JP3634988B2 - Anionic surfactant and detergent composition - Google Patents
Anionic surfactant and detergent composition Download PDFInfo
- Publication number
- JP3634988B2 JP3634988B2 JP29389999A JP29389999A JP3634988B2 JP 3634988 B2 JP3634988 B2 JP 3634988B2 JP 29389999 A JP29389999 A JP 29389999A JP 29389999 A JP29389999 A JP 29389999A JP 3634988 B2 JP3634988 B2 JP 3634988B2
- Authority
- JP
- Japan
- Prior art keywords
- carbon atoms
- alkylene oxide
- average
- aliphatic alcohol
- group
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 239000003945 anionic surfactant Substances 0.000 title claims description 43
- 239000000203 mixture Substances 0.000 title claims description 36
- 239000003599 detergent Substances 0.000 title claims description 20
- -1 sulfosuccinic acid ester salt Chemical class 0.000 claims description 68
- 125000004432 carbon atom Chemical group C* 0.000 claims description 51
- 125000002947 alkylene group Chemical group 0.000 claims description 47
- DNIAPMSPPWPWGF-GSVOUGTGSA-N (R)-(-)-Propylene glycol Chemical compound C[C@@H](O)CO DNIAPMSPPWPWGF-GSVOUGTGSA-N 0.000 claims description 27
- 239000003054 catalyst Substances 0.000 claims description 25
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 18
- 238000006243 chemical reaction Methods 0.000 claims description 18
- 238000009826 distribution Methods 0.000 claims description 15
- 239000002253 acid Substances 0.000 claims description 11
- 238000004140 cleaning Methods 0.000 claims description 11
- 125000002723 alicyclic group Chemical group 0.000 claims description 10
- 125000001931 aliphatic group Chemical group 0.000 claims description 10
- 229910052751 metal Inorganic materials 0.000 claims description 10
- 239000002184 metal Substances 0.000 claims description 10
- 150000001875 compounds Chemical class 0.000 claims description 9
- 238000004519 manufacturing process Methods 0.000 claims description 9
- 150000003839 salts Chemical class 0.000 claims description 9
- 239000003513 alkali Substances 0.000 claims description 8
- 229920000642 polymer Polymers 0.000 claims description 8
- 239000004094 surface-active agent Substances 0.000 claims description 8
- 239000002453 shampoo Substances 0.000 claims description 7
- 239000002280 amphoteric surfactant Substances 0.000 claims description 6
- 150000002500 ions Chemical class 0.000 claims description 6
- 229910019142 PO4 Inorganic materials 0.000 claims description 5
- 238000007259 addition reaction Methods 0.000 claims description 5
- 235000021317 phosphate Nutrition 0.000 claims description 5
- 239000000654 additive Substances 0.000 claims description 4
- 125000002091 cationic group Chemical group 0.000 claims description 4
- 239000003093 cationic surfactant Substances 0.000 claims description 4
- 239000010703 silicon Substances 0.000 claims description 4
- 229910052710 silicon Inorganic materials 0.000 claims description 4
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 claims description 3
- 150000001298 alcohols Chemical class 0.000 claims description 3
- 239000002738 chelating agent Substances 0.000 claims description 3
- 239000003086 colorant Substances 0.000 claims description 3
- 239000003906 humectant Substances 0.000 claims description 3
- 150000002823 nitrates Chemical class 0.000 claims description 3
- 239000002736 nonionic surfactant Substances 0.000 claims description 3
- VLTRZXGMWDSKGL-UHFFFAOYSA-M perchlorate Inorganic materials [O-]Cl(=O)(=O)=O VLTRZXGMWDSKGL-UHFFFAOYSA-M 0.000 claims description 3
- VLTRZXGMWDSKGL-UHFFFAOYSA-N perchloric acid Chemical compound OCl(=O)(=O)=O VLTRZXGMWDSKGL-UHFFFAOYSA-N 0.000 claims description 3
- 150000003013 phosphoric acid derivatives Chemical class 0.000 claims description 3
- 239000003755 preservative agent Substances 0.000 claims description 3
- 150000003467 sulfuric acid derivatives Chemical class 0.000 claims description 3
- 239000006096 absorbing agent Substances 0.000 claims description 2
- 239000013543 active substance Substances 0.000 claims description 2
- 125000000129 anionic group Chemical group 0.000 claims description 2
- 239000003205 fragrance Substances 0.000 claims description 2
- 239000010452 phosphate Substances 0.000 claims description 2
- DXIRVOVNWFDULL-UHFFFAOYSA-N Cl.S(=O)(=O)(O)C(C(=O)O)CC(=O)O Chemical class Cl.S(=O)(=O)(O)C(C(=O)O)CC(=O)O DXIRVOVNWFDULL-UHFFFAOYSA-N 0.000 claims 1
- 229910002651 NO3 Inorganic materials 0.000 claims 1
- NHNBFGGVMKEFGY-UHFFFAOYSA-N Nitrate Chemical compound [O-][N+]([O-])=O NHNBFGGVMKEFGY-UHFFFAOYSA-N 0.000 claims 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 claims 1
- 239000000047 product Substances 0.000 description 29
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 21
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 21
- 235000014113 dietary fatty acids Nutrition 0.000 description 20
- 239000000194 fatty acid Substances 0.000 description 20
- 229930195729 fatty acid Natural products 0.000 description 20
- 239000012190 activator Substances 0.000 description 19
- 239000007795 chemical reaction product Substances 0.000 description 19
- 150000004665 fatty acids Chemical class 0.000 description 19
- LQZZUXJYWNFBMV-UHFFFAOYSA-N dodecan-1-ol Chemical compound CCCCCCCCCCCCO LQZZUXJYWNFBMV-UHFFFAOYSA-N 0.000 description 17
- 238000006116 polymerization reaction Methods 0.000 description 16
- 238000005259 measurement Methods 0.000 description 15
- GEHJYWRUCIMESM-UHFFFAOYSA-L sodium sulfite Chemical compound [Na+].[Na+].[O-]S([O-])=O GEHJYWRUCIMESM-UHFFFAOYSA-L 0.000 description 14
- 239000007864 aqueous solution Substances 0.000 description 11
- 239000003240 coconut oil Substances 0.000 description 11
- 230000000052 comparative effect Effects 0.000 description 11
- 229910052708 sodium Inorganic materials 0.000 description 11
- 239000011734 sodium Substances 0.000 description 11
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 11
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 9
- 125000000217 alkyl group Chemical group 0.000 description 9
- 238000005187 foaming Methods 0.000 description 9
- 238000004817 gas chromatography Methods 0.000 description 9
- 239000011521 glass Substances 0.000 description 9
- 235000019864 coconut oil Nutrition 0.000 description 8
- 238000005227 gel permeation chromatography Methods 0.000 description 8
- FPYJFEHAWHCUMM-UHFFFAOYSA-N maleic anhydride Chemical compound O=C1OC(=O)C=C1 FPYJFEHAWHCUMM-UHFFFAOYSA-N 0.000 description 8
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 7
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 7
- LSNNMFCWUKXFEE-UHFFFAOYSA-N Sulfurous acid Chemical compound OS(O)=O LSNNMFCWUKXFEE-UHFFFAOYSA-N 0.000 description 7
- 229910052799 carbon Inorganic materials 0.000 description 7
- 238000000034 method Methods 0.000 description 7
- IJGRMHOSHXDMSA-UHFFFAOYSA-N nitrogen Substances N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 7
- 229910052757 nitrogen Inorganic materials 0.000 description 7
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 7
- 125000006353 oxyethylene group Chemical group 0.000 description 7
- 235000010265 sodium sulphite Nutrition 0.000 description 7
- 238000003756 stirring Methods 0.000 description 7
- KWIUHFFTVRNATP-UHFFFAOYSA-N Betaine Natural products C[N+](C)(C)CC([O-])=O KWIUHFFTVRNATP-UHFFFAOYSA-N 0.000 description 6
- KWIUHFFTVRNATP-UHFFFAOYSA-O N,N,N-trimethylglycinium Chemical compound C[N+](C)(C)CC(O)=O KWIUHFFTVRNATP-UHFFFAOYSA-O 0.000 description 6
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 6
- 229960003237 betaine Drugs 0.000 description 6
- 230000018044 dehydration Effects 0.000 description 6
- 238000006297 dehydration reaction Methods 0.000 description 6
- 230000000694 effects Effects 0.000 description 6
- 238000011156 evaluation Methods 0.000 description 6
- FSYKKLYZXJSNPZ-UHFFFAOYSA-N sarcosine Chemical compound C[NH2+]CC([O-])=O FSYKKLYZXJSNPZ-UHFFFAOYSA-N 0.000 description 6
- 229910001220 stainless steel Inorganic materials 0.000 description 6
- 239000010935 stainless steel Substances 0.000 description 6
- MPCRDALPQLDDFX-UHFFFAOYSA-L Magnesium perchlorate Chemical compound [Mg+2].[O-]Cl(=O)(=O)=O.[O-]Cl(=O)(=O)=O MPCRDALPQLDDFX-UHFFFAOYSA-L 0.000 description 5
- 230000002378 acidificating effect Effects 0.000 description 5
- 239000012459 cleaning agent Substances 0.000 description 5
- 150000002148 esters Chemical class 0.000 description 5
- 229910052749 magnesium Inorganic materials 0.000 description 5
- 239000011777 magnesium Substances 0.000 description 5
- 238000006277 sulfonation reaction Methods 0.000 description 5
- 239000004215 Carbon black (E152) Substances 0.000 description 4
- 206010015150 Erythema Diseases 0.000 description 4
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 4
- 239000002202 Polyethylene glycol Substances 0.000 description 4
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical compound OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 4
- 229910052783 alkali metal Inorganic materials 0.000 description 4
- 231100000321 erythema Toxicity 0.000 description 4
- 238000005194 fractionation Methods 0.000 description 4
- BXWNKGSJHAJOGX-UHFFFAOYSA-N hexadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCO BXWNKGSJHAJOGX-UHFFFAOYSA-N 0.000 description 4
- 229930195733 hydrocarbon Natural products 0.000 description 4
- 239000007788 liquid Substances 0.000 description 4
- GLDOVTGHNKAZLK-UHFFFAOYSA-N octadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCCCO GLDOVTGHNKAZLK-UHFFFAOYSA-N 0.000 description 4
- 125000005702 oxyalkylene group Chemical group 0.000 description 4
- 229920001223 polyethylene glycol Polymers 0.000 description 4
- 239000007787 solid Substances 0.000 description 4
- HLZKNKRTKFSKGZ-UHFFFAOYSA-N tetradecan-1-ol Chemical compound CCCCCCCCCCCCCCO HLZKNKRTKFSKGZ-UHFFFAOYSA-N 0.000 description 4
- 229910052725 zinc Inorganic materials 0.000 description 4
- KBPLFHHGFOOTCA-UHFFFAOYSA-N 1-Octanol Chemical compound CCCCCCCCO KBPLFHHGFOOTCA-UHFFFAOYSA-N 0.000 description 3
- RFVNOJDQRGSOEL-UHFFFAOYSA-N 2-hydroxyethyl octadecanoate Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCCO RFVNOJDQRGSOEL-UHFFFAOYSA-N 0.000 description 3
- WGVGLSURTKMQQC-UHFFFAOYSA-N 3-aminopropyl-ethyl-dimethylazanium Chemical compound CC[N+](C)(C)CCCN WGVGLSURTKMQQC-UHFFFAOYSA-N 0.000 description 3
- LSNNMFCWUKXFEE-UHFFFAOYSA-M Bisulfite Chemical compound OS([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-M 0.000 description 3
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 description 3
- 235000019482 Palm oil Nutrition 0.000 description 3
- 108010077895 Sarcosine Proteins 0.000 description 3
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 3
- 206010040880 Skin irritation Diseases 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 3
- 238000012644 addition polymerization Methods 0.000 description 3
- 150000001340 alkali metals Chemical class 0.000 description 3
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 3
- 229910052782 aluminium Inorganic materials 0.000 description 3
- 229910052791 calcium Inorganic materials 0.000 description 3
- 239000011575 calcium Substances 0.000 description 3
- 239000003795 chemical substances by application Substances 0.000 description 3
- 239000004205 dimethyl polysiloxane Substances 0.000 description 3
- 235000013870 dimethyl polysiloxane Nutrition 0.000 description 3
- SZXQTJUDPRGNJN-UHFFFAOYSA-N dipropylene glycol Chemical compound OCCCOCCCO SZXQTJUDPRGNJN-UHFFFAOYSA-N 0.000 description 3
- RTZKZFJDLAIYFH-UHFFFAOYSA-N ether Substances CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 3
- 239000006260 foam Substances 0.000 description 3
- VTHJTEIRLNZDEV-UHFFFAOYSA-L magnesium dihydroxide Chemical compound [OH-].[OH-].[Mg+2] VTHJTEIRLNZDEV-UHFFFAOYSA-L 0.000 description 3
- 239000000347 magnesium hydroxide Substances 0.000 description 3
- 229910001862 magnesium hydroxide Inorganic materials 0.000 description 3
- 150000002739 metals Chemical class 0.000 description 3
- 238000002156 mixing Methods 0.000 description 3
- 230000007935 neutral effect Effects 0.000 description 3
- 239000002540 palm oil Substances 0.000 description 3
- 229920000435 poly(dimethylsiloxane) Polymers 0.000 description 3
- 229940043230 sarcosine Drugs 0.000 description 3
- 231100000475 skin irritation Toxicity 0.000 description 3
- 230000036556 skin irritation Effects 0.000 description 3
- 235000019333 sodium laurylsulphate Nutrition 0.000 description 3
- HOVAGTYPODGVJG-UVSYOFPXSA-N (3s,5r)-2-(hydroxymethyl)-6-methoxyoxane-3,4,5-triol Chemical compound COC1OC(CO)[C@@H](O)C(O)[C@H]1O HOVAGTYPODGVJG-UVSYOFPXSA-N 0.000 description 2
- XFRVVPUIAFSTFO-UHFFFAOYSA-N 1-Tridecanol Chemical compound CCCCCCCCCCCCCO XFRVVPUIAFSTFO-UHFFFAOYSA-N 0.000 description 2
- VBICKXHEKHSIBG-UHFFFAOYSA-N 1-monostearoylglycerol Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCC(O)CO VBICKXHEKHSIBG-UHFFFAOYSA-N 0.000 description 2
- PQXKWPLDPFFDJP-UHFFFAOYSA-N 2,3-dimethyloxirane Chemical compound CC1OC1C PQXKWPLDPFFDJP-UHFFFAOYSA-N 0.000 description 2
- XPALGXXLALUMLE-UHFFFAOYSA-N 2-(dimethylamino)tetradecanoic acid Chemical compound CCCCCCCCCCCCC(N(C)C)C(O)=O XPALGXXLALUMLE-UHFFFAOYSA-N 0.000 description 2
- MFGOFGRYDNHJTA-UHFFFAOYSA-N 2-amino-1-(2-fluorophenyl)ethanol Chemical compound NCC(O)C1=CC=CC=C1F MFGOFGRYDNHJTA-UHFFFAOYSA-N 0.000 description 2
- KTCCLEVCNRHWQL-UHFFFAOYSA-N 3-(2-oxotridecylamino)propanoic acid;sodium Chemical compound [Na].CCCCCCCCCCCC(=O)CNCCC(O)=O KTCCLEVCNRHWQL-UHFFFAOYSA-N 0.000 description 2
- DDGPBVIAYDDWDH-UHFFFAOYSA-N 3-[dodecyl(dimethyl)azaniumyl]-2-hydroxypropane-1-sulfonate Chemical compound CCCCCCCCCCCC[N+](C)(C)CC(O)CS([O-])(=O)=O DDGPBVIAYDDWDH-UHFFFAOYSA-N 0.000 description 2
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 2
- QXNVGIXVLWOKEQ-UHFFFAOYSA-N Disodium Chemical compound [Na][Na] QXNVGIXVLWOKEQ-UHFFFAOYSA-N 0.000 description 2
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 2
- 229920002907 Guar gum Polymers 0.000 description 2
- 241000282412 Homo Species 0.000 description 2
- 239000004166 Lanolin Substances 0.000 description 2
- 239000004909 Moisturizer Substances 0.000 description 2
- QZXSMBBFBXPQHI-UHFFFAOYSA-N N-(dodecanoyl)ethanolamine Chemical compound CCCCCCCCCCCC(=O)NCCO QZXSMBBFBXPQHI-UHFFFAOYSA-N 0.000 description 2
- SUZRRICLUFMAQD-UHFFFAOYSA-N N-Methyltaurine Chemical compound CNCCS(O)(=O)=O SUZRRICLUFMAQD-UHFFFAOYSA-N 0.000 description 2
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 2
- VBIIFPGSPJYLRR-UHFFFAOYSA-M Stearyltrimethylammonium chloride Chemical compound [Cl-].CCCCCCCCCCCCCCCCCC[N+](C)(C)C VBIIFPGSPJYLRR-UHFFFAOYSA-M 0.000 description 2
- ULUAUXLGCMPNKK-UHFFFAOYSA-N Sulfobutanedioic acid Chemical compound OC(=O)CC(C(O)=O)S(O)(=O)=O ULUAUXLGCMPNKK-UHFFFAOYSA-N 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 2
- 125000003342 alkenyl group Chemical group 0.000 description 2
- 229940024606 amino acid Drugs 0.000 description 2
- 229910052788 barium Inorganic materials 0.000 description 2
- HUCVOHYBFXVBRW-UHFFFAOYSA-M caesium hydroxide Inorganic materials [OH-].[Cs+] HUCVOHYBFXVBRW-UHFFFAOYSA-M 0.000 description 2
- 239000001913 cellulose Substances 0.000 description 2
- 229920002678 cellulose Polymers 0.000 description 2
- 239000013522 chelant Substances 0.000 description 2
- 239000000460 chlorine Substances 0.000 description 2
- 229910052801 chlorine Inorganic materials 0.000 description 2
- 238000004040 coloring Methods 0.000 description 2
- 125000000753 cycloalkyl group Chemical group 0.000 description 2
- MWKFXSUHUHTGQN-UHFFFAOYSA-N decan-1-ol Chemical compound CCCCCCCCCCO MWKFXSUHUHTGQN-UHFFFAOYSA-N 0.000 description 2
- 125000003438 dodecyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- SYELZBGXAIXKHU-UHFFFAOYSA-N dodecyldimethylamine N-oxide Chemical compound CCCCCCCCCCCC[N+](C)(C)[O-] SYELZBGXAIXKHU-UHFFFAOYSA-N 0.000 description 2
- 238000005886 esterification reaction Methods 0.000 description 2
- 235000011187 glycerol Nutrition 0.000 description 2
- 239000000665 guar gum Substances 0.000 description 2
- 235000010417 guar gum Nutrition 0.000 description 2
- 229960002154 guar gum Drugs 0.000 description 2
- 125000001183 hydrocarbyl group Chemical group 0.000 description 2
- 125000001165 hydrophobic group Chemical group 0.000 description 2
- 229940039717 lanolin Drugs 0.000 description 2
- 235000019388 lanolin Nutrition 0.000 description 2
- HOVAGTYPODGVJG-UHFFFAOYSA-N methyl beta-galactoside Natural products COC1OC(CO)C(O)C(O)C1O HOVAGTYPODGVJG-UHFFFAOYSA-N 0.000 description 2
- 230000001333 moisturizer Effects 0.000 description 2
- 239000012299 nitrogen atmosphere Substances 0.000 description 2
- XGFDHKJUZCCPKQ-UHFFFAOYSA-N nonadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCCCCO XGFDHKJUZCCPKQ-UHFFFAOYSA-N 0.000 description 2
- ZWRUINPWMLAQRD-UHFFFAOYSA-N nonan-1-ol Chemical compound CCCCCCCCCO ZWRUINPWMLAQRD-UHFFFAOYSA-N 0.000 description 2
- 239000006072 paste Substances 0.000 description 2
- 125000003367 polycyclic group Chemical group 0.000 description 2
- 229910052700 potassium Inorganic materials 0.000 description 2
- 239000011591 potassium Substances 0.000 description 2
- 102000004169 proteins and genes Human genes 0.000 description 2
- 108090000623 proteins and genes Proteins 0.000 description 2
- 230000035484 reaction time Effects 0.000 description 2
- 238000000926 separation method Methods 0.000 description 2
- 235000010267 sodium hydrogen sulphite Nutrition 0.000 description 2
- 229940045920 sodium pyrrolidone carboxylate Drugs 0.000 description 2
- HYRLWUFWDYFEES-UHFFFAOYSA-M sodium;2-oxopyrrolidine-1-carboxylate Chemical compound [Na+].[O-]C(=O)N1CCCC1=O HYRLWUFWDYFEES-UHFFFAOYSA-M 0.000 description 2
- 229910052712 strontium Inorganic materials 0.000 description 2
- LSNNMFCWUKXFEE-UHFFFAOYSA-L sulfite Chemical class [O-]S([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-L 0.000 description 2
- 238000010998 test method Methods 0.000 description 2
- 229940087291 tridecyl alcohol Drugs 0.000 description 2
- KJIOQYGWTQBHNH-UHFFFAOYSA-N undecanol Chemical compound CCCCCCCCCCCO KJIOQYGWTQBHNH-UHFFFAOYSA-N 0.000 description 2
- JNYAEWCLZODPBN-JGWLITMVSA-N (2r,3r,4s)-2-[(1r)-1,2-dihydroxyethyl]oxolane-3,4-diol Chemical compound OC[C@@H](O)[C@H]1OC[C@H](O)[C@H]1O JNYAEWCLZODPBN-JGWLITMVSA-N 0.000 description 1
- ALSTYHKOOCGGFT-KTKRTIGZSA-N (9Z)-octadecen-1-ol Chemical compound CCCCCCCC\C=C/CCCCCCCCO ALSTYHKOOCGGFT-KTKRTIGZSA-N 0.000 description 1
- JXNPEDYJTDQORS-HZJYTTRNSA-N (9Z,12Z)-octadecadien-1-ol Chemical compound CCCCC\C=C/C\C=C/CCCCCCCCO JXNPEDYJTDQORS-HZJYTTRNSA-N 0.000 description 1
- QGLWBTPVKHMVHM-KTKRTIGZSA-N (z)-octadec-9-en-1-amine Chemical compound CCCCCCCC\C=C/CCCCCCCCN QGLWBTPVKHMVHM-KTKRTIGZSA-N 0.000 description 1
- UUWJHAWPCRFDHZ-UHFFFAOYSA-N 1-dodecoxydodecane;phosphoric acid Chemical compound OP(O)(O)=O.CCCCCCCCCCCCOCCCCCCCCCCCC UUWJHAWPCRFDHZ-UHFFFAOYSA-N 0.000 description 1
- HPQKGWKGZNXUEF-UHFFFAOYSA-N 1-nonylcyclohexan-1-ol Chemical compound CCCCCCCCCC1(O)CCCCC1 HPQKGWKGZNXUEF-UHFFFAOYSA-N 0.000 description 1
- KHJWSKNOMFJTDN-UHFFFAOYSA-N 2-[2-[bis(carboxymethyl)amino]ethyl-(carboxymethyl)amino]acetic acid;sodium Chemical compound [Na].OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KHJWSKNOMFJTDN-UHFFFAOYSA-N 0.000 description 1
- AEDQNOLIADXSBB-UHFFFAOYSA-N 3-(dodecylazaniumyl)propanoate Chemical compound CCCCCCCCCCCCNCCC(O)=O AEDQNOLIADXSBB-UHFFFAOYSA-N 0.000 description 1
- LIFHMKCDDVTICL-UHFFFAOYSA-N 6-(chloromethyl)phenanthridine Chemical compound C1=CC=C2C(CCl)=NC3=CC=CC=C3C2=C1 LIFHMKCDDVTICL-UHFFFAOYSA-N 0.000 description 1
- XZIIFPSPUDAGJM-UHFFFAOYSA-N 6-chloro-2-n,2-n-diethylpyrimidine-2,4-diamine Chemical compound CCN(CC)C1=NC(N)=CC(Cl)=N1 XZIIFPSPUDAGJM-UHFFFAOYSA-N 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical class N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- HQDYLJNVVXCDAC-UHFFFAOYSA-N CCCCCCCCCCCCCCCCC(C(O)=O)N(CC)CC.CCN.CC(C(O)=O)O Chemical compound CCCCCCCCCCCCCCCCC(C(O)=O)N(CC)CC.CCN.CC(C(O)=O)O HQDYLJNVVXCDAC-UHFFFAOYSA-N 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- PQUCIEFHOVEZAU-UHFFFAOYSA-N Diammonium sulfite Chemical compound [NH4+].[NH4+].[O-]S([O-])=O PQUCIEFHOVEZAU-UHFFFAOYSA-N 0.000 description 1
- KIWBPDUYBMNFTB-UHFFFAOYSA-N Ethyl hydrogen sulfate Chemical compound CCOS(O)(=O)=O KIWBPDUYBMNFTB-UHFFFAOYSA-N 0.000 description 1
- FPVVYTCTZKCSOJ-UHFFFAOYSA-N Ethylene glycol distearate Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCCOC(=O)CCCCCCCCCCCCCCCCC FPVVYTCTZKCSOJ-UHFFFAOYSA-N 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- 229920000663 Hydroxyethyl cellulose Polymers 0.000 description 1
- 239000004354 Hydroxyethyl cellulose Substances 0.000 description 1
- JVTAAEKCZFNVCJ-UHFFFAOYSA-M Lactate Chemical compound CC(O)C([O-])=O JVTAAEKCZFNVCJ-UHFFFAOYSA-M 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- AOMUHOFOVNGZAN-UHFFFAOYSA-N N,N-bis(2-hydroxyethyl)dodecanamide Chemical compound CCCCCCCCCCCC(=O)N(CCO)CCO AOMUHOFOVNGZAN-UHFFFAOYSA-N 0.000 description 1
- 239000004435 Oxo alcohol Substances 0.000 description 1
- UAJFTICTIXLVSD-UHFFFAOYSA-N P(=O)(O)(O)O.OCCC[Na] Chemical compound P(=O)(O)(O)O.OCCC[Na] UAJFTICTIXLVSD-UHFFFAOYSA-N 0.000 description 1
- GOOHAUXETOMSMM-UHFFFAOYSA-N Propylene oxide Chemical compound CC1CO1 GOOHAUXETOMSMM-UHFFFAOYSA-N 0.000 description 1
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 1
- DWAQJAXMDSEUJJ-UHFFFAOYSA-M Sodium bisulfite Chemical compound [Na+].OS([O-])=O DWAQJAXMDSEUJJ-UHFFFAOYSA-M 0.000 description 1
- AWMVMTVKBNGEAK-UHFFFAOYSA-N Styrene oxide Chemical compound C1OC1C1=CC=CC=C1 AWMVMTVKBNGEAK-UHFFFAOYSA-N 0.000 description 1
- QVFOGJXHRGAFSH-UHFFFAOYSA-N acetic acid;1-dodecoxydodecane Chemical compound CC(O)=O.CCCCCCCCCCCCOCCCCCCCCCCCC QVFOGJXHRGAFSH-UHFFFAOYSA-N 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 239000004480 active ingredient Substances 0.000 description 1
- VLLNJDMHDJRNFK-UHFFFAOYSA-N adamantan-1-ol Chemical compound C1C(C2)CC3CC2CC1(O)C3 VLLNJDMHDJRNFK-UHFFFAOYSA-N 0.000 description 1
- 125000005073 adamantyl group Chemical group C12(CC3CC(CC(C1)C3)C2)* 0.000 description 1
- 230000032683 aging Effects 0.000 description 1
- 229910000272 alkali metal oxide Inorganic materials 0.000 description 1
- 229910001860 alkaline earth metal hydroxide Inorganic materials 0.000 description 1
- 150000001342 alkaline earth metals Chemical class 0.000 description 1
- ZRGUXTGDSGGHLR-UHFFFAOYSA-K aluminum;triperchlorate Chemical compound [Al+3].[O-]Cl(=O)(=O)=O.[O-]Cl(=O)(=O)=O.[O-]Cl(=O)(=O)=O ZRGUXTGDSGGHLR-UHFFFAOYSA-K 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- ZETCGWYACBNPIH-UHFFFAOYSA-N azane;sulfurous acid Chemical compound N.OS(O)=O ZETCGWYACBNPIH-UHFFFAOYSA-N 0.000 description 1
- DSAJWYNOEDNPEQ-UHFFFAOYSA-N barium atom Chemical compound [Ba] DSAJWYNOEDNPEQ-UHFFFAOYSA-N 0.000 description 1
- RQPZNWPYLFFXCP-UHFFFAOYSA-L barium dihydroxide Chemical compound [OH-].[OH-].[Ba+2] RQPZNWPYLFFXCP-UHFFFAOYSA-L 0.000 description 1
- 229910001863 barium hydroxide Inorganic materials 0.000 description 1
- YSJGOMATDFSEED-UHFFFAOYSA-M behentrimonium chloride Chemical compound [Cl-].CCCCCCCCCCCCCCCCCCCCCC[N+](C)(C)C YSJGOMATDFSEED-UHFFFAOYSA-M 0.000 description 1
- 238000009835 boiling Methods 0.000 description 1
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- 230000005587 bubbling Effects 0.000 description 1
- AXCZMVOFGPJBDE-UHFFFAOYSA-L calcium dihydroxide Chemical compound [OH-].[OH-].[Ca+2] AXCZMVOFGPJBDE-UHFFFAOYSA-L 0.000 description 1
- 239000000920 calcium hydroxide Substances 0.000 description 1
- 229910001861 calcium hydroxide Inorganic materials 0.000 description 1
- 230000003750 conditioning effect Effects 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- HPXRVTGHNJAIIH-UHFFFAOYSA-N cyclohexanol Chemical compound OC1CCCCC1 HPXRVTGHNJAIIH-UHFFFAOYSA-N 0.000 description 1
- 125000003493 decenyl group Chemical group [H]C([*])=C([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000002704 decyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- SWWQNNDPJXYCNJ-UHFFFAOYSA-N didodecylazanium;chloride Chemical compound Cl.CCCCCCCCCCCCNCCCCCCCCCCCC SWWQNNDPJXYCNJ-UHFFFAOYSA-N 0.000 description 1
- REZZEXDLIUJMMS-UHFFFAOYSA-M dimethyldioctadecylammonium chloride Chemical compound [Cl-].CCCCCCCCCCCCCCCCCC[N+](C)(C)CCCCCCCCCCCCCCCCCC REZZEXDLIUJMMS-UHFFFAOYSA-M 0.000 description 1
- USIUVYZYUHIAEV-UHFFFAOYSA-N diphenyl ether Chemical compound C=1C=CC=CC=1OC1=CC=CC=C1 USIUVYZYUHIAEV-UHFFFAOYSA-N 0.000 description 1
- 238000004851 dishwashing Methods 0.000 description 1
- 229940079886 disodium lauryl sulfosuccinate Drugs 0.000 description 1
- KHIQYZGEUSTKSB-UHFFFAOYSA-L disodium;4-dodecoxy-4-oxo-3-sulfobutanoate Chemical compound [Na+].[Na+].CCCCCCCCCCCCOC(=O)C(S(O)(=O)=O)CC([O-])=O.CCCCCCCCCCCCOC(=O)C(S(O)(=O)=O)CC([O-])=O KHIQYZGEUSTKSB-UHFFFAOYSA-L 0.000 description 1
- YVIGPQSYEAOLAD-UHFFFAOYSA-L disodium;dodecyl phosphate Chemical compound [Na+].[Na+].CCCCCCCCCCCCOP([O-])([O-])=O YVIGPQSYEAOLAD-UHFFFAOYSA-L 0.000 description 1
- 239000004664 distearyldimethylammonium chloride (DHTDMAC) Substances 0.000 description 1
- 125000005066 dodecenyl group Chemical group C(=CCCCCCCCCCC)* 0.000 description 1
- GVGUFUZHNYFZLC-UHFFFAOYSA-N dodecyl benzenesulfonate;sodium Chemical compound [Na].CCCCCCCCCCCCOS(=O)(=O)C1=CC=CC=C1 GVGUFUZHNYFZLC-UHFFFAOYSA-N 0.000 description 1
- JZKFHQMONDVVNF-UHFFFAOYSA-N dodecyl sulfate;tris(2-hydroxyethyl)azanium Chemical compound OCCN(CCO)CCO.CCCCCCCCCCCCOS(O)(=O)=O JZKFHQMONDVVNF-UHFFFAOYSA-N 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 125000000816 ethylene group Chemical group [H]C([H])([*:1])C([H])([H])[*:2] 0.000 description 1
- 230000001815 facial effect Effects 0.000 description 1
- 229960002989 glutamic acid Drugs 0.000 description 1
- 229940075507 glyceryl monostearate Drugs 0.000 description 1
- 229940100608 glycol distearate Drugs 0.000 description 1
- 229910052736 halogen Inorganic materials 0.000 description 1
- 150000002367 halogens Chemical class 0.000 description 1
- 239000008233 hard water Substances 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 235000019447 hydroxyethyl cellulose Nutrition 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- PNDPGZBMCMUPRI-UHFFFAOYSA-N iodine Chemical compound II PNDPGZBMCMUPRI-UHFFFAOYSA-N 0.000 description 1
- 230000007794 irritation Effects 0.000 description 1
- 229940031957 lauric acid diethanolamide Drugs 0.000 description 1
- VMPHSYLJUKZBJJ-UHFFFAOYSA-N lauric acid triglyceride Natural products CCCCCCCCCCCC(=O)OCC(OC(=O)CCCCCCCCCCC)COC(=O)CCCCCCCCCCC VMPHSYLJUKZBJJ-UHFFFAOYSA-N 0.000 description 1
- JXNPEDYJTDQORS-UHFFFAOYSA-N linoleyl alcohol Natural products CCCCCC=CCC=CCCCCCCCCO JXNPEDYJTDQORS-UHFFFAOYSA-N 0.000 description 1
- 125000005645 linoleyl group Chemical group 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- WRUGWIBCXHJTDG-UHFFFAOYSA-L magnesium sulfate heptahydrate Chemical compound O.O.O.O.O.O.O.[Mg+2].[O-]S([O-])(=O)=O WRUGWIBCXHJTDG-UHFFFAOYSA-L 0.000 description 1
- 229940061634 magnesium sulfate heptahydrate Drugs 0.000 description 1
- LPHFLPKXBKBHRW-UHFFFAOYSA-L magnesium;hydrogen sulfite Chemical compound [Mg+2].OS([O-])=O.OS([O-])=O LPHFLPKXBKBHRW-UHFFFAOYSA-L 0.000 description 1
- JESHZQPNPCJVNG-UHFFFAOYSA-L magnesium;sulfite Chemical compound [Mg+2].[O-]S([O-])=O JESHZQPNPCJVNG-UHFFFAOYSA-L 0.000 description 1
- 229910021645 metal ion Inorganic materials 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 230000003020 moisturizing effect Effects 0.000 description 1
- 239000001788 mono and diglycerides of fatty acids Substances 0.000 description 1
- 125000001421 myristyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 229910052759 nickel Inorganic materials 0.000 description 1
- 125000001196 nonadecyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000001400 nonyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- SBOJXQVPLKSXOG-UHFFFAOYSA-N o-amino-hydroxylamine Chemical compound NON SBOJXQVPLKSXOG-UHFFFAOYSA-N 0.000 description 1
- 125000004365 octenyl group Chemical group C(=CCCCCCC)* 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 229940055577 oleyl alcohol Drugs 0.000 description 1
- XMLQWXUVTXCDDL-UHFFFAOYSA-N oleyl alcohol Natural products CCCCCCC=CCCCCCCCCCCO XMLQWXUVTXCDDL-UHFFFAOYSA-N 0.000 description 1
- 125000001117 oleyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])/C([H])=C([H])\C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 125000000962 organic group Chemical group 0.000 description 1
- 125000000913 palmityl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000002304 perfume Substances 0.000 description 1
- 230000035699 permeability Effects 0.000 description 1
- 229920001495 poly(sodium acrylate) polymer Polymers 0.000 description 1
- 229920001296 polysiloxane Polymers 0.000 description 1
- BHZRJJOHZFYXTO-UHFFFAOYSA-L potassium sulfite Chemical compound [K+].[K+].[O-]S([O-])=O BHZRJJOHZFYXTO-UHFFFAOYSA-L 0.000 description 1
- 235000019252 potassium sulphite Nutrition 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 230000002335 preservative effect Effects 0.000 description 1
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical group CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 1
- 150000003242 quaternary ammonium salts Chemical class 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- BTURAGWYSMTVOW-UHFFFAOYSA-M sodium dodecanoate Chemical compound [Na+].CCCCCCCCCCCC([O-])=O BTURAGWYSMTVOW-UHFFFAOYSA-M 0.000 description 1
- 229940080264 sodium dodecylbenzenesulfonate Drugs 0.000 description 1
- 229940079827 sodium hydrogen sulfite Drugs 0.000 description 1
- 229940082004 sodium laurate Drugs 0.000 description 1
- 229910052938 sodium sulfate Inorganic materials 0.000 description 1
- 235000011152 sodium sulphate Nutrition 0.000 description 1
- HLWRUJAIJJEZDL-UHFFFAOYSA-M sodium;2-[2-[bis(carboxymethyl)amino]ethyl-(carboxymethyl)amino]acetate Chemical compound [Na+].OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC([O-])=O HLWRUJAIJJEZDL-UHFFFAOYSA-M 0.000 description 1
- 238000007711 solidification Methods 0.000 description 1
- 230000008023 solidification Effects 0.000 description 1
- 229940035044 sorbitan monolaurate Drugs 0.000 description 1
- 125000004079 stearyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 125000001273 sulfonato group Chemical group [O-]S(*)(=O)=O 0.000 description 1
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 1
- KWXLCDNSEHTOCB-UHFFFAOYSA-J tetrasodium;1,1-diphosphonatoethanol Chemical compound [Na+].[Na+].[Na+].[Na+].[O-]P(=O)([O-])C(O)(C)P([O-])([O-])=O KWXLCDNSEHTOCB-UHFFFAOYSA-J 0.000 description 1
- 125000005040 tridecenyl group Chemical group C(=CCCCCCCCCCCC)* 0.000 description 1
- 125000002889 tridecyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- JSPLKZUTYZBBKA-UHFFFAOYSA-N trioxidane Chemical compound OOO JSPLKZUTYZBBKA-UHFFFAOYSA-N 0.000 description 1
- 239000006097 ultraviolet radiation absorber Substances 0.000 description 1
- 229940057402 undecyl alcohol Drugs 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
Classifications
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P20/00—Technologies relating to chemical industry
- Y02P20/50—Improvements relating to the production of bulk chemicals
- Y02P20/52—Improvements relating to the production of bulk chemicals using catalysts, e.g. selective catalysts
Landscapes
- Cosmetics (AREA)
- Emulsifying, Dispersing, Foam-Producing Or Wetting Agents (AREA)
- Detergent Compositions (AREA)
- Low-Molecular Organic Synthesis Reactions Using Catalysts (AREA)
Description
【0001】
【発明の属する技術分野】
本発明はアニオン性界面活性剤およびそれを含有する洗浄剤組成物に関する。
【0002】
【従来の技術】
従来、脂肪族系アルコール類にアルキレンオキサイドを付加重合させて得られる、脂肪族系アルコールアルキレンオキサイド付加物を疎水基とするスルホコハク酸エステル塩型アニオン性界面活性剤を含有する洗浄剤組成物からなる、シャンプー、食器用洗浄剤、金属等の硬質表面洗浄剤が知られている(例えば、特開平1−259098号公報)。
【0003】
【発明が解決しようとする課題】
しかしながら、これらの洗浄剤組成物に配合されるスルホコハク酸エステル塩型アニオン性界面活性剤は、低温で長期保存した場合に、外観がかすむ等の製品の経日安定性や、起泡性、洗浄性、人体の皮膚刺激性について問題が残されていた。
【0004】
【課題を解決するための手段】
本発明者らは、上記の課題を解決すべく鋭意検討した結果、特定の脂肪族系アルコールアルキレンオキサイド付加物を疎水基とするスルホコハク酸エステル塩型アニオン性界面活性剤が、製品の経日安定性、その他の問題が解決できることを見いだし、本発明に到達した。
【0005】
すなわち本発明は、脂肪族系アルコールアルキレンオキサイド付加物(A)のスルホコハク酸エステル塩(B)からなり;(A)が、触媒(d)の存在下、脂肪族系アルコール(a1)に炭素数2以上のアルキレンオキサイド(b2)を平均1〜2.5モル付加させて直接製造され下記(i)〜(iii)を満たす脂肪族系アルコールアルキレンオキサイド付加物(A1)、または(A1)に、アルカリ触媒(e)の存在下、炭素数2以上のアルキレンオキサイド(b3)を付加反応させてなり下記(i)〜(iii)を満たす脂肪族系アルコールアルキレンオキサイド付加物(A2)であることを特徴とするアニオン性界面活性剤;過ハロゲン酸(塩)、硫酸塩、燐酸塩および硝酸塩からなる群から選ばれる1種以上である触媒(d)の存在下に炭素数8〜24の脂肪族系アルコール(a1)に炭素数2以上のアルキレンオキサイド(b2)を平均1〜2.5モル付加させて直接製造され、下記式(4)から求められる分布定数cが1.0以下である脂肪族系アルコールアルキレンオキサイド付加物(A1)または(A1)にアルカリ触媒(e)の存在下に炭素数2以上のアルキレンオキサイド(b3)[(b3)は(b2)と同じであっても異なっていてもよい]を付加反応させてなる脂肪族系アルコールアルキレンオキサイド付加物(A2)であって、下記(i)〜(iii)を満たす脂肪族系アルコールアルキレンオキサイド付加物(A)を、未反応アルコールや付加モル数の異なるものを分別する操作なしで、スルホコハク酸エステル塩化して、(A1)もしくは(A2)のスルホコハク酸エステル塩(B)を製造することを特徴とするアニオン性界面活性剤の製造方法;上記アニオン性界面活性剤からなる洗浄剤組成物;並びに上記洗浄剤組成物からなる皮膚洗浄剤、毛髪洗浄剤または家庭用洗剤である。
【0006】
▲1▼脂肪族系アルコール(a1)のアルキレンオキサイド(b1)付加物であって、下記一般式(1)で表される化合物の1種または2種以上の混合物からなる。
R1−(OA)k−OH (1)
[式中、R1 は炭素数8〜24の脂肪族炭化水素基または炭素数8〜24の脂環式炭化水素基;Aは炭素数2以上の1種以上のアルキレン基;kは平均が1〜20となる0または1以上の整数]
▲2▼重量平均分子量(Mw)と数平均分子量(Mn)との比:Mw/Mnが下記関係式(2)または(3)を満たす。
Mw/Mn≦0.030×Ln(v)+1.010 (但し、v<10)(2)
Mw/Mn≦−0.026×Ln(v)+1.139(但し、v≧10)(3)
[但し、vは脂肪族系アルコール(a1)1モル当たりに付加したアルキレンオキサイドの平均付加モル数を表し、上記一般式(1)でのkの平均に相当する。]
▲3▼下記式(4)から求められる分布定数cが2.0以下である。(本項はvが12以下の場合のみ適用する。)
c=(v+n0/n00−1)/[Ln(n00/n0)+n0/n00−1] (4)
[但し、vは上記に同じ、n00は反応に用いた(a1)のモル数、n0 は未反応の(a1)のモル数を表す。]
【0007】
【発明の実施の形態】
本発明のアニオン性界面活性剤において、スルホコハク酸エステル塩(B)は、上記▲1▼〜▲3▼を満たす(A)のスルホコハク酸エステル塩であれば、特に限定されないが、下記一般式(5)で表される塩の1種以上であることが界面活性能の点から好ましい。
[式中、R1 は炭素数8〜24の脂肪族炭化水素基または炭素数8〜24の脂環式炭化水素基;Aは炭素数2以上の1種以上のアルキレン基;kは平均が1〜20となる0または1以上の整数;Z1 およびはZ2 は、いずれか一方がH、もう一方がSO3Q1基を表し、XはQ2 またはR1−(OA)k−基を表す。Q1 およびQ2 は、Hまたはカチオン性対イオンを表す。〕
【0008】
(B)は、さらに、下記一般式(5a)および/または(5b)で表される、モノエステル化物を80〜100質量%、特に90〜100質量%含有し、一般式(5c)で表されるジエステル化物を0〜20質量%、特に0〜10質量%含有するものが好ましい。また、(5a)/(5b)の質量%の比は、好ましくは10〜90/90〜10、さらに好ましくは30〜70/70〜30である。
[式中、R1 、A、k、Q1 およびQ2 は一般式(5)に同じ。〕
【0009】
前記一般式(5)において、R1−(OA)k−O−基は、脂肪族系アルコールアルキレンオキサイド付加物(A)の残基である。Z1 またはZ2 基を形成するSO3Q1基は、例えば、(A)と無水マレイン酸とを反応させたエステル化物をスルホン化することにより導入されるスルホン酸塩基である。Q1 およびQ2 基としては、用いるスルホン化剤の種類により異なるが、水素およびカチオン性対イオンが挙げられる。カチオン性対イオンとしては、例えば、アルカリ金属(ナトリウム、カリウム、リチウムなど)イオン、アルカリ土類金属(マグネシウム、カルシウム、バリウムなど)イオンなどの金属イオン;アンモニウムイオンなどが挙げられる。
【0010】
本発明において、(A)は、脂肪族系アルコール(a1)(本明細書中、脂肪族系アルコールとは、脂肪族アルコールと脂環式アルコールの両者を含むものとする。)にアルキレンオキサイド(b1)を付加して得られる脂肪族系アルコールアルキレンオキサイド付加物の1種または2種以上の混合物であり、下記一般式(1)で表される。
R1−(OA)k−OH (1)
[式中、R1 は炭素数8〜24の脂肪族炭化水素基または炭素数8〜24の脂環式炭化水素基;Aは炭素数2以上の1種以上のアルキレン基;kは平均が1〜20となる0または1以上の整数]
【0011】
上記一般式(1)において、R1 は、脂肪族系アルコール(a1)の残基であり、炭素数が通常8〜24の脂肪族炭化水素基(アルキル基、アルケニル基、アルカジエニル基など)または脂環式炭化水素基(シクロアルキル基、多環炭化水素基など)を表す。R1 は、直鎖状と分岐状など2種以上の基の混合物であってもよい。炭素数8未満では、十分な起泡力、洗浄力が得られず、炭素数が24を超えると起泡力、洗浄力が劣り、好ましくない。
R1 の具体例としては、アルキル基としては、オクチル、ノニル、デシル、ラウリル、トリデシル、ミリスチル、セチル、ステアリル、ノナデシル、2−エチルヘキシル、2−エチルオクチル基などが挙げられる。アルケニル基としては、オクテニル、デセニル、ドデセニル、トリデセニル、ペンタデセニル、オレイル、ガドレイル基などが挙げられる。アルカジエニル基としては、リノレイル基などが挙げられる。シクロアルキル基としては、エチルシクロヘキシル、プロピルシクロヘキシル、オクチルシクロヘキシル、ノニルシクロヘキシル基などが挙げられる。多環炭化水素基としては、アダマンチル基などが挙げられる。
【0012】
一般式(1)中、Aは炭素数2以上、好ましくは2〜8、さらに好ましくは2〜4、特に好ましくは炭素数2または3のアルキレン基を表し、(OA)の部分は、炭素数2以上のアルキレンオキサイド(b1)の付加により形成される。このようなアルキレンオキサイド(b1)としては、エチレンオキサイド(以下、EOと略記)、プロピレンオキサイド(以下、POと略記)、1,2−または2,3−ブチレンオキサイド、テトラヒドロフラン、スチレンオキサイドなどが挙げられる。好ましくはEOおよび/またはPOである。
一般式(1)中、kは(b1)の付加モル数に相当し、平均が1〜20となる整数であり、好ましくは1〜12である。kが20を超えると、十分な起泡力、洗浄力が得られず好ましくない。
【0013】
前記脂肪族系アルコール(a1)は、R1 残基を与えるものであり、炭素数が通常8〜24(好ましくは12〜18)のアルコールであり、天然アルコールでも合成アルコール(チーグラーアルコール、オキソアルコールなど)でもよい。具体例としては、オクチルアルコール、ノニルアルコール、デシルアルコール、ウンデシルアルコール、ドデシルアルコール、トリデシルアルコール、テトラデシルアルコール、ヘキサデシルアルコール、オクタデシルアルコール、ノナデシルアルコールなどの飽和脂肪族アルコール;オクテニルアルコール、デセニルアルコール、ドデセニルアルコール、トリデセニルアルコール、ペンタデセニルアルコール、オレイルアルコール、ガドレイルアルコール、リノレイルアルコールなどの不飽和脂肪族アルコール;エチルシクロヘキシルアルコール、プロピルシクロヘキシルアルコール、オクチルシクロヘキシルアルコール、ノニルシクロヘキシルアルコール、アダマンチルアルコールなどの環状脂肪族アルコールが挙げられ、これらの1種または2種以上が使用出来る。これら脂肪族系アルコールは1級または2級が好ましく、さらに1級が好ましい。また、アルキル基部分は直鎖状でも分岐状でもよい。特に好ましくはドデシルアルコール、トリデシルアルコール、テトラデシルアルコール、ヘキサデシルアルコール、オクタデシルアルコールである。
【0014】
(A)としては、工程が煩雑でないことから、(a1)と(b1)から直接製造されたものであることが好ましい。ここで、「直接製造された」とは、上記付加物が、精留などにより未反応アルコールや付加モル数の異なるものを分別する操作なしで、直接得られたものであることを意味する。但し、分別を目的としないで、簡単な操作で未反応アルキレンオキサイドや低沸点物をストリッピングしたものは分別操作に含まれない。
【0015】
また、本発明で、(A)は、重量平均分子量(Mw)と数平均分子量(Mn)との比:Mw/Mnが下記関係式(2)または(3)を満たす必要がある。
Mw/Mn≦0.030×Ln(v)+1.010 (但し、v<10)(2)
Mw/Mn≦−0.026×Ln(v)+1.139(但し、v≧10)(3)
これらの式で、Ln(v)はvの自然対数を意味し、vは脂肪族系アルコール(a1)1モル当たりに付加したアルキレンオキサイドの平均付加モル数を表し、前記一般式(1)でのアルキレンオキサイドの付加モル数であるkの平均に相当する。(以下同じ)
関係式(2)または(3)を満たさない、すなわち分子量分布が広くなると、アニオン性界面活性剤としたときに、充分な界面活性能が得られない。
また、Mw/Mnは下記関係式(2’)または(3’)を満たすことが好ましい。
Mw/Mn≦0.031×Ln(v)+1.000 (但し、v<10)(2’)
Mw/Mn≦−0.026×Ln(v)+1.129(但し、v≧10)(3’)
【0016】
一般式(1)においてAがエチレン基のみである場合、即ち、脂肪族系アルコール(a1)にエチレンオキサイドのみを付加した場合には、重量平均分子量(Mw)と数平均分子量(Mn)との比:Mw/Mnが関係式(2)または(3)に代わって、下記(6)または(7)を満たすことが好ましい。
Mw/Mn≦0.020×Ln(v)+1.010 (但し、v<10)(6)
Mw/Mn≦−0.026×Ln(v)+1.116(但し、v≧10)(7)
関係式(6)または(7)を満たす場合、即ち分子量分布が狭くなると、アニオン性界面活性剤としたときに、良好な界面活性能が得られる。
また、この場合、Mw/Mnは下記関係式(6’)または(7’)を満たすことがさらに好ましい。
Mw/Mn≦0.018×Ln(v)+1.015 (但し、v<10) (6’)
Mw/Mn≦−0.023×Ln(v)+1.113(但し、v≧10)(7’)
【0017】
さらに、(A)は、下記Weibullの分布則の式(8)から導き出される関係式(4)から分布定数cを求めることができるとき、cが2.0以下である必要がある。cは好ましくは1.0以下、さらに好ましくは0.7以下である。関係式(4)において、分布定数cの値が小さい、すなわち未反応の脂肪族系アルコールの含有量が少ないほど分子量分布が狭いことを意味する。
なお、この式は、未反応の脂肪族系アルコール(a1)の量が検出限界(0.001質量%)以上の場合に適用される式であり、(A)の場合はアルキレンオキサイド(b1)の平均付加モル数が12モル程度まで適用可能である。
cが2を超えると、アニオン性界面活性剤としたときに、充分な界面活性能が得られない。
v=c×Ln(n00/n0)−(c−1)×(1−n0/n00) (8)
c=(v+n0/n00−1)/[Ln(n00/n0)+n0/n00−1] (4)
これらの式で、Ln(n00/n0)は(n00/n0)の自然対数を意味し、vは上記に同じ、n00は反応に用いた脂肪族系アルコール(a1)のモル数、n0 は未反応の脂肪族系アルコール(a1)のモル数を表す。
【0018】
本発明において、上記(A)としては、さらに下記一般式(1’)で表される化合物であることが、起泡力の点で好ましい。
R1−[(OC2H4)m/(OD)n]−(OC2H4)p−OH (1’)
[式中、R1 は炭素数8〜24の脂肪族炭化水素基または炭素数8〜24の脂環式炭化水素基;Dは炭素数3または4のアルキレン基;mは平均が0〜5となる0または1以上の整数、nは平均が0〜5となる0または1以上の整数、pは平均が1〜10となる0または1以上の整数であり、(m+n+p)は平均が1〜20となる整数であり、(m+p)/(m+n+p)は平均0.5以上である。{(OC2H4)m/(0D)n}は、m≠0,n≠0のときブロック付加またはランダム付加を表す。]
【0019】
上記一般式(1’)中、(OC2H4)の部分は、EOの付加により形成される。Dは炭素数3または4のアルキレン基を表し、(OD)の部分は、炭素数3または4のアルキレンオキサイドの付加により形成される。このようなアルキレンオキサイドとしては、PO、1,2−もしくは2,3−ブチレンオキサイドなどが挙げられる。
【0020】
上記一般式(1’)中、mは平均が0〜5となる0または1以上の整数であり、さらに好ましくは0〜3の整数である。nは平均が0〜5となる0または1以上の整数であり、さらに好ましくは0または1である。pは平均が1〜10となる0または1以上の整数であり、さらに好ましくは1〜8である。(m+n+p)は平均が1〜20の整数であり、さらに好ましくは1〜12である。(m+p)/(m+n+p)は平均0.5以上であり、さらに好ましくは0.7以上である。0.5以上であると、アニオン性活性剤としたときの洗浄力が良好となる。{(OC2H4)m/(OD)n} の部分は、ブロック付加〔(OC2H4)m、(OD)nの順〕 でもランダム付加でもよいが、さらに好ましくはブロック付加である。
【0021】
本発明において、(A)としては、以下に述べる方法により製造された(A1)または(A2)であることが好ましい。
すなわち、前記の、Weibullの分布則から導かれる式(4)から求められる分布定数cが2.0以下となる付加物を与える触媒(d)の存在下、前記脂肪族系アルコール(a1)にアルキレンオキサイド(b2)を平均1〜2.5モル付加して得られる脂肪族系アルコールアルキレンオキサイド付加物(A1)であるか、(A1)にさらにアルカリ触媒(f)の存在下でアルキレンオキサイド(b3)を付加反応させてなる脂肪族系アルコールアルキレンオキサイド付加物(A2)であるのが好ましい。さらに好ましくは(A2)である。
【0022】
上記アルキレンオキサイド(b2)および(b3)としては、炭素数2以上のアルキレンオキサイドが挙げられる。具体例としては、一般式(1)におけるOAを形成するアルキレンオキサイド(b1)の例として前記したアルキレンオキサイドが挙げられ、2種以上を併用してもよい。2種以上用いる場合は、ブロック付加でもランダム付加でもよいが、好ましくはブロック付加である。これらのうちで好ましくは、EOおよび/またはPOである。
【0023】
触媒(d)としては、得られるアルキレンオキサイド付加物の分布定数cが2.0以下となるものを用いる。好ましくはcが1.0以下、さらに好ましくは0.7以下、特に好ましくは0.45以下となるものである。分布定数cが2.0を超えるものを用いると、アニオン性界面活性剤とした場合に、低温でカスミを生じたり、起泡性、洗浄性が低下する。また、皮膚に対する刺激性が低くならず、問題となることがある。
【0024】
触媒(d)の具体例としては、過ハロゲン酸もしくはその塩、硫酸塩、燐酸塩および硝酸塩が挙げられる。塩を形成する場合の金属は、特に限定されるものではないが、アルカリ金属以外のものが好ましく、2価または3価の金属が好ましい。
これら金属として好ましくは、Mg、Ca、Sr、Ba、Zn、Co、Ni、Cu、Alであり、より好ましくは、Mg、Zn、Ca、Sr、Ba、Alであり、特に好ましくは、Mg、Zn、Alである。過ハロゲン酸(塩)のハロゲンとしては塩素、臭素、沃素が挙げられ、塩素が好ましい。
したがって、(d)としては、2価もしくは3価の金属の過塩素酸塩が好ましく、Mg、ZnおよびAlから選ばれる金属の過塩素酸塩がさらに好ましい。また、(d)に2価もしくは3価の金属アルコラートを併用してもよい。金属アルコラートのアルキル基としては、アルコールとして留去し易い低級(炭素数1〜4)アルキル基、または原料脂肪族系アルコールと同一組成のアルキル基が挙げられる。これらの触媒は1種でもよいが、2種以上の触媒〔たとえば、過塩素酸マグネシウム/硫酸マグネシウム7水塩=95/5〜50/50、過塩素酸マグネシウム/過塩素酸アルミニウム=99/1〜30/70(いずれも質量比)〕を併用した方が好ましい。
【0025】
触媒(d)の使用量としては、反応速度と経済性の点から、(a1)と(b2)の合計100質量部当たり、0.001〜1質量部が好ましい。さらに好ましくは0.003〜0.8質量部、特に好ましくは0.005〜0.5質量部である。
【0026】
(a1)に(b2)を付加して得られるアルキレンオキサイド付加物(A1)に、アルキレンオキサイド(b3)を付加させる際に用いる触媒は、アルカリ触媒(e)である。
アルカリ触媒(e)としては、アルカリ金属およびアルカリ土類金属の水酸化物、たとえば、水酸化リチウム、水酸化ナトリウム、水酸化カリウム、水酸化セシウム、水酸化マグネシウム、水酸化カルシウム、水酸化バリウムなどが挙げられるが、より好ましくは水酸化カリウム、水酸化セシウムである。
触媒(e)の使用量としては、反応速度と経済性の点から、(A1)と(b3)の合計100質量部当たり、0.0001〜1質量部が好ましい。さらに好ましくは0.001〜0.8質量部である。
【0027】
(a1)と(b2)を反応させる場合の反応条件としては、(a1)と(d)を混合し、窒素置換を行った後、−0.8〜5kgf/cm2 Gで、80〜200℃で(b2)を導入し、所定量の(b2)を投入後、80〜200℃で反応系内の圧力が平衡になるまで熟成を行う方法などが挙げられる。
このようにして得られたアルキレンオキサイド付加物(A1)に、アルカリ触媒(e)を添加し、アルキレンオキサイド(b3)を、上記と同様の方法で反応することで、脂肪族系アルコールアルキレンオキサイド付加物(A2)が得られる。
【0028】
(A)からスルホコハク酸エステル塩(B)を得る方法は、特に限定されないが、一例としては、(A)と無水マレイン酸(以下MAと略記することがある)を反応させてエステル化物を得る工程と、水の存在下、亜硫酸塩や酸性亜硫酸塩を用いスルホン化する工程との二段工程で反応する方法が挙げられる。
(A)とMAとの反応は、反応温度が、通常50〜100℃、好ましくは60〜90℃で、反応時間は反応温度によって異なるが、通常1〜15時間、好ましくは3〜7時間で、窒素雰囲気中で行い、(A)のマレイン酸エステルを得る。MAを過剰に加えた場合は、エステル化反応終了後、減圧下脱MAを行う。(A)とMAとのモル比は、前述したように、モノエステル化物の含量が高いものが好ましいことから、好ましくは1.0:0.9〜1.0:1.1、さらに好ましくは1.0:0.95〜1.0:1.05である。
エステル化反応の終点は、[56100/(エステル化物の分子量)]で表される酸価(AV)が、理論値の90〜110%となる点であり、好ましくは
95〜105%となる点である。
【0029】
次いで、亜硫酸塩または酸性亜硫酸塩を用いて生成したエステル化物のスルホン化を行う。亜硫酸塩としては、亜硫酸ナトリウム、亜硫酸カリウム等のアルカリ金属塩、亜硫酸マグネシウム等のアルカリ土類金属塩、亜硫酸アンモニウム等のアンモニアの塩等があげられ、酸性亜硫酸塩としては、亜硫酸水素ナトリウム、亜硫酸水素カリウム、亜硫酸水素アンモニウム、亜硫酸水素マグネシウムなどの亜硫酸水素塩(アルカリ金属塩、アルカリ土類金属塩、アンモニウム塩)が挙げられる。スルホン化は、水の存在下、エステル化物と亜硫酸塩または亜硫酸水素塩を反応させることによって行うことができる。エステル化物と亜硫酸塩もしくは亜硫酸水素塩とのモル比は、通常1.0:0.9〜1.0:1.1、好ましくは1.0:0.95〜1.0:1.05であり、反応温度は、通常30〜90℃、好ましくは40〜80℃であり、通常、窒素雰囲気化で行う。反応時間は反応温度によって異なるが、通常1〜10時間、好ましくは2〜5時間である。
スルホン化の終点は、[(80×100)/スルホン化物の分子量]で表される結合硫酸量が、理論値の90〜110%となる点、好ましくは、95〜105%となる点である。スルホン化が終了した後、クエン酸などの有機酸、またはトリエタノールアミンなどのアルカノールアミンでpHを中性にする。
【0030】
本発明のスルホコハク酸エステル塩(B)からなるアニオン性界面活性剤は、起泡性、洗浄性に優れる。また、▲1▼〜▲3▼を満たす脂肪族系アルコールアルキレンオキサイド付加物(A)中にはその原料の脂肪族系アルコールが少ないので、そのスルホコハク酸エステル塩中にも少なく、人体の皮膚刺激性の少ないものとなる。また、臭気も改善される。
【0031】
本発明の(B)からなるアニオン性界面活性剤は、単独で洗浄剤として用いてもよいが、必要により、本発明の効果を妨げない量の本発明以外のアニオン性界面活性剤、ノニオン性界面活性剤、カチオン性界面活性剤、両性界面活性剤から選ばれる1種以上の界面活性剤を配合して洗浄剤組成物とすることができる。また、洗浄剤組成物のpHは特に限定されないが、人体に対する安全性の面から中性〜弱酸性(例えば、pH4〜9)が好ましい。
洗浄剤組成物中の界面活性剤有効成分(固形分換算)の配合割合(質量%)は、本発明のアニオン性界面活性剤が、好ましくは3〜60%であり、さらに好ましくは5〜50%である。本発明以外のアニオン性界面活性剤を用いる場合、本発明のアニオン性界面活性剤との合計量に対して、好ましくは60%以下、さらに好ましくは50%以下であり、かつアニオン性界面活性剤の合計の配合割合が上記範囲内であることが好ましい。また、ノニオン性界面活性剤は、好ましくは60%以下であり、さらに好ましくは1〜30%である。カチオン性界面活性剤は、好ましくは5%以下であり、さらに好ましくは0.05〜3%である。両性界面活性剤は、好ましくは50%以下であり、さらに好ましくは1〜30%である。
【0032】
具体的には、本発明以外のその他のアニオン性界面活性剤としては、炭素数8〜24の炭化水素系エーテルカルボン酸またはその塩、[(ポリ)オキシエチレン(重合度=1〜100)ラウリルエーテル酢酸ナトリウム、(ポリ)オキシエチレン(重合度=1〜100)ラウリルスルホコハク酸2ナトリウム等]、炭素数8〜24の炭化水素系硫酸エステル塩[ラウリル硫酸ナトリウム、(ポリ)オキシエチレン(重合度=1〜100)ラウリル硫酸ナトリウム、(ポリ)オキシエチレン(重合度=1〜100)ラウリル硫酸トリエタノールアミン、(ポリ)オキシエチレン(重合度=1〜100)ヤシ油脂肪酸モノエタノールアミド硫酸ナトリウム、]、炭素数8〜24の炭化水素系スルホン酸塩[ドデシルベンゼンスルホン酸ナトリウム等]及び炭素数8〜24の炭化水素系リン酸エステル塩[ラウリルリン酸ナトリウム、(ポリ)オキシエチレン(重合度=1〜100)ラウリルエーテルリン酸ナトリウム等]、脂肪酸塩[ラウリン酸ナトリウム、ラウリン酸トリエタノールアミン等]、アシル化アミノ酸塩[ヤシ油脂肪酸メチルタウリンナトリウム、ヤシ油脂肪酸サルコシンナトリウム、ヤシ油脂肪酸サルコシントリエタノールアミン、N−ヤシ油脂肪酸アシル−L−グルタミン酸トリエタノールアミン、N−ヤシ油脂肪酸アシル−L−グルタミン酸ナトリウム、ラウロイルメチル−β−アラニンナトリウム等]、その他[スルホコハク酸(ポリ)オキシエチレン(重合度=1〜100)ラウロイルエタノールアミド2ナトリウム等]等が挙げられる。
【0033】
ノニオン性界面活性剤としては、脂肪族系アルコール(炭素数8〜24)アルキレンオキサイド(炭素数2〜8)付加物(重合度=1〜100)、(ポリ)オキシアルキレン(炭素数2〜8、重合度=1〜100)高級脂肪酸(炭素数8〜24)エステル[モノステアリン酸ポリエチレングリコール(重合度=20)、ジステアリン酸ポリエチレングリコール(重合度=30)等]、多価(2価〜10価またはそれ以上)アルコール脂肪酸(炭素数8〜24)エステル[モノステアリン酸グリセリン、モノステアリン酸エチレングリコール、モノラウリン酸ソルビタン等]、(ポリ)オキシアルキレン(炭素数2〜8,重合度=1〜100)多価(2価〜10価またはそれ以上)アルコール高級脂肪酸(炭素数8〜24)エステル[モノラウリン酸ポリオキシエチレン(重合度=10)ソルビタン、ポリオキシエチレン(重合度=50)ジオレイン酸メチルグルコシド等]、脂肪酸アルカノールアミド[1:1型ヤシ油脂肪酸ジエタノールアミド、1:1型ラウリン酸ジエタノールアミド等]、(ポリ)オキシアルキレン(炭素数2〜8、重合度=1〜100)アルキル(炭素数1〜22)フェニルエーテル、(ポリ)オキシアルキレン(炭素数2〜8、重合度=1〜100)アルキル(炭素数8〜24)アミノエーテルおよびアルキル(炭素数8〜24)ジアルキル(炭素数1〜6)アミンオキシド[ラウリルジメチルアミンオキシド等]等が挙げられる。
【0034】
カチオン性界面活性剤としては、第4級アンモニウム塩型[塩化ステアリルトリメチルアンモニウム、塩化ベヘニルトリメチルアンモニウム、塩化ジステアリルジメチルアンモニウム、エチル硫酸ラノリン脂肪酸アミノプロピルエチルジメチルアンモニウム等]、アミン塩型[ステアリン酸ジエチルアミノエチルアミド乳酸塩、ジラウリルアミン塩酸塩、オレイルアミン乳酸塩等]等が挙げられる。両性界面活性剤としては、ベタイン型両性界面活性剤[ヤシ油脂肪酸アミドプロピルジメチルアミノ酢酸ベタイン、ラウリルジメチルアミノ酢酸ベタイン、2−アルキル−N−カルボキシメチル−N−ヒドロキシエチルイミダゾリニウムベタイン、ラウリルヒドロキシスルホベタイン、ラウロイルアミドエチルヒドロキシエチルカルボキシメチルベタインヒドロキシプロピルリン酸ナトリウム等]、アミノ酸型両性界面活性剤[β−ラウリルアミノプロピオン酸ナトリウム等]が挙げられる。これらの1種または2種以上が使用出来る。
【0035】
また、本発明の洗浄剤組成物には、従来から公知の添加剤を1種以上配合して使用することができる。このようなものとしては、保湿剤としてグリセリン、ピロリドンカルボン酸ナトリウムなど;コンディショニング剤として使用する高分子化合物として、重量平均分子量500〜500万の範囲のカチオン化セルロース、カチオン化グアーガム、ポリエチレングリコール、ポリアクリル酸ナトリウム、ヒドロキシエチルセルロース、タンパク質誘導体など;シリコンとして、ジメチルポリシロキサン、ジメチルポリシロキサンのメチル基の一部に各種の有機基を導入した変性シリコーン、環状ジメチルシロキサンなど;キレート剤として、エチレンジアミン四酢酸ナトリウム、1−ヒドロキシエタン−1、1−ジホスホン酸ナトリウムなど;低級アルコール類として、エタノール、プロピレングリコール、ジプロピレングリコールなどの炭素数2〜6のアルコール類;香料、着色剤、防腐剤、紫外線吸収剤、水などが挙げられる。
【0036】
本発明の洗浄剤組成物の形態は、液体、ペースト、固体、粉末など特に限定されないが、液体およびペーストが使いやすく好ましい。
例えば、液体およびペースト状のシャンプー用組成物の場合、本発明のアニオン性界面活性剤が好ましくは5〜30質量%、本発明の界面活性剤を含む界面活性剤の合計量が好ましくは5〜50質量%、さらに好ましくは、10〜30質量%;高分子化合物、シリコン、香料、着色剤、防腐剤および紫外線吸収剤が好ましくは各々0〜5質量%、保湿剤、キレート剤および低級アルコール類が好ましくは各々0〜10質量%、これらの界面活性剤以外の添加剤の合計が好ましくは0〜30質量%、さらに好ましくは1〜25質量%;水が好ましくは35〜95質量%用いられる。
【0037】
本発明の洗浄剤組成物の用途も特に限定されないが、例えば、ボディシャンプー、洗顔料などの皮膚洗浄剤;シャンプーなどの毛髪洗浄剤;食器用洗剤、衣料用洗剤などの家庭用洗剤として用いられる。
【0038】
【実施例】
以下、実施例により本発明をさらに詳細に説明するが、本発明はこれに限定されるものではない。部は質量部を示す。
ゲルパーミエーションクロマトグラフ(GPC)による分子量の測定、ガスクロマトグラフ(GC)による未反応の脂肪族系アルコールの含有量の測定は次の通りである。
【0039】
【0040】
《 GCの測定条件》
カラム シリコンGE−SE30
検出器 :FID
Injection :280℃
昇温速度 :100〜250℃/10℃毎分
内部標準 :オクタノール
【0041】
製造例1
撹拌および温度調節機能の付いたステンレス製オートクレーブに、ラウリルアルコール186部(1モル)、過塩素酸マグネシウム0.32部および水酸化マグネシウム0.03部を投入し、混合系内を窒素で置換した後、減圧下(20mmHg)、120℃にて1時間脱水を行った。次いでEO88部(2モル)を150℃にて、ゲージ圧が1〜3kgf/cm2 となるように導入した。EOの付加重合に要した時間は10時間であった。
得られた反応物に水酸化カリウム1.3部を加え、EO44部(1モル)を130℃で反応させた。反応物を前記GPCの測定条件によって測定した結果、Mw/Mnは1.037[一般式(2)をみたすMw/Mnの上限計算値1.043]、前記GCの測定条件によって測定した未反応のラウリルアルコールは全反応物中の2.35質量%(0.040モル)であった。(定数c=0.910)ガラス容器に上記反応物176部(0.55モル)と無水マレイン酸57部(0.58モル)を仕込み、65℃で8時間反応させモノエステル化物を得た。その後、亜硫酸ナトリウム77部(0.61モル)を水690部に溶解し、60℃で上記モノエステル化物233部を加え、同温で約5時間反応させスルホン化物を得た。その後、クエン酸でpHを6.5にし、本発明に係わるスルホコハク酸エステル塩の約30%水溶液を得た。
【0042】
製造例2
撹拌および温度調節機能の付いたステンレス製オートクレーブに、ラウリルアルコール186部(1モル)、過塩素酸マグネシウム0.32部および水酸化マグネシウム0.03部を投入し、混合系内を窒素で置換した後、減圧下(20mmHg)、120℃にて1時間脱水を行った。次いでEO88部(2モル)を150℃にて、ゲージ圧が1〜3kgf/cm2 となるように導入した。EOの付加重合に要した時間は10時間であった。
反応物をGPCの測定条件によって測定した結果、Mw/Mnは1.020[一般式(2)をみたすMw/Mnの上限計算値;1.031]、GCの測定条件によって測定した未反応のラウリルアルコールは全反応物中の3.98質量%(0.0586モル)であった。(定数c=0.558)
ガラス容器に上記反応物165部(0.60モル)と無水マレイン酸62部(0.63モル)を仕込み、65℃で8時間反応させモノエステル化物を得た。その後、亜硫酸ナトリウム84部(0.67モル)を水689部に溶解し、60℃で上記モノエステル化物227部を加え、同温で約5時間反応させスルホン化物を得た。その後、クエン酸でpHを7.0にし、本発明に係わるスルホコハク酸エステル塩の約30%水溶液を得た。
【0043】
製造例3
撹拌および温度調節機能の付いたステンレス製オートクレーブに、ラウリルアルコール186部(1モル)、過塩素酸マグネシウム0.05部を投入し、混合系内を窒素で置換した後、減圧下(約20mmHg)、120℃にて1時間脱水を行った。次いでEO88部(2モル)を150℃にて、ゲージ圧が1〜3kgf/cm2 となるように導入した。(定数c=0.558、未反応ラウリルアルコール3.98質量%)
得られた反応物に水酸化カリウム1.3部を加え、PO58部(1モル)次いでEO44部(1モル)の順に130℃にて反応を行った。反応物をGPCの測定条件によって測定した結果、Mw/Mnは1.036[一般式(2)をみたすMw/Mnの上限計算値;1.052]、GCの測定条件によって測定した未反応のラウリルアルコールは0.21質量%であった。(定数c=0.628)
ガラス容器に上記反応物188部(0.54モル)と無水マレイン酸52部(0.53モル)を仕込み、65℃で8時間反応させモノエステル化物を得た。その後、亜硫酸ナトリウム69部(0.55モル)を水691部に溶解し、60℃で上記モノエステル化物240部を加え、同温で約5時間反応させスルホン化物を得た。その後、クエン酸でpHを7.5にし、本発明に係わるスルホコハク酸エステル塩の約30%水溶液を得た。
【0044】
比較製造例1
撹拌および温度調節機能の付いたステンレス製オートクレーブに、ラウリルアルコール186部(1モル)、水酸化カリウム、0.3部を投入し、混合系内を窒素で置換した後、減圧下(20mmHg)、120℃にて1時間脱水を行った。次いでEO176部(4モル)を150℃にて、ゲージ圧が1〜3kgf/cm2 となるように導入した。
反応物をGPCの測定条件によって測定した結果、Mw/Mnは1.10[一般式(2)をみたすMw/Mnの上限計算値;1.052]、GCの測定条件によって測定した未反応のラウリルアルコールは全反応物中の11.0質量%(0.214モル)であった。(定数c=2.26)
ガラス容器に上記反応物185部(0.51モル)と無水マレイン酸53部(0.54モル)を仕込み、65℃で8時間反応させモノエステル化物を得た。その後、亜硫酸ナトリウム71部(0.56モル)を水691部に溶解し、60℃で上記モノエステル化物238部を加え、同温で約5時間反応させスルホン化物を得た。その後、クエン酸でpHを6.5にし、比較のスルホコハク酸エステル塩の約30%水溶液を得た。
【0045】
比較製造例2
撹拌および温度調節機能の付いたステンレス製オートクレーブに、ラウリルアルコール186部(1モル)、水酸化カリウム0.3部を投入し、混合系内を窒素で置換した後、減圧下(20mmHg)、120℃にて1時間脱水を行った。次いで、EO88部(2モル)を150℃にて、ゲージ圧が1〜3kgf/cm2 となるように導入した。
反応物をGPCの測定条件によって測定した結果、Mw/Mnは1.07[一般式(2)をみたすMw/Mnの上限計算値;1.031]、GCの測定条件によって測定した未反応のラウリルアルコールは全反応物中の38.0質量%(0.560モル)であった。(定数c=11.15)
ガラス容器に上記反応物165部(0.6モル)と無水マレイン酸62部(0.63モル)を仕込み、65℃で8時間反応させモノエステル化物を得た。その後、亜硫酸ナトリウム84部(0.67モル)を水690部に溶解し、60℃で上記モノエステル化物227部を加え、同温で約5時間反応させスルホン化物を得た。その後、クエン酸でpHを6.5にし、比較のスルホコハク酸エステル塩の約30%水溶液を得た。
【0046】
比較製造例3
撹拌および温度調節機能の付いたステンレス製オートクレーブに、ラウリルアルコール186部(1モル)、水酸化カリウム0.6部を投入し、混合系内を窒素で置換した後、減圧下(約20mmHg)、120℃にて1時間脱水を行った。次いでEO88部(2モル)、PO58部(1モル)、EO44部(1モル)を130℃にて、ゲージ圧が1〜3kgf/cm2 となるように導入した。
反応物をGPCの測定条件によって測定した結果、Mw/Mnは1.12[一般式(2)をみたすMw/Mnの上限計算値;1.052]、GCの測定条件によって測定した未反応のラウリルアルコールは全反応物中の7.3質量%であった。(定数c=2.97)
ガラス容器に上記反応物188部(0.50モル)と無水マレイン酸52部(0.53モル)を仕込み、65℃で8時間反応させモノエステル化物を得た。その後、亜硫酸ナトリウム69部(0.55モル)を水691部に溶解し、60℃で上記モノエステル化物240部を加え、同温で約5時間反応させスルホン化物を得た。その後、クエン酸でpHを6.5にし、比較のスルホコハク酸エステル塩の約30%水溶液を得た。
【0047】
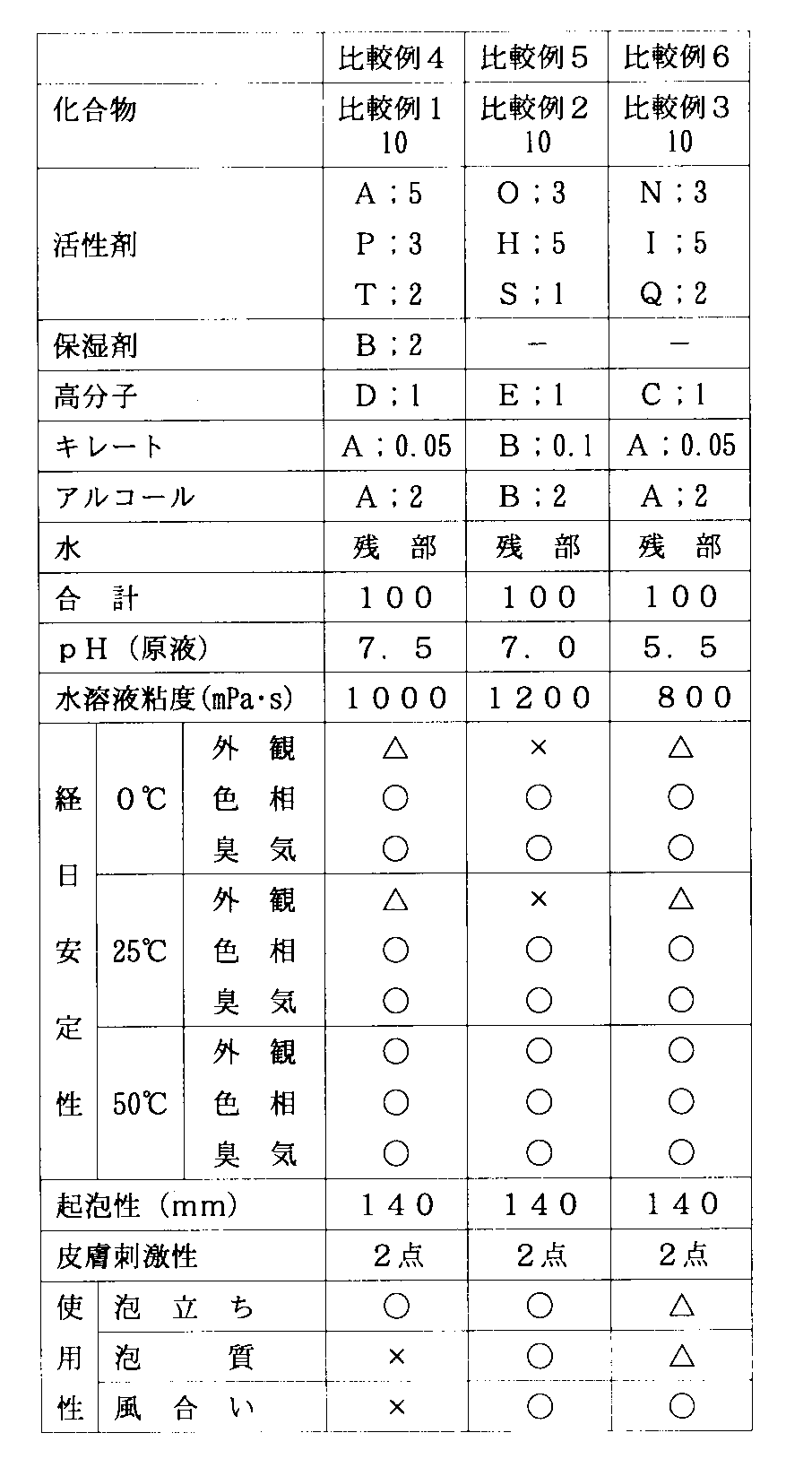
実施例4〜8および比較例4〜6
実施例1〜3の本発明のスルホコハク酸エステル塩からなるアニオン性界面活性剤および比較例1〜3のアニオン性界面活性剤を、表3、4記載の組成(質量部)で配合し、これに水(残部)を加えて全部を100部にすることにより、本発明の洗浄剤組成物および比較の洗浄剤組成物を調整した。
【0048】
実施例1〜8および比較例1〜6のアニオン性界面活性剤または洗浄剤組成物の起泡性、低温および高温での経日安定性、水溶液の粘度、使用性および皮膚刺激性を試験した。その結果を表1〜4に示す。
【0049】
実施例および比較例で用いた界面活性剤、その他の添加剤および試験方法は次の通りである。
活性剤A;ポリオキシエチレン(2モル)ラウリル硫酸ナトリウム
活性剤B;ポリオキシエチレン(4モル)ラウリル硫酸トリエタノールアミン
活性剤C;ポリオキシエチレン(3モル)ラウリルエーテル酢酸ナトリウム
活性剤D;ポリオキシエチレン(3モル)ヤシ油脂肪酸モノエタノールアミド硫酸ナトリウム
活性剤F;スルホコハク酸ポリオキシエチレン(5モル)ラウロイルエタノールアミド2ナトリウム
活性剤G;ヤシ油脂肪酸メチルタウリンナトリウム
活性剤H;ヤシ油脂肪酸サルコシンナトリウム
