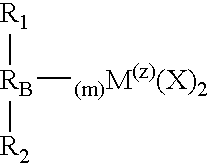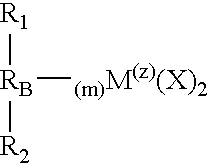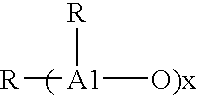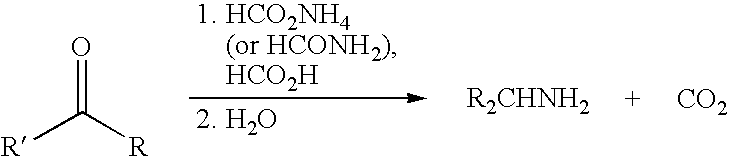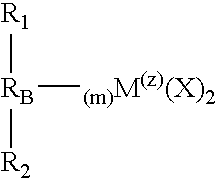US20040102311A1 - Bridged metallocene catalyst component, method of making, polyolefin catalyst having C1, C2, or Cs symmetry, methods of making, methods of polymerizing, olefins and products made thereof - Google Patents
Bridged metallocene catalyst component, method of making, polyolefin catalyst having C1, C2, or Cs symmetry, methods of making, methods of polymerizing, olefins and products made thereof Download PDFInfo
- Publication number
- US20040102311A1 US20040102311A1 US10/301,884 US30188402A US2004102311A1 US 20040102311 A1 US20040102311 A1 US 20040102311A1 US 30188402 A US30188402 A US 30188402A US 2004102311 A1 US2004102311 A1 US 2004102311A1
- Authority
- US
- United States
- Prior art keywords
- bonded
- heteroatoms
- heteroatom
- different
- same
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Abandoned
Links
- 239000003054 catalyst Substances 0.000 title claims abstract description 60
- 150000001336 alkenes Chemical class 0.000 title claims abstract description 12
- 238000000034 method Methods 0.000 title claims description 44
- 229920000098 polyolefin Polymers 0.000 title claims description 11
- 238000004519 manufacturing process Methods 0.000 title claims description 9
- 239000012968 metallocene catalyst Substances 0.000 title abstract description 17
- 230000000379 polymerizing effect Effects 0.000 title description 3
- 125000005842 heteroatom Chemical group 0.000 claims abstract description 69
- 229910052751 metal Inorganic materials 0.000 claims abstract description 30
- 239000002184 metal Substances 0.000 claims abstract description 30
- 125000000058 cyclopentadienyl group Chemical group C1(=CC=CC1)* 0.000 claims abstract description 21
- 239000000203 mixture Substances 0.000 claims description 45
- 150000001875 compounds Chemical class 0.000 claims description 31
- -1 lanthanide metals Chemical class 0.000 claims description 17
- 239000012190 activator Substances 0.000 claims description 13
- 229910052742 iron Inorganic materials 0.000 claims description 11
- 125000004429 atom Chemical group 0.000 claims description 9
- 229910052723 transition metal Inorganic materials 0.000 claims description 9
- 150000003624 transition metals Chemical class 0.000 claims description 9
- 229910052759 nickel Inorganic materials 0.000 claims description 8
- 229910052757 nitrogen Inorganic materials 0.000 claims description 8
- JRZJOMJEPLMPRA-UHFFFAOYSA-N olefin Natural products CCCCCCCC=C JRZJOMJEPLMPRA-UHFFFAOYSA-N 0.000 claims description 7
- 125000001183 hydrocarbyl group Chemical group 0.000 claims description 6
- 229910052747 lanthanoid Inorganic materials 0.000 claims description 6
- 229910052760 oxygen Inorganic materials 0.000 claims description 6
- 150000004820 halides Chemical class 0.000 claims description 5
- 239000000178 monomer Substances 0.000 claims description 5
- 229910052698 phosphorus Inorganic materials 0.000 claims description 5
- 229910052717 sulfur Inorganic materials 0.000 claims description 4
- 150000002736 metal compounds Chemical class 0.000 claims description 2
- 238000006116 polymerization reaction Methods 0.000 abstract description 19
- 239000003446 ligand Substances 0.000 description 23
- 0 [1*]C([2*])C Chemical compound [1*]C([2*])C 0.000 description 19
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 18
- 150000001450 anions Chemical class 0.000 description 14
- 229920000642 polymer Polymers 0.000 description 14
- 239000000243 solution Substances 0.000 description 14
- 230000015572 biosynthetic process Effects 0.000 description 9
- 238000003786 synthesis reaction Methods 0.000 description 9
- KWOLFJPFCHCOCG-UHFFFAOYSA-N Acetophenone Chemical compound CC(=O)C1=CC=CC=C1 KWOLFJPFCHCOCG-UHFFFAOYSA-N 0.000 description 8
- 150000001768 cations Chemical class 0.000 description 8
- 230000007935 neutral effect Effects 0.000 description 8
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 7
- 150000001412 amines Chemical class 0.000 description 7
- 238000006243 chemical reaction Methods 0.000 description 7
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 6
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 6
- CPOFMOWDMVWCLF-UHFFFAOYSA-N methyl(oxo)alumane Chemical compound C[Al]=O CPOFMOWDMVWCLF-UHFFFAOYSA-N 0.000 description 6
- ZVEJRZRAUYJYCO-UHFFFAOYSA-N CC1C2=C(C=CC=C2)C2=C1/C=C\C=C/2 Chemical compound CC1C2=C(C=CC=C2)C2=C1/C=C\C=C/2 ZVEJRZRAUYJYCO-UHFFFAOYSA-N 0.000 description 5
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 5
- 230000009467 reduction Effects 0.000 description 5
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 4
- ZYASLTYCYTYKFC-UHFFFAOYSA-N C=C1C2=C(C=CC=C2)C2=C1/C=C\C=C/2 Chemical compound C=C1C2=C(C=CC=C2)C2=C1/C=C\C=C/2 ZYASLTYCYTYKFC-UHFFFAOYSA-N 0.000 description 4
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 4
- 125000000217 alkyl group Chemical group 0.000 description 4
- 238000004821 distillation Methods 0.000 description 4
- 229910052809 inorganic oxide Inorganic materials 0.000 description 4
- 239000002685 polymerization catalyst Substances 0.000 description 4
- 239000002243 precursor Substances 0.000 description 4
- 150000003623 transition metal compounds Chemical class 0.000 description 4
- 239000004711 α-olefin Substances 0.000 description 4
- YBYIRNPNPLQARY-UHFFFAOYSA-N 1H-indene Natural products C1=CC=C2CC=CC2=C1 YBYIRNPNPLQARY-UHFFFAOYSA-N 0.000 description 3
- HVVZVBWIBBTXAJ-UHFFFAOYSA-N C=C1C=CC2=CC=CC=C12 Chemical compound C=C1C=CC2=CC=CC=C12 HVVZVBWIBBTXAJ-UHFFFAOYSA-N 0.000 description 3
- HBFWNTWSYDZULV-UHFFFAOYSA-N CC1C=CC2=C1CCCC2 Chemical compound CC1C=CC2=C1CCCC2 HBFWNTWSYDZULV-UHFFFAOYSA-N 0.000 description 3
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 3
- 239000005977 Ethylene Substances 0.000 description 3
- 239000002841 Lewis acid Substances 0.000 description 3
- 239000004743 Polypropylene Substances 0.000 description 3
- 229910052782 aluminium Inorganic materials 0.000 description 3
- 150000004985 diamines Chemical class 0.000 description 3
- 238000004992 fast atom bombardment mass spectroscopy Methods 0.000 description 3
- 238000010438 heat treatment Methods 0.000 description 3
- 150000002602 lanthanoids Chemical class 0.000 description 3
- 150000007517 lewis acids Chemical class 0.000 description 3
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 3
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 3
- 229920001155 polypropylene Polymers 0.000 description 3
- 239000000047 product Substances 0.000 description 3
- 150000003839 salts Chemical class 0.000 description 3
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 3
- WKBALTUBRZPIPZ-UHFFFAOYSA-N 2,6-di(propan-2-yl)aniline Chemical compound CC(C)C1=CC=CC(C(C)C)=C1N WKBALTUBRZPIPZ-UHFFFAOYSA-N 0.000 description 2
- BEZVGIHGZPLGBL-UHFFFAOYSA-N 2,6-diacetylpyridine Chemical compound CC(=O)C1=CC=CC(C(C)=O)=N1 BEZVGIHGZPLGBL-UHFFFAOYSA-N 0.000 description 2
- AQZWEFBJYQSQEH-UHFFFAOYSA-N 2-methyloxaluminane Chemical compound C[Al]1CCCCO1 AQZWEFBJYQSQEH-UHFFFAOYSA-N 0.000 description 2
- WSSSPWUEQFSQQG-UHFFFAOYSA-N 4-methyl-1-pentene Chemical compound CC(C)CC=C WSSSPWUEQFSQQG-UHFFFAOYSA-N 0.000 description 2
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 2
- WLZJVEGXORNWGV-UHFFFAOYSA-N C=C1C=CC(C(C)(C)C)=C1 Chemical compound C=C1C=CC(C(C)(C)C)=C1 WLZJVEGXORNWGV-UHFFFAOYSA-N 0.000 description 2
- PGTKVMVZBBZCKQ-UHFFFAOYSA-N C=C1C=CC=C1 Chemical compound C=C1C=CC=C1 PGTKVMVZBBZCKQ-UHFFFAOYSA-N 0.000 description 2
- LNCBEQDVIBTVGB-UHFFFAOYSA-N CC(=N)C1=CC=CC(C(C)=N)=N1.CC(=O)C1=CC=CC(C(C)=O)=N1.O=C=O Chemical compound CC(=N)C1=CC=CC(C(C)=N)=N1.CC(=O)C1=CC=CC(C(C)=O)=N1.O=C=O LNCBEQDVIBTVGB-UHFFFAOYSA-N 0.000 description 2
- TZJURNZIVRWFDW-UHFFFAOYSA-N CC1C=CC(C(C)(C)C)=C1 Chemical compound CC1C=CC(C(C)(C)C)=C1 TZJURNZIVRWFDW-UHFFFAOYSA-N 0.000 description 2
- QVRBGKYLLCLCHL-UHFFFAOYSA-N CC1C=CC=C1 Chemical compound CC1C=CC=C1 QVRBGKYLLCLCHL-UHFFFAOYSA-N 0.000 description 2
- ZHNUHDYFZUAESO-UHFFFAOYSA-N Formamide Chemical compound NC=O ZHNUHDYFZUAESO-UHFFFAOYSA-N 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- CPLXHLVBOLITMK-UHFFFAOYSA-N Magnesium oxide Chemical compound [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 description 2
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 2
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 2
- 238000003833 Wallach reaction Methods 0.000 description 2
- MCMNRKCIXSYSNV-UHFFFAOYSA-N Zirconium dioxide Chemical compound O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 description 2
- 229960000583 acetic acid Drugs 0.000 description 2
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 2
- 229910052768 actinide Inorganic materials 0.000 description 2
- 150000001255 actinides Chemical class 0.000 description 2
- 238000005804 alkylation reaction Methods 0.000 description 2
- AZDRQVAHHNSJOQ-UHFFFAOYSA-N alumane Chemical class [AlH3] AZDRQVAHHNSJOQ-UHFFFAOYSA-N 0.000 description 2
- VZTDIZULWFCMLS-UHFFFAOYSA-N ammonium formate Chemical compound [NH4+].[O-]C=O VZTDIZULWFCMLS-UHFFFAOYSA-N 0.000 description 2
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 125000004432 carbon atom Chemical group C* 0.000 description 2
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 2
- 125000002091 cationic group Chemical group 0.000 description 2
- 150000001805 chlorine compounds Chemical group 0.000 description 2
- 238000001816 cooling Methods 0.000 description 2
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N diphenyl Chemical compound C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 description 2
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 2
- 238000000605 extraction Methods 0.000 description 2
- 239000012362 glacial acetic acid Substances 0.000 description 2
- 150000002367 halogens Chemical group 0.000 description 2
- 150000004678 hydrides Chemical class 0.000 description 2
- 229910052739 hydrogen Inorganic materials 0.000 description 2
- 239000001257 hydrogen Substances 0.000 description 2
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 2
- 150000002466 imines Chemical class 0.000 description 2
- 150000002576 ketones Chemical class 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 229910052573 porcelain Inorganic materials 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- QQONPFPTGQHPMA-UHFFFAOYSA-N propylene Natural products CC=C QQONPFPTGQHPMA-UHFFFAOYSA-N 0.000 description 2
- 125000004805 propylene group Chemical group [H]C([H])([H])C([H])([*:1])C([H])([H])[*:2] 0.000 description 2
- 239000011541 reaction mixture Substances 0.000 description 2
- 239000000377 silicon dioxide Substances 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 125000004209 (C1-C8) alkyl group Chemical group 0.000 description 1
- IDHCQGUWHXGMQW-UHFFFAOYSA-N 1-(2-acetylpyridin-3-yl)ethanone Chemical compound CC(=O)C1=CC=CN=C1C(C)=O IDHCQGUWHXGMQW-UHFFFAOYSA-N 0.000 description 1
- LIKMAJRDDDTEIG-UHFFFAOYSA-N 1-hexene Chemical compound CCCCC=C LIKMAJRDDDTEIG-UHFFFAOYSA-N 0.000 description 1
- YEHGSOZIZRABBU-UHFFFAOYSA-N 1-phenylethanamine;hydrochloride Chemical compound [Cl-].CC([NH3+])C1=CC=CC=C1 YEHGSOZIZRABBU-UHFFFAOYSA-N 0.000 description 1
- RQEUFEKYXDPUSK-UHFFFAOYSA-N 1-phenylethylamine Chemical compound CC(N)C1=CC=CC=C1 RQEUFEKYXDPUSK-UHFFFAOYSA-N 0.000 description 1
- 238000005160 1H NMR spectroscopy Methods 0.000 description 1
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- ATRRKUHOCOJYRX-UHFFFAOYSA-N Ammonium bicarbonate Chemical compound [NH4+].OC([O-])=O ATRRKUHOCOJYRX-UHFFFAOYSA-N 0.000 description 1
- BTBUEUYNUDRHOZ-UHFFFAOYSA-N Borate Chemical compound [O-]B([O-])[O-] BTBUEUYNUDRHOZ-UHFFFAOYSA-N 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-M Bromide Chemical compound [Br-] CPELXLSAUQHCOX-UHFFFAOYSA-M 0.000 description 1
- NVPUVWBDTWBFRF-DNHJVCTKSA-N C/C(=N\C1=C(C(C)C)C=CC=C1C(C)C)C1=CC=CC(/C(C)=N/C2=C(C(C)C)C=CC=C2C(C)C)=N1 Chemical compound C/C(=N\C1=C(C(C)C)C=CC=C1C(C)C)C1=CC=CC(/C(C)=N/C2=C(C(C)C)C=CC=C2C(C)C)=N1 NVPUVWBDTWBFRF-DNHJVCTKSA-N 0.000 description 1
- XQWTYSRPLULDBC-UOSOPFLXSA-N C/C(=N\C1C=CC2=CC=CC=C21)C1=CC=CC(/C(C)=N/C2C=CC3=CC=CC=C32)=N1 Chemical compound C/C(=N\C1C=CC2=CC=CC=C21)C1=CC=CC(/C(C)=N/C2C=CC3=CC=CC=C32)=N1 XQWTYSRPLULDBC-UOSOPFLXSA-N 0.000 description 1
- WTIGZNOJKLRBHI-HNZPQQFJSA-L CC1=N(C2=C(C(C)C)C=CC=C2C(C)C)[Fe]2(Cl)(Cl)N3=C(C=CC=C13)/C(C)=N\2C1=C(C(C)C)C=CC=C1C(C)C Chemical compound CC1=N(C2=C(C(C)C)C=CC=C2C(C)C)[Fe]2(Cl)(Cl)N3=C(C=CC=C13)/C(C)=N\2C1=C(C(C)C)C=CC=C1C(C)C WTIGZNOJKLRBHI-HNZPQQFJSA-L 0.000 description 1
- VYUKLQMLJDFRTQ-UHFFFAOYSA-N CC1=N(C2C=CC3=CC=CC=C32)C2(C)(C)N3=C(C=CC=C13)/C(C)=N\2C1C=CC2=CC=CC=C21 Chemical compound CC1=N(C2C=CC3=CC=CC=C32)C2(C)(C)N3=C(C=CC=C13)/C(C)=N\2C1C=CC2=CC=CC=C21 VYUKLQMLJDFRTQ-UHFFFAOYSA-N 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 239000004215 Carbon black (E152) Substances 0.000 description 1
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 1
- BDAGIHXWWSANSR-UHFFFAOYSA-M Formate Chemical compound [O-]C=O BDAGIHXWWSANSR-UHFFFAOYSA-M 0.000 description 1
- 239000005909 Kieselgur Substances 0.000 description 1
- 239000002879 Lewis base Substances 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- 229910002651 NO3 Inorganic materials 0.000 description 1
- NHNBFGGVMKEFGY-UHFFFAOYSA-N Nitrate Chemical compound [O-][N+]([O-])=O NHNBFGGVMKEFGY-UHFFFAOYSA-N 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- 239000011954 Ziegler–Natta catalyst Substances 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 238000012644 addition polymerization Methods 0.000 description 1
- 125000003342 alkenyl group Chemical group 0.000 description 1
- 150000004703 alkoxides Chemical class 0.000 description 1
- 125000005234 alkyl aluminium group Chemical group 0.000 description 1
- 125000002877 alkyl aryl group Chemical group 0.000 description 1
- 230000029936 alkylation Effects 0.000 description 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 239000001099 ammonium carbonate Substances 0.000 description 1
- 235000012501 ammonium carbonate Nutrition 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 150000001449 anionic compounds Chemical class 0.000 description 1
- 239000011260 aqueous acid Substances 0.000 description 1
- 229910052786 argon Inorganic materials 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 1
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 1
- 239000004305 biphenyl Substances 0.000 description 1
- 235000010290 biphenyl Nutrition 0.000 description 1
- 150000001642 boronic acid derivatives Chemical class 0.000 description 1
- 239000006227 byproduct Substances 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- JYYOBHFYCIDXHH-UHFFFAOYSA-N carbonic acid;hydrate Chemical compound O.OC(O)=O JYYOBHFYCIDXHH-UHFFFAOYSA-N 0.000 description 1
- 150000001728 carbonyl compounds Chemical class 0.000 description 1
- 150000007942 carboxylates Chemical class 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- 239000004927 clay Substances 0.000 description 1
- 239000002734 clay mineral Substances 0.000 description 1
- 229910017052 cobalt Inorganic materials 0.000 description 1
- 239000010941 cobalt Substances 0.000 description 1
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 1
- IBZAAOVLBXXVHW-UHFFFAOYSA-N cobalt;ethene Chemical group [Co].C=C IBZAAOVLBXXVHW-UHFFFAOYSA-N 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 238000012718 coordination polymerization Methods 0.000 description 1
- 238000007334 copolymerization reaction Methods 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 125000004122 cyclic group Chemical group 0.000 description 1
- 125000002704 decyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- ZSWFCLXCOIISFI-UHFFFAOYSA-N endo-cyclopentadiene Natural products C1C=CC=C1 ZSWFCLXCOIISFI-UHFFFAOYSA-N 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- HHFAWKCIHAUFRX-UHFFFAOYSA-N ethoxide Chemical compound CC[O-] HHFAWKCIHAUFRX-UHFFFAOYSA-N 0.000 description 1
- 239000000284 extract Substances 0.000 description 1
- 239000012634 fragment Substances 0.000 description 1
- 150000004795 grignard reagents Chemical class 0.000 description 1
- 229910021482 group 13 metal Inorganic materials 0.000 description 1
- 229910052736 halogen Inorganic materials 0.000 description 1
- 125000003187 heptyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 229930195733 hydrocarbon Natural products 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 238000011065 in-situ storage Methods 0.000 description 1
- 125000003454 indenyl group Chemical group C1(C=CC2=CC=CC=C12)* 0.000 description 1
- 150000008040 ionic compounds Chemical class 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 229910052741 iridium Inorganic materials 0.000 description 1
- 150000002505 iron Chemical class 0.000 description 1
- WSSMOXHYUFMBLS-UHFFFAOYSA-L iron dichloride tetrahydrate Chemical compound O.O.O.O.[Cl-].[Cl-].[Fe+2] WSSMOXHYUFMBLS-UHFFFAOYSA-L 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- 150000007527 lewis bases Chemical class 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- 239000000395 magnesium oxide Substances 0.000 description 1
- NBTOZLQBSIZIKS-UHFFFAOYSA-N methoxide Chemical compound [O-]C NBTOZLQBSIZIKS-UHFFFAOYSA-N 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- CDHCCWRMWKZBGE-UHFFFAOYSA-N n-(1-phenylethyl)formamide Chemical compound O=CNC(C)C1=CC=CC=C1 CDHCCWRMWKZBGE-UHFFFAOYSA-N 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004433 nitrogen atom Chemical group N* 0.000 description 1
- 125000001400 nonyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 150000002902 organometallic compounds Chemical class 0.000 description 1
- 229910052762 osmium Inorganic materials 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 230000001590 oxidative effect Effects 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229910052763 palladium Inorganic materials 0.000 description 1
- NFHFRUOZVGFOOS-UHFFFAOYSA-N palladium;triphenylphosphane Chemical compound [Pd].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 NFHFRUOZVGFOOS-UHFFFAOYSA-N 0.000 description 1
- 125000000913 palmityl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 239000008188 pellet Substances 0.000 description 1
- ISWSIDIOOBJBQZ-UHFFFAOYSA-M phenolate Chemical compound [O-]C1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-M 0.000 description 1
- 229910052697 platinum Inorganic materials 0.000 description 1
- 239000011148 porous material Substances 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- OGHBATFHNDZKSO-UHFFFAOYSA-N propan-2-olate Chemical compound CC(C)[O-] OGHBATFHNDZKSO-UHFFFAOYSA-N 0.000 description 1
- 239000002516 radical scavenger Substances 0.000 description 1
- 229910052761 rare earth metal Inorganic materials 0.000 description 1
- 150000002910 rare earth metals Chemical class 0.000 description 1
- 238000010992 reflux Methods 0.000 description 1
- 229910052703 rhodium Inorganic materials 0.000 description 1
- 229910052707 ruthenium Inorganic materials 0.000 description 1
- 229910052706 scandium Inorganic materials 0.000 description 1
- SIXSYDAISGFNSX-UHFFFAOYSA-N scandium atom Chemical compound [Sc] SIXSYDAISGFNSX-UHFFFAOYSA-N 0.000 description 1
- 150000004760 silicates Chemical class 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 125000001424 substituent group Chemical group 0.000 description 1
- 125000000547 substituted alkyl group Chemical group 0.000 description 1
- 229910021653 sulphate ion Inorganic materials 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 150000007944 thiolates Chemical class 0.000 description 1
- XJDNKRIXUMDJCW-UHFFFAOYSA-J titanium tetrachloride Chemical compound Cl[Ti](Cl)(Cl)Cl XJDNKRIXUMDJCW-UHFFFAOYSA-J 0.000 description 1
- 239000012485 toluene extract Substances 0.000 description 1
- IMFACGCPASFAPR-UHFFFAOYSA-N tributylamine Chemical compound CCCCN(CCCC)CCCC IMFACGCPASFAPR-UHFFFAOYSA-N 0.000 description 1
- ITMCEJHCFYSIIV-UHFFFAOYSA-M triflate Chemical compound [O-]S(=O)(=O)C(F)(F)F ITMCEJHCFYSIIV-UHFFFAOYSA-M 0.000 description 1
- OBAJXDYVZBHCGT-UHFFFAOYSA-N tris(pentafluorophenyl)borane Chemical compound FC1=C(F)C(F)=C(F)C(F)=C1B(C=1C(=C(F)C(F)=C(F)C=1F)F)C1=C(F)C(F)=C(F)C(F)=C1F OBAJXDYVZBHCGT-UHFFFAOYSA-N 0.000 description 1
- 150000003746 yttrium Chemical class 0.000 description 1
- 239000010457 zeolite Substances 0.000 description 1
- QMBQEXOLIRBNPN-UHFFFAOYSA-L zirconocene dichloride Chemical compound [Cl-].[Cl-].[Zr+4].C=1C=C[CH-]C=1.C=1C=C[CH-]C=1 QMBQEXOLIRBNPN-UHFFFAOYSA-L 0.000 description 1
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J31/00—Catalysts comprising hydrides, coordination complexes or organic compounds
- B01J31/16—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes
- B01J31/18—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes containing nitrogen, phosphorus, arsenic or antimony as complexing atoms, e.g. in pyridine ligands, or in resonance therewith, e.g. in isocyanide ligands C=N-R or as complexed central atoms
- B01J31/1805—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes containing nitrogen, phosphorus, arsenic or antimony as complexing atoms, e.g. in pyridine ligands, or in resonance therewith, e.g. in isocyanide ligands C=N-R or as complexed central atoms the ligands containing nitrogen
- B01J31/181—Cyclic ligands, including e.g. non-condensed polycyclic ligands, comprising at least one complexing nitrogen atom as ring member, e.g. pyridine
- B01J31/1815—Cyclic ligands, including e.g. non-condensed polycyclic ligands, comprising at least one complexing nitrogen atom as ring member, e.g. pyridine with more than one complexing nitrogen atom, e.g. bipyridyl, 2-aminopyridine
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J23/00—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group B01J21/00
- B01J23/70—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group B01J21/00 of the iron group metals or copper
- B01J23/74—Iron group metals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F15/00—Compounds containing elements of Groups 8, 9, 10 or 18 of the Periodic Table
- C07F15/02—Iron compounds
- C07F15/025—Iron compounds without a metal-carbon linkage
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F15/00—Compounds containing elements of Groups 8, 9, 10 or 18 of the Periodic Table
- C07F15/06—Cobalt compounds
- C07F15/065—Cobalt compounds without a metal-carbon linkage
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F17/00—Metallocenes
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F10/00—Homopolymers and copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2531/00—Additional information regarding catalytic systems classified in B01J31/00
- B01J2531/02—Compositional aspects of complexes used, e.g. polynuclearity
- B01J2531/0238—Complexes comprising multidentate ligands, i.e. more than 2 ionic or coordinative bonds from the central metal to the ligand, the latter having at least two donor atoms, e.g. N, O, S, P
- B01J2531/0258—Flexible ligands, e.g. mainly sp3-carbon framework as exemplified by the "tedicyp" ligand, i.e. cis-cis-cis-1,2,3,4-tetrakis(diphenylphosphinomethyl)cyclopentane
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2531/00—Additional information regarding catalytic systems classified in B01J31/00
- B01J2531/80—Complexes comprising metals of Group VIII as the central metal
- B01J2531/84—Metals of the iron group
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2531/00—Additional information regarding catalytic systems classified in B01J31/00
- B01J2531/80—Complexes comprising metals of Group VIII as the central metal
- B01J2531/84—Metals of the iron group
- B01J2531/842—Iron
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J31/00—Catalysts comprising hydrides, coordination complexes or organic compounds
- B01J31/16—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes
- B01J31/18—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes containing nitrogen, phosphorus, arsenic or antimony as complexing atoms, e.g. in pyridine ligands, or in resonance therewith, e.g. in isocyanide ligands C=N-R or as complexed central atoms
- B01J31/1845—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes containing nitrogen, phosphorus, arsenic or antimony as complexing atoms, e.g. in pyridine ligands, or in resonance therewith, e.g. in isocyanide ligands C=N-R or as complexed central atoms the ligands containing phosphorus
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J31/00—Catalysts comprising hydrides, coordination complexes or organic compounds
- B01J31/16—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes
- B01J31/22—Organic complexes
- B01J31/2204—Organic complexes the ligands containing oxygen or sulfur as complexing atoms
Definitions
- the present invention relates to catalyst components, to methods of making such components, to catalyst systems comprising such components, to methods of making such systems, to methods of polymerization using such systems, to polymer compositions, and to articles made from such polymer compositions.
- the present invention relates to olefin polymerization catalyst components, to methods of making such components, to olefin polymerization catalyst systems comprising such components, to methods of making such systems, to methods of polymerization using such systems, to polymer compositions, and to articles made from such polymer compositions.
- the present invention relates to chiral catalysts having C 1 , C 2 or C S symmetry, to methods of making such catalysts, methods of polymerization using such catalysts, to polymer compositions, and to articles made from such polymer compositions.
- the present invention relates to chiral polyolefin catalysts having C 1 , C 2 or C S symmetry, to methods of making such catalysts, to methods of polymerizing or copolymerizing olefins, to polyolefin compositions, and to articles made from such polyolefin compositions.
- metallocene catalysts for olefin polymerization comprise a metallocene and an aluminum alkyl component, with the transition metal compound having two or more cyclopentadienyl ring ligands. Accordingly, titanocenes, zirconocenes and hafnocenes have all been utilized as the transition metal component in such “metallocene” containing catalyst systems for the production of polyolefins.
- Metallocene catalysts are cocatalyzed with an alumoxane, rather than an aluminum alkyl, to provide a metallocene catalyst system of high activity for the production of polyolefins.
- Group 3 metal catalysts such as scandium and yttrium complexes; Rare Earth Metal catalysts such as lanthanide and actinide-based catalysts stabilized with substituted cyclopentadienyl ligands; cationic Group 4 metal complexes including carbon-based ligands (such as alkyl ligands, allyl ligands, Cp analogues), including nitrogen-based ligands (such as amide ligands either along or in combination with other ligands, amidinate ligands either alone or in combination with other ligands, and ⁇ -diketimate ligands), and including oxygen-based ligands (such as alkoxide ligands either alone or in combination with other ligands, bis-alkoxides with additional donors); neutral Group 4 metal complexes; Group 5 metal catalysts; Group 6 metal catalysts; Group 8 metal catalysts; Group 9 metal catalysts; Group 10 metal catalysts
- WO 98/30612 published Jul. 16, 1998, discloses selected iron complexes of 2,6-pyridinecarboxaldehydebis(imines) and 2,6-diacyclpyridinebis(imines) as catalysts for the polymerization of propylene.
- WO 99/12981 published Mar. 18, 1999, discloses catalyst complexes having a bridge comprising heteroatoms bridging R groups R 5 and R 7 , with these complexes taught as being useful “especially for polymerizing ethylene alone or for copolymerizing ethylene with higher 1-olefins” (page 2, lines 28-29).
- the bridged R groups R 5 and R 7 are independently selected from hydrogen, halogen, hydrocarbyl, substituted hydrocarbyl, heterohydrocarbyl or substituted heterohydrocarbyl.
- M is a metal
- each X is an atom or group covalently or ionically bonded to M and may be the same or different
- R 1 and R 2 may be the same or each may be different and are substituted or unsubstituted cyclopentadienyl rings
- R B is a structural bridge between the cyclopentadienyl rings R 1 and R 2 and imparts stereorigidity to the rings, and comprises at least one heteroatom bonded to M, with each of R 1 and R 2 bonded to the same or different heteroatom of R B which heteroatom is also bonded to M
- Z is the coordination number of M and is greater than or equal to 4
- m is the number of bonds between M and heteroatoms of R B and to impart sterorigidity m ⁇ 2; because the number of bonds around M cannot exceed its coordination number m+2 ⁇ Z
- R 1 , R 2 and R B selected to provide a catalyst component that is chiral with C 1 , C 2 or C S symmetry
- a method of making a bridged metallocene compound comprising contacting a metal compound of the formula M(X) 2 with a bridged compound R B of the formula
- a catalyst system comprising an activated bridged metallocene compound having the formula:
- a method of making a catalyst system comprising contacting an activator with a bridged metallocene compound having the formula:
- the bridged metallocene catalyst component of the present invention is represented by the following formula EQN. 1:
- M is a metal
- each X is an atom or group covalently or ionically bonded to M and may be the same or different
- R 1 and R 2 may be the same or each may be different and are substituted or unsubstituted cyclopentadienyl rings
- R B is a structural bridge between the cyclopentadienyl rings R 1 and R 2 and imparts stereorigidity to the rings, and comprises at least one heteroatom bonded to M, with each of R 1 and R 2 bonded to the same or different heteroatom of R B which heteroatom is also bonded to M
- Z is the coordination number of M and is greater than or equal to 4
- m is the number of bonds between M and the heteroatom(s) of R B and to impart sterorigidity m ⁇ 2; because the number of bonds around M cannot exceed its coordination number m+2 ⁇ Z; with R 1 , R 2 and R B selected to provide a catalyst component that is chiral with C 1 , C 2 or C S
- the metal M of the present invention may be any suitable metal useful as the metal component in metallocene catalysts.
- M may be selected from among any metal as is known in the prior art to be useful as the metal component in metallocene catalysts.
- M will be selected to have a coordination number Z that is at least equal to the number of substituents bonded to M, that is, m number of R B heteroatom-to-metal bonds plus 2 (for both X's).
- M is selected from among any transition metal. More preferably, M is selected from among transition metals, lanthanides and actinides. Even more preferably, M is selected from transition metals and lanthanides. Still more preferably, M is selected from among group 3d, 4d or 5d transition metals, specifically Fe, Co, Ni, Ru, Rh, Pd, Os, Ir and Pt. Yet more preferably, M is selected from among Fe, Co and Ni. Even still more preferably, M is selected from among Fe and Co.
- R 1 and R 2 may be the same or each may be different and may be generally described as being substituted or unsubstituted cyclopentadienyl rings.
- R 1 and R 2 may be selected from among any substituted or unsubstituted cyclopentadienyl ring as are known in the art to be useful in metallocene catalysts.
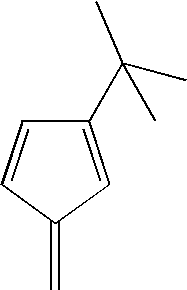
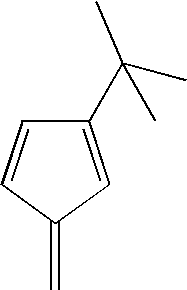
- hydrocarbon radicals suitable for use as R 1 and R 2 are shown in the Examples below.
- R 1 and R 2 may be described as a cyclopentadienyl ring of the form (C 5 (R′) 4 ), wherein each R′ may be the same or each may be different, and R′ is a hydrogen or a substituted or unsubstituted hydrocarbyl radical having 1-20 carbon atoms.
- Non-limiting examples of hydrocarbyl radicals suitable for use as R′ include unsubstituted and substituted alkyl, alkenyl, aryl, alkylaryl or arylalkyl radicals. More specific non-limiting examples of suitable hydrocarbyl radicals include unsubstituted and substituted methyl, ethyl, propyl, butyl, amyl, isoamyl, hexyl, isobutyl, heptyl, octyl, nonyl, decyl, cetyl, phenyl, methylene, ethylene, propylene, and other like groups.
- R B is a structural bridge between the cyclopentadienyl rings R 1 and R 2 and imparts stereorigidity to the rings, and comprises n heteroatoms (“HA”) bonded to M.
- HA heteroatoms
- An example of a suitable structural bridge R B is provided in the examples.
- Heteroatoms useful in structural bridge R B include any that can be coordinated to the metal M by a “dative” bond, that is, a bond formed by the donation of a lone pair of electrons from the heteroatom. Where R B comprises more than one heteroatom bonded to M, they may be the same heteroatom or different heteroatoms.
- suitable heteroatoms include O, N, S, and P.
- the heteroatoms are selected from among O, N, and P, and more preferably is N.
- R 1 is bonded to a heteroatom of R B which heteroatom is also bonded to M.
- R 2 is also bonded to a heteroatom of R B which heteroatom is also bonded to M.
- R 1 and R 2 may be bonded to the same heteroatom that is also bonded to M, or may be bonded to different heteroatoms which different heteroatoms are also bonded to M.
- R 1 , R 2 and R B are selected to provide a catalyst component that is chiral with C 1 , C 2 or C S symmetry.
- Each X may be any atom or group as are known to be utilized with metallocene catalysts, and is generally covalently or ionically bonded to M. Each X may be the same or different, although commonly each X is the same. As a non-limiting example, X may be selected from among halide, sulphate, nitrate, thiolate, thiocarboxylate, BF 4 ⁇ , PF 6 ⁇ , hydride, hydrocarbyloxide, carboxylate, substituted or unsubstituted hydrocarbyl, and heterohydrocarbyl.
- Non-limiting examples of such atoms or groups are chloride, bromide, methyl, ethyl, propyl, butyl, octyl decyl, phenyl, benzyl, methoxide, ethoxide, isopropoxide, toxylate, triflate, formate, acetate, phenoxide and benzoate.
- X is a halide or a C 1 to C 20 hydrocarbyl. More preferably, X is chloride.
- the bridged metallocene catalyst component is generally made by contacting a bridge intermediate with a compound of the form M(X) 2 . More details are provided in the Examples.
- the present invention further includes a catalyst system comprising one or more of the above described bridged metallocene catalyst components and one or more activators and/or cocatalysts (as described in greater detail below) or the reaction product of an activator and/or cocatalyst, such as for example methylaluminoxane (MAO) and optionally an alkylation/scavenging agent such as trialkylaluminum compound (TEAL).
- activator and/or cocatalysts such as for example methylaluminoxane (MAO) and optionally an alkylation/scavenging agent such as trialkylaluminum compound (TEAL).
- MAO methylaluminoxane
- TEAL trialkylaluminum compound
- the above described metallocene catalyst components may also be supported as is known in the metallocene art.
- Typical supports may be any support such as talc, an inorganic oxide, clay, and clay minerals, ion-exchanged layered compounds, diatomaceous earth, silicates, zeolites or a resinous support material such as a polyolefin.
- Specific inorganic oxides include silica and alumina, used alone or in combination with other inorganic oxides such as magnesia, titania, zirconia and the like.
- Non-metallocene transition metal compounds, such as titanium tetrachloride, are also incorporated into the supported catalyst component.
- the inorganic oxides used as support are characterized as having an average particle size ranging from 30-600 microns, desirably from 30-100 microns, a surface area of 50-1,000 square meters per gram, desirably from 100-400 square meters per gram, a pore volume of 0.5-3.5 cc/g, desirably from about 0.5-2 cc/g.
- the bridged metallocenes of the present invention may be used in combination with some form of activator in order to create an active catalyst system.
- activator is defined herein to be any compound or component, or combination of compounds or components, capable of enhancing the ability of one or more metallocenes to polymerize olefins to polyolefins.
- Alklyalumoxanes such as methylalumoxane (MAO) are commonly used as metallocene activators. Generally alkylalumoxanes contain about 5 to 40 of the repeating units.
- AIR 2 for linear species and for cyclic species
- R is a C1-C8 alkyl including mixed alkyls. Particularly desirable are the compounds in which R is methyl.
- Alumoxane solutions, particularly methylalumoxane solutions, may be obtained from commercial vendors as solutions having various concentrations. There are a variety of methods for preparing alumoxane, non-limiting examples of which are described in U.S. Pat. Nos.
- Ionizing activators may also be used to activate the bridged metallocenes.
- activators are neutral or ionic, or are compounds such as tri(n-butyl)ammonium tetrakis(pentaflurophenyl)borate, which ionize the neutral metallocene compound.
- ionizing compounds may contain an active proton, or some other cation associated with, but not coordinated or only loosely coordinated to, the remaining ion of the ionizing compound.
- Combinations of activators may also be used, for example, alumoxane and ionizing activators in combinations, see for example, WO 94/07928.
- ionic catalysts for coordination polymerization comprised of metallocene cations activated by non-coordinating anions appear in the early work in EP-A-0 277 003, EP-A-0 277 004 and U.S. Pat. No. 5,198,401 and WO-A-92/00333 (incorporated herein by reference). These teach a desirable method of preparation wherein metallocenes (bis Cp and monoCp) are protanated by an anion precursor such that an alkyl/hydride group is abstracted from a transition metal to make it both cationic and charge-balanced by the non-coordinating anion.
- Suitable ionic salts include tetrakis-substituted borate or aluminum salts having fluorided aryl-constituents such as phenyl, biphenyl and napthyl.
- noncoordinating anion means an anion which either does not coordinate to said cation or which is only weakly coordinated to said cation thereby remaining sufficiently labile to be displaced by a neutral Lewis base.
- “Compatible” noncoordinating anions are those which are not degraded to neutrality when the initially formed complex decomposes. Further, the anion will not transfer an anionic substituent or fragment to the cation so as to cause it to form a neutral four coordinate metallocene compound and a neutral by-product from the anion.
- ionizing ionic compounds not containing an active proton but capable of producing both the active metallocene cation and a noncoordinating anion. See, for example, EP-A-0 426 637 and EP-A-0 573 403 (incorporated herein by reference).
- An additional method of making the ionic catalysts uses ionizing anion precursors which are initially neutral Lewis acids but form the cation and anion upon ionizing reaction with the metallocene compounds, for example the use of tris(pentafluorophenyl) borane, see EP-A-0 520 732 (incorporated herein by reference).
- Ionic catalysts for addition polymerization can also be prepared by oxidation of the metal centers of transition metal compounds by anion precursors containing metallic oxidizing groups along with the anion groups, see EP-A-0 495 375 (incorporated herein by reference).
- the metal ligands include halogen moieties (for example, bis-cyclopentadienyl zirconium dichloride) which are not capable of ionizing abstraction under standard conditions, they can be converted via known alkylation reactions with organometallic compounds such as lithium or aluminum hydrides or alkyls, alkylalumoxanes, Grignard reagents, etc. See EP-A-O 500 944 and EP-Al-0 570 982 (incorporated herein by reference) for in situ processes describing the reaction of alkyl aluminum compounds with dihalo-substituted metallocene compounds prior to or with the addition of activating anionic compounds.
- organometallic compounds such as lithium or aluminum hydrides or alkyls, alkylalumoxanes, Grignard reagents, etc.
- NCA support methods generally comprise using neutral anion precursors that are sufficiently strong Lewis acids to react with the hydroxyl reactive functionalities present on the silica surface such that the Lewis acid becomes covalently bound.
- the activator for the metallocene supported catalyst composition is a NCA
- the NCA is first added to the support composition followed by the addition of the bridged metallocene catalyst.
- the activator is MAO
- the MAO and bridged metallocene catalyst are dissolved together in solution.
- the support is then contacted with the MAO/metallocene catalyst solution.
- the catalysts of the present invention can be used for the polymerization of any type of ⁇ -olefins or the copolymerization any mixture of ⁇ -olefins.
- the present catalyst is useful for catalyzing ethylene, propylene, butylene, pentene, hexene, 4-methylpentene and other ⁇ -alkenes having at least 2 carbon atoms, and also for mixtures thereof.
- the catalysts of the present invention are utilized for the polymerization of propylene to produce polypropylene, most preferably high crystallinity polypropylene.
- polymerization is accomplished by contacting together either ⁇ -olefin monomer or mixture of ⁇ -olefins in the presence of the above described catalyst system under polymerization conditions.
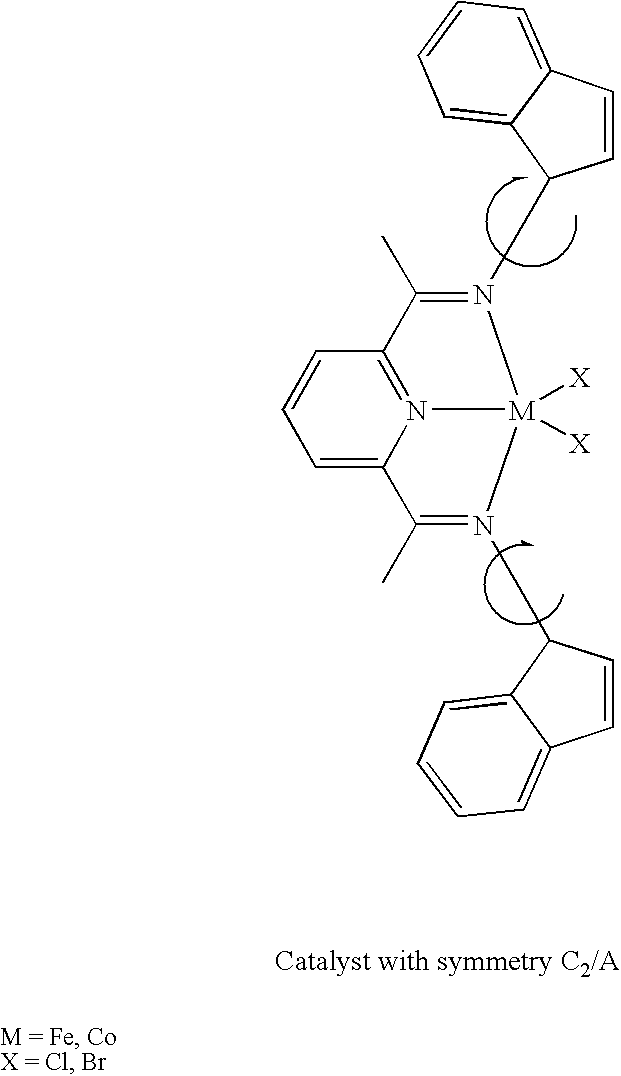
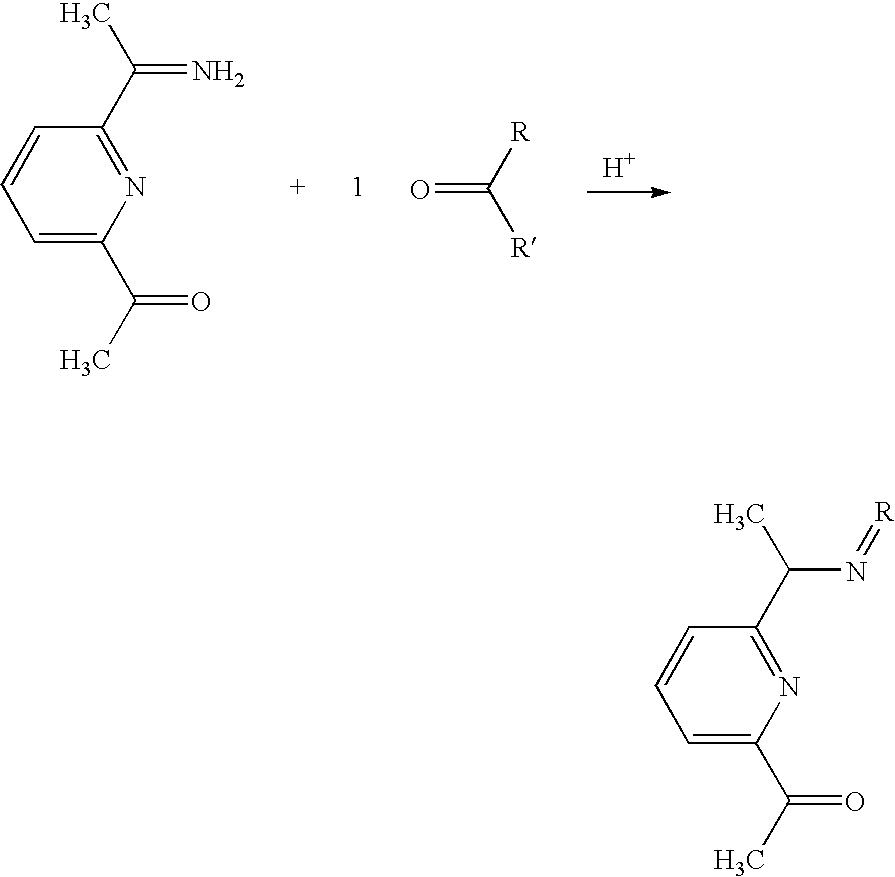
- This example shows creation of a ligand having C2/Class A symmetry.
- the same general synthesis is followed from Example 1, with the exception that the 2,6-diisopropylaniline is replaced with indene.
- Indene (18.4 mmol) is added to a solution of 2,6-diacetylpyridine (9.2 mmol) in absolute. A few drops of glacial acetic acid is added and the solution is refluxed for 48 h. Concentration of the solution to half volume and cooling to room temperature and filtered to give the intermediate shown below.
- a catalyst from the ligand of Example 3 (Intermediate with symmetry C2/Class A) is synthesized by using the same general synthesis as in Example 2, to provide the catalyst component shown below.
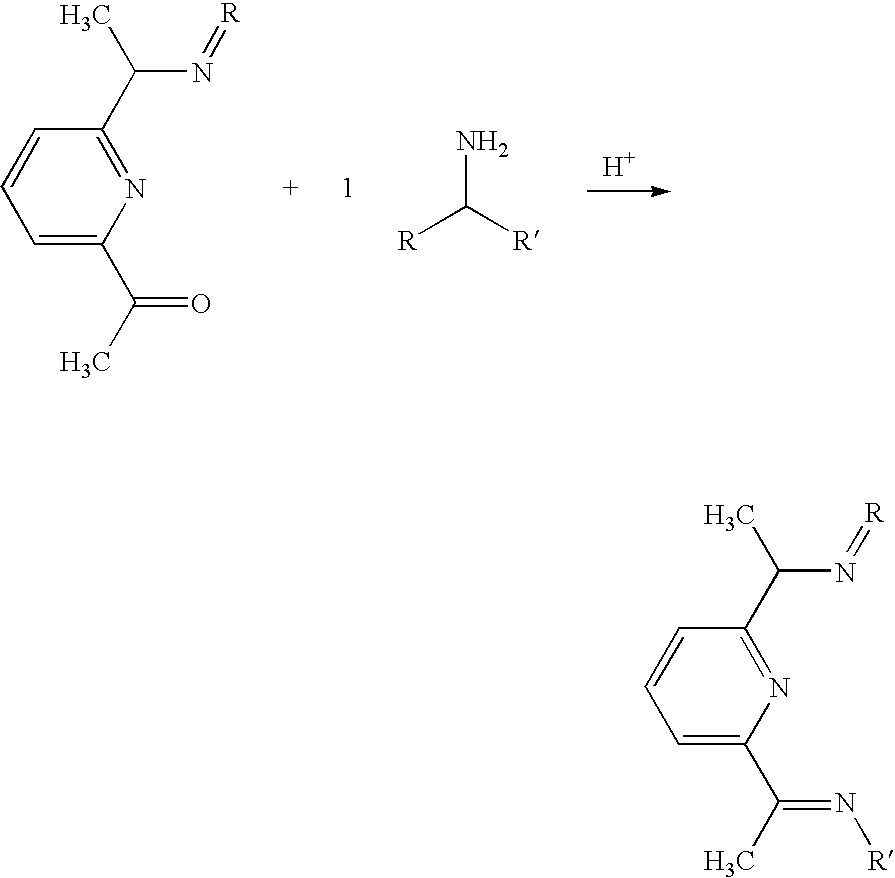
- This example shows creation of a ligand having C2/Class B symmetry.
- the first part of the synthesis for this ligand is different from that of Example 1 above.
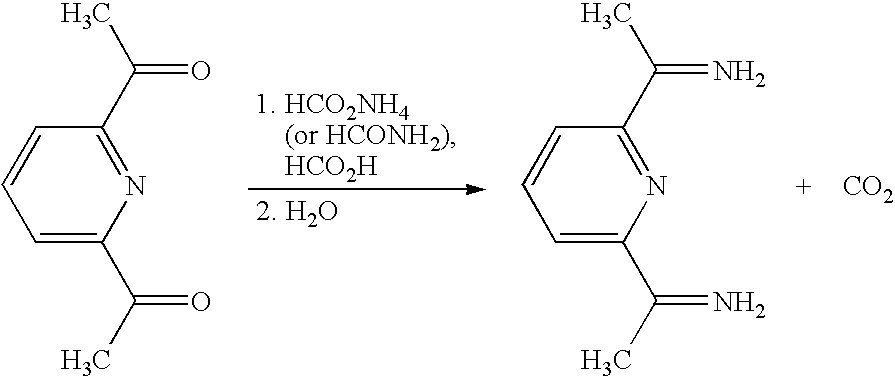
- the first part of the synthesis starts with the reduction of the diacetylpyridine to a diamine by using the Leuckart-Wallach reaction.
- scheme 1 a general reaction is shown for the reduction of a carbonyl to an amine.
- This example illustrates the reduction of a carbonyl to an amine, specifically, the synthesis of 1-phenylethylamine (Vogel's Practical Organic Chemistry including qualitative organic analysis, 4 th Ed, Furniss, B. S., et al., School of Chemistry Thames Polytechnic Longman Scientific and Technical, 1978).
- the crude (1-phenylethyl)formamide is transferred into the original reaction flask; the aqeous layer was extracted with two 20 ml portions of toluene, the toluene extracts were transferred to the flask, 75 ml of concentrated hydrochloric acid and a few chips of porous porcelain were added.
- the mixture was heated cautiously until about 40 ml of toluene was collected, and boiled gently under reflux for a further 40 minutes; hydrolysis proceeded rapidly to 1-phenylethylamine hydrochloride except for a small layer of unchanged acetophenone.
- the reaction mixture was allowed to cool, and the acetopenone was removed by extraction with four 20 ml portions of toluene.
- the aqueous acid solution was transferred to a 500 ml round-bottom flask equipped for stream distillation, a solution of 62.5 g of sodium hydroxide was cautiously added to 125 ml water, and steam distilled: the distillation flask was heated so that the volume remained nearly constant. Most of the amine is contained in the first 500 ml of distillate; the operation was stopped when the distillate was only faintly alkaline. The distillate was extracted with five 25, ml portions of toluene, the extract was dried with sodium hydroxide pellets and fractionally distilled. Toluene distilled over at 111 C., followed by the phylethylamine. The latter was collected as a fraction of b.p. 180-190 C. (the bulk of the product distiled at 184-186 C. (3); the yield was 43 g (59%).
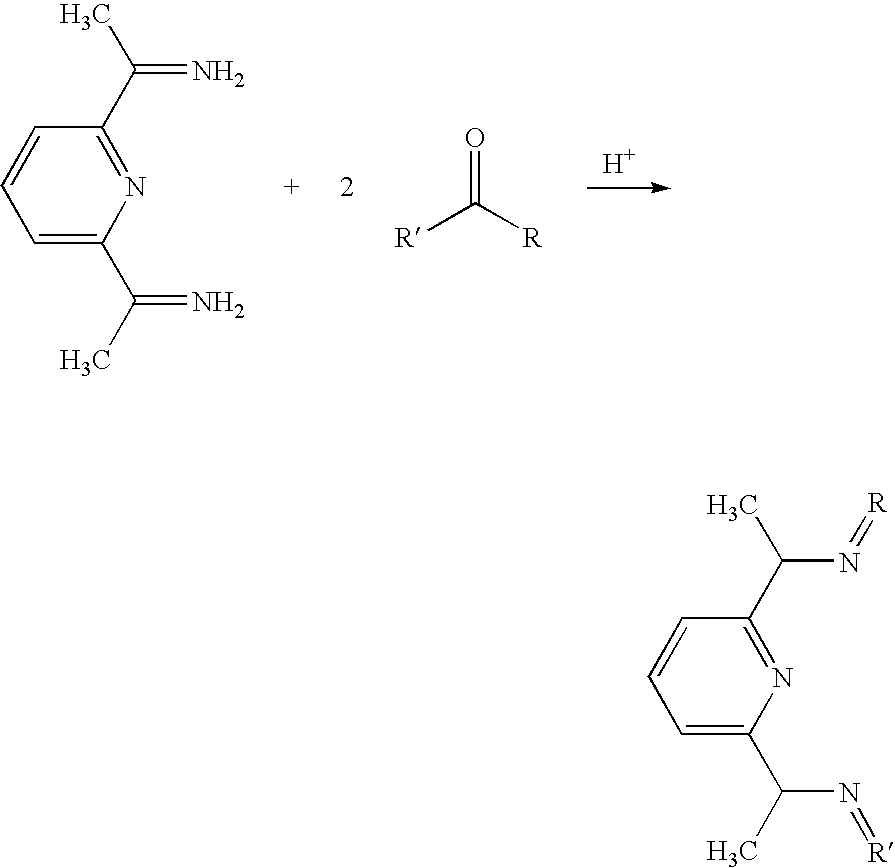
- This example provides a catalyst with symmetry C2/Class B by reacting the ligand of Example 9 with the desired metal.
- Example 11 the amine of Example 11 is reacted with a ketone to provide the R group double bond to the nitrogen.
- a catalyst is then synthesized according to the procedure described in Example 10.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Inorganic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Transition And Organic Metals Composition Catalysts For Addition Polymerization (AREA)
Abstract
Bridged metallocene catalyst component in which a bridge spans two cyclopentadienyl groups, which Cp groups are attached to same or different heteroatoms of the bridge, which heteroatoms are also bonded to the metal. A catalyst systems is made by contacting the bridged component with a cocatalyst. Polymerization of olefins in catalyzed by the system.
Description
- 1. Field of the Invention
- The present invention relates to catalyst components, to methods of making such components, to catalyst systems comprising such components, to methods of making such systems, to methods of polymerization using such systems, to polymer compositions, and to articles made from such polymer compositions. In another aspect, the present invention relates to olefin polymerization catalyst components, to methods of making such components, to olefin polymerization catalyst systems comprising such components, to methods of making such systems, to methods of polymerization using such systems, to polymer compositions, and to articles made from such polymer compositions. In even another aspect, the present invention, the present invention relates to chiral catalysts having C 1, C2 or CS symmetry, to methods of making such catalysts, methods of polymerization using such catalysts, to polymer compositions, and to articles made from such polymer compositions. In still another aspect, the present invention relates to chiral polyolefin catalysts having C1, C2 or CS symmetry, to methods of making such catalysts, to methods of polymerizing or copolymerizing olefins, to polyolefin compositions, and to articles made from such polyolefin compositions.
- 2. Description of the Related Art
- As is well known, various processes and catalysts exist for the production of polyolefins.
- First commercialized in the 1950's, the traditional Ziegler-Natta catalyst systems utilize a transition metal compound cocatalyzed by an aluminum alkyl.
- Commercialized in the 1980's, “metallocene” catalysts for olefin polymerization comprise a metallocene and an aluminum alkyl component, with the transition metal compound having two or more cyclopentadienyl ring ligands. Accordingly, titanocenes, zirconocenes and hafnocenes have all been utilized as the transition metal component in such “metallocene” containing catalyst systems for the production of polyolefins. Metallocene catalysts are cocatalyzed with an alumoxane, rather than an aluminum alkyl, to provide a metallocene catalyst system of high activity for the production of polyolefins.
- In addition to Ziegler-Natta catalysts and metallocene catalysts, a number of “non-metallocene” type catalysts have been suggested for the polymerization of olefins.
- Specifically, for example, in The Search for New-Generation Olefin Polymerization Catalysts: Life Beyond Metallocenes, Angew. Chem. Int. Ed. 1999, 38, 428-447, Britovsek et al. review a number of olefin catalyst systems, including: Group 3 metal catalysts such as scandium and yttrium complexes; Rare Earth Metal catalysts such as lanthanide and actinide-based catalysts stabilized with substituted cyclopentadienyl ligands; cationic Group 4 metal complexes including carbon-based ligands (such as alkyl ligands, allyl ligands, Cp analogues), including nitrogen-based ligands (such as amide ligands either along or in combination with other ligands, amidinate ligands either alone or in combination with other ligands, and β-diketimate ligands), and including oxygen-based ligands (such as alkoxide ligands either alone or in combination with other ligands, bis-alkoxides with additional donors); neutral Group 4 metal complexes; Group 5 metal catalysts; Group 6 metal catalysts; Group 8 metal catalysts; Group 9 metal catalysts; Group 10 metal catalysts; Group 13 metal catalysts.
- Additionally, in Iron and Cobalt Ethylene Polymerization Catalysts Bearing 2,6-Bis (Imino)Pyridyl Ligands; Synthesis, Structures, and Polymerization Studies, J. Am. Chem. Soc. 1999, 121, 8728-8740, Britovsek et al. disclose certain iron and cobalt catalysts for the polymerization of ethylene.
- WO 98/30612, published Jul. 16, 1998, discloses selected iron complexes of 2,6-pyridinecarboxaldehydebis(imines) and 2,6-diacyclpyridinebis(imines) as catalysts for the polymerization of propylene.
- WO 99/12981, published Mar. 18, 1999, discloses catalyst complexes having a bridge comprising heteroatoms bridging R groups R 5 and R7, with these complexes taught as being useful “especially for polymerizing ethylene alone or for copolymerizing ethylene with higher 1-olefins” (page 2, lines 28-29). The bridged R groups R5 and R7 are independently selected from hydrogen, halogen, hydrocarbyl, substituted hydrocarbyl, heterohydrocarbyl or substituted heterohydrocarbyl. There is no teaching or suggestion to make a chiral complex suitable for producing high tacticity, crystallinity polypropylene.
- The following patents disclose bridged metallocene catalyst systems: U.S. Pat. No. 5,145,819, issued Sep. 8, 1992 to Winter et al.; U.S. Pat. No. 5,158,920, issued Oct. 27, 1992 to Razavi; U.S. Pat. No. 5,243,001, issued Sep. 7, 1993 to Winter et al.; U.S. Pat. No. 6,002,033, issued Dec. 14, 1999 to Razavi et al.; U.S. Pat. No. 6,066,588, issued May 23, 2000, to Razavi et al.; U.S. Pat. No. 6,177,529 B1, issued Jan. 23, 2001, to Razavi et al.; U.S. Pat. No. 6,194,343 B1, issued Feb. 27, 2001 to Collins et al.; U.S. Pat. No. 6,211,110 B1, issued Apr. 3, 2001 to Santi et al.; and U.S. Pat. No. 6,268,518 B1, issued Jul. 31, 2001 to Resconi et al.
- However, in spite of the above advancements, there still exists a need in the art for catalyst compositions, methods of making such compositions, methods of polymerization using such compositions, to polymer compositions, and to articles made from such polymer compositions.
- There is another need in the art for catalyst compositions, methods of making such compositions, methods of polymerization using such compositions, to polymer compositions, and to articles made from such polymer compositions, which do not suffer from the disadvantages of the prior art compositions, products and methods.
- These and other needs in the art will become apparent to those of skill in the art upon review of this specification, including its drawings and claims.
- It is an object of the present invention to provide for catalyst compositions, methods of making such compositions, methods of polymerization using such compositions, to polymer compositions, and to articles made from such polymer compositions.
- It is another object of the present invention to provide for catalyst compositions, methods of making such compositions, methods of polymerization using such compositions, to polymer compositions, and to articles made from such polymer compositions, which do not suffer from the disadvantages of the prior art compositions, products and methods.
- These and other objects of the present invention will become apparent to those of skill in the art upon review of this specification, including its drawings and claims.
-
- wherein M is a metal; each X is an atom or group covalently or ionically bonded to M and may be the same or different; R 1 and R2 may be the same or each may be different and are substituted or unsubstituted cyclopentadienyl rings; RB is a structural bridge between the cyclopentadienyl rings R1 and R2 and imparts stereorigidity to the rings, and comprises at least one heteroatom bonded to M, with each of R1 and R2 bonded to the same or different heteroatom of RB which heteroatom is also bonded to M; Z is the coordination number of M and is greater than or equal to 4; m is the number of bonds between M and heteroatoms of RB and to impart sterorigidity m≧2; because the number of bonds around M cannot exceed its coordination number m+2≦Z; and with R1, R2 and RB selected to provide a catalyst component that is chiral with C1, C2 or CS symmetry.
-
- wherein M, X, R 1, R2, m and z are as defined above.
-
- wherein M, X, R 1, R2, m and z are as defined above.
-
- wherein M, X, R 1, R2, m and z are as defined above.
-
- wherein M, X, R 1, R2, m and z are as defined above.
- For all of the above embodiments, various further embodiments are provided by changing M, X, R 1, R2, m and z as described in the detailed description.
-
- wherein M is a metal; each X is an atom or group covalently or ionically bonded to M and may be the same or different; R 1 and R2 may be the same or each may be different and are substituted or unsubstituted cyclopentadienyl rings; RB is a structural bridge between the cyclopentadienyl rings R1 and R2 and imparts stereorigidity to the rings, and comprises at least one heteroatom bonded to M, with each of R1 and R2 bonded to the same or different heteroatom of RB which heteroatom is also bonded to M; Z is the coordination number of M and is greater than or equal to 4; m is the number of bonds between M and the heteroatom(s) of RB and to impart sterorigidity m≧2; because the number of bonds around M cannot exceed its coordination number m+2≦Z; with R1, R2 and RB selected to provide a catalyst component that is chiral with C1, C2 or CS symmetry.
- The metal M of the present invention may be any suitable metal useful as the metal component in metallocene catalysts. As a non-limiting example, M may be selected from among any metal as is known in the prior art to be useful as the metal component in metallocene catalysts.
- M will be selected to have a coordination number Z that is at least equal to the number of substituents bonded to M, that is, m number of R B heteroatom-to-metal bonds plus 2 (for both X's).
- Preferably, M is selected from among any transition metal. More preferably, M is selected from among transition metals, lanthanides and actinides. Even more preferably, M is selected from transition metals and lanthanides. Still more preferably, M is selected from among group 3d, 4d or 5d transition metals, specifically Fe, Co, Ni, Ru, Rh, Pd, Os, Ir and Pt. Yet more preferably, M is selected from among Fe, Co and Ni. Even still more preferably, M is selected from among Fe and Co.
- R 1 and R2 may be the same or each may be different and may be generally described as being substituted or unsubstituted cyclopentadienyl rings. As non-limiting examples, R1 and R2 may be selected from among any substituted or unsubstituted cyclopentadienyl ring as are known in the art to be useful in metallocene catalysts. Non-limiting examples of hydrocarbon radicals suitable for use as R1 and R2 are shown in the Examples below.
- Preferably, R 1 and R2 may be described as a cyclopentadienyl ring of the form (C5(R′)4), wherein each R′ may be the same or each may be different, and R′ is a hydrogen or a substituted or unsubstituted hydrocarbyl radical having 1-20 carbon atoms.
- Non-limiting examples of hydrocarbyl radicals suitable for use as R′ include unsubstituted and substituted alkyl, alkenyl, aryl, alkylaryl or arylalkyl radicals. More specific non-limiting examples of suitable hydrocarbyl radicals include unsubstituted and substituted methyl, ethyl, propyl, butyl, amyl, isoamyl, hexyl, isobutyl, heptyl, octyl, nonyl, decyl, cetyl, phenyl, methylene, ethylene, propylene, and other like groups.
- R B is a structural bridge between the cyclopentadienyl rings R1 and R2 and imparts stereorigidity to the rings, and comprises n heteroatoms (“HA”) bonded to M. Preferably, n≧1, more preferably n≧2, and even more preferably n≧3. An example of a suitable structural bridge RB is provided in the examples.
- Heteroatoms useful in structural bridge R B include any that can be coordinated to the metal M by a “dative” bond, that is, a bond formed by the donation of a lone pair of electrons from the heteroatom. Where RB comprises more than one heteroatom bonded to M, they may be the same heteroatom or different heteroatoms. Non-limiting examples of suitable heteroatoms include O, N, S, and P. Preferably the heteroatoms are selected from among O, N, and P, and more preferably is N.
- R 1 is bonded to a heteroatom of RB which heteroatom is also bonded to M. Likewise, R2 is also bonded to a heteroatom of RB which heteroatom is also bonded to M. R1 and R2 may be bonded to the same heteroatom that is also bonded to M, or may be bonded to different heteroatoms which different heteroatoms are also bonded to M.
- According to the present invention, R 1, R2 and RB are selected to provide a catalyst component that is chiral with C1, C2 or CS symmetry.
- Each X may be any atom or group as are known to be utilized with metallocene catalysts, and is generally covalently or ionically bonded to M. Each X may be the same or different, although commonly each X is the same. As a non-limiting example, X may be selected from among halide, sulphate, nitrate, thiolate, thiocarboxylate, BF 4 −, PF6 −, hydride, hydrocarbyloxide, carboxylate, substituted or unsubstituted hydrocarbyl, and heterohydrocarbyl. Non-limiting examples of such atoms or groups are chloride, bromide, methyl, ethyl, propyl, butyl, octyl decyl, phenyl, benzyl, methoxide, ethoxide, isopropoxide, toxylate, triflate, formate, acetate, phenoxide and benzoate. Preferably, X is a halide or a C1 to C20 hydrocarbyl. More preferably, X is chloride.
- The bridged metallocene catalyst component is generally made by contacting a bridge intermediate with a compound of the form M(X) 2. More details are provided in the Examples.
- The present invention further includes a catalyst system comprising one or more of the above described bridged metallocene catalyst components and one or more activators and/or cocatalysts (as described in greater detail below) or the reaction product of an activator and/or cocatalyst, such as for example methylaluminoxane (MAO) and optionally an alkylation/scavenging agent such as trialkylaluminum compound (TEAL). The above described metallocene catalyst components may also be supported as is known in the metallocene art. Typical supports may be any support such as talc, an inorganic oxide, clay, and clay minerals, ion-exchanged layered compounds, diatomaceous earth, silicates, zeolites or a resinous support material such as a polyolefin. Specific inorganic oxides include silica and alumina, used alone or in combination with other inorganic oxides such as magnesia, titania, zirconia and the like. Non-metallocene transition metal compounds, such as titanium tetrachloride, are also incorporated into the supported catalyst component. The inorganic oxides used as support are characterized as having an average particle size ranging from 30-600 microns, desirably from 30-100 microns, a surface area of 50-1,000 square meters per gram, desirably from 100-400 square meters per gram, a pore volume of 0.5-3.5 cc/g, desirably from about 0.5-2 cc/g.
- The bridged metallocenes of the present invention may be used in combination with some form of activator in order to create an active catalyst system. The term “activator” is defined herein to be any compound or component, or combination of compounds or components, capable of enhancing the ability of one or more metallocenes to polymerize olefins to polyolefins. Alklyalumoxanes such as methylalumoxane (MAO) are commonly used as metallocene activators. Generally alkylalumoxanes contain about 5 to 40 of the repeating units.
AIR2 for linear species and for cyclic species - where R is a C1-C8 alkyl including mixed alkyls. Particularly desirable are the compounds in which R is methyl. Alumoxane solutions, particularly methylalumoxane solutions, may be obtained from commercial vendors as solutions having various concentrations. There are a variety of methods for preparing alumoxane, non-limiting examples of which are described in U.S. Pat. Nos. 4,665,208, 4,952,540, 5,091,352, 5,206,199, 5,204,419, 4,874,734, 4,924,018, 4,908,463, 4,968,827, 5,308,815, 5,329,032, 5,248,801, 5,235,081, 5,103,031 and EP-A-0 561 476, EP 0 279 586, EP-A-0 594 218 and WO 94/10180, each fully incorporated herein by reference. (As used herein unless otherwise stated “solution” refers to any mixture including suspensions.) Ionizing activators may also be used to activate the bridged metallocenes. These activators are neutral or ionic, or are compounds such as tri(n-butyl)ammonium tetrakis(pentaflurophenyl)borate, which ionize the neutral metallocene compound. Such ionizing compounds may contain an active proton, or some other cation associated with, but not coordinated or only loosely coordinated to, the remaining ion of the ionizing compound. Combinations of activators may also be used, for example, alumoxane and ionizing activators in combinations, see for example, WO 94/07928.
- Descriptions of ionic catalysts for coordination polymerization comprised of metallocene cations activated by non-coordinating anions appear in the early work in EP-A-0 277 003, EP-A-0 277 004 and U.S. Pat. No. 5,198,401 and WO-A-92/00333 (incorporated herein by reference). These teach a desirable method of preparation wherein metallocenes (bis Cp and monoCp) are protanated by an anion precursor such that an alkyl/hydride group is abstracted from a transition metal to make it both cationic and charge-balanced by the non-coordinating anion. Suitable ionic salts include tetrakis-substituted borate or aluminum salts having fluorided aryl-constituents such as phenyl, biphenyl and napthyl.
- The term “noncoordinating anion” (“NCA”) means an anion which either does not coordinate to said cation or which is only weakly coordinated to said cation thereby remaining sufficiently labile to be displaced by a neutral Lewis base. “Compatible” noncoordinating anions are those which are not degraded to neutrality when the initially formed complex decomposes. Further, the anion will not transfer an anionic substituent or fragment to the cation so as to cause it to form a neutral four coordinate metallocene compound and a neutral by-product from the anion.
- The use of ionizing ionic compounds not containing an active proton but capable of producing both the active metallocene cation and a noncoordinating anion is also known. See, for example, EP-A-0 426 637 and EP-A-0 573 403 (incorporated herein by reference). An additional method of making the ionic catalysts uses ionizing anion precursors which are initially neutral Lewis acids but form the cation and anion upon ionizing reaction with the metallocene compounds, for example the use of tris(pentafluorophenyl) borane, see EP-A-0 520 732 (incorporated herein by reference). Ionic catalysts for addition polymerization can also be prepared by oxidation of the metal centers of transition metal compounds by anion precursors containing metallic oxidizing groups along with the anion groups, see EP-A-0 495 375 (incorporated herein by reference).
- Where the metal ligands include halogen moieties (for example, bis-cyclopentadienyl zirconium dichloride) which are not capable of ionizing abstraction under standard conditions, they can be converted via known alkylation reactions with organometallic compounds such as lithium or aluminum hydrides or alkyls, alkylalumoxanes, Grignard reagents, etc. See EP-A-O 500 944 and EP-Al-0 570 982 (incorporated herein by reference) for in situ processes describing the reaction of alkyl aluminum compounds with dihalo-substituted metallocene compounds prior to or with the addition of activating anionic compounds.
- Desirable methods for supporting ionic catalysts comprising metallocene cations and NCA are described in U.S. Pat. No. 5,643,847, U.S. patent application No. 09184358, filed Nov. 2, 1998 and U.S. patent application No. 09184389, filed Nov. 2, 1998 (all fully incorporated herein by reference). When using the support composition, these NCA support methods generally comprise using neutral anion precursors that are sufficiently strong Lewis acids to react with the hydroxyl reactive functionalities present on the silica surface such that the Lewis acid becomes covalently bound.
- Additionally, when the activator for the metallocene supported catalyst composition is a NCA, desirably the NCA is first added to the support composition followed by the addition of the bridged metallocene catalyst. When the activator is MAO, desirably the MAO and bridged metallocene catalyst are dissolved together in solution. The support is then contacted with the MAO/metallocene catalyst solution. Other methods and order of addition will be apparent to those skilled in the art.
- The catalysts of the present invention can be used for the polymerization of any type of α-olefins or the copolymerization any mixture of α-olefins. For example, the present catalyst is useful for catalyzing ethylene, propylene, butylene, pentene, hexene, 4-methylpentene and other α-alkenes having at least 2 carbon atoms, and also for mixtures thereof. Preferably, the catalysts of the present invention are utilized for the polymerization of propylene to produce polypropylene, most preferably high crystallinity polypropylene.
- The polymerization and, where applicable, pre-polymerization conditions are well known in the art and need not be described in too much detail here. In general, polymerization is accomplished by contacting together either α-olefin monomer or mixture of α-olefins in the presence of the above described catalyst system under polymerization conditions.
- The invention having been generally described, the following examples are given as particular embodiments of the invention and to demonstrate the practice and advantages thereof. It is understood that the exaples are given by way of illustration and are not intended to limit the specification or the claims to follow in any manner.
- These examples are provided merely to illustrate a few embodiments of the present invention, and are not intended to and do not limit the specification or scope of the claims.
- In these examples, all manipulations of air/moisture-sensitive materials were performed on a conventional vacuum/inert atmosphere line using standard Schlenk line techniques.
-
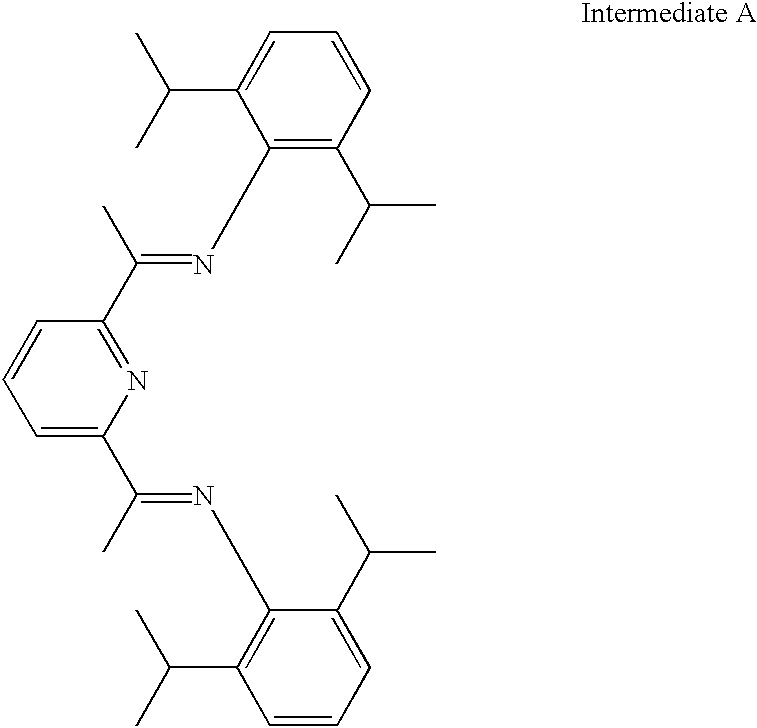
- 2,6-diisopropylaniline (3.46 ml, 18.4 mmol) was added dropwise to a solution of 2,6-diacetylpyridine (1.5 g, 9.2 mmol) in absolute ethanol (25 ml) A few drops of glacial acetic acid was added and the solution was refluxed for 48 h. Concentration of the solution to half volume and cooling to −78° C. gave intermediate A as pale yellow crystals (80%). Calcd for C33H43N3: C, 82.3; H 8.9; N 8.7; Found; C, 81.9; H 8.5; 8.7%. FABMS: M+(481). 1H NMR (CDCl3): 8.6-7.9 [m, 3H, C5H3N], 7.2-6.9 [m, 6H, C6(CHMe2)H3], 2.73[sept, 4H, CHMe2], 2.26[s, 6H, C5H3N(CMeNAr)2] and 1.16 [m, 24H, CHMe2]. FABMS is fast atom bombardment mass spectrometry.
- 250 mg, 1.09 eq. of Intermediate A, and 95 mg of FeCl2.4H2O was weighed into a 10 ml Schlenk flask containing a stirbar. The flask was placed on a Schlenk manifold, backfilled 3 times with argon, and 10 ml of THF were added while stirring. After 2 h, the THF was removed under vacuum. The resulting deep blue solid (formula below), was washed twice with ether and dried under vacuum.
- This example shows creation of a ligand having C2/Class A symmetry. The same general synthesis is followed from Example 1, with the exception that the 2,6-diisopropylaniline is replaced with indene.
- Indene (18.4 mmol) is added to a solution of 2,6-diacetylpyridine (9.2 mmol) in absolute. A few drops of glacial acetic acid is added and the solution is refluxed for 48 h. Concentration of the solution to half volume and cooling to room temperature and filtered to give the intermediate shown below.
-
- This example shows creation of a ligand having C2/Class B symmetry. The first part of the synthesis for this ligand is different from that of Example 1 above. The first part of the synthesis starts with the reduction of the diacetylpyridine to a diamine by using the Leuckart-Wallach reaction. In scheme 1 below, a general reaction is shown for the reduction of a carbonyl to an amine.
-
- This example illustrates the reduction of a carbonyl to an amine, specifically, the synthesis of 1-phenylethylamine (Vogel's Practical Organic Chemistry including qualitative organic analysis, 4 th Ed, Furniss, B. S., et al., School of Chemistry Thames Polytechnic Longman Scientific and Technical, 1978).
- 126 g (2.0 mol) of ammonium formate, 72 g (0.6 mol) of acetophenone and a few chips of porous porcelain were added to a 250 ml flask fitted with a Claisen still-head carrying a short fractionating column; a thermometer expending nearly to the bottom of the flask was inserted, and a short condenser was set for downward distillation to the side arm. The flask was heated (either with a heating mantle or in an air batch); the mixture first melted to two layers and distillation occurs. The mixture becomes homogeneous at 150-155 C. and reaction took place with slight frothing. Heating was continued, until the temperature reached 185 C. (about 2 hours); acetophenone, water and ammonium carbonate distill. The heating was stopped at 185 C., the upper layer of acetophenone was separated from the distillate and returned without drying to the flask. The mixture was heated for 3 hours at 180-185 C. and allowed to cool; the acetophone may be recovered from the distillate by extraction with 20 ml portions of toluene. The reaction mixture is transferred to a 250 ml separatory funnel and shake it with two 75 ml portions of water to remove formamide and ammonium formate. The crude (1-phenylethyl)formamide is transferred into the original reaction flask; the aqeous layer was extracted with two 20 ml portions of toluene, the toluene extracts were transferred to the flask, 75 ml of concentrated hydrochloric acid and a few chips of porous porcelain were added. The mixture was heated cautiously until about 40 ml of toluene was collected, and boiled gently under reflux for a further 40 minutes; hydrolysis proceeded rapidly to 1-phenylethylamine hydrochloride except for a small layer of unchanged acetophenone. The reaction mixture was allowed to cool, and the acetopenone was removed by extraction with four 20 ml portions of toluene. The aqueous acid solution was transferred to a 500 ml round-bottom flask equipped for stream distillation, a solution of 62.5 g of sodium hydroxide was cautiously added to 125 ml water, and steam distilled: the distillation flask was heated so that the volume remained nearly constant. Most of the amine is contained in the first 500 ml of distillate; the operation was stopped when the distillate was only faintly alkaline. The distillate was extracted with five 25, ml portions of toluene, the extract was dried with sodium hydroxide pellets and fractionally distilled. Toluene distilled over at 111 C., followed by the phylethylamine. The latter was collected as a fraction of b.p. 180-190 C. (the bulk of the product distiled at 184-186 C. (3); the yield was 43 g (59%).
- The reduction of 2,6-diacetylpiridine to the diamine is provided using a similar synthetic procedure as described in Example 6 above.
-
-
- This example provides a catalyst with symmetry C2/Class B by reacting the ligand of Example 9 with the desired metal.
- The ligand (1.05 eq.) of Example 9 and the metal salt in its hydrated or anhydrous form are added together in a Schlenk flask under inert atmosphere and then charged with THF. The mixture is stirred for several hours or until no detectable unreacted salts are observed. The mixture is filtered in air and the solids are washed with Et2O and dried under vacuum.
- To synthesize a structure with a C2/Class C symmetry, a similar procedure as the one used for the C2/Class B symmetry is used. The exception is that only one acetyl group is reduced on the 2,6-diacetylanaline. The general procedure for this synthesis is shown in the formula below where only one of the acetyls is reduced to the amine.
-
-
- A catalyst is then synthesized according to the procedure described in Example 10.
- Simply by using different R groups bonded to the nitrogen atoms with a single bond, a double bond, or with one single bond and one double bond, th esymmetries for C 1 and CS may also be obtained. Some examples for the different symmetries is summarized in Table 1 for the structure of EQN. 1.
TABLE 1 Examples of Symmetries Symmetry R1 R2 C2/Class A C2/Class B C2/Class C Cs/Class A Cs/Class B Cs/Class C Cs/Class D C1/Class A C1/Class B C1/Class C C1/Class D - Any patents, patent applications, articles, books, treatises, and any other publications cited herein, are hereby incorporated by reference for all that they teach or suggest.
- While the illustrative embodiments of the invention have been described with particularity, it will be understood that various other modifications will be apparent to and can be readily made by those skilled in the art without departing from the spirit and scope of the invention. Accordingly, it is not intended that the scope of the claims appended hereto be limited to the examples and descriptions set forth herein but rather that the claims be construed as encompassing all the features of patentable novelty which reside in the present invention, including all features which would be treated as equivalents thereof by those skilled in the art to which this invention pertains.
Claims (20)
1. A bridged metallocene compound having the formula:
wherein M is a metal; each X is an atom or group covalently or ionically bonded to M and may be the same or different; R1 and R2 may be the same or each may be different and are substituted or unsubstituted cyclopentadienyl rings; RB is a structural bridge between the cyclopentadienyl rings R1 and R2 and imparts stereorigidity to the rings, and comprises at least one heteroatom bonded to M, with each of R1 and R2 bonded to the same or different heteroatom of RB which heteroatom is also bonded to M; Z is the coordination number of M and is greater than or equal to 4; m is the number of bonds between M and heteroatoms of R1 and to impart sterorigidity m≧2; because the number of bonds around M cannot exceed its coordination number m+2≦Z; and with R1, R2 and RB selected to provide a catalyst component that is chiral with C1, C2 or CS symmetry.
2. The compound of claim 1 , wherein M is selected from the group consisting of transition metals and lanthanide metals, wherein the heteroatoms are selected from the group consisting of O, N, S, and P,
3. The compound of claim 1 , wherein RB comprises three heteroatoms bonded to M, and wherein R1 is bonded to one of the three heteroatoms, and R2 is bonded to a different one of the three heteroatoms.
4. The compound of claim 1 , wherein M is selected from among Fe, Co and Ni.
5. The compound of claim 1 , wherein M is Fe, RB comprises three heteroatoms bonded to M, and wherein R1 is bonded to one of the three heteroatoms, and R2 is bonded to a heteroatom different than the heteroatom to which R1 is bonded; M is selected from among Fe, Co and Ni.
6. The compound of claim 5 , wherein each X is independently selected from among halides and substituted or unsubstituted hydrocarbyls.
7. A method of making a bridged metallocene compound comprising contacting a metal compound of the formula M(X)2 with a bridged compound RB of the formula
wherein M is a metal; each X is an atom or group covalently or ionically bonded to M and may be the same or different; R1 and R2 may be the same or each may be different and are substituted or unsubstituted cyclopentadienyl rings; RB is a structural bridge between the cyclopentadienyl rings R1 and R2 and imparts stereorigidity to the rings, and comprises at least one heteroatom suitable for bonding to M, with each of R1 and R2 bonded to the same or different heteroatom of RB which heteroatom; Z is the coordination number of M and is greater than or equal to 4; and with R1, R2 and RB selected to provide a bridged metallocene compound that is chiral with C1, C2 or CS symmetry.
8. The method of claim 7 , wherein M is selected from the group consisting of transition metals and lanthanide metals, wherein the heteroatoms are selected from the group consisting of O, N, S, and P,
9. The method of claim 7 , wherein RB comprises three heteroatoms suitable for bonding to M, and wherein R1 is bonded to one of the three heteroatoms, and R2 is bonded to a different one of the three heteroatoms.
10. The method of claim 7 , wherein M is selected from among Fe, Co and Ni.
11. The method of claim 7 , wherein M is Fe, RB comprises three heteroatoms suitable for bonding to M, and wherein R1 is bonded to one of the three heteroatoms, and R2 is bonded to a heteroatom different than the heteroatom to which R1 is bonded; M is selected from among Fe, Co and Ni.
12. The method of claim 11 , wherein each X is independently selected from among halides and substituted or unsubstituted hydrocarbyls.
13. A catalyst system comprising an activated bridged metallocene compound having the formula:
wherein M is a metal; each X is an atom or group covalently or ionically bonded to M and may be the same or different; R1 and R2 may be the same or each may be different and are substituted or unsubstituted cyclopentadienyl rings; RB is a structural bridge between the cyclopentadienyl rings R1 and R2 and imparts stereorigidity to the rings, and comprises at least one heteroatom bonded to M, with each of R1 and R2 bonded to the same or different heteroatom of RB which heteroatom is also bonded to M; Z is the coordination number of M and is greater than or equal to 4; m is the number of bonds between M and heteroatoms of RB and to impart sterorigidity m≧2; because the number of bonds around M cannot exceed its coordination number m+2≦Z; and with R1, R2 and RB selected to provide a catalyst component that is chiral with C1, C2 or CS symmetry.
14. The system of claim 13 , wherein M is selected from the group consisting of transition metals and lanthanide metals, wherein the heteroatoms are selected from the group consisting of O, N, S and P,
15. The system of claim 13 , wherein RB comprises three heteroatoms bonded to M, and wherein R1 is bonded to one of the three heteroatoms, and R2 is bonded to a different one of the three heteroatoms.
16. The system of claim 13 , wherein M is selected from among Fe, Co and Ni.
17. The system of claim 13 , wherein M is Fe, RB comprises three heteroatoms bonded to M, and wherein R1 is bonded to one of the three heteroatoms, and R2 is bonded to a heteroatom different than the heteroatom to which R1 is bonded; M is selected from among Fe, Co and Ni.
18. The system of claim 17 , wherein each X is independently selected from among halides and substituted or unsubstituted hydrocarbyls.
19. A method of making a catalyst system comprising contacting an activator with a bridged metallocene compound having the formula:
wherein M is a metal; each X is an atom or group covalently or ionically bonded to M and may be the same or different; R1 and R2 may be the same or each may be different and are substituted or unsubstituted cyclopentadienyl rings; RB is a structural bridge between the cyclopentadienyl rings R1 and R2 and imparts stereorigidity to the rings, and comprises at least one heteroatom bonded to M, with each of R1 and R2 bonded to the same or different heteroatom of RB which heteroatom is also bonded to M; Z is the coordination number of M and is greater than or equal to 4; m is the number of bonds between M and heteroatoms of RB and to impart sterorigidity m≧2; because the number of bonds around M cannot exceed its coordination number m+2≦Z; and with R1, R2 and RB selected to provide a catalyst component that is chiral with C1, C2 or CS symmetry.
20. A method of forming polyolefins comprising contacting olefin monomer or mixture of monomers in the presence of an activated bridged metallocene compound having the formula:
wherein M is a metal; each X is an atom or group covalently or ionically bonded to M and may be the same or different; R1 and R2 may be the same or each may be different and are substituted or unsubstituted cyclopentadienyl rings; RB is a structural bridge between the cyclopentadienyl rings R1 and R2 and imparts stereorigidity to the rings, and comprises at least one heteroatom bonded to M, with each of R1 and R2 bonded to the same or different heteroatom of RB which heteroatom is also bonded to M; Z is the coordination number of M and is greater than or equal to 4; m is the number of bonds between M and heteroatoms of RB and to impart sterorigidity m≧2; because the number of bonds around M cannot exceed its coordination number m+2≦Z; and with R1, R2 and RB selected to provide a catalyst component that is chiral with C1, C2 or CS symmetry.
Priority Applications (10)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US10/301,884 US20040102311A1 (en) | 2002-11-21 | 2002-11-21 | Bridged metallocene catalyst component, method of making, polyolefin catalyst having C1, C2, or Cs symmetry, methods of making, methods of polymerizing, olefins and products made thereof |
| US10/692,068 US6894180B2 (en) | 2002-11-21 | 2003-10-23 | Catalyst structure for olefin polymerization |
| PCT/US2003/036049 WO2004047972A2 (en) | 2002-11-21 | 2003-11-12 | New catalyst structure for olefin polymerization |
| JP2004555427A JP4977322B2 (en) | 2002-11-21 | 2003-11-12 | A new catalyst structure for olefin polymerization. |
| AU2003285202A AU2003285202A1 (en) | 2002-11-21 | 2003-11-12 | New catalyst structure for olefin polymerization |
| CA002503986A CA2503986A1 (en) | 2002-11-21 | 2003-11-12 | New catalyst structure for olefin polymerization |
| KR1020057009228A KR20050085108A (en) | 2002-11-21 | 2003-11-12 | New catalyst structure for olefin polymerization |
| EP03779526A EP1562705A4 (en) | 2002-11-21 | 2003-11-12 | New catalyst structure for olefin polymerization |
| CNB2003801036547A CN100365026C (en) | 2002-11-21 | 2003-11-12 | Catalyst structure for olefin polymerization |
| TW092132162A TW200415162A (en) | 2002-11-21 | 2003-11-17 | New catalyst structure for olefin polymerization |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US10/301,884 US20040102311A1 (en) | 2002-11-21 | 2002-11-21 | Bridged metallocene catalyst component, method of making, polyolefin catalyst having C1, C2, or Cs symmetry, methods of making, methods of polymerizing, olefins and products made thereof |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10/692,068 Continuation-In-Part US6894180B2 (en) | 2002-11-21 | 2003-10-23 | Catalyst structure for olefin polymerization |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| US20040102311A1 true US20040102311A1 (en) | 2004-05-27 |
Family
ID=32324614
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10/301,884 Abandoned US20040102311A1 (en) | 2002-11-21 | 2002-11-21 | Bridged metallocene catalyst component, method of making, polyolefin catalyst having C1, C2, or Cs symmetry, methods of making, methods of polymerizing, olefins and products made thereof |
| US10/692,068 Expired - Fee Related US6894180B2 (en) | 2002-11-21 | 2003-10-23 | Catalyst structure for olefin polymerization |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10/692,068 Expired - Fee Related US6894180B2 (en) | 2002-11-21 | 2003-10-23 | Catalyst structure for olefin polymerization |
Country Status (3)
| Country | Link |
|---|---|
| US (2) | US20040102311A1 (en) |
| CN (1) | CN100365026C (en) |
| TW (1) | TW200415162A (en) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114751943B (en) * | 2022-04-21 | 2023-06-23 | 中国科学院青岛生物能源与过程研究所 | Pyridine-2-aldehyde (ketone) oxime/cobalt complex, preparation method thereof and application thereof in conjugated diene polymerization |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6313243B1 (en) * | 1998-06-16 | 2001-11-06 | Mitsui Chemicals Inc | Catalyst for olefin polymerization and method of polymerizing olefin |
| US6369177B1 (en) * | 1998-06-16 | 2002-04-09 | Mitsui Chemicals Inc | Catalyst for olefin polymerization and method of polymerizing olefin |
Family Cites Families (41)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4665208A (en) | 1985-07-11 | 1987-05-12 | Exxon Chemical Patents Inc. | Process for the preparation of alumoxanes |
| DE3752359T2 (en) | 1986-09-24 | 2003-07-31 | Mitsui Chemicals, Inc. | olefin polymerization |
| JPH0780933B2 (en) | 1986-11-20 | 1995-08-30 | 三井石油化学工業株式会社 | Olefin Polymerization Method |
| US5198401A (en) | 1987-01-30 | 1993-03-30 | Exxon Chemical Patents Inc. | Ionic metallocene catalyst compositions |
| IL85097A (en) | 1987-01-30 | 1992-02-16 | Exxon Chemical Patents Inc | Catalysts based on derivatives of a bis(cyclopentadienyl)group ivb metal compound,their preparation and their use in polymerization processes |
| PL276385A1 (en) | 1987-01-30 | 1989-07-24 | Exxon Chemical Patents Inc | Method for polymerization of olefines,diolefins and acetylene unsaturated compounds |
| JPH0742301B2 (en) | 1987-02-14 | 1995-05-10 | 三井石油化学工業株式会社 | Particulate aluminoxane, its manufacturing method and its use |
| JP2538588B2 (en) | 1987-04-03 | 1996-09-25 | 三井石油化学工業株式会社 | Method for producing solid catalyst for olefin polymerization |
| US5206199A (en) | 1987-04-20 | 1993-04-27 | Mitsui Petrochemical Industries, Ltd. | Catalyst for polymerizing an olefin and process for polymerizing an olefin |
| US5158920A (en) | 1988-07-15 | 1992-10-27 | Fina Technology, Inc. | Process for producing stereospecific polymers |
| US5091352A (en) | 1988-09-14 | 1992-02-25 | Mitsui Petrochemical Industries, Ltd. | Olefin polymerization catalyst component, olefin polymerization catalyst and process for the polymerization of olefins |
| US4908463A (en) | 1988-12-05 | 1990-03-13 | Ethyl Corporation | Aluminoxane process |
| US5103031A (en) | 1989-02-21 | 1992-04-07 | Ethyl Corporation | Falling film aluminoxane process |
| US4968827A (en) | 1989-06-06 | 1990-11-06 | Ethyl Corporation | Alkylaluminoxane process |
| US4924018A (en) | 1989-06-26 | 1990-05-08 | Ethyl Corporation | Alkylaluminoxane process |
| US5763549A (en) | 1989-10-10 | 1998-06-09 | Fina Technology, Inc. | Cationic metallocene catalysts based on organoaluminum anions |
| DK0426637T4 (en) | 1989-10-30 | 2002-01-14 | Fina Technology | Process for the preparation of metallocene catalysts for polymerization of olefins |
| EP0670334A3 (en) | 1990-06-22 | 1995-09-13 | Exxon Chemical Patents Inc. | Aluminum-free monocyclopentadienyl metallocene catalysts for olefin polymerization |
| EP0500944B1 (en) | 1990-07-24 | 1998-10-07 | Mitsui Chemicals, Inc. | Catalyst for alpha-olefin polymerization and production of poly-alpha-olefin therewith |
| ES2071888T3 (en) | 1990-11-12 | 1995-07-01 | Hoechst Ag | BISINDENILMETALOCENOS SUBSTITUTED IN POSITION 2, PROCEDURE FOR ITS PREPARATION AND USE AS CATALYSTS IN THE POLYMERIZATION OF OLEFINS. |
| US5243001A (en) | 1990-11-12 | 1993-09-07 | Hoechst Aktiengesellschaft | Process for the preparation of a high molecular weight olefin polymer |
| US5189192A (en) | 1991-01-16 | 1993-02-23 | The Dow Chemical Company | Process for preparing addition polymerization catalysts via metal center oxidation |
| US5721185A (en) | 1991-06-24 | 1998-02-24 | The Dow Chemical Company | Homogeneous olefin polymerization catalyst by abstraction with lewis acids |
| US5235081A (en) | 1992-03-18 | 1993-08-10 | Ethyl Corporation | Method of removing gel forming materials from methylaluminoxanes |
| US5308815A (en) | 1991-07-26 | 1994-05-03 | Ethyl Corporation | Heterogeneous methylaluminoxane catalyst system |
| IL101215A (en) * | 1992-03-13 | 1995-12-31 | Scitex Corp Ltd | Flexible sheet storage device |
| US5329032A (en) | 1992-03-18 | 1994-07-12 | Akzo Chemicals Inc. | Polymethylaluminoxane of enhanced solution stability |
| US5434115A (en) | 1992-05-22 | 1995-07-18 | Tosoh Corporation | Process for producing olefin polymer |
| US5248801A (en) | 1992-08-27 | 1993-09-28 | Ethyl Corporation | Preparation of methylaluminoxanes |
| ES2105331T3 (en) | 1992-10-02 | 1997-10-16 | Dow Chemical Co | SUPPORTED HOMOGENEOUS CATALYTIC COMPLEXES FOR OLEPHINE POLYMERIZATION. |
| US5939346A (en) | 1992-11-02 | 1999-08-17 | Akzo Nobel N.V. | Catalyst system comprising an aryloxyaluminoxane containing an electron withdrawing group |
| US5643847A (en) | 1994-08-03 | 1997-07-01 | Exxon Chemical Patents Inc. | Supported ionic catalyst composition |
| US6143686A (en) | 1994-08-03 | 2000-11-07 | Exxon Chemical Patents, Inc. | Supported ionic catalyst compositions |
| ES2161919T3 (en) | 1994-11-22 | 2001-12-16 | Atofina Res | METALOCENE BRIDGES FOR USE IN CATALYTIC SYSTEMS FOR POLYMERIZATION OF OLEFINS. |
| US6002033A (en) | 1995-11-22 | 1999-12-14 | Fina Research, S.A. | Bridged metallocenes for use in catalyst systems |
| JP4169791B2 (en) | 1997-03-29 | 2008-10-22 | バセル テクノロジー カンパニー ベスローテン フェンノートシャップ | Metallocenes and catalysts for olefin polymerization. |
| US6228795B1 (en) | 1997-06-05 | 2001-05-08 | Exxon Chemical Patents, Inc. | Polymeric supported catalysts |
| IT1298616B1 (en) | 1998-03-10 | 2000-01-12 | Enichem Spa | BRIDGED METALLOCENIC COMPLEX FOR THE (CO) POLYMERIZATION OF OLEFINS |
| US6194343B1 (en) | 1999-03-11 | 2001-02-27 | University Of Waterloo | Bridged “tethered” metallocenes |
| DE10017430A1 (en) * | 2000-04-07 | 2001-10-11 | Basf Ag | Polymerization catalyst |
| GB0112023D0 (en) * | 2001-05-17 | 2001-07-11 | Bp Chem Int Ltd | Polymerisation catalyst |
-
2002
- 2002-11-21 US US10/301,884 patent/US20040102311A1/en not_active Abandoned
-
2003
- 2003-10-23 US US10/692,068 patent/US6894180B2/en not_active Expired - Fee Related
- 2003-11-12 CN CNB2003801036547A patent/CN100365026C/en not_active Expired - Fee Related
- 2003-11-17 TW TW092132162A patent/TW200415162A/en unknown
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6313243B1 (en) * | 1998-06-16 | 2001-11-06 | Mitsui Chemicals Inc | Catalyst for olefin polymerization and method of polymerizing olefin |
| US6369177B1 (en) * | 1998-06-16 | 2002-04-09 | Mitsui Chemicals Inc | Catalyst for olefin polymerization and method of polymerizing olefin |
Also Published As
| Publication number | Publication date |
|---|---|
| CN100365026C (en) | 2008-01-30 |
| US20040138055A1 (en) | 2004-07-15 |
| TW200415162A (en) | 2004-08-16 |
| US6894180B2 (en) | 2005-05-17 |
| CN1713959A (en) | 2005-12-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US7973116B2 (en) | Pyridyldiamido transition metal complexes, production and use thereof | |
| US7102006B2 (en) | Preparation of metal complexes | |
| AU764658B2 (en) | Production of half-sandwich substituted catalyst precursors | |
| JP2005511867A (en) | Imino-amide catalyst compound for olefin polymerization | |
| US7164020B2 (en) | Preparation of metal amide complexes | |
| US6998363B2 (en) | Polyolefin catalyst component using non-covalent interactions | |
| JP4977322B2 (en) | A new catalyst structure for olefin polymerization. | |
| US20040102311A1 (en) | Bridged metallocene catalyst component, method of making, polyolefin catalyst having C1, C2, or Cs symmetry, methods of making, methods of polymerizing, olefins and products made thereof | |
| US7105672B2 (en) | Cyclometallated catalysts | |
| EP1997834A1 (en) | Catalysts | |
| CN111620914B (en) | Side-arm metallocene tetravalent transition metal complex containing neutral benzyl heteroatom ligand and application thereof | |
| US6683199B1 (en) | Dicationic non-metallocene group 4 metal complexes | |
| US6271322B1 (en) | Monocyclopentadienyl transition metal catalyst and olefin polymerization process | |
| US6087290A (en) | Si-N-Si-bridged metallocenes, their production and use | |
| US7001863B2 (en) | Monoamide based catalyst compositions for the polymerization of olefins | |
| US6391988B1 (en) | Tris(pyrazoyl) based anions | |
| EP1809643A1 (en) | Highly soluble ferrocenyl compounds | |
| Pyridine—Alkoxide | plexes of Tin (II), Gallium (HI), and Indium (III). Syntheses of [(ME) 2ATI] 2GaI and [(Me) 2AT1] 2InCl Using the T in (III) | |
| JP2001181336A (en) | Olefin polymerization catalyst and olefin polymerization method |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: FINA TECHNOLOGY, INC., TEXAS Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:RAZAVI, ABBAS;LOPEZ, MARGARITO;BAEKELMANS, DIDIER;AND OTHERS;REEL/FRAME:014824/0008;SIGNING DATES FROM 20030904 TO 20031107 |
|
| STCB | Information on status: application discontinuation |
Free format text: EXPRESSLY ABANDONED -- DURING EXAMINATION |