US11084974B2 - Use of multiple charged cationic compounds derived from polyamines for clay stabilization in oil and gas operations - Google Patents
Use of multiple charged cationic compounds derived from polyamines for clay stabilization in oil and gas operations Download PDFInfo
- Publication number
- US11084974B2 US11084974B2 US16/554,805 US201916554805A US11084974B2 US 11084974 B2 US11084974 B2 US 11084974B2 US 201916554805 A US201916554805 A US 201916554805A US 11084974 B2 US11084974 B2 US 11084974B2
- Authority
- US
- United States
- Prior art keywords
- treatment composition
- compound
- clay
- clay treatment
- polyamine
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 0 [2*]C(=C)C(=O)C[3*]CC Chemical compound [2*]C(=C)C(=O)C[3*]CC 0.000 description 15
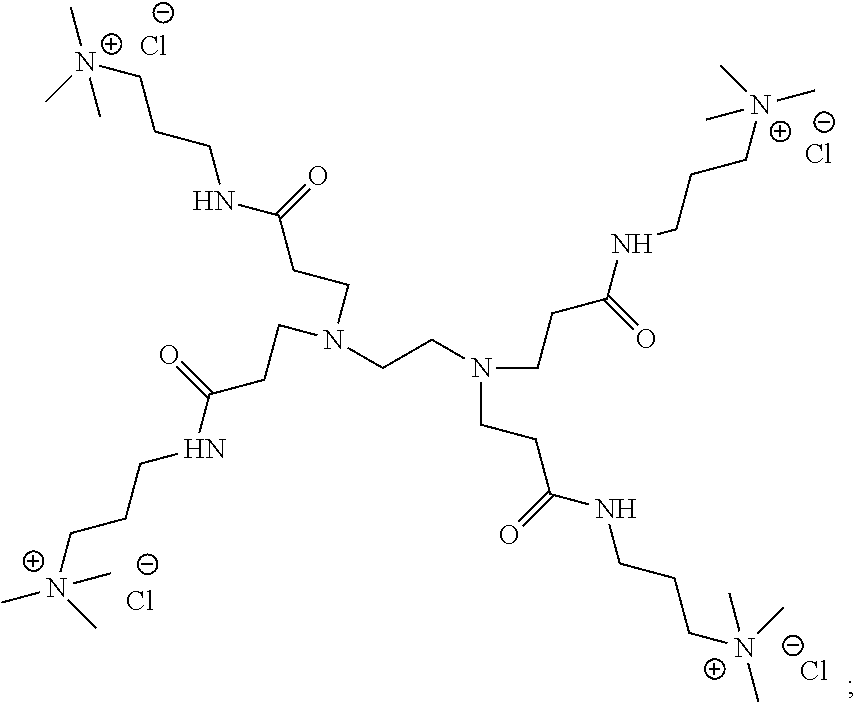
- NOVQCVLRBYHBJT-UHFFFAOYSA-R Cl.[CH2-][N+](C)(C)CCCNC(=O)CCN(CCN(CCC(=O)NCCC[N+](C)(C)C)CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCC(=O)NCCC[N+](C)(C)C.[Cl-].[Cl-].[Cl-].[Cl-] Chemical compound Cl.[CH2-][N+](C)(C)CCCNC(=O)CCN(CCN(CCC(=O)NCCC[N+](C)(C)C)CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCC(=O)NCCC[N+](C)(C)C.[Cl-].[Cl-].[Cl-].[Cl-] NOVQCVLRBYHBJT-UHFFFAOYSA-R 0.000 description 3
- RLKSWKNUKYLHIF-UHFFFAOYSA-R C[N+](C)(C)CCCNC(=O)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCC(=O)NCCC[N+](C)(C)C.[Cl-].[Cl-].[Cl-].[Cl-] Chemical compound C[N+](C)(C)CCCNC(=O)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCC(=O)NCCC[N+](C)(C)C.[Cl-].[Cl-].[Cl-].[Cl-] RLKSWKNUKYLHIF-UHFFFAOYSA-R 0.000 description 2
- KILXRPFYVSQIFC-UHFFFAOYSA-S C[N+](C)(C)CCCNC(=O)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCC(=O)NCCC[N+](C)(C)C.[Cl-].[Cl-].[Cl-].[Cl-].[Cl-] Chemical compound C[N+](C)(C)CCCNC(=O)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCC(=O)NCCC[N+](C)(C)C.[Cl-].[Cl-].[Cl-].[Cl-].[Cl-] KILXRPFYVSQIFC-UHFFFAOYSA-S 0.000 description 2
- KBCNDQHJJOUFJS-UHFFFAOYSA-A C.C.C.C.C[N+](C)(C)CCCNC(=O)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCC(=O)NCCC[N+](C)(C)C.C[N+](C)(C)CCCNC(=O)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCC(=O)NCCC[N+](C)(C)C.C[N+](C)(C)CCCNC(=O)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCC(=O)NCCC[N+](C)(C)C.[Cl-].[Cl-].[Cl-].[Cl-].[Cl-].[Cl-].[Cl-].[Cl-].[Cl-].[Cl-].[Cl-] Chemical compound C.C.C.C.C[N+](C)(C)CCCNC(=O)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCC(=O)NCCC[N+](C)(C)C.C[N+](C)(C)CCCNC(=O)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCC(=O)NCCC[N+](C)(C)C.C[N+](C)(C)CCCNC(=O)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCC(=O)NCCC[N+](C)(C)C.[Cl-].[Cl-].[Cl-].[Cl-].[Cl-].[Cl-].[Cl-].[Cl-].[Cl-].[Cl-].[Cl-] KBCNDQHJJOUFJS-UHFFFAOYSA-A 0.000 description 1
- VSWZOJHMERGUPZ-UHFFFAOYSA-N CCCNCCCCCNCCCCCNC.CCCNCCN(CCN)CCN(CCNCCN)CCN(CCNC)CCN(CCN)CCN.NCCN(CCN)CCN(CCN(CCN)CCN)CCN(CCN(CCN(CCN)CCN)CCN(CCN)CCN)CCN(CCN(CCN(CCN)CCN)CCN(CCN)CCN)CCN(CCN(CCN)CCN)CCN(CCN)CCN Chemical compound CCCNCCCCCNCCCCCNC.CCCNCCN(CCN)CCN(CCNCCN)CCN(CCNC)CCN(CCN)CCN.NCCN(CCN)CCN(CCN(CCN)CCN)CCN(CCN(CCN(CCN)CCN)CCN(CCN)CCN)CCN(CCN(CCN(CCN)CCN)CCN(CCN)CCN)CCN(CCN(CCN)CCN)CCN(CCN)CCN VSWZOJHMERGUPZ-UHFFFAOYSA-N 0.000 description 1
- VMZHPYGPZAROSM-UHFFFAOYSA-V C[N+](C)(C)CCCNC(=O)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCC(=O)NCCC[N+](C)(C)C.[Cl-].[Cl-].[Cl-].[Cl-].[Cl-] Chemical compound C[N+](C)(C)CCCNC(=O)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCC(=O)NCCC[N+](C)(C)C.[Cl-].[Cl-].[Cl-].[Cl-].[Cl-] VMZHPYGPZAROSM-UHFFFAOYSA-V 0.000 description 1
- KJOVUSSIYQSEGL-UHFFFAOYSA-T C[N+](C)(C)CCCNC(=O)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCN(CCC(=O)NCCC[N+](C)(C)C)CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCC(=O)NCCC[N+](C)(C)C.[Cl-].[Cl-].[Cl-].[Cl-].[Cl-].[Cl-] Chemical compound C[N+](C)(C)CCCNC(=O)CCN(CCC(=O)NCCC[N+](C)(C)C)CCN(CCN(CCC(=O)NCCC[N+](C)(C)C)CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCC(=O)NCCC[N+](C)(C)C.[Cl-].[Cl-].[Cl-].[Cl-].[Cl-].[Cl-] KJOVUSSIYQSEGL-UHFFFAOYSA-T 0.000 description 1
- FLKUKMUUKBBJTJ-UHFFFAOYSA-R Cl.[CH2-][N+](C)(C)CCCNC(=O)CCN(CCN(CCC(=O)NCCC[N+](C)(C)C)CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+]([CH2-])(C)C)CCN(CCC(=O)NCCC[N+]([CH2-])(C)C)CCN(CCC(=O)NCCC[N+]([CH2-])(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCC(=O)NCCC[N+](C)(C)C.[Cl-].[Cl-].[Cl-].[Cl-] Chemical compound Cl.[CH2-][N+](C)(C)CCCNC(=O)CCN(CCN(CCC(=O)NCCC[N+](C)(C)C)CCC(=O)NCCC[N+](C)(C)C)CCN(CCC(=O)NCCC[N+]([CH2-])(C)C)CCN(CCC(=O)NCCC[N+]([CH2-])(C)C)CCN(CCC(=O)NCCC[N+]([CH2-])(C)C)CCN(CCC(=O)NCCC[N+](C)(C)C)CCC(=O)NCCC[N+](C)(C)C.[Cl-].[Cl-].[Cl-].[Cl-] FLKUKMUUKBBJTJ-UHFFFAOYSA-R 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K8/00—Compositions for drilling of boreholes or wells; Compositions for treating boreholes or wells, e.g. for completion or for remedial operations
- C09K8/60—Compositions for stimulating production by acting on the underground formation
- C09K8/607—Compositions for stimulating production by acting on the underground formation specially adapted for clay formations
- C09K8/608—Polymer compositions
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B24/00—Use of organic materials as active ingredients for mortars, concrete or artificial stone, e.g. plasticisers
- C04B24/12—Nitrogen containing compounds organic derivatives of hydrazine
- C04B24/123—Amino-carboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B24/00—Use of organic materials as active ingredients for mortars, concrete or artificial stone, e.g. plasticisers
- C04B24/12—Nitrogen containing compounds organic derivatives of hydrazine
- C04B24/124—Amides
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K8/00—Compositions for drilling of boreholes or wells; Compositions for treating boreholes or wells, e.g. for completion or for remedial operations
- C09K8/02—Well-drilling compositions
- C09K8/03—Specific additives for general use in well-drilling compositions
- C09K8/035—Organic additives
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K8/00—Compositions for drilling of boreholes or wells; Compositions for treating boreholes or wells, e.g. for completion or for remedial operations
- C09K8/42—Compositions for cementing, e.g. for cementing casings into boreholes; Compositions for plugging, e.g. for killing wells
- C09K8/46—Compositions for cementing, e.g. for cementing casings into boreholes; Compositions for plugging, e.g. for killing wells containing inorganic binders, e.g. Portland cement
- C09K8/467—Compositions for cementing, e.g. for cementing casings into boreholes; Compositions for plugging, e.g. for killing wells containing inorganic binders, e.g. Portland cement containing additives for specific purposes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K8/00—Compositions for drilling of boreholes or wells; Compositions for treating boreholes or wells, e.g. for completion or for remedial operations
- C09K8/58—Compositions for enhanced recovery methods for obtaining hydrocarbons, i.e. for improving the mobility of the oil, e.g. displacing fluids
- C09K8/588—Compositions for enhanced recovery methods for obtaining hydrocarbons, i.e. for improving the mobility of the oil, e.g. displacing fluids characterised by the use of specific polymers
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K8/00—Compositions for drilling of boreholes or wells; Compositions for treating boreholes or wells, e.g. for completion or for remedial operations
- C09K8/58—Compositions for enhanced recovery methods for obtaining hydrocarbons, i.e. for improving the mobility of the oil, e.g. displacing fluids
- C09K8/594—Compositions used in combination with injected gas, e.g. CO2 orcarbonated gas
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K8/00—Compositions for drilling of boreholes or wells; Compositions for treating boreholes or wells, e.g. for completion or for remedial operations
- C09K8/60—Compositions for stimulating production by acting on the underground formation
- C09K8/62—Compositions for forming crevices or fractures
- C09K8/66—Compositions based on water or polar solvents
- C09K8/68—Compositions based on water or polar solvents containing organic compounds
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K8/00—Compositions for drilling of boreholes or wells; Compositions for treating boreholes or wells, e.g. for completion or for remedial operations
- C09K8/60—Compositions for stimulating production by acting on the underground formation
- C09K8/62—Compositions for forming crevices or fractures
- C09K8/72—Eroding chemicals, e.g. acids
- C09K8/725—Compositions containing polymers
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K8/00—Compositions for drilling of boreholes or wells; Compositions for treating boreholes or wells, e.g. for completion or for remedial operations
- C09K8/60—Compositions for stimulating production by acting on the underground formation
- C09K8/62—Compositions for forming crevices or fractures
- C09K8/72—Eroding chemicals, e.g. acids
- C09K8/74—Eroding chemicals, e.g. acids combined with additives added for specific purposes
-
- E—FIXED CONSTRUCTIONS
- E21—EARTH OR ROCK DRILLING; MINING
- E21B—EARTH OR ROCK DRILLING; OBTAINING OIL, GAS, WATER, SOLUBLE OR MELTABLE MATERIALS OR A SLURRY OF MINERALS FROM WELLS
- E21B33/00—Sealing or packing boreholes or wells
- E21B33/10—Sealing or packing boreholes or wells in the borehole
- E21B33/13—Methods or devices for cementing, for plugging holes, crevices or the like
- E21B33/138—Plastering the borehole wall; Injecting into the formation
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2208/00—Aspects relating to compositions of drilling or well treatment fluids
- C09K2208/12—Swell inhibition, i.e. using additives to drilling or well treatment fluids for inhibiting clay or shale swelling or disintegrating
Definitions
- the present disclosure relates generally to the field of using a clay treatment composition in oil and gas operations.
- the present disclosure relates to using a clay treatment composition comprising one or more multiple charged cationic compounds that are derived from polyamines for preventing clay swelling, clay migration and/or sludge formation in a subterranean formation in oil and gas operations.
- These multiple charged cationic compounds are the products of an aza-Michael Addition reaction between a polyamine and an activated olefin ( ⁇ , ⁇ -unsaturated carbonyl compound).
- the disclosed methods, clay treatment compositions, multiple charged cationic compounds herein are effective to prevent clay swelling, clay migration, and/or sludge formation than the methods, compositions, or compounds that are currently used in oil and gas operations.
- a majority of the oil and gas reservoirs are composed of water-sensitive clay minerals. These clay particles or clays tend to swell, migrate, and form sludge when they meet high volumes of aqueous fluid used in stimulation for oil and gas production. The clay swelling, clay migration, and/or sludge formation results in blockage of pores, thus reducing permeability and thus ultimately production of oil or gas flow, unless a stabilizer is used to prevent clay swelling, clay migration, and/or sludge formation.
- Clay stabilizers are typically cationic in nature. They range from simple single charged compounds like KCl in high concentrations (2% of the stimulation fluid) to high molecular weight polymeric quaternary amines, with some other types of compounds between these extremes.
- the simple single charged clay stabilizers are considered “temporary” in that they are easily washed away via ion exchange but do the job of preventing clay swelling and migration without having adverse effects to the aqueous fluid used for stimulation (stimulation fluid).
- the polymeric clay stabilizers are considered “permanent” in that once the polymeric clay stabilizers attach to clays in a polydentate fashion they become exceedingly difficult to be exchanged out. This affords a longer lasting protection after the stimulation fluid has flowed back.
- clay stabilizers While existing clay stabilizers are useful to prevent clay swelling, they are not idea and effective for every kind of subterranean formation.
- organic cationic polymers can cause formation damage due to their high molecular weights.
- the polymeric cationic materials will plate out on the formation face as they cannot leak off into the formation matrix and hence need to be used along with temporary clay control additives like potassium chloride, ammonium chloride or choline chloride.
- Smaller molecular weight materials such as choline chloride and tetramethyl ammonium chloride have also been utilized as clay stabilizers but provide only temporary clay protection and can get washed away during subsequent acid or fresh water ingression.
- existing clay stabilizers may interfere with or are not compatible with other ingredients in a stimulation fluid.
- acids or emulsifier are usually needed in a stimulation fluid
- existing clay stabilizers may not function well in the presence of these ingredients.
- new and alternative clay stabilizer is always desired for better performance, less cost, and less environmental impact.
- the exemplary multiple charged cationic compounds disclosed herein have a superior performance than some existing clay stabilizers for preventing clay swell, clay migration, and sludge formation in a subterranean formation in oil and gas operations.
- the exemplary multiple charged cationic compounds disclosed here also show an improved performance when they are used as a coagulant, water clarification, corrosion inhibition, reverse emulsion breaker (REB), antimicrobial agent in a water system or in other applications. Therefore, the disclosed clay treatment compositions or methods have an advantage of not only preventing clay swell and migration but also serving other purposes, leading to overall reduction in chemical uses, cost, and operation complexity.
- a clay treatment composition for stabilizing swellable clays and/or reducing formation of sludge in a subterranean formation
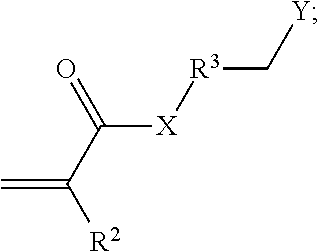
- the clay treatment composition comprises a compound or its salt derived from an aza-Michael Addition Reaction between a polyamine and an ⁇ , ⁇ -unsaturated carbonyl compound according to the following formula
- X is NH or O
- R 2 is H, CH 3 , or an unsubstituted, linear or branched C 2 -C 10 alkyl, alkenyl, or alkynyl group
- R 3 is absent or an unsubstituted, linear C 1 -C 30 alkylene group
- Y is —NR 4 R 5 R 6 (+) , or a salt thereof
- R 4 , R 5 , and R 6 are independently a C 1 -C 10 alkyl group; wherein the compound is a multiple charged cationic compound having 2, 3, or more positive charges.
- a use solution of the clay treatment composition reduces clay swelling, clay migration, and sludge formation.
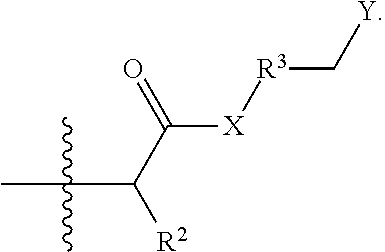
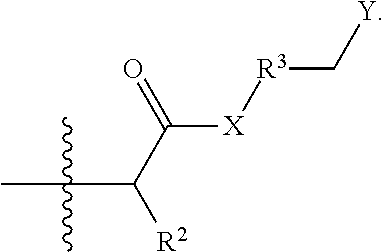
- a method of stabilizing swellable clays and/or reducing formation of sludge in a subterranean formation comprises providing a clay treatment composition into a subterranean formation, wherein the clay treatment composition comprises one or more clay treatment composition agents and a compound or its salt derived from an aza-Michael Addition Reaction between a polyamine and an ⁇ , ⁇ -unsaturated carbonyl compound according to the following formula
- X is NH or O
- R 2 is H, CH 3 , or an unsubstituted, linear or branched C 2 -C 10 alkyl, alkenyl, or alkynyl group
- R 3 is absent or an unsubstituted, linear C 1 -C 30 alkylene group
- Y is —NR 4 R 5 R 6 (+) , or a salt thereof
- R 4 , R 5 , and R 6 are independently a C 1 -C 10 alkyl group; and wherein the compound is a multiple charged cationic compound having 3 or more positive charges.
- a use solution of the clay treatment composition reduces clay swelling, clay migration, and sludge formation.
- FIG. 1 shows a generic reaction scheme to produce a multiple charged cationic compound by an aza-Michael addition reaction between a linear polyethyleneimine and an ⁇ , ⁇ -unsaturated carbonyl compound.
- FIG. 2 shows a generic reaction scheme to produce a multiple charged cationic compound by an aza-Michael addition reaction between a branch polyethyleneimine and an ⁇ , ⁇ -unsaturated carbonyl compound.
- compositions for stabilizing swellable clays and/or reducing formation of sludge in a subterranean formation for oil and gas operations More particularly, one or more multiple charged cationic compounds are used in clay treatment compositions for stabilizing swellable clays and/or reducing formation of sludge during oil and gas operations. These multiple charged cationic compounds are derived from polyamines through an aza-Michael Addition reaction a polyamine and an ⁇ , ⁇ -unsaturated carbonyl compound.
- the term “about,” as used herein, refers to variation in the numerical quantity that can occur, for example, through typical measuring and liquid handling procedures used for making concentrates or use solutions in the real world; through error in these procedures; through differences in the manufacture, source, or purity of the ingredients used to make the compositions or carry out the methods; and the like.
- the term “about” also encompasses amounts that differ due to novel equilibrium conditions for a composition resulting from a particular initial mixture. Whether or not modified by the term “about”, the claims include equivalents to the quantities.
- substituted refers to an organic group as defined below (e.g., an alkyl group) in which one or more bonds to a hydrogen atom contained therein are replaced by a bond to non-hydrogen or non-carbon atoms.
- Substituted groups also include groups in which one or more bonds to carbon(s) or hydrogen(s) atom replaced by one or more bonds, including double or triple bonds, to a heteroatom.
- a substituted group is substituted with one or more substituents, unless otherwise specified.
- a substituted group can be substituted with 1, 2, 3, 4, 5, or 6 substituents.
- Substituted ring groups include rings and ring systems in which a bond to a hydrogen atom is replaced with a bond to a carbon atom. Therefore, substituted cycloalkyl, aryl, heterocyclyl, and heteroaryl groups may also be substituted with substituted or unsubstituted alkyl, alkenyl, and alkynyl groups are defined herein.
- alkyl refers to saturated hydrocarbons having one or more carbon atoms, including straight-chain alkyl groups (e.g., methyl, ethyl, propyl, butyl, pentyl, hexyl, heptyl, octyl, nonyl, decyl, etc.), cyclic alkyl groups (or “cycloalkyl” or “alicyclic” or “carbocyclic” groups) (e.g., cyclopropyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, etc.), branched-chain alkyl groups (e.g., isopropyl, tert-butyl, sec-butyl, isobutyl, etc.), and alkyl-substituted alkyl groups (e.g., alkyl-substituted
- alkyl includes both “unsubstituted alkyls” and “substituted alkyls.”
- substituted alkyls refers to alkyl groups having substituents replacing one or more hydrogens on one or more carbons of the hydrocarbon backbone.
- substituents may include, for example, alkenyl, alkynyl, halogeno, hydroxyl, alkylcarbonyloxy, arylcarbonyloxy, alkoxycarbonyloxy, aryloxy, aryloxycarbonyloxy, carboxylate, alkylcarbonyl, arylcarbonyl, alkoxycarbonyl, aminocarbonyl, alkylaminocarbonyl, dialkylaminocarbonyl, alkylthiocarbonyl, alkoxyl, phosphate, phosphonato, phosphinato, cyano, amino (including alkyl amino, dialkylamino, arylamino, diarylamino, and alkylarylamino), acylamino (including alkylcarbonylamino, arylcarbonylamino, carbamoyl and ureido), imino, sulfhydryl, alkylthio, arylthio, thiocarboxylate,
- substituted alkyls can include a heterocyclic group.
- heterocyclic group includes closed ring structures analogous to carbocyclic groups in which one or more of the carbon atoms in the ring is an element other than carbon, for example, nitrogen, sulfur or oxygen. Heterocyclic groups may be saturated or unsaturated.
- heterocyclic groups include, but are not limited to, aziridine, ethylene oxide (epoxides, oxiranes), thiirane (episulfides), dioxirane, azetidine, oxetane, thietane, dioxetane, dithietane, dithiete, azolidine, pyrrolidine, pyrroline, oxolane, dihydrofuran, and furan.
- aziridine ethylene oxide (epoxides, oxiranes), thiirane (episulfides), dioxirane, azetidine, oxetane, thietane, dioxetane, dithietane, dithiete, azolidine, pyrrolidine, pyrroline, oxolane, dihydrofuran, and furan.
- Alkenyl groups or alkenes are straight chain, branched, or cyclic alkyl groups having two to about 30 carbon atoms, and further including at least one double bond.
- an alkenyl group has from 2 to about 30 carbon atoms, or typically, from 2 to 10 carbon atoms.
- Alkenyl groups may be substituted or unsubstituted.
- the configuration for the double bond can be a trans or cis configuration.
- Alkenyl groups may be substituted similarly to alkyl groups.
- Alkynyl groups are straight chain, branched, or cyclic alkyl groups having two to about 30 carbon atoms, and further including at least one triple bond. In some embodiments, an alkynyl group has from 2 to about 30 carbon atoms, or typically, from 2 to 10 carbon atoms. Alkynyl groups may be substituted or unsubstituted. Alkynyl groups may be substituted similarly to alkyl or alkenyl groups.
- alkylene As used herein, the terms “alkylene”, “cycloalkylene”, “alkynylides”, and “alkenylene”, alone or as part of another substituent, refer to a divalent radical derived from an alkyl, cycloalkyl, or alkenyl group, respectively, as exemplified by —CH 2 CH 2 CH 2 —.
- alkylene, cycloalkylene, alkynylene, and alkenylene groups no orientation of the linking group is implied.
- esters refers to —R 30 COOR 31 group.
- R 30 is absent, a substituted or unsubstituted alkylene, cycloalkylene, alkenylene, alkynylene, arylene, aralkylene, heterocyclylalkylene, or heterocyclylene group as defined herein.
- R 31 is a substituted or unsubstituted alkyl, cycloalkyl, alkenyl, alkynyl, aryl, aralkyl, heterocyclylalkyl, or heterocyclyl group as defined herein.
- amine refers to —R 32 NR 33 R 34 groups.
- R 32 is absent, a substituted or unsubstituted alkylene, cycloalkylene, alkenylene, alkynylene, arylene, aralkylene, heterocyclylalkylene, or heterocyclylene group as defined herein.
- R 33 and R 34 are independently hydrogen, or a substituted or unsubstituted alkyl, cycloalkyl, alkenyl, alkynyl, aryl, aralkyl, heterocyclylalkyl, or heterocyclyl group as defined herein.
- amine as used herein also refers to an independent compound.
- an amine when an amine is a compound, it can be represented by a formula of R 32′ NR 33′ R 34′ groups, wherein R 32′ , R 33′ , and R 34 are independently hydrogen, or a substituted or unsubstituted alkyl, cycloalkyl, alkenyl, alkynyl, aryl, aralkyl, heterocyclylalkyl, or heterocyclyl group as defined herein.
- alcohol refers to —R 35 OH groups.
- R 35 is absent, a substituted or unsubstituted alkylene, cycloalkylene, alkenylene, alkynylene, arylene, aralkylene, heterocyclylalkylene, or heterocyclylene group as defined herein.
- R 36 is absent, a substituted or unsubstituted alkylene, cycloalkylene, alkenylene, alkynylene, arylene, aralkylene, heterocyclylalkylene, or heterocyclylene group as defined herein.
- ether refers to —R 37 OR 38 groups.
- R 37 is absent, a substituted or unsubstituted alkylene, cycloalkylene, alkenylene, alkynylene, arylene, aralkylene, heterocyclylalkylene, or heterocyclylene group as defined herein.
- R 38 is a substituted or unsubstituted alkyl, cycloalkyl, alkenyl, alkynyl, aryl, aralkyl, heterocyclylalkyl, or heterocyclyl group as defined herein.
- solvent refers to any inorganic or organic solvent. Solvents are useful in the disclosed method or composition as reaction solvents or carrier solvents. Suitable solvents include, but are not limited to, oxygenated solvents such as lower alkanols, lower alkyl ethers, glycols, aryl glycol ethers and lower alkyl glycol ethers.
- solvents examples include, but are not limited to, methanol, ethanol, propanol, isopropanol and butanol, isobutanol, ethylene glycol, diethylene glycol, triethylene glycol, propylene glycol, dipropylene glycol, glycol ethers, mixed ethylene-propylene glycol ethers, ethylene glycol phenyl ether, and propylene glycol phenyl ether.
- Water is a solvent too.
- the solvent used herein can be of a single solvent or a mixture of many different solvents.
- Glycol ethers include, but are not limited to, diethylene glycol n-butyl ether, diethylene glycol n-propyl ether, diethylene glycol ethyl ether, diethylene glycol methyl ether, diethylene glycol t-butyl ether, dipropylene glycol n-butyl ether, dipropylene glycol methyl ether, dipropylene glycol ethyl ether, dipropylene glycol propyl ether, dipropylene glycol tert-butyl ether, ethylene glycol butyl ether, ethylene glycol propyl ether, ethylene glycol ethyl ether, ethylene glycol methyl ether, ethylene glycol methyl ether acetate, propylene glycol n-butyl ether, propylene glycol ethyl ether, propylene glycol methyl ether, propylene glycol n-propyl ether, tripropylene glycol methyl
- the term “substantially free”, “free” or “free of” refers to compositions completely lacking the component or having such a small amount of the component that the component does not affect the performance of the composition.
- the component may be present as an impurity or as a contaminant and shall be less than about 0.5 wt-%. In another embodiment, the amount of the component is less than about 0.1 wt-% and in yet another embodiment, the amount of component is less than about 0.01 wt-%.
- weight percent refers to the concentration of a substance as the weight of that substance divided by the total weight of the composition and multiplied by 100. It is understood that, as used here, “percent,” “%,” and the like are intended to be synonymous with “weight percent,” “wt-%,” etc.
- compositions of the present disclosure may comprise, consist essentially of, or consist of the components and ingredients of the disclosed compositions or methods as well as other ingredients described herein.
- “consisting essentially of” means that the methods and compositions may include additional steps, components or ingredients, but only if the additional steps, components or ingredients do not materially alter the basic and novel characteristics of the claimed methods and compositions.
- compositions disclosed herein may include an acid. However, in some embodiments, the compositions disclosed herein are free of an acid.
- acids include both organic and inorganic acids.
- Organic acids include, but not limited to, hydroxyacetic (glycolic) acid, formic acid, acetic acid, propionic acid, butyric acid, valeric acid, caproic acid, gluconic acid, itaconic acid, trichloroacetic acid, urea hydrochloride, and benzoic acid.
- Organic acids also include dicarboxylic acids such as oxalic acid, malonic acid, succinic acid, glutaric acid, maleic acid, fumaric acid, adipic acid, and terephthalic acid. Combinations of these organic acids can also be used.
- Inorganic acids include, but are not limited to, mineral acids, such as phosphoric acid, sulfuric acid, sulfamic acid, methylsulfamic acid, hydrochloric acid, hydrobromic acid, hydrofluoric acid, and nitric acid. Inorganic acids can be used alone, in combination with other inorganic acid(s), or in combination with one or more organic acid. Acid generators can be used to form a suitable acid, including for example generators such as potassium fluoride, sodium fluoride, lithium fluoride, ammonium fluoride, ammonium bifluoride, sodium silicofluoride, etc.
- Examples of particularly suitable acids in this the methods or compositions disclosed herein include inorganic and organic acids.
- Exemplary inorganic acids include phosphoric, phosphonic, sulfuric, sulfamic, methylsulfamic, hydrochloric, hydrobromic, hydrofluoric, and nitric.
- Exemplary organic acids include hydroxyacetic (glycolic), citric, lactic, formic, acetic, propionic, butyric, valeric, caproic, gluconic, itaconic, trichloroacetic, urea hydrochloride, and benzoic.
- Organic dicarboxylic acids can also be used such as oxalic, maleic, fumaric, adipic, and terephthalic acid.
- a peroxycarboxylic acid (i.e. peracid) or peroxycarboxylic acid composition can be included in the articles, products, or compositions disclosed herein.
- the term “peracid” may also be referred to as a “percarboxylic acid,” “peroxycarboxylic acid” or “peroxyacid.”
- Sulfoperoxycarboxylic acids, sulfonated peracids and sulfonated peroxycarboxylic acids are also included within the terms “peroxycarboxylic acid” and “peracid” as used herein.
- a peracid refers to an acid having the hydrogen of the hydroxyl group in carboxylic acid replaced by a hydroxy group. Oxidizing peracids may also be referred to herein as peroxycarboxylic acids.
- a peracid includes any compound of the formula R—(COOOH) n in which R can be hydrogen, alkyl, alkenyl, alkyne, acylic, alicyclic group, aryl, heteroaryl, or heterocyclic group, and n is 1, 2, or 3, and named by prefixing the parent acid with peroxy.
- R includes hydrogen, alkyl, or alkenyl.
- a peroxycarboxylic acid composition refers to any composition that comprises one or more peracids, their corresponding acids, and hydrogen peroxide or or other oxidizing agents.
- a peroxycarboxylic acid composition can also include a stabilizer, fluorescent active tracer or compound, or other ingredients, as one skilled in the other would know.
- the terms “mixed” or “mixture” when used relating to “percarboxylic acid composition,” “percarboxylic acids,” “peroxycarboxylic acid composition” or “peroxycarboxylic acids” refer to a composition or mixture including more than one percarboxylic acid or peroxycarboxylic acid. Peracids such as peroxyacetic acid and peroxyoctanoic acid may also be used. Any combination of these acids may also be used.
- the articles, products, or compositions disclosed herein are free of a peroxycarboxylic acid or peroxycarboxylic acid composition.
- a polyamine can have, but is limited to, a generic formula of NH 2 —[R 10′ ] n —NH 2 , (RNH) n —RNH 2 , H 2 N—(RNH) n —RNH 2 , or H 2 N—(RN(R′)) n —RNH 2 , wherein R 10′ is a linear or branched, unsubstituted or substituted C 2 -C 10 alkylene group, or combination thereof; R is —CH 2 —, —CH 2 CH 2 —, —CH 2 CH 2 CH 2 —, —CH(CH 3 )CH 2 —, a linear or branched, unsubstituted or substituted C 4 -C 10 alkylene group, or combination thereof; R′ is —CH 2 —, —CH 2 CH 2 —, —CH 2 CH 2 CH 2 —, —CH(CH 3 )CH 2 —, a linear or branched, unsubstitute
- the monomer in a polyamine can be the same or different.
- a polyamine refers to both small molecule polyamine when n is from 1 to 9 and polymeric polyamine when n is from 10 to 1,000,000.
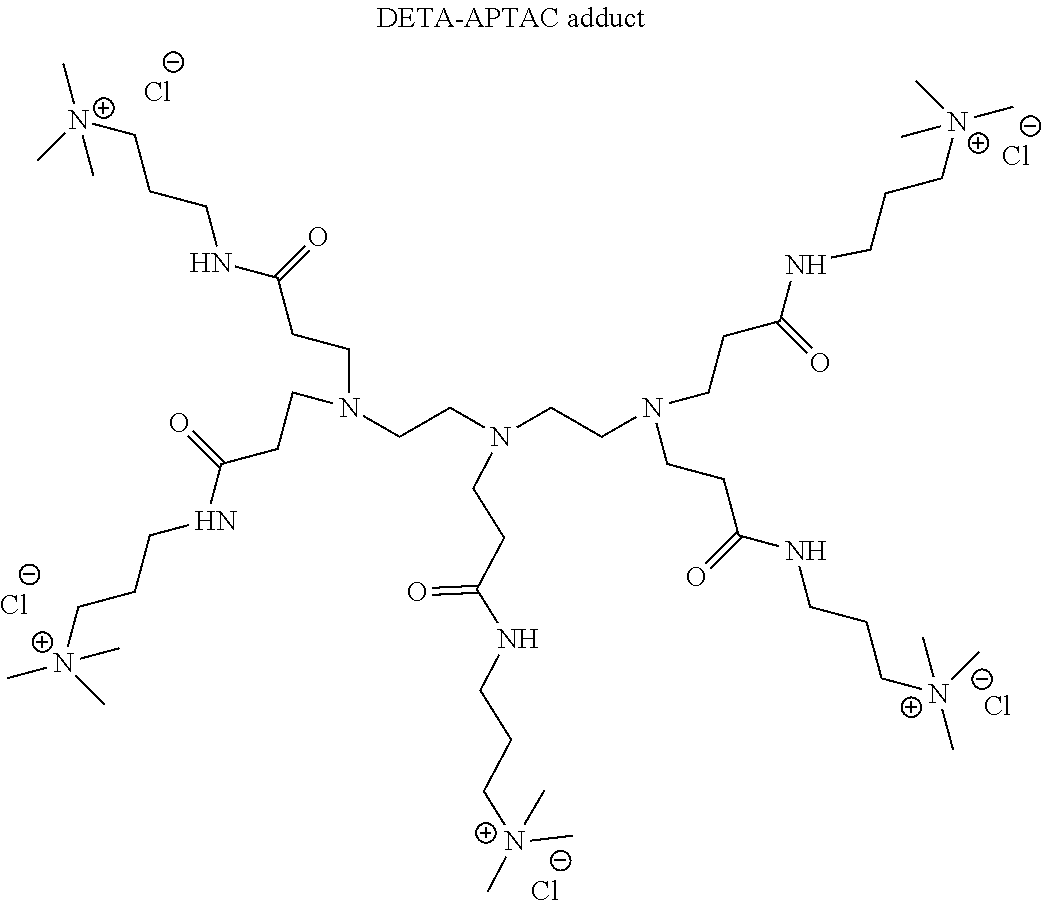
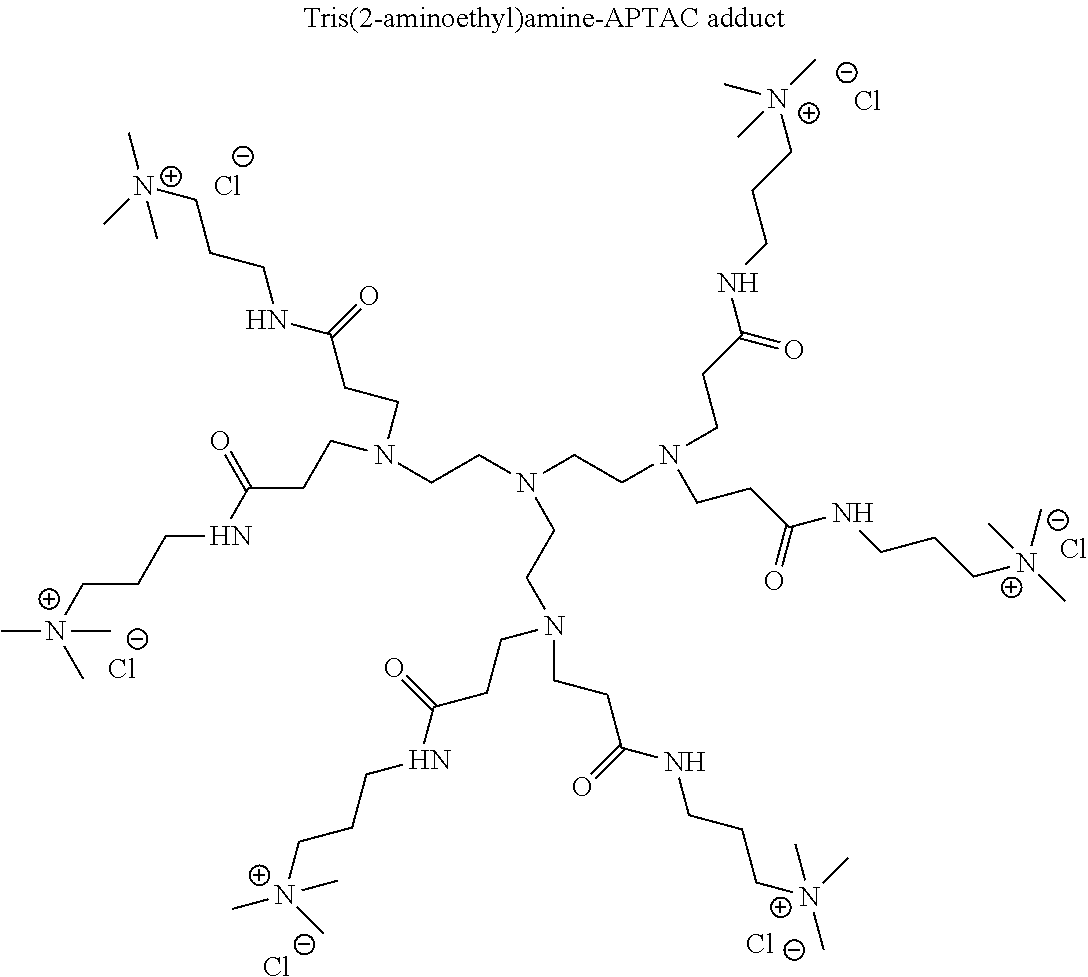
- Small molecule polyamines include, but are not limited to ethylenediamine, diethylenetriamine, triethylenetetramine, tetraethylenepentamine, pentaethylenehexamine, hexaethyleneheptamine, and tris(2-aminoethyl)amine.
- JEFFAMINE® diamines and triamines by Huntsman. These highly versatile products contain primary amino groups attached to the end of a polyether backbone normally based on propylene oxide (PO), ethylene oxide (EO), or a mixture of both oxides.
- JEFFAMINE® amines include a polyetheramine family consisting of monoamines, diamines and triamines based on the core polyether backbone structure.
- JEFFAMINE® amines also include high-conversion, and polytetramethylene glycol (PTMEG) based polyetheramines. These JEFFAMINE® amines have an average molecular weight (M w ) of from about 130 to about 4,000.
- a polyamine used in this disclosure can a polyamine derivative, in which one or more of the NH protons, but not all, in the polyamine is substituted by an unsubstituted or substituted group.
- an alkyl polyamine that contains one or more alkyl group connected to the nitrogen atom can be used to produce the multiple charged cationic polyamine disclosed herein.
- these PEI derivatives only some of primary NH 2 or secondary NH protons are replaced by other non-proton groups and the remaining NH 2 or NH protons can still react with a Michael acceptor, such as an activated olefin containing a hydrophilic (ionic) group, by an aza-Michael Addition reaction.
- polyethyleneimine (PEI) and its derivatives.
- PEI polyethyleneimine
- polyaziridine is a polymer with a repeating unit of CH 2 CH 2 NH and has a general formulation of NH 2 (CH 2 CH 2 NH) n —CH 2 CH 2 NH 2 , wherein n can be from 2 to 10 5 .
- the repeating monomer in PEI has a molecular weight (M w ) of 43.07 and a nitrogen to carbon ratio of 1:2.
- PEI derivatives include ethoxylated/propylated PEIs, polyquats PEI, polyglycerol quats PEI, and other PEI derivatives, salts, or mixtures thereof.
- the molar mass of the polyethyleneimines, including modified polyethyleneimines can vary from about 800 g/mol to about 2,000,000 g/mol.
- SOKALAN® HP20 is an alkoxylated PEI product.
- PEIs and their derivatives can linear, branched, or dendric.
- Linear polyethyleneimines contain all secondary amines, in contrast to branched PEIs which contain primary, secondary and tertiary amino groups. Totally branched, dendrimeric forms also exist and contain primary and tertiary amino groups. Drawings for unmodified linear, branched, and dendrimeric PEI are shown below.
- PEI derivatives are usually obtained by substituting proton(s) on the nitrogen atoms with different group.
- One such PEI derivative is ethoxylated and propoxylated PEI, wherein the polyethyleneimines are derivatized with ethylene oxide (EO) and/or propylene oxide (PO) side chains. Ethoxylation of PEIs can increase the solubility of PEIs.
- PEI derivative usually, not every NH proton in a PEI derivative is substituted.
- a partially derivatized PEI can be used to produce the multiple charged cationic compounds disclosed here.
- PEI is produced on industrial scale.
- Various commercial polyethyleneimines are available, including for example those sold under the tradename Lupasol® (BASF), including for example Lupasol® FG, Lupasol® G, Lupasol® PR 8515, Lupasol® WF, Lupasol® G 20/35/100, Lupasol® HF, Lupasol® P, Lupasol® PS, Lupasol® PO 100, Lupasol® PN 50/60, and Lupasol® SK.
- M w average molecular weights
- Mn number average molecular weight
- M w weight average molecular weight
- D The polydispersity index
- Mn ( ⁇ n i M i )/ ⁇ n i
- M w ( ⁇ n i Mi 2 )/ ⁇ n i M i
- D M w /M n
- the index number, i represents the number of different molecular weights present in the sample and this the total number of moles with the molar mass of M i .
- Mn and M w are usually different.
- a PEI compound can have a M n of about 10,000 by GPC and M w of about 25,000 by LS.
- LS Light Scattering
- GPC gel permeation chromatography
- SEC size exclusion chromatography
- polyethyleneimines are soluble in water and available as anhydrous polyethyleneimines and/or modified polyethyleneimines provided in aqueous solutions or methoxypropanol (as for Lupasol® PO 100).
- PEI and its derivatives find many applications usually derived from its polycationic character. Because of the presence of amine groups, PEI can be protonated with acids to form a PEI salt from the surrounding medium resulting in a product that is partially or fully ionized depending on pH. For example, about 73% of PEI is protonated at pH 2, about 50% of PEI is protonated at pH 4, about 33% of PEI is protonated at pH 5, about 25% of PEI is protonated at pH 8 and about 4% of PEI is protonated at pH 10. In general, PEIs can be purchased as their protonated or unprotonated form with and without water. The commercial PEIs at pH 13 have a charge (cationic) density of about 16-17 meq/g (milliequivalents per gram).
- each protonated nitrogen center is balanced with an anion of an acid obtained during neutralization.
- protonated PEI salts include, but are not limited to, PEI-hydrochloride salt, PEI-sulfuric acid salt, PEI-nitric acid salt, PEI-acetic acid salt PEI fatty acid salt and the like. In fact, any acid can be used to protonate PEIs resulting in the formation of the corresponding PEI salt compound.
- Suitable polyethyleneimine useful in the present disclosure may contain a mixture of primary, secondary, and tertiary amine substituents or mixture of different average molecular weights.
- the mixture of primary, secondary, and tertiary amine substituents may be in any ratio, including for example in the ratio of about 1:1:1 to about 1:2:1 with branching every 3 to 3.5 nitrogen atoms along a chain segment.
- suitable polyethyleneimine compounds may be primarily one of primary, secondary or tertiary amine substituents.
- the polyamine that can be used to make the multiple charged cationic compounds disclosed herein can have a wide range of its average molecular weight. Different multiple charged cationic compounds with their characteristic average molecular weights can be produced by selecting different starting small molecule polyamines, polymeric PEIs, or mixture thereof. Controlling the size of polyamines or PEI and extent of modification by the ⁇ , ⁇ -unsaturated carbonyl compound, one can produce the multiple charged cationic compounds with a similar average molecular weight and multiple cationic charges at any pH. Because of this character, one can produce and use different modified polyamines for a wider range of application that using unmodified polyamine or PEIs.
- the polyamines that can be used to make the multiple charged cationic or anionic compounds disclosed here have an average molecular weight (M w ) of about 60-200, about 100-400, about 100-600, about 600-5,000, about 600-800, about 800-2,000, about 800-5,000, about 100-2,000,000, about 100-25,000, about 600-25,000, about 800-25,000, about 600-750,000, about 800-750,000, about 25,000-750,000, about 750,000-2,000,000, about 100, about 200, about 300, about 400, about 500, about 600, about 700, about 800, about 1,000, about 1,500, about 2,000, about 3,000, about 5,000, about 8,000, about 10,000, about 15,000, about 20,000, about 50,000, about 100,000, about 250,000, about 500,000, about 1,000,000, 2,000,000, or any value there between.
- M w average molecular weight
- the multiple charged cationic compounds used in the clay treatment compositions disclosed herein are derived from an aza-Michael Addition Reaction between a polyamine and an activated olefin, more specifically an ⁇ , ⁇ -unsaturated carbonyl compound containing a hydrophilic ionic group.
- An aliphatic amine group may undergo an aza-Michael Addition reaction when in contact with an unsaturated hydrocarbon moiety (e.g., carbon-carbon double bond) that is in proximity of an electron withdrawing group such as carbonyl, cyano, or nitro group.
- an unsaturated hydrocarbon moiety e.g., carbon-carbon double bond
- an electron withdrawing group such as carbonyl, cyano, or nitro group.
- the Michael addition is a reaction between nucleophiles and activated olefin and alkyne functionalities, wherein the nucleophile adds across a carbon-carbon multiple bond that is adjacent to an electron withdrawing and resonance stabilizing activating group, such as a carbonyl group.
- the Michael addition nucleophile is known as the “Michael donor”
- the activated electrophilic olefin is known as the “Michael acceptor”
- reaction product of the two components is known as the “Michael adduct.”
- Michael donors include, but are not restricted to, amines, thiols, phosphines, carbanions, and alkoxides.
- Michael acceptors include, but are not restricted to, acrylate esters, alkyl methacrylates, acrylonitrile, acrylamides, maleimides, cyanoacrylates and vinyl sulfones, vinyl ketones, nitro ethylenes, ⁇ , ⁇ -unsaturated aldehydes, vinyl phosphonates, acrylonitrile, vinyl pyridines, azo compounds, beta-keto acetylenes and acetylene esters.
- an “activated olefin” refers to a substituted alkene in which at least one of the double-bond carbon has a conjugated electron withdrawing group.
- Aza-Michael addition reaction can be catalyzed by a strong acid or base. In some cases, some ionic liquids can function both as reaction media and catalyst.
- the preferred catalyst for the Aza-Michael addition reaction to synthesize the disclosed compounds is a base. Exemplary base catalyst can be hydroxide and amines. Because the reaction to synthesize the disclosed compounds uses a primary amine, the primary amine itself can function as a catalyst for the reaction. In such embodiments, no additional catalyst is necessary, or an additional catalyst is optional. Other preferred catalysts include amidine and guanidine bases.
- solvent and/or diluent for the reaction is optional.
- a wide range of non-acidic solvents are suitable, such as, for example, water, ethers (e.g., tetrahydrofuran (THF)), aromatic hydrocarbons (e.g., toluene and xylene), alcohols (e.g., n-butanol), esters (e.g., ethyl 3-ethoxypropionate), and the like.
- a wide range of solvents can be used for the reaction because the synthesis process is relatively insensitive to solvent.
- loading levels can range from as low as about 10 wt-% up to about 80 wt-% and higher.
- the solvent loading level can be about 0 wt-%, from about 1 wt-% to about 10 wt-%, from about 10 wt-% to about 20 wt-%, from about 20 wt-% to about 30 wt-%, from about 30 wt-% to about 40 wt-%, from about 40 wt-% to about 50 wt-%, from about 50 wt-% to about 60 wt-%, from about 60 wt-% to about 70 wt-%, from about 70 wt-% to about 80 wt-%, from about 1 wt-% to about 20 wt-%, from about 20 wt-% to about 40 wt-%, from about 40 wt-% to about 60 wt-%, from about 60 wt-% to about 80 wt-%, from about 40 wt-% to about 70 wt-%, about 5 wt-%, about 15 wt-%, about 25
- the reaction can be carried out at a temperature over a wide range of temperatures.
- the reaction temperature can range from about 0° C. to about 150° C., more preferably from about 50° C. to about 80° C.
- the contacting temperature can be from about 10° C. to about 140° C., about 20° C. to about 130° C., about 30° C. to about 120° C., about 40° C. to about 110° C., about 50° C. to about 100° C., about 60° C. to about 90° C., about 70° C. to about 80° C., about 0° C. to about 20° C., about 20° C. to about 40° C., about 40° C. to about 60° C., about 60° C.
- reaction temperature can be about the same from starting of the reaction to end of the reaction and can be changed from one temperature to another while the reaction is going on.
- the reaction time for the synthesis of the compounds disclosed herein can vary widely, depending on such factors as the reaction temperature, the efficacy and amount of the catalyst, the presence or absence of diluent (solvent), and the like.
- the preferred reaction time can be from about 0.5 hours to about 48 hours, from about 1 hour to 40 hours, from about 2 hours to 38 hours, from about 4 hours to about 36 hours, from 6 hours to about 34 hours, from about 8 hours to about 32 hours, from about 10 hours to about 30 hours, from about 12 hours to about 28 hours, from about 14 hours to 26 hours, from about 16 hours to 24 hours, from about 18 hours to 20 hours, from about 1 hour to 8 hours, from 8 hours to 16 hours, from 8 hours to about 24 hours, about 2 hours, about 4 hours, about 6 hours, about 8 hours, about 10 hours, about 14 hours, about 16 hours, about 18 hours, about 24 hours, about 30 hours, about 36 hours, or any values there between.
- reaction for the synthesis of the compounds disclosed herein can go to completion when one mole of the polyamine in mole and two or more moles of the ⁇ , ⁇ -unsaturated carbonyl compound, are mixed together for a sufficient of time at a temperature described above.
- the progression of the reaction can be typically monitored by ESI-MS and/or NMR spectroscopy for consumption of the monomer.
- the reaction products can be purified or separated by HPLC or other methods known by one skilled in the art.
- the formed product was separated by removal of solvent or by precipitation in a non-polar solvent that was the opposite of the reaction media.
- the formed product was precipitated from the aqueous reaction mixture. Higher pressure can speed-up the reaction.
- the reaction can have a product yield of more than 98% in 16 hours.
- a clay treatment composition in the present disclosure includes one or more additional clay treatment composition agents.
- the additional clay treatment composition agent in the disclosed clay treatment compositions can include, but is not limited to, an acid, carrier, dispersant, biocide, inorganic salt, organic salt, emulsifier, additional clay stabilizer, corrosion inhibitor, antioxidant, polymer degradation prevention agent, permeability modifier, foaming agent, antifoaming agent, fracturing proppant, glass particulate, sand, fracture proppant/sand control agent, scavenger for H 2 S, CO 2 , and/or O 2 , gelling agent, lubricant, and friction reducing agent, salt, or mixture thereof.
- the additional clay treatment composition agent in the disclosed clay treatment compositions can also include, but not be limited to, an organic sulfur compound, de-emulsifier, asphaltene inhibitor, paraffin inhibitor, scale inhibitor, water clarifier, emulsion breaker, reverse emulsion breaker, gas hydrate inhibitor, a pH modifier, a surfactant, or a combination thereof.
- the reverse emulsion breakers are organic polymers such as acrylic acid-based polymers, acrylamide-based polymers, polymerized amines, alkanolamines, thiocarbamates, and cationic polymers such as acrylamide diallyldimethylammonium chloride (DADMAC) polymer and/or methylacrylamide [3-(Methacryloylamino)propyl]trimethylammonium chloride (MAPTAC) polymers, copolymers of epichlorohydrin and dimethylamine or trimethylamine, copolymers of acrylamide and dimethylaminoethyl acrylate-methyl chloride quaternized, aluminium chlorohydrate (ACH) and polyaluminium chlorides (PAC), acrylamides—DMAEA.MCQ copolymers.
