US20070173483A1 - Pyrrolopyrimidines and Related Analogs as HSP90-Inhibitors - Google Patents
Pyrrolopyrimidines and Related Analogs as HSP90-Inhibitors Download PDFInfo
- Publication number
- US20070173483A1 US20070173483A1 US11/531,218 US53121806A US2007173483A1 US 20070173483 A1 US20070173483 A1 US 20070173483A1 US 53121806 A US53121806 A US 53121806A US 2007173483 A1 US2007173483 A1 US 2007173483A1
- Authority
- US
- United States
- Prior art keywords
- dimethyl
- aryl
- heteroaryl
- group
- chloro
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Abandoned
Links
- 150000004944 pyrrolopyrimidines Chemical class 0.000 title abstract description 4
- 239000003481 heat shock protein 90 inhibitor Substances 0.000 title description 7
- 150000001875 compounds Chemical class 0.000 claims abstract description 307
- 238000000034 method Methods 0.000 claims abstract description 146
- 101710113864 Heat shock protein 90 Proteins 0.000 claims abstract description 44
- 230000001404 mediated effect Effects 0.000 claims abstract description 10
- 230000002062 proliferating effect Effects 0.000 claims abstract description 9
- -1 lower perhaloalkyl Chemical group 0.000 claims description 706
- 125000000217 alkyl group Chemical group 0.000 claims description 158
- 125000003118 aryl group Chemical group 0.000 claims description 155
- 125000001072 heteroaryl group Chemical group 0.000 claims description 135
- 229910052736 halogen Inorganic materials 0.000 claims description 97
- 150000002367 halogens Chemical class 0.000 claims description 91
- 125000004435 hydrogen atom Chemical class [H]* 0.000 claims description 82
- 229910052739 hydrogen Inorganic materials 0.000 claims description 81
- 239000001257 hydrogen Substances 0.000 claims description 80
- 125000006413 ring segment Chemical group 0.000 claims description 80
- 125000002723 alicyclic group Chemical group 0.000 claims description 73
- 125000000623 heterocyclic group Chemical group 0.000 claims description 72
- 125000003342 alkenyl group Chemical group 0.000 claims description 70
- 125000000304 alkynyl group Chemical group 0.000 claims description 70
- 125000001424 substituent group Chemical group 0.000 claims description 69
- 150000003839 salts Chemical class 0.000 claims description 67
- 229940002612 prodrug Drugs 0.000 claims description 58
- 239000000651 prodrug Substances 0.000 claims description 58
- 239000000203 mixture Substances 0.000 claims description 53
- 229910052757 nitrogen Inorganic materials 0.000 claims description 49
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 48
- 150000002148 esters Chemical class 0.000 claims description 45
- 229910052717 sulfur Inorganic materials 0.000 claims description 43
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims description 40
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims description 40
- 229910052760 oxygen Inorganic materials 0.000 claims description 39
- 125000005842 heteroatom Chemical group 0.000 claims description 38
- 239000012453 solvate Substances 0.000 claims description 35
- ZRALSGWEFCBTJO-UHFFFAOYSA-N Guanidine Chemical compound NC(N)=N ZRALSGWEFCBTJO-UHFFFAOYSA-N 0.000 claims description 32
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Thiophene Chemical compound C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 claims description 32
- SIKJAQJRHWYJAI-UHFFFAOYSA-N Indole Chemical compound C1=CC=C2NC=CC2=C1 SIKJAQJRHWYJAI-UHFFFAOYSA-N 0.000 claims description 30
- 206010028980 Neoplasm Diseases 0.000 claims description 27
- 125000003710 aryl alkyl group Chemical group 0.000 claims description 27
- 208000035475 disorder Diseases 0.000 claims description 24
- 239000002246 antineoplastic agent Substances 0.000 claims description 23
- 125000004076 pyridyl group Chemical group 0.000 claims description 23
- 125000004446 heteroarylalkyl group Chemical group 0.000 claims description 21
- ABLZXFCXXLZCGV-UHFFFAOYSA-N Phosphorous acid Chemical class OP(O)=O ABLZXFCXXLZCGV-UHFFFAOYSA-N 0.000 claims description 20
- 229940127089 cytotoxic agent Drugs 0.000 claims description 19
- 239000008194 pharmaceutical composition Substances 0.000 claims description 19
- 125000002619 bicyclic group Chemical group 0.000 claims description 18
- 125000003282 alkyl amino group Chemical group 0.000 claims description 17
- 150000003457 sulfones Chemical class 0.000 claims description 17
- CHJJGSNFBQVOTG-UHFFFAOYSA-N N-methyl-guanidine Natural products CNC(N)=N CHJJGSNFBQVOTG-UHFFFAOYSA-N 0.000 claims description 16
- 229910019142 PO4 Inorganic materials 0.000 claims description 16
- 125000004663 dialkyl amino group Chemical group 0.000 claims description 16
- SWSQBOPZIKWTGO-UHFFFAOYSA-N dimethylaminoamidine Natural products CN(C)C(N)=N SWSQBOPZIKWTGO-UHFFFAOYSA-N 0.000 claims description 16
- 201000010099 disease Diseases 0.000 claims description 16
- 125000002541 furyl group Chemical group 0.000 claims description 16
- 235000021317 phosphate Nutrition 0.000 claims description 16
- 150000003871 sulfonates Chemical class 0.000 claims description 16
- 229930192474 thiophene Natural products 0.000 claims description 16
- BAXOFTOLAUCFNW-UHFFFAOYSA-N 1H-indazole Chemical compound C1=CC=C2C=NNC2=C1 BAXOFTOLAUCFNW-UHFFFAOYSA-N 0.000 claims description 15
- 150000004657 carbamic acid derivatives Chemical class 0.000 claims description 15
- 235000013877 carbamide Nutrition 0.000 claims description 15
- PZOUSPYUWWUPPK-UHFFFAOYSA-N indole Natural products CC1=CC=CC2=C1C=CN2 PZOUSPYUWWUPPK-UHFFFAOYSA-N 0.000 claims description 15
- RKJUIXBNRJVNHR-UHFFFAOYSA-N indolenine Natural products C1=CC=C2CC=NC2=C1 RKJUIXBNRJVNHR-UHFFFAOYSA-N 0.000 claims description 15
- 150000008039 phosphoramides Chemical class 0.000 claims description 15
- 150000003013 phosphoric acid derivatives Chemical class 0.000 claims description 15
- 150000003456 sulfonamides Chemical class 0.000 claims description 15
- 150000003467 sulfuric acid derivatives Chemical class 0.000 claims description 15
- 150000003556 thioamides Chemical class 0.000 claims description 15
- 150000003585 thioureas Chemical class 0.000 claims description 15
- 150000003672 ureas Chemical class 0.000 claims description 15
- 239000003814 drug Substances 0.000 claims description 14
- 125000001309 chloro group Chemical group Cl* 0.000 claims description 13
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 13
- 125000002950 monocyclic group Chemical group 0.000 claims description 12
- 125000001246 bromo group Chemical group Br* 0.000 claims description 11
- 229940124597 therapeutic agent Drugs 0.000 claims description 9
- 229930194542 Keto Natural products 0.000 claims description 7
- 229940034982 antineoplastic agent Drugs 0.000 claims description 7
- 230000001842 fibrogenetic effect Effects 0.000 claims description 7
- 125000005843 halogen group Chemical group 0.000 claims description 7
- 125000000468 ketone group Chemical group 0.000 claims description 7
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 claims description 7
- 102000004190 Enzymes Human genes 0.000 claims description 5
- 108090000790 Enzymes Proteins 0.000 claims description 5
- 208000015181 infectious disease Diseases 0.000 claims description 5
- 208000020446 Cardiac disease Diseases 0.000 claims description 4
- 208000029523 Interstitial Lung disease Diseases 0.000 claims description 4
- 206010039710 Scleroderma Diseases 0.000 claims description 4
- 208000006011 Stroke Diseases 0.000 claims description 4
- 230000000118 anti-neoplastic effect Effects 0.000 claims description 4
- 208000019425 cirrhosis of liver Diseases 0.000 claims description 4
- 239000002254 cytotoxic agent Substances 0.000 claims description 4
- 231100000599 cytotoxic agent Toxicity 0.000 claims description 4
- 208000019622 heart disease Diseases 0.000 claims description 4
- 201000006334 interstitial nephritis Diseases 0.000 claims description 4
- 208000028867 ischemia Diseases 0.000 claims description 4
- 208000011379 keloid formation Diseases 0.000 claims description 4
- 206010025135 lupus erythematosus Diseases 0.000 claims description 4
- 208000005987 polymyositis Diseases 0.000 claims description 4
- 206010039073 rheumatoid arthritis Diseases 0.000 claims description 4
- 230000009885 systemic effect Effects 0.000 claims description 4
- 208000023275 Autoimmune disease Diseases 0.000 claims description 3
- 201000009030 Carcinoma Diseases 0.000 claims description 3
- 208000012902 Nervous system disease Diseases 0.000 claims description 3
- 208000025966 Neurological disease Diseases 0.000 claims description 3
- 229940100198 alkylating agent Drugs 0.000 claims description 3
- 239000002168 alkylating agent Substances 0.000 claims description 3
- 230000003388 anti-hormonal effect Effects 0.000 claims description 3
- 230000000340 anti-metabolite Effects 0.000 claims description 3
- 229940100197 antimetabolite Drugs 0.000 claims description 3
- 239000002256 antimetabolite Substances 0.000 claims description 3
- 239000003534 dna topoisomerase inhibitor Substances 0.000 claims description 3
- 239000003102 growth factor Substances 0.000 claims description 3
- 239000003966 growth inhibitor Substances 0.000 claims description 3
- 230000003394 haemopoietic effect Effects 0.000 claims description 3
- 230000003054 hormonal effect Effects 0.000 claims description 3
- 239000000367 immunologic factor Substances 0.000 claims description 3
- 208000027866 inflammatory disease Diseases 0.000 claims description 3
- 208000032839 leukemia Diseases 0.000 claims description 3
- 230000003211 malignant effect Effects 0.000 claims description 3
- 208000030159 metabolic disease Diseases 0.000 claims description 3
- 229910052697 platinum Inorganic materials 0.000 claims description 3
- 125000000446 sulfanediyl group Chemical group *S* 0.000 claims description 3
- 229940044693 topoisomerase inhibitor Drugs 0.000 claims description 3
- 239000004037 angiogenesis inhibitor Substances 0.000 claims description 2
- LMBFAGIMSUYTBN-MPZNNTNKSA-N teixobactin Chemical compound C([C@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CO)C(=O)N[C@H](CCC(N)=O)C(=O)N[C@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CO)C(=O)N[C@H]1C(N[C@@H](C)C(=O)N[C@@H](C[C@@H]2NC(=N)NC2)C(=O)N[C@H](C(=O)O[C@H]1C)[C@@H](C)CC)=O)NC)C1=CC=CC=C1 LMBFAGIMSUYTBN-MPZNNTNKSA-N 0.000 claims description 2
- 102100034051 Heat shock protein HSP 90-alpha Human genes 0.000 claims 5
- 230000015572 biosynthetic process Effects 0.000 abstract description 13
- 238000003786 synthesis reaction Methods 0.000 abstract description 11
- 230000002401 inhibitory effect Effects 0.000 abstract description 6
- 230000002265 prevention Effects 0.000 abstract description 2
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 102
- 238000005160 1H NMR spectroscopy Methods 0.000 description 84
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 82
- 238000004128 high performance liquid chromatography Methods 0.000 description 79
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 75
- IAZDPXIOMUYVGZ-WFGJKAKNSA-N Dimethyl sulfoxide Chemical compound [2H]C([2H])([2H])S(=O)C([2H])([2H])[2H] IAZDPXIOMUYVGZ-WFGJKAKNSA-N 0.000 description 64
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 54
- 230000029936 alkylation Effects 0.000 description 48
- 238000005804 alkylation reaction Methods 0.000 description 48
- 125000002147 dimethylamino group Chemical group [H]C([H])([H])N(*)C([H])([H])[H] 0.000 description 48
- 210000004027 cell Anatomy 0.000 description 46
- 0 [1*]C1=C2C=CN([4*][5*])C2=CC([2*])=C1 Chemical compound [1*]C1=C2C=CN([4*][5*])C2=CC([2*])=C1 0.000 description 43
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 42
- 235000019439 ethyl acetate Nutrition 0.000 description 41
- 239000000243 solution Substances 0.000 description 38
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 27
- 101001012157 Homo sapiens Receptor tyrosine-protein kinase erbB-2 Proteins 0.000 description 26
- 102100030086 Receptor tyrosine-protein kinase erbB-2 Human genes 0.000 description 26
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 25
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 24
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 24
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 22
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 22
- XHXFXVLFKHQFAL-UHFFFAOYSA-N phosphoryl trichloride Chemical compound ClP(Cl)(Cl)=O XHXFXVLFKHQFAL-UHFFFAOYSA-N 0.000 description 22
- 238000002360 preparation method Methods 0.000 description 22
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 21
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 20
- 238000000746 purification Methods 0.000 description 20
- 239000000725 suspension Substances 0.000 description 20
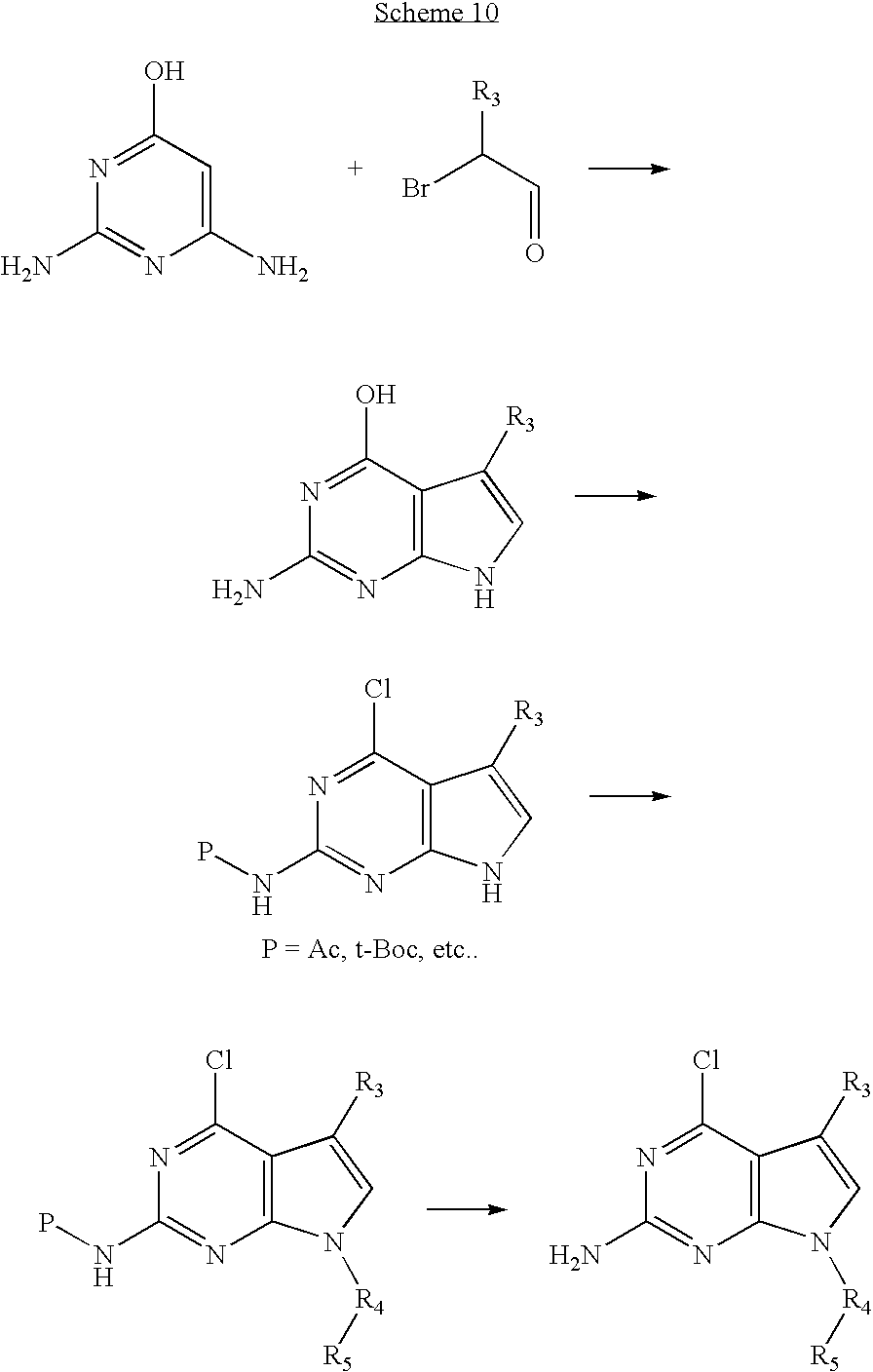
- VIVLSUIQHWGALQ-UHFFFAOYSA-N 4-chloro-7h-pyrrolo[2,3-d]pyrimidin-2-amine Chemical compound NC1=NC(Cl)=C2C=CNC2=N1 VIVLSUIQHWGALQ-UHFFFAOYSA-N 0.000 description 19
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 19
- 238000010626 work up procedure Methods 0.000 description 19
- 230000003647 oxidation Effects 0.000 description 17
- 238000007254 oxidation reaction Methods 0.000 description 17
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 16
- 239000003921 oil Substances 0.000 description 16
- 235000019198 oils Nutrition 0.000 description 16
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 15
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 15
- 108090000623 proteins and genes Proteins 0.000 description 15
- 125000005809 3,4,5-trimethoxyphenyl group Chemical group [H]C1=C(OC([H])([H])[H])C(OC([H])([H])[H])=C(OC([H])([H])[H])C([H])=C1* 0.000 description 14
- 238000003556 assay Methods 0.000 description 14
- 125000004432 carbon atom Chemical group C* 0.000 description 14
- 230000005764 inhibitory process Effects 0.000 description 14
- 239000000546 pharmaceutical excipient Substances 0.000 description 14
- WFDIJRYMOXRFFG-UHFFFAOYSA-N Acetic anhydride Chemical compound CC(=O)OC(C)=O WFDIJRYMOXRFFG-UHFFFAOYSA-N 0.000 description 13
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 13
- 239000002585 base Substances 0.000 description 13
- 238000001704 evaporation Methods 0.000 description 13
- 230000008020 evaporation Effects 0.000 description 13
- 235000018102 proteins Nutrition 0.000 description 13
- 102000004169 proteins and genes Human genes 0.000 description 13
- 239000007787 solid Substances 0.000 description 13
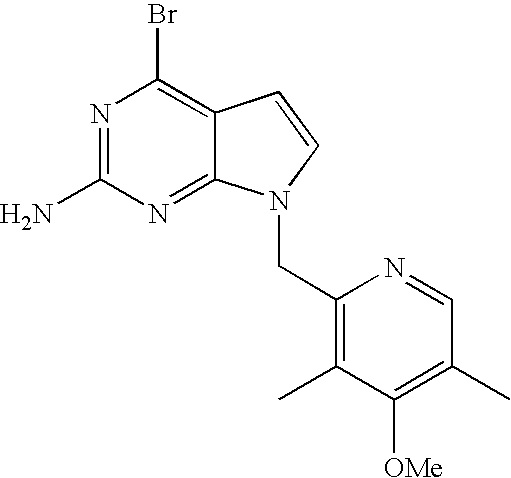
- SRKVJDYNPSMHJM-UHFFFAOYSA-N 2-(chloromethyl)-4-methoxy-3,5-dimethylpyridine Chemical compound COC1=C(C)C=NC(CCl)=C1C SRKVJDYNPSMHJM-UHFFFAOYSA-N 0.000 description 12
- 125000003545 alkoxy group Chemical group 0.000 description 12
- 230000000694 effects Effects 0.000 description 12
- 238000003818 flash chromatography Methods 0.000 description 12
- 229910000027 potassium carbonate Inorganic materials 0.000 description 12
- 238000012746 preparative thin layer chromatography Methods 0.000 description 12
- 125000004178 (C1-C4) alkyl group Chemical group 0.000 description 11
- 229910019213 POCl3 Inorganic materials 0.000 description 11
- 239000003795 chemical substances by application Substances 0.000 description 11
- 125000004122 cyclic group Chemical group 0.000 description 11
- 125000004043 oxo group Chemical group O=* 0.000 description 11
- 239000002904 solvent Substances 0.000 description 11
- 230000001225 therapeutic effect Effects 0.000 description 11
- QMKKJBRRKIKWFK-UHFFFAOYSA-N CNC(=O)C(C)(C)C Chemical compound CNC(=O)C(C)(C)C QMKKJBRRKIKWFK-UHFFFAOYSA-N 0.000 description 10
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 10
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 10
- FJDQFPXHSGXQBY-UHFFFAOYSA-L caesium carbonate Chemical compound [Cs+].[Cs+].[O-]C([O-])=O FJDQFPXHSGXQBY-UHFFFAOYSA-L 0.000 description 10
- 201000011510 cancer Diseases 0.000 description 10
- LOKCTEFSRHRXRJ-UHFFFAOYSA-I dipotassium trisodium dihydrogen phosphate hydrogen phosphate dichloride Chemical compound P(=O)(O)(O)[O-].[K+].P(=O)(O)([O-])[O-].[Na+].[Na+].[Cl-].[K+].[Cl-].[Na+] LOKCTEFSRHRXRJ-UHFFFAOYSA-I 0.000 description 10
- 239000002953 phosphate buffered saline Substances 0.000 description 10
- 230000005855 radiation Effects 0.000 description 10
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 9
- 239000004480 active ingredient Substances 0.000 description 9
- 229910021529 ammonia Inorganic materials 0.000 description 9
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 9
- 239000000463 material Substances 0.000 description 9
- 238000010992 reflux Methods 0.000 description 9
- 238000012360 testing method Methods 0.000 description 9
- 125000004001 thioalkyl group Chemical group 0.000 description 9
- OKNZIPMFCKVSGB-UHFFFAOYSA-N 2-amino-4-chloro-7-[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]-5h-pyrrolo[2,3-d]pyrimidin-6-one Chemical compound COC1=C(C)C=NC(CN2C3=C(C(=NC(N)=N3)Cl)CC2=O)=C1C OKNZIPMFCKVSGB-UHFFFAOYSA-N 0.000 description 8
- JJTNLWSCFYERCK-UHFFFAOYSA-N 7h-pyrrolo[2,3-d]pyrimidine Chemical compound N1=CN=C2NC=CC2=C1 JJTNLWSCFYERCK-UHFFFAOYSA-N 0.000 description 8
- 206010006187 Breast cancer Diseases 0.000 description 8
- 208000026310 Breast neoplasm Diseases 0.000 description 8
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 8
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 8
- JLTDJTHDQAWBAV-UHFFFAOYSA-N N,N-dimethylaniline Chemical compound CN(C)C1=CC=CC=C1 JLTDJTHDQAWBAV-UHFFFAOYSA-N 0.000 description 8
- GXCLVBGFBYZDAG-UHFFFAOYSA-N N-[2-(1H-indol-3-yl)ethyl]-N-methylprop-2-en-1-amine Chemical compound CN(CCC1=CNC2=C1C=CC=C2)CC=C GXCLVBGFBYZDAG-UHFFFAOYSA-N 0.000 description 8
- 239000012267 brine Substances 0.000 description 8
- 239000003153 chemical reaction reagent Substances 0.000 description 8
- 230000001419 dependent effect Effects 0.000 description 8
- 238000001914 filtration Methods 0.000 description 8
- 239000000047 product Substances 0.000 description 8
- LJXQPZWIHJMPQQ-UHFFFAOYSA-N pyrimidin-2-amine Chemical compound NC1=NC=CC=N1 LJXQPZWIHJMPQQ-UHFFFAOYSA-N 0.000 description 8
- JPJALAQPGMAKDF-UHFFFAOYSA-N selenium dioxide Chemical compound O=[Se]=O JPJALAQPGMAKDF-UHFFFAOYSA-N 0.000 description 8
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical compound O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 8
- RIOQSEWOXXDEQQ-UHFFFAOYSA-N triphenylphosphine Chemical compound C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 RIOQSEWOXXDEQQ-UHFFFAOYSA-N 0.000 description 8
- LNHBUZIEWGLSMI-UHFFFAOYSA-N 4-bromo-7h-pyrrolo[2,3-d]pyrimidin-2-amine Chemical compound NC1=NC(Br)=C2C=CNC2=N1 LNHBUZIEWGLSMI-UHFFFAOYSA-N 0.000 description 7
- BAIWBDZQCQHMJV-UHFFFAOYSA-N 4-chloro-2-(chloromethyl)-3,5-dimethylpyridine Chemical compound CC1=CN=C(CCl)C(C)=C1Cl BAIWBDZQCQHMJV-UHFFFAOYSA-N 0.000 description 7
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 7
- 239000002253 acid Substances 0.000 description 7
- 125000004429 atom Chemical group 0.000 description 7
- 230000027455 binding Effects 0.000 description 7
- 229910052794 bromium Inorganic materials 0.000 description 7
- 238000001035 drying Methods 0.000 description 7
- 239000012039 electrophile Substances 0.000 description 7
- 125000000524 functional group Chemical group 0.000 description 7
- 230000004048 modification Effects 0.000 description 7
- 238000012986 modification Methods 0.000 description 7
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 7
- 239000000523 sample Substances 0.000 description 7
- 239000000126 substance Substances 0.000 description 7
- WDYVUKGVKRZQNM-UHFFFAOYSA-N 6-phosphonohexylphosphonic acid Chemical compound OP(O)(=O)CCCCCCP(O)(O)=O WDYVUKGVKRZQNM-UHFFFAOYSA-N 0.000 description 6
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 6
- PCLIMKBDDGJMGD-UHFFFAOYSA-N N-bromosuccinimide Chemical compound BrN1C(=O)CCC1=O PCLIMKBDDGJMGD-UHFFFAOYSA-N 0.000 description 6
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 6
- KAESVJOAVNADME-UHFFFAOYSA-N Pyrrole Chemical compound C=1C=CNC=1 KAESVJOAVNADME-UHFFFAOYSA-N 0.000 description 6
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 6
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 6
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 6
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Chemical compound BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 6
- 239000003054 catalyst Substances 0.000 description 6
- 235000014113 dietary fatty acids Nutrition 0.000 description 6
- 239000000194 fatty acid Substances 0.000 description 6
- 229930195729 fatty acid Natural products 0.000 description 6
- 239000000796 flavoring agent Substances 0.000 description 6
- 230000014509 gene expression Effects 0.000 description 6
- 150000004820 halides Chemical class 0.000 description 6
- 239000005457 ice water Substances 0.000 description 6
- 239000003112 inhibitor Substances 0.000 description 6
- KDLHZDBZIXYQEI-UHFFFAOYSA-N palladium Substances [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 6
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 6
- 125000000246 pyrimidin-2-yl group Chemical group [H]C1=NC(*)=NC([H])=C1[H] 0.000 description 6
- 239000003765 sweetening agent Substances 0.000 description 6
- VZGDMQKNWNREIO-UHFFFAOYSA-N tetrachloromethane Chemical compound ClC(Cl)(Cl)Cl VZGDMQKNWNREIO-UHFFFAOYSA-N 0.000 description 6
- UMGDCJDMYOKAJW-UHFFFAOYSA-N thiourea Chemical compound NC(N)=S UMGDCJDMYOKAJW-UHFFFAOYSA-N 0.000 description 6
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 6
- LWEDMPDRWLOJDD-UHFFFAOYSA-N 2-(chloromethyl)-4-methoxy-3,5-dimethyl-1-oxidopyridin-1-ium Chemical compound COC1=C(C)C=[N+]([O-])C(CCl)=C1C LWEDMPDRWLOJDD-UHFFFAOYSA-N 0.000 description 5
- WUUXYBFKNASCHL-UHFFFAOYSA-N 2-amino-4-chloro-7-[(3,4,5-trimethoxyphenyl)methyl]-5h-pyrrolo[2,3-d]pyrimidin-6-one Chemical compound COC1=C(OC)C(OC)=CC(CN2C3=C(C(=NC(N)=N3)Cl)CC2=O)=C1 WUUXYBFKNASCHL-UHFFFAOYSA-N 0.000 description 5
- GRSASFHRWSWUHX-UHFFFAOYSA-N 4-chloro-5-propan-2-yl-7h-pyrrolo[2,3-d]pyrimidin-2-amine Chemical compound NC1=NC(Cl)=C2C(C(C)C)=CNC2=N1 GRSASFHRWSWUHX-UHFFFAOYSA-N 0.000 description 5
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 5
- CPELXLSAUQHCOX-UHFFFAOYSA-N Hydrogen bromide Chemical compound Br CPELXLSAUQHCOX-UHFFFAOYSA-N 0.000 description 5
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 5
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 5
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 5
- 125000004414 alkyl thio group Chemical group 0.000 description 5
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 5
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 5
- 239000000872 buffer Substances 0.000 description 5
- 229910000024 caesium carbonate Inorganic materials 0.000 description 5
- 230000015556 catabolic process Effects 0.000 description 5
- 238000004587 chromatography analysis Methods 0.000 description 5
- 239000007859 condensation product Substances 0.000 description 5
- 238000006731 degradation reaction Methods 0.000 description 5
- 150000004665 fatty acids Chemical class 0.000 description 5
- 235000003599 food sweetener Nutrition 0.000 description 5
- 238000009472 formulation Methods 0.000 description 5
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 5
- KZDHRXDFCHHFEJ-UHFFFAOYSA-N n-(4-chloro-5-iodo-7h-pyrrolo[2,3-d]pyrimidin-2-yl)-2,2-dimethylpropanamide Chemical compound CC(C)(C)C(=O)NC1=NC(Cl)=C2C(I)=CNC2=N1 KZDHRXDFCHHFEJ-UHFFFAOYSA-N 0.000 description 5
- HNBVXLCVUIJCSK-UHFFFAOYSA-N n-(4-chloro-7h-pyrrolo[2,3-d]pyrimidin-2-yl)-2,2-dimethylpropanamide Chemical compound N1C(NC(=O)C(C)(C)C)=NC(Cl)=C2C=CN=C21 HNBVXLCVUIJCSK-UHFFFAOYSA-N 0.000 description 5
- 239000001301 oxygen Substances 0.000 description 5
- 229920001223 polyethylene glycol Polymers 0.000 description 5
- 238000003752 polymerase chain reaction Methods 0.000 description 5
- 239000003755 preservative agent Substances 0.000 description 5
- 150000004943 pyrrolo[2,3-d]pyrimidines Chemical class 0.000 description 5
- 230000009467 reduction Effects 0.000 description 5
- 230000004044 response Effects 0.000 description 5
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 5
- 239000000758 substrate Substances 0.000 description 5
- 125000004434 sulfur atom Chemical group 0.000 description 5
- 208000024891 symptom Diseases 0.000 description 5
- 230000000699 topical effect Effects 0.000 description 5
- OBGVMNAMBZAWKG-UHFFFAOYSA-N 2-amino-4-chloro-7-[(4-chloro-3,5-dimethylpyridin-2-yl)methyl]-5h-pyrrolo[2,3-d]pyrimidin-6-one Chemical compound CC1=C(Cl)C(C)=CN=C1CN1C(N=C(N)N=C2Cl)=C2CC1=O OBGVMNAMBZAWKG-UHFFFAOYSA-N 0.000 description 4
- NHQDETIJWKXCTC-UHFFFAOYSA-N 3-chloroperbenzoic acid Chemical compound OOC(=O)C1=CC=CC(Cl)=C1 NHQDETIJWKXCTC-UHFFFAOYSA-N 0.000 description 4
- XHYZMYGEIKKHLE-UHFFFAOYSA-N 4-bromo-2-(chloromethyl)-3,5-dimethyl-1-oxidopyridin-1-ium Chemical compound CC1=C[N+]([O-])=C(CCl)C(C)=C1Br XHYZMYGEIKKHLE-UHFFFAOYSA-N 0.000 description 4
- IQQDBXHZFBZFKJ-UHFFFAOYSA-N 4-bromo-2-(chloromethyl)-3,5-dimethylpyridine Chemical compound CC1=CN=C(CCl)C(C)=C1Br IQQDBXHZFBZFKJ-UHFFFAOYSA-N 0.000 description 4
- XCVCBOBAZRPXIT-UHFFFAOYSA-N 4-chloro-2-(chloromethyl)-3,5-dimethyl-1-oxidopyridin-1-ium Chemical compound CC1=C[N+]([O-])=C(CCl)C(C)=C1Cl XCVCBOBAZRPXIT-UHFFFAOYSA-N 0.000 description 4
- GLPLKXYNIUWGGS-UHFFFAOYSA-N 4-chloro-7-[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]-5-[2-(2-methylpropylamino)ethyl]pyrrolo[2,3-d]pyrimidin-2-amine Chemical compound COC1=C(C)C=NC(CN2C3=NC(N)=NC(Cl)=C3C(CCNCC(C)C)=C2)=C1C GLPLKXYNIUWGGS-UHFFFAOYSA-N 0.000 description 4
- MRIZBPRGVMKLON-UHFFFAOYSA-N 4-chloro-7-[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]-5-propan-2-ylpyrrolo[2,3-d]pyrimidin-2-amine Chemical compound COC1=C(C)C=NC(CN2C3=NC(N)=NC(Cl)=C3C(C(C)C)=C2)=C1C MRIZBPRGVMKLON-UHFFFAOYSA-N 0.000 description 4
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 4
- PAYRUJLWNCNPSJ-UHFFFAOYSA-N Aniline Chemical compound NC1=CC=CC=C1 PAYRUJLWNCNPSJ-UHFFFAOYSA-N 0.000 description 4
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 4
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 4
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 4
- 108010010803 Gelatin Proteins 0.000 description 4
- MHAJPDPJQMAIIY-UHFFFAOYSA-N Hydrogen peroxide Chemical compound OO MHAJPDPJQMAIIY-UHFFFAOYSA-N 0.000 description 4
- 238000005684 Liebig rearrangement reaction Methods 0.000 description 4
- 108010006519 Molecular Chaperones Proteins 0.000 description 4
- 108010004729 Phycoerythrin Proteins 0.000 description 4
- SMWDFEZZVXVKRB-UHFFFAOYSA-N Quinoline Chemical compound N1=CC=CC2=CC=CC=C21 SMWDFEZZVXVKRB-UHFFFAOYSA-N 0.000 description 4
- PXIPVTKHYLBLMZ-UHFFFAOYSA-N Sodium azide Chemical compound [Na+].[N-]=[N+]=[N-] PXIPVTKHYLBLMZ-UHFFFAOYSA-N 0.000 description 4
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 4
- 239000003963 antioxidant agent Substances 0.000 description 4
- 235000006708 antioxidants Nutrition 0.000 description 4
- 239000007900 aqueous suspension Substances 0.000 description 4
- 230000037396 body weight Effects 0.000 description 4
- 239000003086 colorant Substances 0.000 description 4
- 239000003085 diluting agent Substances 0.000 description 4
- 239000002270 dispersing agent Substances 0.000 description 4
- 229940079593 drug Drugs 0.000 description 4
- 239000003937 drug carrier Substances 0.000 description 4
- 229940088598 enzyme Drugs 0.000 description 4
- 239000012091 fetal bovine serum Substances 0.000 description 4
- 235000013355 food flavoring agent Nutrition 0.000 description 4
- 239000000499 gel Substances 0.000 description 4
- 239000008273 gelatin Substances 0.000 description 4
- 229920000159 gelatin Polymers 0.000 description 4
- 235000019322 gelatine Nutrition 0.000 description 4
- 235000011852 gelatine desserts Nutrition 0.000 description 4
- QTQAWLPCGQOSGP-GBTDJJJQSA-N geldanamycin Chemical compound N1C(=O)\C(C)=C/C=C\[C@@H](OC)[C@H](OC(N)=O)\C(C)=C/[C@@H](C)[C@@H](O)[C@H](OC)C[C@@H](C)CC2=C(OC)C(=O)C=C1C2=O QTQAWLPCGQOSGP-GBTDJJJQSA-N 0.000 description 4
- 230000026030 halogenation Effects 0.000 description 4
- 238000005658 halogenation reaction Methods 0.000 description 4
- 125000004404 heteroalkyl group Chemical group 0.000 description 4
- 238000002347 injection Methods 0.000 description 4
- 239000007924 injection Substances 0.000 description 4
- 229910052740 iodine Inorganic materials 0.000 description 4
- INQOMBQAUSQDDS-UHFFFAOYSA-N iodomethane Chemical compound IC INQOMBQAUSQDDS-UHFFFAOYSA-N 0.000 description 4
- ZCSHNCUQKCANBX-UHFFFAOYSA-N lithium diisopropylamide Chemical compound [Li+].CC(C)[N-]C(C)C ZCSHNCUQKCANBX-UHFFFAOYSA-N 0.000 description 4
- 239000004530 micro-emulsion Substances 0.000 description 4
- ULWOJODHECIZAU-UHFFFAOYSA-N n,n-diethylpropan-2-amine Chemical compound CCN(CC)C(C)C ULWOJODHECIZAU-UHFFFAOYSA-N 0.000 description 4
- 231100000252 nontoxic Toxicity 0.000 description 4
- 230000003000 nontoxic effect Effects 0.000 description 4
- 238000010534 nucleophilic substitution reaction Methods 0.000 description 4
- 239000004006 olive oil Substances 0.000 description 4
- 235000008390 olive oil Nutrition 0.000 description 4
- 230000002018 overexpression Effects 0.000 description 4
- 230000036961 partial effect Effects 0.000 description 4
- UEZVMMHDMIWARA-UHFFFAOYSA-M phosphonate Chemical group [O-]P(=O)=O UEZVMMHDMIWARA-UHFFFAOYSA-M 0.000 description 4
- 239000002798 polar solvent Substances 0.000 description 4
- 239000000843 powder Substances 0.000 description 4
- 239000002243 precursor Substances 0.000 description 4
- SHNUBALDGXWUJI-UHFFFAOYSA-N pyridin-2-ylmethanol Chemical class OCC1=CC=CC=N1 SHNUBALDGXWUJI-UHFFFAOYSA-N 0.000 description 4
- 238000001959 radiotherapy Methods 0.000 description 4
- LPXPTNMVRIOKMN-UHFFFAOYSA-M sodium nitrite Chemical compound [Na+].[O-]N=O LPXPTNMVRIOKMN-UHFFFAOYSA-M 0.000 description 4
- 235000000346 sugar Nutrition 0.000 description 4
- 239000011593 sulfur Substances 0.000 description 4
- 239000000375 suspending agent Substances 0.000 description 4
- 239000003826 tablet Substances 0.000 description 4
- FYSNRJHAOHDILO-UHFFFAOYSA-N thionyl chloride Chemical compound ClS(Cl)=O FYSNRJHAOHDILO-UHFFFAOYSA-N 0.000 description 4
- 239000000080 wetting agent Substances 0.000 description 4
- YSBDSWBSEXCSSE-UHFFFAOYSA-N 2,3,5-trimethyl-1-oxidopyridin-1-ium Chemical compound CC1=CC(C)=C(C)[N+]([O-])=C1 YSBDSWBSEXCSSE-UHFFFAOYSA-N 0.000 description 3
- ZYOPGXVIDGBHLM-UHFFFAOYSA-N 2-(chloromethyl)-3,5-dimethyl-1h-pyridin-4-one Chemical compound CC1=CNC(CCl)=C(C)C1=O ZYOPGXVIDGBHLM-UHFFFAOYSA-N 0.000 description 3
- LCJDHJOUOJSJGS-UHFFFAOYSA-N 2-(chloromethyl)-4-methoxy-3,5-dimethylpyridin-1-ium;chloride Chemical compound Cl.COC1=C(C)C=NC(CCl)=C1C LCJDHJOUOJSJGS-UHFFFAOYSA-N 0.000 description 3
- GOADXANONHWKFZ-UHFFFAOYSA-N 2-[2-amino-4-chloro-7-[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]pyrrolo[2,3-d]pyrimidin-5-yl]ethanol Chemical compound COC1=C(C)C=NC(CN2C3=NC(N)=NC(Cl)=C3C(CCO)=C2)=C1C GOADXANONHWKFZ-UHFFFAOYSA-N 0.000 description 3
- NKRUETLHIAFSRL-UHFFFAOYSA-N 2-amino-5-propan-2-yl-1,7-dihydropyrrolo[2,3-d]pyrimidin-4-one Chemical compound N1=C(N)NC(=O)C2=C1NC=C2C(C)C NKRUETLHIAFSRL-UHFFFAOYSA-N 0.000 description 3
- FSMOSXMNXZERIL-UHFFFAOYSA-N 2-amino-7-[(2-bromo-3,4,5-trimethoxyphenyl)methyl]-4-chloro-5h-pyrrolo[2,3-d]pyrimidin-6-one Chemical compound COC1=C(OC)C(OC)=CC(CN2C3=C(C(=NC(N)=N3)Cl)CC2=O)=C1Br FSMOSXMNXZERIL-UHFFFAOYSA-N 0.000 description 3
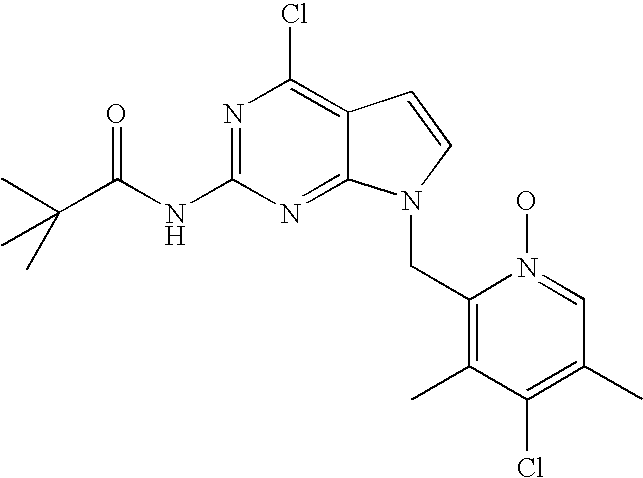
- OMZJYYILWBEANE-UHFFFAOYSA-N 2-chloro-6-(chloromethyl)-4-methoxy-3,5-dimethylpyridine Chemical compound COC1=C(C)C(Cl)=NC(CCl)=C1C OMZJYYILWBEANE-UHFFFAOYSA-N 0.000 description 3
- VESACMRPXUVRKJ-UHFFFAOYSA-N 3-bromo-4-methylpentanal Chemical compound CC(C)C(Br)CC=O VESACMRPXUVRKJ-UHFFFAOYSA-N 0.000 description 3
- WDHFLSDUDGZUKC-UHFFFAOYSA-N 4-bromo-2-(bromomethyl)-3,5-dimethylpyridine Chemical compound CC1=CN=C(CBr)C(C)=C1Br WDHFLSDUDGZUKC-UHFFFAOYSA-N 0.000 description 3
- BXBLDQBGTGOLJL-UHFFFAOYSA-N 4-bromo-7-[(4-bromo-3,5-dimethylpyridin-2-yl)methyl]pyrrolo[2,3-d]pyrimidin-2-amine Chemical compound CC1=C(Br)C(C)=CN=C1CN1C2=NC(N)=NC(Br)=C2C=C1 BXBLDQBGTGOLJL-UHFFFAOYSA-N 0.000 description 3
- UVXHHZHDHDQPMQ-UHFFFAOYSA-N 4-bromo-7-[(4-chloro-3,5-dimethylpyridin-2-yl)methyl]pyrrolo[2,3-d]pyrimidin-2-amine Chemical compound CC1=C(Cl)C(C)=CN=C1CN1C2=NC(N)=NC(Br)=C2C=C1 UVXHHZHDHDQPMQ-UHFFFAOYSA-N 0.000 description 3
- LVELLGFWQLBCMI-UHFFFAOYSA-N 4-bromo-7-[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]pyrrolo[2,3-d]pyrimidin-2-amine Chemical compound COC1=C(C)C=NC(CN2C3=NC(N)=NC(Br)=C3C=C2)=C1C LVELLGFWQLBCMI-UHFFFAOYSA-N 0.000 description 3
- UTYKALJIQPGPEY-UHFFFAOYSA-N 4-chloro-7-[(4-chloro-3,5-dimethylpyridin-2-yl)methyl]pyrrolo[2,3-d]pyrimidin-2-amine Chemical compound CC1=C(Cl)C(C)=CN=C1CN1C2=NC(N)=NC(Cl)=C2C=C1 UTYKALJIQPGPEY-UHFFFAOYSA-N 0.000 description 3
- OSIVBPGKYWLPGE-UHFFFAOYSA-N 4-chloro-7-[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]-6-(pyrrolidin-1-ylmethyl)pyrrolo[2,3-d]pyrimidin-2-amine Chemical compound COC1=C(C)C=NC(CN2C3=NC(N)=NC(Cl)=C3C=C2CN2CCCC2)=C1C OSIVBPGKYWLPGE-UHFFFAOYSA-N 0.000 description 3
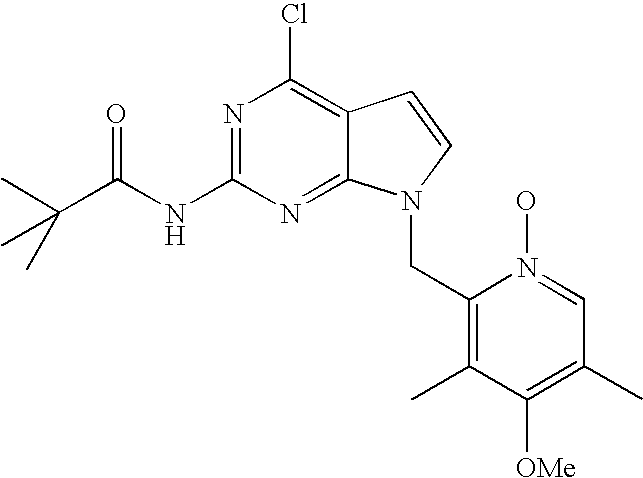
- LHLULKGITMTJMB-UHFFFAOYSA-N 4-chloro-7-[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]pyrrolo[2,3-d]pyrimidin-2-amine Chemical compound COC1=C(C)C=NC(CN2C3=NC(N)=NC(Cl)=C3C=C2)=C1C LHLULKGITMTJMB-UHFFFAOYSA-N 0.000 description 3
- BBUPYQLZNSPTGY-UHFFFAOYSA-N 4-chloro-7-[(4-methoxyphenyl)methyl]pyrrolo[2,3-d]pyrimidin-2-amine Chemical compound C1=CC(OC)=CC=C1CN1C2=NC(N)=NC(Cl)=C2C=C1 BBUPYQLZNSPTGY-UHFFFAOYSA-N 0.000 description 3
- PVDDFQUAZQLROB-UHFFFAOYSA-N 4-chloro-7-[(6-chloro-4-methoxy-3,5-dimethylpyridin-2-yl)methyl]pyrrolo[2,3-d]pyrimidin-2-amine Chemical compound COC1=C(C)C(Cl)=NC(CN2C3=NC(N)=NC(Cl)=C3C=C2)=C1C PVDDFQUAZQLROB-UHFFFAOYSA-N 0.000 description 3
- AIOVNNOZJCABNA-UHFFFAOYSA-N 7-[(4-bromo-3,5-dimethylpyridin-2-yl)methyl]-4-chloropyrrolo[2,3-d]pyrimidin-2-amine Chemical compound CC1=C(Br)C(C)=CN=C1CN1C2=NC(N)=NC(Cl)=C2C=C1 AIOVNNOZJCABNA-UHFFFAOYSA-N 0.000 description 3
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical group [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 3
- YJABUDQVPTVLEI-UHFFFAOYSA-N COC1=C(OC)C(OC)=CC(CN2C=3C(C(=NC(=N)N=3)Cl)=C(O)C2=O)=C1Br Chemical compound COC1=C(OC)C(OC)=CC(CN2C=3C(C(=NC(=N)N=3)Cl)=C(O)C2=O)=C1Br YJABUDQVPTVLEI-UHFFFAOYSA-N 0.000 description 3
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical group [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 3
- UHDGCWIWMRVCDJ-CCXZUQQUSA-N Cytarabine Chemical compound O=C1N=C(N)C=CN1[C@H]1[C@@H](O)[C@H](O)[C@@H](CO)O1 UHDGCWIWMRVCDJ-CCXZUQQUSA-N 0.000 description 3
- 239000006144 Dulbecco’s modified Eagle's medium Substances 0.000 description 3
- 102000001301 EGF receptor Human genes 0.000 description 3
- 108060006698 EGF receptor Proteins 0.000 description 3
- JRZJKWGQFNTSRN-UHFFFAOYSA-N Geldanamycin Natural products C1C(C)CC(OC)C(O)C(C)C=C(C)C(OC(N)=O)C(OC)CCC=C(C)C(=O)NC2=CC(=O)C(OC)=C1C2=O JRZJKWGQFNTSRN-UHFFFAOYSA-N 0.000 description 3
- OAKJQQAXSVQMHS-UHFFFAOYSA-N Hydrazine Chemical compound NN OAKJQQAXSVQMHS-UHFFFAOYSA-N 0.000 description 3
- 238000000719 MTS assay Methods 0.000 description 3
- 231100000070 MTS assay Toxicity 0.000 description 3
- 206010027476 Metastases Diseases 0.000 description 3
- 102000005431 Molecular Chaperones Human genes 0.000 description 3
- JRNVZBWKYDBUCA-UHFFFAOYSA-N N-chlorosuccinimide Chemical compound ClN1C(=O)CCC1=O JRNVZBWKYDBUCA-UHFFFAOYSA-N 0.000 description 3
- LQZMLBORDGWNPD-UHFFFAOYSA-N N-iodosuccinimide Chemical compound IN1C(=O)CCC1=O LQZMLBORDGWNPD-UHFFFAOYSA-N 0.000 description 3
- 108020004711 Nucleic Acid Probes Proteins 0.000 description 3
- 229910019201 POBr3 Inorganic materials 0.000 description 3
- 239000002202 Polyethylene glycol Substances 0.000 description 3
- VMHLLURERBWHNL-UHFFFAOYSA-M Sodium acetate Chemical compound [Na+].CC([O-])=O VMHLLURERBWHNL-UHFFFAOYSA-M 0.000 description 3
- 229920002472 Starch Polymers 0.000 description 3
- 229930006000 Sucrose Natural products 0.000 description 3
- OKJPEAGHQZHRQV-UHFFFAOYSA-N Triiodomethane Natural products IC(I)I OKJPEAGHQZHRQV-UHFFFAOYSA-N 0.000 description 3
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Natural products NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 3
- 125000002015 acyclic group Chemical group 0.000 description 3
- 235000010443 alginic acid Nutrition 0.000 description 3
- 229920000615 alginic acid Polymers 0.000 description 3
- 150000004781 alginic acids Chemical class 0.000 description 3
- 150000001412 amines Chemical class 0.000 description 3
- 230000001093 anti-cancer Effects 0.000 description 3
- 230000003078 antioxidant effect Effects 0.000 description 3
- 125000005110 aryl thio group Chemical group 0.000 description 3
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 3
- 230000004071 biological effect Effects 0.000 description 3
- 201000008275 breast carcinoma Diseases 0.000 description 3
- 229910000019 calcium carbonate Inorganic materials 0.000 description 3
- 239000001506 calcium phosphate Substances 0.000 description 3
- 229910000389 calcium phosphate Inorganic materials 0.000 description 3
- 235000011010 calcium phosphates Nutrition 0.000 description 3
- 230000010261 cell growth Effects 0.000 description 3
- 239000013592 cell lysate Substances 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 229910052801 chlorine Inorganic materials 0.000 description 3
- 238000013270 controlled release Methods 0.000 description 3
- 230000008878 coupling Effects 0.000 description 3
- 238000010168 coupling process Methods 0.000 description 3
- 238000005859 coupling reaction Methods 0.000 description 3
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 3
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 3
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 3
- IJKVHSBPTUYDLN-UHFFFAOYSA-N dihydroxy(oxo)silane Chemical compound O[Si](O)=O IJKVHSBPTUYDLN-UHFFFAOYSA-N 0.000 description 3
- WAFZJLNLVZWUSY-UHFFFAOYSA-N ethyl 2-(2-amino-4,6-dichloropyrimidin-5-yl)acetate Chemical compound CCOC(=O)CC1=C(Cl)N=C(N)N=C1Cl WAFZJLNLVZWUSY-UHFFFAOYSA-N 0.000 description 3
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 3
- MMXKVMNBHPAILY-UHFFFAOYSA-N ethyl laurate Chemical compound CCCCCCCCCCCC(=O)OCC MMXKVMNBHPAILY-UHFFFAOYSA-N 0.000 description 3
- 238000000605 extraction Methods 0.000 description 3
- 238000002509 fluorescent in situ hybridization Methods 0.000 description 3
- 235000011187 glycerol Nutrition 0.000 description 3
- 230000012010 growth Effects 0.000 description 3
- 125000001188 haloalkyl group Chemical group 0.000 description 3
- 150000002391 heterocyclic compounds Chemical class 0.000 description 3
- 239000000543 intermediate Substances 0.000 description 3
- 238000003402 intramolecular cyclocondensation reaction Methods 0.000 description 3
- 238000001990 intravenous administration Methods 0.000 description 3
- 235000013675 iodine Nutrition 0.000 description 3
- 230000000670 limiting effect Effects 0.000 description 3
- 229940057995 liquid paraffin Drugs 0.000 description 3
- 238000004519 manufacturing process Methods 0.000 description 3
- 239000011159 matrix material Substances 0.000 description 3
- 238000005259 measurement Methods 0.000 description 3
- 230000009401 metastasis Effects 0.000 description 3
- 150000007522 mineralic acids Chemical class 0.000 description 3
- VMGAPWLDMVPYIA-HIDZBRGKSA-N n'-amino-n-iminomethanimidamide Chemical compound N\N=C\N=N VMGAPWLDMVPYIA-HIDZBRGKSA-N 0.000 description 3
- FYYGFJXFFZQZSV-UHFFFAOYSA-N n-[4-chloro-6-(pyrrolidin-1-ylmethyl)-7h-pyrrolo[2,3-d]pyrimidin-2-yl]octanamide Chemical compound N1C2=NC(NC(=O)CCCCCCC)=NC(Cl)=C2C=C1CN1CCCC1 FYYGFJXFFZQZSV-UHFFFAOYSA-N 0.000 description 3
- QIOVDTJAHJLIII-UHFFFAOYSA-N n-[4-chloro-7-[(4-chloro-3,5-dimethylpyridin-2-yl)methyl]-5-iodopyrrolo[2,3-d]pyrimidin-2-yl]-2,2-dimethylpropanamide Chemical compound CC1=C(Cl)C(C)=CN=C1CN1C2=NC(NC(=O)C(C)(C)C)=NC(Cl)=C2C(I)=C1 QIOVDTJAHJLIII-UHFFFAOYSA-N 0.000 description 3
- BOCISLNTLNBUHN-UHFFFAOYSA-N n-[4-chloro-7-[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]pyrrolo[2,3-d]pyrimidin-2-yl]-2,2-dimethylpropanamide Chemical compound COC1=C(C)C=NC(CN2C3=NC(NC(=O)C(C)(C)C)=NC(Cl)=C3C=C2)=C1C BOCISLNTLNBUHN-UHFFFAOYSA-N 0.000 description 3
- QWQAKESRAMSQNY-UHFFFAOYSA-N n-[5-[(dibenzylamino)methyl]-4-oxo-1,7-dihydropyrrolo[2,3-d]pyrimidin-2-yl]octanamide Chemical compound C1=2C(=O)NC(NC(=O)CCCCCCC)=NC=2NC=C1CN(CC=1C=CC=CC=1)CC1=CC=CC=C1 QWQAKESRAMSQNY-UHFFFAOYSA-N 0.000 description 3
- 239000013642 negative control Substances 0.000 description 3
- 229910000069 nitrogen hydride Inorganic materials 0.000 description 3
- 239000002853 nucleic acid probe Substances 0.000 description 3
- 108020004707 nucleic acids Proteins 0.000 description 3
- 102000039446 nucleic acids Human genes 0.000 description 3
- 150000007523 nucleic acids Chemical class 0.000 description 3
- 210000000056 organ Anatomy 0.000 description 3
- 150000007524 organic acids Chemical class 0.000 description 3
- 125000004430 oxygen atom Chemical group O* 0.000 description 3
- 229910052763 palladium Inorganic materials 0.000 description 3
- 239000008196 pharmacological composition Substances 0.000 description 3
- UXCDUFKZSUBXGM-UHFFFAOYSA-N phosphoric tribromide Chemical compound BrP(Br)(Br)=O UXCDUFKZSUBXGM-UHFFFAOYSA-N 0.000 description 3
- 230000003389 potentiating effect Effects 0.000 description 3
- 239000002244 precipitate Substances 0.000 description 3
- 230000000069 prophylactic effect Effects 0.000 description 3
- ILVXOBCQQYKLDS-UHFFFAOYSA-N pyridine N-oxide Chemical compound [O-][N+]1=CC=CC=C1 ILVXOBCQQYKLDS-UHFFFAOYSA-N 0.000 description 3
- HMOPBGFCZBTBMX-UHFFFAOYSA-N pyrrolo[2,3-d]pyrimidin-6-one Chemical compound C1=NC=NC2=NC(=O)C=C21 HMOPBGFCZBTBMX-UHFFFAOYSA-N 0.000 description 3
- 230000002829 reductive effect Effects 0.000 description 3
- BOLDJAUMGUJJKM-LSDHHAIUSA-N renifolin D Natural products CC(=C)[C@@H]1Cc2c(O)c(O)ccc2[C@H]1CC(=O)c3ccc(O)cc3O BOLDJAUMGUJJKM-LSDHHAIUSA-N 0.000 description 3
- 229910000029 sodium carbonate Inorganic materials 0.000 description 3
- 238000010561 standard procedure Methods 0.000 description 3
- 235000019698 starch Nutrition 0.000 description 3
- 239000007858 starting material Substances 0.000 description 3
- 239000005720 sucrose Substances 0.000 description 3
- 229960004793 sucrose Drugs 0.000 description 3
- 210000001519 tissue Anatomy 0.000 description 3
- 230000009466 transformation Effects 0.000 description 3
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 3
- 235000015112 vegetable and seed oil Nutrition 0.000 description 3
- 239000008158 vegetable oil Substances 0.000 description 3
- 239000003981 vehicle Substances 0.000 description 3
- 238000001262 western blot Methods 0.000 description 3
- PUPZLCDOIYMWBV-UHFFFAOYSA-N (+/-)-1,3-Butanediol Chemical compound CC(O)CCO PUPZLCDOIYMWBV-UHFFFAOYSA-N 0.000 description 2
- QGIYYKAVFCPEGC-UHFFFAOYSA-N (4-bromo-3,5-dimethylpyridin-2-yl)methanol Chemical compound CC1=CN=C(CO)C(C)=C1Br QGIYYKAVFCPEGC-UHFFFAOYSA-N 0.000 description 2
- 125000000229 (C1-C4)alkoxy group Chemical group 0.000 description 2
- ZORQXIQZAOLNGE-UHFFFAOYSA-N 1,1-difluorocyclohexane Chemical compound FC1(F)CCCCC1 ZORQXIQZAOLNGE-UHFFFAOYSA-N 0.000 description 2
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 2
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 description 2
- GFYHSKONPJXCDE-UHFFFAOYSA-N 2,3,5-trimethylpyridine Chemical compound CC1=CN=C(C)C(C)=C1 GFYHSKONPJXCDE-UHFFFAOYSA-N 0.000 description 2
- SWELIMKTDYHAOY-UHFFFAOYSA-N 2,4-diamino-6-hydroxypyrimidine Chemical compound NC1=CC(=O)N=C(N)N1 SWELIMKTDYHAOY-UHFFFAOYSA-N 0.000 description 2
- OFPWMRMIFDHXFE-UHFFFAOYSA-N 2-(bromomethyl)pyridine Chemical class BrCC1=CC=CC=N1 OFPWMRMIFDHXFE-UHFFFAOYSA-N 0.000 description 2
- HZAXFHJVJLSVMW-UHFFFAOYSA-N 2-Aminoethan-1-ol Chemical compound NCCO HZAXFHJVJLSVMW-UHFFFAOYSA-N 0.000 description 2
- WOXFMYVTSLAQMO-UHFFFAOYSA-N 2-Pyridinemethanamine Chemical class NCC1=CC=CC=N1 WOXFMYVTSLAQMO-UHFFFAOYSA-N 0.000 description 2
- WRYMKDLLJFJZCA-UHFFFAOYSA-N 2-[2-amino-4-chloro-7-[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]pyrrolo[2,3-d]pyrimidin-5-yl]ethyl methanesulfonate Chemical compound COC1=C(C)C=NC(CN2C3=NC(N)=NC(Cl)=C3C(CCOS(C)(=O)=O)=C2)=C1C WRYMKDLLJFJZCA-UHFFFAOYSA-N 0.000 description 2
- JKMHFZQWWAIEOD-UHFFFAOYSA-N 2-[4-(2-hydroxyethyl)piperazin-1-yl]ethanesulfonic acid Chemical compound OCC[NH+]1CCN(CCS([O-])(=O)=O)CC1 JKMHFZQWWAIEOD-UHFFFAOYSA-N 0.000 description 2
- RWVGDJMHCSPSPQ-UHFFFAOYSA-N 2-amino-4-chloro-7-[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]-5,5-dimethylpyrrolo[2,3-d]pyrimidin-6-one Chemical compound COC1=C(C)C=NC(CN2C3=C(C(=NC(N)=N3)Cl)C(C)(C)C2=O)=C1C RWVGDJMHCSPSPQ-UHFFFAOYSA-N 0.000 description 2
- QEACLLJJRTVPRI-UHFFFAOYSA-N 2-amino-5-[2-[tert-butyl(diphenyl)silyl]oxyethyl]-1,7-dihydropyrrolo[2,3-d]pyrimidin-4-one Chemical compound C=1NC=2N=C(N)NC(=O)C=2C=1CCO[Si](C(C)(C)C)(C=1C=CC=CC=1)C1=CC=CC=C1 QEACLLJJRTVPRI-UHFFFAOYSA-N 0.000 description 2
- JGLCDBJMIOJAIF-UHFFFAOYSA-N 2-bromo-4-[tert-butyl(diphenyl)silyl]oxybutanal Chemical compound C=1C=CC=CC=1[Si](OCCC(Br)C=O)(C(C)(C)C)C1=CC=CC=C1 JGLCDBJMIOJAIF-UHFFFAOYSA-N 0.000 description 2
- VHYFNPMBLIVWCW-UHFFFAOYSA-N 4-Dimethylaminopyridine Chemical compound CN(C)C1=CC=NC=C1 VHYFNPMBLIVWCW-UHFFFAOYSA-N 0.000 description 2
- WGBBKMOBGARXLF-UHFFFAOYSA-N 4-[tert-butyl(diphenyl)silyl]oxybutan-1-ol Chemical compound C=1C=CC=CC=1[Si](OCCCCO)(C(C)(C)C)C1=CC=CC=C1 WGBBKMOBGARXLF-UHFFFAOYSA-N 0.000 description 2
- FIGNMIFTOBCQOY-UHFFFAOYSA-N 4-[tert-butyl(diphenyl)silyl]oxybutanal Chemical compound C=1C=CC=CC=1[Si](OCCCC=O)(C(C)(C)C)C1=CC=CC=C1 FIGNMIFTOBCQOY-UHFFFAOYSA-N 0.000 description 2
- XJTFVTIYKKOOCN-UHFFFAOYSA-N 4-bromo-2,3,5-trimethyl-1-oxidopyridin-1-ium Chemical compound CC1=C[N+]([O-])=C(C)C(C)=C1Br XJTFVTIYKKOOCN-UHFFFAOYSA-N 0.000 description 2
- FEWCKGAHYXFBJY-UHFFFAOYSA-N 4-bromo-7-[(3,5-dimethoxyphenyl)methyl]pyrrolo[2,3-d]pyrimidin-2-amine Chemical compound COC1=CC(OC)=CC(CN2C3=NC(N)=NC(Br)=C3C=C2)=C1 FEWCKGAHYXFBJY-UHFFFAOYSA-N 0.000 description 2
- QWFFGACVYBYLBL-UHFFFAOYSA-N 4-chloro-5-[(dibenzylamino)methyl]-7-[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]pyrrolo[2,3-d]pyrimidin-2-amine Chemical compound COC1=C(C)C=NC(CN2C3=NC(N)=NC(Cl)=C3C(CN(CC=3C=CC=CC=3)CC=3C=CC=CC=3)=C2)=C1C QWFFGACVYBYLBL-UHFFFAOYSA-N 0.000 description 2
- QWMUZEWXOHTYEU-UHFFFAOYSA-N 4-chloro-7-[(2-chloro-4,5-dimethoxyphenyl)methyl]pyrrolo[2,3-d]pyrimidin-2-amine Chemical compound C1=C(OC)C(OC)=CC(Cl)=C1CN1C2=NC(N)=NC(Cl)=C2C=C1 QWMUZEWXOHTYEU-UHFFFAOYSA-N 0.000 description 2
- XWPASKSPLMPAFA-UHFFFAOYSA-N 4-chloro-7-[(3,4,5-trimethoxyphenyl)methyl]pyrrolo[2,3-d]pyrimidin-2-amine Chemical compound COC1=C(OC)C(OC)=CC(CN2C3=NC(N)=NC(Cl)=C3C=C2)=C1 XWPASKSPLMPAFA-UHFFFAOYSA-N 0.000 description 2
- OABBVRNSJWLUDO-UHFFFAOYSA-N 4-chloro-7-[(3,4-dichlorophenyl)methyl]pyrrolo[2,3-d]pyrimidin-2-amine Chemical compound C12=NC(N)=NC(Cl)=C2C=CN1CC1=CC=C(Cl)C(Cl)=C1 OABBVRNSJWLUDO-UHFFFAOYSA-N 0.000 description 2
- JJHVDKYZVGILPX-UHFFFAOYSA-N 4-chloro-7-[(3,5-dimethoxyphenyl)methyl]pyrrolo[2,3-d]pyrimidin-2-amine Chemical compound COC1=CC(OC)=CC(CN2C3=NC(N)=NC(Cl)=C3C=C2)=C1 JJHVDKYZVGILPX-UHFFFAOYSA-N 0.000 description 2
- VQFMBSGPLNPGDM-UHFFFAOYSA-N 4-chloro-7-[(3-methoxyphenyl)methyl]pyrrolo[2,3-d]pyrimidin-2-amine Chemical compound COC1=CC=CC(CN2C3=NC(N)=NC(Cl)=C3C=C2)=C1 VQFMBSGPLNPGDM-UHFFFAOYSA-N 0.000 description 2
- JXIKWDVFEDDBEF-UHFFFAOYSA-N 4-chloro-7-[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]-5-[(n-methylanilino)methyl]pyrrolo[2,3-d]pyrimidin-2-amine Chemical compound COC1=C(C)C=NC(CN2C3=NC(N)=NC(Cl)=C3C(CN(C)C=3C=CC=CC=3)=C2)=C1C JXIKWDVFEDDBEF-UHFFFAOYSA-N 0.000 description 2
- AMATXUCYHHHHHB-UHFFFAOYSA-N 5,5-dibromo-1,3-diazinane-2,4,6-trione Chemical compound BrC1(Br)C(=O)NC(=O)NC1=O AMATXUCYHHHHHB-UHFFFAOYSA-N 0.000 description 2
- BIVSHTFKYFJPBM-UHFFFAOYSA-N 5-(anilinomethyl)-4-chloro-7-[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]pyrrolo[2,3-d]pyrimidin-2-amine Chemical compound COC1=C(C)C=NC(CN2C3=NC(N)=NC(Cl)=C3C(CNC=3C=CC=CC=3)=C2)=C1C BIVSHTFKYFJPBM-UHFFFAOYSA-N 0.000 description 2
- XXRUQNNAKXZSOS-UHFFFAOYSA-N 5-(chloromethyl)-1,2,3-trimethoxybenzene Chemical compound COC1=CC(CCl)=CC(OC)=C1OC XXRUQNNAKXZSOS-UHFFFAOYSA-N 0.000 description 2
- KQQKHXJWTLMVNX-UHFFFAOYSA-N 5-[2-[tert-butyl(diphenyl)silyl]oxyethyl]-4-chloro-7-[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]pyrrolo[2,3-d]pyrimidin-2-amine Chemical compound COC1=C(C)C=NC(CN2C3=NC(N)=NC(Cl)=C3C(CCO[Si](C=3C=CC=CC=3)(C=3C=CC=CC=3)C(C)(C)C)=C2)=C1C KQQKHXJWTLMVNX-UHFFFAOYSA-N 0.000 description 2
- RXGJTUSBYWCRBK-UHFFFAOYSA-M 5-methylphenazinium methyl sulfate Chemical compound COS([O-])(=O)=O.C1=CC=C2[N+](C)=C(C=CC=C3)C3=NC2=C1 RXGJTUSBYWCRBK-UHFFFAOYSA-M 0.000 description 2
- NOHITWUPXFNNDC-UHFFFAOYSA-N 7-[(2-bromo-3,4,5-trimethoxyphenyl)methyl]-4-chloropyrrolo[2,3-d]pyrimidin-2-amine Chemical compound COC1=C(OC)C(OC)=CC(CN2C3=NC(N)=NC(Cl)=C3C=C2)=C1Br NOHITWUPXFNNDC-UHFFFAOYSA-N 0.000 description 2
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 2
- 235000003911 Arachis Nutrition 0.000 description 2
- 244000105624 Arachis hypogaea Species 0.000 description 2
- 241000416162 Astragalus gummifer Species 0.000 description 2
- 238000007045 Balz-Schiemann reaction Methods 0.000 description 2
- 108010006654 Bleomycin Proteins 0.000 description 2
- NFCFFILMVCFXLM-UHFFFAOYSA-N COC1=C(C)C=NC(CN2C=3C(C(=NC(=N)N=3)Cl)=C(O)C2=O)=C1C Chemical compound COC1=C(C)C=NC(CN2C=3C(C(=NC(=N)N=3)Cl)=C(O)C2=O)=C1C NFCFFILMVCFXLM-UHFFFAOYSA-N 0.000 description 2
- LMRYOPDATKLWEH-UHFFFAOYSA-N COC1=C(OC)C(OC)=CC(CN2C=3C(C(=NC(=N)N=3)Cl)=C(O)C2=O)=C1 Chemical compound COC1=C(OC)C(OC)=CC(CN2C=3C(C(=NC(=N)N=3)Cl)=C(O)C2=O)=C1 LMRYOPDATKLWEH-UHFFFAOYSA-N 0.000 description 2
- 239000004215 Carbon black (E152) Substances 0.000 description 2
- 208000035473 Communicable disease Diseases 0.000 description 2
- 229920002261 Corn starch Polymers 0.000 description 2
- 108010025464 Cyclin-Dependent Kinase 4 Proteins 0.000 description 2
- 102000013701 Cyclin-Dependent Kinase 4 Human genes 0.000 description 2
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 2
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 2
- HMFHBZSHGGEWLO-SOOFDHNKSA-N D-ribofuranose Chemical compound OC[C@H]1OC(O)[C@H](O)[C@@H]1O HMFHBZSHGGEWLO-SOOFDHNKSA-N 0.000 description 2
- OKKJLVBELUTLKV-MZCSYVLQSA-N Deuterated methanol Chemical compound [2H]OC([2H])([2H])[2H] OKKJLVBELUTLKV-MZCSYVLQSA-N 0.000 description 2
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 2
- 239000001856 Ethyl cellulose Substances 0.000 description 2
- ZZSNKZQZMQGXPY-UHFFFAOYSA-N Ethyl cellulose Chemical compound CCOCC1OC(OC)C(OCC)C(OCC)C1OC1C(O)C(O)C(OC)C(CO)O1 ZZSNKZQZMQGXPY-UHFFFAOYSA-N 0.000 description 2
- QUSNBJAOOMFDIB-UHFFFAOYSA-N Ethylamine Chemical compound CCN QUSNBJAOOMFDIB-UHFFFAOYSA-N 0.000 description 2
- 239000007995 HEPES buffer Substances 0.000 description 2
- MCAHMSDENAOJFZ-UHFFFAOYSA-N Herbimycin A Natural products N1C(=O)C(C)=CC=CC(OC)C(OC(N)=O)C(C)=CC(C)C(OC)C(OC)CC(C)C(OC)C2=CC(=O)C=C1C2=O MCAHMSDENAOJFZ-UHFFFAOYSA-N 0.000 description 2
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 2
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 2
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 2
- 239000002841 Lewis acid Substances 0.000 description 2
- TWRXJAOTZQYOKJ-UHFFFAOYSA-L Magnesium chloride Chemical compound [Mg+2].[Cl-].[Cl-] TWRXJAOTZQYOKJ-UHFFFAOYSA-L 0.000 description 2
- NWIBSHFKIJFRCO-WUDYKRTCSA-N Mytomycin Chemical compound C1N2C(C(C(C)=C(N)C3=O)=O)=C3[C@@H](COC(N)=O)[C@@]2(OC)[C@@H]2[C@H]1N2 NWIBSHFKIJFRCO-WUDYKRTCSA-N 0.000 description 2
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 2
- ZSXGLVDWWRXATF-UHFFFAOYSA-N N,N-dimethylformamide dimethyl acetal Chemical compound COC(OC)N(C)C ZSXGLVDWWRXATF-UHFFFAOYSA-N 0.000 description 2
- AFBPFSWMIHJQDM-UHFFFAOYSA-N N-methylaniline Chemical compound CNC1=CC=CC=C1 AFBPFSWMIHJQDM-UHFFFAOYSA-N 0.000 description 2
- 150000001204 N-oxides Chemical class 0.000 description 2
- 229910004064 NOBF4 Inorganic materials 0.000 description 2
- 108010058765 Oncogene Protein pp60(v-src) Proteins 0.000 description 2
- 235000019483 Peanut oil Nutrition 0.000 description 2
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 2
- GLUUGHFHXGJENI-UHFFFAOYSA-N Piperazine Chemical compound C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 description 2
- 239000004698 Polyethylene Substances 0.000 description 2
- CZPWVGJYEJSRLH-UHFFFAOYSA-N Pyrimidine Chemical compound C1=CN=CN=C1 CZPWVGJYEJSRLH-UHFFFAOYSA-N 0.000 description 2
- 102100033479 RAF proto-oncogene serine/threonine-protein kinase Human genes 0.000 description 2
- PYMYPHUHKUWMLA-LMVFSUKVSA-N Ribose Natural products OC[C@@H](O)[C@@H](O)[C@@H](O)C=O PYMYPHUHKUWMLA-LMVFSUKVSA-N 0.000 description 2
- 238000000297 Sandmeyer reaction Methods 0.000 description 2
- 229910018162 SeO2 Inorganic materials 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- 238000003477 Sonogashira cross-coupling reaction Methods 0.000 description 2
- UCKMPCXJQFINFW-UHFFFAOYSA-N Sulphide Chemical compound [S-2] UCKMPCXJQFINFW-UHFFFAOYSA-N 0.000 description 2
- DKGAVHZHDRPRBM-UHFFFAOYSA-N Tert-Butanol Chemical compound CC(C)(C)O DKGAVHZHDRPRBM-UHFFFAOYSA-N 0.000 description 2
- 229920001615 Tragacanth Polymers 0.000 description 2
- 108091008605 VEGF receptors Proteins 0.000 description 2
- 102000009484 Vascular Endothelial Growth Factor Receptors Human genes 0.000 description 2
- JXLYSJRDGCGARV-WWYNWVTFSA-N Vinblastine Natural products O=C(O[C@H]1[C@](O)(C(=O)OC)[C@@H]2N(C)c3c(cc(c(OC)c3)[C@]3(C(=O)OC)c4[nH]c5c(c4CCN4C[C@](O)(CC)C[C@H](C3)C4)cccc5)[C@@]32[C@H]2[C@@]1(CC)C=CCN2CC3)C JXLYSJRDGCGARV-WWYNWVTFSA-N 0.000 description 2
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 2
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 2
- 125000002252 acyl group Chemical group 0.000 description 2
- 125000004423 acyloxy group Chemical group 0.000 description 2
- 150000001299 aldehydes Chemical class 0.000 description 2
- 239000000783 alginic acid Substances 0.000 description 2
- 229960001126 alginic acid Drugs 0.000 description 2
- 125000004183 alkoxy alkyl group Chemical group 0.000 description 2
- HMFHBZSHGGEWLO-UHFFFAOYSA-N alpha-D-Furanose-Ribose Natural products OCC1OC(O)C(O)C1O HMFHBZSHGGEWLO-UHFFFAOYSA-N 0.000 description 2
- WNROFYMDJYEPJX-UHFFFAOYSA-K aluminium hydroxide Chemical compound [OH-].[OH-].[OH-].[Al+3] WNROFYMDJYEPJX-UHFFFAOYSA-K 0.000 description 2
- 150000001408 amides Chemical class 0.000 description 2
- 230000003321 amplification Effects 0.000 description 2
- 239000003242 anti bacterial agent Substances 0.000 description 2
- 229940088710 antibiotic agent Drugs 0.000 description 2
- 229940045696 antineoplastic drug podophyllotoxin derivative Drugs 0.000 description 2
- 150000001491 aromatic compounds Chemical class 0.000 description 2
- 125000005160 aryl oxy alkyl group Chemical group 0.000 description 2
- 150000005840 aryl radicals Chemical class 0.000 description 2
- 125000004104 aryloxy group Chemical group 0.000 description 2
- 239000011324 bead Substances 0.000 description 2
- 230000009286 beneficial effect Effects 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- HUMNYLRZRPPJDN-UHFFFAOYSA-N benzaldehyde Chemical compound O=CC1=CC=CC=C1 HUMNYLRZRPPJDN-UHFFFAOYSA-N 0.000 description 2
- IOJUPLGTWVMSFF-UHFFFAOYSA-N benzothiazole Chemical compound C1=CC=C2SC=NC2=C1 IOJUPLGTWVMSFF-UHFFFAOYSA-N 0.000 description 2
- 125000001743 benzylic group Chemical group 0.000 description 2
- OYVAGSVQBOHSSS-UAPAGMARSA-O bleomycin A2 Chemical class N([C@H](C(=O)N[C@H](C)[C@@H](O)[C@H](C)C(=O)N[C@@H]([C@H](O)C)C(=O)NCCC=1SC=C(N=1)C=1SC=C(N=1)C(=O)NCCC[S+](C)C)[C@@H](O[C@H]1[C@H]([C@@H](O)[C@H](O)[C@H](CO)O1)O[C@@H]1[C@H]([C@@H](OC(N)=O)[C@H](O)[C@@H](CO)O1)O)C=1N=CNC=1)C(=O)C1=NC([C@H](CC(N)=O)NC[C@H](N)C(N)=O)=NC(N)=C1C OYVAGSVQBOHSSS-UAPAGMARSA-O 0.000 description 2
- QARVLSVVCXYDNA-UHFFFAOYSA-N bromobenzene Chemical compound BrC1=CC=CC=C1 QARVLSVVCXYDNA-UHFFFAOYSA-N 0.000 description 2
- WERYXYBDKMZEQL-UHFFFAOYSA-N butane-1,4-diol Chemical compound OCCCCO WERYXYBDKMZEQL-UHFFFAOYSA-N 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 2
- CREMABGTGYGIQB-UHFFFAOYSA-N carbon carbon Chemical compound C.C CREMABGTGYGIQB-UHFFFAOYSA-N 0.000 description 2
- 239000011203 carbon fibre reinforced carbon Substances 0.000 description 2
- 125000005518 carboxamido group Chemical group 0.000 description 2
- 239000001768 carboxy methyl cellulose Substances 0.000 description 2
- 150000001768 cations Chemical class 0.000 description 2
- 239000006285 cell suspension Substances 0.000 description 2
- 235000010980 cellulose Nutrition 0.000 description 2
- 229920002678 cellulose Polymers 0.000 description 2
- 239000001913 cellulose Substances 0.000 description 2
- 238000012512 characterization method Methods 0.000 description 2
- 235000017168 chlorine Nutrition 0.000 description 2
- 229940110456 cocoa butter Drugs 0.000 description 2
- 235000019868 cocoa butter Nutrition 0.000 description 2
- 238000012875 competitive assay Methods 0.000 description 2
- 238000009833 condensation Methods 0.000 description 2
- 230000005494 condensation Effects 0.000 description 2
- 229910052802 copper Inorganic materials 0.000 description 2
- 239000010949 copper Substances 0.000 description 2
- 239000008120 corn starch Substances 0.000 description 2
- 239000006071 cream Substances 0.000 description 2
- 125000000753 cycloalkyl group Chemical group 0.000 description 2
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 2
- 125000000298 cyclopropenyl group Chemical group [H]C1=C([H])C1([H])* 0.000 description 2
- 238000010511 deprotection reaction Methods 0.000 description 2
- 238000013461 design Methods 0.000 description 2
- 238000001514 detection method Methods 0.000 description 2
- 238000010790 dilution Methods 0.000 description 2
- 239000012895 dilution Substances 0.000 description 2
- 230000006806 disease prevention Effects 0.000 description 2
- 239000003480 eluent Substances 0.000 description 2
- 239000000839 emulsion Substances 0.000 description 2
- 235000019441 ethanol Nutrition 0.000 description 2
- 235000019325 ethyl cellulose Nutrition 0.000 description 2
- 229920001249 ethyl cellulose Polymers 0.000 description 2
- 230000005284 excitation Effects 0.000 description 2
- 235000019000 fluorine Nutrition 0.000 description 2
- 125000001153 fluoro group Chemical group F* 0.000 description 2
- 239000012634 fragment Substances 0.000 description 2
- 239000007903 gelatin capsule Substances 0.000 description 2
- 239000008187 granular material Substances 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- MCAHMSDENAOJFZ-BVXDHVRPSA-N herbimycin Chemical compound N1C(=O)\C(C)=C\C=C/[C@H](OC)[C@@H](OC(N)=O)\C(C)=C\[C@H](C)[C@@H](OC)[C@@H](OC)C[C@H](C)[C@@H](OC)C2=CC(=O)C=C1C2=O MCAHMSDENAOJFZ-BVXDHVRPSA-N 0.000 description 2
- 125000005368 heteroarylthio group Chemical group 0.000 description 2
- BXWNKGSJHAJOGX-UHFFFAOYSA-N hexadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCO BXWNKGSJHAJOGX-UHFFFAOYSA-N 0.000 description 2
- GNOIPBMMFNIUFM-UHFFFAOYSA-N hexamethylphosphoric triamide Chemical compound CN(C)P(=O)(N(C)C)N(C)C GNOIPBMMFNIUFM-UHFFFAOYSA-N 0.000 description 2
- 238000009396 hybridization Methods 0.000 description 2
- 238000006197 hydroboration reaction Methods 0.000 description 2
- 229930195733 hydrocarbon Natural products 0.000 description 2
- NPZTUJOABDZTLV-UHFFFAOYSA-N hydroxybenzotriazole Substances O=C1C=CC=C2NNN=C12 NPZTUJOABDZTLV-UHFFFAOYSA-N 0.000 description 2
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 description 2
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 description 2
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 description 2
- UFVKGYZPFZQRLF-UHFFFAOYSA-N hydroxypropyl methyl cellulose Chemical compound OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC2C(C(O)C(OC3C(C(O)C(O)C(CO)O3)O)C(CO)O2)O)C(CO)O1 UFVKGYZPFZQRLF-UHFFFAOYSA-N 0.000 description 2
- 239000007943 implant Substances 0.000 description 2
- 238000001727 in vivo Methods 0.000 description 2
- 238000011065 in-situ storage Methods 0.000 description 2
- 238000010348 incorporation Methods 0.000 description 2
- 238000011534 incubation Methods 0.000 description 2
- 238000007918 intramuscular administration Methods 0.000 description 2
- 238000002955 isolation Methods 0.000 description 2
- HPIGCVXMBGOWTF-UHFFFAOYSA-N isomaltol Chemical compound CC(=O)C=1OC=CC=1O HPIGCVXMBGOWTF-UHFFFAOYSA-N 0.000 description 2
- 125000002183 isoquinolinyl group Chemical class C1(=NC=CC2=CC=CC=C12)* 0.000 description 2
- 239000008101 lactose Substances 0.000 description 2
- 235000010445 lecithin Nutrition 0.000 description 2
- 239000000787 lecithin Substances 0.000 description 2
- 229940067606 lecithin Drugs 0.000 description 2
