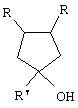RU2250892C2 - Method for production of 1,3,4-trialkylcyclopentane-1-ols - Google Patents
Method for production of 1,3,4-trialkylcyclopentane-1-ols Download PDFInfo
- Publication number
- RU2250892C2 RU2250892C2 RU2003114593/04A RU2003114593A RU2250892C2 RU 2250892 C2 RU2250892 C2 RU 2250892C2 RU 2003114593/04 A RU2003114593/04 A RU 2003114593/04A RU 2003114593 A RU2003114593 A RU 2003114593A RU 2250892 C2 RU2250892 C2 RU 2250892C2
- Authority
- RU
- Russia
- Prior art keywords
- etalcl
- zrcl
- nicl
- production
- general formula
- Prior art date
Links
- 238000004519 manufacturing process Methods 0.000 title abstract description 7
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 claims abstract description 14
- 238000000034 method Methods 0.000 claims abstract description 14
- 239000004711 α-olefin Substances 0.000 claims abstract description 12
- 238000006243 chemical reaction Methods 0.000 claims abstract description 11
- 239000011777 magnesium Substances 0.000 claims abstract description 11
- 239000003054 catalyst Substances 0.000 claims abstract description 10
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 claims abstract description 7
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 claims abstract description 4
- 239000012300 argon atmosphere Substances 0.000 claims abstract description 4
- 229910052749 magnesium Inorganic materials 0.000 claims abstract description 4
- HJIYKFHFIDDFLJ-UHFFFAOYSA-L dichloronickel phosphane Chemical compound P.Cl[Ni]Cl HJIYKFHFIDDFLJ-UHFFFAOYSA-L 0.000 claims abstract description 3
- 229910007926 ZrCl Inorganic materials 0.000 claims description 10
- MGDOJPNDRJNJBK-UHFFFAOYSA-N ethylaluminum Chemical compound [Al].C[CH2] MGDOJPNDRJNJBK-UHFFFAOYSA-N 0.000 claims 1
- 238000003786 synthesis reaction Methods 0.000 abstract description 5
- 230000003993 interaction Effects 0.000 abstract description 4
- 239000007866 anti-wear additive Substances 0.000 abstract description 2
- UAIZDWNSWGTKFZ-UHFFFAOYSA-L ethylaluminum(2+);dichloride Chemical compound CC[Al](Cl)Cl UAIZDWNSWGTKFZ-UHFFFAOYSA-L 0.000 abstract description 2
- 230000007062 hydrolysis Effects 0.000 abstract description 2
- 238000006460 hydrolysis reaction Methods 0.000 abstract description 2
- 239000003921 oil Substances 0.000 abstract description 2
- 229910007928 ZrCl2 Inorganic materials 0.000 abstract 2
- 230000000975 bioactive effect Effects 0.000 abstract 1
- 150000001875 compounds Chemical class 0.000 abstract 1
- 230000000694 effects Effects 0.000 abstract 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 abstract 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 abstract 1
- 239000000126 substance Substances 0.000 abstract 1
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 9
- UGFAIRIUMAVXCW-UHFFFAOYSA-N Carbon monoxide Chemical compound [O+]#[C-] UGFAIRIUMAVXCW-UHFFFAOYSA-N 0.000 description 8
- 229910002091 carbon monoxide Inorganic materials 0.000 description 8
- PTWYHRSIWNDYLT-UHFFFAOYSA-N 3,4-dibutyl-1-methylcyclopentan-1-ol Chemical compound CCCCC1CC(C)(O)CC1CCCC PTWYHRSIWNDYLT-UHFFFAOYSA-N 0.000 description 5
- LIKMAJRDDDTEIG-UHFFFAOYSA-N 1-hexene Chemical compound CCCCC=C LIKMAJRDDDTEIG-UHFFFAOYSA-N 0.000 description 4
- -1 Cyclic Alcohols Chemical class 0.000 description 3
- 150000002148 esters Chemical class 0.000 description 3
- VTBOTOBFGSVRMA-UHFFFAOYSA-N 1-Methylcyclohexanol Chemical compound CC1(O)CCCCC1 VTBOTOBFGSVRMA-UHFFFAOYSA-N 0.000 description 2
- KWKAKUADMBZCLK-UHFFFAOYSA-N 1-octene Chemical compound CCCCCCC=C KWKAKUADMBZCLK-UHFFFAOYSA-N 0.000 description 2
