TW202227445A - Use of inhibiting genetically defective hiv virus - Google Patents
Use of inhibiting genetically defective hiv virus Download PDFInfo
- Publication number
- TW202227445A TW202227445A TW110140359A TW110140359A TW202227445A TW 202227445 A TW202227445 A TW 202227445A TW 110140359 A TW110140359 A TW 110140359A TW 110140359 A TW110140359 A TW 110140359A TW 202227445 A TW202227445 A TW 202227445A
- Authority
- TW
- Taiwan
- Prior art keywords
- alkyl
- halogen
- alkoxy
- deuterium
- independently selected
- Prior art date
Links
- 230000002950 deficient Effects 0.000 title claims abstract description 7
- 241000700605 Viruses Species 0.000 title claims description 15
- 230000002401 inhibitory effect Effects 0.000 title abstract 3
- 241000725303 Human immunodeficiency virus Species 0.000 claims abstract description 19
- 239000003814 drug Substances 0.000 claims abstract description 14
- 229940079593 drug Drugs 0.000 claims abstract description 8
- 125000000217 alkyl group Chemical group 0.000 claims description 216
- 229910052736 halogen Inorganic materials 0.000 claims description 172
- 150000002367 halogens Chemical class 0.000 claims description 172
- 125000003545 alkoxy group Chemical group 0.000 claims description 133
- 229910052805 deuterium Inorganic materials 0.000 claims description 132
- YZCKVEUIGOORGS-OUBTZVSYSA-N Deuterium Chemical compound [2H] YZCKVEUIGOORGS-OUBTZVSYSA-N 0.000 claims description 131
- 150000001875 compounds Chemical class 0.000 claims description 111
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 91
- 150000003839 salts Chemical class 0.000 claims description 81
- 229910052739 hydrogen Inorganic materials 0.000 claims description 76
- 239000001257 hydrogen Substances 0.000 claims description 76
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 71
- 150000002431 hydrogen Chemical class 0.000 claims description 64
- 125000000592 heterocycloalkyl group Chemical group 0.000 claims description 55
- 125000004432 carbon atom Chemical group C* 0.000 claims description 51
- 125000000753 cycloalkyl group Chemical group 0.000 claims description 45
- -1 nitro, hydroxy Chemical group 0.000 claims description 44
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 37
- 125000005843 halogen group Chemical group 0.000 claims description 24
- 125000006272 (C3-C7) cycloalkyl group Chemical group 0.000 claims description 22
- 125000001072 heteroaryl group Chemical group 0.000 claims description 20
- 125000003118 aryl group Chemical group 0.000 claims description 17
- CZFFBEXEKNGXKS-UHFFFAOYSA-N raltegravir Chemical compound O1C(C)=NN=C1C(=O)NC(C)(C)C1=NC(C(=O)NCC=2C=CC(F)=CC=2)=C(O)C(=O)N1C CZFFBEXEKNGXKS-UHFFFAOYSA-N 0.000 claims description 17
- 229960004742 raltegravir Drugs 0.000 claims description 17
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 15
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 13
- 230000035772 mutation Effects 0.000 claims description 12
- 229960003586 elvitegravir Drugs 0.000 claims description 11
- JUZYLCPPVHEVSV-LJQANCHMSA-N elvitegravir Chemical compound COC1=CC=2N([C@H](CO)C(C)C)C=C(C(O)=O)C(=O)C=2C=C1CC1=CC=CC(Cl)=C1F JUZYLCPPVHEVSV-LJQANCHMSA-N 0.000 claims description 11
- 229940124524 integrase inhibitor Drugs 0.000 claims description 11
- 239000002850 integrase inhibitor Substances 0.000 claims description 11
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 10
- 208000031886 HIV Infections Diseases 0.000 claims description 9
- 208000037357 HIV infectious disease Diseases 0.000 claims description 9
- 229960002542 dolutegravir Drugs 0.000 claims description 9
- RHWKPHLQXYSBKR-BMIGLBTASA-N dolutegravir Chemical compound C([C@@H]1OCC[C@H](N1C(=O)C1=C(O)C2=O)C)N1C=C2C(=O)NCC1=CC=C(F)C=C1F RHWKPHLQXYSBKR-BMIGLBTASA-N 0.000 claims description 9
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 9
- 208000033519 human immunodeficiency virus infectious disease Diseases 0.000 claims description 9
- 238000002360 preparation method Methods 0.000 claims description 9
- HBOMLICNUCNMMY-XLPZGREQSA-N zidovudine Chemical compound O=C1NC(=O)C(C)=CN1[C@@H]1O[C@H](CO)[C@@H](N=[N+]=[N-])C1 HBOMLICNUCNMMY-XLPZGREQSA-N 0.000 claims description 8
- 229950004159 bictegravir Drugs 0.000 claims description 7
- SOLUWJRYJLAZCX-LYOVBCGYSA-N bictegravir Chemical compound C([C@H]1O[C@@H]2CC[C@@H](C2)N1C(=O)C1=C(C2=O)O)N1C=C2C(=O)NCC1=C(F)C=C(F)C=C1F SOLUWJRYJLAZCX-LYOVBCGYSA-N 0.000 claims description 7
- 229940124321 AIDS medicine Drugs 0.000 claims description 6
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 6
- NQDJXKOVJZTUJA-UHFFFAOYSA-N nevirapine Chemical compound C12=NC=CC=C2C(=O)NC=2C(C)=CC=NC=2N1C1CC1 NQDJXKOVJZTUJA-UHFFFAOYSA-N 0.000 claims description 6
- 150000002825 nitriles Chemical class 0.000 claims description 6
- 108010002459 HIV Integrase Proteins 0.000 claims description 5
- 125000000623 heterocyclic group Chemical group 0.000 claims description 5
- 108090000623 proteins and genes Proteins 0.000 claims description 5
- 102220076678 rs146651027 Human genes 0.000 claims description 5
- 229960002555 zidovudine Drugs 0.000 claims description 5
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 claims description 4
- 125000006582 (C5-C6) heterocycloalkyl group Chemical group 0.000 claims description 4
- AXRYRYVKAWYZBR-UHFFFAOYSA-N Atazanavir Natural products C=1C=C(C=2N=CC=CC=2)C=CC=1CN(NC(=O)C(NC(=O)OC)C(C)(C)C)CC(O)C(NC(=O)C(NC(=O)OC)C(C)(C)C)CC1=CC=CC=C1 AXRYRYVKAWYZBR-UHFFFAOYSA-N 0.000 claims description 4
- 108010019625 Atazanavir Sulfate Proteins 0.000 claims description 4
- QAGYKUNXZHXKMR-UHFFFAOYSA-N CPD000469186 Natural products CC1=C(O)C=CC=C1C(=O)NC(C(O)CN1C(CC2CCCCC2C1)C(=O)NC(C)(C)C)CSC1=CC=CC=C1 QAGYKUNXZHXKMR-UHFFFAOYSA-N 0.000 claims description 4
- KJHKTHWMRKYKJE-SUGCFTRWSA-N Kaletra Chemical compound N1([C@@H](C(C)C)C(=O)N[C@H](C[C@H](O)[C@H](CC=2C=CC=CC=2)NC(=O)COC=2C(=CC=CC=2C)C)CC=2C=CC=CC=2)CCCNC1=O KJHKTHWMRKYKJE-SUGCFTRWSA-N 0.000 claims description 4
- 229940122313 Nucleoside reverse transcriptase inhibitor Drugs 0.000 claims description 4
- NCDNCNXCDXHOMX-UHFFFAOYSA-N Ritonavir Natural products C=1C=CC=CC=1CC(NC(=O)OCC=1SC=NC=1)C(O)CC(CC=1C=CC=CC=1)NC(=O)C(C(C)C)NC(=O)N(C)CC1=CSC(C(C)C)=N1 NCDNCNXCDXHOMX-UHFFFAOYSA-N 0.000 claims description 4
- SUJUHGSWHZTSEU-UHFFFAOYSA-N Tipranavir Natural products C1C(O)=C(C(CC)C=2C=C(NS(=O)(=O)C=3N=CC(=CC=3)C(F)(F)F)C=CC=2)C(=O)OC1(CCC)CCC1=CC=CC=C1 SUJUHGSWHZTSEU-UHFFFAOYSA-N 0.000 claims description 4
- 229960001830 amprenavir Drugs 0.000 claims description 4
- YMARZQAQMVYCKC-OEMFJLHTSA-N amprenavir Chemical compound C([C@@H]([C@H](O)CN(CC(C)C)S(=O)(=O)C=1C=CC(N)=CC=1)NC(=O)O[C@@H]1COCC1)C1=CC=CC=C1 YMARZQAQMVYCKC-OEMFJLHTSA-N 0.000 claims description 4
- 229960003277 atazanavir Drugs 0.000 claims description 4
- AXRYRYVKAWYZBR-GASGPIRDSA-N atazanavir Chemical compound C([C@H](NC(=O)[C@@H](NC(=O)OC)C(C)(C)C)[C@@H](O)CN(CC=1C=CC(=CC=1)C=1N=CC=CC=1)NC(=O)[C@@H](NC(=O)OC)C(C)(C)C)C1=CC=CC=C1 AXRYRYVKAWYZBR-GASGPIRDSA-N 0.000 claims description 4
- 229960005107 darunavir Drugs 0.000 claims description 4
- CJBJHOAVZSMMDJ-HEXNFIEUSA-N darunavir Chemical compound C([C@@H]([C@H](O)CN(CC(C)C)S(=O)(=O)C=1C=CC(N)=CC=1)NC(=O)O[C@@H]1[C@@H]2CCO[C@@H]2OC1)C1=CC=CC=C1 CJBJHOAVZSMMDJ-HEXNFIEUSA-N 0.000 claims description 4
- 230000007547 defect Effects 0.000 claims description 4
- 229960003142 fosamprenavir Drugs 0.000 claims description 4
- MLBVMOWEQCZNCC-OEMFJLHTSA-N fosamprenavir Chemical compound C([C@@H]([C@H](OP(O)(O)=O)CN(CC(C)C)S(=O)(=O)C=1C=CC(N)=CC=1)NC(=O)O[C@@H]1COCC1)C1=CC=CC=C1 MLBVMOWEQCZNCC-OEMFJLHTSA-N 0.000 claims description 4
- 229960001936 indinavir Drugs 0.000 claims description 4
- CBVCZFGXHXORBI-PXQQMZJSSA-N indinavir Chemical compound C([C@H](N(CC1)C[C@@H](O)C[C@@H](CC=2C=CC=CC=2)C(=O)N[C@H]2C3=CC=CC=C3C[C@H]2O)C(=O)NC(C)(C)C)N1CC1=CC=CN=C1 CBVCZFGXHXORBI-PXQQMZJSSA-N 0.000 claims description 4
- 229960004525 lopinavir Drugs 0.000 claims description 4
- 229960000884 nelfinavir Drugs 0.000 claims description 4
- QAGYKUNXZHXKMR-HKWSIXNMSA-N nelfinavir Chemical compound CC1=C(O)C=CC=C1C(=O)N[C@H]([C@H](O)CN1[C@@H](C[C@@H]2CCCC[C@@H]2C1)C(=O)NC(C)(C)C)CSC1=CC=CC=C1 QAGYKUNXZHXKMR-HKWSIXNMSA-N 0.000 claims description 4
- 229940042402 non-nucleoside reverse transcriptase inhibitor Drugs 0.000 claims description 4
- 239000002726 nonnucleoside reverse transcriptase inhibitor Substances 0.000 claims description 4
- 229960000311 ritonavir Drugs 0.000 claims description 4
- NCDNCNXCDXHOMX-XGKFQTDJSA-N ritonavir Chemical compound N([C@@H](C(C)C)C(=O)N[C@H](C[C@H](O)[C@H](CC=1C=CC=CC=1)NC(=O)OCC=1SC=NC=1)CC=1C=CC=CC=1)C(=O)N(C)CC1=CSC(C(C)C)=N1 NCDNCNXCDXHOMX-XGKFQTDJSA-N 0.000 claims description 4
- 239000003419 rna directed dna polymerase inhibitor Substances 0.000 claims description 4
- 229960001852 saquinavir Drugs 0.000 claims description 4
- QWAXKHKRTORLEM-UGJKXSETSA-N saquinavir Chemical compound C([C@@H]([C@H](O)CN1C[C@H]2CCCC[C@H]2C[C@H]1C(=O)NC(C)(C)C)NC(=O)[C@H](CC(N)=O)NC(=O)C=1N=C2C=CC=CC2=CC=1)C1=CC=CC=C1 QWAXKHKRTORLEM-UGJKXSETSA-N 0.000 claims description 4
- 229960000838 tipranavir Drugs 0.000 claims description 4
- SUJUHGSWHZTSEU-FYBSXPHGSA-N tipranavir Chemical compound C([C@@]1(CCC)OC(=O)C([C@H](CC)C=2C=C(NS(=O)(=O)C=3N=CC(=CC=3)C(F)(F)F)C=CC=2)=C(O)C1)CC1=CC=CC=C1 SUJUHGSWHZTSEU-FYBSXPHGSA-N 0.000 claims description 4
- ZIAOVIPSKUPPQW-UHFFFAOYSA-N 3-chloro-5-[1-[(4-methyl-5-oxo-1h-1,2,4-triazol-3-yl)methyl]-2-oxo-4-(trifluoromethyl)pyridin-3-yl]oxybenzonitrile Chemical compound N1C(=O)N(C)C(CN2C(C(OC=3C=C(C=C(Cl)C=3)C#N)=C(C=C2)C(F)(F)F)=O)=N1 ZIAOVIPSKUPPQW-UHFFFAOYSA-N 0.000 claims description 3
- XPOQHMRABVBWPR-UHFFFAOYSA-N Efavirenz Natural products O1C(=O)NC2=CC=C(Cl)C=C2C1(C(F)(F)F)C#CC1CC1 XPOQHMRABVBWPR-UHFFFAOYSA-N 0.000 claims description 3
- XQSPYNMVSIKCOC-NTSWFWBYSA-N Emtricitabine Chemical compound C1=C(F)C(N)=NC(=O)N1[C@H]1O[C@@H](CO)SC1 XQSPYNMVSIKCOC-NTSWFWBYSA-N 0.000 claims description 3
- 108010032976 Enfuvirtide Proteins 0.000 claims description 3
- WREGKURFCTUGRC-POYBYMJQSA-N Zalcitabine Chemical compound O=C1N=C(N)C=CN1[C@@H]1O[C@H](CO)CC1 WREGKURFCTUGRC-POYBYMJQSA-N 0.000 claims description 3
- 229960004748 abacavir Drugs 0.000 claims description 3
- MCGSCOLBFJQGHM-SCZZXKLOSA-N abacavir Chemical compound C=12N=CN([C@H]3C=C[C@@H](CO)C3)C2=NC(N)=NC=1NC1CC1 MCGSCOLBFJQGHM-SCZZXKLOSA-N 0.000 claims description 3
- 150000001335 aliphatic alkanes Chemical group 0.000 claims description 3
- 229940042399 direct acting antivirals protease inhibitors Drugs 0.000 claims description 3
- 229950003141 doravirine Drugs 0.000 claims description 3
- 229960003804 efavirenz Drugs 0.000 claims description 3
- XPOQHMRABVBWPR-ZDUSSCGKSA-N efavirenz Chemical compound C([C@]1(C2=CC(Cl)=CC=C2NC(=O)O1)C(F)(F)F)#CC1CC1 XPOQHMRABVBWPR-ZDUSSCGKSA-N 0.000 claims description 3
- 229960000366 emtricitabine Drugs 0.000 claims description 3
- PEASPLKKXBYDKL-FXEVSJAOSA-N enfuvirtide Chemical compound C([C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](C)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC=1C=CC=CC=1)C(N)=O)NC(=O)[C@@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(C)=O)[C@@H](C)O)[C@@H](C)CC)C1=CN=CN1 PEASPLKKXBYDKL-FXEVSJAOSA-N 0.000 claims description 3
- 229960002062 enfuvirtide Drugs 0.000 claims description 3
- 229960001627 lamivudine Drugs 0.000 claims description 3
- JTEGQNOMFQHVDC-NKWVEPMBSA-N lamivudine Chemical compound O=C1N=C(N)C=CN1[C@H]1O[C@@H](CO)SC1 JTEGQNOMFQHVDC-NKWVEPMBSA-N 0.000 claims description 3
- 229960000689 nevirapine Drugs 0.000 claims description 3
- 239000000137 peptide hydrolase inhibitor Substances 0.000 claims description 3
- 229940111527 trizivir Drugs 0.000 claims description 3
- 230000009385 viral infection Effects 0.000 claims description 3
- 229960000523 zalcitabine Drugs 0.000 claims description 3
- ONIKNECPXCLUHT-UHFFFAOYSA-N 2-chlorobenzoyl chloride Chemical compound ClC(=O)C1=CC=CC=C1Cl ONIKNECPXCLUHT-UHFFFAOYSA-N 0.000 claims description 2
- 125000004070 6 membered heterocyclic group Chemical group 0.000 claims description 2
- XNKLLVCARDGLGL-JGVFFNPUSA-N Stavudine Chemical compound O=C1NC(=O)C(C)=CN1[C@H]1C=C[C@@H](CO)O1 XNKLLVCARDGLGL-JGVFFNPUSA-N 0.000 claims description 2
- 239000002259 anti human immunodeficiency virus agent Substances 0.000 claims description 2
- 125000006297 carbonyl amino group Chemical group [H]N([*:2])C([*:1])=O 0.000 claims description 2
- 238000012217 deletion Methods 0.000 claims description 2
- 230000037430 deletion Effects 0.000 claims description 2
- 125000004216 fluoromethyl group Chemical group [H]C([H])(F)* 0.000 claims description 2
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 2
- 229960002814 rilpivirine Drugs 0.000 claims description 2
- YIBOMRUWOWDFLG-ONEGZZNKSA-N rilpivirine Chemical compound CC1=CC(\C=C\C#N)=CC(C)=C1NC1=CC=NC(NC=2C=CC(=CC=2)C#N)=N1 YIBOMRUWOWDFLG-ONEGZZNKSA-N 0.000 claims description 2
- 229960001203 stavudine Drugs 0.000 claims description 2
- 108700026220 vif Genes Proteins 0.000 claims description 2
- 229950005928 cabotegravir Drugs 0.000 claims 1
- WCWSTNLSLKSJPK-LKFCYVNXSA-N cabotegravir Chemical compound C([C@H]1OC[C@@H](N1C(=O)C1=C(O)C2=O)C)N1C=C2C(=O)NCC1=CC=C(F)C=C1F WCWSTNLSLKSJPK-LKFCYVNXSA-N 0.000 claims 1
- 150000001924 cycloalkanes Chemical class 0.000 claims 1
- 125000001153 fluoro group Chemical group F* 0.000 claims 1
- 208000010648 susceptibility to HIV infection Diseases 0.000 claims 1
- 238000011282 treatment Methods 0.000 abstract description 7
- 201000010099 disease Diseases 0.000 abstract description 3
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 abstract description 3
- 239000013543 active substance Substances 0.000 abstract 2
- 238000006243 chemical reaction Methods 0.000 description 30
- 239000000243 solution Substances 0.000 description 14
- 238000004949 mass spectrometry Methods 0.000 description 13
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 12
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 10
- 239000012043 crude product Substances 0.000 description 10
- 125000001424 substituent group Chemical group 0.000 description 10
- 239000000203 mixture Substances 0.000 description 8
- 102100034343 Integrase Human genes 0.000 description 7
- 108010061833 Integrases Proteins 0.000 description 7
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 6
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 6
- 206010059866 Drug resistance Diseases 0.000 description 6
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 6
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 6
- 229910052757 nitrogen Inorganic materials 0.000 description 6
- 239000000741 silica gel Substances 0.000 description 6
- 229910002027 silica gel Inorganic materials 0.000 description 6
- 238000004809 thin layer chromatography Methods 0.000 description 6
- 238000005481 NMR spectroscopy Methods 0.000 description 5
- 229910052799 carbon Inorganic materials 0.000 description 5
- 125000006413 ring segment Chemical group 0.000 description 5
- 125000004191 (C1-C6) alkoxy group Chemical group 0.000 description 4
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 4
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 4
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 4
- 125000004429 atom Chemical group 0.000 description 4
- 239000003153 chemical reaction reagent Substances 0.000 description 4
- 230000000694 effects Effects 0.000 description 4
- 239000011737 fluorine Substances 0.000 description 4
- 229910052731 fluorine Inorganic materials 0.000 description 4
- 125000005842 heteroatom Chemical group 0.000 description 4
- 238000000034 method Methods 0.000 description 4
- 239000000126 substance Substances 0.000 description 4
- 230000003612 virological effect Effects 0.000 description 4
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 4
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 3
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical group [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 3
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 3
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 description 3
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 3
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 3
- 239000000460 chlorine Substances 0.000 description 3
- 238000007796 conventional method Methods 0.000 description 3
- 239000003112 inhibitor Substances 0.000 description 3
- 230000010354 integration Effects 0.000 description 3
- 125000002950 monocyclic group Chemical group 0.000 description 3
- 239000012299 nitrogen atmosphere Substances 0.000 description 3
- 229910052760 oxygen Inorganic materials 0.000 description 3
- 239000001301 oxygen Substances 0.000 description 3
- 125000003367 polycyclic group Chemical group 0.000 description 3
- 238000000746 purification Methods 0.000 description 3
- 239000007858 starting material Substances 0.000 description 3
- 238000012360 testing method Methods 0.000 description 3
- KRIWIRSMQRQYJG-DLBZAZTESA-N (2s,3s)-3-[[7-(benzylamino)-3-propan-2-ylpyrazolo[1,5-a]pyrimidin-5-yl]amino]butane-1,2,4-triol Chemical compound C=1C(N[C@@H](CO)[C@H](O)CO)=NC2=C(C(C)C)C=NN2C=1NCC1=CC=CC=C1 KRIWIRSMQRQYJG-DLBZAZTESA-N 0.000 description 2
- OMBVEVHRIQULKW-DNQXCXABSA-M (3r,5r)-7-[3-(4-fluorophenyl)-8-oxo-7-phenyl-1-propan-2-yl-5,6-dihydro-4h-pyrrolo[2,3-c]azepin-2-yl]-3,5-dihydroxyheptanoate Chemical compound O=C1C=2N(C(C)C)C(CC[C@@H](O)C[C@@H](O)CC([O-])=O)=C(C=3C=CC(F)=CC=3)C=2CCCN1C1=CC=CC=C1 OMBVEVHRIQULKW-DNQXCXABSA-M 0.000 description 2
- PNHBRYIAJCYNDA-VQCQRNETSA-N (4r)-6-[2-[2-ethyl-4-(4-fluorophenyl)-6-phenylpyridin-3-yl]ethyl]-4-hydroxyoxan-2-one Chemical compound C([C@H](O)C1)C(=O)OC1CCC=1C(CC)=NC(C=2C=CC=CC=2)=CC=1C1=CC=C(F)C=C1 PNHBRYIAJCYNDA-VQCQRNETSA-N 0.000 description 2
- QVBVQHTXLPNXEY-ZMFCMNQTSA-N (4r)-6-[2-[4-(4-fluorophenyl)-6-phenyl-2-propan-2-ylpyridin-3-yl]ethyl]-4-hydroxyoxan-2-one Chemical compound C([C@H](O)C1)C(=O)OC1CCC=1C(C(C)C)=NC(C=2C=CC=CC=2)=CC=1C1=CC=C(F)C=C1 QVBVQHTXLPNXEY-ZMFCMNQTSA-N 0.000 description 2
- QOLHWXNSCZGWHK-BWBORTOCSA-N (6r,7r)-1-[(4s,5r)-4-acetyloxy-5-methyl-3-methylidene-6-phenylhexyl]-4,7-dihydroxy-6-(11-phenoxyundecylcarbamoyloxy)-2,8-dioxabicyclo[3.2.1]octane-3,4,5-tricarboxylic acid Chemical compound C([C@@H](C)[C@H](OC(C)=O)C(=C)CCC12[C@@H]([C@@H](OC(=O)NCCCCCCCCCCCOC=3C=CC=CC=3)C(O1)(C(O)=O)C(O)(C(O2)C(O)=O)C(O)=O)O)C1=CC=CC=C1 QOLHWXNSCZGWHK-BWBORTOCSA-N 0.000 description 2
- MOQCFMZWVKQBAP-UHFFFAOYSA-N 1-[3,5-bis(trifluoromethyl)benzoyl]-n-(4-chlorophenyl)piperidine-3-carboxamide Chemical compound FC(F)(F)C1=CC(C(F)(F)F)=CC(C(=O)N2CC(CCC2)C(=O)NC=2C=CC(Cl)=CC=2)=C1 MOQCFMZWVKQBAP-UHFFFAOYSA-N 0.000 description 2
