US20080044487A1 - Anti-inflammatory properties of marine lipid compositions - Google Patents
Anti-inflammatory properties of marine lipid compositions Download PDFInfo
- Publication number
- US20080044487A1 US20080044487A1 US11/800,229 US80022907A US2008044487A1 US 20080044487 A1 US20080044487 A1 US 20080044487A1 US 80022907 A US80022907 A US 80022907A US 2008044487 A1 US2008044487 A1 US 2008044487A1
- Authority
- US
- United States
- Prior art keywords
- composition
- omega
- fatty acid
- predicted
- phospholipids
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Abandoned
Links
- 239000000203 mixture Substances 0.000 title claims abstract description 192
- 150000002632 lipids Chemical class 0.000 title claims abstract description 70
- 230000003110 anti-inflammatory effect Effects 0.000 title description 4
- 150000003904 phospholipids Chemical class 0.000 claims abstract description 156
- 235000020660 omega-3 fatty acid Nutrition 0.000 claims abstract description 72
- 150000003626 triacylglycerols Chemical class 0.000 claims abstract description 34
- 235000014113 dietary fatty acids Nutrition 0.000 claims description 51
- 229930195729 fatty acid Natural products 0.000 claims description 51
- 239000000194 fatty acid Substances 0.000 claims description 51
- 150000004665 fatty acids Chemical class 0.000 claims description 49
- 229940012843 omega-3 fatty acid Drugs 0.000 claims description 49
- CYQFCXCEBYINGO-IAGOWNOFSA-N delta1-THC Chemical compound C1=C(C)CC[C@H]2C(C)(C)OC3=CC(CCCCC)=CC(O)=C3[C@@H]21 CYQFCXCEBYINGO-IAGOWNOFSA-N 0.000 claims description 35
- 238000000034 method Methods 0.000 claims description 32
- 239000006014 omega-3 oil Substances 0.000 claims description 26
- 235000015872 dietary supplement Nutrition 0.000 claims description 17
- HZAXFHJVJLSVMW-UHFFFAOYSA-N 2-Aminoethan-1-ol Chemical compound NCCO HZAXFHJVJLSVMW-UHFFFAOYSA-N 0.000 claims description 16
- 241001465754 Metazoa Species 0.000 claims description 16
- 235000013305 food Nutrition 0.000 claims description 13
- MTCFGRXMJLQNBG-REOHCLBHSA-N (2S)-2-Amino-3-hydroxypropansäure Chemical compound OC[C@H](N)C(O)=O MTCFGRXMJLQNBG-REOHCLBHSA-N 0.000 claims description 10
- SQUHHTBVTRBESD-UHFFFAOYSA-N Hexa-Ac-myo-Inositol Natural products CC(=O)OC1C(OC(C)=O)C(OC(C)=O)C(OC(C)=O)C(OC(C)=O)C1OC(C)=O SQUHHTBVTRBESD-UHFFFAOYSA-N 0.000 claims description 10
- MTCFGRXMJLQNBG-UHFFFAOYSA-N Serine Natural products OCC(N)C(O)=O MTCFGRXMJLQNBG-UHFFFAOYSA-N 0.000 claims description 10
- 230000004054 inflammatory process Effects 0.000 claims description 10
- CDAISMWEOUEBRE-GPIVLXJGSA-N inositol Chemical compound O[C@H]1[C@H](O)[C@@H](O)[C@H](O)[C@H](O)[C@@H]1O CDAISMWEOUEBRE-GPIVLXJGSA-N 0.000 claims description 10
- 229960000367 inositol Drugs 0.000 claims description 10
- CDAISMWEOUEBRE-UHFFFAOYSA-N scyllo-inosotol Natural products OC1C(O)C(O)C(O)C(O)C1O CDAISMWEOUEBRE-UHFFFAOYSA-N 0.000 claims description 10
- 229960001153 serine Drugs 0.000 claims description 10
- UFTFJSFQGQCHQW-UHFFFAOYSA-N triformin Chemical compound O=COCC(OC=O)COC=O UFTFJSFQGQCHQW-UHFFFAOYSA-N 0.000 claims description 10
- 206010061218 Inflammation Diseases 0.000 claims description 9
- OEYIOHPDSNJKLS-UHFFFAOYSA-N choline Chemical compound C[N+](C)(C)CCO OEYIOHPDSNJKLS-UHFFFAOYSA-N 0.000 claims description 9
- 229960001231 choline Drugs 0.000 claims description 9
- 229940031098 ethanolamine Drugs 0.000 claims description 8
- 230000008569 process Effects 0.000 claims description 7
- 235000013376 functional food Nutrition 0.000 claims description 5
- 239000000090 biomarker Substances 0.000 claims description 4
- 125000004494 ethyl ester group Chemical group 0.000 claims description 3
- 150000004702 methyl esters Chemical class 0.000 claims description 3
- 238000004806 packaging method and process Methods 0.000 claims description 2
- 230000002829 reductive effect Effects 0.000 abstract description 10
- 230000000770 proinflammatory effect Effects 0.000 abstract description 7
- 102000004127 Cytokines Human genes 0.000 abstract description 4
- 108090000695 Cytokines Proteins 0.000 abstract description 4
- 238000010348 incorporation Methods 0.000 abstract description 4
- 108090000623 proteins and genes Proteins 0.000 description 91
- MBMBGCFOFBJSGT-KUBAVDMBSA-N docosahexaenoic acid Natural products CC\C=C/C\C=C/C\C=C/C\C=C/C\C=C/C\C=C/CCC(O)=O MBMBGCFOFBJSGT-KUBAVDMBSA-N 0.000 description 85
- 235000018102 proteins Nutrition 0.000 description 64
- 102000004169 proteins and genes Human genes 0.000 description 64
- YZXBAPSDXZZRGB-DOFZRALJSA-N arachidonic acid Chemical compound CCCCC\C=C/C\C=C/C\C=C/C\C=C/CCCC(O)=O YZXBAPSDXZZRGB-DOFZRALJSA-N 0.000 description 51
- 239000003921 oil Substances 0.000 description 41
- WTJKGGKOPKCXLL-RRHRGVEJSA-N phosphatidylcholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCCC=CCCCCCCCC WTJKGGKOPKCXLL-RRHRGVEJSA-N 0.000 description 32
- 235000021342 arachidonic acid Nutrition 0.000 description 26
- 229940114079 arachidonic acid Drugs 0.000 description 25
- 235000019198 oils Nutrition 0.000 description 24
- 241000700159 Rattus Species 0.000 description 22
- 230000008859 change Effects 0.000 description 22
- 102000004190 Enzymes Human genes 0.000 description 21
- 108090000790 Enzymes Proteins 0.000 description 21
- 210000000577 adipose tissue Anatomy 0.000 description 18
- 230000001105 regulatory effect Effects 0.000 description 18
- 210000002381 plasma Anatomy 0.000 description 17
- 210000004185 liver Anatomy 0.000 description 16
- 210000001519 tissue Anatomy 0.000 description 16
- 102000004882 Lipase Human genes 0.000 description 14
- 108090001060 Lipase Proteins 0.000 description 14
- 239000004367 Lipase Substances 0.000 description 14
- 238000006243 chemical reaction Methods 0.000 description 14
- 239000002299 complementary DNA Substances 0.000 description 14
- 235000019421 lipase Nutrition 0.000 description 14
- 239000000047 product Substances 0.000 description 14
- 241000255581 Drosophila <fruit fly, genus> Species 0.000 description 13
- 235000021323 fish oil Nutrition 0.000 description 13
- 239000000787 lecithin Substances 0.000 description 13
- 229940067606 lecithin Drugs 0.000 description 13
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 description 12
- 235000010445 lecithin Nutrition 0.000 description 12
- -1 nitrogen containing compound Chemical class 0.000 description 12
- 238000010521 absorption reaction Methods 0.000 description 11
- 210000004556 brain Anatomy 0.000 description 11
- 101150067766 mpl2 gene Proteins 0.000 description 11
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 10
- 210000002216 heart Anatomy 0.000 description 10
- YUFFSWGQGVEMMI-UHFFFAOYSA-N (7Z,10Z,13Z,16Z,19Z)-7,10,13,16,19-docosapentaenoic acid Natural products CCC=CCC=CCC=CCC=CCC=CCCCCCC(O)=O YUFFSWGQGVEMMI-UHFFFAOYSA-N 0.000 description 9
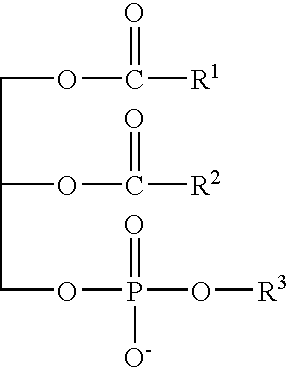
- 0 [1*]C(=O)OCC(COP(=O)([O-])O[3*])OC([2*])=O Chemical compound [1*]C(=O)OCC(COP(=O)([O-])O[3*])OC([2*])=O 0.000 description 9
- 239000002253 acid Substances 0.000 description 9
- 230000027455 binding Effects 0.000 description 9
- KSMVZQYAVGTKIV-UHFFFAOYSA-N decanal Chemical compound CCCCCCCCCC=O KSMVZQYAVGTKIV-UHFFFAOYSA-N 0.000 description 9
- 235000005911 diet Nutrition 0.000 description 9
- 230000037213 diet Effects 0.000 description 9
- 210000001550 testis Anatomy 0.000 description 9
- 238000011282 treatment Methods 0.000 description 9
- 210000004027 cell Anatomy 0.000 description 8
- 101150011281 mpl1 gene Proteins 0.000 description 8
- 238000005809 transesterification reaction Methods 0.000 description 8
- 235000010469 Glycine max Nutrition 0.000 description 7
- 108091000080 Phosphotransferase Proteins 0.000 description 7
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 7
- 230000007423 decrease Effects 0.000 description 7
- 238000000105 evaporative light scattering detection Methods 0.000 description 7
- 230000028709 inflammatory response Effects 0.000 description 7
- 229910052500 inorganic mineral Inorganic materials 0.000 description 7
- 235000010755 mineral Nutrition 0.000 description 7
- 239000011707 mineral Substances 0.000 description 7
- 102000020233 phosphotransferase Human genes 0.000 description 7
- 239000000126 substance Substances 0.000 description 7
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 7
- YBJHBAHKTGYVGT-ZKWXMUAHSA-N (+)-Biotin Chemical compound N1C(=O)N[C@@H]2[C@H](CCCCC(=O)O)SC[C@@H]21 YBJHBAHKTGYVGT-ZKWXMUAHSA-N 0.000 description 6
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 6
- 102100033246 Lysine-specific demethylase 5A Human genes 0.000 description 6
- 239000002775 capsule Substances 0.000 description 6
- 150000001875 compounds Chemical class 0.000 description 6
- 201000010099 disease Diseases 0.000 description 6
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 6
- 238000002474 experimental method Methods 0.000 description 6
- 235000021588 free fatty acids Nutrition 0.000 description 6
- WTEVQBCEXWBHNA-JXMROGBWSA-N geranial Chemical compound CC(C)=CCC\C(C)=C\C=O WTEVQBCEXWBHNA-JXMROGBWSA-N 0.000 description 6
- 230000007062 hydrolysis Effects 0.000 description 6
- 238000006460 hydrolysis reaction Methods 0.000 description 6
- 210000001616 monocyte Anatomy 0.000 description 6
- NLKNQRATVPKPDG-UHFFFAOYSA-M potassium iodide Chemical compound [K+].[I-] NLKNQRATVPKPDG-UHFFFAOYSA-M 0.000 description 6
- 239000008347 soybean phospholipid Substances 0.000 description 6
- 208000024891 symptom Diseases 0.000 description 6
- 229940088594 vitamin Drugs 0.000 description 6
- 229930003231 vitamin Natural products 0.000 description 6
- 235000013343 vitamin Nutrition 0.000 description 6
- 239000011782 vitamin Substances 0.000 description 6
- PZNPLUBHRSSFHT-RRHRGVEJSA-N 1-hexadecanoyl-2-octadecanoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCCCC(=O)O[C@@H](COP([O-])(=O)OCC[N+](C)(C)C)COC(=O)CCCCCCCCCCCCCCC PZNPLUBHRSSFHT-RRHRGVEJSA-N 0.000 description 5
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 5
- 101000892360 Homo sapiens Protein AF-17 Proteins 0.000 description 5
- 102100040638 Protein AF-17 Human genes 0.000 description 5
- 102000001708 Protein Isoforms Human genes 0.000 description 5
- 108010029485 Protein Isoforms Proteins 0.000 description 5
- DTOSIQBPPRVQHS-PDBXOOCHSA-N alpha-linolenic acid Chemical compound CC\C=C/C\C=C/C\C=C/CCCCCCCC(O)=O DTOSIQBPPRVQHS-PDBXOOCHSA-N 0.000 description 5
- 208000037976 chronic inflammation Diseases 0.000 description 5
- 230000006020 chronic inflammation Effects 0.000 description 5
- 239000003623 enhancer Substances 0.000 description 5
- 230000035558 fertility Effects 0.000 description 5
- 230000006870 function Effects 0.000 description 5
- 239000004615 ingredient Substances 0.000 description 5
- 208000032839 leukemia Diseases 0.000 description 5
- CDOSHBSSFJOMGT-UHFFFAOYSA-N linalool Chemical compound CC(C)=CCCC(C)(O)C=C CDOSHBSSFJOMGT-UHFFFAOYSA-N 0.000 description 5
- 239000003960 organic solvent Substances 0.000 description 5
- 235000019629 palatability Nutrition 0.000 description 5
- 239000002245 particle Substances 0.000 description 5
- SATCULPHIDQDRE-UHFFFAOYSA-N piperonal Chemical compound O=CC1=CC=C2OCOC2=C1 SATCULPHIDQDRE-UHFFFAOYSA-N 0.000 description 5
- 229920001184 polypeptide Polymers 0.000 description 5
- 239000000843 powder Substances 0.000 description 5
- 102000004196 processed proteins & peptides Human genes 0.000 description 5
- 108090000765 processed proteins & peptides Proteins 0.000 description 5
- 239000011541 reaction mixture Substances 0.000 description 5
- 230000035484 reaction time Effects 0.000 description 5
- 102000005962 receptors Human genes 0.000 description 5
- 108020003175 receptors Proteins 0.000 description 5
- 230000009467 reduction Effects 0.000 description 5
- 239000012855 volatile organic compound Substances 0.000 description 5
- ADHNUPOJJCKWRT-JLXBFWJWSA-N (2e,4e)-octadeca-2,4-dienoic acid Chemical compound CCCCCCCCCCCCC\C=C\C=C\C(O)=O ADHNUPOJJCKWRT-JLXBFWJWSA-N 0.000 description 4
- IKHGUXGNUITLKF-UHFFFAOYSA-N Acetaldehyde Chemical compound CC=O IKHGUXGNUITLKF-UHFFFAOYSA-N 0.000 description 4
- ROWKJAVDOGWPAT-UHFFFAOYSA-N Acetoin Chemical compound CC(O)C(C)=O ROWKJAVDOGWPAT-UHFFFAOYSA-N 0.000 description 4
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 4
- 240000004160 Capsicum annuum Species 0.000 description 4
- 102000014914 Carrier Proteins Human genes 0.000 description 4
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 4
- 241000195493 Cryptophyta Species 0.000 description 4
- 102100029905 DNA polymerase epsilon subunit 3 Human genes 0.000 description 4
- 244000068988 Glycine max Species 0.000 description 4
- 241000282412 Homo Species 0.000 description 4
- 101710105716 Lysine-specific demethylase 5A Proteins 0.000 description 4
- 102100037611 Lysophospholipase Human genes 0.000 description 4
- 241000124008 Mammalia Species 0.000 description 4
- 208000001145 Metabolic Syndrome Diseases 0.000 description 4
- 235000009421 Myristica fragrans Nutrition 0.000 description 4
- 208000008589 Obesity Diseases 0.000 description 4
- LQKRYVGRPXFFAV-UHFFFAOYSA-N Phenylmethylglycidic ester Chemical compound CCOC(=O)C1OC1(C)C1=CC=CC=C1 LQKRYVGRPXFFAV-UHFFFAOYSA-N 0.000 description 4
- 108010058864 Phospholipases A2 Proteins 0.000 description 4
- AUNGANRZJHBGPY-SCRDCRAPSA-N Riboflavin Chemical compound OC[C@@H](O)[C@@H](O)[C@@H](O)CN1C=2C=C(C)C(C)=CC=2N=C2C1=NC(=O)NC2=O AUNGANRZJHBGPY-SCRDCRAPSA-N 0.000 description 4
- 244000178231 Rosmarinus officinalis Species 0.000 description 4
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 4
- 108060008682 Tumor Necrosis Factor Proteins 0.000 description 4
- 102000000852 Tumor Necrosis Factor-alpha Human genes 0.000 description 4
- 201000000690 abdominal obesity-metabolic syndrome Diseases 0.000 description 4
- 235000010323 ascorbic acid Nutrition 0.000 description 4
- 230000006399 behavior Effects 0.000 description 4
- HUMNYLRZRPPJDN-UHFFFAOYSA-N benzaldehyde Chemical compound O=CC1=CC=CC=C1 HUMNYLRZRPPJDN-UHFFFAOYSA-N 0.000 description 4
- 108091008324 binding proteins Proteins 0.000 description 4
- RMRCNWBMXRMIRW-BYFNXCQMSA-M cyanocobalamin Chemical compound N#C[Co+]N([C@]1([H])[C@H](CC(N)=O)[C@]\2(CCC(=O)NC[C@H](C)OP(O)(=O)OC3[C@H]([C@H](O[C@@H]3CO)N3C4=CC(C)=C(C)C=C4N=C3)O)C)C/2=C(C)\C([C@H](C/2(C)C)CCC(N)=O)=N\C\2=C\C([C@H]([C@@]/2(CC(N)=O)C)CCC(N)=O)=N\C\2=C(C)/C2=N[C@]1(C)[C@@](C)(CC(N)=O)[C@@H]2CCC(N)=O RMRCNWBMXRMIRW-BYFNXCQMSA-M 0.000 description 4
- 230000001419 dependent effect Effects 0.000 description 4
- 229940079593 drug Drugs 0.000 description 4
- 239000003814 drug Substances 0.000 description 4
- 239000008344 egg yolk phospholipid Substances 0.000 description 4
- 150000002066 eicosanoids Chemical class 0.000 description 4
- ZQPPMHVWECSIRJ-MDZDMXLPSA-N elaidic acid Chemical compound CCCCCCCC\C=C\CCCCCCCC(O)=O ZQPPMHVWECSIRJ-MDZDMXLPSA-N 0.000 description 4
- 239000012467 final product Substances 0.000 description 4
- OVBPIULPVIDEAO-LBPRGKRZSA-N folic acid Chemical compound C=1N=C2NC(N)=NC(=O)C2=NC=1CNC1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 OVBPIULPVIDEAO-LBPRGKRZSA-N 0.000 description 4
- 239000000499 gel Substances 0.000 description 4
- 230000014509 gene expression Effects 0.000 description 4
- 230000002401 inhibitory effect Effects 0.000 description 4
- 239000007788 liquid Substances 0.000 description 4
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 4
- 239000011159 matrix material Substances 0.000 description 4
- 108020004999 messenger RNA Proteins 0.000 description 4
- VAMXMNNIEUEQDV-UHFFFAOYSA-N methyl anthranilate Chemical compound COC(=O)C1=CC=CC=C1N VAMXMNNIEUEQDV-UHFFFAOYSA-N 0.000 description 4
- 235000020824 obesity Nutrition 0.000 description 4
- 150000003180 prostaglandins Chemical class 0.000 description 4
- 239000013589 supplement Substances 0.000 description 4
- 238000012360 testing method Methods 0.000 description 4
- MZOFCQQQCNRIBI-VMXHOPILSA-N (3s)-4-[[(2s)-1-[[(2s)-1-[[(1s)-1-carboxy-2-hydroxyethyl]amino]-4-methyl-1-oxopentan-2-yl]amino]-5-(diaminomethylideneamino)-1-oxopentan-2-yl]amino]-3-[[2-[[(2s)-2,6-diaminohexanoyl]amino]acetyl]amino]-4-oxobutanoic acid Chemical compound OC[C@@H](C(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CC(O)=O)NC(=O)CNC(=O)[C@@H](N)CCCCN MZOFCQQQCNRIBI-VMXHOPILSA-N 0.000 description 3
- ULDHMXUKGWMISQ-VIFPVBQESA-N (S)-(+)-Carvone Natural products CC(=C)[C@H]1CC=C(C)C(=O)C1 ULDHMXUKGWMISQ-VIFPVBQESA-N 0.000 description 3
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 3
- TZCPCKNHXULUIY-RGULYWFUSA-N 1,2-distearoyl-sn-glycero-3-phosphoserine Chemical compound CCCCCCCCCCCCCCCCCC(=O)OC[C@H](COP(O)(=O)OC[C@H](N)C(O)=O)OC(=O)CCCCCCCCCCCCCCCCC TZCPCKNHXULUIY-RGULYWFUSA-N 0.000 description 3
- JLPULHDHAOZNQI-ZTIMHPMXSA-N 1-hexadecanoyl-2-(9Z,12Z-octadecadienoyl)-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCC\C=C/C\C=C/CCCCC JLPULHDHAOZNQI-ZTIMHPMXSA-N 0.000 description 3
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 3
- 102100034614 Ankyrin repeat domain-containing protein 11 Human genes 0.000 description 3
- 101710188804 Ankyrin repeat domain-containing protein 11 Proteins 0.000 description 3
- 208000006096 Attention Deficit Disorder with Hyperactivity Diseases 0.000 description 3
- 101710186200 CCAAT/enhancer-binding protein Proteins 0.000 description 3
- 235000008534 Capsicum annuum var annuum Nutrition 0.000 description 3
- 240000008574 Capsicum frutescens Species 0.000 description 3
- 235000002568 Capsicum frutescens Nutrition 0.000 description 3
- WTEVQBCEXWBHNA-UHFFFAOYSA-N Citral Natural products CC(C)=CCCC(C)=CC=O WTEVQBCEXWBHNA-UHFFFAOYSA-N 0.000 description 3
- 235000015655 Crocus sativus Nutrition 0.000 description 3
- 244000124209 Crocus sativus Species 0.000 description 3
- 101100238358 Dictyostelium discoideum mpl3 gene Proteins 0.000 description 3
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 3
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 3
- ZWZWYGMENQVNFU-UHFFFAOYSA-N Glycerophosphorylserin Natural products OC(=O)C(N)COP(O)(=O)OCC(O)CO ZWZWYGMENQVNFU-UHFFFAOYSA-N 0.000 description 3
- 102100025505 Intersectin-2 Human genes 0.000 description 3
- 108010013563 Lipoprotein Lipase Proteins 0.000 description 3
- 102100022119 Lipoprotein lipase Human genes 0.000 description 3
- 239000005913 Maltodextrin Substances 0.000 description 3
- 229920002774 Maltodextrin Polymers 0.000 description 3
- 244000270834 Myristica fragrans Species 0.000 description 3
- ZQPPMHVWECSIRJ-UHFFFAOYSA-N Oleic acid Natural products CCCCCCCCC=CCCCCCCCC(O)=O ZQPPMHVWECSIRJ-UHFFFAOYSA-N 0.000 description 3
- 108700020796 Oncogene Proteins 0.000 description 3
- 108090000315 Protein Kinase C Proteins 0.000 description 3
- 102000003923 Protein Kinase C Human genes 0.000 description 3
- 102000002727 Protein Tyrosine Phosphatase Human genes 0.000 description 3
- 206010037660 Pyrexia Diseases 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 206010049418 Sudden Cardiac Death Diseases 0.000 description 3
- 244000269722 Thea sinensis Species 0.000 description 3
- 102100039411 WW domain-binding protein 4 Human genes 0.000 description 3
- 101710160510 WW domain-binding protein 4 Proteins 0.000 description 3
- 235000006886 Zingiber officinale Nutrition 0.000 description 3
- 244000273928 Zingiber officinale Species 0.000 description 3
- 238000009825 accumulation Methods 0.000 description 3
- 201000006288 alpha thalassemia Diseases 0.000 description 3
- 235000020661 alpha-linolenic acid Nutrition 0.000 description 3
- 239000011668 ascorbic acid Substances 0.000 description 3
- 229960005070 ascorbic acid Drugs 0.000 description 3
- 208000029560 autism spectrum disease Diseases 0.000 description 3
- 229960002685 biotin Drugs 0.000 description 3
- 235000020958 biotin Nutrition 0.000 description 3
- 239000011616 biotin Substances 0.000 description 3
- 210000005013 brain tissue Anatomy 0.000 description 3
- 239000001506 calcium phosphate Substances 0.000 description 3
- 239000001511 capsicum annuum Substances 0.000 description 3
- 235000011089 carbon dioxide Nutrition 0.000 description 3
- 239000000969 carrier Substances 0.000 description 3
- ULDHMXUKGWMISQ-UHFFFAOYSA-N carvone Natural products CC(=C)C1CC=C(C)C(=O)C1 ULDHMXUKGWMISQ-UHFFFAOYSA-N 0.000 description 3
- 206010012601 diabetes mellitus Diseases 0.000 description 3
- POULHZVOKOAJMA-UHFFFAOYSA-N dodecanoic acid Chemical compound CCCCCCCCCCCC(O)=O POULHZVOKOAJMA-UHFFFAOYSA-N 0.000 description 3
- 238000001914 filtration Methods 0.000 description 3
- 235000019152 folic acid Nutrition 0.000 description 3
- 235000008397 ginger Nutrition 0.000 description 3
- 239000011521 glass Substances 0.000 description 3
- QXJSBBXBKPUZAA-UHFFFAOYSA-N isooleic acid Natural products CCCCCCCC=CCCCCCCCCC(O)=O QXJSBBXBKPUZAA-UHFFFAOYSA-N 0.000 description 3
- 235000020978 long-chain polyunsaturated fatty acids Nutrition 0.000 description 3
- 238000012423 maintenance Methods 0.000 description 3
- 229940035034 maltodextrin Drugs 0.000 description 3
- 238000004519 manufacturing process Methods 0.000 description 3
- 230000003340 mental effect Effects 0.000 description 3
- 230000002438 mitochondrial effect Effects 0.000 description 3
- 229910052757 nitrogen Inorganic materials 0.000 description 3
- 229940094443 oxytocics prostaglandins Drugs 0.000 description 3
- SECPZKHBENQXJG-FPLPWBNLSA-N palmitoleic acid Chemical compound CCCCCC\C=C/CCCCCCCC(O)=O SECPZKHBENQXJG-FPLPWBNLSA-N 0.000 description 3
- 239000000546 pharmaceutical excipient Substances 0.000 description 3
- 239000006187 pill Substances 0.000 description 3
- 235000020777 polyunsaturated fatty acids Nutrition 0.000 description 3
- 239000002243 precursor Substances 0.000 description 3
- 108020000494 protein-tyrosine phosphatase Proteins 0.000 description 3
- 239000000377 silicon dioxide Substances 0.000 description 3
- 239000000758 substrate Substances 0.000 description 3
- 208000011580 syndromic disease Diseases 0.000 description 3
- GVZXZHWIIXHZOB-UHFFFAOYSA-N tariric acid Chemical compound CCCCCCCCCCCC#CCCCCC(O)=O GVZXZHWIIXHZOB-UHFFFAOYSA-N 0.000 description 3
- 230000002381 testicular Effects 0.000 description 3
- 238000013518 transcription Methods 0.000 description 3
- 230000035897 transcription Effects 0.000 description 3
- 230000014621 translational initiation Effects 0.000 description 3
- ULDHMXUKGWMISQ-SECBINFHSA-N (-)-carvone Chemical compound CC(=C)[C@@H]1CC=C(C)C(=O)C1 ULDHMXUKGWMISQ-SECBINFHSA-N 0.000 description 2
- SBHCLVQMTBWHCD-METXMMQOSA-N (2e,4e,6e,8e,10e)-icosa-2,4,6,8,10-pentaenoic acid Chemical compound CCCCCCCCC\C=C\C=C\C=C\C=C\C=C\C(O)=O SBHCLVQMTBWHCD-METXMMQOSA-N 0.000 description 2
- LOGFVTREOLYCPF-KXNHARMFSA-N (2s,3r)-2-[[(2r)-1-[(2s)-2,6-diaminohexanoyl]pyrrolidine-2-carbonyl]amino]-3-hydroxybutanoic acid Chemical compound C[C@@H](O)[C@@H](C(O)=O)NC(=O)[C@H]1CCCN1C(=O)[C@@H](N)CCCCN LOGFVTREOLYCPF-KXNHARMFSA-N 0.000 description 2
- YWWVWXASSLXJHU-AATRIKPKSA-N (9E)-tetradecenoic acid Chemical compound CCCC\C=C\CCCCCCCC(O)=O YWWVWXASSLXJHU-AATRIKPKSA-N 0.000 description 2
- GHOKWGTUZJEAQD-ZETCQYMHSA-N (D)-(+)-Pantothenic acid Chemical compound OCC(C)(C)[C@@H](O)C(=O)NCCC(O)=O GHOKWGTUZJEAQD-ZETCQYMHSA-N 0.000 description 2
- WRIDQFICGBMAFQ-UHFFFAOYSA-N (E)-8-Octadecenoic acid Natural products CCCCCCCCCC=CCCCCCCC(O)=O WRIDQFICGBMAFQ-UHFFFAOYSA-N 0.000 description 2
- KJPRLNWUNMBNBZ-QPJJXVBHSA-N (E)-cinnamaldehyde Chemical compound O=C\C=C\C1=CC=CC=C1 KJPRLNWUNMBNBZ-QPJJXVBHSA-N 0.000 description 2
- GVJHHUAWPYXKBD-UHFFFAOYSA-N (±)-α-Tocopherol Chemical compound OC1=C(C)C(C)=C2OC(CCCC(C)CCCC(C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-UHFFFAOYSA-N 0.000 description 2
- FPIPGXGPPPQFEQ-UHFFFAOYSA-N 13-cis retinol Natural products OCC=C(C)C=CC=C(C)C=CC1=C(C)CCCC1(C)C FPIPGXGPPPQFEQ-UHFFFAOYSA-N 0.000 description 2
- WVXRAFOPTSTNLL-NKWVEPMBSA-N 2',3'-dideoxyadenosine Chemical compound C1=NC=2C(N)=NC=NC=2N1[C@H]1CC[C@@H](CO)O1 WVXRAFOPTSTNLL-NKWVEPMBSA-N 0.000 description 2
- LQJBNNIYVWPHFW-UHFFFAOYSA-N 20:1omega9c fatty acid Natural products CCCCCCCCCCC=CCCCCCCCC(O)=O LQJBNNIYVWPHFW-UHFFFAOYSA-N 0.000 description 2
- UNSRRHDPHVZAHH-UHFFFAOYSA-N 5,8,11-eicosatrienoic acid Chemical compound CCCCCCCCC=CCC=CCC=CCCCC(O)=O UNSRRHDPHVZAHH-UHFFFAOYSA-N 0.000 description 2
- 108010042833 7,8-diaminopelargonic acid aminotransferase Proteins 0.000 description 2
- PQMIPLRIRFVQJZ-QBYYVRQOSA-N 7-[2-[(2s,4s)-4-[(2r,3r,4r,5s,6s)-3-fluoro-4,5-dihydroxy-6-methyloxan-2-yl]oxy-2,5,12-trihydroxy-7-methoxy-6,11-dioxo-3,4-dihydro-1h-tetracen-2-yl]-2-oxoethoxy]-7-oxoheptanoic acid Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)COC(=O)CCCCCC(O)=O)[C@@H]1O[C@@H](C)[C@@H](O)[C@@H](O)[C@H]1F PQMIPLRIRFVQJZ-QBYYVRQOSA-N 0.000 description 2
- QSBYPNXLFMSGKH-UHFFFAOYSA-N 9-Heptadecensaeure Natural products CCCCCCCC=CCCCCCCCC(O)=O QSBYPNXLFMSGKH-UHFFFAOYSA-N 0.000 description 2
- YWWVWXASSLXJHU-UHFFFAOYSA-N 9E-tetradecenoic acid Natural products CCCCC=CCCCCCCCC(O)=O YWWVWXASSLXJHU-UHFFFAOYSA-N 0.000 description 2
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 2
- 241000251468 Actinopterygii Species 0.000 description 2
- 102000007469 Actins Human genes 0.000 description 2
- 108010085238 Actins Proteins 0.000 description 2
- 206010048998 Acute phase reaction Diseases 0.000 description 2
- 206010001488 Aggression Diseases 0.000 description 2
- 244000016163 Allium sibiricum Species 0.000 description 2
- 235000001270 Allium sibiricum Nutrition 0.000 description 2
- 208000000044 Amnesia Diseases 0.000 description 2
- 241000189429 Angostura Species 0.000 description 2
- 235000007258 Anthriscus cerefolium Nutrition 0.000 description 2
- 240000002022 Anthriscus cerefolium Species 0.000 description 2
- 239000004475 Arginine Substances 0.000 description 2
- 235000011330 Armoracia rusticana Nutrition 0.000 description 2
- 240000003291 Armoracia rusticana Species 0.000 description 2
- 235000003092 Artemisia dracunculus Nutrition 0.000 description 2
- 240000001851 Artemisia dracunculus Species 0.000 description 2
- 208000036864 Attention deficit/hyperactivity disease Diseases 0.000 description 2
- 241000271566 Aves Species 0.000 description 2
- 102100032430 B-cell CLL/lymphoma 7 protein family member C Human genes 0.000 description 2
- DPUOLQHDNGRHBS-UHFFFAOYSA-N Brassidinsaeure Natural products CCCCCCCCC=CCCCCCCCCCCCC(O)=O DPUOLQHDNGRHBS-UHFFFAOYSA-N 0.000 description 2
- 241000273930 Brevoortia tyrannus Species 0.000 description 2
- 102100021975 CREB-binding protein Human genes 0.000 description 2
- 240000001432 Calendula officinalis Species 0.000 description 2
- 235000005747 Carum carvi Nutrition 0.000 description 2
- 240000000467 Carum carvi Species 0.000 description 2
- 102000052052 Casein Kinase II Human genes 0.000 description 2
- 108010010919 Casein Kinase II Proteins 0.000 description 2
- 102100029297 Cholinephosphotransferase 1 Human genes 0.000 description 2
- 101710155338 Cholinephosphotransferase 1 Proteins 0.000 description 2
- 108010077544 Chromatin Proteins 0.000 description 2
- 244000037364 Cinnamomum aromaticum Species 0.000 description 2
- 235000014489 Cinnamomum aromaticum Nutrition 0.000 description 2
- 241000252203 Clupea harengus Species 0.000 description 2
- 235000002787 Coriandrum sativum Nutrition 0.000 description 2
- 244000018436 Coriandrum sativum Species 0.000 description 2
- 235000007129 Cuminum cyminum Nutrition 0.000 description 2
- 244000304337 Cuminum cyminum Species 0.000 description 2
- 244000163122 Curcuma domestica Species 0.000 description 2
- 235000003405 Curcuma zedoaria Nutrition 0.000 description 2
- 240000009138 Curcuma zedoaria Species 0.000 description 2
- AUNGANRZJHBGPY-UHFFFAOYSA-N D-Lyxoflavin Natural products OCC(O)C(O)C(O)CN1C=2C=C(C)C(C)=CC=2N=C2C1=NC(=O)NC2=O AUNGANRZJHBGPY-UHFFFAOYSA-N 0.000 description 2
- ZZZCUOFIHGPKAK-UHFFFAOYSA-N D-erythro-ascorbic acid Natural products OCC1OC(=O)C(O)=C1O ZZZCUOFIHGPKAK-UHFFFAOYSA-N 0.000 description 2
- 108010014303 DNA-directed DNA polymerase Proteins 0.000 description 2
- 102000016928 DNA-directed DNA polymerase Human genes 0.000 description 2
- 102100021820 E3 ubiquitin-protein ligase RNF4 Human genes 0.000 description 2
- 101710202028 E3 ubiquitin-protein ligase RNF4 Proteins 0.000 description 2
- 102100028591 E3 ubiquitin-protein ligase ZNRF1 Human genes 0.000 description 2
- 241000258955 Echinodermata Species 0.000 description 2
- 240000002943 Elettaria cardamomum Species 0.000 description 2
- 241000196324 Embryophyta Species 0.000 description 2
- 241000195955 Equisetum hyemale Species 0.000 description 2
- URXZXNYJPAJJOQ-UHFFFAOYSA-N Erucic acid Natural products CCCCCCC=CCCCCCCCCCCCC(O)=O URXZXNYJPAJJOQ-UHFFFAOYSA-N 0.000 description 2
- 102100026545 Fibronectin type III domain-containing protein 3B Human genes 0.000 description 2
- 240000006927 Foeniculum vulgare Species 0.000 description 2
- 235000004204 Foeniculum vulgare Nutrition 0.000 description 2
- 102100025444 Gamma-butyrobetaine dioxygenase Human genes 0.000 description 2
- 108010067121 Gamma-butyrobetaine dioxygenase Proteins 0.000 description 2
- 108700039691 Genetic Promoter Regions Proteins 0.000 description 2
- GLZPCOQZEFWAFX-UHFFFAOYSA-N Geraniol Chemical compound CC(C)=CCCC(C)=CCO GLZPCOQZEFWAFX-UHFFFAOYSA-N 0.000 description 2
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 2
- 101000798476 Homo sapiens B-cell CLL/lymphoma 7 protein family member C Proteins 0.000 description 2
- 101000864175 Homo sapiens DNA polymerase epsilon subunit 3 Proteins 0.000 description 2
- 101000915580 Homo sapiens E3 ubiquitin-protein ligase ZNRF1 Proteins 0.000 description 2
- 101000913642 Homo sapiens Fibronectin type III domain-containing protein 3B Proteins 0.000 description 2
- 101001056699 Homo sapiens Intersectin-2 Proteins 0.000 description 2
- 101001024120 Homo sapiens Nipped-B-like protein Proteins 0.000 description 2
- 101001068628 Homo sapiens Protein PRRC2C Proteins 0.000 description 2
- 101000668416 Homo sapiens Regulator of chromosome condensation Proteins 0.000 description 2
- 101000628693 Homo sapiens Serine/threonine-protein kinase 25 Proteins 0.000 description 2
- 101000800113 Homo sapiens THO complex subunit 2 Proteins 0.000 description 2
- 101000772767 Homo sapiens Ubiquitin-like protein 5 Proteins 0.000 description 2
- 101000772888 Homo sapiens Ubiquitin-protein ligase E3A Proteins 0.000 description 2
- 235000010650 Hyssopus officinalis Nutrition 0.000 description 2
- 240000001812 Hyssopus officinalis Species 0.000 description 2
- 235000008227 Illicium verum Nutrition 0.000 description 2
- 240000007232 Illicium verum Species 0.000 description 2
- 208000022559 Inflammatory bowel disease Diseases 0.000 description 2
- 102000003777 Interleukin-1 beta Human genes 0.000 description 2
- 108090000193 Interleukin-1 beta Proteins 0.000 description 2
- 108090001005 Interleukin-6 Proteins 0.000 description 2
- 102000004889 Interleukin-6 Human genes 0.000 description 2
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 2
- UQSXHKLRYXJYBZ-UHFFFAOYSA-N Iron oxide Chemical compound [Fe]=O UQSXHKLRYXJYBZ-UHFFFAOYSA-N 0.000 description 2
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 2
- QNAYBMKLOCPYGJ-REOHCLBHSA-N L-alanine Chemical compound C[C@H](N)C(O)=O QNAYBMKLOCPYGJ-REOHCLBHSA-N 0.000 description 2
- 150000000994 L-ascorbates Chemical class 0.000 description 2
- 244000165082 Lavanda vera Species 0.000 description 2
- 235000010663 Lavandula angustifolia Nutrition 0.000 description 2
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 2
- 235000014837 Malpighia glabra Nutrition 0.000 description 2
- 240000003394 Malpighia glabra Species 0.000 description 2
- 244000137850 Marrubium vulgare Species 0.000 description 2
- 235000005321 Marrubium vulgare Nutrition 0.000 description 2
- 240000004658 Medicago sativa Species 0.000 description 2
- 244000062730 Melissa officinalis Species 0.000 description 2
- 235000010654 Melissa officinalis Nutrition 0.000 description 2
- 235000014749 Mentha crispa Nutrition 0.000 description 2
- 235000004357 Mentha x piperita Nutrition 0.000 description 2
- 108060004795 Methyltransferase Proteins 0.000 description 2
- 241001661345 Moesziomyces antarcticus Species 0.000 description 2
- 101100167320 Monascus purpureus mpl4 gene Proteins 0.000 description 2
- 108020002334 Monoacylglycerol lipase Proteins 0.000 description 2
- 102100029814 Monoglyceride lipase Human genes 0.000 description 2
- 241000498617 Mucor javanicus Species 0.000 description 2
- OVBPIULPVIDEAO-UHFFFAOYSA-N N-Pteroyl-L-glutaminsaeure Natural products C=1N=C2NC(N)=NC(=O)C2=NC=1CNC1=CC=C(C(=O)NC(CCC(O)=O)C(O)=O)C=C1 OVBPIULPVIDEAO-UHFFFAOYSA-N 0.000 description 2
- WHNWPMSKXPGLAX-UHFFFAOYSA-N N-Vinyl-2-pyrrolidone Chemical compound C=CN1CCCC1=O WHNWPMSKXPGLAX-UHFFFAOYSA-N 0.000 description 2
- 108010057466 NF-kappa B Proteins 0.000 description 2
- 102000003945 NF-kappa B Human genes 0.000 description 2
- 102100023031 Neural Wiskott-Aldrich syndrome protein Human genes 0.000 description 2
- 101710134234 Neural Wiskott-Aldrich syndrome protein Proteins 0.000 description 2
- 108010088373 Neurofilament Proteins Proteins 0.000 description 2
- 102000008763 Neurofilament Proteins Human genes 0.000 description 2
- 102100024007 Neurofilament heavy polypeptide Human genes 0.000 description 2
- PVNIIMVLHYAWGP-UHFFFAOYSA-N Niacin Chemical compound OC(=O)C1=CC=CN=C1 PVNIIMVLHYAWGP-UHFFFAOYSA-N 0.000 description 2
- DFPAKSUCGFBDDF-UHFFFAOYSA-N Nicotinamide Chemical compound NC(=O)C1=CC=CN=C1 DFPAKSUCGFBDDF-UHFFFAOYSA-N 0.000 description 2
- 102100035377 Nipped-B-like protein Human genes 0.000 description 2
- 102100023049 Nuclear factor 1 X-type Human genes 0.000 description 2
- 101710140810 Nuclear factor 1 X-type Proteins 0.000 description 2
- 235000010676 Ocimum basilicum Nutrition 0.000 description 2
- 240000007926 Ocimum gratissimum Species 0.000 description 2
- 239000005642 Oleic acid Substances 0.000 description 2
- 108010068204 Peptide Elongation Factors Proteins 0.000 description 2
- 102000002508 Peptide Elongation Factors Human genes 0.000 description 2
- 241000283216 Phocidae Species 0.000 description 2
- 235000006990 Pimenta dioica Nutrition 0.000 description 2
- 240000008474 Pimenta dioica Species 0.000 description 2
- 235000012550 Pimpinella anisum Nutrition 0.000 description 2
- 240000004760 Pimpinella anisum Species 0.000 description 2
- 235000008184 Piper nigrum Nutrition 0.000 description 2
- 244000203593 Piper nigrum Species 0.000 description 2
- WCUXLLCKKVVCTQ-UHFFFAOYSA-M Potassium chloride Chemical compound [Cl-].[K+] WCUXLLCKKVVCTQ-UHFFFAOYSA-M 0.000 description 2
- 102100033952 Protein PRRC2C Human genes 0.000 description 2
- 241000589540 Pseudomonas fluorescens Species 0.000 description 2
- 102100021672 Pumilio homolog 1 Human genes 0.000 description 2
- 101710087391 Pumilio homolog 1 Proteins 0.000 description 2
- 101150061360 RBP1 gene Proteins 0.000 description 2
- 102000009572 RNA Polymerase II Human genes 0.000 description 2
- 108010009460 RNA Polymerase II Proteins 0.000 description 2
- 230000004570 RNA-binding Effects 0.000 description 2
- 102100022127 Radixin Human genes 0.000 description 2
- 101100083561 Rattus norvegicus Pdxp gene Proteins 0.000 description 2
- 102100039977 Regulator of chromosome condensation Human genes 0.000 description 2
- 201000000582 Retinoblastoma Diseases 0.000 description 2
- 108010071010 Retinoblastoma-Binding Protein 2 Proteins 0.000 description 2
- 101000968489 Rhizomucor miehei Lipase Proteins 0.000 description 2
- 241000220317 Rosa Species 0.000 description 2
- 102000037054 SLC-Transporter Human genes 0.000 description 2
- 108091006207 SLC-Transporter Proteins 0.000 description 2
- BUGBHKTXTAQXES-UHFFFAOYSA-N Selenium Chemical compound [Se] BUGBHKTXTAQXES-UHFFFAOYSA-N 0.000 description 2
- 102000013008 Semaphorin-3A Human genes 0.000 description 2
- 108010090319 Semaphorin-3A Proteins 0.000 description 2
- 102100026737 Serine/threonine-protein kinase 25 Human genes 0.000 description 2
- 244000000231 Sesamum indicum Species 0.000 description 2
- 235000003434 Sesamum indicum Nutrition 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- 235000021355 Stearic acid Nutrition 0.000 description 2
- 229930006000 Sucrose Natural products 0.000 description 2
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 2
- 102100021682 Syntaxin-binding protein 5 Human genes 0.000 description 2
- 102000006467 TATA-Box Binding Protein Human genes 0.000 description 2
- 108010044281 TATA-Box Binding Protein Proteins 0.000 description 2
- 102100040296 TATA-box-binding protein Human genes 0.000 description 2
- 102100033491 THO complex subunit 2 Human genes 0.000 description 2
- 102100024817 TNF receptor-associated factor 6 Human genes 0.000 description 2
- 108090000009 TNF receptor-associated factor 6 Proteins 0.000 description 2
- 101000984201 Thermomyces lanuginosus Lipase Proteins 0.000 description 2
- 235000007303 Thymus vulgaris Nutrition 0.000 description 2
- 240000002657 Thymus vulgaris Species 0.000 description 2
- HXWJFEZDFPRLBG-UHFFFAOYSA-N Timnodonic acid Natural products CCCC=CC=CCC=CCC=CCC=CCCCC(O)=O HXWJFEZDFPRLBG-UHFFFAOYSA-N 0.000 description 2
- 235000001484 Trigonella foenum graecum Nutrition 0.000 description 2
- 244000250129 Trigonella foenum graecum Species 0.000 description 2
- 101150089725 Tspoap1 gene Proteins 0.000 description 2
- LFTYTUAZOPRMMI-CFRASDGPSA-N UDP-N-acetyl-alpha-D-glucosamine Chemical compound O1[C@H](CO)[C@@H](O)[C@H](O)[C@@H](NC(=O)C)[C@H]1OP(O)(=O)OP(O)(=O)OC[C@@H]1[C@@H](O)[C@@H](O)[C@H](N2C(NC(=O)C=C2)=O)O1 LFTYTUAZOPRMMI-CFRASDGPSA-N 0.000 description 2
