KR20110062351A - Method for preparing heterobiarylpyridine derivative compound and heterobiarylpyridine derivative compound prepared by such method - Google Patents
Method for preparing heterobiarylpyridine derivative compound and heterobiarylpyridine derivative compound prepared by such method Download PDFInfo
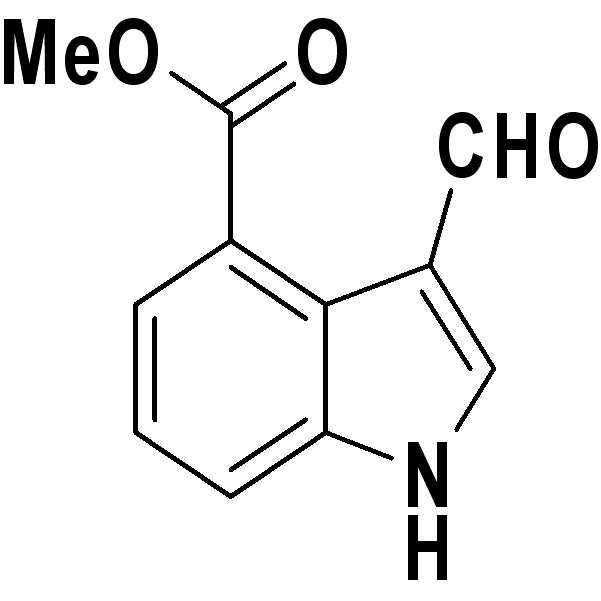
- Publication number
- KR20110062351A KR20110062351A KR1020090119049A KR20090119049A KR20110062351A KR 20110062351 A KR20110062351 A KR 20110062351A KR 1020090119049 A KR1020090119049 A KR 1020090119049A KR 20090119049 A KR20090119049 A KR 20090119049A KR 20110062351 A KR20110062351 A KR 20110062351A
- Authority
- KR
- South Korea
- Prior art keywords
- alkyl
- halogen
- substituted
- unsubstituted
- alkoxy
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/02—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings
- C07D405/04—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings
- C07D409/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings directly linked by a ring-member-to-ring-member bond
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
본 발명은 신규의 헤테로바이아릴피리딘 유도체 화합물의 제조방법 및 이러한 방법에 의해 제조된 헤테로바이아릴피리딘 유도체 화합물에 관한 것이다.The present invention relates to a method for preparing a novel heterobiarylpyridine derivative compound and a heterobiarylpyridine derivative compound prepared by such a method.
바이아릴(biaryls) 및 헤테로바이아릴(hetero-biaryls) 화합물들은 이들의 다양한 생물학적 활성으로 인해 많은 관심이 집중되고 있다. 특히 헤테로바이아릴은 생물활성을 지닌 다수의 작은 분자들에서 빈번하게 확인되고 있으며, 헤테로환(heterocycles)과 융합된 헤테로바이아릴 화합물은 중요한 약물특이분자단(pharmacophores)으로 사용되고 있다.Biaryls and hetero-biaryls compounds are of great interest due to their various biological activities. In particular, heterobiaryl is frequently identified in many small molecules with bioactivity, and heterobiaryl compounds fused with heterocycles are used as important pharmacophores.
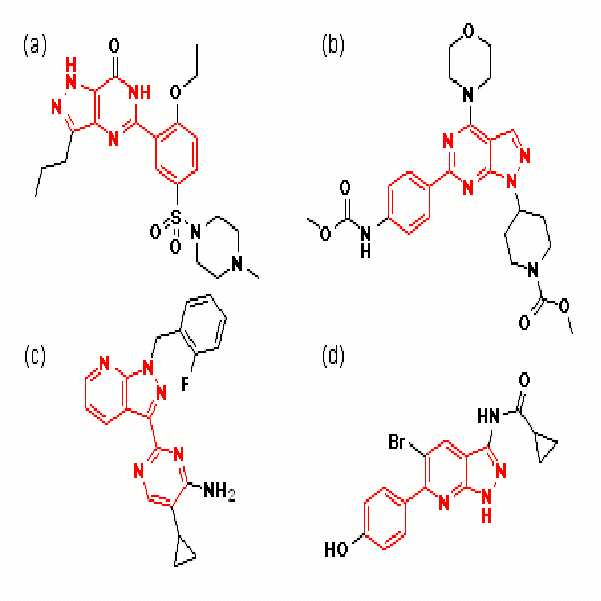
지금까지 공지된 주된 헤테로바이아릴 화합물로는, 예를 들면, 비아그라(ViagraR, sildenafil citrate, 하기 화합물(a)), 강력한 항암제인 WYE-354(하기 화합물 (b)), 가용성 구아닐레이트 사이클라제(sGC) 활성화제인 BAY 41-2272 (하기 화합물 (c)), 글리코겐 합성효소 키나아제-3 (GSK-3)의 강력한 억제제인 6-아릴 피 라졸로[3,4-b]피리딘 (하기 화합물 (d)) 등이 있다.The major heterobiaryl compounds known so far are, for example, Viagra (Viagra R , sildenafil citrate, compound (a)), WYE-354 (compound (b)), a potent anticancer agent, and soluble guanylate. BAY 41-2272, a sGC activator (compound (c)), 6-aryl pyrazolo [3,4-b] pyridine, a potent inhibitor of glycogen synthase kinase-3 (GSK-3) Compound (d)) and the like.
일반적으로, 바이아릴 또는 헤테로바이아릴 화합물은 전이 금속 촉매의 존재하에 교차-결합 반응(cross-coupling reactions)으로 합성된다. 교차-결합 반응이 다양한 이점을 제공하지만, 부피가 큰 기질(bulky substrates)에 대한 낮은 반응효율, 전이금속의 독성/안전성/고비용 문제, 적절한 촉매 등을 찾기 위한 버거운 선별작업 등과 같은 문제점이 존재하며, 나아가 다단계(multi-step) 합성과정을 요구한다.In general, biaryl or heterobiaryl compounds are synthesized in cross-coupling reactions in the presence of transition metal catalysts. While cross-linking reactions offer a variety of advantages, there are problems such as low reaction efficiency to bulky substrates, toxicity / safety / high cost of transition metals, and heavy screening to find suitable catalysts. Furthermore, it requires a multi-step synthesis process.
이에, 본 발명자들은 상기한 문제점을 해결하기 위하여 인돌의 C-3 위치에 알데하이드기를 도입하여 친전자성(dielectrophilic) 인돌 유도체를 만든 다음 이를 극성 양성자성 용매(polar protic solvent)에서 산 촉매(acid catalyst) 존재하에 이친핵체(dinucleophile)인 아미노피라졸 유도체와 반응시킨 결과, 인돌 고 리(indole ring)의 개방을 통해 고수율의 헤테로-바이아릴 피라졸로[3,4-b]피리딘 화합물이 얻어지고 이러한 화합물이 종양세포의 증식을 선택적으로 억제하는 것을 확인함으로써 본 발명을 완성하였다.In order to solve the above problems, the present inventors introduced an aldehyde group to the C-3 position of indole to make a dielectrophilic indole derivative, and then an acid catalyst in a polar protic solvent. Reaction with an aminopyrazole derivative which is a dinucleophile in the presence of a compound yields a high yield of hetero-biaryl pyrazolo [3,4-b] pyridine compound through the opening of an indole ring. The present invention was completed by confirming that these compounds selectively inhibit the proliferation of tumor cells.
따라서, 본 발명의 목적은 생물학적으로 유용한 활성을 지닌 헤테로바이아릴피리딘 유도체 화합물의 제조방법 및 이러한 방법에 의해 제조된 헤테로바이아릴피리딘 유도체 화합물을 제공하는 것이다.Accordingly, it is an object of the present invention to provide a method for preparing a heterobiarylpyridine derivative compound having biologically useful activity and a heterobiarylpyridine derivative compound prepared by such a method.
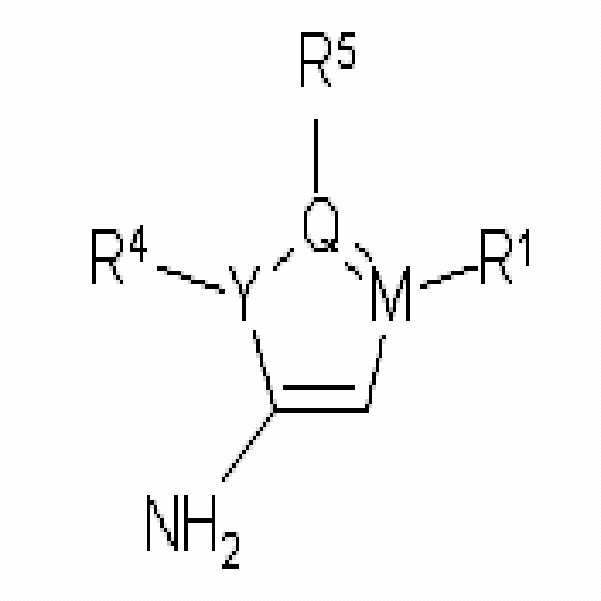
상기 목적에 따라, 본 발명은 극성 양성자성 용매(polar protic solvent)에서 화학식 2의 화합물을 산 촉매(acid catalyst)의 존재하에 화학식 3의 화합물과 반응시키는 단계를 포함하는, 하기 화학식 1의 화합물의 제조방법을 제공한다:According to the above object, the present invention comprises the step of reacting the compound of
<화학식 1><
<화학식 2><
<화학식 3><
상기 식에서,Where
X는 질소(N), 산소(O) 또는 황(S) 원자이고;X is a nitrogen (N), oxygen (O) or sulfur (S) atom;
Y, Q 및 M은 각각 독립적으로 탄소(C), 질소(N), 산소(O) 또는 황(S) 원자이며 (단 Y가 탄소 원자이면, Q 및 M 중 적어도 하나는 질소, 산소 또는 황 원자이고, Q가 탄소 원자이면, Y 및 M 중 적어도 하나는 질소, 산소 또는 황 원자이며, M이 탄소 원자이면, Y 및 Q 중 적어도 하나는 질소, 산소 또는 황 원자이다.);Y, Q and M are each independently carbon (C), nitrogen (N), oxygen (O) or sulfur (S) atoms (provided that if Y is a carbon atom, at least one of Q and M is nitrogen, oxygen or sulfur) And if Q is a carbon atom, then at least one of Y and M is a nitrogen, oxygen or sulfur atom, and if M is a carbon atom, then at least one of Y and Q is a nitrogen, oxygen or sulfur atom.);
R1은 존재하지 않거나(absent), 수소, 히드록시, 비치환되거나 C1-C6 알콕시 또는 할로겐으로 치환된 C1-C6 알킬, 비치환되거나 C1-C6 알콕시, 할로겐 또는 C1-C6 알킬로 치환된 아릴, 또는 비치환되거나 C1-C6 알콕시, 할로겐 또는 C1-C6 알킬로 치환된 헤테로아릴이며;R 1 is absent, hydrogen, hydroxy, aryl unsubstituted or substituted with C1-C6 alkoxy or halogen, C1-C6 alkyl, aryl unsubstituted or substituted with C1-C6 alkoxy, halogen or C1-C6 alkyl Or heteroaryl unsubstituted or substituted with C1-C6 alkoxy, halogen or C1-C6 alkyl;
R2는 수소, 할로겐, 비치환되거나 C1-C6 알킬로 치환된 아민, 니트로, C1-C6 알킬에스테르, 비치환되거나 C1-C6 알킬로 치환된 C1-C6 알콕시, 비치환되거나 C1-C6 알킬, C1-C6 할로알킬, C1-C6 알콕시 또는 할로겐으로 치환된 아릴, 또는 비치환되거나 C1-C6 알킬, C1-C6 할로알킬, C1-C6 알콕시 또는 할로겐으로 치환된 헤테로아릴이며;R 2 is hydrogen, halogen, amine unsubstituted or substituted with C1-C6 alkyl, nitro, C1-C6 alkylester, C1-C6 alkoxy unsubstituted or substituted with C1-C6 alkyl, unsubstituted or C1-C6 alkyl, Aryl substituted with C1-C6 haloalkyl, C1-C6 alkoxy or halogen, or heteroaryl unsubstituted or substituted with C1-C6 alkyl, C1-C6 haloalkyl, C1-C6 alkoxy or halogen;
R3는 X가 O 또는 S이면 존재하지 않고(absent), X가 N이면 수소, 비치환되거나 C1-C6 알킬 또는 할로겐으로 치환된 C1-C6 알킬, 비치환되거나 C1-C6 알킬 또는 할로겐으로 치환된 C1-C6 아랄킬(aralkyl), 비치환되거나 C1-C6 알킬 또는 할로겐으로 치환된 C1-C6 알콕시카보닐, 술포닐, COR6, CONR6R7 또는 CSNR6R7(여기서 R6 및 R7은 각각 독립적으로 수소, 비치환되거나 할로겐, C1-C6 알콕시 또는 C1-C6 알킬로 치환된 C1-C6 알킬, 비치환되거나 할로겐, C1-C6 알콕시 또는 C1-C6 알킬로 치환된 C3-C6 사이클로알킬, 비치환되거나 할로겐, C1-C6 알콕시 또는 C1-C6 알킬로 치환된 아릴, 또는 비치환되거나 C1-C6 알킬, C1-C6 할로알킬, C1-C6 알콕시 또는 할로겐으로 치환된 벤질임)이며;R 3 is absent if X is O or S, and if X is N, then C 1 -C 6 alkyl, unsubstituted or substituted with C 1 -C 6 alkyl or halogen is hydrogen, unsubstituted or substituted with C 1 -C 6 alkyl or halogen; C1-C6 alkoxy, unsubstituted or substituted with C1-C6 alkyl or halogen, C1-C6 alkoxycarbonyl, sulfonyl, COR 6 , CONR 6 R 7 or CSNR 6 R 7 , wherein R 6 and R 7 is each independently hydrogen, C 3 -C 6 alkyl unsubstituted or substituted with halogen, C 1 -C 6 alkoxy or C 1 -C 6 alkyl, C 3 -C 6 cyclo unsubstituted or substituted with halogen, C 1 -C 6 alkoxy or C 1 -C 6 alkyl Alkyl, aryl unsubstituted or substituted with halogen, C1-C6 alkoxy or C1-C6 alkyl, or benzyl unsubstituted or substituted with C1-C6 alkyl, C1-C6 haloalkyl, C1-C6 alkoxy or halogen;
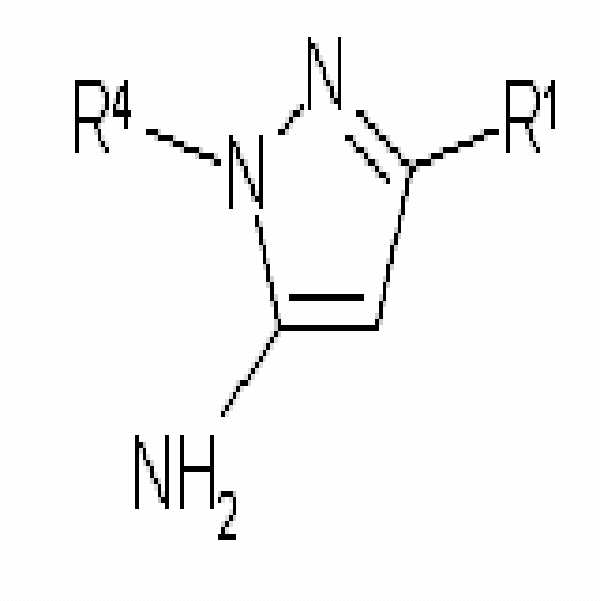
R4 및 R5는 각각 독립적으로 존재하지 않거나(absent), 수소, C1-C6 알킬 또는 비치환되거나 C1-C6 알킬, C1-C6 알콕시 또는 할로겐으로 치환된 아릴이다.R 4 and R 5 are each independently absent, hydrogen, C 1 -C 6 alkyl or aryl unsubstituted or substituted with C 1 -C 6 alkyl, C 1 -C 6 alkoxy or halogen.
본 발명은 또한 하기 화학식 1의 헤테로바이아릴피리딘 유도체 화합물을 제공한다:The present invention also provides heterobiarylpyridine derivative compounds of formula
<화학식 1><
상기 식에서,Where
X, Y, M, Q, R1, R2, R3, R4 및 R5는 앞서 정의된 바와 같다.X, Y, M, Q, R 1 , R 2 , R 3 , R 4 and R 5 are as defined above.
본 발명의 화학식 1의 헤테로바이아릴피리딘 유도체 화합물은 원-스텝(one-step) 개환(ring opening) 및 고리화반응(cyclization)을 통해 위치특이적으로(regioselectively) 고수율로 쉽게 제조될 수 있으며, 나아가 종양세포에 대해 우수한 증식억제활성을 가짐에 따라 항암제용 약학 조성물로 이용될 수 있다.Heterobiarylpyridine derivative compounds of the formula (1) of the present invention can be easily prepared in a high yield regioselectively through one-step ring opening and cyclization Furthermore, it can be used as an anticancer pharmaceutical composition as it has excellent proliferation inhibitory activity against tumor cells.
본 발명의 화학식 1의 헤테로바이아릴피리딘 유도체 화합물의 제조방법에서, 화학식 3의 화합물은 화학식 2의 화합물에 대해 바람직하게는 1.0 내지 1.5 당량, 보다 바람직하게는 1.0 당량으로 사용될 수 있다. In the method for preparing a heterobiarylpyridine derivative compound of
극성 양성자성 용매는 바람직하게는 메탄올, 에탄올, 디클로로에탄, 디메틸포름아마이드, 이들의 혼합물 등일 수 있고, 산 촉매는 AlCl3, AcOH, 포름산(formic acid), 트리플루오로아세트산(trifluoroacetic acid, TFA), HCl 등이 사용될 수 있으며, AlCl3이 바람직하다. 산 촉매의 사용량은, 산 촉매를 함유한 극성 양성자성 용매의 총 몰을 기준으로 5 내지 30 몰%, 바람직하게는 10 몰%일 수 있다.The polar protic solvent may preferably be methanol, ethanol, dichloroethane, dimethylformamide, mixtures thereof, and the like, and the acid catalyst may be AlCl 3 , AcOH, formic acid, trifluoroacetic acid (TFA). , HCl and the like can be used, with AlCl 3 being preferred. The amount of the acid catalyst used may be 5 to 30 mol%, preferably 10 mol% based on the total moles of the polar protic solvent containing the acid catalyst.
반응조건은 특별한 제한은 없으나, 예를 들어 환류(reflux) 조건하에 40 내지 70℃ 온도범위에서 2 내지 5 시간 동안 반응이 수행될 수 있다. The reaction conditions are not particularly limited, but for example, the reaction may be performed for 2 to 5 hours at a temperature range of 40 to 70 ° C. under reflux conditions.
아미노피라졸은 일반적으로 N/N-1 이친핵체(dinucleophile)로 작용하여 피라졸로[1,5-a]피리미딘을 생성하는 것으로 보고되고 있다(Quiroga, J.: Portilla, J.; Abonia, R.; Insuasty, B.; Nogueras, M.; Cobo, J. Tetrahedron Lett. 2008, 49, 6254-6256; Ghotekar, B. K.; Jachak, M. N.; Toche, R. B. J. Heterocyclic Chem. 2009, 46, 708-719). 이와 달리, 본 발명에서는, 화학식 3의 화합물이 N/C-4 이친핵체로서 작용하여 화학식 2의 화합물을 선택적으로 공격하여 우수한 위치특이성(regioselectivity)을 지닌 헤테로-바이아릴피리딘 유도체 화합물을 제조할 수 있다. Aminopyrazoles have generally been reported to act as N / N-1 dinucleophiles to produce pyrazolo [1,5-a] pyrimidines (Quiroga, J .: Portilla, J .; Abonia, R .; Insuasty, B .; Nogueras, M .; Cobo, J. Tetrahedron Lett . 2008, 49, 6254-6256; Ghotekar, BK; Jachak, MN; Toche, RB J. Heterocyclic Chem . 2009, 46, 708-719). In contrast, in the present invention, the compound of Formula 3 may act as an N / C-4 nucleophile to selectively attack the compound of
나아가, 기존의 헤테로바이아릴 화합물의 제조를 위한 교차-결합방법과 달리, 독성을 유발하는 전이 금속 촉매 또는 첨가제를 사용하고 있지 않으며, 입체장애(steric hindrance)로 인해 기존의 교차-결합법에 의해서는 얻기가 힘든 부피가 큰(bulky) 2,6-이치환된 바이아릴피리딘 화합물까지도 효과적으로 제조가 가능하다.Furthermore, unlike the cross-linking method for the preparation of the heterobiaryl compound, it does not use a transition metal catalyst or additive that causes toxicity, and by the conventional cross-linking method due to steric hindrance It is possible to effectively prepare even bulky 2,6-disubstituted biarylpyridine compounds which are difficult to obtain.
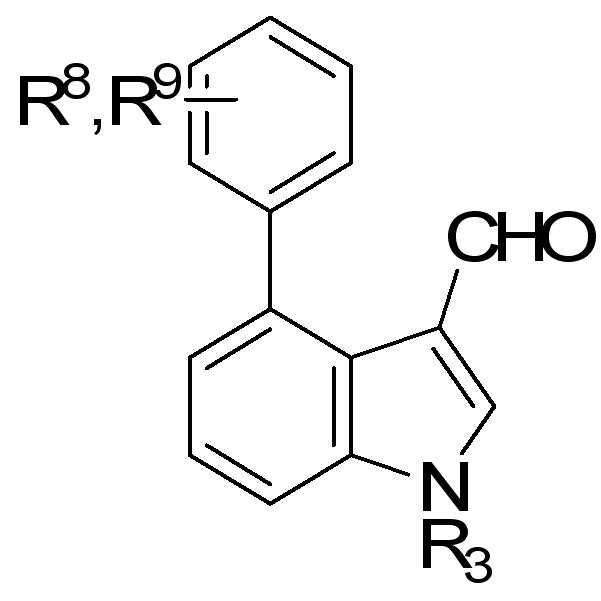
화학식 2의 화합물은 하기 화학식 2a로 표현되는 인돌-3-카복시알데하이드 유도체 화합물일 수 있다.The compound of
상기 식에서, R2 및 R3는 앞서 정의된 바와 같다.Wherein R 2 and R 3 are as defined above.
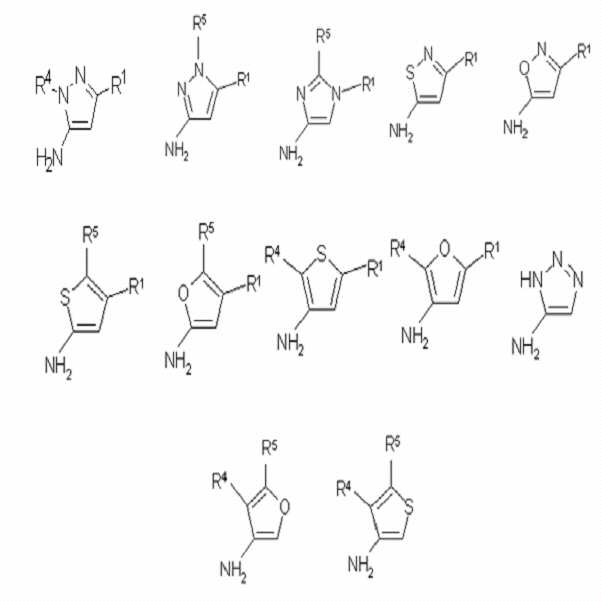
화학식 3의 화합물은 하기 구조식의 화합물들로 구성된 군으로부터 선택된 이친핵성 화합물(dinucleophiles)일 수 있으며, 이에 한정되는 것은 아니다:The compound of Formula 3 may be, but is not limited to, a dinucleophiles selected from the group consisting of compounds of the following structures:
상기 식에서, R1, R4 및 R5는 앞서 정의된 바와 같다.Wherein R 1 , R 4 and R 5 are as defined above.
화학식 3의 화합물은 특히 하기 화학식 3a의 아미노피라졸 유도체 화합물일 수 있다:The compound of
상기 식에서, R1 및 R4는 앞서 정의된 바와 같다.Wherein R 1 and R 4 are as defined above.
출발 물질로 사용된 화학식 2의 화합물과 화학식 3의 화합물은 시판 중인 제품을 구입하거나 당업계에 일반적으로 알려진 방법( Bioorg . Med . Chem . Lett . 15: 5039-5044 (2005))에 의해 합성하여 사용될 수 있다. Compounds of formula (2) and (3) used as starting materials may be obtained by purchasing a commercially available product or by synthesis by methods generally known in the art ( Bioorg . Med . Chem . Lett . 15: 5039-5044 (2005)) . Can be used.
특히, 하기 화학식 2b, 2c 및 2d의 인돌-3-카복시알데하이드 유도체 화합물들은 본 출원인이 제조예 8, 10, 11, 15 등에 기재된 방법에 따라 합성한 것이다.In particular, the indole-3-carboxyaldehyde derivative compounds of the following Chemical Formulas 2b, 2c and 2d are synthesized according to the methods described in Preparation Examples 8, 10, 11, 15, etc. of the applicant.
상기 식에서, Where
R3은 수소, 비치환되거나 C1-C6 알킬 또는 할로겐으로 치환된 C1-C6 알킬, 비치환되거나 C1-C6 알킬 또는 할로겐으로 치환된 C1-C6 아랄킬(aralkyl), 비치환 되거나 C1-C6 알킬 또는 할로겐으로 치환된 C1-C6 알콕시카보닐, 술포닐, COR6, CONR6R7 또는 CSNR6R7(여기서 R6 및 R7은 각각 독립적으로 수소, 비치환되거나 할로겐, C1-C6 알콕시 또는 C1-C6 알킬로 치환된 C1-C6 알킬, 비치환되거나 할로겐, C1-C6 알콕시 또는 C1-C6 알킬로 치환된 C3-C6 사이클로알킬, 비치환되거나 할로겐, C1-C6 알콕시 또는 C1-C6 알킬로 치환된 아릴, 또는 비치환되거나 C1-C6 알킬, C1-C6 할로알킬, C1-C6 알콕시 또는 할로겐으로 치환된 벤질임)이고; R 3 is hydrogen, unsubstituted or substituted by C1-C6 alkyl or halogen, C1-C6 alkyl, unsubstituted or substituted by C1-C6 alkyl or halogen, C1-C6 aralkyl, unsubstituted or C1-C6 alkyl Or C1-C6 alkoxycarbonyl, sulfonyl, COR 6 , CONR 6 R 7 or CSNR 6 R 7 substituted with halogen, wherein R 6 and R 7 are each independently hydrogen, unsubstituted or halogen, C1-C6 alkoxy or C1-C6 alkyl substituted with C1-C6 alkyl, C3-C6 cycloalkyl unsubstituted or substituted with halogen, C1-C6 alkoxy or C1-C6 alkyl, unsubstituted or halogen, C1-C6 alkoxy or C1-C6 alkyl Substituted aryl, or benzyl unsubstituted or substituted with C1-C6 alkyl, C1-C6 haloalkyl, C1-C6 alkoxy or halogen;
R8 및 R9는 각각 독립적으로 수소, 할로겐 원자, C1-C6 알킬, 또는 할로겐으로 치환된 C1-C6 알킬 또는 알콕시알킬이며;R 8 and R 9 are each independently hydrogen, a halogen atom, C 1 -C 6 alkyl, or C 1 -C 6 alkyl or alkoxyalkyl substituted with halogen;
단 R3, R8 및 R9는 동시에 수소인 경우는 제외된다.Except that R 3 , R 8 and R 9 are hydrogen at the same time.
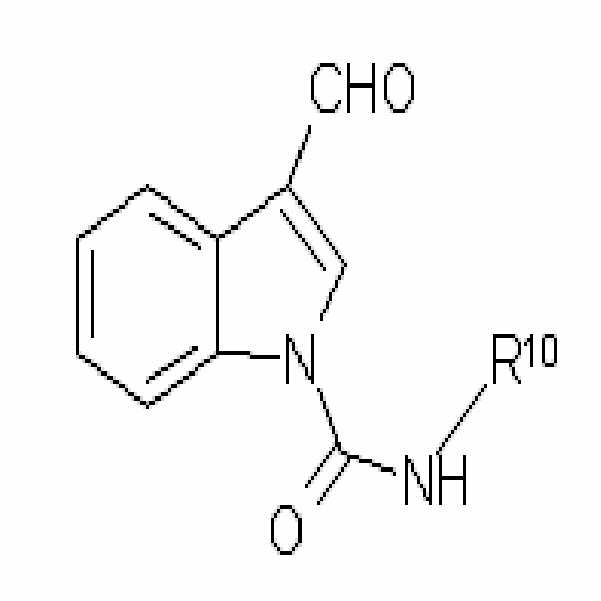
상기 식에서,Where
R3은 수소, 비치환되거나 C1-C6 알킬 또는 할로겐으로 치환된 C1-C6 알킬, 비치환되거나 C1-C6 알킬 또는 할로겐으로 치환된 C1-C6 아랄킬(aralkyl), 비치환되거나 C1-C6 알킬 또는 할로겐으로 치환된 C1-C6 알콕시카보닐, 술포닐, COR6, CONR6R7 또는 CSNR6R7(여기서 R6 및 R7은 각각 독립적으로 수소, 비치환되거나 할로겐, C1-C6 알콕시 또는 C1-C6 알킬로 치환된 C1-C6 알킬, 비치환되거나 할로겐, C1-C6 알콕시 또는 C1-C6 알킬로 치환된 C3-C6 사이클로알킬, 비치환되거나 할로겐, C1-C6 알콕시 또는 C1-C6 알킬로 치환된 아릴, 또는 비치환되거나 C1-C6 알킬, C1-C6 할로알킬, C1-C6 알콕시 또는 할로겐으로 치환된 벤질임)이다.R 3 is hydrogen, unsubstituted or substituted with C1-C6 alkyl or halogen, C1-C6 alkyl, unsubstituted or substituted with C1-C6 alkyl or halogen, C1-C6 aralkyl, unsubstituted or C1-C6 alkyl Or C1-C6 alkoxycarbonyl, sulfonyl, COR 6 , CONR 6 R 7 or CSNR 6 R 7 substituted with halogen, wherein R 6 and R 7 are each independently hydrogen, unsubstituted or halogen, C1-C6 alkoxy or C1-C6 alkyl substituted with C1-C6 alkyl, C3-C6 cycloalkyl unsubstituted or substituted with halogen, C1-C6 alkoxy or C1-C6 alkyl, unsubstituted or halogen, C1-C6 alkoxy or C1-C6 alkyl Substituted aryl, or benzyl unsubstituted or substituted with C1-C6 alkyl, C1-C6 haloalkyl, C1-C6 alkoxy or halogen.
상기 식에서,Where
R10은 수소, 비치환되거나 할로겐 또는 C1-C6 알콕시로 치환된 C1-C6 알킬, 비치환되거나 할로겐 또는 C1-C6 알콕시로 치환된 C3-C6 사이클로알킬, 비치환되거나 할로겐, C1-C6 알킬 또는 C1-C6 알콕시로 치환된 아릴, 또는 비치환되거나 C1-C6 알킬, C1-C6 할로알킬, C1-C6 알콕시 또는 할로겐으로 치환된 벤질이다.R 10 is hydrogen, C 1 -C 6 alkyl unsubstituted or substituted with halogen or C 1 -C 6 alkoxy, C 3 -C 6 cycloalkyl unsubstituted or substituted with halogen or C 1 -C 6 alkoxy, unsubstituted or halogen, C 1 -C 6 alkyl or Aryl substituted with C1-C6 alkoxy, or benzyl unsubstituted or substituted with C1-C6 alkyl, C1-C6 haloalkyl, C1-C6 alkoxy or halogen.
상기와 같은 제조방법에 의해 얻어지는 화학식 1의 대표적인 화합물은 하기 화학식 1a의 헤테로바이아릴 피라졸로[3,4-b] 피리딘 유도체 화합물이다:Representative compounds of formula (I) obtained by the preparation method as described above are heterobiaryl pyrazolo [3,4-b] pyridine derivative compounds of formula (I):
<화학식 1a><Formula 1a>
상기 식에서,Where
R1, R2, R3 및 R4는 앞서 정의된 바와 같다.R 1 , R 2 , R 3 and R 4 are as defined above.
상기 화학식 1a의 화합물을 제조하는 과정을 하기 반응식 1에 개략적으로 나타내고 있다:The process for preparing the compound of Formula 1a is schematically shown in Scheme 1:
상기 식에서, R1, R2, R3 및 R4는 앞서 정의된 바와 같다.Wherein R 1 , R 2 , R 3 and R 4 are as defined above.
상기 반응식 1에 나타낸 바와 같이, 극성 양성자성 용매(polar protic solvent)에서 화학식 2a의 화합물을 산 촉매(acid catalyst)의 존재하에 화학식 3a 의 화합물과 반응시켜 화학식 1a의 헤테로바이아릴 피라졸로[3,4-b] 피리딘 유도체를 제조할 수 있다.As shown in
화학식 1의 화합물의 구체적인 예로는,Specific examples of the compound of Formula 1,
2-(3-페닐-1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민;2- (3-phenyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) benzeneamine;
2-(1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민;2- ( 1H -pyrazolo [3,4-b] pyridin-5-yl) benzeneamine;
2-(3-메틸-1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민;2- (3-methyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) benzeneamine;
5-(2-아미노페닐)-1H-피라졸로[3,4-b]피리딘-3-올;5- (2-amino-phenyl) - 1H - pyrazolo [3,4-b] pyridin-3-ol;
2-(3-(4-메톡시페닐)-1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민;2- (3- (4-methoxyphenyl) - 1H - pyrazolo [3,4-b] pyridin-5-yl) benzene amine;
2-(3-(4-플루오로페닐)-1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민;2- (3- (4-fluorophenyl) - 1H - pyrazolo [3,4-b] pyridin-5-yl) benzene amine;
2-(3-(푸란-2-일)-1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민;2- (3- (furan-2-yl) - 1H - pyrazolo [3,4-b] pyridin-5-yl) benzene amine;
2-(3-(티오펜-2-일)-1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민;2- (3- (thiophen-2-yl) - 1H - pyrazolo [3,4-b] pyridin-5-yl) benzene amine;
3-니트로-2-(3-페닐-1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민;3-nitro-2- (3-phenyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) benzeneamine;
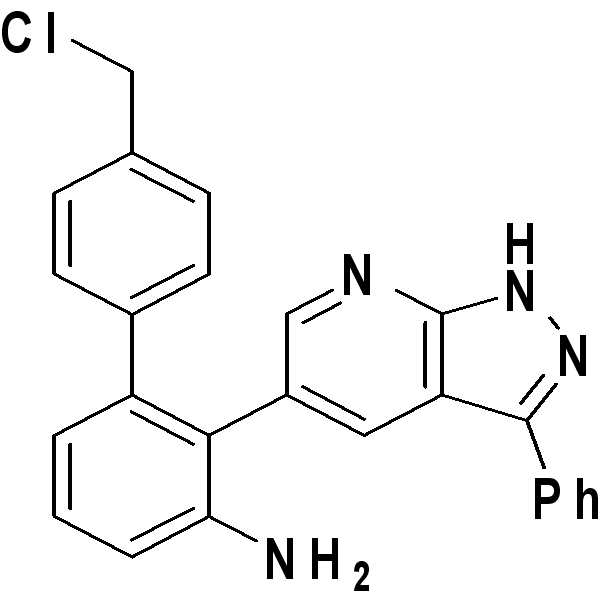
4'-(클로로메틸)-2-(3-페닐-1H-피라졸로[3,4-b]피리딘-5-일)비페닐-3-아민;4 '-(chloromethyl) -2- (3-phenyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) biphenyl-3-amine;
tert-부틸3-아미노-2-(3-페닐-1H-피라졸로[3,4-b]피리딘-5-일)페닐카바메이트;tert-butyl3-amino-2- (3-phenyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) phenylcarbamate;
메틸 3-아미노-2-(3-페닐-1H-피라졸로[3,4-b]피리딘-5-일)벤조에이트;Methyl 3-amino-2- (3-phenyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) benzoate;
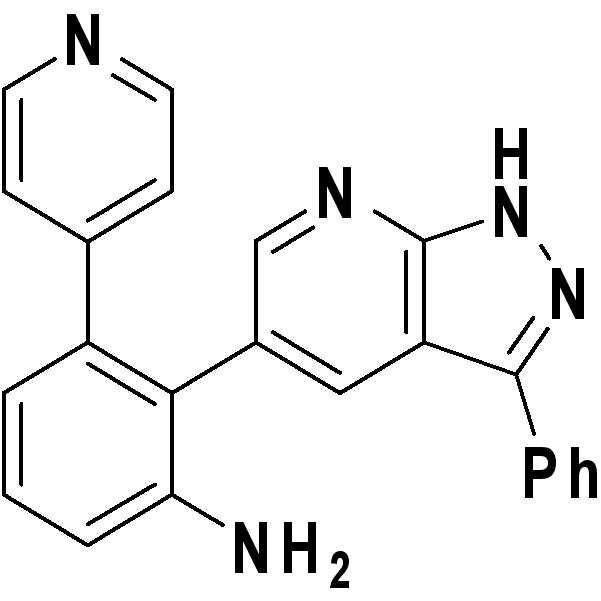
2-(3-페닐-1H-피라졸로[3,4-b]피리딘-5-일)-3-(피리딘-4-일)벤젠아민;2- (3-phenyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) -3- (pyridin-4-yl) benzeneamine;
2',6'-디메틸-2-(3-페닐-1H-피라졸로[3,4-b]피리딘-5-일)비페닐-3-아민;2 ', 6'-dimethyl-2- (3-phenyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) biphenyl-3-amine;
4-니트로-2-(3-페닐-1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민;4-nitro-2- (3-phenyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) benzeneamine;
2-(3-페닐-1H-피라졸로[3,4-b]피리딘-5-일)-4-(티오펜-2-일)벤젠아민;2- (3-phenyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) -4- (thiophen-2-yl) benzeneamine;
4-메톡시-2-(3-페닐-1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민;4-methoxy-2- (3-phenyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) benzeneamine;
4-클로로-2-(3-페닐-1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민;4-chloro-2- (3-phenyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) benzeneamine;
4-브로모-2-(3-페닐-1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민;4-bromo-2- (3-phenyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) benzeneamine;
5-클로로-2-(3-페닐-1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민;5-chloro-2- (3-phenyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) benzeneamine;
2-플루오로-6-(3-페닐-1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민;2-fluoro-6- (3-phenyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) benzeneamine;
2-클로로-6-(3-페닐-1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민;2-chloro-6- (3-phenyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) benzeneamine;
N-메틸-2-(3-페닐-1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민;N-methyl-2- (3-phenyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) benzeneamine;
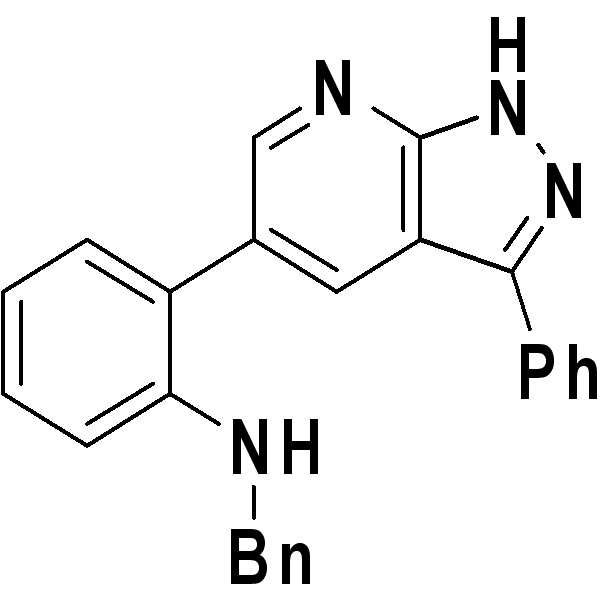
N-벤질-2-(3-페닐-1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민;N-benzyl-2- (3-phenyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) benzeneamine;
1-벤질-3-(2-(3-페닐-1H-피라졸로[3,4-b]피리딘-5-일)페닐)우레아;1-benzyl-3- (2- (3-phenyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) phenyl) urea;
2-(3-메틸-1-페닐-1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민;2- (3-methyl-1-phenyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) benzeneamine;
2-(3-(4-메톡시페닐)-1H-피라졸로[3,4-b]피리딘-5-일)-3-니트로벤젠아민;2- (3- (4-methoxyphenyl) - 1H - pyrazolo [3,4-b] pyridin-5-yl) -3-nitrobenzene-amine;
5-플루오로-2-(3-(4-메톡시페닐)-1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민; 5-fluoro-2- (3- (4-methoxyphenyl) - 1H - pyrazolo [3,4-b] pyridin-5-yl) benzene amine;
2-플루오로-6-(3-메틸-1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민;2-fluoro-6- (3-methyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) benzeneamine;
5-플루오로-2-(3-메틸-1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민;5-fluoro-2- (3-methyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) benzeneamine;
3-니트로-2-(3-메틸-1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민;3-nitro-2- (3-methyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) benzeneamine;
메틸 3-아미노-2-(3-메틸-1H-피라졸로[3,4-b]피리딘-5-일)벤조에이트;Methyl 3-amino-2- (3-methyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) benzoate;
4-브로모-2-(3-메틸-1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민;4-bromo-2- (3-methyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) benzeneamine;
4-클로로-2-(3-메틸-1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민;4-chloro-2- (3-methyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) benzeneamine;
5-클로로-2-(3-메틸-1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민;5-chloro-2- (3-methyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) benzeneamine;
N-메틸-2-(3-메틸-1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민; N-methyl-2- (3-methyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) benzeneamine;
N-벤질-2-(3-메틸-1H-피라졸로[3,4-b]피리딘-5-일)벤젠아민; 및N-benzyl-2- (3-methyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) benzeneamine; And
1-벤질-3-(2-(3-메틸-1H-피라졸로[3,4-b]피리딘-5-일)페닐)우레아1-benzyl-3- (2- (3-methyl- 1H -pyrazolo [3,4-b] pyridin-5-yl) phenyl) urea
를 들 수 있다..
본 발명에서 사용된 “아릴”은 탄소수 5 내지 6의 모노- 또는 폴리-사이클릭 방향족 환을 의미하며, 예를 들어 페닐을 들 수 있다. 아릴은 비치환되거나 1개 이상의 치환 그룹을 가질 수 있다.As used herein, “aryl” refers to a mono- or polycyclic aromatic ring having 5 to 6 carbon atoms, for example phenyl. Aryl may be unsubstituted or have one or more substituent groups.
“헤테로아릴”은 헤테로원자 O, N 또는 S를 1개 이상, 바람직하게는 동일하거나 상이한 헤테로원자를 1개 또는 2개 포함하는 5 내지 6원의 방향족 환을 의미하며, 예를 들어 피롤, 피라졸, 푸란, 티오펜, 피리딘, 피리미딘, 피라진, 피리다진, 옥사졸, 옥사디아졸, 테트라졸, 티아졸, 티아디아졸, 이미다졸, 벤즈이미다졸, 벤조티아펜. 벤조피롤, 벤조푸란 등을 의미한다. “Heteroaryl” means a 5-6 membered aromatic ring containing at least one, preferably one or two, heteroatoms O, N or S, for example pyrrole, pyra Sol, furan, thiophene, pyridine, pyrimidine, pyrazine, pyridazine, oxazole, oxadiazole, tetrazole, thiazole, thiadiazole, imidazole, benzimidazole, benzothiaphene. Benzopyrrole, benzofuran and the like.
이하에서는, 인돌 유도체의 개환(ring-opening) 및 아미노피라졸 유도체의 고리화(cyclization)에 대해 예상되는 원-스텝 메카니즘을 하기 반응식 2를 참조하여 간단히 설명하도록 한다.Hereinafter, one-step mechanisms expected for ring-opening of indole derivatives and cyclization of aminopyrazole derivatives will be briefly described with reference to
상기 반응식 2에 나타낸 바와 같이, 먼저, 이민을 형성한 다음 중간체 (I)의 고리형 자리옮김 반응(pericyclic rearrangement)에 의하거나 이미늄 친전자체(iminium electrophiles) (II)에 피라졸의 C-4 친핵성 첨가에 의해 분자내 고리화반응(intramolecular cyclization)을 유도하여 테트라사이클릭 화합물 (IV)을 얻는다. 마지막으로, 사이클릭 중간체 (IV)를 재방향족화(rearomatization)시켜 인돌 고리를 열고 결과적으로 원하는 헤테로-바이아릴 피라졸로[3,4-b]피리딘 유도체 화합물(1b)를 얻는다. As shown in
한편, 본 출원인은 본 발명의 화학식 1의 화합물의 위치특이성을 입증하기 위해, 하기 반응식 3에 나타낸 바와 같이, 인돌 3-카복시알데하이드(a)를 5-아미노-3-메틸-1-페닐-피라졸(b) 또는 4-(4-플루오로페닐)-1H-피라졸-5-아민(c)과 반응시킨 결과, 피라졸의 N-1 위치에서의 변형(화합물 b에 해당)은 C-4 친핵성 첨가에 의해 원하는 산물을 얻었지만, 피라졸의 C-4 위치에서의 변형(화합물 c에 해당)은 동 일조건하에서 원하는 산물이 얻어지지 않는 것을 확인하였다. 이는 아미노피라졸의 C-4가 친핵체로 작용하여 위치특이적으로 원-스텝 고리화반응을 수행함을 보여준다.On the other hand, Applicant, to demonstrate the positional specificity of the compound of
본 발명의 화학식 1의 헤테로바이아릴피리딘 유도체 화합물은 세포독성시험(cytotoxicity test)에서 정상세포에 비해 종양세포에 대한 증식을 선택적으로 억제하는 것으로 나타났다 (시험예 1 참조). 따라서, 본 발명에서 제조한 화학식 1의 헤테로바이아릴피리딘 유도체 화합물은 암의 예방 또는 치료용 약학 조성물의 유효성분으로 사용될 수 있다.The heterobiarylpyridine derivative compound of
이하, 제조예 및 실시예를 들어 본 발명을 상세히 설명하지만, 본 발명이 이들 실시예로만 한정되는 것은 아니다.Hereinafter, the present invention will be described in detail with reference to production examples and examples, but the present invention is not limited only to these examples.
제조예Manufacturing example 1: 4-니트로-1H-인돌-3- 1: 4-nitro-lH-indole-3- 카복시알데하이드Carboxyaldehyde
건조 아르곤 대기하에 4-니트로-1H-인돌(80 mg)을 무수 DMF중에 첨가하여 교반한 용액에, 0℃에서 인 염화물 산화물(phosphorus chloride oxide, 138 ㎕)을 첨가한 다음, 얻어진 혼합용액을 실온에서 1시간 동안 교반하였다. 이후, 반응 용액을 차가운 포화 탄산수소나트륨(NaHCO3) 수용액에 붓고 30분간 교반하였다.To a stirred solution by adding 4-nitro-1H-indole (80 mg) in anhydrous DMF under a dry argon atmosphere, phosphorus chloride oxide (138 μl) was added at 0 ° C., and the resulting mixed solution was allowed to come to room temperature. Stirred for 1 h. Thereafter, the reaction solution was poured into cold saturated aqueous sodium hydrogen carbonate (NaHCO 3 ) and stirred for 30 minutes.
얻어진 용액을 에틸아세테이트(EtOAc)로 수회 추출하고, 모아진 유기층을 무수 Na2SO4 상에서 건조한 다음 여과하였다. 여액을 감압하에 농축하고 실리카 겔상에서 플래시 컬럼 크로마토그래피(flash column chromatography)에 의해 정제하여 하기 구조식의 표제화합물(86 mg, 수율: 92%)을 수득하였다.The resulting solution was extracted several times with ethyl acetate (EtOAc), and the combined organic layers were dried over anhydrous Na 2 SO 4 and filtered. The filtrate was concentrated under reduced pressure and purified by flash column chromatography on silica gel to give the title compound (86 mg, yield: 92%) of the following structural formula.
1H NMR (500 MHz, Acetone-d 6 ) δ 11.84 (s, 1H) 10.29 (s, 1H) 8.43 (s, 1H) 8.00 (d, J = 8.07 Hz, 1H) 7.96 (d, J = 7.83 Hz, 1H) 7.47 (t, J = 8.07 Hz, 1H); 13C NMR (125 MHz, Acetone-d 6 ) δ 186.4, 140.4, 137.7, 123.4, 119.5, 118.5, 117.6; LRMS (MALDI) m/z calcd for C9H7N2O3 [M+H]+: 191.04; Found:191.39. 1 H NMR (500 MHz, Acetone- d 6 ) δ 11.84 (s, 1H) 10.29 (s, 1H) 8.43 (s, 1H) 8.00 (d, J = 8.07 Hz, 1H) 7.96 (d, J = 7.83 Hz , 1H) 7.47 (t, J = 8.07 Hz, 1H); 13 C NMR (125 MHz, Acetone- d 6) δ 186.4, 140.4, 137.7, 123.4, 119.5, 118.5, 117.6; LRMS (MALDI) m / z calcd for C 9 H 7 N 2 0 3 [M + H] + : 191.04; Found: 191.39.
제조예Manufacturing example 2: 2: 메틸methyl 3- 3- 포밀Formyl -1H-인돌-4--1H-indole-4- 카복실레이트Carboxylate
출발물질로서 인돌-4-카복실산 메틸에스터(175 mg)과 인 염화물 산화물 (280 ㎕)를 사용하는 것을 제외하고는 제조예 1과 동일한 방법으로 하기 구조식의 표제화합물(160 mg, 수율: 79%)을 수득하였다.The title compound (160 mg, Yield: 79%) of the following structural formula in the same manner as in Preparation Example 1, except that indole-4-carboxylic acid methyl ester (175 mg) and phosphorus chloride oxide (280 μl) were used as starting materials. Obtained.
1H NMR (500 MHz, Acetone-d 6 ) δ 11.49 (s, 1H) 10.43 (s, 1H) 8.28 (s, 1H) 7.80 (dd, J = 8.19, 1.10 Hz, 1H) 7.73 (dd, J = 7.46, 1.10 Hz, 1H) 7.34 (t, 1H) 3.94 (s, 3H); 13C NMR (125 MHz, Acetone-d 6 ) δ 187.6, 187.6, 169.5, 139.0, 135.8, 125.6, 124.7, 123.1, 123.1, 120.0, 117.2, 52.2, 52.2; LRMS (MALDI) m/z calcd for C11H9NO3 [M+H]+: 204.06; Found: 204.40. 1 H NMR (500 MHz, Acetone- d 6 ) δ 11.49 (s, 1H) 10.43 (s, 1H) 8.28 (s, 1H) 7.80 (dd, J = 8.19, 1.10 Hz, 1H) 7.73 (dd, J = 7.46, 1.10 Hz, 1 H) 7.34 (t, 1 H) 3.94 (s, 3 H); 13 C NMR (125 MHz, Acetone- d 6) δ 187.6, 187.6, 169.5, 139.0, 135.8, 125.6, 124.7, 123.1, 123.1, 120.0, 117.2, 52.2, 52.2; LRMS (MALDI) m / z calcd for C 11 H 9 NO 3 [M + H] < + >:204.06; Found: 204.40.
제조예Manufacturing example 3: 5-니트로-1H-인돌-3- 3: 5-nitro-1H-indole-3- 카복시알데하이드Carboxyaldehyde
출발물질로서 5-니트로-1H-인돌 (50 mg)과 인 염화물 산화물(86 ㎕)를 사용하는 것을 제외하고는 제조예 1과 동일한 방법으로 하기 구조식의 표제화합물(46 mg, 수율: 76%)을 수득하였다.The title compound (46 mg, yield: 76%) of the following structural formula in the same manner as in Preparation Example 1, except that 5-nitro-1H-indole (50 mg) and phosphorus chloride oxide (86 μl) were used as starting materials. Obtained.
1H NMR (500 MHz, DMSO-d 6) δ 12.70 (br. s., 1H) 10.03 (s, 1H) 8.94 (d, J = 2.21 Hz, 1H) 8.58 (s, 1H) 8.16 (dd, J = 8.98, 2.35 Hz, 1H) 7.72 (d, J = 9.12 Hz, 1H); 13C NMR (125 MHz, DMSO-d 6) δ 185.99, 185.96, 143.31, 141.92, 140.60, 123.97, 119.50, 119.23, 117.50, 113.68; LRMS (MALDI) m/z calcd for C9H6N2O3 [M+H]+: 191.04; Found: 191.40. 1 H NMR (500 MHz, DMSO- d 6 ) δ 12.70 (br.s., 1H) 10.03 (s, 1H) 8.94 (d, J = 2.21 Hz, 1H) 8.58 (s, 1H) 8.16 (dd, J = 8.98, 2.35 Hz, 1H) 7.72 (d, J = 9.12 Hz, 1H); 13 C NMR (125 MHz, DMSO- d 6 ) δ 185.99, 185.96, 143.31, 141.92, 140.60, 123.97, 119.50, 119.23, 117.50, 113.68; LRMS (MALDI) m / z calcd for C 9 H 6 N 2 O 3 [M + H] < + >:191.04; Found: 191.40.
제조예Manufacturing example 4: 5- 4: 5- 클로로Chloro -1H-인돌-3--1H-indole-3- 카복시알데하이드Carboxyaldehyde
출발물질로서 5-클로로-1H-인돌(70 mg)과 인 염화물 산화물 (130 ㎕)를 사용하는 것을 제외하고는 제조예 1과 동일한 방법으로 하기 구조식의 표제화합물(74 mg, 수율: 93%)을 수득하였다.The title compound (74 mg, yield: 93%) of the following structural formula in the same manner as in Preparation Example 1, except that 5-chloro-1H-indole (70 mg) and phosphorus chloride oxide (130 μl) were used as starting materials. Obtained.
1H NMR (500 MHz, Acetone-d 6 ) δ 11.33 (s, 1H) 10.03 (s, 1H) 8.29 (s, 1H) 8.23 (d, J = 1.96 Hz, 1H) 7.58 (d, J=8.56 Hz, 1H) 7.28 (dd, J = 8.68, 2.08 Hz, 1H); 13C NMR (125 MHz, Acetone-d 6 ) δ 185.4, 139.1, 136.7, 128.5, 126.6, 124.7, 121.5, 119.5, 114.5; LRMS (MALDI) m/z calcd for C9H6ClNO [M+H]+: 180.01; Found: 180.40. 1 H NMR (500 MHz, Acetone- d 6 ) δ 11.33 (s, 1H) 10.03 (s, 1H) 8.29 (s, 1H) 8.23 (d, J = 1.96 Hz, 1H) 7.58 (d, J = 8.56 Hz , 1H) 7.28 (dd, J = 8.68, 2.08 Hz, 1H); 13 C NMR (125 MHz, Acetone- d 6) δ 185.4, 139.1, 136.7, 128.5, 126.6, 124.7, 121.5, 119.5, 114.5; LRMS (MALDI) m / z calcd for C 9 H 6 ClNO [M + H] < + >:180.01; Found: 180.40.
제조예Manufacturing example 5: 6- 5: 6- 클로로Chloro -1H-인돌-3--1H-indole-3- 카복시알데하이드Carboxyaldehyde
출발물질로서 6-클로로-1H-인돌(70 mg)과 인 염화물 산화물(130 ㎕)를 사용하는 것을 제외하고는 제조예 1과 동일한 방법으로 하기 구조식의 표제화합물(64 mg, 수율: 77%)을 수득하였다.The title compound (64 mg, Yield: 77%) of the following structural formula in the same manner as in Preparation Example 1, except that 6-chloro-1H-indole (70 mg) and phosphorus chloride oxide (130 μl) were used as starting materials. Obtained.
1H NMR (500 MHz, Acetone-d 6 ) δ 11.27 (s, 1 H) 10.04 (s, 1 H) 8.26 (s, 1H) 8.20 (d, J = 8.56 Hz, 1H) 7.60 (d, J = 1.71 Hz, 1H) 7.26 (dd, J = 8.44, 1.83 Hz, 1H); 13C NMR (125 MHz, Acetone-d 6 ) δ 185.4, 138.8, 138.7, 129.7, 124.1, 123.4, 123.3, 119.9, 113.0; LRMS (MALDI) m/z calcd for C9H6ClNO [M+H]+: 180.01; Found: 180.40. 1 H NMR (500 MHz, Acetone- d 6 ) δ 11.27 (s, 1 H) 10.04 (s, 1 H) 8.26 (s, 1H) 8.20 (d, J = 8.56 Hz, 1H) 7.60 (d, J = 1.71 Hz, 1 H) 7.26 (dd, J = 8.44, 1.83 Hz, 1 H); 13 C NMR (125 MHz, Acetone- d 6) δ 185.4, 138.8, 138.7, 129.7, 124.1, 123.4, 123.3, 119.9, 113.0; LRMS (MALDI) m / z calcd for C 9 H 6 ClNO [M + H] < + >:180.01; Found: 180.40.
제조예Manufacturing example 6: 7- 6: 7- 플루오로Fluoro -1H-인돌-3--1H-indole-3- 카복시알데하이드Carboxyaldehyde
출발물질로서 7-플루오로-1H-인돌(60 mg)과 인 염화물 산화물 (124 ㎕)를 사용하는 것을 제외하고는 제조예 1과 동일한 방법으로 하기 구조식의 표제화합물(59 mg, 수율: 83%)을 수득하였다.The title compound of the following structural formula (59 mg, yield: 83%) in the same manner as in Preparation Example 1, except that 7-fluoro-1H-indole (60 mg) and phosphorus chloride oxide (124 μl) were used as starting materials. ) Was obtained.
1H NMR (500 MHz, Acetone-d 6 ) δ 11.62 (br. s., 1 H) 10.08 (s, 1 H) 8.29 (s, 1H) 8.03 (d, J=7.83 Hz, 1H) 7.18-7.31 (m, 1 H) 7.08 (dd, J=11.25, 7.83 Hz, 1 H); 13C NMR (125 MHz, Acetone-d 6 ) δ 185.6, 151.3, 149.4, 138.5, 129.1, 129.1, 123.8, 123.8, 120.7, 118.3, 118.3, 109.5, 109.4; LRMS (MALDI) m/z calcd for C9H6FNO [M+H]+: 164.04; Found: 164.42. 1 H NMR (500 MHz, Acetone- d 6 ) δ 11.62 (br. S., 1 H) 10.08 (s, 1 H) 8.29 (s, 1H) 8.03 (d, J = 7.83 Hz, 1H) 7.18-7.31 (m, 1H) 7.08 (dd, J = 11.25, 7.83 Hz, 1H); 13 C NMR (125 MHz, Acetone- d 6) δ 185.6, 151.3, 149.4, 138.5, 129.1, 129.1, 123.8, 123.8, 120.7, 118.3, 118.3, 109.5, 109.4; LRMS (MALDI) m / z calcd for C 9 H 6 FNO [M + H] < + >:164.04; Found: 164.42.
제조예Manufacturing example 7: 7- 7: 7- 클로로Chloro -1H-인돌-3--1H-indole-3- 카복시알데하이드Carboxyaldehyde
출발물질로서 7-클로로-1H-인돌(70 mg)과 인 염화물 산화물(130 ㎕)를 사용하는 것을 제외하고는 제조예 1과 동일한 방법으로 하기 구조식의 표제화합물(66 mg, 수율: 79%)을 수득하였다.The title compound (66 mg, yield: 79%) of the following structural formula in the same manner as in Preparation Example 1, except that 7-chloro-1H-indole (70 mg) and phosphorus chloride oxide (130 μl) were used as starting materials. Obtained.
1H NMR (500 MHz, Acetone-d 6 ) δ 11.49 (br. s., 1 H) 10.08 (s, 1 H) 8.30 (s, 1 H) 8.20 (dd, J=7.83, 0.98 Hz, 1 H) 7.34-7.36 (m, 1 H) 7.26 (t, J=7.70 Hz, 1 H); 13C NMR (125 MHz, Acetone-d 6 ) δ 185.6, 138.5, 135.1, 127.2, 124.1, 124.0, 121.1, 120.7, 117.7; LRMS (MALDI) m/z calcd for C9H6ClNO [M+H]+: 180.01; Found: 180.43. 1 H NMR (500 MHz, Acetone- d 6 ) δ 11.49 (br. S., 1 H) 10.08 (s, 1 H) 8.30 (s, 1 H) 8.20 (dd, J = 7.83, 0.98 Hz, 1 H ) 7.34-7.36 (m, 1H) 7.26 (t, J = 7.70 Hz, 1H); 13 C NMR (125 MHz, Acetone- d 6) δ 185.6, 138.5, 135.1, 127.2, 124.1, 124.0, 121.1, 120.7, 117.7; LRMS (MALDI) m / z calcd for C 9 H 6 ClNO [M + H] < + >:180.01; Found: 180.43.
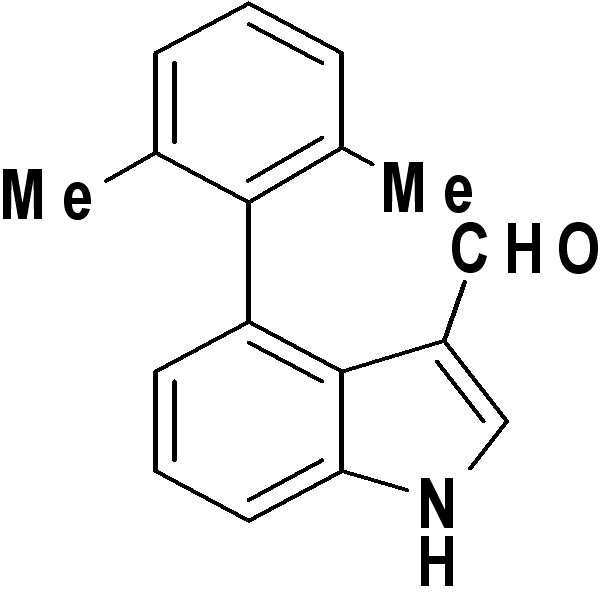
제조예Manufacturing example 8: 4-(4-( 8: 4- (4- ( 클로로메틸Chloromethyl )) 페닐Phenyl )-1H-인돌-3-) -1H-indole-3- 카복시알데하이드Carboxyaldehyde
(8-1) 4-(4-(8-1) 4- (4- 히드록시메틸페닐Hydroxymethylphenyl )) 인돌의Indole 제조 Produce
물, 톨루엔 및 에탄올(부피비: 1:1:3)의 혼합 용매에 4-브로모인돌(1 당량, 125 ㎕)을 용해시켜 얻어진 용액에 4-(히드록시메틸)페닐보론산(1.5 당량, 228 mg), Pd(OAc)2 (10 mol%, 22 mg) 및 Na2SO4(3 당량, 318 mg)을 첨가하고 얻어진 혼합용액을 80℃에서 8시간 동안 교반하였다.4- (hydroxymethyl) phenylboronic acid (1.5 equiv., In a solution obtained by dissolving 4-bromoindole (1 equiv, 125 μl) in a mixed solvent of water, toluene and ethanol (volume ratio 1: 1: 1: 3) 228 mg), Pd (OAc) 2 (10 mol%, 22 mg) and Na 2 SO 4 (3 equiv, 318 mg) were added and the resulting mixed solution was stirred at 80 ° C. for 8 hours.
반응이 종결된 후, 반응 용액을 에틸아세테이트로 희석한 다음 유기층을 분리하여 셀라이트를 통해 여과하였다. 여액을 감압하에 농축한 다음 실리카 겔상에서 플래시 컬럼 크로마토그래피로 정제하여 하기 구조식의 황색 고형물(143 mg, 수율: 64%)을 수득하였다. After the reaction was completed, the reaction solution was diluted with ethyl acetate, and then the organic layer was separated and filtered through celite. The filtrate was concentrated under reduced pressure and then purified by flash column chromatography on silica gel to give a yellow solid (143 mg, yield: 64%) of the following structural formula.
1H NMR (500 MHz, acetone) δ ppm 10.39 (br. s., 1 H) 7.67 (d, J=7.34 Hz, 2 H) 7.49 (d, J=7.58 Hz, 2 H) 7.43 (d, J=8.07 Hz, 1 H) 7.35-7.41 (m, 1 H) 7.20 (t, J=7.58 Hz, 1 H) 7.12 (d, J=7.34 Hz, 1 H) 6.60-6.68 (m, 1 H) 4.71 (d, J=5.38 Hz, 2 H) 4.25 (t, J=5.50 Hz, 1 H); 13C NMR (125 MHz, acetone) δ ppm 141.92 141.12 137.76 134,60 129.13 127.89 127.67 127.09 126.00 125.95 122.48 122.45 119.69 119.67 111.36 101.69 101.68; LRMS (MALDI) m/z calcd for C15H13NO [M+H]+: 224.10; Found: 224.44 1 H NMR (500 MHz, acetone) δ ppm 10.39 (br. S., 1 H) 7.67 (d, J = 7.34 Hz, 2 H) 7.49 (d, J = 7.58 Hz, 2 H) 7.43 (d, J = 8.07 Hz, 1 H) 7.35-7.41 (m, 1 H) 7.20 (t, J = 7.58 Hz, 1 H) 7.12 (d, J = 7.34 Hz, 1 H) 6.60-6.68 (m, 1 H) 4.71 (d, J = 5.38 Hz, 2H) 4.25 (t, J = 5.50 Hz, 1H); 13 C NMR (125 MHz, acetone) δ ppm 141.92 141.12 137.76 134,60 129.13 127.89 127.67 127.09 126.00 125.95 122.48 122.45 119.69 119.67 111.36 101.69 101.68; LRMS (MALDI) m / z calcd for C 15 H 13 NO [M + H] < + >:224.10; Found: 224.44
(8-2) 4-(4-((8-2) 4- (4- ( 클로로메틸Chloromethyl )) 페닐Phenyl )-1H-인돌-3-) -1H-indole-3- 카복시알데하이드의Carboxyaldehyde 제조 Produce
상기 (8-1)에서 얻어진 4-(히드록시메틸페닐)인돌(1 당량, 55 mg)을 건조 아르곤 대기하에 무수 DMF중에 첨가하여 교반한 용액에, 0℃에서 인 염화물 산화물(3 당량, 69 ㎕)을 첨가한 다음, 얻어진 혼합용액을 실온에서 1시간 동안 교반하였다. 이후, 반응 용액을 차가운 포화 탄산수소나트륨(NaHCO3) 수용액에 붓고 30분간 교반하였다.4- (hydroxymethylphenyl) indole (1 equivalent, 55 mg) obtained in the above (8-1) was added to anhydrous DMF under a dry argon atmosphere and stirred, and then, phosphorus chloride oxide (3 equivalents, 69 µl) was added at 0 ° C. ) Was added and the resulting mixed solution was stirred at room temperature for 1 hour. Thereafter, the reaction solution was poured into cold saturated aqueous sodium hydrogen carbonate (NaHCO 3 ) and stirred for 30 minutes.
얻어진 용액을 에틸아세테이트로 수회 추출하고, 모아진 유기층을 무수 Na2SO4 상에서 건조한 다음 여과하였다. 여액을 감압하에 농축하고 실리카 겔상에서 플래시 컬럼 크로마토그래피로 정제하여 하기 구조식의 표제화합물(32 mg, 수율: 52%)을 수득하였다.The obtained solution was extracted several times with ethyl acetate, and the combined organic layers were dried over anhydrous Na 2 SO 4 and filtered. The filtrate was concentrated under reduced pressure and purified by flash column chromatography on silica gel to give the title compound (32 mg, yield: 52%) of the following structural formula.
1H NMR (500 MHz, Acetone-d 6 ) δ 11.41 (s, 1H) 9.54 (s, 1H) 8.16 (d, J = 3.18 Hz, 1H) 7.60 (dd, J = 8.31, 0.98 Hz, 1H) 7.52-7.58 (m, 4H) 7.33 (dd, J = 8.31, 7.34 Hz, 4H) 7.13 (dd, J = 7.34, 0.98 Hz, 1H) 4.81 (s, 2H); 13C NMR (125 MHz, Acetone-d 6 ) δ 185.2, 185.2, 143.5, 138.6, 138.1, 135.6, 133.2, 133.0, 130.2, 124.7, 124.2, 123.8, 119.8, 112.8, 112.7, 46.7; LRMS (MALDI) m/z calcd for C16H12ClNO [M+H]+: 270.06; Found: 270.41 1 H NMR (500 MHz, Acetone- d 6 ) δ 11.41 (s, 1H) 9.54 (s, 1H) 8.16 (d, J = 3.18 Hz, 1H) 7.60 (dd, J = 8.31, 0.98 Hz, 1H) 7.52 -7.58 (m, 4H) 7.33 (dd, J = 8.31, 7.34 Hz, 4H) 7.13 (dd, J = 7.34, 0.98 Hz, 1H) 4.81 (s, 2H); 13 C NMR (125 MHz, Acetone- d 6) δ 185.2, 185.2, 143.5, 138.6, 138.1, 135.6, 133.2, 133.0, 130.2, 124.7, 124.2, 123.8, 119.8, 112.8, 112.7, 46.7; LRMS (MALDI) m / z calcd for C 16 H 12 ClNO [M + H] + : 270.06; Found: 270.41
제조예Manufacturing example 9: 4-(피리딘-4-일)-1H-인돌-3- 9: 4- (pyridin-4-yl) -1H-indole-3- 카복시알데하이드Carboxyaldehyde
(9-1) 4-(4-(9-1) 4- (4- 피리디닐Pyridinyl )) 인돌의Indole 제조 Produce
4-브로모인돌(1 당량, 62 ㎕), 4-피리디닐 보론산 (2 당량, 205 mg), Pd(PPh3)4 (20 mol%, 115 mg), K2CO3(4 당량, 276 mg)을 물, 톨루엔, 에탄올의 혼합용매(부피비: 1:1:3)에 용해시킨 다음 반응 혼합물을 80℃에서 밤새 교반하였다. 4-bromoindole (1 equiv, 62 μl), 4-pyridinyl boronic acid (2 equiv, 205 mg), Pd (PPh 3 ) 4 (20 mol%, 115 mg), K 2 CO 3 (4 equiv, 276 mg) was dissolved in a mixed solvent of water, toluene and ethanol (volume ratio 1: 1: 1) and then the reaction mixture was stirred at 80 ° C. overnight.
반응이 종결된 후, 반응 용액을 에틸아세테이트로 희석한 다음 유기층을 분리하여 셀라이트를 이용하여 여과하였다. 여액을 감압하에 농축하고 실리카겔상에 서 플래시 컬럼 크로마토그래피로 정제하여 하기 구조식의 백색 고형물 (85 mg, 수율: 88%)을 수득하였다.After the reaction was completed, the reaction solution was diluted with ethyl acetate and then the organic layer was separated and filtered using celite. The filtrate was concentrated under reduced pressure and purified by flash column chromatography on silica gel to give a white solid (85 mg, yield: 88%) of the following structural formula.
1H NMR (500 MHz, acetone) δ ppm 10.59 (br. s., 1 H) 8.68 (d, J=4.40 Hz, 2 H) 7.69 (d, J=4.40 Hz, 2 H) 7.56 (d, J=5.87 Hz, 1 H) 7.45-7.50 (m, 1 H) 7.20-7.30 (m, 2 H) 6.71-6.74 (m, 1 H); 13C NMR (125 MHz, DMSO-d6) δ ppm 149.94, 148.21, 136.48, 129.86, 126.62, 125.23, 122.96, 121.36, 118.91, 112.38, 99.77; LRMS (MALDI) m/z calcd for C13H10N2 [M+H]+: 195.08; Found: 195.44. 1 H NMR (500 MHz, acetone) δ ppm 10.59 (br. S., 1 H) 8.68 (d, J = 4.40 Hz, 2 H) 7.69 (d, J = 4.40 Hz, 2 H) 7.56 (d, J = 5.87 Hz, 1H) 7.45-7.50 (m, 1H) 7.20-7.30 (m, 2H) 6.71-6.74 (m, 1H); 13 C NMR (125 MHz, DMSO-d 6 ) δ ppm 149.94, 148.21, 136.48, 129.86, 126.62, 125.23, 122.96, 121.36, 118.91, 112.38, 99.77; LRMS (MALDI) m / z calcd for C 13 H 10 N 2 [M + H] < + >:195.08; Found: 195.44.
(9-2) 4-(피리딘-4-일)-1H-인돌-3-(9-2) 4- (pyridin-4-yl) -1H-indole-3- 카복시알데하이드의Carboxyaldehyde 제조 Produce
건조 아르곤 대기하에 상기 (9-1)에서 얻어진 4-(4-피리디닐)인돌(85 mg)을 무수 DMF중에 첨가하여 교반한 용액에, 0℃에서 인 염화물 산화물(3 당량, 122 ㎕)을 첨가한 다음, 얻어진 혼합용액을 실온에서 1시간 동안 교반하였다. 이후, 반응 용액을 차가운 포화 탄산수소나트륨(NaHCO3) 수용액에 붓고 30분간 교반하였다.Phosphorous chloride oxide (3 equivalents, 122 µl) was added to the stirred solution by adding 4- (4-pyridinyl) indole (85 mg) obtained in the above (9-1) in anhydrous DMF under a dry argon atmosphere. After the addition, the resulting mixed solution was stirred at room temperature for 1 hour. Thereafter, the reaction solution was poured into cold saturated aqueous sodium hydrogen carbonate (NaHCO 3 ) and stirred for 30 minutes.
얻어진 용액을 에틸아세테이트로 수회 추출하고, 모아진 유기층을 무수 Na2SO4 상에서 건조한 다음 여과하였다. 여액을 감압하에 농축하고 실리카 겔상에서 플래시 컬럼 크로마토그래피로 정제하여 하기 구조식의 표제화합물(46 mg, 수율: 47%)을 수득하였다.The obtained solution was extracted several times with ethyl acetate, and the combined organic layers were dried over anhydrous Na 2 SO 4 and filtered. The filtrate was concentrated under reduced pressure and purified by flash column chromatography on silica gel to give the title compound (46 mg, yield: 47%) of the following structural formula.
1H NMR (500 MHz, DMSO-d 6) δ 9.49 (s, 1H) 8.53-8.62 (m, 2H) 8.24 (s, 1H) 7.57 (d, J = 8.07 Hz, 1H) 7.37-7.45 (m, 2H) 7.24 (t, J = 7.70 Hz, 1H) 7.04 (d, J = 7.09 Hz, 1H); 13C NMR (125 MHz, DMSO-d 6) δ 181.8, 150.1, 149.0, 142.2, 141.2, 131.6, 124.1, 124.0, 123.2, 122.4, 121.8, 117.8, 114.6; LRMS (MALDI) m/z calcd for C14H10N2O [M+H]+: 223.08; Found: 223.41. 1 H NMR (500 MHz, DMSO- d 6 ) δ 9.49 (s, 1H) 8.53-8.62 (m, 2H) 8.24 (s, 1H) 7.57 (d, J = 8.07 Hz, 1H) 7.37-7.45 (m, 2H) 7.24 (t, J = 7.70 Hz, 1 H) 7.04 (d, J = 7.09 Hz, 1 H); 13 C NMR (125 MHz, DMSO- d 6) δ 181.8, 150.1, 149.0, 142.2, 141.2, 131.6, 124.1, 124.0, 123.2, 122.4, 121.8, 117.8, 114.6; LRMS (MALDI) m / z calcd for C 14 H 10 N 2 O [M + H] < + >:223.08; Found: 223.41.
제조예Manufacturing example 10: 4-(2,6- 10: 4- (2,6- 디메틸페닐Dimethylphenyl )-1H-인돌-3-) -1H-indole-3- 카복시알데하이드Carboxyaldehyde
(10-1) 4-(2,6-(10-1) 4- (2,6- 디메틸페닐Dimethylphenyl )) 인돌의Indole 제조 Produce
4-브로모인돌(1 당량, 124 ㎕), 2,6-디메틸페닐 보론산 (2 당량, 300 mg), Pd(PPh3)4 (10 mol%, 115 mg), K2CO3(4 당량: 553 mg)을 물, 톨루엔, 에탄올의 혼합용매(부피비: 1:2:2)에 용해시킨 다음 반응 혼합물을 70℃에서 밤새 교반하였다. 4-bromoindole (1 equiv, 124 μl), 2,6-dimethylphenyl boronic acid (2 equiv, 300 mg), Pd (PPh 3 ) 4 (10 mol%, 115 mg), K 2 CO 3 (4 Equivalent: 553 mg) was dissolved in a mixed solvent of water, toluene and ethanol (volume ratio 1: 2: 2) and then the reaction mixture was stirred at 70 ° C. overnight.
반응이 종결된 후, 반응 용액을 에틸아세테이트로 희석하고 유기층을 분리하 여 셀라이트로 여과하였다. 여액을 감압하에 농축한 다음 실리카겔 플래시 컬럼 크로마토그래피로 정제하여 하기 구조식의 백색 고형물 (97 mg, 수율: 44%)을 수득하였다.After the reaction was completed, the reaction solution was diluted with ethyl acetate, the organic layer was separated and filtered through Celite. The filtrate was concentrated under reduced pressure and purified by silica gel flash column chromatography to give a white solid (97 mg, yield: 44%) of the following structural formula.
1H NMR (500 MHz, acetone) δ ppm 10.31 (br. s., 1 H) 7.44 (d, J=8.07 Hz, 1 H) 7.28-7.30 (m, 1 H) 7.11-7.21 (m, 4 H) 6.78 (dd, J=7.21, 0.86 Hz, 1 H) 5.94-5.97 (m, 1 H) 1.96 (s, 6 H); 13C NMR (125 MHz, acetone) δ ppm 141.5, 137.4, 137.1, 133.9, 128.0, 128.0, 127.7, 125.7, 122.3, 120.1, 111.0, 101.5, 101.5, 20.6, 20.6; LRMS (MALDI) m/z calcd for C16H15N [M+H]+: 222.12; Found: 222.47. 1 H NMR (500 MHz, acetone) δ ppm 10.31 (br.s., 1 H) 7.44 (d, J = 8.07 Hz, 1 H) 7.28-7.30 (m, 1 H) 7.11-7.21 (m, 4 H ) 6.78 (dd, J = 7.21, 0.86 Hz, 1H) 5.94-5.97 (m, 1H) 1.96 (s, 6H); 13 C NMR (125 MHz, acetone) δ ppm 141.5, 137.4, 137.1, 133.9, 128.0, 128.0, 127.7, 125.7, 122.3, 120.1, 111.0, 101.5, 101.5, 20.6, 20.6; LRMS (MALDI) m / z calcd for C 16 H 15 N [M + H] < + >:222.12; Found: 222.47.
(10-2) 4-(2,6-(10-2) 4- (2,6- 디메틸페닐Dimethylphenyl )-1H-인돌-3-) -1H-indole-3- 카복시알데하이드의Carboxyaldehyde 제조 Produce
건조 아르곤 대기하에 상기 (10-1)에서 얻어진 4-(2,6-디메틸페닐)인돌 (66 mg)을 무수 DMF중에 첨가하여 교반한 용액에, 0℃에서 인 염화물 산화물(3 당량, 84 ㎕)을 첨가한 다음, 얻어진 혼합용액을 실온에서 1시간 동안 교반하였다. 이후, 반응 용액을 차가운 포화 탄산수소나트륨(NaHCO3) 수용액에 붓고 30분간 교반하 였다.Phosphorous chloride oxide (3 equivalents, 84 µl) was added to the stirred solution by adding 4- (2,6-dimethylphenyl) indole (66 mg) obtained in the above (10-1) in anhydrous DMF under a dry argon atmosphere. ) Was added and the resulting mixed solution was stirred at room temperature for 1 hour. Thereafter, the reaction solution was poured into cold saturated aqueous sodium hydrogen carbonate (NaHCO 3 ) and stirred for 30 minutes.
얻어진 용액을 에틸아세테이트로 수회 추출하고, 모아진 유기층을 무수 Na2SO4 상에서 건조한 다음 여과하였다. 여액을 감압하에 농축하고 실리카 겔상에서 플래시 컬럼 크로마토그래피로 정제하여 하기 구조식의 표제화합물(64 mg, 수율: 85%)을 수득하였다.The obtained solution was extracted several times with ethyl acetate, and the combined organic layers were dried over anhydrous Na 2 SO 4 and filtered. The filtrate was concentrated under reduced pressure and purified by flash column chromatography on silica gel to give the title compound (64 mg, yield: 85%) of the following structural formula.
1H NMR (500 MHz, Acetone-d 6 ) δ 11.39 (s, 1H) 8.92 (s, 1H) 8.11 (s, 1H) 7.60 (dd, J = 8.31, 0.98 Hz, 1H) 7.36 (dd, J = 8.19, 7.21 Hz, 1H) 7.19-7.26 (m, 1H) 7.13-7.20 (m, 2H) 6.99 (dd, J = 7.34, 0.98 Hz, 1H) 1.97 (s, 6H); 13C NMR (125 MHz, Acetone-d 6 ) δ 185.45, 185.44, 141.89, 138.21, 136.8, 133.85, 131.52, 128.4, 128.39, 128.29, 125.26, 123.96, 123.41, 120.01, 112.21, 20.36; LRMS (MALDI) m/z calcd for C17H15NO [M+H]+: 250.12; Found: 250.42. 1 H NMR (500 MHz, Acetone- d 6 ) δ 11.39 (s, 1H) 8.92 (s, 1H) 8.11 (s, 1H) 7.60 (dd, J = 8.31, 0.98 Hz, 1H) 7.36 (dd, J = 8.19, 7.21 Hz, 1H) 7.19-7.26 (m, 1H) 7.13-7.20 (m, 2H) 6.99 (dd, J = 7.34, 0.98 Hz, 1H) 1.97 (s, 6H); 13 C NMR (125 MHz, Acetone- d 6 ) δ 185.45, 185.44, 141.89, 138.21, 136.8, 133.85, 131.52, 128.4, 128.39, 128.29, 125.26, 123.96, 123.41, 120.01, 112.21, 20.36; LRMS (MALDI) m / z calcd for C 17 H 15 NO [M + H] < + >:250.12; Found: 250.42.
제조예Manufacturing example 11: 11: terttert -부틸 4-아미노-3--Butyl 4-amino-3- 포밀Formyl -1H-인돌-1--1H-indole-1- 카복실레이트Carboxylate
4-니트로-1H-인돌(81 mg)과 인 염화물 산화물(140 ㎕)를 이용하여 제조예 1의 과정에 따라 얻어진 조 반응 용액(crude reaction mixture)) 추가 정제없이 아세토니트릴(ACN) (5 ㎖)에 용해시켰다. 여기에 DMAP(0.2 당량, 12 mg) 및 Boc2O(1.5 당량, 164 mg)을 첨가하여 실온에서 밤새 교반하였다. Crude reaction mixture obtained according to the procedure of Preparation Example 1 using 4-nitro-1H-indole (81 mg) and phosphorus chloride oxide (140 μl), without any further purification of acetonitrile (ACN) (5 mL). )). To this was added DMAP (0.2 equiv, 12 mg) and Boc 2 O (1.5 equiv, 164 mg) and stirred overnight at room temperature.
반응이 종결된 후, 반응 용액을 증발시켜 휘발물질을 제거한 다음 실리카 겔상에서 플래시 컬럼 크로마토그래피로 정제하여 황색 고체상의 N-Boc-4-니트로인돌-3-카복시알데하이드(140 mg, 수율: 95%)를 수득하였다.After completion of the reaction, the reaction solution was evaporated to remove volatiles and purified by flash column chromatography on silica gel to give N-Boc-4-nitroindole-3-carboxyaldehyde (140 mg, yield: 95%) as a yellow solid. ) Was obtained.
1H NMR (500 MHz, acetone) δ ppm 10.17(s, 1 H) 8.61-8.64 (m, 2H) 7.98 (d, J=7.83 Hz, 1 H) 7.65 (t, J=8.19 Hz, 1 H) 1.75 (s, 9 H); 13C NMR (125 MHz, acetone) δ ppm 149.79, 141.35, 137.91, 131.31, 125.59, 124.70, 122.43, 120.25, 120.22, 106.83, 86.03, 28.26; LRMS 미검출. 1 H NMR (500 MHz, acetone) δ ppm 10.17 (s, 1H) 8.61-8.64 (m, 2H) 7.98 (d, J = 7.83 Hz, 1H) 7.65 (t, J = 8.19 Hz, 1H) 1.75 (s, 9 H); 13 C NMR (125 MHz, acetone) δ ppm 149.79, 141.35, 137.91, 131.31, 125.59, 124.70, 122.43, 120.25, 120.22, 106.83, 86.03, 28.26; LRMS not detected.
N-Boc-4-니트로인돌-3-카복시알데하이드(122 mg)를 실온에서 30분간 SnCl2·H2O(10 당량, 948 mg)을 함유한 DMF로 처리한 다음, 반응 용액을 NaHCO3 ( aq ) 포화용액에 첨가하고 에틸아세테이트로 수회 추출하였다.N-Boc-4-nitroindole-3-carboxyaldehyde (122 mg) was treated with DMF containing SnCl 2 · H 2 O (10 equiv, 948 mg) at room temperature for 30 minutes, and then the reaction solution was treated with NaHCO 3 ( aq ) was added to a saturated solution and extracted several times with ethyl acetate.
모아진 유기층을 무수 Na2SO4 상에서 건조한 다음 여과하였다. 여액을 감압하에 농축하여 황색 고체상의 표제 화합물(45 mg, 수율: 41%)을 수득하였다.The combined organic layers were dried over anhydrous Na 2 SO 4 and filtered. The filtrate was concentrated under reduced pressure to give the title compound (45 mg, yield: 41%) as a yellow solid.
1H NMR (500 MHz, acetone) δ ppm 9.82 (s, 1 H) 8.44 (s, 1 H) 7.39 (d, J=8.31 Hz, 1 H) 7.11 (t, J=7.95 Hz, 1 H) 6.54 (d, J=7.83 Hz, 1 H) 6.15 (br. s., 2 H) 1.69 (s, 9 H); 13C NMR (75 MHz, acetone) δ ppm 187.6, 149.6, 144.6, 140.2, 139.4, 128.5, 124.4, 113.4, 109.3, 103.6, 86.1, 28.2; LRMS (ESI+) m/z calcd for C14H16N2O3 [M+Na]+: 283.11; Found: 283.10 1 H NMR (500 MHz, acetone) δ ppm 9.82 (s, 1 H) 8.44 (s, 1 H) 7.39 (d, J = 8.31 Hz, 1 H) 7.11 (t, J = 7.95 Hz, 1 H) 6.54 (d, J = 7.83 Hz, 1H) 6.15 (br. s., 2H) 1.69 (s, 9H); 13 C NMR (75 MHz, acetone) δ ppm 187.6, 149.6, 144.6, 140.2, 139.4, 128.5, 124.4, 113.4, 109.3, 103.6, 86.1, 28.2; LRMS (ESI +) m / z calcd for C 14 H 16 N 2 O 3 [M + Na] +: 283.11; Found: 283.10
제조예Manufacturing example 12: 5-(티오펜-2-일)-1H-인돌-3- 12: 5- (thiophen-2-yl) -1H-indole-3- 카복시알데하이드Carboxyaldehyde
20 ml 용량의 바이알(vial)에 5-브로모-1H-인돌-3-카복시알데하이드(224 mg), Pd(OAc)2(2 mol%, 4.5 mg), 티오펜-2-일보론산(1.5 당량, 192 mg), 포스핀 리간드로서 2-디사이클로헥실포스피노-2',6'-디메톡시비페닐(SPhos, 4 몰%, 16 mg) 및 무수 Na2CO3(3 당량, 318 mg)를 충진하고, 고무 격막으로 마개를 씌워두었다. 이후, 공기를 제거한 다음 아르곤으로 재충진시켰다.In a 20 ml vial, 5-bromo-1H-indole-3-carboxyaldehyde (224 mg), Pd (OAc) 2 (2 mol%, 4.5 mg), thiophen-2-ylboronic acid (1.5 Equivalent, 192 mg), 2-dicyclohexylphosphino-2 ', 6'-dimethoxybiphenyl (SPhos, 4 mole%, 16 mg) as an phosphine ligand and anhydrous Na 2 CO 3 (3 equivalents, 318 mg ) Was filled with a rubber diaphragm. The air was then removed and refilled with argon.
시린지를 이용하여 격막을 통해 n-부탄올을 첨가한 다음 격막을 테플론 스크루캡(Teflon Screwcap)으로 교체하였다. 반응 혼합물을 100℃까지 밤새 가열한 다 음 냉각하고 고진공상태에서 농축시켜 용매를 제거하였다.A syringe was used to add n-butanol through the diaphragm and then the diaphragm was replaced with a Teflon Screwcap. The reaction mixture was heated to 100 ° C. overnight, then cooled and concentrated in high vacuum to remove solvent.
조 산물(crude mixture)을 에틸아세테이트로 희석하고 셀라이트로 여과한 다음 아세톤과 메탄올을 이용하여 재결정하여 표제 화합물(137 mg, 수율: 60%)을 수득하였다.The crude mixture was diluted with ethyl acetate, filtered through celite and recrystallized with acetone and methanol to give the title compound (137 mg, yield: 60%).
1H NMR (500 MHz, DMSO-d6) δ ppm 12.24 (s, 1 H) 9.96 (s, 1 H) 8.36 (s, 1 H) 8.32 (s, 1 H) 7.53-7.65 (m, 2 H) 7.48 (d, J=4.89 Hz, 1 H) 7.44 (d, J=3.42 Hz, 1 H) 7.12 (t, J=4.16 Hz, 1 H); 13C NMR (125 MHz, DMSO-d6) δ ppm 185.2, 185.1, 144.5, 139.3, 136.6, 128.5, 128.5, 128.3, 124.8, 124.8, 124.8, 122.9, 122.8, 121.8, 118.3, 117.6, 117.5, 113.1; LRMS (MALDI) m/z calcd for C13H9NOS [M+H]+: 228.04; Found: 228.39. 1 H NMR (500 MHz, DMSO-d 6 ) δ ppm 12.24 (s, 1 H) 9.96 (s, 1 H) 8.36 (s, 1 H) 8.32 (s, 1 H) 7.53-7.65 (m, 2 H ) 7.48 (d, J = 4.89 Hz, 1 H) 7.44 (d, J = 3.42 Hz, 1 H) 7.12 (t, J = 4.16 Hz, 1 H); 13 C NMR (125 MHz, DMSO-d 6 ) δ ppm 185.2, 185.1, 144.5, 139.3, 136.6, 128.5, 128.5, 128.3, 124.8, 124.8, 124.8, 122.9, 122.8, 121.8, 118.3, 117.6, 117.5, 113.1; LRMS (MALDI) m / z calcd for C 13 H 9 NOS [M + H] < + >:228.04; Found: 228.39.
제조예Manufacturing example 13: 1- 13: 1- 메틸methyl -1H-인돌-3--1H-indole-3- 카복시알데하이드Carboxyaldehyde
인돌-3-카복시알데하이드(58 mg)를 NaH(1.2 당량, 12 mg, 미네랄 오일내에 60% 분산액)를 함유한 무수 DMF 용액으로 0℃에서 10분간 처리한 다음, 요오드화메틸 (1.2 당량, 30 ㎕)을 첨가하고 실온에서 1시간동안 교반하였다. Indole-3-carboxyaldehyde (58 mg) was treated with anhydrous DMF solution containing NaH (1.2 equivalents, 12 mg, 60% dispersion in mineral oil) at 0 ° C. for 10 minutes, followed by methyl iodide (1.2 equivalents, 30 μl). ) Was added and stirred at room temperature for 1 hour.
반응 용액을 H2O를 이용하여 식힌 다음 30분간 추가 교반하고, Na2S2O4의 존재하에 에틸아세테이트로 여과하여 요오드화메틸을 냉각시켰다. The reaction solution was cooled using H 2 O and then further stirred for 30 minutes, and filtered through ethyl acetate in the presence of Na 2 S 2 O 4 to cool methyl iodide.
유기층을 무수 Na2SO4 상에서 건조하고 감압하에 농축한 다음 실리카 겔상에서 플래시 컬럼 크로마토그래피로 정제하여 표제 화합물 (46 mg, 수율: 73%)을 수득하였다.The organic layer was dried over anhydrous Na 2 SO 4 , concentrated under reduced pressure and purified by flash column chromatography on silica gel to give the title compound (46 mg, yield: 73%).
1H NMR (500 MHz, acetone) δ ppm 9.96 (s, 1 H) 8.23 (d, J=7.83 Hz, 1 H) 8.03 (s, 1 H) 7.50 (d, J=8.07 Hz, 1 H) 7.32 (t, J=6.97 Hz, 1 H) 7.27 (t, J=6.97 Hz, 1 H) 3.92 (s, 3 H); 13C NMR (125 MHz, acetone) δ ppm 184.7, 141.5, 139.2, 126.2, 124.5, 123.3, 122.4, 118.9, 111.4, 33.9; LRMS (MALDI) m/z calcd for C10H9NO [M+H]+: 160.07; Found: 160.45. 1 H NMR (500 MHz, acetone) δ ppm 9.96 (s, 1 H) 8.23 (d, J = 7.83 Hz, 1 H) 8.03 (s, 1 H) 7.50 (d, J = 8.07 Hz, 1 H) 7.32 (t, J = 6.97 Hz, 1 H) 7.27 (t, J = 6.97 Hz, 1 H) 3.92 (s, 3H); 13 C NMR (125 MHz, acetone) δ ppm 184.7, 141.5, 139.2, 126.2, 124.5, 123.3, 122.4, 118.9, 111.4, 33.9; LRMS (MALDI) m / z calcd for C 10 H 9 NO [M + H] < + >:160.07; Found: 160.45.
제조예Manufacturing example 14: 1-벤질-1H-인돌-3- 14: 1-benzyl-1H-indole-3- 카복시알데하이드Carboxyaldehyde
출발물질로서 인돌-3-카복시알데하이드(145 mg)을 사용하고 요오드화메틸 대신 브롬화벤질(171 mg)을 사용하는 것을 제외하고는 제조예 13의 과정과 동일한 방법으로 표제 화합물(216 mg, 수율: 92%)을 수득하였다.The title compound (216 mg, yield: 92) was obtained in the same manner as in Example 13, except that indole-3-carboxyaldehyde (145 mg) was used as the starting material and benzyl bromide (171 mg) was used instead of methyl iodide. %) Was obtained.
1H NMR (500 MHz, acetone) δ ppm 10.02 (s, 1 H) 8.25 (d, J=6.36 Hz, 1 H) 8.24 (s, 1 H) 7.50-7.53 (m, 1 H) 7.23-7.37 (m, 7 H) 5.56 (s, 2 H); 13C NMR (125 MHz, acetone) δ ppm 185.0 , 140.9, 138.5, 137.7, 129.7, 128.8, 128.2, 126.4, 124.6, 123.4, 122.6, 119.3, 111.9, 51.2; LRMS (MALDI) m/z calcd for C16H13NO [M+H]+: 236.10; Found: 236.43. 1 H NMR (500 MHz, acetone) δ ppm 10.02 (s, 1 H) 8.25 (d, J = 6.36 Hz, 1 H) 8.24 (s, 1 H) 7.50-7.53 (m, 1 H) 7.23-7.37 ( m, 7 H) 5.56 (s, 2H); 13 C NMR (125 MHz, acetone) δ ppm 185.0, 140.9, 138.5, 137.7, 129.7, 128.8, 128.2, 126.4, 124.6, 123.4, 122.6, 119.3, 111.9, 51.2; LRMS (MALDI) m / z calcd for C 16 H 13 NO [M + H] < + >:236.10; Found: 236.43.
제조예Manufacturing example 15: N-벤질-3- 15: N-benzyl-3- 포밀Formyl -1H-인돌-1--1H-indole-1- 카복시아마이드Carboxyxamide
인돌-3-카복시알데하이드와 DIPEA(1.0 당량, 591 ㎕)을 함유한 DCM 용액에, 이소시아나토벤젠(BnNCO, 1.5 당량, 417 ㎕)을 첨가한 다음 실온에서 3시간 동안 교반하였다.To a DCM solution containing indole-3-carboxyaldehyde and DIPEA (1.0 equiv, 591 μl), isocyanatobenzene (BnNCO, 1.5 equiv., 417 μl) was added and stirred at room temperature for 3 hours.
반응이 종결된 후, 반응 용액을 디클로로메탄(DCM)으로 희석하고 포화 시트르산(aq)으로 세척하였다. 모아진 유기층을 무수 Na2SO4 상에서 건조한 다음 여과하였다. 여액을 감압하에 농축하고 DCM과 헥산으로 재결정화하여 백색 고체상의 표제 화합물 (739 mg, 수율: 78%)을 수득하였다.After the reaction was complete, the reaction solution was diluted with dichloromethane (DCM) and washed with saturated citric acid (aq). The combined organic layers were dried over anhydrous Na 2 SO 4 and filtered. The filtrate was concentrated under reduced pressure and recrystallized from DCM and hexanes to give the title compound (739 mg, yield: 78%) as a white solid.
1H NMR (500 MHz, CHLOROFORM-d) δ ppm 10.06 (s, 1 H) 8.31 (d, J=7.34 Hz, 1 H) 8.17 (s, 1 H) 7.92 (d, J=7.58 Hz, 1 H) 7.33-7.45 (m, 7 H) 6.14 (br.s., 1 H) 4.71 (d, J=5.62 Hz, 2 H); 13C NMR (125 MHz, acetone) δ ppm 185.9, 151.2, 137.2, 135.8, 135.4, 129.1, 128.2, 128.1, 126.0, 124.6, 122.3, 121.0, 113.9, 45.4, 45.3; LRMS (MALDI) m/z calcd for C17H14N2O2 [M+H]+: 279.11; Found: 279.41. 1 H NMR (500 MHz, CHLOROFORM-d) δ ppm 10.06 (s, 1 H) 8.31 (d, J = 7.34 Hz, 1 H) 8.17 (s, 1 H) 7.92 (d, J = 7.58 Hz, 1 H ) 7.33-7.45 (m, 7H) 6.14 (br.s., 1H) 4.71 (d, J = 5.62 Hz, 2H); 13 C NMR (125 MHz, acetone) δ ppm 185.9, 151.2, 137.2, 135.8, 135.4, 129.1, 128.2, 128.1, 126.0, 124.6, 122.3, 121.0, 113.9, 45.4, 45.3; LRMS (MALDI) m / z calcd for C 17 H 14 N 2 O 2 [M + H] < + >:279.11; Found: 279.41.
실시예Example 1: 2-(3- 1: 2- (3- 페닐Phenyl -1H--1H- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
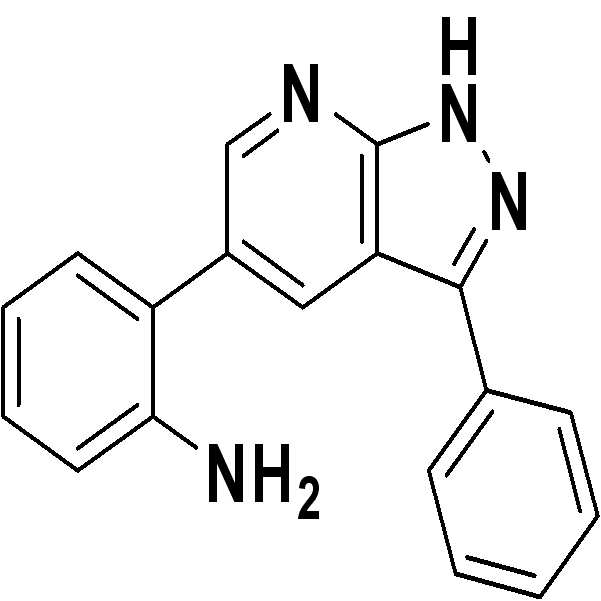
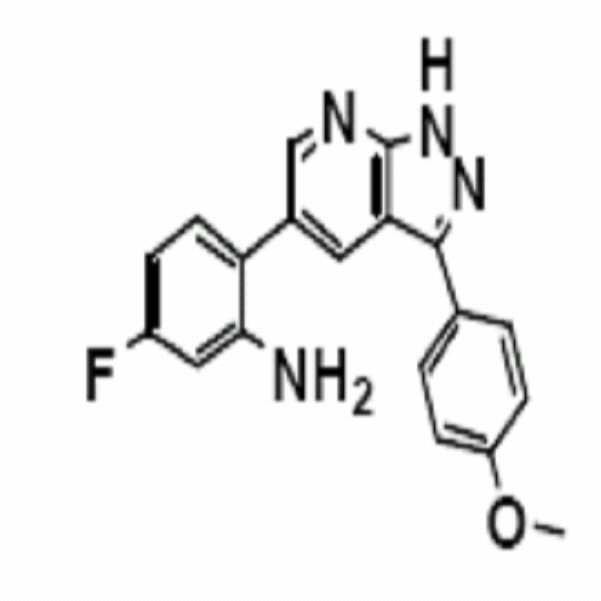
인돌-3-카복시알데하이드(29 mg)와 3-아미노-5-페닐피라졸(1.0 당량, 32 mg)을 함유한 MeOH(0.05M) 용액에, 알루미늄 트리클로라이드(0.10 당량, 2.7 mg)을 첨가한 다음, 반응 혼합물을 70℃로 설정된 20 ml 용량의 바이알에서 환류(reflux) 조건하에 4시간 동안 가열하였다. To a solution of MeOH (0.05M) containing indole-3-carboxyaldehyde (29 mg) and 3-amino-5-phenylpyrazole (1.0 equiv, 32 mg), aluminum trichloride (0.10 equiv, 2.7 mg) was added. The reaction mixture was then heated in a 20 ml volume vial set at 70 ° C. for 4 hours under reflux conditions.
반응이 종결된 후, 반응 용액을 실온으로 냉각하고 얻어진 고형물을 서서히 여과한 다음 냉 메탄올과 디에틸에테르로 세척하였다. 여액을 실리카 겔상에서 플래시 컬럼 크로마토그래피(용리액: 에틸아세테이트/헥산/메탄올을 함유한 1% TEA)에 의해 정제하여 원하는 표제 화합물(34 mg, 수율: 58%)을 수득하였다.After the reaction was completed, the reaction solution was cooled to room temperature and the solid obtained was slowly filtered and washed with cold methanol and diethyl ether. The filtrate was purified by flash column chromatography on silica gel (eluent: 1% TEA with ethyl acetate / hexanes / methanol) to afford the desired title compound (34 mg, yield: 58%).
1H NMR (500 MHz, acetone) δ ppm 12.90 (br. s., 1 H) 8.60 (d, J = 1.96 Hz, 1 H) 8.53 (d, J = 1.96 Hz, 1 H) 8.12 (dd, J = 8.56, 1.22 Hz, 2 H) 7.54 (t, J = 7.70 Hz, 2 H) 7.41-7.45 (m, 1 H) 7.13-7.17 (m, 2 H) 6.89 (dd, J = 8.44, 1.10 Hz, 1 H) 6.76 (td, J = 7.46, 1.22 Hz, 1 H) 4.58-4.64 (m, 2 H); 13C NMR (125 MHz, acetone) δ ppm 151.1, 146.8, 134.8, 131.9, 130.9, 130.5, 129.9, 129.8, 129.1, 127.9, 124.8, 118.5, 116.6, 116.5, 113.5; HRMS (EI+) m/z calcd for C18H14N4 [M]+: 286.1218; Found: 286.1216 1 H NMR (500 MHz, acetone) δ ppm 12.90 (br.s., 1 H) 8.60 (d, J = 1.96 Hz, 1 H) 8.53 (d, J = 1.96 Hz, 1 H) 8.12 (dd, J = 8.56, 1.22 Hz, 2H) 7.54 (t, J = 7.70 Hz, 2H) 7.41-7.45 (m, 1H) 7.13-7.17 (m, 2H) 6.89 (dd, J = 8.44, 1.10 Hz, 1 H) 6.76 (td, J = 7.46, 1.22 Hz, 1 H) 4.58-4.64 (m, 2H); 13 C NMR (125 MHz, acetone) δ ppm 151.1, 146.8, 134.8, 131.9, 130.9, 130.5, 129.9, 129.8, 129.1, 127.9, 124.8, 118.5, 116.6, 116.5, 113.5; HRMS (EI +) m / z calcd for C 18 H 14 N 4 [M] < + >:286.1218; Found: 286.1216
실시예Example 2: 2-(1H- 2: 2- (1H- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
출발물질로서 인돌-3-카복시알데하이드(15 mg)를 사용하고 3-아미노-5-페닐피라졸 대신에 3-아미노피라졸(8.3 mg)을 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(15 mg, 수율: 71%)을 수득하였다.The same procedure as in Example 1 was used except that indole-3-carboxyaldehyde (15 mg) was used as starting material and 3-aminopyrazole (8.3 mg) instead of 3-amino-5-phenylpyrazole. The desired compound was obtained (15 mg, yield 71%).
1H NMR (500 MHz, acetone) δ ppm 12.76 (br. s., 1 H) 8.55 (d, J = 1.96 Hz, 1 H) 8.23 (d, J = 1.96 Hz, 1 H) 8.13 (s, 1 H) 7.13 (td, J = 7.64, 1.59 Hz, 1 H) 7.10 (dd, J = 7.58, 1.47 Hz, 1 H) 6.87 (dd, J = 8.07, 0.98 Hz, 1 H) 6.74 (td, J = 7.46, 1.22 Hz, 1 H) 4.56 (br. s., 2 H); 13C NMR (125 MHz, acetone) δ ppm 150.9, 146.7, 134.5, 132.0, 131.8, 130.4, 130.0, 129.7, 118.5, 116.5, 115.8; HRMS (EI+) m/z calcd for C12H10N4 [M]+: 210.0905; Found:210.0908. 1 H NMR (500 MHz, acetone) δ ppm 12.76 (br.s., 1 H) 8.55 (d, J = 1.96 Hz, 1 H) 8.23 (d, J = 1.96 Hz, 1 H) 8.13 (s, 1 H) 7.13 (td, J = 7.64, 1.59 Hz, 1 H) 7.10 (dd, J = 7.58, 1.47 Hz, 1 H) 6.87 (dd, J = 8.07, 0.98 Hz, 1 H) 6.74 (td, J = 7.46, 1.22 Hz, 1 H) 4.56 (br. S., 2H); 13 C NMR (125 MHz, acetone) δ ppm 150.9, 146.7, 134.5, 132.0, 131.8, 130.4, 130.0, 129.7, 118.5, 116.5, 115.8; HRMS (EI +) m / z calcd for C 12 H 10 N 4 [M] < + >:210.0905; Found: 210.0908.
실시예Example 3: 2-(3- 3: 2- (3- 메틸methyl -1H--1H- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
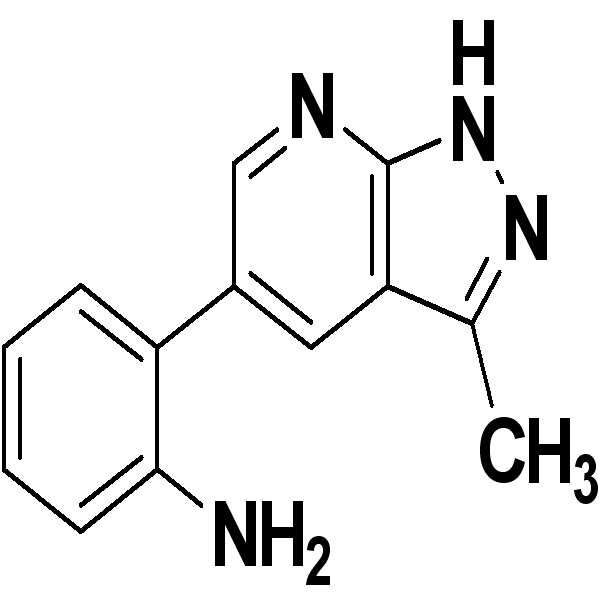
출발물질로서 3-아미노-5-페닐피라졸 대신에 3-아미노-5-메틸피라졸(19 mg)을 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(35 mg, 수율: 79%)을 수득하였다.Except for using 3-amino-5-methylpyrazole (19 mg) instead of 3-amino-5-phenylpyrazole as starting material, the same procedure as in Example 1 was carried out to give the desired title compound (35 mg, Yield: 79%).
1H NMR (500 MHz, acetone) δ ppm 12.34 (br. s., 1 H) 8.51 (d, J = 2.20 Hz, 1 H) 8.15 (d, J = 1.96 Hz, 1 H) 7.11-7.15 (m, 1 H) 7.09 (dd, J = 7.46, 1.59 Hz, 1 H) 6.87 (dd, J = 8.07, 0.98 Hz, 1 H) 6.74 (td, J = 7.40, 1.10 Hz, 1 H) 4.56 (br. s., 2 H) 2.55 (s, 3 H); 13C NMR (125 MHz, acetone) δ ppm 150.6, 146.7, 131.8, 129.8, 129.6, 129.1, 124.9, 118.4, 116.4, 115.3, 12.6; HRMS (EI+) m/z calcd for C13H12N4 [M+H]+: 224.1062; Found: 224.1061. 1 H NMR (500 MHz, acetone) δ ppm 12.34 (br. S., 1 H) 8.51 (d, J = 2.20 Hz, 1 H) 8.15 (d, J = 1.96 Hz, 1 H) 7.11-7.15 (m , 1 H) 7.09 (dd, J = 7.46, 1.59 Hz, 1 H) 6.87 (dd, J = 8.07, 0.98 Hz, 1 H) 6.74 (td, J = 7.40, 1.10 Hz, 1 H) 4.56 (br. s., 2H) 2.55 (s, 3H); 13 C NMR (125 MHz, acetone) δ ppm 150.6, 146.7, 131.8, 129.8, 129.6, 129.1, 124.9, 118.4, 116.4, 115.3, 12.6; HRMS (EI +) m / z calcd for C 13 H 12 N 4 [M + H] < + >:224.1062; Found: 224.1061.
실시예Example 4: 5-(2- 4: 5- (2- 아미노페닐Aminophenyl )-) - 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -3-올-3-ol
출발물질로서 3-아미노-5-페닐피라졸 대신에 3-아미노-5-히드록시피라졸(20 mg)을 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(28 mg, 수율: 62%)을 수득하였다.The same procedure as in Example 1 was carried out except that 3-amino-5-hydroxypyrazole (20 mg) was used instead of 3-amino-5-phenylpyrazole as the starting material (28 mg). , Yield: 62%).
1H NMR (500 MHz, DMSO-d6) δ ppm 12.23 (br. s., 1 H) 10.06 (s, 1 H) 9.66 (s, 1 H) 8.08 (s, 1 H) 8.05 (dd, J = 6.24, 2.81 Hz, 1 H) 7.54 (dd, J = 5.38, 3.67 Hz, 1 H)7.23-7.28 (m, 2H) 5.72 (s, 2 H); 13C NMR (125 MHz, DMSO-d6) δ ppm 164.9, 152.7, 136.1, 136.1, 133.1, 133.1, 128.3, 122.9, 121.4, 118.3, 114.4, 112.7, 111.2; HRMS (EI+) m/z calcd for C12H10N4O [M]+: 226.0854; Found: 226.0855. 1 H NMR (500 MHz, DMSO-d6) δ ppm 12.23 (br. S., 1 H) 10.06 (s, 1 H) 9.66 (s, 1 H) 8.08 (s, 1 H) 8.05 (dd, J = 6.24, 2.81 Hz, 1H) 7.54 (dd, J = 5.38, 3.67 Hz, 1H) 7.33-7.28 (m, 2H) 5.72 (s, 2H); 13 C NMR (125 MHz, DMSO-d 6 ) δ ppm 164.9, 152.7, 136.1, 136.1, 133.1, 133.1, 128.3, 122.9, 121.4, 118.3, 114.4, 112.7, 111.2; HRMS (EI +) m / z calcd for C 12 H 10 N 4 O [M] < + >:226.0854; Found: 226.0855.
실시예Example 5: 2-(3-(4- 5: 2- (3- (4- 메톡시페닐Methoxyphenyl )-) - 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
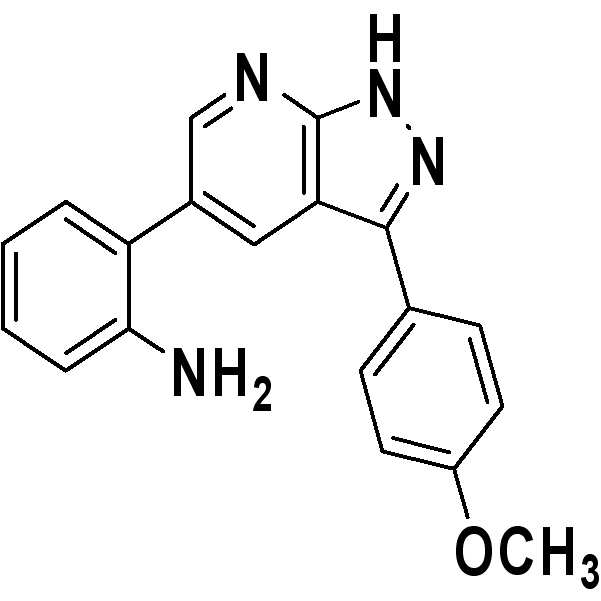
출발물질로서 3-아미노-5-페닐피라졸 대신에 3-아미노-5-(4-메톡시페닐)피라 졸(38 mg)을 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(50 mg, 수율: 78%)을 수득하였다.The same procedure as in Example 1 was carried out except that 3-amino-5- (4-methoxyphenyl) pyrazole (38 mg) was used instead of 3-amino-5-phenylpyrazole as a starting material. The title compound (50 mg, yield: 78%) was obtained.
1H NMR (500 MHz, acetone) δ ppm 12.77 (br. s., 1 H) 8.57 (d, J = 1.47 Hz, 1 H) 8.49 (d, J = 1.71 Hz, 1 H) 8.05 (d, J = 8.80 Hz, 2 H) 7.13-7.17 (m, 2 H) 7.10 (d, J = 9.05 Hz, 2 H) 6.89 (d, J = 8.31 Hz, 1 H) 6.76 (t, J = 7.46 Hz, 1 H) 4.58-4.65 (m, 2 H) 3.87 (s, 3 H); 13C NMR (125 MHz, DMSO-d6) δ ppm 159.2, 149.5, 145.8, 130.8, 129.6, 128.7, 128.5, 127.9, 125.7, 122.9, 116.7, 115.2, 114.3, 114.3, 111.8, 55.1, 55.1; HRMS (EI+) m/z calcd for C19H16N4O [M]+: 316.1324; Found: 316.1323. 1 H NMR (500 MHz, acetone) δ ppm 12.77 (br.s., 1 H) 8.57 (d, J = 1.47 Hz, 1 H) 8.49 (d, J = 1.71 Hz, 1 H) 8.05 (d, J = 8.80 Hz, 2H) 7.13-7.17 (m, 2H) 7.10 (d, J = 9.05 Hz, 2H) 6.89 (d, J = 8.31 Hz, 1H) 6.76 (t, J = 7.46 Hz, 1 H) 4.58-4.65 (m, 2H) 3.87 (s, 3H); 13 C NMR (125 MHz, DMSO-d 6 ) δ ppm 159.2, 149.5, 145.8, 130.8, 129.6, 128.7, 128.5, 127.9, 125.7, 122.9, 116.7, 115.2, 114.3, 114.3, 111.8, 55.1, 55.1; HRMS (EI +) m / z calcd for C 19 H 16 N 4 O [M] < + >:316.1324; Found: 316.1323.
실시예Example 6: 2-(3-(4- 6: 2- (3- (4- 플루오로페닐Fluorophenyl )-) - 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
출발물질로서 3-아미노-5-페닐피라졸 대신에 5-아미노-3-(4-플루오로페닐)피라졸(35 mg)을 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(42 mg, 수율: 69%)을 수득하였다.The same procedure as in Example 1 was carried out except that 5-amino-3- (4-fluorophenyl) pyrazole (35 mg) was used instead of 3-amino-5-phenylpyrazole as a starting material. The title compound (42 mg, yield: 69%) was obtained.
1H NMR (500 MHz, acetone) δ ppm 12.93 (br. s., 1 H) 8.60 (d, J = 1.96 Hz, 1 H) 8.51 (d, J = 1.96 Hz, 1 H) 8.15-8.19 (m, 2 H) 7.28-7.34 (m, 2 H) 7.13-7.17 (m, 2 H) 6.89 (dd, J = 8.44, 1.10 Hz, 1 H) 6.76 (td, J = 7.46, 1.22 Hz, 1 H) 4.59-4.66 (m, 2 H); 13C NMR (125 MHz, acetone) δ ppm 164.7, 162.7, 151.1, 146.8,1 31.9, 131.2, 130.7, 130.6, 129.8, 129.8, 129.8, 124.6, 118.4, 116.7, 116.6, 116.5, 113.3; HRMS (EI+) m/z calcd for C18H13FN4 [M]+: 304.1124; Found: 304.1123. 1 H NMR (500 MHz, acetone) δ ppm 12.93 (br.s., 1 H) 8.60 (d, J = 1.96 Hz, 1 H) 8.51 (d, J = 1.96 Hz, 1 H) 8.15-8.19 (m , 2H) 7.28-7.34 (m, 2H) 7.13-7.17 (m, 2H) 6.89 (dd, J = 8.44, 1.10 Hz, 1H) 6.76 (td, J = 7.46, 1.22 Hz, 1H) 4.59-4.66 (m, 2H); 13 C NMR (125 MHz, acetone) δ ppm 164.7, 162.7, 151.1, 146.8,1 31.9, 131.2, 130.7, 130.6, 129.8, 129.8, 129.8, 124.6, 118.4, 116.7, 116.6, 116.5, 113.3; HRMS (EI +) m / z calcd for C 18 H 13 FN 4 [M] < + >:304.1124; Found: 304.1123.
실시예Example 7: 2-(3-(푸란-2-일)- 7: 2- (3- (furan-2-yl)- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
출발물질로서 인돌-3-카복시알데하이드(15 mg)를 사용하고 3-아미노-5-페닐피라졸 대신에 3-아미노-5-(2-푸릴)피라졸(15 mg)을 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(20 mg, 수율: 72%)을 수득하였다.Except using indole-3-carboxyaldehyde (15 mg) as starting material and 3-amino-5- (2-furyl) pyrazole (15 mg) instead of 3-amino-5-phenylpyrazole Was subjected to the same procedure as in Example 1 to obtain the desired title compound (20 mg, yield: 72%).
1H NMR (500 MHz, acetone) δ ppm 12.93 (br. s., 1 H) 8.60 (d, J = 2.20 Hz, 1 H) 8.52 (d, J = 1.96 Hz, 1 H) 7.76 (dd, J = 1.71, 0.73 Hz, 1 H) 7.13-7.18 (m, 2 H) 7.05 (dd, J = 3.18, 0.73 Hz, 1 H) 6.89 (d, J = 7.83 Hz, 1 H) 6.77 (td, J = 7.46, 1.22 Hz, 1 H) 6.65 (dd, J = 3.42, 1.96 Hz, 1 H) 4.63 (br. s., 2 H); 13C NMR (125 MHz, acetone) δ ppm 152.0, 151.5, 150.1, 146.8, 143.7, 131.8, 130.9, 130.7, 129.8, 124.5, 118.4, 116.5, 112.9, 112.4, 107.8; HRMS (EI+) m/z calcd for C16H12N4O [M]+: 276.1011; Found: 276.1014. 1 H NMR (500 MHz, acetone) δ ppm 12.93 (br. S., 1 H) 8.60 (d, J = 2.20 Hz, 1 H) 8.52 (d, J = 1.96 Hz, 1 H) 7.76 (dd, J = 1.71, 0.73 Hz, 1 H) 7.13-7.18 (m, 2 H) 7.05 (dd, J = 3.18, 0.73 Hz, 1 H) 6.89 (d, J = 7.83 Hz, 1 H) 6.77 (td, J = 7.46, 1.22 Hz, 1 H) 6.65 (dd, J = 3.42, 1.96 Hz, 1 H) 4.63 (br. S., 2 H); 13 C NMR (125 MHz, acetone) δ ppm 152.0, 151.5, 150.1, 146.8, 143.7, 131.8, 130.9, 130.7, 129.8, 124.5, 118.4, 116.5, 112.9, 112.4, 107.8; HRMS (EI +) m / z calcd for C 16 H 12 N 4 O [M] < + >:276.1011; Found: 276.1014.
실시예Example 8: 2-(3-(티오펜-2-일)- 8: 2- (3- (thiophen-2-yl)- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
출발물질로서 인돌-3-카복시알데하이드(15 mg)를 이용하고 3-아미노-5-페닐피라졸 대신에 5-아미노-3-(2-티에닐)피라졸(17 mg)을 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(19 mg, 수율: 65%)을 수득하였다.Except using indole-3-carboxyaldehyde (15 mg) as starting material and 5-amino-3- (2-thienyl) pyrazole (17 mg) instead of 3-amino-5-phenylpyrazole And the same procedure as in Example 1 to give the title compound (19 mg, yield: 65%).
1H NMR (500 MHz, acetone) δ ppm 12.87 (br. s., 1 H) 8.60 (d, J = 1.96 Hz, 1 H) 8.52 (d, J = 1.96 Hz, 1 H) 7.85 (d, J = 3.67 Hz, 1 H) 7.53 (dd, J = 5.01, 1.10 Hz, 1 H) 7.21 (dd, J = 5.01, 3.55 Hz, 1 H) 7.13-7.18 (m, 2 H) 6.89 (d, J = 8.31 Hz, 1 H) 6.77 (t, J = 7.34 Hz, 1 H) 4.65 (br. s., 2 H); 13C NMR (125 MHz, acetone) δ ppm 151.3, 146.7, 137.0, 131.8, 130.6, 130.3, 129.7, 128.7, 126.2, 125.9, 124.4, 118.3, 118.3, 116.4, 116.4, 112.8; HRMS (EI+) m/z calcd for C16H12N4S [M]+: 292.0782; Found: 292.0785 1 H NMR (500 MHz, acetone) δ ppm 12.87 (br. S., 1 H) 8.60 (d, J = 1.96 Hz, 1 H) 8.52 (d, J = 1.96 Hz, 1 H) 7.85 (d, J = 3.67 Hz, 1 H) 7.53 (dd, J = 5.01, 1.10 Hz, 1 H) 7.21 (dd, J = 5.01, 3.55 Hz, 1 H) 7.13-7.18 (m, 2H) 6.89 (d, J = 8.31 Hz, 1 H) 6.77 (t, J = 7.34 Hz, 1 H) 4.65 (br. S., 2 H); 13 C NMR (125 MHz, acetone) δ ppm 151.3, 146.7, 137.0, 131.8, 130.6, 130.3, 129.7, 128.7, 126.2, 125.9, 124.4, 118.3, 118.3, 116.4, 116.4, 112.8; HRMS (EI +) m / z calcd for C 16 H 12 N 4 S [M] < + >:292.0782; Found: 292.0785
실시예Example 9: 3-니트로-2-(3- 9: 3-nitro-2- (3- 페닐Phenyl -- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
출발물질로서 3-아미노-5-페닐파라졸(16 mg)을 이용하고 인돌-3-카복시알데하이드 대신에 제조예 1에서 얻어진 4-니트로-1H-인돌-3-카복시알데하이드(19 mg)를 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(25 mg, 수율: 75%)을 수득하였다.3-amino-5-phenylparasol (16 mg) was used as a starting material, and 4-nitro- 1H -indole-3-carboxyaldehyde (19 mg) obtained in Preparation Example 1 was used instead of indole-3-carboxyaldehyde. The same procedure as in Example 1 was carried out except that the title compound (25 mg, yield: 75%) was obtained.
1H NMR (500 MHz, acetone) δ ppm 13.00 (br. s., 1 H) 8.47 (d, J = 1.96 Hz, 1 H) 8.43 (d, J = 1.96 Hz, 1 H) 8.11 (dd, J = 8.44, 1.34 Hz, 2 H) 7.53 (t, J = 7.70 Hz, 2 H) 7.40-7.45 (m, 1 H) 7.37 (t, J = 8.19 Hz, 1 H) 7.21 (dd, J = 7.95, 1.10 Hz, 1 H) 7.18 (dd, J = 8.19, 1.10 Hz, 1 H) 5.02-5.13 (m, 2 H); 13C NMR (125 MHz, acetone) δ ppm 152.6, 150.9, 149.8, 149.8, 134.5, 132.0, 130.3, 129.9, 129.2, 127.8, 124.7, 119.6, 119.5, 117.0, 113.7, 112.3; HRMS (EI+) m/z calcd for C18H13N5O2 [M]+: 331.1069; Found: 331.1068 1 H NMR (500 MHz, acetone) δ ppm 13.00 (br. S., 1 H) 8.47 (d, J = 1.96 Hz, 1 H) 8.43 (d, J = 1.96 Hz, 1 H) 8.11 (dd, J = 8.44, 1.34 Hz, 2H) 7.53 (t, J = 7.70 Hz, 2H) 7.40-7.45 (m, 1H) 7.37 (t, J = 8.19 Hz, 1H) 7.21 (dd, J = 7.95, 1.10 Hz, 1 H) 7.18 (dd, J = 8.19, 1.10 Hz, 1 H) 5.02-5.13 (m, 2H); 13 C NMR (125 MHz, acetone) δ ppm 152.6, 150.9, 149.8, 149.8, 134.5, 132.0, 130.3, 129.9, 129.2, 127.8, 124.7, 119.6, 119.5, 117.0, 113.7, 112.3; HRMS (EI +) m / z calcd for C 18 H 13 N 5 O 2 [M] < + >:331.1069; Found: 331.1068
실시예Example 10: 4'-( 10: 4 '-( 클로로메틸Chloromethyl )-2-(3-) -2- (3- 페닐Phenyl -- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)비페닐-3- 아민-5-yl) biphenyl-3-amine
출발물질로서 3-아미노-5-페닐파라졸(19 mg)을 이용하고 인돌-3-카복시알데하이드 대신에 제조예 8에서 제조된 4-(4-(클로로메틸)페닐)-1H-인돌-3-카복시알데하이드(30 mg)를 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(28 mg, 수율: 61%)을 수득하였다.4- (4- (chloromethyl) phenyl) -1H-indole-3 prepared in Preparation Example 8 using 3-amino-5-phenylparasol (19 mg) as a starting material and replacing indole-3-carboxyaldehyde. The same procedure as in Example 1 was carried out except for using carboxyaldehyde (30 mg) to give the desired title compound (28 mg, yield: 61%).
1H NMR (500 MHz, DMSO-d6) δ ppm 13.69 (s, 1 H) 8.27 (d, J = 1.96 Hz, 1 H) 8.05 (d, J = 1.96 Hz, 1 H) 7.92 (d, J = 7.09 Hz, 2 H) 7.50 (t, J = 7.58 Hz, 2 H) 7.40 (t, J = 7.34 Hz, 1 H) 7.14-7.21 (m, 3 H) 7.07 (d, J = 8.07 Hz, 2 H) 6.84 (dd, J = 8.07, 1.22 Hz, 1 H) 6.62 (dd, J = 7.46, 1.10 Hz, 1 H) 4.84 (br. s., 2 H) 4.60 (s, 2 H); 13C NMR (125 MHz, acetone) δ ppm 151.6, 151.1, 142.7, 141.8, 135.3, 133.1, 1312.0, 129.8, 128.9, 128.4, 128.2, 128.0, 126.6, 121.5, 118.3, 114.4, 111.9, 45.8; HRMS (EI+) m/z calcd for C25H19ClN4 [M]+: 410.1298 ; Found: 410.1297 1 H NMR (500 MHz, DMSO-d 6 ) δ ppm 13.69 (s, 1 H) 8.27 (d, J = 1.96 Hz, 1 H) 8.05 (d, J = 1.96 Hz, 1 H) 7.92 (d, J = 7.09 Hz, 2H) 7.50 (t, J = 7.58 Hz, 2H) 7.40 (t, J = 7.34 Hz, 1H) 7.14-7.21 (m, 3H) 7.07 (d, J = 8.07 Hz, 2 H) 6.84 (dd, J = 8.07, 1.22 Hz, 1 H) 6.62 (dd, J = 7.46, 1.10 Hz, 1 H) 4.84 (br. S., 2 H) 4.60 (s, 2 H); 13 C NMR (125 MHz, acetone) δ ppm 151.6, 151.1, 142.7, 141.8, 135.3, 133.1, 1312.0, 129.8, 128.9, 128.4, 128.2, 128.0, 126.6, 121.5, 118.3, 114.4, 111.9, 45.8; HRMS (EI +) m / z calcd for C 25 H 19 ClN 4 [M] < + >:410.1298; Found: 410.1297
실시예Example 11: 11: terttert -부틸 3-아미노-2-(3--Butyl 3-amino-2- (3- 페닐Phenyl -- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 페닐카 바메이트Phenyl Carbamate
출발물질로서 3-아미노-5-페닐파라졸(16 mg)을 이용하고 인돌-3-카복시알데하이드 대신에 제조예 11에서 제조된 tert-부틸 4-아미노-3-포밀-1H-인돌-1-카복실레이트(26 mg)를 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(17 mg, 수율: 43%)을 수득하였다. Tert -Butyl 4-amino-3-formyl- 1H -indole- 1 -prepared in Preparation Example 11 using 3-amino-5-phenylparasol (16 mg) as starting material and replacing indole-3-carboxyaldehyde. The same procedure as in Example 1 was carried out except for using carboxylate (26 mg) to afford the title compound (17 mg, yield: 43%).
1H NMR (500 MHz, acetone) δ ppm 12.96 (br. s., 1 H) 8.41 (s, 1 H) 8.39 (s, 1 H) 8.11 (d, J = 7.58 Hz, 2 H) 7.53 (t, J = 7.70 Hz, 2 H) 7.42 (t, J = 8.07 Hz, 1 H) 7.21 (d, J = 8.07 Hz, 1 H) 7.12 (t, J = 8.19 Hz, 1 H) 7.07 (br. s., 1 H) 6.67 (d, J = 8.07 Hz, 1 H) 4.39 (br. s., 2 H) 1.26 (s, 9 H); 13C NMR (125 MHz, acetone) δ ppm 154.0, 152.2, 148.0, 144.6, 138.6, 134.6, 133.4, 129.8, 129.7, 129.1, 127.8, 125.7, 117.4, 114.0, 112.6, 112.0, 79.6, 28.4; HRMS (EI+) m/z calcd for C23H23N5O2 [M]+: 401.1851 ; Found: 401.1851 1 H NMR (500 MHz, acetone) δ ppm 12.96 (br. S., 1 H) 8.41 (s, 1 H) 8.39 (s, 1 H) 8.11 (d, J = 7.58 Hz, 2 H) 7.53 (t , J = 7.70 Hz, 2 H) 7.42 (t, J = 8.07 Hz, 1 H) 7.21 (d, J = 8.07 Hz, 1 H) 7.12 (t, J = 8.19 Hz, 1 H) 7.07 (br. S ., 1 H) 6.67 (d, J = 8.07 Hz, 1 H) 4.39 (br. S., 2 H) 1.26 (s, 9 H); 13 C NMR (125 MHz, acetone) δ ppm 154.0, 152.2, 148.0, 144.6, 138.6, 134.6, 133.4, 129.8, 129.7, 129.1, 127.8, 125.7, 117.4, 114.0, 112.6, 112.0, 79.6, 28.4; HRMS (EI +) m / z calcd for C 23 H 23 N 5 O 2 [M] < + >:401.1851; Found: 401.1851
실시예Example 12: 12: 메틸methyl 3-아미노-2-(3- 3-amino-2- (3- 페닐Phenyl -- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤조에이트Benzoate
출발물질로서 인돌-3-카복시알데하이드 대신에 제조예 2에서 얻어진 메틸 3-포밀-1H-인돌-4-카복실레이트(41 mg)를 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(52 mg, 수율: 76%)을 수득하였다.The same procedure as in Example 1 was conducted except that methyl 3-formyl- 1H -indole-4-carboxylate (41 mg) obtained in Preparation Example 2 was used instead of indole-3-carboxyaldehyde as a starting material. The title compound (52 mg, yield: 76%) was obtained.
1H NMR (500 MHz, acetone) δ ppm 12.90 (br. s., 1 H) 8.35 (s, 2 H) 8.10 (dd, J = 8.31, 1.22 Hz, 2 H) 7.53 (t, J = 7.70 Hz, 2 H) 7.39-7.44 (m, 1 H) 7.26 (t, J = 7.83 Hz, 1 H) 7.19-7.23 (m, 1 H) 7.08 (dd, J = 7.95, 1.34 Hz, 1 H) 4.62-4.67 (m, 2 H); 13C NMR (125 MHz, acetone) δ ppm 168.8, 151.4, 148.4, 148.4, 134.8, 133.5, 131.4, 129.9, 129.5, 129.1, 128.5, 127.8, 124.1, 119.2, 119.2, 119.1, 113.5, 52.0; HRMS (EI+) m/z calcd for C20H16N4O2 [M]+: 344.1273; Found: 344.1275 1 H NMR (500 MHz, acetone) δ ppm 12.90 (br.s., 1 H) 8.35 (s, 2 H) 8.10 (dd, J = 8.31, 1.22 Hz, 2 H) 7.53 (t, J = 7.70 Hz , 2H) 7.39-7.44 (m, 1H) 7.26 (t, J = 7.83 Hz, 1H) 7.19-7.23 (m, 1H) 7.08 (dd, J = 7.95, 1.34 Hz, 1H) 4.62- 4.67 (m, 2H); 13 C NMR (125 MHz, acetone) δ ppm 168.8, 151.4, 148.4, 148.4, 134.8, 133.5, 131.4, 129.9, 129.5, 129.1, 128.5, 127.8, 124.1, 119.2, 119.2, 119.1, 113.5, 52.0; HRMS (EI +) m / z calcd for C 20 H 16 N 4 O 2 [M] +: 344.1273; Found: 344.1275
실시예Example 13: 2-(3- 13: 2- (3- 페닐Phenyl -- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-3-(피리딘-4-일)-5-yl) -3- (pyridin-4-yl) 벤젠아민Benzeneamine
출발물질로서 3-아미노-5-페닐파라졸(16 mg)을 이용하고 인돌-3-카복시알데하이드 대신에 제조예 9에서 제조된 4-(피리딘-4-일)-1H-인돌-3-카복시알데하이드(22 mg)를 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(26 mg, 수율: 71%)을 수득하였다.The use of 3-amino-5-phenyl-para-sol (16 mg) as a starting material and produced in Production Example 9 in place of indole-3-carboxy aldehyde 4- (pyridin-4-yl) - 1H - indole-3-carboxylic The same procedure as in Example 1 was carried out except for the use of aldehyde (22 mg) to give the desired title compound (26 mg, yield: 71%).
1H NMR (500 MHz, DMSO-d6) δ ppm 13.73 (s, 1 H) 8.31 (d, J = 1.47 Hz, 1 H) 8.29 (d, J = 5.87 Hz, 2 H) 8.08 (d, J = 1.96 Hz, 1 H) 7.95 (d, J = 7.34 Hz, 2 H) 7.50 (t, J = 7.83 Hz, 2 H) 7.41 (t, J = 7.58 Hz, 1 H) 7.22 (t, J = 7.83 Hz, 1 H) 7.07 (d, J = 5.62 Hz, 2 H) 6.89 (d, J = 8.07 Hz, 1 H) 6.64 (d, J = 7.09 Hz, 1 H) 4.93 (s, 2H); HRMS (EI+) m/z calcd for C23H17N5 [M+H]+: 363.1484 ; Found: 363.1489 1 H NMR (500 MHz, DMSO-d 6 ) δ ppm 13.73 (s, 1 H) 8.31 (d, J = 1.47 Hz, 1 H) 8.29 (d, J = 5.87 Hz, 2H) 8.08 (d, J = 1.96 Hz, 1 H) 7.95 (d, J = 7.34 Hz, 2 H) 7.50 (t, J = 7.83 Hz, 2 H) 7.41 (t, J = 7.58 Hz, 1 H) 7.22 (t, J = 7.83 Hz, 1H) 7.07 (d, J = 5.62 Hz, 2H) 6.89 (d, J = 8.07 Hz, 1H) 6.64 (d, J = 7.09 Hz, 1H) 4.93 (s, 2H); HRMS (EI +) m / z calcd for C 23 H 17 N 5 [M + H] < + >:363.1484; Found: 363.1489
실시예Example 14: 2',6'-디메틸-2-(3- 14: 2 ', 6'-dimethyl-2- (3- 페닐Phenyl -- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)비페닐-3-아민 -5-yl) biphenyl-3-amine
출발물질로서 3-아미노-5-페닐파라졸(13 mg)을 이용하고 인돌-3-카복시알데하이드 대신에 제조예 10에서 제조된 4-(2,6-디메틸페닐)-1H-인돌-3-카복시알데하이드(20 mg)를 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(19 mg, 수율: 61%)을 수득하였다.The use of 3-amino-5-phenyl-para-sol (13 mg) as a starting material and produced in Production Example 10 in place of indole-3-carboxy-4-aldehyde (2,6-dimethylphenyl) - 1H - indol-3 The same procedure as in Example 1 was carried out except for using carboxyaldehyde (20 mg) to give the desired title compound (19 mg, yield: 61%).
1H NMR (500 MHz, DMSO-d6) δ ppm 13.66 (br. s., 1 H) 8.23 (d, J = 1.96 Hz, 1 H) 7.96 (d, J = 2.20 Hz, 1 H) 7.75 (dd, J = 8.44, 1.34 Hz, 2 H) 7.52 (t, J = 7.70 Hz, 2 H) 7.39 - 7.44 (m, 1 H) 7.18 (dd, J = 8.07, 7.58 Hz, 1 H) 6.81 - 6.95 (m, 4 H) 6.39 (dd, J = 7.46, 1.10 Hz, 1 H) 4.84 (s, 2 H) 2.03 (br. s., 3 H) 1.92 (br. s., 3H); 13C NMR (125 MHz, DMSO-d6) δ ppm 151.65, 149.97, 146.82, 141.05, 140.6, 133.06, 130.44, 128.98, 128.73, 128.11, 126.61, 126.36, 121.43, 117.39, 114.18, 111.64, 20.42 ; HRMS (EI+) m/z calcd for C26H22N4 [M]+: 390.1844; Found: 390.1845 1 H NMR (500 MHz, DMSO-d 6 ) δ ppm 13.66 (br. S., 1 H) 8.23 (d, J = 1.96 Hz, 1 H) 7.96 (d, J = 2.20 Hz, 1 H) 7.75 ( dd, J = 8.44, 1.34 Hz, 2H) 7.52 (t, J = 7.70 Hz, 2H) 7.39-7.44 (m, 1H) 7.18 (dd, J = 8.07, 7.58 Hz, 1H) 6.81-6.95 (m, 4H) 6.39 (dd, J = 7.46, 1.10 Hz, 1H) 4.84 (s, 2H) 2.03 (br. s., 3H) 1.92 (br. s., 3H); 13 C NMR (125 MHz, DMSO-d 6 ) δ ppm 151.65, 149.97, 146.82, 141.05, 140.6, 133.06, 130.44, 128.98, 128.73, 128.11, 126.61, 126.36, 121.43, 117.39, 114.18, 111.64, 20.42; HRMS (EI +) m / z calcd for C 26 H 22 N 4 [M] < + >:390.1844; Found: 390.1845
실시예Example 15: 4-니트로-2-(3- 15: 4-nitro-2- (3- 페닐Phenyl -- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
출발물질로서 3-아미노-5-페닐파라졸(16 mg)을 이용하고 인돌-3-카복시알데하이드 대신에 제조예 3에서 제조된 5-니트로-1H-인돌-3-카복시알데하이드(19 mg)를 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(26 mg, 수율: 80%)을 수득하였다.Using 3-amino-5-phenylparazol (16 mg) as starting material and 5-nitro- 1H -indole-3-carboxyaldehyde (19 mg) prepared in Preparation Example 3 instead of indole-3-carboxyaldehyde. Except for using the same procedure as in Example 1 to give the desired title compound (26 mg, yield: 80%).
1H NMR (500 MHz, acetone) δ ppm 12.99 (br. s., 1 H) 8.64 (d, 1 H) 8.62 (d, 1 H) 8.14 (dd, J = 8.31, 0.98 Hz, 2 H) 8.07-8.11 (m, 2 H) 7.54 (t, J = 7.58 Hz, 2 H) 7.41-7.46 (m, 1 H) 6.99 (d, J = 9.29 Hz, 1 H) 6.04 (br. s., 2 H); 13C NMR (125 MHz, acetone) δ ppm 153.9, 150.8, 138.9, 134.6, 131.7, 129.9, 129.2, 128.4, 128.1, 127.9, 126.4, 115.0, 113.6; HRMS (EI+) m/z calcd for C18H13N5O2 [M]+: 331.1069; Found: 331.1073 1 H NMR (500 MHz, acetone) δ ppm 12.99 (br. S., 1 H) 8.64 (d, 1 H) 8.62 (d, 1 H) 8.14 (dd, J = 8.31, 0.98 Hz, 2 H) 8.07 -8.11 (m, 2H) 7.54 (t, J = 7.58 Hz, 2H) 7.41-7.46 (m, 1H) 6.99 (d, J = 9.29 Hz, 1H) 6.04 (br.s., 2H ); 13 C NMR (125 MHz, acetone) δ ppm 153.9, 150.8, 138.9, 134.6, 131.7, 129.9, 129.2, 128.4, 128.1, 127.9, 126.4, 115.0, 113.6; HRMS (EI +) m / z calcd for C 18 H 13 N 5 O 2 [M] < + >:331.1069; Found: 331.1073
실시예Example 16: 2-(3- 16: 2- (3- 페닐Phenyl -- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-4-(티오펜-2-일)-5-yl) -4- (thiophen-2-yl) 벤젠아민Benzeneamine
출발물질로서 인돌-3-카복시알데하이드 대신에 제조예 12에서 제조된 5-(티오펜-2-일)-1H-인돌-3-카복시알데하이드(45 mg)를 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(60 mg, 수율: 82%)을 수득하였다.As the starting material 5- (thiophen-2-yl) produced in Production Example 12 in place of indole-3-carboxy aldehyde - 1H - and are carried out but using indole-3-carboxy-aldehyde (45 mg) Example 1 The same procedure was followed to obtain the title compound (60 mg, yield: 82%).
1H NMR (500 MHz, acetone) δ ppm 12.96 (br. s., 1 H) 8.66 (d, J = 1.71 Hz, 1 H) 8.60 (d, J = 0.98 Hz, 1 H) 8.15 (dd, J = 8.44, 1.10 Hz, 2 H) 7.54 (t, J = 7.83 Hz, 2 H) 7.45-7.49 (m, 2 H) 7.40-7.45 (m, 1 H) 7.27-7.32 (m, 2 H) 7.03-7.07 (m, 1 H) 6.94 (d, J = 7.83 Hz, 1 H) 4.82-4.91 (m, 2 H); 13C NMR (125 MHz, acetone) δ ppm 151.05, 146.74, 145.82, 134.73, 131.15, 129.98, 129.86, 129.40, 129.11, 128.92, 127.88, 127.46, 125.09, 123.84, 122.14, 116.84, 113.55; HRMS (EI+) m/z calcd for C22H16N4S [M]+: 368.1096; Found: 368.1101 1 H NMR (500 MHz, acetone) δ ppm 12.96 (br. S., 1 H) 8.66 (d, J = 1.71 Hz, 1 H) 8.60 (d, J = 0.98 Hz, 1 H) 8.15 (dd, J = 8.44, 1.10 Hz, 2H) 7.54 (t, J = 7.83 Hz, 2H) 7.45-7.49 (m, 2H) 7.40-7.45 (m, 1H) 7.27-7.32 (m, 2H) 7.03- 7.07 (m, 1H) 6.94 (d, J = 7.83 Hz, 1H) 4.82-4.91 (m, 2H); 13 C NMR (125 MHz, acetone) δ ppm 151.05, 146.74, 145.82, 134.73, 131.15, 129.98, 129.86, 129.40, 129.11, 128.92, 127.88, 127.46, 125.09, 123.84, 122.14, 116.84, 113.55; HRMS (EI +) m / z calcd for C 22 H 16 N 4 S [M] < + >:368.1096; Found: 368.1101
실시예Example 17: 4- 17: 4- 메톡시Methoxy -2-(3--2- (3- 페닐Phenyl -- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
출발물질로서 인돌-3-카복시알데하이드 대신에 5-메톡시-1H-인돌-3-카복시알데하이드(35 mg)를 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(36 mg, 수율: 56%)을 수득하였다.The same procedure as in Example 1 was carried out except that 5-methoxy-1H-indole-3-carboxyaldehyde (35 mg) was used instead of indole-3-carboxyaldehyde as starting material to obtain the desired title compound (36 mg). , Yield: 56%) was obtained.
1H NMR (500 MHz, acetone) δ ppm 12.96 (br. s., 1 H) 8.64 (d, J = 1.96 Hz, 1 H) 8.56 (d, J = 1.96 Hz, 1 H) 8.12 (d, J = 7.09 Hz, 2 H) 7.53 (t, J = 7.70 Hz, 2 H) 7.42 (t, J = 7.46 Hz, 1 H) 6.78-6.87 (m, 3 H) 4.25 (br. s., 2 H) 3.75 (s, 3 H); 13C NMR (125 MHz, acetone) δ ppm 153.4, 151.0, 140.3, 134.7, 130.9, 130.5, 129.8, 129.1, 127.8, 125.7, 117.8, 117.0, 115.8, 113.4, 56.0; HRMS (EI+) m/z calcd for C19H16N4O [M]+: 316.1324; Found: 316.1326 1 H NMR (500 MHz, acetone) δ ppm 12.96 (br.s., 1 H) 8.64 (d, J = 1.96 Hz, 1 H) 8.56 (d, J = 1.96 Hz, 1 H) 8.12 (d, J = 7.09 Hz, 2H) 7.53 (t, J = 7.70 Hz, 2H) 7.42 (t, J = 7.46 Hz, 1H) 6.78-6.87 (m, 3H) 4.25 (br.s., 2H) 3.75 (s, 3H); 13 C NMR (125 MHz, acetone) δ ppm 153.4, 151.0, 140.3, 134.7, 130.9, 130.5, 129.8, 129.1, 127.8, 125.7, 117.8, 117.0, 115.8, 113.4, 56.0; HRMS (EI +) m / z calcd for C 19 H 16 N 4 O [M] < + >:316.1324; Found: 316.1326
실시예Example 18: 4- 18: 4- 클로로Chloro -2-(3--2- (3- 페닐Phenyl -- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
출발물질로서 3-아미노-5-페닐파라졸(16 mg)을 이용하고 인돌-3-카복시알데하이드 대신에 제조예 4에서 제조된 5-클로로-1H-인돌-3-카복시알데하이드(18 mg)를 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(22 mg, 수율: 68%)을 수득하였다.3-amino-5-phenylparazol (16 mg) was used as starting material and 5-chloro-1H-indole-3-carboxyaldehyde (18 mg) prepared in Preparation Example 4 was used instead of indole-3-carboxyaldehyde. Except for using the same procedure as in Example 1 to give the desired title compound (22 mg, yield: 68%).
1H NMR (500 MHz, DMSO-d6) δ ppm 13.88 (br. s., 1 H) 8.56 (d, J = 1.71 Hz, 1 H) 8.50 (d, J = 1.71 Hz, 1 H) 8.06 (d, J = 7.09 Hz, 2 H) 7.52 (t, J = 7.58 Hz, 2 H) 7.42 (t, J = 7.34 Hz, 1 H) 7.15 (d, J = 2.45 Hz, 1 H) 7.11-7.14 (m, 1 H) 6.78-6.82 (m, 1 H) 5.15 (br. s., 2 H); 13C NMR (125 MHz, DMSO-d6) δ ppm152.2, 150.2, 145.9, 143.6, 133.9, 130.8, 130.7, 129.7, 128.8, 128.5, 127.4, 125.1, 120.4, 117.3, 112.8; HRMS (EI+) m/z calcd for C18H13ClN4 [M]+: 320.0829; Found: 320.0827 1 H NMR (500 MHz, DMSO-d6) δ ppm 13.88 (br. S., 1 H) 8.56 (d, J = 1.71 Hz, 1 H) 8.50 (d, J = 1.71 Hz, 1 H) 8.06 (d , J = 7.09 Hz, 2H) 7.52 (t, J = 7.58 Hz, 2H) 7.42 (t, J = 7.34 Hz, 1H) 7.15 (d, J = 2.45 Hz, 1H) 7.11-7.14 (m , 1 H) 6.78-6.82 (m, 1 H) 5.15 (br. S., 2 H); 13 C NMR (125 MHz, DMSO-d 6 ) δ ppm152.2, 150.2, 145.9, 143.6, 133.9, 130.8, 130.7, 129.7, 128.8, 128.5, 127.4, 125.1, 120.4, 117.3, 112.8; HRMS (EI +) m / z calcd for C 18 H 13 ClN 4 [M] < + >:320.0829; Found: 320.0827
실시예Example 19: 4- 19: 4- 브로모Bromo -2-(3--2- (3- 페닐Phenyl -- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
출발물질로서 인돌-3-카복시알데하이드 대신에 5-브로모-1H-인돌-3-카복시알데하이드(45 mg)를 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(51 mg, 수율: 70%)을 수득하였다.Except for using 5-bromo-1H-indole-3-carboxyaldehyde (45 mg) instead of indole-3-carboxyaldehyde as starting material, the same procedure as in Example 1 was carried out to give the desired title compound (51 mg). , Yield: 70%) was obtained.
1H NMR (500 MHz, acetone) δ ppm 12.94 (br. s., 1 H) 8.60 (d, J = 1.96 Hz, 1 H) 8.56 (d, J = 1.96 Hz, 1 H) 8.13 (dd, J = 8.31, 1.22 Hz, 2 H) 7.54 (t, J = 7.58 Hz,2 H) 7.43 (t, J = 7.46 Hz, 1 H) 7.31 (d, J = 2.45 Hz, 1 H) 7.28 (dd, J = 8.56, 2.45 Hz, 1 H) 6.86 (d, J = 8.56 Hz, 1 H) 4.80-4.87 (m, 2 H); 13C NMR (125 MHz, acetone) δ ppm 150.8, 146.4, 134.6, 134.0, 132.3, 131.2, 129.9, 129.2, 127.9, 126.8, 118.1, 118.1, 113.5, 109.1; HRMS (EI+) m/z calcd for C18H13BrN4 [M]+: 364.0323; Found: 364.0326 1 H NMR (500 MHz, acetone) δ ppm 12.94 (br. S., 1 H) 8.60 (d, J = 1.96 Hz, 1 H) 8.56 (d, J = 1.96 Hz, 1 H) 8.13 (dd, J = 8.31, 1.22 Hz, 2H) 7.54 (t, J = 7.58 Hz, 2H) 7.43 (t, J = 7.46 Hz, 1H) 7.31 (d, J = 2.45 Hz, 1H) 7.28 (dd, J = 8.56, 2.45 Hz, 1 H) 6.86 (d, J = 8.56 Hz, 1 H) 4.80-4.87 (m, 2H); 13 C NMR (125 MHz, acetone) δ ppm 150.8, 146.4, 134.6, 134.0, 132.3, 131.2, 129.9, 129.2, 127.9, 126.8, 118.1, 118.1, 113.5, 109.1; HRMS (EI +) m / z calcd for C 18 H 13 BrN 4 [M] < + >:364.0323; Found: 364.0326
실시예Example 20: 5- 20: 5- 클로로Chloro -2-(3--2- (3- 페닐Phenyl -- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
출발물질로서 3-아미노-5-페닐파라졸(16 mg)을 이용하고 인돌-3-카복시알데하이드 대신에 제조예 5에서 얻어진 6-클로로-1H-인돌-3-카복시알데하이드(18 mg)를 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(20 mg, 수율: 61%)을 수득하였다.Use 3-amino-5-phenylparasol (16 mg) as starting material and 6-chloro-1H-indole-3-carboxyaldehyde (18 mg) obtained in Preparation Example 5 instead of indole-3-carboxyaldehyde. The same procedure as in Example 1 was carried out except that the title compound (20 mg, yield: 61%) was obtained.
1H NMR (500 MHz, acetone) δ ppm 12.95 (br. s., 1 H) 8.57 (d, J = 1.96 Hz, 1 H) 8.53 (d, J = 1.96 Hz, 1 H) 8.11 (dd, J = 8.31, 1.22 Hz, 2 H) 7.54 (t, J = 7.70 Hz, 2 H) 7.41-7.45 (m, 1 H) 7.15 (d, J = 8.07 Hz, 1 H) 6.94 (d, J = 2.20 Hz, 1 H) 6.75 (dd, J = 8.07, 2.20 Hz, 1 H) 4.92-4.98 (m, 2 H); 13C NMR (125 MHz, acetone) δ ppm 150.9, 148.4, 134.8, 134.7, 133.3, 131.1, 129.8, 129.3, 129.1, 127.8, 127.8, 123.4, 117.8, 115.6, 113.5; HRMS (EI+) m/z calcd for C18H13ClN4 [M]+: 320.0829; Found: 320.0827 1 H NMR (500 MHz, acetone) δ ppm 12.95 (br.s., 1 H) 8.57 (d, J = 1.96 Hz, 1 H) 8.53 (d, J = 1.96 Hz, 1 H) 8.11 (dd, J = 8.31, 1.22 Hz, 2H) 7.54 (t, J = 7.70 Hz, 2H) 7.41-7.45 (m, 1H) 7.15 (d, J = 8.07 Hz, 1H) 6.94 (d, J = 2.20 Hz , 1 H) 6.75 (dd, J = 8.07, 2.20 Hz, 1 H) 4.92-4.98 (m, 2H); 13 C NMR (125 MHz, acetone) δ ppm 150.9, 148.4, 134.8, 134.7, 133.3, 131.1, 129.8, 129.3, 129.1, 127.8, 127.8, 123.4, 117.8, 115.6, 113.5; HRMS (EI +) m / z calcd for C 18 H 13 ClN 4 [M] < + >:320.0829; Found: 320.0827
실시예Example 21: 2- 21: 2- 플루오로Fluoro -6-(3--6- (3- 페닐Phenyl -- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
출발물질로서 3-아미노-5-페닐파라졸(16 mg)을 이용하고 인돌-3-카복시알데하이드 대신에 제조예 6에서 제조된 7-플루오로-1H-인돌-3-카복시알데하이드(16 mg)를 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(22 mg, 수율: 72%)을 수득하였다.7-fluoro-1H-indole-3-carboxyaldehyde (16 mg) prepared in Preparation Example 6 using 3-amino-5-phenylparazol (16 mg) as starting material and replacing indole-3-carboxyaldehyde. The same procedure as in Example 1 was carried out except that the title compound (22 mg, yield: 72%) was obtained.
1H NMR (500 MHz, acetone) δ ppm 12.98 (br. s., 1 H) 8.61 (d, J = 1.96 Hz, 1 H) 8.57 (d, J = 1.96 Hz, 1 H) 8.12 (dd, J = 8.31, 1.22 Hz, 2 H) 7.53 (t, J = 7.58 Hz, 2 H) 7.41-7.45 (m, 1 H) 7.07 (ddd, J = 11.25, 8.19, 1.35 Hz, 1 H) 7.03 (dd, J = 7.70, 0.86 Hz, 1 H) 6.75 (td, J = 7.89, 5.26 Hz, 1 H) 4.66 (br. s., 2 H); 13C NMR (125 MHz, acetone) δ ppm 153.7, 153.6, 151.7, 150.7, 135.2, 134.6, 131.0, 129.8, 129.2, 129.1, 127.8, 127.4, 127.4, 127.2, 117.8, 117.8, 115.3, 115.1, 113.5; HRMS (EI+) m/z calcd for C18H13FN4 [M]+: 304.1124; Found: 304.1123 1 H NMR (500 MHz, acetone) δ ppm 12.98 (br. S., 1 H) 8.61 (d, J = 1.96 Hz, 1 H) 8.57 (d, J = 1.96 Hz, 1 H) 8.12 (dd, J = 8.31, 1.22 Hz, 2H) 7.53 (t, J = 7.58 Hz, 2H) 7.41-7.45 (m, 1H) 7.07 (ddd, J = 11.25, 8.19, 1.35 Hz, 1H) 7.03 (dd, J = 7.70, 0.86 Hz, 1 H) 6.75 (td, J = 7.89, 5.26 Hz, 1 H) 4.66 (br. S., 2 H); 13 C NMR (125 MHz, acetone) δ ppm 153.7, 153.6, 151.7, 150.7, 135.2, 134.6, 131.0, 129.8, 129.2, 129.1, 127.8, 127.4, 127.4, 127.2, 117.8, 117.8, 115.3, 115.1, 113.5; HRMS (EI +) m / z calcd for C 18 H 13 FN 4 [M] < + >:304.1124; Found: 304.1123
실시예Example 22: 2- 22: 2- 클로로Chloro -6-(3--6- (3- 페닐Phenyl -- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
출발물질로서 3-아미노-5-페닐파라졸(16 mg)을 이용하고 인돌-3-카복시알데하이드 대신에 제조예 7에서 제조된 7-클로로-1H-인돌-3-카복시알데하이드(18 mg)를 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(23 mg, 수율: 71%)을 수득하였다.3-amino-5-phenylparazol (16 mg) was used as starting material and 7-chloro-1H-indole-3-carboxyaldehyde (18 mg) prepared in Preparation Example 7 was used instead of indole-3-carboxyaldehyde. Except for using the same procedure as in Example 1 to give the desired title compound (23 mg, yield: 71%).
1H NMR (500 MHz, acetone) δ ppm 12.95 (br. s., 1 H) 8.58 (d, J = 1.96 Hz, 1 H) 8.56 (d, J = 1.96 Hz, 1 H) 8.13 (dd, J = 8.07, 0.98 Hz, 2 H) 7.54 (t, J = 7.83 Hz, 2 H) 7.43 (t, J = 7.34 Hz, 1 H) 7.33 (dd, J = 8.07, 1.47 Hz, 1 H) 7.15 (dd, J = 7.46, 1.10 Hz, 1 H) 6.79 (t, J = 7.83 Hz, 1 H) 4.82-4.91 (m, 2 H) 13C NMR (125 MHz, DMSO-d6) δ ppm 149.5, 142.0, 133.0, 130.1, 129.9, 128.9, 128.7, 128.1, 126.6, 125.0, 118.2, 117.2, 112.0; HRMS (EI+) m/z calcd for C18H13ClN4 [M]+: 320.0829; Found: 320.0829 1 H NMR (500 MHz, acetone) δ ppm 12.95 (br. S., 1 H) 8.58 (d, J = 1.96 Hz, 1 H) 8.56 (d, J = 1.96 Hz, 1 H) 8.13 (dd, J = 8.07, 0.98 Hz, 2 H) 7.54 (t, J = 7.83 Hz, 2 H) 7.43 (t, J = 7.34 Hz, 1 H) 7.33 (dd, J = 8.07, 1.47 Hz, 1 H) 7.15 (dd , J = 7.46, 1.10 Hz, 1 H) 6.79 (t, J = 7.83 Hz, 1 H) 4.82-4.91 (m, 2H) 13 C NMR (125 MHz, DMSO-d 6 ) δ ppm 149.5, 142.0, 133.0, 130.1, 129.9, 128.9, 128.7, 128.1, 126.6, 125.0, 118.2, 117.2, 112.0; HRMS (EI +) m / z calcd for C 18 H 13 ClN 4 [M] < + >:320.0829; Found: 320.0829
실시예Example 23: N- 23: N- 메틸methyl -2-(3--2- (3- 페닐Phenyl -- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
출발물질로서 인돌-3-카복시알데하이드 대신에 제조예 13에서 얻어진 1-메틸-1H-인돌-3-카복시알데하이드(32 mg)를 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(41 mg, 수율: 68%)을 수득하였다.The same procedure as in Example 1 was carried out except that 1-methyl- 1H -indole-3-carboxyaldehyde (32 mg) obtained in Preparation Example 13 was used instead of indole-3-carboxyaldehyde as the starting material. Compound (41 mg, yield: 68%) was obtained.
1H NMR (500 MHz, acetone) δ ppm 12.92 (br. s., 1 H) 8.51 (d, J = 1.96 Hz, 1 H) 8.46 (d, J = 1.96 Hz, 1 H) 8.11 (dd, J = 8.31, 1.22 Hz, 2 H) 7.53 (t, J = 7.58 Hz, 2 H) 7.40-7.45 (m, 1 H) 7.28 (td, J = 7.83, 1.71 Hz, 1 H) 7.12 (dd, J = 7.34, 1.71 Hz, 1 H) 6.71-6.77 (m, 2 H) 4.74 (d, J = 4.65 Hz, 1 H) 2.79 (d, J = 5.14 Hz, 3 H); 13C NMR (75 MHz, acetone) δ ppm 153.7, 150.3, 147.4, 143.6, 133.7, 130.7, 129.2, 129.2, 128.9, 128.1, 126.8, 124.3, 116.3, 112.5, 109.7, 29.8; HRMS (EI+) m/z calcd for C19H16N4 [M]+: 300.1375; Found: 300.1376 1 H NMR (500 MHz, acetone) δ ppm 12.92 (br.s., 1 H) 8.51 (d, J = 1.96 Hz, 1 H) 8.46 (d, J = 1.96 Hz, 1 H) 8.11 (dd, J = 8.31, 1.22 Hz, 2H) 7.53 (t, J = 7.58 Hz, 2H) 7.40-7.45 (m, 1H) 7.28 (td, J = 7.83, 1.71 Hz, 1H) 7.12 (dd, J = 7.34, 1.71 Hz, 1H) 6.71-6.77 (m, 2H) 4.74 (d, J = 4.65 Hz, 1H) 2.79 (d, J = 5.14 Hz, 3H); 13 C NMR (75 MHz, acetone) δ ppm 153.7, 150.3, 147.4, 143.6, 133.7, 130.7, 129.2, 129.2, 128.9, 128.1, 126.8, 124.3, 116.3, 112.5, 109.7, 29.8; HRMS (EI +) m / z calcd for C 19 H 16 N 4 [M] < + >:300.1375; Found: 300.1376
실시예Example 24: N-벤질-2-(3- 24: N-benzyl-2- (3- 페닐Phenyl -- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
출발물질로서 인돌-3-카복시알데하이드 대신에 제조예 14에서 얻어진 1-벤질-1H-인돌-3-카복시알데하이드(47 mg)를 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(63 mg, 수율: 83%)을 수득하였다.The same procedure as in Example 1 was carried out except that 1-benzyl- 1H -indole-3-carboxyaldehyde (47 mg) obtained in Preparation Example 14 was used instead of indole-3-carboxyaldehyde as the starting material. Compound (63 mg, yield: 83%) was obtained.
1H NMR (500 MHz, acetone) δ ppm 12.93 (br. s., 1 H) 8.61 (d, J = 1.96 Hz, 1 H) 8.56 (d, J = 1.96 Hz, 1 H) 8.13 (dd, J = 8.31, 1.22 Hz, 2 H) 7.55 (t, J = 7.70 Hz, 2 H) 7.44 (tt, J = 7.37, 1.31 Hz, 1 H) 7.40 (d, J = 7.09 Hz, 2 H) 7.26-7.31 (m, 2 H) 7.14-7.22 (m, 3 H) 6.74 (td, J = 7.40, 1.10 Hz, 1 H) 6.69 (d, J = 8.07 Hz, 1 H) 5.31 (t, J = 5.75 Hz, 1 H) 4.39-4.42 (m, 2 H); 13C NMR (125 MHz, acetone) δ ppm 151.3, 146.2, 140.5, 133.9, 131.1, 130.6, 129.4, 129.2, 129.1, 128.6, 128.4, 127.3, 127.1, 126.9, 124.8, 117.0, 117.0, 112.8, 111.3, 47.3, 47.3; HRMS (EI+) m/z calcd for C25H20N4 [M]+: 376.1688; Found: 376.1691 1 H NMR (500 MHz, acetone) δ ppm 12.93 (br. S., 1 H) 8.61 (d, J = 1.96 Hz, 1 H) 8.56 (d, J = 1.96 Hz, 1 H) 8.13 (dd, J = 8.31, 1.22 Hz, 2H) 7.55 (t, J = 7.70 Hz, 2H) 7.44 (tt, J = 7.37, 1.31 Hz, 1H) 7.40 (d, J = 7.09 Hz, 2H) 7.26-7.31 (m, 2H) 7.14-7.22 (m, 3H) 6.74 (td, J = 7.40, 1.10 Hz, 1H) 6.69 (d, J = 8.07 Hz, 1H) 5.31 (t, J = 5.75 Hz, 1 H) 4.39-4. 42 (m, 2H); 13 C NMR (125 MHz, acetone) δ ppm 151.3, 146.2, 140.5, 133.9, 131.1, 130.6, 129.4, 129.2, 129.1, 128.6, 128.4, 127.3, 127.1, 126.9, 124.8, 117.0, 117.0, 112.8, 111.3, 47.3 , 47.3; HRMS (EI +) m / z calcd for C 25 H 20 N 4 [M] < + >:376.1688; Found: 376.1691
실시예Example 25: 1-벤질-3-(2-(3- 25: 1-benzyl-3- (2- (3- 페닐Phenyl -- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 페닐Phenyl )) 우레아Urea
출발물질로서 3-아미노-5-페닐파라졸(16 mg)을 이용하고 인돌-3-카복시알데하이드 대신에 제조예 15에서 얻어진 N-벤질-3-포밀-1H-인돌-1-카복시아마이드(28 mg)를 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(19 mg, 수율: 51%)을 수득하였다.N-benzyl-3-formyl- 1H -indole-1-carboxamide (28) obtained in Preparation Example 15 using 3-amino-5-phenylparasol (16 mg) as a starting material and replacing indole-3-carboxyaldehyde (28) Except for using mg), the same procedure as in Example 1 was carried out to obtain the title compound (19 mg, yield: 51%).
1H NMR (500 MHz, DMSO-d6) δ ppm 13.93 (br. s., 1 H) 8.50 (d, J = 1.71 Hz, 1 H) 8.48 (d, J = 1.71 Hz, 1 H) 8.05 (d, J = 7.09 Hz, 2 H) 7.95 (d, J = 8.07 Hz, 1 H) 7.65 (s, 1 H) 7.51 (t, J = 7.58 Hz, 2 H) 7.42 (t, J = 7.34 Hz, 1 H) 7.32-7.38 (m, 2 H) 7.11-7.22 (m, 6 H) 6.84 (t, J = 5.75 Hz, 1 H) 4.20 (d, J = 5.62 Hz, 2 H); 13C NMR (125 MHz, DMSO-d6) δ ppm 155.5, 152.3, 149.8, 140.0, 137.4, 133.2, 131.0, 130.4, 129.9, 129.0, 128.2, 127.0, 126.6, 122.9, 122.3, 112.1, 42.8, 42.8; HRMS (EI+) m/z calcd for C26H21N5O [M]+: 419.1746; Found: 419.1744. 1 H NMR (500 MHz, DMSO-d 6 ) δ ppm 13.93 (br.s., 1 H) 8.50 (d, J = 1.71 Hz, 1 H) 8.48 (d, J = 1.71 Hz, 1 H) 8.05 ( d, J = 7.09 Hz, 2H) 7.95 (d, J = 8.07 Hz, 1H) 7.65 (s, 1H) 7.51 (t, J = 7.58 Hz, 2H) 7.42 (t, J = 7.34 Hz, 1 H) 7.32-7.38 (m, 2H) 7.11-7.22 (m, 6H) 6.84 (t, J = 5.75 Hz, 1H) 4.20 (d, J = 5.62 Hz, 2H); 13 C NMR (125 MHz, DMSO-d 6 ) δ ppm 155.5, 152.3, 149.8, 140.0, 137.4, 133.2, 131.0, 130.4, 129.9, 129.0, 128.2, 127.0, 126.6, 122.9, 122.3, 112.1, 42.8, 42.8; HRMS (EI +) m / z calcd for C 26 H 21 N 5 O [M] < + >:419.1746; Found: 419.1744.
실시예Example 26: 2-(3- 26: 2- (3- 메틸methyl -1--One- 페닐Phenyl -- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
출발물질로서 인돌-3-카복시알데하이드(17 mg)를 이용하고 3-아미노-5-페닐피라졸 대신에 5-아미노-3-메틸-1-페닐 피라졸(15 mg)을 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(16 mg, 수율: 54%)을 수득하였다.Except using indole-3-carboxyaldehyde (17 mg) as starting material and 5-amino-3-methyl-1-phenyl pyrazole (15 mg) instead of 3-amino-5-phenylpyrazole Was subjected to the same procedure as in Example 1 to obtain the desired title compound (16 mg, yield: 54%).
1H NMR (500 MHz, Acetone-d6) δ 8.66 (d, J = 1.96 Hz, 1 H) 8.45 (d, J = 8.56 Hz, 2H) 8.28 (d, J = 1.96 Hz, 1H) 7.55 (t, J = 7.58Hz, 2H) 7.29 (td, J = 7.46, 0.98 Hz, 1H) 7.11-7.18 (m, 2H) 6.89 (d, J =7.83 Hz, 1H) 6.76 (t, J = 7.46 Hz, 1H) 4.62 (br. s., 2H) 2.66 (s, 3H); 13C NMR (125 MHz, Acetone-d6) δ 175.18, 150.20, 145.99, 143.16, 140.28, 130.99, 130.01, 129.72, 129.12, 129.10, 125.23, 123.56, 119.99, 117.72, 117.37, 115.78, 12.55; LRMS (MALDI) m/z calcd for C19H17N4 [M+H]+: 301.14; Found: 301.45. 1 H NMR (500 MHz, Acetone- d6 ) δ 8.66 (d, J = 1.96 Hz, 1 H) 8.45 (d, J = 8.56 Hz, 2H) 8.28 (d, J = 1.96 Hz, 1H) 7.55 (t, J = 7.58 Hz, 2H) 7.29 (td, J = 7.46, 0.98 Hz, 1H) 7.11-7.18 (m, 2H) 6.89 (d, J = 7.83 Hz, 1H) 6.76 (t, J = 7.46 Hz, 1H) 4.62 (br. S., 2 H) 2.66 (s, 3 H); 13 C NMR (125 MHz, Acetone- d6 ) δ 175.18, 150.20, 145.99, 143.16, 140.28, 130.99, 130.01, 129.72, 129.12, 129.10, 125.23, 123.56, 119.99, 117.72, 117.37, 115.78, 12.55; LRMS (MALDI) m / z calcd for C 19 H 17 N 4 [M + H] +: 301.14; Found: 301.45.
실시예Example 27: 2-(3-(4- 27: 2- (3- (4- 메톡시페닐Methoxyphenyl )-) - 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-3--5-day) -3- 니트로벤젠아민Nitrobenzeneamine
출발물질로서 인돌-3-카복시알데하이드 대신에 제조예 1에서 제조된 4-니트로-1H-인돌-3-카복시알데하이드(19 mg)를 사용하고 3-아미노-5-페닐피라졸 대신에 3-아미노-5-(4-메톡시페닐)피라졸(19 mg)을 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(27 mg, 수율: 74%)을 수득하였다.As a starting material, 4-nitro-1H-indole-3-carboxyaldehyde (19 mg) prepared in Preparation Example 1 was used instead of indole-3-carboxyaldehyde and 3-amino instead of 3-amino-5-phenylpyrazole. Except for using -5- (4-methoxyphenyl) pyrazole (19 mg), the same procedure as in Example 1 was carried out to obtain the title compound (27 mg, yield: 74%).
1H NMR (500 MHz, DMSO-d6) δ 13.81 (br. s., 1H) 8.35 (d, J= 1.96 Hz, 1H) 8.33 (d, J = 1.96 Hz, 1H) 7.93-7.98 (m, 2H) 7.31 (t, J = 8.19 Hz, 1H) 7.14(d, J = 8.07 Hz, 1H) 7.07 (dd, J = 8.68, 2.57 Hz, 3H) 5.37 (s, 2H) 3.78-3.82 (m, 3H); 13C NMR (125 MHz, DMSO-d6) δ 159.42, 152.47, 151.18, 149.62, 148.99, 142.84, 131.06, 129.33, 128.05, 125.71, 123.12, 118.69,115.31, 114.55, 112.23, 110.64, 55.31, 55.28; LRMS (MALDI) m/z calcd for C19H16N5O3 [M+H]+: 362.35; Found: 362.34 1 H NMR (500 MHz, DMSO- d 6) δ 13.81 (br. S., 1H) 8.35 (d, J = 1.96 Hz, 1H) 8.33 (d, J = 1.96 Hz, 1H) 7.93-7.98 (m, 2H) 7.31 (t, J = 8.19 Hz, 1H) 7.14 (d, J = 8.07 Hz, 1H) 7.07 (dd, J = 8.68, 2.57 Hz, 3H) 5.37 (s, 2H) 3.78-3.82 (m, 3H ); 13 C NMR (125 MHz, DMSO- d 6) δ 159.42, 152.47, 151.18, 149.62, 148.99, 142.84, 131.06, 129.33, 128.05, 125.71, 123.12, 118.69,115.31, 114.55, 112.23, 110.64, 55.31, 55.28 LRMS (MALDI) m / z calcd for C 19 H 16 N 5 O 3 [M + H] +: 362.35; Found: 362.34
실시예Example 28: 5- 28: 5- 플루오로Fluoro -2-(3-(4--2- (3- (4- 메톡시페닐Methoxyphenyl )-) - 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
출발물질로서 인돌-3-카복시알데하이드 대신에 6-플루오로인돌-3-카복시알데하이드(16 mg)를 사용하고 3-아미노-5-페닐피라졸 대신에 3-아미노-5-(4-메톡시페닐)피라졸(19 mg)을 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(25 mg, 수율: 74%)을 수득하였다.Use 6-fluoroindole-3-carboxyaldehyde (16 mg) instead of indole-3-carboxyaldehyde as starting material and 3-amino-5- (4-methoxy instead of 3-amino-5-phenylpyrazole The same procedure as in Example 1 was carried out except for using phenyl) pyrazole (19 mg) to give the desired title compound (25 mg, yield: 74%).
1H NMR (500 MHz, Acetone-d6) δ 12.78 (br. s., 1H) 8.53 (d,J = 1.71 Hz, 1H) 8.46 (d, J = 1.71 Hz, 1H) 8.02-8.07 (m, 2H) 7.15 (dd, J =8.31, 6.60 Hz, 1H) 7.07-7.12 (m, 2H) 6.65 (dd, J = 11.37, 2.57 Hz, 1H) 6.49(td, J = 8.50, 2.57 Hz, 1H) 4.90-4.99 (m, 2H) 3.87 (s, 3H); 13C NMR (125MHz, DMSO-d6) δ 163.88, 161.96, 159.34, 152.11, 149.75, 148.21, 148.11,142.81, 132.54, 132.47, 130.07, 128.06, 127.94, 125.9, 119.46, 114.51, 112.05, 103.01, 102.84,101.13, 100.94, 55.29, 55.25; LRMS (MALDI) m/z calcd for C19H16FN4O [M+H]+: 335.35;Found: 335.52 1 H NMR (500 MHz, Acetone- d6 ) δ 12.78 (br. S., 1H) 8.53 (d, J = 1.71 Hz, 1H) 8.46 (d, J = 1.71 Hz, 1H) 8.02-8.07 (m, 2H ) 7.15 (dd, J = 8.31, 6.60 Hz, 1H) 7.07-7.12 (m, 2H) 6.65 (dd, J = 11.37, 2.57 Hz, 1H) 6.49 (td, J = 8.50, 2.57 Hz, 1H) 4.90- 4.99 (m, 2 H) 3.87 (s, 3 H); 13 C NMR (125 MHz, DMSO- d 6) δ 163.88, 161.96, 159.34, 152.11, 149.75, 148.21, 148.11,142.81, 132.54, 132.47, 130.07, 128.06, 127.94, 125.9, 119.46, 114.51, 112.05, 103.01, 102.01, 102.01 , 100.94, 55.29, 55.25; LRMS (MALDI) m / z calcd for C19H16FN4O [M + H] +: 335.35; Found: 335.52
실시예Example 29: 2- 29: 2- 플루오로Fluoro -6-(3--6- (3- 메틸methyl -- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
출발물질로서 인돌-3-카복시알데하이드 대신에 제조예 6에서 제조된 7-플루오로-1H-인돌-3-카복시알데하이드(16 mg)를 사용하고 3-아미노-5-페닐피라졸 대신에 3-아미노-5-메틸피라졸(10 mg)을 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하여 원하는 표제 화합물(13 mg, 수율: 55%)을 수득하였다.Use 7-fluoro- 1H -indole-3-carboxyaldehyde (16 mg) prepared in Preparation Example 6 instead of indole-3-carboxyaldehyde as starting material and 3-in place of 3-amino-5-phenylpyrazole Except for using amino-5-methylpyrazole (10 mg), the same procedure as in Example 1 was carried out to obtain the title compound (13 mg, yield: 55%).
1H NMR (500 MHz, Acetone-d6) δ 12.39 (br. s., 1H) 8.52 (d, J= 1.96 Hz, 1H) 8.19 (d, J = 1.96 Hz, 1H) 7.05 (ddd, J = 11.19, 8.13, 1.22 Hz,1H) 6.97 (d, J = 7.58 Hz, 1H) 6.74 (td, J = 7.95, 5.38 Hz, 1H) 4.58 (br. s., 2H) 2.56 (s, 3H); 13C NMR (125 MHz, Acetone-d6) δ 153.59, 151.7, 150.36, 135.09,134.99, 130, 127.83, 127.81, 127.48, 127.45, 127.36, 127.34, 117.84, 117.82, 117.78, 117.75,115.24, 115.14, 114.99, 12.56; LRMS (MALDI) m/z calcd for C13H12FN4 [M+H]+: 243.25; Found: 243.47. 1 H NMR (500 MHz, Acetone- d6 ) δ 12.39 (br. S., 1H) 8.52 (d, J = 1.96 Hz, 1H) 8.19 (d, J = 1.96 Hz, 1H) 7.05 (ddd, J = 11.19 , 8.13, 1.22 Hz, 1H) 6.97 (d, J = 7.58 Hz, 1H) 6.74 (td, J = 7.95, 5.38 Hz, 1H) 4.58 (br. S., 2H) 2.56 (s, 3H); 13C NMR (125 MHz, Acetone- d6 ) δ 153.59, 151.7, 150.36, 135.09,134.99, 130, 127.83, 127.81, 127.48, 127.45, 127.36, 127.34, 117.84, 117.82, 117.78, 117.75, 115.24, 115.14, 12. ; LRMS (MALDI) m / z calcd for C 13 H 12 FN 4 [M + H] +: 243.25; Found: 243.47.
실시예Example 30: 5- 30: 5- 플루오로Fluoro -2-(3--2- (3- 메틸methyl -1H--1H- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
출발물질로서 3-아미노-5-메틸파라졸(39 mg) 및 6-플루오로-1H-인돌-3-카복시알데하이드(65 mg)를 사용하여 실시예 1과 동일한 과정을 수행하되, 크로마토그래피에 의한 정제없이 재결정에 의해 원하는 표제 화합물(18 mg, 수율: 19%)을 수득하였다. The same procedure as in Example 1 was carried out using 3-amino-5-methylparasol (39 mg) and 6-fluoro-1H-indole-3-carboxyaldehyde (65 mg) as starting materials, but the chromatography was carried out. Recrystallization without purification gave the desired title compound (18 mg, yield: 19%).
1H NMR (500 MHz, acetone) d ppm 2.55 (s, 3 H) 4.89 (br. s., 2 H) 6.47 (tt, J=8.47, 2.05 Hz, 1 H) 6.63 (dt, J=11.31, 2.05 Hz, 1 H) 7.10 (td, J=7.46, 1.47 Hz,1 H) 8.14 (d, J=1.96 Hz, 1 H) 8.47 (d, J=1.96 Hz, 1 H) 12.36 (br. s., 1 H); LRMS (MALDI) m/z calcd for C13H12FN4 [M+H]+: 243.10; Found: 243.40 1 H NMR (500 MHz, acetone) d ppm 2.55 (s, 3 H) 4.89 (br. S., 2 H) 6.47 (tt, J = 8.47, 2.05 Hz, 1 H) 6.63 (dt, J = 11.31, 2.05 Hz, 1H) 7.10 (td, J = 7.46, 1.47 Hz, 1H) 8.14 (d, J = 1.96 Hz, 1H) 8.47 (d, J = 1.96 Hz, 1H) 12.36 (br. S. , 1 H); LRMS (MALDI) m / z calcd for C 13 H 12 FN 4 [M + H] < + >:243.10; Found: 243.40
실시예Example 31: 3-니트로-2-(3- 31: 3-nitro-2- (3- 메틸methyl -1H--1H- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
출발물질로서 3-아미노-5-메틸파라졸(24 mg) 및 4-니트로-1H-인돌-3-카복시알데하이드(48 mg)를 사용하여 실시예 1과 동일한 과정을 수행하되, 크로마토그래피에 의한 정제없이 재결정에 의해 원하는 표제 화합물(6 mg, 수율: 9%)을 수득하였다.The same procedure as in Example 1 was carried out using 3-amino-5-methylparasol (24 mg) and 4-nitro- 1H -indole-3-carboxyaldehyde (48 mg) as starting materials, but by chromatography. Recrystallization without purification gave the desired title compound (6 mg, yield: 9%).
1H NMR (500 MHz, DMSO-d6) d ppm 2.51 (s, 3 H) 5.34 (br. s., 2 H) 7.06 (t, J=6.85 Hz, 1 H) 7.12 (t, J=6.85 Hz, 1 H) 7.27 - 7.34 (m, 1 H) 8.08 (d, J=5.62 Hz, 1 H) 8.25 (d, J=5.38 Hz, 1 H) 13.33 (br. s., 1 H); 13C NMR (125 MHz, DMSO-d6) d ppm 12.67 110.85 114.67 115.74 118.84 122.14 129.59 130.63 141.65 149.30 149.66 151.67 152.37; LRMS (MALDI) m/z calcd for C13H12N5O2 [M+H]+:270.09 ; Found: 270.37 1 H NMR (500 MHz, DMSO-d6) d ppm 2.51 (s, 3 H) 5.34 (br. S., 2 H) 7.06 (t, J = 6.85 Hz, 1 H) 7.12 (t, J = 6.85 Hz , 1 H) 7.27-7.34 (m, 1 H) 8.08 (d, J = 5.82 Hz, 1 H) 8.25 (d, J = 5.38 Hz, 1 H) 13.33 (br. S., 1 H); 13 C NMR (125 MHz, DMSO- d 6) d ppm 12.67 110.85 114.67 115.74 118.84 122.14 129.59 130.63 141.65 149.30 149.66 151.67 152.37; LRMS (MALDI) m / z calcd for C 13 H 12 N 5 0 2 [M + H] < + >:270.09; Found: 270.37
실시예Example 32: 32: 메틸methyl 3-아미노-2-(3- 3-amino-2- (3- 메틸methyl -- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤조에이트Benzoate
출발물질로서 3-아미노-5-메틸파라졸(29 mg) 및 메틸 3-포밀-1H-인돌-4-카복실레이트(61 mg)를 사용하여 실시예 1과 동일한 과정을 수행하되, 크로마토그래피에 의한 정제없이 재결정에 의해 원하는 표제 화합물(39 mg, 수율: 46%)을 수득하였다.The same procedure as in Example 1 was carried out using 3-amino-5-methylparasol (29 mg) and methyl 3-formyl- 1H -indole-4-carboxylate (61 mg) as starting materials, but the chromatography was carried out. Recrystallization without purification gave the desired title compound (39 mg, yield: 46%).
1H NMR (500 MHz, DMSO-d6) d ppm 2.50 (s, 3 H) 3.45 (s, 3 H) 4.88 (s, 2 H) 6.97 (d, J=8.07 Hz, 1 H) 7.04 (d, J=7.83 Hz, 1 H) 7.20 (t, J=7.83 Hz, 1 H) 7.96 (s, 1 H) 8.16 (s, 1 H) 13.20 (s, 1 H); 13C NMR (125 MHz, DMSO-d6) d ppm 114.43 117.48 118.29 122.80 125.77 127.61 128.75 129.96 132.53 141.36 147.91 150.01 152.10 168.34; LRMS (MALDI) m/z calcd for C15H15N4O2 [M+H]+:283.11 ; Found: 283.36 1 H NMR (500 MHz, DMSO-d6) d ppm 2.50 (s, 3 H) 3.45 (s, 3 H) 4.88 (s, 2 H) 6.97 (d, J = 8.07 Hz, 1 H) 7.04 (d, J = 7.83 Hz, 1 H) 7.20 (t, J = 7.83 Hz, 1 H) 7.96 (s, 1 H) 8.16 (s, 1 H) 13.20 (s, 1 H); 13 C NMR (125 MHz, DMSO- d 6) d ppm 114.43 117.48 118.29 122.80 125.77 127.61 128.75 129.96 132.53 141.36 147.91 150.01 152.10 168.34; LRMS (MALDI) m / z calcd for C 15 H 15 N 4 O 2 [M + H] +: 283.11; Found: 283.36
실시예Example 33: 4- 33: 4- 브로모Bromo -2-(3--2- (3- 메틸methyl -- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
출발물질로서 3-아미노-5-메틸파라졸(29 mg) 및 5-브로모-1H-인돌-3-카복시알데하이드(67 mg)를 사용하여 실시예 1과 동일한 과정을 수행하되, 크로마토그래피를 이용한 정제없이 재결정에 의해 원하는 표제 화합물(47 mg, 수율: 52%)을 수득하였다.The same procedure as in Example 1 was carried out using 3-amino-5-methylparasol (29 mg) and 5-bromo-1H-indole-3-carboxyaldehyde (67 mg) as starting materials, but chromatography was performed. Recrystallization without the purification was used to give the desired title compound (47 mg, yield: 52%).
1H NMR (500 MHz, DMSO-d6) d ppm 2.50 (s, 3 H) 5.12 (br. s., 2 H) 6.73 (d, J=8.56 Hz, 1 H) 7.07 - 7.31 (m, 2 H) 8.18 (s, 1 H) 8.45 (s, 1 H) 13.24 (br. s., 1H); 13C NMR (125 MHz, DMSO-d6) d ppm 12.68 69.86 107.46 114.45 117.49 125.72 126.68 129.82 131.31 133.08 145.93 149.53; LRMS (MALDI) m/z calcd for C13H12BrN4 [M+H]+:303.02 ; Found: 303.29 1 H NMR (500 MHz, DMSO-d6) d ppm 2.50 (s, 3 H) 5.12 (br. S., 2 H) 6.73 (d, J = 8.56 Hz, 1 H) 7.07-7.31 (m, 2 H ) 8.18 (s, 1 H) 8.45 (s, 1 H) 13.24 (br. S., 1 H); 13 C NMR (125 MHz, DMSO- d 6) d ppm 12.68 69.86 107.46 114.45 117.49 125.72 126.68 129.82 131.31 133.08 145.93 149.53; LRMS (MALDI) m / z calcd for C 13 H 12 BrN 4 [M + H] < + >:303.02; Found: 303.29
실시예Example 34: 4- 34: 4- 클로로Chloro -2-(3--2- (3- 메틸methyl -- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
출발물질로서 3-아미노-5-메틸파라졸(15 mg) 및 5-클로로-1H-인돌-3-카복시알데하이드(27 mg)를 사용하여 실시예 1과 동일한 과정을 수행하되, 크로마토그래피를 이용한 정제없이 재결정에 의해 원하는 표제 화합물(10 mg, 수율: 26%)을 수득하였다.Using the same procedure as in Example 1 using 3-amino-5-methylparazol (15 mg) and 5-chloro-1H-indole-3-carboxyaldehyde (27 mg) as starting materials, Recrystallization without purification gave the desired title compound (10 mg, yield: 26%).
1H NMR (500 MHz, DMSO-d6) d ppm 2.50 (br. s., 3 H) 5.08 (br. s., 2 H) 6.78 (d, J=8.31 Hz, 1 H) 6.95 - 7.25 (m, 2 H) 8.18 (s, 1 H) 8.45 (s, 1 H) 13.24 (br.s., 1 H); 13C NMR (125 MHz, DMSO-d6) d ppm 12.24 114.00 116.57 119.68 124.69 126.32 128.04 129.91 141.25 145.11 149.09 151.67 1 H NMR (500 MHz, DMSO-d6) d ppm 2.50 (br. S., 3 H) 5.08 (br. S., 2 H) 6.78 (d, J = 8.31 Hz, 1 H) 6.95-7.25 (m , 2H) 8.18 (s, 1H) 8.45 (s, 1H) 13.24 (br.s., 1H); 13 C NMR (125 MHz, DMSO- d 6) d ppm 12.24 114.00 116.57 119.68 124.69 126.32 128.04 129.91 141.25 145.11 149.09 151.67
실시예Example 35: 5- 35: 5- 클로로Chloro -2-(3--2- (3- 메틸methyl -- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
출발물질로서 3-아미노-5-메틸파라졸(10 mg) 및 6-클로로-1H-인돌-3-카복시알데하이드(18 mg)를 사용하여 실시예 1과 동일한 과정을 수행하되, 크로마토그래피를 이용한 정제없이 재결정에 의해 원하는 표제 화합물(5 mg, 수율: 20%)을 수득 하였다.The same procedure as in Example 1 was carried out using 3-amino-5-methylparasol (10 mg) and 6-chloro-1H-indole-3-carboxyaldehyde (18 mg) as starting materials, but using chromatography. Recrystallization without purification gave the desired title compound (5 mg, yield: 20%).
1H NMR (500 MHz, DMSO-d6) d ppm 2.50 (s, 3 H) 5.23 (br. s., 2 H) 6.64 (d, J=8.07 Hz, 1 H) 6.82 (s, 1 H) 7.02 (dd, J=7.95, 1.83 Hz, 1 H) 8.14 (s, 1 H) 8.42(s, 1 H) 13.24 (br. s., 1 H); 13C NMR (125 MHz, DMSO-d6) d ppm 12.66 103.26 114.39 114.50 116.37 122.40 126.85 129.65 132.70 133.23 148.01 149.62; LRMS (MALDI) m/z calcd for C13H12ClN4 [M+H]+:259.07 ; Found: 259.43 1 H NMR (500 MHz, DMSO-d6) d ppm 2.50 (s, 3 H) 5.23 (br. S., 2 H) 6.64 (d, J = 8.07 Hz, 1 H) 6.82 (s, 1 H) 7.02 (dd, J = 7.95, 1.83 Hz, 1 H) 8.14 (s, 1 H) 8.42 (s, 1 H) 13.24 (br. s., 1 H); 13 C NMR (125 MHz, DMSO- d 6) d ppm 12.66 103.26 114.39 114.50 116.37 122.40 126.85 129.65 132.70 133.23 148.01 149.62; LRMS (MALDI) m / z calcd for C 13 H 12 ClN 4 [M + H] +: 259.07; Found: 259.43
실시예Example 36: N- 36: N- 메틸methyl -2-(3--2- (3- 메틸methyl -- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
출발물질로서 3-아미노-5-메틸피라졸(15 mg) 및 1-메틸-1H-인돌-3-카복시알데하이드(24 mg)를 사용하여 실시예 1과 동일한 과정을 수행하되, 크로마토그래피를 이용한 정제없이 재결정에 의해 원하는 표제 화합물(16 mg, 수율: 46%)을 수득하였다.Using the same procedure as in Example 1 using 3-amino-5-methylpyrazole (15 mg) and 1-methyl- 1H -indole-3-carboxyaldehyde (24 mg) as starting materials, Recrystallization without purification gave the desired title compound (16 mg, yield: 46%).
1H NMR (500 MHz, DMSO-d6) d ppm 2.50 (s, 3 H) 2.64 (d, J=3.42 Hz, 3 H) 4.96 (d, J=3.91 Hz, 1 H) 6.62 (d, J=8.07 Hz, 1 H) 6.68 (t, J=7.34 Hz, 1 H) 7.01(d, J=7.34 Hz, 1 H) 7.22 (t, J=7.70 Hz, 1 H) 8.12 (s, 1 H) 8.39 (s, 1 H) 13.22 (br. s., 1 H); 13C NMR (125 MHz, DMSO-d6) d ppm 12.66 30.71 110.00 114.44 116.31 124.48 127.68 129.33 129.95 131.04 147.54 149.98 152.12; LRMS (MALDI) m/z calcd for C14H15N4 [M+H]+:238.12 ; Found: 239.47 1 H NMR (500 MHz, DMSO-d6) d ppm 2.50 (s, 3H) 2.64 (d, J = 3.42 Hz, 3H) 4.96 (d, J = 3.91 Hz, 1H) 6.62 (d, J = 8.07 Hz, 1 H) 6.68 (t, J = 7.34 Hz, 1 H) 7.01 (d, J = 7.34 Hz, 1 H) 7.22 (t, J = 7.70 Hz, 1 H) 8.12 (s, 1 H) 8.39 (s, 1 H) 13.22 (br. s., 1 H); 13 C NMR (125 MHz, DMSO- d 6) d ppm 12.66 30.71 110.00 114.44 116.31 124.48 127.68 129.33 129.95 131.04 147.54 149.98 152.12; LRMS (MALDI) m / z calcd for C 14 H 15 N 4 [M + H] < + >:238.12; Found: 239.47
실시예Example 37: N-벤질-2-(3- 37: N-benzyl-2- (3- 메틸methyl -- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 벤젠아민Benzeneamine
출발물질로서 3-아미노-5-메틸피라졸(15 mg) 및 1-벤질-1H-인돌-3-카복시알데하이드(35 mg)를 사용하는 것을 제외하고는 실시예 1과 동일한 과정을 수행하되, 크로마토그래피를 이용한 정제없이 재결정에 의해 원하는 표제 화합물(13 mg, 수율: 27%)을 수득하였다.The same procedure as in Example 1 was carried out except that 3-amino-5-methylpyrazole (15 mg) and 1-benzyl- 1H -indole-3-carboxyaldehyde (35 mg) were used as starting materials, Recrystallization without purification using chromatography gave the desired title compound (13 mg, yield: 27%).
1H NMR (500 MHz, DMSO-d6) d ppm 2.51 (s, 3 H) 4.26 (d, J=3.91 Hz, 2 H) 5.58 (br. s., 1 H) 6.49 (d, J=8.07 Hz, 1 H) 6.63 (t, J=7.34 Hz, 1 H) 7.02 (d,J=7.34 Hz, 1 H) 7.06 (t, J=7.70 Hz, 1 H) 7.18 (t, J=6.60 Hz, 1 H) 7.25 - 7.36 (m, 4 H) 8.17 (s, 1 H) 8.47 (s, 1 H) 13.24 (br. s., 1 H); 13C NMR (125 MHz, DMSO-d6) d ppm 12.92 47.11 111.42 114.82 116.86 125.02 127.18 127.49 128.01 128.97 129.34 130.29 131.54 141.01 146.30 150.24 152.40; LRMS (MALDI) m/z calcd for C20H19N4 [M+H]+:315.15 ; Found: 315.43 1 H NMR (500 MHz, DMSO-d6) d ppm 2.51 (s, 3 H) 4.26 (d, J = 3.91 Hz, 2 H) 5.58 (br. S., 1 H) 6.49 (d, J = 8.07 Hz , 1 H) 6.63 (t, J = 7.34 Hz, 1 H) 7.02 (d, J = 7.34 Hz, 1 H) 7.06 (t, J = 7.70 Hz, 1 H) 7.18 (t, J = 6.60 Hz, 1 H) 7.25-7.36 (m, 4H) 8.17 (s, 1H) 8.47 (s, 1H) 13.24 (br.s., 1H); 13 C NMR (125 MHz, DMSO- d 6) d ppm 12.92 47.11 111.42 114.82 116.86 125.02 127.18 127.49 128.01 128.97 129.34 130.29 131.54 141.01 146.30 150.24 152.40; LRMS (MALDI) m / z calcd for C 20 H 19 N 4 [M + H] +: 315.15; Found: 315.43
실시예Example 38: 1-벤질-3-(2-(3- 38: 1-benzyl-3- (2- (3- 메틸methyl -- 1H1H -- 피라졸로[3,4-b]피리딘Pyrazolo [3,4-b] pyridine -5-일)-5 days) 페닐Phenyl )) 우레아Urea
출발물질로서 3-아미노-5-메틸파라졸(15 mg) 및 N-벤질-3-포밀-1H-인돌-1-카복시아마이드(42 mg)를 사용하여 실시예 1과 동일한 과정을 수행하되, 크로마토그래피를 이용한 정제없이 재결정에 의해 원하는 표제 화합물(15 mg, 수율: 28%)을 수득하였다.The same procedure as in Example 1 was carried out using 3-amino-5-methylparazol (15 mg) and N-benzyl-3-formyl- 1H -indole- 1 -carboxamide (42 mg) as starting materials, Recrystallization without purification using chromatography gave the desired title compound (15 mg, yield: 28%).
1H NMR (500 MHz, DMSO-d6) d ppm 2.50 (s, 3 H) 4.21 (d, J=5.62 Hz, 2 H) 6.89 (t, J=5.75 Hz, 1 H) 7.10 (t, J=6.97 Hz, 1 H) 7.15 - 7.29 (m, 6 H) 7.32 (t,J=7.70 Hz, 1 H) 7.56 (s, 1 H) 7.97 (d, J=8.31 Hz, 1 H) 8.15 (s, 1 H) 8.40 (s, 1 H) 13.29 (s, 1 H); 13C NMR (125 MHz, DMSO-d6) d ppm 12.77 13.00 43.40 114.71 122.61 127.23 127.38 127.58 127.91 128.77 128.92 129.12 130.40 138.09 140.79 141.89 149.89 150.09 152.42 156.08; LRMS (MALDI) m/z calcd for C21H20N5O [M+H]+:358.16 ; Found: 358.38 1 H NMR (500 MHz, DMSO-d6) d ppm 2.50 (s, 3H) 4.21 (d, J = 5.62 Hz, 2H) 6.89 (t, J = 5.75 Hz, 1H) 7.10 (t, J = 6.97 Hz, 1 H) 7.15-7.29 (m, 6 H) 7.32 (t, J = 7.70 Hz, 1 H) 7.56 (s, 1 H) 7.97 (d, J = 8.31 Hz, 1 H) 8.15 (s, 1 H) 8.40 (s, 1 H) 13.29 (s, 1 H); 13 C NMR (125 MHz, DMSO- d 6) d ppm 12.77 13.00 43.40 114.71 122.61 127.23 127.38 127.58 127.91 128.77 128.92 129.12 130.40 138.09 140.79 141.89 149.89 150.09 152.42 156.08; LRMS (MALDI) m / z calcd for C 21 H 20 N 5O [M + H] +: 358.16; Found: 358.38
시험예Test Example 1: 세포독성시험( 1: cytotoxicity test cytotoxicitycytotoxicity testtest ))
본 발명의 화합물들에 대한 항암활성을 알아보기 위해 CCK(cell counting kit)-8 분석을 다음과 같이 실시하였다.In order to investigate the anticancer activity of the compounds of the present invention, a cell counting kit (CCK) -8 assay was performed.
1) 세포배양1) Cell Culture
인간 폐 흑색종 세포주(A549, human lung melanoma cells), 인간 자궁 경부암 세포주(HeLa, human cervical carcinoma cells), 인간 배아 신장 세포주(293T, human kidney cells) 및 근육세포주(C2C12, myogenic cells)를 ATCC(American Typ Culture Collection, USA)로부터 입수하였다.Human lung melanoma cells (A549), human cervical carcinoma cells (HeLa), human kidney cells (293T) and myogenic cells (C2C12) are identified as ATCCs (ATCCs). American Typ Culture Collection, USA.
HeLa 세포와 A549 세포의 경우, 열-불활성화된 10%(v/v) 태 소 혈청(Gibco, Invitrogen) 및 1%(v/v) 항생제-항미생물 용액(Gibco, Invitrogen)이 보충된 RPMI 1640 배지(Gibco, Invitrogen)에서 배양하였고, 293T 세포와 C2C12 세포의 경우는, 열-불활성화된 10%(v/v) 태 소 혈청(Gibco, Invitrogen) 및 1%(v/v) 항생제-항미생물 용액(Gibco, Invitrogen)이 보충된 DMEM (Gibco, Invitrogen)에서 배양하였다. For HeLa cells and A549 cells, RPMI supplemented with heat-inactivated 10% (v / v) fetal bovine serum (Gibco, Invitrogen) and 1% (v / v) antibiotic-antimicrobial solution (Gibco, Invitrogen) Cultured in 1640 medium (Gibco, Invitrogen), and for 293T cells and C2C12 cells, heat-inactivated 10% (v / v) fetal calf serum (Gibco, Invitrogen) and 1% (v / v) antibiotics- The cells were cultured in DMEM (Gibco, Invitrogen) supplemented with antimicrobial solution (Gibco, Invitrogen).
이렇게 배양한 세포들을 5% CO2 및 95% 공기로 구성된 가습 분위기하에 37℃에서 유지한 다음 T75 플라스크(Nalge Nunc International, USA)에서 배양하였다.The cells thus cultured were maintained at 37 ° C. under a humidified atmosphere consisting of 5% CO 2 and 95% air and then cultured in a T75 flask (Nalge Nunc International, USA).
2) 암세포 증식억제활성 시험2) Cancer cell proliferation inhibitory activity test
CCK(cell counting kit)-8 분석법(Dojindo, Japan)에 따라 세포 생존력을 시험하였다. 구체적으로, 얻어진 세포를 96-웰 플레이트의 각 웰에 2 x 103 세포의 밀도로 접종하고 이를 24시간 동안 배양한 다음, 실시예 3, 28, 29, 30, 35 및 36 에서 얻어진 본 발명의 화합물들(1, 5, 10 uM)을 처리하였다. 어떠한 화합물도 처리되지 않은 세포를 대조군으로 사용하였다. 72시간 후, CCK-8 용액(10 ㎕, 2-(2-메톡시-4-니트로페닐)-3-(4-니트로페닐)-5-(2,4-디술포페닐)-2H-테트라졸리움, 일가나트륨염)을 각 웰에 부가한 다음 37℃에서 1시간 동안 추가 배양하였다. Bio-Tek 모델 ELx800TM 마이크로플레이트 리더기(Bio-Tek Instruments, Inc., USA)를 이용하여 450 nm에서 각 웰의 흡광도를 측정하였다(630 nm 기준). 각 세포주에 대해 시험을 2회 수행하였다. 세포 생존율(%) 및 증식억제율(%)은 하기 식에 따라 계산되었다:Cell viability was tested according to the CCK (cell counting kit) -8 assay (Dojindo, Japan). Specifically, the obtained cells were inoculated in each well of a 96-well plate at a density of 2 × 10 3 cells and incubated for 24 hours, and then the cells of the present invention obtained in Examples 3, 28, 29, 30, 35, and 36 Compounds (1, 5, 10 uM) were treated. Cells not treated with any compound were used as controls. After 72 hours, CCK-8 solution (10 μl, 2- (2-methoxy-4-nitrophenyl) -3- (4-nitrophenyl) -5- (2,4-disulfophenyl) -2H-tetra Jolyum, monosodium salt) was added to each well and then further incubated at 37 ° C. for 1 hour. The absorbance of each well was measured at 450 nm (based on 630 nm) using a Bio-Tek model ELx800TM microplate reader (Bio-Tek Instruments, Inc., USA). The test was performed twice for each cell line. Cell survival rate (%) and proliferation inhibition rate (%) were calculated according to the following formula:
%세포 생존율 = (시험 샘플에서 평균 흡광도)/(대조군 샘플에서 평균 흡광도) x 100% Cell viability = (average absorbance in test sample) / (average absorbance in control sample) x 100
%증식 억제율 = (1 - 시험 샘플에서 평균 흡광도/대조군 샘플에서 평균 흡광도) x 100% Growth inhibition = (1-average absorbance in test sample / average absorbance in control sample) x 100
결과가 하기 표 1, 도 1, 도 2 및 도 3에 나타내고 있다.The results are shown in Tables 1, 1, 2 and 3 below.
(IC50, uM)HeLa / A549 cell inhibition
(IC 50 , uM)
상기 표 1, 도 1, 도 2 및 도 3에서 보는 바와 같이, 본 발명의 화합물은 암세포에 대해 특이적으로 우수한 저해활성을 나타냄을 알 수 있다.As shown in Table 1, Figure 1, Figure 2 and Figure 3 , it can be seen that the compound of the present invention exhibits a particularly excellent inhibitory activity against cancer cells.
도 1, 도 2 및 도 3은 본 발명의 화합물(1, 5 및 10 uM)이 처리된 인간 폐 흑색종 세포주(A549), 인간 자궁 경부암 세포주(HeLa) 및 인간 배아 신장 세포주(293T) 각각에 대한 세포독성시험 결과를 보여주는 그래프이다 (C01: 실시예 3, E02: 실시예 28, C03: 실시예 29, C02: 실시예 30, C08: 실시예 35, C09: 실시예 36). 1, 2 and 3 show human lung melanoma cell lines (A549), human cervical cancer cell lines (HeLa) and human embryonic
도 4는 본 발명의 화합물(1, 5 및 10 uM)이 처리된 근육 세포주(C2C12)에 대한 세포독성시험 결과를 보여주는 그래프이다 (1: 실시예 3, 2: 실시예 28, 3: 실시예 29). Figure 4 is a graph showing the results of cytotoxicity test for the muscle cell line (C2C12) treated with the compound (1, 5 and 10 uM) of the present invention ( 1 : Example 3, 2 : Example 28, 3 : Example 29).
Claims (7)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020090119049A KR101134194B1 (en) | 2009-12-03 | 2009-12-03 | Method of preparing hetero-biaryl pyridine derivative compounds, and hetero-biaryl pyridine derivative compounds prepared thereby |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020090119049A KR101134194B1 (en) | 2009-12-03 | 2009-12-03 | Method of preparing hetero-biaryl pyridine derivative compounds, and hetero-biaryl pyridine derivative compounds prepared thereby |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20110062351A true KR20110062351A (en) | 2011-06-10 |
| KR101134194B1 KR101134194B1 (en) | 2012-04-09 |
Family
ID=44396550
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020090119049A Active KR101134194B1 (en) | 2009-12-03 | 2009-12-03 | Method of preparing hetero-biaryl pyridine derivative compounds, and hetero-biaryl pyridine derivative compounds prepared thereby |
Country Status (1)
| Country | Link |
|---|---|
| KR (1) | KR101134194B1 (en) |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8791112B2 (en) | 2011-03-30 | 2014-07-29 | Arrien Pharmaceuticals Llc | Substituted 5-(pyrazin-2-yl)-1H-pyrazolo [3, 4-B] pyridine and pyrazolo [3, 4-B] pyridine derivatives as protein kinase inhibitors |
| US9029408B2 (en) | 2008-06-16 | 2015-05-12 | Gtx, Inc. | Compounds for treatment of cancer |
| US9334242B2 (en) | 2008-06-16 | 2016-05-10 | Gtx, Inc. | Compounds for treatment of cancer |
| US9447049B2 (en) | 2010-03-01 | 2016-09-20 | University Of Tennessee Research Foundation | Compounds for treatment of cancer |
| KR20200001859A (en) | 2018-06-28 | 2020-01-07 | 영남대학교 산학협력단 | 2-arylpyridines compound, use thereof and preparation method thereof |
| US10865196B2 (en) | 2008-06-16 | 2020-12-15 | University Of Tennessee Research Foundation | Compounds for treatment of cancer |
| CN113429408A (en) * | 2021-07-07 | 2021-09-24 | 南方科技大学 | Nitrogen-containing heterocyclic compound and preparation method, pharmaceutical composition and application thereof |
| WO2023150793A1 (en) * | 2022-02-07 | 2023-08-10 | Cornell University | Inhibitors of malic enzyme 1 |
| CN120058714A (en) * | 2025-01-14 | 2025-05-30 | 安徽中医药大学 | Indole condensed 3-thiopyrazolo [1,5-a ] pyrimidine compound, synthetic method and application |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2007063225A (en) * | 2005-09-01 | 2007-03-15 | Takeda Chem Ind Ltd | Imidazopyridine compound |
-
2009
- 2009-12-03 KR KR1020090119049A patent/KR101134194B1/en active Active
Cited By (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10865196B2 (en) | 2008-06-16 | 2020-12-15 | University Of Tennessee Research Foundation | Compounds for treatment of cancer |
| US9334242B2 (en) | 2008-06-16 | 2016-05-10 | Gtx, Inc. | Compounds for treatment of cancer |
| US10301285B2 (en) | 2008-06-16 | 2019-05-28 | Gtx, Inc. | Compounds for treatment of cancer |
| US9029408B2 (en) | 2008-06-16 | 2015-05-12 | Gtx, Inc. | Compounds for treatment of cancer |
| US11465987B2 (en) | 2010-03-01 | 2022-10-11 | Oncternal Therapeutics, Inc. | Compounds for treatment of cancer |
| US9447049B2 (en) | 2010-03-01 | 2016-09-20 | University Of Tennessee Research Foundation | Compounds for treatment of cancer |
| US9187473B2 (en) | 2011-03-30 | 2015-11-17 | Arrien Pharmaceuticals Llc | Substituted 5-(pyrazin-2-yl)-1H-pyrazolo [3, 4-b] pyridine and pyrazolo [3, 4-b] pyridine derivatives as protein kinase inhibitors |
| US9669028B2 (en) | 2011-03-30 | 2017-06-06 | Arrien Pharmaceuticals Llc | Substituted 5-(pyrazin-2-yl)-1H-pyrazolo [3, 4-B] pyridine and pyrazolo [3, 4-B] pyridine derivatives as protein kinase inhibitors |
| US8791112B2 (en) | 2011-03-30 | 2014-07-29 | Arrien Pharmaceuticals Llc | Substituted 5-(pyrazin-2-yl)-1H-pyrazolo [3, 4-B] pyridine and pyrazolo [3, 4-B] pyridine derivatives as protein kinase inhibitors |
| US9962382B2 (en) | 2011-03-30 | 2018-05-08 | Arrien Pharmaceuticals Llc | Substituted 5-(pyrazin-2-yl)-1H-pyrazolo [3, 4-b] pyridine and pyrazolo [3, 4-b] pyridine derivatives as protein kinase inhibitors |
| KR20200001859A (en) | 2018-06-28 | 2020-01-07 | 영남대학교 산학협력단 | 2-arylpyridines compound, use thereof and preparation method thereof |
| CN113429408A (en) * | 2021-07-07 | 2021-09-24 | 南方科技大学 | Nitrogen-containing heterocyclic compound and preparation method, pharmaceutical composition and application thereof |
| WO2023150793A1 (en) * | 2022-02-07 | 2023-08-10 | Cornell University | Inhibitors of malic enzyme 1 |
| CN120058714A (en) * | 2025-01-14 | 2025-05-30 | 安徽中医药大学 | Indole condensed 3-thiopyrazolo [1,5-a ] pyrimidine compound, synthetic method and application |
Also Published As
| Publication number | Publication date |
|---|---|
| KR101134194B1 (en) | 2012-04-09 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR101134194B1 (en) | Method of preparing hetero-biaryl pyridine derivative compounds, and hetero-biaryl pyridine derivative compounds prepared thereby | |
| CA2446820C (en) | Novel pyrrole derivatives as pharmaceutical agents | |
| JP2022506887A (en) | Nitrogen-containing condensed heterocyclic SHP2 inhibitor compound, production method and use | |
| Abdel‐Aziz et al. | Facile synthesis and in‐vitro antitumor activity of some pyrazolo [3, 4‐b] pyridines and pyrazolo [1, 5‐a] pyrimidines linked to a thiazolo [3, 2‐a] benzimidazole moiety | |
| ES2877678T3 (en) | Preparation method for a chiral pyrrolopyrimidine compound | |
| AU2006320580A1 (en) | Inhibitors of c-Met and uses thereof | |
| KR102826937B1 (en) | Compounds having Axl and c-Met kinase inhibitory activity and their preparation and application | |
| KR20210136995A (en) | Bruton's Tyrosine Kinase Inhibitors | |
| Al-Kamali et al. | Synthesis and antibacterial activity of some novel thieno [2, 3-c] pyridazines using 3-amino-5-phenyl-2-ethoxycarbonylthieno [2, 3-c] pyridazine as a starting material | |
| EP1295878B1 (en) | Pyrazinone derivatives | |
| AU2007227557A1 (en) | Pyrazolo[1,5-a]pyrimidine derivatives and methods of use thereof | |
| AU2014243257A1 (en) | Pyrazolonaphthyridinone derivatives as METAP2 inhibitors (methionine aminopeptidase type-2) | |
| Fahim et al. | Microwave Assisted Synthesis of Pyrazolo [1, 5-a] pyrimidine, Triazolo [1, 5-a] pyrimidine, Pyrimido [1, 2-a] benzimdazole, Triazolo [5, 1-c][1, 2, 4] triazine and Imidazo [2, 1-c][1, 2, 4] triazine | |
| Wang et al. | Identification of 4-(2-furanyl) pyrimidin-2-amines as Janus kinase 2 inhibitors | |
| Pierce et al. | Synthesis of novel 3, 4-diaryl-5-aminopyrazoles as potential kinase inhibitors | |
| Shaaban et al. | Synthesis of novel pyrazolo [3, 4‐d] pyridazine, pyrido [1, 2‐a] benzimidazole, pyrimido [1, 2‐a] benzimidazole and triazolo [4, 3‐a] pyrimidine derivatives | |
| CN104829609B (en) | Substituted Pyridopyrimidine compound and its preparation method and application | |
| CN117645598A (en) | Azadi-and multi-membered fused ring compound, and pharmaceutical composition and application thereof | |
| CN119552175B (en) | PRMT5-MTA inhibitor | |
| Kato et al. | Synthesis of poly-substituted pyrazolo [1, 5-a] quinolines through one-pot two component cascade reaction | |
| CN114957262A (en) | Preparation method of C-6-arylation deazapurine derivative | |
| CN112778308A (en) | Fused tricyclic derivatives as FGFR4 inhibitors | |
| EP1556381B1 (en) | Pyrazole amides for treating hiv infections | |
| CN114848648A (en) | Application of C-6 arylation deazapurine derivative in preparation of antitumor drugs | |
| Mahmoud et al. | Synthesis of novel pyrimidine and fused pyrimidine derivatives |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A201 | Request for examination | ||
| PA0109 | Patent application |
St.27 status event code: A-0-1-A10-A12-nap-PA0109 |
|
| PA0201 | Request for examination |
St.27 status event code: A-1-2-D10-D11-exm-PA0201 |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| PG1501 | Laying open of application |
St.27 status event code: A-1-1-Q10-Q12-nap-PG1501 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-3-3-R10-R13-asn-PN2301 St.27 status event code: A-3-3-R10-R11-asn-PN2301 |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
St.27 status event code: A-1-2-D10-D21-exm-PE0902 |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| R17-X000 | Change to representative recorded |
St.27 status event code: A-3-3-R10-R17-oth-X000 |
|
| E701 | Decision to grant or registration of patent right | ||
| PE0701 | Decision of registration |
St.27 status event code: A-1-2-D10-D22-exm-PE0701 |
|
| GRNT | Written decision to grant | ||
| PR0701 | Registration of establishment |
St.27 status event code: A-2-4-F10-F11-exm-PR0701 |
|
| PR1002 | Payment of registration fee |
St.27 status event code: A-2-2-U10-U11-oth-PR1002 Fee payment year number: 1 |
|
| PG1601 | Publication of registration |
St.27 status event code: A-4-4-Q10-Q13-nap-PG1601 |
|
| P14-X000 | Amendment of ip right document requested |
St.27 status event code: A-5-5-P10-P14-nap-X000 |
|
| P16-X000 | Ip right document amended |
St.27 status event code: A-5-5-P10-P16-nap-X000 |
|
| Q16-X000 | A copy of ip right certificate issued |
St.27 status event code: A-4-4-Q10-Q16-nap-X000 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |
|
| FPAY | Annual fee payment |
Payment date: 20150212 Year of fee payment: 4 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 4 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-5-5-R10-R13-asn-PN2301 St.27 status event code: A-5-5-R10-R11-asn-PN2301 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-5-5-R10-R13-asn-PN2301 St.27 status event code: A-5-5-R10-R11-asn-PN2301 |
|
| FPAY | Annual fee payment |
Payment date: 20160122 Year of fee payment: 5 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 5 |
|
| P22-X000 | Classification modified |
St.27 status event code: A-4-4-P10-P22-nap-X000 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-5-5-R10-R11-asn-PN2301 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-5-5-R10-R14-asn-PN2301 |
|
| FPAY | Annual fee payment |
Payment date: 20170220 Year of fee payment: 6 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 6 |
|
| FPAY | Annual fee payment |
Payment date: 20180323 Year of fee payment: 7 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 7 |
|
| P14-X000 | Amendment of ip right document requested |
St.27 status event code: A-5-5-P10-P14-nap-X000 |
|
| P16-X000 | Ip right document amended |
St.27 status event code: A-5-5-P10-P16-nap-X000 |
|
| Q16-X000 | A copy of ip right certificate issued |
St.27 status event code: A-4-4-Q10-Q16-nap-X000 |
|
| P15-X000 | Request for amendment of ip right document rejected |
St.27 status event code: A-5-5-P10-P15-nap-X000 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 8 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-5-5-R10-R13-asn-PN2301 St.27 status event code: A-5-5-R10-R11-asn-PN2301 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 9 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-5-5-R10-R13-asn-PN2301 St.27 status event code: A-5-5-R10-R11-asn-PN2301 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 10 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 11 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 12 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 13 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 14 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |
|
| R18 | Changes to party contact information recorded |
Free format text: ST27 STATUS EVENT CODE: A-5-5-R10-R18-OTH-X000 (AS PROVIDED BY THE NATIONAL OFFICE) |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |