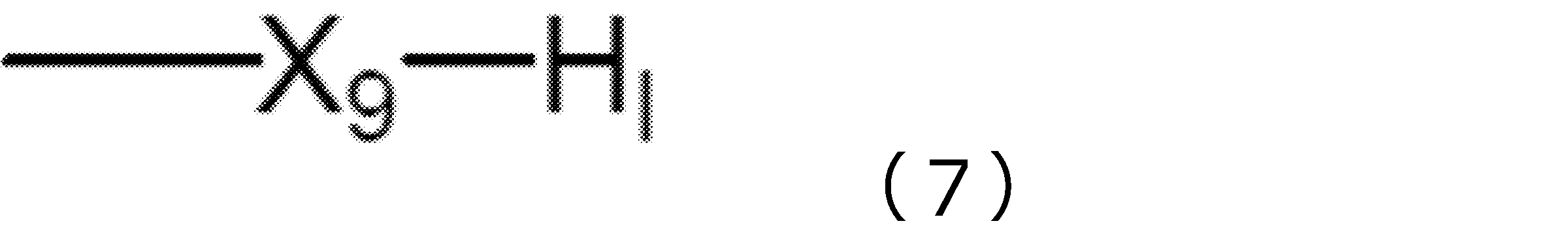JP7102658B2 - Whitening composition - Google Patents
Whitening composition Download PDFInfo
- Publication number
- JP7102658B2 JP7102658B2 JP2018101964A JP2018101964A JP7102658B2 JP 7102658 B2 JP7102658 B2 JP 7102658B2 JP 2018101964 A JP2018101964 A JP 2018101964A JP 2018101964 A JP2018101964 A JP 2018101964A JP 7102658 B2 JP7102658 B2 JP 7102658B2
- Authority
- JP
- Japan
- Prior art keywords
- carbon atoms
- acid
- group
- salt
- general formula
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 230000002087 whitening effect Effects 0.000 title claims description 45
- 239000000203 mixture Substances 0.000 title claims description 35
- 125000004432 carbon atom Chemical group C* 0.000 claims description 75
- 150000001875 compounds Chemical class 0.000 claims description 51
- 125000000217 alkyl group Chemical group 0.000 claims description 39
- 150000003839 salts Chemical class 0.000 claims description 38
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 29
- GYDJEQRTZSCIOI-LJGSYFOKSA-N tranexamic acid Chemical compound NC[C@H]1CC[C@H](C(O)=O)CC1 GYDJEQRTZSCIOI-LJGSYFOKSA-N 0.000 claims description 27
- 229960000401 tranexamic acid Drugs 0.000 claims description 27
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 23
- 125000001424 substituent group Chemical group 0.000 claims description 22
- 239000002253 acid Substances 0.000 claims description 18
- 229910052757 nitrogen Inorganic materials 0.000 claims description 15
- 125000004433 nitrogen atom Chemical group N* 0.000 claims description 15
- FPIPGXGPPPQFEQ-OVSJKPMPSA-N all-trans-retinol Chemical compound OC\C=C(/C)\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C FPIPGXGPPPQFEQ-OVSJKPMPSA-N 0.000 claims description 13
- 125000004430 oxygen atom Chemical group O* 0.000 claims description 12
- 239000002537 cosmetic Substances 0.000 claims description 11
- 125000003545 alkoxy group Chemical group 0.000 claims description 10
- 125000003118 aryl group Chemical group 0.000 claims description 10
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 9
- QIGBRXMKCJKVMJ-UHFFFAOYSA-N Hydroquinone Chemical compound OC1=CC=C(O)C=C1 QIGBRXMKCJKVMJ-UHFFFAOYSA-N 0.000 claims description 8
- FPIPGXGPPPQFEQ-UHFFFAOYSA-N 13-cis retinol Natural products OCC=C(C)C=CC=C(C)C=CC1=C(C)CCCC1(C)C FPIPGXGPPPQFEQ-UHFFFAOYSA-N 0.000 claims description 7
- 125000002252 acyl group Chemical group 0.000 claims description 7
- 150000002148 esters Chemical class 0.000 claims description 7
- WXTMDXOMEHJXQO-UHFFFAOYSA-N 2,5-dihydroxybenzoic acid Chemical compound OC(=O)C1=CC(O)=CC=C1O WXTMDXOMEHJXQO-UHFFFAOYSA-N 0.000 claims description 6
- 125000000623 heterocyclic group Chemical group 0.000 claims description 6
- 235000020944 retinol Nutrition 0.000 claims description 6
- 239000011607 retinol Substances 0.000 claims description 6
- 229960003471 retinol Drugs 0.000 claims description 6
- SNPLKNRPJHDVJA-ZETCQYMHSA-N D-panthenol Chemical compound OCC(C)(C)[C@@H](O)C(=O)NCCCO SNPLKNRPJHDVJA-ZETCQYMHSA-N 0.000 claims description 5
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 claims description 5
- 229930188104 Alkylresorcinol Natural products 0.000 claims description 4
- XBLVHTDFJBKJLG-UHFFFAOYSA-N Ethyl nicotinate Chemical compound CCOC(=O)C1=CC=CN=C1 XBLVHTDFJBKJLG-UHFFFAOYSA-N 0.000 claims description 4
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 claims description 4
- VYGQUTWHTHXGQB-FFHKNEKCSA-N Retinol Palmitate Chemical compound CCCCCCCCCCCCCCCC(=O)OC\C=C(/C)\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C VYGQUTWHTHXGQB-FFHKNEKCSA-N 0.000 claims description 4
- 125000002947 alkylene group Chemical group 0.000 claims description 4
- KVYGGMBOZFWZBQ-UHFFFAOYSA-N benzyl nicotinate Chemical compound C=1C=CN=CC=1C(=O)OCC1=CC=CC=C1 KVYGGMBOZFWZBQ-UHFFFAOYSA-N 0.000 claims description 4
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 4
- YNBADRVTZLEFNH-UHFFFAOYSA-N methyl nicotinate Chemical compound COC(=O)C1=CC=CN=C1 YNBADRVTZLEFNH-UHFFFAOYSA-N 0.000 claims description 4
- 235000004866 D-panthenol Nutrition 0.000 claims description 3
- 239000011703 D-panthenol Substances 0.000 claims description 3
- 125000001931 aliphatic group Chemical group 0.000 claims description 3
- 125000005257 alkyl acyl group Chemical group 0.000 claims description 3
- 125000002521 alkyl halide group Chemical group 0.000 claims description 3
- 125000003277 amino group Chemical group 0.000 claims description 3
- 125000006267 biphenyl group Chemical group 0.000 claims description 3
- 125000003262 carboxylic acid ester group Chemical group [H]C([H])([*:2])OC(=O)C([H])([H])[*:1] 0.000 claims description 3
- 239000000539 dimer Substances 0.000 claims description 3
- 229960005219 gentisic acid Drugs 0.000 claims description 3
- 125000005843 halogen group Chemical group 0.000 claims description 3
- 125000001624 naphthyl group Chemical group 0.000 claims description 3
- 125000004043 oxo group Chemical group O=* 0.000 claims description 3
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims description 3
- QGNJRVVDBSJHIZ-QHLGVNSISA-N retinyl acetate Chemical compound CC(=O)OC\C=C(/C)\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C QGNJRVVDBSJHIZ-QHLGVNSISA-N 0.000 claims description 3
- HBAIZGPCSAAFSU-UHFFFAOYSA-N 1-(2-hydroxyethyl)imidazolidin-2-one Chemical compound OCCN1CCNC1=O HBAIZGPCSAAFSU-UHFFFAOYSA-N 0.000 claims description 2
- WDQFELCEOPFLCZ-UHFFFAOYSA-N 1-(2-hydroxyethyl)pyrrolidin-2-one Chemical compound OCCN1CCCC1=O WDQFELCEOPFLCZ-UHFFFAOYSA-N 0.000 claims description 2
- JBRMEFWJFBHUKG-UHFFFAOYSA-N 4-[(diaminomethylideneamino)methyl]cyclohexane-1-carboxylic acid Chemical compound NC(N)=NCC1CCC(C(O)=O)CC1 JBRMEFWJFBHUKG-UHFFFAOYSA-N 0.000 claims description 2
- UEQVLKWMHAYVLO-MGCOHNPYSA-N C1C[C@@H](CN)CC[C@@H]1C(=O)OC1=CC=C(O)C=C1C(O)=O Chemical compound C1C[C@@H](CN)CC[C@@H]1C(=O)OC1=CC=C(O)C=C1C(O)=O UEQVLKWMHAYVLO-MGCOHNPYSA-N 0.000 claims description 2
- IFYATEIGOYKKKS-KYZUINATSA-N CC(=O)NC[C@H]1CC[C@H](C(O)=O)CC1 Chemical compound CC(=O)NC[C@H]1CC[C@H](C(O)=O)CC1 IFYATEIGOYKKKS-KYZUINATSA-N 0.000 claims description 2
- AVWTXYWHGFHGQD-ZKCHVHJHSA-N CNC(=O)[C@H]1CC[C@H](CN)CC1 Chemical compound CNC(=O)[C@H]1CC[C@H](CN)CC1 AVWTXYWHGFHGQD-ZKCHVHJHSA-N 0.000 claims description 2
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 2
- KLDXJTOLSGUMSJ-JGWLITMVSA-N Isosorbide Chemical compound O[C@@H]1CO[C@@H]2[C@@H](O)CO[C@@H]21 KLDXJTOLSGUMSJ-JGWLITMVSA-N 0.000 claims description 2
- MSCCTZZBYHQMQJ-AZAGJHQNSA-N Tocopheryl nicotinate Chemical compound C([C@@](OC1=C(C)C=2C)(C)CCC[C@H](C)CCC[C@H](C)CCCC(C)C)CC1=C(C)C=2OC(=O)C1=CC=CN=C1 MSCCTZZBYHQMQJ-AZAGJHQNSA-N 0.000 claims description 2
- GYDJEQRTZSCIOI-UHFFFAOYSA-N Tranexamic acid Chemical compound NCC1CCC(C(O)=O)CC1 GYDJEQRTZSCIOI-UHFFFAOYSA-N 0.000 claims description 2
- 125000004429 atom Chemical group 0.000 claims description 2
- 229950004580 benzyl nicotinate Drugs 0.000 claims description 2
- 229940064982 ethylnicotinate Drugs 0.000 claims description 2
- 239000001257 hydrogen Substances 0.000 claims description 2
- 229910052739 hydrogen Inorganic materials 0.000 claims description 2
- 229960002479 isosorbide Drugs 0.000 claims description 2
- 229960001238 methylnicotinate Drugs 0.000 claims description 2
- 235000020945 retinal Nutrition 0.000 claims description 2
- 239000011604 retinal Substances 0.000 claims description 2
- 230000002207 retinal effect Effects 0.000 claims description 2
- NCYCYZXNIZJOKI-OVSJKPMPSA-N retinal group Chemical group C\C(=C/C=O)\C=C\C=C(\C=C\C1=C(CCCC1(C)C)C)/C NCYCYZXNIZJOKI-OVSJKPMPSA-N 0.000 claims description 2
- 229960000342 retinol acetate Drugs 0.000 claims description 2
- 235000019173 retinyl acetate Nutrition 0.000 claims description 2
- 239000011770 retinyl acetate Substances 0.000 claims description 2
- 229940108325 retinyl palmitate Drugs 0.000 claims description 2
- 235000019172 retinyl palmitate Nutrition 0.000 claims description 2
- 239000011769 retinyl palmitate Substances 0.000 claims description 2
- 229950009883 tocopheryl nicotinate Drugs 0.000 claims description 2
- 150000001408 amides Chemical class 0.000 claims 2
- PHJIZHSZHXTTCK-DDHJBXDOSA-N (4s,5s,6s,7s)-2,9-dimethyldeca-2,8-diene-3,4,5,6,7,8-hexol Chemical compound CC(C)=C(O)[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)C(O)=C(C)C PHJIZHSZHXTTCK-DDHJBXDOSA-N 0.000 claims 1
- DFPAKSUCGFBDDF-ZQBYOMGUSA-N [14c]-nicotinamide Chemical compound N[14C](=O)C1=CC=CN=C1 DFPAKSUCGFBDDF-ZQBYOMGUSA-N 0.000 claims 1
- 229930002945 all-trans-retinaldehyde Natural products 0.000 claims 1
- 125000001183 hydrocarbyl group Chemical group 0.000 claims 1
- NCYCYZXNIZJOKI-UHFFFAOYSA-N vitamin A aldehyde Natural products O=CC=C(C)C=CC=C(C)C=CC1=C(C)CCCC1(C)C NCYCYZXNIZJOKI-UHFFFAOYSA-N 0.000 claims 1
- -1 Cyclic carboxamide Chemical class 0.000 description 80
- 239000000284 extract Substances 0.000 description 33
- 210000003491 skin Anatomy 0.000 description 17
- 208000012641 Pigmentation disease Diseases 0.000 description 14
- 238000000605 extraction Methods 0.000 description 12
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 11
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 11
- 230000019612 pigmentation Effects 0.000 description 11
- 235000014113 dietary fatty acids Nutrition 0.000 description 10
- 239000000194 fatty acid Substances 0.000 description 10
- 229930195729 fatty acid Natural products 0.000 description 10
- 230000002401 inhibitory effect Effects 0.000 description 10
- 239000002904 solvent Substances 0.000 description 10
- 239000002994 raw material Substances 0.000 description 9
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Natural products CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 8
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 8
- 230000000694 effects Effects 0.000 description 8
- 229920001296 polysiloxane Polymers 0.000 description 8
- PVNIIMVLHYAWGP-UHFFFAOYSA-N Niacin Chemical class OC(=O)C1=CC=CN=C1 PVNIIMVLHYAWGP-UHFFFAOYSA-N 0.000 description 7
- 102000003425 Tyrosinase Human genes 0.000 description 7
- 108060008724 Tyrosinase Proteins 0.000 description 7
- 239000004359 castor oil Substances 0.000 description 7
- 235000019438 castor oil Nutrition 0.000 description 7
- 235000011187 glycerol Nutrition 0.000 description 7
- ZEMPKEQAKRGZGQ-XOQCFJPHSA-N glycerol triricinoleate Natural products CCCCCC[C@@H](O)CC=CCCCCCCCC(=O)OC[C@@H](COC(=O)CCCCCCCC=CC[C@@H](O)CCCCCC)OC(=O)CCCCCCCC=CC[C@H](O)CCCCCC ZEMPKEQAKRGZGQ-XOQCFJPHSA-N 0.000 description 7
- 210000002752 melanocyte Anatomy 0.000 description 7
- 239000000419 plant extract Substances 0.000 description 7
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 6
- 239000006096 absorbing agent Substances 0.000 description 6
- 239000003795 chemical substances by application Substances 0.000 description 6
- 238000009472 formulation Methods 0.000 description 6
- 230000008099 melanin synthesis Effects 0.000 description 6
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 5
- 239000004480 active ingredient Substances 0.000 description 5
- FKRCODPIKNYEAC-UHFFFAOYSA-N ethyl propionate Chemical compound CCOC(=O)CC FKRCODPIKNYEAC-UHFFFAOYSA-N 0.000 description 5
- 239000000843 powder Substances 0.000 description 5
- 150000005846 sugar alcohols Chemical class 0.000 description 5
- PUPZLCDOIYMWBV-UHFFFAOYSA-N (+/-)-1,3-Butanediol Chemical compound CC(O)CCO PUPZLCDOIYMWBV-UHFFFAOYSA-N 0.000 description 4
- 239000004166 Lanolin Substances 0.000 description 4
- 229910019142 PO4 Inorganic materials 0.000 description 4
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 4
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 description 4
- 230000000052 comparative effect Effects 0.000 description 4
- 239000003814 drug Substances 0.000 description 4
- 239000003112 inhibitor Substances 0.000 description 4
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 4
- 235000019388 lanolin Nutrition 0.000 description 4
- 229940039717 lanolin Drugs 0.000 description 4
- 238000000034 method Methods 0.000 description 4
- YKYONYBAUNKHLG-UHFFFAOYSA-N n-Propyl acetate Natural products CCCOC(C)=O YKYONYBAUNKHLG-UHFFFAOYSA-N 0.000 description 4
- 239000003921 oil Substances 0.000 description 4
- 235000019198 oils Nutrition 0.000 description 4
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 4
- 235000021317 phosphate Nutrition 0.000 description 4
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 4
- 229940090181 propyl acetate Drugs 0.000 description 4
- 208000024891 symptom Diseases 0.000 description 4
- 238000012360 testing method Methods 0.000 description 4
- 229940088594 vitamin Drugs 0.000 description 4
- 229930003231 vitamin Natural products 0.000 description 4
- 235000013343 vitamin Nutrition 0.000 description 4
- 239000011782 vitamin Substances 0.000 description 4
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 3
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 3
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 3
- 208000003351 Melanosis Diseases 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 3
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 3
- 235000019482 Palm oil Nutrition 0.000 description 3
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 3
- 230000009471 action Effects 0.000 description 3
- 239000007844 bleaching agent Substances 0.000 description 3
- BTANRVKWQNVYAZ-UHFFFAOYSA-N butan-2-ol Chemical compound CCC(C)O BTANRVKWQNVYAZ-UHFFFAOYSA-N 0.000 description 3
- FHRSHSOEWXUORL-HDJSIYSDSA-N cetraxate Chemical compound C1C[C@@H](C[NH3+])CC[C@@H]1C(=O)OC1=CC=C(CCC([O-])=O)C=C1 FHRSHSOEWXUORL-HDJSIYSDSA-N 0.000 description 3
- 229950009533 cetraxate Drugs 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 229940079593 drug Drugs 0.000 description 3
- 150000002430 hydrocarbons Chemical group 0.000 description 3
- 230000006872 improvement Effects 0.000 description 3
- 230000010534 mechanism of action Effects 0.000 description 3
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 3
- GLDOVTGHNKAZLK-UHFFFAOYSA-N octadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCCCO GLDOVTGHNKAZLK-UHFFFAOYSA-N 0.000 description 3
- 239000002540 palm oil Substances 0.000 description 3
- 239000010452 phosphate Substances 0.000 description 3
- NQPDZGIKBAWPEJ-UHFFFAOYSA-N valeric acid Chemical compound CCCCC(O)=O NQPDZGIKBAWPEJ-UHFFFAOYSA-N 0.000 description 3
- 150000003722 vitamin derivatives Chemical class 0.000 description 3
- GVJHHUAWPYXKBD-IEOSBIPESA-N α-tocopherol Chemical compound OC1=C(C)C(C)=C2O[C@@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-IEOSBIPESA-N 0.000 description 3
- RBCOYOYDYNXAFA-UHFFFAOYSA-L (5-hydroxy-4,6-dimethylpyridin-3-yl)methyl phosphate Chemical compound CC1=NC=C(COP([O-])([O-])=O)C(C)=C1O RBCOYOYDYNXAFA-UHFFFAOYSA-L 0.000 description 2
- GHOKWGTUZJEAQD-ZETCQYMHSA-N (D)-(+)-Pantothenic acid Chemical compound OCC(C)(C)[C@@H](O)C(=O)NCCC(O)=O GHOKWGTUZJEAQD-ZETCQYMHSA-N 0.000 description 2
- 229940058015 1,3-butylene glycol Drugs 0.000 description 2
- WNWHHMBRJJOGFJ-UHFFFAOYSA-N 16-methylheptadecan-1-ol Chemical compound CC(C)CCCCCCCCCCCCCCCO WNWHHMBRJJOGFJ-UHFFFAOYSA-N 0.000 description 2
- XDOFQFKRPWOURC-UHFFFAOYSA-N 16-methylheptadecanoic acid Chemical compound CC(C)CCCCCCCCCCCCCCC(O)=O XDOFQFKRPWOURC-UHFFFAOYSA-N 0.000 description 2
- ALYNCZNDIQEVRV-UHFFFAOYSA-N 4-aminobenzoic acid Chemical compound NC1=CC=C(C(O)=O)C=C1 ALYNCZNDIQEVRV-UHFFFAOYSA-N 0.000 description 2
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Natural products OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- LSNNMFCWUKXFEE-UHFFFAOYSA-M Bisulfite Chemical compound OS([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-M 0.000 description 2
- DKPFZGUDAPQIHT-UHFFFAOYSA-N Butyl acetate Natural products CCCCOC(C)=O DKPFZGUDAPQIHT-UHFFFAOYSA-N 0.000 description 2
- QFOHBWFCKVYLES-UHFFFAOYSA-N Butylparaben Chemical compound CCCCOC(=O)C1=CC=C(O)C=C1 QFOHBWFCKVYLES-UHFFFAOYSA-N 0.000 description 2
- FERIUCNNQQJTOY-UHFFFAOYSA-M Butyrate Chemical compound CCCC([O-])=O FERIUCNNQQJTOY-UHFFFAOYSA-M 0.000 description 2
- FERIUCNNQQJTOY-UHFFFAOYSA-N Butyric acid Chemical compound CCCC(O)=O FERIUCNNQQJTOY-UHFFFAOYSA-N 0.000 description 2
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 2
- KXDHJXZQYSOELW-UHFFFAOYSA-N Carbamic acid Chemical class NC(O)=O KXDHJXZQYSOELW-UHFFFAOYSA-N 0.000 description 2
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 2
- 206010014970 Ephelides Diseases 0.000 description 2
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 2
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 2
- UQSXHKLRYXJYBZ-UHFFFAOYSA-N Iron oxide Chemical compound [Fe]=O UQSXHKLRYXJYBZ-UHFFFAOYSA-N 0.000 description 2