- LFTYTUAZOPRMMI-UHFFFAOYSA-N UNPD164450 Natural products O1C(CO)C(O)C(O)C(NC(=O)C)C1OP(O)(=O)OP(O)(=O)OCC1C(O)C(O)C(N2C(NC(=O)C=C2)=O)O1 LFTYTUAZOPRMMI-UHFFFAOYSA-N 0.000 description 2
- 102100030580 Ubiquitin-like protein 5 Human genes 0.000 description 2
- 102100030434 Ubiquitin-protein ligase E3A Human genes 0.000 description 2
- 244000290333 Vanilla fragrans Species 0.000 description 2
- 235000009499 Vanilla fragrans Nutrition 0.000 description 2
- FPIPGXGPPPQFEQ-BOOMUCAASA-N Vitamin A Natural products OC/C=C(/C)\C=C\C=C(\C)/C=C/C1=C(C)CCCC1(C)C FPIPGXGPPPQFEQ-BOOMUCAASA-N 0.000 description 2
- 229930003268 Vitamin C Natural products 0.000 description 2
- 208000021017 Weight Gain Diseases 0.000 description 2
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 2
- 230000009471 action Effects 0.000 description 2
- 208000038016 acute inflammation Diseases 0.000 description 2
- 230000006022 acute inflammation Effects 0.000 description 2
- 230000004658 acute-phase response Effects 0.000 description 2
- 125000002252 acyl group Chemical group 0.000 description 2
- 230000010933 acylation Effects 0.000 description 2
- 238000005917 acylation reaction Methods 0.000 description 2
- 238000007259 addition reaction Methods 0.000 description 2
- 235000004279 alanine Nutrition 0.000 description 2
- 150000001299 aldehydes Chemical class 0.000 description 2
- AHANXAKGNAKFSK-PDBXOOCHSA-N all-cis-icosa-11,14,17-trienoic acid Chemical compound CC\C=C/C\C=C/C\C=C/CCCCCCCCCC(O)=O AHANXAKGNAKFSK-PDBXOOCHSA-N 0.000 description 2
- OENHQHLEOONYIE-UKMVMLAPSA-N all-trans beta-carotene Natural products CC=1CCCC(C)(C)C=1/C=C/C(/C)=C/C=C/C(/C)=C/C=C/C=C(C)C=CC=C(C)C=CC1=C(C)CCCC1(C)C OENHQHLEOONYIE-UKMVMLAPSA-N 0.000 description 2
- FPIPGXGPPPQFEQ-OVSJKPMPSA-N all-trans-retinol Chemical compound OC\C=C(/C)\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C FPIPGXGPPPQFEQ-OVSJKPMPSA-N 0.000 description 2
- 229940024606 amino acid Drugs 0.000 description 2
- 235000001014 amino acid Nutrition 0.000 description 2
- 150000001413 amino acids Chemical class 0.000 description 2
- RDOXTESZEPMUJZ-UHFFFAOYSA-N anisole Chemical compound COC1=CC=CC=C1 RDOXTESZEPMUJZ-UHFFFAOYSA-N 0.000 description 2
- 239000001387 apium graveolens Substances 0.000 description 2
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 2
- 238000003491 array Methods 0.000 description 2
- 208000015802 attention deficit-hyperactivity disease Diseases 0.000 description 2
- 230000001363 autoimmune Effects 0.000 description 2
- WQZGKKKJIJFFOK-FPRJBGLDSA-N beta-D-galactose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@H]1O WQZGKKKJIJFFOK-FPRJBGLDSA-N 0.000 description 2
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 2
- 108010005774 beta-Galactosidase Proteins 0.000 description 2
- 235000013734 beta-carotene Nutrition 0.000 description 2
- 239000011648 beta-carotene Substances 0.000 description 2
- TUPZEYHYWIEDIH-WAIFQNFQSA-N beta-carotene Natural products CC(=C/C=C/C=C(C)/C=C/C=C(C)/C=C/C1=C(C)CCCC1(C)C)C=CC=C(/C)C=CC2=CCCCC2(C)C TUPZEYHYWIEDIH-WAIFQNFQSA-N 0.000 description 2
- 229960002747 betacarotene Drugs 0.000 description 2
- 235000013361 beverage Nutrition 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- 210000004369 blood Anatomy 0.000 description 2
- 239000008280 blood Substances 0.000 description 2
- 229910000019 calcium carbonate Inorganic materials 0.000 description 2
- 235000010216 calcium carbonate Nutrition 0.000 description 2
- FDSDTBUPSURDBL-LOFNIBRQSA-N canthaxanthin Chemical compound CC=1C(=O)CCC(C)(C)C=1/C=C/C(/C)=C/C=C/C(/C)=C/C=C/C=C(C)C=CC=C(C)C=CC1=C(C)C(=O)CCC1(C)C FDSDTBUPSURDBL-LOFNIBRQSA-N 0.000 description 2
- 239000001728 capsicum frutescens Substances 0.000 description 2
- 210000002421 cell wall Anatomy 0.000 description 2
- 210000003483 chromatin Anatomy 0.000 description 2
- 210000000349 chromosome Anatomy 0.000 description 2
- KJPRLNWUNMBNBZ-UHFFFAOYSA-N cinnamic aldehyde Natural products O=CC=CC1=CC=CC=C1 KJPRLNWUNMBNBZ-UHFFFAOYSA-N 0.000 description 2
- 229940117916 cinnamic aldehyde Drugs 0.000 description 2
- SECPZKHBENQXJG-UHFFFAOYSA-N cis-palmitoleic acid Natural products CCCCCCC=CCCCCCCCC(O)=O SECPZKHBENQXJG-UHFFFAOYSA-N 0.000 description 2
- 208000010877 cognitive disease Diseases 0.000 description 2
- 230000003931 cognitive performance Effects 0.000 description 2
- 230000001276 controlling effect Effects 0.000 description 2
- 235000003373 curcuma longa Nutrition 0.000 description 2
- VFLDPWHFBUODDF-FCXRPNKRSA-N curcumin Chemical compound C1=C(O)C(OC)=CC(\C=C\C(=O)CC(=O)\C=C\C=2C=C(OC)C(O)=CC=2)=C1 VFLDPWHFBUODDF-FCXRPNKRSA-N 0.000 description 2
- 229960002104 cyanocobalamin Drugs 0.000 description 2
- 235000000639 cyanocobalamin Nutrition 0.000 description 2
- 239000011666 cyanocobalamin Substances 0.000 description 2
- 230000006378 damage Effects 0.000 description 2
- 230000003247 decreasing effect Effects 0.000 description 2
- FMGSKLZLMKYGDP-USOAJAOKSA-N dehydroepiandrosterone Chemical compound C1[C@@H](O)CC[C@]2(C)[C@H]3CC[C@](C)(C(CC4)=O)[C@@H]4[C@@H]3CC=C21 FMGSKLZLMKYGDP-USOAJAOKSA-N 0.000 description 2
- 238000001212 derivatisation Methods 0.000 description 2
- 108010050073 dethiobiotin synthetase Proteins 0.000 description 2
- HOBAELRKJCKHQD-QNEBEIHSSA-N dihomo-γ-linolenic acid Chemical compound CCCCC\C=C/C\C=C/C\C=C/CCCCCCC(O)=O HOBAELRKJCKHQD-QNEBEIHSSA-N 0.000 description 2
- XIVFQYWMMJWUCD-UHFFFAOYSA-N dihydrophaseic acid Natural products C1C(O)CC2(C)OCC1(C)C2(O)C=CC(C)=CC(O)=O XIVFQYWMMJWUCD-UHFFFAOYSA-N 0.000 description 2
- SLPJGDQJLTYWCI-UHFFFAOYSA-N dimethyl-(4,5,6,7-tetrabromo-1h-benzoimidazol-2-yl)-amine Chemical compound BrC1=C(Br)C(Br)=C2NC(N(C)C)=NC2=C1Br SLPJGDQJLTYWCI-UHFFFAOYSA-N 0.000 description 2
- UKMSUNONTOPOIO-UHFFFAOYSA-N docosanoic acid Chemical compound CCCCCCCCCCCCCCCCCCCCCC(O)=O UKMSUNONTOPOIO-UHFFFAOYSA-N 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- JAZBEHYOTPTENJ-UHFFFAOYSA-N eicosapentaenoic acid Natural products CCC=CCC=CCC=CCC=CCC=CCCCC(O)=O JAZBEHYOTPTENJ-UHFFFAOYSA-N 0.000 description 2
- 239000002702 enteric coating Substances 0.000 description 2
- 238000009505 enteric coating Methods 0.000 description 2
- 230000002255 enzymatic effect Effects 0.000 description 2
- DPUOLQHDNGRHBS-KTKRTIGZSA-N erucic acid Chemical compound CCCCCCCC\C=C/CCCCCCCCCCCC(O)=O DPUOLQHDNGRHBS-KTKRTIGZSA-N 0.000 description 2
- 210000003743 erythrocyte Anatomy 0.000 description 2
- CBOQJANXLMLOSS-UHFFFAOYSA-N ethyl vanillin Chemical compound CCOC1=CC(C=O)=CC=C1O CBOQJANXLMLOSS-UHFFFAOYSA-N 0.000 description 2
- 235000019688 fish Nutrition 0.000 description 2
- 239000000796 flavoring agent Substances 0.000 description 2
- 235000019634 flavors Nutrition 0.000 description 2
- 235000013312 flour Nutrition 0.000 description 2
- 229960000304 folic acid Drugs 0.000 description 2
- 239000011724 folic acid Substances 0.000 description 2
- 230000008014 freezing Effects 0.000 description 2
- 238000007710 freezing Methods 0.000 description 2
- 238000000769 gas chromatography-flame ionisation detection Methods 0.000 description 2
- HIGQPQRQIQDZMP-DHZHZOJOSA-N geranyl acetate Chemical compound CC(C)=CCC\C(C)=C\COC(C)=O HIGQPQRQIQDZMP-DHZHZOJOSA-N 0.000 description 2
- 235000013761 grape skin extract Nutrition 0.000 description 2
- 230000012010 growth Effects 0.000 description 2
- CKDDRHZIAZRDBW-UHFFFAOYSA-N henicosanoic acid Chemical compound CCCCCCCCCCCCCCCCCCCCC(O)=O CKDDRHZIAZRDBW-UHFFFAOYSA-N 0.000 description 2
- VXZBFBRLRNDJCS-UHFFFAOYSA-N heptacosanoic acid Chemical compound CCCCCCCCCCCCCCCCCCCCCCCCCCC(O)=O VXZBFBRLRNDJCS-UHFFFAOYSA-N 0.000 description 2
- KEMQGTRYUADPNZ-UHFFFAOYSA-N heptadecanoic acid Chemical compound CCCCCCCCCCCCCCCCC(O)=O KEMQGTRYUADPNZ-UHFFFAOYSA-N 0.000 description 2
- 235000019514 herring Nutrition 0.000 description 2
- XMHIUKTWLZUKEX-UHFFFAOYSA-N hexacosanoic acid Chemical compound CCCCCCCCCCCCCCCCCCCCCCCCCC(O)=O XMHIUKTWLZUKEX-UHFFFAOYSA-N 0.000 description 2
- IPCSVZSSVZVIGE-UHFFFAOYSA-N hexadecanoic acid Chemical compound CCCCCCCCCCCCCCCC(O)=O IPCSVZSSVZVIGE-UHFFFAOYSA-N 0.000 description 2
- 238000004128 high performance liquid chromatography Methods 0.000 description 2
- VKOBVWXKNCXXDE-UHFFFAOYSA-N icosanoic acid Chemical compound CCCCCCCCCCCCCCCCCCCC(O)=O VKOBVWXKNCXXDE-UHFFFAOYSA-N 0.000 description 2
- 238000001727 in vivo Methods 0.000 description 2
- 230000006698 induction Effects 0.000 description 2
- 229940060367 inert ingredients Drugs 0.000 description 2
- 230000002757 inflammatory effect Effects 0.000 description 2
- 230000002452 interceptive effect Effects 0.000 description 2
- 229940100601 interleukin-6 Drugs 0.000 description 2
- 210000000265 leukocyte Anatomy 0.000 description 2
- 239000003446 ligand Substances 0.000 description 2
- XMGQYMWWDOXHJM-UHFFFAOYSA-N limonene Chemical compound CC(=C)C1CCC(C)=CC1 XMGQYMWWDOXHJM-UHFFFAOYSA-N 0.000 description 2
- UWKAYLJWKGQEPM-UHFFFAOYSA-N linalool acetate Natural products CC(C)=CCCC(C)(C=C)OC(C)=O UWKAYLJWKGQEPM-UHFFFAOYSA-N 0.000 description 2
- UWKAYLJWKGQEPM-LBPRGKRZSA-N linalyl acetate Chemical compound CC(C)=CCC[C@](C)(C=C)OC(C)=O UWKAYLJWKGQEPM-LBPRGKRZSA-N 0.000 description 2
- 229960004488 linolenic acid Drugs 0.000 description 2
- KQQKGWQCNNTQJW-UHFFFAOYSA-N linolenic acid Natural products CC=CCCC=CCC=CCCCCCCCC(O)=O KQQKGWQCNNTQJW-UHFFFAOYSA-N 0.000 description 2
- 235000019359 magnesium stearate Nutrition 0.000 description 2
- 235000012054 meals Nutrition 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- 230000001404 mediated effect Effects 0.000 description 2
- 206010027175 memory impairment Diseases 0.000 description 2
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 2
- 235000013336 milk Nutrition 0.000 description 2
- 239000008267 milk Substances 0.000 description 2
- 210000004080 milk Anatomy 0.000 description 2
- 239000003226 mitogen Substances 0.000 description 2
- 238000000199 molecular distillation Methods 0.000 description 2
- 210000000107 myocyte Anatomy 0.000 description 2
- WQEPLUUGTLDZJY-UHFFFAOYSA-N n-Pentadecanoic acid Natural products CCCCCCCCCCCCCCC(O)=O WQEPLUUGTLDZJY-UHFFFAOYSA-N 0.000 description 2
- 230000001537 neural effect Effects 0.000 description 2
- 229960003966 nicotinamide Drugs 0.000 description 2
- 235000005152 nicotinamide Nutrition 0.000 description 2
- 239000011570 nicotinamide Substances 0.000 description 2
- 235000001968 nicotinic acid Nutrition 0.000 description 2
- 229960003512 nicotinic acid Drugs 0.000 description 2
- 239000011664 nicotinic acid Substances 0.000 description 2
- IHEJEKZAKSNRLY-UHFFFAOYSA-N nonacosanoic acid Chemical compound CCCCCCCCCCCCCCCCCCCCCCCCCCCCC(O)=O IHEJEKZAKSNRLY-UHFFFAOYSA-N 0.000 description 2
- ISYWECDDZWTKFF-UHFFFAOYSA-N nonadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCCC(O)=O ISYWECDDZWTKFF-UHFFFAOYSA-N 0.000 description 2
- 210000000633 nuclear envelope Anatomy 0.000 description 2
- UTOPWMOLSKOLTQ-UHFFFAOYSA-N octacosanoic acid Chemical compound CCCCCCCCCCCCCCCCCCCCCCCCCCCC(O)=O UTOPWMOLSKOLTQ-UHFFFAOYSA-N 0.000 description 2
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 2
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 2
- 230000001575 pathological effect Effects 0.000 description 2
- MWMPEAHGUXCSMY-UHFFFAOYSA-N pentacosanoic acid Chemical compound CCCCCCCCCCCCCCCCCCCCCCCCC(O)=O MWMPEAHGUXCSMY-UHFFFAOYSA-N 0.000 description 2
- 239000008194 pharmaceutical composition Substances 0.000 description 2
- 150000003905 phosphatidylinositols Chemical class 0.000 description 2
- SHUZOJHMOBOZST-UHFFFAOYSA-N phylloquinone Natural products CC(C)CCCCC(C)CCC(C)CCCC(=CCC1=C(C)C(=O)c2ccccc2C1=O)C SHUZOJHMOBOZST-UHFFFAOYSA-N 0.000 description 2
- 230000001766 physiological effect Effects 0.000 description 2
- 229940068196 placebo Drugs 0.000 description 2
- 239000000902 placebo Substances 0.000 description 2
- LXNHXLLTXMVWPM-UHFFFAOYSA-N pyridoxine Chemical compound CC1=NC=C(CO)C(CO)=C1O LXNHXLLTXMVWPM-UHFFFAOYSA-N 0.000 description 2
- 108010048484 radixin Proteins 0.000 description 2
- 108010014186 ras Proteins Proteins 0.000 description 2
- 230000003252 repetitive effect Effects 0.000 description 2
- 230000004044 response Effects 0.000 description 2
- 235000019192 riboflavin Nutrition 0.000 description 2
- 239000002151 riboflavin Substances 0.000 description 2
- 229960002477 riboflavin Drugs 0.000 description 2
- 210000003705 ribosome Anatomy 0.000 description 2
- 239000004248 saffron Substances 0.000 description 2
- 235000013974 saffron Nutrition 0.000 description 2
- 235000002020 sage Nutrition 0.000 description 2
- 239000000523 sample Substances 0.000 description 2
- 238000005070 sampling Methods 0.000 description 2
- 239000011669 selenium Substances 0.000 description 2
- 229910052711 selenium Inorganic materials 0.000 description 2
- 229940091258 selenium supplement Drugs 0.000 description 2
- 210000000582 semen Anatomy 0.000 description 2
- 238000000526 short-path distillation Methods 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 229940083466 soybean lecithin Drugs 0.000 description 2
- 241000894007 species Species 0.000 description 2
- 238000012453 sprague-dawley rat model Methods 0.000 description 2
- 239000008117 stearic acid Substances 0.000 description 2
- JIWBIWFOSCKQMA-UHFFFAOYSA-N stearidonic acid Natural products CCC=CCC=CCC=CCC=CCCCCC(O)=O JIWBIWFOSCKQMA-UHFFFAOYSA-N 0.000 description 2
- 239000005720 sucrose Substances 0.000 description 2
- 235000013616 tea Nutrition 0.000 description 2
- JZRWCGZRTZMZEH-UHFFFAOYSA-N thiamine Chemical compound CC1=C(CCO)SC=[N+]1CC1=CN=C(C)N=C1N JZRWCGZRTZMZEH-UHFFFAOYSA-N 0.000 description 2
- 239000001585 thymus vulgaris Substances 0.000 description 2
- RUVINXPYWBROJD-ONEGZZNKSA-N trans-anethole Chemical compound COC1=CC=C(\C=C\C)C=C1 RUVINXPYWBROJD-ONEGZZNKSA-N 0.000 description 2
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 2
- XEZVDURJDFGERA-UHFFFAOYSA-N tricosanoic acid Chemical compound CCCCCCCCCCCCCCCCCCCCCCC(O)=O XEZVDURJDFGERA-UHFFFAOYSA-N 0.000 description 2
- SZHOJFHSIKHZHA-UHFFFAOYSA-N tridecanoic acid Chemical compound CCCCCCCCCCCCC(O)=O SZHOJFHSIKHZHA-UHFFFAOYSA-N 0.000 description 2
- 235000001019 trigonella foenum-graecum Nutrition 0.000 description 2
- 208000001072 type 2 diabetes mellitus Diseases 0.000 description 2
- ZDPHROOEEOARMN-UHFFFAOYSA-N undecanoic acid Chemical compound CCCCCCCCCCC(O)=O ZDPHROOEEOARMN-UHFFFAOYSA-N 0.000 description 2
- 206010047302 ventricular tachycardia Diseases 0.000 description 2
- 235000019155 vitamin A Nutrition 0.000 description 2
- 239000011719 vitamin A Substances 0.000 description 2
- 235000019154 vitamin C Nutrition 0.000 description 2
- 239000011718 vitamin C Substances 0.000 description 2
- 229940045997 vitamin a Drugs 0.000 description 2
- 230000004584 weight gain Effects 0.000 description 2
- 235000019786 weight gain Nutrition 0.000 description 2
- 239000011701 zinc Substances 0.000 description 2
- 229910052725 zinc Inorganic materials 0.000 description 2
- XLOMVQKBTHCTTD-UHFFFAOYSA-N zinc oxide Inorganic materials [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 description 2
- OENHQHLEOONYIE-JLTXGRSLSA-N β-Carotene Chemical compound CC=1CCCC(C)(C)C=1\C=C\C(\C)=C\C=C\C(\C)=C\C=C\C=C(/C)\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C OENHQHLEOONYIE-JLTXGRSLSA-N 0.000 description 2
- XSXIVVZCUAHUJO-AVQMFFATSA-N (11e,14e)-icosa-11,14-dienoic acid Chemical compound CCCCC\C=C\C\C=C\CCCCCCCCCC(O)=O XSXIVVZCUAHUJO-AVQMFFATSA-N 0.000 description 1
- GWHCXVQVJPWHRF-KTKRTIGZSA-N (15Z)-tetracosenoic acid Chemical compound CCCCCCCC\C=C/CCCCCCCCCCCCCC(O)=O GWHCXVQVJPWHRF-KTKRTIGZSA-N 0.000 description 1
- MJYQFWSXKFLTAY-OVEQLNGDSA-N (2r,3r)-2,3-bis[(4-hydroxy-3-methoxyphenyl)methyl]butane-1,4-diol;(2r,3r,4s,5s,6r)-6-(hydroxymethyl)oxane-2,3,4,5-tetrol Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O.C1=C(O)C(OC)=CC(C[C@@H](CO)[C@H](CO)CC=2C=C(OC)C(O)=CC=2)=C1 MJYQFWSXKFLTAY-OVEQLNGDSA-N 0.000 description 1
- LNAZSHAWQACDHT-XIYTZBAFSA-N (2r,3r,4s,5r,6s)-4,5-dimethoxy-2-(methoxymethyl)-3-[(2s,3r,4s,5r,6r)-3,4,5-trimethoxy-6-(methoxymethyl)oxan-2-yl]oxy-6-[(2r,3r,4s,5r,6r)-4,5,6-trimethoxy-2-(methoxymethyl)oxan-3-yl]oxyoxane Chemical compound CO[C@@H]1[C@@H](OC)[C@H](OC)[C@@H](COC)O[C@H]1O[C@H]1[C@H](OC)[C@@H](OC)[C@H](O[C@H]2[C@@H]([C@@H](OC)[C@H](OC)O[C@@H]2COC)OC)O[C@@H]1COC LNAZSHAWQACDHT-XIYTZBAFSA-N 0.000 description 1
- 239000001490 (3R)-3,7-dimethylocta-1,6-dien-3-ol Substances 0.000 description 1
- XUHRVZXFBWDCFB-QRTDKPMLSA-N (3R)-4-[[(3S,6S,9S,12R,15S,18R,21R,24R,27R,28R)-12-(3-amino-3-oxopropyl)-6-[(2S)-butan-2-yl]-3-(2-carboxyethyl)-18-(hydroxymethyl)-28-methyl-9,15,21,24-tetrakis(2-methylpropyl)-2,5,8,11,14,17,20,23,26-nonaoxo-1-oxa-4,7,10,13,16,19,22,25-octazacyclooctacos-27-yl]amino]-3-[[(2R)-2-[[(3S)-3-hydroxydecanoyl]amino]-4-methylpentanoyl]amino]-4-oxobutanoic acid Chemical compound CCCCCCC[C@H](O)CC(=O)N[C@H](CC(C)C)C(=O)N[C@H](CC(O)=O)C(=O)N[C@@H]1[C@@H](C)OC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](CCC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](CO)NC(=O)[C@@H](CC(C)C)NC(=O)[C@@H](CC(C)C)NC1=O)[C@@H](C)CC XUHRVZXFBWDCFB-QRTDKPMLSA-N 0.000 description 1
- UNSRRHDPHVZAHH-YOILPLPUSA-N (5Z,8Z,11Z)-icosatrienoic acid Chemical compound CCCCCCCC\C=C/C\C=C/C\C=C/CCCC(O)=O UNSRRHDPHVZAHH-YOILPLPUSA-N 0.000 description 1
- YUFFSWGQGVEMMI-RCHUDCCISA-N (7e,10e,13e,16e,19e)-docosa-7,10,13,16,19-pentaenoic acid Chemical compound CC\C=C\C\C=C\C\C=C\C\C=C\C\C=C\CCCCCC(O)=O YUFFSWGQGVEMMI-RCHUDCCISA-N 0.000 description 1
- HOBAELRKJCKHQD-UHFFFAOYSA-N (8Z,11Z,14Z)-8,11,14-eicosatrienoic acid Natural products CCCCCC=CCC=CCC=CCCCCCCC(O)=O HOBAELRKJCKHQD-UHFFFAOYSA-N 0.000 description 1
- KSEBMYQBYZTDHS-HWKANZROSA-M (E)-Ferulic acid Natural products COC1=CC(\C=C\C([O-])=O)=CC=C1O KSEBMYQBYZTDHS-HWKANZROSA-M 0.000 description 1
- CDOSHBSSFJOMGT-JTQLQIEISA-N (R)-linalool Natural products CC(C)=CCC[C@@](C)(O)C=C CDOSHBSSFJOMGT-JTQLQIEISA-N 0.000 description 1
- KSDMISMEMOGBFU-UHFFFAOYSA-N (all-Z)-7,10,13-Eicosatrienoic acid Natural products CCCCCCC=CCC=CCC=CCCCCCC(O)=O KSDMISMEMOGBFU-UHFFFAOYSA-N 0.000 description 1
- HQPCSDADVLFHHO-UHFFFAOYSA-N (all-Z)-8,11,14,17-Eicosatetraenoic acid Natural products CCC=CCC=CCC=CCC=CCCCCCCC(O)=O HQPCSDADVLFHHO-UHFFFAOYSA-N 0.000 description 1
- PORPENFLTBBHSG-MGBGTMOVSA-N 1,2-dihexadecanoyl-sn-glycerol-3-phosphate Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP(O)(O)=O)OC(=O)CCCCCCCCCCCCCCC PORPENFLTBBHSG-MGBGTMOVSA-N 0.000 description 1
- MLYCFWZIAJAIGW-UHFFFAOYSA-N 1-(2,5-dimethoxy-4-methylphenyl)butan-2-amine Chemical compound CCC(N)CC1=CC(OC)=C(C)C=C1OC MLYCFWZIAJAIGW-UHFFFAOYSA-N 0.000 description 1
- GYSCBCSGKXNZRH-UHFFFAOYSA-N 1-benzothiophene-2-carboxamide Chemical compound C1=CC=C2SC(C(=O)N)=CC2=C1 GYSCBCSGKXNZRH-UHFFFAOYSA-N 0.000 description 1
- HNAGHMKIPMKKBB-UHFFFAOYSA-N 1-benzylpyrrolidine-3-carboxamide Chemical compound C1C(C(=O)N)CCN1CC1=CC=CC=C1 HNAGHMKIPMKKBB-UHFFFAOYSA-N 0.000 description 1
- WRGQSWVCFNIUNZ-GDCKJWNLSA-N 1-oleoyl-sn-glycerol 3-phosphate Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OC[C@@H](O)COP(O)(O)=O WRGQSWVCFNIUNZ-GDCKJWNLSA-N 0.000 description 1
- MSWZFWKMSRAUBD-GASJEMHNSA-N 2-amino-2-deoxy-D-galactopyranose Chemical compound N[C@H]1C(O)O[C@H](CO)[C@H](O)[C@@H]1O MSWZFWKMSRAUBD-GASJEMHNSA-N 0.000 description 1
- CFWRDBDJAOHXSH-SECBINFHSA-N 2-azaniumylethyl [(2r)-2,3-diacetyloxypropyl] phosphate Chemical compound CC(=O)OC[C@@H](OC(C)=O)COP(O)(=O)OCCN CFWRDBDJAOHXSH-SECBINFHSA-N 0.000 description 1
- TWJNQYPJQDRXPH-UHFFFAOYSA-N 2-cyanobenzohydrazide Chemical compound NNC(=O)C1=CC=CC=C1C#N TWJNQYPJQDRXPH-UHFFFAOYSA-N 0.000 description 1
- MIDXCONKKJTLDX-UHFFFAOYSA-N 3,5-dimethylcyclopentane-1,2-dione Chemical compound CC1CC(C)C(=O)C1=O MIDXCONKKJTLDX-UHFFFAOYSA-N 0.000 description 1
- PMYDPQQPEAYXKD-UHFFFAOYSA-N 3-hydroxy-n-naphthalen-2-ylnaphthalene-2-carboxamide Chemical compound C1=CC=CC2=CC(NC(=O)C3=CC4=CC=CC=C4C=C3O)=CC=C21 PMYDPQQPEAYXKD-UHFFFAOYSA-N 0.000 description 1
- AJBZENLMTKDAEK-UHFFFAOYSA-N 3a,5a,5b,8,8,11a-hexamethyl-1-prop-1-en-2-yl-1,2,3,4,5,6,7,7a,9,10,11,11b,12,13,13a,13b-hexadecahydrocyclopenta[a]chrysene-4,9-diol Chemical compound CC12CCC(O)C(C)(C)C1CCC(C1(C)CC3O)(C)C2CCC1C1C3(C)CCC1C(=C)C AJBZENLMTKDAEK-UHFFFAOYSA-N 0.000 description 1
- AVKOENOBFIYBSA-GUTOPQIJSA-N 4,7,10,13,16-Docosapentaenoic acid Chemical compound CCCCC\C=C\C\C=C\C\C=C\C\C=C\C\C=C\CCC(O)=O AVKOENOBFIYBSA-GUTOPQIJSA-N 0.000 description 1
- AVKOENOBFIYBSA-UHFFFAOYSA-N 4,7,10,13,16-Docosapentaenoic acid Natural products CCCCCC=CCC=CCC=CCC=CCC=CCCC(O)=O AVKOENOBFIYBSA-UHFFFAOYSA-N 0.000 description 1
- JHPNVNIEXXLNTR-UHFFFAOYSA-N 4-(trimethylammonio)butanoate Chemical compound C[N+](C)(C)CCCC([O-])=O JHPNVNIEXXLNTR-UHFFFAOYSA-N 0.000 description 1
- 108010021561 4-Nitrophenylphosphatase Proteins 0.000 description 1
- 102100022721 40S ribosomal protein S25 Human genes 0.000 description 1
- 102100022289 60S ribosomal protein L13a Human genes 0.000 description 1
- 102100031002 60S ribosomal protein L36a Human genes 0.000 description 1
- ZMKDEQUXYDZSNN-OKLKQMLOSA-N 6E,9E-octadecadienoic acid Chemical compound CCCCCCCC\C=C\C\C=C\CCCCC(O)=O ZMKDEQUXYDZSNN-OKLKQMLOSA-N 0.000 description 1
- AUNGANRZJHBGPY-MBNYWOFBSA-N 7,8-dimethyl-10-[(2R,3R,4S)-2,3,4,5-tetrahydroxypentyl]benzo[g]pteridine-2,4-dione Chemical compound OC[C@H](O)[C@H](O)[C@H](O)CN1C=2C=C(C)C(C)=CC=2N=C2C1=NC(=O)NC2=O AUNGANRZJHBGPY-MBNYWOFBSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- JBYXPOFIGCOSSB-GOJKSUSPSA-N 9-cis,11-trans-octadecadienoic acid Chemical compound CCCCCC\C=C\C=C/CCCCCCCC(O)=O JBYXPOFIGCOSSB-GOJKSUSPSA-N 0.000 description 1
- 102100040084 A-kinase anchor protein 9 Human genes 0.000 description 1
- 102100028359 ADP-ribosylation factor-like protein 6 Human genes 0.000 description 1
- 102100032814 ATP-dependent zinc metalloprotease YME1L1 Human genes 0.000 description 1
- 102000016190 Abl interactor 1 Human genes 0.000 description 1
- 108050004693 Abl interactor 1 Proteins 0.000 description 1
- 102100037768 Acetyl-CoA acetyltransferase, mitochondrial Human genes 0.000 description 1
- 102100021029 Activating signal cointegrator 1 complex subunit 3 Human genes 0.000 description 1
- 102100034542 Acyl-CoA (8-3)-desaturase Human genes 0.000 description 1
- 102100028264 Alanyl-tRNA editing protein Aarsd1 Human genes 0.000 description 1
- 240000002234 Allium sativum Species 0.000 description 1
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 1
- 208000024827 Alzheimer disease Diseases 0.000 description 1
- 102000013455 Amyloid beta-Peptides Human genes 0.000 description 1
- 108010090849 Amyloid beta-Peptides Proteins 0.000 description 1
- 244000061520 Angelica archangelica Species 0.000 description 1
- 235000007070 Angelica archangelica Nutrition 0.000 description 1
- 208000008822 Ankylosis Diseases 0.000 description 1
- 102100039158 Ankyrin repeat domain-containing protein 53 Human genes 0.000 description 1
- 108010049777 Ankyrins Proteins 0.000 description 1
- 102000008102 Ankyrins Human genes 0.000 description 1
- 240000007087 Apium graveolens Species 0.000 description 1
- 235000002764 Apium graveolens Nutrition 0.000 description 1
- 102100021569 Apoptosis regulator Bcl-2 Human genes 0.000 description 1
- 101100465060 Arabidopsis thaliana PRK4 gene Proteins 0.000 description 1
- 240000005528 Arctium lappa Species 0.000 description 1
- 235000003130 Arctium lappa Nutrition 0.000 description 1
- 235000008078 Arctium minus Nutrition 0.000 description 1
- 102100023221 Arginine and glutamate-rich protein 1 Human genes 0.000 description 1
- 241001064577 Ariadne <plant> Species 0.000 description 1
- 235000016425 Arthrospira platensis Nutrition 0.000 description 1
- 240000002900 Arthrospira platensis Species 0.000 description 1
- 108090000448 Aryl Hydrocarbon Receptors Proteins 0.000 description 1
- 102100026792 Aryl hydrocarbon receptor Human genes 0.000 description 1
- 241000512259 Ascophyllum nodosum Species 0.000 description 1
- 108010011485 Aspartame Proteins 0.000 description 1
- JEBFVOLFMLUKLF-IFPLVEIFSA-N Astaxanthin Natural products CC(=C/C=C/C(=C/C=C/C1=C(C)C(=O)C(O)CC1(C)C)/C)C=CC=C(/C)C=CC=C(/C)C=CC2=C(C)C(=O)C(O)CC2(C)C JEBFVOLFMLUKLF-IFPLVEIFSA-N 0.000 description 1
- 206010003658 Atrial Fibrillation Diseases 0.000 description 1
- 108091012583 BCL2 Proteins 0.000 description 1
- 101150045555 Bbox1 gene Proteins 0.000 description 1
- 235000021357 Behenic acid Nutrition 0.000 description 1
- 101710117026 Biotin synthase Proteins 0.000 description 1
- 239000004135 Bone phosphate Substances 0.000 description 1
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 1
- 241000219198 Brassica Species 0.000 description 1
- 235000006463 Brassica alba Nutrition 0.000 description 1
- 235000003351 Brassica cretica Nutrition 0.000 description 1
- 244000140786 Brassica hirta Species 0.000 description 1
- 235000011371 Brassica hirta Nutrition 0.000 description 1
- 244000178993 Brassica juncea Species 0.000 description 1
- 235000011332 Brassica juncea Nutrition 0.000 description 1
- 235000014700 Brassica juncea var napiformis Nutrition 0.000 description 1
- 235000011291 Brassica nigra Nutrition 0.000 description 1
- 244000180419 Brassica nigra Species 0.000 description 1
- 235000011299 Brassica oleracea var botrytis Nutrition 0.000 description 1
- 235000017647 Brassica oleracea var italica Nutrition 0.000 description 1
- 240000003259 Brassica oleracea var. botrytis Species 0.000 description 1
- 235000003343 Brassica rupestris Nutrition 0.000 description 1
- 102100029895 Bromodomain-containing protein 4 Human genes 0.000 description 1
- 101710133654 C-5 sterol desaturase Proteins 0.000 description 1
- 102100025905 C-Jun-amino-terminal kinase-interacting protein 4 Human genes 0.000 description 1
- 101710105201 C-Jun-amino-terminal kinase-interacting protein 4 Proteins 0.000 description 1
- 102100026197 C-type lectin domain family 2 member D Human genes 0.000 description 1
- 101710153512 C-type lectin domain family 2 member D Proteins 0.000 description 1
- YEBDWAHEIMUJQT-XVSDJDOKSA-N CCCCCC=CCC=CCC=CCC=CCCCC(O)=O.CCCCC\C=C/C\C=C/C\C=C/C\C=C/CCCC(O)=O Chemical compound CCCCCC=CCC=CCC=CCC=CCCCC(O)=O.CCCCC\C=C/C\C=C/C\C=C/C\C=C/CCCC(O)=O YEBDWAHEIMUJQT-XVSDJDOKSA-N 0.000 description 1
- 108010092574 CD69 antigen Proteins 0.000 description 1
- 108060001253 CD99 Proteins 0.000 description 1
- 102000024905 CD99 Human genes 0.000 description 1
- 108010070033 COP9 Signalosome Complex Proteins 0.000 description 1
- 102000005643 COP9 Signalosome Complex Human genes 0.000 description 1
- 108010040163 CREB-Binding Protein Proteins 0.000 description 1
- 102100028011 CTD small phosphatase-like protein 2 Human genes 0.000 description 1
- 101100009017 Caenorhabditis elegans dcr-1 gene Proteins 0.000 description 1
- 235000014161 Caesalpinia gilliesii Nutrition 0.000 description 1
- 244000003240 Caesalpinia gilliesii Species 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- 102100024436 Caldesmon Human genes 0.000 description 1
- 235000003880 Calendula Nutrition 0.000 description 1
- 235000005881 Calendula officinalis Nutrition 0.000 description 1
- 241000282472 Canis lupus familiaris Species 0.000 description 1
- 235000002566 Capsicum Nutrition 0.000 description 1
- 235000002567 Capsicum annuum Nutrition 0.000 description 1
- 235000002283 Capsicum annuum var aviculare Nutrition 0.000 description 1
- 235000013303 Capsicum annuum var. frutescens Nutrition 0.000 description 1
- 235000002284 Capsicum baccatum var baccatum Nutrition 0.000 description 1
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 1
- 108010078791 Carrier Proteins Proteins 0.000 description 1
- 108010076119 Caseins Proteins 0.000 description 1
- 102000011632 Caseins Human genes 0.000 description 1
- 108010076667 Caspases Proteins 0.000 description 1
- 102000011727 Caspases Human genes 0.000 description 1
- 102000007768 Cellular Retinol-Binding Proteins Human genes 0.000 description 1
- 108010021988 Cellular Retinol-Binding Proteins Proteins 0.000 description 1
- 229920000623 Cellulose acetate phthalate Polymers 0.000 description 1
- 102100028776 Centrosome and spindle pole-associated protein 1 Human genes 0.000 description 1
- 241000283153 Cetacea Species 0.000 description 1
- 235000007866 Chamaemelum nobile Nutrition 0.000 description 1
- 240000003538 Chamaemelum nobile Species 0.000 description 1
- 102000019034 Chemokines Human genes 0.000 description 1
- 108010012236 Chemokines Proteins 0.000 description 1
- GHOKWGTUZJEAQD-UHFFFAOYSA-N Chick antidermatitis factor Natural products OCC(C)(C)C(O)C(=O)NCCC(O)=O GHOKWGTUZJEAQD-UHFFFAOYSA-N 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 description 1
- 229910021555 Chromium Chloride Inorganic materials 0.000 description 1
- 102000017589 Chromo domains Human genes 0.000 description 1
- 108050005811 Chromo domains Proteins 0.000 description 1
- 102100032918 Chromobox protein homolog 5 Human genes 0.000 description 1
- 235000007871 Chrysanthemum coronarium Nutrition 0.000 description 1
- 244000067456 Chrysanthemum coronarium Species 0.000 description 1
- 102100035396 Cingulin-like protein 1 Human genes 0.000 description 1
- 235000021511 Cinnamomum cassia Nutrition 0.000 description 1
- 102100036444 Clathrin interactor 1 Human genes 0.000 description 1
- 101710148711 Clathrin interactor 1 Proteins 0.000 description 1
- 102000004360 Cofilin 1 Human genes 0.000 description 1
- 108090000996 Cofilin 1 Proteins 0.000 description 1
- 102100032377 Coiled-coil domain-containing protein 107 Human genes 0.000 description 1
- 206010062759 Congenital dyskeratosis Diseases 0.000 description 1
- 102100032648 Copine-3 Human genes 0.000 description 1
- 102100029384 Copine-8 Human genes 0.000 description 1
- 101710125091 Copine-8 Proteins 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- OCUCCJIRFHNWBP-IYEMJOQQSA-L Copper gluconate Chemical compound [Cu+2].OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C([O-])=O.OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C([O-])=O OCUCCJIRFHNWBP-IYEMJOQQSA-L 0.000 description 1
- 229920002261 Corn starch Polymers 0.000 description 1
- 229920002785 Croscarmellose sodium Polymers 0.000 description 1
- 241000238424 Crustacea Species 0.000 description 1
- 235000003392 Curcuma domestica Nutrition 0.000 description 1
- 108090000266 Cyclin-dependent kinases Proteins 0.000 description 1
- 102000003903 Cyclin-dependent kinases Human genes 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- HMFHBZSHGGEWLO-SOOFDHNKSA-N D-ribofuranose Chemical compound OC[C@H]1OC(O)[C@H](O)[C@@H]1O HMFHBZSHGGEWLO-SOOFDHNKSA-N 0.000 description 1
- 102100036402 DAP3-binding cell death enhancer 1 Human genes 0.000 description 1
- 108020004414 DNA Proteins 0.000 description 1
- 108090000323 DNA Topoisomerases Proteins 0.000 description 1
- 102100021603 DNA excision repair protein ERCC-6-like 2 Human genes 0.000 description 1
- 101710184972 DNA polymerase epsilon subunit 3 Proteins 0.000 description 1
- 102100024607 DNA topoisomerase 1 Human genes 0.000 description 1
- 102100031230 DNA topoisomerase I, mitochondrial Human genes 0.000 description 1
- 101710094860 DNA topoisomerase I, mitochondrial Proteins 0.000 description 1
- GHVNFZFCNZKVNT-UHFFFAOYSA-N Decanoic acid Natural products CCCCCCCCCC(O)=O GHVNFZFCNZKVNT-UHFFFAOYSA-N 0.000 description 1
- 102100036511 Dehydrodolichyl diphosphate synthase complex subunit DHDDS Human genes 0.000 description 1
- 101710111046 Delta(7)-sterol 5(6)-desaturase Proteins 0.000 description 1
- 108010073542 Delta-5 Fatty Acid Desaturase Proteins 0.000 description 1
- 102100034289 Deoxynucleoside triphosphate triphosphohydrolase SAMHD1 Human genes 0.000 description 1
- 235000019739 Dicalciumphosphate Nutrition 0.000 description 1
- 235000021298 Dihomo-γ-linolenic acid Nutrition 0.000 description 1
- 102000016680 Dioxygenases Human genes 0.000 description 1
- 108010028143 Dioxygenases Proteins 0.000 description 1
- 206010013142 Disinhibition Diseases 0.000 description 1
- 101800001224 Disintegrin Proteins 0.000 description 1
- 102100023316 DnaJ homolog subfamily B member 14 Human genes 0.000 description 1
- 108700019125 Drosophila Em7-HLH Proteins 0.000 description 1
- 101100009019 Drosophila melanogaster Dcr-1 gene Proteins 0.000 description 1
- 102100024827 Dynamin-1-like protein Human genes 0.000 description 1
- 102100025682 Dystroglycan 1 Human genes 0.000 description 1
- 108010071885 Dystroglycans Proteins 0.000 description 1
- 102100023227 E3 SUMO-protein ligase EGR2 Human genes 0.000 description 1
- 102100023149 E3 ubiquitin-protein ligase MARCHF6 Human genes 0.000 description 1
- 102100037715 E3 ubiquitin-protein ligase MSL2 Human genes 0.000 description 1
- 102100031438 E3 ubiquitin-protein ligase RING1 Human genes 0.000 description 1
- 101710093740 E3 ubiquitin-protein ligase RING1 Proteins 0.000 description 1
- 102100034209 E3 ubiquitin-protein ligase RNF126 Human genes 0.000 description 1
- 101710162554 E3 ubiquitin-protein ligase RNF126 Proteins 0.000 description 1
- 102100038662 E3 ubiquitin-protein ligase SMURF2 Human genes 0.000 description 1
- 102100034235 ELAV-like protein 1 Human genes 0.000 description 1
- 102100034232 ER membrane protein complex subunit 9 Human genes 0.000 description 1
- 102100023078 Early endosome antigen 1 Human genes 0.000 description 1
- 101710116100 Ectonucleoside triphosphate diphosphohydrolase 5 Proteins 0.000 description 1
- 102000002322 Egg Proteins Human genes 0.000 description 1
- 108010000912 Egg Proteins Proteins 0.000 description 1
- 235000018602 Elettaria cardamomum Nutrition 0.000 description 1
- 241001632410 Eleutherococcus senticosus Species 0.000 description 1
- 102100030209 Elongin-B Human genes 0.000 description 1
- 102100033183 Epithelial membrane protein 1 Human genes 0.000 description 1
- 206010049466 Erythroblastosis Diseases 0.000 description 1
- 102100036089 Fascin Human genes 0.000 description 1
- 241000282326 Felis catus Species 0.000 description 1
- DKKCQDROTDCQOR-UHFFFAOYSA-L Ferrous lactate Chemical compound [Fe+2].CC(O)C([O-])=O.CC(O)C([O-])=O DKKCQDROTDCQOR-UHFFFAOYSA-L 0.000 description 1
- 201000008808 Fibrosarcoma Diseases 0.000 description 1
- 102100028122 Forkhead box protein P1 Human genes 0.000 description 1
- 241000748691 Furia Species 0.000 description 1
- OPGOLNDOMSBSCW-CLNHMMGSSA-N Fursultiamine hydrochloride Chemical compound Cl.C1CCOC1CSSC(\CCO)=C(/C)N(C=O)CC1=CN=C(C)N=C1N OPGOLNDOMSBSCW-CLNHMMGSSA-N 0.000 description 1
- 102100020976 G kinase-anchoring protein 1 Human genes 0.000 description 1
- 101710184398 G kinase-anchoring protein 1 Proteins 0.000 description 1
- 102100039806 G patch domain-containing protein 1 Human genes 0.000 description 1
- 102000034286 G proteins Human genes 0.000 description 1
- 108091006027 G proteins Proteins 0.000 description 1
- 102100024422 GTPase IMAP family member 7 Human genes 0.000 description 1
- 241000287828 Gallus gallus Species 0.000 description 1
- 102100034004 Gamma-adducin Human genes 0.000 description 1
- 239000005792 Geraniol Substances 0.000 description 1
- GLZPCOQZEFWAFX-YFHOEESVSA-N Geraniol Natural products CC(C)=CCC\C(C)=C/CO GLZPCOQZEFWAFX-YFHOEESVSA-N 0.000 description 1
- 235000008100 Ginkgo biloba Nutrition 0.000 description 1
- 244000194101 Ginkgo biloba Species 0.000 description 1
- JZNWSCPGTDBMEW-UHFFFAOYSA-N Glycerophosphorylethanolamin Natural products NCCOP(O)(=O)OCC(O)CO JZNWSCPGTDBMEW-UHFFFAOYSA-N 0.000 description 1
- 102000003886 Glycoproteins Human genes 0.000 description 1
- 108090000288 Glycoproteins Proteins 0.000 description 1
- 101150007061 Gnai2 gene Proteins 0.000 description 1
- 102100021182 Golgi integral membrane protein 4 Human genes 0.000 description 1
- 101710140584 Golgi integral membrane protein 4 Proteins 0.000 description 1
- 108010067218 Guanine Nucleotide Exchange Factors Proteins 0.000 description 1
- 102000016285 Guanine Nucleotide Exchange Factors Human genes 0.000 description 1
- 102100031249 H/ACA ribonucleoprotein complex subunit DKC1 Human genes 0.000 description 1
- 101710167047 H/ACA ribonucleoprotein complex subunit DKC1 Proteins 0.000 description 1
- 102100030488 HEAT repeat-containing protein 6 Human genes 0.000 description 1
- 108010061414 Hepatocyte Nuclear Factor 1-beta Proteins 0.000 description 1
- 102100022123 Hepatocyte nuclear factor 1-beta Human genes 0.000 description 1
- 244000153234 Hibiscus abelmoschus Species 0.000 description 1
- 235000010616 Hibiscus abelmoschus Nutrition 0.000 description 1
- 102100038885 Histone acetyltransferase p300 Human genes 0.000 description 1
- 102100022103 Histone-lysine N-methyltransferase 2A Human genes 0.000 description 1
- 108050002855 Histone-lysine N-methyltransferase 2A Proteins 0.000 description 1
- 108010033040 Histones Proteins 0.000 description 1
- 101000678929 Homo sapiens 40S ribosomal protein S25 Proteins 0.000 description 1
- 101000681240 Homo sapiens 60S ribosomal protein L13a Proteins 0.000 description 1
- 101001127203 Homo sapiens 60S ribosomal protein L36a Proteins 0.000 description 1
- 101001127258 Homo sapiens 60S ribosomal protein L36a-like Proteins 0.000 description 1
- 101000890598 Homo sapiens A-kinase anchor protein 9 Proteins 0.000 description 1
- 101000769028 Homo sapiens ADP-ribosylation factor-like protein 6 Proteins 0.000 description 1
- 101000708560 Homo sapiens ATP-dependent zinc metalloprotease YME1L1 Proteins 0.000 description 1
- 101000598552 Homo sapiens Acetyl-CoA acetyltransferase, mitochondrial Proteins 0.000 description 1
- 101000784211 Homo sapiens Activating signal cointegrator 1 complex subunit 3 Proteins 0.000 description 1
- 101000724444 Homo sapiens Alanyl-tRNA editing protein Aarsd1 Proteins 0.000 description 1
