KR20140019055A - Pyrazolopyridine or indazole derivatives as protein kinase inhibitors - Google Patents
Pyrazolopyridine or indazole derivatives as protein kinase inhibitors Download PDFInfo
- Publication number
- KR20140019055A KR20140019055A KR1020120077013A KR20120077013A KR20140019055A KR 20140019055 A KR20140019055 A KR 20140019055A KR 1020120077013 A KR1020120077013 A KR 1020120077013A KR 20120077013 A KR20120077013 A KR 20120077013A KR 20140019055 A KR20140019055 A KR 20140019055A
- Authority
- KR
- South Korea
- Prior art keywords
- fluorophenyl
- sulfonyl
- compound
- group
- pyrazolo
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 0 CC(C)(C)C(Nc(c1c2)n[n](*)c1ncc2S(c1cc(F)ccc1)(=O)=O)=O Chemical compound CC(C)(C)C(Nc(c1c2)n[n](*)c1ncc2S(c1cc(F)ccc1)(=O)=O)=O 0.000 description 3
- CXGLOCBJICXMCK-UHFFFAOYSA-N CCCCCC(Nc1n[nH]c(nc2)c1cc2S(c1cc(F)ccc1)(=O)=O)=O Chemical compound CCCCCC(Nc1n[nH]c(nc2)c1cc2S(c1cc(F)ccc1)(=O)=O)=O CXGLOCBJICXMCK-UHFFFAOYSA-N 0.000 description 1
- YOSVQMBYJMICGH-UHFFFAOYSA-N CC[O](C)C(c1n[nH]c(cc2)c1cc2Br)=O Chemical compound CC[O](C)C(c1n[nH]c(cc2)c1cc2Br)=O YOSVQMBYJMICGH-UHFFFAOYSA-N 0.000 description 1
- UFNTYZKSXHAHAW-UHFFFAOYSA-M C[TlH][n](c(c1c2)ncc2S(c2cc(F)ccc2)(=O)=O)nc1NC(c(cc1)ccc1N1CCOCC1)=O Chemical compound C[TlH][n](c(c1c2)ncc2S(c2cc(F)ccc2)(=O)=O)nc1NC(c(cc1)ccc1N1CCOCC1)=O UFNTYZKSXHAHAW-UHFFFAOYSA-M 0.000 description 1
- LYIWIIMIGOHWKX-UHFFFAOYSA-N N#Cc1cc(S(c2cc(F)ccc2)(=O)=O)cnc1Cl Chemical compound N#Cc1cc(S(c2cc(F)ccc2)(=O)=O)cnc1Cl LYIWIIMIGOHWKX-UHFFFAOYSA-N 0.000 description 1
- ONGWZKWZQPAKPA-UHFFFAOYSA-N O=C(Nc1n[nH]c(nc2)c1cc2S(c1cccc(F)c1)(=O)=O)I Chemical compound O=C(Nc1n[nH]c(nc2)c1cc2S(c1cccc(F)c1)(=O)=O)I ONGWZKWZQPAKPA-UHFFFAOYSA-N 0.000 description 1
- BIOMJQNWMMQZLY-UHFFFAOYSA-N O=C(Nc1n[n](C(c2ccccc2)(c2ccccc2)c2ccccc2)c(nc2)c1cc2S(c1cc(F)ccc1)(=O)=O)I Chemical compound O=C(Nc1n[n](C(c2ccccc2)(c2ccccc2)c2ccccc2)c(nc2)c1cc2S(c1cc(F)ccc1)(=O)=O)I BIOMJQNWMMQZLY-UHFFFAOYSA-N 0.000 description 1
- HOYZVAFPGKAPRV-UHFFFAOYSA-N O=C(c(cc1)ccc1F)Nc1n[nH]c(nc2)c1cc2S(c1cc(F)ccc1)(=O)=O Chemical compound O=C(c(cc1)ccc1F)Nc1n[nH]c(nc2)c1cc2S(c1cc(F)ccc1)(=O)=O HOYZVAFPGKAPRV-UHFFFAOYSA-N 0.000 description 1
- DBDYJHQQCLBBET-UHFFFAOYSA-M O=C(c1ccccc1)Nc(c1cc(S(c2cc(F)ccc2)(=O)=O)cnc11)n[n]1[Tl] Chemical compound O=C(c1ccccc1)Nc(c1cc(S(c2cc(F)ccc2)(=O)=O)cnc11)n[n]1[Tl] DBDYJHQQCLBBET-UHFFFAOYSA-M 0.000 description 1
- JTYXMDXGAZQFEQ-UHFFFAOYSA-N O=C(c1n[nH]c(cc2)c1cc2S(c1cccc(F)c1)(=O)=O)Nc(cc1)ccc1N1CCOCC1 Chemical compound O=C(c1n[nH]c(cc2)c1cc2S(c1cccc(F)c1)(=O)=O)Nc(cc1)ccc1N1CCOCC1 JTYXMDXGAZQFEQ-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/4353—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom ortho- or peri-condensed with heterocyclic ring systems
- A61K31/437—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom ortho- or peri-condensed with heterocyclic ring systems the heterocyclic ring system containing a five-membered ring having nitrogen as a ring hetero atom, e.g. indolizine, beta-carboline
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D231/00—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings
- C07D231/54—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings condensed with carbocyclic rings or ring systems
- C07D231/56—Benzopyrazoles; Hydrogenated benzopyrazoles
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings
- C07D409/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings linked by a chain containing hetero atoms as chain links
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
본 발명은 단백질 키나아제에 대한 저해활성을 가지는 신규 피라졸로피리딘 또는 인다졸 유도체 또는 이의 약학적으로 허용 가능한 염에 관한 것이다. 본 발명의 화합물은 단백질 키나아제 예를 들면 ABL, ACK1, ALK, Aurora A, Aurora B, Aurora C, BLK, BMX/ETK, BRSK1, BTK, c-Src, CAMKK, CDK1, CDK2, CDK5, CLK, DDR, DYRK1B, EPHA, EPHB, FAK/PTK2, FER, FES/FPS, FGFR, FGR, FLT3, FLT4/VEGFR3, FMS, FRK/PTK5, FYN, GSK3b, HCK, IGF1R, IR, IRAK1, IRR/INSRR, ITK, JAK2, KHS/MAP4K5, LCK, LYN, PHKg, PLK4/SAK, PYK2, RET, ROS/ROS1, TIE2/TEK, TRK, TXK, TYK, YES/YES1 등에 대하여 우수한 저해활성을 가지므로 각종 암질환의 치료 및 예방을 위한 약물로 유용하다.The present invention relates to novel pyrazolopyridine or indazole derivatives or pharmaceutically acceptable salts thereof having inhibitory activity against protein kinases. Compounds of the present invention are protein kinases such as ABL, ACK1, ALK, Aurora A, Aurora B, Aurora C, BLK, BMX / ETK, BRSK1, BTK, c-Src, CAMKK, CDK1, CDK2, CDK5, CLK, DDR , DYRK1B, EPHA, EPHB, FAK / PTK2, FER, FES / FPS, FGFR, FGR, FLT3, FLT4 / VEGFR3, FMS, FRK / PTK5, FYN, GSK3b, HCK, IGF1R, IR, IRAK1, IRR / INSRR, ITK , JAK2, KHS / MAP4K5, LCK, LYN, PHKg, PLK4 / SAK, PYK2, RET, ROS / ROS1, TIE2 / TEK, TRK, TXK, TYK, YES / YES1, etc. It is useful as a drug for treatment and prevention.
Description
본 발명은 단백질 키나아제에 대한 저해활성을 가지는 신규 피라졸로피리딘 유도체 또는 인다졸 유도체 또는 이의 약학적으로 허용 가능한 염에 관한 것으로, 본 발명의 화합물은 단백질 키나아제에 대한 저해활성을 가지므로 각종 암질환의 치료 및 예방을 위한 약물로 유용하다.
The present invention relates to novel pyrazolopyridine derivatives or indazole derivatives or pharmaceutically acceptable salts thereof having inhibitory activity against protein kinases. Since the compounds of the present invention have inhibitory activity against protein kinases, It is useful as a drug for treatment and prevention.
단백질 키나아제는 단백질의 티로신, 세린 및 트레오닌 잔기에 위치하는 하이드록시 그룹의 인산화를 촉매하는 효소로서, 세포의 성장, 분화 및 증식을 유발하는 성장 인자 신호 전달에 중요한 역할을 담당하고 있다.Protein kinases are enzymes that catalyze the phosphorylation of hydroxy groups located at tyrosine, serine and threonine residues of proteins and play an important role in the growth factor signal transduction that causes cell growth, differentiation and proliferation.
생체의 항상성 유지를 위해서 생체 내 신호 전달 체계는 켜짐과 꺼짐이 원활하게 균형을 이루어야 한다. 그러나 특정 단백질 키나아제의 돌연변이나 과발현은 정상적인 세포 내 신호 전달체계를 붕괴시켜서 (주로 생체 내 신호 전달이 계속 되는 상태) 암, 염증, 대사성 질환, 뇌질환 등 다양한 질병을 유발한다. In order to maintain the homeostasis of the living body, the signaling system in vivo should be smoothly turned on and off. However, mutation or overexpression of certain protein kinases disrupts normal cellular signaling systems (mainly in vivo), leading to a variety of diseases, including cancer, inflammation, metabolic diseases, and brain diseases.
인간 단백질 키나아제는 인간 전체 유전자의 약 1.7%에 해당하는 518 종이 존재하는 것으로 추정되며, 크게 티로신 단백질 키나아제 (90 종 이상)와 세린/트레오닌 단백질 키나아제로 양분된다. 티로신 단백질 키나아제는 20개의 아과로 구분되는 58종의 수용체 티로신 키나아제와 10개의 아과로 구분되는 32종의 세포질/비수용체로 나눌 수 있다. 수용체 티로신 키나아제는 세포 표면에는 성장 인자를 수용할 수 있는 도메인과 세포질에는 티로신 잔기를 인산화 할 수 있는 활성부위를 갖고 있다. 성장 인자가 수용체 티로신 키나아제 세포 표면의 성장인자 수용체 자리에 결합되면, 수용체 티로신 키나아제는 중합체를 형성하고 세포질의 티로신 잔기는 자가인산화 된다. 그리고 하위 계열 단백질들의 순차적인 인산화를 통해 신호 전달이 핵 내로 진행되어서 종국에는 암을 유발하는 전사인자들이 과발현된다. Human protein kinases are estimated to be present in 518 species, corresponding to about 1.7% of the total human gene, and are largely divided into tyrosine protein kinases (more than 90 species) and serine / threonine protein kinases. Tyrosine protein kinases can be divided into 58 receptor tyrosine kinases divided into 20 subfamily and 32 cytoplasmic / non-receptors separated into 10 subfamily. Receptor tyrosine kinases have a domain that can accommodate growth factors on the cell surface and an active site that can phosphorylate tyrosine residues on the cytoplasm. When the growth factor is bound to the growth factor receptor site on the receptor tyrosine kinase cell surface, the receptor tyrosine kinase forms a polymer and the tyrosine residues of the cytoplasm are autophosphorylated. Signal transduction proceeds into the nucleus through sequential phosphorylation of the lower family proteins, resulting in overexpression of cancer-causing transcription factors.
혈관 내피 세포 성장 인자 수용체 (Vascular Endothelial Growth Factors Receptors, VEGFR)는 수용체 티로신 키나아제 (Receptor Tyrosine Kinase, RTK)로서 신생혈관생성 (angiogenesis)을 위한 중요한 조절인자이다. 혈관, 림프관의 발생과 항상성 유지에 관여하며 신경세포에도 중요한 효과를 가진다. VEGF는 저산소 상태 및 TGF, 인터루킨, PDGF와 같은 세포 성장 인자들의 자극에 의해 혈관 내피 세포, 조혈 세포, 기질 (stromal) 세포에서 주로 생성된다. VEGF는 VEGF 수용체(VEGFR)-1, -2, -3에 결합하고, 각각의 VEGF isoform은 특정 수용체에 결합하여 수용체의 동형 혹은 이형 접합체 형성을 유도한 뒤 각각의 신호전달체계를 활성화시킨다. VEGFR의 signal specificity는 뉴로피린(neurophilin), 헤파란설페이트(heparan sulfate), 인테그린(integrin), 카드헤린(cadherin) 등과 같은 보조수용체(coreceptor)에 의해 보다 더 미세하게 조절된다.Vascular Endothelial Growth Factors Receptors (VEGFRs) are receptor tyrosine kinases (RTKs) and are important regulators for angiogenesis. It is involved in the development and maintenance of blood vessels and lymphatic vessels, and has an important effect on nerve cells. VEGF is produced mainly in vascular endothelial cells, hematopoietic cells, and stromal cells by hypoxia and stimulation of cell growth factors such as TGF, interleukin, PDGF. VEGF binds to VEGF receptors (VEGFR) -1, -2, and -3, and each VEGF isoform binds to a specific receptor, inducing the formation of a homozygous or heterozygous receptor, and then activates each signaling system. Signal specificity of VEGFR is more finely regulated by coreceptors such as neurophilin, heparan sulfate, integrin, cadherin and the like.
VEGF의 생물학적 기능은 type Ⅲ RTK, VEGFR-1 (Flt-1), VEGFR-2 (KDR/Flk-1), VEGFR-3 (Flt-4)을 통해 매개된다. EGFR은 Fms, Kit, PDGFR과 밀접하게 관련 되어 있고, VEGF는 각각 특정 수용체에 결합하는데, VEGF-A는 VEGFR-1, -2 및 수용체 이형 중합체와 결합하는 반면, VEGF-C는 VEGF-2, -3에 결합한다. 또한, PIGF 와 VEGF-B는 VEGFR-1에 배타적으로, VEGF-E는 오직 VEGFR-2와 상호 작용한다. VEGF-F variant는 VEGFR-1 혹은 -2와 상호 작용한다. VEGF-A, - B, PIGF는 혈관 형성에 우선적으로 필요한 반면, VEGF-C, -D는 림프관 형성에 필수적이다. 신생혈관은 종양에 영양분과 산소를 공급하며 암세포 전이의 통로를 제공하여 그 증식과 전이에 필수적이다. 혈관형성은 정상적인 경우 생체 내에서 혈관생성 촉진물질 (angiogenic stimulator)과 혈관생성 억제물질 (angiogenic suppressor)의 상호조절에 의해 균형을 이루고 있으나 암세포에서와 같이 그러한 균형이 깨진 경우 혈관내피생장에 가장 큰 영향을 미치는 성장인자 VEGF (Vascular Endothelial Growth Factor)에 의해 그 수용체인 VEGFR이 활성화된다. 여러 작용 기전 중에 저분자 합성물질을 이용한 이러한 VEGF의 수용체인 티로신 키나아제를 억제하는 저해제가 다양하게 연구 개발되고 있으며 이들 대부분은 고형암 (solid tumor)에 공통적으로 사용될 수 있는 가능성과 암세포에서만 활성화된 신생혈관형성을 억제하므로 비교적 적은 부작용으로 효과적인 약효를 기대할 수 있는 장점을 가지고 있다. Biological function of VEGF is mediated through type III RTK, VEGFR-1 (Flt-1), VEGFR-2 (KDR / Flk-1), VEGFR-3 (Flt-4). EGFR is closely related to Fms, Kit and PDGFR, and VEGF binds to specific receptors, respectively, while VEGF-A binds to VEGFR-1, -2 and receptor heteropolymers, while VEGF-C binds to VEGF-2, Bind to -3. Also, PIGF and VEGF-B are exclusive to VEGFR-1, and VEGF-E only interacts with VEGFR-2. The VEGF-F variant interacts with VEGFR-1 or -2. VEGF-A, -B, and PIGF are primarily required for blood vessel formation, while VEGF-C, -D are essential for lymphatic vessel formation. New blood vessels supply nutrients and oxygen to tumors and provide a pathway for cancer cell metastasis, essential for their proliferation and metastasis. Angiogenesis is normally balanced by the mutual regulation of angiogenic stimulators and angiogenic suppressors in vivo, but the greatest impact on vascular endothelial growth when such balances are broken, such as in cancer cells. The receptor, VEGFR, is activated by the Vascular Endothelial Growth Factor (VEGF). Among various mechanisms of action, various inhibitors that inhibit the tyrosine kinase, a receptor of VEGF, using a low molecular weight compound have been researched and developed. Most of them have the potential to be commonly used in solid tumors and neovascularization activated only in cancer cells. Because it suppresses the relatively low side effects have the advantage that you can expect effective efficacy.
ALK (ANAPLASTIC LYMPHOMA KINASE)는 수용체 티로신 키나아제의 일종이다. 1620개의 아미노산으로 구성된 ALK는 약 180kDa이다. ALK와 NPM (nucleophosmin)이 융합된 ALK-NPM은 종양유전자로서 ALCL (anaplastic large cell lymphoma) 질환 유발인자이다. 또한 ALK와 EML4 (echinoderm microtubule-associated proteinALK (ANAPLASTIC LYMPHOMA KINASE) is a type of receptor tyrosine kinase. The ALK, consisting of 1620 amino acids, is about 180 kDa. ALK-NPM, a fusion of ALK and nucleophosmin (NPM), is an oncogene and a trigger for anaplastic large cell lymphoma (ALCL) disease. In addition, ALK and EML4 (echinoderm microtubule-associated proteins
like 4)가 융합된 EML4-ALK도 종양유전자로서, NSCLC (non-small cell lung cancer)질환 유발인자이다. Crizotinib (PF-02341066)는 ALK 수용체 티로신 키나아제의 저해제로서 NSCLC (non-small cell lung cancer)질환치료제로 승인을 받았다. Crizotinib (PF-02341066)는 ALCL (anaplastic large cell lymphoma) 질환 치료 목적으로 임상시험 중이다.Like 4) fused EML4-ALK is also a tumor gene, NSCLC (non-small cell lung cancer) disease causing factor. Crizotinib (PF-02341066) has been approved as a non-small cell lung cancer (NSCLC) treatment as an inhibitor of ALK receptor tyrosine kinase. Crizotinib (PF-02341066) is under clinical trial for the treatment of anaplastic large cell lymphoma (ALCL) disease.
Tie2는 수용체 티로신 키나아제의 일종인데, 신생혈관생성과 혈관배치 (vasculature)과 연관이 깊다. Tie2의 도메인 구조는 모든 척추 동물에 매우 높게 보존되어 있다. Tie2의 리간드는 엔지오포에틴 (angiopoietins, Ang)이다. Ang2는 Tie2의 자가인산화를 유발하지 않고, Ang1이 유발하는 Tie2의 활성화를 방해한다. 내피세포에서 Ang2에 의한 Tie2의 활성화는 PI3K-Akt의 활성화를 유발한다. Tie2의 주 신호전달 체계인 미토젠 활성화 단백질 키나아제 (mitogen-activated protein kinase, MAPK) 신호전달 경로에서, 어댑터 단백질인 GRB2와 단백질 티로신 포스파타제인 SHP2는 Tie2 수용체 티로신 키나아제의 자가인산화를 통한 이합체화 과정에서 중요한 역할을 한다. Ang/Tie2와 혈관 내피 세포 성장 인자 (VEGF) 신호전달 경로는 암세포의 신생혈관생성에 중요한 역할을 한다. Tie2는 혈관내피세포에 발현하는데, 특히 암세포가 침윤하는 자리에서 발현이 극대화된다. Tie2의 과발현은 유방암 (Peters et al., 1998)에서 확인되었으며, 동시에 자궁암, 간암, 뇌암 등에서도 관찰된다. Tie2 is a type of receptor tyrosine kinase that is highly associated with angiogenesis and vasculature. The domain structure of Tie2 is very highly conserved in all vertebrates. Ligands of Tie2 are angiopoietins (Ang). Ang2 does not induce autophosphorylation of Tie2, but interferes with the activation of Tie2 induced by Ang1. Activation of Tie2 by Ang2 in endothelial cells leads to the activation of PI3K-Akt. In the mitogen-activated protein kinase (MAPK) signaling pathway, Tie2's main signaling system, the adapter protein GRB2 and protein tyrosine phosphatase SHP2 are involved in the dimerization process through autophosphorylation of Tie2 receptor tyrosine kinase. Plays an important role. Ang / Tie2 and vascular endothelial growth factor (VEGF) signaling pathways play an important role in the neovascularization of cancer cells. Tie2 is expressed in vascular endothelial cells, especially in the place where cancer cells invade. Overexpression of Tie2 has been identified in breast cancer (Peters et al., 1998), and at the same time in uterine cancer, liver cancer and brain cancer.
현재까지 피라졸로피리딘 또는 인다졸 구조를 모체로 하는 여러 화합물들이 합성된 바는 있으나, 본 발명에서와 같이 모핵의 C3 위치에는 -C(O)NH- 또는 -NHC(O)-의 아미드 그룹이 도입되고, 모핵의 C5 위치에는 설파이드(SR1), 설파이닐(SOR1), 또는 설포닐(SO2R1) 그룹이 동시에 도입된 화합물이 합성된 바는 없으며, 또한 이들 화합물에 대한 단백질 키나아제의 저해활성을 확인하여 종양 치료 및 예방제로 사용할 수 있음에 대해서는 현재까지 어떠한 문헌에도 발표되어 있지 않고 있다.
To date, several compounds having a parent structure of pyrazolopyridine or indazole have been synthesized. However, in the C3 position of the parent nucleus, an amide group of -C (O) NH- or -NHC (O)- No compound was introduced at the same time that the sulfide (SR 1 ), sulfinyl (SOR 1 ), or sulfonyl (SO 2 R 1 ) group was introduced at the C5 position of the parent nucleus, and also the protein kinase for these compounds. It has not been published in any literature to confirm its inhibitory activity and can be used as a tumor treatment and prevention agent.
본 발명의 목적은 신규 피라졸로피리딘 유도체, 인다졸 유도체 또는 이의 약학적으로 허용 가능한 염을 제공하는 것이다.It is an object of the present invention to provide novel pyrazolopyridine derivatives, indazole derivatives or pharmaceutically acceptable salts thereof.
본 발명의 다른 목적은 상기한 신규 화합물 또는 이의 약학적으로 허용 가능한 염을 유효성분으로 함유하는 암질환의 예방 및 치료용 약학적 조성물을 제공하는 것이다.
Another object of the present invention to provide a pharmaceutical composition for the prevention and treatment of cancer diseases containing the novel compound or a pharmaceutically acceptable salt thereof as an active ingredient.
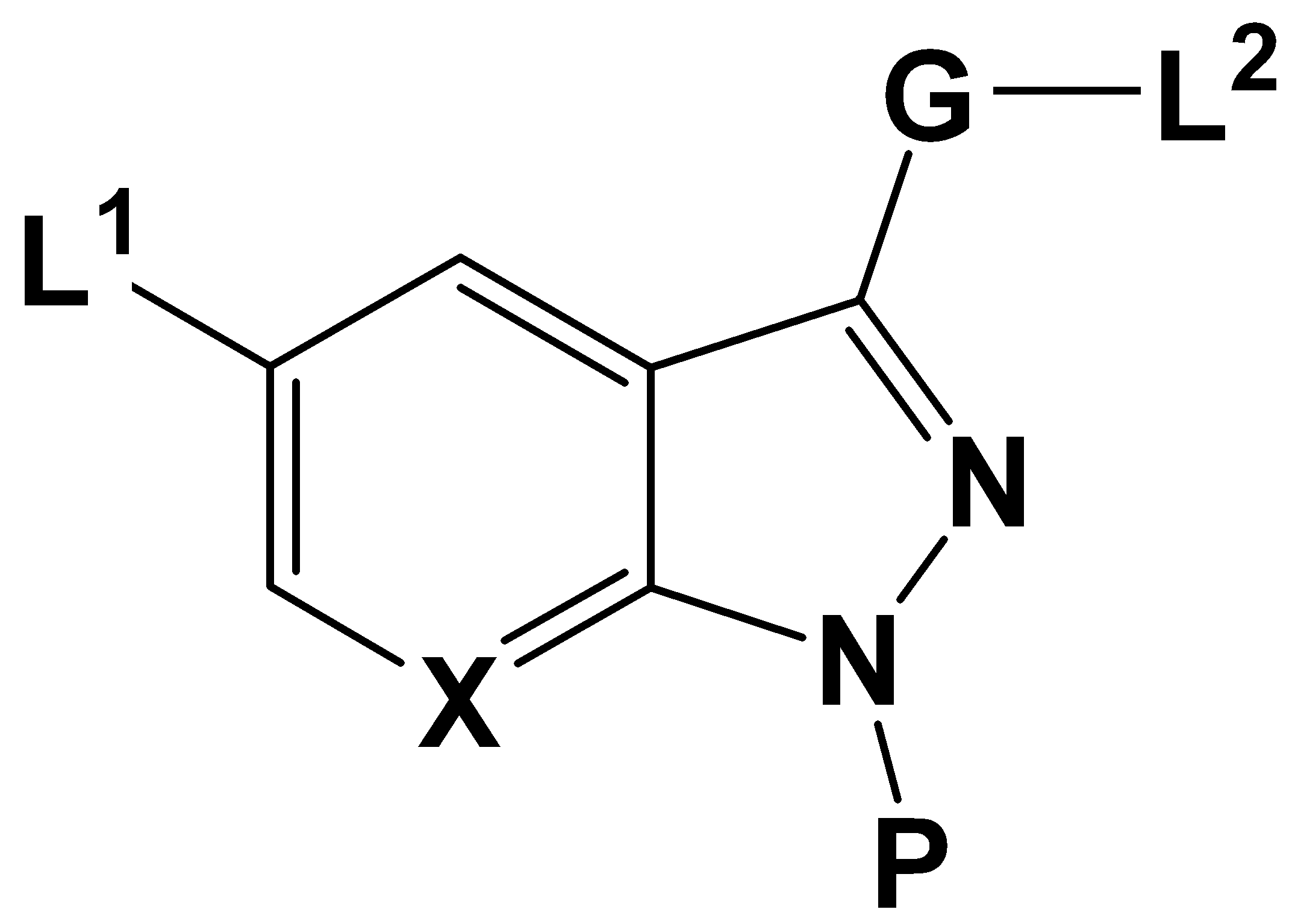
상기한 과제 해결을 위하여, 본 발명은 단백질 키나아제의 활성을 저해하는 하기 화학식 1로 표시되는 화합물, 이의 약학적으로 허용되는 염, 이의 수화물 또는 이의 용매화물을 그 특징으로 한다 : In order to solve the above problems, the present invention is characterized by a compound represented by the following formula (1), pharmaceutically acceptable salts, hydrates or solvates thereof that inhibit the activity of protein kinases:
[화학식 1][Formula 1]
상기 화학식 1에서,In Formula 1,
X는 CH; 또는 N 을 나타내며,X is CH; Or N,
P는 수소원자; C1-C10 알킬기; 1 내지 3개의 페닐이 치환된 C1∼C10 알킬기; 페닐기; 또는 산소원자가 포함된 5 내지 7각형의 헤테로싸이클로알킬기를 나타내며,P is a hydrogen atom; A C 1 -C 10 alkyl group; C 1 -C 10 alkyl group substituted with 1 to 3 phenyl; A phenyl group; Or a 5 to 7 heterocyclic alkyl group containing an oxygen atom,
L1은 -SR1; -S(O)R1; 또는 -S(O)2R1을 나타내며,L 1 is -SR 1 ; -S (O) R 1 ; Or -S (O) 2 R 1 ,
G는 존재하지 않거나; 또는 -NHC(O)-; 또는 -C(O)NH- 를 나타내며,G is absent; Or -NHC (O)-; Or -C (O) NH-,
L2는 -NHR2; -OR3; R4로 치환 또는 비치환된 C1∼C10 알킬기; R5로 치환 또는 비치환된 C3∼C10 싸이클로알킬기; R6으로 치환 또는 비치환될 수 있고 질소원자가 1 내지 2개 포함된5 내지 7각형의 헤테로싸이클로알킬기; R7로 치환 또는 비치환된 C6∼C15 아릴기; 또는 R8로 치환 또는 비치환될 수 있고 황원자 및 질소원자 중에서 선택된 헤테로원자가 1 내지 3개 포함된 5 내지 7각형의 헤테로아릴기를 나타내며,L 2 is -NHR 2 ; -OR 3 ; C 1 -C 10 alkyl group unsubstituted or substituted with R 4 ; C 3 -C 10 cycloalkyl group unsubstituted or substituted with R 5 ; 5-7 hexagonal heterocycloalkyl group which may be unsubstituted or substituted with R 6 , and contains 1 to 2 nitrogen atoms; C 6 -C 15 aryl group unsubstituted or substituted with R 7 ; Or it may be substituted or unsubstituted with R 8 and represents a 5 to 7 heteroaryl group containing 1 to 3 heteroatoms selected from a sulfur atom and a nitrogen atom,
R1은 수소원자; 또는 1 내지 3개의 할로겐원자로 치환 또는 비치환된 C6∼C15 아릴기를 나타내며,R 1 is a hydrogen atom; Or a C 6 to C 15 aryl group unsubstituted or substituted with 1 to 3 halogen atoms,
R2는 수소원자; C1∼C10 알킬기; C3∼C10 싸이클로알킬기; 또는 할로, C1∼C10 알킬, C1∼C10 알콕시, 및 1 내지 10개의 할로겐이 치환된 C1∼C10 할로알킬 중에서 선택된 치환기로 치환 또는 비치환된 페닐기를 나타내며, R 2 is a hydrogen atom; C 1 -C 10 alkyl group; C 3 -C 10 cycloalkyl group; Or a phenyl group unsubstituted or substituted with a substituent selected from halo, C 1 -C 10 alkyl, C 1 -C 10 alkoxy, and C 1 -C 10 haloalkyl substituted with 1 to 10 halogen,
R3은 수소원자; 또는 C1∼C10 알킬기를 나타내며, R 3 is a hydrogen atom; Or a C 1 to C 10 alkyl group,
R4 및 R5는 서로 같거나 다른 것으로서 아미노기; 모노(C1∼C10 알킬)아미노기; 또는 디(C1∼C10 알킬)아미노기를 나타내며,R 4 and R 5 are the same as or different from each other and an amino group; Mono (C 1 -C 10 alkyl) amino group; Or a di (C 1 -C 10 alkyl) amino group,
R6은 수소원자; 또는 C1∼C6 알킬기를 나타내며,R 6 is a hydrogen atom; Or a C 1 to C 6 alkyl group,
R7은 할로겐원자; C1∼C10 알킬기; C1∼C10 알콕시기; 1 내지 10개의 할로겐이 치환된 할로알킬기; C1∼C10 알킬기로 치환 또는 비치환될 수 있고 산소원자 및 질소원자 중에서 선택된 헤테로원자가 1 내지 3개 포함된 5 내지 7각형의 헤테로싸이클로알킬기; 아미노기; 모노(C1∼C10 알킬)아미노기; 또는 디(C1∼C10 알킬)아미노기를 나타내며,R 7 is Halogen atom; C 1 -C 10 alkyl group; C 1 -C 10 alkoxy group; Haloalkyl group substituted with 1 to 10 halogens; A 5 to 7 hexagonal heterocycloalkyl group which may be unsubstituted or substituted with a C 1 to C 10 alkyl group and contains 1 to 3 heteroatoms selected from an oxygen atom and a nitrogen atom; An amino group; Mono (C 1 -C 10 alkyl) amino group; Or a di (C 1 -C 10 alkyl) amino group,
R8은 C1∼C10 알킬기; 또는 산소원자 및 질소원자 중에서 선택된 헤테로원자가 1 내지 3개 포함된 5 내지 7각형의 헤테로싸이클로알킬기를 나타낸다.
R 8 is a C 1 -C 10 alkyl group; Or a 5 to 7 hexagonal heterocycloalkyl group including 1 to 3 heteroatoms selected from an oxygen atom and a nitrogen atom.
본 발명의 신규 화합물은 ABL, ACK1, ALK, Aurora A, Aurora B, Aurora C, BLK, BMX/ETK, BRSK1, BTK, c-Src, CAMKK, CDK1, CDK2, CDK5, CLK, DDR, DYRK1B, EPHA, EPHB, FAK/PTK2, FER, FES/FPS, FGFR, FGR, FLT3, FLT4/VEGFR3, FMS, FRK/PTK5, FYN, GSK3b, HCK, IGF1R, IR, IRAK1, IRR/INSRR, ITK, JAK2, KHS/MAP4K5, LCK, LYN, PHKg, PLK4/SAK, PYK2, RET, ROS/ROS1, TIE2/TEK, TRK, TXK, TYK, YES/YES1 등과 같은 단백질 키나아제의 활성을 저해하는 능력이 우수하므로 암질환의 예방 및 치료를 위한 약제조성물의 유효성분으로 사용될 수 있다. The novel compounds of the present invention are ABL, ACK1, ALK, Aurora A, Aurora B, Aurora C, BLK, BMX / ETK, BRSK1, BTK, c-Src, CAMKK, CDK1, CDK2, CDK5, CLK, DDR, DYRK1B, EPHA , EPHB, FAK / PTK2, FER, FES / FPS, FGFR, FGR, FLT3, FLT4 / VEGFR3, FMS, FRK / PTK5, FYN, GSK3b, HCK, IGF1R, IR, IRAK1, IRR / INSRR, ITK, JAK2, KHS / MAP4K5, LCK, LYN, PHKg, PLK4 / SAK, PYK2, RET, ROS / ROS1, TIE2 / TEK, TRK, TXK, TYK, YES / YES1, etc. It can be used as an active ingredient of a pharmaceutical composition for prevention and treatment.
본 발명에 따른 화합물로부터 예방 및 치료될 수 있는 암질환은 위암, 폐암, 간암, 대장암, 소장암, 췌장암, 뇌암, 뼈암, 흑색종, 유방암, 경화성선증, 자궁암, 자궁경부암, 두경부암, 식도암, 갑상선암, 부갑상선암, 신장암, 육종, 전립선암, 요도암, 방광암, 혈액암(백혈병, 다발성골수종, 골수이형성증후군 포함), 림프종(호치킨병, 비호치킨림프종 포함), 건선, 또는 섬유선종 등이 포함될 수 있다.
Cancer diseases that can be prevented and treated from the compounds according to the present invention are gastric cancer, lung cancer, liver cancer, colon cancer, small intestine cancer, pancreatic cancer, brain cancer, bone cancer, melanoma, breast cancer, scleroderma, uterine cancer, cervical cancer, head and neck cancer, esophageal cancer , Thyroid cancer, parathyroid cancer, kidney cancer, sarcoma, prostate cancer, urethral cancer, bladder cancer, blood cancer (including leukemia, multiple myeloma, myelodysplastic syndrome), lymphoma (including Hochkin's disease, non-Hodgkin's lymphoma), psoriasis, or fibroadenoma Etc. may be included.
본 발명에 따른 상기 화학식 1로 표시되는 화합물의 약학적으로 허용 가능한 염은 당해 기술 분야에서 통상적인 방법에 의해 제조될 수 있다. 약학적으로 허용된 염은 인체에 독성이 낮고 모화합물의 생물학적 활성과 물리화학적 성질에 악영향을 주지 않아야 한다. 약학적으로 허용된 염은 약학적으로 사용 가능한 유리산과 화학식 1의 염기 화합물의 산부가염, 그리고 알칼리 금속염 (나트륨염 등)과 알칼리 토금속염 (칼슘염 등), 그리고 유기염과 화학식 1의 카르복실산의 유기염기부가염, 그리고 아미노산부가염으로 구성된다. 약학적으로 허용된 염 제조에 사용될 수 있는 유리산은 무기산과 유기산으로 나눌 수 있다. 무기산은 염산, 황산, 질산, 인산, 과염소산, 브롬산 등이 사용될 수 있다. 유기산은 초산, 메탄설폰산, 에탄설폰산, p-톨루엔설폰산, 푸마린산, 말레산, 말론산, 프탈산, 숙신산, 젖산, 구연산, 시트르산, 글루콘산, 타타르산, 살리실산, 말산, 옥살산, 벤조산, 엠본산, 아스파르트산, 글루탐산 등이 사용될 수 있다. 유기염기부가염 제조에 사용될 수 있는 유기염기는 트리스(하이드록시메틸)메틸아민, 디시클로헥실아민 등이다. 아미노산부가염기 제조에 사용될 수 있는 아미노산은 알라닌, 글라이신 등의 천연아미노산이다. The pharmaceutically acceptable salts of the compound represented by the formula (1) according to the present invention can be prepared by a conventional method in the art. Pharmacologically acceptable salts should be low in toxicity to humans and should not adversely affect the biological activity and physicochemical properties of the parent compound. Pharmaceutically acceptable salts include pharmaceutically acceptable free acids, acid addition salts of base compounds of formula (I), and alkali metal salts (such as sodium salts) and alkaline earth metal salts (such as calcium salts) Organic base addition salts of acids, and amino acid addition salts. Free acids that can be used in the preparation of pharmaceutically acceptable salts can be divided into inorganic and organic acids. As the inorganic acid, hydrochloric acid, sulfuric acid, nitric acid, phosphoric acid, perchloric acid, bromic acid and the like can be used. The organic acid may be selected from the group consisting of acetic acid, methanesulfonic acid, ethanesulfonic acid, p- toluenesulfonic acid, fumaric acid, maleic acid, malonic acid, phthalic acid, succinic acid, lactic acid, citric acid, citric acid, gluconic acid, tartaric acid, Benzoic acid, embonic acid, aspartic acid, glutamic acid and the like can be used. Organic bases that can be used to prepare organic base addition salts include tris (hydroxymethyl) methylamine, dicyclohexylamine, and the like. Amino acids that can be used to prepare amino acid addition bases are natural amino acids such as alanine, glycine and the like.
본 발명에 따른 상기 화학식 1로 표시되는 화합물 상기한 약학적으로 허용된 염과 더불어 모든 수화물 그리고 용매화물도 포함한다. 상기한 약학적으로 허용된 염은 하기한 통상적인 방법으로 제조될 수 있다. 상기한 화학식 1의 염기 화합물을 메탄올, 에탄올, 아세톤, 1,4-디옥산과 같은 물과 섞일 수 있는 용매에 녹인 다음에 유리산 또는 유리염기를 가한 후에 결정화되거나 또는 재결정화될 수 있다. The compound represented by the formula (1) according to the present invention includes all the hydrates and solvates as well as the pharmaceutically acceptable salts described above. The above-mentioned pharmaceutically acceptable salts can be prepared by the conventional methods described below. The base compound of Formula 1 may be dissolved in a solvent such as methanol, ethanol, acetone, or 1,4-dioxane and then crystallized or recrystallized after addition of a free acid or a free base.
또한, 본 발명에 따른 상기 화학식 1로 표시되는 화합물은 하나 또는 그 이상의 비대칭 중심을 가질 수 있고, 이러한 화합물의 경우 거울상 이성질체 또는 부분입체이성질체가 존재할 수 있다. 따라서, 본 발명은 각 이성질체 또는 이들 이성질체 혼합물을 포함한다. 상이한 이성질체는 통상의 방법에 의해 분리되거나 또는 분해될 수 있거나, 또는 임의의 소정 이성질체는 통상의 합성법에 의해 또는 입체특이적 또는 비대칭적 합성에 의해 수득할 수 있다.In addition, the compound represented by Formula 1 according to the present invention may have one or more asymmetric centers, and in the case of such a compound, an enantiomer or diastereomer may exist. Accordingly, the present invention includes each isomer or a mixture of these isomers. The different isomers may be separated or cleaved by conventional methods, or any desired isomer may be obtained by conventional synthetic methods or by stereospecific or asymmetric synthesis.
또한, 본 발명은 본 발명에 따른 상기 화학식 1로 표시되는 화합물의 방사성 유도체를 포함하며, 이들 방사성 화합물은 생체연구 분야에 유용하다.The present invention also includes a radioactive derivative of the compound represented by Formula 1 according to the present invention, and these radioactive compounds are useful in the field of biomedical research.
본 발명에 따른 화합물을 정의하기 위해 사용된 치환기에 대해 보다 상세히 설명하면 다음과 같다.The substituents used to define the compounds according to the invention are described in more detail below.
본 발명에서의 '할로겐 원자'라 함은 클로로, 플루오로, 브로모, 요오도를 의미한다. In the present invention, the term "halogen atom" means chloro, fluoro, bromo, iodo.
본 발명에서의 '알킬기'라 함은 메틸, 에틸, n-프로필, i-프로필, n-부틸, i-부틸, t-부틸, n-펜틸, i-펜틸, 네오펜틸, t-펜틸, n-헥실, i-헥실, 헵틸, 옥틸 등을 포함하는 1개에서 10개까지의 탄소원자를 가지는 지방족 포화 탄화수소기를 의미한다. La 'alkyl group' in the present invention means the methyl, ethyl, n - propyl, i - propyl, n - butyl, i - butyl, t - butyl, n - pentyl, i - pentyl, neopentyl, t - pentyl, n Means an aliphatic saturated hydrocarbon group having from 1 to 10 carbon atoms including hexyl, i -hexyl, heptyl, octyl, and the like.
본 발명에서의 '싸이클로알킬기'라 함은 싸이클로프로필, 싸이클로부틸, 싸이클로프로필메틸, 싸이클로펜틸, 싸이클로헥실 등을 포함하는 1개에서 10개까지의 탄소원자를 가지는 지방족 고리형 탄화수소기를 의미한다. The term "cycloalkyl group" in the present invention means an aliphatic cyclic hydrocarbon group having 1 to 10 carbon atoms including cyclopropyl, cyclobutyl, cyclopropylmethyl, cyclopentyl, cyclohexyl, and the like.
본 발명에서의'할로알킬기'라 함은 트라이플루오르메틸기와 같이 한개 이상의 할로겐 원자에 의해 수소원자가 치환된 알킬기를 의미한다. In the present invention, the "haloalkyl group" means an alkyl group in which a hydrogen atom is substituted by one or more halogen atoms, such as trifluoromethyl group.
본 발명에서의 '알콕시기'라 함은 메톡시, 에톡시, n-프로폭시, i-프로폭시, n-부톡시, i-부톡시, t-부톡시를 포함하는, C1-C10 알킬기에서 선택된 치환체에 의해 수소원자가 치환된 하이드록시기를 의미한다. La "alkoxy group" in the present invention means the methoxy, ethoxy, n - propoxy, i - propoxy, n - butoxy, i - butoxycarbonyl, t - containing butoxy, C 1 -C 10 Means a hydroxy group substituted with a hydrogen atom by a substituent selected from an alkyl group.
본 발명에서의 '아릴기'라 함은 페닐, 나프틸, 안트라니릴, 페난트리닐 등을 포함하여, 6개에서 15개까지의 탄소원자를 가지는 단일고리, 두고리, 또는 세고리의 방향족 탄화수소기를 의미한다. In the present invention, the "aryl group" means a monocyclic, bicyclic, or tricyclic aromatic hydrocarbon group having 6 to 15 carbon atoms, including phenyl, naphthyl, anthranilyl, phenanthryl, and the like. do.
본 발명에서의 '헤테로아릴기'라 함은 피롤릴, 퓨라닐, 싸이오페닐, 피라졸릴, 이미다졸릴, 옥사졸릴, 이소옥사졸릴, 싸이아졸릴, 이소싸이아졸릴, 트리아졸릴, 옥사디아졸릴, 싸이아디아졸릴, 테트라졸릴, 피리디닐, 피라지닐, 피리다지닐, 피리미디닐, 트리아졸릴, 인돌릴, 이소인돌릴, 벤조퓨라닐, 벤조퓨라자닐, 디벤조퓨라닐, 이소벤조퓨라닐, 인다졸릴, 벤즈이미다졸릴, 벤즈옥사졸릴, 벤즈이소옥사졸릴, 벤조싸이아졸릴, 디벤조싸이오페닐, 나프티리딜, 벤즈이소싸이아졸릴, 퀴놀리닐, 이소퀴놀리닐, 퀴녹살리닐, 프탈라지닐, 치놀리닐, 퀴나졸리닐 등을 포함하여, 헤테로원자가 1개 이상 포함된 5 내지 7각형의 단일고리, 두고리, 또는 세고리 방향족 헤테로탄화수소기를 의미한다. In the present invention, the "heteroaryl group" refers to pyrrolyl, furanyl, thiophenyl, pyrazolyl, imidazolyl, oxazolyl, isoxazolyl, thiazolyl, isothiazolyl, triazolyl, oxadia Zolyl, thiadiazolyl, tetrazolyl, pyridinyl, pyrazinyl, pyridazinyl, pyrimidinyl, triazolyl, indolyl, isoindolyl, benzofuranyl, benzofurazanyl, dibenzofuranyl, isobenzo Furanyl, indazolyl, benzimidazolyl, benzoxazolyl, benzisoxazolyl, benzothiazolyl, dibenzothiophenyl, naphthyridyl, benzisothiazolyl, quinolinyl, isoquinolinyl, quinoxyl It means a 5- to 7-membered monocyclic, bicyclic, or tricyclic aromatic heterohydrocarbon group containing one or more heteroatoms, including salinyl, phthalazinyl, chinolinyl, quinazolinyl, and the like.
본 발명에서의 '헤테로싸이클로알킬기'라 함은 몰포리닐, 피페리딘기, 피페라지닐, N-보호된 피페라지닐 등을 포함하여, 헤테로원자가 1개 이상 포함된 5 내지 7각형의 지방족 헤테로탄화수소 고리기를 의미한다. 피페라지닐의 N-보호기로는 통상적으로 알킬기가 포함될 수 있다.In the present invention, the term "heterocycloalkyl group" includes a morpholinyl, piperidine group, piperazinyl, N -protected piperazinyl, and the like, and a 5- to 7-membered aliphatic hetero-containing group containing one or more heteroatoms. It means a hydrocarbon ring group. The N -protecting group of piperazinyl may typically contain an alkyl group.
본 발명에 따른 상기 화학식 1로 표시되는 화합물에 있어서, 바람직하기로는 다음과 같다.In the compound represented by the formula (1) according to the present invention, Preferably it is as follows.
상기 X는 CH; 또는 N 을 나타내며, 상기 P는 수소원자; C1∼C6 알킬기; 벤질기; 트리틸기; 페닐기; 또는 테트라하이드로피란일기를 나타내며, L1은 -S(O)R1; 또는 -S(O)2R1을 나타내며, 이때 R1은 수소원자; 페닐기; 또는 1 내지 3개의 할로겐원자로 치환된 페닐기를 나타내며, G는 존재하지 않거나; 또는 -NHC(O)-; 또는 -C(O)NH- 를 나타내며, L2는 아미노기; 싸이클로헥실아미노기; 페닐아미노기; C1∼C6 알콕시페닐아미노기; 할로 및 할로알킬 중에서 선택된 치환기로 치환된 페닐아미노기; 하이드록시기; C1∼C6 알콕시기; C1∼C6 알킬기; 디메틸아미노 C1∼C6 알킬기; 싸이클로헥실기; 디메틸아미노싸이클로헥실기; 몰포리노기; 피페리디닐기; 피페라지닐기; 4-(C1∼C6 알킬)피페라지닐기; 페닐기; 클로로페닐기; 플루오로페닐기; 4-클로로-3-트리플루오로메틸페닐기; C1∼C6 알킬페닐기; C1∼C6 알콕시페닐기; 트리플루오로메틸페닐기; 몰포리노페닐기; (4-메틸피페라지닐)페닐기; (4-에틸피페라지닐)페닐기; 아미노페닐기; 디메틸아미노페닐기; 티오페닐기; 피리디닐기; (C1∼C6 알킬)피리디닐기; 또는 몰포리노피리디닐기를 나타내는 화합물이다.X is CH; Or N, wherein P is a hydrogen atom; C 1 -C 6 alkyl group; Benzyl group; Trityl group; A phenyl group; Or a tetrahydropyranyl group, L 1 is -S (O) R 1 ; Or -S (O) 2 R 1 , wherein R 1 is a hydrogen atom; A phenyl group; Or a phenyl group substituted with 1 to 3 halogen atoms, G is absent; Or -NHC (O)-; Or -C (O) NH-, L 2 is an amino group; Cyclohexylamino group; Phenylamino group; C 1 -C 6 alkoxyphenylamino group; Phenylamino group substituted with a substituent selected from halo and haloalkyl; A hydroxyl group; C 1 -C 6 alkoxy group; C 1 -C 6 alkyl group; Dimethylamino C 1 to C 6 alkyl group; Cyclohexyl group; Dimethylaminocyclohexyl group; A morpholino group; Piperidinyl group; Piperazinyl groups; 4- (C 1 -C 6 alkyl) piperazinyl group; A phenyl group; Chlorophenyl group; Fluorophenyl group; 4-chloro-3-trifluoromethylphenyl group; C 1 -C 6 alkylphenyl group; C 1 -C 6 alkoxyphenyl group; Trifluoromethylphenyl group; Morpholinophenyl group; (4-methylpiperazinyl) phenyl group; (4-ethylpiperazinyl) phenyl group; Aminophenyl group; Dimethylaminophenyl group; Thiophenyl group; Pyridinyl group; (C 1 -C 6 alkyl) pyridinyl group; Or a compound showing a morpholino pyridinyl group.
본 발명에 따른 상기 화학식 1로 표시되는 화합물을 구체적으로 예시하면 다음과 같다 : The compound represented by the formula (1) according to the present invention is specifically exemplified as follows:
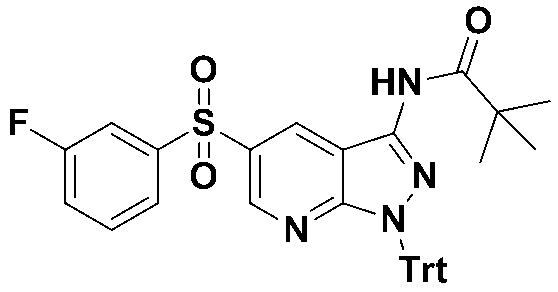
5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-아민 (화합물번호 1);5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-amine (Compound No. 1);
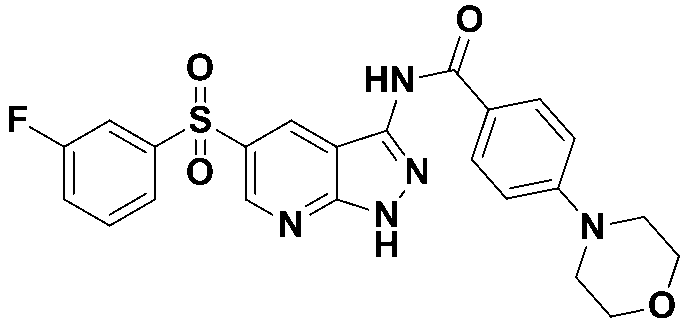
N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-4-(4-메틸피페라진-1-일)벤즈아미드 (화합물번호 2); N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -4- (4-methylpiperazin- 1-yl) benzamide (Compound No. 2);
N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-3-(트리플루오로메틸)벤즈아미드 (화합물번호 3); N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -3- (trifluoromethyl) benz Amides (Compound No. 3);
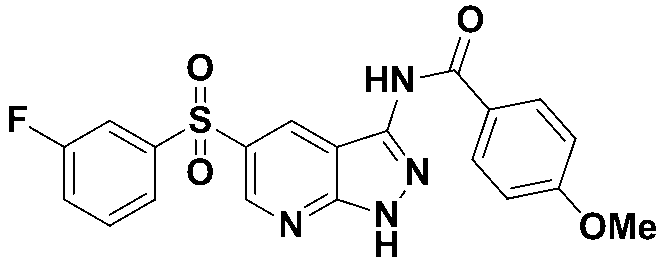
N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-4-메톡시벤즈아미드 (화합물번호 4); N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -4-methoxybenzamide (Compound No. 4);
N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-벤즈아미드 (화합물번호 5); N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -benzamide (Compound No. 5);
4-플루오로-N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-벤즈아미드 (화합물번호 6);4-Fluoro- N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -benzamide (compound Number 6);
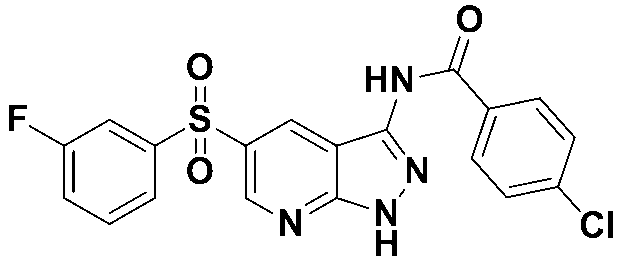
4-클로로-N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-벤즈아미드 (화합물번호 7);4-Chloro- N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -benzamide (Compound No. 7);
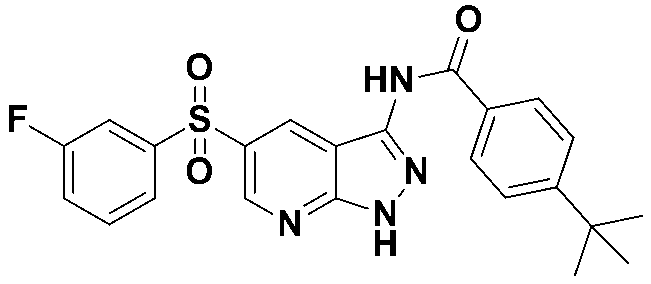
4-(tert-부틸)-N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-벤즈아미드 (화합물번호 8);4- (tert-butyl) - N - (5 - ( (3- fluorophenyl) sulfonyl) -1-trityl -1 H-pyrazolo [3,4- b] pyridin-3-yl) benzamide Amides (Compound No. 8);
N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-4-몰포리노벤즈아미드 (화합물번호 9); N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -4-morpholinobenzamide (Compound No. 9);
4-(디메틸아미노)-N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-벤즈아미드 (화합물번호 10);4- (dimethylamino) - N - (5 - ((3- fluorophenyl) sulfonyl) -1-trityl -1 H-pyrazolo [3,4- b] pyridin-3-yl) -benzamide (Compound number 10);
4-(디메틸아미노)-N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-싸이클로헥산카복스아미드 (화합물번호 11);4- (dimethylamino) - N - (5 - ((3- fluorophenyl) sulfonyl) -1-trityl -1 H-pyrazolo [3,4- b] pyridin-3-yl) cyclo-hexane Carboxamide (Compound No. 11);
4-(디메틸아미노)-N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-피발로아미드 (화합물번호 12);4- (dimethylamino) - to Sangapi - N - (5 - ((3- fluorophenyl) sulfonyl) -1-trityl -1 H-pyrazolo [3,4- b] pyridin-3-yl) Amides (Compound No. 12);
4-(디메틸아미노)-N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-헥산아미드 (화합물번호 13);4- (dimethylamino) - N - (5 - ((3- fluorophenyl) sulfonyl) -1-trityl -1 H-pyrazolo [3,4- b] pyridin-3-yl) -pentanoic amide (Compound number 13);
에틸-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-카바메이트 (화합물번호 14);Ethyl- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -carbamate (Compound No. 14);
tert-부틸-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-카바메이트 (화합물번호 15); tert -butyl- (5-((3-fluorophenyl) sulfonyl) -1-trityl- 1H -pyrazolo [3,4- b ] pyridin-3-yl) -carbamate (Compound No. 15) ;
N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-티오펜-2-카복스아미드 (화합물번호 16); N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -thiophene-2-carboxamide ( Compound no. 16);
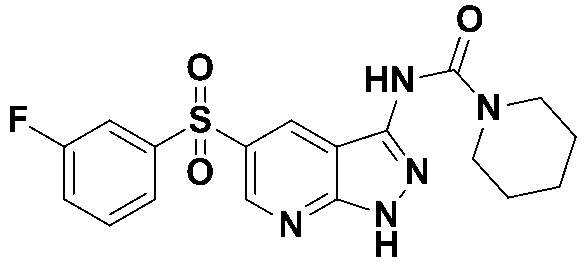
N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-피페리딘-1-카복스아미드 (화합물번호 17); N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -piperidine-1-carboxamide (Compound number 17);
1-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-3-페닐우레아 (화합물번호 18);1- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -3-phenylurea (Compound No. 18) ;
1-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-3-(2-메톡시페닐)우레아 (화합물번호 19);1- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -3- (2-methoxyphenyl) Urea (Compound No. 19);
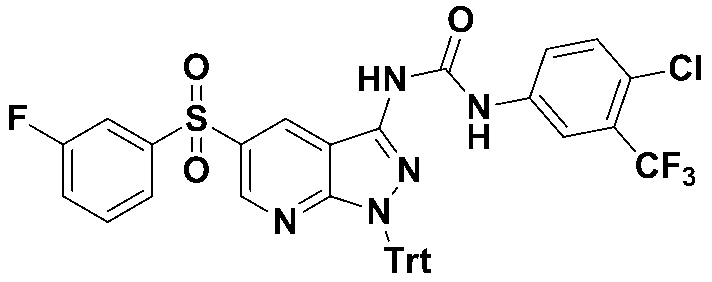
1-(4-클로로-3-(트리플루오로메틸)페닐)-3-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)우레아 (화합물번호 20);1- (4-chloro-3- (trifluoromethyl) phenyl) -3- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) urea (Compound No. 20);
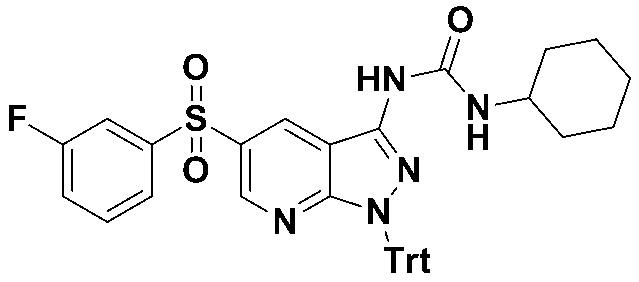
1-싸이클로헥실-3-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)우레아(화합물번호 21);1-cyclohexyl-3- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) urea (Compound No. 21 );
N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)-4-(4-메틸피페라진-1-일)벤즈아미드(화합물번호 22); N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) -4- (4-methylpiperazin-1-yl) benz Amide (Compound No. 22);
N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)-4-메톡시벤즈아미드 (화합물번호 23); N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) -4-methoxybenzamide (Compound No. 23);
N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)-3-(트리플루오로메틸)벤즈아미드 (화합물번호 24); N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) -3- (trifluoromethyl) benzamide (Compound No. 24 );
N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)벤즈아미드 (화합물번호 25); N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) benzamide (Compound No. 25);
4-플루오로-N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)벤즈아미드 (화합물번호 26);4-fluoro- N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) benzamide (Compound No. 26);
4-클로로-N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)벤즈아미드 (화합물번호 27);4-chloro- N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) benzamide (Compound No. 27);
4-(tert-부틸)-N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)벤즈아미드 (화합물번호 28);4- (tert-butyl) - N - (5 - ((3- phenyl) sulfonyl) -1 H-fluoro-pyrazolo [3,4- b] pyridin-3-yl) benzamide (Compound No. 28) ;
N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)-4-몰포리노벤즈아미드 (화합물번호 29); N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) -4-morpholinobenzamide (Compound No. 29);
4-(디메틸아미노)-N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)벤즈아미드 (화합물번호 30);4- (dimethylamino) - N - (5 - ( ( 3-fluorophenyl) sulfonyl) -1 H - pyrazolo [3,4- b] pyridin-3-yl) benzamide (Compound No. 30);
N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)싸이클로헥산카복스아미드 (화합물번호 31); N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) cyclohexanecarboxamide (Compound No. 31);
N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)피발아미드 (화합물번호 32); N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) pivalamide (Compound No. 32);
N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)헥산아미드 (화합물번호 33); N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) hexanamide (Compound No. 33);
에틸-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)카바메이트 (화합물번호 34);Ethyl- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) carbamate (Compound No. 34);
tert-부틸-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)카바메이트 (화합물번호 35); tert -butyl- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) carbamate (Compound No. 35);
N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)티오펜-2-카복스아미드 (화합물번호 36); N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) thiophene-2-carboxamide (Compound No. 36);
N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)피페리딘-1-카복스아미드 (화합물번호 37); N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) piperidine-1-carboxamide (Compound No. 37);
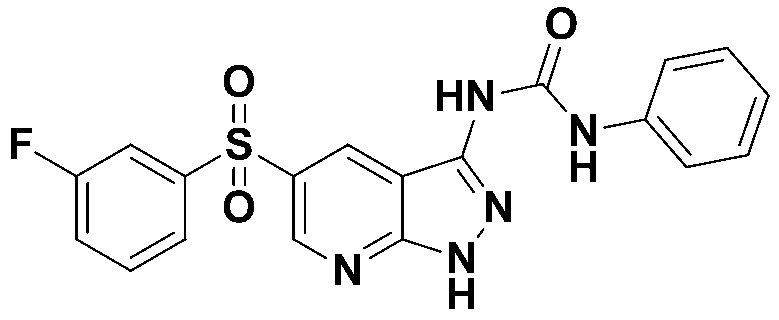
1-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)-3-페닐우레아 (화합물번호 38);1- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) -3-phenylurea (Compound No. 38);
1-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)-3-(2-메톡시페닐)우레아 (화합물번호 39);1- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) -3- (2-methoxyphenyl) urea (Compound No. 39 );
1-(4-클로로-3-(트리플루오로메틸)페닐)-3-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)우레아 (화합물번호 40);1- (4-chloro-3- (trifluoromethyl) phenyl) -3- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridine-3 -Yl) urea (Compound No. 40);
1-싸이클로헥실-3-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)우레아 (화합물번호 41);1-cyclohexyl-3- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) urea (Compound No. 41);
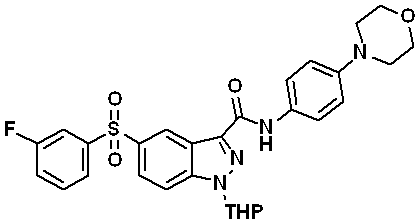
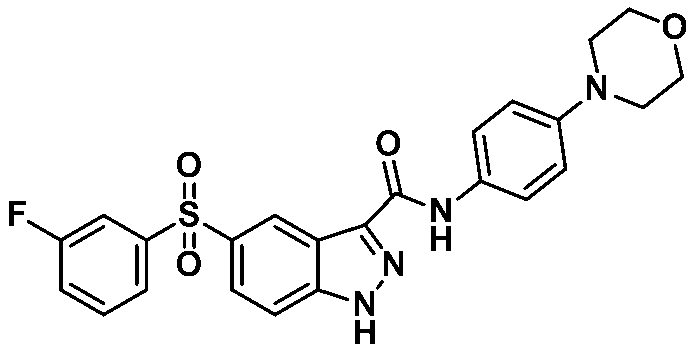
5-((3-플루오로페닐)설포닐)-N-(4-몰포리노페닐)-1-(테트로하이드로-2H-피란-2-일)-1H-인다졸-3-카복스아미드 (화합물번호 42);
5 - ((3-fluorophenyl) sulfonyl) - N - (4- Dimorpholino Reno phenyl) -1- (tetrahydro -2 H in Tet-pyran-2-yl) -1 H-indazol-3-car Voxamide (Compound No. 42);
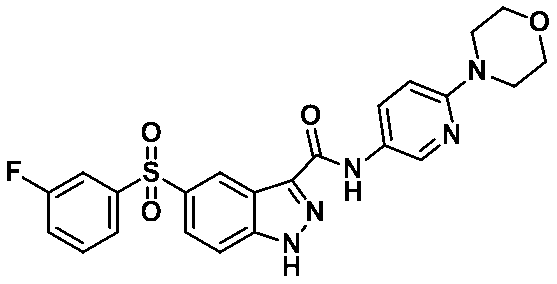
5-((3-플루오로페닐)설포닐)-N-(6-몰포리노피리딘-3-일)-1-(테트로하이드로-2H-피란-2-일)-1H-인다졸-3-카복스아미드 (화합물번호 43);5 - ((3-fluorophenyl) sulfonyl) - N - (6- Reno Dimorpholino-3-yl) -1- (tetrahydro -2 H in Tet-pyran-2-yl) -1 H-indazole -3-carboxamide (Compound No. 43);
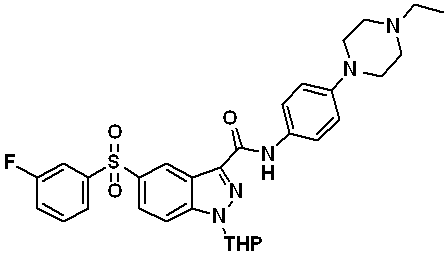
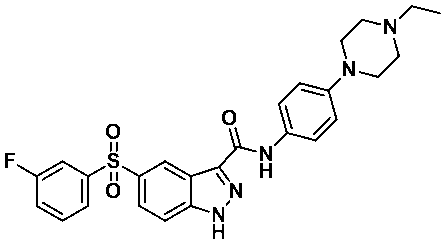
N-(4-(4-에틸피페라진-1-일)페닐)-5-((3-플루오로페닐)설포닐)-1-(테트로하이드로-2H-피란-2-일)-1H-인다졸-3-카복스아미드 (화합물번호 44); N- (4- (4-ethylpiperazin-1-yl) phenyl) -5-((3-fluorophenyl) sulfonyl) -1- (tetrohydro- 2H -pyran-2-yl)- 1 H -indazol-3-carboxamide (Compound No. 44);
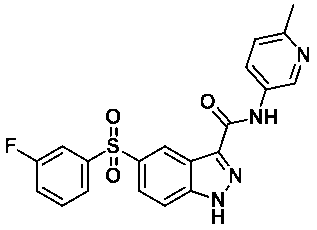
5-((3-플루오로페닐)설포닐)-N-(6-메틸피리딘-3-일)-1-(테트로하이드로-2H-피란-2-일)-1H-인다졸-3-카복스아미드 (화합물번호 45);5 - ((3-fluorophenyl) sulfonyl) - N - (6- methylpyridin-3-yl) -1- (tetrahydro -2 H in Tet-pyran-2-yl) -1 H-indazol- 3-carboxamide (Compound No. 45);
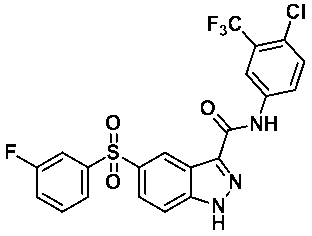
N-(4-클로로-3-(트리프루오로메틸)페닐))-5-((3-플루오로페닐)설포닐)-1-(테트로하이드로-2H-피란-2-일)-1H-인다졸-3-카복스아미드 (화합물번호 46); N- (4-chloro-3- (trifluoromethyl) phenyl))-5-((3-fluorophenyl) sulfonyl) -1- (tetrohydro- 2H -pyran-2-yl) -1 H -indazole-3-carboxamide (Compound No. 46);
5-((3-플루오로페닐)설포닐)-N-(4-몰포리노페닐)-1H-인다졸-3-카복스아미드 (화합물번호 47);5 - ((3-fluorophenyl) sulfonyl) - N - (4- Dimorpholino Reno phenyl) -1 H-indazole-3-carboxamide (Compound No. 47);
5-((3-플루오로페닐)설포닐)-N-(4-몰포리노피리딘-3-일)-1H-인다졸-3-카복스아미드 (화합물번호 48);5 - ((3-fluorophenyl) sulfonyl) - N - (4- Reno Dimorpholino-3-yl) -1 H-indazole-3-carboxamide (Compound No. 48-);
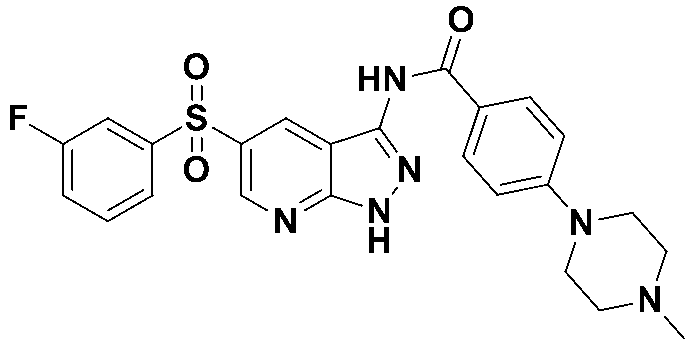
N-(4-(4-에틸피페라진-1-일)페닐)-5-((3-플루오로페닐)설포닐)-1H-인다졸-3-카복스아미드 (화합물번호 49); N- (4- (4-ethylpiperazin-1-yl) phenyl) -5-((3-fluorophenyl) sulfonyl) -1 H -indazol-3-carboxamide (Compound No. 49);
5-((3-플루오로페닐)설포닐)-N-(6-메틸피리딘-3-일)-1H-인다졸-3-카복스아미드 (화합물번호 50); 또는5 - ((3-fluorophenyl) sulfonyl) - N - (6- methylpyridin-3-yl) -1 H-indazole-3-carboxamide (Compound No. 50); or
N-(4-클로로-3-(트리플루오로메틸)페닐)-5-((3-플루오로페닐)설포닐)-1H-인다졸-3-카복스아미드 (화합물번호 51)가 포함될 수 있다.
N- (4-chloro-3- (trifluoromethyl) phenyl) -5-((3-fluorophenyl) sulfonyl) -1 H -indazole-3-carboxamide (Compound No. 51) Can be.
한편, 본 발명은 상기 화학식 1로 표시되는 화합물의 제조방법을 그 특징으로 한다. 본 발명에 따른 제조방법을 구체적으로 설명하면 하기와 같다.
On the other hand, the present invention is characterized by a method for producing a compound represented by the formula (1). Hereinafter, the manufacturing method according to the present invention will be described in detail.
제조방법 1Manufacturing Method 1
하기 반응식 1로 표시되는 바와 같이, 핵심 중간체인 하기 화학식 3으로 표시되는 화합물은 하기 화학식 2로 표시되는 화합물로부터 유기금속화합물을 이용한 결합반응(예, 부크왈드 커플링 반응(Buchwald coupling reaction))을 수행하여 제조할 수 있다.As represented by Scheme 1, the compound represented by the following Chemical Formula 3, which is a core intermediate, may be subjected to a coupling reaction (eg, Buchwald coupling reaction) using an organometallic compound from the compound represented by the following Chemical Formula 2. Can be prepared.
[반응식 1][Reaction Scheme 1]
(상기 반응식 1에서, L1은 상기 화학식 1에서 정의한 바와 같다.)(In Reaction Scheme 1, L 1 is as defined in Formula 1).
상기 반응식 1에 따른 제조방법에서 수행하게 되는 부크왈드 커플링 반응에서는 금속 화합물로서 Pd2(dba)3, Pd(OAc)2, PdCl2(PPh3)2, Pd(PPh3)4 등을 사용할 수 있다. 리간드로서 Xantphos (CAS number: 161265-03-8), Davephos (CAS number: 213697-53-1), Johnphos (CAS number: 224311-51-7), X-phos (CAS number: 564483-18-7), tert-Butyl Xphos (CAS nuber: 564483-19-8) 등을 사용할 수 있다. 그리고 염기로서는 아민류의 유기염기, 알칼리금속 또는 알칼리 토금속의 탐산염, 황산염, 인산염, 알콕사이드류 등을 사용할 수 있다. 그리고, 반응용매로서 테트라하이드로퓨란, 디옥산, N,N-디메틸포름아마이드, N,N-디메틸설폭사이드, 2-부탄올, 2-펜탄올 등이 포함되는 통상의 유기용매를 사용할 수 있다. 반응온도는 50℃ 내지 200℃ 범위이며, 바람직하기로는 80℃ 내지 150℃ 범위를 유지하는 것이다.
In the Buchwald coupling reaction performed in the preparation method according to Scheme 1, Pd 2 (dba) 3 , Pd (OAc) 2 , PdCl 2 (PPh 3 ) 2 , Pd (PPh 3 ) 4, etc. may be used as the metal compound. Can be. As ligand, Xantphos (CAS number: 161265-03-8), Davephos (CAS number: 213697-53-1), Johnphos (CAS number: 224311-51-7), X-phos (CAS number: 564483-18-7 ), tert- Butyl Xphos (CAS nuber: 564483-19-8) and the like can be used. Examples of the base include organic bases of amines, tamate salts of alkali metals or alkaline earth metals, sulfates, phosphates, alkoxides and the like. As the reaction solvent, a conventional organic solvent containing tetrahydrofuran, dioxane, N, N -dimethylformamide, N, N -dimethylsulfoxide, 2-butanol, 2-pentanol and the like can be used. The reaction temperature is in the range of 50 ° C to 200 ° C, and preferably in the range of 80 ° C to 150 ° C.
제조방법 2Manufacturing Method 2
하기 반응식 2로 표시되는 바와 같이, 하기 화학식 6으로 표시되는 3-아미노-5-치환된 1H-피라졸로[3,4-b]피리딘 화합물은 하기의 3단계 제조과정을 수행하여 제조할 수 있다.As represented by Scheme 2 below, the 3-amino-5-substituted 1 H -pyrazolo [3,4- b ] pyridine compound represented by Chemical Formula 6 may be prepared by performing the following three steps. have.
(단계 2-1) 하기 화학식 3으로 표시되는 화합물을 오산화인(PCl5)과 염화포스포릴(POCl3)을 이용하여 반응시켜, 하기 화학식 4로 표시되는 화합물을 제조하는 단계; (Step 2-1) preparing a compound represented by the following Chemical Formula 4 by reacting the compound represented by the following Chemical Formula 3 with phosphorus pentoxide (PCl 5 ) and phosphoryl chloride (POCl 3 );
(단계 2-2) 하기 화학식 4로 표시되는 화합물을 산화 반응하여 설파이드(-S-) 그룹을 설파이닐(-SO-) 또는 설포닐(-SO2-) 그룹으로 전환시켜 하기 화학식 5로 표시되는 화합물을 제조하는 단계;(Step 2-2) The compound represented by the following formula (4) is oxidized to convert the sulfide (-S-) group into a sulfinyl (-SO-) or sulfonyl (-SO 2- ) group, represented by the following formula (5). Preparing a compound;
(단계 2-3) 하기 화학식 5로 표시되는 화합물에 하이드라진 일수화물을 반응시켜 하기 화학식 6으로 표시되는 화합물을 제조하는 단계;(Step 2-3) preparing a compound represented by the following Chemical Formula 6 by reacting a hydrazine monohydrate with a compound represented by the following Chemical Formula 5;
를 거쳐 제조될 수 있다.It can be prepared via.
[반응식 2][Reaction Scheme 2]
(상기 반응식 2에서, L1은 상기 화학식 1에서 정의한 바와 같다.)
(In Scheme 2, L 1 is as defined in the formula (1).)
제조방법 3Manufacturing Method 3
하기 반응식 3으로 표시되는 바와 같이, 하기 화학식 6으로 표시되는 3-아미노-5-치환된 1H-피라졸로[3,4-b]피리딘 화합물은 하기의 2단계 제조과정을 수행하여 제조할 수 있다.As represented by Scheme 3 below, the 3-amino-5-substituted 1 H -pyrazolo [3,4- b ] pyridine compound represented by Formula 6 may be prepared by performing the following two steps. have.
(단계 3-1) 하기 화학식 3으로 표시되는 화합물을 오산화인(PCl5)과 염화포스포릴(POCl3)을 이용하여 반응시켜, 하기 화학식 4로 표시되는 화합물을 제조하는 단계; 및(Step 3-1) reacting a compound represented by Chemical Formula 3 with phosphorus pentoxide (PCl 5 ) and phosphoryl chloride (POCl 3 ) to prepare a compound represented by Chemical Formula 4; And
(단계 2-3) 하기 화학식 4로 표시되는 화합물에 하이드라진 일수화물을 반응시켜 하기 화학식 6으로 표시되는 화합물을 제조하는 단계;(Step 2-3) preparing a compound represented by the following formula (6) by reacting the hydrazine monohydrate to the compound represented by the formula (4);
를 거쳐 제조될 수 있다.It can be prepared via.
[반응식 3]Scheme 3
(상기 반응식 3에서, L1은 상기 화학식 1에서 정의한 바와 같다.)
(In Reaction Scheme 3, L 1 is as defined in Formula 1).
제조방법 4Manufacturing Method 4
하기 반응식 4로 표시되는 바와 같이, C1 위치에 P 그룹과 C3 위치에 -NHC(O)-그룹이 도입된 하기 화학식 9로 표시되는 1,3,5-삼치환된 1H-피라졸로[3,4-b]피리딘 화합물은 하기의 2단계 제조과정을 수행하여 제조할 수 있다.As shown in Scheme 4 below, 1,3,5-trisubstituted 1 H -pyrazolo [3] represented by the following formula (9) in which a P group and a -NHC (O)-group are introduced at a C1 position , 4- b ] pyridine compound can be prepared by performing the following two steps.
(단계 4-1) 하기 화학식 6으로 표시되는 화합물을 무수 트리플루오로아세트산을 이용하여 반응시켜 하기 화학식 8로 표시되는 화합물을 제조하는 단계; (Step 4-1) preparing a compound represented by the following Chemical Formula 8 by reacting the compound represented by the following Chemical Formula 6 with trifluoroacetic anhydride;
(단계 4-2) 상기 화학식 8로 표시되는 화합물에 P-X로 표시되는 화합물을 반응시켜 하기 화학식 9로 표시되는 화합물을 제조하는 단계;(Step 4-2) preparing a compound represented by Formula 9 by reacting the compound represented by P-X with the compound represented by Formula 8;
를 거쳐 제조될 수 있다.It can be prepared via.
[반응식 4][Reaction Scheme 4]
(상기 반응식 4에서, L1 및 P는 상기 화학식 1에서 정의한 바와 같고, X는 할로겐원자 또는 tert-부틸 카보네이트를 나타낸다.)
(In Scheme 4, L 1 and P are as defined in Formula 1, X represents a halogen atom or tert -butyl carbonate.)
제조방법 5Manufacturing Method 5
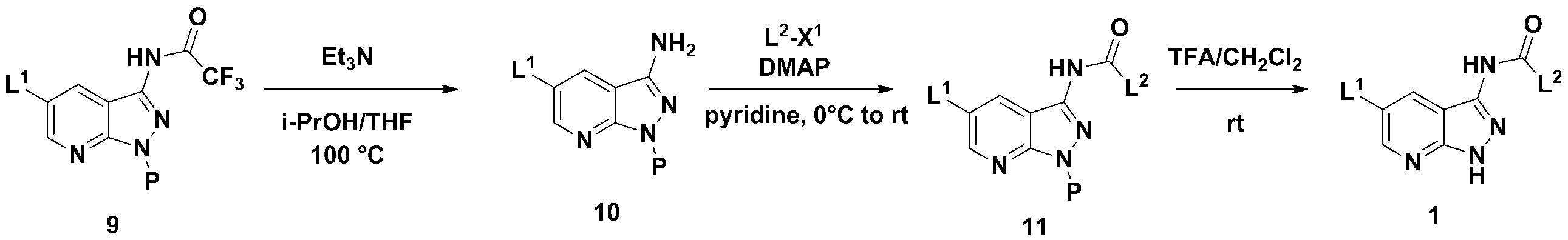
하기 반응식 5로 표시되는 바와 같이, C3 위치에 다양한 L2 그룹이 도입된 본 발명에 따른 상기 화학식 1로 표시되는 화합물은 하기의 3단계 제조과정을 수행하여 상기 화학식 9로 표시되는 화합물로부터 제조할 수 있다.As shown in Scheme 5 below, the compound represented by Formula 1 according to the present invention having various L 2 groups introduced at the C3 position may be prepared from the compound represented by Formula 9 by performing the following three steps. Can be.
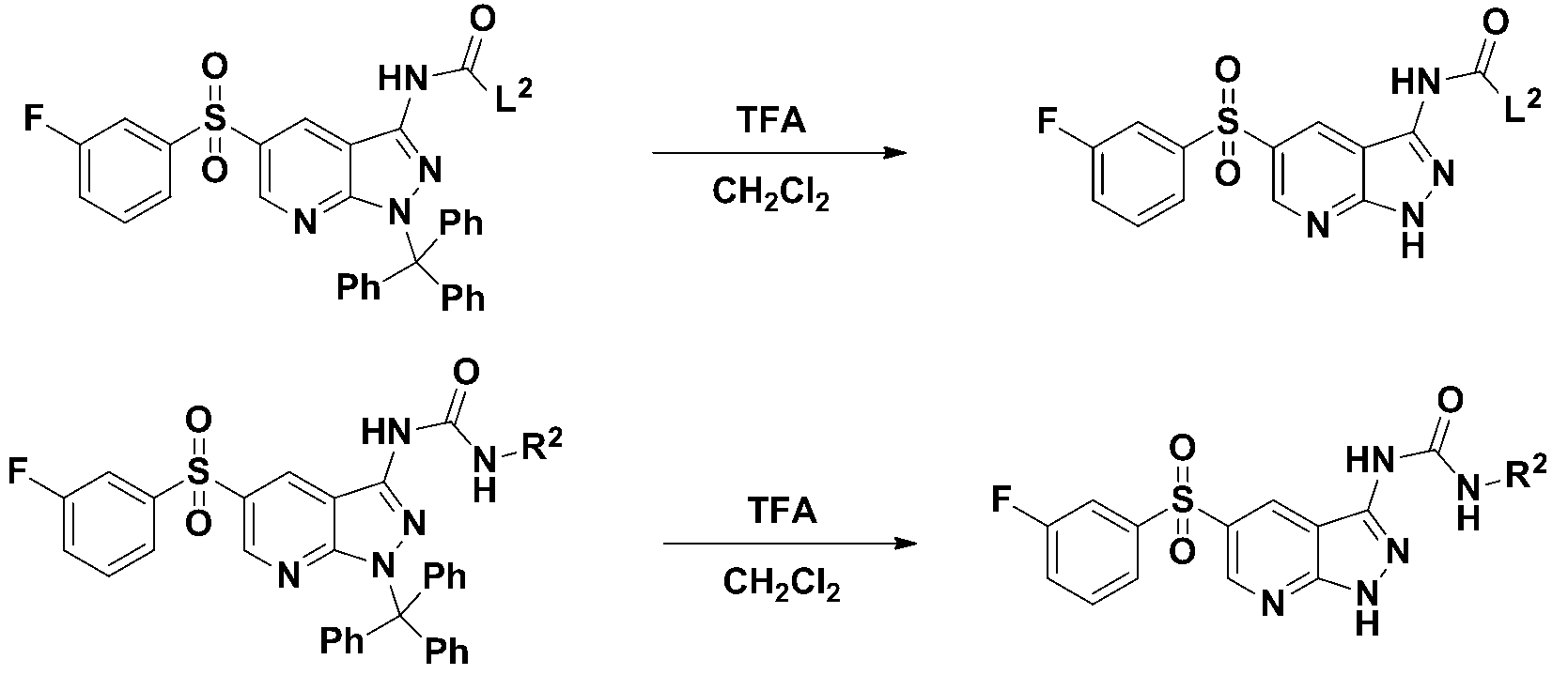
(단계 5-1) 하기 화학식 9로 표시되는 화합물을 유기염기를 이용한 탈보호 반응을 수행하여 하기 화학식 10으로 표시되는 화합물을 제조하는 단계; (Step 5-1) preparing a compound represented by the following Chemical Formula 10 by performing a deprotection reaction using an organic base to the compound represented by the following Chemical Formula 9;
(단계 5-2) 하기 화학식 10으로 표시되는 화합물과 L2-X1로 표시되는 화합물의결합반응을 수행하여 하기 화학식 11로 표시되는 화합물을 제조하는 단계; 및 (Step 5-2) preparing a compound represented by the following Chemical Formula 11 by performing a coupling reaction between the compound represented by the following Chemical Formula 10 and the compound represented by L 2 -X 1 ; And
(단계 5-3) 하기 화학식 11로 표시되는 화합물을 산을 이용한 탈보호 반응을 수행하여 하기 화학식 1로 표시되는 화합물을 제조하는 단계; (Step 5-3) preparing a compound represented by the following Chemical Formula 1 by performing a deprotection reaction using an acid with the compound represented by the following Chemical Formula 11;
를 거쳐 제조될 수 있다.It can be prepared via.
[반응식 5][Reaction Scheme 5]
(상기 반응식 5에서, L1, L2, 및 P는 상기 화학식 1에서 정의한 바와 같고, X1은 아실클로라이이드(C(O)Cl), 이소시아네이트(NCO)를 나타낸다.)
(In Scheme 5, L 1 , L 2 , and P are as defined in Chemical Formula 1, and X 1 represents acyl chloride (C (O) Cl), isocyanate (NCO).)
제조방법 6Manufacturing Method 6
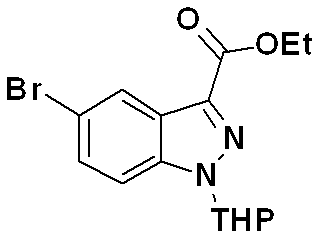
하기 반응식 6으로 표시되는 바와 같이, 하기 화학식 15로 표시되는 에틸 5-브로모-1H-인다졸-3-카복실레이트 화합물은 하기의 2단계 제조과정을 수행하여 제조할 수 있다.As shown in Scheme 6, the ethyl 5-bromo-1 H -indazole-3-carboxylate compound represented by the following Formula 15 may be prepared by performing the following two steps.
(단계 6-1) 하기 화학식 12로 표시되는 화합물로부터 하기 화학식 13으로 표시되는 5-브로모-1H-인다졸-3-카복시산 화합물을 제조하는 단계; (Step 6-1) preparing a 5-bromo-1 H -indazole-3-carboxylic acid compound represented by the following Chemical Formula 13 from the compound represented by the following Chemical Formula 12;
(단계 6-2) 하기 화학식 13으로 표시되는 5-브로모-1H-인다졸-3-카복시산 화합물을 에스테르화 반응하여 하기 화학식 14로 표시되는 에스테르 화합물을 제조하는 단계;(Step 6-2) esterifying a 5-bromo- 1H -indazole-3-carboxylic acid compound represented by the following Chemical Formula 13 to prepare an ester compound represented by the following Chemical Formula 14;
(단계 6-3) 하기 화학식 14로 표시되는 에스테르 화합물을 테트라하이드로피란 화합물과 반응시켜 하기 화학식 15로 표시되는 화합물을 제조하는 단계;(Step 6-3) preparing a compound represented by the following Chemical Formula 15 by reacting an ester compound represented by the following Chemical Formula 14 with a tetrahydropyran compound;
를 거쳐 제조될 수 있다.It can be prepared via.
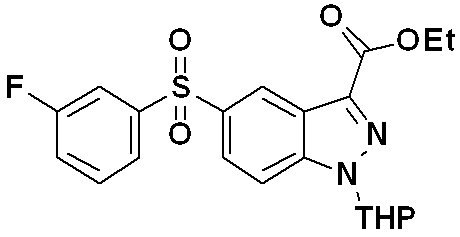
[반응식 6][Reaction Scheme 6]
(상기 반응식 6에서, PTSA는 p-톨루엔설포닐산을 나타내고, THP는 테트라하이드로피란을 나타낸다.)
(In Scheme 6, PTSA represents p- toluenesulfonyl acid and THP represents tetrahydropyran.)
제조방법 7Manufacturing Method 7
하기 반응식 7로 표시되는 바와 같이, C5 위치에 다양한 L1 그룹이 도입된 하기 화학식 17로 표시되는 화합물은 하기의 2단계 제조과정을 수행하여 제조할 수 있다.As shown in Scheme 7 below, the compound represented by the following Formula 17 having various L 1 groups introduced at the C5 position may be prepared by performing the following two steps.
(단계 7-1) 하기 화학식 15로 표시되는 화합물을 부크왈드 커플링 반응을 수행하여, 하기 화학식 16으로 표시되는 화합물을 제조하는 단계; 및(Step 7-1) preparing a compound represented by the following Chemical Formula 16 by performing a Buchwald coupling reaction with the compound represented by the following Chemical Formula 15; And
(단계 7-2) 하기 화학식 16으로 표시되는 화합물을 산화 반응하여 설파이드(-S-) 그룹을 설파이닐(-SO-) 또는 설포닐(-SO2-) 그룹으로 전환시켜 하기 화학식 17로 표시되는 화합물을 제조하는 단계;(Step 7-2) The compound represented by the following formula (16) is oxidized to convert the sulfide (-S-) group into a sulfinyl (-SO-) or sulfonyl (-SO 2- ) group, represented by the following formula (17). Preparing a compound;
를 거쳐 제조할 수 있다. ≪ / RTI >
[반응식 7][Reaction Scheme 7]
(상기 반응식 7에서, L1은 상기 화학식 1에서 정의한 바와 같고, THP는 테트라하이드로피란을 나타낸다.)
(In Reaction Scheme 7, L 1 is as defined in Formula 1, and THP represents tetrahydropyran.)
제조방법 8Manufacturing Method 8
하기 반응식 8로 표시되는 바와 같이, C3 위치에 -C(O)NH- 그룹이 도입된 하기 화학식 20으로 표시되는 화합물은 상기 화학식 17로 표시되는 에스테르 화합물로부터 하기의 2단계 제조과정을 수행하여 제조할 수 있다.As represented by the following Scheme 8, the compound represented by the formula (20) in which the -C (O) NH- group is introduced at the C3 position is prepared by performing the following two-step process from the ester compound represented by the formula (17) can do.
(단계 8-1) 하기 화학식 17로 표시되는 에스테르 화합물을 가수분해 반응하여, 하기 화학식 18로 표시되는 카르복시산 화합물을 제조하는 단계; (Step 8-1) hydrolyzing an ester compound represented by the following formula (17) to prepare a carboxylic acid compound represented by the following formula (18);
(단계 8-2) 하기 화학식 18로 표시되는 카르복시산 화합물을 다양한 아민 화합물과의 아미드결합 반응을 수행하여 하기 화학식 19로 표시되는 화합물을 제조하는 단계;(Step 8-2) preparing a compound represented by the following Chemical Formula 19 by carrying out an amide bond reaction with various amine compounds of the carboxylic acid compound represented by the following Chemical Formula 18;
(단계 8-3) 하기 화학식 19로 표시되는 화합물을 탈보호화 반응을 수행하여 하기 화학식 20으로 표시되는 화합물을 제조하는 단계;(Step 8-3) preparing a compound represented by the following Chemical Formula 20 by performing a deprotection reaction on the compound represented by the following Chemical Formula 19;
를 거쳐 제조할 수 있다. ≪ / RTI >
[반응식 8][Reaction Scheme 8]
(상기 반응식 8에서, L1 및 R2는 상기 화학식 1에서 정의한 바와 같고, THP는 테트라하이드로피란을 나타낸다.)
(In Scheme 8, L 1 and R 2 are as defined in Formula 1, THP represents tetrahydropyran.)
한편, 본 발명은 상기 화학식 1로 표시되는 화합물, 약학적으로 허용되는 이의 염, 이의 용매화물, 이의 수화물이 유효성분으로 포함된 약제조성물을 권리범위로 포함한다. Meanwhile, the scope of the present invention includes a pharmaceutical composition containing the compound represented by the formula (1), a pharmaceutically acceptable salt thereof, a solvate thereof, and a hydrate thereof as an active ingredient.
상기 화학식 1로 표시되는 화합물은 단백질 키나아제에 대한 우수한 억제 활성을 나타내므로, 비정상적인 세포 성장에 의해 유발되는 질환의 예방제 또는 치료제로 사용될 수 있다.Since the compound represented by Chemical Formula 1 exhibits excellent inhibitory activity on protein kinases, it may be used as an agent for preventing or treating a disease caused by abnormal cell growth.
상기 단백질 키나아제는 예를 들면 ABL, ACK1, ALK, Aurora A, Aurora B, Aurora C, BLK, BMX/ETK, BRSK1, BTK, c-Src, CAMKK, CDK1, CDK2, CDK5, CLK, DDR, DYRK1B, EPHA, EPHB, FAK/PTK2, FER, FES/FPS, FGFR, FGR, FLT3, FLT4/VEGFR3, FMS, FRK/PTK5, FYN, GSK3b, HCK, IGF1R, IR, IRAK1, IRR/INSRR, ITK, JAK2, KHS/MAP4K5, LCK, LYN, PHKg, PLK4/SAK, PYK2, RET, ROS/ROS1, TIE2/TEK, TRK, TXK, TYK, YES/YES1 등이 포함될 수 있다.The protein kinase is for example ABL, ACK1, ALK, Aurora A, Aurora B, Aurora C, BLK, BMX / ETK, BRSK1, BTK, c-Src, CAMKK, CDK1, CDK2, CDK5, CLK, DDR, DYRK1B, EPHA, EPHB, FAK / PTK2, FER, FES / FPS, FGFR, FGR, FLT3, FLT4 / VEGFR3, FMS, FRK / PTK5, FYN, GSK3b, HCK, IGF1R, IR, IRAK1, IRR / INSRR, ITK, JAK2, KHS / MAP4K5, LCK, LYN, PHKg, PLK4 / SAK, PYK2, RET, ROS / ROS1, TIE2 / TEK, TRK, TXK, TYK, YES / YES1 and the like.
본 발명에서의 비정상적인 세포 성장에 의해 유발되는 질환은 예를 들면 위암, 폐암, 간암, 대장암, 소장암, 췌장암, 뇌암, 뼈암, 흑색종, 유방암, 경화성선종, 자궁암, 자궁경부암, 두경부암, 식도암, 갑상선암, 부갑상선암, 신장암, 육종, 전립선암, 요도암, 방광암, 백혈병, 다발성골수종, 골수이형성증후군과 같은 혈액암, 호치킨병과 비호치킨림프종과 같은 림프종, 또는 섬유선종 등의 각종 암질환이 포함될 수 있다.Diseases caused by abnormal cell growth in the present invention are, for example, stomach cancer, lung cancer, liver cancer, colon cancer, small intestine cancer, pancreatic cancer, brain cancer, bone cancer, melanoma, breast cancer, sclerotic adenocarcinoma, uterine cancer, cervical cancer, head and neck cancer, Various cancers, such as esophageal cancer, thyroid cancer, parathyroid cancer, kidney cancer, sarcoma, prostate cancer, urethral cancer, bladder cancer, leukemia, multiple myeloma, myelodysplastic syndrome, lymphomas such as Hochkin's disease and non-Hodgkin's lymphoma, or fibroadenoma Diseases may be included.
본 발명의 약제조성물은 상기 화학식 1로 표시되는 화합물, 약학적으로 허용되는 이의 염, 이의 용매화물, 이의 수화물을 유효성분으로 함유하고, 여기에 통상의 무독성 약제학적으로 허용 가능한 담체, 보강제 및 부형제 등을 첨가하여 약제학적 분야에서 통상적인 제제, 예를 들면 정제, 캅셀제, 트로키제, 액제, 현탁제 등의 경구투여용 제제 또는 비경구투여용 제제로 제제화할 수 있다. The pharmaceutical composition of the present invention comprises a compound represented by the general formula (1), a pharmaceutically acceptable salt thereof, a solvate thereof, and a hydrate thereof as an active ingredient, wherein a usual non-toxic pharmaceutically acceptable carrier, adjuvant and excipient And the like can be formulated into preparations for oral administration such as tablets, capsules, troches, liquids and suspensions, or parenteral administration preparations, which are customary in the pharmaceutical field.
본 발명의 약제 조성물에 사용될 수 있는 부형제로는 감미제, 결합제, 용해제, 용해보조제, 습윤제, 유화제, 등장화제, 흡착제, 붕해제, 산화방지제, 방부제, 활탁제, 충진제, 방향제 등이 포함될 수 있다. 예를 들면 락토스, 덱스트로스, 슈크로스, 만니톨, 솔비톨, 셀룰로오스, 글라이신, 실리카, 탈크, 스테아린산, 스테린, 마그네슘 스테아린산염, 마그네슘 알루미늄 규산염, 녹말, 젤라틴, 트라가칸트 고무, 알지닌산, 소디움 알진산염, 메틸셀룰로오스, 소디움 카르복실메틸셀룰로오스, 아가, 물, 에탄올, 폴리에틸렌글리콜, 폴리비닐피롤리돈, 염화나트륨, 염화칼슘, 오렌지 엣센스, 딸기 엣센스, 바닐라 향 등을 들 수 있다. Excipients that may be used in the pharmaceutical compositions of the present invention may include sweeteners, binders, solubilizers, solubilizers, wetting agents, emulsifiers, isotonic agents, adsorbents, disintegrants, antioxidants, preservatives, lubricants, fillers, fragrances and the like. For example, lactose, dextrose, sucrose, mannitol, sorbitol, cellulose, glycine, silica, talc, stearic acid, sterin, magnesium stearate, magnesium aluminum silicate, starch, gelatin, tragacanth rubber, arginine acid, sodium Alginate, methyl cellulose, sodium carboxymethyl cellulose, agar, water, ethanol, polyethylene glycol, polyvinylpyrrolidone, sodium chloride, calcium chloride, orange essence, strawberry essence, vanilla flavor and the like.
또한, 본 발명에 따른 화합물의 인체에 대한 투여용량은 환자의 나이, 몸무게, 성별, 투여형태, 건강상태 및 질환정도에 따라 달라질 수 있으며, 몸무게가 70kg인 성인환자를 기준으로 할 때 일반적으로 0.01 ∼ 1,000 mg/일이며, 의사 또는 약사의 판단에 따라 일정 시간간격으로 1일 1회 내지 수회로 분할 투여할 수도 있다.The dose of the compound according to the present invention to the human body may vary depending on the age, weight, sex, dosage form, health condition and disease severity of the patient, and is generally 0.01 To 1,000 mg / day. Depending on the judgment of a doctor or a pharmacist, it may be administered once to several times a day at a predetermined interval.
이상에서 설명한 바와 같은 본 발명은 하기 실시예, 제제예, 및 실험예에 의거하여 더욱 상세히 설명하겠는 바, 하기의 제조예, 실시예, 제제예, 및 실험예는 본 발명을 예시하는 것일 뿐 본 발명의 범위가 이들에 의해 한정되는 것은 아니다.
The present invention as described above will be described in more detail based on the following Examples, Preparation Examples, and Experimental Examples. The following Preparation Examples, Examples, Preparation Examples, and Experimental Examples are merely illustrative of the present invention. The scope of the invention is not limited by these.
[실시예]
[Example]
실시예 1 : N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-4-(4-메틸피페라진-1-일)벤즈아미드Example 1 N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -4- (4- Methylpiperazin-1-yl) benzamide
상기의 구조식으로 표기되는 실시예 1의 화합물은 하기와 같이 11단계의 합성과정을 통해 제조할 수 있다.Compound of Example 1 represented by the above structural formula can be prepared through the 11-step synthesis process as follows.
단계 1: 2-옥소-1,2-디하이드로피리딘-3-카보니트릴Step 1: 2-oxo-1,2-dihydropyridine-3-carbonitrile
2-클로로니코티노니트릴 (7.25 g, 0.052 mol)에 아세트산 (45 mL)을 첨가하였다. 130 ℃에서 24시간 동안 교반하고 0 ℃로 냉각하였다. 혼합액을 여과하고 아세트산과 물로 세척하여 실온에서 건조하면 목적화합물 (6.28 g)을 흰색 고체로 수득하였다. To 2-chloronicotinonitrile (7.25 g, 0.052 mol) was added acetic acid (45 mL). Stir at 130 ° C for 24 h and cool to 0 ° C. The mixture was filtered, washed with acetic acid and water and dried at room temperature to obtain the target compound (6.28 g) as a white solid.
1H NMR (400 MHz, DMSO-d 6) δ 8.14(m, 1H), 7.78(m, 1H), 6.34(m, 1H), MS m/z : 121.04 [M+1]
1 H NMR (400 MHz, DMSO -d 6 ) δ 8.14 (m, 1H), 7.78 (m, 1H), 6.34 (m, 1H), MS m / z: 121.04 [M + 1]
단계 2: 5-브로모-2-옥소-1,2-디하이드로피리딘-3-카보니트릴Step 2: 5-bromo-2-oxo-1,2-dihydropyridine-3-carbonitrile
2-옥소-1,2-디하이드로피리딘-3-카보니트릴 (6.28 g, 0.052 mol)에 아세트산(34 mL)을 첨가하고 130 ℃로 가온하였다. 고체가 녹으면 브로민 (12.5 g, 0.078 mol)을 천천히 가하고 같은 온도에서 3시간 교반하였다. 반응액을 실온으로 냉각한 후 디클로로메탄과 물을 첨가하였다. 유기층을 분리하고 수층을 디클로로메탄으로 씻어주었다. 유기층을 모아 염수로 세척하고 무수황산나트륨으로 건조하여 농축하여 목적화합물 (9.11 g, 88% 수율)을 노란색 고체로 수득하였다.To 2-oxo-1,2-dihydropyridine-3-carbonitrile (6.28 g, 0.052 mol) was added acetic acid (34 mL) and warmed to 130 ° C. When the solid was dissolved bromine (12.5 g, 0.078 mol) was slowly added and stirred at the same temperature for 3 hours. After cooling the reaction solution to room temperature, dichloromethane and water were added. The organic layer was separated and the aqueous layer was washed with dichloromethane. The combined organic layers were washed with brine, dried over anhydrous sodium sulfate and concentrated to give the target compound (9.11 g, 88% yield) as a yellow solid.
1H NMR (400 MHz, DMSO-d 6) δ 8.91(d, J = 2.48 Hz, 1H), 8.89(d, J = 2.46 Hz, 1H), MS m/z : 199.00 [M+1]
1 H NMR (400 MHz, DMSO -d 6 ) δ 8.91 (d, J = 2.48 Hz, 1H), 8.89 (d, J = 2.46 Hz, 1H), MS m / z: 199.00 [M + 1]
단계 3: 5-((3-플루오로페닐)티오)-2-옥소-1,2-디하이드로피리딘-3-카보니트릴Step 3: 5-((3-fluorophenyl) thio) -2-oxo-1,2-dihydropyridine-3-carbonitrile
밀폐용기에 5-브로모-2-옥소-1,2-디하이드로피리딘-3-카보니트릴 (800 mg, 4.00 mmol)과 Xantphos (116 mg, 0.2 mmol), Pd2(dba)3 (92 mg, 0.1 mmol), 1,4-디옥산 (8.4 mL)을 넣었다. 혼합용액에 있는 기체를 초음파와 질소기체를 이용하여 제거하였다. 디이소프로필에틸아민 (1.4 mL, 8.00 mmol)과 3-플루오로티오페놀 (0.34 mL, 4.00 mmol)를 첨가하고 밀폐시켜 110 ℃에서 36시간 교반하였다. 반응용액을 실온으로 냉각하여 규조토 패드로 여과하고 에틸아세테이트로 여러 번 세척하였다. 유기층에 물을 가하여 추출하였다. 유기층을 분리하고 물층을 에틸아세테이트로 추출하였다. 모아진 유기층을 무수황산마그네슘으로 건조하고 농축하였다. 잔사를 크로마토그래피(silica gel, 디클로로메탄:메탄올=97:3)로 정제하여 목적화합물(424 mg, 43% 수율)을 노란색 고체로 수득하였다. In a closed container, 5-bromo-2-oxo-1,2-dihydropyridine-3-carbonitrile (800 mg, 4.00 mmol), Xantphos (116 mg, 0.2 mmol), Pd 2 (dba) 3 (92 mg , 0.1 mmol) and 1,4-dioxane (8.4 mL) were added. The gas in the mixed solution was removed using ultrasonic waves and nitrogen gas. Diisopropylethylamine (1.4 mL, 8.00 mmol) and 3-fluorothiophenol (0.34 mL, 4.00 mmol) were added, sealed, and stirred at 110 ° C. for 36 hours. The reaction solution was cooled to room temperature, filtered through a pad of diatomaceous earth, and washed several times with ethyl acetate. Water was added to the organic layer and extracted. The organic layer was separated and the water layer was extracted with ethyl acetate. The combined organic layers were dried over anhydrous magnesium sulfate and concentrated. The residue was purified by chromatography (silica gel, dichloromethane: methanol = 97: 3) to afford the desired compound (424 mg, 43% yield) as a yellow solid.
1H NMR (400 MHz, DMSO-d 6) δ 8.31(d, J = 2.60 Hz, 1H), 8.12(d, J = 2.66 Hz, 1H), 7.35(m, 1H), 7.33(m, 1H), 7.24(m, 2H), MS m/z : 247.03 [M+1]
1 H NMR (400 MHz, DMSO -d 6 ) δ 8.31 (d, J = 2.60 Hz, 1H), 8.12 (d, J = 2.66 Hz, 1H), 7.35 (m, 1H), 7.33 (m, 1H), 7.24 (m, 2H), MS m / z: 247.03 [M + l]
단계 4: 2-클로로-5-((3-플루오로페닐)티오)니코티노니트릴Step 4: 2-chloro-5-((3-fluorophenyl) thio) nicotinonitrile
반응용기에 5-((3-플루오로페닐)티오)-2-옥소-1,2-디하이드로피리딘-3-카보니트릴 (2.283 g, 9.27 mmol)과 오염화인 (5.43 g, 26.00 mmol)을 가하고 질소기체를 불어주었다. 혼합물에 염화포스포릴 (11.2 mL, 120.50 mmol)을 가하고 5시간동안 환류교반하였다. 반응액을 실온으로 냉각하고 얼음물 (150 mL)에 천천히 가하였다. 혼합액에 디클로로메탄 (200 mL)를 넣고 2회 추출하였다. 유기층을 모아 염수로 씻어주고 무수황산나트륨으로 건조하고 농축하여 목적화합물(2.165 g, 88% 수율)을 갈색의 오일로 수득하였다. 5-((3-fluorophenyl) thio) -2-oxo-1,2-dihydropyridine-3-carbonitrile (2.283 g, 9.27 mmol) and phosphorus pentachloride (5.43 g, 26.00 mmol) were added to the reaction vessel. And nitrogen gas was blown. Phosphoryl chloride (11.2 mL, 120.50 mmol) was added to the mixture, followed by stirring under reflux for 5 hours. The reaction was cooled to room temperature and slowly added to ice water (150 mL). Dichloromethane (200 mL) was added to the mixture and extracted twice. The combined organic layers were washed with brine, dried over anhydrous sodium sulfate and concentrated to give the target compound (2.165 g, 88% yield) as a brown oil.
1H NMR (400 MHz, DMSO-d 6) δ 8.65(d, J = 2.41 Hz, 1H), 8.55(d, J = 2.42 Hz, 1H), 7.47(m, 1H), 7.33(m, 1H), 7.24(m, 2H), MS m/z : 265.00 [M+1] 1 H NMR (400 MHz, DMSO -d 6 ) δ 8.65 (d, J = 2.41 Hz, 1H), 8.55 (d, J = 2.42 Hz, 1H), 7.47 (m, 1H), 7.33 (m, 1H), 7.24 (m, 2H), MS m / z: 265.00 [M + 1]
단계 5: 2-클로로-5-((3-플루오로페닐)설포닐)니코티노니트릴Step 5: 2-chloro-5-((3-fluorophenyl) sulfonyl) nicotinonitrile
반응용기에 2-클로로-5-((3-플루오로페닐)티오)니코티노니트릴 (2.165 g, 8.179 mmol), 디클로로메탄 40 mL를 가하였다. 실온에서 m-클로로퍼벤조산 (mCPBA(72%), 6.86 g, 28.626 mmol)을 가하고 3시간동안 교반하였다. 반응액에 디클로로메탄 30 mL를 가하고 0 ℃로 냉각하여 10% 아황산나트륨 수용액 30 mL를 가하고 교반하였다. 수층을 제거하고 유기층을 모아 포화 탄산나트륨 수용액 40 mL, 염수 40 mL로 차례로 씻어주었다. 유기층을 무수황산마그네슘으로 건조하고 농축하였다. 잔사를 컬럼크로마토그래피 (silica gel, 에틸아세테이트:n-헥산=4:1)로 정제하여 목적화합물 (1.99 g, 82% 수율)을 흰색 고체로 수득하였다.To the reaction vessel was added 2-chloro-5-((3-fluorophenyl) thio) nicotinonitrile (2.165 g, 8.179 mmol) and 40 mL of dichloromethane. M -chloroperbenzoic acid (mCPBA (72%), 6.86 g, 28.626 mmol) was added at room temperature and stirred for 3 hours. 30 mL of dichloromethane was added to the reaction solution, cooled to 0 ° C., and 30 mL of 10% aqueous sodium sulfite solution was added thereto, followed by stirring. The aqueous layer was removed and the organic layer was collected and washed sequentially with 40 mL of saturated aqueous sodium carbonate solution and 40 mL of brine. The organic layer was dried over anhydrous magnesium sulfate and concentrated. The residue was purified by column chromatography (silica gel, ethyl acetate: n-hexane = 4: 1) to obtain the target compound (1.99 g, 82% yield) as a white solid.
1H NMR (400 MHz, CDCl3) δ 9.06(d, J = 2.48 Hz, 1H), 8.45(d, J = 2.44 Hz, 1H), 7.77(m, 1H), 7.61(m, 1H), 7.40(m, 1H), MS m/z : 296.70 [M+1]
1 H NMR (400 MHz, CDCl 3 ) δ 9.06 (d, J = 2.48 Hz, 1H), 8.45 (d, J = 2.44 Hz, 1H), 7.77 (m, 1H), 7.61 (m, 1H), 7.40 (m, 1H) , MS m / z: 296.70 [M + 1]
단계 6: 5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-아민Step 6: 5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-amine
반응용기에 2-클로로-5-((3-플루오로페닐)설포닐)니코티노니트릴 (860 mg, 2.900 mmol), 에탄올 (2.5 mL)을 가하고 교반하였다. 실온에서 하이드라진 일수화물 (362 mg, 7.255 mmol)을 가하고 80 ℃에서 30분간 교반하였다. 반응액을 실온으로 냉각하고 농축하여 목적화합물 (762 mg, 90% 수율)을 노란색 고체로 수득하였다. 2-Chloro-5-((3-fluorophenyl) sulfonyl) nicotinonitrile (860 mg, 2.900 mmol) and ethanol (2.5 mL) were added to the reaction vessel and stirred. Hydrazine monohydrate (362 mg, 7.255 mmol) was added at room temperature and stirred at 80 ° C for 30 minutes. The reaction solution was cooled to room temperature and concentrated to give the target compound (762 mg, 90% yield) as a yellow solid.
1H NMR (400 MHz, DMSO-d 6) δ 12.68(s, 1H), 8.87(s, 2H), 7.82(m, 2H), 7.69(m, 1H), 7.56(m, 1H), 6.04(s, 2H), MS m/z : 293.05 [M+1]
1 H NMR (400 MHz, DMSO- d 6 ) δ 12.68 (s, 1H), 8.87 (s, 2H), 7.82 (m, 2H), 7.69 (m, 1H), 7.56 (m, 1H), 6.04 (s, 2H), MS m / z: 293.05 [M + 1]
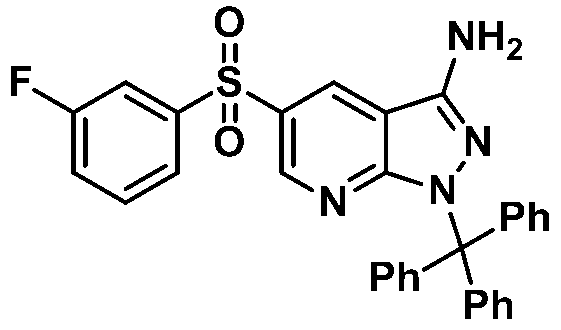
단계 7: 2,2,2-트리플루오로-N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)아세트아미드Step 7: 2,2,2-trifluoro- N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) acetamide
반응용기에 5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-아민 (585 mg, 2.00 mmol), 테트라하이드로퓨란 (24 mL)을 가하고 교반하였다. 혼합액을 0 ℃로 냉각하고 무수트리플루오로아세트산 (0.83 mL, 6.00 mmol)을 가하였다. 반응액을 실온에서 1시간동안 교반하고 농축하고 잔사에 메탄올 (5 mL)를 가하여 농축하여 목적화합물 (700 mg, 90% 수율)을 노란색 고체로 수득하였다. 목적화합물은 정제없이 다음반응에 사용하였다.To the reaction vessel, 5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-amine (585 mg, 2.00 mmol) and tetrahydrofuran (24 mL) were added. Added and stirred. The mixture was cooled to 0 ° C. and trifluoroacetic anhydride (0.83 mL, 6.00 mmol) was added. The reaction solution was stirred at room temperature for 1 hour, concentrated and methanol (5 mL) was added to the residue to give the target compound (700 mg, 90% yield) as a yellow solid. The target compound was used in the next reaction without purification.
MS m/z : 389.03 [M+1]
MS m / z: 389.03 [M + 1]
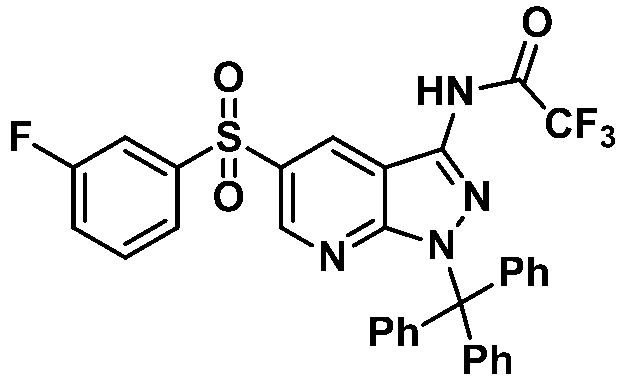
단계 8: 2,2,2-트리플루오로-N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)아세트아미드Step 8: 2,2,2-trifluoro- N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridine-3 Acylamide
반응용기에 2,2,2-트리플루오로-N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)아세트아미드 (700 mg, 1.80 mmol), 디클로로메탄 (3 mL)을 가하고 교반하였다. 혼합액에 트리에틸아민 (0.502 mL, 3.61 mmol)을 가하였다. 반응액을 0 ℃로 냉각하고 트리페닐메틸클로라이드 (502 mg, 1.80 mmol)을 가하고 실온에서 18시간 교반하였다. 반응액에 디클로로메탄 (5 mL)와 포화탄산나트륨용액 (10 mL)를 넣고 교반하였다. 수층을 디클로로메탄 (10 mL)로 두 번 씻어주었다. 유기층을 모아 무수황산나트륨으로 건조하고 잔사를 컬럼크로마토그래피 (silica gel, 에틸아세테이트:n-헥산=1:4 → 1:1)로 정제하여 목적화합물 (766 mg, 67% 수율)을 노란색 고체로 수득하였다.2,2,2-trifluoro-to the reaction vessel - N - (5 - (( 3- fluorophenyl) sulfonyl) -1 H - pyrazolo [3,4- b] pyridin-3-yl) acetamide (700 mg, 1.80 mmol) and dichloromethane (3 mL) were added and stirred. Triethylamine (0.502 mL, 3.61 mmol) was added to the mixture. The reaction solution was cooled to 0 ° C., triphenylmethyl chloride (502 mg, 1.80 mmol) was added, and stirred at room temperature for 18 hours. Dichloromethane (5 mL) and saturated sodium carbonate solution (10 mL) were added to the reaction mixture, followed by stirring. The aqueous layer was washed twice with dichloromethane (10 mL). The combined organic layers were dried over anhydrous sodium sulfate and the residue was purified by column chromatography (silica gel, ethyl acetate: n-hexane = 1: 4 → 1: 1) to obtain the target compound (766 mg, 67% yield) as a yellow solid. It was.
1H NMR (400 MHz, DMSO-d 6) δ 12.64(s, 1H), 8.99(d, J = 2.24 Hz, 1H), 8.87(d, J = 2.2 Hz, 1H), 7.89(m, 2H), 7.68(m, 2H), 7.24(m, 15H), MS m/z : 631.14 [M+1]
1 H NMR (400 MHz, DMSO -d 6 ) δ 12.64 (s, 1H), 8.99 (d, J = 2.24 Hz, 1H), 8.87 (d, J = 2.2 Hz, 1H), 7.89 (m, 2H), 7.68 (m, 2H), 7.24 (m, 15H), MS m / z: 631.14 [M + 1]
단계 9: 5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-아민Step 9: 5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-amine
반응용기에 2,2,2-트리플루오로-N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)아세트아미드 (231 mg, 0.366 mmol), 이소프로필 알코올 (IPA, 2.96 mL), 테트라하이드로퓨란 (THF, 0.74 mL)를 넣고 교반하였다. 혼합액에 트리에틸아민 (0.45 mL, 3.22 mmol)을 가하고 100 ℃로 가온하여 18시간 교반하였다. 반응액을 실온으로 농축하고 잔사를 컬럼크로마토그래피 (silica gel, 에틸아세테이트:n-헥산=1:4 → 1:2)로 정제하여 목적화합물 (100 mg, 50 % 수율)을 노란색 고체로 수득하였다.2,2,2-trifluoro- N- (5-((3-fluorophenyl) sulfonyl) -1-trityl- 1H -pyrazolo [3,4- b ] pyridine-3 in the reaction vessel -Yl) acetamide (231 mg, 0.366 mmol), isopropyl alcohol (IPA, 2.96 mL) and tetrahydrofuran (THF, 0.74 mL) were added and stirred. Triethylamine (0.45 mL, 3.22 mmol) was added to the mixed solution, which was warmed to 100 ° C and stirred for 18 hours. The reaction solution was concentrated to room temperature and the residue was purified by column chromatography (silica gel, ethyl acetate: n-hexane = 1: 4-1: 2) to obtain the target compound (100 mg, 50% yield) as a yellow solid. .
1H NMR (400 MHz, DMSO-d 6) δ 8.82(d, J = 2.24 Hz, 1H), 8.67(d, J = 2.2 Hz, 1H), 7.80(m, 2H), 7.60(m, 2H), 7.24(m, 15H), 6.26(s, 2H), MS m/z : 535.16 [M+1].
1 H NMR (400 MHz, DMSO -d 6 ) δ 8.82 (d, J = 2.24 Hz, 1H), 8.67 (d, J = 2.2 Hz, 1H), 7.80 (m, 2H), 7.60 (m, 2H), 7.24 (m, 15H), 6.26 (s, 2H), MS m / z: 535.16 [M + l].
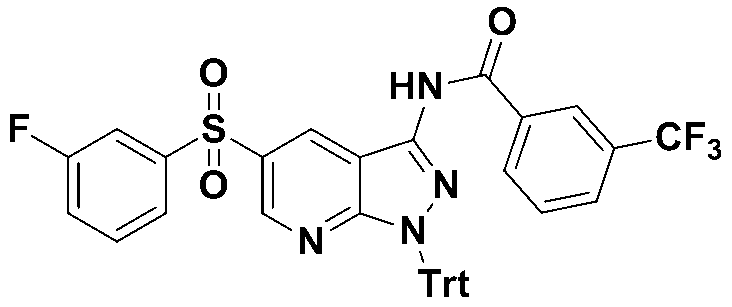
단계 10 : 4-(4-메틸피페라진-1-일)벤조일 클로라이드Step 10: 4- (4-methylpiperazin-1-yl) benzoyl chloride
반응용기에 4-(4-메틸피페라진-1-일)벤조산 (88 mg, 0.4 mmol), 디클로로메탄 (5 mg), 디메틸포름아미드 (0.023 mL)을 가하였다. 혼합액을 0 ℃로 냉각하고 옥살릴클로라이드 (0.36 mL, 4.0 mmol)을 가하고 실온에서 교반하였다. 반응액을 농축하고 목적화합물 (76 mg, 80% 수율)을 정제없이 다음반응에 사용하였다.To the reaction vessel was added 4- (4-methylpiperazin-1-yl) benzoic acid (88 mg, 0.4 mmol), dichloromethane (5 mg) and dimethylformamide (0.023 mL). The mixture was cooled to 0 ° C., oxalylchloride (0.36 mL, 4.0 mmol) was added and stirred at room temperature. The reaction solution was concentrated and the target compound (76 mg, 80% yield) was used for the next reaction without purification.
MS m/z : 240.08 [M+1].
MS m / z: 240.08 [M + l].
단계 11 : N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-4-(4-메틸피페라진-1-일)벤즈아미드Step 11: N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -4- (4-methyl Piperazin-1-yl) benzamide
반응용기에 5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-아민 (53 mg, 0.1 mmol), 디메틸아미노피리딘 (5 mg, 0.04 mmol), 피리딘 (3.2 mL)을 넣고 0 ℃에서 교반하였다. 4-(4-메틸피페라진-1-일)벤조일 클로라이드 (33 mg, 0.14 mmol)을 가하고 실온에서 18시간 교반하였다. 에틸아세테이트 (10 mL)을 가하고 포화탄산나트륨용액 (5 mL)를 첨가하고 교반하였다. 유기층을 모아 염수로 씻어주고 무수황산마그네슘으로 건조하고 농축하였다. 잔사를 컬럼크로마토그래피 (silica gel, 에틸아세테이트:n-헥산 = 2:1 → 디클로메탄:메탄올 = 10:1)로 정제하여 목적화합물 (25 mg, 39 % 수율)을 노란색 고체로 수득하였다.5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-amine (53 mg, 0.1 mmol), dimethylaminopyridine (5 mg, 0.04 mmol) and pyridine (3.2 mL) were added and stirred at 0 ° C. 4- (4-methylpiperazin-1-yl) benzoyl chloride (33 mg, 0.14 mmol) was added and stirred at room temperature for 18 hours. Ethyl acetate (10 mL) was added and saturated sodium carbonate solution (5 mL) was added and stirred. The combined organic layers were washed with brine, dried over anhydrous magnesium sulfate and concentrated. The residue was purified by column chromatography (silica gel, ethyl acetate: n-hexane = 2: 1 → dichloromethane: methanol = 10: 1) to obtain the target compound (25 mg, 39% yield) as a yellow solid.
1H NMR (400 MHz, DMSO-d 6) δ 11.06(s, 1H), 8.94(d, J = 2.24 Hz, 1H), 8.82(d, J = 2.2 Hz, 1H), 7.98(m, 2H), 7.91(m, 1H), 7.86(m, 1H), 7.66(m, 2H), 7.56(m, 2H), 7.10(m, 15H), 6.99(s, 2H), 2.88(m, 4H), 2.44(m, 4H), 2.22(s, 3H), MS m/z : 737.27 [M+1].
1 H NMR (400 MHz, DMSO -d 6 ) δ 11.06 (s, 1H), 8.94 (d, J = 2.24 Hz, 1H), 8.82 (d, J = 2.2 Hz, 1H), 7.98 (m, 2H), 7.91 (m, 1H), 7.86 (m, 1H), 7.66 (m, 2H), 7.56 (m, 2H), 7.10 (m, 15H), 6.99 (s, 2H), 2.88 (m, 4H), 2.44 (m, 4H ), 2.22 (s, 3 H), MS m / z: 737.27 [M + l].
실시예 2∼16Examples 2-16
하기의 화학 반응식과 같이, 상기 실시예 1의 화합물 제조과정의 단계 9에서 합성된 5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-아민과 다양한 아실클로라이드 화합물들 간의 결합반응을 통해서 실시예 2∼16의 목적화합물들을 합성할 수 있다. 본 결합반응의 제조방법은 상기 실시예 1의 화합물 제조과정의 단계 11과 동일하다.As shown in the following chemical scheme, 5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- synthesized in step 9 of the compound preparation process of Example 1 b ] The desired compounds of Examples 2 to 16 can be synthesized through the coupling reaction between pyridin-3-amine and various acyl chloride compounds. The preparation method of the coupling reaction is the same as in step 11 of the compound preparation process of Example 1.
실시예 2 : N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-3-(트리플루오로메틸)벤즈아미드Example 2 N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -3- (trifluoro Rhomethyl) benzamide
MS m/z : 707.17 [M+1]
MS m / z: 707.17 [M + 1]
실시예 3 : N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-4-메톡시벤즈아미드Example 3: N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -4-methoxybenz amides
MS m/z : 669.19 [M+1]
MS m / z: 669.19 [M + 1]
실시예 4 : N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-벤즈아미드Example 4 N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -benzamide
MS m/z : 639.18 [M+1].
MS m / z: 639.18 [M + l].
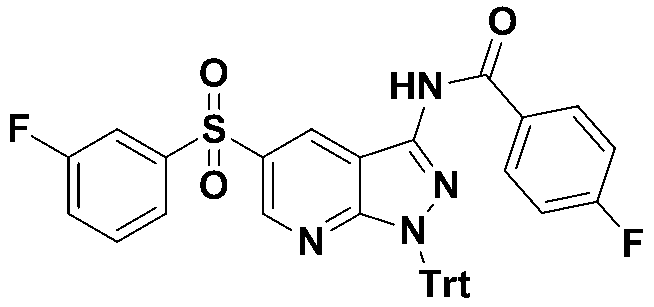
실시예 5 : 4-플루오로-N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-벤즈아미드Example 5: 4-Fluoro- N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl)- Benzamide
MS m/z : 657.17 [M+1]
MS m / z: 657.17 [M + 1]
실시예 6 : 4-클로로-N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-벤즈아미드Example 6 4-Chloro- N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -benz amides
MS m/z : 673.14 [M+1]
MS m / z: 673.14 [M + 1]
실시예 7 : 4-(tert-부틸)-N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-벤즈아미드Example 7: 4- (tert - butyl) - N - (5 - ( ( 3-fluorophenyl) sulfonyl) -1-trityl -1 H - pyrazolo [3,4- b] pyridin-3 Sun) -benzamide
MS m/z : 695.24 [M+1]
MS m / z: 695.24 [M + 1]
실시예 8 : N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-4-몰포리노벤즈아미드Example 8 N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -4-morpholinobenz amides
MS m/z : 724.23 [M+1]
MS m / z: 724.23 [M + l]
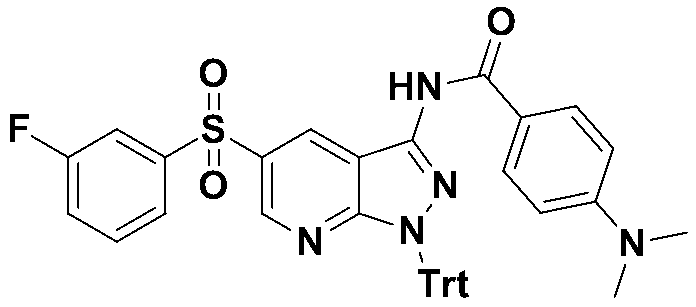
실시예 9 : 4-(디메틸아미노)-N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-벤즈아미드Example 9: 4- (dimethylamino) - N - (5 - ( ( 3-fluorophenyl) sulfonyl) -1-trityl -1 H - pyrazolo [3,4- b] pyridin-3-yl ) -Benzamide
MS m/z : 682.22 [M+1]
MS m / z: 682.22 [M + 1]
실시예 10 : N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-싸이클로헥산카복스아미드Example 10 N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -cyclohexanecarboxamide
MS m/z : 645.23 [M+1]
MS m / z: 645.23 [M + 1]
실시예 11 : N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-피발로아미드Example 11 N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -pivaloamide
MS m/z : 619.21 [M+1]
MS m / z: 619.21 [M + l]
실시예 12 : N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-헥산아미드Example 12 N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -hexanamide
MS m/z : 633.23 [M+1]
MS m / z: 633.23 [M + 1]
실시예 13 : 에틸-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-카바메이트Example 13 ethyl- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -carbamate
MS m/z : 607.18 [M+1]
MS m / z: 607.18 [M + 1]
실시예 14 : tert-부틸-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-카바메이트Example 14 tert -Butyl- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -carbamate
MS m/z : 634.21 [M+1]
MS m / z: 634.21 [M + 1]
실시예 15 : N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-티오펜-2-카복스아미드Example 15 N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -thiophen-2- Carboxamide
MS m/z : 645.14 [M+1].
MS m / z: 645.14 [M + l].
실시예 16 : N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-피페리딘-1-카복스아미드Example 16 N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -piperidine-1 Carboxamide
MS m/z : 646.22 [M+1].
MS m / z: 646.22 [M + l].
실시예 17 : 1-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-3-페닐우레아Example 17 1- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -3-phenylurea
반응용기에 5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-아민 (53 mg, 0.1 mmol)의 THF (1 mL) 혼합용액에 페닐이소시아네이트 (11.8 mg, 0.1 mmol)을 실온에서 첨가하였다. 실온에서 12시간 교반하고 반응액을 감압하에 농축하였다. 잔사를 크로마토그래피 (silica gel, 에틸아세테이트:n-헥산=1:1 → 디클로로메탄:메탄올=20:1)로 정제하여 목적화합물을 수득하였다.THF of 5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-amine (53 mg, 0.1 mmol) in a reaction vessel (1 mL) was added phenyl isocyanate (11.8 mg, 0.1 mmol) at room temperature. The mixture was stirred at room temperature for 12 hours, and the reaction solution was concentrated under reduced pressure. The residue was purified by chromatography (silica gel, ethyl acetate: n-hexane = 1: 1-dichloromethane: methanol = 20: 1) to obtain the target compound.
MS m/z : 654.19 [M+1]
MS m / z: 654.19 [M + 1]
하기의 화학 반응식과 같이, 상기 실시예 1의 화합물 제조과정의 단계 9에서 합성된 5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-아민과 다양한 이소시아네이트 화합물들 간의 결합반응을 통해서 실시예 18∼20의 목적화합물들을 합성할 수 있다. 본 결합반응의 제조방법은 상기 실시예 17의 화합물 제조과정과 동일하다. As shown in the following chemical scheme, 5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- synthesized in step 9 of the compound preparation process of Example 1 b ] The desired compounds of Examples 18 to 20 can be synthesized through the coupling reaction between pyridin-3-amine and various isocyanate compounds. The preparation method of this coupling reaction is the same as the preparation of the compound of Example 17.
실시예 18 : 1-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-3-(2-메톡시페닐)우레아Example 18 1- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -3- (2- Methoxyphenyl) urea
MS m/z : 684.20 [M+1].
MS m / z: 684.20 [M + 1].
실시예 19 : 1-(4-클로로-3-(트리플루오로메틸)페닐)-3-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)우레아Example 19 1- (4-chloro-3- (trifluoromethyl) phenyl) -3- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [ 3,4- b ] pyridin-3-yl) urea
MS m/z : 756.14 [M+1].
MS m / z: 756.14 [M + 1].
실시예 20 : 1-싸이클로헥실-3-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)우레아Example 20 1-cyclohexyl-3- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) urea
MS m/z : 660.24 [M+1]
MS m / z: 660.24 [M + 1]
실시예 21 : N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)-4-(4-메틸피페라진-1-일)벤즈아미드Example 21 N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) -4- (4-methylpiperazin-1 Benzamide
반응용기에 N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-4-(4-메틸피페라진-1-일)벤즈아미드 (22 mg, 0.03 mmol), 디클로로메탄 (1 mL)을 넣고 0 ℃에서 교반하였다. 혼합액에 트리플루오로아세트산 (0.06 mL)을 가하고 실온에서 18시간 교반하였다. 반응액을 농축하고 잔사를 컬럼크로마토그래피 (silica gel, 디클로메탄:메탄올 = 20:1 → 10:1)로 정제하여 목적화합물 (10 mg, 67 % 수율)을 노란색 고체로 수득하였다. N- (5-((3-fluorophenyl) sulfonyl) -1-trityl- 1H -pyrazolo [3,4- b ] pyridin-3-yl) -4- (4-methyl in the reaction vessel Piperazin-1-yl) benzamide (22 mg, 0.03 mmol) and dichloromethane (1 mL) were added and stirred at 0 ° C. Trifluoroacetic acid (0.06 mL) was added to the mixture, and stirred for 18 hours at room temperature. The reaction solution was concentrated and the residue was purified by column chromatography (silica gel, dichloromethane: methanol = 20: 1 to 10: 1) to obtain the target compound (10 mg, 67% yield) as a yellow solid.
1H NMR (400 MHz, DMSO-d 6) δ 11.06(s, 1H), 9.10(d, J = 2.24 Hz, 1H), 9.04(d, J = 2.2 Hz, 1H), 8.00(m, 2H), 7.91(m, 2H), 7.68(m, 1H), 7.56(m, 1H), 7.02(d, J = 9.08 Hz, 2H), 2.49(m, 4H), 2.44(m, 4H), 2.23(s, 3H), MS m/z : 495.16 [M+1].
1 H NMR (400 MHz, DMSO -d 6 ) δ 11.06 (s, 1H), 9.10 (d, J = 2.24 Hz, 1H), 9.04 (d, J = 2.2 Hz, 1H), 8.00 (m, 2H), 7.91 (m, 2H), 7.68 (m, 1H), 7.56 (m, 1H), 7.02 (d, J = 9.08 Hz, 2H), 2.49 (m, 4H), 2.44 (m, 4H), 2.23 (s, 3H), MS m / z: 495.16 [M + 1].
하기의 화학 반응식과 같이, 상기 실시예 1∼20의 화합물은 탈보호반응을 퉁해 실시예 22∼40의 목적화합물들을 합성할 수 있다. 본 결합반응의 제조방법은 상기 실시예 21의 화합물 제조과정과 동일하다. As shown in the following chemical reaction scheme, the compounds of Examples 1 to 20 may be subjected to a deprotection reaction to synthesize target compounds of Examples 22 to 40. The preparation method of this coupling reaction is the same as the preparation of the compound of Example 21.
실시예 22 : N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)-4-(4-메틸피페라진-1-일)-4-메톡시벤즈아미드Example 22 N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) -4- (4-methylpiperazin-1 -Yl) -4-methoxybenzamide
1H NMR (400 MHz, DMSO-d 6) δ 11.23(s, 1H), 9.11(d, J = 2.24 Hz, 1H), 9.07(d, J = 2.2 Hz, 1H), 8.11(m, 2H), 7.92(m, 2H), 7.69(m, 1H), 7.56(m, 1H), 7.09(d, J = 8.92 Hz, 2H), 2.86(s, 3H), MS m/z : 427.08 [M+1].
1 H NMR (400 MHz, DMSO -d 6 ) δ 11.23 (s, 1H), 9.11 (d, J = 2.24 Hz, 1H), 9.07 (d, J = 2.2 Hz, 1H), 8.11 (m, 2H), 7.92 (m, 2H), 7.69 (m, 1H), 7.56 (m, 1H), 7.09 (d, J = 8.92 Hz, 2H), 2.86 (s, 3H), MS m / z: 427.08 [M + 1].
실시예 23 : N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)-3-(트리플루오로메틸)벤즈아미드Example 23 N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) -3- (trifluoromethyl) benzamide
MS m/z : 465.06 [M+1].
MS m / z: 465.06 [M + l].
실시예 24 : N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)벤즈아미드Example 24 N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) benzamide
MS m/z : 397.07 [M+1].
MS m / z: 397.07 [M + l].
실시예 25 : 4-플루오로-N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)벤즈아미드Example 25 4-Fluoro- N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) benzamide
MS m/z : 415.06 [M+1].
MS m / z: 415.06 [M + l].
실시예 26 : 4-클로로-N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)벤즈아미드Example 26 4-chloro- N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) benzamide
MS m/z : 432.03 [M+1].
MS m / z: 432.03 [M + l].
실시예 27 : 4-(tert-부틸)-N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)벤즈아미드Example 27: 4- (tert - butyl) - N - (5 - ( ( 3-fluorophenyl) sulfonyl) -1 H - pyrazolo [3,4- b] pyridin-3-yl) benzamide
MS m/z : 453.14 [M+1].
MS m / z: 453.14 [M + l].
실시예 28 : N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)-4-몰포리노벤즈아미드Example 28 N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) -4-morpholinobenzamide
MS m/z : 482.13 [M+1].
MS m / z: 482.13 [M + l].
실시예 29 : 4-(디메틸아미노)-N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)벤즈아미드Example 29: 4- (dimethylamino) - N - (5 - ( (3- fluorophenyl) sulfonyl) -1 H - pyrazolo [3,4- b] pyridin-3-yl) benzamide
MS m/z : 440.11 [M+1].
MS m / z: 440.11 [M + l].
실시예 30 : N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)싸이클로헥산카복스아미드Example 30 N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) cyclohexanecarboxamide
MS m/z : 403.12 [M+1].
MS m / z: 403.12 [M + l].
실시예 31 : N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)피발아미드Example 31 N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) pivalamide
MS m/z : 377.10 [M+1].
MS m / z: 377.10 [M + 1].
실시예 32 : N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)헥산아미드Example 32 N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) hexanamide
MS m/z : 391.12 [M+1].
MS m / z: 391.12 [M + l].
실시예 33 : 에틸-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)카바메이트Example 33 ethyl- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) carbamate
MS m/z : 365.07 [M+1].
MS m / z: 365.07 [M + l].
실시예 34 : tert-부틸-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)카바메이트Example 34 tert -Butyl- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) carbamate
MS m/z : 393.10 [M+1].
MS m / z: 393.10 [M + 1].
실시예 35 : N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)티오펜-2-카복스아미드Example 35 N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) thiophene-2-carboxamide
MS m/z : 403.03 [M+1].
MS m / z: 403.03 [M + l].
실시예 36 : N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)피페리딘-1-카복스아미드Example 36 N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) piperidine-1-carboxamide
MS m/z : 404.11 [M+1].
MS m / z: 404.11 [M + 1].
실시예 37 : 1-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)-3-페닐우레아Example 37 1- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) -3-phenylurea
MS m/z : 412.08 [M+1].
MS m / z: 412.08 [M + l].
실시예 38 : 1-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)-3-(2-메톡시페닐)우레아Example 38 1- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) -3- (2-methoxyphenyl) urea
MS m/z : 442.09 [M+1].
MS m / z: 442.09 [M + l].
실시예 39 : 1-(4-클로로-3-(트리플루오로메틸)페닐)-3-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)우레아Example 39 1- (4-chloro-3- (trifluoromethyl) phenyl) -3- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] Pyridin-3-yl) urea
MS m/z : 515.03 [M+1].
MS m / z: 515.03 [M + l].
실시예 40 : 1-싸이클로헥실-3-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)우레아Example 40 1-cyclohexyl-3- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) urea
MS m/z : 418.13 [M+1].
MS m / z: 418.13 [M + l].
실시예 41 : 5-((3-플루오로페닐)설포닐)-N-(4-몰포리노페닐)-1-(테트로하이드로-2H-피란-2-일)-1H-인다졸-3-카복스아미드Example 41: 5 - ((3-fluorophenyl) sulfonyl) - N - (4- Dimorpholino Reno phenyl) -1- (tetrahydro -2 H in Tet-pyran-2-yl) -1 H-indazole -3-carboxamide
상기의 구조식으로 표기되는 실시예 41의 화합물은 하기와 같이 9단계의 합성과정을 통해 제조할 수 있다.
Compound of Example 41 represented by the above structural formula can be prepared through a nine-step synthesis process as follows.
단계 1: 6-브로모-1H-인다졸-3-카르복실산Step 1: 6-Bromo-1 H -indazol-3-carboxylic acid
반응용기에 6-브로모이사틴 (10.0 g, 22 mmol), 1N NaOH 수용액 (48 mL)를 투입하고 50 ℃로 가온하여 1시간 교반하였다. 반응액을 0 ℃로 냉각하고 아질산나트륨 (3.0 g, 22 mmol)과 물 (11 mL)의 혼합액을 천천히 가하였다. 같은 온도에서 혼합액에 물 (90 mL)와 황산 (4.6 mL)를 천천히 가하고 교반하였다. 반응액에 염화주석 이수화물 (24.0 g, 53 mmol)과 염산 (40 mL)의 혼합액을 0 ℃에서 가하고 실온에서 교반하였다. 생성된 고체를 여과하고 물로 세척하고 건조하여 목적화합물 (8.53 g, 80 % 수율)을 노란색 고체로 수득하였다.6-bromoissatin (10.0 g, 22 mmol) and 1 N aqueous NaOH solution (48 mL) were added to the reaction vessel, which was then heated to 50 ° C. and stirred for 1 hour. The reaction solution was cooled to 0 ° C. and a mixture of sodium nitrite (3.0 g, 22 mmol) and water (11 mL) was slowly added. Water (90 mL) and sulfuric acid (4.6 mL) were slowly added to the mixture at the same temperature and stirred. A mixture of tin chloride dihydrate (24.0 g, 53 mmol) and hydrochloric acid (40 mL) was added to the reaction solution at 0 ° C., and stirred at room temperature. The resulting solid was filtered, washed with water and dried to give the desired compound (8.53 g, 80% yield) as a yellow solid.
1H NMR (400 MHz, DMSO-d 6) δ 13.99(s, 1H), 13.00(bs, 1H), 8.21(s, 1H), 7.65(d, J = 8 Hz, 1H), 7.57(d, J = 8 Hz, 1H).
1 H NMR (400 MHz, DMSO -d 6 ) δ 13.99 (s, 1H), 13.00 (bs, 1H), 8.21 (s, 1H), 7.65 (d, J = 8 Hz, 1H), 7.57 (d, J = 8 Hz, 1H).
단계 2: 에틸 6-브로모-1H-인다졸-3-카복실레이트Step 2: ethyl 6-bromo-1 H -indazol-3-carboxylate
반응용기에 6-브로모-1H-인다졸-3-카르복실산 (5.0 g, 21 mmol)과 에탄올(100 mL)을 투입하고 교반하였다. 혼합액에 황산 (5 mL)를 가하고 환류교반하였다. 반응액을 실온으로 냉각하고 농축하였다. 잔사에 에틸아세테이트와 포화탄산수소나트륨 용액을 가하고 교반하였다. 유기층을 모아 무수황산마그네슘으로 건조하고 농축하였다. 잔사를 컬럼크로마토그래피 (silica gel, 디클로로메탄:메탄올 = 9:1)로 정제하여 목적화합물 (2.8 g)을 노란색 고체로 수득하였다.6-Bromo- 1H -indazole-3-carboxylic acid (5.0 g, 21 mmol) and ethanol (100 mL) were added to the reaction vessel, followed by stirring. Sulfuric acid (5 mL) was added to the mixture, followed by stirring under reflux. The reaction solution was cooled to room temperature and concentrated. Ethyl acetate and saturated sodium bicarbonate solution were added to the residue, followed by stirring. The organic layers were combined, dried over anhydrous magnesium sulfate and concentrated. The residue was purified by column chromatography (silica gel, dichloromethane: methanol = 9: 1) to give the target compound (2.8 g) as a yellow solid.
1H NMR (400 MHz, CDCl3) δ 14.04(bs, 1H), 8.00(d, J = 8.4 Hz, 1H), 7.91(d, J = 1.2 Hz, 1H), 7.45(dd, J =1.5, 8.7 Hz, 1H), 4.39(q, J = 7.2 Hz, 2H), 1.35(t, J = 7.2 Hz, 3H).
1 H NMR (400 MHz, CDCl 3 ) δ 14.04 (bs, 1H), 8.00 (d, J = 8.4 Hz, 1H), 7.91 (d, J = 1.2 Hz, 1H), 7.45 (dd, J = 1.5, 8.7 Hz, 1H) , 4.39 (q, J = 7.2 Hz, 2H), 1.35 (t, J = 7.2 Hz, 3H).
단계 3: 에틸 5-브로모-1-(테트라하이드로-2H-피란-2-일)-1H-인다졸-3-카복실레이트Step 3: ethyl 5-bromo-1- (tetrahydro-2 H -pyran-2-yl) -1 H -indazol-3-carboxylate
반응용기에 에틸 6-브로모-1H-인다졸-3-카복실레이트 (4.84 g, 18 mmol), 3,4-디하이드로-2H-피란 (3.03 g, 36 mmol)과 아세토나이트릴 (50 mL)을 투입하고 교반하였다. 혼합액에 p-톨루엔설폰산 (0.31 g, 1.8 mmol)을 가하고 실온에서 교반하였다. 반응액을 농축하고 잔사를 컬럼크로마토그래피 (silica gel, 에틸아세테이트:n-헥산 = 3:1)로 정제하여 목적화합물 (3.8 g, 60% 수율)을 노란색 고체로 수득하였다.Ethyl 6-bromo-1 H -indazole-3-carboxylate (4.84 g, 18 mmol), 3,4-dihydro-2 H -pyran (3.03 g, 36 mmol) and acetonitrile ( 50 mL) was added and stirred. P - toluenesulfonic acid (0.31 g, 1.8 mmol) was added to the mixture, and the mixture was stirred at room temperature. The reaction solution was concentrated and the residue was purified by column chromatography (silica gel, ethyl acetate: n-hexane = 3: 1) to obtain the target compound (3.8 g, 60% yield) as a yellow solid.
1H NMR (400 MHz, DMSO-d 6) δ 8.21(d, J = 8.6 Hz, 1H), 7.89(d, J = 8.95 Hz, 1H), 7.66(m, 1H), 6.01(m, 1H), 4.42(m, 2H), 3.88(m, 1H), 3.79(m, 1H), 2.33(m, 1H), 2.00(m, 2H), 1.75(m, 1H), 1.60(m, 2H), 1.37(t, 3H).
1 H NMR (400 MHz, DMSO -d 6 ) δ 8.21 (d, J = 8.6 Hz, 1H), 7.89 (d, J = 8.95 Hz, 1H), 7.66 (m, 1H), 6.01 (m, 1H), 4.42 (m, 2H), 3.88 (m, 1H), 3.79 (m, 1H), 2.33 (m, 1H), 2.00 (m, 2H), 1.75 (m, 1H), 1.60 (m, 2H), 1.37 (t, 3H) ).
단계 4: 에틸 5-((3-플루오로페닐)티오)-1-(테트라하이드로-2H-피란-2-일)-1H-인다졸-3-카복실레이트Step 4: ethyl 5-((3-fluorophenyl) thio) -1- (tetrahydro- 2H -pyran-2-yl) -1 H -indazole-3-carboxylate
밀폐용기에 에틸 5-브로모-1-(테트라하이드로-2H-피란-2-일)-1H-인다졸-3-카복실레이트 (176 mg, 0.5 mmol)과 Xantphos (29 mg, 0.05 mmol), Pd2(dba)3 (23 mg, 0.025 mmol), 1,4-디옥산 (2 mL)을 넣었다. 혼합용액에 있는 기체를 초음파와 질소기체를 이용하여 제거하였다. 디이소프로필에틸아민 (0.18 mL, 1.0 mmol)과 3-플루오로티오페놀 (0.042 mL, 0.5 mmol)을 첨가하고 밀폐시켜 110 ℃에서 36시간 교반하였다. 반응용액을 실온으로 냉각하여 규조토 패드로 여과하고 에틸아세테이트로 여러번 세척하였다. 유기층에 물을 가하여 추출하였다. 유기층을 분리하고 물층을 에틸아세테이트로 추출하였다. 모아진 유기층을 무수황산마그네슘으로 건조하고 농축하였다. 잔사를 크로마토그래피 (silica gel, 에틸아세테이트:n-헥산=6:1→4:1)로 정제하여 목적화합물 (131 mg, 65% 수율)을 노란색 고체로 수득하였다. In an airtight container, ethyl 5-bromo-1- (tetrahydro-2 H -pyran-2-yl) -1 H -indazole-3-carboxylate (176 mg, 0.5 mmol) and Xantphos (29 mg, 0.05 mmol) ), Pd 2 (dba) 3 (23 mg, 0.025 mmol), 1,4-dioxane (2 mL) was added. The gas in the mixed solution was removed using ultrasonic waves and nitrogen gas. Diisopropylethylamine (0.18 mL, 1.0 mmol) and 3-fluorothiophenol (0.042 mL, 0.5 mmol) were added and sealed and stirred at 110 ° C. for 36 hours. The reaction solution was cooled to room temperature, filtered through a pad of diatomaceous earth, and washed several times with ethyl acetate. Water was added to the organic layer and extracted. The organic layer was separated and the water layer was extracted with ethyl acetate. The combined organic layers were dried over anhydrous magnesium sulfate and concentrated. The residue was purified by chromatography (silica gel, ethyl acetate: n-hexane = 6: 1-4: 1) to obtain the target compound (131 mg, 65% yield) as a yellow solid.
1H NMR (400 MHz, DMSO-d 6) δ 8.15(d, J = 1.50 Hz, 1H), 7.97(d, J = 8.83 Hz, 1H), 7.57(m, 1H), 7.39(m, 1H), 7.11(m, 1H), 7.04(m, 2H), 4.38(m, 2H), 3.91(m, 1H), 3.79(m, 1H), 2.36(m, 1H), 2.03(m, 2H), 1.76(m, 1H), 1.60(m, 2H), 1.32(t, 3H).
1 H NMR (400 MHz, DMSO -d 6 ) δ 8.15 (d, J = 1.50 Hz, 1H), 7.97 (d, J = 8.83 Hz, 1H), 7.57 (m, 1H), 7.39 (m, 1H), 7.11 (m, 1H), 7.04 (m, 2H), 4.38 (m, 2H), 3.91 (m, 1H), 3.79 (m, 1H), 2.36 (m, 1H), 2.03 (m, 2H), 1.76 (m, 1H) ), 1.60 (m, 2 H), 1.32 (t, 3 H).
단계 5: 에틸 5-((3-플루오로페닐)설포닐)-1-(테트라하이드로-2H-피란-2-일)-1H-인다졸-3-카복실레이트Step 5: Ethyl 5-((3-fluorophenyl) sulfonyl) -1- (tetrahydro- 2H -pyran-2-yl) -1 H -indazol-3-carboxylate
반응용기에 에틸 5-((3-플루오로페닐)티오)-1-(테트라하이드로-2H-피란-2-일)-1H-인다졸-3-카복실레이트 (125 mg, 0.32 mmol), 디클로로메탄 (2 mL)을 가하였다. 실온에서 m-클로로퍼벤조산 (mCPBA(72%), 244 mg, 1.09 mmol)을 가하고 3시간동안 교반하였다. 반응액에 디클로로메탄 (3 mL)을 가하고 0 ℃로 냉각하여 10% 아황산나트륨 수용액 (3 mL)을 가하고 교반하였다. 수층을 제거하고 유기층을 모아 포화 탄산나트륨 수용액, 염수로 차례로 씻어주었다. 유기층을 무수황산마그네슘으로 건조하고 농축하여 목적화합물 (130 mg, 93% 수율)을 흰색 고체로 수득하였다.To the reaction vessel of ethyl 5 - ((3-phenylpropyl) thio-fluorophenyl) -1- (tetrahydro -2 H-pyran-2-yl) -1 H-indazole-3-carboxylate (125 mg, 0.32 mmol) , Dichloromethane (2 mL) was added. M -Chloroperbenzoic acid (mCPBA (72%), 244 mg, 1.09 mmol) was added at room temperature and stirred for 3 hours. Dichloromethane (3 mL) was added to the reaction mixture, and the mixture was cooled to 0 ° C., followed by addition of 10% aqueous sodium sulfite solution (3 mL), followed by stirring. The aqueous layer was removed, the organic layers were collected and washed sequentially with saturated aqueous sodium carbonate solution and brine. The organic layer was dried over anhydrous magnesium sulfate and concentrated to give the target compound (130 mg, 93% yield) as a white solid.
1H NMR (400 MHz, DMSO-d 6) δ 8.72(s, 1H), 8.08(m, 2H), 7.87(m, 2H), 7.69(m, 1H), 7.58(m, 1H), 6.08(m, 1H), 4.45(m, 2H), 3.87(m, 1H), 3.78(m, 1H), 2.34(m, 1H), 2.02(m, 2H), 1.74(m, 1H), 1.60(m, 2H), 1.40(t, 3H).
1 H NMR (400 MHz, DMSO -d 6 ) δ 8.72 (s, 1H), 8.08 (m, 2H), 7.87 (m, 2H), 7.69 (m, 1H), 7.58 (m, 1H), 6.08 (m, 1H), 4.45 (m, 2H), 3.87 (m, 1H), 3.78 (m, 1H), 2.34 (m, 1H), 2.02 (m, 2H), 1.74 (m, 1H), 1.60 (m, 2H), 1.40 (t, 3H).
단계 6: 5-((3-플루오로페닐)설포닐)-1-(테트라하이드로-2H-피란-2-일)-1H-인다졸-3-카복실산Step 6: 5-((3-fluorophenyl) sulfonyl) -1- (tetrahydro- 2H -pyran-2-yl) -1 H -indazole-3-carboxylic acid
반응용기에 에틸 5-((3-플루오로페닐)설포닐)-1-(테트라하이드로-2H-피란-2-일)-1H-인다졸-3-카복실레이트 (130 mg, 0.3 mmol), 테트라하이드로퓨란 (10 mL), 물 (6 mL), 메탄올 (1 mL)의 혼합액을 가하였다. 혼합액에 수산화리튬 수화물 (28 mg, 0.66 mmol)을 가하고 55 ℃에서 16시간동안 교반하였다. 반응액을 실온으로 냉각하고 농축하였다. 잔사에 1N 염산용액을 천천히 가하여 pH 5를 만들었다. 에틸아세테이트와 염수를 가하고 교반하였다. 유기층을 모아 무수 황산마그네슘으로 건조하고 농축하여 목적화합물 (120 mg, 98% 수율)을 흰색 고체로 수득하였다. 상기 목적화합물은 정제없이 바로 다음 반응에 사용하였다. 5 to a reaction vessel ethyl - ((3-fluorophenyl) sulfonyl) -1- (tetrahydro -2 H-pyran-2-yl) -1 H-indazole-3-carboxylate (130 mg, 0.3 mmol ), Tetrahydrofuran (10 mL), water (6 mL), methanol (1 mL) was added. Lithium hydroxide hydrate (28 mg, 0.66 mmol) was added to the mixture, and the mixture was stirred at 55 ° C. for 16 hours. The reaction solution was cooled to room temperature and concentrated. 1 N hydrochloric acid solution was slowly added to the residue to make pH 5. Ethyl acetate and brine were added and stirred. The combined organic layers were dried over anhydrous magnesium sulfate and concentrated to give the target compound (120 mg, 98% yield) as a white solid. The target compound was used for the next reaction without purification.
MS m/z : 405.09 [M+1].
MS m / z: 405.09 [M + l].
단계 7: 4-(4-니트로페닐)몰포린Step 7: 4- (4-nitrophenyl) morpholine
반응용기에 1-플루오로-4-나이트로벤젠 (1 g, 7.09 mmol)과 디메틸포름아미드 (10 mL)를 넣었다. 혼합액에 몰포린 (0.62 g, 7.09 mmol)과 탄산칼륨 (1.96 g, 14.17 mmol)를 가하고 80 ℃로 가온교반하였다. 반응액을 실온으로 냉각하고 에틸아세테이트, 물을 가하고 교반하였다. 수층을 에틸아세테이트로 씻어주었다. 유기층을 모아 염수로 씻어주었다. 무수황산마그네슘으로 건조하고 농축하여 목적화합물 (1.18 g, 80% 수율)을 노란색 고체로 수득하였다. 상기 화합물은 정제없이 바로 다음 반응에 사용하였다. 1-fluoro-4-nitrobenzene (1 g, 7.09 mmol) and dimethylformamide (10 mL) were added to the reaction vessel. Morpholine (0.62 g, 7.09 mmol) and potassium carbonate (1.96 g, 14.17 mmol) were added to the mixture, and the mixture was warmed and stirred at 80 ° C. The reaction solution was cooled to room temperature, ethyl acetate and water were added and stirred. The aqueous layer was washed with ethyl acetate. The organic layer was collected and washed with brine. Drying with anhydrous magnesium sulfate and concentration gave the desired compound (1.18 g, 80% yield) as a yellow solid. The compound was used directly in the next reaction without purification.
MS m/z : 209.09 [M+1].
MS m / z: 209.09 [M + l].
단계 8: 4-몰포리노아닐린Step 8: 4-morpholinoaniline
반응용기에 4-(4-니트로페닐)몰포린 (1 g, 4.80 mmol)과 메탄올 (30 mL)을 넣었다. 혼합액에 10% Pd/C (100 mg)를 가하고 수소기체를 채운 풍선 압력하에서 실온에서 교반하였다. 반응액을 규조토 패드로 여과하고 농축하였다. 얻어진 목적화합물 (728 mg, 85% 수율)은 정제없이 다음 반응에 사용하였다. 4- (4-nitrophenyl) morpholine (1 g, 4.80 mmol) and methanol (30 mL) were added to the reaction vessel. 10% Pd / C (100 mg) was added to the mixture, and the mixture was stirred at room temperature under a balloon pressure filled with hydrogen gas. The reaction solution was filtered through a pad of diatomaceous earth and concentrated. The desired compound (728 mg, 85% yield) was used in the next reaction without purification.
MS m/z : 179.11 [M+1].
MS m / z: 179.11 [M + l].
단계 9: 5-((3-플루오로페닐)설포닐)-N-(4-몰포리노페닐)-1-(테트로하이드로-2H-피란-2-일)-1H-인다졸-3-카복스아미드Step 9: 5 - ((3-fluorophenyl) sulfonyl) - N - (4- Dimorpholino Reno phenyl) -1- (tetrahydro -2 H in Tet-pyran-2-yl) -1 H-indazol- 3-carboxamide
반응용기에 5-((3-플루오로페닐)설포닐)-1-(테트라하이드로-2H-피란-2-일)-1H-인다졸-3-카복실산 (40 mg, 0.1 mmol), 4-몰포리노아닐린 (29 mg, 0.16 mmol), 디이소프로필에틸아민 (0.035 mL, 0.2 mmol), 1-하이드록시벤조트리아졸 일수화물 (19 mg, 0.12 mmol)과 디클로로메탄 (2 mL)을 넣고 교반하였다. 1-에틸-3-(3-디메틸아미노프로필) 카르보디이미드 염산염 (EDCI??HCl, 23 mg, 0.12 mmol)을 가하고 실온에서 교반하였다. 디클로로메탄 (10 mL)으로 반응액을 희석하고 물 (10 mL)을 가하고 교반하였다. 유기층을 염수로 씻어주고 무수황산마그네슘으로 건조하고 농축하였다. 잔사를 컬럼크로마토그래피 (silica gel, 디클로로메탄:메탄올 = 30:1)로 정제하여 목적화합물 (50 mg, 90% 수율)을 수득하였다. 5-((3-fluorophenyl) sulfonyl) -1- (tetrahydro- 2H -pyran-2-yl) -1 H -indazole-3-carboxylic acid (40 mg, 0.1 mmol) in the reaction vessel, 4-morpholinoaniline (29 mg, 0.16 mmol), diisopropylethylamine (0.035 mL, 0.2 mmol), 1-hydroxybenzotriazole monohydrate (19 mg, 0.12 mmol) and dichloromethane (2 mL) Put and stir. 1-ethyl-3- (3-dimethylaminopropyl) carbodiimide hydrochloride (EDCI ?? HCl, 23 mg, 0.12 mmol) was added and stirred at room temperature. The reaction was diluted with dichloromethane (10 mL), water (10 mL) was added and stirred. The organic layer was washed with brine, dried over anhydrous magnesium sulfate and concentrated. The residue was purified by column chromatography (silica gel, dichloromethane: methanol = 30: 1) to obtain the target compound (50 mg, 90% yield).
1H NMR (400 MHz, CDCl3) δ 9.17(d, J = 1.6 Hz, 1H), 8.67(s, 1H), 8.02(m, 1H), 7.79(m, 2H), 7.69(m, 4H), 7.49(m, 1H), 7.22(m, 1H), 6.97(m, 2H), 5.81(m, 1H), 4.15(m, 1H), 3.90(m, 4H), 3.81(m, 1H), 3.20 (m, 4H), 2.58(m, 1H), 2.17(m, 2H), 1.81(m, 3H), MS m/z : 565.19 [M+1].
1 H NMR (400 MHz, CDCl 3 ) δ 9.17 (d, J = 1.6 Hz, 1H), 8.67 (s, 1H), 8.02 (m, 1H), 7.79 (m, 2H), 7.69 (m, 4H), 7.49 (m, 1H), 7.22 (m, 1H), 6.97 (m, 2H), 5.81 (m, 1H), 4.15 (m, 1H), 3.90 (m, 4H), 3.81 (m, 1H), 3.20 (m, 4H ), 2.58 (m, 1H), 2.17 (m, 2H), 1.81 (m, 3H), MS m / z: 565.19 [M + 1].
상기 실시예 41의 화합물 제조과정의 단계 6에서 합성된 5-((3-플루오로페닐)설포닐)-1-(테트라하이드로-2H-피란-2-일)-1H-인다졸-3-카복실산과 다양한 아닐린 화합물들 간의 결합반응을 통해서 실시예 42∼45의 목적화합물들을 합성할 수 있다. 본 결합반응의 제조방법은 상기 실시예 40의 화합물 제조과정과 동일하다.
5-((3-fluorophenyl) sulfonyl) -1- (tetrahydro- 2H -pyran-2-yl) -1 H -indazole- synthesized in step 6 of the compound preparation of Example 41 above The desired compounds of Examples 42 to 45 can be synthesized through the coupling reaction between 3-carboxylic acid and various aniline compounds. The preparation method of this coupling reaction is the same as the preparation of the compound of Example 40.
실시예 42 : 5-((3-플루오로페닐)설포닐)-N-(6-몰포리노피리딘-3-일)-1-(테트로하이드로-2H-피란-2-일)-1H-인다졸-3-카복스아미드Example 42: 5 - ((3-fluorophenyl) sulfonyl) - N - (6- Reno Dimorpholino-3-yl) -1- (tetrahydro -2 H in Tet-pyran-2-yl) -1 H -indazol-3-carboxamide
1H NMR (400 MHz, CDCl3) δ 9.14(s, 1H), 8.62(s, 1H), 8.02(m, 1H), 8.35(m, 1H), 8.17(m, 1H), 8.01(m, 1H), 7.79(m, 2H), 7.69(m, 1H), 7.49(m, 1H), 7.22(m, 1H), 6.73(m, 1H), 5.82(m, 1H), 4.15(m, 1H), 3.90(m, 4H), 3.81(m, 1H), 3.20 (m, 4H), 2.58(m, 1H), 2.18(m, 2H), 1.80(m, 3H), MS m/z : 566.18 [M+1].
1 H NMR (400 MHz, CDCl 3 ) δ 9.14 (s, 1H), 8.62 (s, 1H), 8.02 (m, 1H), 8.35 (m, 1H), 8.17 (m, 1H), 8.01 (m, 1H), 7.79 ( m, 2H), 7.69 (m, 1H), 7.49 (m, 1H), 7.22 (m, 1H), 6.73 (m, 1H), 5.82 (m, 1H), 4.15 (m, 1H), 3.90 (m , 4H), 3.81 (m, 1H), 3.20 (m, 4H), 2.58 (m, 1H), 2.18 (m, 2H), 1.80 (m, 3H), MS m / z: 566.18 [M + 1] .
실시예 43 : N-(4-(4-에틸피페라진-1-일)페닐)-5-((3-플루오로페닐)설포닐)-1-(테트로하이드로-2H-피란-2-일)-1H-인다졸-3-카복스아미드Example 43 N- (4- (4-ethylpiperazin-1-yl) phenyl) -5-((3-fluorophenyl) sulfonyl) -1- (tetrohydro- 2H -pyran-2 -Yl) -1 H -indazole-3-carboxamide
MS m/z : 592.23 [M+1].
MS m / z: 592.23 [M + l].
실시예 44 : 5-((3-플루오로페닐)설포닐)-N-(6-메틸피리딘-3-일)-1-(테트로하이드로-2H-피란-2-일)-1H-인다졸-3-카복스아미드Example 44: 5 - ((3-fluorophenyl) sulfonyl) - N - (6- methylpyridin-3-yl) -1- (Tet as dihydro -2 H-pyran-2-yl) -1 H -Indazole-3-carboxamide
MS m/z : 495.15 [M+1].
MS m / z: 495.15 [M + 1].
실시예 45 : N-(4-클로로-3-(트리프루오로메틸)페닐))-5-((3-플루오로페닐)설포닐)-1-(테트로하이드로-2H-피란-2-일)-1H-인다졸-3-카복스아미드Example 45 N- (4-Chloro-3- (trifluoromethyl) phenyl))-5-((3-fluorophenyl) sulfonyl) -1- (tetrohydro- 2H -pyran- 2-yl) -1 H -indazol-3-carboxamide
MS m/z : 583.08 [M+1].
MS m / z: 583.08 [M + l].
실시예 46 : 5-((3-플루오로페닐)설포닐)-N-(4-몰포리노페닐)-1H-인다졸-3-카복스아미드Example 46: 5 - ((3-fluorophenyl) sulfonyl) - N - (4- Dimorpholino Reno phenyl) -1 H-indazole-3-carboxamide
반응용기에 5-((3-플루오로페닐)설포닐)-N-(4-몰포리노페닐)-1-(테트로하이드로-2H-피란-2-일)-1H-인다졸-3-카복스아미드 (46.5 mg, 0.082 mmol), p-톨루엔설폰산 일수화물 (233 mg, 1.23 mmol)과 에탄올 (4.8 mL)을 넣고 교반하였다. 혼합액에 아세트산 (2.2 mL)를 가하고 80 ℃로 가온하여 교반하였다. 반응액을 실온으로 냉각하고 농축하였다. 잔사를 컬럼크로마토그래피 (silica gel, 디클로로메탄:메탄올 = 50:1 → 30:1)로 정제하여 목적화합물(19 mg)을 흰색 고체로 수득하였다. To the reaction vessel 5 - ((3-fluorophenyl) sulfonyl) - N - (4- Dimorpholino Reno phenyl) -1- (tetrahydro -2 H in Tet-pyran-2-yl) -1 H-indazol- 3-carboxamide (46.5 mg, 0.082 mmol), p- toluenesulfonic acid monohydrate (233 mg, 1.23 mmol) and ethanol (4.8 mL) were added thereto and stirred. Acetic acid (2.2 mL) was added to the mixed solution, which was then heated to 80 ° C and stirred. The reaction solution was cooled to room temperature and concentrated. The residue was purified by column chromatography (silica gel, dichloromethane: methanol = 50: 1-> 30: 1) to obtain the target compound (19 mg) as a white solid.
1H NMR (400 MHz, DMSO-d 6) δ 14.25(bs, 1H), 10.37(s, 1H), 8.88(d, J = 0.8 Hz, 1H), 7.98(m, 1H), 7.85(m, 3H), 7.76(m, 2H), 7.68(m, 1H), 7.56(m, 1H), 6.95(m, 2H), 3.75(m, 4H), 3.09(m, 4H), MS m/z : 481.13 [M+1].
1 H NMR (400 MHz, DMSO -d 6 ) δ 14.25 (bs, 1H), 10.37 (s, 1H), 8.88 (d, J = 0.8 Hz, 1H), 7.98 (m, 1H), 7.85 (m, 3H), 7.76 ( m, 2H), 7.68 (m, 1H), 7.56 (m, 1H), 6.95 (m, 2H), 3.75 (m, 4H), 3.09 (m, 4H), MS m / z: 481.13 [M + 1 ].
상기 실시예 42∼46의 화합물은 탈보호반응을 통해 실시예 47∼50의 목적화합물들을 합성할 수 있다. 본 결합반응의 제조방법은 상기 실시예 46의 화합물 제조과정과 동일하다.
The compounds of Examples 42 to 46 can synthesize the target compounds of Examples 47 to 50 through a deprotection reaction. The preparation method of this coupling reaction is the same as the preparation of the compound of Example 46.
실시예 47 : 5-((3-플루오로페닐)설포닐)-N-(4-몰포리노피리딘-3-일)-1H-인다졸-3-카복스아미드Example 47: 5 - ((3-fluorophenyl) sulfonyl) - N - (4- Reno Dimorpholino-3-yl) -1 H-indazole-3-carboxamide
1H NMR (400 MHz, DMSO-d 6) δ 14.28(bs, 1H), 10.50(s, 1H), 8.88(s, 1H), 8.61(m, 1H), 8.06(m, 1H), 7.98(m, 1H), 7.86(m, 3H), 7.69(m, 1H), 7.56(m, 1H), 6.88(m, 1H), 3.72(m, 4H), 3.42(m, 4H), MS m/z : 482.13 [M+1].
1 H NMR (400 MHz, DMSO -d 6 ) δ 14.28 (bs, 1H), 10.50 (s, 1H), 8.88 (s, 1H), 8.61 (m, 1H), 8.06 (m, 1H), 7.98 (m, 1H), 7.86 (m, 3H), 7.69 (m, 1H), 7.56 (m, 1H), 6.88 (m, 1H), 3.72 (m, 4H), 3.42 (m, 4H), MS m / z: 482.13 [M +1].
실시예 48 : N-(4-(4-에틸피페라진-1-일)페닐)-5-((3-플루오로페닐)설포닐)-1H-인다졸-3-카복스아미드Example 48 N- (4- (4-ethylpiperazin-1-yl) phenyl) -5-((3-fluorophenyl) sulfonyl) -1 H -indazole-3-carboxamide
MS m/z : 508.18 [M+1].
MS m / z: 508.18 [M + l].
실시예 49 : 5-((3-플루오로페닐)설포닐)-N-(6-메틸피리딘-3-일)-1H-인다졸-3-카복스아미드Example 49: 5 - ((3-fluorophenyl) sulfonyl) - N - (6- methylpyridin-3-yl) -1 H-indazole-3-carboxamide
MS m/z : 411.09 [M+1].
MS m / z: 411.09 [M + l].
실시예 50 : N-(4-클로로-3-(트리플루오로메틸)페닐)-5-((3-플루오로페닐)설포닐)-1H-인다졸-3-카복스아미드Example 50 N- (4-chloro-3- (trifluoromethyl) phenyl) -5-((3-fluorophenyl) sulfonyl) -1 H -indazole-3-carboxamide
MS m/z : 499.02 [M+1]
MS m / z: 499.02 [M + 1]
한편, 본 발명에 따른 상기 화학식 1로 표시되는 신규 화합물은 목적에 따라 여러 형태로 제제화가 가능하다. 다음은 본 발명에 따른 상기 화학식 1로 표시되는 화합물을 활성성분으로 함유시킨 몇몇 제제화 방법을 예시한 것으로 본 발명이 이에 한정되는 것은 아니다.
Meanwhile, the novel compounds represented by Formula 1 according to the present invention can be formulated into various forms according to the purpose. The following is a description of some formulations containing the compound of Formula 1 according to the present invention as an active ingredient, but the present invention is not limited thereto.
[제제예]
[Formulation Example]
제제예 1. 정제(직접 가압)Formulation Example 1 Tablets (direct pressurization)
활성성분 5.0 ㎎을 체로 친 후, 락토스 14.1 ㎎, 크로스포비돈 USNF 0.8 ㎎ 및 마그네슘 스테아레이트 0.1 ㎎을 혼합하고 가압하여 정제로 만들었다.
After 5.0 mg of the active ingredient was sieved, 14.1 mg of lactose, 0.8 mg of crospovidone USNF and 0.1 mg of magnesium stearate were mixed and pressurized to make tablets.
제제예 2. 정제(습식 조립)Formulation Example 2. Tablet (wet assembly)
활성성분 5.0 ㎎을 체로 친 후, 락토스 16.0 ㎎과 녹말 4.0 ㎎을 섞었다. 폴리솔베이트 80 0.3 ㎎을 순수한 물에 녹인 후 이 용액의 적당량을 첨가한 다음, 미립화하였다. 건조 후에 미립을 체질한 후 콜로이달 실리콘 디옥사이드 2.7 ㎎ 및 마그네슘 스테아레이트 2.0 ㎎과 섞었다. 미립을 가압하여 정제로 만들었다.
After 5.0 mg of the active ingredient was sieved, 16.0 mg of lactose and 4.0 mg of starch were mixed. 0.3 mg of Polysorbate 80 was dissolved in pure water, and an appropriate amount of this solution was added, followed by atomization. After drying, the granules were sieved and mixed with 2.7 mg of colloidal silicon dioxide and 2.0 mg of magnesium stearate. The granules were pressurized to make tablets.
제제예 3. 분말과 캡슐제Formulation Example 3 Powders and Capsules
활성성분 5.0 ㎎을 체로 친 후에, 락토스 14.8 ㎎, 폴리비닐 피롤리돈 10.0 ㎎, 마그네슘 스테아레이트 0.2 ㎎와 함께 섞었다. 혼합물을 적당한 장치를 사용하여 단단한 No. 5 젤라틴 캡슐에 채웠다.
5.0 mg of the active ingredient was sieved and mixed with 14.8 mg of lactose, 10.0 mg of polyvinylpyrrolidone and 0.2 mg of magnesium stearate. The mixture was filtered through a hard No. 5 gelatin capsules.
제제예 4. 주사제Formulation Example 4 Injection
활성성분으로서 100 mg을 함유시키고, 그 밖에도 만니톨 180 mg, Na2HPO4·12H2O 26 mg 및 증류수 2974 mg를 함유시켜 주사제를 제조하였다.
100 mg as an active ingredient, 180 mg of mannitol, 26 mg of Na 2 HPO 4 .12H 2 O and 2974 mg of distilled water were added to prepare an injection.
[실험예]
[Experimental Example]
실험예 1. 키나아제 저해 활성 측정Experimental Example 1. Measurement of kinase inhibitory activity
본 발명에 따른 대표화합물로서 N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)-4-(4-메틸피페라진-1-일)벤즈아미드 (화합물번호 22, 실시예 21)에 대하여 다양한 단백질 키나아제의 저해활성(%저해능)을 측정하여 하기 표 1에 나타내었다. As a representative compound according to the present invention, N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) -4- (4-methylpipepe The inhibitory activity (% inhibition) of various protein kinases was measured for razin-1-yl) benzamide (Compound No. 22, Example 21) and is shown in Table 1 below.
Claims (6)
[화학식 1]
상기 화학식 1에서,
X는 CH; 또는 N 을 나타내며,
P는 수소원자; C1-C10 알킬기; 1 내지 3개의 페닐이 치환된 C1∼C10 알킬기; 페닐기; 또는 산소원자가 포함된 5 내지 7각형의 헤테로싸이클로알킬기를 나타내며,
L1은 -SR1; -S(O)R1; 또는 -S(O)2R1을 나타내며,
G는 존재하지 않거나; 또는 -NHC(O)-; 또는 -C(O)NH- 를 나타내며,
L2는 -NHR2; -OR3; R4로 치환 또는 비치환된 C1∼C10 알킬기; R5로 치환 또는 비치환된 C3∼C10 싸이클로알킬기; R6으로 치환 또는 비치환될 수 있고 질소원자가 1 내지 2개 포함된 5 내지 7각형의 헤테로싸이클로알킬기; R7로 치환 또는 비치환된 C6∼C15 아릴기; 또는 R8로 치환 또는 비치환될 수 있고 황원자 및 질소원자 중에서 선택된 헤테로원자가 1 내지 3개 포함된 5 내지 7각형의 헤테로아릴기를 나타내며,
R1은 수소원자; 또는 1 내지 3개의 할로겐원자로 치환 또는 비치환된 C6∼C15 아릴기를 나타내며,
R2는 수소원자; C1∼C10 알킬기; C3∼C10 싸이클로알킬기; 또는 할로, C1∼C10 알킬, C1∼C10 알콕시, 및 1 내지 10개의 할로겐이 치환된 C1∼C10 할로알킬 중에서 선택된 치환기로 치환 또는 비치환된 페닐기를 나타내며,
R3은 수소원자; 또는 C1∼C10 알킬기를 나타내며,
R4 및 R5는 서로 같거나 다른 것으로서 아미노기; 모노(C1∼C10 알킬)아미노기; 또는 디(C1∼C10 알킬)아미노기를 나타내며,
R6은 수소원자; 또는 C1∼C6 알킬기를 나타내며,
R7은 할로겐원자; C1∼C10 알킬기; C1∼C10 알콕시기; 1 내지 10개의 할로겐이 치환된 할로알킬기; C1∼C10 알킬기로 치환 또는 비치환될 수 있고 산소원자 및 질소원자 중에서 선택된 헤테로원자가 1 내지 3개 포함된 5 내지 7각형의 헤테로싸이클로알킬기; 아미노기; 모노(C1∼C10 알킬)아미노기; 또는 디(C1∼C10 알킬)아미노기를 나타내며,
R8은 C1∼C10 알킬기; 또는 산소원자 및 질소원자 중에서 선택된 헤테로원자가 1 내지 3개 포함된 5 내지 7각형의 헤테로싸이클로알킬기를 나타낸다.
A compound selected from a compound represented by Formula 1, a pharmaceutically acceptable salt thereof, a hydrate thereof, or a solvate thereof:
[Chemical Formula 1]
In Formula 1,
X is CH; Or N,
P is a hydrogen atom; A C 1 -C 10 alkyl group; C 1 -C 10 alkyl group substituted with 1 to 3 phenyl; A phenyl group; Or a 5 to 7 heterocyclic alkyl group containing an oxygen atom,
L 1 is -SR 1 ; -S (O) R 1 ; Or -S (O) 2 R 1 ,
G is absent; Or -NHC (O)-; Or -C (O) NH-,
L 2 is -NHR 2 ; -OR 3 ; C 1 -C 10 alkyl group unsubstituted or substituted with R 4 ; C 3 -C 10 cycloalkyl group unsubstituted or substituted with R 5 ; A 5 to 7 hexagonal heterocycloalkyl group which may be unsubstituted or substituted with R 6 , and contains 1 to 2 nitrogen atoms; C 6 -C 15 aryl group unsubstituted or substituted with R 7 ; Or it may be substituted or unsubstituted with R 8 and represents a 5 to 7 heteroaryl group containing 1 to 3 heteroatoms selected from a sulfur atom and a nitrogen atom,
R 1 is a hydrogen atom; Or a C 6 to C 15 aryl group unsubstituted or substituted with 1 to 3 halogen atoms,
R 2 is a hydrogen atom; C 1 -C 10 alkyl group; C 3 -C 10 cycloalkyl group; Or a phenyl group unsubstituted or substituted with a substituent selected from halo, C 1 -C 10 alkyl, C 1 -C 10 alkoxy, and C 1 -C 10 haloalkyl substituted with 1 to 10 halogen,
R 3 is a hydrogen atom; Or a C 1 to C 10 alkyl group,
R 4 and R 5 are the same as or different from each other and an amino group; Mono (C 1 -C 10 alkyl) amino group; Or a di (C 1 -C 10 alkyl) amino group,
R 6 is a hydrogen atom; Or a C 1 to C 6 alkyl group,
R 7 is Halogen atom; C 1 -C 10 alkyl group; C 1 -C 10 alkoxy group; Haloalkyl group substituted with 1 to 10 halogens; A 5 to 7 hexagonal heterocycloalkyl group which may be unsubstituted or substituted with a C 1 to C 10 alkyl group and contains 1 to 3 heteroatoms selected from an oxygen atom and a nitrogen atom; An amino group; Mono (C 1 -C 10 alkyl) amino group; Or a di (C 1 -C 10 alkyl) amino group,
R 8 is a C 1 -C 10 alkyl group; Or a 5 to 7 hexagonal heterocycloalkyl group including 1 to 3 heteroatoms selected from an oxygen atom and a nitrogen atom.
상기 X는 CH; 또는 N 을 나타내며,
상기 P는 수소원자; C1∼C6 알킬기; 벤질기; 트리틸기; 페닐기; 또는 테트라하이드로피란일기를 나타내며,
L1은 -S(O)R1; 또는 -S(O)2R1을 나타내며, 이때 R1은 수소원자; 페닐기; 또는 1 내지 3개의 할로겐원자로 치환된 페닐기를 나타내며,
G는 존재하지 않거나; 또는 -NHC(O)-; 또는 -C(O)NH- 를 나타내며,
L2는 아미노기; 싸이클로헥실아미노기; 페닐아미노기; C1∼C6 알콕시페닐아미노기; 할로 및 할로알킬 중에서 선택된 치환기로 치환된 페닐아미노기; 하이드록시기; C1∼C6 알콕시기; C1∼C6 알킬기; 디메틸아미노 C1∼C6 알킬기; 싸이클로헥실기; 디메틸아미노싸이클로헥실기; 몰포리노기; 피페리디닐기; 피페라지닐기; 4-(C1∼C6 알킬)피페라지닐기; 페닐기; 클로로페닐기; 플루오로페닐기; 4-클로로-3-트리플루오로메틸페닐기; C1∼C6 알킬페닐기; C1∼C6 알콕시페닐기; 트리플루오로메틸페닐기; 몰포리노페닐기; (4-메틸피페라지닐)페닐기; (4-에틸피페라지닐)페닐기; 아미노페닐기; 디메틸아미노페닐기; 티오페닐기; 피리디닐기; (C1∼C6 알킬)피리디닐기; 또는 몰포리노피리디닐기를 나타내는 것을 특징으로 하는 화합물.
The method according to claim 1,
X is CH; Or N,
P is a hydrogen atom; C 1 -C 6 alkyl group; Benzyl group; Trityl group; A phenyl group; Or a tetrahydropyranyl group,
L 1 is —S (O) R 1 ; Or -S (O) 2 R 1 , wherein R 1 is a hydrogen atom; A phenyl group; Or a phenyl group substituted with 1 to 3 halogen atoms,
G is absent; Or -NHC (O)-; Or -C (O) NH-,
L 2 is an amino group; Cyclohexylamino group; Phenylamino group; C 1 -C 6 alkoxyphenylamino group; Phenylamino group substituted with a substituent selected from halo and haloalkyl; A hydroxyl group; C 1 -C 6 alkoxy group; C 1 -C 6 alkyl group; Dimethylamino C 1 to C 6 alkyl group; Cyclohexyl group; Dimethylaminocyclohexyl group; A morpholino group; Piperidinyl group; Piperazinyl groups; 4- (C 1 -C 6 alkyl) piperazinyl group; A phenyl group; Chlorophenyl group; Fluorophenyl group; 4-chloro-3-trifluoromethylphenyl group; C 1 -C 6 alkylphenyl group; C 1 -C 6 alkoxyphenyl group; Trifluoromethylphenyl group; Morpholinophenyl group; (4-methylpiperazinyl) phenyl group; (4-ethylpiperazinyl) phenyl group; Aminophenyl group; Dimethylaminophenyl group; Thiophenyl group; Pyridinyl group; (C 1 -C 6 alkyl) pyridinyl group; Or a morpholino pyridinyl group.
5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-아민 (화합물번호 1);
N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-4-(4-메틸피페라진-1-일)벤즈아미드 (화합물번호 2);
N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-3-(트리플루오로메틸)벤즈아미드 (화합물번호 3);
N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-4-메톡시벤즈아미드 (화합물번호 4);
N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-벤즈아미드 (화합물번호 5);
4-플루오로-N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-벤즈아미드 (화합물번호 6);
4-클로로-N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-벤즈아미드 (화합물번호 7);
4-(tert-부틸)-N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-벤즈아미드 (화합물번호 8);
N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-4-몰포리노벤즈아미드 (화합물번호 9);
4-(디메틸아미노)-N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-벤즈아미드 (화합물번호 10);
4-(디메틸아미노)-N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-싸이클로헥산카복스아미드 (화합물번호 11);
4-(디메틸아미노)-N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-피발로아미드 (화합물번호 12);
4-(디메틸아미노)-N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-헥산아미드 (화합물번호 13);
에틸-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-카바메이트 (화합물번호 14);
tert-부틸-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-카바메이트 (화합물번호 15);
N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-티오펜-2-카복스아미드 (화합물번호 16);
N-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-피페리딘-1-카복스아미드 (화합물번호 17);
1-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-3-페닐우레아 (화합물번호 18);
1-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)-3-(2-메톡시페닐)우레아 (화합물번호 19);
1-(4-클로로-3-(트리플루오로메틸)페닐)-3-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)우레아 (화합물번호 20);
1-싸이클로헥실-3-(5-((3-플루오로페닐)설포닐)-1-트리틸-1H-피라졸로[3,4-b]피리딘-3-일)우레아(화합물번호 21);
N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)-4-(4-메틸피페라진-1-일)벤즈아미드(화합물번호 22);
N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)-4-메톡시벤즈아미드 (화합물번호 23);
N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)-3-(트리플루오로메틸)벤즈아미드 (화합물번호 24);
N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)벤즈아미드 (화합물번호 25);
4-플루오로-N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)벤즈아미드 (화합물번호 26);
4-클로로-N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)벤즈아미드 (화합물번호 27);
4-(tert-부틸)-N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)벤즈아미드 (화합물번호 28);
N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)-4-몰포리노벤즈아미드 (화합물번호 29);
4-(디메틸아미노)-N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)벤즈아미드 (화합물번호 30);
N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)싸이클로헥산카복스아미드 (화합물번호 31);
N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)피발아미드 (화합물번호 32);
N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)펜탄아미드 (화합물번호 33);
에틸-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)카바메이트 (화합물번호 34);
tert-부틸-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)카바메이트 (화합물번호 35);
N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)티오펜-2-카복스아미드 (화합물번호 36);
N-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)피페리딘-1-카복스아미드 (화합물번호 37);
1-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)-3-페닐우레아 (화합물번호 38);
1-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)-3-(2-메톡시페닐)우레아 (화합물번호 39);
1-(4-클로로-3-(트리플루오로메틸)페닐)-3-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)우레아 (화합물번호 40);
1-싸이클로헥실-3-(5-((3-플루오로페닐)설포닐)-1H-피라졸로[3,4-b]피리딘-3-일)우레아 (화합물번호 41);
5-((3-플루오로페닐)설포닐)-N-(4-몰포리노페닐)-1-(테트로하이드로-2H-피란-2-일)-1H-인다졸-3-카복스아미드 (화합물번호 42);
5-((3-플루오로페닐)설포닐)-N-(6-몰포리노피리딘-3-일)-1-(테트로하이드로-2H-피란-2-일)-1H-인다졸-3-카복스아미드 (화합물번호 43);
N-(4-(4-에틸피페라진-1-일)페닐)-5-((3-플루오로페닐)설포닐)-1-(테트로하이드로-2H-피란-2-일)-1H-인다졸-3-카복스아미드 (화합물번호 44);
5-((3-플루오로페닐)설포닐)-N-(6-메틸피리딘-3-일)-1-(테트로하이드로-2H-피란-2-일)-1H-인다졸-3-카복스아미드 (화합물번호 45);
N-(4-클로로-3-(트리프루오로메틸)페닐))-5-((3-플루오로페닐)설포닐)-1-(테트로하이드로-2H-피란-2-일)-1H-인다졸-3-카복스아미드 (화합물번호 46);
5-((3-플루오로페닐)설포닐)-N-(4-몰포리노페닐)-1H-인다졸-3-카복스아미드 (화합물번호 47);
5-((3-플루오로페닐)설포닐)-N-(4-몰포리노피리딘-3-일)-1H-인다졸-3-카복스아미드 (화합물번호 48);
N-(4-(4-에틸피페라진-1-일)페닐)-5-((3-플루오로페닐)설포닐)-1H-인다졸-3-카복스아미드 (화합물번호 49);
5-((3-플루오로페닐)설포닐)-N-(6-메틸피리딘-3-일)-1H-인다졸-3-카복스아미드 (화합물번호 50); 및
N-(4-클로로-3-(트리플루오로메틸)페닐)-5-((3-플루오로페닐)설포닐)-1H-인다졸-3-카복스아미드 (화합물번호 51);
중에서 선택된 것을 특징으로 하는 화합물.
The method according to claim 1,
5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-amine (Compound No. 1);
N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -4- (4-methylpiperazin- 1-yl) benzamide (Compound No. 2);
N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -3- (trifluoromethyl) benz Amides (Compound No. 3);
N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -4-methoxybenzamide (Compound No. 4);
N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -benzamide (Compound No. 5);
4-Fluoro- N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -benzamide (compound Number 6);
4-Chloro- N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -benzamide (Compound No. 7);
4- (tert-butyl) - N - (5 - ( (3- fluorophenyl) sulfonyl) -1-trityl -1 H-pyrazolo [3,4- b] pyridin-3-yl) benzamide Amides (Compound No. 8);
N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -4-morpholinobenzamide (Compound No. 9);
4- (dimethylamino) - N - (5 - ((3- fluorophenyl) sulfonyl) -1-trityl -1 H-pyrazolo [3,4- b] pyridin-3-yl) -benzamide (Compound number 10);
4- (dimethylamino) - N - (5 - ((3- fluorophenyl) sulfonyl) -1-trityl -1 H-pyrazolo [3,4- b] pyridin-3-yl) cyclo-hexane Carboxamide (Compound No. 11);
4- (dimethylamino) - to Sangapi - N - (5 - ((3- fluorophenyl) sulfonyl) -1-trityl -1 H-pyrazolo [3,4- b] pyridin-3-yl) Amides (Compound No. 12);
4- (dimethylamino) - N - (5 - ((3- fluorophenyl) sulfonyl) -1-trityl -1 H-pyrazolo [3,4- b] pyridin-3-yl) -pentanoic amide (Compound number 13);
Ethyl- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -carbamate (Compound No. 14);
tert -butyl- (5-((3-fluorophenyl) sulfonyl) -1-trityl- 1H -pyrazolo [3,4- b ] pyridin-3-yl) -carbamate (Compound No. 15) ;
N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -thiophene-2-carboxamide ( Compound no. 16);
N- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -piperidine-1-carboxamide (Compound number 17);
1- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -3-phenylurea (Compound No. 18) ;
1- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) -3- (2-methoxyphenyl) Urea (Compound No. 19);
1- (4-chloro-3- (trifluoromethyl) phenyl) -3- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) urea (Compound No. 20);
1-cyclohexyl-3- (5-((3-fluorophenyl) sulfonyl) -1-trityl-1 H -pyrazolo [3,4- b ] pyridin-3-yl) urea (Compound No. 21 );
N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) -4- (4-methylpiperazin-1-yl) benz Amide (Compound No. 22);
N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) -4-methoxybenzamide (Compound No. 23);
N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) -3- (trifluoromethyl) benzamide (Compound No. 24 );
N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) benzamide (Compound No. 25);
4-fluoro- N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) benzamide (Compound No. 26);
4-chloro- N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) benzamide (Compound No. 27);
4- (tert-butyl) - N - (5 - ((3- phenyl) sulfonyl) -1 H-fluoro-pyrazolo [3,4- b] pyridin-3-yl) benzamide (Compound No. 28) ;
N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) -4-morpholinobenzamide (Compound No. 29);
4- (dimethylamino) - N - (5 - ( ( 3-fluorophenyl) sulfonyl) -1 H - pyrazolo [3,4- b] pyridin-3-yl) benzamide (Compound No. 30);
N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) cyclohexanecarboxamide (Compound No. 31);
N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) pivalamide (Compound No. 32);
N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) pentanamide (Compound No. 33);
Ethyl- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) carbamate (Compound No. 34);
tert -butyl- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) carbamate (Compound No. 35);
N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) thiophene-2-carboxamide (Compound No. 36);
N- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) piperidine-1-carboxamide (Compound No. 37);
1- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) -3-phenylurea (Compound No. 38);
1- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) -3- (2-methoxyphenyl) urea (Compound No. 39 );
1- (4-chloro-3- (trifluoromethyl) phenyl) -3- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridine-3 -Yl) urea (Compound No. 40);
1-cyclohexyl-3- (5-((3-fluorophenyl) sulfonyl) -1 H -pyrazolo [3,4- b ] pyridin-3-yl) urea (Compound No. 41);
5 - ((3-fluorophenyl) sulfonyl) - N - (4- Dimorpholino Reno phenyl) -1- (tetrahydro -2 H in Tet-pyran-2-yl) -1 H-indazol-3-car Voxamide (Compound No. 42);
5 - ((3-fluorophenyl) sulfonyl) - N - (6- Reno Dimorpholino-3-yl) -1- (tetrahydro -2 H in Tet-pyran-2-yl) -1 H-indazole -3-carboxamide (Compound No. 43);
N- (4- (4-ethylpiperazin-1-yl) phenyl) -5-((3-fluorophenyl) sulfonyl) -1- (tetrohydro- 2H -pyran-2-yl)- 1 H -indazol-3-carboxamide (Compound No. 44);
5 - ((3-fluorophenyl) sulfonyl) - N - (6- methylpyridin-3-yl) -1- (tetrahydro -2 H in Tet-pyran-2-yl) -1 H-indazol- 3-carboxamide (Compound No. 45);
N- (4-chloro-3- (trifluoromethyl) phenyl))-5-((3-fluorophenyl) sulfonyl) -1- (tetrohydro- 2H -pyran-2-yl) -1 H -indazole-3-carboxamide (Compound No. 46);
5 - ((3-fluorophenyl) sulfonyl) - N - (4- Dimorpholino Reno phenyl) -1 H-indazole-3-carboxamide (Compound No. 47);
5 - ((3-fluorophenyl) sulfonyl) - N - (4- Reno Dimorpholino-3-yl) -1 H-indazole-3-carboxamide (Compound No. 48-);
N- (4- (4-ethylpiperazin-1-yl) phenyl) -5-((3-fluorophenyl) sulfonyl) -1 H -indazol-3-carboxamide (Compound No. 49);
5 - ((3-fluorophenyl) sulfonyl) - N - (6- methylpyridin-3-yl) -1 H-indazole-3-carboxamide (Compound No. 50); And
N- (4-chloro-3- (trifluoromethyl) phenyl) -5-((3-fluorophenyl) sulfonyl) -1 H -indazole-3-carboxamide (Compound No. 51);
Compounds characterized in that selected from.
A pharmaceutical composition for preventing and treating cancer diseases containing the compound of any one of claims 1 to 3 as an active ingredient.
단백질 키나아제에 대한 저해활성을 가지는 암질환의 예방 및 치료용 약학적 조성물.
The method of claim 4,
A pharmaceutical composition for preventing and treating cancer diseases having inhibitory activity against protein kinases.
상기 암질환은 위암, 폐암, 간암, 대장암, 소장암, 췌장암, 뇌암, 뼈암, 흑색종, 유방암, 경화성선증, 자궁암, 자궁경부암, 두경부암, 식도암, 갑상선암, 부갑상선암, 신장암, 육종, 전립선암, 요도암, 방광암, 혈액암, 림프종, 건선, 또는 섬유선종 중에서 선택된 것을 특징으로 하는 암질환의 예방 및 치료용 약학적 조성물.The method of claim 4,
The cancer diseases include stomach cancer, lung cancer, liver cancer, colon cancer, small intestine cancer, pancreatic cancer, brain cancer, bone cancer, melanoma, breast cancer, sclerosis, uterine cancer, cervical cancer, head and neck cancer, esophageal cancer, thyroid cancer, parathyroid cancer, kidney cancer, sarcoma, Pharmaceutical composition for the prevention and treatment of cancer diseases, characterized in that selected from prostate cancer, urethral cancer, bladder cancer, hematologic cancer, lymphoma, psoriasis, or fibroadenoma.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020120077013A KR101936851B1 (en) | 2012-07-16 | 2012-07-16 | Pyrazolopyridine or indazole derivatives as protein kinase inhibitors |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020120077013A KR101936851B1 (en) | 2012-07-16 | 2012-07-16 | Pyrazolopyridine or indazole derivatives as protein kinase inhibitors |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20140019055A true KR20140019055A (en) | 2014-02-14 |
| KR101936851B1 KR101936851B1 (en) | 2019-01-11 |
Family
ID=50266750
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020120077013A Expired - Fee Related KR101936851B1 (en) | 2012-07-16 | 2012-07-16 | Pyrazolopyridine or indazole derivatives as protein kinase inhibitors |
Country Status (1)
| Country | Link |
|---|---|
| KR (1) | KR101936851B1 (en) |
Cited By (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2018152220A1 (en) * | 2017-02-15 | 2018-08-23 | Incyte Corporation | Pyrazolopyridine compounds and uses thereof |
| CN109071516A (en) * | 2015-12-15 | 2018-12-21 | 阿斯利康(瑞典)有限公司 | novel compounds |
| US10266530B2 (en) | 2016-09-09 | 2019-04-23 | Incyte Corporation | Pyrazolopyridine compounds and uses thereof |
| KR20190043013A (en) * | 2017-10-17 | 2019-04-25 | 한국화학연구원 | Novel indazole derivatives, and use thereof |
| US10280164B2 (en) | 2016-09-09 | 2019-05-07 | Incyte Corporation | Pyrazolopyridone compounds and uses thereof |
| KR20200046952A (en) | 2018-10-26 | 2020-05-07 | 한국과학기술연구원 | heteroaromatic macrocyclic derivatives as protein kinase inhibitors |
| US10722495B2 (en) | 2017-09-08 | 2020-07-28 | Incyte Corporation | Cyanoindazole compounds and uses thereof |
| US10745388B2 (en) | 2018-02-20 | 2020-08-18 | Incyte Corporation | Indazole compounds and uses thereof |
| US10752635B2 (en) | 2018-02-20 | 2020-08-25 | Incyte Corporation | Indazole compounds and uses thereof |
| US10800761B2 (en) | 2018-02-20 | 2020-10-13 | Incyte Corporation | Carboxamide compounds and uses thereof |
| US10899755B2 (en) | 2018-08-08 | 2021-01-26 | Incyte Corporation | Benzothiazole compounds and uses thereof |
| US11014929B2 (en) | 2016-09-09 | 2021-05-25 | Incyte Corporation | Pyrazolopyrimidine compounds and uses thereof |
| US11066394B2 (en) | 2019-08-06 | 2021-07-20 | Incyte Corporation | Solid forms of an HPK1 inhibitor |
| US11111247B2 (en) | 2018-09-25 | 2021-09-07 | Incyte Corporation | Pyrazolopyrimidine compounds and uses thereof |
| US11242343B2 (en) | 2016-09-09 | 2022-02-08 | Incyte Corporation | Pyrazolopyridine compounds and uses thereof |
| US11299473B2 (en) | 2018-04-13 | 2022-04-12 | Incyte Corporation | Benzimidazole and indole compounds and uses thereof |
| CN114685430A (en) * | 2022-03-18 | 2022-07-01 | 浙江大学 | Fenbendazole analogue, preparation method and application |
| US20230167057A1 (en) * | 2021-08-10 | 2023-06-01 | Ifm Due, Inc. | Compounds and compositions for treating conditions associated with sting activity |
| CN118126038A (en) * | 2024-05-08 | 2024-06-04 | 烟台大学 | Pyrazolopyridine derivatives and preparation methods and applications thereof |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA3161730A1 (en) | 2019-12-16 | 2021-06-24 | Korea Research Institute Of Chemical Technology | Novel indazole derivative, and use thereof |
| KR102334365B1 (en) | 2019-12-27 | 2021-12-06 | 한국화학연구원 | Novel dihydroindeno[1,2-c]pyrazole-3-carboxamide derivatives and pharmaceutical composition for preventing or treating neurodevelopmental disorders or neurodegenerative diseases comprising the same |
| KR102607237B1 (en) | 2023-06-07 | 2023-11-30 | 주식회사 젠센 | New triazole substituted indazole derivatives and use thereof |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20030192893A1 (en) * | 2002-04-15 | 2003-10-16 | Silvio Weder | Product package having multi-layer labeling cover assembly |
| EA017852B1 (en) * | 2006-12-20 | 2013-03-29 | НЕРВИАНО МЕДИКАЛ САЙЕНСИЗ С.р.л. | Substituted indazole derivatives active as kinase inhibitors |
-
2012
- 2012-07-16 KR KR1020120077013A patent/KR101936851B1/en not_active Expired - Fee Related
Cited By (35)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN109071516B (en) * | 2015-12-15 | 2021-08-31 | 阿斯利康(瑞典)有限公司 | novel compound |
| CN109071516A (en) * | 2015-12-15 | 2018-12-21 | 阿斯利康(瑞典)有限公司 | novel compounds |
| US12384778B2 (en) | 2016-09-09 | 2025-08-12 | Incyte Corporation | Pyrazolopyridine compounds and uses thereof |
| US11891388B2 (en) | 2016-09-09 | 2024-02-06 | Incyte Corporation | Pyrazolopyridine compounds and uses thereof |
| US10280164B2 (en) | 2016-09-09 | 2019-05-07 | Incyte Corporation | Pyrazolopyridone compounds and uses thereof |
| US10435405B2 (en) | 2016-09-09 | 2019-10-08 | Incyte Corporation | Pyrazolopyridine compounds and uses thereof |
| US12516059B2 (en) | 2016-09-09 | 2026-01-06 | Incyte Corporation | Pyrazolopyridine compounds and uses thereof |
| US10266530B2 (en) | 2016-09-09 | 2019-04-23 | Incyte Corporation | Pyrazolopyridine compounds and uses thereof |
| US11242343B2 (en) | 2016-09-09 | 2022-02-08 | Incyte Corporation | Pyrazolopyridine compounds and uses thereof |
| US11542265B2 (en) | 2016-09-09 | 2023-01-03 | Incyte Corporation | Pyrazolopyrimidine compounds and uses thereof |
| US11795166B2 (en) | 2016-09-09 | 2023-10-24 | Incyte Corporation | Pyrazolopyridine compounds and uses thereof |
| US11014929B2 (en) | 2016-09-09 | 2021-05-25 | Incyte Corporation | Pyrazolopyrimidine compounds and uses thereof |
| US10934288B2 (en) | 2016-09-09 | 2021-03-02 | Incyte Corporation | Pyrazolopyridine compounds and uses thereof |
| WO2018152220A1 (en) * | 2017-02-15 | 2018-08-23 | Incyte Corporation | Pyrazolopyridine compounds and uses thereof |
| US11406624B2 (en) | 2017-02-15 | 2022-08-09 | Incyte Corporation | Pyrazolopyridine compounds and uses thereof |
| US10722495B2 (en) | 2017-09-08 | 2020-07-28 | Incyte Corporation | Cyanoindazole compounds and uses thereof |
| KR20190043013A (en) * | 2017-10-17 | 2019-04-25 | 한국화학연구원 | Novel indazole derivatives, and use thereof |
| US11731958B2 (en) | 2018-02-20 | 2023-08-22 | Incyte Corporation | Carboxamide compounds and uses thereof |
| US10745388B2 (en) | 2018-02-20 | 2020-08-18 | Incyte Corporation | Indazole compounds and uses thereof |
| US11492354B2 (en) | 2018-02-20 | 2022-11-08 | Incyte Corporation | Indazole compounds and uses thereof |
| US12466815B2 (en) | 2018-02-20 | 2025-11-11 | Incyte Corporation | Carboxamide compounds and uses thereof |
| US10800761B2 (en) | 2018-02-20 | 2020-10-13 | Incyte Corporation | Carboxamide compounds and uses thereof |
| US10752635B2 (en) | 2018-02-20 | 2020-08-25 | Incyte Corporation | Indazole compounds and uses thereof |
| US11299473B2 (en) | 2018-04-13 | 2022-04-12 | Incyte Corporation | Benzimidazole and indole compounds and uses thereof |
| US10899755B2 (en) | 2018-08-08 | 2021-01-26 | Incyte Corporation | Benzothiazole compounds and uses thereof |
| US11866426B2 (en) | 2018-08-08 | 2024-01-09 | Incyte Corporation | Benzothiazole compounds and uses thereof |
| US11111247B2 (en) | 2018-09-25 | 2021-09-07 | Incyte Corporation | Pyrazolopyrimidine compounds and uses thereof |
| US12441731B2 (en) | 2018-09-25 | 2025-10-14 | Incyte Corporation | Pyrazolopyrimidine compounds and uses thereof |
| KR20200046952A (en) | 2018-10-26 | 2020-05-07 | 한국과학기술연구원 | heteroaromatic macrocyclic derivatives as protein kinase inhibitors |
| US11066394B2 (en) | 2019-08-06 | 2021-07-20 | Incyte Corporation | Solid forms of an HPK1 inhibitor |
| US11787784B2 (en) | 2019-08-06 | 2023-10-17 | Incyte Corporation | Solid forms of an HPK1 inhibitor |
| US20230167057A1 (en) * | 2021-08-10 | 2023-06-01 | Ifm Due, Inc. | Compounds and compositions for treating conditions associated with sting activity |
| CN114685430A (en) * | 2022-03-18 | 2022-07-01 | 浙江大学 | Fenbendazole analogue, preparation method and application |
| CN118126038B (en) * | 2024-05-08 | 2024-07-12 | 烟台大学 | Pyrazolopyridine derivative, and preparation method and application thereof |
| CN118126038A (en) * | 2024-05-08 | 2024-06-04 | 烟台大学 | Pyrazolopyridine derivatives and preparation methods and applications thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| KR101936851B1 (en) | 2019-01-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR101936851B1 (en) | Pyrazolopyridine or indazole derivatives as protein kinase inhibitors | |
| JP5557849B2 (en) | Carbazole and carboline kinase inhibitors | |
| CN113454081B (en) | Imidazopyridinyl compounds and their use for the treatment of proliferative diseases | |
| JP6035423B2 (en) | Novel condensed pyrimidine compound or salt thereof | |
| KR101490761B1 (en) | THIENO[3,2-d]PYRIMIDINE DERIVATIVES HAVING INHIBITORY ACTIVITY FOR PROTEIN KINASES | |
| KR101116756B1 (en) | Novel 1,6-disubstituted indole compounds as protein kinase inhibitors | |
| KR101850282B1 (en) | Heteroarylamine derivatives as protein kinase inhibitors | |
| CN112839937B (en) | Isoindolin-1-one derivatives, preparation method and application thereof | |
| KR101965326B1 (en) | Novel 2,3,5-substituted thiophene compounds as protein kinase inhibitors | |
| US9956220B2 (en) | Imidazo-pyridazine derivatives as casein kinase 1 δ/ϵ inhibitors | |
| AU2006232620A1 (en) | Substituted heterocycles and their use as CHK1, PDK1 and PAK inhibitors | |
| JP2010522195A (en) | Pyrazolopyrimidine analogs and their use as mTOR kinase inhibitors and PI3K inhibitors | |
| AU2014370186A1 (en) | Pyrazolo[1,5-a]pyridine derivatives and methods of their use | |
| KR20210015730A (en) | Oxo-pyridine fused ring derivatives and pharmaceutical composition containing the same | |
| WO2013100672A1 (en) | 3,6-disubstituted indazole derivative having protein kinase inhibiting activity | |
| KR20200134179A (en) | Heteroaryl comprising N derivatives and pharmaceutical composition for use in preventing or treating cancer containing the same as an active ingredient | |
| CN104837844A (en) | Pyrazole-substituted imidazopiperazines as casein kinase 1 D/E inhibitors | |
| KR20100057433A (en) | Novel pyrazolo[4,3-b]pyridine derivatives or pharmaceutically acceptable salt thereof, preparation method thereof and pharmaceutical composition containing the same as an active ingredient | |
| JP2012211085A (en) | Hedgehog signal inhibitor | |
| JP6093485B2 (en) | Preventive and / or therapeutic agent for immune disease | |
| CN111499613B (en) | N-carboxamide derivatives, method for the production thereof and their use in medicine | |
| AU2014284013B2 (en) | Five-membered heterocyclic pyridine compounds and preparation method and use thereof | |
| WO2023185073A1 (en) | Parp7 inhibitor and use thereof | |
| AU2006297889A1 (en) | New compounds II | |
| HK40083035B (en) | Parp7 inhibitor and use thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0109 | Patent application |
St.27 status event code: A-0-1-A10-A12-nap-PA0109 |
|
| PG1501 | Laying open of application |
St.27 status event code: A-1-1-Q10-Q12-nap-PG1501 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-3-3-R10-R13-asn-PN2301 St.27 status event code: A-3-3-R10-R11-asn-PN2301 |
|
| R17-X000 | Change to representative recorded |
St.27 status event code: A-3-3-R10-R17-oth-X000 |
|
| P22-X000 | Classification modified |
St.27 status event code: A-2-2-P10-P22-nap-X000 |
|
| A201 | Request for examination | ||
| PA0201 | Request for examination |
St.27 status event code: A-1-2-D10-D11-exm-PA0201 |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
St.27 status event code: A-1-2-D10-D21-exm-PE0902 |
|
| T11-X000 | Administrative time limit extension requested |
St.27 status event code: U-3-3-T10-T11-oth-X000 |
|
| E13-X000 | Pre-grant limitation requested |
St.27 status event code: A-2-3-E10-E13-lim-X000 |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| E701 | Decision to grant or registration of patent right | ||
| PE0701 | Decision of registration |
St.27 status event code: A-1-2-D10-D22-exm-PE0701 |
|
| PR0701 | Registration of establishment |
St.27 status event code: A-2-4-F10-F11-exm-PR0701 |
|
| PR1002 | Payment of registration fee |
St.27 status event code: A-2-2-U10-U11-oth-PR1002 Fee payment year number: 1 |
|
| PG1601 | Publication of registration |
St.27 status event code: A-4-4-Q10-Q13-nap-PG1601 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 4 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 5 |
|
| PC1903 | Unpaid annual fee |
St.27 status event code: A-4-4-U10-U13-oth-PC1903 Not in force date: 20240104 Payment event data comment text: Termination Category : DEFAULT_OF_REGISTRATION_FEE |
|
| PN2301 | Change of applicant |
St.27 status event code: A-5-5-R10-R13-asn-PN2301 St.27 status event code: A-5-5-R10-R11-asn-PN2301 |
|
| PC1903 | Unpaid annual fee |
St.27 status event code: N-4-6-H10-H13-oth-PC1903 Ip right cessation event data comment text: Termination Category : DEFAULT_OF_REGISTRATION_FEE Not in force date: 20240104 |