- OFBQJSOFQDEBGM-UHFFFAOYSA-N Pentane Chemical compound CCCCC OFBQJSOFQDEBGM-UHFFFAOYSA-N 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- 150000001735 carboxylic acids Chemical class 0.000 description 2
- 239000003153 chemical reaction reagent Substances 0.000 description 2
- 125000004494 ethyl ester group Chemical group 0.000 description 2
- FKRCODPIKNYEAC-UHFFFAOYSA-N ethyl propionate Chemical compound CCOC(=O)CC FKRCODPIKNYEAC-UHFFFAOYSA-N 0.000 description 2
- 239000000843 powder Substances 0.000 description 2
- XBDQKXXYIPTUBI-UHFFFAOYSA-N Propionic acid Chemical compound CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 1
- CUJRVFIICFDLGR-UHFFFAOYSA-N acetylacetonate Chemical compound CC(=O)[CH-]C(C)=O CUJRVFIICFDLGR-UHFFFAOYSA-N 0.000 description 1
- 239000013543 active substance Substances 0.000 description 1
- 125000001931 aliphatic group Chemical group 0.000 description 1
- AZDRQVAHHNSJOQ-UHFFFAOYSA-N alumane Chemical class [AlH3] AZDRQVAHHNSJOQ-UHFFFAOYSA-N 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 238000001460 carbon-13 nuclear magnetic resonance spectrum Methods 0.000 description 1
- 230000008878 coupling Effects 0.000 description 1
- 238000010168 coupling process Methods 0.000 description 1
- 238000005859 coupling reaction Methods 0.000 description 1
- ZMMRKRFMSDTOLV-UHFFFAOYSA-N cyclopenta-1,3-diene zirconium Chemical compound [Zr].C1C=CC=C1.C1C=CC=C1 ZMMRKRFMSDTOLV-UHFFFAOYSA-N 0.000 description 1
- 239000004210 ether based solvent Substances 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 150000004820 halides Chemical class 0.000 description 1
- 238000002329 infrared spectrum Methods 0.000 description 1
- LZKLAOYSENRNKR-LNTINUHCSA-N iron;(z)-4-oxoniumylidenepent-2-en-2-olate Chemical compound [Fe].C\C(O)=C\C(C)=O.C\C(O)=C\C(C)=O.C\C(O)=C\C(C)=O LZKLAOYSENRNKR-LNTINUHCSA-N 0.000 description 1
- 238000001819 mass spectrum Methods 0.000 description 1
- TVMXDCGIABBOFY-UHFFFAOYSA-N n-Octanol Natural products CCCCCCCC TVMXDCGIABBOFY-UHFFFAOYSA-N 0.000 description 1
- 239000012044 organic layer Substances 0.000 description 1
- 239000003973 paint Substances 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 238000000425 proton nuclear magnetic resonance spectrum Methods 0.000 description 1
- 239000011541 reaction mixture Substances 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 230000003595 spectral effect Effects 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 239000002966 varnish Substances 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
Landscapes
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
Description
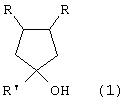
Предлагаемое изобретение относится к органической химии, в частности к способу получения 1,3,4-триалкилциклопентан-1-олов общей формулы (I):The present invention relates to organic chemistry, in particular to a method for producing 1,3,4-trialkylcyclopentan-1-ol of the general formula (I):
, где R=н-С4Н9, н-С6Н13, н-C8H17; R1=СН3, С2Н5 where R = n — C 4 H 9 , n — C 6 H 13 , n — C 8 H 17 ; R 1 = CH 3 , C 2 H 5
Циклоалканолы и их производные могут найти применение в тонком органическом синтезе, в производстве лакокрасочных материалов, высокоэффективных противозадирных и противоизносных присадок к маслам, биологически активных веществ.Cycloalkanols and their derivatives can be used in fine organic synthesis, in the production of paints and varnishes, highly effective anti-seize and anti-wear additives for oils, biologically active substances.
