RU2221566C1 - Medicinal agent for opiomania treatment - Google Patents
Medicinal agent for opiomania treatment Download PDFInfo
- Publication number
- RU2221566C1 RU2221566C1 RU2003104582/15A RU2003104582A RU2221566C1 RU 2221566 C1 RU2221566 C1 RU 2221566C1 RU 2003104582/15 A RU2003104582/15 A RU 2003104582/15A RU 2003104582 A RU2003104582 A RU 2003104582A RU 2221566 C1 RU2221566 C1 RU 2221566C1
- Authority
- RU
- Russia
- Prior art keywords
- carboxylic acids
- esters
- carbon atoms
- acids
- substituted
- Prior art date
Links
Images
Landscapes
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
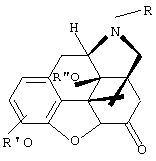
FIELD: medicine, narcology, pharmacy. SUBSTANCE: invention relates to agent used for treatment of opiomania. Medicinal agent consists of antagonists of opiate receptors and pharmaceutically acceptable carrier. As antagonists of opiate receptors esters of N-substituted 14-hydroxy- morphinans are used; as a carrier natural oils, esters of carboxylic acid with 8-16 carbon atoms and low-molecular alcohols are used, sulfolanes, propylene carbonate, N,N- dimethylacetamide or their mixture are used taken in the definite ratio of components. Invention provides the development of highly effective low toxic an antirelapse agent with prolonged opioprotective effect being after a single subcutaneous or intramuscular injection. EFFECT: enhanced effectiveness of agent, valuable medicinal properties. 4 cl, 3 tbl
Description
Текст описания в факсимильном виде (см. графический материал)а Description text in facsimile form (see graphic material) a
Claims (4)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| RU2003104582/15A RU2221566C1 (en) | 2003-02-17 | 2003-02-17 | Medicinal agent for opiomania treatment |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| RU2003104582/15A RU2221566C1 (en) | 2003-02-17 | 2003-02-17 | Medicinal agent for opiomania treatment |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| RU2221566C1 true RU2221566C1 (en) | 2004-01-20 |
Family
ID=32091909
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| RU2003104582/15A RU2221566C1 (en) | 2003-02-17 | 2003-02-17 | Medicinal agent for opiomania treatment |
Country Status (1)
| Country | Link |
|---|---|
| RU (1) | RU2221566C1 (en) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10525055B2 (en) | 2017-11-03 | 2020-01-07 | Nirsum Laboratories, Inc. | Opioid receptor antagonist prodrugs |
| US10533015B1 (en) | 2019-05-07 | 2020-01-14 | Nirsum Laboratories, Inc. | Opioid receptor antagonist prodrugs |
| JP2021514971A (en) * | 2018-02-23 | 2021-06-17 | ロデス テクノロジーズ | New opioid compounds and their use |
| US11186585B2 (en) | 2018-08-17 | 2021-11-30 | Kappa-Pharma LLC | Compositions and methods of enhancing opioid receptor engagement by opioid hexadienoates and optionally substituted hexadienoates |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4889860A (en) * | 1985-09-23 | 1989-12-26 | Nova Pharmaceutical Corporation | Oximes of oxymorphone, naltrexone and naloxone as potent, selective opioid receptor agonists and antagonists |
| WO1999032119A1 (en) * | 1997-12-22 | 1999-07-01 | Euro-Celtique, S.A. | Opioid agonist/antagonist combinations |
| RU2147879C1 (en) * | 1999-11-10 | 2000-04-27 | Мамренко Валерий Русланович | Drug for treatment of patients with opium narcomania and method of their treatment |
| RU2159615C1 (en) * | 1999-11-30 | 2000-11-27 | Смирнов Леонид Дмитриевич | Medical preparation for treating narcomania |
| RU2165270C1 (en) * | 2000-06-28 | 2001-04-20 | Вовк Сергей Мирославович | Method for treating narcotic addiction |
| RU2194538C2 (en) * | 2001-02-08 | 2002-12-20 | Красноярская государственная медицинская академия | Method grot for applying accelerated stress protection type opioid detoxication |
-
2003
- 2003-02-17 RU RU2003104582/15A patent/RU2221566C1/en not_active IP Right Cessation
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4889860A (en) * | 1985-09-23 | 1989-12-26 | Nova Pharmaceutical Corporation | Oximes of oxymorphone, naltrexone and naloxone as potent, selective opioid receptor agonists and antagonists |
| WO1999032119A1 (en) * | 1997-12-22 | 1999-07-01 | Euro-Celtique, S.A. | Opioid agonist/antagonist combinations |
| RU2147879C1 (en) * | 1999-11-10 | 2000-04-27 | Мамренко Валерий Русланович | Drug for treatment of patients with opium narcomania and method of their treatment |
| RU2159615C1 (en) * | 1999-11-30 | 2000-11-27 | Смирнов Леонид Дмитриевич | Medical preparation for treating narcomania |
| RU2165270C1 (en) * | 2000-06-28 | 2001-04-20 | Вовк Сергей Мирославович | Method for treating narcotic addiction |
| RU2194538C2 (en) * | 2001-02-08 | 2002-12-20 | Красноярская государственная медицинская академия | Method grot for applying accelerated stress protection type opioid detoxication |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10525055B2 (en) | 2017-11-03 | 2020-01-07 | Nirsum Laboratories, Inc. | Opioid receptor antagonist prodrugs |
| US10538531B2 (en) | 2017-11-03 | 2020-01-21 | Nirsum Laboratories, Inc. | Opioid receptor antagonist prodrugs |
| US10544154B2 (en) | 2017-11-03 | 2020-01-28 | Nirsum Laboratories, Inc. | Opioid receptor antagonist prodrugs |
| JP2021514971A (en) * | 2018-02-23 | 2021-06-17 | ロデス テクノロジーズ | New opioid compounds and their use |
| US11845759B2 (en) | 2018-02-23 | 2023-12-19 | Rhodes Technologies | Opioid compounds and uses thereof |
| US11186585B2 (en) | 2018-08-17 | 2021-11-30 | Kappa-Pharma LLC | Compositions and methods of enhancing opioid receptor engagement by opioid hexadienoates and optionally substituted hexadienoates |
| US11851439B2 (en) | 2018-08-17 | 2023-12-26 | Kappa-Pharma LLC | Compositions and methods of use for opioid hexadienoates and optionally substituted hexadienoates |
| US10533015B1 (en) | 2019-05-07 | 2020-01-14 | Nirsum Laboratories, Inc. | Opioid receptor antagonist prodrugs |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AR018634A1 (en) | BENZO DERIVATIVES (B) TIEPIN-1,1, -DIOXIDE, PROCEDURE FOR PREPARATION, MEDICATIONS CONTAINING THESE COMPOUNDS, PROCEDURE FOR PREPARATION OF MEDICINES AND THEIR EMPLOYMENT FOR PREPARATION OF MEDICINES. | |
| KR927003585A (en) | Heterocyclic compounds | |
| RU2009125597A (en) | MEDICINES AND METHODS FOR PRODUCING AND USING THEM | |
| RU2221566C1 (en) | Medicinal agent for opiomania treatment | |
| US10568890B2 (en) | Lactate-based fulvestrant or fulvestrant derivative oily preparation and preparation method thereof | |
| CO4950542A1 (en) | IMPROVED PROCESS FOR THE SYNTHESIS OF PROTECTED ESTERS OF ACID (S) -3, 4-DIHIDROXIBUTIRICO | |
| RU2007129090A (en) | ORGANIC COMPOUNDS | |
| ATE493133T1 (en) | ANTIBIOTIC/ANALGESIC FORMULATION AND METHOD FOR PRODUCING SUCH FORMULATION | |
| AR067475A1 (en) | NON BASIC S ANTAGONISTS OF THE RECEIVER OF THE CONCENTRATING HORMONE OF MELANINA 1 | |
| RU2008133761A (en) | PHARMACEUTICAL PREPARATION OF TAXANA, SOLID TAXAN COMPOSITION, METHOD FOR PRODUCING SOLID TAXAN COMPOSITION, COMPOSITION FOR INCREASING THE SOLUBILITY OF THE SPECIFIED SOLID TAXAN SOLID COMPOSITION AND NOREXTARA ELEMENT FOR ( | |
| AR072042A1 (en) | COMPOUNDS OF 2,3,4,5-TETRAHIDRO-1H-PIRIDO (4,3-B) INDOL AND METHODS OF THE SAME USE | |
| AR036939A1 (en) | ANTAGONISTS OF THE MELANINE CONCENTRATION HORMONE (MCH) PHARMACEUTICAL COMPOSITIONS, A PROCESS FOR THEIR DEVELOPMENT AND THE USE OF SUCH COMPOUNDS, ONLY OR IN COMBINATION, FOR THE DEVELOPMENT OF A MEDICINAL PRODUCT FOR THE TREATMENT OF OBESITY | |
| CN1093571A (en) | Parmaceutical agent | |
| PE20060562A1 (en) | AMINO-PIPERIDINE DERIVATIVES AS AGONISTS OF MC3 AND / OR MC4 MELANOCORTIN RECEPTORS | |
| RU2371444C1 (en) | FURO- AND THIENO[2,3-b]-QUINOLINE-2-CARBOXAMIDES, METHOD OF PRODUCTION AND ANTITUBERCULOUS ACTIVITY | |
| KR920702854A (en) | Octapeptide analogues of somatostatin with threonine at position 6 | |
| KR900016202A (en) | Spirocyclic compounds containing 5-membered rings having two heteroatoms | |
| RU2227145C2 (en) | Lhrh antagonists and method for their preparing, pharmaceutical composition, method for preparing medicinal agent | |
| KR880011157A (en) | Heterocyclic Compounds, Preparations and Uses thereof | |
| US20220048946A1 (en) | Cyclic tetrapeptide stereoisomers | |
| AR040126A1 (en) | PHENYL SULFONYL COMPOUND, PHARMACEUTICAL COMPOSITION THAT INCLUDES IT AND ITS USE FOR THE PREPARATION OF A MEDICINAL PRODUCT | |
| RU2215741C1 (en) | N-substituted 14-hydroxymorphinane esters and method for their preparing | |
| CA2561509A1 (en) | Anti-itching agent | |
| AR072802A1 (en) | PIPERIDINIC DERIVATIVES OF ESTERES OF BIFENIL-2-IL-CARBAMIC ACID, ANTAGONISTS OF M3 MUSCARINIC RECEPTORS, PHARMACEUTICAL COMPOSITIONS CONTAINING THEM AND USES OF THE SAME FOR THE TREATMENT OF DIGESTIVE OR URBAN DISORDERS. | |
| RU2008134492A (en) | PHARMACEUTICAL COMPOSITION CONTAINING A BICYCLIC COMPOUND AND METHOD FOR STABILIZING A Bicyclic Compound |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| MM4A | The patent is invalid due to non-payment of fees |
Effective date: 20170218 |