- 101000889569 Homo sapiens Ankyrin repeat domain-containing protein 53 Proteins 0.000 description 1
- 101000685364 Homo sapiens Arginine and glutamate-rich protein 1 Proteins 0.000 description 1
- 101000794024 Homo sapiens Bromodomain-containing protein 4 Proteins 0.000 description 1
- 101000896987 Homo sapiens CREB-binding protein Proteins 0.000 description 1
- 101000859047 Homo sapiens CTD small phosphatase-like protein 2 Proteins 0.000 description 1
- 101000910297 Homo sapiens Caldesmon Proteins 0.000 description 1
- 101000916452 Homo sapiens Centrosome and spindle pole-associated protein 1 Proteins 0.000 description 1
- 101000797581 Homo sapiens Chromobox protein homolog 5 Proteins 0.000 description 1
- 101000737619 Homo sapiens Cingulin-like protein 1 Proteins 0.000 description 1
- 101000868800 Homo sapiens Coiled-coil domain-containing protein 107 Proteins 0.000 description 1
- 101000929221 Homo sapiens DAP3-binding cell death enhancer 1 Proteins 0.000 description 1
- 101000876524 Homo sapiens DNA excision repair protein ERCC-6-like Proteins 0.000 description 1
- 101000898683 Homo sapiens DNA excision repair protein ERCC-6-like 2 Proteins 0.000 description 1
- 101000641031 Homo sapiens Deoxynucleoside triphosphate triphosphohydrolase SAMHD1 Proteins 0.000 description 1
- 101000908034 Homo sapiens DnaJ homolog subfamily B member 14 Proteins 0.000 description 1
- 101000909218 Homo sapiens Dynamin-1-like protein Proteins 0.000 description 1
- 101001049692 Homo sapiens E3 SUMO-protein ligase EGR2 Proteins 0.000 description 1
- 101001027791 Homo sapiens E3 ubiquitin-protein ligase MSL2 Proteins 0.000 description 1
- 101000664952 Homo sapiens E3 ubiquitin-protein ligase SMURF2 Proteins 0.000 description 1
- 101000925870 Homo sapiens ELAV-like protein 1 Proteins 0.000 description 1
- 101000925833 Homo sapiens ER membrane protein complex subunit 9 Proteins 0.000 description 1
- 101001011859 Homo sapiens Elongin-A Proteins 0.000 description 1
- 101001011846 Homo sapiens Elongin-B Proteins 0.000 description 1
- 101000881731 Homo sapiens Elongin-C Proteins 0.000 description 1
- 101001059893 Homo sapiens Forkhead box protein P1 Proteins 0.000 description 1
- 101001034116 Homo sapiens G patch domain-containing protein 1 Proteins 0.000 description 1
- 101000833390 Homo sapiens GTPase IMAP family member 7 Proteins 0.000 description 1
- 101000799011 Homo sapiens Gamma-adducin Proteins 0.000 description 1
- 101000990566 Homo sapiens HEAT repeat-containing protein 6 Proteins 0.000 description 1
- 101000585618 Homo sapiens Leptin receptor gene-related protein Proteins 0.000 description 1
- 101001007394 Homo sapiens Leukocyte receptor cluster member 8 Proteins 0.000 description 1
- 101100129369 Homo sapiens MARCHF6 gene Proteins 0.000 description 1
- 101001116520 Homo sapiens Myotubularin-related protein 11 Proteins 0.000 description 1
- 101000972796 Homo sapiens NF-kappa-B-activating protein Proteins 0.000 description 1
- 101000634091 Homo sapiens Nuclear speckle splicing regulatory protein 1 Proteins 0.000 description 1
- 101000651906 Homo sapiens Paired amphipathic helix protein Sin3a Proteins 0.000 description 1
- 101000801684 Homo sapiens Phospholipid-transporting ATPase ABCA1 Proteins 0.000 description 1
- 101000906619 Homo sapiens Polyribonucleotide 5'-hydroxyl-kinase Clp1 Proteins 0.000 description 1
- 101001077420 Homo sapiens Potassium voltage-gated channel subfamily H member 7 Proteins 0.000 description 1
- 101000605345 Homo sapiens Prefoldin subunit 1 Proteins 0.000 description 1
- 101000983170 Homo sapiens Proliferation-associated protein 2G4 Proteins 0.000 description 1
- 101000912957 Homo sapiens Protein DEK Proteins 0.000 description 1
- 101001061807 Homo sapiens Rab-like protein 6 Proteins 0.000 description 1
- 101000836005 Homo sapiens S-phase kinase-associated protein 1 Proteins 0.000 description 1
- 101000617778 Homo sapiens SNF-related serine/threonine-protein kinase Proteins 0.000 description 1
- 101001041365 Homo sapiens Serine protease HTRA2, mitochondrial Proteins 0.000 description 1
- 101000628647 Homo sapiens Serine/threonine-protein kinase 24 Proteins 0.000 description 1
- 101000987317 Homo sapiens Serine/threonine-protein kinase PAK 1 Proteins 0.000 description 1
- 101000577652 Homo sapiens Serine/threonine-protein kinase PRP4 homolog Proteins 0.000 description 1
- 101000825726 Homo sapiens Structural maintenance of chromosomes protein 4 Proteins 0.000 description 1
- 101000820494 Homo sapiens Syntaxin-binding protein 5 Proteins 0.000 description 1
- 101001099181 Homo sapiens TATA-binding protein-associated factor 2N Proteins 0.000 description 1
- 101000891620 Homo sapiens TBC1 domain family member 1 Proteins 0.000 description 1
- 101000665486 Homo sapiens TBC1 domain family member 12 Proteins 0.000 description 1
- 101000889527 Homo sapiens TOG array regulator of axonemal microtubules protein 1 Proteins 0.000 description 1
- 101000805518 Homo sapiens Transcription cofactor vestigial-like protein 4 Proteins 0.000 description 1
- 101000596771 Homo sapiens Transcription factor 7-like 2 Proteins 0.000 description 1
- 101000715069 Homo sapiens Transcription initiation factor TFIID subunit 10 Proteins 0.000 description 1
- 101000714746 Homo sapiens Transmembrane protein 161B Proteins 0.000 description 1
- 101000577737 Homo sapiens U4/U6 small nuclear ribonucleoprotein Prp4 Proteins 0.000 description 1
- 101000777156 Homo sapiens UBX domain-containing protein 4 Proteins 0.000 description 1
- 101000777138 Homo sapiens Ubiquitin carboxyl-terminal hydrolase 42 Proteins 0.000 description 1
- 101000585623 Homo sapiens Unconventional myosin-X Proteins 0.000 description 1
- 101000771655 Homo sapiens WD repeat and FYVE domain-containing protein 1 Proteins 0.000 description 1
- 101000976607 Homo sapiens Zinc finger protein 135 Proteins 0.000 description 1
- 101000857273 Homo sapiens Zinc finger protein GLIS2 Proteins 0.000 description 1
- 101000743197 Homo sapiens pre-mRNA 3' end processing protein WDR33 Proteins 0.000 description 1
- XSXIVVZCUAHUJO-UHFFFAOYSA-N Homo-gamma-linoleic acid Natural products CCCCCC=CCC=CCCCCCCCCCC(O)=O XSXIVVZCUAHUJO-UHFFFAOYSA-N 0.000 description 1
- 206010020751 Hypersensitivity Diseases 0.000 description 1
- 235000004185 Hyptis suaveolens Nutrition 0.000 description 1
- 108010042653 IgA receptor Proteins 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 206010022489 Insulin Resistance Diseases 0.000 description 1
- 108090000723 Insulin-Like Growth Factor I Proteins 0.000 description 1
- 102100037852 Insulin-like growth factor I Human genes 0.000 description 1
- 102000014150 Interferons Human genes 0.000 description 1
- 108010050904 Interferons Proteins 0.000 description 1
- 102000045959 Interleukin-1 Receptor-Like 1 Human genes 0.000 description 1
- 108700003107 Interleukin-1 Receptor-Like 1 Proteins 0.000 description 1
- 101710088362 Intersectin-2 Proteins 0.000 description 1
- 206010023198 Joint ankylosis Diseases 0.000 description 1
- 108010076876 Keratins Proteins 0.000 description 1
- 102000011782 Keratins Human genes 0.000 description 1
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 description 1
- QIVBCDIJIAJPQS-VIFPVBQESA-N L-tryptophane Chemical compound C1=CC=C2C(C[C@H](N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-VIFPVBQESA-N 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- 101710137448 Lathosterol oxidase Proteins 0.000 description 1
- 102100037199 Lathosterol oxidase Human genes 0.000 description 1
- 239000005639 Lauric acid Substances 0.000 description 1
- 235000017858 Laurus nobilis Nutrition 0.000 description 1
- 244000147568 Laurus nobilis Species 0.000 description 1
- 235000010701 Lavanda vera Nutrition 0.000 description 1
- 108090001090 Lectins Proteins 0.000 description 1
- 102000004856 Lectins Human genes 0.000 description 1
- 102100029825 Leptin receptor gene-related protein Human genes 0.000 description 1
- ROHFNLRQFUQHCH-UHFFFAOYSA-N Leucine Natural products CC(C)CC(N)C(O)=O ROHFNLRQFUQHCH-UHFFFAOYSA-N 0.000 description 1
- 206010024305 Leukaemia monocytic Diseases 0.000 description 1
- 102100028297 Leukocyte receptor cluster member 8 Human genes 0.000 description 1
- 108090000364 Ligases Proteins 0.000 description 1
- 102000003960 Ligases Human genes 0.000 description 1
- 235000021353 Lignoceric acid Nutrition 0.000 description 1
- CQXMAMUUWHYSIY-UHFFFAOYSA-N Lignoceric acid Natural products CCCCCCCCCCCCCCCCCCCCCCCC(=O)OCCC1=CC=C(O)C=C1 CQXMAMUUWHYSIY-UHFFFAOYSA-N 0.000 description 1
- OYHQOLUKZRVURQ-HZJYTTRNSA-N Linoleic acid Chemical compound CCCCC\C=C/C\C=C/CCCCCCCC(O)=O OYHQOLUKZRVURQ-HZJYTTRNSA-N 0.000 description 1
- UPYKUZBSLRQECL-UKMVMLAPSA-N Lycopene Natural products CC(=C/C=C/C=C(C)/C=C/C=C(C)/C=C/C1C(=C)CCCC1(C)C)C=CC=C(/C)C=CC2C(=C)CCCC2(C)C UPYKUZBSLRQECL-UKMVMLAPSA-N 0.000 description 1
- JEVVKJMRZMXFBT-XWDZUXABSA-N Lycophyll Natural products OC/C(=C/CC/C(=C\C=C\C(=C/C=C/C(=C\C=C\C=C(/C=C/C=C(\C=C\C=C(/CC/C=C(/CO)\C)\C)/C)\C)/C)\C)/C)/C JEVVKJMRZMXFBT-XWDZUXABSA-N 0.000 description 1
- 108010018650 MEF2 Transcription Factors Proteins 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- 229930195725 Mannitol Natural products 0.000 description 1
- 235000010624 Medicago sativa Nutrition 0.000 description 1
- 235000017587 Medicago sativa ssp. sativa Nutrition 0.000 description 1
- 108010037255 Member 7 Tumor Necrosis Factor Receptor Superfamily Proteins 0.000 description 1
- 102000018697 Membrane Proteins Human genes 0.000 description 1
- 108010052285 Membrane Proteins Proteins 0.000 description 1
- 208000026139 Memory disease Diseases 0.000 description 1
- ABSPRNADVQNDOU-UHFFFAOYSA-N Menaquinone 1 Natural products C1=CC=C2C(=O)C(CC=C(C)C)=C(C)C(=O)C2=C1 ABSPRNADVQNDOU-UHFFFAOYSA-N 0.000 description 1
- 244000024873 Mentha crispa Species 0.000 description 1
- 244000246386 Mentha pulegium Species 0.000 description 1
- 235000016257 Mentha pulegium Nutrition 0.000 description 1
- 244000078639 Mentha spicata Species 0.000 description 1
- 241001479543 Mentha x piperita Species 0.000 description 1
- 102000036858 Metal ion transporters Human genes 0.000 description 1
- 108091006974 Metal ion transporters Proteins 0.000 description 1
- 108010006035 Metalloproteases Proteins 0.000 description 1
- 102000005741 Metalloproteases Human genes 0.000 description 1
- 206010059521 Methylmalonic aciduria Diseases 0.000 description 1
- 229920000168 Microcrystalline cellulose Polymers 0.000 description 1
- 235000005135 Micromeria juliana Nutrition 0.000 description 1
- 108091022875 Microtubule Proteins 0.000 description 1
- 102000029749 Microtubule Human genes 0.000 description 1
- 108010020004 Microtubule-Associated Proteins Proteins 0.000 description 1
- 102000009664 Microtubule-Associated Proteins Human genes 0.000 description 1
- 101710169105 Minor spike protein Proteins 0.000 description 1
- 101710081079 Minor spike protein H Proteins 0.000 description 1
- ZOKXTWBITQBERF-UHFFFAOYSA-N Molybdenum Chemical compound [Mo] ZOKXTWBITQBERF-UHFFFAOYSA-N 0.000 description 1
- 241000699666 Mus <mouse, genus> Species 0.000 description 1
- 241000699660 Mus musculus Species 0.000 description 1
- 101001060237 Mus musculus F-box/WD repeat-containing protein 7 Proteins 0.000 description 1
- 101000767143 Mus musculus U3 small nucleolar RNA-associated protein 15 homolog Proteins 0.000 description 1
- 208000000112 Myalgia Diseases 0.000 description 1
- 102100039229 Myocyte-specific enhancer factor 2C Human genes 0.000 description 1
- 102100024963 Myotubularin-related protein 11 Human genes 0.000 description 1
- 235000021360 Myristic acid Nutrition 0.000 description 1
- TUNFSRHWOTWDNC-UHFFFAOYSA-N Myristic acid Natural products CCCCCCCCCCCCCC(O)=O TUNFSRHWOTWDNC-UHFFFAOYSA-N 0.000 description 1
- 235000007265 Myrrhis odorata Nutrition 0.000 description 1
- 108010081735 N-Ethylmaleimide-Sensitive Proteins Proteins 0.000 description 1
- 102000004390 N-Ethylmaleimide-Sensitive Proteins Human genes 0.000 description 1
- 102100022580 NF-kappa-B-activating protein Human genes 0.000 description 1
- 102000034570 NR1 subfamily Human genes 0.000 description 1
- 108020001305 NR1 subfamily Proteins 0.000 description 1
- 102000038100 NR2 subfamily Human genes 0.000 description 1
- 108020002076 NR2 subfamily Proteins 0.000 description 1
- 102100036954 Nck-associated protein 1 Human genes 0.000 description 1
- 101710134622 Nck-associated protein 1 Proteins 0.000 description 1
- 101150097297 Nedd4 gene Proteins 0.000 description 1
- XJXROGWVRIJYMO-SJDLZYGOSA-N Nervonic acid Natural products O=C(O)[C@@H](/C=C/CCCCCCCC)CCCCCCCCCCCC XJXROGWVRIJYMO-SJDLZYGOSA-N 0.000 description 1
- 101710159895 Neurofilament heavy polypeptide Proteins 0.000 description 1
- 102000008299 Nitric Oxide Synthase Human genes 0.000 description 1
- 108010021487 Nitric Oxide Synthase Proteins 0.000 description 1
- 102100029230 Nuclear speckle splicing regulatory protein 1 Human genes 0.000 description 1
- 102100036518 Nucleoside diphosphate phosphatase ENTPD5 Human genes 0.000 description 1
- 102100025062 Nucleosome-remodeling factor subunit BPTF Human genes 0.000 description 1
- 108010084438 Oncogene Protein v-maf Proteins 0.000 description 1
- 108700026244 Open Reading Frames Proteins 0.000 description 1
- 235000006297 Origanum majorana Nutrition 0.000 description 1
- 240000000783 Origanum majorana Species 0.000 description 1
- 240000007594 Oryza sativa Species 0.000 description 1
- 235000007164 Oryza sativa Nutrition 0.000 description 1
- 102100029181 PDZ and LIM domain protein 5 Human genes 0.000 description 1
- 101710121657 PDZ and LIM domain protein 5 Proteins 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 102100027334 Paired amphipathic helix protein Sin3a Human genes 0.000 description 1
- 241000206754 Palmaria palmata Species 0.000 description 1
- 235000021314 Palmitic acid Nutrition 0.000 description 1
- 235000021319 Palmitoleic acid Nutrition 0.000 description 1
- 235000008753 Papaver somniferum Nutrition 0.000 description 1
- 244000062780 Petroselinum sativum Species 0.000 description 1
- OOUTWVMJGMVRQF-DOYZGLONSA-N Phoenicoxanthin Natural products CC(=C/C=C/C=C(C)/C=C/C=C(C)/C=C/C1=C(C)C(=O)C(O)CC1(C)C)C=CC=C(/C)C=CC2=C(C)C(=O)CCC2(C)C OOUTWVMJGMVRQF-DOYZGLONSA-N 0.000 description 1
- 102000001105 Phosphofructokinases Human genes 0.000 description 1
- 108010069341 Phosphofructokinases Proteins 0.000 description 1
- 102100036072 Phospholipase A and acyltransferase 1 Human genes 0.000 description 1
- 101710131822 Phospholipase A and acyltransferase 1 Proteins 0.000 description 1
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 1
- 102100030365 Plakophilin-4 Human genes 0.000 description 1
- 108050004723 Plakophilin-4 Proteins 0.000 description 1
- 102100030264 Pleckstrin Human genes 0.000 description 1
- 108010066816 Polypeptide N-acetylgalactosaminyltransferase Proteins 0.000 description 1
- 102100020950 Polypeptide N-acetylgalactosaminyltransferase 2 Human genes 0.000 description 1
- 102100023504 Polyribonucleotide 5'-hydroxyl-kinase Clp1 Human genes 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 102100025133 Potassium voltage-gated channel subfamily H member 7 Human genes 0.000 description 1
- 102100025822 Pre-mRNA-processing factor 40 homolog A Human genes 0.000 description 1
- 101710165431 Pre-mRNA-processing factor 40 homolog A Proteins 0.000 description 1
- 208000008691 Precursor B-Cell Lymphoblastic Leukemia-Lymphoma Diseases 0.000 description 1
- 102100038255 Prefoldin subunit 1 Human genes 0.000 description 1
- 235000016311 Primula vulgaris Nutrition 0.000 description 1
- 244000028344 Primula vulgaris Species 0.000 description 1
- 102100026899 Proliferation-associated protein 2G4 Human genes 0.000 description 1
- 102100034014 Prolyl 3-hydroxylase 3 Human genes 0.000 description 1
- 241000241413 Propolis Species 0.000 description 1
- 102000000471 Prostaglandin F receptors Human genes 0.000 description 1
- 108050008995 Prostaglandin F receptors Proteins 0.000 description 1
- 102100026113 Protein DEK Human genes 0.000 description 1
- 102000001253 Protein Kinase Human genes 0.000 description 1
- 102100034053 Protein PALS2 Human genes 0.000 description 1
- 101710191290 Protein PALS2 Proteins 0.000 description 1
- 108010059000 Protein Phosphatase 1 Proteins 0.000 description 1
- 102000005569 Protein Phosphatase 1 Human genes 0.000 description 1
- 108010058956 Protein Phosphatase 2 Proteins 0.000 description 1
- 102000006478 Protein Phosphatase 2 Human genes 0.000 description 1
- 102100036452 Protein RUFY3 Human genes 0.000 description 1
- 101710179024 Protein RUFY3 Proteins 0.000 description 1
- 108010039518 Proton-Translocating ATPases Proteins 0.000 description 1
- 102000015176 Proton-Translocating ATPases Human genes 0.000 description 1
- 208000001431 Psychomotor Agitation Diseases 0.000 description 1
- REFJWTPEDVJJIY-UHFFFAOYSA-N Quercetin Chemical compound C=1C(O)=CC(O)=C(C(C=2O)=O)C=1OC=2C1=CC=C(O)C(O)=C1 REFJWTPEDVJJIY-UHFFFAOYSA-N 0.000 description 1
- 101710168045 REST corepressor 1 Proteins 0.000 description 1
- 102100024864 REST corepressor 1 Human genes 0.000 description 1
- 101710119903 RING finger protein 39 Proteins 0.000 description 1
- 102100026465 RING finger protein 39 Human genes 0.000 description 1
- 239000013614 RNA sample Substances 0.000 description 1
- 102100025873 RNA-binding protein 27 Human genes 0.000 description 1
- 101710206013 RNA-binding protein 27 Proteins 0.000 description 1
- 102100029618 Rab-like protein 6 Human genes 0.000 description 1
- 241000700157 Rattus norvegicus Species 0.000 description 1
- 101000926909 Rattus norvegicus Golgi resident protein GCP60 Proteins 0.000 description 1
- 101000780661 Rattus norvegicus Protein argonaute-2 Proteins 0.000 description 1
- 101100367090 Rattus norvegicus Suco gene Proteins 0.000 description 1
- 102100030849 Receptor-transporting protein 3 Human genes 0.000 description 1
- 108050009681 Receptor-transporting protein 3 Proteins 0.000 description 1
- 206010038743 Restlessness Diseases 0.000 description 1
- 102100035179 Ribitol-5-phosphate xylosyltransferase 1 Human genes 0.000 description 1
- 101710161553 Ribitol-5-phosphate xylosyltransferase 1 Proteins 0.000 description 1
- 102000004389 Ribonucleoproteins Human genes 0.000 description 1
- 108010081734 Ribonucleoproteins Proteins 0.000 description 1
- PYMYPHUHKUWMLA-LMVFSUKVSA-N Ribose Natural products OC[C@@H](O)[C@@H](O)[C@@H](O)C=O PYMYPHUHKUWMLA-LMVFSUKVSA-N 0.000 description 1
- 102100022010 SNF-related serine/threonine-protein kinase Human genes 0.000 description 1
- 240000007164 Salvia officinalis Species 0.000 description 1
- 235000002912 Salvia officinalis Nutrition 0.000 description 1
- 244000151637 Sambucus canadensis Species 0.000 description 1
- 235000018735 Sambucus canadensis Nutrition 0.000 description 1
- 241001125046 Sardina pilchardus Species 0.000 description 1
- 235000007315 Satureja hortensis Nutrition 0.000 description 1
- 240000002114 Satureja hortensis Species 0.000 description 1
- 102100021117 Serine protease HTRA2, mitochondrial Human genes 0.000 description 1
- 102100026764 Serine/threonine-protein kinase 24 Human genes 0.000 description 1
- 102100027910 Serine/threonine-protein kinase PAK 1 Human genes 0.000 description 1
- 102100028868 Serine/threonine-protein kinase PRP4 homolog Human genes 0.000 description 1
- 102100032015 Serine/threonine-protein kinase tousled-like 1 Human genes 0.000 description 1
- 101710098564 Serine/threonine-protein kinase tousled-like 1 Proteins 0.000 description 1
- 108700000090 Sfrs11 Proteins 0.000 description 1
- 241000320380 Silybum Species 0.000 description 1
- 235000010841 Silybum marianum Nutrition 0.000 description 1
- 102100038626 Sorting nexin-6 Human genes 0.000 description 1
- 108050006109 Sorting nexin-6 Proteins 0.000 description 1
- 108010019965 Spectrin Proteins 0.000 description 1
- 102000005890 Spectrin Human genes 0.000 description 1
- 108010071698 Spermine synthase Proteins 0.000 description 1
- 102100037616 Spermine synthase Human genes 0.000 description 1
- 235000009337 Spinacia oleracea Nutrition 0.000 description 1
- 244000300264 Spinacia oleracea Species 0.000 description 1
- 102100025252 StAR-related lipid transfer protein 13 Human genes 0.000 description 1
- 102100022842 Structural maintenance of chromosomes protein 4 Human genes 0.000 description 1
- 102000004896 Sulfotransferases Human genes 0.000 description 1
- 108090001033 Sulfotransferases Proteins 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- 235000019486 Sunflower oil Nutrition 0.000 description 1
- 102000019215 Supervillin Human genes 0.000 description 1
- 108050006606 Supervillin Proteins 0.000 description 1
- 102100039485 Symplekin Human genes 0.000 description 1
- 101710138921 Symplekin Proteins 0.000 description 1
- 102100030700 Synaptic vesicle glycoprotein 2B Human genes 0.000 description 1
- 101710084139 Synaptic vesicle glycoprotein 2B Proteins 0.000 description 1
- 101710096027 Syntaxin-binding protein 5 Proteins 0.000 description 1
- 102100038721 T-box transcription factor TBX2 Human genes 0.000 description 1
- 101710167702 T-box transcription factor TBX2 Proteins 0.000 description 1
- 102100038917 TATA-binding protein-associated factor 2N Human genes 0.000 description 1
- 102100040238 TBC1 domain family member 1 Human genes 0.000 description 1
- 102100038201 TBC1 domain family member 12 Human genes 0.000 description 1
- 102100039142 TOG array regulator of axonemal microtubules protein 1 Human genes 0.000 description 1
- 235000012308 Tagetes Nutrition 0.000 description 1
- 241000736851 Tagetes Species 0.000 description 1
- 240000001949 Taraxacum officinale Species 0.000 description 1
- 235000005187 Taraxacum officinale ssp. officinale Nutrition 0.000 description 1
- 102100024547 Tensin-1 Human genes 0.000 description 1
- 108010088950 Tensins Proteins 0.000 description 1
- 235000004054 Thymus serpyllum Nutrition 0.000 description 1
- 240000006001 Thymus serpyllum Species 0.000 description 1
- 108010048999 Transcription Factor 3 Proteins 0.000 description 1
- 108010048992 Transcription Factor 4 Proteins 0.000 description 1
- 102100038034 Transcription cofactor vestigial-like protein 4 Human genes 0.000 description 1
- 102100023489 Transcription factor 4 Human genes 0.000 description 1
- 102100035101 Transcription factor 7-like 2 Human genes 0.000 description 1
- 102100038313 Transcription factor E2-alpha Human genes 0.000 description 1
- 102100036677 Transcription initiation factor TFIID subunit 10 Human genes 0.000 description 1
- 102000006612 Transducin Human genes 0.000 description 1
- 108010087042 Transducin Proteins 0.000 description 1
- 102100036388 Transmembrane protein 161B Human genes 0.000 description 1
- 235000015724 Trifolium pratense Nutrition 0.000 description 1
- 102000011408 Tripartite Motif Proteins Human genes 0.000 description 1
- 108010023649 Tripartite Motif Proteins Proteins 0.000 description 1
- QIVBCDIJIAJPQS-UHFFFAOYSA-N Tryptophan Natural products C1=CC=C2C(CC(N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-UHFFFAOYSA-N 0.000 description 1
- 102100033732 Tumor necrosis factor receptor superfamily member 1A Human genes 0.000 description 1
- 102100031308 UBX domain-containing protein 4 Human genes 0.000 description 1
- AXMVYSVVTMKQSL-UHFFFAOYSA-N UNPD142122 Natural products OC1=CC=C(C=CC=O)C=C1O AXMVYSVVTMKQSL-UHFFFAOYSA-N 0.000 description 1
- 102100038458 Ubinuclein-1 Human genes 0.000 description 1
- 101710094188 Ubinuclein-1 Proteins 0.000 description 1
- 108090000848 Ubiquitin Proteins 0.000 description 1
- 102000044159 Ubiquitin Human genes 0.000 description 1
- 102100031310 Ubiquitin carboxyl-terminal hydrolase 42 Human genes 0.000 description 1
- 108010083111 Ubiquitin-Protein Ligases Proteins 0.000 description 1
- 102000006275 Ubiquitin-Protein Ligases Human genes 0.000 description 1
- 108010066496 Ubiquitin-Specific Proteases Proteins 0.000 description 1
- 102000018390 Ubiquitin-Specific Proteases Human genes 0.000 description 1
- 102100029827 Unconventional myosin-X Human genes 0.000 description 1
- 235000009108 Urtica dioica Nutrition 0.000 description 1
- 241000218215 Urticaceae Species 0.000 description 1
- UWHZIFQPPBDJPM-FPLPWBNLSA-M Vaccenic acid Natural products CCCCCC\C=C/CCCCCCCCCC([O-])=O UWHZIFQPPBDJPM-FPLPWBNLSA-M 0.000 description 1
- 235000021322 Vaccenic acid Nutrition 0.000 description 1
- 235000017537 Vaccinium myrtillus Nutrition 0.000 description 1
- 244000078534 Vaccinium myrtillus Species 0.000 description 1
- 235000012036 Vanilla tahitensis Nutrition 0.000 description 1
- 229920001938 Vegetable gum Polymers 0.000 description 1
- 206010047281 Ventricular arrhythmia Diseases 0.000 description 1
- 241000700605 Viruses Species 0.000 description 1
- 206010047571 Visual impairment Diseases 0.000 description 1
- 229930003316 Vitamin D Natural products 0.000 description 1
- QYSXJUFSXHHAJI-XFEUOLMDSA-N Vitamin D3 Natural products C1(/[C@@H]2CC[C@@H]([C@]2(CCC1)C)[C@H](C)CCCC(C)C)=C/C=C1\C[C@@H](O)CCC1=C QYSXJUFSXHHAJI-XFEUOLMDSA-N 0.000 description 1
- 229930003427 Vitamin E Natural products 0.000 description 1
- 229930003448 Vitamin K Natural products 0.000 description 1
- 235000009754 Vitis X bourquina Nutrition 0.000 description 1
- 235000012333 Vitis X labruscana Nutrition 0.000 description 1
- 240000006365 Vitis vinifera Species 0.000 description 1
- 235000014787 Vitis vinifera Nutrition 0.000 description 1
- 102100029468 WD repeat and FYVE domain-containing protein 1 Human genes 0.000 description 1
- 241000269370 Xenopus <genus> Species 0.000 description 1
- 102100023585 Zinc finger CCHC-type and RNA-binding motif-containing protein 1 Human genes 0.000 description 1
- 101710180522 Zinc finger CCHC-type and RNA-binding motif-containing protein 1 Proteins 0.000 description 1
- 101710185494 Zinc finger protein Proteins 0.000 description 1
- 102100023555 Zinc finger protein 135 Human genes 0.000 description 1
- 102100021360 Zinc finger protein 260 Human genes 0.000 description 1
- 101710143808 Zinc finger protein 260 Proteins 0.000 description 1
- 102100028431 Zinc finger protein 292 Human genes 0.000 description 1
- 101710147076 Zinc finger protein 292 Proteins 0.000 description 1
- 102100021355 Zinc finger protein 609 Human genes 0.000 description 1
- 101710144201 Zinc finger protein 609 Proteins 0.000 description 1
- 102100023597 Zinc finger protein 816 Human genes 0.000 description 1
- 102100025884 Zinc finger protein GLIS2 Human genes 0.000 description 1
- 210000001015 abdomen Anatomy 0.000 description 1
- 230000003187 abdominal effect Effects 0.000 description 1
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 1
- 108020002494 acetyltransferase Proteins 0.000 description 1
- 102000005421 acetyltransferase Human genes 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 230000006978 adaptation Effects 0.000 description 1
- 210000001789 adipocyte Anatomy 0.000 description 1
- TWSWSIQAPQLDBP-UHFFFAOYSA-N adrenic acid Natural products CCCCCC=CCC=CCC=CCC=CCCCCCC(O)=O TWSWSIQAPQLDBP-UHFFFAOYSA-N 0.000 description 1
- 230000032683 aging Effects 0.000 description 1
- HQPCSDADVLFHHO-LTKCOYKYSA-N all-cis-8,11,14,17-icosatetraenoic acid Chemical compound CC\C=C/C\C=C/C\C=C/C\C=C/CCCCCCC(O)=O HQPCSDADVLFHHO-LTKCOYKYSA-N 0.000 description 1
- TWSWSIQAPQLDBP-DOFZRALJSA-N all-cis-docosa-7,10,13,16-tetraenoic acid Chemical compound CCCCC\C=C/C\C=C/C\C=C/C\C=C/CCCCCC(O)=O TWSWSIQAPQLDBP-DOFZRALJSA-N 0.000 description 1
- JIWBIWFOSCKQMA-LTKCOYKYSA-N all-cis-octadeca-6,9,12,15-tetraenoic acid Chemical compound CC\C=C/C\C=C/C\C=C/C\C=C/CCCCC(O)=O JIWBIWFOSCKQMA-LTKCOYKYSA-N 0.000 description 1
- SHGAZHPCJJPHSC-YCNIQYBTSA-N all-trans-retinoic acid Chemical compound OC(=O)\C=C(/C)\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C SHGAZHPCJJPHSC-YCNIQYBTSA-N 0.000 description 1
- 208000026935 allergic disease Diseases 0.000 description 1
- 230000007815 allergy Effects 0.000 description 1
- HMFHBZSHGGEWLO-UHFFFAOYSA-N alpha-D-Furanose-Ribose Natural products OCC1OC(O)C(O)C1O HMFHBZSHGGEWLO-UHFFFAOYSA-N 0.000 description 1
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 description 1
- AWUCVROLDVIAJX-UHFFFAOYSA-N alpha-glycerophosphate Natural products OCC(O)COP(O)(O)=O AWUCVROLDVIAJX-UHFFFAOYSA-N 0.000 description 1
- 244000096712 ambrette Species 0.000 description 1
- 235000002783 ambrette Nutrition 0.000 description 1
- 235000005550 amino acid supplement Nutrition 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 229940011037 anethole Drugs 0.000 description 1
- UIERETOOQGIECD-ARJAWSKDSA-N angelic acid group Chemical group C(\C(\C)=C/C)(=O)O UIERETOOQGIECD-ARJAWSKDSA-N 0.000 description 1
- 229940105969 annatto extract Drugs 0.000 description 1
- 229940121363 anti-inflammatory agent Drugs 0.000 description 1
- 239000002260 anti-inflammatory agent Substances 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 235000006708 antioxidants Nutrition 0.000 description 1
- 201000007201 aphasia Diseases 0.000 description 1
- 206010003119 arrhythmia Diseases 0.000 description 1
- 230000006793 arrhythmia Effects 0.000 description 1
- AQLMHYSWFMLWBS-UHFFFAOYSA-N arsenite(1-) Chemical compound O[As](O)[O-] AQLMHYSWFMLWBS-UHFFFAOYSA-N 0.000 description 1
- 239000001181 artemisia dracunculus Substances 0.000 description 1
- 206010003246 arthritis Diseases 0.000 description 1
- IAOZJIPTCAWIRG-QWRGUYRKSA-N aspartame Chemical compound OC(=O)C[C@H](N)C(=O)N[C@H](C(=O)OC)CC1=CC=CC=C1 IAOZJIPTCAWIRG-QWRGUYRKSA-N 0.000 description 1
- 239000000605 aspartame Substances 0.000 description 1
- 235000010357 aspartame Nutrition 0.000 description 1
- 229960003438 aspartame Drugs 0.000 description 1
- 235000013793 astaxanthin Nutrition 0.000 description 1
- 239000001168 astaxanthin Substances 0.000 description 1
- MQZIGYBFDRPAKN-ZWAPEEGVSA-N astaxanthin Chemical compound C([C@H](O)C(=O)C=1C)C(C)(C)C=1/C=C/C(/C)=C/C=C/C(/C)=C/C=C/C=C(C)C=CC=C(C)C=CC1=C(C)C(=O)[C@@H](O)CC1(C)C MQZIGYBFDRPAKN-ZWAPEEGVSA-N 0.000 description 1
- 229940022405 astaxanthin Drugs 0.000 description 1
- 208000006673 asthma Diseases 0.000 description 1
- 230000003143 atherosclerotic effect Effects 0.000 description 1
- 230000004900 autophagic degradation Effects 0.000 description 1
- IRERQBUNZFJFGC-UHFFFAOYSA-L azure blue Chemical compound [Na+].[Na+].[Na+].[Na+].[Na+].[Na+].[Na+].[Na+].[Al+3].[Al+3].[Al+3].[Al+3].[Al+3].[Al+3].[S-]S[S-].[O-][Si]([O-])([O-])[O-].[O-][Si]([O-])([O-])[O-].[O-][Si]([O-])([O-])[O-].[O-][Si]([O-])([O-])[O-].[O-][Si]([O-])([O-])[O-].[O-][Si]([O-])([O-])[O-] IRERQBUNZFJFGC-UHFFFAOYSA-L 0.000 description 1
- 230000001580 bacterial effect Effects 0.000 description 1
- 229940038481 bee pollen Drugs 0.000 description 1
- 235000012677 beetroot red Nutrition 0.000 description 1
- 230000003542 behavioural effect Effects 0.000 description 1
- 229940116226 behenic acid Drugs 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 235000021028 berry Nutrition 0.000 description 1
- MSWZFWKMSRAUBD-UHFFFAOYSA-N beta-D-galactosamine Natural products NC1C(O)OC(CO)C(O)C1O MSWZFWKMSRAUBD-UHFFFAOYSA-N 0.000 description 1
- WHGYBXFWUBPSRW-FOUAGVGXSA-N beta-cyclodextrin Chemical compound OC[C@H]([C@H]([C@@H]([C@H]1O)O)O[C@H]2O[C@@H]([C@@H](O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O3)[C@H](O)[C@H]2O)CO)O[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@@H]3O[C@@H]1CO WHGYBXFWUBPSRW-FOUAGVGXSA-N 0.000 description 1
- 230000008436 biogenesis Effects 0.000 description 1
- 230000004071 biological effect Effects 0.000 description 1
- QKSKPIVNLNLAAV-UHFFFAOYSA-N bis(2-chloroethyl) sulfide Chemical compound ClCCSCCCl QKSKPIVNLNLAAV-UHFFFAOYSA-N 0.000 description 1
- 235000019481 bixa orellana extract Nutrition 0.000 description 1
- 235000013614 black pepper Nutrition 0.000 description 1
- 210000000601 blood cell Anatomy 0.000 description 1
- 244000078885 bloodborne pathogen Species 0.000 description 1
- 210000001185 bone marrow Anatomy 0.000 description 1
- 235000021329 brown rice Nutrition 0.000 description 1
- 239000007853 buffer solution Substances 0.000 description 1
- PASOAYSIZAJOCT-UHFFFAOYSA-N butanoic acid Chemical compound CCCC(O)=O.CCCC(O)=O PASOAYSIZAJOCT-UHFFFAOYSA-N 0.000 description 1
- OBNCKNCVKJNDBV-UHFFFAOYSA-N butanoic acid ethyl ester Natural products CCCC(=O)OCC OBNCKNCVKJNDBV-UHFFFAOYSA-N 0.000 description 1
- 239000006227 byproduct Substances 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- FAPWYRCQGJNNSJ-UBKPKTQASA-L calcium D-pantothenic acid Chemical compound [Ca+2].OCC(C)(C)[C@@H](O)C(=O)NCCC([O-])=O.OCC(C)(C)[C@@H](O)C(=O)NCCC([O-])=O FAPWYRCQGJNNSJ-UBKPKTQASA-L 0.000 description 1
- 239000001639 calcium acetate Substances 0.000 description 1
- 235000011092 calcium acetate Nutrition 0.000 description 1
- 229960005147 calcium acetate Drugs 0.000 description 1
- 229960003563 calcium carbonate Drugs 0.000 description 1
- 229960002079 calcium pantothenate Drugs 0.000 description 1
- 229910000389 calcium phosphate Inorganic materials 0.000 description 1
- 235000011010 calcium phosphates Nutrition 0.000 description 1
- 235000012682 canthaxanthin Nutrition 0.000 description 1
- 239000001659 canthaxanthin Substances 0.000 description 1
- 229940008033 canthaxanthin Drugs 0.000 description 1
- 239000001325 capsicum annuum l. var. longum oleoresin Substances 0.000 description 1
- 239000001390 capsicum minimum Substances 0.000 description 1
- 235000013736 caramel Nutrition 0.000 description 1
- 125000004432 carbon atom Chemical group C* 0.000 description 1
- 239000001569 carbon dioxide Substances 0.000 description 1
- 229910002092 carbon dioxide Inorganic materials 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 1
- 150000001735 carboxylic acids Chemical class 0.000 description 1
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 1
- 235000005300 cardamomo Nutrition 0.000 description 1
- 235000012730 carminic acid Nutrition 0.000 description 1
- 229960004203 carnitine Drugs 0.000 description 1
- 150000001745 carotenals Chemical class 0.000 description 1
- 235000015190 carrot juice Nutrition 0.000 description 1
- 235000013709 carrot oil Nutrition 0.000 description 1
- 239000005018 casein Substances 0.000 description 1
- BECPQYXYKAMYBN-UHFFFAOYSA-N casein, tech. Chemical compound NCCCCC(C(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(CC(C)C)N=C(O)C(CCC(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(C(C)O)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(COP(O)(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(N)CC1=CC=CC=C1 BECPQYXYKAMYBN-UHFFFAOYSA-N 0.000 description 1
- 235000021240 caseins Nutrition 0.000 description 1
- 235000011472 cat’s claw Nutrition 0.000 description 1
- 230000021164 cell adhesion Effects 0.000 description 1
- 210000000170 cell membrane Anatomy 0.000 description 1
- 229940081734 cellulose acetate phthalate Drugs 0.000 description 1
- 239000000460 chlorine Substances 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- 229910052804 chromium Inorganic materials 0.000 description 1
- 239000011651 chromium Substances 0.000 description 1
- QSWDMMVNRMROPK-UHFFFAOYSA-K chromium(3+) trichloride Chemical compound [Cl-].[Cl-].[Cl-].[Cr+3] QSWDMMVNRMROPK-UHFFFAOYSA-K 0.000 description 1
- IJBFSOLHRKELLR-FPLPWBNLSA-N cis-5-dodecenoic acid Chemical compound CCCCCC\C=C/CCCC(O)=O IJBFSOLHRKELLR-FPLPWBNLSA-N 0.000 description 1
- GWHCXVQVJPWHRF-UHFFFAOYSA-N cis-tetracosenoic acid Natural products CCCCCCCCC=CCCCCCCCCCCCCCC(O)=O GWHCXVQVJPWHRF-UHFFFAOYSA-N 0.000 description 1
- DQLATGHUWYMOKM-UHFFFAOYSA-L cisplatin Chemical compound N[Pt](N)(Cl)Cl DQLATGHUWYMOKM-UHFFFAOYSA-L 0.000 description 1
- 229960004316 cisplatin Drugs 0.000 description 1
- 229940043350 citral Drugs 0.000 description 1
- WTEVQBCEXWBHNA-YFHOEESVSA-N citral B Natural products CC(C)=CCC\C(C)=C/C=O WTEVQBCEXWBHNA-YFHOEESVSA-N 0.000 description 1
- 235000012716 cod liver oil Nutrition 0.000 description 1
- 239000003026 cod liver oil Substances 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 238000004040 coloring Methods 0.000 description 1
- 208000004209 confusion Diseases 0.000 description 1
- 229940108924 conjugated linoleic acid Drugs 0.000 description 1
- 235000020940 control diet Nutrition 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 235000019545 cooked cereal Nutrition 0.000 description 1
- 108010039581 copine III Proteins 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 229940108925 copper gluconate Drugs 0.000 description 1
- 229910000365 copper sulfate Inorganic materials 0.000 description 1
- ARUVKPQLZAKDPS-UHFFFAOYSA-L copper(II) sulfate Chemical compound [Cu+2].[O-][S+2]([O-])([O-])[O-] ARUVKPQLZAKDPS-UHFFFAOYSA-L 0.000 description 1
- 235000013712 corn endosperm oil Nutrition 0.000 description 1
- 239000008120 corn starch Substances 0.000 description 1
- 235000012343 cottonseed oil Nutrition 0.000 description 1
- 229960001681 croscarmellose sodium Drugs 0.000 description 1
- 229960000913 crospovidone Drugs 0.000 description 1
- 235000010947 crosslinked sodium carboxy methyl cellulose Nutrition 0.000 description 1
- 239000001812 curcuma zedoaria berg. rosc. Substances 0.000 description 1
- 235000012754 curcumin Nutrition 0.000 description 1
- 239000004148 curcumin Substances 0.000 description 1
- 229940109262 curcumin Drugs 0.000 description 1
- 206010061428 decreased appetite Diseases 0.000 description 1
- 230000002950 deficient Effects 0.000 description 1
- 239000007857 degradation product Substances 0.000 description 1
- 108010040223 dehydrodolichyl diphosphate synthetase Proteins 0.000 description 1