Известен способ ([1] Nenitzescu C.D., Necsoin I. The Synthesis of Cyclic Alcohols and Olefms by the Interaction of Dimagnesium Halides and Esters // J. Amer. Chem. Soc., 1950, 72, 3483-3486) получения циклоалканолов, например, 1-метилциклогексан-1-ола (2) взаимодействием 1,5-димагнийпентана с эквимольным количеством этилацетата по схеме:A known method ([1] Nenitzescu CD, Necsoin I. The Synthesis of Cyclic Alcohols and Olefms by the Interaction of Dimagnesium Halides and Esters // J. Amer. Chem. Soc., 1950, 72, 3483-3486) for producing cycloalkanols, for example , 1-methylcyclohexan-1-ol (2) by the interaction of 1,5-dimagnesium pentane with an equimolar amount of ethyl acetate according to the scheme:
Известный способ не позволяет получать 1,3,4-триалкилциклопентан-1-олы общей формулы (1).The known method does not allow to obtain 1,3,4-trialkylcyclopentan-1-ol of the General formula (1).
Известен способ ([2]. Swanson D.R., Rousset C.J., E.-I. Negishi, Takahashi Т., Seki Т., Saburi M., Uskida J. Regioselective and Diastereoselective Alkyl-Alkene and Alkene-Alkene Coupling Promoted by Zirconocene and Hafnocene // J. Org. Chem. 1989, V.54, №15, Р.3521-3523) получения транс-3,4-диалкилциклопентан-1-олов взаимодействием цирконацендихлорида (Cp2ZrCl2) с двукратным избытком алкильных производных лития с последующей последовательной обработкой реакционной массы α -олефинами, окисью углерода (СО) и водой по схеме:The known method ([2]. Swanson DR, Rousset CJ, E.-I. Negishi, Takahashi T., Seki T., Saburi M., Uskida J. Regioselective and Diastereoselective Alkyl-Alkene and Alkene-Alkene Coupling Promoted by Zirconocene and Hafnocene // J. Org. Chem. 1989, V.54, No. 15, P.3521-3523) for the preparation of trans-3,4-dialkylcyclopentan-1-ol by the reaction of zirconacenedichloride (Cp 2 ZrCl 2 ) with a twofold excess of alkyl lithium derivatives followed by sequential treatment of the reaction mass with α-olefins, carbon monoxide (CO) and water according to the scheme:
R=Et, n-HexR = Et, n-Hex
Известным способом не могут быть получены 1,3,4-триалкилциклопентан-1-олы (1).In a known manner, 1,3,4-trialkylcyclopentan-1-oles (1) cannot be obtained.
Предлагается новый способ синтеза 1,3,4-триалкилциклопентан-1-олов(1).A new method is proposed for the synthesis of 1,3,4-trialkylcyclopentan-1-ol (1).
Сущность способа заключается во взаимодействии α -олефинов общей формулы RCH=CH2, где R=н-C4H9, н-C6H13, н-C8H17, с этилалюминийдихлоридом (EtAlCl2) и магнием (Mg, порошок), взятыми в мольном соотношении RCH=CH2:EtAlCl2:Mg=10:(5-7):(5-7), в присутствии катализатора цирконацендихлорида Cp2ZrCl2, взятом в мольном соотношении Cp2ZrCl2 к α -олефину, равном 0,3-0,7, в атмосфере аргона при комнатной температуре (20-21° С) и атмосферном давлении в тетрагидрофуране (ТГФ) с последующим добавлением к реакционной массе катализатора фосфинового комплекса хлорида никеля [NiCl2· 2Ph3P] при мольном соотношении [NiCl2· 2Ph3P] к α -олефину, равном 0,8-1,2, и этилового эфира карбоновой кислоты (R1CO2Et, где R1=CH3, C2H5) в трехкратном мольном избытке по отношению к EtAlCl2, с последующим перемешиванием реакционной массы при комнатной температуре (20-21° С) в течение 6-8 часов. Общий выход 1,3,4-триалкилциклопентан-1-олов (1) составляет после гидролиза реакционной массы 50-74%. Реакция протекает по схеме:The essence of the method consists in the interaction of α-olefins of the general formula RCH = CH 2 , where R = n-C 4 H 9 , n-C 6 H 13 , n-C 8 H 17 , with ethyl aluminum dichloride (EtAlCl 2 ) and magnesium (Mg, powder), taken in a molar ratio of RCH = CH 2 : EtAlCl 2 : Mg = 10: (5-7) :( 5-7), in the presence of a catalyst of zirconacide dichloride