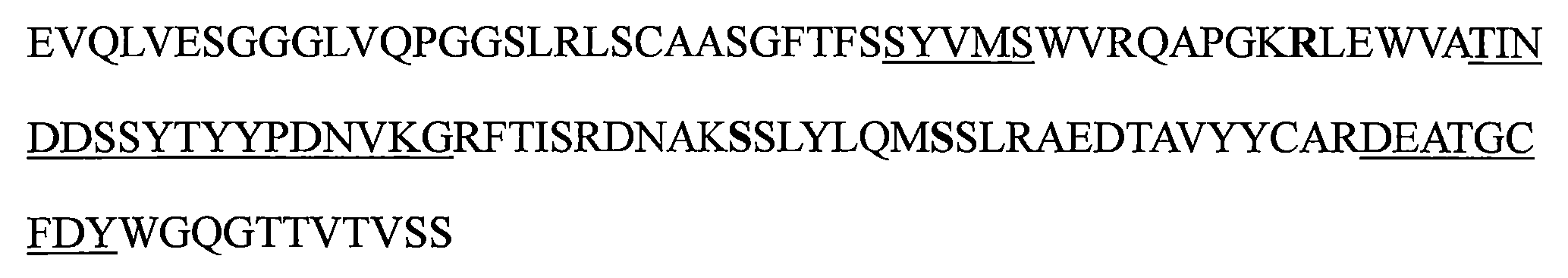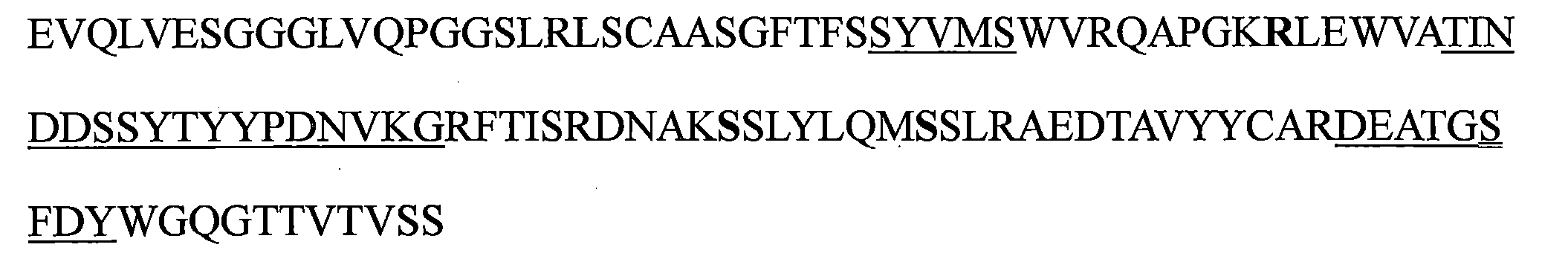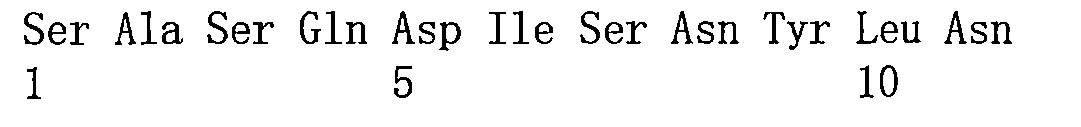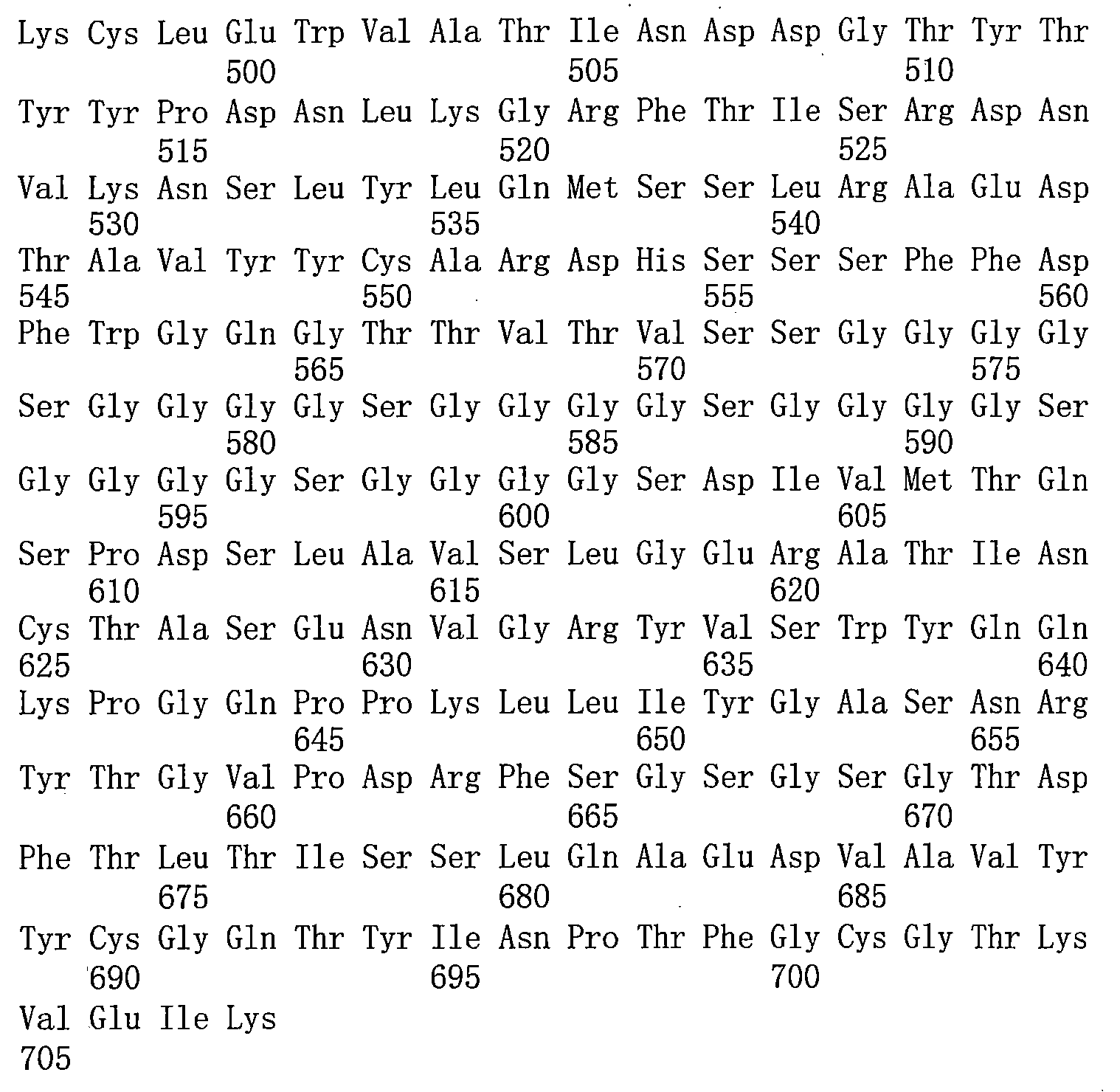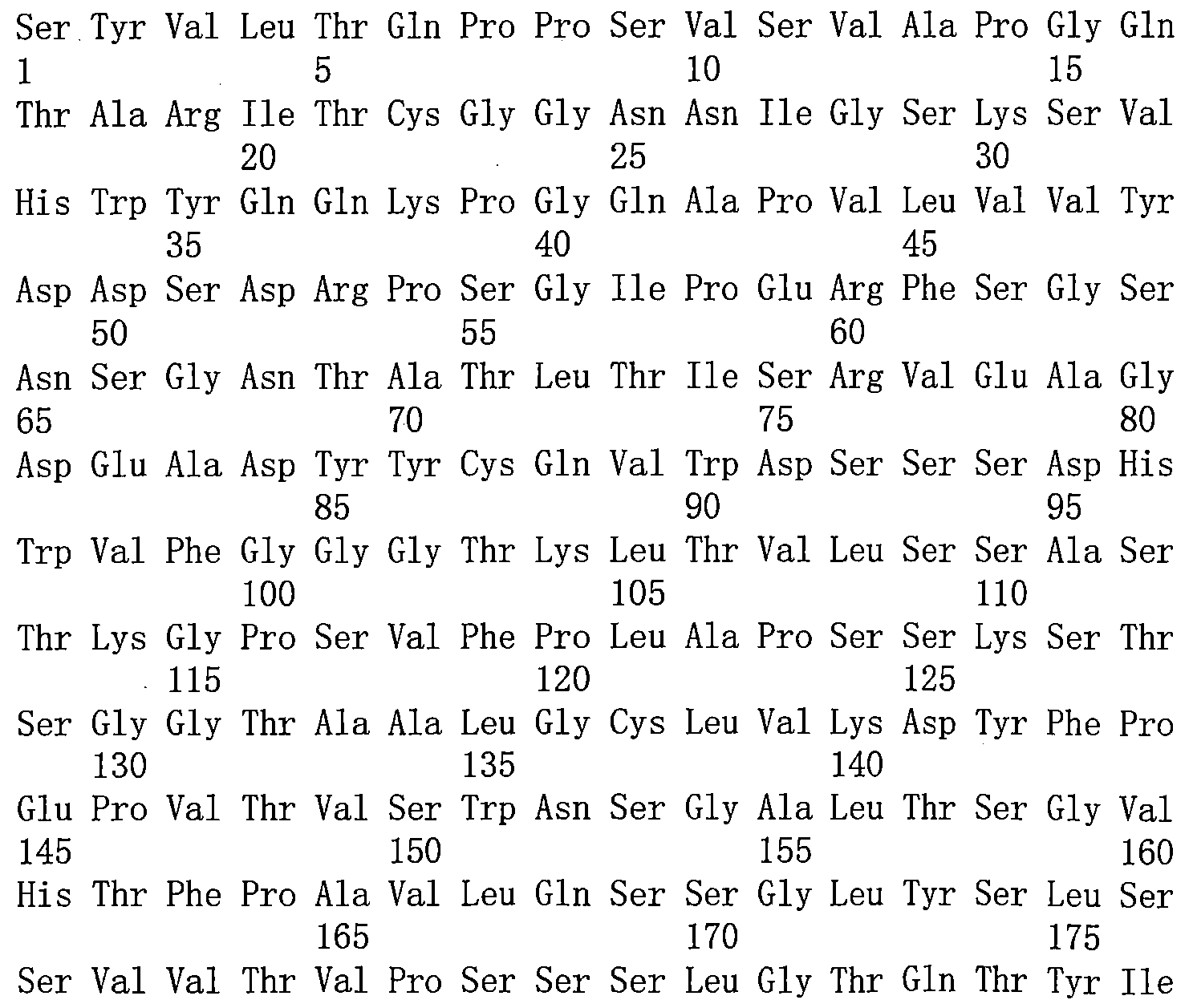TW202221027A - Bispecific antigen binding molecules that specifically bind to vegf and ang-2 - Google Patents
Bispecific antigen binding molecules that specifically bind to vegf and ang-2 Download PDFInfo
- Publication number
- TW202221027A TW202221027A TW110134911A TW110134911A TW202221027A TW 202221027 A TW202221027 A TW 202221027A TW 110134911 A TW110134911 A TW 110134911A TW 110134911 A TW110134911 A TW 110134911A TW 202221027 A TW202221027 A TW 202221027A
- Authority
- TW
- Taiwan
- Prior art keywords
- seq
- variable region
- chain variable
- amino acid
- heavy chain
- Prior art date
Links
- 230000027455 binding Effects 0.000 title claims abstract description 245
- 239000000427 antigen Substances 0.000 title claims abstract description 231
- 102000036639 antigens Human genes 0.000 title claims abstract description 231
- 108091007433 antigens Proteins 0.000 title claims abstract description 231
- 101100372758 Danio rerio vegfaa gene Proteins 0.000 title 1
- 101150030763 Vegfa gene Proteins 0.000 title 1
- 102100034608 Angiopoietin-2 Human genes 0.000 claims abstract description 153
- 108010048036 Angiopoietin-2 Proteins 0.000 claims abstract description 151
- 108010019530 Vascular Endothelial Growth Factors Proteins 0.000 claims abstract description 90
- 102000005789 Vascular Endothelial Growth Factors Human genes 0.000 claims abstract 9
- 125000003275 alpha amino acid group Chemical group 0.000 claims description 256
- 206010028980 Neoplasm Diseases 0.000 claims description 65
- 241000282414 Homo sapiens Species 0.000 claims description 60
- 150000001413 amino acids Chemical class 0.000 claims description 59
- 150000007523 nucleic acids Chemical class 0.000 claims description 30
- 238000000034 method Methods 0.000 claims description 27
- 108020004707 nucleic acids Proteins 0.000 claims description 26
- 102000039446 nucleic acids Human genes 0.000 claims description 26
- 101000924533 Homo sapiens Angiopoietin-2 Proteins 0.000 claims description 22
- 241001529936 Murinae Species 0.000 claims description 20
- 201000011510 cancer Diseases 0.000 claims description 20
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims description 20
- 230000035772 mutation Effects 0.000 claims description 18
- 201000010099 disease Diseases 0.000 claims description 17
- 239000013598 vector Substances 0.000 claims description 15
- 208000005590 Choroidal Neovascularization Diseases 0.000 claims description 13
- 206010060823 Choroidal neovascularisation Diseases 0.000 claims description 13
- 206010064930 age-related macular degeneration Diseases 0.000 claims description 12
- 230000002491 angiogenic effect Effects 0.000 claims description 12
- 208000030533 eye disease Diseases 0.000 claims description 12
- 208000002780 macular degeneration Diseases 0.000 claims description 12
- 206010029113 Neovascularisation Diseases 0.000 claims description 10
- 208000002154 non-small cell lung carcinoma Diseases 0.000 claims description 9
- 239000008194 pharmaceutical composition Substances 0.000 claims description 9
- 208000029729 tumor suppressor gene on chromosome 11 Diseases 0.000 claims description 9
- 208000022211 Arteriovenous Malformations Diseases 0.000 claims description 8
- 101000808011 Homo sapiens Vascular endothelial growth factor A Proteins 0.000 claims description 8
- 239000002253 acid Substances 0.000 claims description 8
- 230000005744 arteriovenous malformation Effects 0.000 claims description 8
- 102000058223 human VEGFA Human genes 0.000 claims description 8
- 230000000711 cancerogenic effect Effects 0.000 claims description 7
- 229910052799 carbon Inorganic materials 0.000 claims description 7
- 206010004593 Bile duct cancer Diseases 0.000 claims description 6
- 206010005003 Bladder cancer Diseases 0.000 claims description 6
- 206010009944 Colon cancer Diseases 0.000 claims description 6
- 206010033128 Ovarian cancer Diseases 0.000 claims description 6
- 206010061535 Ovarian neoplasm Diseases 0.000 claims description 6
- 206010061902 Pancreatic neoplasm Diseases 0.000 claims description 6
- 208000000453 Skin Neoplasms Diseases 0.000 claims description 6
- 208000005718 Stomach Neoplasms Diseases 0.000 claims description 6
- 208000007097 Urinary Bladder Neoplasms Diseases 0.000 claims description 6
- 208000026900 bile duct neoplasm Diseases 0.000 claims description 6
- 206010017758 gastric cancer Diseases 0.000 claims description 6
- 201000007270 liver cancer Diseases 0.000 claims description 6
- 208000014018 liver neoplasm Diseases 0.000 claims description 6
- 208000015486 malignant pancreatic neoplasm Diseases 0.000 claims description 6
- 208000008443 pancreatic carcinoma Diseases 0.000 claims description 6
- 201000000849 skin cancer Diseases 0.000 claims description 6
- 206010041823 squamous cell carcinoma Diseases 0.000 claims description 6
- 201000011549 stomach cancer Diseases 0.000 claims description 6
- 201000005112 urinary bladder cancer Diseases 0.000 claims description 6
- 206010006187 Breast cancer Diseases 0.000 claims description 5
- 208000026310 Breast neoplasm Diseases 0.000 claims description 5
- 208000001333 Colorectal Neoplasms Diseases 0.000 claims description 5
- 201000001342 Fallopian tube cancer Diseases 0.000 claims description 5
- 208000013452 Fallopian tube neoplasm Diseases 0.000 claims description 5
- 208000024447 adrenal gland neoplasm Diseases 0.000 claims description 5
- 125000003277 amino group Chemical group 0.000 claims description 5
- 208000006990 cholangiocarcinoma Diseases 0.000 claims description 5
- 239000003937 drug carrier Substances 0.000 claims description 5
- 201000002528 pancreatic cancer Diseases 0.000 claims description 5
- 230000002207 retinal effect Effects 0.000 claims description 5
- 206010055665 Corneal neovascularisation Diseases 0.000 claims description 4
- 208000010412 Glaucoma Diseases 0.000 claims description 4
- 208000007135 Retinal Neovascularization Diseases 0.000 claims description 4
- 201000000159 corneal neovascularization Diseases 0.000 claims description 4
- 201000003142 neovascular glaucoma Diseases 0.000 claims description 4
- 208000037803 restenosis Diseases 0.000 claims description 4
- 230000002792 vascular Effects 0.000 claims description 4
- 206010012688 Diabetic retinal oedema Diseases 0.000 claims description 3
- 201000011190 diabetic macular edema Diseases 0.000 claims description 3
- 239000000546 pharmaceutical excipient Substances 0.000 claims description 3
- 230000009870 specific binding Effects 0.000 claims description 3
- 206010011017 Corneal graft rejection Diseases 0.000 claims description 2
- 238000012258 culturing Methods 0.000 claims description 2
- 239000003085 diluting agent Substances 0.000 claims description 2
- 208000022873 Ocular disease Diseases 0.000 claims 1
- 208000017572 squamous cell neoplasm Diseases 0.000 claims 1
- 238000002360 preparation method Methods 0.000 abstract description 4
- 210000004027 cell Anatomy 0.000 description 92
- 102000009524 Vascular Endothelial Growth Factor A Human genes 0.000 description 82
- 235000001014 amino acid Nutrition 0.000 description 54
- 101100481410 Mus musculus Tek gene Proteins 0.000 description 35
- 101710117290 Aldo-keto reductase family 1 member C4 Proteins 0.000 description 28
- 102100024952 Protein CBFA2T1 Human genes 0.000 description 28
- 101100481408 Danio rerio tie2 gene Proteins 0.000 description 26
- 238000012360 testing method Methods 0.000 description 26
- 241000699666 Mus <mouse, genus> Species 0.000 description 25
- 108010047041 Complementarity Determining Regions Proteins 0.000 description 24
- 108090000765 processed proteins & peptides Proteins 0.000 description 24
- 102000004196 processed proteins & peptides Human genes 0.000 description 21
- 229960003876 ranibizumab Drugs 0.000 description 21
- 230000000903 blocking effect Effects 0.000 description 19
- 229940116862 faricimab Drugs 0.000 description 17
- 101100440894 Arabidopsis thaliana CP33 gene Proteins 0.000 description 16
- 238000002474 experimental method Methods 0.000 description 16
- 239000012634 fragment Substances 0.000 description 16
- 238000010494 dissociation reaction Methods 0.000 description 15
- 230000005593 dissociations Effects 0.000 description 15
- 108090000623 proteins and genes Proteins 0.000 description 15
- 241000699660 Mus musculus Species 0.000 description 14
- 241000699670 Mus sp. Species 0.000 description 14
- 239000000243 solution Substances 0.000 description 14
- 238000002965 ELISA Methods 0.000 description 13
- 230000014509 gene expression Effects 0.000 description 13
- 239000000203 mixture Substances 0.000 description 13
- 235000018102 proteins Nutrition 0.000 description 13
- 102000004169 proteins and genes Human genes 0.000 description 13
- 241000282693 Cercopithecidae Species 0.000 description 12
- 108091008605 VEGF receptors Proteins 0.000 description 12
- 239000002609 medium Substances 0.000 description 12
- 239000002953 phosphate buffered saline Substances 0.000 description 12
- 229950000449 vanucizumab Drugs 0.000 description 12
- 229920001184 polypeptide Polymers 0.000 description 11
- 108020004705 Codon Proteins 0.000 description 10
- 241000282560 Macaca mulatta Species 0.000 description 10
- 241001465754 Metazoa Species 0.000 description 10
- 102100033177 Vascular endothelial growth factor receptor 2 Human genes 0.000 description 10
- 210000004204 blood vessel Anatomy 0.000 description 10
- 230000000694 effects Effects 0.000 description 10
- 230000004614 tumor growth Effects 0.000 description 10
- 125000000539 amino acid group Chemical group 0.000 description 9
- 229940120638 avastin Drugs 0.000 description 9
- 239000003814 drug Substances 0.000 description 9
- 210000001508 eye Anatomy 0.000 description 9
- 230000026731 phosphorylation Effects 0.000 description 9
- 238000006366 phosphorylation reaction Methods 0.000 description 9
- 230000035755 proliferation Effects 0.000 description 9
- 238000006467 substitution reaction Methods 0.000 description 9
- 210000001519 tissue Anatomy 0.000 description 9
- 238000011282 treatment Methods 0.000 description 9
- NFGXHKASABOEEW-UHFFFAOYSA-N 1-methylethyl 11-methoxy-3,7,11-trimethyl-2,4-dodecadienoate Chemical compound COC(C)(C)CCCC(C)CC=CC(C)=CC(=O)OC(C)C NFGXHKASABOEEW-UHFFFAOYSA-N 0.000 description 8
- 108010048154 Angiopoietin-1 Proteins 0.000 description 8
- 102000009088 Angiopoietin-1 Human genes 0.000 description 8
- 102000018071 Immunoglobulin Fc Fragments Human genes 0.000 description 8
- 108010091135 Immunoglobulin Fc Fragments Proteins 0.000 description 8
- 230000033115 angiogenesis Effects 0.000 description 8
- 108020001507 fusion proteins Proteins 0.000 description 8
- 102000037865 fusion proteins Human genes 0.000 description 8
- 230000002401 inhibitory effect Effects 0.000 description 8
- 229940076783 lucentis Drugs 0.000 description 8
- 208000024891 symptom Diseases 0.000 description 8
- 241000282412 Homo Species 0.000 description 7
- 241000235648 Pichia Species 0.000 description 7
- 238000004113 cell culture Methods 0.000 description 7
- 239000012530 fluid Substances 0.000 description 7
- 210000004602 germ cell Anatomy 0.000 description 7
- 230000012010 growth Effects 0.000 description 7
- 239000000126 substance Substances 0.000 description 7
- 210000004881 tumor cell Anatomy 0.000 description 7
- 239000003981 vehicle Substances 0.000 description 7
- 238000003556 assay Methods 0.000 description 6
- 230000004071 biological effect Effects 0.000 description 6
- 210000002889 endothelial cell Anatomy 0.000 description 6
- 230000006870 function Effects 0.000 description 6
- 230000004927 fusion Effects 0.000 description 6
- 230000005764 inhibitory process Effects 0.000 description 6
- 210000000056 organ Anatomy 0.000 description 6
- 238000012216 screening Methods 0.000 description 6
- 239000006228 supernatant Substances 0.000 description 6
- 108010021625 Immunoglobulin Fragments Proteins 0.000 description 5
- 102000008394 Immunoglobulin Fragments Human genes 0.000 description 5
- 206010027476 Metastases Diseases 0.000 description 5
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 5
- 235000014680 Saccharomyces cerevisiae Nutrition 0.000 description 5
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 5
- 210000001742 aqueous humor Anatomy 0.000 description 5
- 150000001875 compounds Chemical class 0.000 description 5
- UQLDLKMNUJERMK-UHFFFAOYSA-L di(octadecanoyloxy)lead Chemical compound [Pb+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O UQLDLKMNUJERMK-UHFFFAOYSA-L 0.000 description 5
- 230000003834 intracellular effect Effects 0.000 description 5
- 230000009401 metastasis Effects 0.000 description 5
- 238000007920 subcutaneous administration Methods 0.000 description 5
- 229940124597 therapeutic agent Drugs 0.000 description 5
- 238000001890 transfection Methods 0.000 description 5
- 102100033402 Angiopoietin-4 Human genes 0.000 description 4
- 238000011729 BALB/c nude mouse Methods 0.000 description 4
- 108020004414 DNA Proteins 0.000 description 4
- 241000282567 Macaca fascicularis Species 0.000 description 4
- 108010069801 angiopoietin 4 Proteins 0.000 description 4
- 230000000890 antigenic effect Effects 0.000 description 4
- 239000000872 buffer Substances 0.000 description 4
- 238000011161 development Methods 0.000 description 4
- 230000018109 developmental process Effects 0.000 description 4
- 239000012139 lysis buffer Substances 0.000 description 4
- 230000000649 photocoagulation Effects 0.000 description 4
- 230000001737 promoting effect Effects 0.000 description 4
- 238000000159 protein binding assay Methods 0.000 description 4
- RXWNCPJZOCPEPQ-NVWDDTSBSA-N puromycin Chemical compound C1=CC(OC)=CC=C1C[C@H](N)C(=O)N[C@H]1[C@@H](O)[C@H](N2C3=NC=NC(=C3N=C2)N(C)C)O[C@@H]1CO RXWNCPJZOCPEPQ-NVWDDTSBSA-N 0.000 description 4
- 229940081969 saccharomyces cerevisiae Drugs 0.000 description 4
- 241000894007 species Species 0.000 description 4
- 210000003556 vascular endothelial cell Anatomy 0.000 description 4
- 239000012224 working solution Substances 0.000 description 4
- FWMNVWWHGCHHJJ-SKKKGAJSSA-N 4-amino-1-[(2r)-6-amino-2-[[(2r)-2-[[(2r)-2-[[(2r)-2-amino-3-phenylpropanoyl]amino]-3-phenylpropanoyl]amino]-4-methylpentanoyl]amino]hexanoyl]piperidine-4-carboxylic acid Chemical compound C([C@H](C(=O)N[C@H](CC(C)C)C(=O)N[C@H](CCCCN)C(=O)N1CCC(N)(CC1)C(O)=O)NC(=O)[C@H](N)CC=1C=CC=CC=1)C1=CC=CC=C1 FWMNVWWHGCHHJJ-SKKKGAJSSA-N 0.000 description 3
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- 102100035360 Cerebellar degeneration-related antigen 1 Human genes 0.000 description 3
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 3
- 108060003951 Immunoglobulin Proteins 0.000 description 3
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 3
- FFEARJCKVFRZRR-BYPYZUCNSA-N L-methionine Chemical compound CSCC[C@H](N)C(O)=O FFEARJCKVFRZRR-BYPYZUCNSA-N 0.000 description 3
- 241000124008 Mammalia Species 0.000 description 3
- 230000001580 bacterial effect Effects 0.000 description 3
- 239000013060 biological fluid Substances 0.000 description 3
- 230000015572 biosynthetic process Effects 0.000 description 3
- 210000004899 c-terminal region Anatomy 0.000 description 3
- 230000004663 cell proliferation Effects 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 239000011248 coating agent Substances 0.000 description 3
- 238000000576 coating method Methods 0.000 description 3
- 238000001514 detection method Methods 0.000 description 3
- 238000010790 dilution Methods 0.000 description 3
- 239000012895 dilution Substances 0.000 description 3
- 208000035475 disorder Diseases 0.000 description 3
- 230000007717 exclusion Effects 0.000 description 3
- 230000002538 fungal effect Effects 0.000 description 3
- 230000002068 genetic effect Effects 0.000 description 3
- 206010073071 hepatocellular carcinoma Diseases 0.000 description 3
- 102000018358 immunoglobulin Human genes 0.000 description 3
- 238000000338 in vitro Methods 0.000 description 3
- 239000003446 ligand Substances 0.000 description 3
- 229930182817 methionine Natural products 0.000 description 3
- 239000013642 negative control Substances 0.000 description 3
- 238000012014 optical coherence tomography Methods 0.000 description 3
- 239000013612 plasmid Substances 0.000 description 3
- 108091033319 polynucleotide Proteins 0.000 description 3
- 102000040430 polynucleotide Human genes 0.000 description 3
- 239000002157 polynucleotide Substances 0.000 description 3
- 102000005962 receptors Human genes 0.000 description 3
- 108020003175 receptors Proteins 0.000 description 3
- 238000009738 saturating Methods 0.000 description 3
- 238000012163 sequencing technique Methods 0.000 description 3
- 239000011780 sodium chloride Substances 0.000 description 3
- 239000002904 solvent Substances 0.000 description 3
- 230000005747 tumor angiogenesis Effects 0.000 description 3
- 238000005406 washing Methods 0.000 description 3
- YBJHBAHKTGYVGT-ZKWXMUAHSA-N (+)-Biotin Chemical compound N1C(=O)N[C@@H]2[C@H](CCCCC(=O)O)SC[C@@H]21 YBJHBAHKTGYVGT-ZKWXMUAHSA-N 0.000 description 2
- MTCFGRXMJLQNBG-REOHCLBHSA-N (2S)-2-Amino-3-hydroxypropansäure Chemical compound OC[C@H](N)C(O)=O MTCFGRXMJLQNBG-REOHCLBHSA-N 0.000 description 2
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 2
- UHPMCKVQTMMPCG-UHFFFAOYSA-N 5,8-dihydroxy-2-methoxy-6-methyl-7-(2-oxopropyl)naphthalene-1,4-dione Chemical compound CC1=C(CC(C)=O)C(O)=C2C(=O)C(OC)=CC(=O)C2=C1O UHPMCKVQTMMPCG-UHFFFAOYSA-N 0.000 description 2
- 102000009840 Angiopoietins Human genes 0.000 description 2
- 108010009906 Angiopoietins Proteins 0.000 description 2
- 241000283690 Bos taurus Species 0.000 description 2
- 241000288950 Callithrix jacchus Species 0.000 description 2
- 241000222122 Candida albicans Species 0.000 description 2
- 241001674013 Chrysosporium lucknowense Species 0.000 description 2
- 108091026890 Coding region Proteins 0.000 description 2
- 241000699800 Cricetinae Species 0.000 description 2
- 238000012286 ELISA Assay Methods 0.000 description 2
- 241000196324 Embryophyta Species 0.000 description 2
- 241000223218 Fusarium Species 0.000 description 2
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 2
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 2
- 102000003886 Glycoproteins Human genes 0.000 description 2
- 108090000288 Glycoproteins Proteins 0.000 description 2
- 101001037153 Homo sapiens Immunoglobulin heavy variable 3-7 Proteins 0.000 description 2
- 101000604674 Homo sapiens Immunoglobulin kappa variable 4-1 Proteins 0.000 description 2
- 102100040231 Immunoglobulin heavy variable 3-7 Human genes 0.000 description 2
- 102100038198 Immunoglobulin kappa variable 4-1 Human genes 0.000 description 2
- 208000008839 Kidney Neoplasms Diseases 0.000 description 2
- 241000235649 Kluyveromyces Species 0.000 description 2
- QNAYBMKLOCPYGJ-REOHCLBHSA-N L-alanine Chemical compound C[C@H](N)C(O)=O QNAYBMKLOCPYGJ-REOHCLBHSA-N 0.000 description 2
- LRQKBLKVPFOOQJ-YFKPBYRVSA-N L-norleucine Chemical compound CCCC[C@H]([NH3+])C([O-])=O LRQKBLKVPFOOQJ-YFKPBYRVSA-N 0.000 description 2
- 206010025421 Macule Diseases 0.000 description 2
- 101000732672 Mus musculus Angiopoietin-2 Proteins 0.000 description 2
- 101000808007 Mus musculus Vascular endothelial growth factor A Proteins 0.000 description 2
- 229910020700 Na3VO4 Inorganic materials 0.000 description 2
- 241000221961 Neurospora crassa Species 0.000 description 2
- 108091028043 Nucleic acid sequence Proteins 0.000 description 2
- 241000320412 Ogataea angusta Species 0.000 description 2
- 241001489174 Ogataea minuta Species 0.000 description 2
- 241000282577 Pan troglodytes Species 0.000 description 2
- 206010035226 Plasma cell myeloma Diseases 0.000 description 2
- 206010060862 Prostate cancer Diseases 0.000 description 2
- 208000000236 Prostatic Neoplasms Diseases 0.000 description 2
- 108090000412 Protein-Tyrosine Kinases Proteins 0.000 description 2
- 102000004022 Protein-Tyrosine Kinases Human genes 0.000 description 2
- 108010008281 Recombinant Fusion Proteins Proteins 0.000 description 2
- 102000007056 Recombinant Fusion Proteins Human genes 0.000 description 2
- 206010038389 Renal cancer Diseases 0.000 description 2
- 206010039491 Sarcoma Diseases 0.000 description 2
- 206010041067 Small cell lung cancer Diseases 0.000 description 2
- 102000012753 TIE-2 Receptor Human genes 0.000 description 2
- 108010090091 TIE-2 Receptor Proteins 0.000 description 2
- 206010052779 Transplant rejections Diseases 0.000 description 2
- 241000499912 Trichoderma reesei Species 0.000 description 2
- 108010073929 Vascular Endothelial Growth Factor A Proteins 0.000 description 2
- 102000009484 Vascular Endothelial Growth Factor Receptors Human genes 0.000 description 2
- 241000700605 Viruses Species 0.000 description 2
- 238000001042 affinity chromatography Methods 0.000 description 2
- 235000004279 alanine Nutrition 0.000 description 2
- 150000001412 amines Chemical class 0.000 description 2
- 230000003698 anagen phase Effects 0.000 description 2
- 238000004458 analytical method Methods 0.000 description 2
- 238000002583 angiography Methods 0.000 description 2
- 238000010171 animal model Methods 0.000 description 2
- 230000009830 antibody antigen interaction Effects 0.000 description 2
- 102000025171 antigen binding proteins Human genes 0.000 description 2
- 108091000831 antigen binding proteins Proteins 0.000 description 2
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 2
- 238000012925 biological evaluation Methods 0.000 description 2
- 230000037396 body weight Effects 0.000 description 2
- 229940095731 candida albicans Drugs 0.000 description 2
- 239000013592 cell lysate Substances 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- 210000004978 chinese hamster ovary cell Anatomy 0.000 description 2
- 230000009137 competitive binding Effects 0.000 description 2
- 230000000295 complement effect Effects 0.000 description 2
- 230000003247 decreasing effect Effects 0.000 description 2
- 238000000375 direct analysis in real time Methods 0.000 description 2
- 238000012063 dual-affinity re-targeting Methods 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- 239000013604 expression vector Substances 0.000 description 2
- GNBHRKFJIUUOQI-UHFFFAOYSA-N fluorescein Chemical compound O1C(=O)C2=CC=CC=C2C21C1=CC=C(O)C=C1OC1=CC(O)=CC=C21 GNBHRKFJIUUOQI-UHFFFAOYSA-N 0.000 description 2
- 230000005714 functional activity Effects 0.000 description 2
- 231100000844 hepatocellular carcinoma Toxicity 0.000 description 2
- 230000028993 immune response Effects 0.000 description 2
- 238000001727 in vivo Methods 0.000 description 2
- 238000011534 incubation Methods 0.000 description 2
- 238000002347 injection Methods 0.000 description 2
- 239000007924 injection Substances 0.000 description 2
- 238000007918 intramuscular administration Methods 0.000 description 2
- 238000001990 intravenous administration Methods 0.000 description 2
- 210000003734 kidney Anatomy 0.000 description 2
- 201000010982 kidney cancer Diseases 0.000 description 2
- 210000004962 mammalian cell Anatomy 0.000 description 2
- 241001515942 marmosets Species 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 108010082117 matrigel Proteins 0.000 description 2
- 230000001394 metastastic effect Effects 0.000 description 2
- 206010061289 metastatic neoplasm Diseases 0.000 description 2
- 125000003729 nucleotide group Chemical group 0.000 description 2
- 230000002093 peripheral effect Effects 0.000 description 2
- 230000000144 pharmacologic effect Effects 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 238000000746 purification Methods 0.000 description 2
- 229950010131 puromycin Drugs 0.000 description 2
- 229960002633 ramucirumab Drugs 0.000 description 2
- 230000010076 replication Effects 0.000 description 2
- 238000010839 reverse transcription Methods 0.000 description 2
- 102220311238 rs768968410 Human genes 0.000 description 2
- 239000000523 sample Substances 0.000 description 2
- 210000002966 serum Anatomy 0.000 description 2
- 208000000587 small cell lung carcinoma Diseases 0.000 description 2
- 238000002198 surface plasmon resonance spectroscopy Methods 0.000 description 2
- 230000001225 therapeutic effect Effects 0.000 description 2
- IHIXIJGXTJIKRB-UHFFFAOYSA-N trisodium vanadate Chemical compound [Na+].[Na+].[Na+].[O-][V]([O-])([O-])=O IHIXIJGXTJIKRB-UHFFFAOYSA-N 0.000 description 2
- 239000013603 viral vector Substances 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 2
- 239000000080 wetting agent Substances 0.000 description 2
- OZFAFGSSMRRTDW-UHFFFAOYSA-N (2,4-dichlorophenyl) benzenesulfonate Chemical compound ClC1=CC(Cl)=CC=C1OS(=O)(=O)C1=CC=CC=C1 OZFAFGSSMRRTDW-UHFFFAOYSA-N 0.000 description 1
- UKAUYVFTDYCKQA-UHFFFAOYSA-N -2-Amino-4-hydroxybutanoic acid Natural products OC(=O)C(N)CCO UKAUYVFTDYCKQA-UHFFFAOYSA-N 0.000 description 1
- NVKAWKQGWWIWPM-ABEVXSGRSA-N 17-β-hydroxy-5-α-Androstan-3-one Chemical compound C1C(=O)CC[C@]2(C)[C@H]3CC[C@](C)([C@H](CC4)O)[C@@H]4[C@@H]3CC[C@H]21 NVKAWKQGWWIWPM-ABEVXSGRSA-N 0.000 description 1
- QKNYBSVHEMOAJP-UHFFFAOYSA-N 2-amino-2-(hydroxymethyl)propane-1,3-diol;hydron;chloride Chemical compound Cl.OCC(N)(CO)CO QKNYBSVHEMOAJP-UHFFFAOYSA-N 0.000 description 1
- IVLXQGJVBGMLRR-UHFFFAOYSA-N 2-aminoacetic acid;hydron;chloride Chemical compound Cl.NCC(O)=O IVLXQGJVBGMLRR-UHFFFAOYSA-N 0.000 description 1
- 206010069754 Acquired gene mutation Diseases 0.000 description 1
- 241000702423 Adeno-associated virus - 2 Species 0.000 description 1
- 208000007860 Anus Neoplasms Diseases 0.000 description 1
- 108010039627 Aprotinin Proteins 0.000 description 1
- 206010003445 Ascites Diseases 0.000 description 1
- 241000228212 Aspergillus Species 0.000 description 1
- 241000351920 Aspergillus nidulans Species 0.000 description 1
- 241000228245 Aspergillus niger Species 0.000 description 1
- 240000006439 Aspergillus oryzae Species 0.000 description 1
- 235000002247 Aspergillus oryzae Nutrition 0.000 description 1
- 241000151861 Barnettozyma salicaria Species 0.000 description 1
- 206010055113 Breast cancer metastatic Diseases 0.000 description 1
- 241000282465 Canis Species 0.000 description 1
- 241000282472 Canis lupus familiaris Species 0.000 description 1
- 241000283707 Capra Species 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 201000009030 Carcinoma Diseases 0.000 description 1
- 208000017897 Carcinoma of esophagus Diseases 0.000 description 1
- 206010008342 Cervix carcinoma Diseases 0.000 description 1
- 241000699802 Cricetulus griseus Species 0.000 description 1
- 241000938605 Crocodylia Species 0.000 description 1
- 108090000695 Cytokines Proteins 0.000 description 1
- 102000004127 Cytokines Human genes 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- 102000053602 DNA Human genes 0.000 description 1
- 241000702421 Dependoparvovirus Species 0.000 description 1
- 206010061818 Disease progression Diseases 0.000 description 1
- 239000012591 Dulbecco’s Phosphate Buffered Saline Substances 0.000 description 1
- 206010014733 Endometrial cancer Diseases 0.000 description 1
- 206010014759 Endometrial neoplasm Diseases 0.000 description 1
- 241000283073 Equus caballus Species 0.000 description 1
- 208000000461 Esophageal Neoplasms Diseases 0.000 description 1
- 206010015548 Euthanasia Diseases 0.000 description 1
- 108091006020 Fc-tagged proteins Proteins 0.000 description 1
- 241001149959 Fusarium sp. Species 0.000 description 1
- 241000567178 Fusarium venenatum Species 0.000 description 1
- 241000287828 Gallus gallus Species 0.000 description 1
- 102400000921 Gastrin Human genes 0.000 description 1
- 108010052343 Gastrins Proteins 0.000 description 1
- 206010017993 Gastrointestinal neoplasms Diseases 0.000 description 1
- 239000004471 Glycine Substances 0.000 description 1
- 241000238631 Hexapoda Species 0.000 description 1
- 101000690301 Homo sapiens Aldo-keto reductase family 1 member C4 Proteins 0.000 description 1
- 101000878605 Homo sapiens Low affinity immunoglobulin epsilon Fc receptor Proteins 0.000 description 1
- 101001116548 Homo sapiens Protein CBFA2T1 Proteins 0.000 description 1
- PMMYEEVYMWASQN-DMTCNVIQSA-N Hydroxyproline Chemical compound O[C@H]1CN[C@H](C(O)=O)C1 PMMYEEVYMWASQN-DMTCNVIQSA-N 0.000 description 1
- 102000006496 Immunoglobulin Heavy Chains Human genes 0.000 description 1
- 108010019476 Immunoglobulin Heavy Chains Proteins 0.000 description 1
- UGQMRVRMYYASKQ-KQYNXXCUSA-N Inosine Chemical group O[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1N1C2=NC=NC(O)=C2N=C1 UGQMRVRMYYASKQ-KQYNXXCUSA-N 0.000 description 1
- 102100034343 Integrase Human genes 0.000 description 1
- 108091092195 Intron Proteins 0.000 description 1
- PIWKPBJCKXDKJR-UHFFFAOYSA-N Isoflurane Chemical compound FC(F)OC(Cl)C(F)(F)F PIWKPBJCKXDKJR-UHFFFAOYSA-N 0.000 description 1
- 241001138401 Kluyveromyces lactis Species 0.000 description 1
- 241000235058 Komagataella pastoris Species 0.000 description 1
- 229930195714 L-glutamate Natural products 0.000 description 1
- UKAUYVFTDYCKQA-VKHMYHEASA-N L-homoserine Chemical compound OC(=O)[C@@H](N)CCO UKAUYVFTDYCKQA-VKHMYHEASA-N 0.000 description 1
- AYFVYJQAPQTCCC-GBXIJSLDSA-N L-threonine Chemical compound C[C@@H](O)[C@H](N)C(O)=O AYFVYJQAPQTCCC-GBXIJSLDSA-N 0.000 description 1
- QIVBCDIJIAJPQS-VIFPVBQESA-N L-tryptophane Chemical compound C1=CC=C2C(C[C@H](N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-VIFPVBQESA-N 0.000 description 1
- 102100038007 Low affinity immunoglobulin epsilon Fc receptor Human genes 0.000 description 1
- 108060001084 Luciferase Proteins 0.000 description 1
- 208000019693 Lung disease Diseases 0.000 description 1
- 206010058467 Lung neoplasm malignant Diseases 0.000 description 1
- 102220465493 Lymphocyte activation gene 3 protein_D52A_mutation Human genes 0.000 description 1
- 206010025323 Lymphomas Diseases 0.000 description 1
- 239000004472 Lysine Substances 0.000 description 1
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 1
- 229930195725 Mannitol Natural products 0.000 description 1
- 208000000172 Medulloblastoma Diseases 0.000 description 1
- 206010027406 Mesothelioma Diseases 0.000 description 1
- 208000034578 Multiple myelomas Diseases 0.000 description 1
- 241001452677 Ogataea methanolica Species 0.000 description 1
- 241000489470 Ogataea trehalophila Species 0.000 description 1
- 241000826199 Ogataea wickerhamii Species 0.000 description 1
- 241000282579 Pan Species 0.000 description 1
- 241001494479 Pecora Species 0.000 description 1
- 208000002471 Penile Neoplasms Diseases 0.000 description 1
- 108091093037 Peptide nucleic acid Proteins 0.000 description 1
- 241000529953 Phaffomyces thermotolerans Species 0.000 description 1
- 241000195887 Physcomitrella patens Species 0.000 description 1
- 241000235062 Pichia membranifaciens Species 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 229920001213 Polysorbate 20 Polymers 0.000 description 1
- 206010036790 Productive cough Diseases 0.000 description 1
- 108010092799 RNA-directed DNA polymerase Proteins 0.000 description 1
- 239000012980 RPMI-1640 medium Substances 0.000 description 1
- 241000700159 Rattus Species 0.000 description 1
- 108090000873 Receptor Protein-Tyrosine Kinases Proteins 0.000 description 1
- 102000004278 Receptor Protein-Tyrosine Kinases Human genes 0.000 description 1
- 208000015634 Rectal Neoplasms Diseases 0.000 description 1
- 201000000582 Retinoblastoma Diseases 0.000 description 1
- 108091028664 Ribonucleotide Proteins 0.000 description 1
- YDBYJHTYSHBBAU-YFKPBYRVSA-N S-methyl-L-methioninate Chemical compound C[S+](C)CC[C@H](N)C([O-])=O YDBYJHTYSHBBAU-YFKPBYRVSA-N 0.000 description 1
- 208000004337 Salivary Gland Neoplasms Diseases 0.000 description 1
- 206010061934 Salivary gland cancer Diseases 0.000 description 1
- MTCFGRXMJLQNBG-UHFFFAOYSA-N Serine Natural products OCC(N)C(O)=O MTCFGRXMJLQNBG-UHFFFAOYSA-N 0.000 description 1
- 241000256251 Spodoptera frugiperda Species 0.000 description 1
- 108010090804 Streptavidin Proteins 0.000 description 1
- LSNNMFCWUKXFEE-UHFFFAOYSA-N Sulfurous acid Chemical compound OS(O)=O LSNNMFCWUKXFEE-UHFFFAOYSA-N 0.000 description 1
- 102220548254 Tafazzin_E96Q_mutation Human genes 0.000 description 1
- 208000024313 Testicular Neoplasms Diseases 0.000 description 1
- RYYWUUFWQRZTIU-UHFFFAOYSA-N Thiophosphoric acid Chemical class OP(O)(S)=O RYYWUUFWQRZTIU-UHFFFAOYSA-N 0.000 description 1
- AYFVYJQAPQTCCC-UHFFFAOYSA-N Threonine Natural products CC(O)C(N)C(O)=O AYFVYJQAPQTCCC-UHFFFAOYSA-N 0.000 description 1
- 239000004473 Threonine Substances 0.000 description 1
- 208000024770 Thyroid neoplasm Diseases 0.000 description 1
- 241000255993 Trichoplusia ni Species 0.000 description 1
- 102000004142 Trypsin Human genes 0.000 description 1
- 108090000631 Trypsin Proteins 0.000 description 1
- QIVBCDIJIAJPQS-UHFFFAOYSA-N Tryptophan Natural products C1=CC=C2C(CC(N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-UHFFFAOYSA-N 0.000 description 1
- 208000006105 Uterine Cervical Neoplasms Diseases 0.000 description 1
- 208000002495 Uterine Neoplasms Diseases 0.000 description 1
- 241000251539 Vertebrata <Metazoa> Species 0.000 description 1
- 208000014070 Vestibular schwannoma Diseases 0.000 description 1
- 206010047741 Vulval cancer Diseases 0.000 description 1
- 208000004354 Vulvar Neoplasms Diseases 0.000 description 1
- 241000370136 Wickerhamomyces pijperi Species 0.000 description 1
- 241000235015 Yarrowia lipolytica Species 0.000 description 1
- 210000000683 abdominal cavity Anatomy 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- 238000002835 absorbance Methods 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 239000008351 acetate buffer Substances 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 208000004064 acoustic neuroma Diseases 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 208000009956 adenocarcinoma Diseases 0.000 description 1
- 230000001464 adherent effect Effects 0.000 description 1
- 239000002671 adjuvant Substances 0.000 description 1
- 239000000556 agonist Substances 0.000 description 1
- 238000012867 alanine scanning Methods 0.000 description 1
- 229940037003 alum Drugs 0.000 description 1
- 239000005557 antagonist Substances 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- 230000000844 anti-bacterial effect Effects 0.000 description 1
- 230000000259 anti-tumor effect Effects 0.000 description 1
- 239000003429 antifungal agent Substances 0.000 description 1
- 229940121375 antifungal agent Drugs 0.000 description 1
- 229960004405 aprotinin Drugs 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 239000003124 biologic agent Substances 0.000 description 1
- 230000033228 biological regulation Effects 0.000 description 1
- 229960000074 biopharmaceutical Drugs 0.000 description 1
- 238000001574 biopsy Methods 0.000 description 1
- 229960002685 biotin Drugs 0.000 description 1
- 235000020958 biotin Nutrition 0.000 description 1
- 239000011616 biotin Substances 0.000 description 1
- 201000000053 blastoma Diseases 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 210000005252 bulbus oculi Anatomy 0.000 description 1
- 238000004364 calculation method Methods 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- UHBYWPGGCSDKFX-UHFFFAOYSA-N carboxyglutamic acid Chemical compound OC(=O)C(N)CC(C(O)=O)C(O)=O UHBYWPGGCSDKFX-UHFFFAOYSA-N 0.000 description 1
- 208000002458 carcinoid tumor Diseases 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 239000005018 casein Substances 0.000 description 1
- BECPQYXYKAMYBN-UHFFFAOYSA-N casein, tech. Chemical compound NCCCCC(C(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(CC(C)C)N=C(O)C(CCC(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(C(C)O)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(COP(O)(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(N)CC1=CC=CC=C1 BECPQYXYKAMYBN-UHFFFAOYSA-N 0.000 description 1
- 235000021240 caseins Nutrition 0.000 description 1
- 230000010261 cell growth Effects 0.000 description 1
- 230000012292 cell migration Effects 0.000 description 1
- 238000012054 celltiter-glo Methods 0.000 description 1
- 238000005119 centrifugation Methods 0.000 description 1
- 201000010881 cervical cancer Diseases 0.000 description 1
- 210000003756 cervix mucus Anatomy 0.000 description 1
- AOXOCDRNSPFDPE-UKEONUMOSA-N chembl413654 Chemical compound C([C@H](C(=O)NCC(=O)N[C@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@H](CCSC)C(=O)N[C@H](CC(O)=O)C(=O)N[C@H](CC=1C=CC=CC=1)C(N)=O)NC(=O)[C@@H](C)NC(=O)[C@@H](CCC(O)=O)NC(=O)[C@@H](CCC(O)=O)NC(=O)[C@@H](CCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H]1N(CCC1)C(=O)CNC(=O)[C@@H](N)CCC(O)=O)C1=CC=C(O)C=C1 AOXOCDRNSPFDPE-UKEONUMOSA-N 0.000 description 1
- 238000007385 chemical modification Methods 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 235000013330 chicken meat Nutrition 0.000 description 1
- 239000003593 chromogenic compound Substances 0.000 description 1
- 208000029742 colonic neoplasm Diseases 0.000 description 1
- 238000012875 competitive assay Methods 0.000 description 1
- 230000004154 complement system Effects 0.000 description 1
- 239000002299 complementary DNA Substances 0.000 description 1
- 239000003636 conditioned culture medium Substances 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- 239000012228 culture supernatant Substances 0.000 description 1
- 235000018417 cysteine Nutrition 0.000 description 1
- XUJNEKJLAYXESH-UHFFFAOYSA-N cysteine Natural products SCC(N)C(O)=O XUJNEKJLAYXESH-UHFFFAOYSA-N 0.000 description 1
- 230000009089 cytolysis Effects 0.000 description 1
- 230000006735 deficit Effects 0.000 description 1
- 239000005547 deoxyribonucleotide Substances 0.000 description 1
- 125000002637 deoxyribonucleotide group Chemical group 0.000 description 1
- 239000008121 dextrose Substances 0.000 description 1
- 206010012601 diabetes mellitus Diseases 0.000 description 1
- 239000000032 diagnostic agent Substances 0.000 description 1
- 229940039227 diagnostic agent Drugs 0.000 description 1
- LOKCTEFSRHRXRJ-UHFFFAOYSA-I dipotassium trisodium dihydrogen phosphate hydrogen phosphate dichloride Chemical compound P(=O)(O)(O)[O-].[K+].P(=O)(O)([O-])[O-].[Na+].[Na+].[Cl-].[K+].[Cl-].[Na+] LOKCTEFSRHRXRJ-UHFFFAOYSA-I 0.000 description 1
- 230000005750 disease progression Effects 0.000 description 1
- 239000002612 dispersion medium Substances 0.000 description 1
- 150000002019 disulfides Chemical class 0.000 description 1
- PMMYEEVYMWASQN-UHFFFAOYSA-N dl-hydroxyproline Natural products OC1C[NH2+]C(C([O-])=O)C1 PMMYEEVYMWASQN-UHFFFAOYSA-N 0.000 description 1
- 239000012154 double-distilled water Substances 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 239000012636 effector Substances 0.000 description 1
- 238000001962 electrophoresis Methods 0.000 description 1
- 201000008184 embryoma Diseases 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 230000002357 endometrial effect Effects 0.000 description 1
- 210000003989 endothelium vascular Anatomy 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 230000005284 excitation Effects 0.000 description 1
- 239000013613 expression plasmid Substances 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 235000013861 fat-free Nutrition 0.000 description 1
- 210000003608 fece Anatomy 0.000 description 1
- 239000012526 feed medium Substances 0.000 description 1
- 238000000684 flow cytometry Methods 0.000 description 1
- 238000013534 fluorescein angiography Methods 0.000 description 1
- 238000001943 fluorescence-activated cell sorting Methods 0.000 description 1
- 238000002594 fluoroscopy Methods 0.000 description 1
- 230000002496 gastric effect Effects 0.000 description 1
- 238000005227 gel permeation chromatography Methods 0.000 description 1
- 208000005017 glioblastoma Diseases 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 239000001963 growth medium Substances 0.000 description 1
- 201000010536 head and neck cancer Diseases 0.000 description 1
- 208000014829 head and neck neoplasm Diseases 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 230000003862 health status Effects 0.000 description 1
- 238000007490 hematoxylin and eosin (H&E) staining Methods 0.000 description 1
- 230000002440 hepatic effect Effects 0.000 description 1
- 238000010562 histological examination Methods 0.000 description 1
- 102000054751 human RUNX1T1 Human genes 0.000 description 1
- 210000005260 human cell Anatomy 0.000 description 1
- 235000003642 hunger Nutrition 0.000 description 1
- 238000007131 hydrochloric acid regeneration reaction Methods 0.000 description 1
- 229960002591 hydroxyproline Drugs 0.000 description 1
- 230000001900 immune effect Effects 0.000 description 1
- 210000000987 immune system Anatomy 0.000 description 1
- 230000002163 immunogen Effects 0.000 description 1
- 230000005847 immunogenicity Effects 0.000 description 1
- 230000016784 immunoglobulin production Effects 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- ZPNFWUPYTFPOJU-LPYSRVMUSA-N iniprol Chemical compound C([C@H]1C(=O)NCC(=O)NCC(=O)N[C@H]2CSSC[C@H]3C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@H](C(N[C@H](C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=4C=CC(O)=CC=4)C(=O)N[C@@H](CC=4C=CC=CC=4)C(=O)N[C@@H](CC=4C=CC(O)=CC=4)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)NCC(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CSSC[C@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CC=4C=CC=CC=4)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCCCN)NC(=O)[C@H](C)NC(=O)[C@H](CCCNC(N)=N)NC2=O)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CSSC[C@H](NC(=O)[C@H](CC=2C=CC=CC=2)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H]2N(CCC2)C(=O)[C@@H](N)CCCNC(N)=N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N2[C@@H](CCC2)C(=O)N2[C@@H](CCC2)C(=O)N[C@@H](CC=2C=CC(O)=CC=2)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)N2[C@@H](CCC2)C(=O)N3)C(=O)NCC(=O)NCC(=O)N[C@@H](C)C(O)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@H](C(=O)N[C@@H](CC=2C=CC=CC=2)C(=O)N[C@H](C(=O)N1)C(C)C)[C@@H](C)O)[C@@H](C)CC)=O)[C@@H](C)CC)C1=CC=C(O)C=C1 ZPNFWUPYTFPOJU-LPYSRVMUSA-N 0.000 description 1
- 238000011081 inoculation Methods 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 230000004410 intraocular pressure Effects 0.000 description 1
- 238000007912 intraperitoneal administration Methods 0.000 description 1
- 239000007928 intraperitoneal injection Substances 0.000 description 1
- 230000002427 irreversible effect Effects 0.000 description 1
- 229960002725 isoflurane Drugs 0.000 description 1
- 239000007951 isotonicity adjuster Substances 0.000 description 1
- 210000003292 kidney cell Anatomy 0.000 description 1
- 238000002372 labelling Methods 0.000 description 1
- CIPMKIHUGVGQTG-VFFZMTJFSA-N leupeptin hemisulfate Chemical compound OS(O)(=O)=O.CC(C)C[C@H](NC(C)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@H](C=O)CCCN=C(N)N.CC(C)C[C@H](NC(C)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@H](C=O)CCCN=C(N)N CIPMKIHUGVGQTG-VFFZMTJFSA-N 0.000 description 1
- 206010024627 liposarcoma Diseases 0.000 description 1
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 1
- 201000005249 lung adenocarcinoma Diseases 0.000 description 1
- 201000005202 lung cancer Diseases 0.000 description 1
- 208000020816 lung neoplasm Diseases 0.000 description 1
- 201000005243 lung squamous cell carcinoma Diseases 0.000 description 1
- 210000002751 lymph Anatomy 0.000 description 1
- 239000006166 lysate Substances 0.000 description 1
- 125000003588 lysine group Chemical group [H]N([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])(N([H])[H])C(*)=O 0.000 description 1
- 230000036210 malignancy Effects 0.000 description 1
- 230000003211 malignant effect Effects 0.000 description 1
- 239000000594 mannitol Substances 0.000 description 1
- 235000010355 mannitol Nutrition 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 230000001404 mediated effect Effects 0.000 description 1
- 201000001441 melanoma Diseases 0.000 description 1
- 206010027191 meningioma Diseases 0.000 description 1
- MYWUZJCMWCOHBA-VIFPVBQESA-N methamphetamine Chemical compound CN[C@@H](C)CC1=CC=CC=C1 MYWUZJCMWCOHBA-VIFPVBQESA-N 0.000 description 1
- YACKEPLHDIMKIO-UHFFFAOYSA-N methylphosphonic acid Chemical class CP(O)(O)=O YACKEPLHDIMKIO-UHFFFAOYSA-N 0.000 description 1
- 238000013508 migration Methods 0.000 description 1
- 235000013336 milk Nutrition 0.000 description 1
- 210000004080 milk Anatomy 0.000 description 1
- 239000008267 milk Substances 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 108091005601 modified peptides Proteins 0.000 description 1
- 210000003097 mucus Anatomy 0.000 description 1
- 238000011201 multiple comparisons test Methods 0.000 description 1
- 201000000050 myeloid neoplasm Diseases 0.000 description 1
- 201000011519 neuroendocrine tumor Diseases 0.000 description 1
- 238000010606 normalization Methods 0.000 description 1
- 239000002773 nucleotide Substances 0.000 description 1
- 235000015097 nutrients Nutrition 0.000 description 1
- 210000001672 ovary Anatomy 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 201000002530 pancreatic endocrine carcinoma Diseases 0.000 description 1
- 230000001575 pathological effect Effects 0.000 description 1
- 230000007170 pathology Effects 0.000 description 1
- 230000037361 pathway Effects 0.000 description 1
- 230000007030 peptide scission Effects 0.000 description 1
- 210000003516 pericardium Anatomy 0.000 description 1
- 210000004303 peritoneum Anatomy 0.000 description 1
- 201000002628 peritoneum cancer Diseases 0.000 description 1
- 230000035699 permeability Effects 0.000 description 1
- 150000008298 phosphoramidates Chemical class 0.000 description 1
- BZQFBWGGLXLEPQ-REOHCLBHSA-N phosphoserine Chemical compound OC(=O)[C@@H](N)COP(O)(O)=O BZQFBWGGLXLEPQ-REOHCLBHSA-N 0.000 description 1
- 210000002381 plasma Anatomy 0.000 description 1
- 210000004224 pleura Anatomy 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 229920005862 polyol Polymers 0.000 description 1
- 150000003077 polyols Chemical class 0.000 description 1
- 239000000256 polyoxyethylene sorbitan monolaurate Substances 0.000 description 1
- 235000010486 polyoxyethylene sorbitan monolaurate Nutrition 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 230000003449 preventive effect Effects 0.000 description 1
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 238000004393 prognosis Methods 0.000 description 1
- 230000002062 proliferating effect Effects 0.000 description 1
- 230000000069 prophylactic effect Effects 0.000 description 1
- 238000011321 prophylaxis Methods 0.000 description 1
- 108020001580 protein domains Proteins 0.000 description 1
- 230000005180 public health Effects 0.000 description 1
- 238000003127 radioimmunoassay Methods 0.000 description 1
- 238000002708 random mutagenesis Methods 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 238000005215 recombination Methods 0.000 description 1
- 206010038038 rectal cancer Diseases 0.000 description 1
- 201000001275 rectum cancer Diseases 0.000 description 1
- 230000002829 reductive effect Effects 0.000 description 1
- 230000003362 replicative effect Effects 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 238000012552 review Methods 0.000 description 1
- 239000002336 ribonucleotide Substances 0.000 description 1
- 125000002652 ribonucleotide group Chemical group 0.000 description 1
- 102200118232 rs33959340 Human genes 0.000 description 1
- 102220005175 rs33961886 Human genes 0.000 description 1
- 102220141108 rs886056447 Human genes 0.000 description 1
- 108091008601 sVEGFR Proteins 0.000 description 1
- 210000003296 saliva Anatomy 0.000 description 1
- 210000004116 schwann cell Anatomy 0.000 description 1
- 238000007789 sealing Methods 0.000 description 1
- 210000004911 serous fluid Anatomy 0.000 description 1
- 239000012679 serum free medium Substances 0.000 description 1
- 230000001568 sexual effect Effects 0.000 description 1
- 230000019491 signal transduction Effects 0.000 description 1
- 238000002741 site-directed mutagenesis Methods 0.000 description 1
- 235000020183 skimmed milk Nutrition 0.000 description 1
- 230000037439 somatic mutation Effects 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 210000004989 spleen cell Anatomy 0.000 description 1
- 210000003802 sputum Anatomy 0.000 description 1
- 208000024794 sputum Diseases 0.000 description 1
- 238000010561 standard procedure Methods 0.000 description 1
- 230000037351 starvation Effects 0.000 description 1
- 238000007619 statistical method Methods 0.000 description 1
- 239000007929 subcutaneous injection Substances 0.000 description 1
- 238000010254 subcutaneous injection Methods 0.000 description 1
- 235000000346 sugar Nutrition 0.000 description 1
- 150000008163 sugars Chemical class 0.000 description 1
- 239000013595 supernatant sample Substances 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 230000004083 survival effect Effects 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 230000008961 swelling Effects 0.000 description 1
- 210000001179 synovial fluid Anatomy 0.000 description 1
- 210000002437 synoviocyte Anatomy 0.000 description 1
- 230000008685 targeting Effects 0.000 description 1
- 210000001138 tear Anatomy 0.000 description 1
- 201000002510 thyroid cancer Diseases 0.000 description 1
- 238000004448 titration Methods 0.000 description 1
- FGMPLJWBKKVCDB-UHFFFAOYSA-N trans-L-hydroxy-proline Natural products ON1CCCC1C(O)=O FGMPLJWBKKVCDB-UHFFFAOYSA-N 0.000 description 1
- 238000013518 transcription Methods 0.000 description 1
- 230000035897 transcription Effects 0.000 description 1
- 230000002103 transcriptional effect Effects 0.000 description 1
- 230000002463 transducing effect Effects 0.000 description 1
- 239000012096 transfection reagent Substances 0.000 description 1
- 230000009466 transformation Effects 0.000 description 1
- 230000001131 transforming effect Effects 0.000 description 1
- 230000009261 transgenic effect Effects 0.000 description 1
- 238000013519 translation Methods 0.000 description 1
- 239000012588 trypsin Substances 0.000 description 1
- 230000004565 tumor cell growth Effects 0.000 description 1
- 210000002700 urine Anatomy 0.000 description 1
- 206010046766 uterine cancer Diseases 0.000 description 1
- 230000008728 vascular permeability Effects 0.000 description 1
- 230000003612 virological effect Effects 0.000 description 1
- 210000004127 vitreous body Anatomy 0.000 description 1
- 201000005102 vulva cancer Diseases 0.000 description 1
- 230000003442 weekly effect Effects 0.000 description 1
- 238000009736 wetting Methods 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/22—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against growth factors ; against growth regulators
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Immunology (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Mycology (AREA)
- Genetics & Genomics (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Microbiology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Biochemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Biophysics (AREA)
- Epidemiology (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Peptides Or Proteins (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
Abstract
Description
本揭露屬於生物製藥領域,具體地,本揭露關於抗ANG-2抗體以及抗ANG-2和抗VEGF的雙特異性抗原結合分子的製備和應用。 The present disclosure belongs to the field of biopharmaceuticals, and in particular, the present disclosure relates to the preparation and application of anti-ANG-2 antibodies and bispecific antigen-binding molecules of anti-ANG-2 and anti-VEGF.
這裡的陳述僅提供與本揭露有關的背景信息,而不必然地構成現有技術。 The statements herein merely provide background information related to the present disclosure and do not necessarily constitute prior art.
新血管的生成為腫瘤細胞提供氧氣和養料,使得腫瘤細胞獲得生長優勢,從無血管的慢速生長期到有血管的快速增長期。因此,藉由抑制血管生成來抑制腫瘤的生長是一個較有潛力的有效策略。在眾多促進血管生成的相關因子中,血管內皮生長因子VEGF是非常關鍵和重要的促進血管生成的因子。VEGF可以藉由與VEGF受體結合促進細胞的增殖、遷移、增加血管通透性等促進腫瘤細胞的新生血管生成。因此藉由阻斷VEGF可抑制腫瘤血管的生成,進而達到抑制腫瘤生長和轉移的目的。臨床上有很多藉由不同策略阻斷VEGF的生物製劑,諸如針對VEGF的單抗阿瓦斯汀(Avastin)、中和VEGF的可溶性VEGF受體、針對VEGF受體的單抗等都顯示出較好的活性。 The formation of new blood vessels provides oxygen and nutrients to tumor cells, enabling tumor cells to gain growth advantages, from a slow growth period without blood vessels to a rapid growth period with blood vessels. Therefore, inhibiting tumor growth by inhibiting angiogenesis is a potentially effective strategy. Among the many factors related to promoting angiogenesis, vascular endothelial growth factor VEGF is a very critical and important factor promoting angiogenesis. VEGF can promote the neovascularization of tumor cells by binding to VEGF receptors to promote cell proliferation, migration, and increase vascular permeability. Therefore, by blocking VEGF, tumor angiogenesis can be inhibited, thereby achieving the purpose of inhibiting tumor growth and metastasis. There are many biological agents that block VEGF by different strategies, such as Avastin, a monoclonal antibody against VEGF, soluble VEGF receptors that neutralize VEGF, and monoclonal antibodies against VEGF receptors, all of which have shown better results. activity.
但腫瘤血管的生成是由眾多分子、多信號通路參與的複雜過程,藉由阻斷一條通路仍不能達到完全抑制腫瘤的目的,需要同時阻斷其他血管生成相關因子。 However, tumor angiogenesis is a complex process involving many molecules and multiple signaling pathways. By blocking one pathway, the goal of completely suppressing tumors cannot be achieved, and other angiogenesis-related factors need to be blocked at the same time.
Tie2是第二個被鑑定出的血管內皮細胞特異的酪胺酸激酶受體,其與配體血管生成素-1(ANG1)和血管生成素-2(ANG2)的結合對於血管生成也發揮重要作用。ANG1與ANG2都結合Tie2,其中ANG1支持內皮細胞(EC)存活並促進血管的完整性和穩定性,而ANG2具有相反效應,可以使周邊細胞從內皮細胞脫落下來,導致內皮細胞通透性提高,使VEGF發揮促進新生血管形成的作用。ANG2和VEGF在腫瘤的血管形成過程中互補協調,共同作用。因此,同時阻斷VEGF和ANG2可以更有效抑制血管的生成,促進血管的正常化,達到抑制腫瘤生長和轉移的目的。 Tie2 is the second vascular endothelial cell-specific tyrosine kinase receptor identified, and its binding to the ligands angiopoietin-1 (ANG1) and angiopoietin-2 (ANG2) is also important for angiogenesis effect. Both ANG1 and ANG2 bind Tie2, where ANG1 supports endothelial cell (EC) survival and promotes the integrity and stability of blood vessels, while ANG2 has the opposite effect, shedding peripheral cells from endothelial cells, resulting in increased endothelial cell permeability, Make VEGF play a role in promoting the formation of new blood vessels. ANG2 and VEGF complement each other and work together in tumor angiogenesis. Therefore, blocking VEGF and ANG2 at the same time can more effectively inhibit angiogenesis, promote the normalization of blood vessels, and achieve the purpose of inhibiting tumor growth and metastasis.
目前,專利申請WO1998045332、WO2007095338A2、WO201004058、CN102250247A、WO2011117329等中公開了抗ANG-2和VEGF的雙特異性抗體或VEGF抗體,但仍有待於開發新的高效的抗ANG-2和VEGF的雙特異性抗體。 At present, patent applications WO1998045332, WO2007095338A2, WO201004058, CN102250247A, WO2011117329, etc. disclose anti-ANG-2 and VEGF bispecific antibodies or VEGF antibodies, but there is still a need to develop new and efficient anti-ANG-2 and VEGF bispecific antibodies Sexual antibodies.
本揭露提供了一種靶向ANG-2和VEGF的雙特異性抗體,其在體外,具有更好的阻斷ANG-2與其受體Tie2結合的阻斷活性,並可顯著抑制VEGF引起的HUVEC細胞內磷酸化VEGFR水平的升高,抑制VEGF引起的HUVEC細胞的增殖。除此之外,本揭露中的雙特異性抗體,在小鼠體內具有更優異的抑制腫瘤生長的效果,並可用於治療眼部疾病,如AMD。 The present disclosure provides a bispecific antibody targeting ANG-2 and VEGF, which has better blocking activity of blocking the binding of ANG-2 to its receptor Tie2 in vitro, and can significantly inhibit VEGF-induced HUVEC cells Elevated levels of endophosphorylated VEGFR inhibited the proliferation of HUVEC cells induced by VEGF. Besides, the bispecific antibodies in the present disclosure have more excellent tumor growth inhibitory effect in mice, and can be used to treat eye diseases such as AMD.
在一些實施方案中,本揭露提供一種雙特異性抗原結合分子,其包含特異性結合ANG-2的第一抗原結合域和特異性結合VEGF的第二抗原結合域,其中該特異性結合ANG-2的第一抗原結合域包含: In some embodiments, the present disclosure provides a bispecific antigen-binding molecule comprising a first antigen-binding domain that specifically binds ANG-2 and a second antigen-binding domain that specifically binds VEGF, wherein the specifically binds ANG- The first antigen-binding domain of 2 contains:
i)重鏈可變區,其包含分別如SEQ ID NO:7,SEQ ID NO:38和SEQ ID NO:39所示的HCDR1,HCDR2和HCDR3,和 i) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO:7, SEQ ID NO:38 and SEQ ID NO:39, respectively, and
輕鏈可變區,其包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3;其中, A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as shown in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12, respectively; wherein,
SEQ ID NO:38的序列為:TINX1X2SSYTYYPDNVKG; The sequence of SEQ ID NO: 38 is: TINX 1 X 2 SSYTYYPDNVKG;
SEQ ID NO:39的序列為:X3X4ATGX5FDY The sequence of SEQ ID NO: 39 is: X 3 X 4 ATGX 5 FDY
其中,X1為D或E,X2為D或N,X3為D或N,X4為E或Q,X5為C、S或V; Wherein, X 1 is D or E, X 2 is D or N, X 3 is D or N, X 4 is E or Q, and X 5 is C, S or V;
或 or
ii)重鏈可變區,其包含分別如SEQ ID NO:7,SEQ ID NO:13和SEQ ID NO:14所示的HCDR1,HCDR2和HCDR3,和 ii) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO: 7, SEQ ID NO: 13 and SEQ ID NO: 14, respectively, and
輕鏈可變區,其包含分別如SEQ ID NO:15,SEQ ID NO:11和SEQ ID NO:16所示的LCDR1,LCDR2和LCDR3。 A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as set forth in SEQ ID NO: 15, SEQ ID NO: 11 and SEQ ID NO: 16, respectively.
在一些實施方案中,前述的雙特異性抗原結合分子,其中該特異性結合ANG-2的第一抗原結合域包含: In some embodiments, the aforementioned bispecific antigen binding molecule, wherein the first antigen binding domain that specifically binds ANG-2 comprises:
重鏈可變區,其包含如SEQ ID NO:7所示的HCDR1,如SEQ ID NO:8、22或24所示的HCDR2,和如SEQ ID NO:9、23、25、26或27所示的HCDR3;和 A heavy chain variable region comprising HCDR1 as set forth in SEQ ID NO:7, HCDR2 as set forth in SEQ ID NO:8, 22 or 24, and HCDR2 as set forth in SEQ ID NO:9, 23, 25, 26 or 27 HCDR3 shown; and
輕鏈可變區,其包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3。 A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as set forth in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12, respectively.
在一些實施方案中,前述的雙特異性抗原結合分子,其中該特異性結合ANG-2的第一抗原結合域包含: In some embodiments, the aforementioned bispecific antigen binding molecule, wherein the first antigen binding domain that specifically binds ANG-2 comprises:
a)重鏈可變區,其包含分別如SEQ ID NO:7,SEQ ID NO:22和SEQ ID NO:23所示的HCDR1,HCDR2和HCDR3;和 a) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO:7, SEQ ID NO:22 and SEQ ID NO:23, respectively; and
輕鏈可變區,其包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3; A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as shown in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12, respectively;
b)重鏈可變區,其包含分別如SEQ ID NO:7,SEQ ID NO:24和SEQ ID NO:23所示的HCDR1,HCDR2和HCDR3;和 b) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO:7, SEQ ID NO:24 and SEQ ID NO:23, respectively; and
輕鏈可變區,其包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3; A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as shown in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12, respectively;
c)重鏈可變區,其包含分別如SEQ ID NO:7,SEQ ID NO:8和SEQ ID NO:25所示的HCDR1,HCDR2和HCDR3;和 c) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO:7, SEQ ID NO:8 and SEQ ID NO:25, respectively; and
輕鏈可變區,其包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3; A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as shown in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12, respectively;
d)重鏈可變區,其包含分別如SEQ ID NO:7,SEQ ID NO:8和SEQ ID NO:26所示的HCDR1,HCDR2和HCDR3;和 d) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO:7, SEQ ID NO:8 and SEQ ID NO:26, respectively; and
輕鏈可變區,其包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3; A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as shown in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12, respectively;
e)重鏈可變區,其包含分別如SEQ ID NO:7,SEQ ID NO:22和SEQ ID NO:25所示的HCDR1,HCDR2和HCDR3;和 e) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO:7, SEQ ID NO:22 and SEQ ID NO:25, respectively; and
輕鏈可變區,其包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3; A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as shown in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12, respectively;
f)重鏈可變區,其包含分別如SEQ ID NO:7,SEQ ID NO:22和SEQ ID NO:26所示的HCDR1,HCDR2和HCDR3;和 f) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO:7, SEQ ID NO:22 and SEQ ID NO:26, respectively; and
輕鏈可變區,其包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3; A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as shown in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12, respectively;
g)重鏈可變區,其包含分別如SEQ ID NO:7,SEQ ID NO:24和SEQ ID NO:25所示的HCDR1,HCDR2和HCDR3;和 g) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO:7, SEQ ID NO:24 and SEQ ID NO:25, respectively; and
輕鏈可變區,其包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3; A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as shown in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12, respectively;
h)重鏈可變區,包含分別如SEQ ID NO:7,SEQ ID NO:24和SEQ ID NO:26所示的HCDR1,HCDR2和HCDR3;和 h) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO:7, SEQ ID NO:24 and SEQ ID NO:26, respectively; and
輕鏈可變區,其包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3; A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as shown in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12, respectively;
j)重鏈可變區,其包含分別如SEQ ID NO:7,SEQ ID NO:8和SEQ ID NO:23所示的HCDR1,HCDR2和HCDR3;和 j) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO: 7, SEQ ID NO: 8 and SEQ ID NO: 23, respectively; and
輕鏈可變區,其包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3;或 A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as shown in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12, respectively; or
k)重鏈可變區,其包含分別如SEQ ID NO:7,SEQ ID NO:22和SEQ ID NO:27所示的HCDR1,HCDR2和HCDR3;和 k) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO:7, SEQ ID NO:22 and SEQ ID NO:27, respectively; and
輕鏈可變區,其包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3;或 A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as shown in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12, respectively; or
l)重鏈可變區,其包含分別如SEQ ID NO:7,SEQ ID NO:8和SEQ ID NO:9所示的HCDR1,HCDR2和HCDR3;和 1) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO: 7, SEQ ID NO: 8 and SEQ ID NO: 9, respectively; and
輕鏈可變區,其包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3。 A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as set forth in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12, respectively.
在一些實施方式中,前述雙特異性抗原結合分子以等於或小於10-7M解離平衡常數與人ANG-2結合,在一些實施方式中,以等於或小於10-8M或10-9M解離平衡常數與人ANG-2結合。 In some embodiments, the aforementioned bispecific antigen binding molecules bind to human ANG-2 with a dissociation equilibrium constant equal to or less than 10-7 M, and in some embodiments, with a dissociation equilibrium constant equal to or less than 10-8 M or 10-9 M Dissociation equilibrium constants for human ANG-2 binding.
在一些實施方式中,前述雙特異性抗原結合分子以等於或小於10-7M解離平衡常數與人VEGF結合,在一些實施方式中,以等於或小於10-8M、10-9M、10-10M、10-11M、10-12M或10-13M解離平衡常數與人VEGF結合。 In some embodiments, the aforementioned bispecific antigen binding molecules bind to human VEGF with a dissociation equilibrium constant equal to or less than 10-7 M, in some embodiments, with a dissociation equilibrium constant equal to or less than 10-8 M, 10-9 M, 10 -10 M, 10 -11 M, 10 -12 M or 10 -13 M dissociation equilibrium constant for binding to human VEGF.
在一些實施方案中,前述雙特異性抗原結合分子與猴的ANG-2和VEGF可交叉結合。 In some embodiments, the aforementioned bispecific antigen binding molecules are cross-binding to monkey ANG-2 and VEGF.
在一些實施方案中,前述的雙特異性抗原結合分子以小於24.82nM、小於20nM、小於15nM、小於10nM、小於5nM、小於3nM、小於1nM、或小於0.5nM的IC50阻斷ANG-2與ANG-2受體Tie2結合,其中該阻斷活性藉由測試例2該ELISA實驗檢測。 In some embodiments, the aforementioned bispecific antigen binding molecules block ANG-2 and ANG with an IC50 of less than 24.82 nM, less than 20 nM, less than 15 nM, less than 10 nM, less than 5 nM, less than 3 nM, less than 1 nM, or less than 0.5 nM -2 receptor Tie2 binding, wherein the blocking activity was detected by the ELISA assay of Test Example 2.
在一些實施方案中,前述的雙特異性抗原結合分子以小於21.27nM、小於15nM、小於10nM、小於8nM或小於5nM的IC50抑制經Tie2轉染的CHO細胞中ANG-2誘導的Tie2磷酸化。 In some embodiments, the aforementioned bispecific antigen binding molecules inhibit ANG-2-induced Tie2 phosphorylation in Tie2-transfected CHO cells with an IC50 of less than 21.27 nM, less than 15 nM, less than 10 nM, less than 8 nM, or less than 5 nM.
在一些實施方案中,前述的雙特異性抗原結合分子可顯著抑制VEGF引起的HUVEC胞內磷酸化VEGFR水平升高。 In some embodiments, the aforementioned bispecific antigen binding molecules can significantly inhibit VEGF-induced elevation of intracellular phosphorylated VEGFR levels in HUVECs.
在一些實施方案中,前述的雙特異性抗原結合分子以小於18nM、小於10nM、或小於5nM的IC50抑制VEGF誘導的HUVEC細胞增殖。 In some embodiments, the aforementioned bispecific antigen binding molecules inhibit VEGF-induced HUVEC cell proliferation with an IC50 of less than 18 nM, less than 10 nM, or less than 5 nM.
在一些實施方案中,前述的雙特異性抗原結合分子,其中該特異性結合ANG-2的第一抗原結合域包含: In some embodiments, the aforementioned bispecific antigen binding molecule, wherein the first antigen binding domain that specifically binds ANG-2 comprises:
i)重鏈可變區,包含SEQ ID NO:3、20、21、28-37、72或73中任一胺基酸序列,或包含與SEQ ID NO:3、20、21、28-37、72或73中任一序列分別具有至少95%,96%,97%,98%或99%的序列同一性的胺基酸序列;和/或 i) a heavy chain variable region comprising the amino acid sequence of any one of SEQ ID NO: 3, 20, 21, 28-37, 72 or 73, or comprising the same amino acid sequence as SEQ ID NO: 3, 20, 21, 28-37 An amino acid sequence having at least 95%, 96%, 97%, 98% or 99% sequence identity, respectively, in any of the sequences , 72 or 73; and/or
輕鏈可變區,包含SEQ ID NO:4、17、18、19或74的胺基酸序列,或包含與SEQ ID NO:4、17、18、19或74分別具有至少95%、96%、97%、98%或99%的序列同一性的胺基酸序列;或 A light chain variable region comprising the amino acid sequence of SEQ ID NO: 4, 17, 18, 19 or 74, or comprising at least 95%, 96% with SEQ ID NO: 4, 17, 18, 19 or 74, respectively , 97%, 98% or 99% sequence identity of amino acid sequences; or
ii)重鏈可變區,包含SEQ ID NO:5、44、45、46或75的胺基酸序列,或包含與SEQ ID NO:5、44、45、46或75分別具有至少95%、96%、97%、98%或99%的序列同一性的胺基酸序列;和/或 ii) a heavy chain variable region comprising the amino acid sequence of SEQ ID NO: 5, 44, 45, 46 or 75, or comprising at least 95%, amino acid sequences of 96%, 97%, 98% or 99% sequence identity; and/or
輕鏈可變區,包含SEQ ID NO:6、40、41、42、43或76的胺基酸序列,或包含與SEQ ID NO:6、40、41、42、43或76分別具有至少95%、96%、97%、98%或99%的序列同一性的胺基酸序列。 A light chain variable region comprising the amino acid sequence of SEQ ID NO: 6, 40, 41, 42, 43 or 76, or comprising at least 95 SEQ ID NO: 6, 40, 41, 42, 43 or 76, respectively %, 96%, 97%, 98% or 99% sequence identity of amino acid sequences.
在一些實施方案中,前述的雙特異性抗原結合分子,其中該特異性結合ANG-2的第一抗原結合域包含: In some embodiments, the aforementioned bispecific antigen binding molecule, wherein the first antigen binding domain that specifically binds ANG-2 comprises:
i)重鏈可變區,其胺基酸序列如SEQ ID NO:3、20、21、28-37、72或73中任一所示,或與SEQ ID NO:3、20、21、28-37、72或73分別具有至少95%、96%、97%、98%或99%的序列同一性;和/或 i) a heavy chain variable region, the amino acid sequence of which is shown in any of SEQ ID NO: 3, 20, 21, 28-37, 72 or 73, or the same as SEQ ID NO: 3, 20, 21, 28 - 37, 72 or 73 have at least 95%, 96%, 97%, 98% or 99% sequence identity, respectively; and/or
輕鏈可變區,其胺基酸序列如SEQ ID NO:4、17、18、19或74所示,或與SEQ ID NO:4、17、18、19或74分別具有至少95%、96%、97%、98%或99%的序列同一性;或 A light chain variable region, the amino acid sequence of which is shown in SEQ ID NO: 4, 17, 18, 19 or 74, or has at least 95%, 96% and 96% respectively with SEQ ID NO: 4, 17, 18, 19 or 74 %, 97%, 98% or 99% sequence identity; or
ii)重鏈可變區,其胺基酸序列如SEQ ID NO:5、44、45、46或75所示,或與SEQ ID NO:5、44、45、46或75分別具有至少95%、96%、97%、98%或99%的序列同一性;和/或 ii) a heavy chain variable region, the amino acid sequence of which is as shown in SEQ ID NO: 5, 44, 45, 46 or 75, or at least 95% of the same as SEQ ID NO: 5, 44, 45, 46 or 75, respectively , 96%, 97%, 98% or 99% sequence identity; and/or
輕鏈可變區,其胺基酸序列如SEQ ID NO:6、40、41、42、43或76所示,或與SEQ ID NO:6、40、41、42、43或76分別具有至少95%、96%、97%、98%或99%的序列同一性。 A light chain variable region, the amino acid sequence of which is shown in SEQ ID NO: 6, 40, 41, 42, 43 or 76, or has at least SEQ ID NO: 6, 40, 41, 42, 43 or 76, respectively 95%, 96%, 97%, 98% or 99% sequence identity.
在一些實施方案中,前述的雙特異性抗原結合分子,其中該特異性結合ANG-2的第一抗原結合域包含: In some embodiments, the aforementioned bispecific antigen binding molecule, wherein the first antigen binding domain that specifically binds ANG-2 comprises:
a)重鏈可變區,包含SEQ ID NO:3的胺基酸序列;和輕鏈可變區,包含SEQ ID NO:4的胺基酸序列; a) a heavy chain variable region comprising the amino acid sequence of SEQ ID NO:3; and a light chain variable region comprising the amino acid sequence of SEQ ID NO:4;
b)重鏈可變區,包含SEQ ID NO:20或21的胺基酸序列;和輕鏈可變區,包含SEQ ID NO:17、18或19的胺基酸序列; b) a heavy chain variable region comprising the amino acid sequence of SEQ ID NO: 20 or 21; and a light chain variable region comprising the amino acid sequence of SEQ ID NO: 17, 18 or 19;
c)重鏈可變區,包含SEQ ID NO:28-37中任一所示的胺基酸序列;和輕鏈可變區,包含SEQ ID NO:17的胺基酸序列; c) a heavy chain variable region comprising the amino acid sequence set forth in any of SEQ ID NOs: 28-37; and a light chain variable region comprising the amino acid sequence of SEQ ID NO: 17;
d)重鏈可變區,包含SEQ ID NO:72或73的胺基酸序列;和輕鏈可變區,包含SEQ ID NO:74的胺基酸序列; d) a heavy chain variable region comprising the amino acid sequence of SEQ ID NO: 72 or 73; and a light chain variable region comprising the amino acid sequence of SEQ ID NO: 74;
e)重鏈可變區,包含SEQ ID NO:5的胺基酸序列;和輕鏈可變區,包含SEQ ID NO:6的胺基酸序列; e) a heavy chain variable region comprising the amino acid sequence of SEQ ID NO:5; and a light chain variable region comprising the amino acid sequence of SEQ ID NO:6;
f)重鏈可變區,包含SEQ ID NO:44、45或46的胺基酸序列;和輕鏈可變區,包含SEQ ID NO:40、41、42或43的胺基酸序列;或 f) a heavy chain variable region comprising the amino acid sequence of SEQ ID NO: 44, 45 or 46; and a light chain variable region comprising the amino acid sequence of SEQ ID NO: 40, 41, 42 or 43; or
g)重鏈可變區,包含SEQ ID NO:75的胺基酸序列;和輕鏈可變區,包含SEQ ID NO:76的胺基酸序列。在一些實施方案中,前述的雙特異性抗原結合分子,其中該特異性結合ANG-2的第一抗原結合域包含: g) a heavy chain variable region comprising the amino acid sequence of SEQ ID NO:75; and a light chain variable region comprising the amino acid sequence of SEQ ID NO:76. In some embodiments, the aforementioned bispecific antigen binding molecule, wherein the first antigen binding domain that specifically binds ANG-2 comprises:
a)如SEQ ID NO:3所示的重鏈可變區;和如SEQ ID NO:4所示的輕鏈可變區; a) a heavy chain variable region as set forth in SEQ ID NO:3; and a light chain variable region as set forth in SEQ ID NO:4;
b)如SEQ ID NO:20或21所示的重鏈可變區;和如SEQ ID NO:17、18或19所示的輕鏈可變區; b) a heavy chain variable region as set forth in SEQ ID NO: 20 or 21; and a light chain variable region as set forth in SEQ ID NO: 17, 18 or 19;
c)如SEQ ID NO:28-37中任一所示的重鏈可變區;和如SEQ ID NO:17所示的輕鏈可變區; c) a heavy chain variable region as set forth in any of SEQ ID NOs: 28-37; and a light chain variable region as set forth in SEQ ID NO: 17;
d)如SEQ ID NO:72或73所示的重鏈可變區;和如SEQ ID NO:74所示的輕鏈可變區; d) a heavy chain variable region as set forth in SEQ ID NO: 72 or 73; and a light chain variable region as set forth in SEQ ID NO: 74;
e)如SEQ ID NO:5所示的重鏈可變區;和如SEQ ID NO:6所示的輕鏈可變區; e) a heavy chain variable region as set forth in SEQ ID NO:5; and a light chain variable region as set forth in SEQ ID NO:6;
f)如SEQ ID NO:44、45或46所示的重鏈可變區;和如SEQ ID NO:40、41、42或43所示的輕鏈可變區;或 f) a heavy chain variable region as set forth in SEQ ID NO: 44, 45 or 46; and a light chain variable region as set forth in SEQ ID NO: 40, 41, 42 or 43; or
g)如SEQ ID NO:75所示的重鏈可變區;和如SEQ ID NO:76所示的輕鏈可變區。 g) a heavy chain variable region as set forth in SEQ ID NO:75; and a light chain variable region as set forth in SEQ ID NO:76.
在一些實施方案中,前述的雙特異性抗原結合分子,其中該特異性結合ANG-2的第一抗原結合域包含: In some embodiments, the aforementioned bispecific antigen binding molecule, wherein the first antigen binding domain that specifically binds ANG-2 comprises:
a)SEQ ID NO:20、21或33的重鏈可變區和SEQ ID NO:17的輕鏈可變區; a) the heavy chain variable region of SEQ ID NO: 20, 21 or 33 and the light chain variable region of SEQ ID NO: 17;
b)SEQ ID NO:44或45的重鏈可變區和SEQ ID NO:40的輕鏈可變區; b) the heavy chain variable region of SEQ ID NO: 44 or 45 and the light chain variable region of SEQ ID NO: 40;
c)SEQ ID NO:72或73的重鏈可變區和SEQ ID NO:74的輕鏈可變區;或 c) the heavy chain variable region of SEQ ID NO: 72 or 73 and the light chain variable region of SEQ ID NO: 74; or
d)SEQ ID NO:75的重鏈可變區和SEQ ID NO:76的輕鏈可變區。 d) The heavy chain variable region of SEQ ID NO:75 and the light chain variable region of SEQ ID NO:76.
在一些實施方案中,前述的雙特異性抗原結合分子,其中該特異性結合ANG-2的第一抗原結合域包含: In some embodiments, the aforementioned bispecific antigen binding molecule, wherein the first antigen binding domain that specifically binds ANG-2 comprises:
重鏈可變區,包含SEQ ID NO:33的胺基酸序列,和輕鏈可變區,包含SEQ ID NO:17的胺基酸序列;或 A heavy chain variable region, comprising the amino acid sequence of SEQ ID NO: 33, and a light chain variable region, comprising the amino acid sequence of SEQ ID NO: 17; or
重鏈可變區,包含SEQ ID NO:45的胺基酸序列,和輕鏈可變區,包含SEQ ID NO:40的胺基酸序列。在一些實施方案中,前述的雙特異性抗原結合分子,其中該特異性結合ANG-2的第一抗原結合域包含: The heavy chain variable region, comprising the amino acid sequence of SEQ ID NO:45, and the light chain variable region, comprising the amino acid sequence of SEQ ID NO:40. In some embodiments, the aforementioned bispecific antigen binding molecule, wherein the first antigen binding domain that specifically binds ANG-2 comprises:
如SEQ ID NO:33所示的重鏈可變區,和如SEQ ID NO:17所示的輕鏈可變區;或 A heavy chain variable region as set forth in SEQ ID NO:33, and a light chain variable region as set forth in SEQ ID NO:17; or
如SEQ ID NO:45所示的重鏈可變區,和如SEQ ID NO:40所示的輕鏈可變區。 The heavy chain variable region is shown in SEQ ID NO:45, and the light chain variable region is shown in SEQ ID NO:40.
在一些實施方案中,前述的雙特異性抗原結合分子,其中該特異性結合VEGF的第二抗原結合域包含: In some embodiments, the aforementioned bispecific antigen binding molecule, wherein the second antigen binding domain that specifically binds VEGF comprises:
重鏈可變區,包含分別如SEQ ID NO:61,SEQ ID NO:62和SEQ ID NO:63所示的HCDR1,HCDR2和HCDR3;和 a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO: 61, SEQ ID NO: 62 and SEQ ID NO: 63, respectively; and
輕鏈可變區,包含分別如SEQ ID NO:64,SEQ ID NO:65和SEQ ID NO:66所示的LCDR1,LCDR2和LCDR3。 The light chain variable region comprises LCDR1, LCDR2 and LCDR3 as set forth in SEQ ID NO:64, SEQ ID NO:65 and SEQ ID NO:66, respectively.
在一些實施方案中,前述的雙特異性抗原結合分子,其中該特異性結合VEGF的第二抗原結合域包含: In some embodiments, the aforementioned bispecific antigen binding molecule, wherein the second antigen binding domain that specifically binds VEGF comprises:
重鏈可變區,包含SEQ ID NO:58的胺基酸序列,和輕鏈可變區,包含SEQ ID NO:56的胺基酸序列。在一些實施方案中,前述的雙特異性抗原結合分子,其中該特異性結合VEGF的第二抗原結合域包含: The heavy chain variable region, comprising the amino acid sequence of SEQ ID NO:58, and the light chain variable region, comprising the amino acid sequence of SEQ ID NO:56. In some embodiments, the aforementioned bispecific antigen binding molecule, wherein the second antigen binding domain that specifically binds VEGF comprises:
如SEQ ID NO:58所示的重鏈可變區,和如SEQ ID NO:56所示的輕鏈可變區。 The heavy chain variable region is shown in SEQ ID NO:58, and the light chain variable region is shown in SEQ ID NO:56.
在一些實施方案中,前述的雙特異性抗原結合分子,其中該特異性結合人VEGF的第二抗原結合域進一步包含重鏈恆定區和輕鏈恆定區。 In some embodiments, the aforementioned bispecific antigen binding molecule, wherein the second antigen binding domain that specifically binds human VEGF further comprises a heavy chain constant region and a light chain constant region.
在一些實施方案中,前述的雙特異性抗原結合分子,其中該特異性結合人VEGF的第二抗原結合域的重鏈恆定區包含L234A、L235A、I253A、H310A和H435A突變。 In some embodiments, the aforementioned bispecific antigen binding molecule, wherein the heavy chain constant region of the second antigen binding domain that specifically binds human VEGF comprises L234A, L235A, I253A, H310A and H435A mutations.
在一些實施方案中,前述的雙特異性抗原結合分子,其中該特異性結合人VEGF的第二抗原結合域的重鏈恆定區為IgG1恆定區,並包含L234A、L235A、I253A、H310A和H435A突變(EU編號)。 In some embodiments, the aforementioned bispecific antigen binding molecule, wherein the heavy chain constant region of the second antigen binding domain that specifically binds human VEGF is an IgGl constant region and comprises L234A, L235A, I253A, H310A and H435A mutations (EU number).
在一些實施方案中,前述的雙特異性抗原結合分子,其中該特異性結合VEGF的第二抗原結合域包含: In some embodiments, the aforementioned bispecific antigen binding molecule, wherein the second antigen binding domain that specifically binds VEGF comprises:
重鏈,包含SEQ ID NO:59或60的胺基酸序列,或與SEQ ID NO:59或60具有至少95%、96%、97%、98%或99%的序列同一性的胺基酸序列;和 A heavy chain comprising the amino acid sequence of SEQ ID NO: 59 or 60, or an amino acid having at least 95%, 96%, 97%, 98% or 99% sequence identity to SEQ ID NO: 59 or 60 sequence; and
輕鏈,包含SEQ ID NO:57的胺基酸序列,或與SEQ ID NO:57具有至少95%、96%、97%、98%或99%的序列同一性的胺基酸序列。在一些實施方案中,前述的雙特異性抗原結合分子,其中該特異性結合VEGF的第二抗原結合域包含: A light chain comprising the amino acid sequence of SEQ ID NO:57, or an amino acid sequence having at least 95%, 96%, 97%, 98% or 99% sequence identity to SEQ ID NO:57. In some embodiments, the aforementioned bispecific antigen binding molecule, wherein the second antigen binding domain that specifically binds VEGF comprises:
重鏈,其胺基酸序列如SEQ ID NO:59或60所示,或與SEQ ID NO:59或60具有至少95%、96%、97%、98%或99%的序列同一性;和 A heavy chain whose amino acid sequence is set forth in SEQ ID NO: 59 or 60, or has at least 95%, 96%, 97%, 98% or 99% sequence identity to SEQ ID NO: 59 or 60; and
輕鏈,其胺基酸序列如SEQ ID NO:57所示,或與SEQ ID NO:57具有至少95%、96%、97%、98%或99%的序列同一性。 A light chain whose amino acid sequence is set forth in SEQ ID NO:57, or has at least 95%, 96%, 97%, 98% or 99% sequence identity to SEQ ID NO:57.
在一些實施方案中,前述的雙特異性抗原結合分子,其中該特異性結合VEGF的第二抗原結合域包含: In some embodiments, the aforementioned bispecific antigen binding molecule, wherein the second antigen binding domain that specifically binds VEGF comprises:
抗原結合分子,其中該特異性結合VEGF的第二抗原結合域包含: An antigen-binding molecule, wherein the second antigen-binding domain that specifically binds VEGF comprises:
如SEQ ID NO:59所示的重鏈;和 A heavy chain as set forth in SEQ ID NO: 59; and
如SEQ ID NO:57所示的輕鏈。 A light chain as set forth in SEQ ID NO:57.
在一些實施方案中,前述的雙特異性抗原結合分子,其中該特異性結合VEGF的第二抗原結合域包含: In some embodiments, the aforementioned bispecific antigen binding molecule, wherein the second antigen binding domain that specifically binds VEGF comprises:
如SEQ ID NO:60所示的重鏈;和 A heavy chain as set forth in SEQ ID NO: 60; and
如SEQ ID NO:57所示的輕鏈。 A light chain as set forth in SEQ ID NO:57.
前述的雙特異性抗原結合分子可具有本領域中公知的任意的雙特異性抗體的分子結構。具體地,前述的雙特異性抗原結合分子可以具有含Fc片段的雙特異性抗體的分子結構,或者不含Fc片段的雙特異性抗體的分子結構。更具體地,該含Fc片段的雙特異性抗體的分子結構包括但不限於TrioMab、 Crossmab/KIH、DVD-Ig、IgG-scFv、FIT-Ig、mAb-Trap。不含Fc片段的雙特異性抗體的分子結構包括但不限於BiTE、DART、TandAb、ImmTAC、TriKE。 The aforementioned bispecific antigen-binding molecule may have the molecular structure of any bispecific antibody known in the art. Specifically, the aforementioned bispecific antigen-binding molecule may have the molecular structure of a bispecific antibody containing an Fc fragment, or the molecular structure of a bispecific antibody that does not contain an Fc fragment. More specifically, the molecular structure of the Fc fragment-containing bispecific antibody includes but is not limited to TrioMab, Crossmab/KIH, DVD-Ig, IgG-scFv, FIT-Ig, mAb-Trap. Molecular structures of bispecific antibodies without Fc fragments include, but are not limited to, BiTE, DART, TandAb, ImmTAC, TriKE.
在一些實施方案中,前述雙特異性抗原結合分子是IgG-scFv形式。 In some embodiments, the aforementioned bispecific antigen binding molecule is in the form of an IgG-scFv.
在IgG-scFv形式的雙特異性抗體示例性的實施方案中,該特異性結合ANG-2的第一抗原結合域為抗ANG-2單鏈抗體(scFv),該特異性結合VEGF的第二抗原結合域為全長IgG抗體,其中該抗ANG-2單鏈抗體直接地或藉由連接子與特異性結合VEGF的全長IgG抗體重鏈或輕鏈的C端連接。 In an exemplary embodiment of the bispecific antibody in the form of an IgG-scFv, the first antigen-binding domain that specifically binds ANG-2 is an anti-ANG-2 single chain antibody (scFv), the second that specifically binds VEGF The antigen binding domain is a full-length IgG antibody, wherein the anti-ANG-2 single chain antibody is linked directly or via a linker to the C-terminus of a full-length IgG antibody heavy or light chain that specifically binds VEGF.
在一些實施方案中,前述的雙特異性抗原結合分子,其中該特異性結合ANG-2的第一抗原結合域為抗ANG-2單鏈抗體。 In some embodiments, the aforementioned bispecific antigen binding molecule, wherein the first antigen binding domain that specifically binds ANG-2 is an anti-ANG-2 single chain antibody.
在一些實施方案中,前述的雙特異性抗原結合分子,其中該特異性結合ANG-2的第一抗原結合域為抗ANG-2單鏈抗體,其包含: In some embodiments, the aforementioned bispecific antigen-binding molecule, wherein the first antigen-binding domain that specifically binds ANG-2 is an anti-ANG-2 single chain antibody, comprising:
a)SEQ ID NO:20、21或33的重鏈可變區和SEQ ID NO:17的輕鏈可變區; a) the heavy chain variable region of SEQ ID NO: 20, 21 or 33 and the light chain variable region of SEQ ID NO: 17;
b)SEQ ID NO:44或45的重鏈可變區和SEQ ID NO:40的輕鏈可變區; b) the heavy chain variable region of SEQ ID NO: 44 or 45 and the light chain variable region of SEQ ID NO: 40;
c)SEQ ID NO:72或73的重鏈可變區和SEQ ID NO:74的輕鏈可變區;或 c) the heavy chain variable region of SEQ ID NO: 72 or 73 and the light chain variable region of SEQ ID NO: 74; or
d)SEQ ID NO:75的重鏈可變區和SEQ ID NO:76的輕鏈可變區。 d) The heavy chain variable region of SEQ ID NO:75 and the light chain variable region of SEQ ID NO:76.
在一些實施方案中,前述的雙特異性抗原結合分子,其中該抗ANG-2單鏈抗體包含SEQ ID NO:67、68、69、70、71、77、78或79的胺基酸序列。 In some embodiments, the aforementioned bispecific antigen binding molecule, wherein the anti-ANG-2 single chain antibody comprises the amino acid sequence of SEQ ID NO: 67, 68, 69, 70, 71, 77, 78 or 79.
在一些實施方案中,前述的雙特異性抗原結合分子,其中該特異性結合ANG-2的第一抗原結合域直接,或藉由連接子連接至該特異性結合VEGF的第二抗原結合域。 In some embodiments, the aforementioned bispecific antigen-binding molecule, wherein the first antigen-binding domain that specifically binds ANG-2 is linked directly, or via a linker, to the second antigen-binding domain that specifically binds VEGF.
在一些實施方案中,前述的雙特異性抗原結合分子,其中該特異性結合ANG-2的第一抗原結合域直接,或藉由連接子連接至該特異性結合VEGF的第二抗原結合域的重鏈或輕鏈。 In some embodiments, the aforementioned bispecific antigen-binding molecule, wherein the first antigen-binding domain that specifically binds ANG-2 is directly, or is linked to the second antigen-binding domain that specifically binds VEGF via a linker heavy or light chain.
在一些實施方案中,前述的雙特異性抗原結合分子,其中該抗ANG-2單鏈抗體的N端藉由連接子,連接至該特異性結合VEGF的第二抗原結合域的重鏈C-端。 In some embodiments, the aforementioned bispecific antigen binding molecule, wherein the N-terminus of the anti-ANG-2 single chain antibody is linked to the heavy chain C- of the second antigen binding domain that specifically binds VEGF via a linker end.
在一些實施方案中,前述的雙特異性抗原結合分子,其中該連接子為(GG)n,其中n為1-20中的整數。 In some embodiments, the aforementioned bispecific antigen binding molecule, wherein the linker is (GG) n , wherein n is an integer from 1-20.
在一些實施方案中,前述的雙特異性抗原結合分子,其包含: In some embodiments, the aforementioned bispecific antigen binding molecule, comprising:
i)第一條鏈,包含SEQ ID NO:80、81、82或83的胺基酸序列,或與SEQ ID NO:80、81、82或83分別具有至少95%、96%、97%、98%或99%的序列同一性的胺基酸序列;和 i) a first strand comprising the amino acid sequence of SEQ ID NO: 80, 81, 82 or 83, or having at least 95%, 96%, 97%, amino acid sequences of 98% or 99% sequence identity; and
ii)第二條鏈,其包含SEQ ID NO:57的胺基酸序列,或與SEQ ID NO:57具有至少95%、96%、97%、98%或99%的序列同一性的胺基酸序列。 ii) a second strand comprising the amino acid sequence of SEQ ID NO:57, or an amino group having at least 95%, 96%, 97%, 98% or 99% sequence identity to SEQ ID NO:57 acid sequence.
在一些實施方案中,前述的雙特異性抗原結合分子為具有IgG-scFv形式的4肽結構,其包含兩條相同的第一條鏈和兩條相同的第二條鏈,其中, In some embodiments, the aforementioned bispecific antigen binding molecule is a 4-peptide structure in the form of an IgG-scFv comprising two identical first chains and two identical second chains, wherein,
i)第一條鏈,包含SEQ ID NO:80、81、82或83的胺基酸序列,或與SEQ ID NO:80、81、82或83分別具有至少95%、96%、97%、98%或99%的序列同一性的胺基酸序列;和 i) a first strand comprising the amino acid sequence of SEQ ID NO: 80, 81, 82 or 83, or having at least 95%, 96%, 97%, amino acid sequences of 98% or 99% sequence identity; and
ii)第二條鏈,其包含SEQ ID NO:57的胺基酸序列,或與SEQ ID NO:57具有至少95%、96%、97%、98%或99%的序列同一性的胺基酸序列。 ii) a second strand comprising the amino acid sequence of SEQ ID NO:57, or an amino group having at least 95%, 96%, 97%, 98% or 99% sequence identity to SEQ ID NO:57 acid sequence.
在一些實施方案中,前述的雙特異性抗原結合分子包含: In some embodiments, the aforementioned bispecific antigen binding molecule comprises:
i)第一條鏈,其胺基酸序列如SEQ ID NO:80、81、82或83所示,或與SEQ ID NO:80、81、82或83分別具有至少95%、96%、97%、98%或99%的序列同一性;和 i) the first strand, the amino acid sequence of which is as shown in SEQ ID NO: 80, 81, 82 or 83, or at least 95%, 96%, 97% of the same as SEQ ID NO: 80, 81, 82 or 83, respectively %, 98% or 99% sequence identity; and
ii)第二條鏈,其胺基酸序列如SEQ ID NO:57所示,或與SEQ ID NO:57具有至少95%、96%、97%、98%或99%的序列同一性。 ii) a second strand whose amino acid sequence is set forth in SEQ ID NO:57, or has at least 95%, 96%, 97%, 98% or 99% sequence identity to SEQ ID NO:57.
在一些實施方案中,前述的雙特異性抗原結合分子為具有IgG-scFv形式的4肽結構,其包含兩條相同的第一條鏈和兩條相同的第二條鏈,其中, In some embodiments, the aforementioned bispecific antigen binding molecule is a 4-peptide structure in the form of an IgG-scFv comprising two identical first chains and two identical second chains, wherein,
i)第一條鏈的胺基酸序列如SEQ ID NO:80、81、82或83所示,或與SEQ ID NO:80、81、82或83分別具有至少95%、96%、97%、98%或99%的序列同一性;和 i) The amino acid sequence of the first strand is as shown in SEQ ID NO: 80, 81, 82 or 83, or at least 95%, 96%, 97% of SEQ ID NO: 80, 81, 82 or 83, respectively , 98% or 99% sequence identity; and
ii)第二條鏈的胺基酸序列如SEQ ID NO:57所示,或與SEQ ID NO:57具有至少95%、96%、97%、98%或99%的序列同一性。 ii) The amino acid sequence of the second strand is as set forth in SEQ ID NO:57, or has at least 95%, 96%, 97%, 98% or 99% sequence identity to SEQ ID NO:57.
在一些實施方案中,前述的雙特異性抗原結合分子為具有IgG-scFv形式的4肽結構,其包含兩條相同的第一條鏈和兩條相同的第二條鏈,其中, In some embodiments, the aforementioned bispecific antigen binding molecule is a 4-peptide structure in the form of an IgG-scFv comprising two identical first chains and two identical second chains, wherein,
i)第一條鏈包含SEQ ID NO:80的胺基酸序列;和第二條鏈,其包含SEQ ID NO:57的胺基酸序列; i) a first strand comprising the amino acid sequence of SEQ ID NO:80; and a second strand comprising the amino acid sequence of SEQ ID NO:57;
ii)第一條鏈,包含SEQ ID NO:81的胺基酸序列;和第二條鏈,其包含SEQ ID NO:57的胺基酸序列; ii) a first strand comprising the amino acid sequence of SEQ ID NO:81; and a second strand comprising the amino acid sequence of SEQ ID NO:57;
iii)第一條鏈,包含SEQ ID NO:82的胺基酸序列;和第二條鏈,其包含SEQ ID NO:57的胺基酸序列;或 iii) a first strand comprising the amino acid sequence of SEQ ID NO:82; and a second strand comprising the amino acid sequence of SEQ ID NO:57; or
iv)第一條鏈,包含SEQ ID NO:83的胺基酸序列;和第二條鏈,其包含SEQ ID NO:57的胺基酸序列。 iv) a first strand comprising the amino acid sequence of SEQ ID NO:83; and a second strand comprising the amino acid sequence of SEQ ID NO:57.
在一些實施方案中,前述的雙特異性抗原結合分子為具有IgG-scFv形式的4肽結構,其包含兩條相同的第一條鏈和兩條相同的第二條鏈,其中, In some embodiments, the aforementioned bispecific antigen binding molecule is a 4-peptide structure in the form of an IgG-scFv comprising two identical first chains and two identical second chains, wherein,
i)第一條鏈的胺基酸序列如SEQ ID NO:80該;和第二條鏈的胺基酸序列如SEQ ID NO:57所示; i) the amino acid sequence of the first chain is shown in SEQ ID NO: 80; and the amino acid sequence of the second chain is shown in SEQ ID NO: 57;
ii)第一條鏈的胺基酸序列如SEQ ID NO:81所示;和第二條鏈的胺基酸序列如SEQ ID NO:57所示; ii) the amino acid sequence of the first chain is shown in SEQ ID NO: 81; and the amino acid sequence of the second chain is shown in SEQ ID NO: 57;
iii)第一條鏈的胺基酸序列如SEQ ID NO:82所示;和第二條鏈的胺基酸序列如SEQ ID NO:57所示;或 iii) the amino acid sequence of the first chain is shown in SEQ ID NO: 82; and the amino acid sequence of the second chain is shown in SEQ ID NO: 57; or
iv)第一條鏈的胺基酸序列如SEQ ID NO:83所示;和第二條鏈的胺基酸序列如SEQ ID NO:57所示。 iv) The amino acid sequence of the first chain is shown in SEQ ID NO:83; and the amino acid sequence of the second chain is shown in SEQ ID NO:57.
在一些實施方案中,前述的雙特異性抗原結合分子具有Crossmab形式的4肽結構。 In some embodiments, the aforementioned bispecific antigen binding molecules have a 4-peptide structure in the form of a Crossmab.
在一些實施方案中,該Crossmab形式的雙特異性抗原結合分子包含: In some embodiments, the crossmab format bispecific antigen binding molecule comprises:
a)特異性結合ANG-2的第一全長抗體的第一輕鏈和第一重鏈; a) the first light chain and the first heavy chain of the first full-length antibody that specifically binds ANG-2;
b)特異性結合VEGF的第二全長抗體的第二輕鏈和第二重鏈;其中恆定結構域CL和CH1相互替換。 b) The second light chain and second heavy chain of a second full-length antibody that specifically binds VEGF; wherein the constant domains CL and CH1 are substituted for each other.
在一些實施方案中,前述的雙特異性抗原結合分子包含: In some embodiments, the aforementioned bispecific antigen binding molecule comprises:
a)特異性結合ANG-2的第一全長抗體的第一輕鏈和第一重鏈;和 a) a first light chain and a first heavy chain of a first full-length antibody that specifically binds ANG-2; and
b)特異性結合VEGF的第二全長抗體的第二輕鏈和第二重鏈;其中, b) a second light chain and a second heavy chain of a second full-length antibody that specifically binds VEGF; wherein,
該第一輕鏈的恆定結構域CL與第一重鏈的恆定結構域CH1相互替換; The constant domain CL of the first light chain is replaced with the constant domain CH1 of the first heavy chain;
該第二輕鏈的恆定結構域CL與第二重鏈的恆定結構域CH1相互替換; The constant domain CL of the second light chain is replaced with the constant domain CH1 of the second heavy chain;
該第一輕鏈的輕鏈可變區VL與第一重鏈的重鏈可變區VH相互替換;或 The light chain variable region VL of the first light chain is replaced with the heavy chain variable region VH of the first heavy chain; or
該第二輕鏈的輕鏈可變區VL與第二重鏈的重鏈可變區VH相互替換。 The light chain variable region VL of the second light chain is replaced with the heavy chain variable region VH of the second heavy chain.
在一些實施方案中,前述的雙特異性抗原結合分子包含: In some embodiments, the aforementioned bispecific antigen binding molecule comprises:
a)特異性結合ANG-2的第一全長抗體的第一輕鏈和第一重鏈;和 a) a first light chain and a first heavy chain of a first full-length antibody that specifically binds ANG-2; and
b)特異性結合VEGF的第二全長抗體的第二輕鏈和第二重鏈;並且, b) a second light chain and a second heavy chain of a second full-length antibody that specifically binds VEGF; and,
其中該第一輕鏈的恆定結構域CL與第一重鏈的恆定結構域CH1相互替換。 Wherein the constant structural domain CL of the first light chain is replaced with the constant structural domain CH1 of the first heavy chain.
在一些實施方案中,前述的雙特異性抗原結合分子包含: In some embodiments, the aforementioned bispecific antigen binding molecule comprises:
a)特異性結合ANG-2的第一輕鏈和第一重鏈; a) specifically binds the first light chain and the first heavy chain of ANG-2;
b)特異性結合VEGF的第二輕鏈和第二重鏈;其中恆定結構域CL和CH1相互替換。 b) A second light chain and a second heavy chain that specifically binds VEGF; wherein the constant domains CL and CH1 are substituted for each other.
在一些實施方案中,前述的雙特異性抗原結合分子包含: In some embodiments, the aforementioned bispecific antigen binding molecule comprises:
a)特異性結合ANG-2的第一輕鏈和第一重鏈;和 a) specifically binds the first light chain and the first heavy chain of ANG-2; and
b)特異性結合VEGF的第二輕鏈和第二重鏈;其中, b) a second light chain and a second heavy chain that specifically binds VEGF; wherein,
該第一輕鏈的恆定結構域CL與第一重鏈的恆定結構域CH1相互替換; The constant domain CL of the first light chain is replaced with the constant domain CH1 of the first heavy chain;
該第二輕鏈的恆定結構域CL與第二重鏈的恆定結構域CH1相互替換; The constant domain CL of the second light chain is replaced with the constant domain CH1 of the second heavy chain;
該第一輕鏈的輕鏈可變區VL與第一重鏈的重鏈可變區VH相互替換;或 The light chain variable region VL of the first light chain is replaced with the heavy chain variable region VH of the first heavy chain; or
該第二輕鏈的輕鏈可變區VL與第二重鏈的重鏈可變區VH相互替換。 The light chain variable region VL of the second light chain is replaced with the heavy chain variable region VH of the second heavy chain.
在一些實施方案中,前述的雙特異性抗原結合分子包含: In some embodiments, the aforementioned bispecific antigen binding molecule comprises:
a)特異性結合ANG-2的第一輕鏈和第一重鏈;和 a) specifically binds the first light chain and the first heavy chain of ANG-2; and
b)特異性結合VEGF的第二輕鏈和第二重鏈;並且, b) a second light chain and a second heavy chain that specifically binds VEGF; and,
其中該第一輕鏈的恆定結構域CL與第一重鏈的恆定結構域CH1相互替換。 Wherein the constant structural domain CL of the first light chain is replaced with the constant structural domain CH1 of the first heavy chain.
在一些實施方案中,在前述的雙特異性抗原結合分子中: In some embodiments, in the aforementioned bispecific antigen binding molecules:
a)該第一輕鏈包含SEQ ID NO:17的輕鏈可變區,和該第一重鏈包含SEQ ID NO:33的重鏈可變區;或 a) the first light chain comprises the light chain variable region of SEQ ID NO:17, and the first heavy chain comprises the heavy chain variable region of SEQ ID NO:33; or
該第一輕鏈包含SEQ ID NO:40的輕鏈可變區,和該第一重鏈包含SEQ ID NO:45的重鏈可變區; The first light chain comprises the light chain variable region of SEQ ID NO:40, and the first heavy chain comprises the heavy chain variable region of SEQ ID NO:45;
和 and
b)該第二輕鏈包含SEQ ID NO:56的輕鏈可變區,和該第二重鏈包含SEQ ID NO:58的重鏈可變區。 b) the second light chain comprises the light chain variable region of SEQ ID NO:56, and the second heavy chain comprises the heavy chain variable region of SEQ ID NO:58.
在一些實施方案中,前述的雙特異性抗原結合分子,其中, In some embodiments, the aforementioned bispecific antigen binding molecule, wherein,
a)該第一輕鏈包含SEQ ID NO:85的胺基酸序列,或與SEQ ID NO:85具有至少95%、96%、97%、98%或99%的序列同一性的胺基酸序列,和 a) the first light chain comprises the amino acid sequence of SEQ ID NO:85, or an amino acid having at least 95%, 96%, 97%, 98% or 99% sequence identity to SEQ ID NO:85 sequence, and
該第一重鏈包含SEQ ID NO:84胺基酸序列,或與SEQ ID NO:84具有至少95%、96%、97%、98%或99%的序列同一性的胺基酸序列;或 the first heavy chain comprises the amino acid sequence of SEQ ID NO:84, or an amino acid sequence having at least 95%, 96%, 97%, 98% or 99% sequence identity to SEQ ID NO:84; or
該第一輕鏈包含SEQ ID NO:88的胺基酸序列,或與SEQ ID NO:88具有至少95%、96%、97%、98%或99%的序列同一性的胺基酸序列,和 the first light chain comprises the amino acid sequence of SEQ ID NO:88, or an amino acid sequence having at least 95%, 96%, 97%, 98% or 99% sequence identity to SEQ ID NO:88, and
該第一重鏈包含SEQ ID NO:87胺基酸序列,或與SEQ ID NO:87具有至少95%、96%、97%、98%或99%的序列同一性的胺基酸序列;和 the first heavy chain comprises the amino acid sequence of SEQ ID NO:87, or an amino acid sequence having at least 95%, 96%, 97%, 98% or 99% sequence identity to SEQ ID NO:87; and
b)該第一輕鏈包含SEQ ID NO:57的胺基酸序列,或與SEQ ID NO:57具有至少95%、96%、97%、98%或99%的序列同一性的胺基酸序列;和 b) the first light chain comprises the amino acid sequence of SEQ ID NO:57, or an amino acid having at least 95%, 96%, 97%, 98% or 99% sequence identity to SEQ ID NO:57 sequence; and
該第一重鏈包含SEQ ID NO:86的胺基酸序列,或與SEQ ID NO:86具有至少95%、96%、97%、98%或99%的序列同一性的胺基酸序列。在一些實施方案中,在前述的雙特異性抗原結合分子中: The first heavy chain comprises the amino acid sequence of SEQ ID NO:86, or an amino acid sequence having at least 95%, 96%, 97%, 98% or 99% sequence identity to SEQ ID NO:86. In some embodiments, in the aforementioned bispecific antigen binding molecules:
a)該第一輕鏈的胺基酸序列如SEQ ID NO:85所示,或與SEQ ID NO:85具有至少95%、96%、97%、98%或99%的序列同一性,和 a) the amino acid sequence of the first light chain is as set forth in SEQ ID NO:85, or has at least 95%, 96%, 97%, 98% or 99% sequence identity to SEQ ID NO:85, and
該第一重鏈的胺基酸序列如SEQ ID NO:84所示,或與SEQ ID NO:84具有至少95%、96%、97%、98%或99%的序列同一性;或 The amino acid sequence of the first heavy chain is set forth in SEQ ID NO: 84, or has at least 95%, 96%, 97%, 98% or 99% sequence identity to SEQ ID NO: 84; or
該第一輕鏈的胺基酸序列如SEQ ID NO:88所示,或與SEQ ID NO:88具有至少95%、96%、97%、98%或99%的序列同一性,和 The amino acid sequence of the first light chain is set forth in SEQ ID NO:88, or has at least 95%, 96%, 97%, 98% or 99% sequence identity to SEQ ID NO:88, and
該第一重鏈的胺基酸序列如SEQ ID NO:87所示,或與SEQ ID NO:87具有至少95%、96%、97%、98%或99%的序列同一性;和 The amino acid sequence of the first heavy chain is set forth in SEQ ID NO:87, or has at least 95%, 96%, 97%, 98% or 99% sequence identity to SEQ ID NO:87; and
b)該第二輕鏈的胺基酸序列如SEQ ID NO:57所示,或與SEQ ID NO:57具有至少95%、96%、97%、98%或99%的序列同一性;和 b) the amino acid sequence of the second light chain is as set forth in SEQ ID NO:57, or has at least 95%, 96%, 97%, 98% or 99% sequence identity to SEQ ID NO:57; and
該第二重鏈的胺基酸序列如SEQ ID NO:86所示,或與SEQ ID NO:86具有至少95%、96%、97%、98%或99%的序列同一性。 The amino acid sequence of the second heavy chain is set forth in SEQ ID NO:86, or has at least 95%, 96%, 97%, 98% or 99% sequence identity to SEQ ID NO:86.
在一些實施方案中,在前述的雙特異性抗原結合分子中: In some embodiments, in the aforementioned bispecific antigen binding molecules:
i)該第一輕鏈的胺基酸序列如SEQ ID NO:85所示,和該第一重鏈的胺基酸序列如SEQ ID NO:84所示;和 i) the amino acid sequence of the first light chain is shown in SEQ ID NO: 85, and the amino acid sequence of the first heavy chain is shown in SEQ ID NO: 84; and
該第二輕鏈的胺基酸序列如SEQ ID NO:57所示,和該第二重鏈的胺基酸序列如SEQ ID NO:86所示;或 The amino acid sequence of the second light chain is shown in SEQ ID NO: 57, and the amino acid sequence of the second heavy chain is shown in SEQ ID NO: 86; or
ii)該第一輕鏈的胺基酸序列如SEQ ID NO:88所示,和該第一重鏈的胺基酸序列如SEQ ID NO:87所示;和 ii) the amino acid sequence of the first light chain is shown in SEQ ID NO: 88, and the amino acid sequence of the first heavy chain is shown in SEQ ID NO: 87; and
該第二輕鏈的胺基酸序列如SEQ ID NO:57所示,和該第二重鏈的胺基酸序列如SEQ ID NO:86所示。 The amino acid sequence of the second light chain is shown in SEQ ID NO:57, and the amino acid sequence of the second heavy chain is shown in SEQ ID NO:86.
在一些實施方案中,本揭露提供一種特異性結合ANG-2的抗體。 In some embodiments, the present disclosure provides an antibody that specifically binds ANG-2.
在一些實施方案中,前述的特異性結合ANG-2的抗體,其包含: In some embodiments, the aforementioned antibody that specifically binds to ANG-2 comprises:
i)重鏈可變區,包含分別如SEQ ID NO:7,SEQ ID NO:38和SEQ ID NO:39所示的HCDR1,HCDR2和HCDR3,和 i) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO:7, SEQ ID NO:38 and SEQ ID NO:39, respectively, and
輕鏈可變區,包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3;其中, A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as shown in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12 respectively; wherein,
SEQ ID NO:38的序列為:TINX1X2SSYTYYPDNVKG; The sequence of SEQ ID NO: 38 is: TINX 1 X 2 SSYTYYPDNVKG;
SEQ ID NO:39的序列為:X3X4ATGX5FDY The sequence of SEQ ID NO: 39 is: X 3 X 4 ATGX 5 FDY
其中,X1為D或E,X2為D或N,X3為D或N,X4為E或Q,X5為C、S或V; Wherein, X 1 is D or E, X 2 is D or N, X 3 is D or N, X 4 is E or Q, and X 5 is C, S or V;
或 or
ii)重鏈可變區,包含分別如SEQ ID NO:7,SEQ ID NO:13和SEQ ID NO:14所示的HCDR1,HCDR2和HCDR3,和 ii) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO:7, SEQ ID NO:13 and SEQ ID NO:14, respectively, and
輕鏈可變區,包含分別如SEQ ID NO:15,SEQ ID NO:11和SEQ ID NO:16所示的LCDR1,LCDR2和LCDR3。 The light chain variable region comprises LCDR1, LCDR2 and LCDR3 as shown in SEQ ID NO: 15, SEQ ID NO: 11 and SEQ ID NO: 16, respectively.
在一些實施方案中,前述的特異性結合ANG-2的抗體,其包含: In some embodiments, the aforementioned antibody that specifically binds to ANG-2 comprises:
重鏈可變區,包含如SEQ ID NO:7所示的HCDR1,如SEQ ID NO:8、22或24所示的HCDR2,和如SEQ ID NO:9、23、25、26或27所示的HCDR3;和 A heavy chain variable region comprising HCDR1 as set forth in SEQ ID NO:7, HCDR2 as set forth in SEQ ID NO:8, 22 or 24, and HCDR2 as set forth in SEQ ID NO:9, 23, 25, 26 or 27 HCDR3; and
輕鏈可變區,包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3。 The light chain variable region comprises LCDR1, LCDR2 and LCDR3 as shown in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12, respectively.
在一些實施方案中,前述的特異性結合ANG-2的抗體,其包含: In some embodiments, the aforementioned antibody that specifically binds to ANG-2 comprises:
a)重鏈可變區,其包含分別如SEQ ID NO:7,SEQ ID NO:22和SEQ ID NO:23所示的HCDR1,HCDR2和HCDR3;和 a) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO:7, SEQ ID NO:22 and SEQ ID NO:23, respectively; and
輕鏈可變區,其包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3; A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as shown in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12, respectively;
b)重鏈可變區,其包含分別如SEQ ID NO:7,SEQ ID NO:24和SEQ ID NO:23所示的HCDR1,HCDR2和HCDR3;和 b) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO:7, SEQ ID NO:24 and SEQ ID NO:23, respectively; and
輕鏈可變區,其包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3; A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as shown in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12, respectively;
c)重鏈可變區,其包含分別如SEQ ID NO:7,SEQ ID NO:8和SEQ ID NO:25所示的HCDR1,HCDR2和HCDR3;和 c) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO:7, SEQ ID NO:8 and SEQ ID NO:25, respectively; and
輕鏈可變區,其包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3; A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as shown in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12, respectively;
d)重鏈可變區,其包含分別如SEQ ID NO:7,SEQ ID NO:8和SEQ ID NO:26所示的HCDR1,HCDR2和HCDR3;和 d) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO:7, SEQ ID NO:8 and SEQ ID NO:26, respectively; and
輕鏈可變區,其包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3; A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as shown in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12, respectively;
e)重鏈可變區,其包含分別如SEQ ID NO:7,SEQ ID NO:22和SEQ ID NO:25所示的HCDR1,HCDR2和HCDR3;和 e) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO:7, SEQ ID NO:22 and SEQ ID NO:25, respectively; and
輕鏈可變區,其包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3; A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as shown in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12, respectively;
f)重鏈可變區,其包含分別如SEQ ID NO:7,SEQ ID NO:22和SEQ ID NO:26所示的HCDR1,HCDR2和HCDR3;和 f) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO:7, SEQ ID NO:22 and SEQ ID NO:26, respectively; and
輕鏈可變區,其包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3; A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as shown in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12, respectively;
g)重鏈可變區,其包含分別如SEQ ID NO:7,SEQ ID NO:24和SEQ ID NO:25所示的HCDR1,HCDR2和HCDR3;和 g) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO:7, SEQ ID NO:24 and SEQ ID NO:25, respectively; and
輕鏈可變區,其包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3; A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as shown in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12, respectively;
h)重鏈可變區,包含分別如SEQ ID NO:7,SEQ ID NO:24和SEQ ID NO:26所示的HCDR1,HCDR2和HCDR3;和 h) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO:7, SEQ ID NO:24 and SEQ ID NO:26, respectively; and
輕鏈可變區,其包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3; A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as shown in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12, respectively;
j)重鏈可變區,其包含分別如SEQ ID NO:7,SEQ ID NO:8和SEQ ID NO:23所示的HCDR1,HCDR2和HCDR3;和 j) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO: 7, SEQ ID NO: 8 and SEQ ID NO: 23, respectively; and
輕鏈可變區,其包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3;或 A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as shown in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12, respectively; or
k)重鏈可變區,其包含分別如SEQ ID NO:7,SEQ ID NO:22和SEQ ID NO:27所示的HCDR1,HCDR2和HCDR3;和 k) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO:7, SEQ ID NO:22 and SEQ ID NO:27, respectively; and
輕鏈可變區,其包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3;或 A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as shown in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12, respectively; or
l)重鏈可變區,其包含分別如SEQ ID NO:7,SEQ ID NO:8和SEQ ID NO:9所示的HCDR1,HCDR2和HCDR3;和 1) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as set forth in SEQ ID NO: 7, SEQ ID NO: 8 and SEQ ID NO: 9, respectively; and
輕鏈可變區,其包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3。 A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as set forth in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12, respectively.
在一些實施方式中,前述的特異性結合ANG-2的抗體以等於或小於10-7M解離平衡常數與人ANG-2結合,在一些實施方式中,以等於或小於10-8M或10-9M解離平衡常數與人ANG-2結合。 In some embodiments, the aforementioned antibodies that specifically bind to ANG-2 bind to human ANG-2 with a dissociation equilibrium constant equal to or less than 10-7 M, and in some embodiments, with a dissociation equilibrium constant equal to or less than 10-8 M or 10 -9 M dissociation equilibrium constant for human ANG-2 binding.
在一些實施方案中,前述的特異性結合ANG-2的抗體與猴的ANG-2可交叉結合。 In some embodiments, the aforementioned antibodies that specifically bind ANG-2 are cross-binding to monkey ANG-2.
在一些實施方案中,前述的特異性結合ANG-2的抗體以小於24.82nM、小於20nM、小於15nM、小於10nM或小於5nM的IC50阻斷ANG-2與ANG-2受體Tie2結合,其中該阻斷活性藉由測試例2所述的ELISA實驗檢測。 In some embodiments, the aforementioned antibody that specifically binds ANG-2 blocks the binding of ANG-2 to the ANG-2 receptor Tie2 with an IC50 of less than 24.82 nM, less than 20 nM, less than 15 nM, less than 10 nM, or less than 5 nM, wherein the The blocking activity was detected by the ELISA assay described in Test Example 2.
在一些實施方案中,前述的特異性結合ANG-2的抗體以小於21.27nM、小於15nM、小於10nM或小於8nM的IC50抑制經Tie2轉染的CHO細胞中ANG-2誘導的Tie2磷酸化。 In some embodiments, the aforementioned antibodies that specifically bind ANG-2 inhibit ANG-2-induced Tie2 phosphorylation in Tie2-transfected CHO cells with an IC50 of less than 21.27 nM, less than 15 nM, less than 10 nM, or less than 8 nM.
在一些實施方案中,前述的特異性結合ANG-2的抗體,其為鼠源抗體,嵌合抗體,人源化抗體或全人抗體。 In some embodiments, the aforementioned antibody that specifically binds to ANG-2 is a murine antibody, a chimeric antibody, a humanized antibody or a fully human antibody.
在一些實施方案中,前述的特異性結合ANG-2的抗體,其中該抗體包含框架區,其中, In some embodiments, the aforementioned antibody that specifically binds ANG-2, wherein the antibody comprises a framework region, wherein,
i)該重鏈框架區包含選自44R、77S或84S中的一個或更多個胺基酸回復突變;和/或 i) the heavy chain framework region comprises one or more amino acid backmutations selected from 44R, 77S or 84S; and/or
該輕鏈框架區包含選自1N、43S、68A、85D、87H中的一個或更多個胺基酸回復突變;或 The light chain framework region comprises one or more amino acid backmutations selected from IN, 43S, 68A, 85D, 87H; or
ii)該重鏈框架區包含選自2L、44R、74V、82AS或83K中的一個或更多個胺基酸回復突變;和/或 ii) the heavy chain framework region comprises one or more amino acid backmutations selected from 2L, 44R, 74V, 82AS or 83K; and/or
該輕鏈框架區包含選自1N、43S、46V、68A、85D、87H中的一個或更多個胺基酸回復突變。 The light chain framework region comprises one or more amino acid backmutations selected from IN, 43S, 46V, 68A, 85D, 87H.
在一些實施方案中,前述的特異性結合ANG-2的抗體,其包含: In some embodiments, the aforementioned antibody that specifically binds to ANG-2 comprises:
i)重鏈可變區,其包含分別如SEQ ID NO:7,SEQ ID NO:38和SEQ ID NO:39所示的HCDR1,HCDR2和HCDR3,且其重鏈框架區包含選自44R、77S或84S中的一個或更多個胺基酸回復突變;和 i) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as shown in SEQ ID NO: 7, SEQ ID NO: 38 and SEQ ID NO: 39, respectively, and whose heavy chain framework region comprises 44R, 77S or one or more amino acid backmutations in 84S; and
輕鏈可變區,其包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3;且其輕鏈框架區包含選自1N、43S、68A、85D、87H中的一個或更多個胺基酸回復突變;其中, A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as shown in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12 respectively; and its light chain framework region comprises a light chain framework region selected from the group consisting of 1N, 43S, 68A One or more amino acid backmutations in , 85D, 87H; wherein,
SEQ ID NO:38的序列為:TINX1X2SSYTYYPDNVKG; The sequence of SEQ ID NO: 38 is: TINX 1 X 2 SSYTYYPDNVKG;
SEQ ID NO:39的序列為:X3X4ATGX5FDY The sequence of SEQ ID NO: 39 is: X 3 X 4 ATGX 5 FDY
其中,X1為D或E,X2為D或N,X3為D或N,X4為E或Q,X5為C、S或V;或 wherein X 1 is D or E, X 2 is D or N, X 3 is D or N, X 4 is E or Q, and X 5 is C, S or V; or
ii)重鏈可變區,其包含分別如SEQ ID NO:7,SEQ ID NO:13和SEQ ID NO:14所示的HCDR1,HCDR2和HCDR3,且其重鏈框架區包含選自2L、44R、74V、82AS或83K中的一個或更多個胺基酸回復突變;和 ii) a heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as shown in SEQ ID NO: 7, SEQ ID NO: 13 and SEQ ID NO: 14, respectively, and whose heavy chain framework region comprises a heavy chain framework region selected from the group consisting of 2L, 44R one or more amino acid backmutations in , 74V, 82AS or 83K; and
輕鏈可變區,其包含分別如SEQ ID NO:15,SEQ ID NO:11和SEQ ID NO:16所示的LCDR1,LCDR2和LCDR3,且其輕鏈框架區包含選自1N、43S、46V、68A、85D、87H中的一個或更多個胺基酸回復突變。 A light chain variable region comprising LCDR1, LCDR2 and LCDR3 as shown in SEQ ID NO: 15, SEQ ID NO: 11 and SEQ ID NO: 16, respectively, and the light chain framework region thereof comprises a light chain framework region selected from the group consisting of 1N, 43S, 46V One or more amino acid backmutations in , 68A, 85D, 87H.
在一些實施方案中,前述的特異性結合ANG-2的抗體,其包含: In some embodiments, the aforementioned antibody that specifically binds to ANG-2 comprises:
重鏈可變區,其包含分別如SEQ ID NO:7,SEQ ID NO:22和SEQ ID NO:26所示的HCDR1,HCDR2和HCDR3,且其重鏈框架區包含選自44R、77S或84S中的一個或更多個胺基酸回復突變;和 A heavy chain variable region comprising HCDR1, HCDR2 and HCDR3 as shown in SEQ ID NO: 7, SEQ ID NO: 22 and SEQ ID NO: 26, respectively, and whose heavy chain framework region comprises 44R, 77S or 84S one or more amino acid backmutations in; and
輕鏈可變區,其包含分別如SEQ ID NO:10,SEQ ID NO:11和SEQ ID NO:12所示的LCDR1,LCDR2和LCDR3,且輕鏈框架區包含選自1N、43S、68A、85D、87H中的一個或更多個胺基酸回復突變。 A light chain variable region comprising LCDR1, LCDR2 and LCDR3 shown in SEQ ID NO: 10, SEQ ID NO: 11 and SEQ ID NO: 12, respectively, and the light chain framework region comprising a light chain framework region selected from the group consisting of 1N, 43S, 68A, One or more amino acid backmutations in 85D, 87H.
在一些實施方案中,前述的特異性結合ANG-2的抗體,其包含: In some embodiments, the aforementioned antibody that specifically binds to ANG-2 comprises:
重鏈可變區,包含SEQ ID NO:20的胺基酸序列,或在SEQ ID NO:20的胺基酸序列中包含選自44R、77S或84S中的一個或更多個胺基酸回復突變的胺基酸序列;和 A heavy chain variable region comprising the amino acid sequence of SEQ ID NO: 20, or comprising one or more amino acid reversions selected from 44R, 77S or 84S in the amino acid sequence of SEQ ID NO: 20 mutated amino acid sequences; and
輕鏈可變區,包含SEQ ID NO:17的胺基酸序列,或在SEQ ID NO:17的胺基酸序列中包含選自1N、43S、68A、85D、87H中的一個或更多個胺基酸回復突變的胺基酸序列。 A light chain variable region comprising the amino acid sequence of SEQ ID NO: 17, or comprising one or more selected from the group consisting of IN, 43S, 68A, 85D, 87H in the amino acid sequence of SEQ ID NO: 17 The amino acid sequence of the amino acid backmutation.
在一些實施方案中,前述的特異性結合ANG-2的抗體,其包含: In some embodiments, the aforementioned antibody that specifically binds to ANG-2 comprises:
重鏈可變區,其胺基酸序列如SEQ ID NO:20所示,或在SEQ ID NO:20的胺基酸序列中包含選自44R、77S或84S中的一個或更多個胺基酸回復突變;和 Heavy chain variable region, the amino acid sequence of which is shown in SEQ ID NO: 20, or comprises one or more amino groups selected from 44R, 77S or 84S in the amino acid sequence of SEQ ID NO: 20 acid backmutation; and
輕鏈可變區,其胺基酸序列如SEQ ID NO:17所示,或在SEQ ID NO:17的胺基酸序列中包含選自1N、43S、68A、85D、87H中的一個或更多個胺基酸回復突變。 The light chain variable region, the amino acid sequence of which is shown in SEQ ID NO: 17, or the amino acid sequence of SEQ ID NO: 17 comprises one or more selected from the group consisting of 1N, 43S, 68A, 85D, 87H Multiple amino acid backmutations.
在一些實施方案中,前述的特異性結合ANG-2的抗體,其包含: In some embodiments, the aforementioned antibody that specifically binds to ANG-2 comprises:
重鏈可變區,包含SEQ ID NO:33的胺基酸序列;和 A heavy chain variable region comprising the amino acid sequence of SEQ ID NO: 33; and
輕鏈可變區,包含SEQ ID NO:17的胺基酸序列,或在SEQ ID NO:17的胺基酸序列中包含選自1N、43S、68A、85D、87H中的一個或更多個胺基酸回復突變的胺基酸序列。 A light chain variable region comprising the amino acid sequence of SEQ ID NO: 17, or comprising one or more selected from the group consisting of IN, 43S, 68A, 85D, 87H in the amino acid sequence of SEQ ID NO: 17 The amino acid sequence of the amino acid backmutation.
在一些實施方案中,前述的特異性結合ANG-2的抗體,其包含: In some embodiments, the aforementioned antibody that specifically binds to ANG-2 comprises:
SEQ ID NO:33的重鏈可變區;和 The heavy chain variable region of SEQ ID NO: 33; and
輕鏈可變區,其胺基酸序列如SEQ ID NO:17所示,或在SEQ ID NO:17的胺基酸序列中包含選自1N、43S、68A、85D、87H中的一個或更多個胺基酸回復突變。 The light chain variable region, the amino acid sequence of which is shown in SEQ ID NO: 17, or the amino acid sequence of SEQ ID NO: 17 comprises one or more selected from the group consisting of 1N, 43S, 68A, 85D, 87H Multiple amino acid backmutations.
在一些實施方案中,前述的特異性結合ANG-2的抗體,其包含: In some embodiments, the aforementioned antibody that specifically binds to ANG-2 comprises:
重鏈可變區,包含SEQ ID NO:44的胺基酸序列,或在SEQ ID NO:44的胺基酸序列中包含選自2L、44R、74V、82AS或83K中的一個或更多個胺基酸回復突變的胺基酸序列;和 A heavy chain variable region comprising the amino acid sequence of SEQ ID NO: 44, or comprising one or more selected from 2L, 44R, 74V, 82AS or 83K in the amino acid sequence of SEQ ID NO: 44 the amino acid sequence of the amino acid backmutation; and
輕鏈可變區,包含SEQ ID NO:40的胺基酸序列,或在SEQ ID NO:40的胺基酸序列中包含選自1N、43S、46V、68A、85D、87H中的一個或更多個胺基酸回復突變的胺基酸序列。 A light chain variable region comprising the amino acid sequence of SEQ ID NO:40, or one or more selected from the group consisting of IN, 43S, 46V, 68A, 85D, 87H in the amino acid sequence of SEQ ID NO:40 Multiple amino acid backmutated amino acid sequences.
在一些實施方案中,前述的特異性結合ANG-2的抗體,其包含: In some embodiments, the aforementioned antibody that specifically binds to ANG-2 comprises:
重鏈可變區,其胺基酸序列如SEQ ID NO:44所示,或在SEQ ID NO:44的胺基酸序列中包含選自2L、44R、74V、82AS或83K中的一個或更多個胺基酸回復突變;和 Heavy chain variable region, the amino acid sequence of which is shown in SEQ ID NO: 44, or the amino acid sequence of SEQ ID NO: 44 comprises one selected from 2L, 44R, 74V, 82AS or 83K or more amino acid backmutations; and
輕鏈可變區,其胺基酸序列如SEQ ID NO:40所示,或在SEQ ID NO:40的胺基酸序列中包含選自1N、43S、46V、68A、85D、87H中的一個或更多個胺基酸回復突變。 The light chain variable region, the amino acid sequence of which is shown in SEQ ID NO: 40, or the amino acid sequence of SEQ ID NO: 40 comprises one selected from the group consisting of 1N, 43S, 46V, 68A, 85D, and 87H or more amino acid backmutations.
在一些實施方案中,前述的特異性結合ANG-2的抗體,其包含: In some embodiments, the aforementioned antibody that specifically binds ANG-2 comprises:
i)重鏈可變區,包含SEQ ID NO:3、20、21、28-37、72或73中任一胺基酸序列,或包含與SEQ ID NO:3、20、21、28-37、72或73中任一序列分別具有至少95%、96%、97%、98%或99%的序列同一性的胺基酸序列;和/或 i) a heavy chain variable region comprising the amino acid sequence of any one of SEQ ID NO: 3, 20, 21, 28-37, 72 or 73, or comprising the same amino acid sequence as SEQ ID NO: 3, 20, 21, 28-37 amino acid sequences having at least 95%, 96%, 97%, 98% or 99% sequence identity, respectively, in any of the sequences , 72 or 73; and/or
輕鏈可變區,包含SEQ ID NO:4、17、18、19或74的胺基酸序列,或包含與SEQ ID NO:4、17、18、19或74分別具有至少95%、96%、97%、98%或99%的序列同一性的胺基酸序列;或 A light chain variable region comprising the amino acid sequence of SEQ ID NO: 4, 17, 18, 19 or 74, or comprising at least 95%, 96% with SEQ ID NO: 4, 17, 18, 19 or 74, respectively , 97%, 98% or 99% sequence identity of amino acid sequences; or
ii)重鏈可變區,包含SEQ ID NO:5、44、45、46或75的胺基酸序列,或包含與SEQ ID NO:5、44、45、46或75分別具有至少95%、96%、97%、98%或99%的序列同一性的胺基酸序列;和/或 ii) a heavy chain variable region comprising the amino acid sequence of SEQ ID NO: 5, 44, 45, 46 or 75, or comprising at least 95%, amino acid sequences of 96%, 97%, 98% or 99% sequence identity; and/or
輕鏈可變區,包含SEQ ID NO:6、40、41、42、43或76的胺基酸序列,或包含與SEQ ID NO:6、40、41、42、43或76分別具有至少95%、96%、97%、98%或99%的序列同一性的胺基酸序列。 A light chain variable region comprising the amino acid sequence of SEQ ID NO: 6, 40, 41, 42, 43 or 76, or comprising at least 95 SEQ ID NO: 6, 40, 41, 42, 43 or 76, respectively %, 96%, 97%, 98% or 99% sequence identity of amino acid sequences.
在一些實施方案中,前述的特異性結合ANG-2的抗體,其包含: In some embodiments, the aforementioned antibody that specifically binds to ANG-2 comprises:
i)重鏈可變區,其胺基酸序列如SEQ ID NO:3、20、21、28-37、72或73中任一所示,或與SEQ ID NO:3、20、21、28-37、72或73分別具有至少95%、96%、97%、98%或99%的序列同一性;和/或 i) a heavy chain variable region, the amino acid sequence of which is shown in any of SEQ ID NO: 3, 20, 21, 28-37, 72 or 73, or the same as SEQ ID NO: 3, 20, 21, 28 - 37, 72 or 73 have at least 95%, 96%, 97%, 98% or 99% sequence identity, respectively; and/or
輕鏈可變區,其胺基酸序列如SEQ ID NO:4、17、18、19或74所示,或與SEQ ID NO:4、17、18、19或74分別具有至少95%、96%、97%、98%或99%的序列同一性;或 A light chain variable region, the amino acid sequence of which is shown in SEQ ID NO: 4, 17, 18, 19 or 74, or has at least 95%, 96% and 96% respectively with SEQ ID NO: 4, 17, 18, 19 or 74 %, 97%, 98% or 99% sequence identity; or
ii)重鏈可變區,其胺基酸序列如SEQ ID NO:5、44、45、46或75所示,或與SEQ ID NO:5、44、45、46或75分別具有至少95%、96%、97%、98%或99%的序列同一性;和/或 ii) a heavy chain variable region, the amino acid sequence of which is as shown in SEQ ID NO: 5, 44, 45, 46 or 75, or at least 95% of the same as SEQ ID NO: 5, 44, 45, 46 or 75, respectively , 96%, 97%, 98% or 99% sequence identity; and/or
輕鏈可變區,其胺基酸序列如SEQ ID NO:6、40、41、42、43或76所示,或與SEQ ID NO:6、40、41、42、43或76分別具有至少95%、96%、97%、98%或99%的序列同一性。 A light chain variable region, the amino acid sequence of which is shown in SEQ ID NO: 6, 40, 41, 42, 43 or 76, or has at least SEQ ID NO: 6, 40, 41, 42, 43 or 76, respectively 95%, 96%, 97%, 98% or 99% sequence identity.
在一些實施方案中,前述的特異性結合ANG-2的抗體,其包含: In some embodiments, the aforementioned antibody that specifically binds to ANG-2 comprises:
a)重鏈可變區,包含SEQ ID NO:3的胺基酸序列;和輕鏈可變區,包含SEQ ID NO:4的胺基酸序列; a) a heavy chain variable region comprising the amino acid sequence of SEQ ID NO:3; and a light chain variable region comprising the amino acid sequence of SEQ ID NO:4;
b)重鏈可變區,包含SEQ ID NO:20或21的胺基酸序列;和輕鏈可變區,包含SEQ ID NO:17、18或19的胺基酸序列; b) a heavy chain variable region comprising the amino acid sequence of SEQ ID NO: 20 or 21; and a light chain variable region comprising the amino acid sequence of SEQ ID NO: 17, 18 or 19;
c)重鏈可變區,包含SEQ ID NO:28-37中任一所示的胺基酸序列;和輕鏈可變區,包含SEQ ID NO:17的胺基酸序列; c) a heavy chain variable region comprising the amino acid sequence set forth in any of SEQ ID NOs: 28-37; and a light chain variable region comprising the amino acid sequence of SEQ ID NO: 17;
d)重鏈可變區,包含SEQ ID NO:72或73的胺基酸序列;和輕鏈可變區,包含SEQ ID NO:74的胺基酸序列; d) a heavy chain variable region comprising the amino acid sequence of SEQ ID NO: 72 or 73; and a light chain variable region comprising the amino acid sequence of SEQ ID NO: 74;
e)重鏈可變區,包含SEQ ID NO:5的胺基酸序列;和輕鏈可變區,包含SEQ ID NO:6的胺基酸序列; e) a heavy chain variable region comprising the amino acid sequence of SEQ ID NO:5; and a light chain variable region comprising the amino acid sequence of SEQ ID NO:6;
f)重鏈可變區,包含SEQ ID NO:44、45或46的胺基酸序列;和輕鏈可變區,包含SEQ ID NO:40、41、42或43的胺基酸序列;或 f) a heavy chain variable region comprising the amino acid sequence of SEQ ID NO: 44, 45 or 46; and a light chain variable region comprising the amino acid sequence of SEQ ID NO: 40, 41, 42 or 43; or
g)重鏈可變區,包含SEQ ID NO:75的胺基酸序列;和輕鏈可變區,包含SEQ ID NO:76的胺基酸序列。 g) a heavy chain variable region comprising the amino acid sequence of SEQ ID NO:75; and a light chain variable region comprising the amino acid sequence of SEQ ID NO:76.
在一些實施方案中,前述的特異性結合ANG-2的抗體,其包含: In some embodiments, the aforementioned antibody that specifically binds to ANG-2 comprises:
a)SEQ ID NO:3的重鏈可變區;和SEQ ID NO:4的輕鏈可變區; a) the heavy chain variable region of SEQ ID NO:3; and the light chain variable region of SEQ ID NO:4;
b)SEQ ID NO:20或21的重鏈可變區;和SEQ ID NO:17、18或19的輕鏈可變區; b) the heavy chain variable region of SEQ ID NO: 20 or 21; and the light chain variable region of SEQ ID NO: 17, 18 or 19;
c)SEQ ID NO:28-37中任一所示的重鏈可變區;和SEQ ID NO:17的輕鏈可變區; c) the heavy chain variable region set forth in any of SEQ ID NOs: 28-37; and the light chain variable region of SEQ ID NO: 17;
d)SEQ ID NO:72或73的重鏈可變區;和SEQ ID NO:74的輕鏈可變區; d) the heavy chain variable region of SEQ ID NO: 72 or 73; and the light chain variable region of SEQ ID NO: 74;
e)SEQ ID NO:5的重鏈可變區;和SEQ ID NO:6的輕鏈可變區; e) the heavy chain variable region of SEQ ID NO:5; and the light chain variable region of SEQ ID NO:6;
f)SEQ ID NO:44、45或46的重鏈可變區;和SEQ ID NO:40、41、42或43的輕鏈可變區;或 f) the heavy chain variable region of SEQ ID NO: 44, 45 or 46; and the light chain variable region of SEQ ID NO: 40, 41, 42 or 43; or
g)SEQ ID NO:75的重鏈可變區;和SEQ ID NO:76的輕鏈可變區。 g) the heavy chain variable region of SEQ ID NO:75; and the light chain variable region of SEQ ID NO:76.
在一些實施方案中,前述的特異性結合ANG-2的抗體,其包含: In some embodiments, the aforementioned antibody that specifically binds to ANG-2 comprises:
a)SEQ ID NO:20、21或33的重鏈可變區和SEQ ID NO:17的輕鏈可變區; a) the heavy chain variable region of SEQ ID NO: 20, 21 or 33 and the light chain variable region of SEQ ID NO: 17;
b)SEQ ID NO:44或45的重鏈可變區和SEQ ID NO:40的輕鏈可變區; b) the heavy chain variable region of SEQ ID NO: 44 or 45 and the light chain variable region of SEQ ID NO: 40;
c)SEQ ID NO:72或73的重鏈可變區和SEQ ID NO:74的輕鏈可變區;或 c) the heavy chain variable region of SEQ ID NO: 72 or 73 and the light chain variable region of SEQ ID NO: 74; or
d)SEQ ID NO:75的重鏈可變區和SEQ ID NO:76的輕鏈可變區。 d) The heavy chain variable region of SEQ ID NO:75 and the light chain variable region of SEQ ID NO:76.
在一些實施方案中,前述的特異性結合ANG-2的抗體,其包含: In some embodiments, the aforementioned antibody that specifically binds to ANG-2 comprises:
重鏈可變區,包含SEQ ID NO:33的胺基酸序列,和輕鏈可變區,包含SEQ ID NO:17的胺基酸序列;或 A heavy chain variable region, comprising the amino acid sequence of SEQ ID NO: 33, and a light chain variable region, comprising the amino acid sequence of SEQ ID NO: 17; or
重鏈可變區,包含SEQ ID NO:45的胺基酸序列,和輕鏈可變區,包含SEQ ID NO:40的胺基酸序列。 The heavy chain variable region, comprising the amino acid sequence of SEQ ID NO:45, and the light chain variable region, comprising the amino acid sequence of SEQ ID NO:40.
在一些實施方案中,前述的特異性結合ANG-2的抗體,其包含: In some embodiments, the aforementioned antibody that specifically binds to ANG-2 comprises:
SEQ ID NO:33的重鏈可變區,和SEQ ID NO:17的輕鏈可變區;或 The heavy chain variable region of SEQ ID NO:33, and the light chain variable region of SEQ ID NO:17; or
SEQ ID NO:45的重鏈可變區,和SEQ ID NO:40的輕鏈可變區。 The heavy chain variable region of SEQ ID NO:45, and the light chain variable region of SEQ ID NO:40.
在一些實施方案中,前述的特異性結合ANG-2的抗體,其包含恆定區。在另外一些實施方案中,該重鏈恆定區選自人IgG1、IgG2、IgG3和IgG4恆定區及其常規變體,該輕鏈恆定區選自人抗體κ和λ鏈恆定區及其常規變體。 In some embodiments, the aforementioned antibody that specifically binds ANG-2 comprises a constant region. In other embodiments, the heavy chain constant region is selected from the group consisting of human IgGl, IgG2, IgG3 and IgG4 constant regions and conventional variants thereof, and the light chain constant region is selected from the group consisting of human antibody kappa and lambda chain constant regions and conventional variants thereof .
在一些實施方案中,前述的特異性結合ANG-2的抗體,其包含SEQ ID NO:47的重鏈恆定區和SEQ ID NO:48的輕鏈恆定區。 In some embodiments, the aforementioned antibody that specifically binds ANG-2 comprises the heavy chain constant region of SEQ ID NO:47 and the light chain constant region of SEQ ID NO:48.
在一些實施方案中,前述的特異性結合ANG-2的抗體,其包含: In some embodiments, the aforementioned antibody that specifically binds to ANG-2 comprises:
a)重鏈,包含SEQ ID NO:49、51或52的胺基酸序列,或包含與SEQ ID NO:49、51或52分別具有至少95%、96%、97%、98%或99%的序列同一性的胺基酸序列,和/或 a) a heavy chain comprising the amino acid sequence of SEQ ID NO: 49, 51 or 52, or comprising at least 95%, 96%, 97%, 98% or 99% of the amino acid sequence of SEQ ID NO: 49, 51 or 52, respectively the sequence identity of the amino acid sequence, and/or
輕鏈,包含SEQ ID NO:50的胺基酸序列,或包含與SEQ ID NO:50具有至少95%、96%、97%、98%或99%的序列同一性的胺基酸序列;或 A light chain comprising the amino acid sequence of SEQ ID NO:50, or comprising an amino acid sequence having at least 95%, 96%, 97%, 98% or 99% sequence identity to SEQ ID NO:50; or
b)重鏈,包含SEQ ID NO:53或55的胺基酸序列,或包含與SEQ ID NO:53或55分別具有至少95%、96%、97%、98%或99%的序列同一性的胺基酸序列,和/或 b) a heavy chain comprising the amino acid sequence of SEQ ID NO: 53 or 55, or comprising at least 95%, 96%, 97%, 98% or 99% sequence identity to SEQ ID NO: 53 or 55, respectively the amino acid sequence, and/or
輕鏈,包含SEQ ID NO:54的胺基酸序列,或包含與SEQ ID NO:54具有至少95%、96%、97%、98%或99%的序列同一性的胺基酸序列。 A light chain comprising the amino acid sequence of SEQ ID NO:54, or comprising an amino acid sequence having at least 95%, 96%, 97%, 98% or 99% sequence identity to SEQ ID NO:54.
在一些實施方案中,前述的特異性結合ANG-2的抗體,其包含: In some embodiments, the aforementioned antibody that specifically binds to ANG-2 comprises:
a)重鏈,其胺基酸序列如SEQ ID NO:49、51或52所示,或與SEQ ID NO:49、51或52分別具有至少95%、96%、97%、98%或99%的序列同一性,和/或 a) a heavy chain having an amino acid sequence as shown in SEQ ID NO: 49, 51 or 52, or at least 95%, 96%, 97%, 98% or 99% from SEQ ID NO: 49, 51 or 52, respectively % sequence identity, and/or
輕鏈,其胺基酸序列如SEQ ID NO:50所示,或與SEQ ID NO:50具有至少95%、96%、97%、98%或99%的序列同一性;或 A light chain whose amino acid sequence is set forth in SEQ ID NO:50, or has at least 95%, 96%, 97%, 98% or 99% sequence identity to SEQ ID NO:50; or
b)重鏈,其胺基酸序列如SEQ ID NO:53或55所示,或與SEQ ID NO:53或55分別具有至少95%、96%、97%、98%或99%的序列同一性,和/或 b) a heavy chain, the amino acid sequence of which is set forth in SEQ ID NO: 53 or 55, or at least 95%, 96%, 97%, 98% or 99% identical to SEQ ID NO: 53 or 55, respectively sex, and/or
輕鏈,其胺基酸序列如SEQ ID NO:54所示,或與SEQ ID NO:54具有至少95%、96%、97%、98%或99%的序列同一性。 A light chain whose amino acid sequence is set forth in SEQ ID NO:54, or has at least 95%, 96%, 97%, 98% or 99% sequence identity to SEQ ID NO:54.
在一些實施方案中,前述的特異性結合ANG-2的抗體,其包含: In some embodiments, the aforementioned antibody that specifically binds to ANG-2 comprises:
a)重鏈,包含SEQ ID NO:49、51或52的胺基酸序列,和 a) a heavy chain comprising the amino acid sequence of SEQ ID NO: 49, 51 or 52, and
輕鏈,包含SEQ ID NO:50的胺基酸序列;或 A light chain comprising the amino acid sequence of SEQ ID NO: 50; or
b)重鏈,包含SEQ ID NO:53或55的胺基酸序列,和 b) a heavy chain comprising the amino acid sequence of SEQ ID NO: 53 or 55, and
輕鏈,包含SEQ ID NO:54的胺基酸序列。 A light chain comprising the amino acid sequence of SEQ ID NO:54.
在一些實施方案中,前述的特異性結合ANG-2的抗體,其包含: In some embodiments, the aforementioned antibody that specifically binds to ANG-2 comprises:
a)重鏈,其胺基酸序列如SEQ ID NO:49、51或52所示,和 a) a heavy chain, the amino acid sequence of which is shown in SEQ ID NO: 49, 51 or 52, and
輕鏈,其胺基酸序列如SEQ ID NO:50所示;或 A light chain, the amino acid sequence of which is shown in SEQ ID NO: 50; or
b)重鏈,其胺基酸序列如SEQ ID NO:53或55所示,和 b) a heavy chain, the amino acid sequence of which is shown in SEQ ID NO: 53 or 55, and
輕鏈,其胺基酸序列如SEQ ID NO:54所示。 Light chain, the amino acid sequence of which is shown in SEQ ID NO:54.
在一些實施方案中,本揭露提供一種分離的抗體,其與前述的特異性結合ANG-2的抗體競爭結合人ANG-2。 In some embodiments, the present disclosure provides an isolated antibody that competes with the aforementioned antibody that specifically binds ANG-2 for binding to human ANG-2.
在一些實施方案中,本揭露提供一種核酸分子,其編碼根據前述的雙特異性抗原結合分子,或前述的特異性結合ANG-2的抗體。 In some embodiments, the present disclosure provides a nucleic acid molecule encoding a bispecific antigen binding molecule according to the foregoing, or an antibody that specifically binds ANG-2 as described above.
在一些實施方案中,本揭露提供一種載體,其包含前述的核酸分子。 In some embodiments, the present disclosure provides a vector comprising the aforementioned nucleic acid molecule.
在一些實施方案中,本揭露提供一種宿主細胞,其包含前述的載體。 In some embodiments, the present disclosure provides a host cell comprising the aforementioned vector.
在一些實施方案中,本揭露提供一種醫藥組成物,其包含有效量的前述雙特異性抗原結合分子,或前述特異性結合ANG-2的抗體,或前述的核酸分子,以及一種或更多種藥學上可接受的載體、稀釋劑或賦形劑。在一些實施方案中,該治療有效量為單位劑量的組成物中含有0.1-3000mg的如前所述的雙特異性抗原結合分子或特異性結合ANG-2的抗體。 In some embodiments, the present disclosure provides a pharmaceutical composition comprising an effective amount of the aforementioned bispecific antigen-binding molecule, or the aforementioned antibody that specifically binds to ANG-2, or the aforementioned nucleic acid molecule, and one or more A pharmaceutically acceptable carrier, diluent or excipient. In some embodiments, the therapeutically effective amount is 0.1-3000 mg of a bispecific antigen binding molecule or antibody that specifically binds ANG-2 as previously described in a unit dose of the composition.
在一些實施方案中,本揭露提供一種生產前述的雙特異性抗原結合分子,或前述的特異性結合ANG-2的抗體的方法,該方法包括培養前述的宿主細胞以表達該雙特異性抗原結合分子或特異性結合ANG-2的抗體。 In some embodiments, the present disclosure provides a method of producing the aforementioned bispecific antigen binding molecule, or the aforementioned antibody that specifically binds ANG-2, the method comprising culturing the aforementioned host cell to express the bispecific antigen binding molecule Molecules or antibodies that specifically bind ANG-2.
在一些實施方案中,本揭露提供一種預防或治療癌症或血管生成性眼病的方法,該方法包括向受試者施用治療有效量的前述的雙特異性抗原結合分子,或前述的特異性結合ANG-2的抗體,或前述的核酸分子,或前述的醫藥組成物。 In some embodiments, the present disclosure provides a method of preventing or treating cancer or angiogenic eye disease, the method comprising administering to a subject a therapeutically effective amount of the aforementioned bispecific antigen binding molecule, or the aforementioned specifically binding ANG The antibody of -2, or the aforementioned nucleic acid molecule, or the aforementioned pharmaceutical composition.
在一些實施方案中,前述的治療方法,其中該癌症選自乳腺癌、腎上腺腫瘤、輸卵管癌、鱗狀細胞癌、卵巢癌、胃癌、結直腸癌、非小細胞肺癌、膽管癌、膀胱癌、胰腺癌、皮膚癌和肝癌。在一些實施方案中,其中該血管生成性眼病選自新生血管性青光眼、年齡相關的黃斑變性(AMD)、糖尿病性黃斑水腫、角膜新血管生成、角膜移植新血管生成、角膜移植排斥、視網膜/脈絡膜新血管生成、房角的新血管生成(虹膜發紅)、眼部新生血管性疾病、血管再狹窄和動靜脈畸形(AVM)。在一些實施方案中,前述的癌症或血管生成性眼病與VEGF或ANG-2相關。 In some embodiments, the aforementioned method of treatment, wherein the cancer is selected from the group consisting of breast cancer, adrenal tumor, fallopian tube cancer, squamous cell carcinoma, ovarian cancer, gastric cancer, colorectal cancer, non-small cell lung cancer, bile duct cancer, bladder cancer, Pancreatic cancer, skin cancer and liver cancer. In some embodiments, wherein the angiogenic eye disease is selected from the group consisting of neovascular glaucoma, age-related macular degeneration (AMD), diabetic macular edema, corneal neovascularization, corneal transplant neovascularization, corneal transplant rejection, retinal/ Choroidal neovascularization, angle neovascularization (iris redness), ocular neovascular disease, vascular restenosis, and arteriovenous malformations (AVMs). In some embodiments, the aforementioned cancer or angiogenic eye disease is associated with VEGF or ANG-2.
在一些實施方案中,本揭露提供前述的雙特異性抗原結合分子,或前述的特異性結合ANG-2的抗體,或前述的核酸分子,或前述的醫藥組成物在製備用於治療或預防癌症或血管生成性眼病的藥物中的用途。 In some embodiments, the present disclosure provides the aforementioned bispecific antigen-binding molecule, or the aforementioned antibody that specifically binds to ANG-2, or the aforementioned nucleic acid molecule, or the aforementioned pharmaceutical composition in preparation for the treatment or prevention of cancer Or the use in the medicine of angiogenic eye disease.
在一些實施方案中,前述的用途,其中該癌症選自乳腺癌、腎上腺腫瘤、輸卵管癌、鱗狀細胞癌、卵巢癌、胃癌、結直腸癌、非小細胞肺癌、膽管癌、膀胱癌、胰腺癌、皮膚癌和肝癌。在一些實施方案中,其中該血管生成性眼病選自新生血管性青光眼、年齡相關的黃斑變性(AMD)、糖尿病性黃斑水 腫、角膜新血管生成、角膜移植新血管生成、角膜移植排斥、視網膜/脈絡膜新血管生成、房角的新血管生成(虹膜發紅)、眼部新生血管性疾病、血管再狹窄和動靜脈畸形(AVM)。在一些實施方案中,前述的癌症或血管生成性眼病與VEGF或ANG-2相關。 In some embodiments, the aforementioned use, wherein the cancer is selected from the group consisting of breast cancer, adrenal tumor, fallopian tube cancer, squamous cell carcinoma, ovarian cancer, gastric cancer, colorectal cancer, non-small cell lung cancer, bile duct cancer, bladder cancer, pancreas cancer cancer, skin cancer and liver cancer. In some embodiments, wherein the angiogenic eye disease is selected from the group consisting of neovascular glaucoma, age-related macular degeneration (AMD), diabetic macular water Swelling, corneal neovascularization, corneal graft neovascularization, corneal graft rejection, retinal/choroidal neovascularization, angle neovascularization (iris redness), ocular neovascular disease, vascular restenosis, and arteriovenous malformations (AVM). In some embodiments, the aforementioned cancer or angiogenic eye disease is associated with VEGF or ANG-2.
在在一些實施方案中,本揭露提供用作藥物的前述的雙特異性抗原結合分子,或前述的特異性結合ANG-2的抗體,或前述的核酸分子,或前述的醫藥組成物。 In some embodiments, the present disclosure provides the aforementioned bispecific antigen binding molecule, or the aforementioned antibody that specifically binds ANG-2, or the aforementioned nucleic acid molecule, or the aforementioned pharmaceutical composition for use as a medicament.
在一些實施方案中,本揭露提供用作藥物的前述的雙特異性抗原結合分子,或前述的特異性結合ANG-2的抗體,或前述的核酸分子,或前述的醫藥組成物,可用作治療或預防癌症或血管生成性眼病。在一些實施方案中,其中該癌症選自乳腺癌、腎上腺腫瘤、輸卵管癌、鱗狀細胞癌、卵巢癌、胃癌、結直腸癌、非小細胞肺癌、膽管癌、膀胱癌、胰腺癌、皮膚癌和肝癌。在一些實施方案中,其中該血管生成性眼病選自新生血管性青光眼、年齡相關的黃斑變性(AMD)、糖尿病性黃斑水腫、角膜新血管生成、角膜移植新血管生成、角膜移植排斥、視網膜/脈絡膜新血管生成、房角的新血管生成(虹膜發紅)、眼部新生血管性疾病、血管再狹窄和動靜脈畸形(AVM)。在一些實施方案中,前述的癌症或血管生成性眼病與VEGF或ANG-2相關。 In some embodiments, the present disclosure provides the aforementioned bispecific antigen-binding molecule, or the aforementioned antibody that specifically binds to ANG-2, or the aforementioned nucleic acid molecule, or the aforementioned pharmaceutical composition for use as a medicament, useful as a Treat or prevent cancer or angiogenic eye disease. In some embodiments, wherein the cancer is selected from breast cancer, adrenal tumor, fallopian tube cancer, squamous cell carcinoma, ovarian cancer, gastric cancer, colorectal cancer, non-small cell lung cancer, bile duct cancer, bladder cancer, pancreatic cancer, skin cancer and liver cancer. In some embodiments, wherein the angiogenic eye disease is selected from the group consisting of neovascular glaucoma, age-related macular degeneration (AMD), diabetic macular edema, corneal neovascularization, corneal transplant neovascularization, corneal transplant rejection, retinal/ Choroidal neovascularization, angle neovascularization (iris redness), ocular neovascular disease, vascular restenosis, and arteriovenous malformations (AVMs). In some embodiments, the aforementioned cancer or angiogenic eye disease is associated with VEGF or ANG-2.
圖1示出雙特異性抗體抑制ANG-2與Tie2的結合的結果; Figure 1 shows the results of bispecific antibodies inhibiting the binding of ANG-2 to Tie2;
圖2示出雙特異性抗體抑制誘導的Tie2磷酸化結果; Figure 2 shows the results of bispecific antibody inhibition-induced Tie2 phosphorylation;
圖3示出雙特異性抗體顯著抑制VEGF引起的HUVEC胞內磷酸化VEGFR水平升高; Figure 3 shows that bispecific antibodies significantly inhibited VEGF-induced increase in intracellular phosphorylated VEGFR levels in HUVECs;
圖4示出雙特異性抗體可以顯著抑制VEGF引起的HUVEC的增殖; Figure 4 shows that bispecific antibodies can significantly inhibit the proliferation of HUVECs induced by VEGF;
圖5示出本揭露中的雙特異性抗體均可顯著抑制小鼠體內的PC-3腫瘤生長; Figure 5 shows that all of the bispecific antibodies in the present disclosure can significantly inhibit PC-3 tumor growth in mice;
圖6示出本揭露中的雙特異性抗體均可顯著抑制小鼠體內的H460移植腫瘤生長; Figure 6 shows that all of the bispecific antibodies in the present disclosure can significantly inhibit H460 transplanted tumor growth in mice;
圖7示出本揭露中的雙特異性抗體可顯著抑制小鼠體內的A431移植腫瘤生長; Figure 7 shows that bispecific antibodies of the present disclosure can significantly inhibit A431 transplanted tumor growth in mice;
圖8A和圖8B示出本揭露的雙特異性抗體恆河猴脈絡膜新生血管的抑制結果;其中圖8A示出,本揭露中的雙特異性抗體對恆河猴眼中螢光滲漏面積的改善率,圖8B示出本揭露中的雙特異性抗體可顯著減少恆河猴眼中的螢光斑點數; 8A and 8B show the inhibition results of rhesus monkey choroidal neovascularization by the bispecific antibody of the present disclosure; wherein FIG. 8A shows the improvement of the fluorescent leakage area in the eyes of the rhesus monkey by the bispecific antibody of the present disclosure 8B shows that bispecific antibodies of the present disclosure can significantly reduce the number of fluorescent spots in the eyes of rhesus monkeys;
圖9示出本揭露中的雙特異性抗體可顯著降低恆河猴眼睛房水中VEGF的表達量。 Figure 9 shows that the bispecific antibody of the present disclosure can significantly reduce the expression of VEGF in the aqueous humor of rhesus monkey eyes.
圖10A和圖10B示出本揭露中示例性的雙特異性抗體的分子結構;圖10A顯示IgG-scFv類型雙特異性抗體的結構;圖10B顯示Crossmab類型雙特異性抗體的結構。 Figures 10A and 10B show the molecular structures of exemplary bispecific antibodies in the present disclosure; Figure 10A shows the structure of an IgG-scFv type bispecific antibody; Figure 10B shows the structure of a Crossmab type bispecific antibody.
術語 the term
為了更容易理解本揭露,以下具體定義了某些技術和科學術語。除非在本文中另有明確定義,本文使用的所有其它技術和科學術語都具有本揭露所屬技術領域具有通常知識者通常理解的含義。 For easier understanding of the present disclosure, certain technical and scientific terms are specifically defined below. Unless explicitly defined otherwise herein, all other technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this disclosure belongs.
本揭露所用胺基酸三字母代碼和單字母代碼如J.biol.chem,243,p3558(1968)中所述。 The three-letter and one-letter codes for amino acids used in this disclosure are as described in J. biol. chem, 243, p3558 (1968).
