KR101005854B1 - Alkaline degreasing agent composition for plating pretreatment and degreasing method using the same - Google Patents
Alkaline degreasing agent composition for plating pretreatment and degreasing method using the same Download PDFInfo
- Publication number
- KR101005854B1 KR101005854B1 KR1020100069121A KR20100069121A KR101005854B1 KR 101005854 B1 KR101005854 B1 KR 101005854B1 KR 1020100069121 A KR1020100069121 A KR 1020100069121A KR 20100069121 A KR20100069121 A KR 20100069121A KR 101005854 B1 KR101005854 B1 KR 101005854B1
- Authority
- KR
- South Korea
- Prior art keywords
- degreasing
- plating
- sodium
- composition
- present
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23G—CLEANING OR DE-GREASING OF METALLIC MATERIAL BY CHEMICAL METHODS OTHER THAN ELECTROLYSIS
- C23G5/00—Cleaning or de-greasing metallic material by other methods; Apparatus for cleaning or de-greasing metallic material with organic solvents
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25D—PROCESSES FOR THE ELECTROLYTIC OR ELECTROPHORETIC PRODUCTION OF COATINGS; ELECTROFORMING; APPARATUS THEREFOR
- C25D5/00—Electroplating characterised by the process; Pretreatment or after-treatment of workpieces
- C25D5/34—Pretreatment of metallic surfaces to be electroplated
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Metallurgy (AREA)
- Organic Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Mechanical Engineering (AREA)
- Electrochemistry (AREA)
- Detergent Compositions (AREA)
- Cleaning And De-Greasing Of Metallic Materials By Chemical Methods (AREA)
Abstract
본 발명은 도금 전처리용 알칼리계 탈지제 조성물 및 이를 이용한 탈지방법에 관한 것으로서, 수산화나트륨(NaOH) 20~35 g/ℓ, 로릴황산나트륨(Sodium lauryl sulfate) 2~6 g/ℓ, 규산나트륨(Na2SiO3) 0.2~3 g/ℓ, 탄산나트륨(Na2CO3) 20~50 g/ℓ 및 에틸렌다이아민테트라아세트산 (EDTA) 10~40 g/ℓ로 이루어지는 도금 전처리용 알칼리계 탈지제 조성물 및 상기 기재의 조성물에 피도금체를 침지하는 것을 특징으로 하는 도금 전처리 공정 내 탈지방법을 제공함으로써, 도금 공정품의 불량을 최소한으로 줄일 수 있게 한 것이다.The present invention relates to an alkali degreasing agent composition for plating pretreatment and a degreasing method using the same, sodium hydroxide (NaOH) 20 ~ 35 g / L, sodium lauryl sulfate 2 ~ 6 g / l, sodium silicate (Na 2 Alkali-based degreasing agent composition for plating pretreatment consisting of 0.2 to 3 g / l of SiO 3 ), 20 to 50 g / l of sodium carbonate (Na 2 CO 3 ) and 10 to 40 g / l of ethylenediaminetetraacetic acid (EDTA) and the substrate By providing a degreasing method in the plating pretreatment step, the plating material is immersed in the composition of the, it is possible to minimize the defect of the plating process products.
Description
본 발명은 도금 전처리용 알칼리계 탈지제 조성물 및 이를 이용한 탈지방법에 관한 것이다.The present invention relates to an alkaline degreasing agent composition for plating pretreatment and a degreasing method using the same.
도금공정이란 금속 또는 비금속 물질의 표면상태를 개선하기 위해 다른 금속의 얇은층을 피복하는 과정으로 크게 탈지과정-활성화과정-도금과정-후처리과정으로 나누어진다. 이중 탈지과정은 도금공정에 있어서 매우 중요한 부분으로 금속표면에 부착되어 있는 산화물, 수산화물, 금속염 및 유지류 등의 오염물의 제거를 목적으로 하며, 오염물의 종류에 따라 알칼리계, 유기용매계 및 에멀션계 탈지로 분류된다.The plating process is a process of coating a thin layer of another metal to improve the surface state of a metal or nonmetallic material, and is generally divided into a degreasing process, an activation process, a plating process, and a post-treatment process. The degreasing process is a very important part of the plating process and aims to remove contaminants such as oxides, hydroxides, metal salts and fats and oils attached to the metal surface, and degreasing alkalis, organic solvents and emulsions depending on the contaminants. Classified as
유기물 중 광물성 유지(방청유, 그리스 등) 제거를 위한 유기용매계 탈지의 경우 알코올류, 케톤류, 에테르류, 리모넨계, 석유계 등의 유기계 용매나 1,1,1-트리클로르에틸렌(TCE)과 프레온 등의 염소계 용매를 사용하는데, 염소계 용매의 경우 오존층 파괴 및 인체에 대한 유해성 때문에 규제를 받고 있는 실정이다. Organic solvent-based degreasing for the removal of mineral oils (rust preventive oil, grease, etc.) among organic substances, organic solvents such as alcohols, ketones, ethers, limonene and petroleum, and 1,1,1-trichlorethylene (TCE) and Chlorine-based solvents such as Freon are used, but the chlorine-based solvents are regulated due to the ozone layer destruction and the harmfulness to the human body.
에멀션계 탈지의 경우, 주로 연마공정에서 발생되는 유무기 혼합 오염물의 제거를 위하여 사용되는데, 주로 케로신, 크롤계 용매, 계면활성제 및 물을 혼합하여 사용한다.In the case of emulsion degreasing, it is mainly used for the removal of organic-inorganic mixed contaminants generated in the polishing process, and is mainly used by mixing kerosine, crawling solvent, surfactant and water.
알칼리계 탈지과정의 경우 동식물성 유지를 제거하기 위한 수용성 탈지과정으로 친유성 오염물의 경우 계면활성제를 첨가하여 탈지능을 개선한다. In the case of alkaline degreasing, water-soluble degreasing is used to remove animal and vegetable fats and oils. In the case of lipophilic contaminants, surfactants are added to improve degreasing ability.
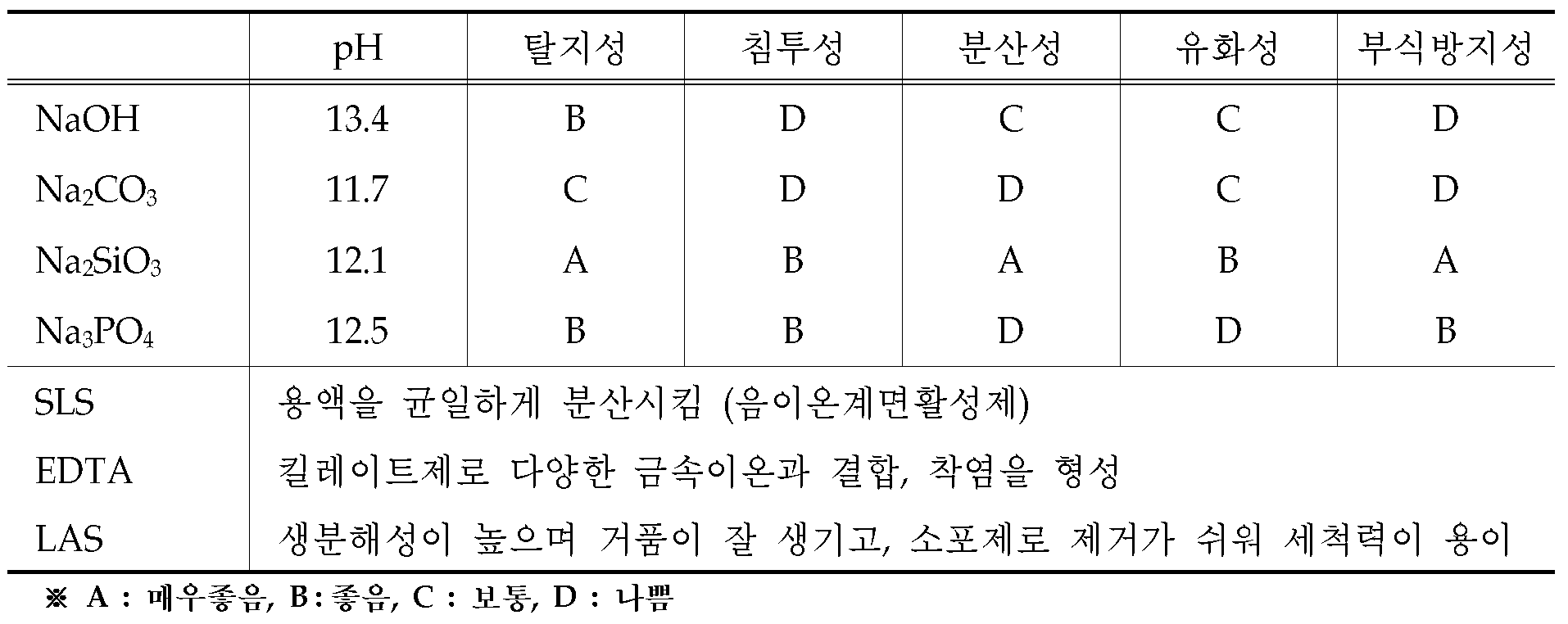
국내에서 판매되고 있는 알칼리계 탈지제는 몇 가지 알칼리염과 한 가지 또는 그 이상의 계면활성제를 넣어서 제조하고 있다. 예를 들어, 철강의 침지 탈지용에는 수산화나트륨(NaOH)이 들어가서 산성의 오물을 중화시켜주고, 오물을 해교시키기 위해서 제3인산나트륨(Na3PO4)를 넣으며, 광물성 유지오염물을 제거하기 위해서 피로인산나트륨(Na4P2O7)을 사용한다. 또한, 경수(硬水)때문에 유기계면활성제도 음이온계, 양이온계, 비이온계를 첨가하여 사용하는데, 최종적으로 알칼리도를 유지하기 위해 탄산나트륨(Na2CO3)을 넣고, 계면활성제도 값싼 약품으로 대치하도록 고려하여 사용하고 있다. 알루미늄의 경우에는 수산화나트륨은 넣지 않고, 메타규산나트륨(Na2SiO3)의 양을 증가시키고, 규산계라도 규산이 많은 것을 넣어 부식을 억제한다. 제3인산나트륨 대신 피로인산나트륨을 사용하고 탄산나트륨도 최소한으로 줄여서 pH를 낮춘다. 그리고, 흔히 계면활성제도 비누계와 비이온계를 첨가하여 사용하고 있다.Alkaline degreasing agents sold in Korea are prepared by adding several alkali salts and one or more surfactants. For example, sodium hydride (NaOH) enters to neutralize acidic dirt, and sodium triphosphate (Na 3 PO 4 ) is added to bridge the dirt, and to remove mineral fats and oils. Sodium pyrophosphate (Na 4 P 2 O 7 ) is used. In addition, due to hard water, organic surfactants are also used by adding anionic, cationic, and nonionic compounds. Finally, in order to maintain alkalinity, sodium carbonate (Na2CO3) is added, and the surfactant is replaced with a cheap chemical agent. I use it. In the case of aluminum, sodium hydroxide is not added, and the amount of sodium metasilicate (Na 2 SiO 3 ) is increased, and even silicic acid-containing silicic acid is added to suppress corrosion. Use sodium pyrophosphate instead of sodium triphosphate and reduce the pH by reducing sodium carbonate to a minimum. In addition, surfactants are often used by adding soap and nonionics.
이러한 알칼리계 탈지제는 탈지공정에서 가격이 저렴하고 작업병이 없으며, 장치가 간편한 장점을 가지고 있어 방청유, 절삭유, 프레스유 등의 유지제거에 가장 널리 사용되고 있다. Such alkali-based degreasing agents are most widely used for the removal and removal of rust-preventive oils, cutting oils, press oils, etc. because of the advantages of low cost and no working bottles in the degreasing process, and a simple device.
일반적으로 사용되는 알칼리계 탈지제의 조성 및 성능을 하기 표 1에 나타내었다.The composition and performance of the alkali degreasing agent generally used are shown in Table 1 below.
하지만, 금속표면의 탈지과정은 단지 탈지효과를 얻기 위한 것이 아니라 비누화 작용, 유화작용, 침투작용, 분산작용 및 기계적 박리작용을 수행한다. 비누화 작용이란 유지류의 불순물을 알칼리수용액 중에서 가열하여 비누와 글리세린으로 분리되는 것을 말한다. 그러나 비누화 작용을 하지 않는 광물성 유지의 경우 유기용매로 용해하거나 유지를 미립화하여 분산시키는 유화작용에 의해서 제거한다. 습윤작용이라고도 불리는 침투작용은 유지 속에 침투해서 유지분자를 파괴하는 작용을 하며, 기계적 박리작용은 공기 교반이나 초음파 세정 등과 같이 기계적인 힘에 의해서 유지를 금속표면으로부터 떼어내는 과정을 말한다. However, the degreasing process of the metal surface is not just to achieve a degreasing effect but to perform saponification, emulsification, infiltration, dispersion and mechanical peeling. The saponification action refers to the separation of soap and glycerin by heating impurities in fats and oils in alkaline aqueous solution. However, in the case of mineral oils that do not saponify, they are dissolved by an organic solvent or removed by emulsification to atomize oils and disperse them. Penetration, also known as wetting, penetrates into oil and destroys oil molecules. Mechanical exfoliation is the process of removing oil from metal surfaces by mechanical forces, such as air agitation or ultrasonic cleaning.
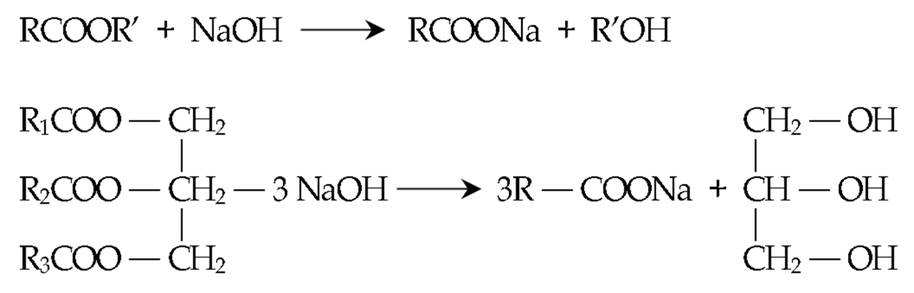
이에, 알칼리계 탈지과정은 하기 식 1과 같이 동식물성 유지를 가열하여 알칼리 수용액과 반응하여 수용성 비누를 만드는 비누화 반응이 그 주체이다. 비누화성 유지는 지방산 글리세린 에스테르이므로 이것이 알칼리 수용액과 반응하여 지방산나트륨인 비누와 글리세린으로 나누어진다. 이 반응에서 생성된 비누는 계면활성제로서 유화력, 분산력을 가지고 있기때문에 2차적으로 알칼리 수용액에 의해서도 유화작용 및 분산작용이 이루어진다.Thus, the alkaline degreasing process is mainly a saponification reaction to heat the animal and vegetable fats and oils and react with an aqueous alkali solution to form a water-soluble soap, as shown in the following
[식 1][Equation 1]
하지만, 이러한 탈지과정이 불충분할 경우 도금층의 밀착불량, 광택불량, 도금면의 흠, 부풀음 등의 발생, 도금의 취화(brittle) 및 부식 등이 발생된다. 금속제품의 표면오염물은 재질, 형상, 제조공정, 제조환경에 따라 달라지며, 오염물의 완전한 제거는 매우 어렵다. However, when the degreasing process is insufficient, poor adhesion of the plating layer, poor gloss, flaws of the plating surface, swelling, etc., brittleness and corrosion of the plating may occur. Surface contaminants in metal products depend on the material, shape, manufacturing process, and manufacturing environment, and it is very difficult to remove the contaminants completely.
따라서 본 발명에서는 도금공정의 전처리과정 중 탈지 공정에 사용할 수 있는 맞춤형 알칼리계 탈지제를 개발하고자 하였다.Therefore, in the present invention, it was intended to develop a customized alkali degreasing agent that can be used in the degreasing process during the pretreatment of the plating process.
이에, 본 발명의 목적은 도금공정의 전처리과정 중 탈지 공정에 사용할 수 있는 맞춤형 알칼리계 탈지제를 제공하는 것이다.Accordingly, an object of the present invention is to provide a customized alkali degreasing agent that can be used in the degreasing step of the pretreatment of the plating process.
또한, 본 발명의 또 다른 목적은 도금공정의 전처리 과정 중 탈지 공정에서 상기 탈지제에 피도금체를 침지하여 탈지하는 방법을 제공하는 것이다.In addition, another object of the present invention is to provide a method for degreasing by immersing the plated body in the degreasing agent in the degreasing step during the pretreatment of the plating process.
상기와 같은 목적을 달성하기 위하여, 본 발명은 수산화나트륨(NaOH) 20~35 g/ℓ, 로릴황산나트륨(Sodium lauryl sulfate) 2~6 g/ℓ, 규산나트륨(Na2SiO3) 0.2~3 g/ℓ, 탄산나트륨(Na2CO3) 20~50 g/ℓ 및 에틸렌다이아민테트라아세트산 (EDTA) 10~40 g/ℓ로 이루어지는 도금 전처리용 알칼리계 탈지제 조성물을 제공한다.In order to achieve the above object, the present invention is sodium hydroxide (NaOH) 20 ~ 35 g / L,
또한, 다른 목적을 달성하기 위하여, 본 발명은 상기 기재의 조성물에 피도금체를 침지하는 것을 특징으로 하는 도금 전처리 공정 내 탈지방법을 제공한다.In addition, in order to achieve another object, the present invention provides a degreasing method in the plating pretreatment process, characterized in that the plated body is immersed in the composition of the substrate.
이하, 본 발명을 상세히 설명하면 다음과 같다.Hereinafter, the present invention will be described in detail.
이때, 사용되는 기술 용어 및 과학 용어에 있어서 다른 정의가 없다면, 이 발명이 속하는 기술 분야에서 통상의 지식을 가진 자가 통상적으로 이해하고 있는 의미를 가진다.At this time, if there is no other definition in the technical terms and scientific terms used, it has a meaning commonly understood by those of ordinary skill in the art.
또한, 종래와 동일한 기술적 구성 및 작용에 대한 반복되는 설명은 생략하기로 한다.Repeated descriptions of the same technical constitution and operation as those of the conventional art will be omitted.
본 발명은 탈지 성능이 매우 우수하면서 사용수명을 예측할 수 있어 도금 공정품의 불량을 최소한으로 줄일 수 있는 도금 전처리용 알칼리계 탈지제 조성물을 개발한 것이다.The present invention has been developed an alkali-based degreasing agent composition for plating pre-treatment that can be used to predict the service life is very excellent degreasing performance to minimize the defect of the plating process products.
구체적으로 본 발명은 종래 도금 전처리 공정 중 탈지공정에서 사용되는 탈지제 성분에서 수산화나트륨(NaOH), 탄산나트륨(Na2CO3), 규산나트륨(Na2SiO3), 로릴황산나트륨(Sodium lauryl sulfate) 및 에틸렌다이아민테트라아세트산(이하, EDTA이라고 함)의 특정 성분을 조합하고 이들 성분을 일정비율로 첨가하여 제조하여 시너지 효과에 의하여 탈지 성능을 향상시키면서 사용주기를 배가되도록 한 것이다.Specifically, the present invention is sodium hydroxide (NaOH), sodium carbonate (Na 2 CO 3 ), sodium silicate (Na 2 SiO 3 ), sodium lauryl sulfate and ethylene in the degreasing agent component used in the degreasing step of the conventional plating pretreatment process It is prepared by combining specific components of diamine tetraacetic acid (hereinafter referred to as EDTA) and adding these components at a certain ratio to double the cycle of use while improving the degreasing performance by synergistic effects.
본 발명에 따른 도금 전처리용 알칼리계 탈지제 조성물에서 수산화나트륨은 비누화 작용을 통해 탈지 효과를 제공하는 알칼리 성분으로서, 전체 조성물 1ℓ 기준에서 규산나트륨(Na2SiO3) 0.2~3 g/ℓ및 탄산나트륨(Na2CO3) 20~50 g/ℓ의 존재하에 20~35 g을 사용하는 것이 바람직하고 보다 바람직하게는 30g을 사용하는데, 이는 20g 미만을 사용하면 탈지 공정 후 피도금체의 표면에 물맺힘 억제와 탈지 효과가 저감되고, 35g을 초과하면 초과량에 비례하여 물맺힘 억제와 탈지 효과가 미미하기 때문이다.In the alkali-based degreasing agent composition for plating pretreatment according to the present invention, sodium hydroxide is an alkali component that provides a degreasing effect through a saponification action, and 0.2 to 3 g / l of sodium silicate (Na 2 SiO 3 ) and sodium carbonate (based on 1 L of the total composition). Na 2 CO 3 ) It is preferable to use 20 to 35 g in the presence of 20 to 50 g / ℓ, more preferably 30 g, which is less than 20 g if the water to the surface of the plated body after the degreasing process This is because the suppression and degreasing effect is reduced, and when it exceeds 35 g, the water squeeze suppression and degreasing effect is insignificant in proportion to the excess amount.
그리고, 탄산나트륨은 수산화나트륨의 탈지 성능을 개선하기 위해 사용하는데, 전체 조성물 1ℓ 기준에서 수산화나트륨 20~35 g/ℓ과 규산나트륨 0.2~3 g/ℓ의 존재하에 20~50 g을 사용하는 것이 바람직하고 보다 바람직하게는 40g을 사용하는데, 이는 상기 사용 범위 내에서 사용하는 것이 탈지 성능의 개선 효과가 가장 우수하기 때문이다.In addition, sodium carbonate is used to improve the degreasing performance of sodium hydroxide. It is preferable to use 20-50 g in the presence of 20-35 g / l sodium hydroxide and 0.2-3 g / l sodium silicate based on 1 l of the total composition. And more preferably 40 g, since the use within the range of use is the most effective in improving the degreasing performance.
또한, 본 발명의 탈지제 조성물에서 규산나트륨은 수산화나트륨과 탄산나트륨의 탈지 성능을 개선하기 위해 사용하는데, 상대적으로 가격이 고가이기 때문에 최소한의 사용량으로 최고의 탈지 성능을 나타내기 위하여, 전체 조성물 1ℓ 기준에서 수산화나트륨 20~35 g/ℓ과 탄산나트륨 20~50 g/ℓ의 존재하에 0.2~3 g을 사용하는 것이 바람직하고 보다 바람직하게는 2g을 사용하는데, 이는 0.2g 미만을 사용하면 탈지 성능의 개선 효과가 미미하고, 3g을 초과하면 초과량에 비례하여 탈지 성능은 미미하면서 생산단가가 높아지기 때문이다.In addition, sodium silicate in the degreasing agent composition of the present invention is used to improve the degreasing performance of sodium hydroxide and sodium carbonate, because it is relatively expensive, in order to show the best degreasing performance with a minimum amount of use, the hydroxide on the basis of 1L total composition It is preferable to use 0.2-3 g in the presence of 20-35 g / l sodium and 20-50 g / l sodium carbonate, and more preferably 2 g. When using less than 0.2 g, the effect of improving degreasing performance is reduced. This is because if it is insignificant and exceeds 3g, the degreasing performance is insignificant in proportion to the excess amount and the production cost is increased.
본 발명에서 로릴황산나트륨(이하, SLS라고 함)은 계면활성제로서 탈지 효율이 좋지만 발암물질로 알려져 있어 최소한의 사용량으로 최고의 탈지 효과를 나타내기 위하여 전체 조성물 1ℓ 기준에서 에틸렌다이아민테트라아세트산 10~40 g/ℓ존재하에 2~6 g을 사용하고 바람직하게는 2g을 사용하는데, 이는 2g 미만을 사용하면 탈지율이 낮아지고 6g을 초과하여 사용하면, 본 발명의 목적에 부합되지 않고 초과 사용량에 대비하여 탈지 효과도 미미하기 때문이다.In the present invention, sodium lauryl sulfate (hereinafter referred to as SLS) is a degreasing efficiency as a surfactant, but is known as a carcinogen, so 10 to 40 g of ethylenediaminetetraacetic acid based on 1 l of the total composition to show the best degreasing effect with a minimum amount of use. 2 to 6 g in the presence of / l, preferably 2 g is used, which is less than 2 g degreasing rate, when used in excess of 6 g, does not meet the purpose of the present invention in preparation for excess usage Degreasing effect is also minimal.
또한, 본 발명에서 음이온 계면활성제로서 에틸렌다이아민테트라아세트산 단독으로는 90% 이상의 탈지 효과가 나타나지 않지만 최소량인 SLS 2~4 g/ℓ과 함께 수산화나트륨(NaOH) 20~35 g/ℓ, 규산나트륨(Na2SiO3) 0.2~3 g/ℓ 및 탄산나트륨(Na2CO3) 20~50 g/ℓ존재 하에서 전체 조성물 1ℓ 기준에서 10~40g의 사용 범위 내에서 바람직하게는 25g을 사용하면 평균적으로 95% 이상의 탈지율이 9번을 반복사용해도 유지될 수 있도록 해 준다. 이에, 본 발명에서 10g 미만을 사용하면 상술한 효과가 미미하고 40g 을 초과하여 사용하면 초과 사용량에 비하여 탈지율 효과가 미미하기 때문에, 상술한 사용 범위 내에서 사용하는 것이 바람직하다.In addition, in the present invention, ethylenediaminetetraacetic acid alone does not show a degreasing effect of 90% or more as an anionic surfactant, but sodium hydroxide (NaOH) 20-35 g / l, sodium silicate with a minimum amount of SLS 2-4 g / l. (Na 2 SiO 3 ) in the presence of 0.2 to 3 g / l and 20 to 50 g / l sodium carbonate (Na 2 CO 3 ) in the use range of 10 to 40 g on the basis of 1 l of the total composition, preferably 25 g on average A degreasing rate of more than 95% is maintained even after 9 repetitions. Therefore, when the use of less than 10g in the present invention, the above-mentioned effect is insignificant, and when it is used in excess of 40g, the degreasing rate effect is insignificant compared to the excess amount used, it is preferable to use within the above-mentioned use range.
따라서, 본 발명에 따른 가장 바람직한 최적의 탈지제 조성물은 수산화나트륨 30 g/ℓ, SLS 2 g/ℓ, 규산나트륨 2 g/ℓ, 탄산나트륨 40 g/ℓ및 EDTA 25 g/ℓ로 이루어지는 것이 바람직하다.Thus, the most preferred optimum degreasing agent composition according to the present invention preferably consists of 30 g / l sodium hydroxide, 2 g / l SLS, 2 g / l sodium silicate, 40 g / l sodium carbonate and 25 g / l EDTA.
한편, 본 발명에 따른 도금 전처리용 알칼리계 탈지제 조성물은 피도금체의 침지형 탈지제로 사용하는 방법을 제공하는데, 바람직하게는 본 발명의 탈지제 조성물 40~55 ℃에서 피도금체를 25~40 분간 침지하여 탈지하는 것이 바람직하다. 이는 상술한 탈지 공정의 최적 조건에서 최적의 탈지 효과가 나타나기 때문이다. 더 바람직하게는 50~55 ℃에서 피도금체를 35분간 침지하여 탈지하는 것이 시간 및 비용적 측면에서 가장 우수한 탈지 효과를 나타낸다.Meanwhile, the alkali degreasing agent composition for plating pretreatment according to the present invention provides a method of using the immersion type degreasing agent of the plated body, and preferably, the plated body is immersed for 25-40 minutes at the degreasing agent composition of 40 to 55 ° C. It is preferable to carry out degreasing. This is because the optimum degreasing effect appears in the optimum conditions of the degreasing process described above. More preferably, degreasing by plating the plated body for 35 minutes at 50-55 ° C. shows the best degreasing effect in terms of time and cost.
또한, 본 발명에 따른 침지형 탈지제 조성물은 95% 이상의 탈지 효율이 나타내는 사용수명이 적어도 9번인 것을 입증함으로써, 정확한 사용수명을 통해 공정품의 불량률을 최소화한 것이다. 이에, 본 발명에 따른 탈지제 조성물은 침지형 탈지공정에서 적어도 9 번을 반복하여 사용할 수 있다.In addition, the immersion type degreasing agent composition according to the present invention is to prove at least nine times the service life of the degreasing efficiency of 95% or more, thereby minimizing the defective rate of the process product through accurate service life. Thus, the degreasing agent composition according to the present invention can be used repeatedly at least 9 times in the immersion type degreasing process.
그리고, 본 발명에 따라 제조된 알칼리계 탈지제는 통상적으로 도금공정의 전처리과정 중 탈지 공정에 사용할 수 있으며, 일례로 자동차 휠너트(wheel nut)의 니켈 도금공정의 전처리과정, 더 구체적으로는 니켈-크롬도금의 하지도금공정인 구리도금 전처리과정 중 탈지과정에 사용될 수 있는 맞춤형 알칼리계 탈지제로 사용하는 것이 바람직하다.In addition, the alkali-based degreasing agent prepared according to the present invention can be generally used in the degreasing step of the pretreatment process of the plating process, for example, the pretreatment process of the nickel plating process of the wheel nut of the automobile (nut), more specifically nickel-chromium It is preferable to use a customized alkali degreasing agent that can be used in the degreasing process of the copper plating pretreatment process, which is a plating process of the base plating.
이상과 같이, 본 발명의 침지형 알칼리계 탈지제는 오염물이 제거된 피도금체에 물방울 맺힘 억제과 Hull-cell 도금분석을 통하여 탈지 성능이 매우 우수하면서 사용수명을 예측할 수 있어, 도금 공정품의 불량을 최소한으로 줄일 수 있게 되었다.As described above, the immersion type alkali-based degreasing agent of the present invention can predict the service life with excellent degreasing performance through suppression of water droplet formation and Hull-cell plating analysis on the plated body from which contaminants have been removed, thereby minimizing defects in the plating process products. Can be reduced.
도 1은 본 발명의 실시예에 따른 수산화나트륨의 농도별 탈지성능을 탈지율과 물방울맺힘 실험 결과로 나타낸 것이다.
도 2는 본 발명의 실시예에 따른 SLS의 농도별 탈지성능을 탈지율과 물방울맺힘 실험 결과로 나타낸 것이다.
도 3은 본 발명의 실시예에 따른 규산나트륨의 농도별 탈지성능을 탈지율과 물방울맺힘 실험 결과로 나타낸 것이다.
도 4는 본 발명의 실시예에 따른 탄산나트륨의 농도별 탈지성능을 탈지율과 물방울맺힘 실험 결과로 나타낸 것이다.
도 5는 본 발명의 다른 실시예에 따른 탄산나트륨의 농도별 탈지성능을 탈지율과 물방울맺힘 실험 결과로 나타낸 것이다.
도 6은 본 발명의 실시예에 따른 LAS의 농도별 탈지성능을 나타낸 것이다.
도 7은 본 발명의 실시예에 따른 EDTA의 농도별 탈지성능을 나타낸 것이다.
도 8은 본 발명의 다른 실시예에 따른 EDTA의 농도별 탈지성능을 나타낸 것이다.
도 9는 본 발명에 알칼리계 탈지제의 탈지 시간에 따른 온도별 탈지율을 나타낸 것이다.
도 10은 본 발명에 알칼리계 탈지제의 온도에 따른 시간별 탈지율을 나타낸 것이다.
도 11은 본 발명에 따른 알칼리계 탈지제의 피도금체의 물방울 맺힘 실험의 결과를 나타낸 것이다.
도 12는 본 발명에 따른 알칼리계 탈지제의 반복 사용 횟수에 따른 탈지율을 나타낸 것이다.Figure 1 shows the degreasing performance of each concentration of sodium hydroxide according to an embodiment of the present invention as a result of the degreasing rate and water droplets.
Figure 2 shows the degreasing performance of each concentration of SLS according to an embodiment of the present invention as a result of the degreasing rate and water droplets.
Figure 3 shows the degreasing performance of each concentration of sodium silicate according to an embodiment of the present invention as a result of the degreasing rate and water droplets.
Figure 4 shows the degreasing performance of each concentration of sodium carbonate according to an embodiment of the present invention as a result of the degreasing rate and water droplets.
Figure 5 shows the degreasing performance of each concentration of sodium carbonate according to another embodiment of the present invention as a result of the degreasing rate and water droplets.
Figure 6 shows the degreasing performance for each concentration of LAS according to an embodiment of the present invention.
Figure 7 shows the degreasing performance by concentration of EDTA according to an embodiment of the present invention.
8 shows degreasing performance for each concentration of EDTA according to another embodiment of the present invention.
Figure 9 shows the degreasing rate for each temperature according to the degreasing time of the alkaline degreasing agent in the present invention.
Figure 10 shows the degreasing rate according to the temperature of the alkaline degreasing agent in the present invention.
Figure 11 shows the results of water droplet condensation experiment of the plated body of the alkali degreasing agent according to the present invention.
Figure 12 shows the degreasing rate according to the number of repeated use of the alkaline degreasing agent according to the present invention.
이하, 본 발명을 구체적인 실시예에 의해 보다 더 상세히 설명하고자 한다. 하지만, 본 발명은 하기 실시예에 의해 한정되는 것은 아니며, 본 발명의 사상과 범위 내에서 여러 가지 변형 또는 수정할 수 있음은 이 분야에서 당업자에게 명백한 것이다.Hereinafter, the present invention will be described in more detail with reference to specific examples. However, the present invention is not limited to the following examples, and it will be apparent to those skilled in the art that various changes or modifications can be made within the spirit and scope of the present invention.
[[ 실험예Experimental Example ]]
1.탈지성능평가1. Degreasing performance evaluation
탈지성능을 평가하는 방법에는 간이검사와 정밀검사가 있다. 간이검사방법으로는 물방울 맺힘 검사, 육안검사, 중량검사, 테스트펜(잉크)검사, 자외선조사법, 접촉각 검사 등이 있으며, 정밀검사방법에는 ESCA, SEM 등의 기기분석방법 및 도금특성 확인을 위한 Hull-cell 도금분석 등이 있다. 본 연구에서는 제조된 탈지제의 탈지성능을 평가하기 위해 물방울맺힘 실험과 Hull-cell 도금분석을 수행하였다. 물방울맺힘 실험은 재료표면에 물방울을 떨어뜨리거나 스프레이를 이용하여 그 퍼짐현상을 관찰하므로써 간접적으로 표면의 청결도를 검사하는 방법이며, Hull-cell 도금분석은 음극과 양극 사이의 거리가 다른 267mL의 Hull-cell 조를 이용하여 한번 도금 시에 여러 가지 전류밀도로 도금표면을 관찰할 수 있는 방법으로 도금된 시편의 표면특성을 관찰할 수 있는 분석방법이다. Hull-cell 도금분석방법은 50℃의 온도에서 전전류(total current)를 2A로 설정하여 5분간 도금하였다. 도금 후 Hull-cell 도금패턴에서 왼쪽(고전류밀도)에서 오른쪽(저전류밀도)까지 균일한 도금특성을 확인함으로써 도금의 표면특성을 관찰하는 시험방법이다.There are two methods for evaluating degreasing performance: simple and overhaul. Simple inspection methods include water droplet condensation inspection, visual inspection, weight inspection, test pen (ink) inspection, ultraviolet irradiation method, contact angle inspection, etc., and detailed inspection methods include device analysis methods such as ESCA and SEM, and Hull for checking plating characteristics. -cell plating analysis. In this study, water drop lumping test and Hull-cell plating analysis were performed to evaluate the degreasing performance of the prepared degreasing agent. The dripping test is a method of indirectly checking the cleanliness of a surface by dropping water droplets on the surface of the material or by observing the spreading phenomenon using a spray. In the Hull-cell plating analysis, a 267 mL Hull having a different distance between a cathode and an anode is used. It is an analysis method that can observe the surface characteristics of plated specimens by the method of observing the plating surface with various current densities at the time of one plating using -cell group. In the Hull-cell plating analysis method, the total current was set to 2A at a temperature of 50 ° C. and plated for 5 minutes. It is a test method to observe the surface characteristics of plating by checking uniform plating characteristics from left (high current density) to right (low current density) in the Hull-cell plating pattern after plating.
[[ 실시예Example ] ] 알칼리계Alkali system 탈지제의 제조 Preparation of Degreasing Agent
본 실시예에 사용된 철시편(6.5×2cm)은 55에서 10분간 초음파로 세척하여 건조한 후 사용하였다. 건조된 철시편에 기계유 및 프레스 공정에 사용되는 식물성유지를 일정하게 도포하여 건조한 후 제조된 알칼리계 침지탈지용액 500mL에 넣어 40~50℃의 온도에서 30~40분간 침지시켜 탈지과정을 수행한 후 탈지성능을 평가하였다. 알칼리계 침지탈지제는 수산화나트륨(NaOH, 20~35g/L), 탄산나트륨(Na2CO3, 0.2~3.0g/L), 규산나트륨(Na2SiO3, 20~50g/L), 제3인산나트륨(Na3PO4,10~30g/L) 등을 사용하였고, 음이온 계면활성제로 SLS(sodium lauric sulfate, 2~8g/L), EDTA(ethylene diamine tetra acetic acid, 10~40g/L), LAS(linear alkylbenzene sulfonic acid, 2.5~10g/L)를 선정하여 물에 일정농도로 변량 혼합하여 제조하였다.Iron specimens used in this example (6.5 × 2cm) were used after washing with ultrasonic waves at 55 minutes for 10 minutes. After applying the vegetable oil used in the machine oil and the press process to dry iron specimens and drying it, put it in 500 mL of the alkali-based degreasing solution, and soak it for 30 to 40 minutes at a temperature of 40-50 ℃ to perform the degreasing process. Degreasing performance was evaluated. Alkaline immersion degreasers include sodium hydroxide (NaOH, 20 to 35 g / L), sodium carbonate (Na 2 CO 3 , 0.2 to 3.0 g / L), sodium silicate (Na 2 SiO 3 , 20 to 50 g / L), triphosphate Sodium (Na 3 PO 4 , 10 ~ 30g / L) was used, and as anionic surfactant, SLS (sodium lauric sulfate, 2 ~ 8g / L), EDTA (ethylene diamine tetra acetic acid, 10 ~ 40g / L), LAS (linear alkylbenzene sulfonic acid, 2.5 ~ 10g / L) was selected and prepared by variable mixing in a constant concentration in water.
(1) (One) NaOHNaOH 의 농도영향Effect of concentration on
알칼리계 탈지제용액의 기본이 되는 NaOH에 대한 금속의 부식성을 알아보기 위하여 20~35g/L NaOH 수용액에 유지가 도포된 철시편을 침지시킨 후 각각의 표면 상태를 관찰하였다. 탈지온도는 50℃로 설정하였으며, 40분 탈지 후 탈지율(degreasing ratio)과 물방울맺힘 실험을 통해 탈지성능을 확인하였다. 본 발명에서는 탈지율은 식(2)와 같이 계산하였다.In order to investigate the corrosiveness of the metal to NaOH, which is the base of the alkaline degreasing agent solution, the surface state was observed after immersing the iron specimen coated with fat or oil in 20 ~ 35g / L NaOH aqueous solution. The degreasing temperature was set at 50 ° C., and the degreasing performance was confirmed by a degreasing ratio and water droplet test after 40 minutes of degreasing. In this invention, the degreasing ratio was computed like Formula (2).
[식 2][Equation 2]
이의 결과, 도 1에 도시한 바와 같이 NaOH의 농도를 20~35g/L의 범위를 설정하였으며, 탈지율 계산결과 56.8~61.2% 범위로 나타났으며, 물방울맺힘 실험결과 모든 조건에서 물방울이 맺혔다. As a result, as shown in FIG. 1, the concentration of NaOH was set in the range of 20 to 35 g / L, the degreasing rate calculation result was found to be 56.8 to 61.2%, and water droplets were formed under all conditions.
따라서 NaOH만으로는 탈지제로의 이용이 불가능하였으며, 계면활성제와 다른 알칼리계 시약들의 첨가가 이루어져야 할 것으로 사료된다.
Therefore, NaOH alone could not be used as a degreasing agent, and it is thought that the addition of surfactant and other alkali reagents should be made.
(2) (2) SLSSLS 의 농도 영향Influence of concentration
탈지제 조성 중 계면활성성제인 SLS의 영향을 알아보기 위해 50℃에서 NaOH의 농도는 30g/L로 고정한 가운데 SLS의 농도를 2~8g/L로 변량하여 40분간 탈지성능을 알아보았다. In order to investigate the effect of SLS, a surfactant, in degreasing agent composition, NaOH concentration was fixed at 30g / L at 50 ° C, and SLS concentration was changed to 2-8g / L for 40 minutes of degreasing performance.
이의 결과, 도 2에 도시한 바와 같이 SLS의 농도가 증가함에 따라 탈지율이 증가하였으나 6.0g/L 이상의 농도에서는 탈지율이 변화하지 않았다. 또한 물방울맺힘 실험 결과 6.0g/L 이상의 SLS가 첨가될 경우 표면에 물방울이 맺히지 않았으며, Hull-cell 도금 분석결과에서도 4.0g/L 이상의 SLS가 첨가될 경우 고전류부분에서 미도금 현상이 관찰되지 않았다. 따라서 SLS의 농도는 6.0g/L를 추천할 수 있었다.
As a result, as shown in FIG. 2, the degreasing rate increased as the concentration of SLS was increased, but the degreasing rate did not change at a concentration of 6.0 g / L or more. In addition, when the SLS of 6.0g / L or more was added as a result of water droplet formation, no water droplets formed on the surface, and in the Hull-cell plating analysis, when the SLS or more was added to 4.0g / L, no plating was observed in the high current part. . Therefore, the concentration of SLS could be recommended 6.0g / L.
(3) (3) 규산나트륨과Sodium Silicate 탄산나트륨의 영향 Effect of Sodium Carbonate
앞선 실시예를 통해 알칼리계 탈지제의 기본조성을 수산화나트륨 30g/L, SLS 6.0g/L로 설정한 후, 50℃에서 40분간 규산나트륨(Na2SiO3) 및 탄산나트륨(Na2CO3) 등 다른 알칼리계 탈지시약을 첨가하고 비율에 따른 탈지성능을 알아보았다.After setting the basic composition of the alkaline degreasing agent to 30g / L sodium hydroxide, SLS 6.0g / L through the previous embodiment, and other such as sodium silicate (Na 2 SiO 3 ) and sodium carbonate (Na 2 CO 3 ) for 40 minutes at 50 ℃ Alkaline degreasing reagent was added and the degreasing performance was examined according to the ratio.
규산나트륨의 농도범위를 0.2~3.0g/L로 하고, 탄산나트륨의 농도범위를 20~50g/L로 변량하여 실험한 결과를 각각 도 3과 도 4에 나타내었다.The concentration range of sodium silicate was 0.2 to 3.0 g / L, and the concentration range of sodium carbonate was varied to 20 to 50 g / L, and the results of experiments are shown in FIGS. 3 and 4, respectively.
도 3과 4에 도시한 바와 같이, 규산나트륨과 탄산나트륨 모두 농도가 증가할수록 탈지율은 증가하였으며, 규산나트륨의 첨가가 탄산나트륨에 비해 우수한 탈지율을 나타냄을 확인할 수 있었다. As shown in Figures 3 and 4, both concentrations of sodium silicate and sodium carbonate increased the degreasing rate, it was confirmed that the addition of sodium silicate showed a better degreasing rate than sodium carbonate.
또한, 물방울 맺힘 실험과 Hull-cell 도금분석 결과, 규산나트륨의 경우에는 2.0g/L이상의 조성에서, 탄산나트륨의 경우에는 40g/L이상의 조성에서 우수한 탈지성능을 나타내었다.
In addition, the result of water droplet condensation experiment and Hull-cell plating showed excellent degreasing performance in the composition of sodium silicate of 2.0g / L or more, and of sodium carbonate in the composition of 40g / L or more.
한편, 규산나트륨의 가격이 탄산나트륨에 비해 고가이므로 첨가비율을 탄산나트륨은 고농도로, 규산나트륨은 저농도로 맞춰 아래와 같이 실험을 수행하였다.On the other hand, since the price of sodium silicate is higher than that of sodium carbonate, the experiment was performed by adjusting the addition ratio to high concentration of sodium carbonate and low concentration of sodium silicate.
NaOH의 농도를 30g/L, SLS 6.0g/L, Na2SiO3 2.0g/L를 기본탈지조성으로 하여 Na2CO3 농도에 따른 탈지성능을 알아보았다. 이때, 탄산나트륨의 농도범위는 20~50g/L로 설정하였고, 탈지조건은 50℃, 40분으로 고정하였다.Degreasing performance according to Na 2 CO 3 concentration was determined by NaOH concentration of 30g / L, SLS 6.0g / L, Na 2 SiO 3 2.0g / L as the basic degreasing composition. At this time, the concentration range of sodium carbonate was set to 20 ~ 50g / L, degreasing conditions were fixed at 50 ℃, 40 minutes.
이의 결과, 도 5에 나타난 바와 같이 탄산나트륨의 농도가 증가함에 따라 탈지성능은 증가하였으나 탄산나트륨의 농도가 40g 이상 첨가할 경우 탈지성능의 변화는 크게 나타나지 않았다. 또한, 물방울 맺힘 실험 및 Hull-cell 도금분석 결과 30g 이상의 탄산나트륨이 첨가될 경우 물방울 맺힘 현상과 저전류 및 고전류 부분의 탄도금 및 미도금 현상이 관찰되지 않았다. As a result, as shown in Figure 5, the degreasing performance increased as the concentration of sodium carbonate increased, but the change in the degreasing performance did not appear significantly when the concentration of sodium carbonate was added more than 40g. In addition, water droplet condensation experiments and Hull-cell plating analysis showed that water droplet condensation and low current and high current portions of carbonized and unplated phenomena were not observed when more than 30 g of sodium carbonate was added.
이에, 본 실시예를 통해 최적 탄산나트륨의 농도는 전체 조성물 1ℓ기준으로 40g임을 알 수 있었다.Thus, through this embodiment it was found that the optimal concentration of sodium carbonate is 40g based on the total composition 1ℓ.
따라서, 상기 실시예의 결과를 통해, 본 발명에 따른 최적의 탈지제의 조성은 수산화나트륨 30g/ℓ, 규산나트륨 2.0g/ℓ, 탄산나트륨 40g/ℓ및 SLS 6.0g/ℓ임을 제시할 수 있었다.Therefore, through the results of the above examples, it could be suggested that the optimal composition of the degreasing agent according to the present invention is sodium hydroxide 30g / L, sodium silicate 2.0g / L, sodium carbonate 40g / L and SLS 6.0g / L.
(4) 음이온 계면활성제의 영향(4) Influence of Anionic Surfactants
본 실시예에서는 발암물질로 분류된 SLS를 대신할 수 있는 음이온 계면활성제로 EDTA와 LAS를 선정하여 탈지성능을 확인하였다. LAS는 금속세정에 가장 널리 사용되고 있는 계면활성제이고, EDTA는 금속이온과 착화합물을 만들어 경수를 연수로 만들어주는 경수연화제로 작용하는 효능이 알려져 있다. 이때, 본 발명의 알칼리계 탈지제의 조성은 수산화나트륨 30g/ℓ, 규산나트륨 2.0g/ℓ, 탄산나트륨 40g/ℓ의 기준으로 EDTA와 LAS를 첨가하고 50℃에서 40분간의 탈지조건에서 탈지 성능을 비교분석하였다.In this example, the degreasing performance was confirmed by selecting EDTA and LAS as an anionic surfactant that can replace SLS classified as a carcinogen. LAS is the most widely used surfactant for metal cleaning, and EDTA is known to act as a water softener that makes hard water soft by making complexes with metal ions. At this time, the composition of the alkaline degreasing agent of the present invention is added to EDTA and LAS on the basis of sodium hydroxide 30g / ℓ, sodium silicate 2.0g / ℓ, sodium carbonate 40g / ℓ and compare the degreasing performance at 40 ℃ degreasing conditions for 40 minutes Analyzed.
도 6에 도시한 바와 같이, 음이온 계면활성제인 LAS의 농도범위를 2.5~10g/L로 변량시켜 탈지 실험을 수행한 결과, SLS 대신 LAS를 사용하여 탈지성능을 평가한 결과 탈지율은 85% 이상을 나타내었으나 물방울 맺힘 실험 및 Hull-cell 도금분석 결과 탈지성능이 저하됨을 확인할 수 있었다.As shown in Figure 6, the result of performing a degreasing experiment by varying the concentration range of the LAS anionic surfactant to 2.5 ~ 10g / L, as a result of evaluating the degreasing performance using LAS instead of SLS, the degreasing rate is more than 85% However, it was confirmed that degreasing performance was deteriorated by water droplet condensation experiment and Hull-cell plating analysis.
도 7에 도시한 바와 같이, EDTA의 농도범위는 10~40g/L로 변량시켜 탈지 실험을 수행한 결과, 90% 이상의 탈지율을 얻기 위해서는 다른 음이온계면활성제에 비해 첨가량의 범위가 크게 나타났으며, EDTA만으로는 우수한 탈지성능을 기대할 수 없음을 알 수 있었다.As shown in FIG. 7, the concentration range of EDTA was 10 to 40 g / L, and the result of the degreasing experiment showed that the range of addition amount was larger than that of other anionic surfactants in order to obtain a degreasing ratio of 90% or more. , EDTA alone could not be expected to excellent degreasing performance.
한편, EDTA의 SLS 저감 효과를 확인하고자 본 발명에 따른 탈지제의 기본 조성을 수산화나트륨 30g/ℓ, 규산나트륨 2.0g/ℓ, 탄산나트륨 40g/ℓ및 SLS(2.0g/L)으로 하고, EDTA의 농도범위를 20~35g/ℓ로 변량시키면서 탈지성능을 확인한 결과, 도 8에 나타난 바와 같이 EDTA만을 사용한 경우에 비해 우수한 탈지율을 나타내었으며, 물방울 맺힘 실험 및 Hull-cell 도금분석 결과 매우 우수한 탈지성능을 나타내었으며, 25g/ℓ를 첨가하는 경우 최적의 탈지 성능을 나타냈다.On the other hand, the basic composition of the degreasing agent according to the present invention to determine the SLS reduction effect of EDTA is sodium hydroxide 30g / L, sodium silicate 2.0g / L, sodium carbonate 40g / L and SLS (2.0g / L), the concentration range of EDTA As a result of confirming the degreasing performance by varying 20 to 35 g / l, as shown in FIG. 8, the degreasing ratio was excellent compared to the case of using only EDTA, and the result of water droplet formation and Hull-cell plating analysis showed very good degreasing performance. The addition of 25 g / ℓ showed the best degreasing performance.
따라서, 본 발명에 따른 침지형 알칼리계 탈지제의 최적 조성은 수산화나트륨 30 g/ℓ, SLS 2 g/ℓ, 규산나트륨 2 g/ℓ, 탄산나트륨 40 g/ℓ및 EDTA 25 g/ℓ로 제시할 수 있다.
Therefore, the optimal composition of the immersed alkaline degreasing agent according to the present invention can be presented as 30 g / L sodium hydroxide, 2 g / L SLS, 2 g / L sodium silicate, 40 g / L sodium carbonate and 25 g / L EDTA. .
(5) (5) 알칼리계Alkali system 탈지제의 탈지조건 결정 Determination of Degreasing Conditions
NaOH 30g/L, Na2SiO3 2.0g/L, Na2CO3 40g/L, SLS 6.0g/L, EDTA 25g/L의 조성으로 제조된 맞춤형 알칼리계 탈지제를 이용하여 탈지온도와 탈지시간에 따른 탈지과정의 최적화실험을 수행하였다. 이때, 탈지온도는 40~55℃의 범위에서 일정하게 유지하였고, 탈지시간은 25~40분으로 설정하였다. NaOH 30g / L, Na 2 SiO 3 2.0g / L, Na 2 CO 3 40g / L, SLS 6.0g / L, EDTA 25g / L with custom alkaline degreasing agent The optimization experiment of the degreasing process was performed. At this time, the degreasing temperature was kept constant in the range of 40 ~ 55 ℃, degreasing time was set to 25 to 40 minutes.
이의 결과, 도 9와 도 10에 나타난 바와 같이, 탈지시간의 경우 시간이 증가함에 따라 탈지율은 증가하는 경향을 나타내었으나 35분 이상의 시간에서는 탈지율의 변화를 관찰할 수 없었다. 또한 탈지온도가 증가함에 따라 탈지율도 역시 증가하였으나 50 ℃이상의 온도에서는 탈지율 변화가 관찰되지 않았다. As a result, as shown in Figure 9 and Figure 10, in the case of degreasing time degreasing rate tended to increase as time increases, but the change in degreasing rate could not be observed at the time more than 35 minutes. The degreasing rate also increased with increasing degreasing temperature, but no change in degreasing rate was observed at temperatures above 50 ℃.
또한, 탈지온도와 탈지시간 변화에 따른 물방울 맺힘 실험과 Hull-cell 도금분석 결과를 도 11에 도시하였다. 이때, Hull-cell 도금분석은 50℃의 온도에서 전전류(total current) 2A로 5분간 도금하였으며 왼쪽(고전류밀도)에서 오른쪽(저전류밀도) 부분의 균일한 도금특성을 관찰하게 위해 수행하였다.Also, the water droplet condensation experiment and the Hull-cell plating analysis according to the degreasing temperature and the degreasing time are shown in FIG. 11. At this time, the Hull-cell plating analysis was plated for 5 minutes with a total current (2A) at a temperature of 50 ℃ and was performed to observe the uniform plating characteristics of the right (low current density) portion from the left (high current density).
도 11에 나타난 바와 같이, 물방울 맺힘 검사의 경우 탈지온도 50℃ 이상의 조건에서 35분 이상 탈지한 경우 우수한 탈지성능을 보임을 확인할 수 있었으며, Hull-cell 도금분석의 경우에도 탈지온도 50℃ 이상의 온도에서 30분 이상 탈지한 경우 저전류밀도에서 고전류밀도까지 균일한 도금특성을 나타내어 탈지효과가 뛰어남을 확인할 수 있었다.As shown in FIG. 11, in the case of the water droplet condensation test, when degreased for more than 35 minutes at a degreasing temperature of 50 ° C. or higher, excellent degreasing performance was observed. When degreased for 30 minutes or more, it was confirmed that the degreasing effect was excellent due to uniform plating characteristics from low current density to high current density.
이에, 본 실시예에서는 최적 탈지온도와 탈지시간을 각각 50℃, 35분으로 설정하였다.Thus, in this embodiment, the optimum degreasing temperature and the degreasing time were set to 50 ° C. and 35 minutes, respectively.
(6) 사용수명(6) Service life
본 실시예에서는 본 발명의 탈지제 조성물은 최적 탈지온도와 탈지시간인 50℃에서 35분간 매회 동일하게 실험을 수행하고 동일 탈지제의 반복 탈지 실험을 통해 목표 탈지율이 95% 이상으로 유지되는 사용 수명을 설정하여 실제 공정에서의 알칼리탈지제의 교체주기를 예측하고자 하였다.In the present embodiment, the degreasing agent composition of the present invention performs the same experiment every 35 minutes at the optimum degreasing temperature and the degreasing time at 50 ° C. and through the repeated degreasing experiment of the same degreasing agent to maintain a service life of 95% or more. The purpose of this study was to predict the replacement cycle of alkaline degreasing agent in the actual process.
이의 결과, 도 12에 나타난 바와 같이, 반복횟수가 증가함에 따라 탈지율은 감소하는 경향을 나타내었으나 9회 반복까지 95% 이상의 탈지율을 나타내었다.As a result, as shown in Figure 12, the degreasing rate tended to decrease as the number of repetitions increased, but the degreasing rate was 95% or more until 9 repetitions.
Claims (6)
수산화나트륨(NaOH) 30 g/ℓ, 로릴황산나트륨(Sodium lauryl sulfate) 2 g/ℓ, 규산나트륨(Na2SiO3) 2 g/ℓ, 탄산나트륨(Na2CO3) 40 g/ℓ및 에틸렌다이아민테트라아세트산(EDTA) 25 g/ℓ로 이루어지는 것을 특징으로 하는 도금 전처리용 알칼리계 탈지제 조성물.The method of claim 1,
30 g / l sodium hydroxide (NaOH), 2 g / l sodium lauryl sulfate, 2 g / l sodium silicate (Na 2 SiO 3 ), 40 g / l sodium carbonate (Na 2 CO 3 ) and ethylenediamine Alkaline degreaser composition for plating pretreatment, characterized in that it consists of 25 g / l of tetraacetic acid (EDTA).
조성물의 온도 40~55 ℃에서 25~40 분간 침지하여 탈지하는 것을 특징으로 하는 도금 전처리 공정 내 탈지방법. The method of claim 3, wherein
A degreasing method in a plating pretreatment process, characterized in that the composition is immersed for 25 to 40 minutes at a temperature of 40 to 55 ° C.
조성물의 온도 50 ℃에서 35 분간 침지하여 탈지하는 것을 특징으로 하는 도금 전처리 공정 내 탈지방법.The method of claim 4, wherein
A degreasing method in a plating pretreatment step, characterized in that the composition is immersed for 35 minutes at a temperature of 50 ° C.
상기 조성물은 탈지 공정에서 반복하여 사용할 수 있는 사용수명이 적어도 9 번인 것을 특징으로 하는 도금 전처리 공정 내 탈지방법.The method of claim 3, wherein
The composition is a degreasing method in the plating pre-treatment process, characterized in that the service life can be used repeatedly in the degreasing process at least nine times.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020100069121A KR101005854B1 (en) | 2010-07-16 | 2010-07-16 | Alkaline degreasing agent composition for plating pretreatment and degreasing method using the same |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020100069121A KR101005854B1 (en) | 2010-07-16 | 2010-07-16 | Alkaline degreasing agent composition for plating pretreatment and degreasing method using the same |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR101005854B1 true KR101005854B1 (en) | 2011-01-05 |
Family
ID=43615809
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020100069121A Expired - Fee Related KR101005854B1 (en) | 2010-07-16 | 2010-07-16 | Alkaline degreasing agent composition for plating pretreatment and degreasing method using the same |
Country Status (1)
| Country | Link |
|---|---|
| KR (1) | KR101005854B1 (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR101070127B1 (en) | 2011-07-28 | 2011-10-07 | 서성택 | Alkaline degreasing additives that can be used at room temperature |
| KR101894488B1 (en) * | 2017-04-20 | 2018-09-05 | 최경수 | Method for treating the high gloss of magnesium alloy by chemical treatment |
| KR20200087363A (en) * | 2019-01-10 | 2020-07-21 | 전북대학교산학협력단 | Method for pretreatment of titanium scrap and titanium-iron alloy ingot manufactured using pretreated titanium scrap |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5080831A (en) | 1989-06-29 | 1992-01-14 | Buckeye International, Inc. | Aqueous cleaner/degreaser compositions |
| KR100250385B1 (en) | 1998-02-09 | 2000-04-01 | 정성구 | Alkaline detergent for oil wash of the metal surface |
| KR100601134B1 (en) | 2004-12-10 | 2006-07-19 | (주)양보 | Surface treatment method of stainless steel tube |
| KR20080107036A (en) * | 2007-06-05 | 2008-12-10 | 엄창훈 | Surface treatment method of stainless steel pipe |
-
2010
- 2010-07-16 KR KR1020100069121A patent/KR101005854B1/en not_active Expired - Fee Related
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5080831A (en) | 1989-06-29 | 1992-01-14 | Buckeye International, Inc. | Aqueous cleaner/degreaser compositions |
| KR100250385B1 (en) | 1998-02-09 | 2000-04-01 | 정성구 | Alkaline detergent for oil wash of the metal surface |
| KR100601134B1 (en) | 2004-12-10 | 2006-07-19 | (주)양보 | Surface treatment method of stainless steel tube |
| KR20080107036A (en) * | 2007-06-05 | 2008-12-10 | 엄창훈 | Surface treatment method of stainless steel pipe |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR101070127B1 (en) | 2011-07-28 | 2011-10-07 | 서성택 | Alkaline degreasing additives that can be used at room temperature |
| KR101894488B1 (en) * | 2017-04-20 | 2018-09-05 | 최경수 | Method for treating the high gloss of magnesium alloy by chemical treatment |
| KR20200087363A (en) * | 2019-01-10 | 2020-07-21 | 전북대학교산학협력단 | Method for pretreatment of titanium scrap and titanium-iron alloy ingot manufactured using pretreated titanium scrap |
| KR102190598B1 (en) | 2019-01-10 | 2020-12-15 | 전북대학교산학협력단 | Method for pretreatment of titanium scrap and titanium-iron alloy ingot manufactured using pretreated titanium scrap |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20080146483A1 (en) | Degreasing agent | |
| CN101956220B (en) | Plating pretreatment oil remover composition and preparation and use methods thereof | |
| CN107022766B (en) | Metal surface cleaning agent, preparation method and metal surface cleaning method | |
| KR101005854B1 (en) | Alkaline degreasing agent composition for plating pretreatment and degreasing method using the same | |
| CN102787322A (en) | Aluminium alloy surface alkaline degreasing agent | |
| CN104294295B (en) | A kind of silver-plated pre-treating method of aluminum alloy cleaning agent and aluminium alloy | |
| CN102220592B (en) | Foamless floating-oil-type alkaline cleaning agent used for pre-treatment of pre-coated steel plate | |
| CN103540947B (en) | A kind of plating substrate degreaser and preparation method thereof | |
| CN103774165A (en) | High-efficiency water-based die-casting aluminum alloy degreasing agent | |
| CN103820800A (en) | Low-temperature degreasing cleaning agent | |
| CN110512219A (en) | A kind of low-temperature environment-friendly liquid automobile pre-treatment degreasing agent | |
| CN105937045A (en) | Die-casting aluminum alloy surface treatment method | |
| CN107384617A (en) | Low temperature high performance surface degreasing powder and preparation method thereof | |
| CN107557851A (en) | A kind of alloy electrolytic degreasing powder and its preparation and application | |
| KR20080107036A (en) | Surface treatment method of stainless steel pipe | |
| CN102965680A (en) | Phosphorus-free oil removing process | |
| CN102888607A (en) | Scavenging agent composite for quickly scavenging nickel-cadmium diffusion coating and deplating method of scavenging agent composite | |
| CN106400034A (en) | Degreaser for surface of brass and preparation method thereof | |
| CN114525501B (en) | Magnesium alloy sealing reinforcing agent and sealing reinforcing treatment process | |
| KR100617704B1 (en) | Low temperature composite degreasing agent in cold rolled steel sheet manufacturing process | |
| KR102924432B1 (en) | Degreasing cleaning composition for copper and iron materials | |
| CN108034952B (en) | Painting dressing automobiles table tune pre-treatment high temperature degreasing agent and its degreasing method | |
| JP5957654B1 (en) | Degreasing composition, degreasing composition, and pretreatment method for metal surface | |
| CN106350826A (en) | A method of cleaning and maintenance for railway box car bearings | |
| JPS5938391A (en) | Alkali rust remover composition |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A201 | Request for examination | ||
| PA0109 | Patent application |
St.27 status event code: A-0-1-A10-A12-nap-PA0109 |
|
| PA0201 | Request for examination |
St.27 status event code: A-1-2-D10-D11-exm-PA0201 |
|
| A302 | Request for accelerated examination | ||
| PA0302 | Request for accelerated examination |
St.27 status event code: A-1-2-D10-D17-exm-PA0302 St.27 status event code: A-1-2-D10-D16-exm-PA0302 |
|
| D13-X000 | Search requested |
St.27 status event code: A-1-2-D10-D13-srh-X000 |
|
| D14-X000 | Search report completed |
St.27 status event code: A-1-2-D10-D14-srh-X000 |
|
| E701 | Decision to grant or registration of patent right | ||
| PE0701 | Decision of registration |
St.27 status event code: A-1-2-D10-D22-exm-PE0701 |
|
| GRNT | Written decision to grant | ||
| PR0701 | Registration of establishment |
St.27 status event code: A-2-4-F10-F11-exm-PR0701 |
|
| PR1002 | Payment of registration fee |
St.27 status event code: A-2-2-U10-U11-oth-PR1002 Fee payment year number: 1 |
|
| PG1601 | Publication of registration |
St.27 status event code: A-4-4-Q10-Q13-nap-PG1601 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-5-5-R10-R13-asn-PN2301 St.27 status event code: A-5-5-R10-R11-asn-PN2301 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-5-5-R10-R13-asn-PN2301 St.27 status event code: A-5-5-R10-R11-asn-PN2301 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-5-5-R10-R13-asn-PN2301 St.27 status event code: A-5-5-R10-R11-asn-PN2301 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-5-5-R10-R13-asn-PN2301 St.27 status event code: A-5-5-R10-R11-asn-PN2301 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-5-5-R10-R13-asn-PN2301 St.27 status event code: A-5-5-R10-R11-asn-PN2301 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |
|
| FPAY | Annual fee payment |
Payment date: 20131014 Year of fee payment: 4 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 4 |
|
| FPAY | Annual fee payment |
Payment date: 20141204 Year of fee payment: 5 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 5 |
|
| FPAY | Annual fee payment |
Payment date: 20151112 Year of fee payment: 6 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 6 |
|
| P22-X000 | Classification modified |
St.27 status event code: A-4-4-P10-P22-nap-X000 |
|
| FPAY | Annual fee payment |
Payment date: 20170628 Year of fee payment: 7 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 7 |
|
| FPAY | Annual fee payment |
Payment date: 20171228 Year of fee payment: 8 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 8 |
|
| P22-X000 | Classification modified |
St.27 status event code: A-4-4-P10-P22-nap-X000 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-5-5-R10-R13-asn-PN2301 St.27 status event code: A-5-5-R10-R11-asn-PN2301 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 9 |
|
| P22-X000 | Classification modified |
St.27 status event code: A-4-4-P10-P22-nap-X000 |
|
| FPAY | Annual fee payment |
Payment date: 20191230 Year of fee payment: 10 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 10 |
|
| PC1903 | Unpaid annual fee |
St.27 status event code: A-4-4-U10-U13-oth-PC1903 Not in force date: 20201229 Payment event data comment text: Termination Category : DEFAULT_OF_REGISTRATION_FEE |
|
| K11-X000 | Ip right revival requested |
St.27 status event code: A-6-4-K10-K11-oth-X000 |
|
| PC1903 | Unpaid annual fee |
St.27 status event code: N-4-6-H10-H13-oth-PC1903 Ip right cessation event data comment text: Termination Category : DEFAULT_OF_REGISTRATION_FEE Not in force date: 20201229 |
|
| PR0401 | Registration of restoration |
St.27 status event code: A-6-4-K10-K13-oth-PR0401 |
|
| R401 | Registration of restoration | ||
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 11 |
|
| PC1903 | Unpaid annual fee |
St.27 status event code: A-4-4-U10-U13-oth-PC1903 Not in force date: 20211229 Payment event data comment text: Termination Category : DEFAULT_OF_REGISTRATION_FEE |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |
|
| PC1903 | Unpaid annual fee |
St.27 status event code: N-4-6-H10-H13-oth-PC1903 Ip right cessation event data comment text: Termination Category : DEFAULT_OF_REGISTRATION_FEE Not in force date: 20211229 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |