JP5417662B2 - ネビラピンの徐放性製剤 - Google Patents
ネビラピンの徐放性製剤 Download PDFInfo
- Publication number
- JP5417662B2 JP5417662B2 JP2009552940A JP2009552940A JP5417662B2 JP 5417662 B2 JP5417662 B2 JP 5417662B2 JP 2009552940 A JP2009552940 A JP 2009552940A JP 2009552940 A JP2009552940 A JP 2009552940A JP 5417662 B2 JP5417662 B2 JP 5417662B2
- Authority
- JP
- Japan
- Prior art keywords
- nevirapine
- tablet
- hypromellose
- max
- sustained release
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/205—Polysaccharides, e.g. alginate, gums; Cyclodextrin
- A61K9/2054—Cellulose; Cellulose derivatives, e.g. hydroxypropyl methylcellulose
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/55—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole
- A61K31/551—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole having two nitrogen atoms, e.g. dilazep
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/14—Antivirals for RNA viruses
- A61P31/18—Antivirals for RNA viruses for HIV
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Medicinal Chemistry (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Chemical & Material Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Virology (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Oncology (AREA)
- Communicable Diseases (AREA)
- Molecular Biology (AREA)
- Tropical Medicine & Parasitology (AREA)
- AIDS & HIV (AREA)
- Medicinal Preparation (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Description
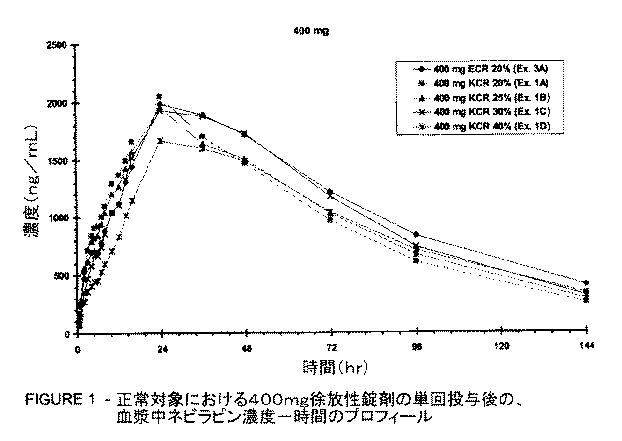
(a)1,000〜2,300ng/mLの幾何平均Cmax、および
(b)73,400〜178,100ng・h/mLの幾何平均AUC0-∞によって
ならびに、400mgのネビラピンを含む剤形では、次の2つのパラメーター:
(a)1,210〜2,740ng/mLの幾何平均Cmax、および
(b)109,000〜237,000ng・h/mLの幾何平均AUC0-∞によって特徴付けられる、特有の薬物動態プロフィールを示す。
300mg用量の場合は
(a)2,300〜3,700ng/mLの幾何平均Cmax,ss、および
(b)45,000〜75,000ng・h/mLの幾何平均AUC0-24h,ssによって、
400mg用量の場合は
(a)3,100〜4,900ng/mLの幾何平均Cmax,ss、および
(b)60,000〜99,000ng・h/mLの幾何平均AUC0-24h,ssによって特徴付けられる。
(a)400mgの無水ネビラピン;
(b)270mgのヒプロメロース2208(Methocel(商標)K4M Premium CR);
(c)400mgの乳糖一水和物;および
(d)10mgのステアリン酸マグネシウム
を含有する医薬錠剤であり、ここで、各錠剤は、約10〜25kNの力で圧縮される。
乳糖、ネビラピン薬物、およびヒプロメロースを、混合し、次いで水を用いて完全に顆粒化する。次いで、顆粒を乾燥する。乾燥した顆粒を、製粉し、滑沢剤(ステアリン酸マグネシウム)と一緒にして混合し、錠剤に圧縮するための最終混合物を調製する。錠剤は、約10〜25kNの力で9.3×19.0mmの錠剤に圧縮される。
乳糖、ネビラピン薬物、およびヒプロメロースを、混合し、次いで水を用いて完全に顆粒化する。次いで、顆粒を乾燥する。乾燥した顆粒を、製粉し、滑沢剤(ステアリン酸マグネシウム)と一緒にして混合し、錠剤に圧縮するための最終混合物を調製する。錠剤は、約10〜25kNの力で9.3×19.0mmの錠剤に圧縮される。
乳糖、ネビラピン薬物、およびヒプロメロースを、混合し、次いで水を用いて完全に顆粒化する。次いで、顆粒を乾燥する。乾燥した顆粒を、製粉し、滑沢剤(ステアリン酸マグネシウム)と一緒にして混合し、錠剤に圧縮するための最終混合物を調製する。錠剤は、9.3×19.0mmの錠剤に圧縮される。
乳糖、ネビラピン薬物、およびヒプロメロースを、混合し、次いで水を用いて完全に顆粒化する。次いで、顆粒を乾燥する。乾燥した顆粒を、製粉し、滑沢剤(ステアリン酸マグネシウム)と一緒にして混合し、錠剤に圧縮するための最終混合物を調製する。錠剤は、約10〜25kNの力で9.3×19.0mmの錠剤に圧縮される。
実施例1、2、3および4で説明した5種の製剤の溶出プロフィールを図4に示す。示した溶出情報は、下記の試験方法によって得られた。
米国薬局方のパドル法は、例えば米国薬局方XXII(1990)中に記載されているパドル法である。
溶出パラメーター:
・媒体容積:900mL
・温度:37.3℃
・回転速度:50rpm
・サンプリング時点:最初の4時間は30分毎、次いで1時間毎(4〜24時間)
・無限大回転:250rpmで15分間
・オンラインフィルター:10μmの全流式フィルター(VanKel社、ノースカロライナ州Cary;またはQuality Lab Accessories L.L.S.)
Claims (6)
- (a)20重量%〜25重量%のヒプロメロースを含む徐放性マトリックス中に300mgのネビラピンを含み、かつ絶食しているヒトに単回投与として投与された場合に、
(1)1,000〜2,300ng/mLの幾何平均Cmax;および
(2)73,400〜178,100hr(ng/mL)の幾何平均AUC0-∞を示し、ヒトに多回投与で投与された場合に、
(1)2,300〜3,700ng/mLの幾何平均Cmax,ss;および
(2)45,000〜75,000hr(ng/mL)の幾何平均AUC0-24,ssを示す;あるいは
(b)20重量%〜25重量%のヒプロメロースを含む徐放性マトリックス中に400mgのネビラピンを含み、かつ絶食しているヒトに単回投与として投与された場合に、
(1)1,210〜2,740ng/mLの幾何平均Cmax;および
(2)109,000〜237,000hr(ng/mL)の幾何平均AUC0-∞を示し、ヒトに多回投与で投与された場合に、
(1)3,100〜4,900ng/mLの幾何平均Cmax,ss;および
(2)60,000〜99,000hr(ng/mL)の幾何平均AUC0-24,ssを示す、錠剤の形態の医薬組成物であって、
(a)及び(b)の両組成物が10時間〜48時間の絶食単回投与Tmaxを示し、多回投与中に測定して1.1〜2の範囲であるCmax,ss/Cmin,ssを示す、医薬組成物。 - 徐放性マトリックス中にネビラピンを含む医薬組成物であって、該組成物が、溶出を、米国薬局方のパドル法を使用して、50rpmで、6%w/wのラウリル硫酸ナトリウムを含む、容積が900mLでpHが6.8の水性緩衝液で、37℃で測定した場合に、2時間の時点で2%w/w以上30%w/w以下のネビラピンが放出され;8時間の時点で20%w/w以上100%w/w以下のネビラピンが放出され;14時間の時点で40%w/w以上100%w/w以下のネビラピンが放出されるようなインビトロでの溶出プロフィールを有する、請求項1記載の医薬組成物。
- 各錠剤が、
(a)400mgの無水ネビラピン;
(b)270mgのヒプロメロース2208;
(c)400mgの乳糖一水和物;および
(d)10mgのステアリン酸マグネシウム
を含有し、
各錠剤が10〜25kNの力で圧縮される、請求項1又は2記載の錠剤型医薬剤形。 - 各錠剤が、
(a)400mgの無水ネビラピン;
(b)202.50mg及び270mgから選択される量のヒプロメロース2208又は2910;
(c)400mgの乳糖一水和物;および
(d)10mgのステアリン酸マグネシウム
を含有し、
各錠剤が10〜25kNの力で圧縮される、請求項1又は2記載の錠剤型医薬剤形。 - 各錠剤が、
(a)400mgの無水ネビラピン;
(b)270mgのヒプロメロース2208;
(c)400mgの乳糖一水和物;および
(d)10mgのステアリン酸マグネシウム
を含有し、
各錠剤が10〜25kNの力で圧縮される、請求項1記載の錠剤型医薬剤形。 - 各錠剤が、
(a)300mgの無水ネビラピン;
(b)151.875mg及び202.50mgから選択される量のヒプロメロース2208又は2910;
(c)300mgの乳糖一水和物;および
(d)7.50mgのステアリン酸マグネシウム
を含有し、
各錠剤が10〜25kNの力で圧縮される、請求項1又は2記載の錠剤型医薬剤形。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US94276507P | 2007-06-08 | 2007-06-08 | |
| US60/942,765 | 2007-06-08 | ||
| PCT/US2008/065705 WO2008154234A2 (en) | 2007-06-08 | 2008-06-04 | Extended release formulation of nevirapine |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2010520891A JP2010520891A (ja) | 2010-06-17 |
| JP5417662B2 true JP5417662B2 (ja) | 2014-02-19 |
Family
ID=39739795
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2009552940A Active JP5417662B2 (ja) | 2007-06-08 | 2008-06-04 | ネビラピンの徐放性製剤 |
Country Status (28)
| Country | Link |
|---|---|
| US (1) | US8460704B2 (ja) |
| EP (1) | EP2155169B1 (ja) |
| JP (1) | JP5417662B2 (ja) |
| KR (1) | KR101017862B1 (ja) |
| CN (1) | CN101784263B (ja) |
| AR (1) | AR066924A1 (ja) |
| AU (1) | AU2008262031B2 (ja) |
| BR (1) | BRPI0811732A2 (ja) |
| CA (1) | CA2687491C (ja) |
| CL (1) | CL2008001678A1 (ja) |
| CO (1) | CO6150128A2 (ja) |
| DK (1) | DK2155169T3 (ja) |
| EA (1) | EA018377B1 (ja) |
| EC (1) | ECSP099561A (ja) |
| ES (1) | ES2574836T3 (ja) |
| HU (1) | HUE028598T2 (ja) |
| IL (1) | IL199924A0 (ja) |
| MA (1) | MA31430B1 (ja) |
| MX (1) | MX2009007764A (ja) |
| NZ (1) | NZ578664A (ja) |
| PE (2) | PE20090371A1 (ja) |
| PL (1) | PL2155169T3 (ja) |
| TN (1) | TN2009000510A1 (ja) |
| TW (1) | TWI419716B (ja) |
| UA (1) | UA97971C2 (ja) |
| UY (1) | UY31128A1 (ja) |
| WO (1) | WO2008154234A2 (ja) |
| ZA (1) | ZA200904939B (ja) |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| MY149159A (en) | 2005-11-15 | 2013-07-31 | Hoffmann La Roche | Method for treating joint damage |
| US20130172274A1 (en) | 2005-12-20 | 2013-07-04 | Duke University | Methods and compositions for delivering active agents with enhanced pharmacological properties |
| NZ578664A (en) | 2007-06-08 | 2011-11-25 | Boehringer Ingelheim Int | Extended release formulation of nevirapine |
| EP4074327A1 (en) | 2008-06-27 | 2022-10-19 | Duke University | Therapeutic agents comprising elastin-like peptides |
| WO2012164241A1 (en) | 2011-05-30 | 2012-12-06 | Cipla Limited | Pharmaceutical antiretroviral composition |
| CN104023784B (zh) * | 2011-08-24 | 2018-05-25 | 费斯生物制药公司 | 供持续释放的活性剂制剂 |
| BR112014021927A2 (pt) * | 2012-03-05 | 2019-09-24 | Cipla Ltd | composição farmacêutica antirretroviral, processo para preparar uma composição farmacêutica antirretroviral, método para o tratamento ou profilaxia de doenças causadas por retrovírus, e, uso de lamivudina, festinavir e nevirapina |
| CN104523630B (zh) * | 2015-01-22 | 2017-08-25 | 山东新时代药业有限公司 | 一种奈韦拉平片剂 |
| EP3153157A1 (en) | 2015-10-09 | 2017-04-12 | Teva Pharmaceutical Works Private Limited Company | Pharmaceutical composition for prolonged release of nevirapine |
Family Cites Families (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5366972A (en) * | 1989-04-20 | 1994-11-22 | Boehringer Ingelheim Pharmaceuticals, Inc. | 5,11-dihydro-6H-dipyrido(3,2-B:2',3'-E)(1,4)diazepines and their use in the prevention or treatment of HIV infection |
| US5133974A (en) * | 1989-05-05 | 1992-07-28 | Kv Pharmaceutical Company | Extended release pharmaceutical formulations |
| CA2030056C (en) | 1989-11-17 | 1995-10-17 | Karl D. Hargrave | 5,11-dihydro-6h-dipyrido[3,2-b:2',3'-e][1,4]diazepines and their use in the prevention or treatment of hiv infection |
| US5419917A (en) * | 1994-02-14 | 1995-05-30 | Andrx Pharmaceuticals, Inc. | Controlled release hydrogel formulation |
| US6514530B2 (en) * | 1997-09-09 | 2003-02-04 | Alza Corporation | Dosage form comprising means for changing drug delivery shape |
| DK1035834T3 (da) * | 1997-12-05 | 2002-07-08 | Alza Corp | Osmotisk doseringsform omfattende en første og anden coating |
| JP2002532406A (ja) * | 1998-12-17 | 2002-10-02 | アルザ・コーポレーション | 複合コーティングによる液体充填ゼラチンカプセルの放出制御システムへの変換 |
| WO2000059477A1 (en) * | 1999-03-31 | 2000-10-12 | Janssen Pharmaceutica N.V. | Pregelatinized starch in a controlled release formulation |
| AU775360B2 (en) * | 1999-09-24 | 2004-07-29 | Janssen Pharmaceutica N.V. | Antiviral compositions |
| AU2084801A (en) | 1999-12-09 | 2001-06-18 | Alza Corporation | Antiviral medication |
| US6541014B2 (en) * | 2000-10-13 | 2003-04-01 | Advancis Pharmaceutical Corp. | Antiviral product, use and formulation thereof |
| WO2002092095A1 (en) | 2001-05-11 | 2002-11-21 | Boehringer Ingelheim International Gmbh | Use of nevirapine to treat or prevent lipid pathology in patients with hiv that is resistant to nevirapine |
| US20030050620A1 (en) | 2001-09-07 | 2003-03-13 | Isa Odidi | Combinatorial type controlled release drug delivery device |
| AU2003287666A1 (en) | 2002-11-13 | 2004-06-03 | Control Delivery Systems, Inc. | Systemic delivery of antiviral agents |
| CA2529852A1 (en) | 2003-06-20 | 2004-12-29 | Viral Genomix, Inc. | Compositions for and methods for treating hiv |
| DE602004005734T2 (de) | 2003-10-01 | 2007-12-27 | Lupin Ltd., Mumbai | Pharmazeutische zusammensetzung mit kontrollierter freisetzung und verfahren zu ihrer herstellung |
| ES2447816T7 (es) | 2003-11-13 | 2014-06-25 | Psivida Us, Inc. | Implante inyectable de liberación prolongada que comprende un núcleo de matriz bioerosionable y una piel bioerosionable |
| CA2577288C (en) * | 2004-09-02 | 2010-11-30 | Janssen Pharmaceutica N.V. | Hydrochloride of 4-[[4-[[4-(2-cyanoethenyl)-2,6-dimethylphenyl]amino]-2-pyrimidinyl]amino]benzonitrile |
| WO2006114709A1 (en) * | 2005-04-25 | 2006-11-02 | Aurobindo Pharma Limited | Pharmaceutical compositions of antiretrovirals |
| BRPI0613070A2 (pt) | 2005-06-29 | 2010-12-21 | Panacea Biotec Ltd | composições farmacêuticas de liberação sustentada e seus processos |
| US9198862B2 (en) | 2005-07-22 | 2015-12-01 | Rubicon Research Private Limited | Dispersible tablet composition |
| FR2891459B1 (fr) * | 2005-09-30 | 2007-12-28 | Flamel Technologies Sa | Microparticules a liberation modifiee d'au moins un principe actif et forme galenique orale en comprenant |
| CA2625776A1 (en) * | 2005-10-14 | 2007-04-26 | Microdose Technologies, Inc. | Pharmaceutical packaging of an oral dosage combination |
| NZ578664A (en) | 2007-06-08 | 2011-11-25 | Boehringer Ingelheim Int | Extended release formulation of nevirapine |
-
2008
- 2008-06-04 NZ NZ578664A patent/NZ578664A/en not_active IP Right Cessation
- 2008-06-04 US US12/523,226 patent/US8460704B2/en active Active
- 2008-06-04 JP JP2009552940A patent/JP5417662B2/ja active Active
- 2008-06-04 HU HUE08756672A patent/HUE028598T2/en unknown
- 2008-06-04 ES ES08756672.5T patent/ES2574836T3/es active Active
- 2008-06-04 CA CA2687491A patent/CA2687491C/en active Active
- 2008-06-04 EA EA200900958A patent/EA018377B1/ru not_active IP Right Cessation
- 2008-06-04 MX MX2009007764A patent/MX2009007764A/es active IP Right Grant
- 2008-06-04 UA UAA200908230A patent/UA97971C2/ru unknown
- 2008-06-04 PL PL08756672.5T patent/PL2155169T3/pl unknown
- 2008-06-04 EP EP08756672.5A patent/EP2155169B1/en not_active Revoked
- 2008-06-04 CN CN2008800193577A patent/CN101784263B/zh active Active
- 2008-06-04 WO PCT/US2008/065705 patent/WO2008154234A2/en not_active Ceased
- 2008-06-04 BR BRPI0811732-2A2A patent/BRPI0811732A2/pt not_active IP Right Cessation
- 2008-06-04 DK DK08756672.5T patent/DK2155169T3/en active
- 2008-06-04 KR KR1020097016610A patent/KR101017862B1/ko active Active
- 2008-06-04 AU AU2008262031A patent/AU2008262031B2/en not_active Ceased
- 2008-06-06 UY UY31128A patent/UY31128A1/es not_active Application Discontinuation
- 2008-06-06 PE PE2008000969A patent/PE20090371A1/es not_active Application Discontinuation
- 2008-06-06 CL CL2008001678A patent/CL2008001678A1/es unknown
- 2008-06-06 AR ARP080102446A patent/AR066924A1/es unknown
- 2008-06-06 PE PE2013000070A patent/PE20131035A1/es not_active Application Discontinuation
- 2008-06-06 TW TW097121294A patent/TWI419716B/zh active
-
2009
- 2009-07-15 ZA ZA200904939A patent/ZA200904939B/xx unknown
- 2009-07-16 IL IL199924A patent/IL199924A0/en unknown
- 2009-07-21 CO CO09075419A patent/CO6150128A2/es unknown
- 2009-08-05 EC EC2009009561A patent/ECSP099561A/es unknown
- 2009-12-04 TN TNP2009000510A patent/TN2009000510A1/fr unknown
- 2009-12-07 MA MA32400A patent/MA31430B1/fr unknown
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5417662B2 (ja) | ネビラピンの徐放性製剤 | |
| WO2011053504A1 (en) | Solid pharmaceutical compositions containing an integrase inhibitor | |
| US20090088424A1 (en) | Methods and compositions for controlling the bioavailability of poorly soluble drugs | |
| WO2006134610A1 (en) | Efavirenz pharmaceutical composition having enhanced dissolution profile | |
| JP2021518422A (ja) | レナリドミドを含む医薬組成物 | |
| CN110913843B (zh) | 药物组合物 | |
| WO2020122244A1 (ja) | 錠剤及びその製造方法 | |
| RU2603469C2 (ru) | Таблетка с контролируемым высвобождением для перорального введения и способ ее приготовления | |
| TWI595871B (zh) | 含有兒茶酚-o-甲基轉移酶抑制劑之口服醫藥組成物及其製造方法 | |
| EP3900708A1 (en) | Extended-release medical composition containing zaltoprofen | |
| EP3335703A1 (en) | Pharmaceutical composition comprising omarigliptin | |
| HK40069975A (en) | Solid pharmaceutical compositions containing an integrase inhibitor | |
| CN119546279A (zh) | Nor-UDCA的药物组合物 | |
| HK1145806B (en) | Extended release formulation of nevirapine | |
| WO2023128898A1 (en) | Pharmaceutical compositions comprising macitentan as active ingredient and other relevant excipients | |
| KR20240141813A (ko) | 경구고형제제 | |
| WO2013100876A1 (en) | Risperidone formulations | |
| CN111759850A (zh) | 草酸替诺福韦的药物组合物 | |
| WO2021253681A1 (zh) | 一种法匹拉韦组合物及其制备方法 | |
| EP3383402A1 (en) | Pharmaceutical composition comprising darunavir and method for the preparation thereof | |
| NZ760868B2 (en) | A solid oral fixed dose composition comprising metformin, valsartan and atorvastatin |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20120717 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20121016 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20121023 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20130117 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20130325 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20130624 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20130701 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20130725 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20131028 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20131101 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 5417662 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |