JP3644809B2 - External preparation for head - Google Patents
External preparation for head Download PDFInfo
- Publication number
- JP3644809B2 JP3644809B2 JP29789697A JP29789697A JP3644809B2 JP 3644809 B2 JP3644809 B2 JP 3644809B2 JP 29789697 A JP29789697 A JP 29789697A JP 29789697 A JP29789697 A JP 29789697A JP 3644809 B2 JP3644809 B2 JP 3644809B2
- Authority
- JP
- Japan
- Prior art keywords
- hair
- aminolevulinic acid
- head
- external preparation
- hair growth
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- 238000002360 preparation method Methods 0.000 title claims description 45
- 229960002749 aminolevulinic acid Drugs 0.000 claims description 46
- ZGXJTSGNIOSYLO-UHFFFAOYSA-N 88755TAZ87 Chemical compound NCC(=O)CCC(O)=O ZGXJTSGNIOSYLO-UHFFFAOYSA-N 0.000 claims description 45
- 230000003779 hair growth Effects 0.000 claims description 38
- -1 5-aminolevulinic acid ester Chemical class 0.000 claims description 22
- 239000003795 chemical substances by application Substances 0.000 claims description 16
- 239000007952 growth promoter Substances 0.000 claims description 12
- 150000003839 salts Chemical class 0.000 claims description 11
- 239000004615 ingredient Substances 0.000 claims description 9
- 150000002148 esters Chemical group 0.000 claims description 6
- 125000004432 carbon atom Chemical group C* 0.000 claims description 3
- 125000006165 cyclic alkyl group Chemical group 0.000 claims 1
- 210000003128 head Anatomy 0.000 description 33
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 31
- 230000000694 effects Effects 0.000 description 20
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 16
- 239000012071 phase Substances 0.000 description 16
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 14
- 239000004480 active ingredient Substances 0.000 description 11
- 230000001737 promoting effect Effects 0.000 description 11
- 238000004519 manufacturing process Methods 0.000 description 10
- 238000002156 mixing Methods 0.000 description 9
- 238000013329 compounding Methods 0.000 description 8
- 235000011187 glycerol Nutrition 0.000 description 8
- 238000000034 method Methods 0.000 description 7
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 6
- 238000005342 ion exchange Methods 0.000 description 6
- NOOLISFMXDJSKH-KXUCPTDWSA-N (-)-Menthol Chemical compound CC(C)[C@@H]1CC[C@@H](C)C[C@H]1O NOOLISFMXDJSKH-KXUCPTDWSA-N 0.000 description 5
- 239000004359 castor oil Substances 0.000 description 5
- 235000019438 castor oil Nutrition 0.000 description 5
- ZEMPKEQAKRGZGQ-XOQCFJPHSA-N glycerol triricinoleate Natural products CCCCCC[C@@H](O)CC=CCCCCCCCC(=O)OC[C@@H](COC(=O)CCCCCCCC=CC[C@@H](O)CCCCCC)OC(=O)CCCCCCCC=CC[C@H](O)CCCCCC ZEMPKEQAKRGZGQ-XOQCFJPHSA-N 0.000 description 5
- 230000003660 hair regeneration Effects 0.000 description 5
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 4
- PVNIIMVLHYAWGP-UHFFFAOYSA-N Niacin Chemical compound OC(=O)C1=CC=CN=C1 PVNIIMVLHYAWGP-UHFFFAOYSA-N 0.000 description 4
- 230000009471 action Effects 0.000 description 4
- QELSKZZBTMNZEB-UHFFFAOYSA-N propylparaben Chemical compound CCCOC(=O)C1=CC=C(O)C=C1 QELSKZZBTMNZEB-UHFFFAOYSA-N 0.000 description 4
- 239000000523 sample Substances 0.000 description 4
- PRAKJMSDJKAYCZ-UHFFFAOYSA-N squalane Chemical compound CC(C)CCCC(C)CCCC(C)CCCCC(C)CCCC(C)CCCC(C)C PRAKJMSDJKAYCZ-UHFFFAOYSA-N 0.000 description 4
- 238000003756 stirring Methods 0.000 description 4
- GVJHHUAWPYXKBD-UHFFFAOYSA-N (±)-α-Tocopherol Chemical compound OC1=C(C)C(C)=C2OC(CCCC(C)CCCC(C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-UHFFFAOYSA-N 0.000 description 3
- NOOLISFMXDJSKH-UHFFFAOYSA-N DL-menthol Natural products CC(C)C1CCC(C)CC1O NOOLISFMXDJSKH-UHFFFAOYSA-N 0.000 description 3
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 3
- 230000003698 anagen phase Effects 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 150000001875 compounds Chemical class 0.000 description 3
- 239000002537 cosmetic Substances 0.000 description 3
- 239000006071 cream Substances 0.000 description 3
- 239000000839 emulsion Substances 0.000 description 3
- 239000003205 fragrance Substances 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 229940041616 menthol Drugs 0.000 description 3
- YUUAYBAIHCDHHD-UHFFFAOYSA-N methyl 5-aminolevulinate Chemical compound COC(=O)CCC(=O)CN YUUAYBAIHCDHHD-UHFFFAOYSA-N 0.000 description 3
- 239000000203 mixture Substances 0.000 description 3
- UTTVXKGNTWZECK-UHFFFAOYSA-N n,n-dimethyloctadecan-1-amine oxide Chemical compound CCCCCCCCCCCCCCCCCC[N+](C)(C)[O-] UTTVXKGNTWZECK-UHFFFAOYSA-N 0.000 description 3
- 235000001968 nicotinic acid Nutrition 0.000 description 3
- GLDOVTGHNKAZLK-UHFFFAOYSA-N octadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCCCO GLDOVTGHNKAZLK-UHFFFAOYSA-N 0.000 description 3
- 239000002453 shampoo Substances 0.000 description 3
- YBJHBAHKTGYVGT-ZKWXMUAHSA-N (+)-Biotin Chemical compound N1C(=O)N[C@@H]2[C@H](CCCCC(=O)O)SC[C@@H]21 YBJHBAHKTGYVGT-ZKWXMUAHSA-N 0.000 description 2
- PUPZLCDOIYMWBV-UHFFFAOYSA-N (+/-)-1,3-Butanediol Chemical compound CC(O)CCO PUPZLCDOIYMWBV-UHFFFAOYSA-N 0.000 description 2
- ALSTYHKOOCGGFT-KTKRTIGZSA-N (9Z)-octadecen-1-ol Chemical compound CCCCCCCC\C=C/CCCCCCCCO ALSTYHKOOCGGFT-KTKRTIGZSA-N 0.000 description 2
- GHOKWGTUZJEAQD-ZETCQYMHSA-N (D)-(+)-Pantothenic acid Chemical compound OCC(C)(C)[C@@H](O)C(=O)NCCC(O)=O GHOKWGTUZJEAQD-ZETCQYMHSA-N 0.000 description 2
- VBICKXHEKHSIBG-UHFFFAOYSA-N 1-monostearoylglycerol Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCC(O)CO VBICKXHEKHSIBG-UHFFFAOYSA-N 0.000 description 2
- MPDGHEJMBKOTSU-YKLVYJNSSA-N 18beta-glycyrrhetic acid Chemical compound C([C@H]1C2=CC(=O)[C@H]34)[C@@](C)(C(O)=O)CC[C@]1(C)CC[C@@]2(C)[C@]4(C)CC[C@@H]1[C@]3(C)CC[C@H](O)C1(C)C MPDGHEJMBKOTSU-YKLVYJNSSA-N 0.000 description 2
- 201000004384 Alopecia Diseases 0.000 description 2
- 241000195940 Bryophyta Species 0.000 description 2
- 108010066551 Cholestenone 5 alpha-Reductase Proteins 0.000 description 2
- 208000001840 Dandruff Diseases 0.000 description 2
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 2
- 108090000790 Enzymes Proteins 0.000 description 2
- 102000004190 Enzymes Human genes 0.000 description 2
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 2
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 2
- 241000699666 Mus <mouse, genus> Species 0.000 description 2
- 241000699670 Mus sp. Species 0.000 description 2
- 229920002125 Sokalan® Polymers 0.000 description 2
- VBIIFPGSPJYLRR-UHFFFAOYSA-M Stearyltrimethylammonium chloride Chemical compound [Cl-].CCCCCCCCCCCCCCCCCC[N+](C)(C)C VBIIFPGSPJYLRR-UHFFFAOYSA-M 0.000 description 2
- 125000002252 acyl group Chemical group 0.000 description 2
- 239000000443 aerosol Substances 0.000 description 2
- 230000000844 anti-bacterial effect Effects 0.000 description 2
- 230000003078 antioxidant effect Effects 0.000 description 2
- 239000008346 aqueous phase Substances 0.000 description 2
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 2
- KVYGGMBOZFWZBQ-UHFFFAOYSA-N benzyl nicotinate Chemical compound C=1C=CN=CC=1C(=O)OCC1=CC=CC=C1 KVYGGMBOZFWZBQ-UHFFFAOYSA-N 0.000 description 2
- 125000001584 benzyloxycarbonyl group Chemical group C(=O)(OCC1=CC=CC=C1)* 0.000 description 2
- 230000017531 blood circulation Effects 0.000 description 2
- 229940117173 croton oil Drugs 0.000 description 2
- SZXQTJUDPRGNJN-UHFFFAOYSA-N dipropylene glycol Chemical compound OCCCOCCCO SZXQTJUDPRGNJN-UHFFFAOYSA-N 0.000 description 2
- LYCAIKOWRPUZTN-UHFFFAOYSA-N ethylene glycol Natural products OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 2
- 238000009472 formulation Methods 0.000 description 2
- 230000003676 hair loss Effects 0.000 description 2
- 208000024963 hair loss Diseases 0.000 description 2
- 239000004009 herbicide Substances 0.000 description 2
- 230000006698 induction Effects 0.000 description 2
- 230000005764 inhibitory process Effects 0.000 description 2
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 2
- 229940057995 liquid paraffin Drugs 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 2
- 230000003020 moisturizing effect Effects 0.000 description 2
- 235000011929 mousse Nutrition 0.000 description 2
- JXTPJDDICSTXJX-UHFFFAOYSA-N n-Triacontane Natural products CCCCCCCCCCCCCCCCCCCCCCCCCCCCCC JXTPJDDICSTXJX-UHFFFAOYSA-N 0.000 description 2
- 229960003512 nicotinic acid Drugs 0.000 description 2
- 239000011664 nicotinic acid Substances 0.000 description 2
- 239000003921 oil Substances 0.000 description 2
- 235000019198 oils Nutrition 0.000 description 2
- 229940055577 oleyl alcohol Drugs 0.000 description 2
- XMLQWXUVTXCDDL-UHFFFAOYSA-N oleyl alcohol Natural products CCCCCCC=CCCCCCCCCCCO XMLQWXUVTXCDDL-UHFFFAOYSA-N 0.000 description 2
- 239000005648 plant growth regulator Substances 0.000 description 2
- 230000003658 preventing hair loss Effects 0.000 description 2
- 235000010232 propyl p-hydroxybenzoate Nutrition 0.000 description 2
- 239000004405 propyl p-hydroxybenzoate Substances 0.000 description 2
- 229960003415 propylparaben Drugs 0.000 description 2
- YGSDEFSMJLZEOE-UHFFFAOYSA-N salicylic acid Chemical compound OC(=O)C1=CC=CC=C1O YGSDEFSMJLZEOE-UHFFFAOYSA-N 0.000 description 2
- 229920002545 silicone oil Polymers 0.000 description 2
- 229910052708 sodium Inorganic materials 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- GCLGEJMYGQKIIW-UHFFFAOYSA-H sodium hexametaphosphate Chemical compound [Na]OP1(=O)OP(=O)(O[Na])OP(=O)(O[Na])OP(=O)(O[Na])OP(=O)(O[Na])OP(=O)(O[Na])O1 GCLGEJMYGQKIIW-UHFFFAOYSA-H 0.000 description 2
- 235000019982 sodium hexametaphosphate Nutrition 0.000 description 2
- 239000000243 solution Substances 0.000 description 2
- 239000007921 spray Substances 0.000 description 2
- 229940032094 squalane Drugs 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 125000001424 substituent group Chemical group 0.000 description 2
- 239000000725 suspension Substances 0.000 description 2
- 238000003786 synthesis reaction Methods 0.000 description 2
- 239000001577 tetrasodium phosphonato phosphate Substances 0.000 description 2
- 230000008719 thickening Effects 0.000 description 2
- 230000001256 tonic effect Effects 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- MTCFGRXMJLQNBG-REOHCLBHSA-N (2S)-2-Amino-3-hydroxypropansäure Chemical compound OC[C@H](N)C(O)=O MTCFGRXMJLQNBG-REOHCLBHSA-N 0.000 description 1
- KIUKXJAPPMFGSW-DNGZLQJQSA-N (2S,3S,4S,5R,6R)-6-[(2S,3R,4R,5S,6R)-3-Acetamido-2-[(2S,3S,4R,5R,6R)-6-[(2R,3R,4R,5S,6R)-3-acetamido-2,5-dihydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-2-carboxy-4,5-dihydroxyoxan-3-yl]oxy-5-hydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-3,4,5-trihydroxyoxane-2-carboxylic acid Chemical compound CC(=O)N[C@H]1[C@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@H](O[C@H]2[C@@H]([C@@H](O[C@H]3[C@@H]([C@@H](O)[C@H](O)[C@H](O3)C(O)=O)O)[C@H](O)[C@@H](CO)O2)NC(C)=O)[C@@H](C(O)=O)O1 KIUKXJAPPMFGSW-DNGZLQJQSA-N 0.000 description 1
- RBCOYOYDYNXAFA-UHFFFAOYSA-L (5-hydroxy-4,6-dimethylpyridin-3-yl)methyl phosphate Chemical compound CC1=NC=C(COP([O-])([O-])=O)C(C)=C1O RBCOYOYDYNXAFA-UHFFFAOYSA-L 0.000 description 1
- 229940058015 1,3-butylene glycol Drugs 0.000 description 1
- VOXZDWNPVJITMN-ZBRFXRBCSA-N 17β-estradiol Chemical compound OC1=CC=C2[C@H]3CC[C@](C)([C@H](CC4)O)[C@@H]4[C@@H]3CCC2=C1 VOXZDWNPVJITMN-ZBRFXRBCSA-N 0.000 description 1
- 239000000263 2,3-dihydroxypropyl (Z)-octadec-9-enoate Substances 0.000 description 1
- LBLYYCQCTBFVLH-UHFFFAOYSA-M 2-methylbenzenesulfonate Chemical compound CC1=CC=CC=C1S([O-])(=O)=O LBLYYCQCTBFVLH-UHFFFAOYSA-M 0.000 description 1
- 125000000094 2-phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 description 1
- RZRNAYUHWVFMIP-GDCKJWNLSA-N 3-oleoyl-sn-glycerol Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OC[C@H](O)CO RZRNAYUHWVFMIP-GDCKJWNLSA-N 0.000 description 1
- 125000006201 3-phenylpropyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- CYDQOEWLBCCFJZ-UHFFFAOYSA-N 4-(4-fluorophenyl)oxane-4-carboxylic acid Chemical compound C=1C=C(F)C=CC=1C1(C(=O)O)CCOCC1 CYDQOEWLBCCFJZ-UHFFFAOYSA-N 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 1
- 239000004475 Arginine Substances 0.000 description 1
- FERIUCNNQQJTOY-UHFFFAOYSA-M Butyrate Chemical compound CCCC([O-])=O FERIUCNNQQJTOY-UHFFFAOYSA-M 0.000 description 1
- FERIUCNNQQJTOY-UHFFFAOYSA-N Butyric acid Natural products CCCC(O)=O FERIUCNNQQJTOY-UHFFFAOYSA-N 0.000 description 1
- 208000019300 CLIPPERS Diseases 0.000 description 1
- GHOKWGTUZJEAQD-UHFFFAOYSA-N Chick antidermatitis factor Natural products OCC(C)(C)C(O)C(=O)NCCC(O)=O GHOKWGTUZJEAQD-UHFFFAOYSA-N 0.000 description 1
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 1
- PMATZTZNYRCHOR-CGLBZJNRSA-N Cyclosporin A Chemical class CC[C@@H]1NC(=O)[C@H]([C@H](O)[C@H](C)C\C=C\C)N(C)C(=O)[C@H](C(C)C)N(C)C(=O)[C@H](CC(C)C)N(C)C(=O)[C@H](CC(C)C)N(C)C(=O)[C@@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)N(C)C(=O)[C@H](C(C)C)NC(=O)[C@H](CC(C)C)N(C)C(=O)CN(C)C1=O PMATZTZNYRCHOR-CGLBZJNRSA-N 0.000 description 1
- 235000017788 Cydonia oblonga Nutrition 0.000 description 1
- ZAKOWWREFLAJOT-CEFNRUSXSA-N D-alpha-tocopherylacetate Chemical compound CC(=O)OC1=C(C)C(C)=C2O[C@@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1C ZAKOWWREFLAJOT-CEFNRUSXSA-N 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 1
- 239000004471 Glycine Substances 0.000 description 1
- AEMRFAOFKBGASW-UHFFFAOYSA-M Glycolate Chemical compound OCC([O-])=O AEMRFAOFKBGASW-UHFFFAOYSA-M 0.000 description 1
- MPDGHEJMBKOTSU-UHFFFAOYSA-N Glycyrrhetinsaeure Natural products C12C(=O)C=C3C4CC(C)(C(O)=O)CCC4(C)CCC3(C)C1(C)CCC1C2(C)CCC(O)C1(C)C MPDGHEJMBKOTSU-UHFFFAOYSA-N 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 1
- FFEARJCKVFRZRR-BYPYZUCNSA-N L-methionine Chemical compound CSCC[C@H](N)C(O)=O FFEARJCKVFRZRR-BYPYZUCNSA-N 0.000 description 1
- JVTAAEKCZFNVCJ-UHFFFAOYSA-M Lactate Chemical compound CC(O)C([O-])=O JVTAAEKCZFNVCJ-UHFFFAOYSA-M 0.000 description 1
- 102000003960 Ligases Human genes 0.000 description 1
- 108090000364 Ligases Proteins 0.000 description 1
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 1
- ZFMITUMMTDLWHR-UHFFFAOYSA-N Minoxidil Chemical compound NC1=[N+]([O-])C(N)=CC(N2CCCCC2)=N1 ZFMITUMMTDLWHR-UHFFFAOYSA-N 0.000 description 1
- 229920000715 Mucilage Polymers 0.000 description 1
- AOMUHOFOVNGZAN-UHFFFAOYSA-N N,N-bis(2-hydroxyethyl)dodecanamide Chemical compound CCCCCCCCCCCC(=O)N(CCO)CCO AOMUHOFOVNGZAN-UHFFFAOYSA-N 0.000 description 1
- SUZRRICLUFMAQD-UHFFFAOYSA-N N-Methyltaurine Chemical compound CNCCS(O)(=O)=O SUZRRICLUFMAQD-UHFFFAOYSA-N 0.000 description 1
- 229910002651 NO3 Inorganic materials 0.000 description 1
- NHNBFGGVMKEFGY-UHFFFAOYSA-N Nitrate Chemical compound [O-][N+]([O-])=O NHNBFGGVMKEFGY-UHFFFAOYSA-N 0.000 description 1
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 1
- MTCFGRXMJLQNBG-UHFFFAOYSA-N Serine Natural products OCC(N)C(O)=O MTCFGRXMJLQNBG-UHFFFAOYSA-N 0.000 description 1
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- 229930003427 Vitamin E Natural products 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- DFPAKSUCGFBDDF-ZQBYOMGUSA-N [14c]-nicotinamide Chemical compound N[14C](=O)C1=CC=CN=C1 DFPAKSUCGFBDDF-ZQBYOMGUSA-N 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- 239000006096 absorbing agent Substances 0.000 description 1
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 1
- OIPILFWXSMYKGL-UHFFFAOYSA-N acetylcholine Chemical class CC(=O)OCC[N+](C)(C)C OIPILFWXSMYKGL-UHFFFAOYSA-N 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000032683 aging Effects 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 1
- 125000003545 alkoxy group Chemical group 0.000 description 1
- 125000005210 alkyl ammonium group Chemical group 0.000 description 1
- 125000005907 alkyl ester group Chemical group 0.000 description 1
- 125000000217 alkyl group Chemical group 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- 238000010171 animal model Methods 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- 229940121363 anti-inflammatory agent Drugs 0.000 description 1
- 239000002260 anti-inflammatory agent Substances 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 235000006708 antioxidants Nutrition 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 108010045569 atelocollagen Proteins 0.000 description 1
- 125000003236 benzoyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C(*)=O 0.000 description 1
- 229950004580 benzyl nicotinate Drugs 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 229960002685 biotin Drugs 0.000 description 1
- 235000020958 biotin Nutrition 0.000 description 1
- 239000011616 biotin Substances 0.000 description 1
- 235000019437 butane-1,3-diol Nutrition 0.000 description 1
- 159000000007 calcium salts Chemical class 0.000 description 1
- RZMKWKZIJJNSLQ-UHFFFAOYSA-M carpronium chloride Chemical compound [Cl-].COC(=O)CCC[N+](C)(C)C RZMKWKZIJJNSLQ-UHFFFAOYSA-M 0.000 description 1
- 229950003631 carpronium chloride Drugs 0.000 description 1
- 239000003518 caustics Substances 0.000 description 1
- 229940082500 cetostearyl alcohol Drugs 0.000 description 1
- 208000021930 chronic lymphocytic inflammation with pontine perivascular enhancement responsive to steroids Diseases 0.000 description 1
- 239000013068 control sample Substances 0.000 description 1
- 239000012043 crude product Substances 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- ZAKOWWREFLAJOT-UHFFFAOYSA-N d-alpha-Tocopheryl acetate Natural products CC(=O)OC1=C(C)C(C)=C2OC(CCCC(C)CCCC(C)CCCC(C)C)(C)CCC2=C1C ZAKOWWREFLAJOT-UHFFFAOYSA-N 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- 230000004069 differentiation Effects 0.000 description 1
- GVGUFUZHNYFZLC-UHFFFAOYSA-N dodecyl benzenesulfonate;sodium Chemical compound [Na].CCCCCCCCCCCCOS(=O)(=O)C1=CC=CC=C1 GVGUFUZHNYFZLC-UHFFFAOYSA-N 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 239000003623 enhancer Substances 0.000 description 1
- 229960003720 enoxolone Drugs 0.000 description 1
- 229960005309 estradiol Drugs 0.000 description 1
- 229930182833 estradiol Natural products 0.000 description 1
- 125000005448 ethoxyethyl group Chemical group [H]C([H])([H])C([H])([H])OC([H])([H])C([H])([H])* 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- 238000000855 fermentation Methods 0.000 description 1
- 230000004151 fermentation Effects 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 235000019634 flavors Nutrition 0.000 description 1
- WIGCFUFOHFEKBI-UHFFFAOYSA-N gamma-tocopherol Natural products CC(C)CCCC(C)CCCC(C)CCCC1CCC2C(C)C(O)C(C)C(C)C2O1 WIGCFUFOHFEKBI-UHFFFAOYSA-N 0.000 description 1
- 229960005150 glycerol Drugs 0.000 description 1
- 229940075507 glyceryl monostearate Drugs 0.000 description 1
- 230000012010 growth Effects 0.000 description 1
- 230000031774 hair cycle Effects 0.000 description 1
- 230000002363 herbicidal effect Effects 0.000 description 1
- OIKBVOIOVNEVJR-UHFFFAOYSA-N hexadecyl 6-methylheptanoate Chemical compound CCCCCCCCCCCCCCCCOC(=O)CCCCC(C)C OIKBVOIOVNEVJR-UHFFFAOYSA-N 0.000 description 1
- 239000005556 hormone Substances 0.000 description 1
- 229940088597 hormone Drugs 0.000 description 1
- 239000003906 humectant Substances 0.000 description 1
- 229920002674 hyaluronan Polymers 0.000 description 1
- 229960003160 hyaluronic acid Drugs 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 125000004029 hydroxymethyl group Chemical group [H]OC([H])([H])* 0.000 description 1
- 230000001939 inductive effect Effects 0.000 description 1
- 239000002917 insecticide Substances 0.000 description 1
- 150000002500 ions Chemical group 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 229940001447 lactate Drugs 0.000 description 1
- 239000004310 lactic acid Substances 0.000 description 1
- 235000014655 lactic acid Nutrition 0.000 description 1
- 229940031957 lauric acid diethanolamide Drugs 0.000 description 1
- 229940049920 malate Drugs 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- BJEPYKJPYRNKOW-UHFFFAOYSA-N malic acid Chemical compound OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 1
- 239000000845 maltitol Substances 0.000 description 1
- VQHSOMBJVWLPSR-WUJBLJFYSA-N maltitol Chemical compound OC[C@H](O)[C@@H](O)[C@@H]([C@H](O)CO)O[C@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O VQHSOMBJVWLPSR-WUJBLJFYSA-N 0.000 description 1
- 235000010449 maltitol Nutrition 0.000 description 1
- 229940035436 maltitol Drugs 0.000 description 1
- 230000007721 medicinal effect Effects 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 229930182817 methionine Natural products 0.000 description 1
- 230000000813 microbial effect Effects 0.000 description 1
- 244000005700 microbiome Species 0.000 description 1
- 229960003632 minoxidil Drugs 0.000 description 1
- 239000001788 mono and diglycerides of fatty acids Substances 0.000 description 1
- RZRNAYUHWVFMIP-UHFFFAOYSA-N monoelaidin Natural products CCCCCCCCC=CCCCCCCCC(=O)OCC(O)CO RZRNAYUHWVFMIP-UHFFFAOYSA-N 0.000 description 1
- GOQYKNQRPGWPLP-UHFFFAOYSA-N n-heptadecyl alcohol Natural products CCCCCCCCCCCCCCCCCO GOQYKNQRPGWPLP-UHFFFAOYSA-N 0.000 description 1
- 125000003136 n-heptyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- 150000002814 niacins Chemical class 0.000 description 1
- LBHIOVVIQHSOQN-UHFFFAOYSA-N nicorandil Chemical compound [O-][N+](=O)OCCNC(=O)C1=CC=CN=C1 LBHIOVVIQHSOQN-UHFFFAOYSA-N 0.000 description 1
- 229960002497 nicorandil Drugs 0.000 description 1
- 239000002674 ointment Substances 0.000 description 1
- 239000004006 olive oil Substances 0.000 description 1
- 235000008390 olive oil Nutrition 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 229940039748 oxalate Drugs 0.000 description 1
- 229940055726 pantothenic acid Drugs 0.000 description 1
- 235000019161 pantothenic acid Nutrition 0.000 description 1
- 239000011713 pantothenic acid Substances 0.000 description 1
- FJKROLUGYXJWQN-UHFFFAOYSA-N papa-hydroxy-benzoic acid Natural products OC(=O)C1=CC=C(O)C=C1 FJKROLUGYXJWQN-UHFFFAOYSA-N 0.000 description 1
- 239000012188 paraffin wax Substances 0.000 description 1
- 239000002304 perfume Substances 0.000 description 1
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 229940093430 polyethylene glycol 1500 Drugs 0.000 description 1
- 229940057838 polyethylene glycol 4000 Drugs 0.000 description 1
- 150000004032 porphyrins Chemical class 0.000 description 1
- 239000013641 positive control Substances 0.000 description 1
- XAEFZNCEHLXOMS-UHFFFAOYSA-M potassium benzoate Chemical compound [K+].[O-]C(=O)C1=CC=CC=C1 XAEFZNCEHLXOMS-UHFFFAOYSA-M 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 230000002335 preservative effect Effects 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 229960004063 propylene glycol Drugs 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- 239000012264 purified product Substances 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 230000000284 resting effect Effects 0.000 description 1
- 229960004889 salicylic acid Drugs 0.000 description 1
- 210000004761 scalp Anatomy 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 230000004215 skin function Effects 0.000 description 1
- 229940080264 sodium dodecylbenzenesulfonate Drugs 0.000 description 1
- 239000001540 sodium lactate Substances 0.000 description 1
- 229940005581 sodium lactate Drugs 0.000 description 1
- 235000011088 sodium lactate Nutrition 0.000 description 1
- 159000000000 sodium salts Chemical class 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 230000035882 stress Effects 0.000 description 1
- 229940086735 succinate Drugs 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-L succinate(2-) Chemical compound [O-]C(=O)CCC([O-])=O KDYFGRWQOYBRFD-UHFFFAOYSA-L 0.000 description 1
- VNOYUJKHFWYWIR-ITIYDSSPSA-N succinyl-CoA Chemical compound O[C@@H]1[C@H](OP(O)(O)=O)[C@@H](COP(O)(=O)OP(O)(=O)OCC(C)(C)[C@@H](O)C(=O)NCCC(=O)NCCSC(=O)CCC(O)=O)O[C@H]1N1C2=NC=NC(N)=C2N=C1 VNOYUJKHFWYWIR-ITIYDSSPSA-N 0.000 description 1
- 229940095064 tartrate Drugs 0.000 description 1
- 238000010998 test method Methods 0.000 description 1
- OULAJFUGPPVRBK-UHFFFAOYSA-N tetratriacontyl alcohol Natural products CCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCO OULAJFUGPPVRBK-UHFFFAOYSA-N 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- 229930003799 tocopherol Natural products 0.000 description 1
- 229960001295 tocopherol Drugs 0.000 description 1
- 235000010384 tocopherol Nutrition 0.000 description 1
- 239000011732 tocopherol Substances 0.000 description 1
- 229940042585 tocopherol acetate Drugs 0.000 description 1
- 230000000699 topical effect Effects 0.000 description 1
- 150000004670 unsaturated fatty acids Chemical class 0.000 description 1
- 235000021122 unsaturated fatty acids Nutrition 0.000 description 1
- 229940070710 valerate Drugs 0.000 description 1
- NQPDZGIKBAWPEJ-UHFFFAOYSA-N valeric acid Chemical compound CCCCC(O)=O NQPDZGIKBAWPEJ-UHFFFAOYSA-N 0.000 description 1
- 229940099259 vaseline Drugs 0.000 description 1
- 229940088594 vitamin Drugs 0.000 description 1
- 235000013343 vitamin Nutrition 0.000 description 1
- 239000011782 vitamin Substances 0.000 description 1
- 229930003231 vitamin Natural products 0.000 description 1
- 229940046009 vitamin E Drugs 0.000 description 1
- 235000019165 vitamin E Nutrition 0.000 description 1
- 239000011709 vitamin E Substances 0.000 description 1
- 239000000230 xanthan gum Substances 0.000 description 1
- 229920001285 xanthan gum Polymers 0.000 description 1
- 235000010493 xanthan gum Nutrition 0.000 description 1
- 229940082509 xanthan gum Drugs 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- GVJHHUAWPYXKBD-IEOSBIPESA-N α-tocopherol Chemical compound OC1=C(C)C(C)=C2O[C@@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-IEOSBIPESA-N 0.000 description 1
Landscapes
- Cosmetics (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Description
【0001】
【発明の属する技術分野】
本発明は、特に養毛料や発毛促進剤として好適な頭部用外用剤に関する技術分野の発明である。より具体的には、5−アミノレブリン酸類を有効成分とすることにより、優れた毛髪伸長促進効果等の養毛料や発毛促進剤として有用な特徴を有する主に頭部に用いる頭部用外用剤に関する発明である。
【0002】
【従来の技術】
高齢化社会、ストレス社会といわれる現代社会では、頭部毛髪が様々な原因により脱毛の危機にさらされる機会がますます多くなってきている。これに対応して、より優れた養毛料等の様々な薬効を有する養毛料等の頭部用外用剤を提供すべく様々な試みがなされている。例えば、養毛料が毛髪に与える効果として主なものに、発毛誘導効果(発毛促進効果,成長期誘導効果)、毛髪を太くする効果、毛髪成長期延長効果、5α−レダクターゼ阻害効果、血行促進効果、殺菌効果、フケ防止効果、保湿効果、抗酸化効果等の効果が挙げられる。
【0003】
しかしながら、前記のように種々の試みがなされているにもかかわらず、従来の養毛料では、その脱毛防止,発毛効果等の養毛作用は必ずしも十分なものではない。これはおそらく脱毛の原因がさまざまであり、また発毛の機構も非常に複雑であるためと考えられている。
従って、このような毛髪の複雑な機構に幅広く対応すべく、様々な用途に応用可能な頭部用外用剤を提供することが望まれている。
【0004】
【発明が解決しようとする課題】
本発明の課題は、発毛促進効果等の頭部外用剤の有効成分として好ましい効能を有する特定の成分をさらに見出し、これを有効成分として配合した頭部用外用剤を提供して、上記の現状を改善することにある。
【0005】
【課題を解決するための手段】
本発明者は、頭髪用外用剤の有効成分として有用な成分の探索対象を、脱毛防止効果及び発毛促進効果等の養毛作用を示すことが既に知られていない化合物にしぼり、上記目的を達成すべく検討した。その結果、驚くべきことに5−アミノレブリン酸及びその誘導体に優れた発毛促進作用が認められることが判明し、この5−アミノレブリン酸類が頭部用外用剤の有効成分として極めて有用であることを見出した。
【0006】
すなわち本発明は、5−アミノレブリン酸及び/又は5−アミノレブリン酸誘導体を有効成分とする頭部用外用剤を提供する。この本発明に関わる頭部用外用剤は、具体的には例えば養毛料又は発毛促進剤としての態様で用いることが好適である。
【0007】
本発明において「頭部用外用剤」とは、凡そ頭部に使用する外用剤の全てを意味するものであり、養毛料,発毛促進剤等の態様を採り得る外用剤である。
また、本発明において「発毛促進剤」とは、特に頭部の発毛作用を促進する発毛促進効果を直接的に発揮させることを一義的な目的とする頭髪用外用剤である。
【0008】
これに対して「養毛料」は、この「発毛促進剤」の概念を包含することは勿論のこと、他の毛髪に関して有用な様々な作用、具体的には上述した毛髪の成長期誘導作用、毛髪を太くする作用、毛髪成長期延長作用、5α−レダクターゼ阻害作用、血行促進作用、殺菌作用、フケ防止作用、保湿作用、抗酸化作用等の作用を発揮して毛髪を養生する頭髪用外用剤を広く意味するものである。
【0009】
また、本発明において「5−アミノレブリン酸及び/又は5−アミノレブリン酸誘導体」(本明細書を通じて,5−アミノレブリン酸類ということもある)とは、「5−アミノレブリン酸」や「5−アミノレブリン酸誘導体」はもちろん、「5−アミノレブリン酸の塩」や「5−アミノレブリン酸誘導体の塩」をも含むものである。この塩の具体的な態様については後述する。
【0010】
【発明の実施の形態】
以下、本発明の実施の形態について説明する。
本発明頭部用外用剤は、前述の通り、5−アミノレブリン酸及び/又はその誘導体を有効成分とする頭部用外用剤である。
【0011】
5−アミノレブリン酸は、δ−アミノレブリン酸ともいい、式
HOOC−(CH2)2 (CO)−CH2 −NH2
で表されるアミノ酸の一種である(以下,5−ALAともいう)。
この5−アミノレブリン酸は、各種の生物に広く分布し、通常は5−アミノレブリン酸シンセターゼにより、スクシニルCoAとグリシンとから生合成される。この5−アミノレブリン酸は、ポルフィリンの生合成の中間体として知られている。
【0012】
5−アミノレブリン酸は、化学合成法,微生物発酵法及び酵素法のいずれの公知の方法により製造して本発明頭部用外用剤の有効成分として用いることができる。なお、微生物又は酵素による生産方法によって5−アミノレブリン酸を製造して本発明において用いる場合は、これらの製造方法によって得られる5−アミノレブリン酸の粗精製物をも、これらの粗精製物が人体に対して有害な物質を含まない限り分離精製することなしに本発明頭部用外用剤中に配合することができる。
【0013】
5−アミノレブリン酸は、除草剤(特表昭61-502814号)、殺虫剤(特開平2-138201号)、植物成長調節剤(特許番号第2613136号)等の有効成分として有用であることが知られている。しかしながら、化粧料等の皮膚外用剤の分野においては、紫外線等の光に対する防御手段として化粧料等中に配合されている(特開平7-10750号)のみであり、本発明において認められた発毛促進効果については全く報告されていない。
【0014】
本発明頭部用外用剤の有効成分として配合され得る5−アミノレブリン酸誘導体の代表的な態様としては、5−アミノレブリン酸のエステルやN−アシル−5−アミノレブリン酸等を挙げることができる。
【0015】
5−アミノレブリン酸エステルの態様として、例えば置換基を有してもよい直鎖,分岐鎖又は環状の炭素数1〜24のアルキルエステル、好ましくは例えばメチル基,エチル基,イソプロピル基,n−ヘキシル基,シクロヘキシル基,n−ヘプチル基,n−オクチル基,n−ノニル基,n−ドデシル基,n−ヘキサデシル基,ベンジル基,フェネチル基,3−フェニルプロピル基,ヒドロキシエチル基,エトキシエチル基等をエステル残基とした5−アミノレブリン酸エステルを挙げることができる。また、これらのアルキル基が有し得る置換基としては、例えばヒドロキシ基,アルコキシ基,フェニル基,ベンジル基,ヒドロキシメチル基等が挙げられる。
【0016】
N−アシル−5−アミノレブリン酸としては、5−アミノレブリン酸のアミノ基が、例えば炭素数1〜24のアルカノイル基,芳香属アシル基,ベンジルオキシカルボニル基等でアシル化された化合物が挙げられる。さらに具体的に好ましいN−アシル基の態様としては、例えばアセチル基,n−ペンタノイル基,n−ヘキサノイル基,n−ノナノイル基,ベンゾイル基,ベンジルオキシカルボニル基等が挙げられる。
【0017】
これらの5−アミノレブリン酸のエステル及びN−アシル−5−アミノレブリン酸は、例えば特開平4−9360号公報等に記載された通常公知の化学合成法によって製造することができる。また、その製造工程において得られる目的物を含む粗精製物をも、これらの粗精製物が人体に対して有害な物質を含まない限り分離精製することなしに本発明頭部用外用剤中に配合することができる。
【0018】
5−アミノレブリン酸誘導体のうち、5−アミノレブリン酸のエステル及びN−アシル−5−アミノレブリン酸から選ばれる化合物の一部は、除草剤(特開平4-9360号)、植物成長調節剤(特願平5-205075号)として有用であることが知られているが、これらの5−アミノレブリン酸誘導体の発毛促進効果については全く報告されていない。
【0019】
これらの5−アミノレブリン酸類の塩としては、製薬学上許容される種々の態様の塩が例示され、これらの塩が人体に対して有害でない限りにおいて特に限定されるものではなく、例えば無機塩,有機酸塩,アルカリ金属塩,アルカリ土類金属塩,アンモニウム又はアルキルアンモニウム塩等が挙げられる。
【0020】
より具体的には、例えば塩酸塩,リン酸塩,硝酸塩,酢酸塩,硫酸塩,プロピオン酸塩,酪酸塩,吉草酸塩,クエン酸塩,フマル酸塩,マレイン酸塩,リンゴ酸塩,臭化水素酸塩,ヨウ化水素酸塩,コハク酸塩,蓚酸塩,乳酸塩,酒石酸塩,グリコール酸塩,メタンスルホン酸塩,トルエンスルホン酸塩等の酸付加塩や、ナトリウム塩,カリウム塩,カルシウム塩等の金属塩等を挙げることができる。
【0021】
本発明頭部用外用剤における5−アミノレブリン酸類の配合量は、外用剤の形態や施用方法等に応じて適宜選択し得るものであり、画一的に限定されるべきものではないが、一般的には外用剤の0.001重量%以上,10.0重量%以下、好ましくは同0.01重量%以上,5.0重量%以下である。この配合量が外用剤の0.001重量%未満であると、本発明の所期の効果である発毛促進作用等が十分に発揮されず好ましくなく、同10.0重量%を超えると製剤化が困難になるため好ましくない。
【0022】
このように、5−アミノレブリン酸類を有効成分として配合することにより、優れた発毛促進作用を示す、養毛料や発毛促進剤として好適に使用することが可能な頭部用外用剤が提供される。
本発明頭部用外用剤中には、その具体的な態様(例えば養毛料及び発毛促進剤)に応じて、通常公知の成分を組み合わせて配合することができる。
【0023】
例えば、高級不飽和脂肪酸,固形パラフィン,流動パラフィン,シリコーン油,スクワラン,モノオレイン酸グリセリル,オリーブ油,高級アルコール等の油分;グリセリン,ヒアルロン酸,プロピレングリコール,マルチトール,アテロコラーゲン,乳酸ナトリウム等の保湿剤;マルメロ粘質物,カルボキシビニルポリマー,キサンタンガム等の増粘剤;ニコチン酸アミド,ニコチン酸ベンジル,ビタミンEアセテート,センブリ抽出物,塩化カルプロニウム,ミノキシジル,ニコランジル,サイクロスポリン誘導体,アセチルコリン誘導体等;セリン,メチオニン,アルギニン等のアミノ酸類;ビタミンB6 ,ビタミンE(若しくはその誘導体),ビオチン,パントテン酸(若しくはその誘導体)等のビタミン類;ニコチン酸,ニコチン酸メチル,ニコチン酸トコフェロール等のニコチン酸エステル類;セファランチン等の皮膚機能亢進剤;エストラジオール等の女性ホルモン剤;グリチルレチン酸(若しくはその誘導体)等の消炎剤;ヒノキチオール,ヘキサクロロフェン,ベンザルコニウムクロリド,ビチオノール等の抗菌剤;メントール等の清涼剤;サリチル酸,亜鉛(若しくはその誘導体),乳酸(若しくはそのアルキルエステル)等;クエン酸等の有機酸類等を配合することができる。
【0024】
これらの諸成分を、例えば本発明頭部用外用剤が発毛促進剤としての態様を採る場合には、特に発毛促進に寄与することが知られている成分を特に選択して、有効成分として配合することが可能である。
【0025】
また、本発明頭部用外用剤が、さらに広く「養毛料」としての態様を採る場合には、発毛促進成分のみならず、他の養毛成分を選択して配合して、養毛料としての特徴付けをすることが可能である(勿論、発毛促進剤としての態様をとる場合にも、これらの養毛成分を必要に応じて配合することも可能である)。
【0026】
本発明頭部用外用剤の形態は、液状,乳液,軟膏等の頭皮に適用できる性状のものであればいずれの形態を採ることも可能であり、一般的にヘアトニック,ヘアリキッド,スカルプトトリートメント,ヘアクリーム,エアゾールムース,エアゾールスプレー,ヘアジェル,スプレームース,シャンプー,リンス等の製品形態をとることができる。
本発明頭部用外用剤の具体的処方は、実施例において例示する。
【0027】
【実施例】
次に実施例をあげて本発明をさらに具体的に説明するが、本発明はこれらにより限定されるものでない。なお、配合量は特記しない限り重量%である。発毛効果試験とその結果について順に説明する。
【0028】
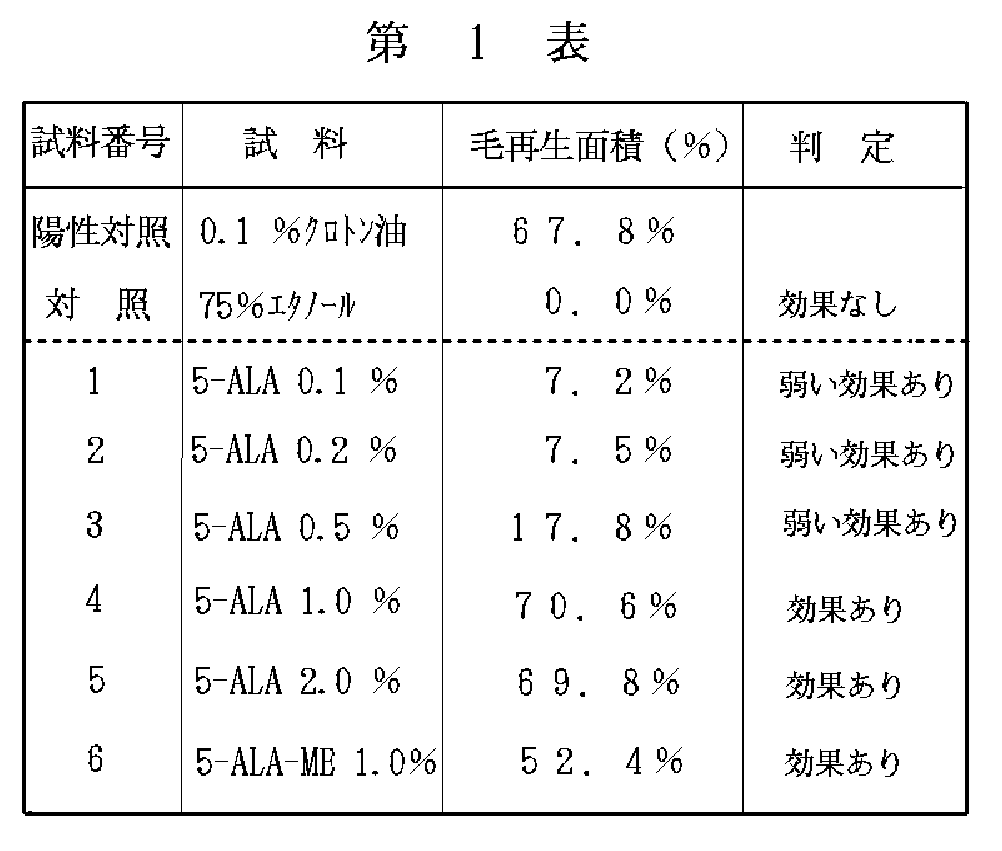
〔発毛試験〕
1.試料の調製
5-アミノレブリン酸(5-ALA)0.1%、0.2%、0.5%、1.0 %、2.0 %を、それぞ
れ75%エタノール溶液に溶解して試料1〜5を得た。また、5-アミノレブリン酸メチルエステル(5-ALAーME)1.0 %を75%エタノール溶液に溶解して試料6を得た。
【0029】
2.発毛試験方法及びその結果
陽性対照試料として 0.1%クロトン油の75%エタノール懸濁液を用い、本発明の液状の試料1〜5(第1表参照のこと)について発毛試験行った。すなわち、実験動物として毛周期の休止期にあるC3H/HeNCrJマウスを使用し、小川らの方法〔ノーマルアンドアブノーマル、エピダーマル、ディファレンシエ−ション(Normal and Abnormal Epidermal Differentiation )、M .Seiji およびI .A .Bernstein 編集、第159 〜170 ページ、1982年、東大出版〕に従って行なった。
【0030】
すなわち、マウスを1群10匹とし、それぞれ被検試料1〜6と対照試料( 0.1%クロトン油の75%エタノール懸濁液および)用の5群に分け、バリカンでマウスの背部を剃毛し、それぞれの試料を1日1回、 0.1mlずつ塗布した。24日後に毛の再生面積を測定した。その結果は再生面積の平均値で表した。これらについて第1表に示す。
【0031】
【表1】
【0032】
<判定基準>
効果あり:毛再生面積が40%以上
弱い効果あり:毛再生面積が5%以上,40%未満
効果なし:毛再生面積が5%未満
【0033】
第1表より、5−アミノレブリン酸及び5−アミノレブリン酸メチルエステルは、マウスの発毛試験において優れた発毛効果を示した。また、5−アミノレブリン酸の効果は1.0%以上の濃度ではその効果の向上は認められなかった。
この結果より、5−アミノレブリン酸又は5−アミノレブリン酸エステルを配合した本発明化粧料は、優れた発毛効果を有することは明らかになり、これらは、養毛用途及び発毛促進用途に適した頭部外用剤であることが判明した。
【0034】
以下に、種々の剤型の本発明頭部外用剤の処方例を実施例として説明する。なお、これらの頭部用外用剤はいずれも、養毛料及び発毛促進剤の双方の態様に用いることが可能である。そして、これらの本発明頭部外用剤に、上述の発毛試験を行ったところ、いずれの処方の本発明頭部外用剤も毛再生面積が40%以上であり、「効果あり」という判定がなされた。
【0035】
〔実施例1〕乳液
配合成分 配合量
(A相)
5−アミノレブリン酸 0.5
ポリオキシエチレン(60モル)付加硬化ヒマシ油 2.0
グリセリン 10.0
ジプロピレングリコール 10.0
1,3−ブチレングリコール 5.0
ポリエチレングリコール1500 5.0
(B相)
セチルイソオクタネート 10.0
スクワラン 5.0
ワセリン 2.0
プロピルパラベン 2.0
(C相)
カルボキシビニルポリマー1%水溶液 30.0
ヘキサメタリン酸ソーダ 0.03
イオン交換水 7.85
(D相)
イオン交換水 5.4
(E相)
カセイカリ 0.12
イオン交換水 5.0
【0036】
(製造法)
A相、B相をそれぞれ60℃で加熱溶解し、混合してホモミキサー処理しゲルを作る。これにD相を徐々に添加しホモミキサーで分散した。
次にこれに溶解したC相を加え、最後に溶解したE相を添加しホモミキサーで乳化してO/W乳液を得た。
【0037】
〔実施例2〕 クリーム
配合成分 配合量
(A相)
流動パラフィン 5.0
セトステアリルアルコール 5.5
グリセリルモノステアレート 3.0
EO(20モル)−2−オクチルドデシルエーテル 8.0
プロピルパラベン 0.3
香料 0.1
(B相)
5−アミノレブリン酸 2.0
グリセリン 8.0
ジプロピレングリコール 20.0
ポリエチレングリコール4000 5.0
ドデシル硫酸ナトリウム 0.1
ヘキサメタリン酸ソーダ 0.005
イオン交換水 42.995
【0038】
(製造法)
A相、B相をそれぞれ加熱溶解して混合し、ホモミキサーで乳化してクリームを得た。
【0039】
〔実施例3〕 ヘアートニック
配合成分 配合量
5−アミノレブリン酸 1.0
ステアリルジメチルアミンオキシド 0.5
硬化ヒマシ油エチレンオキシド(40モル)付加物 1.0
95%エタノール 54.0
イオン交換水 残 量
【0040】
(製造法)
95%エタノールにイオン交換水を加え、これに硬化ヒマシ油エチレンオキシド(40モル)付加物およびステアリルジメチルアミンオキシドを加えた後エキス乾燥物を加え、撹拌溶解してヘアートニックを得た。
【0041】
〔実施例4〕 ヘアートニック
配合成分 配合量
グリセリン 5.0
L−メントール 0.1
5−アミノレブリン酸 0.1
95%エタノール 60.0
香料 0.5
イオン交換水 残 量
【0042】
(製造法)
95%エタノールにグリセリン、L−メントール、香料及びエキス乾燥物を加え、撹拌溶解した後、イオン交換水を加えた。
【0043】
〔実施例5〕 ヘアートニック
配合成分 配合量
N−ヤシラウリル−β−アミノプロピオン酸ソーダ 0.2
5−アミノレブリン酸 5.0
ドデシルベンゼンスルホン酸ナトリウム 0.5
硬化ヒマシ油エチレンオキシド(40モル)付加物 1.0
95%エタノール 70.0
イオン交換水 残 量
【0044】
(製造法)
95%エタノールにイオン交換水を加え、これに硬化ヒマシ油エチレンオキシド(40モル)付加物およびステアリルジメチルアミンオキシド及びN−ヤシラウリル−β−アミノプロピオン酸ソーダを加えた後エキス乾燥物を加え、撹拌溶解してヘアートニックを得た。
【0045】
〔実施例6〕 シャンプー
配合成分 配合量
ココイルメチルタウリンナトリウム 2.0
ポリオキシエチレン(8モル)オレイルアルコール 2.0
ラウリン酸ジエタノールアミド 4.0
エチレングリコール脂肪酸エステル 1.0
グリセリン 0.2
メントール 0.1
5−アミノレブリン酸 0.2
香料 適 量
プロピレングリコール 2.0
イオン交換水 残 量
【0046】
(製造法)
イオン交換水を70℃に加熱し、他の成分を加えて均一に溶解した後、系を冷却してシャンプーを得た。
【0047】
〔実施例7〕 リンス
配合成分 配合量
塩化ステアリルトリメチルアンモニウム 0.7
シリコーン油 3.0
ステアリルアルコール 1.0
ポリオキシエチレン(10モル)オレイルアルコールエーテル 1.0
グリセリン 5.0
5−アミノレブリン酸 0.01
防腐剤 適 量
紫外線吸収剤 適 量
イオン交換水 残 量
【0048】
(製造法)
イオン交換水に塩化ステアリルトリメチルアンモニウム及びグリセリンを加えて70℃に保ち水相を調製した。一方、他の成分を混合し、加熱融解して70℃に保ち油相を調製した。前記水相にこの油相を加えて、ホモミキサーで乳化した後、攪拌しながら冷却してリンスを得た。
【0049】
【発明の効果】
本発明により、優れた発毛促進効果を有する養毛料や発毛促進剤として用いることが好適な頭部用外用剤が提供される。[0001]
BACKGROUND OF THE INVENTION
This invention is invention of the technical field regarding the external preparation for heads especially suitable as a hair nourishing agent and a hair growth promoter. More specifically, by using 5-aminolevulinic acid as an active ingredient, an external preparation for head mainly used in the head having characteristics useful as a hair nourishing agent and hair growth promoting agent such as an excellent hair elongation promoting effect. It is invention regarding.
[0002]
[Prior art]
In the modern society, which is said to be an aging society and a stress society, there are more and more opportunities for the hair of the head to be exposed to the risk of hair loss due to various causes. In response to this, various attempts have been made to provide external preparations for the head such as hair nourishing agents having various medicinal effects such as better hair nourishing agents. For example, the main effects of hair nourishment on hair are hair growth induction effect (hair growth promotion effect, growth phase induction effect), hair thickening effect, hair growth phase extension effect, 5α-reductase inhibition effect, blood circulation Examples of the effect include an accelerating effect, a bactericidal effect, an anti-dandruff effect, a moisturizing effect, and an antioxidant effect.
[0003]
However, in spite of various attempts as described above, conventional hair nourishing agents do not always have sufficient hair nourishing action such as hair loss prevention and hair growth effect. This is probably due to the various causes of hair loss, and the mechanism of hair growth is also very complex.
Therefore, it is desired to provide an external preparation for the head that can be applied to various uses in order to widely cope with such complicated mechanisms of hair.
[0004]
[Problems to be solved by the invention]
The problem of the present invention is to further find a specific component having a preferable effect as an active ingredient of a head external preparation such as a hair growth promoting effect, and provide a head external preparation formulated with this as an active ingredient. It is to improve the current situation.
[0005]
[Means for Solving the Problems]
The present inventor narrows down the search target of an ingredient useful as an active ingredient of an external preparation for hair to a compound that is not already known to exhibit a hair-restoring action such as a hair loss prevention effect and a hair growth promoting effect. Considered to achieve. As a result, it was surprisingly found that 5-aminolevulinic acid and its derivatives have an excellent hair growth promoting action, and that this 5-aminolevulinic acid is extremely useful as an active ingredient for a head external preparation. I found it.
[0006]
That is, this invention provides the external preparation for heads which uses 5-aminolevulinic acid and / or a 5-aminolevulinic acid derivative as an active ingredient. Specifically, the external preparation for head according to the present invention is preferably used, for example, in a form as a hair nourishing agent or a hair growth promoter.
[0007]
In the present invention, the term “external preparation for head” means all external preparations used for the head, and is an external preparation that can take forms such as a hair nourishing agent and a hair growth promoter.
Further, in the present invention, the “hair growth promoter” is an external preparation for hair whose primary purpose is to directly exert the hair growth promoting effect that promotes the hair growth action of the head.
[0008]
On the other hand, “hair growth agent” includes not only the concept of “hair growth promoter” but also various actions useful for other hair, specifically, the above-described hair growth-inducing action. , Hair thickening, hair growth phase extension, 5α-reductase inhibition, blood circulation promotion, bactericidal, anti-dandruff, moisturizing, antioxidant, etc. It means the agent widely.
[0009]
Further, in the present invention, “5-aminolevulinic acid and / or 5-aminolevulinic acid derivative” (also referred to as 5-aminolevulinic acid throughout this specification) means “5-aminolevulinic acid” and “5-aminolevulinic acid derivative”. "Of course," salt of 5-aminolevulinic acid "and" salt of 5-aminolevulinic acid derivative "are also included. Specific embodiments of this salt will be described later.
[0010]
DETAILED DESCRIPTION OF THE INVENTION
Embodiments of the present invention are described below.
As described above, the external preparation for head of the present invention is an external preparation for head containing 5-aminolevulinic acid and / or a derivative thereof as an active ingredient.
[0011]
5-Aminolevulinic acid is also called δ-aminolevulinic acid and has the formula HOOC- (CH 2 ) 2 (CO) —CH 2 —NH 2.
(Hereinafter, also referred to as 5-ALA).
This 5-aminolevulinic acid is widely distributed in various organisms, and is usually biosynthesized from succinyl CoA and glycine by 5-aminolevulinic acid synthetase. This 5-aminolevulinic acid is known as an intermediate of porphyrin biosynthesis.
[0012]
5-Aminolevulinic acid can be produced by any known method such as a chemical synthesis method, a microbial fermentation method, and an enzyme method and used as an active ingredient of the external preparation for head of the present invention. In addition, when 5-aminolevulinic acid is produced by a production method using microorganisms or enzymes and used in the present invention, the crude product of 5-aminolevulinic acid obtained by these production methods is also used in the human body. On the other hand, as long as it does not contain harmful substances, it can be blended in the external preparation for head of the present invention without separation and purification.
[0013]
5-aminolevulinic acid is useful as an active ingredient such as a herbicide (Japanese Patent Publication No. 61-502814), an insecticide (Japanese Patent Laid-Open No. 2-138201), a plant growth regulator (Japanese Patent No. 2613136), etc. Are known. However, in the field of topical skin preparations such as cosmetics, it is only blended in cosmetics and the like as a protective means against light such as ultraviolet rays (Japanese Patent Laid-Open No. 7-10750), and is recognized in the present invention. There has been no report on hair promoting effects.
[0014]
Representative examples of the 5-aminolevulinic acid derivative that can be blended as an active ingredient of the external preparation for the head of the present invention include esters of 5-aminolevulinic acid and N-acyl-5-aminolevulinic acid.
[0015]
As an embodiment of 5-aminolevulinic acid ester, for example, a linear, branched or cyclic alkyl ester having 1 to 24 carbon atoms which may have a substituent, preferably, for example, methyl group, ethyl group, isopropyl group, n-hexyl Group, cyclohexyl group, n-heptyl group, n-octyl group, n-nonyl group, n-dodecyl group, n-hexadecyl group, benzyl group, phenethyl group, 3-phenylpropyl group, hydroxyethyl group, ethoxyethyl group, etc. And 5-aminolevulinic acid ester in which is an ester residue. Examples of the substituent that these alkyl groups may have include a hydroxy group, an alkoxy group, a phenyl group, a benzyl group, and a hydroxymethyl group.
[0016]
Examples of N-acyl-5-aminolevulinic acid include compounds in which the amino group of 5-aminolevulinic acid is acylated with, for example, an alkanoyl group having 1 to 24 carbon atoms, an aromatic acyl group, or a benzyloxycarbonyl group. More specifically, preferred examples of the N-acyl group include an acetyl group, an n-pentanoyl group, an n-hexanoyl group, an n-nonanoyl group, a benzoyl group, and a benzyloxycarbonyl group.
[0017]
These esters of 5-aminolevulinic acid and N-acyl-5-aminolevulinic acid can be produced by a generally known chemical synthesis method described in, for example, JP-A-4-9360. In addition, the crude preparation containing the target product obtained in the production process is also included in the external preparation for head of the present invention without being separated and purified unless the roughly purified product contains a substance harmful to the human body. Can be blended.
[0018]
Among 5-aminolevulinic acid derivatives, some of the compounds selected from esters of 5-aminolevulinic acid and N-acyl-5-aminolevulinic acid are herbicides (Japanese Patent Laid-Open No. 4-9360), plant growth regulators (Japanese Patent Application) Although it is known that the 5-aminolevulinic acid derivative is effective for hair growth, it has not been reported at all.
[0019]
Examples of salts of these 5-aminolevulinic acids include various forms of pharmaceutically acceptable salts, and are not particularly limited as long as these salts are not harmful to the human body. For example, inorganic salts, Examples thereof include organic acid salts, alkali metal salts, alkaline earth metal salts, ammonium or alkylammonium salts.
[0020]
More specifically, for example, hydrochloride, phosphate, nitrate, acetate, sulfate, propionate, butyrate, valerate, citrate, fumarate, maleate, malate, odor Acid addition salts such as hydrohalide, hydroiodide, succinate, oxalate, lactate, tartrate, glycolate, methanesulfonate, toluenesulfonate, sodium salt, potassium salt, Examples thereof include metal salts such as calcium salts.
[0021]
The blending amount of 5-aminolevulinic acids in the external preparation for head of the present invention can be appropriately selected according to the form of external preparation, application method, and the like, and should not be limited uniformly. Specifically, it is 0.001 wt% or more and 10.0 wt% or less, preferably 0.01 wt% or more and 5.0 wt% or less of the external preparation. When the blending amount is less than 0.001% by weight of the external preparation, the hair growth promoting action, which is the intended effect of the present invention, is not sufficiently exhibited, and it is not preferable. It is not preferable because it becomes difficult to make it.
[0022]
Thus, by adding 5-aminolevulinic acid as an active ingredient, there is provided an external preparation for the head that can be suitably used as a hair nourishing agent or a hair growth promoter that exhibits an excellent hair growth promoting action. The
In the external preparation for head of the present invention, generally known components can be combined in combination according to the specific aspects (for example, hair nourishing agent and hair growth promoter).
[0023]
For example, higher unsaturated fatty acids, solid paraffin, liquid paraffin, silicone oil, squalane, glyceryl monooleate, olive oil, higher alcohol, etc .; humectants such as glycerin, hyaluronic acid, propylene glycol, maltitol, atelocollagen, sodium lactate Thickeners such as quince mucilage, carboxyvinyl polymer, xanthan gum; nicotinamide, benzyl nicotinate, vitamin E acetate, assembly extract, carpronium chloride, minoxidil, nicorandil, cyclosporin derivatives, acetylcholine derivatives, etc .; serine, methionine, amino acids such as arginine; vitamin B 6, vitamin E (or derivatives thereof), biotin, vitamins such as pantothenic acid (or derivatives thereof); nicotinic acid, nicotinic Nicotinates such as methyl and nicotinic acid tocopherol; skin function enhancers such as cephalanthin; female hormones such as estradiol; anti-inflammatory agents such as glycyrrhetinic acid (or its derivatives); Antibacterial agents such as menthol; refreshing agents such as menthol; salicylic acid, zinc (or derivatives thereof), lactic acid (or alkyl esters thereof), etc .; organic acids such as citric acid, and the like can be blended.
[0024]
For these various components, for example, when the external preparation for the head of the present invention takes an embodiment as a hair growth promoter, an ingredient that is particularly known to contribute to hair growth promotion is selected in particular. It is possible to mix | blend as.
[0025]
In addition, when the external preparation for the head of the present invention takes a form as a “hair restorer” more widely, not only the hair growth promoting component but also other hair nourishing ingredients are selected and blended, (Of course, when taking the form as a hair growth promoter, these hair nourishing ingredients can be blended as necessary).
[0026]
The form of the external preparation for head of the present invention can take any form as long as it is applicable to the scalp such as liquid, emulsion, ointment, etc., and generally hair tonic, hair liquid, sculpt treatment , Hair cream, aerosol mousse, aerosol spray, hair gel, spray mousse, shampoo, rinse, etc.
The specific formulation of the external preparation for head of the present invention is exemplified in the examples.
[0027]
【Example】
EXAMPLES Next, the present invention will be described more specifically with reference to examples, but the present invention is not limited thereto. The blending amount is% by weight unless otherwise specified. The hair growth effect test and the results will be described in order.
[0028]
[Growth test]
1. Sample preparation
Samples 1 to 5 were obtained by dissolving 5-aminolevulinic acid (5-ALA) 0.1%, 0.2%, 0.5%, 1.0%, and 2.0% in a 75% ethanol solution, respectively. Sample 6 was obtained by dissolving 1.0% of 5-aminolevulinic acid methyl ester (5-ALA-ME) in a 75% ethanol solution.
[0029]
2. Hair growth test method and results As a positive control sample, a 75% ethanol suspension of 0.1% croton oil was used, and hair growth tests were performed on liquid samples 1 to 5 (see Table 1) of the present invention. That is, C3H / HeNCrJ mice in the resting period of the hair cycle were used as experimental animals, and the method of Ogawa et al. (Normal and Abnormal Epidermal Differentiation), M. et al. Seiji and I. A. Edited by Bernstein, pp. 159-170, 1982, The University of Tokyo Press].
[0030]
That is, 10 mice per group were divided into 5 groups for test samples 1 to 6 and a control sample (75% ethanol suspension of 0.1% croton oil), and the back of the mouse was shaved with a clipper. Each sample was applied 0.1 ml once a day. After 24 days, the hair regeneration area was measured. The result was expressed as an average value of the reclaimed area. These are shown in Table 1.
[0031]
[Table 1]
[0032]
<Criteria>
Effective: Hair regeneration area is weaker than 40% Effective: Hair regeneration area is 5% or more, less than 40% No effect: Hair regeneration area is less than 5%
From Table 1, 5-aminolevulinic acid and 5-aminolevulinic acid methyl ester showed excellent hair growth effects in mouse hair growth tests. Further, the effect of 5-aminolevulinic acid was not improved at a concentration of 1.0% or more.
From this result, it is clear that the cosmetic of the present invention containing 5-aminolevulinic acid or 5-aminolevulinic acid ester has an excellent hair growth effect, and these are suitable for hair restoration use and hair growth promotion use. It turned out to be an external preparation for the head.
[0034]
Below, the formulation example of this invention head external preparation of various dosage forms is demonstrated as an Example. In addition, all of these external preparations for heads can be used for both aspects of the hair nourishing agent and the hair growth promoter. And when the above-mentioned hair growth test was performed on these head preparations of the present invention, the hair regeneration area of any head preparation of the present invention of any prescription was 40% or more, and the determination that “effective” was made. Was made.
[0035]
[Example 1] Emulsion
Compounding ingredients blending amount (A phase)
5-Aminolevulinic acid 0.5
Polyoxyethylene (60 mol) addition-cured castor oil 2.0
Glycerin 10.0
Dipropylene glycol 10.0
1,3-butylene glycol 5.0
Polyethylene glycol 1500 5.0
(Phase B)
Cetyl isooctanoate 10.0
Squalane 5.0
Vaseline 2.0
Propylparaben 2.0
(Phase C)
Carboxyvinyl polymer 1% aqueous solution 30.0
Sodium hexametaphosphate 0.03
Ion exchange water 7.85
(Phase D)
Ion exchange water 5.4
(Phase E)
Caustic 0.12
Ion exchange water 5.0
[0036]
(Production method)
A phase and B phase are heated and melted at 60 ° C., mixed and homomixed to prepare a gel. Phase D was gradually added thereto and dispersed with a homomixer.
Next, dissolved C phase was added thereto, and finally dissolved E phase was added and emulsified with a homomixer to obtain an O / W emulsion.
[0037]
[Example 2] Cream
Compounding ingredients blending amount (A phase)
Liquid paraffin 5.0
Cetostearyl alcohol 5.5
Glyceryl monostearate 3.0
EO (20 mol) -2-octyldodecyl ether 8.0
Propylparaben 0.3
Fragrance 0.1
(Phase B)
5-Aminolevulinic acid 2.0
Glycerin 8.0
Dipropylene glycol 20.0
Polyethylene glycol 4000 5.0
Sodium dodecyl sulfate 0.1
Sodium hexametaphosphate 0.005
Ion exchange water 42.995
[0038]
(Production method)
A phase and B phase were heated and dissolved, mixed, and emulsified with a homomixer to obtain a cream.
[0039]
[Example 3] Hair Nick
Compounding component blending amount 5-aminolevulinic acid 1.0
Stearyl dimethylamine oxide 0.5
Hardened castor oil ethylene oxide (40 mol) adduct 1.0
95% ethanol 54.0
Remaining amount of ion-exchanged water [0040]
(Production method)
Ion exchanged water was added to 95% ethanol, hydrogenated castor oil ethylene oxide (40 mol) adduct and stearyl dimethylamine oxide were added thereto, and then the dried extract was added and dissolved by stirring to obtain a hair tonic.
[0041]
[Example 4] Hair Nick
Compounding ingredients blending amount Glycerin 5.0
L-Menthol 0.1
5-Aminolevulinic acid 0.1
95% ethanol 60.0
Fragrance 0.5
Remaining amount of ion-exchanged water [0042]
(Production method)
After adding glycerin, L-menthol, a fragrance | flavor, and a dried extract to 95% ethanol, stirring and dissolving, ion-exchanged water was added.
[0043]
[Example 5] Hair Nick
Compounding component compounding amount N-coconut lauryl-β-aminopropionic acid sodium 0.2
5-Aminolevulinic acid 5.0
Sodium dodecylbenzenesulfonate 0.5
Hardened castor oil ethylene oxide (40 mol) adduct 1.0
95% ethanol 70.0
Remaining amount of ion-exchanged water [0044]
(Production method)
Add ion-exchanged water to 95% ethanol, add hydrogenated castor oil ethylene oxide (40 mol) adduct, stearyl dimethylamine oxide and sodium N-cocolauryl-β-aminopropionate, and then add dry extract and stir to dissolve. And got a hair art nick.
[0045]
[Example 6] Shampoo
Compounding component blending amount <br/> Sodium cocoyl methyl taurine 2.0
Polyoxyethylene (8 mol) oleyl alcohol 2.0
Lauric acid diethanolamide 4.0
Ethylene glycol fatty acid ester 1.0
Glycerin 0.2
Menthol 0.1
5-Aminolevulinic acid 0.2
Perfume proper amount propylene glycol 2.0
Residual amount of ion-exchanged water [0046]
(Production method)
Ion exchange water was heated to 70 ° C., and other components were added and dissolved uniformly, and then the system was cooled to obtain a shampoo.
[0047]
[Example 7] Rinse
Compounding ingredients blending amount Stearyltrimethylammonium chloride 0.7
Silicone oil 3.0
Stearyl alcohol 1.0
Polyoxyethylene (10 mol) oleyl alcohol ether 1.0
Glycerin 5.0
5-Aminolevulinic acid 0.01
Preservative Appropriate amount UV absorber Appropriate amount Ion-exchange water remaining amount
(Production method)
Stearyltrimethylammonium chloride and glycerin were added to ion-exchanged water and kept at 70 ° C. to prepare an aqueous phase. On the other hand, other components were mixed, heated and melted, and kept at 70 ° C. to prepare an oil phase. The oil phase was added to the aqueous phase, emulsified with a homomixer, and then cooled with stirring to obtain a rinse.
[0049]
【The invention's effect】
According to the present invention, a head preparation for external use suitable for use as a hair nourishing agent or a hair growth promoter having an excellent hair growth promoting effect is provided.
Claims (3)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP29789697A JP3644809B2 (en) | 1997-10-15 | 1997-10-15 | External preparation for head |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP29789697A JP3644809B2 (en) | 1997-10-15 | 1997-10-15 | External preparation for head |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JPH11116446A JPH11116446A (en) | 1999-04-27 |
| JP3644809B2 true JP3644809B2 (en) | 2005-05-11 |
Family
ID=17852519
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP29789697A Expired - Lifetime JP3644809B2 (en) | 1997-10-15 | 1997-10-15 | External preparation for head |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP3644809B2 (en) |
Families Citing this family (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP3970492B2 (en) * | 1999-12-14 | 2007-09-05 | コスモ石油株式会社 | Peeling composition |
| DE10003620A1 (en) * | 2000-01-28 | 2001-08-02 | Asat Ag Applied Science & Tech | 5-aminolevulinic acid formulation in non-aqueous solvents |
| US8133479B2 (en) * | 2004-04-28 | 2012-03-13 | Cosmo Oil Co., Ltd. | Hair restorer |
| JP2006151927A (en) * | 2004-12-01 | 2006-06-15 | Toin Gakuen | Photodynamic therapy composition |
| US20090130227A1 (en) | 2005-04-28 | 2009-05-21 | Yoshiyasu Ito | External preparation for skin |
| JP5098051B2 (en) | 2007-04-05 | 2012-12-12 | Sbiファーマ株式会社 | Mitochondrial disorder brain disease therapeutic agent and diagnostic agent |
| JP5820654B2 (en) * | 2011-08-03 | 2015-11-24 | 株式会社ミルボン | External preparation for head |
| JP5782327B2 (en) * | 2011-08-03 | 2015-09-24 | 株式会社ミルボン | Hair restorer composition |
| KR101385196B1 (en) * | 2012-05-17 | 2014-04-14 | 주식회사 유니크메디케어 | Compositions for Improving or Facilitating Hair Growth Comprising a Photosensitizer-Peptide as an Active Ingredient Using Light Irradiation, and Methods thereof |
| CN103655433B (en) * | 2013-04-28 | 2016-03-09 | 钱臻 | A kind of biological fermentation hair shampoo and preparation method thereof |
-
1997
- 1997-10-15 JP JP29789697A patent/JP3644809B2/en not_active Expired - Lifetime
Also Published As
| Publication number | Publication date |
|---|---|
| JPH11116446A (en) | 1999-04-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US8632759B2 (en) | Hair restorer | |
| JPH08502509A (en) | Cosmetic composition | |
| JPH07316023A (en) | Hair tonic | |
| JPH0386812A (en) | Cosmetic compound | |
| JP3644809B2 (en) | External preparation for head | |
| JPH07291823A (en) | Improvement in or concerning cosmetic composition | |
| JP5552741B2 (en) | Whitening agent containing zinc as an active ingredient | |
| JPH0699286B2 (en) | Cosmetic composition | |
| JP2002145719A (en) | Cosmetic material | |
| JP2002037716A (en) | Kaurenes-containing composition, hair-growing agent and skin care agent | |
| JPH11292752A (en) | Whitening agent and skin external composition | |
| JPH0669940B2 (en) | Anti-hair graying agent | |
| JP2001131025A (en) | Cosmetic for scalp and hair | |
| JPH07206647A (en) | Hair tonic | |
| JP4327989B2 (en) | Hair cosmetics | |
| JP2025090529A (en) | Composition for hair growth or hair growth promotion containing a thymol ester compound | |
| JP5145214B2 (en) | Disinfectant / preservative | |
| JPH10245393A (en) | Composition containing triterpenes | |
| HK1098965A (en) | Hair restorer | |
| JP2002114645A (en) | Hair tonic | |
| JPH09227328A (en) | Cosmetics | |
| JP2001122739A (en) | Hair tonic | |
| JPH11322546A (en) | Cosmetic for scalp and hair | |
| JP2001278873A (en) | Hair tonic | |
| JPH07206646A (en) | Hair tonic |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20040922 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20041005 |
|
| A521 | Written amendment |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20041206 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20050111 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20050201 |
|
| R150 | Certificate of patent or registration of utility model |
Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20080210 Year of fee payment: 3 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20090210 Year of fee payment: 4 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20100210 Year of fee payment: 5 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20100210 Year of fee payment: 5 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20110210 Year of fee payment: 6 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20120210 Year of fee payment: 7 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20120210 Year of fee payment: 7 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20130210 Year of fee payment: 8 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20140210 Year of fee payment: 9 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| S111 | Request for change of ownership or part of ownership |
Free format text: JAPANESE INTERMEDIATE CODE: R313111 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| EXPY | Cancellation because of completion of term |