KR20200050893A - Compound and organic light emitting device comprising the same - Google Patents
Compound and organic light emitting device comprising the same Download PDFInfo
- Publication number
- KR20200050893A KR20200050893A KR1020190138726A KR20190138726A KR20200050893A KR 20200050893 A KR20200050893 A KR 20200050893A KR 1020190138726 A KR1020190138726 A KR 1020190138726A KR 20190138726 A KR20190138726 A KR 20190138726A KR 20200050893 A KR20200050893 A KR 20200050893A
- Authority
- KR
- South Korea
- Prior art keywords
- group
- layer
- light emitting
- compound
- formula
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 150000001875 compounds Chemical class 0.000 title claims abstract description 107
- 239000000126 substance Substances 0.000 claims abstract description 62
- 239000010410 layer Substances 0.000 claims description 226
- 238000002347 injection Methods 0.000 claims description 49
- 239000007924 injection Substances 0.000 claims description 49
- 239000011368 organic material Substances 0.000 claims description 49
- 125000004432 carbon atom Chemical group C* 0.000 claims description 41
- 125000003118 aryl group Chemical group 0.000 claims description 37
- 230000005525 hole transport Effects 0.000 claims description 26
- 125000001072 heteroaryl group Chemical group 0.000 claims description 22
- 230000000903 blocking effect Effects 0.000 claims description 20
- 238000000034 method Methods 0.000 claims description 20
- YZCKVEUIGOORGS-OUBTZVSYSA-N Deuterium Chemical compound [2H] YZCKVEUIGOORGS-OUBTZVSYSA-N 0.000 claims description 15
- 229910052805 deuterium Inorganic materials 0.000 claims description 15
- 125000000732 arylene group Chemical group 0.000 claims description 10
- 125000000217 alkyl group Chemical group 0.000 claims description 8
- 125000005549 heteroarylene group Chemical group 0.000 claims description 7
- 229910052739 hydrogen Inorganic materials 0.000 claims description 6
- 239000001257 hydrogen Substances 0.000 claims description 6
- 125000002560 nitrile group Chemical group 0.000 claims description 5
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 4
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 4
- 239000012044 organic layer Substances 0.000 claims description 4
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 72
- 239000002904 solvent Substances 0.000 description 54
- -1 1-methylpentyl Chemical group 0.000 description 38
- 239000000463 material Substances 0.000 description 38
- 230000000052 comparative effect Effects 0.000 description 37
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 35
- 238000006243 chemical reaction Methods 0.000 description 33
- 238000004440 column chromatography Methods 0.000 description 28
- 230000015572 biosynthetic process Effects 0.000 description 27
- 239000000203 mixture Substances 0.000 description 27
- 238000010992 reflux Methods 0.000 description 25
- 125000001424 substituent group Chemical group 0.000 description 25
- 238000003786 synthesis reaction Methods 0.000 description 25
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 25
- 230000032258 transport Effects 0.000 description 25
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 24
- 239000013078 crystal Substances 0.000 description 24
- 239000008096 xylene Substances 0.000 description 24
- SFKMVPQJJGJCMI-UHFFFAOYSA-N 2-chloro-4-phenylquinazoline Chemical compound C=12C=CC=CC2=NC(Cl)=NC=1C1=CC=CC=C1 SFKMVPQJJGJCMI-UHFFFAOYSA-N 0.000 description 14
- 239000002019 doping agent Substances 0.000 description 14
- 239000000758 substrate Substances 0.000 description 12
- MFRIHAYPQRLWNB-UHFFFAOYSA-N sodium tert-butoxide Chemical compound [Na+].CC(C)(C)[O-] MFRIHAYPQRLWNB-UHFFFAOYSA-N 0.000 description 11
- YTTFZSLFNVFHAQ-UHFFFAOYSA-N 2-chloro-4-(4-naphthalen-1-ylphenyl)quinazoline Chemical compound C1=CC=CC2=NC(Cl)=NC(C=3C=CC(=CC=3)C=3C4=CC=CC=C4C=CC=3)=C21 YTTFZSLFNVFHAQ-UHFFFAOYSA-N 0.000 description 10
- 238000002360 preparation method Methods 0.000 description 10
- 229910052782 aluminium Inorganic materials 0.000 description 8
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 8
- 229910052751 metal Inorganic materials 0.000 description 8
- 239000002184 metal Substances 0.000 description 8
- 238000000151 deposition Methods 0.000 description 7
- 229940126062 Compound A Drugs 0.000 description 6
- NLDMNSXOCDLTTB-UHFFFAOYSA-N Heterophylliin A Natural products O1C2COC(=O)C3=CC(O)=C(O)C(O)=C3C3=C(O)C(O)=C(O)C=C3C(=O)OC2C(OC(=O)C=2C=C(O)C(O)=C(O)C=2)C(O)C1OC(=O)C1=CC(O)=C(O)C(O)=C1 NLDMNSXOCDLTTB-UHFFFAOYSA-N 0.000 description 6
- JNGZXGGOCLZBFB-IVCQMTBJSA-N compound E Chemical compound N([C@@H](C)C(=O)N[C@@H]1C(N(C)C2=CC=CC=C2C(C=2C=CC=CC=2)=N1)=O)C(=O)CC1=CC(F)=CC(F)=C1 JNGZXGGOCLZBFB-IVCQMTBJSA-N 0.000 description 6
- 125000002950 monocyclic group Chemical group 0.000 description 6
- YJTKZCDBKVTVBY-UHFFFAOYSA-N 1,3-Diphenylbenzene Chemical group C1=CC=CC=C1C1=CC=CC(C=2C=CC=CC=2)=C1 YJTKZCDBKVTVBY-UHFFFAOYSA-N 0.000 description 5
- 125000001931 aliphatic group Chemical group 0.000 description 5
- 150000004982 aromatic amines Chemical class 0.000 description 5
- 125000000609 carbazolyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3NC12)* 0.000 description 5
- 229910052799 carbon Inorganic materials 0.000 description 5
- TXCDCPKCNAJMEE-UHFFFAOYSA-N dibenzofuran Chemical group C1=CC=C2C3=CC=CC=C3OC2=C1 TXCDCPKCNAJMEE-UHFFFAOYSA-N 0.000 description 5
- IYYZUPMFVPLQIF-ALWQSETLSA-N dibenzothiophene Chemical group C1=CC=CC=2[34S]C3=C(C=21)C=CC=C3 IYYZUPMFVPLQIF-ALWQSETLSA-N 0.000 description 5
- 239000012153 distilled water Substances 0.000 description 5
- PQXKHYXIUOZZFA-UHFFFAOYSA-M lithium fluoride Chemical compound [Li+].[F-] PQXKHYXIUOZZFA-UHFFFAOYSA-M 0.000 description 5
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 5
- JYEUMXHLPRZUAT-UHFFFAOYSA-N 1,2,3-triazine Chemical group C1=CN=NN=C1 JYEUMXHLPRZUAT-UHFFFAOYSA-N 0.000 description 4
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 4
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 description 4
- 125000005264 aryl amine group Chemical group 0.000 description 4
- 125000004429 atom Chemical group 0.000 description 4
- 125000006267 biphenyl group Chemical group 0.000 description 4
- 239000010406 cathode material Substances 0.000 description 4
- 125000000753 cycloalkyl group Chemical group 0.000 description 4
- 230000000694 effects Effects 0.000 description 4
- 125000003983 fluorenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3CC12)* 0.000 description 4
- 125000005842 heteroatom Chemical group 0.000 description 4
- 238000004519 manufacturing process Methods 0.000 description 4
- 125000001624 naphthyl group Chemical group 0.000 description 4
- 229910052760 oxygen Inorganic materials 0.000 description 4
- 125000003367 polycyclic group Chemical group 0.000 description 4
- LVTJOONKWUXEFR-FZRMHRINSA-N protoneodioscin Natural products O(C[C@@H](CC[C@]1(O)[C@H](C)[C@@H]2[C@]3(C)[C@H]([C@H]4[C@@H]([C@]5(C)C(=CC4)C[C@@H](O[C@@H]4[C@H](O[C@H]6[C@@H](O)[C@@H](O)[C@@H](O)[C@H](C)O6)[C@@H](O)[C@H](O[C@H]6[C@@H](O)[C@@H](O)[C@@H](O)[C@H](C)O6)[C@H](CO)O4)CC5)CC3)C[C@@H]2O1)C)[C@H]1[C@H](O)[C@H](O)[C@H](O)[C@@H](CO)O1 LVTJOONKWUXEFR-FZRMHRINSA-N 0.000 description 4
- HRNRMRBZDUOXPS-UHFFFAOYSA-N (2-nitronaphthalen-1-yl) trifluoromethanesulfonate Chemical compound C1=CC=CC2=C(OS(=O)(=O)C(F)(F)F)C([N+](=O)[O-])=CC=C21 HRNRMRBZDUOXPS-UHFFFAOYSA-N 0.000 description 3
- ZTSYGXLLTYTXCS-UHFFFAOYSA-N 1-chloronaphthalen-2-amine Chemical compound C1=CC=CC2=C(Cl)C(N)=CC=C21 ZTSYGXLLTYTXCS-UHFFFAOYSA-N 0.000 description 3
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 3
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 3
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical group C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 3
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 3
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Thiophene Chemical group C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 description 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- 229910045601 alloy Inorganic materials 0.000 description 3
- 239000000956 alloy Substances 0.000 description 3
- IOJUPLGTWVMSFF-UHFFFAOYSA-N benzothiazole Chemical group C1=CC=C2SC=NC2=C1 IOJUPLGTWVMSFF-UHFFFAOYSA-N 0.000 description 3
- 229920001940 conductive polymer Polymers 0.000 description 3
- RAXXELZNTBOGNW-UHFFFAOYSA-N imidazole Natural products C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 description 3
- 125000004957 naphthylene group Chemical group 0.000 description 3
- 125000001792 phenanthrenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3C=CC12)* 0.000 description 3
- 125000000843 phenylene group Chemical group C1(=C(C=CC=C1)*)* 0.000 description 3
- 125000000714 pyrimidinyl group Chemical group 0.000 description 3
- 229910052709 silver Inorganic materials 0.000 description 3
- 239000004332 silver Substances 0.000 description 3
- 239000000243 solution Substances 0.000 description 3
- 239000010409 thin film Substances 0.000 description 3
- TVIVIEFSHFOWTE-UHFFFAOYSA-K tri(quinolin-8-yloxy)alumane Chemical compound [Al+3].C1=CN=C2C([O-])=CC=CC2=C1.C1=CN=C2C([O-])=CC=CC2=C1.C1=CN=C2C([O-])=CC=CC2=C1 TVIVIEFSHFOWTE-UHFFFAOYSA-K 0.000 description 3
- AOSZTAHDEDLTLQ-AZKQZHLXSA-N (1S,2S,4R,8S,9S,11S,12R,13S,19S)-6-[(3-chlorophenyl)methyl]-12,19-difluoro-11-hydroxy-8-(2-hydroxyacetyl)-9,13-dimethyl-6-azapentacyclo[10.8.0.02,9.04,8.013,18]icosa-14,17-dien-16-one Chemical compound C([C@@H]1C[C@H]2[C@H]3[C@]([C@]4(C=CC(=O)C=C4[C@@H](F)C3)C)(F)[C@@H](O)C[C@@]2([C@@]1(C1)C(=O)CO)C)N1CC1=CC=CC(Cl)=C1 AOSZTAHDEDLTLQ-AZKQZHLXSA-N 0.000 description 2
- SZUVGFMDDVSKSI-WIFOCOSTSA-N (1s,2s,3s,5r)-1-(carboxymethyl)-3,5-bis[(4-phenoxyphenyl)methyl-propylcarbamoyl]cyclopentane-1,2-dicarboxylic acid Chemical compound O=C([C@@H]1[C@@H]([C@](CC(O)=O)([C@H](C(=O)N(CCC)CC=2C=CC(OC=3C=CC=CC=3)=CC=2)C1)C(O)=O)C(O)=O)N(CCC)CC(C=C1)=CC=C1OC1=CC=CC=C1 SZUVGFMDDVSKSI-WIFOCOSTSA-N 0.000 description 2
- GHYOCDFICYLMRF-UTIIJYGPSA-N (2S,3R)-N-[(2S)-3-(cyclopenten-1-yl)-1-[(2R)-2-methyloxiran-2-yl]-1-oxopropan-2-yl]-3-hydroxy-3-(4-methoxyphenyl)-2-[[(2S)-2-[(2-morpholin-4-ylacetyl)amino]propanoyl]amino]propanamide Chemical compound C1(=CCCC1)C[C@@H](C(=O)[C@@]1(OC1)C)NC([C@H]([C@@H](C1=CC=C(C=C1)OC)O)NC([C@H](C)NC(CN1CCOCC1)=O)=O)=O GHYOCDFICYLMRF-UTIIJYGPSA-N 0.000 description 2
- ITOFPJRDSCGOSA-KZLRUDJFSA-N (2s)-2-[[(4r)-4-[(3r,5r,8r,9s,10s,13r,14s,17r)-3-hydroxy-10,13-dimethyl-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1h-cyclopenta[a]phenanthren-17-yl]pentanoyl]amino]-3-(1h-indol-3-yl)propanoic acid Chemical compound C([C@H]1CC2)[C@H](O)CC[C@]1(C)[C@@H](CC[C@]13C)[C@@H]2[C@@H]3CC[C@@H]1[C@H](C)CCC(=O)N[C@H](C(O)=O)CC1=CNC2=CC=CC=C12 ITOFPJRDSCGOSA-KZLRUDJFSA-N 0.000 description 2
- WWTBZEKOSBFBEM-SPWPXUSOSA-N (2s)-2-[[2-benzyl-3-[hydroxy-[(1r)-2-phenyl-1-(phenylmethoxycarbonylamino)ethyl]phosphoryl]propanoyl]amino]-3-(1h-indol-3-yl)propanoic acid Chemical compound N([C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)O)C(=O)C(CP(O)(=O)[C@H](CC=1C=CC=CC=1)NC(=O)OCC=1C=CC=CC=1)CC1=CC=CC=C1 WWTBZEKOSBFBEM-SPWPXUSOSA-N 0.000 description 2
- QFLWZFQWSBQYPS-AWRAUJHKSA-N (3S)-3-[[(2S)-2-[[(2S)-2-[5-[(3aS,6aR)-2-oxo-1,3,3a,4,6,6a-hexahydrothieno[3,4-d]imidazol-4-yl]pentanoylamino]-3-methylbutanoyl]amino]-3-(4-hydroxyphenyl)propanoyl]amino]-4-[1-bis(4-chlorophenoxy)phosphorylbutylamino]-4-oxobutanoic acid Chemical compound CCCC(NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@@H](NC(=O)CCCCC1SC[C@@H]2NC(=O)N[C@H]12)C(C)C)P(=O)(Oc1ccc(Cl)cc1)Oc1ccc(Cl)cc1 QFLWZFQWSBQYPS-AWRAUJHKSA-N 0.000 description 2
- IWZSHWBGHQBIML-ZGGLMWTQSA-N (3S,8S,10R,13S,14S,17S)-17-isoquinolin-7-yl-N,N,10,13-tetramethyl-2,3,4,7,8,9,11,12,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-3-amine Chemical compound CN(C)[C@H]1CC[C@]2(C)C3CC[C@@]4(C)[C@@H](CC[C@@H]4c4ccc5ccncc5c4)[C@@H]3CC=C2C1 IWZSHWBGHQBIML-ZGGLMWTQSA-N 0.000 description 2
- ONBQEOIKXPHGMB-VBSBHUPXSA-N 1-[2-[(2s,3r,4s,5r)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]oxy-4,6-dihydroxyphenyl]-3-(4-hydroxyphenyl)propan-1-one Chemical compound O[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1OC1=CC(O)=CC(O)=C1C(=O)CCC1=CC=C(O)C=C1 ONBQEOIKXPHGMB-VBSBHUPXSA-N 0.000 description 2
- UNILWMWFPHPYOR-KXEYIPSPSA-M 1-[6-[2-[3-[3-[3-[2-[2-[3-[[2-[2-[[(2r)-1-[[2-[[(2r)-1-[3-[2-[2-[3-[[2-(2-amino-2-oxoethoxy)acetyl]amino]propoxy]ethoxy]ethoxy]propylamino]-3-hydroxy-1-oxopropan-2-yl]amino]-2-oxoethyl]amino]-3-[(2r)-2,3-di(hexadecanoyloxy)propyl]sulfanyl-1-oxopropan-2-yl Chemical compound O=C1C(SCCC(=O)NCCCOCCOCCOCCCNC(=O)COCC(=O)N[C@@H](CSC[C@@H](COC(=O)CCCCCCCCCCCCCCC)OC(=O)CCCCCCCCCCCCCCC)C(=O)NCC(=O)N[C@H](CO)C(=O)NCCCOCCOCCOCCCNC(=O)COCC(N)=O)CC(=O)N1CCNC(=O)CCCCCN\1C2=CC=C(S([O-])(=O)=O)C=C2CC/1=C/C=C/C=C/C1=[N+](CC)C2=CC=C(S([O-])(=O)=O)C=C2C1 UNILWMWFPHPYOR-KXEYIPSPSA-M 0.000 description 2
- QENGPZGAWFQWCZ-UHFFFAOYSA-N 3-Methylthiophene Chemical compound CC=1C=CSC=1 QENGPZGAWFQWCZ-UHFFFAOYSA-N 0.000 description 2
- VIJYEGDOKCKUOL-UHFFFAOYSA-N 9-phenylcarbazole Chemical group C1=CC=CC=C1N1C2=CC=CC=C2C2=CC=CC=C21 VIJYEGDOKCKUOL-UHFFFAOYSA-N 0.000 description 2
- UJOBWOGCFQCDNV-UHFFFAOYSA-N 9H-carbazole Chemical compound C1=CC=C2C3=CC=CC=C3NC2=C1 UJOBWOGCFQCDNV-UHFFFAOYSA-N 0.000 description 2
- 238000006443 Buchwald-Hartwig cross coupling reaction Methods 0.000 description 2
- OJRUSAPKCPIVBY-KQYNXXCUSA-N C1=NC2=C(N=C(N=C2N1[C@H]3[C@@H]([C@@H]([C@H](O3)COP(=O)(CP(=O)(O)O)O)O)O)I)N Chemical compound C1=NC2=C(N=C(N=C2N1[C@H]3[C@@H]([C@@H]([C@H](O3)COP(=O)(CP(=O)(O)O)O)O)O)I)N OJRUSAPKCPIVBY-KQYNXXCUSA-N 0.000 description 2
- KCBAMQOKOLXLOX-BSZYMOERSA-N CC1=C(SC=N1)C2=CC=C(C=C2)[C@H](C)NC(=O)[C@@H]3C[C@H](CN3C(=O)[C@H](C(C)(C)C)NC(=O)CCCCCCCCCCNCCCONC(=O)C4=C(C(=C(C=C4)F)F)NC5=C(C=C(C=C5)I)F)O Chemical compound CC1=C(SC=N1)C2=CC=C(C=C2)[C@H](C)NC(=O)[C@@H]3C[C@H](CN3C(=O)[C@H](C(C)(C)C)NC(=O)CCCCCCCCCCNCCCONC(=O)C4=C(C(=C(C=C4)F)F)NC5=C(C=C(C=C5)I)F)O KCBAMQOKOLXLOX-BSZYMOERSA-N 0.000 description 2
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 2
- 229940126657 Compound 17 Drugs 0.000 description 2
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 2
- GYHNNYVSQQEPJS-UHFFFAOYSA-N Gallium Chemical compound [Ga] GYHNNYVSQQEPJS-UHFFFAOYSA-N 0.000 description 2
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 2
- OPFJDXRVMFKJJO-ZHHKINOHSA-N N-{[3-(2-benzamido-4-methyl-1,3-thiazol-5-yl)-pyrazol-5-yl]carbonyl}-G-dR-G-dD-dD-dD-NH2 Chemical compound S1C(C=2NN=C(C=2)C(=O)NCC(=O)N[C@H](CCCN=C(N)N)C(=O)NCC(=O)N[C@H](CC(O)=O)C(=O)N[C@H](CC(O)=O)C(=O)N[C@H](CC(O)=O)C(N)=O)=C(C)N=C1NC(=O)C1=CC=CC=C1 OPFJDXRVMFKJJO-ZHHKINOHSA-N 0.000 description 2
- KYQCOXFCLRTKLS-UHFFFAOYSA-N Pyrazine Chemical compound C1=CN=CC=N1 KYQCOXFCLRTKLS-UHFFFAOYSA-N 0.000 description 2
- CZPWVGJYEJSRLH-UHFFFAOYSA-N Pyrimidine Chemical compound C1=CN=CN=C1 CZPWVGJYEJSRLH-UHFFFAOYSA-N 0.000 description 2
- 238000006069 Suzuki reaction reaction Methods 0.000 description 2
- 239000007983 Tris buffer Substances 0.000 description 2
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 2
- LNUFLCYMSVYYNW-ZPJMAFJPSA-N [(2r,3r,4s,5r,6r)-2-[(2r,3r,4s,5r,6r)-6-[(2r,3r,4s,5r,6r)-6-[(2r,3r,4s,5r,6r)-6-[[(3s,5s,8r,9s,10s,13r,14s,17r)-10,13-dimethyl-17-[(2r)-6-methylheptan-2-yl]-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1h-cyclopenta[a]phenanthren-3-yl]oxy]-4,5-disulfo Chemical compound O([C@@H]1[C@@H](COS(O)(=O)=O)O[C@@H]([C@@H]([C@H]1OS(O)(=O)=O)OS(O)(=O)=O)O[C@@H]1[C@@H](COS(O)(=O)=O)O[C@@H]([C@@H]([C@H]1OS(O)(=O)=O)OS(O)(=O)=O)O[C@@H]1[C@@H](COS(O)(=O)=O)O[C@H]([C@@H]([C@H]1OS(O)(=O)=O)OS(O)(=O)=O)O[C@@H]1C[C@@H]2CC[C@H]3[C@@H]4CC[C@@H]([C@]4(CC[C@@H]3[C@@]2(C)CC1)C)[C@H](C)CCCC(C)C)[C@H]1O[C@H](COS(O)(=O)=O)[C@@H](OS(O)(=O)=O)[C@H](OS(O)(=O)=O)[C@H]1OS(O)(=O)=O LNUFLCYMSVYYNW-ZPJMAFJPSA-N 0.000 description 2
- MWPLVEDNUUSJAV-UHFFFAOYSA-N anthracene Chemical compound C1=CC=CC2=CC3=CC=CC=C3C=C21 MWPLVEDNUUSJAV-UHFFFAOYSA-N 0.000 description 2
- XRWSZZJLZRKHHD-WVWIJVSJSA-N asunaprevir Chemical compound O=C([C@@H]1C[C@H](CN1C(=O)[C@@H](NC(=O)OC(C)(C)C)C(C)(C)C)OC1=NC=C(C2=CC=C(Cl)C=C21)OC)N[C@]1(C(=O)NS(=O)(=O)C2CC2)C[C@H]1C=C XRWSZZJLZRKHHD-WVWIJVSJSA-N 0.000 description 2
- 125000002529 biphenylenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3C12)* 0.000 description 2
- 229910052791 calcium Inorganic materials 0.000 description 2
- 239000011575 calcium Substances 0.000 description 2
- 150000001721 carbon Chemical group 0.000 description 2
- WDECIBYCCFPHNR-UHFFFAOYSA-N chrysene Chemical compound C1=CC=CC2=CC=C3C4=CC=CC=C4C=CC3=C21 WDECIBYCCFPHNR-UHFFFAOYSA-N 0.000 description 2
- 238000000576 coating method Methods 0.000 description 2
- 229940125904 compound 1 Drugs 0.000 description 2
- 229940125773 compound 10 Drugs 0.000 description 2
- 229940125797 compound 12 Drugs 0.000 description 2
- 229940126543 compound 14 Drugs 0.000 description 2
- 229940125758 compound 15 Drugs 0.000 description 2
- 229940126142 compound 16 Drugs 0.000 description 2
- 229940125782 compound 2 Drugs 0.000 description 2
- 229940125810 compound 20 Drugs 0.000 description 2
- 229940126086 compound 21 Drugs 0.000 description 2
- 229940126208 compound 22 Drugs 0.000 description 2
- 229940125833 compound 23 Drugs 0.000 description 2
- 229940125961 compound 24 Drugs 0.000 description 2
- 229940126214 compound 3 Drugs 0.000 description 2
- 229940125898 compound 5 Drugs 0.000 description 2
- 150000004696 coordination complex Chemical class 0.000 description 2
- 238000005859 coupling reaction Methods 0.000 description 2
- 230000008021 deposition Effects 0.000 description 2
- 239000003599 detergent Substances 0.000 description 2
- 150000002219 fluoranthenes Chemical class 0.000 description 2
- 229910052733 gallium Inorganic materials 0.000 description 2
- 125000005843 halogen group Chemical group 0.000 description 2
- 150000002391 heterocyclic compounds Chemical class 0.000 description 2
- 125000000623 heterocyclic group Chemical group 0.000 description 2
- 150000002430 hydrocarbons Chemical group 0.000 description 2
- AMGQUBHHOARCQH-UHFFFAOYSA-N indium;oxotin Chemical compound [In].[Sn]=O AMGQUBHHOARCQH-UHFFFAOYSA-N 0.000 description 2
- 229910052741 iridium Inorganic materials 0.000 description 2
- GKOZUEZYRPOHIO-UHFFFAOYSA-N iridium atom Chemical compound [Ir] GKOZUEZYRPOHIO-UHFFFAOYSA-N 0.000 description 2
- 125000004491 isohexyl group Chemical group C(CCC(C)C)* 0.000 description 2
- ZLVXBBHTMQJRSX-VMGNSXQWSA-N jdtic Chemical compound C1([C@]2(C)CCN(C[C@@H]2C)C[C@H](C(C)C)NC(=O)[C@@H]2NCC3=CC(O)=CC=C3C2)=CC=CC(O)=C1 ZLVXBBHTMQJRSX-VMGNSXQWSA-N 0.000 description 2
- 229910052744 lithium Inorganic materials 0.000 description 2
- IMKMFBIYHXBKRX-UHFFFAOYSA-M lithium;quinoline-2-carboxylate Chemical compound [Li+].C1=CC=CC2=NC(C(=O)[O-])=CC=C21 IMKMFBIYHXBKRX-UHFFFAOYSA-M 0.000 description 2
- 229910044991 metal oxide Inorganic materials 0.000 description 2
- 150000004706 metal oxides Chemical class 0.000 description 2
- 150000002739 metals Chemical class 0.000 description 2
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 2
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 2
- 239000007773 negative electrode material Substances 0.000 description 2
- WCPAKWJPBJAGKN-UHFFFAOYSA-N oxadiazole Chemical group C1=CON=N1 WCPAKWJPBJAGKN-UHFFFAOYSA-N 0.000 description 2
- 125000002080 perylenyl group Chemical group C1(=CC=C2C=CC=C3C4=CC=CC5=CC=CC(C1=C23)=C45)* 0.000 description 2
- LFSXCDWNBUNEEM-UHFFFAOYSA-N phthalazine Chemical group C1=NN=CC2=CC=CC=C21 LFSXCDWNBUNEEM-UHFFFAOYSA-N 0.000 description 2
- 238000005240 physical vapour deposition Methods 0.000 description 2
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 2
- 229920000553 poly(phenylenevinylene) Polymers 0.000 description 2
- 229920000767 polyaniline Polymers 0.000 description 2
- 239000007774 positive electrode material Substances 0.000 description 2
- BBEAQIROQSPTKN-UHFFFAOYSA-N pyrene Chemical compound C1=CC=C2C=CC3=CC=CC4=CC=C1C2=C43 BBEAQIROQSPTKN-UHFFFAOYSA-N 0.000 description 2
- BWESROVQGZSBRX-UHFFFAOYSA-N pyrido[3,2-d]pyrimidine Chemical group C1=NC=NC2=CC=CN=C21 BWESROVQGZSBRX-UHFFFAOYSA-N 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 229910052717 sulfur Inorganic materials 0.000 description 2
- 125000001973 tert-pentyl group Chemical group [H]C([H])([H])C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- 150000003852 triazoles Chemical group 0.000 description 2
- 238000004506 ultrasonic cleaning Methods 0.000 description 2
- 238000005406 washing Methods 0.000 description 2
- 229910052725 zinc Inorganic materials 0.000 description 2
- 239000011701 zinc Substances 0.000 description 2
- 239000011787 zinc oxide Substances 0.000 description 2
- XXHOARFXPOMACV-UHFFFAOYSA-N (3-nitronaphthalen-2-yl) trifluoromethanesulfonate Chemical compound FC(S(=O)(=O)OC1=CC2=CC=CC=C2C=C1[N+](=O)[O-])(F)F XXHOARFXPOMACV-UHFFFAOYSA-N 0.000 description 1
- UWRZIZXBOLBCON-VOTSOKGWSA-N (e)-2-phenylethenamine Chemical compound N\C=C\C1=CC=CC=C1 UWRZIZXBOLBCON-VOTSOKGWSA-N 0.000 description 1
- BCMCBBGGLRIHSE-UHFFFAOYSA-N 1,3-benzoxazole Chemical compound C1=CC=C2OC=NC2=C1 BCMCBBGGLRIHSE-UHFFFAOYSA-N 0.000 description 1
- 125000000355 1,3-benzoxazolyl group Chemical group O1C(=NC2=C1C=CC=C2)* 0.000 description 1
- IANQTJSKSUMEQM-UHFFFAOYSA-N 1-benzofuran Chemical group C1=CC=C2OC=CC2=C1 IANQTJSKSUMEQM-UHFFFAOYSA-N 0.000 description 1
- FCEHBMOGCRZNNI-UHFFFAOYSA-N 1-benzothiophene Chemical group C1=CC=C2SC=CC2=C1 FCEHBMOGCRZNNI-UHFFFAOYSA-N 0.000 description 1
- XIWWHUAOOZIREN-UHFFFAOYSA-N 1-bromotriphenylene Chemical group C1=CC=CC2=C3C(Br)=CC=CC3=C(C=CC=C3)C3=C21 XIWWHUAOOZIREN-UHFFFAOYSA-N 0.000 description 1
- 125000006218 1-ethylbutyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])[H] 0.000 description 1
- MYKQKWIPLZEVOW-UHFFFAOYSA-N 11h-benzo[a]carbazole Chemical group C1=CC2=CC=CC=C2C2=C1C1=CC=CC=C1N2 MYKQKWIPLZEVOW-UHFFFAOYSA-N 0.000 description 1
- HIYWOHBEPVGIQN-UHFFFAOYSA-N 1h-benzo[g]indole Chemical group C1=CC=CC2=C(NC=C3)C3=CC=C21 HIYWOHBEPVGIQN-UHFFFAOYSA-N 0.000 description 1
- UWIWGZCNYRBVJL-UHFFFAOYSA-N 2-chloronaphthalen-1-amine Chemical compound C1=CC=C2C(N)=C(Cl)C=CC2=C1 UWIWGZCNYRBVJL-UHFFFAOYSA-N 0.000 description 1
- 125000006176 2-ethylbutyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(C([H])([H])*)C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000005916 2-methylpentyl group Chemical group 0.000 description 1
- DMEVMYSQZPJFOK-UHFFFAOYSA-N 3,4,5,6,9,10-hexazatetracyclo[12.4.0.02,7.08,13]octadeca-1(18),2(7),3,5,8(13),9,11,14,16-nonaene Chemical group N1=NN=C2C3=CC=CC=C3C3=CC=NN=C3C2=N1 DMEVMYSQZPJFOK-UHFFFAOYSA-N 0.000 description 1
- ISWSFPQWSDIVGH-UHFFFAOYSA-N 3-chloronaphthalen-2-amine Chemical compound C1=CC=C2C=C(Cl)C(N)=CC2=C1 ISWSFPQWSDIVGH-UHFFFAOYSA-N 0.000 description 1
- DDTHMESPCBONDT-UHFFFAOYSA-N 4-(4-oxocyclohexa-2,5-dien-1-ylidene)cyclohexa-2,5-dien-1-one Chemical compound C1=CC(=O)C=CC1=C1C=CC(=O)C=C1 DDTHMESPCBONDT-UHFFFAOYSA-N 0.000 description 1
- NJBMMMJOXRZENQ-UHFFFAOYSA-N 6H-pyrrolo[2,3-f]quinoline Chemical compound c1cc2ccc3[nH]cccc3c2n1 NJBMMMJOXRZENQ-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- 239000005725 8-Hydroxyquinoline Substances 0.000 description 1
- ROFVEXUMMXZLPA-UHFFFAOYSA-N Bipyridyl Chemical group N1=CC=CC=C1C1=CC=CC=N1 ROFVEXUMMXZLPA-UHFFFAOYSA-N 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- YLQBMQCUIZJEEH-UHFFFAOYSA-N Furan Chemical group C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 1
- 229910052688 Gadolinium Inorganic materials 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- MVXGYSWVPDGOIQ-UHFFFAOYSA-N N1=CN=CC=2C1=CC1=CC=CCC1=2 Chemical compound N1=CN=CC=2C1=CC1=CC=CCC1=2 MVXGYSWVPDGOIQ-UHFFFAOYSA-N 0.000 description 1
- ZCQWOFVYLHDMMC-UHFFFAOYSA-N Oxazole Chemical compound C1=COC=N1 ZCQWOFVYLHDMMC-UHFFFAOYSA-N 0.000 description 1
- YXLXNENXOJSQEI-UHFFFAOYSA-L Oxine-copper Chemical compound [Cu+2].C1=CN=C2C([O-])=CC=CC2=C1.C1=CN=C2C([O-])=CC=CC2=C1 YXLXNENXOJSQEI-UHFFFAOYSA-L 0.000 description 1
- PCNDJXKNXGMECE-UHFFFAOYSA-N Phenazine Natural products C1=CC=CC2=NC3=CC=CC=C3N=C21 PCNDJXKNXGMECE-UHFFFAOYSA-N 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- KAESVJOAVNADME-UHFFFAOYSA-N Pyrrole Chemical group C=1C=CNC=1 KAESVJOAVNADME-UHFFFAOYSA-N 0.000 description 1
- NRCMAYZCPIVABH-UHFFFAOYSA-N Quinacridone Chemical compound N1C2=CC=CC=C2C(=O)C2=C1C=C1C(=O)C3=CC=CC=C3NC1=C2 NRCMAYZCPIVABH-UHFFFAOYSA-N 0.000 description 1
- 229910052772 Samarium Inorganic materials 0.000 description 1
- 229910006404 SnO 2 Inorganic materials 0.000 description 1
- FZWLAAWBMGSTSO-UHFFFAOYSA-N Thiazole Chemical group C1=CSC=N1 FZWLAAWBMGSTSO-UHFFFAOYSA-N 0.000 description 1
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 1
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 1
- 229910052769 Ytterbium Inorganic materials 0.000 description 1
- DGEZNRSVGBDHLK-UHFFFAOYSA-N [1,10]phenanthroline Chemical group C1=CN=C2C3=NC=CC=C3C=CC2=C1 DGEZNRSVGBDHLK-UHFFFAOYSA-N 0.000 description 1
- AZDRQVAHHNSJOQ-UHFFFAOYSA-N alumane Chemical class [AlH3] AZDRQVAHHNSJOQ-UHFFFAOYSA-N 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- 239000010405 anode material Substances 0.000 description 1
- 150000001454 anthracenes Chemical class 0.000 description 1
- 125000002178 anthracenyl group Chemical group C1(=CC=CC2=CC3=CC=CC=C3C=C12)* 0.000 description 1
- PYKYMHQGRFAEBM-UHFFFAOYSA-N anthraquinone Natural products CCC(=O)c1c(O)c2C(=O)C3C(C=CC=C3O)C(=O)c2cc1CC(=O)OC PYKYMHQGRFAEBM-UHFFFAOYSA-N 0.000 description 1
- 150000004056 anthraquinones Chemical class 0.000 description 1
- RJGDLRCDCYRQOQ-UHFFFAOYSA-N anthrone Chemical compound C1=CC=C2C(=O)C3=CC=CC=C3CC2=C1 RJGDLRCDCYRQOQ-UHFFFAOYSA-N 0.000 description 1
- 125000003609 aryl vinyl group Chemical group 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 229910052788 barium Inorganic materials 0.000 description 1
- DSAJWYNOEDNPEQ-UHFFFAOYSA-N barium atom Chemical compound [Ba] DSAJWYNOEDNPEQ-UHFFFAOYSA-N 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 150000001556 benzimidazoles Chemical class 0.000 description 1
- 125000003785 benzimidazolyl group Chemical group N1=C(NC2=C1C=CC=C2)* 0.000 description 1
- XSCHRSMBECNVNS-UHFFFAOYSA-N benzopyrazine Natural products N1=CC=NC2=CC=CC=C21 XSCHRSMBECNVNS-UHFFFAOYSA-N 0.000 description 1
- GQVWHWAWLPCBHB-UHFFFAOYSA-L beryllium;benzo[h]quinolin-10-olate Chemical compound [Be+2].C1=CC=NC2=C3C([O-])=CC=CC3=CC=C21.C1=CC=NC2=C3C([O-])=CC=CC3=CC=C21 GQVWHWAWLPCBHB-UHFFFAOYSA-L 0.000 description 1
- 150000001616 biphenylenes Chemical group 0.000 description 1
- UFVXQDWNSAGPHN-UHFFFAOYSA-K bis[(2-methylquinolin-8-yl)oxy]-(4-phenylphenoxy)alumane Chemical compound [Al+3].C1=CC=C([O-])C2=NC(C)=CC=C21.C1=CC=C([O-])C2=NC(C)=CC=C21.C1=CC([O-])=CC=C1C1=CC=CC=C1 UFVXQDWNSAGPHN-UHFFFAOYSA-K 0.000 description 1
- 229920001400 block copolymer Polymers 0.000 description 1
- 150000001638 boron Chemical class 0.000 description 1
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 229910052792 caesium Inorganic materials 0.000 description 1
- TVFDJXOCXUVLDH-UHFFFAOYSA-N caesium atom Chemical compound [Cs] TVFDJXOCXUVLDH-UHFFFAOYSA-N 0.000 description 1
- 150000001716 carbazoles Chemical group 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 239000000460 chlorine Substances 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- XOYLJNJLGBYDTH-UHFFFAOYSA-M chlorogallium Chemical compound [Ga]Cl XOYLJNJLGBYDTH-UHFFFAOYSA-M 0.000 description 1
- 229910052804 chromium Inorganic materials 0.000 description 1
- 239000011651 chromium Substances 0.000 description 1
- 125000002676 chrysenyl group Chemical group C1(=CC=CC=2C3=CC=C4C=CC=CC4=C3C=CC12)* 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000004210 cyclohexylmethyl group Chemical group [H]C([H])(*)C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 125000000640 cyclooctyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000004851 cyclopentylmethyl group Chemical group C1(CCCC1)C* 0.000 description 1
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- 150000004826 dibenzofurans Chemical class 0.000 description 1
- 238000003618 dip coating Methods 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- 239000010408 film Substances 0.000 description 1
- GVEPBJHOBDJJJI-UHFFFAOYSA-N fluoranthrene Natural products C1=CC(C2=CC=CC=C22)=C3C2=CC=CC3=C1 GVEPBJHOBDJJJI-UHFFFAOYSA-N 0.000 description 1
- YLQWCDOCJODRMT-UHFFFAOYSA-N fluoren-9-one Chemical compound C1=CC=C2C(=O)C3=CC=CC=C3C2=C1 YLQWCDOCJODRMT-UHFFFAOYSA-N 0.000 description 1
- 125000005567 fluorenylene group Chemical group 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 150000002240 furans Chemical class 0.000 description 1
- UIWYJDYFSGRHKR-UHFFFAOYSA-N gadolinium atom Chemical compound [Gd] UIWYJDYFSGRHKR-UHFFFAOYSA-N 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 1
- 229910052737 gold Inorganic materials 0.000 description 1
- 239000010931 gold Substances 0.000 description 1
- 125000003187 heptyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- 229910052738 indium Inorganic materials 0.000 description 1
- APFVFJFRJDLVQX-UHFFFAOYSA-N indium atom Chemical compound [In] APFVFJFRJDLVQX-UHFFFAOYSA-N 0.000 description 1
- 229910003437 indium oxide Inorganic materials 0.000 description 1
- PJXISJQVUVHSOJ-UHFFFAOYSA-N indium(iii) oxide Chemical compound [O-2].[O-2].[O-2].[In+3].[In+3] PJXISJQVUVHSOJ-UHFFFAOYSA-N 0.000 description 1
- 125000001041 indolyl group Chemical group 0.000 description 1
- 238000007641 inkjet printing Methods 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 125000002183 isoquinolinyl group Chemical group C1(=NC=CC2=CC=CC=C12)* 0.000 description 1
- 238000010030 laminating Methods 0.000 description 1
- 239000011133 lead Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- XNUVVHVFAAQPQY-UHFFFAOYSA-L manganese(2+) quinolin-8-olate Chemical compound N1=CC=CC2=CC=CC(=C12)[O-].[Mn+2].N1=CC=CC2=CC=CC(=C12)[O-] XNUVVHVFAAQPQY-UHFFFAOYSA-L 0.000 description 1
- 238000004776 molecular orbital Methods 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000003136 n-heptyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 150000002790 naphthalenes Chemical class 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 125000004433 nitrogen atom Chemical group N* 0.000 description 1
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000005416 organic matter Substances 0.000 description 1
- 150000004866 oxadiazoles Chemical class 0.000 description 1
- 125000001715 oxadiazolyl group Chemical group 0.000 description 1
- 125000002971 oxazolyl group Chemical group 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 125000004430 oxygen atom Chemical group O* 0.000 description 1
- 229960003540 oxyquinoline Drugs 0.000 description 1
- 150000002964 pentacenes Chemical class 0.000 description 1
- 125000003538 pentan-3-yl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 1
- FVDOBFPYBSDRKH-UHFFFAOYSA-N perylene-3,4,9,10-tetracarboxylic acid Chemical compound C=12C3=CC=C(C(O)=O)C2=C(C(O)=O)C=CC=1C1=CC=C(C(O)=O)C2=C1C3=CC=C2C(=O)O FVDOBFPYBSDRKH-UHFFFAOYSA-N 0.000 description 1
- CSHWQDPOILHKBI-UHFFFAOYSA-N peryrene Natural products C1=CC(C2=CC=CC=3C2=C2C=CC=3)=C3C2=CC=CC3=C1 CSHWQDPOILHKBI-UHFFFAOYSA-N 0.000 description 1
- 150000005041 phenanthrolines Chemical class 0.000 description 1
- 125000005561 phenanthryl group Chemical group 0.000 description 1
- 229910052697 platinum Inorganic materials 0.000 description 1
- 229920002098 polyfluorene Polymers 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 229920000128 polypyrrole Polymers 0.000 description 1
- 229920000123 polythiophene Polymers 0.000 description 1
- 150000004032 porphyrins Chemical class 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000001042 pteridinyl group Chemical group N1=C(N=CC2=NC=CN=C12)* 0.000 description 1
- FQOBINBWTPHVEO-UHFFFAOYSA-N pyrazino[2,3-b]pyrazine Chemical compound N1=CC=NC2=NC=CN=C21 FQOBINBWTPHVEO-UHFFFAOYSA-N 0.000 description 1
- 125000003373 pyrazinyl group Chemical group 0.000 description 1
- 150000003220 pyrenes Chemical class 0.000 description 1
- 125000001725 pyrenyl group Chemical group 0.000 description 1
- PBMFSQRYOILNGV-UHFFFAOYSA-N pyridazine Chemical group C1=CC=NN=C1 PBMFSQRYOILNGV-UHFFFAOYSA-N 0.000 description 1
- 229940083082 pyrimidine derivative acting on arteriolar smooth muscle Drugs 0.000 description 1
- 150000003230 pyrimidines Chemical class 0.000 description 1
- 125000002294 quinazolinyl group Chemical group N1=C(N=CC2=CC=CC=C12)* 0.000 description 1
- MCJGNVYPOGVAJF-UHFFFAOYSA-N quinolin-8-ol Chemical compound C1=CN=C2C(O)=CC=CC2=C1 MCJGNVYPOGVAJF-UHFFFAOYSA-N 0.000 description 1
- 125000002943 quinolinyl group Chemical group N1=C(C=CC2=CC=CC=C12)* 0.000 description 1
- 125000001567 quinoxalinyl group Chemical group N1=C(C=NC2=CC=CC=C12)* 0.000 description 1
- 239000000376 reactant Substances 0.000 description 1
- YYMBJDOZVAITBP-UHFFFAOYSA-N rubrene Chemical compound C1=CC=CC=C1C(C1=C(C=2C=CC=CC=2)C2=CC=CC=C2C(C=2C=CC=CC=2)=C11)=C(C=CC=C2)C2=C1C1=CC=CC=C1 YYMBJDOZVAITBP-UHFFFAOYSA-N 0.000 description 1
- KZUNJOHGWZRPMI-UHFFFAOYSA-N samarium atom Chemical compound [Sm] KZUNJOHGWZRPMI-UHFFFAOYSA-N 0.000 description 1
- 238000007650 screen-printing Methods 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 125000003548 sec-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 229910052711 selenium Inorganic materials 0.000 description 1
- 229910052710 silicon Inorganic materials 0.000 description 1
- 125000003808 silyl group Chemical group [H][Si]([H])([H])[*] 0.000 description 1
- 239000002356 single layer Substances 0.000 description 1
- 210000000813 small intestine Anatomy 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 238000004528 spin coating Methods 0.000 description 1
- 150000003413 spiro compounds Chemical class 0.000 description 1
- 125000003003 spiro group Chemical group 0.000 description 1
- 238000005507 spraying Methods 0.000 description 1
- 238000004544 sputter deposition Methods 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 125000005504 styryl group Chemical group 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 125000004434 sulfur atom Chemical group 0.000 description 1
- 229940042055 systemic antimycotics triazole derivative Drugs 0.000 description 1
- 238000010345 tape casting Methods 0.000 description 1
- 125000006836 terphenylene group Chemical group 0.000 description 1
- 125000001113 thiadiazolyl group Chemical group 0.000 description 1
- 125000000335 thiazolyl group Chemical group 0.000 description 1
- 229930192474 thiophene Natural products 0.000 description 1
- IBBLKSWSCDAPIF-UHFFFAOYSA-N thiopyran Chemical compound S1C=CC=C=C1 IBBLKSWSCDAPIF-UHFFFAOYSA-N 0.000 description 1
- 229910052718 tin Inorganic materials 0.000 description 1
- 239000011135 tin Substances 0.000 description 1
- 229910052719 titanium Inorganic materials 0.000 description 1
- 239000010936 titanium Substances 0.000 description 1
- KWQNQSDKCINQQP-UHFFFAOYSA-K tri(quinolin-8-yloxy)gallane Chemical compound C1=CN=C2C(O[Ga](OC=3C4=NC=CC=C4C=CC=3)OC=3C4=NC=CC=C4C=CC=3)=CC=CC2=C1 KWQNQSDKCINQQP-UHFFFAOYSA-K 0.000 description 1
- BDZBKCUKTQZUTL-UHFFFAOYSA-N triethyl phosphite Chemical compound CCOP(OCC)OCC BDZBKCUKTQZUTL-UHFFFAOYSA-N 0.000 description 1
- FJYFPBQHYWBQFL-UHFFFAOYSA-N triphenylen-1-ylboronic acid Chemical compound C1(=CC=CC=2C3=CC=CC=C3C3=CC=CC=C3C1=2)B(O)O FJYFPBQHYWBQFL-UHFFFAOYSA-N 0.000 description 1
- 125000005580 triphenylene group Chemical group 0.000 description 1
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical compound OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 1
- 238000001771 vacuum deposition Methods 0.000 description 1
- 229910052720 vanadium Inorganic materials 0.000 description 1
- GPPXJZIENCGNKB-UHFFFAOYSA-N vanadium Chemical compound [V]#[V] GPPXJZIENCGNKB-UHFFFAOYSA-N 0.000 description 1
- 238000007740 vapor deposition Methods 0.000 description 1
- NAWDYIZEMPQZHO-UHFFFAOYSA-N ytterbium Chemical compound [Yb] NAWDYIZEMPQZHO-UHFFFAOYSA-N 0.000 description 1
- 229910052727 yttrium Inorganic materials 0.000 description 1
- VWQVUPCCIRVNHF-UHFFFAOYSA-N yttrium atom Chemical compound [Y] VWQVUPCCIRVNHF-UHFFFAOYSA-N 0.000 description 1
- YVTHLONGBIQYBO-UHFFFAOYSA-N zinc indium(3+) oxygen(2-) Chemical compound [O--].[Zn++].[In+3] YVTHLONGBIQYBO-UHFFFAOYSA-N 0.000 description 1
- HTPBWAPZAJWXKY-UHFFFAOYSA-L zinc;quinolin-8-olate Chemical compound [Zn+2].C1=CN=C2C([O-])=CC=CC2=C1.C1=CN=C2C([O-])=CC=CC2=C1 HTPBWAPZAJWXKY-UHFFFAOYSA-L 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/04—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/656—Aromatic compounds comprising a hetero atom comprising two or more different heteroatoms per ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/10—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings linked by a carbon chain containing aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/14—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing three or more hetero rings
-
- H01L51/0071—
-
- H01L51/0072—
-
- H01L51/5012—
-
- H01L51/5048—
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/14—Carrier transporting layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6572—Polycyclic condensed heteroaromatic hydrocarbons comprising only nitrogen in the heteroaromatic polycondensed ring system, e.g. phenanthroline or carbazole
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Physics & Mathematics (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Optics & Photonics (AREA)
- Electroluminescent Light Sources (AREA)
Abstract
Description
본 명세서는 화합물 및 이를 포함하는 유기 발광 소자에 관한 것이다. The present specification relates to a compound and an organic light emitting device including the same.
본 출원은 2018년 11월 02일 한국특허청에 제출된 한국 특허 출원 제10-2018-0133636호의 출원일의 이익을 주장하며, 그 내용 전부는 본 명세서에 포함된다.This application claims the benefit of the filing date of Korean Patent Application No. 10-2018-0133636 filed with the Korean Intellectual Property Office on November 02, 2018, all of which is included in this specification.
일반적으로 유기 발광 현상이란 유기 물질을 이용하여 전기에너지를 빛에너지로 전환시켜주는 현상을 말한다. 유기 발광 현상을 이용하는 유기 발광 소자는 통상 양극과 음극 및 이 사이에 유기물층을 포함하는 구조를 가진다. 여기서 유기물층은 유기 발광 소자의 효율과 안정성을 높이기 위하여 각기 다른 물질로 구성된 다층의 구조로 이루어진 경우가 많으며, 예컨대 정공 주입층, 정공 수송층, 발광층, 전자 수송층, 전자 주입층 등으로 이루어 질 수 있다. 이러한 유기 발광 소자의 구조에서 두 전극 사이에 전압을 걸어주게 되면 양극에서는 정공이, 음극에서는 전자가 유기물층에 주입되게 되고, 주입된 정공과 전자가 만났을 때 엑시톤(exciton)이 형성되며, 이 엑시톤이 다시 바닥상태로 떨어질 때 빛이 나게 된다. In general, the organic light emitting phenomenon refers to a phenomenon that converts electrical energy into light energy using an organic material. An organic light emitting device using an organic light emitting phenomenon usually has a structure including an anode and a cathode and an organic material layer therebetween. Here, in order to increase the efficiency and stability of the organic light emitting device, the organic material layer is often composed of a multi-layered structure composed of different materials, for example, may be formed of a hole injection layer, a hole transport layer, a light emitting layer, an electron transport layer, an electron injection layer, or the like. When a voltage is applied between two electrodes in the structure of the organic light emitting device, holes are injected at the anode, and electrons are injected at the cathode, and an exciton is formed when the injected holes meet the electrons. When it falls to the ground again, it glows.
상기와 같은 유기 발광 소자를 위한 새로운 재료의 개발이 계속 요구되고 있다. The development of new materials for such organic light emitting devices continues to be required.
본 명세서는 화합물 및 이를 포함하는 유기 발광 소자를 제공하고자 한다.The present specification is intended to provide a compound and an organic light emitting device including the same.
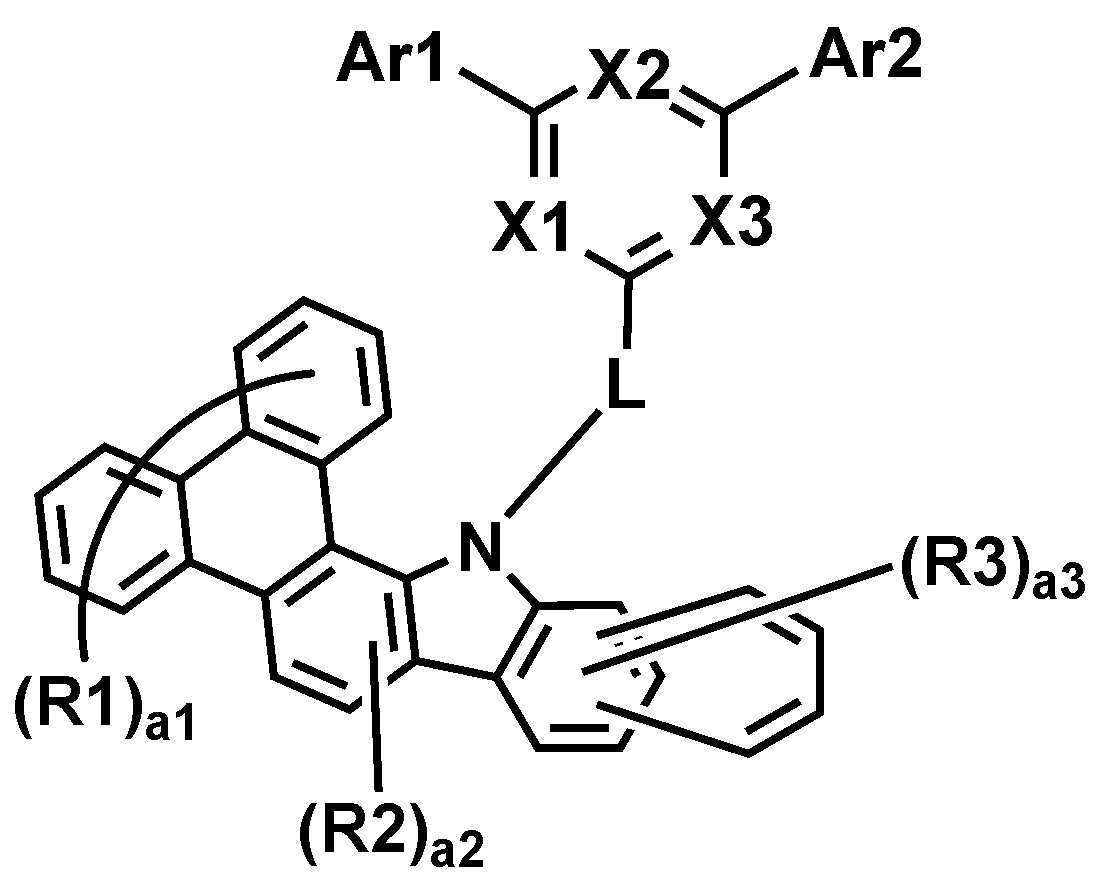
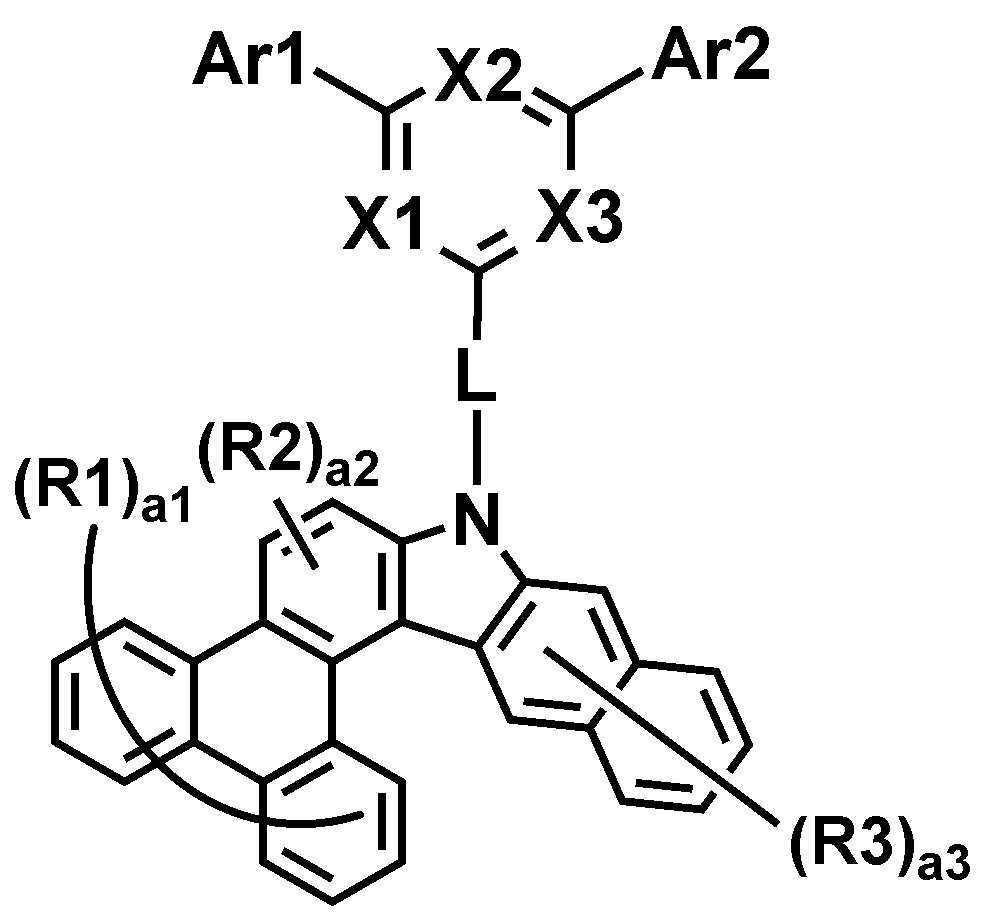
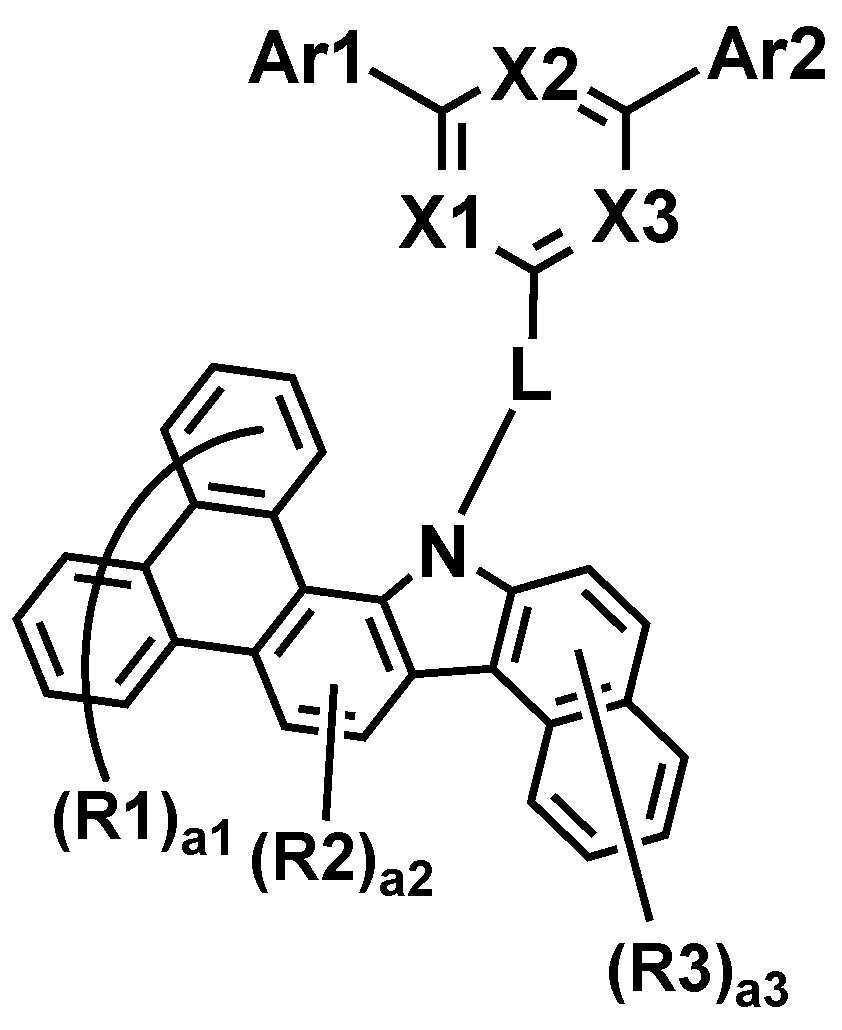
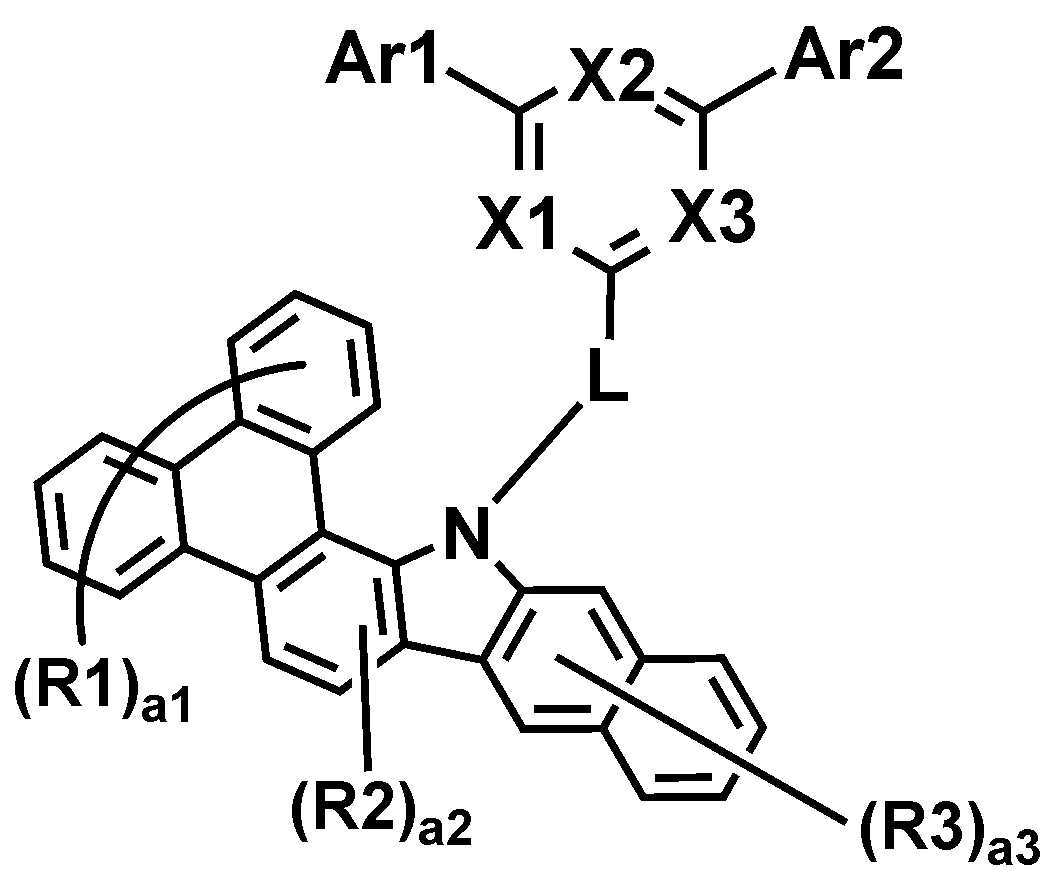
본 명세서는 하기 화학식 1로 표시되는 화합물을 제공한다.The present specification provides a compound represented by Formula 1 below.
상기 화학식 1에 있어서,In Chemical Formula 1,
*은 상기 화학식 1-1의 *와 축합하는 위치이며,* Is a position condensed with * in Chemical Formula 1-1,
R1 및 R2는 서로 동일하거나 상이하고, 각각 독립적으로 수소; 또는 중수소이고,R1 and R2 are the same as or different from each other, and each independently hydrogen; Or deuterium,
a1은 0 내지 8의 정수이고, a1이 2 이상인 경우, R1은 서로 동일하거나 상이하고,a1 is an integer from 0 to 8, and when a1 is 2 or more, R1 is the same as or different from each other,
a2은 0 내지 2의 정수이고, a2이 2인 경우, R2은 서로 동일하거나 상이하고,a2 is an integer from 0 to 2, and when a2 is 2, R2 is the same or different from each other,
상기 화학식 1-1에 있어서,In Chemical Formula 1-1,
X1 내지 X3는 서로 동일하거나 상이하고, 각각 독립적으로 N; 또는 CH이고, X1 내지 X3 중 2 이상은 N이고,X1 to X3 are the same as or different from each other, and each independently N; Or CH, and 2 or more of X1 to X3 are N,
Ar1 및 Ar2는 서로 동일하거나 상이하고, 각각 독립적으로 치환 또는 비치환된 아릴기; 또는 치환 또는 비치환된 헤테로아릴기이고,Ar1 and Ar2 are the same as or different from each other, and each independently a substituted or unsubstituted aryl group; Or a substituted or unsubstituted heteroaryl group,
L은 직접결합; 치환 또는 비치환된 아릴렌기; 또는 치환 또는 비치환된 헤테로아릴렌기이고,L is a direct bond; A substituted or unsubstituted arylene group; Or a substituted or unsubstituted heteroarylene group,
R3는 수소; 또는 중수소이고,R3 is hydrogen; Or deuterium,
a3은 0 내지 6의 정수이고, a3이 2 이상인 경우, R3은 서로 동일하거나 상이하다.a3 is an integer of 0 to 6, and when a3 is 2 or more, R3 is the same or different from each other.
또한, 본 명세서는 제1 전극; 상기 제1 전극과 대향하여 구비된 제2 전극; 및 상기 제1 전극과 상기 제2 전극 사이에 구비된 1층 이상의 유기물층을 포함하는 유기 발광 소자로서, 상기 유기물층 중 1층 이상은 상기 화합물을 포함하는 것인 유기 발광 소자를 제공한다.In addition, the present specification is a first electrode; A second electrode provided to face the first electrode; And an organic light emitting device including at least one organic material layer provided between the first electrode and the second electrode, wherein at least one layer of the organic material layer includes the compound.
본 명세서의 일 실시상태에 따른 화합물은 유기 발광 소자에 사용되어, 유기 발광 소자의 구동전압을 낮출 수 있으며, 광효율을 향상시킬 수 있다. 또한, 화합물의 열적 안정성에 의하여 소장의 수명 특성을 향상시킬 수 있다. The compound according to one embodiment of the present specification is used in an organic light emitting device, so that the driving voltage of the organic light emitting device can be lowered, and light efficiency can be improved. In addition, the lifespan characteristics of the small intestine can be improved by the thermal stability of the compound.
도 1 내지 도 3은 본 명세서의 일 실시상태에 따른 유기 발광 소자의 예를 도시한 것이다.1 to 3 show an example of an organic light emitting device according to an exemplary embodiment of the present specification.
이하, 본 명세서에 대하여 더욱 상세하게 설명한다.Hereinafter, the present specification will be described in more detail.
본 명세서는 상기 화학식 1로 표시되는 화합물을 제공한다.The present specification provides a compound represented by Chemical Formula 1.
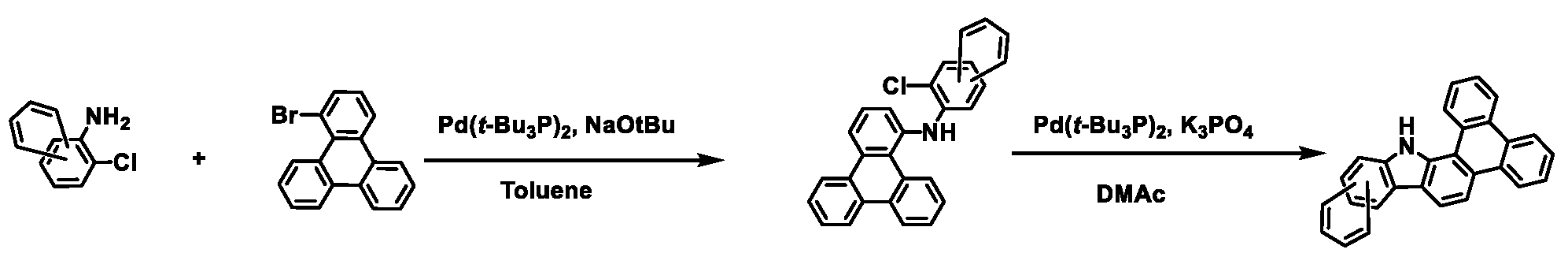
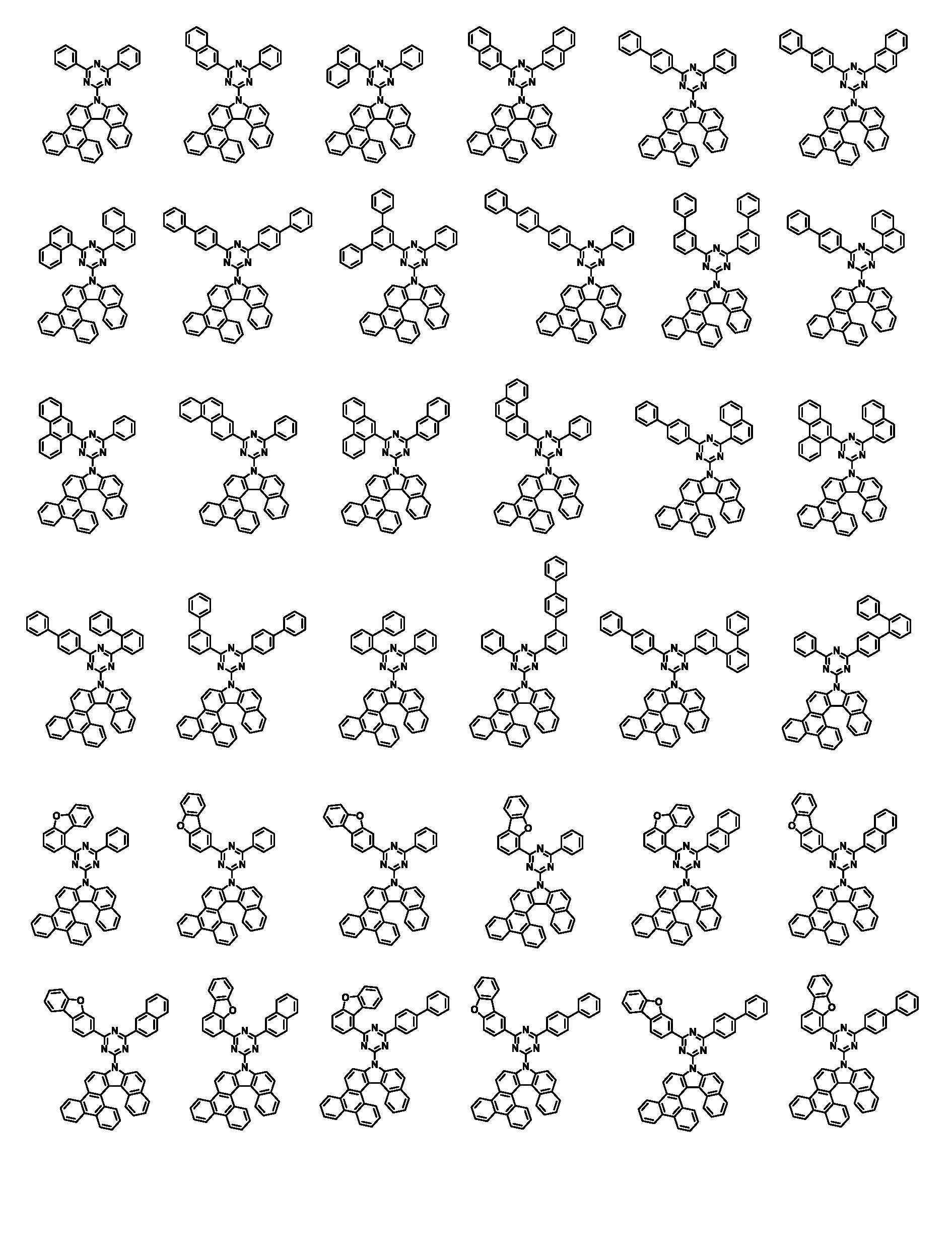
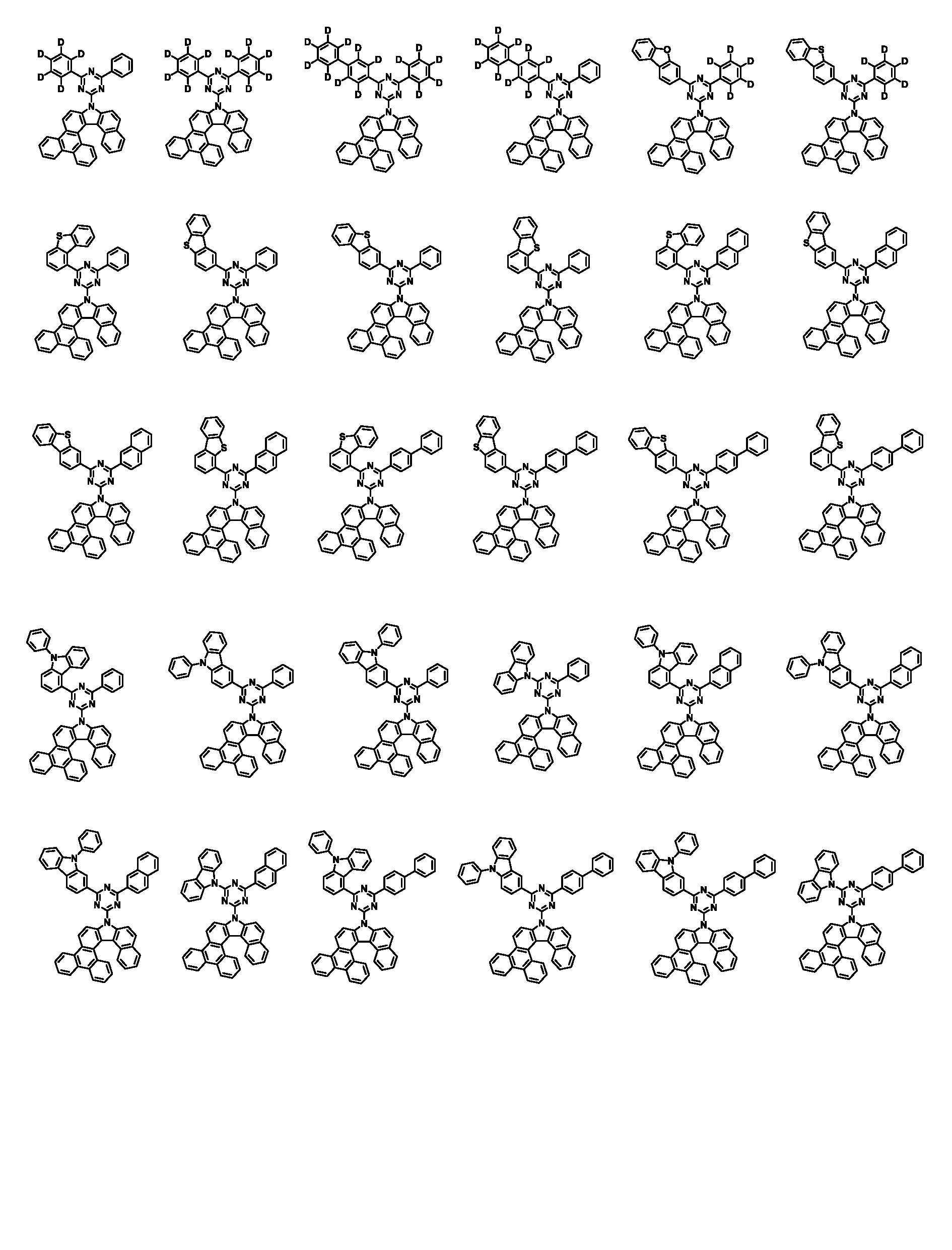
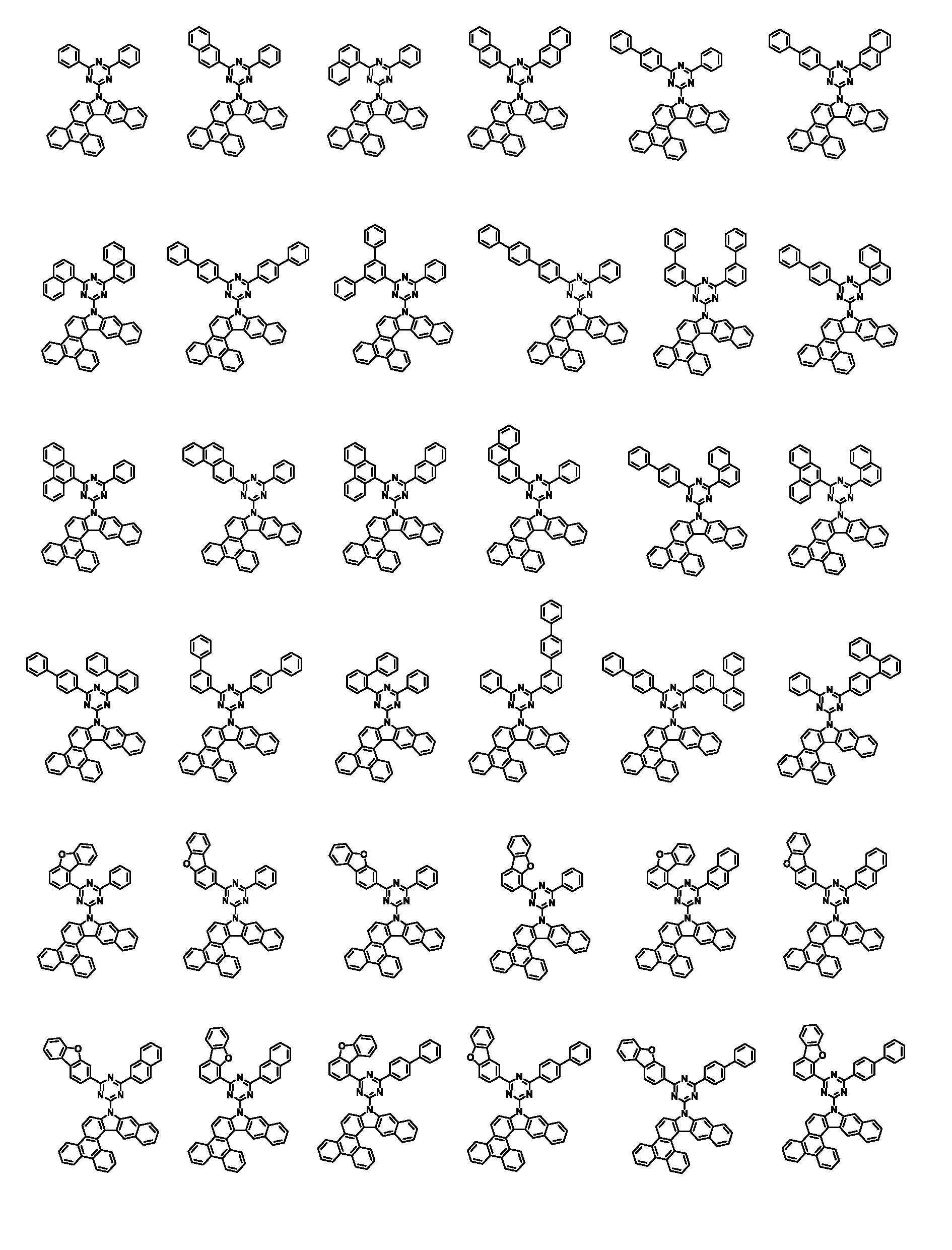
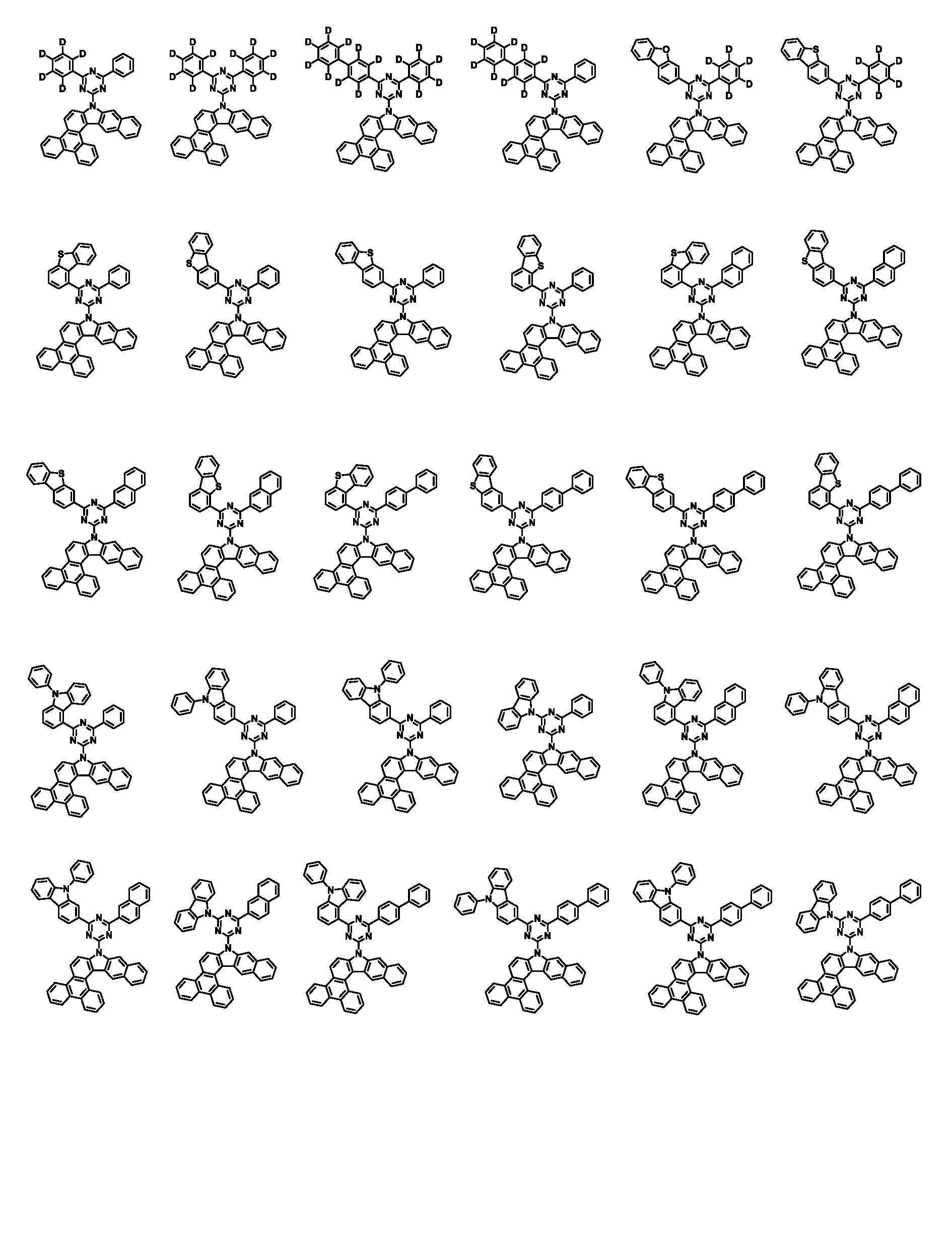
상기 화학식 1로 표시되는 화합물은 트리페닐렌기의 1번 및 2번 탄소에 벤조인돌기가 축합된 코어 구조에 아릴기/헤테로아릴기가 치환된 피리미딘/트리아진이 연결되어 있다. 상기 화학식 1의 화합물이 발광층의 호스트로 사용될 때, 전자 및 정공에 대한 안정도가 높으며, 전자 및 정공의 균형을 잘 맞출수 있어, 소자의 전압, 발광 효율 및 수명이 개선된다.In the compound represented by Chemical Formula 1, a pyrimidine / triazine substituted with an aryl group / heteroaryl group is connected to a core structure in which benzoindol groups are condensed on carbons 1 and 2 of the triphenylene group. When the compound of Formula 1 is used as a host for a light emitting layer, stability to electrons and holes is high, and the balance of electrons and holes can be well balanced, thereby improving the voltage, luminous efficiency, and lifetime of the device.
본 명세서에서 치환기의 예시들은 아래에서 설명하나, 이에 한정되는 것은 아니다. Examples of substituents herein are described below, but are not limited thereto.
본 명세서에 있어서, 는 연결되는 부위를 의미한다.In this specification, Means the site to be connected.
상기 "치환"이라는 용어는 화합물의 탄소 원자에 결합된 수소 원자가 다른 치환기로 바뀌는 것을 의미하며, 치환되는 위치는 수소 원자가 치환되는 위치 즉, 치환기가 치환 가능한 위치라면 한정하지 않으며, 2 이상 치환되는 경우, 2 이상의 치환기는 서로 동일하거나 상이할 수 있다.The term "substitution" means that the hydrogen atom bonded to the carbon atom of the compound is replaced with another substituent, and the position to be substituted is not limited to a position where the hydrogen atom is substituted, that is, a position where the substituent can be substituted, and when two or more are substituted , 2 or more substituents may be the same or different from each other.
본 명세서에서 "치환 또는 비치환된" 이라는 용어는 중수소; 할로겐기; 니트릴기; 알킬기; 시클로알킬기; 아민기; 아릴기; 및 N, O, 및 S 원자 중 1개 이상을 포함하는 헤테로아릴기로 이루어진 군에서 선택된 1 또는 2 이상의 치환기로 치환되었거나 상기 예시된 치환기 중 2 이상의 치환기가 연결된 치환기로 치환되거나, 또는 어떠한 치환기도 갖지 않는 것을 의미한다. The term "substituted or unsubstituted" as used herein refers to deuterium; Halogen group; Nitrile group; Alkyl groups; Cycloalkyl group; Amine group; Aryl group; And one or two or more substituents selected from the group consisting of heteroaryl groups containing one or more of N, O, and S atoms, or substituted with a substituent connected by two or more of the exemplified substituents, or have no substituents. It means not.
본 명세서에 있어서, 할로겐기의 예로는 불소, 염소, 브롬, 또는 요오드가 있다. In the present specification, examples of the halogen group include fluorine, chlorine, bromine, or iodine.
본 명세서에 있어서, 알킬기는 직쇄 또는 분지쇄일 수 있고, 탄소수는 특별히 한정되지 않으나 1 내지 50인 것이 바람직하고, 1 내지 30이 더욱 바람직하다. 구체적인 예로는 메틸, 에틸, 프로필, n-프로필, 이소프로필, 부틸, n-부틸, 이소부틸, tert-부틸, sec-부틸, 1-메틸부틸, 1-에틸부틸, 펜틸, n-펜틸, 이소펜틸, 네오펜틸, tert-펜틸, 헥실, n-헥실, 1-메틸펜틸, 2-메틸펜틸, 4-메틸펜틸, 3,3-디메틸부틸, 2-에틸부틸, 헵틸, n-헵틸, 1-메틸헥실, 시클로펜틸메틸, 시클로헥실메틸, 옥틸, n-옥틸, tert-옥틸, 1-메틸헵틸, 2-에틸헥실, 2-프로필펜틸, n-노닐, 2,2-디메틸헵틸, 1-에틸프로필, 1,1-디메틸프로필, 이소헥실, 4-메틸헥실, 5-메틸헥실 등이 있으나, 이에 한정되는 것은 아니다. In the present specification, the alkyl group may be straight chain or branched chain, and the number of carbon atoms is not particularly limited, but is preferably 1 to 50, more preferably 1 to 30. Specific examples are methyl, ethyl, propyl, n-propyl, isopropyl, butyl, n-butyl, isobutyl, tert-butyl, sec-butyl, 1-methylbutyl, 1-ethylbutyl, pentyl, n-pentyl, iso Pentyl, neopentyl, tert-pentyl, hexyl, n-hexyl, 1-methylpentyl, 2-methylpentyl, 4-methylpentyl, 3,3-dimethylbutyl, 2-ethylbutyl, heptyl, n-heptyl, 1- Methylhexyl, cyclopentylmethyl, cyclohexylmethyl, octyl, n-octyl, tert-octyl, 1-methylheptyl, 2-ethylhexyl, 2-propylpentyl, n-nonyl, 2,2-dimethylheptyl, 1-ethyl Propyl, 1,1-dimethylpropyl, isohexyl, 4-methylhexyl, 5-methylhexyl, and the like, but is not limited thereto.
본 명세서에 있어서, 시클로알킬기는 특별히 한정되지 않으나, 탄소수 3 내지 60인 것이 바람직하며, 3 내지 30이 더욱 바람직하다. 구체적으로, 시클로프로필, 시클로부틸, 시클로펜틸, 3-메틸시클로펜틸, 2,3-디메틸시클로펜틸, 시클로헥실, 3-메틸시클로헥실, 4-메틸시클로헥실, 2,3-디메틸시클로헥실, 3,4,5-트리메틸시클로헥실, 4-tert-부틸시클로헥실, 시클로헵틸, 시클로옥틸 등이 있으나, 이에 한정되는 것은 아니다.In the present specification, the cycloalkyl group is not particularly limited, but is preferably 3 to 60 carbon atoms, and more preferably 3 to 30 carbon atoms. Specifically, cyclopropyl, cyclobutyl, cyclopentyl, 3-methylcyclopentyl, 2,3-dimethylcyclopentyl, cyclohexyl, 3-methylcyclohexyl, 4-methylcyclohexyl, 2,3-dimethylcyclohexyl, 3 , 4,5-trimethylcyclohexyl, 4-tert-butylcyclohexyl, cycloheptyl, cyclooctyl, and the like, but is not limited thereto.
본 명세서에서 아릴기가 단환식 아릴기인 경우 탄소수는 특별히 한정되지 않으나, 탄소수 6 내지 50인 것이 바람직하고, 6 내지 30이 더욱 바람직하다. 구체적으로, 단환식 아릴기로는 페닐기, 비페닐기, 터페닐기, 쿼터페닐기 등이 될 수 있으나, 이에 한정되는 것은 아니다. In the present specification, when the aryl group is a monocyclic aryl group, the number of carbon atoms is not particularly limited, but is preferably 6 to 50 carbon atoms, more preferably 6 to 30 carbon atoms. Specifically, the monocyclic aryl group may be a phenyl group, a biphenyl group, a terphenyl group, a quarterphenyl group, and the like, but is not limited thereto.
상기 아릴기가 다환식 아릴기인 경우 탄소수는 특별히 한정되지 않으나. 탄소수 10 내지 50인 것이 바람직하고, 10 내지 30이 더욱 바람직하다. 구체적으로, 다환식 아릴기로는 나프틸기, 안트라세닐기, 페난트릴기, 파이레닐기, 페릴레닐기, 트리페닐기, 크라이세닐기, 플루오레닐기 등이 될 수 있으나, 이에 한정되는 것은 아니다.When the aryl group is a polycyclic aryl group, the number of carbon atoms is not particularly limited. It is preferable that it is 10-50 carbon atoms, and 10-30 is more preferable. Specifically, the polycyclic aryl group may be a naphthyl group, anthracenyl group, phenanthryl group, pyrenyl group, perylene group, triphenyl group, chrysenyl group, fluorenyl group, but is not limited thereto.
본 명세서에 있어서, 상기 플루오레닐기는 치환될 수 있으며, 인접한 치환기들이 서로 결합하여 고리를 형성할 수 있다.In the present specification, the fluorenyl group may be substituted, and adjacent substituents may combine with each other to form a ring.
상기 플루오레닐기가 치환되는 경우, 9번 탄소의 치환기가 고리를 형성하여 스피로구조를 형성할 수 있다. 스피로비플루오레닐기 등이 될 수 있으나, 이에 한정되는 것은 아니다.When the fluorenyl group is substituted, the substituent of carbon 9 may form a ring to form a spiro structure. Spirobifluorenyl group and the like, but is not limited thereto.
본 명세서에 있어서, 헤테로아릴기는 이종원자로 N, O, S, Si 및 Se 중 1개 이상을 포함하는 것으로서, 탄소수는 특별히 한정되지 않으나 탄소수 2 내지 60인 것이 바람직하고, 2 내지 30인 것이 더욱 바람직하다. 헤테로아릴기의 예로는, 티오펜기, 퓨란기, 피롤기, 이미다졸기, 티아졸기, 옥사졸기, 옥사디아졸기, 트리아졸기, 피리딘기, 비피리딘기, 피리미딘기, 트리아진기, 아크리딘기, 피리다진기, 피라진기, 퀴놀린기, 퀴나졸린기, 퀴녹살린기, 프탈라진기(phthalazine), 프테리딘기(pteridine), 피리도 피리미딘기(pyrido pyrimidine), 피리도 피라진기(pyrido pyrazine), 피라지노 피라진기(pyrazino pyrazine), 이소퀴놀린기, 인돌기, 피리도 인돌기(pyrido indole), 인데노 피리미딘(5H-indeno pyrimidine), 카바졸기, 벤즈옥사졸기, 벤즈이미다졸기, 벤조티아졸기, 벤조카바졸기, 벤조티오펜기, 디벤조티오펜기, 벤조퓨란기, 디벤조퓨란기, 페난쓰롤린기(phenanthroline), 티아졸릴기, 이소옥사졸릴기, 옥사디아졸릴기 및 티아디아졸릴기 등이 있으나, 이들에만 한정되는 것은 아니다.In the present specification, the heteroaryl group is a heteroatom containing at least one of N, O, S, Si, and Se, and carbon number is not particularly limited, but is preferably 2 to 60 carbon atoms, and more preferably 2 to 30 carbon atoms. Do. Examples of the heteroaryl group include thiophene group, furan group, pyrrol group, imidazole group, thiazole group, oxazole group, oxadiazole group, triazole group, pyridine group, bipyridine group, pyrimidine group, triazine group, and acry Din group, pyridazine group, pyrazine group, quinoline group, quinazoline group, quinoxaline group, phthalazine group, phthalazine, pteridine group, pyrido pyrimidine group, pyrido pyrimidine group pyrazine), pyrazino pyrazine, isoquinoline group, indole group, pyrido indole, indo pyrimidine, 5H-indeno pyrimidine, carbazole group, benzoxazole group, benzimidazole group , Benzothiazole group, benzocarbazole group, benzothiophene group, dibenzothiophene group, benzofuran group, dibenzofuran group, phenanthroline group, thiazolyl group, isooxazolyl group, oxadiazolyl group and Thiadiazolyl groups, and the like, but are not limited to these.
본 명세서에 있어서, 아릴렌기는 아릴기에 결합 위치가 두 개 있는 것 즉 2가기를 의미한다. 이들은 각각 2가기인 것을 제외하고는 전술한 아릴기의 설명이 적용될 수 있다.In the present specification, the arylene group means that the aryl group has two bonding positions, that is, a divalent group. These may be applied to the description of the aryl group described above, except that each is a divalent group.
본 명세서에 있어서, 헤테로아릴렌기는 헤테로아릴기에 결합 위치가 두 개 있는 것 즉 2가기를 의미한다. 이들은 각각 2가기인 것을 제외하고는 전술한 헤테로아릴기의 설명이 적용될 수 있다.In the present specification, the heteroarylene group means that the heteroaryl group has two bonding positions, that is, a divalent group. These may be applied to the description of the heteroaryl group described above, except that each is a divalent group.
본 명세서에 있어서, "인접한" 기는 해당 치환기가 치환된 원자와 직접 연결된 원자에 치환된 치환기, 해당 치환기와 입체구조적으로 가장 가깝게 위치한 치환기, 또는 해당 치환기가 치환된 원자에 치환된 다른 치환기를 의미할 수 있다. 예컨대, 벤젠고리에서 오르토(ortho)위치로 치환된 2개의 치환기 및 지방족 고리에서 동일 탄소에 치환된 2개의 치환기는 서로 "인접한" 기로 해석될 수 있다.In the present specification, the “adjacent” group refers to a substituent substituted on an atom directly connected to an atom in which the substituent is substituted, a substituent positioned closest to the substituent and the other substituent substituted on the atom in which the substituent is substituted. Can be. For example, two substituents substituted in the ortho position on the benzene ring and two substituents substituted on the same carbon in the aliphatic ring may be interpreted as "adjacent" groups to each other.
본 명세서에 있어서, 인접한 기가 서로 결합하여 형성되는 치환 또는 비치환된 고리에서, "고리"는 치환 또는 비치환된 탄화수소고리; 또는 치환 또는 비치환된 헤테로고리를 의미한다.In the present specification, in a substituted or unsubstituted ring formed by bonding adjacent groups to each other, "ring" is a substituted or unsubstituted hydrocarbon ring; Or a substituted or unsubstituted hetero ring.
본 명세서에 있어서, 탄화수소고리는 방향족, 지방족 또는 방향족과 지방족의 축합고리일 수 있으며, 상기 1가가 아닌 것을 제외하고 상기 시클로알킬기 또는 아릴기의 예시 중에서 선택될 수 있다.In the present specification, the hydrocarbon ring may be an aromatic, aliphatic or aromatic and aliphatic condensed ring, and may be selected from examples of the cycloalkyl group or aryl group except for the non-monovalent.
본 명세서에 있어서, 방향족고리는 단환 또는 다환일 수 있으며, 1가가 아닌 것을 제외하고 상기 아릴기의 예시 중에서 선택될 수 있다.In the present specification, the aromatic ring may be monocyclic or polycyclic, and may be selected from examples of the aryl group, except that it is not monovalent.
본 명세서에 있어서, 헤테로고리는 탄소가 아닌 원자, 이종원자를 1 이상 포함하는 것으로서, 구체적으로 상기 이종 원자는 O, N 및 S 등으로 이루어진 군에서 선택되는 원자를 1 이상 포함할 수 있다. 상기 헤테로고리는 단환 또는 다환일 수 있으며, 방향족, 지방족 또는 방향족과 지방족의 축합고리일 수 있으며, 1가가 아닌 것을 제외하고 상기 헤테로아릴기의 예시 중에서 선택될 수 있다.In the present specification, the heterocycle is a non-carbon atom, and contains one or more heteroatoms. Specifically, the heteroatom may include one or more atoms selected from the group consisting of O, N, and S. The heterocycle may be monocyclic or polycyclic, and may be aromatic, aliphatic or aromatic and aliphatic condensed ring, and may be selected from examples of the heteroaryl group except that it is not monovalent.
본 명세서의 일 실시상태에 있어서, 상기 화학식 1의 *은 상기 화학식 1-1의 *와 축합되는 위치이다.In one embodiment of the present specification, * in Chemical Formula 1 is a position condensed with * in Chemical Formula 1-1.
본 명세서의 일 실시상태에 있어서, L은 직접결합; 치환 또는 비치환된 아릴렌기; 또는 치환 또는 비치환된 헤테로아릴렌기이다.In one embodiment of the present specification, L is a direct bond; A substituted or unsubstituted arylene group; Or a substituted or unsubstituted heteroarylene group.
본 명세서의 일 실시상태에 있어서, L은 직접결합; 치환 또는 비치환된 탄소수 6 내지 30의 아릴렌기; 또는 치환 또는 비치환된 탄소수 2 내지 30의 헤테로아릴렌기이다.In one embodiment of the present specification, L is a direct bond; A substituted or unsubstituted arylene group having 6 to 30 carbon atoms; Or a substituted or unsubstituted heteroarylene group having 2 to 30 carbon atoms.
본 명세서의 일 실시상태에 있어서, L은 직접결합; 탄소수 6 내지 20의 아릴렌기; 또는 탄소수 2 내지 20의 헤테로아릴렌기이다. 상기 아릴렌기 또는 헤테로아릴렌기는 탄소수 1 내지 10의 알킬기, 탄소수 6 내지 20의 아릴기 또는 탄소수 2 내지 20의 헤테로아릴기로 치환되거나 비치환된다.In one embodiment of the present specification, L is a direct bond; An arylene group having 6 to 20 carbon atoms; Or a heteroarylene group having 2 to 20 carbon atoms. The arylene group or heteroarylene group is substituted or unsubstituted with an alkyl group having 1 to 10 carbon atoms, an aryl group having 6 to 20 carbon atoms, or a heteroaryl group having 2 to 20 carbon atoms.
본 명세서의 일 실시상태에 있어서, L은 직접결합; 치환 또는 비치환된 페닐렌기; 치환 또는 비치환된 비페닐렌기; 치환 또는 비치환된 터페닐렌기; 치환 또는 비치환된 나프틸렌기; 치환 또는 비치환된 플루오레닐렌기; 치환 또는 비치환된 2가의 카바졸기; 치환 또는 비치환된 2가의 디벤조퓨란기; 치환 또는 비치환된 2가의 디벤조티오펜기; 치환 또는 비치환된 2가의 피리딘기; 치환 또는 비치환된 2가의 피리미딘기; 또는 치환 또는 비치환된 2가의 트리아진기이다. 또 하나의 일 실시상태에 있어서, 상기 '치환 또는 비치환된'은 탄소수 1 내지 5의 알킬기; 탄소수 6 내지 20의 아릴기; 또는 탄소수 2 내지 20의 헤테로아릴기로 치환되거나 비치환되는 것을 말한다.In one embodiment of the present specification, L is a direct bond; A substituted or unsubstituted phenylene group; A substituted or unsubstituted biphenylene group; A substituted or unsubstituted terphenylene group; A substituted or unsubstituted naphthylene group; A substituted or unsubstituted fluorenylene group; A substituted or unsubstituted divalent carbazole group; A substituted or unsubstituted divalent dibenzofuran group; A substituted or unsubstituted divalent dibenzothiophene group; A substituted or unsubstituted divalent pyridine group; A substituted or unsubstituted divalent pyrimidine group; Or a substituted or unsubstituted divalent triazine group. In another exemplary embodiment, the 'substituted or unsubstituted' is an alkyl group having 1 to 5 carbon atoms; An aryl group having 6 to 20 carbon atoms; Or it is substituted or unsubstituted with a heteroaryl group having 2 to 20 carbon atoms.
본 명세서의 일 실시상태에 있어서, L은 직접결합; 중수소 또는 니트릴기로 치환 또는 비치환된 탄소수 6 내지 20의 아릴렌기이다.In one embodiment of the present specification, L is a direct bond; It is an arylene group having 6 to 20 carbon atoms unsubstituted or substituted with deuterium or nitrile groups.
본 명세서의 일 실시상태에 있어서, L은 직접결합; 니트릴기로 치환 또는 비치환된 탄소수 6 내지 20의 아릴렌기이다.In one embodiment of the present specification, L is a direct bond; It is an arylene group having 6 to 20 carbon atoms unsubstituted or substituted with a nitrile group.
본 명세서의 일 실시상태에 있어서, L은 직접결합; 니트릴기로 치환 또는 비치환된 페닐렌기; 비페닐렌기; 또는 나프틸렌기이다.In one embodiment of the present specification, L is a direct bond; A phenylene group unsubstituted or substituted with a nitrile group; Biphenylene group; Or a naphthylene group.
본 명세서의 일 실시상태에 있어서, L은 직접결합; 페닐렌기; 비페닐렌기; 또는 나프틸렌기이다.In one embodiment of the present specification, L is a direct bond; Phenylene group; Biphenylene group; Or a naphthylene group.
본 명세서의 일 실시상태에 있어서, L은 직접결합이다.In one embodiment of the present specification, L is a direct bond.
본 명세서의 일 실시상태에 있어서, X1 내지 X3는 서로 동일하거나 상이하고, 각각 독립적으로 N; 또는 CH이고, 2 이상은 N이다. In one embodiment of the present specification, X1 to X3 are the same as or different from each other, and each independently N; Or CH, and 2 or more are N.
본 명세서의 일 실시상태에 있어서, X1 및 X2는 N이고, X3는 CH이다.In one embodiment of the present specification, X1 and X2 are N, and X3 is CH.
본 명세서의 일 실시상태에 있어서, X1 및 X3는 N이고, X2는 CH이다.In one embodiment of the present specification, X1 and X3 are N, and X2 is CH.
본 명세서의 일 실시상태에 있어서, X2 및 X3는 N이고, X1는 CH이다.In one embodiment of the present specification, X2 and X3 are N, and X1 is CH.
본 명세서의 일 실시상태에 있어서, X1 내지 X3는 N이다. In one embodiment of the present specification, X1 to X3 are N.
본 명세서의 일 실시상태에 있어서, Ar1 및 Ar2는 서로 동일하거나 상이하고, 각각 독립적으로 치환 또는 비치환된 아릴기; 또는 치환 또는 비치환된 헤테로아릴기이다.In one embodiment of the present specification, Ar1 and Ar2 are the same as or different from each other, and each independently a substituted or unsubstituted aryl group; Or a substituted or unsubstituted heteroaryl group.
본 명세서의 일 실시상태에 있어서, Ar1 및 Ar2는 서로 동일하거나 상이하고, 각각 독립적으로 치환 또는 비치환된 탄소수 6 내지 30의 아릴기; 또는 치환 또는 비치환된 탄소수 2 내지 30의 헤테로아릴기이다.In one embodiment of the present specification, Ar1 and Ar2 are the same as or different from each other, and each independently a substituted or unsubstituted aryl group having 6 to 30 carbon atoms; Or a substituted or unsubstituted heteroaryl group having 2 to 30 carbon atoms.
본 명세서의 일 실시상태에 있어서, Ar1 및 Ar2는 서로 동일하거나 상이하고, 각각 독립적으로 치환 또는 비치환된 단환 내지 3환의 아릴기; 또는 치환 또는 비치환된 단환 내지 3환의 헤테로아릴기이다.In one embodiment of the present specification, Ar1 and Ar2 are the same or different from each other, and each independently a substituted or unsubstituted monocyclic to tricyclic aryl group; Or a substituted or unsubstituted monocyclic to tricyclic heteroaryl group.
본 명세서의 일 실시상태에 있어서, Ar1 및 Ar2는 서로 동일하거나 상이하고, 각각 독립적으로 탄소수 6 내지 20의 아릴기; 또는 탄소수 2 내지 20의 헤테로아릴기이다. 상기 아릴기 또는 헤테로아릴기는 중수소, 탄소수 1 내지 10의 알킬기, 탄소수 6 내지 20의 아릴기 또는 탄소수 2 내지 20의 헤테로아릴기로 치환되거나 비치환된다.In one embodiment of the present specification, Ar1 and Ar2 are the same as or different from each other, and each independently an aryl group having 6 to 20 carbon atoms; Or a heteroaryl group having 2 to 20 carbon atoms. The aryl group or heteroaryl group is substituted or unsubstituted with deuterium, an alkyl group having 1 to 10 carbon atoms, an aryl group having 6 to 20 carbon atoms, or a heteroaryl group having 2 to 20 carbon atoms.
본 명세서의 일 실시상태에 있어서, Ar1 및 Ar2는 서로 동일하거나 상이하고, 각각 독립적으로 치환 또는 비치환된 페닐기; 치환 또는 비치환된 비페닐기; 치환 또는 비치환된 터페닐기; 치환 또는 비치환된 나프틸기; 치환 또는 비치환된 플루오레닐기; 치환 또는 비치환된 카바졸기; 치환 또는 비치환된 디벤조퓨란기; 치환 또는 비치환된 디벤조티오펜기; 치환 또는 비치환된 피리딘기; 치환 또는 비치환된 피리미딘기; 또는 치환 또는 비치환된 트리아진기이다. 또 하나의 일 실시상태에 있어서, 상기 '치환 또는 비치환된'은 탄소수 1 내지 5의 알킬기; 탄소수 6 내지 20의 아릴기; 또는 탄소수 2 내지 20의 헤테로아릴기로 치환되거나 비치환되는 것을 말한다.In one embodiment of the present specification, Ar1 and Ar2 are the same as or different from each other, and each independently a substituted or unsubstituted phenyl group; A substituted or unsubstituted biphenyl group; A substituted or unsubstituted terphenyl group; A substituted or unsubstituted naphthyl group; A substituted or unsubstituted fluorenyl group; A substituted or unsubstituted carbazole group; A substituted or unsubstituted dibenzofuran group; A substituted or unsubstituted dibenzothiophene group; A substituted or unsubstituted pyridine group; A substituted or unsubstituted pyrimidine group; Or a substituted or unsubstituted triazine group. In another exemplary embodiment, the 'substituted or unsubstituted' is an alkyl group having 1 to 5 carbon atoms; An aryl group having 6 to 20 carbon atoms; Or it is substituted or unsubstituted with a heteroaryl group having 2 to 20 carbon atoms.
본 명세서의 일 실시상태에 있어서, Ar1 및 Ar2는 서로 동일하거나 상이하고, 각각 독립적으로 중수소 또는 메틸기로 치환 또는 비치환된 탄소수 6 내지 20의 아릴기; 또는 탄소수 6 내지 20의 아릴기로 치환 또는 비치환된 탄소수 2 내지 20의 헤테로아릴기이다. In one embodiment of the present specification, Ar1 and Ar2 are the same as or different from each other, and each independently an aryl group having 6 to 20 carbon atoms unsubstituted or substituted with deuterium or a methyl group; Or a heteroaryl group having 2 to 20 carbon atoms unsubstituted or substituted with an aryl group having 6 to 20 carbon atoms.
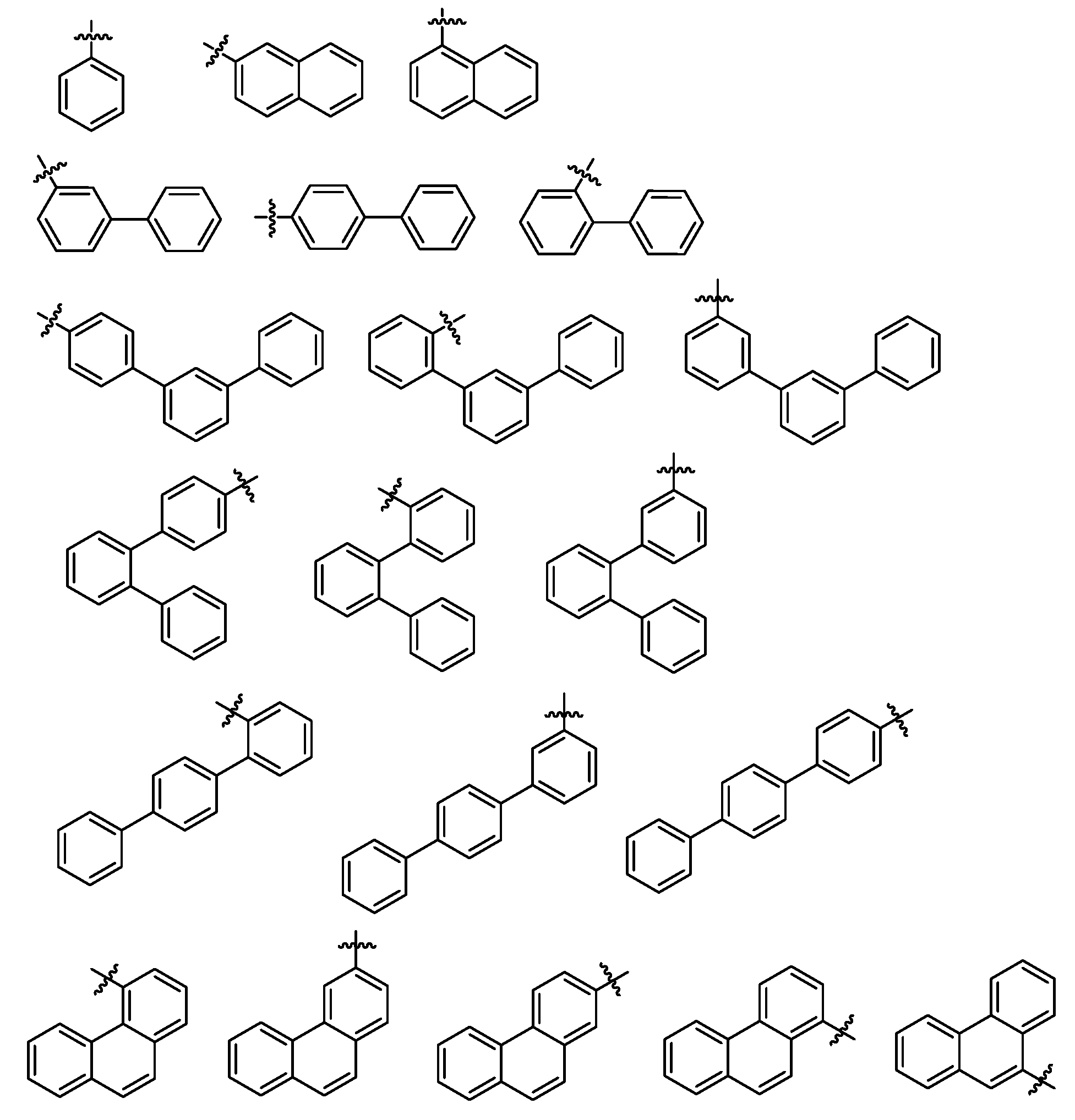
본 명세서의 일 실시상태에 있어서, Ar1 및 Ar2는 서로 동일하거나 상이하고, 각각 독립적으로 페닐기; 비페닐기; 터페닐기; 나프틸기; 페난트레닐기; 디메틸플루오레닐기; 디벤조퓨란기; 디벤조티오펜기; N-페닐카바졸기; 또는 카바졸기이고, 상기 Ar1 및 Ar2는 중수소로 치환되거나 비치환된다.In one embodiment of the present specification, Ar1 and Ar2 are the same as or different from each other, and each independently a phenyl group; Biphenyl group; Terphenyl group; Naphthyl group; Phenanthrenyl group; Dimethylfluorenyl group; Dibenzofuran group; Dibenzothiophene group; N-phenylcarbazole group; Or a carbazole group, wherein Ar1 and Ar2 are substituted with deuterium or unsubstituted.
본 명세서의 일 실시상태에 있어서, Ar1 및 Ar2는 서로 동일하거나 상이하고, 각각 독립적으로 페닐기; 비페닐기; 터페닐기; 나프틸기; 페난트레닐기; 디벤조퓨란기; 디벤조티오펜기; N-페닐카바졸기; 또는 카바졸기이고, 상기 Ar1 및 Ar2는 중수소로 치환되거나 비치환된다.In one embodiment of the present specification, Ar1 and Ar2 are the same as or different from each other, and each independently a phenyl group; Biphenyl group; Terphenyl group; Naphthyl group; Phenanthrenyl group; Dibenzofuran group; Dibenzothiophene group; N-phenylcarbazole group; Or a carbazole group, wherein Ar1 and Ar2 are substituted with deuterium or unsubstituted.
본 명세서의 일 실시상태에 있어서, Ar1 및 Ar2는 서로 동일하거나 상이하고, 각각 독립적으로 하기 구조에서 선택된 하나이다.In one embodiment of the present specification, Ar1 and Ar2 are the same as or different from each other, and each independently selected from the following structures.
상기 구조는 중수소로 치환 또는 비치환된다,The structure is substituted or unsubstituted with deuterium,
본 명세서의 일 실시상태에 있어서, Ar1 및 Ar2는 서로 상이하다.In one embodiment of the present specification, Ar1 and Ar2 are different from each other.
본 명세서의 일 실시상태에 있어서, R1 및 R2는 서로 동일하거나 상이하고, 각각 독립적으로 수소; 또는 중수소이다.In one embodiment of the present specification, R1 and R2 are the same as or different from each other, and each independently hydrogen; Or deuterium.
본 명세서의 일 실시상태에 있어서, R1 및 R2는 수소이다.In one embodiment of the present specification, R1 and R2 are hydrogen.
본 명세서의 일 실시상태에 있어서, R1 및 R2는 중수소이다.In one embodiment of the present specification, R1 and R2 are deuterium.
본 명세서의 일 실시상태에 있어서, a1은 0 내지 8의 정수이고, a1이 2 이상인 경우, R1은 서로 동일하거나 상이하다.In one embodiment of the present specification, a1 is an integer from 0 to 8, and when a1 is 2 or more, R1 is the same or different from each other.
본 명세서의 일 실시상태에 있어서, a2은 0 내지 2의 정수이고, a2이 2인 경우, R2은 서로 동일하거나 상이하다.In one embodiment of the present specification, a2 is an integer from 0 to 2, and when a2 is 2, R2 is the same or different from each other.
본 명세서의 일 실시상태에 있어서, a3은 0 내지 6의 정수이고, a3이 2 이상인 경우, R3은 서로 동일하거나 상이하다.In one embodiment of the present specification, a3 is an integer from 0 to 6, and when a3 is 2 or more, R3 is the same or different from each other.
본 명세서의 일 실시상태에 있어서, a1 내지 a3은 0이다.In one embodiment of the present specification, a1 to a3 are 0.
본 명세서의 일 실시상태에 있어서, a1은 8이다.In one embodiment of the present specification, a1 is 8.
본 명세서의 일 실시상태에 있어서, a2는 2이다,In one embodiment of the present specification, a2 is 2,
본 명세서의 일 실시상태에 있어서, a3은 6이다.In one embodiment of the present specification, a3 is 6.
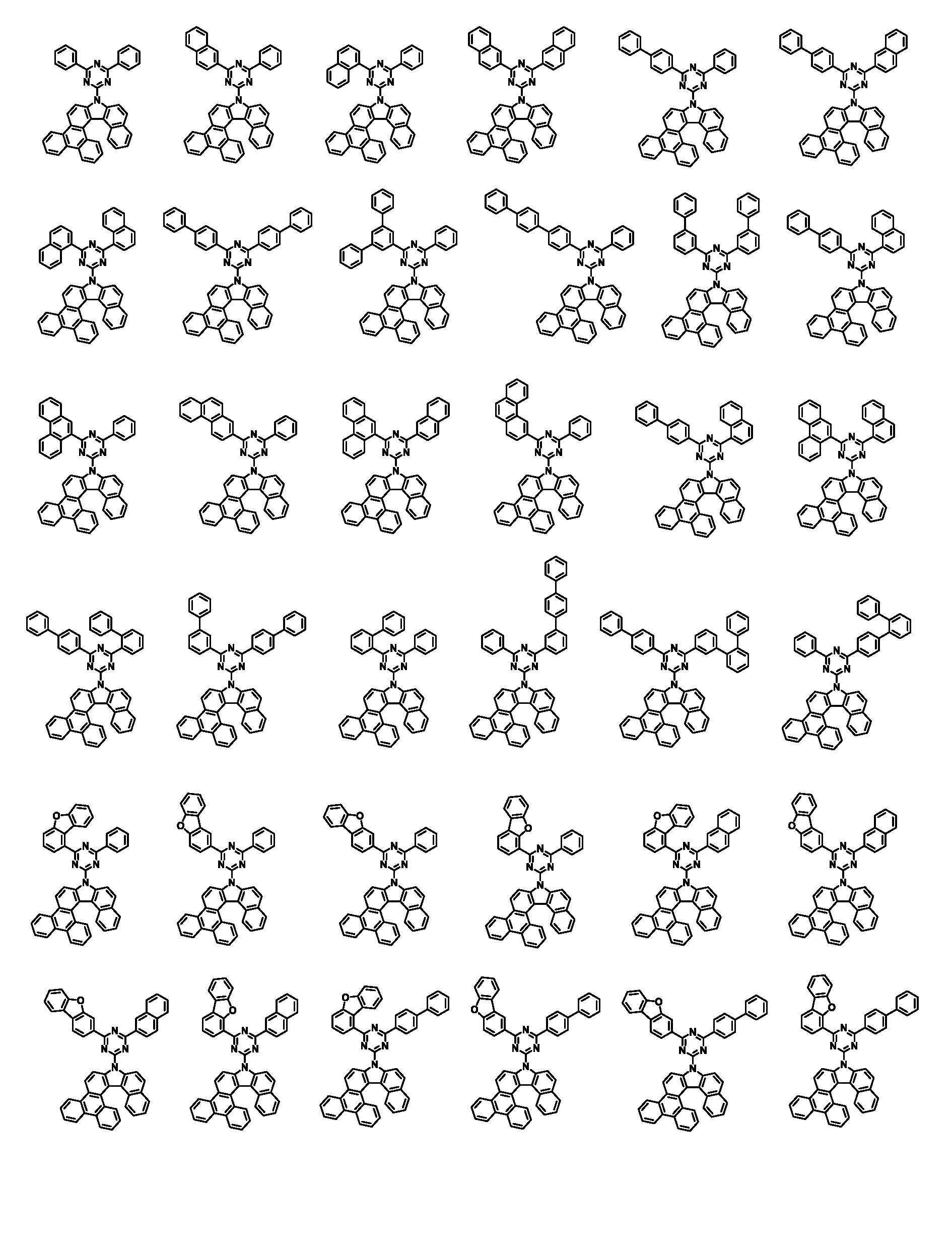
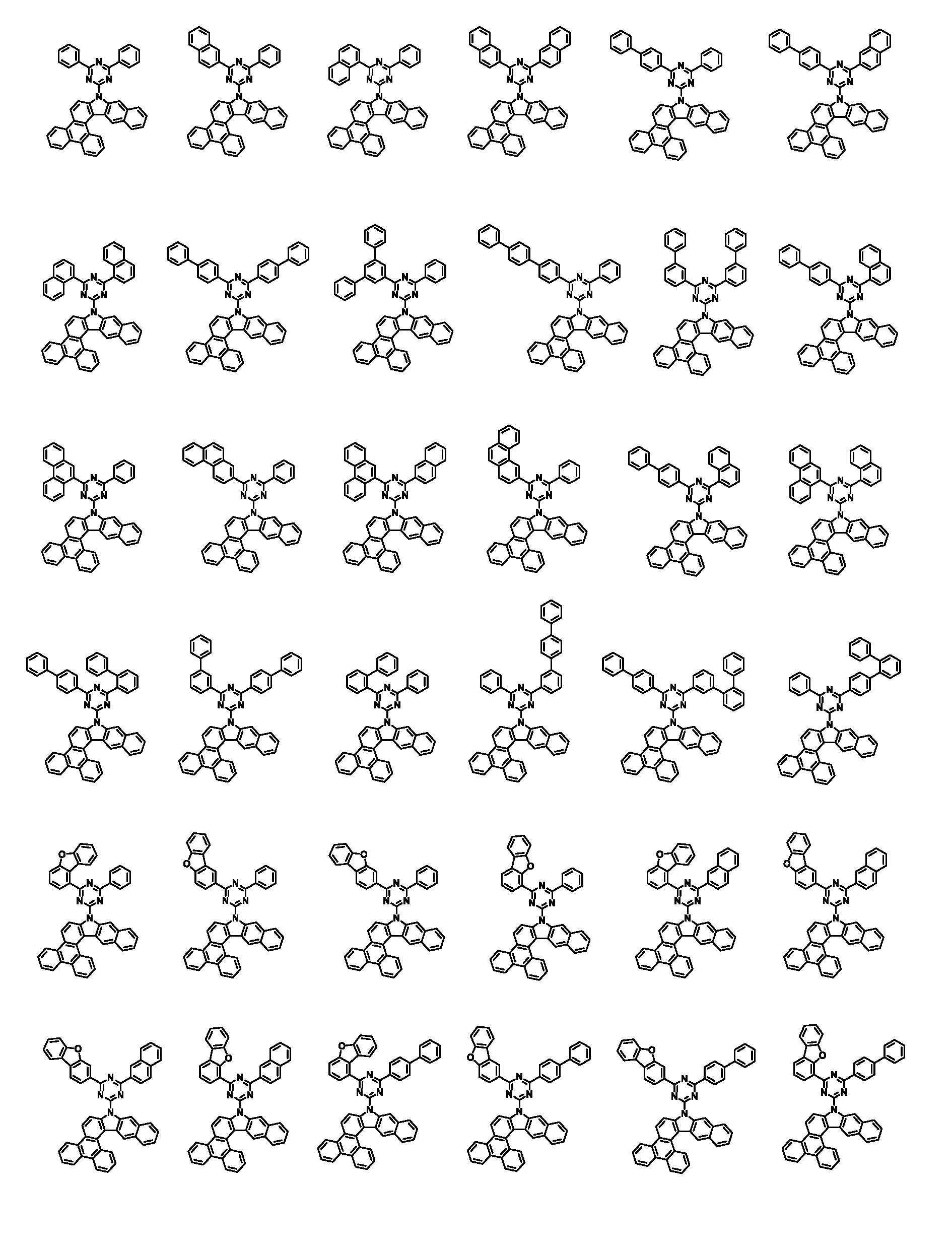
본 명세서의 일 실시상태에 있어서, 상기 화학식 1은 하기 화학식 3-1 또는 3-2로 표시된다.In one embodiment of the present specification, Chemical Formula 1 is represented by the following Chemical Formula 3-1 or 3-2.
[화학식 3-1][Formula 3-1]
[화학식 3-2][Formula 3-2]
상기 화학식 3-1 및 3-2에 있어서, In Chemical Formulas 3-1 and 3-2,
X1 내지 X3, Ar1, Ar2, L, R1 내지 R3 및 a1 내지 a3의 정의는 화학식 1에서 정의한 바와 같다.X1 to X3, Ar1, Ar2, L, R1 to R3 and a1 to a3 are defined as defined in Chemical Formula 1.
본 명세서의 일 실시상태에 있어서, 상기 화학식 1은 하기 화학식 2-1 내지 2-6 중 어느 하나로 표시된다.In one embodiment of the present specification, Chemical Formula 1 is represented by any one of the following Chemical Formulas 2-1 to 2-6.
[화학식 2-1] [Formula 2-1]
[화학식 2-2][Formula 2-2]
[화학식 2-3][Formula 2-3]
[화학식 2-4][Formula 2-4]
[화학식 2-5][Formula 2-5]
[화학식 2-6][Formula 2-6]
상기 화학식 2-1 내지 2-6에 있어서,In Chemical Formulas 2-1 to 2-6,
X1 내지 X3, Ar1, Ar2, L, R1 내지 R3 및 a1 내지 a3의 정의는 화학식 1에서 정의한 바와 같다.X1 to X3, Ar1, Ar2, L, R1 to R3 and a1 to a3 are defined as defined in Chemical Formula 1.
본 명세서의 일 실시상태에 있어서, 상기 화학식 3-1은 상기 화학식 2-1 내지 2-3 중 어느 하나로 표시된다.In one embodiment of the present specification, Chemical Formula 3-1 is represented by any one of Chemical Formulas 2-1 to 2-3.
본 명세서의 일 실시상태에 있어서, 상기 화학식 3-2는 상기 화학식 2-4 내지 2-6 중 어느 하나로 표시된다.In one embodiment of the present specification, Chemical Formula 3-2 is represented by any one of Chemical Formulas 2-4 to 2-6.
본 명세서의 일 실시상태에 있어서, 상기 화학식 1로 표시되는 화합물은 하기 화합물 중에서 선택되는 어느 하나이다.In one embodiment of the present specification, the compound represented by Chemical Formula 1 is any one selected from the following compounds.
본 명세서의 일 실시상태에 따른 화합물은 후술하는 제조 방법으로 제조될 수 있다. 후술하는 제조예들에서는 대표적인 예시들을 기재하지만, 필요에 따라, 치환기를 추가하거나 제외할 수 있으며, 치환기의 위치를 변경할 수 있다. 또한, 당기술분야에 알려져 있는 기술을 기초로, 출발물질, 반응물질, 반응 조건 등을 변경할 수 있다. The compound according to an exemplary embodiment of the present specification may be prepared by a manufacturing method described later. Representative examples are described in the manufacturing examples described below, but if necessary, a substituent may be added or excluded, and the position of the substituent may be changed. In addition, based on techniques known in the art, it is possible to change starting materials, reactants, reaction conditions, and the like.
예컨대, 상기 화학식 1로 표시되는 화합물은 하기 일반식 1 또는 2와 같이 코어 구조가 제조될 수 있다. 치환기는 당기술분야에 알려져 있는 방법에 의하여 결합될 수 있으며, 치환기의 종류, 위치 또는 개수는 당기술분야에 알려져 있는 기술에 따라 변경될 수 있다. 하기 일반식 1 또는 2와 같이 치환기를 결합시킬 수 있으나, 이에 한정되는 것은 아니다.For example, the compound represented by Chemical Formula 1 may have a core structure as shown in General Formula 1 or 2 below. Substituents can be combined by methods known in the art, and the type, location, or number of substituents can be changed according to techniques known in the art. Substituents may be bonded as in the following general formula 1 or 2, but are not limited thereto.
[일반식 1][Formula 1]
[일반식 2] [Formula 2]
상기와 같은 일반식 1 및 2에 의하여 코어구조를 제조한 뒤, 본 발명의 화합물은 Buchwald-Hartwig coupling reaction, Suzuki coupling reaction, Heck coupling reaction 등을 이용하여 치환기를 연결시킬 수 있다.After preparing the core structure according to the general formulas 1 and 2 as described above, the compounds of the present invention can be connected to a substituent using a Buchwald-Hartwig coupling reaction, Suzuki coupling reaction, Heck coupling reaction and the like.
또한, 본 명세서는 상기 전술한 화합물을 포함하는 유기 발광 소자를 제공한다.In addition, the present specification provides an organic light emitting device comprising the above-described compound.
본 명세서의 일 실시상태에 있어서, 제1 전극; 상기 제1 전극과 대향하여 구비된 제2 전극; 및 상기 제1 전극과 상기 제2 전극 사이에 구비된 1층 이상의 유기물층을 포함하는 유기 발광 소자로서, 상기 유기물층 중 1 층 이상은 상기 화합물을 포함하는 것인 유기 발광 소자를 제공한다. In one embodiment of the present specification, the first electrode; A second electrode provided to face the first electrode; And an organic light emitting device including one or more organic material layers provided between the first electrode and the second electrode, wherein at least one layer of the organic material layer includes the compound.
본 명세서에서 어떤 부재가 다른 부재 "상에" 위치하고 있다고 할 때, 이는 어떤 부재가 다른 부재에 접해 있는 경우뿐 아니라 두 부재 사이에 또 다른 부재가 존재하는 경우도 포함한다.When a member is referred to herein as being “on” another member, this includes not only the case where one member abuts another member, but also the case where another member exists between the two members.
본 명세서에서 어떤 부분이 어떤 구성요소를 "포함" 한다고 할 때, 이는 특별히 반대되는 기재가 없는 한 다른 구성요소를 제외하는 것이 아니라 다른 구성 요소를 더 포함할 수 있는 것을 의미한다. In the present specification, when a part “includes” a certain component, it means that the component may further include other components, rather than excluding other components, unless otherwise specified.
본 명세서의 유기 발광 소자의 유기물층은 단층 구조로 이루어질 수도 있으나, 2층 이상의 유기물층이 적층된 다층 구조로 이루어질 수 있다. 예컨대, 정공 주입층, 정공 수송층, 발광층, 전자 수송층, 전자 주입층, 전자 차단층, 정공 차단층 등을 포함하는 구조를 가질 수 있다. 그러나 유기 발광 소자의 구조는 이에 한정되지 않고 더 적은 수의 유기층을 포함할 수 있다.The organic material layer of the organic light emitting device of the present specification may have a single layer structure, but may have a multi-layer structure in which two or more organic material layers are stacked. For example, it may have a structure including a hole injection layer, a hole transport layer, a light emitting layer, an electron transport layer, an electron injection layer, an electron blocking layer, a hole blocking layer, and the like. However, the structure of the organic light emitting device is not limited to this, and may include fewer organic layers.
본 명세서의 일 실시상태에 있어서, 상기 유기물층은 발광층을 포함하고, 상기 발광층은 상기 화학식 1로 표시되는 화합물을 포함한다. In one embodiment of the present specification, the organic material layer includes a light emitting layer, and the light emitting layer includes a compound represented by Chemical Formula 1.
본 명세서의 일 실시상태에 있어서, 상기 유기물층은 발광층을 포함하고, 상기 발광층은 상기 화학식 1로 표시되는 화합물을 포함하고, 상기 화학식 1로 표시되는 화합물을 포함한 발광층은 적색을 띤다.In one embodiment of the present specification, the organic material layer includes a light emitting layer, the light emitting layer includes a compound represented by Chemical Formula 1, and a light emitting layer including a compound represented by Chemical Formula 1 is red.
본 명세서의 일 실시상태에 따르면, 상기 유기물층은 발광층을 포함하고, 상기 발광층은 상기 화학식 1로 표시되는 화합물을 발광층의 호스트로서 포함한다.According to the exemplary embodiment of the present specification, the organic material layer includes a light emitting layer, and the light emitting layer includes a compound represented by Chemical Formula 1 as a host of the light emitting layer.
본 명세서의 일 실시상태에 따르면, 상기 유기물층은 발광층을 포함하고, 상기 발광층은 상기 화학식 1로 표시되는 화합물; 및 도펀트를 포함한다.According to an exemplary embodiment of the present specification, the organic material layer includes a light emitting layer, the light emitting layer is a compound represented by the formula (1); And dopants.
본 명세서의 일 실시상태에 따르면, 상기 도펀트의 함량은 상기 화학식 1로 표시되는 화합물의 100 중량부 대비 0.01 중량부 내지 20 중량부, 더욱 바람직하게는 0.1 중량부 내지 10 중량부이다. 상기 범위인 경우, 호스트에서 도펀트로의 에너지 전달이 잘 일어난다.According to an exemplary embodiment of the present specification, the content of the dopant is 0.01 parts by weight to 20 parts by weight, more preferably 0.1 parts by weight to 10 parts by weight based on 100 parts by weight of the compound represented by Chemical Formula 1 above. In the above range, energy transfer from the host to the dopant occurs well.
본 명세서의 일 실시상태에 따르면, 상기 도펀트의 발광 파장은 한정되지 않으며, 상기 도펀트는 인광 도판트 또는 형광 도펀트일 수 있다.According to an exemplary embodiment of the present specification, the emission wavelength of the dopant is not limited, and the dopant may be a phosphorescent dopant or a fluorescent dopant.
본 명세서의 일 실시상태에 따르면, 상기 도펀트는 이리듐계 착제일 수 있다.According to an exemplary embodiment of the present specification, the dopant may be an iridium-based complex.
본 명세서의 일 실시상태에 따르면, 상기 도펀트는 하기 구조들 중에서 선택된 1종 또는 2종 이상일 수 있으나, 이에 한정되지 않는다.According to an exemplary embodiment of the present specification, the dopant may be one or two or more selected from the following structures, but is not limited thereto.
본 명세서의 일 실시상태에 있어서, 상기 유기물층은 2층 이상의 발광층을 포함하고, 상기 2층 이상의 발광층 중 적어도 하나는 상기 화학식 1로 표시되는 화합물을 포함한다. 상기 화학식 1로 표시되는 화합물을 포함한 발광층은 적색을 띠며, 상기 화학식 1로 표시되는 화합물을 포함하지 않은 발광층은 당업계에 알려진 청색, 적색 또는 녹색 발광 화합물을 포함할 수 있다.In one embodiment of the present specification, the organic material layer includes two or more light-emitting layers, and at least one of the two or more light-emitting layers includes a compound represented by Chemical Formula 1. The light emitting layer including the compound represented by Chemical Formula 1 has a red color, and the light emitting layer not containing the compound represented by Chemical Formula 1 may include a blue, red or green light emitting compound known in the art.
본 명세서의 일 실시상태에 있어서, 상기 유기물층은 정공 주입층 또는 정공 수송층을 포함하고, 상기 정공 주입층 또는 정공 수송층은 상기 화학식 1로 표시되는 화합물을 포함한다. In one embodiment of the present specification, the organic material layer includes a hole injection layer or a hole transport layer, and the hole injection layer or the hole transport layer includes a compound represented by Chemical Formula 1.
본 명세서의 일 실시상태에 있어서, 상기 유기물층은 전자 주입층 또는 전자 수송층을 포함하고, 상기 전자 주입층 또는 전자 수송층은 상기 화학식 1로 표시되는 화합물을 포함한다. In one embodiment of the present specification, the organic material layer includes an electron injection layer or an electron transport layer, and the electron injection layer or electron transport layer includes a compound represented by Chemical Formula 1.
본 명세서의 일 실시상태에 있어서, 상기 유기물층은 전자 차단층을 포함하고, 상기 전자 차단층은 상기 화학식 1로 표시되는 화합물을 포함한다. In one embodiment of the present specification, the organic material layer includes an electron blocking layer, and the electron blocking layer includes a compound represented by Chemical Formula 1.
본 명세서의 일 실시상태에 있어서, 상기 유기물층은 정공 차단층을 포함하고, 상기 정공 차단층은 상기 화학식 1로 표시되는 화합물을 포함한다. In one embodiment of the present specification, the organic material layer includes a hole blocking layer, and the hole blocking layer includes a compound represented by Chemical Formula 1.
본 명세서의 일 실시상태에 있어서, 상기 유기 발광 소자는 정공 주입층, 정공 수송층. 발광층, 전자 수송층, 전자 주입층, 정공 차단층 및 전자 차단층으로 이루어진 군에서 선택되는 1층 또는 2층 이상을 더 포함한다.In one embodiment of the present specification, the organic light emitting device is a hole injection layer, a hole transport layer. It further includes at least one layer or two or more layers selected from the group consisting of a light emitting layer, an electron transport layer, an electron injection layer, a hole blocking layer, and an electron blocking layer.
본 명세서의 일 실시상태에 있어서, 상기 유기 발광 소자는 제1 전극; 상기 제1 전극과 대향하여 구비된 제2 전극; 상기 제1 전극과 상기 제2 전극 사이에 구비된 발광층; 및 상기 발광층과 상기 제1 전극 사이, 또는 상기 발광층과 상기 제2 전극 사이에 구비된 2층 이상의 유기물층을 포함하고, 상기 2층 이상의 유기물층 중 적어도 하나는 상기 화학식 1로 표시되는 화합물을 포함한다. In one embodiment of the present specification, the organic light emitting device includes a first electrode; A second electrode provided to face the first electrode; A light emitting layer provided between the first electrode and the second electrode; And two or more organic material layers provided between the light emitting layer and the first electrode, or between the light emitting layer and the second electrode, and at least one of the two or more organic material layers includes a compound represented by Chemical Formula 1.
본 명세서의 일 실시상태에 있어서, 상기 2층 이상의 유기물층은 발광층, 정공 수송층, 정공 주입층, 정공 수송과 정공 주입을 동시에 하는 층 및 전자 차단층으로 이루어진 군에서 2 이상이 선택될 수 있다.In one embodiment of the present specification, two or more organic material layers may be selected from the group consisting of a light emitting layer, a hole transport layer, a hole injection layer, a layer simultaneously performing hole transport and hole injection, and an electron blocking layer.
본 명세서의 일 실시상태에 있어서, 상기 유기물층은 2층 이상의 전자 수 송층을 포함하고, 상기 2층 이상의 전자 수송층 중 적어도 하나는 상기 화학식 1로 표시되는 화합물을 포함한다. 구체적으로 본 명세서의 일 실시상태에 있어서, 상기 화학식 1로 표시되는 화합물은 상기 2층 이상의 전자 수송층 중 1층에 포함될 수도 있으며, 각각의 2층 이상의 전자 수송층에 포함될 수 있다. In one embodiment of the present specification, the organic material layer includes two or more electron transport layers, and at least one of the two or more electron transport layers includes a compound represented by Chemical Formula 1. Specifically, in one embodiment of the present specification, the compound represented by Chemical Formula 1 may be included in one layer of the two or more electron transport layers, or may be included in each of the two or more electron transport layers.
또한, 본 명세서의 일 실시상태에 있어서, 상기 화학식 1로 표시되는 화합물이 상기 각각의 2층 이상의 전자 수송층에 포함되는 경우, 상기 화학식 1로 표시되는 화합물을 제외한 다른 재료들은 서로 동일하거나 상이할 수 있다.In addition, in one embodiment of the present specification, when the compound represented by Chemical Formula 1 is included in each of the two or more electron transport layers, other materials except the compound represented by Chemical Formula 1 may be the same or different from each other. have.
상기 화학식 1로 표시되는 화합물을 포함하는 유기물층이 전자 수송층인 경우, 상기 전자 수송층은 n형 도펀트를 더 포함할 수 있다. 상기 n형 도펀트는 당 기술분야에 알려져 있는 것들을 사용할 수 있으며, 예컨대 금속 또는 금속착체를 사용할 수 있다. 예를 들어, 상기 화학식 1로 표시되는 화합물을 포함하는 전자 수송층은 LiQ(Lithium Quinolate)를 더 포함할 수 있다.When the organic material layer including the compound represented by Chemical Formula 1 is an electron transport layer, the electron transport layer may further include an n-type dopant. The n-type dopant may use those known in the art, for example, a metal or a metal complex. For example, the electron transport layer including the compound represented by Chemical Formula 1 may further include LiQ (Lithium Quinolate).
본 명세서의 일 실시상태에 있어서, 상기 유기물층은 2층 이상의 정공수송층을 포함하고, 상기 2층 이상의 정공 수송층 중 적어도 하나는 상기 화학식 1로 표시되는 화합물을 포함한다. 구체적으로, 본 명세서의 일 실시상태에 있어서, 상기 화학식 1로 표시되는 화합물은 상기 2층 이상의 정공 수송층 중 1층에 포함될 수도 있으며, 각각의 2층 이상의 정공 수송층에 포함될 수 있다. In one embodiment of the present specification, the organic material layer includes two or more hole transport layers, and at least one of the two or more hole transport layers includes a compound represented by Chemical Formula 1. Specifically, in one embodiment of the present specification, the compound represented by Chemical Formula 1 may be included in one layer of the two or more hole transport layers, or may be included in each of the two or more hole transport layers.
또한, 본 명세서의 일 실시상태에 있어서, 상기 화학식 1로 표시되는 화합물이 상기 각각의 2층 이상의 정공 수송층에 포함되는 경우, 상기 화학식 1로 표시되는 화합물을 제외한 다른 재료들은 서로 동일하거나 상이할 수 있다.In addition, in one embodiment of the present specification, when the compound represented by Chemical Formula 1 is included in each of the two or more hole transport layers, other materials except the compound represented by Chemical Formula 1 may be the same or different from each other. have.
본 명세서의 일 실시상태에 있어서, 상기 유기물층은 상기 화학식 1로 표시되는 화합물을 포함하는 유기물층 이외에 아릴아민기, 카바졸릴기 또는 벤조카바졸릴기를 포함하는 화합물을 포함하는 정공 주입층 또는 정공 수송층을 더 포함할 수 있다.In one embodiment of the present specification, the organic material layer further comprises a hole injection layer or a hole transport layer containing a compound containing an arylamine group, a carbazolyl group, or a benzocarbazolyl group in addition to the organic material layer containing the compound represented by Formula 1 It can contain.
본 명세서의 일 실시상태에 있어서, 상기 제1 전극은 양극이고, 상기 제2 전극은 음극이다.In one embodiment of the present specification, the first electrode is an anode, and the second electrode is a cathode.
본 명세서의 일 실시상태에 있어서, 상기 제1 전극은 음극이고, 상기 제2 전극은 양극이다. In one embodiment of the present specification, the first electrode is a cathode, and the second electrode is an anode.
본 명세서의 일 실시상태에 있어서, 상기 유기 발광 소자는 기판 상에 양극, 1층 이상의 유기물층 및 음극이 순차적으로 적층된 구조(normal type)의 유기 발광 소자일 수 있다. In one embodiment of the present specification, the organic light emitting device may be an organic light emitting device having a structure in which an anode, one or more organic material layers, and a cathode are sequentially stacked on a substrate.
본 명세서의 일 실시상태에 있어서, 상기 유기 발광 소자는 기판 상에 양극, 1층 이상의 유기물층 및 음극이 순차적으로 적층된 역방향 구조(inverted type)의 유기 발광 소자일 수 있다.In one embodiment of the present specification, the organic light emitting device may be an inverted type organic light emitting device in which an anode, one or more organic material layers, and a cathode are sequentially stacked on a substrate.
예컨대, 본 명세서의 일 실시상태에 따른 유기 발광 소자의 구조가 도 1 내지 3에 예시되어 있다. 상기 도 1 내지 도 3은 유기 발광 소자를 예시한 것이며 이에 한정되는 것은 아니다. For example, the structure of the organic light emitting device according to the exemplary embodiment of the present specification is illustrated in FIGS. 1 to 3. 1 to 3 illustrate an organic light emitting device and are not limited thereto.
도 1에는 기판(101) 위에 양극(102), 발광층(106) 및 음극(110)이 순차적으로 적층된 유기 발광 소자의 구조가 예시되어 있다. 상기 화학식 1로 표시되는 화합물은 발광층에 포함된다.1, a structure of an organic light emitting device in which an
도 2에는 기판(101) 위에 양극(102), 정공 주입층(103), 정공 수송층(104), 발광층(106) 및 음극(110)이 순차적으로 적층된 유기 발광 소자의 구조가 예시되어 있다. 본 발명의 일 실시상태에 따르면, 상기 화학식 1로 표시되는 화합물은 상기 유기물층 중 1층 이상에 포함된다. 또 하나의 실시상태에 따르면, 상기 화학식 1로 표시되는 화합물은 발광층에 포함될 수 있으나, 이에 한정되는 것은 아니다.2 illustrates a structure of an organic light emitting device in which an
도 3에는 기판(101) 위에 양극(102), 정공 주입층(103), 정공 수송층(104), 전자 차단층(105), 발광층(106), 정공 차단층(107), 전자 주입 및 수송층(108) 및 음극(110)이 순차적으로 적층된 유기 발광 소자의 구조가 예시되어 있다. 본 발명의 일 실시상태에 따르면, 상기 화학식 1로 표시되는 화합물은 상기 유기물층 중 1층 이상에 포함된다. 또 하나의 실시상태에 따르면, 상기 화학식 1로 표시되는 화합물은 발광층에 포함될 수 있으나, 이에 한정되는 것은 아니다. 3, the
본 명세서의 유기 발광 소자는 유기물층 중 1층 이상이 상기 화합물, 즉 상기 화학식 1로 표시되는 화합물을 포함하는 것을 제외하고는 당 기술분야에 알려져 있는 재료와 방법으로 제조될 수 있다. The organic light emitting device of the present specification may be made of materials and methods known in the art, except that at least one layer of the organic material layer includes the compound, that is, the compound represented by Formula 1.
상기 유기 발광 소자가 복수개의 유기물층을 포함하는 경우, 상기 유기물층은 동일한 물질 또는 다른 물질로 형성될 수 있다. When the organic light emitting device includes a plurality of organic material layers, the organic material layers may be formed of the same material or different materials.
예컨대, 본 명세서의 유기 발광 소자는 기판 상에 제1 전극, 유기물층 및 제2 전극을 순차적으로 적층시킴으로써 제조할 수 있다. 이 때, 스퍼터링법(sputtering)이나 전자빔 증발법(e-beam evaporation)과 같은 PVD(physical Vapor Deposition)방법을 이용하여, 기판 상에 금속 또는 전도성을 가지는 금속 산화물 또는 이들의 합금을 증착시켜 양극을 형성하고, 그 위에 정공 주입층, 정공 수송층, 발광층 및 전자 수송층을 포함하는 유기물층을 형성한 후, 그 위에 음극로 사용할 수 있는 물질을 증착시킴으로써 제조될 수 있다. 이와 같은 방법 외에도, 기판 상에 음극 물질, 유기물층 및 양극 물질을 차례로 증착시켜 유기 발광 소자를 제조할 수 있다. For example, the organic light emitting device of the present specification can be manufactured by sequentially laminating a first electrode, an organic material layer, and a second electrode on a substrate. At this time, a positive electrode is deposited by depositing a metal or conductive metal oxide or an alloy thereof on a substrate using a physical vapor deposition (PVD) method such as sputtering or e-beam evaporation. It can be produced by forming and forming an organic material layer including a hole injection layer, a hole transport layer, a light emitting layer, and an electron transport layer thereon, and then depositing a material that can be used as a cathode thereon. In addition to this method, an organic light emitting device may be manufactured by sequentially depositing a cathode material, an organic material layer, and an anode material on a substrate.
또한, 상기 화학식 1로 표시되는 화합물은 유기 발광 소자의 제조시 진공 증착법 뿐만 아니라 용액 도포법에 의하여 유기물층으로 형성될 수 있다. 여기서, 용액 도포법이라 함은 스핀 코팅, 딥코팅, 닥터 블레이딩, 잉크젯프린팅, 스크린 프린팅, 스프레이법, 롤 코팅 등을 의미하나, 이에 한정되는 것은 아니다. In addition, the compound represented by Chemical Formula 1 may be formed as an organic material layer by a solution coating method as well as a vacuum deposition method when manufacturing an organic light emitting device. Here, the solution application method means spin coating, dip coating, doctor blading, inkjet printing, screen printing, spraying, roll coating, and the like, but is not limited thereto.
이와 같은 방법 외에도, 기판 상에 음극 물질로부터 유기물층, 양극 물질을 차례로 증착시켜 유기 발광 소자를 만들 수도 있다 (국제 특허 출원 공개 제 2003/012890호). 다만, 제조 방법이 이에 한정되는 것은 아니다. In addition to this method, an organic light emitting device may also be formed by sequentially depositing an organic material layer and a cathode material from a cathode material on a substrate (International Patent Application Publication No. 2003/012890). However, the manufacturing method is not limited thereto.
상기 양극 물질로는 통상 유기물층으로 정공 주입이 원활할 수 있도록 일함수가 큰 물질이 바람직하다. 예를 들어, 바나듐, 크롬, 구리, 아연, 금과 같은 금속 또는 이들의 합금; 아연 산화물, 인듐 산화물, 인듐주석 산화물(ITO), 인듐아연 산화물(IZO)과 같은 금속 산화물; ZnO:Al 또는 SnO2 : Sb와 같은 금속과 산화물의 조합; 폴리(3-메틸티오펜), 폴리[3,4-(에틸렌-1,2-디옥시)티오펜](PEDOT), 폴리피롤 및 폴리아닐린과 같은 전도성 고분자 등이 있으나, 이에 한정되는 것은 아니다. The positive electrode material is usually a material having a large work function to facilitate hole injection into the organic material layer. Metals such as vanadium, chromium, copper, zinc, gold or alloys thereof; Metal oxides such as zinc oxide, indium oxide, indium tin oxide (ITO), and indium zinc oxide (IZO); ZnO: Al or SnO 2 : Combination of metal and oxide such as Sb; Conductive polymers such as poly (3-methylthiophene), poly [3,4- (ethylene-1,2-dioxy) thiophene] (PEDOT), polypyrrole, and polyaniline, but are not limited thereto.
상기 음극 물질로는 통상 유기물층으로 전자 주입이 용이하도록 일함수가 작은 물질인 것이 바람직하다. 예를 들어, 마그네슘, 칼슘, 나트륨, 칼륨, 티타늄, 인듐, 이트륨, 리튬, 가돌리늄, 알루미늄, 은, 주석 및 납과 같은 금속 또는 이들의 합금; LiF/Al 또는 LiO2/Al과 같은 다층 구조 물질 등이 있으나, 이에 한정되는 것은 아니다.The cathode material is preferably a material having a small work function to facilitate electron injection into the organic material layer. Metals such as magnesium, calcium, sodium, potassium, titanium, indium, yttrium, lithium, gadolinium, aluminum, silver, tin and lead or alloys thereof; There is a multilayer structure material such as LiF / Al or LiO 2 / Al, but is not limited thereto.
상기 발광층은 호스트 재료 및 도펀트 재료를 포함할 수 있다. 호스트 재료는 축합 방향족환 유도체 또는 헤테로환 함유 화합물 등이 있다. 구체적으로, 축합 방향족환 유도체로는 안트라센 유도체, 피렌 유도체, 나프탈렌 유도체, 펜타센 유도체, 페난트렌 화합물, 플루오란텐 화합물 등이 있고, 헤테로환 함유 화합물로는 디벤조퓨란 유도체, 래더형 퓨란 화합물, 피리미딘 유도체 등이 있으나, 이에 한정되는 것은 아니다. The light emitting layer may include a host material and a dopant material. The host material may be a condensed aromatic ring derivative or a heterocyclic compound. Specifically, condensed aromatic ring derivatives include anthracene derivatives, pyrene derivatives, naphthalene derivatives, pentacene derivatives, phenanthrene compounds, fluoranthene compounds, etc., and heterocyclic compounds include dibenzofuran derivatives, ladder-type furan compounds, Pyrimidine derivatives, and the like, but are not limited thereto.
상기 도펀트 재료로는 방향족 아민 유도체, 스트릴아민 화합물, 붕소 착체, 플루오란텐 화합물, 금속 착체 등이 있다. 구체적으로, 방향족 아민 유도체로는 치환 또는 비치환된 아릴아민기를 갖는 축합 방향족환 유도체로서, 아릴아민기를 갖는 피렌, 안트라센, 크리센, 페리플란텐 등이 있다. 또한, 스티릴아민 화합물은 치환 또는 비치환된 아릴아민에 적어도 1개의 아릴비닐기가 치환되어 있는 화합물로, 아릴기, 실릴기, 알킬기, 시클로알킬기 및 아릴아민기로 이루어진 군에서 1 또는 2 이상 선택되는 치환기가 치환 또는 비치환된다. 구체적으로 스티릴아민, 스티릴디아민, 스티릴트리아민, 스티릴테트라아민 등이 있으나, 이에 한정되지 않는다. 또한, 금속 착체로는 이리듐 착체, 백금 착체 등이 있으나, 이에 한정되는 것은 아니다. Examples of the dopant material include aromatic amine derivatives, strylamine compounds, boron complexes, fluoranthene compounds, and metal complexes. Specifically, aromatic amine derivatives include condensed aromatic ring derivatives having substituted or unsubstituted arylamine groups, such as pyrene, anthracene, chrysene, and periplanene having arylamine groups. In addition, the styrylamine compound is a compound in which at least one arylvinyl group is substituted with a substituted or unsubstituted arylamine, and one or two or more are selected from the group consisting of aryl groups, silyl groups, alkyl groups, cycloalkyl groups, and arylamine groups. The substituent is substituted or unsubstituted. Specifically, styrylamine, styryldiamine, styryltriamine, styryltetraamine, and the like, but are not limited thereto. In addition, examples of the metal complex include an iridium complex, a platinum complex, and the like, but are not limited thereto.
본 명세서에서, 상기 화학식 1로 표시되는 화합물이 발광층 이외의 유기물층에 포함되거나, 추가의 발광층이 구비되는 경우, 상기 발광층의 발광 물질로는 정공 수송층과 전자 수송층으로부터 정공과 전자를 각각 수송 받아 결합시킴으로써 가시광선 영역의 빛을 낼 수 있는 물질로서, 형광이나 인광에 대한 양자 효율이 좋은 물질이 바람직하다. 예를 들어, 8-히드록시퀴놀린 알루미늄 착물(Alq3); 카르바졸 계열 화합물; 이량체화 스티릴(dimerized styryl) 화합물; BAlq; 10-히드록시벤조 퀴놀린-금속 화합물; 벤족사졸, 벤즈티아졸 및 벤즈이미다졸 계열의 화합물; 폴리(p-페닐렌비닐렌)(PPV) 계열의 고분자; 스피로(spiro) 화합물; 폴리플루오렌; 및 루브렌 등이 있으나, 이에 한정되는 것은 아니다.In the present specification, when the compound represented by Chemical Formula 1 is included in an organic material layer other than the light emitting layer, or when an additional light emitting layer is provided, the light emitting material of the light emitting layer is transported and combined with holes and electrons from the hole transport layer and the electron transport layer, respectively. As a material capable of emitting light in the visible light region, a material having good quantum efficiency for fluorescence or phosphorescence is preferable. For example, 8-hydroxyquinoline aluminum complex (Alq3); Carbazole-based compounds; Dimerized styryl compounds; BAlq; 10-hydroxybenzo quinoline-metal compound; Benzoxazole, benzthiazole and benzimidazole compounds; Poly (p-phenylenevinylene) (PPV) polymers; Spiro compounds; Polyfluorene; And rubrene, but is not limited thereto.
상기 정공 주입층은 전극으로부터 정공을 수취하는 층이다. 정공 주입 물질은 정공을 수송하는 능력을 가져 양극으로부터 정공 수취 효과 및 발광층 또는 발광 재료에 대하여 우수한 정공 주입 효과를 갖는 것이 바람직하다. 또한, 발광층에서 생성된 엑시톤의 전자 주입층 또는 전자 주입 재료에의 이동을 방지할 수 있는 능력이 우수한 물질이 바람직하다. 또한, 박막 형성 능력이 우수한 물질이 바람직하다. 또한, 정공 주입 물질의 HOMO(highest occupied molecular orbital)가 양극 물질의 일함수와 주변 유기물층의 HOMO 사이인 것이 바람직하다. 정공 주입 물질의 구체적인 예로는, 금속 포피린(porphyrin), 올리고티오펜, 아릴아민 계열의 유기물; 헥사니트릴헥사아자트리페닐렌 계열의 유기물; 퀴나크리돈(quinacridone)계열의 유기물; 페릴렌(perylene) 계열의 유기물; 안트라퀴논, 폴리아닐린과 같은 폴리티오펜 계열의 전도성 고분자 등이 있으나, 이에 한정 되는 것은 아니다. The hole injection layer is a layer that receives holes from the electrode. It is preferable that the hole injection material has the ability to transport holes and thus has a hole receiving effect from the anode and an excellent hole injection effect for the light emitting layer or the light emitting material. In addition, a material having excellent ability to prevent movement of the exciton generated in the light emitting layer to the electron injection layer or the electron injection material is preferable. Also, a material having excellent thin film formation ability is preferred. In addition, it is preferable that the high-occupied molecular orbital (HOMO) of the hole injection material is between the work function of the positive electrode material and the HOMO of the surrounding organic material layer. Specific examples of the hole injection material include metal porphyrins, oligothiophenes, and arylamine-based organic materials; Hexanitrile hexaaza triphenylene series organics; Quinacridone-based organic matter; Perylene-based organics; Polythiophene-based conductive polymers such as anthraquinone and polyaniline, but are not limited thereto.
상기 정공 수송층은 정공 주입층으로부터 정공을 수취하여 발광층까지 정공을 수송하는 층이다. 정공 수송 물질로는 양극이나 정공 주입층으로부터 정공을 수취하여 발광층으로 옮겨줄 수 있는 물질로 정공에 대한 이동성이 큰 물질이 바람직하다. 구체적인 예로는, 아릴아민 계열의 유기물, 전도성 고분자, 및 공액 부분과 비공액 부분이 함께 있는 블록 공중합체 등이 있으나, 이에 한정되는 것은 아니다.The hole transport layer is a layer that receives holes from the hole injection layer and transports the holes to the light emitting layer. As the hole transport material, a material capable of receiving holes from the anode or the hole injection layer and transferring them to the light emitting layer is preferably a material having high mobility for holes. Specific examples include, but are not limited to, arylamine-based organic materials, conductive polymers, and block copolymers having conjugated and non-conjugated portions.
상기 전자 수송층은 전자 주입층으로부터 전자를 수취하여 발광층까지 전자를 수송하는 층이다. 전자 수송 물질로는 음극으로부터 전자를 잘 주입 받아 발광층으로 옮겨줄 수 있는 물질로서, 전자에 대한 이동성이 큰 물질이 바람직하다. 구체적인 예로는, 8-히드록시퀴놀린의 Al착물; Alq3를 포함한 착물; 유기 라디칼 화합물; 히드록시플라본-금속 착물 등이 있으나, 이에 한정되는 것은 아니다. 전자 수송층은 종래기술에 따라 사용된 바와 같이, 임의의 원하는 음극 물질과 함께 사용할 수 있다. 특히, 적절한 음극 물질은 낮은 일함수를 가지며, 알루미늄층 또는 실버층이 뒤따르는 통상적인 물질이다. 구체적으로, 세슘, 바륨, 칼슘, 이테르븀 및 사마륨 등이 있고, 각 경우 알루미늄층 또는 실버층이 뒤따른다.The electron transport layer is a layer that receives electrons from the electron injection layer and transports electrons to the light emitting layer. As the electron transport material, a material capable of receiving electrons well from the cathode and transferring them to the light emitting layer, a material having high mobility for electrons is preferable. Specific examples include the Al complex of 8-hydroxyquinoline; Complexes including Alq3; Organic radical compounds; Hydroxyflavone-metal complexes, and the like, but are not limited thereto. The electron transport layer can be used with any desired negative electrode material, as used according to the prior art. Particularly, a suitable negative electrode material has a low work function and is a common material followed by an aluminum layer or a silver layer. Specifically, there are cesium, barium, calcium, ytterbium, and samarium, and in each case, an aluminum layer or a silver layer follows.
상기 전자 주입층은 전극으로부터 전자를 수취하는 층이다. 전자 주입물로는 전자를 수송하는 능력이 우수하고, 제2 전극으로부터의 전자 수취 효과, 발광층 또는 발광 재료에 대하여 우수한 전자주입 효과를 갖는 것이 바람직하다. 또한, 발광층에서 생성된 엑시톤이 정공 주입층으로 이동하는 것을 방지하고, 박막 형성 능력이 우수한 물질이 바람직하다. 구체적으로는, 플루오레논, 안트라퀴노다이메탄, 다이페노퀴논, 티오피란 다이옥사이드, 옥사졸, 옥사다이아졸, 트리아졸, 이미다졸, 페릴렌테트라카복실산, 프레오레닐리덴 메탄, 안트론 등과 그들의 유도체, 금속 착체 화합물 및 함질소 5원환 유도체 등이 있으나, 이에 한정되는 것은 아니다. The electron injection layer is a layer that receives electrons from an electrode. It is preferable that the electron injecting agent has an excellent electron transporting ability and an electron receiving effect from the second electrode, and an excellent electron injection effect with respect to the light emitting layer or the light emitting material. In addition, a material that prevents exciton generated in the light emitting layer from moving to the hole injection layer and has excellent thin film formation ability is preferable. Specifically, fluorenone, anthraquinodimethane, diphenoquinone, thiopyran dioxide, oxazole, oxadiazole, triazole, imidazole, perylenetetracarboxylic acid, preorenylidene methane, anthrone and the like and their derivatives, Metal complex compounds, nitrogen-containing 5-membered ring derivatives, and the like, but are not limited thereto.
상기 금속 착체 화합물로는 8-히드록시퀴놀리나토 리튬, 비스(8-히드록시퀴놀리나토)아연, 비스(8-히드록시퀴놀리나토)구리, 비스(8-히드록시퀴놀리나토)망간, 트리스(8-히드록시퀴놀리나토)알루미늄, 트리스(2-메틸-8-히드록시퀴놀리나토)알루미늄, 트리스(8-히드록시퀴놀리나토)갈륨, 비스(10-히드록시벤조[h]퀴놀리나토)베릴륨, 비스(10-히드록시벤조[h]퀴놀리나토)아연, 비스(2-메틸-8-퀴놀리나토)클로로갈륨, 비스(2-메틸-8-퀴놀리나토)(o-크레졸라토)갈륨, 비스(2-메틸-8-퀴놀리나토)(1-나프톨라토)알루미늄, 비스(2-메틸-8-퀴놀리나토)(2-나프톨라토)갈륨 등이 있으나, 이에 한정되는 것은 아니다.Examples of the metal complex compound include 8-hydroxyquinolinato lithium, bis (8-hydroxyquinolinato) zinc, bis (8-hydroxyquinolinato) copper, and bis (8-hydroxyquinolinato) manganese , Tris (8-hydroxyquinolinato) aluminum, tris (2-methyl-8-hydroxyquinolinato) aluminum, tris (8-hydroxyquinolinato) gallium, bis (10-hydroxybenzo [h ] Quinolinato) beryllium, bis (10-hydroxybenzo [h] quinolinato) zinc, bis (2-methyl-8-quinolinato) chlorogallium, bis (2-methyl-8-quinolinato) (o-cresolato) gallium, bis (2-methyl-8-quinolinato) (1-naphtholato) aluminum, bis (2-methyl-8-quinolinato) (2-naphtolato) gallium, etc. , But is not limited thereto.
상기 전자 차단층은 전자 주입층으로부터 주입된 전자가 발광층을 지나 정공 주입층으로 진입하는 것을 방지하여 소자의 수명과 효율을 향상시킬 수 있는 층이다. 공지된 재료는 제한 없이 사용 가능하며, 발광층과 정공 주입층 사이에, 또는 발광층과 정공 주입 및 정공 수송을 동시에 하는 층 사이에 형성될 수 있다. The electron blocking layer is a layer capable of improving the life and efficiency of the device by preventing electrons injected from the electron injection layer from entering the hole injection layer through the light emitting layer. Known materials can be used without limitation, and can be formed between the light emitting layer and the hole injection layer, or between the light emitting layer and the layer simultaneously performing hole injection and hole transport.
상기 정공 차단층은 정공의 음극으로 도달을 저지하는 층으로, 일반적으로 전자 주입층과 동일한 조건으로 형성될 수 있다. 구체적으로, 옥사디아졸 유도체나 트리아졸 유도체, 페난트롤린 유도체, 알루미늄 착물 (aluminum complex) 등이 있으나, 이에 한정되는 것은 아니다. The hole blocking layer is a layer that blocks reaching the cathode of the hole, and may be generally formed under the same conditions as the electron injection layer. Specifically, there are oxadiazole derivatives, triazole derivatives, phenanthroline derivatives, aluminum complexes, and the like, but are not limited thereto.
본 명세서에 따른 유기 발광 소자는 사용되는 재료에 따라 전면 발광형, 후면 발광형 또는 양면 발광형일 수 있다.The organic light emitting device according to the present specification may be a front emission type, a back emission type, or a double-sided emission type, depending on the material used.
이하, 본 명세서를 구체적으로 설명하기 위해 실시예 및 비교예 등을 들어 상세하게 설명하기로 한다. 그러나, 본 명세서에 따른 실시예 및 비교예는 여러 가지 다른 형태로 변형될 수 있으며, 본 명세서의 범위가 아래에서 상술하는 실시예 및 비교예에 한정되는 것으로 해석되지 않는다. 본 명세서의 실시예 및 비교예는 당업계에서 평균적인 지식을 가진 자에게 본 명세서를 보다 완전하게 설명하기 위해 제공되는 것이다.Hereinafter, examples and comparative examples will be described in detail to specifically describe the present specification. However, the examples and comparative examples according to the present specification may be modified in various other forms, and the scope of the present specification is not interpreted to be limited to the examples and comparative examples described below. The examples and comparative examples in the present specification are provided to more fully describe the present specification to those skilled in the art.
본 발명의 화합물은 대표적인 반응으로 Buchwald-Hartwig coupling reaction, Suzuki coupling reaction, Heck coupling reaction 등을 이용하여 제조되었다.The compounds of the present invention were prepared using Buchwald-Hartwig coupling reaction, Suzuki coupling reaction, Heck coupling reaction, and the like as representative reactions.
제조예 1.Preparation Example 1.
2-nitronaphthalen-1-yl trifluoromethanesulfonate 100.0g (1.0 eq), triphenylen-1-ylboronic acid 93.17 g (1.1 eq)을 THF 1000ml 에 녹인 후 물 300ml 에 녹인 K2CO3 86.05 g (2.0 eq)을 같이 넣어 주었다. Pd(t-Bu3P)2 1.59 g (0.005 eq) 을 넣어 주고 환류하여 교반했다. 반응이 종료되면 감압하여 용매를 제거하였다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 무수황산마그네슘으로 처리하여 다시 감압하여 용매를 제거하고 이를 컬럼크로마토그래피하여 화합물 A-1 90.77 g (수율 73 %)를 얻었다. [M+H]=4002-nitronaphthalen-1-yl trifluoromethanesulfonate 100.0 g (1.0 eq), triphenylen-1-ylboronic acid 93.17 g (1.1 eq) was dissolved in 1000 ml of THF, and 86.05 g (2.0 eq) of K 2 CO 3 dissolved in 300 ml of water was added together. gave. Pd ( t -Bu 3 P) 2 1.59 g (0.005 eq) was added and refluxed and stirred. When the reaction was completed, the solvent was removed under reduced pressure. Thereafter, the mixture was completely dissolved in CHCl 3 , washed with water, treated with anhydrous magnesium sulfate, and then decompressed again to remove the solvent, and subjected to column chromatography to obtain 90.77 g of Compound A-1 (yield 73%). [M + H] = 400
화학식 A-1 90.77g (1.0 eq) Triethylphosphite 200 mL에 넣고 환류하여 교반했다. 2시간 후 반응 종결하고 에탄올 2L에 반응물을 부어서 고체를 떨어트렸다. 이 고체를 CHCl3에 완전히 녹인 후 물로 씻어주고 무수황산마그네슘으로 처리하여 용액을 감압 농축하고 컬럼크로마토그래피를 이용하여 정제하였다. 화합물 A 53.44g (수율 63 %)을 얻었다. [M+H]=218Formula A-1 90.77g (1.0 eq) Triethylphosphite was added to 200 mL and stirred under reflux. After 2 hours, the reaction was terminated and the reaction was poured into 2 L of ethanol to drop a solid. The solid was completely dissolved in CHCl 3 , washed with water, treated with anhydrous magnesium sulfate, and the solution was concentrated under reduced pressure and purified by column chromatography. Compound A 53.44 g (yield 63%) was obtained. [M + H] = 218
제조예 2. Preparation Example 2.
제조예 1에서 2-nitronaphthalen-1-yl trifluoromethanesulfonate 대신 3-nitronaphthalen-2-yl trifluoromethanesulfonate 을 사용하여 화학식 A의 제조 방법과 같은 방법으로 화학식 B 를 합성했다.In Preparation Example 1, instead of 2-nitronaphthalen-1-yl trifluoromethanesulfonate, 3-nitronaphthalen-2-yl trifluoromethanesulfonate was used to synthesize Formula B in the same manner as in Formula A.
제조예 3. Preparation Example 3.
제조예 1에서 2-nitronaphthalen-1-yl trifluoromethanesulfonate 대신 3- 1-nitronaphthalen-2-yl trifluoromethanesulfonate 을 사용하여 화학식 A의 제조 방법과 같은 방법으로 화학식 C 를 합성했다In Preparation Example 1, instead of 2-nitronaphthalen-1-yl trifluoromethanesulfonate, 3-C- 1-nitronaphthalen-2-yl trifluoromethanesulfonate was used to synthesize Formula C in the same manner as in Formula A.
제조예 4.Preparation Example 4.
1-chloronaphthalen-2-amine 100.0g (1.0 eq), 1-bromotriphenylene 190.13 g (1.1 eq)을 톨루엔 1000ml 에 녹인 후 NaOtBu 108.58 g (2.0 eq)을 같이 넣어 주었다. Pd(t-Bu3P)2 2.88 g (0.01 eq) 을 넣어 주고 환류하여 교반했다. 반응이 종료되면 감압하여 용매를 제거하였다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 무수황산마그네슘으로 처리하여 다시 감압하여 용매를 제거하고 이를 컬럼크로마토그래피하여 화합물 D-1 152.56 g (수율 67 %)를 얻었다. [M+H]=404100.0 g (1.0 eq) of 1-chloronaphthalen-2-amine and 190.13 g (1.1 eq) of 1-bromotriphenylene were dissolved in 1000 ml of toluene, and then NaOtBu 108.58 g (2.0 eq) was added together. Pd ( t -Bu 3 P) 2 2.88 g (0.01 eq) was added and refluxed and stirred. When the reaction was completed, the solvent was removed under reduced pressure. Thereafter, the mixture was completely dissolved in CHCl 3 , washed with water, treated with anhydrous magnesium sulfate, and then decompressed again to remove the solvent, and subjected to column chromatography to obtain 152.56 g of Compound D-1 (yield 67%). [M + H] = 404
화합물 D-1 152.56 g (1.0 eq) 을 DMAc 1000ml 에 녹인 후 K3PO4 160.67 g (2.0 eq), Pd(t-Bu3P)2 1.93 g (0.01 eq) 을 넣어 주고 환류하여 교반했다. 반응이 종료되면 식힌 후 물에 부어서 교반 및 고체화 진행 후 여과했다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 무수황산마그네슘으로 처리하여 다시 감압하여 용매를 제거하고 이를 컬럼크로마토그래피하여 화합물 D 94.48 g (수율 68 %)를 얻었다. [M+H]=368After dissolving 152.56 g (1.0 eq) of Compound D-1 in 1000 ml of DMAc, K 3 PO 4 160.67 g (2.0 eq), Pd ( t -Bu 3 P) 2 1.93 g (0.01 eq) was added and refluxed and stirred. After the reaction was completed, it was cooled, poured into water, stirred and solidified, and filtered. Thereafter, the mixture was completely dissolved in CHCl 3 , washed with water, treated with anhydrous magnesium sulfate, and then decompressed again to remove the solvent. This was subjected to column chromatography, thereby obtaining Compound D 94.48 g (68% yield). [M + H] = 368
제조예 5.Preparation Example 5.
제조예 4에서 1-chloronaphthalen-2-amine 대신 3-chloronaphthalen-2-amine 을 사용하여 화학식 D의 제조 방법과 같은 방법으로 화학식 E 를 합성했다.In Preparation Example 4, 3-chloronaphthalen-2-amine was used instead of 1-chloronaphthalen-2-amine to synthesize Formula E in the same manner as in Formula D.
제조예 6.Preparation Example 6.
제조예 4에서 1-chloronaphthalen-2-amine 대신 2-chloronaphthalen-1-amine 을 사용하여 화학식 D의 제조 방법과 같은 방법으로 화학식 F 를 합성했다.In Preparation Example 4, instead of 1-chloronaphthalen-2-amine, 2-chloronaphthalen-1-amine was used to synthesize Formula F in the same manner as in Formula D.
<합성예><Synthesis example>
합성예 1Synthesis Example 1
화합물 A 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 8.00 g (1.1 eq), Pd(t-Bu3P)2 0.13 g (0.01 eq), K3PO4 11.56 g (2.0 eq)를 Xylene 250ml에 넣어서 환류하여 교반했다. 2 시간 후 반응이 종료되면 감압하여 용매를 제거했다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 다시 감압하여 용매를 50% 정도 제거했다. 다시 환류 상태에서 에틸아세테이트를 넣어주며 결정을 떨어트려 식힌 후 여과했다. 이를 컬럼크로마토그래피하여 화합물 1 12.22 g (수율 75 %)를 얻었다. [M+H]=599Compound A 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 8.00 g (1.1 eq), Pd (t-Bu 3 P) 2 0.13 g (0.01 eq), K 3 PO 4 11.56 g (2.0 eq) It was put in 250 ml of Xylene and refluxed and stirred. When the reaction was completed after 2 hours, the solvent was removed under reduced pressure. After that, the mixture was completely dissolved in CHCl 3 , washed with water, and then reduced again to remove about 50% of the solvent. Ethyl acetate was added again under reflux, the crystals were dropped, cooled, and filtered. This was subjected to column chromatography, thereby obtaining 12.22 g of a compound 1 (75% yield). [M + H] = 599
합성예 2Synthesis Example 2
화합물 A 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 10.69 g (1.1 eq), Pd(t-Bu3P)2 0.13 g (0.01 eq), K3PO4 11.56 g (2.0 eq)를 Xylene 250ml에 넣어서 환류하여 교반했다. 2 시간 후 반응이 종료되면 감압하여 용매를 제거했다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 다시 감압하여 용매를 50% 정도 제거했다. 다시 환류 상태에서 에틸아세테이트를 넣어주며 결정을 떨어트려 식힌 후 여과했다. 이를 컬럼크로마토그래피하여 화합물 2 13.30 g (수율 71 %)를 얻었다. [M+H]=689Compound A 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 10.69 g (1.1 eq), Pd (t-Bu 3 P) 2 0.13 g (0.01 eq), K 3 PO 4 11.56 g (2.0 eq) It was put in 250 ml of Xylene and refluxed and stirred. When the reaction was completed after 2 hours, the solvent was removed under reduced pressure. After that, the mixture was completely dissolved in CHCl 3 , washed with water, and then reduced again to remove about 50% of the solvent. Ethyl acetate was added again under reflux, the crystals were dropped, cooled, and filtered. This was subjected to column chromatography, thereby obtaining 13.30 g of compound 2 (yield 71%). [M + H] = 689
합성예 3Synthesis Example 3
화합물 B 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 9.49 g (1.1 eq), Pd(t-Bu3P)2 0.13 g (0.01 eq), K3PO4 11.56 g (2.0 eq)를 Xylene 250ml에 넣어서 환류하여 교반했다. 2 시간 후 반응이 종료되면 감압하여 용매를 제거했다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 다시 감압하여 용매를 50% 정도 제거했다. 다시 환류 상태에서 에틸아세테이트를 넣어주며 결정을 떨어트려 식힌 후 여과했다. 이를 컬럼크로마토그래피하여 화합물 3 12.88 g (수율 73 %)를 얻었다. [M+H]=649Compound B 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 9.49 g (1.1 eq), Pd (t-Bu 3 P) 2 0.13 g (0.01 eq), K 3 PO 4 11.56 g (2.0 eq) It was put in 250 ml of Xylene and refluxed and stirred. When the reaction was completed after 2 hours, the solvent was removed under reduced pressure. After that, the mixture was completely dissolved in CHCl 3 , washed with water, and then reduced again to remove about 50% of the solvent. Ethyl acetate was added again under reflux, the crystals were dropped, cooled, and filtered. This was subjected to column chromatography, thereby obtaining 12.88 g of a compound 3 (yield 73%). [M + H] = 649
합성예 4Synthesis Example 4
화합물 B 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 12.49 g (1.1 eq), Pd(t-Bu3P)2 0.13 g (0.01 eq), K3PO4 11.56 g (2.0 eq)를 Xylene 250ml에 넣어서 환류하여 교반했다. 2 시간 후 반응이 종료되면 감압하여 용매를 제거했다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 다시 감압하여 용매를 50% 정도 제거했다. 다시 환류 상태에서 에틸아세테이트를 넣어주며 결정을 떨어트려 식힌 후 여과했다. 이를 컬럼크로마토그래피하여 화합물 4 13.65 g (수율 67 %)를 얻었다. [M+H]=749Compound B 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 12.49 g (1.1 eq), Pd (t-Bu 3 P) 2 0.13 g (0.01 eq), K 3 PO 4 11.56 g (2.0 eq) It was put in 250 ml of Xylene and refluxed and stirred. When the reaction was completed after 2 hours, the solvent was removed under reduced pressure. After that, the mixture was completely dissolved in CHCl 3 , washed with water, and then reduced again to remove about 50% of the solvent. Ethyl acetate was added again under reflux, the crystals were dropped, cooled, and filtered. This was subjected to column chromatography, thereby obtaining 13.65 g of Compound 4 (yield 67%). [M + H] = 749
합성예 5Synthesis Example 5
화합물 B 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 12.97 g (1.1 eq), Pd(t-Bu3P)2 0.13 g (0.01 eq), K3PO4 11.56 g (2.0 eq)를 Xylene 250ml에 넣어서 환류하여 교반했다. 2 시간 후 반응이 종료되면 감압하여 용매를 제거했다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 다시 감압하여 용매를 50% 정도 제거했다. 다시 환류 상태에서 에틸아세테이트를 넣어주며 결정을 떨어트려 식힌 후 여과했다. 이를 컬럼크로마토그래피하여 화합물 5 15.19 g (수율 73 %)를 얻었다. [M+H]=765Compound B 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 12.97 g (1.1 eq), Pd (t-Bu 3 P) 2 0.13 g (0.01 eq), K 3 PO 4 11.56 g (2.0 eq) It was put in 250 ml of Xylene and refluxed and stirred. When the reaction was completed after 2 hours, the solvent was removed under reduced pressure. After that, the mixture was completely dissolved in CHCl 3 , washed with water, and then reduced again to remove about 50% of the solvent. Ethyl acetate was added again under reflux, the crystals were dropped, cooled, and filtered. This was subjected to column chromatography, thereby obtaining 15.19 g of Compound 5 (yield 73%). [M + H] = 765
합성예 6Synthesis Example 6
화합물 C 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 12.56 g (1.1 eq), Pd(t-Bu3P)2 0.13 g (0.01 eq), K3PO4 11.56 g (2.0 eq)를 Xylene 250ml에 넣어서 환류하여 교반했다. 2 시간 후 반응이 종료되면 감압하여 용매를 제거했다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 다시 감압하여 용매를 50% 정도 제거했다. 다시 환류 상태에서 에틸아세테이트를 넣어주며 결정을 떨어트려 식힌 후 여과했다. 이를 컬럼크로마토그래피하여 화합물 6 12.46 g (수율 61 %)를 얻었다. [M+H]=751Compound C 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 12.56 g (1.1 eq), Pd (t-Bu 3 P) 2 0.13 g (0.01 eq), K 3 PO 4 11.56 g (2.0 eq) It was put in 250 ml of Xylene and refluxed and stirred. When the reaction was completed after 2 hours, the solvent was removed under reduced pressure. After that, the mixture was completely dissolved in CHCl 3 , washed with water, and then reduced again to remove about 50% of the solvent. Ethyl acetate was added again under reflux, the crystals were dropped, cooled, and filtered. This was subjected to column chromatography, thereby obtaining 12.46 g of a compound 6 (yield 61%). [M + H] = 751
합성예 7Synthesis Example 7
화합물 C 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 12.67 g (1.1 eq), Pd(t-Bu3P)2 0.13 g (0.01 eq), K3PO4 11.56 g (2.0 eq)를 Xylene 250ml에 넣어서 환류하여 교반했다. 2 시간 후 반응이 종료되면 감압하여 용매를 제거했다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 다시 감압하여 용매를 50% 정도 제거했다. 다시 환류 상태에서 에틸아세테이트를 넣어주며 결정을 떨어트려 식힌 후 여과했다. 이를 컬럼크로마토그래피하여 화합물 7 12.94 g (수율 63 %)를 얻었다. [M+H]=755Compound C 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 12.67 g (1.1 eq), Pd (t-Bu 3 P) 2 0.13 g (0.01 eq), K 3 PO 4 11.56 g (2.0 eq) It was put in 250 ml of Xylene and refluxed and stirred. When the reaction was completed after 2 hours, the solvent was removed under reduced pressure. After that, the mixture was completely dissolved in CHCl 3 , washed with water, and then reduced again to remove about 50% of the solvent. Ethyl acetate was added again under reflux, the crystals were dropped, cooled, and filtered. This was subjected to column chromatography, thereby obtaining 12.94 g of Compound 7 (63% yield). [M + H] = 755
합성예 8Synthesis Example 8
화합물 C 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 12.94 g (1.1 eq), Pd(t-Bu3P)2 0.13 g (0.01 eq), K3PO4 11.56 g (2.0 eq)를 Xylene 250ml에 넣어서 환류하여 교반했다. 2 시간 후 반응이 종료되면 감압하여 용매를 제거했다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 다시 감압하여 용매를 50% 정도 제거했다. 다시 환류 상태에서 에틸아세테이트를 넣어주며 결정을 떨어트려 식힌 후 여과했다. 이를 컬럼크로마토그래피하여 화합물 8 12.68 g (수율 61 %)를 얻었다. [M+H]=764Compound C 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 12.94 g (1.1 eq), Pd (t-Bu 3 P) 2 0.13 g (0.01 eq), K 3 PO 4 11.56 g (2.0 eq) It was put in 250 ml of Xylene and refluxed and stirred. When the reaction was completed after 2 hours, the solvent was removed under reduced pressure. After that, the mixture was completely dissolved in CHCl 3 , washed with water, and then reduced again to remove about 50% of the solvent. Ethyl acetate was added again under reflux, the crystals were dropped, cooled, and filtered. This was subjected to column chromatography, thereby obtaining 12.68 g of a compound 8 (yield 61%). [M + H] = 764
합성예 9Synthesis Example 9
화합물 A 10.0 g (1.0 eq), 2-chloro-4-(4-(naphthalen-1-yl)phenyl)quinazoline 12.34 g (1.1 eq), Pd(t-Bu3P)2 0.14 g (0.01 eq), NaOtBu 5.23 g (2.0 eq)를 Xylene 250ml에 넣어서 환류하여 교반했다. 2 시간 후 반응이 종료되면 감압하여 용매를 제거했다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 다시 감압하여 용매를 50% 정도 제거했다. 다시 환류 상태에서 에틸아세테이트를 넣어주며 결정을 떨어트려 식힌 후 여과했다. 이를 컬럼크로마토그래피하여 화합물 9 12.37 g (수율 65 %)를 얻었다. [M+H]=700Compound A 10.0 g (1.0 eq), 2-chloro-4- (4- (naphthalen-1-yl) phenyl) quinazoline 12.34 g (1.1 eq), Pd (t-Bu 3 P) 2 0.14 g (0.01 eq) , NaOtBu 5.23 g (2.0 eq) was added to 250 ml of Xylene and refluxed and stirred. When the reaction was completed after 2 hours, the solvent was removed under reduced pressure. After that, the mixture was completely dissolved in CHCl 3 , washed with water, and then reduced again to remove about 50% of the solvent. Ethyl acetate was added again under reflux, the crystals were dropped, cooled, and filtered. This was subjected to column chromatography, thereby obtaining 12.37 g of Compound 9 (65% yield). [M + H] = 700
합성예 10Synthesis Example 10
화합물 A 10.0 g (1.0 eq), 2-chloro-4-(4-(naphthalen-1-yl)phenyl)quinazoline 13.09 g (1.1 eq), Pd(t-Bu3P)2 0.14 g (0.01 eq), NaOtBu 5.23 g (2.0 eq)를 Xylene 250ml에 넣어서 환류하여 교반했다. 2 시간 후 반응이 종료되면 감압하여 용매를 제거했다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 다시 감압하여 용매를 50% 정도 제거했다. 다시 환류 상태에서 에틸아세테이트를 넣어주며 결정을 떨어트려 식힌 후 여과했다. 이를 컬럼크로마토그래피하여 화합물 10 12.03 g (수율 61 %)를 얻었다. [M+H]=725Compound A 10.0 g (1.0 eq), 2-chloro-4- (4- (naphthalen-1-yl) phenyl) quinazoline 13.09 g (1.1 eq), Pd (t-Bu 3 P) 2 0.14 g (0.01 eq) , NaOtBu 5.23 g (2.0 eq) was added to 250 ml of Xylene and refluxed and stirred. When the reaction was completed after 2 hours, the solvent was removed under reduced pressure. After that, the mixture was completely dissolved in CHCl 3 , washed with water, and then reduced again to remove about 50% of the solvent. Ethyl acetate was added again under reflux, the crystals were dropped, cooled, and filtered. This was subjected to column chromatography, thereby obtaining 12.03 g of a compound 10 (yield 61%). [M + H] = 725
합성예 11Synthesis Example 11
화합물 A 10.0 g (1.0 eq), 2-chloro-4-(4-(naphthalen-1-yl)phenyl)quinazoline 13.24 g (1.1 eq), Pd(t-Bu3P)2 0.14 g (0.01 eq), NaOtBu 5.23 g (2.0 eq)를 Xylene 250ml에 넣어서 환류하여 교반했다. 2 시간 후 반응이 종료되면 감압하여 용매를 제거했다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 다시 감압하여 용매를 50% 정도 제거했다. 다시 환류 상태에서 에틸아세테이트를 넣어주며 결정을 떨어트려 식힌 후 여과했다. 이를 컬럼크로마토그래피하여 화합물 11 13.30 g (수율 67 %)를 얻었다. [M+H]=730Compound A 10.0 g (1.0 eq), 2-chloro-4- (4- (naphthalen-1-yl) phenyl) quinazoline 13.24 g (1.1 eq), Pd (t-Bu 3 P) 2 0.14 g (0.01 eq) , NaOtBu 5.23 g (2.0 eq) was added to 250 ml of Xylene and refluxed and stirred. When the reaction was completed after 2 hours, the solvent was removed under reduced pressure. After that, the mixture was completely dissolved in CHCl 3 , washed with water, and then reduced again to remove about 50% of the solvent. Ethyl acetate was added again under reflux, the crystals were dropped, cooled, and filtered. This was subjected to column chromatography to obtain 13.30 g of compound 11 (yield 67%). [M + H] = 730
합성예 12Synthesis Example 12
화합물 C 10.0 g (1.0 eq), 2-chloro-4-(4-(naphthalen-1-yl)phenyl)quinazoline 14.29 g (1.1 eq), Pd(t-Bu3P)2 0.14 g (0.01 eq), NaOtBu 5.23 g (2.0 eq)를 Xylene 250ml에 넣어서 환류하여 교반했다. 2 시간 후 반응이 종료되면 감압하여 용매를 제거했다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 다시 감압하여 용매를 50% 정도 제거했다. 다시 환류 상태에서 에틸아세테이트를 넣어주며 결정을 떨어트려 식힌 후 여과했다. 이를 컬럼크로마토그래피하여 화합물 12 13.32 g (수율 64 %)를 얻었다. [M+H]=765Compound C 10.0 g (1.0 eq), 2-chloro-4- (4- (naphthalen-1-yl) phenyl) quinazoline 14.29 g (1.1 eq), Pd (t-Bu 3 P) 2 0.14 g (0.01 eq) , NaOtBu 5.23 g (2.0 eq) was added to 250 ml of Xylene and refluxed and stirred. When the reaction was completed after 2 hours, the solvent was removed under reduced pressure. After that, the mixture was completely dissolved in CHCl 3 , washed with water, and then reduced again to remove about 50% of the solvent. Ethyl acetate was added again under reflux, the crystals were dropped, cooled, and filtered. This was subjected to column chromatography, thereby obtaining 13.32 g of compound 12 (yield 64%). [M + H] = 765
합성예 13Synthesis Example 13
화합물 D 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 12.55 g (1.1 eq), Pd(t-Bu3P)2 0.13 g (0.01 eq), K3PO4 11.56 g (2.0 eq)를 Xylene 250ml에 넣어서 환류하여 교반했다. 2 시간 후 반응이 종료되면 감압하여 용매를 제거했다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 다시 감압하여 용매를 50% 정도 제거했다. 다시 환류 상태에서 에틸아세테이트를 넣어주며 결정을 떨어트려 식힌 후 여과했다. 이를 컬럼크로마토그래피하여 화합물 13 12.26 g (수율 60 %)를 얻었다. [M+H]=751Compound D 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 12.55 g (1.1 eq), Pd (t-Bu 3 P) 2 0.13 g (0.01 eq), K 3 PO 4 11.56 g (2.0 eq) It was put in 250 ml of Xylene and refluxed and stirred. When the reaction was completed after 2 hours, the solvent was removed under reduced pressure. After that, the mixture was completely dissolved in CHCl 3 , washed with water, and then reduced again to remove about 50% of the solvent. Ethyl acetate was added again under reflux, the crystals were dropped, cooled, and filtered. This was subjected to column chromatography, thereby obtaining 12.26 g of a compound 13 (60% yield). [M + H] = 751
합성예 14Synthesis Example 14
화합물 D 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 12.94 g (1.1 eq), Pd(t-Bu3P)2 0.13 g (0.01 eq), K3PO4 11.56 g (2.0 eq)를 Xylene 250ml에 넣어서 환류하여 교반했다. 2 시간 후 반응이 종료되면 감압하여 용매를 제거했다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 다시 감압하여 용매를 50% 정도 제거했다. 다시 환류 상태에서 에틸아세테이트를 넣어주며 결정을 떨어트려 식힌 후 여과했다. 이를 컬럼크로마토그래피하여 화합물 14 13.09 g (수율 63 %)를 얻었다. [M+H]=764Compound D 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 12.94 g (1.1 eq), Pd (t-Bu 3 P) 2 0.13 g (0.01 eq), K 3 PO 4 11.56 g (2.0 eq) It was put in 250 ml of Xylene and refluxed and stirred. When the reaction was completed after 2 hours, the solvent was removed under reduced pressure. After that, the mixture was completely dissolved in CHCl 3 , washed with water, and then reduced again to remove about 50% of the solvent. Ethyl acetate was added again under reflux, the crystals were dropped, cooled, and filtered. This was subjected to column chromatography to obtain 13.09 g of compound 14 (63% yield). [M + H] = 764
합성예 15Synthesis Example 15
화합물 E 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 10.99 g (1.1 eq), Pd(t-Bu3P)2 0.13 g (0.01 eq), K3PO4 11.56 g (2.0 eq)를 Xylene 250ml에 넣어서 환류하여 교반했다. 2 시간 후 반응이 종료되면 감압하여 용매를 제거했다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 다시 감압하여 용매를 50% 정도 제거했다. 다시 환류 상태에서 에틸아세테이트를 넣어주며 결정을 떨어트려 식힌 후 여과했다. 이를 컬럼크로마토그래피하여 화합물 15 11.41 g (수율 60 %)를 얻었다. [M+H]=699Compound E 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 10.99 g (1.1 eq), Pd (t-Bu 3 P) 2 0.13 g (0.01 eq), K 3 PO 4 11.56 g (2.0 eq) It was put in 250 ml of Xylene and refluxed and stirred. When the reaction was completed after 2 hours, the solvent was removed under reduced pressure. After that, the mixture was completely dissolved in CHCl 3 , washed with water, and then reduced again to remove about 50% of the solvent. Ethyl acetate was added again under reflux, the crystals were dropped, cooled, and filtered. This was subjected to column chromatography, thereby obtaining 11.41 g of compound 15 (yield 60%). [M + H] = 699
합성예 16Synthesis Example 16
화합물 E 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 11.18 g (1.1 eq), Pd(t-Bu3P)2 0.13 g (0.01 eq), K3PO4 11.56 g (2.0 eq)를 Xylene 250ml에 넣어서 환류하여 교반했다. 2 시간 후 반응이 종료되면 감압하여 용매를 제거했다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 다시 감압하여 용매를 50% 정도 제거했다. 다시 환류 상태에서 에틸아세테이트를 넣어주며 결정을 떨어트려 식힌 후 여과했다. 이를 컬럼크로마토그래피하여 화합물 16 10.93 g (수율 57 %)를 얻었다. [M+H]=705Compound E 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 11.18 g (1.1 eq), Pd (t-Bu 3 P) 2 0.13 g (0.01 eq), K 3 PO 4 11.56 g (2.0 eq) It was put in 250 ml of Xylene and refluxed and stirred. When the reaction was completed after 2 hours, the solvent was removed under reduced pressure. After that, the mixture was completely dissolved in CHCl 3 , washed with water, and then reduced again to remove about 50% of the solvent. Ethyl acetate was added again under reflux, the crystals were dropped, cooled, and filtered. This was subjected to column chromatography, thereby obtaining 10.93 g of Compound 16 (yield 57%). [M + H] = 705
합성예 17Synthesis Example 17
화합물 E 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 11.32 g (1.1 eq), Pd(t-Bu3P)2 0.13 g (0.01 eq), K3PO4 11.56 g (2.0 eq)를 Xylene 250ml에 넣어서 환류하여 교반했다. 2 시간 후 반응이 종료되면 감압하여 용매를 제거했다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 다시 감압하여 용매를 50% 정도 제거했다. 다시 환류 상태에서 에틸아세테이트를 넣어주며 결정을 떨어트려 식힌 후 여과했다. 이를 컬럼크로마토그래피하여 화합물 17 10.62 g (수율 55 %)를 얻었다. [M+H]=710Compound E 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 11.32 g (1.1 eq), Pd (t-Bu 3 P) 2 0.13 g (0.01 eq), K 3 PO 4 11.56 g (2.0 eq) It was put in 250 ml of Xylene and refluxed and stirred. When the reaction was completed after 2 hours, the solvent was removed under reduced pressure. After that, the mixture was completely dissolved in CHCl 3 , washed with water, and then reduced again to remove about 50% of the solvent. Ethyl acetate was added again under reflux, the crystals were dropped, cooled, and filtered. This was subjected to column chromatography, thereby obtaining 10.62 g of Compound 17 (yield 55%). [M + H] = 710
합성예 18Synthesis Example 18
화합물 F 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 10.99 g (1.1 eq), Pd(t-Bu3P)2 0.13 g (0.01 eq), K3PO4 11.56 g (2.0 eq)를 Xylene 250ml에 넣어서 환류하여 교반했다. 2 시간 후 반응이 종료되면 감압하여 용매를 제거했다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 다시 감압하여 용매를 50% 정도 제거했다. 다시 환류 상태에서 에틸아세테이트를 넣어주며 결정을 떨어트려 식힌 후 여과했다. 이를 컬럼크로마토그래피하여 화합물 18 10.84 g (수율 57 %)를 얻었다. [M+H]=699Compound F 10.0 g (1.0 eq), 2-chloro-4-phenylquinazoline 10.99 g (1.1 eq), Pd (t-Bu 3 P) 2 0.13 g (0.01 eq), K 3 PO 4 11.56 g (2.0 eq) It was put in 250 ml of Xylene and refluxed and stirred. When the reaction was completed after 2 hours, the solvent was removed under reduced pressure. After that, the mixture was completely dissolved in CHCl 3 , washed with water, and then reduced again to remove about 50% of the solvent. Ethyl acetate was added again under reflux, the crystals were dropped, cooled, and filtered. This was subjected to column chromatography, thereby obtaining 10.84 g of a compound 18 (yield 57%). [M + H] = 699
합성예 19Synthesis Example 19
화합물 D 10.0 g (1.0 eq), 2-chloro-4-(4-(naphthalen-1-yl)phenyl)quinazoline 12.34 g (1.1 eq), Pd(t-Bu3P)2 0.14 g (0.01 eq), NaOtBu 5.23 g (2.0 eq)를 Xylene 250ml에 넣어서 환류하여 교반했다. 2 시간 후 반응이 종료되면 감압하여 용매를 제거했다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 다시 감압하여 용매를 50% 정도 제거했다. 다시 환류 상태에서 에틸아세테이트를 넣어주며 결정을 떨어트려 식힌 후 여과했다. 이를 컬럼크로마토그래피하여 화합물 19 9.71 g (수율 51 %)를 얻었다. [M+H]=700Compound D 10.0 g (1.0 eq), 2-chloro-4- (4- (naphthalen-1-yl) phenyl) quinazoline 12.34 g (1.1 eq), Pd (t-Bu 3 P) 2 0.14 g (0.01 eq) , NaOtBu 5.23 g (2.0 eq) was added to 250 ml of Xylene and refluxed and stirred. When the reaction was completed after 2 hours, the solvent was removed under reduced pressure. After that, the mixture was completely dissolved in CHCl 3 , washed with water, and then reduced again to remove about 50% of the solvent. Ethyl acetate was added again under reflux, the crystals were dropped, cooled, and filtered. This was subjected to column chromatography, thereby obtaining 9.71 g (Yield 51%) of Compound 19. [M + H] = 700
합성예 20Synthesis Example 20
화합물 D 10.0 g (1.0 eq), 2-chloro-4-(4-(naphthalen-1-yl)phenyl)quinazoline 19.59 g (1.1 eq), Pd(t-Bu3P)2 0.14 g (0.01 eq), NaOtBu 5.23 g (2.0 eq)를 Xylene 250ml에 넣어서 환류하여 교반했다. 2 시간 후 반응이 종료되면 감압하여 용매를 제거했다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 다시 감압하여 용매를 50% 정도 제거했다. 다시 환류 상태에서 에틸아세테이트를 넣어주며 결정을 떨어트려 식힌 후 여과했다. 이를 컬럼크로마토그래피하여 화합물 20 12.69 g (수율 50 %)를 얻었다. [M+H]=933Compound D 10.0 g (1.0 eq), 2-chloro-4- (4- (naphthalen-1-yl) phenyl) quinazoline 19.59 g (1.1 eq), Pd (t-Bu 3 P) 2 0.14 g (0.01 eq) , NaOtBu 5.23 g (2.0 eq) was added to 250 ml of Xylene and refluxed and stirred. When the reaction was completed after 2 hours, the solvent was removed under reduced pressure. After that, the mixture was completely dissolved in CHCl 3 , washed with water, and then reduced again to remove about 50% of the solvent. Ethyl acetate was added again under reflux, the crystals were dropped, cooled, and filtered. This was subjected to column chromatography, thereby obtaining 12.69 g of a compound 20 (50% yield). [M + H] = 933
합성예 21Synthesis Example 21
화합물 E 10.0 g (1.0 eq), 2-chloro-4-(4-(naphthalen-1-yl)phenyl)quinazoline 17.05 g (1.1 eq), Pd(t-Bu3P)2 0.14 g (0.01 eq), NaOtBu 5.23 g (2.0 eq)를 Xylene 250ml에 넣어서 환류하여 교반했다. 2 시간 후 반응이 종료되면 감압하여 용매를 제거했다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 다시 감압하여 용매를 50% 정도 제거했다. 다시 환류 상태에서 에틸아세테이트를 넣어주며 결정을 떨어트려 식힌 후 여과했다. 이를 컬럼크로마토그래피하여 화합물 21 15.86 g (수율 68 %)를 얻었다. [M+H]=857Compound E 10.0 g (1.0 eq), 2-chloro-4- (4- (naphthalen-1-yl) phenyl) quinazoline 17.05 g (1.1 eq), Pd (t-Bu 3 P) 2 0.14 g (0.01 eq) , NaOtBu 5.23 g (2.0 eq) was added to 250 ml of Xylene and refluxed and stirred. When the reaction was completed after 2 hours, the solvent was removed under reduced pressure. After that, the mixture was completely dissolved in CHCl 3 , washed with water, and then reduced again to remove about 50% of the solvent. Ethyl acetate was added again under reflux, the crystals were dropped, cooled, and filtered. This was subjected to column chromatography, thereby obtaining 15.86 g of a compound 21 (68% yield). [M + H] = 857
합성예 22Synthesis Example 22
화합물 E 10.0 g (1.0 eq), 2-chloro-4-(4-(naphthalen-1-yl)phenyl)quinazoline 17.05 g (1.1 eq), Pd(t-Bu3P)2 0.14 g (0.01 eq), NaOtBu 5.23 g (2.0 eq)를 Xylene 250ml에 넣어서 환류하여 교반했다. 2 시간 후 반응이 종료되면 감압하여 용매를 제거했다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 다시 감압하여 용매를 50% 정도 제거했다. 다시 환류 상태에서 에틸아세테이트를 넣어주며 결정을 떨어트려 식힌 후 여과했다. 이를 컬럼크로마토그래피하여 화합물 22 12.36 g (수율 53 %)를 얻었다. [M+H]=857Compound E 10.0 g (1.0 eq), 2-chloro-4- (4- (naphthalen-1-yl) phenyl) quinazoline 17.05 g (1.1 eq), Pd (t-Bu 3 P) 2 0.14 g (0.01 eq) , NaOtBu 5.23 g (2.0 eq) was added to 250 ml of Xylene and refluxed and stirred. When the reaction was completed after 2 hours, the solvent was removed under reduced pressure. After that, the mixture was completely dissolved in CHCl 3 , washed with water, and then reduced again to remove about 50% of the solvent. Ethyl acetate was added again under reflux, the crystals were dropped, cooled, and filtered. This was subjected to column chromatography, thereby obtaining 12.36 g of Compound 22 (yield 53%). [M + H] = 857
합성예 23Synthesis Example 23
화합물 E 10.0 g (1.0 eq), 2-chloro-4-(4-(naphthalen-1-yl)phenyl)quinazoline 20.31 g (1.1 eq), Pd(t-Bu3P)2 0.14 g (0.01 eq), NaOtBu 5.23 g (2.0 eq)를 Xylene 250ml에 넣어서 환류하여 교반했다. 2 시간 후 반응이 종료되면 감압하여 용매를 제거했다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 다시 감압하여 용매를 50% 정도 제거했다. 다시 환류 상태에서 에틸아세테이트를 넣어주며 결정을 떨어트려 식힌 후 여과했다. 이를 컬럼크로마토그래피하여 화합물 23 15.51 g (수율 59 %)를 얻었다. [M+H]=966Compound E 10.0 g (1.0 eq), 2-chloro-4- (4- (naphthalen-1-yl) phenyl) quinazoline 20.31 g (1.1 eq), Pd (t-Bu 3 P) 2 0.14 g (0.01 eq) , NaOtBu 5.23 g (2.0 eq) was added to 250 ml of Xylene and refluxed and stirred. When the reaction was completed after 2 hours, the solvent was removed under reduced pressure. After that, the mixture was completely dissolved in CHCl 3 , washed with water, and then reduced again to remove about 50% of the solvent. Ethyl acetate was added again under reflux, the crystals were dropped, cooled, and filtered. This was subjected to column chromatography, thereby obtaining 15.51 g of a compound 23 (59% yield). [M + H] = 966
합성예 24Synthesis Example 24
화합물 F 10.0 g (1.0 eq), 2-chloro-4-(4-(naphthalen-1-yl)phenyl)quinazoline 16.57 g (1.1 eq), Pd(t-Bu3P)2 0.14 g (0.01 eq), NaOtBu 5.23 g (2.0 eq)를 Xylene 250ml에 넣어서 환류하여 교반했다. 2 시간 후 반응이 종료되면 감압하여 용매를 제거했다. 이 후 CHCl3에 완전히 녹여 물로 씻어주고 다시 감압하여 용매를 50% 정도 제거했다. 다시 환류 상태에서 에틸아세테이트를 넣어주며 결정을 떨어트려 식힌 후 여과했다. 이를 컬럼크로마토그래피하여 화합물 24 14.64 g (수율 64 %)를 얻었다. [M+H]=841Compound F 10.0 g (1.0 eq), 2-chloro-4- (4- (naphthalen-1-yl) phenyl) quinazoline 16.57 g (1.1 eq), Pd (t-Bu 3 P) 2 0.14 g (0.01 eq) , NaOtBu 5.23 g (2.0 eq) was added to 250 ml of Xylene and refluxed and stirred. When the reaction was completed after 2 hours, the solvent was removed under reduced pressure. After that, the mixture was completely dissolved in CHCl 3 , washed with water, and then reduced again to remove about 50% of the solvent. Ethyl acetate was added again under reflux, the crystals were dropped, cooled, and filtered. This was subjected to column chromatography, thereby obtaining 14.64 g of Compound 24 (Yield 64%). [M + H] = 841
<실험예><Experimental Example>
비교예 1Comparative Example 1
ITO(indium tin oxide)가 1,000Å의 두께로 박막 코팅된 유리 기판을 세제를 녹인 증류수에 넣고 초음파로 세척하였다. 이때, 세제로는 피셔사(Fischer Co.) 제품을 사용하였으며, 증류수로는 밀러포어사(Millipore Co.) 제품의 필터(Filter)로 2차로 걸러진 증류수를 사용하였다. ITO를 30분간 세척한 후 증류수로 2회 반복하여 초음파 세척을 10분간 진행하였다. 증류수 세척이 끝난 후, 이소프로필알콜, 아세톤, 메탄올의 용제로 초음파 세척을 하고 건조시킨 후 플라즈마 세정기로 수송시켰다. 또한, 산소 플라즈마를 이용하여 상기 기판을 5분간 세정한 후 진공 증착기로 기판을 수송시켰다.A glass substrate coated with a thin film coated with ITO (indium tin oxide) at a thickness of 1,000 에 was put in distilled water in which detergent was dissolved and washed with ultrasonic waves. At this time, Fischer Co. product was used as the detergent, and distilled water filtered secondarily by a filter of Millipore Co. was used as the distilled water. After washing the ITO for 30 minutes, ultrasonic cleaning was repeated twice with distilled water for 10 minutes. After washing with distilled water, ultrasonic cleaning was performed with a solvent of isopropyl alcohol, acetone, and methanol, followed by drying and then transported to a plasma cleaner. In addition, the substrate was washed for 5 minutes using oxygen plasma, and then transferred to a vacuum evaporator.
이렇게 준비된 ITO 투명 전극 위에 정공주입층으로 하기 HI-1 화합물을 1150Å의 두께로 형성하되 하기 A-1 화합물을 1.5% 농도로 p-도핑 하였다. 상기 정공주입층 위에 하기 HT-1 화합물을 진공 증착하여 막 두께 800Å 의 정공수송층을 형성하였다. 이어서, 상기 정공수송층 위에 막 두께 150Å으로 하기 EB-1 화합물을 진공 증착하여 전자차단층을 형성하였다. 이어서, 상기 EB-1 증착막 위에 하기 RH-1 화합물과 하기 Dp-7 화합물을 98:2의 중량비로 진공 증착하여 400Å 두께의 적색 발광층을 형성하였다. 상기 발광층 위에 막 두께 30Å으로 하기 HB-1 화합물을 진공 증착하여 정공차단층을 형성하였다. 이어서, 상기 정공차단층 위에 하기 ET-1 화합물과 하기 LiQ 화합물을 2:1의 중량비로 진공 증착하여 300Å의 두께로 전자 주입 및 수송층을 형성하였다. 상기 전자 주입 및 수송층 위에 순차적으로 12Å 두께로 리튬플로라이드(LiF)와 1,000Å 두께로 알루미늄을 증착하여 음극을 형성하였다. The following HI-1 compound was formed as a hole injection layer on the prepared ITO transparent electrode to a thickness of 1150Å, but the following A-1 compound was p-doped at a concentration of 1.5%. The following HT-1 compound was vacuum deposited on the hole injection layer to form a hole transport layer having a thickness of 800 mm 2. Subsequently, the following EB-1 compound was vacuum-deposited to a thickness of 150 mm 2 on the hole transport layer to form an electron blocking layer. Subsequently, the following RH-1 compound and the following Dp-7 compound were vacuum deposited on the EB-1 deposition film at a weight ratio of 98: 2 to form a red light emitting layer having a thickness of 400 Pa. A hole blocking layer was formed by vacuum-depositing the following HB-1 compound with a thickness of 30 Pa on the light emitting layer. Subsequently, the following ET-1 compound and the following LiQ compound were vacuum deposited on the hole blocking layer in a weight ratio of 2: 1 to form an electron injection and transport layer with a thickness of 300 Pa. On the electron injection and transport layer, lithium fluoride (LiF) with a thickness of 12 Å and aluminum with a thickness of 1,000 순차적 were sequentially deposited to form a negative electrode.
상기의 과정에서 유기물의 증착속도는 0.4 내지 0.7Å/sec를 유지하였고, 음극의 리튬플로라이드는 0.3Å/sec, 알루미늄은 2Å/sec의 증착 속도를 유지하였으며, 증착시 진공도는 2 X 10-7 내지 5 X 10-6 torr를 유지하여, 유기 발광 소자를 제작하였다Was maintained at a vapor deposition rate of 0.4 to 0.7Å / sec for organic material in the above process, the lithium fluoride of the cathode was 0.3Å / sec, aluminum is deposited at a rate of 2Å / sec, During the deposition, a vacuum 10 2 X - An organic light emitting device was manufactured by maintaining 7 to 5 X 10 -6 torr.
실시예 1 내지 실시예 24Examples 1 to 24
비교예 1의 유기 발광 소자에서 RH-1 대신 하기 표 1에 기재된 화합물을 사용하는 것을 제외하고는, 상기 비교예 1과 동일한 방법으로 유기 발광 소자를 제조하였다. An organic light emitting diode was manufactured according to the same method as Comparative Example 1 except for using the compound shown in Table 1 instead of RH-1 in the organic light emitting diode of Comparative Example 1.
비교예 2 내지 비교예 21Comparative Examples 2 to 21
비교예 1의 유기 발광 소자에서 RH-1 대신 하기 표 1에 기재된 화합물을 사용하는 것을 제외하고는, 상기 비교예 1과 동일한 방법으로 유기 발광 소자를 제조하였다. An organic light emitting diode was manufactured according to the same method as Comparative Example 1 except for using the compound shown in Table 1 instead of RH-1 in the organic light emitting diode of Comparative Example 1.
상기 실시예 1 내지 실시예 24 및 비교예 1 내지 비교예 21 에서 제조한 유기 발광 소자에 전류를 인가하였을 때, 전압, 효율, 수명을 측정하고 그 결과를 하기 표 1에 나타내었다. T95은 휘도가 초기 휘도(10,000 nit)에서 95%로 감소되는데 소요되는 시간을 의미한다. When current was applied to the organic light emitting devices manufactured in Examples 1 to 24 and Comparative Examples 1 to 21, voltage, efficiency, and life were measured and the results are shown in Table 1 below. T95 means the time required for the luminance to decrease from the initial luminance (10,000 nit) to 95%.
실시예 1 내지 24 및 비교예 1 내지 21에 의해 제작된 유기 발광 소자에 전류를 인가하였을 때, 상기 표 1의 결과를 얻었다. 상기 비교예 1의 적색 유기 발광 소자는 종래 널리 사용되고 있는 물질을 사용하였다. 비교예 2 내지 21은 RH-1 대신 C-1 내지 C-20을 사용하여 유기 발광 소자를 제조하였다. 상기 표 1의 결과를 보면 본 발명의 화합물이 적색 발광층의 호스트로 사용 했을 때 비교예 물질에 비해서 구동전압이 크게는 30% 가까이 낮아졌으며, 효율 측면에서도 30% 이상 상승을 한 것으로 보아 호스트에서 적색 도펀트로의 에너지 전달이 잘 이뤄진다는 것을 알 수 있었다. 또한 높은 효율을 유지하면서도 수명 특성을 크게 개선 시킬 수 있는 것을 알 수 있었다. 이것은 결국 비교예 화합물 보다 본 발명의 화합물이 전자와 정공에 대한 안정도가 높으며 OLED Red 소자내에서 전자와 정공 이동의 균형이 잘 맞기 때문이라 판단 할 수 있다. 결론적으로 본 발명의 화합물을 적색 발광층의 호스트로 사용하였을 때 유기 발광 소자의 구동전압, 발광 효율 및 수명 특성을 개선할 수 있다는 것을 확인할 수 있다.When current was applied to the organic light emitting devices manufactured by Examples 1 to 24 and Comparative Examples 1 to 21, the results of Table 1 were obtained. In the red organic light emitting device of Comparative Example 1, a material widely used in the related art was used. In Comparative Examples 2 to 21, organic light emitting devices were manufactured using C-1 to C-20 instead of RH-1. Looking at the results of Table 1, when the compound of the present invention was used as a host for a red light-emitting layer, the driving voltage was significantly lower than 30% compared to the comparative example material, and the efficiency was also increased by 30% or more, so that the host was red. It was found that the energy transfer to the dopant was well achieved. In addition, it was found that while maintaining high efficiency, the lifespan characteristics could be greatly improved. It can be judged that this is because the compound of the present invention has higher stability to electrons and holes than the comparative example compound, and the balance of electron and hole movement in the OLED Red device is well balanced. In conclusion, it can be seen that when the compound of the present invention is used as a host of a red light emitting layer, it is possible to improve driving voltage, light emission efficiency, and life characteristics of an organic light emitting device.
101: 기판
102: 양극
103: 정공 주입층
104: 정공 수송층
105: 전자 차단층
106: 발광층
107: 정공 차단층
108: 전자 주입 및 수송층
110: 음극101: substrate
102: anode
103: hole injection layer
104: hole transport layer
105: electron blocking layer
106: emitting layer
107: hole blocking layer
108: electron injection and transport layer
110: cathode
Claims (13)
상기 화학식 1에 있어서,
*은 상기 화학식 1-1의 *와 축합하는 위치이며,
R1 및 R2는 서로 동일하거나 상이하고, 각각 독립적으로 수소; 또는 중수소이고,
a1은 0 내지 8의 정수이고, a1이 2 이상인 경우, R1은 서로 동일하거나 상이하고,
a2은 0 내지 2의 정수이고, a2이 2인 경우, R2은 서로 동일하거나 상이하고,
상기 화학식 1-1에 있어서,
X1 내지 X3는 서로 동일하거나 상이하고, 각각 독립적으로 N; 또는 CH이고, X1 내지 X3 중 2 이상은 N이고,
Ar1 및 Ar2는 서로 동일하거나 상이하고, 각각 독립적으로 치환 또는 비치환된 아릴기; 또는 치환 또는 비치환된 헤테로아릴기이고,
L은 직접결합; 치환 또는 비치환된 아릴렌기; 또는 치환 또는 비치환된 헤테로아릴렌기이고,
R3는 수소; 또는 중수소이고,
a3은 0 내지 6의 정수이고, a3이 2 이상인 경우, R3은 서로 동일하거나 상이하다.Compound represented by the formula (1):
In Chemical Formula 1,
* Is a position condensed with * in Chemical Formula 1-1,
R1 and R2 are the same as or different from each other, and each independently hydrogen; Or deuterium,
a1 is an integer from 0 to 8, and when a1 is 2 or more, R1 is the same as or different from each other,
a2 is an integer from 0 to 2, and when a2 is 2, R2 is the same or different from each other,
In Chemical Formula 1-1,
X1 to X3 are the same as or different from each other, and each independently N; Or CH, and 2 or more of X1 to X3 are N,
Ar1 and Ar2 are the same as or different from each other, and each independently a substituted or unsubstituted aryl group; Or a substituted or unsubstituted heteroaryl group,
L is a direct bond; A substituted or unsubstituted arylene group; Or a substituted or unsubstituted heteroarylene group,
R3 is hydrogen; Or deuterium,
a3 is an integer of 0 to 6, and when a3 is 2 or more, R3 is the same or different from each other.
[화학식 3-1]
[화학식 3-2]
상기 화학식 3-1 및 3-2에 있어서,
X1 내지 X3, Ar1, Ar2, L, R1 내지 R3 및 a1 내지 a3의 정의는 화학식 1에서 정의한 바와 같다.The method according to claim 1, wherein Formula 1 is a compound represented by the following Formula 3-1 or 3-2:
[Formula 3-1]
[Formula 3-2]
In Chemical Formulas 3-1 and 3-2,
X1 to X3, Ar1, Ar2, L, R1 to R3 and a1 to a3 are defined as defined in Chemical Formula 1.
[화학식 2-1]
[화학식 2-2]
[화학식 2-3]
[화학식 2-4]
[화학식 2-5]
[화학식 2-6]
상기 화학식 2-1 내지 2-6에 있어서,
X1 내지 X3, Ar1, Ar2, L, R1 내지 R3 및 a1 내지 a3의 정의는 화학식 1에서 정의한 바와 같다.Chemical Formula 1 is a compound represented by any one of the following Chemical Formulas 2-1 to 2-6:
[Formula 2-1]
[Formula 2-2]
[Formula 2-3]
[Formula 2-4]
[Formula 2-5]
[Formula 2-6]
In Chemical Formulas 2-1 to 2-6,
X1 to X3, Ar1, Ar2, L, R1 to R3 and a1 to a3 are defined as defined in Chemical Formula 1.
L은 직접결합; 중수소 또는 니트릴기로 치환 또는 비치환된 탄소수 6 내지 20의 아릴렌기인 화합물.The method according to claim 1,
L is a direct bond; A compound having an arylene group having 6 to 20 carbon atoms unsubstituted or substituted with deuterium or nitrile groups.
X1 내지 X3는 N인 화합물.The method according to claim 1,
X1 to X3 are N compounds.
Ar1 및 Ar2는 서로 동일하거나 상이하고, 각각 독립적으로 탄소수 6 내지 20의 아릴기; 또는 탄소수 2 내지 20의 헤테로아릴기이고, 상기 아릴기 또는 헤테로아릴기는 중수소, 탄소수 1 내지 10의 알킬기, 탄소수 6 내지 20의 아릴기 또는 탄소수 2 내지 20의 헤테로아릴기로 치환되거나 비치환되는 것인 화합물.The method according to claim 1,
Ar1 and Ar2 are the same as or different from each other, and each independently an aryl group having 6 to 20 carbon atoms; Or a heteroaryl group having 2 to 20 carbon atoms, the aryl group or heteroaryl group being substituted or unsubstituted with deuterium, an alkyl group having 1 to 10 carbon atoms, an aryl group having 6 to 20 carbon atoms, or a heteroaryl group having 2 to 20 carbon atoms. compound.
.The compound according to claim 1, wherein the compound represented by Formula 1 is any one selected from the following compounds:
.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020180133636 | 2018-11-02 | ||
| KR20180133636 | 2018-11-02 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20200050893A true KR20200050893A (en) | 2020-05-12 |
| KR102312476B1 KR102312476B1 (en) | 2021-10-14 |
Family
ID=70461854
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020190138726A Active KR102312476B1 (en) | 2018-11-02 | 2019-11-01 | Compound and organic light emitting device comprising the same |
Country Status (3)
| Country | Link |
|---|---|
| KR (1) | KR102312476B1 (en) |
| CN (1) | CN112334463B (en) |
| WO (1) | WO2020091521A1 (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2021527635A (en) * | 2018-07-13 | 2021-10-14 | エルジー・ケム・リミテッド | Heterocyclic compounds and organic light emitting devices containing them |
| WO2022025021A1 (en) * | 2020-07-29 | 2022-02-03 | 出光興産株式会社 | Organic electroluminescent element, organic electroluminescent light-emitting device, and electronic equipment |
| JP2023544188A (en) * | 2020-10-09 | 2023-10-20 | 三星ディスプレイ株式會社 | Organic molecules for optoelectronic devices |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20200023984A (en) * | 2018-08-27 | 2020-03-06 | 삼성전자주식회사 | Heterocyclic compound and organic light emitting device including the same |
| KR102031300B1 (en) * | 2018-12-21 | 2019-10-11 | 삼성에스디아이 주식회사 | Compound for organic optoelectronic device, composition for organic optoelectronic device and organic optoelectronic device and display device |
| CN114369088B (en) * | 2020-10-15 | 2024-04-23 | 江苏三月科技股份有限公司 | Compound containing triazine structure and application thereof |
| CN112300162B (en) | 2020-10-30 | 2022-04-05 | 武汉天马微电子有限公司 | Compound, electron transport material, display panel, and display device |
| CN114685484B (en) * | 2020-12-28 | 2023-09-01 | 宁波卢米蓝新材料有限公司 | Organic electroluminescent compound and organic electroluminescent device comprising same |
| WO2022178442A1 (en) | 2021-02-19 | 2022-08-25 | Advanced Battery Concepts, LLC | Method for forming a reusable battery assembly |
| CN115636818A (en) * | 2021-07-20 | 2023-01-24 | 北京鼎材科技有限公司 | Organic compound and application thereof |
| CN113512043A (en) * | 2021-08-17 | 2021-10-19 | 浙江华显光电科技有限公司 | Organic compound, preparation, organic light-emitting device, and display or lighting device |
| WO2024111223A1 (en) * | 2022-11-22 | 2024-05-30 | 株式会社Kyulux | Compound, light-emitting material and light-emitting element |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2003012890A2 (en) | 2001-07-20 | 2003-02-13 | Novaled Gmbh | Light emitting component with organic layers |
| KR20120072784A (en) * | 2010-12-24 | 2012-07-04 | 에스에프씨 주식회사 | Heterocyclic compounds and organic light-emitting diode including the same |
| WO2013009095A1 (en) * | 2011-07-11 | 2013-01-17 | 주식회사 두산 | Triphenylene-based compound and organic electroluminescent diode using same |
| WO2016105036A1 (en) * | 2014-12-22 | 2016-06-30 | 주식회사 두산 | Organic electroluminescent element |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20120038056A (en) * | 2010-10-13 | 2012-04-23 | 롬엔드하스전자재료코리아유한회사 | Novel compounds for organic electronic material and organic electroluminescent device using the same |
| KR102360228B1 (en) * | 2014-10-22 | 2022-02-09 | 솔루스첨단소재 주식회사 | Organic electro luminescence device |
| CN108137558B (en) * | 2016-01-07 | 2021-04-16 | 广州华睿光电材料有限公司 | Carbazole derivatives, polymers, mixtures, compositions, organic electronic devices and their applications |
-
2019
- 2019-11-01 WO PCT/KR2019/014745 patent/WO2020091521A1/en not_active Ceased
- 2019-11-01 KR KR1020190138726A patent/KR102312476B1/en active Active
- 2019-11-01 CN CN201980043253.8A patent/CN112334463B/en active Active
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2003012890A2 (en) | 2001-07-20 | 2003-02-13 | Novaled Gmbh | Light emitting component with organic layers |
| KR20120072784A (en) * | 2010-12-24 | 2012-07-04 | 에스에프씨 주식회사 | Heterocyclic compounds and organic light-emitting diode including the same |
| WO2013009095A1 (en) * | 2011-07-11 | 2013-01-17 | 주식회사 두산 | Triphenylene-based compound and organic electroluminescent diode using same |
| WO2016105036A1 (en) * | 2014-12-22 | 2016-06-30 | 주식회사 두산 | Organic electroluminescent element |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2021527635A (en) * | 2018-07-13 | 2021-10-14 | エルジー・ケム・リミテッド | Heterocyclic compounds and organic light emitting devices containing them |
| WO2022025021A1 (en) * | 2020-07-29 | 2022-02-03 | 出光興産株式会社 | Organic electroluminescent element, organic electroluminescent light-emitting device, and electronic equipment |
| JP2023544188A (en) * | 2020-10-09 | 2023-10-20 | 三星ディスプレイ株式會社 | Organic molecules for optoelectronic devices |
Also Published As
| Publication number | Publication date |
|---|---|
| CN112334463A (en) | 2021-02-05 |
| CN112334463B (en) | 2023-09-01 |
| KR102312476B1 (en) | 2021-10-14 |
| WO2020091521A1 (en) | 2020-05-07 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR102312476B1 (en) | Compound and organic light emitting device comprising the same | |
| KR102119352B1 (en) | Fused cyclic compound including nitrogen and organic light emitting device using the same | |
| KR102209929B1 (en) | Organic light emitting device | |
| KR102100008B1 (en) | Anthracene based compound and organic light emitting device comprising the same | |
| KR102486517B1 (en) | Novel compound and organic light emitting device comprising the same | |
| KR20200038872A (en) | Hetero cyclic compound and organic light emitting device comprising the same | |
| KR102466029B1 (en) | Anthracene compound and organic light emitting device comprising same | |
| KR101920143B1 (en) | Heterocyclic compound and organic light emitting device comprising the same | |
| KR20200096173A (en) | Compound and organic light emitting device comprising the same | |
| KR20200037734A (en) | Heterocyclic compound and organic light emitting device comprising the same | |
| KR102312477B1 (en) | Compound and organic light emitting device comprising the same | |
| KR102428976B1 (en) | Compound and organic light emitting device comprising same | |
| KR102780445B1 (en) | Novel compound and organic light emitting device comprising the same | |
| KR102209924B1 (en) | Compound and organic light emitting device comprising same | |
| KR20220066648A (en) | Anthracene compound and organic light emitting device comprising same | |
| KR102123742B1 (en) | Compound and organic light emitting device comprising the same | |
| KR102274880B1 (en) | Compound and organic electronic device comprising the same | |
| KR20200088234A (en) | Heterocyclic compound and organic light emitting device comprising same | |
| KR102564847B1 (en) | Novel compound and organic light emitting device comprising the same | |
| KR20190103997A (en) | Heterocyclic compound and organic light emitting device comprising the same | |
| KR102312478B1 (en) | Compound and organic light emitting device comprising the same | |
| KR20190000832A (en) | Compound and organic light emitting device comprising the same | |
| KR20200088235A (en) | Organic light emitting device | |
| KR20200021424A (en) | Organic light emitting device | |
| KR20190141582A (en) | Compound and organic light emitting device comprising same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0109 | Patent application |
Patent event code: PA01091R01D Comment text: Patent Application Patent event date: 20191101 |
|
| PA0201 | Request for examination | ||
| PG1501 | Laying open of application | ||
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20210427 Patent event code: PE09021S01D |
|
| E701 | Decision to grant or registration of patent right | ||
| PE0701 | Decision of registration |
Patent event code: PE07011S01D Comment text: Decision to Grant Registration Patent event date: 20210917 |
|
| GRNT | Written decision to grant | ||
| PR0701 | Registration of establishment |
Comment text: Registration of Establishment Patent event date: 20211007 Patent event code: PR07011E01D |
|
| PR1002 | Payment of registration fee |
Payment date: 20211007 End annual number: 3 Start annual number: 1 |
|
| PG1601 | Publication of registration |