- XUMBMVFBXHLACL-UHFFFAOYSA-N Melanin Chemical compound O=C1C(=O)C(C2=CNC3=C(C(C(=O)C4=C32)=O)C)=C2C4=CNC2=C1C XUMBMVFBXHLACL-UHFFFAOYSA-N 0.000 description 2
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 2
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 description 2
- DFPAKSUCGFBDDF-UHFFFAOYSA-N Nicotinamide Chemical compound NC(=O)C1=CC=CN=C1 DFPAKSUCGFBDDF-UHFFFAOYSA-N 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- 239000002202 Polyethylene glycol Substances 0.000 description 2
- 229920001214 Polysorbate 60 Polymers 0.000 description 2
- ROJKPKOYARNFNB-UHFFFAOYSA-N Propyl pentanoate Chemical compound CCCCC(=O)OCCC ROJKPKOYARNFNB-UHFFFAOYSA-N 0.000 description 2
- HVUMOYIDDBPOLL-XWVZOOPGSA-N Sorbitan monostearate Chemical compound CCCCCCCCCCCCCCCCCC(=O)OC[C@@H](O)[C@H]1OC[C@H](O)[C@H]1O HVUMOYIDDBPOLL-XWVZOOPGSA-N 0.000 description 2
- 206010042496 Sunburn Diseases 0.000 description 2
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 2
- 230000002159 abnormal effect Effects 0.000 description 2
- 150000001298 alcohols Chemical class 0.000 description 2
- 229910052783 alkali metal Inorganic materials 0.000 description 2
- 150000005215 alkyl ethers Chemical class 0.000 description 2
- 239000002280 amphoteric surfactant Substances 0.000 description 2
- RWZYAGGXGHYGMB-UHFFFAOYSA-N anthranilic acid Chemical compound NC1=CC=CC=C1C(O)=O RWZYAGGXGHYGMB-UHFFFAOYSA-N 0.000 description 2
- 229940121363 anti-inflammatory agent Drugs 0.000 description 2
- 239000002260 anti-inflammatory agent Substances 0.000 description 2
- 235000010323 ascorbic acid Nutrition 0.000 description 2
- 239000011668 ascorbic acid Substances 0.000 description 2
- 229960005070 ascorbic acid Drugs 0.000 description 2
- 125000003236 benzoyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C(*)=O 0.000 description 2
- 235000019437 butane-1,3-diol Nutrition 0.000 description 2
- 150000007942 carboxylates Chemical class 0.000 description 2
- 238000007796 conventional method Methods 0.000 description 2
- 125000004122 cyclic group Chemical group 0.000 description 2
- 238000000151 deposition Methods 0.000 description 2
- 238000013461 design Methods 0.000 description 2
- XBDQKXXYIPTUBI-UHFFFAOYSA-N dimethylselenoniopropionate Natural products CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 2
- NOPFSRXAKWQILS-UHFFFAOYSA-N docosan-1-ol Chemical compound CCCCCCCCCCCCCCCCCCCCCCO NOPFSRXAKWQILS-UHFFFAOYSA-N 0.000 description 2
- POULHZVOKOAJMA-UHFFFAOYSA-N dodecanoic acid Chemical compound CCCCCCCCCCCC(O)=O POULHZVOKOAJMA-UHFFFAOYSA-N 0.000 description 2
- 125000004185 ester group Chemical group 0.000 description 2
- 150000002170 ethers Chemical class 0.000 description 2
- 150000004665 fatty acids Chemical class 0.000 description 2
- 230000014509 gene expression Effects 0.000 description 2
- KWIUHFFTVRNATP-UHFFFAOYSA-N glycine betaine Chemical compound C[N+](C)(C)CC([O-])=O KWIUHFFTVRNATP-UHFFFAOYSA-N 0.000 description 2
- BXWNKGSJHAJOGX-UHFFFAOYSA-N hexadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCO BXWNKGSJHAJOGX-UHFFFAOYSA-N 0.000 description 2
- IPCSVZSSVZVIGE-UHFFFAOYSA-N hexadecanoic acid Chemical compound CCCCCCCCCCCCCCCC(O)=O IPCSVZSSVZVIGE-UHFFFAOYSA-N 0.000 description 2
- 239000004615 ingredient Substances 0.000 description 2
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- 210000002510 keratinocyte Anatomy 0.000 description 2
- TYQCGQRIZGCHNB-JLAZNSOCSA-N l-ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(O)=C(O)C1=O TYQCGQRIZGCHNB-JLAZNSOCSA-N 0.000 description 2
- 239000004310 lactic acid Substances 0.000 description 2
- 235000014655 lactic acid Nutrition 0.000 description 2
- 239000006210 lotion Substances 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- 230000004060 metabolic process Effects 0.000 description 2
- 229940017219 methyl propionate Drugs 0.000 description 2
- LXCFILQKKLGQFO-UHFFFAOYSA-N methylparaben Chemical compound COC(=O)C1=CC=C(O)C=C1 LXCFILQKKLGQFO-UHFFFAOYSA-N 0.000 description 2
- 239000010445 mica Substances 0.000 description 2
- 229910052618 mica group Inorganic materials 0.000 description 2
- 150000007522 mineralic acids Chemical class 0.000 description 2
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 2
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- 235000001968 nicotinic acid Nutrition 0.000 description 2
- 229960003512 nicotinic acid Drugs 0.000 description 2
- 239000011664 nicotinic acid Substances 0.000 description 2
- PGMYKACGEOXYJE-UHFFFAOYSA-N pentyl acetate Chemical compound CCCCCOC(C)=O PGMYKACGEOXYJE-UHFFFAOYSA-N 0.000 description 2
- 229920001223 polyethylene glycol Polymers 0.000 description 2
- 238000002360 preparation method Methods 0.000 description 2
- 230000002265 prevention Effects 0.000 description 2
- HUAZGNHGCJGYNP-UHFFFAOYSA-N propyl butyrate Chemical compound CCCOC(=O)CCC HUAZGNHGCJGYNP-UHFFFAOYSA-N 0.000 description 2
- 238000000746 purification Methods 0.000 description 2
- 229940109850 royal jelly Drugs 0.000 description 2
- YGSDEFSMJLZEOE-UHFFFAOYSA-N salicylic acid Chemical compound OC(=O)C1=CC=CC=C1O YGSDEFSMJLZEOE-UHFFFAOYSA-N 0.000 description 2
- 159000000000 sodium salts Chemical class 0.000 description 2
- 239000001587 sorbitan monostearate Substances 0.000 description 2
- 235000011076 sorbitan monostearate Nutrition 0.000 description 2
- 229940035048 sorbitan monostearate Drugs 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- HLZKNKRTKFSKGZ-UHFFFAOYSA-N tetradecan-1-ol Chemical compound CCCCCCCCCCCCCCO HLZKNKRTKFSKGZ-UHFFFAOYSA-N 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- 238000011282 treatment Methods 0.000 description 2
- 229940096998 ursolic acid Drugs 0.000 description 2
- PLSAJKYPRJGMHO-UHFFFAOYSA-N ursolic acid Natural products CC1CCC2(CCC3(C)C(C=CC4C5(C)CCC(O)C(C)(C)C5CCC34C)C2C1C)C(=O)O PLSAJKYPRJGMHO-UHFFFAOYSA-N 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 2
- 239000001993 wax Substances 0.000 description 2
- 239000011787 zinc oxide Substances 0.000 description 2
- WGVKWNUPNGFDFJ-DQCZWYHMSA-N β-tocopherol Chemical compound OC1=CC(C)=C2O[C@@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1C WGVKWNUPNGFDFJ-DQCZWYHMSA-N 0.000 description 2
- YBJHBAHKTGYVGT-ZKWXMUAHSA-N (+)-Biotin Chemical compound N1C(=O)N[C@@H]2[C@H](CCCCC(=O)O)SC[C@@H]21 YBJHBAHKTGYVGT-ZKWXMUAHSA-N 0.000 description 1
- KIUKXJAPPMFGSW-DNGZLQJQSA-N (2S,3S,4S,5R,6R)-6-[(2S,3R,4R,5S,6R)-3-Acetamido-2-[(2S,3S,4R,5R,6R)-6-[(2R,3R,4R,5S,6R)-3-acetamido-2,5-dihydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-2-carboxy-4,5-dihydroxyoxan-3-yl]oxy-5-hydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-3,4,5-trihydroxyoxane-2-carboxylic acid Chemical compound CC(=O)N[C@H]1[C@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@H](O[C@H]2[C@@H]([C@@H](O[C@H]3[C@@H]([C@@H](O)[C@H](O)[C@H](O3)C(O)=O)O)[C@H](O)[C@@H](CO)O2)NC(C)=O)[C@@H](C(O)=O)O1 KIUKXJAPPMFGSW-DNGZLQJQSA-N 0.000 description 1
- CUNWUEBNSZSNRX-RKGWDQTMSA-N (2r,3r,4r,5s)-hexane-1,2,3,4,5,6-hexol;(z)-octadec-9-enoic acid Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO.OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO.CCCCCCCC\C=C/CCCCCCCC(O)=O.CCCCCCCC\C=C/CCCCCCCC(O)=O.CCCCCCCC\C=C/CCCCCCCC(O)=O CUNWUEBNSZSNRX-RKGWDQTMSA-N 0.000 description 1
- BJDAUCLANVMIOB-UHFFFAOYSA-N (3-decanoyloxy-2,2-dimethylpropyl) decanoate Chemical compound CCCCCCCCCC(=O)OCC(C)(C)COC(=O)CCCCCCCCC BJDAUCLANVMIOB-UHFFFAOYSA-N 0.000 description 1
- WCGUUGGRBIKTOS-GPOJBZKASA-N (3beta)-3-hydroxyurs-12-en-28-oic acid Chemical compound C1C[C@H](O)C(C)(C)[C@@H]2CC[C@@]3(C)[C@]4(C)CC[C@@]5(C(O)=O)CC[C@@H](C)[C@H](C)[C@H]5C4=CC[C@@H]3[C@]21C WCGUUGGRBIKTOS-GPOJBZKASA-N 0.000 description 1
- WRIDQFICGBMAFQ-UHFFFAOYSA-N (E)-8-Octadecenoic acid Natural products CCCCCCCCCC=CCCCCCCC(O)=O WRIDQFICGBMAFQ-UHFFFAOYSA-N 0.000 description 1
- PSBDWGZCVUAZQS-UHFFFAOYSA-N (dimethylsulfonio)acetate Chemical compound C[S+](C)CC([O-])=O PSBDWGZCVUAZQS-UHFFFAOYSA-N 0.000 description 1
- WBYWAXJHAXSJNI-VOTSOKGWSA-M .beta-Phenylacrylic acid Natural products [O-]C(=O)\C=C\C1=CC=CC=C1 WBYWAXJHAXSJNI-VOTSOKGWSA-M 0.000 description 1
- ZORQXIQZAOLNGE-UHFFFAOYSA-N 1,1-difluorocyclohexane Chemical compound FC1(F)CCCCC1 ZORQXIQZAOLNGE-UHFFFAOYSA-N 0.000 description 1
- 229940015975 1,2-hexanediol Drugs 0.000 description 1
- 229940031723 1,2-octanediol Drugs 0.000 description 1
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 1
- 125000004955 1,4-cyclohexylene group Chemical group [H]C1([H])C([H])([H])C([H])([*:1])C([H])([H])C([H])([H])C1([H])[*:2] 0.000 description 1
- XLTMWFMRJZDFFD-UHFFFAOYSA-N 1-[(2-chloro-4-nitrophenyl)diazenyl]naphthalen-2-ol Chemical compound OC1=CC=C2C=CC=CC2=C1N=NC1=CC=C([N+]([O-])=O)C=C1Cl XLTMWFMRJZDFFD-UHFFFAOYSA-N 0.000 description 1
- HNAGHMKIPMKKBB-UHFFFAOYSA-N 1-benzylpyrrolidine-3-carboxamide Chemical compound C1C(C(=O)N)CCN1CC1=CC=CC=C1 HNAGHMKIPMKKBB-UHFFFAOYSA-N 0.000 description 1
- VBICKXHEKHSIBG-UHFFFAOYSA-N 1-monostearoylglycerol Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCC(O)CO VBICKXHEKHSIBG-UHFFFAOYSA-N 0.000 description 1
- MPDGHEJMBKOTSU-YKLVYJNSSA-N 18beta-glycyrrhetic acid Chemical class C([C@H]1C2=CC(=O)[C@H]34)[C@@](C)(C(O)=O)CC[C@]1(C)CC[C@@]2(C)[C@]4(C)CC[C@@H]1[C@]3(C)CC[C@H](O)C1(C)C MPDGHEJMBKOTSU-YKLVYJNSSA-N 0.000 description 1
- RKJGFHYCZPZJPE-UHFFFAOYSA-N 2,2-bis(16-methylheptadecanoyloxymethyl)butyl 16-methylheptadecanoate Chemical compound CC(C)CCCCCCCCCCCCCCC(=O)OCC(CC)(COC(=O)CCCCCCCCCCCCCCC(C)C)COC(=O)CCCCCCCCCCCCCCC(C)C RKJGFHYCZPZJPE-UHFFFAOYSA-N 0.000 description 1
- XFOQWQKDSMIPHT-UHFFFAOYSA-N 2,3-dichloro-6-(trifluoromethyl)pyridine Chemical compound FC(F)(F)C1=CC=C(Cl)C(Cl)=N1 XFOQWQKDSMIPHT-UHFFFAOYSA-N 0.000 description 1
- UHOPWFKONJYLCF-UHFFFAOYSA-N 2-(2-sulfanylethyl)isoindole-1,3-dione Chemical compound C1=CC=C2C(=O)N(CCS)C(=O)C2=C1 UHOPWFKONJYLCF-UHFFFAOYSA-N 0.000 description 1
- OBETXYAYXDNJHR-UHFFFAOYSA-N 2-Ethylhexanoic acid Chemical compound CCCCC(CC)C(O)=O OBETXYAYXDNJHR-UHFFFAOYSA-N 0.000 description 1
- FKOKUHFZNIUSLW-UHFFFAOYSA-N 2-Hydroxypropyl stearate Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCC(C)O FKOKUHFZNIUSLW-UHFFFAOYSA-N 0.000 description 1
- UDWLULWHUVTZJX-UHFFFAOYSA-N 2-[4-[4-(aminomethyl)cyclohexanecarbonyl]phenyl]acetic acid Chemical compound C1CC(CN)CCC1C(=O)C1=CC=C(CC(O)=O)C=C1 UDWLULWHUVTZJX-UHFFFAOYSA-N 0.000 description 1
- JXDOAAYLPIIEKK-UHFFFAOYSA-N 2-[4-[4-(aminomethyl)cyclohexanecarbonyl]phenyl]propanoic acid Chemical compound C1=CC(C(C(O)=O)C)=CC=C1C(=O)C1CCC(CN)CC1 JXDOAAYLPIIEKK-UHFFFAOYSA-N 0.000 description 1
- FMKGJQHNYMWDFJ-CVEARBPZSA-N 2-[[4-(2,2-difluoropropoxy)pyrimidin-5-yl]methylamino]-4-[[(1R,4S)-4-hydroxy-3,3-dimethylcyclohexyl]amino]pyrimidine-5-carbonitrile Chemical compound FC(COC1=NC=NC=C1CNC1=NC=C(C(=N1)N[C@H]1CC([C@H](CC1)O)(C)C)C#N)(C)F FMKGJQHNYMWDFJ-CVEARBPZSA-N 0.000 description 1
- NCLNAHJFXIKYBY-UHFFFAOYSA-N 2-hexyldecyl 16-methylheptadecanoate Chemical compound CCCCCCCCC(CCCCCC)COC(=O)CCCCCCCCCCCCCCC(C)C NCLNAHJFXIKYBY-UHFFFAOYSA-N 0.000 description 1
- MUHFRORXWCGZGE-KTKRTIGZSA-N 2-hydroxyethyl (z)-octadec-9-enoate Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OCCO MUHFRORXWCGZGE-KTKRTIGZSA-N 0.000 description 1
- QCDWFXQBSFUVSP-UHFFFAOYSA-N 2-phenoxyethanol Chemical compound OCCOC1=CC=CC=C1 QCDWFXQBSFUVSP-UHFFFAOYSA-N 0.000 description 1
- LQJBNNIYVWPHFW-UHFFFAOYSA-N 20:1omega9c fatty acid Natural products CCCCCCCCCCC=CCCCCCCCC(O)=O LQJBNNIYVWPHFW-UHFFFAOYSA-N 0.000 description 1
- ZGIGZINMAOQWLX-NCZFFCEISA-N 3,7,11-Trimethyl-2,6,10-dodecatrienyl acetate Chemical compound CC(C)=CCC\C(C)=C\CC\C(C)=C\COC(C)=O ZGIGZINMAOQWLX-NCZFFCEISA-N 0.000 description 1
- NVBZUCIQNYPGCI-UHFFFAOYSA-N 3-[4-[4-(aminomethyl)cyclohexanecarbonyl]phenyl]propanoic acid Chemical compound C1CC(CN)CCC1C(=O)C1=CC=C(CCC(O)=O)C=C1 NVBZUCIQNYPGCI-UHFFFAOYSA-N 0.000 description 1
- BMYNFMYTOJXKLE-UHFFFAOYSA-N 3-azaniumyl-2-hydroxypropanoate Chemical compound NCC(O)C(O)=O BMYNFMYTOJXKLE-UHFFFAOYSA-N 0.000 description 1
- XPFCZYUVICHKDS-UHFFFAOYSA-N 3-methylbutane-1,3-diol Chemical compound CC(C)(O)CCO XPFCZYUVICHKDS-UHFFFAOYSA-N 0.000 description 1
- CYDQOEWLBCCFJZ-UHFFFAOYSA-N 4-(4-fluorophenyl)oxane-4-carboxylic acid Chemical compound C=1C=C(F)C=CC=1C1(C(=O)O)CCOCC1 CYDQOEWLBCCFJZ-UHFFFAOYSA-N 0.000 description 1
- HBTAOSGHCXUEKI-UHFFFAOYSA-N 4-chloro-n,n-dimethyl-3-nitrobenzenesulfonamide Chemical compound CN(C)S(=O)(=O)C1=CC=C(Cl)C([N+]([O-])=O)=C1 HBTAOSGHCXUEKI-UHFFFAOYSA-N 0.000 description 1
- HIQIXEFWDLTDED-UHFFFAOYSA-N 4-hydroxy-1-piperidin-4-ylpyrrolidin-2-one Chemical compound O=C1CC(O)CN1C1CCNCC1 HIQIXEFWDLTDED-UHFFFAOYSA-N 0.000 description 1
- CSHZYWUPJWVTMQ-UHFFFAOYSA-N 4-n-Butylresorcinol Chemical compound CCCCC1=CC=C(O)C=C1O CSHZYWUPJWVTMQ-UHFFFAOYSA-N 0.000 description 1
- QSBYPNXLFMSGKH-UHFFFAOYSA-N 9-Heptadecensaeure Natural products CCCCCCCC=CCCCCCCCC(O)=O QSBYPNXLFMSGKH-UHFFFAOYSA-N 0.000 description 1
- 244000298715 Actinidia chinensis Species 0.000 description 1
- 235000009434 Actinidia chinensis Nutrition 0.000 description 1
- 235000009436 Actinidia deliciosa Nutrition 0.000 description 1
- 241000251468 Actinopterygii Species 0.000 description 1
- 239000005995 Aluminium silicate Substances 0.000 description 1
- 240000005528 Arctium lappa Species 0.000 description 1
- 239000004475 Arginine Substances 0.000 description 1
- 241001474374 Blennius Species 0.000 description 1
- SGHZXLIDFTYFHQ-UHFFFAOYSA-L Brilliant Blue Chemical compound [Na+].[Na+].C=1C=C(C(=C2C=CC(C=C2)=[N+](CC)CC=2C=C(C=CC=2)S([O-])(=O)=O)C=2C(=CC=CC=2)S([O-])(=O)=O)C=CC=1N(CC)CC1=CC=CC(S([O-])(=O)=O)=C1 SGHZXLIDFTYFHQ-UHFFFAOYSA-L 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- GHOKWGTUZJEAQD-UHFFFAOYSA-N Chick antidermatitis factor Natural products OCC(C)(C)C(O)C(=O)NCCC(O)=O GHOKWGTUZJEAQD-UHFFFAOYSA-N 0.000 description 1
- 206010008570 Chloasma Diseases 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- WBYWAXJHAXSJNI-SREVYHEPSA-N Cinnamic acid Chemical compound OC(=O)\C=C/C1=CC=CC=C1 WBYWAXJHAXSJNI-SREVYHEPSA-N 0.000 description 1
- 235000008733 Citrus aurantifolia Nutrition 0.000 description 1
- 235000005979 Citrus limon Nutrition 0.000 description 1
- 244000131522 Citrus pyriformis Species 0.000 description 1
- 102000008186 Collagen Human genes 0.000 description 1
- 108010035532 Collagen Proteins 0.000 description 1
- 240000008067 Cucumis sativus Species 0.000 description 1
- 235000010799 Cucumis sativus var sativus Nutrition 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- ZAKOWWREFLAJOT-CEFNRUSXSA-N D-alpha-tocopherylacetate Chemical compound CC(=O)OC1=C(C)C(C)=C2O[C@@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1C ZAKOWWREFLAJOT-CEFNRUSXSA-N 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- 244000000626 Daucus carota Species 0.000 description 1
- 235000002767 Daucus carota Nutrition 0.000 description 1
- XMSXQFUHVRWGNA-UHFFFAOYSA-N Decamethylcyclopentasiloxane Chemical compound C[Si]1(C)O[Si](C)(C)O[Si](C)(C)O[Si](C)(C)O[Si](C)(C)O1 XMSXQFUHVRWGNA-UHFFFAOYSA-N 0.000 description 1
- 201000004624 Dermatitis Diseases 0.000 description 1
- QXNVGIXVLWOKEQ-UHFFFAOYSA-N Disodium Chemical class [Na][Na] QXNVGIXVLWOKEQ-UHFFFAOYSA-N 0.000 description 1
- 241000196324 Embryophyta Species 0.000 description 1
- 206010015150 Erythema Diseases 0.000 description 1
- 239000004386 Erythritol Substances 0.000 description 1
- UNXHWFMMPAWVPI-UHFFFAOYSA-N Erythritol Natural products OCC(O)C(O)CO UNXHWFMMPAWVPI-UHFFFAOYSA-N 0.000 description 1
- ICMAFTSLXCXHRK-UHFFFAOYSA-N Ethyl pentanoate Chemical compound CCCCC(=O)OCC ICMAFTSLXCXHRK-UHFFFAOYSA-N 0.000 description 1
- ZGIGZINMAOQWLX-UHFFFAOYSA-N Farnesyl acetate Natural products CC(C)=CCCC(C)=CCCC(C)=CCOC(C)=O ZGIGZINMAOQWLX-UHFFFAOYSA-N 0.000 description 1
- 235000001637 Ganoderma lucidum Nutrition 0.000 description 1
- 240000008397 Ganoderma lucidum Species 0.000 description 1
- 239000006000 Garlic extract Substances 0.000 description 1
- 229940122498 Gene expression inhibitor Drugs 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-L L-tartrate(2-) Chemical compound [O-]C(=O)[C@H](O)[C@@H](O)C([O-])=O FEWJPZIEWOKRBE-JCYAYHJZSA-L 0.000 description 1
- 240000008415 Lactuca sativa Species 0.000 description 1
- 235000003228 Lactuca sativa Nutrition 0.000 description 1
- 239000005639 Lauric acid Substances 0.000 description 1
- 239000002841 Lewis acid Substances 0.000 description 1
- 235000017617 Lonicera japonica Nutrition 0.000 description 1
- 244000167230 Lonicera japonica Species 0.000 description 1
- 235000007688 Lycopersicon esculentum Nutrition 0.000 description 1
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 1
- 239000004472 Lysine Substances 0.000 description 1
- 235000018330 Macadamia integrifolia Nutrition 0.000 description 1
- 235000003800 Macadamia tetraphylla Nutrition 0.000 description 1
