JP7527559B2 - Adhesion-inducing peptides for barnacle cypris larvae and their applications - Google Patents
Adhesion-inducing peptides for barnacle cypris larvae and their applications Download PDFInfo
- Publication number
- JP7527559B2 JP7527559B2 JP2020098675A JP2020098675A JP7527559B2 JP 7527559 B2 JP7527559 B2 JP 7527559B2 JP 2020098675 A JP2020098675 A JP 2020098675A JP 2020098675 A JP2020098675 A JP 2020098675A JP 7527559 B2 JP7527559 B2 JP 7527559B2
- Authority
- JP
- Japan
- Prior art keywords
- peptide
- barnacle
- amino acid
- cypris larvae
- tag
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 241000238586 Cirripedia Species 0.000 title claims description 172
- 108090000765 processed proteins & peptides Proteins 0.000 title claims description 148
- 241001195836 Cypris Species 0.000 title claims description 88
- 230000001939 inductive effect Effects 0.000 title claims description 44
- 102000004196 processed proteins & peptides Human genes 0.000 title description 24
- 125000000539 amino acid group Chemical group 0.000 claims description 30
- 150000001413 amino acids Chemical group 0.000 claims description 29
- UIPYIFZPHJHVQB-KOKHDOHPSA-N C[C@H](N)C(O)=O.CC(C)C[C@H](N)C(O)=O.N[C@@H](CC(O)=O)C(O)=O Chemical group C[C@H](N)C(O)=O.CC(C)C[C@H](N)C(O)=O.N[C@@H](CC(O)=O)C(O)=O UIPYIFZPHJHVQB-KOKHDOHPSA-N 0.000 claims description 5
- 208000021048 Dianzani autoimmune lymphoproliferative disease Diseases 0.000 claims description 4
- 239000000411 inducer Substances 0.000 claims 3
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 105
- 239000007787 solid Substances 0.000 description 33
- 229940024606 amino acid Drugs 0.000 description 32
- 125000003275 alpha amino acid group Chemical group 0.000 description 30
- 235000001014 amino acid Nutrition 0.000 description 29
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 23
- 238000010511 deprotection reaction Methods 0.000 description 23
- VHYFNPMBLIVWCW-UHFFFAOYSA-N 4-Dimethylaminopyridine Chemical compound CN(C)C1=CC=NC=C1 VHYFNPMBLIVWCW-UHFFFAOYSA-N 0.000 description 20
- 238000000034 method Methods 0.000 description 19
- 238000006243 chemical reaction Methods 0.000 description 16
- 238000010586 diagram Methods 0.000 description 15
- 239000000499 gel Substances 0.000 description 15
- -1 hydroxymethylene group Chemical group 0.000 description 15
- 102000007079 Peptide Fragments Human genes 0.000 description 14
- 108010033276 Peptide Fragments Proteins 0.000 description 14
- 238000010647 peptide synthesis reaction Methods 0.000 description 14
- 239000002904 solvent Substances 0.000 description 14
- 238000003786 synthesis reaction Methods 0.000 description 14
- 230000006698 induction Effects 0.000 description 13
- 239000000203 mixture Substances 0.000 description 13
- 239000000047 product Substances 0.000 description 13
- 230000015572 biosynthetic process Effects 0.000 description 11
- 238000003756 stirring Methods 0.000 description 11
- BDNKZNFMNDZQMI-UHFFFAOYSA-N 1,3-diisopropylcarbodiimide Chemical compound CC(C)N=C=NC(C)C BDNKZNFMNDZQMI-UHFFFAOYSA-N 0.000 description 10
- 229960000549 4-dimethylaminophenol Drugs 0.000 description 10
- 238000004128 high performance liquid chromatography Methods 0.000 description 10
- 238000000746 purification Methods 0.000 description 10
- 238000000926 separation method Methods 0.000 description 10
- 238000001269 time-of-flight mass spectrometry Methods 0.000 description 10
- 230000000694 effects Effects 0.000 description 8
- 239000011734 sodium Substances 0.000 description 8
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 7
- 230000002209 hydrophobic effect Effects 0.000 description 7
- 239000007791 liquid phase Substances 0.000 description 7
- 239000000126 substance Substances 0.000 description 7
- OBMZMSLWNNWEJA-XNCRXQDQSA-N C1=CC=2C(C[C@@H]3NC(=O)[C@@H](NC(=O)[C@H](NC(=O)N(CC#CCN(CCCC[C@H](NC(=O)[C@@H](CC4=CC=CC=C4)NC3=O)C(=O)N)CC=C)NC(=O)[C@@H](N)C)CC3=CNC4=C3C=CC=C4)C)=CNC=2C=C1 Chemical compound C1=CC=2C(C[C@@H]3NC(=O)[C@@H](NC(=O)[C@H](NC(=O)N(CC#CCN(CCCC[C@H](NC(=O)[C@@H](CC4=CC=CC=C4)NC3=O)C(=O)N)CC=C)NC(=O)[C@@H](N)C)CC3=CNC4=C3C=CC=C4)C)=CNC=2C=C1 OBMZMSLWNNWEJA-XNCRXQDQSA-N 0.000 description 6
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 6
- 101710176384 Peptide 1 Proteins 0.000 description 6
- 239000013535 sea water Substances 0.000 description 6
- VZQHRKZCAZCACO-PYJNHQTQSA-N (2s)-2-[[(2s)-2-[2-[[(2s)-2-[[(2s)-2-amino-5-(diaminomethylideneamino)pentanoyl]amino]propanoyl]amino]prop-2-enoylamino]-3-methylbutanoyl]amino]propanoic acid Chemical compound OC(=O)[C@H](C)NC(=O)[C@H](C(C)C)NC(=O)C(=C)NC(=O)[C@H](C)NC(=O)[C@@H](N)CCCNC(N)=N VZQHRKZCAZCACO-PYJNHQTQSA-N 0.000 description 5
- 125000003088 (fluoren-9-ylmethoxy)carbonyl group Chemical group 0.000 description 5
- DLZKEQQWXODGGZ-KCJUWKMLSA-N 2-[[(2r)-2-[[(2s)-2-amino-3-(4-hydroxyphenyl)propanoyl]amino]propanoyl]amino]acetic acid Chemical compound OC(=O)CNC(=O)[C@@H](C)NC(=O)[C@@H](N)CC1=CC=C(O)C=C1 DLZKEQQWXODGGZ-KCJUWKMLSA-N 0.000 description 5
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 5
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 description 5
- OUYCCCASQSFEME-QMMMGPOBSA-N L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-QMMMGPOBSA-N 0.000 description 5
- UCMIRNVEIXFBKS-UHFFFAOYSA-N beta-alanine Chemical compound NCCC(O)=O UCMIRNVEIXFBKS-UHFFFAOYSA-N 0.000 description 5
- BTCSSZJGUNDROE-UHFFFAOYSA-N gamma-aminobutyric acid Chemical compound NCCCC(O)=O BTCSSZJGUNDROE-UHFFFAOYSA-N 0.000 description 5
- 125000006239 protecting group Chemical group 0.000 description 5
- FSYKKLYZXJSNPZ-UHFFFAOYSA-N sarcosine Chemical compound C[NH2+]CC([O-])=O FSYKKLYZXJSNPZ-UHFFFAOYSA-N 0.000 description 5
- ZDXPYRJPNDTMRX-VKHMYHEASA-N L-glutamine Chemical compound OC(=O)[C@@H](N)CCC(N)=O ZDXPYRJPNDTMRX-VKHMYHEASA-N 0.000 description 4
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 description 4
- NQRYJNQNLNOLGT-UHFFFAOYSA-N Piperidine Chemical compound C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 description 4
- 125000003277 amino group Chemical group 0.000 description 4
- 150000001875 compounds Chemical class 0.000 description 4
- 125000004122 cyclic group Chemical group 0.000 description 4
- COLNVLDHVKWLRT-QMMMGPOBSA-N phenylalanine group Chemical group N[C@@H](CC1=CC=CC=C1)C(=O)O COLNVLDHVKWLRT-QMMMGPOBSA-N 0.000 description 4
- 241000251468 Actinopterygii Species 0.000 description 3
- 239000004475 Arginine Substances 0.000 description 3
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 3
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 3
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 3
- ODKSFYDXXFIFQN-BYPYZUCNSA-P L-argininium(2+) Chemical compound NC(=[NH2+])NCCC[C@H]([NH3+])C(O)=O ODKSFYDXXFIFQN-BYPYZUCNSA-P 0.000 description 3
- DCXYFEDJOCDNAF-REOHCLBHSA-N L-asparagine Chemical compound OC(=O)[C@@H](N)CC(N)=O DCXYFEDJOCDNAF-REOHCLBHSA-N 0.000 description 3
- AGPKZVBTJJNPAG-WHFBIAKZSA-N L-isoleucine Chemical compound CC[C@H](C)[C@H](N)C(O)=O AGPKZVBTJJNPAG-WHFBIAKZSA-N 0.000 description 3
- AYFVYJQAPQTCCC-GBXIJSLDSA-N L-threonine Chemical compound C[C@@H](O)[C@H](N)C(O)=O AYFVYJQAPQTCCC-GBXIJSLDSA-N 0.000 description 3
- 239000004472 Lysine Substances 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- 102000007056 Recombinant Fusion Proteins Human genes 0.000 description 3
- 108010008281 Recombinant Fusion Proteins Proteins 0.000 description 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- KZSNJWFQEVHDMF-UHFFFAOYSA-N Valine Chemical compound CC(C)C(N)C(O)=O KZSNJWFQEVHDMF-UHFFFAOYSA-N 0.000 description 3
- QWCKQJZIFLGMSD-UHFFFAOYSA-N alpha-aminobutyric acid Chemical compound CCC(N)C(O)=O QWCKQJZIFLGMSD-UHFFFAOYSA-N 0.000 description 3
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 3
- 235000003704 aspartic acid Nutrition 0.000 description 3
- OQFSQFPPLPISGP-UHFFFAOYSA-N beta-carboxyaspartic acid Natural products OC(=O)C(N)C(C(O)=O)C(O)=O OQFSQFPPLPISGP-UHFFFAOYSA-N 0.000 description 3
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 3
- PMMYEEVYMWASQN-UHFFFAOYSA-N dl-hydroxyproline Natural products OC1C[NH2+]C(C([O-])=O)C1 PMMYEEVYMWASQN-UHFFFAOYSA-N 0.000 description 3
- WVDDGKGOMKODPV-UHFFFAOYSA-N hydroxymethyl benzene Natural products OCC1=CC=CC=C1 WVDDGKGOMKODPV-UHFFFAOYSA-N 0.000 description 3
- 125000001360 methionine group Chemical group N[C@@H](CCSC)C(=O)* 0.000 description 3
- 230000003287 optical effect Effects 0.000 description 3
- 108090000623 proteins and genes Proteins 0.000 description 3
- 102000004169 proteins and genes Human genes 0.000 description 3
- 239000007790 solid phase Substances 0.000 description 3
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 3
- FODJWPHPWBKDON-IBGZPJMESA-N (2s)-2-(9h-fluoren-9-ylmethoxycarbonylamino)-4-[(2-methylpropan-2-yl)oxy]-4-oxobutanoic acid Chemical compound C1=CC=C2C(COC(=O)N[C@@H](CC(=O)OC(C)(C)C)C(O)=O)C3=CC=CC=C3C2=C1 FODJWPHPWBKDON-IBGZPJMESA-N 0.000 description 2
- CBPJQFCAFFNICX-IBGZPJMESA-N (2s)-2-(9h-fluoren-9-ylmethoxycarbonylamino)-4-methylpentanoic acid Chemical compound C1=CC=C2C(COC(=O)N[C@@H](CC(C)C)C(O)=O)C3=CC=CC=C3C2=C1 CBPJQFCAFFNICX-IBGZPJMESA-N 0.000 description 2
- OTKXCALUHMPIGM-FQEVSTJZSA-N (2s)-2-(9h-fluoren-9-ylmethoxycarbonylamino)-5-[(2-methylpropan-2-yl)oxy]-5-oxopentanoic acid Chemical compound C1=CC=C2C(COC(=O)N[C@@H](CCC(=O)OC(C)(C)C)C(O)=O)C3=CC=CC=C3C2=C1 OTKXCALUHMPIGM-FQEVSTJZSA-N 0.000 description 2
- KSDTXRUIZMTBNV-INIZCTEOSA-N (2s)-2-(9h-fluoren-9-ylmethoxycarbonylamino)butanedioic acid Chemical compound C1=CC=C2C(COC(=O)N[C@@H](CC(=O)O)C(O)=O)C3=CC=CC=C3C2=C1 KSDTXRUIZMTBNV-INIZCTEOSA-N 0.000 description 2
- FUOOLUPWFVMBKG-UHFFFAOYSA-N 2-Aminoisobutyric acid Chemical compound CC(C)(N)C(O)=O FUOOLUPWFVMBKG-UHFFFAOYSA-N 0.000 description 2
- OYIFNHCXNCRBQI-UHFFFAOYSA-N 2-aminoadipic acid Chemical compound OC(=O)C(N)CCCC(O)=O OYIFNHCXNCRBQI-UHFFFAOYSA-N 0.000 description 2
- RDFMDVXONNIGBC-UHFFFAOYSA-N 2-aminoheptanoic acid Chemical compound CCCCCC(N)C(O)=O RDFMDVXONNIGBC-UHFFFAOYSA-N 0.000 description 2
- PECYZEOJVXMISF-UHFFFAOYSA-N 3-aminoalanine Chemical compound [NH3+]CC(N)C([O-])=O PECYZEOJVXMISF-UHFFFAOYSA-N 0.000 description 2
- 241001523707 Balanidae Species 0.000 description 2
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 2
- JUQLUIFNNFIIKC-YFKPBYRVSA-N L-2-aminopimelic acid Chemical compound OC(=O)[C@@H](N)CCCCC(O)=O JUQLUIFNNFIIKC-YFKPBYRVSA-N 0.000 description 2
- AHLPHDHHMVZTML-BYPYZUCNSA-N L-Ornithine Chemical compound NCCC[C@H](N)C(O)=O AHLPHDHHMVZTML-BYPYZUCNSA-N 0.000 description 2
- SNDPXSYFESPGGJ-UHFFFAOYSA-N L-norVal-OH Natural products CCCC(N)C(O)=O SNDPXSYFESPGGJ-UHFFFAOYSA-N 0.000 description 2
- LRQKBLKVPFOOQJ-YFKPBYRVSA-N L-norleucine Chemical compound CCCC[C@H]([NH3+])C([O-])=O LRQKBLKVPFOOQJ-YFKPBYRVSA-N 0.000 description 2
- ROHFNLRQFUQHCH-UHFFFAOYSA-N Leucine Natural products CC(C)CC(N)C(O)=O ROHFNLRQFUQHCH-UHFFFAOYSA-N 0.000 description 2
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 2
- OLNLSTNFRUFTLM-UHFFFAOYSA-N N-ethylasparagine Chemical compound CCNC(C(O)=O)CC(N)=O OLNLSTNFRUFTLM-UHFFFAOYSA-N 0.000 description 2
- YPIGGYHFMKJNKV-UHFFFAOYSA-N N-ethylglycine Chemical compound CC[NH2+]CC([O-])=O YPIGGYHFMKJNKV-UHFFFAOYSA-N 0.000 description 2
- AKCRVYNORCOYQT-YFKPBYRVSA-N N-methyl-L-valine Chemical compound CN[C@@H](C(C)C)C(O)=O AKCRVYNORCOYQT-YFKPBYRVSA-N 0.000 description 2
- KSPIYJQBLVDRRI-UHFFFAOYSA-N N-methylisoleucine Chemical compound CCC(C)C(NC)C(O)=O KSPIYJQBLVDRRI-UHFFFAOYSA-N 0.000 description 2
- AHLPHDHHMVZTML-UHFFFAOYSA-N Orn-delta-NH2 Natural products NCCCC(N)C(O)=O AHLPHDHHMVZTML-UHFFFAOYSA-N 0.000 description 2
- UTJLXEIPEHZYQJ-UHFFFAOYSA-N Ornithine Natural products OC(=O)C(C)CCCN UTJLXEIPEHZYQJ-UHFFFAOYSA-N 0.000 description 2
- 108010077895 Sarcosine Proteins 0.000 description 2
- 241001454402 Tetraclita Species 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 230000002378 acidificating effect Effects 0.000 description 2
- 125000003295 alanine group Chemical group N[C@@H](C)C(=O)* 0.000 description 2
- 125000001931 aliphatic group Chemical group 0.000 description 2
- 125000003368 amide group Chemical group 0.000 description 2
- 150000001555 benzenes Chemical class 0.000 description 2
- 229940000635 beta-alanine Drugs 0.000 description 2
- 238000005859 coupling reaction Methods 0.000 description 2
- YSMODUONRAFBET-UHFFFAOYSA-N delta-DL-hydroxylysine Natural products NCC(O)CCC(N)C(O)=O YSMODUONRAFBET-UHFFFAOYSA-N 0.000 description 2
- VEVRNHHLCPGNDU-MUGJNUQGSA-O desmosine Chemical compound OC(=O)[C@@H](N)CCCC[N+]1=CC(CC[C@H](N)C(O)=O)=C(CCC[C@H](N)C(O)=O)C(CC[C@H](N)C(O)=O)=C1 VEVRNHHLCPGNDU-MUGJNUQGSA-O 0.000 description 2
- 235000013922 glutamic acid Nutrition 0.000 description 2
- 239000004220 glutamic acid Substances 0.000 description 2
- 125000000291 glutamic acid group Chemical group N[C@@H](CCC(O)=O)C(=O)* 0.000 description 2
- ZDXPYRJPNDTMRX-UHFFFAOYSA-N glutamine Natural products OC(=O)C(N)CCC(N)=O ZDXPYRJPNDTMRX-UHFFFAOYSA-N 0.000 description 2
- HNDVDQJCIGZPNO-UHFFFAOYSA-N histidine Natural products OC(=O)C(N)CC1=CN=CN1 HNDVDQJCIGZPNO-UHFFFAOYSA-N 0.000 description 2
- 150000002430 hydrocarbons Chemical group 0.000 description 2
- 125000002883 imidazolyl group Chemical group 0.000 description 2
- RGXCTRIQQODGIZ-UHFFFAOYSA-O isodesmosine Chemical compound OC(=O)C(N)CCCC[N+]1=CC(CCC(N)C(O)=O)=CC(CCC(N)C(O)=O)=C1CCCC(N)C(O)=O RGXCTRIQQODGIZ-UHFFFAOYSA-O 0.000 description 2
- 229960000310 isoleucine Drugs 0.000 description 2
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 2
- 235000005772 leucine Nutrition 0.000 description 2
- 125000003588 lysine group Chemical group [H]N([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])(N([H])[H])C(*)=O 0.000 description 2
- 238000004519 manufacturing process Methods 0.000 description 2
- 125000001570 methylene group Chemical group [H]C([H])([*:1])[*:2] 0.000 description 2
- 230000007935 neutral effect Effects 0.000 description 2
- 229960003104 ornithine Drugs 0.000 description 2
- 239000012071 phase Substances 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 235000018102 proteins Nutrition 0.000 description 2
- UQDJGEHQDNVPGU-UHFFFAOYSA-N serine phosphoethanolamine Chemical compound [NH3+]CCOP([O-])(=O)OCC([NH3+])C([O-])=O UQDJGEHQDNVPGU-UHFFFAOYSA-N 0.000 description 2
- 241000894007 species Species 0.000 description 2
- OUYCCCASQSFEME-UHFFFAOYSA-N tyrosine Natural products OC(=O)C(N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-UHFFFAOYSA-N 0.000 description 2
- XUJHKPSBHDQIOD-UHFFFAOYSA-N (2-bromo-7,7-dimethyl-3-oxo-4-bicyclo[2.2.1]heptanyl)methanesulfonic acid Chemical compound C1CC2(CS(O)(=O)=O)C(=O)C(Br)C1C2(C)C XUJHKPSBHDQIOD-UHFFFAOYSA-N 0.000 description 1
- QBYIENPQHBMVBV-HFEGYEGKSA-N (2R)-2-hydroxy-2-phenylacetic acid Chemical compound O[C@@H](C(O)=O)c1ccccc1.O[C@@H](C(O)=O)c1ccccc1 QBYIENPQHBMVBV-HFEGYEGKSA-N 0.000 description 1
- BJBUEDPLEOHJGE-UHFFFAOYSA-N (2R,3S)-3-Hydroxy-2-pyrolidinecarboxylic acid Natural products OC1CCNC1C(O)=O BJBUEDPLEOHJGE-UHFFFAOYSA-N 0.000 description 1
- XXMYDXUIZKNHDT-QNGWXLTQSA-N (2s)-2-(9h-fluoren-9-ylmethoxycarbonylamino)-3-(1-tritylimidazol-4-yl)propanoic acid Chemical compound C([C@@H](C(=O)O)NC(=O)OCC1C2=CC=CC=C2C2=CC=CC=C21)C(N=C1)=CN1C(C=1C=CC=CC=1)(C=1C=CC=CC=1)C1=CC=CC=C1 XXMYDXUIZKNHDT-QNGWXLTQSA-N 0.000 description 1
- ADOHASQZJSJZBT-SANMLTNESA-N (2s)-2-(9h-fluoren-9-ylmethoxycarbonylamino)-3-[1-[(2-methylpropan-2-yl)oxycarbonyl]indol-3-yl]propanoic acid Chemical compound C12=CC=CC=C2N(C(=O)OC(C)(C)C)C=C1C[C@@H](C(O)=O)NC(=O)OCC1C2=CC=CC=C2C2=CC=CC=C21 ADOHASQZJSJZBT-SANMLTNESA-N 0.000 description 1
- YMYLGVIBUGDMLT-QMMMGPOBSA-N (2s)-2-(phenylmethoxyamino)propanoic acid Chemical compound OC(=O)[C@H](C)NOCC1=CC=CC=C1 YMYLGVIBUGDMLT-QMMMGPOBSA-N 0.000 description 1
- DVBUCBXGDWWXNY-SFHVURJKSA-N (2s)-5-(diaminomethylideneamino)-2-(9h-fluoren-9-ylmethoxycarbonylamino)pentanoic acid Chemical compound C1=CC=C2C(COC(=O)N[C@@H](CCCN=C(N)N)C(O)=O)C3=CC=CC=C3C2=C1 DVBUCBXGDWWXNY-SFHVURJKSA-N 0.000 description 1
- HNICLNKVURBTKV-NDEPHWFRSA-N (2s)-5-[[amino-[(2,2,4,6,7-pentamethyl-3h-1-benzofuran-5-yl)sulfonylamino]methylidene]amino]-2-(9h-fluoren-9-ylmethoxycarbonylamino)pentanoic acid Chemical compound C12=CC=CC=C2C2=CC=CC=C2C1COC(=O)N[C@H](C(O)=O)CCCN=C(N)NS(=O)(=O)C1=C(C)C(C)=C2OC(C)(C)CC2=C1C HNICLNKVURBTKV-NDEPHWFRSA-N 0.000 description 1
- QXVFEIPAZSXRGM-DJJJIMSYSA-N (2s,3s)-2-(9h-fluoren-9-ylmethoxycarbonylamino)-3-methylpentanoic acid Chemical compound C1=CC=C2C(COC(=O)N[C@@H]([C@@H](C)CC)C(O)=O)C3=CC=CC=C3C2=C1 QXVFEIPAZSXRGM-DJJJIMSYSA-N 0.000 description 1
- XABCFXXGZPWJQP-BYPYZUCNSA-N (S)-3-aminoadipic acid Chemical compound OC(=O)C[C@@H](N)CCC(O)=O XABCFXXGZPWJQP-BYPYZUCNSA-N 0.000 description 1
- MIOPJNTWMNEORI-GMSGAONNSA-N (S)-camphorsulfonic acid Chemical compound C1C[C@@]2(CS(O)(=O)=O)C(=O)C[C@@H]1C2(C)C MIOPJNTWMNEORI-GMSGAONNSA-N 0.000 description 1
- BJEPYKJPYRNKOW-REOHCLBHSA-N (S)-malic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O BJEPYKJPYRNKOW-REOHCLBHSA-N 0.000 description 1
- UKAUYVFTDYCKQA-UHFFFAOYSA-N -2-Amino-4-hydroxybutanoic acid Natural products OC(=O)C(N)CCO UKAUYVFTDYCKQA-UHFFFAOYSA-N 0.000 description 1
- JHTPBGFVWWSHDL-UHFFFAOYSA-N 1,4-dichloro-2-isothiocyanatobenzene Chemical compound ClC1=CC=C(Cl)C(N=C=S)=C1 JHTPBGFVWWSHDL-UHFFFAOYSA-N 0.000 description 1
- IXPNQXFRVYWDDI-UHFFFAOYSA-N 1-methyl-2,4-dioxo-1,3-diazinane-5-carboximidamide Chemical compound CN1CC(C(N)=N)C(=O)NC1=O IXPNQXFRVYWDDI-UHFFFAOYSA-N 0.000 description 1
- OGNSCSPNOLGXSM-UHFFFAOYSA-N 2,4-diaminobutyric acid Chemical compound NCCC(N)C(O)=O OGNSCSPNOLGXSM-UHFFFAOYSA-N 0.000 description 1
- GMKMEZVLHJARHF-UHFFFAOYSA-N 2,6-diaminopimelic acid Chemical compound OC(=O)C(N)CCCC(N)C(O)=O GMKMEZVLHJARHF-UHFFFAOYSA-N 0.000 description 1
- XABCFXXGZPWJQP-UHFFFAOYSA-N 3-aminoadipic acid Chemical compound OC(=O)CC(N)CCC(O)=O XABCFXXGZPWJQP-UHFFFAOYSA-N 0.000 description 1
- QCHPKSFMDHPSNR-UHFFFAOYSA-N 3-aminoisobutyric acid Chemical compound NCC(C)C(O)=O QCHPKSFMDHPSNR-UHFFFAOYSA-N 0.000 description 1
- FWMNVWWHGCHHJJ-SKKKGAJSSA-N 4-amino-1-[(2r)-6-amino-2-[[(2r)-2-[[(2r)-2-[[(2r)-2-amino-3-phenylpropanoyl]amino]-3-phenylpropanoyl]amino]-4-methylpentanoyl]amino]hexanoyl]piperidine-4-carboxylic acid Chemical compound C([C@H](C(=O)N[C@H](CC(C)C)C(=O)N[C@H](CCCCN)C(=O)N1CCC(N)(CC1)C(O)=O)NC(=O)[C@H](N)CC=1C=CC=CC=1)C1=CC=CC=C1 FWMNVWWHGCHHJJ-SKKKGAJSSA-N 0.000 description 1
- JJMDCOVWQOJGCB-UHFFFAOYSA-N 5-aminopentanoic acid Chemical compound [NH3+]CCCCC([O-])=O JJMDCOVWQOJGCB-UHFFFAOYSA-N 0.000 description 1
- SLXKOJJOQWFEFD-UHFFFAOYSA-N 6-aminohexanoic acid Chemical compound NCCCCCC(O)=O SLXKOJJOQWFEFD-UHFFFAOYSA-N 0.000 description 1
- HRPVXLWXLXDGHG-UHFFFAOYSA-N Acrylamide Chemical compound NC(=O)C=C HRPVXLWXLXDGHG-UHFFFAOYSA-N 0.000 description 1
- 229920000936 Agarose Polymers 0.000 description 1
- 241001168843 Amphibalanus Species 0.000 description 1
- 241000739363 Archaeobalanidae Species 0.000 description 1
- 241000238421 Arthropoda Species 0.000 description 1
- DCXYFEDJOCDNAF-UHFFFAOYSA-N Asparagine Natural products OC(=O)C(N)CC(N)=O DCXYFEDJOCDNAF-UHFFFAOYSA-N 0.000 description 1
- 241000238588 Balanus Species 0.000 description 1
- LSNNMFCWUKXFEE-UHFFFAOYSA-M Bisulfite Chemical compound OS([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-M 0.000 description 1
- 125000001433 C-terminal amino-acid group Chemical group 0.000 description 1
- 239000004215 Carbon black (E152) Substances 0.000 description 1
- 241001523453 Chelonibia Species 0.000 description 1
- 241001189840 Chionelasmatidae Species 0.000 description 1
- 241001523447 Chthamalidae Species 0.000 description 1
- 241001523449 Chthamalus Species 0.000 description 1
- 241001193189 Coronula Species 0.000 description 1
- 241001523451 Coronulidae Species 0.000 description 1
- 241000238424 Crustacea Species 0.000 description 1
- ZAFNJMIOTHYJRJ-UHFFFAOYSA-N Diisopropyl ether Chemical compound CC(C)OC(C)C ZAFNJMIOTHYJRJ-UHFFFAOYSA-N 0.000 description 1
- 241000588724 Escherichia coli Species 0.000 description 1
- 229920002148 Gellan gum Polymers 0.000 description 1
- 241000238631 Hexapoda Species 0.000 description 1
- LCWXJXMHJVIJFK-UHFFFAOYSA-N Hydroxylysine Natural products NCC(O)CC(N)CC(O)=O LCWXJXMHJVIJFK-UHFFFAOYSA-N 0.000 description 1
- PMMYEEVYMWASQN-DMTCNVIQSA-N Hydroxyproline Chemical compound O[C@H]1CN[C@H](C(O)=O)C1 PMMYEEVYMWASQN-DMTCNVIQSA-N 0.000 description 1
- SNDPXSYFESPGGJ-BYPYZUCNSA-N L-2-aminopentanoic acid Chemical compound CCC[C@H](N)C(O)=O SNDPXSYFESPGGJ-BYPYZUCNSA-N 0.000 description 1
- QNAYBMKLOCPYGJ-REOHCLBHSA-N L-alanine Chemical compound C[C@H](N)C(O)=O QNAYBMKLOCPYGJ-REOHCLBHSA-N 0.000 description 1
- FFFHZYDWPBMWHY-VKHMYHEASA-N L-homocysteine Chemical compound OC(=O)[C@@H](N)CCS FFFHZYDWPBMWHY-VKHMYHEASA-N 0.000 description 1
- UKAUYVFTDYCKQA-VKHMYHEASA-N L-homoserine Chemical compound OC(=O)[C@@H](N)CCO UKAUYVFTDYCKQA-VKHMYHEASA-N 0.000 description 1
- FFEARJCKVFRZRR-BYPYZUCNSA-N L-methionine Chemical compound CSCC[C@H](N)C(O)=O FFEARJCKVFRZRR-BYPYZUCNSA-N 0.000 description 1
- QIVBCDIJIAJPQS-VIFPVBQESA-N L-tryptophane Chemical compound C1=CC=C2C(C[C@H](N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-VIFPVBQESA-N 0.000 description 1
- KZSNJWFQEVHDMF-BYPYZUCNSA-N L-valine Chemical compound CC(C)[C@H](N)C(O)=O KZSNJWFQEVHDMF-BYPYZUCNSA-N 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-N Malonic acid Chemical compound OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 1
- 241000238589 Megabalanus Species 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- PQNASZJZHFPQLE-LURJTMIESA-N N(6)-methyl-L-lysine Chemical compound CNCCCC[C@H](N)C(O)=O PQNASZJZHFPQLE-LURJTMIESA-N 0.000 description 1
- 108010065338 N-ethylglycine Proteins 0.000 description 1
- KSPIYJQBLVDRRI-WDSKDSINSA-N N-methyl-L-isoleucine Chemical compound CC[C@H](C)[C@H](NC)C(O)=O KSPIYJQBLVDRRI-WDSKDSINSA-N 0.000 description 1
- 125000000729 N-terminal amino-acid group Chemical group 0.000 description 1
- 241001193172 Platylepadidae Species 0.000 description 1
- 239000004793 Polystyrene Substances 0.000 description 1
- 241000131106 Pyrgomatidae Species 0.000 description 1
- IWYDHOAUDWTVEP-UHFFFAOYSA-N R-2-phenyl-2-hydroxyacetic acid Natural products OC(=O)C(O)C1=CC=CC=C1 IWYDHOAUDWTVEP-UHFFFAOYSA-N 0.000 description 1
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 1
- MTCFGRXMJLQNBG-UHFFFAOYSA-N Serine Natural products OCC(N)C(O)=O MTCFGRXMJLQNBG-UHFFFAOYSA-N 0.000 description 1
- 244000191761 Sida cordifolia Species 0.000 description 1
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- 241000036188 Tetraclitella Species 0.000 description 1
- AYFVYJQAPQTCCC-UHFFFAOYSA-N Threonine Natural products CC(O)C(N)C(O)=O AYFVYJQAPQTCCC-UHFFFAOYSA-N 0.000 description 1
- 239000004473 Threonine Substances 0.000 description 1
- QIVBCDIJIAJPQS-UHFFFAOYSA-N Tryptophan Natural products C1=CC=C2C(CC(N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-UHFFFAOYSA-N 0.000 description 1
- PAKFDDLYENSQHL-UHFFFAOYSA-N [2,4-di(docosoxy)phenyl]methanol Chemical compound CCCCCCCCCCCCCCCCCCCCCCOC1=CC=C(CO)C(OCCCCCCCCCCCCCCCCCCCCCC)=C1 PAKFDDLYENSQHL-UHFFFAOYSA-N 0.000 description 1
- GPDHNZNLPKYHCN-DZOOLQPHSA-N [[(z)-(1-cyano-2-ethoxy-2-oxoethylidene)amino]oxy-morpholin-4-ylmethylidene]-dimethylazanium;hexafluorophosphate Chemical compound F[P-](F)(F)(F)(F)F.CCOC(=O)C(\C#N)=N/OC(=[N+](C)C)N1CCOCC1 GPDHNZNLPKYHCN-DZOOLQPHSA-N 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- 235000004279 alanine Nutrition 0.000 description 1
- 230000001476 alcoholic effect Effects 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 150000001371 alpha-amino acids Chemical class 0.000 description 1
- 235000008206 alpha-amino acids Nutrition 0.000 description 1
- BJEPYKJPYRNKOW-UHFFFAOYSA-N alpha-hydroxysuccinic acid Natural products OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 1
- 229960002684 aminocaproic acid Drugs 0.000 description 1
- 239000000010 aprotic solvent Substances 0.000 description 1
- 125000000637 arginyl group Chemical group N[C@@H](CCCNC(N)=N)C(=O)* 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 235000009582 asparagine Nutrition 0.000 description 1
- 229960001230 asparagine Drugs 0.000 description 1
- 125000000613 asparagine group Chemical group N[C@@H](CC(N)=O)C(=O)* 0.000 description 1
- 239000005667 attractant Substances 0.000 description 1
- 235000019445 benzyl alcohol Nutrition 0.000 description 1
- 229960004217 benzyl alcohol Drugs 0.000 description 1
- 150000001576 beta-amino acids Chemical class 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 125000004432 carbon atom Chemical group C* 0.000 description 1
- 235000010418 carrageenan Nutrition 0.000 description 1
- 239000000679 carrageenan Substances 0.000 description 1
- 229920001525 carrageenan Polymers 0.000 description 1
- 229940113118 carrageenan Drugs 0.000 description 1
- 210000004027 cell Anatomy 0.000 description 1
- 239000004568 cement Substances 0.000 description 1
- 238000005119 centrifugation Methods 0.000 description 1
- 230000031902 chemoattractant activity Effects 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- BJBUEDPLEOHJGE-IUYQGCFVSA-N cis-3-hydroxy-D-proline zwitterion Chemical compound O[C@H]1CCN[C@H]1C(O)=O BJBUEDPLEOHJGE-IUYQGCFVSA-N 0.000 description 1
- 238000004440 column chromatography Methods 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 238000010168 coupling process Methods 0.000 description 1
- 238000004132 cross linking Methods 0.000 description 1
- 238000012258 culturing Methods 0.000 description 1
- 125000000151 cysteine group Chemical group N[C@@H](CS)C(=O)* 0.000 description 1
- YSMODUONRAFBET-UHNVWZDZSA-N erythro-5-hydroxy-L-lysine Chemical compound NC[C@H](O)CC[C@H](N)C(O)=O YSMODUONRAFBET-UHNVWZDZSA-N 0.000 description 1
- JBTWLSYIZRCDFO-UHFFFAOYSA-N ethyl methyl carbonate Chemical compound CCOC(=O)OC JBTWLSYIZRCDFO-UHFFFAOYSA-N 0.000 description 1
- 239000000706 filtrate Substances 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 229960003692 gamma aminobutyric acid Drugs 0.000 description 1
- 238000001641 gel filtration chromatography Methods 0.000 description 1
- 238000001879 gelation Methods 0.000 description 1
- 235000010492 gellan gum Nutrition 0.000 description 1
- 239000000216 gellan gum Substances 0.000 description 1
- 210000004907 gland Anatomy 0.000 description 1
- 125000000404 glutamine group Chemical group N[C@@H](CCC(N)=O)C(=O)* 0.000 description 1
- 102000035122 glycosylated proteins Human genes 0.000 description 1
- 108091005608 glycosylated proteins Proteins 0.000 description 1
- 125000003630 glycyl group Chemical group [H]N([H])C([H])([H])C(*)=O 0.000 description 1
- 230000005484 gravity Effects 0.000 description 1
- 125000000487 histidyl group Chemical group [H]N([H])C(C(=O)O*)C([H])([H])C1=C([H])N([H])C([H])=N1 0.000 description 1
- 229930195733 hydrocarbon Natural products 0.000 description 1
- 239000000017 hydrogel Substances 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- QJHBJHUKURJDLG-UHFFFAOYSA-N hydroxy-L-lysine Natural products NCCCCC(NO)C(O)=O QJHBJHUKURJDLG-UHFFFAOYSA-N 0.000 description 1
- 229960002591 hydroxyproline Drugs 0.000 description 1
- 125000001841 imino group Chemical group [H]N=* 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 239000012442 inert solvent Substances 0.000 description 1
- 238000005342 ion exchange Methods 0.000 description 1
- AGPKZVBTJJNPAG-UHFFFAOYSA-N isoleucine Natural products CCC(C)C(N)C(O)=O AGPKZVBTJJNPAG-UHFFFAOYSA-N 0.000 description 1
- 125000000741 isoleucyl group Chemical group [H]N([H])C(C(C([H])([H])[H])C([H])([H])C([H])([H])[H])C(=O)O* 0.000 description 1
- 239000004310 lactic acid Substances 0.000 description 1
- 235000014655 lactic acid Nutrition 0.000 description 1
- 125000001909 leucine group Chemical group [H]N(*)C(C(*)=O)C([H])([H])C(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 239000001630 malic acid Substances 0.000 description 1
- 235000011090 malic acid Nutrition 0.000 description 1
- 229960002510 mandelic acid Drugs 0.000 description 1
- 229930182817 methionine Natural products 0.000 description 1
- 150000002762 monocarboxylic acid derivatives Chemical class 0.000 description 1
- 102000039446 nucleic acids Human genes 0.000 description 1
- 108020004707 nucleic acids Proteins 0.000 description 1
- 150000007523 nucleic acids Chemical class 0.000 description 1
- 239000003973 paint Substances 0.000 description 1
- 235000010987 pectin Nutrition 0.000 description 1
- 229920001277 pectin Polymers 0.000 description 1
- 239000001814 pectin Substances 0.000 description 1
- COLNVLDHVKWLRT-UHFFFAOYSA-N phenylalanine Natural products OC(=O)C(N)CC1=CC=CC=C1 COLNVLDHVKWLRT-UHFFFAOYSA-N 0.000 description 1
- 239000003016 pheromone Substances 0.000 description 1
- 229920002223 polystyrene Polymers 0.000 description 1
- 125000001500 prolyl group Chemical group [H]N1C([H])(C(=O)[*])C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 230000004952 protein activity Effects 0.000 description 1
- 239000011347 resin Substances 0.000 description 1
- 229920005989 resin Polymers 0.000 description 1
- 238000004366 reverse phase liquid chromatography Methods 0.000 description 1
- 238000004007 reversed phase HPLC Methods 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 229940043230 sarcosine Drugs 0.000 description 1
- 125000003607 serino group Chemical group [H]N([H])[C@]([H])(C(=O)[*])C(O[H])([H])[H] 0.000 description 1
- 238000010898 silica gel chromatography Methods 0.000 description 1
- 235000010413 sodium alginate Nutrition 0.000 description 1
- 239000000661 sodium alginate Substances 0.000 description 1
- 229940005550 sodium alginate Drugs 0.000 description 1
- 238000010532 solid phase synthesis reaction Methods 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 238000000967 suction filtration Methods 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 230000004083 survival effect Effects 0.000 description 1
- 230000002194 synthesizing effect Effects 0.000 description 1
- 235000002906 tartaric acid Nutrition 0.000 description 1
- 239000011975 tartaric acid Substances 0.000 description 1
- 125000000341 threoninyl group Chemical group [H]OC([H])(C([H])([H])[H])C([H])(N([H])[H])C(*)=O 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 125000000430 tryptophan group Chemical group [H]N([H])C(C(=O)O*)C([H])([H])C1=C([H])N([H])C2=C([H])C([H])=C([H])C([H])=C12 0.000 description 1
- 125000001493 tyrosinyl group Chemical group [H]OC1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])(N([H])[H])C(*)=O 0.000 description 1
- 239000004474 valine Substances 0.000 description 1
- 125000002987 valine group Chemical group [H]N([H])C([H])(C(*)=O)C([H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- 239000002699 waste material Substances 0.000 description 1
- UHVMMEOXYDMDKI-JKYCWFKZSA-L zinc;1-(5-cyanopyridin-2-yl)-3-[(1s,2s)-2-(6-fluoro-2-hydroxy-3-propanoylphenyl)cyclopropyl]urea;diacetate Chemical compound [Zn+2].CC([O-])=O.CC([O-])=O.CCC(=O)C1=CC=C(F)C([C@H]2[C@H](C2)NC(=O)NC=2N=CC(=CC=2)C#N)=C1O UHVMMEOXYDMDKI-JKYCWFKZSA-L 0.000 description 1
Landscapes
- Catching Or Destruction (AREA)
- Peptides Or Proteins (AREA)
Description
本発明は、フジツボ類キプリス幼生の付着を誘引する機能を有するペプチド、及び当該ペプチドを利用したフジツボ類トラップ装置に関する。 The present invention relates to a peptide that has the function of inducing the attachment of barnacle cypris larvae, and a barnacle trapping device that uses the peptide.
フジツボ類は自由遊泳性のノープリウス幼生として孵化し、海水中の植物プランクトンなどを捕食する。その後、約1ヶ月程度で、キプリス幼生に変態する。キプリス幼生は、既に成体が固着生活を営んでいる場所が見つかると、その近傍で頭部の触角にあるセメント腺から固着物質を分泌して接着する。このように、フジツボ類は、種の存続のために同種が近接して生息するといった特徴を有する。 Barnacles hatch as free-swimming nauplii larvae that feed on phytoplankton and other organisms in seawater. After about one month, they metamorphose into cypris larvae. When a cypris larva finds a location where an adult is already living a sessile life, it secretes an adhesive substance from the cement glands in the antennae on its head and adheres to the location nearby. In this way, barnacles have the characteristic of living in close proximity to the same species in order to ensure the survival of the species.
これに関して、キプリス幼生は、フジツボ成体が分泌するフェロモン様の物質に誘起されて、フジツボ成体の近くに生息するといわれている。実際に、フジツボ成体の抽出液より、76、88及び98KDaの3個のサブユニットからなる糖鎖結合型タンパク質としてSettlement Inducing Protein Complex(SIPC)が単離されている(非特許文献1:Matsumura, K. et al., J. Exp. Zool., 1998, 281及び非特許文献2:Dreanno, C. et al., Proc. Natl. Acad. Sci, USA, 2006, 103, 14396)。 In this regard, it is said that cypris larvae are attracted by a pheromone-like substance secreted by adult barnacles and live near the adult barnacles. In fact, Settlement Inducing Protein Complex (SIPC) has been isolated from the extract of adult barnacles as a glycosylated protein consisting of three subunits of 76, 88, and 98 KDa (Non-Patent Document 1: Matsumura, K. et al., J. Exp. Zool., 1998, 281 and Non-Patent Document 2: Dreanno, C. et al., Proc. Natl. Acad. Sci, USA, 2006, 103, 14396).
また、フジツボ成体が分泌するフェロモン様の物質として、SIPCよりも拡散性の高い、分子量32KDaのWaterborne Settlement Pheromone(WSP)も単離されている(非特許文献3:Endo, N. et al., Biofouling, 2009, 25, 429及び特許文献1:特許第5931584号)。 In addition, Waterborne Settlement Pheromone (WSP), a pheromone-like substance secreted by adult barnacles and with a molecular weight of 32 KDa that is more diffusible than SIPC, has also been isolated (Non-Patent Document 3: Endo, N. et al., Biofouling, 2009, 25, 429 and Patent Document 1: Japanese Patent No. 5931584).
しかしながら、上述したSIPC及びWSPは、分子量が大きいことから化学合成により製造することは非常に困難であり、コスト面から非現実的であった。また、上述したSIPC及びWSPを組み換えタンパク質として製造するとしても、組み換えタンパク質を産生する生物の培養や、組み換えタンパク質の分離精製等が必要でありコストがかかるといった問題がある。 However, because the above-mentioned SIPC and WSP have large molecular weights, it is extremely difficult to produce them by chemical synthesis, and this is unrealistic from a cost perspective. Furthermore, even if the above-mentioned SIPC and WSP were to be produced as recombinant proteins, there would be problems such as the need to cultivate organisms that produce the recombinant proteins and to separate and purify the recombinant proteins, which is costly.
以上のように、上述したSIPC及びWSPといったフジツボ類に対する誘起物質は、製造コストの面から現実的に利用することが困難であるといった問題があった。そこで、本発明は、上述した実情に鑑み、低コストで製造することができる、フジツボ類に対する誘起能を有する新規物質及びその利用を提供することを目的とする。 As described above, there is a problem with the above-mentioned SIPC and WSP, which are inducing substances for barnacles, in that they are difficult to use in practice due to the cost of production. In view of the above-mentioned circumstances, the present invention aims to provide a new substance that can be produced at low cost and has the ability to induce barnacles, and the use of the same.
上述した目的を達成するために本発明者等が鋭意検討した結果、所定の配列を有するペプチド断片がフジツボ類に対する誘起物質として機能することを見いだし、本発明を完成するに至った。 As a result of intensive research conducted by the inventors in order to achieve the above-mentioned objective, they discovered that a peptide fragment having a specific sequence functions as an attractant for barnacles, and thus completed the present invention.
本発明は以下を包含する。
(1)DAL(アスパラギン酸-アラニン-ロイシン)配列を含む3~12アミノ酸残基のアミノ酸配列を含む、フジツボ類キプリス幼生の付着誘起ペプチド。
(2)上記のアミノ酸配列は、ELQDALDIHWRE(配列番号1)から選択されることを特徴とする(1)記載のフジツボ類キプリス幼生の付着誘起ペプチド。
(3)上記アミノ酸配列は、ELQDALDIHWRE(配列番号1)、ELQDAL(配列番号2)、LQDALD(配列番号3)、QDALDI(配列番号4)、DALDIH(配列番号5)、DAL(配列番号9)及びDALD(配列番号10)からなる群から選ばれる1つのアミノ酸配列であることを特徴とする(1)記載のフジツボ類キプリス幼生の付着誘起ペプチド。
(4)上記(1)乃至(3)いずれかに記載のフジツボ類キプリス幼生の付着誘起ペプチドを備えるフジツボ類トラップ装置。
(5)上記フジツボ類キプリス幼生の付着誘起ペプチドを含浸させたゲルを備えることを特徴とする(4)記載のフジツボ類トラップ装置。
The present invention encompasses the following:
(1) A peptide that induces adhesion of barnacle cypris larvae, which comprises an amino acid sequence of 3 to 12 amino acid residues including the DAL (aspartic acid-alanine-leucine) sequence.
(2) The adhesion-inducing peptide for barnacle cypris larvae according to (1), characterized in that the amino acid sequence is selected from ELQDALDIHWRE (SEQ ID NO: 1).
(3) The adhesion-inducing peptide of barnacle cypris larvae described in (1), characterized in that the amino acid sequence is one amino acid sequence selected from the group consisting of ELQDALDIHWRE (sequence number 1), ELQDAL (sequence number 2), LQDALD (sequence number 3), QDALDI (sequence number 4), DALDIH (sequence number 5), DAL (sequence number 9) and DALD (sequence number 10).
(4) A barnacle trapping device comprising the attachment-inducing peptide for barnacle cypris larvae described in any one of (1) to (3) above.
(5) A barnacle trapping device according to (4), comprising a gel impregnated with a peptide that induces adhesion of the barnacle cypris larvae.
本発明に係るフジツボ類キプリス幼生の付着誘起ペプチドは、3~12アミノ酸残基の範囲の長さを有するペプチド断片であり、低コストに化学合成することができる。また、フジツボ類キプリス幼生の付着誘起ペプチドは安全性に優れるため、フジツボの付着を防止したい海洋に広く適用することができる。 The peptides inducing attachment of barnacle cypris larvae according to the present invention are peptide fragments having a length ranging from 3 to 12 amino acid residues, and can be chemically synthesized at low cost. In addition, the peptides inducing attachment of barnacle cypris larvae are highly safe, and therefore can be widely applied in the ocean where it is desired to prevent the attachment of barnacles.
本発明に係るフジツボ類トラップ装置は、3~12アミノ酸残基の範囲の長さを有する低コストに化学合成可能なフジツボ類キプリス幼生の付着誘起ペプチドを備える。また、フジツボ類トラップ装置は、フジツボ類キプリス幼生の付着誘起ペプチドが安全性に優れるため、フジツボの付着を防止したい海洋に広く適用することができる。 The barnacle trapping device according to the present invention is equipped with a peptide that induces attachment of barnacle cypris larvae and has a length ranging from 3 to 12 amino acid residues, which can be chemically synthesized at low cost. In addition, since the peptide that induces attachment of barnacle cypris larvae is highly safe, the barnacle trapping device can be widely applied to marine environments where it is desired to prevent attachment of barnacles.
以下、本発明に係るフジツボ類キプリス幼生の付着誘起ペプチド及びこれを用いたフジツボ類トラップ装置について詳細に説明する。 The following provides a detailed explanation of the peptide that induces adhesion of barnacle cypris larvae and the barnacle trapping device that uses the peptide according to the present invention.
本発明に係るフジツボ類キプリス幼生の付着誘起ペプチドは、DAL(アスパラギン酸-アラニン-ロイシン)配列を含む3~12アミノ酸残基のアミノ酸配列を有するペプチド断片である。 The adhesion-inducing peptide of barnacle cypris larvae according to the present invention is a peptide fragment having an amino acid sequence of 3 to 12 amino acid residues including the DAL (aspartic acid-alanine-leucine) sequence.
ここで、ペプチドのアミノ酸配列は、常法に従って、N末端アミノ酸のアミノ酸残基が左側に位置し、C末端アミノ酸のアミノ酸残基が右側に位置するように記述する。したがって、DAL配列とは、N末端側からC末端側に向かってアスパラギン酸、アラニン及びロイシンがこの順にペプチド結合した配列を意味する。 Here, the amino acid sequence of a peptide is written in the usual way so that the N-terminal amino acid residue is located on the left and the C-terminal amino acid residue is located on the right. Therefore, the DAL sequence means a sequence in which aspartic acid, alanine, and leucine are peptide-bonded in this order from the N-terminus to the C-terminus.
DAL配列を含む3~12アミノ酸残基のアミノ酸配列とは、DAL配列からなるアミノ酸配列、又は、DAL配列のN末端側及び/又はC末端側に任意のアミノ酸がペプチド結合して全体が4~12アミノ酸残基となってアミノ酸配列を意味する。 An amino acid sequence of 3 to 12 amino acid residues containing a DAL sequence means an amino acid sequence consisting of the DAL sequence, or an amino acid sequence in which any amino acid is peptide-bonded to the N-terminus and/or C-terminus of the DAL sequence, resulting in a total of 4 to 12 amino acid residues.
また、「アミノ酸残基」とは、ペプチド鎖を構成しているアミノ酸の一単位に当たる部分を意味する。「アミノ酸残基」としては、天然若しくは非天然のα-アミノ酸、β-アミノ酸、γ-アミノ酸、δ-アミノ酸、オルニチン、ホモセリン、ホモシステイン、β-アラニン、γ-アミノブタン酸又はδ-アミノペンタン酸などが挙げられる。アミノ酸残基は、アミノ酸に光学活性体があり得る場合、L体及びD体の何れであってもよいが、L体が好ましい。 The term "amino acid residue" refers to a portion of an amino acid that constitutes a peptide chain. Examples of "amino acid residues" include natural or unnatural α-amino acids, β-amino acids, γ-amino acids, δ-amino acids, ornithine, homoserine, homocysteine, β-alanine, γ-aminobutanoic acid, and δ-aminopentanoic acid. When the amino acid may have optically active forms, the amino acid residue may be either the L-form or the D-form, with the L-form being preferred.
本発明に係るフジツボ類キプリス幼生の付着誘起ペプチドに含まれる天然のアミノ酸残基を略号で表示する場合、次の略号で記述する。アラニン残基:Ala又はA、アルギニン残基:Arg又はR、アスパラギン残基:Asn又はN、アスパラギン酸残基:Asp又はD、システイン残基:Cys又はC、グルタミン残基:Gln又はQ、グルタミン酸残基:GluまたはE、グリシン残基:Gly又はG、ヒスチジン残基:His又はH、イソロイシン残基:Ile又はI、ロイシン残基:Leu又はL、リジン残基:Lys又はK、メチオニン残基:Met又はM、フェニルアラニン残基:Phe又はF、プロリン残基:Pro又はP、セリン残基:Ser又はS、スレオニン残基:Thr又はT、トリプトファン残基:Trp又はW、チロシン残基:Tyr又はY、バリン残基:Val又はV。 The natural amino acid residues contained in the peptide for inducing attachment of barnacle cypris larvae according to the present invention are represented by the following abbreviations: alanine residue: Ala or A, arginine residue: Arg or R, asparagine residue: Asn or N, aspartic acid residue: Asp or D, cysteine residue: Cys or C, glutamine residue: Gln or Q, glutamic acid residue: Glu or E, glycine residue: Gly or G, histidine residue: His or H, isoleucine residue: Ile or I, leucine residue: Leu or L, lysine residue: Lys or K, methionine residue: Met or M, phenylalanine residue: Phe or F, proline residue: Pro or P, serine residue: Ser or S, threonine residue: Thr or T, tryptophan residue: Trp or W, tyrosine residue: Tyr or Y, valine residue: Val or V.
また、任意のアミノ酸としては、上述した天然のアミノ酸以外に2-アミノアジピン酸(略号:Aad)、3-アミノアジピン酸(略号:bAad)、β-アラニン(略号:bAla)、2-アミノ酪酸(略号:Abu)、4-アミノ酪酸或いはピペリジン酸(略号:4Abu)、6-アミノカプロン酸(略号:Acp)、2-アミノヘプタン酸(略号:Ahe)、2-アミノイソ酪酸(略号:Aib)、3-アミノイソ酪酸(略号:bAib)、2-アミノピメリン酸(略号:Apm)、2,4-ジアミノ酪酸(略号:Dbu)、デスモシン(略号:Des)、2,2'-ジアミノピメリン酸(略号:Dpm)、2,3-ジアミノプロピオン酸(略号:Dpr)、N-エチルグリシン(略号:EtGly)、N-エチルアスパラギン(EtAsn)、ヒドロキシリジン(Hyl)、アローヒドロキシリジン(略号:aHyl)、3-ヒドロキシプロリン(3Hyp)、4-ヒドロキシプロリン(略号:4Hyp)、イソデスモシン(略号:Ide)、アローイソロイシン(略号:aIle)、N-メチルグリシン或いはサルコシン(略号:MeGly)、N-メチルイソロイシン(略号:MeIle)、6-N-メチルリジン(略号:MeLys)、N-メチルバリン(略号:MeVal)、ノルバリン(略号:Nva)、ノルロイシン(略号:Nle)、オルニチン(略号:Orn)を挙げることができる。 In addition to the above-mentioned natural amino acids, the optional amino acids include 2-aminoadipic acid (abbreviation: Aad), 3-aminoadipic acid (abbreviation: bAad), β-alanine (abbreviation: bAla), 2-aminobutyric acid (abbreviation: Abu), 4-aminobutyric acid or piperidinic acid (abbreviation: 4Abu), 6-aminocaproic acid (abbreviation: Acp), 2-aminoheptanoic acid (abbreviation: Ahe), 2-aminoisobutyric acid (abbreviation: Aib), 3-aminoisobutyric acid (abbreviation: bAib), 2-aminopimelic acid (abbreviation: Apm), 2,4-diaminobutyric acid (abbreviation: Dbu), desmosine (abbreviation: Des), 2,2'-diaminopimelic acid (abbreviation: Dpm), 2,3-diaminopropionic acid (abbreviation: D Examples include N-ethylglycine (abbreviation: EtGly), N-ethylasparagine (EtAsn), hydroxylysine (Hyl), a-hydroxylysine (abbreviation: aHyl), 3-hydroxyproline (3Hyp), 4-hydroxyproline (abbreviation: 4Hyp), isodesmosine (abbreviation: Ide), a-isoleucine (abbreviation: aIle), N-methylglycine or sarcosine (abbreviation: MeGly), N-methylisoleucine (abbreviation: MeIle), 6-N-methyllysine (abbreviation: MeLys), N-methylvaline (abbreviation: MeVal), norvaline (abbreviation: Nva), norleucine (abbreviation: Nle), and ornithine (abbreviation: Orn).
フジツボ類キプリス幼生の付着誘起ペプチドにおいて、DAL配列以外のアミノ酸残基の種類は、フジツボ類キプリス幼生の付着誘起活性を有する限り任意である。また、フジツボ類キプリス幼生の付着誘起ペプチドにおけるDAL配列の位置は、フジツボ類キプリス幼生の付着誘起活性を有する限り任意である。 In a peptide that induces adhesion of barnacle cypris larvae, the type of amino acid residues other than the DAL sequence may be any as long as it has the activity of inducing adhesion of barnacle cypris larvae. In addition, the position of the DAL sequence in a peptide that induces adhesion of barnacle cypris larvae may be any as long as it has the activity of inducing adhesion of barnacle cypris larvae.
フジツボ類キプリス幼生の付着誘起ペプチドの一例としては、ELQDALDIHWRE(配列番号1)からなるペプチド断片を挙げることができる。また、フジツボ類キプリス幼生の付着誘起ペプチドの他の例としては、ELQDALDIHWRE(配列番号1)におけるDAL配列を含む連続する部分アミノ酸配列からなり、フジツボ類キプリス幼生の付着誘起活性を有するペプチドとすることができる。このようなフジツボ類キプリス幼生の付着誘起ペプチドの他の例としては、より具体的に、ELQDAL(配列番号2)、LQDALD(配列番号3)、QDALDI(配列番号4)、DALDIH(配列番号5)、DAL(配列番号9)及びDALD(配列番号10)を挙げることができる。 An example of a peptide that induces adhesion of barnacle cypris larvae is a peptide fragment consisting of ELQDALDIHWRE (SEQ ID NO: 1). Another example of a peptide that induces adhesion of barnacle cypris larvae is a peptide consisting of a continuous partial amino acid sequence containing the DAL sequence in ELQDALDIHWRE (SEQ ID NO: 1) and has adhesion-inducing activity for barnacle cypris larvae. More specifically, other examples of such peptides that induce adhesion of barnacle cypris larvae are ELQDAL (SEQ ID NO: 2), LQDALD (SEQ ID NO: 3), QDALDI (SEQ ID NO: 4), DALDIH (SEQ ID NO: 5), DAL (SEQ ID NO: 9), and DALD (SEQ ID NO: 10).
ただし、フジツボ類キプリス幼生の付着誘起ペプチドは、これら配列番号1~6のアミノ酸配列に限定されず、1又は複数のアミノ酸残基が置換したアミノ酸配列からなるものであっても良い。ここで、これらアミノ酸配列において置換する1又は複数のアミノ酸残基とはDAL配列を除くアミノ酸残基である。これらアミノ酸配列において置換するアミノ酸残基は、1又は2つのアミノ酸残基とすることが好ましく、1つのアミノ酸残基とすることがより好ましい。 However, the adhesion-inducing peptides for barnacle cypris larvae are not limited to the amino acid sequences of SEQ ID NOs: 1 to 6, and may be composed of amino acid sequences in which one or more amino acid residues are substituted. Here, the one or more amino acid residues substituted in these amino acid sequences are amino acid residues excluding the DAL sequence. It is preferable that one or two amino acid residues are substituted in these amino acid sequences, and it is more preferable that one amino acid residue is substituted.
これらアミノ酸配列において所定のアミノ酸残基を置換する場合、置換前のアミノ酸残基と性質の類似するアミノ酸残基に置換することが好ましい。例えば、「マッキー生化学」第6版「5.アミノ酸・ペプチド・タンパク質」監修:市川厚、監訳:福岡伸一、発行者:曽根良介、発行所:(株)化学同)に記載されているように、アミノ酸は同様の性質(化学的性質や物理的大きさ)を持つ側鎖に従って分類される。また、タンパク質の活性を保持したまま、所定のグループに分類されるアミノ酸残基間における分子進化上の置換が頻度高く起こることがよく知られる。 When replacing a given amino acid residue in these amino acid sequences, it is preferable to replace it with an amino acid residue that has similar properties to the amino acid residue before replacement. For example, as described in "Mackey Biochemistry" 6th Edition, "5. Amino Acids, Peptides, and Proteins" (edited by Ichikawa Atsushi, translated by Fukuoka Shinichi, published by Sone Ryosuke, published by Kagaku Do Co., Ltd.), amino acids are classified according to side chains that have similar properties (chemical properties and physical size). It is also well known that molecular evolutionary substitutions occur frequently between amino acid residues classified into certain groups while maintaining protein activity.
具体的に、中性非極性アミノ酸のうち、脂肪属性の疎水性側鎖をもつアミノ酸であるV(Val、バリン)、L(Leu、ロイシン)、I(Ile、イソロイシン)及びM(Met、メチオニン)からなる脂肪族疎水性アミノ酸グループが挙げられる。また、中性極性アミノ酸のうちヒドロキシメチレン基を側鎖に持つアミノ酸であるS(Ser、セリン)とT(Thr、スレオニン)から構成されるヒドロキシメチレン基グループが挙げられる。さらに、酸性であるカルボキシル基を側鎖に持つアミノ酸であるD(Asp、アスパラギン酸)とE(Glu、グルタミン酸)から構成される。酸性アミノ酸グループが挙げられる。さらにまた、塩基性のアミノ酸であるK(Lys、リジン)とR(Arg、アルギニン)から構成される塩基性アミノ酸グループが挙げられる。さらにまた、α位の炭素元素に側鎖としてメチレン基が結合しその先に極性基を有するアミノ酸であるN(Asn、アスパラギン、極性基はアミド基)、D(Asp、アスパラギン酸、極性基はカルボキシル基)及びH(His、ヒスチジン、極性基はイミダゾール基)から構成されるメチレン基=極性基グループが挙げられる。さらにまた、α位の炭素元素に側鎖としてジメチレン基以上の直鎖炭化水素が結合しその先に極性基を有するアミノ酸であるE(Glu、グルタミン酸、極性基はカルボキシル基)、K(Lys、リジン、極性基はアミノ基)、Q(Gln、グルタミン、極性基はアミド基)、R(Arg、アルギニン、極性基はイミノ基とアミノ基)から構成されるジメチレン基=極性基グループが挙げられる。さらにまた、側鎖にベンゼン核を持つ芳香族アミノ酸であるF(Phe、フェニルアラニン)、Y(Tyr、チロシン)及びW(Trp、トリプトファン)から構成される芳香族グループが挙げられる。さらにまた、側鎖に環状構造を有し、極性も有するアミノ酸であるH(H、ヒスチジン、環状構造と極性基は共にイミダゾール基)及びY(Tyr、チロシン、環状構造はベンゼン核で極性基は水酸基)から構成される環状&極性基グループが挙げられる。 Specific examples of such amino acids include the aliphatic hydrophobic amino acid group, which is made up of neutral nonpolar amino acids with hydrophobic side chains of aliphatic nature, V (Val, valine), L (Leu, leucine), I (Ile, isoleucine), and M (Met, methionine). Another example is the hydroxymethylene group, which is made up of neutral polar amino acids with hydroxymethylene groups in their side chains, S (Ser, serine) and T (Thr, threonine). Another example is the acidic amino acid group, which is made up of amino acids with acidic carboxyl groups in their side chains, D (Asp, aspartic acid) and E (Glu, glutamic acid). Another example is the basic amino acid group, which is made up of basic amino acids K (Lys, lysine) and R (Arg, arginine). Further examples include the methylene group = polar group group, which is composed of amino acids having a methylene group bonded to the carbon atom at the α position as a side chain and a polar group at the end of the side chain, N (Asn, asparagine, polar group is amide group), D (Asp, aspartic acid, polar group is carboxyl group), and H (His, histidine, polar group is imidazole group).Further examples include the dimethylene group = polar group group, which is composed of amino acids having a dimethylene group or more as a side chain and a polar group at the end of the side chain, E (Glu, glutamic acid, polar group is carboxyl group), K (Lys, lysine, polar group is amino group), Q (Gln, glutamine, polar group is amide group), and R (Arg, arginine, polar group is imino group and amino group). Furthermore, there is an aromatic group consisting of aromatic amino acids with a benzene nucleus in the side chain, F (Phe, phenylalanine), Y (Tyr, tyrosine), and W (Trp, tryptophan). There is also a cyclic and polar group group consisting of amino acids with a cyclic structure in the side chain, H (H, histidine, both the cyclic structure and the polar group are imidazole groups) and Y (Tyr, tyrosine, the cyclic structure is a benzene nucleus and the polar group is a hydroxyl group).
以上のように分類されるグループは、それぞれ機能的に類似したアミノ酸から構成される。よって、所定のアミノ酸配列からなるフジツボ類キプリス幼生の付着誘起ペプチドにおいて、所定のアミノ酸残基を置換する場合、同じグループ内のアミノ酸から選択することで、フジツボ類キプリス幼生の付着誘起機能を維持できる蓋然性が高い。一例として、フジツボ類キプリス幼生の付着誘起ペプチド:ELQDALDIHWRE(配列番号1)においては、N末端又はC末端のグルタミン酸残基を、ジメチレン基=極性基グループを構成するリジン、グルタミン又はアルギニンに置換してもフジツボ類キプリス幼生の付着誘起機能を維持する蓋然性が高い。 Each of the groups classified as above is composed of functionally similar amino acids. Therefore, when replacing a specific amino acid residue in a peptide that induces adhesion of barnacle cypris larvae consisting of a specific amino acid sequence, there is a high probability that the adhesion-inducing function of barnacle cypris larvae can be maintained by selecting an amino acid from the same group. As an example, in the adhesion-inducing peptide of barnacle cypris larvae: ELQDALDIHWRE (SEQ ID NO: 1), even if the glutamic acid residue at the N-terminus or C-terminus is replaced with lysine, glutamine, or arginine that constitutes a dimethylene group = polar group group, there is a high probability that the adhesion-inducing function of barnacle cypris larvae can be maintained.
所定のアミノ酸配列からなるペプチド断片がフジツボ類キプリス幼生の付着誘起機能を有するかは、例えば以下の方法により確認することができる。先ず、供試するペプチド断片を含む海水を樹脂製の容器内に充填する。そして、容器内の海水にフジツボ類キプリス幼生を加え、所定時間経過した後の当該容器内面に付着したキプリス幼生を計測する。また、比較のため、ペプチド断片を含まない以外は同様な方法で容器内面に付着したキプリス幼生を計測する。供試するペプチド断片が存在する条件下で、容器内面に付着したキプリス幼生の数が統計的に有意に増加しているならば、当該ペプチド断片についてフジツボ類キプリス幼生の付着誘起機能を有すると判断できる。 Whether a peptide fragment consisting of a specific amino acid sequence has the function of inducing the attachment of barnacle cypris larvae can be confirmed, for example, by the following method. First, seawater containing the peptide fragment to be tested is filled into a resin container. Then, barnacle cypris larvae are added to the seawater in the container, and the number of cypris larvae attached to the inner surface of the container after a specified time has passed is counted. For comparison, the number of cypris larvae attached to the inner surface of the container is counted using a similar method except that the peptide fragment is not included. If there is a statistically significant increase in the number of cypris larvae attached to the inner surface of the container in the presence of the peptide fragment to be tested, the peptide fragment can be determined to have the function of inducing the attachment of barnacle cypris larvae.
また、この試験において、キプリス幼生の死滅数を計測し、当該ペプチド断片についてフジツボ類キプリス幼生に対する毒性を有するか判定することができる。この結果に基づけば、供試したペプチド断片について、フジツボ類キプリス幼生に対する毒性がなく、且つ、付着誘起する機能を有するか判断することができる。 In addition, in this test, the number of dead cypris larvae can be counted to determine whether the peptide fragment has toxicity to barnacle cypris larvae. Based on this result, it can be determined whether the peptide fragment tested is not toxic to barnacle cypris larvae and has the function of inducing attachment.
本発明に係るフジツボ類キプリス幼生の付着誘起ペプチドは、以上で説明したように最大でも12アミノ酸残基からなるペプチド断片であるため、化学合成により非常に容易に且つ低コストに製造することができる。 As described above, the adhesion-inducing peptide of the present invention for barnacle cypris larvae is a peptide fragment consisting of a maximum of 12 amino acid residues, and therefore can be produced very easily and at low cost by chemical synthesis.
ペプチド断片を化学的に合成する手法は、特に限定されず、従来公知の手法を適用することができる。ペプチドの化学的合成法としては、後述の実施例に記載した方法、本技術分野において固相ペプチド合成法と総称される方法、本技術分野において液相ペプチド合成法と総称される方法を挙げることができる。固相ペプチド合成法及び液相ペプチド合成法のいずれも、C末端からN末端に向かって、アミノ基に保護基を結合させたアミノ酸を順次結合させる点で共通しており、その後、アミノ基に結合した保護基を除去(脱保護)して次の反応点とする点で共通している。 The method for chemically synthesizing peptide fragments is not particularly limited, and conventionally known methods can be applied. Examples of chemical peptide synthesis methods include the method described in the Examples below, the method generally known in this technical field as solid-phase peptide synthesis, and the method generally known in this technical field as liquid-phase peptide synthesis. Both solid-phase peptide synthesis and liquid-phase peptide synthesis methods have in common that amino acids having protecting groups bound to their amino groups are sequentially bound from the C-terminus to the N-terminus, and then the protecting groups bound to the amino groups are removed (deprotected) to provide the next reaction point.
特に、本発明に係るフジツボ類キプリス幼生の付着誘起ペプチドは、以上で説明したように最大でも12アミノ酸残基からなるペプチド断片であるため、液相ペプチド合成法による大量合成に適している。液相ペプチド合成法のなかでも、いわゆる疎水性タグを用いた液相ペプチド合成法(Y. Okada, H. Suzuki, T. Nakae, S. Fujita, H. Abe, K. Nagano, T. Yamada, N. Ebata, S. Kim and K. Chiba, Tag-Assisted Liquid-Phase Peptide Synthesis Using Hydrophobic Bnezyl Alcohols as Supports, J. Org. Chem., 2013, 78, 320-327.)を適用することが好ましい。 In particular, the adhesion-inducing peptide of the present invention for barnacle cypris larvae is a peptide fragment consisting of at most 12 amino acid residues as explained above, and is therefore suitable for mass synthesis by liquid-phase peptide synthesis. Among liquid-phase peptide synthesis methods, it is preferable to apply a liquid-phase peptide synthesis method using a so-called hydrophobic tag (Y. Okada, H. Suzuki, T. Nakae, S. Fujita, H. Abe, K. Nagano, T. Yamada, N. Ebata, S. Kim and K. Chiba, Tag-Assisted Liquid-Phase Peptide Synthesis Using Hydrophobic Benezyl Alcohols as Supports, J. Org. Chem., 2013, 78, 320-327.).
疎水性タグとは、可溶性の担体であって固相法で使用される反応を適用できる化合物であり、1つめのアミノ酸のC末端に結合可能な分子である。疎水性タグとしては、3,4,5-三置換ベンジルアルコールを炭化水素鎖で疎水化した化合物を使用することができる。ペプチド合成反応では、当該疎水性タグのベンジルアルコール基に一つ目のアミノ酸のC末端を結合させ、これを基点にして順次、保護基を有するアミノ酸を結合させていき(脱保護したアミノ酸を反応基点とする)、目的とするアミノ酸配列のペプチドを合成する。 A hydrophobic tag is a soluble carrier that can be used in the reactions used in the solid-phase method, and is a molecule that can be bound to the C-terminus of the first amino acid. A compound in which 3,4,5-trisubstituted benzyl alcohol is hydrophobized with a hydrocarbon chain can be used as a hydrophobic tag. In the peptide synthesis reaction, the C-terminus of the first amino acid is bound to the benzyl alcohol group of the hydrophobic tag, and using this as a base point, amino acids with protecting groups are sequentially bound (the deprotected amino acid serves as the reaction base point) to synthesize a peptide of the desired amino acid sequence.
ここで保護基としては、一般的な固相ペプチド合成法や液相ペプチド合成法で利用されているものを使用することができる。例えば、保護基としては、9-フルオレニルメトキシカルボニル(Fmoc)やtert-ブトキシカルボニル基(Boc)を挙げることができる。Fmoc基をペプチド鎖から外す場合(脱保護)には、通常DMF/20%ピペリジン条件が用いられる。またBoc基をペプチド鎖から外す場合(脱保護)には通常トリフルオロ酢酸などが用いられる。 The protecting groups used here can be those used in general solid-phase peptide synthesis methods or liquid-phase peptide synthesis methods. For example, protecting groups include 9-fluorenylmethoxycarbonyl (Fmoc) and tert-butoxycarbonyl (Boc) groups. When removing the Fmoc group from the peptide chain (deprotection), DMF/20% piperidine conditions are usually used. When removing the Boc group from the peptide chain (deprotection), trifluoroacetic acid or the like is usually used.
ペプチド合成法により合成されたペプチドは、従来公知の方法に準じて精製することができる。例えば、種々のクロマトグラフィー(例えば、シリカゲルカラムクロマトグラフィー、イオン交換カラムクロマトグラフィー、ゲルろ過、もしくは逆相クロマトグラフィー)等で精製することができる。 Peptides synthesized by peptide synthesis methods can be purified according to conventional methods. For example, they can be purified by various types of chromatography (e.g., silica gel column chromatography, ion exchange column chromatography, gel filtration, or reverse phase chromatography).
なお、設計したペプチドにおいて、1つ以上の不斉点がある場合、その不斉点を有するアミノ酸を用いることによって、目的の不斉点を有するペプチドを製造することができる。また、合成したペプチドの光学純度を上げるために、製造工程の適当な段階で光学分割などを行ってもよい。光学分割法として例えば、不活性溶媒中(例えばメタノール、エタノール、もしくは2-プロパノールなどのアルコール系溶媒、ジエチルエーテルなどのエーテル系溶媒、酢酸エチルなどのエステル系溶媒、トルエンなどの炭化水素系溶媒又はアセトニトリルなどの非プロトン系溶媒、およびこれらの混合溶媒)、光学活性な酸(例えば、マンデル酸、N-ベンジルオキシアラニン、もしくは乳酸などのモノカルボン酸、酒石酸、o-ジイソプロピリデン酒石酸若しくはリンゴ酸などのジカルボン酸、又はカンファースルフォン酸若しくはブロモカンファースルフォン酸などのスルホン酸)と塩を形成させるジアステレオマー法により行うことができる。 When the designed peptide has one or more asymmetric points, a peptide having the desired asymmetric point can be produced by using an amino acid having the asymmetric point. In addition, in order to increase the optical purity of the synthesized peptide, optical resolution or the like may be performed at an appropriate stage in the production process. As an optical resolution method, for example, a diastereomeric method can be used in which a salt is formed with an optically active acid (for example, a monocarboxylic acid such as mandelic acid, N-benzyloxyalanine, or lactic acid, a dicarboxylic acid such as tartaric acid, o-diisopropylidenetartaric acid, or malic acid, or a sulfonic acid such as camphorsulfonic acid or bromocamphorsulfonic acid) in an inert solvent (for example, an alcoholic solvent such as methanol, ethanol, or 2-propanol, an ether solvent such as diethyl ether, an ester solvent such as ethyl acetate, a hydrocarbon solvent such as toluene, or an aprotic solvent such as acetonitrile, or a mixture of these).
なお、本発明に係るフジツボ類キプリス幼生の付着誘起ペプチドは、上述のように化学合成法により製造されたものに限定されず、当該付着誘起ペプチドを発現する形質転換体を用いて製造しても良い。形質転換体は、上述した付着誘起ペプチドをコードする核酸を宿主内で発現可能なようにベクターに挿入し、大腸菌、酵母、昆虫細胞等の宿主中で発現させることにより得られる。そして、得られた形質転換体を培養することにより、目的の付着誘起ペプチドを培養物に蓄積させることで生産することができる。また、無細胞タンパク質発現系を適用して、本発明に係るフジツボ類キプリス幼生の付着誘起ペプチドを製造しても良い。 The adhesion-inducing peptide for barnacle cypris larvae according to the present invention is not limited to those produced by chemical synthesis as described above, and may be produced using a transformant that expresses the adhesion-inducing peptide. The transformant is obtained by inserting a nucleic acid encoding the above-mentioned adhesion-inducing peptide into a vector so that it can be expressed in a host, and expressing it in a host such as E. coli, yeast, or insect cells. The desired adhesion-inducing peptide can then be produced by accumulating it in the culture by culturing the obtained transformant. The adhesion-inducing peptide for barnacle cypris larvae according to the present invention may also be produced by applying a cell-free protein expression system.
本発明に係るフジツボ類キプリス幼生の付着誘起ペプチドは、海洋中に存在するフジツボ類キプリス幼生の固定物への付着を誘起する。ここで、対象となるフジツボ類とは、節足動物門甲殻亜門におけるフジツボ亜目に分類される動物を意味する。より具体的に、フジツボ類としては、特に限定されないが、タテジマフジツボ属(Amphibalanus)、フジツボ属(Balanus)及びオオアカフジツボ属(Megabalanus)を含むフジツボ科(Balanidae)のフジツボ;カメフジツボ属(Chelonibia)を含むカメフジツボ科(Chelonibiidae);アカツキフジツボ科(Chionelasmatidae)のフジツボ;イワフジツボ属(Chthamalus)を含むイワフジツボ科(Chthamalidae)のフジツボ;オニフジツボ属(Coronula)を含むオニフジツボ科(Coronulidae)のフジツボ;サラフジツボ科(Platylepadidae)のフジツボ;サンゴフジツボ科(Pyrgomatidae)のフジツボ;クロフジツボ属(Tetraclita)及びヒラフジツボ属(Tetraclitella)を含むクロフジツボ科(Tetraclitidae)のフジツボ;ムカシフジツボ科(Archaeobalanidae)のフジツボからなる群から選ばれる少なくとも1種のフジツボを挙げることができる。 The peptide for inducing adhesion of barnacle cypris larvae according to the present invention induces adhesion of barnacle cypris larvae present in the ocean to fixed objects. Here, the target barnacles refer to animals classified in the suborder Balanidae in the subphylum Crustacea of the phylum Arthropoda. More specifically, barnacles include, but are not limited to, barnacles of the family Balanidae including the genera Amphibalanus, Balanus, and Megabalanus; barnacles of the family Chelonibiidae including Chelonibia; barnacles of the family Chionelasmatidae; barnacles of the family Chthamalidae including Chthamalus; At least one type of barnacle may be selected from the group consisting of barnacles of the Coronulidae family, including the Coronula genus; barnacles of the Platylepadidae family; barnacles of the Pyrgomatidae family; barnacles of the Tetraclita family, including the Tetraclita and Tetraclitella genera; and barnacles of the Archaeobalanidae family.
したがって、フジツボ類キプリス幼生の付着誘起ペプチドは、フジツボ類トラップ装置に利用することができる。フジツボ類トラップ装置は、海洋中に設置され、フジツボ類キプリス幼生の付着を誘起する機能を有する装置である。すなわち、フジツボ類トラップ装置を海洋に設置することで、フジツボ類トラップ装置及びその周囲の構造物に対してフジツボ類キプリス幼生の付着を誘起することができる。その結果、フジツボ類の付着を防止したい構造物に対するフジツボ類キプリス幼生の付着を防止することができる。 Therefore, the peptides that induce attachment of barnacle cypris larvae can be used in a barnacle trap device. A barnacle trap device is a device that is installed in the ocean and has the function of inducing attachment of barnacle cypris larvae. In other words, by installing a barnacle trap device in the ocean, it is possible to induce attachment of barnacle cypris larvae to the barnacle trap device and its surrounding structures. As a result, it is possible to prevent attachment of barnacle cypris larvae to structures on which it is desired to prevent barnacle attachment.
例えば、図1に概略的に示すように、フジツボ類トラップ装置1としては、一主面に複数の開口部2を有す蓋部材3と、フジツボ類キプリス幼生の付着誘起ペプチドを含むゲル4を収容する筐体5とから構成される装置を挙げることができる。フジツボ類トラップ装置1は、開口部2からゲル4が臨むため、海洋に浸漬されるとゲル4内のフジツボ類キプリス幼生の付着誘起ペプチドが拡散することとなる。 For example, as shown diagrammatically in FIG. 1, a barnacle trapping device 1 can be a device that is composed of a cover member 3 having a plurality of openings 2 on one main surface, and a housing 5 that contains a gel 4 that contains an attachment-inducing peptide for barnacle cypris larvae. Since the gel 4 faces the barnacle trapping device 1 from the opening 2, when the barnacle trapping device 1 is immersed in the ocean, the attachment-inducing peptide for barnacle cypris larvae in the gel 4 will diffuse.
ここで、ゲル4は、上述した付着誘起ペプチドを所定の濃度で分散させた高分子化合物溶液を作製し、高分子化合物が架橋して形成されるハイドロゲルを意味する。ゲル4を構成する高分子化合物は、特に限定されず、デンプン、アガロース、ペクチン、カラギナン、ジェランガム、アルギン酸ナトリウム、アクリルアミド及びコラーゲン様ペプチド等を挙げることができる。また、ゲル4は、特許第5931584号に開示された硬脆性ゲル及び軟伸性ゲルの混合相と前記軟伸性ゲルの単相とからなる親水性ダブルネットワークゲルを適用してもよい。 Here, gel 4 refers to a hydrogel formed by crosslinking a polymer compound solution prepared by dispersing the above-mentioned adhesion-inducing peptide at a predetermined concentration. The polymer compound constituting gel 4 is not particularly limited, and examples thereof include starch, agarose, pectin, carrageenan, gellan gum, sodium alginate, acrylamide, and collagen-like peptides. Gel 4 may also be a hydrophilic double network gel consisting of a mixed phase of a hard brittle gel and a soft extensible gel and a single phase of the soft extensible gel, as disclosed in Patent No. 5931584.
以上のように構成されたフジツボ類トラップ装置を海洋中に設置されると、ゲル4に含まれるフジツボ類キプリス幼生の付着誘起ペプチドが周囲の海中に拡散する。これにより、海中に存在するフジツボ類キプリス幼生は、フジツボ類トラップ装置及びその周囲の構造物に付着する。例えば、フジツボ類トラップ装置を構造物とともに海底に沈めることで、当該構造物の表面にキプリス幼生を付着させることができる。 When the barnacle trapping device configured as described above is installed in the ocean, the adhesion-inducing peptides for barnacle cypris larvae contained in the gel 4 diffuse into the surrounding ocean. As a result, barnacle cypris larvae present in the ocean attach to the barnacle trapping device and surrounding structures. For example, by sinking the barnacle trapping device together with a structure to the ocean bottom, the cypris larvae can be attached to the surface of the structure.
このように、フジツボ類トラップ装置によれば、当該装置及び当該装置に近接する構造物に対してフジツボの付着を誘起できるため、当該装置及び当該構造物から離間した施設・設備に対してフジツボの付着を防止することができる。フジツボの付着を防止する施設・設備としては、特に限定されないが、発電所における機器冷却用海水の取水装置並びに排水装置、工場取水・排水設備、定置網施設(網や錨、網の固定具)、排水溝、港湾施設、船舶係留施設等を挙げることができる。 In this way, the barnacle trapping device can induce barnacles to attach to the device itself and structures adjacent to the device, and can therefore prevent barnacles from attaching to facilities and equipment distant from the device and structure. Examples of facilities and equipment that prevent barnacle attachment include, but are not limited to, seawater intake and discharge systems for cooling equipment at power plants, factory water intake and discharge systems, fixed net facilities (nets, anchors, net fasteners), drains, port facilities, ship mooring facilities, etc.
これら施設・設備に対するフジツボの付着を防止するには、より具体的には図2に示すように、フジツボ類トラップ装置1を構造体10の一主面に取り付けた状態で、当該施設・設備の周囲の海底に沈めるといった方法・方式を採用することができる。構造体10は、例えばコンクリート成形体とすることができ、コンクリートからなる建築廃材としてもよい。また、図2の例では、構造体10の上面にフジツボ類トラップ装置1を配設したが、フジツボ類トラップ装置1は構造体10の上面及び/又は側面に複数個配設しても良い。 To prevent barnacles from attaching to these facilities and equipment, more specifically, as shown in Figure 2, a method or system can be adopted in which a barnacle trapping device 1 is attached to one main surface of a structure 10 and then submerged in the seabed around the facility or equipment. The structure 10 can be, for example, a concrete molded body, or it may be construction waste material made of concrete. In the example of Figure 2, the barnacle trapping device 1 is disposed on the top surface of the structure 10, but multiple barnacle trapping devices 1 may be disposed on the top surface and/or sides of the structure 10.
さらにまた、フジツボ類トラップ装置1は、図3に示すように、構造体10に対して着脱自在とすることが好ましい。この場合、フジツボ類トラップ装置1は、所望のタイミングで構造体10から脱離することができる。例えば、フジツボ類トラップ装置1を取り付けた構造体10を海底に沈め、構造体10の表面にフジツボ類キプリス幼生が付着し、フジツボ成体まで成長した段階でフジツボ類トラップ装置1を取り外すことができる。一旦、構造体10表面にフジツボ成体が付着すれば、フジツボ類キプリス幼生はフジツボ成体に近接して構造体10表面に付着できるためである。構造体10から取り外したフジツボ類トラップ装置1は、異なる構造体10等に取り付けられ再利用することができる。 Furthermore, as shown in FIG. 3, it is preferable that the barnacle trapping device 1 is detachable from the structure 10. In this case, the barnacle trapping device 1 can be detached from the structure 10 at a desired timing. For example, the structure 10 to which the barnacle trapping device 1 is attached is sunk to the bottom of the sea, and the barnacle cypris larvae attach to the surface of the structure 10, and the barnacle trapping device 1 can be removed when the barnacle cypris larvae grow into adult barnacles. Once the adult barnacles attach to the surface of the structure 10, the barnacle cypris larvae can attach to the surface of the structure 10 in close proximity to the adult barnacles. The barnacle trapping device 1 detached from the structure 10 can be attached to a different structure 10, etc., and reused.
図2及び/又は図3に示したフジツボ類トラップ装置1を取り付けた構造体10は、例えば、発電所の取水装置の近傍に設置することで、当該取水装置に対するフジツボの付着を防止することができる。 The structure 10 to which the barnacle trapping device 1 shown in Figures 2 and/or 3 is attached can be installed, for example, near the water intake device of a power plant to prevent barnacles from attaching to the water intake device.
一方、フジツボ付着による被害としては、漁網の目を詰まらせ養殖魚を窒息させることが大きな問題となっている。例えば、図4に示すように、海水より比重を軽くした浮遊型のフジツボ類トラップ装置11とすることができる。より具体的に、浮遊型のフジツボ類トラップ装置11としては、一般的に浮きやブイと呼称される球形のフロート部材の表面に図1に示したようなフジツボ類キプリス幼生の付着誘起ペプチドを含むゲル4を臨ませても良いし、フジツボ類キプリス幼生の付着誘起ペプチドを含む塗料を塗布しても良い。 On the other hand, damage caused by barnacle attachment is a major problem, as it clogs the meshes of fishing nets and suffocates farmed fish. For example, as shown in Figure 4, a floating barnacle trapping device 11 with a specific gravity lighter than seawater can be used. More specifically, the floating barnacle trapping device 11 can be made by having a gel 4 containing the attachment-inducing peptides of barnacle cypris larvae as shown in Figure 1 on the surface of a spherical float member, generally called a float or buoy, or by applying a paint containing the attachment-inducing peptides of barnacle cypris larvae.
図4に示したフジツボ類トラップ装置11は、アンカー12に対して縄13によって連結されている。縄13の中途部にはフジツボ類トラップ装置11の着脱のための連結環15が配設されており、連結環15を介してフジツボ類トラップ装置11が縄13に連結されている。図4に示したフジツボ類トラップ装置11は、アンカー12を海底に沈めることで、海底から所定の高さに位置することができる。図4に示したフジツボ類トラップ装置11は、縄3の長さを調節することで、海底からの所望の高さに設定することができる。したがって、養殖魚を飼育する漁網の下部にフジツボ類トラップ装置11を位置させることで、漁網へのフジツボの付着を防止し、フジツボ類トラップ装置11の表面にフジツボの付着を誘起することができる。 The barnacle trapping device 11 shown in FIG. 4 is connected to the anchor 12 by a rope 13. A connecting ring 15 for attaching and detaching the barnacle trapping device 11 is provided in the middle of the rope 13, and the barnacle trapping device 11 is connected to the rope 13 via the connecting ring 15. The barnacle trapping device 11 shown in FIG. 4 can be positioned at a predetermined height from the seabed by sinking the anchor 12 to the seabed. The barnacle trapping device 11 shown in FIG. 4 can be set to a desired height from the seabed by adjusting the length of the rope 3. Therefore, by positioning the barnacle trapping device 11 at the bottom of a fishing net for raising farmed fish, it is possible to prevent barnacles from attaching to the fishing net and induce barnacles to attach to the surface of the barnacle trapping device 11.
また、図4に示したフジツボ類トラップ装置11は連結環15を介して縄13に連結しているため、容易に取り外すことができる。すなわち、フジツボ類トラップ装置11は、適宜取り替えることができ、長期間にわたって漁網へのフジツボの付着を防止することができる。 The barnacle trapping device 11 shown in FIG. 4 is connected to the rope 13 via a connecting ring 15, so it can be easily removed. In other words, the barnacle trapping device 11 can be replaced as needed, and barnacles can be prevented from attaching to fishing nets for a long period of time.
さらにまた、漁網などの海洋に設置する設備に対するフジツボ類の付着を防止するには、図4に示したフジツボ類トラップ装置11に限定されず、例えば、図5(A)及び(B)に示すように、縄状或いは網状のフジツボ類トラップ装置20とすることができる。図5(A)及び(B)に示すフジツボ類トラップ装置20は、フジツボ類キプリス幼生の付着誘起ペプチドを含む溶液を縄や網に含浸させて作製することができる。このとき溶液としては、ゲル化前の溶液とすることができ、当該溶液を縄や網に含浸させた後にゲル化するものであってもよい。また、図5(A)及び(B)に示したフジツボ類トラップ装置20では、適切な溶媒にフジツボ類キプリス幼生の付着誘起ペプチドを溶解させた溶液に縄や網に含浸させたものであってもよい。 Furthermore, in order to prevent barnacles from attaching to marine equipment such as fishing nets, the barnacle trapping device 11 shown in FIG. 4 is not limited to the barnacle trapping device 11 shown in FIG. 4, but may be, for example, a rope- or net-shaped barnacle trapping device 20 as shown in FIG. 5(A) and (B). The barnacle trapping device 20 shown in FIG. 5(A) and (B) can be produced by impregnating a rope or net with a solution containing an attachment-inducing peptide for barnacle cypris larvae. In this case, the solution may be a solution before gelation, or may be one that gels after the rope or net is impregnated with the solution. In addition, in the barnacle trapping device 20 shown in FIG. 5(A) and (B), the rope or net may be impregnated with a solution in which an attachment-inducing peptide for barnacle cypris larvae is dissolved in an appropriate solvent.
図5(A)及び(B)に示したフジツボ類トラップ装置20は、例えば球形のフロート部材21(浮きやブイとの称される)に着脱自在に連結され海面近傍に設置される。フロート部材21は、図示しないが、海底に対してアンカー等で連結されることで、フジツボ類トラップ装置20を所定の位置に設置することができる。例えば、養殖魚を飼育する漁網を囲うように図5(A)及び(B)に示したフジツボ類トラップ装置20を設置することができる。これにより、フジツボ類トラップ装置20の表面にフジツボの付着を誘起することができ、漁網へのフジツボの付着を防止することができる。なお、図5(A)及び(B)に示したフジツボ類トラップ装置20は、漁網以外にも、停泊中の船舶の周囲に設置したり、海洋上に設置した建造物の周囲に設置したりすることで、船底や建造物へのフジツボの付着を防止することができる。また、この場合にも、フロート部材21に連結したフジツボ類トラップ装置20を適宜、取り替えることで長期にわたってフジツボの付着を防止することができる。 The barnacle trapping device 20 shown in Figs. 5(A) and (B) is detachably connected to, for example, a spherical float member 21 (also called a float or buoy) and installed near the sea surface. The float member 21 is connected to the seabed with an anchor or the like (not shown), so that the barnacle trapping device 20 can be installed at a predetermined position. For example, the barnacle trapping device 20 shown in Figs. 5(A) and (B) can be installed so as to surround a fishing net for raising farmed fish. This can induce barnacles to attach to the surface of the barnacle trapping device 20, and prevent barnacles from attaching to the fishing net. The barnacle trapping device 20 shown in Figs. 5(A) and (B) can be installed around a ship at anchor or around a structure installed on the sea, in addition to a fishing net, to prevent barnacles from attaching to the bottom of the ship or the structure. In this case, too, barnacle attachment can be prevented for a long period of time by replacing the barnacle trap device 20 connected to the float member 21 as needed.
〔実施例〕
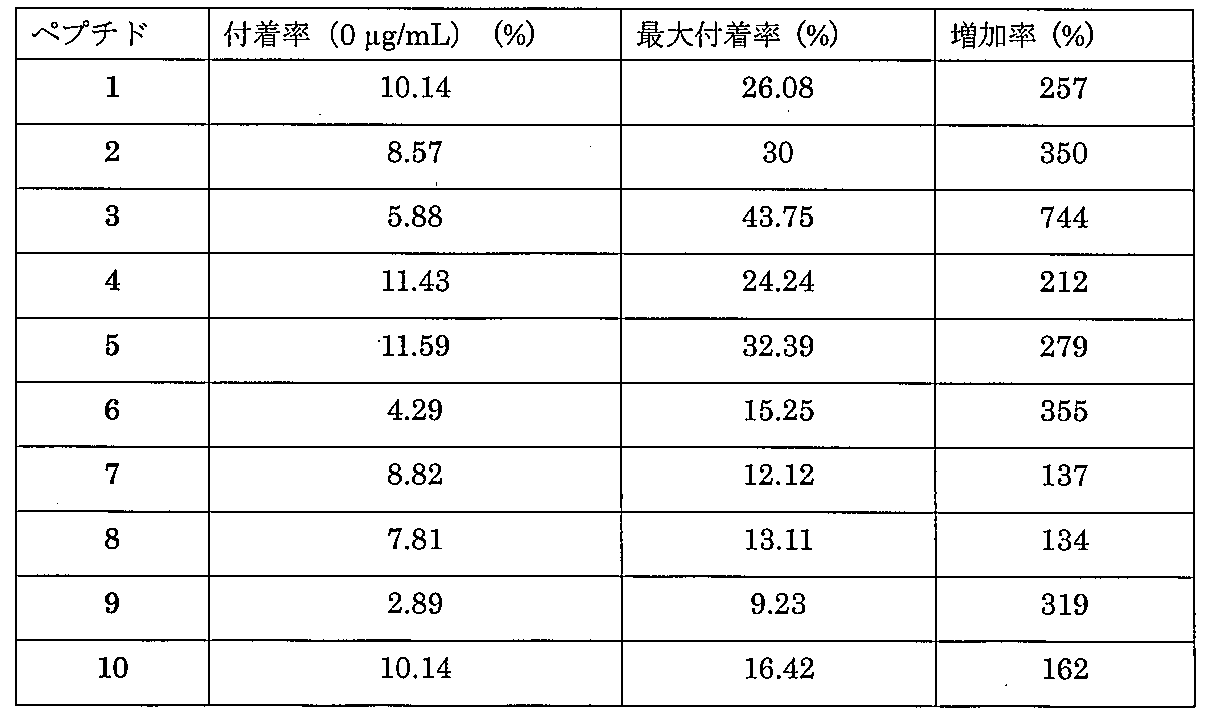
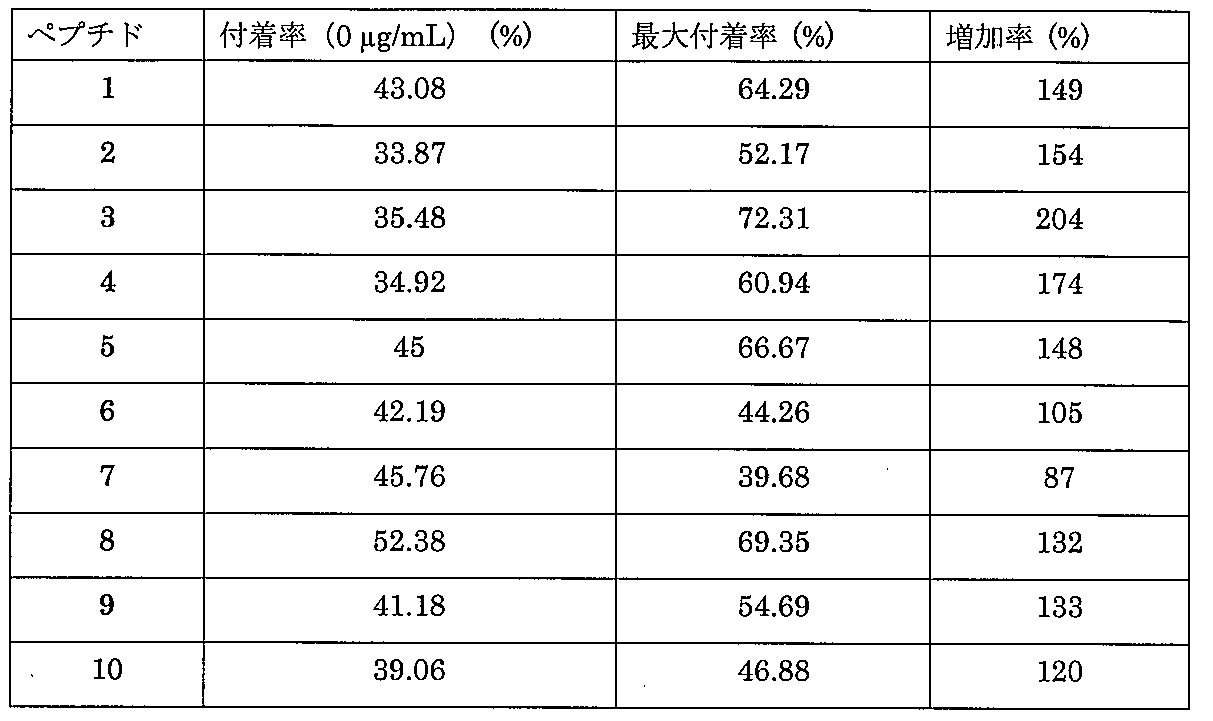
以下、実施例により本発明を詳細に説明するが、本発明の技術的範囲は以下の実施例に限定されるものではない。本実施例では、下記のタグペプチドを利用した方法により、ペプチド1~10(表1、配列番号1~10)を化学的に合成した。
〔Example〕
The present invention will be described in detail below with reference to examples, but the technical scope of the present invention is not limited to the following examples. In these examples, peptides 1 to 10 (Table 1, SEQ ID NOs: 1 to 10) were chemically synthesized by a method using the following tag peptides.
ペプチド合成に使用したタグペプチド、アミノ酸のカップリング反応、Fmoc基脱保護反応、全脱保護反応について説明する。 We will explain the tag peptide, amino acid coupling reaction, Fmoc group deprotection reaction, and total deprotection reaction used in peptide synthesis.
〔タグペプチド〕
下記に示すTAG-OH(2,4-bis(docosyloxy)benzylalcohol)とFmoc-アミノ酸をC末端で結合させたFmoc-AA-TAG及び脱保護と伸長を行い生成したペプチドを本実施例においてタグペプチドと称することとする。
[Tag peptide]
In this example, Fmoc-AA-TAG, in which TAG-OH (2,4-bis(docosyloxy)benzylalcohol) and an Fmoc-amino acid are bound at the C-terminus, and the peptide generated by deprotection and elongation are referred to as tag peptides.
〔カップリング反応〕
Fmoc化/Boc化アミノ酸のカップリング方法では、先ず、脱保護したタグペプチド(1.0 eq)とFmoc化アミノ酸(1.3 eq)、COMU(1.3 eq)をdry THF(20 mL)に溶解させ、DIPEA(2.0 eq)を添加し、室温で30~60分攪拌した。TLCによる反応終了確認後、Acetonitrileを加えて目的物を析出させ、Acetonitrileで吸引ろ過、洗浄を行い、固体を捕集した。なお、本実施例では、詳細を後述するようにペプチド1~10の10種類を合成した。ペプチド1を合成する際には12残基目、ペプチド2~7を合成する際には6残基目、ペプチド9を合成する際には3残基目、そして、ペプチド10を合成する際には10及び11残基目については、Fmoc化アミノ酸に代えてBoc化アミノ酸を使用した。
[Coupling reaction]
In the coupling method of Fmoc/Boc amino acids, first, deprotected tag peptide (1.0 eq), Fmoc amino acid (1.3 eq), and COMU (1.3 eq) were dissolved in dry THF (20 mL), DIPEA (2.0 eq) was added, and the mixture was stirred at room temperature for 30 to 60 minutes. After confirming the completion of the reaction by TLC, acetonitrile was added to precipitate the target product, and the solid was collected by suction filtration and washing with acetonitrile. In this example, 10 types of peptides 1 to 10 were synthesized as described in detail below. Boc amino acids were used instead of Fmoc amino acids for the 12th residue when peptide 1 was synthesized, the 6th residue when peptides 2 to 7 were synthesized, the 3rd residue when peptide 9 was synthesized, and the 10th and 11th residues when peptide 10 was synthesized.
〔Fmoc基脱保護反応〕
Fmoc化を末端に有するタグペプチドをdry THFに溶解させ、室温で撹拌しながらPiperidine(1%)、DBU(1%)を添加し、室温で5~15分攪拌した。TLCによる反応終了確認後、1N HClで塩基を中和し、Acetonitrileを加えて目的物を析出させ、Acetonitrileで吸引ろ過、洗浄を行い、固体を捕集した。
[Fmoc group deprotection reaction]
The tag peptide with Fmoc at the end was dissolved in dry THF, and piperidine (1%) and DBU (1%) were added while stirring at room temperature, and the mixture was stirred at room temperature for 5 to 15 minutes. After confirming the completion of the reaction by TLC, the base was neutralized with 1N HCl, and acetonitrile was added to precipitate the target product. The product was filtered by suction and washed with acetonitrile, and the solid was collected.
〔全脱保護反応〕
タグペプチドをChloroform 5 mLに溶解させ、TFA(19mL、95%)、TIS(250 μL、2.5%)、H2O(250μL、2.5%)を添加し、室温で1~2時間攪拌した。TLCによる反応終了確認後、セライトろ過により不溶性不純物を除去した。得られたろ液に冷diisopropyletherを加えてペプチドを析出させ、溶液をファルコンチューブに移し、凍結遠心(3500 rpm、15 min、-9℃)を行った。上清を捨て、同様の操作をさらに2回行い、固体を得た。得られた化合物はこの後逆相HPLCにより精製を行った。
[Total deprotection reaction]
The tag peptide was dissolved in 5 mL of Chloroform, and TFA (19 mL, 95%), TIS (250 μL, 2.5%), and H 2 O (250 μL, 2.5%) were added and stirred at room temperature for 1 to 2 hours. After confirming the completion of the reaction by TLC, insoluble impurities were removed by celite filtration. The resulting filtrate was added with cold diisopropylether to precipitate the peptide, and the solution was transferred to a Falcon tube and subjected to freeze centrifugation (3500 rpm, 15 min, -9°C). The supernatant was discarded, and the same procedure was repeated twice more to obtain a solid. The resulting compound was then purified by reversed-phase HPLC.
〔ペプチド1(ELQDALDIHWRE)の合成〕
ペプチド1の構造式を下記に示した。
[Synthesis of peptide 1 (ELQDALDIHWRE)]
The structural formula of peptide 1 is shown below.
先ず、TAG-OH(1514.9mg、2.0mmol)とFmoc-Glu(OtBu)-OH・H2O(1154.0mg、2.6mmol、1.3eq)をdry DCM(48mL)に溶解させ、撹拌しながらDMAP(24.7mg、0.2mmol、0.1eq)、DIPCI(400μL、2.6mmol、1.3eq)を添加し、室温で1.75時間攪拌した。TLCによる反応終了確認後、Acetonitrileを加えて目的物を析出させ、Acetonitrileで吸引ろ過、洗浄を行い、Fmoc-Glu(OtBu)-TAGの固体を捕集した。 First, TAG-OH (1514.9 mg, 2.0 mmol) and Fmoc-Glu(OtBu)-OH.H 2 O (1154.0 mg, 2.6 mmol, 1.3 eq) were dissolved in dry DCM (48 mL), and DMAP (24.7 mg, 0.2 mmol, 0.1 eq) and DIPCI (400 μL, 2.6 mmol, 1.3 eq) were added while stirring, and the mixture was stirred at room temperature for 1.75 hours. After confirming the completion of the reaction by TLC, acetonitrile was added to precipitate the target product, which was then suction filtered and washed with acetonitrile to collect the solid Fmoc-Glu(OtBu)-TAG.
その後、ペプチド1のアミノ酸配列に従って、順次、ペプチドの脱保護と伸長を11サイクル行い、Boc-E(OtBu)-L-Q(Trt)-D(OtBu)-A-L-D(OtBu)-I-H(Trt)-W(Boc)-R(Pbf)-E(OtBu)-TAGの固体(1785.9mg、0.5213mmol、over 23 steps、overall yield 26%)を得た。最後にBoc-E(OtBu)-L-Q(Trt)-D(OtBu)-A-L-D(OtBu)-I-H(Trt)-W(Boc)-R(Pbf)-E(OtBu)-TAG(83.7 mg)の全脱保護を行い、ペプチド1の固体(29.4mg)を得た。 After that, 11 cycles of deprotection and elongation of the peptide were performed in sequence according to the amino acid sequence of peptide 1, and a solid of Boc-E(OtBu)-L-Q(Trt)-D(OtBu)-A-L-D(OtBu)-I-H(Trt)-W(Boc)-R(Pbf)-E(OtBu)-TAG (1785.9 mg, 0.5213 mmol, over 23 steps, overall yield 26%) was obtained. Finally, full deprotection of Boc-E(OtBu)-L-Q(Trt)-D(OtBu)-A-L-D(OtBu)-I-H(Trt)-W(Boc)-R(Pbf)-E(OtBu)-TAG (83.7 mg) was performed to obtain a solid of peptide 1 (29.4 mg).
HPLCによる分離精製は、溶媒をA液:H2O(0.1% TFA)、B液:Acetonitrile(0.1% TFA)とし、B液の比率;start~10min:20% → 10min~40min:20%~50% → 40 min~60min:50%(finish)で行った。
・MALDI-TOFMS Calcd. for [C67H102N19O22]+: 1524.7442; found: 1524.7438
Separation and purification by HPLC was performed using solvents A: H2O (0.1% TFA) and B: Acetonitrile (0.1% TFA), with the ratio of B solution being: start to 10 min: 20% → 10 min to 40 min: 20% to 50% → 40 min to 60 min: 50% (finish).
・MALDI-TOFMS Calcd. for [C 67 H 102 N 19 O 22 ] + : 1524.7442; found: 1524.7438
〔ペプチド2(ELQDAL)の合成〕
ペプチド2の構造式を下記に示した。
[Synthesis of peptide 2 (ELQDAL)]
The structural formula of peptide 2 is shown below.
先ず、TAG-OH(757.9mg、1.0mmol)とFmoc-Leu-OH(459.8mg、1.3mmol、1.3eq)をdry DCM(22mL)に溶解させ、撹拌しながらDMAP(12.5mg、0.1mmol、0.1eq)、DIPCI(200μL、1.3mmol、1.3eq)を添加し、室温で1時間攪拌した。TLCによる反応終了確認後、Acetonitrileを加えて目的物を析出させ、Acetonitrileで吸引ろ過、洗浄を行い、Fmoc-Leu-TAGの固体を捕集した。 First, TAG-OH (757.9 mg, 1.0 mmol) and Fmoc-Leu-OH (459.8 mg, 1.3 mmol, 1.3 eq) were dissolved in dry DCM (22 mL), and DMAP (12.5 mg, 0.1 mmol, 0.1 eq) and DIPCI (200 μL, 1.3 mmol, 1.3 eq) were added while stirring, and the mixture was stirred at room temperature for 1 hour. After confirming the completion of the reaction by TLC, acetonitrile was added to precipitate the target product, which was then suction filtered and washed with acetonitrile to collect the solid Fmoc-Leu-TAG.
その後、ペプチド2のアミノ酸配列に従って、順次、ペプチドの脱保護と伸長を5サイクル行い、Boc-E(OtBu)-L-Q(Trt)-D(OtBu)-A-L-TAGの固体(1343.7mg、0.7140mmol、over 11 steps、overall yield 71%)を得た。最後にBoc-E(OtBu)-L-Q(Trt)-D(OtBu)-A-L-TAG(110.8 mg)の全脱保護を行い、ペプチド2の固体(28.9 mg)を得た。 After that, five cycles of peptide deprotection and elongation were performed in sequence according to the amino acid sequence of peptide 2, yielding a solid of Boc-E(OtBu)-L-Q(Trt)-D(OtBu)-A-L-TAG (1343.7 mg, 0.7140 mmol, over 11 steps, overall yield 71%). Finally, Boc-E(OtBu)-L-Q(Trt)-D(OtBu)-A-L-TAG (110.8 mg) was completely deprotected to obtain a solid of peptide 2 (28.9 mg).
HPLCによる分離精製は溶媒をA液:H2O(0.1% TFA)、B液:Acetonitrile(0.1% TFA)とし、B液の比率;start~15min:20% → 15min~75min:20%~50% → 75min~100min:50%(finish)で行った。
・MALDI-TOFMS Calcd. for [C29H49N7O12Na]+: 710.3331; found: 710.3338
Separation and purification by HPLC was performed using solvents A: H 2 O (0.1% TFA) and B: Acetonitrile (0.1% TFA), with the ratio of B solution being: start to 15 min: 20% → 15 min to 75 min: 20% to 50% → 75 min to 100 min: 50% (finish).
・MALDI-TOFMS Calcd. for [C 29 H 49 N 7 O 12 Na] + : 710.3331; found: 710.3338
〔ペプチド3(LQDALD)の合成〕
ペプチド3の構造式を下記に示した。
[Synthesis of peptide 3 (LQDALD)]
The structural formula of peptide 3 is shown below.
先ず、TAG-OH(757.9mg、1.0mmol)とFmoc-Asp(OtBu)-OH(535.0mg、1.3mmol、1.3eq)をdry DCM(30mL)に溶解させ、撹拌しながらDMAP(12.2mg、0.1mmol、0.1eq)、DIPCI(200 μL、1.3mmol、1.3 eq)を添加し、室温で1時間攪拌した。TLCによる反応終了確認後、Acetonitrileを加えて目的物を析出させ、Acetonitrileで吸引ろ過、洗浄を行い、Fmoc-Asp(OtBu)-TAGの固体を捕集した。 First, TAG-OH (757.9 mg, 1.0 mmol) and Fmoc-Asp(OtBu)-OH (535.0 mg, 1.3 mmol, 1.3 eq) were dissolved in dry DCM (30 mL), and DMAP (12.2 mg, 0.1 mmol, 0.1 eq) and DIPCI (200 μL, 1.3 mmol, 1.3 eq) were added while stirring, and the mixture was stirred at room temperature for 1 hour. After confirming the completion of the reaction by TLC, acetonitrile was added to precipitate the target product, which was then suction filtered and washed with acetonitrile to collect the solid Fmoc-Asp(OtBu)-TAG.
その後、ペプチド3のアミノ酸配列に従って、順次、ペプチドの脱保護と伸長を5サイクル行い、Boc-L-Q(Trt)-D(OtBu)-A-L-D(OtBu)-TAGの固体(1337.9mg、0.6724mmol、over 11 steps、overall yield 67%)を得た。最後にBoc-L-Q(Trt)-D(OtBu)-ALD-(OtBu)-TAG(104.1 mg)の全脱保護を行い、ペプチド3の固体(22.7mg)を得た。 After that, five cycles of peptide deprotection and elongation were performed in sequence according to the amino acid sequence of peptide 3, yielding a solid of Boc-L-Q(Trt)-D(OtBu)-A-L-D(OtBu)-TAG (1337.9 mg, 0.6724 mmol, over 11 steps, overall yield 67%). Finally, full deprotection of Boc-L-Q(Trt)-D(OtBu)-ALD-(OtBu)-TAG (104.1 mg) was performed to obtain a solid of peptide 3 (22.7 mg).
HPLCによる分離精製は溶媒をA液:H2O(0.1% TFA)、B液:Acetonitrile(0.1% TFA)とし、B液の比率;start~15min:20% → 15min~75min:20%~50% →75min~100min:50%(finish)で行った。
・MALDI-TOFMS Calcd. for [C28H47N7O12Na]+: 696.3175; found: 696.3495
Separation and purification by HPLC was performed using solvents A: H2O (0.1% TFA) and B: Acetonitrile (0.1% TFA), with the ratio of B solution being: start to 15 min: 20% → 15 min to 75 min: 20% to 50% → 75 min to 100 min: 50% (finish).
・MALDI-TOFMS Calcd. for [C 28 H 47 N 7 O 12 Na] + : 696.3175; found: 696.3495
〔ペプチド4(QDALDI)の合成〕
ペプチド4の構造式を下記に示した。
[Synthesis of peptide 4 (QDALDI)]
The structural formula of peptide 4 is shown below.
先ず、TAG-OH(757.0 mg、1.0mmol)とFmoc-Ile-OH(460.2mg、1.3mmol、1.3eq)をdry DCM(31mL)に溶解させ、撹拌しながらDMAP(12.7mg、0.1mmol、0.1eq)、DIPCI(200μL、1.3mmol、1.3eq)を添加し、室温で1時間攪拌した。TLCによる反応終了確認後、Acetonitrileを加えて目的物を析出させ、Acetonitrileで吸引ろ過、洗浄を行い、Fmoc-Ile-TAGの固体を捕集した。 First, TAG-OH (757.0 mg, 1.0 mmol) and Fmoc-Ile-OH (460.2 mg, 1.3 mmol, 1.3 eq) were dissolved in dry DCM (31 mL), and DMAP (12.7 mg, 0.1 mmol, 0.1 eq) and DIPCI (200 μL, 1.3 mmol, 1.3 eq) were added while stirring, and the mixture was stirred at room temperature for 1 hour. After confirming the completion of the reaction by TLC, acetonitrile was added to precipitate the target product, which was then suction filtered and washed with acetonitrile to collect the solid Fmoc-Ile-TAG.
その後、ペプチド4のアミノ酸配列に従って、順次、ペプチドの脱保護と伸長を5サイクル行い、Boc-Q(Trt)-D(OtBu)-A-L-D(OtBu)-I-TAGの固体(1477.6mg、0.7911mmol、over 11 steps、overall yield 79%)を得た。最後にBoc-Q(Trt)-D(OtBu)-A-L-D(OtBu)-I-TAG(91.9mg)の全脱保護を行い、ペプチド4の固体(25.4mg)を得た。 After that, five cycles of peptide deprotection and elongation were performed in sequence according to the amino acid sequence of peptide 4, yielding a solid of Boc-Q(Trt)-D(OtBu)-A-L-D(OtBu)-I-TAG (1477.6 mg, 0.7911 mmol, over 11 steps, overall yield 79%). Finally, full deprotection of Boc-Q(Trt)-D(OtBu)-A-L-D(OtBu)-I-TAG (91.9 mg) was performed to obtain a solid of peptide 4 (25.4 mg).
HPLCによる分離精製は溶媒をA液:H2O(0.1% TFA)、B液:Acetonitrile(0.1% TFA)とし、B液の比率;start~10 min:20% → 10min~40min:20%~50% →40min ~60min:50%(finish)で行った。
・MALDI-TOFMS Calcd. for [C28H47N7O12Na]+: 696.3175; found: 696.3332
Separation and purification by HPLC was performed using solvents A: H2O (0.1% TFA) and B: Acetonitrile (0.1% TFA), with the ratio of B solution being: start to 10 min: 20% → 10 min to 40 min: 20% to 50% → 40 min to 60 min: 50% (finish).
・MALDI-TOFMS Calcd. for [C 28 H 47 N 7 O 12 Na] + : 696.3175; found: 696.3332
〔ペプチド5(DALDIH)の合成〕
ペプチド5の構造式を下記に示した。
[Synthesis of peptide 5 (DALDIH)]
The structural formula of peptide 5 is shown below.
先ず、TAG-OH(757.8mg、1.0mmol)とFmoc-His(Trt)-OH(806.0mg、1.3mmol、1.3eq)をdry DCM(23mL)に溶解させ、撹拌しながらDMAP(12.2mg、0.1mmol、0.1eq)、DIPCI(200μL、1.3mmol、1.3eq)を添加し、室温で1時間攪拌した。TLCによる反応終了確認後、Acetonitrileを加えて目的物を析出させ、Acetonitrileで吸引ろ過、洗浄を行い、Fmoc-His(Trt)-TAGの固体を捕集した。 First, TAG-OH (757.8 mg, 1.0 mmol) and Fmoc-His(Trt)-OH (806.0 mg, 1.3 mmol, 1.3 eq) were dissolved in dry DCM (23 mL), and DMAP (12.2 mg, 0.1 mmol, 0.1 eq) and DIPCI (200 μL, 1.3 mmol, 1.3 eq) were added while stirring, and the mixture was stirred at room temperature for 1 hour. After confirming the completion of the reaction by TLC, acetonitrile was added to precipitate the target product, which was then suction filtered and washed with acetonitrile to collect the solid Fmoc-His(Trt)-TAG.
その後、ペプチド5のアミノ酸配列に従って、順次、ペプチドの脱保護と伸長を5サイクル行い、Boc-D(OtBu)-A-L-D(OtBu)-I-H(Trt)-TAGの固体(1368.9mg、0.7294mmol、over 11 steps、overall yield 73%)を得た。最後にBoc-D(OtBu)-A-L-D(OtBu)-I-H(Trt)-TAG(64.5mg)の全脱保護を行い、ペプチド5の固体(15.3mg)を得た。 After that, five cycles of peptide deprotection and elongation were performed in sequence according to the amino acid sequence of peptide 5, yielding a solid of Boc-D(OtBu)-A-L-D(OtBu)-I-H(Trt)-TAG (1368.9 mg, 0.7294 mmol, over 11 steps, overall yield 73%). Finally, Boc-D(OtBu)-A-L-D(OtBu)-I-H(Trt)-TAG (64.5 mg) was completely deprotected to obtain a solid of peptide 5 (15.3 mg).
HPLCによる分離精製は溶媒をA液:H2O(0.1% TFA)、B液:Acetonitrile(0.1% TFA)とし、B液の比率;start~15 min:20% → 15min~75 min:20%~50% → 75min~100min:50%(finish)で行った。
・MALDI-TOFMS Calcd. for [C29H46N8O11Na]+: 705.3178; found: 705.3247
Separation and purification by HPLC was performed using solvents A: H2O (0.1% TFA) and B: Acetonitrile (0.1% TFA), with the ratio of B solution being: start to 15 min: 20% → 15 min to 75 min: 20% to 50% → 75 min to 100 min: 50% (finish).
・MALDI-TOFMS Calcd. for [C 29 H 46 N 8 O 11 Na] + : 705.3178; found: 705.3247
〔ペプチド6(ALDIHW)の合成〕
ペプチド6の構造式を下記に示した。
[Synthesis of peptide 6 (ALDIHW)]
The structural formula of peptide 6 is shown below.
先ず、TAG-OH(758.6mg、1.0mmol)とFmoc-Trp(Boc)-OH(684.6mg、1.3mmol、1.3eq)をdry DCM(26mL)に溶解させ、撹拌しながらDMAP(12.9mg、0.1mmol、0.1eq)、DIPCI(200μL、1.3mmol、1.3eq)を添加し、室温で3時間攪拌した。TLCによる反応終了確認後、Acetonitrileを加えて目的物を析出させ、Acetonitrileで吸引ろ過、洗浄を行い、Fmoc-Trp(Boc)-TAGの固体を捕集した。 First, TAG-OH (758.6 mg, 1.0 mmol) and Fmoc-Trp(Boc)-OH (684.6 mg, 1.3 mmol, 1.3 eq) were dissolved in dry DCM (26 mL), and DMAP (12.9 mg, 0.1 mmol, 0.1 eq) and DIPCI (200 μL, 1.3 mmol, 1.3 eq) were added while stirring, and the mixture was stirred at room temperature for 3 hours. After confirming the completion of the reaction by TLC, acetonitrile was added to precipitate the target product, which was then suction filtered and washed with acetonitrile to collect the solid Fmoc-Trp(Boc)-TAG.
その後、ペプチド6のアミノ酸配列に従って、順次、ペプチドの脱保護と伸長を5サイクル行い、Boc-A-L-D(OtBu)-I-H(Trt)-W(Boc)-TAGの固体(1486.0mg、0.7460mmol、over 11 steps、overall yield 75%)を得た。最後にBoc-A-L-D(OtBu)-I-H(Trt)-W(Boc)-TAG(62.5mg)の全脱保護を行い、ペプチド6の固体(15.4mg)を得た。 After that, five cycles of peptide deprotection and elongation were performed in sequence according to the amino acid sequence of peptide 6, to obtain a solid of Boc-A-L-D(OtBu)-I-H(Trt)-W(Boc)-TAG (1486.0 mg, 0.7460 mmol, over 11 steps, overall yield 75%). Finally, full deprotection of Boc-A-L-D(OtBu)-I-H(Trt)-W(Boc)-TAG (62.5 mg) was performed to obtain a solid of peptide 6 (15.4 mg).
HPLCによる分離精製は溶媒をA液:H2O(0.1% TFA)、B液:Acetonitrile(0.1% TFA)とし、B液の比率;start~15min:20% → 15min~75min:20%~50% → 75min~100min:50%(finish)で行った。
・MALDI-TOFMS Calcd. for [C36H51N9O9Na]+: 776.3702; found: 776.4098
Separation and purification by HPLC was performed using solvents A: H 2 O (0.1% TFA) and B: Acetonitrile (0.1% TFA), with the ratio of B solution being: start to 15 min: 20% → 15 min to 75 min: 20% to 50% → 75 min to 100 min: 50% (finish).
・MALDI-TOFMS Calcd. for [C 36 H 51 N 9 O 9 Na] + : 776.3702; found: 776.4098
〔ペプチド7(LDIHWR)の合成〕
ペプチド7の構造式を下記に示した。
[Synthesis of peptide 7 (LDIHWR)]
The structural formula of peptide 7 is shown below.
先ず、TAG-OH(758.1 mg, 1.0mmol)とFmoc-Arg(Pbf)-OH(924.1 mg, 1.3 mmol, 1.3eq)をdry DCM(24 mL)に溶解させ、撹拌しながらDMAP(12.4 mg, 0.1 mmol, 0.1 eq)、DIPCI(200 μL, 1.3 mmol, 1.3 eq)を添加し、室温で1.75時間攪拌した。TLCによる反応終了確認後、Acetonitrileを加えて目的物を析出させ、Acetonitrileで吸引ろ過、洗浄を行い、Fmoc-Arg(Pbf)-TAGの固体を捕集した。 First, TAG-OH (758.1 mg, 1.0 mmol) and Fmoc-Arg(Pbf)-OH (924.1 mg, 1.3 mmol, 1.3 eq) were dissolved in dry DCM (24 mL), and DMAP (12.4 mg, 0.1 mmol, 0.1 eq) and DIPCI (200 μL, 1.3 mmol, 1.3 eq) were added while stirring, and the mixture was stirred at room temperature for 1.75 hours. After confirming the completion of the reaction by TLC, acetonitrile was added to precipitate the target product, which was then suction filtered and washed with acetonitrile to collect the solid Fmoc-Arg(Pbf)-TAG.
その後、ペプチドの脱保護と伸長を5 サイクル行い、Boc-L-D(OtBu)-I-H (Trt)-W(Boc)-Arg(Pbf)-TAGの固体(1481.1 mg, 0.6358 mmol, over 11 steps, overall yield 64%)を得た。最後にBoc-L-D(OtBu)-I-H (Trt)-W(Boc)-Arg(Pbf)-TAG(101.6 mg, 43.62 μmol)の全脱保護を行い、ペプチド7の固体(27.1 mg)を得た。 After that, five cycles of peptide deprotection and elongation were performed to obtain a solid of Boc-L-D(OtBu)-I-H (Trt)-W(Boc)-Arg(Pbf)-TAG (1481.1 mg, 0.6358 mmol, over 11 steps, overall yield 64%). Finally, full deprotection of Boc-L-D(OtBu)-I-H (Trt)-W(Boc)-Arg(Pbf)-TAG (101.6 mg, 43.62 μmol) was performed to obtain a solid of peptide 7 (27.1 mg).
HPLCによる分離精製は溶媒をA液:H2O(0.1% TFA)、B液:Acetonitrile(0.1% TFA)とし、B液の比率;start~15min:20% → 15min~75min:20%~50% → 75min~100min:50%(finish)で行った。
・MALDI-TOFMS Calcd. for [C39H58N12O9]+: 838.4450; found: 839.4566
Separation and purification by HPLC was performed using solvents A: H 2 O (0.1% TFA) and B: Acetonitrile (0.1% TFA), with the ratio of B solution being: start to 15 min: 20% → 15 min to 75 min: 20% to 50% → 75 min to 100 min: 50% (finish).
・MALDI-TOFMS Calcd. for [C 39 H 58 N 12 O 9 ] + : 838.4450; found: 839.4566
〔ペプチド8(DIHWRE)の合成〕
ペプチド8の構造式を下記に示した。
[Synthesis of peptide 8 (DIHWRE)]
The structural formula of peptide 8 is shown below.
先ず、TAG-OH(757.3mg、1.0mmol)とFmoc-Glu(OtBu)-OH・H2O(576.8mg、1.3mmol、1.3eq)をdry DCM(23mL)に溶解させ、撹拌しながらDMAP(12.8mg、0.1mmol、0.1eq)、DIPCI(200μL、1.3mmol、1.3eq)を添加し、室温で2時間攪拌した。TLCによる反応終了確認後、Acetonitrileを加えて目的物を析出させ、Acetonitrileで吸引ろ過、洗浄を行い、Fmoc-Glu(OtBu)-TAGの固体を捕集した。 First, TAG-OH (757.3 mg, 1.0 mmol) and Fmoc-Glu(OtBu) -OH.H2O (576.8 mg, 1.3 mmol, 1.3 eq) were dissolved in dry DCM (23 mL), and DMAP (12.8 mg, 0.1 mmol, 0.1 eq) and DIPCI (200 μL, 1.3 mmol, 1.3 eq) were added while stirring, and the mixture was stirred at room temperature for 2 hours. After confirming the completion of the reaction by TLC, acetonitrile was added to precipitate the target product, which was then suction filtered and washed with acetonitrile to collect the solid Fmoc-Glu(OtBu)-TAG.
その後、ペプチド8のアミノ酸配列に従って、順次、ペプチドの脱保護と伸長を5サイクル行い、Boc-D(OtBu)-I-H(Trt)-W(Boc)-R(Pbf)-E(OtBu)-TAGの固体(1540.8mg、0.6416mmol、over 11 steps、overall yield 64%)を得た。最後にBoc-D(OtBu)-I-H(Trt)-W(Boc)-R(Pbf)-E(OtBu)-TAG(91.4 mg)の全脱保護を行い、ペプチド8の固体(21.9mg)を得た。 After that, five cycles of peptide deprotection and elongation were performed in sequence according to the amino acid sequence of peptide 8, yielding a solid of Boc-D(OtBu)-I-H(Trt)-W(Boc)-R(Pbf)-E(OtBu)-TAG (1540.8 mg, 0.6416 mmol, over 11 steps, overall yield 64%). Finally, full deprotection of Boc-D(OtBu)-I-H(Trt)-W(Boc)-R(Pbf)-E(OtBu)-TAG (91.4 mg) was performed to obtain a solid of peptide 8 (21.9 mg).
HPLCによる分離精製は溶媒をA液:H2O(0.1% TFA)、B液:Acetonitrile(0.1% TFA)とし、B液の比率;start~10min:20% → 10min~40min:20%~50% → 40min~45min:50%(finish)で行った。
・MALDI-TOFMS Calcd. for [C38H54N12O11Na]+: 877.3927; found: 877.4598
Separation and purification by HPLC was performed using solvents A: H 2 O (0.1% TFA) and B: Acetonitrile (0.1% TFA), with the ratio of B solution being: start to 10 min: 20% → 10 min to 40 min: 20% to 50% → 40 min to 45 min: 50% (finish).
・MALDI-TOFMS Calcd. for [C 38 H 54 N 12 O 11 Na] + : 877.3927; found: 877.4598
〔ペプチド9(DAL)の合成〕
ペプチド9の構造式を下記に示した。
Synthesis of peptide 9 (DAL)
The structural formula of peptide 9 is shown below.
先ず、TAG-OH(755.3mg、1.0mmol)とFmoc-Leu-OH(458.6mg、1.3mmol、1.3eq)をdry DCM(22mL)に溶解させ、撹拌しながらDMAP(13.7mg、0.1mmol、0.1eq)、DIPCI(200μL、1.3mmol、1.3eq)を添加し、室温で1時間攪拌した。TLCによる反応終了確認後、Acetonitrileを加えて目的物を析出させ、Acetonitrileで吸引ろ過、洗浄を行い、Fmoc-Leu-TAGの固体を捕集した。 First, TAG-OH (755.3 mg, 1.0 mmol) and Fmoc-Leu-OH (458.6 mg, 1.3 mmol, 1.3 eq) were dissolved in dry DCM (22 mL), and DMAP (13.7 mg, 0.1 mmol, 0.1 eq) and DIPCI (200 μL, 1.3 mmol, 1.3 eq) were added while stirring, and the mixture was stirred at room temperature for 1 hour. After confirming the completion of the reaction by TLC, acetonitrile was added to precipitate the target product, which was then suction filtered and washed with acetonitrile to collect the solid Fmoc-Leu-TAG.
その後、ペプチド9のアミノ酸配列に従って、順次、ペプチドの脱保護と伸長を2サイクル行い、Boc-D(OtBu)-A-L-TAGの固体(1041.0mg、0.8582mmol、over 5 steps、overall yield 86%)を得た。最後にBoc-D(OtBu)-A-L-TAG(102.1mg)の全脱保護を行い、ペプチド9の固体(16.8mg)を得た。 After that, two cycles of peptide deprotection and elongation were performed in sequence according to the amino acid sequence of peptide 9, yielding a solid of Boc-D(OtBu)-A-L-TAG (1041.0 mg, 0.8582 mmol, over 5 steps, overall yield 86%). Finally, Boc-D(OtBu)-A-L-TAG (102.1 mg) was completely deprotected to obtain a solid of peptide 9 (16.8 mg).
HPLCによる分離精製は溶媒をA液:H2O(0.1% TFA)、B液:Acetonitrile(0.1% TFA)とし、B液の比率;start~10 min:20% → 10min~40min:20%~50% → 40min~45min:50%(finish)で行った。
・MALDI-TOFMS Calcd. for [C13H23N3O6Na]+: 340.1479; found: 340.1232
Separation and purification by HPLC was performed using solvents A: H 2 O (0.1% TFA) and B: Acetonitrile (0.1% TFA), with the ratio of B solution being: start to 10 min: 20% → 10 min to 40 min: 20% to 50% → 40 min to 45 min: 50% (finish).
・MALDI-TOFMS Calcd. for [C 13 H 23 N 3 O 6 Na] + : 340.1479; found: 340.1232
〔ペプチド10(DALD)の合成〕
ペプチド10の構造式を下記に示した。
[Synthesis of peptide 10 (DALD)]
The structural formula of peptide 10 is shown below.
先ず、TAG-OH(756.6mg、1.0mmol)とFmoc-Asp(OtBu)-OH(536.0mg、1.3mmol、1.3eq)をdry DCM(26mL)に溶解させ、撹拌しながらDMAP(13.0mg、0.1mmol、0.1eq)、DIPCI(200μL、1.3mmol、1.3eq)を添加し、室温で1時間攪拌した。TLCによる反応終了確認後、Acetonitrileを加えて目的物を析出させ、Acetonitrileで吸引ろ過、洗浄を行い、Fmoc-Asp(OtBu)-TAGの固体を捕集した。 First, TAG-OH (756.6 mg, 1.0 mmol) and Fmoc-Asp(OtBu)-OH (536.0 mg, 1.3 mmol, 1.3 eq) were dissolved in dry DCM (26 mL), and DMAP (13.0 mg, 0.1 mmol, 0.1 eq) and DIPCI (200 μL, 1.3 mmol, 1.3 eq) were added while stirring, and the mixture was stirred at room temperature for 1 hour. After confirming the completion of the reaction by TLC, acetonitrile was added to precipitate the target product, which was then suction filtered and washed with acetonitrile to collect the solid Fmoc-Asp(OtBu)-TAG.
その後、ペプチド10のアミノ酸配列に従って、順次、ペプチドの脱保護と伸長を3サイクル行い、Boc-D(OtBu)-A-L-D(OtBu)-TAGの固体(1184.6mg、0.8558mmol、over 7 steps、overall yield 86%)を得た。最後にBoc-D(OtBu)-A-L-D(OtBu)-TAG(100.6mg)の全脱保護を行い、ペプチド10の固体(20.8mg)を得た。 After that, three cycles of peptide deprotection and elongation were performed in sequence according to the amino acid sequence of peptide 10, to obtain a solid of Boc-D(OtBu)-A-L-D(OtBu)-TAG (1184.6 mg, 0.8558 mmol, over 7 steps, overall yield 86%). Finally, Boc-D(OtBu)-A-L-D(OtBu)-TAG (100.6 mg) was completely deprotected to obtain a solid of peptide 10 (20.8 mg).
HPLCによる分離精製は溶媒をA液:H2O(0.1% TFA)、B液:Acetonitrile(0.1% TFA)とし、B液の比率;start~10 min:20% → 10min~40min:20%~50% → 40min~45min:50%(finish)で行った。
・MALDI-TOFMS Calcd. for [C17H28N4O9Na]+: 455.1748; found: 455.1732.
Separation and purification by HPLC was performed using solvents A: H 2 O (0.1% TFA) and B: Acetonitrile (0.1% TFA), with the ratio of B solution being: start to 10 min: 20% → 10 min to 40 min: 20% to 50% → 40 min to 45 min: 50% (finish).
・MALDI-TOFMS Calcd. for [C 17 H 28 N 4 O 9 Na] + : 455.1748; found: 455.1732.
〔キプリス幼生付着誘起試験〕
以上のように化学的に合成したペプチド1~10について、それぞれキプリス幼生に対する付着誘起作用を有するか試験した。先ず、ポリスチレン製24穴プレートの各ウェルに、各ペプチドを0.1、1.0、10(μg/mL)の濃度になるように溶解したDMSO 2%溶液を含む80%ろ過海水2mLを加えた。その後、各穴にタテジマフジツボキプリス幼生を6匹ずつ加え、25℃で暗所静置し、6時間後及び24時間後にキプリス幼生の観察を行った。各穴におけるキプリス幼生の付着個体数と死亡個体数を計測し、各サンプル濃度における付着誘起率と死亡率と算出した。
[Cypris larvae attachment induction test]
The peptides 1 to 10 thus chemically synthesized were tested for their adhesion-inducing effect on cypris larvae. First, 2 mL of 80% filtered seawater containing a 2% DMSO solution in which each peptide was dissolved to a concentration of 0.1, 1.0, or 10 (μg/mL) was added to each well of a 24-well polystyrene plate. Six Balsaminella cypris larvae were then added to each well, and the wells were left to stand in the dark at 25°C. The cypris larvae were observed 6 and 24 hours later. The number of attached and dead cypris larvae in each well was counted, and the adhesion induction rate and mortality rate at each sample concentration were calculated.
ペプチド1~10について付着誘起率と死亡率を算出した結果を図6~15に示した。また、ペプチドを含まないコントロール溶液を用いたときの付着誘起率と、ペプチドを含む溶液を用いたときの付着誘起率の最大値から付着増加率を算出した結果を表2及び3に示した。表2及び3は、それぞれキプリス幼生を添加してから6時間後及び24時間後に計測した結果を示している。 The results of calculating the attachment induction rate and mortality rate for peptides 1 to 10 are shown in Figures 6 to 15. In addition, the results of calculating the attachment increase rate from the attachment induction rate when a control solution containing no peptide was used and the maximum attachment induction rate when a solution containing peptide was used are shown in Tables 2 and 3. Tables 2 and 3 show the results measured 6 hours and 24 hours, respectively, after the addition of cypris larvae.
図6~15及び表2並びに3から、ペプチド1~5、9及び10は、キプリス幼生を溶液に加えた後24時間以内の条件下では、少なくとも20%以上の付着率向上効果を有することが理解できる。また、供試したペプチドの中でも特にペプチド3は、キプリス幼生の付着率向上効果が極めて優れていることが理解できる。一方、ペプチド6及び7については、キプリス幼生を溶液に加えた後6時間以内の条件下では付着率向上効果が見られるものの、同24時間後においては付着率向上効果が見られなかった。 From Figures 6 to 15 and Tables 2 and 3, it can be seen that peptides 1 to 5, 9, and 10 have an effect of improving the attachment rate by at least 20% or more within 24 hours after the cypris larvae are added to the solution. It can also be seen that, among the peptides tested, peptide 3 in particular has an extremely excellent effect of improving the attachment rate of cypris larvae. On the other hand, for peptides 6 and 7, although an effect of improving the attachment rate was observed within 6 hours after the cypris larvae were added to the solution, no effect of improving the attachment rate was observed 24 hours later.
この結果から、DAL(アスパラギン酸-アラニン-ロイシン)配列を含む3~12アミノ酸残基のアミノ酸配列からなるペプチドには、フジツボ類キプリス幼生の付着を誘起する作用を有することが明らかとなった。これらの結果から、DAL配列を含む3~12アミノ酸残基のアミノ酸配列からなるペプチドを用いてフジツボ類キプリス幼生の付着を誘起し、フジツボの付着を防止したい場所に対するフジツボ類の付着を防止することができることが示された。また、特に、LQDALD配列(配列番号3)からなるペプチドには、フジツボ類キプリス幼生の付着を誘起する極めて優れた作用を有することが明らかとなった。 These results demonstrate that peptides consisting of an amino acid sequence of 3 to 12 amino acid residues including the DAL (aspartic acid-alanine-leucine) sequence have the effect of inducing the attachment of barnacle cypris larvae. These results demonstrate that it is possible to induce the attachment of barnacle cypris larvae using peptides consisting of an amino acid sequence of 3 to 12 amino acid residues including the DAL sequence, and to prevent barnacle attachment to locations where barnacle attachment is desired to be prevented. In particular, it has been demonstrated that a peptide consisting of the LQDALD sequence (SEQ ID NO: 3) has an extremely excellent effect of inducing the attachment of barnacle cypris larvae.
1…フジツボ類トラップ装置、2…開口部、3…蓋部材、4…ゲル、5…筐体、10…構造体、11…浮遊型のフジツボ類トラップ装置、12…アンカー、13…縄、15…連結環、20…縄状或いは網状のフジツボ類トラップ装置、21…フロート部材 1...barnacle trapping device, 2...opening, 3...lid member, 4...gel, 5...casing, 10...structure, 11...floating barnacle trapping device, 12...anchor, 13...rope, 15...connecting ring, 20...rope-like or net-like barnacle trapping device, 21...float member
Claims (4)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2020098675A JP7527559B2 (en) | 2020-06-05 | 2020-06-05 | Adhesion-inducing peptides for barnacle cypris larvae and their applications |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2020098675A JP7527559B2 (en) | 2020-06-05 | 2020-06-05 | Adhesion-inducing peptides for barnacle cypris larvae and their applications |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2021191737A JP2021191737A (en) | 2021-12-16 |
| JP7527559B2 true JP7527559B2 (en) | 2024-08-05 |
Family
ID=78945696
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2020098675A Active JP7527559B2 (en) | 2020-06-05 | 2020-06-05 | Adhesion-inducing peptides for barnacle cypris larvae and their applications |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP7527559B2 (en) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN117624394B (en) * | 2023-11-28 | 2025-07-08 | 中国水产科学研究院黄海水产研究所 | A Navicula active substance for promoting the attachment and metamorphosis of bivalve molluscs and its application |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2001518287A (en) | 1997-09-30 | 2001-10-16 | ペビバ、アクチボラグ | Apoptosis-related compounds and uses thereof |
| JP2009155295A (en) | 2007-12-27 | 2009-07-16 | Central Res Inst Of Electric Power Ind | Proteins that induce the establishment of larvae |
| JP5931584B2 (en) | 2012-05-21 | 2016-06-08 | 一般財団法人電力中央研究所 | Pheromone trap for barnacle larvae and its production method |
-
2020
- 2020-06-05 JP JP2020098675A patent/JP7527559B2/en active Active
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2001518287A (en) | 1997-09-30 | 2001-10-16 | ペビバ、アクチボラグ | Apoptosis-related compounds and uses thereof |
| JP2009155295A (en) | 2007-12-27 | 2009-07-16 | Central Res Inst Of Electric Power Ind | Proteins that induce the establishment of larvae |
| JP5931584B2 (en) | 2012-05-21 | 2016-06-08 | 一般財団法人電力中央研究所 | Pheromone trap for barnacle larvae and its production method |
Non-Patent Citations (2)
| Title |
|---|
| BIOFOULING,2019年,VOL.35, NO.4,pp.416-428,https://doi.org/10.1080/08927014.2019.1602123 |
| Peptides,1989年,Vol.9,pp.1403-1406 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2021191737A (en) | 2021-12-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US6569993B1 (en) | Process for the preparation of resin-bound cyclic peptides | |
| US9206222B2 (en) | Solid phase peptide synthesis of peptide alcohols | |
| JP6150726B2 (en) | Cross-linked peptide containing a novel non-peptidic cross-linked structure, method for synthesizing the cross-linked peptide, and novel organic compound used in the method | |
| Goodman et al. | New reagents, reactions, and peptidomimetics for drug design | |
| US20200317721A1 (en) | A process for preparing a glucagon-like peptide | |
| JPH04500357A (en) | Atrial cyclic analogs of natriuretic peptides | |
| CA1268898A (en) | Growth hormone-releasing peptides and method of treating mammals therewith | |
| RS55193B1 (en) | PROCEDURE FOR THE PRODUCTION OF DEGARELIX AND ITS INTERMEDIATES | |
| RS55457B1 (en) | PROCEDURE FOR THE PRODUCTION OF DEGARELIX AND ITS INTERMEDIATES | |
| US20100197891A1 (en) | Method for peptide synthesis | |
| JP7527559B2 (en) | Adhesion-inducing peptides for barnacle cypris larvae and their applications | |
| Spiess et al. | Sequence analysis of rat hypothalamic corticotropin-releasing factor with the o-phthalaldehyde strategy | |
| CA2115246A1 (en) | Lanthionine bridged peptides | |
| US20160137689A1 (en) | Peptide-resin conjugate and use thereof | |
| US6673769B2 (en) | Lanthionine bridged peptides | |
| CN101072791A (en) | S-alkyl-sulphenyl protection groups in solid-phase synthesis | |
| CN118027136A (en) | A paper-assisted integrated green synthesis method for peptides | |
| ISOSTERE | SYNTHETIC APPROACHES TO BRIDGES IN PEPTIDES | |
| Bobrova et al. | A structure–activity study of nociceptin-(1–13)-peptide amide. Synthesis of analogues substituted in positions 0, 1, 3, 4 and 10 | |
| EGGLESTONE et al. | Peptide Synthesis | |
| Hlaváček et al. | Synthesis and utilization of peptides containing 3, 4-dehydroproline | |
| CN112500453A (en) | Solid-phase synthesis method of fragment of antibacterial peptide Iseganan | |
| Perlman | The syntheses and binding studies of fourteen peptide analogs of the C-terminal segments of avian pancreatic polypeptide and neuropeptide [gamma] | |
| MXPA00010056A (en) | Process for the preparation of resin-bound cyclic peptides | |
| JPH09221500A (en) | Calcitonin analogue |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20230421 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20240319 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20240319 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20240514 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20240606 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20240702 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20240711 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 7527559 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |