Real Estate AB | Bis(sulfonamide) derivatives and their use as mPGES inhibitors |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2011507837A (en) | 2011-03-10 |
| WO2009082347A1 (en) | 2009-07-02 |
| JP5555175B2 (en) | 2014-07-23 |
| BRPI0821299A2 (en) | 2015-06-16 |
| ECSP10010272A (en) | 2010-08-31 |
| CO6270307A2 (en) | 2011-04-20 |
| CA2709510A1 (en) | 2009-07-02 |
| TW200932210A (en) | 2009-08-01 |
| CN101945859A (en) | 2011-01-12 |
| CR11517A (en) | 2010-08-05 |
| EP2234993B1 (en) | 2012-11-07 |
| NI201000109A (en) | 2012-05-14 |
| CL2008003848A1 (en) | 2010-02-05 |
| HK1149259A1 (en) | 2011-09-30 |
| AU2008341173B2 (en) | 2011-12-15 |
| ZA201004226B (en) | 2011-04-28 |
| ES2397502T3 (en) | 2013-03-07 |
| PE20091175A1 (en) | 2009-09-03 |
| EA017940B1 (en) | 2013-04-30 |
| EP2234993A1 (en) | 2010-10-06 |
| US9145380B2 (en) | 2015-09-29 |
| AU2008341173A1 (en) | 2009-07-02 |
| CN101945859B (en) | 2014-04-09 |
| KR20100098711A (en) | 2010-09-08 |
| AR069919A1 (en) | 2010-03-03 |
| US20100331321A1 (en) | 2010-12-30 |
| DOP2010000191A (en) | 2010-08-31 |
| UY31546A1 (en) | 2009-08-03 |
| EA201001013A1 (en) | 2010-12-30 |
| EP2234993A4 (en) | 2012-02-08 |
| IL205758A0 (en) | 2010-11-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US9145380B2 (en) | Bis-(sulfonylamino) derivatives for use in therapy | |
| US20110021540A1 (en) | Bis-(Sulfonylamino) Derivatives in Therapy 066 | |
| US20090131468A1 (en) | Bis-(Sulfonylamino) Derivatives in Therapy 065 | |
| CA2489276C (en) | Tricyclic steroid hormone nuclear receptor modulators | |
| US20100137298A1 (en) | Biaryl Sulfonamides and Methods for Using Same | |
| RU2372330C2 (en) | SUBSTITUTED PHENOXY-ACETIC ACIDS, WITH MODULATING EFFECT ON CRTh2 RECEPTORS | |
| US20140057953A1 (en) | Biaryl derivatives as selective 17beta-hydroxysteroid dehydrogenase type 2 inhibitors | |
| US20100105657A1 (en) | Compounds, Process for their Preparation, Intermediates, Pharmaceutical Compositions and their use in the Treatment of 5-HT6 Mediated Disorders such as Alzheimer's Disease, Cognitive Disorders, Cognitive Impairment Associated with Schizophrenia, Obesity and Parkinson's Disease | |
| TW201100073A (en) | Bis-(sulfonylamino) derivatives in therapy 735 | |
| AU2010212970A1 (en) | Phenoxy acetic acid derivatives | |
| US20200030303A1 (en) | Compositions and methods for inhibiting protein kinases | |
| CZ259399A3 (en) | Derivative of 5-aroylnaphthalene, process of its preparation and use | |
| HK1149259B (en) | Bis-(sulfonylamino) derivatives for use in therapy | |
| PT2177512E (en) | Inhibitor of binding of s1p1 | |
| SK83399A3 (en) | Nitromethylthiobenzene derivatives as inhibitors of aldose reductase |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: ASTRAZENECA AB,SWEDEN Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:BYLUND, JOHAN;EK, MARIA E.;GRAVENFORS, YLVA;AND OTHERS;SIGNING DATES FROM 20081203 TO 20081215;REEL/FRAME:022376/0934 |
|
| STCB | Information on status: application discontinuation |
Free format text: ABANDONED -- FAILURE TO RESPOND TO AN OFFICE ACTION |