- YLLRPQWLASQXSI-UHFFFAOYSA-N 3-(4b,8a,9,9a-tetrahydro-4aH-pyrido[2,3-b]indol-4-ylamino)phenol Chemical compound Oc1cccc(NC2=CC=NC3NC4C=CC=CC4C23)c1 YLLRPQWLASQXSI-UHFFFAOYSA-N 0.000 description 2
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 2
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 2
- 108091026890 Coding region Proteins 0.000 description 2
- 229940126559 Compound 4e Drugs 0.000 description 2
- 206010013710 Drug interaction Diseases 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 2
- UFWIBTONFRDIAS-UHFFFAOYSA-N Naphthalene Chemical compound C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 2
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 2
- 108091005804 Peptidases Proteins 0.000 description 2
- 239000004365 Protease Substances 0.000 description 2
- KYQCOXFCLRTKLS-UHFFFAOYSA-N Pyrazine Chemical compound C1=CN=CC=N1 KYQCOXFCLRTKLS-UHFFFAOYSA-N 0.000 description 2
- 102100037486 Reverse transcriptase/ribonuclease H Human genes 0.000 description 2
- WQDUMFSSJAZKTM-UHFFFAOYSA-N Sodium methoxide Chemical compound [Na+].[O-]C WQDUMFSSJAZKTM-UHFFFAOYSA-N 0.000 description 2
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 2
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 2
- 208000036142 Viral infection Diseases 0.000 description 2
- 239000012298 atmosphere Substances 0.000 description 2
- 239000004202 carbamide Substances 0.000 description 2
- 239000012069 chiral reagent Substances 0.000 description 2
- 229910052801 chlorine Inorganic materials 0.000 description 2
- 238000004587 chromatography analysis Methods 0.000 description 2
- 229940126540 compound 41 Drugs 0.000 description 2
- 229940125872 compound 4d Drugs 0.000 description 2
- 229940126115 compound 4f Drugs 0.000 description 2
- 229940125880 compound 4j Drugs 0.000 description 2
- 238000001212 derivatisation Methods 0.000 description 2
- 125000000524 functional group Chemical group 0.000 description 2
- 239000011259 mixed solution Substances 0.000 description 2
- 230000001566 pro-viral effect Effects 0.000 description 2
- 125000004076 pyridyl group Chemical group 0.000 description 2
- 229920006395 saturated elastomer Polymers 0.000 description 2
- 238000000926 separation method Methods 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 125000003003 spiro group Chemical group 0.000 description 2
- 238000006467 substitution reaction Methods 0.000 description 2
- 229910052717 sulfur Inorganic materials 0.000 description 2
- 239000011593 sulfur Substances 0.000 description 2
- DYHSDKLCOJIUFX-UHFFFAOYSA-N tert-butoxycarbonyl anhydride Chemical compound CC(C)(C)OC(=O)OC(=O)OC(C)(C)C DYHSDKLCOJIUFX-UHFFFAOYSA-N 0.000 description 2
- QDZZDVQGBKTLHV-UHFFFAOYSA-N (2,4-difluorophenyl)methanamine Chemical compound NCC1=CC=C(F)C=C1F QDZZDVQGBKTLHV-UHFFFAOYSA-N 0.000 description 1
- GHYOCDFICYLMRF-UTIIJYGPSA-N (2S,3R)-N-[(2S)-3-(cyclopenten-1-yl)-1-[(2R)-2-methyloxiran-2-yl]-1-oxopropan-2-yl]-3-hydroxy-3-(4-methoxyphenyl)-2-[[(2S)-2-[(2-morpholin-4-ylacetyl)amino]propanoyl]amino]propanamide Chemical compound C1(=CCCC1)C[C@@H](C(=O)[C@@]1(OC1)C)NC([C@H]([C@@H](C1=CC=C(C=C1)OC)O)NC([C@H](C)NC(CN1CCOCC1)=O)=O)=O GHYOCDFICYLMRF-UTIIJYGPSA-N 0.000 description 1
- QFLWZFQWSBQYPS-AWRAUJHKSA-N (3S)-3-[[(2S)-2-[[(2S)-2-[5-[(3aS,6aR)-2-oxo-1,3,3a,4,6,6a-hexahydrothieno[3,4-d]imidazol-4-yl]pentanoylamino]-3-methylbutanoyl]amino]-3-(4-hydroxyphenyl)propanoyl]amino]-4-[1-bis(4-chlorophenoxy)phosphorylbutylamino]-4-oxobutanoic acid Chemical compound CCCC(NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@@H](NC(=O)CCCCC1SC[C@@H]2NC(=O)N[C@H]12)C(C)C)P(=O)(Oc1ccc(Cl)cc1)Oc1ccc(Cl)cc1 QFLWZFQWSBQYPS-AWRAUJHKSA-N 0.000 description 1
- VIMMECPCYZXUCI-MIMFYIINSA-N (4s,6r)-6-[(1e)-4,4-bis(4-fluorophenyl)-3-(1-methyltetrazol-5-yl)buta-1,3-dienyl]-4-hydroxyoxan-2-one Chemical compound CN1N=NN=C1C(\C=C\[C@@H]1OC(=O)C[C@@H](O)C1)=C(C=1C=CC(F)=CC=1)C1=CC=C(F)C=C1 VIMMECPCYZXUCI-MIMFYIINSA-N 0.000 description 1
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 description 1
- 125000006507 2,4-difluorobenzyl group Chemical group [H]C1=C(F)C([H])=C(F)C(=C1[H])C([H])([H])* 0.000 description 1
- SOLCGTLCIBOSQW-UHFFFAOYSA-N 2-acetyloxyethylazanium;chloride Chemical compound Cl.CC(=O)OCCN SOLCGTLCIBOSQW-UHFFFAOYSA-N 0.000 description 1
- 125000003542 3-methylbutan-2-yl group Chemical group [H]C([H])([H])C([H])(*)C([H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- 208000030507 AIDS Diseases 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 102100027211 Albumin Human genes 0.000 description 1
- 108010088751 Albumins Proteins 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- COTNUBDHGSIOTA-LHHVLQQYSA-N CO.[2H]OC([2H])([2H])[2H] Chemical class CO.[2H]OC([2H])([2H])[2H] COTNUBDHGSIOTA-LHHVLQQYSA-N 0.000 description 1
- VPTMFAQTRZTWOA-UHFFFAOYSA-N COC1=C(OC=C(C1=C=O)C(=O)OC)C(=O)OC Chemical compound COC1=C(OC=C(C1=C=O)C(=O)OC)C(=O)OC VPTMFAQTRZTWOA-UHFFFAOYSA-N 0.000 description 1
- 108020004414 DNA Proteins 0.000 description 1
- IAZDPXIOMUYVGZ-WFGJKAKNSA-N Dimethyl sulfoxide Chemical compound [2H]C([2H])([2H])S(=O)C([2H])([2H])[2H] IAZDPXIOMUYVGZ-WFGJKAKNSA-N 0.000 description 1
- 230000005526 G1 to G0 transition Effects 0.000 description 1
- 229940099797 HIV integrase inhibitor Drugs 0.000 description 1
- 101000801643 Homo sapiens Retinal-specific phospholipid-transporting ATPase ABCA4 Proteins 0.000 description 1
- 101000652482 Homo sapiens TBC1 domain family member 8 Proteins 0.000 description 1
- 241000713772 Human immunodeficiency virus 1 Species 0.000 description 1
- 102000012330 Integrases Human genes 0.000 description 1
- LOMVENUNSWAXEN-UHFFFAOYSA-N Methyl oxalate Chemical compound COC(=O)C(=O)OC LOMVENUNSWAXEN-UHFFFAOYSA-N 0.000 description 1
- ZSXGLVDWWRXATF-UHFFFAOYSA-N N,N-dimethylformamide dimethyl acetal Chemical compound COC(OC)N(C)C ZSXGLVDWWRXATF-UHFFFAOYSA-N 0.000 description 1
- 102000011931 Nucleoproteins Human genes 0.000 description 1
- 108010061100 Nucleoproteins Proteins 0.000 description 1
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 1
- PCNDJXKNXGMECE-UHFFFAOYSA-N Phenazine Natural products C1=CC=CC2=NC3=CC=CC=C3N=C21 PCNDJXKNXGMECE-UHFFFAOYSA-N 0.000 description 1
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 1
- 108010092799 RNA-directed DNA polymerase Proteins 0.000 description 1
- 102100033617 Retinal-specific phospholipid-transporting ATPase ABCA4 Human genes 0.000 description 1
- UIIMBOGNXHQVGW-DEQYMQKBSA-M Sodium bicarbonate-14C Chemical class [Na+].O[14C]([O-])=O UIIMBOGNXHQVGW-DEQYMQKBSA-M 0.000 description 1
- 102100030302 TBC1 domain family member 8 Human genes 0.000 description 1
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical class [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 1
- 241000009298 Trigla lyra Species 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 239000008186 active pharmaceutical agent Substances 0.000 description 1
- 229940050528 albumin Drugs 0.000 description 1
- 125000001931 aliphatic group Chemical group 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- VZTDIZULWFCMLS-UHFFFAOYSA-N ammonium formate Chemical compound [NH4+].[O-]C=O VZTDIZULWFCMLS-UHFFFAOYSA-N 0.000 description 1
- 230000003698 anagen phase Effects 0.000 description 1
- 230000000798 anti-retroviral effect Effects 0.000 description 1
- 230000000840 anti-viral effect Effects 0.000 description 1
- 239000004599 antimicrobial Substances 0.000 description 1
- 238000011225 antiretroviral therapy Methods 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 238000011914 asymmetric synthesis Methods 0.000 description 1
- 230000004888 barrier function Effects 0.000 description 1
- 238000012925 biological evaluation Methods 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- UORVGPXVDQYIDP-BJUDXGSMSA-N borane Chemical class [10BH3] UORVGPXVDQYIDP-BJUDXGSMSA-N 0.000 description 1
- UWTDFICHZKXYAC-UHFFFAOYSA-N boron;oxolane Chemical class [B].C1CCOC1 UWTDFICHZKXYAC-UHFFFAOYSA-N 0.000 description 1
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- 150000001721 carbon Chemical group 0.000 description 1
- 239000001569 carbon dioxide Substances 0.000 description 1
- 229910002092 carbon dioxide Inorganic materials 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- PBAYDYUZOSNJGU-UHFFFAOYSA-N chelidonic acid Natural products OC(=O)C1=CC(=O)C=C(C(O)=O)O1 PBAYDYUZOSNJGU-UHFFFAOYSA-N 0.000 description 1
- 238000002512 chemotherapy Methods 0.000 description 1
- 238000004440 column chromatography Methods 0.000 description 1
- 239000002299 complementary DNA Substances 0.000 description 1
- 229940125797 compound 12 Drugs 0.000 description 1
- 239000012141 concentrate Substances 0.000 description 1
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000003678 cyclohexadienyl group Chemical group C1(=CC=CCC1)* 0.000 description 1
- 125000000596 cyclohexenyl group Chemical group C1(=CCCCC1)* 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000002433 cyclopentenyl group Chemical group C1(=CCCC1)* 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- 150000001975 deuterium Chemical group 0.000 description 1
- 238000003745 diagnosis Methods 0.000 description 1
- HPNMFZURTQLUMO-UHFFFAOYSA-N diethylamine Chemical compound CCNCC HPNMFZURTQLUMO-UHFFFAOYSA-N 0.000 description 1
- 125000004852 dihydrofuranyl group Chemical group O1C(CC=C1)* 0.000 description 1
- 125000005047 dihydroimidazolyl group Chemical group N1(CNC=C1)* 0.000 description 1
- 125000005052 dihydropyrazolyl group Chemical group N1(NCC=C1)* 0.000 description 1
- 125000005054 dihydropyrrolyl group Chemical group [H]C1=C([H])C([H])([H])C([H])([H])N1* 0.000 description 1
- 231100000676 disease causative agent Toxicity 0.000 description 1
- MGPSIDGTLFKDEY-UHFFFAOYSA-N ethyl 5-oxohexanoate Chemical compound CCOC(=O)CCCC(C)=O MGPSIDGTLFKDEY-UHFFFAOYSA-N 0.000 description 1
- KFEZBDVRVGQKOV-UHFFFAOYSA-N ethyl 6-[(2-ethoxy-2-oxoethyl)amino]hexanoate Chemical compound CCOC(=O)CCCCCNCC(=O)OCC KFEZBDVRVGQKOV-UHFFFAOYSA-N 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 238000011049 filling Methods 0.000 description 1
- 125000002541 furyl group Chemical group 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 239000003084 hiv integrase inhibitor Substances 0.000 description 1
- 238000005984 hydrogenation reaction Methods 0.000 description 1
- 125000002632 imidazolidinyl group Chemical group 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- 210000000987 immune system Anatomy 0.000 description 1
- 238000010348 incorporation Methods 0.000 description 1
- 238000011534 incubation Methods 0.000 description 1
- 125000003392 indanyl group Chemical group C1(CCC2=CC=CC=C12)* 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 230000005764 inhibitory process Effects 0.000 description 1
- 238000003780 insertion Methods 0.000 description 1
- 230000037431 insertion Effects 0.000 description 1
- 239000000543 intermediate Substances 0.000 description 1
- PNDPGZBMCMUPRI-UHFFFAOYSA-N iodine Chemical compound II PNDPGZBMCMUPRI-UHFFFAOYSA-N 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- HVTICUPFWKNHNG-BJUDXGSMSA-N iodoethane Chemical class [11CH3]CI HVTICUPFWKNHNG-BJUDXGSMSA-N 0.000 description 1
- INQOMBQAUSQDDS-BJUDXGSMSA-N iodomethane Chemical class I[11CH3] INQOMBQAUSQDDS-BJUDXGSMSA-N 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 238000006317 isomerization reaction Methods 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 229940067606 lecithin Drugs 0.000 description 1
- 239000000787 lecithin Substances 0.000 description 1
- 235000010445 lecithin Nutrition 0.000 description 1
- OTCKOJUMXQWKQG-UHFFFAOYSA-L magnesium bromide Chemical compound [Mg+2].[Br-].[Br-] OTCKOJUMXQWKQG-UHFFFAOYSA-L 0.000 description 1
- 238000013508 migration Methods 0.000 description 1
- 230000005012 migration Effects 0.000 description 1
- 125000006578 monocyclic heterocycloalkyl group Chemical group 0.000 description 1
- 125000002757 morpholinyl group Chemical group 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 239000002773 nucleotide Substances 0.000 description 1
- 125000003729 nucleotide group Chemical group 0.000 description 1
- 150000002894 organic compounds Chemical class 0.000 description 1
- 125000002971 oxazolyl group Chemical group 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 239000011574 phosphorus Substances 0.000 description 1
- 125000004193 piperazinyl group Chemical group 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 125000003226 pyrazolyl group Chemical group 0.000 description 1
- 125000000714 pyrimidinyl group Chemical group 0.000 description 1
- 125000000719 pyrrolidinyl group Chemical group 0.000 description 1
- 125000000168 pyrrolyl group Chemical group 0.000 description 1
- 238000010791 quenching Methods 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 238000010992 reflux Methods 0.000 description 1
- 230000008439 repair process Effects 0.000 description 1
- 230000010076 replication Effects 0.000 description 1
- 230000001177 retroviral effect Effects 0.000 description 1
- 238000004366 reverse phase liquid chromatography Methods 0.000 description 1
- 238000010839 reverse transcription Methods 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- BEOOHQFXGBMRKU-UHFFFAOYSA-N sodium cyanoborohydride Chemical compound [Na+].[B-]C#N BEOOHQFXGBMRKU-UHFFFAOYSA-N 0.000 description 1
- MFRIHAYPQRLWNB-UHFFFAOYSA-N sodium tert-butoxide Chemical compound [Na+].CC(C)(C)[O-] MFRIHAYPQRLWNB-UHFFFAOYSA-N 0.000 description 1
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical class O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 238000010186 staining Methods 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 238000004808 supercritical fluid chromatography Methods 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 238000003419 tautomerization reaction Methods 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000001973 tert-pentyl group Chemical group [H]C([H])([H])C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 238000010998 test method Methods 0.000 description 1
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 1
- 125000003718 tetrahydrofuranyl group Chemical group 0.000 description 1
- 125000001712 tetrahydronaphthyl group Chemical group C1(CCCC2=CC=CC=C12)* 0.000 description 1
- 125000005958 tetrahydrothienyl group Chemical group 0.000 description 1
- CZDYPVPMEAXLPK-UHFFFAOYSA-N tetramethylsilane Chemical compound C[Si](C)(C)C CZDYPVPMEAXLPK-UHFFFAOYSA-N 0.000 description 1
- 125000003831 tetrazolyl group Chemical group 0.000 description 1
- VLLMWSRANPNYQX-UHFFFAOYSA-N thiadiazole Chemical compound C1=CSN=N1.C1=CSN=N1 VLLMWSRANPNYQX-UHFFFAOYSA-N 0.000 description 1
- 125000000335 thiazolyl group Chemical group 0.000 description 1
- 125000001544 thienyl group Chemical group 0.000 description 1
- 125000004568 thiomorpholinyl group Chemical group 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 238000011269 treatment regimen Methods 0.000 description 1
- 241001430294 unidentified retrovirus Species 0.000 description 1
- 244000052613 viral pathogen Species 0.000 description 1
- 239000003643 water by type Substances 0.000 description 1
- 239000008096 xylene Substances 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/4985—Pyrazines or piperazines ortho- or peri-condensed with heterocyclic ring systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/55—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/14—Antivirals for RNA viruses
- A61P31/18—Antivirals for RNA viruses for HIV
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- Medicinal Chemistry (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Virology (AREA)
- Oncology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- AIDS & HIV (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Tropical Medicine & Parasitology (AREA)
- Communicable Diseases (AREA)
- Molecular Biology (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
本揭露屬於醫藥領域,尤係關於一種抑制基因缺陷的HIV病毒的用途。 The present disclosure belongs to the field of medicine, and particularly relates to the use of a gene-deficient HIV virus.
HIV逆轉錄病毒是複雜性疾病的病原體,其逐步破壞免疫系統。逆轉錄病毒複製的特點包括病毒基因組逆轉錄到前病毒DNA中以及其整合到宿主細胞基因組中。其中,前病毒基因組整合到宿主細胞基因組中需要整合酶的作用,其以至少三個或四個步驟進行此過程。步驟一涉及將病毒基因組裝配到穩定的核蛋白複合物中,步驟二涉及加工來自基因組的3’末端的兩個核苷酸以產生具有自由3’OH殘基的交錯末端,以及步驟三涉及將這些末端轉移到宿主細胞基因組中,最後宿主基因組中插入位點的間隙充填和修復。 HIV retroviruses are causative agents of complex diseases that progressively destroy the immune system. Features of retroviral replication include reverse transcription of the viral genome into proviral DNA and its integration into the host cell genome. Among others, the integration of the proviral genome into the host cell genome requires the action of an integrase, which performs this process in at least three or four steps. Step one involves assembling the viral genome into a stable nucleoprotein complex, step two involves processing two nucleotides from the 3' end of the genome to create staggered ends with free 3'OH residues, and step three involves adding These ends are transferred into the host cell genome, and finally gap filling and repair of the insertion site in the host genome.
目前,可以用在市場上的許多抑制劑來治療HIV感染,其中上述抑制劑靶向逆轉錄酶、蛋白酶,或進入細胞。已知用這些藥物或這些藥物的組合來治療HIV感染可以有效治療AIDS和類似疾病。目前的抑制劑的缺點包括耐藥性和許多副作用的快速出現和發生率增加。 Currently, HIV infection can be treated with a number of inhibitors on the market that target reverse transcriptase, protease, or entry into cells. Treatment of HIV infection with these drugs or combinations of these drugs is known to be effective in the treatment of AIDS and similar diseases. Disadvantages of current inhibitors include drug resistance and the rapid onset and increased incidence of many side effects.
已知在野生型病毒整合酶中的某些突變會賦予對已知整合抑制劑的抗性。例如,在整合酶中包含Q148H/G140S雙突變以及在整合酶中包含N155H/E92Q雙突變的病毒變體為代表的患者中,雷特格韋(Raltegravir)或MK-0518治療無效(參見WO2010000032)。這導致患者的HIV感染治療策略複雜,而且HIV感染患者可能因患有其他病症而需要接受其他藥物治療,藥物的相互作用會導致抗逆轉錄病毒治療的評價標準失效。需要開發更為有效的、降低藥物相互作用的抗逆轉錄病毒的方法。 Certain mutations in wild-type viral integrases are known to confer resistance to known inhibitors of integration. For example, treatment with Raltegravir or MK-0518 was ineffective in patients represented by viral variants containing the Q148H/G140S double mutation in the integrase and N155H/E92Q double mutations in the integrase (see WO2010000032) . This complicates treatment strategies for patients with HIV infection, and HIV-infected patients may require additional drug treatment due to other medical conditions, and drug interactions can render the evaluation criteria for antiretroviral therapy invalid. There is a need to develop more effective antiretroviral methods with reduced drug interactions.
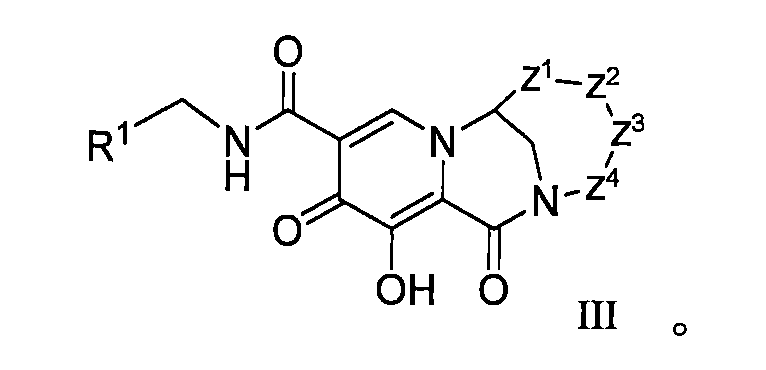
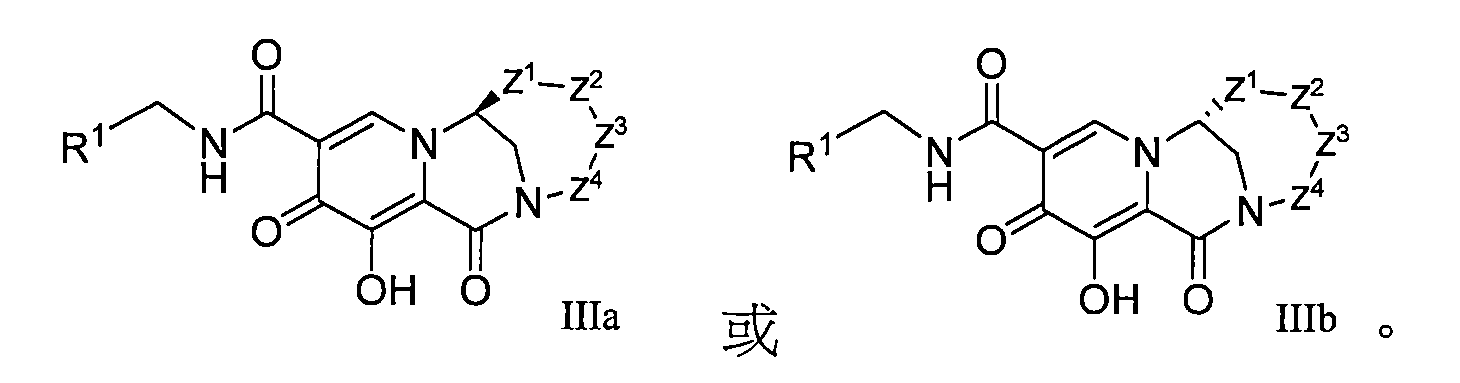
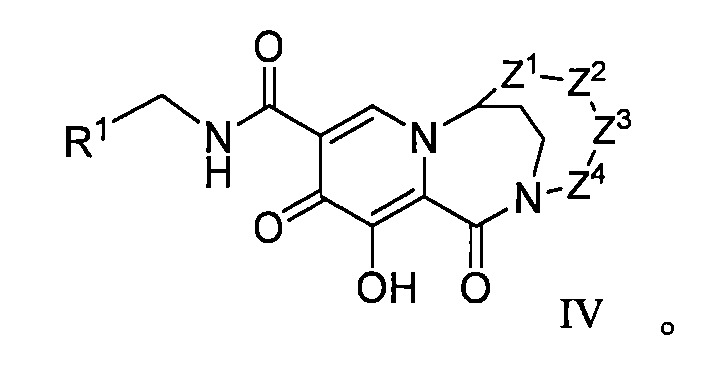
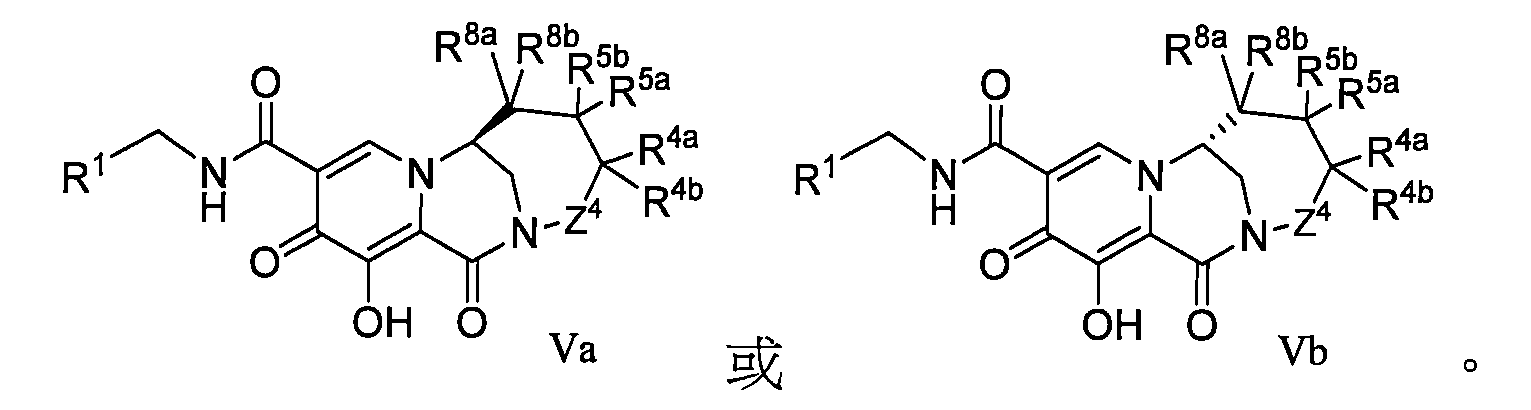
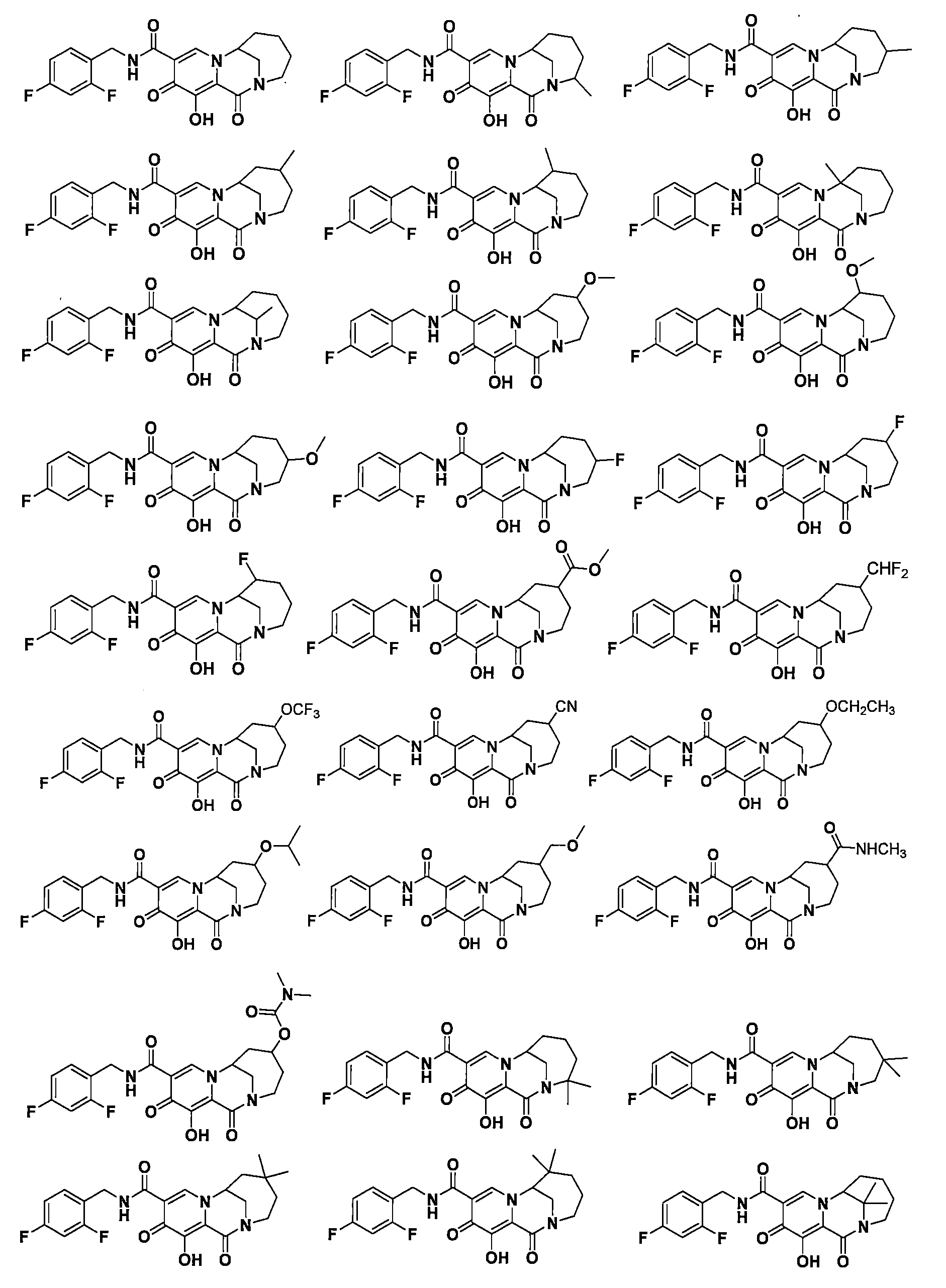
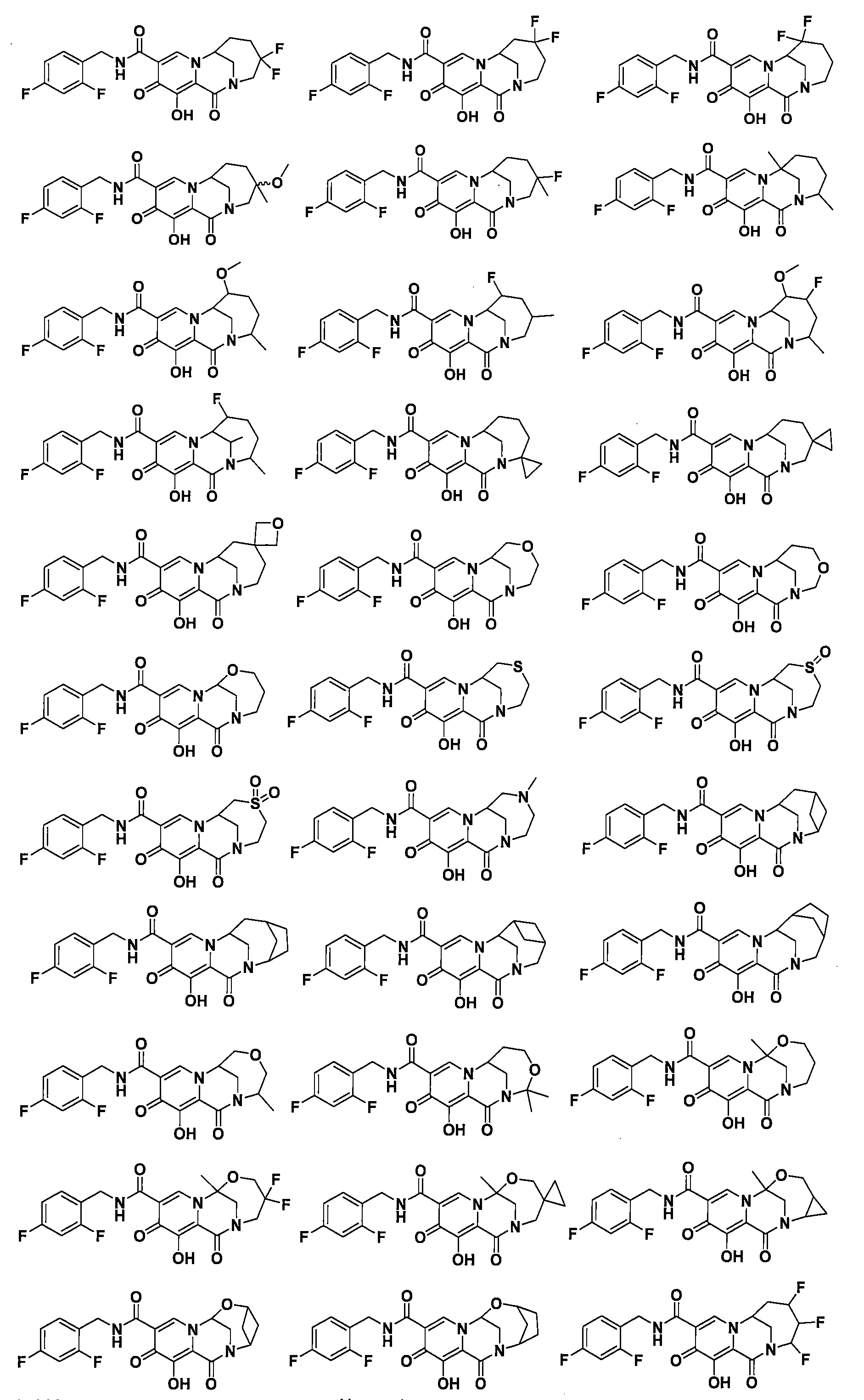
WO2020197991和WO2020221294描述了一類HIV整合酶抑制劑化合物,本揭露發明人發現這些子類化合物在整合酶包含突變位點的病毒病體表現出優異的耐藥性能,預期可用於對某些藥物產生耐藥的患者的治療,進一步改善或提高HIV患者生存質量。 WO2020197991 and WO2020221294 describe a class of HIV integrase inhibitor compounds, the inventors of the present disclosure have found that these subclasses of compounds show excellent drug resistance in viral pathogens containing integrase mutation sites, and are expected to be used to develop resistance to certain drugs The treatment of patients with HIV can further improve or improve the quality of life of HIV patients.
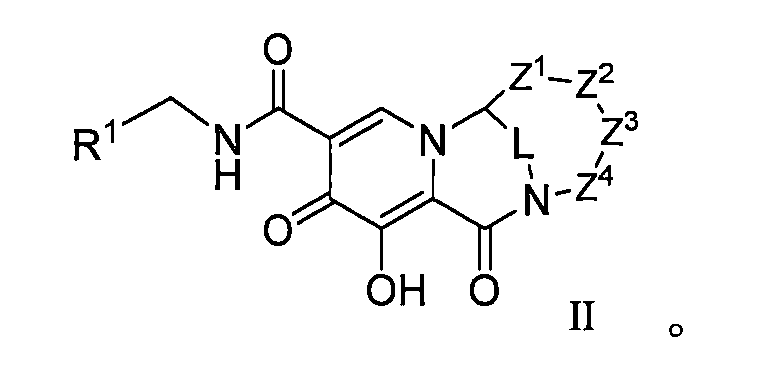
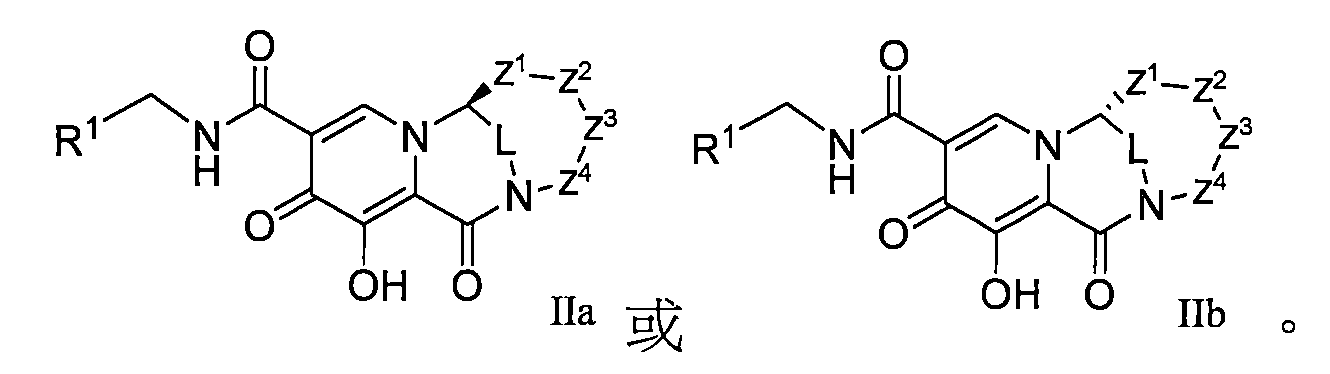
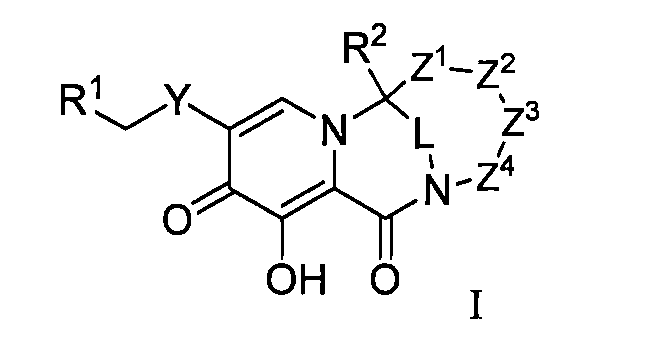
本揭露(The disclosure)提供了式I所示化合物或其可藥用鹽在製備用於治療或者預防HIV病毒感染的藥物中的用途,該HIV病毒包括N155H、E92Q、G140S、Q148H、Y143H、Y143C中的一種或多種基因中具有缺陷, The disclosure provides the use of a compound represented by formula I or a pharmaceutically acceptable salt thereof in the preparation of a medicament for the treatment or prevention of HIV infection, the HIV virus including N155H, E92Q, G140S, Q148H, Y143H, Y143C have a defect in one or more of the genes,
其中,R1選自氫或C6-10芳基,該芳基視需要被一個或多個RA1所取代,RA1選自氘、鹵素、硝基、氰基、C1-6烷基、C1-6烷氧基,該烷基或烷氧基視需要被鹵素、硝基、氰基或C1-6烷氧基所取代; Wherein, R 1 is selected from hydrogen or C 6-10 aryl, the aryl is optionally substituted by one or more R A1 , R A1 is selected from deuterium, halogen, nitro, cyano, C 1-6 alkyl , C 1-6 alkoxy, the alkyl or alkoxy is optionally substituted by halogen, nitro, cyano or C 1-6 alkoxy;
R2選自氫、氘、鹵素或烷基,該烷基視需要被一個或多個硝基、腈基、羥基或鹵素所取代; R 2 is selected from hydrogen, deuterium, halogen or alkyl optionally substituted by one or more nitro, nitrile, hydroxyl or halogen;
L選自-CR3aR3b-、-C(O)-、-SO2-、-CH2-CH2-或-N(Ra)-; L is selected from -CR 3a R 3b -, -C(O)-, -SO 2 -, -CH 2 -CH 2 - or -N(R a )-;
Z3選自鍵或-CR4aR4b-; Z 3 is selected from a bond or -CR 4a R 4b -;
Z2選自鍵或-CR5aR5b-、-CR5aR5bCR5cR5d-、-CR6a=CR6b-、-N(R7)-、-O-、-C(O)-、-SOn-、-C(O)O-、-C(O)NH-、-CR5aR5b-N(R7)-、-OCR5aR5b-、-C(O)-CR5aR5b-、-SOn-CR5aR5b-、-C(O)O-CR5aR5b-、-OC(O)O-CR5aR5b-、-(O)NH-CR5aR5b-或-NHC(O)-CR5aR5b-; Z 2 is selected from a bond or -CR 5a R 5b -, -CR 5a R 5b CR 5c R 5d -, -CR 6a =CR 6b -, -N(R 7 )-, -O-, -C(O)- , -SO n -, -C(O)O-, -C(O)NH-, -CR 5a R 5b -N(R 7 )-, -OCR 5a R 5b -, -C(O)-CR 5a R 5b -, -SO n -CR 5a R 5b -, -C(O)O-CR 5a R 5b -, -OC(O)O-CR 5a R 5b -, -(O)NH-CR 5a R 5b - or -NHC(O)-CR 5a R 5b -;
Z1選自鍵或-CR8aR8b-; Z 1 is selected from a bond or -CR 8a R 8b -;
Y選自-C(O)NH-或五員至六員雜環,該五員至六員雜環選自 Y is selected from -C(O)NH- or a five- to six-membered heterocycle selected from
Z4選自-CR9aR9b-、-CR9aR9bCR9cR9d-或-CR10a=CR10b-; Z 4 is selected from -CR 9a R 9b -, -CR 9a R 9b CR 9c R 9d - or -CR 10a =CR 10b -;
R3a和R3b各自獨立選自氫、氘、鹵素或C1-6烷基; R 3a and R 3b are each independently selected from hydrogen, deuterium, halogen or C 1-6 alkyl;
或者,R3a和R3b與相鄰碳原子一起形成3至7員環烷基或雜環烷基,該環烷基或雜環烷基視需要被一個或多個RA2所取代,RA2各自獨立選自氘、鹵素、硝基、氰基、C1-6烷基、C1-6烷氧基,該烷基或烷氧基視需要被鹵素、硝基、氰基或C1-6烷氧基所取代; Alternatively, R 3a and R 3b together with adjacent carbon atoms form a 3- to 7-membered cycloalkyl or heterocycloalkyl group optionally substituted with one or more R A2 , R A2 Each is independently selected from deuterium, halogen, nitro, cyano, C 1-6 alkyl, C 1-6 alkoxy, the alkyl or alkoxy optionally being replaced by halogen, nitro, cyano or C 1- 6 alkoxy substituted;
R4a和R4b各自獨立選自氫、氘、鹵素或烷基,該烷基視需要被一個或多個硝基、腈基、羥基或鹵素所取代; R 4a and R 4b are each independently selected from hydrogen, deuterium, halogen or alkyl optionally substituted with one or more nitro, nitrile, hydroxy or halogen;
R5a、R5b、R5c和R5d各自獨立選自氘、氫、鹵素、硝基、羥基、氰基、C1-6烷基、C1-6烷氧基、SR'、NR'(R")、OCOR'、OCOOR'、COOR'、CONR'(R")或OCONR'(R"),該烷基或烷氧基視需要被鹵素、硝基、氰基或C1-6烷氧基所取代; R 5a , R 5b , R 5c and R 5d are each independently selected from deuterium, hydrogen, halogen, nitro, hydroxy, cyano, C 1-6 alkyl, C 1-6 alkoxy, SR', NR' ( R"), OCOR', OCOOR', COOR', CONR'(R") or OCONR'(R"), the alkyl or alkoxy is optionally replaced by halogen, nitro, cyano or C 1-6 alkane substituted by oxy;
或者,R5a、R5b、R5c和R5d中任意兩個與相鄰碳原子一起形成3至7員環烷基或雜環烷基,該環烷基或雜環烷基視需要被一個或多個RA3所取代,RA3各自獨立選自氘、鹵素、硝基、氰基、C1-6烷基、C1-6烷氧基,該烷基或烷氧基視需要被鹵素、硝基、氰基或C1-6烷氧基所取代; Alternatively, any two of R 5a , R 5b , R 5c and R 5d are taken together with adjacent carbon atoms to form a 3- to 7-membered cycloalkyl or heterocycloalkyl which is optionally replaced by a or multiple R A3 substituted, R A3 are each independently selected from deuterium, halogen, nitro, cyano, C 1-6 alkyl, C 1-6 alkoxy, the alkyl or alkoxy is optionally halogenated , nitro, cyano or C 1-6 alkoxy substituted;
R6a和R6b各自獨立選自氫、氘、鹵素或烷基,該烷基視需要被一個或多個硝基、腈基、羥基或鹵素所取代; R 6a and R 6b are each independently selected from hydrogen, deuterium, halogen or alkyl optionally substituted with one or more nitro, nitrile, hydroxy or halogen;
或者,R6a、R6b與相鄰碳原子一起形成5至10員環烷基、雜環烷基、芳基或雜芳基,其視需要被一個或多個RA4所取代,RA4各自獨立選自氘、鹵素、硝基、氰基、C1-6烷基、C1-6烷氧基; Alternatively, R 6a , R 6b together with adjacent carbon atoms form a 5- to 10-membered cycloalkyl, heterocycloalkyl, aryl or heteroaryl group optionally substituted with one or more R A4 , each of which R A4 independently selected from deuterium, halogen, nitro, cyano, C 1-6 alkyl, C 1-6 alkoxy;
R7選自氫、氘、鹵素、硝基、氰基、C1-6烷基、C1-6烷氧基,該烷基或烷氧基視需要被鹵素、硝基、氰基或C1-6烷氧基所取代; R 7 is selected from hydrogen, deuterium, halogen, nitro, cyano, C 1-6 alkyl, C 1-6 alkoxy, the alkyl or alkoxy optionally replaced by halogen, nitro, cyano or C 1-6 alkoxy substituted;
R8a和R8b各自獨立選自氫、氘、羥基、鹵素、硝基、羥基、氰基、C1-6烷基、C1-6烷氧基,該烷基或烷氧基視需要被鹵素、硝基、氰基或C1-6烷氧基所取代; R 8a and R 8b are each independently selected from hydrogen, deuterium, hydroxy, halogen, nitro, hydroxy, cyano, C 1-6 alkyl, C 1-6 alkoxy, the alkyl or alkoxy optionally being substituted by halogen, nitro, cyano or C 1-6 alkoxy;
或者,R8a、R8b與相鄰碳原子一起形成3至7員環烷基或雜環烷基,該環烷基或雜環烷基視需要被一個或多個RA5所取代,RA5各自獨立選自氘、 鹵素、硝基、氰基、C1-6烷基、C1-6烷氧基,該烷基或烷氧基視需要被鹵素、硝基、氰基或C1-6烷氧基所取代; Alternatively, R 8a , R 8b and adjacent carbon atoms together form a 3- to 7-membered cycloalkyl or heterocycloalkyl, the cycloalkyl or heterocycloalkyl may be optionally substituted by one or more R A5 , R A5 each independently selected from deuterium, halogen, nitro, cyano, C 1-6 alkyl, C 1-6 alkoxy, the alkyl or alkoxy optionally replaced by halogen, nitro, cyano or C 1- 6 alkoxy substituted;
或者,R8b與R5a、R5b、R5c、R5d和R7中任一個基團一起形成3至7員環烷基或雜環烷基,該環烷基或雜環烷基視需要被一個或多個RA5所取代,RA5各自獨立選自氘、鹵素、硝基、氰基、C1-6烷基、C1-6烷氧基,該烷基或烷氧基視需要被鹵素、硝基、氰基或C1-6烷氧基所取代; Alternatively, R 8b is taken together with any of R 5a , R 5b , R 5c , R 5d and R 7 to form a 3- to 7-membered cycloalkyl or heterocycloalkyl, optionally Replaced by one or more R A5 , each R A5 is independently selected from deuterium, halogen, nitro, cyano, C 1-6 alkyl, C 1-6 alkoxy, the alkyl or alkoxy is optional substituted by halogen, nitro, cyano or C 1-6 alkoxy;