- 150000007517 lewis acids Chemical class 0.000 description 2
- 239000003446 ligand Substances 0.000 description 2
- 125000005647 linker group Chemical group 0.000 description 2
- 239000002502 liposome Substances 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- YNESATAKKCNGOF-UHFFFAOYSA-N lithium bis(trimethylsilyl)amide Chemical compound [Li+].C[Si](C)(C)[N-][Si](C)(C)C YNESATAKKCNGOF-UHFFFAOYSA-N 0.000 description 2
- 239000006166 lysate Substances 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 231100000682 maximum tolerated dose Toxicity 0.000 description 2
- 239000002609 medium Substances 0.000 description 2
- SGDBTWWWUNNDEQ-LBPRGKRZSA-N melphalan Chemical compound OC(=O)[C@@H](N)CC1=CC=C(N(CCCl)CCCl)C=C1 SGDBTWWWUNNDEQ-LBPRGKRZSA-N 0.000 description 2
- 229960001924 melphalan Drugs 0.000 description 2
- 239000012528 membrane Substances 0.000 description 2
- GLVAUDGFNGKCSF-UHFFFAOYSA-N mercaptopurine Chemical compound S=C1NC=NC2=C1NC=N2 GLVAUDGFNGKCSF-UHFFFAOYSA-N 0.000 description 2
- 229910052751 metal Inorganic materials 0.000 description 2
- 239000002184 metal Substances 0.000 description 2
- QARBMVPHQWIHKH-UHFFFAOYSA-N methanesulfonyl chloride Chemical compound CS(Cl)(=O)=O QARBMVPHQWIHKH-UHFFFAOYSA-N 0.000 description 2
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 2
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 2
- 239000002480 mineral oil Substances 0.000 description 2
- 235000010446 mineral oil Nutrition 0.000 description 2
- RCTXKMDREXEBSU-UHFFFAOYSA-N n-[4-chloro-5-[(dibenzylamino)methyl]-7h-pyrrolo[2,3-d]pyrimidin-2-yl]octanamide Chemical compound C=1NC2=NC(NC(=O)CCCCCCC)=NC(Cl)=C2C=1CN(CC=1C=CC=CC=1)CC1=CC=CC=C1 RCTXKMDREXEBSU-UHFFFAOYSA-N 0.000 description 2
- SFFVAIRPVGDCMO-UHFFFAOYSA-N n-[4-chloro-5-[(n-methylanilino)methyl]-7h-pyrrolo[2,3-d]pyrimidin-2-yl]octanamide Chemical compound C=1NC2=NC(NC(=O)CCCCCCC)=NC(Cl)=C2C=1CN(C)C1=CC=CC=C1 SFFVAIRPVGDCMO-UHFFFAOYSA-N 0.000 description 2
- TVCDBMPMXKLKGL-UHFFFAOYSA-N n-[4-chloro-5-iodo-7-[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]pyrrolo[2,3-d]pyrimidin-2-yl]-2,2-dimethylpropanamide Chemical compound COC1=C(C)C=NC(CN2C3=NC(NC(=O)C(C)(C)C)=NC(Cl)=C3C(I)=C2)=C1C TVCDBMPMXKLKGL-UHFFFAOYSA-N 0.000 description 2
- SGMPXYBFAZJVAV-UHFFFAOYSA-N n-[5-(anilinomethyl)-4-chloro-7h-pyrrolo[2,3-d]pyrimidin-2-yl]octanamide Chemical compound C=1NC2=NC(NC(=O)CCCCCCC)=NC(Cl)=C2C=1CNC1=CC=CC=C1 SGMPXYBFAZJVAV-UHFFFAOYSA-N 0.000 description 2
- APLCHODOBFLXLN-UHFFFAOYSA-N n-[5-[2-[tert-butyl(diphenyl)silyl]oxyethyl]-4-chloro-7-[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]pyrrolo[2,3-d]pyrimidin-2-yl]acetamide Chemical compound COC1=C(C)C=NC(CN2C3=NC(NC(C)=O)=NC(Cl)=C3C(CCO[Si](C=3C=CC=CC=3)(C=3C=CC=CC=3)C(C)(C)C)=C2)=C1C APLCHODOBFLXLN-UHFFFAOYSA-N 0.000 description 2
- QSGQVHOVXMKGCZ-UHFFFAOYSA-N n-[5-[2-[tert-butyl(diphenyl)silyl]oxyethyl]-4-chloro-7h-pyrrolo[2,3-d]pyrimidin-2-yl]acetamide Chemical compound C=1NC2=NC(NC(=O)C)=NC(Cl)=C2C=1CCO[Si](C(C)(C)C)(C=1C=CC=CC=1)C1=CC=CC=C1 QSGQVHOVXMKGCZ-UHFFFAOYSA-N 0.000 description 2
- HUAFOQNMDYHQGE-UHFFFAOYSA-N n-[7-acetyl-5-[2-[tert-butyl(diphenyl)silyl]oxyethyl]-4-chloropyrrolo[2,3-d]pyrimidin-2-yl]acetamide Chemical compound C=1N(C(C)=O)C2=NC(NC(=O)C)=NC(Cl)=C2C=1CCO[Si](C(C)(C)C)(C=1C=CC=CC=1)C1=CC=CC=C1 HUAFOQNMDYHQGE-UHFFFAOYSA-N 0.000 description 2
- UAZPXAHOHCWSSF-UHFFFAOYSA-N n-[7-acetyl-5-[2-[tert-butyl(diphenyl)silyl]oxyethyl]-4-oxo-1h-pyrrolo[2,3-d]pyrimidin-2-yl]acetamide Chemical compound C1=2C(=O)NC(NC(=O)C)=NC=2N(C(C)=O)C=C1CCO[Si](C(C)(C)C)(C=1C=CC=CC=1)C1=CC=CC=C1 UAZPXAHOHCWSSF-UHFFFAOYSA-N 0.000 description 2
- 230000010807 negative regulation of binding Effects 0.000 description 2
- 229910052759 nickel Inorganic materials 0.000 description 2
- PXHVJJICTQNCMI-UHFFFAOYSA-N nickel Substances [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 2
- 239000000346 nonvolatile oil Substances 0.000 description 2
- 238000003199 nucleic acid amplification method Methods 0.000 description 2
- 238000007899 nucleic acid hybridization Methods 0.000 description 2
- 239000012038 nucleophile Substances 0.000 description 2
- 239000002674 ointment Substances 0.000 description 2
- CTSLXHKWHWQRSH-UHFFFAOYSA-N oxalyl chloride Chemical compound ClC(=O)C(Cl)=O CTSLXHKWHWQRSH-UHFFFAOYSA-N 0.000 description 2
- 230000001590 oxidative effect Effects 0.000 description 2
- 230000007170 pathology Effects 0.000 description 2
- 239000000312 peanut oil Substances 0.000 description 2
- 229940124531 pharmaceutical excipient Drugs 0.000 description 2
- 229910052698 phosphorus Inorganic materials 0.000 description 2
- 239000011574 phosphorus Substances 0.000 description 2
- YJGVMLPVUAXIQN-XVVDYKMHSA-N podophyllotoxin Chemical compound COC1=C(OC)C(OC)=CC([C@@H]2C3=CC=4OCOC=4C=C3[C@H](O)[C@@H]3[C@@H]2C(OC3)=O)=C1 YJGVMLPVUAXIQN-XVVDYKMHSA-N 0.000 description 2
- 239000003600 podophyllotoxin derivative Substances 0.000 description 2
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 2
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 2
- 239000013641 positive control Substances 0.000 description 2
- IUBQJLUDMLPAGT-UHFFFAOYSA-N potassium bis(trimethylsilyl)amide Chemical compound C[Si](C)(C)N([K])[Si](C)(C)C IUBQJLUDMLPAGT-UHFFFAOYSA-N 0.000 description 2
- QELSKZZBTMNZEB-UHFFFAOYSA-N propylparaben Chemical compound CCCOC(=O)C1=CC=C(O)C=C1 QELSKZZBTMNZEB-UHFFFAOYSA-N 0.000 description 2
- 238000000159 protein binding assay Methods 0.000 description 2
- 125000000561 purinyl group Chemical group N1=C(N=C2N=CNC2=C1)* 0.000 description 2
- 125000003373 pyrazinyl group Chemical group 0.000 description 2
- KEYZUMLVDCHSTP-UHFFFAOYSA-N pyridin-2-ylmethyl acetate Chemical compound CC(=O)OCC1=CC=CC=N1 KEYZUMLVDCHSTP-UHFFFAOYSA-N 0.000 description 2
- JQRYUMGHOUYJFW-UHFFFAOYSA-N pyridine;trihydrobromide Chemical compound [Br-].[Br-].[Br-].C1=CC=[NH+]C=C1.C1=CC=[NH+]C=C1.C1=CC=[NH+]C=C1 JQRYUMGHOUYJFW-UHFFFAOYSA-N 0.000 description 2
- 125000000714 pyrimidinyl group Chemical group 0.000 description 2
- 125000000168 pyrrolyl group Chemical group 0.000 description 2
- 150000003254 radicals Chemical class 0.000 description 2
- 230000001105 regulatory effect Effects 0.000 description 2
- ATEBXHFBFRCZMA-VXTBVIBXSA-N rifabutin Chemical compound O([C@](C1=O)(C)O/C=C/[C@@H]([C@H]([C@@H](OC(C)=O)[C@H](C)[C@H](O)[C@H](C)[C@@H](O)[C@@H](C)\C=C\C=C(C)/C(=O)NC(=C2N3)C(=O)C=4C(O)=C5C)C)OC)C5=C1C=4C2=NC13CCN(CC(C)C)CC1 ATEBXHFBFRCZMA-VXTBVIBXSA-N 0.000 description 2
- 229960000885 rifabutin Drugs 0.000 description 2
- 229920006395 saturated elastomer Polymers 0.000 description 2
- 239000008159 sesame oil Substances 0.000 description 2
- 235000011803 sesame oil Nutrition 0.000 description 2
- 102000034285 signal transducing proteins Human genes 0.000 description 2
- 108091006024 signal transducing proteins Proteins 0.000 description 2
- 230000019491 signal transduction Effects 0.000 description 2
- 239000000741 silica gel Substances 0.000 description 2
- 229910002027 silica gel Inorganic materials 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 235000015424 sodium Nutrition 0.000 description 2
- 229910052708 sodium Inorganic materials 0.000 description 2
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 description 2
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 description 2
- 235000011069 sorbitan monooleate Nutrition 0.000 description 2
- 239000001593 sorbitan monooleate Substances 0.000 description 2
- 229940035049 sorbitan monooleate Drugs 0.000 description 2
- 235000010356 sorbitol Nutrition 0.000 description 2
- 239000000600 sorbitol Substances 0.000 description 2
- 239000003549 soybean oil Substances 0.000 description 2
- 235000012424 soybean oil Nutrition 0.000 description 2
- 241000894007 species Species 0.000 description 2
- 239000008107 starch Substances 0.000 description 2
- 102000005969 steroid hormone receptors Human genes 0.000 description 2
- 238000007920 subcutaneous administration Methods 0.000 description 2
- 150000008163 sugars Chemical class 0.000 description 2
- 239000000829 suppository Substances 0.000 description 2
- 238000001356 surgical procedure Methods 0.000 description 2
- 239000006188 syrup Substances 0.000 description 2
- 235000020357 syrup Nutrition 0.000 description 2
- 239000000454 talc Substances 0.000 description 2
- 235000012222 talc Nutrition 0.000 description 2
- 229910052623 talc Inorganic materials 0.000 description 2
- 150000003512 tertiary amines Chemical class 0.000 description 2
- DPKBAXPHAYBPRL-UHFFFAOYSA-M tetrabutylazanium;iodide Chemical compound [I-].CCCC[N+](CCCC)(CCCC)CCCC DPKBAXPHAYBPRL-UHFFFAOYSA-M 0.000 description 2
- 125000003831 tetrazolyl group Chemical group 0.000 description 2
- 125000001544 thienyl group Chemical group 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- 238000000844 transformation Methods 0.000 description 2
- ITMCEJHCFYSIIV-UHFFFAOYSA-M triflate Chemical compound [O-]S(=O)(=O)C(F)(F)F ITMCEJHCFYSIIV-UHFFFAOYSA-M 0.000 description 2
- 229910052725 zinc Inorganic materials 0.000 description 2
- 239000011701 zinc Substances 0.000 description 2
- GVJHHUAWPYXKBD-IEOSBIPESA-N α-tocopherol Chemical compound OC1=C(C)C(C)=C2O[C@@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-IEOSBIPESA-N 0.000 description 2
- AADVCYNFEREWOS-UHFFFAOYSA-N (+)-DDM Natural products C=CC=CC(C)C(OC(N)=O)C(C)C(O)C(C)CC(C)=CC(C)C(O)C(C)C=CC(O)CC1OC(=O)C(C)C(O)C1C AADVCYNFEREWOS-UHFFFAOYSA-N 0.000 description 1
- LSPHULWDVZXLIL-UHFFFAOYSA-N (+/-)-Camphoric acid Chemical compound CC1(C)C(C(O)=O)CCC1(C)C(O)=O LSPHULWDVZXLIL-UHFFFAOYSA-N 0.000 description 1
- LNAZSHAWQACDHT-XIYTZBAFSA-N (2r,3r,4s,5r,6s)-4,5-dimethoxy-2-(methoxymethyl)-3-[(2s,3r,4s,5r,6r)-3,4,5-trimethoxy-6-(methoxymethyl)oxan-2-yl]oxy-6-[(2r,3r,4s,5r,6r)-4,5,6-trimethoxy-2-(methoxymethyl)oxan-3-yl]oxyoxane Chemical compound CO[C@@H]1[C@@H](OC)[C@H](OC)[C@@H](COC)O[C@H]1O[C@H]1[C@H](OC)[C@@H](OC)[C@H](O[C@H]2[C@@H]([C@@H](OC)[C@H](OC)O[C@@H]2COC)OC)O[C@@H]1COC LNAZSHAWQACDHT-XIYTZBAFSA-N 0.000 description 1
- MCEHFIXEKNKSRW-LBPRGKRZSA-N (2s)-2-[[3,5-dichloro-4-[(2,4-diaminopteridin-6-yl)methyl-methylamino]benzoyl]amino]pentanedioic acid Chemical compound C=1N=C2N=C(N)N=C(N)C2=NC=1CN(C)C1=C(Cl)C=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1Cl MCEHFIXEKNKSRW-LBPRGKRZSA-N 0.000 description 1
- YUPUSBMJCFBHAP-UHFFFAOYSA-N (3,4,5-trimethoxyphenyl)methanamine Chemical compound COC1=CC(CN)=CC(OC)=C1OC YUPUSBMJCFBHAP-UHFFFAOYSA-N 0.000 description 1
- CNPVFVHYBKQINB-UHFFFAOYSA-N (4-methoxy-3,5-dimethylpyridin-2-yl)methanamine Chemical compound COC1=C(C)C=NC(CN)=C1C CNPVFVHYBKQINB-UHFFFAOYSA-N 0.000 description 1
- OMJKFYKNWZZKTK-POHAHGRESA-N (5z)-5-(dimethylaminohydrazinylidene)imidazole-4-carboxamide Chemical compound CN(C)N\N=C1/N=CN=C1C(N)=O OMJKFYKNWZZKTK-POHAHGRESA-N 0.000 description 1
- WRIDQFICGBMAFQ-UHFFFAOYSA-N (E)-8-Octadecenoic acid Natural products CCCCCCCCCC=CCCCCCCC(O)=O WRIDQFICGBMAFQ-UHFFFAOYSA-N 0.000 description 1
- LKJPYSCBVHEWIU-KRWDZBQOSA-N (R)-bicalutamide Chemical compound C([C@@](O)(C)C(=O)NC=1C=C(C(C#N)=CC=1)C(F)(F)F)S(=O)(=O)C1=CC=C(F)C=C1 LKJPYSCBVHEWIU-KRWDZBQOSA-N 0.000 description 1
- SCYULBFZEHDVBN-UHFFFAOYSA-N 1,1-Dichloroethane Chemical compound CC(Cl)Cl SCYULBFZEHDVBN-UHFFFAOYSA-N 0.000 description 1
- FNQJDLTXOVEEFB-UHFFFAOYSA-N 1,2,3-benzothiadiazole Chemical compound C1=CC=C2SN=NC2=C1 FNQJDLTXOVEEFB-UHFFFAOYSA-N 0.000 description 1
- SLLFVLKNXABYGI-UHFFFAOYSA-N 1,2,3-benzoxadiazole Chemical compound C1=CC=C2ON=NC2=C1 SLLFVLKNXABYGI-UHFFFAOYSA-N 0.000 description 1
- 150000000183 1,3-benzoxazoles Chemical class 0.000 description 1
- ARKHLKHORHLJGX-UHFFFAOYSA-N 1-(bromomethyl)-2-chloro-4,5-dimethoxybenzene Chemical compound COC1=CC(Cl)=C(CBr)C=C1OC ARKHLKHORHLJGX-UHFFFAOYSA-N 0.000 description 1
- UEKFEYNZISYRRH-UHFFFAOYSA-N 1-(bromomethyl)-4,5-dimethoxy-2-nitrobenzene Chemical compound COC1=CC(CBr)=C([N+]([O-])=O)C=C1OC UEKFEYNZISYRRH-UHFFFAOYSA-N 0.000 description 1
- NTCULFTXJBHUAF-UHFFFAOYSA-N 1-(chloromethyl)-2-iodo-3,4,5-trimethoxybenzene Chemical compound COC1=CC(CCl)=C(I)C(OC)=C1OC NTCULFTXJBHUAF-UHFFFAOYSA-N 0.000 description 1
- CCAWDIFJOBKBSE-UHFFFAOYSA-N 1-(chloromethyl)-3,5-dimethoxybenzene Chemical compound COC1=CC(CCl)=CC(OC)=C1 CCAWDIFJOBKBSE-UHFFFAOYSA-N 0.000 description 1
- VGISFWWEOGVMED-UHFFFAOYSA-N 1-(chloromethyl)-3-methoxybenzene Chemical compound COC1=CC=CC(CCl)=C1 VGISFWWEOGVMED-UHFFFAOYSA-N 0.000 description 1
- MOHYOXXOKFQHDC-UHFFFAOYSA-N 1-(chloromethyl)-4-methoxybenzene Chemical compound COC1=CC=C(CCl)C=C1 MOHYOXXOKFQHDC-UHFFFAOYSA-N 0.000 description 1
- LMDZBCPBFSXMTL-UHFFFAOYSA-N 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide Substances CCN=C=NCCCN(C)C LMDZBCPBFSXMTL-UHFFFAOYSA-N 0.000 description 1
- 102100025573 1-alkyl-2-acetylglycerophosphocholine esterase Human genes 0.000 description 1
- JLPULHDHAOZNQI-ZTIMHPMXSA-N 1-hexadecanoyl-2-(9Z,12Z-octadecadienoyl)-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCC\C=C/C\C=C/CCCCC JLPULHDHAOZNQI-ZTIMHPMXSA-N 0.000 description 1
- IXPNQXFRVYWDDI-UHFFFAOYSA-N 1-methyl-2,4-dioxo-1,3-diazinane-5-carboximidamide Chemical compound CN1CC(C(N)=N)C(=O)NC1=O IXPNQXFRVYWDDI-UHFFFAOYSA-N 0.000 description 1
- HYZJCKYKOHLVJF-UHFFFAOYSA-N 1H-benzimidazole Chemical compound C1=CC=C2NC=NC2=C1 HYZJCKYKOHLVJF-UHFFFAOYSA-N 0.000 description 1
- APXRHPDHORGIEB-UHFFFAOYSA-N 1H-pyrazolo[4,3-d]pyrimidine Chemical class N1=CN=C2C=NNC2=C1 APXRHPDHORGIEB-UHFFFAOYSA-N 0.000 description 1
- SXFWKZNLYYRHMK-UHFFFAOYSA-N 1h-indolo[7,6-f]quinoline Chemical class C1=CC=C2C3=C(NC=C4)C4=CC=C3C=CC2=N1 SXFWKZNLYYRHMK-UHFFFAOYSA-N 0.000 description 1
- JVSFQJZRHXAUGT-UHFFFAOYSA-N 2,2-dimethylpropanoyl chloride Chemical compound CC(C)(C)C(Cl)=O JVSFQJZRHXAUGT-UHFFFAOYSA-N 0.000 description 1
- HMBHAQMOBKLWRX-UHFFFAOYSA-N 2,3-dihydro-1,4-benzodioxine-3-carboxylic acid Chemical compound C1=CC=C2OC(C(=O)O)COC2=C1 HMBHAQMOBKLWRX-UHFFFAOYSA-N 0.000 description 1
- NJBICLSFUUJIDH-UHFFFAOYSA-N 2-(chloromethyl)-1,4-dimethoxybenzene Chemical compound COC1=CC=C(OC)C(CCl)=C1 NJBICLSFUUJIDH-UHFFFAOYSA-N 0.000 description 1
- MWFCWXUIMDWPQD-UHFFFAOYSA-N 2-(chloromethyl)-3,5-dimethyl-1-oxidopyridin-1-ium Chemical compound CC1=CC(C)=C(CCl)[N+]([O-])=C1 MWFCWXUIMDWPQD-UHFFFAOYSA-N 0.000 description 1
- NJWIMFZLESWFIM-UHFFFAOYSA-N 2-(chloromethyl)pyridine Chemical class ClCC1=CC=CC=N1 NJWIMFZLESWFIM-UHFFFAOYSA-N 0.000 description 1
- WVAKRQOMAINQPU-UHFFFAOYSA-N 2-[4-[2-[5-(2,2-dimethylbutyl)-1h-imidazol-2-yl]ethyl]phenyl]pyridine Chemical compound N1C(CC(C)(C)CC)=CN=C1CCC1=CC=C(C=2N=CC=CC=2)C=C1 WVAKRQOMAINQPU-UHFFFAOYSA-N 0.000 description 1
- GOJUJUVQIVIZAV-UHFFFAOYSA-N 2-amino-4,6-dichloropyrimidine-5-carbaldehyde Chemical group NC1=NC(Cl)=C(C=O)C(Cl)=N1 GOJUJUVQIVIZAV-UHFFFAOYSA-N 0.000 description 1
- KGVGCKFEDOHXRF-UHFFFAOYSA-N 2-amino-4-chloro-7-[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]-5-methyl-5h-pyrrolo[2,3-d]pyrimidin-6-one Chemical compound COC1=C(C)C=NC(CN2C3=C(C(=NC(N)=N3)Cl)C(C)C2=O)=C1C KGVGCKFEDOHXRF-UHFFFAOYSA-N 0.000 description 1
- UQYUBOPIPNYYEX-UHFFFAOYSA-N 2-amino-4-chloro-7h-pyrrolo[2,3-d]pyrimidine-5,6-dione Chemical compound N1C(=O)C(O)=C2C1=NC(=N)N=C2Cl UQYUBOPIPNYYEX-UHFFFAOYSA-N 0.000 description 1
- HNEAWTLAJHJLIK-UHFFFAOYSA-N 2-bromo-1-(chloromethyl)-3,4,5-trimethoxybenzene Chemical compound COC1=CC(CCl)=C(Br)C(OC)=C1OC HNEAWTLAJHJLIK-UHFFFAOYSA-N 0.000 description 1
- 125000000069 2-butynyl group Chemical group [H]C([H])([H])C#CC([H])([H])* 0.000 description 1
- RYILSNRQSLTWPT-UHFFFAOYSA-N 2-chloro-1-(chloromethyl)-3,4,5-trimethoxybenzene Chemical compound COC1=CC(CCl)=C(Cl)C(OC)=C1OC RYILSNRQSLTWPT-UHFFFAOYSA-N 0.000 description 1
- 150000005759 2-chloropyridine Chemical class 0.000 description 1
- OKDGRDCXVWSXDC-UHFFFAOYSA-N 2-chloropyridine Chemical compound ClC1=CC=CC=N1 OKDGRDCXVWSXDC-UHFFFAOYSA-N 0.000 description 1
- KDSNLYIMUZNERS-UHFFFAOYSA-N 2-methylpropanamine Chemical compound CC(C)CN KDSNLYIMUZNERS-UHFFFAOYSA-N 0.000 description 1
- 125000000094 2-phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001494 2-propynyl group Chemical group [H]C#CC([H])([H])* 0.000 description 1
- LQJBNNIYVWPHFW-UHFFFAOYSA-N 20:1omega9c fatty acid Natural products CCCCCCCCCCC=CCCCCCCCC(O)=O LQJBNNIYVWPHFW-UHFFFAOYSA-N 0.000 description 1
- MGADZUXDNSDTHW-UHFFFAOYSA-N 2H-pyran Chemical compound C1OC=CC=C1 MGADZUXDNSDTHW-UHFFFAOYSA-N 0.000 description 1
- 125000004189 3,4-dichlorophenyl group Chemical group [H]C1=C([H])C(Cl)=C(Cl)C([H])=C1* 0.000 description 1
- NDMPLJNOPCLANR-UHFFFAOYSA-N 3,4-dihydroxy-15-(4-hydroxy-18-methoxycarbonyl-5,18-seco-ibogamin-18-yl)-16-methoxy-1-methyl-6,7-didehydro-aspidospermidine-3-carboxylic acid methyl ester Natural products C1C(CC)(O)CC(CC2(C(=O)OC)C=3C(=CC4=C(C56C(C(C(O)C7(CC)C=CCN(C67)CC5)(O)C(=O)OC)N4C)C=3)OC)CN1CCC1=C2NC2=CC=CC=C12 NDMPLJNOPCLANR-UHFFFAOYSA-N 0.000 description 1
- FPQQSJJWHUJYPU-UHFFFAOYSA-N 3-(dimethylamino)propyliminomethylidene-ethylazanium;chloride Chemical compound Cl.CCN=C=NCCCN(C)C FPQQSJJWHUJYPU-UHFFFAOYSA-N 0.000 description 1
- ZRPLANDPDWYOMZ-UHFFFAOYSA-N 3-cyclopentylpropionic acid Chemical compound OC(=O)CCC1CCCC1 ZRPLANDPDWYOMZ-UHFFFAOYSA-N 0.000 description 1
- XMIIGOLPHOKFCH-UHFFFAOYSA-M 3-phenylpropionate Chemical compound [O-]C(=O)CCC1=CC=CC=C1 XMIIGOLPHOKFCH-UHFFFAOYSA-M 0.000 description 1
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 1
- XLWSBDFQAJXCQX-UHFFFAOYSA-N 4-(bromomethyl)-1,2-dichlorobenzene Chemical compound ClC1=CC=C(CBr)C=C1Cl XLWSBDFQAJXCQX-UHFFFAOYSA-N 0.000 description 1
- APRZHQXAAWPYHS-UHFFFAOYSA-N 4-[5-[3-(carboxymethoxy)phenyl]-3-(4,5-dimethyl-1,3-thiazol-2-yl)tetrazol-3-ium-2-yl]benzenesulfonate Chemical compound S1C(C)=C(C)N=C1[N+]1=NC(C=2C=C(OCC(O)=O)C=CC=2)=NN1C1=CC=C(S([O-])(=O)=O)C=C1 APRZHQXAAWPYHS-UHFFFAOYSA-N 0.000 description 1
- GQGVBSHMRYHBTF-UOWFLXDJSA-N 4-amino-1-[(2r,4r,5r)-3,3-difluoro-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-1,3,5-triazin-2-one Chemical compound O=C1N=C(N)N=CN1[C@H]1C(F)(F)[C@H](O)[C@@H](CO)O1 GQGVBSHMRYHBTF-UOWFLXDJSA-N 0.000 description 1
- TVZGACDUOSZQKY-LBPRGKRZSA-N 4-aminofolic acid Chemical compound C1=NC2=NC(N)=NC(N)=C2N=C1CNC1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 TVZGACDUOSZQKY-LBPRGKRZSA-N 0.000 description 1
- LLFALGZRMACGJA-UHFFFAOYSA-N 4-chloro-7-[(2-iodo-3,4,5-trimethoxyphenyl)methyl]pyrrolo[2,3-d]pyrimidin-2-amine Chemical compound COC1=C(OC)C(OC)=CC(CN2C3=NC(N)=NC(Cl)=C3C=C2)=C1I LLFALGZRMACGJA-UHFFFAOYSA-N 0.000 description 1
- FDTTVIPMHKXALC-UHFFFAOYSA-N 4-chloro-7-[(4-chloro-3-methylpyridin-2-yl)methyl]-5-propan-2-ylpyrrolo[2,3-d]pyrimidin-2-amine Chemical compound C12=NC(N)=NC(Cl)=C2C(C(C)C)=CN1CC1=NC=CC(Cl)=C1C FDTTVIPMHKXALC-UHFFFAOYSA-N 0.000 description 1
- GIICXTUQQULGHY-UHFFFAOYSA-N 4-chloropyrrolo[2,3-d]pyrimidin-6-one Chemical compound ClC1=NC=NC2=NC(=O)C=C12 GIICXTUQQULGHY-UHFFFAOYSA-N 0.000 description 1
- 229960000549 4-dimethylaminophenol Drugs 0.000 description 1
- JGEGJYXHCFUMJF-UHFFFAOYSA-N 4-methylpentanal Chemical compound CC(C)CCC=O JGEGJYXHCFUMJF-UHFFFAOYSA-N 0.000 description 1
- FHVDTGUDJYJELY-UHFFFAOYSA-N 6-{[2-carboxy-4,5-dihydroxy-6-(phosphanyloxy)oxan-3-yl]oxy}-4,5-dihydroxy-3-phosphanyloxane-2-carboxylic acid Chemical compound O1C(C(O)=O)C(P)C(O)C(O)C1OC1C(C(O)=O)OC(OP)C(O)C1O FHVDTGUDJYJELY-UHFFFAOYSA-N 0.000 description 1
- STQGQHZAVUOBTE-UHFFFAOYSA-N 7-Cyan-hept-2t-en-4,6-diinsaeure Natural products C1=2C(O)=C3C(=O)C=4C(OC)=CC=CC=4C(=O)C3=C(O)C=2CC(O)(C(C)=O)CC1OC1CC(N)C(O)C(C)O1 STQGQHZAVUOBTE-UHFFFAOYSA-N 0.000 description 1
- QSBYPNXLFMSGKH-UHFFFAOYSA-N 9-Heptadecensaeure Natural products CCCCCCCC=CCCCCCCCC(O)=O QSBYPNXLFMSGKH-UHFFFAOYSA-N 0.000 description 1
- 108091006112 ATPases Proteins 0.000 description 1
- 244000215068 Acacia senegal Species 0.000 description 1
- 235000006491 Acacia senegal Nutrition 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 239000005964 Acibenzolar-S-methyl Substances 0.000 description 1
- 102000057290 Adenosine Triphosphatases Human genes 0.000 description 1
- 229920001817 Agar Polymers 0.000 description 1
- 239000005995 Aluminium silicate Substances 0.000 description 1
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 1
- 102400000068 Angiostatin Human genes 0.000 description 1
- 108010079709 Angiostatins Proteins 0.000 description 1
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Natural products OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 1
- 108010024976 Asparaginase Proteins 0.000 description 1
- 108010011485 Aspartame Proteins 0.000 description 1
- 101150033765 BAG1 gene Proteins 0.000 description 1
- 239000005711 Benzoic acid Substances 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-M Bicarbonate Chemical compound OC([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-M 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-M Bromide Chemical compound [Br-] CPELXLSAUQHCOX-UHFFFAOYSA-M 0.000 description 1
- FERIUCNNQQJTOY-UHFFFAOYSA-M Butyrate Chemical compound CCCC([O-])=O FERIUCNNQQJTOY-UHFFFAOYSA-M 0.000 description 1
- FERIUCNNQQJTOY-UHFFFAOYSA-N Butyric acid Natural products CCCC(O)=O FERIUCNNQQJTOY-UHFFFAOYSA-N 0.000 description 1
- JKJQYINUFPFPAJ-UHFFFAOYSA-N CC(=O)OCC1=C(C)C(Br)=C(C)C=N1.CC1=C(Br)C(C)=C(C)N(O)=C1.CC1=C(Br)C(C)=C(CBr)N=C1.CC1=C(Br)C(C)=C(CO)N=C1.CC1=CC(C)=C(C)N(O)=C1.CC1=CC(C)=C(C)N=C1 Chemical compound CC(=O)OCC1=C(C)C(Br)=C(C)C=N1.CC1=C(Br)C(C)=C(C)N(O)=C1.CC1=C(Br)C(C)=C(CBr)N=C1.CC1=C(Br)C(C)=C(CO)N=C1.CC1=CC(C)=C(C)N(O)=C1.CC1=CC(C)=C(C)N=C1 JKJQYINUFPFPAJ-UHFFFAOYSA-N 0.000 description 1
- DXWYWFJRGRMURH-UHFFFAOYSA-N CC(C)C(Br)CC=O.CC(C)C(C)CC=O.CC(C)C1=CNC2=C1C(=O)NC(N)=N2.CC(C)C1=CNC2=C1C(Cl)=NC(N)=N2.COC1=C(C)C(CCl)=NC=C1C.COC1=C(C)C=NC(CN2C=C(C(C)C)C3=C2N=C(N)N=C3Cl)=C1C.ClCCl.NC1=NC(N)=NC(O)=C1 Chemical compound CC(C)C(Br)CC=O.CC(C)C(C)CC=O.CC(C)C1=CNC2=C1C(=O)NC(N)=N2.CC(C)C1=CNC2=C1C(Cl)=NC(N)=N2.COC1=C(C)C(CCl)=NC=C1C.COC1=C(C)C=NC(CN2C=C(C(C)C)C3=C2N=C(N)N=C3Cl)=C1C.ClCCl.NC1=NC(N)=NC(O)=C1 DXWYWFJRGRMURH-UHFFFAOYSA-N 0.000 description 1
- RUWRIQQLOUHDLX-UHFFFAOYSA-N CC(C)C1=CNC2=C1C(Cl)=NC(N)=N2.CC1=C(Cl)C(C)=C(CN2C=C(C(C)C)C3=C2N=C(N)N=C3Cl)N=C1.CC1=CN=C(CCl)C(C)=C1Cl Chemical compound CC(C)C1=CNC2=C1C(Cl)=NC(N)=N2.CC1=C(Cl)C(C)=C(CN2C=C(C(C)C)C3=C2N=C(N)N=C3Cl)N=C1.CC1=CN=C(CCl)C(C)=C1Cl RUWRIQQLOUHDLX-UHFFFAOYSA-N 0.000 description 1
- VAJKTJNWEQDIOA-UHFFFAOYSA-N CC(C)C1=CNC2=C1C(Cl)=NC(N)=N2.CC1=C(Cl)C(C)=C(CN2C=C(C(C)C)C3=C2N=C(N)N=C3Cl)[N+]([O-])=C1.CC1=C[N+]([O-])=C(CCl)C(C)=C1Cl Chemical compound CC(C)C1=CNC2=C1C(Cl)=NC(N)=N2.CC1=C(Cl)C(C)=C(CN2C=C(C(C)C)C3=C2N=C(N)N=C3Cl)[N+]([O-])=C1.CC1=C[N+]([O-])=C(CCl)C(C)=C1Cl VAJKTJNWEQDIOA-UHFFFAOYSA-N 0.000 description 1
- QZRVQIARSNARRE-UHFFFAOYSA-N CC1=C(Br)C(C)=C(CCl)N=C1.CC1=C(O)C(C)=C(CCl)N=C1.COC1=C(C)C=NC(CCl)=C1C.Cl Chemical compound CC1=C(Br)C(C)=C(CCl)N=C1.CC1=C(O)C(C)=C(CCl)N=C1.COC1=C(C)C=NC(CCl)=C1C.Cl QZRVQIARSNARRE-UHFFFAOYSA-N 0.000 description 1
- YCXJCSHCLTYOET-UHFFFAOYSA-N CC1=C(Br)C(C)=C(CCl)N=C1.COC1=C(C)C=NC(CCl)=C1C.Cl Chemical compound CC1=C(Br)C(C)=C(CCl)N=C1.COC1=C(C)C=NC(CCl)=C1C.Cl YCXJCSHCLTYOET-UHFFFAOYSA-N 0.000 description 1
- CQTBNXHMRRCSQH-UHFFFAOYSA-N CC1=C(Br)C(C)=C(CN2C=C(I)C3=C(Cl)N=C(NC(=O)C(C)(C)C)N=C32)N(O)=C1 Chemical compound CC1=C(Br)C(C)=C(CN2C=C(I)C3=C(Cl)N=C(NC(=O)C(C)(C)C)N=C32)N(O)=C1 CQTBNXHMRRCSQH-UHFFFAOYSA-N 0.000 description 1
- RDBQTVIOIQJPGK-UHFFFAOYSA-N CC1=C(Br)C(C)=C(CN2C=CC3=C(Br)N=C(N)N=C32)N(O)=C1 Chemical compound CC1=C(Br)C(C)=C(CN2C=CC3=C(Br)N=C(N)N=C32)N(O)=C1 RDBQTVIOIQJPGK-UHFFFAOYSA-N 0.000 description 1
- QBFLCTMDUAWEHS-UHFFFAOYSA-N CC1=C(Br)C(C)=C(CN2C=CC3=C(Br)N=C(N)N=C32)[N+]([O-])=C1 Chemical compound CC1=C(Br)C(C)=C(CN2C=CC3=C(Br)N=C(N)N=C32)[N+]([O-])=C1 QBFLCTMDUAWEHS-UHFFFAOYSA-N 0.000 description 1
- ZNEVKLCNJGGTBM-UHFFFAOYSA-N CC1=C(Br)C(C)=C(CN2C=CC3=C(Cl)N=C(N)N=C32)N(O)=C1 Chemical compound CC1=C(Br)C(C)=C(CN2C=CC3=C(Cl)N=C(N)N=C32)N(O)=C1 ZNEVKLCNJGGTBM-UHFFFAOYSA-N 0.000 description 1
- STTBUEYZFNYDKP-UHFFFAOYSA-N CC1=C(Br)C(C)=C(CN2C=CC3=C(Cl)N=C(N)N=C32)[N+]([O-])=C1 Chemical compound CC1=C(Br)C(C)=C(CN2C=CC3=C(Cl)N=C(N)N=C32)[N+]([O-])=C1 STTBUEYZFNYDKP-UHFFFAOYSA-N 0.000 description 1
- XRXQJHNZUBQDSS-UHFFFAOYSA-N CC1=C(Br)C(C)=C(CN2C=CC3=C(Cl)N=C(NC(=O)C(C)(C)C)N=C32)N(O)=C1 Chemical compound CC1=C(Br)C(C)=C(CN2C=CC3=C(Cl)N=C(NC(=O)C(C)(C)C)N=C32)N(O)=C1 XRXQJHNZUBQDSS-UHFFFAOYSA-N 0.000 description 1
- RYWGELXBAUNCNA-UHFFFAOYSA-N CC1=C(Cl)C(C)=C(CCl)N=C1.CC1=C(O)C(C)=C(CCl)N=C1 Chemical compound CC1=C(Cl)C(C)=C(CCl)N=C1.CC1=C(O)C(C)=C(CCl)N=C1 RYWGELXBAUNCNA-UHFFFAOYSA-N 0.000 description 1
- BEODOKLZFCXBCQ-UHFFFAOYSA-N CC1=C(Cl)C(C)=C(CN2C=C(C(C)C)C3=C(Cl)N=C(N)N=C32)[N+]([O-])=C1 Chemical compound CC1=C(Cl)C(C)=C(CN2C=C(C(C)C)C3=C(Cl)N=C(N)N=C32)[N+]([O-])=C1 BEODOKLZFCXBCQ-UHFFFAOYSA-N 0.000 description 1
- HZZZMESVNNZQBB-UHFFFAOYSA-N CC1=C(Cl)C(C)=C(CN2C=C(I)C3=C(Cl)N=C(NC(=O)C(C)(C)C)N=C32)N(O)=C1 Chemical compound CC1=C(Cl)C(C)=C(CN2C=C(I)C3=C(Cl)N=C(NC(=O)C(C)(C)C)N=C32)N(O)=C1 HZZZMESVNNZQBB-UHFFFAOYSA-N 0.000 description 1
- VCXGUCMJKIBPOS-UHFFFAOYSA-N CC1=C(Cl)C(C)=C(CN2C=CC3=C(Br)N=C(N)N=C32)N(O)=C1 Chemical compound CC1=C(Cl)C(C)=C(CN2C=CC3=C(Br)N=C(N)N=C32)N(O)=C1 VCXGUCMJKIBPOS-UHFFFAOYSA-N 0.000 description 1
- ZYKXLRKWHRHZCK-UHFFFAOYSA-N CC1=C(Cl)C(C)=C(CN2C=CC3=C(Br)N=C(N)N=C32)[N+]([O-])=C1 Chemical compound CC1=C(Cl)C(C)=C(CN2C=CC3=C(Br)N=C(N)N=C32)[N+]([O-])=C1 ZYKXLRKWHRHZCK-UHFFFAOYSA-N 0.000 description 1
- TWIBXWIGBNKQPJ-UHFFFAOYSA-N CC1=C(Cl)C(C)=C(CN2C=CC3=C(Cl)/N=C(N)/N=C\32)[N+]([O-])=C1 Chemical compound CC1=C(Cl)C(C)=C(CN2C=CC3=C(Cl)/N=C(N)/N=C\32)[N+]([O-])=C1 TWIBXWIGBNKQPJ-UHFFFAOYSA-N 0.000 description 1
- KSIFSADZZREBMQ-UHFFFAOYSA-N CC1=C(Cl)C(C)=C(CN2C=CC3=C(Cl)N=C(N)N=C32)N(O)=C1 Chemical compound CC1=C(Cl)C(C)=C(CN2C=CC3=C(Cl)N=C(N)N=C32)N(O)=C1 KSIFSADZZREBMQ-UHFFFAOYSA-N 0.000 description 1
- SYHKCYUXSHCIPL-UHFFFAOYSA-N CC1=C(Cl)C(C)=C(CN2C=CC3=C(Cl)N=C(NC(=O)C(C)(C)C)N=C32)N(O)=C1 Chemical compound CC1=C(Cl)C(C)=C(CN2C=CC3=C(Cl)N=C(NC(=O)C(C)(C)C)N=C32)N(O)=C1 SYHKCYUXSHCIPL-UHFFFAOYSA-N 0.000 description 1
- NKEZVMXYCTWQRS-UHFFFAOYSA-N CC1=C(Cl)C(C)=C(CN2C=CC3=C(Cl)N=C(NC(=O)C(C)(C)C)N=C32)N=C1 Chemical compound CC1=C(Cl)C(C)=C(CN2C=CC3=C(Cl)N=C(NC(=O)C(C)(C)C)N=C32)N=C1 NKEZVMXYCTWQRS-UHFFFAOYSA-N 0.000 description 1
- LUDZCVWUAYKJNS-UHFFFAOYSA-N CC1=CN(O)=C(CN2C(=O)CC3=C(Cl)N=C(N)N=C32)C(C)=C1Cl Chemical compound CC1=CN(O)=C(CN2C(=O)CC3=C(Cl)N=C(N)N=C32)C(C)=C1Cl LUDZCVWUAYKJNS-UHFFFAOYSA-N 0.000 description 1
- NUJGXTPIMDUCQC-UHFFFAOYSA-N CCCC(Br)C=O.CCCC1=CN(C(C)=O)C2=C1C(=O)NC(NC(C)=O)=N2.CCCC1=CNC2=C1C(=O)NC(N)=N2.CCCC1=CNC2=C1C(Cl)=NC(NC(C)=O)=N2.COC1=C(C)C=NC(CN2C=C(CCO)C3=C2N=C(N)N=C3Cl)=C1C.ClC[Ar].NC1=CC(=O)NC(N)=N1.OCCCCO Chemical compound CCCC(Br)C=O.CCCC1=CN(C(C)=O)C2=C1C(=O)NC(NC(C)=O)=N2.CCCC1=CNC2=C1C(=O)NC(N)=N2.CCCC1=CNC2=C1C(Cl)=NC(NC(C)=O)=N2.COC1=C(C)C=NC(CN2C=C(CCO)C3=C2N=C(N)N=C3Cl)=C1C.ClC[Ar].NC1=CC(=O)NC(N)=N1.OCCCCO NUJGXTPIMDUCQC-UHFFFAOYSA-N 0.000 description 1
- VSEMECRRGOYMBT-UHFFFAOYSA-N CCCCCCCC(=O)NC1=NC2=C(C(=O)N1)C(CN(C)C1=CC=CC=C1)=CN2.CCCCCCCC(=O)NC1=NC2=C(C(=O)N1)C(CN(CC1=CC=CC=C1)CC1=CC=CC=C1)=CN2.CNC1=CC=CC=C1.COC1=C(C)C(CCl)=NC=C1C.COC1=C(C)C=NC(CN2C=C(CN(C)C3=CC=CC=C3)C3=C2N=C(N)N=C3Cl)=C1C Chemical compound CCCCCCCC(=O)NC1=NC2=C(C(=O)N1)C(CN(C)C1=CC=CC=C1)=CN2.CCCCCCCC(=O)NC1=NC2=C(C(=O)N1)C(CN(CC1=CC=CC=C1)CC1=CC=CC=C1)=CN2.CNC1=CC=CC=C1.COC1=C(C)C(CCl)=NC=C1C.COC1=C(C)C=NC(CN2C=C(CN(C)C3=CC=CC=C3)C3=C2N=C(N)N=C3Cl)=C1C VSEMECRRGOYMBT-UHFFFAOYSA-N 0.000 description 1
- PHYNCAOYPPILPM-UHFFFAOYSA-N CCCCCCCC(=O)NC1=NC2=C(C(=O)N1)C(CN(CC1=CC=CC=C1)CC1=CC=CC=C1)=CN2.CCCCCCCC(=O)NC1=NC2=C(C(=O)N1)C(CNC1=CC=CC=C1)=CN2.COC1=C(C)C(CCl)=NC=C1C.COC1=C(C)C=NC(CN2C=C(CNC3=CC=CC=C3)C3=C2N=C(N)N=C3Cl)=C1C.NC1=CC=CC=C1 Chemical compound CCCCCCCC(=O)NC1=NC2=C(C(=O)N1)C(CN(CC1=CC=CC=C1)CC1=CC=CC=C1)=CN2.CCCCCCCC(=O)NC1=NC2=C(C(=O)N1)C(CNC1=CC=CC=C1)=CN2.COC1=C(C)C(CCl)=NC=C1C.COC1=C(C)C=NC(CN2C=C(CNC3=CC=CC=C3)C3=C2N=C(N)N=C3Cl)=C1C.NC1=CC=CC=C1 PHYNCAOYPPILPM-UHFFFAOYSA-N 0.000 description 1
- AHZOXLZFNJQTEJ-UHFFFAOYSA-N CCCCCCCC(=O)NC1=NC2=C(C(=O)N1)C(CN(CC1=CC=CC=C1)CC1=CC=CC=C1)=CN2.CCCCCCCC(=O)NC1=NC2=C(C(CN(CC3=CC=CC=C3)CC3=CC=CC=C3)=CN2)C(Cl)=N1.COC1=C(C)C(CCl)=NC=C1C.COC1=C(C)C=NC(CN2C=C(CN(CC3=CC=CC=C3)CC3=CC=CC=C3)C3=C2N=C(N)N=C3Cl)=C1C Chemical compound CCCCCCCC(=O)NC1=NC2=C(C(=O)N1)C(CN(CC1=CC=CC=C1)CC1=CC=CC=C1)=CN2.CCCCCCCC(=O)NC1=NC2=C(C(CN(CC3=CC=CC=C3)CC3=CC=CC=C3)=CN2)C(Cl)=N1.COC1=C(C)C(CCl)=NC=C1C.COC1=C(C)C=NC(CN2C=C(CN(CC3=CC=CC=C3)CC3=CC=CC=C3)C3=C2N=C(N)N=C3Cl)=C1C AHZOXLZFNJQTEJ-UHFFFAOYSA-N 0.000 description 1
- IPMWTZIETCDUFF-UHFFFAOYSA-N CCCCCCCC(=O)NC1=NC2=C(C=C(CN3CCCC3)N2)C(=O)N1.COC1=C(C)C(CCl)=NC=C1C.COC1=C(C)C=NC(CN2C(CN3CCCC3)=CC3=C2N=C(N)N=C3Cl)=C1C Chemical compound CCCCCCCC(=O)NC1=NC2=C(C=C(CN3CCCC3)N2)C(=O)N1.COC1=C(C)C(CCl)=NC=C1C.COC1=C(C)C=NC(CN2C(CN3CCCC3)=CC3=C2N=C(N)N=C3Cl)=C1C IPMWTZIETCDUFF-UHFFFAOYSA-N 0.000 description 1
- PHYBTJHGXXIDRQ-UHFFFAOYSA-N COC1=C(C)C(CN2C(=O)CC3=C(Cl)N=C(N)N=C32)=[N+](O)C=C1C Chemical compound COC1=C(C)C(CN2C(=O)CC3=C(Cl)N=C(N)N=C32)=[N+](O)C=C1C PHYBTJHGXXIDRQ-UHFFFAOYSA-N 0.000 description 1
- LLADMNDTEVYVBB-UHFFFAOYSA-N COC1=C(C)C(Cl)=NC(CCl)=C1C.COC1=C(C)C=N(O)C(CCl)=C1C.COC1=C(C)C=NC(CCl)=C1C.O=P(Cl)(Cl)Cl Chemical compound COC1=C(C)C(Cl)=NC(CCl)=C1C.COC1=C(C)C=N(O)C(CCl)=C1C.COC1=C(C)C=NC(CCl)=C1C.O=P(Cl)(Cl)Cl LLADMNDTEVYVBB-UHFFFAOYSA-N 0.000 description 1
- IVFQOFMDBPWTQG-UHFFFAOYSA-N COC1=C(C)C=N(O)C(CN2C=C(I)C3=C(Cl)N=C(NC(=O)C(C)(C)C)N=C32)=C1C Chemical compound COC1=C(C)C=N(O)C(CN2C=C(I)C3=C(Cl)N=C(NC(=O)C(C)(C)C)N=C32)=C1C IVFQOFMDBPWTQG-UHFFFAOYSA-N 0.000 description 1
- MENZQXMVFABHEC-UHFFFAOYSA-N COC1=C(C)C=N(O)C(CN2C=CC3=C(Br)N=C(N)N=C32)=C1C Chemical compound COC1=C(C)C=N(O)C(CN2C=CC3=C(Br)N=C(N)N=C32)=C1C MENZQXMVFABHEC-UHFFFAOYSA-N 0.000 description 1
- YOUZNLGQNGHHQV-UHFFFAOYSA-N COC1=C(C)C=N(O)C(CN2C=CC3=C(Cl)N=C(N)N=C32)=C1C Chemical compound COC1=C(C)C=N(O)C(CN2C=CC3=C(Cl)N=C(N)N=C32)=C1C YOUZNLGQNGHHQV-UHFFFAOYSA-N 0.000 description 1
- LETCZQHHKHSYCG-UHFFFAOYSA-N COC1=C(C)C=N(O)C(CN2C=CC3=C(Cl)N=C(NC(=O)C(C)(C)C)N=C32)=C1C Chemical compound COC1=C(C)C=N(O)C(CN2C=CC3=C(Cl)N=C(NC(=O)C(C)(C)C)N=C32)=C1C LETCZQHHKHSYCG-UHFFFAOYSA-N 0.000 description 1
- MJGFAULXIIORAM-UHFFFAOYSA-N COC1=C(C)C=NC(CN2C3=NC(N)=NC(Cl)=C3C(C)=C2O)=C1C Chemical compound COC1=C(C)C=NC(CN2C3=NC(N)=NC(Cl)=C3C(C)=C2O)=C1C MJGFAULXIIORAM-UHFFFAOYSA-N 0.000 description 1
- ZBPVFBGYKPAYQH-UHFFFAOYSA-N COC1=C(C)C=[N+]([O-])C(CN2C(=O)CC3=C(Cl)N=C(N)N=C32)=C1C Chemical compound COC1=C(C)C=[N+]([O-])C(CN2C(=O)CC3=C(Cl)N=C(N)N=C32)=C1C ZBPVFBGYKPAYQH-UHFFFAOYSA-N 0.000 description 1
- NOHSBEAPVWNLHY-UHFFFAOYSA-N COC1=C(C)C=[N+]([O-])C(CN2C=CC3=C(Br)N=C(N)N=C32)=C1C Chemical compound COC1=C(C)C=[N+]([O-])C(CN2C=CC3=C(Br)N=C(N)N=C32)=C1C NOHSBEAPVWNLHY-UHFFFAOYSA-N 0.000 description 1
- VCVHHLOHUGPNKS-UHFFFAOYSA-N COC1=C(C)C=[N+]([O-])C(CN2C=CC3=C(Cl)N=C(N)N=C32)=C1C Chemical compound COC1=C(C)C=[N+]([O-])C(CN2C=CC3=C(Cl)N=C(N)N=C32)=C1C VCVHHLOHUGPNKS-UHFFFAOYSA-N 0.000 description 1
- GJWXMOBMWVUVBS-UHFFFAOYSA-N COC1=C(OC)C(OC)=C(C)C(CN2C=CC3=C(Cl)N=C(N)N=C32)=C1 Chemical compound COC1=C(OC)C(OC)=C(C)C(CN2C=CC3=C(Cl)N=C(N)N=C32)=C1 GJWXMOBMWVUVBS-UHFFFAOYSA-N 0.000 description 1
- FFFQHIFSJBSLRX-UHFFFAOYSA-N COC1=C(OC)C(OC)=C(Cl)C(CCl)=C1.COC1=CC(CCl)=CC(OC)=C1OC Chemical compound COC1=C(OC)C(OC)=C(Cl)C(CCl)=C1.COC1=CC(CCl)=CC(OC)=C1OC FFFQHIFSJBSLRX-UHFFFAOYSA-N 0.000 description 1
- YTYHSFMZYIZYOF-UHFFFAOYSA-N COC1=CC(CN2C(=O)C(C)(C)C3=C(Cl)N=C(N)N=C32)=C(Br)C(OC)=C1OC Chemical compound COC1=CC(CN2C(=O)C(C)(C)C3=C(Cl)N=C(N)N=C32)=C(Br)C(OC)=C1OC YTYHSFMZYIZYOF-UHFFFAOYSA-N 0.000 description 1
- LPMACFYMWYJPNN-UHFFFAOYSA-N COC1=CC(OC)=C([N+](=O)[O-])C(CN2C=CC3=C(Cl)N=C(N)N=C32)=C1 Chemical compound COC1=CC(OC)=C([N+](=O)[O-])C(CN2C=CC3=C(Cl)N=C(N)N=C32)=C1 LPMACFYMWYJPNN-UHFFFAOYSA-N 0.000 description 1
- JZMSKNRSJFMYRS-UHFFFAOYSA-N COC1=CC=C(OC)C(CN2C=CC3=C(Cl)N=C(N)N=C32)=C1 Chemical compound COC1=CC=C(OC)C(CN2C=CC3=C(Cl)N=C(N)N=C32)=C1 JZMSKNRSJFMYRS-UHFFFAOYSA-N 0.000 description 1
- 101100311260 Caenorhabditis elegans sti-1 gene Proteins 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- KLWPJMFMVPTNCC-UHFFFAOYSA-N Camptothecin Natural products CCC1(O)C(=O)OCC2=C1C=C3C4Nc5ccccc5C=C4CN3C2=O KLWPJMFMVPTNCC-UHFFFAOYSA-N 0.000 description 1
- KXDHJXZQYSOELW-UHFFFAOYSA-M Carbamate Chemical compound NC([O-])=O KXDHJXZQYSOELW-UHFFFAOYSA-M 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 1
- 102000053642 Catalytic RNA Human genes 0.000 description 1
- 108090000994 Catalytic RNA Proteins 0.000 description 1
- 102100025064 Cellular tumor antigen p53 Human genes 0.000 description 1
- 101710163595 Chaperone protein DnaK Proteins 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 1
- 102100031162 Collagen alpha-1(XVIII) chain Human genes 0.000 description 1
- CMSMOCZEIVJLDB-UHFFFAOYSA-N Cyclophosphamide Chemical compound ClCCN(CCCl)P1(=O)NCCCO1 CMSMOCZEIVJLDB-UHFFFAOYSA-N 0.000 description 1
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 1
- OLAFFPNXVJANFR-UHFFFAOYSA-N DG Chemical compound N1C(N)=NC(=O)C2=C1NC=C2 OLAFFPNXVJANFR-UHFFFAOYSA-N 0.000 description 1
- 108010054814 DNA Gyrase Proteins 0.000 description 1
- 108020001019 DNA Primers Proteins 0.000 description 1
- 239000003155 DNA primer Substances 0.000 description 1
- 101710088194 Dehydrogenase Proteins 0.000 description 1
- BWGNESOTFCXPMA-UHFFFAOYSA-N Dihydrogen disulfide Chemical compound SS BWGNESOTFCXPMA-UHFFFAOYSA-N 0.000 description 1
- XTHFKEDIFFGKHM-UHFFFAOYSA-N Dimethoxyethane Chemical compound COCCOC XTHFKEDIFFGKHM-UHFFFAOYSA-N 0.000 description 1
- AADVCYNFEREWOS-OBRABYBLSA-N Discodermolide Chemical compound C=C\C=C/[C@H](C)[C@H](OC(N)=O)[C@@H](C)[C@H](O)[C@@H](C)C\C(C)=C/[C@H](C)[C@@H](O)[C@@H](C)\C=C/[C@@H](O)C[C@@H]1OC(=O)[C@H](C)[C@@H](O)[C@H]1C AADVCYNFEREWOS-OBRABYBLSA-N 0.000 description 1
- 102100020977 DnaJ homolog subfamily A member 1 Human genes 0.000 description 1
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 1
- 101150029707 ERBB2 gene Proteins 0.000 description 1
- LVGKNOAMLMIIKO-UHFFFAOYSA-N Elaidinsaeure-aethylester Natural products CCCCCCCCC=CCCCCCCCC(=O)OCC LVGKNOAMLMIIKO-UHFFFAOYSA-N 0.000 description 1
- 108010079505 Endostatins Proteins 0.000 description 1
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical compound NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 description 1
- GHASVSINZRGABV-UHFFFAOYSA-N Fluorouracil Chemical compound FC1=CNC(=O)NC1=O GHASVSINZRGABV-UHFFFAOYSA-N 0.000 description 1
- BDAGIHXWWSANSR-UHFFFAOYSA-M Formate Chemical compound [O-]C=O BDAGIHXWWSANSR-UHFFFAOYSA-M 0.000 description 1
- 238000005863 Friedel-Crafts acylation reaction Methods 0.000 description 1
- 238000003547 Friedel-Crafts alkylation reaction Methods 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- AEMRFAOFKBGASW-UHFFFAOYSA-M Glycolate Chemical compound OCC([O-])=O AEMRFAOFKBGASW-UHFFFAOYSA-M 0.000 description 1
- 102000009465 Growth Factor Receptors Human genes 0.000 description 1
- 108010009202 Growth Factor Receptors Proteins 0.000 description 1
- 229920000084 Gum arabic Polymers 0.000 description 1
- 101150054472 HER2 gene Proteins 0.000 description 1
- 229910004373 HOAc Inorganic materials 0.000 description 1
- 108010042283 HSP40 Heat-Shock Proteins Proteins 0.000 description 1
- 102000004447 HSP40 Heat-Shock Proteins Human genes 0.000 description 1
- 101710178376 Heat shock 70 kDa protein Proteins 0.000 description 1
- 101710152018 Heat shock cognate 70 kDa protein Proteins 0.000 description 1
- 101000931227 Homo sapiens DnaJ homolog subfamily A member 1 Proteins 0.000 description 1
- 229920002153 Hydroxypropyl cellulose Polymers 0.000 description 1
- 102000000521 Immunophilins Human genes 0.000 description 1
- 108010016648 Immunophilins Proteins 0.000 description 1
- 102000014150 Interferons Human genes 0.000 description 1
- 108010050904 Interferons Proteins 0.000 description 1
- 102000015696 Interleukins Human genes 0.000 description 1
- 108010063738 Interleukins Proteins 0.000 description 1
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 1
- FBOZXECLQNJBKD-ZDUSSCGKSA-N L-methotrexate Chemical compound C=1N=C2N=C(N)N=C(N)C2=NC=1CN(C)C1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 FBOZXECLQNJBKD-ZDUSSCGKSA-N 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-L L-tartrate(2-) Chemical compound [O-]C(=O)[C@H](O)[C@@H](O)C([O-])=O FEWJPZIEWOKRBE-JCYAYHJZSA-L 0.000 description 1
- JVTAAEKCZFNVCJ-UHFFFAOYSA-M Lactate Chemical compound CC(O)C([O-])=O JVTAAEKCZFNVCJ-UHFFFAOYSA-M 0.000 description 1
- 240000007472 Leucaena leucocephala Species 0.000 description 1
- 235000010643 Leucaena leucocephala Nutrition 0.000 description 1
- 108010000817 Leuprolide Proteins 0.000 description 1
- JXLYSJRDGCGARV-PJXZDTQASA-N Leurosidine Natural products O=C(O[C@H]1[C@](O)(C(=O)OC)[C@@H]2N(C)c3c(cc(c(OC)c3)[C@]3(C(=O)OC)c4[nH]c5c(c4CCN4C[C@@](O)(CC)C[C@H](C3)C4)cccc5)[C@@]32[C@H]2[C@@]1(CC)C=CCN2CC3)C JXLYSJRDGCGARV-PJXZDTQASA-N 0.000 description 1
- LPGWZGMPDKDHEP-HLTPFJCJSA-N Leurosine Chemical compound C([C@]1([C@@H]2O1)CC)N(CCC=1C3=CC=CC=C3NC=11)C[C@H]2C[C@]1(C(=O)OC)C1=CC([C@]23[C@H]([C@@]([C@H](OC(C)=O)[C@]4(CC)C=CCN([C@H]34)CC2)(O)C(=O)OC)N2C)=C2C=C1OC LPGWZGMPDKDHEP-HLTPFJCJSA-N 0.000 description 1
- LPGWZGMPDKDHEP-GKWAKPNHSA-N Leurosine Natural products O=C(O[C@H]1[C@](O)(C(=O)OC)[C@@H]2N(C)c3c(cc(c(OC)c3)[C@]3(C(=O)OC)c4[nH]c5c(c4CCN4C[C@]6(CC)O[C@@H]6[C@H](C3)C4)cccc5)[C@@]32[C@H]2[C@@]1(CC)C=CCN2CC3)C LPGWZGMPDKDHEP-GKWAKPNHSA-N 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-L Malonate Chemical compound [O-]C(=O)CC([O-])=O OFOBLEOULBTSOW-UHFFFAOYSA-L 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- 238000006683 Mannich reaction Methods 0.000 description 1
- 229930195725 Mannitol Natural products 0.000 description 1
- 239000012359 Methanesulfonyl chloride Substances 0.000 description 1
- 229920000168 Microcrystalline cellulose Polymers 0.000 description 1
- 229930192392 Mitomycin Natural products 0.000 description 1
- HRHKSTOGXBBQCB-UHFFFAOYSA-N Mitomycin E Natural products O=C1C(N)=C(C)C(=O)C2=C1C(COC(N)=O)C1(OC)C3N(C)C3CN12 HRHKSTOGXBBQCB-UHFFFAOYSA-N 0.000 description 1
- MZRVEZGGRBJDDB-UHFFFAOYSA-N N-Butyllithium Chemical compound [Li]CCCC MZRVEZGGRBJDDB-UHFFFAOYSA-N 0.000 description 1
- 229910017849 NH2—NH2 Inorganic materials 0.000 description 1
- 229910002651 NO3 Inorganic materials 0.000 description 1
- PVNIIMVLHYAWGP-UHFFFAOYSA-N Niacin Chemical compound OC(=O)C1=CC=CN=C1 PVNIIMVLHYAWGP-UHFFFAOYSA-N 0.000 description 1
- NHNBFGGVMKEFGY-UHFFFAOYSA-N Nitrate Chemical compound [O-][N+]([O-])=O NHNBFGGVMKEFGY-UHFFFAOYSA-N 0.000 description 1
- 238000000636 Northern blotting Methods 0.000 description 1
- 239000005642 Oleic acid Substances 0.000 description 1
- ZQPPMHVWECSIRJ-UHFFFAOYSA-N Oleic acid Natural products CCCCCCCCC=CCCCCCCCC(O)=O ZQPPMHVWECSIRJ-UHFFFAOYSA-N 0.000 description 1
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 1
- 229910020667 PBr3 Inorganic materials 0.000 description 1
- 229930012538 Paclitaxel Natural products 0.000 description 1
- 241000237988 Patellidae Species 0.000 description 1
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 1
- 102000004245 Proteasome Endopeptidase Complex Human genes 0.000 description 1
- 108090000708 Proteasome Endopeptidase Complex Proteins 0.000 description 1
- 102000001253 Protein Kinase Human genes 0.000 description 1
- 102000004022 Protein-Tyrosine Kinases Human genes 0.000 description 1
- 108090000412 Protein-Tyrosine Kinases Proteins 0.000 description 1
- 101710141955 RAF proto-oncogene serine/threonine-protein kinase Proteins 0.000 description 1
- 101150101372 RAF1 gene Proteins 0.000 description 1
- 229910006124 SOCl2 Inorganic materials 0.000 description 1
- 235000019485 Safflower oil Nutrition 0.000 description 1
- 239000002262 Schiff base Substances 0.000 description 1
- 150000004753 Schiff bases Chemical class 0.000 description 1
- BUGBHKTXTAQXES-UHFFFAOYSA-N Selenium Chemical compound [Se] BUGBHKTXTAQXES-UHFFFAOYSA-N 0.000 description 1
- 238000002105 Southern blotting Methods 0.000 description 1
- 235000021355 Stearic acid Nutrition 0.000 description 1
- 108010085012 Steroid Receptors Proteins 0.000 description 1
- 108010090804 Streptavidin Proteins 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- 238000006069 Suzuki reaction reaction Methods 0.000 description 1
- ZMZDMBWJUHKJPS-UHFFFAOYSA-M Thiocyanate anion Chemical compound [S-]C#N ZMZDMBWJUHKJPS-UHFFFAOYSA-M 0.000 description 1
- FOCVUCIESVLUNU-UHFFFAOYSA-N Thiotepa Chemical compound C1CN1P(N1CC1)(=S)N1CC1 FOCVUCIESVLUNU-UHFFFAOYSA-N 0.000 description 1
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 1
- 102000004142 Trypsin Human genes 0.000 description 1
- 108090000631 Trypsin Proteins 0.000 description 1
- 102000044159 Ubiquitin Human genes 0.000 description 1
- 108090000848 Ubiquitin Proteins 0.000 description 1
- 241000863480 Vinca Species 0.000 description 1
- WERKSKAQRVDLDW-ANOHMWSOSA-N [(2s,3r,4r,5r)-2,3,4,5,6-pentahydroxyhexyl] (z)-octadec-9-enoate Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO WERKSKAQRVDLDW-ANOHMWSOSA-N 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- 238000002835 absorbance Methods 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- JXLYSJRDGCGARV-KSNABSRWSA-N ac1l29ym Chemical compound C([C@H](C[C@]1(C(=O)OC)C=2C(=CC3=C([C@]45[C@H]([C@@]([C@H](OC(C)=O)[C@]6(CC)C=CCN([C@H]56)CC4)(O)C(=O)OC)N3C)C=2)OC)C[C@](C2)(O)CC)N2CCC2=C1NC1=CC=CC=C21 JXLYSJRDGCGARV-KSNABSRWSA-N 0.000 description 1
- 235000010489 acacia gum Nutrition 0.000 description 1
- 239000008351 acetate buffer Substances 0.000 description 1
- XPOLVIIHTDKJRY-UHFFFAOYSA-N acetic acid;methanimidamide Chemical compound NC=N.CC(O)=O XPOLVIIHTDKJRY-UHFFFAOYSA-N 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 230000004913 activation Effects 0.000 description 1
- 239000011149 active material Substances 0.000 description 1
- 230000010933 acylation Effects 0.000 description 1
- 125000005041 acyloxyalkyl group Chemical group 0.000 description 1
- WNLRTRBMVRJNCN-UHFFFAOYSA-L adipate(2-) Chemical compound [O-]C(=O)CCCCC([O-])=O WNLRTRBMVRJNCN-UHFFFAOYSA-L 0.000 description 1
- 238000012382 advanced drug delivery Methods 0.000 description 1
- 239000008272 agar Substances 0.000 description 1
- 229940072056 alginate Drugs 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 150000001336 alkenes Chemical class 0.000 description 1
- 150000001345 alkine derivatives Chemical class 0.000 description 1
- 125000005205 alkoxycarbonyloxyalkyl group Chemical group 0.000 description 1
- 125000002877 alkyl aryl group Chemical group 0.000 description 1
- 125000005907 alkyl ester group Chemical group 0.000 description 1
- 150000001350 alkyl halides Chemical class 0.000 description 1
- 125000002947 alkylene group Chemical group 0.000 description 1
- 229940087168 alpha tocopherol Drugs 0.000 description 1
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 description 1
- AWUCVROLDVIAJX-UHFFFAOYSA-N alpha-glycerophosphate Natural products OCC(O)COP(O)(O)=O AWUCVROLDVIAJX-UHFFFAOYSA-N 0.000 description 1
- 229960000473 altretamine Drugs 0.000 description 1
- AZDRQVAHHNSJOQ-UHFFFAOYSA-N alumane Chemical class [AlH3] AZDRQVAHHNSJOQ-UHFFFAOYSA-N 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 235000012211 aluminium silicate Nutrition 0.000 description 1
- 150000001409 amidines Chemical class 0.000 description 1
- 229940024606 amino acid Drugs 0.000 description 1
- 235000001014 amino acid Nutrition 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- 238000005902 aminomethylation reaction Methods 0.000 description 1
- 229960003896 aminopterin Drugs 0.000 description 1
- 230000033115 angiogenesis Effects 0.000 description 1
- 229940045799 anthracyclines and related substance Drugs 0.000 description 1
- 125000005428 anthryl group Chemical group [H]C1=C([H])C([H])=C2C([H])=C3C(*)=C([H])C([H])=C([H])C3=C([H])C2=C1[H] 0.000 description 1
- 230000002924 anti-infective effect Effects 0.000 description 1
- 239000002260 anti-inflammatory agent Substances 0.000 description 1
- 230000001028 anti-proliverative effect Effects 0.000 description 1
- 230000000692 anti-sense effect Effects 0.000 description 1
- 229940111121 antirheumatic drug quinolines Drugs 0.000 description 1
- 230000006907 apoptotic process Effects 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 150000007860 aryl ester derivatives Chemical class 0.000 description 1
- 235000010323 ascorbic acid Nutrition 0.000 description 1
- 229960005070 ascorbic acid Drugs 0.000 description 1
- 239000011668 ascorbic acid Substances 0.000 description 1
- IAOZJIPTCAWIRG-QWRGUYRKSA-N aspartame Chemical compound OC(=O)C[C@H](N)C(=O)N[C@H](C(=O)OC)CC1=CC=CC=C1 IAOZJIPTCAWIRG-QWRGUYRKSA-N 0.000 description 1
- 239000000605 aspartame Substances 0.000 description 1
- 235000010357 aspartame Nutrition 0.000 description 1
- 229960003438 aspartame Drugs 0.000 description 1
- 229940009098 aspartate Drugs 0.000 description 1
- FZCSTZYAHCUGEM-UHFFFAOYSA-N aspergillomarasmine B Natural products OC(=O)CNC(C(O)=O)CNC(C(O)=O)CC(O)=O FZCSTZYAHCUGEM-UHFFFAOYSA-N 0.000 description 1
- 230000005784 autoimmunity Effects 0.000 description 1
- 210000003719 b-lymphocyte Anatomy 0.000 description 1
- HNYOPLTXPVRDBG-UHFFFAOYSA-N barbituric acid Chemical compound O=C1CC(=O)NC(=O)N1 HNYOPLTXPVRDBG-UHFFFAOYSA-N 0.000 description 1
- 235000013871 bee wax Nutrition 0.000 description 1
- 239000012166 beeswax Substances 0.000 description 1
- SRSXLGNVWSONIS-UHFFFAOYSA-M benzenesulfonate Chemical compound [O-]S(=O)(=O)C1=CC=CC=C1 SRSXLGNVWSONIS-UHFFFAOYSA-M 0.000 description 1
- 229940077388 benzenesulfonate Drugs 0.000 description 1
- 229940050390 benzoate Drugs 0.000 description 1
- 125000003310 benzodiazepinyl group Chemical group N1N=C(C=CC2=C1C=CC=C2)* 0.000 description 1
- 125000000499 benzofuranyl group Chemical group O1C(=CC2=C1C=CC=C2)* 0.000 description 1
- 235000010233 benzoic acid Nutrition 0.000 description 1
- QRUDEWIWKLJBPS-UHFFFAOYSA-N benzotriazole Chemical compound C1=CC=C2N[N][N]C2=C1 QRUDEWIWKLJBPS-UHFFFAOYSA-N 0.000 description 1
- 239000012964 benzotriazole Substances 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- XMIIGOLPHOKFCH-UHFFFAOYSA-N beta-phenylpropanoic acid Natural products OC(=O)CCC1=CC=CC=C1 XMIIGOLPHOKFCH-UHFFFAOYSA-N 0.000 description 1
- 229960000997 bicalutamide Drugs 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 229960001561 bleomycin Drugs 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 230000036765 blood level Effects 0.000 description 1
- 210000000133 brain stem Anatomy 0.000 description 1
- 239000006172 buffering agent Substances 0.000 description 1
- 244000309464 bull Species 0.000 description 1
- 125000004369 butenyl group Chemical group C(=CCC)* 0.000 description 1
- 239000006227 byproduct Substances 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 235000010216 calcium carbonate Nutrition 0.000 description 1
- MIOPJNTWMNEORI-UHFFFAOYSA-N camphorsulfonic acid Chemical compound C1CC2(CS(O)(=O)=O)C(=O)CC1C2(C)C MIOPJNTWMNEORI-UHFFFAOYSA-N 0.000 description 1
- VSJKWCGYPAHWDS-FQEVSTJZSA-N camptothecin Chemical compound C1=CC=C2C=C(CN3C4=CC5=C(C3=O)COC(=O)[C@]5(O)CC)C4=NC2=C1 VSJKWCGYPAHWDS-FQEVSTJZSA-N 0.000 description 1
- 229940127093 camptothecin Drugs 0.000 description 1
- RYTLGWCJESCDMY-UHFFFAOYSA-N carbamimidoyl chloride Chemical compound NC(Cl)=N RYTLGWCJESCDMY-UHFFFAOYSA-N 0.000 description 1
- 150000004649 carbonic acid derivatives Chemical class 0.000 description 1
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 1
- YAYRGNWWLMLWJE-UHFFFAOYSA-L carboplatin Chemical compound O=C1O[Pt](N)(N)OC(=O)C11CCC1 YAYRGNWWLMLWJE-UHFFFAOYSA-L 0.000 description 1
- 229960004562 carboplatin Drugs 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 230000021523 carboxylation Effects 0.000 description 1
- 238000006473 carboxylation reaction Methods 0.000 description 1
- XREUEWVEMYWFFA-CSKJXFQVSA-N carminomycin Chemical compound C1[C@H](N)[C@H](O)[C@H](C)O[C@H]1O[C@@H]1C2=C(O)C(C(=O)C3=C(O)C=CC=C3C3=O)=C3C(O)=C2C[C@@](O)(C(C)=O)C1 XREUEWVEMYWFFA-CSKJXFQVSA-N 0.000 description 1
- 229930188550 carminomycin Natural products 0.000 description 1
- XREUEWVEMYWFFA-UHFFFAOYSA-N carminomycin I Natural products C1C(N)C(O)C(C)OC1OC1C2=C(O)C(C(=O)C3=C(O)C=CC=C3C3=O)=C3C(O)=C2CC(O)(C(C)=O)C1 XREUEWVEMYWFFA-UHFFFAOYSA-N 0.000 description 1
- 229950001725 carubicin Drugs 0.000 description 1
- 230000003197 catalytic effect Effects 0.000 description 1
- 238000006555 catalytic reaction Methods 0.000 description 1
- 239000006143 cell culture medium Substances 0.000 description 1
- 230000022131 cell cycle Effects 0.000 description 1
- 230000024245 cell differentiation Effects 0.000 description 1
- 238000002701 cell growth assay Methods 0.000 description 1
- 230000011748 cell maturation Effects 0.000 description 1
- 230000004663 cell proliferation Effects 0.000 description 1
- 238000001516 cell proliferation assay Methods 0.000 description 1
- 230000001413 cellular effect Effects 0.000 description 1
- 229920002301 cellulose acetate Polymers 0.000 description 1
- 229920006217 cellulose acetate butyrate Polymers 0.000 description 1
- 210000003169 central nervous system Anatomy 0.000 description 1
- 229960000541 cetyl alcohol Drugs 0.000 description 1
- 238000010568 chiral column chromatography Methods 0.000 description 1
- 238000005660 chlorination reaction Methods 0.000 description 1
- 229940075419 choline hydroxide Drugs 0.000 description 1
- 125000004230 chromenyl group Chemical group O1C(C=CC2=CC=CC=C12)* 0.000 description 1
- 125000002676 chrysenyl group Chemical group C1(=CC=CC=2C3=CC=C4C=CC=CC4=C3C=CC12)* 0.000 description 1
- JAWGVVJVYSANRY-UHFFFAOYSA-N cobalt(3+) Chemical compound [Co+3] JAWGVVJVYSANRY-UHFFFAOYSA-N 0.000 description 1
- 239000003240 coconut oil Substances 0.000 description 1
- 235000019864 coconut oil Nutrition 0.000 description 1
- 239000012230 colorless oil Substances 0.000 description 1
- 238000002648 combination therapy Methods 0.000 description 1
- 230000009137 competitive binding Effects 0.000 description 1
- 230000002860 competitive effect Effects 0.000 description 1
- 239000002299 complementary DNA Substances 0.000 description 1
- 238000002591 computed tomography Methods 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 235000005687 corn oil Nutrition 0.000 description 1
- 239000002285 corn oil Substances 0.000 description 1
- 235000012343 cottonseed oil Nutrition 0.000 description 1
- 239000002385 cottonseed oil Substances 0.000 description 1
- 239000013058 crude material Substances 0.000 description 1
- 239000010779 crude oil Substances 0.000 description 1
- 239000012043 crude product Substances 0.000 description 1
- 150000004292 cyclic ethers Chemical class 0.000 description 1
- 125000000392 cycloalkenyl group Chemical group 0.000 description 1
- 125000000058 cyclopentadienyl group Chemical group C1(=CC=CC1)* 0.000 description 1
- 229960004397 cyclophosphamide Drugs 0.000 description 1
- 229960000684 cytarabine Drugs 0.000 description 1
- 231100000433 cytotoxic Toxicity 0.000 description 1
- 230000001472 cytotoxic effect Effects 0.000 description 1
- 231100000135 cytotoxicity Toxicity 0.000 description 1
- 230000003013 cytotoxicity Effects 0.000 description 1
- 229960003901 dacarbazine Drugs 0.000 description 1
- STQGQHZAVUOBTE-VGBVRHCVSA-N daunorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(C)=O)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 STQGQHZAVUOBTE-VGBVRHCVSA-N 0.000 description 1
- 229960000975 daunorubicin Drugs 0.000 description 1
- 125000004855 decalinyl group Chemical group C1(CCCC2CCCCC12)* 0.000 description 1
- 238000000326 densiometry Methods 0.000 description 1
- 238000001212 derivatisation Methods 0.000 description 1
- 239000007933 dermal patch Substances 0.000 description 1
- 230000001687 destabilization Effects 0.000 description 1
- 230000000368 destabilizing effect Effects 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- 238000003745 diagnosis Methods 0.000 description 1
- LUHUASWMJCSDTG-UHFFFAOYSA-N dicyclohexylboron Chemical compound C1CCCCC1[B]C1CCCCC1 LUHUASWMJCSDTG-UHFFFAOYSA-N 0.000 description 1
- ZBCBWPMODOFKDW-UHFFFAOYSA-N diethanolamine Chemical compound OCCNCCO ZBCBWPMODOFKDW-UHFFFAOYSA-N 0.000 description 1
- HPNMFZURTQLUMO-UHFFFAOYSA-N diethylamine Chemical compound CCNCC HPNMFZURTQLUMO-UHFFFAOYSA-N 0.000 description 1
- 125000005043 dihydropyranyl group Chemical group O1C(CCC=C1)* 0.000 description 1
- 125000005879 dioxolanyl group Chemical group 0.000 description 1
- BFMYDTVEBKDAKJ-UHFFFAOYSA-L disodium;(2',7'-dibromo-3',6'-dioxido-3-oxospiro[2-benzofuran-1,9'-xanthene]-4'-yl)mercury;hydrate Chemical compound O.[Na+].[Na+].O1C(=O)C2=CC=CC=C2C21C1=CC(Br)=C([O-])C([Hg])=C1OC1=C2C=C(Br)C([O-])=C1 BFMYDTVEBKDAKJ-UHFFFAOYSA-L 0.000 description 1
- VSJKWCGYPAHWDS-UHFFFAOYSA-N dl-camptothecin Natural products C1=CC=C2C=C(CN3C4=CC5=C(C3=O)COC(=O)C5(O)CC)C4=NC2=C1 VSJKWCGYPAHWDS-UHFFFAOYSA-N 0.000 description 1
- MOTZDAYCYVMXPC-UHFFFAOYSA-N dodecyl hydrogen sulfate Chemical compound CCCCCCCCCCCCOS(O)(=O)=O MOTZDAYCYVMXPC-UHFFFAOYSA-N 0.000 description 1
- 229940043264 dodecyl sulfate Drugs 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 231100000673 dose–response relationship Toxicity 0.000 description 1
- 238000009510 drug design Methods 0.000 description 1
- 238000009509 drug development Methods 0.000 description 1
- 125000006575 electron-withdrawing group Chemical group 0.000 description 1
- 238000002330 electrospray ionisation mass spectrometry Methods 0.000 description 1
- 239000003974 emollient agent Substances 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- ZSWFCLXCOIISFI-UHFFFAOYSA-N endo-cyclopentadiene Natural products C1C=CC=C1 ZSWFCLXCOIISFI-UHFFFAOYSA-N 0.000 description 1
- YJGVMLPVUAXIQN-UHFFFAOYSA-N epipodophyllotoxin Natural products COC1=C(OC)C(OC)=CC(C2C3=CC=4OCOC=4C=C3C(O)C3C2C(OC3)=O)=C1 YJGVMLPVUAXIQN-UHFFFAOYSA-N 0.000 description 1
- 229930013356 epothilone Natural products 0.000 description 1
- HESCAJZNRMSMJG-KKQRBIROSA-N epothilone A Chemical class C/C([C@@H]1C[C@@H]2O[C@@H]2CCC[C@@H]([C@@H]([C@@H](C)C(=O)C(C)(C)[C@@H](O)CC(=O)O1)O)C)=C\C1=CSC(C)=N1 HESCAJZNRMSMJG-KKQRBIROSA-N 0.000 description 1
- 108700020302 erbB-2 Genes Proteins 0.000 description 1
- 125000004185 ester group Chemical group 0.000 description 1
- FRPJXPJMRWBBIH-RBRWEJTLSA-N estramustine Chemical compound ClCCN(CCCl)C(=O)OC1=CC=C2[C@H]3CC[C@](C)([C@H](CC4)O)[C@@H]4[C@@H]3CCC2=C1 FRPJXPJMRWBBIH-RBRWEJTLSA-N 0.000 description 1
- 229960001842 estramustine Drugs 0.000 description 1
- CCIVGXIOQKPBKL-UHFFFAOYSA-M ethanesulfonate Chemical compound CCS([O-])(=O)=O CCIVGXIOQKPBKL-UHFFFAOYSA-M 0.000 description 1
- LVGKNOAMLMIIKO-QXMHVHEDSA-N ethyl oleate Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OCC LVGKNOAMLMIIKO-QXMHVHEDSA-N 0.000 description 1
- 229940093471 ethyl oleate Drugs 0.000 description 1
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 description 1
- VJJPUSNTGOMMGY-MRVIYFEKSA-N etoposide Chemical compound COC1=C(O)C(OC)=CC([C@@H]2C3=CC=4OCOC=4C=C3[C@@H](O[C@H]3[C@@H]([C@@H](O)[C@@H]4O[C@H](C)OC[C@H]4O3)O)[C@@H]3[C@@H]2C(OC3)=O)=C1 VJJPUSNTGOMMGY-MRVIYFEKSA-N 0.000 description 1
- 229960005420 etoposide Drugs 0.000 description 1
- LIQODXNTTZAGID-OCBXBXKTSA-N etoposide phosphate Chemical compound COC1=C(OP(O)(O)=O)C(OC)=CC([C@@H]2C3=CC=4OCOC=4C=C3[C@@H](O[C@H]3[C@@H]([C@@H](O)[C@@H]4O[C@H](C)OC[C@H]4O3)O)[C@@H]3[C@@H]2C(OC3)=O)=C1 LIQODXNTTZAGID-OCBXBXKTSA-N 0.000 description 1
- 229960000752 etoposide phosphate Drugs 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 230000007717 exclusion Effects 0.000 description 1
- 230000001747 exhibiting effect Effects 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 238000000855 fermentation Methods 0.000 description 1
- 230000004151 fermentation Effects 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 239000000706 filtrate Substances 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- GNBHRKFJIUUOQI-UHFFFAOYSA-N fluorescein Chemical compound O1C(=O)C2=CC=CC=C2C21C1=CC=C(O)C=C1OC1=CC(O)=CC=C21 GNBHRKFJIUUOQI-UHFFFAOYSA-N 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 229960002949 fluorouracil Drugs 0.000 description 1
- MKXKFYHWDHIYRV-UHFFFAOYSA-N flutamide Chemical compound CC(C)C(=O)NC1=CC=C([N+]([O-])=O)C(C(F)(F)F)=C1 MKXKFYHWDHIYRV-UHFFFAOYSA-N 0.000 description 1
- 229960002074 flutamide Drugs 0.000 description 1
- 235000019253 formic acid Nutrition 0.000 description 1
- 238000001640 fractional crystallisation Methods 0.000 description 1
- 239000012458 free base Substances 0.000 description 1
- VZCYOOQTPOCHFL-OWOJBTEDSA-L fumarate(2-) Chemical compound [O-]C(=O)\C=C\C([O-])=O VZCYOOQTPOCHFL-OWOJBTEDSA-L 0.000 description 1
- 238000002825 functional assay Methods 0.000 description 1
- 210000001035 gastrointestinal tract Anatomy 0.000 description 1
- SDUQYLNIPVEERB-QPPQHZFASA-N gemcitabine Chemical compound O=C1N=C(N)C=CN1[C@H]1C(F)(F)[C@H](O)[C@@H](CO)O1 SDUQYLNIPVEERB-QPPQHZFASA-N 0.000 description 1
- 229960005277 gemcitabine Drugs 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 150000002334 glycols Chemical class 0.000 description 1
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 1
- 229910052737 gold Inorganic materials 0.000 description 1
- 239000010931 gold Substances 0.000 description 1
- 239000001963 growth medium Substances 0.000 description 1
- LHGVFZTZFXWLCP-UHFFFAOYSA-N guaiacol Chemical class COC1=CC=CC=C1O LHGVFZTZFXWLCP-UHFFFAOYSA-N 0.000 description 1
- 125000000262 haloalkenyl group Chemical group 0.000 description 1
- 125000004438 haloalkoxy group Chemical group 0.000 description 1
- 125000000232 haloalkynyl group Chemical group 0.000 description 1
- 230000002140 halogenating effect Effects 0.000 description 1
- 239000007902 hard capsule Substances 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 108060003552 hemocyanin Proteins 0.000 description 1
- 230000002440 hepatic effect Effects 0.000 description 1
- MNWFXJYAOYHMED-UHFFFAOYSA-N heptanoic acid Chemical compound CCCCCCC(O)=O MNWFXJYAOYHMED-UHFFFAOYSA-N 0.000 description 1
- 125000003187 heptyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- UUVWYPNAQBNQJQ-UHFFFAOYSA-N hexamethylmelamine Chemical compound CN(C)C1=NC(N(C)C)=NC(N(C)C)=N1 UUVWYPNAQBNQJQ-UHFFFAOYSA-N 0.000 description 1
- FBPFZTCFMRRESA-UHFFFAOYSA-N hexane-1,2,3,4,5,6-hexol Chemical compound OCC(O)C(O)C(O)C(O)CO FBPFZTCFMRRESA-UHFFFAOYSA-N 0.000 description 1
- FUZZWVXGSFPDMH-UHFFFAOYSA-N hexanoic acid Chemical compound CCCCCC(O)=O FUZZWVXGSFPDMH-UHFFFAOYSA-N 0.000 description 1
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- BHEPBYXIRTUNPN-UHFFFAOYSA-N hydridophosphorus(.) (triplet) Chemical compound [PH] BHEPBYXIRTUNPN-UHFFFAOYSA-N 0.000 description 1
- 150000002430 hydrocarbons Chemical group 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- ZMZDMBWJUHKJPS-UHFFFAOYSA-N hydrogen thiocyanate Natural products SC#N ZMZDMBWJUHKJPS-UHFFFAOYSA-N 0.000 description 1
- 239000008172 hydrogenated vegetable oil Substances 0.000 description 1
- 238000005984 hydrogenation reaction Methods 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-M hydrogensulfate Chemical compound OS([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-M 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-M hydroxide Chemical compound [OH-] XLYOFNOQVPJJNP-UHFFFAOYSA-M 0.000 description 1
- 235000010977 hydroxypropyl cellulose Nutrition 0.000 description 1
- 239000001863 hydroxypropyl cellulose Substances 0.000 description 1
- 238000003384 imaging method Methods 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- 230000003100 immobilizing effect Effects 0.000 description 1
- 238000003119 immunoblot Methods 0.000 description 1
- 230000002055 immunohistochemical effect Effects 0.000 description 1
- 238000001114 immunoprecipitation Methods 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 238000000338 in vitro Methods 0.000 description 1
- 238000000099 in vitro assay Methods 0.000 description 1
- 238000005462 in vivo assay Methods 0.000 description 1
- 125000003392 indanyl group Chemical group C1(CCC2=CC=CC=C12)* 0.000 description 1
- 150000002475 indoles Chemical class 0.000 description 1
- 125000003406 indolizinyl group Chemical group C=1(C=CN2C=CC=CC12)* 0.000 description 1
- 125000001041 indolyl group Chemical group 0.000 description 1
- 239000003701 inert diluent Substances 0.000 description 1
- 230000008595 infiltration Effects 0.000 description 1
- 238000001764 infiltration Methods 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 230000000977 initiatory effect Effects 0.000 description 1
- 229940102223 injectable solution Drugs 0.000 description 1
- 229940102213 injectable suspension Drugs 0.000 description 1
- 230000000266 injurious effect Effects 0.000 description 1
- 150000007529 inorganic bases Chemical class 0.000 description 1
- 229940047124 interferons Drugs 0.000 description 1
- 229940047122 interleukins Drugs 0.000 description 1
- 238000006361 intramolecular Friedel-Crafts acylation reaction Methods 0.000 description 1
- 238000007912 intraperitoneal administration Methods 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- UWKQSNNFCGGAFS-XIFFEERXSA-N irinotecan Chemical compound C1=C2C(CC)=C3CN(C(C4=C([C@@](C(=O)OC4)(O)CC)C=4)=O)C=4C3=NC2=CC=C1OC(=O)N(CC1)CCC1N1CCCCC1 UWKQSNNFCGGAFS-XIFFEERXSA-N 0.000 description 1
- 230000007794 irritation Effects 0.000 description 1
- SUMDYPCJJOFFON-UHFFFAOYSA-N isethionic acid Chemical compound OCCS(O)(=O)=O SUMDYPCJJOFFON-UHFFFAOYSA-N 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- QXJSBBXBKPUZAA-UHFFFAOYSA-N isooleic acid Natural products CCCCCCCC=CCCCCCCCCC(O)=O QXJSBBXBKPUZAA-UHFFFAOYSA-N 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 125000000842 isoxazolyl group Chemical group 0.000 description 1
- 235000015110 jellies Nutrition 0.000 description 1
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 1
- 150000002576 ketones Chemical class 0.000 description 1
- TYQCGQRIZGCHNB-JLAZNSOCSA-N l-ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(O)=C(O)C1=O TYQCGQRIZGCHNB-JLAZNSOCSA-N 0.000 description 1
- GFIJNRVAKGFPGQ-LIJARHBVSA-N leuprolide Chemical compound CCNC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](CC(C)C)NC(=O)[C@@H](NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1N=CNC=1)NC(=O)[C@H]1NC(=O)CC1)CC1=CC=C(O)C=C1 GFIJNRVAKGFPGQ-LIJARHBVSA-N 0.000 description 1
- 229960004338 leuprorelin Drugs 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- 239000007937 lozenge Substances 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 210000004324 lymphatic system Anatomy 0.000 description 1
- 230000002934 lysing effect Effects 0.000 description 1
- 239000012139 lysis buffer Substances 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 229910001629 magnesium chloride Inorganic materials 0.000 description 1
- VTHJTEIRLNZDEV-UHFFFAOYSA-L magnesium dihydroxide Chemical compound [OH-].[OH-].[Mg+2] VTHJTEIRLNZDEV-UHFFFAOYSA-L 0.000 description 1
- 239000000347 magnesium hydroxide Substances 0.000 description 1
- 229910001862 magnesium hydroxide Inorganic materials 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 238000002595 magnetic resonance imaging Methods 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000000594 mannitol Substances 0.000 description 1
- 235000010355 mannitol Nutrition 0.000 description 1
- 230000000873 masking effect Effects 0.000 description 1
- 229960001428 mercaptopurine Drugs 0.000 description 1
- 108020004999 messenger RNA Proteins 0.000 description 1
- 239000002207 metabolite Substances 0.000 description 1
- 238000006263 metalation reaction Methods 0.000 description 1
- FBOZXECLQNJBKD-UHFFFAOYSA-N methotrexate Chemical compound C=1N=C2N=C(N)N=C(N)C2=NC=1CN(C)C1=CC=C(C(=O)NC(CCC(O)=O)C(O)=O)C=C1 FBOZXECLQNJBKD-UHFFFAOYSA-N 0.000 description 1
- 229960000485 methotrexate Drugs 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 235000010981 methylcellulose Nutrition 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- HRHKSTOGXBBQCB-VFWICMBZSA-N methylmitomycin Chemical compound O=C1C(N)=C(C)C(=O)C2=C1[C@@H](COC(N)=O)[C@@]1(OC)[C@H]3N(C)[C@H]3CN12 HRHKSTOGXBBQCB-VFWICMBZSA-N 0.000 description 1
- 229940016286 microcrystalline cellulose Drugs 0.000 description 1
- 235000019813 microcrystalline cellulose Nutrition 0.000 description 1
- 239000008108 microcrystalline cellulose Substances 0.000 description 1
- 230000005012 migration Effects 0.000 description 1
- 238000013508 migration Methods 0.000 description 1
- 229960004857 mitomycin Drugs 0.000 description 1
- KKZJGLLVHKMTCM-UHFFFAOYSA-N mitoxantrone Chemical compound O=C1C2=C(O)C=CC(O)=C2C(=O)C2=C1C(NCCNCCO)=CC=C2NCCNCCO KKZJGLLVHKMTCM-UHFFFAOYSA-N 0.000 description 1
- 229960001156 mitoxantrone Drugs 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 238000010369 molecular cloning Methods 0.000 description 1
- 239000003068 molecular probe Substances 0.000 description 1
- 230000000877 morphologic effect Effects 0.000 description 1
- 239000002324 mouth wash Substances 0.000 description 1
- 201000006417 multiple sclerosis Diseases 0.000 description 1
- ZLSLXRZFIXTXQE-UHFFFAOYSA-N n-[4-oxo-6-(pyrrolidin-1-ylmethyl)-1,7-dihydropyrrolo[2,3-d]pyrimidin-2-yl]octanamide Chemical compound C=1C=2C(=O)NC(NC(=O)CCCCCCC)=NC=2NC=1CN1CCCC1 ZLSLXRZFIXTXQE-UHFFFAOYSA-N 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001624 naphthyl group Chemical group 0.000 description 1
- 230000001613 neoplastic effect Effects 0.000 description 1
- 210000005036 nerve Anatomy 0.000 description 1
- 238000006386 neutralization reaction Methods 0.000 description 1
- 235000001968 nicotinic acid Nutrition 0.000 description 1
- 239000011664 nicotinic acid Substances 0.000 description 1
- 238000006396 nitration reaction Methods 0.000 description 1
- 231100000344 non-irritating Toxicity 0.000 description 1
- 108091008569 nuclear steroid hormone receptors Proteins 0.000 description 1
- 239000002777 nucleoside Substances 0.000 description 1
- 125000003835 nucleoside group Chemical group 0.000 description 1
- GYCKQBWUSACYIF-UHFFFAOYSA-N o-hydroxybenzoic acid ethyl ester Natural products CCOC(=O)C1=CC=CC=C1O GYCKQBWUSACYIF-UHFFFAOYSA-N 0.000 description 1
- 239000007764 o/w emulsion Substances 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 1
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 150000007530 organic bases Chemical class 0.000 description 1
- 239000012044 organic layer Substances 0.000 description 1
- 150000002905 orthoesters Chemical class 0.000 description 1
- 125000002971 oxazolyl group Chemical group 0.000 description 1
- 239000007800 oxidant agent Substances 0.000 description 1
- 229960001592 paclitaxel Drugs 0.000 description 1
- QNGNSVIICDLXHT-UHFFFAOYSA-N para-ethylbenzaldehyde Natural products CCC1=CC=C(C=O)C=C1 QNGNSVIICDLXHT-UHFFFAOYSA-N 0.000 description 1
- 239000012188 paraffin wax Substances 0.000 description 1
- 230000037361 pathway Effects 0.000 description 1
- 239000013610 patient sample Substances 0.000 description 1
- 125000006340 pentafluoro ethyl group Chemical group FC(F)(F)C(F)(F)* 0.000 description 1
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 1
- 230000002093 peripheral effect Effects 0.000 description 1
- 229940083251 peripheral vasodilators purine derivative Drugs 0.000 description 1
- JRKICGRDRMAZLK-UHFFFAOYSA-L peroxydisulfate Chemical compound [O-]S(=O)(=O)OOS([O-])(=O)=O JRKICGRDRMAZLK-UHFFFAOYSA-L 0.000 description 1
- 239000000825 pharmaceutical preparation Substances 0.000 description 1
- 230000000144 pharmacologic effect Effects 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 239000008363 phosphate buffer Substances 0.000 description 1
- 150000003014 phosphoric acid esters Chemical class 0.000 description 1
- IPNPIHIZVLFAFP-UHFFFAOYSA-N phosphorus tribromide Chemical compound BrP(Br)Br IPNPIHIZVLFAFP-UHFFFAOYSA-N 0.000 description 1
- CFZKDDTWZYUZKS-UHFFFAOYSA-N picoline N-oxide Chemical compound CC1=CC=CC=[N+]1[O-] CFZKDDTWZYUZKS-UHFFFAOYSA-N 0.000 description 1
- 229940075930 picrate Drugs 0.000 description 1
- OXNIZHLAWKMVMX-UHFFFAOYSA-M picrate anion Chemical compound [O-]C1=C([N+]([O-])=O)C=C([N+]([O-])=O)C=C1[N+]([O-])=O OXNIZHLAWKMVMX-UHFFFAOYSA-M 0.000 description 1
- 125000004193 piperazinyl group Chemical group 0.000 description 1
- 125000003386 piperidinyl group Chemical group 0.000 description 1
- IUGYQRQAERSCNH-UHFFFAOYSA-M pivalate Chemical compound CC(C)(C)C([O-])=O IUGYQRQAERSCNH-UHFFFAOYSA-M 0.000 description 1
- 229950010765 pivalate Drugs 0.000 description 1
- YVCVYCSAAZQOJI-UHFFFAOYSA-N podophyllotoxin Natural products COC1=C(O)C(OC)=CC(C2C3=CC=4OCOC=4C=C3C(O)C3C2C(OC3)=O)=C1 YVCVYCSAAZQOJI-UHFFFAOYSA-N 0.000 description 1
- 239000003880 polar aprotic solvent Substances 0.000 description 1
- 125000003367 polycyclic group Chemical group 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 229920005862 polyol Polymers 0.000 description 1
- 150000003077 polyols Chemical class 0.000 description 1
- 235000010482 polyoxyethylene sorbitan monooleate Nutrition 0.000 description 1
- 239000000244 polyoxyethylene sorbitan monooleate Substances 0.000 description 1
- 229920000053 polysorbate 80 Polymers 0.000 description 1
- 229950004406 porfiromycin Drugs 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 229920001592 potato starch Polymers 0.000 description 1
- 230000001855 preneoplastic effect Effects 0.000 description 1
- 230000002335 preservative effect Effects 0.000 description 1
- 150000003141 primary amines Chemical class 0.000 description 1
- 239000013615 primer Substances 0.000 description 1
- CPTBDICYNRMXFX-UHFFFAOYSA-N procarbazine Chemical compound CNNCC1=CC=C(C(=O)NC(C)C)C=C1 CPTBDICYNRMXFX-UHFFFAOYSA-N 0.000 description 1
- 229960000624 procarbazine Drugs 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 230000002035 prolonged effect Effects 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- 125000004368 propenyl group Chemical group C(=CC)* 0.000 description 1
- 235000013772 propylene glycol Nutrition 0.000 description 1
- 230000012846 protein folding Effects 0.000 description 1
- 108060006633 protein kinase Proteins 0.000 description 1
- 238000000425 proton nuclear magnetic resonance spectrum Methods 0.000 description 1
- 150000003195 pteridines Chemical class 0.000 description 1
- 150000003212 purines Chemical class 0.000 description 1
- 125000003226 pyrazolyl group Chemical group 0.000 description 1
- 150000003222 pyridines Chemical class 0.000 description 1
- 150000003230 pyrimidines Chemical class 0.000 description 1
- 150000003233 pyrroles Chemical class 0.000 description 1
- 125000000719 pyrrolidinyl group Chemical group 0.000 description 1
- 238000011002 quantification Methods 0.000 description 1
- 238000005956 quaternization reaction Methods 0.000 description 1
- 150000003248 quinolines Chemical class 0.000 description 1
- 125000002943 quinolinyl group Chemical group N1=C(C=CC2=CC=CC=C12)* 0.000 description 1
- 238000003127 radioimmunoassay Methods 0.000 description 1
- 239000011541 reaction mixture Substances 0.000 description 1
- 210000000664 rectum Anatomy 0.000 description 1
- 230000008929 regeneration Effects 0.000 description 1
- 238000011069 regeneration method Methods 0.000 description 1
- 230000022983 regulation of cell cycle Effects 0.000 description 1
- 230000022532 regulation of transcription, DNA-dependent Effects 0.000 description 1
- 238000003757 reverse transcription PCR Methods 0.000 description 1
- 238000012552 review Methods 0.000 description 1
- PYWVYCXTNDRMGF-UHFFFAOYSA-N rhodamine B Chemical compound [Cl-].C=12C=CC(=[N+](CC)CC)C=C2OC2=CC(N(CC)CC)=CC=C2C=1C1=CC=CC=C1C(O)=O PYWVYCXTNDRMGF-UHFFFAOYSA-N 0.000 description 1
- 108091092562 ribozyme Proteins 0.000 description 1
- 229910001927 ruthenium tetroxide Inorganic materials 0.000 description 1
- CVHZOJJKTDOEJC-UHFFFAOYSA-N saccharin Chemical compound C1=CC=C2C(=O)NS(=O)(=O)C2=C1 CVHZOJJKTDOEJC-UHFFFAOYSA-N 0.000 description 1
- 235000019204 saccharin Nutrition 0.000 description 1
- 229940081974 saccharin Drugs 0.000 description 1
- 239000000901 saccharin and its Na,K and Ca salt Substances 0.000 description 1
- 235000005713 safflower oil Nutrition 0.000 description 1
- 239000003813 safflower oil Substances 0.000 description 1
- YGSDEFSMJLZEOE-UHFFFAOYSA-M salicylate Chemical compound OC1=CC=CC=C1C([O-])=O YGSDEFSMJLZEOE-UHFFFAOYSA-M 0.000 description 1
- 229960001860 salicylate Drugs 0.000 description 1
- 239000012723 sample buffer Substances 0.000 description 1
- 229930195734 saturated hydrocarbon Natural products 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 150000003335 secondary amines Chemical class 0.000 description 1
- 125000000467 secondary amino group Chemical group [H]N([*:1])[*:2] 0.000 description 1
- 229910052711 selenium Inorganic materials 0.000 description 1
- 239000011669 selenium Substances 0.000 description 1
- 230000011664 signaling Effects 0.000 description 1
- 239000002002 slurry Substances 0.000 description 1
- AWUCVROLDVIAJX-GSVOUGTGSA-N sn-glycerol 3-phosphate Chemical compound OC[C@@H](O)COP(O)(O)=O AWUCVROLDVIAJX-GSVOUGTGSA-N 0.000 description 1
- 235000010413 sodium alginate Nutrition 0.000 description 1
- 239000000661 sodium alginate Substances 0.000 description 1
- 229940005550 sodium alginate Drugs 0.000 description 1
- 229910001467 sodium calcium phosphate Inorganic materials 0.000 description 1
- 238000002415 sodium dodecyl sulfate polyacrylamide gel electrophoresis Methods 0.000 description 1
- 229910000104 sodium hydride Inorganic materials 0.000 description 1
- 239000001488 sodium phosphate Substances 0.000 description 1
- GEHJYWRUCIMESM-UHFFFAOYSA-L sodium sulfite Chemical compound [Na+].[Na+].[O-]S([O-])=O GEHJYWRUCIMESM-UHFFFAOYSA-L 0.000 description 1
- AKHNMLFCWUSKQB-UHFFFAOYSA-L sodium thiosulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=S AKHNMLFCWUSKQB-UHFFFAOYSA-L 0.000 description 1
- 239000007901 soft capsule Substances 0.000 description 1
- 229940083466 soybean lecithin Drugs 0.000 description 1
- 238000010186 staining Methods 0.000 description 1
- 239000008117 stearic acid Substances 0.000 description 1
- 108020003113 steroid hormone receptors Proteins 0.000 description 1
- 125000000547 substituted alkyl group Chemical group 0.000 description 1
- 125000005346 substituted cycloalkyl group Chemical group 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-L succinate(2-) Chemical compound [O-]C(=O)CCC([O-])=O KDYFGRWQOYBRFD-UHFFFAOYSA-L 0.000 description 1
- 150000003462 sulfoxides Chemical class 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 230000002459 sustained effect Effects 0.000 description 1
- 230000008685 targeting Effects 0.000 description 1
- 229940095064 tartrate Drugs 0.000 description 1
- RCINICONZNJXQF-MZXODVADSA-N taxol Chemical compound O([C@@H]1[C@@]2(C[C@@H](C(C)=C(C2(C)C)[C@H](C([C@]2(C)[C@@H](O)C[C@H]3OC[C@]3([C@H]21)OC(C)=O)=O)OC(=O)C)OC(=O)[C@H](O)[C@@H](NC(=O)C=1C=CC=CC=1)C=1C=CC=CC=1)O)C(=O)C1=CC=CC=C1 RCINICONZNJXQF-MZXODVADSA-N 0.000 description 1
- NRUKOCRGYNPUPR-QBPJDGROSA-N teniposide Chemical compound COC1=C(O)C(OC)=CC([C@@H]2C3=CC=4OCOC=4C=C3[C@@H](O[C@H]3[C@@H]([C@@H](O)[C@@H]4O[C@@H](OC[C@H]4O3)C=3SC=CC=3)O)[C@@H]3[C@@H]2C(OC3)=O)=C1 NRUKOCRGYNPUPR-QBPJDGROSA-N 0.000 description 1
- 229960001278 teniposide Drugs 0.000 description 1
- DYHSDKLCOJIUFX-UHFFFAOYSA-N tert-butoxycarbonyl anhydride Chemical compound CC(C)(C)OC(=O)OC(=O)OC(C)(C)C DYHSDKLCOJIUFX-UHFFFAOYSA-N 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- HJUGFYREWKUQJT-UHFFFAOYSA-N tetrabromomethane Chemical compound BrC(Br)(Br)Br HJUGFYREWKUQJT-UHFFFAOYSA-N 0.000 description 1
- FPGGTKZVZWFYPV-UHFFFAOYSA-M tetrabutylammonium fluoride Chemical compound [F-].CCCC[N+](CCCC)(CCCC)CCCC FPGGTKZVZWFYPV-UHFFFAOYSA-M 0.000 description 1
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 1
- 125000003718 tetrahydrofuranyl group Chemical group 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- 238000004809 thin layer chromatography Methods 0.000 description 1
- 125000000101 thioether group Chemical group 0.000 description 1
- 150000003568 thioethers Chemical class 0.000 description 1
- 229960001196 thiotepa Drugs 0.000 description 1
- 230000036964 tight binding Effects 0.000 description 1
- 229960000984 tocofersolan Drugs 0.000 description 1
- 125000003944 tolyl group Chemical group 0.000 description 1
- UCFGDBYHRUNTLO-QHCPKHFHSA-N topotecan Chemical compound C1=C(O)C(CN(C)C)=C2C=C(CN3C4=CC5=C(C3=O)COC(=O)[C@]5(O)CC)C4=NC2=C1 UCFGDBYHRUNTLO-QHCPKHFHSA-N 0.000 description 1
- 229960000303 topotecan Drugs 0.000 description 1
- 125000002088 tosyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1C([H])([H])[H])S(*)(=O)=O 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 235000010487 tragacanth Nutrition 0.000 description 1
- 239000000196 tragacanth Substances 0.000 description 1
- 229940116362 tragacanth Drugs 0.000 description 1
- 230000037317 transdermal delivery Effects 0.000 description 1
- GKASDNZWUGIAMG-UHFFFAOYSA-N triethyl orthoformate Chemical compound CCOC(OCC)OCC GKASDNZWUGIAMG-UHFFFAOYSA-N 0.000 description 1
- CWMFRHBXRUITQE-UHFFFAOYSA-N trimethylsilylacetylene Chemical group C[Si](C)(C)C#C CWMFRHBXRUITQE-UHFFFAOYSA-N 0.000 description 1
- NOYPYLRCIDNJJB-UHFFFAOYSA-N trimetrexate Chemical compound COC1=C(OC)C(OC)=CC(NCC=2C(=C3C(N)=NC(N)=NC3=CC=2)C)=C1 NOYPYLRCIDNJJB-UHFFFAOYSA-N 0.000 description 1
- 229960001099 trimetrexate Drugs 0.000 description 1
- RIOQSEWOXXDEQQ-UHFFFAOYSA-O triphenylphosphanium Chemical compound C1=CC=CC=C1[PH+](C=1C=CC=CC=1)C1=CC=CC=C1 RIOQSEWOXXDEQQ-UHFFFAOYSA-O 0.000 description 1
- RYFMWSXOAZQYPI-UHFFFAOYSA-K trisodium phosphate Chemical compound [Na+].[Na+].[Na+].[O-]P([O-])([O-])=O RYFMWSXOAZQYPI-UHFFFAOYSA-K 0.000 description 1
- 239000012588 trypsin Substances 0.000 description 1
- 210000004881 tumor cell Anatomy 0.000 description 1
- 230000004614 tumor growth Effects 0.000 description 1
- 238000000825 ultraviolet detection Methods 0.000 description 1
- ZDPHROOEEOARMN-UHFFFAOYSA-N undecanoic acid Chemical compound CCCCCCCCCCC(O)=O ZDPHROOEEOARMN-UHFFFAOYSA-N 0.000 description 1
- 230000003827 upregulation Effects 0.000 description 1
- 229960003048 vinblastine Drugs 0.000 description 1
- JXLYSJRDGCGARV-XQKSVPLYSA-N vincaleukoblastine Chemical compound C([C@@H](C[C@]1(C(=O)OC)C=2C(=CC3=C([C@]45[C@H]([C@@]([C@H](OC(C)=O)[C@]6(CC)C=CCN([C@H]56)CC4)(O)C(=O)OC)N3C)C=2)OC)C[C@@](C2)(O)CC)N2CCC2=C1NC1=CC=CC=C21 JXLYSJRDGCGARV-XQKSVPLYSA-N 0.000 description 1
- OGWKCGZFUXNPDA-XQKSVPLYSA-N vincristine Chemical compound C([N@]1C[C@@H](C[C@]2(C(=O)OC)C=3C(=CC4=C([C@]56[C@H]([C@@]([C@H](OC(C)=O)[C@]7(CC)C=CCN([C@H]67)CC5)(O)C(=O)OC)N4C=O)C=3)OC)C[C@@](C1)(O)CC)CC1=C2NC2=CC=CC=C12 OGWKCGZFUXNPDA-XQKSVPLYSA-N 0.000 description 1
- 229960004528 vincristine Drugs 0.000 description 1
- OGWKCGZFUXNPDA-UHFFFAOYSA-N vincristine Natural products C1C(CC)(O)CC(CC2(C(=O)OC)C=3C(=CC4=C(C56C(C(C(OC(C)=O)C7(CC)C=CCN(C67)CC5)(O)C(=O)OC)N4C=O)C=3)OC)CN1CCC1=C2NC2=CC=CC=C12 OGWKCGZFUXNPDA-UHFFFAOYSA-N 0.000 description 1
- HHJUWIANJFBDHT-KOTLKJBCSA-N vindesine Chemical compound C([N@]1C[C@@H](C[C@]2(C(=O)OC)C=3C(=CC4=C([C@]56[C@H]([C@@]([C@H](O)[C@]7(CC)C=CCN([C@H]67)CC5)(O)C(N)=O)N4C)C=3)OC)C[C@@](C1)(O)CC)CC1=C2NC2=CC=CC=C12 HHJUWIANJFBDHT-KOTLKJBCSA-N 0.000 description 1
- 229960004355 vindesine Drugs 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- 239000001993 wax Substances 0.000 description 1
- 230000036642 wellbeing Effects 0.000 description 1
- 125000005023 xylyl group Chemical group 0.000 description 1
- 239000002076 α-tocopherol Substances 0.000 description 1
- 235000004835 α-tocopherol Nutrition 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/16—Drugs for disorders of the alimentary tract or the digestive system for liver or gallbladder disorders, e.g. hepatoprotective agents, cholagogues, litholytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/12—Drugs for disorders of the urinary system of the kidneys
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/02—Drugs for dermatological disorders for treating wounds, ulcers, burns, scars, keloids, or the like
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/04—Drugs for skeletal disorders for non-specific disorders of the connective tissue
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P21/00—Drugs for disorders of the muscular or neuromuscular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/08—Antiepileptics; Anticonvulsants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/14—Antivirals for RNA viruses
- A61P31/16—Antivirals for RNA viruses for influenza or rhinoviruses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/02—Antineoplastic agents specific for leukemia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/06—Immunosuppressants, e.g. drugs for graft rejection
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D473/00—Heterocyclic compounds containing purine ring systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D473/00—Heterocyclic compounds containing purine ring systems
- C07D473/02—Heterocyclic compounds containing purine ring systems with oxygen, sulphur, or nitrogen atoms directly attached in positions 2 and 6
- C07D473/18—Heterocyclic compounds containing purine ring systems with oxygen, sulphur, or nitrogen atoms directly attached in positions 2 and 6 one oxygen and one nitrogen atom, e.g. guanine
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D473/00—Heterocyclic compounds containing purine ring systems
- C07D473/02—Heterocyclic compounds containing purine ring systems with oxygen, sulphur, or nitrogen atoms directly attached in positions 2 and 6
- C07D473/24—Heterocyclic compounds containing purine ring systems with oxygen, sulphur, or nitrogen atoms directly attached in positions 2 and 6 one nitrogen and one sulfur atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D473/00—Heterocyclic compounds containing purine ring systems
- C07D473/26—Heterocyclic compounds containing purine ring systems with an oxygen, sulphur, or nitrogen atom directly attached in position 2 or 6, but not in both
- C07D473/32—Nitrogen atom
Definitions
- the invention relates in general to pyrrolopyrimidines and their broad-spectrum utility, e.g., in inhibiting heat shock protein 90 (HSP90) to thereby treat or prevent HSP90-mediated diseases.
- HSP90 heat shock protein 90
- HSP90s are ubiquitous chaperone proteins that are involved in folding, activation and assembly of a wide range of proteins, including key proteins involved in signal transduction, cell cycle control and transcriptional regulation.
- HSP90 chaperone proteins are associated with important signaling proteins, such as steroid hormone receptors and protein kinases, including, e.g., Raf-1, EGFR, v-Src family kinases, Cdk4, and ErbB-2 (Buchner J. TIBS 1999, 24, 136-141; Stepanova, L. et al. Genes Dev . 1996, 10, 1491-502; Dai, K. et al. J. Biol. Chem . 1996, 271, 22030-4).
- HSP70 e.g., HSP70, p60/Hop/Sti1, Hip, Bag1, HSP40/Hdj2/Hsj1, immunophilins, p23, and p50
- HSP90 may assist HSP90 in its function (see, e.g., Caplan, A. Trends in Cell Biol . 1999, 9, 262-68).
- Ansamycin antibiotics e.g., herbimycin A (HA), geldanamycin (GM), and 17-allylamninogeldanamycin (17-AAG) are thought to exert their anticancerous effects by tight binding of the N-terminus pocket of HSP90, thereby destabilizing substrates that normally interact with HSP90 (Stebbins, C. et al. Cell 1997, 89, 239-250).
- This pocket is highly conserved and has weak homology to the ATP-binding site of DNA gyrase (Stebbins, C. et al., supra; Grenert, J. P. et al. J. Biol. Chem . 1997, 272, 23843-50).
- ATP and ADP have both been shown to bind this pocket with low affinity and to have weak ATPase activity (Proromou, C. et al. Cell 1997, 90, 65-75; Panaretou, B. et al. EMBO J . 1998, 17, 4829-36).
- In vitro and in vivo studies have demonstrated that occupancy of this N-terminal pocket by ansamycins and other HSP90 inhibitors alters HSP90 function and inhibits protein folding.
- ansamycins and other HSP90 inhibitors have been shown to prevent binding of protein substrates to HSP90 (Scheibel, T. H. et al. Proc. Natl. Acad. Sci.
- the substrates are degraded by a ubiquitin-dependent process in the proteasome (Schneider, C. L., supra; Sepp-Lorenzino, L., et al. J. Biol. Chem . 1995, 270, 16580-16587; Whitesell, L. et al. Proc. Natl. Acad. Sci. USA 1994, 91, 8324-8328).
- HSP90 substrate destabilization occurs in tumor and non-transformed cells alike and has been shown to be especially effective on a subset of signaling regulators, e.g., Raf (Schulte, T. W. et al. Biochem. Biophys. Res. Commun . 1997, 239, 655-9; Schulte, T. W., et al. J. Biol. Chem. 1995, 270, 24585-8), nuclear steroid receptors (Segnitz, B.; U. Gehring J. Biol. Chem . 1997, 272, 18694-18701; Smith, D. F. et al. Mol. Cell. Biol .
- Ansamycins thus hold great promise for the treatment and/or prevention of many types of cancers and proliferative disorders, and also hold promise as traditional antibiotics.
- their relative insolubility makes them difficult to formulate and administer, and they are not easily synthesized and currently must, at least in part, be generated through fermentation.
- the hepatic toxicity of ansamyins is dose limiting.
- HSP90 inhibitors have also been implicated in a wide variety of other utilities, including use as anti-inflammation agents, anti-infectious disease agents, agents for treating autoimmunity, agents for treating stroke, ischemia, multiple sclerosis, cardiac disorders, central nervous system related disorders and agents useful in promoting nerve regeneration (See, e.g., Rosen et al. WO 02/09696 (PCT/US01/23640); Degranco et al. WO 99/51223 (PCT/US99/07242); Gold, U.S. Pat. No. 6,210,974 B1; DeFranco et al., U.S. Pat. No. 6,174,875.
- fibrogenetic disorders including but not limited to scleroderma, polymyositis, systemic lupus, rheumatoid arthritis, liver cirrhosis, keloid formation, interstitial nephritis, and pulmonary fibrosis also may be treatable with HSP90 inhibitors.
- Still further HSP90 modulation, modulators and uses thereof are reported in Application Nos.
- the present invention is directed towards heterocyclic compounds, in particular, pyrrolopyrimidines and related compounds that show broad utility, e.g., by inhibiting HSP90 and treating diseases that are HSP90-dependent.
- the invention comprises heterocyclic compounds as specified below in Formulae A, I, II, III and IV.
- stereoisomic forms including the individual enantiomers and diastereomers, racemic mixtures, and diasteromeric mixtures, and combinations thereof, where appropriate, as well as polymorphs, specific racemates and stereoisomers, solvates, esters, tautomers, pharmaceutically acceptable salts and prodrugs of these compounds.
- Stereoisomers of the compounds of the present invention may be isolated by standard resolution techniques such as, for example, fractional crystallization and chiral column chromatography.
- the invention provides compounds of Formula A, or a polymorph, solvate, ester, tautomer, diastereomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, which show utility by inhibiting HSP90 and treating and preventing diseases that are HSP90-dependent.
- the invention features compounds of Formulae I, II, III, & IV: or a polymorph, solvate, ester, diastereomer, enantiomer, tautomer, pharmaceutically acceptable salt or prodrug thereof, wherein:
- the invention features pharmaceutical compositions comprising the compounds of the invention, in particular, the compounds of Formulae A, I, II, III or IV, or a polymorph, solvate, ester, tautomer, diastereoisomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, and one or more pharmaceutical excipients, for use in treatment or prevention of diseases that are HSP90-dependent.
- the invention features a method of treating an individual having an HSP90-mediated disorder by administering to the individual a pharmaceutical composition that comprises a pharmaceutically effective amount of a compound of Formula A, I, II, III or IV, or a polymorph, solvate, ester, tautomer, diastereomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof.
- the invention provides a method for treating an individual having a disorder selected from the group of inflammatory diseases, infections, autoimmune disorders, stroke, ischemia, cardiac disorders, neurological disorders, fibrogenetic disorders, proliferative disorders, tumors, leukemias, neoplasms, cancers, carcinomas, metabolic diseases, and malignant diseases.
- the invention provides a method for treating an individual having a fibrogenetic disorder, such as, for example, scleroderma, polymyositis, systemic lupus, rheumatoid arthritis, liver cirrhosis, keloid formation, interstitial nephritis or pulmonary fibrosis.
- a fibrogenetic disorder such as, for example, scleroderma, polymyositis, systemic lupus, rheumatoid arthritis, liver cirrhosis, keloid formation, interstitial nephritis or pulmonary fibrosis.
- the invention provides a combination therapy comprising the administration of a pharmaceutically effective amount of a compound of Formula I, II, III, or IV, or a solvate, tautomer, diastereomer, enantiomer, pharmaceutically acceptable salt, polymorph, or prodrug thereof according to any of the preceding aspects or embodiments, and at least one therapeutic agent selected from the group of cytotoxic agents, anti-angiogenesis agents and anti-neoplastic agents.
- the anti-neoplastic agent may be selected from the group of alkylating agents, anti-metabolites, epidophyllotoxins antineoplastic enzymes, topoisomerase inhibitors, procarbazines, mitoxantrones, platinum coordination complexes, biological response modifiers and growth inhibitors, hormonal/anti-hormonal therapeutic agents, and haematopoietic growth factors.
- Advantages of the invention depend on the specific aspect and embodiment and may include one or more of: ease of synthesis and/or formulation, solubility, and IC 50 relative to previously existing compounds in the same or different classes of HSP90 inhibitors.
- a “pharmaceutically acceptable derivative or prodrug” means any pharmaceutically acceptable salt, ester, salt of an ester or other derivative of a compound of this invention, which, upon administration to a recipient, is capable of providing, either directly or indirectly, a compound of this invention or a pharmaceutically active metabolite or residue thereof.
- Particularly favored derivatives or prodrugs are those that increase the bioavailability of the compounds of this invention when such compounds are administered to a patient (e.g., by allowing orally administered compound to be more readily absorbed into blood) or which enhance delivery of the parent compound to a biological compartment (e.g., the brain or lymphatic system).
- a “pharmaceutically acceptable salt” may be prepared for any compound of the invention having a functionality capable of forming a salt, for example, an acid or base functionality.
- Pharmaceutically acceptable salts may be derived from organic or inorganic acids and bases.
- Compounds of the invention that contain one or more basic functional groups, e.g., amino or alkylamino, are capable of forming pharmaceutically acceptable salts with pharmaceutically acceptable organic and inorganic acids.
- These salts can be prepared in situ during the final isolation and purification of the compounds of the invention, or by separately reacting a purified compound of the invention in its free base form with a suitable organic or inorganic acid, and isolating the salt thus formed.
- Suitable acid salts include acetate, adipate, alginate, aspartate, benzoate, benzenesulfonate, bisulfate, butyrate, citrate, camphorate, camphorsulfonate, cyclopentanepropionate, digluconate, dodecylsulfate, ethanesulfonate, formate, fumarate, glucoheptanoate, glycerophosphate, glycolate, hemisulfate, heptanoate, hexanoate, hydrochloride, hydrobromide, hydroiodide, 2-hydroxyethanesulfonate, lactate, maleate, malonate, methanesulfonate, 2-napthalenesulfonate, nicotinate, nitrate, oxalate, palmoate, pectinate, persulfate, 3-phenylpropionate, phosphate, picrate, pivalate, prop
- compositions of the present invention that contain one or more acidic functional groups are capable of forming pharmaceutically acceptable salts with pharmaceutically acceptable bases.
- pharmaceutically acceptable salts refers to the relatively non-toxic, inorganic and organic base addition salts of compounds of the present invention. These salts can likewise be prepared in situ during the final isolation and purification of the compounds, or by separately reacting the purified compound in its free acid form with a suitable base, such as the hydroxide, carbonate or bicarbonate of a pharmaceutically acceptable metal cation, with ammonia, or with a pharmaceutically acceptable organic primary, secondary or tertiary amine.
- Representative pharmaceutically acceptable cations include alkali or alkaline earth salts such as the lithium, sodium, potassium, calcium, magnesium, and aluminum salts and the like.
- Illustrative examples of some of the bases that can be used include sodium hydroxide, potassium hydroxide, choline hydroxide, sodium carbonate, N + (C 1-4 alkyl) 4 , and the like.
- Representative organic amines useful for the formation of base addition salts include ethylamine, diethylamine, ethylenediamine, ethanolamine, diethanolamine, piperazine and the like. This invention also envisions the quatemization of any basic nitrogen-containing groups of the compounds disclosed herein. Water or oil-soluble or dispersible products may be obtained by such quaternization. See, for example, Berge et al., supra.
- prodrugs of the compounds of this invention include, but are not limited to, esters, carbonates, thiocarbonates, N-acyl derivatives, N-acyloxyalkyl derivatives, quaternary derivatives of tertiary amines, N-Mannich bases, Schiff bases, aminoacid conjugates, phosphate esters, metal salts and sulfonate esters.
- Suitable positions for derivatization of the compounds of the invention to create “prodrugs” include but are not limited, to, 2-amino substitution. Those of ordinary skill in the art have the knowledge and means to accomplish this without undue experimentation. Various forms of prodrugs are well known in the art. For examples of such prodrug derivatives, see, e.g.,
- Bundgaard H. “Design and Application of Prodrugs” in A Textbook of Drug Design and Development , Krosgaard-Larsen and H. Bundgaard, Ed., 1991, Chapter 5, p. 113-191; and
- prodrugs as employed herein includes, but is not limited to, the following groups and combinations of these groups:
- alkyl refers to an optionally substituted straight-chain, or optionally substituted branched-chain saturated hydrocarbon radical having from one to thirty carbons, more preferably one to twelve carbons.
- alkyl radicals include methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, tert-amyl, pentyl, hexyl, heptyl, octyl and the like.
- cycloalkyl embraces cyclic alkyl radicals which include monocyclic, bicyclic, tricyclic, and higher multicyclic alkyl radicals wherein each cyclic moiety has from three to eight carbon atoms.
- examples of cycloalkyl radicals include cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and the like.
- a “lower alkyl” is a shorter alkyl, e.g., one containing from one to six carbon atoms.
- alkenyl refers to an optionally substituted straight-chain, or optionally substituted branched-chain hydrocarbon radical having one or more carbon-carbon double-bonds and having from two to thirty carbon atoms, more preferably two to eighteen carbons.
- alkenyl radicals include ethenyl, propenyl, butenyl, 1,3-butadienyl and the like.
- cycloalkenyl refers to cyclic alkenyl radicals which include monocyclic, bicyclic, tricyclic, and higher multicyclic alkenyl radicals wherein each cyclic moiety has from three to eight carbon atoms.
- a “lower alkenyl” refers to an alkenyl having from two to six carbons.
- alkynyl refers to an optionally substituted straight-chain or optionally substituted branched-chain hydrocarbon radical having one or more carbon-carbon triple-bonds and having from two to thirty carbon atoms, more preferably from two to twelve carbon atoms, or from two to six carbon atoms, as well as those having from two to four carbon atoms.
- alkynyl radicals include ethynyl, 2-propynyl, 2-butynyl, 1,3-butadiynyl and the like.
- cycloalkynyl refers to cyclic alkynyl radicals which include monocyclic, bicyclic, tricyclic, and higher multicyclic alkynyl radicals wherein each cyclic moiety has from three to eight carbon atoms.
- a “lower alkynyl” refers to an alkynyl having from two to six carbons.
- heteroalkyl, heteroalkenyl and heteroalkynyl include optionally substituted alkyl, alkenyl and alkynyl structures, as described above, and which have one or more skeletal chain atoms selected from an atom other than carbon, e.g., oxygen, nitrogen, sulfur, phosphorous or combinations thereof.
- carbon chain embraces any alkyl, alkenyl, alkynyl, or heteroalkyl, heteroalkenyl, or heteroalkynyl group, which are linear, cyclic, or any combination thereof. If the chain is part of a linker and that linker comprises one or more rings as part of the core backbone, for purposes of calculating chain length, the “chain” only includes those carbon atoms that compose the bottom or top of a given ring and not both, and where the top and bottom of the ring(s) are not equivalent in length, the shorter distance shall be used in determining the chain length. If the chain contains heteroatoms as part of the backbone, those atoms are not calculated as part of the carbon chain length.
- membered ring can embrace any cyclic structure, including aromatic, heteroaromatic, alicyclic, heterocyclic and polycyclic fused ring systems as described below.
- membered is meant to denote the number of skeletal atoms that constitute the ring.
- pyridine, pyran, and pyrimidine are six-membered rings and pyrrole, tetrahydrofuran, and thiophene are five-membered rings.
- aryl refers to an optionally substituted aromatic hydrocarbon radical of six to twenty ring atoms, and includes mono-aromatic rings and fused aromatic rings.
- a fuised aromatic ring radical contains from two to four fused rings where the ring of attachment is an aromatic ring, and the other individual rings within the fused ring may be aromatic, heteroaromatic, alicyclic or heterocyclic.
- aryl includes mono-aromatic rings and fused aromatic rings containing from six to twelve carbon atoms, as well as those containing from six to ten carbon atoms.
- aryl groups include, without limitation, phenyl, naphthyl, anthryl, chrysenyl, and benzopyrenyl ring systems.
- lower aryl refers to an aryl having six to ten skeletal ring carbons, e.g., phenyl and naphthyl ring systems.
- heteroaryl refers to optionally substituted aromatic radicals containing from five to twenty skeletal ring atoms and where one or more of the ring atoms is a heteroatom such as, for example, oxygen, nitrogen, sulfur, selenium or phosphorus.
- heteroaryl includes optionally substituted mono-heteroaryl radicals and fused heteroaryl radicals having at least one heteroatom (e.g., quinoline, benzothiazole).
- a fused heteroaryl radical may contain from two to four fused rings where the ring of attachment is a heteroaromatic ring, the other individual rings within the fused ring system may be aromatic, heteroaromatic, alicyclic or heterocyclic.
- heteroaryl also includes mono-heteroaryls or fused heteroaryls having from five to twelve skeletal ring atoms, as well as those having from five to ten skeletal ring atoms.
- heteroaryls include, without limitation, furanyl, benzofuranyl, chromenyl, pyridyl, pyrrolyl, indolyl, quinolinyl, pyridyl-N-oxide, pyrimidyl, pyrazinyl, imidazolyl, pyrazolyl, oxazolyl, isoxazolyl, benzothiozole, benzimidazole, benzoxazoles, benzothiadiazole, benzoxadiazole, benzotriazole, quinolines, isoquinolines, indoles, purinyl, indolizinyl, thienyl and the like and their oxides.
- lower heteroaryl refers to a heteroaryl having five to ten skeletal ring atoms, e.g., pyridyl, thienyl, pyrimidyl, pyrazinyl, pyrrolyl, or furanyl.
- alicyclic alone or in combination, refers to an optionally substituted saturated or unsaturated nonaromatic hydrocarbon ring system containing from three to twenty ring atoms.
- the term alicyclic includes mono-alicyclic and fused alicyclic radicals.
- a fused alicyclic may contain from two to four fused rings where the ring of attachment is an alicyclic ring, and the other individual rings within the fused-alicyclic radical may be aromatic, heteroaromatic, alicyclic and heterocyclic.
- the term alicyclic also includes mono-alicyclic and fuised alicyclic radicals containing from three to twelve carbon atoms, as well as those containing from three to ten carbon atoms.
- alicyclics include, without limitation, cyclopropyl, cyclopropenyl, cyclobutyl, cyclopentyl, cyclodecyl, cyclododecyl, cyclopentadienyl, indanyl, and cyclooctatetraenyl ring systems.
- the term “lower alicyclic” refers to an alicyclic having three to ten skeletal ring carbons, e.g., cyclopropyl, cyclopropenyl, cyclobutyl, cyclopentyl, decalinyl, and cyclohexyl.
- heterocyclic refers to optionally substituted saturated or unsaturated nonaromatic ring radicals containing from five to twenty ring atoms where one or more of the ring atoms are heteroatoms such as, for example, oxygen, nitrogen, sulfur, and phosphorus.
- alicyclic includes mono-heterocyclic and fused heterocyclic ring radicals.
- a fused heterocyclic radical may contain from two to four fused rings where the attaching ring is a heterocyclic, and the other individual rings within the fused heterocyclic radical may be aromatic, heteroaromatic, alicyclic or heterocyclic.
- heterocyclic also includes mono-heterocyclic and fused alicyclic radicals having from five to twelve skeletal ring atoms, as well as those having from five to ten skeletal ring atoms.
- Example of heterocyclics include without limitation, tetrahydrofuranyl, benzodiazepinyl, tetrahydroindazolyl, dihyroquinolinyl, and the like.
- the term “lower heterocyclic” refers to a heterocyclic ring system having five to ten skeletal ring atoms, e.g., dihydropyranyl, pyrrolidinyl, dioxolanyl, piperidinyl, piperazinyl, and the like.
- alkylaryl or “araalkyl,” alone or in combination, refers to an aryl radical as defined above in which at least one H atom is replaced by an alkyl radical as defined above, such as, for example, tolyl, xylyl and the like.
- arylalkyl refers to an alkyl radical as defined above in which at least one H atom is replaced by an aryl radical as defined above, such as, for example, benzyl, 2-phenylethyl and the like.
- heteroarylalkyl refers to an alkyl radical as defined above in which at least one H atom is replaced by a heteroaryl radical as defined above, each of which may be optionally substituted.
- alkoxy refers to an alkyl ether radical, alkyl-O—, wherein the term alkyl is defined as above.
- alkoxy radicals include methoxy, ethoxy, n-propoxy, isopropoxy, n-butoxy, iso-butoxy, sec-butoxy, tert-butoxy and the like.
- aryloxy refers to an aryl ether radical wherein the term aryl is defined as above.
- aryloxy radicals include phenoxy, thienyloxy and the like.
- alkylthio refers to an alkyl thio radical, alkyl-S—, wherein the term alkyl is as defined above.
- arylthio refers to an aryl thio radical, aryl-S—, wherein the term aryl is as defined above.
- heteroarylthio refers to the group heteroaryl-S—, wherein the term heteroaryl is as defined above.
- acyl refers to a radical —C(O)R where R includes alkyl, alkenyl, alkynyl, aryl, heteroaryl, alicyclic, heterocyclic, arylalkyl or heteroarylalkyl wherein the alkyl, alkenyl, alkynyl, aryl, heteroaryl, alicyclic, heterocyclic, arylalkyl or heteroaryl alkyl groups may be optionally substituted.
- acyloxy refers to the ester group —OC(O)R, where R is H, alkyl, alkenyl, alkynyl, aryl, heteroaryl, alicyclic, heterocyclic, arylalkyl, or heteroarylalkyl wherein the alkyl, alkenyl, alkynyl, aryl, heteroaryl, alicyclic, heterocyclic, arylalkyl or heteroarylalkyl may be optionally substituted.
- carboxy esters refers to —C(O)OR where R is alkyl, aryl or arylalkyl, wherein the alkyl, aryl and arylalkyl groups may be optionally substituted.
- R and R′ are independently selected from the group consisting of H, alkyl, aryl, heteroaryl, alicyclic, heterocyclic, arylalkyl and heteroarylalkyl, wherein the alkyl, aryl, heteroaryl, alicyclic, heterocyclic, or arylalkyl groups may be optionally substituted.
- halogen includes F, Cl, Br and I.
- haloalkyl, haloalkenyl, haloalkynyl and haloalkoxy include alkyl, alkenyl, alkynyl and alkoxy structures, as described above, that are substituted with one or more fluorines, chlorines, bromines or iodines, or with combinations thereof.
- perhaloalkyl, perhaloalkyloxy and perhaloacyl refer to alkyl, alkyloxy and acyl radicals as described above, in which all the H atoms are replaced by fluorines, chlorines, bromines or iodines, or combinations thereof.
- cycloalkyl, arylalkyl, aryl, heteroaryl, alicyclic, heterocyclic, alkyl, alkynyl, alkenyl, haloalkyl, and heteroalkyl include optionally substituted cycloalkyl, arylalkyl, aryl, heteroaryl, alicyclic, heterocyclic, alkyl, alkynyl, alkenyl, haloalkyl and heteroalkyl groups.
- alkylamino refers to the group —NHR where R is alkyl.
- dialkylamino refers to the group —NRR′ where R and R′ are alkyls.
- sulfuride refers to a sulfur atom covalently linked to two atoms; the formal oxidation state of said sulfur is (II).
- thioether may be used interchangeably with the term “sulfide.”
- sulfoxide refers to a sulfur atom covalently linked to three atoms, at least one of which is an oxygen atom; the formal oxidation state of said sulfur atom is (IV).
- sulfurone refers to a sulfur atom covalently linked to four atoms, at least two of which are oxygen atoms; the formal oxidation state of said sulfur atom is (VI).
- aryl optionally mono- or di-substituted with an alkyl means that the alkyl may but need not be present, or either one alkyl or two may be present, and the description includes situations where the aryl is substituted with one or two alkyls and situations where the aryl is not substituted with an alkyl.
- “Optionally substituted” groups may be substituted or unsubstituted.
- the substituents of an “optionally substituted” group may include, without limitation, one or more substituents independently selected from the following groups or designated subsets thereof: lower alkyl, lower alkenyl, lower alkynyl, lower aryl, heteroaryl, alicyclic, heterocyclic, arylalkyl, heteroarylalkyl, lower alkoxy, lower aryloxy, amino, alkylamino, dialkylamino, diarylalkylamino, alkylthio, arylthio, heteroarylthio, oxo, oxa, acyl (—C(O)R), (—C(O)), carboxyesters (—C(O)OR), carboxamido (—C(O)NH 2 ), carboxy, acyloxy, —H, halo, —CN, —NO 2 , —N 3 , —SH, —
- An optionally substituted group may be unsubstituted (e.g., —CH 2 CH 3 ), fully substituted (e.g., —CF 2 CF 3 ), monosubstituted (e.g., —CH 2 CH 2 F) or substituted at a level anywhere in-between fully substituted and monosubstituted (e.g., —CH 2 CF 3 ).
- pyridine-1-oxy also means “pyridine-N-oxy.”
- Some of the compounds of the present invention may contain one or more chiral centers and therefore may exist in enantiomeric and diastereomeric forms.
- the scope of the present invention is intended to cover all isomers per se, as well as mixtures of cis and trans isomers, mixtures of diastereomers and racemic mixtures of enantiomers (optical isomers) as well. Further, it is possible using well known techniques to separate the various forms, and some embodiments of the invention may feature purified or enriched species of a given enantiomer or diastereomer.
- a “pharmacological composition” refers to a mixture of one or more of the compounds described herein, or pharmaceutically acceptable salts thereof, with other chemical components, such as pharmaceutically acceptable carriers and/or excipients.
- the purpose of a pharmacological composition is to facilitate administration of a compound to an organism.
- pharmaceutically acceptable carrier means a pharmaceutically-acceptable material, composition or vehicle, such as a liquid or solid filler, diluent, excipient, solvent or encapsulating material, involved in carrying or transporting the subject agent from one organ, or portion of the body, to another organ, or portion of the body.
- a pharmaceutically-acceptable material such as a liquid or solid filler, diluent, excipient, solvent or encapsulating material, involved in carrying or transporting the subject agent from one organ, or portion of the body, to another organ, or portion of the body.
- Each carrier must be “acceptable” in the sense of being compatible with the other ingredients of the formulation and not injurious to the patient.
- materials which can serve as pharmaceutically-acceptable carriers include: (1) sugars, such as lactose, glucose and sucrose; (2) starches, such as corn starch and potato starch; (3) cellulose, and its derivatives, such as sodium carboxymethyl cellulose, ethyl cellulose and cellulose acetate; (4) powdered tragacanth; (5) malt; (6) gelatin; (7) talc; (8) excipients, such as cocoa butter and suppository waxes; (9) oils, such as peanut oil, cottonseed oil, safflower oil, sesame oil, olive oil, corn oil and soybean oil; (10) glycols, such as propylene glycol; (11) polyols, such as glycerin, sorbitol, mannitol and polyethylene glycol; (12) esters, such as ethyl oleate and ethyl laurate; (13) agar; (14) buffering agents, such as magnesium hydroxide and aluminum hydrox
- excipient refers to an inert substance added to a pharmacological composition to further facilitate administration of a compound.
- excipients include but are not limited to calcium carbonate, calcium phosphate, various sugars and types of starch, cellulose derivatives, gelatin, vegetable oils and polyethylene glycols.
- a “pharmaceutically effective amount” means an amount which is capable of providing a therapeutic and/or prophylactic effect.
- the specific dose of compound administered according to this invention to obtain therapeutic and/or prophylactic effect will, of course, be determined by the particular circumstances surrounding the case, including, for example, the specific compound administered, the route of administration, the condition being treated, and the individual being treated.
- a typical daily dose (administered in single or divided doses) will contain a dosage level of from about 0.01 mg/kg to about 50-100 mg/kg of body weight of an active compound of the invention.
- Preferred daily doses generally will be from about 0.05 mg/kg to about 20 mg/kg and ideally from about 0.1 mg/kg to about 10 mg/kg.
- Factors such as clearance rate, half-life and maximum tolerated dose (MTD) have yet to be determined but one of ordinary skill in the art can determine these using standard procedures.
- the preferred therapeutic effect is the inhibition, to some extent, of the growth of cells characteristic of a proliferative disorder, e.g., breast cancer.
- a therapeutic effect will also normally, but need not, relieve to some extent one or more of the symptoms other than cell growth or size of cell mass.
- a therapeutic effect may include, for example, one or more of 1) a reduction in the number of cells; 2) a reduction in cell size; 3) inhibition (i.e., slowing to some extent, preferably stopping) of cell infiltration into peripheral organs, e.g., in the instance of cancer metastasis; 3) inhibition (i.e., slowing to some extent, preferably stopping) of tumor metastasis; 4) inhibition, to some extent, of cell growth; and/or 5) relieving to some extent one or more of the symptoms associated with the disorder.
- IC 50 refers to an amount, concentration or dosage of a particular test compound that achieves a 50% inhibition of a maximal response in an assay that measures such response.
- the “IC 50 ” value of a compound of the invention can be greater for normal cells than for cells exhibiting a proliferative disorder, e.g., breast cancer cells. The value depends on the assay used.
- a “standard” is meant a positive or negative control.
- a negative control in the context of HER2 expression levels is, e.g., a sample possessing an amount of HER2 protein that correlates with a normal cell.
- a negative control may also include a sample that contains no HER2 protein.
- a positive control does contain HER2 protein, preferably of an amount that correlates with overexpression as found in proliferative disorders, e.g., breast cancers.
- the controls may be from cell or tissue samples, or else contain purified ligand (or absent ligand), immobilized or otherwise.
- one or more of the controls may be in the form of a diagnostic “dipstick.”By “selectively targeting” is meant affecting one type of cell to a greater extent than another, e.g., in the case of cells with high as opposed to relatively low or normal HER2 levels.
- One embodiment of the compounds of the invention is of Formula A: or a polymorph, solvate, ester, tautomer, diastereomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, wherein:
- R 5 is alkyl, aryl, heteroaryl, alicyclic, or heterocyclic, each of which is optionally bi- or tricyclic, and optionally substituted with H, halogen, lower alkyl, lower alkenyl, lower alkynyl, lower aryl, lower alicyclic, araalkyl, aryloxyalkyl, alkoxyalkyl, perhaloalkyl, perhaloalkyloxy, perhaloacyl, —N 3 , —SR 8 , —OR 8 , —CN, —CO 2 R 9 , —NO 2 , or —NR 8 R 10 ;
- the compound is not one found or described in one or more of JP 10025294; U.S. Pat. No. 4,748,177; U.S. Pat. No. 4,748,177; U.S. Pat. No. 6,369,092; WO 00/06573; WO 02/055521; WO 02/055082; WO 02/055083 ; Eur. J. Med. Chem ., 1994, 29(1), 3-9; and J. Het. Chem . 1990, 27(5), 1409;
- -R 4 R 5 is not a ribose or derivative thereof, or a sugar or derivative thereof;
- -R 4 R 5 is not a phosphonate or phosphonic acid, or a group substituted with a phosphonate or phosphonic acid
- X 1 and X 2 are the same or different and each is nitrogen or —CR 6 ;
- R 1 is halogen, —OR 8 , —SR 8 , or lower alkyl;
- R 2 is —NR 8 R 10 ;
- R3 is hydrogen, —OH or keto tautomer, —OR 8 , halogen, —CN, lower alkyl, or —C(O)R 9 ;
- R 5 is alkyl, aromatic, heteroaromatic, alicyclic, heterocyclic, each of which is optionally bi- or tricyclic, and optionally substituted with H, halogen, lower alkyl, —SR 8 , —OR 8 , —CN, —CO 2 R
- R 1 is selected from halogen, hydroxyl, lower alkoxy, lower thioalkyl and C 1-4 alkyl; and R 2 is —NH 2 .
- R 1 is selected from halogen, hydroxyl, lower alkoxy, lower thioalkyl or C 1-4 alkyl; optionally wherein R 2 is NH 2 .
- R 1 is halogen, hydroxyl, lower alkoxy, lower thioalkyl, or C 1-4 alkyl; and R 2 is optionally NH 2 , R 4 is —(CH 2 )—, and R 5 is phenyl, benzyl, or pyridyl, all optionally substituted with H, halogen, lower alkyl, —SR 8 , —OR 8 (or cyclic ethers such as methylenedioxy), —CN, —C0 2 R 9 , —NO 2 , or —NR 8 R 10 ; R 8 is hydrogen, lower alkyl, lower aryl or —(CO)R 9 ; R 9 is lower alkyl, lower aryl, lower heteroaryl, —NR 8 R 10 or —OR 11 ; R 11 is lower alkyl or lower aryl; and R 10 is hydrogen or lower alkyl.
- R 1 is halogen
- R 2 is —NH 2
- R 4 is —CH 2 —
- R 6 is H or halogen
- R 5 is phenyl optionally substituted with H, halogen, C 1-4 alkyl, C 1-4 alkoxy, C 1-4 alkylthio, perhaloalkyl, perhaloalkyloxy, —CN, —NO 2 , —NH 2 or —CO 2 R 11 .
- R 1 is halogen
- R 2 is —NH 2
- R 4 is —CH 2 —
- R 6 is H
- R 5 is 2-halo-3, 5-dimethoxyphenyl optionally substituted with H, halogen, C 1-4 alkyl, C 1-4 alkoxy, C 1-4 alkylthio, perhaloalkyl, perhaloalkyloxy, —CN, —NO 2 , —NH 2 , or —CO 2 R 11 at the para (4-) position.
- R 1 is chloro
- R 2 is —NH 2
- R 4 is —CH 2 —
- R 6 is H
- R 5 is 2-chloro-3, 4,5-trimethoxyphenyl.
- R 1 is chloro
- R 2 is —NH 2
- R 4 is —CH 2 —
- R 6 is H
- R 5 is 2-bromo-3, 4,5-trimethoxyphenyl.
- R 5 is selected from 2-iodo-3,4,5-trimethoxyphenyl, 2-fluoro-3,4,5-trimethoxyphenyl, and 2-bromo-3,4,5-trimethoxyphenyl.
- the invention provides compounds of Formula A1: or a tautomer, pharmaceutically acceptable salt, or prodrug thereof, wherein:
- R 1 is halogen, hydroxyl, lower alkoxy, lower thioalkyl, or C 1-4 alkyl; and R 2 is NH 2 .
- R 1 is halogen;
- R 2 is NH 2 ,
- R 4 is —CH 2 —.
- the invention provides compounds of Formula A2. or a tautomer, pharmaceutically acceptable salt, or prodrug thereof, wherein:
- X 1 and X 2 are the same or different and each is nitrogen or —CR 6 ;
- R 1 is halogen, —OR 8 , —SR 8 or lower alkyl
- R 2 is —NR 8 R 10 ;
- R 3 is hydrogen, OH or a keto tautomer, —OR 8 , halogen, —CN, lower alkyl or —C(O)R 9 ;
- R 6 is hydrogen, halogen, lower alkyl, —SR 8 , —OR 8 , —NR 8 R 10 , —N 3 , or —C(O)R 9 ;
- R 5 is alkyl, aromatic, heteroaromatic, alicyclic, heterocyclic, all optionally bi- or tricyclic, and all optionally substituted with H, halogen, lower alkyl, —SR 8 , —OR 8 , —CN, —CO 2 R 9 , —NO 2 , or —NR 8 R 10 ;
- R 8 is hydrogen, lower alkyl, lower aryl, or —(CO)R 9 ;
- R 9 is lower alkyl, lower aryl, lower heteroaryl, —NR 8 R 10 or —OR 11 ;
- R 11 is lower alkyl or lower aryl
- R 10 is hydrogen or lower alkyl.
- R 1 is halogen, hydroxyl, lower alkoxy, lower thioalkyl or C 1-4 alkyl; and wherein R 2 is NH 2 .
- R 4 is —(CH 2 )—.
- R 1 is halogen, hydroxyl, lower alkoxy, lower thioalkyl or C 1-4 alkyl;
- R 2 is NH 2 ,
- R 4 is —(CH 2 )—.
- Another embodiment of the invention is compounds of Formula I: or a polymorph, solvate, ester, tautomer, diastereomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, wherein:
- each of the aryl, heteroaryl, alicyclic or heterocyclic group is monocyclic or bicyclic.
- R 0 is hydrogen, halogen, —SH, —OH, or —CN; R 1 is halogen; and R 2 is —NHR 8 , where R 8 is hydrogen or —C(O)R 9 .
- R 1 is chloro or bromo
- R 2 is —NHR 8 , where R 8 is hydrogen or —C(O)R 9
- R 3 is hydrogen, halogen, OR 8 , SR 8 , NR 8 R 10 , lower alkyl, lower alkenyl, lower alkynyl, lower perhaloalkyl, lower aryl, or lower heteroaryl.
- R 0 is hydrogen, halogen or —CN;
- R 2 is —NHR 8 , where R 8 is hydrogen or —C(O)R 9 ; and
- R 4 is —CH 2 —.
- R 0 is hydrogen, halogen, —SH, —OH or —CN;
- R 1 is halogen;
- R 2 is —NH 2 ,
- R 3 is hydrogen, halogen, —OR 8 , —SR 8 , —NR 8 R 10 , lower alkyl, lower alkenyl, lower alkynyl, perhaloalkyl, lower aryl, or lower heteroaryl, wherein R 8 is hydrogen, lower alkyl, lower aryl, or —C(O)R 9 ;
- R 4 is —CH 2 —;
- R 5 is aryl or heteroaryl, wherein each of said aryl and heteroaryl is monocyclic or bicyclic and is substituted with 3 to 5 substituents.
- R 1 is chloro or bromo
- R 2 is —NH 2
- R 5 is a phenyl having at least three substituents.
- R 1 is chloro or bromo
- R 2 is —NH 2
- R 5 is a pyridyl having at least two substituents.
- R 1 is chloro or bromo
- R 2 is —NH 2
- R 5 is 1-oxy-pyridyl (N-oxy-pyridyl) having at least two substituents.
- Another embodiment of the invention is a compound of Formula II: or a polymorph, solvate, ester, tautomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, wherein:
- R 1 is halogen or lower alkyl
- R 4 is —CHR 12 —
- R 5 is aryl or heteroaryl, wherein each of said aryl and heteroaryl is monocyclic or bicyclic and is substituted with 3 to 5 substituents.
- R 0 is hydrogen or —NHR 8
- R 1 is halogen, —OR 11 , —SR 11 or lower alkyl
- R 10 is hydrogen or lower alkyl.
- R 0 is hydrogen;
- R 1 is halogen;
- R 4 is —CH 2 —; and
- R 5 is aryl or heteroaryl, wherein each of said aryl and heteroaryl is monocyclic or bicyclic and is substituted with 3 to 5 substituents; and
- R 10 is hydrogen.
- R 1 is chloro or bromo
- R 5 is phenyl, pyridyl or 1-oxy-pyridyl (N-oxy-pyridyl) each of which has at least two substituents.
- Another embodiment of the invention is a compound represented by Formula III: or a polymorph, solvate, ester, tautomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, wherein:
- R 1 is halogen
- R 3 is hydrogen, halogen, —OR 8 , —SR 8 , —NR 8 R 10 , lower alkyl, lower alkenyl, lower alkynyl, lower perhaloalkyl, lower aryl, or lower heteroaryl, wherein R 8 is hydrogen, lower alkyl, lower alkenyl, or lower alkynyl, lower aryl, lower heteroaryl, or —C(O)R 9
- R 4 is —CH 2 —
- R 5 is aryl or heteroaryl, wherein each of said aryl and heteroaryl is monocyclic or bicyclic and is substituted with 3 to 5 substituents
- R 10 is hydrogen or lower alkyl.
- R 1 is halogen;
- R 4 is —CH 2 —;
- R 5 is aryl or heteroaryl, wherein each of said aryl and heteroaryl is monocyclic or bicyclic and is substituted with 3 to 5 substituents; and
- R 10 is hydrogen.
- R 1 is halogen;
- R 3 is hydrogen;
- R 4 is —CH 2 —;
- R 5 is aryl or heteroaryl, wherein each of said aryl and heteroaryl is monocyclic or bicyclic and is substituted with 3 to 5 substituents; and
- R 10 is hydrogen.
- Another embodiment of the invention is compounds represented by Formula IV: or a polymorph, solvate, ester, tautomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, wherein:
- R 1 is halogen;
- R 4 is —CH 2 —;
- R 5 is aryl or heteroaryl, wherein each of said aryl and heteroaryl is monocyclic or bicyclic and is substituted with 3 to 5 substituents.
- R 1 is chloro or bromo
- R 5 is phenyl, pyridyl or 1-oxy-pyridyl (N-oxy-pyridyl), each of which has at least two substituents.
- the compounds of Formula I of the present invention may be synthesized by various methods known in the art.
- the general strategy is outlined in Scheme 1 and consists of three parts: (1) constructing the bicyclic system, starting from either a pyridine or a pyrrole, or an acyclic precursor (2) appending the R 5 -R 4 -group, and (3) further elaborating the ring systems.
- sequence of events is not necessarily (1)-(2)-(3), and that these events may be interchanged, provided there be no incompatibility between the reagents and the functional groups specific to the case in point.
- the compounds of Formula 4 can be prepared from pyrimidines as outlined in Scheme 2. For instance:
- the compounds of Formula 4 can be made by intramolecular cyclization of an aldehyde or ketone, possibly protected, as in Formula 6.
- an aldehyde or ketone possibly protected, as in Formula 6.
- the compounds of Formula 4, wherein R 3 is H, R 6 is Cl, and R 7 is NH 2 can be prepared by treating compounds of Formula 7 wherein R is a halogen or a leaving group with ammonia.
- compounds of Formula I wherein R 3 is H, R 1 is Cl, R 2 is NH 2 can be prepared by treating the compound of Formula 7 wherein R is a halogen or leaving group with R 5 -R 4 —NH 2 in butanol at reflux in presence of a base such as K 2 CO 3 , Cs 2 CO 3 or iPrNEt 2 .
- a base such as K 2 CO 3 , Cs 2 CO 3 or iPrNEt 2 .
- the compounds of Formula 4 can be obtained by treatment of a ⁇ -haloketone of Formula 8 wherein X is a halogen with ammonia or a synthetic equivalent thereof.
- the compounds of Formula 4 wherein R 0 is methyl can be obtained by a tandem Pd-mediated intramolecular cyclization/double-bond migration of alkenes of Formula 9 (S. E. Watson, Synth. Commun . 1998, 28, 3885).
- the compounds of Formula 4 wherein R 3 is H can be obtained by Pd-mediated intramolecular cyclization of alkynes of Formula 10, wherein Z in as electron-withdrawing group such as, e.g., tosyl-, or —CO 2 Et.
- the compounds of Formula 4 wherein R 3 is AcO— can be obtained by intramolecular Friedel-Crafts acylation of precursors of Formula 11 (E. D. Edstrom, J. Org. Chem . 1993, 58, 403).
- the compound of Formula 4 wherein R 0 is H, R 6 is OH, and R 7 is NH 2 , can be prepared by treating the compound of Formula 12 with an ⁇ -haloaldehyde of the formula R 3 —CHX-CHO. See, D. M. Williams, J. Chem. Soc., Perkin Trans 1, 1997, 1171; C. J. Barnett, Org. Proc. Res. Devop . 1999, 3, 184; A. Gangjee, J. Med. Chem . 2001, 44, 1993.
- the compounds of Formula 4, wherein R 6 is OH and R 7 is NH 2 can be obtained by treating the compound of Formula 13 with an aldehyde of the formula R 3 —CHO. See, A. Gangjee, J. Med. Chem . 2003, 46, 591; E. C. Taylor, Heterocycles 1996, 43, 323.
- the compounds of Formula 4 can also be made from pyrroles of Formula 2.
- pyrroles of Formula 2 There is a variety of methods by which the 6-membered ring can be formed (e.g. R. J. Bontems, J. Med. Chem , 1990, 33, 2174 and references therein). For instance:
- the compounds of Formula 14 can be prepared from an acyclic precursor as outlined in Scheme 4 (T. Miwa, J. Med. Chem . 1991, 34,555). 2. Incorporation of the -R 4 -R 5 fragment.
- Compounds of Formula 4 can be alkylated in the presence of a base such as K 2 CO 3 , NaH, Cs 2 CO 3 , DBU etc. with/without the presence of a catalyst such as NaI, KI, (Bu) 4 NI etc., and in a polar solvent such as DMF, THF, DMSO etc. using electrophiles such as L 1 -R-R 5 where L 1 is a leaving group.
- Leaving groups include but are not limited to, e.g., halogen, triflate, tosylate, mesylate, triphenylphosphonium (generated under Mitsunobu conditions, e.g. PPh 3 /DEAD) etc. See Kasibhatla, PCT publication number WO 03/037860.
- the electrophiles can be prepared from the substituted benzene derivatives using various methods reported in the literature, see Jerry March, Advanced Organic Chemistry , 4 th edition; Larock, Comprehensive Organic Transformations , 1989, VCH, New York.
- L 1 is Br
- benzyl derivatives can also be prepared by benzylic oxidation or benzylic halogenation. Further modification of the benzyl ring can be done before or after the pyrrolo[2,3-d]pyrimidine alkylation step.
- the -R 4 -R 5 group can be appended before the bicyclic pyrrolo[2,3-d]pyrimidine bicyclic ring is constructed, and this is further detailed below (paragraph 4, schemes 8 and 9).
- the -R 4 -R 5 group can be appended by an aromatic nucleophilic substitution using NH 2 —R 4 -R 5 .
- the compound NH 2 —R 4 —R 5 is obtained by treating L 1 -R 4 -R 5 with ammonia at temperatures of 20-160° C. in a pressure vessel.
- L 1 is —NH 2
- L 1 is leaving group such as chloride, bromide, tosylate, mesylate etc. using ammonia, or with sodium azide followed by hydrogenation.
- R 0 is H
- electrophiles e.g., I 2 , ArCHO
- R 0 is, e.g. —I or —CH(OH)Ar.
- HMPT/CCl 4 Perkin Trans 1, 1994, 923
- HMPT/NBS E. A. Veliz, Tetrahedron Lett , 2000, 41, 1695
- PPh 3 /I 2 X. Lin, Org. Letters , 2000, 2, 3497
- Compounds of Formula I, wherein R 1 is NH 2 can be converted to halides by a Balz-Schiemann (F) or Sandmeyer reaction (Cl, Br, I) by means of a nitrosylating agent (NaNO 2 /H + , NOBF 4 , RONO) and a halogen donor (BF 4 ⁇ , CuX 2 , SbX 3 ).
- F Balz-Schiemann
- Sandmeyer reaction Cl, Br, I
- a nitrosylating agent NaNO 2 /H + , NOBF 4 , RONO
- a halogen donor BF 4 ⁇ , CuX 2 , SbX 3
- R 1 is alkyl
- R 1 is halogen and trialkyl aluminum or dialkyl zinc (A. Holy, J. Med. Chem . 1999, 42, 2064).
- R 1 is a halide
- R 1 is —NH 2 , —OH, —SH, —OR 8 , —SR 8 with standard reagents, e.g., NH 3 , NaOH, thiourea, R 8 O ⁇ , R 8 S ⁇ , with or without a catalyst (e.g. Pd, Ni, Cu, Lewis acid, H + ) (e.g., B. G. Ugarkar, J. Med. Chem . 2000, 43, 2883-2893 and 2894-2905).
- a catalyst e.g. Pd, Ni, Cu, Lewis acid, H +
- Compounds of Formula I, wherein R 2 is NH 2 can be temporarily protected, e.g. as an amide (Ac 2 O, PivCl), a carbamate (tBoc) 2 O) or amidine (DMF-DMA).
- Compounds of Formula I, wherein R 2 is NH 2 can be converted to halides by a Balz-Schiemann (F) or Sandmeyer reaction (Cl, Br, I) by means of a nitrosylating agent (NaNO 2 /H + , NOBF 4 , RONO) and a halogen donor (BF4 ⁇ , CuX 2 , SbX 3 ).
- F Balz-Schiemann
- Sandmeyer reaction Cl, Br, I
- a nitrosylating agent NaNO 2 /H + , NOBF 4 , RONO
- a halogen donor BF4 ⁇ , CuX 2 , SbX 3
- R 2 is a halide
- R 2 is NH 2 , OH, SH, OR 8 , SR 8 with standard reagents, e.g. NH 3 , NaOH, thiourea, R 8 O ⁇ , R 8 S ⁇ , with or without a catalyst (e.g. Pd, Ni, Cu, Lewis acid, H + ).
- a catalyst e.g. Pd, Ni, Cu, Lewis acid, H + .
- R 2 is a sulfide, e.g., MeS—
- a sulfone e.g MeSO 2 —
- a nucleophile e.g. NH 3 or NH 2 —NH 2 , N 3 —, CN—.
- Compounds of Formula I, wherein R 0 is OH and R 3 is H can be be monalkylated or bis-alkylated to give compounds of Formula III, wherein R 1 is an alkyl group.
- the alkylation can be effected in the presence of a base such as KHMDS, LHMDS, LDA etc. with/without the presence of a catalyst such as NaI, KI, (Bu) 4 NI etc., and in a polar solvent such as THF, DMSO etc. using electrophiles such as L 1 -R 3 where L 1 is a leaving group.
- Leaving groups include but are not limited to, e.g., halogen, triflate, tosylate or mesylate.
- Compounds of Formula I, wherein R 0 is H and R 3 is H can be oxidized to compounds of Formula 16/IV, with an oxidizing reagent such as ruthenium tetroxide in a binary solvent such as acetonitrile/water. (G. W. Gribble Org. Prep. Proced. Int . 2001, 33(6), 615).
- an oxidizing reagent such as ruthenium tetroxide in a binary solvent such as acetonitrile/water.
- Compounds of Formula I, wherein R 0 is OH and R 3 is H can be oxidized to compounds of Formula II, (wherein R 3 , R 3 is an oxo group) with an oxidizing reagent such as selenium dioxide or oxygen in presence of a cobalt (III) catalyst.
- an oxidizing reagent such as selenium dioxide or oxygen in presence of a cobalt (III) catalyst.
- R 5 especially when it is aryl or heteroaryl, can be further modified as needed, for example by halogenation, nitration, palladium coupling of halogen, Friedel-Crafts alkylation/acylation, etc. or these modifications can also be done before alkylation, see Jerry March, Advanced Organic Chemistry .
- the heteroaromatic rings can also be oxidized to their corresponding N-oxides using various oxidizing agents such as H 2 O 2 , O 3 , MCPBA etc. in polar solvents such as CH 2 Cl 2 , CHCl 3 , CF 3 COOH etc. See Jerry March, Advanced Organic Chemistry , 4th edition, Chapter 19. Examples of modifications are suggested in Scheme 6. 4. Permutations of the order of events
- the events (1) assembly of the bicyclic system (2) appendage of the R 5 -R 4 -moiety, and (3) further elaboration of the ring systems do not necessarily have to be made in the sequence (1)-(2)-(3), and it may be beneficial to proceed in a different sequence.
- Scheme 8 shows a synthesis in which the order of events is not (1)-(2)-(3), but is (2)-(1)-(3).
- First R 5 is appended via an aromatic nucleophilic substitution, then the bicyclic system is constructed, and finally it is elaborated.
- the compound of Formula 18, wherein R 1 is Cl and R 2 is NH 2 can be prepared by treating the compound of Formula 17 wherein R 2 ⁇ NH 2 , and R 1 ⁇ X ⁇ Cl, with R 5 -R 4 —NH 2 in butanol at reflux in presence of a base such as K 2 CO 3 , Cs 2 CO 3 or iPrNEt 2 . (A. B. Reitz J. Med. Chem . 1994, 37, 3561).
- an halogenating reagent such as bromine, N-bromosuccinimide, iodine or N-iodosuccinimide
- an acid such as acetic acid or p-toluenesulfonic acid.
- the compound of Formula 20, wherein R 1 is Cl and R 2 is NH 2 can be prepared by coupling the compound of Formula 19 with trimethylsilylacetylene under Sonogashira conditions followed by hydroboration using dicylohexylborane and oxidation using hydrogen peroxide in presence of sodium hydroxide.
- Sonogashira coupling E. C. Taylor Tetrahedron , 1992, 48, 8089.
- Hydroboration/oxidation G. Zweifel J. Am. Chem. Soc . 1976, 98, 3184).
- the compound of Formula 21, wherein R 1 is Cl and R 2 is NH 2 can be prepared by heating the compound of Formula 20 in a polar aprotic solvent such as THF, DME or dioxane in presence of oxalyl chloride, thionyl chloride, mesyl chloride or alkyl chloroformate and a base such as iPrNEt 2 or pyridine. It can also be prepared by treating the compound of Formula 20 with coupling reagents such DCC/HOBt, DCC/DMAP or EDCI/HOBt. (R. C. Larock Comprehensive Organic Transformations , Second Edition, p. 1870).
- Scheme 10 shows a putative synthesis in which the order of events is not (1)-(2)-(3), but is (1)-(3)-(2)-(3).
- the bicyclic ring is constructed, then it is elaborated, then the R 4 -R 5 moiety is appended, and finally the bicyclic ring system is further elaborated (deprotection).
- R 5 is for instance a pyridine, it can be converted to a N-oxide either before or after alkylation.
- the present invention is directed to the clinical use of the heterocyclics, in particular, the pyrazolopyrimidines and their related analogs of Formulae A, I, II, III and IV, and their polymorphs, solvates, esters, tautomers, diastereomers, enantiomers, pharmaceutically acceptable salts and prodrugs thereof, for use in treatment or prevention of diseases that are HSP90-dependent.
- diseases include disorders such as inflammatory diseases, infections, autoimmune disorders, stroke, ischemia, cardiac disorder, neurological disorders, fibrogenetic disorders, proliferative disorders, tumors, leukemias, neoplasms, cancers, carcinomas, metabolic diseases, and malignant disease.
- the fibrogenetic disorders include but are not limited to scleroderma, polymyositis, systemic lupus, rheumatoid arthritis, liver cirrhosis, keloid formation, interstitial nephritis and pulmonary fibrosis.
- the present invention features pharmaceutical compositions comprising the compound of Formulae A, I, II, III and IV, or a polymorph, solvate, ester, tautomer, enantiomer, diastereomer, pharmaceutically acceptable salt thereof, or prodrug thereof, of any of the preceding aspects and embodiments and one or more pharmaceutical excipients.
- the compounds utilized in the methods of the instant invention may be administered either alone or in combination with pharmaceutically acceptable carriers, excipients or diluents, in a pharmaceutical composition, according to standard pharmaceutical practice.
- the compounds can be administered orally or parenterally, including the intravenous, intramuscular, intraperitoneal, subcutaneous, rectal and topical routes of administration.
- the therapeutic or pharmaceutical compositions of the invention can be administered locally to the area in need of treatment.
- This may be achieved by, for example, but not limited to, local infuision during surgery, topical application, e.g., cream, ointment, injection, catheter, or implant, said implant made, e.g., out of a porous, non-porous, or gelatinous material, including membranes, such as sialastic membranes, or fibers.
- the administration can also be by direct injection at the site (or former site) of a tumor or neoplastic or pre-neoplastic tissue.
- the compounds or compositions of the invention can be delivered in a vesicle, e.g., a liposome (see, for example, Langer, Science 1990, 249,1527-1533; Treat et al., Liposomes in the Therapy of Infectious Disease and Cancer , Lopez-Bernstein and Fidler, Ed., Liss, N.Y., pp. 353-365, 1989).
- a liposome see, for example, Langer, Science 1990, 249,1527-1533; Treat et al., Liposomes in the Therapy of Infectious Disease and Cancer , Lopez-Bernstein and Fidler, Ed., Liss, N.Y., pp. 353-365, 1989).
- the compounds and pharmaceutical compositions used in the methods of the present invention can also be delivered in a controlled release system.
- a pump may be used (see, Sefton, 1987, CRC Crit. Ref. Biomed. Eng. 14:201; Buchwald et al. Surgery, 1980 88, 507; Saudek et al. N. Engl. J. Med. 1989, 321, (574).
- a controlled release system can be placed in proximity of the therapeutic target. (See, Goodson, Medical Applications of Controlled Release , 1984, Vol. 2, pp. 115-138).
- compositions used in the methods of the instant invention can also contain the active ingredient in a form suitable for oral use, for example, as tablets, troches, lozenges, aqueous or oily suspensions, dispersible powders or granules, emulsions, hard or soft capsules, or syrups or elixirs.
- Compositions intended for oral use may be prepared according to any method known in the art for the manufacture of pharmaceutical compositions, and such compositions may contain one or more agents selected from the group consisting of sweetening agents, flavoring agents, coloring agents and preserving agents in order to provide pharmaceutically elegant and palatable preparations. Tablets contain the active ingredient in admixture with non-toxic pharmaceutically acceptable excipients which are suitable for the manufacture of tablets.
- excipients may be, for example, inert diluents, such as calcium carbonate, sodium carbonate, lactose, calcium phosphate or sodium phosphate; granulating and disintegrating agents, such as microcrystalline cellulose, sodium crosscarmellose, corn starch, or alginic acid; binding agents, for example starch, gelatin, polyvinyl-pyrrolidone or acacia, and lubricating agents, for example, magnesium stearate, stearic acid or talc.
- the tablets may be un-coated or coated by known techniques to mask the taste of the drug or delay disintegration and absorption in the gastrointestinal tract and thereby provide a sustained action over a longer period.
- a water soluble taste masking material such as hydroxypropylmethyl-cellulose or hydroxypropylcellulose, or a time delay material such as ethyl cellulose, or cellulose acetate butyrate may be employed as appropriate.
- Formulations for oral use may also be presented as hard gelatin capsules wherein the active ingredient is mixed with an inert solid diluent, for example, calcium carbonate, calcium phosphate or kaolin, or as soft gelatin capsules wherein the active ingredient is mixed with water soluble carrier such as polyethyleneglycol or an oil medium, for example peanut oil, liquid paraffin, or olive oil.
- an inert solid diluent for example, calcium carbonate, calcium phosphate or kaolin
- water soluble carrier such as polyethyleneglycol or an oil medium, for example peanut oil, liquid paraffin, or olive oil.
- Aqueous suspensions contain the active material in admixture with excipients suitable for the manufacture of aqueous suspensions.
- excipients are suspending agents, for example sodium carboxymethylcellulose, methylcellulose, hydroxypropylmethyl-cellulose, sodium alginate, polyvinyl-pyrrolidone, gum tragacanth and gum acacia; dispersing or wetting agents may be a naturally-occurring phosphatide, for example lecithin, or condensation products of an alkylene oxide with fatty acids, for example polyoxyethylene stearate, or condensation products of ethylene oxide with long chain aliphatic alcohols, for example heptadecaethylene-oxycetanol, or condensation products of ethylene oxide with partial esters derived from fatty acids and a hexitol such as polyoxyethylene sorbitol monooleate, or condensation products of ethylene oxide with partial esters derived from fatty acids and hexitol anhydrides, for example polyethylene sorbitan mono
- the aqueous suspensions may also contain one or more preservatives, for example ethyl, or n-propyl p-hydroxybenzoate, one or more coloring agents, one or more flavoring agents, and one or more sweetening agents, such as sucrose, saccharin or aspartame.
- preservatives for example ethyl, or n-propyl p-hydroxybenzoate
- coloring agents for example ethyl, or n-propyl p-hydroxybenzoate
- flavoring agents such as sucrose, saccharin or aspartame.
- sweetening agents such as sucrose, saccharin or aspartame.
- Oily suspensions may be formulated by suspending the active ingredient in a vegetable oil, for example arachis oil, olive oil, sesame oil or coconut oil, or in mineral oil such as liquid paraffin.
- the oily suspensions may contain a thickening agent, for example beeswax, hard paraffin or cetyl alcohol.
- Sweetening agents such as those set forth above, and flavoring agents may be added to provide a palatable oral preparation.
- These compositions may be preserved by the addition of an anti-oxidant such as butylated hydroxyanisol or alpha-tocopherol.
- Dispersible powders and granules suitable for preparation of an aqueous suspension by the addition of water provide the active ingredient in admixture with a dispersing or wetting agent, suspending agent and one or more preservatives.
- Suitable dispersing or wetting agents and suspending agents are exemplified by those already mentioned above. Additional excipients, for example sweetening, flavoring and coloring agents, may also be present. These compositions may be preserved by the addition of an anti-oxidant such as ascorbic acid.
- the compounds and pharmaceutical compositions used in the methods of the instant invention may also be in the form of an oil-in-water emulsion.
- the oily phase may be a vegetable oil, for example olive oil or arachis oil, or a mineral oil, for example liquid paraffin, or mixtures of these.
- Suitable emulsifying agents may be naturally-occurring phosphatides, for example soybean lecithin, and esters or partial esters derived from fatty acids and hexitol anhydrides, for example sorbitan monooleate, and condensation products of the said partial esters with ethylene oxide, for example polyoxyethylene sorbitan monooleate.
- the emulsions may also contain sweetening agents, flavoring agents, preservatives and antioxidants.
- Syrups and elixirs may be formulated with sweetening agents, for example glycerol, propylene glycol, sorbitol or sucrose. Such formulations may also contain a demulcent, a preservative, flavoring and coloring agents and antioxidant.
- sweetening agents for example glycerol, propylene glycol, sorbitol or sucrose.
- Such formulations may also contain a demulcent, a preservative, flavoring and coloring agents and antioxidant.
- compositions may be in the form of a sterile injectable aqueous solution.
- acceptable vehicles and solvents that may be employed are water, Ringer's solution and isotonic sodium chloride solution.
- the sterile injectable preparation may also be a sterile injectable oil-in-water microemulsion where the active ingredient is dissolved in the oily phase.
- the active ingredient may be first dissolved in a mixture of soybean oil and lecithin.
- the oil solution may then be introduced into a water and glycerol mixture and processed to form a microemulsion.
- the injectable solutions or microemulsions may be introduced into a patient's blood-stream by local bolus injection.
- a continuous intravenous delivery device may be utilized.
- An example of such a device is the Deltec CADD-PLUSTM model 5400 intravenous pump.
- the pharmaceutical compositions may be in the form of a sterile injectable aqueous or oleagenous suspension for intramuscular and subcutaneous administration.
- This suspension may be formulated according to the known art using those suitable dispersing or wetting agents and suspending agents which have been mentioned above.
- the sterile injectable preparation may also be a sterile injectable solution or suspension in a non-toxic parenterally-acceptable diluent or solvent, for example as a solution in 1,3-butanediol.
- sterile, fixed oils are conventionally employed as a solvent or suspending medium.
- any bland fixed oil may be employed including synthetic mono- or diglycerides.
- fatty acids such as oleic acid find use in the preparation of injectables.
- compositions can be prepared by mixing the inhibitors with a suitable non-irritating excipient which is solid at ordinary temperatures but liquid at the rectal temperature and will therefore melt in the rectum to release the drug.
- suitable non-irritating excipient include cocoa butter, glycerinated gelatin, hydrogenated vegetable oils, mixtures of polyethylene glycols of various molecular weights and fatty acid esters of polyethylene glycol.
- creams, ointments, jellies, solutions or suspensions, etc., containing a compound or composition of the invention can be used.
- topical application can include mouth washes and gargles.
- the compounds used in the methods of the present invention can be administered in intranasal form via topical use of suitable intranasal vehicles and delivery devices, or via transdermal routes, using those forms of transdermal skin patches well known to those of ordinary skill in the art.
- the dosage administration will, of course, be continuous rather than intermittent throughout the dosage regimen.
- the methods, compounds and compositions of the instant invention may also be used in conjunction with other well known therapeutic agents that are selected for their particular usefulness against the condition that is being treated.
- the instant compounds may be useful in combination with known anti-cancer and cytotoxic agents.
- the instant methods and compounds may also be usefuil in combination with other inhibitors of parts of the signaling pathway that links cell surface growth factor receptors to nuclear signals initiating cellular proliferation.
- the methods of the present invention may also be useful with other agents that inhibit angiogenesis and thereby inhibit the growth and invasiveness of tumor cells, including, but not limited to VEGF receptor inhibitors, including ribozymes and antisense targeted to VEGF receptors, angiostatin and endostatin.
- VEGF receptor inhibitors including ribozymes and antisense targeted to VEGF receptors, angiostatin and endostatin.
- antineoplastic agents examples include, in general, and as appropriate, alkylating agents, anti-metabolites, epidophyllotoxins, an antineoplastic enzyme, a topoisomerase inhibitor, procarbazine, mitoxantrone, platinum coordination complexes, biological response modifiers and growth inhibitors, hormonal/anti-hormonal therapeutic agents and haematopoietic growth factors.
- alkylating agents include, in general, and as appropriate, alkylating agents, anti-metabolites, epidophyllotoxins, an antineoplastic enzyme, a topoisomerase inhibitor, procarbazine, mitoxantrone, platinum coordination complexes, biological response modifiers and growth inhibitors, hormonal/anti-hormonal therapeutic agents and haematopoietic growth factors.
- alkylating agents examples include, in general, and as appropriate, alkylating agents, anti-metabolites, epidophyllotoxins, an antineoplastic enzyme, a topoisomerase inhibitor, procarbazine
- Particularly usefuil members of those classes include, for example, carminomycin, daunorubicin, aminopterin, methotrexate, methopterin, dichloromethotrexate, mitomycin C, porfiromycin, 5-fluorouracil, 6-mercaptopurine, gemcitabine, cytosine arabinoside, podophyllotoxin or podo-phyllotoxin derivatives such as etoposide, etoposide phosphate or teniposide, melphalan, vinblastine, vincristine, leurosidine, vindesine, leurosine, paclitaxel and the like.
- antineoplastic agents include estramustine, carboplatin, cyclophosphamide, bleomycin, gemcitibine, ifosamide, melphalan, hexamethyl melamine, thiotepa, cytarabin, idatrexate, trimetrexate, dacarbazine, L-asparaginase, camptothecin, CPT-11, topotecan, ara-C, bicalutamide, flutamide, leuprolide, pyridobenzoindole derivatives, interferons and interleukins.
- the daily dosage will normally be determined by the prescribing physician with the dosage generally varying according to the age, weight, and response of the individual patient, as well as the severity of the patient's symptoms.
- a suitable amount of compound is administered to a mammal undergoing treatment for cancer, for example, breast cancer.
- Administration typically occurs in an amount of between about 0.01 mg/kg of body weight to about 100 mg/kg of body weight per day (administered in single or divided doses), more preferably at least about 0.1 mg/kg of body weight per day.
- a particular therapeutic dosage can include, e.g., from about 0.01 mg to about 1000 mg of compound, and preferably includes, e.g., from about 1 mg to about 1000 mg.
- the quantity of active compound in a unit dose of preparation may be varied or adjusted from about 0.1 mg to 1000 mg, preferably from about 1 mg to 300 mg, more preferably 10 mg to 200 mg, according to the particular application.
- the amount administered will vary depending on the particular IC 50 value of the compound used and the judgment of the attending clinician taking into consideration factors such as health, weight, and age. In combinational applications in which the compound is not the sole active ingredient, it may be possible to administer lesser amounts of compound and still have therapeutic or prophylactic effect.
- the pharmaceutical preparation is in unit dosage form.
- the preparation is subdivided into unit doses containing appropriate quantities of the active component, e.g., an effective amount to achieve the desired purpose.
- the actual dosage employed may be varied depending upon the requirements of the patient and the severity of the condition being treated. Determination of the proper dosage for a particular situation is within the skill of the art. Generally, treatment is initiated with smaller dosages which are less than the optimum dose of the compound. Thereafter, the dosage is increased by small amounts until the optimum effect under the circumstances is reached. For convenience, the total daily dosage may be divided and administered in portions during the day if desired.
- the amount and frequency of administration of the compounds and compositions of the present invention used in the methods of the present invention, and if applicable other chemotherapeutic agents and/or radiation therapy, will be regulated according to the judgment of the attending clinician (physician) considering such factors as age, condition and size of the patient as well as severity of the disease being treated.
- the chemotherapeutic agent and/or radiation therapy can be administered according to therapeutic protocols well known in the art. It will be apparent to those skilled in the art that the administration of the chemotherapeutic agent and/or radiation therapy can be varied depending on the disease being treated and the known effects of the chemotherapeutic agent and/or radiation therapy on that disease. Also, in accordance with the knowledge of the skilled clinician, the therapeutic protocols (e.g., dosage amounts and times of administration) can be varied in view of the observed effects of the administered therapeutic agents (i.e., antineoplastic agent or radiation) on the patient, and in view of the observed responses of the disease to the administered therapeutic agents.
- the administered therapeutic agents i.e., antineoplastic agent or radiation
- the compounds of the invention need not be administered in the same pharmaceutical composition as a chemotherapeutic agent, and may, because of different physical and chemical characteristics, be administered by a different route.
- the compounds/compositions may be administered orally to generate and maintain good blood levels thereof, while the chemotherapeutic agent may be administered intravenously.
- the determination of the mode of administration and the advisability of administration, where possible, in the same pharmaceutical composition, is well within the knowledge of the skilled clinician.
- the initial administration can be made according to established protocols known in the art, and then, based upon the observed effects, the dosage, modes of administration and times of administration can be modified by the skilled clinician.
- compositions of the invention may be administered concurrently (e.g., simultaneously, essentially simultaneously or within the same treatment protocol) or sequentially, depending upon the nature of the proliferative disease, the condition of the patient, and the actual choice of chemotherapeutic agent and/or radiation to be administered in conjunction (i.e., within a single treatment protocol) with the compound/composition.
- the compound/composition and the chemotherapeutic agent and/or radiation need not be administered simultaneously or essentially simultaneously, and the initial order of administration of the compound/composition, and the chemotherapeutic agent and/or radiation, may not be important.
- the compounds/compositions of the invention may be administered first followed by the administration of the chemotherapeutic agent and/or radiation; or the chemotherapeutic agent and/or radiation may be administered first followed by the administration of the compounds/compositions of the invention.
- This alternate administration may be repeated during a single treatment protocol.
- the determination of the order of administration, and the number of repetitions of administration of each therapeutic agent during a treatment protocol is well within the knowledge of the skilled physician after evaluation of the disease being treated and the condition of the patient.
- the chemotherapeutic agent and/or radiation may be administered first, especially if it is a cytotoxic agent, and then the treatment continued with the administration of the compounds/compositions of the invention followed, where determined advantageous, by the administration of the chemotherapeutic agent and/or radiation, and so on until the treatment protocol is complete.
- the practicing physician can modify each protocol for the administration of a compound/composition for treatment according to the individual patient's needs, as the treatment proceeds.
- the attending clinician in judging whether treatment is effective at the dosage administered, will consider the general well-being of the patient as well as more definite signs such as relief of disease-related symptoms, inhibition of tumor growth, actual shrinkage of the tumor, or inhibition of metastasis. Size of the tumor can be measured by standard methods such as radiological studies, e.g., CAT or MRI scan, and successive measurements can be used to judge whether or not growth of the tumor has been retarded or even reversed. Relief of disease-related symptoms such as pain, and improvement in overall condition can also be used to help judge effectiveness of treatment.
- HSP90 competitive binding assays and functional assays can be performed as known in the art by substituting in the compounds of the invention. Chiosis et al. Chemistry & Biology 2001, 8, 289-299, describe some of the known ways in which this can be done.
- competition binding assays using, e.g., geldanamycin or 17-AAG as a competitive binding inhibitor of HSP90 can be used to determine relative HSP90 affinity of the compounds of the invention by immobilizing the compound of interest or other competitive inhibitor on a gel or solid matrix, preincubating HSP90 with the other inhibitor, passing the preincubated mix over the gel or matrix, and then measuring the amount of HSP90 that retains or does not retain on the gel or matrix.
- Downstream effects can also be evaluated based on the known effect of HSP90 inhibition on function and stability of various steroid receptors and signaling proteins including, e.g., Raf1 and HER2.
- Compounds of the present invention induce dose-dependent degradation of these molecules, which can be measured using standard techniques. Inhibition of HSP90 also results in up-regulation of HSP90 and related chaperone proteins that can similarly be measured.
- Antiproliferative activity on various cancer cell lines can also be measured, as can morphological and functional differentiation related to HSP90 inhibition.
- Indirect techniques include nucleic acid hybridization and amplification using, e.g., polymerase chain reaction (PCR). These techniques are known to the person of skill and are discussed, e.g., in Sambrook, Fritsch & Maniatis Molecular Cloning: A Laboratory Manual, 2nd ed., Cold Spring Harbor Laboratory, Cold Spring Harbor, N.Y., 1989; Ausubel, et al.
- HER2 expression in breast cancer cells can be determined with the use of an immunohistochemical assay, such as the Dako HercepTM test (Dako Corp., Carpinteria, Calif.
- the HercepTM test is an antibody staining assay designed to detect HER2 overexpression in tumor tissue specimens. This particular assay grades HER2 expression into four levels: 0, 1, 2, and 3, with level 3 representing the highest level of HER2 expression.
- Accurate quantitation can be enhanced by employing an Automated Cellular Imaging System (ACIS) as described, e.g., by Press, M. et al. Modern Pathology 2000, 13, 225A.
- ACIS Automated Cellular Imaging System
- Antibodies polyclonal or monoclonal, can be purchased from a variety of commercial suppliers, or may be manufactured using well-known methods, e.g., as described in Harlow et al. Antibodies: A Laboratory Manual , 2nd ed; Cold Spring Harbor Laboratory, Cold Spring Harbor, N.Y., 1988.
- HER2 overexpression can also be determined at the nucleic acid level since there is a reported high correlation between overexpression of the HER2 protein and amplification of the gene that codes for it.
- One way to test this is by using RT-PCR.
- the genomic and cDNA sequences for HER2 are known.
- Specific DNA primers can be generated using standard, well-known techniques, and can then be used to amplify template already present in the cell. An example of this is described in Kurokawa, H. et al. Cancer Res . 2000, 60, 5887-5894.
- PCR can be standardized such that quantitative differences are observed as between normal and abnormal cells, e.g., cancerous and noncancerous cells.
- Well known methods employing, e.g., densitometry can be used to quantitate and/or compare nucleic acid levels amplified using PCR.
- FISH fluorescent in situ hybridization
- other assays can be used, e.g., Northern and/or Southern blotting.
- FISH fluorescent in situ hybridization
- this nucleic acid probe can be conjugated to a fluorescent molecule, e.g., fluorescein and/or rhodamine, that preferably does not interfere with hybridization, and which fluorescence can later be measured following hybridization.
- Immuno and nucleic acid detection can also be directed against proteins other than HSP90 and HER2, which proteins are nevertheless affected in response to HSP90 inhibition.
- the final compounds were usually purified by preparative TLC (silica gel 60 ⁇ , Whatman Partisil PK6F) or flash chromatography (silica gel 60 ⁇ , EMD Chemicals) using EtOAc/hexane or MeOH/CH 2 Cl 2 as eluents. Rf's were measured using silica gel TLC plates (silica gel 60 ⁇ , EMD Chemicals). Analytical HPLC chromatograms were obtained using a C18 column (Agilent Zorbax 300SB-C18; 5 microns; 4.6 mm ⁇ 150 mm).
- the samples were diluted to typically 0.1-1 mg/mL in MeOH or CH 3 CN and the injection volumes were typically 10 ⁇ L.
- the column was not heated, and UV detection was effected at 254 nm.
- 1 H-NMR spectra were recorded on a Bruker Avance 400 MHz spectrometer.
- the 2-(chloromethyl)-pyridine derivative in a solution of ammonia in MeOH was heated at 100° C. overnight whereupon it was concentrated under reduced pressure and purified by flash chromatography (MeOH/CH 2 Cl 2 ) to afford the 2-(aminomethyl)-pyridine derivative.
- Variant 1 A solution of the aromatic compound in MeOH/THF/acetate buffer (1N in each AcOH and AcONa) was treated with Br 2 (1.3-equiv) at r.t. for 5 min. The excess bromine and solvent were removed on a rotary evaporator. Work-up (CHCl 3 ) and flash chromatography afforded the desired bromobenzene.
- Variant 2 A solution of the aromatic compound (7 mmol) and N-halosuccinimide (NCS, NBS, or NIS, 1.06 equiv) in acetic acid (40 mL) was heated to 4-90° C. for 0.3-1 h. Evaporation, work-up (EtOAc) and flash chromatography afforded the desired halogenated benzene.
- the title compound was obtained by chlorination of 5-chloromethyl-1,2,3-trimethoxy-benzene with NCS according to the general procedure 3.1.
- Step 3 Acetic acid 4-bromo-3,5-dimethyl-pyridin-2-yl methyl ester
- Step 4 4-Bromo-3,5-dimethyl-pyridin-2-yl methanol
- the title compound was obtained by heating 2-chloromethyl-4-methoxy-3,5-dimethyl-pyridine hydrochloride in toluene as described in the patent by Tarbit, et al. WO 99/10326.
- Step 2 4-Chloro-7-(4-methoxy-3,5-dimethyl-pyridin-2-ylmethyl)-5-[(methyl-phenyl-amino)-methyl]-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine
- rHSP90 protein Stressgen, BC, Canada, #SPP-770
- PBS phosphate buffered saline
- biotin-GM biotinylated-geldanamycin
- biotin-GM biotinylated-geldanamycin
- Relative fluorescence units was measured using a SpectraMax Gemini XS Spectrofluorometer (Molecular Devices, Sunnyvale, Calif.) with an excitation at 485 nm and emission at 580 nm; data was acquired using SOFTmax®PRO software (Molecular Devices Corporation, Sunnyvale, Calif.).
- the background was defined as the RFU generated from wells that were not coated with HSP90 but were treated with the biotin-GM and streptavidin-PE.
- MCF7 breast carcinoma cell lysates were prepared by douncing in lysing buffer (20 mM HEPES, pH 7.3, 1 mM EDTA, 5 mM MgCl 2 , 100 mM KCl), and then incubated with or without test compound for 30 mins at 4° C., followed by incubation with biotin-GM linked to BioMagTM streptavidin magnetic beads (Qiagen) for 1 hr at 4° C. The tubes were placed on a magnetic rack, and the unbound supernatant removed. The magnetic beads were washed three times in lysis buffer and boiled for 5 mins at 95° C. in SDS-PAGE sample buffer.
- the lysate binding ability of selected compounds of the invention based on the above assay is summarized in Table 2.
- the IC 50 reported is the concentration of test compound needed to achieve 50% inhibition of the biotin-GM binding to rHSP90 in the MCF7 cell lysates.
- MCF7 breast carcinoma cells were grown in Dulbecco's modified Eagle's medium (DMEM) containing 10% fetal bovine serum (FBS) and 10 mM HEPES, and plated in 24 well plates (50% confluent). Twenty-four hrs later (cells are 65-70% confluent), test compounds were added and incubated overnight for 16 h. For the less potent compounds, the amounts added were 100 ⁇ M, 30 ⁇ M, 10 ⁇ M and 1 ⁇ M, and for more potent compounds, the amounts added were 1 ⁇ M, 0.3 ⁇ M, 0.1 ⁇ M, 0.03 ⁇ M, 0.01 ⁇ M and 0.003 ⁇ M.
- DMEM Dulbecco's modified Eagle's medium
- FBS fetal bovine serum
- HEPES fetal bovine serum
- the wells were washed with 1 mL phosphate buffered saline (PBS), and 200 ⁇ L trypsin was added to each well. After trypsinization was complete, 50 ⁇ L of FBS was added to each well. Then 200 ⁇ L cells was transferred to 96 well plates. The cells were pipetted up and down to obtain a single cell suspension. The plates were centrifuiged at 2,500 rpm for 1 min using a Sorvall Legend RTTM tabletop centrifuge (Kendro Laboratory Products, Asheville, N.C.). The cells were then washed once in PBS containing 0.2% BSA and 0.2% sodium azide (BA buffer).
- PBS phosphate buffered saline
- PE conjugated anti HER2/Neu antibody Becton Dickinson, #340552
- PE conjugated anti-keyhole limpet hemocyanin [KLH] Becton Dickinson, #340761
- control antibody was added at a dilution of 1:20 and 1:40 respectively (final concentration was 1 ⁇ g/mL) and the cells were pipeted up and down to form a single cell suspension, and incubated for 15 mins. The cells were washed twice with 200 ⁇ L BA buffer, and resuspended in 200 ⁇ L BA buffer, and transferred to FACSCAN tubes with an additional 250 ⁇ L BA buffer.
- IC 50 is defined as the concentration at which there was 50% degradation of the HER2/Neu protein.
- MTS assays measure the cytotoxicity of geldanamycin derivatives.
- MTS (3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium) is a tetrazolium dye that is converted to a formazan product by dehydrogenase enzymes of metabolically active cells (Corey, A. et al. “Use of an aqueous soluble tetrazolium/formazan assay for cell growth assays in culture,” Cancer Commun . 1991, 3, 207-212).
- % viable cells (Abs at 490 nm treated cells/Abs at 490 nm untreated cells) ⁇ 100
- IC 50 was defined as the concentration of the compound which gave rise to 50% reduction in viable cell number.
Landscapes
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Public Health (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Immunology (AREA)
- Oncology (AREA)
- Virology (AREA)
- Neurology (AREA)
- Biomedical Technology (AREA)
- Physical Education & Sports Medicine (AREA)
- Heart & Thoracic Surgery (AREA)
- Dermatology (AREA)
- Hematology (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Communicable Diseases (AREA)
- Pain & Pain Management (AREA)
- Cardiology (AREA)
- Rheumatology (AREA)
- Neurosurgery (AREA)
- Pulmonology (AREA)
- Urology & Nephrology (AREA)
- Molecular Biology (AREA)
- Vascular Medicine (AREA)
- Transplantation (AREA)
- Diabetes (AREA)
- Gastroenterology & Hepatology (AREA)
- Obesity (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
Abstract
Pyrrolopyrimidines and related analogs are described and demonstrated to have utility as Heat Shock Protein 90 (HSP90) inhibiting agents used in the treatment and prevention of various HSP90 mediated disorders, e.g., proliferative disorders. Methods of synthesis and use of such compounds are also described and claimed.
Description
- This application relates and claims priority to U.S. application Ser. No. 10/945,851 filed Sep. 20, 2004 and U.S. Provisional Application Ser. No. 60/504,135, filed Sep. 18, 2003, and U.S. Provisional Application Ser. No. 60/591,467, filed Jul. 26, 2004. This application also relates to three other U.S. Utility Applications Ser. No. 10/946,645 filed Sep 20, 2004 (now Publication No. 20050113340; 10/946,637 filed Sep. 20, 2004 (now Publication No. 2005-119282) and 10/946,628 filed Sep. 20, 2004 (now Publication No. This application further relates to International Application PCT/US02/35069, filed Oct. 30, 2002 (now Publication No. WO03/37860. All the above cited U.S. utility applications, provisional applications and international application are expressly incorporated herein by reference in their entirety.
- The invention relates in general to pyrrolopyrimidines and their broad-spectrum utility, e.g., in inhibiting heat shock protein 90 (HSP90) to thereby treat or prevent HSP90-mediated diseases.
- HSP90s are ubiquitous chaperone proteins that are involved in folding, activation and assembly of a wide range of proteins, including key proteins involved in signal transduction, cell cycle control and transcriptional regulation. Researchers have reported that HSP90 chaperone proteins are associated with important signaling proteins, such as steroid hormone receptors and protein kinases, including, e.g., Raf-1, EGFR, v-Src family kinases, Cdk4, and ErbB-2 (Buchner J. TIBS 1999, 24, 136-141; Stepanova, L. et al. Genes Dev. 1996, 10, 1491-502; Dai, K. et al. J. Biol. Chem. 1996, 271, 22030-4). Studies further indicate that certain co-chaperones, e.g., HSP70, p60/Hop/Sti1, Hip, Bag1, HSP40/Hdj2/Hsj1, immunophilins, p23, and p50, may assist HSP90 in its function (see, e.g., Caplan, A. Trends in Cell Biol. 1999, 9, 262-68).
- Ansamycin antibiotics, e.g., herbimycin A (HA), geldanamycin (GM), and 17-allylamninogeldanamycin (17-AAG) are thought to exert their anticancerous effects by tight binding of the N-terminus pocket of HSP90, thereby destabilizing substrates that normally interact with HSP90 (Stebbins, C. et al. Cell 1997, 89, 239-250). This pocket is highly conserved and has weak homology to the ATP-binding site of DNA gyrase (Stebbins, C. et al., supra; Grenert, J. P. et al. J. Biol. Chem. 1997, 272, 23843-50). Further, ATP and ADP have both been shown to bind this pocket with low affinity and to have weak ATPase activity (Proromou, C. et al. Cell 1997, 90, 65-75; Panaretou, B. et al. EMBO J. 1998, 17, 4829-36). In vitro and in vivo studies have demonstrated that occupancy of this N-terminal pocket by ansamycins and other HSP90 inhibitors alters HSP90 function and inhibits protein folding. At high concentrations, ansamycins and other HSP90 inhibitors have been shown to prevent binding of protein substrates to HSP90 (Scheibel, T. H. et al. Proc. Natl. Acad. Sci. USA 1999, 96, 1297-302; Schulte, T. W. et al. J. Biol. Chem. 1995, 270, 24585-8; Whitesell, L., et al. Proc. Natl. Acad. Sci. USA 1994, 91, 8324-8328). Ansamycins have also been demonstrated to inhibit the ATP-dependent release of chaperone-associated protein substrates (Schneider, C. L. et al. Proc. Natl. Acad. Sci., USA 1996, 93, 14536-41; Sepp-Lorenzino et al. J. Biol. Chem. 1995, 270, 16580-16587). In either event, the substrates are degraded by a ubiquitin-dependent process in the proteasome (Schneider, C. L., supra; Sepp-Lorenzino, L., et al. J. Biol. Chem. 1995, 270, 16580-16587; Whitesell, L. et al. Proc. Natl. Acad. Sci. USA 1994, 91, 8324-8328).
- HSP90 substrate destabilization occurs in tumor and non-transformed cells alike and has been shown to be especially effective on a subset of signaling regulators, e.g., Raf (Schulte, T. W. et al. Biochem. Biophys. Res. Commun. 1997, 239, 655-9; Schulte, T. W., et al. J. Biol. Chem. 1995, 270, 24585-8), nuclear steroid receptors (Segnitz, B.; U. Gehring J. Biol. Chem. 1997, 272, 18694-18701; Smith, D. F. et al. Mol. Cell. Biol. 1995, 15, 6804-12), v-Src (Whitesell, L., et al. Proc. Natl. Acad. Sci. USA 1994, 91, 8324-8328) and certain transmembrane tyrosine kinases (Sepp-Lorenzino, L. et al. J. Biol. Chem. 1995, 270, 16580-16587) such as EGF receptor (EGFR) and HER2/Neu (Hartmann, F., et al. Int. J. Cancer 1997, 70, 221-9; Miller, P. et al. Cancer Res. 1994, 54, 2724-2730; Mimnaugh, E. G., et al. J. Biol. Chem. 1996, 271, 22796-801; Schnur, R. et al. J. Med. Chem. 1995, 38, 3806-3812), CDK4, and mutant p53. Erlichman et al. Proc. AACR 2001, 42, abstract 4474. The ansamycin-induced loss of these proteins leads to the selective disruption of certain regulatory pathways and results in growth arrest at specific phases of the cell cycle (Muise-Heimericks, R. C. et al. J. Biol. Chem. 1998, 273, 29864-72), and apoptosis, and/or differentiation of cells so treated (Vasilevskaya, A. et al. Cancer Res., 1999, 59, 3935-40). Ansamycins thus hold great promise for the treatment and/or prevention of many types of cancers and proliferative disorders, and also hold promise as traditional antibiotics. However, their relative insolubility makes them difficult to formulate and administer, and they are not easily synthesized and currently must, at least in part, be generated through fermentation. Further, the hepatic toxicity of ansamyins is dose limiting.
- In addition to anti-cancer and antitumorgenic activity, HSP90 inhibitors have also been implicated in a wide variety of other utilities, including use as anti-inflammation agents, anti-infectious disease agents, agents for treating autoimmunity, agents for treating stroke, ischemia, multiple sclerosis, cardiac disorders, central nervous system related disorders and agents useful in promoting nerve regeneration (See, e.g., Rosen et al. WO 02/09696 (PCT/US01/23640); Degranco et al. WO 99/51223 (PCT/US99/07242); Gold, U.S. Pat. No. 6,210,974 B1; DeFranco et al., U.S. Pat. No. 6,174,875. Overlapping somewhat with the above, there are reports in the literature that fibrogenetic disorders including but not limited to scleroderma, polymyositis, systemic lupus, rheumatoid arthritis, liver cirrhosis, keloid formation, interstitial nephritis, and pulmonary fibrosis also may be treatable with HSP90 inhibitors. Strehlow, WO 02/02123 (PCT/US01/20578). Still further HSP90 modulation, modulators and uses thereof are reported in Application Nos. PCT/US03/04283, PCT/US02/35938, PCT/US02/16287, PCT/US02/06518, PCT/US98/09805, PCT/US00/09512, PCT/US01/09512, PCT/US01/23640, PCT/US01/46303, PCT/US01/46304, PCT/US02/06518, PCT/US02/29715, PCT/US02/35069, PCT/US02/35938, PCT/US02/39993, 60/293,246, 60/371,668, 60/335,391, 60/128,593, 60/337,919, 60/340,762, 60/359,484 and 60/331,893.
- Recently, purine derivatives showing HSP90 inhibitory activity have been reported, e.g., in PCT/US02/35069; PCT/US02/36075. Purine moieties are well accepted bioisosteres for a variety of ATP-dependent molecular targets, see, JP 10025294; U.S. Pat. No. 4,748,177; U.S. Pat. No. 4,772,606; U.S. Pat. No. 6,369,092; WO 00/06573; WO 02/055521; WO 02/055082; WO 02/055083; European Patent 0178178; Eur. J. Med. Chem. 1994, 29(1), 3-9; and J. Het. Chem. 1990, 27(5), 1409. However, compounds having the desired potency, selectivity and pharmaceutical properties required for effective HSP90 inhibition in vivo have not been reported. Therefore, a need remains for additional novel and potent HSP90 inhibitors that meet the demanding biological and pharmaceutical criteria required to proceed towards human clinical trials.
- The present invention is directed towards heterocyclic compounds, in particular, pyrrolopyrimidines and related compounds that show broad utility, e.g., by inhibiting HSP90 and treating diseases that are HSP90-dependent.
- In one aspect, the invention comprises heterocyclic compounds as specified below in Formulae A, I, II, III and IV. Also included in the scope of the present invention are stereoisomic forms, including the individual enantiomers and diastereomers, racemic mixtures, and diasteromeric mixtures, and combinations thereof, where appropriate, as well as polymorphs, specific racemates and stereoisomers, solvates, esters, tautomers, pharmaceutically acceptable salts and prodrugs of these compounds. Stereoisomers of the compounds of the present invention may be isolated by standard resolution techniques such as, for example, fractional crystallization and chiral column chromatography.
-
-
- X1 and X2 are the same or different and each is nitrogen or —CR6;
- X3 is nitrogen or —CR3 wherein R3 is hydrogen, OH, a keto tautomer, —OR8, —CN, halogen, lower alkyl, or —C(O)R9;
- X4 is nitrogen or —CR6 when X3 is nitrogen, and X4 is —CR6R7 when X3 is —CR3;
- R1 is halogen, —OR8, —SR8, or lower alkyl;
- R2 is —NR8R10;
- R4 is —(CH2)n— wherein n=0-3, —C(O), —C(S), —SO2—, or —SO2N—; and
- R5 is alkyl, aromatic, heteroaromatic, alicyclic, or heterocyclic, each of which is optionally bi- or tri-cyclic, and optionally substituted with H, halogen, lower alkyl, lower alkenyl, lower alkynyl, lower aryl, lower alicyclic, aralkyl, aryloxyalkyl, alkoxyalkyl, perhaloalkyl, perhaloalkyloxy, perhaloacyl, —N3, —SR8, —OR8, —CN, —CO2R9, —NO2, or —NR8R10.
- In certain embodiments, there are exclusionary provisos with respect to compounds disclosed in JP 10025294; U.S. Pat. No. 4,748,177; U.S. Pat. No. 4,748,177; U.S. Pat. No. 6,369,092; WO 00/06573; WO 02/055521; WO 02/055082; WO 02/055083; Eur. J. Med. Chem. 1994, 29(1), 3-9; and J. Het. Chem. 1990, 27(5), 1409, which disclose compounds with, -R4R5 comprising ribose or a derivative thereof, or a sugar or derivative thereof; and compounds where -R4R5 is a phosphonate or phosphonic acid, or is substituted with a phosphonate or phosphonic acid; or compounds connected where R4 is —CH2— or —(CH2)n— that are connected through an oxygen atom to another group.
-
-
- R0 is selected from hydrogen, halogen, lower alkyl, —SR8, —OR8, —CN, and —NHR8,
- R1 is halogen, —OR11, —SR11 or lower alkyl;
- R2 is —NHR8;
- R3 is selected from the group consisting of hydrogen, halogen, —SR8, —OR8, —CN, —C(O)R9, —C(O)OH, —NO2, —NR8R10, lower alkyl, lower alkenyl, lower alkynyl, lower perhaloalkyl, aryl, heteroaryl, alicyclic, and heterocyclic, all optionally substituted, wherein:
- the aryl, heteroaryl, alicyclic and heterocyclic groups are optionally mono-, bi- or tri-cyclic;
- R8 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N; and
- the optional substituents on R3 are selected from the group consisting of halogen, lower alkyl, lower alkenyl, lower alkynyl, —SR8, —OR8, —CN, —C(O)OH, —C(O)R9, —NO2, —NR8R10, lower aryl, heteroaryl, alicyclic, lower heterocyclic, arylalkyl, heteroarylalkyl, amino, alkylamino, dialkylamino, diarylalkylamino, oxo, oxa, perhaloalkyl, perhaloalkoxy, perhaloacyl, guanidine, pyridinyl, thiophene, furanyl, indole, indazole, phosphonates, phosphates, phosphoramides, sulfonates, sulfones, sulfates, sulphonamides, carbamates, ureas, thioureas and thioamides, wherein R8 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N;
- when R0 or R3 is —OH or —SH, the compound may exist as the corresponding (thio)keto tautomer or a mixture of keto-enol tautomers;
- R4 is —CHR12—, —C(O)—, —C(S)—, —S(O)— or —SO2—;
- R5 is aryl, heteroaryl, alicyclic, or heterocyclic, wherein:
- the aryl group is substituted with 3 to 5 substituents,
- the heteroaryl group is substituted with 2 to 5 substituents,
- the alicyclic group is substituted with 3 to 5 substituents,
- the heterocyclic group is substituted with 3 to 5 substituents, and
- the substituents are selected from the group consisting of halogen, lower alkyl, lower alkenyl, lower alkynyl, —SR8, —OR8, —CN, —C(O)OH, —C(O)R9, —NO2, —NR8R10, lower aryl, heteroaryl, alicyclic, lower heterocyclic, arylalkyl, heteroarylalkyl, amino, alkylamino, dialkylamino, diarylalkylarnino, oxo, oxa, perhaloalkyl, perhaloalkoxy, perhaloacyl, guanidine, pyridinyl, thiophene, furanyl, indole, indazole, phosphonates, phosphates, phosphoramides, sulfonates, sulfones, sulfates, sulphonamides, carbamates, ureas, thioureas and thioamides, wherein R8 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N;
- R8 is hydrogen, lower alkyl, lower alkenyl, lower alkynyl, lower aryl, lower heteroaryl, or —C(O)R9;
- R9 is H, lower alkyl, lower alkenyl, lower alkynyl, lower aryl, lower heteroaryl, —NR10R10, or —OR11, wherein R10 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N;
- R10 is hydrogen, lower alkyl, lower alkenyl, lower alkynyl, lower aryl or lower heteroaryl;
- R11 is lower alkyl, lower alkenyl, lower alkynyl, lower heteroaryl or lower aryl;
- R12 is hydrogen or lower alkyl;
- R0 and R10 taken together optionally form an exocyclic double bond which is optionally substituted, or optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N; and
- R3 and R10 taken together optionally form an exocyclic double bond which is optionally substituted, or optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N.
- In another aspect, the invention features pharmaceutical compositions comprising the compounds of the invention, in particular, the compounds of Formulae A, I, II, III or IV, or a polymorph, solvate, ester, tautomer, diastereoisomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, and one or more pharmaceutical excipients, for use in treatment or prevention of diseases that are HSP90-dependent.
- In another aspect, the invention features a method of treating an individual having an HSP90-mediated disorder by administering to the individual a pharmaceutical composition that comprises a pharmaceutically effective amount of a compound of Formula A, I, II, III or IV, or a polymorph, solvate, ester, tautomer, diastereomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof.
- In one embodiment, the invention provides a method for treating an individual having a disorder selected from the group of inflammatory diseases, infections, autoimmune disorders, stroke, ischemia, cardiac disorders, neurological disorders, fibrogenetic disorders, proliferative disorders, tumors, leukemias, neoplasms, cancers, carcinomas, metabolic diseases, and malignant diseases.
- In another embodiment, the invention provides a method for treating an individual having a fibrogenetic disorder, such as, for example, scleroderma, polymyositis, systemic lupus, rheumatoid arthritis, liver cirrhosis, keloid formation, interstitial nephritis or pulmonary fibrosis.
- In another embodiment, the invention provides a combination therapy comprising the administration of a pharmaceutically effective amount of a compound of Formula I, II, III, or IV, or a solvate, tautomer, diastereomer, enantiomer, pharmaceutically acceptable salt, polymorph, or prodrug thereof according to any of the preceding aspects or embodiments, and at least one therapeutic agent selected from the group of cytotoxic agents, anti-angiogenesis agents and anti-neoplastic agents. The anti-neoplastic agent may be selected from the group of alkylating agents, anti-metabolites, epidophyllotoxins antineoplastic enzymes, topoisomerase inhibitors, procarbazines, mitoxantrones, platinum coordination complexes, biological response modifiers and growth inhibitors, hormonal/anti-hormonal therapeutic agents, and haematopoietic growth factors.
- Any of the above described aspects and embodiments of the invention can be combined where practical.
- The individual compounds, methods and compositions prescribed do not preclude the utilization of other, unspecified steps and agents, and those of ordinary skill in the art will appreciate that additional steps and compounds may also be combined usefully within the spirit of various aspects and embodiments of the invention.
- Advantages of the invention depend on the specific aspect and embodiment and may include one or more of: ease of synthesis and/or formulation, solubility, and IC50 relative to previously existing compounds in the same or different classes of HSP90 inhibitors.
- I. Definitions
- A “pharmaceutically acceptable derivative or prodrug” means any pharmaceutically acceptable salt, ester, salt of an ester or other derivative of a compound of this invention, which, upon administration to a recipient, is capable of providing, either directly or indirectly, a compound of this invention or a pharmaceutically active metabolite or residue thereof. Particularly favored derivatives or prodrugs are those that increase the bioavailability of the compounds of this invention when such compounds are administered to a patient (e.g., by allowing orally administered compound to be more readily absorbed into blood) or which enhance delivery of the parent compound to a biological compartment (e.g., the brain or lymphatic system).
- A “pharmaceutically acceptable salt” may be prepared for any compound of the invention having a functionality capable of forming a salt, for example, an acid or base functionality. Pharmaceutically acceptable salts may be derived from organic or inorganic acids and bases. Compounds of the invention that contain one or more basic functional groups, e.g., amino or alkylamino, are capable of forming pharmaceutically acceptable salts with pharmaceutically acceptable organic and inorganic acids. These salts can be prepared in situ during the final isolation and purification of the compounds of the invention, or by separately reacting a purified compound of the invention in its free base form with a suitable organic or inorganic acid, and isolating the salt thus formed. Examples of suitable acid salts include acetate, adipate, alginate, aspartate, benzoate, benzenesulfonate, bisulfate, butyrate, citrate, camphorate, camphorsulfonate, cyclopentanepropionate, digluconate, dodecylsulfate, ethanesulfonate, formate, fumarate, glucoheptanoate, glycerophosphate, glycolate, hemisulfate, heptanoate, hexanoate, hydrochloride, hydrobromide, hydroiodide, 2-hydroxyethanesulfonate, lactate, maleate, malonate, methanesulfonate, 2-napthalenesulfonate, nicotinate, nitrate, oxalate, palmoate, pectinate, persulfate, 3-phenylpropionate, phosphate, picrate, pivalate, propionate, salicylate, succinate, sulfate, tartrate, thiocyanate, tosylate and undecanoate. Other acids, such as oxalic, while not in themselves pharmaceutically acceptable, may be employed in the preparation of salts useful as intermediates in obtaining the compounds of the invention and their pharmaceutically acceptable acid addition salts. See, e.g., Berge et al. “Pharmaceutical Salts”, J. Pharm. Sci. 1977, 66:1-19.
- Compounds of the present invention that contain one or more acidic functional groups are capable of forming pharmaceutically acceptable salts with pharmaceutically acceptable bases. The term “pharmaceutically acceptable salts” in these instances refers to the relatively non-toxic, inorganic and organic base addition salts of compounds of the present invention. These salts can likewise be prepared in situ during the final isolation and purification of the compounds, or by separately reacting the purified compound in its free acid form with a suitable base, such as the hydroxide, carbonate or bicarbonate of a pharmaceutically acceptable metal cation, with ammonia, or with a pharmaceutically acceptable organic primary, secondary or tertiary amine. Representative pharmaceutically acceptable cations include alkali or alkaline earth salts such as the lithium, sodium, potassium, calcium, magnesium, and aluminum salts and the like. Illustrative examples of some of the bases that can be used include sodium hydroxide, potassium hydroxide, choline hydroxide, sodium carbonate, N+(C1-4 alkyl)4, and the like. Representative organic amines useful for the formation of base addition salts include ethylamine, diethylamine, ethylenediamine, ethanolamine, diethanolamine, piperazine and the like. This invention also envisions the quatemization of any basic nitrogen-containing groups of the compounds disclosed herein. Water or oil-soluble or dispersible products may be obtained by such quaternization. See, for example, Berge et al., supra.
- Pharmaceutically acceptable prodrugs of the compounds of this invention include, but are not limited to, esters, carbonates, thiocarbonates, N-acyl derivatives, N-acyloxyalkyl derivatives, quaternary derivatives of tertiary amines, N-Mannich bases, Schiff bases, aminoacid conjugates, phosphate esters, metal salts and sulfonate esters.
- Suitable positions for derivatization of the compounds of the invention to create “prodrugs” include but are not limited, to, 2-amino substitution. Those of ordinary skill in the art have the knowledge and means to accomplish this without undue experimentation. Various forms of prodrugs are well known in the art. For examples of such prodrug derivatives, see, e.g.,
- a) Design of Prodrugs, Bundgaard, A. Ed., Elseview, 1985 and Method in Enzymology, Widder, K. et al., Ed.; Academic, 1985, vol. 42, p.309-396;
- b) Bundgaard, H. “Design and Application of Prodrugs” in A Textbook of Drug Design and Development, Krosgaard-Larsen and H. Bundgaard, Ed., 1991, Chapter 5, p. 113-191; and
- c) Bundgaard, H., Advanced Drug Delivery Review, 1992, 8, 1-38.
- Each of which is incorporated herein by reference.
- The tenn “prodrugs” as employed herein includes, but is not limited to, the following groups and combinations of these groups:
-
- Hydroxy Prodrugs:
-
- Acyloxyalkyl esters;
- Alkoxycarbonyloxyalkyl esters;
- Alkyl esters;
- Aryl esters; and
- Disulfide containing esters.
- The term “alkyl,” alone or in combination, refers to an optionally substituted straight-chain, or optionally substituted branched-chain saturated hydrocarbon radical having from one to thirty carbons, more preferably one to twelve carbons. Examples of alkyl radicals include methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, tert-amyl, pentyl, hexyl, heptyl, octyl and the like. The term “cycloalkyl” embraces cyclic alkyl radicals which include monocyclic, bicyclic, tricyclic, and higher multicyclic alkyl radicals wherein each cyclic moiety has from three to eight carbon atoms. Examples of cycloalkyl radicals include cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and the like. A “lower alkyl” is a shorter alkyl, e.g., one containing from one to six carbon atoms.
- The term “alkenyl,” alone or in combination, refers to an optionally substituted straight-chain, or optionally substituted branched-chain hydrocarbon radical having one or more carbon-carbon double-bonds and having from two to thirty carbon atoms, more preferably two to eighteen carbons. Examples of alkenyl radicals include ethenyl, propenyl, butenyl, 1,3-butadienyl and the like. The term “cycloalkenyl” refers to cyclic alkenyl radicals which include monocyclic, bicyclic, tricyclic, and higher multicyclic alkenyl radicals wherein each cyclic moiety has from three to eight carbon atoms. A “lower alkenyl” refers to an alkenyl having from two to six carbons.
- The term “alkynyl,” alone or in combination, refers to an optionally substituted straight-chain or optionally substituted branched-chain hydrocarbon radical having one or more carbon-carbon triple-bonds and having from two to thirty carbon atoms, more preferably from two to twelve carbon atoms, or from two to six carbon atoms, as well as those having from two to four carbon atoms. Examples of alkynyl radicals include ethynyl, 2-propynyl, 2-butynyl, 1,3-butadiynyl and the like. The term “cycloalkynyl” refers to cyclic alkynyl radicals which include monocyclic, bicyclic, tricyclic, and higher multicyclic alkynyl radicals wherein each cyclic moiety has from three to eight carbon atoms. A “lower alkynyl” refers to an alkynyl having from two to six carbons.
- The terms “heteroalkyl, heteroalkenyl and heteroalkynyl” include optionally substituted alkyl, alkenyl and alkynyl structures, as described above, and which have one or more skeletal chain atoms selected from an atom other than carbon, e.g., oxygen, nitrogen, sulfur, phosphorous or combinations thereof.
- The term “carbon chain” embraces any alkyl, alkenyl, alkynyl, or heteroalkyl, heteroalkenyl, or heteroalkynyl group, which are linear, cyclic, or any combination thereof. If the chain is part of a linker and that linker comprises one or more rings as part of the core backbone, for purposes of calculating chain length, the “chain” only includes those carbon atoms that compose the bottom or top of a given ring and not both, and where the top and bottom of the ring(s) are not equivalent in length, the shorter distance shall be used in determining the chain length. If the chain contains heteroatoms as part of the backbone, those atoms are not calculated as part of the carbon chain length.
- The term “membered ring” can embrace any cyclic structure, including aromatic, heteroaromatic, alicyclic, heterocyclic and polycyclic fused ring systems as described below. The term “membered” is meant to denote the number of skeletal atoms that constitute the ring. Thus, for example, pyridine, pyran, and pyrimidine are six-membered rings and pyrrole, tetrahydrofuran, and thiophene are five-membered rings.
- The term “aryl,” alone or in combination, refers to an optionally substituted aromatic hydrocarbon radical of six to twenty ring atoms, and includes mono-aromatic rings and fused aromatic rings. A fuised aromatic ring radical contains from two to four fused rings where the ring of attachment is an aromatic ring, and the other individual rings within the fused ring may be aromatic, heteroaromatic, alicyclic or heterocyclic. Further, the term aryl includes mono-aromatic rings and fused aromatic rings containing from six to twelve carbon atoms, as well as those containing from six to ten carbon atoms. Examples of aryl groups include, without limitation, phenyl, naphthyl, anthryl, chrysenyl, and benzopyrenyl ring systems. The term “lower aryl” refers to an aryl having six to ten skeletal ring carbons, e.g., phenyl and naphthyl ring systems.
- The term “heteroaryl” refers to optionally substituted aromatic radicals containing from five to twenty skeletal ring atoms and where one or more of the ring atoms is a heteroatom such as, for example, oxygen, nitrogen, sulfur, selenium or phosphorus. The term heteroaryl includes optionally substituted mono-heteroaryl radicals and fused heteroaryl radicals having at least one heteroatom (e.g., quinoline, benzothiazole). A fused heteroaryl radical may contain from two to four fused rings where the ring of attachment is a heteroaromatic ring, the other individual rings within the fused ring system may be aromatic, heteroaromatic, alicyclic or heterocyclic. The term heteroaryl also includes mono-heteroaryls or fused heteroaryls having from five to twelve skeletal ring atoms, as well as those having from five to ten skeletal ring atoms. Examples of heteroaryls include, without limitation, furanyl, benzofuranyl, chromenyl, pyridyl, pyrrolyl, indolyl, quinolinyl, pyridyl-N-oxide, pyrimidyl, pyrazinyl, imidazolyl, pyrazolyl, oxazolyl, isoxazolyl, benzothiozole, benzimidazole, benzoxazoles, benzothiadiazole, benzoxadiazole, benzotriazole, quinolines, isoquinolines, indoles, purinyl, indolizinyl, thienyl and the like and their oxides. The term “lower heteroaryl” refers to a heteroaryl having five to ten skeletal ring atoms, e.g., pyridyl, thienyl, pyrimidyl, pyrazinyl, pyrrolyl, or furanyl.
- The term “alicyclic” alone or in combination, refers to an optionally substituted saturated or unsaturated nonaromatic hydrocarbon ring system containing from three to twenty ring atoms. The term alicyclic includes mono-alicyclic and fused alicyclic radicals. A fused alicyclic may contain from two to four fused rings where the ring of attachment is an alicyclic ring, and the other individual rings within the fused-alicyclic radical may be aromatic, heteroaromatic, alicyclic and heterocyclic. The term alicyclic also includes mono-alicyclic and fuised alicyclic radicals containing from three to twelve carbon atoms, as well as those containing from three to ten carbon atoms. Examples of alicyclics include, without limitation, cyclopropyl, cyclopropenyl, cyclobutyl, cyclopentyl, cyclodecyl, cyclododecyl, cyclopentadienyl, indanyl, and cyclooctatetraenyl ring systems. The term “lower alicyclic” refers to an alicyclic having three to ten skeletal ring carbons, e.g., cyclopropyl, cyclopropenyl, cyclobutyl, cyclopentyl, decalinyl, and cyclohexyl.
- The term “heterocyclic” refers to optionally substituted saturated or unsaturated nonaromatic ring radicals containing from five to twenty ring atoms where one or more of the ring atoms are heteroatoms such as, for example, oxygen, nitrogen, sulfur, and phosphorus. The term alicyclic includes mono-heterocyclic and fused heterocyclic ring radicals. A fused heterocyclic radical may contain from two to four fused rings where the attaching ring is a heterocyclic, and the other individual rings within the fused heterocyclic radical may be aromatic, heteroaromatic, alicyclic or heterocyclic. The term heterocyclic also includes mono-heterocyclic and fused alicyclic radicals having from five to twelve skeletal ring atoms, as well as those having from five to ten skeletal ring atoms. Example of heterocyclics include without limitation, tetrahydrofuranyl, benzodiazepinyl, tetrahydroindazolyl, dihyroquinolinyl, and the like. The term “lower heterocyclic” refers to a heterocyclic ring system having five to ten skeletal ring atoms, e.g., dihydropyranyl, pyrrolidinyl, dioxolanyl, piperidinyl, piperazinyl, and the like.
- The term “alkylaryl,” or “araalkyl,” alone or in combination, refers to an aryl radical as defined above in which at least one H atom is replaced by an alkyl radical as defined above, such as, for example, tolyl, xylyl and the like.
- The term “arylalkyl,” alone or in combination, refers to an alkyl radical as defined above in which at least one H atom is replaced by an aryl radical as defined above, such as, for example, benzyl, 2-phenylethyl and the like.
- The termn “heteroarylalkyl” refers to an alkyl radical as defined above in which at least one H atom is replaced by a heteroaryl radical as defined above, each of which may be optionally substituted.
- The term “alkoxy,” alone or in combination, refers to an alkyl ether radical, alkyl-O—, wherein the term alkyl is defined as above. Examples of alkoxy radicals include methoxy, ethoxy, n-propoxy, isopropoxy, n-butoxy, iso-butoxy, sec-butoxy, tert-butoxy and the like.
- The term “aryloxy,” alone or in combination, refers to an aryl ether radical wherein the term aryl is defined as above. Examples of aryloxy radicals include phenoxy, thienyloxy and the like.
- The term “alkylthio,” alone or in combination, refers to an alkyl thio radical, alkyl-S—, wherein the term alkyl is as defined above.
- The term “arylthio,” alone or in combination, refers to an aryl thio radical, aryl-S—, wherein the term aryl is as defined above.
- The term “heteroarylthio” refers to the group heteroaryl-S—, wherein the term heteroaryl is as defined above.
- The term “acyl” refers to a radical —C(O)R where R includes alkyl, alkenyl, alkynyl, aryl, heteroaryl, alicyclic, heterocyclic, arylalkyl or heteroarylalkyl wherein the alkyl, alkenyl, alkynyl, aryl, heteroaryl, alicyclic, heterocyclic, arylalkyl or heteroaryl alkyl groups may be optionally substituted.
- The term “acyloxy” refers to the ester group —OC(O)R, where R is H, alkyl, alkenyl, alkynyl, aryl, heteroaryl, alicyclic, heterocyclic, arylalkyl, or heteroarylalkyl wherein the alkyl, alkenyl, alkynyl, aryl, heteroaryl, alicyclic, heterocyclic, arylalkyl or heteroarylalkyl may be optionally substituted.
- The term “carboxy esters” refers to —C(O)OR where R is alkyl, aryl or arylalkyl, wherein the alkyl, aryl and arylalkyl groups may be optionally substituted.
-
- where each of R and R′ are independently selected from the group consisting of H, alkyl, aryl, heteroaryl, alicyclic, heterocyclic, arylalkyl and heteroarylalkyl, wherein the alkyl, aryl, heteroaryl, alicyclic, heterocyclic, or arylalkyl groups may be optionally substituted.
- The term “oxo” refers to ═O.
- The term “halogen” includes F, Cl, Br and I.
- The terms “haloalkyl, haloalkenyl, haloalkynyl and haloalkoxy” include alkyl, alkenyl, alkynyl and alkoxy structures, as described above, that are substituted with one or more fluorines, chlorines, bromines or iodines, or with combinations thereof.
- The terms “perhaloalkyl, perhaloalkyloxy and perhaloacyl” refer to alkyl, alkyloxy and acyl radicals as described above, in which all the H atoms are replaced by fluorines, chlorines, bromines or iodines, or combinations thereof.
- The terms “cycloalkyl, arylalkyl, aryl, heteroaryl, alicyclic, heterocyclic, alkyl, alkynyl, alkenyl, haloalkyl, and heteroalkyl” include optionally substituted cycloalkyl, arylalkyl, aryl, heteroaryl, alicyclic, heterocyclic, alkyl, alkynyl, alkenyl, haloalkyl and heteroalkyl groups.
- The terms “alkylamino”, refers to the group —NHR where R is alkyl.
- The terms “dialkylamino”, refers to the group —NRR′ where R and R′ are alkyls.
- The term “sulfide” refers to a sulfur atom covalently linked to two atoms; the formal oxidation state of said sulfur is (II). The term “thioether” may be used interchangeably with the term “sulfide.”
- The term “sulfoxide” refers to a sulfur atom covalently linked to three atoms, at least one of which is an oxygen atom; the formal oxidation state of said sulfur atom is (IV).
- The term “sulfone” refers to a sulfur atom covalently linked to four atoms, at least two of which are oxygen atoms; the formal oxidation state of said sulfur atom is (VI).
- The terms “optional” or “optionally” mean that the subsequently described event or circumstance may but need not occur, and that the description includes instances where the event or circumstance occurs and instances in which it does not. For example, “aryl optionally mono- or di-substituted with an alkyl” means that the alkyl may but need not be present, or either one alkyl or two may be present, and the description includes situations where the aryl is substituted with one or two alkyls and situations where the aryl is not substituted with an alkyl.
- “Optionally substituted” groups may be substituted or unsubstituted. The substituents of an “optionally substituted” group may include, without limitation, one or more substituents independently selected from the following groups or designated subsets thereof: lower alkyl, lower alkenyl, lower alkynyl, lower aryl, heteroaryl, alicyclic, heterocyclic, arylalkyl, heteroarylalkyl, lower alkoxy, lower aryloxy, amino, alkylamino, dialkylamino, diarylalkylamino, alkylthio, arylthio, heteroarylthio, oxo, oxa, acyl (—C(O)R), (—C(O)), carboxyesters (—C(O)OR), carboxamido (—C(O)NH2), carboxy, acyloxy, —H, halo, —CN, —NO2, —N3, —SH, —OH, —C(O)CH3, perhaloalkyl, perhaloalkoxy, perhaloacyl, guanidine, pyridinyl, thiophene, furanyl, indole, indazole, esters, amides, phosphonates, phosphonic acid, phosphates, phosphoramides, sulfonates, sulfones, sulfates, sulphonamides, carbamates, ureas, thioureas, thioamides and thioalkyls. An optionally substituted group may be unsubstituted (e.g., —CH2CH3), fully substituted (e.g., —CF2CF3), monosubstituted (e.g., —CH2CH2F) or substituted at a level anywhere in-between fully substituted and monosubstituted (e.g., —CH2CF3).
- The term “pyridine-1-oxy” also means “pyridine-N-oxy.”
- Some of the compounds of the present invention may contain one or more chiral centers and therefore may exist in enantiomeric and diastereomeric forms. The scope of the present invention is intended to cover all isomers per se, as well as mixtures of cis and trans isomers, mixtures of diastereomers and racemic mixtures of enantiomers (optical isomers) as well. Further, it is possible using well known techniques to separate the various forms, and some embodiments of the invention may feature purified or enriched species of a given enantiomer or diastereomer.
- A “pharmacological composition” refers to a mixture of one or more of the compounds described herein, or pharmaceutically acceptable salts thereof, with other chemical components, such as pharmaceutically acceptable carriers and/or excipients. The purpose of a pharmacological composition is to facilitate administration of a compound to an organism.
- The phrase “pharmaceutically acceptable carrier” as used herein means a pharmaceutically-acceptable material, composition or vehicle, such as a liquid or solid filler, diluent, excipient, solvent or encapsulating material, involved in carrying or transporting the subject agent from one organ, or portion of the body, to another organ, or portion of the body. Each carrier must be “acceptable” in the sense of being compatible with the other ingredients of the formulation and not injurious to the patient. Some examples of materials which can serve as pharmaceutically-acceptable carriers include: (1) sugars, such as lactose, glucose and sucrose; (2) starches, such as corn starch and potato starch; (3) cellulose, and its derivatives, such as sodium carboxymethyl cellulose, ethyl cellulose and cellulose acetate; (4) powdered tragacanth; (5) malt; (6) gelatin; (7) talc; (8) excipients, such as cocoa butter and suppository waxes; (9) oils, such as peanut oil, cottonseed oil, safflower oil, sesame oil, olive oil, corn oil and soybean oil; (10) glycols, such as propylene glycol; (11) polyols, such as glycerin, sorbitol, mannitol and polyethylene glycol; (12) esters, such as ethyl oleate and ethyl laurate; (13) agar; (14) buffering agents, such as magnesium hydroxide and aluminum hydroxide; (15) alginic acid; (16) pyrogen-free water; (17) isotonic saline; (18) Ringer's solution; (19) ethyl alcohol; (20) phosphate buffer solutions; and (21) other non-toxic compatible substances employed in pharmaceutical formulations. A physiologically acceptable carrier should not cause significant irritation to an organism and does not abrogate the biological activity and properties of the administered compound.
- An “excipient” refers to an inert substance added to a pharmacological composition to further facilitate administration of a compound. Examples of excipients include but are not limited to calcium carbonate, calcium phosphate, various sugars and types of starch, cellulose derivatives, gelatin, vegetable oils and polyethylene glycols.
- A “pharmaceutically effective amount” means an amount which is capable of providing a therapeutic and/or prophylactic effect. The specific dose of compound administered according to this invention to obtain therapeutic and/or prophylactic effect will, of course, be determined by the particular circumstances surrounding the case, including, for example, the specific compound administered, the route of administration, the condition being treated, and the individual being treated. A typical daily dose (administered in single or divided doses) will contain a dosage level of from about 0.01 mg/kg to about 50-100 mg/kg of body weight of an active compound of the invention. Preferred daily doses generally will be from about 0.05 mg/kg to about 20 mg/kg and ideally from about 0.1 mg/kg to about 10 mg/kg. Factors such as clearance rate, half-life and maximum tolerated dose (MTD) have yet to be determined but one of ordinary skill in the art can determine these using standard procedures.
- In some method embodiments, the preferred therapeutic effect is the inhibition, to some extent, of the growth of cells characteristic of a proliferative disorder, e.g., breast cancer. A therapeutic effect will also normally, but need not, relieve to some extent one or more of the symptoms other than cell growth or size of cell mass. A therapeutic effect may include, for example, one or more of 1) a reduction in the number of cells; 2) a reduction in cell size; 3) inhibition (i.e., slowing to some extent, preferably stopping) of cell infiltration into peripheral organs, e.g., in the instance of cancer metastasis; 3) inhibition (i.e., slowing to some extent, preferably stopping) of tumor metastasis; 4) inhibition, to some extent, of cell growth; and/or 5) relieving to some extent one or more of the symptoms associated with the disorder.
- As used herein, the term IC50 refers to an amount, concentration or dosage of a particular test compound that achieves a 50% inhibition of a maximal response in an assay that measures such response. In some method embodiments of the invention, the “IC50” value of a compound of the invention can be greater for normal cells than for cells exhibiting a proliferative disorder, e.g., breast cancer cells. The value depends on the assay used.
- By a “standard” is meant a positive or negative control. A negative control in the context of HER2 expression levels is, e.g., a sample possessing an amount of HER2 protein that correlates with a normal cell. A negative control may also include a sample that contains no HER2 protein. By contrast, a positive control does contain HER2 protein, preferably of an amount that correlates with overexpression as found in proliferative disorders, e.g., breast cancers. The controls may be from cell or tissue samples, or else contain purified ligand (or absent ligand), immobilized or otherwise. In some embodiments, one or more of the controls may be in the form of a diagnostic “dipstick.”By “selectively targeting” is meant affecting one type of cell to a greater extent than another, e.g., in the case of cells with high as opposed to relatively low or normal HER2 levels.
- II. Compounds of the Invention
- Compounds of the invention and their polymorphs, solvates, esters, tautomers, diastereomers, enantiomers, pharmaceutically acceptable salts or prodrugs show utility for inhibiting HSP90 and treating and preventing diseases that are HSP90-dependent.
-
-
- X1 and X2 are the same or different and each is nitrogen or —CR6;
- X3 is nitrogen or —CR3 wherein R3 is hydrogen, OH, a keto tautomer, —OR8, —CN, halogen, lower alkyl, or —C(O)R9;
- X4 is nitrogen or a group CR6 when X3 is nitrogen, and X4 is —CR6R7 when X3 is —CR3;
- R1 is halogen, —OR8, —SR8, or lower alkyl;
- R2 is —NR8R10;
- R4 is —(CH2)n— wherein n=0-3, —C(O), —C(S), —SO2—, or —SO2N—; and
- R5 is alkyl, aryl, heteroaryl, alicyclic, or heterocyclic, each of which is optionally bi- or tricyclic, and optionally substituted with H, halogen, lower alkyl, lower alkenyl, lower alkynyl, lower aryl, lower alicyclic, araalkyl, aryloxyalkyl, alkoxyalkyl, perhaloalkyl, perhaloalkyloxy, perhaloacyl, —N3, —SR8, —OR8, —CN, —CO2R9, —NO2, or —NR8R10;
- with the provisos that:
- the compound is not one found or described in one or more of JP 10025294; U.S. Pat. No. 4,748,177; U.S. Pat. No. 4,748,177; U.S. Pat. No. 6,369,092; WO 00/06573; WO 02/055521; WO 02/055082; WO 02/055083; Eur. J. Med. Chem., 1994, 29(1), 3-9; and J. Het. Chem. 1990, 27(5), 1409;
- -R4R5 is not a ribose or derivative thereof, or a sugar or derivative thereof;
- -R4R5 is not a phosphonate or phosphonic acid, or a group substituted with a phosphonate or phosphonic acid; and
- when R4 is (CH2)n where n=0 or 1, then R4 and R5 are not connected with ‘O’, e.g., —CH2—O—CH2— or —CH2—CH2—O—CH2—.
- In one embodiment of, the compound, tautomer, pharmaceutically acceptable salt thereof, or prodrug thereof of Formula A, X1 and X2 are the same or different and each is nitrogen or —CR6; R1 is halogen, —OR8, —SR8, or lower alkyl; R2 is —NR8R10; R3 is hydrogen, —OH or keto tautomer, —OR8, halogen, —CN, lower alkyl, or —C(O)R9; R4 is —(CH2)n— wherein n=0-3, —C(O), —C(S), —SO2—, or —SO2N—; and R5 is alkyl, aromatic, heteroaromatic, alicyclic, heterocyclic, each of which is optionally bi- or tricyclic, and optionally substituted with H, halogen, lower alkyl, —SR8, —OR8, —CN, —CO2R9, —NO2 or —NR8R10; R8 is hydrogen, lower alkyl, lower aryl or —(CO)R9; R9 is lower alkyl, lower aryl, lower heteroaryl, —NR8R10 or OR11; R11 is lower alkyl or lower aryl; and R10 is hydrogen or lower alkyl.
- In one embodiment, the compound, tautomer, pharmaceutically acceptable salt thereof, or prodrug thereof of Formula A, R1 is selected from halogen, hydroxyl, lower alkoxy, lower thioalkyl and C1-4 alkyl; and R2 is —NH2.
- In another embodiment, R4 is —(CH2)n—, wherein n=0-3.
- In another embodiment, R1 is selected from halogen, hydroxyl, lower alkoxy, lower thioalkyl or C1-4 alkyl; optionally wherein R2 is NH2.
- In another embodiment, R4 is —(CH2)n—, wherein n=0-3.
- In another embodiment, R4 is —(CH2)n—, wherein n=0-3, R1 is selected from halogen, hydroxyl, lower alkoxy, lower thioalkyl, and C1-4 alkyl, and R2 is optionally NH2.
- In another embodiment, R1 is halogen, hydroxyl, lower alkoxy, lower thioalkyl, or C1-4 alkyl; and R2 is optionally NH2, R4 is —(CH2)—, and R5 is phenyl, benzyl, or pyridyl, all optionally substituted with H, halogen, lower alkyl, —SR8, —OR8 (or cyclic ethers such as methylenedioxy), —CN, —C02R9, —NO2, or —NR8R10; R8 is hydrogen, lower alkyl, lower aryl or —(CO)R9; R9 is lower alkyl, lower aryl, lower heteroaryl, —NR8R10 or —OR11; R11 is lower alkyl or lower aryl; and R10 is hydrogen or lower alkyl.
- In another embodiment R1 is halogen, R2 is —NH2, R4 is —CH2—, R6 is H or halogen, and R5 is phenyl optionally substituted with H, halogen, C1-4 alkyl, C1-4 alkoxy, C1-4 alkylthio, perhaloalkyl, perhaloalkyloxy, —CN, —NO2, —NH2 or —CO2R11.
- In another embodiment, R1 is halogen, R2 is —NH2, R4 is —CH2—, R6 is H, and R5 is 2-halo-3, 5-dimethoxyphenyl optionally substituted with H, halogen, C1-4 alkyl, C1-4 alkoxy, C1-4 alkylthio, perhaloalkyl, perhaloalkyloxy, —CN, —NO2, —NH2, or —CO2R11 at the para (4-) position.
- In another embodiment, R1 is chloro, R2 is —NH2, R4 is —CH2—, R6 is H and R5 is 2-chloro-3, 4,5-trimethoxyphenyl.
- In another embodiment, R1 is chloro, R2 is —NH2, R4 is —CH2—, R6 is H and R5 is 2-bromo-3, 4,5-trimethoxyphenyl. In other embodiments, R5 is selected from 2-iodo-3,4,5-trimethoxyphenyl, 2-fluoro-3,4,5-trimethoxyphenyl, and 2-bromo-3,4,5-trimethoxyphenyl.
- Any of the foregoing embodiments can be combined where feasible and appropriate.
-
-
- X1 and X2 are the same or different and each is nitrogen or a group —CR6;
- R1 is halogen, —OR8, —SR8, or lower alkyl;
- R2 is —NR8R10;
- R4 is —(CH2)n— where n=0-3, —C(O), —C(S), —SO2— or —SO2N—;
- R5 is alkyl, aromatic, heteroaromatic, alicyclic, heterocyclic, all optionally bi- or tricyclic, and all optionally substituted with H, halogen, lower alkyl, —SR8, —OR8, —CN, —CO2R9, —NO2, or —NR8R10;
- R6 is hydrogen, halogen, lower alkyl, —SR8, —OR8, —NR8R10, —N3, —CN, —C(O)R9, or taken together with R7 is carbonyl (C═O);
- R7 is independently selected from hydrogen, lower alkyl or taken together with R6 is —C(O);
- R8 is hydrogen, lower alkyl, lower aryl, or —(CO)R9;
- R9 is lower alkyl, lower aryl, lower heteroaryl, —NR8R10 or —OR11;
- R10 is hydrogen or lower alkyl, and
- R11 is lower alkyl or lower aryl.
- In one embodiment of the compounds of Formula A1, or a tautomer, pharmaceutically acceptable salt, or prodrug thereof, R1 is halogen, hydroxyl, lower alkoxy, lower thioalkyl, or C1-4 alkyl; and R2 is NH2.
- In another embodiment of the compounds of Formula A1, or a tautomer, pharmaceutically acceptable salt thereof, or prodrug thereof, R4 is —(CH2)n—, where n=0-3.
- In another embodiment of the compounds of Formula A1, or a tautomer, pharmaceutically acceptable salt, or prodrug thereof, R1 is halogen, hydroxyl, lower alkoxy, lower thioalkyl, or C1-4 alkyl; and R2 is NH2; R4 is —(CH2)n—, and wherein n=0-3.
- In another embodiment of the compounds of Formula A1, or a tautomer, pharmaceutically acceptable salt thereof, R1 is halogen; R2 is NH2, R4 is —CH2—.
-
- X1 and X2 are the same or different and each is nitrogen or —CR6;
- R1 is halogen, —OR8, —SR8 or lower alkyl;
- R2 is —NR8R10;
- R3 is hydrogen, OH or a keto tautomer, —OR8, halogen, —CN, lower alkyl or —C(O)R9;
- R4 is —(CH2)n— where n=0-3, —C(O), —C(S), —SO2— or —SO2N—;
- R6 is hydrogen, halogen, lower alkyl, —SR8, —OR8, —NR8R10, —N3, or —C(O)R9;
- R5 is alkyl, aromatic, heteroaromatic, alicyclic, heterocyclic, all optionally bi- or tricyclic, and all optionally substituted with H, halogen, lower alkyl, —SR8, —OR8, —CN, —CO2R9, —NO2, or —NR8R10;
- R8 is hydrogen, lower alkyl, lower aryl, or —(CO)R9;
- R9 is lower alkyl, lower aryl, lower heteroaryl, —NR8R10 or —OR11;
- R11 is lower alkyl or lower aryl; and
- R10 is hydrogen or lower alkyl.
- In another embodiment of the compounds of Formula A2, or a tautomer, pharmaceutically acceptable salt, or prodrug thereof, R1 is halogen, hydroxyl, lower alkoxy, lower thioalkyl or C1-4 alkyl; and wherein R2 is NH2.
- In another embodiment of the compounds of Formula A2, or a tautomer, pharmaceutically acceptable salt or prodrug thereof, R4 is —(CH2)n—, wherein n=0-3.
- In another embodiment of the compounds of Formula A2, or a tautomer, pharmaceutically acceptable salt, or prodrug thereof, R4 is —(CH2)—.
- In another embodiment of the compounds of Formula A2, or a tautomer, pharmaceutically acceptable salt or prodrug thereof, R1 is halogen, hydroxyl, lower alkoxy, lower thioalkyl or C1-4 alkyl; R2 is NH2, R4 is —(CH2)—.
-
-
- R0 is selected from hydrogen, halogen, lower alkyl, —SR8, —OR8, —CN, and —NHR8,
- R1 is halogen, —OR11, —SR11 or lower alkyl;
- R2 is —NHR8;
- R3 is selected from the group consisting of hydrogen, halogen, —SR8, —OR8, —CN, —C(O)R9, —C(O)OH, —NO2, —NR8R10, lower alkyl, lower alkenyl, lower alkynyl, aryl, lower perhaloalkyl, heteroaryl, alicyclic, heterocyclic, all optionally substituted, wherein:
- the aryl, heteroaryl, alicyclic and heterocyclic groups are optionally mono-, bi- or tri-cyclic,
- R8 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N, and
- the optional substituents on R3 are selected from the group consisting of halogen, lower alkyl, lower alkenyl, lower alkynyl, —SR8, —OR8, —CN, —C(O)OH, —C(O)R9, —NO2, —NR8R10, lower aryl, heteroaryl, alicyclic, lower heterocyclic, arylalkyl, heteroarylalkyl, amino, alkylamino, dialkylamino, diarylalkylamino, oxo, oxa, perhaloalkyl, perhaloalkoxy, perhaloacyl, guanidine, pyridinyl, thiophene, furanyl, indole, indazole, phosphonates, phosphates, phosphoramides, sulfonates, sulfones, sulfates, sulphonamides, carbamates, ureas, thioureas and thioamides, wherein R8 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N;
- R0 or R3 is —OH or —SH, the compound may exist as the corresponding (thio)keto tautomer or a mixture of keto-enol tautomers;
- R4 is —CHR12—, —C(O)—, —C(S)—, —S(O)— or —SO2—;
- R5 is aryl, heteroaryl, alicyclic, or heterocyclic, wherein
- the aryl group is substituted with 3 to 5 substituents,
- the heteroaryl group is substituted with 2 to 5 substituents,
- the alicyclic group is substituted with 3 to 5 substituents,
- the heterocyclic group is substituted with 3 to 5 substituents, and
- the substituents are selected from the group consisting of halogen, lower alkyl, lower alkenyl, lower alkynyl, —SR8, —OR8, —CN, —C(O)OH, —C(O)R9, —NO2, —NR8R10, lower aryl, heteroaryl, alicyclic, lower heterocyclic, arylalkyl, heteroarylalkyl, amino, alkylamino, dialkylamino, diarylalkylamino, oxo, oxa, perhaloalkyl, perhaloalkoxy, perhaloacyl, guanidine, pyridinyl, thiophene, furanyl, indole, indazole, phosphonates, phosphates, phosphoramides, sulfonates, sulfones, sulfates, sulphonamides, carbamates, ureas, thioureas and thioamides, wherein R8 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N;
- R8 is hydrogen, lower alkyl, lower alkenyl, lower alkynyl, lower aryl, lower heteroaryl, or —C(O)R9;
- R9 is H, lower alkyl, lower alkenyl, or lower alkynyl, lower aryl, lower heteroaryl, —NR10R10, or —OR11, wherein R10 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N;
- R10 is hydrogen, lower alkyl, lower alkenyl, lower alkynyl, lower aryl or lower heteroaryl;
- R11 is lower alkyl, lower alkenyl, or lower alkynyl, lower heteroaryl or lower aryl; and
- R12 is hydrogen or lower alkyl.
- In one embodiment of the compounds of Formula I, or a polymorph, solvate, ester, tautomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, each of the aryl, heteroaryl, alicyclic or heterocyclic group is monocyclic or bicyclic.
- In another embodiment of the compounds of Formula I, or a polymorph, solvate, ester, tautomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, R0 is hydrogen, halogen, —SH, —OH, or —CN; R1 is halogen; and R2 is —NHR8, where R8 is hydrogen or —C(O)R9.
- In another embodiment of the compounds of Formula I, or a polymorph, solvate, ester, tautomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, R1 is chloro or bromo, R2 is —NHR8, where R8 is hydrogen or —C(O)R9; R3is hydrogen, halogen, OR8, SR8, NR8R10, lower alkyl, lower alkenyl, lower alkynyl, lower perhaloalkyl, lower aryl, or lower heteroaryl.
- In another embodiment of the compounds of Formula I, or a polymorph, solvate, ester, tautomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, R0 is hydrogen, halogen or —CN; R2 is —NHR8, where R8 is hydrogen or —C(O)R9; and R4 is —CH2—.
- In another embodiment of the compounds of Formula I, or a polymorph, solvate, ester, tautomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, R0 is hydrogen, halogen, —SH, —OH or —CN; R1 is halogen; R2 is —NH2, R3 is hydrogen, halogen, —OR8, —SR8, —NR8R10, lower alkyl, lower alkenyl, lower alkynyl, perhaloalkyl, lower aryl, or lower heteroaryl, wherein R8 is hydrogen, lower alkyl, lower aryl, or —C(O)R9; R4 is —CH2—; and R5 is aryl or heteroaryl, wherein each of said aryl and heteroaryl is monocyclic or bicyclic and is substituted with 3 to 5 substituents.
- In another embodiment of the compounds of Formula I, or a polymorph, solvate, ester, tautomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, R1 is chloro or bromo, R2 is —NH2, and R5 is a phenyl having at least three substituents.
- In another embodiment of the compounds of Formula I, or a polymorph, solvate, ester, tautomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, R1 is chloro or bromo, R2 is —NH2 and R5 is a pyridyl having at least two substituents.
- In another embodiment of the compounds of Formula I, or a polymorph, solvate, ester, tautomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, R1 is chloro or bromo, R2 is —NH2, and R5 is 1-oxy-pyridyl (N-oxy-pyridyl) having at least two substituents.
-
-
- R0 is hydrogen, halogen, lower alkyl, —SR8, —OR8, —CN or —NHR8,
- R1 is halogen, —OR11, —SR11 or lower alkyl;
- R2 is —NH2;
- R4 is —CHR12—, —C(O)—, —C(S)—, —S(O)— or —SO2—;
- R5 is aryl, heteroaryl, alicyclic, or heterocyclic, wherein:
- the aryl group is substituted with 3 to 5 substituents,
- the heteroaryl group is substituted with 2 to 5 substituents,
- the alicyclic group is substituted with 3 to 5 substituents,
- the heterocyclic group is substituted with 3 to 5 substituents, and
- the substituents on R5 are selected from the group consisting of halogen, lower alkyl, lower alkenyl, lower alkynyl, —SR8, —OR8, —CN, —C(O)OH, —C(O)R9, —NO2, —NR8R10, lower aryl, heteroaryl, alicyclic, lower heterocyclic, arylalkyl, heteroarylalkyl, amino, alkylamino, dialkylamino, diarylalkylamino, oxo, oxa, perhaloalkyl, perhaloalkoxy, perhaloacyl, guanidine, pyridinyl, thiophene, furanyl, indole, indazole, phosphonates, phosphates, phosphoramides, sulfonates, sulfones, sulfates, sulphonamides, carbamates, ureas, thioureas and thioamides, wherein R8 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N;
- R8 is hydrogen, lower alkyl, lower alkenyl, lower alkynyl, lower aryl, lower heteroaryl, or —C(O)R9;
- R9 is H, lower alkyl, lower aryl, lower heteroaryl, —NR10R10, or —OR11, wherein R10 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N;
- R10 is hydrogen, lower alkyl, lower heteroaryl, lower aryl, lower alkenyl, or lower alkynyl,
- R11 is lower alkyl, lower alkenyl, lower alkynyl, lower heteroaryl or lower aryl;
- R12 is hydrogen or lower alkyl; and
- R0 and R10 taken together optionally form an exocyclic double bond which is optionally substituted, or optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N.
- In one embodiment of the compounds of Formula II, or a polymorph, solvate, ester, tautomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, R1 is halogen or lower alkyl; R4 is —CHR12—; R5 is aryl or heteroaryl, wherein each of said aryl and heteroaryl is monocyclic or bicyclic and is substituted with 3 to 5 substituents.
- In another embodiment of the compounds of Formula II, or a polymorph, solvate, ester, tautomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, R0 is hydrogen or —NHR 8, R1 is halogen, —OR11, —SR11 or lower alkyl; R10 is hydrogen or lower alkyl.
- In another embodiment of the compounds of Formula II, or a polymorph, solvate, ester, tautomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, R0 is hydrogen; R1 is halogen; R4 is —CH2—; and R5 is aryl or heteroaryl, wherein each of said aryl and heteroaryl is monocyclic or bicyclic and is substituted with 3 to 5 substituents; and R10 is hydrogen.
- In another embodiment of the compounds of Formula II, or a polymorph, solvate, ester, tautomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, R1 is chloro or bromo, R5is phenyl, pyridyl or 1-oxy-pyridyl (N-oxy-pyridyl) each of which has at least two substituents.
-
-
- R1 is halogen, —OR11, —SR11 or lower alkyl;
- R2 is —NH2;
- R3 is selected from the group consisting of hydrogen, halogen, —SR8, —OR8, —CN, —C(O)R9, —C(O)OH, —NO2, —NR8R10, lower alkyl, lower alkenyl, lower alkynyl, lower perhaloalkyl, aryl, heteroaryl, alicyclic, heterocyclic, all optionally substituted, wherein:
- the aryl, heteroaryl, alicyclic and heterocyclic groups are optionally mono-, bi- or tri-cyclic,
- R8 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N, and
- the optional substituents on R3 are selected from the group consisting of halogen, lower alkyl, lower alkenyl, lower alkynyl, —SR8, —OR8, —CN, —C(O)OH, —C(O)R9, —NO2, —NR8R10, lower aryl, heteroaryl, alicyclic, lower heterocyclic, arylalkyl, heteroarylalkyl, amino, alkylamino, dialkylamino, diarylalkylamino, oxo, oxa, perhaloalkyl, perhaloalkoxy, perhaloacyl, guanidine, pyridinyl, thiophene, furanyl, indole, indazole, phosphonates, phosphates, phosphoramides, sulfonates, sulfones, sulfates, sulphonamides, carbamates, ureas, thioureas and thioamides, wherein R8 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N;
- R4 is —CHR12—, —C(O)—, —C(S)—, —S(O)— or —SO2—;
- R5 is aryl, heteroaryl, alicyclic, or heterocyclic, wherein
- the aryl group is substituted with 3 to 5 substituents,
- the heteroaryl group is substituted with 2 to 5 substituents,
- the alicyclic group is substituted with 3 to 5 substituents,
- the heterocyclic group is substituted with 3 to 5 substituents, and
- the substituents on R5 are selected from the group consisting of halogen, lower alkyl, lower alkenyl, lower alkynyl, —SR8, —OR8, —CN, —C(O)OH, —C(O)R9, —NO2, —NR8R10, lower aryl, heteroaryl, alicyclic, lower heterocyclic, arylalkyl, heteroarylalkyl, amino, alkylamino, dialkylamino, diarylalkylamino, oxo, oxa, perhaloalkyl, perhaloalkoxy, perhaloacyl, guanidine, pyridinyl, thiophene, furanyl, indole, indazole, phosphonates, phosphates, phosphoramides, sulfonates, sulfones, sulfates, sulphonamides, carbamates, ureas, thioureas and thioamides, wherein R8 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N;
- R8 is hydrogen, lower alkyl, lower alkenyl, or lower alkynyl, lower aryl, lower heteroaryl, or —C(O)R9;
- R9 is H, lower alkyl, lower aryl, lower heteroaryl, —NR10R10, or —OR11, wherein R10 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N;
- R10 is hydrogen, lower alkyl, lower heteroaryl, lower aryl, lower alkenyl, or lower alkynyl,
- R11 is lower alkyl, lower alkenyl, lower alkynyl, lower heteroaryl or lower aryl;
- R12 is hydrogen or lower alkyl; and
- R3 and R10 taken together optionally form an exocyclic double bond which is optionally substituted, or optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N.
- In one embodiment of the compounds of Formula III, or a polymorph, solvate, ester, tautomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, R1 is halogen; R3 is hydrogen, halogen, —OR8, —SR8, —NR8R10, lower alkyl, lower alkenyl, lower alkynyl, lower perhaloalkyl, lower aryl, or lower heteroaryl, wherein R8 is hydrogen, lower alkyl, lower alkenyl, or lower alkynyl, lower aryl, lower heteroaryl, or —C(O)R9; R4 is —CH2—; R5 is aryl or heteroaryl, wherein each of said aryl and heteroaryl is monocyclic or bicyclic and is substituted with 3 to 5 substituents; and R10 is hydrogen or lower alkyl.
- In another embodiment of the compounds of Formula III, or a polymorph, solvate, ester, tautomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, R1 is halogen; R4 is —CH2—; R5 is aryl or heteroaryl, wherein each of said aryl and heteroaryl is monocyclic or bicyclic and is substituted with 3 to 5 substituents; and R10 is hydrogen.
- In another embodiment of the compounds of Formula III, or a polymorph, solvate, ester, tautomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, R1 is halogen; R3 is hydrogen; R4 is —CH2—; R5 is aryl or heteroaryl, wherein each of said aryl and heteroaryl is monocyclic or bicyclic and is substituted with 3 to 5 substituents; and R10 is hydrogen.
- In another embodiment of the compounds of Formula III, or a polymorph, solvate, ester, tautomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, wherein R1 is chloro or bromo, R5 is phenyl, pyridyl or 1-oxy-pyridyl (N-oxy-pyridyl), each of which has at least two substituents.
-
-
- R1 is halogen, —OR11, —SR11 or lower alkyl;
- R2 is —NH2;
- R4 is —CHR12—, —C(O)—, —C(S)—, —S(O)— or —SO2—;
- R5 is aryl, heteroaryl, alicyclic, or heterocyclic, wherein
- the aryl group is substituted with 3 to 5 substituents,
- the heteroaryl group is substituted with 2 to 5 substituents,
- the alicyclic group is substituted with 3 to 5 substituents,
- the heterocyclic group is substituted with 3 to 5 substituents, and
- the substituents on R5 are selected from the group consisting of halogen, lower alkyl, lower alkenyl, lower alkynyl, —SR8, —OR8, —CN, —C(O)OH, —C(O)R9, —NO2, —NR8R10, lower aryl, heteroaryl, alicyclic, lower heterocyclic, arylalkyl, heteroarylalkyl, amino, alkylamino, dialkylamino, diarylalkylamino, oxo, oxa, perhaloalkyl, perhaloalkoxy, perhaloacyl, guanidine, pyridinyl, thiophene, furanyl, indole, indazole, phosphonates, phosphates, phosphoramides, sulfonates, sulfones, sulfates, sulphonamides, carbamates, ureas, thioureas and thioamides, wherein R8 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N;
- R8 is hydrogen, lower alkyl, lower alkenyl, or lower alkynyl, lower aryl, lower heteroaryl, or —C(O)R9;
- R9 is H, lower alkyl, lower aryl, lower heteroaryl, —NR10R10, or —OR11, wherein R10 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N;
- R10 is hydrogen, lower alkyl, lower heteroaryl, lower aryl, lower alkenyl, or lower alkynyl,
- R11 is lower alkyl, lower alkenyl, or lower alkynyl, lower heteroaryl lower aryl; and
- R12 is hydrogen or lower alkyl.
- In one embodiment of the compounds of Formula IV, or a polymorph, solvate, ester, tautomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, R1 is halogen; R4 is —CH2—; R5 is aryl or heteroaryl, wherein each of said aryl and heteroaryl is monocyclic or bicyclic and is substituted with 3 to 5 substituents.
- In another embodiment of the compounds of Formula IV, or a polymorph, solvate, ester, tautomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, R1 is chloro or bromo, and R5 is phenyl, pyridyl or 1-oxy-pyridyl (N-oxy-pyridyl), each of which has at least two substituents.
- It should be understood that any of the foregoing embodiments can be combined where feasible and appropriate.
- Illustrative species of the compounds of the invention that are based on Formula I are described in TABLE 1. Prodrugs which can be employed with the compound of the invention include, but are not limited to, those listed in the Definition section above.
TABLE 1 Exemplary Compounds based on Formula I I No. Ex R1 R2 R3 R4 R5 R0 1 9 Cl NH2 H CH2 3,4,5-Trimethoxyphenyl H 2 Cl NH2 H CH2 2-Chloro-3,4,5-trimethoxyphenyl H 3 6 Cl NH2 H CH2 2-Bromo-3,4,5-trimethoxyphenyl H 4 7 Cl NH2 H CH2 2-Iodo-3,4,5-trimethoxyphenyl H 5 Cl NH2 H CH2 2-Fluoro-3,4,5-trimethoxyphenyl H 6 Cl NH2 H CH2 3,4,5-Trimethylphenyl H 7 Cl NH2 H CH2 2-Chloro-3,4,5-trimethylphenyl H 8 Cl NH2 H CH2 2-Bromo-3,4,5-trimethylphenyl H 9 Cl NH2 H CH2 2-Iodo-3,4,5-trimethylphenyl H 10 Cl NH2 H CH2 2-Fluoro-3,4,5-trimethylphenyl H 11 Cl NH2 H CH2 3,5-Dimethoxy-4-methylphenyl H 12 Cl NH2 H CH2 2-Chloro-3,5-dimethoxy-4-methylphenyl H 13 Cl NH2 H CH2 2-Bromo-3,5-dimethoxy-4-methylphenyl H 14 Cl NH2 H CH2 2-Iodo-3,5-dimethoxy-4-methylphenyl H 15 Cl NH2 H CH2 2-Fluoro-3,5-dimethoxy-4-methylphenyl H 16 Cl NH2 i-pr CH2 3,4,5-Trimethoxyphenyl H 17 Cl NH2 i-pr CH2 2-Chloro-3,4,5-trimethoxyphenyl H 18 Cl NH2 i-pr CH2 2-Bromo-3,4,5-trimethoxyphenyl H 19 Cl NH2 i-pr CH2 2-Iodo-3,4,5-trimethoxyphenyl H 20 Cl NH2 i-pr CH2 2-Fluoro-3,4,5-trimethoxyphenyl H 21 Cl NH2 i-pr CH2 3,4,5-Trimethylphenyl H 22 Cl NH2 i-pr CH2 2-Chloro-3,4,5-trimethylphenyl H 23 Cl NH2 i-pr CH2 2-Bromo-3,4,5-trimethylphenyl H 24 Cl NH2 i-pr CH2 2-Iodo-3,4,5-trimethylphenyl H 25 Cl NH2 i-pr CH2 2-Fluoro-3,4,5-trimethylphenyl H 26 Cl NH2 i-pr CH2 3,5-Dimethoxy-4-methylphenyl H 27 Cl NH2 i-pr CH2 2-Chloro-3,5-dimethoxy-4-methylphenyl H 28 Cl NH2 i-pr CU2 2-Bromo-3,5-dimethoxy-4-methylphenyl H 29 Cl NH2 i-pr CU2 2-Iodo-3,5-dimethoxy-4-methylphenyl H 30 Cl NH2 i-pr CU2 2-Fluoro-3,5-dimethoxy-4-methylphenyl H 31 Cl NH2 Et CH2 3,4,5-Trimethoxyphenyl H 32 Cl NH2 Et CH2 2-Chloro-3,4,5-trimethoxyphenyl H 33 Cl NH2 Et CH2 2-Bromo-3,4,5-trimethoxyphenyl H 34 Cl NH2 Et CH2 2-Iodo-3,4,5-trimethoxyphenyl H 35 Cl NH2 Et CH2 2-Fluoro-3,4,5-trimethoxyphenyl H 36 Cl NH2 Et CH2 3,4,5-Trimethylphenyl H 37 Cl NH2 Et CH2 2-Chloro-3,4,5-trimethylphenyl H 38 Cl NH2 Et CH2 2-Bromo-3,4,5-trimethylphenyl H 39 Cl NH2 Et CH2 2-Iodo-3,4,5-trimethylphenyl H 40 Cl NH2 Et CH2 2-Fluoro-3,4,5-trimethylphenyl H 41 Cl NH2 Et CH2 3,5-Dimethoxy-4-methylphenyl H 42 Cl NH2 Et CH2 2-Chloro-3,5-dimethoxy-4-methylphenyl H 43 Cl NH2 Et CH2 2-Bromo-3,5-dimethoxy-4-methylphenyl H 44 Cl NH2 Et CH2 2-Iodo-3,5-dimethoxy-4-methylphenyl H 45 Cl NH2 Et CH2 2-Fluoro-3,5-dimethoxy-4-methylphenyl H 46 Cl NH2 Me CH2 3,4,5-Trimethoxyphenyl H 47 Cl NH2 Me CH2 2-Chloro-3,4,5-trimethoxyphenyl H 48 Cl NH2 Me CH2 2-Bromo-3,4,5-trimethoxyphenyl H 49 Cl NH2 Me CH2 2-Iodo-3,4,5-trimethoxyphenyl H 50 Cl NH2 Me CH2 2-Fluoro-3,4,5-trimethoxyphenyl H 51 Cl NH2 Me CH2 3,4,5-Trimethylphenyl H 52 Cl NH2 Me CH2 2-Chloro-3,4,5-trimethylphenyl H 53 Cl NH2 Me CH2 2-Bromo-3,4,5-trimethylphenyl H 54 Cl NH2 Me CH2 2-Iodo-3,4,5-trimethylphenyl H 55 Cl NH2 Me CH2 2-Fluoro-3,4,5-trimethylphenyl H 56 Cl NH2 Me CH2 3,5-Dimethoxy-4-methylphenyl H 57 Cl NH2 Me CH2 2-Chloro-3,5-dimethoxy-4-methylphenyl H 58 Cl NH2 Me CH2 2-Bromo-3,5-dimethoxy-4-methylphenyl H 59 Cl NH2 Me CH2 2-Iodo-3,5-dimethoxy-4-methylphenyl H 60 Cl NH2 Me CH2 2-Fluoro-3,5-dimethoxy-4-methylphenyl H 61 Cl NH2 Ph CH2 3,4,5-Trimethoxyphenyl H 62 Cl NH2 Ph CH2 2-Chloro-3,4,5-trimethoxyphenyl H 63 Cl NH2 Ph CH2 2-Bromo-3,4,5-trimethoxyphenyl H 64 Cl NH2 Ph CH2 2-Iodo-3,4,5-trimethoxyphenyl H 65 Cl NH2 Ph CH2 2-Fluoro-3,4,5-trimethoxyphenyl H 66 Cl NH2 Ph CH2 3,4,5-Trimethylphenyl H 67 Cl NH2 Ph CH2 2-Chloro-3,4,5-trimethylphenyl H 68 Cl NH2 Ph CH2 2-Bromo-3,4,5-trimethylphenyl H 69 Cl NH2 Ph CH2 2-Iodo-3,4,5-trimethylphenyl H 70 Cl NH2 Ph CH2 2-Fluoro-3,4,5-trimethylphenyl H 71 Cl NH2 Ph CH2 3,5-Dimethoxy-4-methylphenyl H 72 Cl NH2 Ph CH2 2-Chloro-3,5-dimethoxy-4-methylphenyl H 73 Cl NH2 Ph CH2 2-Bromo-3,5-dimethoxy-4-methylphenyl H 74 Cl NH2 Ph CH2 2-Iodo-3,5-dimethoxy-4-methylphenyl H 75 Cl NH2 Ph CH2 2-Fluoro-3,5-dimethoxy-4-methylphenyl H 76 Cl NH2 2-Py CH2 3,4,5-Trimethoxyphenyl H 77 Cl NH2 2-Py CH2 2-Chloro-3,4,5-trimethoxyphenyl H 78 Cl NH2 2-Py CH2 2-Bromo-3,4,5-trimethoxyphenyl H 79 Cl NH2 2-Py CH2 2-Iodo-3,4,5-trimethoxyphenyl H 80 Cl NH2 2-Py CH2 2-Fluoro-3,4,5-trimethoxyphenyl H 81 Cl NH2 2-Py CH2 3,4,5-Trimethylphenyl H 82 Cl NH2 2-Py CH2 2-Chloro-3,4,5-trimethylphenyl H 83 Cl NH2 2-Py CH2 2-Bromo-3,4,5-trimethylphenyl H 84 Cl NH2 2-Py CH2 2-Iodo-3,4,5-trimethylphenyl H 85 Cl NH2 2-Py CH2 2-Fluoro-3,4,5-trimethylphenyl H 86 Cl NH2 2-Py CH2 3,5-Dimethoxy-4-methylphenyl H 87 Cl NH2 2-Py CH2 2-Chloro-3,5-dimethoxy-4-methylphenyl H 88 Cl NH2 2-Py CH2 2-Bromo-3,5-dimethoxy-4-methylphenyl H 89 Cl NH2 2-Py CH2 2-Iodo-3,5-dimethoxy-4-methylphenyl H 90 Cl NH2 2-Py CU2 2-Fluoro-3,5-dimethoxy-4-methylphenyl H 91 Cl NH2 4-Py CH2 3,4,5-Trimethoxyphenyl H 92 Cl NH2 4-Py CH2 2-Chloro-3,4,5-trimethoxyphenyl H 93 Cl NH2 4-Py CH2 2-Bromo-3,4,5-trimethoxyphenyl H 94 Cl NH2 4-Py CH2 2-Iodo-3,4,5-trimethoxyphenyl H 95 Cl NH2 4-Py CH2 2-Fluoro-3,4,5-trimethoxyphenyl H 96 Cl NH2 Ph CH2 3,4,5-Trimethylphenyl H 97 Cl NH2 Ph CH2 2-Chloro-3,4,5-trimethylphenyl H 98 Cl NH2 Ph CH2 2-Bromo-3,4,5-trimethylphenyl H 99 Cl NH2 Ph CH2 2-Iodo-3,4,5-trimethylphenyl H 100 Cl NH2 Ph CH2 2-Fluoro-3,4,5-trimethylphenyl H 101 Cl NH2 Ph CH2 3,5-Dimethoxy-4-methylphenyl H 102 Cl NH2 Ph CH2 2-Chloro-3,5-dimethoxy-4-methylphenyl H 103 Cl NH2 Ph CH2 2-Bromo-3,5-dimethoxy-4-methylphenyl H 104 Cl NH2 Ph CH2 2-Iodo-3,5-dimethoxy-4-methylphenyl H 105 Cl NH2 Pr CH2 2-Fluoro-3,5-dimethoxy-4-methylphenyl H 106 Cl NH2 Pr CH2 3,5-Dimethoxy-4-methylphenyl H 107 Cl NH2 Pr CH2 2-Chloro-3,5-dimethoxy-4-methylphenyl H 108 Cl NH2 Pr CH2 2-Bromo-3,5-dimethoxy-4-methylphenyl H 109 Cl NH2 Pr CH2 2-Iodo-3,5-dimethoxy-4-methylphenyl H 110 Cl NH2 Pr CH2 2-Fluoro-3,5-dimethoxy-4-methylphenyl H 111 Cl NH2 Pr CH2 3,4,5-Trimethoxyphenyl H 112 Cl NH2 Pr CH2 2-Chloro-3,4,5-trimethoxyphenyl H 113 Cl NH2 Pr CH2 2-Bromo-3,4,5-trimethoxyphenyl H 114 Cl NH2 Pr CH2 2-Iodo-3,4,5-trimethoxyphenyl H 115 Cl NH2 Pr CH2 2-Fluoro-3,4,5-trimethoxyphenyl H 116 Cl NH2 Pr CH2 3,4,5-Trimethylphenyl H 117 Cl NH2 Pr CH2 2-Chloro-3,4,5-trimethylphenyl H 118 Cl NH2 Pr CH2 2-Bromo-3,4,5-trimethylphenyl H 119 Cl NH2 Pr CH2 2-Iodo-3,4,5-trimethylphenyl H 120 Cl NH2 Pr CH2 2-Fluoro-3,4,5-trimethylphenyl H 121 Cl NH2 Pr CH2 3,5-Dimethoxy-4-methylphenyl H 122 Cl NH2 Pr CH2 2-Chloro-3,5-dimethoxy-4-methylphenyl H 123 Cl NH2 Pr CH2 2-Bromo-3,5-dimethoxy-4-methylphenyl H 124 Cl NH2 Pr CH2 2-Iodo-3,5-dimethoxy-4-methylphenyl H 125 Cl NH2 Pr CH2 2-Fluoro-3,5-dimethoxy-4-methylphenyl H 126 Br NH2 H CH2 3,4,5-Trimethoxyphenyl H 127 Br NH2 H CH2 2-Chloro-3,4,5-trimethoxyphenyl H 128 Br NH2 H CH2 2-Bromo-3,4,5-trimethoxyphenyl H 129 Br NH2 H CH2 2-Iodo-3,4,5-trimethoxyphenyl H 130 Br NH2 H CH2 2-Fluoro-3,4,5-trimethoxyphenyl H 131 Br NH2 H CH2 3,4,5-Trimethylphenyl H 132 Br NH2 H CH2 2-Chloro-3,4,5-trimethylphenyl H 133 Br NH2 H CH2 2-Bromo-3,4,5-trimethylphenyl H 134 Br NH2 H CH2 2-Iodo-3,4,5-trimethylphenyl H 135 Br NH2 H CH2 2-Fluoro-3,4,5-trimethylphenyl H 136 Br NH2 H CH2 3,5-Dimethoxy-4-methylphenyl H 137 Br NH2 H CH2 2-Chloro-3,5-dimethoxy-4-methylphenyl H 138 Br NH2 H CH2 2-Bromo-3,5-dimethoxy-4-methylphenyl H 139 Br NH2 H CH2 2-Iodo-3,5-dimethoxy-4-methylphenyl H 140 Br NH2 H CH2 2-Fluoro-3,5-dimethoxy-4-methylphenyl H 141 Cl NH2 i-Bu CH2 3,4,5-Trimethoxyphenyl H 142 Cl NH2 i-Bu CH2 2-Chloro-3,4,5-trimethoxyphenyl H 143 Cl NH2 i-Bu CH2 2-Bromo-3,4,5-trimethoxyphenyl H 144 Cl NH2 i-Bu CH2 2-Iodo-3,4,5-trimethoxyphenyl H 145 Cl NH2 i-Bu CH2 2-Fluoro-3,4,5-trimethoxyphenyl H 146 Cl NH2 i-Bu CH2 3,4,5-Trimethylphenyl H 147 Cl NH2 i-Bu CH2 2-Chloro-3,4,5-trimethylphenyl H 148 Cl NH2 i-Bu CH2 2-Bromo-3,4,5-trimethylphenyl H 149 Cl NH2 i-Bu CH2 2-Iodo-3,4,5-trimethylphenyl H 150 Cl NH2 i-Bu CH2 2-Fluoro-3,4,5-trimethylphenyl H 151 Cl NH2 i-Bu CH2 3,5-Dimethoxy-4-methylphenyl H 152 Cl NH2 i-Bu CH2 2-Chloro-3,5-dimethoxy-4-methylphenyl H 153 Cl NH2 i-Bu CH2 2-Bromo-3,5-dimethoxy-4-methylphenyl H 154 Cl NH2 i-Bu CH2 2-Iodo-3,5-dimethoxy-4-methylphenyl H 155 Cl NH2 i-Bu CH2 2-Fluoro-3,5-dimethoxy-4-methylphenyl H 156 Cl NH2 CN CH2 3,4,5-Trimethoxyphenyl H 157 Cl NH2 CN CH2 2-Chloro-3,4,5-trimethoxyphenyl H 158 Cl NH2 CN CH2 2-Bromo-3,4,5-trimethoxyphenyl H 159 Cl NH2 CN CH2 2-Iodo-3,4,5-trimethoxyphenyl H 160 Cl NH2 CN CH2 3,4,5-Trimethoxyphenyl H 161 Cl NH2 CN CH2 2-Chloro-3,4,5-trimethoxyphenyl H 162 Cl NH2 CN CH2 2-Bromo-3,4,5-trimethoxyphenyl H 163 Cl NH2 CN CH2 2-Iodo-3,4,5-trimethoxyphenyl H 164 Cl NH2 CN CH2 2-Fluoro-3,4,5-trimethoxyphenyl H 165 Cl NH2 CN CH2 3,4,5-Trimethylphenyl H 166 Cl NH2 CN CH2 2-Chloro-3,4,5-trimethylphenyl H 167 Cl NH2 CN CH2 2-Bromo-3,4,5-trimethylphenyl H 168 Cl NH2 CN CH2 2-Iodo-3,4,5-trimethylphenyl H 169 Cl NH2 CN CH2 2-Fluoro-3,4,5-trimethylphenyl H 170 Cl NH2 CN CH2 3,5-Dimethoxy-4-methylphenyl H 171 Cl NH2 CN CH2 2-Chloro-3,5-dimethoxy-4-methylphenyl H 172 Cl NH2 CN CH2 2-Bromo-3,5-dimethoxy-4-methylphenyl H 173 Cl NH2 CN CH2 2-Iodo-3,5-dimethoxy-4-methylphenyl H 174 Cl NH2 CN CH2 2-Fluoro-3,5-dimethoxy-4-methylphenyl H 175 Cl NH2 Cl CH2 3,4,5-Trimethoxyphenyl H 176 Cl NH2 Cl CH2 2-Chloro-3,4,5-trimethoxyphenyl H 177 Cl NH2 Cl CH2 2-Bromo-3,4,5-trimethoxyphenyl H 178 Cl NH2 Cl CH2 2-Iodo-3,4,5-trimethoxyphenyl H 179 Cl NH2 Cl CH2 2-Fluoro-3,4,5-trimethoxyphenyl H 180 Cl NH2 Cl CH2 3,4,5-Trimethylphenyl H 181 Cl NH2 Cl CH2 2-Chloro-3,4,5-trimethylphenyl H 182 Cl NH2 Cl CH2 2-Bromo-3,4,5-trimethylphenyl H 183 Cl NH2 Cl CH2 2-Iodo-3,4,5-trimethylphenyl H 184 Cl NH2 Cl CH2 2-Fluoro-3,4,5-trimethylphenyl H 185 Cl NH2 Cl CH2 3,5-Dimethoxy-4-methylphenyl H 186 Cl NH2 Cl CH2 2-Chloro-3,5-dimethoxy-4-methylphenyl H 187 Cl NH2 Cl CH2 2-Bromo-3,5-dimethoxy-4-methylphenyl H 188 Cl NH2 Cl CH2 2-Iodo-3,5-dimethoxy-4-methylphenyl H 189 Cl NH2 Cl CH2 2-Fluoro-3,5-dimethoxy-4-methylphenyl H 190 Cl NH2 Br CH2 3,4,5-Trimethoxyphenyl H 191 Cl NH2 Br CH2 2-Chloro-3,4,5-trimethoxyphenyl H 192 Cl NH2 Br CH2 2-Bromo-3,4,5-trimethoxyphenyl H 193 Cl NH2 Br CH2 2-Iodo-3,4,5-trimethoxyphenyl H 194 Cl NH2 Br CH2 2-Fluoro-3,4,5-trimethoxyphenyl H 195 Cl NH2 Br CH2 3,4,5-Trimethylphenyl H 196 Cl NH2 Br CH2 2-Chloro-3,4,5-trimethylphenyl H 197 Cl NH2 Br CH2 2-Bromo-3,4,5-trimethylphenyl H 198 Cl NH2 Br CH2 2-Iodo-3,4,5-trimethylphenyl H 199 Cl NH2 Br CH2 2-Fluoro-3,4,5-trimethylphenyl H 200 Cl NH2 Br CH2 3,5-Dimethoxy-4-methylphenyl H 201 Cl NH2 Br CH2 2-Chloro-3,5-dimethoxy-4-methylphenyl H 202 Cl NH2 Br CH2 2-Bromo-3,5-dimethoxy-4-methylphenyl H 203 Cl NH2 Br CH2 2-Iodo-3,5-dimethoxy-4-methylphenyl H 204 Cl NH2 Br CH2 2-Fluoro-3,5-dimethoxy-4-methylphenyl H 205 Cl NH2 I CH2 3,4,5-Trimethoxyphenyl H 206 Cl NH2 I CH2 2-Chloro-3,4,5-trimethoxyphenyl H 207 Cl NH2 I CH2 2-Bromo-3,4,5-trimethoxyphenyl H 208 Cl NH2 I CH2 2-Iodo-3,4,5-trimethoxyphenyl H 209 Cl NH2 I CH2 2-Fluoro-3,4,5-trimethoxyphenyl H 210 Cl NH2 I CH2 3,4,5-Trimethylphenyl H 211 Cl NH2 I CH2 2-Chloro-3,4,5-trimethylphenyl H 212 Cl NH2 I CH2 2-Bromo-3,4,5-trimethylphenyl H 213 Cl NH2 I CH2 2-Iodo-3,4,5-trimethylphenyl H 214 Cl NH2 I CH2 2-Fluoro-3,4,5-trimethylphenyl H 215 Cl NH2 I CH2 3,5-Dimethoxy-4-methylphenyl H 216 Cl NH2 I CH2 2-Chloro-3,5-dimethoxy-4-methylphenyl H 217 Cl NH2 I CH2 2-Bromo-3,5-dimethoxy-4-methylphenyl H 218 Cl NH2 I CH2 2-Iodo-3,5-dimethoxy-4-methylphenyl H 219 Cl NH2 I CH2 2-Fluoro-3,5-dimethoxy-4-methylphenyl H 220 Cl NH2 CH2—NMe2 CH2 3,4,5-Trimethoxyphenyl H 221 Cl NH2 CH2—NMe2 CH2 2-Chloro-3,4,5-trimethoxyphenyl H 222 Cl NH2 CH2—NMe2 CH2 2-Bromo-3,4,5-trimethoxyphenyl H 223 Cl NH2 CH2—NMe2 CH2 2-Iodo-3,4,5-trimethoxyphenyl H 224 Cl NH2 CH2—NMe2 CH2 2-Fluoro-3,4,5-trimethoxyphenyl H 225 Cl NH2 CH2—NMe2 CH2 3,4,5-Trimethylphenyl H 226 Cl NH2 CH2—NMe2 CH2 2-Chloro-3,4,5-trimethoxyphenyl H 227 Cl NH2 CH2—NMe2 CH2 2-Bromo-3,4,5-trimethoxyphenyl H 228 Cl NH2 CH2—NMe2 CH2 2-Iodo-3,4,5-trimethylphenyl H 229 Cl NH2 CH2—NMe2 CH2 2-Fluoro-3,4,5-trimethoxyphenyl H 230 Cl NH2 CH2—NMe2 CH2 3,5-Dimethoxy-4-methylphenyl H 231 Cl NH2 CH2—NMe2 CH2 2-Chloro-3,5-dimethoxy-4-methylphenyl H 232 Cl NH2 CH2—NMe2 CH2 2-Bromo-3,5-dimethoxy-4-methylphenyl H 233 Cl NH2 CH2—NMe2 CH2 2-Iodo-3,5-dimethoxy-4-methylphenyl H 234 Cl NH2 CH2—NMe2 CH2 2-Fluoro-3,5-dimethoxy-4-methylphenyl H 235 Cl NH2 3-Py CH2 3,4,5-Trimethoxyphenyl H 236 Cl NH2 3-Py CH2 2-Chloro-3,4,5-trimethoxyphenyl H 237 Cl NH2 3-Py CH2 2-Bromo-3,4,5-trimethoxyphenyl H 238 Cl NH2 3-Py CH2 2-Iodo-3,4,5-trimethoxyphenyl H 239 Cl NH2 3-Py CH2 2-Fluoro-3,4,5-trimethoxyphenyl H 240 5 Cl NH2 H CH2 3,5-Dimethyl-4-methoxypyridin-2-yl H 241 8 Cl NH2 H CH2 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl H 242 Cl NH2 H CH2 6-Bromo-3,5-dimethyl-4-methoxypyridin-2-yl H 243 10 Cl NH2 H CH2 6-Chloro-3,5-dimethyl-4-methoxypyridin-2-yl H 244 13 Cl NH2 H CH2 3,5-Dimethyl-4-bromopyridin-2-yl H 245 15 Cl NH2 H CH2 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl H 246 11 Cl NH2 H CH2 3,5-Dimethyl-4-chloropyridin-2-yl H 247 14 Cl NH2 H CH2 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl H 248 Cl NH2 H CH2 3,5-Dimethyl-4-iodopyridin-2-yl H 249 Cl NH2 H CH2 3,5-Dimethyl-4-iodo-1-oxypyridin-2-yl H 250 Cl NH2 H CH2 3,5-Dimethyl-4-thiomethyl-pyridin-2-yl H 251 Cl NH2 H CH2 3,5-Dimethyl-4-thiomethyl-1-oxypyridin-2-yl H 252 Cl NH2 H CH2 3,4,5-Trimethyl-pyridin-2-yl H 253 Cl NH2 H CH2 3,4,5-Trimethyl-1-oxypyridin-2-yl H 254 Cl NH2 H CH2 4,5,6-Trimethoxypyridin-2-yl H 255 Cl NH2 H CH2 4,5,6-Trimethoxy-1-oxypyridin-2-yl H 256 Cl NH2 H CH2 3-Bromo-4,5,6-trimethoxypyridin-2-yl H 257 Cl NH2 H CH2 3-Chloro-4,5,6-trimethoxypyridin-2-yl H 258 Cl NH2 H CH2 3,4,5-Trimethoxy-pyridin-2-yl H 259 Cl NH2 H CH2 3,4,5-Trimethoxy-1-oxypyridin-2-yl H 260 Cl NH2 H CH2 3-Bromo-3,4,5-trimethoxy-pyridin-2-yl H 261 Cl NH2 H CH2 3-Chloro-3,4,5-trimethoxy-pyridin-2-yl H 262 Cl NH2 H CH2 4,5,6-Trimethyl-pyridin-2-yl H 263 Cl NH2 H CH2 4,5,6-Trimethyl-1-oxypyridin-2-yl H 264 Cl NH2 H CH2 4,6-Dimethyl-5-methoxy-pyridin-2-yl H 265 Cl NH2 H CH2 4,6-Dimethyl-5-methoxypyridin-3-yl H 266 Cl NH2 H CH2 4,6-Dimethyl-5-methoxy-1-oxypyridin-3-yl H 267 Cl NH2 H CH2 4,6-Dimethyl-5-bromopyridin-3-yl H 268 Cl NH2 H CH2 4,6-Dimethyl-5-chloropyridin-3-yl H 269 Cl NH2 H CH2 5,6-Dimethyl-4-bromopyridin-3-yl H 270 Cl NH2 H CH2 5,6-Dimethyl-4-chloropyridin-3-yl H 271 Cl NH2 H CH2 2,6-Dimethyl-3-methoxypyridin-4-yl H 272 Cl NH2 H CH2 2,6-Dimethyl-pyridin-4-yl H 273 Cl NH2 H CH2 2,3,6-Trimethyl-pyridin-4-yl H 274 Cl NH2 H CH2 2,3,6-Trimethoxy-pyridin-4-yl H 275 Cl NH2 H CH2 2,6-Dimethyl-3-bromopyridin-4-yl H 276 Cl NH2 H CH2 2,6-Dimethyl-3-chloropyridin-4-yl H 277 Cl NH2 H CH2 2,6-Dimethyl-3-methoxy-1-oxy-pyridin-4-yl H 278 Cl NH2 H CH2 2,6-Dimethyl-1-oxy-pyridin-4-yl H 279 Cl NH2 H CH2 2,3,6-Trimethyl-1-oxy-pyridin-4-yl H 280 Cl NH2 H CH2 2,3,6-Trimethoxy-1-oxy-pyridin-4-yl H 281 Cl NH2 H CH2 2,6-Dimethyl-3-bromo1-oxy-pyridin-4-yl H 282 Cl NH2 H CH2 2,6-Dimethyl-3-chloro1-oxy-pyridin-4-yl H 283 Cl NH2 H CH2 4,6-Dimethyl-5-iodopyridin-3-yl H 284 Cl NH2 H CH2 3,5-Dimethyl-4-aminopyridin-2-yl H 285 Cl NH2 i-pr CH2 3,5-Dimethyl-4-methoxypyridin-2-yl H 286 Cl NH2 i-pr CH2 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl H 287 Cl NH2 i-pr CH2 6-Bromo-3,5-dimethyl-4-methoxypyridin-2-yl H 288 Cl NH2 i-pr CH2 6-Chloro-3,5-dimethyl-4-methoxypyridin-2-yl H 289 Cl NH2 i-pr CH2 3,5-Dimethyl-4-bromopyridin-2-yl H 290 Cl NH2 i-pr CH2 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl H 291 Cl NH2 i-pr CH2 3,5-Dimethyl-4-chloropyridin-2-yl H 292 Cl NH2 i-pr CH2 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl H 293 Cl NH2 i-pr CH2 3,5-Dimethyl-4-iodopyridin-2-yl H 294 Cl NH2 i-pr CH2 3,5-Dimethyl-4-iodo-1-oxypyridin-2-yl H 295 Cl NH2 i-pr CH2 3,5-Dimethyl-4-thiomethyl-pyridin-2-yl H 296 Cl NH2 i-pr CH2 3,5-Dimethyl-4-thiomethyl-1-oxypyridin-2-yl H 297 Cl NH2 i-pr CH2 3,4,5-Trimethyl-pyridin-2-yl H 298 Cl NH2 i-pr CH2 3,4,5-Trimethyl-1-oxypyridin-2-yl H 299 Cl NH2 i-pr CH2 4,5,6-Trimethoxypyridin-2-yl H 300 Cl NH2 i-pr CH2 4,5,6-Trimethoxy-1-oxypyridin-2-yl H 301 Cl NH2 i-pr CH2 3-Bromo-4,5,6-trimethoxypyridin-2-yl H 302 Cl NH2 i-pr CH2 3-Chloro-4,5,6-trimethoxypyridin-2-yl H 303 Cl NH2 i-pr CH2 3,4,5-Trimethoxy-pyridin-2-yl H 304 Cl NH2 i-pr CH2 3,4,5-Trimethoxy-1-oxypyridin-2-yl H 305 Cl NH2 i-pr CH2 3-Bromo-3,4,5-trimethoxy-pyridin-2-yl H 306 Cl NH2 i-pr CH2 3-Chloro-3,4,5-trimethoxy-pyridin-2-yl H 307 Cl NH2 i-pr CH2 4,5,6-Trimethyl-pyridin-2-yl H 308 Cl NH2 i-pr CH2 4,5,6-Trimethyl-1-oxypyridin-2-yl H 309 Cl NH2 i-pr CH2 4,6-Dimethyl-5-methoxy-pyridin-2-yl H 310 Cl NH2 i-pr CH2 4,6-Dimethyl-5-methoxypyridin-3-yl H 311 Cl NH2 i-pr CH2 4,6-Dimethyl-5-methoxy-1-oxypyridin-3-yl H 312 Cl NH2 i-pr CH2 4,6-Dimethyl-5-bromopyridin-3-yl H 313 Cl NH2 i-pr CH2 4,6-Dimethyl-5-chloropyridin-3-yl H 314 Cl NH2 i-pr CH2 5,6-Dimethyl-4-bromopyridin-3-yl H 315 Cl NH2 i-pr CH2 5,6-Dimethyl-4-chloropyridin-3-yl H 316 Cl NH2 i-pr CH2 2,6-Dimethyl-3-methoxypyridin-4-yl H 317 Cl NH2 i-pr CH2 2,6-Dimethyl-pyridin-4-yl H 318 Cl NH2 i-pr CH2 2,3,6-Trimethyl-pyridin-4-yl H 319 Cl NH2 i-pr CH2 2,3,6-Trimethoxy-pyridin-4-yl H 320 Cl NH2 i-pr CH2 2,6-Dimethyl-3-bromopyridin-4-yl H 321 Cl NH2 i-pr CH2 2,6-Dimethyl-3-chloropyridin-4-yl H 322 Cl NH2 i-pr CH2 2,6-Dimethyl-3-methoxy-1-oxy-pyridin-4-yl H 323 Cl NH2 i-pr CH2 2,6-Dimethyl-1-oxy-pyridin-4-yl H 324 Cl NH2 i-pr CH2 2,3,6-Trimethyl-1-oxy-pyridin-4-yl H 325 Cl NH2 i-pr CH2 2,3,6-Trimethoxy-1-oxy-pyridin-4-yl H 326 Cl NH2 i-pr CH2 2,6-Dimethyl-3-bromo1-oxy-pyridin-4-yl H 327 Cl NH2 i-pr CH2 2,6-Dimethyl-3-chloro1-oxy-pyridin-4-yl H 328 Cl NH2 i-pr CH2 4,6-Dimethyl-5-iodopyridin-3-yl H 329 Cl NH2 i-pr CH2 3,5-Dimethyl-4-aminopyridin-2-yl H 330 Cl NH2 Me CH2 3,5-Dimethyl-4-methoxypyridin-2-yl H 331 Cl NH2 Me CH2 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl H 332 Cl NH2 Me CH2 3,5-Dimethyl-4-bromopyridin-2-yl H 333 Cl NH2 Me CH2 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl H 334 Cl NH2 Me CH2 3,5-Dimethyl-4-chloropyridin-2-yl H 335 Cl NH2 Me CH2 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl H 336 Cl NH2 Me CH2 3,5-Dimethyl-4-iodopyridin-2-yl H 337 Cl NH2 Me CH2 3,5-Dimethyl-4-iodo-1-oxypyridin-2-yl H 338 Cl NH2 Me CH2 3,5-Dimethyl-4-thiomethyl-pyridin-2-yl H 339 Cl NH2 Me CH2 3,5-Dimethyl-4-thiomethyl-1-oxypyridin-2-yl H 340 Cl NH2 Me CH2 3,4,5-Trimethyl-pyridin-2-yl H 341 Cl NH2 Me CH2 3,4,5-Trimethyl-1-oxypyridin-2-yl H 342 Cl NH2 Me CH2 4,5,6-Trimethoxypyridin-2-yl H 343 Cl NH2 Me CH2 4,5,6-Trimethoxy-1-oxypyridin-2-yl H 344 Cl NH2 Me CH2 3,4,5-Trimethoxy-pyridin-2-yl H 345 Cl NH2 Me CH2 3,4,5-Trimethoxy-1-oxypyridin-2-yl H 346 Cl NH2 Me CH2 4,5,6-Trimethyl-pyridin-2-yl H 347 Cl NH2 Me CH2 4,5,6-Trimethyl-1-oxypyridin-2-yl H 348 Cl NH2 Me CH2 4,6-Dimethyl-5-methoxy-pyridin-2-yl H 349 Cl NH2 Me CH2 4,6-Dimethyl-5-methoxypyridin-3-yl H 350 Cl NH2 Me CH2 4,6-Dimethyl-5-methoxy-1-oxypyridin-3-yl H 351 Cl NH2 Me CH2 4,6-Dimethyl-5-bromopyridin-3-yl H 352 Cl NH2 Me CH2 4,6-Dimethyl-5-chloropyridin-3-yl H 353 Cl NH2 Me CH2 5,6-Dimethyl-4-bromopyridin-3-yl H 354 Cl NH2 Me CH2 5,6-Dimethyl-4-chloropyridin-3-yl H 355 Cl NH2 Me CH2 2,6-Dimethyl-3-methoxypyridin-4-yl H 356 Cl NH2 Me CH2 2,6-Dimethyl-pyridin-4-yl H 357 Cl NH2 Me CH2 2,3,6-Trimethyl-pyridin-4-yl H 358 Cl NH2 Me CH2 2,3,6-Trimethoxy-pyridin-4-yl H 359 Cl NH2 Me CH2 2,6-Dimethyl-3-bromopyridin-4-yl H 360 Cl NH2 Me CH2 2,6-Dimethyl-3-chloropyridin-4-yl H 361 Cl NH2 Me CH2 4,6-Dimethyl-5-iodopyridin-3-yl H 362 Cl NH2 Me CH2 3,5-Dimethyl-4-aminopyridin-2-yl H 363 Cl NH2 Et CH2 3,5-Dimethyl-4-methoxypyridin-2-yl H 364 Cl NH2 Et CH2 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl H 365 Cl NH2 Et CH2 3,5-Dimethyl-4-bromopyridin-2-yl H 366 Cl NH2 Et CH2 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl H 367 Cl NH2 Et CH2 3,5-Dimethyl-4-chloropyridin-2-yl H 368 Cl NH2 Et CH2 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl H 369 Cl NH2 Et CH2 3,5-Dimethyl-4-iodopyridin-2-yl H 370 Cl NH2 Et CH2 3,5-Dimethyl-4-iodo-1-oxypyridin-2-yl H 371 Cl NH2 Ef CH2 3,5-Dimethyl-4-thiomethyl-pyridin-2-yl H 372 Cl NH2 Et CH2 3,5-Dimethyl-4-thiomethyl-1-oxypyridin-2-yl H 373 Cl NH2 Et CH2 3,4,5-Trimethyl-pyridin-2-yl H 374 Cl NH2 Et CH2 3,4,5-Trimethyl-1-oxypyridin-2-yl H 375 Cl NH2 Et CH2 4,5,6-Trimethoxypyridin-2-yl H 376 Cl NH2 Et CH2 4,5,6-Trimethoxy-1-oxypyridin-2-yl H 377 Cl NH2 Et CH2 3,4,5-Trimethoxy-pyridin-2-yl H 378 Cl NH2 Et CH2 3,4,5-Trimethoxy-1-oxypyridin-2-yl H 379 Cl NH2 Et CH2 4,5,6-Trimethyl-pyridin-2-yl H 380 Cl NH2 Et CH2 4,5,6-Trimethyl-1-oxypyridin-2-yl H 381 Cl NH2 Et CH2 4,6-Dimethyl-5-methoxy-pyridin-2-yl H 382 Cl NH2 Et CH2 4,6-Dimethyl-5-methoxypyridin-3-yl H 383 Cl NH2 Et CH2 4,6-Dimethyl-5-methoxy-1-oxypyridin-3-yl H 384 Cl NH2 Et CH2 4,6-Dimethyl-5-bromopyridin-3-yl H 385 Cl NH2 Et CH2 4,6-Dimethyl-5-chloropyridin-3-yl H 386 Cl NH2 Et CH2 5,6-Dimethyl-4-bromopyridin-3-yl H 387 Cl NH2 Et CH2 5,6-Dimethyl-4-chloropyridin-3-yl H 388 Cl NH2 Et CH2 2,6-Dimethyl-3-methoxypyridin-4-yl H 389 Cl NH2 Et CH2 2,6-Dimethyl-pyridin-4-yl H 390 Cl NH2 Et CH2 2,3,6-Trimethyl-pyridin-4-yl H 391 Cl NH2 Et CH2 2,3,6-Trimethoxy-pyridin-4-yl H 392 Cl NH2 Et CH2 2,6-Dimethyl-3-bromopyridin-4-yl H 393 Cl NH2 Et CH2 2,6-Dimethyl-3-chloropyridin-4-yl H 394 Cl NH2 Et CH2 4,6-Dimethyl-5-iodopyridin-3-yl H 395 Cl NH2 Et CH2 3,5-Dimethyl-4-aminopyridin-2-yl H 396 Cl NH2 2-Py CH2 3,5-Dimethyl-4-methoxypyridin-2-yl H 397 Cl NH2 2-Py CH2 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl H 398 Cl NH2 2-Py CH2 3,5-Dimethyl-4-bromopyridin-2-yl H 399 Cl NH2 2-Py CH2 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl H 400 Cl NH2 2-Py CH2 3,5-Dimethyl-4-chloropyridin-2-yl H 401 Cl NH2 2-Py CH2 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl H 402 Cl NH2 2-Py CH2 3,5-Dimethyl-4-iodopyridin-2-yl H 403 Cl NH2 2-Py CH2 3,5-Dimethyl-4-iodo-1-oxypyridin-2-yl H 404 Cl NH2 2-Py CH2 3,5-Dimethyl-4-thiomethyl-pyridin-2-yl H 405 Cl NH2 2-Py CH2 3,5-Dimethyl-4-thiomethyl-1-oxypyridin-2-yl H 406 Cl NH2 2-Py CH2 3,4,5-Trimethyl-pyridin-2-yl H 407 Cl NH2 2-Py CH2 3,4,5-Trimethyl-1-oxypyridin-2-yl H 408 Cl NH2 2-Py CH2 4,5,6-Trimethoxypyridin-2-yl H 409 Cl NH2 2-Py CH2 4,5,6-Trimethoxy-1-oxypyridin-2-yl H 410 Cl NH2 2-Py CH2 3,4,5-Trimethoxy-pyridin-2-yl H 411 Cl NH2 2-Py CH2 3,4,5-Trimethoxy-1-oxypyridin-2-yl H 412 Cl NH2 2-Py CH2 4,5,6-Trimethyl-pyridin-2-yl H 413 Cl NH2 2-Py CH2 4,5,6-Trimethyl-1-oxypyridin-2-yl H 414 Cl NH2 2-Py CH2 4,6-Dimethyl-5-methoxy-pyridin-2-yl H 415 Cl NH2 2-Py CH2 4,6-Dimethyl-5-methoxypyridin-3-yl H 416 Cl NH2 2-Py CH2 4,6-Dimethyl-5-methoxy-1-oxypyridin-3-yl H 417 Cl NH2 2-Py CH2 4,6-Dimethyl-5-bromopyridin-3-yl H 418 Cl NH2 2-Py CH2 4,6-Dimethyl-5-chloropyridin-3-yl H 419 Cl NH2 2-Py CH2 5,6-Dimethyl-4-bromopyridin-3-yl H 420 Cl NH2 2-Py CH2 5,6-Dimethyl-4-chloropyridin-3-yl H 421 Cl NH2 2-Py CH2 2,6-Dimethyl-3-methoxypyridin-4-yl H 422 Cl NH2 2-Py CH2 2,6-Dimethyl-pyridin-4-yl H 423 Cl NH2 2-Py CH2 2,3,6-Trimethyl-pyridin-4-yl H 424 Cl NH2 2-Py CH2 2,3,6-Trimethoxy-pyridin-4-yl H 425 Cl NH2 2-Py CH2 2,6-Dimethyl-3-bromopyridin-4-yl H 426 Cl NH2 2-Py CH2 2,6-Dimethyl-3-chloropyridin-4-yl H 427 Cl NH2 2-Py CH2 4,6-Dimethyl-5-iodopyridin-3-yl H 428 Cl NH2 2-Py CH2 3,5-Dimethyl-4-aminopyridin-2-yl H 429 Cl NH2 Ph CH2 3,5-Dimethyl-4-methoxypyridin-2-yl H 430 Cl NH2 Ph CH2 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl H 431 Cl NH2 Ph CH2 3,5-Dimethyl-4-bromopyridin-2-yl H 432 Cl NH2 Ph CH2 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl H 433 Cl NH2 Ph CH2 3,5-Dmethyl-4-chloropyridin-2-yl H 434 Cl NH2 Ph CH2 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl H 435 Cl NH2 Ph CH2 3,5-Dimethyl-4-iodopyridin-2-yl H 436 Cl NH2 Ph CH2 3,5-Dimethyl-4-iodo-1-oxypyridin-2-yl H 437 Cl NH2 Ph CH2 3,5-Dimethyl-4-thiomethyl-pyridin-2-yl H 438 Cl NH2 Ph CH2 3,5-Dimethyl-4-thiomethyl-1-oxypyridin-2-yl H 439 Cl NH2 Ph CH2 3,4,5-Trimethyl-pyridin-2-yl H 440 Cl NH2 Ph CH2 3,4,5-Trimethyl-1-oxypyridin-2-yl H 441 Cl NH2 Ph CH2 4,5,6-Trimethoxypyridin-2-yl H 442 Cl NH2 Ph CH2 4,5,6-Trimethoxy-1-oxypyridin-2-yl H 443 Cl NH2 Ph CH2 3,4,5-Trimethoxy-pyridin-2-yl H 444 Cl NH2 Ph CH2 3,4,5-Trimethoxy-1-oxypyridin-2-yl H 445 Cl NH2 Ph CH2 4,5,6-Trimethyl-pyridin-2-yl H 446 Cl NH2 Ph CH2 4,5,6-Trimethyl-1-oxypyridin-2-yl H 447 Cl NH2 Ph CH2 4,6-Dimethyl-5-methoxy-pyridin-2-yl H 448 Cl NH2 Ph CH2 4,6-Dimethyl-5-methoxypyridin-3-yl H 449 Cl NH2 Ph CH2 4,6-Dimethyl-5-methoxy-1-oxypyridin-3-yl H 450 Cl NH2 Ph CH2 4,6-Dimethyl-5-bromopyridin-3-yl H 451 Cl NH2 Ph CH2 4,6-Dimethyl-5-chloropyridin-3-yl H 452 Cl NH2 Ph CH2 5,6-Dimethyl-4-bromopyridin-3-yl H 453 Cl NH2 Ph CH2 5,6-Dimethyl-4-chloropyridin-3-yl H 454 Cl NH2 Ph CH2 2,6-Dimethyl-3-methoxypyridin-4-yl H 455 Cl NH2 Ph CH2 2,6-Dimethyl-pyridin-4-yl H 456 Cl NH2 Ph CH2 2,3,6-Trimethyl-pyridin-4-yl H 457 Cl NH2 Ph CH2 2,3,6-Trimethoxy-pyridin-4-yl H 458 Cl NH2 Ph CH2 2,6-Dimethyl-3-bromopyridin-4-yl H 459 Cl NH2 Ph CH2 2,6-Dimethyl-3-chloropyridin-4-yl H 460 Cl NH2 Ph CH2 4,6-Dimethyl-5-iodopyridin-3-yl H 461 Cl NH2 Ph CH2 3,5-Dimethyl-4-aminopyridin-2-yl H 462 Cl NH2 3-Py CH2 3,5-Dimethyl-4-methoxypyridin-2-yl H 463 Cl NH2 3-Py CH2 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl H 464 Cl NH2 3-Py CH2 3,5-Dimethyl-4-bromopyridin-2-yl H 465 Cl NH2 3-Py CH2 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl H 466 Cl NH2 3-Py CH2 3,5-Dimethyl-4-chloropyridin-2-yl H 467 Cl NH2 3-Py CH2 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl H 468 Cl NH2 3-Py CH2 3,5-Dimethyl-4-iodopyridin-2-yl H 469 Cl NH2 3-Py CH2 3,5-Dimethyl-4-iodo-1-oxypyridin-2-yl H 470 Cl NH2 3-Py CH2 3,5-Dimethyl-4-thiomethyl-pyridin-2-yl H 471 Cl NH2 3-Py CH2 3,5-Dimethyl-4-thiomethyl-1-oxypyridin-2-yl H 472 Cl NH2 3-Py CH2 3,4,5-Trimethyl-pyridin-2-yl H 473 Cl NH2 3-Py CH2 3,4,5-Trimethyl-1-oxypyridin-2-yl H 474 Cl NH2 3-Py CH2 4,5,6-Trimethoxypyridin-2-yl H 475 Cl NH2 3-Py CH2 4,5,6-Trimethoxy-1-oxypyridin-2-yl H 476 Cl NH2 3-Py CH2 3,4,5-Trimethoxy-pyridin-2-yl H 477 Cl NH2 3-Py CH2 3,4,5-Trimethoxy-1-oxypyridin-2-yl H 478 Cl NH2 3-Py CH2 4,5,6-Trimethyl-pyridin-2-yl H 479 Cl NH2 3-Py CH2 4,5,6-Trimethyl-1-oxypyridin-2-yl H 480 Cl NH2 3-Py CH2 4,6-Dimethyl-5-methoxy-pyridin-2-yl H 481 Cl NH2 3-Py CH2 4,6-Dimethyl-5-methoxypyridin-3-yl H 482 Cl NH2 3-Py CH2 4,6-Dimethyl-5-methoxy-1-oxypyridin-3-yl H 483 Cl NH2 3-Py CH2 4,6-Dimethyl-5-bromopyridin-3-yl H 484 Cl NH2 3-Py CH2 4,6-Dimethyl-5-chloropyridin-3-yl H 485 Cl NH2 3-Py CH2 5,6-Dimethyl-4-bromopyridin-3-yl H 486 Cl NH2 3-Py CH2 5,6-Dimethyl-4-chloropyridin-3-yl H 487 Cl NH2 3-Py CH2 2,6-Dimethyl-3-methoxypyridin-4-yl H 488 Cl NH2 3-Py CH2 2,6-Dimethyl-pyridin-4-yl H 489 Cl NH2 3-Py CH2 2,3,6-Trimethyl-pyridin-4-yl H 490 Cl NH2 3-Py CH2 2,3,6-Trimethoxy-pyridin-4-yl H 491 Cl NH2 3-Py CH2 2,6-Dimethyl-3-bromopyridin-4-yl H 492 Cl NH2 3-Py CH2 2,6-Dimethyl-3-chloropyridin-4-yl H 493 Cl NH2 3-Py CH2 4,6-Dimethyl-5-iodopyridin-3-yl H 494 Cl NH2 3-Py CH2 3,5-Dimethyl-4-aminopyridin-2-yl H 495 Cl NH2 CH2—NMe2 CH2 3,5-Dimethyl-4-methoxypyridin-2-yl H 496 Cl NH2 CH2—NMe2 CH2 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl H 497 Cl NH2 CH2—NMe2 CH2 3,5-Dimethyl-4-bromopyridin-2-yl H 498 Cl NH2 CH2—NMe2 CH2 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl H 499 Cl NH2 CH2—NMe2 CH2 3,5-Dimethyl-4-chloropyridin-2-yl H 500 Cl NH2 CH2—NMe2 CH2 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl H 501 Cl NH2 CH2—NMe2 CH2 3,5-Dimethyl-4-iodopyridin-2-yl H 502 Cl NH2 CH2—NMe2 CH2 3,5-Dimethyl-4-iodo-1-oxypyridin-2-yl H 503 Cl NH2 CH2—NMe2 CH2 3,5-Dimethyl-4-thiomethyl-pyridin-2-yl H 504 Cl NH2 CH2—NMe2 CH2 3,5-Dimethyl-4-thiomethyl-1-oxypyridin-2-yl H 505 Cl NH2 CH2—NMe2 CH2 3,4,5-Trimethyl-pyridin-2-yl H 506 Cl NH2 CH2—NMe2 CH2 3,4,5-Trimethyl-pyridin-2-yl H 507 Cl NH2 CH2—NMe2 CH2 4,5,6-Trimethoxypyridin-2-yl H 508 Cl NH2 CH2—NMe2 CH2 4,5,6-Trimethoxy-1-oxypyridin-2-yl H 509 Cl NH2 CH2—NMe2 CH2 3,4,5-Trimethoxy-pyridin-2-yl H 510 Cl NH2 CH2—NMe2 CH2 3,4,5-Trimethoxy-1-oxypyridin-2-yl H 511 Cl NH2 CH2—NMe2 CH2 4,5,6-Trimethyl-pyridin-2-yl H 512 Cl NH2 CH2—NMe2 CH2 4,5,6-Trimethyl-1-oxypyridin-2-yl H 513 Cl NH2 CH2—NMe2 CH2 4,6-Dimethyl-5-methoxy-pyridin-2-yl H 514 Cl NH2 CH2—NMe2 CH2 4,6-Dimethyl-5-methoxypyridin-3-yl H 515 Cl NH2 CH2—NMe2 CH2 4,6-Dimethyl-5-methoxy-1-oxypyridin-3-yl H 516 Cl NH2 CH2—NMe2 CH2 4,6-Dimethyl-5-bromopyridin-3-yl H 517 Cl NH2 CH2—NMe2 CH2 4,6-Dimethyl-5-chloropyridin-3-yl H 518 Cl NH2 CH2—NMe2 CH2 5,6-Dimethyl-4-bromopyridin-3-yl H 519 Cl NH2 CH2—NMe2 CH2 5,6-Dimethyl-4-chloropyridin-3-yl H 520 Cl NH2 CH2—NMe2 CH2 2,6-Dimethyl-3-methoxypyridin-4-yl H 521 Cl NH2 CH2—NMe2 CH2 2,6-Dimethyl-pyridin-4-yl H 522 Cl NH2 CH2—NMe2 CH2 2,3,6-Trimethyl-pyridin-4-yl H 523 Cl NH2 CH2—NMe2 CH2 2,3,6-Trimethoxy-pyridin-4-yl H 524 Cl NH2 CH2—NMe2 CH2 2,6-Dimethyl-3-bromopyridin-4-yl H 525 Cl NH2 CH2—NMe2 CH2 2,6-Dimethyl-3-chloropyridin-4-yl H 526 Cl NH2 CH2—NMe2 CH2 4,6-Dimethyl-5-iodopyridin-3-yl H 527 Cl NH2 CH2—NMe2 CH2 3,5-Dimethyl-4-aminopyridin-2-yl H 528 Cl NH2 2-furanyl CH2 3,5-Dimethyl-4-methoxypyridin-2-yl H 529 Cl NH2 2-furanyl CH2 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl H 530 Cl NH2 2-furanyl CH2 3,5-Dimethyl-4-bromopyridin-2-yl H 531 Cl NH2 2-furanyl CH2 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl H 532 Cl NH2 2-furanyl CH2 3,5-Dimethyl-4-chloropyridin-2-yl H 533 Cl NH2 2-furanyl CH2 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl H 534 Cl NH2 2-furanyl CH2 3,5-Dimethyl-4-iodopyridin-2-yl H 535 Cl NH2 2-furanyl CH2 3,5-Dimethyl-4-iodo-1-oxypyridin-2-yl H 536 Cl NH2 2-furanyl CH2 3,5-Dimethyl-4-thiomethyl-pyridin-2-yl H 537 Cl NH2 2-furanyl CH2 3,5-Dimethyl-4-thiomethyl-1-oxypyridin-2-yl H 538 Cl NH2 2-furanyl CH2 3,4,5-Trimethyl-pyridin-2-yl H 539 Cl NH2 2-furanyl CH2 3,4,5-Trimethyl-1-oxypyridin-2-yl H 540 Cl NH2 2-furanyl CH2 4,5,6-Trimethoxypyridin-2-yl H 541 Cl NH2 2-furanyl CH2 4,5,6-Trimethoxy-1-oxypyridin-2-yl H 542 Cl NH2 2-furanyl CH2 3,4,5-Trimethoxy-pyridin-2-yl H 543 Cl NH2 2-furanyl CH2 3,4,5-Trimethoxy-1-oxypyridin-2-yl H 544 Cl NH2 2-furanyl CH2 4,5,6-Trimethyl-pyridin-2-yl H 545 Cl NH2 2-furanyl CH2 4,5,6-Trimethyl-1-oxypyridin-2-yl H 546 Cl NH2 2-furanyl CH2 4,6-Dimethyl-5-methoxy-pyridin-2-yl H 547 Cl NH2 2-furanyl CH2 4,6-Dimethyl-5-methoxypyridin-3-yl H 548 Cl NH2 2-furanyl CH2 4,6-Dimethyl-5-methoxy-1-oxypyridin-3-yl H 549 Cl NH2 2-furanyl CH2 4,6-Dimethyl-5-bromopyridin-3-yl H 550 Cl NH2 2-furanyl CH2 4,6-Dimethyl-5-chloropyridin-3-yl H 551 Cl NH2 2-furanyl CH2 5,6-Dimethyl-4-bromopyridin-3-yl H 552 Cl NH2 2-furanyl CH2 5,6-Dimethyl-4-chloropyridin-3-yl H 553 Cl NH2 2-furanyl CH2 2,6-Dimethyl-3-methoxypyridin-4-yl H 554 Cl NH2 2-furanyl CH2 2,6-Dimethyl-pyridin-4-yl H 555 Cl NH2 2-furanyl CH2 2,3,6-Trimethyl-pyridin-4-yl H 556 Cl NH2 2-furanyl CH2 2,3,6-Trimethoxy-pyridin-4-yl H 557 Cl NH2 2-furanyl CH2 2,6-Dimethyl-3-bromopyridin-4-yl H 558 Cl NH2 2-furanyl CH2 2,6-Dimethyl-3-chloropyridin-4-yl H 559 Cl NH2 2-furanyl CH2 3,5-Dimethyl-4-aminopyridin-2-yl H 560 Cl NH2 Cl CH2 3,5-Dimethyl-4-methoxypyridin-2-yl H 561 Cl NH2 Cl CH2 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl H 562 Cl NH2 Cl CH2 3,5-Dimethyl-4-bromopyridin-2-yl H 563 Cl NH2 Cl CH2 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl H 564 Cl NH2 Cl CH2 3,5-Dimethyl-4-chloropyridin-2-yl H 565 Cl NH2 Cl CH2 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl H 566 Cl NH2 Cl CH2 3,5-Dimethyl-4-iodopyridin-2-yl H 567 Cl NH2 Cl CH2 3,5-Dimethyl-4-iodo-1-oxypyridin-2-yl H 568 Cl NH2 Cl CH2 3,5-Dimethyl-4-thiomethyl-pyridin-2-yl H 569 Cl NH2 Cl CH2 3,5-Dimethyl-4-thiomethyl-1-oxypyridin-2-yl H 570 Cl NH2 Cl CH2 3,4,5-Trimethyl-pyridin-2-yl H 571 Cl NH2 Cl CH2 3,4,5-Trimethyl-1-oxypyridin-2-yl H 572 Cl NH2 Cl CH2 4,6-Dimethyl-5-methoxypyridin-3-yl H 573 Cl NH2 Cl CH2 4,6-Dimethyl-5-methoxy-1-oxypyridin-3-yl H 574 Cl NH2 Cl CH2 3,5-Dimethyl-4-aminopyridin-2-yl H 575 Cl NH2 Br CH2 3,5-Dimethyl-4-methoxypyridin-2-yl H 576 Cl NH2 Br CH2 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl H 577 Cl NH2 Br CH2 3,5-Dimethyl-4-bromopyridin-2-yl H 578 Br NH2 Br CH2 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl H 579 Cl NH2 Br CH2 3,5-Dimethyl-4-chloropyridin-2-yl H 580 Br NH2 Br CH2 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl H 581 Cl NH2 Br CH2 3,5-Dimethyl-4-iodopyridin-2-yl H 582 Br NH2 Br CH2 3,5-Dimethyl-4-iodo-1-oxypyridin-2-yl H 583 Cl NH2 Br CH2 3,5-Dimethyl-4-thiomethyl-pyridin-2-yl H 584 Br NH2 Br CH2 3,5-Dimethyl-4-thiomethyl-1-oxypyridin-2-yl H 585 Cl NH2 Br CH2 3,4,5-Trimethyl-pyridin-2-yl H 586 Br NH2 Br CH2 3,4,5-Trimethyl-1-oxypyridin-2-yl H 587 Cl NH2 Br CH2 4,6-Dimethyl-5-methoxypyridin-3-yl H 588 Cl NH2 Br CH2 4,6-Dimethyl-5-methoxy-1-oxypyridin-3-yl H 589 Cl NH2 Br CH2 3,5-Dimethyl-4-aminopyridin-2-yl H 590 Cl NH2 I CH2 3,5-Dimethyl-4-methoxypyridin-2-yl H 591 Cl NH2 I CH2 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl H 592 Cl NH2 I CH2 3,5-Dimethyl-4-bromopyridin-2-yl H 593 Cl NH2 I CH2 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl H 594 Cl NH2 I CH2 3,5-Dimethyl-4-chloropyridin-2-yl H 595 Cl NH2 I CH2 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl H 596 Cl NH2 I CH2 3,5-Dimethyl-4-iodopyridin-2-yl H 597 Cl NH2 I CH2 3,5-Dimethyl-4-iodo-1-oxypyridin-2-yl H 598 Cl NH2 I CH2 3,5-Dimethyl-4-thiomethyl-pyridin-2-yl H 599 Cl NH2 I CH2 3,5-Dimethyl-4-thiomethyl-1-oxypyridin-2-yl H 600 Cl NH2 I CH2 3,4,5-Trimethyl-pyridin-2-yl H 601 Cl NH2 I CH2 3,4,5-Trimethyl-1-oxypyridin-2-yl H 602 Cl NH2 I CH2 4,6-Dimethyl-5-methoxypyridin-3-yl H 603 Cl NH2 I CH2 4,6-Dimethyl-5-methoxy-1-oxypyridin-3-yl H 604 Cl NH2 I CH2 3,5-Dimethyl-4-aminopyridin-2-yl H 605 Cl NH2 CN CH2 3,5-Dimethyl-4-methoxypyridin-2-yl H 606 Cl NH2 CN CH2 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl H 607 Cl NH2 CN CH2 3,5-Dimethyl-4-bromopyridin-2-yl H 608 Cl NH2 CN CH2 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl H 609 Cl NH2 CN CH2 3,5-Dimethyl-4-chloropyridin-2-yl H 610 Cl NH2 CN CH2 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl H 611 Cl NH2 CN CH2 3,5-Dimethyl-4-iodopyridin-2-yl H 612 Cl NH2 CN CH2 3,5-Dimethyl-4-iodo-1-oxypyridin-2-yl H 613 Cl NH2 CN CH2 3,5-Dimethyl-4-thiomethyl-pyridin-2-yl H 614 Cl NH2 CN CH2 3,5-Dimethyl-4-thiomethyl-1-oxypyridin-2-yl H 615 Cl NH2 CN CH2 3,4,5-Trimethyl-pyridin-2-yl H 616 Cl NH2 CN CH2 3,4,5-Trimethyl-1-oxypyridin-2-yl H 617 Cl NH2 CN CH2 4,6-Dimethyl-5-methoxypyridin-3-yl H 618 Cl NH2 CN CH2 4,6-Dimethyl-5-methoxy-1-oxypyridin-3-yl H 619 Cl NH2 CN CH2 3,5-Dimethyl-4-aminopyridin-2-yl H 620 Cl NH2 H C(O) 3,5-Dimethyl-4-methoxypyridin-2-yl H 621 Cl NH2 H C(O) 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl H 622 Cl NH2 H C(O) 3,5-Dimethyl-4-bromopyridin-2-yl H 623 Cl NH2 H C(O) 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl H 624 Cl NH2 H C(O) 3,5-Dimethyl-4-chloropyridin-2-yl H 625 Cl NH2 H C(O) 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl H 626 Cl NH2 H C(O) 3,5-Dimethyl-4-iodopyridin-2-yl H 627 Cl NH2 H C(O) 3,5-Dimethyl-4-iodo-1-oxypyridin-2-yl H 628 Cl NH2 H C(O) 3,5-Dimethyl-4-thiomethyl-pyridin-2-yl H 629 Cl NH2 H C(O) 3,5-Dimethyl-4-thiomethyl-1-oxypyridin-2-yl H 630 Cl NH2 H C(O) 3,4,5-Trimethyl-pyridin-2-yl H 631 Cl NH2 H C(O) 3,4,5-Trimethyl-1-oxypyridin-2-yl H 632 Cl NH2 H C(O) 4,6-Dimethyl-5-methoxypyridin-3-yl H 633 Cl NH2 H C(O) 4,6-Dimethyl-5-methoxy-1-oxypyridin-3-yl H 634 Cl NH2 H C(O) 3,5-Dimethyl-4-aminopyridin-2-yl H 635 Cl NH2 H S(O) 3,5-Dimethyl-4-methoxypyridin-2-yl H 636 Cl NH2 H S(O) 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl H 637 Cl NH2 H S(O) 3,5-Dimethyl-4-bromopyridin-2-yl H 638 Cl NH2 H S(O) 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl H 639 Cl NH2 H S(O) 3,5-Dimethyl-4-chloropyridin-2-yl H 640 Cl NH2 H S(O) 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl H 641 Cl NH2 H S(O) 3,5-Dimethyl-4-iodopyridin-2-yl H 642 Cl NH2 H S(O) 3,5-Dimethyl-4-iodo-1-oxypyridin-2-yl H 643 Cl NH2 H S(O) 3,5-Dimethyl-4-thiomethyl-pyridin-2-yl H 644 Cl NH2 H S(O) 3,5-Dimethyl-4-thiomethyl-1-oxypyridin-2-yl H 645 Cl NH2 Br S(O) 3,4,5-Trimethyl-pyridin-2-yl H 646 Cl NH2 H S(O) 3,4,5-Trimethyl-1-oxypyridin-2-yl H 647 Cl NH2 Br S(O) 4,6-Dimethyl-5-methoxypyridin-3-yl H 648 Cl NH2 H S(O) 4,6-Dimethyl-5-methoxy-1-oxypyridin-3-yl H 649 Cl NH2 H SO2 3,5-Dimethyl-4-methoxypyridin-2-yl H 650 Cl NH2 H SO2 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl H 651 Cl NH2 H SO2 3,5-Dimethyl-4-bromopyridin-2-yl H 652 Cl NH2 H SO2 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl H 653 Cl NH2 Br SO2 3,5-Dimethyl-4-chloropyridin-2-yl H 654 Cl NH2 H SO2 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl H 655 Cl NH2 H SO2 3,5-Dimethyl-4-iodopyridin-2-yl H 656 Cl NH2 H SO2 3,5-Dimethyl-4-iodo-1-oxypyridin-2-yl H 657 Cl NH2 H SO2 3,5-Dimethyl-4-thiomethyl-pyridin-2-yl H 658 Cl NH2 H SO2 3,5-Dimethyl-4-thiomethyl-1-oxypyridin-2-yl H 659 Cl NH2 H SO2 3,4,5-Trimethyl-pyridin-2-yl H 660 Cl NH2 H SO2 3,4,5-Trimethyl-1-oxypyridin-2-yl H 661 Cl NH2 H SO2 4,6-Dimethyl-5-methoxypyridin-3-yl H 662 Cl NH2 H SO2 4,6-Dimethyl-5-methoxy-1-oxypyridin-3-yl H 663 Cl NH2 i-pr C(O) 3,5-Dimethyl-4-methoxypyridin-2-yl H 664 Cl NH2 i-pr C(O) 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl H 665 Cl NH2 i-pr C(O) 3,5-Dimethyl-4-bromopyridin-2-yl H 666 Cl NH2 i-pr C(O) 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl H 667 Cl NH2 i-pr C(O) 3,5-Dimethyl-4-chloropyridin-2-yl H 668 Cl NH2 i-pr C(O) 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl H 669 Cl NH2 i-pr C(O) 3,5-Dimethyl-4-iodopyridin-2-yl H 670 Cl NH2 i-pr C(O) 3,5-Dimethyl-4-iodo-1-oxypyridin-2-yl H 671 Cl NH2 i-pr C(O) 3,5-Dimethyl-4-thiomethyl-pyridin-2-yl H 672 Cl NH2 i-pr C(O) 3,5-Dimethyl-4-thiomethyl-1-oxypyridin-2-yl H 673 Cl NH2 i-pr C(O) 3,4,5-Trimethyl-pyridin-2-yl H 674 Cl NH2 i-pr C(O) 3,4,5-Trimethyl-1-oxypyridin-2-yl H 675 Cl NH2 i-pr C(O) 4,6-Dimethyl-5-methoxypyridin-3-yl H 676 Cl NH2 i-pr C(O) 4,6-Dimethyl-5-methoxy-1-oxypyridin-3-yl H 677 Cl NH2 i-pr C(O) 3,5-Dimethyl-4-aminopyridin-2-yl H 678 Cl NH2 i-pr S(O) 3,5-Dimethyl-4-methoxypyridin-2-yl H 679 Cl NH2 i-pr S(O) 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl H 680 Cl NH2 i-pr S(O) 3,5-Dimethyl-4-bromopyridin-2-yl H 681 Cl NH2 i-pr S(O) 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl H 682 Cl NH2 i-pr S(O) 3,5-Dimethyl-4-chloropyridin-2-yl H 683 Cl NH2 i-pr S(O) 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl H 684 Cl NH2 i-pr S(O) 3,5-Dimethyl-4-iodopyridin-2-yl H 685 Cl NH2 i-pr S(O) 3,5-Dimethyl-4-iodo-1-oxypyridin-2-yl H 686 Cl NH2 i-pr S(O) 3,5-Dimethyl-4-thiomethyl-pyridin-2-yl H 687 Cl NH2 i-pr S(O) 3,5-Dimethyl-4-thiomethyl-1-oxypyridin-2-yl H 688 Cl NH2 i-pr S(O) 3,4,5-Trimethyl-pyridin-2-yl H 689 Cl NH2 i-pr S(O) 3,4,5-Trimethyl-1-oxypyridin-2-yl H 690 Cl NH2 i-pr S(O) 4,6-Dimethyl-5-methoxypyridin-3-yl H 691 Cl NH2 i-pr S(O) 4,6-Dimethyl-5-methoxy-1-oxypyridin-3-yl H 692 Cl NH2 i-pr SO2 3,5-Dimethyl-4-methoxypyridin-2-yl H 693 Cl NH2 i-pr SO2 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl H 694 Cl NH2 i-pr SO2 3,5-Dimethyl-4-bromopyridin-2-yl H 695 Cl NH2 i-pr SO2 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl H 696 Cl NH2 i-pr SO2 3,5-Dimethyl-4-chloropyridin-2-yl H 697 Cl NH2 i-pr SO2 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl H 698 Cl NH2 i-pr SO2 3,5-Dimethyl-4-iodopyridin-2-yl H 699 Cl NH2 i-pr SO2 3,5-Dimethyl-4-iodo-1-oxypyridin-2-yl H 700 Cl NH2 i-pr SO2 3,5-Dimethyl-4-thiomethyl-pyridin-2-yl H 701 Cl NH2 i-pr SO2 3,5-Dimethyl-4-thiomethyl-1-oxypyridin-2-yl H 702 Cl NH2 i-pr SO2 3,4,5-Trimethyl-pyridin-2-yl H 703 Cl NH2 i-pr SO2 3,4,5-Trimethyl-1-oxypyridin-2-yl H 704 Cl NH2 i-pr SO2 4,6-Dimethyl-5-methoxypyridin-3-yl H 705 Cl NH2 i-pr SO2 4,6-Dimethyl-5-methoxy-1-oxypyridin-3-yl H 706 Cl NH2 H C(O) 3,4,5-Trimethoxyphenyl H 707 Cl NH2 H C(O) 2-Chloro-3,4,5-trimethoxyphenyl H 708 Cl NH2 H C(O) 2-Bromo-3,4,5-trimethoxyphenyl H 709 Cl NH2 H C(O) 3,5-Dimethyl-4-methoxyphenyl H 710 Cl NH2 H C(O) 2-Chloro-3,5-Dimethyl-4-methoxyphenyl H 711 Cl NH2 H C(O) 2-Bromo-3,5-Dimethyl-4-methoxyphenyl H 712 Cl NH2 H SO2 3,4,5-Trimethoxyphenyl H 713 Cl NH2 H SO2 2-Chloro-3,4,5-trimethoxyphenyl H 714 Cl NH2 H SO2 2-Bromo-3,4,5-trimethoxyphenyl H 715 Cl NH2 H SO2 3,5-Dimethyl-4-methoxyphenyl H 716 Cl NH2 H SO2 2-Chloro-3,5-Dimethyl-4-methoxyphenyl H 717 Cl NH2 H SO2 2-Bromo-3,5-Dimethyl-4-methoxyphenyl H 718 Cl NH2 H CH2 3,5-Dimethyl-4-methoxypyridin-2-yl Br 719 Cl NH2 H CH2 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl Br 720 Cl NH2 H CH2 3,5-Dimethyl-4-bromopyridin-2-yl Br 721 Cl NH2 H CH2 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl Br 722 Cl NH2 H CH2 3,5-Dimethyl-4-chloropyridin-2-yl Br 723 Cl NH2 H CH2 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl Br 724 Cl NH2 H CH2 3,5-Dimethyl-4-iodopyridin-2-yl Br 725 Cl NH2 H CH2 3,5-Dimethyl-4-iodo-1-oxypyridin-2-yl Br 726 Cl NH2 H CH2 3,5-Dimethyl-4-thiomethyl-pyridin-2-yl Br 727 Cl NH2 H CH2 3,5-Dimethyl-4-thiomethyl-1-oxypyridin-2-yl Br 728 Cl NH2 H CH2 3,4,5-Trimethyl-pyridin-2-yl Br 729 Cl NH2 H CH2 3,4,5-Trimethyl-1-oxypyridin-2-yl Br 730 Cl NH2 H CH2 4,6-Dimethyl-5-methoxypyridin-3-yl Br 731 Cl NH2 H CH2 4,6-Dimethyl-5-methoxy-1-oxypyridin-3-yl Br 732 Cl NH2 H CH2 3,5-Dimethyl-4-methoxypyridin-2-yl Cl 733 Cl NH2 H CH2 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl Cl 734 Cl NH2 H CH2 3,5-Dimethyl-4-bromopyridin-2-yl Cl 735 Cl NH2 H CH2 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl Cl 736 Cl NH2 H CH2 3,5-Dimethyl-4-chloropyridin-2-yl Cl 737 Cl NH2 H CH2 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl Cl 738 Cl NH2 H CH2 3,5-Dimethyl-4-iodopyridin-2-yl Cl 739 Cl NH2 H CH2 3,5-Dimethyl-4-iodo-1-oxypyridin-2-yl Cl 740 Cl NH2 H CH2 3,5-Dimethyl-4-thiomethyl-pyridin-2-yl Cl 741 Cl NH2 H CH2 3,5-Dimethyl-4-thiomethyl-1-oxypyridin-2-yl Cl 742 Cl NH2 H CH2 3,4,5-Trimethyl-pyridin-2-yl Cl 743 Cl NH2 H CH2 3,4,5-Trimethyl-1-oxypyridin-2-yl Cl 744 Cl NH2 H CH2 4,6-Dimethyl-5-methoxypyridin-3-yl Cl 745 Cl NH2 H CH2 4,6-Dimethyl-5-methoxy-1-oxypyridin-3-yl Cl 746 Cl NH2 H CH2 3,5-Dimethyl-4-methoxypyridin-2-yl CN 747 Cl NH2 H CH2 3, 5-Dimethyl-4-methoxy- 1 -oxypyridin-2-yl CN 748 Cl NH2 H CH2 3,5-Dimethyl-4-bromopyridin-2-yl CN 749 Cl NH2 H CH2 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl CN 750 Cl NH2 H CH2 3,5-Dimethyl-4-chloropyridin-2-yl CN 751 Cl NH2 H CH2 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl CN 752 Cl NH2 H CH2 3,5-Dimethyl-4-iodopyridin-2-yl CN 753 25 Br NH2 H CH2 3,5-Dimethyl-4-methoxypyridin-2-yl H 754 20 Br NH2 H CH2 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl H 755 Br NH2 H CH2 6-Bromo-3,5-dimethyl-4-methoxypyridin-2-yl H 756 Br NH2 H CH2 6-Chloro-3,5-dimethyl-4-methoxypyridin-2-yl H 757 23 Br NH2 H CH2 3,5-Dimethyl-4-bromopyridin-2-yl H 758 24 Br NH2 H CH2 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl H 759 21 Br NH2 H CH2 3,5-Dimethyl-4-chloropyridin-2-yl H 760 22 Br NH2 H CH2 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl H 761 Br NH2 H CH2 3,5-Dimethyl-4-iodopyridin-2-yl H 762 Br NH2 H CH2 3,5-Dimethyl-4-iodo-1-oxypyridin-2-yl H 763 Br NH2 H CH2 3,5-Dimethyl-4-thiomethyl-pyridin-2-yl H 764 Br NH2 H CH2 3,5-Dimethyl-4-thiomethyl-1-oxypyridin-2-yl H 765 Br NH2 H CH2 3,4,5-Trimethyl-pyridin-2-yl H 766 Br NH2 H CH2 3,4,5-Trimethyl-1-oxypyridin-2-yl H 767 Br NH2 H CH2 4,5,6-Trimethoxypyridin-2-yl H 768 Br NH2 H CH2 4,5,6-Trimethoxy-1-oxypyridin-2-yl H 769 Br NH2 H CH2 3-Bromo-4,5,6-trimethoxypyridin-2-yl H 770 Br NH2 H CH2 3-Chloro-4,5,6-trimethoxypyridin-2-yl H 771 Br NH2 H CH2 3,4,5-Trimethoxy-pyridin-2-yl H 772 Br NH2 H CH2 3,4,5-Trimethoxy-1-oxypyridin-2-yl H 773 Br NH2 H CH2 3-Bromo-3,4,5-trimethoxy-pyridin-2-yl H 774 Br NH2 H CH2 3-Chloro-3,4,5-trimethoxy-pyridin-2-yl H 775 Br NH2 H CH2 4,5,6-Trimethyl-pyridin-2-yl H 776 Br NH2 H CH2 4,5,6-Trimethyl-1-oxypyridin-2-yl H 777 Br NH2 H CH2 4,6-Dimethyl-5-methoxy-pyridin-2-yl H 778 Br NH2 H CH2 4,6-Dimethyl-5-methoxypyridin-3-yl H 779 Br NH2 H CH2 4,6-Dimethyl-5-methoxy-1-oxypyridin-3-yl H 780 Br NH2 H CH2 4,6-Dimethyl-5-bromopyridin-3-yl H 781 Br NH2 H CH2 4,6-Dimethyl-5-chloropyridin-3-yl H 782 Br NH2 H CH2 5,6-Dimethyl-4-bromopyridin-3-yl H 783 Br NH2 H CH2 5,6-Dimethyl-4-chloropyridin-3-yl H 784 Br NH2 H CH2 2,6-Dimethyl-3-methoxypyridin-4-yl H 785 Br NH2 H CH2 2,6-Dimethyl-pyridin-4-yl H 786 Br NH2 H CH2 2,3,6-Trimethyl-pyridin-4-yl H 787 Br NH2 H CH2 2,3,6-Trimethoxy-pyridin-4-yl H 788 Br NH2 H CH2 2,6-Dimethyl-3-bromopyridin-4-yl H 789 Br NH2 H CH2 2,6-Dimethyl-3-chloropyridin-4-yl H 790 Br NH2 H CH2 2,6-Dimethyl-3-methoxy-1-oxy-pyridin-4-yl H 791 Br NH2 H CH2 2,6-Dimethyl-1-oxy-pyridin-4-yl H 792 Br NH2 H CH2 2,3,6-Trimethyl-1-oxy-pyridin-4-yl H 793 Br NH2 H CH2 2,3,6-Trimethoxy-1-oxy-pyridin-4-yl H 794 Br NH2 H CH2 2,6-Dimethyl-3-bromo1-oxy-pyridin-4-yl H 795 Br NH2 H CH2 2,6-Dimethyl-3-chloro1-oxy-pyridin-4-yl H 796 Br NH2 H CH2 4,6-Dimethyl-5-iodopyridin-3-yl H 797 Br NH2 H CH2 3,5-Dimethyl-4-aminopyridin-2-yl H 798 Br NH2 i-pr CH2 3,5-Dimethyl-4-methoxypyridin-2-yl H 799 Br NH2 i-pr CH2 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl H 800 Br NH2 i-pr CH2 6-Bromo-3,5-dimethyl-4-methoxypyridin-2-yl H 801 Br NH2 i-pr CH2 6-Chloro-3,5-dimethyl-4-methoxypyridin-2-yl H 802 Br NH2 i-pr CH2 3,5-Dimethyl-4-bromopyridin-2-yl H 803 Br NH2 i-pr CH2 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl H 804 Br NH2 i-pr CH2 3,5-Dimethyl-4-chloropyridin-2-yl H 805 Br NH2 i-pr CH2 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl H 806 Br NH2 i-pr CH2 3,5-Dimethyl-4-iodopyridin-2-yl H 807 Br NH2 i-pr CH2 3,5-Dimethyl-4-iodo-1-oxypyridin-2-yl H 808 Br NH2 i-pr CH2 3,5-Dimethyl-4-thiomethyl-pyridin-2-yl H 809 Br NH2 i-pr CH2 3,5-Dimethyl-4-thiomethyl-1-oxypyridin-2-yl H 810 Br NH2 i-pr CH2 3,4,5-Trimethyl-pyridin-2-yl H 811 Br NH2 i-pr CH2 3,4,5-Trimethyl-1-oxypyridin-2-yl H 812 Br NH2 i-pr CH2 4,5,6-Trimethoxypyridin-2-yl H 813 Br NH2 i-pr CH2 4,5,6-Trimethoxy-1-oxypyridin-2-yl H 814 Br NH2 i-pr CH2 3-Bromo-4,5,6-trimethoxypyridin-2-yl H 815 Br NH2 i-pr CH2 3-Chloro-4,5,6-trimethoxypyridin-2-yl H 816 Br NH2 i-pr CH2 3,4,5-Trimethoxy-pyridin-2-yl H 817 Br NH2 i-pr CH2 3,4,5-Trimethoxy-1-oxypyridin-2-yl H 818 Br NH2 i-pr CH2 3-Bromo-3,4,5-trimethoxy-pyridin-2-yl H 819 Br NH2 i-pr CH2 3-Chloro-3,4,5-trimethoxy-pyridin-2-yl H 820 Br NH2 i-pr CH2 4,5,6-Trimethyl-pyridin-2-yl H 821 Br NH2 i-pr CH2 4,5,6-Trimethyl-1-oxypyridin-2-yl H 822 Br NH2 i-pr CH2 4,6-Dimethyl-5-methoxy-pyridin-2-yl H 823 Br NH2 i-pr CH2 4,6-Dimethyl-5-methoxypyridin-3-yl H 824 Br NH2 i-pr CH2 4,6-Dimethyl-5-methoxy-1-oxypyridin-3-yl H 825 Br NH2 i-pr CH2 4,6-Dimethyl-5-bromopyridin-3-yl H 826 Br NH2 i-pr CH2 4,6-Dimethyl-5-chloropyridin-3-yl H 827 Br NH2 i-pr CH2 5,6-Dimethyl-4-bromopyridin-3-yl H 828 Br NH2 i-pr CH2 5,6-Dimethyl-4-chloropyridin-3-yl H 829 Br NH2 i-pr CH2 2,6-Dimethyl-3-methoxypyridin-4-yl H 830 Br NH2 i-pr CH2 2,6-Dimethyl-pyridin-4-yl H 831 Br NH2 i-pr CH2 2,3,6-Trimethyl-pyridin-4-yl H 832 Br NH2 i-pr CH2 2,3,6-Trimethoxy-pyridin-4-yl H 833 Br NH2 i-pr CH2 2,6-Dimethyl-3-bromopyridin-4-yl H 834 Br NH2 i-pr CH2 2,6-Dimethyl-3-chloropyridin-4-yl H 835 Br NH2 i-pr CH2 2,6-Dimethyl-3-methoxy-1-oxypyridin-4-yl H 836 Br NH2 i-pr CH2 2,6-Dimethyl-1-oxy-pyridin-4-yl H 837 Br NH2 i-pr CH2 2,3,6-Trimethyl-1-oxypyridin-4-yl H 838 Br NH2 i-pr CH2 2,3,6-Trimethoxy-1-oxypyridin-4-yl H 839 Br NH2 i-pr CH2 2,6-Dimethyl-3-bromo1-oxypyridin-4-yl H 840 Br NH2 i-pr CH2 2,6-Dimethyl-3-chloro1-oxypyridin-4-yl H 841 Br NH2 i-pr CH2 4,6-Dimethyl-5-iodopyridin-3-yl H 842 Br NH2 i-pr CH2 3,5-Dimethyl-4-aminopyridin-2-yl H 843 Br NH2 Ph CH2 3,5-Dimethyl-4-methoxypyridin-2-yl H 844 Br NH2 Ph CH2 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl H 845 Br NH2 Ph CH2 6-Bromo-3,5-dimethyl-4-methoxypyridin-2-yl H 846 Br NH2 Ph CH2 6-Chloro-3,5-dimethyl-4-methoxypyridin-2-yl H 847 Br NH2 Ph CH2 3,5-Dimethyl-4-bromopyridin-2-yl H 848 Br NH2 Ph CH2 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl H 849 Br NH2 Ph CH2 3,5-Dimethyl-4-chloropyridin-2-yl H 850 Br NH2 Ph CH2 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl H 851 Br NH2 Ph CH2 3,5-Dimethyl-4-iodopyridin-2-yl H 852 Br NH2 Ph CH2 3,5-Dimethyl-4-iodo-1-oxypyridin-2-yl H 853 Br NH2 Ph CH2 3,5-Dimethyl-4-thiomethyl-pyridin-2-yl H 854 Br NH2 Ph CH2 3,5-Dimethyl-4-thiomethyl-1-oxypyridin-2-yl H 855 Br NH2 Ph CH2 3,4,5-Trimethyl-pyridin-2-yl H 856 Br NH2 Ph CH2 3,4,5-Trimethyl-1-oxypyridin-2-yl H 857 Br NH2 Ph CH2 4,5,6-Trimethoxypyridin-2-yl H 858 Br NH2 Ph CH2 4,5,6-Trimethoxy-1-oxypyridin-2-yl H 859 Br NH2 Ph CH2 3-Bromo-4,5,6-trimethoxypyridin-2-yl H 860 Br NH2 Ph CH2 3-Chloro-4,5,6-trimethoxypyridin-2-yl H 861 Br NH2 Ph CH2 4,6-Dimethyl-5-methoxypyridin-3-yl H 862 Br NH2 Ph CH2 4,6-Dimethyl-5-methoxy-1-oxypyridin-3-yl H 863 Br NH2 Me CH2 3,5-Dimethyl-4-methoxypyridin-2-yl H 864 Br NH2 Me CH2 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl H 865 Br NH2 Me CH2 6-Bromo-3,5-dimethyl-4-methoxypyridin-2-yl H 866 Br NH2 Me CH2 6-Chloro-3,5-dimethyl-4-methoxypyridin-2-yl H 867 Br NH2 Me CH2 3,5-Dimethyl-4-bromopyridin-2-yl H 868 Br NH2 Me CH2 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl H 869 Br NH2 Me CH2 3,5-Dimethyl-4-chloropyridin-2-yl H 870 Br NH2 Me CH2 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl H 871 Br NH2 Me CH2 3,5-Dimethyl-4-iodopyridin-2-yl H 872 Br NH2 Me CH2 3,5-Dimethyl-4-iodo-1-oxypyridin-2-yl H 873 Br NH2 Me CH2 4,6-Dimethyl-5-methoxypyridin-3-yl H 874 Br NH2 Me CH2 4,6-Dimethyl-5-methoxy-1-oxypyridin-3-yl H 875 39 Cl NH2 CH2(Bn)2 CH2 3,5-Dimethyl-4-methoxypyridin-2-yl H 876 12 Cl NH2 H CH2 2-Chloro-4,5-dimethoxylphenyl H 877 16 Cl NH2 H CH2 2-Nitro-4,5-dimethoxylphenyl H 878 17 Cl NH2 H CH2 3,4-Dichlorophenyl H 879 18 Cl NH2 H CH2 3,5-DIMETHOXYLPHENYL H 880 19 Cl NH2 H CH2 2,5-DIMETHOXYLPHENYL H 881 26 Br NH2 H CH2 3,5-DIMETHOXYLPHENYL H 882 27 Cl NH2 H CH2 3-METHOXYLPHENYL H 883 28 Cl NH2 H CH2 4-METHOXYLPIENYL H 884 29 Cl I CH2 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl H 885 30 Cl I CH2 3,5-Dimethyl-4-bromo-1-oxypyridin-2-yl H 886 31 Cl I CH2 3,5-Dimethyl-4-methoxypyridin-2-yl H 887 32 Cl H CH2 3,5-DIMETHYL-4-BROMO-1-OXYPYRIDIN-2-YL H 888 33 Cl H CH2 3,5-Dimethyl-4-methoxypyridin-2-yl H 889 34 Cl H CH2 3,5-Dimethyl-4-methoxy-1-oxypyridin-2-yl H 890 35 Cl H CH2 3,5-Dimethyl-4-chloropyridin-2-yl H 891 36 Cl I CH2 3,5-Dimethyl-4-chloropyridin-2-yl H 892 37 Cl H CH2 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl H 893 38 Cl I CH2 3,5-Dimethyl-4-chloro-1-oxypyridin-2-yl H
Compounds of interest in Table 1 are compounds 2, 3, 17, 18, 27, 28, 62, 63, 77, 78, 92, 93, 129, 130, 238, 239, 242, 243, 245, 246, 247, 248, 249, 250, 251, 252, 253, 267, 268, 287, 288, 291, 292, 293, 294, 295, 296, 297, 298, 312,313, 332,333, 334, 335, 336, 337, 338, 339, 351, 352, 365, 366, 384, 385, 398, 399, 400, 401, 402, 403, 404, 405, 417, 418, 431, 432, 433, 434, 435, 436, 437, 438, 450, 451, 464, 465, 483, 484, 497, 498, 530, 531, 549, 550, 562, 563,574, 575, 577, 578, 589, 590, 592, 593, 604, 605, 607, 608, 619, 620, 755, 756, 759, 760, 761, 762, 763, 764, 765, 766, 780, 781, 800, 801, 804, 805, 806, 807, 808, 809, 810, 811, 825, 826, 845, 846, 863, 864, 865, 866, 875, and 876 with the selected ones being 17, 18, 27, 28, 62, 63, 77, 78, 242, 243, 245, 246, 247, 248, 249, 250, 251, 252, 253, 267, 268, 287, 288, 291, 292, 293, 294, 295, 296, 312,313, 431, 432, 755, 756, 759, 760, 761, 762, 763, 764, 800, and 801.
III. Synthesis of the Compounds of the Invention - The compounds of Formula I of the present invention may be synthesized by various methods known in the art. The general strategy is outlined in Scheme 1 and consists of three parts: (1) constructing the bicyclic system, starting from either a pyridine or a pyrrole, or an acyclic precursor (2) appending the R5-R4-group, and (3) further elaborating the ring systems.
-
-
- 1.1. Assembly of the pyrrolo[2,3-d]pyrimidine starting from a pyrimidine
-
- Method 1.1.1:
- The compounds of Formula 4 can be made by intramolecular cyclization of an aldehyde or ketone, possibly protected, as in Formula 6. (See, J. Davoll, J. Chem. Soc. 1960, 131; J. A. Montgomery, J. Chem. Soc. 1967, 665; G. Cristalli, J. Med. Chem. 1988, 31, 390; T. Miwa, J. Org. Chem. 1993,58, 1696; D. M. Williams, J. Chem. Soc., Perkin Trans 1, 1997, 1171).
- Method 1.1.2
- The compounds of Formula 4, wherein R3 is H, R6 is Cl, and R7 is NH2 can be prepared by treating compounds of Formula 7 wherein R is a halogen or a leaving group with ammonia. Similarly, compounds of Formula I wherein R3 is H, R1 is Cl, R2 is NH2 can be prepared by treating the compound of Formula 7 wherein R is a halogen or leaving group with R5-R4—NH2 in butanol at reflux in presence of a base such as K2CO3, Cs2CO3 or iPrNEt2. (A. B. Reitz J. Med. Chem. 1994, 37, 3561). Compounds of Formula 7 can in turn be prepared as taught by G. W. Craig, J. Prakt. Chem. 2000, 342, 504 and M. Semonsky, Coll. Czech. Chem. Commun. 1980, 45, 3583).
- Method 1.1.3:
- The compounds of Formula 4 can be obtained by treatment of a α-haloketone of Formula 8 wherein X is a halogen with ammonia or a synthetic equivalent thereof.
- Method 1.1.4:
- The compounds of Formula 4 wherein R0 is methyl can be obtained by a tandem Pd-mediated intramolecular cyclization/double-bond migration of alkenes of Formula 9 (S. E. Watson, Synth. Commun. 1998, 28, 3885).
- Method 1.1.5:
- The compounds of Formula 4 wherein R3 is H can be obtained by Pd-mediated intramolecular cyclization of alkynes of Formula 10, wherein Z in as electron-withdrawing group such as, e.g., tosyl-, or —CO2Et.
- Method 1.1.6:
- The compounds of Formula 4 wherein R3 is AcO— can be obtained by intramolecular Friedel-Crafts acylation of precursors of Formula 11 (E. D. Edstrom, J. Org. Chem. 1993, 58, 403).
- Method 1.1.7:
- The compound of Formula 4, wherein R0 is H, R6 is OH, and R7 is NH2, can be prepared by treating the compound of Formula 12 with an α-haloaldehyde of the formula R3—CHX-CHO. See, D. M. Williams, J. Chem. Soc., Perkin Trans 1, 1997, 1171; C. J. Barnett, Org. Proc. Res. Devop. 1999, 3, 184; A. Gangjee, J. Med. Chem. 2001, 44, 1993.
- Method 1.1.8:
- The compounds of Formula 4, wherein R6 is OH and R7 is NH2 can be obtained by treating the compound of Formula 13 with an aldehyde of the formula R3—CHO. See, A. Gangjee, J. Med. Chem. 2003, 46, 591; E. C. Taylor, Heterocycles 1996, 43, 323.
- 1.2: Assembly of the pyrrolo[2,3-d]pyrimidine starting from a pyrrole
-
- Compounds of Formula 2 wherein R13 is —CN and R14 is R—NH—CR7═N— can be cyclized and rearranged to give compounds of Formula 4 where R6 is R—NH—. See, E. C. Taylor, J. Am. Chem. Soc. 1965, 87,1995.
- Compounds of Formula 2 wherein R13 is —CN and R14 is NH2 can be treated with thiourea, guanidine, or chloroformamidine to give compounds of Formula 4 in which R6 is —NH2 and R7 is —NH2. See, H. Kosaku, Heterocycles, 2001, 55, 2279; A. Gangjee, U.S. Pat. No. 5,939,420 (1999).
- Compounds of Formula 2 wherein R13 is —CN and R14 is NH2 can be treated with formamidine acetate to give compounds of Formula 4 wherein R6 is NH2 and R7 is H (J. A. Montgomery, J. Chem. Soc. 1967, 665). The same transformation can be accomplished by treatment with DMF-DMA or an orthoester such as (EtO)3CH, followed by treatment with ammonia. See, E. C. Taylor, J. Am. Chem. Soc, 1965, 87, 1995.
- Compounds of Formula 2 wherein R13 is —CN and R14 is NH2 can be treated with formic acid to give compounds of Formula 4 wherein R6 is OH and R7 is H (K. A. M. El-Bayouki, J. Chem. Res. Miniprint, 1995, 1901).
- Compounds of Formula 2 wherein R13 is —CO2NH2 and R14 is —NH2 can be treated under Vilsmeyer-Haack conditions (DMF/POCl3) to give compounds of Formula 4 wherein R6 is OH or Cl and R7 is H. See, K. A. M. El-Bayouki, J. Chem. Res. Miniprint, 1995, 1901.
- Compounds of Formula 2 wherein R13 is —CONH2 and R14 is —NH2 can be treated with CS2 or EtOCS2K to give compounds of Formula 4 in which R6 is —OH and R7 is —SH. See, S. M. Bennett, J. Med. Chem. 1990, 33, 2162.
- 1.3. Assembly of the pyrrolo[2,3-d]pyrimidine starting from an acyclic precursor
-
- 2.1. Alkylation of compounds of Formula 4.
- Compounds of Formula 4 can be alkylated in the presence of a base such as K2CO3, NaH, Cs2CO3, DBU etc. with/without the presence of a catalyst such as NaI, KI, (Bu)4NI etc., and in a polar solvent such as DMF, THF, DMSO etc. using electrophiles such as L1-R-R5 where L1 is a leaving group. See Scheme 5. Leaving groups include but are not limited to, e.g., halogen, triflate, tosylate, mesylate, triphenylphosphonium (generated under Mitsunobu conditions, e.g. PPh3/DEAD) etc. See Kasibhatla, PCT publication number WO 03/037860.
2.2. Preparation of electrophiles L1-R4-R5 wherein L1 is a leaving group -
- The electrophiles can be prepared from the substituted benzene derivatives using various methods reported in the literature, see Jerry March, Advanced Organic Chemistry, 4th edition; Larock, Comprehensive Organic Transformations, 1989, VCH, New York. For example the compounds wherein L1 is Br can be prepared by reduction of the corresponding benzoic acid or benzaldehyde, followed by halogenation. These benzyl derivatives can also be prepared by benzylic oxidation or benzylic halogenation. Further modification of the benzyl ring can be done before or after the pyrrolo[2,3-d]pyrimidine alkylation step.
- These compounds can be prepared by many methods reported in the literature.
- Morisawa, J. Med. Chem. 1974, 17, 1083; Klaus, W., J. Med. Chem. 1992, 35, 438; Abramovitch, R. A.; Smith, E. M. “Pyridine-1-oxide in Pyridine and its Derivatives,” in The Chemistry of Heterocyclic Compounds; Weissberger, A., Taylor, E. C., Eds.; John Wiley, New York, 1974, Pt. 2, pp 1-261; Jeromin, G. E., Chem. Ber. 1987, 120, 649. Blanz, E. J., J. Med. Chem. 1970, 13, 1124; Smith, Kline and French, EP Application EP 0184322, 1986; Abblard, J., Bull. Soc. Chim. Fr. 1972, 2466; Fisher, B. E., The Structure of Isomaltol. J. Org. Chem. 1964, 29, 776. De Cat, A., Bull. Soc. Chim. Belg. 1965, 74, 270; Looker, J. H., J. Org. Chem. 1979, 44, 3407. Ackerman, J. F. Ph.D. Dissertation, University of Notre Dame, June, 1949. These methods can be applied to the synthesis of quinoline and isoquinoline type compounds.
- 2.3. Incorporation of the -R4-R5 fragment by nucleophilic substitution.
- In some cases, the -R4-R5 group can be appended before the bicyclic pyrrolo[2,3-d]pyrimidine bicyclic ring is constructed, and this is further detailed below (paragraph 4, schemes 8 and 9). In these cases the -R4-R5 group can be appended by an aromatic nucleophilic substitution using NH2—R4-R5. The compound NH2—R4—R5 is obtained by treating L1-R4-R5 with ammonia at temperatures of 20-160° C. in a pressure vessel. The corresponding amines where L1 is —NH2 can be prepared by a variety of methods, for instance from compounds where L1 is leaving group such as chloride, bromide, tosylate, mesylate etc. using ammonia, or with sodium azide followed by hydrogenation.
- 3. Further Elaboration of the Ring Systems.
- 3.1. Functional Group Interconversions of R0:
- Compounds of Formula I, wherein R0 is H can be oxidized to compounds of Formula I wherein R0 is OH with pyridinium tribromide or polymer supported pyridinium tribromide in tert-butanol/acetic acid mixture followed by zinc reduction. See, C. Liang, U.S. Pat No. 6,610,688 (2000); L. Sun, Bioorg. Med. Chem Lett., 2002, 12, 2153.
- Compounds of Formula I, wherein R0 is H can be treated under Mannich conditions (HCHO+HNRR′) to give compounds Formula I wherein R0 is —CH—NRR′. See, F. Seela, Synthesis, 1997, 1067.
- Compounds of Formula I, wherein R0 is H can be lithiated and treated with electrophiles (e.g., I2, ArCHO) to provide compounds of Formula I wherein R0 is, e.g. —I or —CH(OH)Ar. See, E. Bisagni, Tetrahedron, 1983, 39, 1777; T, Sakamoto, Tetrahedron Lett. 1994, 35, 2919; T. Sakamoto, J. Chem. Soc., Perkin Trans 1, 1996, 459.
- 3.2. Functional group interconversions of R1:
- Compounds of Formula I, wherein R1 is OH, can be converted to halides using standard conditions POCl3, POBr3 etc. with/without a base such as Et3N, N,N-dimethylaniline, (iPr)2NEt etc. and with/without a catalyst such as BnEt3N+Cl−, in polar solvents such as CH3CN, CH2Cl2 etc. Related methods include, but are not limited to, SOCl2/DMF (M. J. Robins, Can. J. Chem. 1973, 12, 3161), PPh3 /CCl4 (L. De Napoli, J. Chem. Soc. Perkin Trans 1, 1994, 923), HMPT/CCl4 or HMPT/NBS (E. A. Veliz, Tetrahedron Lett, 2000, 41, 1695) or PPh3/I2 (X. Lin, Org. Letters, 2000, 2, 3497).
- Compounds of Formula I, wherein R1 is NH2, can be converted to halides by a Balz-Schiemann (F) or Sandmeyer reaction (Cl, Br, I) by means of a nitrosylating agent (NaNO2/H+, NOBF4, RONO) and a halogen donor (BF4 −, CuX2, SbX3).
- Compounds of Formula I, wherein R1 is alkyl can be prepared from compounds of Formula 4 where R1 is halogen and trialkyl aluminum or dialkyl zinc (A. Holy, J. Med. Chem. 1999, 42, 2064).
- Compounds of Formula I, wherein R1 is a halide can be converted to compounds wherein R1 is —NH2, —OH, —SH, —OR8 , —SR8 with standard reagents, e.g., NH3, NaOH, thiourea, R8O−, R8S−, with or without a catalyst (e.g. Pd, Ni, Cu, Lewis acid, H+) (e.g., B. G. Ugarkar, J. Med. Chem. 2000, 43, 2883-2893 and 2894-2905).
- Compounds of Formula I, wherein R1 is halogen or another leaving group can be treated with ammonia to provide compounds of Formula I wherein R1 is NH2 (F. Seela, Liebigs. Ann. Chem. 1985, 315).
- 3.2. Functional group interconversions of R2:
- Compounds of Formula I, wherein R2 is NH2 can be temporarily protected, e.g. as an amide (Ac2O, PivCl), a carbamate (tBoc)2O) or amidine (DMF-DMA).
- Compounds of Formula I, wherein R2 is NH2 can be converted to halides by a Balz-Schiemann (F) or Sandmeyer reaction (Cl, Br, I) by means of a nitrosylating agent (NaNO2/H+, NOBF4, RONO) and a halogen donor (BF4−, CuX2, SbX3).
- Compounds of Formula I, wherein R2 is a halide can be converted to compounds wherein R2 is NH2, OH, SH, OR8, SR8 with standard reagents, e.g. NH3, NaOH, thiourea, R8O−, R8S−, with or without a catalyst (e.g. Pd, Ni, Cu, Lewis acid, H+).
- Compounds of Formula I, wherein R2 is SH can be converted to halides (Br2). They can also be oxidized (e.g., H2O2) and treated with ammonia to give a NH2 group (S. M. Bennett, J. Med. Chem. 1990, 33, 2162).
- Compounds of Formula I, wherein R2 is a sulfide, e.g., MeS—, can be converted to a sulfone, e.g MeSO2—, and displaced with a nucleophile, e.g. NH3 or NH2—NH2, N3—, CN—.
- 3.3. Functional group interconversions of R3:
- Compounds of Formula I, wherein R3 is H can be halogenated (J. F. Gerster, J. Chem. Soc. 1969, 207) and further functionalized by Pd-catalyzed reactions ((a) Sonogashira coupling: E. C. Taylor et al, Tetrahedron, 1992, 48, 8089; (b) carboxylation: J. W. Pawlik, J. Heterocycl. Chem. 1992, 29, 1357; (c) Suzuki coupling: T. Y. I Wu, Org. Lett., 2003,5, 3587) or by addition of nucleophiles (e.g. hydrazine, B. M. Lynch, Can. J. Chem. 1988, 66, 420).
- Compounds of Formula I wherein R3 is —CHO can be sujected to a Bayer-Villiger oxidation to provide compounds of Formula I wherein R3 is —O—CHO. The latter can be hydrolyzed to R3 is —OH. (A. S. Bourlot, E. Desarbre, J. Y. Mérour Synthesis 1994, 411)
- Compounds of Formula I, wherein R3is H can be treated under Mannich condition (HCHO+HNRR′) to give compounds Formula I wherein R3 is —CH—NRR′ (F. Seela, Synthesis, 1997, 10-67)
- Compounds of Formula I, wherein R3 is —CH2—NBn2 can be obtained by Mannich reaction and further treated with an aniline of Formula NH2—Ar to give compounds of Formula I wherein R3 is —CH2—NH—Ar (D. C. Miller, J. Med. Chem. 2002, 45, 90).
- Compounds of Formula I, wherein R3 is Br can be metallated with BuLi, and treated with an electrophile such as MeI to give a compound of Formula I, wherein R3 is Me. Compounds of Formula I, wherein R1 is Cl and R3 is Br can undergo selective metallation at R3 (J. S. Pudlo, J. Med. Chem. 1990, 33, 1984).
- Compounds of Formula I, wherein R0 is OH and R3 is H can be be monalkylated or bis-alkylated to give compounds of Formula III, wherein R1 is an alkyl group. The alkylation can be effected in the presence of a base such as KHMDS, LHMDS, LDA etc. with/without the presence of a catalyst such as NaI, KI, (Bu)4NI etc., and in a polar solvent such as THF, DMSO etc. using electrophiles such as L1-R3 where L1 is a leaving group. Leaving groups include but are not limited to, e.g., halogen, triflate, tosylate or mesylate.
- Compounds of Formula I, wherein R0 is H and R3 is H can be oxidized to compounds of Formula 16/IV, with an oxidizing reagent such as ruthenium tetroxide in a binary solvent such as acetonitrile/water. (G. W. Gribble Org. Prep. Proced. Int. 2001, 33(6), 615).
- Compounds of Formula I, wherein R0 is OH and R3 is H can be oxidized to compounds of Formula II, (wherein R3, R3 is an oxo group) with an oxidizing reagent such as selenium dioxide or oxygen in presence of a cobalt (III) catalyst. (SeO2 oxidation: Romeo Helv. Chim. Acta. 1955, 38, 463, 465. Oxygen oxidation: A. Inada Heterocycles 1982, 19, 2139).
- 3.4. Further elaboration of R5:
- R5, especially when it is aryl or heteroaryl, can be further modified as needed, for example by halogenation, nitration, palladium coupling of halogen, Friedel-Crafts alkylation/acylation, etc. or these modifications can also be done before alkylation, see Jerry March, Advanced Organic Chemistry. The heteroaromatic rings can also be oxidized to their corresponding N-oxides using various oxidizing agents such as H2O2, O3, MCPBA etc. in polar solvents such as CH2Cl2, CHCl3, CF3COOH etc. See Jerry March, Advanced Organic Chemistry, 4th edition, Chapter 19. Examples of modifications are suggested in Scheme 6.
4. Permutations of the order of events - As mentioned above, the events (1) assembly of the bicyclic system (2) appendage of the R5-R4-moiety, and (3) further elaboration of the ring systems do not necessarily have to be made in the sequence (1)-(2)-(3), and it may be beneficial to proceed in a different sequence.
- Method 4.1.
-
- Method 4.1.1
- The compound of Formula 18, wherein R1 is Cl and R2 is NH2, can be prepared by treating the compound of Formula 17 wherein R2═NH2, and R1═X═Cl, with R5-R4—NH2 in butanol at reflux in presence of a base such as K2CO3, Cs2CO3 or iPrNEt2. (A. B. Reitz J. Med. Chem. 1994, 37, 3561).
- Method 4.1.2
- The compound of Formula 19, wherein R1 is Cl and R2 is NH2 and L1=Br on I, can be prepared by refluxing the compound of Formula 18 in chloroform or dichloroethane in presence of an halogenating reagent such as bromine, N-bromosuccinimide, iodine or N-iodosuccinimide and an acid such as acetic acid or p-toluenesulfonic acid. (A. P. Phillips J. Am. Chem. Soc. 1952, 74, 3922).
- Method 4.1.3
- The compound of Formula 20, wherein R1 is Cl and R2 is NH2, can be prepared by coupling the compound of Formula 19 with trimethylsilylacetylene under Sonogashira conditions followed by hydroboration using dicylohexylborane and oxidation using hydrogen peroxide in presence of sodium hydroxide. (Sonogashira coupling: E. C. Taylor Tetrahedron, 1992, 48, 8089. Hydroboration/oxidation: G. Zweifel J. Am. Chem. Soc. 1976, 98, 3184).
- Method 4.1.4
- The compound of Formula 21, wherein R1 is Cl and R2 is NH2, can be prepared by heating the compound of Formula 20 in a polar aprotic solvent such as THF, DME or dioxane in presence of oxalyl chloride, thionyl chloride, mesyl chloride or alkyl chloroformate and a base such as iPrNEt2 or pyridine. It can also be prepared by treating the compound of Formula 20 with coupling reagents such DCC/HOBt, DCC/DMAP or EDCI/HOBt. (R. C. Larock Comprehensive Organic Transformations, Second Edition, p. 1870).
- Method 4.2
- Again, as mentioned above, the events (1) assembly of the bicyclic system (2) appendage of the R5-R4-moiety, and (3) further elaboration of the ring systems do not necessarily have to be made in the sequence (1)-(2)-(3), and it may be beneficial to proceed in a different sequence. For illustrative purposes, Scheme 9 shows a putative synthesis in which the order of events is not (1)-(2)-(3), but is (2)-(1)-(3). First R5 is appended via an aromatic nucleophilic substitution, then the bicyclic system is constructed, and finally it is elaborated.
- Method 4.3
- For illustrative purposes, Scheme 10 shows a putative synthesis in which the order of events is not (1)-(2)-(3), but is (1)-(3)-(2)-(3). First the bicyclic ring is constructed, then it is elaborated, then the R4-R5 moiety is appended, and finally the bicyclic ring system is further elaborated (deprotection).
- Also, if R5 is for instance a pyridine, it can be converted to a N-oxide either before or after alkylation.
- IV. Pharmaceutical Compositions, Dosaging, and Modes of Administration
- The present invention is directed to the clinical use of the heterocyclics, in particular, the pyrazolopyrimidines and their related analogs of Formulae A, I, II, III and IV, and their polymorphs, solvates, esters, tautomers, diastereomers, enantiomers, pharmaceutically acceptable salts and prodrugs thereof, for use in treatment or prevention of diseases that are HSP90-dependent. Examples of such diseases include disorders such as inflammatory diseases, infections, autoimmune disorders, stroke, ischemia, cardiac disorder, neurological disorders, fibrogenetic disorders, proliferative disorders, tumors, leukemias, neoplasms, cancers, carcinomas, metabolic diseases, and malignant disease. The fibrogenetic disorders include but are not limited to scleroderma, polymyositis, systemic lupus, rheumatoid arthritis, liver cirrhosis, keloid formation, interstitial nephritis and pulmonary fibrosis.
- The present invention features pharmaceutical compositions comprising the compound of Formulae A, I, II, III and IV, or a polymorph, solvate, ester, tautomer, enantiomer, diastereomer, pharmaceutically acceptable salt thereof, or prodrug thereof, of any of the preceding aspects and embodiments and one or more pharmaceutical excipients.
- Those of ordinary skill in the art are familiar with formulation and administration techniques that can be employed with the compounds and methods of the invention, e.g., as discussed in Goodman and Gilman, The Pharmacological Basis of Therapeutics, current ed.; Pergamon; and Remington's, Pharmaceutical Sciences (current edition), Mack Publishing Co., Easton, Pa.
- The compounds utilized in the methods of the instant invention may be administered either alone or in combination with pharmaceutically acceptable carriers, excipients or diluents, in a pharmaceutical composition, according to standard pharmaceutical practice. The compounds can be administered orally or parenterally, including the intravenous, intramuscular, intraperitoneal, subcutaneous, rectal and topical routes of administration.
- For example, the therapeutic or pharmaceutical compositions of the invention can be administered locally to the area in need of treatment. This may be achieved by, for example, but not limited to, local infuision during surgery, topical application, e.g., cream, ointment, injection, catheter, or implant, said implant made, e.g., out of a porous, non-porous, or gelatinous material, including membranes, such as sialastic membranes, or fibers. The administration can also be by direct injection at the site (or former site) of a tumor or neoplastic or pre-neoplastic tissue.
- Still further, the compounds or compositions of the invention can be delivered in a vesicle, e.g., a liposome (see, for example, Langer, Science 1990, 249,1527-1533; Treat et al., Liposomes in the Therapy of Infectious Disease and Cancer, Lopez-Bernstein and Fidler, Ed., Liss, N.Y., pp. 353-365, 1989).
- The compounds and pharmaceutical compositions used in the methods of the present invention can also be delivered in a controlled release system. In one embodiment, a pump may be used (see, Sefton, 1987, CRC Crit. Ref. Biomed. Eng. 14:201; Buchwald et al. Surgery, 1980 88, 507; Saudek et al. N. Engl. J. Med. 1989, 321, (574). Additionally, a controlled release system can be placed in proximity of the therapeutic target. (See, Goodson, Medical Applications of Controlled Release, 1984, Vol. 2, pp. 115-138).
- The pharmaceutical compositions used in the methods of the instant invention can also contain the active ingredient in a form suitable for oral use, for example, as tablets, troches, lozenges, aqueous or oily suspensions, dispersible powders or granules, emulsions, hard or soft capsules, or syrups or elixirs. Compositions intended for oral use may be prepared according to any method known in the art for the manufacture of pharmaceutical compositions, and such compositions may contain one or more agents selected from the group consisting of sweetening agents, flavoring agents, coloring agents and preserving agents in order to provide pharmaceutically elegant and palatable preparations. Tablets contain the active ingredient in admixture with non-toxic pharmaceutically acceptable excipients which are suitable for the manufacture of tablets. These excipients may be, for example, inert diluents, such as calcium carbonate, sodium carbonate, lactose, calcium phosphate or sodium phosphate; granulating and disintegrating agents, such as microcrystalline cellulose, sodium crosscarmellose, corn starch, or alginic acid; binding agents, for example starch, gelatin, polyvinyl-pyrrolidone or acacia, and lubricating agents, for example, magnesium stearate, stearic acid or talc. The tablets may be un-coated or coated by known techniques to mask the taste of the drug or delay disintegration and absorption in the gastrointestinal tract and thereby provide a sustained action over a longer period. For example, a water soluble taste masking material such as hydroxypropylmethyl-cellulose or hydroxypropylcellulose, or a time delay material such as ethyl cellulose, or cellulose acetate butyrate may be employed as appropriate.
- Formulations for oral use may also be presented as hard gelatin capsules wherein the active ingredient is mixed with an inert solid diluent, for example, calcium carbonate, calcium phosphate or kaolin, or as soft gelatin capsules wherein the active ingredient is mixed with water soluble carrier such as polyethyleneglycol or an oil medium, for example peanut oil, liquid paraffin, or olive oil.
- Aqueous suspensions contain the active material in admixture with excipients suitable for the manufacture of aqueous suspensions. Such excipients are suspending agents, for example sodium carboxymethylcellulose, methylcellulose, hydroxypropylmethyl-cellulose, sodium alginate, polyvinyl-pyrrolidone, gum tragacanth and gum acacia; dispersing or wetting agents may be a naturally-occurring phosphatide, for example lecithin, or condensation products of an alkylene oxide with fatty acids, for example polyoxyethylene stearate, or condensation products of ethylene oxide with long chain aliphatic alcohols, for example heptadecaethylene-oxycetanol, or condensation products of ethylene oxide with partial esters derived from fatty acids and a hexitol such as polyoxyethylene sorbitol monooleate, or condensation products of ethylene oxide with partial esters derived from fatty acids and hexitol anhydrides, for example polyethylene sorbitan monooleate. The aqueous suspensions may also contain one or more preservatives, for example ethyl, or n-propyl p-hydroxybenzoate, one or more coloring agents, one or more flavoring agents, and one or more sweetening agents, such as sucrose, saccharin or aspartame.
- Oily suspensions may be formulated by suspending the active ingredient in a vegetable oil, for example arachis oil, olive oil, sesame oil or coconut oil, or in mineral oil such as liquid paraffin. The oily suspensions may contain a thickening agent, for example beeswax, hard paraffin or cetyl alcohol. Sweetening agents such as those set forth above, and flavoring agents may be added to provide a palatable oral preparation. These compositions may be preserved by the addition of an anti-oxidant such as butylated hydroxyanisol or alpha-tocopherol.
- Dispersible powders and granules suitable for preparation of an aqueous suspension by the addition of water provide the active ingredient in admixture with a dispersing or wetting agent, suspending agent and one or more preservatives. Suitable dispersing or wetting agents and suspending agents are exemplified by those already mentioned above. Additional excipients, for example sweetening, flavoring and coloring agents, may also be present. These compositions may be preserved by the addition of an anti-oxidant such as ascorbic acid.
- The compounds and pharmaceutical compositions used in the methods of the instant invention may also be in the form of an oil-in-water emulsion. The oily phase may be a vegetable oil, for example olive oil or arachis oil, or a mineral oil, for example liquid paraffin, or mixtures of these. Suitable emulsifying agents may be naturally-occurring phosphatides, for example soybean lecithin, and esters or partial esters derived from fatty acids and hexitol anhydrides, for example sorbitan monooleate, and condensation products of the said partial esters with ethylene oxide, for example polyoxyethylene sorbitan monooleate. The emulsions may also contain sweetening agents, flavoring agents, preservatives and antioxidants.
- Syrups and elixirs may be formulated with sweetening agents, for example glycerol, propylene glycol, sorbitol or sucrose. Such formulations may also contain a demulcent, a preservative, flavoring and coloring agents and antioxidant.
- The pharmaceutical compositions may be in the form of a sterile injectable aqueous solution. Among the acceptable vehicles and solvents that may be employed are water, Ringer's solution and isotonic sodium chloride solution.
- The sterile injectable preparation may also be a sterile injectable oil-in-water microemulsion where the active ingredient is dissolved in the oily phase. For example, the active ingredient may be first dissolved in a mixture of soybean oil and lecithin. The oil solution may then be introduced into a water and glycerol mixture and processed to form a microemulsion.
- The injectable solutions or microemulsions may be introduced into a patient's blood-stream by local bolus injection. Alternatively, it may be advantageous to administer the solution or microemulsion in such a way as to maintain a constant circulating concentration of the instant compound. In order to maintain such a constant concentration, a continuous intravenous delivery device may be utilized. An example of such a device is the Deltec CADD-PLUS™ model 5400 intravenous pump.
- The pharmaceutical compositions may be in the form of a sterile injectable aqueous or oleagenous suspension for intramuscular and subcutaneous administration. This suspension may be formulated according to the known art using those suitable dispersing or wetting agents and suspending agents which have been mentioned above. The sterile injectable preparation may also be a sterile injectable solution or suspension in a non-toxic parenterally-acceptable diluent or solvent, for example as a solution in 1,3-butanediol. In addition, sterile, fixed oils are conventionally employed as a solvent or suspending medium. For this purpose any bland fixed oil may be employed including synthetic mono- or diglycerides. In addition, fatty acids such as oleic acid find use in the preparation of injectables.
- The compounds of the present invention used in the methods of the present invention may also be administered in the form of suppositories for rectal administration of the drug. These compositions can be prepared by mixing the inhibitors with a suitable non-irritating excipient which is solid at ordinary temperatures but liquid at the rectal temperature and will therefore melt in the rectum to release the drug. Such materials include cocoa butter, glycerinated gelatin, hydrogenated vegetable oils, mixtures of polyethylene glycols of various molecular weights and fatty acid esters of polyethylene glycol.
- For topical use, creams, ointments, jellies, solutions or suspensions, etc., containing a compound or composition of the invention can be used. As used herein, topical application can include mouth washes and gargles.
- The compounds used in the methods of the present invention can be administered in intranasal form via topical use of suitable intranasal vehicles and delivery devices, or via transdermal routes, using those forms of transdermal skin patches well known to those of ordinary skill in the art. To be administered in the form of a transdermal delivery system, the dosage administration will, of course, be continuous rather than intermittent throughout the dosage regimen.
- The methods, compounds and compositions of the instant invention may also be used in conjunction with other well known therapeutic agents that are selected for their particular usefulness against the condition that is being treated. For example, the instant compounds may be useful in combination with known anti-cancer and cytotoxic agents. Further, the instant methods and compounds may also be usefuil in combination with other inhibitors of parts of the signaling pathway that links cell surface growth factor receptors to nuclear signals initiating cellular proliferation.
- The methods of the present invention may also be useful with other agents that inhibit angiogenesis and thereby inhibit the growth and invasiveness of tumor cells, including, but not limited to VEGF receptor inhibitors, including ribozymes and antisense targeted to VEGF receptors, angiostatin and endostatin.
- Examples of antineoplastic agents that can be used in combination with the compounds and methods of the present invention include, in general, and as appropriate, alkylating agents, anti-metabolites, epidophyllotoxins, an antineoplastic enzyme, a topoisomerase inhibitor, procarbazine, mitoxantrone, platinum coordination complexes, biological response modifiers and growth inhibitors, hormonal/anti-hormonal therapeutic agents and haematopoietic growth factors. Exemplary classes of antineoplastic include the anthracyclines, vinca drugs, mitomycins, bleomycins, cytotoxic nucleosides, epothilones, discodermolide, pteridines, diynenes and podophyllotoxins. Particularly usefuil members of those classes include, for example, carminomycin, daunorubicin, aminopterin, methotrexate, methopterin, dichloromethotrexate, mitomycin C, porfiromycin, 5-fluorouracil, 6-mercaptopurine, gemcitabine, cytosine arabinoside, podophyllotoxin or podo-phyllotoxin derivatives such as etoposide, etoposide phosphate or teniposide, melphalan, vinblastine, vincristine, leurosidine, vindesine, leurosine, paclitaxel and the like. Other useful antineoplastic agents include estramustine, carboplatin, cyclophosphamide, bleomycin, gemcitibine, ifosamide, melphalan, hexamethyl melamine, thiotepa, cytarabin, idatrexate, trimetrexate, dacarbazine, L-asparaginase, camptothecin, CPT-11, topotecan, ara-C, bicalutamide, flutamide, leuprolide, pyridobenzoindole derivatives, interferons and interleukins.
- When a compound or composition of the invention is administered into a human subject, the daily dosage will normally be determined by the prescribing physician with the dosage generally varying according to the age, weight, and response of the individual patient, as well as the severity of the patient's symptoms.
- In one exemplary application, a suitable amount of compound is administered to a mammal undergoing treatment for cancer, for example, breast cancer. Administration typically occurs in an amount of between about 0.01 mg/kg of body weight to about 100 mg/kg of body weight per day (administered in single or divided doses), more preferably at least about 0.1 mg/kg of body weight per day. A particular therapeutic dosage can include, e.g., from about 0.01 mg to about 1000 mg of compound, and preferably includes, e.g., from about 1 mg to about 1000 mg. The quantity of active compound in a unit dose of preparation may be varied or adjusted from about 0.1 mg to 1000 mg, preferably from about 1 mg to 300 mg, more preferably 10 mg to 200 mg, according to the particular application. The amount administered will vary depending on the particular IC50 value of the compound used and the judgment of the attending clinician taking into consideration factors such as health, weight, and age. In combinational applications in which the compound is not the sole active ingredient, it may be possible to administer lesser amounts of compound and still have therapeutic or prophylactic effect.
- Preferably, the pharmaceutical preparation is in unit dosage form. In such form, the preparation is subdivided into unit doses containing appropriate quantities of the active component, e.g., an effective amount to achieve the desired purpose.
- The actual dosage employed may be varied depending upon the requirements of the patient and the severity of the condition being treated. Determination of the proper dosage for a particular situation is within the skill of the art. Generally, treatment is initiated with smaller dosages which are less than the optimum dose of the compound. Thereafter, the dosage is increased by small amounts until the optimum effect under the circumstances is reached. For convenience, the total daily dosage may be divided and administered in portions during the day if desired.
- The amount and frequency of administration of the compounds and compositions of the present invention used in the methods of the present invention, and if applicable other chemotherapeutic agents and/or radiation therapy, will be regulated according to the judgment of the attending clinician (physician) considering such factors as age, condition and size of the patient as well as severity of the disease being treated.
- The chemotherapeutic agent and/or radiation therapy can be administered according to therapeutic protocols well known in the art. It will be apparent to those skilled in the art that the administration of the chemotherapeutic agent and/or radiation therapy can be varied depending on the disease being treated and the known effects of the chemotherapeutic agent and/or radiation therapy on that disease. Also, in accordance with the knowledge of the skilled clinician, the therapeutic protocols (e.g., dosage amounts and times of administration) can be varied in view of the observed effects of the administered therapeutic agents (i.e., antineoplastic agent or radiation) on the patient, and in view of the observed responses of the disease to the administered therapeutic agents.
- Also, in general, the compounds of the invention need not be administered in the same pharmaceutical composition as a chemotherapeutic agent, and may, because of different physical and chemical characteristics, be administered by a different route. For example, the compounds/compositions may be administered orally to generate and maintain good blood levels thereof, while the chemotherapeutic agent may be administered intravenously. The determination of the mode of administration and the advisability of administration, where possible, in the same pharmaceutical composition, is well within the knowledge of the skilled clinician. The initial administration can be made according to established protocols known in the art, and then, based upon the observed effects, the dosage, modes of administration and times of administration can be modified by the skilled clinician.
- The particular choice of compound (and where appropriate, chemotherapeutic agent and/or radiation) will depend upon the diagnosis of the attending physicians and their judgment of the condition of the patient and the appropriate treatment protocol.
- The compounds/compositions of the invention (and where appropriate chemotherapeutic agent and/or radiation) may be administered concurrently (e.g., simultaneously, essentially simultaneously or within the same treatment protocol) or sequentially, depending upon the nature of the proliferative disease, the condition of the patient, and the actual choice of chemotherapeutic agent and/or radiation to be administered in conjunction (i.e., within a single treatment protocol) with the compound/composition.
- In combinational applications and uses, the compound/composition and the chemotherapeutic agent and/or radiation need not be administered simultaneously or essentially simultaneously, and the initial order of administration of the compound/composition, and the chemotherapeutic agent and/or radiation, may not be important. Thus, the compounds/compositions of the invention may be administered first followed by the administration of the chemotherapeutic agent and/or radiation; or the chemotherapeutic agent and/or radiation may be administered first followed by the administration of the compounds/compositions of the invention. This alternate administration may be repeated during a single treatment protocol. The determination of the order of administration, and the number of repetitions of administration of each therapeutic agent during a treatment protocol, is well within the knowledge of the skilled physician after evaluation of the disease being treated and the condition of the patient. For example, the chemotherapeutic agent and/or radiation may be administered first, especially if it is a cytotoxic agent, and then the treatment continued with the administration of the compounds/compositions of the invention followed, where determined advantageous, by the administration of the chemotherapeutic agent and/or radiation, and so on until the treatment protocol is complete.
- Thus, in accordance with experience and knowledge, the practicing physician can modify each protocol for the administration of a compound/composition for treatment according to the individual patient's needs, as the treatment proceeds.
- The attending clinician, in judging whether treatment is effective at the dosage administered, will consider the general well-being of the patient as well as more definite signs such as relief of disease-related symptoms, inhibition of tumor growth, actual shrinkage of the tumor, or inhibition of metastasis. Size of the tumor can be measured by standard methods such as radiological studies, e.g., CAT or MRI scan, and successive measurements can be used to judge whether or not growth of the tumor has been retarded or even reversed. Relief of disease-related symptoms such as pain, and improvement in overall condition can also be used to help judge effectiveness of treatment.
- V. Assays for Determining HSP90 Binding and Downstream Effect
- A variety of in vitro and in vivo assays are available to test the effect of the compounds of the invention on HSP90. HSP90 competitive binding assays and functional assays can be performed as known in the art by substituting in the compounds of the invention. Chiosis et al. Chemistry & Biology 2001, 8, 289-299, describe some of the known ways in which this can be done. For example, competition binding assays using, e.g., geldanamycin or 17-AAG as a competitive binding inhibitor of HSP90 can be used to determine relative HSP90 affinity of the compounds of the invention by immobilizing the compound of interest or other competitive inhibitor on a gel or solid matrix, preincubating HSP90 with the other inhibitor, passing the preincubated mix over the gel or matrix, and then measuring the amount of HSP90 that retains or does not retain on the gel or matrix.
- Downstream effects can also be evaluated based on the known effect of HSP90 inhibition on function and stability of various steroid receptors and signaling proteins including, e.g., Raf1 and HER2. Compounds of the present invention induce dose-dependent degradation of these molecules, which can be measured using standard techniques. Inhibition of HSP90 also results in up-regulation of HSP90 and related chaperone proteins that can similarly be measured. Antiproliferative activity on various cancer cell lines can also be measured, as can morphological and functional differentiation related to HSP90 inhibition.
- Many different types of methods are known in the art for determining protein concentrations and measuring or predicting the level of proteins within cells and in fluid samples. Indirect techniques include nucleic acid hybridization and amplification using, e.g., polymerase chain reaction (PCR). These techniques are known to the person of skill and are discussed, e.g., in Sambrook, Fritsch & Maniatis Molecular Cloning: A Laboratory Manual, 2nd ed., Cold Spring Harbor Laboratory, Cold Spring Harbor, N.Y., 1989; Ausubel, et al. Current Protocols in Molecular Biology, John Wiley & Sons, N.Y., 1994, and, as specifically applied to the quantification, detection, and relative activity of HER2/Neu in patient samples, e.g., in U.S. Pat. Nos. 4,699,877, 4,918,162, 4,968,603, and 5,846,749. A brief discussion of two generic techniques that can be used follows.
- The determination of whether cells overexpress or contain elevated levels of HER2 can be determined using well known antibody techniques such as immunoblotting, radioimmunoassays, western blotting, immunoprecipitation, enzyme-linked immunosorbant assays (ELISA), and derivative techniques that make use of antibodies directed against HER2. As an example, HER2 expression in breast cancer cells can be determined with the use of an immunohistochemical assay, such as the Dako Hercep™ test (Dako Corp., Carpinteria, Calif. The Hercep™ test is an antibody staining assay designed to detect HER2 overexpression in tumor tissue specimens. This particular assay grades HER2 expression into four levels: 0, 1, 2, and 3, with level 3 representing the highest level of HER2 expression. Accurate quantitation can be enhanced by employing an Automated Cellular Imaging System (ACIS) as described, e.g., by Press, M. et al. Modern Pathology 2000, 13, 225A.
- Antibodies, polyclonal or monoclonal, can be purchased from a variety of commercial suppliers, or may be manufactured using well-known methods, e.g., as described in Harlow et al. Antibodies: A Laboratory Manual, 2nd ed; Cold Spring Harbor Laboratory, Cold Spring Harbor, N.Y., 1988.
- HER2 overexpression can also be determined at the nucleic acid level since there is a reported high correlation between overexpression of the HER2 protein and amplification of the gene that codes for it. One way to test this is by using RT-PCR. The genomic and cDNA sequences for HER2 are known. Specific DNA primers can be generated using standard, well-known techniques, and can then be used to amplify template already present in the cell. An example of this is described in Kurokawa, H. et al. Cancer Res. 2000, 60, 5887-5894. PCR can be standardized such that quantitative differences are observed as between normal and abnormal cells, e.g., cancerous and noncancerous cells. Well known methods employing, e.g., densitometry, can be used to quantitate and/or compare nucleic acid levels amplified using PCR.
- Similarly, fluorescent in situ hybridization (FISH) assays and other assays can be used, e.g., Northern and/or Southern blotting. These rely on nucleic acid hybridization between the HER2 gene or mRNA and a corresponding nucleic acid probe that can be designed in the same or a similar way as for PCR primers, above. See, e.g., Mitchell M S, and Press M. F. Oncol., Suppl. 1999, 12, 108-116. For FISH, this nucleic acid probe can be conjugated to a fluorescent molecule, e.g., fluorescein and/or rhodamine, that preferably does not interfere with hybridization, and which fluorescence can later be measured following hybridization. See, e.g., Kurokawa, H et al, Cancer Res. 2000, 60, 5887-5894 (describing a specific nucleic acid probe having sequence 5′-FAM-NucleicAcid-TAMRA-p-3′ sequence). ACIS-based approaches as described above can be employed to make the assay more quantitative (de la Torre-Bueno, J., et al. Modern Pathology 2000, 13, 221A).
- Immuno and nucleic acid detection can also be directed against proteins other than HSP90 and HER2, which proteins are nevertheless affected in response to HSP90 inhibition.
- The following examples are offered by way of illustration only and are not intended to be limiting of the full scope and spirit of the invention.
- I. Materials and Methods
- The chemical reagents used to create the novel products of the invention below are all available commercially, e.g., from Aldrich Chemical Co., Milwaukee, Wis., USA. Otherwise their preparation is facile and known to one of ordinary skill in the art, or it is referenced or described herein.
- The final compounds were usually purified by preparative TLC (silica gel 60 Å, Whatman Partisil PK6F) or flash chromatography (silica gel 60 Å, EMD Chemicals) using EtOAc/hexane or MeOH/CH2Cl2 as eluents. Rf's were measured using silica gel TLC plates (silica gel 60 Å, EMD Chemicals). Analytical HPLC chromatograms were obtained using a C18 column (Agilent Zorbax 300SB-C18; 5 microns; 4.6 mm×150 mm). A gradient was applied between solvent A (0.1% TFA in H2O) and solvent B (0.5% TFA in CH3CN) increasing the proportion of A linearly from 5% (t=0) to 100% (t=7.00 min), with a constant flow rate of 1 mL/min. The samples were diluted to typically 0.1-1 mg/mL in MeOH or CH3CN and the injection volumes were typically 10 μL. The column was not heated, and UV detection was effected at 254 nm. 1H-NMR spectra were recorded on a Bruker Avance 400 MHz spectrometer.
- The chemical names were generated using the Beilstein Autonom 2.1 software.
- II. General Procedures
- 1. General Procedures to Prepare and Manipulate the pyrrolo[2,3-d]pyrimidine Ring
-
- A suspension of 4-diamino-6-hydroxypyrimidine (6 mmol), AcONa (12 mmol) and α-haloaldehyde (6 mmol ) in CH3CN (20 mL) and H2O (20 mL) was stirred at 22-40° C. overnight whereupon the starting materials gradually dissolved and the desired pyrrolo[2,3-d]pyrimidine precipitated. The precipitate was collected by filtration and washed (water, acetontrile, ether) and air-dried ((a) C. J. Barnett, Org. Proc. Res. Develop. 1999, 3, 184. (b) F. Seela, Liebigs Ann. Chem. 1987, 15).
-
- A suspension of (2-amino-4,6-dichloro-pyrimidin-5-yl)-acetic acid ethyl ester, R5—R4—NH2 and EtN(i-Pr)2 in BuOH was heated at reflux for 24 h whereupon the solvent was removed under reduced pressure. The residue was then dissolved in CH2Cl2 and washed with sat. NaHCO3 solution and dried with Na2SO4. The crude material was purified by preparative TLC or flash chromatography (EtOAc/hexane or MeOH/CH2Cl2) to give the pure pyrrolo[3,4-d]pyrimidin-6-one.
-
- A suspension of the 4-chloro-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine (1 mmol), benzyl halide (1 mmol) and K2CO3 or Cs2CO3 (1-5 mmol) in dry DMF (5 mL) was heated to 40° C. for 3 to 10 h. Work-up (EtOAc) and purification by preparative TLC or flash chromatography (EtOAc/hexane or MeOH/CH2Cl2) yielded the pure N-7 alkylated product.
-
- A solution of 2-amino-7H-pyrrolo[2,3-d]pyrimidin-4-ol, formaldehyde (2-5 equiv.) and HNR9R9 (2-5 equiv.) in 80% aq. acetic acid was heated in a sealed tube at 60° C. overnight, concentrated, extracted in MeOH:CH2Cl2 (1:10), washed with sat. NaHCO3 and concentrated. See H. Akimoto, J. Chem. Soc. Perkin Trans 1. 1998, 1637.
-
- To a solution of pyrrolo[2,3-d]pyrimidin-6-one in THF at −78° C. was added a base such as LDA, LHMDS or KHMDS and after 30 min, the alkyl halide was further added to give monoalkylated and bisalkylated pyrrolo[2,3-d]pyrimidin-6-ones which were purified by preparative TLC or flash chromatography (EtOAc/hexane or MeOH/CH2Cl2).
-
- A solution of 2-amino-4-chloro-pyrrolo[3,4-d]pyrimidin-6-one and SeO2 in dioxane was heated at reflux until completion of the reaction, (1 h) whereupon the solvent was removed under reduced pressure. The crude was purified by preparative TLC or flash chromatography (EtOAc/hexane or MeOH/CH2Cl2) to give the pure 4-chloro-5-hydroxy-2-imino-2,7-dihydro-pyrrolo[2,3-d]pyrimidin-6-one.
- 2. General Procedures to Manipulate the Pyridine Ring
- General Procedure 2.1: Preparation of Pyridine N-oxides
- A solution of the pyridine derivative (1 mmol) in dichloromethane or chloroform (5 mL) was cooled by means of an ice-bath, treated with m-CPBA (1.1 to 3 mmol) in three portions, and allowed to warm to r.t. The mixture was extracted with dichloromethane and washed with aqueous NaOH, followed by water. Drying (Na2SO4) and concentration afforded the pyridine N-oxide.
- General Procedure 2.2: Preparation of 2-(acetoxymethyl)-piridines
- A solution of the 2-methylpyridine N-oxide (1.0 mmol) in acetic anhydride (5 mL) was heated to reflux for 0.5 h. Work-up (EtOAc), drying (MgSO4), evaporation and purification by preparative TLC or flash chromatography afforded the 2-(acetoxymethyl) pyridine.
- General Procedure 2.3: Preparation of 2-(hydroxymethyl)-pyridines
- A suspension of 2-acetoxymethyl-pyridine derivative and solid K2CO3 in methanol was heated to 50° C. for 5-30 min. Evaporation, work-up (EtOAc), and drying (MgSO4) afforded the 2-(hydroxymethyl)-pyridine.
- General Procedure 2.4: Preparation of 2-(bromomethyl)-pyridines
- A solution of 2-(hydroxymethyl)-pyridine (1.0 mmol) and triphenyl phosphine (1.2 mmol) in dichloromethane or chloroform (5 mL) was cooled to 0° C. A solution of CBr4 (1.5 mmol) in dichloromethane or chlorofornm was added dopwise, and the resulting mixture was stirred at 0° C. for 0.5-1 h. Work-up and purification by flash chromatography afforded the 2-(bromomethyl)-pyridine.
- General Procedure 2.5: Preparation of 2-(aminomethyl)-pyridines
- The 2-(chloromethyl)-pyridine derivative in a solution of ammonia in MeOH was heated at 100° C. overnight whereupon it was concentrated under reduced pressure and purified by flash chromatography (MeOH/CH2Cl2) to afford the 2-(aminomethyl)-pyridine derivative.
- General Procedure 2.6: Preparation of 2-chloropyridines
- A suspension of 2-(hydroxymethyl)-pyridine (10 g) in POCl3 (30 mL) was stirred at 110° C. for 1.5 h. The resulting viscous oil was cooled to r.t. and poured onto ice water (500 g). The pH was adjusted to 10 with solid KOH. Work-up (CHCl3), drying (MgSO4) and evaporation gave the 2-chloropyridine, which was used without purification.
- General Procedure 2.7: Preparation of pyridinium salts.
- A solution of the pyridine was heated in MeOH until it dissolved. A methanolic solution of acid (1.0 equiv of e.g. HCl, MsOH) was added, and the solvent was evaporated to give the pyridinium salt.
- 3. General Procedure to Manipulate Benzene Rings
- General Procedure 3.1: Halogenation of Benzene Rings.
- Variant 1: A solution of the aromatic compound in MeOH/THF/acetate buffer (1N in each AcOH and AcONa) was treated with Br2 (1.3-equiv) at r.t. for 5 min. The excess bromine and solvent were removed on a rotary evaporator. Work-up (CHCl3) and flash chromatography afforded the desired bromobenzene.
- Variant 2: A solution of the aromatic compound (7 mmol) and N-halosuccinimide (NCS, NBS, or NIS, 1.06 equiv) in acetic acid (40 mL) was heated to 4-90° C. for 0.3-1 h. Evaporation, work-up (EtOAc) and flash chromatography afforded the desired halogenated benzene.
- Preparation of Intermediates
-
- The title compound was obtained by chlorination of 5-chloromethyl-1,2,3-trimethoxy-benzene with NCS according to the general procedure 3.1.
- 1H-NMR (CDCl3): δ 6.82 (s, 1H), 4.70 (s, 1H), 3.93 (s, 3H), 3.90 (s, 3H) 3.87 (s, 3H).
-
- The title compound was obtained by oxidation of 2-chloromethyl-4-methoxy-3,5-dimethyl-pyridine according to the general procedure 2.1. HPLC Rt: 4.46 min. 1H-NMR (CDCl3): δ 8.05 (s, 1H), 4.93 (s, 2H), 3.77 (s, 3H), 2.37 (s, 3H), 2.24 (s, 3H).
- The title compound was obtained by treating 2-chloromethyl-4-methoxy-3,5-dimethylpyridine-1-oxide with POCl3 according to the general procedure 2.6. HPLC Rt: 6.757 min. 1H-NMR (CDCl3): δ 4.64 (s, 2H), 3.79 (s, 3H), 2.35 (s, 3H), 2.33 (s, 3H).
-
- The title compound was obtained by treating 2-chloromethyl-3,5-dimethyl-pyridin-4-ol (Tarbit, et al. WO 99/10326) with POCl3 in the same manner as in the general procedure 2.6 (74% yield). HPLC Rt: 5.54 min. 1H-NMR (CDCl3): 8.24 (s, 1H), 4.71 (s, 2H), 2.48 (s, 3H), 2.36 (s, 3H).
-
- 2,3,5-Collidine-N-oxide was obtained by oxidation of 2,3,5-collidine according to the general procedure 2.1 in 70% yield. HPLC Rt: 3.96 min. 1H-NMR (CDCl3): δ 8.03 (s, 1H), 6.90 (s, 1H), 2.47 (s, 3H), 2.31 (s, 3H), 2.24 (s, 3H). m/z (%) 138.2 (M+1, 100%). Rf (20% MeOH/EtOAc): 0.35.
- 2,3,5-collidine-N-oxide (1.3 g, 10 mmol) and K2CO3 (2.9 g, 20 mmol) were suspended in 10 mL of CCl4. Bromine (1 mL, 20 mmol) was added dropwise, and the reaction mixture was heated to reflux for 2 h. Work-up (EtOAc) and flash chromatography (10% MeOH/EtOAc) afforded the title compound as a solid (1.05 g, 51% yield). HPLC Rt: 5.24 min. 1H-NMR (CDCl3): δ 8.06 (s, 1H), 2.56 (s, 3H), 2.43 (s, 3H), 2.31 (s, 3H). m/z (%) 216.2 (M+1, 100%), 218.2 (M+3, 100%). Rf (20% MeOH/EtOAc): 0.45.
- 4-Bromo-2,3,5-collidine-N-oxide (0.25 g, 11 mmol) was dissolved in acetic anhydride (5 mL) and the solution was heated to reflux for 30 min. Work-up and flash chromatography (50% Hexane/EtOAc) afforded the title compound (0.27 g, 96% yield). Rf (50% Hexane/EtOAc): 0.70. HPLC Rt: 4.76 min. 1H-NMR (CDCl3): δ 8.26 (s, 1H), 5.27 (s, 2H), 2.46 (s, 3H), 2.41 (s, 3H), 2.14 (s, 3H).
- A suspension of acetic acid 4-bromo-3,5-dimethyl-pyridin-2-yl methyl ester (0.26 g, 1.0 mmol) and K2CO3 (excess) in MeOH (5 mL) was heated to 50° C. for 15 min. Work-up (CHCl3), evaporation, and filtration through a silica gel pad (eluent: 100% EtOAc) gave the title compound as a white solid (0.19 g, 88% yield). Rf (50% Hexane/EtOAc): 0.5. HPLC Rt: 3.80 min. 1H-NMR (CDCl3): δ 8.23 (s, 1H), 4.70 (s, 2H), 2.46 (s, 3H), 2.30 (s, 3H).
-
- The title compound was obtained by heating 2-chloromethyl-4-methoxy-3,5-dimethyl-pyridine hydrochloride in toluene as described in the patent by Tarbit, et al. WO 99/10326.
- A mixture of 2-chloromethyl-3,5-dimethyl-pyridin-4-ol (8.2 g, 47.8 mmol) and POBr3 (60 g, 209 mmol) was stirred at 130° C. for 3 h. The resulting viscous oil was cooled to r.t. and poured onto ice water. The pH was adjusted to 10 with solid KOH. Work-up (CHCl3), drying (MgSO4) and evaporation afforded the title compound as a purple solid (8.7 g, 78% yield) which was used without purification. HPLC Rt: 6.03 min. 1H-NMR (CDCl3): 8.20 (s, 1H), 4.62 (s, 2H), 2.50 (s, 3H), 2.38 (s, 3H).
- A suspension of 2-chloromethyl-4-methoxy-3,5-dimethyl-pyridine (3.24 g, 14.6 mmol) in PBr3 (8.0 mL, 85.1 mmol, 5.8 equiv.) was heated to 80° C. under nitrogen. A catalytic amount of DMF (0.50 mL, 6.4 mmol, 0.44 equiv.) was added, whereupon the suspension rapidly turned into an orange solution. After 40 min., the reaction was still incomplete as judged by HPLC. The temperature was raised to 110° C. and the reaction was prolonged for 30 min, at which point it was complete. The mixture was poured over ice, made basic with conc. aq. NH4OH and extracted into EtOAc. Washing with water, drying (brine, MgSO4) and concentration gave the title compound as a pink solid (1.51 g, 44%) containing 10% of an impurity by 1H-NMR. The crude was used without further purification. 1H-NMR (CDCl3) δ 8.19 (s, 1H), 4.59 (s, 2H), 2.48 (s, 3H), 2.37 (s, 3H).
- Preparation of Final Compounds
-
- The title compound was obtained by alkylation of 4-chloro-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine (F. Seela,Liebigs Ann. Chem. 1987, 15) with 2-chloromethyl-4-methoxy-3,5-dimethyl-pyridine hydrochloride according to the general procedure 1.3. HPLC Rt: 4.709 min. 1H-NMR (CDCl3): δ 8.23 (s, 1H), 6.90 (m, 1H), 6.38 (m 1H), 5.35 (s, 2H), 4.99 (s, 2H), 3.75 (s, 3H), 2.26 (s, 3H), 2.21 (s, 3H).
-
- The title compound was obtained by alkylation of 4-chloro-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine with 2-bromo-1-chloromethyl-3,4,5-trimethoxy-benzene according to the general procedure 1.3. HPLC Rt: 6.937 min. 1H-NMR (DMSO-d6): δ 7.11 (m 1H), 6.73(s, 2H), 6.42 (s, 1H), 6.37 (m 1H), 5.23 (s, 2H), 3.79 (s, 3H), 3.75 (s, 3H), 3.61 (s, 3H).
-
- The title compound was obtained by alkylation of 4-chloro-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine with 1-chloromethyl-2-iodo-3,4,5-trimethoxy-benzene according to the general procedure 1.3. HPLC Rt: 7.069 min. 1H-NMR (DMSO-d6): δ 7.08 (m 1H), 6.74 (s, 2H), 6.38 (m 1H), 6.36 (s, 1H), 5.19 (s, 2H), 3.80 (s, 3H), 3.77 (s, 3H), 3.60 (s, 3H).
-
- The title compound was obtained by alkylation of 4-chloro-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine with 2-chloromethyl-4-methoxy-3,5-dimethyl-pyridine I-oxide according to the general procedure 1.3. HPLC Rt: 5.079 min. 1H-NMR (DMSO-d6): δ 8.18 (s, 1H), 7.29 (m, 1H), 6.68(s, 2H), 6.24 (m, 1H), 5.38 (s, 2H), 3.70 (s, 3H), 2.42 (s, 3H), 2.17 (s, 3H). ESI-MS 334.2 (M+1).
-
- The title compound was obtained by alkylation of 4-chloro-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine with 5-chloromethyl-1,2,3-trimethoxy-benzene according to the general procedure 1.3. HPLC Rt: 6.036 min. 1H-NMR (CDCl3): δ 6.82 (m, 1H), 6.41(s, 2H), 6.40 (m, 1H), 5.36 (s, 2H), 5.16 (s, 2H), 3.81 (s, 3H), 3.78 (s, 6H).
-
- The title compound was obtained by alkylation of 4-chloro-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine with 2-chloro-6-chloromethyl-4-methoxy-3,5-dimethyl-pyridine according to the general procedure 1.3. HPLC Rt: 6.880 min. 1H-NMR (DMSO-d6): δ 7.06 (m, 1H), 6.63(s, 2H), 6.32 (m, 1H), 5.29 (s, 2H), 3.74 (s, 3H), 2.25 (s, 3H), 2.21 (s, 3H).
-
- The title compound was obtained by alkylation of 4-chloro-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine with 4-chloro-2-chloromethyl-3,5-dimethyl-pyridine according to the general procedure 1.3. HPLC Rt: 5.878 min. 1H-NMR (CDCl3): δ 8.27 (s, 1H), 6.89 (m, 1H), 6.40 (m, 1H), 5.40 (s, 2H), 4.94 (s, 2H), 2.39 (s, 3H), 2.37 (s, 3H).
-
- The title compound was obtained by alkylation of 4-chloro-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine with 1-bromomethyl-2-chloro-4,5-dimethoxy-benzene according to the general procedure 1.3. HPLC Rt: 6.635 min. 1H-NMR (CDCl3): δ 6.91 (m, 1H), 6.90 (s, 1H), 6.71 (s, 1H), 6.42 (m, 1H), 5.30 (s, 2H), 4.97 (s, 2H), 3.88 (s, 3H), 3.75 (s, 3H).
-
- The title compound was obtained by alkylation of 4-chloro-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine with 4-bromo-2-chloromethyl-3,5-dimethyl-pyridine according to the general procedure 1.3. HPLC Rt: 6.072 min. 1H-NMR (DMSO-d6): δ 8.15 (s, 1H), 7.10 (m, 1H), 6.60(s, 1H), 6.30 (m, 1H), 5.40(s, 2H), 2.46 (s, 3H), 2.30 (s, 3H).
-
- The title compound was obtained by alkylation of 4-chloro-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine with 4-chloro-2-chloromethyl-3,5-dimethyl-pyridine 1-oxide according to the general procedure 1.3. HPLC Rt: 5.610 min. 1H-NMR (DMSO-d6): δ 8.36 (s, 1H), 7.26 (m, 1H), 6.69(s, 1H), 6.21 (m, 1H), 5.43(s, 2H), 2.60 (s, 3H), 2.27 (s, 3H).
-
- The title compound was obtained by alkylation of 4-chloro-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine with 4-bromo-2-chloromethyl-3,5-dimethyl-pyridine 1-oxide according to the general procedure 1.3. HPLC Rt: 5.734 min. 1H-NMR (DMSO-d6): δ 8.33 (s, 1H), 7.24 (m, 1H), 6.69 (s, 1H), 6.25 (m, 1H), 5.47(s, 2H), 2.65 (s, 3H), 2.29 (s, 3H).
-
- The title compound was obtained by alkylation of 4-chloro-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine with 1-bromomethyl-4,5-dimethoxy-2-nitro-benzene according to the general procedure 1.3. HPLC Rt: 6.345 min. 1H-NMR (DMSO-d6): δ 7.73 (s, 1H), 7.16 (in, 1H), 6.72 (s, 2H), 6.41 (s, 1H), 6.40 (m, 1H), 5.58(s, 2H), 3.92 (s, 3H), 3.62 (s, 3H).
-
- The title compound was obtained by alkylation of 4-chloro-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine with 4-bromomethyl-1,2-dichloro-benzene according to the general procedure 1.3. HPLC Rt: 7.148 min. 1H-NMR (DMSO-d6): δ 7.60 (m, 1H), 7.59 (in, 1H), 7.25(q, 1H), 7.12(m, 1H), 6.71 (s, 2H), 6.37 (q, 1H), 5.26(s, 2H).
-
- The title compound was obtained by alkylation of 4-chloro-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine with 1-chloromethyl-3,5-dimethoxy-benzene according to the general procedure 1.3. HPLC Rt: 6.423 min. 1H-NMR (DMSO-d6): δ 7.21(m, 1H), 6.69 (s, 2H), 6.40 (m,3H), 6.34 (m, 1H), 5.34 (s, 2H), 3.68 (s, 6H).
-
- The title compound was obtained by alkylation of 4-chloro-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine with 2-chloromethyl-1,4-dimethoxy-benzene according to the general procedure 1.3. HPLC Rt: 6.537 min. 1H-NMR (DMSO-d6): δ 7.13 (m, 1H), 6.85 (d, 1H), 6.82 (m, 1H), 6.68 (s, 2H), 6.35 (m, 1H), 6.22 (d, 1H), 3.78 (s, 3H), 3.60 (s, 3H).
-
- The title compound was obtained by alkylation of 4-bromo-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine (obtained as described in F. Seela, Liebigs Ann. Chem. 1987, 15 for 4-chloro-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine, but replacing POCl3 with POBr3) with 2-chloromethyl-4-methoxy-3,5-dimethyl-pyridine-1-oxide according to the general procedure 1.3. HPLC Rt: 5.158 min. 1H-NMR (DMSO-d6): δ 8.18 (s, 1H), 7.29 (m, 1H), 6.69 (s, 2H), 6.15 (m, 1H), 5.37(s, 2H), 3.70 (s, 3H), 2.42 (s, 3H), 2.17 (s, 3H).
-
- The title compound was obtained by alkylation of 4-bromo-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine with 4-chloro-2-chloromethyl-3,5-dimethyl-pyridine according to the general procedure 1.3. HPLC Rt: 5.803 min. 1H-NMR (DMSO-d6): δ 8.20 (s, 1H), 7.04 (m, 11H), 6.61 (s, 2H), 6.21 (m, 1H), 5.38(s, 2H), 2.42 (s, 3H), 2.28 (s, 3H).
-
- The title compound was obtained by alkylation of 4-bromo-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine with 4-chloro-2-chloromethyl-3,5-dimethyl-pyridine-1-oxide according to the general procedure 1.3. HPLC Rt: 5.688 min. 1H-NMR (DMSO-d6): δ 8.35 (s, 1H), 7.25 (m, 1H), 6.70 (s, 2H), 6.15 (m, 1H), 5.43 (s, 2H), 2.60 (s, 3H), 2.27 (s, 3H).
-
- The title compound was obtained by alkylation of 4-bromo-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine with 4-bromo-2-chloromethyl-3,5-dimethyl-pyridine according to the general procedure 1.3. HPLC Rt: 5.996 min. 1H-NMR (DMSO-d6): δ 8.15 (s, 1H), 7.05 (m, 1H), 6.61 (s, 2H), 6.21 (m, 1H), 5.43 (s, 2H), 2.46 (s, 3H), 2.30 (s, 3H).
-
- The title compound was obtained by alkylation of 4-bromo-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine with 4-bromo-2-chloromethyl-3,5-dimethyl-pyridine-1-oxide according to the general procedure 1.3. HPLC Rt: 5.798 min. 1H-NMR (DMSO-d6): δ 8.33 (s, 1H), 7.24 (m, 1H), 6.71 (s, 2H), 6.15 (m, 1H), 5.46 (s, 2H), 2.64 (s, 3H), 2.29 (s, 3H).
-
- The title compound was obtained by alkylation of 4-bromo-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine with 2-chloromethyl-4-methoxy-3,5-dimethyl-pyridine according to the general procedure 1.3. HPLC Rt: 4.847 min. 1H-NMR (DMSO-d6): δ 8.07 (s, 1H), 7.03 (m, 1H), 6.60 (s, 2H), 6.20 (m, 1H), 5.29 (s, 2H), 3.72 (s, 3H), 2.24 (s, 3H), 2.17 (s, 3H).
-
- The title compound was obtained by alkylation of 4-bromo-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine with 1-cloromethyl-3,5-dimethoxy-benzene according to the general procedure 1.3. HPLC Rt: 6.490 min. 1H-NMR (CDCl3): δ 7.20 (m, 1H), 6.70 (s, 2H), 6.40 (s, 1H), 6.34 (s, 2H), 6.23 (m, 1H), 5.16 (s, 2H), 3.69 (s, 6H).
-
- The title compound was obtained by alkylation of 4-chloro-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine with 1-chloromethyl-3-methoxy-benzene according to the general procedure 1.3. HPLC Rt: 7.177 min. 1H-NMR (DMSO-d6): δ 7.26-7.18 (m, 2H), 6.82-6.80 (m, 1H), 6.67 (s, 1H), 6.70-6.67 (m, 3H), 6.32-6.30 (m, 1H), 5.20 (s, 2H), 3.68 (s, 3H).
-
- The title compound was obtained by alkylation of 4-chloro-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine with 1-chloromethyl-4-methoxy-benzene according to the general procedure 1.3. HPLC Rt: 6.889 min. H-NMR (DMSO-d6): δ 7.19-7.16 (m, 3H), 6.90-6.88 (m, 2H), 6.69 (s, 2H), 6.32-6.30 (m, 1H), 5.18 (s, 2H), 3.71 (s, 3H).
-
- The title compound was obtained by alkylation of N-(4-chloro-5-iodo-7H-pyrrolo[2,3-d]pyrimidin-2-yl)-2,2-dimethyl-propionamide with 2-chloromethyl-4-methoxy-3,5-dimethyl-pyridine-1-oxide according to the general procedure 1.3. HPLC Rt: 6.812min. 1H-NMR (DMSO-d6): δ 10.20 (s, 1H), 8.13(s, 1H), 7.97 (s, 1H), 5.50 (s, 2H), 3.72 (s, 3H), 2.50 (s, 3H), 2.16 (s, 3H), 1.22 (s, 9H).
-
- The title compound was obtained by alkylation of N-(4-chloro-5-iodo-7H-pyrrolo[2,3-d]pyrimidin-2-yl)-2,2-dimethyl-propionamide with 4-bromo-2-chloromethyl-3,5-dimethyl-pyridine-1-oxide according to the general procedure 1.3. HPLC Rt: 7.630 min. H-NMR (DMSO-d6): δ 10.21 (s, 1H), 8.30(s, 1H), 7.91 (s, 1H), 5.59 (s, 2H), 2.72 (s, 3H), 2.28 (s, 3H), 1.22 (s, 9H).
-
- The title compound was obtained by alkylation of N-(4-chloro-5-iodo-7H-pyrrolo[2,3-d]pyrimidin-2-yl)-2,2-dimethyl-propionamide (A. Gangjee, J. Med. Chem. 2003, 46, 591) with 2-chloromethyl-4-methoxy-3,5-dimethyl-pyridine according to the general procedure 1.3. HPLC Rt: 6.627 min. 1H-NMR (DMSO-d6): δ 10.15 (s, 1H), 8.05(s, 1H), 7.73 (s, 1H), 5.46 (s, 2H), 3.74 (s, 3H), 2.33 (s, 3H), 2.16 (s, 3H), 1.21 (s, 9H).
-
- The title compound was obtained by alkylation of N-(4-chloro-7H-pyrrolo[2,3-d]pyrimidin-2-yl)-2,2-dimethyl-propionamide with 4-bromo-2-chloromethyl-3,5-dimethyl-pyridine-1-oxide according to the general procedure 1.3. HPLC Rt: 6.806 min. 1H-NMR (DMSO-d6): δ 10.13 (s, 1H), 8.30(s, 1H), 7.74 (m, 1H), 6.52 (m, 1H), 5.62 (s, 2H), 2.73 (s, 3H), 2.28 (s, 3H), 1.23 (s, 9H).
-
- The title compound was obtained by alkylation of N-(4-chloro-7H-pyrrolo[2,3-d]pyrimidin-2-yl)-2,2-dimethyl-propionamide with 2-chloromethyl-4-methoxy-3,5-dimethyl-pyridine according to the general procedure 1.3. HPLC Rt: 6.087 min. 1H-NMR (CDCl3): δ 8.18 (s, 1H), 8.13(s, 1H), 7.18 (m, 1H), 6.49 (m, 1H), 5.50 (s, 2H), 3.72 (s, 3H), 2.26 (s, 3H), 2.22 (s, 3H), 1.34 (s, 9H).
-
- The title compound was obtained by alkylation of N-(4-chloro-7H-pyrrolo[2,3-d]pyrimidin-2-yl)-2,2-dimethyl-propionamide with 2-chloromethyl-4-methoxy-3,5-dimethyl-pyridine-1-oxide according to the general procedure 1.3. HPLC Rt: 6.115 min. 1H-NMR (CDCl3): δ 8.12 (s, 1H), 8.02(s, 1H), 7.93 (m, 1H), 6.49 (m, 1H), 5.71 (s, 2H), 3.76 (s, 3H), 2.70 (s, 3H), 2.22 (s, 3H), 1.36 (s, 9H).
-
- The title compound was obtained by alkylation of N-(4-chloro-7H-pyrrolo[2,3-d]pyrimidin-2-yl)-2,2-dimethyl-propionamide with 4-chloro-2-chloromethyl-3,5-dimethyl-pyridine according to the general procedure 1.3. HPLC Rt: 6.761 min. 1H-NMR (CDCl3): δ 8.23 (s, 1H), 8.11(s, 1H), 7.15 (m, 1H), 6.50 (m, 1H), 5.71 (s, 2H), 2.43 (s, 3H), 2.33 (s, 3H), 1.35 (s, 9H).
-
- The title compound was obtained by alkylation of N-(4-chloro-5-iodo-7H-pyrrolo[2,3-d]pyrimidin-2-yl)-2,2-dimethyl-propionamide with 4-chloro-2-chloromethyl-3,5-dimethyl-pyridine according to the general procedure 1.3. HPLC Rt: 7.508 min. 1H-NMR (CDCl3): δ 8.17 (s, 1H), 8.11 (s, 1H), 8.07 (s, 1H), 5.77 (s, 2H), 2.81 (s, 3H), 2.33 (s, 3H), 1.37 (s, 9H).
-
- The title compound was obtained by alkylation of N-(4-chloro-7H-pyrrolo[2,3-d]pyrimidin-2-yl)-2,2-dimethyl-propionamide with 4-chloro-2-chloromethyl-3,5-dimethyl-pyridine-1-oxide according to the general procedure 1.3. HPLC Rt: 6.688 min. 1H-NMR (CDCl3): δ 8.15 (s, 1H), 8.09 (s, 1H), 7.87 (m, 1H), 6.47 (m, 1H), 5.77 (s, 2H), 2.84 (s, 3H), 2.31 (s, 3H), 1.37(s,9H).
-
- The title compound was obtained by alkylation of N-(4-chloro-5-iodo-7H-pyrrolo[2,3-d]pyrimidin-2-yl)-2,2-dimethyl-propionamide with 4-chloro-2-chloromethyl-3,5-dimethyl-pyridine-1-oxide according to the general procedure 1.3. HPLC Rt: 7.619 min. 1H-NMR (CDCl3): δ 8.25 (s, 1H), 8.13 (s, 1H), 7.33 (s, 1H), 5.55 (s, 2H), 2.47 (s, 3H), 2.36 (s, 3H), 1.36 (s, 9H).
-
- A solution of octanoic acid {5-[(dibenzylamino)-methyl]-4-oxo-4,7-dihydro-3H-pyrrolo[2,3-d]pyrimidin-2-yl}-amide (1.0 g, 2 mmol; J. Chem. Soc. Perkin Trans. 1 1998, 1637), BnNEt3Cl (1.4 g, 4 mmol), PhNMe2 (0.5 mL) and POCl3 (1.73 mL, 12 mmol) in CH3CN (9.2 mL) was heated to 100° C. for 40 min and concentrated. The residue was poured into ice water and neutralized with 2N NaOH, extracted with EtOAc (50 mL×3), and evaporated, to give the title compound (0.80 g, 76%). HPLC Rt: 6.868 min. 1H-NMR (DMSO-d6): δ 12.19 (s, 1H), 10.49 (s, 1H), 1.45-7.21 (m, 11H), 3.80 (s, 2H), 3.59 (s, 4H), 2.41 (t, 2H), 1,56 (m, 2H), 1.27 (br s 8H), 0.85 (t, 3H).
- A suspension of octanoic acid {4-chloro-5-[(dibenzylamino)-methyl]-7H-pyrrolo[2,3-d]pyrimidin-2-yl}-amide (150 mg, 0.30 mmol), 2-chloromethyl-4-methoxy-3,5-dimethyl-pyridine (56 mg, 0.30 mmol) and K2CO3 (84 mg, 0.60 mmol) in dry DMF (1 mL) was heated to 45° C. overnight. After work-up (EtOAc) and evaporation, the residue was taken up in methanolic 4N HCl (1 mL), stirred at room temperature for 1 h, and neutralized to pH 7 with 2N NaOH. Extraction with EtOAc (10 mL×3), evaporation and purification by preparative TLC (MeOH/CH2Cl2 10:1) gave the title compound (70.5 mg, 45%). HPLC Rt: 5.362 min. 1H-NMR (DMSO-d6): δ 8.05 (s, 1H), 7.30-7.22 (m, 10H), 6.99(s, 1H), 6.57s, 2H), 5.27 (s 2H), 3.70 (s, 2H), 3.65 (s, 3H), 3.54 (s, 4H), 2.17 (s, 3H), 2.15 (s, 3H).
-
- A solution of octanoic acid {5-[(dibenzylamino)-methyl]-4-oxo-4,7-dihydro-3H-pyrrolo[2,3-d]pyrimidin-2-yl}-amide (2.42 g, 3 mmol) and aniline (10 mL) was heated to 90° C. in a sealed tube overnight, concentrated, filtered, and washed with MeOH (2 mL×3) to give the title compound (1.1 g, 57%). HPLC Rt: 6.327 min. 1H-NMR (DMSO-d6): δ 11.77 (s, 1H), 11.47 (s, 1H), 11.37 (s, 1H), 7.05 (m, 2H), 6.85 (s, 1H), 6.62 (m, 2H), 6.52 (m, 1H), 5.58 (t, 1H), 4.31 (d, 2H), 2.43 (t, 2H), 1.58 (m, 2H), 1.27 (m, 8H), 0.86 (t, 3H).
- A solution of octanoic acid (4-chloro-5-phenylaminomethyl-7H-pyrrolo[2,3-d]pyrimidin-2-yl)-amide (270 mg, 0.68 mmol), BnNEt3Cl (0.48 g, 1.36 mmol), PhNMe2 (0.17 mL) and POCl3 (0.59 mL, 4.08 mmol) in CH3CN (3 mL) was heated to 100° C. for 40 min and concentrated. The residue was poured into ice water and neutralized with 2N NaOH, extracted with EtOAc (20 mL×3), evaporated, to give 2-chloromethyl-4-methoxy-3,5-dimethyl-pyridine as a crude oil (282 mg) which was used without purification. A suspension of this crude (282 mg, 0.68 mmol), 2-chloromethyl-4-methoxy-3,5-dimethyl-pyridine (140 mg, 0.68 mmol) and Cs2CO3 (266 mg, 0.68 mmol) in dry DMF (1 mL) was heated to 45° C. overnight. After work-up (EtOAc) and evaporation, the residue was taken up in methanolic 4N HCl (1 mL), stirred at room temperature for 1 h, and neutralized to pH 7 with 2N NaOH. Extraction with EtOAc (10 mL×3), evaporation and purification by preparative TLC (MeOH/CH2Cl2 10: 1) gave the title compound (4.8 mg, 1.6%). HPLC Rt: 4.785 min. 1H-NMR (CDCl3): δ 8.12 (s, 1H), 7.18(m, 2H), 6.75(m, 2H), 6.60 (s, 1H), 5.26(s, 2H), 4.93 (s, 2H), 4.74 (s, 2H), 3.70 (s, 3H), 3.00 (s, 3H), 2.23 (s, 3H), 2.16 (s, 3H).
-
- The title compound was obtained by treating octanoic acid {5-[(dibenzylamino)-methyl]-4-oxo-4,7-dihydro-3H-pyrrolo[2,3-d]pyrimidin-2-yl}-amide (2.42 g, 3 mmol) and N-methylaniline (10 mL) as in step 1 of the previous example. HPLC Rt: 6.325 min. 1H-NMR (DMSO-d6): δ 11.73 (s, 1H), 11.47 (s, 1H), 11.35 (s, 1H), 7.13 (m, 2H), 6.78 (s, 2H), 6.60 (m, 2H), 4.64(s, 2H), 2.98 (s, 3H), 2.43 (t, 2H), 1.58 (m, 2H), 1.27 (m, 8H), 0.86 (t, 3H).
- The title compound was obtained by alkylation of octanoic acid {4-chloro-5-[(methyl-phenyl-amino)-methyl]-7H-pyrrolo[2,3-d]pyrimidin-2-yl}-amide with 2-chloromethyl-4-methoxy-3,5-dimethyl-pyridine and deprotection with 4N HCl as in step 2 of the previous example. HPLC 4.844 min. 1H-NMR (CDCl3): δ 8.20 (s, 1H), 7.16 (m, 2H), 6.87 (s, 1H), 6.70-6.64 (m, 2H), 5.28 (s, 2H), 5.13 (s, 2H), 4.46 (s, 2H), 4.15 (br s, 1H), 3.73 (s, 3H), 2.25 (s, 3H), 2.18 (s, 3H).
-
- A solution of octanoic acid (4-oxo-6-pyrrolidin-1-ylmethyl-4,7-dihydro-3H-pyrrolo[2,3-d]pyrimidin-2-yl)-amide (0.36 g 1 mmol; J. Chem. Soc. Perkin Trans. 1 1998, 1637), BnNEt3Cl (0.70 g, 2 mmol), PhNMe2 (0.25 mL) and POCl3 (0.86 mL, 6 mmol) in CH3CN (5 mL) was heated to 100° C. for 40 min and concentrated. The residue was poured into ice water and neutralized with 2N NaOH, extracted with EtOAc (50 mL ×3), and evaporated to give octanoic acid (4-chloro-6-pyrrolidin-1-ylmethyl-7H-pyrrolo[2,3-d]pyrimidin-2-yl)-amide as a crude (0.33 g) which was used without purification. HPLC Rt: 6.737 min. 1H-NMR (CDCl3): δ 11.60 (br s, 1H), 10.20 (br s, 1H), 6.38 (s, 1H), 3.86 (s, 2H), 2.90 (m, 2H), 2.70 9s, 4H), 1.86 (s, 4H), 1.78 (t, 2H), 1.32-1.29 (m, 8H), 0.90 (t, 3H).
- Step 2. 4-Chloro-7-(4-methoxy-3,5-dimethyl-pyridin-2-ylmethyl)-6-pyrrolidin-1-ylmethyl-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine
- A suspension of the crude octanoic acid (4-chloro-6-pyrrolidin-1-ylmethyl-7H-pyrrolo[2,3-d]pyrimidin-2-yl)-amide (330 mg, 0.87 mmol), 2-chloromethyl-4-methoxy-3,5-dimethyl-pyridine (162 mg, 0.87 mmol) and K2CO3 (121 mg, 0.87 mmol) in dry DMF (1 mL) was heated to 45° C. overnight. After work-up (EtOAc) and evaporation, the residue was taken up in 6N methanolic HCl (1 mL), stirred at room temperature for 1 h, and neutralized to pH 7 with 2N NaOH. Extraction with EtOAc (10 mL×3), evaporation and purification by preparative TLC (MeOH/CH2Cl2 10:1) yielded 4-chloro-7-(4-methoxy-3,5-dimethyl-pyridin-2-ylmethyl)-6-pyrrolidin-1-ylmethyl-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine (4.1 mg, yield 1.0%). HPLC Rt: 6.092 min. 1H-NMR (CDCl3): δ 8.09 (s, 1H), 6.31(s, 1H), 5.55(s, 2H), 4.85(s, 2H), 3.77 (s, 3H), 3.52(brs, 2H), 2.43 (m, 4H), 1.76-1.71(m, 4H).
-
- A mixture of 4-methyl-pentanal (8.60 g, 0.10 mol), 5,5-dibromobarbituric acid (DBBA, 17.15 g, 0.06 mol), 40% HBr (2 mL) and HOAc (1 mL) in CH2Cl2 (180 mL) was stirred at 25° C. for 5 h. after filtration, the filtrate was washed with 1N Na2SO3, Na2CO3, and brine, dried with Na2SO4, and evaporated to give 3-bromo-4-methyl-pentanal (8.76 g, 53%). 1H-NMR (CDCl3): δ 9.40 (s, 1H), 4.40 (t, 1H), 2.10 (m, 1H), 1.06 (s, 3H), 1.05 (s, 3H).
- A suspension of 2,4-diamino-6-hydroxypyrimidine (6.68 g, 50 mmol), AcONa (8.3 g 100 mmol) and 3-bromo-4-methyl-pentanal (8.76 g, 50 mmol) in CH3CN (100 mL) and H2O (100 mL) was stirred at 25° C. overnight whereupon the starting materials gradually dissolved and the desired pyrrolo[2,3-d]pyrimidine precipitated. The precipitate was collected by filtration and washed with MeOH to give 2-amino-5-isopropyl-3,7-dihydro-pyrrolo[2,3-d]pyrimidin-4-one (3.80 g, 40%). HPLC Rt: 4.408 min. 1H-NMR (DMSO-d6): δ 10.58 (s, 1H), 10.10 (s, 1H), 6.30 (s, 1H), 5.97 (s, 2H), 3.03 (7, 1H), 1.20 (s, 3H), 1.19 (s, 3H).
- A mixture of 2-amino-5-isopropyl-3,7-dihydro-pyrrolo [2,3-d]pyrimidin-4-one and acetic anhydride (20 mL) was heated to reflux for 3 h and evaporated. The residue was treated with BnNEt3Cl (8.99 g, 40 mmol), PhNMe2 (4.9 mL) and POCl3 (17 mL, 120 mmol) in CH3CN (100 mL) at 100° C. for 40 min and concentrated. The residue was poured into ice water and neutralized with 2N NaOH, extracted with EtOAc (80 mL×3), and evaporated to give an oil which was digested with methanolic 4N HCl (50 mL) at 50° C. for 2 h. After cooling, and neutralization to pH 7 with 2N NaOH, the solid was collected by filtration and dried to give 4-chloro-5-isopropyl-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine (1.88 g, 45%). HPLC Rt: 5.796 min. 1H-NMR (DMSO-d6): δ 11.170 (s, 1H), 6.82 (s, 1H), 6.42 (s, 2H), 3.24 (7, 1H), 1.25 (s, 3H), 1.23 (s, 3H).
- A suspension of 4-chloro-5-isopropyl-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine (105 mg, 0.5 mmol), 2-chloromethyl-4-methoxy-3,5-dimethyl-pyridine (93 mg, 0.5 mmol) and K2CO3 (85 mg, 0.6 mmol) in dry DMF (1 mL) was heated to 45° C. overnight, Work-up (EtOAc), evaporation, and purification by preparative TLC (MeOH/CH2Cl2 10:1) gave 4-chloro-5-isopropyl-7-(4-methoxy-3,5-dimethyl-pyridin-2-ylmethyl)-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine (36 mg). HPLC Rt: 5.867 min. 1H-NMR (DMSO-d6): δ 8.07 (s, 1H), 6.74(s, 1H), 6.51(s, 2H), 5.22 (s, 2H), 3.70 0s, 3H), 3.23 (7, 1H), 2.21 (s, 3H), 2.15 (s, 3H).
-
- The title compound was obtained by alkylation of 4-chloro-5-isopropyl-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine with 4-chloro-2-chloromethyl-3,5-dimethyl-pyridine according to the general procedure 1.3. HPLC Rt: 6.997 min. 1H-NMR (CDCl3): δ 8.27 (s, 1H), 6.61 (s, 1H), 5.33 (s, 2H), 5.09 (s, 2H), 3.35 (7, 1H), 2.35 (s, 6H), 1.25 (s, 3H), 1.23 (s, 3H).
-
- The title compound was obtained by alkylation of 4-chloro-5-isopropyl-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine with 2-chloromethyl-3,5-dimethyl-pyridine-1-oxide according to the general procedure 1.3. HPLC Rt: 6.753 min. 1H-NMR (DMSO-d6): δ 8.37 (s, 1H), 7.03 (s, 1H), 6.63 (s, 2H), 5.40 (s, 2H), 3.20 (7, 1H), 2.59 (s, 3H), 2.27 (s, 3H), 1.20 (s, 3H), 1.19 (s, 3H).
-
- A mixture of tBuPh2SiCl (25 mL, 98 mmol), 1,4-butanediol (25 mL, 281 mmol), iPrNEt2 (50 mL, 303 mmol) and CH2Cl2 (50 mL) was stirred at rt for 14 h, concentrated, diluted with diethyl ether, washed with water (3×) and brine. Drying (Na2SO4) and concentration afforded the title compound as a clear oil (29.8 g, 93%) which was used without further purification. Rf (EtOAc:hexane 1:4) 0.3. 1H-NMR (CDCl3): δ 7.71 (dd, 4H), 7.43 (m, 6H), 3.74 (t, 2H), 3.70 (q, 2H), 2.10 (br. t, 1H), 1.69 (m, 4H), 1.08 (s, 9H).
- A solution of 4-(tert-butyl-diphenyl-silanyloxy)-butan-1-ol (29.8 g, 91 mmol) in CH2Cl2 (70 mL) was added to a slurry of PCC (21.5 g, 100 mmol), celite (50 g) and CH2Cl2 (300 mL). The mixture was stirred for 2 h at rt, and the celite was removed by filtration and washed with CH2Cl2 (300 mL). Concentration and chromatography (EtOAc/hexane 1:4) afforded the title compound as a clear oil (22.2 g, 75%). Rf (EtOAc:hexane 1:4) 0.7. 1H-NMR (CDCl3): δ 9.82 (t, 1H), 7.68 (dd, 4H), 7.41 (m, 6H), 3.71 (t, 2H), 2.57 (t, 2H), 1.91 (q, 2H), 1.07 (s, 9H).
- A mixture of 4-(tert-butyl-diphenyl-silanyloxy)-butyraldehyde (22.2 g, 68 mmol), 5,5-dibromobarbituric acid (12.1 g, 43 mmol) and CH2Cl2 (80 mL) was treated with 70% aq HBr (1 mL, 14 mmol) and stirred at rt for 1 h. The by-product (barbituric acid) was removed by filtration and washed with CH2Cl2 (100 mL). The combined organic layers were washed (1N Na2S2O3, 5% NaHCO3, half-sat. brine) and dried (Na2SO4). Concentration gave the title compound as a clear oil (25.3 g, 92%) which was used without further purification. Rf (EtOAc:hexane 1:4) 0.7. 1H-NMR (CDCl3): δ 9.55 (d, 1H), 7.68 (dd, 4H), 7.41 (m, 6H), 4.60 (ddd, 1H), 3.84 (m, 2H), 2.35 (m, 1H), 2.10 (m, 1H), 1.07 (s, 9H).
- The title compound was obtained by treating 2-bromo-4-(tert-butyl-diphenyl-silanyloxy)-butyraldehyde (25.3 g, 62 mmol) with 2,4-diamino-6-hydroxypyrimidine (10.2 g, 124 mmol) according to the general procedure 1.1 (23.4 g, 87%). HPLC Rt: 6.981 min. H-NMR (CDCl3): δ 10.67 (s, 1H), 10.14 (s, 1H), 7.55 (m, 4H), 7.38 (m, 6H), 6.37 (s, 1H), 5.98 (s, 2H), 3.86 (t, 2H), 2.86 (t, 2H), 0.95 (s, 9H).
- A solution of 2-amino-5-[2-(tert-butyl-diphenyl-silanyloxy)-ethyl]-3,7-dihydro-pyrrolo[2,3-d]pyrimidin-4-one (22.7 g, 52 mmol) in Ac2O (200 mL) was heated to 110° C. for 2.5 h, concentrated, diluted with toluene (300 mL) and concentrated again to afford the title compound to afford the title compound as a crude brown oil (27 g) which was used without further purification. An aliquot was purified by chromatography for characterization. HPLC Rt: 8.349 min. 1H-NMR (CDCl3): δ 11.77 (s, 1H), 8.81 (s, 1H), 7.61 (dd, 4H), 7.30 (m, 7H), 6.37 (s, 1H), 4.00 (t, 2H), 3.02 (t, 2H), 2.70 (s, 3H), 2.23 (s, 3H), 1.04 (s, 9H).
- A solution of crude N-{7-acetyl-5-[2-(tert-butyl-diphenyl-silanyloxy)-ethyl]-4-oxo-4,7-dihydro-3H-pyrrolo[2,3-d]pyrimidin-2-yl}-acetamide (26.4 g, 51 mmol), BnNEt3Cl (23.2 g, 102 mmol), PhNMe2 (19.6 mL, 153 mmol) and POCl3 (9.3 mL, 77 mmol) in CH3CN (100 mL) was heated to 80° C. for 1.5 h. The mixture was diluted with EtOAc (800 mL), washed (sat. NaHCO3, brine) and concentrated to afford the title compound as an oil (46 g) which was used without further purification. An aliquot was purified by chromatography for characterization. HPLC Rt: 8.562 min. 1H-NMR (CDCl3): δ 8.05 (s, 1H), 7.68 (s, 1H), 7.57 (dd, 4H), 7.40 (m, 6H), 3.99 (t, 2H), 3.06 (t, 2H), 2.98 (s, 3H), 2.52 (s, 3H), 1.04 (s, 9H).
- A solution of crude N-{7-acetyl-5-[2-(tert-butyl-diphenyl-silanyloxy)-ethyl]-4-chloro-7H-pyrrolo[2,3-d]pyrimidin-2-yl}-acetamide (46 g) in MeOH (150 mL) was treated with K2CO3 (8.0 g, 58 mmol) at rt for 15 min. Filtration, concentration, and chromatography (EtOAc/hexane 1:1) afforded the title compound as an oil contaminated with residual PhNMe2 from step 6. The oil was diluted with EtOAc (40 mL) and treated with hexane (40 mL) to obtained the desired product as a pale yellow precipitate (4.2 g, 16% over 3 steps). HPLC Rt: 8.558 min. 1H-NMR (CDCl3): δ 11.75 (br. s, 1H), 11.35 (br. s, 1H), 7.60 (dd, 4H), 7.37 (m, 6H), 3.97 (t, 2H), 3.10 (t, 2H), 2.57 (s, 3H), 1.06 (s, 9H).
- A mixture of N-{5-[2-(tert-butyl-diphenyl-silanyloxy)-ethyl]-4-chloro-7H-pyrrolo[2,3-d]pyrimidin-2-yl}-acetamide (344 mg, 0.70 mmol), 2-chloromethyl-4-methoxy-3,5-dimethyl-pyridine hydrochloride (175 mg, 0.77 mmol), K2CO3 (516 mg, 3.7 mmol) and DMF (3.0 mL) was stirred at rt overnight. Work-up (EtOAc/water; brine) afforded the title compound as an off-white solid which was used without further purification (516 mg, “115%”). HPLC Rt: 8.419 min. 1H-NMR (CDCl3): δ 8.20 (s, 1H), 7.95 (s, 1H), 7.55 (dd, 4H), 7.32 (m, 6H), 7.04 (s, 1H), 5.37 (s, 2H), 3.91 (t, 2H), 3.73 (s, 3H), 3.06 (t, 2H), 2.57 (s, 3H), 2.26 (s, 3H), 2.25 (s, 3H), 0.97 (s, 9H).
- A solution of N-[5-[2-(tert-Butyl-diphenyl-silanyloxy)-ethyl]-4-chloro-7-(4-methoxy-3,5-dimethyl-pyridin-2-ylmethyl)-7H-pyrrolo[2,3-d]pyrimidin-2-yl]-acetamide (511 mg) in THF (3 mL) and MeOH (3 mL) was treated with NaOH 2M (3 mL) at 45° C. for 1.5 h. Work-up and chromatography (EtOAc/hexane 1:1) afforded the title compound as a white powder (290 mg, 69% over 2 steps). HPLC Rt: 8.198 min. 1H-NMR (CDCl3): δ 8.20 (s, 1H), 7.57 (dd, 4H), 7.40 (m, 2H), 7.32 (m, 4H), 6.69 (s, 1H), 5.26 (s, 2H), 4.90 (s, 2H), 3.98 (t, 2H), 3.66 (s, 3H), 3.00 (t, 2H), 2.24 (s, 3H), 2.18 (s, 3H), 0.96 (s, 9H).
- A solution of 5-[2-(tert-butyl-diphenyl-silanyloxy)-ethyl]-4-chloro-7-(4-methoxy-3,5-dimethyl-pyridin-2-ylmethyl)-7H-pyrrolo[2,3-d]pyrimidin-2-ylamine (246 mg, 0.41 mmol) in THF (5 mL) was treated with TBAF (1N in THF, 0.5 mL, 0.50 mmol) at rt for 1 h. Work-up (EtOAc/water, brine) gave the crude product as an oil, which was diluted with diethyl ether (15 mL) whereupon the desired product precipitated out of solution as a white powder (110 mg, 74%). HPLC Rt: 4.474 min. 1H-NMR (CDCl3): δ 8.21 (s, 1H), 6.78 (s, 1H), 5.30 (s, 2H), 4.93 (s, 2H), 3.87 (t, 2H), 3.76 (s, 3H), 3.03 (t, 2H), 2.26 (s, 3H), 2.23 (s, 3H).
- A solution of 2-[2-amino-4-chloro-7-(4-methoxy-3,5-dimethyl-pyridin-2-ylmethyl)-7H-pyrrolo[2,3-d]pyrimidin-5-yl]-ethanol (11.6 mg, 0.031 mmol) and Et3N (30 ul, 0.22 mmol) in THF (2 mL) was treated with MsCl (11 uL, 0.14 mmol) at rt for 15 min to give a solution of the title compound which was used without further purification. In a separate experiment, the material was purified by preparative TLC (EtOAc 100%). HPLC Rt: 4.765 min. 1H-NMR (CDCl3): δ 8.22 (s, 1H), 6.80 (s, 1H), 5.31 (s, 2H), 4.93 (s, 2H), 4.42 (t, 2H), 3.77 (s, 3H), 2.98 (t, 2H), 2.85 (s, 3H), 2.23 (s, 3H), 2.07 (s, 3H).
- The solution of methanesulfonic acid 2-[2-amino-4-chloro-7-(4-methoxy-3,5-dimethyl-pyridin-2-ylmethyl)-7H-pyrrolo[2,3-d]pyrimidin-5-yl]-ethyl ester in THF obtained from step 11 was diluted with i-BuNH2 (4 mL) and heated to 50° C. for 15 h. Concentration, work-up (EtOAc/NaHCO3 sat.; brine) and preparative TLC (MeOH:Et3N:CCH2Cl2 7:3:100) gave the title compound as a colorless oil (6 mg, 50%). HPLC Rt: 4.263 min. 1H-NMR (CDCl3): δ 8.20 (s, 1H), 6.79 (s, 1H), 5.29 (s, 2H), 4.97 (s, 2H), 3.76 (s, 3H), 3.04 (t, 2H), 2.96 (t, 2H), 2.53 (d, 2H), 2.26 (s, 3H), 2.22 (s, 3H), 1.85 (oct., 1H), 0.90 (d, 6H).
-
- The title compound was obtained by condensation between (2-amino-4,6-dichloro-pyrimidin-5-yl)-acetic acid ethyl ester and (4-methoxy-3,5-dimethyl-pyridin-2-yl)-methylamine according to the general procedure 1.2. HPLC Rt: 4.893 min. 1H-NMR (CDCl3): δ 8.07 (s, 1H), 5.03 (s, 2H), 4.92 (s, 2H), 3.77 (s, 3H), 3.57 (s, 2H), 2.31 (s, 3H), 2.20 (s, 3H).
-
- The title compound was obtained by alkylation of 4-chloro-pyrrolo[2,3-d]pyrimidin-6-one with 2-chloromethyl-4-chloro-3,5-dimethyl-pyridine according to the general procedure 1.3. HPLC Rt: 5.367 min. 1H-NMR (CDCl3): δ 8.09 (s, 1H), 5.02 (s, 2H), 4.96 (s, 2H), 3.57 (s, 2H), 2.45 (s, 3H), 2.29 (s, 3H).
-
- The title compound was obtained by oxidation of 2-amino-4-chloro-7-(4-methoxy-3,5-dimethyl-pyridin-2-ylmethyl)-5,7-dihydro-pyrrolo[2,3-d]pyrimidin-6-one with m-CPBA according to the general procedure 2.1. HPLC Rt: 4.763 min. 1H-NMR (DMSO-d6): δ 8.01 (s, 1H), 7.01 (s, 2H), 4.93 (s, 2H), 3.73 (s, 3H), 3.46 (s, 2H), 2.40 (s, 3H), 2.19 (s, 3H).
-
- The title compound was obtained by oxidation of 2-amino-4-chloro-7-(4-chloro-3,5-dimethyl-pyridin-2-ylmethyl)-5,7-dihydro-pyrrolo[2,3-d]pyrimidin-6-one with m-CPBA according to the general procedure 2.1. HPLC Rt: 4.90 min. 1H-NMR (CDCl3/CD3OD): δ 7.96 (s, 1H), 5.12 (s, 2H), 3.38 (s, 2H), 2.47 (s, 3H), 2.27 (s, 3H).
-
- The title compound was obtained by condensation between (2-Amino-4,6-dichloro-pyrimidin-5-yl)-acetic acid ethyl ester and 3,4,5-Trimethoxy-benzylamine according to the general procedure 1.2. HPLC Rt: 6.391 min. 1H-NMR (CDCl3): δ 6.70 (s, 2H), 5.14 (s, 2H), 4.77 (s, 2H), 3.84 (s, 6H), 3.81 (s, 3H), 3.47 (s, 2H).
-
- The title compound was obtained by treating 2-amino-4-chloro-7-(3,4,5-trimethoxy-benzyl)-5,7-dihydro-pyrrolo[2,3-d]pyrimidin-6-one with bromine in acetic acid according to the general procedure 3.1. HPLC Rt: 7.150 min. 1H-NMR (CDCl3): δ 6.49 (s, 1H), 5.14 (s, 2H), 4.94 (s, 2H), 3.90 (s, 3H), 3.86 (s, 3H), 3.75 (s, 3H), 3.55 (s, 2H).
-
- The title compound was obtained by alkylation of 2-amino-4-chloro-7-(4-methoxy-3,5-dimethyl-pyridin-2-ylmethyl)-5,7-dihydro-pyrrolo[2,3-d]pyrimidin-6-one with iodomethane according to the general procedure 1.5. HPLC Rt: 4.091 min. 1H-NMR (CDCl3): δ 8.01 (s, 1H), 5.15 (s, 2H), 4.93 (d, 1H), 4.87 (d, 1H), 3.76 (s, 3H), 3.51 (s, 1H), 2.29 (s, 3H), 2.20 (s, 3H), 1.78 (s, 3H).
-
- The title compound was obtained by alkylation of 2-amino-4-chloro-7-(4-methoxy-3,5-dimethyl-pyridin-2-ylmethyl)-5,7-dihydro-pyrrolo[2,3-d]pyrimidin-6-one with iodomethane according to the general procedure 1.5. HPLC Rt: 5.002 min. 1H-NMR (CDCl3): δ 8.02 (s, 1H), 5.02 (s, 2H), 4.90 (s, 2H), 3.75 (s, 3H), 2.29 (s, 3H), 2.18 (s, 3H), 1.53 (s, 6H).
-
- The title compound was obtained by alkylation of 2-amino-4-chloro-7-(3,4,5-trimethoxy-benzyl)-5,7-dihydro-pyrrolo[2,3-d]pyrimidin-6-one with iodomethane according to the general procedure 1.5. HPLC Rt: 6.944 min. 1H-NMR (CDCl3): δ 6.34 (s, 1H), 5.09 (s, 2H), 4.93 (s, 2H), 3.90 (s, 3H), 3.86 (s, 3H), 3.71 (s, 3H), 1.52 (s, 6H).
-
- The title compound was obtained by oxidation of 2-amino-4-chloro-7-(4-methoxy-3,5-dimethyl-pyridin-2-ylmethyl)-5,7-dihydro-pyrrolo[2,3-d]pyrimidin-6-one with selenium dioxide according to the general procedure 1.6. HPLC Rt: 4.294 min. 1H-NMR (CDCl3): δ 8.04 (s, 1H), 5.93 (s, 1H), 5.76 (s, 1H), 4.97 (s, 2H), 3.765 (s, 3H), 2.29 (s, 3H), 2.20 (s, 3H).
-
- The title compound was obtained by oxidation of 2-amino-4-chloro-7-(3,4,5-trimethoxy-benzyl)-5,7-dihydro-pyrrolo[2,3-d]pyrimidin-6-one with selenium dioxide according to the general procedure 1.6. HPLC Rt: 6.156 min. 1H-NMR (CDCl3): δ 6.68 (s, 2H), 6.12 (s, 1H), 5.93 (s, 1H), 4.84 (s, 2H), 3.86 (s, 6H), 3.83 (s, 3H).
-
- The title compound was obtained by oxidation of 4-chloro-5-hydroxy-2-imino-7-(2-bromo-3,4,5-trimethoxy-benzyl)-2,7-dihydro-pyrrolo[2,3-d]pyrimidin-6-one according to the general procedure 1.6. HPLC Rt: 6.230 min. 1H-NMR (CDCl3): δ 6.57 (s, 1H), 6.14 (s, 1H), 5.91 (s, 1H), 5.01 (s, 2H), 3.90 (s, 3H), 3.87 (s, 3H), 3.78 (s, 3H).
- Five microgram of purified rHSP90 protein (Stressgen, BC, Canada, #SPP-770) in phosphate buffered saline (PBS) was coated on 96 well plates by incubating overnight at 4° C. Unbound protein was removed and the coated wells were washed twice with 200 μL PBS. DMSO controls (considered as untreated samples) or test compounds were then added at 100-30-10-3-1-0.3 μM dilutions (in PBS), the plates mixed for 30 seconds on the plate shaker, and then incubated for 60 min. at 37° C. The wells were washed twice with 200 μL PBS, and 10 μM biotinylated-geldanamycin (biotin-GM) was added and incubated for 60 min. at 37° C. The wells were washed again twice with 200 μL PBS, before the addition of 20 μg/mL streptavidin-phycoerythrin (streptavidin-PE) (Molecular Probes, Eugene, Oreg.) and incubation for 60 min. at 37° C. The wells were washed again twice with 200 μL PBS. Relative fluorescence units (RFU) was measured using a SpectraMax Gemini XS Spectrofluorometer (Molecular Devices, Sunnyvale, Calif.) with an excitation at 485 nm and emission at 580 nm; data was acquired using SOFTmax®PRO software (Molecular Devices Corporation, Sunnyvale, Calif.). The background was defined as the RFU generated from wells that were not coated with HSP90 but were treated with the biotin-GM and streptavidin-PE. The background measurements were subtracted from each sample treated with biotin-GM and streptavidin-PE measurements before other computation. Percent inhibition of binding for each sample was calculated from the background subtracted values as follows:
% binding inhibition=[(RFU untreated−RFU treated)/RFU untreated]×100. - MCF7 breast carcinoma cell lysates were prepared by douncing in lysing buffer (20 mM HEPES, pH 7.3, 1 mM EDTA, 5 mM MgCl2, 100 mM KCl), and then incubated with or without test compound for 30 mins at 4° C., followed by incubation with biotin-GM linked to BioMag™ streptavidin magnetic beads (Qiagen) for 1 hr at 4° C. The tubes were placed on a magnetic rack, and the unbound supernatant removed. The magnetic beads were washed three times in lysis buffer and boiled for 5 mins at 95° C. in SDS-PAGE sample buffer. Samples were analyzed on SDS protein gels, and Western blots were done for rHSP90. Bands in the Western Blots were quantitated using the Bio-rad Fluor-S MultiImager, and the % inhibition of binding of rHSP90 to the biotin-GM was calculated.
- The lysate binding ability of selected compounds of the invention based on the above assay is summarized in Table 2. The IC50 reported is the concentration of test compound needed to achieve 50% inhibition of the biotin-GM binding to rHSP90 in the MCF7 cell lysates.
- MCF7 breast carcinoma cells (ATCC) were grown in Dulbecco's modified Eagle's medium (DMEM) containing 10% fetal bovine serum (FBS) and 10 mM HEPES, and plated in 24 well plates (50% confluent). Twenty-four hrs later (cells are 65-70% confluent), test compounds were added and incubated overnight for 16 h. For the less potent compounds, the amounts added were 100 μM, 30 μM, 10 μM and 1 μM, and for more potent compounds, the amounts added were 1 μM, 0.3 μM, 0.1 μM, 0.03 μM, 0.01 μM and 0.003 μM. The wells were washed with 1 mL phosphate buffered saline (PBS), and 200 μL trypsin was added to each well. After trypsinization was complete, 50 μL of FBS was added to each well. Then 200 μL cells was transferred to 96 well plates. The cells were pipetted up and down to obtain a single cell suspension. The plates were centrifuiged at 2,500 rpm for 1 min using a Sorvall Legend RT™ tabletop centrifuge (Kendro Laboratory Products, Asheville, N.C.). The cells were then washed once in PBS containing 0.2% BSA and 0.2% sodium azide (BA buffer). Phycoerythrin (PE) conjugated anti HER2/Neu antibody (Becton Dickinson, #340552), or PE conjugated anti-keyhole limpet hemocyanin [KLH] (Becton Dickinson, #340761) control antibody was added at a dilution of 1:20 and 1:40 respectively (final concentration was 1 μg/mL) and the cells were pipeted up and down to form a single cell suspension, and incubated for 15 mins. The cells were washed twice with 200 μL BA buffer, and resuspended in 200 μL BA buffer, and transferred to FACSCAN tubes with an additional 250 μL BA buffer. Samples were analyzed using a FACSCalibur™ flow cytometer (Becton Dickinson, San Jose, Calif.) equipped with Argon-ion laser that emits 15 mW of 488 nm light for excitation of the PE fluorochrome. 10,000 events were collected per sample. A fluorescence histogram was generated and the mean fluorescence intensity (MFI) of each sample was determined using Cellquest software. The background was defined as the MFI generated from cells incubated with control IgG-PE, and was subtracted from each sample stained with the HER2/Neu antibody. Cells incubated with DMSO were used as untreated controls since the compounds were resuspended in DMSO. Percent degradation of HER2 was calculated as follows:
% HER2 degraded=[(MF1 untreated cells−MF1 treated cells)/MF1 untreated cell]×100 - The HER2 degradation ability of selected compounds of the invention based on this assay is summarized in Table 2. IC50 is defined as the concentration at which there was 50% degradation of the HER2/Neu protein.
- MTS assays measure the cytotoxicity of geldanamycin derivatives. MTS (3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium) is a tetrazolium dye that is converted to a formazan product by dehydrogenase enzymes of metabolically active cells (Corey, A. et al. “Use of an aqueous soluble tetrazolium/formazan assay for cell growth assays in culture,” Cancer Commun. 1991, 3, 207-212). Cells were seeded in 96 well plates at 2000 cells/well and allowed to adhere overnight in Dulbecco's modified Eagle's medium supplemented with 10% fetal bovine serum. The final culture volume was 100 μl. Viable cell number was determined by using the Celltiter 96 AQueous Non-radioactive Cell Proliferation Assay (Promega, Madison Wis.). The MTS/PMS (phenazine methosulfate) solution was mixed at a ratio of 20:1, and 20 μL was added per well to 100 μl of culture medium. After 2-4 hours, the formation of the formazan product was measured at 490 nm absorbance using a multiwell plate spectrophotometer. Background was determined by measuring the Abs 490 nm of cell culture medium and MTS-PMS in the absence of cells and was subtracted from all values. Percent viable cells was calculated as follows:
% viable cells=(Abs at 490 nm treated cells/Abs at 490 nm untreated cells)×100 - The effect of selected compounds of the invention on MCF7 breast carcinoma cells according to the MTS assay is summarized in Table 2. IC50 was defined as the concentration of the compound which gave rise to 50% reduction in viable cell number.
TABLE 2 Biological Activities of Selected Compounds of the Invention Lysate HER2 MIS binding IC50 IC50 S. No. Ex # Structure (μM) (μM) (μM) 1 8 ND 0.023 0.1 2 10 ND 0.25 0.6 3 11 0.09 0.08 0.2 4 5 0.15 0.095 0.3 5 13 0.09 0.05 1.0 6 14 0.05 0.038 1.0 7 15 0.03 0.015 0.023 8 20 ND 0.042 1.0 9 21 ND 0.17 >10.0 10 22 ND 0.065 1.0 11 23 ND 0.13 >10.0 12 24 ND 0.025 0.3 13 25 ND 0.15 >10.0 14 46 ND 0.07 ND 16 45 ND 0.02 ND 17 43 ND 0.13 ND 18 47 ND 0.45 ND 19 48 ND 1.5 ND 20 52 ND 0.4 10.0 21 49 ND 0.18 0.9
ND not determined.
- The foregoing examples are not limiting and are merely illustrative of various aspects and embodiments of the present invention. All documents cited herein are indicative of the levels of skill in the art to which the invention pertains and are incorporated by reference herein in their entireties. None, however, is admitted to be prior art.
- One skilled in the art will readily appreciate that the present invention is well adapted to carry out the objects and obtain the ends and advantages mentioned, as well as those inherent therein. The methods and compositions described illustrate preferred embodiments, are exemplary, and are not intended as limitations on the scope of the invention. Certain modifications and other uses will occur to those skilled in the art, and are encompassed within the spirit of the invention, as defined by the scope of the claims.
- The invention illustratively described herein suitably may be practiced in the absence of any element or elements, limitation or limitations which is not specifically disclosed herein. The terms and expressions which have been employed are used as terms of description and not of limitation, and there is no intention in the use of such terms and expressions of excluding any equivalents of the features shown and described, or portions thereof. It is recognized that various modifications are possible within the scope of the invention claimed. Thus, it should be understood that although the present invention has been specifically disclosed by preferred embodiments, optional features, modifications and variations of the concepts herein disclosed may be resorted to by those skilled in the art, and that such modifications and variations are considered to be within the scope of this invention as defined by the description and the appended claims.
- In addition, where features or aspects of the invention are described in terms of Markush groups or other grouping of alternatives, e.g., genuses, those skilled in the art will recognize that the invention is also thereby described in terms of any individual member or subgroup of members of the Markush group or subgenus, and exclusions of individual members as appropriate, e.g., by proviso.
- Other embodiments are within the following claims.
Claims (17)
1-48. (canceled)
49. A method of treating an individual having an HSP90 mediated disorder comprising administering to said individual a pharmaceutical composition comprising a pharmaceutically effective amount of a compound of Formula I:
or a polymorph, solvate, ester, tautomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, wherein:
R0 is selected from hydrogen, halogen, lower alkyl, —SR8, —OR8, —CN, and —NHR8,
R1 is halogen, —OR11, —SR11 or lower alkyl;
R2 is —NHR8;
R3is selected from the group consisting of hydrogen, halogen, —SR8, —OR8, —CN, —C(O)R9, —C(O)OH, —NO2, —NR8R10, lower alkyl, lower alkenyl, lower alkynyl, lower perhaloalkyl, aryl, heteroaryl, alicyclic and heterocyclic, all optionally substituted, wherein:
the aryl, heteroaryl, alicyclic and heterocyclic groups are optionally mono-, bi- or tri-cyclic,
R8 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N, and
the optional substituents on R3 are selected from the group consisting of halogen, lower alkyl, lower alkenyl, lower alkynyl, —SR8, —OR8, —CN, —C(O)OH, —C(O)R9, —NO2, —NR8R10, lower aryl, heteroaryl, alicyclic, lower heterocyclic, arylalkyl, heteroarylalkyl, amino, alkylamino, dialkylamino, diarylalkylamino, oxo, oxa, perhaloalkyl, perhaloalkoxy, perhaloacyl, guanidine, pyridinyl, thiophene, furanyl, indole, indazole, phosphonates, phosphates, phosphoramides, sulfonates, sulfones, sulfates, sulphonamides, carbamates, ureas, thioureas and thioamides, wherein R8 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N;
R0 or R3 is —OH or —SH, the compound may exist as the corresponding (thio)keto tautomer or a mixture of keto-enol tautomers;
R4 is —CHR12—, —C(O)—, —C(S)—, —S(O)— or —SO2—;
R5 is aryl, heteroaryl, alicyclic, or heterocyclic, wherein
the aryl group is substituted with 3 to 5 substituents,
the heteroaryl group is substituted with 2 to 5 substituents,
the alicyclic group is substituted with 3 to 5 substituents,
the heterocyclic group is substituted with 3 to 5 substituents, and
the substituents are selected from the group consisting of halogen, lower alkyl, lower alkenyl, lower alkynyl, —SR8, —OR8, —CN, —C(O)OH, —C(O)R, —NO2, —NR8R10, lower aryl, heteroaryl, alicyclic, lower heterocyclic, arylalkyl, heteroarylalkyl, amino, alkylamino, dialkylamino, diarylalkylamino, oxo, oxa, perhaloalkyl, perhaloalkoxy, perhaloacyl, guanidine, pyridinyl, thiophene, furanyl, indole, indazole, phosphonates, phosphates, phosphoramides, sulfonates, sulfones, sulfates, sulphonamides, carbamates, ureas, thioureas and thioamides, wherein R8 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N;
R8 is hydrogen, lower alkyl, lower alkenyl, or lower alkynyl, lower aryl, lower heteroaryl, or —C(O)R9;
R9 is H, lower alkyl, lower alkenyl, lower alkynyl, lower aryl, lower heteroaryl, —NR10R10, or —OR11, wherein R10 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N;
R10 is hydrogen, lower alkyl, lower alkenyl, lower alkynyl, lower aryl or lower heteroaryl;
R11 is lower alkyl, lower alkenyl, or lower alkynyl, lower heteroaryl or lower aryl; and
R12 is hydrogen or lower alkyl.
50. The method of claim 49 , wherein:
R0 is hydrogen, halogen, —SH, —OH, or —CN,
R1 is halogen; and
R2 is —NHR8, where R8 is hydrogen or —C(O)R9.
51. The method of claim 49 , wherein:
R1 is chloro or bromo,
R2 is —NHR8, where R8 is hydrogen or —C(O)R9,
R3 is hydrogen, halogen, —OR8, —SR8, —NR8R10, lower alkyl, lower alkenyl, lower alkynyl, lower perhaloalkyl, lower aryl, or lower heteroaryl.
52. The method of claim 49 , wherein:
R0 is hydrogen, halogen or —CN,
R2 is —NHR8, where R8 is hydrogen or —C(O)R9; and
R4 is —CH2—.
53. The method of claim 49 , wherein:
R0 is hydrogen, halogen, —SH, —OH or —CN;
R1 is halogen;
R2 is —NH2;
R3 is hydrogen, halogen, —OR8, —SR8, —NR8R8, lower alkyl, lower alkenyl, lower alkynyl, lower perhaloalkyl, lower aryl, or lower heteroaryl, wherein R8 is hydrogen, lower alkyl, lower aryl, or —C(O)R9;
R4 is —CH2—; and
R5 is aryl or heteroaryl, wherein each of said aryl and heteroaryl is monocyclic or bicyclic and is substituted with 3 to 5 substituents.
54. The method of claim 53 , wherein:
R1 is chloro or bromo;
R2 is —NH2; and
R5 is a phenyl having at least three substituents, a pyridyl having at least two substituents, or 1-oxy-pyridyl (N-oxy-pyridyl) having at least two substituents.
55. The method of claim 49 , wherein the HSP90 mediated disorder is selected from the group consisting of inflammatory diseases, infections, autoimmune disorders, stroke, ischemia, cardiac disorders, neurological disorders, fibrogenetic disorders, proliferative disorders, tumors, leukemias, neoplasms, cancers, carcinomas, metabolic diseases, and malignant diseases.
56. The method of claim 55 , wherein the fibrogenetic disorder is further selected from the group consisting of scleroderma, polymyositis, systemic lupus, rheumatoid arthritis, liver cirrhosis, keloid formation, interstitial nephritis and pulmonary fibrosis.
57. The method of claim 49 , further comprising administering at least one therapeutic agent selected from the group consisting of cytotoxic agents, anti-angiogenesis agents and anti-neoplastic agents.
58. The method of claim 57 , wherein the at least one anti-neoplastic agent is selected from the group consisting of alkylating agents, anti-metabolites, epidophyllotoxins, antineoplastic enzymes, topoisomerase inhibitors, procarbazines, mitoxantrones, platinum coordination complexes, biological response modifiers and growth inhibitors, hormonal/anti-hormonal therapeutic agents, and haematopoietic growth factors.
59-68. (canceled)
69. A method of treating an individual having an HSP90 mediated disorder comprising administering to said individual a pharmaceutical composition comprising a pharmaceutically effective amount of a compound of Formula II:
wherein:
R0 is hydrogen, halogen, lower alkyl, —SR8, —OR8, —CN or —NHR8;
R1 is halogen, —OR11, —SR11 or lower alkyl;
R2 is —NH2;
R4 is —CHR12—, —C(O)—, —C(S)—, —S(O)— or —SO2—;
R5 is aryl, heteroaryl, alicyclic, or heterocyclic, wherein:
the aryl group is substituted with 3 to 5 substituents,
the heteroaryl group is substituted with 2 to 5 substituents,
the alicyclic group is substituted with 3 to 5 substituents,
the heterocyclic group is substituted with 3 to 5 substituents, and
the substituents on R5 are selected from the group consisting of halogen, lower alkyl, lower alkenyl, lower alkynyl, —SR8, —OR8, —CN, —C(O)OH, —C(O)R9, —NO2 and —NR8R10, lower aryl, heteroaryl, alicyclic, lower heterocyclic, arylalkyl, heteroarylalkyl, amino, alkylamino, dialkylamino, diarylalkylamino, oxo, oxa, perhaloalkyl, perhaloalkoxy, perhaloacyl, guanidine, pyridinyl, thiophene, furanyl, indole, indazole, phosphonates, phosphates, phosphoramides, sulfonates, sulfones, sulfates, sulphonamides, carbamates, ureas, thioureas and thioamides, wherein R8 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N;
R8 is hydrogen, lower alkyl, lower alkenyl, lower alkynyl, lower aryl, lower heteroaryl, or —C(O)R9;
R9 is H, lower alkyl, lower aryl, lower heteroaryl, —NR10R10, or —OR11, wherein R10 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N;
R10 is hydrogen, lower alkyl, lower heteroaryl, lower aryl, lower alkenyl, or lower alkynyl,
R11 is lower alkyl, lower alkenyl, lower alkynyl, lower heteroaryl or lower aryl;
R12 is hydrogen or lower alkyl; and
R0 and R10 taken together optionally form an exocyclic double bond which is optionally substituted, or optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N.
70-75. (canceled)
76. A method of treating an individual having an HSP90 mediated disorder comprising administering to said individual a pharmaceutical composition comprising a pharmaceutically effective amount of a compound of Formula III:
or a polymorph, solvate, ester, tantomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, wherein:
R1 is halogen, —OR11, —SR11 or lower alkyl;
R2 is —NH2;
R3 is selected from the group consisting of hydrogen, halogen, —SR8, —OR8, —CN, —C(O)R9, —C(O)OH, —NO2, —NR8R10, lower alkyl, lower alkenyl, lower alkynyl, lower perhaloalkyl, aryl, heteroaryl, alicyclic, heterocyclic, all optionally substituted, wherein:
the aryl, heteroaryl, alicyclic and heterocyclic groups are optionally mono-, bi-or tri-cyclic;
R8 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N, and
the optional substituents on R3 are selected from the group consisting of halogen, lower alkyl, lower alkenyl, lower alkynyl, —SR8, —OR8, —CN, —C(O)OH, —C(O)R9, —NO2, —NR8R10, lower aryl, heteroaryl, alicyclic, lower heterocyclic, arylalkyl, heteroarylalkyl, amino, alkylamino, dialkylamino, diarylalkylamino, oxo, oxa, perhaloalkyl, perhaloalkoxy, perhaloacyl, guanidine, pyridinyl, thiophene, furanyl, indole, indazole, phosphonates, phosphates, phosphoramides, sulfonates, sulfones, sulfates, sulphonamides, carbamates, ureas, thioureas and thioamides, wherein R8 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N;
R4 is —CHR12—, —C(O)—, —C(S)—, —S(O)— or —SO2—;
R5 is aryl, heteroaryl, alicyclic, or heterocyclic, wherein
the aryl group is substituted with 3 to 5 substituents,
the heteroaryl group is substituted with 2 to 5 substituents,
the alicyclic group is substituted with 3 to 5 substituents,
the heterocyclic group is substituted with 3 to 5 substituents, and
the substituents on R5 are selected from the group consisting of halogen, lower alkyl, lower alkenyl, lower alkynyl, —SR8, —OR8, —CN, —C(O)OH, —C(O)R9, —NO2 and —NR8R10, lower aryl, heteroaryl, alicyclic, lower heterocyclic, arylalkyl, heteroarylalkyl, amino, alkylamino, dialkylamino, diarylalkylamino, oxo, oxa, perhaloalkyl, perhaloalkoxy, perhaloacyl, guanidine, pyridinyl, thiophene, firanyl, indole, indazole, phosphonates, phosphates, phosphoramides, sulfonates, sulfones, sulfates, sulphonamides, carbamates, ureas, thioureas and thioamides, wherein R8 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N;
R8 is hydrogen, lower alkyl, lower alkenyl, lower alkynyl, lower aryl, lower heteroaryl, or —C(O)R9;
R9 is H, lower alkyl, lower aryl, lower heteroaryl, —NR10R10, or —OR11, wherein R10 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N;
R10 is hydrogen, lower alkyl, lower heteroaryl, lower aryl, lower alkenyl, or lower alkynyl,
R11 is lower alkyl, lower alkenyl, lower alkynyl, lower heteroaryl or lower aryl;
R12 is hydrogen or lower alkyl; and
R3 and R10 taken together optionally form an exocyclic double bond which is optionally substituted, or optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N.
77-80. (canceled)
81. A method of treating an individual having an HSP90 mediated disorder comprising administering to said individual a pharmaceutical composition comprising a pharmaceutically effective amount of a compound of Formula IV:
or a polymorph, solvate, ester, tantomer, enantiomer, pharmaceutically acceptable salt or prodrug thereof, wherein:
R1 is halogen, —OR11, —SR11 or lower alkyl;
R2 is —NH2;
R4 is —CHR12—, —C(O)—, —C(S)—, —S(O)— or —SO2—;
R5 is aryl, heteroaryl, alicyclic, or heterocyclic, wherein
the aryl group is substituted with 3 to 5 substituents,
the heteroaryl group is substituted with 2 to 5 substituents,
the alicyclic group is substituted with 3 to 5 substituents,
the heterocyclic group is substituted with 3 to 5 substituents, and
the substituents on R5 are selected from the group consisting of halogen, lower alkyl, lower alkenyl, lower alkynyl, —SR8, —OR8, —CN, —C(O)OH, —C(O)R9, —NO2 and —NR8R10, lower aryl, heteroaryl, alicyclic, lower heterocyclic, arylalkyl, heteroarylalkyl, amino, alkylamino, dialkylamino, diarylalkylamino, oxo, oxa, perhaloalkyl, perhaloalkoxy, perhaloacyl, guanidine, pyridinyl, thiophene, furanyl, indole, indazole, phosphonates, phosphates, phosphoramides, sulfonates, sulfones, sulfates, sulphonamides, carbamates, ureas, thioureas and thioamides, wherein R8 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N;
R8 is hydrogen, lower alkyl, lower alkenyl, lower alkynyl, lower aryl, lower heteroaryl, or —C(O)R9;
R9 is H, lower alkyl, lower aryl, lower heteroaryl, —NR10R10, or —OR11, wherein R10 and R10 taken together optionally form a ring of 3-7 ring atoms and optionally 1-3 of the ring atoms are heteroatoms selected from the group of O, S and N;
R10 is hydrogen, lower alkyl, lower heteroaryl, lower aryl, lower alkenyl, or lower alkynyl;
R11 is lower alkyl, lower alkenyl, lower alkynyl, lower heteroaryl or lower aryl; and
R12 is hydrogen or lower alkyl.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US11/531,218 US20070173483A1 (en) | 2002-10-30 | 2006-09-12 | Pyrrolopyrimidines and Related Analogs as HSP90-Inhibitors |
Applications Claiming Priority (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| USPCT/US02/35069 | 2002-10-30 | ||
| PCT/US2002/035069 WO2003037860A2 (en) | 2001-10-30 | 2002-10-30 | Purine analogs having hsp90-inhibiting activity |
| US50413503P | 2003-09-18 | 2003-09-18 | |
| US59146704P | 2004-07-26 | 2004-07-26 | |
| US10/945,851 US7138402B2 (en) | 2003-09-18 | 2004-09-20 | Pyrrolopyrimidines and related analogs as HSP90-inhibitors |
| US11/531,218 US20070173483A1 (en) | 2002-10-30 | 2006-09-12 | Pyrrolopyrimidines and Related Analogs as HSP90-Inhibitors |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10/945,851 Continuation US7138402B2 (en) | 2002-10-30 | 2004-09-20 | Pyrrolopyrimidines and related analogs as HSP90-inhibitors |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| US20070173483A1 true US20070173483A1 (en) | 2007-07-26 |
Family
ID=34381103
Family Applications (8)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10/946,637 Expired - Lifetime US7148228B2 (en) | 2003-09-18 | 2004-09-20 | Pyrazolopyrimidines and related analogs as HSP90-inhibitors |
| US10/946,645 Expired - Lifetime US7138401B2 (en) | 2003-09-18 | 2004-09-20 | 2-aminopurine analogs having HSP90-inhibiting activity |
| US10/945,851 Expired - Lifetime US7138402B2 (en) | 2002-10-30 | 2004-09-20 | Pyrrolopyrimidines and related analogs as HSP90-inhibitors |
| US10/946,628 Expired - Lifetime US7129244B2 (en) | 2003-09-18 | 2004-09-20 | Triazolopyrimidines and related analogs as HSP90-inhibitors |
| US11/531,218 Abandoned US20070173483A1 (en) | 2002-10-30 | 2006-09-12 | Pyrrolopyrimidines and Related Analogs as HSP90-Inhibitors |
| US11/531,221 Abandoned US20070185064A1 (en) | 2003-09-18 | 2006-09-12 | 2-Aminopurine Analogs Having HSP90-Inhibiting Activity |
| US11/537,017 Abandoned US20070111997A1 (en) | 2003-09-18 | 2006-09-29 | Triazolopyrimidines and related analogs as HSP90-inhibitors |
| US11/537,002 Abandoned US20070111996A1 (en) | 2003-09-18 | 2006-09-29 | Pyrazolopyrimidines and related analogs as HSP90-inhibitors |
Family Applications Before (4)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10/946,637 Expired - Lifetime US7148228B2 (en) | 2003-09-18 | 2004-09-20 | Pyrazolopyrimidines and related analogs as HSP90-inhibitors |
| US10/946,645 Expired - Lifetime US7138401B2 (en) | 2003-09-18 | 2004-09-20 | 2-aminopurine analogs having HSP90-inhibiting activity |
| US10/945,851 Expired - Lifetime US7138402B2 (en) | 2002-10-30 | 2004-09-20 | Pyrrolopyrimidines and related analogs as HSP90-inhibitors |
| US10/946,628 Expired - Lifetime US7129244B2 (en) | 2003-09-18 | 2004-09-20 | Triazolopyrimidines and related analogs as HSP90-inhibitors |
Family Applications After (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/531,221 Abandoned US20070185064A1 (en) | 2003-09-18 | 2006-09-12 | 2-Aminopurine Analogs Having HSP90-Inhibiting Activity |
| US11/537,017 Abandoned US20070111997A1 (en) | 2003-09-18 | 2006-09-29 | Triazolopyrimidines and related analogs as HSP90-inhibitors |
| US11/537,002 Abandoned US20070111996A1 (en) | 2003-09-18 | 2006-09-29 | Pyrazolopyrimidines and related analogs as HSP90-inhibitors |
Country Status (14)
| Country | Link |
|---|---|
| US (8) | US7148228B2 (en) |
| EP (2) | EP1670802A4 (en) |
| JP (1) | JP2007505933A (en) |
| KR (1) | KR20060070572A (en) |
| CN (1) | CN101906106A (en) |
| AU (2) | AU2004274507B2 (en) |
| BR (1) | BRPI0414533A (en) |
| CA (1) | CA2539548A1 (en) |
| EA (1) | EA010160B1 (en) |
| IL (1) | IL174375A0 (en) |
| MX (1) | MXPA06002997A (en) |
| NO (1) | NO20061396L (en) |
| NZ (1) | NZ546611A (en) |
| WO (1) | WO2005028434A2 (en) |
Cited By (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20060223797A1 (en) * | 2005-03-30 | 2006-10-05 | Conforma Therapeutics Corporation | Alkynyl pyrrolopyrimidines and related analogs as hsp90-inhibitors |
| US20090202626A1 (en) * | 2008-02-07 | 2009-08-13 | Carson Dennis A | Treatment of bladder diseases with a tlr7 activator |
| WO2009139834A1 (en) * | 2008-05-13 | 2009-11-19 | Poniard Pharmaceuticals, Inc. | Bioactive compounds for treatment of cancer and neurodegenerative diseases |
| US20090324551A1 (en) * | 2005-08-22 | 2009-12-31 | The Regents Of The University Of California Office Of Technology Transfer | Tlr agonists |
| US20100210598A1 (en) * | 2009-02-11 | 2010-08-19 | Regents Of The University Of California, San Diego | Toll-like receptor modulators and treatment of diseases |
| WO2010117425A1 (en) * | 2009-03-31 | 2010-10-14 | Biogen Idec Ma Inc. | Certain substituted pyrimidines, pharmaceutical compositions thereof, and methods for their use |
| US20110098294A1 (en) * | 2006-05-31 | 2011-04-28 | Carson Dennis A | Purine analogs |
| US8357374B2 (en) | 2007-02-07 | 2013-01-22 | The Regents Of The University Of California | Conjugates of synthetic TLR agonists and uses therefor |
| US9050319B2 (en) | 2010-04-30 | 2015-06-09 | Telormedix, Sa | Phospholipid drug analogs |
| US9066940B2 (en) | 2009-02-06 | 2015-06-30 | Telormedix, Sa | Pharmaceutical compositions comprising imidazoquinolin(amines) and derivatives thereof suitable for local administration |
| US9173935B2 (en) | 2010-04-30 | 2015-11-03 | Telormedix Sa | Phospholipid drug analogs |
| US11697851B2 (en) | 2016-05-24 | 2023-07-11 | The Regents Of The University Of California | Early ovarian cancer detection diagnostic test based on mRNA isoforms |
Families Citing this family (178)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6992188B1 (en) * | 1995-12-08 | 2006-01-31 | Pfizer, Inc. | Substituted heterocyclic derivatives |
| KR100850727B1 (en) * | 2000-11-02 | 2008-08-06 | 슬로안-케테링인스티튜트퍼캔서리서치 | Small molecule compositions for binding to hsp90 |
| EP1463742A4 (en) * | 2001-06-21 | 2006-05-10 | Ariad Pharma Inc | Novel pyrazolo-and pyrrolo-pyrimidines and uses thereof |
| EP1440072A4 (en) * | 2001-10-30 | 2005-02-02 | Conforma Therapeutic Corp | Purine analogs having hsp90-inhibiting activity |
| US20070129334A1 (en) * | 2001-10-30 | 2007-06-07 | Conforma Therapeutics Corporation | Orally Active Purine-Based Inhibitors of Heat Shock Protein 90 |
| BRPI0414533A (en) | 2003-09-18 | 2006-11-07 | Conforma Therapeutics Corp | compound, pharmaceutical composition, and methods for inhibiting an hsp90 and treating an individual having an hsp90 mediated disorder |
| US8741578B2 (en) * | 2004-11-02 | 2014-06-03 | The Regents Of The University Of California | Methods of detecting chronic lymphocytic leukemia with Hsp90 and ZAP-70 |
| AU2005302000A1 (en) * | 2004-11-02 | 2006-05-11 | Conforma Therapeutics Corporation | Methods and compositions for treating chronic lymphocytic leukemia |
| US9120774B2 (en) | 2004-11-03 | 2015-09-01 | University Of Kansas | Novobiocin analogues having modified sugar moieties |
| US8212012B2 (en) * | 2004-11-03 | 2012-07-03 | University Of Kansas | Novobiocin analogues having modified sugar moieties |
| WO2006109075A2 (en) * | 2005-04-13 | 2006-10-19 | Astex Therapeutics Limited | Hydroxybenzamide derivatives and their use as inhibitors of hsp90 |
| AU2006236557A1 (en) * | 2005-04-14 | 2006-10-26 | Novartis Vaccines And Diagnostics Inc. | 2-amino-quinazolin-5-ones as HSP90 inhibitors useful in treating proliferation diseases |
| WO2006124897A2 (en) * | 2005-05-13 | 2006-11-23 | Lexicon Genetics Incorporated | Methods and compositions for improving cognition |
| CA2615946A1 (en) * | 2005-07-22 | 2007-02-01 | Sunesis Pharmaceuticals, Inc. | Pyrazolo pyrimidines useful as aurora kinase inhibitors |
| AU2006283592A1 (en) | 2005-08-22 | 2007-03-01 | Amgen Inc. | Pyrazolopyridine and pyrazolopyrimidine compounds useful as kinase enzymes modulators |
| EP1919482A1 (en) * | 2005-08-24 | 2008-05-14 | Lexicon Pharmaceuticals, Inc. | Pyrrolopyridine, pyrrolopyrimidine and pyrazolopyridine compounds, compositions comprising them, and methods of their use |
| WO2007035963A2 (en) * | 2005-09-23 | 2007-03-29 | Conforma Therapeutics Corporation | Anti-tumor methods using multi drug resistance independent synthetic hsp90 inhibitors |
| JP2009515884A (en) * | 2005-11-10 | 2009-04-16 | スミスクライン・ビーチャム・コーポレイション | Inhibitor of AKT activity |
| JP2009517342A (en) * | 2005-11-10 | 2009-04-30 | スミスクライン・ビーチャム・コーポレイション | Inhibitor of AKT activity |
| EP1962863A4 (en) * | 2005-12-22 | 2010-11-24 | Conforma Therapeutics Corp | Orally active purine-based inhibitors of heat shock protein 90 |
| WO2007092496A2 (en) * | 2006-02-07 | 2007-08-16 | Conforma Therapeutics Corporation | 7,9-dihydro-purin-8-one and related analogs as hsp90-inhibitors |
| US8658633B2 (en) * | 2006-02-16 | 2014-02-25 | Massachusetts Eye And Ear Infirmary | Methods and compositions for treating conditions of the eye |
| NZ572600A (en) * | 2006-05-12 | 2011-08-26 | Myrexis Inc | 6-Amino-purine derivatives for the treatment of cancer and fibrogenetic disorders |
| US10336757B2 (en) | 2006-06-30 | 2019-07-02 | Sloan-Kettering Institute For Cancer Research | Treatment of neurodegenerative diseases through inhibition of HSP90 |
| US20080009508A1 (en) * | 2006-07-10 | 2008-01-10 | Lucie Szucova | 6,9-Disubstituted Purine Derivatives And Their Use For Treating Skin |
| ATE414281T1 (en) * | 2006-08-03 | 2008-11-15 | Cellzome Ag | METHOD FOR IDENTIFYING MOLECULES THAT INTERACT WITH PI3K AND METHOD FOR PURIFYING PI3K |
| KR20140127367A (en) | 2006-09-19 | 2014-11-03 | 다이이찌 산쿄 가부시키가이샤 | Pyrazolopyrimidine derivative |
| WO2008045529A1 (en) * | 2006-10-12 | 2008-04-17 | Serenex, Inc. | Purine and pyrimidine derivatives for treatment of cancer and inflammatory diseases |
| CL2007002994A1 (en) * | 2006-10-19 | 2008-02-08 | Wyeth Corp | HETEROCICLIC DERIVATIVE COMPOUNDS CONTAINING SULFAMOIL, INHIBITORS OF HSP90; PHARMACEUTICAL COMPOSITION; AND USE FOR THE TREATMENT OF CANCER, SUCH AS CANCER OF BREAST, COLON AND PROSTATE, BETWEEN OTHERS. |
| FR2907453B1 (en) * | 2006-10-24 | 2008-12-26 | Sanofi Aventis Sa | NOVEL FLUORENE DERIVATIVES, COMPOSITIONS CONTAINING SAME AND USE THEREOF |
| GB0622084D0 (en) * | 2006-11-06 | 2006-12-13 | Chroma Therapeutics Ltd | Inhibitors of HSP90 |
| EP2108019A2 (en) * | 2007-01-30 | 2009-10-14 | Biogen Idec MA, Inc. | 1-h-pyrazolo[3,4b]pyrimidine derivatives and their use as modulators of mitotic kinases |
| DE602008003404D1 (en) * | 2007-02-01 | 2010-12-23 | Astrazeneca Ab | 5,6,7,8-TETRAHYDROPTERIDINE DERIVATIVES AS HSP90 INHIBITORS |
| KR101313804B1 (en) * | 2007-03-20 | 2013-10-01 | 쿠리스 인코퍼레이션 | Fused amino pyridine as hsp90 inhibitors |
| CA2684169C (en) | 2007-04-12 | 2012-06-19 | Joyant Pharmaceuticals, Inc. | Smac mimetic dimers and trimers useful as anti-cancer agents |
| US8143265B2 (en) * | 2007-04-16 | 2012-03-27 | Meharry Medical College | Method of treating atherosclerosis |
| US7960353B2 (en) * | 2007-05-10 | 2011-06-14 | University Of Kansas | Novobiocin analogues as neuroprotective agents and in the treatment of autoimmune disorders |
| DE102007028521A1 (en) * | 2007-06-21 | 2008-12-24 | Merck Patent Gmbh | Indazolamidderivate |
| EP2200653A2 (en) | 2007-09-10 | 2010-06-30 | University of Massachusetts | Mitochondria-targeted anti-tumour agents |
| US8119616B2 (en) * | 2007-09-10 | 2012-02-21 | Curis, Inc. | Formulation of quinazoline based EGFR inhibitors containing a zinc binding moiety |
| GB2467670B (en) | 2007-10-04 | 2012-08-01 | Intellikine Inc | Chemical entities and therapeutic uses thereof |
| WO2009086170A1 (en) | 2007-12-21 | 2009-07-09 | Joyant Pharmaceuticals, Inc. | Diazonamide analogs with improved solubility |
| US8895701B2 (en) | 2008-01-05 | 2014-11-25 | Sloan-Kettering Institute For Cancer Research | Peptide-conjugated oligonucleotide therapeutic and method of making and using same |
| US8190543B2 (en) * | 2008-03-08 | 2012-05-29 | Tokyo Electron Limited | Autonomous biologically based learning tool |
| WO2009114470A2 (en) * | 2008-03-10 | 2009-09-17 | Curis, Inc. | Tetrahydroindole and tetrahdyroindazole as hsp90 inhibitors containing a zinc binding moiety |
| US8993580B2 (en) | 2008-03-14 | 2015-03-31 | Intellikine Llc | Benzothiazole kinase inhibitors and methods of use |
| WO2009114870A2 (en) | 2008-03-14 | 2009-09-17 | Intellikine, Inc. | Kinase inhibitors and methods of use |
| JP5123429B2 (en) | 2008-04-29 | 2013-01-23 | ジョイアント ファーマスーティカルズ、インク. | Indoline anticancer agent |
| LT5623B (en) | 2008-04-30 | 2010-01-25 | Biotechnologijos Institutas, , | 5-aryl-4-(5-substituted 2,4-dihydroxyfenil)-1,2,3-thiadiazoles as inhibitors of hsp90 chaperone and the intermediates for production thereof |
| WO2009143485A1 (en) | 2008-05-22 | 2009-11-26 | Joyant Pharmaceuticals, Inc. | Diazonamide analogs |
| WO2010006086A2 (en) | 2008-07-08 | 2010-01-14 | Intellikine, Inc. | Kinase inhibitors and methods of use |
| WO2010014617A1 (en) * | 2008-07-28 | 2010-02-04 | University Of Kansas | Heat shock protein 90 inhibitor dosing methods |
| GB0819102D0 (en) * | 2008-10-17 | 2008-11-26 | Chroma Therapeutics Ltd | Pyrrolo-pyrimidine compounds |
| US8476431B2 (en) | 2008-11-03 | 2013-07-02 | Itellikine LLC | Benzoxazole kinase inhibitors and methods of use |
| WO2010065275A1 (en) * | 2008-11-25 | 2010-06-10 | Merck Sharp & Dohme Corp. | Soluble guanylate cyclase activators |
| CA2786715C (en) * | 2009-01-16 | 2019-05-21 | Curis, Inc. | Fused amino pyridines for the treatment of brain tumors |
| WO2010098344A1 (en) * | 2009-02-25 | 2010-09-02 | 第一三共株式会社 | Tricyclic pyrazolopyrimidine derivative |
| EP3578664B1 (en) * | 2009-03-30 | 2024-10-30 | Nordic Bioscience A/S | Collagen iv neo-epitope fibrosis assay |
| CA2766100C (en) | 2009-06-29 | 2018-05-22 | Incyte Corporation | Pyrimidinones as pi3k inhibitors |
| AU2010269674B2 (en) * | 2009-07-10 | 2014-01-16 | Taiho Pharmaceutical Co., Ltd. | Azabicyclo compound and salt thereof |
| PE20121148A1 (en) | 2009-08-17 | 2012-09-07 | Intellikine Llc | HETEROCYCLIC COMPOUNDS AND USES OF THEM |
| WO2011041593A1 (en) * | 2009-09-30 | 2011-04-07 | University Of Kansas | Novobiocin analogues and treatment of polycystic kidney disease |
| LT2486039T (en) | 2009-10-07 | 2016-10-10 | Sloan Kettering Institute For Cancer Research | Purine derivatives useful as hsp90 inhibitors |
| EP2493879B1 (en) | 2009-10-28 | 2014-05-14 | Joyant Pharmaceuticals, Inc. | Dimeric smac mimetics |
| US20110130711A1 (en) * | 2009-11-19 | 2011-06-02 | Follica, Inc. | Hair growth treatment |
| WO2011075643A1 (en) | 2009-12-18 | 2011-06-23 | Incyte Corporation | Substituted heteroaryl fused derivatives as pi3k inhibitors |
| WO2011075630A1 (en) * | 2009-12-18 | 2011-06-23 | Incyte Corporation | Substituted fused aryl and heteroaryl derivatives as pi3k inhibitors |
| AR079545A1 (en) | 2009-12-21 | 2012-02-01 | Bayer Cropscience Ag | TIENILPIRI (MI) DINILAZOL |
| WO2011130342A1 (en) | 2010-04-14 | 2011-10-20 | Incyte Corporation | FUSED DERIVATIVES AS ΡI3Κδ INHIBITORS |
| EP2571361A4 (en) | 2010-05-19 | 2013-11-13 | Univ North Carolina | PYRAZOLOPYRIMIDINE COMPOUNDS FOR THE TREATMENT OF CANCER |
| WO2011163195A1 (en) | 2010-06-21 | 2011-12-29 | Incyte Corporation | Fused pyrrole derivatives as pi3k inhibitors |
| KR20130099006A (en) | 2010-08-23 | 2013-09-05 | 다이이찌 산쿄 가부시키가이샤 | Free crystal of tricyclic pyrazolopyrimidine derivative |
| JP5701884B2 (en) | 2010-08-23 | 2015-04-15 | 第一三共株式会社 | Crystals of tricyclic pyrazolopyrimidine derivatives |
| EP2638014B1 (en) | 2010-11-08 | 2017-01-04 | Lycera Corporation | N-sulfonylated tetrahydroquinolines and related bicyclic compounds for inhibition of ror-gamma activity and the treatment of diseases |
| KR20140099556A (en) | 2010-12-16 | 2014-08-12 | 에프. 호프만-라 로슈 아게 | Tricyclic pi3k inhibitor compounds and methods of use |
| WO2012083181A1 (en) * | 2010-12-16 | 2012-06-21 | Indiana University Research And Technology Corporation | Alpha helix mimetics and methods for using |
| ES2764848T3 (en) | 2010-12-20 | 2020-06-04 | Incyte Holdings Corp | N- (1- (substituted phenyl) ethyl) -9H-purine-6-amines as PI3K inhibitors |
| US8754114B2 (en) | 2010-12-22 | 2014-06-17 | Incyte Corporation | Substituted imidazopyridazines and benzimidazoles as inhibitors of FGFR3 |
| US8362023B2 (en) | 2011-01-19 | 2013-01-29 | Hoffmann-La Roche Inc. | Pyrazolo pyrimidines |
| CA2825028A1 (en) * | 2011-02-09 | 2012-08-16 | F. Hoffman-La Roche Ag | Heterocyclic compounds as pi3 kinase inhibitors |
| TWI592411B (en) | 2011-02-23 | 2017-07-21 | 英特爾立秦有限責任公司 | Combination of kinase inhibitors and uses thereof |
| WO2012125629A1 (en) | 2011-03-14 | 2012-09-20 | Incyte Corporation | Substituted diamino-pyrimidine and diamino-pyridine derivatives as pi3k inhibitors |
| US9126948B2 (en) | 2011-03-25 | 2015-09-08 | Incyte Holdings Corporation | Pyrimidine-4,6-diamine derivatives as PI3K inhibitors |
| KR101984480B1 (en) | 2011-04-05 | 2019-05-31 | 슬로안-케테링인스티튜트퍼캔서리서치 | Hsp90 inhibitors |
| MX360390B (en) | 2011-04-05 | 2018-10-31 | Sloan Kettering Inst Cancer Res | Hsp90 inhibitors. |
| LT2699580T (en) | 2011-04-22 | 2018-01-10 | Joyant Pharmaceuticals, Inc. | Diazonamide analogs |
| EP2714688B1 (en) | 2011-05-26 | 2016-02-24 | Daiichi Sankyo Company, Limited | Heterocyclic compounds as protein kinase inhibitors |
| US9309250B2 (en) | 2011-06-22 | 2016-04-12 | Vertex Pharmaceuticals Incorporated | Substituted pyrrolo[2,3-b]pyrazines as ATR kinase inhibitors |
| ITTO20110652A1 (en) * | 2011-07-20 | 2013-01-21 | Medestea Res & Production S P A | COMPOUND DERIVED FROM THE ADENINE HAVING IMMUNO-MODULATING ACTION, ANTI-INFLAMMATORY AND ANTI-DOLUTION ACTION |
| KR102131612B1 (en) | 2011-09-02 | 2020-07-08 | 인사이트 홀딩스 코포레이션 | Heterocyclylamines as pi3k inhibitors |
| MX2014004086A (en) | 2011-10-03 | 2014-09-22 | Univ North Carolina | Pyrrolopyrimidine compounds for the treatment of cancer. |
| AR090548A1 (en) | 2012-04-02 | 2014-11-19 | Incyte Corp | BICYCLIC AZAHETEROCICLOBENCILAMINS AS PI3K INHIBITORS |
| RU2014149136A (en) | 2012-05-08 | 2016-07-10 | Мерк Шарп И Доум Корп. | TETRAHYDRONAFTHYRIDINE AND RELATED Bicyclic Compounds for Inhibition of Rorγ Activity and Treatment of a Disease |
| JP6242868B2 (en) | 2012-05-08 | 2017-12-06 | リセラ・コーポレイションLycera Corporation | Tetrahydro [1,8] naphthyridinesulfonamide and related compounds for use as agonists of RORγ and for the treatment of diseases |
| JP2015516464A (en) * | 2012-05-15 | 2015-06-11 | カルアジア ファーマシューティカルズ,インコーポレイテッド | Pyrimidinediamine derivatives as inhibitors of cytoplasmic Hsp90 |
| WO2013177168A1 (en) | 2012-05-22 | 2013-11-28 | The University Of North Carolina At Chapel Hill | Pyrimidine compounds for the treatment of cancer |
| CN107652289B (en) | 2012-06-13 | 2020-07-21 | 因塞特控股公司 | Substituted tricyclic compounds as FGFR inhibitors |
| US9388185B2 (en) | 2012-08-10 | 2016-07-12 | Incyte Holdings Corporation | Substituted pyrrolo[2,3-b]pyrazines as FGFR inhibitors |
| EP2904119B1 (en) | 2012-10-02 | 2020-06-17 | The General Hospital Corporation d/b/a Massachusetts General Hospital | Methods relating to dna-sensing pathway related conditions |
| JP2015531395A (en) | 2012-10-04 | 2015-11-02 | ファイザー・リミテッドPfizer Limited | Pyrrolo [3,2-C] pyridine tropomyosin-related kinase inhibitor |
| CN103724269B (en) * | 2012-10-11 | 2016-12-21 | 中国科学院上海药物研究所 | Phenyl 1, 2-isoxazole or phenyl 1, 2-pyrazole compound and application thereof |
| US9562047B2 (en) | 2012-10-17 | 2017-02-07 | The University Of North Carolina At Chapel Hill | Pyrazolopyrimidine compounds for the treatment of cancer |
| WO2014085225A1 (en) | 2012-11-27 | 2014-06-05 | The University Of North Carolina At Chapel Hill | Pyrimidine compounds for the treatment of cancer |
| HRP20180859T1 (en) | 2012-12-07 | 2018-07-13 | Vertex Pharmaceuticals Incorporated | 2-Amino-6-fluoro-N- (5-fluoro-4- (4- (4- (3-oxetanyl-yl) piperazine-1-carbonyl) piperidin-1-yl) pyridin-3-yl) pyrazolo [ 1,5ALFA] PIRIMIDINE-3-CARBOXAMIDE AS ATR KINASE INHIBITOR |
| US9266892B2 (en) | 2012-12-19 | 2016-02-23 | Incyte Holdings Corporation | Fused pyrazoles as FGFR inhibitors |
| WO2014144715A1 (en) | 2013-03-15 | 2014-09-18 | Memorial Sloan-Kettering Cancer Center | Hsp90-targeted cardiac imaging and therapy |
| EP2970286A1 (en) | 2013-03-15 | 2016-01-20 | Vertex Pharmaceuticals Inc. | Fused pyrazolopyrimidine derivatives useful as inhibitors of atr kinase |
| JP2016512239A (en) | 2013-03-15 | 2016-04-25 | バーテックス ファーマシューティカルズ インコーポレイテッドVertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| EP2970288A1 (en) | 2013-03-15 | 2016-01-20 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of atr kinase |
| WO2014145576A2 (en) * | 2013-03-15 | 2014-09-18 | Northwestern University | Substituted pyrrolo(2,3-d)pyrimidines for the treatment of cancer |
| US9724354B2 (en) | 2013-03-22 | 2017-08-08 | Millennium Pharmaceuticals, Inc. | Combination of catalytic mTORC1/2 inhibitors and selective inhibitors of Aurora A kinase |
| KR102269032B1 (en) | 2013-04-19 | 2021-06-24 | 인사이트 홀딩스 코포레이션 | Bicyclic heterocycles as fgfr inhibitors |
| AU2014293011A1 (en) | 2013-07-26 | 2016-03-17 | Race Oncology Ltd. | Compositions to improve the therapeutic benefit of bisantrene |
| CN105899503B (en) | 2013-08-16 | 2020-08-18 | 纪念斯隆-凯特琳癌症中心 | Selective GRP94 inhibitors and uses thereof |
| HRP20192183T1 (en) | 2013-12-06 | 2020-02-21 | Vertex Pharmaceuticals Inc. | COMPOUND 2-AMINO-6-FLUORO-N- [5-FLUORO-PYRIDIN-3-YL] PYRAZOLO [1,5-A] PYRIMIDINE-3-CARBOXAMIDE USEFUL AS AN ATR KINASE INHIBITOR, ITS PREPARATION, VARIOUS DIFFERENT MARKED DERIVATIVES |
| WO2015095795A1 (en) | 2013-12-20 | 2015-06-25 | Merck Sharp & Dohme Corp. | TETRAHYDRONAPHTHYRIDINE, BENZOXAZINE, AZA-BENZOXAZINE, AND RELATED BICYCLIC COMPOUNDS FOR INHIBITION OF RORgamma ACTIVITY AND THE TREATMENT OF DISEASE |
| WO2015095792A1 (en) | 2013-12-20 | 2015-06-25 | Merck Sharp & Dohme Corp. | Carbamate benzoxaxine propionic acids and acid derivatives for modulation of rorgamma activity and the treatment of disease |
| WO2015095788A1 (en) | 2013-12-20 | 2015-06-25 | Merck Sharp & Dohme Corp. | 2-ACYLAMIDOMETHYL AND SULFONYLAMIDOMETHYL BENZOXAZINE CARBAMATES FOR INHIBITION OF RORgamma ACTIVITY AND THE TREATMENT OF DISEASE |
| US10532088B2 (en) | 2014-02-27 | 2020-01-14 | Lycera Corporation | Adoptive cellular therapy using an agonist of retinoic acid receptor-related orphan receptor gamma and related therapeutic methods |
| ES2548927B1 (en) * | 2014-03-21 | 2016-08-11 | Universidad De Granada | Purine derivatives as Dapk-1 inhibitors |
| WO2015157127A1 (en) | 2014-04-11 | 2015-10-15 | The University Of North Carolina At Chapel Hill | Therapuetic uses of selected pyrimidine compounds with anti-mer tyrosine kinase activity |
| US9896441B2 (en) | 2014-05-05 | 2018-02-20 | Lycera Corporation | Tetrahydroquinoline sulfonamide and related compounds for use as agonists of RORγ and the treatment of disease |
| US10189777B2 (en) | 2014-05-05 | 2019-01-29 | Lycera Corporation | Benzenesulfonamido and related compounds for use as agonists of RORγ and the treatment of disease |
| EP3152212B9 (en) | 2014-06-05 | 2020-05-27 | Vertex Pharmaceuticals Inc. | Radiolabelled derivatives of a 2-amino-6-fluoro-n-[5-fluoro-pyridin-3-yl]- pyrazolo[1,5-a]pyrimidin-3-carboxamide compound useful as atr kinase inhibitor, the preparation of said compound and different solid forms thereof |
| WO2015191677A1 (en) | 2014-06-11 | 2015-12-17 | Incyte Corporation | Bicyclic heteroarylaminoalkyl phenyl derivatives as pi3k inhibitors |
| EP3157566B1 (en) | 2014-06-17 | 2019-05-01 | Vertex Pharmaceuticals Incorporated | Method for treating cancer using a combination of chk1 and atr inhibitors |
| KR20170052687A (en) | 2014-09-17 | 2017-05-12 | 메모리얼 슬로안-케터링 캔서 센터 | Hsp90-targeted inflammation and infection imaging and therapy |
| US10851105B2 (en) | 2014-10-22 | 2020-12-01 | Incyte Corporation | Bicyclic heterocycles as FGFR4 inhibitors |
| KR101713638B1 (en) * | 2014-11-18 | 2017-03-08 | 한국과학기술연구원 | Novel pyrrolo pyrimidine derivatives and composition for preventing or treating cancer comprising the same |
| CA2975997A1 (en) | 2015-02-11 | 2016-08-18 | Merck Sharp & Dohme Corp. | Substituted pyrazole compounds as rorgammat inhibitors and uses thereof |
| PE20171514A1 (en) | 2015-02-20 | 2017-10-20 | Incyte Corp | BICYCLE HETEROCYCLES AS FGFR INHIBITORS |
| WO2016134294A1 (en) | 2015-02-20 | 2016-08-25 | Incyte Corporation | Bicyclic heterocycles as fgfr4 inhibitors |
| MA41551A (en) | 2015-02-20 | 2017-12-26 | Incyte Corp | BICYCLIC HETEROCYCLES USED AS FGFR4 INHIBITORS |
| SI3831833T1 (en) | 2015-02-27 | 2023-03-31 | Incyte Holdings Corporation | Processes for the preparation of a pi3k inhibitor |
| US10421751B2 (en) | 2015-05-05 | 2019-09-24 | Lycera Corporation | Dihydro-2H-benzo[b][1,4]oxazine sulfonamide and related compounds for use as agonists of RORγ and the treatment of disease |
| WO2016183060A1 (en) | 2015-05-11 | 2016-11-17 | Incyte Corporation | Process for the synthesis of a phosphoinositide 3-kinase inhibitor |
| WO2016183063A1 (en) | 2015-05-11 | 2016-11-17 | Incyte Corporation | Crystalline forms of a pi3k inhibitor |
| WO2016201225A1 (en) | 2015-06-11 | 2016-12-15 | Lycera Corporation | Aryl dihydro-2h-benzo[b][1,4]oxazine sulfonamide and related compounds for use as agonists of rory and the treatment of disease |
| CN107922396B (en) * | 2015-07-20 | 2022-08-05 | 建新公司 | Colony stimulating factor-1 receptor (CSF-1R) inhibitors |
| HK1258570A1 (en) | 2015-09-30 | 2019-11-15 | Vertex Pharmaceuticals Inc. | Method for treating cancer using a combination of dna damaging agents and atr inhibitors |
| US10287272B2 (en) | 2015-10-27 | 2019-05-14 | Merck Sharp & Dohme Corp. | Substituted indazole compounds as RORgammaT inhibitors and uses thereof |
| EP3368535B1 (en) | 2015-10-27 | 2020-12-02 | Merck Sharp & Dohme Corp. | Heteroaryl substituted benzoic acids as rorgammat inhibitors and uses thereof |
| WO2017075178A1 (en) | 2015-10-27 | 2017-05-04 | Merck Sharp & Dohme Corp. | SUBSTITUTED BICYCLIC PYRAZOLE COMPOUNDS AS RORgammaT INHIBITORS AND USES THEREOF |
| US10709708B2 (en) | 2016-03-17 | 2020-07-14 | The University Of North Carolina At Chapel Hill | Method of treating cancer with a combination of MER tyrosine kinase inhibitor and an epidermal growth factor receptor (EGFR) inhibitor |
| KR101896616B1 (en) * | 2016-10-11 | 2018-09-07 | 한국과학기술연구원 | 2-Amino-purine derivative compound, preparing method and use thereof |
| KR102053933B1 (en) * | 2016-10-26 | 2019-12-11 | 울산과학기술원 | Compound as TRAP1 inhibitor and pharmaceutical composition for anticancer comprising the same |
| SMT201900517T1 (en) | 2017-03-20 | 2019-11-13 | Forma Therapeutics Inc | Pyrrolopyrrole compositions as pyruvate kinase (pkr) activators |
| AR111960A1 (en) | 2017-05-26 | 2019-09-04 | Incyte Corp | CRYSTALLINE FORMS OF A FGFR INHIBITOR AND PROCESSES FOR ITS PREPARATION |
| US10472361B2 (en) * | 2017-08-16 | 2019-11-12 | Bristol-Myers Squibb Company | Toll-like receptor 7 (TLR7) agonists having a benzotriazole moiety, conjugates thereof, and methods and uses therefor |
| JOP20180094A1 (en) | 2017-10-18 | 2019-04-18 | Hk Inno N Corp | Heterocyclic compound as a protein kinase inhibito |
| CN107857763A (en) * | 2017-11-28 | 2018-03-30 | 江苏杏睿生物科技有限公司 | A kind of adjacent nitro aroylation derivatives quasi-compound, preparation method and applications |
| FR3075794A1 (en) * | 2017-12-21 | 2019-06-28 | Galderma Research & Development | NEW MTOR INHIBITOR COMPOUNDS |
| FR3075795A1 (en) | 2017-12-21 | 2019-06-28 | Galderma Research & Development | NEW MTOR INHIBITOR COMPOUNDS |
| EP3762385B1 (en) | 2018-03-09 | 2025-02-12 | Recurium IP Holdings, LLC | Substituted 1,2-dihydro-3h-pyrazolo[3,4-d]pyrimidin-3-ones |
| SG11202010636VA (en) | 2018-05-04 | 2020-11-27 | Incyte Corp | Solid forms of an fgfr inhibitor and processes for preparing the same |
| BR112020022373A2 (en) | 2018-05-04 | 2021-02-02 | Incyte Corporation | salts of a fgfr inhibitor |
| IL278889B2 (en) | 2018-06-01 | 2025-11-01 | Incyte Corp | Dosage regimen for the treatment of PI3K-related disorders |
| US12324807B2 (en) | 2018-06-01 | 2025-06-10 | Cornell University | Combination therapy for PI3K-associated disease or disorder |
| US20200129485A1 (en) | 2018-09-19 | 2020-04-30 | Forma Therapeutics, Inc. | Treating sickle cell disease with a pyruvate kinase r activating compound |
| CN113226356B (en) | 2018-09-19 | 2025-03-04 | 诺沃挪第克健康护理股份公司 | Pyruvate kinase R |
| KR102195348B1 (en) | 2018-11-15 | 2020-12-24 | 에이치케이이노엔 주식회사 | Novel compound as a protein kinase inhibitor, and the pharmaceutical composition comprising thereof |
| WO2020185532A1 (en) | 2019-03-08 | 2020-09-17 | Incyte Corporation | Methods of treating cancer with an fgfr inhibitor |
| WO2021007269A1 (en) | 2019-07-09 | 2021-01-14 | Incyte Corporation | Bicyclic heterocycles as fgfr inhibitors |
| CN114615977B (en) | 2019-09-19 | 2025-01-14 | 诺沃挪第克健康护理股份公司 | Pyruvate Kinase R (PKR) activating compositions |
| WO2021067374A1 (en) | 2019-10-01 | 2021-04-08 | Incyte Corporation | Bicyclic heterocycles as fgfr inhibitors |
| WO2021076602A1 (en) | 2019-10-14 | 2021-04-22 | Incyte Corporation | Bicyclic heterocycles as fgfr inhibitors |
| WO2021076728A1 (en) | 2019-10-16 | 2021-04-22 | Incyte Corporation | Bicyclic heterocycles as fgfr inhibitors |
| JP7720840B2 (en) | 2019-12-04 | 2025-08-08 | インサイト・コーポレイション | Tricyclic heterocycles as FGFR inhibitors |
| CN115151539A (en) | 2019-12-04 | 2022-10-04 | 因赛特公司 | Derivatives of FGFR Inhibitors |
| WO2021146424A1 (en) | 2020-01-15 | 2021-07-22 | Incyte Corporation | Bicyclic heterocycles as fgfr inhibitors |
| KR102516260B1 (en) * | 2020-07-10 | 2023-03-31 | 울산과학기술원 | Compounds as a TRAP1 selective inhibitor, and composition for preventing or treating cancer comprising the same |
| EP4267573A1 (en) | 2020-12-23 | 2023-11-01 | Genzyme Corporation | Deuterated colony stimulating factor-1 receptor (csf-1r) inhibitors |
| US12128035B2 (en) | 2021-03-19 | 2024-10-29 | Novo Nordisk Health Care Ag | Activating pyruvate kinase R |
| US12065494B2 (en) | 2021-04-12 | 2024-08-20 | Incyte Corporation | Combination therapy comprising an FGFR inhibitor and a Nectin-4 targeting agent |
| AR126102A1 (en) | 2021-06-09 | 2023-09-13 | Incyte Corp | TRICYCLIC HETEROCYCLES AS FGFR INHIBITORS |
| CA3220155A1 (en) | 2021-06-09 | 2022-12-15 | Incyte Corporation | Tricyclic heterocycles as fgfr inhibitors |
| CN115925724B (en) * | 2022-11-16 | 2024-09-06 | 成都中医药大学 | Adenine compound with anti-inflammatory effect and preparation method and application thereof |
| CN116332940B (en) * | 2023-02-14 | 2024-09-13 | 广西民族大学 | 7-Deazapurine derivative and preparation method and application thereof |
Citations (53)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4495190A (en) * | 1980-12-22 | 1985-01-22 | Astra Lakemedel Aktiebolag | Derivatives of guanine for combating herpes virus infections |
| US4547573A (en) * | 1983-12-02 | 1985-10-15 | Ici Pharma | Process for preparing cephalosporin derivatives |
| US4617304A (en) * | 1984-04-10 | 1986-10-14 | Merck & Co., Inc. | Purine derivatives |
| US4699877A (en) * | 1982-11-04 | 1987-10-13 | The Regents Of The University Of California | Methods and compositions for detecting human tumors |
| US4748177A (en) * | 1984-03-26 | 1988-05-31 | Warner-Lambert Company | Guanine derivatives |
| US4772606A (en) * | 1985-08-22 | 1988-09-20 | Warner-Lambert Company | Purine derivatives |
| US4774325A (en) * | 1984-09-20 | 1988-09-27 | Pierrel Spa | New 8-substituted nucleoside and purine derivatives, the process for the preparation thereof and the pharmaceutical compositions containing them |
| US4806642A (en) * | 1984-10-05 | 1989-02-21 | Warner-Lambert Company | Purine derivatives |
| US4918162A (en) * | 1986-05-06 | 1990-04-17 | The Regents Of The University Of California | Assays and antibodies for N-MYC proteins |
| US4921859A (en) * | 1983-10-31 | 1990-05-01 | Warner-Lambert Company | Purine derivatives |
| US4923885A (en) * | 1988-08-19 | 1990-05-08 | Merck & Co., Inc. | 5-amino-1-(4-naphthoylbenzyl)-1,2,3-triazole-4-carboxamides and analogs as antiproliferative agents |
| US4968603A (en) * | 1986-12-31 | 1990-11-06 | The Regents Of The University Of California | Determination of status in neoplastic disease |
| US5002950A (en) * | 1986-10-24 | 1991-03-26 | Warner-Lambert Co. | 7-deazaguanines as immunomodulators |
| US5098906A (en) * | 1983-10-31 | 1992-03-24 | Warner-Lambert Company | Purine derivatives |
| US5110818A (en) * | 1988-10-06 | 1992-05-05 | Ciba-Geigy Corporation | Anticonvulsive substituted-9-benzyl-9h-purines |
| US5204353A (en) * | 1987-04-07 | 1993-04-20 | Ciba-Geigy Corporation | 3-benzyl-3H-1,2,3-triazolo[4,5-d]pyrimidines, compositions thereof, and method of treating epilepsy therewith |
| US5217866A (en) * | 1985-03-15 | 1993-06-08 | Anti-Gene Development Group | Polynucleotide assay reagent and method |
| US5332744A (en) * | 1989-05-30 | 1994-07-26 | Merck & Co., Inc. | Substituted imidazo-fused 6-membered heterocycles as angiotensin II antagonists |
| US5369008A (en) * | 1991-05-24 | 1994-11-29 | Board Of Regents, The University Of Texas System | Methods for the detection of BCR-ABL and abnormal ABL proteins in leukemia patients |
| US5602156A (en) * | 1993-09-17 | 1997-02-11 | The United States Of America As Represented By The Department Of Health And Human Services | Method for inhibiting metalloproteinase expression |
| US5656629A (en) * | 1995-03-10 | 1997-08-12 | Sanofi Winthrop, Inc. | 6-substituted pyrazolo (3,4-d)pyrimidin-4-ones and compositions and methods of use thereof |
| US5789394A (en) * | 1992-12-23 | 1998-08-04 | Nguyen-Ba; Nghe | Anti-viral compounds |
| US5846749A (en) * | 1994-10-12 | 1998-12-08 | The Regents Of The University Of California | Quantitative measurement of tissue protein identified by immunohistochemistry and standardized protein determination |
| US5861503A (en) * | 1997-04-30 | 1999-01-19 | The Regents Of The University Of California | Process for producing 8-fluoropurines |
| US5917042A (en) * | 1994-02-04 | 1999-06-29 | Glaxo Wellcome Inc. | Process for the preparation of 2,5-diamino-4,6-dichloropyrimidine |
| US5932566A (en) * | 1994-06-16 | 1999-08-03 | Pfizer Inc. | Ansamycin derivatives as antioncogene and anticancer agents |
| US5994361A (en) * | 1994-06-22 | 1999-11-30 | Biochem Pharma | Substituted purinyl derivatives with immunomodulating activity |
| US6005107A (en) * | 1992-12-23 | 1999-12-21 | Biochem Pharma, Inc. | Antiviral compounds |
| US6025126A (en) * | 1991-10-28 | 2000-02-15 | Arch Development Corporation | Methods and compositions for the detection of chromosomal aberrations |
| US6143743A (en) * | 1997-07-03 | 2000-11-07 | Dupont Pharmaceuticals Company | Imidazopyrimidines and imidazopyridines for the treatment of neurological disorders |
| US6174875B1 (en) * | 1999-04-01 | 2001-01-16 | University Of Pittsburgh | Benzoquinoid ansamycins for the treatment of cardiac arrest and stroke |
| US6177460B1 (en) * | 1995-04-12 | 2001-01-23 | The Procter & Gamble Company | Method of treatment for cancer or viral infections |
| US6210974B1 (en) * | 1997-10-24 | 2001-04-03 | Oregon Health Sciences University | Compositions and methods for promoting nerve regeneration |
| US6333331B1 (en) * | 1994-08-01 | 2001-12-25 | The United States Of America As Represented By The Department Of Health And Human Services | Substituted O6-benzylguanines |
| US6369092B1 (en) * | 1998-11-23 | 2002-04-09 | Cell Pathways, Inc. | Method for treating neoplasia by exposure to substituted benzimidazole derivatives |
| US6444656B1 (en) * | 1992-12-23 | 2002-09-03 | Biochem Pharma, Inc. | Antiviral phosphonate nucleotides |
| US20020156277A1 (en) * | 2001-04-20 | 2002-10-24 | Fick David B. | Synthesis and methods of use of purine analogues and derivatives |
| US20020161014A1 (en) * | 2000-04-25 | 2002-10-31 | Chanchal Sadhu | Inhibitors of human phosphatidylinositol 3-kinase delta |
| US20030022864A1 (en) * | 2001-04-24 | 2003-01-30 | Ishaq Khalid S. | 9-[(5-dihydroxyboryl)-pentyl] purines, useful as an inhibitor of inflammatory cytokines |
| US20030078413A1 (en) * | 2001-09-12 | 2003-04-24 | Epoch Biosciences, Inc. | Process for the synthesis of pyrazolopyrimidines |
| US20030113331A1 (en) * | 1999-01-06 | 2003-06-19 | Brooks Peter C. | Method and composition for angiogenesis inhibition |
| US20040063668A1 (en) * | 2001-01-19 | 2004-04-01 | Jong-Ryoo Choi | Novel acyclic nucleoside phosphonate derivatives, salts thereof and process for the preparation of the same |
| US6723727B1 (en) * | 1996-12-20 | 2004-04-20 | Hoechst Aktiengesellschaft | Substituted purine derivatives, processes for their preparation, their use, and compositions comprising them |
| US20040097526A1 (en) * | 2001-01-10 | 2004-05-20 | Gillespie Roger John | Triazolo[4,5-d]pyrimidine derivatives and their use as purinergic receptor antagonists |
| US20040102458A1 (en) * | 2000-11-02 | 2004-05-27 | Gabriela Chiosis | Small molecule compositions for binding to hsp90 |
| US20040242490A1 (en) * | 2003-02-20 | 2004-12-02 | Brooks Peter C. | CLK-peptide and SLK-peptide |
| US20040241706A1 (en) * | 2002-09-13 | 2004-12-02 | Irm, Llc | Highly specific modulators of GTPases for target validation |
| US20050043239A1 (en) * | 2003-08-14 | 2005-02-24 | Jason Douangpanya | Methods of inhibiting immune responses stimulated by an endogenous factor |
| US20050049263A1 (en) * | 2001-10-30 | 2005-03-03 | Kasibhatla Srinivas Rao | Purine analogs having hsp90-inhibiting activity |
| US20050113340A1 (en) * | 2003-09-18 | 2005-05-26 | Conforma Therapeutics Corporation | 2-Aminopurine analogs having HSP90-inhibiting activity |
| US20050256183A1 (en) * | 2001-11-09 | 2005-11-17 | Kasibhatla Srinivas R | Hsp90-inhibiting zearalanol compounds and methods of producing and using same |
| US20060035909A1 (en) * | 2002-08-23 | 2006-02-16 | Kveta Fuksova | Azapurine derivatives |
| US7087754B2 (en) * | 2001-01-10 | 2006-08-08 | Vernalis Research Limited | Pyrazolo[3,4-d]pyrimidine derivatives and their use as purinergic receptor antagonists |
Family Cites Families (74)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB884151A (en) | 1957-12-06 | 1961-12-06 | Ciba Ltd | New pyrazolo-pyrimidines substituted in the pyrazole nucleus, and process for their manufacture |
| DE3578499D1 (en) * | 1984-02-02 | 1990-08-09 | Merck & Co Inc | 5- (AMINO OR SUBSTITUTED AMINO) -1,2,3-TRIAZOLE. |
| ZA857647B (en) * | 1984-10-12 | 1986-05-28 | Warner Lambert Co | Purine derivatives |
| IL76546A (en) | 1984-10-12 | 1988-12-30 | Warner Lambert Co | 9-(heteroarylalkyl)-6-purine(thi)one derivatives,their preparation and pharmaceutical compositions containing them |
| JO1406B1 (en) | 1984-11-02 | 1986-11-30 | سميث كلاين اند فرينش لابوراتوريز ليمتد | Chemical compounds |
| DE3687030T2 (en) | 1985-03-15 | 1993-03-11 | Eugene Stirchak | STEREO-REGULAR POLYNUCLEOTID-BINDING POLYMERS. |
| IL78643A0 (en) | 1985-05-02 | 1986-08-31 | Wellcome Found | Purine derivatives,their preparation and pharmaceutical compositions containing them |
| GB8515934D0 (en) | 1985-06-24 | 1985-07-24 | Janssen Pharmaceutica Nv | (4-piperidinomethyl and-hetero)purines |
| DE3623099C3 (en) * | 1986-07-09 | 1998-08-13 | Spoetzl Markus Dipl Ing Fh | Blow molding machine |
| JPH0821101B2 (en) | 1987-07-21 | 1996-03-04 | 三洋電機株式会社 | Coin sorter |
| SE8801729D0 (en) | 1988-05-06 | 1988-05-06 | Astra Ab | PURINE DERIVATIVES FOR USE IN THERAPY |
| EP0465297B1 (en) | 1990-07-04 | 1996-01-31 | Merrell Dow Pharmaceuticals Inc. | 9-Purinyl phosphonic acid derivatives |
| GB9020931D0 (en) | 1990-09-26 | 1990-11-07 | Wellcome Found | Heterocyclic compounds |
| ATE187173T1 (en) | 1991-03-05 | 1999-12-15 | Ajinomoto Kk | CYCLOPROPANE DERIVATIVE |
| US5939420A (en) | 1991-04-08 | 1999-08-17 | Duquesne University Of The Holy Ghost | Pyrrolo 2,3d!derivatives |
| CA2093403C (en) | 1992-04-08 | 1999-08-10 | Fumio Suzuki | Therapeutic agent for parkinson's disease |
| JPH0680670A (en) | 1992-09-03 | 1994-03-22 | Ajinomoto Co Inc | Cyclopropane derivative and its production |
| US5744492A (en) | 1993-09-17 | 1998-04-28 | United States Of America | Method for inhibiting angiogenesis |
| JP3769737B2 (en) | 1994-03-30 | 2006-04-26 | 味の素株式会社 | Cyclopropane derivative and process for producing the same |
| JPH0841035A (en) | 1994-08-05 | 1996-02-13 | Ajinomoto Co Inc | Cyclopropane derivative and its production |
| JPH08208687A (en) | 1994-11-25 | 1996-08-13 | Sankyo Co Ltd | Glyceryl oligonucleotide |
| JPH0920776A (en) | 1995-06-30 | 1997-01-21 | Nippon Paper Ind Co Ltd | New purine nucleoside derivative, its production and antiviral agent with the same as active ingredient |
| JPH09169758A (en) | 1995-10-18 | 1997-06-30 | Nippon Paper Ind Co Ltd | New purine nucleoside derivative, its production and antivirus using the same |
| JPH1025294A (en) | 1996-03-26 | 1998-01-27 | Akira Matsuda | Fused heterocyclic derivative, method for producing the same, and therapeutic agent for malignant tumor containing the same |
| CA2230808C (en) | 1996-07-03 | 2006-08-15 | Japan Energy Corporation | A novel purine derivative |
| US6284748B1 (en) | 1997-03-07 | 2001-09-04 | Metabasis Therapeutics, Inc. | Purine inhibitors of fructose 1,6-bisphosphatase |
| CA2287387C (en) | 1997-05-14 | 2010-02-16 | Sloan-Kettering Institute For Cancer Research | Methods and compositions for destruction of selected proteins |
| ES2253821T3 (en) | 1997-07-12 | 2006-06-01 | Cancer Research Technology Limited | PURINE DERIVATIVES QUINASE INHIBITORS THAT DEPENDS ON CYCLINE. |
| GB9717849D0 (en) | 1997-08-23 | 1997-10-29 | Seal Sands Chemicals Limited | Preparation of pyridene derivatives |
| AUPO912997A0 (en) | 1997-09-11 | 1997-10-02 | Commonwealth Scientific And Industrial Research Organisation | Antiviral agents |
| CA2309350C (en) | 1997-11-12 | 2007-04-03 | Mitsubishi Chemical Corporation | Purine derivatives and medicaments comprising the same as active ingredient |
| TW572758B (en) | 1997-12-22 | 2004-01-21 | Sumitomo Pharma | Type 2 helper T cell-selective immune response inhibitors comprising purine derivatives |
| AU3378599A (en) | 1998-04-03 | 1999-10-25 | University Of Pittsburgh | Benzoquinoid ansamycins for the treatment of cardiac arrest and stroke |
| US5985060A (en) | 1998-07-25 | 1999-11-16 | Breed Automotive Technology, Inc. | Gas generant compositions containing guanidines |
| SI20022A (en) | 1998-07-29 | 2000-02-29 | Kemijski inštitut | Alkyl substituted purine derivatives and their preparation |
| JP2000072773A (en) | 1998-08-28 | 2000-03-07 | Zeria Pharmaceut Co Ltd | Purine derivatives |
| CZ27399A3 (en) | 1999-01-26 | 2000-08-16 | Ústav Experimentální Botaniky Av Čr | Substituted nitrogen heterocyclic derivatives process of their preparation, the derivatives employed as medicaments, pharmaceutical composition and a compound pharmaceutical preparation in which these derivatives are comprised as well as use of these derivatives for preparing medicaments |
| RU2001124352A (en) | 1999-02-01 | 2004-02-20 | Си Ви Терапьютикс, Инк. (Us) | Purine inhibitors of cyclin-dependent kinase 2 and Ikb-alpha |
| FR2790702B1 (en) | 1999-03-08 | 2001-07-20 | Sidel Sa | MOLDING UNIT AND EXTRUSION-BLOWING MACHINE PROVIDED WITH SUCH A UNIT |
| AU4589800A (en) | 1999-05-05 | 2000-11-21 | Darwin Discovery Limited | 9-(1,2,3,4-tetrahydronaphthalen-1-yl)-1,9-dihydropurin-6-one derivatives as pde7inhibitors |
| US6660845B1 (en) * | 1999-11-23 | 2003-12-09 | Epoch Biosciences, Inc. | Non-aggregating, non-quenching oligomers comprising nucleotide analogues; methods of synthesis and use thereof |
| CO5271715A1 (en) | 1999-12-21 | 2003-04-30 | Sugen Inc | 7-AZA-INDOLIN-2-WAVES SUBSTITUTED IN 4 AND ITS USE AS PROTEIUNA QUINASA INHIBITORS |
| US20030187261A1 (en) | 2000-01-07 | 2003-10-02 | Libor Havlicek | Purine derivatives, process for their preparation and use thereof |
| EP1265913A4 (en) | 2000-03-24 | 2004-07-14 | Univ Duke | CHARACTERIZATION OF LIGANDS-GRP94 INTERACTIONS AND RELATED PURIFICATION, SCREENING AND PROCESSING METHODS |
| NZ522076A (en) | 2000-04-25 | 2005-08-26 | Icos Corp | Inhibitors of human phosphatidyl-inositol 3-kinase delta |
| WO2002002123A1 (en) | 2000-06-29 | 2002-01-10 | Trustees Of Boston University | Use of geldanamycin and related compounds for prophylaxis or treatment of fibrogenic disorders |
| WO2002009696A1 (en) | 2000-07-28 | 2002-02-07 | Sloan-Kettering Institute For Cancer Research | Methods for treating cell proliferative disorders and viral infections |
| US6613089B1 (en) | 2000-10-25 | 2003-09-02 | Sdgi Holdings, Inc. | Laterally expanding intervertebral fusion device |
| EP1337275A4 (en) | 2000-11-02 | 2007-05-09 | Sloan Kettering Inst Cancer | METHOD FOR IMPROVING THE EFFICACY OF CYTOTOXIC AGENTS BY USING HSP90 INHIBITORS |
| GB0100623D0 (en) | 2001-01-10 | 2001-02-21 | Vernalis Res Ltd | Chemical compounds IV |
| CA2440809A1 (en) | 2001-03-01 | 2002-09-12 | Conforma Therapeutics Corp. | Methods for treating genetically-defined proliferative disorders with hsp90 inhibitors |
| US6881567B2 (en) * | 2001-03-16 | 2005-04-19 | Her Majesty The Queen In Right Of Canada As Represented By The Minister Of Agricultural And Agri-Food Canada | Pre-emergent biological control agents |
| CA2444130C (en) | 2001-04-17 | 2010-12-21 | Sumitomo Pharmaceuticals Company, Limited | Adenine derivatives |
| WO2002088079A2 (en) | 2001-05-01 | 2002-11-07 | Bristol-Myers Squibb Company | Dual inhibitors of pde 7 and pde 4 |
| EP1404871A4 (en) | 2001-05-23 | 2006-10-04 | Sloan Kettering Inst Cancer | METHOD FOR THE TREATMENT OF CANCER IN CONNECTION WITH INCREASED HER 2 VALUES |
| PE20030008A1 (en) | 2001-06-19 | 2003-01-22 | Bristol Myers Squibb Co | DUAL INHIBITORS OF PDE 7 AND PDE 4 |
| EP1478322A4 (en) | 2001-06-22 | 2007-08-08 | Pharmasset Ltd | Beta-2'-OR 3'-HALONUCLEOSIDES |
| JP4381804B2 (en) | 2001-06-27 | 2009-12-09 | サイクラセル リミテッド | 2,6,9-substituted purine derivatives and their use in the treatment of proliferative diseases |
| KR20040054692A (en) | 2001-09-24 | 2004-06-25 | 콘포마 세러퓨틱스 코포레이션 | Process for preparing 17-allyl amino geldanamycin (17-aag) and other ansamycins |
| DE60230818D1 (en) | 2001-09-24 | 2009-02-26 | Imp Innovations Ltd | PYY3-36 FOR REDUCING OR PREVENTING GREASE LUBRICITY |
| JP2003113181A (en) | 2001-10-04 | 2003-04-18 | Sumika Fine Chemicals Co Ltd | Method for producing 6-halopurine |
| DE10152275A1 (en) | 2001-10-23 | 2003-04-30 | Bayer Ag | Process for the electrolysis of aqueous solutions of hydrogen chloride |
| EP1519735A4 (en) | 2001-12-12 | 2006-01-11 | Conforma Therapeutic Corp | TESTS AND ENFORCEMENTS FOR DETERMINING AND MODULATING HSP90 BINDING ACTIVITY |
| DE60327994D1 (en) | 2002-02-08 | 2009-07-30 | Conforma Therapeutics Corp | ANSAMYCINE WITH IMPROVED PHARMACOLOGICAL AND BIOLOGICAL PROPERTIES |
| JP2004018453A (en) | 2002-06-14 | 2004-01-22 | Sumika Fine Chemicals Co Ltd | Alkynylpurine compound and its manufacturing method |
| CN1688580A (en) | 2002-08-08 | 2005-10-26 | 记忆药物公司 | 2-Trifluoromethyl-6-aminopurine derivatives as phosphodiesterase 4 inhibitors |
| RU2005108601A (en) | 2002-09-26 | 2006-01-20 | Эл Джи Лайф Сайенсиз Лтд. (Kr) | (+) - TRANS-ISOMERS (1-PHOSPHONOMETOXY-2-ALKYL CYCLOPROPYL) METHYL NUCLEOSIDE DERIVATIVES, METHOD FOR PRODUCING THEIR STEREOISOMERS AND APPLICATION OF THESE COMPOUNDS AS A RELATED TO |
| EP1781659B1 (en) * | 2003-07-16 | 2008-11-05 | Janssen Pharmaceutica N.V. | Triazolopyrimidine derivatives as glycogen synthase kinase 3 inhibitors |
| PT1658292E (en) * | 2003-07-16 | 2007-10-25 | Janssen Pharmaceutica Nv | Triazolopyrimidine derivatives as glycogen synthase kinase 3 inhibitors |
| US7579332B2 (en) | 2003-07-30 | 2009-08-25 | Gilead Sciences, Inc. | Nucleobase phosphonate analogs for antiviral treatment |
| US20050054614A1 (en) | 2003-08-14 | 2005-03-10 | Diacovo Thomas G. | Methods of inhibiting leukocyte accumulation |
| EP1772454A4 (en) * | 2004-07-23 | 2009-02-25 | Mitsubishi Tanabe Pharma Corp | BICYCLIC NITROGEN COMPOUND |
| WO2007035963A2 (en) * | 2005-09-23 | 2007-03-29 | Conforma Therapeutics Corporation | Anti-tumor methods using multi drug resistance independent synthetic hsp90 inhibitors |
| US9809805B2 (en) | 2015-03-13 | 2017-11-07 | Acro Biotech Co., Ltd. | Zinc binding fusion protein of glutathione S-transferase and metallothionein |
-
2004
- 2004-09-20 BR BRPI0414533-0A patent/BRPI0414533A/en not_active IP Right Cessation
- 2004-09-20 AU AU2004274507A patent/AU2004274507B2/en not_active Ceased
- 2004-09-20 WO PCT/US2004/031248 patent/WO2005028434A2/en not_active Ceased
- 2004-09-20 US US10/946,637 patent/US7148228B2/en not_active Expired - Lifetime
- 2004-09-20 EP EP04788954A patent/EP1670802A4/en not_active Withdrawn
- 2004-09-20 EP EP09157018A patent/EP2145888A1/en not_active Withdrawn
- 2004-09-20 US US10/946,645 patent/US7138401B2/en not_active Expired - Lifetime
- 2004-09-20 EA EA200600594A patent/EA010160B1/en not_active IP Right Cessation
- 2004-09-20 US US10/945,851 patent/US7138402B2/en not_active Expired - Lifetime
- 2004-09-20 CA CA002539548A patent/CA2539548A1/en not_active Abandoned
- 2004-09-20 US US10/946,628 patent/US7129244B2/en not_active Expired - Lifetime
- 2004-09-20 CN CN2010101575972A patent/CN101906106A/en active Pending
- 2004-09-20 MX MXPA06002997A patent/MXPA06002997A/en active IP Right Grant
- 2004-09-20 KR KR1020067007478A patent/KR20060070572A/en not_active Ceased
- 2004-09-20 JP JP2006527162A patent/JP2007505933A/en not_active Ceased
- 2004-09-20 NZ NZ546611A patent/NZ546611A/en not_active IP Right Cessation
-
2006
- 2006-03-16 IL IL174375A patent/IL174375A0/en unknown
- 2006-03-27 NO NO20061396A patent/NO20061396L/en not_active Application Discontinuation
- 2006-09-12 US US11/531,218 patent/US20070173483A1/en not_active Abandoned
- 2006-09-12 US US11/531,221 patent/US20070185064A1/en not_active Abandoned
- 2006-09-29 US US11/537,017 patent/US20070111997A1/en not_active Abandoned
- 2006-09-29 US US11/537,002 patent/US20070111996A1/en not_active Abandoned
-
2010
- 2010-06-30 AU AU2010202750A patent/AU2010202750B2/en not_active Ceased
Patent Citations (57)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4495190A (en) * | 1980-12-22 | 1985-01-22 | Astra Lakemedel Aktiebolag | Derivatives of guanine for combating herpes virus infections |
| US4699877A (en) * | 1982-11-04 | 1987-10-13 | The Regents Of The University Of California | Methods and compositions for detecting human tumors |
| US4921859A (en) * | 1983-10-31 | 1990-05-01 | Warner-Lambert Company | Purine derivatives |
| US5098906A (en) * | 1983-10-31 | 1992-03-24 | Warner-Lambert Company | Purine derivatives |
| US4547573A (en) * | 1983-12-02 | 1985-10-15 | Ici Pharma | Process for preparing cephalosporin derivatives |
| US4748177A (en) * | 1984-03-26 | 1988-05-31 | Warner-Lambert Company | Guanine derivatives |
| US4617304A (en) * | 1984-04-10 | 1986-10-14 | Merck & Co., Inc. | Purine derivatives |
| US4774325A (en) * | 1984-09-20 | 1988-09-27 | Pierrel Spa | New 8-substituted nucleoside and purine derivatives, the process for the preparation thereof and the pharmaceutical compositions containing them |
| US4806642A (en) * | 1984-10-05 | 1989-02-21 | Warner-Lambert Company | Purine derivatives |
| US5217866A (en) * | 1985-03-15 | 1993-06-08 | Anti-Gene Development Group | Polynucleotide assay reagent and method |
| US4772606A (en) * | 1985-08-22 | 1988-09-20 | Warner-Lambert Company | Purine derivatives |
| US4918162A (en) * | 1986-05-06 | 1990-04-17 | The Regents Of The University Of California | Assays and antibodies for N-MYC proteins |
| US5002950A (en) * | 1986-10-24 | 1991-03-26 | Warner-Lambert Co. | 7-deazaguanines as immunomodulators |
| US4968603A (en) * | 1986-12-31 | 1990-11-06 | The Regents Of The University Of California | Determination of status in neoplastic disease |
| US5204353A (en) * | 1987-04-07 | 1993-04-20 | Ciba-Geigy Corporation | 3-benzyl-3H-1,2,3-triazolo[4,5-d]pyrimidines, compositions thereof, and method of treating epilepsy therewith |
| US4923885A (en) * | 1988-08-19 | 1990-05-08 | Merck & Co., Inc. | 5-amino-1-(4-naphthoylbenzyl)-1,2,3-triazole-4-carboxamides and analogs as antiproliferative agents |
| US5110818A (en) * | 1988-10-06 | 1992-05-05 | Ciba-Geigy Corporation | Anticonvulsive substituted-9-benzyl-9h-purines |
| US5332744A (en) * | 1989-05-30 | 1994-07-26 | Merck & Co., Inc. | Substituted imidazo-fused 6-membered heterocycles as angiotensin II antagonists |
| US5369008A (en) * | 1991-05-24 | 1994-11-29 | Board Of Regents, The University Of Texas System | Methods for the detection of BCR-ABL and abnormal ABL proteins in leukemia patients |
| US6025126A (en) * | 1991-10-28 | 2000-02-15 | Arch Development Corporation | Methods and compositions for the detection of chromosomal aberrations |
| US6005107A (en) * | 1992-12-23 | 1999-12-21 | Biochem Pharma, Inc. | Antiviral compounds |
| US5955610A (en) * | 1992-12-23 | 1999-09-21 | Biochem Pharma, Inc. | Antiviral compounds |
| US5789394A (en) * | 1992-12-23 | 1998-08-04 | Nguyen-Ba; Nghe | Anti-viral compounds |
| US6444656B1 (en) * | 1992-12-23 | 2002-09-03 | Biochem Pharma, Inc. | Antiviral phosphonate nucleotides |
| US5602156A (en) * | 1993-09-17 | 1997-02-11 | The United States Of America As Represented By The Department Of Health And Human Services | Method for inhibiting metalloproteinase expression |
| US5917042A (en) * | 1994-02-04 | 1999-06-29 | Glaxo Wellcome Inc. | Process for the preparation of 2,5-diamino-4,6-dichloropyrimidine |
| US5932566A (en) * | 1994-06-16 | 1999-08-03 | Pfizer Inc. | Ansamycin derivatives as antioncogene and anticancer agents |
| US5994361A (en) * | 1994-06-22 | 1999-11-30 | Biochem Pharma | Substituted purinyl derivatives with immunomodulating activity |
| US6333331B1 (en) * | 1994-08-01 | 2001-12-25 | The United States Of America As Represented By The Department Of Health And Human Services | Substituted O6-benzylguanines |
| US5846749A (en) * | 1994-10-12 | 1998-12-08 | The Regents Of The University Of California | Quantitative measurement of tissue protein identified by immunohistochemistry and standardized protein determination |
| US5656629A (en) * | 1995-03-10 | 1997-08-12 | Sanofi Winthrop, Inc. | 6-substituted pyrazolo (3,4-d)pyrimidin-4-ones and compositions and methods of use thereof |
| US6177460B1 (en) * | 1995-04-12 | 2001-01-23 | The Procter & Gamble Company | Method of treatment for cancer or viral infections |
| US6723727B1 (en) * | 1996-12-20 | 2004-04-20 | Hoechst Aktiengesellschaft | Substituted purine derivatives, processes for their preparation, their use, and compositions comprising them |
| US5861503A (en) * | 1997-04-30 | 1999-01-19 | The Regents Of The University Of California | Process for producing 8-fluoropurines |
| US6262254B1 (en) * | 1997-04-30 | 2001-07-17 | The Regents Of Univ. Of California | 8-fluoropurine compounds |
| US6143743A (en) * | 1997-07-03 | 2000-11-07 | Dupont Pharmaceuticals Company | Imidazopyrimidines and imidazopyridines for the treatment of neurological disorders |
| US6210974B1 (en) * | 1997-10-24 | 2001-04-03 | Oregon Health Sciences University | Compositions and methods for promoting nerve regeneration |
| US6369092B1 (en) * | 1998-11-23 | 2002-04-09 | Cell Pathways, Inc. | Method for treating neoplasia by exposure to substituted benzimidazole derivatives |
| US20030113331A1 (en) * | 1999-01-06 | 2003-06-19 | Brooks Peter C. | Method and composition for angiogenesis inhibition |
| US6174875B1 (en) * | 1999-04-01 | 2001-01-16 | University Of Pittsburgh | Benzoquinoid ansamycins for the treatment of cardiac arrest and stroke |
| US20020161014A1 (en) * | 2000-04-25 | 2002-10-31 | Chanchal Sadhu | Inhibitors of human phosphatidylinositol 3-kinase delta |
| US20040102458A1 (en) * | 2000-11-02 | 2004-05-27 | Gabriela Chiosis | Small molecule compositions for binding to hsp90 |
| US20040097526A1 (en) * | 2001-01-10 | 2004-05-20 | Gillespie Roger John | Triazolo[4,5-d]pyrimidine derivatives and their use as purinergic receptor antagonists |
| US7087754B2 (en) * | 2001-01-10 | 2006-08-08 | Vernalis Research Limited | Pyrazolo[3,4-d]pyrimidine derivatives and their use as purinergic receptor antagonists |
| US20040063668A1 (en) * | 2001-01-19 | 2004-04-01 | Jong-Ryoo Choi | Novel acyclic nucleoside phosphonate derivatives, salts thereof and process for the preparation of the same |
| US20020156277A1 (en) * | 2001-04-20 | 2002-10-24 | Fick David B. | Synthesis and methods of use of purine analogues and derivatives |
| US20030022864A1 (en) * | 2001-04-24 | 2003-01-30 | Ishaq Khalid S. | 9-[(5-dihydroxyboryl)-pentyl] purines, useful as an inhibitor of inflammatory cytokines |
| US20030078413A1 (en) * | 2001-09-12 | 2003-04-24 | Epoch Biosciences, Inc. | Process for the synthesis of pyrazolopyrimidines |
| US20050049263A1 (en) * | 2001-10-30 | 2005-03-03 | Kasibhatla Srinivas Rao | Purine analogs having hsp90-inhibiting activity |
| US20050256183A1 (en) * | 2001-11-09 | 2005-11-17 | Kasibhatla Srinivas R | Hsp90-inhibiting zearalanol compounds and methods of producing and using same |
| US20060035909A1 (en) * | 2002-08-23 | 2006-02-16 | Kveta Fuksova | Azapurine derivatives |
| US20040241706A1 (en) * | 2002-09-13 | 2004-12-02 | Irm, Llc | Highly specific modulators of GTPases for target validation |
| US20040242490A1 (en) * | 2003-02-20 | 2004-12-02 | Brooks Peter C. | CLK-peptide and SLK-peptide |
| US20050043239A1 (en) * | 2003-08-14 | 2005-02-24 | Jason Douangpanya | Methods of inhibiting immune responses stimulated by an endogenous factor |
| US20050119282A1 (en) * | 2003-09-18 | 2005-06-02 | Conforma Therapeutics Corporation | Pyrazolopyrimidines and related analogs as HSP90-inhibitors |
| US20050113339A1 (en) * | 2003-09-18 | 2005-05-26 | Kasibhatla Srinivas R. | Triazolopyrimidines and related analogs as HSP90-inhibitors |
| US20050113340A1 (en) * | 2003-09-18 | 2005-05-26 | Conforma Therapeutics Corporation | 2-Aminopurine analogs having HSP90-inhibiting activity |
Cited By (23)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7544672B2 (en) | 2005-03-30 | 2009-06-09 | Conforma Therapeutics Corporation | Alkynyl pyrrolo[2,3-d]pyrimidines and related analogs as HSP90-inhibitors |
| US20060223797A1 (en) * | 2005-03-30 | 2006-10-05 | Conforma Therapeutics Corporation | Alkynyl pyrrolopyrimidines and related analogs as hsp90-inhibitors |
| US8093229B2 (en) | 2005-03-30 | 2012-01-10 | Conforma Therapeutics Corporation | Alkynyl pyrrolo[2,3-d]pyrimidines and related analogs as HSP90-inhibitors |
| US9359360B2 (en) | 2005-08-22 | 2016-06-07 | The Regents Of The University Of California | TLR agonists |
| US20090324551A1 (en) * | 2005-08-22 | 2009-12-31 | The Regents Of The University Of California Office Of Technology Transfer | Tlr agonists |
| US20110098294A1 (en) * | 2006-05-31 | 2011-04-28 | Carson Dennis A | Purine analogs |
| US8846697B2 (en) | 2006-05-31 | 2014-09-30 | The Regents Of The University Of California | Purine analogs |
| US9050376B2 (en) | 2007-02-07 | 2015-06-09 | The Regents Of The University Of California | Conjugates of synthetic TLR agonists and uses therefor |
| US8357374B2 (en) | 2007-02-07 | 2013-01-22 | The Regents Of The University Of California | Conjugates of synthetic TLR agonists and uses therefor |
| US8790655B2 (en) | 2007-02-07 | 2014-07-29 | The Regents Of The University Of California | Conjugates of synthetic TLR agonists and uses therefor |
| US20090202626A1 (en) * | 2008-02-07 | 2009-08-13 | Carson Dennis A | Treatment of bladder diseases with a tlr7 activator |
| WO2009139834A1 (en) * | 2008-05-13 | 2009-11-19 | Poniard Pharmaceuticals, Inc. | Bioactive compounds for treatment of cancer and neurodegenerative diseases |
| US20110124634A1 (en) * | 2008-05-13 | 2011-05-26 | Poniard Pharmaceuticals, Inc. | Bioactive compounds for treatment of cancer and neurodegenerative diseases |
| US9107919B2 (en) | 2009-02-06 | 2015-08-18 | Telormedix Sa | Pharmaceutical compositions comprising imidazoquinolin(amines) and derivatives thereof suitable for local administration |
| US9066940B2 (en) | 2009-02-06 | 2015-06-30 | Telormedix, Sa | Pharmaceutical compositions comprising imidazoquinolin(amines) and derivatives thereof suitable for local administration |
| US8729088B2 (en) | 2009-02-11 | 2014-05-20 | The Regents Of The University Of California | Toll-like receptor modulators and treatment of diseases |
| US20100210598A1 (en) * | 2009-02-11 | 2010-08-19 | Regents Of The University Of California, San Diego | Toll-like receptor modulators and treatment of diseases |
| WO2010117425A1 (en) * | 2009-03-31 | 2010-10-14 | Biogen Idec Ma Inc. | Certain substituted pyrimidines, pharmaceutical compositions thereof, and methods for their use |
| US9050319B2 (en) | 2010-04-30 | 2015-06-09 | Telormedix, Sa | Phospholipid drug analogs |
| US9173935B2 (en) | 2010-04-30 | 2015-11-03 | Telormedix Sa | Phospholipid drug analogs |
| US9173936B2 (en) | 2010-04-30 | 2015-11-03 | Telormedix Sa | Phospholipid drug analogs |
| US9180183B2 (en) | 2010-04-30 | 2015-11-10 | Telormedix Sa | Phospholipid drug analogs |
| US11697851B2 (en) | 2016-05-24 | 2023-07-11 | The Regents Of The University Of California | Early ovarian cancer detection diagnostic test based on mRNA isoforms |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2145888A1 (en) | 2010-01-20 |
| NO20061396L (en) | 2006-06-16 |
| US20050119282A1 (en) | 2005-06-02 |
| US20050107343A1 (en) | 2005-05-19 |
| US7138402B2 (en) | 2006-11-21 |
| US20070111996A1 (en) | 2007-05-17 |
| IL174375A0 (en) | 2006-08-01 |
| US7148228B2 (en) | 2006-12-12 |
| CN101906106A (en) | 2010-12-08 |
| EP1670802A4 (en) | 2010-07-07 |
| US20070185064A1 (en) | 2007-08-09 |
| EA010160B1 (en) | 2008-06-30 |
| BRPI0414533A (en) | 2006-11-07 |
| AU2004274507B2 (en) | 2010-08-19 |
| WO2005028434A2 (en) | 2005-03-31 |
| US7129244B2 (en) | 2006-10-31 |
| US20050113340A1 (en) | 2005-05-26 |
| AU2004274507A1 (en) | 2005-03-31 |
| US20050113339A1 (en) | 2005-05-26 |
| AU2010202750A1 (en) | 2010-07-22 |
| JP2007505933A (en) | 2007-03-15 |
| EA200600594A1 (en) | 2006-10-27 |
| KR20060070572A (en) | 2006-06-23 |
| MXPA06002997A (en) | 2007-02-08 |
| WO2005028434A3 (en) | 2006-03-23 |
| NZ546611A (en) | 2010-02-26 |
| CA2539548A1 (en) | 2005-03-31 |
| US20070111997A1 (en) | 2007-05-17 |
| US7138401B2 (en) | 2006-11-21 |
| AU2010202750B2 (en) | 2011-09-01 |
| EP1670802A2 (en) | 2006-06-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US7138402B2 (en) | Pyrrolopyrimidines and related analogs as HSP90-inhibitors | |
| US8093229B2 (en) | Alkynyl pyrrolo[2,3-d]pyrimidines and related analogs as HSP90-inhibitors | |
| US20250129085A1 (en) | Substituted imidazo[1,2-b]pyridazines as protein kinase inhibitors | |
| US20080096903A1 (en) | Sulfamoyl-containing derivatives and uses thereof | |
| US20070253896A1 (en) | 7,9-Dihydro-Purin-8-One and Related Analogs as HSP90-Inhibitors | |
| SK3542002A3 (en) | Pteridinones as kinase inhibitors | |
| US20120058997A1 (en) | Imidazo[1,2-b]pyridazine and pyrazolo[1,5-a]pyrimidine derivatives and their use as protein kinase inhibitors | |
| CN1882589B (en) | Novel heterocyclic compounds as HSP 90-inhibitors | |
| HK1140753A (en) | Deazapurine derivatives as hsp90-inhibitors |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| STCB | Information on status: application discontinuation |
Free format text: ABANDONED -- FAILURE TO RESPOND TO AN OFFICE ACTION |