- 230000003111 delayed effect Effects 0.000 description 1
- 230000001627 detrimental effect Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- 239000008121 dextrose Substances 0.000 description 1
- NEFBYIFKOOEVPA-UHFFFAOYSA-K dicalcium phosphate Chemical compound [Ca+2].[Ca+2].[O-]P([O-])([O-])=O NEFBYIFKOOEVPA-UHFFFAOYSA-K 0.000 description 1
- 229940038472 dicalcium phosphate Drugs 0.000 description 1
- 229910000390 dicalcium phosphate Inorganic materials 0.000 description 1
- 230000000378 dietary effect Effects 0.000 description 1
- VFLDPWHFBUODDF-UHFFFAOYSA-N diferuloylmethane Natural products C1=C(O)C(OC)=CC(C=CC(=O)CC(=O)C=CC=2C=C(OC)C(O)=CC=2)=C1 VFLDPWHFBUODDF-UHFFFAOYSA-N 0.000 description 1
- ZPWVASYFFYYZEW-UHFFFAOYSA-L dipotassium hydrogen phosphate Chemical compound [K+].[K+].OP([O-])([O-])=O ZPWVASYFFYYZEW-UHFFFAOYSA-L 0.000 description 1
- BVTBRVFYZUCAKH-UHFFFAOYSA-L disodium selenite Chemical compound [Na+].[Na+].[O-][Se]([O-])=O BVTBRVFYZUCAKH-UHFFFAOYSA-L 0.000 description 1
- 206010013395 disorientation Diseases 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- MBMBGCFOFBJSGT-SFGLVEFQSA-N docosa-4,7,10,13,16,19-hexaenoic acid Chemical compound CC\C=C\C\C=C\C\C=C\C\C=C\C\C=C\C\C=C\CCC(O)=O MBMBGCFOFBJSGT-SFGLVEFQSA-N 0.000 description 1
- KFEVDPWXEVUUMW-UHFFFAOYSA-N docosanoic acid Natural products CCCCCCCCCCCCCCCCCCCCCC(=O)OCCC1=CC=C(O)C=C1 KFEVDPWXEVUUMW-UHFFFAOYSA-N 0.000 description 1
- 235000013760 dried algae meal Nutrition 0.000 description 1
- 239000001921 dulse Substances 0.000 description 1
- 208000009356 dyskeratosis congenita Diseases 0.000 description 1
- 108010037434 early endosome antigen 1 Proteins 0.000 description 1
- 230000001819 effect on gene Effects 0.000 description 1
- 235000013345 egg yolk Nutrition 0.000 description 1
- 210000002969 egg yolk Anatomy 0.000 description 1
- 235000013601 eggs Nutrition 0.000 description 1
- BITHHVVYSMSWAG-UHFFFAOYSA-N eicosenoic acid Natural products CCCCCCCCC=CCCCCCCCCCC(O)=O BITHHVVYSMSWAG-UHFFFAOYSA-N 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 238000006911 enzymatic reaction Methods 0.000 description 1
- 108010008594 epithelial membrane protein-1 Proteins 0.000 description 1
- 230000032050 esterification Effects 0.000 description 1
- 238000005886 esterification reaction Methods 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 229940073505 ethyl vanillin Drugs 0.000 description 1
- 235000008995 european elder Nutrition 0.000 description 1
- 235000008524 evening primrose extract Nutrition 0.000 description 1
- 239000010475 evening primrose oil Substances 0.000 description 1
- 229940089020 evening primrose oil Drugs 0.000 description 1
- 239000000284 extract Substances 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 208000009866 extrahepatic cholestasis Diseases 0.000 description 1
- 150000002190 fatty acyls Chemical group 0.000 description 1
- 235000013924 ferrous gluconate Nutrition 0.000 description 1
- 239000004222 ferrous gluconate Substances 0.000 description 1
- 229960001645 ferrous gluconate Drugs 0.000 description 1
- 235000013925 ferrous lactate Nutrition 0.000 description 1
- 239000004225 ferrous lactate Substances 0.000 description 1
- 229940037907 ferrous lactate Drugs 0.000 description 1
- 229940114124 ferulic acid Drugs 0.000 description 1
- KSEBMYQBYZTDHS-HWKANZROSA-N ferulic acid Chemical compound COC1=CC(\C=C\C(O)=O)=CC=C1O KSEBMYQBYZTDHS-HWKANZROSA-N 0.000 description 1
- KSEBMYQBYZTDHS-UHFFFAOYSA-N ferulic acid Natural products COC1=CC(C=CC(O)=O)=CC=C1O KSEBMYQBYZTDHS-UHFFFAOYSA-N 0.000 description 1
- 235000001785 ferulic acid Nutrition 0.000 description 1
- 108010072251 fetal Alzheimer antigen Proteins 0.000 description 1
- 239000012091 fetal bovine serum Substances 0.000 description 1
- 210000003754 fetus Anatomy 0.000 description 1
- 235000004426 flaxseed Nutrition 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 150000002224 folic acids Chemical class 0.000 description 1
- 230000037406 food intake Effects 0.000 description 1
- 235000003599 food sweetener Nutrition 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 235000015203 fruit juice Nutrition 0.000 description 1
- 235000011389 fruit/vegetable juice Nutrition 0.000 description 1
- 230000002538 fungal effect Effects 0.000 description 1
- 108020001507 fusion proteins Proteins 0.000 description 1
- 102000037865 fusion proteins Human genes 0.000 description 1
- WIGCFUFOHFEKBI-UHFFFAOYSA-N gamma-tocopherol Natural products CC(C)CCCC(C)CCCC(C)CCCC1CCC2C(C)C(O)C(C)C(C)C2O1 WIGCFUFOHFEKBI-UHFFFAOYSA-N 0.000 description 1
- 235000004611 garlic Nutrition 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 239000007903 gelatin capsule Substances 0.000 description 1
- 239000007897 gelcap Substances 0.000 description 1
- HIGQPQRQIQDZMP-UHFFFAOYSA-N geranil acetate Natural products CC(C)=CCCC(C)=CCOC(C)=O HIGQPQRQIQDZMP-UHFFFAOYSA-N 0.000 description 1
- 229940113087 geraniol Drugs 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 229960001031 glucose Drugs 0.000 description 1
- 235000001727 glucose Nutrition 0.000 description 1
- 150000002327 glycerophospholipids Chemical class 0.000 description 1
- 210000000224 granular leucocyte Anatomy 0.000 description 1
- 210000003714 granulocyte Anatomy 0.000 description 1
- 235000002532 grape seed extract Nutrition 0.000 description 1
- 229940087603 grape seed extract Drugs 0.000 description 1
- 229940107702 grapefruit seed extract Drugs 0.000 description 1
- 235000009569 green tea Nutrition 0.000 description 1
- UYTPUPDQBNUYGX-UHFFFAOYSA-N guanine Chemical class O=C1NC(N)=NC2=C1N=CN2 UYTPUPDQBNUYGX-UHFFFAOYSA-N 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 231100000206 health hazard Toxicity 0.000 description 1
- 210000005003 heart tissue Anatomy 0.000 description 1
- 235000008216 herbs Nutrition 0.000 description 1
- 235000001050 hortel pimenta Nutrition 0.000 description 1
- 150000002430 hydrocarbons Chemical group 0.000 description 1
- 230000003301 hydrolyzing effect Effects 0.000 description 1
- GFAZHVHNLUBROE-UHFFFAOYSA-N hydroxymethyl propionaldehyde Natural products CCC(=O)CO GFAZHVHNLUBROE-UHFFFAOYSA-N 0.000 description 1
- 210000003016 hypothalamus Anatomy 0.000 description 1
- XUJWOMMOEOHPFP-UHFFFAOYSA-N icosa-8,11-dienoic acid Chemical compound CCCCCCCCC=CCC=CCCCCCCC(O)=O XUJWOMMOEOHPFP-UHFFFAOYSA-N 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 208000000509 infertility Diseases 0.000 description 1
- 230000036512 infertility Effects 0.000 description 1
- 231100000535 infertility Toxicity 0.000 description 1
- 208000027866 inflammatory disease Diseases 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 238000003780 insertion Methods 0.000 description 1
- 230000037431 insertion Effects 0.000 description 1
- 229940079322 interferon Drugs 0.000 description 1
- 238000007912 intraperitoneal administration Methods 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- 235000013980 iron oxide Nutrition 0.000 description 1
- YAQXGBBDJYBXKL-UHFFFAOYSA-N iron(2+);1,10-phenanthroline;dicyanide Chemical compound [Fe+2].N#[C-].N#[C-].C1=CN=C2C3=NC=CC=C3C=CC2=C1.C1=CN=C2C3=NC=CC=C3C=CC2=C1 YAQXGBBDJYBXKL-UHFFFAOYSA-N 0.000 description 1
- WBJZTOZJJYAKHQ-UHFFFAOYSA-K iron(3+) phosphate Chemical compound [Fe+3].[O-]P([O-])([O-])=O WBJZTOZJJYAKHQ-UHFFFAOYSA-K 0.000 description 1
- 229910000399 iron(III) phosphate Inorganic materials 0.000 description 1
- VRIVJOXICYMTAG-IYEMJOQQSA-L iron(ii) gluconate Chemical compound [Fe+2].OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C([O-])=O.OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C([O-])=O VRIVJOXICYMTAG-IYEMJOQQSA-L 0.000 description 1
- 229930013032 isoflavonoid Natural products 0.000 description 1
- 150000003817 isoflavonoid derivatives Chemical class 0.000 description 1
- 235000012891 isoflavonoids Nutrition 0.000 description 1
- 150000002576 ketones Chemical class 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 239000001102 lavandula vera Substances 0.000 description 1
- 235000018219 lavender Nutrition 0.000 description 1
- 239000002523 lectin Substances 0.000 description 1
- 231100000518 lethal Toxicity 0.000 description 1
- 230000001665 lethal effect Effects 0.000 description 1
- 150000002617 leukotrienes Chemical class 0.000 description 1
- 235000001510 limonene Nutrition 0.000 description 1
- 229940087305 limonene Drugs 0.000 description 1
- 229930007744 linalool Natural products 0.000 description 1
- 239000000865 liniment Substances 0.000 description 1
- OYHQOLUKZRVURQ-IXWMQOLASA-N linoleic acid Natural products CCCCC\C=C/C\C=C\CCCCCCCC(O)=O OYHQOLUKZRVURQ-IXWMQOLASA-N 0.000 description 1
- ZMKDEQUXYDZSNN-UHFFFAOYSA-N linolelaidic acid Natural products CCCCCCCCC=CCC=CCCCCC(O)=O ZMKDEQUXYDZSNN-UHFFFAOYSA-N 0.000 description 1
- 235000021388 linseed oil Nutrition 0.000 description 1
- 239000000944 linseed oil Substances 0.000 description 1
- 235000019626 lipase activity Nutrition 0.000 description 1
- 230000007787 long-term memory Effects 0.000 description 1
- 235000012661 lycopene Nutrition 0.000 description 1
- 239000001751 lycopene Substances 0.000 description 1
- 229960004999 lycopene Drugs 0.000 description 1
- OAIJSZIZWZSQBC-GYZMGTAESA-N lycopene Chemical compound CC(C)=CCC\C(C)=C\C=C\C(\C)=C\C=C\C(\C)=C\C=C\C=C(/C)\C=C\C=C(/C)\C=C\C=C(/C)CCC=C(C)C OAIJSZIZWZSQBC-GYZMGTAESA-N 0.000 description 1
- 210000004698 lymphocyte Anatomy 0.000 description 1
- 210000003712 lysosome Anatomy 0.000 description 1
- 230000001868 lysosomic effect Effects 0.000 description 1
- 239000001115 mace Substances 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 239000000395 magnesium oxide Substances 0.000 description 1
- CPLXHLVBOLITMK-UHFFFAOYSA-N magnesium oxide Inorganic materials [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 description 1
- 229940057948 magnesium stearate Drugs 0.000 description 1
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 1
- 235000019341 magnesium sulphate Nutrition 0.000 description 1
- AXZKOIWUVFPNLO-UHFFFAOYSA-N magnesium;oxygen(2-) Chemical class [O-2].[Mg+2] AXZKOIWUVFPNLO-UHFFFAOYSA-N 0.000 description 1
- WPBNNNQJVZRUHP-UHFFFAOYSA-L manganese(2+);methyl n-[[2-(methoxycarbonylcarbamothioylamino)phenyl]carbamothioyl]carbamate;n-[2-(sulfidocarbothioylamino)ethyl]carbamodithioate Chemical compound [Mn+2].[S-]C(=S)NCCNC([S-])=S.COC(=O)NC(=S)NC1=CC=CC=C1NC(=S)NC(=O)OC WPBNNNQJVZRUHP-UHFFFAOYSA-L 0.000 description 1
- 239000000594 mannitol Substances 0.000 description 1
- 235000010355 mannitol Nutrition 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 210000004379 membrane Anatomy 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 230000006984 memory degeneration Effects 0.000 description 1
- 208000023060 memory loss Diseases 0.000 description 1
- 239000001771 mentha piperita Substances 0.000 description 1
- 239000001220 mentha spicata Substances 0.000 description 1
- 230000002503 metabolic effect Effects 0.000 description 1
- GBMDVOWEEQVZKZ-UHFFFAOYSA-N methanol;hydrate Chemical compound O.OC GBMDVOWEEQVZKZ-UHFFFAOYSA-N 0.000 description 1
- UZKWTJUDCOPSNM-UHFFFAOYSA-N methoxybenzene Substances CCCCOC=C UZKWTJUDCOPSNM-UHFFFAOYSA-N 0.000 description 1
- 229940102398 methyl anthranilate Drugs 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 235000010981 methylcellulose Nutrition 0.000 description 1
- 201000003694 methylmalonic acidemia Diseases 0.000 description 1
- 239000008108 microcrystalline cellulose Substances 0.000 description 1
- 229940016286 microcrystalline cellulose Drugs 0.000 description 1
- 235000019813 microcrystalline cellulose Nutrition 0.000 description 1
- 210000004688 microtubule Anatomy 0.000 description 1
- 235000021243 milk fat Nutrition 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 229910052750 molybdenum Inorganic materials 0.000 description 1
- 239000011733 molybdenum Substances 0.000 description 1
- 201000006894 monocytic leukemia Diseases 0.000 description 1
- 210000005087 mononuclear cell Anatomy 0.000 description 1
- 208000015001 muscle soreness Diseases 0.000 description 1
- 235000010460 mustard Nutrition 0.000 description 1
- 210000004165 myocardium Anatomy 0.000 description 1
- YWWVWXASSLXJHU-WAYWQWQTSA-N myristoleic acid Chemical compound CCCC\C=C/CCCCCCCC(O)=O YWWVWXASSLXJHU-WAYWQWQTSA-N 0.000 description 1
- 239000000974 natural food coloring agent Substances 0.000 description 1
- 230000003880 negative regulation of appetite Effects 0.000 description 1
- 210000005044 neurofilament Anatomy 0.000 description 1
- 239000001702 nutmeg Substances 0.000 description 1
- 235000016709 nutrition Nutrition 0.000 description 1
- BNDAMTLLUULVLF-UHFFFAOYSA-N octa-3,6-dien-1-ol Chemical compound CC=CCC=CCCO BNDAMTLLUULVLF-UHFFFAOYSA-N 0.000 description 1
- XVEIGUQEXNENQF-HPFCUAHCSA-N octadeca-6,9,12-trienoic acid;(6z,9z,12z)-octadeca-6,9,12-trienoic acid Chemical compound CCCCCC=CCC=CCC=CCCCCC(O)=O.CCCCC\C=C/C\C=C/C\C=C/CCCCC(O)=O XVEIGUQEXNENQF-HPFCUAHCSA-N 0.000 description 1
- VJZWIFWPGRIJSN-NBTZWHCOSA-N octadeca-9,12-dienoic acid;(9z,12z)-octadeca-9,12-dienoic acid Chemical compound CCCCCC=CCC=CCCCCCCCC(O)=O.CCCCC\C=C/C\C=C/CCCCCCCC(O)=O VJZWIFWPGRIJSN-NBTZWHCOSA-N 0.000 description 1
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 description 1
- 239000008601 oleoresin Substances 0.000 description 1
- 235000021354 omega 7 monounsaturated fatty acids Nutrition 0.000 description 1
- 235000020665 omega-6 fatty acid Nutrition 0.000 description 1
- 108010057248 oncogene proteins v-ets Proteins 0.000 description 1
- 210000003463 organelle Anatomy 0.000 description 1
- 150000002894 organic compounds Chemical class 0.000 description 1
- 108010024073 ornithine transporter Proteins 0.000 description 1
- 201000008482 osteoarthritis Diseases 0.000 description 1
- 210000002997 osteoclast Anatomy 0.000 description 1
- 235000019161 pantothenic acid Nutrition 0.000 description 1
- 229940055726 pantothenic acid Drugs 0.000 description 1
- 239000011713 pantothenic acid Substances 0.000 description 1
- 235000012658 paprika extract Nutrition 0.000 description 1
- DYUUPIKEWLHQGQ-SDXBLLFJSA-N paprika oleoresin Chemical compound C(\[C@]12[C@@](O1)(C)C[C@@H](O)CC2(C)C)=C/C(/C)=C/C=C/C(/C)=C/C=C/C=C(C)C=CC=C(C)C=C[C@H]1C(C)=C[C@H](O)CC1(C)C DYUUPIKEWLHQGQ-SDXBLLFJSA-N 0.000 description 1
- QNGNSVIICDLXHT-UHFFFAOYSA-N para-ethylbenzaldehyde Natural products CCC1=CC=C(C=O)C=C1 QNGNSVIICDLXHT-UHFFFAOYSA-N 0.000 description 1
- RUVINXPYWBROJD-UHFFFAOYSA-N para-methoxyphenyl Natural products COC1=CC=C(C=CC)C=C1 RUVINXPYWBROJD-UHFFFAOYSA-N 0.000 description 1
- 230000007170 pathology Effects 0.000 description 1
- 235000011197 perejil Nutrition 0.000 description 1
- 210000003200 peritoneal cavity Anatomy 0.000 description 1
- 210000002824 peroxisome Anatomy 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 150000008104 phosphatidylethanolamines Chemical class 0.000 description 1
- 239000011574 phosphorus Substances 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- MBWXNTAXLNYFJB-NKFFZRIASA-N phylloquinone Chemical compound C1=CC=C2C(=O)C(C/C=C(C)/CCC[C@H](C)CCC[C@H](C)CCCC(C)C)=C(C)C(=O)C2=C1 MBWXNTAXLNYFJB-NKFFZRIASA-N 0.000 description 1
- 235000019175 phylloquinone Nutrition 0.000 description 1
- 239000011772 phylloquinone Substances 0.000 description 1
- 229960001898 phytomenadione Drugs 0.000 description 1
- 229940068065 phytosterols Drugs 0.000 description 1
- 239000001909 pimpinella anisum Substances 0.000 description 1
- 229940081310 piperonal Drugs 0.000 description 1
- 210000004180 plasmocyte Anatomy 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 108010026735 platelet protein P47 Proteins 0.000 description 1
- 229920001197 polyacetylene Polymers 0.000 description 1
- 150000008442 polyphenolic compounds Chemical class 0.000 description 1
- 235000013824 polyphenols Nutrition 0.000 description 1
- 235000013809 polyvinylpolypyrrolidone Nutrition 0.000 description 1
- 229920000523 polyvinylpolypyrrolidone Polymers 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- SCVFZCLFOSHCOH-UHFFFAOYSA-M potassium acetate Substances [K+].CC([O-])=O SCVFZCLFOSHCOH-UHFFFAOYSA-M 0.000 description 1
- 235000011056 potassium acetate Nutrition 0.000 description 1
- 239000001103 potassium chloride Substances 0.000 description 1
- 235000011164 potassium chloride Nutrition 0.000 description 1
- 229940069328 povidone Drugs 0.000 description 1
- 102100038155 pre-mRNA 3' end processing protein WDR33 Human genes 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 230000000750 progressive effect Effects 0.000 description 1
- 229940069949 propolis Drugs 0.000 description 1
- 108020003519 protein disulfide isomerase Proteins 0.000 description 1
- 108060006633 protein kinase Proteins 0.000 description 1
- 238000001243 protein synthesis Methods 0.000 description 1
- 235000011962 puddings Nutrition 0.000 description 1
- 235000008160 pyridoxine Nutrition 0.000 description 1
- 239000011677 pyridoxine Substances 0.000 description 1
- ZUFQODAHGAHPFQ-UHFFFAOYSA-N pyridoxine hydrochloride Chemical compound Cl.CC1=NC=C(CO)C(CO)=C1O ZUFQODAHGAHPFQ-UHFFFAOYSA-N 0.000 description 1
- 229960004172 pyridoxine hydrochloride Drugs 0.000 description 1
- 235000019171 pyridoxine hydrochloride Nutrition 0.000 description 1
- 239000011764 pyridoxine hydrochloride Substances 0.000 description 1
- 235000005875 quercetin Nutrition 0.000 description 1
- 150000004053 quinones Chemical class 0.000 description 1
- 230000006798 recombination Effects 0.000 description 1
- 238000005215 recombination Methods 0.000 description 1
- 230000007115 recruitment Effects 0.000 description 1
- 235000013526 red clover Nutrition 0.000 description 1
- 230000008929 regeneration Effects 0.000 description 1
- 238000011069 regeneration method Methods 0.000 description 1
- 230000018406 regulation of metabolic process Effects 0.000 description 1
- 230000008439 repair process Effects 0.000 description 1
- 239000013557 residual solvent Substances 0.000 description 1
- 229930002330 retinoic acid Natural products 0.000 description 1
- 206010039073 rheumatoid arthritis Diseases 0.000 description 1
- 108010004650 rho GTPase-activating protein Proteins 0.000 description 1
- 235000009566 rice Nutrition 0.000 description 1
- WBHHMMIMDMUBKC-XLNAKTSKSA-N ricinelaidic acid Chemical compound CCCCCC[C@@H](O)C\C=C\CCCCCCCC(O)=O WBHHMMIMDMUBKC-XLNAKTSKSA-N 0.000 description 1
- 229960003656 ricinoleic acid Drugs 0.000 description 1
- FEUQNCSVHBHROZ-UHFFFAOYSA-N ricinoleic acid Natural products CCCCCCC(O[Si](C)(C)C)CC=CCCCCCCCC(=O)OC FEUQNCSVHBHROZ-UHFFFAOYSA-N 0.000 description 1
- 235000015639 rosmarinus officinalis Nutrition 0.000 description 1
- 229940109850 royal jelly Drugs 0.000 description 1
- CVHZOJJKTDOEJC-UHFFFAOYSA-N saccharin Chemical compound C1=CC=C2C(=O)NS(=O)(=O)C2=C1 CVHZOJJKTDOEJC-UHFFFAOYSA-N 0.000 description 1
- 235000019204 saccharin Nutrition 0.000 description 1
- 229940081974 saccharin Drugs 0.000 description 1
- 239000000901 saccharin and its Na,K and Ca salt Substances 0.000 description 1
- 235000014438 salad dressings Nutrition 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 235000019512 sardine Nutrition 0.000 description 1
- 235000015067 sauces Nutrition 0.000 description 1
- 230000003248 secreting effect Effects 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 230000021317 sensory perception Effects 0.000 description 1
- 230000035939 shock Effects 0.000 description 1
- 235000021391 short chain fatty acids Nutrition 0.000 description 1
- 150000004666 short chain fatty acids Chemical class 0.000 description 1
- 230000006403 short-term memory Effects 0.000 description 1
- 230000019491 signal transduction Effects 0.000 description 1
- 239000000741 silica gel Substances 0.000 description 1
- 229910002027 silica gel Inorganic materials 0.000 description 1
- 235000012239 silicon dioxide Nutrition 0.000 description 1
- 235000020183 skimmed milk Nutrition 0.000 description 1
- 210000000813 small intestine Anatomy 0.000 description 1
- 210000002460 smooth muscle Anatomy 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 239000011684 sodium molybdate Substances 0.000 description 1
- 235000015393 sodium molybdate Nutrition 0.000 description 1
- TVXXNOYZHKPKGW-UHFFFAOYSA-N sodium molybdate (anhydrous) Chemical compound [Na+].[Na+].[O-][Mo]([O-])(=O)=O TVXXNOYZHKPKGW-UHFFFAOYSA-N 0.000 description 1
- 239000011655 sodium selenate Substances 0.000 description 1
- 235000018716 sodium selenate Nutrition 0.000 description 1
- 229960001881 sodium selenate Drugs 0.000 description 1
- 239000011781 sodium selenite Substances 0.000 description 1
- 235000015921 sodium selenite Nutrition 0.000 description 1
- 229960001471 sodium selenite Drugs 0.000 description 1
- 229920003109 sodium starch glycolate Polymers 0.000 description 1
- 229940079832 sodium starch glycolate Drugs 0.000 description 1
- 239000008109 sodium starch glycolate Substances 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 235000010356 sorbitol Nutrition 0.000 description 1
- 235000003687 soy isoflavones Nutrition 0.000 description 1
- 235000012424 soybean oil Nutrition 0.000 description 1
- 239000003549 soybean oil Substances 0.000 description 1
- 229940082787 spirulina Drugs 0.000 description 1
- 230000037078 sports performance Effects 0.000 description 1
- 238000013222 sprague-dawley male rat Methods 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 229960004274 stearic acid Drugs 0.000 description 1
- 230000004936 stimulating effect Effects 0.000 description 1
- 230000000638 stimulation Effects 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 210000002784 stomach Anatomy 0.000 description 1
- 229960004793 sucrose Drugs 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- 229910052717 sulfur Inorganic materials 0.000 description 1
- 239000002600 sunflower oil Substances 0.000 description 1
- 230000009469 supplementation Effects 0.000 description 1
- 230000001629 suppression Effects 0.000 description 1
- 239000003765 sweetening agent Substances 0.000 description 1
- 210000002504 synaptic vesicle Anatomy 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 230000009885 systemic effect Effects 0.000 description 1
- GDBJCCBRRCYCEG-UHFFFAOYSA-N tariric acid Natural products CCCCCCCCCCCCC#CCCCC(O)=O GDBJCCBRRCYCEG-UHFFFAOYSA-N 0.000 description 1
- 150000003505 terpenes Chemical class 0.000 description 1
- 235000007586 terpenes Nutrition 0.000 description 1
- QZZGJDVWLFXDLK-UHFFFAOYSA-N tetracosanoic acid Chemical compound CCCCCCCCCCCCCCCCCCCCCCCC(O)=O QZZGJDVWLFXDLK-UHFFFAOYSA-N 0.000 description 1
- 238000009482 thermal adhesion granulation Methods 0.000 description 1
- 235000019157 thiamine Nutrition 0.000 description 1
- 239000011721 thiamine Substances 0.000 description 1
- UIERGBJEBXXIGO-UHFFFAOYSA-N thiamine mononitrate Chemical compound [O-][N+]([O-])=O.CC1=C(CCO)SC=[N+]1CC1=CN=C(C)N=C1N UIERGBJEBXXIGO-UHFFFAOYSA-N 0.000 description 1
- 150000003595 thromboxanes Chemical class 0.000 description 1
- QURCVMIEKCOAJU-UHFFFAOYSA-N trans-isoferulic acid Natural products COC1=CC=C(C=CC(O)=O)C=C1O QURCVMIEKCOAJU-UHFFFAOYSA-N 0.000 description 1
- ZCIHMQAPACOQHT-ZGMPDRQDSA-N trans-isorenieratene Natural products CC(=C/C=C/C=C(C)/C=C/C=C(C)/C=C/c1c(C)ccc(C)c1C)C=CC=C(/C)C=Cc2c(C)ccc(C)c2C ZCIHMQAPACOQHT-ZGMPDRQDSA-N 0.000 description 1
- UWHZIFQPPBDJPM-BQYQJAHWSA-N trans-vaccenic acid Chemical compound CCCCCC\C=C\CCCCCCCCCC(O)=O UWHZIFQPPBDJPM-BQYQJAHWSA-N 0.000 description 1
- 230000005029 transcription elongation Effects 0.000 description 1
- 230000002103 transcriptional effect Effects 0.000 description 1
- 230000014616 translation Effects 0.000 description 1
- 229960001727 tretinoin Drugs 0.000 description 1
- 229940078499 tricalcium phosphate Drugs 0.000 description 1
- 229910000391 tricalcium phosphate Inorganic materials 0.000 description 1
- 235000019731 tricalcium phosphate Nutrition 0.000 description 1
- 239000001226 triphosphate Substances 0.000 description 1
- 235000011178 triphosphate Nutrition 0.000 description 1
- UNXRWKVEANCORM-UHFFFAOYSA-N triphosphoric acid Chemical compound OP(O)(=O)OP(O)(=O)OP(O)(O)=O UNXRWKVEANCORM-UHFFFAOYSA-N 0.000 description 1
- 150000003648 triterpenes Chemical class 0.000 description 1
- 235000013976 turmeric Nutrition 0.000 description 1
- 235000013799 ultramarine blue Nutrition 0.000 description 1
- 241001529453 unidentified herpesvirus Species 0.000 description 1
- 210000004291 uterus Anatomy 0.000 description 1
- 235000012141 vanillin Nutrition 0.000 description 1
- MWOOGOJBHIARFG-UHFFFAOYSA-N vanillin Chemical compound COC1=CC(C=O)=CC=C1O MWOOGOJBHIARFG-UHFFFAOYSA-N 0.000 description 1
- FGQOOHJZONJGDT-UHFFFAOYSA-N vanillin Natural products COC1=CC(O)=CC(C=O)=C1 FGQOOHJZONJGDT-UHFFFAOYSA-N 0.000 description 1
- 235000015112 vegetable and seed oil Nutrition 0.000 description 1
- 235000015192 vegetable juice Nutrition 0.000 description 1
- 239000008158 vegetable oil Substances 0.000 description 1
- 230000003612 virological effect Effects 0.000 description 1
- 230000004393 visual impairment Effects 0.000 description 1
- 208000002670 vitamin B12 deficiency Diseases 0.000 description 1
- 235000019166 vitamin D Nutrition 0.000 description 1
- 239000011710 vitamin D Substances 0.000 description 1
- 150000003710 vitamin D derivatives Chemical class 0.000 description 1
- QYSXJUFSXHHAJI-YRZJJWOYSA-N vitamin D3 Chemical compound C1(/[C@@H]2CC[C@@H]([C@]2(CCC1)C)[C@H](C)CCCC(C)C)=C\C=C1\C[C@@H](O)CCC1=C QYSXJUFSXHHAJI-YRZJJWOYSA-N 0.000 description 1
- 239000011647 vitamin D3 Substances 0.000 description 1
- 235000019165 vitamin E Nutrition 0.000 description 1
- 229940046009 vitamin E Drugs 0.000 description 1
- 239000011709 vitamin E Substances 0.000 description 1
- 235000019168 vitamin K Nutrition 0.000 description 1
- 239000011712 vitamin K Substances 0.000 description 1
- 150000003721 vitamin K derivatives Chemical class 0.000 description 1
- 229940011671 vitamin b6 Drugs 0.000 description 1
- 229940046008 vitamin d Drugs 0.000 description 1
- 150000003722 vitamin derivatives Chemical class 0.000 description 1
- 229940046010 vitamin k Drugs 0.000 description 1
- 239000001717 vitis vinifera seed extract Substances 0.000 description 1
- 239000003039 volatile agent Substances 0.000 description 1
- 239000000341 volatile oil Substances 0.000 description 1
- 235000019509 white turmeric Nutrition 0.000 description 1
- NWONKYPBYAMBJT-UHFFFAOYSA-L zinc sulfate Chemical compound [Zn+2].[O-]S([O-])(=O)=O NWONKYPBYAMBJT-UHFFFAOYSA-L 0.000 description 1
- 229910000368 zinc sulfate Inorganic materials 0.000 description 1
- 229960001763 zinc sulfate Drugs 0.000 description 1
- 239000001841 zingiber officinale Substances 0.000 description 1
- DTOSIQBPPRVQHS-UHFFFAOYSA-N α-Linolenic acid Chemical compound CCC=CCC=CCC=CCCCCCCCC(O)=O DTOSIQBPPRVQHS-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/66—Phosphorus compounds
- A61K31/683—Diesters of a phosphorus acid with two hydroxy compounds, e.g. phosphatidylinositols
- A61K31/685—Diesters of a phosphorus acid with two hydroxy compounds, e.g. phosphatidylinositols one of the hydroxy compounds having nitrogen atoms, e.g. phosphatidylserine, lecithin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/02—Nutrients, e.g. vitamins, minerals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic Table
- C07F9/02—Phosphorus compounds
- C07F9/06—Phosphorus compounds without P—C bonds
- C07F9/08—Esters of oxyacids of phosphorus
- C07F9/09—Esters of phosphoric acids
- C07F9/10—Phosphatides, e.g. lecithin
Definitions
- the present invention relates to novel marine lipid compositions comprising combinations of omega-3 fatty acid rich functional phospholipids and omega-3 fatty acid rich triglycerides.
- food supplements, functional food, drugs and feed products comprising such compositions are provided along with methods of their use.
- Marine lipids such as omega-3 rich triglycerides and omega-3 rich phospholipids can be isolated from a number of different natural sources such as fish, crustaceans, plankton, seals, whales as well as algae using extraction technologies. In addition, they can be prepared industrially using chemical or bio-catalytical methods such as enzyme catalyzed transesterification of crude soy lecithin with fish oil fatty acids [1].
- omega-3 fatty acids are well known and the use as an anti-inflammatory agent has been described both for triglycerides and phospholipids [2-3].
- omega-3 fatty acids are famous for their anti-inflammatory properties, and it has been shown that omega-3 fatty acids alleviate the symptoms of a series of autoimmune, atherosclerotic and inflammatory diseases including inflammatory bowel diseases and rheumatoid arthritis [4-6]. Suppression of inflammation has been proposed as one of the strategies to slow down the progress of these diseases.
- this invention discloses the effect on marine lipid compositions on the concentration of markers of inflammation such as TNF- ⁇ and other cytokines such as interleukin-1 ⁇ and interleukin 6.
- AA arachidonic acid
- prostaglandins prostaglandins, thromboxanes and leukotrienes
- this invention discloses the reduction of AA level and the improvement in the EPA/AA ratio in different lipid pools in tissues such as in the phospholipids isolated from adipose tissue, heart, testicles, plasma, brain and liver.
- the invention provides a composition comprising a triglyceride and a phospholipids in a ratio ranging from 1:10 to 10:1; said phospholipids having the following structure: wherein R1 is OH or a fatty acid, R2 is OH or a fatty acid, and R3 is a mixture of H, choline, ethanolamine, inositol and serine, said phospholipid having at least 1% of DHA/EPA, said phospholipids have a concentration of OH in the range of 25-50%.
- the invention provides a marine lipid composition characterized by providing higher uptake of omega-3 fatty acids into plasma as compared to administration of purified triglycerides, phospholipids, or natural marine phospholipids.
- the invention provides a composition characterized by efficiently improving the AA/EPA ratio in plasma phospholipids as compared to administration of purified triglycerides, phospholipids, or natural marine phospholipids.
- the invention is a marine lipid composition characterized by efficiently increasing the concentration of omega-3 fatty acids in tissues as compared to administration of purified triglycerides, phospholipids, or natural marine phospholipids.
- the composition is characterized by reducing the concentration of biomarkers of inflammation as compared to administration of purified triglycerides, phospholipids, or natural marine phospholipids.
- the marine lipid composition is formulated into an animal feed, a food product, a food supplement and a drug.
- the present invention provides a composition comprising phospholipids having the following structure: wherein R1 is OH or a fatty acid, R2 is OH or a fatty acid, and R3 is a mixture of H, choline, ethanolamine, inositol and serine, said phospholipid having at least 1% of DHA/EPA at positions R1 and/or R2 and from about 20-50% of OH at positions R1 and/or R2.
- the composition is acylated in a range from about 55% to about 85%.
- the omega-3 fatty acids are selected from the group consisting of EPA, DHA, DPA and ⁇ -linolenic acid (ALA).
- the composition is substantially free of organic solvents and volatile organic compounds such as short chain fatty acids, short chain aldehydes and short chain ketones.
- the composition has at least 5% of a combination of EPA and DHA esterified.
- the composition has at least 10% of a combination of EPA and DHA esterified.
- the composition has at least 20% of a combination of EPA and DHA esterified.
- the composition has at least 30% of a combination of EPA and DHA esterified.
- said composition contains from about 5%, 10%, 20% and 30% EPA/DHA attached to position 1 and/ or position 2.
- the composition has a ratio of EPA/DHA ranging from 1:1 to 4:1. In some embodiments, the composition has a ratio of EPA/DHA ranging from 2:1 to 4:1. In some embodiments, the composition is acylated in a range from 60% to 80%. In some embodiments, the composition is acylated in a range from 50% to 75%.
- the composition further comprises a lipid carrier in a ratio of from 1:10 to 10:1 to said phospholipids.
- the lipid carrier and said phospholipids are in a ratio of from about 5:1 to 1:5.
- the composition comprises from about 20% to about 90% of said phospholipid composition and from about 10% to about 50% of said lipid carrier.
- the present invention is not limited to any particular lipid carrier.
- the lipid carrier is selected from the group consisting of a triglyceride, a diglyceride, an ethyl ester, and a methyl ester and combinations thereof.
- the composition provides higher uptake of omega-3 fatty acids into plasma as compared to natural marine phospholipids when administered to subjects. In some embodiments, the composition improves the AA/EPA ratio in plasma phospholipids when administered to subjects as compared to natural marine phospholipids. In some embodiments, the composition increases the concentration of omega-3 fatty acids in tissues when administered to subjects as compared to natural marine phospholipids. In some embodiments, the composition reduces the concentration of biomarkers of inflammation when administered to subjects as compared to natural marine phospholipids. In some embodiments, the present invention provides a food product comprising the foregoing compositions. In some embodiments, the present invention provides an animal feed comprising the foregoing compositions. In some embodiments, the present invention provides a food supplement comprising the foregoing compositions. In some embodiments, the present invention provides a pharmaceutical composition comprising the foregoing compositions.
- the present invention provides methods of preparing a bioavailable omega-3 fatty acid composition comprising: a) providing a purified phospholipid composition comprising omega-3 fatty acid residues and a purified triglyceride composition comprising omega-3 fatty acid residues; b) combining said phospholipid composition and said triglyceride composition to form a bioavailable omega-3 fatty acid composition.
- the bioavailable phospholipid composition is one of the compositions described above.
- the methods further comprise the step of encapsulating said bioavailable omega-3 fatty acid composition.
- the bioavailable omega-3 fatty acid composition has increased bioavailability as compared to purified triglycerides or phospholipids comprising omega-3 fatty acid residues.
- the methods further comprise the step of packaging the bioavailable omega-3 fatty acid composition for use in functional foods.
- the methods further comprise the step of assaying the bioavailable omega-3 fatty acid composition for bioavailability.
- the methods further comprise administering the bioavailable omega-3 fatty acid composition to a patient.
- the present invention provides a food product, animal feed, food supplement or pharmaceutical composition made by the foregoing process.
- the present invention provides methods for reducing symptoms of cognitive dysfunction in a child comprising administering an effective amount of a marine phospholipid composition, wherein said symptoms are selected from the group consisting of ability to complete task, ability to stay on task, ability to follow instructions, ability to complete assignments, psychomotor function, long term memory, short term memory, ability to make a decision, ability to follow through on decision, ability to self-sustain attention, ability to engage in conversations, sensitivity to surroundings, ability to plan, ability to carry out plan, ability to listen, interruptions in social situations, temper tantrums, level/frequency of frustration, level/frequency restlessness, frequency/level fidgeting, ability to exhibit delayed gratification, aggressiveness, demanding behavior/frequency of demanding behavior, sleep patterns, restive sleep, interrupted sleep, awakening behavior, disruptive behavior, ability to exhibit control in social situations, ability to extrapolate information and ability to integrate information.
- the child exhibits one or more symptoms of Attention Deficit Hyperactivity Disorder (ADHD), is suspected of having ADHD, or has been diagnosed with ADHD.
- ADHD Attention Deficit Hyperactivity Disorder
- autistic spectrum disorder is suspected of having autistic spectrum disorder, or has been diagnosed with autistic spectrum disorder.
- the present invention provides methods of increasing cognitive performance in an aging mammal comprising administering an effective amount of a marine phospholipid composition.
- the cognitive performance is selected from the group consisting of memory loss, forgetfulness, short-term memory loss, aphasia, disorientation, disinhibition, and behavioral changes.
- the mammal is a human.
- the mammal is a pet selected from the group consisting of cats and dogs.
- the mammal has symptoms of age-associated memory impairment or decline.
- the marine phospholipid composition comprises phospholipids having the following structure: wherein R1 is OH or a fatty acid, R2 is OH or a fatty acid, and R3 is a mixture of H, choline, ethanolamine, inositol and serine, said phospholipid having at least 1% of omega-3 fatty acid moieties at positions R1 and/or R2.
- the phospholipid composition comprises from about 20-50% of OH at positions R1 and/or R2.
- the phospholipid composition further comprises a lipid carrier.
- the phospholipid composition is prepared from natural marine phospholipids isolated from a marine organism. In some embodiments, the phospholipid composition is enzymatically prepared by reacting lecithin with DHA and EPA in the presence of an enzyme. In some embodiments, the lecithin is soybean or egg lecithin. In some embodiments, the omega-3 fatty acid moieties are selected from the group of EPA and DHA and combination thereof. In some embodiments, the effective amount of said phospholipid composition comprises from about 300 to about 1000 mg omega-3 fatty acids. In some embodiments, the phospholipid composition is administered orally. In some embodiments, the phospholipid composition is provided in a gel capsule or pill.
- the present invention provides methods of treating a subject by administration of a marine phospholipid composition comprising administering a marine phospholipid composition to said subject under conditions such that a desired condition is improved, wherein said conditions is selected from the group consisting of fertility, physical endurance, sports performance, muscle soreness, inflammation, auto-immune stimulation, metabolic syndrome, obesity and type II diabetes.
- the subject is a human.
- the subject is a companion animal.
- the present invention is not limited to any particular marine phospholipid composition.
- the marine phospholipid composition comprises phospholipids having the following structure: wherein R1 is OH or a fatty acid, R2 is OH or a fatty acid, and R3 is a mixture of H, choline, ethanolamine, inositol and serine, said phospholipid having at least 1% of omega-3 fatty acid moieties at positions R1 and/or R2.
- the phospholipid composition comprises from about 20-50% of OH at positions R1 and/or R2.
- the phospholipid composition is prepared from natural marine phospholipids isolated from a marine organism.
- the composition further comprises a lipid carrier.
- the phospholipid composition is enzymatically prepared by reacting lecithin with DHA and EPA in the presence of an enzyme.
- the-lecithin is soybean or egg lecithin.
- the omega-3 fatty acid moieties are selected from the group of EPA and DHA and combination thereof.
- the effective amount of said phospholipid composition comprises from about 300 to about 1000 mg omega-3 fatty acids.
- the phospholipid composition is administered orally.
- the phospholipid composition is provided in a gel capsule or pill.
- the human is a male.
- the present invention provides methods for prophylactically treating a subject by administration of a marine phospholipid composition
- a marine phospholipid composition comprising administering a marine phospholipid composition to a subject under conditions such that an undesirable condition is prevented, wherein said undesirable condition is selected from the group consisting of weight gain, infertility, obesity, metabolic syndrome, diabetes type II, mortality in subjects with a high risk of sudden cardiac death, and induction of sustained ventricular tachycardia.
- the subject is at risk for developing a condition selected from the group consisting of weight gain, obesity, metabolic syndrome, diabetes type II, mortality in subjects with a high risk of sudden cardiac death, and induction of sustained ventricular tachycardia.
- the subject is a human. In some embodiments, the subject is a companion animal.
- the present invention is not limited to any particular marine phospholipid composition.
- the marine phospholipid composition comprises phospholipids having the following structure: wherein R1 is OH or a fatty acid, R2 is OH or a fatty acid, and R3 is a mixture of H, choline, ethanolamine, inositol and serine, said phospholipid having at least 1% of omega-3 fatty acid moieties at positions R1 and/or R2.
- the phospholipid composition comprises from about 20-50% of OH at positions R1 and/or R2.
- the phospholipid composition is prepared from natural marine phospholipids isolated from a marine organism.
- the composition further comprises a lipid carrier.
- the phospholipid composition is enzymatically prepared by reacting lecithin with DHA and EPA in the presence of an enzyme.
- the lecithin is soybean or egg lecithin.
- the omega-3 fatty acid moieties are selected from the group of EPA and DHA and combination thereof.
- the effective amount of said phospholipid composition comprises from about 300 to about 1000 mg omega-3 fatty acids.
- the phospholipid composition is administered orally.
- the phospholipid composition is provided in a gel capsule or pill.
- FIG. 1 The total amount of EPA consumed during the four-week rat trial (mean ⁇ SE).
- FIG. 5 Schematic drawing of experimental set-up.
- FIG. 6 EPA levels in plasma as a function of hours after one bolus intake of a marine phospholipid composition.
- FIG. 7 EPA levels in plasma as a function of hours after one bolus intake of a marine phospholipid composition.
- FIG. 8 ARA levels in plasma as a function of hours after one bolus intake of a marine phospholipid composition.
- phospholipid refers to an organic compound having the following general structure: wherein R1 is a fatty acid residue or —OH, R2 is a fatty acid residue or —OH, and R3 is a —H or a nitrogen containing compound such as choline (HOCH 2 CH 2 N + (CH 3 ) 3 OH ⁇ ), ethanolamine (HOCH 2 CH 2 NH 2 ), inositol or serine. R1 and R2 cannot simultaneously be OH.
- R3 is an —OH
- the compound is a diacylglycerophosphate
- R3 is a nitrogen-containing compound
- the compound is a phosphatide such as lecithin, cephalin, phosphatidyl serine or plasmalogen.
- the R1 site is herein referred to as position 1 of the phospholipid
- the R2 site is herein referred to as position 2 of the phospholipid
- the R3 site is herein referred to as position 3 of the phospholipid.
- omega-3 fatty acid refers to polyunsaturated fatty acids that have the final double bond in the hydrocarbon chain between the third and fourth carbon atoms from the methyl end of the molecule.
- Non-limiting examples of omega-3 fatty acids include, 5,8,11,14,17-eicosapentaenoic acid (EPA), 4,7,10,13,16,19-docosahexanoic acid (DHA) and 7,10,13,16,19-docosapentanoic acid (DPA).
- bioavailability refers to the degree and rate at which a substance (as a drug) is absorbed into a living system or is made available at the site of physiological activity.
- the term “functional food” refers to a food product to which a biologically active supplement has been added.
- fish oil refers to any oil obtained from a marine source e.g. tuna oil, seal oil and algae oil.
- lipase refers to any enzyme capable of hydrolyzing fatty acid esters
- the term “food supplement” refers to a food product formulated as a dietary or nutritional supplement to be used as part of a diet.
- acylation means fatty acids attached to the phospholipid. 100% acylation means that there are no lyso- or glycerol-phospholipids.
- This invention discloses that the uptake/absorption of omega-3 fatty acids attached to phospholipids are dependent on the level of LPL and GPL.
- the level of LPL should be in the range of 15-45% and the level of GPL should be 0%.
- this invention discloses that the pure PC transesterified with EPA/DHA have a different effect on gene expression in the liver than 40% PC transesterified with EPA/DHA. It is disclosed that the two compositions regulated around 40 genes differently.
- the invention discloses that the EPA/DHA ratio is important. The treatment containing a EPA/DHA ratio of 2:1 regulated key enzymes involved in the inflammatory response (NF- ⁇ B) in a positive way, the treatment containing a EPA/DHA ratio of 1:1 did not.
- the present invention describes novel marine lipid compositions comprising an omega-3 containing phospholipid and a triacylglyceride (TG) in a ratio from about 1:10 to 10:1.
- the ratio is in the range of from about 3:1 to 1:3, more preferably the ratio is in the range of about 1:2 to 2:1.
- the TG is a fish oil such as tuna oil, herring oil, menhaden oil, cod liver oil and algae oil.
- this invention is not limited to omega-3 containing oils as other TG sources are contemplated such as vegetable oils.
- the phospholipids in the composition have the following structure: wherein R1 is OH or a fatty acid, R2 is OH or a fatty acid, and R3 is a mixture of H, choline, ethanolamine, inositol and serine. Attached to position 1 or position 2 are least 1% omega-3 fatty acids, preferably at least 5%, more preferably at least 10% omega-3 fatty acids, up to about 15%, 20%, 30%, 40%, 50%, or 60% omega-3 fatty acids.
- the omega-3 fatty acids can be EPA, DHA, DPA or C18:3 (n-3), most preferably the omega-3 fatty acids are EPA and DHA.
- the phospholipid composition preferably contains OH in position 1 or position 2 in a range of 25% to 50% in order to maximize absorption in-vivo.
- the present invention provides bioavailable and bioefficient omega-3 fatty acids.
- This invention shows that the novel marine lipid composition disclosed above enhances the uptake of the omega-3 fatty acid in vivo and incorporates omega-3 fatty acids more efficiently into tissues of adult rats than pure fish oil does.
- An embodiment of the invention is to use the marine lipid composition for efficient increase of omega-3 fatty acids in the liver, brain, adipose tissue, plasma, testicles and heart.
- this invention also discloses that the marine lipid compositions efficiently reduced the concentration of the pro-inflammatory precursor AA in total lipids and in phospholipids in tissues.
- the concentration of AA in the different lipid pools in the liver, brain, adipose tissue, plasma, testicles and heart can be more efficiently reduced than using fish oil.
- the composition can be used to improve the EPA/AA ratio, which is a bio-marker of silent inflammation.
- the invention also discloses that the incorporation of the omega-3 fatty acids into monocytes is also more efficient using the claimed marine lipid composition as opposed to the fish oil.
- Yet another embodiment of the invention is to use the marine lipid composition to reduce chronic and acute inflammation in humans and in animals. Acute inflammation is mediated by granulocytes or polymorphonuclear leukocytes, while chronic inflammation is mediated by mononuclear cells such as monocytes.
- Monocytes protect against blood-borne pathogens and moves quickly to sites of infection in the tissues, secreting large amounts of pro-inflammatory prostaglandins. Furthermore, low grade chronic inflammation may be the underlying cause of many life-style related diseases such as obesity, arthritis, diabetes type II, metabolic syndrome, Alzheimer's disease, osteoarthritis, inflammatory bowel disease, allergy and asthma [14].
- the marine lipid composition can be used to treat and prevent diseases linked to chronic inflammation.
- This invention discloses that the inflammatory response of monocytes harvested from animals in lower in animals treated with the marine lipid composition compared to fish oil.
- the concentrations of the pro-inflammatory cytokines such as interleukin-1 ⁇ , interleukin-6 as well as tumor necrosis factor ⁇ (TNF- ⁇ ) were reduced for the group fed the marine lipid composition compared to fish oil.
- These cytokines are important markers of real inflammation as for examples I1-1 ⁇ induces fever.
- I1-6 also induces fever in addition to being linked to the acute phase response.
- TNF- ⁇ is involved in systemic inflammation as well and is released by white blood cells in the case of damage. It has a range of different biological effects such as increasing insulin resistance, stimulating the acute phase response in the liver and affecting the hypothalamus causing appetite suppression and fever.
- This invention also discloses that the fatty acid composition of the brain and adipose tissue phospholipids changes after in take of omega-3 fatty acids for 30 days.
- a significant reduction of the arachidonic acids can be found in the phospholipids in the brain and adipose tissue for the rats given either the EPA- or DHA-rich PL diets (PL 1 and PL 2, respectively). This may affect the inflammatory response in this tissue and thereby have a great impact on cognitive diseases/conditions such as Parkinson's or and Alzheimer's where the inflammatory component is fundamental for the progression of the disease.
- This invention also discloses that the reduction of ARA is present also in the sn-2 position of the phospholipids in the brain.
- adipose tissue In adipose tissue, accumulation of EPA and DHA in both total lipids (table 3) and PLs (table 8) is substantial when omega-3 supplements were fed and negligible when the control diet was fed. The increase was more pronounced in total lipids, which mainly consists of triglycerides (99% of fat cell lipid content).
- This invention demonstrates that omega-3 phospholipids can increase the accumulation of EPA/DHA into adipose tissue. This is important as the adipose tissue can function as a reservoir for these fatty acids.
- Arachidonic acid concentration in total lipids was higher in omega-3 supplemented animals, showing probably an increase of lipoprotein lipase activity, in agreement with the ability of omega 3 in decreasing plasma TAGs concentration.
- arachidonic acid levels in adipose tissue PLs were significantly lower in omega-3 supplemented animals than the levels in controls. Peculiar enough, the PL-EPA diet was the most efficient in decreasing arachidonic acid.
- the invention discloses that the reduction of ARA is also observed in the sn-2 position of the phospholipids of the omega-3 supplemented animals. This is very important as the pro-inflammatory eicosanoids are produced from ARA, which are catalytically hydrolyzed from position 2 on the phospholipid by the action of phospholipase A2. The phospholipase A2 is released after stimuli at the cell wall, it then moves to the nuclear membrane where the hydrolysis of the phospholipid takes place. The reduction of ARA in position 2 on the phospholipids may affect the inflammatory response in this tissue, which may have practical application in different pathologies of the adipose tissue and in its physiological activity of accumulation and release of fatty acids.
- Fatty acid data from brain are well in line with the data from adipose tissue. Also in this tissue, we found a significant decrease of arachidonic acid in PLs, but surprisingly, only with PL-EPA and PL-DHA (table 7). On the other hand, DHA levels in both total lipids and PLs were not influenced by the omega-3 diets, while there was a small but significant increase in EPA levels. Lack of increase in DHA levels is likely to be attributable to the fact that the rats in this study were adults and pass the stage in development where they incorporate DHA in the brain (mainly PE). On the other hand, EPA being present at low concentration has more margin to increase.
- liver as expected, we found for all omega-3 groups a significant increase of EPA and DHA and decrease of arachidonic acid. No great differences were expected between total lipids and PLs because about 80% of liver total lipids are PLs (table 4, 9 and 14).
- Heart total lipids and PLs showed a strong increase of EPA and DHA with a concomitant decrease of arachidonic acid when omega-3 supplements were fed.
- the strong decrease in the omega-6/omega-3 ratio in heart lipids is important considering the possible impact on the anti-inflammatory potential.
- Observed change in heart tissue fatty acids increase of fatty acids with 6 or 5 double bonds
- This change was most striking in the PL-DHA group where the increase of DHA was significantly higher than the increase in the TG-oil and PL-EPA groups.
- the fluidity of myocardium cell membrane seems to play an important role in controlling arrhythmia. Ventricular arrhythmia, is one of the main causes of sudden cardiac death. Furthermore, atrial fibrillation is another pathological state with a high incidence and important health consequences.
- Testicular long chain PUFAs are of special interest because there is a high rate of production of prostaglandins from the omega-6 PUFA (arachidonic acid mainly) into the semen or seminal fluid.
- High rate of prostaglandin production does not indicate an active inflammatory process but a stimulus for the uterus smooth muscle to favor male fertility.
- An omega-3 induced decrease of arachidonic acid as observed in other tissue could be detrimental to the male fertility if it occurred also in testis.
- testicular tissue has also a high level of DPA (22:5 omega-6), which may serve as a reservoir for arachidonic acid.
- Arachidonic acid could be formed according to the need, through the retroconversion mechanism in the peroxisomes.
- Another embodiment of the invention is to formulate the marine lipid compositions into a feed product for the purpose of reducing low-grade chronic inflammation in animals. It can also be formulated into a food product and given to humans for the same purpose. Furthermore, it can be formulated as a functional food product, as a drug or as food supplement.
- the compositions of this invention are contained in acceptable excipients and/or carriers for oral consumption.
- the actual form of the carrier, and thus, the compositions itself, is not critical.
- the carrier may be a liquid, gel, gelcap, capsule, powder, solid tablet (coated or non-coated), tea, or the like.
- the composition is preferably in the form of a tablet or capsule and most preferably in the form of a hard gelatin capsule.
- Suitable excipient and/or carriers include maltodextrin, calcium carbonate, dicalcium phosphate, tricalcium phosphate, microcrystalline cellulose, dextrose, rice flour, magnesium stearate, stearic acid, croscarmellose sodium, sodium starch glycolate, crospovidone, sucrose, vegetable gums, lactose, methylcellulose, povidone, carboxymethylcellulose, corn starch, and the like (including mixtures thereof).
- Preferred carriers include calcium carbonate, magnesium stearate, maltodextrin, and mixtures thereof.
- the various ingredients and the excipient and/or carrier are mixed and formed into the desired form using conventional techniques.
- the tablet or capsule of the present invention may be coated with an enteric coating that dissolves at a pH of about 6.0 to 7.0.
- a suitable enteric coating that dissolves in the small intestine but not in the stomach is cellulose acetate phthalate. Further details on techniques for formulation for and administration may be found in the latest edition of Remington's Pharmaceutical Sciences (Maack Publishing Co., Easton, Pa.).
- the composition contains no traces of organic solvents which is an important property regarding the safety of consuming such compounds.
- Phospholipids prepared using chemical or enzymatic methods in the presence of organic solvents may contain residual solvents that may be a health hazard.
- VOC are often co-extracted when marine phospholipids are extracted, such VOC's may contribute to the smell taste of the phospholipids.
- the supplement is provided as a powder or liquid suitable for adding by the consumer to a food or beverage.
- the dietary supplement can be administered to an individual in the form of a powder, for instance to be used by mixing into a beverage, or by stirring into a semi-solid food such as a pudding, topping, sauce, puree, cooked cereal, or salad dressing, for instance, or by otherwise adding to a food.
- compositions of the present invention may also be formulated with a number of other compounds. These compounds and substances add to the palatability or sensory perception of the particles (e.g., flavorings and colorings) or improve the nutritional value of the particles (e.g., minerals, vitamins, phytonutrients, antioxidants, etc.).
- the dietary supplement may comprise one or more inert ingredients, especially if it is desirable to limit the number of calories added to the diet by the dietary supplement.
- the dietary supplement of the present invention may also contain optional ingredients including, for example, herbs, vitamins, minerals, enhancers, colorants, sweeteners, flavorants, inert ingredients, and the like.
- the dietary supplement of the present invention may contain one or more of the following: ascorbates (ascorbic acid, mineral ascorbate salts, rose hips, acerola, and the like), dehydroepiandosterone (DHEA), Fo-Ti or Ho Shu Wu (herb common to traditional Asian treatments), Cat's Claw ( ancient herbal ingredient), green tea (polyphenols), inositol, kelp, dulse, bioflavinoids, maltodextrin, nettles, niacin, niacinamide, rosemary, selenium, silica (silicon dioxide, silica gel, horsetail, shavegrass, and the like), spirulina, zinc, and the like.
- Such optional ingredients may be either naturally occurring or concentrated forms.
- the dietary supplements further comprise vitamins and minerals including, but not limited to, calcium phosphate or acetate, tribasic; potassium phosphate, dibasic; magnesium sulfate or oxide; salt (sodium chloride); potassium chloride or acetate; ascorbic acid; ferric orthophosphate; niacinamide; zinc sulfate or oxide; calcium pantothenate; copper gluconate; riboflavin; beta-carotene; pyridoxine hydrochloride; thiamin mononitrate; folic acid; biotin; chromium chloride or picolonate; potassium iodide; sodium selenate; sodium molybdate; phylloquinone; vitamin D 3 ; cyanocobalamin; sodium selenite; copper sulfate; vitamin A; vitamin C; inositol; potassium iodide.
- vitamins and minerals including, but not limited to, calcium phosphate or acetate, tribasic; potassium
- compositions comprise at least one food flavoring such as acetaldehyde(ethanal), acetoin(acetyl methylcarbinol), anethole(parapropenyl anisole), benzaldehyde(benzoic aldehyde), N-butyric acid (butanoic acid), d- or l-carvone(carvol), cinnamaldehyde(cinnamic aldehyde), citral(2,6-dimethyloctadien-2,6-al-8, gera-nial, neral), decanal(N-decylaldehyde, capraldehyde, capric aldehyde, caprinaldehyde, aldehyde C-10), ethyl acetate, ethyl butyrate, 3-methyl-3-phenyl glycidic acid ethyl ester(ethyl-methyl-phenyl-glycid)
- compositions comprise at least one synthetic or natural food coloring (e.g., annatto extract, astaxanthin, beet powder, ultramarine blue, canthaxanthin, caramel, carotenal, beta carotene, carmine, toasted cottonseed flour, ferrous gluconate, ferrous lactate, grape color extract, grape skin extract, iron oxide, fruit juice, vegetable juice, dried algae meal, tagetes meal, carrot oil, corn endosperm oil, paprika, paprika oleoresin, riboflavin, saffron, tumeric, tumeric and oleoresin).
- synthetic or natural food coloring e.g., annatto extract, astaxanthin, beet powder, ultramarine blue, canthaxanthin, caramel, carotenal, beta carotene, carmine, toasted cottonseed flour, ferrous gluconate, ferrous lactate, grape color extract, grape skin extract, iron oxide, fruit juice, vegetable juice, dried algae
- compositions comprise at least one phytonutrient (e.g., soy isoflavonoids, oligomeric proanthcyanidins, indol-3-carbinol, sulforaphone, fibrous ligands, plant phytosterols, ferulic acid, anthocyanocides, triterpenes, omega 3/6 fatty acids, conjugated fatty acids such as conjugated linoleic acid and conjugated linolenic acid, polyacetylene, quinones, terpenes, cathechins, gallates, and quercitin).
- phytonutrient e.g., soy isoflavonoids, oligomeric proanthcyanidins, indol-3-carbinol, sulforaphone, fibrous ligands, plant phytosterols, ferulic acid, anthocyanocides, triterpenes, omega 3/6 fatty acids, conjugated fatty acids such as conjugated linole
- Sources of plant phytonutrients include, but are not limited to, soy lecithin, soy isoflavones, brown rice germ, royal jelly, bee propolis, acerola berry juice powder, Japanese green tea, grape seed extract, grape skin extract, carrot juice, bilberry, flaxseed meal, bee pollen, ginkgo biloba, primrose (evening primrose oil), red clover, burdock root, dandelion, parsley, rose hips, milk thistle, ginger, Siberian ginseng, rosemary, curcumin, garlic, lycopene, grapefruit seed extract, spinach, and broccoli.
- the compositions comprise at least one vitamin (e.g., vitamin A, thiamin (B1), riboflavin (B2), pyridoxine (B6), cyanocobalamin (B12), biotin, ascorbic acid (vitamin C), retinoic acid (vitamin D), vitamin E, folic acid and other folates, vitamin K, niacin, and pantothenic acid).
- the particles comprise at least one mineral (e.g., sodium, potassium, magnesium, calcium, phosphorus, chlorine, iron, zinc, manganese, flourine, copper, molybdenum, chromium, selenium, and iodine).
- a dosage of a plurality of particles includes vitamins or minerals in the range of the recommended daily allowance (RDA) as specified by the United States Department of Agriculture.
- the particles comprise an amino acid supplement formula in which at least one amino acid is included (e.g., 1-carnitine or tryptophan).
- Thermomyces Lanuginosus lipase a variety of lipases is contemplated, including, but not limited to, Thermomyces Lanuginosus lipase, Rhizomucor miehei lipase, Candida Antarctica lipase, Pseudomonas fluorescence lipase, and Mucor javanicus lipase.
- free fatty acids are added such as EPA and DHA, however any omega-3 fatty acid is contemplated.
- a strong vacuum is applied to the reaction vessel for 72 hours.
- the reaction length can be varied in order to obtain a composition with the desired amount of phospholipids and lyso-phospholipids. By extending the reaction time beyond 72 hours, a product comprising more than 65% phospholipids can be obtained.
- a lipid carrier is added to the reaction mixture in order to reduce the viscosity of the solution.
- the added amount of triglycerides can be 10%, 20%, 30%, 40% or more, it depends on the requested viscosity of the final product.
- the lipid carrier can be a fish oil such as tuna oil, menhaden oil and herring oil, or any triglyceride, diglyceride, ethyl- or methylester of a fatty acid.
- the product is subjected to a molecular distillation and the free fatty acids are removed, resulting in a final product comprising of phospholipids (lyso-phospholipids and phospholipids) and triglycerides in a ratio of preferably 2:1.
- This invention further discloses a process for the enzymatic transesterification/esterification of phospholipids with fatty acids alkyl esters or free fatty acids in an evacuated vessel (B).
- a reduced pressure is applied to the vessel B (0.001-30 mbar) and water vapor (moisture) is allowed to enter the reaction mixture through a tube from a second vessel (A) ( FIG. 5 for schematic drawing of the experimental setup).
- the water in vessel A is heated to 25-30° C.
- the rate of reaction could either be increased of the lipase dosage could be reduced.
- the reuse of the enzymes was improved.
- a novel marine phospholipid composition was prepared characterized by being acylated in the range of 55%-85%, having at least 5% EPA and/or DHA esterified, having a EPA/DHA ratio of at least 1.
- the present invention utilizes a phospholipid, preferably a phosphatide such as lecithin.
- a phospholipid preferably a phosphatide such as lecithin.
- the present invention is not limited to the use of any particular phospholipid. Indeed, the use of a variety of phospholipids is contemplated.
- the phospholipid is a phosphatidic or lysophosphatidic acid.
- the phospholipid is a mixture of phosphatides such as phosphatidylcholine, phosphatidylethnolamine, phosphatidylserine and phosphatidylinositol.
- the present invention is not limited to the use of any particular source of phospholipids.
- the phospholipids are from soybeans, while in other embodiments, the phospholipids are from eggs.
- the phospholipids utilized are commercially available, such as Alcolec 40P® from American Lecithin Company Inc (Oxford, Conn., USA).
- Alcolec 40P® from American Lecithin Company Inc (Oxford, Conn., USA).
- the present invention is not limited to the use of any particular enzyme. Indeed, the use of a variety of enzymes is contemplated, including, but not limited to Thermomyces Lanuginosus lipase, Rhizomucor miehei lipase, Candida Antarctica lipase, Pseudomonas fluorescence lipase, and Mucor javanicus lipase.
- This invention is not limited to any particular fatty acid alkyl ester either. This includes, but not limited to: decanoic acid (10:0), undecanoic acid (11:0), 10-undecanoic acid (11:1), lauric acid (12:0), cis-5-dodecanoic acid (12:1), tridecanoic acid (13:0), myristic acid (14:0), myristoleic acid (cis-9-tetradecenoic acid, 14:1), pentadecanoic acid (15:0), palmitic acid (16:0), palmitoleic acid (cis-9-hexadecenoic acid, 16:1), heptadecanoic acid (17:1), stearic acid (18:0), elaidic acid (trans-9-octadecenoic acid, 18:1), oleic acid (cis-9-octadecanoic acid, 18:1), nonadecanoic acid (19:0), eicosanoi
- the synthetic marine phospholipid compositions of the present invention are substantially free of volatile organic compounds and are therefore much more suitable as a food supplement for humans and animals. Accordingly, in preferred compositions, the present invention provides synthetic marine phospholipids compositions having high or increased palatability, wherein the high or increased palatability is due to low levels of organic solvents and/or volatile organic compounds. In preferred embodiments, palatability is assayed by feeding the composition to a panel of subjects, preferably human. In more preferred embodiments, the phospholipids compositions have high or increased palatability as compared to naturally extracted marine phospholipids. In other preferred embodiments, the synthetic marine phospholipids compositions of the present invention are safe for oral administration.
- the difference in bioavailability and bioefficacy between the marine lipid composition of the present invention and a fish oil were investigated in a rat experiment.
- the rat feed was prepared using AIN-93 except that soybean oil was removed from the feed.
- the pelleted AIN-93 diet was ground and the marine lipid compositions (PL 1 and PL 2) as well as fish oil (TG oil) and control were added to this ground feed.
- the marine lipid compositions were prepared using enzymatic (lipase) catalyzed transesterification of soy lecithin with fish oil fatty acids according to the method described in Example 4, followed by the addition of a triglyceride carrier and short path distillation.
- Sprague Dawley rats Thirty six newly weaned male Sprague Dawley rats (start weight 168 ⁇ 11 g) were used in the experiment.
- the rats were initially given low-essential oil rat feed, containing 20 g of sunflower oil and 10 g of flaxseed oil per kg of feed, for one week.
- modified AIN-93 diet powder without the test oil was given to rats ad libitum until the start of the experiment. Feeding of rats was stopped 12 hours before the sampling, 30 days after the start of feeding.
- Each rat was individually anesthetized with carbon dioxide, weighed and euthanized with cervical dislocation. Next, blood was sampled and centrifuged to separate plasma and blood cells.
- the total fatty acid profile for the lipids in the brains (table 2), adipose tissue (table 3), liver (table 4), testicles (table 5) and heart (table 6) were isolated from the rats in example 1.
- the PL 1 composition increases the DHA content in brain and adipose tissue more than the TG composition.
- the PL 1 composition increase the EPA content in the adipose tissue more than the TG composition. It is to be observed that the PL1 composition increases the EPA/DHA content in the phospholipids and in the total lipids of the different tissues as well as reduces the AA/EPA ratio more than the TG oil composition.
- TABLE 2 Fatty acid profile of the total lipids isolated from the rat brain (mmol/g lipids).
- a marine phospholipid composition containing 8.4% EPA and 1.2% DHA was prepared using a crude soybean lecithin as a starting material according to [19].
- a marine oil was added to the phospholipid mixture (30% w/w) so that the total level of EPA was 21.9% and for DHA 9.4%.
- soy lecithin and lyso-phospholipids prepared according to [20] were added to the mixture in variable amounts so that a range of PC/LPC/GPC ratios could be obtained (Table 17).
- all the treatments (MPL1-MPL5) contained exactly the same amount EPA and DHA.
- Lipid compositions were consumed as a single bolus by adult Sprague-Dawley rats and the appearance of EPA/DHA in blood was measured at different time points from 1 to 12 hours after ingestion. The concentration of EPA/DHA was determined using GC-FID and reported as area percentage.
- FIGS. 6 and 7 show that composition MPL2 and MPL3 results in the highest concentration of EPA/DHA in plasma after uptake. Comparing the surface area under each curve it is clear that MPL2 and MPL3 demonstrates a higher bioavailability of EPA/DHA than the other composition MPL1, MPL4 and MPL5.
- Marine phospholipids were prepared using either 40% soy PC (American Lecithin Company Inc, Oxford, Conn., USA) (MPL1) or 96% pure soy PC (Phospholipid GmbH, GmbH, Germany) (MPL2) according to a method described by others [4]. Fatty acid content and the level of bi-products are shown in table 18.
- the MPL treatments consisted of a mixture of phospholipids, lyso-phospholipids and glycerol-phospholipids. Looking only at the PC/LPC/GPC relationship, it was 64/33/2 and 42/40/18 for MPL1 and MPL2, respectively. Finally, all three treatments were emulsified into skimmed milk.
- Sprague-Dawley rats were fed the milk emulsions for 1 week. Each rat was placed in its own cage to ensure that they got an even amount of test substance and the milk was consumed by the rat pups ad libitum. After 1 week the experiment was terminated and the rats were decapitated. The animals were kept without food for 24 hours before sampling. Entire livers were collected and frozen immediately using liquid nitrogen (stored at ⁇ 65° C.). Total RNA was isolated from the liver samples according to the Quiagen Rnaesy Midi Kit Protocol. The RNA samples were quantified and quality measured by NanoDrop and Bioanalyzer.
- FDR False Discovery Rate
- MPL1 and MPL2 are biologically different compounds due to the fact that over 40 genes were differentially expressed (table 19).
- TABLE 19 List of genes differentially expressed (DE) by MPL1 versus MPL2. The list is sorted according to increasing p-values.
- SLR Estimated signal log-ratio ( ⁇ 0: down regulated gene, >0: up regulated gene).
- Fold change Estimated fold change corresponding to the parameter ( ⁇ 1: down regulated gene, >1: up regulated gene).
- Affy fold change Estimated fold change using the Affymetrix definition ( ⁇ 1: down regulated gene, >1: up regulated gene)
- MPL2 regulates 401 genes versus the control (table 20).
- a number of genes listed are involved maintenance of the cell, in transcription and protein synthesis as well as signaling pathways. Others are involved in regulation of metabolism and the inflammatory response such as Tnf receptor-associated factor 6 (Traf6_predicted) (fold change of 0.53), guanine nucleotide binding protein alpha inhibiting 2 (Gnai2) (fold change of 0.6, gamma-butyrobetaine hydroxylase (Bbox1) (fold change of 1.32), monoglyceride lipase (Mg11) (fold change 0.52), nuclear NF-kappaB activating protein (fold change 0.65) and CCAAT/enhancer binding protein (C/EBP) (fold change of 0.66).
- Tnf receptor-associated factor 6 Traf6_predicted
- Gnai2 guanine nucleotide binding protein alpha inhibiting 2
- Bbox1 gamma-butyrobetaine hydroxylase
- fatty acid ethyl ester consisting of 10% EPA and 50% DHA (FAEE 10-50), obtained from Napro Pharma (Brattvaag, Norway) and 15 g TL-IM obtained from Novozymes (Bagsvaerd, Denmark) were mixed in an evacuated round bottomed glass flask for 15 minutes. Next, N 2 was released into the glass flask and the mixture was heated to 65° C. 20 g Alcolec 40P® from American Lecithin Company Inc (Oxford, Conn., USA) was then added to the reaction mixture.
- Alcolec 40P® is a crude soybean phospholipid product containing 40% PC, 26% phosphatidylethanolamine, 11% phosphatidylinositol, 1% phosphatidylserine, 13% phytoglycolipids as well as 14% other phosphatides (w,w).
- the glass flask was evacuated (20-30 mbar).
- a second vessel containing water (30° C.) was connected to the reaction vessel through a plastic tube ( FIG. 1 ). The reduced pressure allowed moisture from the headspace of the second vessel to be added through the reaction mixture continuously. In order to obtain the final product the enzymes were removed by filtration.
- a triglyceride carrier was added to the product, followed by removal of the residual free fatty acids and/or esters by short path distillation.
- the isolated PC+LPC fraction was then dried under nitrogen prior to derivatization; finally the fatty acid profile was determined by analyzing the derivatives using GC-FID.
- the relationship between PC, LPC and GPC was determined using HPLC with the method above, except that the UV detector was replaced by an evaporative light scattering detection (ELSD).
- ELSD evaporative light scattering detection
- the enzymes from example 7 were isolated by filtration and the possibility of reuse was determined in the following experiment. 30 g FAEE (10-50), 10 g Alcolec and 15 g used enzymes (equivalent to 7.5 g enzyme because the used enzymes had absorbed product from the first reaction). The reaction was performed at 65° C. and stirred at 200 rpm using a shaker incubator. The transesterified phospholipids were analyzed as in the previous example and the results are shown in table 21 below.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Molecular Biology (AREA)
- Biochemistry (AREA)
- Epidemiology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Engineering & Computer Science (AREA)
- Obesity (AREA)
- Nutrition Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Pain & Pain Management (AREA)
- Rheumatology (AREA)
- Diabetes (AREA)
- Hematology (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Coloring Foods And Improving Nutritive Qualities (AREA)
Abstract
Novel marine lipid compositions comprising triglycerides and omega-3 rich phospholipids are described. The compositions are characterized by providing highly bioavailable omega-3, increased tissue incorporation of omega-3 and reduced concentration of pro-inflammatory cytokines.
Description
- This application claims the benefit of U.S. Provisional Applications 60/798,026, 60/798,027, and 60/798,030, all filed May 5, 2006, and U.S. Provisional Application 60/872,096, filed Dec. 1, 2006, each of which is incorporated herein by reference in their entirety.
- The present invention relates to novel marine lipid compositions comprising combinations of omega-3 fatty acid rich functional phospholipids and omega-3 fatty acid rich triglycerides. In addition, food supplements, functional food, drugs and feed products comprising such compositions are provided along with methods of their use.
- Marine lipids such as omega-3 rich triglycerides and omega-3 rich phospholipids can be isolated from a number of different natural sources such as fish, crustaceans, plankton, seals, whales as well as algae using extraction technologies. In addition, they can be prepared industrially using chemical or bio-catalytical methods such as enzyme catalyzed transesterification of crude soy lecithin with fish oil fatty acids [1].
- The anti-inflammatory properties of omega-3 fatty acids are well known and the use as an anti-inflammatory agent has been described both for triglycerides and phospholipids [2-3]. Actually, omega-3 fatty acids are famous for their anti-inflammatory properties, and it has been shown that omega-3 fatty acids alleviate the symptoms of a series of autoimmune, atherosclerotic and inflammatory diseases including inflammatory bowel diseases and rheumatoid arthritis [4-6]. Suppression of inflammation has been proposed as one of the strategies to slow down the progress of these diseases. Hence, this invention discloses the effect on marine lipid compositions on the concentration of markers of inflammation such as TNF-α and other cytokines such as interleukin-1β and
interleukin 6. In addition, since arachidonic acid (AA) is the predominant precursor of the eicosanoid mediators of inflammatory responses (prostaglandins, thromboxanes and leukotrienes), this invention discloses the reduction of AA level and the improvement in the EPA/AA ratio in different lipid pools in tissues such as in the phospholipids isolated from adipose tissue, heart, testicles, plasma, brain and liver. - The bioavailability of EPA and DHA from fish oil triglycerides have been reported to be high in healthy adults. However, for certain conditions i.e. pathological conditions such as extrahepatic cholestasis and for pre-term infants the absorption can be low. For example it was shown that the absorption of DHA from egg lecithin in pre-term infants was 90% compared to 80% from triglycerides [7]. Absorption of long chain PUFA (AA and DHA) is less (75% and 62%, respectively) than the absorption of C18 PUFA (94%) in pre-term infants [8]. The difference between C18 PUFA and long chain PUFA absorption is likely to become less apparent in older children and adults. Sala-Vila et al [9-10] investigated the bioavailabilities of DHA-PL and DHA-TG in full term infants and found no differences based on plasma lipid enrichments. Valenzuela et al. [11] supplemented female rats with different forms of DHA including egg yolk PL and single cell algae TG. They found also no difference in absorption of DHA from PL and TG based on plasma lipid enrichments. However, the tissue and milk fat levels were higher in PL-DHA compared to the TG-DHA supplemented rats. These data indicate that although there were no differences in the bioavailability, efficacy with respect to tissue enrichment was higher for PL-DHA compared to TG-DHA. Furthermore, the relative absorption of EPA and DHA ethyl esters (4 g/d) compared to oleic acid calculated from peak concentrations was 94 and 100%, respectively. Estimates of relative absorption based on the area under the concentration curve indicated a relative absorption of 91% for EPA and 93% for DHA [12]. Bioavailability of C18:1, C18:2 and C18:3 in adult humans are close to 100% (note 94% in preterm infants). Thus the bioavailability of EPA and DHA delivered in different forms is, according to previous, work likely to be over 90%.
- In some embodiments, the invention provides a composition comprising a triglyceride and a phospholipids in a ratio ranging from 1:10 to 10:1; said phospholipids having the following structure:
wherein R1 is OH or a fatty acid, R2 is OH or a fatty acid, and R3 is a mixture of H, choline, ethanolamine, inositol and serine, said phospholipid having at least 1% of DHA/EPA, said phospholipids have a concentration of OH in the range of 25-50%. In further embodiments, the invention provides a marine lipid composition characterized by providing higher uptake of omega-3 fatty acids into plasma as compared to administration of purified triglycerides, phospholipids, or natural marine phospholipids. In further embodiments, the invention provides a composition characterized by efficiently improving the AA/EPA ratio in plasma phospholipids as compared to administration of purified triglycerides, phospholipids, or natural marine phospholipids. In still other embodiments, the invention is a marine lipid composition characterized by efficiently increasing the concentration of omega-3 fatty acids in tissues as compared to administration of purified triglycerides, phospholipids, or natural marine phospholipids. In still further embodiments, the invention the composition is characterized by reducing the concentration of biomarkers of inflammation as compared to administration of purified triglycerides, phospholipids, or natural marine phospholipids. In other embodiments of the invention, the marine lipid composition is formulated into an animal feed, a food product, a food supplement and a drug. - In some embodiments, the present invention provides a composition comprising phospholipids having the following structure:
wherein R1 is OH or a fatty acid, R2 is OH or a fatty acid, and R3 is a mixture of H, choline, ethanolamine, inositol and serine, said phospholipid having at least 1% of DHA/EPA at positions R1 and/or R2 and from about 20-50% of OH at positions R1 and/or R2. In some embodiments, the composition is acylated in a range from about 55% to about 85%. In some embodiments, the omega-3 fatty acids are selected from the group consisting of EPA, DHA, DPA and α-linolenic acid (ALA). In some embodiments, the composition is substantially free of organic solvents and volatile organic compounds such as short chain fatty acids, short chain aldehydes and short chain ketones. In some embodiments, the composition has at least 5% of a combination of EPA and DHA esterified. In some embodiments, the composition has at least 10% of a combination of EPA and DHA esterified. In some embodiments, the composition has at least 20% of a combination of EPA and DHA esterified. In some embodiments, the composition has at least 30% of a combination of EPA and DHA esterified. In yet other embodiments, said composition contains from about 5%, 10%, 20% and 30% EPA/DHA attached toposition 1 and/ orposition 2. In some embodiments, the composition has a ratio of EPA/DHA ranging from 1:1 to 4:1. In some embodiments, the composition has a ratio of EPA/DHA ranging from 2:1 to 4:1. In some embodiments, the composition is acylated in a range from 60% to 80%. In some embodiments, the composition is acylated in a range from 50% to 75%. - In some embodiments, the composition further comprises a lipid carrier in a ratio of from 1:10 to 10:1 to said phospholipids. In some embodiments, the lipid carrier and said phospholipids are in a ratio of from about 5:1 to 1:5. In some embodiments, the composition comprises from about 20% to about 90% of said phospholipid composition and from about 10% to about 50% of said lipid carrier. The present invention is not limited to any particular lipid carrier. In some embodiments, the lipid carrier is selected from the group consisting of a triglyceride, a diglyceride, an ethyl ester, and a methyl ester and combinations thereof. In some embodiments, the composition provides higher uptake of omega-3 fatty acids into plasma as compared to natural marine phospholipids when administered to subjects. In some embodiments, the composition improves the AA/EPA ratio in plasma phospholipids when administered to subjects as compared to natural marine phospholipids. In some embodiments, the composition increases the concentration of omega-3 fatty acids in tissues when administered to subjects as compared to natural marine phospholipids. In some embodiments, the composition reduces the concentration of biomarkers of inflammation when administered to subjects as compared to natural marine phospholipids. In some embodiments, the present invention provides a food product comprising the foregoing compositions. In some embodiments, the present invention provides an animal feed comprising the foregoing compositions. In some embodiments, the present invention provides a food supplement comprising the foregoing compositions. In some embodiments, the present invention provides a pharmaceutical composition comprising the foregoing compositions.
- In some embodiments, the present invention provides methods of preparing a bioavailable omega-3 fatty acid composition comprising: a) providing a purified phospholipid composition comprising omega-3 fatty acid residues and a purified triglyceride composition comprising omega-3 fatty acid residues; b) combining said phospholipid composition and said triglyceride composition to form a bioavailable omega-3 fatty acid composition. In some embodiments, the bioavailable phospholipid composition is one of the compositions described above. In some embodiments, the methods further comprise the step of encapsulating said bioavailable omega-3 fatty acid composition. In some embodiments, the bioavailable omega-3 fatty acid composition has increased bioavailability as compared to purified triglycerides or phospholipids comprising omega-3 fatty acid residues. In some embodiments, the methods further comprise the step of packaging the bioavailable omega-3 fatty acid composition for use in functional foods. In some embodiments, the methods further comprise the step of assaying the bioavailable omega-3 fatty acid composition for bioavailability. In some embodiments, the methods further comprise administering the bioavailable omega-3 fatty acid composition to a patient. In some embodiments, the present invention provides a food product, animal feed, food supplement or pharmaceutical composition made by the foregoing process.
- In some embodiments, the present invention provides methods for reducing symptoms of cognitive dysfunction in a child comprising administering an effective amount of a marine phospholipid composition, wherein said symptoms are selected from the group consisting of ability to complete task, ability to stay on task, ability to follow instructions, ability to complete assignments, psychomotor function, long term memory, short term memory, ability to make a decision, ability to follow through on decision, ability to self-sustain attention, ability to engage in conversations, sensitivity to surroundings, ability to plan, ability to carry out plan, ability to listen, interruptions in social situations, temper tantrums, level/frequency of frustration, level/frequency restlessness, frequency/level fidgeting, ability to exhibit delayed gratification, aggressiveness, demanding behavior/frequency of demanding behavior, sleep patterns, restive sleep, interrupted sleep, awakening behavior, disruptive behavior, ability to exhibit control in social situations, ability to extrapolate information and ability to integrate information. In some embodiments, the child exhibits one or more symptoms of Attention Deficit Hyperactivity Disorder (ADHD), is suspected of having ADHD, or has been diagnosed with ADHD. In some embodiments, the child exhibits one or more symptoms of autistic spectrum disorder, is suspected of having autistic spectrum disorder, or has been diagnosed with autistic spectrum disorder. In further embodiments, the present invention provides methods of increasing cognitive performance in an aging mammal comprising administering an effective amount of a marine phospholipid composition. In some embodiments, the cognitive performance is selected from the group consisting of memory loss, forgetfulness, short-term memory loss, aphasia, disorientation, disinhibition, and behavioral changes. In some embodiments, the mammal is a human. In some embodiments, the mammal is a pet selected from the group consisting of cats and dogs. In some embodiments, the mammal has symptoms of age-associated memory impairment or decline.
- The foregoing methods are not limited to the use of any particular marine phospholipid composition. In some embodiments, the marine phospholipid composition comprises phospholipids having the following structure:
wherein R1 is OH or a fatty acid, R2 is OH or a fatty acid, and R3 is a mixture of H, choline, ethanolamine, inositol and serine, said phospholipid having at least 1% of omega-3 fatty acid moieties at positions R1 and/or R2. In some embodiments, the phospholipid composition comprises from about 20-50% of OH at positions R1 and/or R2. In some embodiments, the phospholipid composition further comprises a lipid carrier. In some embodiments, the phospholipid composition is prepared from natural marine phospholipids isolated from a marine organism. In some embodiments, the phospholipid composition is enzymatically prepared by reacting lecithin with DHA and EPA in the presence of an enzyme. In some embodiments, the lecithin is soybean or egg lecithin. In some embodiments, the omega-3 fatty acid moieties are selected from the group of EPA and DHA and combination thereof. In some embodiments, the effective amount of said phospholipid composition comprises from about 300 to about 1000 mg omega-3 fatty acids. In some embodiments, the phospholipid composition is administered orally. In some embodiments, the phospholipid composition is provided in a gel capsule or pill. - In some embodiments, the present invention provides methods of treating a subject by administration of a marine phospholipid composition comprising administering a marine phospholipid composition to said subject under conditions such that a desired condition is improved, wherein said conditions is selected from the group consisting of fertility, physical endurance, sports performance, muscle soreness, inflammation, auto-immune stimulation, metabolic syndrome, obesity and type II diabetes. In some embodiments, the subject is a human. In some embodiments, the subject is a companion animal. The present invention is not limited to any particular marine phospholipid composition. In some embodiments, the marine phospholipid composition comprises phospholipids having the following structure:
wherein R1 is OH or a fatty acid, R2 is OH or a fatty acid, and R3 is a mixture of H, choline, ethanolamine, inositol and serine, said phospholipid having at least 1% of omega-3 fatty acid moieties at positions R1 and/or R2. In some embodiments, the phospholipid composition comprises from about 20-50% of OH at positions R1 and/or R2. In some embodiments, the phospholipid composition is prepared from natural marine phospholipids isolated from a marine organism. In some embodiments, the composition further comprises a lipid carrier. In some embodiments, the phospholipid composition is enzymatically prepared by reacting lecithin with DHA and EPA in the presence of an enzyme. In some embodiments, the-lecithin is soybean or egg lecithin. In some embodiments, the omega-3 fatty acid moieties are selected from the group of EPA and DHA and combination thereof. In some embodiments, the effective amount of said phospholipid composition comprises from about 300 to about 1000 mg omega-3 fatty acids. In some embodiments, the phospholipid composition is administered orally. In some embodiments, the phospholipid composition is provided in a gel capsule or pill. In some embodiments, the human is a male. - In some embodiments, the present invention provides methods for prophylactically treating a subject by administration of a marine phospholipid composition comprising administering a marine phospholipid composition to a subject under conditions such that an undesirable condition is prevented, wherein said undesirable condition is selected from the group consisting of weight gain, infertility, obesity, metabolic syndrome, diabetes type II, mortality in subjects with a high risk of sudden cardiac death, and induction of sustained ventricular tachycardia. In some embodiments, the subject is at risk for developing a condition selected from the group consisting of weight gain, obesity, metabolic syndrome, diabetes type II, mortality in subjects with a high risk of sudden cardiac death, and induction of sustained ventricular tachycardia.
- In some embodiments, the subject is a human. In some embodiments, the subject is a companion animal. The present invention is not limited to any particular marine phospholipid composition. In some embodiments, the marine phospholipid composition comprises phospholipids having the following structure:
wherein R1 is OH or a fatty acid, R2 is OH or a fatty acid, and R3 is a mixture of H, choline, ethanolamine, inositol and serine, said phospholipid having at least 1% of omega-3 fatty acid moieties at positions R1 and/or R2. In some embodiments, the phospholipid composition comprises from about 20-50% of OH at positions R1 and/or R2. In some embodiments, the phospholipid composition is prepared from natural marine phospholipids isolated from a marine organism. In some embodiments, the composition further comprises a lipid carrier. In some embodiments, the phospholipid composition is enzymatically prepared by reacting lecithin with DHA and EPA in the presence of an enzyme. In some embodiments, the lecithin is soybean or egg lecithin. In some embodiments, the omega-3 fatty acid moieties are selected from the group of EPA and DHA and combination thereof. In some embodiments, the effective amount of said phospholipid composition comprises from about 300 to about 1000 mg omega-3 fatty acids. In some embodiments, the phospholipid composition is administered orally. In some embodiments, the phospholipid composition is provided in a gel capsule or pill. -
FIG. 1 . The total amount of EPA consumed during the four-week rat trial (mean±SE). -
FIG. 2 . Relative EPA (20:5) content of plasma (mean±SE; n=6). -
FIG. 3 . Relative 20:5 content of red blood cells (mean±SE; n=5-6). -
FIG. 4 . Relative 20:5 content of monocytes (mean±SE; n=5-6). -
FIG. 5 . Schematic drawing of experimental set-up. -
FIG. 6 . EPA levels in plasma as a function of hours after one bolus intake of a marine phospholipid composition. -
FIG. 7 . EPA levels in plasma as a function of hours after one bolus intake of a marine phospholipid composition. -
FIG. 8 . ARA levels in plasma as a function of hours after one bolus intake of a marine phospholipid composition. - As used herein, “phospholipid” refers to an organic compound having the following general structure:
wherein R1 is a fatty acid residue or —OH, R2 is a fatty acid residue or —OH, and R3 is a —H or a nitrogen containing compound such as choline (HOCH2CH2N+(CH3)3OH−), ethanolamine (HOCH2CH2NH2), inositol or serine. R1 and R2 cannot simultaneously be OH. When R3 is an —OH, the compound is a diacylglycerophosphate, while when R3 is a nitrogen-containing compound, the compound is a phosphatide such as lecithin, cephalin, phosphatidyl serine or plasmalogen. - The R1 site is herein referred to as
position 1 of the phospholipid, the R2 site is herein referred to asposition 2 of the phospholipid, and the R3 site is herein referred to asposition 3 of the phospholipid. - As used herein, the term omega-3 fatty acid refers to polyunsaturated fatty acids that have the final double bond in the hydrocarbon chain between the third and fourth carbon atoms from the methyl end of the molecule. Non-limiting examples of omega-3 fatty acids include, 5,8,11,14,17-eicosapentaenoic acid (EPA), 4,7,10,13,16,19-docosahexanoic acid (DHA) and 7,10,13,16,19-docosapentanoic acid (DPA).
- As used herein, the term “bioavailability” refers to the degree and rate at which a substance (as a drug) is absorbed into a living system or is made available at the site of physiological activity.
- As used herein, the term “functional food” refers to a food product to which a biologically active supplement has been added.
- As used herein, the term “fish oil” refers to any oil obtained from a marine source e.g. tuna oil, seal oil and algae oil.
- As used herein, the term “lipase” refers to any enzyme capable of hydrolyzing fatty acid esters
- As used herein, the term “food supplement” refers to a food product formulated as a dietary or nutritional supplement to be used as part of a diet.
- As used herein, the term “acylation” means fatty acids attached to the phospholipid. 100% acylation means that there are no lyso- or glycerol-phospholipids.
- This invention discloses that the uptake/absorption of omega-3 fatty acids attached to phospholipids are dependent on the level of LPL and GPL. Preferably, in order to ensure maximum uptake the level of LPL should be in the range of 15-45% and the level of GPL should be 0%. Furthermore, this invention discloses that the pure PC transesterified with EPA/DHA have a different effect on gene expression in the liver than 40% PC transesterified with EPA/DHA. It is disclosed that the two compositions regulated around 40 genes differently. Furthermore, the invention discloses that the EPA/DHA ratio is important. The treatment containing a EPA/DHA ratio of 2:1 regulated key enzymes involved in the inflammatory response (NF-κB) in a positive way, the treatment containing a EPA/DHA ratio of 1:1 did not.
- The present invention describes novel marine lipid compositions comprising an omega-3 containing phospholipid and a triacylglyceride (TG) in a ratio from about 1:10 to 10:1. Preferably the ratio is in the range of from about 3:1 to 1:3, more preferably the ratio is in the range of about 1:2 to 2:1. Preferably, the TG is a fish oil such as tuna oil, herring oil, menhaden oil, cod liver oil and algae oil. However, this invention is not limited to omega-3 containing oils as other TG sources are contemplated such as vegetable oils. The phospholipids in the composition have the following structure:
wherein R1 is OH or a fatty acid, R2 is OH or a fatty acid, and R3 is a mixture of H, choline, ethanolamine, inositol and serine. Attached toposition 1 orposition 2 are least 1% omega-3 fatty acids, preferably at least 5%, more preferably at least 10% omega-3 fatty acids, up to about 15%, 20%, 30%, 40%, 50%, or 60% omega-3 fatty acids. The omega-3 fatty acids can be EPA, DHA, DPA or C18:3 (n-3), most preferably the omega-3 fatty acids are EPA and DHA. The phospholipid composition preferably contains OH inposition 1 orposition 2 in a range of 25% to 50% in order to maximize absorption in-vivo. - In some embodiments, the present invention provides bioavailable and bioefficient omega-3 fatty acids. This invention shows that the novel marine lipid composition disclosed above enhances the uptake of the omega-3 fatty acid in vivo and incorporates omega-3 fatty acids more efficiently into tissues of adult rats than pure fish oil does. An embodiment of the invention is to use the marine lipid composition for efficient increase of omega-3 fatty acids in the liver, brain, adipose tissue, plasma, testicles and heart. Furthermore, this invention also discloses that the marine lipid compositions efficiently reduced the concentration of the pro-inflammatory precursor AA in total lipids and in phospholipids in tissues. It is disclosed that the concentration of AA in the different lipid pools in the liver, brain, adipose tissue, plasma, testicles and heart can be more efficiently reduced than using fish oil. Hence, the composition can be used to improve the EPA/AA ratio, which is a bio-marker of silent inflammation. The invention also discloses that the incorporation of the omega-3 fatty acids into monocytes is also more efficient using the claimed marine lipid composition as opposed to the fish oil. Yet another embodiment of the invention is to use the marine lipid composition to reduce chronic and acute inflammation in humans and in animals. Acute inflammation is mediated by granulocytes or polymorphonuclear leukocytes, while chronic inflammation is mediated by mononuclear cells such as monocytes. Monocytes protect against blood-borne pathogens and moves quickly to sites of infection in the tissues, secreting large amounts of pro-inflammatory prostaglandins. Furthermore, low grade chronic inflammation may be the underlying cause of many life-style related diseases such as obesity, arthritis, diabetes type II, metabolic syndrome, Alzheimer's disease, osteoarthritis, inflammatory bowel disease, allergy and asthma [14]. Hence, the marine lipid composition can be used to treat and prevent diseases linked to chronic inflammation. This invention discloses that the inflammatory response of monocytes harvested from animals in lower in animals treated with the marine lipid composition compared to fish oil. The concentrations of the pro-inflammatory cytokines such as interleukin-1β, interleukin-6 as well as tumor necrosis factor α (TNF-α) were reduced for the group fed the marine lipid composition compared to fish oil. These cytokines are important markers of real inflammation as for examples I1-1β induces fever. I1-6 also induces fever in addition to being linked to the acute phase response. TNF-α is involved in systemic inflammation as well and is released by white blood cells in the case of damage. It has a range of different biological effects such as increasing insulin resistance, stimulating the acute phase response in the liver and affecting the hypothalamus causing appetite suppression and fever.
- This invention also discloses that the fatty acid composition of the brain and adipose tissue phospholipids changes after in take of omega-3 fatty acids for 30 days. A significant reduction of the arachidonic acids can be found in the phospholipids in the brain and adipose tissue for the rats given either the EPA- or DHA-rich PL diets (
PL 1 andPL 2, respectively). This may affect the inflammatory response in this tissue and thereby have a great impact on cognitive diseases/conditions such as Parkinson's or and Alzheimer's where the inflammatory component is fundamental for the progression of the disease. This invention also discloses that the reduction of ARA is present also in the sn-2 position of the phospholipids in the brain. This is very important as the pro-inflammatory eicosanoids are produced from ARA, which are catalytically hydrolyzed fromposition 2 on the phospholipid by the action of phospholipase A2. The phospholipase A2 is released after stimuli at the cell wall, it then moves to the nuclear membrane where the hydrolysis of the phospholipid takes place. - In adipose tissue, accumulation of EPA and DHA in both total lipids (table 3) and PLs (table 8) is substantial when omega-3 supplements were fed and negligible when the control diet was fed. The increase was more pronounced in total lipids, which mainly consists of triglycerides (99% of fat cell lipid content). This invention demonstrates that omega-3 phospholipids can increase the accumulation of EPA/DHA into adipose tissue. This is important as the adipose tissue can function as a reservoir for these fatty acids. Arachidonic acid concentration in total lipids was higher in omega-3 supplemented animals, showing probably an increase of lipoprotein lipase activity, in agreement with the ability of
omega 3 in decreasing plasma TAGs concentration. On the other hand, arachidonic acid levels in adipose tissue PLs were significantly lower in omega-3 supplemented animals than the levels in controls. Peculiar enough, the PL-EPA diet was the most efficient in decreasing arachidonic acid. In addition, the invention discloses that the reduction of ARA is also observed in the sn-2 position of the phospholipids of the omega-3 supplemented animals. This is very important as the pro-inflammatory eicosanoids are produced from ARA, which are catalytically hydrolyzed fromposition 2 on the phospholipid by the action of phospholipase A2. The phospholipase A2 is released after stimuli at the cell wall, it then moves to the nuclear membrane where the hydrolysis of the phospholipid takes place. The reduction of ARA inposition 2 on the phospholipids may affect the inflammatory response in this tissue, which may have practical application in different pathologies of the adipose tissue and in its physiological activity of accumulation and release of fatty acids. - Fatty acid data from brain are well in line with the data from adipose tissue. Also in this tissue, we found a significant decrease of arachidonic acid in PLs, but surprisingly, only with PL-EPA and PL-DHA (table 7). On the other hand, DHA levels in both total lipids and PLs were not influenced by the omega-3 diets, while there was a small but significant increase in EPA levels. Lack of increase in DHA levels is likely to be attributable to the fact that the rats in this study were adults and pass the stage in development where they incorporate DHA in the brain (mainly PE). On the other hand, EPA being present at low concentration has more margin to increase. Furthermore, this may affect the inflammatory response in this tissue, which may have a great impact in such diseases as Parkinson's and Alzheimer's where the inflammatory component is fundamental for the progression of the disease. Positional distribution of arachidonic acid show that the ARA content is reduced for the EPA-PL groups, as stated before this is very important as it influences the pro-inflammatory eicosanoid production.
- In liver, as expected, we found for all omega-3 groups a significant increase of EPA and DHA and decrease of arachidonic acid. No great differences were expected between total lipids and PLs because about 80% of liver total lipids are PLs (table 4, 9 and 14).
- Heart total lipids and PLs (table 6 and table 11, respectively) showed a strong increase of EPA and DHA with a concomitant decrease of arachidonic acid when omega-3 supplements were fed. The strong decrease in the omega-6/omega-3 ratio in heart lipids is important considering the possible impact on the anti-inflammatory potential. Observed change in heart tissue fatty acids (increase of fatty acids with 6 or 5 double bonds) also suggests a possible increase in membrane fluidity. This change was most striking in the PL-DHA group where the increase of DHA was significantly higher than the increase in the TG-oil and PL-EPA groups. The fluidity of myocardium cell membrane seems to play an important role in controlling arrhythmia. Ventricular arrhythmia, is one of the main causes of sudden cardiac death. Furthermore, atrial fibrillation is another pathological state with a high incidence and important health consequences.
- Testicular long chain PUFAs are of special interest because there is a high rate of production of prostaglandins from the omega-6 PUFA (arachidonic acid mainly) into the semen or seminal fluid. High rate of prostaglandin production does not indicate an active inflammatory process but a stimulus for the uterus smooth muscle to favor male fertility. An omega-3 induced decrease of arachidonic acid as observed in other tissue could be detrimental to the male fertility if it occurred also in testis. Furthermore, testicular tissue has also a high level of DPA (22:5 omega-6), which may serve as a reservoir for arachidonic acid. Arachidonic acid could be formed according to the need, through the retroconversion mechanism in the peroxisomes. A similar mechanism may take place with DHA to form EPA in other tissues. Our data (table 5) show an increase of EPA and DHA and a small decrease of arachidonic acid in the total lipids fraction when omega-3 fatty acids are fed. However, there is no change in arachidonic acid levels in PL when TG-oil and PL-EPA are fed and interestingly a significant increase in the PL-DHA group. Furthermore, DPA n-6 concentration in total lipids was not influenced by omega-3 supplementation but there was a significant increase in DPA in the PL-EPA group (table 5). Overall, these data seem to indicate that the diets with omega-3 did not change the arachidonic and DPA n-6 concentrations in a way that would predict negative effects on male fertility. In contrast, increase in arachidonic acid content of testicular PLs (table 10) when PL-DHA was fed and increase in DPA when PL-EPA was fed could be interpreted to be positively associated with male fertility.
- Another embodiment of the invention is to formulate the marine lipid compositions into a feed product for the purpose of reducing low-grade chronic inflammation in animals. It can also be formulated into a food product and given to humans for the same purpose. Furthermore, it can be formulated as a functional food product, as a drug or as food supplement.
- In some embodiments, the compositions of this invention are contained in acceptable excipients and/or carriers for oral consumption. The actual form of the carrier, and thus, the compositions itself, is not critical. The carrier may be a liquid, gel, gelcap, capsule, powder, solid tablet (coated or non-coated), tea, or the like. The composition is preferably in the form of a tablet or capsule and most preferably in the form of a hard gelatin capsule. Suitable excipient and/or carriers include maltodextrin, calcium carbonate, dicalcium phosphate, tricalcium phosphate, microcrystalline cellulose, dextrose, rice flour, magnesium stearate, stearic acid, croscarmellose sodium, sodium starch glycolate, crospovidone, sucrose, vegetable gums, lactose, methylcellulose, povidone, carboxymethylcellulose, corn starch, and the like (including mixtures thereof). Preferred carriers include calcium carbonate, magnesium stearate, maltodextrin, and mixtures thereof. The various ingredients and the excipient and/or carrier are mixed and formed into the desired form using conventional techniques. The tablet or capsule of the present invention may be coated with an enteric coating that dissolves at a pH of about 6.0 to 7.0. A suitable enteric coating that dissolves in the small intestine but not in the stomach is cellulose acetate phthalate. Further details on techniques for formulation for and administration may be found in the latest edition of Remington's Pharmaceutical Sciences (Maack Publishing Co., Easton, Pa.).
- In other embodiments, the composition contains no traces of organic solvents which is an important property regarding the safety of consuming such compounds. Phospholipids prepared using chemical or enzymatic methods in the presence of organic solvents may contain residual solvents that may be a health hazard. VOC are often co-extracted when marine phospholipids are extracted, such VOC's may contribute to the smell taste of the phospholipids.
- In other embodiments, the supplement is provided as a powder or liquid suitable for adding by the consumer to a food or beverage. For example, in some embodiments, the dietary supplement can be administered to an individual in the form of a powder, for instance to be used by mixing into a beverage, or by stirring into a semi-solid food such as a pudding, topping, sauce, puree, cooked cereal, or salad dressing, for instance, or by otherwise adding to a food.
- The compositions of the present invention may also be formulated with a number of other compounds. These compounds and substances add to the palatability or sensory perception of the particles (e.g., flavorings and colorings) or improve the nutritional value of the particles (e.g., minerals, vitamins, phytonutrients, antioxidants, etc.).
- The dietary supplement may comprise one or more inert ingredients, especially if it is desirable to limit the number of calories added to the diet by the dietary supplement. For example, the dietary supplement of the present invention may also contain optional ingredients including, for example, herbs, vitamins, minerals, enhancers, colorants, sweeteners, flavorants, inert ingredients, and the like. For example, the dietary supplement of the present invention may contain one or more of the following: ascorbates (ascorbic acid, mineral ascorbate salts, rose hips, acerola, and the like), dehydroepiandosterone (DHEA), Fo-Ti or Ho Shu Wu (herb common to traditional Asian treatments), Cat's Claw (ancient herbal ingredient), green tea (polyphenols), inositol, kelp, dulse, bioflavinoids, maltodextrin, nettles, niacin, niacinamide, rosemary, selenium, silica (silicon dioxide, silica gel, horsetail, shavegrass, and the like), spirulina, zinc, and the like. Such optional ingredients may be either naturally occurring or concentrated forms.
- In some embodiments, the dietary supplements further comprise vitamins and minerals including, but not limited to, calcium phosphate or acetate, tribasic; potassium phosphate, dibasic; magnesium sulfate or oxide; salt (sodium chloride); potassium chloride or acetate; ascorbic acid; ferric orthophosphate; niacinamide; zinc sulfate or oxide; calcium pantothenate; copper gluconate; riboflavin; beta-carotene; pyridoxine hydrochloride; thiamin mononitrate; folic acid; biotin; chromium chloride or picolonate; potassium iodide; sodium selenate; sodium molybdate; phylloquinone; vitamin D3; cyanocobalamin; sodium selenite; copper sulfate; vitamin A; vitamin C; inositol; potassium iodide. Suitable dosages for vitamins and minerals may be obtained, for example, by consulting the U.S. RDA guidelines.
- In further embodiments, the compositions comprise at least one food flavoring such as acetaldehyde(ethanal), acetoin(acetyl methylcarbinol), anethole(parapropenyl anisole), benzaldehyde(benzoic aldehyde), N-butyric acid (butanoic acid), d- or l-carvone(carvol), cinnamaldehyde(cinnamic aldehyde), citral(2,6-dimethyloctadien-2,6-al-8, gera-nial, neral), decanal(N-decylaldehyde, capraldehyde, capric aldehyde, caprinaldehyde, aldehyde C-10), ethyl acetate, ethyl butyrate, 3-methyl-3-phenyl glycidic acid ethyl ester(ethyl-methyl-phenyl-glycidate, strawberry aldehyde, C-16 aldehyde), ethyl vanillin, geraniol(3,7-dimethyl-2,6 and 3,6-octadien-1-ol), geranyl acetate (geraniol acetate), limonene (d-, l-, and dl-), linalool (linalol, 3,7-dimethyl-1,6-octadien-3-ol), linalyl acetate(bergamol), methyl anthranilate(methyl-2-aminobenzoate), piperonal(3,4-methylenedioxy-benzaldehyde, heliotropin), vanillin, alfalfa (Medicago sativa L.), allspice (Pimenta officinalis), ambrette seed (Hibiscus abelmoschus), angelic (Angelica archangelica), Angostura (Galipea officinalis), anise (Pimpinella anisum), star anise (Illicium verum), balm (Melissa officinalis), basil (Ocimum basilicum), bay (Laurus nobilis), calendula (Calendula officinalis), (Anthemis nobilis), capsicum (Capsicum frutescens), caraway (Carum carvi), cardamom (Elettaria cardamomum), cassia, (Cinnamomum cassia), cayenne pepper (Capsicum frutescens), Celery seed (Apium graveolens), chervil (Anthriscus cerefolium), chives (Allium schoenoprasum), coriander (Coriandrum sativum), cumin (Cuminum cyminum), elder flowers (Sambucus canadensis), fennel (Foeniculum vulgare), fenugreek (Trigonella foenum-graecum), ginger (Zingiber officinale), horehound (Marrubium vulgare), horseradish (Armoracia lapathifolia), hyssop (Hyssopus officinalis), lavender (Lavandula officinalis), mace (Myristica fragrans), maroram (Majorana hortensis), mustard (Brassica nigra, Brassica juncea, Brassica hirta), nutmeg (Myristica fragrans), paprika (Capsicum annuum), black pepper (Piper nigrum), peppermint (Mentha piperita), poppy seed (Papayer somniferum), rosemary (Rosmarinus officinalis), saffron (Crocus sativus), sage (Salvia officinalis), savory (Satureia hortensis, Satureia montana), sesame (Sesamum indicum), spearmint (Mentha spicata), tarragon (Artemisia dracunculus), thyme (Thymus vulgaris, Thymus serpyllum), turmeric (Curcuma longa), vanilla (Vanilla planifolia), zedoary (Curcuma zedoaria), sucrose, glucose, saccharin, sorbitol, mannitol, aspartame. Other suitable flavoring are disclosed in such references as Remington's Pharmaceutical Sciences, 18th Edition, Mack Publishing, p. 1288-1300 (1990), and Furia and Pellanca, Fenaroli's Handbook of Flavor Ingredients, The Chemical Rubber Company, Cleveland, Ohio, (1971), known to those skilled in the art.
- In other embodiments, the compositions comprise at least one synthetic or natural food coloring (e.g., annatto extract, astaxanthin, beet powder, ultramarine blue, canthaxanthin, caramel, carotenal, beta carotene, carmine, toasted cottonseed flour, ferrous gluconate, ferrous lactate, grape color extract, grape skin extract, iron oxide, fruit juice, vegetable juice, dried algae meal, tagetes meal, carrot oil, corn endosperm oil, paprika, paprika oleoresin, riboflavin, saffron, tumeric, tumeric and oleoresin).
- In still further embodiments, the compositions comprise at least one phytonutrient (e.g., soy isoflavonoids, oligomeric proanthcyanidins, indol-3-carbinol, sulforaphone, fibrous ligands, plant phytosterols, ferulic acid, anthocyanocides, triterpenes,
omega 3/6 fatty acids, conjugated fatty acids such as conjugated linoleic acid and conjugated linolenic acid, polyacetylene, quinones, terpenes, cathechins, gallates, and quercitin). Sources of plant phytonutrients include, but are not limited to, soy lecithin, soy isoflavones, brown rice germ, royal jelly, bee propolis, acerola berry juice powder, Japanese green tea, grape seed extract, grape skin extract, carrot juice, bilberry, flaxseed meal, bee pollen, ginkgo biloba, primrose (evening primrose oil), red clover, burdock root, dandelion, parsley, rose hips, milk thistle, ginger, Siberian ginseng, rosemary, curcumin, garlic, lycopene, grapefruit seed extract, spinach, and broccoli. - In still other embodiments, the compositions comprise at least one vitamin (e.g., vitamin A, thiamin (B1), riboflavin (B2), pyridoxine (B6), cyanocobalamin (B12), biotin, ascorbic acid (vitamin C), retinoic acid (vitamin D), vitamin E, folic acid and other folates, vitamin K, niacin, and pantothenic acid). In some embodiments, the particles comprise at least one mineral (e.g., sodium, potassium, magnesium, calcium, phosphorus, chlorine, iron, zinc, manganese, flourine, copper, molybdenum, chromium, selenium, and iodine). In some particularly preferred embodiments, a dosage of a plurality of particles includes vitamins or minerals in the range of the recommended daily allowance (RDA) as specified by the United States Department of Agriculture. In still other embodiments, the particles comprise an amino acid supplement formula in which at least one amino acid is included (e.g., 1-carnitine or tryptophan).
- Transesterification of phosphatidylcholine (PC) under solvent free conditions has been performed by Haraldsson et al in 1999 [15], with the results of high incorporation of EPA/DHA and with the following hydrolysis profile PC/LPC/GPC=39/44/17. Extensive hydrolysis and by-product formation is generally considered a problem with transesterification reactions, resulting in low product yields. This invention discloses a process for transesterification of crude soybean lecithin (mixture of PC, PE and PI). In the first step, the lecithin is hydrolyzed using a lipase in the presence of water (pH=8). The use of a variety of lipases is contemplated, including, but not limited to, Thermomyces Lanuginosus lipase, Rhizomucor miehei lipase, Candida Antarctica lipase, Pseudomonas fluorescence lipase, and Mucor javanicus lipase. The first step takes around 24 hours and results in a product comprising predominantly of lyso-phospholipids and glycerophospholipids such as PC/LPC/GPC=0/15/85. In the second step, free fatty acids are added such as EPA and DHA, however any omega-3 fatty acid is contemplated. Next a strong vacuum is applied to the reaction vessel for 72 hours. However, the reaction length can be varied in order to obtain a composition with the desired amount of phospholipids and lyso-phospholipids. By extending the reaction time beyond 72 hours, a product comprising more than 65% phospholipids can be obtained. Next, a lipid carrier is added to the reaction mixture in order to reduce the viscosity of the solution. The added amount of triglycerides can be 10%, 20%, 30%, 40% or more, it depends on the requested viscosity of the final product. The lipid carrier can be a fish oil such as tuna oil, menhaden oil and herring oil, or any triglyceride, diglyceride, ethyl- or methylester of a fatty acid. In the final step, the product is subjected to a molecular distillation and the free fatty acids are removed, resulting in a final product comprising of phospholipids (lyso-phospholipids and phospholipids) and triglycerides in a ratio of preferably 2:1.
- This invention further discloses a process for the enzymatic transesterification/esterification of phospholipids with fatty acids alkyl esters or free fatty acids in an evacuated vessel (B). A reduced pressure is applied to the vessel B (0.001-30 mbar) and water vapor (moisture) is allowed to enter the reaction mixture through a tube from a second vessel (A) (
FIG. 5 for schematic drawing of the experimental setup). The water in vessel A is heated to 25-30° C. By adding moisture to an evacuated reaction vessel the rate of reaction could either be increased of the lipase dosage could be reduced. In addition, the reuse of the enzymes was improved. Finally, a novel marine phospholipid composition was prepared characterized by being acylated in the range of 55%-85%, having at least 5% EPA and/or DHA esterified, having a EPA/DHA ratio of at least 1. - Accordingly, in preferred embodiments, the present invention utilizes a phospholipid, preferably a phosphatide such as lecithin. The present invention is not limited to the use of any particular phospholipid. Indeed, the use of a variety of phospholipids is contemplated. In some embodiments, the phospholipid is a phosphatidic or lysophosphatidic acid. In more preferred embodiments, the phospholipid is a mixture of phosphatides such as phosphatidylcholine, phosphatidylethnolamine, phosphatidylserine and phosphatidylinositol. The present invention is not limited to the use of any particular source of phospholipids. In some embodiments, the phospholipids are from soybeans, while in other embodiments, the phospholipids are from eggs. In particularly preferred embodiments, the phospholipids utilized are commercially available, such as Alcolec 40P® from American Lecithin Company Inc (Oxford, Conn., USA). The present invention is not limited to the use of any particular enzyme. Indeed, the use of a variety of enzymes is contemplated, including, but not limited to Thermomyces Lanuginosus lipase, Rhizomucor miehei lipase, Candida Antarctica lipase, Pseudomonas fluorescence lipase, and Mucor javanicus lipase. This invention is not limited to any particular fatty acid alkyl ester either. This includes, but not limited to: decanoic acid (10:0), undecanoic acid (11:0), 10-undecanoic acid (11:1), lauric acid (12:0), cis-5-dodecanoic acid (12:1), tridecanoic acid (13:0), myristic acid (14:0), myristoleic acid (cis-9-tetradecenoic acid, 14:1), pentadecanoic acid (15:0), palmitic acid (16:0), palmitoleic acid (cis-9-hexadecenoic acid, 16:1), heptadecanoic acid (17:1), stearic acid (18:0), elaidic acid (trans-9-octadecenoic acid, 18:1), oleic acid (cis-9-octadecanoic acid, 18:1), nonadecanoic acid (19:0), eicosanoic acid (20:0), cis-11-eicosenoic acid (20:1), 11,14-eicosadienoic acid (20:2), heneicosanoic acid (21:0), docosanoic acid (22:0), erucic acid (cis-13-docosenoic acid, 22:1), tricosanoic acid (23:0), tetracosanoic acid (24:0), nervonic acid (24:1), pentacosanoic acid (25:0), hexacosanoic acid (26:0), heptacosanoic acid (27:0), octacosanoic acid (28:0), nonacosanoic acid (29:0), triacosanoic acid (30:0), vaccenic acid (t-11-octadecenoic acid, 18:1), tariric acid (octadec-6-ynoic acid, 18:1), and ricinoleic acid (12-hydroxyoctadec-cis-9-enoic acid, 18:1) and ω3, ω6, and ω9 fatty acyl residues such as 9,12,15-octadecatrienoic acid (α-linolenic acid) [18:3, ω3]; 6,9,12,15-octadecatetraenoic acid (stearidonic acid) [18:4, ω3]; 11,14,17-eicosatrienoic acid (dihomo-α-linolenic acid) [20:3, ω3]; 8,11,14,17-eicosatetraenoic acid [20:4, ω3], 5,8,11,14,17-eicosapentaenoic acid [20:5, ω3]; 7,10,13,16,19-docosapentaenoic acid [22:5, ω3]; 4,7,10,13,16,19-docosahexaenoic acid [22:6, ω3]; 9,12-octadecadienoic acid (linoleic acid) [18:2, ω6]; 6,9,12-octadecatrienoic acid (γ-linolenic acid) [18:3, ω6]; 8,11,14-eicosatrienoic acid (dihomo-γ-linolenic acid) [20:3 ω6]; 5,8,11,14-eicosatetraenoic acid (arachidonic acid) [20:4, ω6]; 7,10,13,16-docosatetraenoic acid [22:4, ω6]; 4,7,10,13,16-docosapentaenoic acid [22:5, ω6]; 6,9-octadecadienoic acid [18:2, ω9]; 8,1 1-eicosadienoic acid [20:2, ω9]; 5,8,1 1-eicosatrienoic acid (Mead acid) [20:3, ω9]; t10,c12 octadecadienoic acid; c10,t12 octadecadienoic acid; c9,t11 octadecadienoic acid; and t9,c11 octadecadienoic acid. Moreover, acyl residues may be conjugated, hydroxylated, epoxidated or hydroxyepoxidated acyl residues.
- Marine phospholipids extracted from marine sources have a characteristic smell and taste of rancid fish. The GC profile of the volatiles confirms the presence of these degradation products, such as short chain aldehydes and carboxylic acids. In preferred embodiments, the synthetic marine phospholipid compositions of the present invention are substantially free of volatile organic compounds and are therefore much more suitable as a food supplement for humans and animals. Accordingly, in preferred compositions, the present invention provides synthetic marine phospholipids compositions having high or increased palatability, wherein the high or increased palatability is due to low levels of organic solvents and/or volatile organic compounds. In preferred embodiments, palatability is assayed by feeding the composition to a panel of subjects, preferably human. In more preferred embodiments, the phospholipids compositions have high or increased palatability as compared to naturally extracted marine phospholipids. In other preferred embodiments, the synthetic marine phospholipids compositions of the present invention are safe for oral administration.
- Experimental
- The difference in bioavailability and bioefficacy between the marine lipid composition of the present invention and a fish oil were investigated in a rat experiment. The rat feed was prepared using AIN-93 except that soybean oil was removed from the feed. The pelleted AIN-93 diet was ground and the marine lipid compositions (
PL 1 and PL 2) as well as fish oil (TG oil) and control were added to this ground feed. The marine lipid compositions were prepared using enzymatic (lipase) catalyzed transesterification of soy lecithin with fish oil fatty acids according to the method described in Example 4, followed by the addition of a triglyceride carrier and short path distillation. The concentration of EPA, DHA and 18:3 n-3 in the different diets can be seen in the table below (table 1).TABLE 1 Amount of different fatty acid in the final feed products g/100 g g/100 g g/100 g SUM g/100 g EPA DHA 18:3n3 EPA + DHA + 18:3n3 Control T4 0 0 0.26 0.26 TG Oil T1 0.61 0.39 0.24 1.23 PL 1T2 0.61 0.35 0.26 1.22 PL 2T3 0.24 0.73 0.26 1.23 - Thirty six newly weaned male Sprague Dawley rats (start weight 168±11 g) were used in the experiment. The rats were initially given low-essential oil rat feed, containing 20 g of sunflower oil and 10 g of flaxseed oil per kg of feed, for one week. After the first week, modified AIN-93 diet powder without the test oil was given to rats ad libitum until the start of the experiment. Feeding of rats was stopped 12 hours before the sampling, 30 days after the start of feeding. Each rat was individually anesthetized with carbon dioxide, weighed and euthanized with cervical dislocation. Next, blood was sampled and centrifuged to separate plasma and blood cells. Then abdominal skin was removed and 70 ml of sterile Hepes-Hanks was injected into the peritoneal cavity to collect intraperitoneal lymphocytes. The abdomen was gently massaged for about 3 minutes after which the buffer solution was drained and centrifuged in Falcon tubes (200×g, 10 min) to collect the cells. The cells were resuspended into 1 ml of freezing fluid (10% DMSO, 90% fetal bovine serum) in 1.5 ml Eppendorf tubes. These tubes were then frozen to dry ice temperature for one hour by immersing the tubes in isopropanol placed on dry ice. This enabled a slower freezing rate than by putting the cells directly on dry ice. In the laboratory, the cells were stored overnight at −80° C. and then stored in liquid nitrogen. The derivatization of the lipids in order to perform gas chromatographic (GC) analysis was carried accordingly to [16]. The run conditions for the GC were according to [17]. The growth of rats did not differ between the feeding groups (data not shown). The intake of feed, and the intake fatty acids thereof, was monitored by keeping the rats in metabolic cages which allows the measurement of eaten and uneaten portion of feed. The
PL 1 test group consumed somewhat less EPA than the TG oil group, whereas thePL 2 and the control group consumed much less EPA than both thePL 1 and the TG oil groups (FIG. 1 ). The amount of EPA in plasma varied between the groups and the results are shownFIG. 2 . Even though the estimated intake of EPA was higher in the TG Oil group than thePL 1 group, the area % of EPA measured in plasma for PL1 was higher than for TG oil. Indicating a higher bioavailability of EPA from ofPL 1 than from the TG oil. Furthermore, this was also observed in the FA profile of the red blood cells and the monocytes (FIG. 3 andFIG. 4 , respectively). Demonstrating that thePL 1 composition was more efficient in enriching these cells with omega-3 than the TG oil group, hence being more bioefficient than TG. - The total fatty acid profile for the lipids in the brains (table 2), adipose tissue (table 3), liver (table 4), testicles (table 5) and heart (table 6) were isolated from the rats in example 1. The
PL 1 composition increases the DHA content in brain and adipose tissue more than the TG composition. ThePL 1 composition increase the EPA content in the adipose tissue more than the TG composition. It is to be observed that the PL1 composition increases the EPA/DHA content in the phospholipids and in the total lipids of the different tissues as well as reduces the AA/EPA ratio more than the TG oil composition.TABLE 2 Fatty acid profile of the total lipids isolated from the rat brain (mmol/g lipids). 18:1 18:2 18:3 n3 20:3 ARA EPA 22:4 22:5 n6 22:5 n3 DHA TG-oil 370 12 0.35 5.7 153.7 2.2 68.5 5.6 5.0 191.4 PL-1 399 12 0.33 7.2 160.7 2.2 70.2 7.2 5.7 203.3 PL-2 345 13 0.33 5.7 141.6 1.7 60.8 5.8 3.4 190.3 Control 363 12 0.30 5.2 174.9 0.1 81.4 7.6 1.7 189.6 -
TABLE 3 Fatty acid profile of the total lipids isolated from adipose tissue (mmol/g lipids). 18:1 18:2 18:3 n3 20:3 ARA EPA 22:4 22:5 n6 22:5 n3 DHA TG-oil 350 306 40.2 1.4 6.3 21.7 6.3 0.9 11.3 27.5 PL-1 500 236 58.2 1.9 7.0 24.0 7.0 1.0 7.2 30.1 PL-2 420 536 45.7 2.4 6.8 12.8 6.8 2.7 104.0 50.9 Control 419 113 28.2 0.8 3.8 0.3 3.8 0.6 26.1 0.8 -
TABLE 4 Fatty acid profile of the total lipids in liver (mmol/g lipids). 18:1 18:2 18:3 n3 20:3 ARA EPA 22:4 22:5 n6 22:5 n3 DHA TG-oil 304.2 514.1 31.9 13.2 278.9 147.8 2.5 2.9 49.9 272.8 PL-1 264.3 501.6 28.1 12.8 317.3 120.2 2.4 2.6 49.8 259.0 PL-2 226.6 462.7 19.9 12.3 289.0 80.4 3.0 5.5 30.4 263.1 Control 359.2 493.7 21.7 8.9 480.9 9.2 12.0 4.5 14.7 145.8 -
TABLE 5 Fatty acid profile of the total lipids in testicles. nmoles FA/mg n3 n6 20:3 lipids 18:4 20:5 18:3 18:3 22:6 16:1 20:4 18:2 22:5 20:3 n9 22:4 18:1 TG-oil 3.2 17.0 11.7 1.8 56.6 47.6 280.1 195.1 6.9 285.6 3.7 56.2 326.8 PL-1 0.6 10.8 9.2 1.7 52.4 35.6 294.9 210.1 6.1 294.2 3.4 61.3 348.3 PL-2 2.4 6.1 1.7 2.0 56.0 0.0 310.8 156.5 2.4 345.3 6.2 59.5 332.5 Control 1.8 1.1 1.6 2.0 27.2 0.0 335.5 162.2 1.3 319.1 5.8 78.7 330.1 -
TABLE 6 Fatty acid profile of the total lipids in heart. nmoles FA/mg n3 n6 20:3 lipids 18:4 20:5 18:3 18:3 22:6 16:1 20:4 18:2 22:5 20:3 n9 22:4 18:1 TG-oil 2.0 44.7 20.2 0.9 314.0 49.7 279.6 787.9 4.0 6.8 3.0 288.9 PL-1 1.5 45.0 25.6 0.8 314.4 44.6 300.4 789.0 4.4 7.2 3.3 276.6 PL-2 0.2 29.1 9.9 0.4 372.1 30.1 291.0 690.3 9.0 6.3 1.5 4.4 237.0 Control 0.0 2.9 7.2 0.7 209.9 495.8 756.2 14.7 6.2 2.4 30.3 289.5 - The fatty acid profile of the phospholipids isolated from the brain (table 7), adipose tissue (table 8), liver (table 9), testicles (table 10) and heart (Table 11) in the rats from example 1 were determined.
TABLE 7 Fatty acid profile of the phospholipids isolated from the brain (mmol/g lipids). 18:1 18:2 18:3 n3 20:3 ARA EPA 22:4 22:5 n6 22:5 n3 DHA TG-oil 260.8 9.9 0.2 4.5 147.17 1.9 72.8 5.6 5.3 196.8 PL-1 289.9 10.9 0.1 4.2 115.07 1.4 54.7 4.3 4.5 170.1 PL-2 232.6 2.1 0.6 4.2 122.38 1.2 57.4 4.4 0.7 175.9 Control 288.8 4.4 3.5 181.60 0.1 86.8 7.3 0.5 213.1 -
TABLE 8 Fatty acid profile of the phospholipids isolated from the adipose tissue (mmol/g lipids). 18:1 18:2 18:3 n3 20:3 ARA EPA 22:4 22:5 n6 22:5 n3 DHA TG-oil 0.7 0.9 0.04 0.04 0.45 0.10 0.0 0.0 0.0 0.17 PL-1 0.6 0.5 0.04 0.02 0.24 0.06 0.0 0.0 0.0 0.09 PL-2 0.5 0.6 0.03 0.04 0.37 0.05 0.0 0.0 0.0 0.13 Control 0.5 0.4 0.04 0.03 0.57 0.01 0.0 0.0 0.0 0.04 -
TABLE 9 Fatty acid profile of the phospholipids isolated from the liver (mmol/g lipids). 18:1 18:2 18:3 n3 20:3 ARA EPA 22:4 22:5 n6 22:5 n3 DHA TG-oil 84.2 165.4 4.4 11.5 256.9 61.6 1.7 1.69 20.5 186.5 PL-1 107.2 227.6 4.6 11.4 287.3 67.9 2.0 1.62 29.1 207.7 PL-2 117.0 347.7 6.6 12.3 314.7 51.7 2.7 5.12 26.0 247.7 Control 86.7 204.4 1.6 7.6 393.6 2.8 8.0 3.41 11.0 125.9 -
TABLE 10 Fatty acid profile of the phospholipids isolated from the testicles nmoles FA/mg lipids 20:5 22:6 20:4 18:2 22:5 n3 22:5 n6 22:4 18:1 TG-oil 3.18 23.00 173.10 55.30 1.55 234.24 20.50 118.11 PL-1 3.71 31.41 227.44 82.56 2.05 317.90 28.27 143.41 PL-2 3.339 46.79 335.87 127.50 2.04 265.36 45.48 155.71 Control 0.430 14.08 204.27 49.39 0.36 161.20 35.70 118.18 -
TABLE 11 Fatty acid profile of the phospholipids isolated from the heart. nmoles 20:3 FA/mg lipids 20:5 n3 18:3 22:6 20:4 18:2 22:5 20:3 n9 22:4 18:1 TG-oil 35.0 6.4 286.3 240.3 877.5 4.2 5.1 3.2 119.6 PL-1 27.5 6.0 261.6 205.1 754.6 3.4 4.2 2.6 96.3 PL-2 20.8 4.1 331.1 227.6 570.3 7.9 5.0 1.2 4.1 115.7 Control 1.6 3.1 187.1 348.8 505.9 13.7 3.2 1.9 22.0 128.6 -
TABLE 12 Fatty acid profile of the sn-2 position on the phospholipids isolated from the adipose tissue sn-2 EPA n3 18:3 DHA ARA 18:2 TG-oil 0.06 0.03 0.09 0.24 0.57 PL-1 0.09 0.04 0.12 0.29 0.70 PL-2 0.04 0.11 0.35 0.60 Control 0.00 0.04 0.45 0.49 -
TABLE 13 Fatty acid profile of the sn-2 position on the phospholipids isolated from the brain sn-2 EPA DHA ARA 18:2 22:5 20:3 22:4 18:1 TG-oil 1.6 164.7 123.6 9.8 5.0 3.9 72.8 197.4 PL-1 1.3 146.3 96.5 11.6 3.7 4.1 51.8 260.2 PL-2 1.2 173.5 120.3 3.2 4.4 4.2 57.4 223.4 Control 0.1 203.0 171.6 4.4 7.1 3.2 81.1 258.4 -
TABLE 14 Fatty acid profile of the sn-2 position of the phospholipids in the liver. n3 n6 20:3 sn-2 18:4 20:5 18:3 18:3 22:6 16:1 20:4 18:2 22:5n3 22:5 20:3 n9 22:4 18:1 20:2 TG-oil 0.5 50.2 2.3 0.3 145.3 6.9 188.6 149.5 18.6 0.9 6.4 0.5 0.9 45.1 2.7 PL-1 0.4 60.1 2.9 0.4 176.7 0.0 235.6 193.0 24.3 1.2 8.3 0.8 1.6 77.9 2.5 PL-2 0.1 45.8 4.2 0.5 212.4 22.2 264.5 343.7 25.6 3.7 9.6 0.6 2.0 87.0 2.5 Control 0.0 2.5 1.3 0.4 108.6 31.7 330.9 204.4 11.0 2.7 6.0 1.2 6.4 62.6 3.7 -
TABLE 15 Fatty acid profile of the sn-2 position of the phospholipids in the testis. sn-2 20:5 22:6 20:4 18:2 22:5 22:4 18:1 TG-oil 2.9 21.8 167.6 60.7 224.4 20.5 109.3 PL-1 3.4 28.4 210.7 77.2 290.6 28.2 120.3 PL-2 2.5 41.6 304.7 110.4 244.1 45.4 137.8 Control 0.4 13.6 202.1 50.9 157.2 35.6 113.7 -
TABLE 16 Fatty acid profile of the sn-2 position of the phospholipids in the heart sn-2 20:5 n3 18:3 22:6 20:4 18:2 22:5 20:3 20:3 n9 22:4 18:1 TG-oil 28.6 5.3 215.4 164.9 741.7 2.9 3.2 3.2 68.2 PL-1 22.2 4.3 193.4 131.5 601.8 2.1 2.2 2.5 52.5 PL-2 17.6 3.4 256.7 160.4 519.5 5.7 3.2 1.2 4.1 70.0 Control 1.4 1.9 163.7 283.9 467.0 8.4 2.9 1.9 21.9 96.0 - 50 g of soy lecithin from American Lecithin Company Inc (Oxford, Conn., USA), 40 g of TL-IM lipase from Novozymes (Bagsvaerd, Denmark) and 5 g of water (adjusted to pH=8 using NaOH) were mixed in a reaction vessel at 50° C. for 24 hours. Next, 10 g of free fatty acids containing 10% EPA and 50% DHA from Napro Pharma (Brattvaag, Norway) was added, followed by application of vacuum to the reaction vessel. After 72 hours the reaction was terminated and the phospholipid mixture was analyzed using HPLC and GC. The results showed that the relationship between PC/LPC/GPC was 65/35/0, and that the content of EPA and DHA was around 10% and 12%, respectively. Next, 20 g of sardine oil was added to the reaction mixture which comprised of 18% EPA and 12% DHA (relative GC peak area), followed by molecular distillation. The final product contained around 70% acetone insolubles, around 30% triglycerides and traces of free fatty acids.
- A marine phospholipid composition containing 8.4% EPA and 1.2% DHA was prepared using a crude soybean lecithin as a starting material according to [19]. A marine oil was added to the phospholipid mixture (30% w/w) so that the total level of EPA was 21.9% and for DHA 9.4%. Furthermore, soy lecithin and lyso-phospholipids prepared according to [20] were added to the mixture in variable amounts so that a range of PC/LPC/GPC ratios could be obtained (Table 17). By using this method, all the treatments (MPL1-MPL5) contained exactly the same amount EPA and DHA. Lipid compositions were consumed as a single bolus by adult Sprague-Dawley rats and the appearance of EPA/DHA in blood was measured at different time points from 1 to 12 hours after ingestion. The concentration of EPA/DHA was determined using GC-FID and reported as area percentage.
FIGS. 6 and 7 show that composition MPL2 and MPL3 results in the highest concentration of EPA/DHA in plasma after uptake. Comparing the surface area under each curve it is clear that MPL2 and MPL3 demonstrates a higher bioavailability of EPA/DHA than the other composition MPL1, MPL4 and MPL5.TABLE 17 Hydrolysis profile of the compositions tested Treatment MPL1 MPL2 MPL3 MPL4 MPL5 PC/LPC/GPC 85/15/0 70/30/0 55/45/0 40/60/0 47/37/16 - Marine phospholipids were prepared using either 40% soy PC (American Lecithin Company Inc, Oxford, Conn., USA) (MPL1) or 96% pure soy PC (Phospholipid GmbH, Köln, Germany) (MPL2) according to a method described by others [4]. Fatty acid content and the level of bi-products are shown in table 18. The MPL treatments consisted of a mixture of phospholipids, lyso-phospholipids and glycerol-phospholipids. Looking only at the PC/LPC/GPC relationship, it was 64/33/2 and 42/40/18 for MPL1 and MPL2, respectively. Finally, all three treatments were emulsified into skimmed milk.
TABLE 18 Composition of the phospholipids used in example 2 PC/LPC/ 18:2 Composition GPC (n − 6) 18:3 (n − 3) EPA DHA MPL1 64/33/2 129 mg/ g 9 mg/g 51 mg/g 171 mg/g MPL2 42/40/18 124 mg/ g 9 mg/g 96 mg/g 96 mg/g - 18 newly weaned Sprague-Dawley rats were fed the milk emulsions for 1 week. Each rat was placed in its own cage to ensure that they got an even amount of test substance and the milk was consumed by the rat pups ad libitum. After 1 week the experiment was terminated and the rats were decapitated. The animals were kept without food for 24 hours before sampling. Entire livers were collected and frozen immediately using liquid nitrogen (stored at −65° C.). Total RNA was isolated from the liver samples according to the Quiagen Rnaesy Midi Kit Protocol. The RNA samples were quantified and quality measured by NanoDrop and Bioanalyzer. The isolated RNA was hybridized onto a gene chip RAE230 2.0 from Affymetrix (Santa Clara, Calif., USA). The expression level of each gene was measured using an Affymetrix GeneChip 3000 7G scanner. The results were suitable for all chips except 2 and they were excluded from the trial. Using statistical tools a list of genes expressed differentially between MPL1 versus MPL2 (Table 3) was obtained. The results are based on (log) probe set summary expression measures, computed by RMA, and linear models are fitted using Empirical Bayes methods for borrowing strength across genes (using the Limma package in R). The p-value are adjusted for multiple testing using the Benjamini-Hochberg-method, controlling the False Discovery Rate (FDR), where FDR=the proportion of null-hypotheses of no DE that are falsely rejected.
- It was observed that MPL1 and MPL2 are biologically different compounds due to the fact that over 40 genes were differentially expressed (table 19).
TABLE 19 List of genes differentially expressed (DE) by MPL1 versus MPL2. The list is sorted according to increasing p-values. SLR: Estimated signal log-ratio (<0: down regulated gene, >0: up regulated gene). Fold change: Estimated fold change corresponding to the parameter (<1: down regulated gene, >1: up regulated gene). Affy fold change: Estimated fold change using the Affymetrix definition (<−1: down regulated gene, >1: up regulated gene) df: Degrees of freedom (= number of arrays − number of estimated parameters). Fold Affy Gene-name t p-value FDR SLR change Fold.change df Wiskott-Aldrich syndrome-like 8.35 0.00000 0.00261 1.02 2.02 2.02 14 Similar to osteoclast inhibitory lectin 7.40 0.00000 0.00392 1.49 2.80 2.80 14 neuron-glia-CAM-related cell adhesion −7.14 0.00000 0.00392 −0.55 0.68 −1.46 14 molecule dehydrodolichyl diphosphate synthase 7.09 0.00000 0.00392 0.57 1.49 1.49 14 (predicted) casein kinase II, alpha 1 polypeptide 7.05 0.00000 0.00392 1.47 2.77 2.77 14 similar to cisplatin resistance-associated 7.03 0.00000 0.00392 0.94 1.92 1.92 14 overexpressed protein (predicted) similar to Retinoblastoma-binding protein 2 7.01 0.00000 0.00392 0.67 1.59 1.59 14 (RBBP-2) serine/threonine kinase 25 (STE20 homolog, 6.90 0.00000 0.00392 0.43 1.35 1.35 14 yeast) Delangin (predicted) 6.89 0.00000 0.00392 0.69 1.61 1.61 14 similar to Hypothetical protein MGC30714 −6.80 0.00000 0.00401 −0.40 0.76 −1.32 14 myeloid/lymphoid or mixed-lineage leukemia 5 6.77 0.00000 0.00401 0.68 1.60 1.60 14 (trithorax homolog, Drosophila) (predicted) Radixin 6.76 0.00000 0.00401 0.74 1.67 1.67 14 similar to myocyte enhancer factor 2C 6.70 0.00000 0.00420 0.60 1.52 1.52 14 WD repeat and FYVE domain containing 1 6.64 0.00000 0.00443 0.80 1.74 1.74 14 (predicted) similar to Retinoblastoma-binding protein 2 6.55 0.00000 0.00509 0.87 1.82 1.82 14 (RBBP-2) zinc and ring finger 1 (predicted) −6.50 0.00000 0.00528 −0.51 0.70 −1.42 14 pumilio 1 (Drosophila) (predicted) 6.45 0.00000 0.00535 0.59 1.51 1.51 14 leukocyte receptor cluster (LRC) member 8 6.44 0.00000 0.00535 1.06 2.08 2.08 14 (predicted) Similar to collapsin response mediator protein- −6.40 0.00000 0.00542 −0.52 0.70 −1.43 14 2A SWI/SNF related, matrix associated, actin 6.37 0.00000 0.00542 0.74 1.67 1.67 14 dependent regulator of chromatin, subfamily a, member 4 B-cell CLL/lymphoma 7C (predicted) 6.37 0.00000 0.00542 0.51 1.42 1.42 14 synaptic vesicle glycoprotein 2b 6.33 0.00000 0.00568 0.97 1.96 1.96 14 leptin receptor overlapping transcript −6.31 0.00000 0.00570 −0.58 0.67 −1.50 14 Transcribed locus 6.22 0.00001 0.00658 0.44 1.36 1.36 14 similar to mKIAA1321 protein 6.20 0.00001 0.00658 0.99 1.99 1.99 14 retinol binding protein 2, cellular 6.18 0.00001 0.00660 0.59 1.51 1.51 14 similar to Safb2 protein 6.16 0.00001 0.00660 0.62 1.53 1.53 14 similar to Zbtb20 protein 6.15 0.00001 0.00660 0.49 1.40 1.40 14 phosphofructokinase, liver, B-type 6.13 0.00001 0.00665 0.67 1.60 1.60 14 Transcription elongation factor B (SIII), 6.10 0.00001 0.00668 0.43 1.35 1.35 14 polypeptide 3 Echinoderm microtubule associated protein like 6.10 0.00001 0.00668 1.00 2.00 2.00 14 4 (predicted) DNA topoisomerase I, mitochondrial 6.08 0.00001 0.00678 0.61 1.53 1.53 14 ectonucleoside triphosphate diphosphohydrolase 5 5.96 0.00001 0.00849 0.74 1.67 1.67 14 nuclear factor I/X 5.94 0.00001 0.00859 0.65 1.57 1.57 14 WW domain binding protein 4 5.90 0.00001 0.00914 0.58 1.50 1.50 14 Acetyl-coenzyme A acetyltransferase 1 5.84 0.00001 0.00967 0.69 1.61 1.61 14 similar to THO complex 2 5.84 0.00001 0.00967 0.74 1.67 1.67 14 - MPL2 regulates 401 genes versus the control (table 20). A number of genes listed are involved maintenance of the cell, in transcription and protein synthesis as well as signaling pathways. Others are involved in regulation of metabolism and the inflammatory response such as Tnf receptor-associated factor 6 (Traf6_predicted) (fold change of 0.53), guanine nucleotide binding protein alpha inhibiting 2 (Gnai2) (fold change of 0.6, gamma-butyrobetaine hydroxylase (Bbox1) (fold change of 1.32), monoglyceride lipase (Mg11) (fold change 0.52), nuclear NF-kappaB activating protein (fold change 0.65) and CCAAT/enhancer binding protein (C/EBP) (fold change of 0.66). The data listed in table 4 show that a omega-3 rich phospholipid with an EPA/DHA ratio of 2:1 behaves differently compared to placebo. A phospholipid composition with an EPA/DHA ratio o 1:1 did not show any difference versus placebo on gene expression. Consequently, the EPA/DHA ratio is important and should preferably be 2:1.
TABLE 20 List of genes differentially expressed (DE) by MPL2 versus control. SLR: Estimated signal log-ratio (<0: down regulated gene, >0: up regulated gene). Fold change: Estimated fold change corresponding to the parameter (<1: down regulated gene, >1: up regulated gene). Affy fold change: Estimated fold change using the Affymetrix definition (<−1: down regulated gene, >1: up regulated gene) df: Degrees of freedom (= number of arrays − number of estimated parameters) Fold Affy Gene-ID Gene name t p-value FDR SLR change Fold.change df 1367588_a_at ribosomal protein L13A −5.22 0.00005 0.00568 −0.44 0.74 −1.36 14 1367844_at guanine nucleotide binding −5.47 0.00003 0.00417 −0.54 0.69 −1.46 14 protein, alpha inhibiting 2 1367958_at abl-interactor 1 −6.42 0.00000 0.00119 −0.69 0.62 −1.62 14 1367971_at protein tyrosine phosphatase −6.01 0.00001 0.00190 −0.36 0.78 −1.28 14 4a2 1368057_at ATP-binding cassette, sub- −5.06 0.00007 0.00688 −0.54 0.69 −1.45 14 family D (ALD), member 3 1368405_at v-ral simian leukemia viral −4.86 0.00011 0.00913 −0.44 0.74 −1.36 14 oncogene homolog A (ras related) 1368646_at mitogen-activated protein 4.98 0.00008 0.00772 0.70 1.62 1.62 14 kinase 9 1368649_at dyskeratosis congenita 1, −6.73 0.00000 0.00080 −0.53 0.69 −1.44 14 dyskerin 1368662_at ring finger protein 39 −7.01 0.00000 0.00053 −0.61 0.66 −1.52 14 1368703_at enigma homolog −5.38 0.00003 0.00450 −0.76 0.59 −1.70 14 1368824_at caldesmon 1 −7.18 0.00000 0.00043 −1.00 0.50 −2.00 14 1368841_at transcription factor 4 −4.94 0.00009 0.00828 −0.38 0.77 −1.31 14 1368867_at GERp95 −7.83 0.00000 0.00019 −0.85 0.56 −1.80 14 1369094_a_at protein tyrosine phosphatase, −7.22 0.00000 0.00042 −0.97 0.51 −1.96 14 receptor type, D 1369127_a_at prostaglandin F receptor 4.85 0.00011 0.00921 0.45 1.37 1.37 14 1369174_at SMAD, mothers against DPP −5.19 0.00005 0.00581 −0.38 0.77 −1.30 14 homolog 1 (Drosophila) 1369227_at Choroidermia 5.04 0.00007 0.00718 0.47 1.39 1.39 14 1369249_at progressive ankylosis homolog 5.36 0.00004 0.00467 0.48 1.39 1.39 14 (mouse) 1369501_at zinc finger protein 260 5.17 0.00005 0.00595 0.41 1.33 1.33 14 1369517_at pleckstrin homology, Sec7 and 4.93 0.00009 0.00829 0.48 1.40 1.40 14 coiled/coil domains 1 1369546_at butyrobetaine (gamma), 2- 4.96 0.00009 0.00811 0.40 1.32 1.32 14 oxoglutarate dioxygenase 1 (gamma-butyrobetaine hydroxylase) 1369628_at synaptic vesicle glycoprotein −7.20 0.00000 0.00042 −1.11 0.46 −2.15 14 2b 1369689_at N-ethylmaleimide sensitive 6.22 0.00001 0.00155 0.66 1.58 1.58 14 fusion protein 1369736_at epithelial membrane protein 1 5.74 0.00002 0.00286 0.62 1.54 1.54 14 1369775_at nuclear ubiquitous casein −7.56 0.00000 0.00027 −0.79 0.58 −1.73 14 kinase and cyclin-dependent kinase substrate 1370184_at cofilin 1 −6.07 0.00001 0.00178 −0.38 0.77 −1.30 14 1370260_at adducin 3 (gamma) −5.50 0.00003 0.00399 −0.76 0.59 −1.70 14 1370328_at Dickkopf homolog 3 (Xenopus 4.80 0.00012 0.00964 0.59 1.51 1.51 14 laevis) 1370717_at AP1 gamma subunit binding 6.00 0.00001 0.00192 0.58 1.50 1.50 14 protein 1 1370831_at monoglyceride lipase −5.47 0.00003 0.00414 −0.94 0.52 −1.92 14 1370901_at similar to hypothetical protein −4.83 0.00012 0.00948 −0.34 0.79 −1.27 14 MGC36831 (predicted) 1370946_at nuclear factor I/X −10.64 0.00000 0.00002 −1.17 0.45 −2.25 14 1370949_at myristoylated alanine rich −7.58 0.00000 0.00026 −1.17 0.44 −2.26 14 protein kinase C substrate 1370993_at laminin, gamma 1 6.13 0.00001 0.00171 0.63 1.54 1.54 14 1371034_at one cut domain, family −5.65 0.00002 0.00327 −1.77 0.29 −3.40 14 member 1 1371059_at protein kinase, cAMP- 5.24 0.00005 0.00556 0.48 1.40 1.40 14 dependent, regulatory, type 2, alpha 1371345_at methyl-CpG binding domain −5.32 0.00004 0.00491 −0.34 0.79 −1.27 14 protein 3 (predicted) 1371361_at similar to tensin −7.21 0.00000 0.00042 −0.60 0.66 −1.51 14 1371394_x_at similar to Ab2-143 −5.11 0.00006 0.00645 −0.63 0.64 −1.55 14 1371397_at nitric oxide synthase −5.53 0.00002 0.00383 −0.34 0.79 −1.26 14 interacting protein (predicted) 1371428_at −5.76 0.00001 0.00276 −0.37 0.77 −1.29 14 1371430_at dystroglycan 1 −5.46 0.00003 0.00417 −0.62 0.65 −1.53 14 1371432_at −4.95 0.00009 0.00811 −0.36 0.78 −1.28 14 1371452_at bone marrow stromal cell- −5.05 0.00007 0.00705 −0.46 0.73 −1.37 14 derived ubiquitin-like protein 1371573_at ribosomal protein L36a −5.90 0.00001 0.00221 −0.40 0.76 −1.32 14 (predicted) 1371589_at Ubiquitin-Like 5 Protein −5.28 0.00004 0.00518 −0.57 0.68 −1.48 14 1371590_s_at Ubiquitin-Like 5 Protein −4.94 0.00009 0.00829 −0.39 0.76 −1.31 14 1371779_at sorting nexin 6 (predicted) 5.64 0.00002 0.00329 0.63 1.55 1.55 14 1371826_at Transcribed locus −5.58 0.00002 0.00359 −0.48 0.72 −1.39 14 1371896_at growth arrest and DNA- −6.02 0.00001 0.00189 −0.43 0.74 −1.35 14 damage-inducible, gamma interacting protein 1 (predicted) 1371918_at CD99 −5.35 0.00004 0.00476 −0.37 0.77 −1.29 14 1372057_at CDNA clone MGC: 124976 −6.12 0.00001 0.00173 −0.38 0.77 −1.30 14 IMAGE: 7110947 1372137_at biogenesis of lysosome-related −6.03 0.00001 0.00187 −0.41 0.75 −1.32 14 organelles complex-1, subunit 1 (predicted) 1372142_at arsA arsenite transporter, ATP- −4.93 0.00009 0.00829 −0.37 0.77 −1.30 14 binding, homolog 1 (bacterial) (predicted) 1372236_at Similar to Caspase recruitment −4.90 0.00010 0.00871 −0.36 0.78 −1.29 14 domain protein 4 1372469_at Transcribed locus −4.84 0.00011 0.00945 −0.36 0.78 −1.28 14 1372697_at mitochondrial ribosomal −5.70 0.00002 0.00299 −0.58 0.67 −1.49 14 protein S15 1373031_at tripartite motif protein 8 −5.13 0.00006 0.00628 −0.44 0.74 −1.36 14 (predicted) 1373105_at interleukin 1 receptor-like 1 −5.01 0.00008 0.00742 −0.37 0.77 −1.30 14 ligand (predicted) 1373135_at similar to hypothetical protein −5.30 0.00004 0.00503 −0.55 0.68 −1.46 14 MGC2744 1373206_at similar to FAD104 (predicted) 6.73 0.00000 0.00080 0.64 1.56 1.56 14 1373303_at similar to mKIAA3013 protein −5.28 0.00004 0.00514 −0.48 0.72 −1.39 14 1373347_at DMT1-associated protein −6.18 0.00001 0.00162 −0.73 0.60 −1.66 14 1373378_at ATP/GTP binding protein 1 5.39 0.00003 0.00449 0.51 1.42 1.42 14 (predicted) 1373804_at Forkhead box P1 (predicted) −5.28 0.00004 0.00518 −0.59 0.66 −1.51 14 1373885_at chromobox homolog 5 −5.94 0.00001 0.00208 −1.04 0.48 −2.06 14 (Drosophila HP1a) (predicted) 1374002_at −6.78 0.00000 0.00074 −0.86 0.55 −1.82 14 1374283_at fetal Alzheimer antigen −7.44 0.00000 0.00032 −0.74 0.60 −1.67 14 (predicted) 1374425_at transducin-like enhancer of −4.91 0.00010 0.00849 −0.40 0.76 −1.32 14 split 1, homolog of Drosophila E(spl) (predicted) 1374509_at Similar to RIKEN cDNA −5.62 0.00002 0.00337 −0.47 0.72 −1.39 14 1110018O08 1374511_at 5.60 0.00002 0.00345 0.55 1.47 1.47 14 1374657_at Transcribed locus −4.88 0.00010 0.00890 −0.34 0.79 −1.27 14 1374733_at symplekin (predicted) −5.04 0.00007 0.00716 −0.36 0.78 −1.28 14 1374772_at similar to Chromosome 13 5.18 0.00005 0.00581 0.46 1.38 1.38 14 open reading frame 21 1374837_at B-cell CLL/lymphoma 7C −8.92 0.00000 0.00006 −0.71 0.61 −1.63 14 (predicted) 1374851_at similar to RIKEN cDNA −4.89 0.00010 0.00879 −0.39 0.76 −1.31 14 2810405O22 (predicted) 1374852_at hypothetical LOC362592 −5.20 0.00005 0.00579 −0.37 0.78 −1.29 14 1375214_at UDP-N-acetyl-alpha-D- −5.31 0.00004 0.00500 −0.58 0.67 −1.50 14 galactosamine:polypeptide N- acetylgalactosaminyltransferase 2 (predicted) 1375335_at heat shock 90 kDa protein 1, −5.26 0.00004 0.00538 −0.55 0.68 −1.46 14 beta 1375396_at pumilio 1 (Drosophila) −10.05 0.00000 0.00003 −0.92 0.53 −1.89 14 (predicted) 1375421_a_at praja 2, RING-H2 motif −6.51 0.00000 0.00102 −0.60 0.66 −1.52 14 containing 1375453_at −12.32 0.00000 0.00000 −1.02 0.49 −2.02 14 1375469_at SWI/SNF related, matrix −7.97 0.00000 0.00017 −0.93 0.53 −1.90 14 associated, actin dependent regulator of chromatin, subfamily a, member 4 1375533_at vestigial like 4 (Drosophila) −5.30 0.00004 0.00505 −0.61 0.66 −1.52 14 (predicted) 1375548_at similar to RIKEN cDNA −5.64 0.00002 0.00328 −0.58 0.67 −1.50 14 4732418C07 (predicted) 1375621_at −7.05 0.00000 0.00051 −0.96 0.51 −1.95 14 1375632_at similar to 60S ribosomal −4.85 0.00011 0.00921 −0.29 0.82 −1.22 14 protein L38 1375650_at bromodomain containing 4 −6.64 0.00000 0.00088 −0.48 0.71 −1.40 14 (predicted) 1375658_at Transcribed locus −5.00 0.00008 0.00756 −0.44 0.74 −1.35 14 1375696_at interferon (alpha and beta) 4.81 0.00012 0.00958 0.59 1.51 1.51 14 receptor 1 (predicted) 1375703_at myeloid/lymphoid or mixed- −10.20 0.00000 0.00003 −1.02 0.49 −2.03 14 lineage leukemia 5 (trithorax homolog, Drosophila) (predicted) 1375706_at −5.01 0.00008 0.00743 −0.49 0.71 −1.40 14 1375763_at similar to 2700008B19Rik −7.08 0.00000 0.00050 −0.54 0.69 −1.45 14 protein 1375958_at −5.13 0.00006 0.00628 −0.65 0.64 −1.57 14 1376059_at 5.33 0.00004 0.00483 0.35 1.28 1.28 14 1376256_at WD repeat and FYVE domain −9.16 0.00000 0.00005 −1.10 0.47 −2.15 14 containing 1 (predicted) 1376299_at similar to Retinoblastoma- −9.22 0.00000 0.00005 −0.89 0.54 −1.85 14 binding protein 2 (RBBP-2) 1376450_at transmembrane protein 5 −6.26 0.00001 0.00147 −0.55 0.68 −1.46 14 (predicted) 1376523_at At rich interactive domain 4A −5.53 0.00002 0.00383 −0.77 0.59 −1.70 14 (Rbp1 like) (predicted) 1376524_at hypothetical protein Dd25 −6.69 0.00000 0.00082 −0.66 0.63 −1.58 14 1376532_at similar to FAD104 (predicted) 6.06 0.00001 0.00178 0.56 1.47 1.47 14 1376728_at Transcribed locus −4.80 0.00012 0.00966 −0.35 0.78 −1.27 14 1376917_at zinc finger protein 292 −5.21 0.00005 0.00571 −0.66 0.63 −1.58 14 1376982_at Transcribed locus −5.49 0.00003 0.00405 −0.45 0.73 −1.37 14 1377105_at −6.97 0.00000 0.00056 −0.89 0.54 −1.85 14 1377302_a_at methylmalonic aciduria −5.10 0.00006 0.00660 −0.52 0.70 −1.43 14 (cobalamin deficiency) type A (predicted) 1377524_at similar to CG18661-PA −5.36 0.00003 0.00465 −0.43 0.74 −1.35 14 (predicted) 1377663_at ras homolog gene family, −5.00 0.00008 0.00756 −0.87 0.55 −1.82 14 member E 1377683_at similar to hypothetical protein −6.63 0.00000 0.00088 −0.56 0.68 −1.47 14 FLJ13045 (predicted) 1377728_at LOC499567 −5.45 0.00003 0.00419 −1.03 0.49 −2.04 14 1377766_at Transcribed locus 4.80 0.00012 0.00964 0.37 1.29 1.29 14 1377899_at similar to RIKEN cDNA −4.99 0.00008 0.00760 −0.46 0.73 −1.38 14 2810025M15 (predicted) 1377906_at DEAH (Asp-Glu-Ala-His) box −4.82 0.00012 0.00950 −0.73 0.60 −1.66 14 polypeptide 36 (predicted) 1377914_at serine/arginine repetitive −6.41 0.00000 0.00120 −0.98 0.51 −1.97 14 matrix 1 (predicted) 1378155_at similar to KIAA1096 protein −5.68 0.00002 0.00313 −0.89 0.54 −1.86 14 1378163_at Transcribed locus −4.86 0.00011 0.00913 −0.78 0.58 −1.71 14 1378170_at Transcribed locus −5.00 0.00008 0.00756 −0.92 0.53 −1.90 14 1378194_a_at rap2 interacting protein x −4.82 0.00012 0.00950 −0.72 0.61 −1.65 14 1378361_at chromodomain helicase DNA −7.32 0.00000 0.00039 −0.73 0.60 −1.66 14 binding protein 7 (predicted) 1378453_at −4.84 0.00011 0.00938 −0.74 0.60 −1.66 14 1378504_at Insulin-like growth factor I −5.41 0.00003 0.00440 −0.96 0.51 −1.95 14 mRNA, 3′ end of mRNA 1378786_at Transcribed locus, weakly 4.89 0.00010 0.00879 0.33 1.25 1.25 14 similar to NP_780607.2 hypothetical protein LOC10905 [Mus musculus] 1379062_at similar to Expressed sequence −6.60 0.00000 0.00090 −1.08 0.47 −2.12 14 AU019823 1379073_at Similar to RIKEN cDNA −5.51 0.00003 0.00394 −0.49 0.71 −1.40 14 2310067G05 1379101_at DEAH (Asp-Glu-Ala-His) box −5.55 0.00002 0.00375 −0.87 0.55 −1.82 14 polypeptide 36 (predicted) 1379112_At At rich interactive domain 4A −5.70 0.00002 0.00299 −0.44 0.74 −1.35 14 (Rbp1 like) (predicted) 1379232_at TBC1D12: TBC1 domain −6.98 0.00000 0.00056 −1.40 0.38 −2.63 14 family, member 12 (predicted) 1379330_s_at CDNA clone IMAGE: 7316839 −4.80 0.00012 0.00967 −0.36 0.78 −1.28 14 1379332_at Transcribed locus, strongly −4.88 0.00010 0.00886 −0.61 0.66 −1.52 14 similar to XP_417265.1 PREDICTED: similar to F- box-WD40 repeat protein 6 [Gallus gallus] 1379399_at similar to cDNA sequence −5.37 0.00003 0.00459 −0.42 0.75 −1.34 14 BC016188 (predicted) 1379457_at neural precursor cell expressed, −5.39 0.00003 0.00449 −0.56 0.68 −1.48 14 developmentally down- regulated gene 1 (predicted) 1379469_at similar to transducin (beta)-like −6.23 0.00001 0.00153 −0.91 0.53 −1.88 14 1 X-linked 1379485_at eukaryotic translation initiation −7.08 0.00000 0.00050 −1.68 0.31 −3.21 14 factor 3, subunit 10 (theta) (predicted) 1379571_at plakophilin 4 (predicted) −5.42 0.00000 0.00436 −0.74 0.60 −1.67 14 1379578_at similar to Zbtb20 protein −8.89 0.00000 0.00006 −0.71 0.61 −1.63 14 1379662_a_at SNF related kinase 4.93 0.00009 0.00829 0.36 1.29 1.29 14 1379715_at similar to CG9346-PA −4.93 0.00009 0.00829 −0.71 0.61 −1.63 14 (predicted) 1379826_at similar to hypothetical protein −5.95 0.00001 0.00208 −0.62 0.65 −1.54 14 MGC31967 1380008_at similar to Neurofilament triplet −5.11 0.00006 0.00645 −0.60 0.66 −1.52 14 H protein (20 kDa neurofilament protein) (Neurofilament heavy polypeptide) (NF-H) (predicted) 1380060_at DNA topoisomerase I, −5.23 0.00005 0.00566 −0.53 0.69 −1.44 14 mitochondrial 1380062_at membrane protein, −6.88 0.00000 0.00065 −0.75 0.59 −1.68 14 palmitoylated 6 (MAGUK p55 subfamily member 6) (predicted) 1380166_at Similar to hypothetical protein 5.63 0.00002 0.00333 0.34 1.27 1.27 14 FLJ12056 1380371_at delangin (predicted) −9.37 0.00000 0.00005 −0.94 0.52 −1.91 14 1380446_at myeloid/lymphoid or mixed- −5.00 0.00008 0.00756 −0.62 0.65 −1.54 14 lineage leukemia (trithorax homolog, Drosophila); translocated to, 10 (predicted) 1380503_at hypothetical LOC305452 −6.07 0.00001 0.00178 −0.62 0.65 −1.53 14 (predicted) 1380728_at Similar to collapsin response 6.09 0.00001 0.00178 0.49 1.41 1.41 14 mediator protein-2A 1381469_a_at PERQ amino acid rich, with −5.49 0.00003 0.00405 −0.51 0.70 −1.43 14 GYF domain 1 (predicted) 1381525_at −4.82 0.00012 0.00952 −0.41 0.75 −1.33 14 1381542_at UBX domain containing 2 −6.15 0.00001 0.00171 −0.83 0.56 −1.78 14 (predicted) 1381548_at golgi phosphoprotein 4 −5.81 0.00001 0.00256 −0.69 0.62 −1.61 14 (predicted) 1381567_at hypothetical LOC294390 4.97 0.00008 0.00800 0.36 1.29 1.29 14 (predicted) 1381764_s_at ring finger protein 126 −5.54 0.00002 0.00382 −0.51 0.70 −1.42 14 (predicted) 1381809_at ankyrin repeat domain 11 −5.94 0.00001 0.00209 −1.11 0.46 −2.17 14 (predicted) 1381829_at −6.27 0.00000 0.00145 −1.07 0.48 −2.10 14 1381878_at ubinuclein 1 (predicted) −5.82 0.00001 0.00252 −1.18 0.44 −2.26 14 1381958_at similar to mKIAA0259 protein −6.90 0.00000 0.00062 −1.27 0.41 −2.42 14 1382000_at 4.82 0.00012 0.00950 0.41 1.33 1.33 14 1382009_at Transcribed locus −5.39 0.00003 0.00449 −0.69 0.62 −1.62 14 1382027_at LOC498010 −6.28 0.00000 0.00144 −0.76 0.59 −1.70 14 1382056_at similar to splicing factor p54 −8.13 0.00000 0.00016 −0.97 0.51 −1.96 14 1382109_at nuclear NF-kappaB activating −5.83 0.00001 0.00250 −0.62 0.65 −1.53 14 protein 1382155_at 6.37 0.00000 0.00126 0.58 1.50 1.50 14 1382193_at Transcribed locus −6.07 0.00001 0.00178 −1.42 0.37 −2.67 14 1382306_at Ariadne ubiquitin-conjugating 6.59 0.00000 0.00090 0.59 1.50 1.50 14 enzyme E2 binding protein homolog 1 (Drosophila) (predicted) 1382307_at protein phosphatase 1, −4.79 0.00013 0.00976 −0.47 0.72 −1.39 14 regulatory (inhibitor) subunit 12A 1382358_at Similar to SRY (sex −5.34 0.00004 0.00482 −0.65 0.64 −1.57 14 determining region Y)-box 5 isoform a 1382372_at Aryl hydrocarbon receptor −5.07 0.00007 0.00680 −0.74 0.60 −1.67 14 1382430_at similar to KIAA1585 protein −5.62 0.00002 0.00338 −0.58 0.67 −1.50 14 (predicted) 1382434_at ectonucleoside triphosphate −5.89 0.00001 0.00229 −0.73 0.60 −1.66 14 diphosphohydrolase 5 1382466_at similar to RIKEN cDNA −5.43 0.00003 0.00433 −0.98 0.51 −1.97 14 6530403A03 (predicted) 1382551_at similar to Intersectin 2 (SH3 −6.72 0.00000 0.00080 −1.41 0.38 −2.67 14 domain-containing protein 1B) (SH3P18) (SH3P18-like WASP associated protein) 1382558_at transcription factor 3 −6.13 0.00001 0.00171 −0.62 0.65 −1.54 14 (predicted) 1382573_at Transcribed locus 5.08 0.00007 0.00677 0.38 1.30 1.30 14 1382584_at similar to mKIAA1321 protein −7.22 0.00000 0.00042 −1.15 0.45 −2.22 14 1382620_at ankyrin repeat domain 11 −9.69 0.00000 0.00003 −0.95 0.52 −1.93 14 (predicted) 1382797_at similar to 1500019C06Rik −5.02 0.00008 0.00742 −0.47 0.72 −1.39 14 protein 1382813_at similar to RIKEN cDNA −5.36 0.00004 0.00467 −0.46 0.73 −1.38 14 4930444A02 (predicted) 1382862_at Transcribed locus −6.23 0.00001 0.00153 −1.16 0.45 −2.23 14 1382904_at similar to hypothetical protein −9.04 0.00000 0.00005 −0.85 0.56 −1.80 14 DKFZp434K1421 (predicted) 1382935_at similar to Hypothetical protein −6.54 0.00000 0.00097 −0.64 0.64 −1.56 14 KIAA0141 1382939_at translocated promoter region −5.18 0.00005 0.00581 −1.13 0.46 −2.19 14 (predicted) 1382957_at similar to cisplatin resistance- −8.04 0.00000 0.00016 −1.08 0.47 −2.11 14 associated overexpressed protein (predicted) 1382960_at Transcribed locus −5.95 0.00001 0.00208 −0.77 0.59 −1.70 14 1382972_at Transcribed locus, strongly 5.17 0.00005 0.00595 0.37 1.29 1.29 14 similar to XP_226713.2 PREDICTED: similar to Src- associated protein SAW [Rattus norvegicus] 1383008_at SMC4 structural maintenance −5.19 0.00005 0.00581 −0.98 0.51 −1.97 14 of chromosomes 4-like 1 (yeast) (predicted) 1383040_a_at −5.46 0.00003 0.00419 −0.47 0.72 −1.38 14 1383052_a_at −6.54 0.00000 0.00097 −0.62 0.65 −1.53 14 1383054_at −7.86 0.00000 0.00019 −0.76 0.59 −1.70 14 1383060_at G kinase anchoring protein 1 −5.82 0.00001 0.00255 −0.44 0.74 −1.35 14 (predicted) 1383085_at Similar to Sh3bgrl protein −5.20 0.00005 0.00576 −0.86 0.55 −1.81 14 1383179_at Similar to hypothetical protein −5.10 0.00006 0.00660 −0.75 0.60 −1.68 14 HSPC129 (predicted) 1383184_at zinc and ring finger 1 5.03 0.00007 0.00731 0.39 1.31 1.31 14 (predicated) 1383334_at Transcribed locus −5.37 0.00003 0.00461 −0.46 0.73 −1.37 14 1383455_at glutamyl-prolyl-tRNA −6.80 0.00000 0.00072 −0.72 0.61 −1.65 14 synthetase (predicted) 1383535_at ankyrin repeat and SOCS box- 6.24 0.00001 0.00152 0.38 1.30 1.30 14 containing protein 8 (predicted) 1383615_a_at similar to HECT domain −6.06 0.00001 0.00178 −1.08 0.47 −2.11 14 containing 1 1383687_at −5.40 0.00003 0.00441 −0.43 0.74 −1.34 14 1383776_at Transcribed locus 6.41 0.00000 0.00120 0.62 1.54 1.54 14 1383786_at Transcribed locus −5.13 0.00006 0.00628 −0.50 0.71 −1.41 14 1383825_at radixin −9.20 0.00000 0.00005 −1.01 0.50 −2.01 14 1383827_at tousled-like kinase 1 −6.09 0.00001 0.00178 −1.25 0.42 −2.37 14 (predicted) 1384125_at myeloid/lymphoid or mixed- −5.97 0.00001 0.00202 −0.51 0.70 −1.42 14 lineage leukemia 5 (trithorax homolog, Drosophila) (predicted) 1384131_at ADP-ribosylation factor-like 6 6.10 0.00001 0.00176 0.70 1.63 1.63 14 interacting protein 2 (predicted) 1384146_at Similar to CD69 antigen (p60, −5.22 0.00005 0.00568 −1.35 0.39 −2.55 14 early T-cell activation antigen) 1384154_at WW domain binding protein 4 −5.33 0.00004 0.00483 −0.52 0.70 −1.43 14 1384260_at Transcribed locus −6.36 0.00000 0.00126 −0.66 0.63 −1.58 14 1384263_at ATP-binding cassette, sub- −7.27 0.00000 0.00040 −0.72 0.61 −1.64 14 family A (ABC1), member 13 (predicted) similar to hypothetical protein MGC33214 (predicted) 1384339_s_at casein kinase II, alpha 1 −8.81 0.00000 0.00006 −1.83 0.28 −3.56 14 polypeptide 1384376_at similar to FLJ14281 protein −5.42 0.00003 0.00436 −0.65 0.64 −1.57 14 1384394_at −7.21 0.00000 0.00042 −0.61 0.65 −1.53 14 1384609_a_at similar to RIKEN cDNA −6.61 0.00000 0.00090 −0.91 0.53 −1.87 14 B230380D07 (predicted) 1384766_a_at similar to PHD finger protein −5.18 0.00005 −0.00581 −0.70 0.61 −1.63 14 14 isoform 1 1384791_at UDP-GlcNAc:betaGal beta- −5.08 0.00006 0.00671 −0.75 0.60 −1.68 14 1,3-N- acetylglucosaminyltransferase 1 (predicted) 1384792_at formin binding protein 3 −6.70 0.00000 0.00082 −0.97 0.51 −1.96 14 (predicted) 1384857_at A kinase (PRKA) anchor −5.62 0.00002 0.00338 −1.05 0.48 −2.07 14 protein (yotiao) 9 1385006_at alpha thalassemia/mental −4.94 0.00009 0.00829 −0.45 0.73 −1.37 14 retardation syndrome X-linked homolog (human) 1385038_at similar to hedgehog-interacting −9.94 0.00000 0.00003 −0.80 0.57 −1.75 14 protein 1385076_at −5.78 0.00001 0.00271 −0.57 0.68 −1.48 14 1385077_at similar to golgi-specific −7.83 0.00000 0.00019 −1.05 0.48 −2.07 14 brefeldin A-resistance guanine nucleotide exchange factor 1 (predicted) 1385101_a_at Unknown (protein for −5.53 0.00002 0.00383 −0.97 0.51 −1.96 14 MGC: 73017) 1385108_at Transcribed locus −4.83 0.00011 0.00946 −1.27 0.42 −2.40 14 1385240_at WD repeat domain 33 −4.81 0.00012 0.00958 −0.93 0.52 −1.91 14 (predicted) 1385320_at similar to Pdz-containing −5.26 0.00004 0.00530 −0.47 0.72 −1.38 14 protein 1385407_at TCDD-inducible poly(ADP- −5.46 0.00003 0.00417 −1.34 0.39 −2.54 14 ribose) polymerase (predicted) 1385408_at similar to mKIAA0518 protein −5.86 0.00001 0.00236 −1.31 0.40 −2.49 14 1385689_at Transcribed locus −4.83 0.00011 0.00948 −0.68 0.62 −1.60 14 1385852_at CREB binding protein −4.85 0.00011 0.00927 −0.53 0.69 −1.44 14 hypothetical gene supported by NM_133381 1385931_at hook homolog 3 −7.30 0.00000 0.00040 −1.66 0.32 −3.16 14 1385999_at YME1-like 1 (S. cerevisiae) −4.80 0.00012 0.00964 −0.63 0.64 −1.55 14 1386191_a_at Transcribed locus 5.22 0.00005 0.00568 0.46 1.37 1.37 14 1386641_at Transcribed locus −5.41 0.00003 0.00440 −0.97 0.51 −1.95 14 1386793_at similar to zinc finger protein 61 −5.28 0.00004 0.00514 −0.59 0.66 −1.51 14 1387087_at CCAAT/enhancer binding −5.43 0.00003 0.00435 −0.61 0.66 −1.52 14 protein (C/EBP), beta 1387306_a_at early growth response 2 4.82 0.00012 0.00950 0.33 1.26 1.26 14 1387365_at nuclear receptor subfamily 1, −4.86 0.00011 0.00913 −0.35 0.78 −1.27 14 group H, member 3 1387415_a_at syntaxin binding protein 5 4.86 0.00011 0.00913 0.40 1.32 1.32 14 (tomosyn) 1387458_at ring finger protein 4 7.05 0.00000 0.00051 0.75 1.69 1.69 14 1387664_at ATPase, H+ transporting, V1 5.55 0.00002 0.00375 0.46 1.38 1.38 14 subunit B, isoform 2 1387757_at liver regeneration p-53 related 5.19 0.00005 0.00581 0.51 1.42 1.42 14 protein 1387760_a_at one cut domain, family −6.12 0.00001 0.00171 −1.58 0.34 −2.98 14 member 1 1387789_at v-ets erythroblastosis virus E26 −6.61 0.00000 0.00090 −0.58 0.67 −1.50 14 oncogene like (avian) 1387915_at Ratsg2 −4.79 0.00013 0.00976 −0.33 0.79 −1.26 14 1387947_at v-maf musculoaponeurotic −5.08 0.00007 0.00674 −0.80 0.57 −1.74 14 fibrosarcoma oncogene family, protein B (avian) 1388022_a_at dynamin 1-like 4.95 0.00009 0.00811 0.43 1.35 1.35 14 1388059_a_at solute carrier family 11 5.75 0.00001 0.00280 0.43 1.35 1.35 14 (proton-coupled divalent metal ion transporters), member 2 1388089_a_at ring finger protein 4 5.73 0.00002 0.00289 0.50 1.41 1.41 14 1388157_at myristoylated alanine rich −5.76 0.00001 0.00276 −0.51 0.70 −1.43 14 protein kinase C substrate 1388196_at NCK-associated protein 1 5.31 0.00004 0.00500 0.48 1.40 1.40 14 1388251_at protein kinase C, lambda 5.21 0.00005 0.00571 0.55 1.47 1.47 14 1388313_at ribosomal protein s25 −4.83 0.00011 0.00946 −0.63 0.65 −1.54 14 1388353_at proliferation-associated 2G4, −6.35 0.00000 0.00127 −0.67 0.63 −1.59 14 38 kDa 1388388_at Protein phosphatase 2, −5.32 0.00004 0.00487 −0.41 0.75 −1.33 14 regulatory subunit B (B56), delta isoform (predicted) 1388396_at serine/threonine kinase 25 −5.49 0.00003 0.00404 −0.34 0.79 −1.27 14 (STE20 homolog, yeast) 1388503_at similar to CREBBP/EP300 −6.06 0.00001 0.00178 −0.40 0.76 −1.32 14 inhibitory protein 1 1388714_at elongation factor RNA −5.88 0.00001 0.00229 −0.46 0.73 −1.37 14 polymerase II (predicted) 1388735_at Similar to keratin associated 4.84 0.00011 0.00945 0.50 1.41 1.41 14 protein 10-6 1388752_at BCL2-associated transcription −4.81 0.00012 0.00958 −0.40 0.76 −1.32 14 factor 1 (predicted) 1388849_at Protease, serine, 25 (predicted) −5.97 0.00001 0.00202 −0.50 0.71 −1.41 14 1388888_at Transcribed locus 5.13 0.00006 0.00632 0.40 1.32 1.32 14 1389268_at similar to DNA polymerase −5.21 0.00005 0.00571 −0.37 0.77 −1.29 14 lambda 1389307_at similar to Amyloid beta (A4) −4.91 0.00010 0.00854 −0.49 0.71 −1.40 14 precursor-like protein 1 1389419_at Transcribed locus −6.54 0.00000 0.00097 −1.30 0.40 −2.47 14 1389432_at pre-B-cell leukemia −4.93 0.00009 0.00829 −0.55 0.69 −1.46 14 transcription factor 2 1389444_at Transcribed locus −6.84 0.00000 0.00068 −1.06 0.48 −2.09 14 1389806_at Transcribed locus −7.63 0.00000 0.00026 −0.54 0.69 −1.46 14 1389868_at similar to RCK −6.34 0.00000 0.00128 −1.47 0.36 −2.76 14 1389963_at P55 mRNA for p55 protein −5.54 0.00002 0.00378 −0.44 0.74 −1.35 14 1389986_at LOC499304 −5.39 0.00003 0.00449 −2.57 0.17 −5.93 14 1389989_at alpha thalassemia/mental −4.93 0.00009 0.00829 −0.54 0.69 −1.45 14 retardation syndrome X-linked homolog (human) 1389998_at Nuclear receptor subfamily 2, −6.03 0.00001 0.00187 −0.69 0.62 −1.61 14 group F, member 2 1390048_at serine/arginine repetitive −5.81 0.00001 0.00256 −1.04 0.49 −2.05 14 matrix 2 (predicted) 1390120_a_at ring finger protein 1 −5.70 0.00002 0.00299 −0.36 0.78 −1.28 14 1390121_at GLIS family zinc finger 2 4.81 0.00012 0.00958 0.43 1.34 1.34 14 (predicted) 1390227_at CDNA clone IMAGE: 7300848 −5.91 0.00001 0.00219 −1.03 0.49 −2.04 14 1390360_a_at similar to Safb2 protein −4.79 0.00013 0.00976 −0.48 0.72 −1.39 14 1390410_at Transcribed locus −4.79 0.00012 0.00973 −0.49 0.71 −1.40 14 1390436_at Autophagy 7-like (S. cerevisiae) −7.88 0.00000 0.00019 −1.46 0.36 −2.76 14 (predicted) 1390448_at similar to 1110065L07Rik 5.08 0.00007 0.00674 0.32 1.25 1.25 14 protein (predicted) 1390454_at 4-nitrophenylphosphatase −5.47 0.00003 0.00415 −0.41 0.75 −1.33 14 domain and non-neuronal SNAP25-like protein homolog 1 (C. elegans) (predicted) 1390576_at Transcribed locus −5.10 0.00006 0.00660 −0.67 0.63 −1.59 14 1390660_at T-box 2 (predicted) 5.01 0.00008 0.00752 0.40 1.32 1.32 14 1390706_at spectrin beta 2 −5.55 0.00002 0.00376 −0.71 0.61 −1.64 14 1390739_at similar to zinc finger protein −5.51 0.00003 0.00395 −0.52 0.70 −1.43 14 609 similar to zinc finger protein 609 1390777_at sterol-C5-desaturase (fungal −6.59 0.00000 0.00090 −0.70 0.61 −1.63 14 ERG3, delta-5-desaturase) homolog (S. cerevisae) 1390779_at Similar to phosphoseryl-tRNA −4.86 0.00011 0.00913 −0.64 0.64 −1.56 14 kinase 1390813_at Similar to RNA-binding −5.19 0.00005 0.00581 −0.62 0.65 −1.54 14 protein Musashi2-S 1390884_a_at UDP-GlcNAc: betaGal beta- 4.87 0.00010 0.00904 0.49 1.40 1.40 14 1,3-N- acetylglucosaminyltransferase 7 (predicted) 1391021_at similar to KIAA1749 protein −7.55 0.00000 0.00027 −0.74 0.60 −1.67 14 (predicted) 1391170_at similar to mKIAA1757 protein −9.21 0.00000 0.00005 −2.01 0.25 −4.04 14 (predicted) 1391222_at similar to Nedd4 binding −5.91 0.00001 0.00219 −0.81 0.57 −1.76 14 protein 1 (predicted) 1391297_at REST corepressor 1 (predicted) −5.17 0.00005 0.00595 −0.92 0.53 −1.89 14 1391578_at −8.48 0.00000 0.00009 −1.11 0.46 −2.15 14 1391584_at Transcribed locus 6.04 0.00001 0.00185 0.45 1.37 1.37 14 1391625_at Wiskott-Aldrich syndrome-like −10.52 0.00000 0.00002 −1.28 0.41 −2.43 14 (human) 1391669_at protein tyrosine phosphatase, −6.21 0.00001 0.00156 −0.82 0.56 −1.77 14 receptor type, B (predicted) 1391689_at similar to Retinoblastoma- −9.06 0.00000 0.00005 −1.20 0.44 −2.30 14 binding protein 2 (RBBP-2) 1391701_at MYST histone −5.18 0.00005 0.00581 −0.98 0.51 −1.97 14 acetyltransferase (monocytic leukemia) 3 (predicted) 1391743_at ELAV (embryonic lethal, −4.80 0.00012 0.00964 −1.36 0.39 −2.58 14 abnormal vision, Drosophila)- like 1 (Hu antigen R) (predicted) 1391830_at copine VIII (predicted) −5.22 0.00005 0.00568 −1.08 0.47 −2.12 14 1391838_at ankyrin repeat domain 11 −7.81 0.00000 0.00019 −1.15 0.45 −2.23 14 (predicted) 1391848_at RNA binding motif protein 27 −7.02 0.00000 0.00053 −0.76 0.59 −1.70 14 (predicted) 1391968_at Similar to expressed sequence −4.89 0.00010 0.00883 −0.69 0.62 −1.62 14 AA415817 1392000_at Similar to PHD finger protein 5.01 0.00008 0.00742 0.45 1.37 1.37 14 14 isoform 1 1392061_at minichromosome maintenance 5.34 0.00004 0.00482 0.54 1.46 1.46 14 deficient 10 (S. cerevisiae) (predicted) 1392269_at transcriptional regulator, −6.23 0.00001 0.00153 −1.13 0.46 −2.19 14 SIN3A (yeast) (predicted) 1392277_at −7.29 0.00000 0.00040 −0.48 0.72 −1.40 14 1392322_at GTPase, IMAP family member 7 −4.83 0.00012 0.00948 −0.29 0.82 −1.22 14 1392472_at similar to myocyte enhancer −9.77 0.00000 0.00003 −0.88 0.54 −1.84 14 factor 2C 1392552_at similar to transcription −6.15 0.00001 0.00169 −0.96 0.51 −1.95 14 repressor p66 (predicted) 1392564_at myeloid/lymphoid or mixed- −6.13 0.00001 0.00171 −0.57 0.68 −1.48 14 lineage leukemia 5 (trithorax homolog, Drosophila) (predicted) 1392629_a_at similar to MADP-1 protein −4.93 0.00009 0.00829 −0.82 0.57 −1.77 14 (predicted) 1392738_at similar to KIAA1096 protein −5.88 0.00001 0.00231 −0.75 0.59 −1.68 14 1392825_at LOC499256 −5.20 0.00005 0.00580 −0.93 0.53 −1.90 14 1392864_at Rho GTPase activating protein −8.05 0.00000 0.00016 −1.37 0.39 −2.58 14 5 (predicted) 1392932_at leukocyte receptor cluster −4.81 0.00012 0.00958 −0.79 0.58 −1.73 14 (LRC) member 8 (predicted) 1392936_at similar to RNA binding motif −4.82 0.00012 0.00950 −0.88 0.54 −1.85 14 protein 25 1392984_at copine III (predicted) −7.83 0.00000 0.00019 −0.95 0.52 −1.93 14 1393151_at 5.03 0.00007 0.00726 0.65 1.57 1.57 14 1393226_at Transcribed locus −4.94 0.00009 0.00828 −0.73 0.60 −1.66 14 1393290_at similar to myocyte enhancer −5.65 0.00002 0.00327 −0.50 0.71 −1.42 14 factor 2C 1393322_at TAF15 RNA polymerase II, −6.18 0.00001 0.00162 −1.00 0.50 −2.00 14 TATA box binding protein (TBP)-associated factor (predicted) 1393378_at −5.72 0.00002 0.00293 −0.52 0.70 −1.43 14 1393443_a_at similar to CGI-112 protein −5.33 0.00004 0.00483 −0.47 0.72 −1.39 14 (predicted) 1393505_x_at similar to RIKEN cDNA −7.60 0.00000 0.00026 −0.69 0.62 −1.61 14 B230380D07 (predicted) 1393511_at similar to galactose-3-O- 5.10 0.00006 0.00655 0.41 1.33 1.33 14 sulfotransferase 4 1393560_at −4.91 0.00010 0.00852 −0.51 0.70 −1.42 14 1393576_at Transcribed locus −4.82 0.00012 0.00950 −0.62 0.65 −1.54 14 1393593_at similar to KIAA0597 protein 5.43 0.00003 0.00435 0.57 1.48 1.48 14 1393639_at myosin X (predicted) −4.95 0.00009 0.00811 −0.59 0.67 −1.50 14 1393790_at HRAS-like suppressor 5.44 0.00003 0.00432 0.44 1.35 1.35 14 (predicted) 1393798_at alpha thalassemia/mental −5.00 0.00008 0.00757 −0.84 0.56 −1.79 14 retardation syndrome X-linked homolog (human) 1393804_at similar to hypothetical protein −6.79 0.00000 0.00073 −0.85 0.56 −1.80 14 FLJ22490 (predicted) 1393809_at Tnf receptor-associated factor 6 −8.48 0.00000 0.00009 −0.90 0.53 −1.87 14 (predicted) 1393811_at similar to putative repair and −6.08 0.00001 0.00178 −0.79 0.58 −1.73 14 recombination helicase RAD26L 1393910_at similar to Fam13a1 protein −4.85 0.00011 0.00921 −0.81 0.57 −1.75 14 (predicted) 1393981_at similar to KIAA0423 −5.24 0.00005 0.00556 −0.57 0.68 −1.48 14 (predicted) 1394003_at similar to DNA polymerase −5.59 0.00002 0.00349 −0.59 0.67 −1.50 14 epsilon p17 subunit (DNA polymerase epsilon subunit 3) (Chromatin accessibility complex 17) (HuCHRAC17) (CHRAC-17) 1394220_at Similar to hypothetical protein 5.46 0.00003 0.00417 0.43 1.34 1.34 14 (predicted) 1394243_at similar to spermine synthase −6.11 0.00001 0.00175 −0.60 0.66 −1.51 14 1394436_at sperm associated antigen 9 −6.60 0.00000 0.00090 −0.91 0.53 −1.88 14 (predicted) 1394497_at similar to TCF7L2 protein −8.03 0.00000 0.00016 −1.06 0.48 −2.08 14 1394594_at Transcribed locus 5.09 0.00006 0.00671 0.42 1.34 1.34 14 1394715_at Dicer1, Dcr-1 homolog 5.14 0.00006 0.00627 0.54 1.46 1.46 14 (Drosophila) (predicted) 1394740_at 5.41 0.00003 0.00440 0.52 1.43 1.43 14 1394742_at Transcribed locus −5.73 0.00002 0.00289 −0.98 0.51 −1.98 14 1394746_at hect (homologous to the E6-AP −7.32 0.00000 0.00039 −0.94 0.52 −1.91 14 (UBE3A) carboxyl terminus) domain and RCC1 (CHC1)-like domain (RLD) 1 (predicted) 1394814_at translocated promoter region −6.13 0.00001 0.00171 −0.63 0.64 −1.55 14 (predicted) 1394849_at Transcribed locus −5.22 0.00005 0.00569 −1.61 0.33 −3.05 14 1394865_at Transmembrane protein 7 −7.85 0.00000 0.00019 −0.92 0.53 −1.90 14 (predicted) 1394965_at enthoprotin 5.30 0.00004 0.00503 0.40 1.32 1.32 14 1394969_at Transcribed locus 5.40 0.00003 0.00441 0.39 1.31 1.31 14 1394985_at early endosome antigen 1 −7.60 0.00000 0.00026 −1.00 0.50 −2.00 14 (predicted) 1395211_s_at supervillin (predicted) −8.74 0.00000 0.00007 −0.98 0.51 −1.97 14 1395237_at eukaryotic translation initiation −8.31 0.00000 0.00012 −0.87 0.55 −1.83 14 factor 5B 1395264_at similar to Rap1-interacting −6.85 0.00000 0.00067 −0.95 0.52 −1.93 14 factor 1 1395331_at similar to hypothetical protein 4.84 0.00011 0.00945 0.31 1.24 1.24 14 CL25084 (predicted) 1395338_at leucine-rich PPR-motif 5.24 0.00005 0.00555 0.75 1.68 1.68 14 containing (predicted) 1395516_at similar to hypothetical protein −4.89 0.00010 0.00883 −0.59 0.66 −1.51 14 FLJ10154 (predicted) 1395565_at COP9 signalosome subunit 4 5.55 0.00002 0.00376 0.40 1.32 1.32 14 1395610_at similar to Hypothetical protein 5.66 0.00002 0.00325 0.33 1.26 1.26 14 MGC30714 1395616_at similar to Ab2-008 (predicted) −5.03 0.00007 0.00729 −0.50 0.71 −1.42 14 1395625_at Transcribed locus −6.03 0.00001 0.00187 −0.76 0.59 −1.70 14 1395739_at similar to RIKEN cDNA 5.05 0.00007 0.00698 0.54 1.46 1.46 14 C920006C10 (predicted) 1395814_at Transcribed locus −5.09 0.00006 0.00663 −0.78 0.58 −1.71 14 1395976_at similar to phosphoinositol 4- −6.37 0.00000 0.00126 −0.57 0.67 −1.49 14 phosphate adaptor protein-2 1395981_at helicase, ATP binding 1 −5.76 0.00001 0.00276 −0.62 0.65 −1.54 14 (predicted) 1396036_at Ral GEF with PH domain and −6.67 0.00000 0.00084 −1.04 0.49 −2.06 14 SH3 binding motif 2 (predicted) 1396063_at DEK oncogene (DNA binding) −4.82 0.00012 0.00952 −0.63 0.65 −1.55 14 1396100_at similar to RIKEN cDNA −5.15 0.00006 0.00610 −0.56 0.68 −1.47 14 2010009L17 (predicted) 1396170_at WW domain binding protein 4 −7.78 0.00000 0.00020 −0.77 0.59 −1.71 14 1396187_at Hypothetical protein 5.14 0.00006 0.00622 0.51 1.43 1.43 14 LOC606294 1396202_at Transcribed locus 4.97 0.00008 0.00795 0.52 1.44 1.44 14 1396403_at −9.07 0.00000 0.00005 −1.01 0.50 −2.02 14 1396803_at similar to THO complex 2 −7.09 0.00000 0.00050 −0.90 0.54 −1.86 14 1397203_at PRP4 pre-mRNA processing −6.18 0.00001 0.00162 −0.67 0.63 −1.59 14 factor 4 homolog B (yeast) (predicted) 1397234_at G patch domain containing 1 −5.65 0.00002 0.00326 −0.49 0.71 −1.40 14 (predicted) 1397367_at A disintegrin and 5.05 0.00007 0.00698 0.47 1.38 1.38 14 metalloprotease domain 23 (predicted) 1397508_at similar to RIKEN cDNA −5.08 0.00006 0.00671 −0.62 0.65 −1.54 14 2310005B10 1397552_at echinoderm microtubule −8.47 0.00000 0.00009 −1.39 0.38 −2.62 14 associated protein like 4 (predicted) 1397627_at diaphanous homolog 1 −5.07 0.00007 0.00680 −0.52 0.70 −1.43 14 (Drosophila) (predicted) 1397647_at solute carrier family 25 5.51 0.00003 0.00395 0.62 1.54 1.54 14 (mitochondrial carrier; ornithine transporter) member 15 (predicted) 1397669_at Chemokine (C—C motif) 5.78 0.00001 0.00271 0.51 1.43 1.43 14 receptor 6 (predicted) 1397674_at eukaryotic translation initiation −6.44 0.00000 0.00116 −0.76 0.59 −1.69 14 factor 3, subunit 8, 110 kDa (predicted) 1397676_at Similar to osteoclast inhibitory −6.68 0.00000 0.00084 −1.34 0.39 −2.54 14 lectin 1397758_at Similar to choline −4.83 0.00011 0.00946 −0.38 0.77 −1.30 14 phosphotransferase 1; cholinephosphotransferase 1 alpha; cholinephosphotransferase 1 1397959_at similar to RIKEN cDNA −6.39 0.00000 0.00123 −1.14 0.45 −2.20 14 D130059P03 gene (predicted) 1398311_a_at kinase D-interacting substance 5.14 0.00006 0.00627 0.44 1.36 1.36 14 220 1398351_at Ubiquitin specific protease 7 −5.60 0.00002 0.00349 −0.42 0.75 −1.34 14 (herpes virus-associated) (predicted) 1398420_at Similar to E3 ubiquitin ligase −5.33 0.00004 0.00483 −0.94 0.52 −1.92 14 SMURF2 (predicted) 1398436_at ubiquitin specific protease 42 −6.36 0.00000 0.00126 −0.76 0.59 −1.69 14 (predicted) 1398486_at CDNA clone MGC: 93990 −8.09 0.00000 0.00016 −1.53 0.35 −2.89 14 IMAGE: 7115381 1398522_at similar to Ab2-034 (predicted) −4.92 0.00009 0.00832 −0.51 0.70 −1.42 14 1398553_at similar to CGI-100-like protein −6.91 0.00000 0.00062 −1.68 0.31 −3.20 14 1398834_at mitogen activated protein −4.94 0.00009 0.00828 −0.32 0.80 −1.25 14 kinase kinase 2 1398926_at prefoldin 1 (predicted) −5.95 0.00001 0.00208 −0.48 0.72 −1.40 14 1398963_at TAF10 RNA polymerase II, −5.42 0.00003 0.00436 −0.41 0.75 −1.33 14 TATA box binding protein (TBP)-associated factor (predicted) 1399099_at heterogeneous nuclear −4.94 0.00009 0.00829 −0.54 0.69 −1.46 14 ribonucleoprotein U-like 1 (predicted) 1399140_at Transcribed locus −5.16 0.00005 0.00597 −0.49 0.71 −1.40 14 AFFX-BioB- Biotin synthase −4.89 0.00010 0.00879 −0.64 0.64 −1.56 14 M_at biotin synthesis, sulfur insertion? AFFX- dethiobiotin synthetase −4.92 0.00009 0.00834 −0.70 0.62 −1.62 14 BioDn-5_at AFFX-r2-Ec- dethiobiotin synthetase −5.41 0.00003 0.00440 −0.51 0.70 −1.43 14 bioD-5_at - 60 g fatty acid ethyl ester consisting of 10% EPA and 50% DHA (FAEE 10-50), obtained from Napro Pharma (Brattvaag, Norway) and 15 g TL-IM obtained from Novozymes (Bagsvaerd, Denmark) were mixed in an evacuated round bottomed glass flask for 15 minutes. Next, N2 was released into the glass flask and the mixture was heated to 65° C. 20 g Alcolec 40P® from American Lecithin Company Inc (Oxford, Conn., USA) was then added to the reaction mixture. Alcolec 40P® is a crude soybean phospholipid product containing 40% PC, 26% phosphatidylethanolamine, 11% phosphatidylinositol, 1% phosphatidylserine, 13% phytoglycolipids as well as 14% other phosphatides (w,w). Next, the glass flask was evacuated (20-30 mbar). Finally, a second vessel containing water (30° C.), was connected to the reaction vessel through a plastic tube (
FIG. 1 ). The reduced pressure allowed moisture from the headspace of the second vessel to be added through the reaction mixture continuously. In order to obtain the final product the enzymes were removed by filtration. Finally, a triglyceride carrier was added to the product, followed by removal of the residual free fatty acids and/or esters by short path distillation. In order to analyze the product, the sample was fractionated by HPLC-UV (λ=207 nm) with a silica column and methanol-water (92:8, v/v) as mobile phase. The isolated PC+LPC fraction was then dried under nitrogen prior to derivatization; finally the fatty acid profile was determined by analyzing the derivatives using GC-FID. Furthermore, the relationship between PC, LPC and GPC was determined using HPLC with the method above, except that the UV detector was replaced by an evaporative light scattering detection (ELSD). Integrated ELSD peak areas were reported for PC/LPC/GPC (total 100%); however for simplicity other PL species were not analyzed. The results obtained for example 7 is shown in table 20 below.TABLE 20 Results obtained after transesterification using vacuum and water addition Reaction time PC/LPC/GPC* EPA/DHA** Acid value 1 day 65/31/4 3.6/2.6 43 2 days 52/45/3 5.3/4.7 55 5 days 78/22/0 5.2/4.6 65 6 days 72/26/2 6.0/5.3 75
*ELSD peak area (total 100%). Only peaks relating to PC, LPC and GPC are integrated.
**EPA/DHA attached to PC + LPC
- The enzymes from example 7 were isolated by filtration and the possibility of reuse was determined in the following experiment. 30 g FAEE (10-50), 10 g Alcolec and 15 g used enzymes (equivalent to 7.5 g enzyme because the used enzymes had absorbed product from the first reaction). The reaction was performed at 65° C. and stirred at 200 rpm using a shaker incubator. The transesterified phospholipids were analyzed as in the previous example and the results are shown in table 21 below.
TABLE 21 Results obtained with reused enzymes using incubator shaker Reaction time PC/LPC/GPC* EPA/DHA** Acid value 1 day 94/5/1 0.4/0.5 55 2 days 87/11/2 0.7/0.7 76 5 days 68/26/6 1.0/0.8 85
*ELSD peak area (total 100%). Only peaks relating to PC, LPC and GPC are integrated.
**EPA/DHA attached to PC + LPC
- The same conditions as in example 1 were used, except that the amount of lipase was 10 g. The results are shown in table 22.
TABLE 22 Results obtained with reduced lipase dosage after transesterification using vacuum and water addition Reaction time PC/LPC/GPC* EPA/DHA** Acid value 1 day 90/10/0 0.9/0.7 29 2 days 74/24/2 2.1/1.5 87 3 days 49/27/24 4.6/4.4 102 6 days 25/32/43 6.7/6.9 115
*ELSD peak area (total 100%). Only peaks relating to PC, LPC and GPC are integrated.
**EPA/DHA attached to PC + LPC
- The enzymes from example 7 were isolated by filtration and the possibility of reuse was determined in the following experiment. 30 g FAEE (10-50), 10 g Alcolec and 15 g used enzymes (equivalent to 7.5 g enzyme because the used enzymes had absorbed product from the first reaction). The reaction was performed using the same conditions as in example 3. See table 23 below for results.
TABLE 23 Reuse of enzymes from example 7 using vapor addition into evacuated reaction vessel. Reaction time PC/LPC/GPC* EPA/DHA** Acid value 1 day 79/17/4 1.0/0.9 55 2 days 59/31/10 2.8/2.6 76 3 days 52/34/14 3.8/3.5 85 6 days 37/43/20 5.6/5.7 95
*ELSD peak area (total 100%). Only peaks relating to PC, LPC and GPC are integrated.
**EPA/DHA attached the fraction consisting of PC + LPC
- The same conditions as in example 7 are applied, except that the pressure in the reaction vessel is 1 mbar. The results obtained are similar to the results in Table 23, except that the hydrolysis and the acid values are reduced. After 6 days the relationship between PC species is 80/10/0 and the acids value is 40. The incorporation of EPA/DHA is the same.
- The safety of omega-3 rich phospholipids prepared in the presence of chloroform and omega-7 rich phospholipids prepared under solvent free conditions is to be examined by feeding pregnant rats for 1 week. It is to be found that the treatment containing omega-3 rich phospholipds with traces of chloroform will result in damage to the developing fetus than the treatment containing essentially no traces of organic solvents.
- Having now fully described this invention, it will be appreciated by those skilled in the art that the same can be performed within a wide range of equivalent parameters, concentrations, and conditions without departing from the spirit and scope of the invention and without undue experimentation.
- While this invention has been described in connection with specific embodiments thereof, it will be understood that it is capable of further modifications. This application is intended to cover any variations, uses, or adaptations of the invention following, in general, the principles of the invention and including such departures from the present disclosure as come within known or customary practice within the art to which the invention pertains and as may be applied to the essential features hereinbefore set forth.
- [1] WO 2006054183
- [2] P. C. Calder. Prostaglandins, Leukotrienes and Essential Fatty Acids 2006; 75; 197-202.
- [3] U.S. Pat. No. 5,434,183
- [4] Alexander J W. Nutrition 14 (1998) 627.
- [5] Belluzi A, Boschi S, Brignola C, Munarini A, Cariani G and Miglio F. Am J Clin Nutr 71 (2000) 339.
- [6] Kremer J M. Am J Clin Nutr 71 (2000) 249
- [7] V P Carnielli, G. Verlato, F. Pederzini, I. Luijendijk, A. Boerlage, D. Pedrotti and P. Sauer Am J Clin Nutr 1998;67; 97-103.
- [8] M. Moya, E. Cortes, M. Juste, J. G. De Dios and A Vera Eur. J. Clin. Nutr. 2001; 55; 755-762.
- [9] A. Sala-Vila, A. I. Castello, C. Campoy, M. Rivero, M. Rodriguez-Palermoe and M. C. Lopez-Sabater. J. Nutr. 2004; 134; 868-873.
- [10] A. Sala-Vila, C. Campoy, A. I. Castellote, F. J. Garrido, M. Rivero, M. Rodriguez-Palmero and M. C. Lopez-Sabater. Prostaglandins, Leukotrienes and Essential Fatty Acids; 2006; 74; 143-148.
- [11] A. Valenzuela, S. Nieto, J. Sanhueza, M. J. Nunez and C. Ferrer. Annals of Nutrition & Metabolism; 2005; 49; 325-332.
- [12] J. B. Hansen, S: Grimsgaard, H. Nilsen, A. Nordoy and K. H. Bonaa. Lipids; 1998; 33; 131-138.
- [13] U.S. provisional application entitled “Functional Phospholipid Compositions” with Ser. No. 60/798,027 filed May 5, 2006.
- [14] P. C Calder, Philip C. Am. J. Clin. Nutr; 2006; 83; 1505S-1519S.
- [15] G. G. Haraldsson, A. Thorarensen, JAOCS 75 (1999) 1143-1149.
- [16] G. Lepage and C. C. Roy; J. Lipid Res; 1986; 27; 114-120.
- [17] T. Moriguchi, S-Y Lim, R. Greiner, W. Lefkowitz, J. Loewke, J. Hoshiba and N. Salem. J. Lipid Res; 2004;. 45; 1437-1445.
- [18] H. Salman, M. Bergman, H Bessler, S Alexandrova, B. Beilin, M. Djaldetti. Acta Phys Scand; 2000; 168, 431-436.
- [19] Haraldsson G G and Thorarensen A, JAOCS 75 (1999) 1143.
- [20] Samey D B, Fregapane G and Vulfson E N. JAOCS 71 (1994) 93.
Claims (26)
1. A composition comprising phospholipids having the following structure:
wherein R1 is OH or a fatty acid, R2 is OH or a fatty acid, and R3 is a mixture of H, choline, ethanolamine, inositol and serine, said phospholipid having at least 1% of DHA/EPA at positions R1 and/or R2 and from about 20-50% of OH at positions R1 and/or R2.
2. The composition as claimed in claim 1 , wherein said composition is acylated in a range from about 55% to about 85%.
3. The composition as claimed in claim 2 , wherein said composition has a ratio of EPA/DHA ranging from 1:1 to 4:1.
4. The composition of claim 2 , wherein said composition having a ratio of EPA/DHA ranging from 2:1 to 4:1.
5. The composition of claim 1 , wherein said composition further comprises a lipid carrier in a ratio of from 1:10 to 10:1 to said phospholipids.
6. The composition of claim 5 , wherein said lipid carrier is selected from the group consisting of a triglyceride, a diglyceride, an ethyl ester, and a methyl ester and combinations thereof.
7. The composition in claim 1 , wherein said composition provides higher uptake of omega-3 fatty acids into plasma as compared to natural marine phospholipids.
8. The composition in claim 1 , wherein said composition improves the AA/EPA ratio in plasma phospholipids as compared to natural marine phospholipids.
9. The composition in claim 1 , wherein said composition increases the concentration of omega-3 fatty acids in tissues as compared to natural marine phospholipids.
10. The composition in claim 1 , wherein said composition reduces the concentration of biomarkers of inflammation as compared to natural marine phospholipids.
11. A food product comprising the composition in claim 1 .
12. An animal feed comprising the composition in claim 1 .
13. A food supplement comprising the composition in claim 1 .
14. A pharmaceutical comprising the composition in claim 1 .
15. The composition of claim 5 , wherein said lipid carrier and said phospholipids are in a ratio of from about 5:1 to 1:5.
16. The composition of claim 5 , wherein said composition comprises from about 20% to about 90% of said phospholipid composition and from about 10% to about 50% of said lipid carrier.
17. A method of preparing a bioavailable omega-3 fatty acid composition comprising:
a) providing a purified phospholipid composition comprising omega-3 fatty acid residues and a purified triglyceride composition comprising omega-3 fatty acid residues;
b) combining said phospholipid composition and said triglyceride composition to form a bioavailable omega-3 fatty acid composition.
18. The method of claim 17 , further comprising the step of encapsulating said bioavailable omega-3 fatty acid composition.
19. The method of claim 17 , wherein said bioavailable omega-3 fatty acid composition has increased bioavailability as compared to purified triglycerides or phospholipids comprising omega-3 fatty acid residues.
20. The method of claim 17 , further comprising the step of packaging the bioavailable omega-3 fatty acid composition for use in functional foods.
21. The method of claim 17 , further comprising the step of assaying the bioavailable omega-3 fatty acid composition for bioavailability.
22. The method of claim 17 , further comprising administering the bioavailable omega-3 fatty acid composition to a patient.
23. A food product comprising a bioavailable omega-3 fatty acid composition made the process of claim 17 .
24. An animal feed comprising a bioavailable omega-3 fatty acid composition made the process of claim 17 .
25. A food supplement comprising a bioavailable omega-3 fatty acid composition made the process of claim 17 .
26. A pharmaceutical comprising a bioavailable omega-3 fatty acid composition made the process of claim 17.
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US11/800,229 US20080044487A1 (en) | 2006-05-05 | 2007-05-04 | Anti-inflammatory properties of marine lipid compositions |
| US12/849,950 US10117882B2 (en) | 2006-05-05 | 2010-08-04 | Anti-inflammatory properties of marine lipid compositions |
| US16/180,564 US10525068B2 (en) | 2006-05-05 | 2018-11-05 | Anti-inflammatory properties of marine lipid compositions |
| US16/734,850 US11400105B2 (en) | 2006-05-05 | 2020-01-06 | Anti-inflammatory properties of marine lipid compositions |
| US17/874,677 US20230047556A1 (en) | 2006-05-05 | 2022-07-27 | Anti-inflammatory properties of marine lipid compositions |
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US79803006P | 2006-05-05 | 2006-05-05 | |
| US79802706P | 2006-05-05 | 2006-05-05 | |
| US79802606P | 2006-05-05 | 2006-05-05 | |
| US87209606P | 2006-12-01 | 2006-12-01 | |
| US11/800,229 US20080044487A1 (en) | 2006-05-05 | 2007-05-04 | Anti-inflammatory properties of marine lipid compositions |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/849,950 Continuation US10117882B2 (en) | 2006-05-05 | 2010-08-04 | Anti-inflammatory properties of marine lipid compositions |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| US20080044487A1 true US20080044487A1 (en) | 2008-02-21 |
Family
ID=39101658
Family Applications (5)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/800,229 Abandoned US20080044487A1 (en) | 2006-05-05 | 2007-05-04 | Anti-inflammatory properties of marine lipid compositions |
| US12/849,950 Active 2028-05-13 US10117882B2 (en) | 2006-05-05 | 2010-08-04 | Anti-inflammatory properties of marine lipid compositions |
| US16/180,564 Active US10525068B2 (en) | 2006-05-05 | 2018-11-05 | Anti-inflammatory properties of marine lipid compositions |
| US16/734,850 Active 2027-11-15 US11400105B2 (en) | 2006-05-05 | 2020-01-06 | Anti-inflammatory properties of marine lipid compositions |
| US17/874,677 Pending US20230047556A1 (en) | 2006-05-05 | 2022-07-27 | Anti-inflammatory properties of marine lipid compositions |
Family Applications After (4)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/849,950 Active 2028-05-13 US10117882B2 (en) | 2006-05-05 | 2010-08-04 | Anti-inflammatory properties of marine lipid compositions |
| US16/180,564 Active US10525068B2 (en) | 2006-05-05 | 2018-11-05 | Anti-inflammatory properties of marine lipid compositions |
| US16/734,850 Active 2027-11-15 US11400105B2 (en) | 2006-05-05 | 2020-01-06 | Anti-inflammatory properties of marine lipid compositions |
| US17/874,677 Pending US20230047556A1 (en) | 2006-05-05 | 2022-07-27 | Anti-inflammatory properties of marine lipid compositions |
Country Status (1)
| Country | Link |
|---|---|
| US (5) | US20080044487A1 (en) |
Cited By (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20100010088A1 (en) * | 2007-11-01 | 2010-01-14 | Wake Forest University School Of Medicine | Compositions and Methods for Prevention and Treatment of Mammalian Diseases |
| WO2010077358A1 (en) * | 2009-01-02 | 2010-07-08 | Nestec S.A. | Methods for increasing endogenous plasmalogen levels |
| WO2011139010A1 (en) * | 2010-05-03 | 2011-11-10 | 연세대학교 산학협력단 | Composition for the prevention or treatment of obesity, dyslipidaemia, fatty liver or insulin resistance syndrome, comprising piperonal as an active ingredient |
| US8168611B1 (en) | 2011-09-29 | 2012-05-01 | Chemo S.A. France | Compositions, kits and methods for nutrition supplementation |
| US8183227B1 (en) | 2011-07-07 | 2012-05-22 | Chemo S. A. France | Compositions, kits and methods for nutrition supplementation |
| WO2012095749A1 (en) * | 2011-01-14 | 2012-07-19 | Pharma Marine As | Removal of monoglycerides from fatty acid concentrates |
| US8343753B2 (en) | 2007-11-01 | 2013-01-01 | Wake Forest University School Of Medicine | Compositions, methods, and kits for polyunsaturated fatty acids from microalgae |
| CN103355254A (en) * | 2013-06-21 | 2013-10-23 | 四川大学华西医院 | Preparation method of rhesus monkey metabolic syndrome model |
| EP2934505A4 (en) * | 2012-12-24 | 2016-11-30 | Qualitas Health Ltd | EICOSAPENTAENOIC ACID FORMULATIONS (EPA) |
| US9629820B2 (en) | 2012-12-24 | 2017-04-25 | Qualitas Health, Ltd. | Eicosapentaenoic acid (EPA) formulations |
| US10123986B2 (en) | 2012-12-24 | 2018-11-13 | Qualitas Health, Ltd. | Eicosapentaenoic acid (EPA) formulations |
| CN110538185A (en) * | 2019-10-09 | 2019-12-06 | 广东海洋大学深圳研究院 | Application of sea cucumber phospholipids in the preparation of anti-inflammatory drugs |
| US10653725B2 (en) | 2015-08-17 | 2020-05-19 | Aker Biomarine Antarctic As | Methods for improving health of farmed fish |
| JP2021502819A (en) * | 2017-11-17 | 2021-02-04 | ヒルズ・ペット・ニュートリシャン・インコーポレーテッド | Compositions containing omega-3 polyunsaturated and medium chain fatty acids |
| US11065267B2 (en) | 2017-12-21 | 2021-07-20 | Aker Biomarine Antarctic As | Lysophosphatidylcholine compositions |
| EP3958973A4 (en) * | 2019-04-26 | 2023-06-07 | Cytosolve, Inc. | COMPOSITIONS TO IMPROVE BRAIN HEALTH AND MEMORY |
| US12023359B2 (en) | 2022-11-01 | 2024-07-02 | Aker Biomarine Human Ingredients As | Phospholipid compositions for delivery of therapeutic compounds |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20080044487A1 (en) * | 2006-05-05 | 2008-02-21 | Natural Asa | Anti-inflammatory properties of marine lipid compositions |
| US8846604B2 (en) | 2011-09-02 | 2014-09-30 | Artic Nutrition AS | Lipid compositions with high DHA content |
| US9233114B2 (en) | 2013-12-05 | 2016-01-12 | Buriva, LLC | Dietary supplement containing phospholipid-DHA derived from eggs |
| US9610302B2 (en) | 2013-12-05 | 2017-04-04 | Buriva, LLC. | Composition containing phospholipid-DHA and B vitamins |
| US9549937B2 (en) | 2013-12-05 | 2017-01-24 | Burvia, LLC. | Composition containing phospholipid-DHA and folate |
| US9216199B2 (en) | 2013-12-05 | 2015-12-22 | Buriva, LLC | Nutritional supplement containing phospholipid-DHA derived from eggs |
| MX2021015311A (en) * | 2019-06-20 | 2022-02-21 | Aker Biomarine Antarctic As | Parenteral lysophosphatidylcholine formulations such as lpc-dha, lpc-epa and their use in therapy. |
| CN110564786B (en) * | 2019-08-02 | 2022-06-03 | 大渔华创(广州)海洋生物科技有限公司 | EPA/DHA type lysophospholipid composition and preparation method thereof |
| WO2023079332A1 (en) * | 2021-11-02 | 2023-05-11 | MOSSAVI, Seid Mohamad Hosein | Composition for burns, bed sores and diabetic foot ulcers |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20050130937A1 (en) * | 2003-10-22 | 2005-06-16 | Enzymotec Ltd. | Lipids containing omega-3 and omega-6 fatty acids |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| SE9101642D0 (en) | 1991-05-30 | 1991-05-30 | Kabi Pharmacia Ab | phospholipids |
| ID16442A (en) | 1996-01-22 | 1997-10-02 | Dow Chemical Co | WEIGHT POLYMER ETHYLENE ULTRA LOW MOLECULES |
| DK1417211T3 (en) | 2001-07-27 | 2007-10-08 | Neptune Technologies & Bioress | Natural marine phospholipids containing flavonoids and polyunsaturated phospholipids and their use |
| CA2587730A1 (en) | 2004-11-17 | 2006-05-26 | Natural Asa | Enzymatically synthesized marine phospholipids |
| US20080044487A1 (en) * | 2006-05-05 | 2008-02-21 | Natural Asa | Anti-inflammatory properties of marine lipid compositions |
-
2007
- 2007-05-04 US US11/800,229 patent/US20080044487A1/en not_active Abandoned
-
2010
- 2010-08-04 US US12/849,950 patent/US10117882B2/en active Active
-
2018
- 2018-11-05 US US16/180,564 patent/US10525068B2/en active Active
-
2020
- 2020-01-06 US US16/734,850 patent/US11400105B2/en active Active
-
2022
- 2022-07-27 US US17/874,677 patent/US20230047556A1/en active Pending
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20050130937A1 (en) * | 2003-10-22 | 2005-06-16 | Enzymotec Ltd. | Lipids containing omega-3 and omega-6 fatty acids |
Cited By (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8343753B2 (en) | 2007-11-01 | 2013-01-01 | Wake Forest University School Of Medicine | Compositions, methods, and kits for polyunsaturated fatty acids from microalgae |
| US20100010088A1 (en) * | 2007-11-01 | 2010-01-14 | Wake Forest University School Of Medicine | Compositions and Methods for Prevention and Treatment of Mammalian Diseases |
| WO2010077358A1 (en) * | 2009-01-02 | 2010-07-08 | Nestec S.A. | Methods for increasing endogenous plasmalogen levels |
| US9884041B2 (en) * | 2010-05-03 | 2018-02-06 | Yonsei University Technology Holdings, Inc. | Composition for the prevention or treatment of obesity, dyslipidaemia, fatty liver or insulin resistance syndrome, comprising piperonal as an active ingredient |
| WO2011139010A1 (en) * | 2010-05-03 | 2011-11-10 | 연세대학교 산학협력단 | Composition for the prevention or treatment of obesity, dyslipidaemia, fatty liver or insulin resistance syndrome, comprising piperonal as an active ingredient |
| WO2012095749A1 (en) * | 2011-01-14 | 2012-07-19 | Pharma Marine As | Removal of monoglycerides from fatty acid concentrates |
| US8183227B1 (en) | 2011-07-07 | 2012-05-22 | Chemo S. A. France | Compositions, kits and methods for nutrition supplementation |
| US8168611B1 (en) | 2011-09-29 | 2012-05-01 | Chemo S.A. France | Compositions, kits and methods for nutrition supplementation |
| US8545896B2 (en) | 2011-09-29 | 2013-10-01 | Chemo S. A. France | Compositions, kits and methods for nutrition supplementation |
| US10039734B2 (en) | 2012-12-24 | 2018-08-07 | Qualitas Health, Ltd. | Eicosapentaenoic acid (EPA) formulations |
| US10123986B2 (en) | 2012-12-24 | 2018-11-13 | Qualitas Health, Ltd. | Eicosapentaenoic acid (EPA) formulations |
| EP2934505A4 (en) * | 2012-12-24 | 2016-11-30 | Qualitas Health Ltd | EICOSAPENTAENOIC ACID FORMULATIONS (EPA) |
| US9629820B2 (en) | 2012-12-24 | 2017-04-25 | Qualitas Health, Ltd. | Eicosapentaenoic acid (EPA) formulations |
| WO2014202023A1 (en) * | 2013-06-21 | 2014-12-24 | 四川大学华西医院 | Method for preparing rhesus macaque metabolic syndrome model |
| CN103355254A (en) * | 2013-06-21 | 2013-10-23 | 四川大学华西医院 | Preparation method of rhesus monkey metabolic syndrome model |
| US10653725B2 (en) | 2015-08-17 | 2020-05-19 | Aker Biomarine Antarctic As | Methods for improving health of farmed fish |
| JP2021502819A (en) * | 2017-11-17 | 2021-02-04 | ヒルズ・ペット・ニュートリシャン・インコーポレーテッド | Compositions containing omega-3 polyunsaturated and medium chain fatty acids |
| JP7030194B2 (en) | 2017-11-17 | 2022-03-04 | ヒルズ・ペット・ニュートリシャン・インコーポレーテッド | Compositions containing omega-3 polyunsaturated and medium chain fatty acids |
| US11065267B2 (en) | 2017-12-21 | 2021-07-20 | Aker Biomarine Antarctic As | Lysophosphatidylcholine compositions |
| US12076333B2 (en) | 2017-12-21 | 2024-09-03 | Aker Biomarine Human Ingredients As | Lysophosphatidylcholine compositions |
| EP3958973A4 (en) * | 2019-04-26 | 2023-06-07 | Cytosolve, Inc. | COMPOSITIONS TO IMPROVE BRAIN HEALTH AND MEMORY |
| US12521365B2 (en) | 2019-04-26 | 2026-01-13 | Cytosolve, Inc. | Compositions for improving brain health and memory |
| CN110538185A (en) * | 2019-10-09 | 2019-12-06 | 广东海洋大学深圳研究院 | Application of sea cucumber phospholipids in the preparation of anti-inflammatory drugs |
| US12023359B2 (en) | 2022-11-01 | 2024-07-02 | Aker Biomarine Human Ingredients As | Phospholipid compositions for delivery of therapeutic compounds |
Also Published As
| Publication number | Publication date |
|---|---|
| US11400105B2 (en) | 2022-08-02 |
| US20190070203A1 (en) | 2019-03-07 |
| US20200179416A1 (en) | 2020-06-11 |
| US10117882B2 (en) | 2018-11-06 |
| US20230047556A1 (en) | 2023-02-16 |
| US20100316680A1 (en) | 2010-12-16 |
| US10525068B2 (en) | 2020-01-07 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20230047556A1 (en) | Anti-inflammatory properties of marine lipid compositions | |
| Paul et al. | Plasmalogens: A potential therapeutic target for neurodegenerative and cardiometabolic disease | |
| US11865143B2 (en) | Bioeffective krill oil compositions | |
| WO2008149177A2 (en) | Marine lipid compositions and uses thereof | |
| EP3727037B1 (en) | Lysophosphatidylcholine compositions | |
| IL176054A (en) | Oil or fat compositions containing phospholipids and long-chain polyunsaturated fatty acid supplying compound and food using the same | |
| CN105189768A (en) | Natural algae oil composition | |
| US20080058286A1 (en) | Novel applications of omega-3 rich phospholipids | |
| Wang et al. | Digestion, absorption, and metabolism characteristics of EPA-enriched phosphoethanolamine plasmalogens based on gastrointestinal functions in healthy mice | |
| AU2014256345C1 (en) | Bio effective krill oil compositions | |
| WO2025148196A1 (en) | Use of microalgae extract for promoting absorption and permeation through blood-brain barrier of fatty acid and composition thereof | |
| US20080070870A1 (en) | Use of omega-3 rich phospholipids in the area of cognitive function | |
| Zhang | Omega-3 phospholipids | |
| AU2012101335A4 (en) | Processes and products thereof | |
| HK1187809A (en) | Bioeffective krill oil compositions |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: AKER BIOMARINE ASA, NORWAY Free format text: CHANGE OF NAME;ASSIGNOR:NATURAL ASA;REEL/FRAME:019805/0418 Effective date: 20070707 Owner name: AKER BIOMARINE ASA, NORWAY Free format text: CHANGE OF NAME;ASSIGNOR:NATURAL ASA;REEL/FRAME:019805/0274 Effective date: 20070707 |
|
| STCB | Information on status: application discontinuation |
Free format text: ABANDONED -- FAILURE TO RESPOND TO AN OFFICE ACTION |