KR20080106506A - 만성 신장병의 치료 방법 - Google Patents
만성 신장병의 치료 방법 Download PDFInfo
- Publication number
- KR20080106506A KR20080106506A KR1020087018633A KR20087018633A KR20080106506A KR 20080106506 A KR20080106506 A KR 20080106506A KR 1020087018633 A KR1020087018633 A KR 1020087018633A KR 20087018633 A KR20087018633 A KR 20087018633A KR 20080106506 A KR20080106506 A KR 20080106506A
- Authority
- KR
- South Korea
- Prior art keywords
- kidney disease
- chronic kidney
- ferric
- organic compound
- patients
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 208000020832 chronic kidney disease Diseases 0.000 title claims abstract description 125
- 150000002894 organic compounds Chemical class 0.000 claims abstract description 108
- NPFOYSMITVOQOS-UHFFFAOYSA-K iron(III) citrate Chemical compound [Fe+3].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O NPFOYSMITVOQOS-UHFFFAOYSA-K 0.000 claims abstract description 94
- 229960002413 ferric citrate Drugs 0.000 claims abstract description 91
- 238000004090 dissolution Methods 0.000 claims abstract description 37
- 238000000034 method Methods 0.000 claims description 71
- 210000002966 serum Anatomy 0.000 claims description 48
- 238000011282 treatment Methods 0.000 claims description 40
- 239000002775 capsule Substances 0.000 claims description 38
- DDRJAANPRJIHGJ-UHFFFAOYSA-N creatinine Chemical compound CN1CC(=O)NC1=N DDRJAANPRJIHGJ-UHFFFAOYSA-N 0.000 claims description 36
- 150000007524 organic acids Chemical class 0.000 claims description 36
- 239000002244 precipitate Substances 0.000 claims description 31
- 239000003814 drug Substances 0.000 claims description 27
- 239000000203 mixture Substances 0.000 claims description 24
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical group [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 claims description 23
- 238000000502 dialysis Methods 0.000 claims description 20
- 239000011575 calcium Substances 0.000 claims description 19
- 210000003734 kidney Anatomy 0.000 claims description 19
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 claims description 18
- 229940109239 creatinine Drugs 0.000 claims description 18
- 239000008194 pharmaceutical composition Substances 0.000 claims description 18
- 208000017169 kidney disease Diseases 0.000 claims description 17
- 239000000725 suspension Substances 0.000 claims description 16
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 claims description 15
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 claims description 15
- 229910052791 calcium Inorganic materials 0.000 claims description 15
- 239000003960 organic solvent Substances 0.000 claims description 15
- 239000011574 phosphorus Substances 0.000 claims description 15
- 229910052698 phosphorus Inorganic materials 0.000 claims description 15
- -1 troche Substances 0.000 claims description 15
- 208000010444 Acidosis Diseases 0.000 claims description 13
- 150000003839 salts Chemical class 0.000 claims description 13
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 claims description 12
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 claims description 12
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 12
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 claims description 12
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 claims description 12
- 229910000000 metal hydroxide Inorganic materials 0.000 claims description 12
- 150000004692 metal hydroxides Chemical class 0.000 claims description 12
- 206010027417 Metabolic acidosis Diseases 0.000 claims description 11
- 239000001506 calcium phosphate Substances 0.000 claims description 11
- 229910000389 calcium phosphate Inorganic materials 0.000 claims description 11
- 235000011010 calcium phosphates Nutrition 0.000 claims description 11
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 claims description 11
- 230000015572 biosynthetic process Effects 0.000 claims description 10
- 239000000843 powder Substances 0.000 claims description 10
- 238000001556 precipitation Methods 0.000 claims description 10
- 239000003826 tablet Substances 0.000 claims description 10
- VTLYFUHAOXGGBS-UHFFFAOYSA-N Fe3+ Chemical class [Fe+3] VTLYFUHAOXGGBS-UHFFFAOYSA-N 0.000 claims description 9
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 claims description 9
- 238000011161 development Methods 0.000 claims description 9
- 239000007788 liquid Substances 0.000 claims description 9
- 238000004519 manufacturing process Methods 0.000 claims description 9
- 239000000047 product Substances 0.000 claims description 9
- 239000006188 syrup Substances 0.000 claims description 9
- 235000020357 syrup Nutrition 0.000 claims description 9
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 claims description 8
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 claims description 8
- 239000000839 emulsion Substances 0.000 claims description 8
- 239000008187 granular material Substances 0.000 claims description 8
- 239000006187 pill Substances 0.000 claims description 8
- 241001465754 Metazoa Species 0.000 claims description 7
- 239000007937 lozenge Substances 0.000 claims description 7
- 235000015096 spirit Nutrition 0.000 claims description 7
- 239000000126 substance Substances 0.000 claims description 7
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 claims description 6
- 208000000913 Kidney Calculi Diseases 0.000 claims description 6
- 206010029148 Nephrolithiasis Diseases 0.000 claims description 6
- 208000020084 Bone disease Diseases 0.000 claims description 5
- 201000002980 Hyperparathyroidism Diseases 0.000 claims description 5
- 229940044631 ferric chloride hexahydrate Drugs 0.000 claims description 5
- NQXWGWZJXJUMQB-UHFFFAOYSA-K iron trichloride hexahydrate Chemical group O.O.O.O.O.O.[Cl-].Cl[Fe+]Cl NQXWGWZJXJUMQB-UHFFFAOYSA-K 0.000 claims description 5
- 238000010438 heat treatment Methods 0.000 claims description 4
- 230000002829 reductive effect Effects 0.000 claims description 4
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 claims description 4
- ODBLHEXUDAPZAU-ZAFYKAAXSA-N D-threo-isocitric acid Chemical compound OC(=O)[C@H](O)[C@@H](C(O)=O)CC(O)=O ODBLHEXUDAPZAU-ZAFYKAAXSA-N 0.000 claims description 3
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 claims description 3
- ODBLHEXUDAPZAU-FONMRSAGSA-N Isocitric acid Natural products OC(=O)[C@@H](O)[C@H](C(O)=O)CC(O)=O ODBLHEXUDAPZAU-FONMRSAGSA-N 0.000 claims description 3
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 claims description 3