或者,R8b和R2與相鄰碳原子一起形成3至7員環烷基或雜環烷基,該環烷基或雜環烷基視需要被一個或多個RA5所取代,RA5各自獨立選自氘、鹵素、硝基、氰基、C1-6烷基、C1-6烷氧基,該烷基或烷氧基視需要被鹵素、硝基、氰基或C1-6烷氧基所取代; Alternatively, R 8b and R 2 together with adjacent carbon atoms form a 3- to 7-membered cycloalkyl or heterocycloalkyl group optionally substituted with one or more R A5 , R A5 Each is independently selected from deuterium, halogen, nitro, cyano, C 1-6 alkyl, C 1-6 alkoxy, the alkyl or alkoxy optionally being replaced by halogen, nitro, cyano or C 1- 6 alkoxy substituted;
或者,R8a和R4a或R8b和R4b與相鄰碳原子一起形成3至7員環烷基或雜環烷基,該環烷基或雜環烷基視需要被一個或多個RA5所取代,RA5各自獨立選自氘、鹵素、硝基、氰基、C1-6烷基、C1-6烷氧基,該烷基或烷氧基視需要被鹵素、硝基、氰基或C1-6烷氧基所取代; Alternatively, R 8a and R 4a or R 8b and R 4b are taken together with adjacent carbon atoms to form a 3- to 7-membered cycloalkyl or heterocycloalkyl group optionally separated by one or more R Replaced by A5 , R and A5 are each independently selected from deuterium, halogen, nitro, cyano, C 1-6 alkyl, C 1-6 alkoxy, and the alkyl or alkoxy is optionally substituted by halogen, nitro, substituted by cyano or C 1-6 alkoxy;
R9a、R9b、R9c和R9d各自獨立選自氫、氘、羥基、鹵素、硝基、羥基、氰基、C1-6烷基、C1-6烷氧基,該烷基或烷氧基視需要被鹵素、硝基、氰基或C1-6烷氧基所取代; R 9a , R 9b , R 9c and R 9d are each independently selected from hydrogen, deuterium, hydroxy, halogen, nitro, hydroxy, cyano, C 1-6 alkyl, C 1-6 alkoxy, the alkyl or Alkoxy is optionally substituted by halogen, nitro, cyano or C 1-6 alkoxy;
或者,R9a和R9b或R9c和R9d與相鄰碳原子一起形成3至7員環烷基或雜環烷基,該環烷基或雜環烷基視需要被一個或多個RA6所取代,RA6各自獨立選自氘、鹵素、硝基、氰基、C1-6烷基、C1-6烷氧基,該烷基或烷氧基視需要被鹵素、硝基、氰基或C1-6烷氧基所取代; Alternatively, R 9a and R 9b or R 9c and R 9d are taken together with adjacent carbon atoms to form a 3- to 7-membered cycloalkyl or heterocycloalkyl group optionally separated by one or more R Replaced by A6 , R and A6 are each independently selected from deuterium, halogen, nitro, cyano, C 1-6 alkyl, C 1-6 alkoxy, and the alkyl or alkoxy is optionally substituted by halogen, nitro, substituted by cyano or C 1-6 alkoxy;
或者,R9a、R9b、R9c和R9d中任一個與R4a、R4b、R5a、R5b和R7中任一個一起形成3至7員環烷基或雜環烷基,該環烷基或雜環烷基視需要被一個或多個RA6所取代,RA6各自獨立選自氘、鹵素、硝基、氰基、C1-6烷基、C1-6烷氧基,該烷基或烷氧基視需要被鹵素、硝基、氰基或C1-6烷氧基所取代; Alternatively, any of R 9a , R 9b , R 9c and R 9d together with any of R 4a , R 4b , R 5a , R 5b and R 7 form a 3- to 7-membered cycloalkyl or heterocycloalkyl, the Cycloalkyl or heterocycloalkyl is optionally substituted by one or more R A6 , each R A6 is independently selected from deuterium, halogen, nitro, cyano, C 1-6 alkyl, C 1-6 alkoxy , the alkyl or alkoxy is optionally substituted by halogen, nitro, cyano or C 1-6 alkoxy;
R10a和R10b各自獨立選自氫、氘、羥基、鹵素、硝基、羥基、氰基、C1-6烷基、C1-6烷氧基,該烷基或烷氧基視需要被鹵素、硝基、氰基或C1-6烷氧基所取代; R 10a and R 10b are each independently selected from hydrogen, deuterium, hydroxyl, halogen, nitro, hydroxyl, cyano, C 1-6 alkyl, C 1-6 alkoxy, the alkyl or alkoxy optionally being substituted by halogen, nitro, cyano or C 1-6 alkoxy;
或者,R10a、R10b與相鄰碳原子一起形成5至10員環烷基、雜環烷基、芳基或雜芳基,其視需要被一個或多個RA7所取代,RA7各自獨立選自氘、鹵素、硝基、氰基、C1-6烷基、C1-6烷氧基; Alternatively, R 10a , R 10b together with adjacent carbon atoms form a 5- to 10-membered cycloalkyl, heterocycloalkyl, aryl or heteroaryl group, optionally substituted with one or more R A7 , each R A7 independently selected from deuterium, halogen, nitro, cyano, C 1-6 alkyl, C 1-6 alkoxy;
Rb選自氫或C1-6烷基; R b is selected from hydrogen or C 1-6 alkyl;
R'或R"獨立地選自氫、羥基、C1-6烷基、C1-6烷氧基; R' or R" is independently selected from hydrogen, hydroxyl, C 1-6 alkyl, C 1-6 alkoxy;
n選自0-2之間整數,例如1或2。 n is selected from an integer between 0-2, such as 1 or 2.
本揭露中該缺陷是該基因中的突變、該基因中的缺失或有缺陷的表達。 The defect in the present disclosure is a mutation in the gene, a deletion in the gene, or defective expression.
在一些實施方案中,式I所示化合物或其可藥用鹽中R2選自氫、氘、甲基、乙基或氟甲基。在一些實施方案中,式I所示化合物或其可藥用鹽中R2選自氫或甲基,更佳為氫。 In some embodiments, in the compound of formula I or a pharmaceutically acceptable salt thereof, R 2 is selected from hydrogen, deuterium, methyl, ethyl, or fluoromethyl. In some embodiments, in the compound of formula I or a pharmaceutically acceptable salt thereof, R 2 is selected from hydrogen or methyl, more preferably hydrogen.
在一些實施方案中,式I所示化合物或其可藥用鹽中Y選自-C(O)NH-。 In some embodiments, Y in the compound of formula I or a pharmaceutically acceptable salt thereof is selected from -C(O)NH-.
在一些實施方案中,式I所示化合物或其可藥用鹽中Y選自 In some embodiments, Y in the compound of formula I or a pharmaceutically acceptable salt thereof is selected from
在一些實施方案中,式I所示化合物或其可藥用鹽中Y選自 In some embodiments, Y in the compound of formula I or a pharmaceutically acceptable salt thereof is selected from
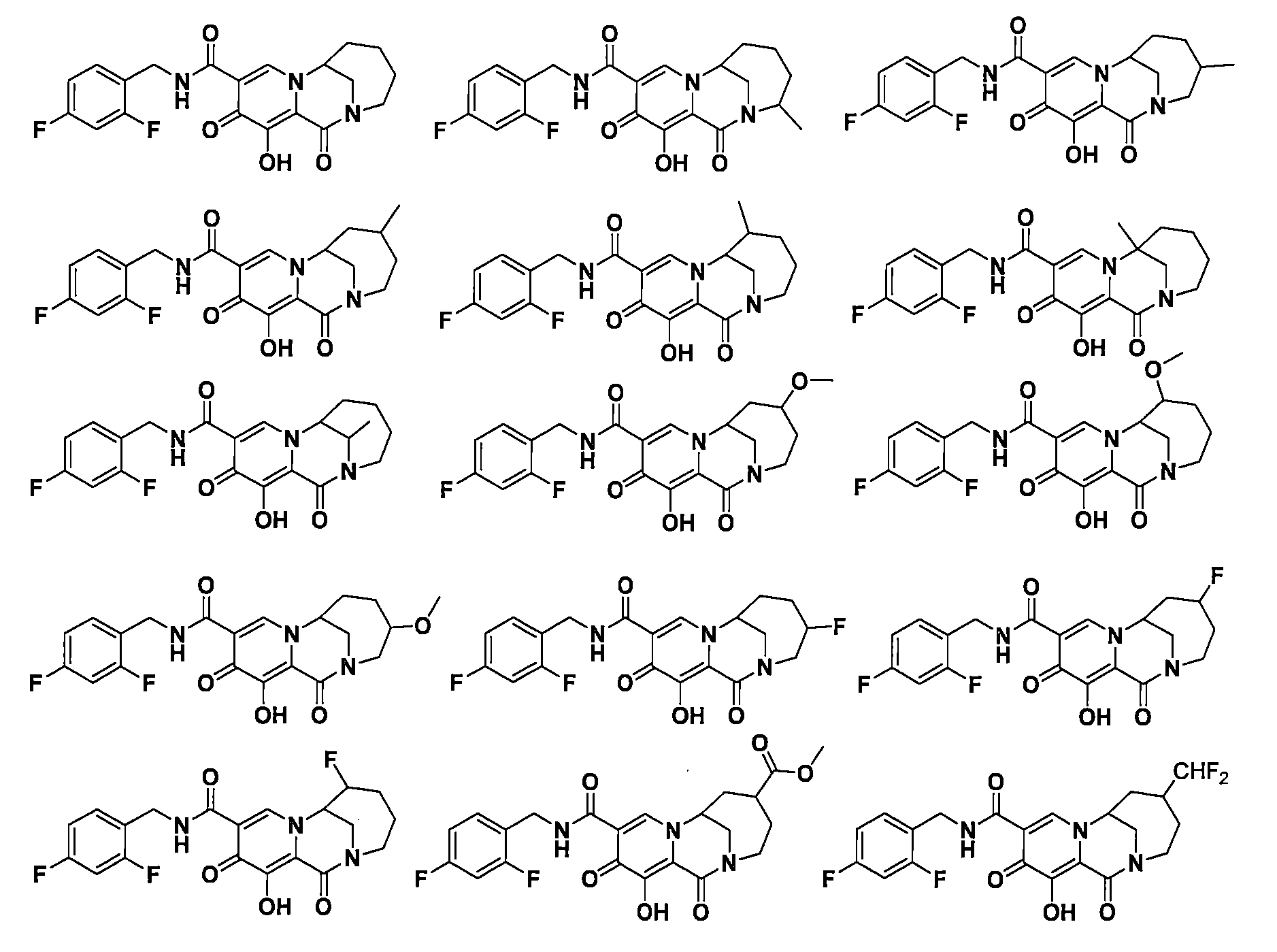
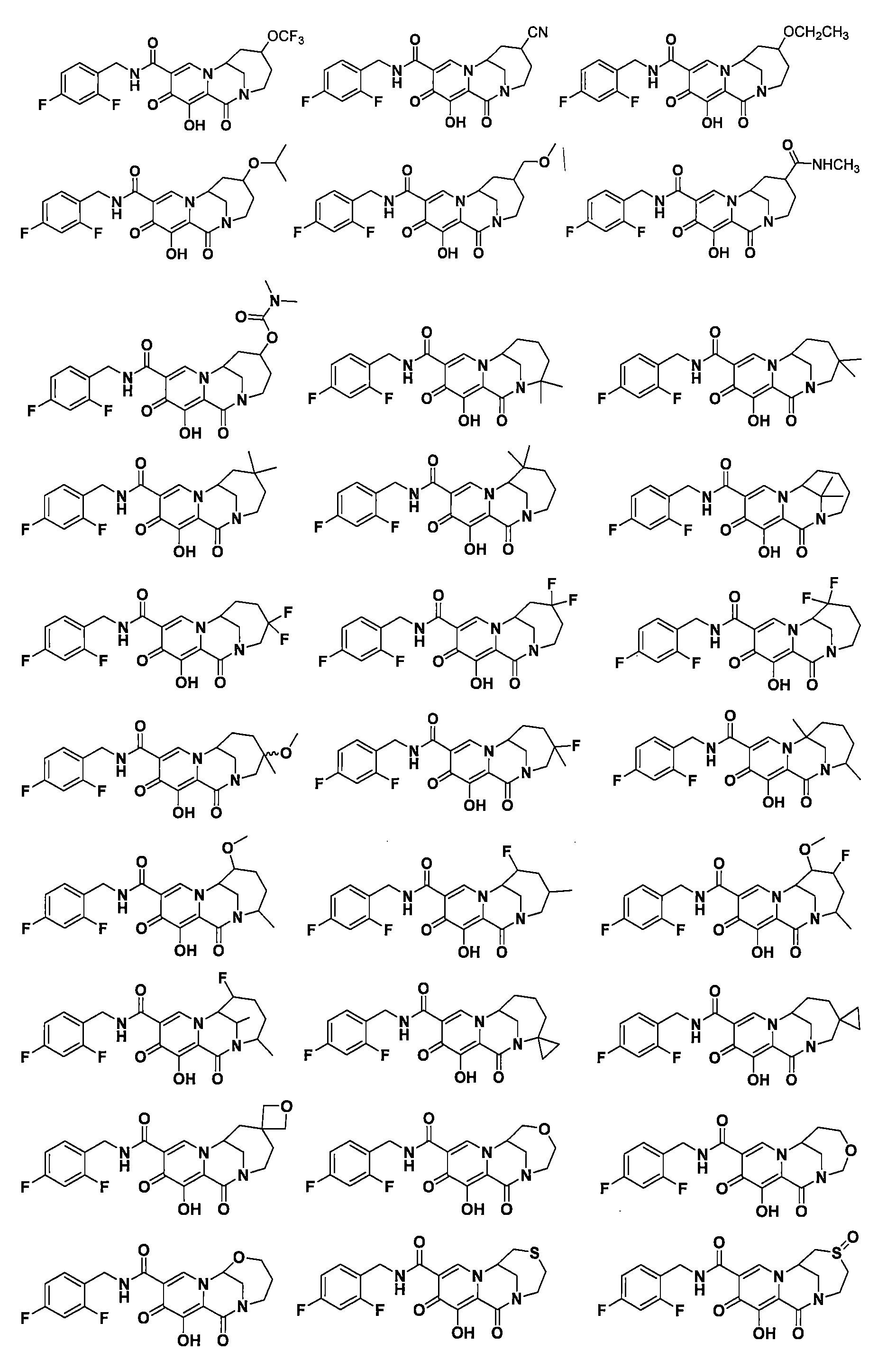
在一些實施方案中,式I所示化合物或其可藥用鹽為 In some embodiments, the compound of formula I or a pharmaceutically acceptable salt thereof is
在一些實施方案中,式I所示化合物或其可藥用鹽為 In some embodiments, the compound of formula I or a pharmaceutically acceptable salt thereof is
在一些實施方案中,式I所示化合物或其可藥用鹽中R1選自苯基,該苯基視需要被一個或多個RA1所取代,RA1各自獨立選自氘、鹵素、硝基、氰基、C1-6烷基、C1-6烷氧基,該烷基或烷氧基視需要被鹵素、硝基、氰基或C1-6烷氧基所取代。 In some embodiments, in the compound represented by formula I or a pharmaceutically acceptable salt thereof, R 1 is selected from phenyl, and the phenyl group is optionally substituted by one or more R A1 , and R A1 is independently selected from deuterium, halogen, Nitro group, cyano group, C 1-6 alkyl group, C 1-6 alkoxy group, the alkyl or alkoxy group is optionally substituted by halogen, nitro group, cyano group or C 1-6 alkoxy group.
在一些實施方案中,式I所示化合物或其可藥用鹽中R1選自苯基,該苯基視需要被1至4個RA1所取代,RA1各自獨立選自氘、鹵素、硝基、氰基、C1-6烷基、鹵C1-6烷基或C1-6烷氧基。 In some embodiments, in the compound represented by formula I or a pharmaceutically acceptable salt thereof, R 1 is selected from phenyl, and the phenyl group is optionally substituted by 1 to 4 R A1 , and R A1 is independently selected from deuterium, halogen, nitro, cyano, C 1-6 alkyl, halo C 1-6 alkyl or C 1-6 alkoxy.
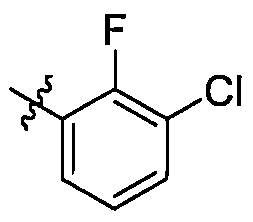
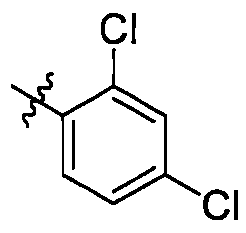
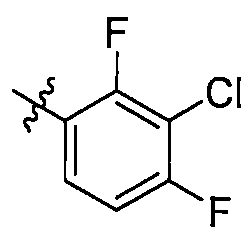
在一些實施方案中,式I所示化合物或其可藥用鹽中R1選自苯基,該苯基視需要被1至4個RA1所取代,RA1各自獨立選自鹵素或C1-6烷氧基; 進一步地,R1較佳,,,,, In some embodiments, in the compound represented by formula I or a pharmaceutically acceptable salt thereof, R 1 is selected from phenyl, and the phenyl group is optionally substituted by 1 to 4 R A1 , and R A1 is independently selected from halogen or C 1 -6 alkoxy; further, R 1 is preferred , , , , ,
在一些實施方案中,式I所示化合物或其可藥用鹽中R1選自 In some embodiments, in the compound of formula I or a pharmaceutically acceptable salt thereof, R 1 is selected from
在一些實施方案中,式I所示化合物或其可藥用鹽中R1選自 In some embodiments, in the compound of formula I or a pharmaceutically acceptable salt thereof, R 1 is selected from
在一些實施方案中,式I所示化合物或其可藥用鹽為 In some embodiments, the compound of formula I or a pharmaceutically acceptable salt thereof is
在一些實施方案中,式I所示化合物或其可藥用鹽中L選自-CH2-、-CH(CH3)-、-C(O)-或-CH2CH2-。 In some embodiments, in the compound of formula I or a pharmaceutically acceptable salt thereof, L is selected from -CH2- , -CH( CH3 )-, -C (O)- or -CH2CH2- .
在一些實施方案中,式I所示化合物或其可藥用鹽中L選自-CH2-。 In some embodiments, in the compound of formula I or a pharmaceutically acceptable salt thereof, L is selected from -CH2- .
在一些實施方案中,式I所示化合物或其可藥用鹽中L選自-CH2-、-CH(CH3)-、-C(CH3)2-或。 In some embodiments, in the compound of formula I or a pharmaceutically acceptable salt thereof, L is selected from -CH 2 -, -CH(CH 3 )-, -C(CH 3 ) 2 - or .
在一些實施方案中,式I所示化合物或其可藥用鹽為 In some embodiments, the compound of formula I or a pharmaceutically acceptable salt thereof is
在一些實施方案中,式I所示化合物或其可藥用鹽為 In some embodiments, the compound of formula I or a pharmaceutically acceptable salt thereof is
在一些實施方案中,式I所示化合物或其可藥用鹽為 In some embodiments, the compound of formula I or a pharmaceutically acceptable salt thereof is
在一些實施方案中,式I所示化合物或其可藥用鹽中L選自-CH2CH2-。 In some embodiments, in the compound of formula I or a pharmaceutically acceptable salt thereof, L is selected from -CH 2 CH 2 -.
在一些實施方案中,式I所示化合物或其可藥用鹽為 In some embodiments, the compound of formula I or a pharmaceutically acceptable salt thereof is
在一些實施方案中,式I所示化合物或其可藥用鹽為 In some embodiments, the compound of formula I or a pharmaceutically acceptable salt thereof is
在一些實施方案中,式I所示化合物或其可藥用鹽中Z3選自鍵或-CR4aR4b-,R4a和R4b各自獨立選自氫、氘、鹵素或C1-6烷基。 In some embodiments, in the compound of formula I or a pharmaceutically acceptable salt thereof, Z 3 is selected from a bond or -CR 4a R 4b -, and R 4a and R 4b are each independently selected from hydrogen, deuterium, halogen or C 1-6 alkyl.
在一些實施方案中,式I所示化合物或其可藥用鹽中Z3選自鍵或-CR4aR4b-,R4a和R4b各自獨立選自氫、氘、鹵素或甲基。 In some embodiments, in the compound of formula I or a pharmaceutically acceptable salt thereof, Z 3 is selected from a bond or -CR 4a R 4b -, and R 4a and R 4b are each independently selected from hydrogen, deuterium, halogen or methyl.
在一些實施方案中,式I所示化合物或其可藥用鹽中Z3選自鍵、-CH2-、-CF2-、-CH(F)-、-CH(CH3)-或-CF(CH3)-,較佳鍵、-CH2-、-CF2-或-CH(F)-。 In some embodiments, in the compound of formula I or a pharmaceutically acceptable salt thereof, Z 3 is selected from bond, -CH 2 -, -CF 2 -, -CH(F)-, -CH(CH 3 )- or - CF(CH 3 )-, preferably a bond, -CH 2 -, -CF 2 - or -CH(F)-.
另一方面,在一些實施方案中,式I所示化合物或其可藥用鹽中Z2選自-CR5aR5b-、-CR5aR5bCR5cR5d-、-CR6a=CR6b-、-N(R7)-、-O-、-C(O)-、-SOn-、-C(O)O-、-O-CR5aR5b-或-C(O)-CR5aR5b-, On the other hand, in some embodiments, in the compound shown in formula I or a pharmaceutically acceptable salt thereof, Z 2 is selected from -CR 5a R 5b -, -CR 5a R 5b CR 5c R 5d -, -CR 6a =CR 6b -, -N(R 7 )-, -O-, -C(O)-, -SO n -, -C(O)O-, -O-CR 5a R 5b - or -C(O)-CR 5a R 5b -,
R5a、R5b、R5c和R5d各自獨立選自氫、氘、鹵素、硝基、羥基、氰基、C1-6烷基、鹵C1-6烷基或C1-6烷氧基; R 5a , R 5b , R 5c and R 5d are each independently selected from hydrogen, deuterium, halogen, nitro, hydroxy, cyano, C 1-6 alkyl, halo C 1-6 alkyl or C 1-6 alkoxy base;
或者,R5a、R5b、R5c和R5d與相鄰碳原子一起形成3至7員環烷基或雜環烷基,該環烷基或雜環烷基視需要被一個或多個RA3所取代,RA3各自獨立選自氘、鹵素、硝基、氰基、C1-6烷基、鹵C1-6烷基、C1-6烷氧基; Alternatively, R 5a , R 5b , R 5c and R 5d are taken together with adjacent carbon atoms to form a 3- to 7-membered cycloalkyl or heterocycloalkyl group optionally substituted by one or more R Replaced by A3 , R and A3 are each independently selected from deuterium, halogen, nitro, cyano, C 1-6 alkyl, halogen C 1-6 alkyl, C 1-6 alkoxy;
R6a和R6b各自獨立選自氫、氘、鹵素、C1-6烷基或鹵C1-6烷基; R 6a and R 6b are each independently selected from hydrogen, deuterium, halogen, C 1-6 alkyl or halo C 1-6 alkyl;
或者,R6a、R6b與相鄰碳原子一起形成5至10員環烷基、雜環烷基、芳基或雜芳基,其視需要被一個或多個RA4所取代,RA4各自獨立選自氘、鹵素、氰基、C1-6烷基; Alternatively, R 6a , R 6b together with adjacent carbon atoms form a 5- to 10-membered cycloalkyl, heterocycloalkyl, aryl or heteroaryl group optionally substituted with one or more R A4 , each of which R A4 independently selected from deuterium, halogen, cyano, C 1-6 alkyl;
R7選自氫、氘、鹵素、C1-6烷基或鹵C1-6烷基; R 7 is selected from hydrogen, deuterium, halogen, C 1-6 alkyl or halogen C 1-6 alkyl;
n選自0、1或2。 n is selected from 0, 1 or 2.
在一些實施方案中,式I所示化合物或其可藥用鹽中Z2選自-CR5aR5b-、-CR5aR5bCR5cR5d-、-CR6a=CR6b-、-N(R7)-、-O-、-C(O)-、-SOn-、-C(O)O-或-O-CR5aR5b-, In some embodiments, in the compound shown in formula I or a pharmaceutically acceptable salt thereof, Z 2 is selected from -CR 5a R 5b -, -CR 5a R 5b CR 5c R 5d -, -CR 6a =CR 6b -, -N (R 7 )-, -O-, -C(O)-, -SO n -, -C(O)O- or -O-CR 5a R 5b -,
R5a、R5b、R5c和R5d各自獨立選自氫、氘、鹵素、羥基或C1-6烷基; R 5a , R 5b , R 5c and R 5d are each independently selected from hydrogen, deuterium, halogen, hydroxy or C 1-6 alkyl;
或者R5a、R5b、R5c和R5d與相鄰碳原子一起形成3員環烷基,該環烷基視需要被一個或多個RA3所取代,RA3各自獨立選自氘、鹵素、C1-6烷基或鹵C1-6烷基; Or R 5a , R 5b , R 5c and R 5d together with adjacent carbon atoms form a 3-membered cycloalkyl, which is optionally substituted by one or more R A3 , each R A3 is independently selected from deuterium, halogen , C 1-6 alkyl or halogen C 1-6 alkyl;
R6a和R6b各自獨立選自氫、氘、鹵素或C1-6烷基或鹵C1-6烷基; R 6a and R 6b are each independently selected from hydrogen, deuterium, halogen or C 1-6 alkyl or halo C 1-6 alkyl;
或者,R6a、R6b與相鄰碳原子一起形成5至6員雜環烷基或雜芳基,其視需要被一個或多個RA4所取代,RA4各自獨立選自氘、鹵素或C1-6烷基; Alternatively, R 6a , R 6b together with adjacent carbon atoms form a 5- to 6-membered heterocycloalkyl or heteroaryl group, optionally substituted by one or more R A4 , each independently selected from deuterium, halogen or C 1-6 alkyl;
R7選自氫、氘、C1-6烷基或鹵C1-6烷基; R 7 is selected from hydrogen, deuterium, C 1-6 alkyl or halo C 1-6 alkyl;
n選自0、1或2。 n is selected from 0, 1 or 2.
在一些實施方案中,式I所示化合物或其可藥用鹽中Z2選自-CR5aR5b-、-CR5aR5bCR5cR5d-、-CR6a=CR6b-、-N(R7)-、-O-、-C(O)-、-SOn-、-C(O)O-或-O-CR5aR5b-, In some embodiments, in the compound shown in formula I or a pharmaceutically acceptable salt thereof, Z 2 is selected from -CR 5a R 5b -, -CR 5a R 5b CR 5c R 5d -, -CR 6a =CR 6b -, -N (R 7 )-, -O-, -C(O)-, -SO n -, -C(O)O- or -O-CR 5a R 5b -,
R5a、R5b、R5c和R5d各自獨立選自氫、氘、鹵素、羥基或C1-6烷基; R 5a , R 5b , R 5c and R 5d are each independently selected from hydrogen, deuterium, halogen, hydroxy or C 1-6 alkyl;
或者R5a、R5b、R5c和R5d中任意兩個與相鄰碳原子一起形成5員環烷基,該環烷基視需要被一個或多個RA3所取代,RA3各自獨立選自氘、鹵素、C1-6烷基或鹵C1-6烷基; Or any two of R 5a , R 5b , R 5c and R 5d together with adjacent carbon atoms form a 5-membered cycloalkyl group, the cycloalkyl group is optionally substituted by one or more R A3 , and R A3 is independently selected from deuterium, halogen, C 1-6 alkyl or halogen C 1-6 alkyl;
R6a和R6b各自獨立選自氫、氘、鹵素或C1-6烷基或鹵C1-6烷基; R 6a and R 6b are each independently selected from hydrogen, deuterium, halogen or C 1-6 alkyl or halo C 1-6 alkyl;
或者,R6a、R6b與相鄰碳原子一起形成5至6員雜環烷基或雜芳基,其視需要被一個或多個RA4所取代,RA4選自氘、鹵素或C1-6烷基; Alternatively, R 6a , R 6b together with adjacent carbon atoms form a 5- to 6-membered heterocycloalkyl or heteroaryl group, optionally substituted by one or more R A4 selected from deuterium, halogen or C 1 -6 alkyl;
R7選自氫、氘、C1-6烷基或鹵C1-6烷基; R 7 is selected from hydrogen, deuterium, C 1-6 alkyl or halo C 1-6 alkyl;
n選自0、1或2。 n is selected from 0, 1 or 2.
在一些實施方案中,式I所示化合物或其可藥用鹽中Z2選自-CR5aR5b-、-CR5aR5bCR5cR5d-、-CR6a=CR6b-、-N(R7)-、-O-、-C(O)-、-SOn-、-C(O)O-或-O-CR5aR5b-,R5a、R5b、R5c和R5d各自獨立選自氫、氘、鹵素、羥基或C1-6烷基;R6a和R6b各自獨立選自氫、氘、鹵素或C1-6烷基或鹵C1-6烷基;R7選自氫、氘、C1-6烷基或鹵C1-6烷基;n選自0、1或2。 In some embodiments, in the compound shown in formula I or a pharmaceutically acceptable salt thereof, Z 2 is selected from -CR 5a R 5b -, -CR 5a R 5b CR 5c R 5d -, -CR 6a =CR 6b -, -N (R 7 )-, -O-, -C(O)-, -SO n -, -C(O)O- or -O-CR 5a R 5b -, R 5a , R 5b , R 5c and R 5d Each is independently selected from hydrogen, deuterium, halogen, hydroxyl or C 1-6 alkyl; R 6a and R 6b are each independently selected from hydrogen, deuterium, halogen or C 1-6 alkyl or halogen C 1-6 alkyl; R 7 is selected from hydrogen, deuterium, C 1-6 alkyl or halo C 1-6 alkyl; n is selected from 0, 1 or 2.
在一些實施方案中,式I所示化合物或其可藥用鹽中Z2選自-CH2-、-CH2CH2-、-CH2=CH2-、-NH-、-N(CH3)-、-N(CH(CH3)2)-、-N(C(O)CH3)-、-N(SO2CH3)-、-O-、-OCH2-或。 In some embodiments, in the compound of formula I or a pharmaceutically acceptable salt thereof, Z 2 is selected from -CH 2 -, -CH 2 CH 2 -, -CH 2 =CH 2 -, -NH-, -N(CH 3 )-, -N(CH(CH 3 ) 2 )-, -N(C(O)CH 3 )-, -N(SO 2 CH 3 )-, -O-, -OCH 2 - or .
在一些實施方案中,式I所示化合物或其可藥用鹽中Z1選自鍵。 In some embodiments, Z 1 in the compound of formula I or a pharmaceutically acceptable salt thereof is selected from a bond.
在一些實施方案中,式I所示化合物或其可藥用鹽中Z1選自-CR8aR8b-,R8a和R8b各自獨立選自氫、氘、羥基、鹵素、羥基、C1-6烷基、C1-6烷氧基,該烷基或烷氧基視需要被鹵素所取代。 In some embodiments, in the compound represented by formula I or a pharmaceutically acceptable salt thereof, Z 1 is selected from -CR 8a R 8b -, and R 8a and R 8b are each independently selected from hydrogen, deuterium, hydroxyl, halogen, hydroxyl, C 1 -6 alkyl, C 1-6 alkoxy, the alkyl or alkoxy is optionally substituted by halogen.
在一些實施方案中,式I所示化合物或其可藥用鹽中Z1選自-CR8aR8b-,R8a、R8b與相鄰碳原子一起形成3至6員雜環烷基,該雜環烷基視需要被一個或多個RA5所取代,RA5各自獨立選自氘、鹵素、C1-6烷基、C1-6烷氧基,該烷基或烷氧基視需要被鹵素所取代。 In some embodiments, in the compound represented by formula I or a pharmaceutically acceptable salt thereof, Z 1 is selected from -CR 8a R 8b -, R 8a and R 8b together with adjacent carbon atoms form a 3- to 6-membered heterocycloalkyl, The heterocycloalkyl is optionally substituted by one or more R A5 , and R A5 is independently selected from deuterium, halogen, C 1-6 alkyl, C 1-6 alkoxy, and the alkyl or alkoxy is optionally need to be replaced by halogen.
在一些實施方案中,式I所示化合物或其可藥用鹽中Z1選自-CR8aR8b-,R8a、R8b與相鄰碳原子一起形成3至6員環烷基,該環烷基視需要被 一個或多個RA5所取代,RA5各自獨立選自氘、鹵素、C1-6烷基、C1-6烷氧基,該烷基或烷氧基視需要被鹵素所取代。 In some embodiments, in the compound represented by formula I or a pharmaceutically acceptable salt thereof, Z 1 is selected from -CR 8a R 8b -, R 8a , R 8b and adjacent carbon atoms together form a 3- to 6-membered cycloalkyl, the Cycloalkyl is optionally substituted by one or more R A5 , and R A5 is independently selected from deuterium, halogen, C 1-6 alkyl, C 1-6 alkoxy, and the alkyl or alkoxy is optionally substituted by substituted by halogen.
在一些實施方案中,式I所示化合物或其可藥用鹽中Z1選自R8b和R2與相鄰碳原子一起形成3至6員環烷基或雜環烷基,該環烷基或雜環烷基視需要被一個或多個RA5所取代,RA5各自獨立選自氘、鹵素、C1-6烷基、C1-6烷氧基,該烷基或烷氧基視需要被鹵素所取代。 In some embodiments, in the compound of formula I or a pharmaceutically acceptable salt thereof, Z 1 is selected from R 8b and R 2 together with adjacent carbon atoms form a 3- to 6-membered cycloalkyl or heterocycloalkyl, the cycloalkane Radical or heterocycloalkyl is optionally substituted by one or more R A5 , each R A5 is independently selected from deuterium, halogen, C 1-6 alkyl, C 1-6 alkoxy, the alkyl or alkoxy Optionally substituted with halogen.
在一些實施方案中,式I所示化合物或其可藥用鹽中Z1選自-CH2-、-CF2-、-CHF-、-CH(OH)-、-CH(CH3)-、-CH(CH2F)-、-C(CH2F)(OH)-、-C(CH3)(OH)-、-C(CH2CH3)(OCH3)-、-CH(CH2CH3)-、-C(CH3)(OCH3)-或-CF(CH3)-,較佳-CH2-、-CF2-、-CHF-、-CH(OH)-、-CH(CH3)-或-CF(CH3)-。 In some embodiments, in the compound of formula I or a pharmaceutically acceptable salt thereof, Z 1 is selected from -CH 2 -, -CF 2 -, -CHF-, -CH(OH)-, -CH(CH 3 )- , -CH(CH 2 F)-, -C(CH 2 F)(OH)-, -C(CH 3 )(OH)-, -C(CH 2 CH 3 )(OCH 3 )-, -CH( CH 2 CH 3 )-, -C(CH 3 )(OCH 3 )- or -CF(CH 3 )-, preferably -CH 2 -, -CF 2 -, -CHF-, -CH(OH)-, -CH( CH3 )- or -CF( CH3 )-.
另一方面,在一些實施方案中,式I所示化合物或其可藥用鹽中Z4選自-CR9aR9b-。 On the other hand, in some embodiments, Z 4 in the compound of formula I or a pharmaceutically acceptable salt thereof is selected from -CR 9a R 9b -.
在一些實施方案中,式I所示化合物或其可藥用鹽中Z4選自-CR9aR9bCR9cR9d-。 In some embodiments, Z 4 in the compound of formula I or a pharmaceutically acceptable salt thereof is selected from -CR 9a R 9b CR 9c R 9d -.
在一些實施方案中,式I所示化合物或其可藥用鹽中R9a、R9b、R9c和R9d各自獨立選自氫、氘、羥基、鹵素、羥基、C1-6烷基、C1-6烷氧基,該烷基或烷氧基視需要被鹵素所取代。 In some embodiments, each of R 9a , R 9b , R 9c and R 9d in the compound of formula I or a pharmaceutically acceptable salt thereof is independently selected from hydrogen, deuterium, hydroxyl, halogen, hydroxyl, C 1-6 alkyl, C 1-6 alkoxy, the alkyl or alkoxy is optionally substituted by halogen.
在一些實施方案中,式I所示化合物或其可藥用鹽中R9a、R9b、R9c和R9d各自獨立選自氫、氘、羥基、氟、甲基、乙基、-CHF2或-CH2F。 In some embodiments, each of R 9a , R 9b , R 9c and R 9d in the compound of formula I or a pharmaceutically acceptable salt thereof is independently selected from hydrogen, deuterium, hydroxyl, fluorine, methyl, ethyl, -CHF 2 or -CH 2 F.
在一些實施方案中,式I所示化合物或其可藥用鹽為 In some embodiments, the compound of formula I or a pharmaceutically acceptable salt thereof is
在一些實施方案中,式I所示化合物或其可藥用鹽為 In some embodiments, the compound of formula I or a pharmaceutically acceptable salt thereof is
在一些實施方案中,式Va或Vb所示化合物或其可藥用鹽中Z4選自-CR9aR9b-。 In some embodiments, Z 4 in the compound of formula Va or Vb or a pharmaceutically acceptable salt thereof is selected from -CR 9a R 9b -.
在一些實施方案中,式Va或Vb所示化合物或其可藥用鹽中Z4選自-CR9aR9bCR9cR9d-。 In some embodiments, Z 4 in the compound of formula Va or Vb or a pharmaceutically acceptable salt thereof is selected from -CR 9a R 9b CR 9c R 9d -.
在一些實施方案中,式Va或Vb所示化合物或其可藥用鹽中R9a、R9b、R9c和R9d各自獨立選自氫、氘、羥基、鹵素、羥基、C1-6烷基、C1-6烷氧基,該烷基或烷氧基視需要被鹵素所取代。 In some embodiments, each of R 9a , R 9b , R 9c and R 9d in the compound represented by formula Va or Vb or a pharmaceutically acceptable salt thereof is independently selected from hydrogen, deuterium, hydroxyl, halogen, hydroxyl, C 1-6 alkane group, C 1-6 alkoxy group, the alkyl group or alkoxy group is optionally substituted by halogen.
在一些實施方案中,式Va或Vb所示化合物或其可藥用鹽中R9a、R9b、R9c和R9d各自獨立選自氫、氘、羥基、氟、甲基、乙基。-CHF2或-CH2F。 In some embodiments, each of R 9a , R 9b , R 9c and R 9d in the compound of formula Va or Vb or a pharmaceutically acceptable salt thereof is independently selected from hydrogen, deuterium, hydroxyl, fluorine, methyl, and ethyl. -CHF2 or -CH2F .
在一些實施方案中,式I所示化合物或其可藥用鹽中Z4選自-CH2-、-CH(CH3)-、-CH2CH2-、、、、或。 In some embodiments, in the compound of formula I or a pharmaceutically acceptable salt thereof, Z 4 is selected from -CH 2 -, -CH(CH 3 )-, -CH 2 CH 2 -, , , , or .
在一些實施方案中,式I所示化合物或其可藥用鹽中Z4選自-CH2-、-CH(CH3)-、-CH2CH2-、、、。 In some embodiments, in the compound of formula I or a pharmaceutically acceptable salt thereof, Z 4 is selected from -CH 2 -, -CH(CH 3 )-, -CH 2 CH 2 -, , , .
在一些實施方案中,式I所示化合物或其可藥用鹽中Z4選自-CH2-、-CH(CH3)-、-CH2CH2-。 In some embodiments, in the compound of formula I or a pharmaceutically acceptable salt thereof, Z 4 is selected from -CH 2 -, -CH(CH 3 )-, -CH 2 CH 2 -.
在一些實施方案中,式I所示化合物或其可藥用鹽中Z4選自-CH2-、-CF2-、-CHF-、-CH(OH)-、-CH(CH3)-、-CH(CH2F)-、-C(CH2F)(OH)-、-C(CH3)(OH)-、-C(CH2CH3)(OCH3)-、-CH(CH2CH3)-、-C(CH3)(OCH3)-或-CF(CH3)-。在一些實施方案中,式I所示化合物或其可藥用鹽中Z4選自-CH2-、-CF2-、-CHF-、-CH(OH)-、-CH(CH3)-或-CF(CH3)-。 In some embodiments, in the compound of formula I or a pharmaceutically acceptable salt thereof, Z 4 is selected from -CH 2 -, -CF 2 -, -CHF-, -CH(OH)-, -CH(CH 3 )- , -CH(CH 2 F)-, -C(CH 2 F)(OH)-, -C(CH 3 )(OH)-, -C(CH 2 CH 3 )(OCH 3 )-, -CH( CH2CH3 )-, -C( CH3 )( OCH3 )- or -CF( CH3 ) -. In some embodiments, in the compound of formula I or a pharmaceutically acceptable salt thereof, Z 4 is selected from -CH 2 -, -CF 2 -, -CHF-, -CH(OH)-, -CH(CH 3 )- or -CF(CH 3 )-.
在一些實施方案中,式I所示化合物或其可藥用鹽中Z4選自-N(R7)-、-O-、-C(O)-、-SOn-,n選自0、1或2。 In some embodiments, in the compound represented by formula I or a pharmaceutically acceptable salt thereof, Z 4 is selected from -N(R 7 )-, -O-, -C(O)-, -SO n -, and n is selected from 0 , 1 or 2.
在一些實施方案中,式I所示化合物或其可藥用鹽中R5a、R5b與相鄰碳原子一起形成3至6員環烷基或雜環烷基,該環烷基或雜環烷基視需要被1至3個RA3所取代,RA3各自獨立選自氘、鹵素、C1-6烷基或鹵C1-6烷基。 In some embodiments, in the compound of formula I or a pharmaceutically acceptable salt thereof, R 5a , R 5b and adjacent carbon atoms together form a 3- to 6-membered cycloalkyl or heterocycloalkyl, the cycloalkyl or heterocycle The alkyl group is optionally substituted with 1 to 3 R A3 , each R A3 is independently selected from deuterium, halogen, C 1-6 alkyl or halo C 1-6 alkyl.
在一些實施方案中,式I所示化合物或其可藥用鹽中R4a、R4b與相鄰碳原子一起形成3至6員環烷基或雜環烷基,該環烷基或雜環烷基視需要被1至3個RA3所取代,RA3各自獨立選自氘、鹵素、C1-6烷基或鹵C1-6烷基。 In some embodiments, in the compound represented by formula I or a pharmaceutically acceptable salt thereof, R 4a , R 4b and adjacent carbon atoms together form a 3- to 6-membered cycloalkyl or heterocycloalkyl, the cycloalkyl or heterocycle The alkyl group is optionally substituted with 1 to 3 R A3 , each R A3 is independently selected from deuterium, halogen, C 1-6 alkyl or halo C 1-6 alkyl.
在一些實施方案中,式I所示化合物或其可藥用鹽中R8a、R8b與相鄰碳原子一起形成3至6員環烷基或雜環烷基,該環烷基或雜環烷基視需要被1至3個RA3所取代,RA3各自獨立選自氘、鹵素、C1-6烷基或鹵C1-6烷基。 In some embodiments, in the compound of formula I or a pharmaceutically acceptable salt thereof, R 8a , R 8b and adjacent carbon atoms together form a 3- to 6-membered cycloalkyl or heterocycloalkyl, the cycloalkyl or heterocycle The alkyl group is optionally substituted with 1 to 3 R A3 , each R A3 is independently selected from deuterium, halogen, C 1-6 alkyl or halo C 1-6 alkyl.
在一些實施方案中,式I所示化合物或其可藥用鹽中Z2選自-CR5aR5b-,R5a、R5b各自選自OCOC1-6烷基、OCOOC1-6烷基、COOC1-6烷基、CONH(C1-6烷基)或OCONC1-6烷基(C1-6烷基),該C1-6烷基選自甲基、乙基或丙基。 In some embodiments, in the compound represented by formula I or a pharmaceutically acceptable salt thereof, Z 2 is selected from -CR 5a R 5b -, and R 5a and R 5b are each selected from OCOC 1-6 alkyl, OCOOC 1-6 alkyl , COOC 1-6 alkyl, CONH (C 1-6 alkyl) or OCONC 1-6 alkyl (C 1-6 alkyl), the C 1-6 alkyl is selected from methyl, ethyl or propyl .
在一些實施方案中,式I所示化合物或其可藥用鹽中Z1選自鍵,Z2選自-OCR5aR5b-,Z4選自-CR9aR9b-,其中R5a與R9a或R5b與R9b與相鄰碳原子一起形成3至7員環烷基或雜環烷基,該環烷基或雜環烷基視需要被1至3個氘、鹵素、氰基、C1-6烷基所取代。 In some embodiments, in the compound represented by formula I or a pharmaceutically acceptable salt thereof, Z 1 is selected from a bond, Z 2 is selected from -OCR 5a R 5b -, and Z 4 is selected from -CR 9a R 9b -, wherein R 5a and R 9a or R 5b and R 9b together with adjacent carbon atoms form a 3- to 7-membered cycloalkyl or heterocycloalkyl group optionally substituted by 1 to 3 deuterium, halogen, cyano groups , C 1-6 alkyl substituted.
在一些實施方案中,式I所示化合物或其可藥用鹽中Z1選自-CR8aR8b-,Z3選自-CR4aR4b-,其中R8a和R4a與相鄰碳原子一起形成3至7員環烷基,該環烷基視需要被1至3個氘、鹵素、氰基、C1-6烷基所取代。 In some embodiments, in the compound represented by formula I or a pharmaceutically acceptable salt thereof, Z 1 is selected from -CR 8a R 8b -, and Z 3 is selected from -CR 4a R 4b -, wherein R 8a and R 4a are adjacent to the adjacent carbon The atoms together form a 3 to 7 membered cycloalkyl group optionally substituted with 1 to 3 deuterium, halogen, cyano, C1-6 alkyl groups.
在一些實施方案中,式I所示化合物或其可藥用鹽中Z2選自-N(R7)-、-O-、-C(O)-、-SOn-,R7選自氫、C1-6烷基或鹵C1-6烷基,n=0、1或2。 In some embodiments, in the compound represented by formula I or a pharmaceutically acceptable salt thereof, Z 2 is selected from -N(R 7 )-, -O-, -C(O)-, -SO n -, and R 7 is selected from Hydrogen, C 1-6 alkyl or halo C 1-6 alkyl, n=0, 1 or 2.
在一些實施方案中,式I所示典型化合物或其可藥用的鹽包括但不限於: In some embodiments, typical compounds of formula I or pharmaceutically acceptable salts thereof include, but are not limited to:
在一些實施方案中,該HIV病毒是對整合酶抑制劑具有抗性,該整合酶抑制劑選自雷特格韋、埃替拉韋、比克替拉韋、多替拉韋、卡博特韋。在一些實施方案中,該HIV病毒是對雷特格韋或埃替拉韋具有抗性。 In some embodiments, the HIV virus is resistant to an integrase inhibitor selected from the group consisting of raltegravir, elvitegravir, bictegravir, dolutegravir, cabot Wei. In some embodiments, the HIV virus is resistant to raltegravir or elvitegravir.
在一些實施方案中,該HIV病毒是對卡博特韋具有抗性的病毒株,或者說,該病毒株對整合酶抑制劑卡博特韋具有抗性。 In some embodiments, the HIV virus is a cabotevir resistant strain, or the virus strain is resistant to the integrase inhibitor cabotevir.
在一些實施方案中,該HIV病毒是對比克替拉韋或多替拉韋具有抗性的病毒株,或者說,該病毒株對整合酶抑制劑比克替拉韋或多替拉韋具有抗性。在一些實施方案中,該HIV病毒是對整合酶抑制劑具有抗性,該整合酶抑制劑較佳雷特格韋或埃替拉韋。 In some embodiments, the HIV virus is a strain resistant to bictegravir or dolutegravir, or the virus strain is resistant to the integrase inhibitor bictegravir or dolutegravir sex. In some embodiments, the HIV virus is resistant to an integrase inhibitor, preferably raltegravir or elvitegravir.
該HIV病毒是對抗艾滋病藥物具有抗性,該抗艾滋病藥物包括雷特格韋、埃替拉韋、比克替拉韋、多替拉韋、卡博特韋、Trizivir、Nevirapine、恩夫韋肽、艾博韋泰、達蘆那韋、替拉那韋、福沙那韋、阿紮那韋、洛匹那韋、安普那韋、奈非那韋、茚地那韋、利托那韋、沙奎那韋、磷坦姆沙韋、齊多夫定、拉米夫定、阿巴卡韋、依非韋倫、司他夫定、紮西他濱、恩曲他濱、利匹韋林、多拉韋林。 The HIV virus is resistant to anti-AIDS drugs, including raltegravir, elvitegravir, bictegravir, dolutegravir, cabotevir, Trizivir, Nevirapine, enfuvirtide , albovir, darunavir, tipranavir, fosamprenavir, atazanavir, lopinavir, amprenavir, nelfinavir, indinavir, ritonavir , saquinavir, fostamsavir, zidovudine, lamivudine, abacavir, efavirenz, stavudine, zalcitabine, emtricitabine, rilpivir Lin, Doravirine.
在一些實施方案中,該病毒是對整合酶抑制劑具有抗性的病毒株,該病毒株包括HIV整合酶,該HIV整合酶包括Y143C突變、E92Q/N155H兩重突變或者G140S/Y143H/Q148H三重突變。 In some embodiments, the virus is a strain resistant to an integrase inhibitor, the strain comprising HIV integrase comprising the Y143C mutation, the E92Q/N155H double mutation, or the G140S/Y143H/Q148H triplet mutation.
本揭露還提供了式I所示化合物或其可藥用鹽在製備用於抗艾滋病藥物具有抗性的HIV病毒感染的藥物中的用途, The present disclosure also provides the use of the compound represented by formula I or a pharmaceutically acceptable salt thereof in the preparation of a drug for HIV infection with resistance to an anti-AIDS drug,
其中,R1、R2、L、Z1、Z2、Z3、Z4、Y如前所定義。 Wherein, R 1 , R 2 , L, Z 1 , Z 2 , Z 3 , Z 4 , and Y are as defined above.
在一些實施方案中,該抗艾滋病藥物選自整合酶抑制劑、蛋白酶抑制劑、非核苷類反轉錄酶抑制劑或核苷類反轉錄酶抑制劑。 In some embodiments, the anti-AIDS drug is selected from integrase inhibitors, protease inhibitors, non-nucleoside reverse transcriptase inhibitors or nucleoside reverse transcriptase inhibitors.
在一些實施方案中,該蛋白酶抑制包括但不限於達蘆那韋、替拉那韋、福沙那韋、阿紮那韋、洛匹那韋、安普那韋、奈非那韋、茚地那韋、利托那韋、沙奎那韋。 In some embodiments, the protease inhibition includes, but is not limited to, darunavir, tipranavir, fosamprenavir, atazanavir, lopinavir, amprenavir, nelfinavir, indinavir Navir, ritonavir, saquinavir.
在一些實施方案中,該非核苷類反轉錄酶抑制劑或核苷類反轉錄酶抑制劑包括但不限於齊多夫定、拉米夫定、阿巴卡韋、依非韋倫、司他夫定、紮西他濱、恩曲他濱、利匹韋林、多拉韋林。 In some embodiments, the non-nucleoside reverse transcriptase inhibitor or nucleoside reverse transcriptase inhibitor includes, but is not limited to, zidovudine, lamivudine, abacavir, efavirenz, lecithin Fudine, zalcitabine, emtricitabine, rilpivirine, doravirine.
在一些實施方案中,該整合酶抑制劑選自但不限於雷特格韋、埃替拉韋、比克替拉韋、多替拉韋、卡博特韋,較佳雷特格韋或埃替拉韋。 In some embodiments, the integrase inhibitor is selected from but not limited to raltegravir, elvitegravir, bictegravir, dolutegravir, cabotevir, preferably raltegravir or elvitegravir Telavir.
在另一些實施方案中,該抗艾滋病藥物選自雷特格韋、埃替拉韋、比克替拉韋、多替拉韋、卡博特韋、Trizivir、Nevirapine、恩夫韋肽、艾博韋泰、達蘆那韋、替拉那韋、福沙那韋、阿紮那韋、洛匹那韋、安普那韋、奈非那韋、茚地那韋、利托那韋、沙奎那韋、磷坦姆沙韋。 In other embodiments, the anti-AIDS drug is selected from the group consisting of raltegravir, elvitegravir, bictegravir, dolutegravir, cabotevir, Trizivir, Nevirapine, enfuvirtide, albumin Vitavir, darunavir, tipranavir, fosamprenavir, atazanavir, lopinavir, amprenavir, nelfinavir, indinavir, ritonavir, saqui Navir, fostamsavir.
在一些實施方案中,該抗艾滋病藥物選自雷特格韋或埃替拉韋。 In some embodiments, the anti-AIDS drug is selected from raltegravir or elvitegravir.
本揭露還提供了式I所示化合物或其可藥用鹽在製備用於對整合酶抑制劑具有抗性的HIV病毒感染的藥物中的用途。 The present disclosure also provides use of the compound represented by formula I or a pharmaceutically acceptable salt thereof in the preparation of a medicament for HIV infection resistant to an integrase inhibitor.
本揭露還提供了式I所示化合物或其可藥用鹽在製備用於對對非核苷類反轉錄酶抑制劑或核苷類反轉錄酶抑制劑具有抗性的HIV病毒感染的藥物中的用途。 The present disclosure also provides a compound of formula I or a pharmaceutically acceptable salt thereof in the preparation of a medicament for HIV infection resistant to non-nucleoside reverse transcriptase inhibitors or nucleoside reverse transcriptase inhibitors use.
本揭露還提供了式I所示化合物或其可藥用鹽在製備用於對蛋白酶抑制劑具有抗性的HIV病毒感染的藥物中的用途。 The present disclosure also provides the use of the compound represented by formula I or a pharmaceutically acceptable salt thereof in the preparation of a medicament for HIV infection resistant to protease inhibitors.
本揭露所述HIV病毒感染包括HIV-1病毒感染。 The HIV viral infection described in the present disclosure includes HIV-1 viral infection.
另一方面,本揭露中所述化合物可藥用鹽選自無機鹽或有機鹽。 In another aspect, the pharmaceutically acceptable salts of the compounds described in this disclosure are selected from inorganic or organic salts.
另一方面,本揭露化合物可以存在特定的幾何或立體異構體形式。本揭露設想所有的這類化合物,包括順式和反式異構體、(-)-和(+)-對對映體、(R)-和(S)-對映體、非對映異構體、(D)-異構體、(L)-異構體,及其外消旋混合物和其他混合物,例如對映異構體或非對映體富集的混合物,所有這些混合物都屬於本揭露的範圍之內。烷基等取代基中可存在另外的不對稱碳原子。所有這些異構體以及它們的混合物,均包括在本揭露的範圍之內。 On the other hand, compounds of the present disclosure may exist in specific geometric or stereoisomeric forms. This disclosure contemplates all such compounds, including cis and trans isomers, (-)- and (+)-enantiomers, (R)- and (S)-enantiomers, diastereomers isomers, (D)-isomers, (L)-isomers, and racemic and other mixtures thereof, such as enantiomerically or diastereomerically enriched mixtures, all of which belong to within the scope of this disclosure. Additional asymmetric carbon atoms may be present in substituents such as alkyl. All such isomers, as well as mixtures thereof, are included within the scope of this disclosure.
另外,本揭露的化合物和中間體還可以以不同的互變異構體形式存在,並且所有這樣的形式包含於本揭露的範圍內。術語“互變異構體”或“互變異構體形式”是指可經由低能壘互變的不同能量的結構異構體。例如,質子互變異構體(也稱為質子轉移互變異構體)包括經由質子遷移的互變,如酮-烯醇及亞胺-烯胺異構化。 In addition, the compounds and intermediates of the present disclosure may also exist in different tautomeric forms, and all such forms are included within the scope of the present disclosure. The term "tautomer" or "tautomeric form" refers to structural isomers of different energies that are interconvertible via a low energy barrier. For example, proton tautomers (also known as proton tautomers) include interconversions via migration of protons, such as keto-enol and imine-enamine isomerizations.
本揭露化合物可以是不對稱的,例如,具有一個或多個立體異構體。除非另有說明,所有立體異構體都包括,如對映異構體和非對映異構體。本揭露的含有不對稱碳原子的化合物可以以光學活性純的形式或外消旋形式被分離出來。光學活性純的形式可以從外消旋混合物拆分,或藉由使用手性原料或手性試劑合成。 Compounds of the present disclosure may be asymmetric, eg, have one or more stereoisomers. Unless otherwise specified, all stereoisomers include, such as enantiomers and diastereomers. Compounds of the present disclosure containing asymmetric carbon atoms can be isolated in optically pure or racemic forms. Optically pure forms can be resolved from racemic mixtures or synthesized by using chiral starting materials or chiral reagents.
可以藉由的手性合成或手性試劑或者其他常規技術製備光學活性的(R)-和(S)-異構體以及D和L異構體。如果想得到本揭露某化合物的一種對映體,可以藉由不對稱合成或者具有手性助劑的衍生作用來製備,其中將所得非對映體混合物分離,並且輔助基團裂開以提供純的所需對映異構體。或者,當分 子中含有鹼性官能團(如胺基)或酸性官能團(如羧基)時,與適當的光學活性的酸或鹼形成非對映異構體的鹽,然後藉由本領域所公知的常規方法進行非對映異構體拆分,然後回收得到純的對映體。此外,對映異構體和非對映異構體的分離通常是藉由使用色譜法完成的,該色譜法採用手性固定相,並視需要地與化學衍生法相結合(例如由胺生成胺基甲酸鹽)。 Optically active (R)- and (S)-isomers, as well as D and L isomers, can be prepared by chiral synthesis or chiral reagents or other conventional techniques. If an enantiomer of a compound of the present disclosure is desired, it can be prepared by asymmetric synthesis or derivatization with a chiral auxiliary, wherein the resulting mixture of diastereomers is separated and the auxiliary group is cleaved to provide pure desired enantiomer. Or, when points When the molecule contains a basic functional group (such as an amine group) or an acidic functional group (such as a carboxyl group), a diastereoisomeric salt is formed with an appropriate optically active acid or base, and then the non-stereomeric salt is carried out by conventional methods known in the art. The enantiomers are resolved and then recovered to give the pure enantiomer. In addition, separation of enantiomers and diastereomers is usually accomplished by the use of chromatography employing a chiral stationary phase, optionally combined with chemical derivatization (eg from amine to amine carboxylate).
雖然為簡便起見將全部上述結構式畫成某些異構體形式,但是本發明可以包括所有的異構體,如互變異構體、旋轉異構體、幾何異構體、非對映異構體、外消旋體和對映異構體。 Although all of the above formulae are drawn as certain isomeric forms for simplicity, the present invention encompasses all isomers such as tautomers, rotamers, geometric isomers, diastereomers isomers, racemates and enantiomers.
互變異構體是有機化合物的結構異構體,藉由被稱為互變異構化的化學反應容易相互轉化。這種反應常導致氫原子或質子的形式遷移,伴隨著單鍵和鄰近的雙鍵的轉換。一些常見的互變異構對為:酮-烯醇、內醯胺-內醯亞胺。內醯胺-內醯亞胺平衡實例是在如下所示的A和B之間。 Tautomers are structural isomers of organic compounds that are readily interconverted by a chemical reaction called tautomerization. This reaction often results in the transfer of hydrogen atoms or protons in the form of a single bond and an adjacent double bond. Some common tautomeric pairs are: keto-enol, lactam-lactamide. An example of a lactam-lactamine equilibrium is between A and B as shown below.
本發明中的所有化合物可以被畫成A型或B型。所有的互變異構形式在本發明的範圍內。化合物的命名不排除任何互變異構體。” All compounds in the present invention can be drawn as A or B type. All tautomeric forms are within the scope of the present invention. The naming of compounds does not exclude any tautomers. "
本揭露還包括一些與本文中記載的那些相同的,但一個或多個原子被原子量或質量數不同於自然中通常發現的原子量或質量數的原子置換的同位素標記的本揭露化合物。可結合到本揭露化合物的同位素的實例包括氫、碳、氮、氧、磷、硫、氟、碘和氯的同位素,諸如分別為2H、3H、11C、13C、14C、13N、15N、15O、17O、18O、31P、32P、35S、18F、123I、125I和36Cl等。 The present disclosure also includes certain isotopically-labeled compounds of the present disclosure that are identical to those described herein, but wherein one or more atoms are replaced by an atom having an atomic weight or mass number different from that normally found in nature. Examples of isotopes that can be incorporated into the compounds of the present disclosure include isotopes of hydrogen, carbon, nitrogen, oxygen, phosphorus, sulfur, fluorine, iodine, and chlorine, such as 2H, 3H , 11C , 13C , 14C , 13 , respectively N, 15 N, 15 O, 17 O, 18 O, 31 P, 32 P, 35 S, 18 F, 123 I, 125 I and 36 Cl and the like.
除另有說明,當一個位置被特別地指定為氘(D)時,該位置應理解為具有大於氘的天然豐度(其為0.015%)至少1000倍的豐度的氘(即,至少10%的氘摻入)。示例中化合物的具有大於氘的天然豐度可以是至少1000倍的豐度的氘、至少2000倍的豐度的氘、至少3000倍的豐度的氘、至少4000倍的豐度的氘、至少5000倍的豐度的氘、至少6000倍的豐度的氘或更高豐度的氘。本揭露還包括各種氘化形式的式(I)化合物。與碳原子連接的各個可用的氫原子可獨立地被氘原子替換。所屬技術領域具有通常知識者能夠參考相關文獻合成氘化形式的式(I)化合物。在製備氘代形式的式(I)化合物時可使用市售的氘代起始物質,或它們可使用常規技術採用氘代試劑合成,氘代試劑包括但不限於氘代硼烷、三氘代硼烷四氫呋喃溶液、氘代氫化鋰鋁、氘代碘乙烷和氘代碘甲烷等。 Unless otherwise stated, when a position is specifically designated as deuterium (D), the position is understood to have an abundance of deuterium (ie, at least 10 times greater than the natural abundance of deuterium, which is 0.015%), at least 1000 times greater % deuterium incorporation). Exemplary compounds having natural abundance greater than deuterium may be at least 1000 times more abundant deuterium, at least 2000 times more abundant deuterium, at least 3000 times more abundant deuterium, at least 4000 times more abundant deuterium, at least 4000 times more abundant 5000 times more abundant deuterium, at least 6000 times more abundant deuterium or more abundant deuterium. The present disclosure also includes compounds of formula (I) in various deuterated forms. Each available hydrogen atom attached to a carbon atom can be independently replaced by a deuterium atom. One of ordinary skill in the art can refer to the relevant literature to synthesize the compound of formula (I) in deuterated form. Commercially available deuterated starting materials can be used in the preparation of deuterated forms of compounds of formula (I), or they can be synthesized using conventional techniques using deuterated reagents including, but not limited to, deuterated borane, trideuterated Borane tetrahydrofuran solution, deuterated lithium aluminum hydride, deuterated iodoethane and deuterated iodomethane, etc.
“視需要地”或“視需要”是指意味著隨後所描述的事件或環境可以但不必發生,該說明包括該事件或環境發生或不發生的場合。例如“視需要的被鹵素或者氰基取代的C1-6烷基”是指鹵素或者氰基可以但不必須存在,該說明包括烷基被鹵素或者氰基取代的情形和烷基不被鹵素和氰基取代的情形。 "Optionally" or "optionally" is meant to mean that the subsequently described event or circumstance can but need not occur, and that the description includes instances where the event or circumstance does or does not occur. For example, "optionally substituted C 1-6 alkyl substituted by halogen or cyano" means that halogen or cyano may but need not be present, and the description includes the case where the alkyl is substituted by halogen or cyano and the case where the alkyl is not substituted by halogen and cyano substitution.
本發明所述化合物的化學結構中,鍵“/”表示未指定構型,即如果化學結構中存在手性異構體,鍵“/”可以為“”或“”,或者同時包含“”和“”兩種構型。 In the chemical structure of the compounds described in the present invention, the bond "/" represents an unspecified configuration, that is, if there is a chiral isomer in the chemical structure, the bond "/" can be "/" "or" ", or both " "and" "Two configurations.
術語解釋: Terminology Explanation:
本揭露中所述“有效量”或“有效治療量”包含足以改善或預防醫學病症的症狀或病症的量。有效量還意指足以允許或促進診斷的量。用於特定患者或獸醫學受試者的有效量可依據以下因素而變化:如待治療的病症、患者的總體健康情 況、給藥的方法途徑和劑量以及副作用嚴重性。有效量可以是避免顯著副作用或毒性作用的最大劑量或給藥方案。 An "effective amount" or "therapeutically effective amount" as used in this disclosure includes an amount sufficient to ameliorate or prevent a symptom or condition of a medical condition. An effective amount also means an amount sufficient to allow or facilitate diagnosis. The effective amount for a particular patient or veterinary subject may vary depending on factors such as the condition being treated, the patient's general health condition, route of administration and dosage, and severity of side effects. An effective amount can be the maximum dose or dosing regimen that avoids significant side effects or toxic effects.
“烷基”指飽和的脂族烴基團,包括1至20個碳原子的直鏈和支鏈基團。含有1至6個碳原子的烷基。非限制性實施例包括甲基、乙基、正丙基、異丙基、正丁基、異丁基、第三丁基、第二丁基、正戊基、1,1-二甲基丙基、1,2-二甲基丙基、2,2-二甲基丙基及其各種支鏈異構體等。烷基可以是取代的或未取代的,當被取代時,取代基可以在任何可使用的連接點上被取代,較佳一個或多個以下基團,獨立地選自氘、鹵素、硝基、羥基、氰基、C1-6烷基、C1-6烷氧基。 "Alkyl" refers to saturated aliphatic hydrocarbon groups, including straight and branched chain groups of 1 to 20 carbon atoms. An alkyl group containing 1 to 6 carbon atoms. Non-limiting examples include methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, tert-butyl, sec-butyl, n-pentyl, 1,1-dimethylpropyl base, 1,2-dimethylpropyl, 2,2-dimethylpropyl and various branched chain isomers thereof. Alkyl groups may be substituted or unsubstituted, and when substituted, the substituents may be substituted at any available point of attachment, preferably one or more of the following groups, independently selected from deuterium, halogen, nitro , hydroxyl, cyano, C 1-6 alkyl, C 1-6 alkoxy.
術語“環烷基”指飽和或部分不飽和單環或多環環狀烴取代基,環烷基環包含3至20個碳原子,較佳包含3至7個碳原子。單環環烷基的非限制性實例包括環丙基、環丁基、環戊基、環戊烯基、環己基、環己烯基、環己二烯基等;多環環烷基包括螺環、稠環和橋環的環烷基。環烷基可以是取代的或未取代的,當被取代時,取代基可以在任何可使用的連接點上被取代,較佳一個或多個以下基團,獨立地選自氘、鹵素、硝基、羥基、氰基、C1-6烷基、C1-6烷氧基。 The term "cycloalkyl" refers to a saturated or partially unsaturated monocyclic or polycyclic cyclic hydrocarbon substituent, the cycloalkyl ring containing 3 to 20 carbon atoms, preferably 3 to 7 carbon atoms. Non-limiting examples of monocyclic cycloalkyl groups include cyclopropyl, cyclobutyl, cyclopentyl, cyclopentenyl, cyclohexyl, cyclohexenyl, cyclohexadienyl, and the like; polycyclic cycloalkyl groups include spiro Ring, fused and bridged cycloalkyl groups. Cycloalkyl may be substituted or unsubstituted, and when substituted, the substituents may be substituted at any available point of attachment, preferably one or more of the following groups, independently selected from deuterium, halogen, nitro group, hydroxyl group, cyano group, C 1-6 alkyl group, C 1-6 alkoxy group.
該環烷基環可以稠合於芳基或雜芳基環上,其中與母體結構連接在一起的環為環烷基,非限制性實例包括茚滿基、四氫萘基、苯并環庚烷基等。環烷基可以是視需要取代的或非取代的,當被取代時,取代基較佳為一個或多個以下基團,其獨立地選自氘、鹵素、硝基、羥基、氰基、C1-6烷基、C1-6烷氧基。 The cycloalkyl ring can be fused to an aryl or heteroaryl ring, wherein the ring attached to the parent structure is a cycloalkyl, non-limiting examples include indanyl, tetrahydronaphthyl, benzocycloheptyl Alkyl etc. Cycloalkyl may be optionally substituted or unsubstituted, and when substituted, the substituents are preferably one or more of the following groups independently selected from deuterium, halogen, nitro, hydroxyl, cyano, C 1-6 alkyl, C 1-6 alkoxy.
術語“雜環烷基(Heterocycloalkyl)”指飽和或部分不飽和單環或多環環狀烴取代基,其包含3至20個環原子,其中一個或多個環原子為選自氮、氧或S(O)m(其中m是整數0至2)的雜原子,但不包括-O-O-、-O-S-或-S-S-的環部分,其餘環原子為碳。較佳包含3至12個環原子,其中1~4個是雜原子;更 佳包含3至7個環原子。單環雜環烷基的非限制性實例包括吡咯烷基、咪唑烷基、四氫呋喃基、四氫噻吩基、二氫咪唑基、二氫呋喃基、二氫吡唑基、二氫吡咯基、哌啶基、哌嗪基、嗎啉基、硫嗎啉基、高哌嗪基等。多環雜環烷基包括螺環、稠環和橋環的雜環烷基。“雜環烷基”非限制性實例包括: The term "Heterocycloalkyl" refers to a saturated or partially unsaturated monocyclic or polycyclic cyclic hydrocarbon substituent containing from 3 to 20 ring atoms, one or more of which is selected from nitrogen, oxygen or Heteroatoms of S(O) m (where m is an integer from 0 to 2), excluding ring moieties of -OO-, -OS- or -SS-, the remaining ring atoms being carbon. It preferably contains 3 to 12 ring atoms, of which 1 to 4 are heteroatoms; more preferably contains 3 to 7 ring atoms. Non-limiting examples of monocyclic heterocycloalkyl include pyrrolidinyl, imidazolidinyl, tetrahydrofuranyl, tetrahydrothienyl, dihydroimidazolyl, dihydrofuranyl, dihydropyrazolyl, dihydropyrrolyl, piper pyridyl, piperazinyl, morpholinyl, thiomorpholinyl, homopiperazinyl, etc. Polycyclic heterocycloalkyl groups include spiro, fused and bridged ring heterocycloalkyl groups. Non-limiting examples of "heterocycloalkyl" include:
該雜環烷基環可以稠合於芳基或雜芳基環上,其中與母體結構連接在一起的環為雜環烷基,其非限制性實例包括: The heterocycloalkyl ring can be fused to an aryl or heteroaryl ring, wherein the ring attached to the parent structure is a heterocycloalkyl, non-limiting examples of which include:
和等。 and Wait.
雜環烷基可以是視需要取代的或非取代的,當被取代時,取代基較佳為一個或多個以下基團,其獨立地選自氘、鹵素、硝基、羥基、氰基、C1-6烷基、C1-6烷氧基。 Heterocycloalkyl can be optionally substituted or unsubstituted, and when substituted, the substituents are preferably one or more of the following groups independently selected from deuterium, halogen, nitro, hydroxyl, cyano, C 1-6 alkyl, C 1-6 alkoxy.
術語“芳基”指具有共軛的π電子體系的6至14員全碳單環或稠合多環(也就是共享毗鄰碳原子對的環)基團,較佳為6至12員,例如苯基和萘 基。該芳基環可以稠合於雜芳基、雜環烷基或環烷基環上,其中與母體結構連接在一起的環為芳基環,其非限制性實例包括: The term "aryl" refers to a 6- to 14-membered all-carbon monocyclic or fused polycyclic (ie, rings sharing adjacent pairs of carbon atoms) group having a conjugated pi-electron system, preferably 6 to 12 membered, e.g. Phenyl and Naphthalene base. The aryl ring can be fused to a heteroaryl, heterocycloalkyl or cycloalkyl ring, wherein the ring linked to the parent structure is an aryl ring, non-limiting examples of which include:
芳基可以是取代的或非取代的,當被取代時,取代基較佳為一個或多個以下基團,其獨立地選自氘、鹵素、硝基、羥基、氰基、C1-6烷基、C1-6烷氧基,該烷基或烷氧基視需要被鹵素、硝基、氰基或C1-6烷氧基所取代。 Aryl can be substituted or unsubstituted, and when substituted, the substituents are preferably one or more of the following groups independently selected from deuterium, halogen, nitro, hydroxyl, cyano, C 1-6 Alkyl, C 1-6 alkoxy, optionally substituted by halogen, nitro, cyano or C 1-6 alkoxy.
術語“雜芳基”指包含1至4個雜原子、5至14個環原子的雜芳族體系,其中雜原子選自氧、硫和氮。雜芳基較佳為6至12員,更佳為5員或6員。例如。其非限制性實例包括:咪唑基、呋喃基、噻吩基、噻唑基、吡唑基、噁唑基、吡咯基、四唑基、吡啶基、嘧啶基、噻二唑、吡嗪,,,,等等。 The term "heteroaryl" refers to a heteroaromatic system comprising 1 to 4 heteroatoms, 5 to 14 ring atoms, wherein the heteroatoms are selected from oxygen, sulfur and nitrogen. The heteroaryl group is preferably 6 to 12 members, more preferably 5 or 6 members. E.g. Non-limiting examples thereof include: imidazolyl, furyl, thienyl, thiazolyl, pyrazolyl, oxazolyl, pyrrolyl, tetrazolyl, pyridyl, pyrimidinyl, thiadiazole, pyrazine, , , ,and many more.
該雜芳基環可以稠合於芳基、雜環烷基或環烷基環上,其中與母體結構連接在一起的環為雜芳基環,其非限制性實例包括: The heteroaryl ring can be fused to an aryl, heterocycloalkyl or cycloalkyl ring, wherein the ring linked to the parent structure is a heteroaryl ring, non-limiting examples of which include:
雜芳基可以是視需要取代的或非取代的,當被取代時,取代基較佳為一個或多個以下基團,其獨立地選自氘、鹵素、硝基、羥基、氰基、C1-6烷基、C1-6烷氧基,該烷基或烷氧基視需要被鹵素、硝基、氰基或C1-6烷氧基所取代。 Heteroaryl groups can be optionally substituted or unsubstituted, and when substituted, the substituents are preferably one or more of the following groups independently selected from deuterium, halogen, nitro, hydroxyl, cyano, C 1-6 alkyl, C 1-6 alkoxy, the alkyl or alkoxy is optionally substituted by halogen, nitro, cyano or C 1-6 alkoxy.
術語“烷氧基”指-O-(烷基)和-O-(非取代的環烷基),其中烷基的定義如上所述。烷氧基的非限制性實例包括:甲氧基、乙氧基、丙氧基、丁氧基、環丙氧基、環丁氧基、環戊氧基、環己氧基。烷氧基可以是視需要取代的或非取代的,當被取代時,取代基較佳為一個或多個以下基團,其獨立地選自氘、鹵素、硝基、羥基、氰基、C1-6烷基、C1-6烷氧基。 The term "alkoxy" refers to -O-(alkyl) and -O-(unsubstituted cycloalkyl), wherein alkyl is as defined above. Non-limiting examples of alkoxy groups include: methoxy, ethoxy, propoxy, butoxy, cyclopropoxy, cyclobutoxy, cyclopentyloxy, cyclohexyloxy. Alkoxy can be optionally substituted or unsubstituted, and when substituted, the substituents are preferably one or more of the following groups independently selected from deuterium, halogen, nitro, hydroxyl, cyano, C 1-6 alkyl, C 1-6 alkoxy.
術語“雜環”指構成環的原子除碳原子外還有其他原子,其包括雜環烷基和雜芳環。 The term "heterocycle" refers to the atoms that make up the ring in addition to carbon atoms, and includes heterocycloalkyl and heteroaromatic rings.
術語“羥基”指-OH基團。 The term "hydroxy" refers to the -OH group.
術語“鹵素”指氟、氯、溴或碘。 The term "halogen" refers to fluorine, chlorine, bromine or iodine.
術語“氰基”指-CN。 The term "cyano" refers to -CN.
術語“硝基”指-NO2。 The term "nitro" refers to -NO2 .
“取代的”指基團中的一個或多個氫原子,較佳為最多5個,更佳為1~3個氫原子彼此獨立地被相應數目的取代基取代。不言而喻,取代基僅處在它們的可能的化學位置,所屬技術領域具有通常知識者能夠在不付出過多努力的情況下確定(藉由實驗或理論)可能或不可能的取代。 "Substituted" means that one or more hydrogen atoms in a group, preferably up to 5, more preferably 1 to 3 hydrogen atoms are independently substituted with a corresponding number of substituents. It goes without saying that the substituents are only in their possible chemical positions, and those of ordinary skill in the art can determine (either experimentally or theoretically) possible or impossible substitutions without undue effort.
以下結合實施例進一步描述本揭露中,但這些實施例並非限制本揭露中的範圍。 The present disclosure is further described below with reference to the embodiments, but these embodiments do not limit the scope of the present disclosure.
本揭露中實施例中未註明具體條件的實驗方法,通常按照常規條件,或按照原料或商品製造廠商所建議的條件。未註明具體來源的試劑,為市場購買的常規試劑。 The experimental methods for which specific conditions are not indicated in the embodiments of the present disclosure are generally based on conventional conditions or conditions suggested by raw material or commodity manufacturers. Reagents with no specific source indicated are conventional reagents purchased in the market.
化合物的結構是藉由核磁共振(NMR)或/和質譜(MS)來確定的。NMR位移(δ)以10-6(ppm)的單位給出。NMR的測定是用Bruker AVANCE-400核磁儀,測定溶劑為氘代二甲基亞碸(DMSO-d 6 ),氘代氯仿(CDCl3),氘代甲醇(Methanol-d 4 ),內標為四甲基矽烷(TMS)。 The structures of the compounds were determined by nuclear magnetic resonance (NMR) or/and mass spectrometry (MS). NMR shifts ([delta]) are given in units of 10<" 6 > (ppm). NMR was measured by Bruker AVANCE-400 nuclear magnetic instrument, and the solvent was deuterated dimethyl sulfoxide (DMSO- d 6 ), deuterated chloroform (CDCl 3 ), deuterated methanol (Methanol- d 4 ), and the internal standard was Tetramethylsilane (TMS).
MS的測定用Waters Micromass Quattro micro API三重四級杆質譜儀,以正/負離子模式掃描,質量掃描範圍為120~1300。 MS was measured with a Waters Micromass Quattro micro API triple quadrupole mass spectrometer, scanned in positive/negative ion mode, with a mass scan range of 120–1300.
薄層層析矽膠板使用煙臺黃海HSGF254矽膠板,薄層色譜法(TLC)使用矽膠板採用規格是0.2mm±0.03mm,薄層層析分離純化產品採用的規格是0.4mm-0.5mm。 The thin layer chromatography silica gel plate uses Yantai Huanghai HSGF254 silica gel plate, the thin layer chromatography (TLC) uses the silica gel plate with a specification of 0.2mm±0.03mm, and the thin layer chromatography separation and purification product adopts a specification of 0.4mm-0.5mm.
快速管柱純化系統使用Combiflash Rf150(TELEDYNE ISCO)或者Isolara one(Biotage)。 The flash column purification system used Combiflash Rf150 (TELEDYNE ISCO) or Isolara one (Biotage).
正向管柱層析一般使用煙臺黃海矽膠200~300目或300~400目矽膠為載體,或者使用常州三泰預填預填超純正相矽膠管柱(40-63μm,60g,24g,40g,120g或其它規格)。 Forward column chromatography generally uses Yantai Huanghai silica gel 200~300 mesh or 300~400 mesh silica gel as the carrier, or Changzhou Santai pre-packed pre-packed ultra-pure normal phase silica gel column (40-63μm, 60g, 24g, 40g, 120g or other specifications).
本揭露中的已知的起始原料可以採用或按照本領域已知的方法來合成,或可購買自上海泰坦科技,ABCR GmbH & Co.KG,Acros Organics, Aldrich Chemical Company,韶遠化學科技(Accela ChemBio Inc),畢得醫藥等公司。 The known starting materials in the present disclosure can be synthesized using or according to methods known in the art, or can be purchased from Shanghai Titan Technology, ABCR GmbH & Co.KG, Acros Organics, Aldrich Chemical Company, Accela ChemBio Inc, Bide Medicine and other companies.
實施例中無特殊說明,反應能夠均在氮氣氛下進行。 There is no special description in the examples, and the reactions can all be carried out under nitrogen atmosphere.
氮氣氛是指反應瓶連接一個約1L容積的氮氣氣球。 Nitrogen atmosphere means that the reaction flask is connected to a nitrogen balloon with a volume of about 1 L.
氫氣氛是指反應瓶連接一個約1L容積的氫氣氣球。 Hydrogen atmosphere means that the reaction flask is connected to a hydrogen balloon with a volume of about 1 L.
氫氣是由上海全浦科學儀器公司QPH-1L型氫氣發生儀製得。 Hydrogen was produced by a QPH-1L hydrogen generator from Shanghai Quanpu Scientific Instrument Company.
氮氣氛或氫化氛通常抽真空,充入氮氣或氫氣,重複操作3次。 The nitrogen atmosphere or hydrogenation atmosphere is usually evacuated, filled with nitrogen or hydrogen, and the operation is repeated 3 times.
實施例中無特殊說明,溶液是指水溶液。 There is no special description in the examples, and the solution refers to an aqueous solution.
實施例中無特殊說明,反應的溫度為室溫,為20℃~30℃。 There is no special description in the examples, and the temperature of the reaction is room temperature, which is 20°C to 30°C.
實施例中的反應進程的監測採用薄層色譜法(TLC)。 The progress of the reaction in the examples was monitored by thin layer chromatography (TLC).
實施例1 Example 1
(3S,7S)-N-(2,4-二氟苯甲基)-12-羥基-3-甲基-1,11-二羰基-1,4,5,6,7,11-六氫-3H-2,7-亞甲基吡啶并[1,2-a][1,4]重氮基壬英-10-甲醯胺 ( 3S,7S )-N-(2,4 - difluorobenzyl)-12-hydroxy-3-methyl-1,11-dicarbonyl-1,4,5,6,7,11-hexahydro -3H-2,7-Methylenepyrido[1,2-a][1,4] diazononin -10-carbamide
第一步 first step
2-甲基5-甲基3-甲氧基-4-羰基-4H-吡喃-2,5-二羧酸酯4 a 2-Methyl 5-methyl 3-methoxy-4-carbonyl-4H-pyran-2,5-dicarboxylate 4 a
將4-甲氧基乙醯乙酸甲酯(17.71mL,136.85mmol)和N,N-二甲基甲醯胺二甲縮醛(18.32mL,136.85mmol)的混合物在85℃加熱攪拌,將草酸二甲酯(32.32g,273.70mmol)加入反應液中,攪拌反應至反應完全,降至室溫,將含量30%的甲醇鈉甲醇溶液(52.1542mL,273.7082mmol)加入到反應液中,繼續攪拌反應2-4h,用50ml醋酸淬滅反應,經C18反相色譜管柱純化得到標題化合物4a(15.8g,產率48%)。 A mixture of methyl 4-methoxyacetate acetate (17.71 mL, 136.85 mmol) and N,N-dimethylformamide dimethyl acetal (18.32 mL, 136.85 mmol) was heated and stirred at 85 °C, and the oxalic acid Dimethyl ester (32.32 g, 273.70 mmol) was added to the reaction solution, and the reaction was stirred until the reaction was complete, then lowered to room temperature, and a methanol solution of sodium methoxide (52.1542 mL, 273.7082 mmol) with a content of 30% was added to the reaction solution, and the stirring was continued. The reaction was carried out for 2-4 h, quenched with 50 ml of acetic acid, and purified by C18 reverse phase chromatography column to obtain the title compound 4a (15.8 g, 48% yield).
MS(ESI)m/z 265.3[M+Na]+ MS(ESI)m/z 265.3[M+Na] +
第二步 second step
6-[(2-乙氧基-2-氧乙基)胺基]己酸乙酯4d Ethyl 6-[(2-ethoxy-2-oxoethyl)amino]hexanoate 4d
將5-氧己酸乙酯(3g,18.96mmol)溶於10mL甲醇中,再依次加入2-胺基乙酸乙酯鹽酸鹽(2.65g,18.96mmol),三乙胺(2.64mL,18.96mmol)和氰基硼氫化鈉(2.38g,37.93mmol),℃室溫攪拌反應18-24h,用20ml飽和碳酸氫鈉淬滅反應,濃縮混合溶液,加入二氯甲烷(20mL×2),飽和鹽水洗滌,無水硫酸鈉乾燥,濃縮得到標題化合物4d(4g),直接用於下一步。 Ethyl 5-oxohexanoate (3 g, 18.96 mmol) was dissolved in 10 mL of methanol, followed by adding 2-aminoethyl acetate hydrochloride (2.65 g, 18.96 mmol), triethylamine (2.64 mL, 18.96 mmol) ) and sodium cyanoborohydride (2.38 g, 37.93 mmol), stirred at room temperature for 18-24 h, quenched with 20 ml of saturated sodium bicarbonate, concentrated the mixed solution, added dichloromethane (20 mL×2), saturated brine Washed, dried over anhydrous sodium sulfate, and concentrated to give the title compound 4d (4 g), which was used directly in the next step.
MS(ESI)m/z 246.2[M+H]+ MS(ESI)m/z 246.2[M+H] +
第三步 third step
5-((第三-丁氧基羰基)(2-乙氧基-2-羰基乙基)胺基)己酸乙酯4e Ethyl 5-((3-butoxycarbonyl)(2-ethoxy-2-carbonylethyl)amino)hexanoate 4e
將化合物4d(4g,16.30mmol)溶於20mL二氯甲烷中,並依次加入二碳酸二第三丁酯(4.19mL,19.57mmol)和三乙胺(6.80mL,48.92mmol),℃室溫下反應4-6小時,加水淬滅反應,分液,水洗,無水硫酸鈉乾燥,濃縮得到標題化合物4e(6g),直接用於下一步。 Compound 4d (4 g, 16.30 mmol) was dissolved in 20 mL of dichloromethane, and di-tert-butyl dicarbonate (4.19 mL, 19.57 mmol) and triethylamine (6.80 mL, 48.92 mmol) were added successively, at room temperature The reaction was carried out for 4-6 hours, water was added to quench the reaction, the mixture was separated, washed with water, dried over anhydrous sodium sulfate, and concentrated to obtain the title compound 4e (6 g), which was directly used in the next step.
MS(ESI)m/z 368.3[M+H]+ MS(ESI)m/z 368.3[M+H] +
第四步 the fourth step
1-(第三-丁基)4-乙基7-甲基-3-羰基吖庚環-1,4-二羧酸酯4f 1-(Third-butyl) 4-ethyl 7-methyl-3-carbonylazepan-1,4-dicarboxylate 4f
1-(第三-丁基)2-乙基7-甲基-3-羰基吖庚環-1,2-二羧酸酯4g 1-(Third-butyl) 2-ethyl 7-methyl-3-carbonyl azepan-1,2-dicarboxylate 4g
將化合物4e(4g,11.5797mmol)溶於10mL甲苯中,將第三丁醇鈉(1.78g,18.53mmol)加入到反應,反應液在110℃反應4小時。濃縮直接得到粗品標題化合物4f和化合物4g的混合物(6g),直接用於下一步。 Compound 4e (4 g, 11.5797 mmol) was dissolved in 10 mL of toluene, sodium tertiary butoxide (1.78 g, 18.53 mmol) was added to the reaction, and the reaction solution was reacted at 110° C. for 4 hours. Concentration directly gave a mixture of crude title compound 4f and compound 4g (6g), which was used directly in the next step.
MS(ESI)m/z 322.3[M+Na]+ MS(ESI)m/z 322.3[M+Na] +
第五步 the fifth step
第三-丁基2-甲基-6-羰基吖庚環-1-羧酸酯4h 3-Butyl 2-methyl-6-carbonylazepine-1-carboxylate 4h
將化合物4f和化合物4g的混合粗品(6g)溶於10mL水和10mL四氫呋喃的混合溶液中,再加入氫氧化鈉(2.40g,60.13mmol),反應在70℃下反應12小時。用乙酸乙酯(20mL×2)萃取,無水硫酸鈉乾燥,濃縮得到粗品。粗品藉由C18反相進行純化,得到標題化合物4h(0.9g,四步總產率31%)。 The mixed crude product (6g) of compound 4f and compound 4g was dissolved in a mixed solution of 10 mL of water and 10 mL of tetrahydrofuran, sodium hydroxide (2.40 g, 60.13 mmol) was added, and the reaction was carried out at 70° C. for 12 hours. Extract with ethyl acetate (20 mL×2), dry over anhydrous sodium sulfate, and concentrate to obtain crude product. The crude product was purified by C18 reverse phase to give the title compound 4h (0.9 g, 31% overall yield over four steps).
MS(ESI)m/z 250.2[M+H]+ MS(ESI)m/z 250.2[M+H] +
第六步 Step 6
第三-丁基6-胺基-2-甲基吖庚環-1-羧酸酯4i 3-butyl 6-amino-2-methylazepan-1-carboxylate 4i
將化合物11(0.9g,3.96mmol)溶於5毫升甲醇中,然後依次加入甲酸銨(2.50g,39.60mmol)和10%鈀碳(0.18g),攪拌加熱反應,過濾,濃縮得到粗品,粗品藉由C18反相純化,得到標題化合物4i(400mg,產率44.24%)。 Compound 11 (0.9 g, 3.96 mmol) was dissolved in 5 mL of methanol, then ammonium formate (2.50 g, 39.60 mmol) and 10% palladium on carbon (0.18 g) were added successively, the reaction was stirred and heated, filtered, and concentrated to obtain the crude product, the crude product Reverse phase purification by C18 afforded the title compound 4i (400 mg, 44.24% yield).
MS(ESI)m/z 229.3[M+H]+ MS(ESI)m/z 229.3[M+H] +
第七步 Step 7
二甲基1-(1-(第三-丁氧基羰基)-7-甲基吖庚環-3-基)-3-甲氧基-4-羰基-1,4-二氫吡啶-2,5-二羧酸酯4j Dimethyl 1-(1-(3-butoxycarbonyl)-7-methylazepan-3-yl)-3-methoxy-4-carbonyl-1,4-dihydropyridine-2 ,5-Dicarboxylate 4j
將化合物4i(424.25mg,1.75mmol)溶於5毫升乙醇中,再將化合物12(400mg,1.75mmol)加入反應液中,在80℃反應5小時。反應液藉由減壓濃縮得到粗品,經C18反相純化,得到標題化合物4j(400mg,產率50.46%)。 Compound 4i (424.25 mg, 1.75 mmol) was dissolved in 5 mL of ethanol, and compound 12 (400 mg, 1.75 mmol) was added to the reaction solution, and the reaction was carried out at 80° C. for 5 hours. The reaction solution was concentrated under reduced pressure to obtain the crude product, which was purified by C18 reverse phase to obtain the title compound 4j (400 mg, yield 50.46%).
MS(ESI)m/z 453.5[M+H]+ MS(ESI)m/z 453.5[M+H] +
第八步 Step 8
二甲基1-(1-(第三-丁氧基羰基)-7-甲基吖庚環-3-基)-3-甲氧基-4-羰基-1,4-二氫吡啶-2,5-二羧酸酯4k Dimethyl 1-(1-(3-butoxycarbonyl)-7-methylazepan-3-yl)-3-methoxy-4-carbonyl-1,4-dihydropyridine-2 ,5-dicarboxylate 4k
將化合物4j(400mg,0.88mmol)溶於5毫升二甲苯,然後依次加入醋酸(530.84mg,8.84mmol)和2,4-二氟苄胺(126.53mg,0.88mmol),反應加熱回流5小時。反應液藉由減壓濃縮得到粗品,粗品藉由C18反相純化,得到標題化合物4k(350mg,70.25%)。 Compound 4j (400 mg, 0.88 mmol) was dissolved in 5 mL of xylene, then acetic acid (530.84 mg, 8.84 mmol) and 2,4-difluorobenzylamine (126.53 mg, 0.88 mmol) were added successively, and the reaction was heated to reflux for 5 hours. The reaction solution was concentrated under reduced pressure to obtain the crude product, and the crude product was purified by C18 reverse phase to obtain the title compound 4k (350 mg, 70.25%).
MS(ESI)m/z 564.5[M+H]+ MS(ESI)m/z 564.5[M+H] +
第九步 Step 9
5-((2,4-二氟苯甲基)胺基甲醯)-3-甲氧基-1-(7-甲基吖庚環-3-基)-4-羰基-1,4-二氫吡啶-2-羧酸4l 5-((2,4-Difluorobenzyl)aminocarbamide)-3-methoxy-1-(7-methylazeptan-3-yl)-4-carbonyl-1,4- Dihydropyridine-2-carboxylic acid 4l
將化合物4k(350mg,0.6210mmol)溶於10毫升甲醇中,依次加入氫氧化鋰(52mg,1.2420mmol)和水(22mg,1.2420mmol),在70℃下反應3小時。反應液藉由減壓濃縮得到粗品,再藉由4莫耳/升的鹽酸甲醇溶液(10mL)稀釋,在室溫下繼續反應3小時。反應液藉由濃縮得到化合物4l(370mg)。 Compound 4k (350 mg, 0.6210 mmol) was dissolved in 10 mL of methanol, lithium hydroxide (52 mg, 1.2420 mmol) and water (22 mg, 1.2420 mmol) were sequentially added, and the reaction was carried out at 70° C. for 3 hours. The reaction solution was concentrated under reduced pressure to obtain a crude product, which was then diluted with a 4 mol/L methanolic hydrochloric acid solution (10 mL), and the reaction was continued at room temperature for 3 hours. The reaction solution was concentrated to obtain compound 41 (370 mg).
MS(ESI)m/z 450.4[M+H]+ MS(ESI)m/z 450.4[M+H] +
第十步 Step 10
N-(2,4-二氟苯甲基)-12-甲氧基-3-甲基-1,11-二羰基-1,4,5,6,7,11-六氫-3H-2,7-亞甲基吡啶并[1,2-a][1,4]重氮基壬英-10-甲醯胺4m N -(2,4-Difluorobenzyl)-12-methoxy-3-methyl-1,11-dicarbonyl-1,4,5,6,7,11-hexahydro-3H-2 ,7-Methylenepyrido[1,2-a][1,4]diazononin-10-carboxamide 4m
將化合物4l(320mg,0.71mmol)溶於N,N-二甲基甲醯胺(5mL),依次加入O-(7-氮雜苯并三唑-1-YL)-N,N,N,N-四甲基脲六氟膦鹽(541.45mg,1.42mmol)和N,N-二異丙基乙胺(0.35mL,2.14mmol),在室溫下反應1-2小時。反應液藉由C18反相純化,得到標題化合物4m(200mg,產率65.11%)。 Compound 41 (320 mg, 0.71 mmol) was dissolved in N,N -dimethylformamide (5 mL), followed by adding O- (7-azabenzotriazole-1-YL) -N,N,N, N -tetramethylurea hexafluorophosphine salt (541.45 mg, 1.42 mmol) and N,N -diisopropylethylamine (0.35 mL, 2.14 mmol) were reacted at room temperature for 1-2 hours. The reaction solution was reverse-phase purified by C18 to obtain the title compound 4m (200 mg, yield 65.11%).
MS(ESI)m/z 432.4[M+H]+ MS(ESI)m/z 432.4[M+H] +
第十一步 Step 11
(3R,7R)-N-(2,4-二氟苯甲基)-12-甲氧基-3-甲基-1,11-二羰基-1,4,5,6,7,11-六氫-3H-2,7-亞甲基吡啶并[1,2-a][1,4]重氮基壬英-10-甲醯胺4n ( 3R,7R )-N-(2,4 - difluorobenzyl)-12-methoxy-3-methyl-1,11-dicarbonyl-1,4,5,6,7,11- Hexahydro-3H-2,7-methylenepyrido[1,2-a][1,4]diazononin-10-carboxamide 4n
(3S,7S)-N-(2,4-二氟苯甲基)-12-甲氧基-3-甲基-1,11-二羰基-1,4,5,6,7,11-六氫-3H-2,7-亞甲基吡啶并[1,2-a][1,4]重氮基壬英-10-甲醯胺4o ( 3S,7S )-N-(2,4 - difluorobenzyl)-12-methoxy-3-methyl-1,11-dicarbonyl-1,4,5,6,7,11- Hexahydro-3H-2,7-methylenepyrido[1,2-a][1,4]diazononin-10-carboxamide 4o
將化合物4m(60mg,0.138mmol)用超臨界流體色譜在Chiralpak AD手性管柱上進行拆分,得到標題化合物4n(23.3mg,產率38.83%)和標題化合物4o(19.4mg,32.33%)。 Compound 4m (60 mg, 0.138 mmol) was resolved by supercritical fluid chromatography on a Chiralpak AD chiral column to give title compound 4n (23.3 mg, 38.83% yield) and title compound 4o (19.4 mg, 32.33%) .
色譜條件: Chromatographic conditions:
色譜管柱:Chiralpak AD-3 50×4.6mm I.D.,3um Chromatography column: Chiralpak AD-3 50×4.6mm I.D., 3um
流動相:A:二氧化碳;B:乙醇(0.05%二乙胺) Mobile phase: A: carbon dioxide; B: ethanol (0.05% diethylamine)
第十二步 Step 12
(3S,7S)-N-(2,4-二氟苯甲基)-12-羥基-3-甲基-1,11-二羰基-1,4,5,6,7,11-六氫-3H-2,7-亞甲基吡啶并[1,2-a][1,4]重氮基壬英-10-甲醯胺4 ( 3S,7S )-N-(2,4 - difluorobenzyl)-12-hydroxy-3-methyl-1,11-dicarbonyl-1,4,5,6,7,11-hexahydro -3H-2,7-Methylenepyrido[1,2-a][1,4] diazononin -10-carbamide 4
將化合物4o(46mg,0.11mmol)溶於3毫升乙腈,再加入二溴化鎂(39.25mg,0.21mmol),在50℃下反應1小時。反應液濃縮得到粗品,再藉由C18反相純化,得到標題化合物4(31mg,收率70%) Compound 4o (46 mg, 0.11 mmol) was dissolved in 3 mL of acetonitrile, magnesium dibromide (39.25 mg, 0.21 mmol) was added, and the reaction was carried out at 50° C. for 1 hour. The reaction solution was concentrated to obtain the crude product, which was then purified by C18 reverse phase to obtain the title compound 4 (31 mg, yield 70%)
MS(ESI)m/z 418.5[M+H]+ MS(ESI)m/z 418.5[M+H] +
1H NMR(400MHz,DMSO-d 6 )d 10.39-10.46(m,1 H)8.49(s,1 H)7.36-7.45(m,1 H)7.20-7.28(m,1 H)7.02-7.11(m,1 H)4.77(br s,1 H)4.55(br d,J=5.77Hz,2 H)4.42-4.50(m,1 H)3.64-3.75(m,2 H)1.97-2.07(m,2 H)1.76-1.85(m,1 H)1.57-1.67(m,1 H)1.40-1.51(m,1 H)1.18(d,J=6.53Hz,3 H)0.97-1.01(m,1 H). 1 H NMR (400MHz, DMSO- d 6 )d 10.39-10.46(m,1H) 8.49(s,1H) 7.36-7.45(m,1H) 7.20-7.28(m,1H) 7.02-7.11( m,1H)4.77(br s,1H)4.55(br d,J=5.77Hz,2H)4.42-4.50(m,1H)3.64-3.75(m,2H)1.97-2.07(m, 2H)1.76-1.85(m,1H)1.57-1.67(m,1H)1.40-1.51(m,1H)1.18(d,J=6.53Hz,3H)0.97-1.01(m,1H) ).
生物學評價Biological evaluation
以下結合測試例進一步描述解釋本揭露中,但這些實施例並非意味著限制本揭露中的範圍。 The present disclosure is further described and explained below in conjunction with test examples, but these examples are not intended to limit the scope of the present disclosure.
測試例1 Test Example 1
HIV整合酶突變病毒: HIV integrase mutant virus:
根據文獻報導(ANTIMICROBIAL AGENTS AND CHEMOTHERAPY,Vol.54,No.2,p.934-936),將來源於HIV感染患者的整合酶編碼區域cDNA,連接在HIV整合酶編碼區域缺失的pNL4.3△IN載體上,製備病毒儲備液後,在MT-4細胞感染體系中確定化合物的活性(EC50)。 According to literature reports ( ANTIMICROBIAL AGENTS AND CHEMOTHERAPY , Vol.54, No.2, p.934-936), the cDNA of the integrase coding region derived from HIV-infected patients was ligated into pNL4.3△ which was deleted in the HIV integrase coding region. Compound activity ( EC50 ) was determined in MT-4 cell infection system after virus stock preparation on IN vector.
病毒株 virus strain
註:五個病毒株4736_2,4736_4,8070_1,8070_2和1556_1由Robert W.Shafer博士(斯坦福大學)貢獻。 Note: Five virus strains 4736_2, 4736_4, 8070_1, 8070_2 and 1556_1 were contributed by Dr. Robert W. Shafer (Stanford University).
在研究中將野生型HIV NL4-3病毒作為對照。 Wild-type HIV NL4-3 virus was used as a control in the study.
測定方法: test methods:
將處於對數生長期的MT-4細胞以5000細胞/孔的濃度鋪板至96孔板中,並加入特定濃度的待測試化合物和野生型或整合酶突變病毒,在37℃,5%CO2培養箱中孵育6天後,用MTS(CellTiter®96 Reagent,Promega)染色法檢測化合物對細胞的保護能力,從而確定化合物的抗病毒活性。根據化合物對於野生型病毒和突變病毒的EC50比值確定耐藥倍數。 MT-4 cells in logarithmic growth phase were plated into 96-well plates at a concentration of 5000 cells/well, and the specific concentration of the test compound and wild-type or integrase mutant virus was added, and incubated at 37 °C, 5% CO 2 After 6 days of incubation in the box, the antiviral activity of the compounds was determined by detecting the protective ability of the compounds on cells by MTS (CellTiter® 96 Reagent, Promega) staining. The fold resistance was determined based on the ratio of the EC50 of the compound against the wild-type virus and the mutant virus.
註:DTG多替拉韋;RAL拉替拉韋或雷特格韋;AZT齊多夫定;NA尚未計算. Note: DTG dolutegravir; RAL raltegravir or raltegravir; AZT zidovudine; NA not yet calculated.
本揭露中化合物4在4736_4、1556_1和8070_1病毒株中均展現耐藥性,相比於拉替拉韋,化合物4在4736_4、1556_1和8070_1病毒株中均展現優異的耐藥性,尤其對Y143C突變和G140S、Y143H、Q148H三重突變病毒株。 In the present disclosure, compound 4 exhibited drug resistance in strains 4736_4, 1556_1 and 8070_1, and compared with raltegravir, compound 4 exhibited superior drug resistance in strains 4736_4, 1556_1 and 8070_1, especially against Y143C Mutant and G140S, Y143H, Q148H triple mutant strains.
其它化合物可以參照WO2020197991或WO2020221294的方法製備獲得,並測試相應耐藥性,預期展現耐藥性能。 Other compounds can be prepared with reference to the method of WO2020197991 or WO2020221294, and the corresponding drug resistance is tested, and it is expected to exhibit drug resistance.
Claims (55)
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202011196367 | 2020-10-30 | ||
| CN202011196367.7 | 2020-10-30 | ||
| CN202110018293 | 2021-01-07 | ||
| CN202110018293.6 | 2021-01-07 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| TW202227445A true TW202227445A (en) | 2022-07-16 |
Family
ID=81383583
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| TW110140359A TW202227445A (en) | 2020-10-30 | 2021-10-29 | Use of inhibiting genetically defective hiv virus |
Country Status (2)
| Country | Link |
|---|---|
| TW (1) | TW202227445A (en) |
| WO (1) | WO2022089562A1 (en) |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| TW202544011A (en) | 2019-03-22 | 2025-11-16 | 美商基利科學股份有限公司 | Bridged tricyclic carbamoylpyridone compounds and their pharmaceutical use |
| JP7453399B2 (en) | 2020-02-24 | 2024-03-19 | ギリアード サイエンシーズ, インコーポレイテッド | Tetracyclic compounds and their use for treating HIV infection |
| KR20230079137A (en) | 2020-09-30 | 2023-06-05 | 길리애드 사이언시즈, 인코포레이티드 | Crosslinked Tricyclic Carbamoylpyridone Compounds and Uses Thereof |
| PL4196479T3 (en) | 2021-01-19 | 2024-03-18 | Gilead Sciences, Inc. | Substituted pyridotriazine compounds and uses thereof |
| TWI856796B (en) | 2022-04-06 | 2024-09-21 | 美商基利科學股份有限公司 | Bridged tricyclic carbamoylpyridone compounds and uses thereof |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| UA96568C2 (en) * | 2005-04-28 | 2011-11-25 | Глаксосмиткляйн Ллк | Polycyclic carbamoyl pyridone derivative as hiv-integrase inhibitor |
| PE20151499A1 (en) * | 2012-12-21 | 2015-10-29 | Gilead Sciences Inc | POLYCYCLICAL CARBAMOYLPYRIDONE COMPOUNDS AND THEIR PHARMACEUTICAL USE |
| EP3229804B1 (en) * | 2014-12-09 | 2020-05-06 | Merck Sharp & Dohme Corp. | Spirocyclic heterocycle compounds useful as hiv integrase inhibitors |
| KR20190057158A (en) * | 2015-04-02 | 2019-05-27 | 길리애드 사이언시즈, 인코포레이티드 | Polycyclic-carbamoylpyridone compounds and their pharmaceutical use |
| TW202544011A (en) * | 2019-03-22 | 2025-11-16 | 美商基利科學股份有限公司 | Bridged tricyclic carbamoylpyridone compounds and their pharmaceutical use |
| CN113795491B (en) * | 2019-04-30 | 2024-09-03 | 上海拓界生物医药科技有限公司 | Bridged ring-3, 4-dihydro-pyrido [1,2-a ] pyrazine-1, 8-dione compounds and pharmaceutical uses thereof |
| WO2021093846A1 (en) * | 2019-11-13 | 2021-05-20 | 上海拓界生物医药科技有限公司 | Novel tetracyclic heterocyclic compounds and pharmaceutical use thereof |
-
2021
- 2021-10-29 TW TW110140359A patent/TW202227445A/en unknown
- 2021-10-29 WO PCT/CN2021/127270 patent/WO2022089562A1/en not_active Ceased
Also Published As
| Publication number | Publication date |
|---|---|
| WO2022089562A1 (en) | 2022-05-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| TW202227445A (en) | Use of inhibiting genetically defective hiv virus | |
| US20180127412A1 (en) | Maleate salts of a b-raf kinase inhibitor, crystalline forms, methods of preparation, and uses therefore | |
| WO2019024908A1 (en) | Substituted penta- and hexa-heterocyclic compounds, preparation method therefor, drug combination and use thereof | |
| TWI823420B (en) | Compounds useful as CDK kinase inhibitors and uses thereof | |
| CN115413279A (en) | P2X3 modulators | |
| AU2017405244B9 (en) | Novel pyrrolopyridine derivative, method for producing same, and use thereof | |
| CN105218548A (en) | A kind of novel heterocyclic compounds and preparation method thereof and the purposes as kinase inhibitor | |
| JP6697808B2 (en) | Compounds and methods for preventing or treating sensory hair cell death | |
| CN112313233A (en) | Small molecule inhibitors of JAK family kinases | |
| WO2024046366A1 (en) | Selective parp1 inhibitor | |
| TW202404604A (en) | New solid forms of [(1s)-1-[(2s,4r,5r)-5-(5-amino-2-oxo-thiazolo[4,5-d]pyrimidin-3-yl)-4-hydroxy-tetrahydrofuran-2-yl]propyl] acetate | |
| RU2672249C1 (en) | Polymorph of yonkenafil hydrochloride, method for its preparation and composition on its basis and its application | |
| JP6275644B2 (en) | N- [2-({2-[(2S) -2-cyanopyrrolidin-1-yl] -2-oxoethyl} amino) -2-methylpropyl] -2-methylpyrazolo [1,5-a] pyrimidine-6 -Carboxamide crystals | |
| WO2018072742A1 (en) | Crystalline form of free base of imidazo isoindole derivative and preparation method therefor | |
| CN111393447A (en) | Pyrimidopyrazole compound, and preparation method and application thereof | |
| JP2025533996A (en) | Crystals of polypeptide compounds containing cyano substituents and their method of manufacture | |
| US20220119415A1 (en) | Solid forms of [(1s)-1-[(2s,4r,5r)-5-(5-amino-2-oxo-thiazolo[4,5-d]pyrimidin-3-yl)-4-hydroxy-tetrahydrofuran-2-yl]propyl] acetate | |
| US20220119414A1 (en) | Solid forms of [(1s)-1-[(2s,4r,5r)-5-(5-amino-2-oxo-thiazolo[4,5-d]pyrimidin-3-yl)-4-hydroxy-tetrahydrofuran-2-yl]propyl] acetate | |
| WO2023116763A1 (en) | Pyridazine compound, and pharmaceutical composition and use thereof | |
| WO2022148439A1 (en) | Heterocyclic compound as bcl-2 inhibitor | |
| WO2021219100A1 (en) | Compound containing fused tricyclic structure | |
| WO2021150700A1 (en) | N-substituted-3-tricyclyl piperidine derivatives as anticancer and neuroprotective agents | |
| CN116041322B (en) | N-substituted fluorine-containing piperidine derivative, preparation method and application thereof | |
| KR102683706B1 (en) | Substituted fluorine-containing imidazole salt compounds, methods for their preparation, pharmaceutical compositions thereof and uses thereof | |
| WO2021182491A1 (en) | Crystal of hypoxanthine compound |