術語“胺基酸”是指天然存在的和合成的胺基酸,以及以與天然存在的胺基酸類似的方式起作用的胺基酸類似物和胺基酸模擬物。天然存在的胺基酸是由遺傳密碼編碼的那些胺基酸,以及後來修飾的那些胺基酸,例如羥脯胺酸、γ-羧基谷胺酸和O-鄰磷酸絲胺酸。胺基酸類似物是指與天然存在的胺基酸具有相同基本化學結構(即與氫結合的α碳、羧基、胺基和R基團,例如高絲胺酸、正亮胺酸、甲硫胺酸亞碸、甲硫胺酸甲基鋶)的化合物。此類類似物具有修飾的R基團(例如,正亮胺酸)或修飾的肽骨架,但保留與天然存在的胺基酸相同的基本化學結構。胺基酸模擬物是指具有如下結構的化合物,該結構與胺基酸的一般化學結構不同但是以與天然存在的胺基酸類似的方式起作用。 The term "amino acid" refers to naturally occurring and synthetic amino acids, as well as amino acid analogs and amino acid mimetics that function in a similar manner to naturally occurring amino acids. Naturally occurring amino acids are those encoded by the genetic code, as well as those amino acids that have been modified later, such as hydroxyproline, gamma-carboxyglutamate, and O-phosphoserine. Amino acid analogs are those that have the same basic chemical structure as naturally occurring amino acids (i.e., hydrogen-bonded alpha carbon, carboxyl, amine, and R groups, such as homoserine, norleucine, methionine, etc.) Compounds of sulfite, methyl methionine, methionine. Such analogs have modified R groups (eg, norleucine) or modified peptide backbones, but retain the same basic chemical structure as a naturally occurring amino acid. An amino acid mimetic refers to a compound having a structure that differs from the general chemical structure of amino acids but functions in a similar manner to naturally occurring amino acids.
術語“ANG-2”指血管生成素-2(ANGPT2或ANG2),其記載於例如Maisonpierre,P.C.等,Science 277(1997)55-60和Cheung,A.H.等,Genomics 48(1998)389-91。發現血管生成素-1和-2為Tie(即,一種在血管內皮內選擇性表達的酪胺酸激酶家族)的配體,Yancopoulos,G.D.等,Nature 407(2000)242-48。血管生成素家族有四種確定的成員,血管生成素-3和-4(ANG-3和ANG-4)可以代表小鼠和人中相同基因基因座的廣泛區域的對應物。Kim,I.等,FEBS Let,443(1999)353-56;Kim,I.等,J Biol Chem 274(1999)26523-28。ANG1和ANG2最初是在組織培養實驗中分別作為激動劑和拮抗劑鑑定的(對於ANG1,參見, Davis,S.等,Cell 87(1996)1161-69;對於ANG2,參見Maisonpierre,P.C.等,Science 277(1997)55-60)。所有已知的血管生成素主要結合Tie2,而ANG1和2兩者都以3nM(Kd)的親和力結合Tie2,Maisonpierre,P.C.等,Science 277(1997)55-60。 The term "ANG-2" refers to Angiopoietin-2 (ANGPT2 or ANG2), which is described, for example, in Maisonpierre, P.C. et al., Science 277 (1997) 55-60 and Cheung, A.H. et al., Genomics 48 (1998) 389-91. Angiopoietin-1 and -2 were found to be ligands for Tie, a family of tyrosine kinases selectively expressed within the vascular endothelium, Yancopoulos, G.D. et al., Nature 407 (2000) 242-48. There are four defined members of the angiopoietin family, and angiopoietin-3 and -4 (ANG-3 and ANG-4) may represent counterparts in a broad region of the same genetic locus in mice and humans. Kim, I. et al, FEBS Let, 443 (1999) 353-56; Kim, I. et al, J Biol Chem 274 (1999) 26523-28. ANG1 and ANG2 were originally identified as agonists and antagonists, respectively, in tissue culture experiments (for ANG1, see, Davis, S. et al., Cell 87 (1996) 1161-69; for ANG2, see Maisonpierre, P.C. et al., Science 277 (1997) 55-60). All known angiopoietins primarily bind Tie2, while both ANG1 and 2 bind Tie2 with an affinity of 3 nM (Kd), Maisonpierre, P.C. et al., Science 277 (1997) 55-60.
術語“VEGF”指人血管內皮生長因子(VEGF/VEGF-A),其記載於例如Leung,D.W.等,Science 246(1989)1306-9;Keck,P.J.等,Science 246(1989)1309-12和Connolly,D.T.等,J.Biol.Chem.264(1989)20017-24。VEGF參與調節正常的和異常的血管發生和與腫瘤和眼內病症有關的新血管化(Ferrara,N.,Endocr.Rev.18(1997)4-25;Berkman,R.A.,J.Clin.Invest.91(1993)153-159;Brown,L.F.等,HumanPathol.26(1995)86-91;Brown,L.F.等,Cancer Res.53(1993)4727-4735;Mattern,J.等,Brit.J.Cancer.73(1996)931-934:及Dvorak,H.F.等,Am.J.Pathol.146(1995)1029-1039)。VEGF是一種同二聚體糖蛋白,VEGF可促進對內皮細胞的促有絲分裂。 The term "VEGF" refers to human vascular endothelial growth factor (VEGF/VEGF-A), which is described, for example, in Leung, D.W. et al., Science 246 (1989) 1306-9; Keck, P.J. et al., Science 246 (1989) 1309-12 and Connolly, D.T. et al., J. Biol. Chem. 264 (1989) 20017-24. VEGF is involved in the regulation of normal and abnormal angiogenesis and neovascularization associated with tumors and intraocular disorders (Ferrara, N., Endocr. Rev. 18 (1997) 4-25; Berkman, R.A., J. Clin. Invest. 91 (1993) 153-159; Brown, L.F. et al., Human Pathol. 26 (1995) 86-91; Brown, L.F. et al., Cancer Res. 53 (1993) 4727-4735; Matter, J. et al., Brit. J. Cancer .73 (1996) 931-934: and Dvorak, H.F. et al., Am. J. Pathol. 146 (1995) 1029-1039). VEGF is a homodimeric glycoprotein that promotes mitogenicity to endothelial cells.
本揭露所述的“抗原結合分子”在本文中以最廣意義指特異性結合抗原的分子,抗原結合分子的例子是抗體、抗體片段、抗體融合蛋白或融合蛋白。示例性的,本文中的雙特異性抗原結合分子包含雙特異性抗體和抗體融合蛋白。示例性的,本文的雙特異性抗體或抗體融合蛋白可以包含第一條鏈與第二條鏈,該第一條鏈是抗體的重鏈或包含抗體重鏈的多肽,第二條鏈是抗體的輕鏈或包含抗體輕鏈的多肽。示例性的,在一些實施方案中,本文的雙特異性抗原結合分子具有全長抗體的基本結構,是由兩條相同的第一條鏈和兩條相同的第二條鏈藉由鏈間二硫鍵連接而成的四肽鏈結構;其中,第一條鏈包含抗VEGF抗體重鏈和抗ANG-2的單鏈抗體,第二條鏈包含抗VEGF抗體的輕鏈。 An "antigen-binding molecule" as used in the present disclosure is used herein in the broadest sense to refer to a molecule that specifically binds an antigen, examples of antigen-binding molecules are antibodies, antibody fragments, antibody fusion proteins, or fusion proteins. Exemplarily, bispecific antigen binding molecules herein include bispecific antibodies and antibody fusion proteins. Exemplarily, a bispecific antibody or antibody fusion protein herein can comprise a first chain, the first chain being the heavy chain of an antibody or a polypeptide comprising an antibody heavy chain, and a second chain being the antibody the light chain or a polypeptide comprising the light chain of an antibody. Illustratively, in some embodiments, the bispecific antigen-binding molecules herein have the basic structure of a full-length antibody, formed by two identical first chains and two identical second chains via interchain disulfides. A tetrapeptide chain structure formed by bonds; wherein, the first chain comprises the heavy chain of anti-VEGF antibody and the single-chain antibody of anti-ANG-2, and the second chain comprises the light chain of anti-VEGF antibody.
如本揭露所述的“雙特異性抗原結合分子”是指能同時結合兩個抗原或抗原決定簇的抗原結合分子,其包含結合第一抗原或抗原決定簇的第一抗原結合域和結合第二抗原或抗原決定簇的第二抗原結合域。在本揭露的一些實施方案中,第一抗原結合域特異性結合ANG-2,第二抗原結合域特異性結合VEGF;或者,在在本揭露的一些實施方案中,第一抗原結合域特異性結合VEGF,第二抗原結合域特異性結合ANG-2。 The "bispecific antigen-binding molecule" as used in the present disclosure refers to an antigen-binding molecule capable of simultaneously binding two antigens or antigenic determinants, comprising a first antigen-binding domain that binds to the first antigen or antigenic determinant and a second antigen-binding domain that binds to the first antigen or antigenic determinant. The second antigen-binding domain of a secondary antigen or antigenic determinant. In some embodiments of the present disclosure, the first antigen binding domain specifically binds ANG-2 and the second antigen binding domain specifically binds VEGF; alternatively, in some embodiments of the present disclosure, the first antigen binding domain specifically binds Binding to VEGF, the second antigen binding domain specifically binds ANG-2.
在一些實施方案中,本揭露所述的雙特異性抗原結合分子為雙特異性二價抗體或雙特異性四價抗體。 In some embodiments, the bispecific antigen binding molecules described in the present disclosure are bispecific bivalent antibodies or bispecific tetravalent antibodies.
在本領域中公知,根據不同結構可將雙特異性抗原結合分子分為2大類:含Fc片段的雙特異性抗原結合分子與不含Fc片段的雙特異性抗原結合分子。含Fc片段的雙特異性抗原結合分子結構包括但不限於TrioMab、Crossmab/KIH、DVD-Ig、IgG-scFv、FIT-Ig、mAb-Trap等。不含Fc片段的雙特異性抗原結合分子結構包括但不限於BiTE、DART、TandAb、ImmTAC、TriKE等。 It is well known in the art that bispecific antigen-binding molecules can be divided into two categories according to different structures: bispecific antigen-binding molecules containing an Fc fragment and bispecific antigen-binding molecules without an Fc fragment. Fc fragment-containing bispecific antigen-binding molecular structures include, but are not limited to, TrioMab, Crossmab/KIH, DVD-Ig, IgG-scFv, FIT-Ig, mAb-Trap, and the like. Bispecific antigen binding molecular structures without Fc fragments include, but are not limited to, BiTE, DART, TandAb, ImmTAC, TriKE, and the like.
如本揭露所述的“Crossmab”是存在Fc區域的IgG樣雙特異性抗原結合分子結構,其藉由在第一抗原結合域的Fab區內交換重鏈和輕鏈結構,從而導致VH-VL和CH1-CL之間界面的分子結構發生變化。經過交換的抗體輕鏈由於相互排斥的原理,即VH與VH相互排斥、CL與CL相互排斥,不易與未改造抗體的重鏈發生錯配,從而產生輕重鏈的正確配對。 A "Crossmab" as described in the present disclosure is an IgG-like bispecific antigen-binding molecule structure in the presence of an Fc region, which results in VH-VL by exchanging heavy and light chain structures within the Fab region of the first antigen-binding domain The molecular structure of the interface between CH1-CL and CH1-CL changes. Due to the principle of mutual exclusion, that is, the mutual exclusion of VH and VH, and the mutual exclusion of CL and CL, the exchanged antibody light chain is not prone to mismatch with the heavy chain of the unmodified antibody, resulting in the correct pairing of the light and heavy chains.
在另一些實施方案中,本揭露的雙特異性抗原結合分子是Crossmab形式,由第一重鏈,第一輕鏈,第二重鏈和第二輕鏈組成的四肽結構,其中,第一重鏈和第一輕鏈組成結合ANG-2的第一抗原結合域,第二重鏈和第 二輕鏈組成結合VEGF的第二抗原結合域,並且第一重鏈的CH1與第一輕鏈的CL互相交換。 In other embodiments, the bispecific antigen binding molecules of the present disclosure are in the form of a Crossmab, a tetrapeptide structure consisting of a first heavy chain, a first light chain, a second heavy chain, and a second light chain, wherein the first The heavy chain and the first light chain constitute the first antigen-binding domain that binds ANG-2, the second heavy chain and the first The two light chains constitute the second antigen-binding domain that binds VEGF, and the CH1 of the first heavy chain is exchanged with the CL of the first light chain.
如本揭露所述的“IgG-scFv”是存在Fc區域的IgG樣雙特異性抗原結合分子結構,其中一個抗原結合域的scFv融合到另一個抗原結合域IgG重鏈或輕鏈的C端,以構成IgG-scFv雙特異性抗原結合分子。 An "IgG-scFv" as described in the present disclosure is an IgG-like bispecific antigen-binding molecule structure with an Fc region, wherein the scFv of one antigen-binding domain is fused to the C-terminus of the IgG heavy or light chain of the other antigen-binding domain, To form an IgG-scFv bispecific antigen-binding molecule.
在另一些實施方案中,本揭露的雙特異性抗原結合分子是IgG-scFv形式。在示例性的實施方案中,特異性結合ANG-2的scFv直接地或藉由連接子與特異性結合VEGF的抗體重鏈C端連接,構成IgG-scFv形式雙特異性抗原結合分子。 In other embodiments, the bispecific antigen binding molecules of the present disclosure are in the form of IgG-scFv. In an exemplary embodiment, the scFv that specifically binds ANG-2 is linked directly or via a linker to the C-terminus of the heavy chain of an antibody that specifically binds VEGF to form a bispecific antigen binding molecule in the form of an IgG-scFv.
術語“價”指抗原結合分子中存在規定數目的結合位點。因此,術語“二價”、“四價”和“六價”分別指抗原結合分子中存在兩個結合位點、四個結合位點和六個結合位點。 The term "valency" refers to the presence of a defined number of binding sites in an antigen-binding molecule. Thus, the terms "bivalent", "tetravalent" and "hexavalent" refer to the presence of two, four and six binding sites, respectively, in an antigen-binding molecule.
本揭露所述的“能夠特異性結合ANG-2的第一抗原結合域”或“能夠特異性結合VEGF的第二抗原結合域”指抗原結合分子中包含與ANG-2或VEGF的全部或部分特異性結合的區域。例如,該抗原結合域可以包含一個或多個抗體可變區。特別的,能夠特異性結合抗原的抗原結合域包含抗體輕鏈可變區和抗體重鏈可變區,其可以形成scFv、Fab等構型。 The "first antigen-binding domain capable of specifically binding to ANG-2" or "the second antigen-binding domain capable of specifically binding to VEGF" in the present disclosure refers to the antigen-binding molecule comprising all or part of ANG-2 or VEGF specific binding region. For example, the antigen binding domain may comprise one or more antibody variable regions. In particular, the antigen binding domain capable of specifically binding an antigen comprises an antibody light chain variable region and an antibody heavy chain variable region, which can form scFv, Fab and other configurations.
術語“抗體融合蛋白”是指將目的蛋白質(多肽)與抗體連接形成的具有生物活性的融合蛋白,該融合蛋白具有所連接的蛋白質的生物學活性以及免疫球蛋白活性。 The term "antibody fusion protein" refers to a biologically active fusion protein formed by linking a protein (polypeptide) of interest with an antibody, and the fusion protein has the biological activity and immunoglobulin activity of the linked protein.
術語“抗體”以最廣義使用,並且涵蓋各種抗體結構,包括但不限於單株抗體、多株抗體、多特異性抗體(例如雙特異性抗體)、和抗體片段,只要它們展現出期望的抗原結合活性。 The term "antibody" is used in the broadest sense and encompasses a variety of antibody structures, including but not limited to monoclonal antibodies, polyclonal antibodies, multispecific antibodies (eg, bispecific antibodies), and antibody fragments so long as they exhibit the desired antigen binding activity.
“天然抗體”指具有不同結構的天然存在的免疫球蛋白分子。例如,天然IgG抗體是約150,000道爾頓的異四聚糖蛋白,由二硫化物鍵合的兩條相同輕鏈和兩條相同重鏈構成。從N至C端,每條重鏈具有一個可變區(VH),又稱作可變重域或重鏈可變域,接著是三個恆定域(CH1,CH2,和CH3)。類似地,從N至C端,每條輕鏈具有一個可變區(VL),又稱作可變輕域,或輕鏈可變域,接著是一個恆定輕(CL)域。根據其恆定域胺基酸序列,抗體輕鏈包括兩種類型,卡帕(κ)和拉姆達(λ)。根據抗體重鏈恆定區的胺基酸組成和排列順序不同,可將抗體分為五類,或稱為抗體同種型,即IgM、IgD、IgG、IgA和IgE,其相應的重鏈分別為μ鏈、δ鏈、γ鏈、α鏈、和ε鏈。同一類Ig根據其鉸鏈區胺基酸組成和重鏈二硫鍵的數目和位置的差別,又可分為不同的亞類,如IgG可分為IgG1、IgG2、IgG3、IgG4。五類Ig中每類Ig都可以有κ鏈或λ鏈。 "Native antibody" refers to naturally-occurring immunoglobulin molecules with different structures. For example, native IgG antibodies are heterotetrameric glycoproteins of approximately 150,000 Daltons composed of two identical light chains and two identical heavy chains that are disulfide-bonded. From N to C-terminus, each heavy chain has a variable domain (VH), also known as a variable heavy domain or heavy chain variable domain, followed by three constant domains (CH1, CH2, and CH3). Similarly, from N to C-terminus, each light chain has a variable region (VL), also known as a variable light domain, or light chain variable domain, followed by a constant light (CL) domain. Antibody light chains include two types, kappa (κ) and lambda (λ), based on their constant domain amino acid sequences. According to the different amino acid composition and arrangement sequence of the constant region of the antibody heavy chain, antibodies can be divided into five categories, or antibody isotypes, namely IgM, IgD, IgG, IgA and IgE, and their corresponding heavy chains are μ chain, delta chain, gamma chain, alpha chain, and epsilon chain. The same type of Ig can be divided into different subclasses according to the difference in the amino acid composition of its hinge region and the number and position of disulfide bonds in the heavy chain. For example, IgG can be divided into IgG1, IgG2, IgG3, and IgG4. Each of the five classes of Ig can have a kappa chain or a lambda chain.
抗體重鏈和輕鏈靠近N端的約110個胺基酸的序列變化很大,為可變區;靠近C端的其餘胺基酸序列相對穩定,為恆定區。每條重鏈由重鏈可變區(本文中縮寫為VH)和重鏈恆定區組成。重鏈恆定區由三個結構域(CH1、CH2和CH3)組成。每條輕鏈由輕鏈可變區(本文中縮寫為VL)和輕鏈恆定區組成。輕鏈恆定區包含一個結構域,CL。VH和VL區可進一步細分為高變區,稱為互補性決定區(CDR),其間穿插有稱為框架區(FR)的較保守區。每條輕鏈的包含3個CDR區:LCDR1、LCDR2、和LCDR3;每條重鏈的包含3個CDR區:HCDR1、HCDR2和HCDR3。每個VH和VL由從胺基末端排到羧基末端按以下順序排列 的三個CDR和四個FR構成:FR1、CDR1、FR2、CDR2、FR3、CDR3、FR4。重鏈和輕鏈的可變區含有與抗原相互作用的結合結構域。抗體的恆定區可以介導免疫球蛋白與宿主組織或因子(包括免疫系統的各種細胞(例如,效應細胞)和經典補體系統的第一組分(Clq))的結合。 The sequences of about 110 amino acids near the N-terminus of the antibody heavy and light chains vary greatly and are variable regions; the remaining amino acid sequences near the C-terminus are relatively stable and are constant regions. Each heavy chain consists of a heavy chain variable region (abbreviated herein as VH) and a heavy chain constant region. The heavy chain constant region consists of three domains (CH1, CH2 and CH3). Each light chain consists of a light chain variable region (abbreviated herein as VL) and a light chain constant region. The light chain constant region contains one domain, CL. The VH and VL regions can be further subdivided into hypervariable regions, termed complementarity determining regions (CDRs), interspersed with more conserved regions termed framework regions (FRs). Each light chain contains 3 CDR regions: LCDR1, LCDR2, and LCDR3; each heavy chain contains 3 CDR regions: HCDR1, HCDR2, and HCDR3. Each VH and VL is arranged in the following order from the amino terminus to the carboxy terminus It consists of three CDRs and four FRs: FR1, CDR1, FR2, CDR2, FR3, CDR3, FR4. The variable regions of the heavy and light chains contain binding domains that interact with the antigen. The constant regions of the antibodies can mediate the binding of the immunoglobulin to host tissues or factors, including various cells of the immune system (eg, effector cells) and the first component (Clq) of the classical complement system.
術語“互補決定區”、“CDR”或“高變區”是指可變結構域內主要促成抗原結合的區域。通常,每個重鏈可變區中存在三個CDR(HCDR1、HCDR2、HCDR3),每個輕鏈可變區中存在三個CDR(LCDR1、LCDR2、LCDR3)。可以各種公知方案來確定CDR的胺基酸序列邊界,例如:“Kabat”編號規則(參見Kabat等(1991),“Sequences of Proteins of Immunological Interest”,第5版,Public Health Service,National Institutes of Health,Bethesda,MD)、“Chothia”編號規則(參見Martin,ACR.Protein Sequence and Structure Analysis of Antibody Variable Domains[J].2001)和ImMunoGenTics(IMGT)編號規則(Lefranc,M.P.等,Dev.Comp.Immunol.,27,55-77(2003);Front Immunol.2018 Oct 16;9:2278等)。包括例如Kabat編號和IMGT獨特編號系統在內的編號系統之間的關係是所屬技術領域具有通常知識者熟知的。 The terms "complementarity determining regions", "CDRs" or "hypervariable regions" refer to regions within a variable domain that primarily contribute to antigen binding. Typically, there are three CDRs (HCDR1, HCDR2, HCDR3) in each heavy chain variable region and three CDRs (LCDR1, LCDR2, LCDR3) in each light chain variable region. The amino acid sequence boundaries of CDRs can be determined by various well-known schemes, for example: the "Kabat" numbering convention (see Kabat et al. (1991), "Sequences of Proteins of Immunological Interest", 5th ed., Public Health Service, National Institutes of Health , Bethesda, MD), the "Chothia" numbering scheme (see Martin, ACR. Protein Sequence and Structure Analysis of Antibody Variable Domains [J]. 2001) and the ImMunoGenTics (IMGT) numbering scheme (Lefranc, M.P. et al., Dev.Comp.Immunol ., 27, 55-77 (2003); Front Immunol. 2018 Oct 16; 9:2278 et al). The relationship between numbering systems, including, for example, the Kabat numbering and the IMGT unique numbering system is well known to those of ordinary skill in the art.
例如,對於經典格式,遵循Kabat規則,該重鏈可變域(VH)中的CDR胺基酸殘基編號為31-35(HCDR1)、50-65(HCDR2)和95-102(HCDR3);輕鏈可變域(VL)中的CDR胺基酸殘基編號為24-34(LCDR1)、50-56(LCDR2)和89-97(LCDR3)。 For example, for the classical format, following Kabat's rules, the CDR amino acid residues in the heavy chain variable domain (VH) are numbered 31-35 (HCDR1), 50-65 (HCDR2) and 95-102 (HCDR3); The CDR amino acid residues in the light chain variable domain (VL) are numbered 24-34 (LCDR1), 50-56 (LCDR2) and 89-97 (LCDR3).
遵循Chothia規則,VH中的CDR胺基酸編號為26-32(HCDR1)、52-56(HCDR2)和95-102(HCDR3);並且VL中的胺基酸殘基編號為24-34(LCDR1)、50-56(LCDR2)和89-97(LCDR3)。 Following Chothia's rule, the CDR amino acids in VH are numbered 26-32 (HCDR1), 52-56 (HCDR2) and 95-102 (HCDR3); and the amino acid residues in VL are numbered 24-34 (LCDR1 ), 50-56 (LCDR2) and 89-97 (LCDR3).
藉由組合Kabat和Chothia兩者的CDR定義,CDR由人VH中的胺基酸殘基26-35(HCDR1)、50-65(HCDR2)和95-102(HCDR3)和人VL中的胺基酸殘基24-34(LCDR1)、50-56(LCDR2)和89-97(LCDR3)構成。 Defined by combining the CDRs of both Kabat and Chothia, the CDRs are defined by amino acid residues 26-35 (HCDR1), 50-65 (HCDR2) and 95-102 (HCDR3) in human VH and amine groups in human VL Acid residues 24-34 (LCDR1), 50-56 (LCDR2) and 89-97 (LCDR3) constitute.
遵循IMGT規則,VH中的CDR胺基酸殘基編號大致為27-38(CDR1)、56-65(CDR2)和105-117(CDR3),VL中的CDR胺基酸殘基編號大致為27-38(CDR1)、56-65(CDR2)和105-117(CDR3)。遵循IMGT規則,抗體的CDR區可以使用程序IMGT/DomainGap Align確定。 Following IMGT rules, CDR amino acid residues in VH are numbered approximately 27-38 (CDR1), 56-65 (CDR2), and 105-117 (CDR3), and CDR amino acid residues in VL are approximately numbered 27 -38 (CDR1), 56-65 (CDR2) and 105-117 (CDR3). Following the rules of IMGT, the CDR regions of an antibody can be determined using the program IMGT/DomainGap Align.
遵循AbM規則,VH中的CDR胺基酸編號為26-35(HCDR1)、50-58(HCDR2)和95-102(HCDR3);並且VL中的胺基酸殘基編號為24-34(LCDR1)、50-56(LCDR2)和89-97(LCDR3)。 Following AbM rules, the CDR amino acids in VH are numbered 26-35 (HCDR1), 50-58 (HCDR2) and 95-102 (HCDR3); and the amino acid residues in VL are numbered 24-34 (LCDR1 ), 50-56 (LCDR2) and 89-97 (LCDR3).
除非另有說明,本揭露實施例中的可變區和CDR序列均適用“Kabat”編號規則。 Unless otherwise specified, the "Kabat" numbering convention applies to the variable regions and CDR sequences in the embodiments of the present disclosure.
術語“抗體框架”或“FR區”,是指可變結構域VL或VH的一部分,其用作該可變結構域的抗原結合環(CDR)的支架。從本質上講,其是不具有CDR的可變結構域。 The term "antibody framework" or "FR region" refers to the portion of a variable domain VL or VH that serves as a scaffold for the antigen binding loops (CDRs) of the variable domain. Essentially, it is a variable domain without CDRs.
“抗體恆定區結構域”指來源於抗體的輕鏈和重鏈的恆定區的結構域,包括CL和來源於不同類抗體的CH1、CH2、CH3和CH4結構域。本揭露的恆定區還包括該人抗體重鏈恆定區和人抗體輕鏈恆定區的“常規變體”,其指現有技術已公開的來源於人的不改變抗體可變區結構和功能的重鏈恆定區或輕鏈恆定區的變體,示例性變體包括對重鏈恆定區進行定點改造和胺基酸替換的IgG1、IgG2、IgG3或IgG4重鏈恆定區變體,具體替換如現有技術已知的YTE突變、L234A和/或L235A突變、S228P突變、和/或獲得knob-into-hole結構的 突變(使得抗體重鏈具有knob-Fc和hole-Fc組合),這些突變已被證實使得抗體具有新的性能,但不改變抗體可變區的功能。 "Antibody constant region domain" refers to domains derived from the constant regions of the light and heavy chains of antibodies, including CL and CH1, CH2, CH3 and CH4 domains derived from different classes of antibodies. The constant regions of the present disclosure also include "conventional variants" of the human antibody heavy chain constant region and the human antibody light chain constant region, which refer to the human-derived recombinants disclosed in the prior art that do not change the structure and function of the antibody variable region. Variants of chain constant regions or light chain constant regions, exemplary variants include IgG1, IgG2, IgG3 or IgG4 heavy chain constant region variants with site-directed reengineering and amino acid substitutions of the heavy chain constant region, specific replacements as in the prior art Known YTE mutation, L234A and/or L235A mutation, S228P mutation, and/or acquired knob-into-hole structure Mutations (giving the antibody heavy chain a combination of knob-Fc and hole-Fc) that have been shown to confer new properties to the antibody without altering the function of the variable region of the antibody.
“抗體片段”指不同於完整抗體的分子,其包含完整抗體的部分,該部分與完整抗體所結合的抗原相結合。抗體片段的實例包括但不限於Fv、Fab、Fab’、Fab’-SH、F(ab')2、Fd、dAb;駝類VHH結構域;雙抗體;線性抗體;單鏈抗體分子(例如scFv);以及由抗體片段形成的多特異性抗體。 An "antibody fragment" refers to a molecule other than an intact antibody that comprises a portion of the intact antibody that binds to the antigen to which the intact antibody binds. Examples of antibody fragments include, but are not limited to, Fv, Fab, Fab', Fab'-SH, F(ab ' )2, Fd, dAb; camelid VHH domains; diabodies; linear antibodies; ); and multispecific antibodies formed from antibody fragments.
“單鏈可變片段(scFv)”,也稱“單鏈抗體”,是藉由接頭連接的抗體的重鏈可變區(VH)和輕鏈可變區(VL)的融合蛋白。特別地,接頭是10至25個胺基酸的短多肽且通常富含有關柔性的甘胺酸,以及有關溶解性的絲胺酸或蘇胺酸,並且可將VH的N端與VL的C端連接,或反之亦然。儘管去除了恆定區並引入了接頭,但這種蛋白質保留了原始抗體的特異性。關於scFv片段的綜述,見例如Pluckthün,於ThePharmacology of Monoclonal Antibodies,第113卷,Rosenburg和Moore編,(Springer-Verlag,New York),第269-315頁(1994);還可見WO 93/16185;和美國專利No.5,571,894和5,587,458。 "Single-chain variable fragments (scFv)", also referred to as "single-chain antibodies", are fusion proteins of the variable heavy (VH) and light chain (VL) regions of an antibody linked by a linker. In particular, linkers are short polypeptides of 10 to 25 amino acids and are usually rich in glycine for flexibility, and serine or threonine for solubility, and can link the N-terminus of VH to the C of VL end connections, or vice versa. This protein retains the specificity of the original antibody despite the removal of the constant region and the introduction of linkers. For a review of scFv fragments see, eg, Pluckthün, in The Pharmacology of Monoclonal Antibodies, Vol. 113, Rosenburg and Moore, eds. (Springer-Verlag, New York), pp. 269-315 (1994); see also WO 93/16185; and US Patent Nos. 5,571,894 and 5,587,458.
“接頭”或“連接子”指用於連接多肽(如蛋白質結構域)的多肽序列,通常具有一定的柔性,接頭的使用不會使多肽原有結構和功能喪失。 "Linker" or "linker" refers to a polypeptide sequence used to connect polypeptides (such as protein domains), usually with a certain flexibility, and the use of the linker will not cause the loss of the original structure and function of the polypeptide.
術語“Fc區”在本文中用於定義免疫球蛋白重鏈的C端區域,其含有恆定區的至少部分。該術語包括天然序列Fc區和變體Fc區。Fc區的C末端賴胺酸(Lys447)可以存在或不存在。除非本文另有說明,Fc區或恆定區中的胺基酸殘基編號根據EU編號系統,也稱為EU索引。 The term "Fc region" is used herein to define the C-terminal region of an immunoglobulin heavy chain, which contains at least part of the constant region. The term includes native sequence Fc regions and variant Fc regions. The C-terminal lysine (Lys447) of the Fc region may or may not be present. Unless otherwise stated herein, amino acid residues in the Fc region or constant region are numbered according to the EU numbering system, also known as the EU index.
術語“結合位點”或“抗原結合位點”指抗體分子與抗原實際結合的區域。術語“抗原結合位點”包含抗體重鏈可變域(VH)和抗體輕鏈可變域(VL),或僅包含抗體重鏈可變域或輕鏈可變結構域。 The term "binding site" or "antigen binding site" refers to the region of an antibody molecule that actually binds to an antigen. The term "antigen binding site" comprises an antibody heavy chain variable domain (VH) and an antibody light chain variable domain (VL), or only the antibody heavy chain variable domain or the light chain variable domain.
“嵌合”抗體指其中的重鏈和/或輕鏈的一部分自特定的來源或物種衍生,而重鏈和/或輕鏈的剩餘部分自不同來源或物種衍生的抗體。 A "chimeric" antibody refers to an antibody in which a portion of the heavy and/or light chain is derived from a particular source or species, and the remainder of the heavy and/or light chain is derived from a different source or species.
術語“人源化”抗體是保留非人抗體的反應性同時在人中具有較低免疫原性的抗體。例如,這可以藉由保留非人CDR區並用其人對應物(即,恆定區以及可變區的框架部分)替換抗體的其餘部分來實現。參見,例如,Morrison等人,Proc.Natl.Acad.Sci.USA[美國國家科學院院刊],81:6851-6855,1984;Morrison和Oi,Adv.Immunol.[免疫學進展],44:65-92,1988;Verhoeyen等人,Science[科學],239:1534-1536,1988;Padlan,Molec.Immun.[分子免疫學],28:489-498,1991;以及Padlan,Molec.Immun.[分子免疫學],31:169-217,1994。人工程化技術的其他實例包括但不限於在US 5,766,886中披露的Xoma技術。 The term "humanized" antibody is an antibody that retains the reactivity of a non-human antibody while being less immunogenic in humans. For example, this can be accomplished by retaining the non-human CDR regions and replacing the rest of the antibody with their human counterparts (ie, the constant regions and framework portions of the variable regions). See, eg, Morrison et al., Proc. Natl. Acad. Sci. USA, 81:6851-6855, 1984; Morrison and Oi, Adv. Immunol. -92, 1988; Verhoeyen et al, Science, 239: 1534-1536, 1988; Padlan, Molec. Immun. [Molecular Immunology], 28: 489-498, 1991; and Padlan, Molec. Immun. [ Molecular Immunology], 31: 169-217, 1994. Other examples of human engineering techniques include, but are not limited to, the Xoma technique disclosed in US 5,766,886.
通常,將非人抗體人源化以降低對人的免疫原性,同時保留親本非人抗體的特異性和親和力。一般地,人源化抗體包含一個或多個可變域,其中HVR(重鏈可變區),例如CDR(或其部分)自非人抗體衍生,而FR(或其部分)自人抗體序列衍生。視需要地,人源化抗體還會至少包含人恆定區的一部分。在一些實施方案中,將人源化抗體中的一些FR殘基用來自非人抗體(例如衍生HVR殘基的抗體)的相應殘基替代,例如以恢復或改善抗體特異性或親和力。 Typically, non-human antibodies are humanized to reduce immunogenicity to humans while retaining the specificity and affinity of the parental non-human antibody. Generally, humanized antibodies comprise one or more variable domains, wherein HVRs (heavy chain variable regions), eg, CDRs (or portions thereof), are derived from non-human antibodies, and FRs (or portions thereof) are derived from human antibody sequences derivative. Optionally, humanized antibodies will also contain at least a portion of human constant regions. In some embodiments, some FR residues in a humanized antibody are replaced with corresponding residues from a non-human antibody (eg, an antibody from which the HVR residues are derived), eg, to restore or improve antibody specificity or affinity.
術語“人抗體”旨在包括具有可變區的抗體,其中框架區和CDR區兩者均衍生自人來源的序列。此外,如果抗體含有恆定區,則恆定區還衍生自此類人序列,例如人種系序列或突變形式的人種系序列。本揭露的人抗體可以包括 不是由人序列編碼的胺基酸殘基(例如,藉由在體外隨機誘變或位點特異性誘變或藉由在體內體細胞突變來引入的突變)。 The term "human antibody" is intended to include antibodies having variable regions in which both the framework and CDR regions are derived from sequences of human origin. Furthermore, if the antibody contains a constant region, the constant region is also derived from such human sequences, eg, human germline sequences or mutant forms of human germline sequences. Human antibodies of the present disclosure can include Amino acid residues not encoded by human sequences (eg, mutations introduced by random or site-specific mutagenesis in vitro or by somatic mutation in vivo).
術語“全長抗體”,“完整抗體”和“全抗體”在本文中可互換使用,指與天然抗體結構具有基本上類似的結構或者具有含有如本文中所限定的Fc區的重鏈的抗體。 The terms "full length antibody", "intact antibody" and "whole antibody" are used interchangeably herein to refer to an antibody having a substantially similar structure to that of a native antibody or having a heavy chain containing an Fc region as defined herein.
術語“抗原”是指能夠由諸如抗原結合蛋白(包括例如抗體)的選擇性結合劑結合且另外能夠用於動物中以產生能夠結合該抗原的抗體的分子或分子部分。抗原可具有一個或多個能夠與不同的抗原結合蛋白(例如抗體)相互作用的表位。 The term "antigen" refers to a molecule or molecular portion capable of being bound by a selective binding agent such as an antigen-binding protein (including, for example, an antibody) and otherwise capable of being used in an animal to generate an antibody capable of binding the antigen. Antigens can have one or more epitopes capable of interacting with different antigen binding proteins (eg, antibodies).
術語“表位”指能夠與抗體或其抗原結合片段特異性結合的抗原上的區域(area或region)。表位可以自連續胺基酸串(線性表位)形成或包含非連續胺基酸(構象表位),例如因抗原的折疊(即藉由蛋白質性質的抗原的三級折疊)而變成空間接近。構象表位和線性表位的差別在於:在變性溶劑的存在下,抗體對構象表位的結合喪失。例如,表位包含處於獨特空間構象的至少3、至少4、至少5、至少6、至少7、或8-10個胺基酸。 The term "epitope" refers to an area or region on an antigen that is capable of specific binding by an antibody or antigen-binding fragment thereof. Epitopes can be formed from contiguous strings of amino acids (linear epitopes) or comprise non-contiguous amino acids (conformational epitopes), eg, brought into steric proximity due to folding of the antigen (ie, tertiary folding of the antigen by proteinaceous nature) . A conformational epitope differs from a linear epitope in that in the presence of a denaturing solvent, binding of the antibody to the conformational epitope is lost. For example, an epitope comprises at least 3, at least 4, at least 5, at least 6, at least 7, or 8-10 amino acids in a unique spatial conformation.
篩選結合特定表位的抗體(即那些結合相同表位的)可以使用本領域例行方法來進行,例如但不限於丙胺酸掃描、肽印跡(見Meth.Mol.Biol.248(2004)443-463)、肽切割分析、表位切除、表位提取、抗原的化學修飾(見Prot.Sci.9(2000)487-496)、和交叉阻斷(見“Antibodies,”Harlow and Lane(Cold Spring Harbor Press,Cold Spring Harb.,NY))。 Screening for antibodies that bind a particular epitope (ie, those that bind the same epitope) can be performed using methods routine in the art, such as, but not limited to, alanine scanning, peptide blotting (see Meth. Mol. Biol. 248 (2004) 443- 463), peptide cleavage analysis, epitope excision, epitope extraction, chemical modification of antigens (see Prot. Sci. 9 (2000) 487-496), and cross-blocking (see "Antibodies," Harlow and Lane (Cold Spring) Harbor Press, Cold Spring Harb., NY)).
還可以使用競爭性結合來鑑定抗體是否與參照抗抗體結合相同表位或競爭結合。例如,與參照抗體“結合相同表位的抗體”指在競爭測定法中將 參照抗體對其抗原的結合阻斷50%或更多的抗體,且相反,參照抗體在競爭測定法中將該抗體對其抗原的結合阻斷50%或更多。還例如,為了測定待測抗體是否與參照抗體結合相同表位,在飽和條件下容許參照抗體結合抗原。在去除過量的參照抗體後,評估待測抗體結合抗原的能力。如果該待測抗體能夠在參照抗體的飽和結合之後結合抗原,那麼可以得出結論,該待測抗體與參照抗體結合不同表位。但是,如果該待測抗體在參照抗體的飽和結合之後不能夠結合抗原,那麼該待測抗體可結合與參照抗體結合相同的表位。為了確認待測抗體是否結合相同表位或僅僅受到空間原因阻礙結合,可以使用例行實驗(例如,肽突變和使用ELISA、RIA、表面電漿共振、流式細胞術或本領域可獲得的任何其它定量或定性抗體結合測定的結合分析)。這種測定法應當以兩種設置中進行,即兩種抗體均作為飽和抗體。如果在兩種設置中,均只有第一(飽和)抗體能夠結合抗原,那麼可以得出結論,該待測抗體和參照抗體競爭結合該抗原。 Competitive binding can also be used to identify whether an antibody binds the same epitope or competes for binding as a reference anti-antibody. For example, an antibody that "binds the same epitope" as a reference antibody means that in a competition assay The reference antibody blocks binding of the antibody to its antigen by 50% or more, and in contrast, the reference antibody blocks the binding of the antibody to its antigen by 50% or more in a competition assay. Also for example, to determine whether a test antibody binds to the same epitope as a reference antibody, the reference antibody is allowed to bind to the antigen under saturating conditions. After removal of excess reference antibody, the ability of the test antibody to bind the antigen is assessed. If the test antibody is able to bind the antigen after saturation binding of the reference antibody, then it can be concluded that the test antibody binds to different epitopes than the reference antibody. However, if the test antibody is unable to bind the antigen after saturation binding of the reference antibody, then the test antibody can bind to the same epitope as the reference antibody. To confirm whether the antibody to be tested binds to the same epitope or is merely hindered by steric reasons, routine experiments (eg, peptide mutation and use of ELISA, RIA, surface plasmon resonance, flow cytometry, or any other method available in the art can be used) Binding assays for other quantitative or qualitative antibody binding assays). This assay should be performed in both settings, i.e. both antibodies as saturating antibodies. If, in both settings, only the first (saturating) antibody is able to bind the antigen, then it can be concluded that the test antibody and the reference antibody compete for binding to the antigen.
在一些實施方案中,如在競爭性結合測定中所測量的(參見例如,Junghans等人,Cancer Res.50(1990)1495-1502),如果一個抗體的1倍、5倍、10倍、20倍或100倍過量抑制另一個抗體的結合至少50%、至少75%、至少90%或甚至99%或更多,則認為兩個抗體結合相同或重疊的表位。 In some embodiments, as measured in a competitive binding assay (see, eg, Junghans et al., Cancer Res. 50 (1990) 1495-1502), if an antibody is 1-fold, 5-fold, 10-fold, 20-fold Two antibodies are considered to bind the same or overlapping epitope if they inhibit binding of the other antibody by at least 50%, at least 75%, at least 90%, or even 99% or more by a fold or 100-fold excess.
如此處該,抗體和/或其抗原結合片段之間的“競爭”意味著兩種抗體(或其結合片段)結合相同或重疊的表位(例如,如藉由競爭性結合測定,藉由所屬技術領域具有通常知識者所熟知的任一方法所確定的)。如果該競爭性抗體或其抗原結合片段與本揭露的抗體或抗原結合片段結合相同的表位或重疊的表位,則該抗體或其抗原結合片段還與本揭露的抗體或抗原結合片段“競爭”。如本文所使用的,競爭性抗體或其抗原結合片段還可包括如下:(i)空間阻斷 本揭露的抗體或抗原結合片段結合其靶標的競爭性抗體或其抗原結合片段(例如,如果該競爭性抗體結合臨近的、非重疊的和/或相同表位並且以物理的方式防止本揭露的抗體或抗原結合片段結合其靶標);和/或(ii)結合不同的、非重疊的表位並且誘導抗原發生如下構象變化的競爭性抗體或其抗原結合片段,該構象變化使得該抗原不再以無該構象變化時具有的方式結合本揭露的抗體或抗原結合片段。 As used herein, "competition" between antibodies and/or antigen-binding fragments thereof means that two antibodies (or binding fragments thereof) bind the same or overlapping epitopes (eg, as determined by competitive binding, by belonging to as determined by any method known to those of ordinary skill in the art). If the competing antibody or antigen-binding fragment thereof binds the same epitope or overlapping epitope as the antibody or antigen-binding fragment of the present disclosure, the antibody or antigen-binding fragment thereof also "competes with the antibody or antigen-binding fragment of the present disclosure" ". As used herein, a competing antibody or antigen-binding fragment thereof may also include the following: (i) steric blocking An antibody or antigen-binding fragment of the present disclosure binds a competing antibody or antigen-binding fragment thereof to its target (eg, if the competing antibody binds adjacent, non-overlapping, and/or the same epitope and physically prevents the antibody or antigen-binding fragment that binds its target); and/or (ii) a competing antibody or antigen-binding fragment thereof that binds to distinct, non-overlapping epitopes and induces a conformational change in the antigen that renders the antigen no longer The antibodies or antigen-binding fragments of the present disclosure are bound in a manner that would be present in the absence of such conformational changes.
“特異性地結合”、“特異性結合”或“結合”是指抗體以比針對其他抗原或表位更高的親和力結合至某個抗原或該抗原內的表位。通常,抗體以約1×10-7M或更小(例如約1×10-8M或更小、約1×10-9M或更小、約1×10-10M或更小、約1×10-11M或更小、或者約1×10-12M或更小)的平衡解離常數(KD)結合抗原或抗原內的表位,通常KD為該抗體結合至非特異性抗原(例如BSA、酪蛋白)的KD的至少百分之一。可使用標準程序來測量KD。然而,特異性結合至抗原或抗原內的表位的抗體可能對其它相關的抗原具有交叉反應性,例如,對來自其它物種(同源)(諸如人或猴,例如食蟹獼猴(Macaca fascicularis)(cynomolgus,cyno)、黑猩猩(Pan troglodytes)(chimpanzee,chimp)或狨猴(Callithrix jacchus)(commonmarmoset,marmoset)的相同抗原具有交叉反應性。 "Specifically binds", "specifically binds" or "binds" means that an antibody binds to an antigen or epitope within that antigen with a higher affinity than for other antigens or epitopes. Typically, antibodies are prepared at about 1 x 10-7 M or less (eg, about 1 x 10-8 M or less, about 1 x 10-9 M or less, about 1 x 10-10 M or less, about 1 x 10-10 M or less, about An equilibrium dissociation constant (KD) of 1 x 10-11 M or less, or about 1 x 10-12 M or less) binds an antigen or an epitope within an antigen, typically the KD is the binding of the antibody to a nonspecific antigen ( For example, at least one percent of the KD of BSA, casein). KD can be measured using standard procedures. However, antibodies that specifically bind to an antigen or to an epitope within an antigen may be cross-reactive to other related antigens, eg, to those from other species (homologous) such as humans or monkeys, eg, Macaca fascicularis (cynomolgus, cyno), chimpanzees (Pan troglodytes) (chimpanzee, chimp) or marmosets (Callithrix jacchus) (common marmoset, marmoset) are cross-reactive.
“親和力”指分子(例如抗體)的單一結合位點與其結合配偶體(例如抗原)之間全部非共價相互作用總和的強度。分子X對其配偶體Y的親和力通常可以用解離常數(KD)來表述。親和力可以藉由本領域知道的常用方法來測量,包括本文中所描述的方法。 "Affinity" refers to the strength of the sum of all non-covalent interactions between a single binding site of a molecule (eg, an antibody) and its binding partner (eg, an antigen). The affinity of a molecule X for its partner Y can generally be expressed in terms of the dissociation constant (KD). Affinity can be measured by common methods known in the art, including those described herein.
術語“kassoc”或“ka”意在是指特定抗體-抗原相互作用的締合速率,而如本文所使用的術語“kdis”或“kd”意在是指特定抗體-抗原相互作用的解離 速率。如本文所使用的,術語“KD”意在是指解離常數,其獲得自kd與ka的比率(即kd/ka)並且表示為莫耳濃度(M)。可以使用本領域良好建立的方法測定抗體的KD值。用於測定抗體KD的方法包括使用生物傳感系統例如系統測量表面電漿共振,或藉由溶液平衡滴定法(SET)測量溶液中的親和力。 The term "kassoc" or "ka" is intended to refer to the rate of association of a particular antibody-antigen interaction, while the term "kdis" or "kd" as used herein is intended to refer to the dissociation of a particular antibody-antigen interaction rate. As used herein, the term "KD" is intended to refer to the dissociation constant, which is obtained from the ratio of kd to ka (ie, kd/ka) and expressed as molar concentration (M). The KD value of an antibody can be determined using methods well established in the art. Methods for determining antibody KD include measuring surface plasmon resonance using a biosensing system such as a system, or measuring affinity in solution by solution equilibrium titration (SET).
術語“核酸”在本文中可與術語“多核苷酸”互換使用,並且是指呈單鏈或雙鏈形式的脫氧核糖核苷酸或核糖核苷酸及其聚合物。該術語涵蓋含有已知核苷酸類似物或修飾的骨架殘基或連接的核酸,該核酸是合成的、天然存在的和非天然存在的,具有與參考核酸相似的結合特性,並且以類似於參考核苷酸的方式代謝。此類類似物的實例包括但不限於硫代磷酸酯、胺基磷酸酯、甲基膦酸酯、手性-甲基膦酸酯、2-O-甲基核糖核苷酸、肽-核酸(PNA)。 The term "nucleic acid" is used interchangeably herein with the term "polynucleotide" and refers to deoxyribonucleotides or ribonucleotides and polymers thereof in single- or double-stranded form. The term encompasses nucleic acids containing known nucleotide analogs or modified backbone residues or linkages, which are synthetic, naturally occurring and non-naturally occurring, have binding properties similar to the reference nucleic acid, and which are similar to the reference nucleic acid. Refers to the way nucleotides are metabolized. Examples of such analogs include, but are not limited to, phosphorothioates, phosphoramidates, methylphosphonates, chiral-methylphosphonates, 2-O-methylribonucleotides, peptide-nucleic acids ( PNA).
除非另有說明,否則特定的核酸序列還隱含地涵蓋其保守修飾的變體(例如,簡並密碼子取代)和互補序列以及明確指明的序列。具體地,如下詳述,簡併密碼子取代可以藉由產生如下序列而獲得,在這些序列中,一個或多個所選的(或全部)密碼子的第三位被混合鹼基和/或脫氧肌苷殘基取代(Batzer等人,Nucleic Acid Res[核酸研究].19:5081,1991;Ohtsuka等人,J.Biol.Chem[生物化學雜誌].260:2605-2608,1985;和Rossolini等人,Mol.Cell.Probes[分子與細胞探針]8:91-98,1994)。 Unless otherwise stated, a particular nucleic acid sequence also implicitly encompasses conservatively modified variants thereof (eg, degenerate codon substitutions) and complementary sequences as well as explicitly indicated sequences. In particular, as detailed below, degenerate codon substitutions can be obtained by generating sequences in which one or more selected (or all) codons are mixed base and/or deoxygenated at the third position Inosine residue substitution (Batzer et al., Nucleic Acid Res. 19:5081, 1991; Ohtsuka et al., J. Biol. Chem. 260:2605-2608, 1985; and Rossolini et al. Human, Mol. Cell. Probes 8:91-98, 1994).
序列“同一性”指,當對兩條序列進行最佳比對時,必要時引入間隙,以獲取最大序列同一性百分比,且不將任何保守性取代視為序列同一性的一部分,兩條序列的胺基酸/核酸在等價位置相同的程度(百分比)。為測定序列同一性百分比,比對可以藉由屬本領域技術的範圍內的多種方式來實現,例如使用公開可得到的計算機軟體,諸如BLAST、BLAST-2、ALIGN、ALIGN-2或 Megalign(DNASTAR)軟體。所屬技術領域具有通常知識者可確定適用於測量比對的參數,包括在所比較的序列全長上達成最大比對所需的任何算法。 Sequence "identity" means that when two sequences are optimally aligned, gaps are introduced as necessary to obtain the maximum percent sequence identity, and any conservative substitutions are not considered to be part of the sequence identity, the two sequences The degree (percent) of amino acids/nucleic acids at equivalent positions identical. To determine percent sequence identity, alignment can be accomplished in a variety of ways that are within the skill in the art, for example, using publicly available computer software such as BLAST, BLAST-2, ALIGN, ALIGN-2 or Megalign (DNASTAR) software. One of ordinary skill in the art can determine parameters suitable for measuring alignment, including any algorithms needed to achieve maximal alignment over the full length of the sequences being compared.
“保守修飾的變體”或“保守性取代”指使用具有相似特徵(例如,電荷、側鏈尺寸、親水性/疏水性、骨架構型和剛性等)的其他胺基酸置換蛋白中的胺基酸,使得通常可以做出這樣的變化而不改變蛋白的生物活性。所屬技術領域具有通常知識者知曉,在通常情況下,在多肽的非必需區域中的單胺基酸置換基本上不改變生物活性(參見例如,Watson等,(1987)Molecular Biology of the Gene,The Benjamin/Cummings Pub.Co.,p.224(4th Ed.))。此外,結構或功能上類似的胺基酸的置換不太可能破壞生物活性。示例性的保守性置換如下表中所示。 "Conservatively modified variants" or "conservative substitutions" refer to the replacement of amines in a protein with other amino acids with similar characteristics (eg, charge, side chain size, hydrophilicity/hydrophobicity, backbone structure, rigidity, etc.) base acid, so that such changes can often be made without altering the biological activity of the protein. It is known to those of ordinary skill in the art that, in general, monoamino acid substitutions in non-essential regions of polypeptides do not substantially alter biological activity (see, eg, Watson et al., (1987) Molecular Biology of the Gene, The Benjamin/Cummings Pub. Co., p. 224 (4th Ed.)). Furthermore, substitution of structurally or functionally similar amino acids is unlikely to disrupt biological activity. Exemplary conservative substitutions are shown in the table below.
術語“保守修飾的變體”當適用核酸序列時,保守修飾的變體是指那些編碼相同或基本上相同的胺基酸序列的核酸,或在該核酸不編碼胺基酸序列的情況下,是指基本相同的序列。由於遺傳密碼的簡並性,任何給定的蛋白質均可以由多個功能相同的核酸編碼。例如,密碼子GCA、GCC、GCG和GCU都編碼胺基酸丙胺酸。因此,在密碼子指定丙胺酸的每個位置,該密碼子可以改變為任何該相應密碼子而不改變編碼的多肽。此類核酸變異是“沉默變異”,它們是保守修飾變異中的一種。本文中編碼多肽的每個核酸序列也描述了核酸的每種可能的沉默變異。所屬技術領域具有通常知識者將認識到,核酸中的每個密碼子(除了AUG--通常是甲硫胺酸的唯一密碼子;和TGG--通常是色胺酸的唯一密碼子)均可以被修飾以產生功能相同的分子。因此,在每個該序列中均隱含了編碼多肽的核酸的每一種沉默變異。 The term "conservatively modified variant" when applied to nucleic acid sequences refers to those nucleic acids encoding the same or substantially the same amino acid sequence, or in the case of the nucleic acid not encoding an amino acid sequence, refers to substantially the same sequence. Due to the degeneracy of the genetic code, any given protein can be encoded by multiple functionally identical nucleic acids. For example, the codons GCA, GCC, GCG, and GCU all encode the amino acid alanine. Thus, at each position where a codon specifies an alanine, the codon can be changed to any of the corresponding codons without changing the encoded polypeptide. Such nucleic acid variations are "silent variations," which are one type of conservatively modified variation. Every nucleic acid sequence herein that encodes a polypeptide also describes every possible silent variation of the nucleic acid. One of ordinary skill in the art will recognize that every codon in a nucleic acid (except AUG--usually the only codon for methionine; and TGG--usually the only codon for tryptophan) can be are modified to produce functionally equivalent molecules. Thus, within each such sequence is implied every silent variation of the nucleic acid encoding the polypeptide.
術語“載體”意指能夠轉運與其連接的另一多核苷酸的多核苷酸分子。一種類型的載體是“質粒”,其是指環狀雙鏈DNA環,其中可以連接附加的DNA區段。另一種類型的載體是病毒載體,例如腺相關病毒載體(AAV或AAV2),其中另外的DNA區段可以連接到病毒基因組中。某些載體能夠在引入它們的宿主細胞中自主複製(例如,具有細菌複製起點的細菌載體和附加型哺乳動物載體)。其他載體(例如,非附加型哺乳動物載體)可以在引入宿主細胞中後整合到宿主細胞的基因組中,從而與宿主基因組一起複製。 The term "vector" means a polynucleotide molecule capable of transporting another polynucleotide to which it is linked. One type of vector is a "plasmid," which refers to a circular double-stranded DNA loop into which additional DNA segments can be ligated. Another type of vector is a viral vector, such as an adeno-associated viral vector (AAV or AAV2), in which additional DNA segments can be ligated into the viral genome. Certain vectors are capable of autonomous replication in the host cell into which they are introduced (eg, bacterial vectors with bacterial origins of replication and episomal mammalian vectors). Other vectors (eg, non-episomal mammalian vectors) can integrate into the genome of the host cell upon introduction into the host cell, thereby replicating together with the host genome.
術語“表達載體”或“表達構建體”是指適用於對宿主細胞進行轉化且含有指導及/或控制(連同宿主細胞一起)與其可操作地連接的一個或多個異源編碼區的表達的核酸序列的載體。表達構建體可以包括但不限於影響或控制轉錄、翻譯且在存在內含子時影響與其可操作地連接的編碼區的RNA剪接的序列。 The term "expression vector" or "expression construct" refers to a device suitable for transforming a host cell and containing one or more heterologous coding regions that direct and/or control (along with the host cell) the expression of which is operably linked to it. Vectors of nucleic acid sequences. Expression constructs can include, but are not limited to, sequences that affect or control transcription, translation, and when introns are present, RNA splicing of the coding region to which they are operably linked.
如本文中所使用,“可操作地連接”意指該術語所適用的組分呈允許其在適合條件下執行其固有功能的關係。舉例而言,載體中與蛋白質編碼序列“可操作地連接”的控制序列是與其連結,從而在與該控制序列的轉錄活性兼容的條件下達成該蛋白質編碼序列的表達。 As used herein, "operably linked" means that the components to which the term applies are in a relationship that allows them to perform their inherent functions under suitable conditions. For example, a control sequence in a vector that is "operably linked" to a protein-coding sequence is linked thereto such that expression of the protein-coding sequence is achieved under conditions compatible with the transcriptional activity of the control sequence.
術語“宿主細胞”,“宿主細胞系”和“宿主細胞培養物”可互換使用,並且指已經導入外源核酸的細胞,包括此類細胞的後代。宿主細胞包括“轉化體”和“經轉化的細胞”,其包括原代的經轉化的細胞及自其衍生的後代而不考慮傳代的次數。後代在核酸內容物上可以與親本細胞不完全相同,而是可以含有突變。本文中包括具有與在初始轉化細胞中篩選或選擇的相同功能或生物學活性的突變體後代。宿主細胞包括原核和真核宿主細胞,其中真核宿主細胞包括但不限於哺乳動物細胞、昆蟲細胞系(例如,草地貪夜蛾(Spodoptera frugiperda)或粉紋夜蛾(Trichoplusia ni))、兩棲動物細胞、細菌細胞、植物細胞和真菌細胞。哺乳動物宿主細胞包括人、小鼠、大鼠、犬、猴、豬、山羊、牛、馬和倉鼠細胞,包括但不限於中國倉鼠卵巢(CHO)細胞、NSO、SP2細胞、HeLa細胞、幼倉鼠腎(BHK)細胞、猴腎細胞(COS)、人肝細胞癌細胞(例如,Hep G2)、A549細胞、3T3細胞和HEK-293細胞。真菌細胞包括酵母和絲狀真菌細胞,包括例如巴氏畢赤酵母(Pichiapastoris)、芬蘭畢赤酵母(Pichia finlandica)、海藻畢赤酵母(Pichia trehalophila)、科克拉馬畢赤酵母(Pichia koclamae)、膜狀畢赤酵母(Pichia membranaefaciens)、小畢赤酵母(Pichia minuta)(Ogataea minuta、Pichia lindneri)、仙人掌畢赤酵母(Pichiaopuntiae)、耐熱畢赤酵母(Pichia thermotolerans)、柳畢赤酵母(Pichia salictaria)、Pichia guercuum、皮傑普畢赤酵母(Pichia pijperi)、具柄畢赤酵母(Pichia stiptis)、甲醇畢赤酵母(Pichia methanolica)、畢赤酵母屬、釀酒酵母 (Saccharomycescerevisiae)、釀酒酵母屬、多形漢遜酵母(Hansenula polymorpha)、克魯維酵母屬、乳酸克魯維酵母(Kluyveromyces lactis)、白色念珠菌(Candida albicans)、構巢麯黴(Aspergillus nidulans)、黑麯黴(Aspergillus niger)、米麯黴(Aspergillus oryzae)、裡氏木黴(Trichoderma reesei)、勒克氏菌(Chrysosporium lucknowense)、鐮刀菌屬(Fusarium sp.)、禾穀鐮刀菌(Fusarium gramineum)、菜鐮刀菌(Fusarium venenatum)、小立碗蘚(Physcomitrella patens)和粗糙脈孢菌(Neurospora crassa)。畢赤酵母屬、任何釀酒酵母屬、多形漢遜酵母(Hansenula polymorpha)、任何克魯維酵母屬、白色念珠菌(Candida albicans)、任何麯黴屬、裡氏木黴(Trichoderma reesei)、勒克黴菌(Chrysosporium lucknowense)、任何鐮刀菌屬、解脂耶氏酵母(Yarrowia lipolytica)和粗糙脈孢菌(Neurospora crassa)。在一些實施方案中,該宿主細胞是非人細胞。 The terms "host cell", "host cell line" and "host cell culture" are used interchangeably and refer to cells into which exogenous nucleic acid has been introduced, including the progeny of such cells. Host cells include "transformants" and "transformed cells," which include the primary transformed cell and progeny derived therefrom without regard to the number of passages. The progeny may not be identical in nucleic acid content to the parent cell, but may contain mutations. Mutant progeny that have the same function or biological activity as screened or selected in the original transformed cell are included herein. Host cells include prokaryotic and eukaryotic host cells, wherein eukaryotic host cells include but are not limited to mammalian cells, insect cell lines (eg, Spodoptera frugiperda or Trichoplusia ni), amphibians cells, bacterial cells, plant cells and fungal cells. Mammalian host cells include human, mouse, rat, canine, monkey, porcine, goat, bovine, equine and hamster cells, including but not limited to Chinese hamster ovary (CHO) cells, NSO, SP2 cells, HeLa cells, baby hamsters Kidney (BHK) cells, monkey kidney cells (COS), human hepatocellular carcinoma cells (eg, Hep G2), A549 cells, 3T3 cells and HEK-293 cells. Fungal cells include yeast and filamentous fungal cells including, for example, Pichia pastoris, Pichia finlandica, Pichia trehalophila, Pichia koclamae, Pichia membranaefaciens, Pichia minuta (Ogataea minuta, Pichia lindneri), Pichia puntiae, Pichia thermotolerans, Pichia salictaria ), Pichia guercuum, Pichia pijperi, Pichia stiptis, Pichia methanolica, Pichia, Saccharomyces cerevisiae (Saccharomycescerevisiae), Saccharomyces cerevisiae, Hansenula polymorpha, Kluyveromyces, Kluyveromyces lactis, Candida albicans, Aspergillus nidulans, Aspergillus niger, Aspergillus oryzae, Trichoderma reesei, Chrysosporium lucknowense, Fusarium sp., Fusarium gramineum, Fusarium venenatum, Physcomitrella patens and Neurospora crassa. Pichia, any Saccharomyces cerevisiae, Hansenula polymorpha, any Kluyveromyces, Candida albicans, any Aspergillus, Trichoderma reesei, Lux Mold (Chrysosporium lucknowense), any Fusarium spp., Yarrowia lipolytica and Neurospora crassa. In some embodiments, the host cell is a non-human cell.
如在本申請中所使用的,表述“細胞”、“細胞系”和“細胞培養物”可以互換使用,並且所有這樣的名稱均包括子代。因而,詞語“轉化體”和“轉化的細胞”包括原代受試者細胞和來源於其的培養物,而與傳代的次數無關。還應理解的是,由於有意或無意的突變,使得並非所有子代均具有完全相同的DNA內容物。包括與篩選出其的原始轉化細胞具有相同功能或生物活性的突變子代。 As used in this application, the expressions "cell", "cell line" and "cell culture" are used interchangeably and all such designations include progeny. Thus, the words "transformants" and "transformed cells" include primary subject cells and cultures derived therefrom, regardless of the number of passages. It will also be appreciated that not all progeny will have exactly the same DNA content due to intentional or unintentional mutations. Mutant progeny that have the same function or biological activity as the original transformed cells from which they were screened are included.
“視需要”或“視需要地”意味著隨後所描述地事件或環境可以但不必發生,該說明包括該事件或環境發生或不發生的場合。 "Optional" or "optionally" means that the subsequently described event or circumstance can but need not occur, and that the description includes instances where the event or circumstance does or does not occur.
“醫藥組成物”表示含有一種或多種本文所述的抗體或其抗原結合片段與其他化學組分的混合物,該其他組分例如生理學/可藥用的載體和賦形劑。 "Pharmaceutical composition" means a mixture comprising one or more of the antibodies or antigen-binding fragments thereof described herein and other chemical components, such as physiological/pharmaceutically acceptable carriers and excipients.
術語“藥學上可接受的載體”意指生理學上相容的任何溶劑、分散介質、塗層、抗細菌和抗真菌劑、等滲和吸收增強或延遲劑等。藥學上可接受的 載體的一些實例為水、鹽水、磷酸鹽緩衝鹽水、具有氯化鈉的乙酸鹽緩衝液、右旋糖、甘油、聚乙二醇、乙醇等以及其組合。在許多情況下,組成物中較佳包含等滲劑,例如糖、多元醇(例如甘露醇、山梨糖醇)或氯化鈉。藥學上可接受的物質的其他實例為表面活性劑、濕潤劑或少量輔助物質例如濕潤劑或乳化劑、防腐劑或緩衝劑,其增強抗體的保質期或有效性。 The term "pharmaceutically acceptable carrier" means any solvent, dispersion medium, coating, antibacterial and antifungal agents, isotonic and absorption enhancing or delaying agents, and the like that are physiologically compatible. pharmaceutically acceptable Some examples of carriers are water, saline, phosphate buffered saline, acetate buffer with sodium chloride, dextrose, glycerol, polyethylene glycol, ethanol, and the like, and combinations thereof. In many cases, it is preferred to include isotonic agents such as sugars, polyols (eg, mannitol, sorbitol) or sodium chloride in the composition. Other examples of pharmaceutically acceptable substances are surfactants, wetting agents or minor amounts of auxiliary substances such as wetting or emulsifying agents, preservatives or buffers, which enhance the shelf-life or effectiveness of the antibody.
本揭露的醫藥組成物可藉由本領域已知的各種方法施與。施與途徑和/或方式根據所希望的結果而變化。較佳地,施與可以是玻璃體內、靜脈內、肌肉內、腹膜內或皮下或在靶標部位附近施與。藥學上可接受的載體應適合於玻璃體內、靜脈內、肌肉內、皮下、腸胃外、脊柱或表皮施與(例如,藉由注射或輸注)。根據施與途徑,活性化合物(即抗體,雙特異性和多特異性分子)可以包被在材料中以保護化合物免受酸和可能使化合物失活的其他自然條件的作用。 The pharmaceutical compositions of the present disclosure can be administered by various methods known in the art. The route and/or mode of administration will vary depending on the desired result. Preferably, administration may be intravitreal, intravenous, intramuscular, intraperitoneal or subcutaneous or near the target site. A pharmaceutically acceptable carrier should be suitable for intravitreal, intravenous, intramuscular, subcutaneous, parenteral, spinal or epidermal administration (eg, by injection or infusion). Depending on the route of administration, the active compound (ie, antibodies, bispecific and multispecific molecules) can be coated in materials to protect the compound from the action of acids and other natural conditions that may inactivate the compound.
術語“受試者”包括人類和非人類動物。非人動物包括所有脊椎動物(例如哺乳動物和非哺乳動物)例如非人靈長類(例如,食蟹猴)、綿羊、狗、牛、雞、兩棲動物和爬行動物。除非指出時,否則該術語“患者”或“受試者”在本文中可互換地使用。如本文所使用的,術語“食蟹猴(cyno)”或“食蟹猴(cynomolgus)”是指食蟹猴(Macaca fascicularis)。在某些實施方案中,個體或受試者是人。 The term "subject" includes humans and non-human animals. Non-human animals include all vertebrates (eg, mammals and non-mammals) such as non-human primates (eg, cynomolgus monkeys), sheep, dogs, cows, chickens, amphibians, and reptiles. Unless indicated, the terms "patient" or "subject" are used interchangeably herein. As used herein, the term "cyno" or "cynomolgus" refers to cynomolgus monkey (Macaca fascicularis). In certain embodiments, the individual or subject is a human.
“施用”、“給予”和“處理”,當其應用於動物、人、實驗受試者、細胞、組織、器官或生物流體時,是指外源性藥物、治療劑、診斷劑或組成物與動物、人、受試者、細胞、組織、器官或生物流體的接觸。 "Administering," "administering," and "treating," when applied to animals, humans, experimental subjects, cells, tissues, organs, or biological fluids, refer to exogenous drugs, therapeutic agents, diagnostic agents, or compositions Contact with animals, humans, subjects, cells, tissues, organs or biological fluids.
“樣本”是指從受試者分離的類似流體、細胞、或組織的採集物,以及存在於受試者體內的流體、細胞或組織。示例性樣本為生物流體,諸如血液、血清和漿膜液、血漿、淋巴液、尿液、唾液、囊液、淚液、排泄物、痰、分泌組 織和器官的黏膜分泌物、陰道分泌物、腹水、胸膜、心包、腹膜、腹腔和其它體腔的流體、由支氣管灌洗液收集的流體、滑液、與受試者或生物來源接觸的液體溶液,例如細胞和器官培養基(包括細胞或器官條件培養基)、灌洗液等,組織活檢樣本、細針穿刺、手術切除的組織、器官培養物或細胞培養物。 "Sample" refers to a collection of similar fluids, cells, or tissues isolated from a subject, as well as fluids, cells, or tissues present in a subject. Exemplary samples are biological fluids such as blood, serum and serous fluid, plasma, lymph, urine, saliva, cystic fluid, tears, feces, sputum, secretome Mucosal secretions of tissues and organs, vaginal secretions, ascites, pleura, pericardium, peritoneum, fluids in the abdominal cavity and other body cavities, fluids collected from bronchial lavage, synovial fluid, fluid solutions in contact with subjects or biological sources , such as cell and organ culture media (including cell or organ conditioned media), lavage fluid, etc., tissue biopsy samples, fine needle aspiration, surgically resected tissue, organ cultures or cell cultures.
“治療/處理”(及其語法變型)指試圖改變所治療個體的天然過程的臨床干預,並且可以為了預防或者在臨床病理學的過程期間實施。治療的期望效果包括但不限於預防疾病的發生或再發生,減輕症狀,減輕/減少疾病的任何直接或間接病理後果,預防轉移,降低疾病進展速率,改善或減輕疾病狀態,和消退或改善的預後。在一些實施方案中,使用本揭露的抗體來延遲疾病的形成或減緩疾病的進展。 "Treatment/treatment" (and grammatical variants thereof) refers to clinical interventions that attempt to alter the natural course of the individual being treated, and may be performed for prophylaxis or during the course of clinical pathology. Desired effects of treatment include, but are not limited to, preventing the occurrence or recurrence of the disease, alleviating symptoms, alleviating/reducing any direct or indirect pathological consequences of the disease, preventing metastasis, reducing the rate of disease progression, ameliorating or alleviating the disease state, and remission or amelioration of Prognosis. In some embodiments, the antibodies of the present disclosure are used to delay the development or slow the progression of a disease.
“有效量”一般是足以降低症狀的嚴重程度及/或頻率、消除這些症狀及/或潛在病因、預防症狀及/或其潛在病因出現及/或改良或改善由疾病狀態引起或與其相關的損傷(例如肺病)的量。在一些實施例中,有效量是治療有效量或預防有效量。“治療有效量”是足以治療疾病狀態或症狀、尤其與該疾病狀態相關的狀態或症狀,或者以其他方式預防、阻礙、延遲或逆轉該疾病狀態或以任何方式與該疾病相關的任何其他不理想症狀的進展的量。“預防有效量”是當給予受試者時將具有預定預防效應,例如預防或延遲該疾病狀態的發作(或復發),或者降低該疾病狀態或相關症狀的發作(或復發)可能性的量。完全治療或預防效應未必因給予一個劑量便發生,而且可能僅在給予一系列劑量之後發生。因而,治療或預防有效量可以一次或多次給予的方式給予。“治療有效量”和“預防有效量”可取決於以下因素變化:諸如個體的疾病狀態、年齡、性別和體重,以及治療劑或治 療劑組合在個體中引發期望的應答的能力。有效治療劑或治療劑組合的示例性指標包括例如患者改善的健康狀況。 An "effective amount" is generally sufficient to reduce the severity and/or frequency of symptoms, eliminate those symptoms and/or underlying causes, prevent the appearance of symptoms and/or their underlying causes, and/or ameliorate or ameliorate impairments caused by or associated with a disease state (eg lung disease). In some embodiments, the effective amount is a therapeutically effective amount or a prophylactically effective amount. A "therapeutically effective amount" is sufficient to treat a disease state or symptom, particularly a state or symptom associated with the disease state, or to otherwise prevent, retard, delay or reverse the disease state or any other irreversible disorder in any way associated with the disease state Amount of progression of desired symptoms. A "prophylactically effective amount" is an amount that, when administered to a subject, will have a predetermined preventive effect, such as preventing or delaying the onset (or recurrence) of the disease state, or reducing the likelihood of the onset (or recurrence) of the disease state or associated symptoms . A complete therapeutic or prophylactic effect does not necessarily occur with the administration of a single dose, and may occur only after a series of doses have been administered. Thus, a therapeutically or prophylactically effective amount can be administered in one or more administrations. A "therapeutically effective amount" and a "prophylactically effective amount" may vary depending on factors such as the individual's disease state, age, sex, and weight, as well as the therapeutic or therapeutic agent. The ability of a combination of therapeutic agents to elicit a desired response in an individual. Exemplary indicators of an effective therapeutic agent or combination of therapeutic agents include, for example, improved health status in a patient.
術語“癌(症)”和“癌(性)的”指向或描述哺乳動物中典型的以不受調節的細胞生長為特徵的生理疾患。此定義中包括良性和惡性癌症。“早期癌症”或“早期腫瘤”指非侵入性的或轉移性的,或者歸為0期、I期、或II期癌症的癌症。癌症的例子包括但不限於淋巴瘤、母細胞瘤(包括髓母細胞瘤和視網膜母細胞瘤)、肉瘤(包括脂肪肉瘤和滑膜細胞肉瘤)、神經內分泌腫瘤(包括類癌瘤、胃泌素瘤和胰島細胞癌)、間皮瘤、施旺氏細胞瘤(包括聽神經瘤)、腦膜瘤、腺癌、黑素瘤、腎上腺腫瘤、和白血病或淋巴樣惡性腫瘤。此類癌症的更具體例子包括但不限於鱗狀細胞癌(例如上皮鱗狀細胞癌);肺癌包括小細胞肺癌(SCLC)、非小細胞肺癌(NSCLC)、肺的腺癌和肺的鱗癌;腹膜癌;肝細胞癌;胃癌(gastric or stomach cancer)包括胃腸癌;胰腺癌;成膠質細胞瘤;宮頸癌;卵巢癌;肝癌(liver cancer or hepatic carcinoma)、膀胱癌;肝瘤(hepatoma);乳腺癌(包括轉移性乳腺癌);結腸癌;直腸癌;結腸直腸癌;子宮內膜癌或子宮癌;唾液腺癌;腎癌(kidney or renal cancer);前列腺癌;外陰癌;甲狀腺癌;肛門癌;陰莖癌;睾丸癌;食道癌;膽管腫瘤;輸卵管癌;卵巢癌;膽管癌;膀胱癌;胰腺癌;皮膚癌;及頭和頸癌和多發性骨髓瘤。
The terms "cancer" and "cancerous" refer to or describe a physiological disorder that is typically characterized by unregulated cell growth in mammals. Benign and malignant cancers are included in this definition. "Early stage cancer" or "early stage tumor" refers to cancer that is either non-invasive or metastatic, or classified as
實施例和測試例Examples and Test Cases
以下結合實施例和測試例進一步描述本揭露,但這些實施例和測試例並非限制著本揭露的範圍。本揭露實施例或測試例中未註明具體條件的實驗方法,通常按照常規條件,如冷泉港的抗體技術實驗手冊,分子選殖手冊;或按照原料或商品製造廠商所建議的條件;未註明具體來源的試劑材料,為市場購買獲得。 The present disclosure is further described below with reference to the embodiments and test examples, but these embodiments and test examples do not limit the scope of the present disclosure. The experimental methods for which specific conditions are not specified in the examples or test examples of the present disclosure are usually based on conventional conditions, such as the Antibody Technology Experiment Manual of Cold Spring Harbor, and the molecular colonization manual; or according to the conditions suggested by the manufacturers of raw materials or commodities; The source reagent materials are purchased from the market.
實施例1. ANG-2和ANG-2受體Tie2的表達Example 1. Expression of ANG-2 and the ANG-2 receptor Tie2
編碼帶人IgG1-Fc標簽的人ANG-2和人ANG-2受體Tie2胞外區序列插入phr載體中,構建成表達質粒,然後轉染HEK293。具體轉染步驟為:前一天將HEK293E細胞以1×106/mL接種於freestyle表達培養基(含有1% FBS,Gibco,12338-026),放置於37度恆溫搖床(120rpm)繼續培養24小時。24小時後,將轉染質粒和轉染試劑PEI混合物緩慢加入200mL HEK293E的細胞中,放入8% CO2、120rpm、37℃的搖床中培養。轉染第3天,補充10%體積的補料培養基(20mM葡萄糖+2mM L-谷胺酸)。待轉染第6天,取樣4500rpm離心10分鐘收集細胞上清,將重組的ANG-2和Tie2受體蛋白按照實施例2所述的方法進行純化。
The sequences encoding human ANG-2 with human IgG1-Fc tag and the extracellular domain of human ANG-2 receptor Tie2 were inserted into the phr vector, constructed into an expression plasmid, and then transfected into HEK293. The specific transfection steps are as follows: the day before, HEK293E cells were inoculated in freestyle expression medium (containing 1% FBS, Gibco, 12338-026) at 1×10 6 /mL, and placed in a constant temperature shaker (120 rpm) at 37 degrees for 24 hours. . After 24 hours, the mixture of transfection plasmid and transfection reagent PEI was slowly added to 200 mL HEK293E cells, and cultured in a shaker at 8% CO 2 , 120 rpm, and 37°C. On
其中人ANG-2胺基酸序列如SEQ ID NO:1所示,Tie2胞外區Fc融合蛋白胺基酸序列如SEQ ID NO:2所示。 The amino acid sequence of human ANG-2 is shown in SEQ ID NO: 1, and the amino acid sequence of Tie2 extracellular domain Fc fusion protein is shown in SEQ ID NO: 2.
相關序列如下所示: The relevant sequence looks like this:
(1)帶人Fc標簽的人ANG-2胺基酸序列(huANG-2-Fc) (1) Amino acid sequence of human ANG-2 with human Fc tag (huANG-2-Fc)
SEQ ID NO:1 SEQ ID NO: 1
註釋:横線部分為ANG-2蛋白全常序列,點線為接頭,斜體部分為人IgG1Fc標簽。 Note: The horizontal line part is the full normal sequence of ANG-2 protein, the dotted line is the linker, and the italic part is the human IgG1Fc tag.
(2)帶人Fc標簽的Tie2胺基酸序列(huTie2-Fc) (2) Tie2 amino acid sequence with human Fc tag (huTie2-Fc)
SEQ ID NO:2 SEQ ID NO: 2
註釋:下劃線部分為Tie2的胞外區,斜體部分為人IgG1 Fc標簽。 Note: The underlined part is the extracellular region of Tie2, and the italicized part is the human IgG1 Fc tag.
實施例2. Protein A親和層析純化帶Fc標簽的重組蛋白或抗體Example 2. Protein A affinity chromatography for purification of Fc-tagged recombinant proteins or antibodies
將表達抗體或huANG-2-Fc、huTie2-Fc的細胞上清樣品高速離心去除雜質,藉由Protein A管柱進行純化。用PBS沖洗管柱,至A280讀數降至基線。用100mM乙酸pH 3.5沖提目的蛋白,用1M Tris-HCl,pH8.0中和。沖提樣品適當濃縮後利用PBS平衡好的凝膠層析Superdex200(GE)進一步純化,將得到的蛋白經電泳,肽圖,LC-MS鑑定為正確後分裝備用。 The supernatant samples of cells expressing antibodies or huANG-2-Fc and huTie2-Fc were centrifuged at high speed to remove impurities, and purified by Protein A column. The column was flushed with PBS until the A280 reading dropped to baseline. The protein of interest was extracted with 100 mM acetic acid pH 3.5 and neutralized with 1 M Tris-HCl, pH 8.0. The washed samples were properly concentrated and further purified by gel chromatography Superdex200 (GE) equilibrated with PBS, and the obtained proteins were identified as correct by electrophoresis, peptide map, and LC-MS, and then separated for use.
實施例3. 表達重組ANG-2受體Tie2的細胞系的構建和鑑定Example 3. Construction and identification of cell lines expressing recombinant ANG-2 receptor Tie2
為篩選有功能的抗體,本揭露構建了表達Tie2的CHO-K1/Tie2細胞株。 To screen for functional antibodies, the present disclosure constructed a CHO-K1/Tie2 cell line expressing Tie2.
將人Tie2全長基因選殖到哺乳動物細胞表達載體pBABE上,用pVSV-G,pGag-pol和pBABE-Tie2三種質粒共同轉染HEK293T細胞(ATCC,CRL-3216)包裝病毒,轉染48小時後,收集病毒感染CHOK1細胞(ATCC,CRL-9618)。感染72小時後用10μg/mL嘌呤黴素加壓篩選,待純株團擴增生長後,消化細胞用FACS檢測表達量,陽性率約40%,然後分選單純株細胞,得到表達人Tie2的單株1B11。 The full-length human Tie2 gene was cloned into the mammalian cell expression vector pBABE, and the three plasmids pVSV-G, pGag-pol and pBABE-Tie2 were used to co-transfect HEK293T cells (ATCC, CRL-3216) to package the virus, and 48 hours after transfection , the virus was collected to infect CHOK1 cells (ATCC, CRL-9618). After 72 hours of infection, 10 μg/mL puromycin was used for pressure screening. After the pure strain was expanded and grown, the expression level of the digested cells was detected by FACS, and the positive rate was about 40%. Then the pure strain cells were sorted to obtain the expression of human Tie2. Individual plant 1B11.
實施例4. 抗人ANG-2融合瘤抗體的篩選和鑑定Example 4. Screening and identification of anti-human ANG-2 fusion tumor antibodies
本揭露藉由融合瘤技術製備了針對人ANG-2的單株抗體,所得抗體與人ANG-2以較高的親和力,並且可以與食蟹猴ANG-2有交叉反應,能夠阻斷ANG-2與其受體結合,且可以抑制ANG-2誘導的Tie2的磷酸化。 In the present disclosure, a monoclonal antibody against human ANG-2 has been prepared by fusion tumor technology. 2 binds to its receptor and can inhibit ANG-2-induced phosphorylation of Tie2.
用重組蛋白huANG-2-Fc(100/50/50μg),與TiterMax/Alum/CpG佐劑免疫SJL小鼠。ANG-2的特異性免疫反應藉由檢測血清效價的ELISA和配體受體阻斷實驗來測定。選取有較好特異性免疫反應的小鼠,處死後,取脾細胞,與骨髓瘤細胞融合。 SJL mice were immunized with recombinant protein huANG-2-Fc ( 100/50/50 μg) with TiterMax/Alum/CpG adjuvant. Specific immune responses to ANG-2 were determined by ELISA and ligand receptor blockade assays for serum titers. Mice with better specific immune response were selected, and after being sacrificed, spleen cells were taken and fused with myeloma cells.
初次篩選用針對人和鼠ANG-2的ELISA結合實驗,阻斷人ANG-2結合其受體Tie2的實驗進行篩選。當將融合瘤細胞轉移到24孔板後,用針對人和鼠ANG-2的ELISA結合實驗,基於ELISA的阻斷ANG-2結合其受體Tie2的受體阻斷實驗對其細胞上清進行複篩。篩選出的陽性純株經過兩輪亞選殖後,得到融合瘤純株,用於抗體生產,並藉由親和方法純化。 The primary screening was performed with an ELISA binding assay for human and murine ANG-2, an assay that blocks binding of human ANG-2 to its receptor Tie2. After the fusion tumor cells were transferred to 24-well plates, the cell supernatants were subjected to an ELISA binding assay against human and murine ANG-2, an ELISA-based receptor blocking assay that blocks ANG-2 binding to its receptor Tie2 Rescreen. After two rounds of sub-selection, the screened positive clones were used to obtain pure fusion tumor clones, which were used for antibody production and purified by affinity method.
篩選出活性好的單株融合瘤細胞株HR54和CP33,分別收集對數生長期融合瘤細胞,用NucleoZol(MN)提取RNA(按照試劑盒說明書步驟),並進行反轉錄(PrimeScriptTM Reverse Transcriptase,Takara,cat # 2680A)。將反轉錄得到的cDNA採用鼠Ig-Primer Set(Novagen,TB326 Rev.B 0503)進行PCR擴增後送測序公司測序。經測序得到鼠源抗ANG-2抗體:HR54和CP33,其可變區胺基酸序列如下: The single-clone fusion tumor cell lines HR54 and CP33 with good activity were screened, and the logarithmic growth phase fusion tumor cells were collected respectively, RNA was extracted with NucleoZol (MN) (following the steps of the kit instructions), and reverse transcription (PrimeScript TM Reverse Transcriptase, Takara) was performed. , cat # 2680A). The cDNA obtained by reverse transcription was amplified by PCR using mouse Ig-Primer Set (Novagen, TB326 Rev. B 0503) and then sent to a sequencing company for sequencing. The mouse-derived anti-ANG-2 antibodies were obtained by sequencing: HR54 and CP33, and the amino acid sequences of their variable regions are as follows:
>HR54鼠源重鏈可變區序列: >HR54 murine heavy chain variable region sequence:
SEQ ID NO:3 SEQ ID NO: 3
>HR54鼠源輕鏈可變區序列: >HR54 murine light chain variable region sequence:
SEQ ID NO:4 SEQ ID NO: 4
>CP33鼠源重鏈可變區序列: >CP33 murine heavy chain variable region sequence:
SEQ ID NO:5 SEQ ID NO: 5
>CP33鼠源輕鏈可變區序列: >CP33 murine light chain variable region sequence:
SEQ ID NO:6 SEQ ID NO: 6
其中,劃線表示CDR,斜體部分表示連接子,其餘部分為FR。 The dashed lines represent CDRs, the italicized parts represent linkers, and the rest are FRs.
根據Kabat編號規則獲得的CDR區胺基酸序列如下表所示: The amino acid sequences of the CDR regions obtained according to the Kabat numbering rules are shown in the following table:
將上述鼠源抗體的輕重鏈可變區與人源抗體的輕、重鏈恆定區(如SEQ ID NO:47所示的重鏈恆定區和如SEQ ID NO:48所示的輕鏈恆定區)連接後形成嵌合抗體,HR54純株對應的嵌合抗體命名為CHR54,其他抗體類推。 The light and heavy chain variable regions of the above-mentioned murine antibodies were combined with the light and heavy chain constant regions of human antibodies (heavy chain constant regions shown in SEQ ID NO: 47 and light chain constant regions shown in SEQ ID NO: 48) ) is connected to form a chimeric antibody, the chimeric antibody corresponding to the HR54 pure strain is named CHR54, and other antibodies are analogous.
實施例5. 抗人ANG-2單株抗體的人源化Example 5. Humanization of anti-human ANG-2 monoclonal antibody
鼠源單株抗體人源化,可以根據本領域許多文獻公示的方法進行。簡言之,在所獲得的鼠源抗體VH/VL CDR典型結構的基礎上,從人源germline數據庫中搜索輕鏈可變區(VL)和重鏈可變區(VH)的同源序列,選取FR同源性高的germline作為模板,將鼠源抗體的CDR區移植到人源模板上,並對FR區的某些 胺基酸進行回復突變,將鼠源抗體的恆定區替換為人恆定區,得到最終的人源化分子。 Humanization of murine monoclonal antibodies can be carried out according to the methods disclosed in many documents in the art. Briefly, on the basis of the obtained murine antibody VH/VL CDR canonical structures, the homologous sequences of the light chain variable region (VL) and heavy chain variable region (VH) were searched from the human germline database, The germline with high FR homology was selected as the template, the CDR region of the mouse antibody was transplanted to the human template, and some parts of the FR region were transplanted. The amino acid is backmutated to replace the constant region of the murine antibody with the human constant region to obtain the final humanized molecule.
5.1 HR54鼠源抗體的人源化5.1 Humanization of HR54 murine antibody
HR54的人源化VH模板為IGHV3-7*01,人源化VL的模板為IGKV4-1*01,將HR54鼠源抗體的CDR移植到人源模板上,並對框架區胺基酸進行回復突變,具體回復突變如下表: The humanized VH template of HR54 is IGHV3-7*01, and the humanized VL template is IGKV4-1*01. The CDRs of HR54 mouse antibody are grafted onto the human template, and the amino acids in the framework region are restored. Mutations, specific reversion mutations are as follows:
註:Grafted代表鼠抗體CDR植入人種系FR區序列,N84S表示依照Kabat編號系統,將84位N突變回S。 Note: Grafted represents the CDR of murine antibody implanted into the human germline FR region sequence, N84S represents the mutation of the 84th N back to S according to the Kabat numbering system.
鼠源抗體HR54人源化抗體的可變區序列如下: The variable region sequence of the mouse antibody HR54 humanized antibody is as follows:
>huHR54VL1(hu HR54 VL-CDR grafted) >huHR54VL1(hu HR54VL-CDR grafted)
SEQ ID NO:17 SEQ ID NO: 17
>huHR54VL2 >huHR54VL2
SEQ ID NO:18 SEQ ID NO: 18
>huHR54VL3 >huHR54VL3
SEQ ID NO:19 SEQ ID NO: 19
>huHR54VH1(huHR54 VH-CDR Grafted) >huHR54VH1(huHR54 VH-CDR Grafted)
SEQ ID NO:20 SEQ ID NO: 20
>huHR54VH2 >huHR54VH2
SEQ ID NO:21 SEQ ID NO: 21
其中,劃線表示CDR,斜體部分表示連接子,其餘部分為FR。 The dashed lines represent CDRs, the italicized parts represent linkers, and the rest are FRs.
將上述輕、重鏈可變區與人種系輕鏈、重恆定區序列組合形成最終的完整輕、重鏈序列,進而得到全長序列的抗體。在本揭露中如無明確說明時,形成全長抗體時重鏈可變區與SEQ ID NO:47所示恆定區連接形成抗體重鏈,輕鏈可變區與SEQ ID NO:48所示的恆定區連接形成抗體輕鏈鏈,得到的HR54的人源化抗體(huHR54),不同huHR54抗體的重、輕鏈可變區見下表。 The above-mentioned light and heavy chain variable regions are combined with the human germline light chain and heavy constant region sequences to form the final complete light and heavy chain sequences, and then antibodies with full-length sequences are obtained. Unless otherwise specified in the present disclosure, when forming a full-length antibody, the variable region of the heavy chain is linked with the constant region shown in SEQ ID NO: 47 to form an antibody heavy chain, and the variable region of the light chain is connected with the constant region shown in SEQ ID NO: 48 to form an antibody heavy chain. The regions are connected to form an antibody light chain chain, and the obtained humanized antibody of HR54 (huHR54), the variable regions of the heavy and light chains of different huHR54 antibodies are shown in the following table.
註:huHR54-01抗體,其重鏈可變區為huHR54VH1、輕鏈可變區為huHR54VL1,重鏈恆定區為SEQ ID NO:47,輕鏈恆定區為SEQ ID NO:48,其他類推。 Note: huHR54-01 antibody, its heavy chain variable region is huHR54VH1, light chain variable region is huHR54VL1, heavy chain constant region is SEQ ID NO: 47, light chain constant region is SEQ ID NO: 48, and so on.
藉由ELISA實驗檢測HR54人源化抗體或嵌合抗體與人ANG-2的結合活性。方法如下: The binding activity of HR54 humanized antibody or chimeric antibody to human ANG-2 was detected by ELISA. Methods as below:
用鏈黴親和素(abcam,ab123480),濃度1ng/μL,每孔100μL,4℃過夜包被平板後,除去上清液,加250μL 5%脫脂奶粉37℃封閉1小時,洗板機洗板3遍。加0.5ng/μL生物素-hAng2-His(sinobiologican,10691-H07H),37℃孵育1小時。洗板機洗板3遍,加100μL1:1稀釋的噬菌體上清液,37℃孵育1小時。洗板機洗板3遍,每孔加100μL 1:10000稀釋的抗M13-HRP(GE,27-9421-01),37℃孵育1小時。洗板機洗板3遍,每孔加100μL TMB顯色。5-10分鐘後每孔加100μL 1M H2SO4終止顯色,酶標儀測OD450值。其結果如下表所示:
The plate was coated with streptavidin ( abcam , ab123480 ) at a concentration of 1 ng/μL, 100 μL per well, overnight at 4°C, the supernatant was removed, and 250 μL of 5% nonfat dry milk was added to block for 1 hour at 37°C. Wash the
結果顯示,本揭露中的HR54嵌合抗體和人源化抗體均可與人ANG-2特異性結合。 The results show that both the HR54 chimeric antibody and the humanized antibody in the present disclosure can specifically bind to human ANG-2.
5.2 huHR54人源化抗體的改造5.2 Transformation of huHR54 humanized antibody
對huHR54-04抗體的HCDR2進行D52AE或D53N突變,對HCDR3進行D95N、E96Q、C100S或C100V突變,突變後得到的huHR54-04突變體的CDR序列如下: The HCDR2 of the huHR54-04 antibody was mutated with D52A E or D53N, and the HCDR3 was mutated with D95N, E96Q, C100S or C100V. The CDR sequences of the huHR54-04 mutant obtained after mutation were as follows:
示例性的,huR54的突變後的重鏈可變區如下: Exemplarily, the mutated heavy chain variable region of huR54 is as follows:
>huHR54VH2a >huHR54VH2a
SEQ ID NO:28 SEQ ID NO: 28
>huHR54VH2b >huHR54VH2b
SEQ ID NO:29 SEQ ID NO: 29
>huHR54VH2c >huHR54VH2c
SEQ ID NO:30 SEQ ID NO: 30
>huHR54VH2d >huHR54VH2d
SEQ ID NO:31 SEQ ID NO: 31
>huHR54VH2e >huHR54VH2e
SEQ ID NO:32 SEQ ID NO: 32
>huHR54VH2f >huHR54VH2f
SEQ ID NO:33 SEQ ID NO: 33
>huHR54VH2g >huHR54VH2g
SEQ ID NO:34 SEQ ID NO: 34
>huHR54VH2k >huHR54VH2k
SEQ ID NO:35 SEQ ID NO: 35
>huHR54VH2m >huHR54VH2m
SEQ ID NO:36 SEQ ID NO: 36
>huHR54VH2n >huHR54VH2n
SEQ ID NO:37 SEQ ID NO: 37
其中,劃線表示CDR,斜體部分表示連接子,其餘部分為FR。 The dashed lines represent CDRs, the italicized parts represent linkers, and the rest are FRs.
將上述huHR54-04突變體的重鏈可變區與重鏈恆定區組合形成完整重鏈序列,再與huHR54-04人源化抗體的輕鏈重組,進而得到全長抗體。在本揭露中如無明確說明時,形成全長抗體時重鏈可變區與SEQ ID NO:47所示恆定區連接形成抗體重鏈,輕鏈可變區與SEQ ID NO:48所示的恆定區連接形成抗體輕鏈,得到的人源化抗體的重、輕鏈可變區組合見下表。 The heavy chain variable region and heavy chain constant region of the above-mentioned huHR54-04 mutant were combined to form a complete heavy chain sequence, and then recombined with the light chain of the huHR54-04 humanized antibody to obtain a full-length antibody. Unless otherwise specified in the present disclosure, when forming a full-length antibody, the variable region of the heavy chain is linked with the constant region shown in SEQ ID NO: 47 to form an antibody heavy chain, and the variable region of the light chain is connected with the constant region shown in SEQ ID NO: 48 to form an antibody heavy chain. The regions are connected to form an antibody light chain, and the combination of the heavy and light chain variable regions of the obtained humanized antibody is shown in the following table.
註:huHR54-07抗體,其重鏈可變區為huHR54VH2a、輕鏈可變區為huHR54VL1,重鏈恆定區為SEQ ID NO:47,輕鏈恆定區為SEQ ID NO:48,其他類推。 Note: huHR54-07 antibody, its heavy chain variable region is huHR54VH2a, light chain variable region is huHR54VL1, heavy chain constant region is SEQ ID NO: 47, light chain constant region is SEQ ID NO: 48, and so on.
藉由ELISA實驗檢測huHR54抗體突變體與人ANG-2的結合活性,其結果如下表所示: The binding activity of huHR54 antibody mutants to human ANG-2 was detected by ELISA, and the results are shown in the following table:
結果顯示,在huHR54抗體的HCDR2,HCDR3上的點突變,並不會影響huHR54抗體與人ANG-2的結合活性。 The results showed that point mutations on HCDR2 and HCDR3 of huHR54 antibody did not affect the binding activity of huHR54 antibody to human ANG-2.
huHR54人源化抗體的CDR區可如下表: The CDR regions of the huHR54 humanized antibody can be shown in the following table:
其中X1為D或E,X2為D或N,X3為D或N,X4為E或Q,X5為C,S或V。 Wherein X 1 is D or E, X 2 is D or N, X 3 is D or N, X 4 is E or Q, and X 5 is C, S or V.
5.3 CP33鼠源抗體的人源化5.3 Humanization of CP33 murine antibody
CP33的人源化VH模板為IGHV3-7*01,VL的模板為IGKV4-1*01,將CP33的CDR移植到人源模板上,並對框架區胺基酸進行回復突變,具體回復突變見下表: The humanized VH template of CP33 is IGHV3-7*01, and the VL template is IGKV4-1*01. The CDRs of CP33 are transplanted to the human template, and the amino acids in the framework region are back-mutated. For specific back-mutations, see The following table:
註:Grafted代表鼠抗體CDR植入人種系FR區序列,N84S表示依照Kabat編號系統,將84位N突變回S。 Note: Grafted represents the CDR of murine antibody implanted into the human germline FR region sequence, N84S represents the mutation of the 84th N back to S according to the Kabat numbering system.
鼠源抗體CP33人源化抗體可變區序列如下: The variable region sequence of the mouse antibody CP33 humanized antibody is as follows:
>huCP33VL1(hu CP33 VL-CDR grafted) >huCP33VL1(huCP33VL-CDR grafted)
SEQ ID NO:40 SEQ ID NO: 40
>huCP33VL2 >huCP33VL2
SEQ ID NO:41 SEQ ID NO: 41
>huCP33VL3 >huCP33VL3
SEQ ID NO:42 SEQ ID NO: 42
>huCP33VL4 >huCP33VL4
SEQ ID NO:43 SEQ ID NO: 43
>huCP33VH1(huCP33 VH-CDR Grafted) >huCP33VH1(huCP33 VH-CDR Grafted)
SEQ ID NO:44 SEQ ID NO: 44
>huCP33VH2 >huCP33VH2
SEQ ID NO:45 SEQ ID NO: 45
>huCP33VH3 >huCP33VH3
SEQ ID NO:46 SEQ ID NO: 46
其中,劃線表示CDR,斜體部分表示連接子,其餘部分為FR。 The dashed lines represent CDRs, the italicized parts represent linkers, and the rest are FRs.
將上述輕、重鏈可變區與人輕鏈、重恆定區序列組合形成最終的完整輕、重鏈序列,進而得到全長序列的抗體。在本揭露中如無明確說明時,形成全長抗體時重鏈可變區與SEQ ID NO:47所示恆定區連接形成抗體重鏈,輕鏈可變區與SEQ ID NO:48所示的恆定區連接形成抗體輕鏈鏈,得到的CP33的人源化抗體(huCP33),不同huCP33抗體的重、輕鏈可變區見下表。 The above-mentioned light and heavy chain variable regions are combined with the human light chain and heavy constant region sequences to form the final complete light and heavy chain sequences, and then antibodies with full-length sequences are obtained. Unless otherwise specified in the present disclosure, when forming a full-length antibody, the variable region of the heavy chain is linked with the constant region shown in SEQ ID NO: 47 to form an antibody heavy chain, and the variable region of the light chain is connected with the constant region shown in SEQ ID NO: 48 to form an antibody heavy chain. The regions are connected to form an antibody light chain chain, and the obtained humanized antibody of CP33 (huCP33), the heavy and light chain variable regions of different huCP33 antibodies are shown in the following table.
註:huCP33-01抗體,其重鏈可變區為huCP33VH1、輕鏈可變區為huCP33VL1,重鏈恆定區為SEQ ID NO:47,輕鏈恆定區為SEQ ID NO:48,其他類推。 Note: huCP33-01 antibody, its heavy chain variable region is huCP33VH1, light chain variable region is huCP33VL1, heavy chain constant region is SEQ ID NO: 47, light chain constant region is SEQ ID NO: 48, and so on.
藉由ELISA實驗檢測huCP33抗體及嵌合抗體與人ANG-2的結合活性,其結果如下表所示: The binding activity of huCP33 antibody and chimeric antibody to human ANG-2 was detected by ELISA, and the results are shown in the following table:
結果顯示,CP33人源化抗體與人ANG-2具有親和力與ChCP33相當,均可特異性識別人ANG-2。 The results showed that the CP33 humanized antibody had an affinity for human ANG-2 comparable to that of ChCP33, and both could specifically recognize human ANG-2.
5.4. 抗ANG-2抗體的恆定區及全長抗體序列5.4. Constant region and full-length antibody sequence of anti-ANG-2 antibody
示例性的抗體的抗體的重、輕鏈恆定區如下: The heavy and light chain constant regions of the antibodies of the exemplary antibodies are as follows:
重鏈恆定區: Heavy chain constant region:
SEQ ID NO:47 SEQ ID NO: 47
輕鏈恆定區: Light chain constant region:
SEQ ID NO:48 SEQ ID NO: 48
示例性的huHR54全長抗體和huCP33全長抗體的序列如下: The sequences of exemplary huHR54 full-length antibodies and huCP33 full-length antibodies are as follows:
(a)huHR54-01抗體(a) huHR54-01 antibody
huHR54-01重鏈: huHR54-01 heavy chain:
SEQ ID NO:49 SEQ ID NO: 49
huHR54-01輕鏈: huHR54-01 light chain:
SEQ ID NO:50 SEQ ID NO: 50
(b)huHR54-04抗體(b) huHR54-04 antibody
huHR54-04重鏈: huHR54-04 heavy chain:
SEQ ID NO:51 SEQ ID NO: 51
huHR54-04輕鏈: huHR54-04 light chain:
SEQ ID NO:50 SEQ ID NO: 50
(c)huHR54-12抗體(c) huHR54-12 antibody
huHR54-12重鏈: huHR54-12 heavy chain:
SEQ ID NO:52 SEQ ID NO: 52
huHR54-12輕鏈: huHR54-12 light chain:
SEQ ID NO:50 SEQ ID NO: 50
(d)huCP33-01抗體(d) huCP33-01 antibody
huCP33-01抗體重鏈: huCP33-01 antibody heavy chain:
SEQ ID NO:53 SEQ ID NO: 53
huCP33-01抗體輕鏈: huCP33-01 antibody light chain:
SEQ ID NO:54 SEQ ID NO: 54
(e)huCP33-05抗體(e) huCP33-05 antibody
huCP33-05重鏈: huCP33-05 heavy chain:
SEQ ID NO:55 SEQ ID NO: 55
huCP33-05輕鏈: huCP33-05 light chain:
SEQ ID NO:54 SEQ ID NO: 54
其中,劃線表示CDR,其餘部分為FR,點劃線部分為恆定區。 The dashed lines represent CDRs, the rest are FRs, and the dashed-dotted lines represent constant regions.
實施例6. 抗VEGF/ANG-2雙特異性抗體的製備及鑑定Example 6. Preparation and identification of anti-VEGF/ANG-2 bispecific antibodies
本揭露構建了針對VEGF和ANG-2的不同形式的雙特異性抗體,包括但不限於IgG-scFv、DVD和crosmab形式。 The present disclosure constructs bispecific antibodies against VEGF and ANG-2 in different formats, including but not limited to IgG-scFv, DVD and crosmab formats.
抗VEGF抗體可以是目前已知的針對VEGF的任何抗體,比如阿瓦斯汀(Avastin)、RAZUMAB(Axxiom Inc)、GNR-011(Affitech A/S)、R-TPR-024(Reliance Life Sciences Grou)、雷莫蘆單抗(ramucirumab,ImClone Systems)等。 Anti-VEGF antibody can be any antibody against VEGF currently known, such as Avastin (Avastin), RAZUMAB (Axxiom Inc), GNR-011 (Affitech A/S), R-TPR-024 (Reliance Life Sciences Grou) , Ramucirumab (ramucirumab, ImClone Systems) and the like.
示例性的抗體為基因泰克已上市Fab抗體雷珠單抗(Lucentis),其輕鏈可變區序列如SEQ ID NO:56所示(參見WO1998045332或CAS Registry Number:347396-82-1)。抗VEGF抗體的重鏈是將雷珠單抗的重鏈可變區(序列如SEQ ID NO:58所示)與不同的人IgG1恆定區組合成完整IgG1重鏈。其具體序列如下: An exemplary antibody is the Genentech marketed Fab antibody ranibizumab (Lucentis), whose light chain variable region sequence is shown in SEQ ID NO: 56 (see WO1998045332 or CAS Registry Number: 347396-82-1). The heavy chain of the anti-VEGF antibody was the combination of the heavy chain variable region of ranibizumab (sequence shown in SEQ ID NO: 58) with various human IgG1 constant regions to form a complete IgG1 heavy chain. Its specific sequence is as follows:
(1)雷珠單抗的輕鏈可變區 (1) Light chain variable region of ranibizumab
SEQ ID NO:56 SEQ ID NO: 56
(2)雷珠單抗的輕鏈 (2) Light chain of ranibizumab
SEQ ID NO:57 SEQ ID NO: 57
(3)雷珠單抗的重鏈可變區 (3) The heavy chain variable region of ranibizumab
SEQ ID NO:58 SEQ ID NO: 58
(4)雷珠單抗的重鏈可變區+IgG1恆定區(本文稱為雷珠單抗重鏈1) (4) Ranibizumab heavy chain variable region + IgG1 constant region (herein referred to as Ranibizumab heavy chain 1)
SEQ ID NO:59 SEQ ID NO: 59
(5)雷珠單抗的重鏈可變區+IgG1恆定區變體(本文稱為雷珠單抗重鏈2) (5) Ranibizumab heavy chain variable region + IgG1 constant region variant (herein referred to as Ranibizumab heavy chain 2)
SEQ ID NO:60 SEQ ID NO: 60
其中,劃線表示CDR,斜體部分表示連接子,其餘部分為FR。 The dashed lines represent CDRs, the italicized parts represent linkers, and the rest are FRs.
為了製備IgG-scFv構型的雙特異性抗體,將篩選得到的抗ANG-2抗體的重鏈可變區和輕鏈可變區藉由連接子連接,得到抗ANG-2單鏈抗體。 其中,連接子可以是本領域公知任何連接子,示例性的連接子為(GGGGS)n,其中n為1-10中的整數。示例性的ANG-2單鏈抗體序列如下所示: In order to prepare a bispecific antibody in IgG-scFv configuration, the heavy chain variable region and light chain variable region of the anti-ANG-2 antibody obtained by screening were connected by a linker to obtain an anti-ANG-2 single-chain antibody. The linker can be any linker known in the art, and an exemplary linker is (GGGGS)n, where n is an integer from 1 to 10. Exemplary ANG-2 single chain antibody sequences are shown below:
huHR54-01-scFv huHR54-01-scFv
SEQ ID NO:67 SEQ ID NO: 67
huHR54-04-scFv huHR54-04-scFv
SEQ ID NO:68 SEQ ID NO: 68
huHR54-12-scFv: huHR54-12-scFv:
SEQ ID NO:69 SEQ ID NO: 69
huCP33-01-scFv: huCP33-01-scFv:
SEQ ID NO:70 SEQ ID NO: 70
huCP33-05-scFv: huCP33-05-scFv:
SEQ ID NO:71 SEQ ID NO: 71
其中,劃線表示CDR,斜體部分表示連接子,其餘部分為FR。 The dashed lines represent CDRs, the italicized parts represent linkers, and the rest are FRs.
此外,在抗ANG-2的單鏈抗體的重、輕鏈可變區中引入半胱胺酸突變,示例性的序列如下: In addition, cysteine mutations were introduced into the heavy and light chain variable regions of the anti-ANG-2 single chain antibody, and the exemplary sequences are as follows:
huHR54VH2-C huHR54VH2-C
SEQ ID NO:72 SEQ ID NO: 72
huHR54VH2f-C huHR54VH2f-C
SEQ ID NO:73 SEQ ID NO: 73
huHR54VL1-C huHR54VL1-C
SEQ ID NO:74 SEQ ID NO: 74
huCP33VH2-C huCP33VH2-C
SEQ ID NO:75 SEQ ID NO: 75
huCP33VL1-C huCP33VL1-C
SEQ ID NO:76 SEQ ID NO: 76
其中,劃線表示CDR,其餘部分為FR。 Among them, the dashed lines represent CDRs, and the rest are FRs.
示例性的,抗ANG-2單鏈抗體的序列如下: Exemplarily, the sequence of the anti-ANG-2 single chain antibody is as follows:
huHR54-04-scFv-CC: huHR54-04-scFv-CC:
SEQ ID NO:77 SEQ ID NO: 77
huHR54-12-scFv-CC huHR54-12-scFv-CC
SEQ ID NO:78 SEQ ID NO: 78
huCP33-05-scFv-CC huCP33-05-scFv-CC
SEQ ID NO:79 SEQ ID NO: 79
其中,劃線表示CDR,斜體部分表示連接子,其餘部分為FR。 The dashed lines represent CDRs, the italicized parts represent linkers, and the rest are FRs.
藉由基因重組技術將抗ANG-2單鏈抗體直接地(藉由肽鍵)或間接地(藉由接頭)連接至抗VEGF抗體的重鏈或輕鏈,藉由293表達系統進行表達,得到雙特異性抗體。 The anti-ANG-2 single-chain antibody is directly (by peptide bond) or indirectly (by linker) linked to the heavy chain or light chain of the anti-VEGF antibody by gene recombination technology, and expressed by 293 expression system to obtain Bispecific antibodies.
示例性的,可將抗ANG-2單鏈抗體的N-端藉由接頭(如(GG)n,其中n為1-20)連接至抗VEGF抗體的重鏈C-端,得到雙特異性抗體,其序列如下所示: Exemplarily, the N-terminus of the anti-ANG-2 single chain antibody can be linked to the C-terminus of the heavy chain of the anti-VEGF antibody via a linker (eg (GG)n, where n is 1-20), resulting in a bispecific Antibodies, whose sequences are shown below:
(1)雙特異抗體1(1)
>雙特異性抗體1第一條鏈:
>
SEQ ID NO:80 SEQ ID NO: 80
其中,波浪下劃線部分表示抗VEGF抗體的HCDR,點下劃線表示恆定區,雙下劃線表示連接子,斜體部分表示ANG2-scFv,劃線部分表示ANG2-scFv的CDR。 The wavy underlined part represents the HCDR of the anti-VEGF antibody, the dotted underlined part represents the constant region, the double underlined part represents the linker, the italicized part represents ANG2-scFv, and the underlined part represents the CDR of ANG2-scFv.
>雙特異抗體1第二條鏈:
>
SEQ ID NO:57 SEQ ID NO: 57
其中,點下劃線表示恆定區,劃線部分表示CDR。 Among them, the dotted underline represents the constant region, and the dashed portion represents the CDR.
(2)雙特異性抗體2(2)
>雙特異性抗體2第一條鏈:
>
SEQ ID NO:81 SEQ ID NO: 81
其中,波浪下劃線部分表示抗VEGF抗體的HCDR,點下劃線表示恆定區,雙下劃線表示連接子,斜體部分表示ANG2-scFv,劃線部分表示ANG2-scFv的CDR。 The wavy underlined part represents the HCDR of the anti-VEGF antibody, the dotted underlined part represents the constant region, the double underlined part represents the linker, the italicized part represents ANG2-scFv, and the underlined part represents the CDR of ANG2-scFv.
>雙特異性抗體2第二條鏈:
>
SEQ ID NO:57 SEQ ID NO: 57
其中,點下劃線表示恆定區,劃線部分表示CDR。 Among them, the dotted underline represents the constant region, and the dashed portion represents the CDR.
(3)雙特異性抗體3(3)
>雙特異抗體3第一條鏈:
>
SEQ ID NO:82 SEQ ID NO: 82
其中,波浪下劃線部分表示抗VEGF抗體的HCDR,點下劃線表示恆定區,雙下劃線表示連接子,斜體部分表示ANG2-scFv,劃線部分表示ANG2-scFv的CDR。 The wavy underlined part represents the HCDR of the anti-VEGF antibody, the dotted underlined part represents the constant region, the double underlined part represents the linker, the italicized part represents ANG2-scFv, and the underlined part represents the CDR of ANG2-scFv.
>雙特異抗體3第二條鏈:
>
SEQ ID NO:57 SEQ ID NO: 57
其中,點下劃線表示恆定區,劃線部分表示CDR。 Among them, the dotted underline represents the constant region, and the dashed portion represents the CDR.
(4)雙特異性抗體4(4) Bispecific Antibody 4
>雙特異性抗體4第一條鏈: >Bispecific antibody 4 first chain:
SEQ ID NO:83 SEQ ID NO: 83
其中,波浪下劃線部分表示抗VEGF抗體的HCDR,點下劃線表示恆定區,雙下劃線表示連接子,斜體部分表示ANG2-scFv,劃線部分表示ANG2-scFv的CDR。 The wavy underlined part represents the HCDR of the anti-VEGF antibody, the dotted underlined part represents the constant region, the double underlined part represents the linker, the italicized part represents ANG2-scFv, and the underlined part represents the CDR of ANG2-scFv.
>雙特異性抗體4第二條鏈: >Bispecific antibody 4 second chain:
SEQ ID NO:57 SEQ ID NO: 57
其中,點下劃線表示恆定區,劃線部分表示CDR。 Among them, the dotted underline represents the constant region, and the dashed portion represents the CDR.
同時,利用篩選得到的抗ANG-2抗體和VEGF抗體,本揭露構建了Crossmab形式的雙特異性抗體,示例性的Crossmab形式的雙特異性抗體的序列如下: At the same time, using the anti-ANG-2 antibody and VEGF antibody obtained by screening, the present disclosure has constructed a bispecific antibody in the form of Crossmab. The sequence of an exemplary bispecific antibody in the form of Crossmab is as follows:
(5)雙特異性抗體5(5)
>雙特異性抗體5第一重鏈:抗ANG-2重鏈
>
SEQ ID NO:84 SEQ ID NO: 84
>雙特異性抗體5第一輕鏈:抗ANG-2輕鏈
>
SEQ ID NO:85 SEQ ID NO: 85
>雙特異性抗體5第二重鏈:抗VEGF重鏈
>
SEQ ID NO:86 SEQ ID NO: 86
>雙特異性抗體5第二輕鏈:抗VEGF輕鏈
>
SEQ ID NO:57 SEQ ID NO: 57
其中,點下劃線表示恆定區,劃線部分表示CDR。 Among them, the dotted underline represents the constant region, and the dashed portion represents the CDR.
(6)雙特異性抗體6(6) Bispecific Antibody 6
>雙特異性抗體6第一重鏈:抗ANG-2重鏈 >Bispecific antibody 6 first heavy chain: anti-ANG-2 heavy chain
SEQ ID NO:87 SEQ ID NO: 87
>雙特異性抗體6第一輕鏈:抗ANG-2輕鏈 >Bispecific antibody 6 first light chain: anti-ANG-2 light chain
>雙特異性抗體6第二重鏈:抗VEGF重鏈 >Bispecific antibody 6 second heavy chain: anti-VEGF heavy chain
SEQ ID NO:86 SEQ ID NO: 86
>雙特異性抗體6第二輕鏈:抗VEGF輕鏈 > Bispecific Antibody 6 Second Light Chain: Anti-VEGF Light Chain
SEQ ID NO:57 SEQ ID NO: 57
其中,點下劃線表示恆定區,劃線部分表示CDR。 Among them, the dotted underline represents the constant region, and the dashed portion represents the CDR.
按照實施例2中的純化方式,使用proteinA親和層析純化即可得到純度>98%的雙特異性抗體分子。 According to the purification method in Example 2, a bispecific antibody molecule with a purity of >98% can be obtained by using proteinA affinity chromatography to purify.
本揭露還使用RG7716,阿瓦斯汀和RG7221(vanucizumab)作為對照分子,其序列如下所示: The present disclosure also uses RG7716, Avastin, and RG7221 (vanucizumab) as control molecules, the sequences of which are shown below:
>RG7716抗-ANG-2重鏈: >RG7716 anti-ANG-2 heavy chain:
SEQ ID NO:89 SEQ ID NO: 89
>RG7716抗ANG-2輕鏈: >RG7716 anti-ANG-2 light chain:
SEQ ID NO:90 SEQ ID NO: 90
>RG7716抗VEGF重鏈 >RG7716 anti-VEGF heavy chain
SEQ ID NO:91 SEQ ID NO: 91
>RG7716抗VEGF輕鏈 >RG7716 anti-VEGF light chain
SEQ ID NO:92 SEQ ID NO: 92
>阿瓦斯汀重鏈 >Avastin Heavy Chain
SEQ ID NO:93 SEQ ID NO: 93
>阿瓦斯汀輕鏈 >Avastin Light Chain
SEQ ID NO:94 SEQ ID NO: 94
>RG7221抗ANG2重鏈 >RG7221 anti-ANG2 heavy chain
SEQ ID NO:95 SEQ ID NO: 95
>RG7221抗ANG2輕鏈 >RG7221 anti-ANG2 light chain
SEQ ID NO:96 SEQ ID NO: 96
>RG7221抗VEGF重鏈 >RG7221 anti-VEGF heavy chain
SEQ ID NO:97 SEQ ID NO: 97
>RG7221抗VEGF輕鏈 >RG7221 anti-VEGF light chain
SEQ ID NO:98 SEQ ID NO: 98
另外,本揭露中使用的陰性對照(NC)是指針對HIV的一個IgG單株抗體。 Additionally, the negative control (NC) used in this disclosure refers to an IgG monoclonal antibody against HIV.
體外活性生物學評價Biological evaluation of in vitro activity
測試例1. Biacore測定VEGF/ANG-2雙特異性抗體與不同種屬VEGF/ANG-2的親和力Test Example 1. Biacore determination of the affinity of VEGF/ANG-2 bispecific antibody to different species of VEGF/ANG-2
用Biacore T200(GE)測定VEGF/ANG-2雙特異性抗體與人、猴和小鼠VEGF和ANG-2的親和力。 The affinity of VEGF/ANG-2 bispecific antibodies to human, monkey and mouse VEGF and ANG-2 was determined using a Biacore T200 (GE).
用Protein A生物傳感芯片親和捕獲抗體,然後於芯片表面流經抗原人VEGF(R&D,293-VE),猴VEGF(sinobiological,11066),小鼠VEGF(sinobiological,51059),人ANG-2(sinobiological,10691-H08H),猴ANG-2(sinobiological,90026-C07H),小鼠ANG-2(sinobiological,50298-M07H)。用Biacore T200儀器實時檢測反應信號,獲得結合和解離曲線。在每個實驗循環解離完成後,用10mM甘胺酸-鹽酸再生溶液(pH 1.5)將生物傳感芯片洗淨再生。用BIA評估版本4.1,GE軟體以(1:1)Langmuir模型擬合數據,得出親和力數值,如表14所示。 The antibody was affinity-captured with Protein A biosensor chip, and then the antigens human VEGF (R&D, 293-VE), monkey VEGF (sinobiological, 11066), mouse VEGF (sinobiological, 51059), human ANG-2 ( sinobiological, 10691-H08H), monkey ANG-2 (sinobiological, 90026-C07H), mouse ANG-2 (sinobiological, 50298-M07H). Reaction signals were detected in real time with a Biacore T200 instrument, and binding and dissociation curves were obtained. After the dissociation in each experimental cycle, the biosensor chip was washed and regenerated with 10 mM glycine-hydrochloric acid regeneration solution (pH 1.5). Using BIA evaluation version 4.1, the GE software fitted the data with a (1:1) Langmuir model, resulting in affinity values as shown in Table 14.
結果顯示,雙特異性抗體1,雙特異性抗體5,雙特異性抗體2和雙特異性抗體3與人和猴的VEGF和ANG-2均有較高的親和力。
The results showed that
測試例2. 基於ELISA的抗體阻斷ANG-2結合Tie2受體實驗Test Example 2. ELISA-based antibody blocking experiment of ANG-2 binding to Tie2 receptor
ANG-2與血管內皮細胞表面的ANG-2受體Tie2結合,引發Tie2細胞內酪胺酸激酶發生磷酸化,繼而傳導信號使外周細胞從血管內皮細胞脫落,使血管處於不穩定易於增殖的狀態。因此,藉由抗體阻斷ANG-2與Tie2的結合可以使血管更穩定,抑制新生血管的生成。本實驗鑑定結果表明雙特異性抗體可以阻斷ANG-2結合到重組表達的Tie2蛋白胞外區。 ANG-2 binds to the ANG-2 receptor Tie2 on the surface of vascular endothelial cells, triggering the phosphorylation of tyrosine kinase in Tie2 cells, and then transducing signals to make peripheral cells shed from vascular endothelial cells, leaving blood vessels in an unstable and proliferating state . Therefore, blocking the binding of ANG-2 to Tie2 by antibodies can make blood vessels more stable and inhibit the formation of new blood vessels. The results of this experiment indicated that the bispecific antibody could block the binding of ANG-2 to the extracellular domain of recombinantly expressed Tie2 protein.
具體方法:用Tie2-Fc(SEQ ID NO:2,3μg/mL溶於PBS,100μL/孔,)包被ELISA板,4℃包被過夜,去除包被液後,加入用PBS稀釋的5%脫脂牛奶封閉液200μL/孔,37℃孵育箱孵育2小時進行封閉。封閉結束後,棄去封閉液,並用PBST緩衝液(含0.05%吐溫-20的PBS,pH7.4)洗板5次後,加入50μL用1% BSA稀釋生物素標記試劑盒(東仁化學,LK03)標記的huANG-
2-Fc(SEQ ID NO:1,bio-huANG-2-Fc,終濃度為0.15μg/mL)和50μL待測抗體(始濃度為10μg/mL,3倍比稀釋),混勻後37℃孵育15分鐘,加至ELISA板,37℃孵育1小時。孵育結束後,棄去酶標板中的反應液,用PBST洗板5次後,加入100μL/孔1:4000稀釋的鏈黴親和素-過氧化物酶聚合物(Sigma,S2438-250UG)於37℃孵育1小時。用PBST洗板5次後,加入100μL/孔TMB顯色受質(KPL,52-00-03),於室溫孵育3-10分鐘,加入100μL/孔1M H2SO4終止反應,用NOVOStar酶標儀在450nm處讀取吸收值,計算抗體阻斷ANG-2與Tie2結合的IC50值。
Specific method: Coat ELISA plate with Tie2-Fc (SEQ ID NO: 2, 3 μg /mL dissolved in PBS, 100 μL /well), coat overnight at 4°C, remove the coating solution, add PBS Dilute 5% skim
結果顯示,雙特異性抗體均可較強地抑制ANG-2與Tie2的結合(見表15和圖1)。 The results showed that both bispecific antibodies could strongly inhibit the binding of ANG-2 to Tie2 (see Table 15 and Figure 1).
測試例3. 雙特異性抗體抑制ANG-2誘導的Tie2磷酸化Test Example 3. Bispecific antibody inhibits ANG-2-induced Tie2 phosphorylation
將過表達huTie2的穩轉株CHO-Tie2#1B11-1消化重新懸浮後,用完全培養基調整密度為2.5×105細胞/mL,100μL/孔鋪入96孔細胞培養板,即2.5×104細胞/孔。4~5小時後換液(DME/F-12,HyClone,Cat#SH30023.01+0.1%BSA+20μg/mL嘌呤黴素,Gibco,Cat# A1113803),饑餓過夜。4.0μg/mL,100μL/孔抗huTie2 capture(R&D Systems,Cat# DYC2720E)包板,常溫過夜。去除包被液,250μL/孔1% BSA+0.05% NaN3封閉液,常溫封閉2小時。將25μL huANG-2-
Fc(終濃度2.5μg/mL)和25μL待測抗體(3倍比稀釋抗體,最高濃度50.0nM)等體積混勻,37℃孵育15分鐘後,再加入Na3VO4(1.0mM,Sigma,Cat# S6508),混勻。過夜培養的細胞,棄去50μL培養上清後,加入50μL準備好的抗原抗體混合液,37℃孵育10分鐘。200μL/孔洗液(PBS+2.0mM Na3VO4)洗兩次。加入90μL裂解液((1×裂解緩衝液+10μg/mL Leupeptin hemisulfate(Tocris,Cat# 1167)+10.0μg/mL APROTININ,Sigma,Cat# SRE0050))冰上裂解10~15分鐘,4000g離心5分鐘。收集細胞裂解液,加入已封閉好的ELISA板中,常溫孵育2h。PBST洗板5次,加入1:1000稀釋的二抗抗PY-HRP(R&D Systems,Cat# DYC2720E),常溫孵育1~2小時。PBST洗板5次,TMB顯色15~30分鐘,1M H2SO4終止顯色。用Versa Max酶標儀讀取OD450,計算IC50。
After digesting and resuspending the stable transgenic CHO-Tie2 #1B11-1 overexpressing huTie2 , the density was adjusted to 2.5×10 5 cells/mL with complete medium, and 100 μL/well was spread into a 96-well cell culture plate, that is, 2.5× 10 4 cells/well. After 4-5 hours, the medium was changed (DME/F-12, HyClone, Cat# SH30023.01 +0.1%BSA+20 μg/mL puromycin, Gibco, Cat#A1113803) and starved overnight. 4.0 μg/mL, 100 μL/well anti- huTie2 capture (R&D Systems, Cat# DYC2720E ) was coated on the plate, overnight at room temperature. The coating solution was removed, and 250 μL /well of 1% BSA+0.05% NaN 3 blocking solution was used for blocking at room temperature for 2 hours.
結果(見圖2)顯示,雙特異性抗體顯示出很強的抑制ANG-2介導的Tie2磷酸化的能力。 The results (see Figure 2) show that the bispecific antibody displayed a strong ability to inhibit ANG-2 mediated phosphorylation of Tie2.
測試例4. 雙特異性抗體抑制VEGF誘導的VEGFR的磷酸化Test Example 4. Bispecific antibody inhibits VEGF-induced phosphorylation of VEGFR
VEGF與血管內皮細胞上的VEGFR結合,使VEGFR胞內區生磷酸化,促進內皮細胞增殖形成新的血管,進而促進腫瘤細胞的生長和轉移。本實驗用來鑑定雙特異性抗體可以阻止VEGF誘導的VEGFR的磷酸化。 VEGF binds to VEGFR on vascular endothelial cells, phosphorylates the intracellular region of VEGFR, and promotes endothelial cell proliferation to form new blood vessels, thereby promoting tumor cell growth and metastasis. This experiment was used to identify bispecific antibodies that prevent VEGF-induced phosphorylation of VEGFR.
具體為:將HUVEC細胞(PromoCell/妙通生物,C-12205)消化後用完全培養基將細胞密度調整為每500μL中含1.5×105個細胞,加到24孔板 中,每孔500μL。在37℃培養箱中培養過夜後,棄去培養基,用500μL冰DPBS(Gibco,14190-250)洗一遍,每孔加200μL含0.1% BSA的基本培養基饑餓培養30分鐘。將待測抗體、陰性對照抗體用基本培養基稀釋至10nM、1nM和0.1nM(RG7221和雙特異性抗體5為20nM、2nM和0.2nM),使用PBS作為對照(ctrl)。將VEGF(R&D system,Cat#293-VE)用基本培養基稀釋至400ng/mL。取等體積稀釋好的VEGF和抗體混勻後取200μL加到培養板對應的孔中,37℃孵育5分鐘。將4×裂解緩衝液#1(cisbio,63ADK041PEG)用dd H2O稀釋成1×。將封閉液用1×裂解緩衝液稀釋100倍配製成裂解液。取出細胞培養板,棄去細胞培養板中的培養基,加入冰PBS 500μL,輕微晃動後,棄去PBS。立即加入50μL配製好的裂解液,放在振盪器上,室溫孵育30分鐘。2400g離心10分鐘,收集上清。使用Phospho-VEGFR2(Tyr1175)試劑盒(cisbio,63ADK041PEG)檢測上清中p-VEGFR。檢測方法為取10μL磷酸-VEGFR2(Tyr1175)d2抗體,加入200μL檢測緩衝液,配製成工作液。取10μL磷酸-VEGFR2(Tyr1175)Cryptate抗體,加入200μL檢測緩衝液,配製成工作液。將d2抗體工作液與Cryptate抗體工作液等體積混合,在HTRF96孔微孔板中,加入16μL細胞裂解液,加入4μL d2抗體與Cryptate抗體混合液,用封板膜封好,微孔板離心機離心1分鐘,室溫避光孵育4-24小時,用PHERAstar多功能酶標儀讀取340nm波長激發,665nm和620nm波長發射的螢光值。 Specifically: after digesting HUVEC cells (PromoCell/Miaotong Bio, C-12205), the cell density was adjusted to contain 1.5×10 5 cells per 500 μL with complete medium, and added to a 24-well plate with 500 cells per well. μL . After overnight incubation in a 37°C incubator, the medium was discarded, washed with 500 μL of ice DPBS (Gibco, 14190-250 ), and 200 μL of minimal medium containing 0.1% BSA was added to each well for starvation for 30 minutes. Test antibody, negative control antibody were diluted with minimal medium to 10 nM, 1 nM and 0.1 nM (20 nM, 2 nM and 0.2 nM for RG7221 and bispecific antibody 5), PBS was used as control (ctrl). VEGF (R&D system, Cat#293-VE) was diluted to 400 ng/mL with minimal medium. Take an equal volume of diluted VEGF and the antibody, mix well, add 200 μL to the corresponding well of the culture plate, and incubate at 37°C for 5 minutes. 4X Lysis Buffer # 1 (cisbio, 63ADK041PEG) was diluted to 1X with ddH2O. The blocking solution was diluted 100 times with 1× lysis buffer to prepare a lysis solution. The cell culture plate was taken out, the medium in the cell culture plate was discarded, 500 μL of ice-cold PBS was added, and PBS was discarded after slight shaking. Immediately add 50 μL of the prepared lysate, place on a shaker, and incubate at room temperature for 30 minutes. Centrifuge at 2400g for 10 minutes and collect the supernatant. The Phospho-VEGFR2 (Tyr1175) kit (cisbio, 63ADK041PEG) was used to detect p-VEGFR in the supernatant. The detection method is to take 10 μL of phospho-VEGFR2( Tyr1175 )d2 antibody, add 200 μL of detection buffer, and prepare a working solution. Take 10 μL of phospho-VEGFR2 ( Tyr1175 ) Cryptate antibody, add 200 μL detection buffer, and prepare a working solution. Mix equal volumes of d2 antibody working solution and Cryptate antibody working solution, add 16 μL of cell lysate and 4 μL of d2 antibody and Cryptate antibody mixture to HTRF96-well microplate, seal with a plate-sealing film, and microplate the solution. Centrifuge for 1 minute in a plate centrifuge, incubate at room temperature for 4-24 hours in the dark, and use a PHERAstar multifunctional microplate reader to read the fluorescence values of excitation at 340 nm and emission at 665 nm and 620 nm.
數據處理:比=信號665nm/信號620nm×10000,用Graphpad Prism 5繪製柱狀圖。結果如圖3所示。
Data processing: ratio = signal 665 nm/signal 620 nm x 10000, histograms were drawn with
結果表明,雙特異性抗體可以顯著抑制VEGF引起的HUVEC胞內磷酸化VEGFR水平升高。 The results showed that the bispecific antibody could significantly inhibit the VEGF-induced increase in the level of intracellular phosphorylated VEGFR in HUVECs.
測試例5. 雙特異性抗體抑制VEGF誘導的HUVEC的增殖Test Example 5. Bispecific antibody inhibits VEGF-induced proliferation of HUVEC
VEGF與HUVEC上的VEGFR結合,使VEGFR胞內區發生磷酸化,促進HUVEC增殖,本實驗用來鑑定雙特異性抗體可以阻止VEGF誘導的HUVEC的增殖。 VEGF binds to VEGFR on HUVEC, phosphorylates the intracellular region of VEGFR, and promotes the proliferation of HUVEC. This experiment is used to identify bispecific antibodies that can prevent the proliferation of HUVEC induced by VEGF.
具體方法如下: The specific method is as follows:
將對數生長期的HUVEC細胞用0.08%胰酶消化,室溫大約1-2分鐘,加10% FBS終止。收集上述消化後的HUVEC,800rpm/分鐘,離心5分鐘,用PBS洗三遍除去培養基中刺激HUVEC增殖的細胞因子(800rpm/分鐘,離心5分鐘)。將HUVEC細胞重新懸浮於6%FBS培養基中,細胞計數後,按照4000細胞/50μL/孔接種在白色96孔細胞培養板中,培養箱中培養2小時。將VEGF起始濃度調整為300ng/mL,120μL/孔加入無菌96孔板中。將待測抗體梯度稀釋:抗體起始濃度為600nM,4倍比進行梯度稀釋,將稀釋好的抗體等體積加入上述96孔板中,室溫孵育30min。將孵育好的抗體、VEGF混合物100μL/孔加入貼壁後的HUVEC細胞中,培養箱中培養5天,培養結束後加入CellTiter-Glo®(G7573,PROMEGA),50μL/孔,室溫避光孵育10min,用Cytation5細胞成像儀Luminescence程序檢測。結果如圖4所示。 HUVEC cells in logarithmic growth phase were digested with 0.08% trypsin for about 1-2 minutes at room temperature, and stopped by adding 10% FBS. The above-digested HUVECs were collected, centrifuged at 800 rpm/min for 5 minutes, washed three times with PBS to remove cytokines that stimulate HUVEC proliferation in the medium (800 rpm/min, centrifuged for 5 minutes). The HUVEC cells were resuspended in 6% FBS medium, and after cell counting, 4000 cells/50 μL /well were seeded in a white 96-well cell culture plate, and cultured in an incubator for 2 hours. The initial concentration of VEGF was adjusted to 300 ng/mL, and 120 μL /well was added to a sterile 96-well plate. Gradient dilution of the antibody to be tested: the initial concentration of the antibody is 600 nM, and the gradient is diluted by 4 times. The diluted antibody is added in an equal volume to the above 96-well plate, and incubated at room temperature for 30 min. Add 100 μL/well of the incubated antibody and VEGF mixture to the adherent HUVEC cells, culture in an incubator for 5 days, and add CellTiter-Glo® ( G7573 , PROMEGA ), 50 μL/well, chamber Incubate in the dark for 10 min, and use the Cytation5 cell imager Luminescence program to detect. The results are shown in Figure 4.
結果顯示,雙特異性抗體可以顯著抑制VEGF引起的HUVEC的增殖。 The results showed that the bispecific antibody could significantly inhibit the proliferation of HUVEC induced by VEGF.
體內活性生物學評價Biological evaluation of in vivo activity
測試例6:雙特異性抗體在前列腺癌細胞小鼠皮下移植瘤模型上的藥效Test Example 6: Pharmacological effects of bispecific antibodies on mouse subcutaneous xenograft model of prostate cancer cells
將5×106 PC-3細胞(ATCC)接種於Balb/c裸鼠右肋部皮下,當荷瘤小鼠腫瘤體積達到100mm3左右時,將小鼠分別隨機分為6組,每組8隻。將分組當天定義第0天,分組當日開始腹腔注射等莫耳量的各抗體,每週2次,共給藥6次。每週2次監測腫瘤體積、動物重量並記錄數據。當腫瘤體積超過1000mm3或多數腫瘤出現破潰或體重下降20%時,將荷瘤動物進行安樂死作為實驗終點。所有數據使用Excel和GraphPad Prism 5軟體進行作圖及統計分析。
5×10 6 PC-3 cells (ATCC) were inoculated subcutaneously in the right flank of Balb/c nude mice. When the tumor volume of the tumor-bearing mice reached about 100 mm 3 , the mice were randomly divided into 6 groups, with 8 in each group. Only. The day of grouping was defined as
腫瘤體積(V)計算公式為:V=1/2×a×b2,其中a、b分別表示長、寬。 The calculation formula of tumor volume (V) is: V=1/2×a×b 2 , where a and b represent length and width, respectively.
相對腫瘤增殖率T/C(%)=(T-T0)/(C-C0)×100,其中T、C為實驗結束時治療組和對照組的腫瘤體積;T0、C0為實驗開始時的腫瘤體積。 Relative tumor proliferation rate T/C(%)=(T-T0)/(C-C0)×100, where T and C are the tumor volumes of the treatment group and control group at the end of the experiment; T0 and C0 are the tumor volumes at the beginning of the experiment. tumor volume.
抑瘤率TGI(%)=1-T/C(%)。 Tumor inhibition rate TGI(%)=1-T/C(%).
分組情況和給藥方案如表18所示,腫瘤生長曲線和抑瘤率見圖5和表19。 The grouping situation and dosing schedule are shown in Table 18, and the tumor growth curve and tumor inhibition rate are shown in Figure 5 and Table 19.
結果顯示,本揭露中的雙特異性抗體5和雙特異性抗體2均可顯著抑制PC-3腫瘤的生長。雙特異性抗體5的抑瘤效果顯著優於與阿瓦斯汀單抗,以及RG7221雙抗。
The results show that both
測試例7 雙特異性抗體在高轉移非小細胞肺癌H460-Luc細胞株BALB/c裸鼠皮下移植瘤模型上的藥效Test Example 7 Efficacy of bispecific antibodies on the subcutaneous xenograft model of highly metastatic non-small cell lung cancer H460-Luc cell line BALB/c nude mice
本實驗評價ANG-2/VEGF雙特異抗體以腹腔注射給藥後抑制人非小細胞肺癌H460移植瘤生長和轉移的效果。 This experiment evaluated the effect of ANG-2/VEGF bispecific antibody in inhibiting the growth and metastasis of human non-small cell lung cancer H460 xenograft after intraperitoneal injection.
BALB/c裸鼠雌性,4-5週,18-20克,購自上海靈暢生物科技有限公司。人非小細胞肺癌H460-Luc(穩定轉染螢光素酶基因)細胞在添加10% FBS的RPMI 1640培養基中培養。細胞連續培養5代,接種於小鼠皮下。接種前用3-4%異氟烷將小鼠麻醉。將約1×106個H460-Luc細胞重新懸浮於無血清培養基和基質膠(Matrigel)混懸液中(培養基:Matrigel=50%:50%),藉由皮下注射接種小鼠,接種體積為200μL。當腫瘤生長到平均約100-150mm3左右時,隨機分成3組,每組8隻。分組給藥當天定義為第0天。分組情況和給藥方案如表20所示。
Female BALB/c nude mice, 4-5 weeks, 18-20 g, were purchased from Shanghai Lingchang Biotechnology Co., Ltd. Human non-small cell lung cancer H460-Luc (stably transfected luciferase gene) cells were cultured in RPMI 1640 medium supplemented with 10% FBS. Cells were continuously cultured for 5 passages and inoculated into mice subcutaneously. Mice were anesthetized with 3-4% isoflurane prior to inoculation. About 1×10 6 H460-Luc cells were resuspended in serum-free medium and Matrigel suspension (medium:Matrigel=50%:50%), and mice were inoculated by subcutaneous injection in a volume of 200 μL . When the tumors grew to an average of about 100-150 mm 3 , they were randomly divided into 3 groups with 8 animals in each group. The day of group dosing was defined as
分組後每週測量腫瘤體積兩次,連續3週。腫瘤體積(V)的計算方法如下: Tumor volumes were measured twice a week for 3 consecutive weeks after grouping. Tumor volume (V) was calculated as follows:
V=(長×寬2)/2。 V=(length×width2)/ 2 .
每隻小鼠相對腫瘤體積(RTV)的計算方法是: The relative tumor volume (RTV) per mouse was calculated as:
RTV=Vt/V0,其中Vt為每天的測量體積,V0為治療開始時的體積。 RTV=Vt/V0, where Vt is the measured volume per day and V0 is the volume at the start of treatment.
實驗結束時,所有荷瘤動物拍照,將所有腫瘤取出,稱重並拍照。 At the end of the experiment, all tumor-bearing animals were photographed, and all tumors were removed, weighed and photographed.
統計分析 Statistical Analysis
結果將以平均值±S.E.M的方式呈現。兩組間比較將用Dunnett多重比較檢驗進行檢驗。如果p<0.05則認為有統計學顯著性差異。 Results will be presented as mean ± S.E.M. Comparisons between two groups will be tested with Dunnett's multiple comparisons test. Differences were considered statistically significant if p<0.05.
測試結果如圖6所示。 The test results are shown in Figure 6.
圖6顯示,本揭露雙特異性抗體1和雙特異性抗體2抗體均可顯著抑制腫瘤的生長。
Figure 6 shows that both
測試例8:雙特異性抗體在人皮膚癌細胞小鼠皮下移植瘤模型上的藥效Test Example 8: Pharmacological effects of bispecific antibodies on mouse subcutaneous xenograft model of human skin cancer cells
將A431細胞(ATCC)2×106細胞/小鼠/100μL接種於Balb/c裸鼠右肋部皮下,當荷瘤小鼠腫瘤體積達到100mm3左右時將小鼠分別隨機分為3組:載劑(PBS)、雙特異性抗體2 4mpk和雙特異性抗體5 6mpk,每組8隻。將分組當
天定義第0天,分組當日開始腹腔注射各抗體,每週2次,共給藥6次,每週2次監測腫瘤體積、動物重量並記錄數據。當腫瘤體積超過1000mm3或多數腫瘤出現破潰或體重下降20%時,將荷瘤動物進行安樂死作為實驗終點。所有數據使用Excel和GraphPad Prism 5軟體進行作圖及統計分析。腫瘤體積(V)計算公式為:
A431 cells (ATCC) 2×10 6 cells/mouse/100 μL were inoculated subcutaneously in the right flank of Balb/c nude mice, and when the tumor volume of the tumor-bearing mice reached about 100 mm, the mice were randomly divided into 3 groups. Groups: vehicle (PBS),
V=1/2×a×b2,其中a、b分別表示長、寬。 V=1/2×a×b 2 , where a and b represent length and width, respectively.
相對腫瘤增殖率T/C(%)=(T-T0)/(C-C0)×100,其中T、C為實驗結束時治療組和對照組的腫瘤體積;T0、C0為實驗開始時的腫瘤體積。 Relative tumor proliferation rate T/C(%)=(T-T0)/(C-C0)×100, where T and C are the tumor volumes of the treatment group and control group at the end of the experiment; T0 and C0 are the tumor volumes at the beginning of the experiment. tumor volume.
抑瘤率TGI(%)=1-T/C(%)。 Tumor inhibition rate TGI(%)=1-T/C(%).
分組情況和給藥方案如表21所示,腫瘤生長曲線如圖7所示。 The grouping and dosing schedule are shown in Table 21, and the tumor growth curve is shown in Figure 7.
結果見圖7,顯示本揭露中的雙特異性抗體2和雙特異性抗體5均可顯著抑制A431腫瘤的生長。
The results are shown in Figure 7, showing that both
測試例9. 雙特異性抗體對激光致恆河猴脈絡膜新生血管抑制功能的測試Test Example 9. Test of Bispecific Antibody on Laser-induced Choroidal Neovascularization in Rhesus Monkeys
藉由眼玻璃體注射給藥,檢測對恆河猴激光致脈絡膜新生血管滲漏和生長的影響,以驗證雙特異性抗體可玻璃體注射用於老年性黃斑變性(Age-related macular degeneration,AMD)等疾病的治療。具體方法如下: By vitreous injection, the effect on the leakage and growth of choroidal neovascularization induced by laser in rhesus monkeys was detected to verify that the bispecific antibody can be injected into the vitreous for age-related macular degeneration (AMD), etc. disease treatment. The specific method is as follows:
藉由激光圍繞恆河猴眼底黃斑中心凹光凝,誘導眼底脈絡膜血管新生,建立與人類脈絡膜新生血管類似的動物模型。光凝前及光凝後20天進行螢光素眼底血管造影判定造模情況,選擇造模成功的16隻恆河猴(四川格林豪斯生物科技有限公司,生產許可證號:SCXK(川)2014-013,實驗動物質量合格證編號:No:0022202),將恆河猴隨機分為溶媒對照組、雷珠單抗-IgG1(Lucentis,96μg,2μM)組、RG7716(292μg,2μM)組、雙特異性抗體3(396μg,2μM)組,共4組,每組4隻猴。 By laser photocoagulation around the fovea of the rhesus macaque, fundus choroidal angiogenesis was induced, and an animal model similar to human choroidal neovascularization was established. Fluorescein angiography was performed before photocoagulation and 20 days after photocoagulation to determine the condition of modeling, and 16 rhesus monkeys with successful modeling were selected (Sichuan Greenhouse Biotechnology Co., Ltd., production license number: SCXK (Chuan) 2014-013, experimental animal quality certificate number: No: 0022202), rhesus monkeys were randomly divided into vehicle control group, ranibizumab-IgG1 (Lucentis, 96 μg, 2 μM) group, RG7716 ( 292 μg, 2 μM ) group, bispecific antibody 3 ( 396 μg, 2 μM ) group, a total of 4 groups, 4 monkeys in each group.
光凝後21天,Lucentis組、RG7716組、雙特異性抗體3組,分別按96μg、292μg、396μg/眼,雙眼玻璃體注射給予50μL濃度為1.92mg/mL的Lucentis,5.84mg/mL的RG7716,7.92mg/mL的雙特異性抗體3,溶媒對照組給與等體積溶劑。各組動物於給藥後7、14、28天進行眼壓檢查、眼底彩色照相、螢光素眼底血管造影檢查、光學相干斷層掃描(OCT),觀察抗體對脈絡膜新生血管的抑制情況。於給藥後28天取房水100~200μL,分裝100μL進行房水VEGF測定。於給藥後29天實施安樂死後每組取3隻眼球進行HE染色組織學檢查。結果如下:
21 days after photocoagulation, Lucentis group, RG7716 group and 3 bispecific antibody groups were given 96 μg, 292 μg and 396 μg/eye respectively, and 50 μL of 1.92 mg/mL was injected into the vitreous body of both eyes. Lucentis, RG7716 at 5.84 mg/mL,
AMD造模AMD modeling
激光造模後20天,所納入試驗的16隻猴雙眼眼底彩色照相均可見黃斑周圍各9個激光斑,眼底黃斑周圍均可見有激光斑呈高螢光,明顯的螢光素滲漏,且滲漏超過光斑邊緣,激光造模後20天(給藥前),溶媒對照組、Lucentis組、
RG7716組、雙特異性抗體3組4級螢光斑點數分別為46、42、40、45個。上述改變類似臨床脈絡膜新生血管(Choroidal Neovascularization,CNV)改變,提示造模成功。
20 days after laser modeling, 9 laser spots around the macula were seen in the color photography of the fundus of both eyes of the 16 monkeys included in the experiment, and there were laser spots around the macula with high fluorescence, obvious fluorescein leakage, and leakage. Leakage beyond the edge of the spot, 20 days after laser modeling (before administration), vehicle control group, Lucentis group,
The number of grade 4 fluorescent spots in the RG7716 group and the
螢光造影檢查fluoroscopy
Lucentis組、RG7716組、雙特異性抗體3組在給藥後7、14、28天螢光斑面積均有一定程度的縮小,各組螢光滲漏面積改善率、螢光素滲漏面積減少量均優於溶媒對照組,各組4級螢光斑點數與溶媒對照組相比明顯降低。
Lucentis group, RG7716 group, and
28天後,給藥量為2μM雙特異性抗體3組與給藥量2μM的雷珠單抗組和給藥量為2μM的RG7716組,均可降低螢光滲漏面積,並減少螢光斑點數,結果見圖8A和8B。
After 28 days, the 2 μM bispecific
房水VEGFAqueous VEGF
Lucentis組、RG7716組、雙特異性抗體3組在給藥後28天房水VEGF表達量均明顯低於溶媒對照組。雙特異性抗體3組房水VEGF表達量均明顯低於Lucentis組和RG7716組,結果見圖9。 The expression of VEGF in the aqueous humor of Lucentis group, RG7716 group and bispecific antibody group 28 days after administration was significantly lower than that of vehicle control group. The expression of VEGF in the aqueous humor of the three bispecific antibody groups was significantly lower than that of the Lucentis group and the RG7716 group, and the results are shown in Figure 9.
綜上所述,在本試驗條件下,激光CNV模型的恆河猴經雙眼玻璃體單次注射給予396μg/眼劑量的雙特異性抗體3,經視網膜血管螢光造影、光學相干斷層成像、房水VEGF及眼組織病理學檢查,雙特異性抗體3在396μg/眼劑量下對猴CNV具有明顯的抑制作用。
To sum up, under the conditions of this experiment, the rhesus monkeys of the laser CNV model were given a single dose of 396 μg/eye
<110> 大陸商江蘇恆瑞醫藥股份有限公司(JIANGSU HENGRUI MEDICINE CO.,LTD.) 大陸商上海恆瑞醫藥有限公司(SHANGHAI HENGRUI PHARMACEUTICAL CO.,LTD.) <110> JIANGSU HENGRUI MEDICINE CO.,LTD. Mainland business Shanghai Hengrui Pharmaceutical Co., Ltd. (SHANGHAI HENGRUI PHARMACEUTICAL CO.,LTD.)
<120> 特異性结合VEGF和ANG-2的雙特異性抗原结合分子 <120> Bispecific antigen-binding molecule that specifically binds VEGF and ANG-2
<130> 721109CPCT <130> 721109CPCT
<150> CN202010980869.2 <150> CN202010980869.2
<151> 2020-09-17 <151> 2020-09-17
<160> 98 <160> 98
<170> SIPOSequenceListing 1.0 <170> SIPOSequenceListing 1.0
<210> 1 <210> 1
<211> 723 <211> 723
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 肽 <221> Peptides
<223> 帶人Fc標簽的人ANG-2胺基酸序列 <223> Human Fc-tagged human ANG-2 amino acid sequence
<400> 1 <400> 1
<210> 2 <210> 2
<211> 953 <211> 953
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 肽 <221> Peptides
<223> 帶人Fc標簽的Tie2胺基酸序列 <223> Tie2 amino acid sequence with human Fc tag
<400> 2 <400> 2
<210> 3 <210> 3
<211> 118 <211> 118
<212> PRT <212> PRT
<213> 小鼠(Mus musculus) <213> Mouse (Mus musculus)
<220> <220>
<221> 結構域 <221> Domain
<223> HR54鼠源重鏈可變區序列 <223> HR54 murine heavy chain variable region sequence
<400> 3 <400> 3
<210> 4 <210> 4
<211> 106 <211> 106
<212> PRT <212> PRT
<213> 小鼠(Mus musculus) <213> Mouse (Mus musculus)
<220> <220>
<221> 結構域 <221> Domain
<223> HR54鼠源輕鏈可變區序列 <223> HR54 murine light chain variable region sequence
<400> 4 <400> 4
<210> 5 <210> 5
<211> 118 <211> 118
<212> PRT <212> PRT
<213> 小鼠(Mus musculus) <213> Mouse (Mus musculus)
<220> <220>
<221> 結構域 <221> Domain
<223> CP33鼠源重鏈可變區序列 <223> CP33 murine heavy chain variable region sequence
<400> 5 <400> 5
<210> 6 <210> 6
<211> 106 <211> 106
<212> PRT <212> PRT
<213> 小鼠(Mus musculus) <213> Mouse (Mus musculus)
<220> <220>
<221> 結構域 <221> Domain
<223> CP33鼠源輕鏈可變區序列 <223> CP33 murine light chain variable region sequence
<400> 6 <400> 6
<210> 7 <210> 7
<211> 5 <211> 5
<212> PRT <212> PRT
<213> 小鼠(Mus musculus) <213> Mouse (Mus musculus)
<220> <220>
<221> 結構域 <221> Domain
<223> HR54 HCDR1 <223> HR54 HCDR1
<400> 7 <400> 7
<210> 8 <210> 8
<211> 17 <211> 17
<212> PRT <212> PRT
<213> 小鼠(Mus musculus) <213> Mouse (Mus musculus)
<220> <220>
<221> 結構域 <221> Domain
<223> HR54 HCDR2 <223> HR54 HCDR2
<400> 8 <400> 8
<210> 9 <210> 9
<211> 9 <211> 9
<212> PRT <212> PRT
<213> 小鼠(Mus musculus) <213> Mouse (Mus musculus)
<220> <220>
<221> 結構域 <221> Domain
<223> HR54 HCDR3 <223> HR54 HCDR3
<400> 9 <400> 9
<210> 10 <210> 10
<211> 11 <211> 11
<212> PRT <212> PRT
<213> 小鼠(Mus musculus) <213> Mouse (Mus musculus)
<220> <220>
<221> 結構域 <221> Domain
<223> HR54 LCDR1 <223> HR54 LCDR1
<400> 10 <400> 10
<210> 11 <210> 11
<211> 7 <211> 7
<212> PRT <212> PRT
<213> 小鼠(Mus musculus) <213> Mouse (Mus musculus)
<220> <220>
<221> 結構域 <221> Domain
<223> HR54 LCDR2 <223> HR54 LCDR2
<400> 11 <400> 11
<210> 12 <210> 12
<211> 8 <211> 8
<212> PRT <212> PRT
<213> 小鼠(Mus musculus) <213> Mouse (Mus musculus)
<220> <220>
<221> 結構域 <221> Domain
<223> HR54 LCDR3 <223> HR54 LCDR3
<400> 12 <400> 12
<210> 13 <210> 13
<211> 17 <211> 17
<212> PRT <212> PRT
<213> 小鼠(Mus musculus) <213> Mouse (Mus musculus)
<220> <220>
<221> 結構域 <221> Domain
<223> CP33 HCDR2 <223> CP33 HCDR2
<400> 13 <400> 13
<210> 14 <210> 14
<211> 9 <211> 9
<212> PRT <212> PRT
<213> 小鼠(Mus musculus) <213> Mouse (Mus musculus)
<220> <220>
<221> 結構域 <221> Domain
<223> CP33 HCDR3 <223> CP33 HCDR3
<400> 14 <400> 14
<210> 15 <210> 15
<211> 11 <211> 11
<212> PRT <212> PRT
<213> 小鼠(Mus musculus) <213> Mouse (Mus musculus)
<220> <220>
<221> 結構域 <221> Domain
<223> CP33 LCDR1 <223> CP33 LCDR1
<400> 15 <400> 15
<210> 16 <210> 16
<211> 8 <211> 8
<212> PRT <212> PRT
<213> 小鼠(Mus musculus) <213> Mouse (Mus musculus)
<220> <220>
<221> 結構域 <221> Domain
<223> CP33 LCDR3 <223> CP33 LCDR3
<400> 16 <400> 16
<210> 17 <210> 17
<211> 106 <211> 106
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> hu HR54VL1 <223> hu HR54VL1
<400> 17 <400> 17
<210> 18 <210> 18
<211> 106 <211> 106
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> hu HR54VL2 <223> hu HR54VL2
<400> 18 <400> 18
<210> 19 <210> 19
<211> 106 <211> 106
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huHR54VL3 <223> huHR54VL3
<400> 19 <400> 19
<210> 20 <210> 20
<211> 118 <211> 118
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huHR54VH1 <223> huHR54VH1
<400> 20 <400> 20
<210> 21 <210> 21
<211> 118 <211> 118
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huHR54VH2 <223> huHR54VH2
<400> 21 <400> 21
<210> 22 <210> 22
<211> 17 <211> 17
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> HR54a HCDR2 <223> HR54a HCDR2
<400> 22 <400> 22
<210> 23 <210> 23
<211> 9 <211> 9
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> HR54a HCDR3 <223> HR54a HCDR3
<400> 23 <400> 23
<210> 24 <210> 24
<211> 17 <211> 17
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> HR54b HCDR2 <223> HR54b HCDR2
<400> 24 <400> 24
<210> 25 <210> 25
<211> 9 <211> 9
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> HR54c HCDR3 <223> HR54c HCDR3
<400> 25 <400> 25
<210> 26 <210> 26
<211> 9 <211> 9
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> HR54d HCDR3 <223> HR54d HCDR3
<400> 26 <400> 26
<210> 27 <210> 27
<211> 9 <211> 9
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> HR54n HCDR3 <223> HR54n HCDR3
<400> 27 <400> 27
<210> 28 <210> 28
<211> 118 <211> 118
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huHR54VH2a <223> huHR54VH2a
<400> 28 <400> 28
<210> 29 <210> 29
<211> 118 <211> 118
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huHR54VH2b <223> huHR54VH2b
<400> 29 <400> 29
<210> 30 <210> 30
<211> 118 <211> 118
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huHR54VH2c <223> huHR54VH2c
<400> 30 <400> 30
<210> 31 <210> 31
<211> 118 <211> 118
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huHR54VH2d <223> huHR54VH2d
<400> 31 <400> 31
<210> 32 <210> 32
<211> 118 <211> 118
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huHR54VH2e <223> huHR54VH2e
<400> 32 <400> 32
<210> 33 <210> 33
<211> 118 <211> 118
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huHR54VH2f <223> huHR54VH2f
<400> 33 <400> 33
<210> 34 <210> 34
<211> 118 <211> 118
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huHR54VH2g <223> huHR54VH2g
<400> 34 <400> 34
<210> 35 <210> 35
<211> 118 <211> 118
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huHR54VH2k <223> huHR54VH2k
<400> 35 <400> 35
<210> 36 <210> 36
<211> 118 <211> 118
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huHR54VH2m <223> huHR54VH2m
<400> 36 <400> 36
<210> 37 <210> 37
<211> 118 <211> 118
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huHR54VH2n <223> huHR54VH2n
<400> 37 <400> 37
<210> 38 <210> 38
<211> 17 <211> 17
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huHR54 HCDR2通式 <223> huHR54 HCDR2 general formula
<220> <220>
<221> 結構域 <221> Domain
<222> (4)..(4) <222> (4)..(4)
<223> Xaa選自Asp或Glu。 <223> Xaa is selected from Asp or Glu.
<220> <220>
<221> 結構域 <221> Domain
<222> (5)..(5) <222> (5)..(5)
<223> X選自Asp或Asn。 <223> X is selected from Asp or Asn.
<400> 38 <400> 38
<210> 39 <210> 39
<211> 9 <211> 9
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huHR54 HCDR3通式 <223> huHR54 HCDR3 general formula
<220> <220>
<221> 結構域 <221> Domain
<222> (1)..(1) <222> (1)..(1)
<223> Xaa選自Asp或Asn。 <223> Xaa is selected from Asp or Asn.
<220> <220>
<221> 結構域 <221> Domain
<222> (2)..(2) <222> (2)..(2)
<223> Xaa選自Glu或Gln。 <223> Xaa is selected from Glu or Gln.
<220> <220>
<221> 結構域 <221> Domain
<222> (6)..(6) <222> (6)..(6)
<223> Xaa選自Cys,Ser或Val。 <223> Xaa is selected from Cys, Ser or Val.
<400> 39 <400> 39
<210> 40 <210> 40
<211> 106 <211> 106
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huCP33VL1 <223> huCP33VL1
<400> 40 <400> 40
<210> 41 <210> 41
<211> 106 <211> 106
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huCP33VL2 <223> huCP33VL2
<400> 41 <400> 41
<210> 42 <210> 42
<211> 106 <211> 106
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huCP33VL3 <223> huCP33VL3
<400> 42 <400> 42
<210> 43 <210> 43
<211> 106 <211> 106
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huCP33VL4 <223> huCP33VL4
<400> 43 <400> 43
<210> 44 <210> 44
<211> 118 <211> 118
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huCP33VH1 <223> huCP33VH1
<400> 44 <400> 44
<210> 45 <210> 45
<211> 118 <211> 118
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huCP33VH2 <223> huCP33VH2
<400> 45 <400> 45
<210> 46 <210> 46
<211> 118 <211> 118
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huCP33VH3 <223> huCP33VH3
<400> 46 <400> 46
<210> 47 <210> 47
<211> 330 <211> 330
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> 重鏈恆定區 <223> Heavy chain constant region
<400> 47 <400> 47
<210> 48 <210> 48
<211> 107 <211> 107
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> 輕鏈恆定區 <223> Light chain constant region
<400> 48 <400> 48
<210> 49 <210> 49
<211> 448 <211> 448
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> huHR54-01重鏈 <223> huHR54-01 heavy chain
<400> 49 <400> 49
<210> 50 <210> 50
<211> 213 <211> 213
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huHR54-01輕鏈 <223> huHR54-01 light chain
<400> 50 <400> 50
<210> 51 <210> 51
<211> 448 <211> 448
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> huHR54-04重鏈 <223> huHR54-04 heavy chain
<400> 51 <400> 51
<210> 52 <210> 52
<211> 448 <211> 448
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> huHR54-12重鏈 <223> huHR54-12 heavy chain
<400> 52 <400> 52
<210> 53 <210> 53
<211> 448 <211> 448
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> huCP33-01抗體重鏈 <223> huCP33-01 antibody heavy chain
<400> 53 <400> 53
<210> 54 <210> 54
<211> 213 <211> 213
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> huCP33-01抗體輕鏈 <223> huCP33-01 antibody light chain
<400> 54 <400> 54
<210> 55 <210> 55
<211> 448 <211> 448
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> huCP33-05重鏈 <223> huCP33-05 heavy chain
<400> 55 <400> 55
<210> 56 <210> 56
<211> 107 <211> 107
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> 雷珠單抗的輕鏈可變區 <223> Light chain variable region of ranibizumab
<400> 56 <400> 56
<210> 57 <210> 57
<211> 214 <211> 214
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> 雷珠單抗的輕鏈 <223> Light chain of ranibizumab
<400> 57 <400> 57
<210> 58 <210> 58
<211> 123 <211> 123
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> 雷珠單抗的重鏈可變區 <223> Heavy chain variable region of ranibizumab
<400> 58 <400> 58
<210> 59 <210> 59
<211> 453 <211> 453
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> 雷珠單抗重鏈1
<223> ranibizumab
<400> 59 <400> 59
<210> 60 <210> 60
<211> 453 <211> 453
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> 雷珠單抗重鏈2
<223> ranibizumab
<400> 60 <400> 60
<210> 61 <210> 61
<211> 5 <211> 5
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> 雷珠單抗HCDR1 <223> ranibizumab HCDR1
<400> 61 <400> 61
<210> 62 <210> 62
<211> 17 <211> 17
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> 雷珠單抗HCDR2 <223> ranibizumab HCDR2
<400> 62 <400> 62
<210> 63 <210> 63
<211> 14 <211> 14
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> 雷珠單抗HCDR3 <223> ranibizumab HCDR3
<400> 63 <400> 63
<210> 64 <210> 64
<211> 11 <211> 11
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> 雷珠單抗LCDR1 <223> ranibizumab LCDR1
<400> 64 <400> 64
<210> 65 <210> 65
<211> 7 <211> 7
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> 雷珠單抗LCDR2 <223> ranibizumab LCDR2
<400> 65 <400> 65
<210> 66 <210> 66
<211> 10 <211> 10
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> 雷珠單抗LCDR3 <223> ranibizumab LCDR3
<400> 66 <400> 66
<210> 67 <210> 67
<211> 254 <211> 254
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 肽 <221> Peptides
<223> huHR54-01-scFv <223> huHR54-01-scFv
<400> 67 <400> 67
<210> 68 <210> 68
<211> 254 <211> 254
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 肽 <221> Peptides
<223> huHR54-04-scFv <223> huHR54-04-scFv
<400> 68 <400> 68
<210> 69 <210> 69
<211> 254 <211> 254
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 肽 <221> Peptides
<223> huHR54-12-scFv <223> huHR54-12-scFv
<400> 69 <400> 69
<210> 70 <210> 70
<211> 254 <211> 254
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 肽 <221> Peptides
<223> huCP33-01-scFv <223> huCP33-01-scFv
<400> 70 <400> 70
<210> 71 <210> 71
<211> 254 <211> 254
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 肽 <221> Peptides
<223> huCP33-05-scFv <223> huCP33-05-scFv
<400> 71 <400> 71
<210> 72 <210> 72
<211> 118 <211> 118
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huHR54VH2-C <223> huHR54VH2-C
<400> 72 <400> 72
<210> 73 <210> 73
<211> 118 <211> 118
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huHR54VH2f-C <223> huHR54VH2f-C
<400> 73 <400> 73
<210> 74 <210> 74
<211> 106 <211> 106
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huHR54VL1-C <223> huHR54VL1-C
<400> 74 <400> 74
<210> 75 <210> 75
<211> 118 <211> 118
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huCP33VH2-C <223> huCP33VH2-C
<400> 75 <400> 75
<210> 76 <210> 76
<211> 106 <211> 106
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 結構域 <221> Domain
<223> huCP33VL1-C <223> huCP33VL1-C
<400> 76 <400> 76
<210> 77 <210> 77
<211> 254 <211> 254
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 肽 <221> Peptides
<223> huHR54-04-scFv-CC <223> huHR54-04-scFv-CC
<400> 77 <400> 77
<210> 78 <210> 78
<211> 254 <211> 254
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 肽 <221> Peptides
<223> huHR54-12-scFv-CC <223> huHR54-12-scFv-CC
<400> 78 <400> 78
<210> 79 <210> 79
<211> 254 <211> 254
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 肽 <221> Peptides
<223> huCP33-05-scFv-CC <223> huCP33-05-scFv-CC
<400> 79 <400> 79
<210> 80 <210> 80
<211> 708 <211> 708
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> 雙特異性抗體1第一條鏈
<223>
<400> 80 <400> 80
<210> 81 <210> 81
<211> 708 <211> 708
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> 雙特異性抗體2第一條鏈
<223>
<400> 81 <400> 81
<210> 82 <210> 82
<211> 708 <211> 708
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> 雙特異抗體3第一條鏈
<223>
<400> 82 <400> 82
<210> 83 <210> 83
<211> 708 <211> 708
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> 雙特異性抗體4第一條鏈 <223> Bispecific antibody 4 first chain
<400> 83 <400> 83
<210> 84 <210> 84
<211> 452 <211> 452
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> 雙特異性抗體5第一重鏈:抗ANG-2重鏈
<223>
<400> 84 <400> 84
<210> 85 <210> 85
<211> 211 <211> 211
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> 雙特異性抗體5第一輕鏈:抗ANG-2輕鏈
<223>
<400> 85 <400> 85
<210> 86 <210> 86
<211> 453 <211> 453
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> 雙特異性抗體5第二重鏈:抗VEGF重鏈
<223>
<400> 86 <400> 86
<210> 87 <210> 87
<211> 452 <211> 452
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> 雙特異性抗體6第一重鏈:抗ANG-2重鏈 <223> Bispecific antibody 6 first heavy chain: anti-ANG-2 heavy chain
<400> 87 <400> 87
<210> 88 <210> 88
<211> 211 <211> 211
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> 雙特異性抗體6第一輕鏈:抗ANG-2輕鏈 <223> Bispecific antibody 6 first light chain: anti-ANG-2 light chain
<400> 88 <400> 88
<210> 89 <210> 89
<211> 463 <211> 463
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> RG7716抗-ANG-2重鏈 <223> RG7716 anti-ANG-2 heavy chain
<400> 89 <400> 89
<210> 90 <210> 90
<211> 213 <211> 213
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> RG7716抗ANG-2輕鏈 <223> RG7716 anti-ANG-2 light chain
<400> 90 <400> 90
<210> 91 <210> 91
<211> 453 <211> 453
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> RG7716抗VEGF重鏈 <223> RG7716 anti-VEGF heavy chain
<400> 91 <400> 91
<210> 92 <210> 92
<211> 214 <211> 214
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> RG7716抗VEGF輕鏈 <223> RG7716 anti-VEGF light chain
<400> 92 <400> 92
<210> 93 <210> 93
<211> 453 <211> 453
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> 阿瓦斯汀重鏈 <223> Avastin heavy chain
<400> 93 <400> 93
<210> 94 <210> 94
<211> 214 <211> 214
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> 阿瓦斯汀輕鏈 <223> Avastin Light Chain
<400> 94 <400> 94
<210> 95 <210> 95
<211> 463 <211> 463
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> RG7221抗ANG2重鏈 <223> RG7221 anti-ANG2 heavy chain
<400> 95 <400> 95
<210> 96 <210> 96
<211> 213 <211> 213
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> RG7221抗ANG2輕鏈 <223> RG7221 anti-ANG2 light chain
<400> 96 <400> 96
<210> 97 <210> 97
<211> 453 <211> 453
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> RG7221抗VEGF重鏈 <223> RG7221 anti-VEGF heavy chain
<400> 97 <400> 97
<210> 98 <210> 98
<211> 214 <211> 214
<212> PRT <212> PRT
<213> 人工序列(Artificial Sequence) <213> Artificial Sequence
<220> <220>
<221> 鏈 <221> Chain
<223> RG7221抗VEGF輕鏈 <223> RG7221 anti-VEGF light chain
<400> 98 <400> 98
Claims (32)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010980869 | 2020-09-17 | ||
| CN202010980869.2 | 2020-09-17 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| TW202221027A true TW202221027A (en) | 2022-06-01 |
Family
ID=80775921
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| TW110134911A TW202221027A (en) | 2020-09-17 | 2021-09-17 | Bispecific antigen binding molecules that specifically bind to vegf and ang-2 |
Country Status (3)
| Country | Link |
|---|---|
| CN (1) | CN116234574B (en) |
| TW (1) | TW202221027A (en) |
| WO (1) | WO2022057888A1 (en) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU2023404529A1 (en) * | 2022-12-01 | 2025-07-10 | Innovent Biologics (Suzhou) Co., Ltd. | Preparation containing bispecific binding molecule binding to vegf and ang2 and use thereof |
Family Cites Families (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8268314B2 (en) * | 2008-10-08 | 2012-09-18 | Hoffmann-La Roche Inc. | Bispecific anti-VEGF/anti-ANG-2 antibodies |
| UY32920A (en) * | 2009-10-02 | 2011-04-29 | Boehringer Ingelheim Int | BISPECIFIC UNION MOLECULES FOR ANTI-ANGIOGENESIS THERAPY |
| TWI426920B (en) * | 2010-03-26 | 2014-02-21 | Hoffmann La Roche | Bispecific, bivalent anti-VEGF/anti-ANG-2 antibody |
| CN102250247B (en) * | 2011-06-15 | 2013-06-19 | 常州亚当生物技术有限公司 | Bispecific antibody to VEGF/ANG2 and application thereof |
| SMT202100621T1 (en) * | 2012-07-13 | 2022-01-10 | Roche Glycart Ag | Bispecific anti-vegf/anti-ang-2 antibodies and their use in the treatment of ocular vascular diseases |
| KR20150063728A (en) * | 2013-12-02 | 2015-06-10 | 삼성전자주식회사 | Anti-VEGF-C/anti-Ang2 bispecific antibody |
| WO2018114728A1 (en) * | 2016-12-20 | 2018-06-28 | F. Hoffmann-La Roche Ag | Combination therapy with a bispecific anti-ang2/vegf antibody and a bispecific anti-her2 antibody |
| CN119733046A (en) * | 2018-02-06 | 2025-04-01 | 豪夫迈·罗氏有限公司 | Treatment of eye diseases |
| MX2021004774A (en) * | 2018-10-29 | 2021-08-24 | Hoffmann La Roche | Antibody formulation. |
-
2021
- 2021-09-17 TW TW110134911A patent/TW202221027A/en unknown
- 2021-09-17 WO PCT/CN2021/119017 patent/WO2022057888A1/en not_active Ceased
- 2021-09-17 CN CN202180054890.2A patent/CN116234574B/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| WO2022057888A1 (en) | 2022-03-24 |
| CN116234574B (en) | 2024-12-20 |
| CN116234574A (en) | 2023-06-06 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN112955473B (en) | Bispecific antibody that specifically binds VEGF and ANG2 | |
| US20250376519A1 (en) | Antigen-binding molecule specifically binding to dll3 and cd3, and pharmaceutical use thereof | |
| TW202400648A (en) | Anti-ccr8 antibody and uses thereof | |
| KR20230132544A (en) | Novel anti-gremlin 1 antibody | |
| CN113840836A (en) | Anti-connective tissue growth factor antibody and its application | |
| CN118043350A (en) | Anti-VEGF A and VEGF C bispecific antibodies and uses thereof | |
| CN116234574B (en) | Bispecific antigen binding molecules specifically binding to VEGF and ANG-2 | |
| WO2022156739A1 (en) | Antigen binding molecule specifically binding to bcma and cd3, and medical use thereof | |
| US20250215083A1 (en) | Antigen-binding molecule specifically binding to gprc5d and cd3 and medical use thereof | |
| US20250043022A1 (en) | Antigen-binding molecule specifically binding to psma and cd3, and pharmaceutical use thereof | |
| TW202413437A (en) | Antigen binding molecules specifically binding to gucy2c and cd3 and their medical uses | |
| TW202504927A (en) | Anti-npr1 antibody and the pharmaceutical use thereof | |
| WO2026008055A1 (en) | Anti-kit antibody and pharmaceutical use thereof | |
| HK40051458A (en) | Bispecific antibody specifically bound to vegf and ang2 | |
| WO2023198042A1 (en) | Antigen-binding molecule specifically binding to egfr and cd28, and medical use thereof | |
| WO2024222409A1 (en) | Prolactin receptor antibody and use thereof | |
| HK40105696A (en) | Novel mesothelin antibody and composition comprising the same | |
| JP2024544961A (en) | Anti-CLDN18.2 monoclonal antibodies and uses thereof | |
| HK40054516B (en) | Novel mesothelin antibody and composition comprising the same | |
| CN115572330A (en) | A kind of specific antibody and its preparation method and application | |
| CN115850499A (en) | Antigen binding molecule specifically binding to ANGPTL3 and PCSK9 and medical application thereof |