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 claims description 3
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical compound O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 claims description 3
- 239000001530 fumaric acid Substances 0.000 claims description 3
- 235000002906 tartaric acid Nutrition 0.000 claims description 3
- 239000011975 tartaric acid Substances 0.000 claims description 3
- ODBLHEXUDAPZAU-UHFFFAOYSA-N threo-D-isocitric acid Natural products OC(=O)C(O)C(C(O)=O)CC(O)=O ODBLHEXUDAPZAU-UHFFFAOYSA-N 0.000 claims description 3
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 claims description 3
- 238000001816 cooling Methods 0.000 claims description 2
- 208000024172 Cardiovascular disease Diseases 0.000 claims 2
- 238000012423 maintenance Methods 0.000 claims 2
- 230000002265 prevention Effects 0.000 claims 2
- 229910019142 PO4 Inorganic materials 0.000 description 39
- 239000010452 phosphate Substances 0.000 description 37
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 37
- 235000021317 phosphate Nutrition 0.000 description 34
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 25
- 229940079593 drug Drugs 0.000 description 23
- 239000000902 placebo Substances 0.000 description 20
- 229940068196 placebo Drugs 0.000 description 20
- 201000000523 end stage renal failure Diseases 0.000 description 15
- 208000028208 end stage renal disease Diseases 0.000 description 13
- 229910052742 iron Inorganic materials 0.000 description 13
- 235000005911 diet Nutrition 0.000 description 12
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 10
- 230000003907 kidney function Effects 0.000 description 9
- 230000003111 delayed effect Effects 0.000 description 8
- 230000000378 dietary effect Effects 0.000 description 8
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 8
- 230000000694 effects Effects 0.000 description 8
- 238000011156 evaluation Methods 0.000 description 8
- 201000005991 hyperphosphatemia Diseases 0.000 description 8
- 235000014413 iron hydroxide Nutrition 0.000 description 8
- NCNCGGDMXMBVIA-UHFFFAOYSA-L iron(ii) hydroxide Chemical compound [OH-].[OH-].[Fe+2] NCNCGGDMXMBVIA-UHFFFAOYSA-L 0.000 description 8
- 150000001875 compounds Chemical class 0.000 description 7
- 201000010099 disease Diseases 0.000 description 7
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 6
- 210000001035 gastrointestinal tract Anatomy 0.000 description 6
- 230000024924 glomerular filtration Effects 0.000 description 6
- 238000012216 screening Methods 0.000 description 6
- BVKZGUZCCUSVTD-UHFFFAOYSA-M Bicarbonate Chemical compound OC([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-M 0.000 description 5
- 102000008857 Ferritin Human genes 0.000 description 5
- 108050000784 Ferritin Proteins 0.000 description 5
- 238000008416 Ferritin Methods 0.000 description 5
- 102000008133 Iron-Binding Proteins Human genes 0.000 description 5
- 108010035210 Iron-Binding Proteins Proteins 0.000 description 5
- 208000001647 Renal Insufficiency Diseases 0.000 description 5
- 102000004338 Transferrin Human genes 0.000 description 5
- 108090000901 Transferrin Proteins 0.000 description 5
- 229910021529 ammonia Inorganic materials 0.000 description 5
- 230000006378 damage Effects 0.000 description 5
- 238000001631 haemodialysis Methods 0.000 description 5
- 230000000322 hemodialysis Effects 0.000 description 5
- 201000006370 kidney failure Diseases 0.000 description 5
- 230000000750 progressive effect Effects 0.000 description 5
- 239000006228 supernatant Substances 0.000 description 5
- 238000003786 synthesis reaction Methods 0.000 description 5
- 239000012581 transferrin Substances 0.000 description 5
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 4
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 4
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 4
- 102000003929 Transaminases Human genes 0.000 description 4
- 108090000340 Transaminases Proteins 0.000 description 4
- 230000002411 adverse Effects 0.000 description 4
- 230000008901 benefit Effects 0.000 description 4
- 235000021152 breakfast Nutrition 0.000 description 4
- CVSVTCORWBXHQV-UHFFFAOYSA-N creatine Chemical compound NC(=[NH2+])N(C)CC([O-])=O CVSVTCORWBXHQV-UHFFFAOYSA-N 0.000 description 4
- 230000037213 diet Effects 0.000 description 4
- 208000015181 infectious disease Diseases 0.000 description 4
- 239000011777 magnesium Substances 0.000 description 4
- 229910052749 magnesium Inorganic materials 0.000 description 4
- 235000012054 meals Nutrition 0.000 description 4
- 239000011591 potassium Substances 0.000 description 4
- 229910052700 potassium Inorganic materials 0.000 description 4
- 230000008569 process Effects 0.000 description 4
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 3
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 3
- 238000008214 LDL Cholesterol Methods 0.000 description 3
- 102000003982 Parathyroid hormone Human genes 0.000 description 3
- 108090000445 Parathyroid hormone Proteins 0.000 description 3
- 102000007562 Serum Albumin Human genes 0.000 description 3
- 108010071390 Serum Albumin Proteins 0.000 description 3
- 238000008050 Total Bilirubin Reagent Methods 0.000 description 3
- 230000009102 absorption Effects 0.000 description 3
- 238000010521 absorption reaction Methods 0.000 description 3
- 150000008044 alkali metal hydroxides Chemical class 0.000 description 3
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 3
- 210000004369 blood Anatomy 0.000 description 3
- 239000008280 blood Substances 0.000 description 3
- 238000004820 blood count Methods 0.000 description 3
- 210000000988 bone and bone Anatomy 0.000 description 3
- 230000008859 change Effects 0.000 description 3
- 230000001684 chronic effect Effects 0.000 description 3
- 230000003247 decreasing effect Effects 0.000 description 3
- 238000001035 drying Methods 0.000 description 3
- 238000009472 formulation Methods 0.000 description 3
- 239000007789 gas Substances 0.000 description 3
- 239000008103 glucose Substances 0.000 description 3
- WBJZTOZJJYAKHQ-UHFFFAOYSA-K iron(3+) phosphate Chemical compound [Fe+3].[O-]P([O-])([O-])=O WBJZTOZJJYAKHQ-UHFFFAOYSA-K 0.000 description 3
- 239000000199 parathyroid hormone Substances 0.000 description 3
- 229960001319 parathyroid hormone Drugs 0.000 description 3
- 239000002245 particle Substances 0.000 description 3
- 230000001376 precipitating effect Effects 0.000 description 3