- DADMAC acrylamide diallyldimethylammonium chloride
- MTYAC methylacrylamide [3-(Methacryloylamino)propyl]trimethylammonium chlor
- the additional clay treatment composition agent can be a sequestrant, solubilizer, lubricant, buffer, cleaning agent, rinse aid, preservative, binder, thickener or other viscosity modifier, processing aid, carrier, water-conditioning agent, foam inhibitor or foam generator, threshold agent or system, aesthetic enhancing agent (e.g., dye, odorant, perfume), other additive suitable for formulation with a clay stabilizer, or mixtures thereof.
- solubilizer e.g., solubilizer, lubricant, buffer, cleaning agent, rinse aid, preservative, binder, thickener or other viscosity modifier, processing aid, carrier, water-conditioning agent, foam inhibitor or foam generator, threshold agent or system, aesthetic enhancing agent (e.g., dye, odorant, perfume), other additive suitable for formulation with a clay stabilizer, or mixtures thereof.
- the additional clay treatment composition agent in a clay treatment composition will vary according to the specific clay treatment composition being manufactured and its intend use as one skilled in the art will appreciate.
- the clay treatment composition does not contain or is free of one or more of the additional clay treatment composition agents.
- one or more additional clay treatment composition agents When one or more additional clay treatment composition agents are used for clay treatment, they can be formulated together with the multiple charged cationic compounds derived from a polyamine as described here in the same clay treatment composition and be supplied to the stimulation fluid. Alternatively, some or all the additional clay treatment composition agent can be formulated into one or more different formulations and be supplied to the stimulation fluid. In other words, the additional clay treatment composition agents can be provided into a subterranean formation independently, simultaneously, or sequentially.
- the clay treatment compositions disclosed herein further include a biocide. In some other embodiments, the disclosed clay treatment compositions herein further include a carrier. In some other embodiments, the disclosed clay treatment compositions herein further include a biocide and carrier. In some embodiments, the disclosed methods or clay treatment compositions herein may consist of one or more multiple charged cationic compounds disclosed herein and carrier. In some embodiments, the clay treatment compositions disclosed herein consist of one or more multiple charged cationic compounds disclosed herein, a carrier, and a biocide.
- Biocides suitable for use may be oxidizing or non-oxidizing biocides.
- Oxidizing biocides can include, but are not limited to, bleach, chlorine, bromine, chlorine dioxide, peroxycarboxylic acid, peroxycarboxylic acid composition, and materials capable of releasing chlorine, bromine, or peroxide.
- Non-oxidizing biocides include, but are not limited to, glutaraldehyde, isothiazolin, 2,2-dibromo-3-nitrilopropionamide, 2-bromo-2-nitropropane-1,3 diol, 1-bromo-1-(bromomethyl)-1,3-propanedicarbonitrile, tetrachloroisophthalonitrile, alkyldimethylbenzylammonium chloride, dimethyl dialkyl ammonium chloride, didecyl dimethyl ammonium chloride, poly(oxyethylene(dimethyliminio)ethylene(dimethyliminio)ethylene dichloride, methylene bisthiocyanate, 2-decylthioethanamine, tetrakishydroxymethyl phosphonium sulfate, dithiocarbamate, cyanodithioimidocarbonate, 2-methyl-5-nitroimidazole-1-ethanol, 2-(2-bromo-2-nitroethenyl
- Suitable non-oxidizing biocides also include, for example, aldehydes (e.g., formaldehyde, glutaraldehyde, and acrolein), amine-type compounds (e.g., quaternary amine compounds and cocodiamine), halogenated compounds (e.g., 2-bromo-2-nitropropane-3-diol (Bronopol) and 2-2-dibromo-3-nitrilopropionamide (DBNPA)), sulfur compounds (e.g., isothiazolone, carbamates, and metronidazole), and quaternary phosphonium salts (e.g., tetrakis(hydroxymethyl)-phosphonium sulfate (THPS)).
- aldehydes e.g., formaldehyde, glutaraldehyde, and acrolein
- amine-type compounds e.g., quaternary amine compounds and cocodiamine
- Suitable oxidizing biocides include, for example, sodium hypochlorite, trichloroisocyanuric acids, dichloroisocyanuric acid, calcium hypochlorite, lithium hypochlorite, chlorinated hydantoins, stabilized sodium hypobromite, activated sodium bromide, brominated hydantoins, chlorine dioxide, ozone, peroxycarboxylic acid, peroxycarboxylic acid composition, and peroxides.
- the composition can comprise from about 0.1 wt-% to about 10 wt-%, from about 0.5 wt-% to about 5 wt-%, or from about 0.5 wt-% to about 4 wt-% of a biocide, based on total weight of the composition.
- a carrier in the disclosed clay treatment composition can be water, an organic solvent, or a combination of water and an organic solvent.
- the organic solvent can be an alcohol, a hydrocarbon, a ketone, an ether, an alkylene glycol, a glycol ether, an amide, a nitrile, a sulfoxide, an ester, or a combination thereof.
- suitable organic solvents include, but are not limited to, methanol, ethanol, propanol, isopropanol, butanol, 2-ethylhexanol, hexanol, octanol, decanol, 2-butoxyethanol, methylene glycol, ethylene glycol, 1,2-propylene glycol, 1,3-propylene glycol, diethyleneglycol monomethyl ether, diethylene glycol monoethyl ether, ethylene glycol monobutyl ether, ethylene glycol dibutyl ether, pentane, hexane, cyclohexane, methylcyclohexane, heptane, decane, dodecane, diesel, toluene, xylene, heavy aromatic naphtha, cyclohexanone, diisobutylketone, diethyl ether, propylene carbonate, N-methylpyrrolidinone, N,N-dimethyl
- the composition can comprise from about 1 wt-% to about 80 wt-%, from about 5 wt-% to about 50 wt-%, from about 5 wt-% to about 45 wt-%, from about 5 wt-% to about 30 wt-%, from about 5 wt-% to about 25 wt-%, from about 5 wt-% to about 20 wt-%, from about 5 wt-% to about 15 wt-%, from about 5 wt-% to about 10 wt-%, from about 10 wt-% to about 35 wt-%, from about 10 wt-% to about 25 wt-%, or from about 10 wt-% to about 35 wt-% of the one or more carriers, based on total weight of the composition.
- the clay treatment compositions disclosed herein further include a corrosion inhibitor. In some other embodiments, the disclosed clay treatment compositions herein further include a corrosion inhibitor and carrier. In some other embodiments, the disclosed clay treatment compositions herein further include a corrosion inhibitor, biocide, and carrier. In some embodiments, the disclosed clay treatment compositions herein may consist of one or more multiple charged cationic compounds disclosed herein, one or more corrosion inhibitors and carrier. In some embodiments, the clay treatment compositions disclosed herein consist of one or more multiple charged cationic compounds disclosed herein, a carrier, corrosion inhibitor, and a biocide.
- the clay treatment composition can comprise from about 0.1 wt-% to about 20 wt-%, from about 0.1 wt-% to about 10 wt-%, or from 0.1 to about 5 wt-% of the one or more corrosion inhibitors, based on total weight of the composition.
- a composition of the disclosure can comprise from about 0 wt-% to about 10 wt-% of the one or more corrosion inhibitors, based on total weight of the composition.
- the composition can comprise about 1.0 wt-%, about 1.5 wt-%, about 2.0 wt-%, about 2.5 wt-%, about 3.0 wt-%, about 3.5 wt-%, about 4.0 wt-%, about 4.5 wt-%, about 5.0 wt-%, about 5.5 wt-%, about 6.0 wt-%, about 6.5 wt-%, about 7.0 wt-%, about 7.5 wt-%, about 8.0 wt-%, about 8.5 wt-%, about 9.0 wt-%, about 9.5 wt-%, about 10.0 wt-%, about 10.5 wt-%, about 11.0 wt-%, about 11.5 wt-%, about 12.0 wt-%, about 12.5 wt-%, about 13.0 wt-%, about 13.5 wt-%, about 14.0 wt-%, about 14.5 wt-%, or
- Corrosion inhibitors for multi-metal protection are typically triazoles, such as, but not limited to, benzotriazole, halogenated triazoles, and nitro-substituted azoles.
- the one or more corrosion inhibitors can be an imidazoline compound, a quaternary ammonium compound, a pyridinium compound, or a combination thereof.
- the one or more corrosion inhibitor component can be an imidazoline.
- the imidazoline can be, for example, imidazoline derived from a diamine, such as ethylene diamine (EDA), diethylene triamine (DETA), triethylene tetraamine (TETA) etc. and a long chain fatty acid such as tall oil fatty acid (TOFA).
- the imidazoline can be an imidazoline of Formula (1A) or an imidazoline derivative.
- Representative imidazoline derivatives include an imidazolinium compound of Formula (2A) or a bis-quaternized compound of Formula (3A).
- the one or more corrosion inhibitor component can include an imidazoline of Formula (1A):
- R 10a is a C 1 -C 20 alkyl or a C 1 -C 20 alkoxyalkyl group
- R 11a is hydrogen, C 1 -C 6 alkyl, C 1 -C 6 hydroxyalkyl, or C 1 -C 6 arylalkyl
- R 12a and R 13a are independently hydrogen or a C 1 -C 6 alkyl group.
- the imidazoline includes an R 10a which is the alkyl mixture typical in tall oil fatty acid (TOFA), and R 11a , R 12a and R 13a are each hydrogen.
- the one or more additional corrosion inhibitor component can be an imidazolinium compound of Formula (2A):
- R 10a is a C 1 -C 20 alkyl or a C 1 -C 20 alkoxyalkyl group
- R 11a and R 14a are independently hydrogen, C 1 -C 6 alkyl, C 1 -C 6 hydroxyalkyl, or C 1 -C 6 arylalkyl
- R 12a and R 13a are independently hydrogen or a C 1 -C 6 alkyl group
- X ⁇ is a halide (such as chloride, bromide, or iodide), carbonate, sulfonate, phosphate, or the anion of an organic carboxylic acid (such as acetate).
- the imidazolinium compound includes 1-benzyl-1-(2-hydroxyethyl)-2-tall-oil-2-imidazolinium chloride.
- the one or more additional corrosion inhibitors can be a bis-quaternized compound having the formula (3A):
- R 1a and R 2a are each independently unsubstituted branched, chain or ring alkyl or alkenyl having from 1 to about 29 carbon atoms; partially or fully oxygenized, sulfurized, and/or phosphorylized branched, chain, or ring alkyl or alkenyl having from 1 to about 29 carbon atoms; or a combination thereof;
- R 3a and R 4a are each independently unsubstituted branched, chain or ring alkylene or alkenylene having from 1 to about 29 carbon atoms; partially or fully oxygenized, sulfurized, and/or phosphorylized branched, chain, or ring alkylene or alkenylene having from 1 to about 29 carbon atoms; or a combination thereof;
- L 1 and L 2 are each independently absent, H, —COOH, —SO 3 H, —PO 3 H, —COOR 5a , —CONH 2 , —CONHR 5a , or —CON(R 5a ) 2
- R 1a and R 2a are each independently C 6 -C 22 alkyl, C 8 -C 20 alkyl, C 12 -C 18 alkyl, C 16 -C 18 alkyl, or a combination thereof;
- R 1a and R 4a are C 1 -C 10 alkylene, C 2 -C 8 alkylene, C 2 -C 6 alkylene, or C 2 -C 3 alkylene;
- n is 0 or 1;
- x is 2;
- y is 1;
- R 3 and R 4 are —C 2 H 2 —;
- L 1 is —COOH, —SO 3 H, or —PO 3 H; and
- L 2 is absent, H, —COOH, —SO 3 H, or —PO 3 H.
- R 1a and R 2a can be derived from a mixture of tall oil fatty acids and are predominantly a mixture of C 17 H 33 and C 17 H 31 or can be C 16 -C 18 alkyl;
- R 1a and R 4a can be C 2 -C 3 alkylene such as —C 2 H 2 —; n is 1 and L 2 is —COOH or n is 0 and L 2 is absent or H; x is 2; y is 1; R 1a and R 4a are —C 2 H 2 —; and L 1 is —COOH.
- the one or more corrosion inhibitors can be a bis-quaternized imidazoline compound having the formula (3A) wherein R 1a and R 2a are each independently C 6 -C 22 alkyl, C 8 -C 20 alkyl, C 12 -C 18 alkyl, or C 16 -C 18 alkyl or a combination thereof; R 4a is C 1 -C 10 alkylene, C 2 -C 8 alkylene, C 2 -C 6 alkylene, or C 2 -C 3 alkylene; x is 2; y is 1; n is 0; L 1 is —COOH, —SO 3 H, or —PO 3 H; and L 2 is absent or H.
- R 1a and R 2a are each independently C 6 -C 22 alkyl, C 8 -C 20 alkyl, C 12 -C 18 alkyl, or C 16 -C 18 alkyl or a combination thereof; R 4a is C 1 -C 10 alkylene, C 2 -C 8 al
- a bis-quaternized compound has the formula (3A) wherein R 1a and R 2a are each independently C 16 -C 18 alkyl; R 4a is —C 2 H 2 —; x is 2; y is 1; n is 0; L 1 is —COOH, —SO 3 H, or —PO 3 H and L 2 is absent or H.
- the one or more corrosion inhibitors can be a quaternary ammonium compound of Formula (4A):
- R 1a , R 2a , and R 3a are independently C 1 to C 20 alkyl, R 4a is methyl or benzyl, and X ⁇ is a halide or methosulfate.
- Suitable alkyl, hydroxyalkyl, alkylaryl, arylalkyl or aryl amine quaternary salts include those alkylaryl, arylalkyl and aryl amine quaternary salts of the formula [N + R 5a R 6a R 7a R 8a ][X ⁇ ] wherein R 5a , R 7a , and R 8a contain one to 18 carbon atoms, and X is Cl, Br or I.
- R 5a , R 6a , R 7a , and R 8a can each be independently alkyl (e.g., C 1 -C 18 alkyl), hydroxyalkyl (e.g., C 1 -C 18 hydroxyalkyl), and arylalkyl (e.g., benzyl).
- the mono or polycyclic aromatic amine salt with an alkyl or alkylaryl halide include salts of the formula [N + R 5a R 6a R 7a R 8a ][X ⁇ ] wherein R 5a , R 6a , R 7a , and R 8a contain one to 18 carbon atoms and at least one aryl group, and X is Cl, Br or I.
- Suitable quaternary ammonium salts include, but are not limited to, a tetramethyl ammonium salt, a tetraethyl ammonium salt, a tetrapropyl ammonium salt, a tetrabutyl ammonium salt, a tetrahexyl ammonium salt, a tetraoctyl ammonium salt, a benzyltrimethyl ammonium salt, a benzyltriethyl ammonium salt, a phenyltrimethyl ammonium salt, a phenyltriethyl ammonium salt, a cetyl benzyldimethyl ammonium salt, a hexadecyl trimethyl ammonium salt, a dimethyl alkyl benzyl quaternary ammonium salt, a monomethyl dialkyl benzyl quaternary ammonium salt, or a trialkyl benzyl quaternary ammonium
- the quaternary ammonium salt can be a benzyl trialkyl quaternary ammonium salt, a benzyl triethanolamine quaternary ammonium salt, or a benzyl dimethylaminoethanolamine quaternary ammonium salt.
- the one or more corrosion inhibitor component can be a pyridinium salt such as those represented by Formula (5A):
- R 9a is an alkyl group, an aryl group, or an arylalkyl group, wherein said alkyl groups have from 1 to about 18 carbon atoms and X ⁇ is a halide such as chloride, bromide, or iodide.
- X ⁇ is a halide such as chloride, bromide, or iodide.
- alkyl pyridinium salts and alkyl pyridinium benzyl quats.
- Exemplary compounds include methyl pyridinium chloride, ethyl pyridinium chloride, propyl pyridinium chloride, butyl pyridinium chloride, octyl pyridinium chloride, decyl pyridinium chloride, lauryl pyridinium chloride, cetyl pyridinium chloride, benzyl pyridinium chloride and an alkyl benzyl pyridinium chloride, preferably wherein the alkyl is a C 1 -C 6 hydrocarbyl group.
- the pyridinium compound includes benzyl pyridinium chloride.
- the one or more additional corrosion inhibitors can be a phosphate ester, monomeric or polymeric fatty acid, alkoxylated amine, or mixture thereof.
- the one or more corrosion inhibitor component can be a phosphate ester.
- Suitable mono-, di- and tri-alkyl as well as alkylaryl phosphate esters and phosphate esters of mono, di, and triethanolamine typically contain between from 1 to about 18 carbon atoms.
- Preferred mono-, di-and trialkyl phosphate esters, alkylaryl or arylalkyl phosphate esters are those prepared by reacting a C 3 -C 18 aliphatic alcohol with phosphorous pentoxide.
- the phosphate intermediate interchanges its ester groups with triethylphosphate producing a broader distribution of alkyl phosphate esters.
- the phosphate ester can be made by admixing with an alkyl diester, a mixture of low molecular weight alkyl alcohols or diols.
- the low molecular weight alkyl alcohols or diols preferably include C 6 to C 10 alcohols or diols.
- phosphate esters of polyols and their salts containing one or more 2-hydroxyethyl groups, and hydroxylamine phosphate esters obtained by reacting polyphosphoric acid or phosphorus pentoxide with hydroxylamines such as diethanolamine or triethanolamine are preferred.
- the one or more corrosion inhibitors can be a monomeric or polymeric fatty acid.
- Preferred monomeric fatty acids are C 14 -C 22 saturated and unsaturated fatty acids as well as polymeric products obtained by polymerizing one or more of such fatty acids.
- the one or more corrosion inhibitors can be an alkoxylated amine.
- the alkoxylated amine can be an ethoxylated alkyl amine.
- the alkoxylated amine can be ethoxylated tallow amine.
- the clay treatment compositions disclosed herein can further comprise a dispersant.
- a dispersant keeps particulate matter present in the water of a subterranean formation or stimulation fluid dispersed, so that it does not agglomerate.
- the composition can comprise from about 0.1 wt-% to about 10 wt-%, from about 0.5 wt-% to about 5 wt-%, or from about 0.5 wt-% to about 4 wt-% of a dispersant, based on total weight of the composition.
- a dispersant may be an acrylic acid polymer, maleic acid polymer, copolymer of acrylic acid with sulfonated monomers, alkyl esters thereof, or combination thereof. These polymers may include terpolymers of acrylic acid, acrylamide and sulfonated monomers. These polymers may also include quad-polymers consisting of acrylic acid and three other monomers.
- Suitable dispersants include, but are not limited to, aliphatic phosphonic acids with 2-50 carbons, such as hydroxyethyl diphosphonic acid, and aminoalkyl phosphonic acids, e.g., polyaminomethylene phosphonates with 2-10 N atoms e.g., each bearing at least one methylene phosphonic acid group; examples of the latter are ethylenediamine tetra(methylene phosphonate), diethylenetriamine penta(methylene phosphonate), and the triamine- and tetramine-polymethylene phosphonates with 2-4 methylene groups between each N atom, at least 2 of the numbers of methylene groups in each phosphonate being different.
- Other suitable dispersion agents include lignin, or derivatives of lignin such as lignosulfonate and naphthalene sulfonic acid and derivatives.
- the clay treatment composition can further comprise an organic sulfur compound, such as a mercaptoalkyl alcohol, mercaptoacetic acid, thioglycolic acid, 3,3′-dithiodipropionic acid, sodium thiosulfate, thiourea, L-cysteine, tert-butyl mercaptan, sodium thiosulfate, ammonium thiosulfate, sodium thiocyanate, ammonium thiocyanate, sodium metabisulfite, or a combination thereof.
- the mercaptoalkyl alcohol comprises 2-mercaptoethanol. Such compounds are used as synergists in the composition.
- the organic sulfur compound can constitute from about 0.5 wt-% to about 15 wt-% of the composition, based on total weight of the composition, preferably from about 1 wt-% to about 10 wt-% and more preferably from about 1 wt-% to about 5 wt-%.
- the organic sulfur compound can constitute about 1 wt-%, about 2 wt-%, about 3 wt-%, about 4 wt-%, about 5 wt-%, about 6 wt-%, about 7 wt-%, about 8 wt-%, about 9 wt-%, about 10 wt-%, about 11 wt-%, about 12 wt-%, about 13 wt-%, about 14 wt-%, or about 15 wt-% of the composition.
- the clay treatment composition can further comprise a de-emulsifier.
- the de-emulsifier comprises an oxyalkylate polymer, such as a polyalkylene glycol.
- the de-emulsifier can constitute from about 0.1 wt-% to about 10 wt-%, from about 0.5 wt-% to about 5 wt. %, or from about 0.5 wt-% to about 4 wt-% of the composition, based on total weight of the composition.
- the de-emulsifier can constitute about 0.5 wt-%, about 1 wt-%, about 1.5 wt-%, about 2 wt-%, about 2.5 wt-%, about 3 wt-%, about 3.5 wt-%, about 4 wt-%, about 4.5 wt-% or about 5 wt-% of the composition.
- the clay treatment composition can further comprise an asphaltene inhibitor.
- the composition can comprise from about 0.1 wt-% to about 10 wt-%, from about 0.1 wt-% to about 5 wt-%, or from about 0.5 wt-% to about 4 wt-% of an asphaltene inhibitor, based on total weight of the composition.
- Suitable asphaltene inhibitors include, but are not limited to, aliphatic sulfonic acids; alkyl aryl sulfonic acids; aryl sulfonates; lignosulfonates; alkylphenol/aldehyde resins and similar sulfonated resins; polyolefin esters; polyolefin imides; polyolefin esters with alkyl, alkylenephenyl or alkylenepyridyl functional groups; polyolefin amides; polyolefin amides with alkyl, alkylenephenyl or alkylenepyridyl functional groups; polyolefin imides with alkyl, alkylenephenyl or alkylenepyridyl functional groups; alkenyl/vinyl pyrrolidone copolymers; graft polymers of polyolefins with maleic anhydride or vinyl imidazole; hyperbranched polyester amides; polyalkoxylated
- the clay treatment composition can further comprise a paraffin inhibitor.
- the composition can comprise from about 0.1 wt-% to about 10 wt-%, from about 0.1 wt-% to about 5 wt-%, or from about 0.5 wt-% to about 4 wt-% of a paraffin inhibitor, based on total weight of the composition.
- Suitable paraffin inhibitors include, but are not limited to, paraffin crystal modifiers, and dispersant/crystal modifier combinations.
- Suitable paraffin crystal modifiers include, but are not limited to, alkyl acrylate copolymers, alkyl acrylate vinylpyridine copolymers, ethylene vinyl acetate copolymers, maleic anhydride ester copolymers, branched polyethylenes, naphthalene, anthracene, microcrystalline wax and/or asphaltenes.
- Suitable paraffin dispersants include, but are not limited to, dodecyl benzene sulfonate, oxyalkylated alkylphenols, and oxyalkylated alkylphenolic resins.
- the clay treatment composition can further comprise a scale inhibitor.
- the composition can comprise from about 0.1 wt-% to about 20 wt-%, from about 0.5 wt-% to about 10 wt-%, or from about 1 wt-% to about 10 wt-% of a scale inhibitor, based on total weight of the composition.
- Suitable scale inhibitors include, but are not limited to, phosphates, phosphate esters, phosphoric acids, phosphonates, phosphonic acids, polyacrylamides, salts of acrylamidomethyl propane sulfonate/acrylic acid copolymer (AMPS/AA), phosphinated maleic copolymer (PHOS/MA), mono-, bis- and oligomeric phosphinosuccinic acid (PSO) derivatives, polycarboxylic acid, hydrophobically modified polycarboxylic acid, and salts of a polymaleic acid/acrylic acid/acrylamidomethyl propane sulfonate terpolymer (PMA/AA/AMPS).
- AMPS/AA acrylamidomethyl propane sulfonate/acrylic acid copolymer
- PHOS/MA phosphinated maleic copolymer
- PSO mono-, bis- and oligomeric phosphinosuccinic acid
- the clay treatment composition can further comprise an emulsifier.
- the composition can comprise from about 0.1 wt-% to about 10 wt-%, from about 0.5 wt-% to about 5 wt-%, or from about 0.5 wt-% to about 4 wt-% of an emulsifier, based on total weight of the composition.
- Suitable emulsifiers include, but are not limited to, salts of carboxylic acids, products of acylation reactions between carboxylic acids or carboxylic anhydrides and amines, and alkyl, acyl and amide derivatives of saccharides (alkyl-saccharide emulsifiers).
- the clay treatment composition can further comprise a water clarifier.
- the composition can comprise from about 0.1 to 10 wt-%, from about 0.5 to 5 wt-%, or from about 0.5 to 4 wt-% of a water clarifier, based on total weight of the composition.
- Suitable water clarifiers include, but are not limited to, inorganic metal salts such as alum, aluminum chloride, and aluminum chlorohydrate, or organic polymers such as acrylic acid-based polymers, acrylamide-based polymers, polymerized amines, alkanolamines, thiocarbamates, and cationic polymers such as diallyldimethylammonium chloride (DADMAC).
- DADMAC diallyldimethylammonium chloride
- the clay treatment composition can further comprise an emulsion breaker.
- the composition can comprise from about 0.1 wt-% to about 10 wt-%, from about 0.5 wt-% to about 5 wt-%, or from about 0.5 wt-% to about 4 wt-% of an emulsion breaker, based on total weight of the composition.
- Suitable emulsion breakers include, but are not limited to, dodecylbenzylsulfonic acid (DDBSA), the sodium salt of xylenesulfonic acid (NAXSA), epoxylated and propoxylated compounds, anionic, cationic and nonionic surfactants, and resins, such as phenolic and epoxide resins.
- the clay treatment composition can further comprise a hydrogen sulfide scavenger.
- the composition can comprise from about 1 wt-% to about 50 wt-%, from about 1 wt-% to about 40 wt-%, or from about 1 wt-% to about 30 wt-% of a hydrogen sulfide scavenger, based on total weight of the composition.
- Suitable additional hydrogen sulfide scavengers include, but are not limited to, oxidants (e.g., inorganic peroxides such as sodium peroxide or chlorine dioxide); aldehydes (e.g., of 1-10 carbons such as formaldehyde, glyoxal, glutaraldehyde, acrolein, or methacrolein; triazines (e.g., monoethanolamine triazine, monomethylamine triazine, and triazines from multiple amines or mixtures thereof); condensation products of secondary or tertiary amines and aldehydes, and condensation products of alkyl alcohols and aldehydes.
- oxidants e.g., inorganic peroxides such as sodium peroxide or chlorine dioxide
- aldehydes e.g., of 1-10 carbons such as formaldehyde, glyoxal, glutaraldehyde, acrolein, or
- the clay treatment composition can further comprise a gas hydrate inhibitor.
- the composition can comprise from about 0.1 wt-% to about 25 wt-%, from about 0.5 wt-% to about 20 wt-%, or from about 1 wt-% to about 10 wt-% of a gas hydrate inhibitor, based on total weight of the composition.
- Suitable gas hydrate inhibitors include, but are not limited to, thermodynamic hydrate inhibitors (THI), kinetic hydrate inhibitors (KHI), and anti-agglomerates (AA).
- Suitable thermodynamic hydrate inhibitors include, but are not limited to, sodium chloride, potassium chloride, calcium chloride, magnesium chloride, sodium bromide, formate brines (e.g.
- polyols such as glucose, sucrose, fructose, maltose, lactose, gluconate, monoethylene glycol, diethylene glycol, triethylene glycol, mono-propylene glycol, dipropylene glycol, tripropylene glycols, tetrapropylene glycol, monobutylene glycol, dibutylene glycol, tributylene glycol, glycerol, diglycerol, triglycerol, and sugar alcohols (e.g.
- sorbitol, mannitol methanol
- propanol ethanol
- glycol ethers such as diethyleneglycol monomethylether, ethyleneglycol monobutylether
- alkyl or cyclic esters of alcohols such as ethyl lactate, butyl lactate, methylethyl benzoate
- the clay treatment composition can further comprise a kinetic hydrate inhibitor.
- the composition can comprise from about 0.1 wt-% to about 25 wt-%, from about 0.5 wt-% to about 20 wt-%, or from about 1 wt-% to about 10 wt-% of a kinetic hydrate inhibitor, based on total weight of the composition.
- Suitable kinetic hydrate inhibitors and anti-agglomerates include, but are not limited to, polymers and copolymers, polysaccharides (such as hydroxyethylcellulose (HEC), carboxymethylcellulose (CMC), starch, starch derivatives, and xanthan), lactams (such as polyvinylcaprolactam, polyvinyl lactam), pyrrolidones (such as polyvinyl pyrrolidone of various molecular weights), surfactants (such as fatty acid salts, ethoxylated alcohols, propoxylated alcohols, sorbitan esters, ethoxylated sorbitan esters, polyglycerol esters of fatty acids, alkyl glucosides, alkyl polyglucosides, alkyl sulfates, alkyl sulfonates, alkyl ester sulfonates, alkyl aromatic sulfonates, alkyl betaine, alkyl amid
- the clay treatment composition can further comprise a pH modifier.
- the composition can comprise from about 0.1 wt-% to about 20 wt-%, from about 0.5 wt-% to about 10 wt-%, or from about 0.5 wt-% to about 5 wt-% of a pH modifier, based on total weight of the composition.
- Suitable pH modifiers include, but are not limited to, alkali hydroxides, alkali carbonates, alkali bicarbonates, alkaline earth metal hydroxides, alkaline earth metal carbonates, alkaline earth metal bicarbonates and mixtures or combinations thereof.
- Exemplary pH modifiers include sodium hydroxide, potassium hydroxide, calcium hydroxide, calcium oxide, sodium carbonate, potassium carbonate, sodium bicarbonate, potassium bicarbonate, magnesium oxide, and magnesium hydroxide.
- the clay treatment composition can further comprise a surfactant.
- the composition can comprise from about 0.1 wt-% to about 10 wt-%, from about 0.5 wt-% to about 5 wt-%, or from about 0.5 wt-% to about 4 wt-% of a surfactant, based on total weight of the composition.
- Suitable surfactants include, but are not limited to, anionic surfactants and nonionic surfactants.
- Anionic surfactants include alkyl aryl sulfonates, olefin sulfonates, paraffin sulfonates, alcohol sulfates, alcohol ether sulfates, alkyl carboxylates and alkyl ether carboxylates, and alkyl and ethoxylated alkyl phosphate esters, and mono and dialkyl sulfosuccinates and sulfosuccinamates.
- Nonionic surfactants include alcohol alkoxylates, alkylphenol alkoxylates, block copolymers of ethylene, propylene and butylene oxides, alkyl dimethyl amine oxides, alkyl-bis(2-hydroxyethyl) amine oxides, alkyl amidopropyl dimethyl amine oxides, alkylamidopropyl-bis(2-hydroxyethyl) amine oxides, alkyl polyglucosides, polyalkoxylated glycerides, sorbitan esters and polyalkoxylated sorbitan esters, and alkoyl polyethylene glycol esters and diesters.
- amphoteric surfactants such as alkyl amphoacetates and amphodiacetates, alkyl amphopropionates and amphodipropionates, and alkyliminodipropionate.
- the clay treatment composition can further comprise a friction reducer, cross linker, flowback aid, viscosifier, and the like.
- the clay treatment composition can be formulated into compositions comprising the following components shown in Table 1. These formulations include the ranges of the components listed and can optionally include additional agents. The values in Tables 1A-1E below are weight percentages.
- Exemplary Clay Treatment Compositions Composition ID 8 9 10 11 12 13 14 15 16 Multiple 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 Charged Cationic Compound(s) Friction 0 0 0.025-0.4 0.025-0.4 0.025-0.4 0.025-0.4 0.025-0.4 0.025-0.4 0.025-0.4 0.025-0.4 0.025-0.4 0.025-0.4 reducing agent Flowback aid 0.025-0.2 0.025-0.2 0 0 0 0 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 Viscosifier 0.025-1.0 0.025-1.0 0.025-1.0 0.025-1.0 0.025-1.0 0.025-1.0 0 0.025-1.0 0.025-1.0 0.025-1.0 0.025-1.0 0.0
- Exemplary Clay Treatment Compositions Composition ID 17 18 19 20 21 22 23 24 Multiple 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 Charged Cationic Compound(s) Friction 0 0 0 0.025-0.4 0.025-0.4 0.025-0.4 0.025-0.4 reducer Flowback aid 0 0 0 0 0 0 0 0 0.025-0.2 Viscosifier 0.025-1.0 0.025-1.0 0.025-1.0 0 0.025-1.0 0.025-1.0 0.025-1.0 0 Crosslinker 0 0.010-0.3 0.010-0.3 0 0 0 0.010-0.3 0 Scale 0.025-0.2 0 0.025-0.2 0.025-0.2 0 0.025-0.2 0 0.025-0.2 0 0 inhibitor Biocide 0.025-0.2 0.025
- Exemplary Clay Treatment Compositions Composition ID 25 26 27 28 29 30 31 32 Multiple 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 0.025-0.2 Charged Cationic Compound(s) Friction 0.025-0.4 0.025-0.4 0 0 0 0.025-0.4 0.025-0.4 0.025-0.4 reducer Flowback aid 0.025-0.2 0.025-0.2 0 0 0 0 0 0 0 0 Viscosifier 0 0.025-1.0 0.025-1.0 0.025-1.0 0.025-1.0 0 0 0.025-1.0 Crosslinker 0 0 0 0 0.010-0.3 0 0 0 0 Scale 0.025-0.2 0 0 0.025-0.2 0 0 0.025-0.2 0 inhibitor Biocide 0 0 0.025-0.2
- Exemplary Clay Treatment Compositions Composition ID 33 34 35 Multiple 0.025-0.2 0.025-0.2 0.025-0.2 Charged Cationic Compound(s) Friction 0.025-0.4 0.025-0.4 0 reducer Flowback aid 0.025-0.2 0 0 Viscosifier 0 0 0.025-1.0 Crosslinker 0 0 0 0 Scale inhibitor 0 0 0 Biocide 0 0 0 Water 99.2-99.925 99.4-99.95 98.8-99.95
- the clay treatment composition or the multiple charged cationic compounds may be added to a fluid or stimulation fluid for oil and gas operations, so the concentration of the composition or the compound in the fluid or stimulation fluid is from about 1 ppm to about 2,000 ppm or from about 400 ppm to about 7,000 ppm.
- the amount of the clay treatment composition or the multiple charged cationic compounds in the fluid may range from about 5 ppm to about 2,000 ppm, from about 50 ppm to about 2,000 ppm, from about 100 ppm to about 2,000 ppm, from about 200 ppm to about 2,000 ppm, from about 250 ppm to about 2,000 ppm, from about 300 ppm to about 2,000 ppm, from about 400 ppm to about 2,000 ppm, from about 100 ppm to about 1,000 ppm, from about 200 ppm to about 800 ppm, from about 300 ppm to about 700 ppm, from about 400 ppm to about 600 ppm, or from about 100 ppm to about 500 ppm.
- the clay treatment composition or the multiple charged cationic compounds may be added to the water of the subterranean formation to an amount ranging from about 50 ppm to about 2,000 ppm, from about 100 ppm to about 500 ppm, from about 250 ppm to about 2,000 ppm, or from about 200 ppm to about 800 ppm.
- the clay treatment composition or the multiple charged cationic compounds can be applied to any fluid or stimulation fluid used in crude oil or natural gas productions.
- a fluid to which the clay treatment composition or the multiple charged cationic compounds can be introduced can be an aqueous medium.
- the aqueous medium can comprise water, oil, and optionally liquid hydrocarbon.
- a fluid or gas treated with the clay treatment composition or the multiple charged cationic compounds can be at any selected temperature, such as ambient temperature or an elevated temperature.
- the fluid (e.g., liquid hydrocarbon) or gas can be at a temperature of from about 40° C. to about 250° C.
- the fluid or gas can be at a temperature of from about ⁇ 50° C. to about 300° C., from about 0° C. to about 200° C., from about 10° C. to about 100° C., or from about 20° C. to about 90° C.
- the fluid or gas can be at a temperature of about 22° C., about 23° C., about 24° C., about 25° C., about 26° C., about 27° C., about 28° C., about 29° C., about 30° C., about 31° C., about 32° C., about 33° C., about 34° C., about 35° C., about 36° C., about 37° C., about 38° C., about 39° C., or about 40° C.
- the fluid or gas can be at a temperature of about 85° C., about 86° C., about 87° C., about 88° C., about 89° C., about 90° C., about 91° C., about 92° C., about 93° C., about 94° C., about 95° C., about 96° C., about 97° C., about 98° C., about 99° C., or about 100° C.
- the clay treatment composition or the multiple charged cationic compounds can be added to a fluid or stimulation fluid at various levels of water cut.
- the water cut can be from 0% to 100% volume/volume (v/v), from 1% to 80% v/v, or from 1% to 60% v/v.
- the fluid can be an aqueous medium that contains various levels of salinity.
- the fluid can have a salinity of 0% to 25%, about 1% to 24%, or about 10% to 25% weight/weight (w/w) total dissolved solids (TDS).
- the clay treatment composition or the multiple charged cationic compounds can be introduced into a fluid, stimulation fluid, or gas by any appropriate method for ensuring dispersal through the fluid or gas.
- the clay treatment composition or the multiple charged cationic compounds can be added to a drilling fluid or stimulation fluid before the drilling or stimulation fluid contacts the subterranean formation.
- the clay treatment composition or the multiple charged cationic compounds can be added at a point in a flow line upstream from the point at which the fluid is used for oil and gas productions.
- the clay treatment composition or the multiple charged cationic compounds can be injected using mechanical equipment such as chemical injection pumps, piping tees, injection fittings, atomizers, quills, and the like.
- the clay treatment composition or the multiple charged cationic compounds can be pumped into an oil and/or gas pipeline using an umbilical line.
- a capillary injection system can be used to deliver the clay treatment composition or the multiple charged cationic compounds to a selected fluid.
- a fluid to which the clay treatment composition or the multiple charged cationic compounds can be introduced can be an aqueous medium.
- the aqueous medium can comprise water, oil, and optionally liquid hydrocarbon.
- a fluid to the clay treatment composition or the multiple charged cationic compounds can be introduced can be fracturing fluid, acidizing fluid, drilling fluid, drill-in fluid, stimulation fluid, gravel pack fluid, completion fluid, cementing fluid, other oil gas operation fluid, any other fluid for oil and gas production, or mixture thereof.
- the clay treatment composition or the multiple charged cationic compounds can be introduced into a liquid and a mixture of several liquids, a liquid and gas, liquid, solid, and gas.
- the clay treatment composition or the multiple charged cationic compounds can be injected into a gas stream as an aqueous or non-aqueous solution, mixture, or slurry.
- the fluid or gas can be passed through an absorption tower comprising the clay treatment composition or the multiple charged cationic compounds.
- the clay treatment composition or the multiple charged cationic compounds can be applied to a fluid or gas to provide any selected concentration.
- the clay treatment composition or the multiple charged cationic compounds are typically added to a flow line to provide an effective treating dose of the clay treatment composition or the multiple charged cationic compounds from about 0.01 ppm to about 5,000 ppm.
- the clay treatment composition or the multiple charged cationic compounds can be applied to a fluid or gas to provide an active concentration of about 1 parts per million (ppm) to about 1,000,000 ppm, from about 1 parts per million (ppm) to about 100,000 ppm, or from about 10 ppm to about 75,000 ppm.
- the polymer salts/compositions can be applied to a fluid to provide an actives concentration of from about 100 ppm to about 10,000 ppm, from about 200 ppm to about 8,000 ppm, or from about 500 ppm to about 6,000 ppm.
- the actives concentration means the concentration of clay treatment composition or the multiple charged cationic compounds.
- the clay treatment composition or the multiple charged cationic compounds can be applied to a fluid or gas to provide an active concentration of about 0.1 ppm, about 0.5 ppm, about 1 ppm, about 2 ppm, about 5 ppm, about 10 ppm, about 20 ppm, about 100 ppm, about 200 ppm, about 500 ppm, or about 1,000 ppm.
- the multiple charged cationic compounds, their salt or clay treatment composition can be applied to a fluid or gas to provide an actives concentration of about 0.125 ppm, about 0.25 ppm, about 0.625 ppm, about 1 ppm, about 1.25 ppm, about 2.5 ppm, about 5 ppm, about 10 ppm, about 20 ppm, about 40 ppm, about 60 ppm, about 100 ppm, about 200 ppm, about 400 ppm, about 600 ppm, about 800 ppm, about 1,000 ppm in the fluid or gas.
- Each fluid can have its own dose level requirements, and the effective dose level of the clay treatment composition or the multiple charged cationic compounds to sufficiently prevent clay swell, clay migration, or sludge formation can vary with the subterranean system in which it is used.
- the clay treatment composition or the multiple charged cationic compounds can be applied continuously, in batch, or a combination thereof.
- the clay treatment composition or the multiple charged cationic compounds dosing can be continuous.
- the clay treatment composition or the multiple charged cationic compounds dosing can be intermittent (e.g., batch treatment) or can be continuous/maintained and/or intermittent.
- Dosage rates for continuous treatments typically range from about 10 ppm to about 500 ppm, or from about 10 ppm to about 200 ppm.
- Dosage rates for batch treatments typically range from about 10 ppm to about 400,000 ppm, or from about 10 to about 20,000 ppm.
- the clay treatment composition or the multiple charged cationic compounds can be applied as a pill to a pipeline, providing a high dose (e.g., 20,000 ppm) of the composition.
- the flow rate of a flow line in which the clay treatment composition or the multiple charged cationic compounds is used can be between about 0.1 feet per second and about 100 feet per second, or between about 0.1 feet per second and about 50 feet per second.
- the clay treatment composition or the multiple charged cationic compounds can also be formulated with water to facilitate addition to the flow line.
- the clay treatment composition or the multiple charged cationic compounds can be dispensed in any suitable method generally known by one skilled in the art.
- a spray-type dispenser can be used.
- a spray-type dispenser functions by impinging a water spray upon an exposed surface of a composition to dissolve a portion of the composition, and then immediately directing the concentrate solution including the composition out of the dispenser to a storage reservoir or directly to a point of use.
- the clay treatment composition or the multiple charged cationic compounds can be dispensed by immersing either intermittently or continuously in a fluid used in oil and gas productions.
- the clay treatment composition or the multiple charged cationic compounds can then dissolve, for example, at a controlled or predetermined rate. The rate can be effective to maintain a concentration of the dissolved compounds or compositions that are effective for use according to the methods disclosed herein.
- the clay treatment composition disclosed herein can comprise from about 10 wt-% to about 90 wt-% of the additional clay treatment composition agent(s) and from about 10 wt-% to about 90 wt-% of one or more multiple charged cationic compounds.
- the clay treatment composition disclosed herein can comprise from about 20 wt-% to about 80 wt-% of the carrier, biocide, corrosion inhibitor, additional clay treatment composition agent, a combination thereof; from about 20 wt-% to about 80 wt-% of one or more multiple charged cationic compounds, from about 30 wt-% to about 70 wt-% of the carrier, biocide, corrosion inhibitor, additional clay treatment composition agent, a combination thereof and from about 30 wt-% to about 70 wt-% of one or more multiple charged cationic compounds, from about 40 wt-% to about 60 wt-% of the carrier, biocide, corrosion inhibitor, additional clay treatment composition agent, a combination thereof and from about 40 wt-% to about 60
- the clay treatment composition can comprise an oxyalkylated phenol-formaldehyde resin, a resin ester, an oxyalkylated polyalkylamine, a polyol, a cross-linked polyol with a di- or multi-functional cross linker, an isocyanate, an acid, or a combination thereof.
- the clay treatment composition can comprise one or more additional clay stabilizers depending on the properties of the subterranean formation.
- the multiple charged cationic compound and the additional clay treatment composition agent have a synergistic effect for preventing clay swell, clay migration, and sludge formation in a specific subterranean formation.
- a clay treatment composition for stabilizing swellable clays and/or reducing formation of sludge in a subterranean formation
- the clay treatment composition comprises a compound or its salt derived from an aza-Michael Addition Reaction between a polyamine and an ⁇ , ⁇ -unsaturated carbonyl compound according to the following formula
- X is NH or O
- R 2 is H, CH 3 , or an unsubstituted, linear or branched C 2 -C 10 alkyl, alkenyl, or alkynyl group
- R 3 is absent or an unsubstituted, linear C 1 -C 30 alkylene group
- Y is —NR 4 R 5 R 6 (+) , or a salt thereof
- R 4 , R 5 , and R 6 are independently a C 1 -C 10 alkyl group; wherein the compound is a multiple charged cationic compound having 2, 3, or more positive charges.
- a use solution of the clay treatment composition reduces clay swelling, clay migration, and sludge formation in the subterranean formation.
- a method of stabilizing swellable clays and/or reducing formation of sludge in a subterranean formation comprises providing a clay treatment composition into a subterranean formation, wherein the clay treatment composition comprises one or more additional clay treatment composition agents and a compound or its salt derived from an aza-Michael Addition Reaction between a polyamine and an ⁇ , ⁇ -unsaturated carbonyl compound according to the following formula
- X is NH or O;
- R 2 is H, CH 3 , or an unsubstituted, linear or branched C 2 -C 10 alkyl, alkenyl, or alkynyl group;
- R 3 is absent or an unsubstituted, linear C 1 -C 30 alkylene group;
- Y is —NR 4 R 5 R 6 (+) , or a salt thereof; and
- R 4 , R 5 , and R 6 are independently a C 1 -C 10 alkyl group; wherein the compound is a multiple charged cationic compound having 2, 3, or more positive charges.
- a use solution of the clay treatment composition reduces clay swelling, clay migration, and sludge formation in the subterranean formation.
- the polyamine is NH 2 —[R 10′ ] n —NH 2 , (RNH) n —RNH 2 , H 2 N—(RNH) n —RNH 2 , or H 2 N—(RN(R′)) n —RNH 2 , wherein R 10′ is a linear or branched, unsubstituted or substituted C 2 -C 10 alkylene group, or combination thereof; R is —CH 2 —, —CH 2 CH 2 —, —CH 2 CH 2 CH 2 —, —CH(CH 3 )CH 2 —, a linear or branched, unsubstituted or substituted C 4 -C 10 alkylene group, or combination thereof; R′ is —CH 2 —, —CH 2 CH 2 —, —CH 2 CH 2 CH 2 —, —CH(CH 3 )CH 2 —, a linear or branched, unsubstituted or substituted C 4 -
- FIG. 1 The structures of and the reactions leading to the exemplary multiple charged cationic compounds (I) using a linear polyethyleneimine is shown in FIG. 1 .
- FIG. 2 The scheme for preparation of exemplary cationic polymer compositions (II) using a branched polyethyleneimine is shown in FIG. 2 .
- k, 1, m, n, o, or p is an integer of 1-100;
- X is NH or O;
- R 2 is H, CH 3 , or an unsubstituted, linear or branched C 2 -C 10 alkyl group;
- R 3 is absent or an unsubstituted, linear C 1 -C 30 alkylene group;
- Y is —NR 4 R 5 R 6(+) or a salt thereof;
- R 4 , R 5 , and R 6 are independently C 1 -C 10 alkyl group or benzyl group.
- the structures I and II in FIG. 1 and FIG. 2 are depiction of generalized and exemplary reaction products.
- all the secondary and primary amines in the polyethyleneimine react with the activated olefins so that no secondary amines remain. It is possible that in the disclosed multiple charged cationic or anionic compounds, some secondary or primary amine groups do not react completely with the activated olefins and remain as primary or secondary amines in multiple charged cationic or anionic compounds or their salts.
- the multiple charged cationic or anionic compounds have one of the generic formula of NA 2 -[R 10′ ] n -NA 2 , (RNA) n -RNA 2 , A 2 N—(RNA) n -RNA 2 , or A 2 N—(RN(R′)) n —RNA 2 , wherein R 10′ is a linear or branched, unsubstituted or substituted C 2 -C 10 alkylene group, or combination thereof; R is —CH 2 —, —CH 2 CH 2 —, —CH 2 CH 2 CH 2 —, —CH(CH 3 )CH 2 —, a linear or branched, unsubstituted or substituted C 4 -C 10 alkylene group, or combination thereof; R′ is —CH 2 —, —CH 2 CH 2 —, —CH 2 CH 2 CH 2 —, —CH(CH 3 )CH 2 —, a linear or branched, unsub
- each of the compounds contain at least 2 non-proton and cationic A groups, at least 3 non-proton and cationic A groups, at least 4 non-proton and cationic A groups, at least 5 non-proton and cationic A groups, or more than 6 non-proton and cationic
- At least two of the primary NH 2 protons are
- water can be used as solvent.
- other solvent and/or diluent for the reaction is optional.
- a wide range of non-acidic solvents are suitable, such as, for example, acetonitrile, ethers (e.g., tetrahydrofuran (THF)), alcohols (e.g., methanol, ethanol, isopropanol, n-butanol, glycol, PEG, or a mixture) and the like
- reaction temperature can range from about 20° C. to about 150° C., more preferably from about 50° C. to about 100° C.
- the polyethyleneimine can be added to the solution of the quat-monomer.
- solution of the quat-monomer could be added to polyethyleneimine or both reagents can be added simultaneously.
- the polyamine is a linear, branched, or dendrimer polyamine with a general formula of —[RNH] n —, wherein R is —CH 2 CH 2 —, —CH 2 CH 2 CH 2 —, —CH(CH 3 )CH 2 —, a linear or branched, unsubstituted or substituted C 4 -C 10 alkylene group, or combination thereof and n is an integer of 3, 4, 5, 6, 7-9, or 10 to 1,000,000.
- the polyamine is a linear, branched, or dendrimer polyamine with a general formula of (RNH) n —RNH 2 , wherein R is —CH 2 —, —CH 2 CH 2 —, —CH 2 CH 2 CH 2 —, —CH(CH 3 )CH 2 —, a linear or branched, unsubstituted or substituted C 4 -C 10 alkylene group, or combination thereof and n can be from 2 to 1,000,000.
- R is the same in each monomer. In some other embodiments, R can be different from one monomer to another monomer.
- the polyamine is a linear, branched, or dendrimer polyamine with a general formula of H 2 N—(RNH) n —RNH 2 , wherein R is —CH 2 —, —CH 2 CH 2 —, —CH 2 CH 2 CH 2 —, —CH(CH 3 )CH 2 —, a linear or branched, unsubstituted or substituted C 4 -C 10 alkylene group, or combination thereof and n can be from 2 to 1,000,000.
- R is the same in each monomer. In some other embodiments, R can be different from one monomer to another monomer.
- the polyamine is a linear, branched, or dendrimer polyamine with a general formula of H 2 N—(RN(R′) n —RNH 2 , wherein R is —CH 2 —, —CH 2 CH 2 —, —CH 2 CH 2 CH 2 —, —CH(CH 3 )CH 2 —, a linear or branched, unsubstituted or substituted C 4 -C 10 alkylene group, or combination thereof; R′ is —CH 2 —, —CH 2 CH 2 —, —CH 2 CH 2 CH 2 —, —CH(CH 3 )CH 2 —, a linear or branched, unsubstituted or substituted C 4 -C 10 alkyl group, RNH 2 , RNHRNH 2 , or RN(RNH 2 ) 2 ; and n can be from 2 to 1,000,000.
- R or R′ is the same in each monomer.
- R or R′ is the same in each monomer.
- the polyamine is one with a general formula of NH 2 —[R 10′ ] n -Nth, wherein R 10′ is a linear or branched, unsubstituted or substituted C 4 -C 10 alkylene group, or combination thereof and n is an integer of 3, 4, 5, 6, 7-9, or 10 to 1,000,000.
- R 10′ can be different from one monomer to another monomer.
- the polyamine is one or more of polyamines under JEFFAMINE® by Huntsman.
- the polyamine comprises an alkyleneamine, the alkyleneamine comprising ethylenediamine, diethylenetriamine, triethylenetetramine, tetraethylenepentamine, pentaethylenehexamine, hexaethyleneheptamine, polyethyleneimine, tris(2-aminoethyl)amine, or a mixture thereof.
- the polyamine is a mixture of monoamine, diamine, and triamine with a polyether backbone or with a polyether backbone based on propylene oxide (PO), ethylene oxide (EO), or a mixture of both oxides.
- PO propylene oxide
- EO ethylene oxide
- the polyamine is an unmodified polyamine. In some other embodiments, the polyamine is a modified polyamine. As used herein, a “modified polyamine” refers to a polyamine in which one or more NH protons is substituted by a non-proton group, such as an alkyl.
- the polyamine is an ethoxylated polyamine, propylated polyamine, polyamine with polyquat, polyamine with polyglycerol, or combination thereof.
- the polyamine is a linear, branched, or dendrimer polyethyleneimine. In some other embodiments, the polyamine comprises only primary and secondary amine groups. In some embodiments, the polyamine comprises only primary, secondary, and tertiary amine groups. In some other embodiments, the polyamine comprises only primary and tertiary amine groups.
- the polyamine is a single compound. In some other embodiments, the polyamine is a mixture of two or more different polyamines, wherein the different polyamines have different molecular weight, different structure, or both.
- the polyamine has an average molecular weight (M w ) of from about 60 to about 2,000,000 Da. In some other embodiments, the polyamine has an average molecular weight (M w ) of from about 60 to about 5,000 Da. In yet some other embodiments, the polyamine has an average molecular weight (M w ) of from about 60 to about 25,000 Da.
- the polyamine has an average molecular weight (M w ) of about 60-200, about 100-400, about 100-600, about 600-5,000, about 600-800, about 800-2,000, about 800-5,000, about 100-2,000,000, about 100-25,000, about 600-25,000, about 800-25,000, about 600-750,000, about 800-750,000, about 25,000-750,000, about 750,000-2,000,000, about 100, about 200, about 300, about 400, about 500, about 600, about 700, about 800, about 1,000, about 1,500, about 2,000, about 3,000, about 5,000, about 8,000, about 10,000, about 15,000, about 20,000, about 50,000, about 100,000, about 250,000, about 500,000, about 1,000,000, about 2,000,000, or any value there between.
- M w average molecular weight
- the polyamine is diamine or triamine having an average molecular weight (M w ) of from about 60 to about 4,000 or from about 60 to about 1,300.
- the compound is a mixture derived from a linear polyethyleneimine and (3-Acrylamidopropyl)trimethylammonium chloride (APTAC). In some other embodiments, the compound is a mixture derived from a linear polyethyleneimine and [3-(Methacryloylamino)propyl]trimethylammonium chloride (MAPTAC).
- the multiple charged cationic compound is a mixture derived from a branched polyethyleneimine and 3-Acrylamidopropyl)trimethylammonium chloride (APTAC).
- the compound is a mixture derived from a linear polyethyleneimine and [3-(methacryloylamino)propyl]trimethylammonium chloride (MAPTAC).
- the ⁇ , ⁇ -unsaturated carbonyl compound is (3-Acrylamidopropyl)trimethylammonium chloride (APTAC), [3-(Methacryloylamino)propyl]trimethylammonium chloride (MAPTAC), 2-(acryloyloxy)-N,N,N-trimethylethanaminium chloride (DMAEA-MCQ), N,N-dimethylaminoethyl acrylate benzyl chloride quaternary salt (DMAEA-BCQ), or 2-(methacryloyloxy)-N,N,N-trimethylethan-1-aminium methyl sulfate (DMAEA-MSQ).
- APITAC 3-Acrylamidopropyl)trimethylammonium chloride
- MAEA-MCQ 2-(acryloyloxy)-N,N,N-trimethylethanaminium chloride
- DMAEA-BCQ N,N-dimethylamino
- the ⁇ , ⁇ -unsaturated carbonyl compound is (3-Acrylamidopropyl)trimethylammonium chloride (APTAC), [3-(Methacryloylamino)propyl]trimethylammonium chloride (MAPTAC), or mixture thereof.
- the ⁇ , ⁇ -unsaturated carbonyl compound is 2-(acryloyloxy)-N,N,N-trimethylethanaminium chloride (DMAEA-MCQ), N,N-dimethylaminoethyl acrylate benzyl chloride quaternary salt (DMAEA-BCQ), 2-(methacryloyloxy)-N,N,N-trimethylethan-1-aminium methyl sulfate (DMAEA-MSQ), or a mixture thereof.
- DAEA-MCQ 2-(acryloyloxy)-N,N,N-trimethylethanaminium chloride
- DMAEA-BCQ N,N-dimethylaminoethyl acrylate benzyl chloride quaternary salt
- DMAEA-MSQ 2-(methacryloyloxy)-N,N,N-trimethylethan-1-aminium methyl sulfate
- X is NH. In some other embodiments, X is O.
- R 2 is H. In some embodiments, R 2 is CH 3 . In yet some other embodiments, R 2 is CH 3 CH 3 , CH 2 CH 2 CH 3 , or CH(CH 3 ) 2 .
- Y is —NR 4 R 5 R 6 (+) , and R 4 , R 5 , and R 6 are independently CH 3 .
- Y is —NR 4 R 5 R 6 (+) , and R 4 and R 5 , independently CH 3 , and R 6 is a C 2 -C 12 aromatic alkyl.
- Y is —NR 4 R 5 R 6 (+) , and R 4 and R 5 , independently CH 3 , and R 6 is —CH 2 —C 6 H 6 .
- Y is —NR 4 R 5 R 6 (+) and the counter ion for Y is any negative charged ion or species.
- the counter ion for Y is chloride, bromide, fluoride, iodide, acetate, aluminate, cyanate, cyanide, dihydrogen phosphate, dihydrogen phosphite, formate, carbonate, hydrogen carbonate, hydrogen oxalate, hydrogen sulfate, hydroxide, nitrate, nitrite, thiocyanate, or a combination thereof.
- R 3 is CH 2 . In some other embodiments, R 3 is CH 2 CH 2 . In other embodiments, R 3 is C(CH 3 ) 2 . In yet some other embodiments, R 3 is an unsubstituted, linear, and saturated C 1 -C 20 alkylene group. In some embodiments, R 3 is an unsubstituted, linear, and unsaturated C 1 -C 20 alkylene group.
- R 3 is a linear C 8 -C 18 alkyl, alkenyl, or alkynyl group. In some other embodiments, R 3 is a branched C 8 -C 20 alkyl, alkenyl, or alkynyl group. In some embodiments, the compound is a single multiple charged cationic compound.
- the compound is a mixture of two or more multiple charged cationic compounds, wherein the two or more different compounds differ from other by molecular weight, structure, net charge, or combination thereof.
- the compound is a mixture of at least two different multiple charged cationic compounds derived from the same polyamine and the ⁇ , ⁇ -unsaturated carbonyl compound.
- the compound is a mixture of two or more different multiple charged cationic compounds derived from different polyamines and the same ⁇ , ⁇ -unsaturated carbonyl compound.
- the compound is a mixture of two or more different multiple charged cationic compounds derived from different polyamines and different ⁇ , ⁇ -unsaturated carbonyl compounds.
- the compound has an average molecular weight (M w ) of from about 100 to about 2,000,000 Da. In some other embodiments, the compound or modified compound has an average molecular weight (M w ) of from about 100 to about 5,500 Da or from about 100 to about 10,000 Da.
- the compound has at least 10, 15, 20, or 30 positive charges. In some other embodiments, the compound has at least 4, 5, 6, 7, or 8 positive charges. In yet some other embodiments, the compound has average net charges of from 3 to 100. In some other embodiments, the compound has average net charges of from 3 to 15.
- the compound has average net charges of from 3 to 100. In some embodiments, the compound has average net charges of from 3 to 15.
- the compound has average net charge of from 3 to 10 and an average molecular weight (M w ) of from about 200 to about 500, from about 500 to about 1,000, from about 1,000 to about 2,000, from about 2,000 to about 3,000, from about 3,000, to about 4,000, from about 4,000 to about 5,000, from about 5,000 to about 6,000, from about 6,000 to about 8,000, from about 8,000 to about 10,000, from about 10,000 to about 20,000, from about 20,000 to about 40,000, from about 40,000 to about 80,000, from 80,000 to about 100,000, from 100,000 to about 500,000, or from 500,000 to about 2,000,000.
- M w average molecular weight
- the compound has average net charges of from 10 to 30 and an average molecular weight (M w ) of from about 200 to about 500, from about 500 to about 1,000, from about 1,000 to about 2,000, to about 3,000, from about 3,000, to about 4,000, or from about 4,000 to about 5,000.
- M w average molecular weight
- R 2 is H
- X is NH
- R 3 is CH 2 CH 2
- Y is CH 2 —N + (CH 3 ) 3 Cl ⁇ .
- the polyamine is ethylenediamine, diethylenetriamine, triethylenetetramine, tetraethylenepentamine, pentaethylenehexamine, hexaethyleneheptamine, polyethyleneimine, tris(2-aminoethyl)amine, or a mixture thereof
- the polyamine is a polyethyleneimine or a mixture of polyethyleneimines.
- the polyamine is a polyethyleneimine having an average molecular weight (M w ) of from about 1,000 to about 5,000.
- the polyamine is diamine or triamine having an average molecular weight (M w ) of from about 130 to about 4,000.
- the compound is soluble or dispersible in water or the clay treatment composition.
- the clay treatment composition further comprises one or more additional clay treatment composition agents.
- the clay treatment composition comprises a carrier, wherein the carrier is water, an organic solvent, or a mixture thereof. In some embodiments, the clay treatment composition further comprises an organic solvent. In some other embodiments, the clay treatment composition further comprises an organic solvent and water.
- the organic solvent is an alcohol, a hydrocarbon, a ketone, an ether, an alkylene glycol, a glycol ether, an amide, a nitrile, a sulfoxide, an ester, or any combination thereof.
- the organic solvent is an alcohol, an alkylene glycol, an alkyleneglycol alkyl ether, or a combination thereof.
- the organic solvent is methanol, ethanol, propanol, isopropanol, butanol, isobutanol, monoethyleneglycol, ethyleneglycol monobutyl ether, or a combination thereof.
- the organic solvent is methanol, ethanol, propanol, isopropanol, butanol, 2-ethylhexanol, hexanol, octanol, decanol, 2-butoxyethanol, methylene glycol, ethylene glycol, 1,2-propylene glycol, 1,3-propylene glycol, diethyleneglycol monomethyl ether, diethylene glycol monoethyl ether, ethylene glycol monobutyl ether, ethylene glycol dibutyl ether, pentane, hexane, cyclohexane, methylcyclohexane, heptane, decane, dodecane, diesel, toluene, xylene, heavy aromatic naphtha, cyclohexanone, diisobutylketone, diethyl ether, propylene carbonate, N-methylpyrrolidinone, N,N-dimethylformamide, a
- the clay treatment composition further comprises one or more of corrosion inhibitors. In some embodiments, wherein the clay treatment composition further comprises one or more of corrosion inhibitors and a carrier. In some embodiments, the corrosion inhibitor is an imidazoline compound, a pyridinium compound, or a combination thereof.
- the clay treatment composition further comprises a biocide. In some embodiments, the clay treatment composition further comprises a biocide and carrier. In some other embodiments, the clay treatment composition further comprises a biocide, corrosion inhibitor, and carrier.
- the biocide is chlorine, hypochlorite, ClO 2 , bromine, ozone, hydrogen peroxide, peracetic acid, a peroxycarboxylic acid, peroxycarboxylic acid composition, peroxysulphate, glutaraldehyde, dibromonitrilopropionamide, isothiazolone, terbutylazine, polymeric biguanide, methylene bisthiocyanate, tetrakis hydroxymethyl phosphonium sulphate, and any combination thereof.
- the clay treatment composition further comprises an organic sulfur compound.
- the organic sulfur compound is a mercaptoalkyl alcohol, mercaptoacetic acid, thioglycolic acid, 3,3′-dithiodipropionic acid, sodium thiosulfate, thiourea, L-cysteine, tert-butyl mercaptan, sodium thiosulfate, ammonium thiosulfate, sodium thiocyanate, ammonium thiocyanate, sodium metabisulfite, or a combination thereof.
- the clay treatment composition further comprises an acid. In some embodiments, the clay treatment composition further comprises an inorganic acid, mineral acid, organic acid, or mixture thereof. In some embodiments, the clay treatment composition comprises from about 1 wt-% to about 20 wt-%, from about 1 wt-% to about 15 wt-%, from about 1 wt-% to about 10 wt-%, from about 1 wt-% to about 5 wt-% of the acid.
- the acid is hydrochloric acid, hydrofluoric acid, citric acid, formic acid, acetic acid, or mixture thereof.
- the clay treatment composition further comprises a hydrogen sulfide scavenger.
- the hydrogen sulfide scavenger is an oxidant, inorganic peroxide, sodium peroxide, chlorine dioxide; a C 1 -C 10 aldehyde, formaldehyde, glyoxal, glutaraldehyde, acrolein, or methacrolein, a triazine, monoethanolamine triazine, monomethylamine triazine, or a mixture thereof.
- the clay treatment composition further comprises a surfactant. In some embodiments, the clay treatment composition further comprises a surfactant, biocide, and carrier.
- the surfactant is a nonionic, semi-nonionic, cationic, anionic, amphoteric, zwitterionic, Gemini, di-cationic, di-anionic surfactant, or mixtures thereof.
- the clay treatment composition comprises a nonionic surfactant, particularly alcohol and alkylphenol ethoxylate, cationic, anionic, or amphoteric surfactant, a carrier, or a microemulsions thereof, as flowback aids.
- a nonionic surfactant particularly alcohol and alkylphenol ethoxylate, cationic, anionic, or amphoteric surfactant, a carrier, or a microemulsions thereof, as flowback aids.
- the clay treatment composition further comprises a viscosifier.
- suitable viscosifiers include, but are not limited to, polysaccharides, particularly sugar and sugar derivatized with glycidyl-bearing molecules, an acrylamide polymer, acrylamide copolymer (with acrylate, AMPS, DMAEA-MCQ, DMAEM-MCQ, MAPTAC, etc.), and viscoelastic surfactant (such as fatty diethanolamide).
- the clay treatment composition further comprises a viscosifier and crosslinkers.
- suitable crosslinkers include, but are not limited to, boron oxide, zirconium oxide, or salt thereof.
- the clay treatment composition further comprises a friction reducing agent.
- Suitable friction reducing agents include, but are not limited to, an acrylamide homopolymer, acrylamide (with acrylate, AMPS, DMAEA-MCQ, DMAEM-MCQ, MAPTAC, etc.) copolymer, and alkylene oxide polymer or copolymer.
- the surfactant is an alkyl phenol, fatty acid, or mixture thereof.
- the clay treatment composition further comprises an asphaltene inhibitor, a paraffin inhibitor, a scale inhibitor, a gas hydrate inhibitor, a pH modifier, or any combination thereof.
- the clay treatment composition further comprises a coagulant/flocculant agent, water clarifier, or mixture thereof. In some embodiments, the clay treatment composition further comprises an additional clay stabilizer, carrier, water clarifier, corrosion inhibitor, or mixture thereof.
- the water clarifier is diallyldimethyl ammonium chloride.
- the additional clay stabilizer choline chloride, tetramethylammonium chloride, or mixture thereof.
- the clay treatment composition further comprises a dispersant.
- the clay treatment composition further comprises antioxidant, polymer degradation prevention agent, permeability modifier, foaming agent, antifoaming agent, scavenger agent for CO 2 , and/or O 2 , gelling agent, lubricant, friction reducing agent, salt, alkalinity source, or mixture thereof.
- the clay treatment composition is a liquid, gel, or a mixture comprising liquid/gel and solid. In some embodiments, the clay treatment composition is a solid or liquid.
- the clay treatment composition or a use solution thereof has a pH of from about 2 to about 11.
- a use solution of a clay treatment composition or multiple charged cationic compounds as used herein refers to a diluted solution for the clay treatment composition or compounds by a diluent.
- a diluent as used herein refers to water, a stimulation fluid, any other fluid used for oil and gas operations, or one of the carriers or solvents defined herein.
- the clay treatment composition or the compounds can be diluted by a factor of 0.5, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11-1,000,000, or any value there between to generate a use solution and then provide the use solution to a subterranean formation.
- a clay treatment composition or multiple charged cationic compounds are applied, either the composition/compounds or use solution thereof is applied.
- the clay treatment composition further comprises a polymeric, polyquat, another cationic clay stabilizer, or mixture thereof.
- the clay treatment composition further comprises a low molecular weight inorganic salt, bis-quaternary ammonium compound, or mixture thereof. In some other embodiments, the clay treatment composition further comprises from about 0.1 wt-% to about 10 wt-% of KCl.
- the clay treatment composition is diluted with water to create a use solution of the clay treatment composition, the use solution is then provided into the subterranean formation.
- the water to dilute the clay treatment composition comprises fresh water, recycled water, salt water, surface water, produced water, or mixture thereof.
- the water to dilute the clay treatment composition is a fracturing fluid, acidizing fluid, drilling fluid, drill-in fluid, stimulation fluid, gravel pack fluid, completion fluid, cementing fluid, other oil gas operation fluid, or mixture thereof.
- the clay treatment composition or its use solution is injected into the fracturing fluid, acidizing fluid, drilling fluid, drill-in fluid, stimulation fluid, gravel pack fluid, completion fluid, cementing fluid, other oil gas operation fluid, or mixture thereof, before these fluids are injected into the subterranean formation for oil and gas production. In these situations, these fluids are considered to be the use solution of the clay treatment compositions.
- the concentration of the multiple charged cationic compound or the clay treatment composition is between about 1 ppm to about 1,000 ppm in these fluids.
- the subterranean formation comprises montmorillonite, smectite, saponite, nontronite, hectorite, and sauconite, kaolin, kaolinite, nacrite, dickite, hydrousmica, hydrobiotite, glauconite, illite and brammallite, chlorite, chamosite, vermiculite, attapulgite, palygoskite, sepiolite or mixture thereof.
- the clay treatment composition comprises from about 10 wt-% to about 80 wt-% of the compound or salt thereof. In some embodiments, the clay treatment composition comprises from about 30 wt-% to about 70 wt-%, from about 20 wt-% to about 40 wt-%, from about 25 wt-% to about 30 wt-%, from about 10 wt-% to about 70 wt-%, from about 30 wt-% to about 60 wt-%, from about 40 wt-% to about 50 wt-%, from about 10 wt-% to about 30 wt-%, from about 20 wt-% to about 40 wt-%, from 30 wt-% to about 50 wt-%, from about 40 wt-% to about 60 wt-%, from about 50 wt-% to 70 wt-%, about 10 wt-%, about 15 wt-%, about 20 wt-%, about 25 wt-%
- the clay treatment composition or multiple charged cationic compound(s) disclosed herein has a concentration of from about 1 ppm to about 1000 ppm in the stimulation fluid or any fluid for oil and gas operations. In the other embodiments, wherein the multiple charged cationic compound has a concentration of from about 0.5 ppm to about 100 ppm in the treated stimulation fluid after the clay treatment composition is provided into the stimulation fluid any fluid for oil and gas operations. In yet some other embodiments, the multiple charged cationic compounds disclosed herein has a concentration of from about 5 ppm to about 60 ppm in the stimulation fluid or the fluid for oil and gas operations after the clay treatment composition is applied to the stimulation fluid or the other fluid.
- the clay treatment composition is provided to the stimulation fluid or any other fluid for oil and gas operations independently, simultaneously, or sequentially with one or more additional clay treatment composition agents of the clay treatment composition.
- the clay treatment composition or multiple charged cationic compounds disclosed herein can reduces clay swelling, clay migration, and sludge formation as indicated by the capillary suction time (CST) measurement as described in the Examples section of this disclosure, when the subterranean formation has a multiple charged cationic compound or mixture thereof concentration of from about 1 ppm to about 1,000 ppm, from about 1 to about 900 ppm, from about 1 ppm to about 800 ppm, from about 1 ppm to about 700 ppm, from about 1 ppm to about 600 ppm, from about 1 ppm to about 500 ppm, from about 1 ppm to about 400 ppm, from about 1 ppm to about 300 ppm, from about 1 ppm to about 250 ppm, from about 1 ppm to about 200 ppm, from about 1 ppm to about 150 ppm, from about 1 ppm to about 100 ppm, from about 1 ppm to about 50 ppm, about 950 ppm,
- clay treatment compositions are provided to stabilize swellable clays and/or reduce formation of sludge in a subterranean formation.
- the methods of stabilizing swellable clays and/or reducing formation of sludge in a subterranean formation comprise: providing a clay treatment composition into a subterranean formation, wherein the clay treatment composition comprises a compound or its salt derived from an aza-Michael Addition Reaction between a polyamine and an ⁇ , ⁇ -unsaturated carbonyl compound according to the following formula
- X is NH or O
- R 2 is H, CH 3 , or an unsubstituted, linear or branched C 2 -C 10 alkyl, alkenyl, or alkynyl group
- R 3 is absent or an unsubstituted, linear C 1 -C 30 alkylene group
- Y is —NR 4 R 5 R 6 (+) or a salt thereof
- R 4 , R 5 , and R 6 are independently a C 1 -C 10 alkyl group, wherein the compound is a multiple charged cationic compound having 2 or more positive charges
- the clay treatment composition stabilizes swellable clays and reduces formation of sludge, or both.
- the polyamine is a linear, branched, or dendrimer polyamine with a general formula of NH 2 —[R 10′ ] n —NH 2 , (RNH) n —RNH 2 , H 2 N—(RNH) n —RNH 2 , or H 2 N—(RN(R′)) n —RNH 2 , wherein R 10′ is a linear or branched, unsubstituted or substituted C 2 -C 10 alkylene group, or combination thereof; R is —CH 2 —, —CH 2 CH 2 —, —CH 2 CH 2 CH 2 —, —CH(CH 3 )CH 2 —, a linear or branched, unsubstituted or substituted C 4 -C 10 alkylene group, or combination thereof; R′ is —CH 2 —, —CH 2 CH 2 —, —CH 2 CH 2 CH 2 —, —CH(CH 3 )CH 2 —,
- the polyamine is an unmodified polyamine, a modified polyamine, an ethoxylated polyamine, propylated polyamine, polyamine with polyquat, polyamine with polyglycerol, or combination thereof.
- the polyamine is a linear, branched, or dendrimer polyethyleneimine.
- the polyamine comprises only primary and secondary amine groups; the polyamine comprises only primary, secondary, and tertiary amine groups; or the polyamine comprises only primary and tertiary amine groups.
- the polyamine is one or more of polyamines under the tradename JEFFAMINE® by Huntsman.
- the polyamine is a polyamine with a general formula of NH 2 —[R 10′ ] n —NH 2 , wherein R 10′ is a linear or branched, unsubstituted or substituted C 2 -C 10 alkylene group, or combination thereof
- the polyamine is a polyamine with a general formula of (RNH) n —RNH 2 or H 2 N—(RNH) n —RNH 2 , wherein R is —CH 2 —, —CH 2 CH 2 —, —CH 2 CH 2 CH 2 —, —CH(CH 3 )CH 2 —, a linear or branched, unsubstituted or substituted C 4 -C 10 alkylene group, or combination thereof
- the polyamine is a polyamine with a general formula of H 2 N—(RN(R′)) n —R
- the ⁇ , ⁇ -unsaturated carbonyl compound has X is NH or O; R 2 is H or CH 3 ; Y is —NR 4 R 5 R 6(+) , and R 4 , R 5 , and R 6 are independently CH 3 ; Y is —NR 4 R 5 R 6(+) , R 4 and R 5 are independently CH 3 , and R 6 is a C 2 -C 12 aromatic alkyl; Y is —NR 4 R 5 R 6(+) , R 4 and R 5 are independently CH 3 , and R 6 is —CH 2 —C 6 H 6 ; the counter ion for Y is chloride, bromide, fluoride, iodide, acetate, aluminate, cyanate, cyanide, dihydrogen phosphate, dihydrogen phosphite, formate, hydrogen carbonate, hydrogen oxalate, hydrogen sulfate, hydroxide, nitrate, nitrite,
- the ⁇ , ⁇ -unsaturated carbonyl compound is (3-Acrylamidopropyl)trimethylammonium chloride (APTAC) or [3-(Methacryloylamino)propyl]trimethylammonium chloride (MAPTAC); and/or the ⁇ , ⁇ -unsaturated carbonyl compound is (3-Acrylamidopropyl)trimethylammonium chloride (APTAC), [3-(methacryloylamino)propyl]trimethylammonium chloride (MAPTAC), 2-(acryloyloxy)-N,N,N-trimethylethanaminium chloride (DMAEA-MCQ), N,N-dimethylaminoethyl acrylate benzyl chloride quaternary salt (DMAEA-BCQ), or 2-(methacryloyloxy)-N,N,N-trimethylethan-1-aminium methyl sulfate (DMAEA-MS
- the stabilizing swellable clays and/or reducing formation of sludge are in a subterranean formation
- the compound is a single molecule; the compound is a mixture of at least two multiple charged cationic compounds; or the compound is a mixture of at least two multiple charged cationic compounds derived from the same polyamine and the ⁇ , ⁇ -unsaturated carbonyl compound.
- the product is a mixture of at least two multiple charged cationic compounds derived from different polyamines and the same ⁇ , ⁇ -unsaturated carbonyl compound; or the product is a mixture of at least two multiple charged cationic compounds derived from different polyamines and different ⁇ , ⁇ -unsaturated carbonyl compounds.
- the compound has an average molecular weight of from about 100 to about 2,000,000 Da; or from about 100 to about 5,500 Da.
- the compound has at least 10, 15, 20, or 30 positive charges; or the compound has at least 4, 5, 6, 7, or 8 positive charges.
- the compound is one or more of
- the compound is derived from a polyethyleneimine and (3-Acrylamidopropyl)trimethylammonium chloride (APTAC), wherein the polyethyleneimine is a linear PEI and has an average molecular weight (M w ) of about 5,000.
- the compound is derived from a polyethyleneimine and (3-Acrylamidopropyl)trimethylammonium chloride (APTAC), wherein the polyethyleneimine is a linear PEI and has an average molecular weight (M w ) of about 750,000.
- the compound is derived from a polyethyleneimine and (3-Acrylamidopropyl)trimethylammonium chloride (APTAC), wherein the polyethyleneimine is a branched PEI and has an average molecular weight (M w ) of about 25,000.
- the compound is soluble in water.
- the clay treatment composition further comprises a carrier comprising one or more of water, an alcohol, an alkylene glycol, an alkyleneglycol alkyl ether, or a combination thereof.
- the carrier is water, methanol, ethanol, propanol, isopropanol, butanol, isobutanol, monoethyleneglycol, ethyleneglycol monobutyl ether, or a combination thereof.
- the clay treatment composition further comprises one or more of additional functional ingredients, wherein the additional functional ingredient is a flowback aid, friction reducing agent, crosslinker, additional clay stabilizer, viscosifier, reverse emulsion breaker, coagulant/flocculant agent, biocide, corrosion inhibitor, antioxidant, polymer degradation prevention agent, permeability modifier, foaming agent, antifoaming agent, emulsifying agent, fracturing proppant, glass particulate, sand, fracture proppant/sand control agent, scavenger for H 2 S, CO 2 , and/or O 2 , gelling agent, lubricant, salt thereof, or mixture thereof.
- additional functional ingredient is a flowback aid, friction reducing agent, crosslinker, additional clay stabilizer, viscosifier, reverse emulsion breaker, coagulant/flocculant agent, biocide, corrosion inhibitor, antioxidant, polymer degradation prevention agent, permeability modifier, foaming agent, antifoaming agent, emulsifying agent,
- the clay treatment composition further comprises an acid from about 1 wt-% to about 20 wt-%, wherein the acid is hydrochloric acid, hydrofluoric acid, citric acid, formic acid, acetic acid, or mixture thereof.
- the clay treatment composition further comprises an alkyl phenol, fatty acid, or mixture thereof.
- the clay treatment composition further comprises a surfactant, wherein the surfactant is a nonionic, semi-nonionic, cationic, anionic, amphoteric, zwitterionic, Gemini, di-cationic, di-anionic surfactant, or mixtures thereof.
- the clay treatment composition further comprises a polymer, polymeric polyquat, or another cationic clay stabilizer.
- the clay treatment composition further comprises a bis-quaternary ammonium compound or salt thereof.
- the clay treatment composition contacts the subterranean formation independently, simultaneously, or sequentially with the additional functional ingredient.
- the additional functional ingredient contacts the subterranean formation with the multiple charged compound in the treatment composition or through an additional composition.
- the treatment composition further comprises water, including fresh water, recycled water, salt water, surface water, produced water, or mixture thereof.
- the clay treatment composition is a fracturing fluid, acidizing fluid, drilling fluid, drill-in fluid, stimulation fluid, gravel pack fluid, completion fluid, cementing fluid, or other oil and gas operation fluid.
- the clay treatment composition has a pH of from about 2 to about 11.
- the swellable clays comprises montmorillonite, smectite, saponite, nontronite, hectorite, and sauconite, kaolin, kaolinite, nacrite, dickite, hydrousmica, hydrobiotite, glauconite, illite and brammallite, chlorite, chamosite, vermiculite, attapulgite, palygoskite, sepiolite or mixture thereof.
- the clay treatment composition is a liquid, gel, or a mixture comprising liquid/gel and solid. In further embodiments, the clay treatment composition comprises from about 40 wt-% to about 60 wt-% of the multiple charged cationic compound or its salt.
- Embodiments of the present disclosure are further defined in the following non-limiting Examples. These Examples, while indicating certain embodiments of the disclosure, are given by way of illustration only. From the above discussion and these Examples, one skilled in the art can ascertain the essential characteristics of this disclosure, and without departing from the spirit and scope thereof, can make various changes and modifications of the embodiments of the disclosure to adapt it to various usages and conditions. Thus, various modifications of the embodiments of the disclosure, in addition to those shown and described herein, will be apparent to those skilled in the art from the foregoing description. Such modifications are also intended to fall within the scope of the appended claims.
- Ethyleneamine E-100 from Huntsman was used for this reaction.
- E-100 is a mixture of tetraethylenepentamine (TEPA), pentaethylenehexamine (PEHA), hexaethyleneheptamine (HEHA), and other higher molecular weight amines.
- TEPA tetraethylenepentamine
- PEHA pentaethylenehexamine
- HEHA hexaethyleneheptamine
- E-100 is a complex mixture of various linear, cyclic, and branched amines with a number-average molecular weight (Mn) of 250-300 g/mole.
- BASF Lupasol G20 (50% aqueous solution of polyethylenimine with a weight-average molecular weight (M w ) around 1,300 g/mole) was used for this reaction.
- BASF Lupasol G100 50% aqueous solution of a polyethyleneimine with a weight-average molecular weight (M w ) around 5000 g/mole was used for this reaction.
- CST Capillary Suction Timer
- Capillary suction time (CST) measurement is used as a rapid screening test for clay formation-stabilizing chemical additives.
- CST is a static filtration test that is used to measure the permeability of a filter cake that forms at the interface of a slurry and chromatographic paper.
- the fine particulate material adsorbs and retains water more effectively, forms tighter filter cakes, and more easily moves into the pore space of the paper, thereby hindering the flow of water from the slurry into the paper.
- Coarse and coagulated particulates by contrast, will form a looser filter cake with larger intra-particle spacing, and will not be able to fit into the paper pores and clog them, thereby yielding faster flow through the paper.
- the flow of water through the paper is measured using a set of three electrodes that are placed in contact with the paper at fixed distances from the slurry-containing metal funnel. Two of the electrodes are closer.
- the flow of water through the paper is measured using a set of three electrodes that are placed in contact with the paper at fixed distances from the slurry-containing metal funnel. Two of the electrodes are closer and equidistant from the funnel, the third electrode is placed at a greater distance from the funnel and equidistant from the first two electrodes.
- the measured time reflects the properties of both the solid and liquid in the slurry, depending on their cumulative impact on filter cake formation and flow through microscale pores. Individual tests, not including preparation and cleanup time, range from tens of seconds to minutes. When measuring with water and variable formation materials, shorter times typically reflect reduced need for clay or formation stabilization, and longer measured times reflect increased need for stabilization. If the formation material is held constant and the fluid is varied, lower times reflect improved clay and formation stabilization properties.
- Mineral class Common native minerals Surrogate mineral Tectosilicates Quartz Silica Feldspars Other Pyrite, marcasite Silica Apatite, gypsum, anhydrite Calcite Carbonates Calcite, dolomite, siderite, Calcite ankerite Phyllosilicates [Non-swelling] Illite, mica, Kaolinite (clays) kaolinite, chlorite [Swelling] Smectite Bentonite
- the CST test therefore begins with the identification of the target formation mineral composition.
- a target formation is analyzed, typically based on a sample of drill cuttings that have been cleaned and dried.
- the composition is identified using quantitative X-ray diffraction (XRD) methods that include phyllosilicate (clay) speciation, yielding mass fractions of the various minerals that comprise the sample.
- XRD quantitative X-ray diffraction
- the determined composition is then used to create a “surrogate” that matches the original target composition as much as possible, using the classification scheme shown in Table 3, by weighing out the limbate amount of each surrogate mineral and mixing them well in a container.
- the clay treatment compositions can be prepared by standard procedures. For each CST measurement, 1 gram of the surrogate is treated with 4.0 mL of a solution of the tested composition. Usually, after mixing the solution with the surrogate mineral in a container for an hour, during which the mixture should be vortexed a few times, the mixture is then vortexed again and then immediately poured into the funnel of the CST measuring device for its CST measurement. For each solution, three independent measurements were taken.
- CST Capillary Suction Time
- CST Capillary Sample Description suction time
- Sample ID (concentration) (CST) Stdev Compound 1 EDA - APTAC (0.1%) 50 Compound 2 DETA - APTAC (0.1%) 48 Compound 3 PEHA - APTAC (0.1%) 41 Compound 4 Tris(2- 47 aminomethyl)amine- APTAC (0.1%) Compound 5 E-100 - APTAC (0.1%) 50 Compound 6 G-20 - APTAC (0.1%) 46 Compound 7 G-100 - APTAC (0.1%) 69 KCl (2%) 67 6 Choline chloride (0.1%) 76 2 Fresh water 90 3
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- Materials Engineering (AREA)
- Mining & Mineral Resources (AREA)
- Geology (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Ceramic Engineering (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Dispersion Chemistry (AREA)
- Inorganic Chemistry (AREA)
- Fluid Mechanics (AREA)
- Environmental & Geological Engineering (AREA)
- Geochemistry & Mineralogy (AREA)
- Physics & Mathematics (AREA)
- Structural Engineering (AREA)
- Macromolecular Compounds Obtained By Forming Nitrogen-Containing Linkages In General (AREA)
Abstract
Description
and
one or more additional clay treatment composition agents,
wherein X is NH or O; R2 is H, CH3, or an unsubstituted, linear or branched C2-C10 alkyl, alkenyl, or alkynyl group; R3 is absent or an unsubstituted, linear C1-C30 alkylene group; Y is —NR4R5R6 (+), or a salt thereof; and R4, R5, and R6 are independently a C1-C10 alkyl group; wherein the compound is a multiple charged cationic compound having 2, 3, or more positive charges. In some embodiments, a use solution of the clay treatment composition reduces clay swelling, clay migration, and sludge formation.
wherein X is NH or O; R2 is H, CH3, or an unsubstituted, linear or branched C2-C10 alkyl, alkenyl, or alkynyl group; R3 is absent or an unsubstituted, linear C1-C30 alkylene group; Y is —NR4R5R6 (+), or a salt thereof; and R4, R5, and R6 are independently a C1-C10 alkyl group; and wherein the compound is a multiple charged cationic compound having 3 or more positive charges. In some embodiments, a use solution of the clay treatment composition reduces clay swelling, clay migration, and sludge formation.
wherein R10a is a C1-C20 alkyl or a C1-C20 alkoxyalkyl group; R11a and R14a are independently hydrogen, C1-C6 alkyl, C1-C6 hydroxyalkyl, or C1-C6 arylalkyl; R12a and R13a are independently hydrogen or a C1-C6 alkyl group; and X− is a halide (such as chloride, bromide, or iodide), carbonate, sulfonate, phosphate, or the anion of an organic carboxylic acid (such as acetate). Preferably, the imidazolinium compound includes 1-benzyl-1-(2-hydroxyethyl)-2-tall-oil-2-imidazolinium chloride.
wherein R1a and R2a are each independently unsubstituted branched, chain or ring alkyl or alkenyl having from 1 to about 29 carbon atoms; partially or fully oxygenized, sulfurized, and/or phosphorylized branched, chain, or ring alkyl or alkenyl having from 1 to about 29 carbon atoms; or a combination thereof; R3a and R4a are each independently unsubstituted branched, chain or ring alkylene or alkenylene having from 1 to about 29 carbon atoms; partially or fully oxygenized, sulfurized, and/or phosphorylized branched, chain, or ring alkylene or alkenylene having from 1 to about 29 carbon atoms; or a combination thereof; L1 and L2 are each independently absent, H, —COOH, —SO3H, —PO3H, —COOR5a, —CONH2, —CONHR5a, or —CON(R5a)2; R5a is each independently a branched or unbranched alkyl, aryl, alkylaryl, alkylheteroaryl, cycloalkyl, or heteroaryl group having from 1 to about 10 carbon atoms; n is 0 or 1, and when n is 0, L2 is absent or H; x is from 1 to about 10; and y is from 1 to about 5. Preferably, R1a and R2a are each independently C6-C22 alkyl, C8-C20 alkyl, C12-C18 alkyl, C16-C18 alkyl, or a combination thereof; R1a and R4a are C1-C10 alkylene, C2-C8 alkylene, C2-C6 alkylene, or C2-C3 alkylene; n is 0 or 1; x is 2; y is 1; R3 and R4 are —C2H2—; L1 is —COOH, —SO3H, or —PO3H; and L2 is absent, H, —COOH, —SO3H, or —PO3H. For example, R1a and R2a can be derived from a mixture of tall oil fatty acids and are predominantly a mixture of C17H33 and C17H31 or can be C16-C18 alkyl; R1a and R4a can be C2-C3 alkylene such as —C2H2—; n is 1 and L2 is —COOH or n is 0 and L2 is absent or H; x is 2; y is 1; R1a and R4a are —C2H2—; and L1 is —COOH.
wherein R1a, R2a, and R3a are independently C1 to C20 alkyl, R4a is methyl or benzyl, and X− is a halide or methosulfate.
wherein R9a is an alkyl group, an aryl group, or an arylalkyl group, wherein said alkyl groups have from 1 to about 18 carbon atoms and X− is a halide such as chloride, bromide, or iodide. Among these compounds are alkyl pyridinium salts and alkyl pyridinium benzyl quats. Exemplary compounds include methyl pyridinium chloride, ethyl pyridinium chloride, propyl pyridinium chloride, butyl pyridinium chloride, octyl pyridinium chloride, decyl pyridinium chloride, lauryl pyridinium chloride, cetyl pyridinium chloride, benzyl pyridinium chloride and an alkyl benzyl pyridinium chloride, preferably wherein the alkyl is a C1-C6 hydrocarbyl group. Preferably, the pyridinium compound includes benzyl pyridinium chloride.
| TABLE 1 |
| A. Exemplary Clay Treatment Compositions |
| Composition ID |
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | |
| Multiple | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 |
| Charged | ||||||||
| Cationic | ||||||||
| Compound(s) | ||||||||
| Friction | 0.025-0.4 | 0 | 0.025-0.4 | 0.025-0.4 | 0.025-0.4 | 0.025-0.4 | 0 | 0 |
| reducer | ||||||||
| Flowback aid | 0.025-0.2 | 0.025-0.2 | 0 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0 | 0.025-0.2 |
| Viscosifier | 0.025-1.0 | 0.025-1.0 | 0.025-1.0 | 0.025-1.0 | 0.025-1.0 | 0.025-1.0 | 0.025-1.0 | 0.025-1.0 |
| Crosslinker | 0.010-0.3 | 0.010-0.3 | 0.010-0.3 | 0 | 0.010-0.3 | 0.010-0.3 | 0.010-0.3 | 0 |
| Scale | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 |
| inhibitor | ||||||||
| Biocide | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0 | 0.025-0.2 | 0.025-0.2 |
| Water | 97.5- | 97.9- | 97.7- | 97.8- | 97.7- | 97.7- | 98.1- | 98.2- |
| 99.84 | 99.865 | 99.865 | 99.85 | 99.865 | 99.865 | 99.89 | 99.875 | |
| B. Exemplary Clay Treatment Compositions |
| Composition ID |
| 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | |
| Multiple | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 |
| Charged | |||||||||
| Cationic | |||||||||
| Compound(s) | |||||||||
| Friction | 0 | 0 | 0.025-0.4 | 0.025-0.4 | 0.025-0.4 | 0.025-0.4 | 0.025-0.4 | 0.025-0.4 | 0.025-0.4 |
| reducing agent | |||||||||
| Flowback aid | 0.025-0.2 | 0.025-0.2 | 0 | 0 | 0 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 |
| Viscosifier | 0.025-1.0 | 0.025-1.0 | 0.025-1.0 | 0.025-1.0 | 0.025-1.0 | 0 | 0.025-1.0 | 0.025-1.0 | 0.025-1.0 |
| Crosslinker | 0.010-0.3 | 0.010-0.3 | 0 | 0.010-0.3 | 0.010-0.3 | 0 | 0 | 0 | 0.010-0.3 |
| Scale inhibitor | 0 | 0.025-0.2 | 0.025-0.2 | 0 | 0.025-0.2 | 0.025-0.2 | 0 | 0.025-0.2 | 0 |
| Biocide | 0.025-0.2 | 0 | 0.025-0.2 | 0.025-0.2 | 0 | 0.025-0.2 | 0.025-0.2 | 0 | 0 |
| Water | 98.1- | 98.1- | 98- | 97.9- | 97.9- | 98.8- | 98- | 98- | 97.9- |
| 99.89 | 99.89 | 99.875 | 99.89 | 99.89 | 99.875 | 99.875 | 99.875 | 99.89 | |
| C. Exemplary Clay Treatment Compositions |
| Composition ID |
| 17 | 18 | 19 | 20 | 21 | 22 | 23 | 24 | |
| Multiple | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 |
| Charged | ||||||||
| Cationic | ||||||||
| Compound(s) | ||||||||
| Friction | 0 | 0 | 0 | 0.025-0.4 | 0.025-0.4 | 0.025-0.4 | 0.025-0.4 | 0.025-0.4 |
| reducer | ||||||||
| Flowback aid | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0.025-0.2 |
| Viscosifier | 0.025-1.0 | 0.025-1.0 | 0.025-1.0 | 0 | 0.025-1.0 | 0.025-1.0 | 0.025-1.0 | 0 |
| Crosslinker | 0 | 0.010-0.3 | 0.010-0.3 | 0 | 0 | 0 | 0.010-0.3 | 0 |
| Scale | 0.025-0.2 | 0 | 0.025-0.2 | 0.025-0.2 | 0 | 0.025-0.2 | 0 | 0 |
| inhibitor | ||||||||
| Biocide | 0.025-0.2 | 0.025-0.2 | 0 | 0.025-0.2 | 0.025-0.2 | 0 | 0 | 0.025-0.2 |
| Water | 98.4- | 98.3- | 98.3- | 99- | 98.2- | 98.2- | 98.1- | 99- |
| 99.9 | 99.915 | 99.915 | 99.9 | 99.9 | 99.9 | 99.915 | 99.9 | |
| D. Exemplary Clay Treatment Compositions |
| Composition ID |
| 25 | 26 | 27 | 28 | 29 | 30 | 31 | 32 | |
| Multiple | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 |
| Charged | ||||||||
| Cationic | ||||||||
| Compound(s) | ||||||||
| Friction | 0.025-0.4 | 0.025-0.4 | 0 | 0 | 0 | 0.025-0.4 | 0.025-0.4 | 0.025-0.4 |
| reducer | ||||||||
| Flowback aid | 0.025-0.2 | 0.025-0.2 | 0 | 0 | 0 | 0 | 0 | 0 |
| Viscosifier | 0 | 0.025-1.0 | 0.025-1.0 | 0.025-1.0 | 0.025-1.0 | 0 | 0 | 0.025-1.0 |
| Crosslinker | 0 | 0 | 0 | 0 | 0.010-0.3 | 0 | 0 | 0 |
| Scale | 0.025-0.2 | 0 | 0 | 0.025-0.2 | 0 | 0 | 0.025-0.2 | 0 |
| inhibitor | ||||||||
| Biocide | 0 | 0 | 0.025-0.2 | 0 | 0 | 0.025-0.2 | 0 | 0 |
| Water | 99- | 98.2- | 98.6- | 98.6- | 98.5- | 99.2- | 99.2- | 98.4- |
| 99.9 | 99.9 | 99.925 | 99.925 | 99.94 | 99.925 | 99.925 | 99.925 | |
| E. Exemplary Clay Treatment Compositions |
| Composition ID |
| 33 | 34 | 35 | |
| Multiple | 0.025-0.2 | 0.025-0.2 | 0.025-0.2 |
| Charged | |||
| Cationic | |||
| Compound(s) | |||
| Friction | 0.025-0.4 | 0.025-0.4 | 0 |
| reducer | |||
| Flowback aid | 0.025-0.2 | 0 | 0 |
| Viscosifier | 0 | 0 | 0.025-1.0 |
| Crosslinker | 0 | 0 | 0 |
| Scale inhibitor | 0 | 0 | 0 |
| Biocide | 0 | 0 | 0 |
| Water | 99.2-99.925 | 99.4-99.95 | 98.8-99.95 |
and
one or more additional clay treatment composition agents, wherein X is NH or O; R2 is H, CH3, or an unsubstituted, linear or branched C2-C10 alkyl, alkenyl, or alkynyl group; R3 is absent or an unsubstituted, linear C1-C30 alkylene group; Y is —NR4R5R6 (+), or a salt thereof; and R4, R5, and R6 are independently a C1-C10 alkyl group; wherein the compound is a multiple charged cationic compound having 2, 3, or more positive charges. In some embodiments, a use solution of the clay treatment composition reduces clay swelling, clay migration, and sludge formation in the subterranean formation.
wherein X is NH or O; R2 is H, CH3, or an unsubstituted, linear or branched C2-C10 alkyl, alkenyl, or alkynyl group; R3 is absent or an unsubstituted, linear C1-C30 alkylene group; Y is —NR4R5R6 (+), or a salt thereof; and R4, R5, and R6 are independently a C1-C10 alkyl group; wherein the compound is a multiple charged cationic compound having 2, 3, or more positive charges. In some embodiments, a use solution of the clay treatment composition reduces clay swelling, clay migration, and sludge formation in the subterranean formation.
or a combination thereof, each of the compounds contain at least 2 non-proton and cationic A groups, at least 3 non-proton and cationic A groups, at least 4 non-proton and cationic A groups, at least 5 non-proton and cationic A groups, or more than 6 non-proton and cationic
and the rest of primary NH2 protons remains. In some embodiments, some of primary NH2 and secondary NH proton are replaced by
and
one or more clay treatment composition agents, wherein: X is NH or O; R2 is H, CH3, or an unsubstituted, linear or branched C2-C10 alkyl, alkenyl, or alkynyl group; R3 is absent or an unsubstituted, linear C1-C30 alkylene group; Y is —NR4R5R6 (+) or a salt thereof; and R4, R5, and R6 are independently a C1-C10 alkyl group, wherein the compound is a multiple charged cationic compound having 2 or more positive charges; and wherein the clay treatment composition stabilizes swellable clays and reduces formation of sludge, or both. In some embodiments, the polyamine is a linear, branched, or dendrimer polyamine with a general formula of NH2—[R10′]n—NH2, (RNH)n—RNH2, H2N—(RNH)n—RNH2, or H2N—(RN(R′))n—RNH2, wherein R10′ is a linear or branched, unsubstituted or substituted C2-C10 alkylene group, or combination thereof; R is —CH2—, —CH2CH2—, —CH2CH2CH2—, —CH(CH3)CH2—, a linear or branched, unsubstituted or substituted C4-C10 alkylene group, or combination thereof; R′ is —CH2—, —CH2CH2—, —CH2CH2CH2—, —CH(CH3)CH2—, a linear or branched, unsubstituted or substituted C4-C10 alkyl group, RNH2, RNHRNH2, or RN(RNH2)2; and n can be from 2 to 1,000,000. In some embodiments, the polyamine is an unmodified polyamine, a modified polyamine, an ethoxylated polyamine, propylated polyamine, polyamine with polyquat, polyamine with polyglycerol, or combination thereof. In some embodiments, the polyamine is a linear, branched, or dendrimer polyethyleneimine. In some embodiments, the polyamine comprises only primary and secondary amine groups; the polyamine comprises only primary, secondary, and tertiary amine groups; or the polyamine comprises only primary and tertiary amine groups.
| TABLE 2 |
| Mineral classes of frequently-encountered native minerals in oil |
| and gas reservoirs, common minerals within each class, and the |
| surrogate minerals selected for representation of each native mineral. |
| Mineral class | Common native minerals | Surrogate mineral |
| Tectosilicates | Quartz | Silica |
| Feldspars | ||
| Other | Pyrite, marcasite | Silica |
| Apatite, gypsum, anhydrite | Calcite | |
| Carbonates | Calcite, dolomite, siderite, | Calcite |
| ankerite | ||
| Phyllosilicates | [Non-swelling] Illite, mica, | Kaolinite |
| (clays) | kaolinite, chlorite | |
| [Swelling] Smectite | Bentonite | |
| TABLE 3 |
| List of the exemplary multiple charged cationic compounds tested for clay treatment. |
| Sample | Structure |
| Compound | |
| 1 |
|
| Compound 2 |
|
| Compound 3 |
|
| Compound 4 |
|
| Compound 5 |
|
| Compound | Polyethylenimine; MW 1300 (BASF- |
| 6 | Lupasol G20)-APTAC Adduct |
| Compound | Polyethylenimine; MW 5000 (BASF- |
| 7 | Lupasol G100)-APTAC Adduct |
| TABLE 4 |
| Capillary Suction Time (CST) for the exemplary |
| multiple charged compounds |
| Capillary | ||||
| Sample Description | suction time | |||
| Sample ID | (concentration) | (CST) (s) | | |
| Compound | ||||
| 1 | EDA - APTAC (0.1%) | 50 | ||
| Compound 2 | DETA - APTAC (0.1%) | 48 | ||
| Compound 3 | PEHA - APTAC (0.1%) | 41 | ||
| Compound 4 | Tris(2- | 47 | ||
| aminomethyl)amine- | ||||
| APTAC (0.1%) | ||||
| Compound 5 | E-100 - APTAC (0.1%) | 50 | ||
| Compound 6 | G-20 - APTAC (0.1%) | 46 | ||
| Compound 7 | G-100 - APTAC (0.1%) | 69 | ||
| KCl (2%) | 67 | 6 | ||
| Choline chloride (0.1%) | 76 | 2 | ||
| Fresh water | 90 | 3 | ||
Claims (24)
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US16/554,805 US11084974B2 (en) | 2018-08-29 | 2019-08-29 | Use of multiple charged cationic compounds derived from polyamines for clay stabilization in oil and gas operations |
| US17/305,069 US11702586B2 (en) | 2018-08-29 | 2021-06-30 | Use of multiple charged cationic compounds derived from polyamines for clay stabilization in oil and gas operations |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201862724365P | 2018-08-29 | 2018-08-29 | |
| US16/554,805 US11084974B2 (en) | 2018-08-29 | 2019-08-29 | Use of multiple charged cationic compounds derived from polyamines for clay stabilization in oil and gas operations |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US17/305,069 Division US11702586B2 (en) | 2018-08-29 | 2021-06-30 | Use of multiple charged cationic compounds derived from polyamines for clay stabilization in oil and gas operations |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| US20200071602A1 US20200071602A1 (en) | 2020-03-05 |
| US11084974B2 true US11084974B2 (en) | 2021-08-10 |
Family
ID=69642269
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US16/554,805 Active US11084974B2 (en) | 2018-08-29 | 2019-08-29 | Use of multiple charged cationic compounds derived from polyamines for clay stabilization in oil and gas operations |
| US17/305,069 Active US11702586B2 (en) | 2018-08-29 | 2021-06-30 | Use of multiple charged cationic compounds derived from polyamines for clay stabilization in oil and gas operations |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US17/305,069 Active US11702586B2 (en) | 2018-08-29 | 2021-06-30 | Use of multiple charged cationic compounds derived from polyamines for clay stabilization in oil and gas operations |
Country Status (1)
| Country | Link |
|---|---|
| US (2) | US11084974B2 (en) |
Cited By (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20220002175A1 (en) * | 2019-01-29 | 2022-01-06 | Ecolab Usa Inc. | Use of cationic sugar-based compounds as corrosion inhibitors in a water system |
| US20220002174A1 (en) * | 2019-01-29 | 2022-01-06 | Ecolab Usa Inc. | Use of cationic sugar-based compounds for microbial fouling control in a water system |
| US11565958B2 (en) | 2017-08-30 | 2023-01-31 | Ecolab Usa Inc. | Use of di-ionic compounds as corrosion inhibitors in a water system |
| US11639553B2 (en) | 2019-04-16 | 2023-05-02 | Ecolab Usa Inc. | Compositions comprising multiple charged cationic compounds derived from polyamines for corrosion inhibition in a water system |
| US20230192929A1 (en) * | 2021-12-22 | 2023-06-22 | Ecolab Usa Inc. | Compositions comprising multiple charged cationic compounds for soil release |
| US11685709B2 (en) * | 2018-08-29 | 2023-06-27 | Ecolab Usa Inc. | Multiple charged ionic compounds derived from polyamines and compositions thereof and use thereof as reverse emulsion breakers in oil and gas operations |
| US11702586B2 (en) | 2018-08-29 | 2023-07-18 | Championx Usa Inc. | Use of multiple charged cationic compounds derived from polyamines for clay stabilization in oil and gas operations |
| US11926543B2 (en) | 2018-08-29 | 2024-03-12 | Ecolab Usa Inc. | Use of multiple charged ionic compounds derived from polyamines for waste water clarification |
| US11981857B1 (en) * | 2023-05-11 | 2024-05-14 | King Fahd University Of Petroleum And Minerals | Drilling fluid composition and method of making the composition |
| WO2024124129A1 (en) * | 2022-12-09 | 2024-06-13 | Championx Llc | Corrosion inhibitor having silica nanoparticles |
| US12082580B2 (en) | 2018-08-29 | 2024-09-10 | Ecolab Usa Inc. | Use of multiple charged cationic compounds derived from primary amines or polyamines for microbial fouling control in a water system |
| US12180424B1 (en) | 2023-08-22 | 2024-12-31 | Halliburton Energy Services, Inc. | Methods and compositions used in controlling formation sand production, proppant flowback, and fines migration |
| US12202784B2 (en) | 2018-08-29 | 2025-01-21 | Ecolab Usa Inc. | Multiple charged ionic compounds derived from polyamines and compositions thereof and methods of preparation thereof |
Families Citing this family (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA3132997A1 (en) * | 2019-03-20 | 2020-09-24 | Kao Specialties Americas Llc | Stable and flowable water-free based slurry compositions capable of suspending up to 75 wt.% of organic and inorganic materials for deliveryin a concentrated liquid form useful in a wide range of oilfield applications containing such material |
| CN112080259B (en) * | 2020-04-14 | 2022-04-01 | 西南石油大学 | Shale intercalation inhibitor prepared from environment-friendly hyperbranched polyquaternary ammonium salt amino acid |
| CN111978939B (en) * | 2020-09-16 | 2022-04-19 | 西南石油大学 | A polyionic liquid as a surface hydration inhibitor and a water-based drilling fluid system |
| US11492536B2 (en) * | 2021-02-16 | 2022-11-08 | Saudi Arabian Oil Company | Cement slurries and methods for cementing a casing in a wellbore |
| US11566157B2 (en) | 2021-02-16 | 2023-01-31 | Saudi Arabian Oil Company | Water-based drilling fluid compositions and methods for drilling subterranean wells |
| US11608467B2 (en) * | 2021-02-16 | 2023-03-21 | Saudi Arabian Oil Company | Hydraulic fracturing fluids with an aqueous base fluid and clay stabilizer and methods for hydraulic fracturing using the same |
| GB2606338B (en) * | 2021-04-09 | 2025-08-06 | Championx Llc | Crude oil production using a combination of emulsion viscosity reducer and scale inhibitor |
| US11535787B2 (en) | 2021-05-12 | 2022-12-27 | Saudi Arabian Oil Company | Spacer fluids and methods for cementing a casing in a wellbore |
| CN113403046B (en) * | 2021-05-12 | 2023-01-06 | 中国石油大学(华东) | Shrinkage-expansion agent for preventing borehole wall instability in oil production and drilling processes of high-argillaceous oil reservoirs and preparation method of shrinkage-expansion agent |
| CN113563859B (en) * | 2021-07-16 | 2022-07-12 | 济南雅歌新材料科技有限公司 | Application of carbon-rich fluid in improving petroleum recovery |
| CN114042538A (en) * | 2021-11-16 | 2022-02-15 | 六盘水师范学院 | A method for efficiently inhibiting long-flame coal sliming |
| US12065929B2 (en) * | 2022-01-12 | 2024-08-20 | Saudi Arabian Oil Company | Petro-steering methodologies during under balanced coiled tubing (UBTC) drilling operations |
| CN114524944B (en) * | 2022-01-24 | 2023-05-26 | 成都昂赛福科技有限公司 | Synthesis of tertiary amine end-capped environment-friendly hyperbranched polyamine intercalation inhibitor and water-based drilling fluid |
| EP4511356A1 (en) | 2022-04-22 | 2025-02-26 | Ecolab USA Inc. | Polyhydroxy multi-ionic compounds and methods of making and use thereof |
| US11807803B1 (en) * | 2022-08-02 | 2023-11-07 | Saudi Arabian Oil Company | Cement spacer fluid with polyethyleneimine hydrochloride salt as a shale inhibitor |
| CN118440676B (en) * | 2024-04-30 | 2025-02-18 | 寿光新海能源技术有限公司 | Clay stabilizer composition and preparation method thereof |
| CN118751412B (en) * | 2024-07-30 | 2025-03-18 | 中国矿业大学 | A composite collector and deep flotation process for high-ash difficult-to-clean coal |
Citations (87)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB847321A (en) | 1957-08-30 | 1960-09-07 | Gen Mills Inc | Process for the inhibition of corrosion of metallic substances |
| US4075136A (en) * | 1974-01-25 | 1978-02-21 | Calgon Corporation | Functional ionene compositions and their use |
| GB1550420A (en) | 1975-12-19 | 1979-08-15 | Ciba Geigy Ag | Aminodi-and polyalkylamidoalkane sulphonic acids process for their manufacture and their use |
| US4166894A (en) * | 1974-01-25 | 1979-09-04 | Calgon Corporation | Functional ionene compositions and their use |
| US4259217A (en) | 1978-03-07 | 1981-03-31 | The Procter & Gamble Company | Laundry detergent compositions having enhanced greasy and oily soil removal performance |
| US4355071A (en) * | 1978-05-03 | 1982-10-19 | E. I. Dupont De Nemours And Company | Clear coat/color coat finish containing ultraviolet light stabilizer |
| JPS6116351B2 (en) | 1980-02-23 | 1986-04-30 | Noatsuku Furitsutsu | |
| JPS6116898B2 (en) | 1981-09-10 | 1986-05-02 | Matsushita Electric Ind Co Ltd | |
| EP0296441A2 (en) | 1987-06-22 | 1988-12-28 | Henkel Kommanditgesellschaft auf Aktien | Use of insoluble, polyfunctional quaternary ammonium compounds for the adsorptive binding of microorganisms |
| US5097904A (en) | 1991-02-28 | 1992-03-24 | Halliburton Company | Method for clay stabilization with quaternary amines |
| US5192798A (en) | 1988-02-19 | 1993-03-09 | The Upjohn Company | Lipophilic polyamines useful for treating hypercholesterolemia |
| US5342530A (en) | 1991-02-25 | 1994-08-30 | Nalco Chemical Company | Clay stabilizer |
| US5399746A (en) | 1994-02-07 | 1995-03-21 | Witco Corporation | Diquaternary bleach activators and compositions containing them |
| US5614616A (en) | 1990-08-23 | 1997-03-25 | Wolff Walsrode Aktiengesellschaft | Process for the production of polysaccharides |
| US5738795A (en) | 1996-06-14 | 1998-04-14 | Betzdearborn Inc. | Compositions and methods for water clarification |
| US6054054A (en) | 1994-09-06 | 2000-04-25 | Nalco Chemical Company | Chemical for the prevention of attachment of microorganisms to surfaces |
| JP2001187751A (en) | 1999-12-28 | 2001-07-10 | Nippon Shokubai Co Ltd | New tren derivative and method for producing the same |
| US20010044393A1 (en) | 2000-02-18 | 2001-11-22 | Peterson Robert Frederick | Rinse-off antimicrobial liquid cleansing composition |
| US20020155978A1 (en) | 1999-05-07 | 2002-10-24 | Ecolab Inc. | Detergent composition and method for removing soil |
| US6503880B1 (en) | 1997-08-27 | 2003-01-07 | Akzo Nobel N.V. | Cationic sugar surfactants from ethoxylated ammonium compounds and reducing saccharides |
| WO2004056843A2 (en) | 2002-12-20 | 2004-07-08 | Alpharma Aps | 10-substituted macrolide antibiotics |
| US6797785B1 (en) | 1999-03-29 | 2004-09-28 | Snf S.A. | Methods for synthesizing polyvinyl amine (PVA) type flocculating and coagulating agents, novel agents thus obtained, uses thereof and improved paper types thus obtained |
| US20040235677A1 (en) | 2003-05-23 | 2004-11-25 | Nguyen Philip D. | Methods and compositions for stabilizing swelling clays or migrating fines in formations |
| US6881710B1 (en) | 2004-03-04 | 2005-04-19 | Colonial Chemical Inc. | Personal care products based upon surfactants based upon alkyl polyglucoside quaternary compounds |
| US20050215461A1 (en) | 1997-05-20 | 2005-09-29 | Novapharm Research (Australia) Pty. Ltd. | Alkylpolyglucosides containing disinfectant compositions active against pseudomonas microorganism |
| US20060008496A1 (en) | 2004-07-06 | 2006-01-12 | Sandeep Kulkarni | Insulation paper facing containing an antimicotic or fungicide and methods of making and using the same |
| US7084129B1 (en) | 2003-09-15 | 2006-08-01 | Colonial Chemical | Antimicrobial quaternary surfactants based upon alkyl polyglycoside |
| US20060289359A1 (en) | 2005-06-23 | 2006-12-28 | Manek Maria B | Method of clarifying oily waste water |
| US20060289164A1 (en) * | 2005-06-24 | 2006-12-28 | Innovative Chemical Technologies Canada Ltd. | Clay control additive for wellbore fluids |
| JP2007054710A (en) | 2005-08-23 | 2007-03-08 | Research Institute Of Innovative Technology For The Earth | Gas separation membrane and gas separation method |
| US7507399B1 (en) | 2004-08-05 | 2009-03-24 | Surfatech Corporation | Functionalized polymeric surfactants based upon alkyl polyglycosides |
| US20100004316A1 (en) | 2006-09-29 | 2010-01-07 | University Of Utah Research Foundation | Multifunctional carriers for the delivery of nucleic acids and methods of use thereof |
| US20100029530A1 (en) | 2008-07-30 | 2010-02-04 | Reginald Keith Whiteley | Biostatic Medical Cleaning Products |
| US20100305014A1 (en) | 2009-06-02 | 2010-12-02 | Ecolab Inc. | Biodegradable surfactant blend |
| CN101972612A (en) | 2010-11-10 | 2011-02-16 | 郑州轻工业学院 | Sugar-based quaternary ammonium salt gemini surfactant and synthesis method thereof |
| US20110112007A1 (en) | 2009-11-06 | 2011-05-12 | Ecolab Inc. | Alkyl polyglucosides and a propoxylated-ethoxylated extended chain surfactant |
| US20120053111A1 (en) | 2010-06-21 | 2012-03-01 | Ecolab Usa Inc. | Alkyl polypentosides and alkyl polyglucosides (c8-c11) used for enhanced food soil removal |
| US20120070341A1 (en) | 2010-09-22 | 2012-03-22 | Ecolab Usa Inc. | Antimicrobial compositions containing cationic active ingredients and quaternary sugar derived surfactants |
| US20120115962A1 (en) | 2010-10-18 | 2012-05-10 | Rutgers, The State University Of New Jersey | Cyclodextrin-modified polyamines for delivery of therapeutic molecules |
| WO2012083497A1 (en) | 2010-12-22 | 2012-06-28 | General Electric Company | Methods of removing microbes from surfaces |
| JP2012136504A (en) | 2010-12-08 | 2012-07-19 | Lion Corp | Composition for oral cavity and oral cavity biofilm fungicide |
| CN102675535A (en) | 2012-05-22 | 2012-09-19 | 西南石油大学 | Branched polyacrylamide and preparation method thereof |
| US20120285690A1 (en) | 2011-05-12 | 2012-11-15 | Halliburton Energy Services, Inc. | Multi-Stage Methods and Compositions for Desensitizing Subterranean Formations Faces |
| WO2013087287A1 (en) | 2011-12-12 | 2013-06-20 | Unilever Plc | Laundry compositions |
| CN103288672A (en) | 2013-03-15 | 2013-09-11 | 山东大学(威海) | A preparation method for a novel dendritic oligomeric quaternary ammonium surfactant |
| US20130266669A1 (en) | 2010-12-22 | 2013-10-10 | Juan Jiang | Methods of removing microbes from surfaces |
| JP2014009177A (en) | 2012-06-28 | 2014-01-20 | Lion Corp | Hair detergent composition |
| US8631868B1 (en) | 2013-02-27 | 2014-01-21 | Polymer Ventures Inc. | Treatment of subterranean formations |
| US20140124454A1 (en) | 2012-11-01 | 2014-05-08 | Halosource, Inc. | Water treatment composition and method of using same |
| JP2014093768A (en) | 2012-10-31 | 2014-05-19 | Hyundai Motor Company Co Ltd | Apparatus and method for controlling image on room mirror |
| WO2014079621A1 (en) | 2012-11-20 | 2014-05-30 | Unilever Plc | Laundry compositions |
| US20140224733A1 (en) | 2013-02-14 | 2014-08-14 | Baker Hughes Incorporated | Colloidal silica addition to promote the separation of oil from water |
| CN104130335A (en) | 2014-07-23 | 2014-11-05 | 威海汉邦生物环保科技有限公司 | High-substituted quaternary ammonium polysaccharide bioflocculant and preparation method thereof |
| CN104130351A (en) | 2014-07-23 | 2014-11-05 | 威海汉邦生物环保科技有限公司 | Grafted copolymerized cationic polysaccharide bioflocculant and preparation method thereof |
| JP2014221859A (en) | 2013-05-13 | 2014-11-27 | ライオン株式会社 | Liquid detergent |
| WO2015084304A1 (en) | 2013-12-02 | 2015-06-11 | Empire Technology Development Llc | Novel gemini surfactants and their use |
| US20150203738A1 (en) | 2012-07-20 | 2015-07-23 | Dow Global Technologies Llc | Clay and shale inhibition agents and method of use |
| US20150210913A1 (en) | 2014-01-30 | 2015-07-30 | D.V. Satyanarayana Gupta | Clay stabilizer and method of use |
| CN105076201A (en) | 2015-09-09 | 2015-11-25 | 郑州轻工业学院 | Complex oilfield water flooding bactericide |
| US20160010035A1 (en) | 2014-07-14 | 2016-01-14 | Air Products And Chemicals, Inc. | Copper corrosion inhibition system |
| US20160030315A1 (en) | 2014-08-01 | 2016-02-04 | Ecolab Usa Inc. | Antimicrobial foaming compositions containing cationic active ingredients |
| CN105523956A (en) | 2014-09-30 | 2016-04-27 | 山东大学(威海) | Dendritic tetrameric quaternary ammonium salt surfactant preparation method |
| US20160145610A1 (en) | 2014-06-18 | 2016-05-26 | Case Western Reserve University | Compositions and methods for the delivery of nucleic acids |
| US20160262999A1 (en) | 2014-03-25 | 2016-09-15 | Ecolab Usa Inc. | Antimicrobial compositions containing cationic active ingredients |
| US20160264744A1 (en) | 2015-03-10 | 2016-09-15 | International Business Machines Corporation | Polyhemiaminal and polyhexahydrotriazine materials from 1,4 conjugate addition reactions |
| CN106172434A (en) | 2016-06-27 | 2016-12-07 | 宁德师范学院 | A kind of QIANGYI biocide mildewcide and preparation method thereof |
| WO2016205513A1 (en) | 2015-06-18 | 2016-12-22 | Ecolab Usa Inc. | Reverse emulsion breaker copolymers |
| WO2017003639A2 (en) | 2015-06-02 | 2017-01-05 | University Of Washington | Free-standing non-fouling polymers, their compositions, and related monomers |
| CN106423284A (en) | 2015-08-12 | 2017-02-22 | 中国石油化工股份有限公司 | Vinyl acetate catalyst and preparing method thereof |
| CN106423269A (en) | 2015-08-12 | 2017-02-22 | 中国石油化工股份有限公司 | Unsaturated acetic ester catalyst and preparing method thereof |
| US20170121560A1 (en) | 2015-10-28 | 2017-05-04 | Cabot Microelectronics Corporation | Tungsten-processing slurry with cationic surfactant and cyclodextrin |
| CN106634929A (en) | 2016-12-20 | 2017-05-10 | 陕西延长石油(集团)有限责任公司研究院 | Foaming agent for CO2 oil displacement system and preparation method thereof |
| CN106946743A (en) | 2017-05-02 | 2017-07-14 | 中国科学技术大学 | A kind of gemini cationic surfactant and preparation method thereof and fracturing fluid |
| US20170233643A1 (en) * | 2015-08-14 | 2017-08-17 | Halliburton Energy Services, Inc. | Biopolymer Based Cationic Surfactant for Clay Stabilization and Prevention of Sludging |
| WO2017201076A1 (en) | 2016-05-16 | 2017-11-23 | The Board Of Regents Of The University Of Texas System | Cationic sulfonamide amino lipids and amphiphilic zwitterionic amino lipids |
| CN107440935A (en) | 2017-07-28 | 2017-12-08 | 澳宝化妆品(惠州)有限公司 | A kind of creams hair-washing composition for strengthening anti-dandruff agent deposition |
| US20170360040A1 (en) | 2014-12-02 | 2017-12-21 | B.G. Negev Technologies And Applications Ltd., At Ben-Gurion University | Modified polysaccharides for use as anti-microbial agents |
| US20180066211A1 (en) | 2016-09-07 | 2018-03-08 | The Procter & Gamble Company | Liquid laundry Detergent Composition Comprising A First Polymer And A Second Polymer |
| US20180105629A1 (en) | 2015-07-17 | 2018-04-19 | Fujifilm Corporation | Curable composition |
| US20180118999A1 (en) * | 2016-11-01 | 2018-05-03 | Pfp Technology, Llc | Compositions and methods for stabilizing water sensitive clays and migrating fines in subterranean formations |
| CN108033895A (en) | 2017-11-30 | 2018-05-15 | 山东大学 | A kind of preparation method of four polycation quaternary surface active monomer of dendroid |
| CN108048249A (en) | 2017-12-06 | 2018-05-18 | 成都顺发消洗科技有限公司 | One kind washes shield type liquid detergent |
| US20180163020A1 (en) | 2016-12-14 | 2018-06-14 | Ecolab USA, Inc. | Quaternary cationic polymers |
| WO2018112548A1 (en) | 2016-12-22 | 2018-06-28 | Whiteley Corporation Pty Ltd | Biofilm disrupting composition for use on a surface |
| CN108938662A (en) | 2018-07-03 | 2018-12-07 | 南京大学 | A kind of purposes of cationised polysaccharide in the drug of preparation prevention and treatment bacterial biof iotalm |
| US20190062187A1 (en) | 2017-08-30 | 2019-02-28 | Ecolab Usa Inc. | Use of di-ionic compounds as corrosion inhibitors in a water system |
| US20190223434A1 (en) | 2018-01-19 | 2019-07-25 | Ecolab Usa Inc. | Compositions and methods for biofilm removal |
Family Cites Families (51)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2195974A (en) | 1936-05-19 | 1940-04-02 | Ig Farbenindustrie Ag | Process of producing new amino-carboxylic acids |
| US3077487A (en) | 1959-06-17 | 1963-02-12 | Victor Chemical Works | Lower alkylene and lower alkylenephenylene-lower alkylene polyamine bis n, n'-lower alkylene di and tri carboxylic acids, esters, salts, and chelates |
| JPS518966B1 (en) | 1971-03-18 | 1976-03-23 | ||
| US3793194A (en) | 1972-02-28 | 1974-02-19 | Hercules Inc | Scale and corrosion control in flowing waters |
| US4246030A (en) | 1978-12-08 | 1981-01-20 | The Mogul Corporation | Corrosion inhibiting compositions and the process for using same |
| JPS57185322A (en) | 1981-05-09 | 1982-11-15 | Miyoshi Oil & Fat Co Ltd | Amphoteric high polymer and its preparation |
| US4784797A (en) | 1984-03-29 | 1988-11-15 | The Dow Chemical Company | Process for inhibiting corrosion of ferrous metals |
| GB2159511B (en) | 1984-04-25 | 1988-09-21 | Dearborn Chemicals Ltd | A method of inhibiting corrosion in aqueous systems |
| DE3473365D1 (en) | 1984-05-26 | 1988-09-15 | Honeywell Bull Spa | Single error correction circuit for system memory |
| JPS6116898A (en) | 1984-07-04 | 1986-01-24 | 株式会社 昭和紙工 | Manufacture of cover of filing tool |
| NO173146C (en) | 1984-11-07 | 1993-11-10 | Berol Kemi Ab | PROCEDURE FOR ACID TREATMENT OF AN UNDERGRADUAL FORM |
| JPH0696839B2 (en) | 1985-09-09 | 1994-11-30 | ハリマ化成株式会社 | Papermaking additives |
| US4798675A (en) | 1987-10-19 | 1989-01-17 | The Mogul Corporation | Corrosion inhibiting compositions containing carboxylated phosphonic acids and sequestrants |
| US5053150A (en) | 1988-02-29 | 1991-10-01 | Exxon Chemical Patents Inc. | Polyepoxide modified adducts or reactants and oleaginous compositions containing same |
| US5019343A (en) | 1989-12-15 | 1991-05-28 | W. R. Grace & Co.-Conn. | Control of corrosion in aqueous systems using certain phosphonomethyl amines |
| US5670464A (en) | 1993-01-25 | 1997-09-23 | Kao Corporation | Additive for lubricating oils for diesel engines and lubricating oil compositions containing the same |
| JPH0913066A (en) | 1995-06-26 | 1997-01-14 | Kao Corp | Lubricating oil additive and lubricating oil composition for diesel engine |
| US6896826B2 (en) | 1997-01-09 | 2005-05-24 | Advanced Technology Materials, Inc. | Aqueous cleaning composition containing copper-specific corrosion inhibitor for cleaning inorganic residues on semiconductor substrate |
| US6238621B1 (en) | 1998-05-27 | 2001-05-29 | Solutia Inc. | Corrosion inhibiting compositions |
| US6080323A (en) | 1999-02-17 | 2000-06-27 | Nalco Chemical Company | Method of removing biofilms from surfaces submerged in a fouled water system |
| GB9924358D0 (en) | 1999-10-14 | 1999-12-15 | Brad Chem Technology Ltd | Corrosion inhibiting compositions |
| DE10043040A1 (en) | 2000-09-01 | 2002-03-28 | Goldschmidt Ag Th | Use of N-alkyl-beta-alanine derivatives for the production of cleaning anti-corrosion agents |
| EP1441988A1 (en) | 2001-10-09 | 2004-08-04 | Albemarle Corporation | Control of biofilms in industrial water systems |
| US20040009139A1 (en) | 2002-07-11 | 2004-01-15 | Colgate-Palmolive Company | Nongermicidal compositions |
| US6627612B1 (en) | 2002-10-01 | 2003-09-30 | Colonial Chemical Inc | Surfactants based upon alkyl polyglycosides |
| AU2003291567A1 (en) | 2002-11-19 | 2004-06-15 | Genzyme Corporation | Polymeric boronic acid derivatives as lipase inhibitors |
| DE102004025195A1 (en) | 2004-05-22 | 2005-12-08 | Goldschmidt Gmbh | Process for the preparation of alkyl glycosides |
| US20060180794A1 (en) | 2005-02-15 | 2006-08-17 | Goddard Richard J | Polyamine-based corrosion inhibitors |
| WO2006092736A2 (en) | 2005-03-02 | 2006-09-08 | Mondi Packaging South Africa (Proprietary) Limited | Preparation of polymer latexes |
| JP2007077082A (en) | 2005-09-14 | 2007-03-29 | Fujifilm Corp | Hair cosmetics |
| JP2007256445A (en) | 2006-03-22 | 2007-10-04 | Mitsubishi Paper Mills Ltd | Photopolymerizable composition and photosensitive lithographic printing plate material |
| US7851655B2 (en) | 2006-12-19 | 2010-12-14 | Nalco Company | Functionalized amine-based corrosion inhibitors for galvanized metal surfaces and method of using same |
| US7375064B1 (en) | 2007-02-20 | 2008-05-20 | Surfatech Corporation | Functionalized polymeric surfactants based upon alkyl polyglycosides |
| US8227637B2 (en) | 2008-03-19 | 2012-07-24 | Epolin, Inc. | Stable, water-soluble near infrared dyes |
| WO2009153209A1 (en) | 2008-06-19 | 2009-12-23 | Henkel Ag & Co. Kgaa | Modified polymeric pro-fragrance |
| JP5914907B2 (en) | 2008-08-11 | 2016-05-11 | 株式会社オートネットワーク技術研究所 | Rust preventive and surface-treated metal |
| MX2010006074A (en) | 2010-06-03 | 2011-12-16 | Mexicano Inst Petrol | Amino and imino propionic acids, process of preparation and use. |
| JP6242184B2 (en) | 2013-11-22 | 2017-12-06 | Kjケミカルズ株式会社 | Method for producing N-substituted (meth) acrylamide |
| WO2015171105A1 (en) | 2014-05-05 | 2015-11-12 | Multi-Chem Group, Llc | Multiple hydrophilic head corrosion inhibitors |
| MX2017000444A (en) | 2014-07-15 | 2017-05-01 | Akzo Nobel Coatings Int Bv | Antifouling coating composition comprising at least two salt-containing copolymers. |
| US10094203B2 (en) | 2014-11-06 | 2018-10-09 | Ces Technology S.À R.L. | Corrosion inhibitors for drilling fluid brines |
| CN104744709B (en) | 2015-04-10 | 2021-08-20 | 上海汇锐化工科技有限公司 | Dendritic polyamide type ester quaternary ammonium salt and synthesis process thereof |
| CN105884640B (en) | 2015-06-12 | 2018-02-27 | 宁波大学 | A kind of dianion interfacial initiation agent and preparation method thereof |
| CA2993189C (en) | 2015-07-27 | 2023-10-24 | Ecolab Usa Inc. | Cleaning and removal of wax deposits in oil and gas wells using cationic polymers |
| US10174429B2 (en) | 2015-11-05 | 2019-01-08 | Chemtreat, Inc | Corrosion control for water systems using tin corrosion inhibitor with a hydroxycarboxylic acid |
| US10499647B2 (en) | 2017-01-10 | 2019-12-10 | Ecolab Usa Inc. | Use of an encapsulated chlorine bleach particle to reduce gas evolution during dispensing |
| US11084974B2 (en) | 2018-08-29 | 2021-08-10 | Championx Usa Inc. | Use of multiple charged cationic compounds derived from polyamines for clay stabilization in oil and gas operations |
| EP3844112A1 (en) | 2018-08-29 | 2021-07-07 | Ecolab USA Inc. | Use of multiple charged cationic compounds derived from primary amines or polyamines for microbial fouling control in a water system |
| WO2020047181A1 (en) | 2018-08-29 | 2020-03-05 | Ecolab Usa Inc. | Use of multiple charged ionic compounds derived from polyamines for waste water clarification |
| WO2020047021A1 (en) | 2018-08-29 | 2020-03-05 | Ecolab Usa Inc. | Multiple charged ionic compounds derived from polyamines and compositions thereof and methods of preparation thereof |
| CN113840945B (en) | 2019-04-16 | 2023-09-26 | 埃科莱布美国股份有限公司 | Use of multiply charged cationic compounds derived from polyamines and compositions thereof for corrosion inhibition in water systems |
-
2019
- 2019-08-29 US US16/554,805 patent/US11084974B2/en active Active
-
2021
- 2021-06-30 US US17/305,069 patent/US11702586B2/en active Active
Patent Citations (91)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB847321A (en) | 1957-08-30 | 1960-09-07 | Gen Mills Inc | Process for the inhibition of corrosion of metallic substances |
| US4075136A (en) * | 1974-01-25 | 1978-02-21 | Calgon Corporation | Functional ionene compositions and their use |
| US4166894A (en) * | 1974-01-25 | 1979-09-04 | Calgon Corporation | Functional ionene compositions and their use |
| GB1550420A (en) | 1975-12-19 | 1979-08-15 | Ciba Geigy Ag | Aminodi-and polyalkylamidoalkane sulphonic acids process for their manufacture and their use |
| US4259217A (en) | 1978-03-07 | 1981-03-31 | The Procter & Gamble Company | Laundry detergent compositions having enhanced greasy and oily soil removal performance |
| US4355071A (en) * | 1978-05-03 | 1982-10-19 | E. I. Dupont De Nemours And Company | Clear coat/color coat finish containing ultraviolet light stabilizer |
| JPS6116351B2 (en) | 1980-02-23 | 1986-04-30 | Noatsuku Furitsutsu | |
| JPS6116898B2 (en) | 1981-09-10 | 1986-05-02 | Matsushita Electric Ind Co Ltd | |
| EP0296441A2 (en) | 1987-06-22 | 1988-12-28 | Henkel Kommanditgesellschaft auf Aktien | Use of insoluble, polyfunctional quaternary ammonium compounds for the adsorptive binding of microorganisms |
| US5192798A (en) | 1988-02-19 | 1993-03-09 | The Upjohn Company | Lipophilic polyamines useful for treating hypercholesterolemia |
| US5614616A (en) | 1990-08-23 | 1997-03-25 | Wolff Walsrode Aktiengesellschaft | Process for the production of polysaccharides |
| US5342530A (en) | 1991-02-25 | 1994-08-30 | Nalco Chemical Company | Clay stabilizer |
| US5097904A (en) | 1991-02-28 | 1992-03-24 | Halliburton Company | Method for clay stabilization with quaternary amines |
| US5399746A (en) | 1994-02-07 | 1995-03-21 | Witco Corporation | Diquaternary bleach activators and compositions containing them |
| US6054054A (en) | 1994-09-06 | 2000-04-25 | Nalco Chemical Company | Chemical for the prevention of attachment of microorganisms to surfaces |
| US5738795A (en) | 1996-06-14 | 1998-04-14 | Betzdearborn Inc. | Compositions and methods for water clarification |
| US20050215461A1 (en) | 1997-05-20 | 2005-09-29 | Novapharm Research (Australia) Pty. Ltd. | Alkylpolyglucosides containing disinfectant compositions active against pseudomonas microorganism |
| US6503880B1 (en) | 1997-08-27 | 2003-01-07 | Akzo Nobel N.V. | Cationic sugar surfactants from ethoxylated ammonium compounds and reducing saccharides |
| US6797785B1 (en) | 1999-03-29 | 2004-09-28 | Snf S.A. | Methods for synthesizing polyvinyl amine (PVA) type flocculating and coagulating agents, novel agents thus obtained, uses thereof and improved paper types thus obtained |
| US20020155978A1 (en) | 1999-05-07 | 2002-10-24 | Ecolab Inc. | Detergent composition and method for removing soil |
| JP2001187751A (en) | 1999-12-28 | 2001-07-10 | Nippon Shokubai Co Ltd | New tren derivative and method for producing the same |
| US20010044393A1 (en) | 2000-02-18 | 2001-11-22 | Peterson Robert Frederick | Rinse-off antimicrobial liquid cleansing composition |
| WO2004056843A2 (en) | 2002-12-20 | 2004-07-08 | Alpharma Aps | 10-substituted macrolide antibiotics |
| US20040235677A1 (en) | 2003-05-23 | 2004-11-25 | Nguyen Philip D. | Methods and compositions for stabilizing swelling clays or migrating fines in formations |
| US7084129B1 (en) | 2003-09-15 | 2006-08-01 | Colonial Chemical | Antimicrobial quaternary surfactants based upon alkyl polyglycoside |
| US6881710B1 (en) | 2004-03-04 | 2005-04-19 | Colonial Chemical Inc. | Personal care products based upon surfactants based upon alkyl polyglucoside quaternary compounds |
| US20060008496A1 (en) | 2004-07-06 | 2006-01-12 | Sandeep Kulkarni | Insulation paper facing containing an antimicotic or fungicide and methods of making and using the same |
| US7507399B1 (en) | 2004-08-05 | 2009-03-24 | Surfatech Corporation | Functionalized polymeric surfactants based upon alkyl polyglycosides |
| US20060289359A1 (en) | 2005-06-23 | 2006-12-28 | Manek Maria B | Method of clarifying oily waste water |
| US20060289164A1 (en) * | 2005-06-24 | 2006-12-28 | Innovative Chemical Technologies Canada Ltd. | Clay control additive for wellbore fluids |
| JP2007054710A (en) | 2005-08-23 | 2007-03-08 | Research Institute Of Innovative Technology For The Earth | Gas separation membrane and gas separation method |
| US20100004316A1 (en) | 2006-09-29 | 2010-01-07 | University Of Utah Research Foundation | Multifunctional carriers for the delivery of nucleic acids and methods of use thereof |
| US20100029530A1 (en) | 2008-07-30 | 2010-02-04 | Reginald Keith Whiteley | Biostatic Medical Cleaning Products |
| US20100305014A1 (en) | 2009-06-02 | 2010-12-02 | Ecolab Inc. | Biodegradable surfactant blend |
| US20110112007A1 (en) | 2009-11-06 | 2011-05-12 | Ecolab Inc. | Alkyl polyglucosides and a propoxylated-ethoxylated extended chain surfactant |
| US20120053111A1 (en) | 2010-06-21 | 2012-03-01 | Ecolab Usa Inc. | Alkyl polypentosides and alkyl polyglucosides (c8-c11) used for enhanced food soil removal |
| US20150290100A1 (en) | 2010-09-22 | 2015-10-15 | Ecolab Usa Inc. | Antimicrobial compositions containing cationic active ingredients and quaternary sugar derived surfactants |
| US20120070341A1 (en) | 2010-09-22 | 2012-03-22 | Ecolab Usa Inc. | Antimicrobial compositions containing cationic active ingredients and quaternary sugar derived surfactants |
| US20120115962A1 (en) | 2010-10-18 | 2012-05-10 | Rutgers, The State University Of New Jersey | Cyclodextrin-modified polyamines for delivery of therapeutic molecules |
| CN101972612A (en) | 2010-11-10 | 2011-02-16 | 郑州轻工业学院 | Sugar-based quaternary ammonium salt gemini surfactant and synthesis method thereof |
| JP2012136504A (en) | 2010-12-08 | 2012-07-19 | Lion Corp | Composition for oral cavity and oral cavity biofilm fungicide |
| WO2012083497A1 (en) | 2010-12-22 | 2012-06-28 | General Electric Company | Methods of removing microbes from surfaces |
| US20130266669A1 (en) | 2010-12-22 | 2013-10-10 | Juan Jiang | Methods of removing microbes from surfaces |
| US20120285690A1 (en) | 2011-05-12 | 2012-11-15 | Halliburton Energy Services, Inc. | Multi-Stage Methods and Compositions for Desensitizing Subterranean Formations Faces |
| WO2013087287A1 (en) | 2011-12-12 | 2013-06-20 | Unilever Plc | Laundry compositions |
| CN102675535B (en) | 2012-05-22 | 2013-11-27 | 西南石油大学 | A kind of branched polyacrylamide and preparation method thereof |
| CN102675535A (en) | 2012-05-22 | 2012-09-19 | 西南石油大学 | Branched polyacrylamide and preparation method thereof |
| JP2014009177A (en) | 2012-06-28 | 2014-01-20 | Lion Corp | Hair detergent composition |
| US20150203738A1 (en) | 2012-07-20 | 2015-07-23 | Dow Global Technologies Llc | Clay and shale inhibition agents and method of use |
| JP2014093768A (en) | 2012-10-31 | 2014-05-19 | Hyundai Motor Company Co Ltd | Apparatus and method for controlling image on room mirror |
| US20140124454A1 (en) | 2012-11-01 | 2014-05-08 | Halosource, Inc. | Water treatment composition and method of using same |
| WO2014079621A1 (en) | 2012-11-20 | 2014-05-30 | Unilever Plc | Laundry compositions |
| US20140224733A1 (en) | 2013-02-14 | 2014-08-14 | Baker Hughes Incorporated | Colloidal silica addition to promote the separation of oil from water |
| US8631868B1 (en) | 2013-02-27 | 2014-01-21 | Polymer Ventures Inc. | Treatment of subterranean formations |
| CN103288672A (en) | 2013-03-15 | 2013-09-11 | 山东大学(威海) | A preparation method for a novel dendritic oligomeric quaternary ammonium surfactant |
| JP2014221859A (en) | 2013-05-13 | 2014-11-27 | ライオン株式会社 | Liquid detergent |
| WO2015084304A1 (en) | 2013-12-02 | 2015-06-11 | Empire Technology Development Llc | Novel gemini surfactants and their use |
| US20150210913A1 (en) | 2014-01-30 | 2015-07-30 | D.V. Satyanarayana Gupta | Clay stabilizer and method of use |
| US20160262999A1 (en) | 2014-03-25 | 2016-09-15 | Ecolab Usa Inc. | Antimicrobial compositions containing cationic active ingredients |
| US20160145610A1 (en) | 2014-06-18 | 2016-05-26 | Case Western Reserve University | Compositions and methods for the delivery of nucleic acids |
| US20160010035A1 (en) | 2014-07-14 | 2016-01-14 | Air Products And Chemicals, Inc. | Copper corrosion inhibition system |
| CN104130335A (en) | 2014-07-23 | 2014-11-05 | 威海汉邦生物环保科技有限公司 | High-substituted quaternary ammonium polysaccharide bioflocculant and preparation method thereof |
| CN104130351A (en) | 2014-07-23 | 2014-11-05 | 威海汉邦生物环保科技有限公司 | Grafted copolymerized cationic polysaccharide bioflocculant and preparation method thereof |
| US20160030315A1 (en) | 2014-08-01 | 2016-02-04 | Ecolab Usa Inc. | Antimicrobial foaming compositions containing cationic active ingredients |
| CN105523956A (en) | 2014-09-30 | 2016-04-27 | 山东大学(威海) | Dendritic tetrameric quaternary ammonium salt surfactant preparation method |
| US20170360040A1 (en) | 2014-12-02 | 2017-12-21 | B.G. Negev Technologies And Applications Ltd., At Ben-Gurion University | Modified polysaccharides for use as anti-microbial agents |
| US20160264744A1 (en) | 2015-03-10 | 2016-09-15 | International Business Machines Corporation | Polyhemiaminal and polyhexahydrotriazine materials from 1,4 conjugate addition reactions |
| WO2017003639A2 (en) | 2015-06-02 | 2017-01-05 | University Of Washington | Free-standing non-fouling polymers, their compositions, and related monomers |
| WO2016205513A1 (en) | 2015-06-18 | 2016-12-22 | Ecolab Usa Inc. | Reverse emulsion breaker copolymers |
| US20180105629A1 (en) | 2015-07-17 | 2018-04-19 | Fujifilm Corporation | Curable composition |
| CN106423284A (en) | 2015-08-12 | 2017-02-22 | 中国石油化工股份有限公司 | Vinyl acetate catalyst and preparing method thereof |
| CN106423269A (en) | 2015-08-12 | 2017-02-22 | 中国石油化工股份有限公司 | Unsaturated acetic ester catalyst and preparing method thereof |
| US20170233643A1 (en) * | 2015-08-14 | 2017-08-17 | Halliburton Energy Services, Inc. | Biopolymer Based Cationic Surfactant for Clay Stabilization and Prevention of Sludging |
| CN105076201A (en) | 2015-09-09 | 2015-11-25 | 郑州轻工业学院 | Complex oilfield water flooding bactericide |
| US20170121560A1 (en) | 2015-10-28 | 2017-05-04 | Cabot Microelectronics Corporation | Tungsten-processing slurry with cationic surfactant and cyclodextrin |
| US20170349543A1 (en) | 2016-05-16 | 2017-12-07 | The Board Of Regents Of The University Of Texas System | Cationic sulfonamide amino lipids and amphiphilic zwitterionic amino lipids |
| WO2017201076A1 (en) | 2016-05-16 | 2017-11-23 | The Board Of Regents Of The University Of Texas System | Cationic sulfonamide amino lipids and amphiphilic zwitterionic amino lipids |
| CN106172434A (en) | 2016-06-27 | 2016-12-07 | 宁德师范学院 | A kind of QIANGYI biocide mildewcide and preparation method thereof |
| US20180066211A1 (en) | 2016-09-07 | 2018-03-08 | The Procter & Gamble Company | Liquid laundry Detergent Composition Comprising A First Polymer And A Second Polymer |
| US20180118999A1 (en) * | 2016-11-01 | 2018-05-03 | Pfp Technology, Llc | Compositions and methods for stabilizing water sensitive clays and migrating fines in subterranean formations |
| US20180163020A1 (en) | 2016-12-14 | 2018-06-14 | Ecolab USA, Inc. | Quaternary cationic polymers |
| CN106634929A (en) | 2016-12-20 | 2017-05-10 | 陕西延长石油(集团)有限责任公司研究院 | Foaming agent for CO2 oil displacement system and preparation method thereof |
| WO2018112548A1 (en) | 2016-12-22 | 2018-06-28 | Whiteley Corporation Pty Ltd | Biofilm disrupting composition for use on a surface |
| CN106946743A (en) | 2017-05-02 | 2017-07-14 | 中国科学技术大学 | A kind of gemini cationic surfactant and preparation method thereof and fracturing fluid |
| CN107440935A (en) | 2017-07-28 | 2017-12-08 | 澳宝化妆品(惠州)有限公司 | A kind of creams hair-washing composition for strengthening anti-dandruff agent deposition |
| US20190062187A1 (en) | 2017-08-30 | 2019-02-28 | Ecolab Usa Inc. | Use of di-ionic compounds as corrosion inhibitors in a water system |
| WO2019046409A1 (en) | 2017-08-30 | 2019-03-07 | Ecolab Usa Inc. | Molecules having one hydrophobic group and two identical hydrophilic ionic groups and compositions thereof |
| CN108033895A (en) | 2017-11-30 | 2018-05-15 | 山东大学 | A kind of preparation method of four polycation quaternary surface active monomer of dendroid |
| CN108048249A (en) | 2017-12-06 | 2018-05-18 | 成都顺发消洗科技有限公司 | One kind washes shield type liquid detergent |
| US20190223434A1 (en) | 2018-01-19 | 2019-07-25 | Ecolab Usa Inc. | Compositions and methods for biofilm removal |
| CN108938662A (en) | 2018-07-03 | 2018-12-07 | 南京大学 | A kind of purposes of cationised polysaccharide in the drug of preparation prevention and treatment bacterial biof iotalm |
Non-Patent Citations (21)
| Title |
|---|
| "Azamethonium", http://pubchem.ncbi.nlm.nih.gov/compound/9383, last modified Oct. 6, 2018 and accessed by Applicant Oct. 11, 2018. |
| Bi et al., "Dendrimer-Based Demulsifiers for Polymer Flooding Oil-in-Water Emulsions", Energy Fuels, vol. 31. No. 5, pp. 5395-5401, Apr. 20, 2017. |
| Brycki et al., "The biodegradation of monomeric and dimeric alkylammonium surfactants", Journal of Hazardous Materials, vol. 280, pp. 797-815, Aug. 6, 2014. |
| Fan et al., "Synthesis and Aggregation Behavior of a Hexameric Quaternary Ammonium Surfactant", Langmuir, vol. 27, pp. 10570-10579, Jul. 28, 2011. |
| Gan et al., "Sugar-Based Ester Quaternary Ammonium Compounds and Their Surfactant Properties", Journal of Surfactants and Detergents, vol. 17, Issue 3, pp. 465-470, Jan. 3, 2014. |
| Kawakami et al., "Antibacterial Activity of Radial Compounds with Peripheral Quaternary Ammonium Units", Transactions of the Materials Research Society of Japan, vol. 35[4], pp. 885-887, 2010. |
| Kramer et al., "Dendritic polyamines: simple access to new materials with defined treelike structures for application in nonviral gene delivery", Chembiochem, vol. 5(8), pp. 1081-1087, Aug. 6, 2004. |
| Labade et al., "Cesium fluoride catalyzed Aza-Michael addition reaction in aqueous media", Monatsh Chem., vol. 142, pp. 1055-1059, Jul. 19, 2011. |
| Miller et al., "Non-viral CRISPR/Cas gene editing in vitro and in vivo enabled by synthetic nanoparticle co-delivery of Cas9 mRNA and sgRNA", Angew Chem Int Ed Engl., vol. 56(4), pp. 1059-1063, Jan. 19, 2017. |
| Negm et al., "Synthesis, Characterization and Biological Activity of Sugar-Based Gemini Cationic Amphiphiles", Journal of Surfactants and Detergents, vol. 11, Issue 3, pp. 215-221, Apr. 26, 2008. |
| Ning et al., "Synthesis and characterization of a novel non-polyether demulsifier", Chemical Engineer, 3 pages, 2013. |
| Somerscales, Euan F.C., "Fundamentals of Corrosion Fouling", Experimental Thermal and Fluid Science, vol. 14, pp. 335-355, 1997. |
| Tan et al., "The use of quaternised chitosan-loaded PMMA to inhibit biofilm formation and downregulate the virulence-associated gene expression of antibiotic-resistant Staphylococcus", Biomaterials, vol. 33, Issue 2, pp. 365-377, Jan. 2012. |
| Wang et al., "A novel environment-sensitive biodegradable polydisulfide with protonatable pendants for nucleic acid delivery", Journal of Controlled Release, vol. 120, pp. 250-258, May 11, 2007. |
| Zaky, Mohamad, "Biocidal Activities of Cationic Surface Active Starch and Its Transition Metal Complexes Against Different Bacterial Strains", Journal of Surfactants and Detergents, vol. 13, Issue 3, pp. 255-260, Jul. 2010. |
| Zhang et al., "PAMAM-Based Dendrimers with Different Alkyl Chains Self-Assemble on Silica Surfaces: Controllable Layer Structure and Molecular Aggregation", J. Phys. Chem. B, vol. 122, pp. 6648-6655, Jun. 13, 2018. |
| Zhang et al., Supporting Information, Beijing National Laboratory for Molecular Sciences, published with Controllable Self-Assembly of Amphiphilic Dendrimers on a Silica Surface, 2016 (Year: 2016). * |
| Zhang, et al. Controllable Self-Assembly of Amphiphilic Dendrimers on a Silica Surface: The Effect of Molecular Topological Structure and Salinity, Oct. 5, 2016, The Journal of Physical Chemistry, 8, pp. 10990-10999 (Year: 2016). * |
| Zhi et al., "Self-aggregation and antimicrobial activity of saccharide-cationic surfactants", Colloids and Surfaces A: Physicochemical and Engineering Aspects, vol. 456, pp. 231-237, Aug. 2014. |
| Zhou et al., "Cooperative binding and self-assembling behavior of cationic low molecular-weight dendrons with RNA molecules", Organic & Biomolecular Chemistry, vol. 4, pp. 581-585, 2006. |
| Zielinski et al., "Synthesis of new quaternary ammonium salts for organophilization of fillers for polymeric nanocomposites", www.miesiecznikchemik.pl, 2007. |
Cited By (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11565958B2 (en) | 2017-08-30 | 2023-01-31 | Ecolab Usa Inc. | Use of di-ionic compounds as corrosion inhibitors in a water system |
| US12103881B2 (en) | 2017-08-30 | 2024-10-01 | Ecolab Usa Inc. | Molecules having one hydrophobic group and two identical hydrophilic ionic groups and compositions thereof and methods of preparation thereof |
| US12082580B2 (en) | 2018-08-29 | 2024-09-10 | Ecolab Usa Inc. | Use of multiple charged cationic compounds derived from primary amines or polyamines for microbial fouling control in a water system |
| US11685709B2 (en) * | 2018-08-29 | 2023-06-27 | Ecolab Usa Inc. | Multiple charged ionic compounds derived from polyamines and compositions thereof and use thereof as reverse emulsion breakers in oil and gas operations |
| US11702586B2 (en) | 2018-08-29 | 2023-07-18 | Championx Usa Inc. | Use of multiple charged cationic compounds derived from polyamines for clay stabilization in oil and gas operations |
| US11926543B2 (en) | 2018-08-29 | 2024-03-12 | Ecolab Usa Inc. | Use of multiple charged ionic compounds derived from polyamines for waste water clarification |
| US12202784B2 (en) | 2018-08-29 | 2025-01-21 | Ecolab Usa Inc. | Multiple charged ionic compounds derived from polyamines and compositions thereof and methods of preparation thereof |
| US20220002174A1 (en) * | 2019-01-29 | 2022-01-06 | Ecolab Usa Inc. | Use of cationic sugar-based compounds for microbial fouling control in a water system |
| US20220002175A1 (en) * | 2019-01-29 | 2022-01-06 | Ecolab Usa Inc. | Use of cationic sugar-based compounds as corrosion inhibitors in a water system |
| US12365608B2 (en) * | 2019-01-29 | 2025-07-22 | Ecolab Usa Inc. | Use of cationic sugar-based compounds for microbial fouling control in a water system |
| US11639553B2 (en) | 2019-04-16 | 2023-05-02 | Ecolab Usa Inc. | Compositions comprising multiple charged cationic compounds derived from polyamines for corrosion inhibition in a water system |
| US20230192929A1 (en) * | 2021-12-22 | 2023-06-22 | Ecolab Usa Inc. | Compositions comprising multiple charged cationic compounds for soil release |
| WO2024124129A1 (en) * | 2022-12-09 | 2024-06-13 | Championx Llc | Corrosion inhibitor having silica nanoparticles |
| US12037535B1 (en) * | 2023-05-11 | 2024-07-16 | King Fahd University Of Petroleum And Minerals | Method of drilling hydrocarbon bearing geological formation with horizontal wellbore |
| US12116523B1 (en) | 2023-05-11 | 2024-10-15 | King Fahd University Of Petroleum And Minerals | Method of making drilling fluid composition with functionalized carbon |
| US20240376366A1 (en) * | 2023-05-11 | 2024-11-14 | King Fahd University Of Petroleum And Minerals | Method of drilling a shale formation with a drilling fluid composition |
| US12180415B2 (en) * | 2023-05-11 | 2024-12-31 | King Fahd University Of Petroleum And Minerals | Method of drilling a shale formation with a drilling fluid composition |
| US11981857B1 (en) * | 2023-05-11 | 2024-05-14 | King Fahd University Of Petroleum And Minerals | Drilling fluid composition and method of making the composition |
| US12180424B1 (en) | 2023-08-22 | 2024-12-31 | Halliburton Energy Services, Inc. | Methods and compositions used in controlling formation sand production, proppant flowback, and fines migration |
Also Published As
| Publication number | Publication date |
|---|---|
| US11702586B2 (en) | 2023-07-18 |
| US20200071602A1 (en) | 2020-03-05 |
| US20210332288A1 (en) | 2021-10-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US11702586B2 (en) | Use of multiple charged cationic compounds derived from polyamines for clay stabilization in oil and gas operations | |
| US20230278949A1 (en) | Multiple charged ionic compounds derived from polyamines and compositions thereof and use thereof as reverse emulsion breakers in oil and gas operations | |
| US11639553B2 (en) | Compositions comprising multiple charged cationic compounds derived from polyamines for corrosion inhibition in a water system | |
| AU2019331448B2 (en) | Use of multiple charged cationic compounds derived from primary amines or polyamines for microbial fouling control in a water system | |
| CA3110365C (en) | Use of multiple charged ionic compounds derived from polyamines for waste water clarification | |
| WO2017079594A1 (en) | Friction-reducing compositions formulated with highly concentrated brine | |
| US12359113B2 (en) | Method for dispersing kinetic hydrate inhibitors |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| FEPP | Fee payment procedure |
Free format text: ENTITY STATUS SET TO UNDISCOUNTED (ORIGINAL EVENT CODE: BIG.); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY |
|
| AS | Assignment |
Owner name: ECOLAB USA INC., MINNESOTA Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:DHAWAN, ASHISH;HARKNESS, KELLEN;SILVERNAIL, CARTER M.;AND OTHERS;REEL/FRAME:050223/0280 Effective date: 20190821 |
|
| AS | Assignment |
Owner name: BANK OF AMERICA, N.A., NORTH CAROLINA Free format text: SECURITY INTEREST;ASSIGNOR:CHAMPIONX USA INC.;REEL/FRAME:052848/0368 Effective date: 20200603 Owner name: JPMORGAN CHASE BANK, N.A., NEW YORK Free format text: SECURITY INTEREST;ASSIGNOR:CHAMPIONX USA INC.;REEL/FRAME:053250/0001 Effective date: 20200603 |
|
| AS | Assignment |
Owner name: CHAMPIONX USA INC., TEXAS Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNOR:ECOLAB USA INC.;REEL/FRAME:053639/0001 Effective date: 20200825 |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: NON FINAL ACTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: NON FINAL ACTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: RESPONSE TO NON-FINAL OFFICE ACTION ENTERED AND FORWARDED TO EXAMINER |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: NOTICE OF ALLOWANCE MAILED -- APPLICATION RECEIVED IN OFFICE OF PUBLICATIONS |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: PUBLICATIONS -- ISSUE FEE PAYMENT RECEIVED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: PUBLICATIONS -- ISSUE FEE PAYMENT VERIFIED |
|
| STCF | Information on status: patent grant |
Free format text: PATENTED CASE |
|
| AS | Assignment |
Owner name: CHAMPIONX USA INC., TEXAS Free format text: RELEASE BY SECURED PARTY;ASSIGNOR:BANK OF AMERICA, N.A.;REEL/FRAME:060304/0267 Effective date: 20220607 Owner name: CHAMPIONX USA INC., TEXAS Free format text: RELEASE OF SECURITY INTEREST;ASSIGNOR:BANK OF AMERICA, N.A.;REEL/FRAME:060304/0267 Effective date: 20220607 |
|
| AS | Assignment |
Owner name: CHAMPIONX LLC, TEXAS Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNOR:CHAMPIONX USA INC.;REEL/FRAME:065869/0839 Effective date: 20231114 Owner name: CHAMPIONX LLC, TEXAS Free format text: ASSIGNMENT OF ASSIGNOR'S INTEREST;ASSIGNOR:CHAMPIONX USA INC.;REEL/FRAME:065869/0839 Effective date: 20231114 |
|
| MAFP | Maintenance fee payment |
Free format text: PAYMENT OF MAINTENANCE FEE, 4TH YEAR, LARGE ENTITY (ORIGINAL EVENT CODE: M1551); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY Year of fee payment: 4 |
|
| CC | Certificate of correction | ||
| AS | Assignment |
Owner name: CHAMPIONX LLC, TEXAS Free format text: RELEASE OF SECURITY INTEREST IN PATENTS;ASSIGNOR:JPMORGAN CHASE BANK, N.A.;REEL/FRAME:072004/0019 Effective date: 20250716 Owner name: APERGY ESP SYSTEMS, LLC, TEXAS Free format text: RELEASE OF SECURITY INTEREST IN PATENTS;ASSIGNOR:JPMORGAN CHASE BANK, N.A.;REEL/FRAME:072004/0019 Effective date: 20250716 Owner name: APERGY BMCS ACQUISITION CORP, TEXAS Free format text: RELEASE OF SECURITY INTEREST IN PATENTS;ASSIGNOR:JPMORGAN CHASE BANK, N.A.;REEL/FRAME:072004/0019 Effective date: 20250716 Owner name: HARBISON-FISCHER, INC., TEXAS Free format text: RELEASE OF SECURITY INTEREST IN PATENTS;ASSIGNOR:JPMORGAN CHASE BANK, N.A.;REEL/FRAME:072004/0019 Effective date: 20250716 Owner name: NORRIS RODS, INC.,, TEXAS Free format text: RELEASE OF SECURITY INTEREST IN PATENTS;ASSIGNOR:JPMORGAN CHASE BANK, N.A.;REEL/FRAME:072004/0019 Effective date: 20250716 Owner name: NORRISEAL-WELLMARK, INC., TEXAS Free format text: RELEASE OF SECURITY INTEREST IN PATENTS;ASSIGNOR:JPMORGAN CHASE BANK, N.A.;REEL/FRAME:072004/0019 Effective date: 20250716 Owner name: PCS FERGUSON, INC., TEXAS Free format text: RELEASE OF SECURITY INTEREST IN PATENTS;ASSIGNOR:JPMORGAN CHASE BANK, N.A.;REEL/FRAME:072004/0019 Effective date: 20250716 Owner name: QUARTZDYNE, INC., TEXAS Free format text: RELEASE OF SECURITY INTEREST IN PATENTS;ASSIGNOR:JPMORGAN CHASE BANK, N.A.;REEL/FRAME:072004/0019 Effective date: 20250716 Owner name: US SYNTHETIC CORPORATION, TEXAS Free format text: RELEASE OF SECURITY INTEREST IN PATENTS;ASSIGNOR:JPMORGAN CHASE BANK, N.A.;REEL/FRAME:072004/0019 Effective date: 20250716 |