Cp 2 ZrCl 2 , taken in a molar ratio of Cp 2 ZrCl 2 to α α-olefin, equal to 0.3-0.7, in an argon atmosphere at room temperature (20-21 ° C) and atmospheric pressure in tetrahydrofuran (THF), followed by the addition of nickel chloride phosphine complex [NiCl 2 · 2Ph 3 to the reaction mass of the catalyst P] when the molar ratio of [NiCl 2 · 2Ph 3 P] to the α-olefin, equal to 0.8-1.2, and ethyl carboxylic acid ester (R 1 CO 2 Et, where R 1 = CH 3 , C 2 H 5 ) in triplicate molar excess with respect to EtAlCl 2 , followed by stirring of the reaction mixture at room temperature (20-21 ° C) for 6-8 hours. The total yield of 1,3,4-trialkylcyclopentan-1-ol (1) after hydrolysis of the reaction mass is 50-74%. The reaction proceeds according to the scheme:
R=н-C4H9, н-С6Н13, н-C8H17; R=CH3, С2Н5 R = n-C 4 H 9 , n-C 6 H 13 , n-C 8 H 17 ; R = CH 3 , C 2 H 5
1,3,4-триалкилциклопентан-1-олы (1) образуются только лишь с участием EtAlCl2, Mg, α -олефинов, этиловых эфиров карбоновых кислот и катализаторов Cp2ZrCl2, [NiCl2· 2Ph3P]. В присутствии других соединений алюминия (например, Bu
Реакции проводили с использованием эфирных растворителей (ТГФ). В других растворителях (например, алифатические, ароматические) реакция не идет.Reactions were carried out using ether solvents (THF). In other solvents (for example, aliphatic, aromatic), the reaction does not proceed.
Существенные отличия предлагаемого способа.Significant differences of the proposed method.
В предлагаемом способе используются в качестве исходных соединений EtAlCl2, магний, сложные эфиры (этиловые эфиры карбоновых кислот) и катализаторы Cp2ZrCl2, [NiCl2· 2Ph3P]. В известном способе в качестве исходных реагентов применяются алкильные производные лития, газообразный СО и Cp2ZrCl2 в стехиометрических количествах.In the proposed method, EtAlCl 2 , magnesium, esters (ethyl esters of carboxylic acids) and catalysts Cp 2 ZrCl 2 , [NiCl 2 · 2Ph 3 P] are used as starting compounds. In the known method, alkyl lithium derivatives, gaseous CO, and Cp 2 ZrCl 2 in stoichiometric amounts are used as starting reagents.
Предлагаемый способ позволяет получать 1,3,4-триалкилциклопентан-1-олы (1) исходя из доступных исходных реагентов в мягких условиях (20-21° С).The proposed method allows to obtain 1,3,4-trialkylcyclopentan-1-ol (1) based on the available starting reagents under mild conditions (20-21 ° C).
Способ поясняется следующими примерами:The method is illustrated by the following examples:
ПРИМЕР 1. В стеклянный реактор объемом 50 мл, установленный на магнитной мешалке, в атмосфере аргона помещают 10 мл ТГФ, 10 ммолей 1-гексена, 0.5 ммолей катализатора Cp2ZrCl2, 6 ммолей Mg (порошок), при температуре ~0° С 6 ммолей EtAlCl2, перемешивают 8 часов при комнатной температуре 20-21° С, затем при температуре -15° С добавляют 1 ммоль катализатора [NiСl2· 2Рh3Р] и 18 ммолей этилацетата, перемешивают 7 часов при температуре 20-21° С, реакционную массу гидролизуют водным раствором (5-7%) НСl. Из органического слоя выделяют 1-метил-3,4-ди(н-бутил)циклопентан-1-ол с выходом 64%.EXAMPLE 1. In a 50 ml glass reactor mounted on a magnetic stirrer, 10 ml of THF, 10 mmol of 1-hexene, 0.5 mmol of catalyst Cp 2 ZrCl 2 , 6 mmol of Mg (powder) at a temperature of ~ 0 ° C are placed in an argon atmosphere 6 mmol EtAlCl 2 , stirred for 8 hours at room temperature 20-21 ° C, then at a temperature of -15 ° C add 1 mmol of catalyst [NiCl 2 · 2Ph 3 P] and 18 mmol of ethyl acetate, stirred for 7 hours at a temperature of 20-21 ° C, the reaction mass is hydrolyzed with an aqueous solution (5-7%) of Hcl. 1-methyl-3,4-di (n-butyl) cyclopentan-1-ol is isolated from the organic layer in 64% yield.
Спектральные характеристики 1-метил-3,4-ди(н-бутил)циклопентан-1-ола(1):Spectral characteristics of 1-methyl-3,4-di (n-butyl) cyclopentan-1-ol (1):
ИК-спектр (ν , см-1): 3345, 2945, 2990, 2850, 1730, 1445, 1385, 1230, 1030, 930, 725. Спектр ЯМР 1Н (δ , м.д.): 0.84-0.96 (м, 9Н, СН3), 1.28 (м, 12Н, СН2), 2.02-2.54 (м, 6Н, СН, СН2 в цикле), 3.51-3.64 (м, СН-ОН). Спектр ЯМР 13С (δ , м.д.): 72.24 (С1), 42.52 (С2), 37.36 (С3), 26.62 (С4), 31.98 (С5), 22.44 (С6), 14.08 (С7), 26.29 (С8). Масс-спектр, m/z: 194[М-18]+. IR spectrum (ν, cm -1 ): 3345, 2945, 2990, 2850, 1730, 1445, 1385, 1230, 1030, 930, 725. 1 H NMR spectrum (δ, ppm): 0.84-0.96 ( m, 9H, CH 3 ), 1.28 (m, 12H, CH 2 ), 2.02-2.54 (m, 6H, CH, CH 2 in the cycle), 3.51-3.64 (m, CH-OH). 13 C NMR spectrum (δ, ppm): 72.24 (C 1 ), 42.52 (C 2 ), 37.36 (C 3 ), 26.62 (C 4 ), 31.98 (C 5 ), 22.44 (C 6 ), 14.08 (C 7 ), 26.29 (C 8 ). Mass spectrum, m / z: 194 [M-18] + .
Другие примеры, подтверждающие способ, приведены в табл.1.Other examples confirming the method are given in table 1.
Опыты проводили в ТГФ при комнатной температуре (20-21° С).The experiments were carried out in THF at room temperature (20-21 ° C).
Claims (1)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| RU2003114593/04A RU2250892C2 (en) | 2003-05-16 | 2003-05-16 | Method for production of 1,3,4-trialkylcyclopentane-1-ols |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| RU2003114593/04A RU2250892C2 (en) | 2003-05-16 | 2003-05-16 | Method for production of 1,3,4-trialkylcyclopentane-1-ols |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| RU2003114593A RU2003114593A (en) | 2004-11-27 |
| RU2250892C2 true RU2250892C2 (en) | 2005-04-27 |
Family
ID=35636208
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| RU2003114593/04A RU2250892C2 (en) | 2003-05-16 | 2003-05-16 | Method for production of 1,3,4-trialkylcyclopentane-1-ols |
Country Status (1)
| Country | Link |
|---|---|
| RU (1) | RU2250892C2 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| RU2433990C2 (en) * | 2009-12-07 | 2011-11-20 | Учреждение Российской Академии Наук Институт Нефтехимии И Катализа Ран | Method of producing 1-alkyl-2-alkyl(cycloalkenyl, benzyl)cyclopropanols |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0237978A3 (en) * | 1986-03-21 | 1988-01-20 | Henkel Kommanditgesellschaft auf Aktien | Isomeric 1-alkyl/alkenyl-2,2,4(2,4,4)-trimethyl-cyclopentan-1-ols as perfuming agents |
| US5345007A (en) * | 1992-11-06 | 1994-09-06 | Elf Atochem S.A. | Process for the preparation of octadienols |
| RU2198159C2 (en) * | 1996-11-26 | 2003-02-10 | Шелл Интернэшнл Рисерч Маатсхаппий Б.В. | Mixture of branched primary alcohols, methods of its synthesis, mixtures of biologically decomposable detergents, detergent composite |
-
2003
- 2003-05-16 RU RU2003114593/04A patent/RU2250892C2/en not_active IP Right Cessation
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0237978A3 (en) * | 1986-03-21 | 1988-01-20 | Henkel Kommanditgesellschaft auf Aktien | Isomeric 1-alkyl/alkenyl-2,2,4(2,4,4)-trimethyl-cyclopentan-1-ols as perfuming agents |
| US5345007A (en) * | 1992-11-06 | 1994-09-06 | Elf Atochem S.A. | Process for the preparation of octadienols |
| RU2198159C2 (en) * | 1996-11-26 | 2003-02-10 | Шелл Интернэшнл Рисерч Маатсхаппий Б.В. | Mixture of branched primary alcohols, methods of its synthesis, mixtures of biologically decomposable detergents, detergent composite |
Non-Patent Citations (1)
| Title |
|---|
| C.D. Nenitzescu et al. "The Synthesis of Cyclic Alcohols and Olefins by the Interaction of Dimagnesium Halides and Esters", J. Amer. Chem. Soc., 1950, №72, p.3483-3486. * |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| RU2433990C2 (en) * | 2009-12-07 | 2011-11-20 | Учреждение Российской Академии Наук Институт Нефтехимии И Катализа Ран | Method of producing 1-alkyl-2-alkyl(cycloalkenyl, benzyl)cyclopropanols |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| IL153578A (en) | Synthesis of functionalized and unfunctionalized olefins via cross and ring-closing metathesis | |
| Happ et al. | On the Reactivity of Acetylenes Coordinated to Cobalt. 9. Effects of Substitution and Coordination on the 13C-NMR Chemical Shifts of the sp Carbons of (. mu. 2-R1C2R2) Co2 (CO) 6 Complexes. Molecular Structure of (. mu. 2-PhC2SiPh3) Co2 (CO) 6 | |
| CA2932568A1 (en) | Metal-ligand cooperative catalysis through n-h arm deprotonation/pyridine dearomatiztion for efficient hydrogen generation from formic acid | |
| KR101676835B1 (en) | Ligand compound, catalyst system for olefin oligomerization, and method for olefin oligomerization using the same | |
| Govender et al. | Monometallic and mixed-valence bimetallic Rh (I/III) complexes: Synthesis, structure, electrochemistry and application in 1-octene hydroformylation | |
| RU2250892C2 (en) | Method for production of 1,3,4-trialkylcyclopentane-1-ols | |
| US4245131A (en) | Dimerization of olefins using tantalum and niobium catalysts | |
| US4231947A (en) | Tantalum and niobium catalysts or catalyst precursors | |
| RU2247705C2 (en) | Method of preparing trans-3,4-dialkylcyclopentan-1-ols | |
| RU2313531C1 (en) | Method for preparing 1-ethyl-3,4-di-(7-octenyl)-aluminacyclopentane | |
| RU2307825C2 (en) | METHOD FOR PRODUCTION OF 3-n-ALKYL-1-HYDROXYCYCLOPENTANE-n-ALKYLCARBOXYLATES | |
| SU1664797A1 (en) | Method for obtaining 3-ethyl-3 alumotricyclo-(5 | |
| Dervisi et al. | Synthesis and chemistry of diphenyl-2-pyridylphosphine complexes of palladium (0). X-Ray characterisation of Pd (Ph 2 Ppy) 2 (η 2-DMAD) and trans-Pd (Ph 2 Ppy) 2 (PhC [double bond, length half m-dash] CH 2)(CF 3 CO 2) | |
| RU2342393C2 (en) | METHOD OF OBTAINING 1-ETHYL-1,2,3,3a,4,5,6,7,8,9-DECAHYDROCYCLONONE[b]ALUMINACYCLOPENTANE | |
| WO2022219173A1 (en) | Production of panthenol | |
| CN107935878A (en) | A kind of method that primary amide is prepared by alkene, carbon monoxide and ammonia | |
| RU2238262C1 (en) | Method for preparing 2,3-fullereno[60]cyclopentanols | |
| US4996340A (en) | Intramolecular migration reactions | |
| RU2378245C2 (en) | Spiro[3,4]octan-6-ol synthesis method | |
| RU2355677C1 (en) | Method of 6-hydroxyspiro[3,4]octane-6-alkylcarboxylates obtainment | |
| RU2342392C2 (en) | Method of 6-ethyl-6-aluminaspiro[3,4] octane | |
| US4681708A (en) | Process for converting an alkoxyalkadiene to an alkyl dialkoxyalkanoate | |
| Braunstein et al. | Synthesis of nickel phenyl complexes with new chelating κ2-P, N ligands derived from α-iminoazatriphenylphosphoranes | |
| RU2376311C2 (en) | Method of combined production of tetracyclo [5.4.1.02,6.08,11]dodec-3-ene-9-spiro(3'-ethyl-3'-aluminacyclopentane) and tetracyclo [5.4.1.02,6.08,11]dodec-4-ene-9-spiro(3'-ethyl-3'-aluminacyclopentane) | |
| RU2191171C1 (en) | Method of synthesis of saturated branched hydrocarbons |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| MM4A | The patent is invalid due to non-payment of fees |
Effective date: 20050517 |