活性剤I;N−ヤシ油脂肪酸アシル−L−グルタミン酸ナトリウム
活性剤J;ラウロイルメチル−β−アラニンナトリウム
活性剤K;ヤシ油脂肪酸アミドプロピルジメチルアミノ酢酸ベタイン
活性剤L;ラウリルジメチルアミノ酢酸ベタイン
活性剤M;2−アルキル−N−カルボキシメチル−N−ヒドロキシエチルイミダゾリニウムベタイン
活性剤N;ラウリルヒドロキシスルホベタイン
活性剤O;1:1型ヤシ油脂肪酸ジエタノールアミド
活性剤P;ラウリルジメチルアミンオキシド
活性剤Q;モノステアリン酸エチレングリコール
活性剤R;ポリオキシエチレン(120モル)ジオレイン酸メチルグルコシド
活性剤S;塩化ステアリルトリメチルアンモニウム
活性剤T;エチル硫酸ラノリン脂肪酸アミノプロピルエチルジメチルアンモニウム
【0050】
保湿剤A;グリセリン
保湿剤B;ピロリドンカルボン酸ナトリウム
高分子A;カチオン化セルロース
高分子B;カチオン化グアーガム
高分子C;ポリエチレングリコール(分子量1500)
高分子D;ジメチルポリシロキサン
高分子E;加水分解プロテイン
キレートA ;エチレンジアミン四酢酸ナトリウム
キレートB ;1−ヒドロキシエタン−1、1−ジホスホン酸ナトリウム
アルコールA ;プロピレングリコール
アルコールB ;ジプロピレングリコール
【0051】
試験方法
<起泡性>
CaO換算15ppmの硬水を用いて、アニオン性界面活性剤または洗浄剤組成物の0.3%(固形分)水溶液200mlを調整し、ジューサーミキサー(東芝製MX−390GX)で30秒間撹拌し、その時の泡高さ(mm)により起泡性を評価した。
【0052】
<経日安定性;外観>
アニオン性界面活性剤または洗浄剤組成物を100mlのガラス性ボトルに入れ、0℃、25℃および50℃の恒温槽中に30日間放置した後の外観を肉眼で観察し、次の基準で評価した。
評価基準
○;透明液状
△;若干濁りが生じる/若干分離傾向
×;著しく濁りが生じる/分離
<経日安定性;色相>
アニオン性界面活性剤または洗浄剤組成物を100mlのガラス性ボトルに入れ、0℃、25℃および50℃の恒温槽中に30日間放置した後、着色の度合を肉眼で観察し、次の基準で評価した。
評価基準
○;着色無し
△;若干着色
×;著しく着色
<経日安定性;臭気>
アニオン性界面活性剤または洗浄剤組成物を100mlのガラス性ボトルに入れ、0℃、25℃および50℃の恒温槽中に30日間放置した後、着臭の度合を官能検査行い、次の基準で評価した。
評価基準
○;変化なし
△;若干着臭
×;著しく着臭
【0053】
<粘度>
粘度は、ブルックフィールド型粘度計を用い、25℃で測定した。
<使用性>
使用性は、男女各10名のパネルに、表1〜表4に示すアニオン性界面活性剤または洗浄剤組成物を用いシャンプーさせ、使用時の「泡立ち」、「泡質」、「風合い」につき次の基準で評価した。
評価基準
「泡立ち」および「風合い」
○;良好
△;普通
×;劣っている
「泡質」
○;クリーミー
△;普通
×;荒い
【0054】
<皮膚刺激性>
表1〜4に示すアニオン性界面活性剤または洗浄剤組成物の1.0%(固形分)水溶液を調整し、男女各5名による人パッチテスト(クローズド、48時間、上腕内側)を行い、次の基準で評価しその合計点で表した。
評価基準
0;反応(紅斑)無し
1;ごく軽度の紅斑
2;明瞭な紅斑
3;強度の紅斑
【0055】
【表1】
【0056】
【表2】
【0057】
【表3】
【0058】
【表4】
【0059】
この結果から、本発明のアニオン性界面活性剤および洗浄剤組成物は、中性〜弱酸性のpH領域で、起泡性、低温および高温での経日安定性、水溶液へのしやすさ、使用性および人に対する安全性が良好であることがわかる。
【0060】
【発明の効果】
従来から使用されているスルホコハク酸エステル塩は、中性〜弱酸性のpH領域での起泡性、低温および高温での経日安定性、水溶液のハンドリング性に問題があった。本発明のアニオン性界面活性剤は、これらの問題点を解決するばかりでなく、人に対する安全性を満足した洗浄剤組成物を得ることができるものである。
また、スルホコハク酸エステル塩は、従来低温および高温下での長期保存安定性(特に低温でのかすみないし固化)に問題があったが、本発明品は著しく改善されている。さらに、本発明のアニオン性界面活性剤および洗浄剤組成物は、上記洗浄性、起泡性等に加えて、浸透性などの界面活性能にも優れるので、シャンプー、食器用洗剤、金属等の硬質表面の洗浄剤等の洗浄剤として特に有用である。[0001]
BACKGROUND OF THE INVENTION
The present invention relates to an anionic surfactant and a detergent composition containing the anionic surfactant.
[0002]
[Prior art]
Conventionally, it comprises a detergent composition containing a sulfosuccinic acid ester salt type anionic surfactant having a hydrophobic group as an aliphatic alcohol alkylene oxide adduct obtained by addition polymerization of an alkylene oxide to an aliphatic alcohol. Further, hard surface cleaners such as shampoos, tableware cleaners, and metals are known (for example, JP-A-1-259098).
[0003]
[Problems to be solved by the invention]
However, the sulfosuccinic acid ester salt type anionic surfactant blended in these detergent compositions is stable over time, such as a product with a faint appearance when stored at low temperatures for a long period of time, foaming properties, and washing. There was a problem about sex and skin irritation of the human body.
[0004]
[Means for Solving the Problems]
As a result of intensive studies to solve the above-mentioned problems, the present inventors have found that a sulfosuccinic acid ester salt type anionic surfactant having a specific aliphatic alcohol alkylene oxide adduct as a hydrophobic group is stable over time. Thus, the present inventors have found that the present invention can solve the problems of the nature and other problems.
[0005]
That is, the present invention comprises a sulfosuccinic acid ester salt (B) of an aliphatic alcohol alkylene oxide adduct (A); (A) contains an aliphatic alcohol (a1) having a carbon number in the presence of a catalyst (d). An aliphatic alcohol alkylene oxide adduct (A1) or (A1) directly produced by adding 1 to 2.5 moles of an average of 2 or more alkylene oxides (b2) and satisfying the following (i) to (iii): It is an aliphatic alcohol alkylene oxide adduct (A2) obtained by subjecting an alkylene oxide (b3) having 2 or more carbon atoms to an addition reaction in the presence of an alkali catalyst (e) and satisfying the following (i) to (iii): Anionic surfactant characterized by having 8 to 24 carbon atoms in the presence of catalyst (d) which is one or more selected from the group consisting of perhalogenates (salts), sulfates, phosphates and nitrates Directly produced by adding 1 to 2.5 moles of alkylene oxide (b2) having 2 or more carbon atoms to aliphatic alcohol (a1), and the distribution constant c obtained from the following formula (4) is 1.0 or less. A certain aliphatic alcohol alkylene oxide adduct (A1) or (A1) in the presence of an alkali catalyst (e), an alkylene oxide having 2 or more carbon atoms (b3) [(b3) is the same as (b2) An aliphatic alcohol alkylene oxide adduct (A2) obtained by addition reaction of (which may be different)], and an aliphatic alcohol alkylene oxide adduct (A) satisfying the following (i) to (iii): Without fractionation of unreacted alcohol and those with different added mole numbers, sulfosuccinic acid ester chloride is obtained, and the sulfosuccinic acid ester salt (B1) (B1) is obtained. A method for producing an anionic surfactant, comprising: a detergent composition comprising the anionic surfactant; and a skin cleanser, a hair cleanser or a household detergent comprising the detergent composition. is there.
[0006]
(1) Alkylene oxide (b1) adduct of aliphatic alcohol (a1), which is composed of one or a mixture of two or more compounds represented by the following general formula (1).
R 1 -(OA) k-OH (1)
[Wherein R 1 Is an aliphatic hydrocarbon group having 8 to 24 carbon atoms or an alicyclic hydrocarbon group having 8 to 24 carbon atoms; A is one or more alkylene groups having 2 or more carbon atoms; k is 0 having an average of 1 to 20 Or an integer greater than or equal to 1]
(2) Ratio of weight average molecular weight (Mw) to number average molecular weight (Mn): Mw / Mn satisfies the following relational expression (2) or (3).
Mw / Mn ≦ 0.030 × Ln (v) +1.010 (where v <10) (2)
Mw / Mn ≦ −0.026 × Ln (v) +1.139 (provided that v ≧ 10) (3)
[Wherein v represents the average number of moles of alkylene oxide added per mole of the aliphatic alcohol (a1), and corresponds to the average of k in the above general formula (1). ]
(3) The distribution constant c obtained from the following formula (4) is 2.0 or less. (This item applies only when v is 12 or less.)
c = (v + n 0 / N 00 -1) / [Ln (n 00 / N 0 ) + N 0 / N 00 -1] (4)
[Where v is the same as above, n 00 Is the number of moles of (a1) used in the reaction, n 0 Represents the number of moles of unreacted (a1). ]
[0007]
DETAILED DESCRIPTION OF THE INVENTION
In the anionic surfactant of the present invention, the sulfosuccinic acid ester salt (B) is not particularly limited as long as it is a sulfosuccinic acid ester salt of (A) satisfying the above (1) to (3), but the following general formula ( It is preferable from the point of surface active ability that it is 1 or more types of the salt represented by 5).
[Wherein R 1 Is an aliphatic hydrocarbon group having 8 to 24 carbon atoms or an alicyclic hydrocarbon group having 8 to 24 carbon atoms; A is one or more alkylene groups having 2 or more carbon atoms; k is 0 having an average of 1 to 20 Or an integer greater than or equal to 1; Z 1 And Z 2 Either one is H and the other is SO 3 Q 1 Represents a group, X is Q 2 Or R 1 -Represents an (OA) k- group. Q 1 And Q 2 Represents H or a cationic counter ion. ]
[0008]
(B) further contains 80 to 100% by mass, particularly 90 to 100% by mass of a monoester product represented by the following general formula (5a) and / or (5b), and is represented by the general formula (5c). What contains 0-20 mass%, especially 0-10 mass% of the diesterified product made is preferable. Further, the mass% ratio of (5a) / (5b) is preferably 10 to 90/90 to 10, more preferably 30 to 70/70 to 30.
[Wherein R 1 , A, k, Q 1 And Q 2 Is the same as in general formula (5). ]
[0009]
In the general formula (5), R 1 The — (OA) k—O— group is a residue of the aliphatic alcohol alkylene oxide adduct (A). Z 1 Or Z 2 SO forming group 3 Q 1 The group is, for example, a sulfonate group introduced by sulfonating an esterified product obtained by reacting (A) with maleic anhydride. Q 1 And Q 2 The group includes hydrogen and a cationic counter ion depending on the type of sulfonating agent used. Examples of the cationic counter ion include metal ions such as alkali metal (sodium, potassium, lithium, etc.) ions, alkaline earth metal (magnesium, calcium, barium, etc.) ions; ammonium ions, and the like.
[0010]
In the present invention, (A) is an aliphatic alcohol (a1) (in the present specification, an aliphatic alcohol includes both an aliphatic alcohol and an alicyclic alcohol) and an alkylene oxide (b1). Is one or a mixture of two or more aliphatic alcohol alkylene oxide adducts obtained by the addition of and is represented by the following general formula (1).
R 1 -(OA) k-OH (1)
[Wherein R 1 Is an aliphatic hydrocarbon group having 8 to 24 carbon atoms or an alicyclic hydrocarbon group having 8 to 24 carbon atoms; A is one or more alkylene groups having 2 or more carbon atoms; k is 0 having an average of 1 to 20 Or an integer greater than 1]
[0011]
In the general formula (1), R 1 Is a residue of an aliphatic alcohol (a1), and usually an aliphatic hydrocarbon group (alkyl group, alkenyl group, alkadienyl group, etc.) having 8 to 24 carbon atoms or an alicyclic hydrocarbon group (cycloalkyl group). Represents a polycyclic hydrocarbon group. R 1 May be a mixture of two or more groups such as linear and branched. When the number of carbon atoms is less than 8, sufficient foaming power and detergency cannot be obtained, and when the number of carbon atoms exceeds 24, foaming power and detergency are inferior.
R 1 Specific examples of the alkyl group include octyl, nonyl, decyl, lauryl, tridecyl, myristyl, cetyl, stearyl, nonadecyl, 2-ethylhexyl, 2-ethyloctyl group and the like. Examples of the alkenyl group include octenyl, decenyl, dodecenyl, tridecenyl, pentadecenyl, oleyl, gadryl group and the like. Examples of the alkadienyl group include a linoleyl group. Examples of the cycloalkyl group include ethylcyclohexyl, propylcyclohexyl, octylcyclohexyl, nonylcyclohexyl group and the like. Examples of the polycyclic hydrocarbon group include an adamantyl group.
[0012]
In the general formula (1), A represents an alkylene group having 2 or more carbon atoms, preferably 2 to 8, more preferably 2 to 4 and particularly preferably 2 or 3 carbon atoms. It is formed by the addition of two or more alkylene oxides (b1). Examples of the alkylene oxide (b1) include ethylene oxide (hereinafter abbreviated as EO), propylene oxide (hereinafter abbreviated as PO), 1,2- or 2,3-butylene oxide, tetrahydrofuran, styrene oxide, and the like. It is done. Preferred is EO and / or PO.
In the general formula (1), k corresponds to the number of added moles of (b1) and is an integer having an average of 1 to 20, preferably 1 to 12. When k exceeds 20, it is not preferable because sufficient foaming power and cleaning power cannot be obtained.
[0013]
The aliphatic alcohol (a1) is R 1 It gives a residue and is usually an alcohol having 8 to 24 (preferably 12 to 18) carbon atoms, and may be a natural alcohol or a synthetic alcohol (Ziegler alcohol, oxo alcohol, etc.). Specific examples include saturated aliphatic alcohols such as octyl alcohol, nonyl alcohol, decyl alcohol, undecyl alcohol, dodecyl alcohol, tridecyl alcohol, tetradecyl alcohol, hexadecyl alcohol, octadecyl alcohol, nonadecyl alcohol; octenyl alcohol, Unsaturated aliphatic alcohols such as decenyl alcohol, dodecenyl alcohol, tridecenyl alcohol, pentadecenyl alcohol, oleyl alcohol, gadryl alcohol, linoleyl alcohol; ethyl cyclohexyl alcohol, propyl cyclohexyl alcohol, octyl cyclohexyl And cyclic aliphatic alcohols such as alcohol, nonylcyclohexyl alcohol, adamantyl alcohol, etc. The other is 2 or more can be used. These aliphatic alcohols are preferably primary or secondary, and more preferably primary. Further, the alkyl group portion may be linear or branched. Particularly preferred are dodecyl alcohol, tridecyl alcohol, tetradecyl alcohol, hexadecyl alcohol and octadecyl alcohol.
[0014]
(A) is preferably produced directly from (a1) and (b1) because the process is not complicated. Here, “directly produced” means that the adduct is obtained directly without fractionation of unreacted alcohol or different added moles by rectification or the like. However, the fractionation operation does not include those obtained by stripping unreacted alkylene oxide or low-boiling substances by a simple operation without the purpose of fractionation.
[0015]
In the present invention, (A) is a ratio of the weight average molecular weight (Mw) to the number average molecular weight (Mn): Mw / Mn needs to satisfy the following relational expression (2) or (3).
Mw / Mn ≦ 0.030 × Ln (v) +1.010 (where v <10) (2)
Mw / Mn ≦ −0.026 × Ln (v) +1.139 (provided that v ≧ 10) (3)
In these formulas, Ln (v) means the natural logarithm of v, v represents the average number of moles of alkylene oxide added per mole of aliphatic alcohol (a1), and in the general formula (1), It corresponds to the average of k, which is the number of moles of alkylene oxide added. (same as below)
If the relational expression (2) or (3) is not satisfied, that is, the molecular weight distribution becomes wide, sufficient surface activity cannot be obtained when an anionic surfactant is used.
Mw / Mn preferably satisfies the following relational expression (2 ′) or (3 ′).
Mw / Mn ≦ 0.031 × Ln (v) +1.000 (provided that v <10) (2 ′)
Mw / Mn ≦ −0.026 × Ln (v) +1.129 (provided that v ≧ 10) (3 ′)
[0016]
In the general formula (1), when A is an ethylene group only, that is, when only ethylene oxide is added to the aliphatic alcohol (a1), the weight average molecular weight (Mw) and the number average molecular weight (Mn) Ratio: Mw / Mn preferably satisfies the following (6) or (7) instead of the relational expression (2) or (3).
Mw / Mn ≦ 0.020 × Ln (v) +1.010 (provided that v <10) (6)
Mw / Mn ≦ −0.026 × Ln (v) +1.116 (provided that v ≧ 10) (7)
When the relational expression (6) or (7) is satisfied, that is, when the molecular weight distribution is narrow, good surface activity can be obtained when an anionic surfactant is used.
In this case, it is more preferable that Mw / Mn satisfies the following relational expression (6 ′) or (7 ′).
Mw / Mn ≦ 0.018 × Ln (v) +1.015 (provided that v <10) (6 ′)
Mw / Mn ≦ −0.023 × Ln (v) +1.113 (provided that v ≧ 10) (7 ′)
[0017]
Furthermore, in (A), when the distribution constant c can be obtained from the relational expression (4) derived from the following Weibull distribution law expression (8), c needs to be 2.0 or less. c is preferably 1.0 or less, more preferably 0.7 or less. In relational expression (4), the smaller the value of distribution constant c, that is, the smaller the content of unreacted aliphatic alcohol, the narrower the molecular weight distribution.
This formula is applied when the amount of the unreacted aliphatic alcohol (a1) is not less than the detection limit (0.001% by mass). In the case of (A), the alkylene oxide (b1) Is applicable up to about 12 moles.
When c exceeds 2, sufficient surface activity cannot be obtained when an anionic surfactant is used.
v = c × Ln (n 00 / N 0 )-(C-1) × (1-n 0 / N 00 (8)
c = (v + n 0 / N 00 -1) / [Ln (n 00 / N 0 ) + N 0 / N 00 -1] (4)
In these equations, Ln (n 00 / N 0 ) Is (n 00 / N 0 ) Is the natural logarithm of v), v is the same as above, n 00 Is the number of moles of the aliphatic alcohol (a1) used in the reaction, n 0 Represents the number of moles of the unreacted aliphatic alcohol (a1).
[0018]
In the present invention, the above (A) is preferably a compound represented by the following general formula (1 ′) in terms of foaming power.
R 1 -[(OC 2 H 4 ) M / (OD) n]-(OC 2 H 4 P-OH (1 ')
[Wherein R 1 Is an aliphatic hydrocarbon group having 8 to 24 carbon atoms or an alicyclic hydrocarbon group having 8 to 24 carbon atoms; D is an alkylene group having 3 or 4 carbon atoms; m is 0 or 1 or more with an average of 0 to 5 , N is an integer of 0 or 1 or more with an average of 0 to 5, p is an integer of 0 or 1 or more with an average of 1 to 10, and (m + n + p) is an integer with an average of 1 to 20 Yes, (m + p) / (m + n + p) is 0.5 or more on average. {(OC 2 H 4 ) M / (0D) n} represents block addition or random addition when m ≠ 0 and n ≠ 0. ]
[0019]
In the general formula (1 ′), (OC 2 H 4 ) Is formed by the addition of EO. D represents an alkylene group having 3 or 4 carbon atoms, and the (OD) moiety is formed by addition of an alkylene oxide having 3 or 4 carbon atoms. Examples of such alkylene oxide include PO, 1,2- or 2,3-butylene oxide.
[0020]
In the general formula (1 ′), m is 0 or an integer of 1 or more with an average of 0 to 5, more preferably an integer of 0 to 3. n is 0 or an integer greater than or equal to 1 with an average of 0 to 5, more preferably 0 or 1. p is 0 or an integer of 1 or more with an average of 1 to 10, more preferably 1 to 8. (M + n + p) is an integer having an average of 1 to 20, more preferably 1 to 12. (M + p) / (m + n + p) is an average of 0.5 or more, more preferably 0.7 or more. When it is 0.5 or more, the detergency when an anionic active agent is used is good. {(OC 2 H 4 ) M / (OD) n} is a block addition [(OC 2 H 4 ) M, (OD) n]] or random addition, but more preferably block addition.
[0021]
In the present invention, (A) is preferably (A1) or (A2) produced by the method described below.
That is, the aliphatic alcohol (a1) is added to the aliphatic alcohol (a1) in the presence of the catalyst (d) that gives an adduct having a distribution constant c determined by the formula (4) derived from the Weibull's distribution rule of 2.0 or less. It is an aliphatic alcohol alkylene oxide adduct (A1) obtained by adding an average of 1 to 2.5 moles of alkylene oxide (b2), or an alkylene oxide (A1) in the presence of an alkali catalyst (f). It is preferable that it is an aliphatic alcohol alkylene oxide adduct (A2) formed by addition reaction of b3). More preferably (A2).
[0022]
Examples of the alkylene oxides (b2) and (b3) include alkylene oxides having 2 or more carbon atoms. Specific examples include the alkylene oxides described above as examples of the alkylene oxide (b1) that forms OA in the general formula (1), and two or more of them may be used in combination. When two or more types are used, block addition or random addition may be used, but block addition is preferred. Of these, EO and / or PO are preferred.
[0023]
As the catalyst (d), a catalyst in which the resulting alkylene oxide adduct has a distribution constant c of 2.0 or less is used. Preferably, c is 1.0 or less, more preferably 0.7 or less, and particularly preferably 0.45 or less. When the distribution constant c is more than 2.0, when an anionic surfactant is used, it causes fogging at a low temperature, and foaming properties and detergency deteriorate. Moreover, the irritation to the skin is not lowered, which may cause a problem.
[0024]
Specific examples of the catalyst (d) include perhalogen acids or salts thereof, sulfates, phosphates and nitrates. The metal for forming the salt is not particularly limited, but is preferably other than an alkali metal, and is preferably a divalent or trivalent metal.
These metals are preferably Mg, Ca, Sr, Ba, Zn, Co, Ni, Cu, Al, more preferably Mg, Zn, Ca, Sr, Ba, Al, and particularly preferably Mg, Zn and Al. Examples of the halogen of the perhalogen acid (salt) include chlorine, bromine and iodine, with chlorine being preferred.
Accordingly, (d) is preferably a divalent or trivalent metal perchlorate, and more preferably a metal perchlorate selected from Mg, Zn and Al. Further, (d) may be used in combination with a divalent or trivalent metal alcoholate. Examples of the alkyl group of the metal alcoholate include a lower (1 to 4 carbon atoms) alkyl group that can be easily distilled off as an alcohol, or an alkyl group having the same composition as that of the starting aliphatic alcohol. These catalysts may be one kind, but two or more kinds of catalysts [for example, magnesium perchlorate / magnesium sulfate heptahydrate = 95 / 5-50 / 50, magnesium perchlorate / aluminum perchlorate = 99/1. ~ 30/70 (both mass ratios)] are preferably used in combination.
[0025]
The amount of the catalyst (d) used is preferably 0.001 to 1 part by mass per 100 parts by mass in total of (a1) and (b2) from the viewpoint of reaction rate and economy. More preferably, it is 0.003-0.8 mass part, Most preferably, it is 0.005-0.5 mass part.
[0026]
The catalyst used when the alkylene oxide (b3) is added to the alkylene oxide adduct (A1) obtained by adding (b2) to (a1) is the alkali catalyst (e).
Examples of the alkali catalyst (e) include alkali metal and alkaline earth metal hydroxides such as lithium hydroxide, sodium hydroxide, potassium hydroxide, cesium hydroxide, magnesium hydroxide, calcium hydroxide, barium hydroxide and the like. More preferred are potassium hydroxide and cesium hydroxide.
The amount of the catalyst (e) used is preferably 0.0001 to 1 part by mass per 100 parts by mass in total of (A1) and (b3) from the viewpoint of reaction rate and economy. More preferably, it is 0.001-0.8 mass part.
[0027]
As reaction conditions in the case of reacting (a1) and (b2), (a1) and (d) are mixed, and after nitrogen substitution, -0.8 to 5 kgf / cm 2 In G, (b2) is introduced at 80 to 200 ° C., a predetermined amount of (b2) is introduced, and then aging is performed at 80 to 200 ° C. until the pressure in the reaction system becomes equilibrium.
By adding an alkali catalyst (e) to the alkylene oxide adduct (A1) thus obtained and reacting the alkylene oxide (b3) in the same manner as described above, an aliphatic alcohol alkylene oxide addition is performed. A product (A2) is obtained.
[0028]
The method for obtaining the sulfosuccinic acid ester salt (B) from (A) is not particularly limited. As an example, an esterified product is obtained by reacting (A) with maleic anhydride (hereinafter sometimes abbreviated as MA). There is a method of reacting in a two-stage process including a process and a sulfonation process using sulfite or acidic sulfite in the presence of water.
In the reaction of (A) with MA, the reaction temperature is usually 50 to 100 ° C., preferably 60 to 90 ° C., and the reaction time varies depending on the reaction temperature, but usually 1 to 15 hours, preferably 3 to 7 hours. The reaction is carried out in a nitrogen atmosphere to obtain the maleic ester (A). When MA is excessively added, de-MA is performed under reduced pressure after the esterification reaction is completed. As described above, the molar ratio of (A) to MA is preferably 1.0: 0.9 to 1.0: 1.1, and more preferably, since a high monoester content is preferable. 1.0: 0.95-1.0: 1.05.
The end point of the esterification reaction is that the acid value (AV) represented by [56100 / (molecular weight of esterified product)] is 90 to 110% of the theoretical value, preferably
It is a point which becomes 95 to 105%.
[0029]
Subsequently, sulfonation of the esterified product produced using sulfite or acidic sulfite is carried out. Examples of sulfites include alkali metal salts such as sodium sulfite and potassium sulfite, alkaline earth metal salts such as magnesium sulfite, and ammonia salts such as ammonium sulfite. Acid sulfites include sodium bisulfite and hydrogen sulfite. Examples thereof include bisulfites (alkali metal salts, alkaline earth metal salts, ammonium salts) such as potassium, ammonium hydrogen sulfite, and magnesium hydrogen sulfite. Sulfonation can be performed by reacting an esterified product with sulfite or bisulfite in the presence of water. The molar ratio of the esterified product to sulfite or bisulfite is usually 1.0: 0.9 to 1.0: 1.1, preferably 1.0: 0.95 to 1.0: 1.05. Yes, the reaction temperature is usually 30 to 90 ° C., preferably 40 to 80 ° C., and the reaction is usually performed in a nitrogen atmosphere. While the reaction time varies depending on the reaction temperature, it is generally 1 to 10 hours, preferably 2 to 5 hours.
The end point of sulfonation is that the amount of bound sulfuric acid represented by [(80 × 100) / sulfonated molecular weight] is 90 to 110% of the theoretical value, preferably 95 to 105%. . After sulfonation is complete, the pH is neutralized with an organic acid such as citric acid or an alkanolamine such as triethanolamine.
[0030]
The anionic surfactant comprising the sulfosuccinic acid ester salt (B) of the present invention is excellent in foamability and detergency. In addition, since the aliphatic alcohol alkylene oxide adduct (A) satisfying (1) to (3) contains little aliphatic alcohol as a raw material, it is also less in the sulfosuccinic acid ester salt and irritate the human skin. It will be less prone. Odor is also improved.
[0031]
The anionic surfactant comprising (B) of the present invention may be used alone as a cleaning agent, but if necessary, an anionic surfactant other than the present invention in an amount that does not interfere with the effects of the present invention, nonionic properties One or more surfactants selected from surfactants, cationic surfactants and amphoteric surfactants can be blended to form a detergent composition. The pH of the cleaning composition is not particularly limited, but is preferably neutral to weakly acidic (for example, pH 4 to 9) from the viewpoint of safety to the human body.
The anionic surfactant of the present invention is preferably 3 to 60%, and more preferably 5 to 50, as the blending ratio (mass%) of the surfactant active ingredient (solid content conversion) in the cleaning composition. %. When an anionic surfactant other than the present invention is used, it is preferably 60% or less, more preferably 50% or less, based on the total amount with the anionic surfactant of the present invention, and an anionic surfactant. It is preferable that the total blending ratio is within the above range. Further, the nonionic surfactant is preferably 60% or less, and more preferably 1 to 30%. The cationic surfactant is preferably 5% or less, more preferably 0.05 to 3%. The amphoteric surfactant is preferably 50% or less, more preferably 1 to 30%.
[0032]
Specifically, other anionic surfactants other than the present invention include hydrocarbon ether carboxylic acids having 8 to 24 carbon atoms or salts thereof, [(poly) oxyethylene (degree of polymerization = 1 to 100) lauryl. Sodium ether acetate, (poly) oxyethylene (degree of polymerization = 1-100) disodium lauryl sulfosuccinate, etc., C8-24 hydrocarbon sulfate salt [sodium lauryl sulfate, (poly) oxyethylene (degree of polymerization) = 1-100) Sodium lauryl sulfate, (Poly) oxyethylene (degree of polymerization = 1-100) Triethanolamine lauryl sulfate, (Poly) oxyethylene (degree of polymerization = 1-100) Palm oil fatty acid monoethanolamide sodium sulfate, ], C8-24 hydrocarbon sulfonate [sodium dodecylbenzenesulfonate ] And C8-24 hydrocarbon phosphate ester salt [sodium lauryl phosphate, (poly) oxyethylene (degree of polymerization = 1-100) sodium lauryl ether phosphate, etc.], fatty acid salt [sodium laurate, laurin] Acid triethanolamine, etc.], acylated amino acid salts [coconut oil fatty acid methyl taurine sodium, coconut oil fatty acid sarcosine sodium, coconut oil fatty acid sarcosine triethanolamine, N-coconut oil fatty acid acyl-L-glutamic acid triethanolamine, N-coconut Oil fatty acid acyl-sodium L-glutamate, sodium lauroylmethyl-β-alanine, etc.] and others [sulfosuccinic acid (poly) oxyethylene (degree of polymerization = 1-100) disodium lauroylethanolamide, etc.] and the like.
[0033]
Nonionic surfactants include aliphatic alcohols (8 to 24 carbon atoms) alkylene oxide (2 to 8 carbon atoms) adducts (degree of polymerization = 1 to 100), (poly) oxyalkylenes (2 to 8 carbon atoms). , Degree of polymerization = 1 to 100) higher fatty acid (8 to 24 carbon atoms) ester [polyethylene glycol monostearate (polymerization degree = 20), polyethylene glycol distearate (degree of polymerization = 30), etc.], polyvalent (bivalent to 10-valent or higher alcohol alcohol (8 to 24 carbon atoms) ester [glyceryl monostearate, ethylene glycol monostearate, sorbitan monolaurate, etc.], (poly) oxyalkylene (2 to 8 carbon atoms, degree of polymerization = 1) To 100) polyvalent (divalent to 10-valent or higher) alcohol higher fatty acid (8 to 24 carbon atoms) ester [monolau Polyoxyethylene acid (degree of polymerization = 10) sorbitan, polyoxyethylene (degree of polymerization = 50) dioleic acid methyl glucoside, etc.], fatty acid alkanolamide [1: 1 type coconut oil fatty acid diethanolamide, 1: 1 type lauric acid diethanolamide Etc.], (poly) oxyalkylene (carbon number 2-8, polymerization degree = 1-100) alkyl (carbon number 1-22) phenyl ether, (poly) oxyalkylene (carbon number 2-8, polymerization degree = 1) -100) alkyl (carbon number 8-24) amino ether and alkyl (carbon number 8-24) dialkyl (carbon number 1-6) amine oxide [lauryl dimethylamine oxide etc.] etc. are mentioned.
[0034]
Cationic surfactants include quaternary ammonium salt type [stearyl trimethyl ammonium chloride, behenyl trimethyl ammonium chloride, distearyl dimethyl ammonium chloride, ethyl lanolin sulfate fatty acid aminopropylethyl dimethyl ammonium, etc.], amine salt type [diethylamino stearate] Ethylamide lactate, dilaurylamine hydrochloride, oleylamine lactate, etc.]. Amphoteric surfactants include betaine-type amphoteric surfactants [coconut oil fatty acid amidopropyldimethylaminoacetic acid betaine, lauryldimethylaminoacetic acid betaine, 2-alkyl-N-carboxymethyl-N-hydroxyethylimidazolinium betaine, laurylhydroxy Sulfobetaine, lauroylamidoethylhydroxyethylcarboxymethylbetaine hydroxypropyl sodium phosphate and the like] and amino acid type amphoteric surfactants [sodium β-laurylaminopropionate and the like]. One or more of these can be used.
[0035]
The cleaning composition of the present invention can be used by blending one or more conventionally known additives. Examples of such a humectant include glycerin and sodium pyrrolidonecarboxylate as a moisturizing agent; as a polymer compound used as a conditioning agent, cationized cellulose having a weight average molecular weight in the range of 5 to 5 million, cationized guar gum, polyethylene glycol, poly Sodium acrylate, hydroxyethyl cellulose, protein derivatives, etc .; Silicon, dimethylpolysiloxane, modified silicones with various organic groups introduced into part of the methyl group of dimethylpolysiloxane, cyclic dimethylsiloxane, etc .; Chelating agent, ethylenediaminetetraacetic acid Sodium, 1-hydroxyethane-1, sodium 1-diphosphonate, etc .; lower alcohols such as ethanol, propylene glycol, dipropylene glycol, etc. 2-6 alcohol; perfumes, colorants, preservatives, ultraviolet absorbers, water and the like.
[0036]
The form of the cleaning composition of the present invention is not particularly limited to liquids, pastes, solids, powders and the like, but liquids and pastes are preferable because they are easy to use.
For example, in the case of a liquid and paste-like shampoo composition, the anionic surfactant of the present invention is preferably 5 to 30% by mass, and the total amount of the surfactant containing the surfactant of the present invention is preferably 5 to 5%. 50% by mass, more preferably 10-30% by mass; polymer compound, silicon, fragrance, colorant, preservative and ultraviolet absorber are each preferably 0-5% by mass, humectant, chelating agent and lower alcohols Are preferably 0 to 10% by mass, and the total of additives other than these surfactants is preferably 0 to 30% by mass, more preferably 1 to 25% by mass; water is preferably 35 to 95% by mass. .
[0037]
Although the use of the cleaning composition of the present invention is not particularly limited, for example, it is used as a skin cleaning agent such as body shampoo and facial cleanser; a hair cleaning agent such as shampoo; .
[0038]
【Example】
EXAMPLES Hereinafter, although an Example demonstrates this invention further in detail, this invention is not limited to this. “Part” indicates part by mass.
Measurement of the molecular weight by gel permeation chromatography (GPC) and measurement of the content of unreacted aliphatic alcohol by gas chromatography (GC) are as follows.
[0039]
[0040]
<< GC measurement conditions >>
Column Silicon GE-SE30
Detector: FID
Injection: 280 ° C
Temperature increase rate: 100-250 ° C / 10 ° C per minute
Internal standard: Octanol
[0041]
Production Example 1
186 parts (1 mole) of lauryl alcohol, 0.32 part of magnesium perchlorate and 0.03 part of magnesium hydroxide were charged into a stainless steel autoclave having a stirring and temperature control function, and the inside of the mixed system was replaced with nitrogen. Thereafter, dehydration was performed at 120 ° C. for 1 hour under reduced pressure (20 mmHg). Next, 88 parts (2 mol) of EO at 150 ° C. and the gauge pressure is 1 to 3 kgf / cm. 2 Introduced to become. The time required for the addition polymerization of EO was 10 hours.
To the obtained reaction product, 1.3 parts of potassium hydroxide was added, and 44 parts (1 mol) of EO was reacted at 130 ° C. As a result of measuring the reaction product under the GPC measurement conditions, Mw / Mn was 1.037 [upper calculated value of Mw / Mn satisfying general formula (2) 1.043], unreacted measured under the GC measurement conditions. Of lauryl alcohol was 2.35% by mass (0.040 mol) in the total reaction product. (Constant c = 0.910) 176 parts (0.55 mol) of the reaction product and 57 parts (0.58 mol) of maleic anhydride were charged in a glass container and reacted at 65 ° C. for 8 hours to obtain a monoester product. . Thereafter, 77 parts (0.61 mol) of sodium sulfite was dissolved in 690 parts of water, 233 parts of the monoester product was added at 60 ° C., and reacted at the same temperature for about 5 hours to obtain a sulfonated product. Thereafter, the pH was adjusted to 6.5 with citric acid to obtain an approximately 30% aqueous solution of the sulfosuccinic acid ester salt according to the present invention.
[0042]
Production Example 2
186 parts (1 mole) of lauryl alcohol, 0.32 part of magnesium perchlorate and 0.03 part of magnesium hydroxide were charged into a stainless steel autoclave having a stirring and temperature control function, and the inside of the mixed system was replaced with nitrogen. Thereafter, dehydration was performed at 120 ° C. for 1 hour under reduced pressure (20 mmHg). Next, 88 parts (2 mol) of EO at 150 ° C. and the gauge pressure is 1 to 3 kgf / cm. 2 Introduced to become. The time required for the addition polymerization of EO was 10 hours.
As a result of measuring the reaction product under GPC measurement conditions, Mw / Mn was 1.020 [upper calculated value of Mw / Mn satisfying general formula (2); 1.031], unreacted measured by GC measurement conditions. Lauryl alcohol was 3.98% by mass (0.0586 mol) in the total reaction product. (Constant c = 0.558)
A glass container was charged with 165 parts (0.60 mol) of the reaction product and 62 parts (0.63 mol) of maleic anhydride and reacted at 65 ° C. for 8 hours to obtain a monoester product. Thereafter, 84 parts (0.67 mol) of sodium sulfite was dissolved in 689 parts of water, 227 parts of the monoester product was added at 60 ° C., and reacted at the same temperature for about 5 hours to obtain a sulfonated product. Thereafter, the pH was adjusted to 7.0 with citric acid to obtain an approximately 30% aqueous solution of the sulfosuccinic acid ester salt according to the present invention.
[0043]
Production Example 3
186 parts (1 mole) of lauryl alcohol and 0.05 part of magnesium perchlorate were charged into a stainless steel autoclave equipped with stirring and temperature control functions, and the inside of the mixed system was replaced with nitrogen, and then under reduced pressure (about 20 mmHg) And dehydration at 120 ° C. for 1 hour. Next, 88 parts (2 mol) of EO at 150 ° C. and the gauge pressure is 1 to 3 kgf / cm. 2 Introduced to become. (Constant c = 0.558, unreacted lauryl alcohol 3.98% by mass)
To the obtained reaction product, 1.3 parts of potassium hydroxide was added, and the reaction was carried out at 130 ° C. in the order of 58 parts (1 mole) of PO and then 44 parts (1 mole) of EO. As a result of measuring the reaction product under GPC measurement conditions, Mw / Mn was 1.036 [upper calculation value of Mw / Mn satisfying general formula (2); 1.052], unreacted measured by GC measurement conditions. Lauryl alcohol was 0.21% by mass. (Constant c = 0.628)
A glass container was charged with 188 parts (0.54 mol) of the reaction product and 52 parts (0.53 mol) of maleic anhydride and reacted at 65 ° C. for 8 hours to obtain a monoester product. Thereafter, 69 parts (0.55 mol) of sodium sulfite was dissolved in 691 parts of water, 240 parts of the monoester product was added at 60 ° C., and reacted at the same temperature for about 5 hours to obtain a sulfonated product. Thereafter, the pH was adjusted to 7.5 with citric acid to obtain an about 30% aqueous solution of the sulfosuccinic acid ester salt according to the present invention.
[0044]
Comparative production example 1
186 parts (1 mol) of lauryl alcohol and 0.3 part of potassium hydroxide were charged into a stainless steel autoclave having a stirring and temperature control function, and the mixed system was replaced with nitrogen, and then under reduced pressure (20 mmHg). Dehydration was performed at 120 ° C. for 1 hour. Next, 176 parts (4 moles) of EO at 150 ° C. and a gauge pressure of 1 to 3 kgf / cm 2 Introduced to become.
As a result of measuring the reaction product under GPC measurement conditions, Mw / Mn was 1.10 [the upper limit calculation value of Mw / Mn satisfying general formula (2); 1.052], unreacted measured by GC measurement conditions. Lauryl alcohol was 11.0% by mass (0.214 mol) in the total reaction product. (Constant c = 2.26)
A glass container was charged with 185 parts (0.51 mol) of the reaction product and 53 parts (0.54 mol) of maleic anhydride and reacted at 65 ° C. for 8 hours to obtain a monoester product. Thereafter, 71 parts (0.56 mol) of sodium sulfite was dissolved in 691 parts of water, 238 parts of the monoester product was added at 60 ° C., and reacted at the same temperature for about 5 hours to obtain a sulfonated product. Thereafter, the pH was adjusted to 6.5 with citric acid to obtain an approximately 30% aqueous solution of a comparative sulfosuccinic acid ester salt.
[0045]
Comparative production example 2
186 parts (1 mol) of lauryl alcohol and 0.3 part of potassium hydroxide were put into a stainless steel autoclave having a stirring and temperature control function, and the inside of the mixed system was replaced with nitrogen. Then, under reduced pressure (20 mmHg), 120 Dehydration was performed at 0 ° C. for 1 hour. Next, 88 parts (2 moles) of EO at 150 ° C. and a gauge pressure of 1 to 3 kgf / cm 2 Introduced to become.
As a result of measuring the reaction product under GPC measurement conditions, Mw / Mn was 1.07 [upper calculation value of Mw / Mn satisfying general formula (2); 1.031], unreacted measured by GC measurement conditions. Lauryl alcohol was 38.0 mass% (0.560 mol) in the total reaction product. (Constant c = 11.15)
A glass container was charged with 165 parts (0.6 mol) of the reaction product and 62 parts (0.63 mol) of maleic anhydride and reacted at 65 ° C. for 8 hours to obtain a monoester product. Thereafter, 84 parts (0.67 mol) of sodium sulfite was dissolved in 690 parts of water, 227 parts of the above monoester product was added at 60 ° C., and reacted at the same temperature for about 5 hours to obtain a sulfonated product. Thereafter, the pH was adjusted to 6.5 with citric acid to obtain an approximately 30% aqueous solution of a comparative sulfosuccinic acid ester salt.
[0046]
Comparative production example 3
Into a stainless steel autoclave with stirring and temperature control function, 186 parts (1 mol) of lauryl alcohol and 0.6 part of potassium hydroxide were added, the inside of the mixed system was replaced with nitrogen, and then under reduced pressure (about 20 mmHg). Dehydration was performed at 120 ° C. for 1 hour. Next, 88 parts (2 mol) of EO, 58 parts (1 mol) of PO, 44 parts (1 mol) of EO at 130 ° C., and the gauge pressure is 1 to 3 kgf / cm. 2 Introduced to become.
As a result of measuring the reaction product under GPC measurement conditions, Mw / Mn was 1.12 [upper calculation value of Mw / Mn satisfying general formula (2); 1.052], unreacted measured by GC measurement conditions. Lauryl alcohol was 7.3% by mass in the total reaction product. (Constant c = 2.97)
A glass container was charged with 188 parts (0.50 mol) of the reaction product and 52 parts (0.53 mol) of maleic anhydride, and reacted at 65 ° C. for 8 hours to obtain a monoester product. Thereafter, 69 parts (0.55 mol) of sodium sulfite was dissolved in 691 parts of water, 240 parts of the monoester product was added at 60 ° C., and reacted at the same temperature for about 5 hours to obtain a sulfonated product. Thereafter, the pH was adjusted to 6.5 with citric acid to obtain an approximately 30% aqueous solution of a comparative sulfosuccinic acid ester salt.
[0047]
Examples 4-8 and Comparative Examples 4-6
The anionic surfactants comprising the sulfosuccinic acid ester salts of Examples 1 to 3 of the present invention and the anionic surfactants of Comparative Examples 1 to 3 were blended in the compositions (parts by mass) described in Tables 3 and 4, and this The cleaning composition of the present invention and the comparative cleaning composition were prepared by adding water (remainder) to 100 parts.
[0048]
The anionic surfactants or detergent compositions of Examples 1-8 and Comparative Examples 1-6 were tested for foamability, low and high temperature stability, aqueous solution viscosity, usability and skin irritation. . The results are shown in Tables 1-4.
[0049]
Surfactants, other additives, and test methods used in Examples and Comparative Examples are as follows.
Activator A; polyoxyethylene (2 mol) sodium lauryl sulfate
Activator B; polyoxyethylene (4 mol) lauryl sulfate triethanolamine
Activator C; sodium polyoxyethylene (3 mol) lauryl ether acetate
Activator D: Polyoxyethylene (3 mol) coconut oil fatty acid sodium monoethanolamide sulfate
Activator F; polyoxyethylene sulfosuccinate (5 mol) lauroylethanolamide disodium
Activator G: Palm oil fatty acid methyl taurine sodium
Activator H; coconut oil fatty acid sarcosine sodium
Activator I; N-coconut oil fatty acid acyl-L-glutamate sodium salt
Activator J; sodium lauroylmethyl-β-alanine
Activator K; palm oil fatty acid amidopropyldimethylaminoacetic acid betaine
Activator L; lauryldimethylaminoacetic acid betaine
Activator M; 2-alkyl-N-carboxymethyl-N-hydroxyethylimidazolinium betaine
Activator N; lauryl hydroxysulfobetaine
Activator O; 1: 1 type coconut oil fatty acid diethanolamide
Activator P; Lauryldimethylamine oxide
Activator Q; ethylene glycol monostearate
Activator R; polyoxyethylene (120 mol) dioleic acid methyl glucoside
Activator S; stearyltrimethylammonium chloride
Activator T; Ethanol sulfate lanolin fatty acid aminopropylethyldimethylammonium
[0050]
Moisturizer A; glycerin
Moisturizer B; sodium pyrrolidonecarboxylate
Polymer A; cationized cellulose
Polymer B; Cationized guar gum
Polymer C: polyethylene glycol (molecular weight 1500)
Polymer D: dimethylpolysiloxane
Polymer E; hydrolyzed protein
Chelate A; ethylenediaminetetraacetic acid sodium salt
Chelate B; 1-hydroxyethane-1, 1-diphosphonate sodium
Alcohol A: propylene glycol
Alcohol B: Dipropylene glycol
[0051]
Test method
<Foamability>
Using hard water of 15 ppm in terms of CaO, prepare 200 ml of 0.3% (solid content) aqueous solution of an anionic surfactant or detergent composition, and stir for 30 seconds with a juicer mixer (Toshiba MX-390GX). The foamability was evaluated by the foam height (mm).
[0052]
<Stability over time; Appearance>
An anionic surfactant or detergent composition is placed in a 100 ml glass bottle and the appearance after standing in a thermostatic bath at 0 ° C., 25 ° C. and 50 ° C. for 30 days is observed with the naked eye and evaluated according to the following criteria: did.
Evaluation criteria
○: Transparent liquid
Δ: Slight turbidity / slight separation tendency
×: Significant turbidity / separation
<Stability over time; Hue>
An anionic surfactant or detergent composition is placed in a 100 ml glass bottle and left in a thermostatic bath at 0 ° C., 25 ° C. and 50 ° C. for 30 days, and then the degree of coloring is observed with the naked eye. It was evaluated with.
Evaluation criteria
○: No coloring
Δ: Slightly colored
×: Remarkably colored
<Stability over time; odor>
An anionic surfactant or detergent composition is placed in a 100 ml glass bottle and left in a thermostatic bath at 0 ° C., 25 ° C. and 50 ° C. for 30 days. It was evaluated with.
Evaluation criteria
○: No change
Δ: Slight odor
×: Remarkably odor
[0053]
<Viscosity>
The viscosity was measured at 25 ° C. using a Brookfield viscometer.
<Usability>
Usability was determined by using a panel of 10 men and women with shampoos using the anionic surfactants or detergent compositions shown in Tables 1 to 4 for “foaming”, “foam quality”, and “texture” at the time of use. Evaluation was made according to the following criteria.
Evaluation criteria
"Bubbling" and "Texture"
○: Good
△: Normal
×: Inferior
"Foam quality"
○: Creamy
△: Normal
×; rough
[0054]
<Skin irritation>
A 1.0% (solid content) aqueous solution of the anionic surfactant or detergent composition shown in Tables 1 to 4 was prepared, and a human patch test (closed, 48 hours, upper arm inner side) by 5 men and women, It was evaluated according to the following criteria and expressed as the total score.
Evaluation criteria
0: No reaction (erythema)
1; Very mild erythema
2; Clear erythema
3; Strong erythema
[0055]
[Table 1]
[0056]
[Table 2]
[0057]
[Table 3]
[0058]
[Table 4]
[0059]
From this result, the anionic surfactant and the detergent composition of the present invention are foamable, stable at low temperatures and high temperatures, easy to be dissolved in an aqueous solution, in a neutral to slightly acidic pH range, It can be seen that the usability and safety to humans are good.
[0060]
【The invention's effect】
Conventionally used sulfosuccinic acid ester salts have problems in foaming properties in a neutral to weakly acidic pH range, stability over time at low and high temperatures, and handling properties of aqueous solutions. The anionic surfactant of the present invention not only solves these problems, but also provides a cleaning composition that satisfies safety for humans.
In addition, sulfosuccinic acid ester salts have been problematic in terms of long-term storage stability at low temperatures and high temperatures (particularly, haze or solidification at low temperatures), but the products of the present invention are remarkably improved. Furthermore, since the anionic surfactant and the detergent composition of the present invention are excellent in surface activity such as permeability in addition to the above-described detergency and foaming property, shampoos, dishwashing detergents, metals, etc. It is particularly useful as a cleaning agent such as a hard surface cleaning agent.
Claims (11)
(i)脂肪族系アルコール(a1)のアルキレンオキサイド(b1)付加物であって、下記一般式(1)で表される化合物の1種または2種以上の混合物からなる。
R1−(OA)k−OH (1)
[式中、R1は炭素数8〜24の脂肪族炭化水素基または炭素数8〜24の脂環式炭化水素基;Aは炭素数2以上の1種以上のアルキレン基;kは、平均が1〜20となる、0または1以上の整数]
(ii)重量平均分子量(Mw)と数平均分子量(Mn)との比:Mw/Mnが下記関係式(2)または(3)を満たす。
Mw/Mn≦0.030×Ln(v)+1.010 (但し、v<10)(2)
Mw/Mn≦−0.026×Ln(v)+1.139(但し、v≧10)(3)
[但し、vは脂肪族系アルコール(a1)1モル当たりに付加したアルキレンオキサイドの平均付加モル数を表し、一般式(1)でのkの平均に相当する。]
(iii)下記式(4)から求められる分布定数cが2.0以下である。(本項はvが12以下の場合のみ適用する。)
c=(v+n0/n00−1)/[Ln(n00/n0)+n0/n00−1] (4)
[但し、vは上記に同じ、n00は反応に用いた(a1)のモル数、n0は未反応の(a1)のモル数を表す。]The sulfosuccinic acid ester salt (B) of the aliphatic alcohol alkylene oxide adduct (A); (A) is an alkylene oxide having 2 or more carbon atoms in the aliphatic alcohol (a1) in the presence of the catalyst (d). An aliphatic catalyst alkylene oxide adduct (A1) or (A1) directly produced by adding (b2) in an average of 1 to 2.5 mol and satisfying the following (i) to (iii) is added to an alkali catalyst (e) An anionic property which is an aliphatic alcohol alkylene oxide adduct (A2) obtained by addition reaction of alkylene oxide (b3) having 2 or more carbon atoms in the presence of Surfactant.
(i) An alkylene oxide (b1) adduct of an aliphatic alcohol (a1), which is composed of one or a mixture of two or more compounds represented by the following general formula (1).
R 1 - (OA) k- OH (1)
[In the formula, R 1 is an aliphatic hydrocarbon group having 8 to 24 carbon atoms or an alicyclic hydrocarbon group having 8 to 24 carbon atoms; A is one or more alkylene groups having 2 or more carbon atoms; Is an integer of 0 or 1 or more]
(ii) Ratio of weight average molecular weight (Mw) to number average molecular weight (Mn): Mw / Mn satisfies the following relational expression (2) or (3).
Mw / Mn ≦ 0.030 × Ln (v) +1.010 (where v <10) (2)
Mw / Mn ≦ −0.026 × Ln (v) +1.139 (where v ≧ 10) (3)
[Wherein v represents the average number of moles of alkylene oxide added per mole of the aliphatic alcohol (a1) and corresponds to the average of k in the general formula (1). ]
(iii) The distribution constant c obtained from the following formula (4) is 2.0 or less. (This item applies only when v is 12 or less.)
c = (v + n 0 / n 00 −1) / [Ln (n 00 / n 0 ) + n 0 / n 00 −1] (4)
[Wherein v is the same as above, n 00 represents the number of moles of (a1) used in the reaction, and n 0 represents the number of moles of unreacted (a1). ]
[式中、R1は炭素数8〜24の脂肪族炭化水素基または炭素数8〜24の脂環式炭化水素基;Aは炭素数2以上の1種以上のアルキレン基;kは平均が1〜20となる0または1以上の整数;Z1およびはZ2は、いずれか一方がH、もう一方がSO3Q1基を表し、XはQ2またはR1−(OA)k−基を表す。Q1およびQ2は、Hまたはカチオン性対イオンを表す。]The anionic surfactant according to any one of claims 1 to 4, wherein the sulfosuccinic acid ester salt (B) comprises at least one salt represented by the following general formula (5).
[Wherein, R 1 is an aliphatic hydrocarbon group having 8 to 24 carbon atoms or an alicyclic hydrocarbon group having 8 to 24 carbon atoms; A is one or more alkylene groups having 2 or more carbon atoms; 0 or an integer of 1 or more and 1 to 20; one of Z 1 and Z 2 represents H, the other represents a SO 3 Q 1 group, and X represents Q 2 or R 1- (OA) k- Represents a group. Q 1 and Q 2 represent H or a cationic counter ion. ]
R1−[(OC2H4)m/(OD)n]−(OC2H4)p−OH (1’)
[式中、R1は炭素数8〜24の脂肪族炭化水素基または炭素数8〜24の脂環式炭化水素基;Dは炭素数3または4のアルキレン基;mは平均が0〜5となる0または1以上の整数、nは平均が0〜5となる0または1以上の整数、pは平均が1〜10となる0または1以上の整数であり、(m+n+p)は平均が1〜20となる整数であり、(m+p)/(m+n+p)は平均0.5以上である。{(OC2H4)m/(OD)n}は、m≠0,n≠0のときブロック付加またはランダム付加を表す。]The anionic surfactant according to any one of claims 1 to 5, wherein the compound represented by the general formula (1) is a compound represented by the following general formula (1 ').
R 1 - [(OC 2 H 4) m / (OD) n] - (OC 2 H 4) p-OH (1 ')
Wherein R 1 is an aliphatic hydrocarbon group having 8 to 24 carbon atoms or an alicyclic hydrocarbon group having 8 to 24 carbon atoms; D is an alkylene group having 3 or 4 carbon atoms; m is an average of 0 to 5 0 or an integer of 1 or more, n is an integer of 0 or 1 or more whose average is 0 to 5, p is an integer of 0 or 1 or more whose average is 1 to 10, and (m + n + p) is 1 It is an integer which becomes -20, (m + p) / (m + n + p) is 0.5 or more on average. {(OC 2 H 4 ) m / (OD) n} represents block addition or random addition when m ≠ 0 and n ≠ 0. ]
(i)脂肪族系アルコール(a1)のアルキレンオキサイド(b1)付加物であって、下記一般式(1)で表される化合物の1種または2種以上の混合物からなる。
R1−(OA)k−OH (1)
[式中、R1は炭素数8〜24の脂肪族炭化水素基または炭素数8〜24の脂環式炭化水素基;Aは炭素数2以上の1種以上のアルキレン基;kは、平均が1〜20となる、0または1以上の整数]
(ii)重量平均分子量(Mw)と数平均分子量(Mn)との比:Mw/Mnが下記関係式(2)または(3)を満たす。
Mw/Mn≦0.030×Ln(v)+1.010 (但し、v<10)(2)
Mw/Mn≦−0.026×Ln(v)+1.139(但し、v≧10)(3)
[但し、vは脂肪族系アルコール(a1)1モル当たりに付加したアルキレンオキサイドの平均付加モル数を表し、一般式(1)でのkの平均に相当する。]
(iii)下記式(4)から求められる分布定数cが2.0以下である。(本項はvが12以下の場合のみ適用する。)
c=(v+n0/n00−1)/[Ln(n00/n0)+n0/n00−1] (4)
[但し、vは上記に同じ、n00は反応に用いた(a1)のモル数、n0は未反応の(a1)のモル数を表す。]The aliphatic alcohol (a1) having 8 to 24 carbon atoms in the presence of one or more catalysts (d) selected from the group consisting of perhalogenic acid (salt), sulfate, phosphate and nitrate has 2 carbon atoms. An aliphatic alcohol alkylene oxide adduct (A1) produced directly by adding 1 to 2.5 moles of the above alkylene oxide (b2) on average and having a distribution constant c calculated from the following formula (4) of 1.0 or less. ) Or (A1) is subjected to an addition reaction with an alkylene oxide (b3) having 2 or more carbon atoms [(b3) may be the same as or different from (b2)] in the presence of an alkali catalyst (e). An aliphatic alcohol alkylene oxide adduct (A2) that satisfies the following (i) to (iii), an unreacted alcohol or an added mole number That Is without operating fractionating, and chloride sulfosuccinates, (A1) or (A2) method for producing an anionic surfactant, characterized in that the production of sulfosuccinic acid ester salt (B) of the.
(i) An alkylene oxide (b1) adduct of an aliphatic alcohol (a1), which is composed of one or a mixture of two or more compounds represented by the following general formula (1).
R 1 - (OA) k- OH (1)
[In the formula, R 1 is an aliphatic hydrocarbon group having 8 to 24 carbon atoms or an alicyclic hydrocarbon group having 8 to 24 carbon atoms; A is one or more alkylene groups having 2 or more carbon atoms; Is an integer of 0 or 1 or more]
(ii) Ratio of weight average molecular weight (Mw) to number average molecular weight (Mn): Mw / Mn satisfies the following relational expression (2) or (3).
Mw / Mn ≦ 0.030 × Ln (v) +1.010 (where v <10) (2)
Mw / Mn ≦ −0.026 × Ln (v) +1.139 (where v ≧ 10) (3)
[Wherein v represents the average number of moles of alkylene oxide added per mole of the aliphatic alcohol (a1) and corresponds to the average of k in the general formula (1). ]
(iii) The distribution constant c obtained from the following formula (4) is 2.0 or less. (This item applies only when v is 12 or less.)
c = (v + n 0 / n 00 −1) / [Ln (n 00 / n 0 ) + n 0 / n 00 −1] (4)
[Wherein v is the same as above, n 00 represents the number of moles of (a1) used in the reaction, and n 0 represents the number of moles of unreacted (a1). ]
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP29389999A JP3634988B2 (en) | 1999-10-15 | 1999-10-15 | Anionic surfactant and detergent composition |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP29389999A JP3634988B2 (en) | 1999-10-15 | 1999-10-15 | Anionic surfactant and detergent composition |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2001115189A JP2001115189A (en) | 2001-04-24 |
| JP3634988B2 true JP3634988B2 (en) | 2005-03-30 |
Family
ID=17800604
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP29389999A Expired - Fee Related JP3634988B2 (en) | 1999-10-15 | 1999-10-15 | Anionic surfactant and detergent composition |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP3634988B2 (en) |
Cited By (28)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10311575B2 (en) | 2016-03-23 | 2019-06-04 | The Procter And Gamble Company | Imaging method for determining stray fibers |
| US10426713B2 (en) | 2017-10-10 | 2019-10-01 | The Procter And Gamble Company | Method of treating hair or skin with a personal care composition in a foam form |
| US10441519B2 (en) | 2016-10-21 | 2019-10-15 | The Procter And Gamble Company | Low viscosity hair care composition comprising a branched anionic/linear anionic surfactant mixture |
| US10653590B2 (en) | 2016-10-21 | 2020-05-19 | The Procter And Gamble Company | Concentrated shampoo dosage of foam for providing hair care benefits comprising an anionic/zwitterionic surfactant mixture |
| US10799434B2 (en) | 2016-10-21 | 2020-10-13 | The Procter & Gamble Company | Concentrated shampoo dosage of foam for providing hair care benefits |
| US10842720B2 (en) | 2016-10-21 | 2020-11-24 | The Procter And Gamble Company | Dosage of foam comprising an anionic/zwitterionic surfactant mixture |
| US10888505B2 (en) | 2016-10-21 | 2021-01-12 | The Procter And Gamble Company | Dosage of foam for delivering consumer desired dosage volume, surfactant amount, and scalp health agent amount in an optimal formulation space |
| US10912732B2 (en) | 2017-12-20 | 2021-02-09 | The Procter And Gamble Company | Clear shampoo composition containing silicone polymers |
| US11116705B2 (en) | 2017-10-10 | 2021-09-14 | The Procter And Gamble Company | Compact shampoo composition containing sulfate-free surfactants |
| US11116704B2 (en) | 2017-10-10 | 2021-09-14 | The Procter And Gamble Company | Compact shampoo composition |
| US11116703B2 (en) | 2017-10-10 | 2021-09-14 | The Procter And Gamble Company | Compact shampoo composition containing sulfate-free surfactants |
| US11129783B2 (en) | 2016-10-21 | 2021-09-28 | The Procter And Gamble Plaza | Stable compact shampoo products with low viscosity and viscosity reducing agent |
| US11141370B2 (en) | 2017-06-06 | 2021-10-12 | The Procter And Gamble Company | Hair compositions comprising a cationic polymer mixture and providing improved in-use wet feel |
| US11141361B2 (en) | 2016-10-21 | 2021-10-12 | The Procter And Gamble Plaza | Concentrated shampoo dosage of foam designating hair volume benefits |
| US11154467B2 (en) | 2016-10-21 | 2021-10-26 | The Procter And Gamble Plaza | Concentrated shampoo dosage of foam designating hair conditioning benefits |
| US11224567B2 (en) | 2017-06-06 | 2022-01-18 | The Procter And Gamble Company | Hair compositions comprising a cationic polymer/silicone mixture providing improved in-use wet feel |
| US11291616B2 (en) | 2015-04-23 | 2022-04-05 | The Procter And Gamble Company | Delivery of surfactant soluble anti-dandruff agent |
| US11318073B2 (en) | 2018-06-29 | 2022-05-03 | The Procter And Gamble Company | Low surfactant aerosol antidandruff composition |
| US11446217B2 (en) | 2016-03-03 | 2022-09-20 | The Procter & Gamble Company | Aerosol antidandruff composition |
| US11679073B2 (en) | 2017-06-06 | 2023-06-20 | The Procter & Gamble Company | Hair compositions providing improved in-use wet feel |
| US11679065B2 (en) | 2020-02-27 | 2023-06-20 | The Procter & Gamble Company | Compositions with sulfur having enhanced efficacy and aesthetics |
| US11771635B2 (en) | 2021-05-14 | 2023-10-03 | The Procter & Gamble Company | Shampoo composition |
| US11819474B2 (en) | 2020-12-04 | 2023-11-21 | The Procter & Gamble Company | Hair care compositions comprising malodor reduction materials |
| US11980679B2 (en) | 2019-12-06 | 2024-05-14 | The Procter & Gamble Company | Sulfate free composition with enhanced deposition of scalp active |
| US11986543B2 (en) | 2021-06-01 | 2024-05-21 | The Procter & Gamble Company | Rinse-off compositions with a surfactant system that is substantially free of sulfate-based surfactants |
| US12226505B2 (en) | 2018-10-25 | 2025-02-18 | The Procter & Gamble Company | Compositions having enhanced deposition of surfactant-soluble anti-dandruff agents |
| US12427099B2 (en) | 2020-11-23 | 2025-09-30 | The Procter & Gamble Company | Personal care composition |
| US12458575B2 (en) | 2021-12-09 | 2025-11-04 | The Procter & Gamble Company | Sulfate free personal cleansing composition comprising effective preservation |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP3635064B2 (en) * | 1998-09-29 | 2005-03-30 | 三洋化成工業株式会社 | Method for producing nonionic surfactant |
| JP5272676B2 (en) * | 2008-11-19 | 2013-08-28 | 日油株式会社 | Cleaning composition |
| EP3285723A1 (en) * | 2015-04-23 | 2018-02-28 | The Procter and Gamble Company | A low viscosity hair care composition |
| JP6910190B2 (en) * | 2017-04-25 | 2021-07-28 | ライオン株式会社 | Liquid detergent composition |
-
1999
- 1999-10-15 JP JP29389999A patent/JP3634988B2/en not_active Expired - Fee Related
Cited By (34)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11291616B2 (en) | 2015-04-23 | 2022-04-05 | The Procter And Gamble Company | Delivery of surfactant soluble anti-dandruff agent |
| US11446217B2 (en) | 2016-03-03 | 2022-09-20 | The Procter & Gamble Company | Aerosol antidandruff composition |
| US10311575B2 (en) | 2016-03-23 | 2019-06-04 | The Procter And Gamble Company | Imaging method for determining stray fibers |
| US10653590B2 (en) | 2016-10-21 | 2020-05-19 | The Procter And Gamble Company | Concentrated shampoo dosage of foam for providing hair care benefits comprising an anionic/zwitterionic surfactant mixture |
| US10799434B2 (en) | 2016-10-21 | 2020-10-13 | The Procter & Gamble Company | Concentrated shampoo dosage of foam for providing hair care benefits |
| US10842720B2 (en) | 2016-10-21 | 2020-11-24 | The Procter And Gamble Company | Dosage of foam comprising an anionic/zwitterionic surfactant mixture |
| US10888505B2 (en) | 2016-10-21 | 2021-01-12 | The Procter And Gamble Company | Dosage of foam for delivering consumer desired dosage volume, surfactant amount, and scalp health agent amount in an optimal formulation space |
| US11154467B2 (en) | 2016-10-21 | 2021-10-26 | The Procter And Gamble Plaza | Concentrated shampoo dosage of foam designating hair conditioning benefits |
| US10441519B2 (en) | 2016-10-21 | 2019-10-15 | The Procter And Gamble Company | Low viscosity hair care composition comprising a branched anionic/linear anionic surfactant mixture |
| US11202740B2 (en) | 2016-10-21 | 2021-12-21 | The Procter And Gamble Company | Concentrated shampoo dosage of foam for providing hair care benefits |
| US11129783B2 (en) | 2016-10-21 | 2021-09-28 | The Procter And Gamble Plaza | Stable compact shampoo products with low viscosity and viscosity reducing agent |
| US11141361B2 (en) | 2016-10-21 | 2021-10-12 | The Procter And Gamble Plaza | Concentrated shampoo dosage of foam designating hair volume benefits |
| US11679073B2 (en) | 2017-06-06 | 2023-06-20 | The Procter & Gamble Company | Hair compositions providing improved in-use wet feel |
| US11224567B2 (en) | 2017-06-06 | 2022-01-18 | The Procter And Gamble Company | Hair compositions comprising a cationic polymer/silicone mixture providing improved in-use wet feel |
| US11141370B2 (en) | 2017-06-06 | 2021-10-12 | The Procter And Gamble Company | Hair compositions comprising a cationic polymer mixture and providing improved in-use wet feel |
| US11116705B2 (en) | 2017-10-10 | 2021-09-14 | The Procter And Gamble Company | Compact shampoo composition containing sulfate-free surfactants |
| US11129775B2 (en) | 2017-10-10 | 2021-09-28 | The Procter And Gamble Company | Method of treating hair or skin with a personal care composition in a foam form |
| US11116703B2 (en) | 2017-10-10 | 2021-09-14 | The Procter And Gamble Company | Compact shampoo composition containing sulfate-free surfactants |
| US11116704B2 (en) | 2017-10-10 | 2021-09-14 | The Procter And Gamble Company | Compact shampoo composition |
| US11992540B2 (en) | 2017-10-10 | 2024-05-28 | The Procter & Gamble Company | Sulfate free personal cleansing composition comprising low inorganic salt |
| US11607373B2 (en) | 2017-10-10 | 2023-03-21 | The Procter & Gamble Company | Sulfate free clear personal cleansing composition comprising low inorganic salt |
| US10426713B2 (en) | 2017-10-10 | 2019-10-01 | The Procter And Gamble Company | Method of treating hair or skin with a personal care composition in a foam form |
| US11904036B2 (en) | 2017-10-10 | 2024-02-20 | The Procter & Gamble Company | Sulfate free clear personal cleansing composition comprising low inorganic salt |
| US10912732B2 (en) | 2017-12-20 | 2021-02-09 | The Procter And Gamble Company | Clear shampoo composition containing silicone polymers |
| US11318073B2 (en) | 2018-06-29 | 2022-05-03 | The Procter And Gamble Company | Low surfactant aerosol antidandruff composition |
| US12226505B2 (en) | 2018-10-25 | 2025-02-18 | The Procter & Gamble Company | Compositions having enhanced deposition of surfactant-soluble anti-dandruff agents |
| US11980679B2 (en) | 2019-12-06 | 2024-05-14 | The Procter & Gamble Company | Sulfate free composition with enhanced deposition of scalp active |
| US11679065B2 (en) | 2020-02-27 | 2023-06-20 | The Procter & Gamble Company | Compositions with sulfur having enhanced efficacy and aesthetics |
| US12427099B2 (en) | 2020-11-23 | 2025-09-30 | The Procter & Gamble Company | Personal care composition |
| US11819474B2 (en) | 2020-12-04 | 2023-11-21 | The Procter & Gamble Company | Hair care compositions comprising malodor reduction materials |
| US11771635B2 (en) | 2021-05-14 | 2023-10-03 | The Procter & Gamble Company | Shampoo composition |
| US12409125B2 (en) | 2021-05-14 | 2025-09-09 | The Procter & Gamble Company | Shampoo compositions containing a sulfate-free surfactant system and sclerotium gum thickener |
| US11986543B2 (en) | 2021-06-01 | 2024-05-21 | The Procter & Gamble Company | Rinse-off compositions with a surfactant system that is substantially free of sulfate-based surfactants |
| US12458575B2 (en) | 2021-12-09 | 2025-11-04 | The Procter & Gamble Company | Sulfate free personal cleansing composition comprising effective preservation |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2001115189A (en) | 2001-04-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP3634988B2 (en) | Anionic surfactant and detergent composition | |
| JP3634991B2 (en) | Anionic surfactant and detergent composition | |
| JP3634996B2 (en) | Anionic surfactant and detergent and water-soluble lubricant composition containing the same | |
| KR101646802B1 (en) | Surfactant composition | |
| US4372869A (en) | Detergent compositions | |
| JP2964226B2 (en) | Detergent composition | |
| US4186113A (en) | Low irritating detergent compositions | |
| JP6014380B2 (en) | Foam enhancer and detergent composition containing the same | |
| GB2035362A (en) | Detergent compositions | |
| EP0068868A1 (en) | Detergent compounds and compositions | |
| JPH0753997A (en) | Isoprene glycol-containing fatty acid salt-based solid transparent soap composition | |
| JPH02175799A (en) | Detergent composition | |
| US5922659A (en) | Cleanser composition | |
| EP0496359B1 (en) | Liquid detergent composition | |
| EP0612518A1 (en) | Cosmetic composition containing a phosphate surfactant and a co-surfactant | |
| JP3525848B2 (en) | Anionic surfactant and detergent composition | |
| USRE30641E (en) | Low irritating detergent compositions | |
| JP2005206804A (en) | Cleaning composition | |
| US4812254A (en) | Detergent composition | |
| JP7708490B2 (en) | Skin cleansing composition | |
| JP4647808B2 (en) | Thickener for liquid detergent and liquid detergent composition containing the same | |
| KR102644492B1 (en) | Liquid skin cleanser composition | |
| JPH0292998A (en) | liquid cleaning composition | |
| JP3995814B2 (en) | Foam booster | |
| JP2006022116A (en) | Cleaning composition |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20040203 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20040210 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20040406 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20041221 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20041227 |
|
| R150 | Certificate of patent or registration of utility model |
Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20080107 Year of fee payment: 3 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20090107 Year of fee payment: 4 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20100107 Year of fee payment: 5 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20100107 Year of fee payment: 5 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20110107 Year of fee payment: 6 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20110107 Year of fee payment: 6 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20120107 Year of fee payment: 7 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20120107 Year of fee payment: 7 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20130107 Year of fee payment: 8 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20140107 Year of fee payment: 9 |
|
| LAPS | Cancellation because of no payment of annual fees |