JP4215912B2 - Thermal shock resistant UV blocking glass and UV blocking filter - Google Patents
Thermal shock resistant UV blocking glass and UV blocking filter Download PDFInfo
- Publication number
- JP4215912B2 JP4215912B2 JP30282699A JP30282699A JP4215912B2 JP 4215912 B2 JP4215912 B2 JP 4215912B2 JP 30282699 A JP30282699 A JP 30282699A JP 30282699 A JP30282699 A JP 30282699A JP 4215912 B2 JP4215912 B2 JP 4215912B2
- Authority
- JP
- Japan
- Prior art keywords
- glass
- ultraviolet
- blocking
- transmittance
- thermal shock
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 239000011521 glass Substances 0.000 title claims description 36
- 230000000903 blocking effect Effects 0.000 title claims description 26
- 230000035939 shock Effects 0.000 title claims description 13
- 238000002834 transmittance Methods 0.000 claims description 24
- 229910004298 SiO 2 Inorganic materials 0.000 claims description 4
- 229910018072 Al 2 O 3 Inorganic materials 0.000 claims description 3
- 238000005286 illumination Methods 0.000 claims description 3
- 238000012216 screening Methods 0.000 claims 1
- 239000005388 borosilicate glass Substances 0.000 description 24
- 239000000126 substance Substances 0.000 description 13
- 239000000203 mixture Substances 0.000 description 7
- 230000003595 spectral effect Effects 0.000 description 4
- 239000011358 absorbing material Substances 0.000 description 3
- 238000004040 coloring Methods 0.000 description 3
- 230000007423 decrease Effects 0.000 description 3
- 230000009931 harmful effect Effects 0.000 description 3
- 238000004519 manufacturing process Methods 0.000 description 3
- 229910010413 TiO 2 Inorganic materials 0.000 description 2
- 239000003513 alkali Substances 0.000 description 2
- 238000007796 conventional method Methods 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 238000010828 elution Methods 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- 239000006096 absorbing agent Substances 0.000 description 1
- 230000004888 barrier function Effects 0.000 description 1
- 239000006121 base glass Substances 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 230000000052 comparative effect Effects 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 238000005520 cutting process Methods 0.000 description 1
- 230000002542 deteriorative effect Effects 0.000 description 1
- 238000004031 devitrification Methods 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- 230000008018 melting Effects 0.000 description 1
- QSHDDOUJBYECFT-UHFFFAOYSA-N mercury Chemical compound [Hg] QSHDDOUJBYECFT-UHFFFAOYSA-N 0.000 description 1
- 229910052753 mercury Inorganic materials 0.000 description 1
- 238000000034 method Methods 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C03—GLASS; MINERAL OR SLAG WOOL
- C03C—CHEMICAL COMPOSITION OF GLASSES, GLAZES OR VITREOUS ENAMELS; SURFACE TREATMENT OF GLASS; SURFACE TREATMENT OF FIBRES OR FILAMENTS MADE FROM GLASS, MINERALS OR SLAGS; JOINING GLASS TO GLASS OR OTHER MATERIALS
- C03C3/00—Glass compositions
- C03C3/04—Glass compositions containing silica
- C03C3/076—Glass compositions containing silica with 40% to 90% silica, by weight
- C03C3/095—Glass compositions containing silica with 40% to 90% silica, by weight containing rare earths
-
- C—CHEMISTRY; METALLURGY
- C03—GLASS; MINERAL OR SLAG WOOL
- C03C—CHEMICAL COMPOSITION OF GLASSES, GLAZES OR VITREOUS ENAMELS; SURFACE TREATMENT OF GLASS; SURFACE TREATMENT OF FIBRES OR FILAMENTS MADE FROM GLASS, MINERALS OR SLAGS; JOINING GLASS TO GLASS OR OTHER MATERIALS
- C03C4/00—Compositions for glass with special properties
- C03C4/08—Compositions for glass with special properties for glass selectively absorbing radiation of specified wave lengths
- C03C4/085—Compositions for glass with special properties for glass selectively absorbing radiation of specified wave lengths for ultraviolet absorbing glass
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Geochemistry & Mineralogy (AREA)
- Materials Engineering (AREA)
- Organic Chemistry (AREA)
- Glass Compositions (AREA)
- Optical Filters (AREA)
Description
【0001】
【発明の属する技術分野】
本発明は、耐熱衝撃性に優れた紫外線遮断ガラスおよび光源として放電灯を用いた照明装置のための紫外線遮断フィルターに関するものである。
【0002】
なお、この明細書では、紫外線遮断能が利用されるガラス製品はその主たる機能や形状に関係なく紫外線遮断フィルターという。
【0003】
【従来の技術】
水銀灯など、紫外線放射を伴うランプからの光を照明に使用するには、ランプが発する光の中の有害紫外線を遮断し可視光線だけを透過させるためのフィルターまたはそのようなフィルターとしても機能するランプ保護部材が必要である。
【0004】
紫外線遮断用のフィルターとしては用途に応じて様々なタイプのものが使われているが、照明装置用のものはランプの発熱による温度上昇が激しく、また使用環境も屋外の場合が多く使用中に雨水などで濡れたりして急冷されることが多いから、軟化温度が高いだけでなく急加熱・急冷によっても割れたりしない耐熱衝撃性にも優れたガラスを基礎ガラスとしそれに耐熱性と耐久性のよい無機質紫外線吸収物質を含有させたものでなければならない。
【0005】
耐熱衝撃性の指標となる熱膨張係数が小さく化学的耐久性にも優れているため上記用途の紫外線遮断ガラスの基礎ガラスとして使われるものの代表的な例は、いわゆるホウケイ酸ガラス(Na2O−B2O3−SiO2系ガラス)であり、これに添加される紫外線吸収物質の例としては、CeO2、Fe2O3、V2O5、TiO2等がある(特公昭61−24344号,特開平6−56467号,特開平8−12369号等)。
【0006】
しかしながら、ホウケイ酸ガラスを基礎ガラスとして紫外線遮断用ガラスを製造する場合の解決困難な課題の一つは、添加する紫外線吸収物質による可視光線透過率の低下を防止することである。すなわち、紫外線遮断に十分な量の紫外線吸収物質を含有させたホウケイ酸ガラスはしばしば波長400nm以上の可視光線も遮断するようになり、それにより、フィルターとして通常使われる厚さのものを目で見てもわかるほど黄色に着色してしまうが、汎用照明装置のためのフィルターは事実上無色透明であることが望ましいから、紫外線吸収物質による可視光線領域の透過率低下は極力少なくしなければならない。
【0007】
この課題を解決するため、特開平6−56467号や特開平8−12369号の発明では限定された量の複数の紫外線吸収物質を併用しているが、シャープカット性に問題が残り、したがって、このような手段を採用したとしても着色回避を優先させると近紫外領域における紫外線遮断能はある程度犠牲にせざるを得なかった。
【0008】
【発明が解決しようとする課題】
本発明の目的は、ホウケイ酸ガラスと同等の低い熱膨張係数および高度の化学的耐久性を備え、シャープ且つ十分な紫外線遮断能を有し、実質的に無着色の紫外線遮断ガラスを提供することにある。
【0009】
本発明の他の目的は、上記紫外線遮断ガラスからなる実質的に無着色の高性能紫外線遮断フィルターを提供することにある。
【0010】
【課題を解決するための手段】
本発明は、ホウケイ酸ガラスに紫外線吸収物質としてCeO2を含有させる場合においてホウケイ酸ガラスの主要構成成分であるB2O3の一部をSb2O3で置換すると、透過光と非透過光の境界波長が360〜380nm付近にあってシャープカット性に優れた実質的に無着色のガラスが得られ、しかも熱膨張係数はホウケイ酸ガラスとほとんど変わらず、化学的耐久性はホウケイ酸ガラスに勝るとも劣らない紫外線遮断ガラスが得られるという、新規な知見に基づき完成されたものである。
【0011】
すなわち、本発明は、SiO2 65〜75%、Na2O 5〜10%、ZnO 0〜3%、Al2O3 1.5〜3%、CeO2 0.5〜2.5%、B2O3 0〜10%、及びB2O3との合計量が15〜25%になる量のSb2O3を含有し、それにより波長320nm以下の紫外線の透過率が0%、波長360nmの近紫外線透過率が50%以下、波長400nmの可視光線の透過率が72%以上であり、且つ熱膨張係数が60×10−7/℃以下であることを特徴とする実質的に無色の耐熱衝撃性紫外線遮断ガラス、及び、該紫外線遮断ガラスよりなる放電灯使用照明装置用紫外線遮断フィルターを提供するものである(但し、ガラス組成を表す%は重量%であり透過率は厚さ3mmの板について測定される値。以下同じ。)。
【0012】
【発明の実施の形態】
上記組成の本発明による紫外線遮断ガラスは、CeO2を含有させたホウケイ酸ガラスに類似するがホウケイ酸ガラスに特徴的な多量のB2O3のかなりの部分がSb2O3で置換されており、しかもSb2O3はガラスのネットワーク構成成分になると考えられるから、もはやホウケイ酸ガラスの範疇には属しないものと言える。しかしながら、製法上は特に困難な点はなく、一般的なホウケイ酸ガラスを製造する場合の常法に従って容易に製造することができる。
【0013】
構成成分について説明すると、まずSiO2は65〜75%とするが、75%以上ではガラスの溶融温度が高くなりすぎるので好ましくなく、また65%以下では熱膨張係数が大きくなるほか構造的に弱くなり、化学的耐久性も悪化し、失透を起こしやすくなる。
【0014】
Na2Oは、少なすぎるとガラスの粘度が高くなり溶解性が悪くなるが、多すぎると熱膨張係数を大きくするので、5〜10%にする。
【0015】
上記2成分と共に基材ガラスの主要構成成分であるB2O3は、典型的なホウケイ酸ガラスの場合よりも少なく、10%以下、望ましくは3〜10%とする。これよりも多くすると、Sb2O3を添加してもシャープな紫外線遮断特性が得られず、また黄色に着色するようになる。
【0016】
Sb2O3は、CeO2を含まないホウケイ酸ガラスに添加した場合、紫外線および可視光線の透過率に影響を及ぼすことはほとんどない。しかし、CeO2添加ホウケイ酸ガラスの場合は、Sb2O3を添加すると波長約350nm以上の近紫外から可視光線にわたる広い波長範囲で透過率が顕著に向上し、それにより、CeO2による紫外線遮断のシャープカット性が向上すると共に着色も事実上解消する。その理由としては、添加されたSb2O3により黄色のCe4+が無色のCe3+に変わることが考えられるが、確認されているわけではない。可視光線と共に波長約350nm以上の近紫外線の透過率も増加させることは紫外線の完全遮断という観点からは不利に見えるが、この波長範囲の紫外線は有効に利用できる場合があり(特開平8−12369号)、有害作用も弱い。また、有害度が高い波長320nm以下の紫外線の透過率は全く上昇させないので、実用上問題になることはない。
【0017】
Sb2O3添加に基づく上記効果は、約5%以上のSb2O3により現れるが添加量20%前後で飽和する。なお、Sb2O3添加量の増加に伴い、波長350〜380nm付近の分光透過率曲線は若干長波長側へシフトする。
【0018】
Sb2O3の添加はまた化学的耐久性の向上にも役立ち、ホウケイ酸ガラスよりも化学的耐久性に優れたガラスを与える。
【0019】
なお、ホウケイ酸ガラスにCeO2とSb2O3を添加しただけではガラスの熱膨張係数が大きくなり、耐熱衝撃性が悪化するが、それは、Sb2O3添加量に応じてB2O3の量を減じ、Sb2O3とB2O3の合計量が15〜25%(好ましくは約17〜20%)の範囲内にあるようにすればほぼ完全に防止することができ、熱膨張係数が60×10−7/℃以下のものを容易に得ることができる(一般的なホウケイ酸ガラスの熱膨張係数は約50×10−7/℃である)。
【0020】
ZnOとAl2O3は紫外線遮断能とは関係がないが、少量を配合するとガラスの化学的耐久性を向上させるのに有効である。
【0021】
紫外線遮断のための主剤であるCeO2は約0.5〜1.6%が適量であり、多くても2.5%程度とする。これよりも多くすると、Sb2O3を多量に添加しても可視光線領域に及ぶ透過率低下が大きくなり、フィルターとして使用する場合の板厚が大きい場合には着色が目立つようになる。
【0022】
上述の組成になるように、かつガラス製造の常法に従って選択され調製された原料混合物を加熱して溶融状態にすることによりガラス化させれば、波長320nm以下の全紫外線の透過率が0%、波長360nmの近紫外線透過率が50%以下、波長400nmの可視光線の透過率が72%以上、熱膨張係数が60×10−7/℃以下の、実質的に無色である耐熱衝撃性紫外線遮断ガラスを得ることができる。
【0023】
これを任意の方法で円板状その他ランプやフィルターを製造するのに必要な形状に成形し、必要に応じて研磨すれば、高度の耐熱衝撃性を有する実質的に無色の紫外線遮断フィルターを得ることができる。
【0024】
【実施例】
次に具体例を示して本発明を説明する。
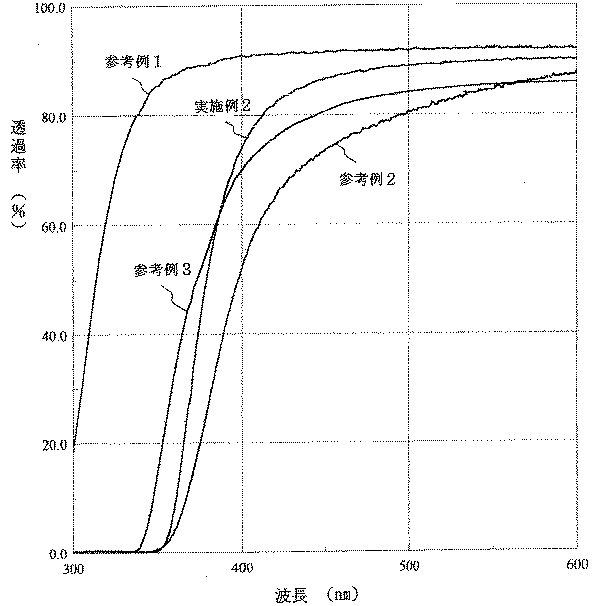
表1に示した組成の標準的なホウケイ酸ガラス(参考例1)およびその組成の一部を変えると共にCeO2を含有させた8種類の紫外線遮断ガラス(比較例1〜3,実施例1〜5)を製造した。得られたガラスおよび参考例1のホウケイ酸ガラスについて熱膨張係数およびアルカリ溶出量を測定し、さらに、波長300以上の紫外線および可視光線領域の分光透過率の測定を行なった。なお、熱膨張係数はJISR3102に、またアルカリ溶出量はJIS R3502に、それぞれ規定されている方法により測定した。その結果を表1に示す。全可視光線透過率は波長380〜780nmの範囲の平均透過率である。
【0025】
また、参考例1のホウケイ酸ガラスを基礎ガラスとしそれに紫外線吸収物質を含有させた下記参考例2,3のガラス、および、上記参考例1と実施例2について測定された分光透過率曲線を図1に示した。
参考例2:CeO2 1.4%を含有させたホウケイ酸ガラス
参考例3:CeO2 0.8%およびTiO2 0.1%を含有させたホウケイ酸ガラス
【0026】
【表1】
【0027】
【発明の効果】
ホウケイ酸ガラスに紫外線吸収物質としてCeO2を含有させる場合にガラスの主要構成成分であるB2O3の一部をSb2O3で置換する本発明によれば、ホウケイ酸ガラスの特長である耐熱衝撃性を悪化させることなしに、シャープ且つ十分な紫外線遮断能と高度の化学的耐久性を備えた実質的に無着色の紫外線遮断ガラスを提供することができる。
【図面の簡単な説明】
【図1】 参考例1〜3および実施例2のガラスの分光透過率曲線である。[0001]
BACKGROUND OF THE INVENTION
The present invention relates to an ultraviolet shielding glass having excellent thermal shock resistance and an ultraviolet shielding filter for an illumination device using a discharge lamp as a light source.
[0002]
In this specification, a glass product using the ultraviolet blocking ability is referred to as an ultraviolet blocking filter regardless of its main function and shape.
[0003]
[Prior art]
In order to use light from a lamp with ultraviolet radiation, such as a mercury lamp, for illumination, a filter that blocks harmful UV rays in the light emitted by the lamp and transmits only visible light, or a lamp that also functions as such a filter. A protective member is required.
[0004]
Various types of UV blocking filters are used depending on the application, but those for lighting devices have a severe temperature rise due to the heat generated by the lamp and are often used outdoors. Since it is often quickly cooled by getting wet with rainwater, etc., it is not only high in softening temperature, but also has excellent thermal shock resistance that does not break due to rapid heating / cooling. Must contain a good inorganic UV absorber.
[0005]
A typical example of what is used as the basic glass of the ultraviolet blocking glass for the above-mentioned uses because it has a small thermal expansion coefficient as an index of thermal shock resistance and excellent chemical durability is so-called borosilicate glass (Na 2 O— a B 2 O 3 -SiO 2 -based glass), examples of the ultraviolet absorbing material added thereto, there is CeO 2, Fe 2 O 3, V 2 O 5, TiO 2 , etc. (JP-B-61-24344 No. 6, JP-A-6-56467, JP-A-8-12369, etc.).
[0006]
However, one of the problems that are difficult to solve in the case of producing an ultraviolet blocking glass using borosilicate glass as a basic glass is to prevent a decrease in visible light transmittance due to an ultraviolet absorbing material to be added. In other words, borosilicate glass containing a sufficient amount of UV-absorbing substance for blocking ultraviolet rays often blocks visible light having a wavelength of 400 nm or more, so that the thickness of the filter usually used can be visually observed. However, since it is desirable that the filter for the general-purpose lighting device is practically colorless and transparent, a decrease in transmittance in the visible light region due to the ultraviolet absorbing material must be minimized.
[0007]
In order to solve this problem, in the inventions of JP-A-6-56467 and JP-A-8-12369, a limited amount of a plurality of ultraviolet absorbing substances are used in combination, but there remains a problem in sharp-cut properties. Even if such a means is adopted, if priority is given to avoiding coloring, the ultraviolet blocking ability in the near ultraviolet region has to be sacrificed to some extent.
[0008]
[Problems to be solved by the invention]
An object of the present invention is to provide a substantially uncolored UV-blocking glass having a low thermal expansion coefficient and a high degree of chemical durability equivalent to those of borosilicate glass, having a sharp and sufficient UV-blocking ability. It is in.
[0009]
Another object of the present invention is to provide a substantially uncolored high performance UV blocking filter comprising the above UV blocking glass.
[0010]
[Means for Solving the Problems]
In the present invention, when CeO 2 is contained in the borosilicate glass as an ultraviolet absorbing substance, when a part of B 2 O 3 which is a main component of the borosilicate glass is replaced with Sb 2 O 3 , transmitted light and non-transmitted light are obtained. A boundary-free wavelength of about 360 to 380 nm is obtained, and a substantially uncolored glass excellent in sharp-cut property is obtained. Moreover, the thermal expansion coefficient is almost the same as that of borosilicate glass, and chemical durability is similar to that of borosilicate glass. It was completed on the basis of a novel finding that an ultraviolet blocking glass that is as good as it can be obtained.
[0011]
That is, the present invention, SiO 2 65~75%, Na 2 O 5~10%, 0~3% ZnO, Al 2 O 3 1.5~3%, CeO 2 0.5~2.5%, B 2 O 3 0 to 10%, and the total amount of B 2 O 3 is 15 to 25% of Sb 2 O 3 is contained, whereby the transmittance of ultraviolet rays with a wavelength of 320 nm or less is 0%, wavelength 360 nm The composition is substantially colorless, characterized by having a near-ultraviolet transmittance of 50% or less, a transmittance of visible light having a wavelength of 400 nm of 72% or more, and a thermal expansion coefficient of 60 × 10 −7 / ° C. or less. The present invention provides a heat-resistant shock-resistant UV-blocking glass and an UV-blocking filter for a lighting device using a discharge lamp comprising the UV-blocking glass (provided that% representing glass composition is% by weight and transmittance is 3 mm thick). The value measured for the plate, and so on. .
[0012]
DETAILED DESCRIPTION OF THE INVENTION
The ultraviolet blocking glass according to the present invention having the above composition is similar to a borosilicate glass containing CeO 2 , but a large part of a large amount of B 2 O 3 characteristic of the borosilicate glass is substituted with Sb 2 O 3. In addition, since Sb 2 O 3 is considered to be a glass network component, it can be said that it no longer belongs to the category of borosilicate glass. However, there is no particular difficulty in the production method, and it can be easily produced according to a conventional method for producing a general borosilicate glass.
[0013]
The constituent components will be described. First, SiO 2 is set to 65 to 75%. However, if it is 75% or more, the melting temperature of the glass becomes too high, which is not preferable, and if it is 65% or less, the thermal expansion coefficient is increased and the structure is weak. Therefore, chemical durability is deteriorated and devitrification is likely to occur.
[0014]
If the Na 2 O content is too small, the viscosity of the glass increases and the solubility becomes poor.
[0015]
B 2 O 3 which is the main component of the base glass together with the above two components is less than that of a typical borosilicate glass, and is 10% or less, preferably 3 to 10%. If it is more than this, even if Sb 2 O 3 is added, no sharp ultraviolet blocking property can be obtained, and it will be colored yellow.
[0016]
When Sb 2 O 3 is added to a borosilicate glass not containing CeO 2 , it hardly affects the transmittance of ultraviolet rays and visible light. However, in the case of CeO 2 -added borosilicate glass, when Sb 2 O 3 is added, the transmittance is remarkably improved in a wide wavelength range from near ultraviolet to visible light having a wavelength of about 350 nm or more, thereby blocking ultraviolet rays by CeO 2. As well as improving the sharp cutting property, the coloring is virtually eliminated. The reason may be that yellow Ce 4+ is changed to colorless Ce 3+ by the added Sb 2 O 3 , but it is not confirmed. Increasing the transmittance of near-ultraviolet light having a wavelength of about 350 nm or more together with visible light seems disadvantageous from the viewpoint of complete blocking of ultraviolet light, but there are cases where ultraviolet light in this wavelength range can be used effectively (Japanese Patent Laid-Open No. 8-12369). No.), harmful effects are also weak. Further, since the transmittance of ultraviolet rays having a wavelength of 320 nm or less, which is highly harmful, is not increased at all, there is no practical problem.
[0017]
Said based on Sb 2 O 3 addition effect is manifested by Sb 2 O 3 of more than about 5% saturated with amount around 20%. As the Sb 2 O 3 addition amount increases, the spectral transmittance curve near the wavelength of 350 to 380 nm slightly shifts to the longer wavelength side.
[0018]
The addition of Sb 2 O 3 also helps to improve chemical durability and gives a glass with better chemical durability than borosilicate glass.
[0019]
Incidentally, only the addition of CeO 2 and Sb 2 O 3 in the borosilicate glass becomes large, the thermal expansion coefficient of the glass, but the thermal shock resistance is deteriorated, it is, B 2 O 3 in accordance with Sb 2 O 3 added amount If the total amount of Sb 2 O 3 and B 2 O 3 is in the range of 15 to 25% (preferably about 17 to 20%), it can be almost completely prevented, Those having an expansion coefficient of 60 × 10 −7 / ° C. or less can be easily obtained (the thermal expansion coefficient of general borosilicate glass is about 50 × 10 −7 / ° C.).
[0020]
ZnO and Al 2 O 3 are not related to the ultraviolet blocking ability, but if a small amount is added, it is effective to improve the chemical durability of the glass.
[0021]
An appropriate amount of CeO 2 which is the main agent for blocking ultraviolet rays is about 0.5 to 1.6%, and at most about 2.5%. If the amount is larger than this, even if a large amount of Sb 2 O 3 is added, the transmittance decreases over the visible light region, and coloring becomes conspicuous when the plate thickness when used as a filter is large.
[0022]
When the raw material mixture selected and prepared according to the conventional method for glass production is vitrified by heating to a molten state so as to have the above composition, the transmittance of all ultraviolet rays having a wavelength of 320 nm or less is 0%. A substantially colorless thermal shock resistant ultraviolet ray having a near-ultraviolet ray transmittance at a wavelength of 360 nm of 50% or less, a visible light transmittance at a wavelength of 400 nm of 72% or more, and a thermal expansion coefficient of 60 × 10 −7 / ° C. or less. A barrier glass can be obtained.
[0023]
If this is formed into a disk shape or any other shape necessary for manufacturing lamps and filters by any method, and polished as necessary, a substantially colorless UV blocking filter having high thermal shock resistance can be obtained. be able to.
[0024]
【Example】
Next, the present invention will be described with reference to specific examples.
Standard borosilicate glass (Reference Example 1) having the composition shown in Table 1 and eight kinds of ultraviolet blocking glasses (Comparative Examples 1 to 3 and Examples 1 to 1) containing CeO 2 while changing part of the composition. 5) was produced. The obtained glass and the borosilicate glass of Reference Example 1 were measured for thermal expansion coefficient and alkali elution amount, and further measured for spectral transmittance in the ultraviolet and visible light regions having a wavelength of 300 or more. The thermal expansion coefficient was measured according to JIS R3102, and the alkali elution amount was measured according to JIS R3502, respectively. The results are shown in Table 1. The total visible light transmittance is an average transmittance in a wavelength range of 380 to 780 nm.
[0025]
Moreover, the spectral transmittance curve measured about the glass of the following reference examples 2 and 3 which made the borosilicate glass of the reference example 1 the basic glass and contained the ultraviolet-absorbing substance, and the said reference example 1 and Example 2 is shown. It was shown in 1.
Reference Example 2: Borosilicate glass containing 1.4% CeO 2 Reference Example 3: Borosilicate glass containing 0.8% CeO 2 and 0.1% TiO 2
[Table 1]
[0027]
【The invention's effect】
According to the present invention, a part of B 2 O 3 which is a main component of glass is substituted with Sb 2 O 3 when CeO 2 is contained in the borosilicate glass as an ultraviolet absorbing substance. Without deteriorating the thermal shock resistance, it is possible to provide a substantially non-colored UV blocking glass having a sharp and sufficient UV blocking ability and a high chemical durability.
[Brief description of the drawings]
1 is a spectral transmittance curve of glasses of Reference Examples 1 to 3 and Example 2. FIG.
Claims (5)
65〜75重量%、Na2O 5〜10重量%、ZnO 0〜3重量%、Al2O3 1.5〜3重量%、CeO2
0.5〜2.5重量%、B2O3 0〜10重量%、及びB2O3との合計量が15〜25重量%になる量のSb2O3
を含有し、それにより波長320nm以下の紫外線の透過率が0%、波長360nmの近紫外線透過率が50%以下、波長400nmの可視光線の透過率が72%以上であり、且つ熱膨張係数が60×10−7/℃以下であることを特徴とする実質的に無色の耐熱衝撃性紫外線遮断ガラス(但し透過率は厚さ3mmの板について測定される値)。SiO 2
65-75 wt%, Na 2 O 5~10 wt%, ZnO 0 to 3 wt%, Al 2 O 3 1.5~3 wt%, CeO 2
0.5-2.5 wt%, B 2 O 3 0 wt%, and B 2 O 3 the total amount of is 15 to 25 wt% amount of Sb 2 O 3
Therefore, the transmittance of ultraviolet light having a wavelength of 320 nm or less is 0%, the transmittance of near ultraviolet light having a wavelength of 360 nm is 50% or less, the transmittance of visible light having a wavelength of 400 nm is 72% or more, and the thermal expansion coefficient is A substantially colorless thermal shock-resistant UV-blocking glass characterized in that it is 60 × 10 −7 / ° C. or less (however, the transmittance is a value measured for a plate having a thickness of 3 mm).
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP30282699A JP4215912B2 (en) | 1999-09-20 | 1999-09-20 | Thermal shock resistant UV blocking glass and UV blocking filter |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP30282699A JP4215912B2 (en) | 1999-09-20 | 1999-09-20 | Thermal shock resistant UV blocking glass and UV blocking filter |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2001089185A JP2001089185A (en) | 2001-04-03 |
| JP4215912B2 true JP4215912B2 (en) | 2009-01-28 |
Family
ID=17913569
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP30282699A Expired - Fee Related JP4215912B2 (en) | 1999-09-20 | 1999-09-20 | Thermal shock resistant UV blocking glass and UV blocking filter |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP4215912B2 (en) |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP4104142B2 (en) * | 2003-08-29 | 2008-06-18 | Hoya株式会社 | Sharp cut filter glass and sharp cut filter |
| KR102558749B1 (en) | 2017-03-31 | 2023-07-24 | 코닝 인코포레이티드 | high transmittance glass |
| WO2020184216A1 (en) * | 2019-03-08 | 2020-09-17 | 日本電気硝子株式会社 | Wavelength-conversion member and light-emitting device |
| CN113045199B (en) * | 2021-03-23 | 2022-04-15 | 成都光明光电股份有限公司 | Ultraviolet-transmitting glass |
-
1999
- 1999-09-20 JP JP30282699A patent/JP4215912B2/en not_active Expired - Fee Related
Also Published As
| Publication number | Publication date |
|---|---|
| JP2001089185A (en) | 2001-04-03 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CA2239342C (en) | Lead and arsenic free borosilicate glass and lamp containing same | |
| KR100254933B1 (en) | UV absorbing glass and its manufacturing method | |
| EP0522859B1 (en) | Colored glass | |
| JP2528351B2 (en) | UV transparent glass and its batch composition | |
| JP2005529048A (en) | UV-shielding borosilicate glass, its use, and fluorescent lamp | |
| MXPA00006579A (en) | Neodymium glass for tungsten-halogen lamp envelopes and filters. | |
| JPH0812369A (en) | Glass for hid lamp | |
| JP4215912B2 (en) | Thermal shock resistant UV blocking glass and UV blocking filter | |
| US5213440A (en) | Method of making yellow transparent tempered glass and glass product | |
| JP4412747B2 (en) | Lead-free crown glass | |
| JPH0656467A (en) | Ultraviolet light absorbing glass | |
| JPH0616452A (en) | Front glass for hid lamp | |
| KR19990087157A (en) | Method for preparing UV absorbing solution | |
| JP2004123525A (en) | Borosilicate glass and its use | |
| CN1625532A (en) | Alkaline-earth aluminosilicate glass and its application | |
| JP2002173338A (en) | Front glass for illumination | |
| JP4237920B2 (en) | UV blocking glass | |
| JP3749276B2 (en) | Infrared transmission glass | |
| JP4104142B2 (en) | Sharp cut filter glass and sharp cut filter | |
| CN110278930A (en) | A kind of unleaded quartz burner of outdoor trap lamp tool | |
| JP3255664B2 (en) | Lighting equipment | |
| RU2280625C1 (en) | Color filter glass | |
| JPS6124344B2 (en) | ||
| JPH0834633A (en) | Optical glass free from solarization | |
| KR20250009685A (en) | Ultra high refractive index glass beads using the ultra high refractive index glass frit and manufacturing method of the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| RD01 | Notification of change of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7421 Effective date: 20060217 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20060316 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20080722 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20080827 |
|
| A521 | Written amendment |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20080911 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20081030 |
|
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20081105 |
|
| R150 | Certificate of patent or registration of utility model |
Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20111114 Year of fee payment: 3 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20111114 Year of fee payment: 3 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20121114 Year of fee payment: 4 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20121114 Year of fee payment: 4 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20121114 Year of fee payment: 4 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20131114 Year of fee payment: 5 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| LAPS | Cancellation because of no payment of annual fees |