- 102000004169 proteins and genes Human genes 0.000 description 3
- 108090000623 proteins and genes Proteins 0.000 description 3
- 210000005084 renal tissue Anatomy 0.000 description 3
- 239000011734 sodium Substances 0.000 description 3
- 229910052708 sodium Inorganic materials 0.000 description 3
- 239000007787 solid Substances 0.000 description 3
- 239000002904 solvent Substances 0.000 description 3
- 208000024891 symptom Diseases 0.000 description 3
- 230000002194 synthesizing effect Effects 0.000 description 3
- 238000012360 testing method Methods 0.000 description 3
- 238000002560 therapeutic procedure Methods 0.000 description 3
- 210000005239 tubule Anatomy 0.000 description 3
- 210000002700 urine Anatomy 0.000 description 3
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 3
- 239000005541 ACE inhibitor Substances 0.000 description 2
- 102000002260 Alkaline Phosphatase Human genes 0.000 description 2
- 108020004774 Alkaline Phosphatase Proteins 0.000 description 2
- 101710129690 Angiotensin-converting enzyme inhibitor Proteins 0.000 description 2
- 101710086378 Bradykinin-potentiating and C-type natriuretic peptides Proteins 0.000 description 2
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 2
- 208000031229 Cardiomyopathies Diseases 0.000 description 2
- 239000005955 Ferric phosphate Substances 0.000 description 2
- 208000021908 Myocardial disease Diseases 0.000 description 2
- TXXIWPTXQBKYOE-UHFFFAOYSA-N OC(=O)CC(CC(O)=O)(OP(=O)=O)C(O)=O Chemical compound OC(=O)CC(CC(O)=O)(OP(=O)=O)C(O)=O TXXIWPTXQBKYOE-UHFFFAOYSA-N 0.000 description 2
- 206010038378 Renal artery stenosis Diseases 0.000 description 2
- 206010061481 Renal injury Diseases 0.000 description 2
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 2
- PNNCWTXUWKENPE-UHFFFAOYSA-N [N].NC(N)=O Chemical compound [N].NC(N)=O PNNCWTXUWKENPE-UHFFFAOYSA-N 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 230000007950 acidosis Effects 0.000 description 2
- 208000026545 acidosis disease Diseases 0.000 description 2
- 230000009471 action Effects 0.000 description 2
- BWZOPYPOZJBVLQ-UHFFFAOYSA-K aluminium glycinate Chemical compound O[Al+]O.NCC([O-])=O BWZOPYPOZJBVLQ-UHFFFAOYSA-K 0.000 description 2
- 229940125364 angiotensin receptor blocker Drugs 0.000 description 2
- 229940044094 angiotensin-converting-enzyme inhibitor Drugs 0.000 description 2
- 239000002585 base Substances 0.000 description 2
- 239000011230 binding agent Substances 0.000 description 2
- 230000002308 calcification Effects 0.000 description 2
- 238000006243 chemical reaction Methods 0.000 description 2
- 208000022831 chronic renal failure syndrome Diseases 0.000 description 2
- 229960003624 creatine Drugs 0.000 description 2
- 239000006046 creatine Substances 0.000 description 2
- 238000013523 data management Methods 0.000 description 2
- 206010012601 diabetes mellitus Diseases 0.000 description 2
- 239000012153 distilled water Substances 0.000 description 2
- 239000002552 dosage form Substances 0.000 description 2
- 239000003937 drug carrier Substances 0.000 description 2
- 229940032958 ferric phosphate Drugs 0.000 description 2
- 238000007429 general method Methods 0.000 description 2
- 230000036541 health Effects 0.000 description 2
- 239000012535 impurity Substances 0.000 description 2
- 210000000936 intestine Anatomy 0.000 description 2
- 229910000399 iron(III) phosphate Inorganic materials 0.000 description 2
- 210000000231 kidney cortex Anatomy 0.000 description 2
- 208000037806 kidney injury Diseases 0.000 description 2
- 230000000670 limiting effect Effects 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- 235000005985 organic acids Nutrition 0.000 description 2
- 239000012071 phase Substances 0.000 description 2
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 2
- 239000011148 porous material Substances 0.000 description 2
- 230000022558 protein metabolic process Effects 0.000 description 2
- 201000001474 proteinuria Diseases 0.000 description 2
- 230000008085 renal dysfunction Effects 0.000 description 2
- 230000002441 reversible effect Effects 0.000 description 2
- 230000028327 secretion Effects 0.000 description 2
- 239000002356 single layer Substances 0.000 description 2
- 239000007858 starting material Substances 0.000 description 2
- 238000011285 therapeutic regimen Methods 0.000 description 2
- 210000000626 ureter Anatomy 0.000 description 2
- 230000002485 urinary effect Effects 0.000 description 2
- 208000009304 Acute Kidney Injury Diseases 0.000 description 1
- 208000037157 Azotemia Diseases 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- 208000030453 Drug-Related Side Effects and Adverse reaction Diseases 0.000 description 1
- 102000003951 Erythropoietin Human genes 0.000 description 1
- 108090000394 Erythropoietin Proteins 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- 206010020772 Hypertension Diseases 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- 206010022489 Insulin Resistance Diseases 0.000 description 1
- 239000002841 Lewis acid Substances 0.000 description 1
- 208000029725 Metabolic bone disease Diseases 0.000 description 1
- 206010049088 Osteopenia Diseases 0.000 description 1
- 208000004531 Renal Artery Obstruction Diseases 0.000 description 1
- 208000033626 Renal failure acute Diseases 0.000 description 1
- 206010062237 Renal impairment Diseases 0.000 description 1
- 208000000223 Solitary Kidney Diseases 0.000 description 1
- 208000025865 Ulcer Diseases 0.000 description 1
- LEHOTFFKMJEONL-UHFFFAOYSA-N Uric Acid Chemical compound N1C(=O)NC(=O)C2=C1NC(=O)N2 LEHOTFFKMJEONL-UHFFFAOYSA-N 0.000 description 1
- 229930003316 Vitamin D Natural products 0.000 description 1
- QYSXJUFSXHHAJI-XFEUOLMDSA-N Vitamin D3 Natural products C1(/[C@@H]2CC[C@@H]([C@]2(CCC1)C)[C@H](C)CCCC(C)C)=C/C=C1\C[C@@H](O)CCC1=C QYSXJUFSXHHAJI-XFEUOLMDSA-N 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- 230000004913 activation Effects 0.000 description 1
- 239000008186 active pharmaceutical agent Substances 0.000 description 1
- 201000011040 acute kidney failure Diseases 0.000 description 1
- 208000012998 acute renal failure Diseases 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 208000007502 anemia Diseases 0.000 description 1
- 238000010171 animal model Methods 0.000 description 1
- 229940069428 antacid Drugs 0.000 description 1
- 239000003159 antacid agent Substances 0.000 description 1
- 239000002220 antihypertensive agent Substances 0.000 description 1
- 229940030600 antihypertensive agent Drugs 0.000 description 1
- 239000000090 biomarker Substances 0.000 description 1
- 230000037396 body weight Effects 0.000 description 1
- GMRQFYUYWCNGIN-NKMMMXOESA-N calcitriol Chemical compound C1(/[C@@H]2CC[C@@H]([C@]2(CCC1)C)[C@@H](CCCC(C)(C)O)C)=C\C=C1\C[C@@H](O)C[C@H](O)C1=C GMRQFYUYWCNGIN-NKMMMXOESA-N 0.000 description 1
- 235000020964 calcitriol Nutrition 0.000 description 1
- 239000011612 calcitriol Substances 0.000 description 1
- 229960005084 calcitriol Drugs 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 238000003759 clinical diagnosis Methods 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 230000000295 complement effect Effects 0.000 description 1
- 238000007405 data analysis Methods 0.000 description 1
- 230000007850 degeneration Effects 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 230000006866 deterioration Effects 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- 208000035475 disorder Diseases 0.000 description 1
- 239000012738 dissolution medium Substances 0.000 description 1
- 229940088679 drug related substance Drugs 0.000 description 1
- 239000003792 electrolyte Substances 0.000 description 1
- 229940105423 erythropoietin Drugs 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 229910001447 ferric ion Inorganic materials 0.000 description 1
- 229940116007 ferrous phosphate Drugs 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 239000012458 free base Substances 0.000 description 1
- 230000002496 gastric effect Effects 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 230000036571 hydration Effects 0.000 description 1
- 238000006703 hydration reaction Methods 0.000 description 1
- 230000006698 induction Effects 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 230000000968 intestinal effect Effects 0.000 description 1
- 238000007912 intraperitoneal administration Methods 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- 229910000155 iron(II) phosphate Inorganic materials 0.000 description 1
- JEIPFZHSYJVQDO-UHFFFAOYSA-N iron(III) oxide Inorganic materials O=[Fe]O[Fe]=O JEIPFZHSYJVQDO-UHFFFAOYSA-N 0.000 description 1
- SDEKDNPYZOERBP-UHFFFAOYSA-H iron(ii) phosphate Chemical compound [Fe+2].[Fe+2].[Fe+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O SDEKDNPYZOERBP-UHFFFAOYSA-H 0.000 description 1
- 238000009533 lab test Methods 0.000 description 1
- 239000010410 layer Substances 0.000 description 1
- 150000007517 lewis acids Chemical class 0.000 description 1
- 231100000053 low toxicity Toxicity 0.000 description 1
- 235000020905 low-protein-diet Nutrition 0.000 description 1
- 230000002101 lytic effect Effects 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 238000002483 medication Methods 0.000 description 1
- 239000002207 metabolite Substances 0.000 description 1
- 238000003801 milling Methods 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 235000010755 mineral Nutrition 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012544 monitoring process Methods 0.000 description 1
- 239000004570 mortar (masonry) Substances 0.000 description 1
- 230000003589 nefrotoxic effect Effects 0.000 description 1
- 239000000041 non-steroidal anti-inflammatory agent Substances 0.000 description 1
- 229940021182 non-steroidal anti-inflammatory drug Drugs 0.000 description 1
- 239000006186 oral dosage form Substances 0.000 description 1
- 210000002990 parathyroid gland Anatomy 0.000 description 1
- 230000007170 pathology Effects 0.000 description 1
- 239000002694 phosphate binding agent Substances 0.000 description 1
- 230000004962 physiological condition Effects 0.000 description 1
- 231100000857 poor renal function Toxicity 0.000 description 1
- OXCMYAYHXIHQOA-UHFFFAOYSA-N potassium;[2-butyl-5-chloro-3-[[4-[2-(1,2,4-triaza-3-azanidacyclopenta-1,4-dien-5-yl)phenyl]phenyl]methyl]imidazol-4-yl]methanol Chemical compound [K+].CCCCC1=NC(Cl)=C(CO)N1CC1=CC=C(C=2C(=CC=CC=2)C2=N[N-]N=N2)C=C1 OXCMYAYHXIHQOA-UHFFFAOYSA-N 0.000 description 1
- 230000003449 preventive effect Effects 0.000 description 1
- 230000009103 reabsorption Effects 0.000 description 1
- 238000011084 recovery Methods 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 210000000813 small intestine Anatomy 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- 235000017557 sodium bicarbonate Nutrition 0.000 description 1
- 239000007790 solid phase Substances 0.000 description 1
- 238000007614 solvation Methods 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 239000013589 supplement Substances 0.000 description 1
- 208000011580 syndromic disease Diseases 0.000 description 1
- 238000001308 synthesis method Methods 0.000 description 1
- 230000001225 therapeutic effect Effects 0.000 description 1
- 238000004448 titration Methods 0.000 description 1
- 238000002054 transplantation Methods 0.000 description 1
- 238000011269 treatment regimen Methods 0.000 description 1
- 208000001072 type 2 diabetes mellitus Diseases 0.000 description 1
- 208000009852 uremia Diseases 0.000 description 1
- 210000001635 urinary tract Anatomy 0.000 description 1
- 235000019166 vitamin D Nutrition 0.000 description 1
- 239000011710 vitamin D Substances 0.000 description 1
- 150000003710 vitamin D derivatives Chemical class 0.000 description 1
- 229940046008 vitamin d Drugs 0.000 description 1
- 239000002699 waste material Substances 0.000 description 1
- 230000004580 weight loss Effects 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/28—Compounds containing heavy metals
- A61K31/295—Iron group metal compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0053—Mouth and digestive tract, i.e. intraoral and peroral administration
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2121/00—Preparations for use in therapy
Landscapes
- Health & Medical Sciences (AREA)
- Epidemiology (AREA)
- Animal Behavior & Ethology (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Veterinary Medicine (AREA)
- Life Sciences & Earth Sciences (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Physiology (AREA)
- Nutrition Science (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Medicinal Preparation (AREA)
Abstract
Description
Claims (45)
- 2 mg/cm2/분 이상의 용해 속도를 갖는 제2철 유기 화합물 유효량을 환자에게 투여하는 것을 포함하는만성 신장병 환자의 치료 방법.
- 제1항에 있어서,상기 제2철 유기 화합물의 용해 속도가 2.5 mg/cm2/분 내지 3 mg/cm2/분인만성 신장병 환자의 치료 방법.
- 제1항에 있어서,상기 제2철 유기 화합물의 용해 속도가 3.0 mg/cm2/분 내지 3.5 mg/cm2/분인만성 신장병 환자의 치료 방법.
- 제1항에 있어서,상기 제2철 유기 화합물의 용해 속도가 3.5 mg/cm2/분 내지 4.0 mg/cm2/분인만성 신장병 환자의 치료 방법.
- 제1항 내지 제4항 중 어느 한 항에 있어서,상기 제2철 유기 화합물은(a) 제2철(ferric iron) 염을 얻는 단계;(b) 다중철 산화물을 포함하는 혼합물을 생성하기 위해 효과적인 조건하에서 상기 제2철 염에 알칼리성 금속 수산화물을 부가하는 단계;(c) 상기 혼합물로부터 석출물을 단리하는 단계;(d) 유기산을 상기 석출물에 부가하는 단계;(e) 상기 유기산 및 상기 석출물을 가열함으로써 제2철 유기 산 용액을 형성하는 단계; 및(f) 상기 제2철 유기산 용액으로부터 유기 용매에 의해 제2철 유기 화합물을 석출하는 단계를 포함하는 방법에 따라 제조되는만성 신장병 환자의 치료 방법.
- 제5항에 있어서,상기 알칼리성 금속 수산화물이 수산화 나트륨 또는 수산화 칼륨인만성 신장병 환자의 치료 방법.
- 제5항 또는 제6항에 있어서,상기 알칼리성 금속 수산화물은 20ml/분 미만의 속도로 부가되며, 또 상기 알칼리성 금속 수산화물은 40℃ 미만의 온도에서 상기 제2철 염에 부가되는만성 신장병 환자의 치료 방법.
- 제5항 내지 제7항 중 어느 한 항에 있어서,상기 유기산 및 석출물은 80℃ 내지 90℃ 사이의 온도로 가열되고, 또 상기 유기 용매에 의해 상기 제2철 유기산 용액으로부터 상기 제2철 유기 화합물을 석출하는 것은 상기 유기 용매를 부가하기 전에 30℃ 미만으로 상기 제2철 유기산 용액을 냉각하는 것을 포함하는만성 신장병 환자의 치료 방법.
- 제 5항 내지 제 8항 중 어느 한 항에 있어서,상기 유기산이 결정 형태인만성 신장병 환자의 치료 방법.
- 제 5항 내지 제 9항 중 어느 한 항에 있어서,상기 유기산이 시트르산, 아세트산, 이소시트르산, 숙신산, 푸마르산 및 타르타르산으로 구성된 군으로부터 선택되는만성 신장병 환자의 치료 방법.
- 제 5항 내지 제 10항 중 어느 한 항에 있어서,상기 유기 용매는 에탄올, 메탄올, 부탄올, 이소프로필 알코올, 아세톤 및 테트라히드로푸란으로 구성된 군으로부터 선택되는만성 신장병 환자의 치료 방법.
- 제 5항 내지 제 11항 중 어느 한 항에 있어서,상기 제2철 염은 염화제2철 육수화물이고, 상기 알칼리성 금속 수산화물은 수산화 나트륨이며 또 상기 유기산은 결정성 시트르산인만성 신장병 환자의 치료 방법.
- 제1항 내지 제12항 중 어느 한 항에 있어서,상기 환자는 사람 또는 동물인만성 신장병 환자의 치료 방법.
- 제1항 내지 제13항 중 어느 한 항에 있어서,상기 만성 신장병은 만성 신장병의 임의 단계 및 말기 신장병을 포함하는만성 신장병 환자의 치료 방법.
- 제1항 내지 제14항 중 어느 한 항에 있어서,상기 환자는 신장 투석을 받는만성 신장병 환자의 치료 방법.
- 제1항 내지 제15항 중 어느 한 항에 있어서,상기 제2철 유기 화합물이 2-20 gm/일의 투여량으로 투여되는만성 신장병 환자의 치료 방법.
- 제1항 내지 제16항 중 어느 한 항에 있어서,상기 제2철 유기 화합물이 경구적으로 또는 기타 적합한 경로를 통하여 투여되는만성 신장병 환자의 치료 방법.
- 제1항 내지 제17항 중 어느 한 항에 있어서,상기 제2철 유기 화합물이 정제, 분말, 현탁액, 에멀젼, 캡슐, 로젠지, 과립, 트로체, 환약, 액체, 스피리트 또는 시럽인만성 신장병 환자의 치료 방법.
- 제1항 내지 제18항 중 어느 한 항에 있어서,상기 제2철 유기 화합물을 사용한 치료는 상기 환자에서 크레아티닌, BUN, 인 및 칼슘과 인 생성물(CaxP)로 구성된 군으로부터 선택되는 1 이상의 물질의 감소된 혈청 수준을 초래하는만성 신장병 환자의 치료 방법.
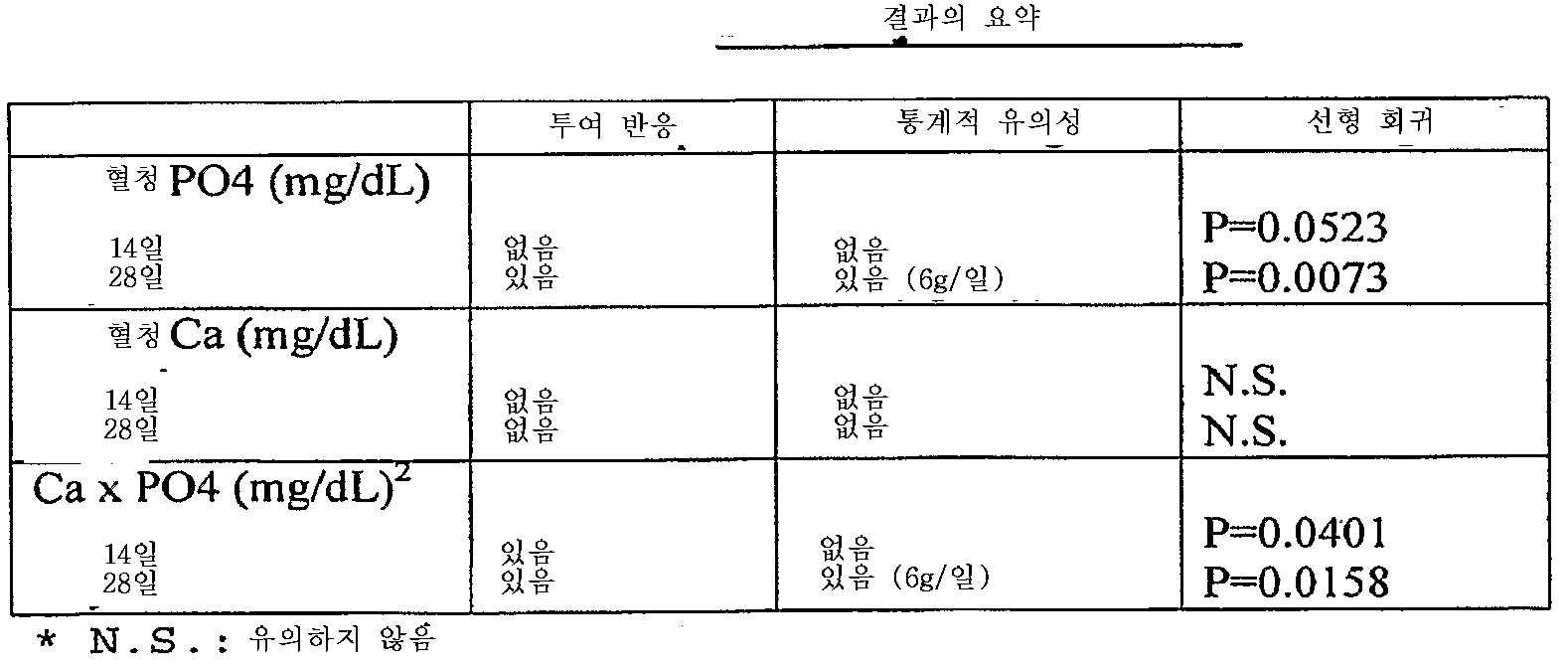
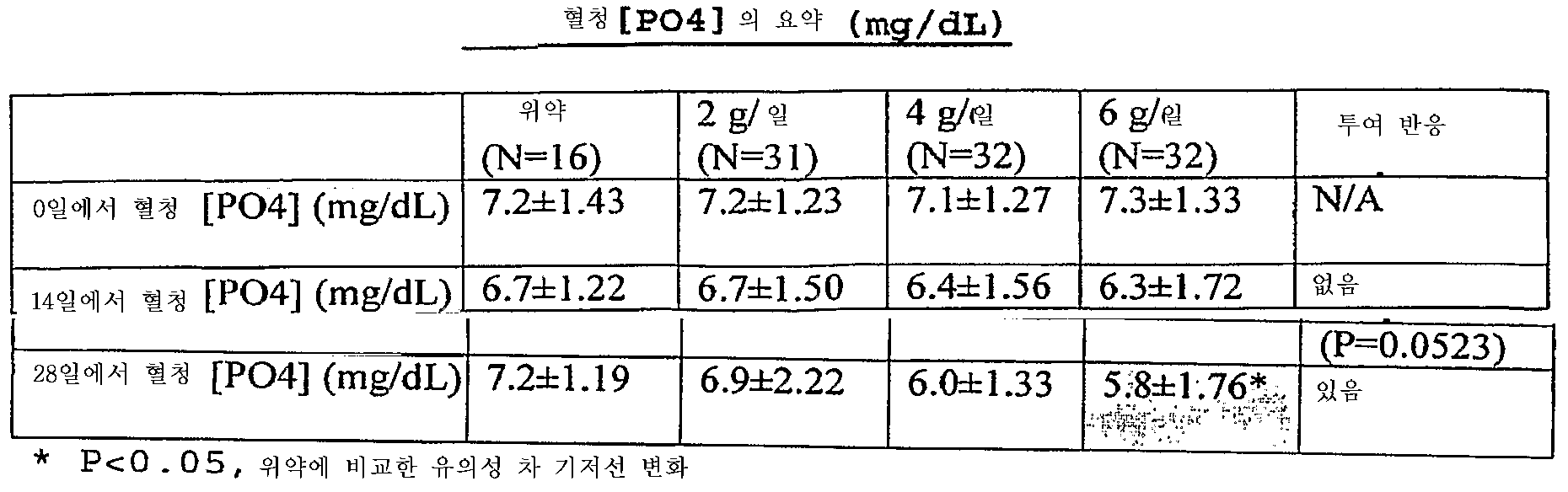
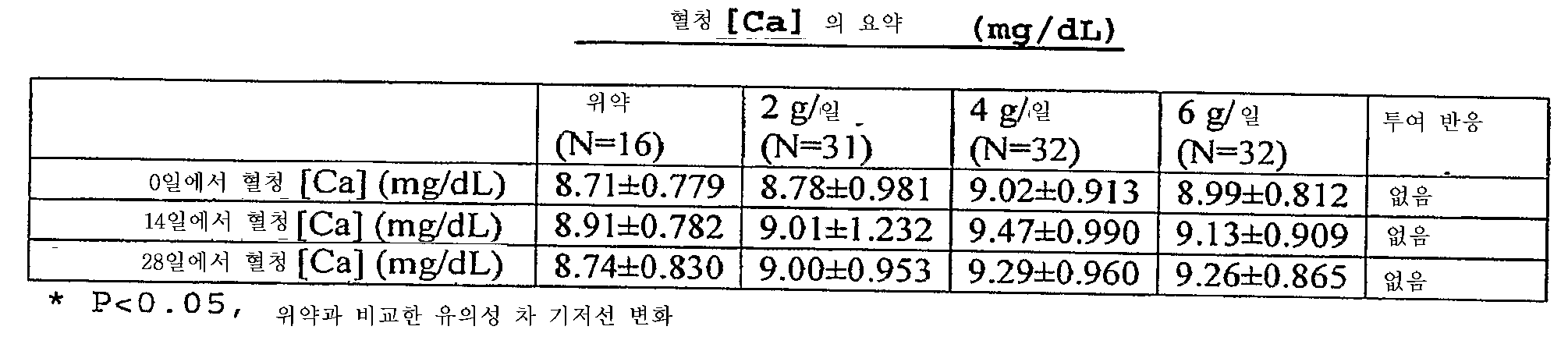
- 제1항 내지 제18항 중 어느 한 항에 있어서,상기 제2철 유기 화합물을 사용한 치료는 만성 신장병의 진행, 부갑상선기능항진증의 발생, 뼈 질병의 발생, 심혈관 질병의 발생, 신장병에서 칼슘 포스페이트 석출, 신장 결석 형성 및 대사성 산증의 발생으로 구성된 군으로부터 선택된 환자에서 1 이상의 상태의 예방, 역전, 유지 또는 지연을 초래하는만성 신장병 환자의 치료 방법.
- 환자에게 제2철 유기 화합물 유효량을 투여하는 것을 포함하는만성 신장병 환자의 치료 방법.
- 제21항에 있어서,상기 제2철 유기 화합물이 경구적으로 투여되는만성 신장병 환자의 치료 방법.
- 제21항에 있어서,상기 환자는 인간 또는 동물인만성 신장병 환자의 치료 방법.
- 제21항에 있어서,상기 환자는 만성 신장병의 임의 단계에 있거나, 또는 신장 투석을 받는만성 신장병 환자의 치료 방법.
- 제21항에 있어서,상기 제2철 유기 화합물이 정제, 분말, 현탁액, 에멀젼, 캡슐, 로젠지, 과립, 트로체, 환약, 액체, 스피리트 또는 시럽으로 제형화되는만성 신장병 환자의 치료 방법.
- 제21항에 있어서,상기 제2철 유기 화합물이 2 mg/cm2/분 이상의 용해 속도를 갖는만성 신장병 환자의 치료 방법.
- 제21항 내지 제26항 중 어느 한 항에 있어서,상기 제2철 유기 화합물을 사용한 치료는 환자에서 크레아티닌, BUN, 인 및 칼슘과 인 생성물(CaxP)로 구성된 군으로부터 선택되는 1 이상의 물질의 감소된 혈청 수준을 초래하는만성 신장병 환자의 치료 방법.
- 제21항 내지 제26항 중 어느 한 항에 있어서,상기 제2철 유기 화합물을 사용한 치료는 만성 신장병의 진행, 부갑상선기능항진증의 발생, 뼈 질병의 발생, 심장혈관 질병의 발생, 신장병에서 칼슘 포스페이트 석출, 신장 결석 형성, 및 대사성 산증의 발생으로 구성된 군으로부터 선택된 환자에서 1 이상의 상태의 예방, 역전, 유지 또는 지연을 초래하는만성 신장병 환자의 치료 방법.
- 제21항 내지 제28항 중 어느 한 항에 있어서,상기 제2철 유기 화합물이 시트르산제2철인만성 신장병 환자의 치료 방법.
- 허용되는 담체 및 2 mg/cm2/분 이상의 용해 속도를 갖는 제2철 유기 화합물 유효량을 포함하는 약학적 조성물을 환자에게 단일 또는 다수회 용량 투여하는 것을 포함하는만성 신장병 환자의 치료 처방.
- 제30항에 있어서,상기 제2철 유기 화합물의 상기 용해 속도가 2.5 mg/cm2/분 내지 3.0 mg/cm2/분인만성 신장병 환자의 치료 처방.
- 제30항에 있어서,상기 제2철 유기 화합물의 상기 용해 속도가 3.0 mg/cm2/분 내지 3.5 mg/cm2/분인만성 신장병 환자의 치료 처방.
- 제30항에 있어서,상기 제2철 유기 화합물의 용해 속도가 3.5 mg/cm2/분 내지 4.0 mg/cm2/분인만성 신장병 환자의 치료 처방.
- 제30항 내지 제33항 중 어느 한 항에 있어서,상기 약학적 조성물의 적어도 일부가 경구적으로 투여되는만성 신장병 환자의 치료 처방.
- 제30항에 있어서,상기 만성 신장병은 만성 신장병의 임의 단계 및 말기 신장병을 포함하는만성 신장병 환자의 치료 처방.
- 제30항 내지 제35항 중 어느 한 항에 있어서,신장 투석 또는 복강 투석을 더 포함하는만성 신장병 환자의 치료 처방.
- 제30항 내지 제36항 중 어느 한 항에 있어서,상기 제2철 유기 화합물은 정제, 분말, 현탁액, 에멀젼, 캡슐, 로젠지, 과립, 트로체, 환약, 액체, 스피리트 또는 시럽으로 제형화된만성 신장병 환자의 치료 처방.
- 제30항 내지 제37항 중 어느 한 항에 있어서,상기 제2철 유기 화합물이 시트르산제2철인만성 신장병 환자의 치료 처방.
- 2 mg/cm2/분 이상의 용해 속도를 갖는 제2철 유기 화합물 유효량을 포함하는만성 신장병 환자를 치료하기 위한 약학적 조성물.
- 제39항에 있어서,상기 제2철 유기 화합물의 상기 용해 속도가 2 mg/cm2/분 내지 4 mg/cm2/분인만성 신장병 환자를 치료하기 위한 약학적 조성물.
- 제39항 또는 제40항에 있어서,상기 제2철 유기 화합물이 시트르산제2철인만성 신장병 환자를 치료하기 위한 약학적 조성물.
- 제39항 내지 제41항 중 어느 한 항에 있어서,상기 조성물은 경구 투여에 적합한 형태인만성 신장병 환자를 치료하기 위한 약학적 조성물.
- 제 42항에 있어서,경구 투여에 적합한 상기 형태가 정제, 분말, 현탁액, 에멀젼, 캡슐, 로젠지, 과립, 트로체, 환약, 액체, 스피리트 또는 시럽인만성 신장병 환자를 치료하기 위한 약학적 조성물.
- 만성 신장병 환자를 치료하기 위한 의약 제조에서 제 39항 내지 제 43항 중 어느 한 항에 따른약학적 조성물의 용도.
- 제44항에 있어서,상기 환자는 만성 신장병의 임의 단계이거나, 또는 신장 투석을 받는약학적 조성물의 용도.
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US76325306P | 2006-01-30 | 2006-01-30 | |
| US60/763,253 | 2006-01-30 | ||
| US63238506A | 2006-08-18 | 2006-08-18 | |
| USPCT/US2006/032385 | 2006-08-18 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20080106506A true KR20080106506A (ko) | 2008-12-08 |
Family
ID=40154338
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020087018633A Ceased KR20080106506A (ko) | 2006-01-30 | 2007-01-26 | 만성 신장병의 치료 방법 |
| KR1020087018634A Ceased KR20080094013A (ko) | 2006-01-30 | 2007-01-26 | 연조직의 석회화 역전, 예방, 지연 또는 안정화 방법 |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020087018634A Ceased KR20080094013A (ko) | 2006-01-30 | 2007-01-26 | 연조직의 석회화 역전, 예방, 지연 또는 안정화 방법 |
Country Status (1)
| Country | Link |
|---|---|
| KR (2) | KR20080106506A (ko) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7767851B2 (en) | 2003-02-19 | 2010-08-03 | Panion & Bf Biotech, Inc. | Ferric organic compounds, uses thereof and methods of making same |
| US8093423B2 (en) | 2003-02-19 | 2012-01-10 | Globoasia, Llc | Pharmaceutical-grade ferric organic compounds, uses thereof and method of making same |
| US9387191B2 (en) | 2009-07-21 | 2016-07-12 | Keryx Biopharmaceuticals, Inc. | Ferric citrate dosage forms |
| KR20160096597A (ko) * | 2013-11-04 | 2016-08-16 | 케릭스 바이오파마슈티컬스 인코포레이티드 | 만성 신장 질환 환자의 심부전 감소를 위한 시트르산 제2철 |
| US9750715B2 (en) | 2006-01-30 | 2017-09-05 | Panion & Biotech Inc. | Method of reversing, preventing, delaying or stabilizing soft tissue calcification |
-
2007
- 2007-01-26 KR KR1020087018633A patent/KR20080106506A/ko not_active Ceased
- 2007-01-26 KR KR1020087018634A patent/KR20080094013A/ko not_active Ceased
Cited By (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8901349B2 (en) | 2003-02-19 | 2014-12-02 | Panion & Bf Biotech Inc. | Ferric organic compounds, uses thereof and methods of making same |
| US9050316B2 (en) | 2003-02-19 | 2015-06-09 | Panion & Bf Biotech Inc. | Pharmaceutical-grade ferric organic compounds, uses thereof and methods of making same |
| US8299298B2 (en) | 2003-02-19 | 2012-10-30 | Globoasia, Llc | Pharmaceutical-grade ferric organic compounds, uses thereof and method of making same |
| US8338642B2 (en) | 2003-02-19 | 2012-12-25 | Panion & Bf Biotech, Inc. | Ferric organic compounds, uses thereof and methods of making same |
| US8609896B2 (en) | 2003-02-19 | 2013-12-17 | Panion & Bf Biotech Inc. | Ferric organic compounds, uses thereof and methods of making same |
| US8754258B2 (en) | 2003-02-19 | 2014-06-17 | Panion & Bf Biotech Inc. | Ferric organic compounds, uses thereof and methods of making same |
| US8093423B2 (en) | 2003-02-19 | 2012-01-10 | Globoasia, Llc | Pharmaceutical-grade ferric organic compounds, uses thereof and method of making same |
| US8754257B2 (en) | 2003-02-19 | 2014-06-17 | Panion & Bf Biotech Inc. | Pharmaceutical-grade ferric organic compounds, uses thereof and methods of making same |
| US7767851B2 (en) | 2003-02-19 | 2010-08-03 | Panion & Bf Biotech, Inc. | Ferric organic compounds, uses thereof and methods of making same |
| US8846976B2 (en) | 2003-02-19 | 2014-09-30 | Panion & Bf Biotech Inc. | Ferric organic compounds, uses thereof and methods of making same |
| US9328133B2 (en) | 2003-02-19 | 2016-05-03 | Panion & Bf Biotech Inc. | Ferric organic compounds, uses thereof and methods of making same |
| US9757416B2 (en) | 2003-02-19 | 2017-09-12 | Panion & Bf Biotech Inc. | Pharmaceutical-grade ferric organic compounds, uses thereof and methods of making same |
| US9750715B2 (en) | 2006-01-30 | 2017-09-05 | Panion & Biotech Inc. | Method of reversing, preventing, delaying or stabilizing soft tissue calcification |
| US9387191B2 (en) | 2009-07-21 | 2016-07-12 | Keryx Biopharmaceuticals, Inc. | Ferric citrate dosage forms |
| US10300039B2 (en) | 2009-07-21 | 2019-05-28 | Keryx Biopharmaceuticals, Inc. | Ferric citrate dosage forms |
| KR20160096597A (ko) * | 2013-11-04 | 2016-08-16 | 케릭스 바이오파마슈티컬스 인코포레이티드 | 만성 신장 질환 환자의 심부전 감소를 위한 시트르산 제2철 |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20080094013A (ko) | 2008-10-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20250144065A1 (en) | Method of treating chronic kidney disease | |
| US20190269722A1 (en) | Pharmaceutical-grade ferric organic compounds, uses thereof and methods of making same | |
| US9757416B2 (en) | Pharmaceutical-grade ferric organic compounds, uses thereof and methods of making same | |
| TWI375555B (en) | Pharmaceutical-grade ferric organic compounds, uses thereof and methods of making same | |
| KR20080106506A (ko) | 만성 신장병의 치료 방법 | |
| HK1228669A1 (en) | Method of treating chronic kidney disease | |
| HK1228669A (en) | Method of treating chronic kidney disease | |
| HK1228669B (en) | Method of treating chronic kidney disease | |
| US20220144872A1 (en) | Pharmaceutical-Grade Ferric Organic Compounds, Uses Thereof and Methods of Making Same | |
| KR20160096178A (ko) | 리튬 공동 투여 없이 고칼륨혈증을 치료하기 위한 규산지르코늄 | |
| Gui | Phosphate binders in chronic kidney disease |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
Patent event date: 20080729 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PG1501 | Laying open of application | ||
| A201 | Request for examination | ||
| PA0201 | Request for examination |
Patent event code: PA02012R01D Patent event date: 20111103 Comment text: Request for Examination of Application |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20121217 Patent event code: PE09021S01D |
|
| AMND | Amendment | ||
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20130628 Patent event code: PE09021S01D |
|
| AMND | Amendment | ||
| E601 | Decision to refuse application | ||
| PE0601 | Decision on rejection of patent |
Patent event date: 20131120 Comment text: Decision to Refuse Application Patent event code: PE06012S01D Patent event date: 20130628 Comment text: Notification of reason for refusal Patent event code: PE06011S01I Patent event date: 20121217 Comment text: Notification of reason for refusal Patent event code: PE06011S01I |
|
| AMND | Amendment | ||
| J201 | Request for trial against refusal decision | ||
| PJ0201 | Trial against decision of rejection |
Patent event date: 20131219 Comment text: Request for Trial against Decision on Refusal Patent event code: PJ02012R01D Patent event date: 20131120 Comment text: Decision to Refuse Application Patent event code: PJ02011S01I Appeal kind category: Appeal against decision to decline refusal Appeal identifier: 2013101008822 Request date: 20131219 |
|
| PB0901 | Examination by re-examination before a trial |
Comment text: Amendment to Specification, etc. Patent event date: 20131219 Patent event code: PB09011R02I Comment text: Request for Trial against Decision on Refusal Patent event date: 20131219 Patent event code: PB09011R01I Comment text: Amendment to Specification, etc. Patent event date: 20130827 Patent event code: PB09011R02I Comment text: Amendment to Specification, etc. Patent event date: 20130214 Patent event code: PB09011R02I |
|
| B601 | Maintenance of original decision after re-examination before a trial | ||
| PB0601 | Maintenance of original decision after re-examination before a trial | ||
| J301 | Trial decision |
Free format text: TRIAL DECISION FOR APPEAL AGAINST DECISION TO DECLINE REFUSAL REQUESTED 20131219 Effective date: 20150612 |
|
| PJ1301 | Trial decision |
Patent event code: PJ13011S01D Patent event date: 20150612 Comment text: Trial Decision on Objection to Decision on Refusal Appeal kind category: Appeal against decision to decline refusal Request date: 20131219 Decision date: 20150612 Appeal identifier: 2013101008822 |