- 240000000912 Macadamia tetraphylla Species 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- 102400000740 Melanocyte-stimulating hormone alpha Human genes 0.000 description 1
- 101710200814 Melanotropin alpha Proteins 0.000 description 1
- RJUFJBKOKNCXHH-UHFFFAOYSA-N Methyl propionate Chemical compound CCC(=O)OC RJUFJBKOKNCXHH-UHFFFAOYSA-N 0.000 description 1
- SUZRRICLUFMAQD-UHFFFAOYSA-N N-Methyltaurine Chemical compound CNCCS(O)(=O)=O SUZRRICLUFMAQD-UHFFFAOYSA-N 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- 239000004677 Nylon Substances 0.000 description 1
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 1
- 239000005642 Oleic acid Substances 0.000 description 1
- ZQPPMHVWECSIRJ-UHFFFAOYSA-N Oleic acid Natural products CCCCCCCCC=CCCCCCCCC(O)=O ZQPPMHVWECSIRJ-UHFFFAOYSA-N 0.000 description 1
- 241000596467 Ononis Species 0.000 description 1
- 235000021314 Palmitic acid Nutrition 0.000 description 1
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 102100021904 Potassium-transporting ATPase alpha chain 1 Human genes 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 1
- 108010083204 Proton Pumps Proteins 0.000 description 1
- 235000019484 Rapeseed oil Nutrition 0.000 description 1
- AUNGANRZJHBGPY-SCRDCRAPSA-N Riboflavin Chemical compound OC[C@@H](O)[C@@H](O)[C@@H](O)CN1C=2C=C(C)C(C)=CC=2N=C2C1=NC(=O)NC2=O AUNGANRZJHBGPY-SCRDCRAPSA-N 0.000 description 1
- 235000019485 Safflower oil Nutrition 0.000 description 1
- 206010039897 Sedation Diseases 0.000 description 1
- 241000238371 Sepiidae Species 0.000 description 1
- 206010040880 Skin irritation Diseases 0.000 description 1
- 240000003768 Solanum lycopersicum Species 0.000 description 1
- 235000021355 Stearic acid Nutrition 0.000 description 1
- VBIIFPGSPJYLRR-UHFFFAOYSA-M Stearyltrimethylammonium chloride Chemical compound [Cl-].CCCCCCCCCCCCCCCCCC[N+](C)(C)C VBIIFPGSPJYLRR-UHFFFAOYSA-M 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- 229920002359 Tetronic® Polymers 0.000 description 1
- 244000269722 Thea sinensis Species 0.000 description 1
- 235000011941 Tilia x europaea Nutrition 0.000 description 1
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 1
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 1
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical compound OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 1
- ZJCCRDAZUWHFQH-UHFFFAOYSA-N Trimethylolpropane Chemical compound CCC(CO)(CO)CO ZJCCRDAZUWHFQH-UHFFFAOYSA-N 0.000 description 1
- 101710147108 Tyrosinase inhibitor Proteins 0.000 description 1
- FPIPGXGPPPQFEQ-BOOMUCAASA-N Vitamin A Natural products OC/C=C(/C)\C=C\C=C(\C)/C=C/C1=C(C)CCCC1(C)C FPIPGXGPPPQFEQ-BOOMUCAASA-N 0.000 description 1
- 229930003270 Vitamin B Natural products 0.000 description 1
- 229930003756 Vitamin B7 Natural products 0.000 description 1
- 235000009754 Vitis X bourquina Nutrition 0.000 description 1
- 235000012333 Vitis X labruscana Nutrition 0.000 description 1
- 235000014787 Vitis vinifera Nutrition 0.000 description 1
- 240000006365 Vitis vinifera Species 0.000 description 1
- TVXBFESIOXBWNM-UHFFFAOYSA-N Xylitol Natural products OCCC(O)C(O)C(O)CCO TVXBFESIOXBWNM-UHFFFAOYSA-N 0.000 description 1
- GCNCUMCXAALSTO-UHFFFAOYSA-N [4-(aminomethyl)cyclohexyl]-[4-(3-hydroxy-2-methylpropyl)phenyl]methanone Chemical compound CC(CO)CC1=CC=C(C=C1)C(=O)C1CCC(CC1)CN GCNCUMCXAALSTO-UHFFFAOYSA-N 0.000 description 1
- HTFBWGFLJPQQDS-UHFFFAOYSA-N [4-(aminomethyl)cyclohexyl]-[4-(3-hydroxypropyl)phenyl]methanone Chemical compound C1CC(CN)CCC1C(=O)C1=CC=C(CCCO)C=C1 HTFBWGFLJPQQDS-UHFFFAOYSA-N 0.000 description 1
- RJDOZRNNYVAULJ-UHFFFAOYSA-L [O--].[O--].[O--].[O--].[O--].[O--].[O--].[O--].[O--].[O--].[F-].[F-].[Mg++].[Mg++].[Mg++].[Al+3].[Si+4].[Si+4].[Si+4].[K+] Chemical compound [O--].[O--].[O--].[O--].[O--].[O--].[O--].[O--].[O--].[O--].[F-].[F-].[Mg++].[Mg++].[Mg++].[Al+3].[Si+4].[Si+4].[Si+4].[K+] RJDOZRNNYVAULJ-UHFFFAOYSA-L 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 238000009825 accumulation Methods 0.000 description 1
- KXKVLQRXCPHEJC-UHFFFAOYSA-N acetic acid trimethyl ester Natural products COC(C)=O KXKVLQRXCPHEJC-UHFFFAOYSA-N 0.000 description 1
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 1
- 125000003647 acryloyl group Chemical group O=C([*])C([H])=C([H])[H] 0.000 description 1
- 239000012190 activator Substances 0.000 description 1
- 238000005917 acylation reaction Methods 0.000 description 1
- 150000001338 aliphatic hydrocarbons Chemical class 0.000 description 1
- 150000001340 alkali metals Chemical class 0.000 description 1
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 1
- 229940087168 alpha tocopherol Drugs 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 235000012211 aluminium silicate Nutrition 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- 239000003945 anionic surfactant Substances 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- 230000000767 anti-ulcer Effects 0.000 description 1
- 229940053202 antiepileptics carboxamide derivative Drugs 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 230000003078 antioxidant effect Effects 0.000 description 1
- 235000006708 antioxidants Nutrition 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- 150000004945 aromatic hydrocarbons Chemical class 0.000 description 1
- 235000020739 avocado extract Nutrition 0.000 description 1
- 235000021302 avocado oil Nutrition 0.000 description 1
- 239000008163 avocado oil Substances 0.000 description 1
- IRERQBUNZFJFGC-UHFFFAOYSA-L azure blue Chemical compound [Na+].[Na+].[Na+].[Na+].[Na+].[Na+].[Na+].[Na+].[Al+3].[Al+3].[Al+3].[Al+3].[Al+3].[Al+3].[S-]S[S-].[O-][Si]([O-])([O-])[O-].[O-][Si]([O-])([O-])[O-].[O-][Si]([O-])([O-])[O-].[O-][Si]([O-])([O-])[O-].[O-][Si]([O-])([O-])[O-].[O-][Si]([O-])([O-])[O-] IRERQBUNZFJFGC-UHFFFAOYSA-L 0.000 description 1
- 229910052788 barium Inorganic materials 0.000 description 1
- DSAJWYNOEDNPEQ-UHFFFAOYSA-N barium atom Chemical compound [Ba] DSAJWYNOEDNPEQ-UHFFFAOYSA-N 0.000 description 1
- POJOORKDYOPQLS-UHFFFAOYSA-L barium(2+) 5-chloro-2-[(2-hydroxynaphthalen-1-yl)diazenyl]-4-methylbenzenesulfonate Chemical compound [Ba+2].C1=C(Cl)C(C)=CC(N=NC=2C3=CC=CC=C3C=CC=2O)=C1S([O-])(=O)=O.C1=C(Cl)C(C)=CC(N=NC=2C3=CC=CC=C3C=CC=2O)=C1S([O-])(=O)=O POJOORKDYOPQLS-UHFFFAOYSA-L 0.000 description 1
- 235000013871 bee wax Nutrition 0.000 description 1
- 239000012166 beeswax Substances 0.000 description 1
- SRSXLGNVWSONIS-UHFFFAOYSA-N benzenesulfonic acid Chemical compound OS(=O)(=O)C1=CC=CC=C1 SRSXLGNVWSONIS-UHFFFAOYSA-N 0.000 description 1
- 229940092714 benzenesulfonic acid Drugs 0.000 description 1
- RWCCWEUUXYIKHB-UHFFFAOYSA-N benzophenone Chemical compound C=1C=CC=CC=1C(=O)C1=CC=CC=C1 RWCCWEUUXYIKHB-UHFFFAOYSA-N 0.000 description 1
- 239000012965 benzophenone Substances 0.000 description 1
- 229940066595 beta tocopherol Drugs 0.000 description 1
- 229960003237 betaine Drugs 0.000 description 1
- HGKOWIQVWAQWDS-UHFFFAOYSA-N bis(16-methylheptadecyl) 2-hydroxybutanedioate Chemical compound CC(C)CCCCCCCCCCCCCCCOC(=O)CC(O)C(=O)OCCCCCCCCCCCCCCCC(C)C HGKOWIQVWAQWDS-UHFFFAOYSA-N 0.000 description 1
- 229940073609 bismuth oxychloride Drugs 0.000 description 1
- 238000009835 boiling Methods 0.000 description 1
- DBZJJPROPLPMSN-UHFFFAOYSA-N bromoeosin Chemical compound O1C(=O)C2=CC=CC=C2C21C1=CC(Br)=C(O)C(Br)=C1OC1=C(Br)C(O)=C(Br)C=C21 DBZJJPROPLPMSN-UHFFFAOYSA-N 0.000 description 1
- OBNCKNCVKJNDBV-UHFFFAOYSA-N butanoic acid ethyl ester Natural products CCCC(=O)OCC OBNCKNCVKJNDBV-UHFFFAOYSA-N 0.000 description 1
- 229940067596 butylparaben Drugs 0.000 description 1
- 125000004063 butyryl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- 239000004204 candelilla wax Substances 0.000 description 1
- 235000013868 candelilla wax Nutrition 0.000 description 1
- 229940073532 candelilla wax Drugs 0.000 description 1
- SHZIWNPUGXLXDT-UHFFFAOYSA-N caproic acid ethyl ester Natural products CCCCCC(=O)OCC SHZIWNPUGXLXDT-UHFFFAOYSA-N 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 239000004203 carnauba wax Substances 0.000 description 1
- 235000013869 carnauba wax Nutrition 0.000 description 1
- 229940008396 carrot extract Drugs 0.000 description 1
- 239000003093 cationic surfactant Substances 0.000 description 1
- 229940082500 cetostearyl alcohol Drugs 0.000 description 1
- 229960000541 cetyl alcohol Drugs 0.000 description 1
- 150000001841 cholesterols Chemical class 0.000 description 1
- 229930016911 cinnamic acid Natural products 0.000 description 1
- 235000013985 cinnamic acid Nutrition 0.000 description 1
- 229910000428 cobalt oxide Inorganic materials 0.000 description 1
- FDJOLVPMNUYSCM-UVKKECPRSA-L cobalt(3+);[(2r,3s,4r,5s)-5-(5,6-dimethylbenzimidazol-1-yl)-4-hydroxy-2-(hydroxymethyl)oxolan-3-yl] [(2r)-1-[3-[(2r,3r,4z,7s,9z,12s,13s,14z,17s,18s,19r)-2,13,18-tris(2-amino-2-oxoethyl)-7,12,17-tris(3-amino-3-oxopropyl)-3,5,8,8,13,15,18,19-octamethyl-2,7, Chemical compound [Co+3].N#[C-].C1([C@H](CC(N)=O)[C@@]2(C)CCC(=O)NC[C@@H](C)OP([O-])(=O)O[C@H]3[C@H]([C@H](O[C@@H]3CO)N3C4=CC(C)=C(C)C=C4N=C3)O)[N-]\C2=C(C)/C([C@H](C\2(C)C)CCC(N)=O)=N/C/2=C\C([C@H]([C@@]/2(CC(N)=O)C)CCC(N)=O)=N\C\2=C(C)/C2=N[C@]1(C)[C@@](C)(CC(N)=O)[C@@H]2CCC(N)=O FDJOLVPMNUYSCM-UVKKECPRSA-L 0.000 description 1
- IVMYJDGYRUAWML-UHFFFAOYSA-N cobalt(ii) oxide Chemical compound [Co]=O IVMYJDGYRUAWML-UHFFFAOYSA-N 0.000 description 1
- 229920001436 collagen Polymers 0.000 description 1
- 229940125904 compound 1 Drugs 0.000 description 1
- 229940125782 compound 2 Drugs 0.000 description 1
- 229940126214 compound 3 Drugs 0.000 description 1
- 229940125898 compound 5 Drugs 0.000 description 1
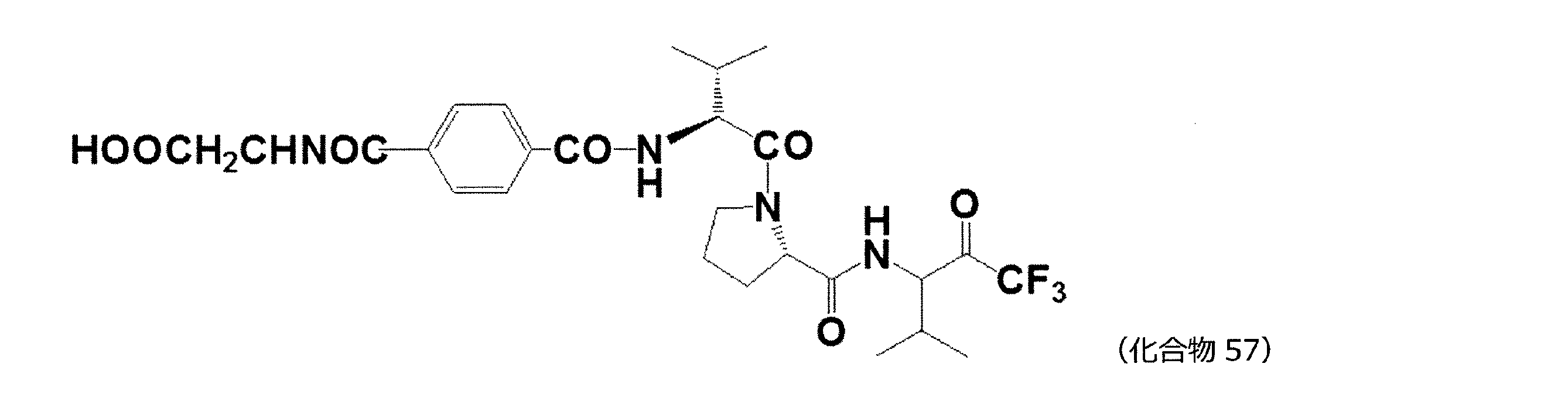
- 229940127113 compound 57 Drugs 0.000 description 1
- 235000005687 corn oil Nutrition 0.000 description 1
- 239000002285 corn oil Substances 0.000 description 1
- 235000012343 cottonseed oil Nutrition 0.000 description 1
- 239000002385 cottonseed oil Substances 0.000 description 1
- 239000006071 cream Substances 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- ZAKOWWREFLAJOT-UHFFFAOYSA-N d-alpha-Tocopheryl acetate Natural products CC(=O)OC1=C(C)C(C)=C2OC(CCCC(C)CCCC(C)CCCC(C)C)(C)CCC2=C1C ZAKOWWREFLAJOT-UHFFFAOYSA-N 0.000 description 1
- GVJHHUAWPYXKBD-UHFFFAOYSA-N d-alpha-tocopherol Natural products OC1=C(C)C(C)=C2OC(CCCC(C)CCCC(C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-UHFFFAOYSA-N 0.000 description 1
- 238000004042 decolorization Methods 0.000 description 1
- 238000004332 deodorization Methods 0.000 description 1
- 230000008021 deposition Effects 0.000 description 1
- 239000003599 detergent Substances 0.000 description 1
- MTHSVFCYNBDYFN-UHFFFAOYSA-N diethylene glycol Chemical compound OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 description 1
- 229940105990 diglycerin Drugs 0.000 description 1
- GPLRAVKSCUXZTP-UHFFFAOYSA-N diglycerol Chemical compound OCC(O)COCC(O)CO GPLRAVKSCUXZTP-UHFFFAOYSA-N 0.000 description 1
- 229940031578 diisopropyl adipate Drugs 0.000 description 1
- 239000004205 dimethyl polysiloxane Substances 0.000 description 1
- 235000013870 dimethyl polysiloxane Nutrition 0.000 description 1
- XEYBRNLFEZDVAW-ARSRFYASSA-N dinoprostone Chemical compound CCCCC[C@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1C\C=C/CCCC(O)=O XEYBRNLFEZDVAW-ARSRFYASSA-N 0.000 description 1
- 229960002986 dinoprostone Drugs 0.000 description 1
- USIUVYZYUHIAEV-UHFFFAOYSA-N diphenyl ether Chemical compound C=1C=CC=CC=1OC1=CC=CC=C1 USIUVYZYUHIAEV-UHFFFAOYSA-N 0.000 description 1
- POLCUAVZOMRGSN-UHFFFAOYSA-N dipropyl ether Chemical compound CCCOCCC POLCUAVZOMRGSN-UHFFFAOYSA-N 0.000 description 1
- SZXQTJUDPRGNJN-UHFFFAOYSA-N dipropylene glycol Chemical compound OCCCOCCCO SZXQTJUDPRGNJN-UHFFFAOYSA-N 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 1
- FHRUGNCCGSEPPE-UHFFFAOYSA-L disodium;2-(4,5-dibromo-3,6-dioxido-9h-xanthen-9-yl)benzoate;hydron Chemical compound [H+].[Na+].[Na+].[O-]C(=O)C1=CC=CC=C1C1C2=CC=C([O-])C(Br)=C2OC2=C(Br)C([O-])=CC=C21 FHRUGNCCGSEPPE-UHFFFAOYSA-L 0.000 description 1
- FPAYXBWMYIMERV-UHFFFAOYSA-L disodium;5-methyl-2-[[4-(4-methyl-2-sulfonatoanilino)-9,10-dioxoanthracen-1-yl]amino]benzenesulfonate Chemical compound [Na+].[Na+].[O-]S(=O)(=O)C1=CC(C)=CC=C1NC(C=1C(=O)C2=CC=CC=C2C(=O)C=11)=CC=C1NC1=CC=C(C)C=C1S([O-])(=O)=O FPAYXBWMYIMERV-UHFFFAOYSA-L 0.000 description 1
- 229960000735 docosanol Drugs 0.000 description 1
- SYELZBGXAIXKHU-UHFFFAOYSA-N dodecyldimethylamine N-oxide Chemical compound CCCCCCCCCCCC[N+](C)(C)[O-] SYELZBGXAIXKHU-UHFFFAOYSA-N 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 229920001971 elastomer Polymers 0.000 description 1
- 239000000806 elastomer Substances 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 231100000321 erythema Toxicity 0.000 description 1
- UNXHWFMMPAWVPI-ZXZARUISSA-N erythritol Chemical compound OC[C@H](O)[C@H](O)CO UNXHWFMMPAWVPI-ZXZARUISSA-N 0.000 description 1
- 229940009714 erythritol Drugs 0.000 description 1
- 235000019414 erythritol Nutrition 0.000 description 1
- 239000010696 ester oil Substances 0.000 description 1
- CCIVGXIOQKPBKL-UHFFFAOYSA-M ethanesulfonate Chemical compound CCS([O-])(=O)=O CCIVGXIOQKPBKL-UHFFFAOYSA-M 0.000 description 1
- TUKWPCXMNZAXLO-UHFFFAOYSA-N ethyl 2-nonylsulfanyl-4-oxo-1h-pyrimidine-6-carboxylate Chemical compound CCCCCCCCCSC1=NC(=O)C=C(C(=O)OCC)N1 TUKWPCXMNZAXLO-UHFFFAOYSA-N 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 229960001617 ethyl hydroxybenzoate Drugs 0.000 description 1
- 239000004403 ethyl p-hydroxybenzoate Substances 0.000 description 1
- 235000010228 ethyl p-hydroxybenzoate Nutrition 0.000 description 1
- 125000000816 ethylene group Chemical group [H]C([H])([*:1])C([H])([H])[*:2] 0.000 description 1
- NUVBSKCKDOMJSU-UHFFFAOYSA-N ethylparaben Chemical compound CCOC(=O)C1=CC=C(O)C=C1 NUVBSKCKDOMJSU-UHFFFAOYSA-N 0.000 description 1
- 229940007703 farnesyl acetate Drugs 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 239000011888 foil Substances 0.000 description 1
- 230000002431 foraging effect Effects 0.000 description 1
- WBJINCZRORDGAQ-UHFFFAOYSA-N formic acid ethyl ester Natural products CCOC=O WBJINCZRORDGAQ-UHFFFAOYSA-N 0.000 description 1
- 125000002485 formyl group Chemical group [H]C(*)=O 0.000 description 1
- 235000011389 fruit/vegetable juice Nutrition 0.000 description 1
- 239000001530 fumaric acid Substances 0.000 description 1
- 235000010382 gamma-tocopherol Nutrition 0.000 description 1
- WIGCFUFOHFEKBI-UHFFFAOYSA-N gamma-tocopherol Natural products CC(C)CCCC(C)CCCC(C)CCCC1CCC2C(C)C(O)C(C)C(C)C2O1 WIGCFUFOHFEKBI-UHFFFAOYSA-N 0.000 description 1
- 235000020706 garlic extract Nutrition 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 229930182478 glucoside Natural products 0.000 description 1
- 229960005150 glycerol Drugs 0.000 description 1
- LPLVUJXQOOQHMX-QWBHMCJMSA-N glycyrrhizinic acid Chemical class O([C@@H]1[C@@H](O)[C@H](O)[C@H](O[C@@H]1O[C@@H]1C([C@H]2[C@]([C@@H]3[C@@]([C@@]4(CC[C@@]5(C)CC[C@@](C)(C[C@H]5C4=CC3=O)C(O)=O)C)(C)CC2)(C)CC1)(C)C)C(O)=O)[C@@H]1O[C@H](C(O)=O)[C@@H](O)[C@H](O)[C@H]1O LPLVUJXQOOQHMX-QWBHMCJMSA-N 0.000 description 1
- 229940065115 grapefruit extract Drugs 0.000 description 1
- 150000008282 halocarbons Chemical class 0.000 description 1
- IUJAMGNYPWYUPM-UHFFFAOYSA-N hentriacontane Chemical compound CCCCCCCCCCCCCCCCCCCCCCCCCCCCCCC IUJAMGNYPWYUPM-UHFFFAOYSA-N 0.000 description 1
- OIKBVOIOVNEVJR-UHFFFAOYSA-N hexadecyl 6-methylheptanoate Chemical compound CCCCCCCCCCCCCCCCOC(=O)CCCCC(C)C OIKBVOIOVNEVJR-UHFFFAOYSA-N 0.000 description 1
- FHKSXSQHXQEMOK-UHFFFAOYSA-N hexane-1,2-diol Chemical compound CCCCC(O)CO FHKSXSQHXQEMOK-UHFFFAOYSA-N 0.000 description 1
- FUZZWVXGSFPDMH-UHFFFAOYSA-M hexanoate Chemical compound CCCCCC([O-])=O FUZZWVXGSFPDMH-UHFFFAOYSA-M 0.000 description 1
- AOGQPLXWSUTHQB-UHFFFAOYSA-N hexyl acetic acid ester Natural products CCCCCCOC(C)=O AOGQPLXWSUTHQB-UHFFFAOYSA-N 0.000 description 1
- 229920002674 hyaluronan Polymers 0.000 description 1
- 229960003160 hyaluronic acid Drugs 0.000 description 1
- 229930195733 hydrocarbon Natural products 0.000 description 1
- 150000003840 hydrochlorides Chemical class 0.000 description 1
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 1
- 125000004029 hydroxymethyl group Chemical group [H]OC([H])([H])* 0.000 description 1
- VVIUBCNYACGLLV-UHFFFAOYSA-N hypotaurine Chemical compound [NH3+]CCS([O-])=O VVIUBCNYACGLLV-UHFFFAOYSA-N 0.000 description 1
- MTNDZQHUAFNZQY-UHFFFAOYSA-N imidazoline Chemical compound C1CN=CN1 MTNDZQHUAFNZQY-UHFFFAOYSA-N 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 239000001023 inorganic pigment Substances 0.000 description 1
- WTFXARWRTYJXII-UHFFFAOYSA-N iron(2+);iron(3+);oxygen(2-) Chemical compound [O-2].[O-2].[O-2].[O-2].[Fe+2].[Fe+3].[Fe+3] WTFXARWRTYJXII-UHFFFAOYSA-N 0.000 description 1
- LDHBWEYLDHLIBQ-UHFFFAOYSA-M iron(3+);oxygen(2-);hydroxide;hydrate Chemical compound O.[OH-].[O-2].[Fe+3] LDHBWEYLDHLIBQ-UHFFFAOYSA-M 0.000 description 1
- SZVJSHCCFOBDDC-UHFFFAOYSA-N iron(II,III) oxide Inorganic materials O=[Fe]O[Fe]O[Fe]=O SZVJSHCCFOBDDC-UHFFFAOYSA-N 0.000 description 1
- JEIPFZHSYJVQDO-UHFFFAOYSA-N iron(III) oxide Inorganic materials O=[Fe]O[Fe]=O JEIPFZHSYJVQDO-UHFFFAOYSA-N 0.000 description 1
- 230000007794 irritation Effects 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- QXJSBBXBKPUZAA-UHFFFAOYSA-N isooleic acid Natural products CCCCCCCC=CCCCCCCCCC(O)=O QXJSBBXBKPUZAA-UHFFFAOYSA-N 0.000 description 1
- 229940119170 jojoba wax Drugs 0.000 description 1
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 1
- 150000002576 ketones Chemical class 0.000 description 1
- 229960000448 lactic acid Drugs 0.000 description 1
- 150000007517 lewis acids Chemical class 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 229940057995 liquid paraffin Drugs 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- ZLNQQNXFFQJAID-UHFFFAOYSA-L magnesium carbonate Chemical compound [Mg+2].[O-]C([O-])=O ZLNQQNXFFQJAID-UHFFFAOYSA-L 0.000 description 1
- 239000001095 magnesium carbonate Substances 0.000 description 1
- 229910000021 magnesium carbonate Inorganic materials 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000011976 maleic acid Substances 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- HEBKCHPVOIAQTA-UHFFFAOYSA-N meso ribitol Natural products OCC(O)C(O)C(O)CO HEBKCHPVOIAQTA-UHFFFAOYSA-N 0.000 description 1
- 239000002207 metabolite Substances 0.000 description 1
- 229940098779 methanesulfonic acid Drugs 0.000 description 1
- 239000004292 methyl p-hydroxybenzoate Substances 0.000 description 1
- 235000010270 methyl p-hydroxybenzoate Nutrition 0.000 description 1
- WBYWAXJHAXSJNI-UHFFFAOYSA-N methyl p-hydroxycinnamate Natural products OC(=O)C=CC1=CC=CC=C1 WBYWAXJHAXSJNI-UHFFFAOYSA-N 0.000 description 1
- 125000001570 methylene group Chemical group [H]C([H])([*:1])[*:2] 0.000 description 1
- 229960002216 methylparaben Drugs 0.000 description 1
- 239000012046 mixed solvent Substances 0.000 description 1
- 230000003020 moisturizing effect Effects 0.000 description 1
- 229940043348 myristyl alcohol Drugs 0.000 description 1
- WQEPLUUGTLDZJY-UHFFFAOYSA-N n-Pentadecanoic acid Natural products CCCCCCCCCCCCCCC(O)=O WQEPLUUGTLDZJY-UHFFFAOYSA-N 0.000 description 1
- GOQYKNQRPGWPLP-UHFFFAOYSA-N n-heptadecyl alcohol Natural products CCCCCCCCCCCCCCCCCO GOQYKNQRPGWPLP-UHFFFAOYSA-N 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 235000013557 nattō Nutrition 0.000 description 1
- 235000005152 nicotinamide Nutrition 0.000 description 1
- 239000011570 nicotinamide Substances 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- 239000002736 nonionic surfactant Substances 0.000 description 1
- 125000001400 nonyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000010466 nut oil Substances 0.000 description 1
- 229920001778 nylon Polymers 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 1
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 1
- HMMGMWAXVFQUOA-UHFFFAOYSA-N octamethylcyclotetrasiloxane Chemical compound C[Si]1(C)O[Si](C)(C)O[Si](C)(C)O[Si](C)(C)O1 HMMGMWAXVFQUOA-UHFFFAOYSA-N 0.000 description 1
- AEIJTFQOBWATKX-UHFFFAOYSA-N octane-1,2-diol Chemical compound CCCCCCC(O)CO AEIJTFQOBWATKX-UHFFFAOYSA-N 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 description 1
- 239000004006 olive oil Substances 0.000 description 1
- 235000008390 olive oil Nutrition 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 239000012860 organic pigment Substances 0.000 description 1
- 235000006408 oxalic acid Nutrition 0.000 description 1
- TWNQGVIAIRXVLR-UHFFFAOYSA-N oxo(oxoalumanyloxy)alumane Chemical compound O=[Al]O[Al]=O TWNQGVIAIRXVLR-UHFFFAOYSA-N 0.000 description 1
- BWOROQSFKKODDR-UHFFFAOYSA-N oxobismuth;hydrochloride Chemical compound Cl.[Bi]=O BWOROQSFKKODDR-UHFFFAOYSA-N 0.000 description 1
- 229940055726 pantothenic acid Drugs 0.000 description 1
- 235000019161 pantothenic acid Nutrition 0.000 description 1
- 239000011713 pantothenic acid Substances 0.000 description 1
- 150000002948 pantothenic acids Chemical class 0.000 description 1
- FJKROLUGYXJWQN-UHFFFAOYSA-N papa-hydroxy-benzoic acid Natural products OC(=O)C1=CC=C(O)C=C1 FJKROLUGYXJWQN-UHFFFAOYSA-N 0.000 description 1
- WCVRQHFDJLLWFE-UHFFFAOYSA-N pentane-1,2-diol Chemical compound CCCC(O)CO WCVRQHFDJLLWFE-UHFFFAOYSA-N 0.000 description 1
- TWSRVQVEYJNFKQ-UHFFFAOYSA-N pentyl propanoate Chemical compound CCCCCOC(=O)CC TWSRVQVEYJNFKQ-UHFFFAOYSA-N 0.000 description 1
- 229940116257 pepper extract Drugs 0.000 description 1
- 239000000825 pharmaceutical preparation Substances 0.000 description 1
- 229940127557 pharmaceutical product Drugs 0.000 description 1
- 229960005323 phenoxyethanol Drugs 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 239000011574 phosphorus Substances 0.000 description 1
- 239000003504 photosensitizing agent Substances 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- 210000004694 pigment cell Anatomy 0.000 description 1
- 229920001983 poloxamer Polymers 0.000 description 1
- 229920000435 poly(dimethylsiloxane) Polymers 0.000 description 1
- 229920003229 poly(methyl methacrylate) Polymers 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 239000004926 polymethyl methacrylate Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- ONQDVAFWWYYXHM-UHFFFAOYSA-M potassium lauryl sulfate Chemical compound [K+].CCCCCCCCCCCCOS([O-])(=O)=O ONQDVAFWWYYXHM-UHFFFAOYSA-M 0.000 description 1
- 229940116985 potassium lauryl sulfate Drugs 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 235000019260 propionic acid Nutrition 0.000 description 1
- 125000001501 propionyl group Chemical group O=C([*])C([H])([H])C([H])([H])[H] 0.000 description 1
- 229940093625 propylene glycol monostearate Drugs 0.000 description 1
- XEYBRNLFEZDVAW-UHFFFAOYSA-N prostaglandin E2 Natural products CCCCCC(O)C=CC1C(O)CC(=O)C1CC=CCCCC(O)=O XEYBRNLFEZDVAW-UHFFFAOYSA-N 0.000 description 1
- 230000002797 proteolythic effect Effects 0.000 description 1
- ZUFQODAHGAHPFQ-UHFFFAOYSA-N pyridoxine hydrochloride Chemical compound Cl.CC1=NC=C(CO)C(CO)=C1O ZUFQODAHGAHPFQ-UHFFFAOYSA-N 0.000 description 1
- IUVKMZGDUIUOCP-BTNSXGMBSA-N quinbolone Chemical compound O([C@H]1CC[C@H]2[C@H]3[C@@H]([C@]4(C=CC(=O)C=C4CC3)C)CC[C@@]21C)C1=CCCC1 IUVKMZGDUIUOCP-BTNSXGMBSA-N 0.000 description 1
- PYWVYCXTNDRMGF-UHFFFAOYSA-N rhodamine B Chemical compound [Cl-].C=12C=CC(=[N+](CC)CC)C=C2OC2=CC(N(CC)CC)=CC=C2C=1C1=CC=CC=C1C(O)=O PYWVYCXTNDRMGF-UHFFFAOYSA-N 0.000 description 1
- 235000005713 safflower oil Nutrition 0.000 description 1
- 239000003813 safflower oil Substances 0.000 description 1
- 229960004889 salicylic acid Drugs 0.000 description 1
- 229940058287 salicylic acid derivative anticestodals Drugs 0.000 description 1
- 150000003872 salicylic acid derivatives Chemical class 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- UQDJGEHQDNVPGU-UHFFFAOYSA-N serine phosphoethanolamine Chemical compound [NH3+]CCOP([O-])(=O)OCC([NH3+])C([O-])=O UQDJGEHQDNVPGU-UHFFFAOYSA-N 0.000 description 1
- 239000008159 sesame oil Substances 0.000 description 1
- 235000011803 sesame oil Nutrition 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- 229920002545 silicone oil Polymers 0.000 description 1
- 239000000344 soap Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- BTURAGWYSMTVOW-UHFFFAOYSA-M sodium dodecanoate Chemical compound [Na+].CCCCCCCCCCCC([O-])=O BTURAGWYSMTVOW-UHFFFAOYSA-M 0.000 description 1
- 239000001540 sodium lactate Substances 0.000 description 1
- 235000011088 sodium lactate Nutrition 0.000 description 1
- 229940005581 sodium lactate Drugs 0.000 description 1
- 229940082004 sodium laurate Drugs 0.000 description 1
- 229940045870 sodium palmitate Drugs 0.000 description 1
- 229940045920 sodium pyrrolidone carboxylate Drugs 0.000 description 1
- HYRLWUFWDYFEES-UHFFFAOYSA-M sodium;2-oxopyrrolidine-1-carboxylate Chemical compound [Na+].[O-]C(=O)N1CCCC1=O HYRLWUFWDYFEES-UHFFFAOYSA-M 0.000 description 1
- GGXKEBACDBNFAF-UHFFFAOYSA-M sodium;hexadecanoate Chemical compound [Na+].CCCCCCCCCCCCCCCC([O-])=O GGXKEBACDBNFAF-UHFFFAOYSA-M 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000001593 sorbitan monooleate Substances 0.000 description 1
- 235000011069 sorbitan monooleate Nutrition 0.000 description 1
- 229940035049 sorbitan monooleate Drugs 0.000 description 1
- 229960005078 sorbitan sesquioleate Drugs 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 229960002920 sorbitol Drugs 0.000 description 1
- 235000010356 sorbitol Nutrition 0.000 description 1
- 239000008117 stearic acid Substances 0.000 description 1
- 230000001954 sterilising effect Effects 0.000 description 1
- 238000004659 sterilization and disinfection Methods 0.000 description 1
- 150000003431 steroids Chemical class 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- 229940117986 sulfobetaine Drugs 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 230000002194 synthesizing effect Effects 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 229940095064 tartrate Drugs 0.000 description 1
- TUNFSRHWOTWDNC-HKGQFRNVSA-N tetradecanoic acid Chemical compound CCCCCCCCCCCCC[14C](O)=O TUNFSRHWOTWDNC-HKGQFRNVSA-N 0.000 description 1
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 1
- OULAJFUGPPVRBK-UHFFFAOYSA-N tetratriacontyl alcohol Natural products CCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCO OULAJFUGPPVRBK-UHFFFAOYSA-N 0.000 description 1
- SHWIJIJNPFXOFS-UHFFFAOYSA-N thiotaurine Chemical compound NCCS(O)(=O)=S SHWIJIJNPFXOFS-UHFFFAOYSA-N 0.000 description 1
- 239000010936 titanium Substances 0.000 description 1
- 229910052719 titanium Inorganic materials 0.000 description 1
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 description 1
- 229960000984 tocofersolan Drugs 0.000 description 1
- 229930003799 tocopherol Natural products 0.000 description 1
- 235000010384 tocopherol Nutrition 0.000 description 1
- 239000011732 tocopherol Substances 0.000 description 1
- 229960001295 tocopherol Drugs 0.000 description 1
- 229940042585 tocopherol acetate Drugs 0.000 description 1
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 229940118594 trimethylolpropane triisostearate Drugs 0.000 description 1
- 230000007306 turnover Effects 0.000 description 1
- 125000003774 valeryl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 235000019155 vitamin A Nutrition 0.000 description 1
- 239000011719 vitamin A Substances 0.000 description 1
- 235000019156 vitamin B Nutrition 0.000 description 1
- 239000011720 vitamin B Substances 0.000 description 1
- 235000011912 vitamin B7 Nutrition 0.000 description 1
- 239000011735 vitamin B7 Substances 0.000 description 1
- 229940045997 vitamin a Drugs 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- 229920003169 water-soluble polymer Polymers 0.000 description 1
- 230000037303 wrinkles Effects 0.000 description 1
- 239000008096 xylene Substances 0.000 description 1
- 239000000811 xylitol Substances 0.000 description 1
- 229960002675 xylitol Drugs 0.000 description 1
- HEBKCHPVOIAQTA-SCDXWVJYSA-N xylitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)CO HEBKCHPVOIAQTA-SCDXWVJYSA-N 0.000 description 1
- 235000010447 xylitol Nutrition 0.000 description 1
- 239000009538 yokuinin Substances 0.000 description 1
- WHNFPRLDDSXQCL-UAZQEYIDSA-N α-msh Chemical compound C([C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC=1NC=NC=1)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](C(C)C)C(N)=O)NC(=O)[C@H](CO)NC(C)=O)C1=CC=C(O)C=C1 WHNFPRLDDSXQCL-UAZQEYIDSA-N 0.000 description 1
- 235000004835 α-tocopherol Nutrition 0.000 description 1
- 239000002076 α-tocopherol Substances 0.000 description 1
- 235000007680 β-tocopherol Nutrition 0.000 description 1
- 239000011590 β-tocopherol Substances 0.000 description 1
- 239000002478 γ-tocopherol Substances 0.000 description 1
- QUEDXNHFTDJVIY-DQCZWYHMSA-N γ-tocopherol Chemical compound OC1=C(C)C(C)=C2O[C@@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1 QUEDXNHFTDJVIY-DQCZWYHMSA-N 0.000 description 1
Landscapes
- Cosmetics (AREA)
Description
本発明は、優れた美白作用を有する美白用組成物に関する。 The present invention relates to a whitening composition having an excellent whitening effect.
紫外線曝露による日焼けのほか、シミ、そばかす、肝斑、薬剤による皮膚の黒化症等の色素沈着による皮膚症状は、色素細胞(メラノサイト)のメラニン産生が亢進された後、産生されたメラニン色素がターンオーバー異常等によって皮膚に沈着することにより生じることが知られている。
このような色素沈着が関連する皮膚症状は外観から容易に認められるため、顔の印象に大きく影響する。そのため、皮膚の色素沈着の予防や改善に対する関心は、肌を美しく見せたいという願望を持つ人を中心にとても高く、美白剤や美白用化粧料の需要は近年大きいものとなっている。
In addition to sunburn caused by exposure to ultraviolet rays, skin symptoms caused by pigmentation such as spots, freckles, chloasma, and skin blackening caused by drugs are caused by the melanin pigment produced after the melanin production of pigment cells (melanocytes) is enhanced. It is known that it is caused by depositing on the skin due to abnormal turnover or the like.
Such skin symptoms associated with pigmentation are easily observed from the appearance and therefore greatly affect the impression of the face. Therefore, interest in the prevention and improvement of skin pigmentation is very high, especially for those who have a desire to make the skin look beautiful, and the demand for whitening agents and whitening cosmetics has been increasing in recent years.
皮膚の色素沈着の予防又は改善を目的とする美白剤としては、アスコルビン酸やハイドロキノン等が古くから知られており、これらを配合した皮膚外用剤が、皮膚の色素沈着の予防又は改善用に広く使用されてきた。また、近年、色素沈着が生じる作用機序の解明により、メラニン産生抑制剤、チロシナ-ゼ阻害剤、チロシナ-ゼ遺伝子発現抑制剤、α-MSH阻害剤、抗酸化剤、メラノサイトのデンドライド伸長抑制剤、メラノサイトのケラチノサイトへのメラノソ-ム受け渡し阻害剤等の従来の美白剤とは異なる作用機序を有する美白剤の開発も盛んに行われている。
セトラキサート又はその塩も、近年開発された美白剤の有効成分の1つであり、代謝によりトラネキサム酸に分解される化合物である(特許文献1)。トラネキサム酸は、メラノサイトを活性化するプロスタグランジンE2の産生抑制作用と、チロシナーゼ阻害作用とを有し、美白効果を奏することが知られている。セトラキサートはその代謝物であるトラネキサム酸による美白作用に加え、化合物本体の活性による美白作用も有することが報告されている。
Ascorbic acid, hydroquinone, and the like have long been known as whitening agents for the purpose of preventing or improving skin pigmentation, and external skin preparations containing these have been widely used for preventing or improving skin pigmentation. Has been used. In recent years, by elucidating the mechanism of action that causes pigmentation, melanin production inhibitor, tyrosinase inhibitor, tyrosinase gene expression inhibitor, α-MSH inhibitor, antioxidant, melanocyte dendride elongation inhibitor. , A whitening agent having a mechanism of action different from that of conventional whitening agents such as an inhibitor of melanocyte delivery to keratinocytes of melanocytes is being actively developed.
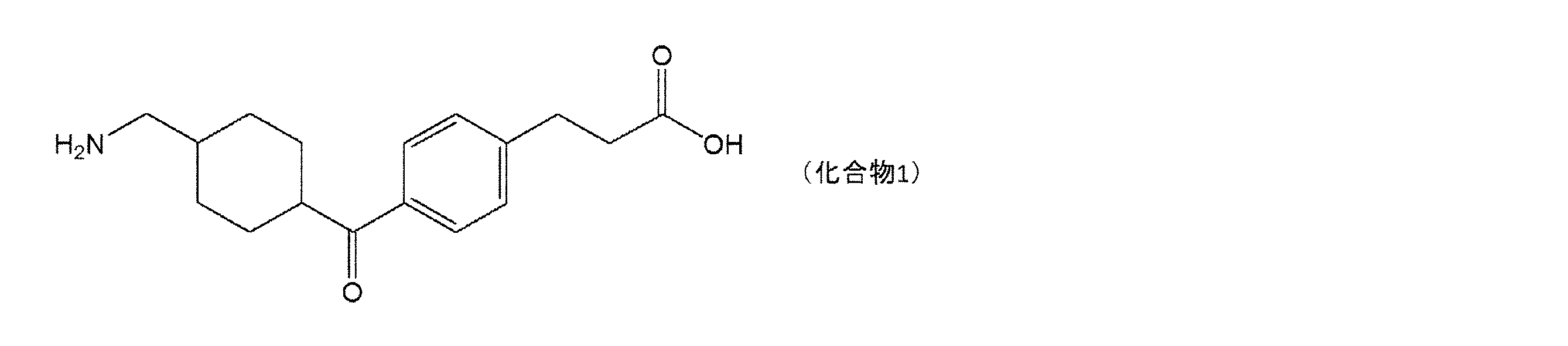
Cetraxate or a salt thereof is also one of the active ingredients of a whitening agent developed in recent years, and is a compound that is decomposed into tranexamic acid by metabolism (Patent Document 1). Tranexamic acid has an action of suppressing the production of prostaglandin E2 that activates melanocytes and an action of inhibiting tyrosinase, and is known to exert a whitening effect. It has been reported that cetraxate has a whitening effect due to the activity of the compound itself in addition to the whitening effect due to its metabolite tranexamic acid.
ところで、特許文献2に記載のアミノカルボン酸誘導体は、抗潰瘍作用があることが確認されており医薬品としての用途が提案されている。なお、該化合物の構造はトラネキサム酸と部分的に一致する。 By the way, the aminocarboxylic acid derivative described in Patent Document 2 has been confirmed to have an anti-ulcer effect, and its use as a pharmaceutical product has been proposed. The structure of the compound is partially consistent with tranexamic acid.
本発明は、優れた美白作用を有する美白用組成物を提供することを課題とする。 An object of the present invention is to provide a whitening composition having an excellent whitening effect.
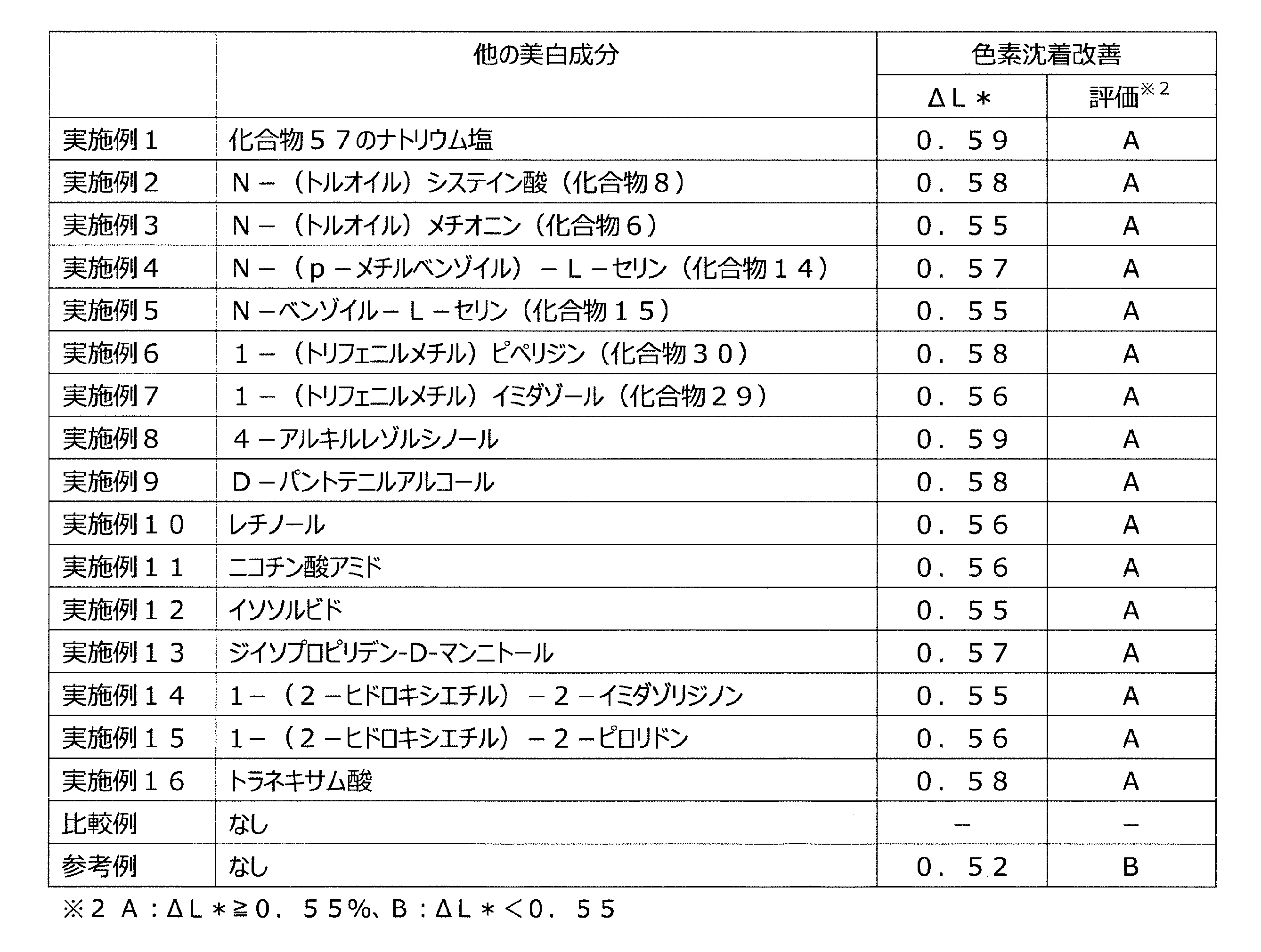
本発明者らは、美白作用を有する化合物を求めて鋭意研究を重ねた結果、特定の構造を有するアミノカルボン酸誘導体及びその酸付加塩が、優れた美白作用を発揮することを見出した。さらに、該化合物と他の美白作用を有する成分とを併用するとより優れた美白効果が得られることに想到し、本発明を完成させるに至った。 As a result of intensive studies for a compound having a whitening effect, the present inventors have found that an aminocarboxylic acid derivative having a specific structure and an acid addition salt thereof exhibit an excellent whitening effect. Furthermore, he came up with the idea that a more excellent whitening effect can be obtained by using the compound in combination with another component having a whitening action, and has completed the present invention.
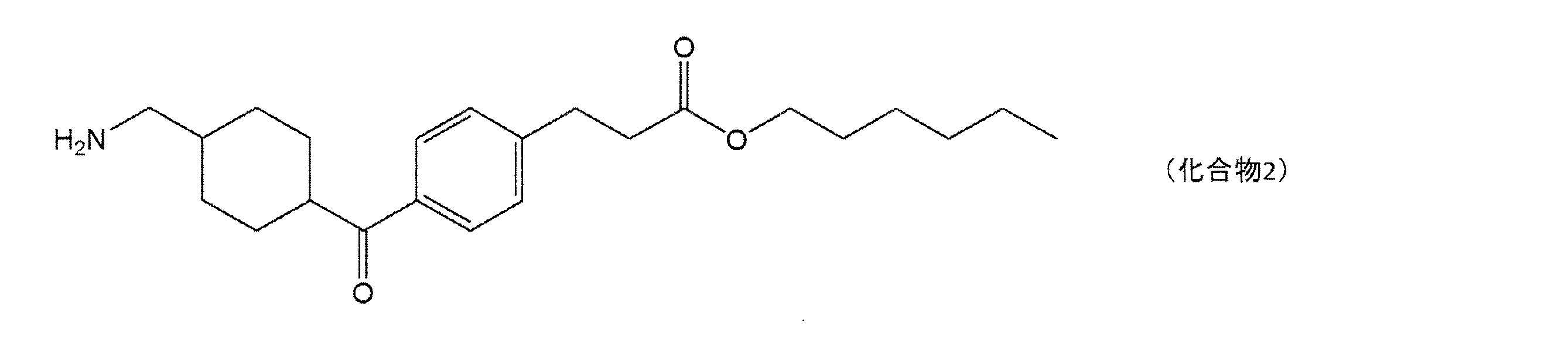
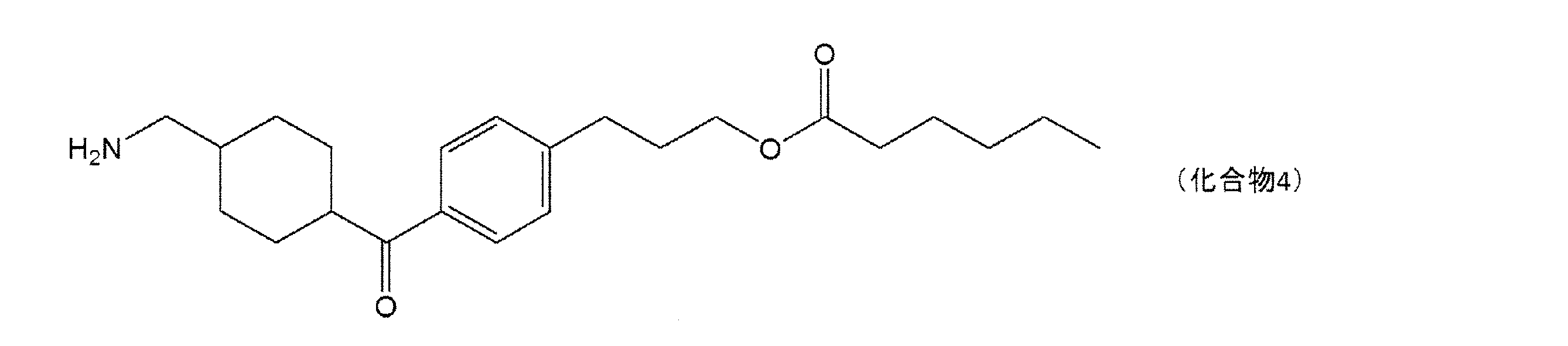
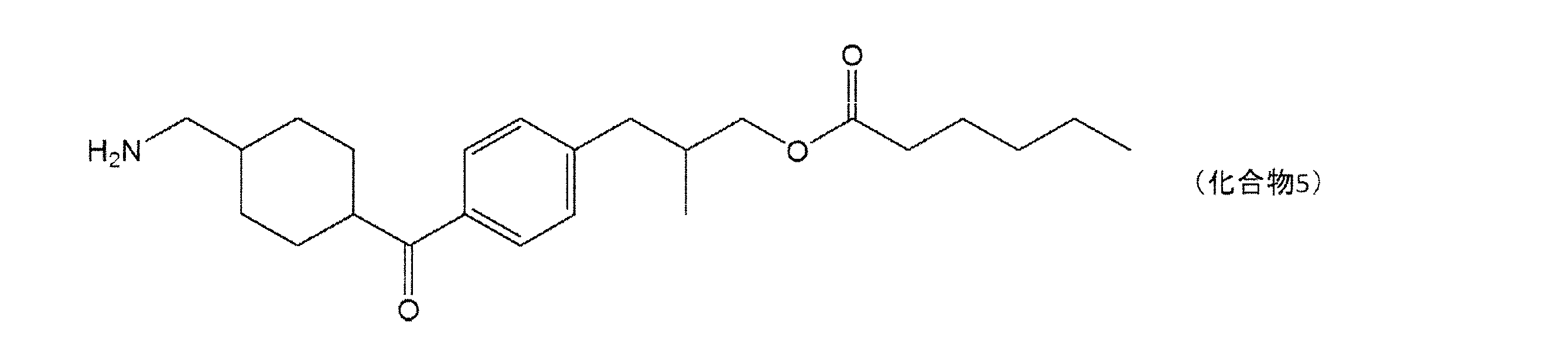
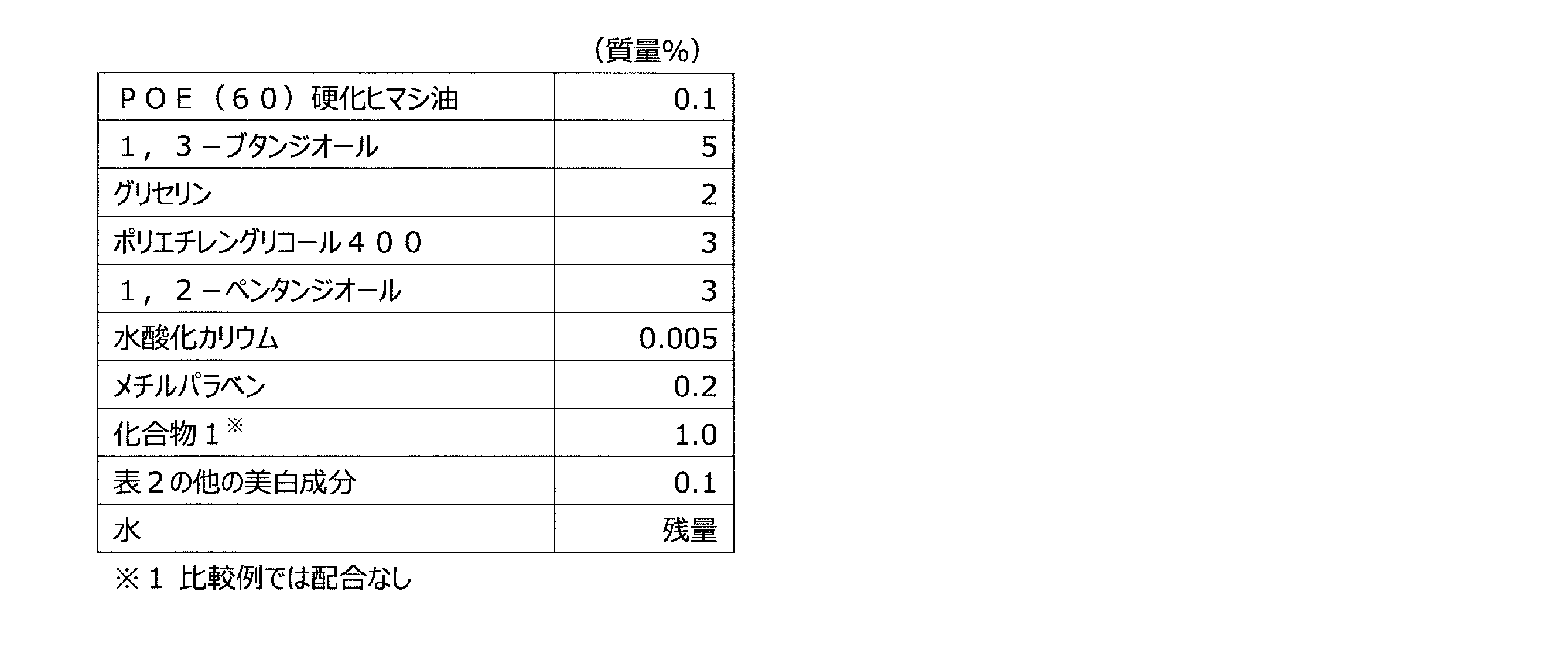
すなわち、本発明は、下記一般式(1)で表される化合物又はその酸付加塩と、他の美
白作用を有する成分とを含有する、美白用組成物である。
That is, the present invention is a whitening composition containing a compound represented by the following general formula (1) or an acid addition salt thereof, and other components having a whitening action.
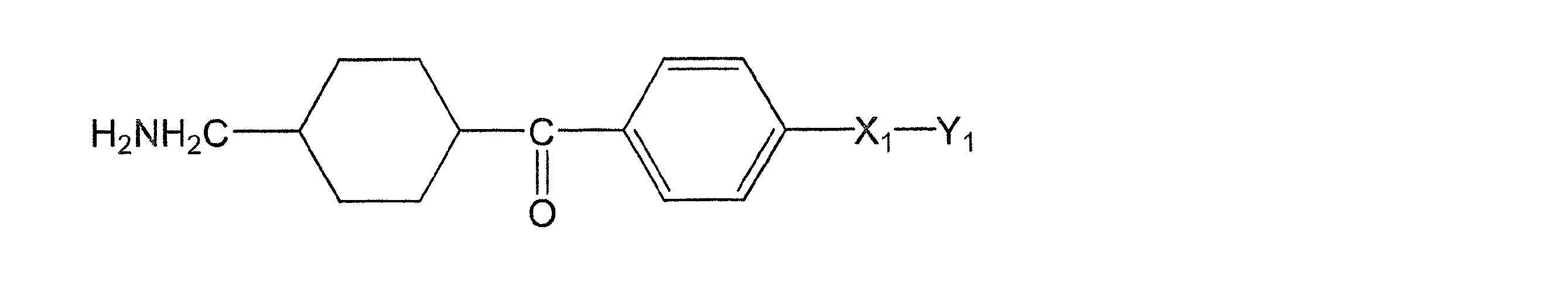
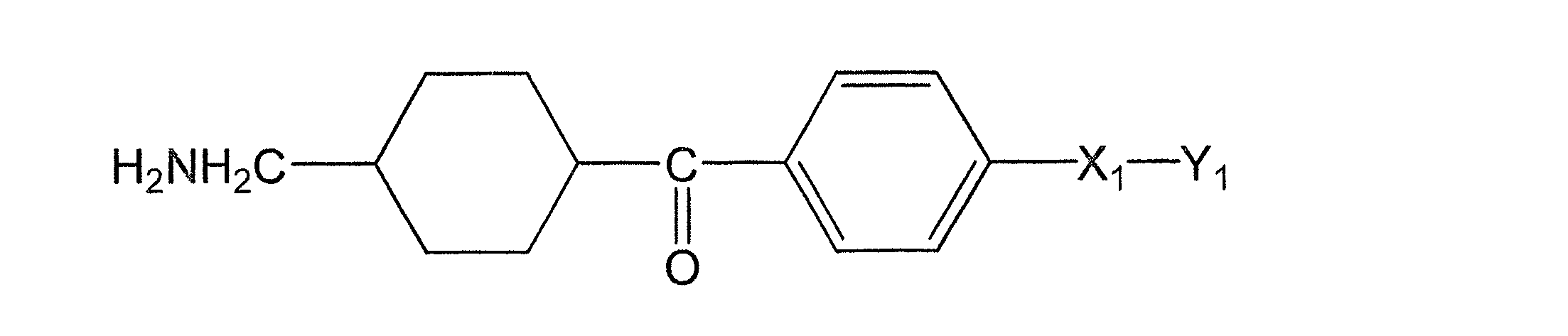
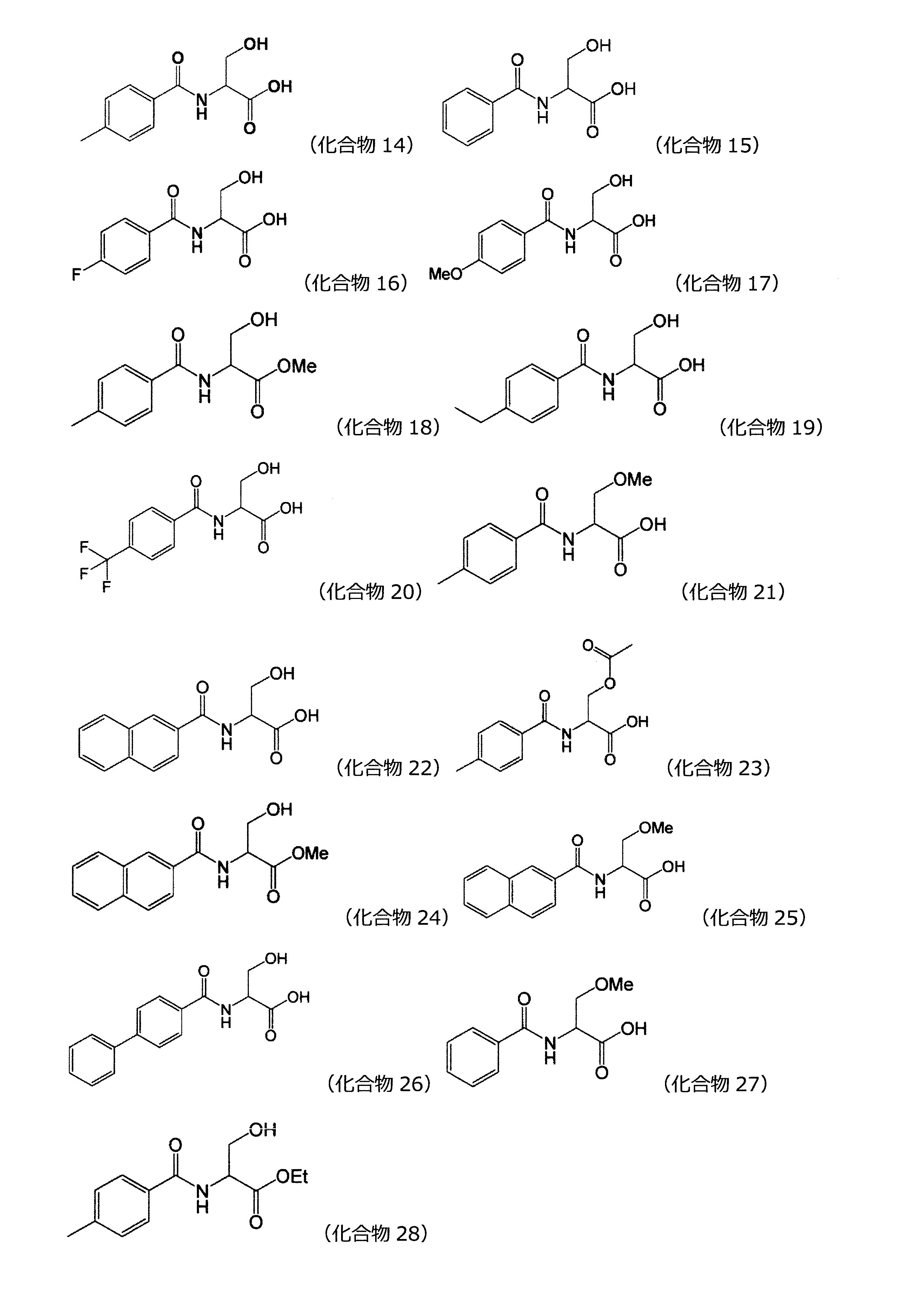
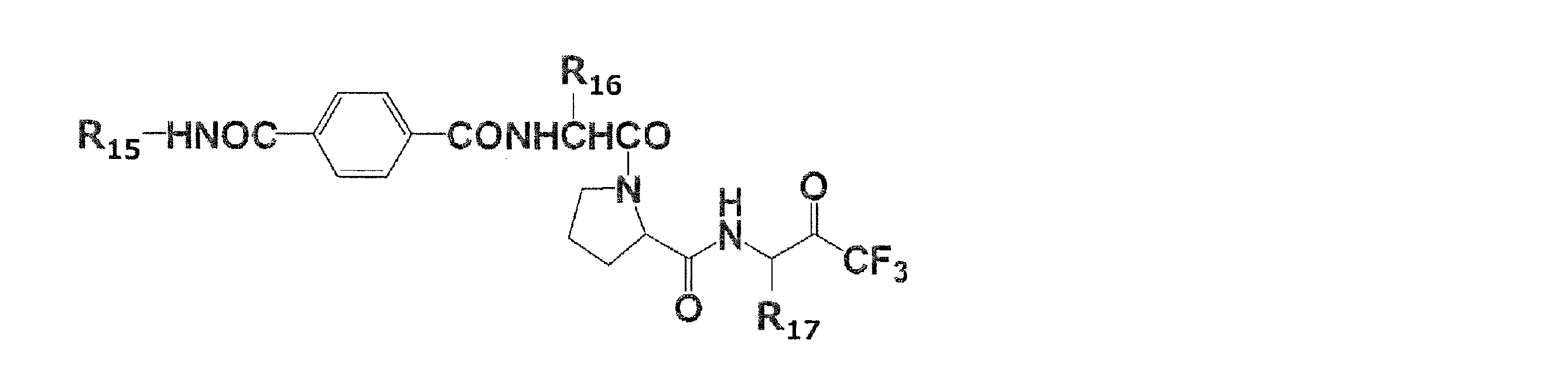
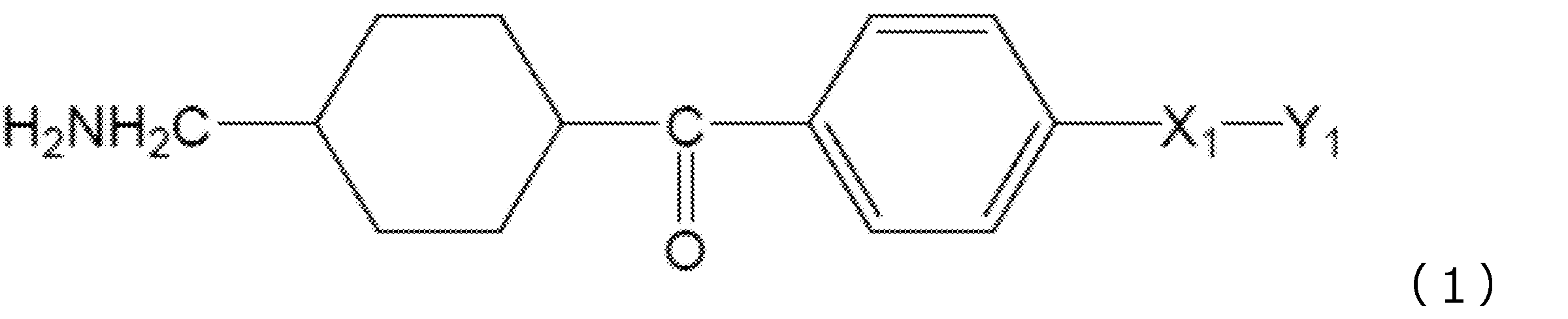
(式(1)中、X1は水素原子がメチル基で置換されていてもよい炭素数1~2のアルキレン基を表し、Y1はCOOR1又はCH2OR2を表し、R1は水素原子又は分岐を有してもよい炭素数1~6のアルキル基を表し、R2は水素原子又は分岐を有してもよい炭素数1~6のアシル基を表す。)
(In the formula (1), X 1 represents an alkylene group having 1 to 2 carbon atoms in which a hydrogen atom may be substituted with a methyl group, Y 1 represents COOR 1 or CH 2 OR 2 , and R 1 represents hydrogen. It represents an alkyl group having 1 to 6 carbon atoms which may have an atom or a branch, and R 2 represents an acyl group having 1 to 6 carbon atoms which may have a hydrogen atom or a branch.)
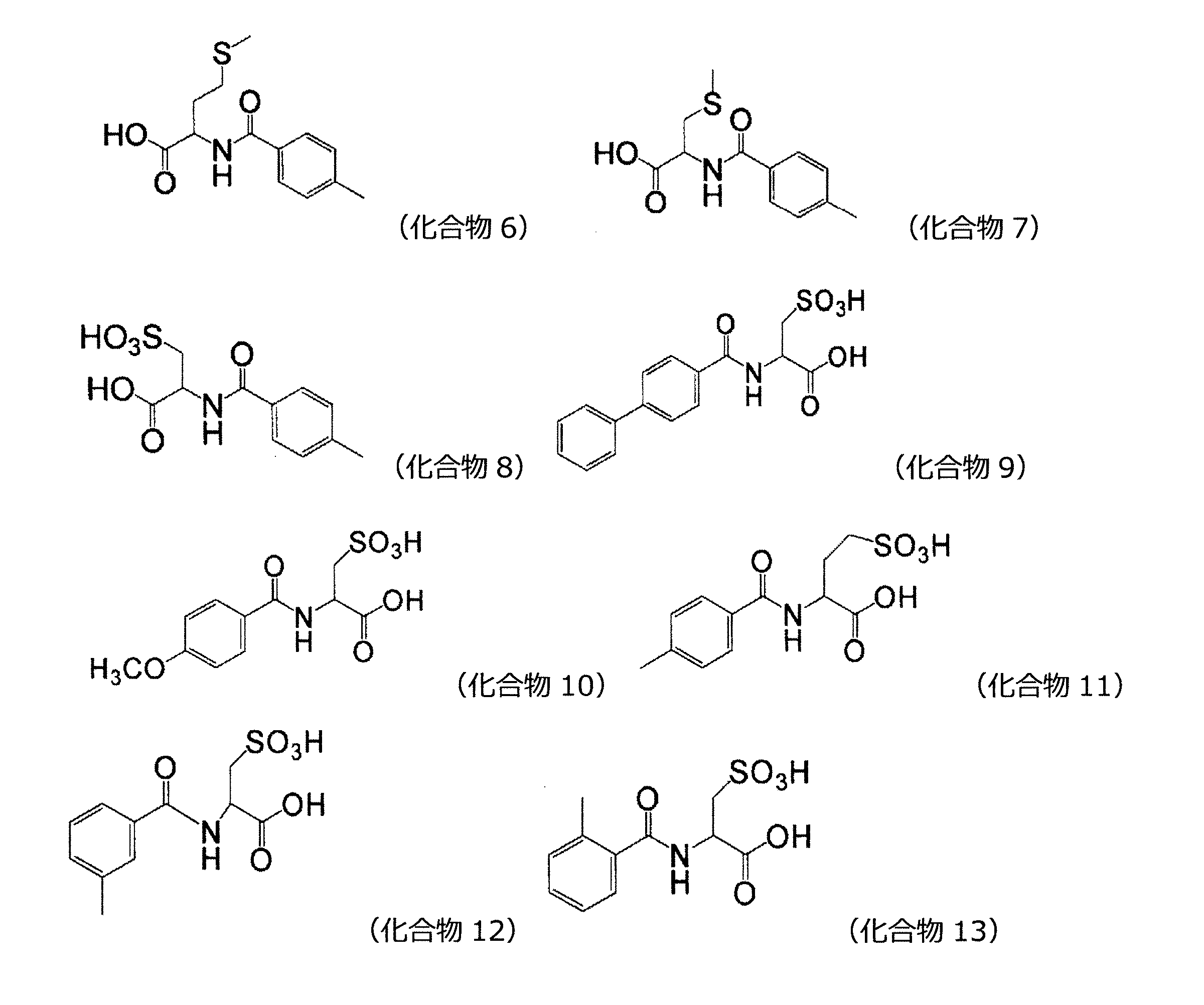
本発明の好ましい態様において、前記他の美白作用を有する成分は、以下の(a)~(l)から選択される1種以上である。
(a)下記一般式(2)で表される化合物又はその塩
(b)下記一般式(3)で表される化合物又はその塩
(c)下記一般式(4)で表される化合物又はその塩
(d)下記一般式(8)で表される化合物又はその塩
(e)アルキルレゾルシノール
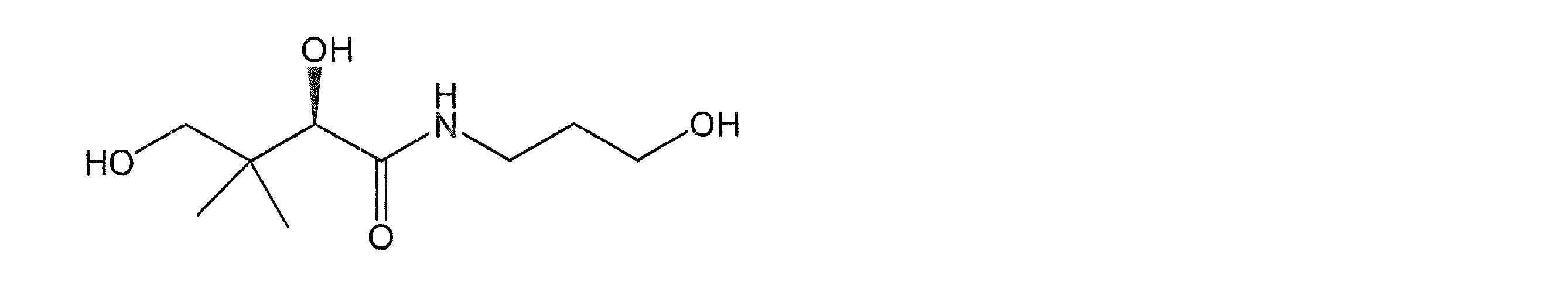
(f)D-パントテニルアルコール
(g)レチノール及びその誘導体
(f)ニコチン酸誘導体
(h)糖アルコール誘導体
(i)環状カルボキサミド誘導体
(j)トラネキサム酸及びその誘導体
(k)ウルソール酸リン酸エステル
(l)メラニン産生抑制作用を有する植物抽出物
In a preferred embodiment of the present invention, the other component having a whitening effect is one or more selected from the following (a) to (l).
(A) Compound represented by the following general formula (2) or a salt thereof (b) Compound represented by the following general formula (3) or a salt thereof (c) Compound represented by the following general formula (4) or a salt thereof Salt (d) Compound represented by the following general formula (8) or a salt thereof (e) Alkyl resorcinol (f) D-pantothenyl alcohol (g) Retinol and its derivative (f) Nicotinic acid derivative (h) Sugar alcohol derivative (I) Cyclic carboxamide derivative (j) Tranexamic acid and its derivative (k) Ursoleic acid phosphate ester (l) Plant extract having melanin production inhibitory action
[式(2)中、R3は、-SH、-SO3H、-S-S-X2、-S-X3、-SO-X4、又は-SO2-X5を表し、前記X2~X5は、独立して、水素原子又は炭素数1~8の脂肪族炭化水素基である。R4は無置換若しくは置換基を有していてもよい、炭素数5~12の芳香族基を表す。nは、1又は2の整数を表す。]
[In formula (2), R 3 represents -SH, -SO 3 H, -S-S-X 2 , -S-X 3 , -SO-X 4 , or -SO 2 -X 5 , as described above. X2 to X5 are independently hydrogen atoms or aliphatic hydrocarbon groups having 1 to 8 carbon atoms. R 4 represents an aromatic group having 5 to 12 carbon atoms, which may have an unsubstituted or substituent. n represents an integer of 1 or 2. ]
[式(3)中、R5は、無置換の又は置換基を有する芳香族基を表し、該置換基は炭素数1~4の直鎖若しくは分岐のアルキル基、炭素数1~4の直鎖若しくは分岐のアルコキシ基、ハロゲン原子、又は炭素数1~4のハロゲン化アルキル基であり、該芳香族基はフェニル基、ナフチル基、又はビフェニル基である。
R6は、水素原子、炭素数1~4の直鎖若しくは分岐のアルキル基、又は炭素数1~3の直鎖若しくは分岐のアルキルアシル基を表す。
R7は、水素原子又は炭素数1~4の直鎖若しくは分岐のアルキル基を表す。]
[In the formula (3), R 5 represents an aromatic group having an unsubstituted or substituent, and the substituent is a linear or branched alkyl group having 1 to 4 carbon atoms and a direct group having 1 to 4 carbon atoms. It is a chain or branched alkoxy group, a halogen atom, or an alkyl halide group having 1 to 4 carbon atoms, and the aromatic group is a phenyl group, a naphthyl group, or a biphenyl group.
R 6 represents a hydrogen atom, a linear or branched alkyl group having 1 to 4 carbon atoms, or a linear or branched alkyl acyl group having 1 to 3 carbon atoms.
R 7 represents a hydrogen atom or a linear or branched alkyl group having 1 to 4 carbon atoms. ]
[式(4)中、A1、A2、A3は独立して、置換基を有していてもよいフェニルを表す。前記置換基はヒドロキシル、炭素数1~4のアルキル、及び炭素数1~4のアルキルオキシから選択される。X6は窒素原子又は酸素原子である。-X6-R8は、下記一般式(5)、(6)又は(7)で表される。]
[In formula (4), A 1 , A 2 , and A 3 independently represent phenyl, which may have a substituent. The substituent is selected from hydroxyl, alkyl having 1 to 4 carbon atoms, and alkyloxy having 1 to 4 carbon atoms. X 6 is a nitrogen atom or an oxygen atom. -X 6 -R 8 is represented by the following general formulas (5), (6) or (7). ]
[式(5)中、X7は、窒素原子である。R9及びR10は互いに結合して、X7とともに、置換基を有していてもよい、炭素数2~8の複素環又は炭化水素環を形成する。前記置換基は、炭素数1~4のアルキル、炭素数1~4のアルキルオキシ、ヒドロキシル、アミノ、及びオキソから選ばれる。前記複素環の炭素数は3~5である。]
[In formula (5), X 7 is a nitrogen atom. R 9 and R 10 bond with each other to form a heterocycle or hydrocarbon ring having 2 to 8 carbon atoms, which may have a substituent, together with X 7 . The substituent is selected from alkyl having 1 to 4 carbon atoms, alkyloxy having 1 to 4 carbon atoms, hydroxyl, amino, and oxo. The heterocycle has 3 to 5 carbon atoms. ]
[式(6)中、X8は、窒素原子又は酸素原子である。mは、1~5の整数である。Y2は、ヒドロキシル基又はアミノ基である。R11は、X8が窒素原子のとき存在し、水素原子を表す。R11は、X8が酸素原子のときは存在しない。]
[In formula (6), X 8 is a nitrogen atom or an oxygen atom. m is an integer from 1 to 5. Y 2 is a hydroxyl group or an amino group. R 11 exists when X 8 is a nitrogen atom and represents a hydrogen atom. R 11 does not exist when X 8 is an oxygen atom. ]
[式(7)中、X9は、酸素原子又は窒素原子である。一般式(7)中、lは、X9に応じて存在する整数である。]
[In formula (7), X 9 is an oxygen atom or a nitrogen atom. In the general formula (7), l is an integer existing according to X 9 . ]
[式(8)中、R12は、カルボキシル基により置換された炭素数1~4の直鎖若しくは
分岐のアルキル基、又は炭素数1~4のアルキル鎖を有するカルボン酸エステル基により置換された炭素数1~4の直鎖若しくは分岐のアルキル基を表し、R13及びR14は、それぞれ独立に、炭素数1~4の直鎖又は分岐のアルキル基を表す。]
[In formula (8), R 12 was substituted with a linear or branched alkyl group having 1 to 4 carbon atoms substituted with a carboxyl group, or a carboxylic acid ester group having an alkyl chain having 1 to 4 carbon atoms. Represents a linear or branched alkyl group having 1 to 4 carbon atoms, and R 13 and R 14 independently represent a linear or branched alkyl group having 1 to 4 carbon atoms. ]
好ましい態様において、本発明の組成物は皮膚外用組成物であり、より好ましくは化粧料である。 In a preferred embodiment, the composition of the present invention is a composition for external use on the skin, more preferably a cosmetic.
本発明により、優れた美白作用を有する美白用組成物が提供される。 INDUSTRIAL APPLICABILITY The present invention provides a whitening composition having an excellent whitening effect.
本発明の美白用組成物は、下記一般式(1)で表される化合物又はその酸付加塩を含有する。 The whitening composition of the present invention contains a compound represented by the following general formula (1) or an acid addition salt thereof.
一般式(1)中、X1は、水素原子がメチル基で置換されていてもよい炭素数1~2のアルキレン基を表す。炭素数1~2のアルキレン基としては、メチレン基、及びエチレン基である。X1は-CH(CH3)-又は-CH2-CH2-が好ましい。 In the general formula (1), X 1 represents an alkylene group having 1 to 2 carbon atoms in which a hydrogen atom may be substituted with a methyl group. Examples of the alkylene group having 1 to 2 carbon atoms are a methylene group and an ethylene group. X 1 is preferably -CH (CH 3 )-or -CH 2 -CH 2- .
一般式(1)中、Y1は、COOR1又はCH2OR2を表し、R1は水素原子又は分岐を有してもよい炭素数1~6のアルキル基を表し、R2は水素原子又は分岐を有してもよい炭素数1~6のアシル基を表す。 In the general formula (1), Y 1 represents COOR 1 or CH 2 OR 2 , R 1 represents a hydrogen atom or an alkyl group having 1 to 6 carbon atoms which may have a branch, and R 2 represents a hydrogen atom. Alternatively, it represents an acyl group having 1 to 6 carbon atoms which may have a branch.
すなわち、Y1がCOOR1の場合、R1が水素原子のときY1はカルボキシル基であり、R1が分岐を有してもよい炭素数1~6のアルキル基のときY1はエステル基である。分岐を有してもよい炭素数1~6のアルキル基としては、例えば、メチル基、エチル基、n-プロピル基、iso-プロピル基、n-ブチル基、iso-ブチル基、tert-ブチル基、n-ペンチル基、n-ヘキシル基等が挙げられる。美白作用の観点から、R1は水素原子であることが特に好ましく、またR1がアルキル基のときはその炭素数は小さい方がより好ましい。 That is, when Y 1 is COOR 1 , Y 1 is a carboxyl group when R 1 is a hydrogen atom, and Y 1 is an ester group when R 1 is an alkyl group having 1 to 6 carbon atoms which may have a branch. Is. Examples of the alkyl group having 1 to 6 carbon atoms which may have a branch include a methyl group, an ethyl group, an n-propyl group, an iso-propyl group, an n-butyl group, an iso-butyl group and a tert-butyl group. , N-pentyl group, n-hexyl group and the like. From the viewpoint of whitening action, it is particularly preferable that R 1 is a hydrogen atom, and when R 1 is an alkyl group, it is more preferable that the number of carbon atoms is small.
また、Y1がCH2OR2の場合、R2が水素原子のときY1はヒドロキシメチル基であり、R2が分岐を有してもよい炭素数1~6のアシル基のときY1はエステル基である。分岐を有してもよい炭素数1~6のアシル基としては、例えば、ホルミル基、アセチル基、アクリロイル基、プロピオニル基、プロピオロイル基、ブチリル基、イソブチリル基、メタクリロイル基、バレリル基、カプロイル基等が挙げられる。美白作用の観点から、R2は水素原子であることが特に好ましく、またR2がアシル基のときはその炭素数は小さい方がより好ましい。 Further, when Y 1 is CH 2 OR 2 , Y 1 is a hydroxymethyl group when R 2 is a hydrogen atom, and Y 1 is an acyl group having 1 to 6 carbon atoms which may have a branch. Is an ester group. Examples of the acyl group having 1 to 6 carbon atoms which may have a branch include a formyl group, an acetyl group, an acryloyl group, a propionyl group, a propioloyl group, a butyryl group, an isobutyryl group, a methacryloyl group, a valeryl group and a caproyl group. Can be mentioned. From the viewpoint of whitening action, it is particularly preferable that R 2 is a hydrogen atom, and when R 2 is an acyl group, it is more preferable that the number of carbon atoms is small.
一般式(1)において、1,4-シクロヘキシレン基は、いす型又はボート型のいずれの形態でもよい。また、2つの結合手は、シス又はトランスのいずれの関係でもよい。好ましくは、いす型の形態でトランスの関係にあるものである。 In the general formula (1), the 1,4-cyclohexylene group may be in either a chair type or a boat type. Also, the two bonds may be in a cis or trans relationship. Preferably, it is in the form of a chair and has a transformer relationship.
下記一般式(1)で表される化合物の酸付加塩としては、無機酸、有機カルボン酸塩、又は有機スルホン酸との塩が挙げられる。無機酸としては、塩酸、臭化水素酸、硫酸、硝酸、リン酸等が挙げられ、有機カルボン酸塩としては、酢酸、プロピオン酸、マレイン酸、フマル酸、シュウ酸、クエン酸、酪酸、乳酸、酒石酸等が挙げられ、有機スルホン酸としては、メタンスルホン酸、エタンスルホン酸、ベンゼンスルホン酸、パラトルエンスルホン酸等が挙げられる。これらのうち無機酸塩が好ましく、塩酸塩がより好ましい。
本発明の美白用組成物の有効成分としては、一般式(1)で表される化合物又はその酸付加塩のいずれでもよいが、酸付加塩のほうがより好ましい。
Examples of the acid addition salt of the compound represented by the following general formula (1) include a salt with an inorganic acid, an organic carboxylate, or an organic sulfonic acid. Examples of the inorganic acid include hydrochloric acid, hydrobromic acid, sulfuric acid, nitric acid, phosphoric acid and the like, and examples of the organic carboxylate include acetic acid, propionic acid, maleic acid, fumaric acid, oxalic acid, citric acid, butyric acid and lactic acid. , Tartrate acid and the like, and examples of the organic sulfonic acid include methanesulfonic acid, ethanesulfonic acid, benzenesulfonic acid, paratoluenesulfonic acid and the like. Of these, inorganic acid salts are preferable, and hydrochlorides are more preferable.
As the active ingredient of the whitening composition of the present invention, either the compound represented by the general formula (1) or an acid addition salt thereof may be used, but the acid addition salt is more preferable.
一般式(1)で表される化合物の具体例を以下に挙げるが、これらにのみ限定されないことはいうまでもない。
2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]酢酸、2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]酢酸メチル、2-[p-(
4-アミノメチルシクロヘキシルカルボニル)フェニル]酢酸エチル、2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]酢酸プロピル、2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]酢酸ブチル、2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]酢酸ペンチル、2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]酢酸ヘキシル;
2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]エタノール、2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]エチルホルメート、2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]エチルアセテート、2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]エチルプロピオネート、2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]エチルブチレート、2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]エチルペンタノエート、2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]エチルヘキサノエート;
2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピオン酸、2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピオン酸メチル、2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピオン酸エチル、2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピオン酸プロピル、2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピオン酸ブチル、2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピオン酸ペンチル、2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピオン酸ヘキシル;
2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロパノール、2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピルホルメート、2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピルアセテート、2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピルプロピオネート、2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピルブチレート、2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピルペンタノエート、2-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピルヘキサノエート;
3-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピオン酸(化合物1)、3-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピオン酸メチル、3-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピオン酸エチル、3-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピオン酸プロピル、3-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピオン酸ブチル、3-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピオン酸ペンチル、3-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピオン酸ヘキシル(化合物2);
3-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロパノール(化合物3)、3-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピルホルメート、3-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピルアセテート、3-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピルプロピオネート、3-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピルブチレート、3-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピルペンタノエート、3-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピルヘキサノエート(化合物4);
2-メチル-3-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロパノール、2-メチル-3-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピルホルメート、2-メチル-3-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピルアセテート、2-メチル-3-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピルプロピオネート、2-メチル-3-
[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピルブチレート、2-メチル-3-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピルペンタノエート、2-メチル-3-[p-(4-アミノメチルシクロヘキシルカルボニル)フェニル]プロピルヘキサノエート(化合物5)。
Specific examples of the compound represented by the general formula (1) are given below, but it goes without saying that the compound is not limited to these.
2- [p- (4-Aminomethylcyclohexylcarbonyl) phenyl] acetic acid, 2- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] methyl acetate, 2- [p-(
4-Aminomethylcyclohexylcarbonyl) phenyl] ethyl acetate, 2- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] propyl acetate, 2- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] butyl acetate, 2- [P- (4-Aminomethylcyclohexylcarbonyl) phenyl] pentyl acetate, 2- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] hexyl acetate;
2- [p- (4-Aminomethylcyclohexylcarbonyl) phenyl] ethanol, 2- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] ethylformate, 2- [p- (4-aminomethylcyclohexylcarbonyl) phenyl ] Ethyl acetate, 2- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] ethyl propionate, 2- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] ethylbutyrate, 2- [p- (4) -Aminomethylcyclohexylcarbonyl) phenyl] ethylpentanoate, 2- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] ethylhexanoate;
2- [p- (4-Aminomethylcyclohexylcarbonyl) phenyl] propionic acid, 2- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] methyl propionate, 2- [p- (4-aminomethylcyclohexylcarbonyl) Phenyl] ethyl propionate, 2- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] propyl propionate, 2- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] butyl propionate, 2- [p-( 4-Aminomethylcyclohexylcarbonyl) phenyl] pentyl propionate, 2- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] hexyl propionate;
2- [p- (4-Aminomethylcyclohexylcarbonyl) phenyl] propanol, 2- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] propylformate, 2- [p- (4-aminomethylcyclohexylcarbonyl) phenyl ] Propyl acetate, 2- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] propylpropionate, 2- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] propylbutyrate, 2- [p- (4) -Aminomethylcyclohexylcarbonyl) phenyl] propylpentanoate, 2- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] propylhexanoate;
3- [p- (4-Aminomethylcyclohexylcarbonyl) phenyl] propionic acid (Compound 1), 3- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] methyl propionate, 3- [p- (4-amino) Methylcyclohexylcarbonyl) phenyl] ethyl propionate, 3- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] propyl propionate, 3- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] butyl propionate, 3- [P- (4-Aminomethylcyclohexylcarbonyl) phenyl] pentyl propionate, 3- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] hexyl propionate (Compound 2);
3- [p- (4-Aminomethylcyclohexylcarbonyl) phenyl] propanol (Compound 3), 3- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] propylformate, 3- [p- (4-aminomethyl) Cyclohexylcarbonyl) phenyl] propyl acetate, 3- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] propylpropionate, 3- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] propylbutyrate, 3-[ p- (4-Aminomethylcyclohexylcarbonyl) phenyl] propylpentanoate, 3- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] propylhexanoate (Compound 4);
2-Methyl-3- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] propanol, 2-methyl-3- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] propylformate, 2-methyl-3- [P- (4-Aminomethylcyclohexylcarbonyl) phenyl] propylacetate, 2-methyl-3- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] propylpropionate, 2-methyl-3-
[P- (4-Aminomethylcyclohexylcarbonyl) phenyl] propylbutyrate, 2-methyl-3- [p- (4-aminomethylcyclohexylcarbonyl) phenyl] propylpentanoate, 2-methyl-3- [p- (4-Aminomethylcyclohexylcarbonyl) phenyl] propylhexanoate (Compound 5).
一般式(1)で表される化合物は、定法により合成・精製して取得することができる。例えば、特許文献2に記載の、アミノカルボン酸ハライドの酸付加塩のルイス酸存在下でのアシル化反応によって合成し、適当な単離・精製方法を経て製造することができる。 The compound represented by the general formula (1) can be obtained by synthesizing and purifying by a conventional method. For example, it can be synthesized by an acylation reaction of an acid addition salt of an aminocarboxylic acid halide in the presence of Lewis acid described in Patent Document 2, and can be produced by an appropriate isolation / purification method.
一般式(1)で表される化合物及びその酸付加物は、優れた美白作用を有するため、美白用組成物の有効成分となる。
なお、本明細書において美白とは、色素沈着の予防及び/又は改善をいい、より具体的には、シミ、くすみ、そばかす、日焼け、皮膚の炎症や刺激による黒ずみ等のメラニン産生亢進、過剰蓄積、及び沈着異常等により生じる色素沈着症状、並びにステロイド等の薬物による皮膚の黒化症等の色素沈着をもたらす疾患等による色素沈着症状などの、予防及び/又は改善をいう。
一般式(1)で表される化合物及びその酸付加物による美白作用の機序については、詳細は不明であるが、チロシナ-ゼ阻害作用、チロシナ-ゼ遺伝子発現抑制作用、チロシナ-ゼ蛋白発現抑制作用、チロシナ-ゼ関連蛋白分解作用等のチロシナーゼ活性阻害作用、プロトンポンプ阻害作用、メラノサイトのケラチノサイトへのメラノソ-ム受け渡し阻害作用、又は新たな作用機序により、色素沈着を予防及び/又は改善することが推測される。
Since the compound represented by the general formula (1) and its acid adduct have an excellent whitening effect, they are active ingredients of the whitening composition.
In the present specification, whitening refers to prevention and / or improvement of pigmentation, and more specifically, enhancement of melanin production such as spots, dullness, freckles, sunburn, darkening due to skin inflammation and irritation, and excessive accumulation. , And pigmentation symptoms caused by abnormal deposition, etc., and pigmentation symptoms caused by diseases that cause pigmentation such as skin blackening caused by drugs such as steroids, etc., are prevented and / or improved.
The mechanism of the whitening action of the compound represented by the general formula (1) and its acid adduct is unknown, but it has a tyrosinase inhibitory action, a tyrosinase gene expression inhibitory action, and a tyrosinase protein expression. Prevents and / or improves pigmentation by inhibitory action, tyrosinase activity inhibitory action such as tyrosinase-related proteolytic action, proton pump inhibitory action, melanocyte transfer inhibitory action of melanocytes to keratinocytes, or a new mechanism of action. It is speculated that it will be done.
一般式(1)で表される化合物の構造は、トラネキサム酸と部分的に一致するが、生体内でトラネキサム酸には代謝されないことが分かっている。すなわち、一般式(1)で表される化合物の美白作用は、トラネキサム酸とは異なるメカニズムによるものと考えられ、少なくとも(4-アミノメチルシクロヘキシルカルボニル)フェニル基の構造が該作用の発揮に関与するものと推測される。
なお、特許文献1に記載のセトラキサートは、トラネキサム酸のエステル誘導体であり、それ自体の構造により、及び代謝によりエステル結合が切断されてトラネキサム酸になることにより美白作用を発揮する。
It is known that the structure of the compound represented by the general formula (1) is partially consistent with tranexamic acid, but is not metabolized to tranexamic acid in vivo. That is, the whitening action of the compound represented by the general formula (1) is considered to be due to a mechanism different from that of tranexamic acid, and at least the structure of the (4-aminomethylcyclohexylcarbonyl) phenyl group is involved in exerting the action. It is presumed to be.
The cetraxate described in Patent Document 1 is an ester derivative of tranexamic acid, and exerts a whitening effect by its own structure and by breaking the ester bond by metabolism to become tranexamic acid.
本発明の美白用組成物において、一般式(1)で表される化合物又はその酸付加塩の含有量は、組成物全量に対して総量で、好ましくは0.01%~20質量%、より好ましくは0.1~10質量%、さらに好ましくは1~5質量%である。かかる範囲において、所望の効果を得やすく、また処方設計の自由度を確保できる。 In the whitening composition of the present invention, the content of the compound represented by the general formula (1) or an acid addition salt thereof is the total amount, preferably 0.01% to 20% by mass, based on the total amount of the composition. It is preferably 0.1 to 10% by mass, more preferably 1 to 5% by mass. Within such a range, the desired effect can be easily obtained, and the degree of freedom in formulation design can be ensured.
本発明の美白用組成物は、一般式(1)で表される化合物又はその酸付加塩とともに、他の美白作用を有する成分を含有する。他の美白作用を有する成分は、好ましくは以下の(a)~(l)から選択される1種以上である。
(a)一般式(2)で表される化合物又はその塩
(b)一般式(3)で表される化合物又はその塩
(c)一般式(4)で表される化合物又はその塩
(d)一般式(8)で表される化合物又はその塩
(e)アルキルレゾルシノール
(f)D-パントテニルアルコール
(g)レチノール及びその誘導体
(f)ニコチン酸誘導体
(h)糖アルコール誘導体
(i)環状カルボキサミド誘導体
(j)トラネキサム酸及びその誘導体
(k)ウルソール酸リン酸エステル
(l)メラニン産生抑制作用を有する植物抽出物
The whitening composition of the present invention contains a compound represented by the general formula (1) or an acid addition salt thereof, as well as other components having a whitening effect. The other component having a whitening effect is preferably one or more selected from the following (a) to (l).
(A) Compound represented by general formula (2) or salt thereof (b) Compound represented by general formula (3) or salt thereof (c) Compound represented by general formula (4) or salt thereof (d) ) Compound represented by the general formula (8) or a salt thereof (e) Alkyl resorcinol (f) D-pantothenyl alcohol (g) Retinol and its derivative (f) Nicotinic acid derivative (h) Sugar alcohol derivative (i) Cyclic Carboxamide derivative (j) Tranexamic acid and its derivative (k) Ursoleic acid phosphate (l) Plant extract having melanin production inhibitory effect
なお、特に記載のない限り、光学異性体の存在する化合物については、L体、D体、及びラセミ体(DL体)の何れをも本発明に含むものとする。
また、ここでいう塩は、皮膚外用剤に使用されるものであれば、特段の限定無く使用できる。例えば、ナトリウム、カリウム等のアルカリ金属塩、カルシウム、マグネシウム等のアルカリ土類金属塩、アンモニウム塩、トリエチルアミン、トリエタノールアミン等の有機アミン塩、リジン、アルギニン等の塩基性アミノ酸塩等が好ましく例示される。これらの塩の内、特に好ましいものは、アルカリ金属であり、中でもナトリウム塩が特に好ましい。
Unless otherwise specified, the present invention includes any of L-form, D-form, and racemic-form (DL-form) for compounds in which optical isomers are present.
Further, the salt referred to here can be used without particular limitation as long as it is used as an external preparation for skin. For example, alkali metal salts such as sodium and potassium, alkaline earth metal salts such as calcium and magnesium, organic amine salts such as ammonium salt, triethylamine and triethanolamine, and basic amino acid salts such as lysine and arginine are preferably exemplified. To. Of these salts, particularly preferable ones are alkali metals, and among them, sodium salts are particularly preferable.
(a)一般式(2)において、R3は、-SH、-SO3H、-S-S-X2、-S-X3、-SO-X4、又は-SO2-X5を表し、前記X2~X5は、独立して、水素原子又は炭素数1~8の脂肪族炭化水素基である。R4は無置換若しくは置換基を有していてもよい、炭素数5~12の芳香族基を表し、前記芳香族基はフェニル基が好ましい。nは、1又は2の整数を表す。前記置換基は、炭素数1~4のアルキル基、炭素数1~4のアルコキシ基、又はフェニル基が好ましい。
一般式(2)で表される化合物についてより詳細には、国際公開2010/058730号を参照されたい。
一般式(2)で表される化合物の好ましい具体例は以下の化合物6~13である。
(A) In the general formula (2), R 3 is -SH, -SO 3 H, -S-S-X 2 , -S-X 3 , -SO-X 4 , or -SO 2 -X 5 . Represented, X2 to X5 are independently hydrogen atoms or aliphatic hydrocarbon groups having 1 to 8 carbon atoms. R 4 represents an aromatic group having 5 to 12 carbon atoms, which may be unsubstituted or may have a substituent, and the aromatic group is preferably a phenyl group. n represents an integer of 1 or 2. The substituent is preferably an alkyl group having 1 to 4 carbon atoms, an alkoxy group having 1 to 4 carbon atoms, or a phenyl group.
For more details on the compound represented by the general formula (2), refer to International Publication No. 2010/058730.
Preferred specific examples of the compound represented by the general formula (2) are the following compounds 6 to 13.
(b)下記一般式(3)において、R5は、無置換の又は置換基を有する芳香族基を表し、該置換基は炭素数1~4の直鎖若しくは分岐のアルキル基、炭素数1~4の直鎖若しくは分岐のアルコキシ基、ハロゲン原子、又は炭素数1~4のハロゲン化アルキル基であり、該芳香族基はフェニル基、ナフチル基、又はビフェニル基である。R6は、水素原子、炭素数1~4の直鎖若しくは分岐のアルキル基、又は炭素数1~3の直鎖若しくは分岐のアルキルアシル基を表す。R7は、水素原子又は炭素数1~4の直鎖若しくは分岐のアルキル基を表す。
一般式(3)で表される化合物についてより詳細には、国際公開2011/074643号を参照されたい。
一般式(3)で表される化合物の好ましい具体例は以下の化合物14~28である。
(B) In the following general formula (3), R 5 represents an unsubstituted or aromatic group having a substituent, and the substituent is a linear or branched alkyl group having 1 to 4 carbon atoms and 1 carbon number. It is a linear or branched alkoxy group of ~ 4, a halogen atom, or an alkyl halide group having 1 to 4 carbon atoms, and the aromatic group is a phenyl group, a naphthyl group, or a biphenyl group. R 6 represents a hydrogen atom, a linear or branched alkyl group having 1 to 4 carbon atoms, or a linear or branched alkyl acyl group having 1 to 3 carbon atoms. R 7 represents a hydrogen atom or a linear or branched alkyl group having 1 to 4 carbon atoms.
For more details on the compound represented by the general formula (3), refer to International Publication No. 2011/0746443.
Preferred specific examples of the compound represented by the general formula (3) are the following compounds 14 to 28.
(c)一般式(4)において、A1、A2、A3は独立して、置換基を有していてもよいフェニルを表す。前記置換基はヒドロキシル、炭素数1~4のアルキル、及び炭素数1~4のアルキルオキシから選択される。X6は窒素原子又は酸素原子である。-X6-R
8は、下記一般式(5)、(6)又は(7)で表される。
(C) In the general formula (4), A 1 , A 2 and A 3 independently represent phenyl which may have a substituent. The substituent is selected from hydroxyl, alkyl having 1 to 4 carbon atoms, and alkyloxy having 1 to 4 carbon atoms. X 6 is a nitrogen atom or an oxygen atom. -X 6 -R
8 is represented by the following general formula (5), (6) or (7).
一般式(5)において、X7は、窒素原子である。R9及びR10は互いに結合して、X7とともに、置換基を有していてもよい、炭素数2~8の複素環又は炭化水素環を形成する。前記置換基は、炭素数1~4のアルキル、炭素数1~4のアルキルオキシ、ヒドロキシル、アミノ、及びオキソから選ばれる。前記複素環の炭素数は3~5である。
一般式(4)中の-X6-R8が、一般式(5)で表される化合物の好ましい具体例は以下の化合物29~40である。
In the general formula (5), X 7 is a nitrogen atom. R 9 and R 10 bond with each other to form a heterocycle or hydrocarbon ring having 2 to 8 carbon atoms, which may have a substituent, together with X 7 . The substituent is selected from alkyl having 1 to 4 carbon atoms, alkyloxy having 1 to 4 carbon atoms, hydroxyl, amino, and oxo. The heterocycle has 3 to 5 carbon atoms.
Preferred specific examples of the compounds in which -X 6 -R 8 in the general formula (4) are represented by the general formula (5) are the following compounds 29 to 40.
一般式(6)において、X8は、窒素原子又は酸素原子である。mは、1~5の整数である。mは、好ましくは1~3の整数である。Y2は、ヒドロキシル基又はアミノ基である。R11は、X8が窒素原子のとき存在し、水素原子を表す。R11は、X8が酸素原子のときは存在しない。
一般式(4)中の-X6-R8が、一般式(6)で表される化合物の好ましい具体例は以下の化合物41~49である。
In the general formula (6), X 8 is a nitrogen atom or an oxygen atom. m is an integer from 1 to 5. m is preferably an integer of 1 to 3. Y 2 is a hydroxyl group or an amino group. R 11 exists when X 8 is a nitrogen atom and represents a hydrogen atom. R 11 does not exist when X 8 is an oxygen atom.
Preferred specific examples of the compounds in which -X 6 -R 8 in the general formula (4) are represented by the general formula (6) are the following compounds 41 to 49.
一般式(7)において、X9は、酸素原子又は窒素原子である。一般式(7)中、lは、X9に応じて存在する整数である。
一般式(4)中の-X6-R8が、一般式(6)で表される化合物の好ましい具体例は以下の化合物50~56である。
In the general formula (7), X 9 is an oxygen atom or a nitrogen atom. In the general formula (7), l is an integer existing according to X 9 .
Preferred specific examples of the compounds in which -X 6 -R 8 in the general formula (4) are represented by the general formula (6) are the following compounds 50 to 56.
一般式(4)で表される化合物についてより詳細には、国際公開2010/074052号を参照されたい。 For more details on the compound represented by the general formula (4), refer to International Publication No. 2010/074052.
(d)一般式(8)において、R12は、カルボキシル基により置換された炭素数1~4の直鎖若しくは分岐のアルキル基、又は炭素数1~4のアルキル鎖を有するカルボン酸エステル基により置換された炭素数1~4の直鎖若しくは分岐のアルキル基を表し、R13及びR14は、それぞれ独立に、炭素数1~4の直鎖又は分岐のアルキル基を表す。
一般式(8)で表される化合物は、好ましくは一般式(9)で表される化合物である。
一般式(9)において、R15は、カルボキシル基により置換された炭素数1~4の直鎖又は分岐のアルキル基を表し、R16及びR17は、それぞれ独立に、炭素数1~4の直鎖又は分岐のアルキル基を表す。
The compound represented by the general formula (8) is preferably a compound represented by the general formula (9).
In the general formula (9), R 15 represents a linear or branched alkyl group having 1 to 4 carbon atoms substituted with a carboxyl group, and R 16 and R 17 independently have 1 to 4 carbon atoms, respectively. Represents a linear or branched alkyl group.
一般式(9)で表される化合物としてより好ましいのは、3-[[4-(カルボキシメチルアミノカルボニル)フェニルカルボニル]-バリル-プロリル]アミノ-1,1,1-トリフルオロ-4-メチル-2-オキソペンタンであり、さらに好ましくは3(RS)-[[4-(カルボキシメチルアミノカルボニル)フェニルカルボニル]-L-バリル-L-プ
ロリル]アミノ-1,1,1-トリフルオロ-4-メチル-2-オキソペンタン(化合物
57)又はそのナトリウム塩である。
一般式(8)で表される化合物についてより詳細には、特公平06-99378号公報及び国際公開2017/221973号を参照されたい。
More preferable compounds represented by the general formula (9) are 3-[[4- (carboxymethylaminocarbonyl) phenylcarbonyl] -valyl-prolyl] amino-1,1,1-trifluoro-4-methyl. -2-oxopentane, more preferably 3 (RS)-[[4- (carboxymethylaminocarbonyl) phenylcarbonyl] -L-valyl-L-prolyl] amino-1,1,1-trifluoro-4. -Methyl-2-oxopentane (Compound 57) or a sodium salt thereof.
For more details on the compound represented by the general formula (8), refer to Japanese Patent Publication No. 06-99378 and International Publication No. 2017/221973.
(e)アルキルレゾルシノールについてより詳細には、国際公開2007/148472号を参照されたい。
4-アルキルレゾルシノールにおけるアルキル基は、炭素数3~10のアルキル基が好ましく、炭素数3~6のアルキル基がより好ましい。具体的には、n-プロピル基、イソプロピル基、n-ブチル基、イソブチル基、sec-ブチル基、tert-ブチル基、アミル基、n-ヘキシル基、シクロヘキシル基、オクチル基、イソオクチル基などが例示できる。本発明においては、特に4-n-ブチルレゾルシノールが特に好ましい。
(E) For more details on alkylresorcinol, see WO 2007/148472.
The alkyl group in 4-alkylresorcinol is preferably an alkyl group having 3 to 10 carbon atoms, and more preferably an alkyl group having 3 to 6 carbon atoms. Specific examples thereof include n-propyl group, isopropyl group, n-butyl group, isobutyl group, sec-butyl group, tert-butyl group, amyl group, n-hexyl group, cyclohexyl group, octyl group and isooctyl group. can. In the present invention, 4-n-butyl resorcinol is particularly preferable.
(f)D-パントテニルアルコールは、パントテン酸のアルコール型誘導体であり、以下に示す構造を有する。 (F) D-pantothenic alcohol is an alcohol-type derivative of pantothenic acid and has the structure shown below.
(g)レチノール及びその誘導体としては、レチノール、パルミチン酸レチノール、酢酸レチノール、レチナール等が好ましく挙げられる。 (G) As the retinol and its derivative, retinol, retinol palmitate, retinol acetate, retinal and the like are preferably mentioned.
(f)ニコチン酸誘導体としては、ニコチン酸アミド、ニコチン酸エステル等が好ましく挙げられ、であり、ニコチン酸エステルとしては、ニコチン酸トコフェロール、ニコチン酸ベンジル、ニコチン酸メチル、ニコチン酸エチル等が好ましい。 (F) As the nicotinic acid derivative, nicotinic acid amide, nicotinic acid ester and the like are preferably mentioned, and as the nicotinic acid ester, tocopherol nicotinate, benzyl nicotinate, methyl nicotinate, ethyl nicotinate and the like are preferable.
(h)糖アルコール誘導体としては、イソソルビド、ジイソプロピリデン-D-マンニトール等が好ましく挙げられる。糖アルコール誘導体についてより詳細には、特開第2008-081474号公報を参照されたい。 As the sugar alcohol derivative, isosorbide, diisopropyridene-D-mannitol and the like are preferably mentioned. For more details on sugar alcohol derivatives, refer to Japanese Patent Application Laid-Open No. 2008-081474.
(i)環状カルボキサミド誘導体としては、1-(2-ヒドロキシエチル)-2-イミダゾリジノン、1-(2-ヒドロキシエチル)-2-ピロリドン等が好ましく挙げられる。環状カルボキサミド誘導体についてより詳細には、国際公開2011-040496号を参照されたい。 As the cyclic carboxamide derivative (i), 1- (2-hydroxyethyl) -2-imidazolidinone, 1- (2-hydroxyethyl) -2-pyrrolidone and the like are preferably mentioned. For more details on cyclic carboxamide derivatives, see International Publication 2011-0404996.
(j)トラネキサム酸及びその誘導体としては、トラネキサム酸、トラネキサム酸の二量体(塩酸トランス-4-(トランス-4-アミノメチルシクロヘキサンカルボニル)アミノメチルシクロヘキサンカルボン酸)、トラネキサム酸とハイドロキノンのエステル体(トランス-4-アミノメチルシクロヘキサンカルボン酸4’-ヒドロキシフェニルエステル)、トラネキサム酸とゲンチシン酸のエステル体(2-(トランス-4-アミノメチルシクロヘキシルカルボニルオキシ)-5-ヒドロキシ安息香酸およびその塩)、トラネキサム酸のアミド体(トランス-4-アミノメチルシクロヘキサンカルボン酸メチルアミド及びその塩、トランス-4-アセチルアミノメチルシクロヘキサンカルボン酸及びその塩、トランス-4-(p-メトキシベンゾイル)アミノメチルシクロヘキサンカルボン酸及びその塩、トランス-4-グアニジノメチルシクロヘキサンカルボン酸及びその塩等)等が好ましく挙げられる。トラネキサム酸及びその誘導体についてより詳細には、特開平10-265321号公報を参照されたい。 (J) Tranexamic acid and its derivatives include tranexamic acid, dimer of tranexamic acid (trans-4- (trans-4-aminomethylcyclohexanecarbonyl) aminomethylcyclohexanecarboxylic acid), ester of tranexamic acid and hydroquinone. (Trans-4-aminomethylcyclohexanecarboxylic acid 4'-hydroxyphenyl ester), ester of tranexamic acid and gentisic acid (2- (trans-4-aminomethylcyclohexylcarbonyloxy) -5-hydroxybenzoic acid and its salt) , Tranexamic acid amide (trans-4-aminomethylcyclohexanecarboxylic acid methylamide and its salt, trans-4-acetylaminomethylcyclohexanecarboxylic acid and its salt, trans-4- (p-methoxybenzoyl) aminomethylcyclohexanecarboxylic acid And salts thereof, trans-4-guanidinomethylcyclohexanecarboxylic acid and salts thereof, etc.) and the like are preferably mentioned. For more details on tranexamic acid and its derivatives, refer to Japanese Patent Application Laid-Open No. 10-265321.
(k)ウルソール酸リン酸エステルは、以下に示す構造を有する。
ウルソール酸リン酸エステルについてより詳細には、国際公開2006/132033号を参照されたい。
(K) Ursolic acid phosphate has the structure shown below.
For more information on ursolic acid phosphates, see WO 2006/132033.
(l)メラニン産生抑制作用を有する植物抽出物としては、アセンヤクエキス、ヒキオコシエキス、エチナシ葉エキス、シコンエキス、ベニバナエキス、アボカドエキス、オクラエキス、キウイエキス、ゲットウ葉エキス、サボンソウエキス、スイカズラエキス、チャエキス、トウガシエキス、ニンニクエキス、ライム果汁エキス、納豆エキス、オレンジエキス、カノコソウエキス、キューカンバーエキス、キョウニンエキス、クチナシエキス、グレープフルーツエキス、ゴボウエキス、紅茶エキス、スギナエキス、ゼニアオイエキス、タイソウエキス、トマトエキス、ニンジンエキス、ブクリョウエキス、ユリエキス、レイシエキス、レタスエキス、レモンエキス、又はローヤルゼリーエキス等が好ましく挙げられる。これら植物抽出物は、1種のみを用いてもよく、2種以上を組み合わせて用い
てもよい。
(L) Examples of plant extracts having a melanin production inhibitory effect include Asenyaku extract, Hikiokoshi extract, Echinashi leaf extract, Shikon extract, Benibana extract, Avocado extract, Okla extract, Kiwi extract, Getto leaf extract, Sabonsou extract, and Suikazura extract. , Cha extract, pepper extract, garlic extract, lime juice extract, natto extract, orange extract, kanokosou extract, cucumber extract, kyonin extract, cuttlefish extract, grapefruit extract, gobo extract, tea extract, sugina extract, zegnaoi extract, taiso extract, tomato extract , Carrot extract, Bukuryo extract, Yuri extract, Reishi extract, Lettuce extract, Lemon extract, Royal jelly extract and the like are preferably mentioned. As these plant extracts, only one kind may be used, or two or more kinds may be used in combination.
これらの植物抽出物の抽出方法は特に限定されるものではないが、溶媒を用いた抽出法が好ましい。抽出を行う際には、原材料植物をそのまま使用することもできるが、粉末状に粉砕・細断して抽出に供した方が、穏和な条件で短時間に高い抽出効率で有効成分の抽出を行うことができる。
抽出温度は特に限定されるものではなく、原材料の粉砕物の大きさや溶媒の種類等に応じて適宜設定すればよい。通常は、室温から溶媒の沸点までの範囲内で設定される。また、抽出時間も特に限定されるものではなく、原材料の粉砕物の大きさ、溶媒の種類、抽出温度等に応じて適宜設定すればよい。さらに、抽出時には、撹拌を行ってもよいし、撹拌せず静置してもよいし、超音波を加えてもよい。
The extraction method of these plant extracts is not particularly limited, but an extraction method using a solvent is preferable. When extracting, the raw material plant can be used as it is, but it is better to crush and shred it into powder and use it for extraction under mild conditions in a short time with high extraction efficiency. It can be carried out.
The extraction temperature is not particularly limited, and may be appropriately set according to the size of the pulverized raw material, the type of solvent, and the like. Usually, it is set in the range from room temperature to the boiling point of the solvent. Further, the extraction time is not particularly limited, and may be appropriately set according to the size of the pulverized raw material, the type of solvent, the extraction temperature, and the like. Further, at the time of extraction, stirring may be performed, the mixture may be allowed to stand without stirring, or ultrasonic waves may be applied.
例えば、上記植物抽出物は、原材料を溶媒中に浸漬し、室温又は80℃~100℃にて抽出することができる。抽出処理により得られた抽出液をろ過後、そのまま又は必要に応じて濃縮若しくは乾固したものを、活性成分として使用することができる。なお、この抽出処理の際には、原材料は細断又は粉砕したものを用いてもよい。また、生の原材料又は乾燥した原材料を用いてもよいし、あるいは焙煎した原材料を用いてもよい。焙煎方法は特に限定されるものではないが、80~120℃で0.5時間~2時間焙煎する方法が挙げられる。 For example, the above plant extract can be extracted at room temperature or 80 ° C. to 100 ° C. by immersing the raw material in a solvent. The extract obtained by the extraction treatment is filtered and then concentrated or dried as it is, or if necessary, it can be used as an active ingredient. In this extraction process, the raw material may be shredded or crushed. In addition, raw raw materials or dried raw materials may be used, or roasted raw materials may be used. The roasting method is not particularly limited, and examples thereof include a method of roasting at 80 to 120 ° C. for 0.5 to 2 hours.
抽出に使用される溶媒の種類は特に限定されるものではないが、水(熱水等を含む)、アルコール(例えばメタノール、エタノール、1-プロパノール、2-プロパノール、1-ブタノール、2-ブタノール)、グリコール(例えば1,3-ブチレングリコール、プロピレングリコール)、グリセリン、ケトン(例えばアセトン、メチルエチルケトン)、エーテル(例えばジエチルエーテル、ジオキサン、テトラヒドロフラン、プロピルエーテル)、アセトニトリル、エステル(例えば酢酸エチル、酢酸ブチル)、脂肪族炭化水素(例えばヘキサン、ヘプタン、流動パラフィン)、芳香族炭化水素(例えばトルエン、キシレン)、ハロゲン化炭化水素(例えばクロロホルム)、又はこれらのうち2種以上の混合溶媒が好ましい。 The type of solvent used for extraction is not particularly limited, but water (including hot water, etc.), alcohol (for example, methanol, ethanol, 1-propanol, 2-propanol, 1-butanol, 2-butanol). , Glycol (eg 1,3-butylene glycol, propylene glycol), glycerin, ketones (eg acetone, methyl ethyl ketone), ethers (eg diethyl ether, dioxane, tetrahydrofuran, propyl ether), acetonitrile, esters (eg ethyl acetate, butyl acetate) , An aliphatic hydrocarbon (for example, hexane, heptane, liquid paraffin), an aromatic hydrocarbon (for example, toluene, xylene), a halogenated hydrocarbon (for example, chloroform), or a mixed solvent of two or more of these is preferable.
このような抽出操作により、原材料から有効成分が抽出され、溶媒に溶け込む。抽出物を含む溶媒は、そのまま使用してもよいが、数日静置して熟成させてから用いてもよい。さらに滅菌、洗浄、濾過、脱色、脱臭等の慣用の精製処理を加えてから使用してもよい。また、必要により濃縮又は希釈してから使用してもよい。さらに、溶媒を全て揮発させて固体状(乾燥物)としてから使用してもよいし、該乾燥物を任意の溶媒に再溶解して使用してもよい。 By such an extraction operation, the active ingredient is extracted from the raw material and dissolved in the solvent. The solvent containing the extract may be used as it is, or it may be allowed to stand for several days for aging before use. Further, it may be used after being subjected to conventional purification treatments such as sterilization, washing, filtration, decolorization and deodorization. Further, it may be used after being concentrated or diluted if necessary. Further, the solvent may be completely volatilized to form a solid (dried product) before use, or the dried product may be redissolved in an arbitrary solvent before use.
本発明の美白用組成物において、(a)~(l)から選択される美白作用を有する成分の含有量は、組成物全量に対して総量で、好ましくは0.01%~20質量%、より好ましくは0.1~10質量%、さらに好ましくは1~5質量%である。かかる範囲において、所望の効果を得やすく、また処方設計の自由度を確保できる。なお、(l)植物抽出物
の場合は、乾燥重量換算の量とする。
In the whitening composition of the present invention, the content of the component having a whitening action selected from (a) to (l) is the total amount, preferably 0.01% to 20% by mass, based on the total amount of the composition. It is more preferably 0.1 to 10% by mass, still more preferably 1 to 5% by mass. Within such a range, the desired effect can be easily obtained, and the degree of freedom in formulation design can be ensured. In the case of (l) plant extract, the amount is converted to dry weight.
本発明の美白用組成物は、特に経皮吸収による効果が期待できる皮膚外用組成物とすることが好ましい。皮膚外用組成物の態様としては、皮膚に外用で適用されるものであれば特に限定されないが、化粧料(医薬部外品を含む)、医薬品等が好ましく挙げられる。一般式(1)で表される化合物は、高い安全性が確認されており、日常的に使用される化粧料の態様で連続的に塗布することが可能である。
皮膚外用組成物の剤型としては、特に限定されず、例えば、ローション剤型、乳液やクリーム等の乳化剤型、オイル剤型、ジェル剤型、パック、洗浄料等が挙げられる。
The whitening composition of the present invention is preferably a composition for external use on the skin, which is expected to have an effect of percutaneous absorption. The mode of the composition for external use on the skin is not particularly limited as long as it is applied to the skin for external use, but cosmetics (including quasi-drugs), pharmaceuticals and the like are preferably mentioned. The compound represented by the general formula (1) has been confirmed to have high safety, and can be continuously applied in the form of cosmetics used on a daily basis.
The dosage form of the external composition for skin is not particularly limited, and examples thereof include a lotion formulation, an emulsifier form such as a milky lotion or cream, an oil formulation, a gel formulation, a pack, and a cleansing agent.
本発明の美白用組成物は、上述した以外にも通常の皮膚外用組成物に配合される成分を、本発明の効果を損なわない限りにおいて任意に含有することができる。
かかる成分としては、例えば、マカデミアナッツ油、アボカド油、トウモロコシ油、オリーブ油、ナタネ油、ゴマ油、ヒマシ油、サフラワー油、綿実油、ホホバ油、ヤシ油、パーム油、液状ラノリン、硬化ヤシ油、硬化油、モクロウ、硬化ヒマシ油、ミツロウ、キャンデリラロウ、カルナウバロウ、イボタロウ、ラノリン、還元ラノリン、硬質ラノリン、ホホバロウ等のオイル、ワックス類;流動パラフィン、スクワラン、プリスタン、オゾケライト、パラフィン、セレシン、ワセリン、マイクロクリスタリンワックス等の炭化水素類;オレイン酸、イソステアリン酸、ラウリン酸、ミリスチン酸、パルミチン酸、ステアリン酸、ベヘン酸、ウンデシレン酸等の高級脂肪酸類;セチルアルコール、ステアリルアルコール、イソステアリルアルコール、ベヘニルアルコール、オクチルドデカノール、ミリスチルアルコール、セトステアリルアルコール等の高級アルコール等;イソオクタン酸セチル、ミリスチン酸イソプロピル、イソステアリン酸ヘキシルデシル、アジピン酸ジイソプロピル、セバチン酸ジ-2-エチルヘキシル、乳酸セチル、リンゴ酸ジイソステアリル、ジ-2-エチルヘキサン酸エチレングリコール、ジカプリン酸ネオペンチルグリコール、ジ-2-ヘプチルウンデカン酸グリセリン、トリ-2-エチルヘキサン酸グリセリン、トリ-2-エチルヘキサン酸トリメチロールプロパン、トリイソステアリン酸トリメチロールプロパン、テトラ-2-エチルヘキサン酸ペンタンエリトリット等の合成エステル油類;ジメチルポリシロキサン、メチルフェニルポリシロキサン、ジフェニルポリシロキサン等の鎖状ポリシロキサン;オクタメチルシクロテトラシロキサン、デカメチルシクロペンタシロキサン、ドデカメチルシクロヘキサンシロキサン等の環状ポリシロキサン;アミノ変性ポリシロキサン、ポリエーテル変性ポリシロキサン、アルキル変性ポリシロキサン、フッ素変性ポリシロキサン等の変性ポリシロキサン等のシリコーン油等の油剤類;
In addition to the above, the whitening composition of the present invention may optionally contain components other than those described above that are blended in ordinary external composition for skin, as long as the effects of the present invention are not impaired.
Such ingredients include, for example, macadamia nut oil, avocado oil, corn oil, olive oil, rapeseed oil, sesame oil, castor oil, safflower oil, cottonseed oil, jojoba oil, palm oil, palm oil, liquid lanolin, hardened palm oil, hardened oil. , Mokuro, hardened castor oil, beeswax, candelilla wax, carnauba wax, ibotarou, lanolin, reduced lanolin, hard lanolin, jojobaro and other oils and waxes; Hydrocarbons such as wax; higher fatty acids such as oleic acid, isostearic acid, lauric acid, myristic acid, palmitic acid, stearic acid, bechenic acid, undesylene acid; cetyl alcohol, stearyl alcohol, isostearyl alcohol, behenyl alcohol, octyldodeca Higher alcohols such as ol, myristyl alcohol, cetostearyl alcohol, etc .; cetyl isooctanoate, isopropyl myristate, hexyldecyl isostearate, diisopropyl adipate, di-2-ethylhexyl sebatate, cetyl lactate, diisostearyl malate, di- Ethylene glycol 2-ethylhexanoate, neopentylglycol dicaprate, glycerin di-2-heptylundecanoate, glycerin tri-2-ethylhexanoate, trimethylolpropane tri-2-ethylhexanoate, trimethylolpropane triisostearate, Synthetic ester oils such as penta-2-ethylhexanoic acid pentanelitrit; chain polysiloxanes such as dimethylpolysiloxane, methylphenylpolysiloxane, diphenylpolysiloxane; octamethylcyclotetrasiloxane, decamethylcyclopentasiloxane, dodecamethyl Cyclic polysiloxanes such as cyclohexanesiloxane; oils such as silicone oils such as amino-modified polysiloxanes, polyether-modified polysiloxanes, alkyl-modified polysiloxanes, and modified polysiloxanes such as fluorine-modified polysiloxanes;
脂肪酸セッケン(ラウリン酸ナトリウム、パルミチン酸ナトリウム等)、ラウリル硫酸カリウム、アルキル硫酸トリエタノールアミンエーテル等のアニオン界面活性剤類;塩化ステアリルトリメチルアンモニウム、塩化ベンザルコニウム、ラウリルアミンオキサイド等のカチオン界面活性剤類;イミダゾリン系両性界面活性剤(2-ココイル-2-イミダゾリニウムヒドロキサイド-1-カルボキシエチロキシ2ナトリウム塩等)、ベタイン系界面活性剤(アルキルベタイン、アミドベタイン、スルホベタイン等)、アシルメチルタウリン等の両性界面活性剤類;ソルビタン脂肪酸エステル類(ソルビタンモノステアレート、セスキオレイン酸ソルビタン等)、グリセリン脂肪酸類(モノステアリン酸グリセリン等)、プロピレングリコール脂肪酸エステル類(モノステアリン酸プロピレングリコール等)、硬化ヒマシ油誘導体、グリセリンアルキルエーテル、POEソルビタン脂肪酸エステル類(POEソルビタンモノオレエート、モノステアリン酸ポリオキエチレンソルビタン等)、POEソルビット脂肪酸エステル類(POE-ソルビットモノラウレート等)、POEグリセリン脂肪酸エステル類(POE-グリセリンモノイソステアレート等)、POE脂肪酸エステル類(ポリエチレングリコールモノオレート、POEジステアレート等)、POEアルキルエーテル類(POE2-オクチルドデシルエーテル等)、POEアルキルフェニルエーテル類(POEノニルフェニルエーテル等)、プルロニック型類、P
OE・POPアルキルエーテル類(POE・POP2-デシルテトラデシルエーテル等)、テトロニック類、POEヒマシ油・硬化ヒマシ油誘導体(POEヒマシ油、POE硬化ヒマシ油等)、ショ糖脂肪酸エステル、アルキルグルコシド等の非イオン界面活性剤類;ポリエチレングリコール、グリセリン、1,3-ブチレングリコール、エリスリトール、ソルビトール、キシリトール、マルチトール、プロピレングリコール、ジプロピレングリコール、ジグリセリン、イソプレングリコール、1,2-ペンタンジオール、2,4-ヘキサンジオール、1,2-ヘキサンジオール、1,2-オクタンジオール等の多価アルコール類;
Anionic surfactants such as fatty acid soap (sodium laurate, sodium palmitate, etc.), potassium lauryl sulfate, triethanolamine ether alkylsulfate; cationic surfactants such as stearyltrimethylammonium chloride, benzalconium chloride, laurylamine oxide, etc. Kind: Imidazoline-based amphoteric surfactant (2-cocoyl-2-imidazolinium hydroxide-1-carboxyethyroxy disodium salt, etc.), betaine-based surfactant (alkylbetaine, amidobetaine, sulfobetaine, etc.), acyl Amphoteric surfactants such as methyltaurin; sorbitan fatty acid esters (sorbitan monostearate, sorbitan sesquioleate, etc.), glycerin fatty acids (glycerin monostearate, etc.), propylene glycol fatty acid esters (propylene glycol monostearate, etc.) ), Hardened castor oil derivative, glycerin alkyl ether, POE sorbitan fatty acid esters (POE sorbitan monooleate, polyochiethylene sorbitan monostearate, etc.), POE sorbit fatty acid esters (POE-sorbit monolaurate, etc.), POE glycerin Fatty acid esters (POE-glycerin monoisostearate, etc.), POE fatty acid esters (polyethylene glycol monooleate, POE distearate, etc.), POE alkyl ethers (POE2-octyldodecyl ether, etc.), POE alkylphenyl ethers (POE nonyl, etc.) Phenyl ether, etc.), Pluronic type, P
OE / POP alkyl ethers (POE / POP2-decyltetradecyl ethers, etc.), tetronics, POE castor oil / cured castor oil derivatives (POE castor oil, POE cured castor oil, etc.), sucrose fatty acid esters, alkyl glucosides, etc. Nonionic surfactants; polyethylene glycol, glycerin, 1,3-butylene glycol, erythritol, sorbitol, xylitol, martitol, propylene glycol, dipropylene glycol, diglycerin, isoprene glycol, 1,2-pentanediol, 2, , 4-Hexanediol, 1,2-hexanediol, 1,2-octanediol and other polyhydric alcohols;
ピロリドンカルボン酸ナトリウム、乳酸、乳酸ナトリウム等の保湿成分類;表面を処理されていてもよい、マイカ、タルク、カオリン、合成雲母、炭酸カルシウム、炭酸マグネシウム、無水ケイ酸(シリカ)、酸化アルミニウム、硫酸バリウム等の粉体類;表面を処理されていてもよい、ベンガラ、黄酸化鉄、黒酸化鉄、酸化コバルト、群青、紺青、酸化チタン、酸化亜鉛の無機顔料類;表面を処理されていてもよい、雲母チタン、魚燐箔、オキシ塩化ビスマス等のパ-ル剤類;レ-キ化されていてもよい赤色202号、赤色228号、赤色226号、黄色4号、青色404号、黄色5号、赤色505号、赤色230号、赤色223号、橙色201号、赤色213号、黄色204号、黄色203号、青色1号、緑色201号、紫色201号、赤色204号等の有機色素類;ポリエチレン末、ポリメタクリル酸メチル、ナイロン粉末、オルガノポリシロキサンエラストマー等の有機粉体類;パラアミノ安息香酸系紫外線吸収剤;アントラニル酸系紫外線吸収剤;サリチル酸系紫外線吸収剤;桂皮酸系紫外線吸収剤;ベンゾフェノン系紫外線吸収剤;糖系紫外線吸収剤;2-(2'-ヒドロキシ-5'-t-オクチルフェニル)ベンゾトリアゾール、4-メトキ
シ-4'-t-ブチルジベンゾイルメタン等の紫外線吸収剤類;
Moisturizing ingredients such as sodium pyrrolidone carboxylate, lactic acid, sodium lactate; surface treated, mica, talc, kaolin, synthetic mica, calcium carbonate, magnesium carbonate, silicic anhydride (silica), aluminum oxide, sulfuric acid Powders such as barium; surface-treated inorganic pigments such as red iron oxide, yellow iron oxide, black iron oxide, cobalt oxide, ultramarine, dark blue, titanium oxide, zinc oxide; even if the surface is treated Good, pearl agents such as mica titanium, fish phosphorus foil, bismuth oxychloride; may be raked Red No. 202, Red No. 228, Red No. 226, Yellow No. 4, Blue No. 404, Yellow Organic pigments such as No. 5, Red No. 505, Red No. 230, Red No. 223, Orange No. 201, Red No. 213, Yellow No. 204, Yellow No. 203, Blue No. 1, Green No. 201, Purple No. 201, Red No. 204, etc. Kind; Organic powders such as polyethylene powder, polymethyl methacrylate, nylon powder, organopolysiloxane elastomer; paraaminobenzoic acid-based UV absorber; anthranilic acid-based UV absorber; salicylic acid-based UV absorber; cinnamic acid-based UV absorber Agent; Benzophenone-based UV absorber; Sugar-based UV absorber; 2- (2'-Hydroxy-5'-t-octylphenyl) benzotriazole, 4-methoxy-4'-t-butyldibenzoylmethane, etc. Agents;
エタノール、イソプロパノール等の低級アルコール類;ビタミンA又はその誘導体、ビタミンB6塩酸塩、ビタミンB6トリパルミテート、ビタミンB6ジオクタノエート、ビタミンB2又はその誘導体、ビタミンB12、ビタミンB15又はその誘導体等のビタミンB類;α-トコフェロール、β-トコフェロール、γ-トコフェロール、ビタミンEアセテート等のビタミンE類、ビタミンD類、ビタミンH、パントテン酸、パンテチン、ピロロキノリンキノン等のビタミン類等;メチルパラベン、エチルパラベン、ブチルパラベン、フェノキシエタノール等の抗菌剤(防腐剤);グリチルリチン酸誘導体、グリチルレチン酸誘導体、サリチル酸誘導体、ヒノキチオール、酸化亜鉛、アラントイン等の消炎剤;レチノール、アスコルビン酸、トコフェロール、又はファルネシル酢酸エステル等のシワ改善剤;各種抽出物(例えば、オウバク、オウレン、シコン、シャクヤク、センブリ、バーチ、セージ、ビワ、ニンジン、アロエ、ゼニアオイ、アイリス、ブドウ、ヨクイニン、ヘチマ、ユリ、サフラン、センキュウ、ショウキュウ、オトギリソウ、オノニス、ニンニク、トウガラシ、チンピ、トウキ、海藻等);ローヤルゼリー、感光素、コレステロール誘導体等の賦活剤;ノニル酸ワレニルアミド、カプサイシン、ジンゲロン、タンニン酸等の血行促進剤;硫黄、チアントール等の抗脂漏剤;トラネキサム酸、チオタウリン、ヒポタウリン等の抗炎症剤;コラーゲン、ヒアルロン酸等の水溶性高分子;などが挙げられる。 Lower alcohols such as ethanol and isopropanol; Vitamin A or its derivatives, Vitamin B 6 hydrochloride, Vitamin B 6 trypalmitate, Vitamin B 6 dioctanoate, Vitamin B 2 or its derivatives, Vitamin B 12 , Vitamin B 15 or its derivatives Vitamin Bs such as α-tocopherol, β-tocopherol, γ-tocopherol, vitamin E acetate and other vitamin Es, vitamin Ds, vitamin H, pantothenic acid, pantetin, pyroquinolinquinone and other vitamins; methylparaben, Antibacterial agents (preservatives) such as ethylparaben, butylparaben, phenoxyethanol; anti-inflammatory agents such as glycyrrhizic acid derivatives, glycyrrhetinic acid derivatives, salicylic acid derivatives, hinokithiol, zinc oxide, allantin; retinol, ascorbic acid, tocopherol, farnesyl acetate, etc. Wrinkle improver; various extracts (eg, Oubaku, Ouren, Shikon, Shakuyaku, Senburi, Birch, Sage, Biwa, Carrot, Aloe, Zeniaoi, Iris, Grape, Yokuinin, Hechima, Yuri, Saffron, Senkyu, Shokyu, Otogirisou, Ononis, Carrot, Togarashi, Chimpi, Touki, Seaweed, etc.); Activators such as royal jelly, photosensitizers, cholesterol derivatives; Fat leaking agents; anti-inflammatory agents such as tranexamic acid, thiotaurine and hypotaurin; water-soluble polymers such as collagen and hyaluronic acid; and the like.
以下、具体的な実験例をあげて、本発明をさらに詳細に説明するが、本発明は以下の態様にのみ限定されない。 Hereinafter, the present invention will be described in more detail with reference to specific experimental examples, but the present invention is not limited to the following aspects.
表1に記載の化粧料(実施例1~16、比較例、及び参考例)を、それぞれ定法で調製した。
調製した各化粧料について、以下の方法で美白効果(色素沈着の改善効果)を評価した。すなわち、自由意思で参加したパネラー8名の左右上腕内側部に0.5cm×0.5c
mの試験部位を合計8ヶ所設けた。設けた部位に最少紅斑量(1MED)の紫外線照射を1日1回、3日連続して3回照射した。試験1日目の紫外線照射終了時(1回目照射終了後)より、1日2回25日連続して試験部位1ヶ所に比較例の製剤を、残りの7ヶ所それぞれに実施例1~9及び参考例のいずれか1つの化粧料を、各50μL塗布した(比較例:n=8、各実施例及び参考例:n=3)。25日間の塗布終了24時間後に色彩色差計(CR-300、コニカミノルタ株式会社)にて各試験部位の皮膚明度(L*値)を測定
し、実施例又は参考例の化粧料の塗布部位のL*値から比較例の化粧料の塗布部位のL*値を減じることにより皮膚明度の差(ΔL*値)を算出した。L*値は、色素沈着の程度が強いほど低い値となるため、ΔL*値が大きいほど、色素沈着が改善されたと判断することができる。
The cosmetics shown in Table 1 (Examples 1 to 16, Comparative Examples, and Reference Examples) were prepared by a conventional method.
The whitening effect (improvement effect of pigmentation) was evaluated for each of the prepared cosmetics by the following method. That is, 0.5 cm x 0.5 c on the medial side of the left and right upper arms of eight panelists who participated voluntarily.
A total of 8 test sites of m were provided. The provided site was irradiated with ultraviolet rays having a minimum amount of erythema (1 MED) once a day, three times in a row for three days. From the end of the ultraviolet irradiation on the first day of the test (after the end of the first irradiation), the formulation of the comparative example was applied to one test site twice a day for 25 consecutive days, and Examples 1 to 9 and each of the remaining 7 sites. 50 μL of each of the cosmetics of any one of the reference examples was applied (comparative example: n = 8, each example and reference example: n = 3). Twenty-four hours after the end of application for 25 days, the skin brightness (L * value) of each test site was measured with a color difference meter (CR-300, Konica Minolta Co., Ltd.), and the application site of the cosmetic of the example or reference example was measured. The difference in skin lightness (ΔL * value) was calculated by subtracting the L * value of the application site of the cosmetic in the comparative example from the L * value. Since the L * value becomes lower as the degree of pigmentation is stronger, it can be judged that the pigmentation is improved as the ΔL * value is larger.
本発明の美白用組成物は、優れた美白効果を奏するため、美白用化粧料等の皮膚外用組成物として好適であり、産業上非常に有用である。 Since the whitening composition of the present invention exerts an excellent whitening effect, it is suitable as an external composition for skin such as whitening cosmetics, and is very useful industrially.
Claims (4)
前記他の美白作用を有する成分が、以下の(a)~(k)から選択される1種以上である、美白用組成物。
(a)下記一般式(2)で表される化合物又はその塩
(b)下記一般式(3)で表される化合物又はその塩
(c)下記一般式(4)で表される化合物又はその塩
(d)下記一般式(8)で表される化合物又はその塩
(e)アルキルレゾルシノール
(f)D-パントテニルアルコール
(g)レチノール、パルミチン酸レチノール、酢酸レチノール、又はレチナール
(h)ニコチン酸アミド、ニコチン酸トコフェロール、ニコチン酸ベンジル、ニコチン酸メチル、又はニコチン酸エチル
(i)イソソルビド、又はジイソプロピリデン-D-マンニトール
(j)1-(2-ヒドロキシエチル)-2-イミダゾリジノン、又は1-(2-ヒドロキシエチル)-2-ピロリドン
(k)トラネキサム酸、トラネキサム酸の二量体、トラネキサム酸とハイドロキノンのエステル体、トラネキサム酸とゲンチシン酸のエステル体、又はトラネキサム酸のアミド体
[式(1)中、X1は水素原子がメチル基で置換されていてもよい炭素数1~2のアルキレン基を表し、Y1はCOOR1又はCH2OR2を表し、R1は水素原子又は分岐を有してもよい炭素数1~6のアルキル基を表し、R2は水素原子又は分岐を有してもよい炭
素数1~6のアシル基を表す。]
[式(2)中、R 3 は、-SH、-SO 3 H、-S-S-X 2 、-S-X 3 、-SO-X 4 、又は-SO 2 -X 5 を表し、前記X 2 ~X 5 は、独立して、水素原子又は炭素数1~8の脂肪族炭化水素基である。R 4 は無置換若しくは置換基を有していてもよい、炭素数5~12の芳香族基を表す。nは、1又は2の整数を表す。]
[式(3)中、R 5 は、無置換の又は置換基を有する芳香族基を表し、該置換基は炭素数1~4の直鎖若しくは分岐のアルキル基、炭素数1~4の直鎖若しくは分岐のアルコキシ基、ハロゲン原子、又は炭素数1~4のハロゲン化アルキル基であり、該芳香族基はフェニル基、ナフチル基、又はビフェニル基である。
R 6 は、水素原子、炭素数1~4の直鎖若しくは分岐のアルキル基、又は炭素数1~3の直鎖若しくは分岐のアルキルアシル基を表す。
R 7 は、水素原子又は炭素数1~4の直鎖若しくは分岐のアルキル基を表す。]
[式(4)中、A 1 、A 2 、A 3 は独立して、置換基を有していてもよいフェニルを表す。前記置換基はヒドロキシル、炭素数1~4のアルキル、及び炭素数1~4のアルキルオキシから選択される。X 6 は窒素原子又は酸素原子である。-X 6 -R 8 は、下記一般式(5)、(6)又は(7)で表される。]
[式(5)中、X 7 は、窒素原子である。R 9 及びR 10 は互いに結合して、X 7 とともに、置換基を有していてもよい、炭素数2~8の複素環又は炭化水素環を形成する。前記置換基は、炭素数1~4のアルキル、炭素数1~4のアルキルオキシ、ヒドロキシル、アミノ、及びオキソから選ばれる。前記複素環の炭素数は3~5である。]
[式(6)中、X 8 は、窒素原子又は酸素原子である。mは、1~5の整数である。Y 2 は、ヒドロキシル基又はアミノ基である。R 11 は、X 8 が窒素原子のとき存在し、水素原子を表す。R 11 は、X 8 が酸素原子のときは存在しない。]
[式(7)中、X 9 は、酸素原子又は窒素原子である。一般式(7)中、lは、X 9 に応じて存在する整数である。]
[式(8)中、R 12 は、カルボキシル基により置換された炭素数1~4の直鎖若しくは
分岐のアルキル基、又は炭素数1~4のアルキル鎖を有するカルボン酸エステル基により置換された炭素数1~4の直鎖若しくは分岐のアルキル基を表し、R 13 及びR 14 は、それぞれ独立に、炭素数1~4の直鎖又は分岐のアルキル基を表す。] It contains a compound represented by the following general formula (1) or an acid addition salt thereof, and other components having a whitening effect.
A whitening composition in which the other component having a whitening action is one or more selected from the following (a) to (k) .
(A) A compound represented by the following general formula (2) or a salt thereof.
(B) A compound represented by the following general formula (3) or a salt thereof.
(C) A compound represented by the following general formula (4) or a salt thereof.
(D) A compound represented by the following general formula (8) or a salt thereof.
(E) Alkylresorcinol
(F) D-pantothenyl alcohol
(G) Retinol, retinol palmitate, retinyl acetate, or retinal
(H) Nicotinamide, tocopherol nicotinate, benzyl nicotinate, methyl nicotinate, or ethyl nicotinate
(I) Isosorbide or diisopropylidene-D-mannitol
(J) 1- (2-Hydroxyethyl) -2-imidazolidinone, or 1- (2-hydroxyethyl) -2-pyrrolidone
(K) Tranexamic acid, dimer of tranexamic acid, ester of tranexamic acid and hydroquinone, ester of tranexamic acid and gentisic acid, or amide of tranexamic acid
[In formula (1), X 1 represents an alkylene group having 1 to 2 carbon atoms in which a hydrogen atom may be substituted with a methyl group, Y 1 represents COOR 1 or CH 2 OR 2 , and R 1 represents hydrogen. It represents an alkyl group having 1 to 6 carbon atoms which may have an atom or a branch, and R 2 represents an acyl group having 1 to 6 carbon atoms which may have a hydrogen atom or a branch. ]
[In formula (2), R 3 represents -SH, -SO 3 H, -S-S-X 2 , -S-X 3 , -SO-X 4 , or -SO 2 - X 5 , as described above. X2 to X5 are independently hydrogen atoms or aliphatic hydrocarbon groups having 1 to 8 carbon atoms. R 4 represents an aromatic group having 5 to 12 carbon atoms, which may have an unsubstituted or substituent. n represents an integer of 1 or 2. ]
[In the formula (3), R 5 represents an aromatic group having an unsubstituted or substituent, and the substituent is a linear or branched alkyl group having 1 to 4 carbon atoms and a direct group having 1 to 4 carbon atoms. It is a chain or branched alkoxy group, a halogen atom, or an alkyl halide group having 1 to 4 carbon atoms, and the aromatic group is a phenyl group, a naphthyl group, or a biphenyl group.
R 6 represents a hydrogen atom, a linear or branched alkyl group having 1 to 4 carbon atoms, or a linear or branched alkyl acyl group having 1 to 3 carbon atoms.
R 7 represents a hydrogen atom or a linear or branched alkyl group having 1 to 4 carbon atoms. ]
[In formula (4), A 1 , A 2 , and A 3 independently represent phenyl, which may have a substituent. The substituent is selected from hydroxyl, alkyl having 1 to 4 carbon atoms, and alkyloxy having 1 to 4 carbon atoms. X 6 is a nitrogen atom or an oxygen atom. -X 6 - R 8 is represented by the following general formulas (5), (6) or (7). ]
[In formula (5), X 7 is a nitrogen atom. R 9 and R 10 bond with each other to form a heterocycle or hydrocarbon ring having 2 to 8 carbon atoms, which may have a substituent, together with X 7 . The substituent is selected from alkyl having 1 to 4 carbon atoms, alkyloxy having 1 to 4 carbon atoms, hydroxyl, amino, and oxo. The heterocycle has 3 to 5 carbon atoms. ]
[In formula (6), X 8 is a nitrogen atom or an oxygen atom. m is an integer from 1 to 5. Y 2 is a hydroxyl group or an amino group. R 11 exists when X 8 is a nitrogen atom and represents a hydrogen atom. R 11 does not exist when X 8 is an oxygen atom. ]
[In formula (7), X 9 is an oxygen atom or a nitrogen atom. In the general formula (7), l is an integer existing according to X 9 . ]
[In the formula (8), R 12 is a straight chain having 1 to 4 carbon atoms substituted with a carboxyl group or
Represents a branched alkyl group or a linear or branched alkyl group having 1 to 4 carbon atoms substituted with a carboxylic acid ester group having an alkyl chain having 1 to 4 carbon atoms, and R 13 and R 14 are independent of each other. , Represents a linear or branched alkyl group having 1 to 4 carbon atoms. ]
前記トラネキサム酸とハイドロキノンのエステル体が、トランス-4-アミノメチルシクロヘキサンカルボン酸4’-ヒドロキシフェニルエステルであり、The ester of tranexamic acid and hydroquinone is a trans-4-aminomethylcyclohexanecarboxylic acid 4'-hydroxyphenyl ester.
前記トラネキサム酸とゲンチシン酸のエステル体が、2-(トランス-4-アミノメチルシクロヘキシルカルボニルオキシ)-5-ヒドロキシ安息香酸およびその塩であり、The ester of tranexamic acid and gentisic acid is 2- (trans-4-aminomethylcyclohexylcarbonyloxy) -5-hydroxybenzoic acid and a salt thereof.
前記トラネキサム酸のアミド体が、トランス-4-アミノメチルシクロヘキサンカルボン酸メチルアミド及びその塩、トランス-4-アセチルアミノメチルシクロヘキサンカルボン酸及びその塩、トランス-4-(p-メトキシベンゾイル)アミノメチルシクロヘキサンカルボン酸及びその塩、並びにトランス-4-グアニジノメチルシクロヘキサンカルボン酸及びその塩から選択される、請求項1に記載の組成物。The amide form of tranexamic acid is trans-4-aminomethylcyclohexanecarboxylic acid methylamide and its salt, trans-4-acetylaminomethylcyclohexanecarboxylic acid and its salt, trans-4- (p-methoxybenzoyl) aminomethylcyclohexanecarboxylic. The composition according to claim 1, which is selected from an acid and a salt thereof, and a trans-4-guanidinomethylcyclohexanecarboxylic acid and a salt thereof.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2018101964A JP7102658B2 (en) | 2018-05-29 | 2018-05-29 | Whitening composition |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2018101964A JP7102658B2 (en) | 2018-05-29 | 2018-05-29 | Whitening composition |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2019206484A JP2019206484A (en) | 2019-12-05 |
| JP7102658B2 true JP7102658B2 (en) | 2022-07-20 |
Family
ID=68767365
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2018101964A Active JP7102658B2 (en) | 2018-05-29 | 2018-05-29 | Whitening composition |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP7102658B2 (en) |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP7778509B2 (en) * | 2021-09-24 | 2025-12-02 | 株式会社 資生堂 | Cosmetics |
| WO2023120204A1 (en) * | 2021-12-21 | 2023-06-29 | 株式会社 資生堂 | Oil-in-water emulsion cosmetic |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20100016442A1 (en) | 2008-07-16 | 2010-01-21 | Dermworx Incorporated | Small molecule solubilization system |
| JP2013001660A (en) | 2011-06-14 | 2013-01-07 | Pola Chemical Industries Inc | Skin care preparation |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6036418B2 (en) * | 1980-07-21 | 1985-08-20 | 帝人株式会社 | Aminocarboxylic acid derivatives, their production methods, and antiulcer agents containing them as active ingredients |
| JPS5846049A (en) * | 1981-09-11 | 1983-03-17 | Teijin Ltd | Aminocarboxylic acid derivative and its preparation |
| JPS63270650A (en) * | 1987-04-30 | 1988-11-08 | Asahi Chem Ind Co Ltd | P-(trans-4-aminomethylcyclohexylcarbonyl) phenylpropionic acid |
| JP2999203B2 (en) * | 1989-08-29 | 2000-01-17 | 第一製薬株式会社 | Sunshine composition |
| ES2171459T3 (en) * | 1993-07-30 | 2002-09-16 | Teijin Ltd | NASAL ADMINISTRATION POWDER FOR PEPTIDIC PHARMACO OR PROTEINACEO. |
-
2018
- 2018-05-29 JP JP2018101964A patent/JP7102658B2/en active Active
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20100016442A1 (en) | 2008-07-16 | 2010-01-21 | Dermworx Incorporated | Small molecule solubilization system |
| JP2013001660A (en) | 2011-06-14 | 2013-01-07 | Pola Chemical Industries Inc | Skin care preparation |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2019206484A (en) | 2019-12-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP4605774B2 (en) | Whitening agent, whitening skin external preparation and whitening method | |
| JP2020180103A (en) | Wrinkle-improving composition | |
| JP7102658B2 (en) | Whitening composition | |
| JP7098537B2 (en) | Drugs containing sclareolide | |
| JP7102657B2 (en) | Wrinkle improving composition | |
| JP7239573B2 (en) | whitening agent | |
| TWI871483B (en) | Composition for brightening containing sodium pyruvate | |
| JP4959104B2 (en) | Whitening agent and external preparation for skin | |
| JP4959103B2 (en) | Whitening agent and external preparation for skin | |
| HK40070862A (en) | Wrinkle ameliorating agent | |
| HK40090230B (en) | Whitening cosmetic composition comprising sodium pyruvate as active ingredient | |
| HK40090230A (en) | Whitening cosmetic composition comprising sodium pyruvate as active ingredient | |
| HK40049697A (en) | Skin lightening agent | |
| HK40049697B (en) | Skin lightening agent | |
| KR20040108405A (en) | Cosmetic Composition for Skin-Whitening Comprising Raspberry Ketone as Active Ingredient | |
| JP2020029400A (en) | Whitening agent |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20210420 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20220126 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20220222 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20220415 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20220607 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20220614 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 7102658 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |