JP2018024660A - Amide compound and pest control agent - Google Patents
Amide compound and pest control agent Download PDFInfo
- Publication number
- JP2018024660A JP2018024660A JP2017151963A JP2017151963A JP2018024660A JP 2018024660 A JP2018024660 A JP 2018024660A JP 2017151963 A JP2017151963 A JP 2017151963A JP 2017151963 A JP2017151963 A JP 2017151963A JP 2018024660 A JP2018024660 A JP 2018024660A
- Authority
- JP
- Japan
- Prior art keywords
- halo
- alkyl
- hydrogen atom
- cycloalkyl
- oet
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- -1 Amide compound Chemical class 0.000 title claims abstract description 208
- 241000607479 Yersinia pestis Species 0.000 title claims abstract description 32
- 125000000217 alkyl group Chemical group 0.000 claims abstract description 34
- 150000003839 salts Chemical class 0.000 claims abstract description 34
- 239000004480 active ingredient Substances 0.000 claims abstract description 19
- 150000001204 N-oxides Chemical class 0.000 claims abstract description 12
- 241000124008 Mammalia Species 0.000 claims abstract description 11
- 125000005843 halogen group Chemical group 0.000 claims description 122
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 85
- 239000003795 chemical substances by application Substances 0.000 claims description 54
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 claims description 34
- 125000005913 (C3-C6) cycloalkyl group Chemical group 0.000 claims description 32
- 125000000392 cycloalkenyl group Chemical group 0.000 claims description 30
- 125000006727 (C1-C6) alkenyl group Chemical group 0.000 claims description 26
- 125000006728 (C1-C6) alkynyl group Chemical group 0.000 claims description 26
- 150000002391 heterocyclic compounds Chemical class 0.000 claims description 25
- 125000004916 (C1-C6) alkylcarbonyl group Chemical group 0.000 claims description 22
- 125000004454 (C1-C6) alkoxycarbonyl group Chemical group 0.000 claims description 20
- 125000004739 (C1-C6) alkylsulfonyl group Chemical group 0.000 claims description 20
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 claims description 18
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 16
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 14
- 125000000753 cycloalkyl group Chemical group 0.000 claims description 14
- 239000002917 insecticide Substances 0.000 claims description 11
- 244000078703 ectoparasite Species 0.000 claims description 10
- 125000000304 alkynyl group Chemical group 0.000 claims description 8
- 239000002689 soil Substances 0.000 claims description 8
- 230000000895 acaricidal effect Effects 0.000 claims description 7
- 239000003905 agrochemical Substances 0.000 claims description 7
- 239000000642 acaricide Substances 0.000 claims description 6
- 125000003342 alkenyl group Chemical group 0.000 claims description 6
- 125000004453 alkoxycarbonyl group Chemical group 0.000 claims description 6
- 238000007598 dipping method Methods 0.000 claims description 6
- 230000002262 irrigation Effects 0.000 claims description 2
- 238000003973 irrigation Methods 0.000 claims description 2
- 150000001875 compounds Chemical class 0.000 abstract description 225
- 241000238631 Hexapoda Species 0.000 abstract description 19
- 230000000694 effects Effects 0.000 abstract description 12
- 241000251468 Actinopterygii Species 0.000 abstract description 6
- 230000009286 beneficial effect Effects 0.000 abstract description 6
- 231100000053 low toxicity Toxicity 0.000 abstract description 3
- 229910052736 halogen Inorganic materials 0.000 abstract 2
- 125000001475 halogen functional group Chemical group 0.000 abstract 2
- 150000002367 halogens Chemical class 0.000 abstract 2
- 239000000460 chlorine Substances 0.000 description 360
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 78
- 239000011541 reaction mixture Substances 0.000 description 69
- 230000015572 biosynthetic process Effects 0.000 description 62
- 238000003786 synthesis reaction Methods 0.000 description 62
- 238000006243 chemical reaction Methods 0.000 description 59
- 239000000243 solution Substances 0.000 description 59
- 239000002904 solvent Substances 0.000 description 59
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 58
- 239000007787 solid Substances 0.000 description 47
- 238000012360 testing method Methods 0.000 description 47
- 239000000203 mixture Substances 0.000 description 42
- 125000000738 acetamido group Chemical group [H]C([H])([H])C(=O)N([H])[*] 0.000 description 37
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 36
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 36
- 125000004432 carbon atom Chemical group C* 0.000 description 36
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 34
- 238000000034 method Methods 0.000 description 31
- 238000005160 1H NMR spectroscopy Methods 0.000 description 29
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 29
- 238000002360 preparation method Methods 0.000 description 29
- 239000000047 product Substances 0.000 description 29
- 239000000126 substance Substances 0.000 description 29
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 28
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 27
- 238000004519 manufacturing process Methods 0.000 description 26
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 description 25
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 24
- 239000011259 mixed solution Substances 0.000 description 23
- 239000012044 organic layer Substances 0.000 description 23
- 241000254173 Coleoptera Species 0.000 description 21
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 21
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 21
- 239000000839 emulsion Substances 0.000 description 21
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 20
- 238000009472 formulation Methods 0.000 description 19
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N silicon dioxide Inorganic materials O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 19
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 18
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 18
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 18
- 238000010576 medium-pressure preparative liquid chromatography Methods 0.000 description 18
- 239000007788 liquid Substances 0.000 description 16
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 15
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 15
- 125000006125 ethylsulfonyl group Chemical group 0.000 description 15
- 238000010992 reflux Methods 0.000 description 15
- 125000004705 ethylthio group Chemical group C(C)S* 0.000 description 14
- 238000002844 melting Methods 0.000 description 14
- 230000008018 melting Effects 0.000 description 14
- 241000282472 Canis lupus familiaris Species 0.000 description 13
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 13
- 241000255925 Diptera Species 0.000 description 13
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 13
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical group C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 13
- 239000002585 base Substances 0.000 description 13
- 239000000543 intermediate Substances 0.000 description 13
- 229920006395 saturated elastomer Polymers 0.000 description 13
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 12
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 12
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 12
- 235000005911 diet Nutrition 0.000 description 12
- 230000037213 diet Effects 0.000 description 12
- 239000004094 surface-active agent Substances 0.000 description 12
- 239000004563 wettable powder Substances 0.000 description 12
- VHYFNPMBLIVWCW-UHFFFAOYSA-N 4-Dimethylaminopyridine Chemical compound CN(C)C1=CC=NC=C1 VHYFNPMBLIVWCW-UHFFFAOYSA-N 0.000 description 11
- 241000238876 Acari Species 0.000 description 11
- 239000008187 granular material Substances 0.000 description 11
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 11
- 235000017557 sodium bicarbonate Nutrition 0.000 description 11
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 10
- 241001674048 Phthiraptera Species 0.000 description 10
- 239000002552 dosage form Substances 0.000 description 10
- OAYLNYINCPYISS-UHFFFAOYSA-N ethyl acetate;hexane Chemical compound CCCCCC.CCOC(C)=O OAYLNYINCPYISS-UHFFFAOYSA-N 0.000 description 10
- 238000000605 extraction Methods 0.000 description 10
- 239000003921 oil Substances 0.000 description 10
- 235000019198 oils Nutrition 0.000 description 10
- IBBMAWULFFBRKK-UHFFFAOYSA-N picolinamide Chemical compound NC(=O)C1=CC=CC=N1 IBBMAWULFFBRKK-UHFFFAOYSA-N 0.000 description 10
- 239000000843 powder Substances 0.000 description 10
- 230000007480 spreading Effects 0.000 description 10
- 238000003892 spreading Methods 0.000 description 10
- GQHTUMJGOHRCHB-UHFFFAOYSA-N 2,3,4,6,7,8,9,10-octahydropyrimido[1,2-a]azepine Chemical compound C1CCCCN2CCCN=C21 GQHTUMJGOHRCHB-UHFFFAOYSA-N 0.000 description 9
- OISVCGZHLKNMSJ-UHFFFAOYSA-N 2,6-dimethylpyridine Chemical compound CC1=CC=CC(C)=N1 OISVCGZHLKNMSJ-UHFFFAOYSA-N 0.000 description 9
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 9
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 9
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 9
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 9
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 9
- 229910052799 carbon Inorganic materials 0.000 description 9
- 235000020971 citrus fruits Nutrition 0.000 description 9
- 239000011521 glass Substances 0.000 description 9
- 241000256837 Apidae Species 0.000 description 8
- XTHFKEDIFFGKHM-UHFFFAOYSA-N Dimethoxyethane Chemical compound COCCOC XTHFKEDIFFGKHM-UHFFFAOYSA-N 0.000 description 8
- 241001465754 Metazoa Species 0.000 description 8
- 241000244206 Nematoda Species 0.000 description 8
- OFBQJSOFQDEBGM-UHFFFAOYSA-N Pentane Chemical compound CCCCC OFBQJSOFQDEBGM-UHFFFAOYSA-N 0.000 description 8
- APEPLROGLDYWBS-UHFFFAOYSA-N [2,3,5,6-tetrafluoro-4-(methoxymethyl)phenyl]methyl 2,2,3,3-tetramethylcyclopropane-1-carboxylate Chemical compound FC1=C(F)C(COC)=C(F)C(F)=C1COC(=O)C1C(C)(C)C1(C)C APEPLROGLDYWBS-UHFFFAOYSA-N 0.000 description 8
- 239000002253 acid Substances 0.000 description 8
- QARVLSVVCXYDNA-UHFFFAOYSA-N bromobenzene Chemical compound BrC1=CC=CC=C1 QARVLSVVCXYDNA-UHFFFAOYSA-N 0.000 description 8
- MVPPADPHJFYWMZ-UHFFFAOYSA-N chlorobenzene Chemical compound ClC1=CC=CC=C1 MVPPADPHJFYWMZ-UHFFFAOYSA-N 0.000 description 8
- DNJIEGIFACGWOD-UHFFFAOYSA-N ethanethiol Chemical compound CCS DNJIEGIFACGWOD-UHFFFAOYSA-N 0.000 description 8
- 238000002347 injection Methods 0.000 description 8
- 239000007924 injection Substances 0.000 description 8
- 239000010410 layer Substances 0.000 description 8
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 8
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 8
- 241000239290 Araneae Species 0.000 description 7
- 241000207199 Citrus Species 0.000 description 7
- 240000007594 Oryza sativa Species 0.000 description 7
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 7
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 7
- 150000001338 aliphatic hydrocarbons Chemical class 0.000 description 7
- 150000004945 aromatic hydrocarbons Chemical class 0.000 description 7
- 238000010438 heat treatment Methods 0.000 description 7
- 230000000749 insecticidal effect Effects 0.000 description 7
- 238000005507 spraying Methods 0.000 description 7
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 6
- NHQDETIJWKXCTC-UHFFFAOYSA-N 3-chloroperbenzoic acid Chemical compound OOC(=O)C1=CC=CC(Cl)=C1 NHQDETIJWKXCTC-UHFFFAOYSA-N 0.000 description 6
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 6
- MHAJPDPJQMAIIY-UHFFFAOYSA-N Hydrogen peroxide Chemical compound OO MHAJPDPJQMAIIY-UHFFFAOYSA-N 0.000 description 6
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 6
- 235000007164 Oryza sativa Nutrition 0.000 description 6
- 241001414989 Thysanoptera Species 0.000 description 6
- 239000003945 anionic surfactant Substances 0.000 description 6
- 150000001721 carbon Chemical group 0.000 description 6
- 239000004927 clay Substances 0.000 description 6
- 150000002148 esters Chemical class 0.000 description 6
- 239000003112 inhibitor Substances 0.000 description 6
- LULAYUGMBFYYEX-UHFFFAOYSA-N metachloroperbenzoic acid Natural products OC(=O)C1=CC=CC(Cl)=C1 LULAYUGMBFYYEX-UHFFFAOYSA-N 0.000 description 6
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 6
- 235000009566 rice Nutrition 0.000 description 6
- AKHNMLFCWUSKQB-UHFFFAOYSA-L sodium thiosulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=S AKHNMLFCWUSKQB-UHFFFAOYSA-L 0.000 description 6
- 235000019345 sodium thiosulphate Nutrition 0.000 description 6
- 239000000725 suspension Substances 0.000 description 6
- 239000008096 xylene Substances 0.000 description 6
- 241000271566 Aves Species 0.000 description 5
- YIIBGNBAMHOSNI-UHFFFAOYSA-N C(C)S(=O)(=O)C1=C(N=C2N1C=CC(=C2)C(F)(F)F)C(=O)Cl Chemical compound C(C)S(=O)(=O)C1=C(N=C2N1C=CC(=C2)C(F)(F)F)C(=O)Cl YIIBGNBAMHOSNI-UHFFFAOYSA-N 0.000 description 5
- 241000258924 Ctenocephalides felis Species 0.000 description 5
- 241000256602 Isoptera Species 0.000 description 5
- FXHOOIRPVKKKFG-UHFFFAOYSA-N N,N-Dimethylacetamide Chemical compound CN(C)C(C)=O FXHOOIRPVKKKFG-UHFFFAOYSA-N 0.000 description 5
- 241001454295 Tetranychidae Species 0.000 description 5
- 150000001298 alcohols Chemical class 0.000 description 5
- 150000001408 amides Chemical class 0.000 description 5
- 230000000844 anti-bacterial effect Effects 0.000 description 5
- 239000003899 bactericide agent Substances 0.000 description 5
- 239000011230 binding agent Substances 0.000 description 5
- 239000004359 castor oil Substances 0.000 description 5
- 235000019438 castor oil Nutrition 0.000 description 5
- 238000001816 cooling Methods 0.000 description 5
- 238000000354 decomposition reaction Methods 0.000 description 5
- 235000014113 dietary fatty acids Nutrition 0.000 description 5
- 239000003814 drug Substances 0.000 description 5
- 239000000428 dust Substances 0.000 description 5
- FQYYIPZPELSLDK-UHFFFAOYSA-N ethyl pyridine-2-carboxylate Chemical compound CCOC(=O)C1=CC=CC=N1 FQYYIPZPELSLDK-UHFFFAOYSA-N 0.000 description 5
- 239000000194 fatty acid Substances 0.000 description 5
- 229930195729 fatty acid Natural products 0.000 description 5
- 239000000499 gel Substances 0.000 description 5
- ZEMPKEQAKRGZGQ-XOQCFJPHSA-N glycerol triricinoleate Natural products CCCCCC[C@@H](O)CC=CCCCCCCCC(=O)OC[C@@H](COC(=O)CCCCCCCC=CC[C@@H](O)CCCCCC)OC(=O)CCCCCCCC=CC[C@H](O)CCCCCC ZEMPKEQAKRGZGQ-XOQCFJPHSA-N 0.000 description 5
- 238000002156 mixing Methods 0.000 description 5
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 5
- 239000007800 oxidant agent Substances 0.000 description 5
- 244000045947 parasite Species 0.000 description 5
- 235000012239 silicon dioxide Nutrition 0.000 description 5
- 150000003462 sulfoxides Chemical class 0.000 description 5
- 229930195735 unsaturated hydrocarbon Natural products 0.000 description 5
- WSLDOOZREJYCGB-UHFFFAOYSA-N 1,2-Dichloroethane Chemical compound ClCCCl WSLDOOZREJYCGB-UHFFFAOYSA-N 0.000 description 4
- CYSGHNMQYZDMIA-UHFFFAOYSA-N 1,3-Dimethyl-2-imidazolidinon Chemical compound CN1CCN(C)C1=O CYSGHNMQYZDMIA-UHFFFAOYSA-N 0.000 description 4
- SGUVLZREKBPKCE-UHFFFAOYSA-N 1,5-diazabicyclo[4.3.0]-non-5-ene Chemical compound C1CCN=C2CCCN21 SGUVLZREKBPKCE-UHFFFAOYSA-N 0.000 description 4
- FVBQAOHNUSJHJQ-UHFFFAOYSA-N 3-ethylsulfanyl-7-(trifluoromethyl)imidazo[1,2-a]pyridine-2-carboxylic acid Chemical compound C(C)SC1=C(N=C2N1C=CC(=C2)C(F)(F)F)C(=O)O FVBQAOHNUSJHJQ-UHFFFAOYSA-N 0.000 description 4
- 241000283690 Bos taurus Species 0.000 description 4
- CBXLBLHFESTOOA-UHFFFAOYSA-N CCSC1=C(N=C2N1C=CC(=C2)I)C(=O)O Chemical compound CCSC1=C(N=C2N1C=CC(=C2)I)C(=O)O CBXLBLHFESTOOA-UHFFFAOYSA-N 0.000 description 4
- 241000242722 Cestoda Species 0.000 description 4
- 241001498622 Cixius wagneri Species 0.000 description 4
- XDTMQSROBMDMFD-UHFFFAOYSA-N Cyclohexane Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 description 4
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 4
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 4
- JRNVZBWKYDBUCA-UHFFFAOYSA-N N-chlorosuccinimide Chemical compound ClN1C(=O)CCC1=O JRNVZBWKYDBUCA-UHFFFAOYSA-N 0.000 description 4
- 241001556089 Nilaparvata lugens Species 0.000 description 4
- 241001494479 Pecora Species 0.000 description 4
- 239000002202 Polyethylene glycol Substances 0.000 description 4
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical compound [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 description 4
- 241000985245 Spodoptera litura Species 0.000 description 4
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 4
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 4
- 239000000654 additive Substances 0.000 description 4
- 239000000443 aerosol Substances 0.000 description 4
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 description 4
- 239000003963 antioxidant agent Substances 0.000 description 4
- 235000006708 antioxidants Nutrition 0.000 description 4
- 239000007864 aqueous solution Substances 0.000 description 4
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 4
- 239000002775 capsule Substances 0.000 description 4
- 239000000969 carrier Substances 0.000 description 4
- 239000003054 catalyst Substances 0.000 description 4
- 229920002678 cellulose Polymers 0.000 description 4
- 239000001913 cellulose Substances 0.000 description 4
- XBDQKXXYIPTUBI-UHFFFAOYSA-N dimethylselenoniopropionate Natural products CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 4
- 229940079593 drug Drugs 0.000 description 4
- 244000079386 endoparasite Species 0.000 description 4
- 150000002170 ethers Chemical class 0.000 description 4
- 238000001914 filtration Methods 0.000 description 4
- 235000013305 food Nutrition 0.000 description 4
- 150000008282 halocarbons Chemical class 0.000 description 4
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 4
- UTCSSFWDNNEEBH-UHFFFAOYSA-N imidazo[1,2-a]pyridine Chemical compound C1=CC=CC2=NC=CN21 UTCSSFWDNNEEBH-UHFFFAOYSA-N 0.000 description 4
- JEIPFZHSYJVQDO-UHFFFAOYSA-N iron(III) oxide Inorganic materials O=[Fe]O[Fe]=O JEIPFZHSYJVQDO-UHFFFAOYSA-N 0.000 description 4
- 229940057995 liquid paraffin Drugs 0.000 description 4
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 4
- 125000002950 monocyclic group Chemical group 0.000 description 4
- 239000002736 nonionic surfactant Substances 0.000 description 4
- 239000002674 ointment Substances 0.000 description 4
- CTSLXHKWHWQRSH-UHFFFAOYSA-N oxalyl chloride Chemical compound ClC(=O)C(Cl)=O CTSLXHKWHWQRSH-UHFFFAOYSA-N 0.000 description 4
- 239000006187 pill Substances 0.000 description 4
- 229920001223 polyethylene glycol Polymers 0.000 description 4
- 229920001296 polysiloxane Polymers 0.000 description 4
- 229910000027 potassium carbonate Inorganic materials 0.000 description 4
- 235000011181 potassium carbonates Nutrition 0.000 description 4
- FVSKHRXBFJPNKK-UHFFFAOYSA-N propionitrile Chemical compound CCC#N FVSKHRXBFJPNKK-UHFFFAOYSA-N 0.000 description 4
- 230000035484 reaction time Effects 0.000 description 4
- 239000012312 sodium hydride Substances 0.000 description 4
- 229910000104 sodium hydride Inorganic materials 0.000 description 4
- 239000007921 spray Substances 0.000 description 4
- 239000000758 substrate Substances 0.000 description 4
- 239000003826 tablet Substances 0.000 description 4
- 239000002562 thickening agent Substances 0.000 description 4
- 238000005406 washing Methods 0.000 description 4
- KAATUXNTWXVJKI-NSHGMRRFSA-N (1R)-cis-(alphaS)-cypermethrin Chemical compound CC1(C)[C@@H](C=C(Cl)Cl)[C@H]1C(=O)O[C@H](C#N)C1=CC=CC(OC=2C=CC=CC=2)=C1 KAATUXNTWXVJKI-NSHGMRRFSA-N 0.000 description 3
- CYPYTURSJDMMMP-WVCUSYJESA-N (1e,4e)-1,5-diphenylpenta-1,4-dien-3-one;palladium Chemical compound [Pd].[Pd].C=1C=CC=CC=1\C=C\C(=O)\C=C\C1=CC=CC=C1.C=1C=CC=CC=1\C=C\C(=O)\C=C\C1=CC=CC=C1.C=1C=CC=CC=1\C=C\C(=O)\C=C\C1=CC=CC=C1 CYPYTURSJDMMMP-WVCUSYJESA-N 0.000 description 3
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 3
- XWKFPIODWVPXLX-UHFFFAOYSA-N 2-methyl-5-methylpyridine Natural products CC1=CC=C(C)N=C1 XWKFPIODWVPXLX-UHFFFAOYSA-N 0.000 description 3
- QCDWFXQBSFUVSP-UHFFFAOYSA-N 2-phenoxyethanol Chemical compound OCCOC1=CC=CC=C1 QCDWFXQBSFUVSP-UHFFFAOYSA-N 0.000 description 3
- WFDIJRYMOXRFFG-UHFFFAOYSA-N Acetic anhydride Chemical compound CC(=O)OC(C)=O WFDIJRYMOXRFFG-UHFFFAOYSA-N 0.000 description 3
- 241001124076 Aphididae Species 0.000 description 3
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 3
- WVDDGKGOMKODPV-UHFFFAOYSA-N Benzyl alcohol Chemical compound OCC1=CC=CC=C1 WVDDGKGOMKODPV-UHFFFAOYSA-N 0.000 description 3
- 241000256593 Brachycaudus schwartzi Species 0.000 description 3
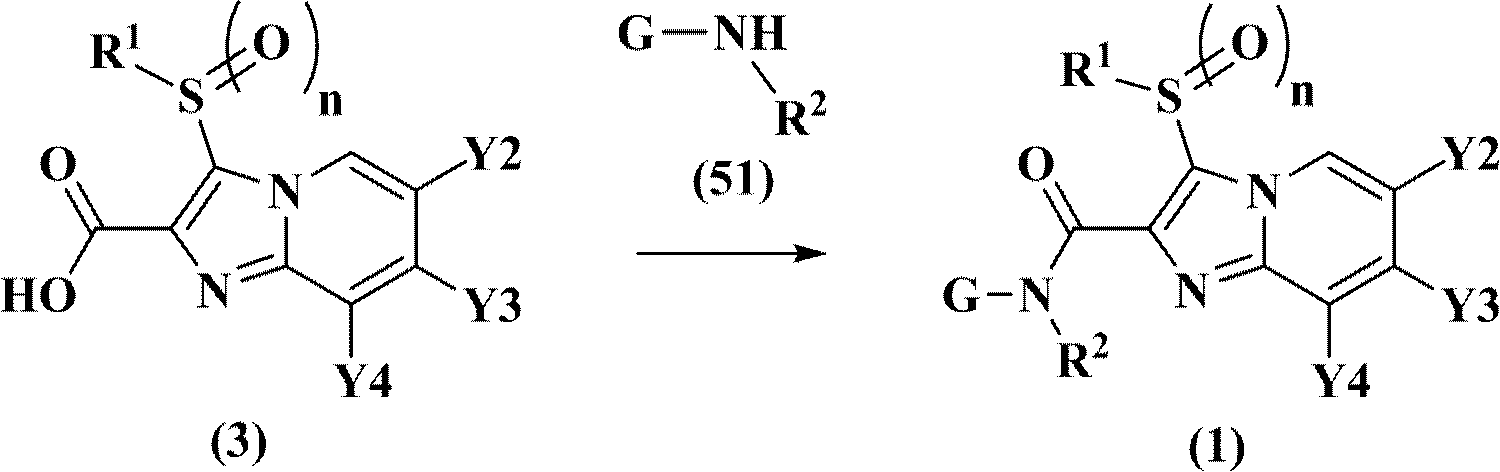
- 0 C*(C12)C1(*)S2(*I)c1c(C(N(*)*)=O)nc2[n]1cc(*)c(*)c2* Chemical compound C*(C12)C1(*)S2(*I)c1c(C(N(*)*)=O)nc2[n]1cc(*)c(*)c2* 0.000 description 3
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 3
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 3
- 241001414720 Cicadellidae Species 0.000 description 3
- 229920000742 Cotton Polymers 0.000 description 3
- 241000238424 Crustacea Species 0.000 description 3
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 3
- 241000257324 Glossina <genus> Species 0.000 description 3
- 241000257303 Hymenoptera Species 0.000 description 3
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 description 3
- 235000007688 Lycopersicon esculentum Nutrition 0.000 description 3
- UFWIBTONFRDIAS-UHFFFAOYSA-N Naphthalene Chemical compound C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 3
- 229910019142 PO4 Inorganic materials 0.000 description 3
- 241000935974 Paralichthys dentatus Species 0.000 description 3
- 241000545593 Scolytinae Species 0.000 description 3
- 240000003768 Solanum lycopersicum Species 0.000 description 3
- 229920002472 Starch Polymers 0.000 description 3
- PPBRXRYQALVLMV-UHFFFAOYSA-N Styrene Chemical compound C=CC1=CC=CC=C1 PPBRXRYQALVLMV-UHFFFAOYSA-N 0.000 description 3
- BABJTMNVJXLAEX-UHFFFAOYSA-N Triamiphos Chemical compound N1=C(N)N(P(=O)(N(C)C)N(C)C)N=C1C1=CC=CC=C1 BABJTMNVJXLAEX-UHFFFAOYSA-N 0.000 description 3
- 238000010521 absorption reaction Methods 0.000 description 3
- 235000011054 acetic acid Nutrition 0.000 description 3
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 3
- 230000002411 adverse Effects 0.000 description 3
- 238000003975 animal breeding Methods 0.000 description 3
- FJDQFPXHSGXQBY-UHFFFAOYSA-L caesium carbonate Chemical compound [Cs+].[Cs+].[O-]C([O-])=O FJDQFPXHSGXQBY-UHFFFAOYSA-L 0.000 description 3
- 229910000024 caesium carbonate Inorganic materials 0.000 description 3
- 239000011575 calcium Substances 0.000 description 3
- 229910052791 calcium Inorganic materials 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 238000011161 development Methods 0.000 description 3
- MTHSVFCYNBDYFN-UHFFFAOYSA-N diethylene glycol Chemical compound OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 description 3
- 229910001873 dinitrogen Inorganic materials 0.000 description 3
- 244000013123 dwarf bean Species 0.000 description 3
- 150000002191 fatty alcohols Chemical class 0.000 description 3
- NYPJDWWKZLNGGM-UHFFFAOYSA-N fenvalerate Aalpha Natural products C=1C=C(Cl)C=CC=1C(C(C)C)C(=O)OC(C#N)C(C=1)=CC=CC=1OC1=CC=CC=C1 NYPJDWWKZLNGGM-UHFFFAOYSA-N 0.000 description 3
- 230000009969 flowable effect Effects 0.000 description 3
- 239000000417 fungicide Substances 0.000 description 3
- 235000011187 glycerol Nutrition 0.000 description 3
- 150000002430 hydrocarbons Chemical group 0.000 description 3
- BEHYAANJUKYBTH-UHFFFAOYSA-N imidazo[1,2-a]pyridine-2-carboxamide Chemical compound C1=CC=CC2=NC(C(=O)N)=CN21 BEHYAANJUKYBTH-UHFFFAOYSA-N 0.000 description 3
- 150000007529 inorganic bases Chemical class 0.000 description 3
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 3
- 125000002816 methylsulfanyl group Chemical group [H]C([H])([H])S[*] 0.000 description 3
- 239000012046 mixed solvent Substances 0.000 description 3
- 239000005645 nematicide Substances 0.000 description 3
- 239000000546 pharmaceutical excipient Substances 0.000 description 3
- WVDDGKGOMKODPV-ZQBYOMGUSA-N phenyl(114C)methanol Chemical compound O[14CH2]C1=CC=CC=C1 WVDDGKGOMKODPV-ZQBYOMGUSA-N 0.000 description 3
- 239000010452 phosphate Substances 0.000 description 3
- 229920000151 polyglycol Polymers 0.000 description 3
- 239000010695 polyglycol Substances 0.000 description 3
- 239000005077 polysulfide Substances 0.000 description 3
- 229920001021 polysulfide Polymers 0.000 description 3
- 150000008117 polysulfides Polymers 0.000 description 3
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 3
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 3
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 3
- 230000001737 promoting effect Effects 0.000 description 3
- 239000003223 protective agent Substances 0.000 description 3
- FBQQHUGEACOBDN-UHFFFAOYSA-N quinomethionate Chemical compound N1=C2SC(=O)SC2=NC2=CC(C)=CC=C21 FBQQHUGEACOBDN-UHFFFAOYSA-N 0.000 description 3
- RMAQACBXLXPBSY-UHFFFAOYSA-N silicic acid Chemical compound O[Si](O)(O)O RMAQACBXLXPBSY-UHFFFAOYSA-N 0.000 description 3
- 239000000377 silicon dioxide Substances 0.000 description 3
- 125000001424 substituent group Chemical group 0.000 description 3
- 235000000346 sugar Nutrition 0.000 description 3
- IMFACGCPASFAPR-UHFFFAOYSA-N tributylamine Chemical compound CCCCN(CCCC)CCCC IMFACGCPASFAPR-UHFFFAOYSA-N 0.000 description 3
- IMNIMPAHZVJRPE-UHFFFAOYSA-N triethylenediamine Chemical compound C1CN2CCN1CC2 IMNIMPAHZVJRPE-UHFFFAOYSA-N 0.000 description 3
- SBNFWQZLDJGRLK-RTWAWAEBSA-N (1R)-trans-phenothrin Chemical compound CC1(C)[C@H](C=C(C)C)[C@H]1C(=O)OCC1=CC=CC(OC=2C=CC=CC=2)=C1 SBNFWQZLDJGRLK-RTWAWAEBSA-N 0.000 description 2
- AGMMRUPNXPWLGF-AATRIKPKSA-N (2,3,5,6-tetrafluoro-4-methylphenyl)methyl 2,2-dimethyl-3-[(e)-prop-1-enyl]cyclopropane-1-carboxylate Chemical compound CC1(C)C(/C=C/C)C1C(=O)OCC1=C(F)C(F)=C(C)C(F)=C1F AGMMRUPNXPWLGF-AATRIKPKSA-N 0.000 description 2
- JNYAEWCLZODPBN-JGWLITMVSA-N (2r,3r,4s)-2-[(1r)-1,2-dihydroxyethyl]oxolane-3,4-diol Polymers OC[C@@H](O)[C@H]1OC[C@H](O)[C@H]1O JNYAEWCLZODPBN-JGWLITMVSA-N 0.000 description 2
- CXNPLSGKWMLZPZ-GIFSMMMISA-N (2r,3r,6s)-3-[[(3s)-3-amino-5-[carbamimidoyl(methyl)amino]pentanoyl]amino]-6-(4-amino-2-oxopyrimidin-1-yl)-3,6-dihydro-2h-pyran-2-carboxylic acid Chemical compound O1[C@@H](C(O)=O)[C@H](NC(=O)C[C@@H](N)CCN(C)C(N)=N)C=C[C@H]1N1C(=O)N=C(N)C=C1 CXNPLSGKWMLZPZ-GIFSMMMISA-N 0.000 description 2
- PGOOBECODWQEAB-UHFFFAOYSA-N (E)-clothianidin Chemical compound [O-][N+](=O)\N=C(/NC)NCC1=CN=C(Cl)S1 PGOOBECODWQEAB-UHFFFAOYSA-N 0.000 description 2
- NWWZPOKUUAIXIW-DHZHZOJOSA-N (E)-thiamethoxam Chemical compound [O-][N+](=O)/N=C/1N(C)COCN\1CC1=CN=C(Cl)S1 NWWZPOKUUAIXIW-DHZHZOJOSA-N 0.000 description 2
- FTTATHOUSOIFOQ-UHFFFAOYSA-N 1,2,3,4,6,7,8,8a-octahydropyrrolo[1,2-a]pyrazine Chemical compound C1NCCN2CCCC21 FTTATHOUSOIFOQ-UHFFFAOYSA-N 0.000 description 2
- ULCWZQJLFZEXCS-UHFFFAOYSA-N 1-[[2-(2,4-dichlorophenyl)-5-(2,2,2-trifluoroethoxy)oxolan-2-yl]methyl]-1,2,4-triazole Chemical compound O1C(OCC(F)(F)F)CCC1(C=1C(=CC(Cl)=CC=1)Cl)CN1N=CN=C1 ULCWZQJLFZEXCS-UHFFFAOYSA-N 0.000 description 2
- ARXJGSRGQADJSQ-UHFFFAOYSA-N 1-methoxypropan-2-ol Chemical compound COCC(C)O ARXJGSRGQADJSQ-UHFFFAOYSA-N 0.000 description 2
- VBICKXHEKHSIBG-UHFFFAOYSA-N 1-monostearoylglycerol Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCC(O)CO VBICKXHEKHSIBG-UHFFFAOYSA-N 0.000 description 2
- JNYAEWCLZODPBN-UHFFFAOYSA-N 2-(1,2-dihydroxyethyl)oxolane-3,4-diol Polymers OCC(O)C1OCC(O)C1O JNYAEWCLZODPBN-UHFFFAOYSA-N 0.000 description 2
- LZILFXHGPBJADI-UHFFFAOYSA-N 2-(2-hydroxypropoxy)propan-1-ol;nonanoic acid Chemical compound CC(O)COC(C)CO.CCCCCCCCC(O)=O LZILFXHGPBJADI-UHFFFAOYSA-N 0.000 description 2
- AWSZRJQNBMEZOI-UHFFFAOYSA-N 2-methoxyethyl 2-(4-tert-butylphenyl)-2-cyano-3-oxo-3-[2-(trifluoromethyl)phenyl]propanoate Chemical compound C=1C=C(C(C)(C)C)C=CC=1C(C#N)(C(=O)OCCOC)C(=O)C1=CC=CC=C1C(F)(F)F AWSZRJQNBMEZOI-UHFFFAOYSA-N 0.000 description 2
- YEKPNMQQSPHKBP-UHFFFAOYSA-N 2-methyl-6-nitrobenzoic anhydride Chemical compound CC1=CC=CC([N+]([O-])=O)=C1C(=O)OC(=O)C1=C(C)C=CC=C1[N+]([O-])=O YEKPNMQQSPHKBP-UHFFFAOYSA-N 0.000 description 2
- SVTBMSDMJJWYQN-UHFFFAOYSA-N 2-methylpentane-2,4-diol Chemical compound CC(O)CC(C)(C)O SVTBMSDMJJWYQN-UHFFFAOYSA-N 0.000 description 2
- WRMNZCZEMHIOCP-UHFFFAOYSA-N 2-phenylethanol Chemical compound OCCC1=CC=CC=C1 WRMNZCZEMHIOCP-UHFFFAOYSA-N 0.000 description 2
- XCGBHLLWJZOLEM-UHFFFAOYSA-N 3-(difluoromethyl)-N-(7-fluoro-1,1,3-trimethyl-2,3-dihydro-1H-inden-4-yl)-1-methyl-1H-pyrazole-4-carboxamide Chemical compound CC1CC(C)(C)C(C(=CC=2)F)=C1C=2NC(=O)C1=CN(C)N=C1C(F)F XCGBHLLWJZOLEM-UHFFFAOYSA-N 0.000 description 2
- YEJRWHAVMIAJKC-UHFFFAOYSA-N 4-Butyrolactone Chemical compound O=C1CCCO1 YEJRWHAVMIAJKC-UHFFFAOYSA-N 0.000 description 2
- HIQIXEFWDLTDED-UHFFFAOYSA-N 4-hydroxy-1-piperidin-4-ylpyrrolidin-2-one Chemical compound O=C1CC(O)CN1C1CCNCC1 HIQIXEFWDLTDED-UHFFFAOYSA-N 0.000 description 2
- PCCSBWNGDMYFCW-UHFFFAOYSA-N 5-methyl-5-(4-phenoxyphenyl)-3-(phenylamino)-1,3-oxazolidine-2,4-dione Chemical compound O=C1C(C)(C=2C=CC(OC=3C=CC=CC=3)=CC=2)OC(=O)N1NC1=CC=CC=C1 PCCSBWNGDMYFCW-UHFFFAOYSA-N 0.000 description 2
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical group [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 2
- 244000215068 Acacia senegal Species 0.000 description 2
- KWOLFJPFCHCOCG-UHFFFAOYSA-N Acetophenone Chemical compound CC(=O)C1=CC=CC=C1 KWOLFJPFCHCOCG-UHFFFAOYSA-N 0.000 description 2
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 2
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonia chloride Chemical compound [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 2
- 241000256186 Anopheles <genus> Species 0.000 description 2
- 241001600408 Aphis gossypii Species 0.000 description 2
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Natural products OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- 244000063299 Bacillus subtilis Species 0.000 description 2
- 235000014469 Bacillus subtilis Nutrition 0.000 description 2
- 239000005735 Benalaxyl-M Substances 0.000 description 2
- 239000005711 Benzoic acid Substances 0.000 description 2
- 241000238662 Blatta orientalis Species 0.000 description 2
- 241000238657 Blattella germanica Species 0.000 description 2
- 241001674044 Blattodea Species 0.000 description 2
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical group [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 2
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 2
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 2
- 241001367803 Chrysodeixis includens Species 0.000 description 2
- 239000005888 Clothianidin Substances 0.000 description 2
- 241000490513 Ctenocephalides canis Species 0.000 description 2
- 241000254171 Curculionidae Species 0.000 description 2
- 239000005655 Cyflumetofen Substances 0.000 description 2
- 239000005946 Cypermethrin Substances 0.000 description 2
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 2
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 2
- RGHNJXZEOKUKBD-SQOUGZDYSA-N D-gluconic acid Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C(O)=O RGHNJXZEOKUKBD-SQOUGZDYSA-N 0.000 description 2
- 241000489972 Diabrotica barberi Species 0.000 description 2
- 241000586568 Diaspidiotus perniciosus Species 0.000 description 2
- MDNWOSOZYLHTCG-UHFFFAOYSA-N Dichlorophen Chemical compound OC1=CC=C(Cl)C=C1CC1=CC(Cl)=CC=C1O MDNWOSOZYLHTCG-UHFFFAOYSA-N 0.000 description 2
- 241001549209 Echidnophaga gallinacea Species 0.000 description 2
- 241000223933 Eimeria bovis Species 0.000 description 2
- 241000196324 Embryophyta Species 0.000 description 2
- 241000995027 Empoasca fabae Species 0.000 description 2
- 241001522296 Erithacus rubecula Species 0.000 description 2
- 239000005772 Famoxadone Substances 0.000 description 2
- 241000242711 Fasciola hepatica Species 0.000 description 2
- 241000282326 Felis catus Species 0.000 description 2
- HMIBKHHNXANVHR-UHFFFAOYSA-N Fenothiocarb Chemical compound CN(C)C(=O)SCCCCOC1=CC=CC=C1 HMIBKHHNXANVHR-UHFFFAOYSA-N 0.000 description 2
- 239000005779 Fenpyrazamine Substances 0.000 description 2
- KRHYYFGTRYWZRS-UHFFFAOYSA-N Fluorane Chemical compound F KRHYYFGTRYWZRS-UHFFFAOYSA-N 0.000 description 2
- 241001466042 Fulgoromorpha Species 0.000 description 2
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 2
- 241000287828 Gallus gallus Species 0.000 description 2
- 241000237858 Gastropoda Species 0.000 description 2
- 108010010803 Gelatin Proteins 0.000 description 2
- 241000224466 Giardia Species 0.000 description 2
- 241001442497 Globodera rostochiensis Species 0.000 description 2
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 description 2
- 229920000084 Gum arabic Polymers 0.000 description 2
- XLYOFNOQVPJJNP-ZSJDYOACSA-N Heavy water Chemical compound [2H]O[2H] XLYOFNOQVPJJNP-ZSJDYOACSA-N 0.000 description 2
- 241000258937 Hemiptera Species 0.000 description 2
- 241001508566 Hypera postica Species 0.000 description 2
- 239000005907 Indoxacarb Substances 0.000 description 2
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 2
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 2
- 241001177117 Lasioderma serricorne Species 0.000 description 2
- 240000000599 Lentinula edodes Species 0.000 description 2
- 235000001715 Lentinula edodes Nutrition 0.000 description 2
- 241000255777 Lepidoptera Species 0.000 description 2
- 241000258916 Leptinotarsa decemlineata Species 0.000 description 2
- 239000005912 Lufenuron Substances 0.000 description 2
- 241000721703 Lymantria dispar Species 0.000 description 2
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 2
- 241000255908 Manduca sexta Species 0.000 description 2
- 241001422926 Mayetiola hordei Species 0.000 description 2
- 241000771994 Melophagus ovinus Species 0.000 description 2
- 239000005808 Metalaxyl-M Substances 0.000 description 2
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 2
- LGDSHSYDSCRFAB-UHFFFAOYSA-N Methyl isothiocyanate Chemical compound CN=C=S LGDSHSYDSCRFAB-UHFFFAOYSA-N 0.000 description 2
- 241000952627 Monomorium pharaonis Species 0.000 description 2
- 241000257159 Musca domestica Species 0.000 description 2
- 241000721621 Myzus persicae Species 0.000 description 2
- LQZMLBORDGWNPD-UHFFFAOYSA-N N-iodosuccinimide Chemical compound IN1C(=O)CCC1=O LQZMLBORDGWNPD-UHFFFAOYSA-N 0.000 description 2
- 241000543819 Oestrus ovis Species 0.000 description 2
- 241001012098 Omiodes indicata Species 0.000 description 2
- 241000963706 Onychiurus Species 0.000 description 2
- 241000176318 Ornithonyssus bacoti Species 0.000 description 2
- 241000273374 Ornithonyssus sylviarum Species 0.000 description 2
- 239000005950 Oxamyl Substances 0.000 description 2
- 241000320508 Pentatomidae Species 0.000 description 2
- 241001177886 Penthaleus Species 0.000 description 2
- KFSLWBXXFJQRDL-UHFFFAOYSA-N Peracetic acid Chemical compound CC(=O)OO KFSLWBXXFJQRDL-UHFFFAOYSA-N 0.000 description 2
- HEMINMLPKZELPP-UHFFFAOYSA-N Phosdiphen Chemical compound C=1C=C(Cl)C=C(Cl)C=1OP(=O)(OCC)OC1=CC=C(Cl)C=C1Cl HEMINMLPKZELPP-UHFFFAOYSA-N 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- 241000691880 Planococcus citri Species 0.000 description 2
- 239000004372 Polyvinyl alcohol Substances 0.000 description 2
- 241000254101 Popillia japonica Species 0.000 description 2
- WCUXLLCKKVVCTQ-UHFFFAOYSA-M Potassium chloride Chemical compound [Cl-].[K+] WCUXLLCKKVVCTQ-UHFFFAOYSA-M 0.000 description 2
- 239000005820 Prochloraz Substances 0.000 description 2
- 241000718000 Pulex irritans Species 0.000 description 2
- 235000014443 Pyrus communis Nutrition 0.000 description 2
- 241001136903 Rhagoletis pomonella Species 0.000 description 2
- 241000555745 Sciuridae Species 0.000 description 2
- 241000258242 Siphonaptera Species 0.000 description 2
- 241000254179 Sitophilus granarius Species 0.000 description 2
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 2
- PXIPVTKHYLBLMZ-UHFFFAOYSA-N Sodium azide Chemical compound [Na+].[N-]=[N+]=[N-] PXIPVTKHYLBLMZ-UHFFFAOYSA-N 0.000 description 2
- 241001521235 Spodoptera eridania Species 0.000 description 2
- 241000256250 Spodoptera littoralis Species 0.000 description 2
- 241001494115 Stomoxys calcitrans Species 0.000 description 2
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 2
- 229930006000 Sucrose Natural products 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- 241000255626 Tabanus <genus> Species 0.000 description 2
- 241000270666 Testudines Species 0.000 description 2
- 241000488589 Tetranychus kanzawai Species 0.000 description 2
- 241000843170 Togo hemipterus Species 0.000 description 2
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 2
- 241000261593 Tyrophagus neiswanderi Species 0.000 description 2
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 2
- FMPFURNXXAKYNE-UHFFFAOYSA-N [2-ethyl-3,7-dimethyl-6-[4-(trifluoromethoxy)phenoxy]quinolin-4-yl] methyl carbonate Chemical compound C1=C2C(OC(=O)OC)=C(C)C(CC)=NC2=CC(C)=C1OC1=CC=C(OC(F)(F)F)C=C1 FMPFURNXXAKYNE-UHFFFAOYSA-N 0.000 description 2
- 235000010489 acacia gum Nutrition 0.000 description 2
- 239000000205 acacia gum Substances 0.000 description 2
- 239000013543 active substance Substances 0.000 description 2
- WNLRTRBMVRJNCN-UHFFFAOYSA-N adipic acid Chemical compound OC(=O)CCCCC(O)=O WNLRTRBMVRJNCN-UHFFFAOYSA-N 0.000 description 2
- 235000010443 alginic acid Nutrition 0.000 description 2
- 229920000615 alginic acid Polymers 0.000 description 2
- 125000003545 alkoxy group Chemical group 0.000 description 2
- 125000004448 alkyl carbonyl group Chemical group 0.000 description 2
- FPIPGXGPPPQFEQ-OVSJKPMPSA-N all-trans-retinol Chemical compound OC\C=C(/C)\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C FPIPGXGPPPQFEQ-OVSJKPMPSA-N 0.000 description 2
- 235000001014 amino acid Nutrition 0.000 description 2
- 150000001413 amino acids Chemical class 0.000 description 2
- 239000002280 amphoteric surfactant Substances 0.000 description 2
- 235000010323 ascorbic acid Nutrition 0.000 description 2
- 239000011668 ascorbic acid Substances 0.000 description 2
- 229960005070 ascorbic acid Drugs 0.000 description 2
- 239000012752 auxiliary agent Substances 0.000 description 2
- 229910052788 barium Inorganic materials 0.000 description 2
- DSAJWYNOEDNPEQ-UHFFFAOYSA-N barium atom Chemical compound [Ba] DSAJWYNOEDNPEQ-UHFFFAOYSA-N 0.000 description 2
- 238000003287 bathing Methods 0.000 description 2
- CJPQIRJHIZUAQP-MRXNPFEDSA-N benalaxyl-M Chemical compound CC=1C=CC=C(C)C=1N([C@H](C)C(=O)OC)C(=O)CC1=CC=CC=C1 CJPQIRJHIZUAQP-MRXNPFEDSA-N 0.000 description 2
- 239000000440 bentonite Substances 0.000 description 2
- 229910000278 bentonite Inorganic materials 0.000 description 2
- SVPXDRXYRYOSEX-UHFFFAOYSA-N bentoquatam Chemical compound O.O=[Si]=O.O=[Al]O[Al]=O SVPXDRXYRYOSEX-UHFFFAOYSA-N 0.000 description 2
- 235000010233 benzoic acid Nutrition 0.000 description 2
- 229960004365 benzoic acid Drugs 0.000 description 2
- FFBHFFJDDLITSX-UHFFFAOYSA-N benzyl N-[2-hydroxy-4-(3-oxomorpholin-4-yl)phenyl]carbamate Chemical compound OC1=C(NC(=O)OCC2=CC=CC=C2)C=CC(=C1)N1CCOCC1=O FFBHFFJDDLITSX-UHFFFAOYSA-N 0.000 description 2
- SESFRYSPDFLNCH-UHFFFAOYSA-N benzyl benzoate Chemical compound C=1C=CC=CC=1C(=O)OCC1=CC=CC=C1 SESFRYSPDFLNCH-UHFFFAOYSA-N 0.000 description 2
- OMFRMAHOUUJSGP-IRHGGOMRSA-N bifenthrin Chemical compound C1=CC=C(C=2C=CC=CC=2)C(C)=C1COC(=O)[C@@H]1[C@H](\C=C(/Cl)C(F)(F)F)C1(C)C OMFRMAHOUUJSGP-IRHGGOMRSA-N 0.000 description 2
- LLEMOWNGBBNAJR-UHFFFAOYSA-N biphenyl-2-ol Chemical compound OC1=CC=CC=C1C1=CC=CC=C1 LLEMOWNGBBNAJR-UHFFFAOYSA-N 0.000 description 2
- CXNPLSGKWMLZPZ-UHFFFAOYSA-N blasticidin-S Natural products O1C(C(O)=O)C(NC(=O)CC(N)CCN(C)C(N)=N)C=CC1N1C(=O)N=C(N)C=C1 CXNPLSGKWMLZPZ-UHFFFAOYSA-N 0.000 description 2
- GZUXJHMPEANEGY-UHFFFAOYSA-N bromomethane Chemical compound BrC GZUXJHMPEANEGY-UHFFFAOYSA-N 0.000 description 2
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical compound O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 description 2
- DUEPRVBVGDRKAG-UHFFFAOYSA-N carbofuran Chemical compound CNC(=O)OC1=CC=CC2=C1OC(C)(C)C2 DUEPRVBVGDRKAG-UHFFFAOYSA-N 0.000 description 2
- 239000001768 carboxy methyl cellulose Substances 0.000 description 2
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 2
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 2
- ULDHMXUKGWMISQ-UHFFFAOYSA-N carvone Chemical compound CC(=C)C1CC=C(C)C(=O)C1 ULDHMXUKGWMISQ-UHFFFAOYSA-N 0.000 description 2
- 239000003093 cationic surfactant Substances 0.000 description 2
- CWFOCCVIPCEQCK-UHFFFAOYSA-N chlorfenapyr Chemical compound BrC1=C(C(F)(F)F)N(COCC)C(C=2C=CC(Cl)=CC=2)=C1C#N CWFOCCVIPCEQCK-UHFFFAOYSA-N 0.000 description 2
- 229910052801 chlorine Inorganic materials 0.000 description 2
- 125000001309 chloro group Chemical group Cl* 0.000 description 2
- 239000003086 colorant Substances 0.000 description 2
- 229920001577 copolymer Polymers 0.000 description 2
- 229940120693 copper naphthenate Drugs 0.000 description 2
- 229910000365 copper sulfate Inorganic materials 0.000 description 2
- ARUVKPQLZAKDPS-UHFFFAOYSA-L copper(II) sulfate Chemical compound [Cu+2].[O-][S+2]([O-])([O-])[O-] ARUVKPQLZAKDPS-UHFFFAOYSA-L 0.000 description 2
- SEVNKWFHTNVOLD-UHFFFAOYSA-L copper;3-(4-ethylcyclohexyl)propanoate;3-(3-ethylcyclopentyl)propanoate Chemical compound [Cu+2].CCC1CCC(CCC([O-])=O)C1.CCC1CCC(CCC([O-])=O)CC1 SEVNKWFHTNVOLD-UHFFFAOYSA-L 0.000 description 2
- 125000004122 cyclic group Chemical group 0.000 description 2
- JHIVVAPYMSGYDF-UHFFFAOYSA-N cyclohexanone Chemical compound O=C1CCCCC1 JHIVVAPYMSGYDF-UHFFFAOYSA-N 0.000 description 2
- YPHMISFOHDHNIV-FSZOTQKASA-N cycloheximide Chemical compound C1[C@@H](C)C[C@H](C)C(=O)[C@@H]1[C@H](O)CC1CC(=O)NC(=O)C1 YPHMISFOHDHNIV-FSZOTQKASA-N 0.000 description 2
- KAATUXNTWXVJKI-UHFFFAOYSA-N cypermethrin Chemical compound CC1(C)C(C=C(Cl)Cl)C1C(=O)OC(C#N)C1=CC=CC(OC=2C=CC=CC=2)=C1 KAATUXNTWXVJKI-UHFFFAOYSA-N 0.000 description 2
- 229960005424 cypermethrin Drugs 0.000 description 2
- 239000012973 diazabicyclooctane Substances 0.000 description 2
- DOIRQSBPFJWKBE-UHFFFAOYSA-N dibutyl phthalate Chemical compound CCCCOC(=O)C1=CC=CC=C1C(=O)OCCCC DOIRQSBPFJWKBE-UHFFFAOYSA-N 0.000 description 2
- 229960003887 dichlorophen Drugs 0.000 description 2
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N diphenyl Chemical compound C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 description 2
- DMBHHRLKUKUOEG-UHFFFAOYSA-N diphenylamine Chemical compound C=1C=CC=CC=1NC1=CC=CC=C1 DMBHHRLKUKUOEG-UHFFFAOYSA-N 0.000 description 2
- 239000007884 disintegrant Substances 0.000 description 2
- 239000003995 emulsifying agent Substances 0.000 description 2
- 230000007613 environmental effect Effects 0.000 description 2
- LZCLXQDLBQLTDK-UHFFFAOYSA-N ethyl 2-hydroxypropanoate Chemical compound CCOC(=O)C(C)O LZCLXQDLBQLTDK-UHFFFAOYSA-N 0.000 description 2
- MVLVMROFTAUDAG-UHFFFAOYSA-N ethyl octadecanoate Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCC MVLVMROFTAUDAG-UHFFFAOYSA-N 0.000 description 2
- FKRCODPIKNYEAC-UHFFFAOYSA-N ethyl propionate Chemical compound CCOC(=O)CC FKRCODPIKNYEAC-UHFFFAOYSA-N 0.000 description 2
- 238000001125 extrusion Methods 0.000 description 2
- 239000003925 fat Substances 0.000 description 2
- 235000019197 fats Nutrition 0.000 description 2
- JFSPBVWPKOEZCB-UHFFFAOYSA-N fenfuram Chemical compound O1C=CC(C(=O)NC=2C=CC=CC=2)=C1C JFSPBVWPKOEZCB-UHFFFAOYSA-N 0.000 description 2
- XQUXKZZNEFRCAW-UHFFFAOYSA-N fenpropathrin Chemical compound CC1(C)C(C)(C)C1C(=O)OC(C#N)C1=CC=CC(OC=2C=CC=CC=2)=C1 XQUXKZZNEFRCAW-UHFFFAOYSA-N 0.000 description 2
- UTOHZQYBSYOOGC-UHFFFAOYSA-N fenpyrazamine Chemical compound O=C1N(C(C)C)N(C(=O)SCC=C)C(N)=C1C1=CC=CC=C1C UTOHZQYBSYOOGC-UHFFFAOYSA-N 0.000 description 2
- XDNBJTQLKCIJBV-UHFFFAOYSA-N fensulfothion Chemical compound CCOP(=S)(OCC)OC1=CC=C(S(C)=O)C=C1 XDNBJTQLKCIJBV-UHFFFAOYSA-N 0.000 description 2
- 239000003337 fertilizer Substances 0.000 description 2
- 239000000706 filtrate Substances 0.000 description 2
- 235000013312 flour Nutrition 0.000 description 2
- MXWAGQASUDSFBG-RVDMUPIBSA-N fluacrypyrim Chemical compound CO\C=C(\C(=O)OC)C1=CC=CC=C1COC1=CC(C(F)(F)F)=NC(OC(C)C)=N1 MXWAGQASUDSFBG-RVDMUPIBSA-N 0.000 description 2
- 229910052731 fluorine Inorganic materials 0.000 description 2
- 125000004216 fluoromethyl group Chemical group [H]C([H])(F)* 0.000 description 2
- 230000000855 fungicidal effect Effects 0.000 description 2
- 239000008273 gelatin Substances 0.000 description 2
- 229920000159 gelatin Polymers 0.000 description 2
- 235000019322 gelatine Nutrition 0.000 description 2
- 235000011852 gelatine desserts Nutrition 0.000 description 2
- 235000013922 glutamic acid Nutrition 0.000 description 2
- 239000004220 glutamic acid Substances 0.000 description 2
- KWIUHFFTVRNATP-UHFFFAOYSA-N glycine betaine Chemical compound C[N+](C)(C)CC([O-])=O KWIUHFFTVRNATP-UHFFFAOYSA-N 0.000 description 2
- 235000021331 green beans Nutrition 0.000 description 2
- 230000002140 halogenating effect Effects 0.000 description 2
- 239000004009 herbicide Substances 0.000 description 2
- 125000000623 heterocyclic group Chemical group 0.000 description 2
- 238000003898 horticulture Methods 0.000 description 2
- 229910052739 hydrogen Inorganic materials 0.000 description 2
- 230000002209 hydrophobic effect Effects 0.000 description 2
- HICUREFSAIZXFQ-JOWPUVSESA-N i9z29i000j Chemical compound C1C[C@H](C)[C@@H](CC)O[C@@]21O[C@H](C\C=C(C)\[C@H](OC(=O)C(=N/OC)\C=1C=CC=CC=1)[C@@H](C)\C=C\C=C/1[C@]3([C@H](C(=O)O4)C=C(C)[C@@H](O)[C@H]3OC\1)O)C[C@H]4C2 HICUREFSAIZXFQ-JOWPUVSESA-N 0.000 description 2
- 238000007654 immersion Methods 0.000 description 2
- VBCVPMMZEGZULK-NRFANRHFSA-N indoxacarb Chemical compound C([C@@]1(OC2)C(=O)OC)C3=CC(Cl)=CC=C3C1=NN2C(=O)N(C(=O)OC)C1=CC=C(OC(F)(F)F)C=C1 VBCVPMMZEGZULK-NRFANRHFSA-N 0.000 description 2
- 229910052500 inorganic mineral Inorganic materials 0.000 description 2
- 229910052740 iodine Inorganic materials 0.000 description 2
- QBSJMKIUCUGGNG-UHFFFAOYSA-N isoprocarb Chemical compound CNC(=O)OC1=CC=CC=C1C(C)C QBSJMKIUCUGGNG-UHFFFAOYSA-N 0.000 description 2
- SDMSCIWHRZJSRN-UHFFFAOYSA-N isoxathion Chemical compound O1N=C(OP(=S)(OCC)OCC)C=C1C1=CC=CC=C1 SDMSCIWHRZJSRN-UHFFFAOYSA-N 0.000 description 2
- 229910052622 kaolinite Inorganic materials 0.000 description 2
- TYQCGQRIZGCHNB-JLAZNSOCSA-N l-ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(O)=C(O)C1=O TYQCGQRIZGCHNB-JLAZNSOCSA-N 0.000 description 2
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 2
- 239000008101 lactose Substances 0.000 description 2
- 244000144972 livestock Species 0.000 description 2
- 239000006210 lotion Substances 0.000 description 2
- 239000000314 lubricant Substances 0.000 description 2
- 229960000521 lufenuron Drugs 0.000 description 2
- PWPJGUXAGUPAHP-UHFFFAOYSA-N lufenuron Chemical compound C1=C(Cl)C(OC(F)(F)C(C(F)(F)F)F)=CC(Cl)=C1NC(=O)NC(=O)C1=C(F)C=CC=C1F PWPJGUXAGUPAHP-UHFFFAOYSA-N 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- ZQEIXNIJLIKNTD-GFCCVEGCSA-N metalaxyl-M Chemical compound COCC(=O)N([C@H](C)C(=O)OC)C1=C(C)C=CC=C1C ZQEIXNIJLIKNTD-GFCCVEGCSA-N 0.000 description 2
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 2
- 229920000609 methyl cellulose Polymers 0.000 description 2
- 239000001923 methylcellulose Substances 0.000 description 2
- 235000010981 methylcellulose Nutrition 0.000 description 2
- 235000010755 mineral Nutrition 0.000 description 2
- 239000011707 mineral Substances 0.000 description 2
- 231100000989 no adverse effect Toxicity 0.000 description 2
- 230000035764 nutrition Effects 0.000 description 2
- 235000016709 nutrition Nutrition 0.000 description 2
- TVMXDCGIABBOFY-UHFFFAOYSA-N octane Chemical compound CCCCCCCC TVMXDCGIABBOFY-UHFFFAOYSA-N 0.000 description 2
- 150000007530 organic bases Chemical class 0.000 description 2
- 239000003960 organic solvent Substances 0.000 description 2
- KZAUOCCYDRDERY-UHFFFAOYSA-N oxamyl Chemical compound CNC(=O)ON=C(SC)C(=O)N(C)C KZAUOCCYDRDERY-UHFFFAOYSA-N 0.000 description 2
- PMCVMORKVPSKHZ-UHFFFAOYSA-N oxydemeton-methyl Chemical compound CCS(=O)CCSP(=O)(OC)OC PMCVMORKVPSKHZ-UHFFFAOYSA-N 0.000 description 2
- IZUPBVBPLAPZRR-UHFFFAOYSA-N pentachlorophenol Chemical compound OC1=C(Cl)C(Cl)=C(Cl)C(Cl)=C1Cl IZUPBVBPLAPZRR-UHFFFAOYSA-N 0.000 description 2
- VLTRZXGMWDSKGL-UHFFFAOYSA-N perchloric acid Chemical compound OCl(=O)(=O)=O VLTRZXGMWDSKGL-UHFFFAOYSA-N 0.000 description 2
- 230000002688 persistence Effects 0.000 description 2
- 229960003536 phenothrin Drugs 0.000 description 2
- 229960005323 phenoxyethanol Drugs 0.000 description 2
- SIOXPEMLGUPBBT-UHFFFAOYSA-N picolinic acid Chemical compound OC(=O)C1=CC=CC=N1 SIOXPEMLGUPBBT-UHFFFAOYSA-N 0.000 description 2
- 229920000058 polyacrylate Polymers 0.000 description 2
- JPFWJDMDPLEUBD-ITJAGOAWSA-N polyoxorim Polymers O[C@@H]1[C@H](O)[C@@H]([C@H](NC(=O)[C@H]([C@H](O)[C@@H](O)COC(N)=O)N)C(O)=O)O[C@H]1N1C(=O)NC(=O)C(C(O)=O)=C1 JPFWJDMDPLEUBD-ITJAGOAWSA-N 0.000 description 2
- 229920001451 polypropylene glycol Polymers 0.000 description 2
- 229920002451 polyvinyl alcohol Polymers 0.000 description 2
- 235000019422 polyvinyl alcohol Nutrition 0.000 description 2
- 239000004540 pour-on Substances 0.000 description 2
- TVLSRXXIMLFWEO-UHFFFAOYSA-N prochloraz Chemical compound C1=CN=CN1C(=O)N(CCC)CCOC1=C(Cl)C=C(Cl)C=C1Cl TVLSRXXIMLFWEO-UHFFFAOYSA-N 0.000 description 2
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 235000018102 proteins Nutrition 0.000 description 2
- 108090000623 proteins and genes Chemical class 0.000 description 2
- 102000004169 proteins and genes Human genes 0.000 description 2
- 238000010298 pulverizing process Methods 0.000 description 2
- 238000000746 purification Methods 0.000 description 2
- 239000002728 pyrethroid Substances 0.000 description 2
- 229910052903 pyrophyllite Inorganic materials 0.000 description 2
- HNJBEVLQSNELDL-UHFFFAOYSA-N pyrrolidin-2-one Chemical compound O=C1CCCN1 HNJBEVLQSNELDL-UHFFFAOYSA-N 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 229920002545 silicone oil Polymers 0.000 description 2
- QDRKDTQENPPHOJ-UHFFFAOYSA-N sodium ethoxide Chemical compound [Na+].CC[O-] QDRKDTQENPPHOJ-UHFFFAOYSA-N 0.000 description 2
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical class O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 2
- JNYAEWCLZODPBN-CTQIIAAMSA-N sorbitan Polymers OCC(O)C1OCC(O)[C@@H]1O JNYAEWCLZODPBN-CTQIIAAMSA-N 0.000 description 2
- 239000000600 sorbitol Substances 0.000 description 2
- 239000004544 spot-on Substances 0.000 description 2
- 235000019698 starch Nutrition 0.000 description 2
- UCSJYZPVAKXKNQ-HZYVHMACSA-N streptomycin Chemical compound CN[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O[C@H]1O[C@@H]1[C@](C=O)(O)[C@H](C)O[C@H]1O[C@@H]1[C@@H](NC(N)=N)[C@H](O)[C@@H](NC(N)=N)[C@H](O)[C@H]1O UCSJYZPVAKXKNQ-HZYVHMACSA-N 0.000 description 2
- 238000006467 substitution reaction Methods 0.000 description 2
- 239000005720 sucrose Substances 0.000 description 2
- 150000008163 sugars Chemical class 0.000 description 2
- 125000000446 sulfanediyl group Chemical group *S* 0.000 description 2
- 239000004546 suspension concentrate Substances 0.000 description 2
- 239000000454 talc Substances 0.000 description 2
- 229910052623 talc Inorganic materials 0.000 description 2
- XLNZEKHULJKQBA-UHFFFAOYSA-N terbufos Chemical compound CCOP(=S)(OCC)SCSC(C)(C)C XLNZEKHULJKQBA-UHFFFAOYSA-N 0.000 description 2
- 229960005199 tetramethrin Drugs 0.000 description 2
- 239000004308 thiabendazole Substances 0.000 description 2
- 235000010296 thiabendazole Nutrition 0.000 description 2
- WJCNZQLZVWNLKY-UHFFFAOYSA-N thiabendazole Chemical compound S1C=NC(C=2NC3=CC=CC=C3N=2)=C1 WJCNZQLZVWNLKY-UHFFFAOYSA-N 0.000 description 2
- 229960004546 thiabendazole Drugs 0.000 description 2
- 239000010409 thin film Substances 0.000 description 2
- FYSNRJHAOHDILO-UHFFFAOYSA-N thionyl chloride Chemical compound ClS(Cl)=O FYSNRJHAOHDILO-UHFFFAOYSA-N 0.000 description 2
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- DDVNRFNDOPPVQJ-HQJQHLMTSA-N transfluthrin Chemical compound CC1(C)[C@H](C=C(Cl)Cl)[C@H]1C(=O)OCC1=C(F)C(F)=CC(F)=C1F DDVNRFNDOPPVQJ-HQJQHLMTSA-N 0.000 description 2
- NKNFWVNSBIXGLL-UHFFFAOYSA-N triazamate Chemical compound CCOC(=O)CSC1=NC(C(C)(C)C)=NN1C(=O)N(C)C NKNFWVNSBIXGLL-UHFFFAOYSA-N 0.000 description 2
- CXNIUSPIQKWYAI-UHFFFAOYSA-N xantphos Chemical compound C=12OC3=C(P(C=4C=CC=CC=4)C=4C=CC=CC=4)C=CC=C3C(C)(C)C2=CC=CC=1P(C=1C=CC=CC=1)C1=CC=CC=C1 CXNIUSPIQKWYAI-UHFFFAOYSA-N 0.000 description 2
- ZCVAOQKBXKSDMS-PVAVHDDUSA-N (+)-trans-(S)-allethrin Chemical compound CC1(C)[C@H](C=C(C)C)[C@H]1C(=O)O[C@@H]1C(C)=C(CC=C)C(=O)C1 ZCVAOQKBXKSDMS-PVAVHDDUSA-N 0.000 description 1
- CXBMCYHAMVGWJQ-CABCVRRESA-N (1,3-dioxo-4,5,6,7-tetrahydroisoindol-2-yl)methyl (1r,3r)-2,2-dimethyl-3-(2-methylprop-1-enyl)cyclopropane-1-carboxylate Chemical compound CC1(C)[C@H](C=C(C)C)[C@H]1C(=O)OCN1C(=O)C(CCCC2)=C2C1=O CXBMCYHAMVGWJQ-CABCVRRESA-N 0.000 description 1
- FJDPATXIBIBRIM-QFMSAKRMSA-N (1R)-trans-cyphenothrin Chemical compound CC1(C)[C@H](C=C(C)C)[C@H]1C(=O)OC(C#N)C1=CC=CC(OC=2C=CC=CC=2)=C1 FJDPATXIBIBRIM-QFMSAKRMSA-N 0.000 description 1
- ZXQYGBMAQZUVMI-RDDWSQKMSA-N (1S)-cis-(alphaR)-cyhalothrin Chemical compound CC1(C)[C@H](\C=C(/Cl)C(F)(F)F)[C@@H]1C(=O)O[C@@H](C#N)C1=CC=CC(OC=2C=CC=CC=2)=C1 ZXQYGBMAQZUVMI-RDDWSQKMSA-N 0.000 description 1
- XERJKGMBORTKEO-VZUCSPMQSA-N (1e)-2-(ethylcarbamoylamino)-n-methoxy-2-oxoethanimidoyl cyanide Chemical compound CCNC(=O)NC(=O)C(\C#N)=N\OC XERJKGMBORTKEO-VZUCSPMQSA-N 0.000 description 1
- MYUPFXPCYUISAG-UHFFFAOYSA-N (2,4-dichlorophenyl)(phenyl)pyrimidin-5-ylmethanol Chemical compound C=1N=CN=CC=1C(C=1C(=CC(Cl)=CC=1)Cl)(O)C1=CC=CC=C1 MYUPFXPCYUISAG-UHFFFAOYSA-N 0.000 description 1
- PTKVPQXEKZOPNA-UHFFFAOYSA-N (2,4-dinitro-6-octan-2-ylphenyl) methylsulfanylformate Chemical compound CCCCCCC(C)C1=CC([N+]([O-])=O)=CC([N+]([O-])=O)=C1OC(=O)SC PTKVPQXEKZOPNA-UHFFFAOYSA-N 0.000 description 1
- YHVJMJFNSRLYIC-WEVVVXLNSA-N (2,6-dinitro-4-octan-2-ylphenyl) (e)-but-2-enoate Chemical compound CCCCCCC(C)C1=CC([N+]([O-])=O)=C(OC(=O)\C=C\C)C([N+]([O-])=O)=C1 YHVJMJFNSRLYIC-WEVVVXLNSA-N 0.000 description 1
- NHOWDZOIZKMVAI-UHFFFAOYSA-N (2-chlorophenyl)(4-chlorophenyl)pyrimidin-5-ylmethanol Chemical compound C=1N=CN=CC=1C(C=1C(=CC=CC=1)Cl)(O)C1=CC=C(Cl)C=C1 NHOWDZOIZKMVAI-UHFFFAOYSA-N 0.000 description 1
- SAPGTCDSBGMXCD-UHFFFAOYSA-N (2-chlorophenyl)-(4-fluorophenyl)-pyrimidin-5-ylmethanol Chemical compound C=1N=CN=CC=1C(C=1C(=CC=CC=1)Cl)(O)C1=CC=C(F)C=C1 SAPGTCDSBGMXCD-UHFFFAOYSA-N 0.000 description 1
- IXNUTQCZWAHNPN-UHFFFAOYSA-N (2-tert-butyl-4,6-dinitrophenyl) ethyl carbonate Chemical compound CCOC(=O)OC1=C([N+]([O-])=O)C=C([N+]([O-])=O)C=C1C(C)(C)C IXNUTQCZWAHNPN-UHFFFAOYSA-N 0.000 description 1
- QBYIENPQHBMVBV-HFEGYEGKSA-N (2R)-2-hydroxy-2-phenylacetic acid Chemical compound O[C@@H](C(O)=O)c1ccccc1.O[C@@H](C(O)=O)c1ccccc1 QBYIENPQHBMVBV-HFEGYEGKSA-N 0.000 description 1
- ZMYFCFLJBGAQRS-IRXDYDNUSA-N (2R,3S)-epoxiconazole Chemical compound C1=CC(F)=CC=C1[C@@]1(CN2N=CN=C2)[C@H](C=2C(=CC=CC=2)Cl)O1 ZMYFCFLJBGAQRS-IRXDYDNUSA-N 0.000 description 1
- RYAUSSKQMZRMAI-ALOPSCKCSA-N (2S,6R)-4-[3-(4-tert-butylphenyl)-2-methylpropyl]-2,6-dimethylmorpholine Chemical compound C=1C=C(C(C)(C)C)C=CC=1CC(C)CN1C[C@H](C)O[C@H](C)C1 RYAUSSKQMZRMAI-ALOPSCKCSA-N 0.000 description 1
- URDNHJIVMYZFRT-KGLIPLIRSA-N (2r,3r)-1-(2,4-dichlorophenyl)-4,4-dimethyl-2-(1,2,4-triazol-1-yl)pentan-3-ol Chemical compound C([C@H]([C@H](O)C(C)(C)C)N1N=CN=C1)C1=CC=C(Cl)C=C1Cl URDNHJIVMYZFRT-KGLIPLIRSA-N 0.000 description 1
- DBTMGCOVALSLOR-DEVYUCJPSA-N (2s,3r,4s,5r,6r)-4-[(2s,3r,4s,5r,6r)-3,5-dihydroxy-6-(hydroxymethyl)-4-[(2s,3r,4s,5s,6r)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxyoxan-2-yl]oxy-6-(hydroxymethyl)oxane-2,3,5-triol Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](O)[C@H](O[C@H]2[C@@H]([C@@H](CO)O[C@H](O)[C@@H]2O)O)O[C@H](CO)[C@H]1O DBTMGCOVALSLOR-DEVYUCJPSA-N 0.000 description 1
- LDVVMCZRFWMZSG-OLQVQODUSA-N (3ar,7as)-2-(trichloromethylsulfanyl)-3a,4,7,7a-tetrahydroisoindole-1,3-dione Chemical compound C1C=CC[C@H]2C(=O)N(SC(Cl)(Cl)Cl)C(=O)[C@H]21 LDVVMCZRFWMZSG-OLQVQODUSA-N 0.000 description 1
- XUNYDVLIZWUPAW-UHFFFAOYSA-N (4-chlorophenyl) n-(4-methylphenyl)sulfonylcarbamate Chemical compound C1=CC(C)=CC=C1S(=O)(=O)NC(=O)OC1=CC=C(Cl)C=C1 XUNYDVLIZWUPAW-UHFFFAOYSA-N 0.000 description 1
- UGJQNSILMBYITH-UHFFFAOYSA-N (4-hydroxyphenyl)iminourea Chemical compound NC(=O)N=Nc1ccc(O)cc1 UGJQNSILMBYITH-UHFFFAOYSA-N 0.000 description 1
- PWWPULQZEAPTTB-UHFFFAOYSA-N (4-phenoxyphenyl)methyl 2-amino-6-methylpyridine-3-carboxylate Chemical compound NC1=NC(C)=CC=C1C(=O)OCC(C=C1)=CC=C1OC1=CC=CC=C1 PWWPULQZEAPTTB-UHFFFAOYSA-N 0.000 description 1
- PPDBOQMNKNNODG-NTEUORMPSA-N (5E)-5-(4-chlorobenzylidene)-2,2-dimethyl-1-(1,2,4-triazol-1-ylmethyl)cyclopentanol Chemical compound C1=NC=NN1CC1(O)C(C)(C)CC\C1=C/C1=CC=C(Cl)C=C1 PPDBOQMNKNNODG-NTEUORMPSA-N 0.000 description 1
- ALSTYHKOOCGGFT-KTKRTIGZSA-N (9Z)-octadecen-1-ol Chemical compound CCCCCCCC\C=C/CCCCCCCCO ALSTYHKOOCGGFT-KTKRTIGZSA-N 0.000 description 1
- BKBSMMUEEAWFRX-NBVRZTHBSA-N (E)-flumorph Chemical compound C1=C(OC)C(OC)=CC=C1C(\C=1C=CC(F)=CC=1)=C\C(=O)N1CCOCC1 BKBSMMUEEAWFRX-NBVRZTHBSA-N 0.000 description 1
- CFRPSFYHXJZSBI-DHZHZOJOSA-N (E)-nitenpyram Chemical compound [O-][N+](=O)/C=C(\NC)N(CC)CC1=CC=C(Cl)N=C1 CFRPSFYHXJZSBI-DHZHZOJOSA-N 0.000 description 1
- IQVNEKKDSLOHHK-FNCQTZNRSA-N (E,E)-hydramethylnon Chemical compound N1CC(C)(C)CNC1=NN=C(/C=C/C=1C=CC(=CC=1)C(F)(F)F)\C=C\C1=CC=C(C(F)(F)F)C=C1 IQVNEKKDSLOHHK-FNCQTZNRSA-N 0.000 description 1
- BJEPYKJPYRNKOW-REOHCLBHSA-N (S)-malic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O BJEPYKJPYRNKOW-REOHCLBHSA-N 0.000 description 1
- XGWIJUOSCAQSSV-XHDPSFHLSA-N (S,S)-hexythiazox Chemical compound S([C@H]([C@@H]1C)C=2C=CC(Cl)=CC=2)C(=O)N1C(=O)NC1CCCCC1 XGWIJUOSCAQSSV-XHDPSFHLSA-N 0.000 description 1
- ZFHGXWPMULPQSE-UKSCLKOJSA-N (Z)-(1R)-cis-tefluthrin Chemical compound FC1=C(F)C(C)=C(F)C(F)=C1COC(=O)[C@H]1C(C)(C)[C@H]1\C=C(/Cl)C(F)(F)F ZFHGXWPMULPQSE-UKSCLKOJSA-N 0.000 description 1
- QNBTYORWCCMPQP-JXAWBTAJSA-N (Z)-dimethomorph Chemical compound C1=C(OC)C(OC)=CC=C1C(\C=1C=CC(Cl)=CC=1)=C/C(=O)N1CCOCC1 QNBTYORWCCMPQP-JXAWBTAJSA-N 0.000 description 1
- PCKNFPQPGUWFHO-SXBRIOAWSA-N (Z)-flucycloxuron Chemical compound FC1=CC=CC(F)=C1C(=O)NC(=O)NC(C=C1)=CC=C1CO\N=C(C=1C=CC(Cl)=CC=1)\C1CC1 PCKNFPQPGUWFHO-SXBRIOAWSA-N 0.000 description 1
- HOKKPVIRMVDYPB-UVTDQMKNSA-N (Z)-thiacloprid Chemical compound C1=NC(Cl)=CC=C1CN1C(=N/C#N)/SCC1 HOKKPVIRMVDYPB-UVTDQMKNSA-N 0.000 description 1
- CKPCAYZTYMHQEX-NBVRZTHBSA-N (e)-1-(2,4-dichlorophenyl)-n-methoxy-2-pyridin-3-ylethanimine Chemical compound C=1C=C(Cl)C=C(Cl)C=1C(=N/OC)/CC1=CC=CN=C1 CKPCAYZTYMHQEX-NBVRZTHBSA-N 0.000 description 1
- IKNXXTIMVROREQ-WXXKFALUSA-N (e)-but-2-enedioic acid;[2-[3-(4-chlorophenyl)propyl]-2,4,4-trimethyl-1,3-oxazolidin-3-yl]-imidazol-1-ylmethanone Chemical compound OC(=O)\C=C\C(O)=O.C1=CN=CN1C(=O)N1C(C)(C)COC1(C)CCCC1=CC=C(Cl)C=C1.C1=CN=CN1C(=O)N1C(C)(C)COC1(C)CCCC1=CC=C(Cl)C=C1 IKNXXTIMVROREQ-WXXKFALUSA-N 0.000 description 1
- IGOWHGRNPLFNDJ-ZPHPHTNESA-N (z)-9-tricosene Chemical compound CCCCCCCCCCCCC\C=C/CCCCCCCC IGOWHGRNPLFNDJ-ZPHPHTNESA-N 0.000 description 1
- IAKOZHOLGAGEJT-UHFFFAOYSA-N 1,1,1-trichloro-2,2-bis(p-methoxyphenyl)-Ethane Chemical compound C1=CC(OC)=CC=C1C(C(Cl)(Cl)Cl)C1=CC=C(OC)C=C1 IAKOZHOLGAGEJT-UHFFFAOYSA-N 0.000 description 1
- WBEJYOJJBDISQU-UHFFFAOYSA-N 1,2-Dibromo-3-chloropropane Chemical compound ClCC(Br)CBr WBEJYOJJBDISQU-UHFFFAOYSA-N 0.000 description 1
- GNPWYHFXSMINJQ-UHFFFAOYSA-N 1,2-dimethyl-3-(1-phenylethyl)benzene Chemical compound C=1C=CC(C)=C(C)C=1C(C)C1=CC=CC=C1 GNPWYHFXSMINJQ-UHFFFAOYSA-N 0.000 description 1
- 125000004521 1,3,4-thiadiazol-2-yl group Chemical group S1C(=NN=C1)* 0.000 description 1
- TUBQDCKAWGHZPF-UHFFFAOYSA-N 1,3-benzothiazol-2-ylsulfanylmethyl thiocyanate Chemical compound C1=CC=C2SC(SCSC#N)=NC2=C1 TUBQDCKAWGHZPF-UHFFFAOYSA-N 0.000 description 1
- NCJRWSQYSLEKEI-UHFFFAOYSA-N 1-(1,3-benzothiazol-2-yl)-3-propan-2-ylurea Chemical compound C1=CC=C2SC(NC(=O)NC(C)C)=NC2=C1 NCJRWSQYSLEKEI-UHFFFAOYSA-N 0.000 description 1
- JWUCHKBSVLQQCO-UHFFFAOYSA-N 1-(2-fluorophenyl)-1-(4-fluorophenyl)-2-(1H-1,2,4-triazol-1-yl)ethanol Chemical compound C=1C=C(F)C=CC=1C(C=1C(=CC=CC=1)F)(O)CN1C=NC=N1 JWUCHKBSVLQQCO-UHFFFAOYSA-N 0.000 description 1
- OWEGWHBOCFMBLP-UHFFFAOYSA-N 1-(4-chlorophenoxy)-1-(1H-imidazol-1-yl)-3,3-dimethylbutan-2-one Chemical compound C1=CN=CN1C(C(=O)C(C)(C)C)OC1=CC=C(Cl)C=C1 OWEGWHBOCFMBLP-UHFFFAOYSA-N 0.000 description 1
- WURBVZBTWMNKQT-UHFFFAOYSA-N 1-(4-chlorophenoxy)-3,3-dimethyl-1-(1,2,4-triazol-1-yl)butan-2-one Chemical compound C1=NC=NN1C(C(=O)C(C)(C)C)OC1=CC=C(Cl)C=C1 WURBVZBTWMNKQT-UHFFFAOYSA-N 0.000 description 1
- RURQAJURNPMSSK-UHFFFAOYSA-N 1-(4-chlorophenoxy)-3-{[2-(4-ethoxyphenyl)-3,3,3-trifluoropropoxy]methyl}benzene Chemical compound C1=CC(OCC)=CC=C1C(C(F)(F)F)COCC1=CC=CC(OC=2C=CC(Cl)=CC=2)=C1 RURQAJURNPMSSK-UHFFFAOYSA-N 0.000 description 1
- PXMNMQRDXWABCY-UHFFFAOYSA-N 1-(4-chlorophenyl)-4,4-dimethyl-3-(1H-1,2,4-triazol-1-ylmethyl)pentan-3-ol Chemical compound C1=NC=NN1CC(O)(C(C)(C)C)CCC1=CC=C(Cl)C=C1 PXMNMQRDXWABCY-UHFFFAOYSA-N 0.000 description 1
- IAQLCKZJGNTRDO-UHFFFAOYSA-N 1-(4-{4-[5-(2,6-difluorophenyl)-4,5-dihydro-1,2-oxazol-3-yl]-1,3-thiazol-2-yl}piperidin-1-yl)-2-[5-methyl-3-(trifluoromethyl)-1H-pyrazol-1-yl]ethanone Chemical compound CC1=CC(C(F)(F)F)=NN1CC(=O)N1CCC(C=2SC=C(N=2)C=2CC(ON=2)C=2C(=CC=CC=2F)F)CC1 IAQLCKZJGNTRDO-UHFFFAOYSA-N 0.000 description 1
- VGPIBGGRCVEHQZ-UHFFFAOYSA-N 1-(biphenyl-4-yloxy)-3,3-dimethyl-1-(1,2,4-triazol-1-yl)butan-2-ol Chemical compound C1=NC=NN1C(C(O)C(C)(C)C)OC(C=C1)=CC=C1C1=CC=CC=C1 VGPIBGGRCVEHQZ-UHFFFAOYSA-N 0.000 description 1
- OZOMQRBLCMDCEG-VIZOYTHASA-N 1-[(e)-[5-(4-nitrophenyl)furan-2-yl]methylideneamino]imidazolidine-2,4-dione Chemical compound C1=CC([N+](=O)[O-])=CC=C1C(O1)=CC=C1\C=N\N1C(=O)NC(=O)C1 OZOMQRBLCMDCEG-VIZOYTHASA-N 0.000 description 1
- HVQHXBNMBZJPLK-UHFFFAOYSA-N 1-[2,6-dichloro-4-(trifluoromethyl)phenyl]-5-[(2-methylprop-2-en-1-yl)amino]-4-[(trifluoromethyl)sulfinyl]-1H-pyrazole-3-carbonitrile Chemical compound CC(=C)CNC1=C([S+]([O-])C(F)(F)F)C(C#N)=NN1C1=C(Cl)C=C(C(F)(F)F)C=C1Cl HVQHXBNMBZJPLK-UHFFFAOYSA-N 0.000 description 1
- LQDARGUHUSPFNL-UHFFFAOYSA-N 1-[2-(2,4-dichlorophenyl)-3-(1,1,2,2-tetrafluoroethoxy)propyl]1,2,4-triazole Chemical compound C=1C=C(Cl)C=C(Cl)C=1C(COC(F)(F)C(F)F)CN1C=NC=N1 LQDARGUHUSPFNL-UHFFFAOYSA-N 0.000 description 1
- WKBPZYKAUNRMKP-UHFFFAOYSA-N 1-[2-(2,4-dichlorophenyl)pentyl]1,2,4-triazole Chemical compound C=1C=C(Cl)C=C(Cl)C=1C(CCC)CN1C=NC=N1 WKBPZYKAUNRMKP-UHFFFAOYSA-N 0.000 description 1
- PZBPKYOVPCNPJY-UHFFFAOYSA-N 1-[2-(allyloxy)-2-(2,4-dichlorophenyl)ethyl]imidazole Chemical compound ClC1=CC(Cl)=CC=C1C(OCC=C)CN1C=NC=C1 PZBPKYOVPCNPJY-UHFFFAOYSA-N 0.000 description 1
- MGNFYQILYYYUBS-UHFFFAOYSA-N 1-[3-(4-tert-butylphenyl)-2-methylpropyl]piperidine Chemical compound C=1C=C(C(C)(C)C)C=CC=1CC(C)CN1CCCCC1 MGNFYQILYYYUBS-UHFFFAOYSA-N 0.000 description 1
- RWNUSVWFHDHRCJ-UHFFFAOYSA-N 1-butoxypropan-2-ol Chemical compound CCCCOCC(C)O RWNUSVWFHDHRCJ-UHFFFAOYSA-N 0.000 description 1
- YIKWKLYQRFRGPM-UHFFFAOYSA-N 1-dodecylguanidine acetate Chemical compound CC(O)=O.CCCCCCCCCCCCN=C(N)N YIKWKLYQRFRGPM-UHFFFAOYSA-N 0.000 description 1
- JOLQKTGDSGKSKJ-UHFFFAOYSA-N 1-ethoxypropan-2-ol Chemical class CCOCC(C)O JOLQKTGDSGKSKJ-UHFFFAOYSA-N 0.000 description 1
- LMDZBCPBFSXMTL-UHFFFAOYSA-N 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide Chemical compound CCN=C=NCCCN(C)C LMDZBCPBFSXMTL-UHFFFAOYSA-N 0.000 description 1
- 125000004776 1-fluoroethyl group Chemical group [H]C([H])([H])C([H])(F)* 0.000 description 1
- 125000006432 1-methyl cyclopropyl group Chemical group [H]C([H])([H])C1(*)C([H])([H])C1([H])[H] 0.000 description 1
- PFFIDZXUXFLSSR-UHFFFAOYSA-N 1-methyl-N-[2-(4-methylpentan-2-yl)-3-thienyl]-3-(trifluoromethyl)pyrazole-4-carboxamide Chemical compound S1C=CC(NC(=O)C=2C(=NN(C)C=2)C(F)(F)F)=C1C(C)CC(C)C PFFIDZXUXFLSSR-UHFFFAOYSA-N 0.000 description 1
- 125000006018 1-methyl-ethenyl group Chemical group 0.000 description 1
- NFGXHKASABOEEW-UHFFFAOYSA-N 1-methylethyl 11-methoxy-3,7,11-trimethyl-2,4-dodecadienoate Chemical compound COC(C)(C)CCCC(C)CC=CC(C)=CC(=O)OC(C)C NFGXHKASABOEEW-UHFFFAOYSA-N 0.000 description 1
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 description 1
- IBLKWZIFZMJLFL-UHFFFAOYSA-N 1-phenoxypropan-2-ol Chemical compound CC(O)COC1=CC=CC=C1 IBLKWZIFZMJLFL-UHFFFAOYSA-N 0.000 description 1
- YKMMLFOYDTYAGR-UHFFFAOYSA-N 1-phenyl-2-(propan-2-ylamino)pentan-1-one Chemical compound CCCC(NC(C)C)C(=O)C1=CC=CC=C1 YKMMLFOYDTYAGR-UHFFFAOYSA-N 0.000 description 1
- 125000006017 1-propenyl group Chemical group 0.000 description 1
- XUJLWPFSUCHPQL-UHFFFAOYSA-N 11-methyldodecan-1-ol Chemical compound CC(C)CCCCCCCCCCO XUJLWPFSUCHPQL-UHFFFAOYSA-N 0.000 description 1
- 125000000453 2,2,2-trichloroethyl group Chemical group [H]C([H])(*)C(Cl)(Cl)Cl 0.000 description 1
- 125000004793 2,2,2-trifluoroethoxy group Chemical group FC(CO*)(F)F 0.000 description 1
- 125000004206 2,2,2-trifluoroethyl group Chemical group [H]C([H])(*)C(F)(F)F 0.000 description 1
- 125000004778 2,2-difluoroethyl group Chemical group [H]C([H])(*)C([H])(F)F 0.000 description 1
- YQTCQNIPQMJNTI-UHFFFAOYSA-N 2,2-dimethylpropan-1-one Chemical group CC(C)(C)[C]=O YQTCQNIPQMJNTI-UHFFFAOYSA-N 0.000 description 1
- PGZVFRAEAAXREB-UHFFFAOYSA-N 2,2-dimethylpropanoyl 2,2-dimethylpropanoate Chemical compound CC(C)(C)C(=O)OC(=O)C(C)(C)C PGZVFRAEAAXREB-UHFFFAOYSA-N 0.000 description 1
- SVPKNMBRVBMTLB-UHFFFAOYSA-N 2,3-dichloronaphthalene-1,4-dione Chemical compound C1=CC=C2C(=O)C(Cl)=C(Cl)C(=O)C2=C1 SVPKNMBRVBMTLB-UHFFFAOYSA-N 0.000 description 1
- NIOPZPCMRQGZCE-WEVVVXLNSA-N 2,4-dinitro-6-(octan-2-yl)phenyl (E)-but-2-enoate Chemical compound CCCCCCC(C)C1=CC([N+]([O-])=O)=CC([N+]([O-])=O)=C1OC(=O)\C=C\C NIOPZPCMRQGZCE-WEVVVXLNSA-N 0.000 description 1
- XEPBBUCQCXXTGR-UHFFFAOYSA-N 2,5-dimethyl-n-phenylfuran-3-carboxamide Chemical compound O1C(C)=CC(C(=O)NC=2C=CC=CC=2)=C1C XEPBBUCQCXXTGR-UHFFFAOYSA-N 0.000 description 1
- STMIIPIFODONDC-UHFFFAOYSA-N 2-(2,4-dichlorophenyl)-1-(1H-1,2,4-triazol-1-yl)hexan-2-ol Chemical compound C=1C=C(Cl)C=C(Cl)C=1C(O)(CCCC)CN1C=NC=N1 STMIIPIFODONDC-UHFFFAOYSA-N 0.000 description 1
- DNBMPXLFKQCOBV-UHFFFAOYSA-N 2-(2-ethoxyethoxy)ethyl n-(1h-benzimidazol-2-yl)carbamate Chemical compound C1=CC=C2NC(NC(=O)OCCOCCOCC)=NC2=C1 DNBMPXLFKQCOBV-UHFFFAOYSA-N 0.000 description 1
- RUGYNGIMTAFTLP-UHFFFAOYSA-N 2-(3,5-dimethylpyrazol-1-yl)-1h-benzimidazole Chemical compound N1=C(C)C=C(C)N1C1=NC2=CC=CC=C2N1 RUGYNGIMTAFTLP-UHFFFAOYSA-N 0.000 description 1
- UFNOUKDBUJZYDE-UHFFFAOYSA-N 2-(4-chlorophenyl)-3-cyclopropyl-1-(1H-1,2,4-triazol-1-yl)butan-2-ol Chemical compound C1=NC=NN1CC(O)(C=1C=CC(Cl)=CC=1)C(C)C1CC1 UFNOUKDBUJZYDE-UHFFFAOYSA-N 0.000 description 1
- KWLVWJPJKJMCSH-UHFFFAOYSA-N 2-(4-chlorophenyl)-N-{2-[3-methoxy-4-(prop-2-yn-1-yloxy)phenyl]ethyl}-2-(prop-2-yn-1-yloxy)acetamide Chemical compound C1=C(OCC#C)C(OC)=CC(CCNC(=O)C(OCC#C)C=2C=CC(Cl)=CC=2)=C1 KWLVWJPJKJMCSH-UHFFFAOYSA-N 0.000 description 1
- YABFPHSQTSFWQB-UHFFFAOYSA-N 2-(4-fluorophenyl)-1-(1,2,4-triazol-1-yl)-3-(trimethylsilyl)propan-2-ol Chemical compound C=1C=C(F)C=CC=1C(O)(C[Si](C)(C)C)CN1C=NC=N1 YABFPHSQTSFWQB-UHFFFAOYSA-N 0.000 description 1
- NGLCOYIAJMJYQI-UHFFFAOYSA-N 2-(4-methoxyiminocyclohexyl)-2-(3,3,3-trifluoropropylsulfonyl)acetonitrile Chemical compound CON=C1CCC(C(C#N)S(=O)(=O)CCC(F)(F)F)CC1 NGLCOYIAJMJYQI-UHFFFAOYSA-N 0.000 description 1
- OTWBGXPTCSGQEJ-UHFFFAOYSA-N 2-[(dimethylcarbamothioyldisulfanyl)carbothioylamino]ethylcarbamothioylsulfanyl n,n-dimethylcarbamodithioate Chemical compound CN(C)C(=S)SSC(=S)NCCNC(=S)SSC(=S)N(C)C OTWBGXPTCSGQEJ-UHFFFAOYSA-N 0.000 description 1
- MNHVNIJQQRJYDH-UHFFFAOYSA-N 2-[2-(1-chlorocyclopropyl)-3-(2-chlorophenyl)-2-hydroxypropyl]-1,2-dihydro-1,2,4-triazole-3-thione Chemical compound N1=CNC(=S)N1CC(C1(Cl)CC1)(O)CC1=CC=CC=C1Cl MNHVNIJQQRJYDH-UHFFFAOYSA-N 0.000 description 1
- BOTNFCTYKJBUMU-UHFFFAOYSA-N 2-[4-(2-methylpropyl)piperazin-4-ium-1-yl]-2-oxoacetate Chemical compound CC(C)C[NH+]1CCN(C(=O)C([O-])=O)CC1 BOTNFCTYKJBUMU-UHFFFAOYSA-N 0.000 description 1
- JERZEQUMJNCPRJ-UHFFFAOYSA-N 2-[4-(4-chlorophenoxy)-2-(trifluoromethyl)phenyl]-1-(1H-1,2,4-triazol-1-yl)propan-2-ol Chemical compound C=1C=C(OC=2C=CC(Cl)=CC=2)C=C(C(F)(F)F)C=1C(O)(C)CN1C=NC=N1 JERZEQUMJNCPRJ-UHFFFAOYSA-N 0.000 description 1
- SIIJJFOXEOHODQ-UHFFFAOYSA-N 2-[4-(4-chlorophenoxy)-2-(trifluoromethyl)phenyl]-3-methyl-1-(1,2,4-triazol-1-yl)butan-2-ol Chemical compound C=1C=C(OC=2C=CC(Cl)=CC=2)C=C(C(F)(F)F)C=1C(O)(C(C)C)CN1C=NC=N1 SIIJJFOXEOHODQ-UHFFFAOYSA-N 0.000 description 1
- ULNWEXDKQHEUSK-UHFFFAOYSA-N 2-[ethoxy-(propan-2-ylamino)phosphoryl]sulfanyl-n-methyl-n-phenylacetamide Chemical compound CCOP(=O)(NC(C)C)SCC(=O)N(C)C1=CC=CC=C1 ULNWEXDKQHEUSK-UHFFFAOYSA-N 0.000 description 1
- 125000005999 2-bromoethyl group Chemical group 0.000 description 1
- 125000004974 2-butenyl group Chemical group C(C=CC)* 0.000 description 1
- POAOYUHQDCAZBD-UHFFFAOYSA-N 2-butoxyethanol Chemical compound CCCCOCCO POAOYUHQDCAZBD-UHFFFAOYSA-N 0.000 description 1
- 125000000069 2-butynyl group Chemical group [H]C([H])([H])C#CC([H])([H])* 0.000 description 1
- 125000004780 2-chloro-2,2-difluoroethyl group Chemical group [H]C([H])(*)C(F)(F)Cl 0.000 description 1
- OGBSAJWRIPNIER-UHFFFAOYSA-N 2-chloro-6-(furan-2-ylmethoxy)-4-(trichloromethyl)pyridine Chemical compound ClC1=CC(C(Cl)(Cl)Cl)=CC(OCC=2OC=CC=2)=N1 OGBSAJWRIPNIER-UHFFFAOYSA-N 0.000 description 1
- 125000006012 2-chloroethoxy group Chemical group 0.000 description 1
- 125000001340 2-chloroethyl group Chemical group [H]C([H])(Cl)C([H])([H])* 0.000 description 1
- SUODCTNNAKSRHB-UHFFFAOYSA-N 2-ethylhexyl 3-sulfanylpropanoate Chemical compound CCCCC(CC)COC(=O)CCS SUODCTNNAKSRHB-UHFFFAOYSA-N 0.000 description 1
- 125000004791 2-fluoroethoxy group Chemical group FCCO* 0.000 description 1
- 125000004777 2-fluoroethyl group Chemical group [H]C([H])(F)C([H])([H])* 0.000 description 1
- 125000006022 2-methyl-2-propenyl group Chemical group 0.000 description 1
- QTWJRLJHJPIABL-UHFFFAOYSA-N 2-methylphenol;3-methylphenol;4-methylphenol Chemical compound CC1=CC=C(O)C=C1.CC1=CC=CC(O)=C1.CC1=CC=CC=C1O QTWJRLJHJPIABL-UHFFFAOYSA-N 0.000 description 1
- 229940044120 2-n-octyl-4-isothiazolin-3-one Drugs 0.000 description 1
- PIAOLBVUVDXHHL-UHFFFAOYSA-N 2-nitroethenylbenzene Chemical compound [O-][N+](=O)C=CC1=CC=CC=C1 PIAOLBVUVDXHHL-UHFFFAOYSA-N 0.000 description 1
- AVGVFDSUDIUXEU-UHFFFAOYSA-N 2-octyl-1,2-thiazolidin-3-one Chemical compound CCCCCCCCN1SCCC1=O AVGVFDSUDIUXEU-UHFFFAOYSA-N 0.000 description 1
- LEACJMVNYZDSKR-UHFFFAOYSA-N 2-octyldodecan-1-ol Chemical compound CCCCCCCCCCC(CO)CCCCCCCC LEACJMVNYZDSKR-UHFFFAOYSA-N 0.000 description 1
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 1
- YEYKMVJDLWJFOA-UHFFFAOYSA-N 2-propoxyethanol Chemical compound CCCOCCO YEYKMVJDLWJFOA-UHFFFAOYSA-N 0.000 description 1
- 125000001494 2-propynyl group Chemical group [H]C#CC([H])([H])* 0.000 description 1
- ZRDUSMYWDRPZRM-UHFFFAOYSA-N 2-sec-butyl-4,6-dinitrophenyl 3-methylbut-2-enoate Chemical compound CCC(C)C1=CC([N+]([O-])=O)=CC([N+]([O-])=O)=C1OC(=O)C=C(C)C ZRDUSMYWDRPZRM-UHFFFAOYSA-N 0.000 description 1
- PDPWCKVFIFAQIQ-UHFFFAOYSA-N 2-{2-[(2,5-dimethylphenoxy)methyl]phenyl}-2-methoxy-N-methylacetamide Chemical compound CNC(=O)C(OC)C1=CC=CC=C1COC1=CC(C)=CC=C1C PDPWCKVFIFAQIQ-UHFFFAOYSA-N 0.000 description 1
- SOUGWDPPRBKJEX-UHFFFAOYSA-N 3,5-dichloro-N-(1-chloro-3-methyl-2-oxopentan-3-yl)-4-methylbenzamide Chemical compound ClCC(=O)C(C)(CC)NC(=O)C1=CC(Cl)=C(C)C(Cl)=C1 SOUGWDPPRBKJEX-UHFFFAOYSA-N 0.000 description 1
- OVFHHJZHXHZIHT-UHFFFAOYSA-N 3-(2,4-dichlorophenyl)-2-(1,2,4-triazol-1-yl)quinazolin-4-one Chemical compound ClC1=CC(Cl)=CC=C1N1C(=O)C2=CC=CC=C2N=C1N1N=CN=C1 OVFHHJZHXHZIHT-UHFFFAOYSA-N 0.000 description 1
- SWBHWUYHHJCADA-UHFFFAOYSA-N 3-(2-chlorophenyl)-6-(2,6-difluorophenyl)-1,2,4,5-tetrazine Chemical compound FC1=CC=CC(F)=C1C1=NN=C(C=2C(=CC=CC=2)Cl)N=N1 SWBHWUYHHJCADA-UHFFFAOYSA-N 0.000 description 1
- BZGLBXYQOMFXAU-UHFFFAOYSA-N 3-(2-methylpiperidin-1-yl)propyl 3,4-dichlorobenzoate Chemical compound CC1CCCCN1CCCOC(=O)C1=CC=C(Cl)C(Cl)=C1 BZGLBXYQOMFXAU-UHFFFAOYSA-N 0.000 description 1
- GPUHJQHXIFJPGN-UHFFFAOYSA-N 3-(3,5-dichlorophenyl)-1-(3-methylbutanoyl)imidazolidine-2,4-dione Chemical compound O=C1N(C(=O)CC(C)C)CC(=O)N1C1=CC(Cl)=CC(Cl)=C1 GPUHJQHXIFJPGN-UHFFFAOYSA-N 0.000 description 1
- FSCWZHGZWWDELK-UHFFFAOYSA-N 3-(3,5-dichlorophenyl)-5-ethenyl-5-methyl-2,4-oxazolidinedione Chemical compound O=C1C(C)(C=C)OC(=O)N1C1=CC(Cl)=CC(Cl)=C1 FSCWZHGZWWDELK-UHFFFAOYSA-N 0.000 description 1
- QCAHUFWKIQLBNB-UHFFFAOYSA-N 3-(3-methoxypropoxy)propan-1-ol Chemical compound COCCCOCCCO QCAHUFWKIQLBNB-UHFFFAOYSA-N 0.000 description 1
- HBLSGPQATABNRY-UHFFFAOYSA-N 3-(4-chlorophenyl)-2-n-methyl-4-n,5-n-bis(trifluoromethyl)-1,3-thiazolidine-2,4,5-triimine Chemical compound CN=C1SC(=NC(F)(F)F)C(=NC(F)(F)F)N1C1=CC=C(Cl)C=C1 HBLSGPQATABNRY-UHFFFAOYSA-N 0.000 description 1
- XTDZGXBTXBEZDN-UHFFFAOYSA-N 3-(difluoromethyl)-N-(9-isopropyl-1,2,3,4-tetrahydro-1,4-methanonaphthalen-5-yl)-1-methylpyrazole-4-carboxamide Chemical compound CC(C)C1C2CCC1C1=C2C=CC=C1NC(=O)C1=CN(C)N=C1C(F)F XTDZGXBTXBEZDN-UHFFFAOYSA-N 0.000 description 1
- DGOAXBPOVUPPEB-UHFFFAOYSA-N 3-(difluoromethyl)-N-methoxy-1-methyl-N-[1-(2,4,6-trichlorophenyl)propan-2-yl]pyrazole-4-carboxamide Chemical compound C=1N(C)N=C(C(F)F)C=1C(=O)N(OC)C(C)CC1=C(Cl)C=C(Cl)C=C1Cl DGOAXBPOVUPPEB-UHFFFAOYSA-N 0.000 description 1
- PKLPQOJFHFGVBS-UHFFFAOYSA-N 3-acetyl-4-hydroxy-6-methyl-2h-pyran-2-one Chemical compound CC(=O)C1=C(O)C=C(C)OC1=O PKLPQOJFHFGVBS-UHFFFAOYSA-N 0.000 description 1
- BMYNFMYTOJXKLE-UHFFFAOYSA-N 3-azaniumyl-2-hydroxypropanoate Chemical compound NCC(O)C(O)=O BMYNFMYTOJXKLE-UHFFFAOYSA-N 0.000 description 1
- RAMUASXTSSXCMB-UHFFFAOYSA-N 3-bromo-N-{2-bromo-4-chloro-6-[(1-cyclopropylethyl)carbamoyl]phenyl}-1-(3-chloropyridin-2-yl)-1H-pyrazole-5-carboxamide Chemical compound C1CC1C(C)NC(=O)C1=CC(Cl)=CC(Br)=C1NC(=O)C1=CC(Br)=NN1C1=NC=CC=C1Cl RAMUASXTSSXCMB-UHFFFAOYSA-N 0.000 description 1
- 125000000474 3-butynyl group Chemical group [H]C#CC([H])([H])C([H])([H])* 0.000 description 1
- LDMRLRNXHLPZJN-UHFFFAOYSA-N 3-propoxypropan-1-ol Chemical compound CCCOCCCO LDMRLRNXHLPZJN-UHFFFAOYSA-N 0.000 description 1
- SWTPIYGGSMJRTB-UHFFFAOYSA-N 4,4-difluoro-3,3-dimethyl-1-quinolin-3-ylisoquinoline Chemical compound C12=CC=CC=C2C(F)(F)C(C)(C)N=C1C1=CN=C(C=CC=C2)C2=C1 SWTPIYGGSMJRTB-UHFFFAOYSA-N 0.000 description 1
- NMWKWBPNKPGATC-UHFFFAOYSA-N 4,5,6,7-tetrachloro-2-benzofuran-1(3H)-one Chemical compound ClC1=C(Cl)C(Cl)=C2COC(=O)C2=C1Cl NMWKWBPNKPGATC-UHFFFAOYSA-N 0.000 description 1
- ZXVONLUNISGICL-UHFFFAOYSA-N 4,6-dinitro-o-cresol Chemical compound CC1=CC([N+]([O-])=O)=CC([N+]([O-])=O)=C1O ZXVONLUNISGICL-UHFFFAOYSA-N 0.000 description 1
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 1
- RQDJADAKIFFEKQ-UHFFFAOYSA-N 4-(4-chlorophenyl)-2-phenyl-2-(1,2,4-triazol-1-ylmethyl)butanenitrile Chemical compound C1=CC(Cl)=CC=C1CCC(C=1C=CC=CC=1)(C#N)CN1N=CN=C1 RQDJADAKIFFEKQ-UHFFFAOYSA-N 0.000 description 1
- UJVMVEZTSSMXAC-UHFFFAOYSA-N 4-[(3-chlorophenyl)diazenyl]-3-methyl-2H-1,2-oxazol-5-one Chemical compound ClC=1C=C(C=CC=1)N=NC1=C(NOC1=O)C UJVMVEZTSSMXAC-UHFFFAOYSA-N 0.000 description 1
- QDFVXXBCJYNKKC-UHFFFAOYSA-N 4-[4-(4-chlorophenyl)-4-cyclopropylbutyl]-1-fluoro-2-phenoxybenzene Chemical compound C1=C(OC=2C=CC=CC=2)C(F)=CC=C1CCCC(C=1C=CC(Cl)=CC=1)C1CC1 QDFVXXBCJYNKKC-UHFFFAOYSA-N 0.000 description 1
- OXDDDHGGRFRLEE-UHFFFAOYSA-N 4-[5-[3-chloro-5-(trifluoromethyl)phenyl]-5-(trifluoromethyl)-4h-1,2-oxazol-3-yl]-n-[2-oxo-2-(2,2,2-trifluoroethylamino)ethyl]naphthalene-1-carboxamide Chemical compound C12=CC=CC=C2C(C(=O)NCC(=O)NCC(F)(F)F)=CC=C1C(C1)=NOC1(C(F)(F)F)C1=CC(Cl)=CC(C(F)(F)F)=C1 OXDDDHGGRFRLEE-UHFFFAOYSA-N 0.000 description 1
- QKNNWDFHKRIYJH-UHFFFAOYSA-N 4-bromo-3-fluorobenzoyl chloride Chemical compound FC1=CC(C(Cl)=O)=CC=C1Br QKNNWDFHKRIYJH-UHFFFAOYSA-N 0.000 description 1
- ZOMKCDYJHAQMCU-UHFFFAOYSA-N 4-butyl-1,2,4-triazole Chemical compound CCCCN1C=NN=C1 ZOMKCDYJHAQMCU-UHFFFAOYSA-N 0.000 description 1
- QCPASDYEQAVIJF-UHFFFAOYSA-N 4-chloro-3-methyl-1,3-benzothiazol-2-one Chemical compound C1=CC=C2SC(=O)N(C)C2=C1Cl QCPASDYEQAVIJF-UHFFFAOYSA-N 0.000 description 1
- HBTAOSGHCXUEKI-UHFFFAOYSA-N 4-chloro-n,n-dimethyl-3-nitrobenzenesulfonamide Chemical compound CN(C)S(=O)(=O)C1=CC=C(Cl)C([N+]([O-])=O)=C1 HBTAOSGHCXUEKI-UHFFFAOYSA-N 0.000 description 1
- SBUKOHLFHYSZNG-UHFFFAOYSA-N 4-dodecyl-2,6-dimethylmorpholine Chemical compound CCCCCCCCCCCCN1CC(C)OC(C)C1 SBUKOHLFHYSZNG-UHFFFAOYSA-N 0.000 description 1
- 229940090248 4-hydroxybenzoic acid Drugs 0.000 description 1
- RSMQQONIFJLFAK-UHFFFAOYSA-N 4-iodopyridin-2-amine Chemical compound NC1=CC(I)=CC=N1 RSMQQONIFJLFAK-UHFFFAOYSA-N 0.000 description 1
- LHZOTJOOBRODLL-UHFFFAOYSA-N 4-oxo-1-(pyrimidin-5-ylmethyl)-3-[3-(trifluoromethyl)phenyl]pyrido[1,2-a]pyrimidin-5-ium-2-olate Chemical compound O=C1[N+]2=CC=CC=C2N(CC=2C=NC=NC=2)C([O-])=C1C1=CC=CC(C(F)(F)F)=C1 LHZOTJOOBRODLL-UHFFFAOYSA-N 0.000 description 1
- BFTGQIQVUVTBJU-UHFFFAOYSA-N 5,6-dihydroimidazo[2,1-c][1,2,4]dithiazole-3-thione Chemical compound C1CN2C(=S)SSC2=N1 BFTGQIQVUVTBJU-UHFFFAOYSA-N 0.000 description 1
- ZOCSXAVNDGMNBV-UHFFFAOYSA-N 5-amino-1-[2,6-dichloro-4-(trifluoromethyl)phenyl]-4-[(trifluoromethyl)sulfinyl]-1H-pyrazole-3-carbonitrile Chemical compound NC1=C(S(=O)C(F)(F)F)C(C#N)=NN1C1=C(Cl)C=C(C(F)(F)F)C=C1Cl ZOCSXAVNDGMNBV-UHFFFAOYSA-N 0.000 description 1
- XJFIKRXIJXAJGH-UHFFFAOYSA-N 5-chloro-1,3-dihydroimidazo[4,5-b]pyridin-2-one Chemical group ClC1=CC=C2NC(=O)NC2=N1 XJFIKRXIJXAJGH-UHFFFAOYSA-N 0.000 description 1
- NRTLIYOWLVMQBO-UHFFFAOYSA-N 5-chloro-1,3-dimethyl-N-(1,1,3-trimethyl-1,3-dihydro-2-benzofuran-4-yl)pyrazole-4-carboxamide Chemical compound C=12C(C)OC(C)(C)C2=CC=CC=1NC(=O)C=1C(C)=NN(C)C=1Cl NRTLIYOWLVMQBO-UHFFFAOYSA-N 0.000 description 1
- NEKULYKCZPJMMJ-UHFFFAOYSA-N 5-chloro-N-{1-[4-(difluoromethoxy)phenyl]propyl}-6-methylpyrimidin-4-amine Chemical compound C=1C=C(OC(F)F)C=CC=1C(CC)NC1=NC=NC(C)=C1Cl NEKULYKCZPJMMJ-UHFFFAOYSA-N 0.000 description 1
- GOFJDXZZHFNFLV-UHFFFAOYSA-N 5-fluoro-1,3-dimethyl-N-[2-(4-methylpentan-2-yl)phenyl]pyrazole-4-carboxamide Chemical compound CC(C)CC(C)C1=CC=CC=C1NC(=O)C1=C(F)N(C)N=C1C GOFJDXZZHFNFLV-UHFFFAOYSA-N 0.000 description 1
- SJDGOKGRYGWNTC-UHFFFAOYSA-N 5-isothiocyanato-2-methoxy-n,n,3-trimethylbenzamide Chemical compound COC1=C(C)C=C(N=C=S)C=C1C(=O)N(C)C SJDGOKGRYGWNTC-UHFFFAOYSA-N 0.000 description 1
- IBSREHMXUMOFBB-JFUDTMANSA-N 5u8924t11h Chemical compound O1[C@@H](C)[C@H](O)[C@@H](OC)C[C@@H]1O[C@@H]1[C@@H](OC)C[C@H](O[C@@H]2C(=C/C[C@@H]3C[C@@H](C[C@@]4(O3)C=C[C@H](C)[C@@H](C(C)C)O4)OC(=O)[C@@H]3C=C(C)[C@@H](O)[C@H]4OC\C([C@@]34O)=C/C=C/[C@@H]2C)/C)O[C@H]1C.C1=C[C@H](C)[C@@H]([C@@H](C)CC)O[C@]11O[C@H](C\C=C(C)\[C@@H](O[C@@H]2O[C@@H](C)[C@H](O[C@@H]3O[C@@H](C)[C@H](O)[C@@H](OC)C3)[C@@H](OC)C2)[C@@H](C)\C=C\C=C/2[C@]3([C@H](C(=O)O4)C=C(C)[C@@H](O)[C@H]3OC\2)O)C[C@H]4C1 IBSREHMXUMOFBB-JFUDTMANSA-N 0.000 description 1
- FHVDTGUDJYJELY-UHFFFAOYSA-N 6-{[2-carboxy-4,5-dihydroxy-6-(phosphanyloxy)oxan-3-yl]oxy}-4,5-dihydroxy-3-phosphanyloxane-2-carboxylic acid Chemical compound O1C(C(O)=O)C(P)C(O)C(O)C1OC1C(C(O)=O)OC(OP)C(O)C1O FHVDTGUDJYJELY-UHFFFAOYSA-N 0.000 description 1
- VSVKOUBCDZYAQY-UHFFFAOYSA-N 7-chloro-1,2-benzothiazole Chemical compound ClC1=CC=CC2=C1SN=C2 VSVKOUBCDZYAQY-UHFFFAOYSA-N 0.000 description 1
- GJCOSYZMQJWQCA-UHFFFAOYSA-N 9H-xanthene Chemical compound C1=CC=C2CC3=CC=CC=C3OC2=C1 GJCOSYZMQJWQCA-UHFFFAOYSA-N 0.000 description 1
- 239000005660 Abamectin Substances 0.000 description 1
- 241000967305 Acaphylla theavagrans Species 0.000 description 1
- 239000005651 Acequinocyl Substances 0.000 description 1
- 241001558877 Aceria tulipae Species 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 241000237364 Achatina fulica Species 0.000 description 1
- 239000005964 Acibenzolar-S-methyl Substances 0.000 description 1
- 208000002874 Acne Vulgaris Diseases 0.000 description 1
- 239000005652 Acrinathrin Substances 0.000 description 1
- 241000580761 Acrolepiopsis Species 0.000 description 1
- 241000540209 Acrolepiopsis sapporensis Species 0.000 description 1
- 241000079319 Aculops lycopersici Species 0.000 description 1
- 241001159389 Aculops pelekassi Species 0.000 description 1
- 241001506414 Aculus Species 0.000 description 1
- 241000515126 Acusta despecta sieboldiana Species 0.000 description 1
- 241001672674 Adoxophyes honmai Species 0.000 description 1
- 241000175828 Adoxophyes orana Species 0.000 description 1
- 241000256111 Aedes <genus> Species 0.000 description 1
- 241000256118 Aedes aegypti Species 0.000 description 1
- 241000256173 Aedes albopictus Species 0.000 description 1
- 229920001817 Agar Polymers 0.000 description 1
- 241000993143 Agromyza Species 0.000 description 1
- 241000566547 Agrotis ipsilon Species 0.000 description 1
- 241000218475 Agrotis segetum Species 0.000 description 1
- YRRKLBAKDXSTNC-UHFFFAOYSA-N Aldicarb sulfonyl Natural products CNC(=O)ON=CC(C)(C)S(C)(=O)=O YRRKLBAKDXSTNC-UHFFFAOYSA-N 0.000 description 1
- YRRKLBAKDXSTNC-WEVVVXLNSA-N Aldoxycarb Chemical compound CNC(=O)O\N=C\C(C)(C)S(C)(=O)=O YRRKLBAKDXSTNC-WEVVVXLNSA-N 0.000 description 1
- 241000292372 Aleurocanthus spiniferus Species 0.000 description 1
- 235000019489 Almond oil Nutrition 0.000 description 1
- 239000005877 Alpha-Cypermethrin Substances 0.000 description 1
- 239000005995 Aluminium silicate Substances 0.000 description 1
- 241000238679 Amblyomma Species 0.000 description 1
- 239000005726 Ametoctradin Substances 0.000 description 1
- 239000005727 Amisulbrom Substances 0.000 description 1
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 1
- 241001147657 Ancylostoma Species 0.000 description 1
- 241000519879 Anomala cuprea Species 0.000 description 1
- 241000519878 Anomala rufocuprea Species 0.000 description 1
- 241001463392 Anoplophora macularia Species 0.000 description 1
- 241000254175 Anthonomus grandis Species 0.000 description 1
- 241001414828 Aonidiella aurantii Species 0.000 description 1
- 241000276082 Apethymus Species 0.000 description 1
- 241000134843 Aphelenchoides besseyi Species 0.000 description 1
- 241000273311 Aphis spiraecola Species 0.000 description 1
- 241000838579 Arboridia Species 0.000 description 1
- 241001002469 Archips Species 0.000 description 1
- 241001423665 Archips fuscocupreana Species 0.000 description 1
- 241000291885 Arge pagana Species 0.000 description 1
- 241000722809 Armadillidium vulgare Species 0.000 description 1
- 241000238421 Arthropoda Species 0.000 description 1
- 241000204727 Ascaridia Species 0.000 description 1
- 241000244186 Ascaris Species 0.000 description 1
- 241001002591 Ascotis selenaria Species 0.000 description 1
- 241001485662 Asphondylia yushimai Species 0.000 description 1
- 241000238017 Astacoidea Species 0.000 description 1
- 241000258957 Asteroidea Species 0.000 description 1
- 241001503477 Athalia rosae Species 0.000 description 1
- 241000902805 Aulacophora Species 0.000 description 1
- 241001367035 Autographa nigrisigna Species 0.000 description 1
- 239000005730 Azoxystrobin Substances 0.000 description 1
- 241000193388 Bacillus thuringiensis Species 0.000 description 1
- 241001124181 Bactrocera dorsalis Species 0.000 description 1
- 241001260001 Balclutha Species 0.000 description 1
- 239000005734 Benalaxyl Substances 0.000 description 1
- MQIVNMHALJNFMG-ILYHAJGMSA-N Benquinox Chemical compound O/N=C(\C=C1)/C=C/C\1=N\NC(C1=CC=CC=C1)=O MQIVNMHALJNFMG-ILYHAJGMSA-N 0.000 description 1
- 239000005737 Benzovindiflupyr Substances 0.000 description 1
- 239000005884 Beta-Cyfluthrin Substances 0.000 description 1
- 239000005653 Bifenazate Substances 0.000 description 1
- 239000005874 Bifenthrin Substances 0.000 description 1
- 239000005738 Bixafen Substances 0.000 description 1
- 239000005739 Bordeaux mixture Substances 0.000 description 1
- 239000005740 Boscalid Substances 0.000 description 1
- 241001217818 Bourletiella hortensis Species 0.000 description 1
- 241000322476 Bovicola bovis Species 0.000 description 1
- 240000007124 Brassica oleracea Species 0.000 description 1
- 235000003899 Brassica oleracea var acephala Nutrition 0.000 description 1
- 235000011301 Brassica oleracea var capitata Nutrition 0.000 description 1
- 235000001169 Brassica oleracea var oleracea Nutrition 0.000 description 1
- 235000010149 Brassica rapa subsp chinensis Nutrition 0.000 description 1
- 235000000536 Brassica rapa subsp pekinensis Nutrition 0.000 description 1
- 241000499436 Brassica rapa subsp. pekinensis Species 0.000 description 1
- 239000005741 Bromuconazole Substances 0.000 description 1
- LVDKZNITIUWNER-UHFFFAOYSA-N Bronopol Chemical compound OCC(Br)(CO)[N+]([O-])=O LVDKZNITIUWNER-UHFFFAOYSA-N 0.000 description 1
- 241001517925 Bucculatrix Species 0.000 description 1
- 241000931178 Bunostomum Species 0.000 description 1
- 239000005742 Bupirimate Substances 0.000 description 1
- 239000005885 Buprofezin Substances 0.000 description 1
- 241000243770 Bursaphelenchus Species 0.000 description 1
- 241000243771 Bursaphelenchus xylophilus Species 0.000 description 1
- ZZVVDIVWGXTDRQ-BSYVCWPDSA-N Buthiobate Chemical compound C=1C=CN=CC=1\N=C(/SCCCC)SCC1=CC=C(C(C)(C)C)C=C1 ZZVVDIVWGXTDRQ-BSYVCWPDSA-N 0.000 description 1
- DKPFZGUDAPQIHT-UHFFFAOYSA-N Butyl acetate Natural products CCCCOC(C)=O DKPFZGUDAPQIHT-UHFFFAOYSA-N 0.000 description 1
- NLZUEZXRPGMBCV-UHFFFAOYSA-N Butylhydroxytoluene Chemical compound CC1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 NLZUEZXRPGMBCV-UHFFFAOYSA-N 0.000 description 1
- MWPPEOGOSMKXOT-UHFFFAOYSA-N CCOC(=O)C1=C(N2C=CC(=CC2=N1)I)SCC Chemical compound CCOC(=O)C1=C(N2C=CC(=CC2=N1)I)SCC MWPPEOGOSMKXOT-UHFFFAOYSA-N 0.000 description 1
- JFLRKDZMHNBDQS-UCQUSYKYSA-N CC[C@H]1CCC[C@@H]([C@H](C(=O)C2=C[C@H]3[C@@H]4C[C@@H](C[C@H]4C(=C[C@H]3[C@@H]2CC(=O)O1)C)O[C@H]5[C@@H]([C@@H]([C@H]([C@@H](O5)C)OC)OC)OC)C)O[C@H]6CC[C@@H]([C@H](O6)C)N(C)C.CC[C@H]1CCC[C@@H]([C@H](C(=O)C2=C[C@H]3[C@@H]4C[C@@H](C[C@H]4C=C[C@H]3C2CC(=O)O1)O[C@H]5[C@@H]([C@@H]([C@H]([C@@H](O5)C)OC)OC)OC)C)O[C@H]6CC[C@@H]([C@H](O6)C)N(C)C Chemical compound CC[C@H]1CCC[C@@H]([C@H](C(=O)C2=C[C@H]3[C@@H]4C[C@@H](C[C@H]4C(=C[C@H]3[C@@H]2CC(=O)O1)C)O[C@H]5[C@@H]([C@@H]([C@H]([C@@H](O5)C)OC)OC)OC)C)O[C@H]6CC[C@@H]([C@H](O6)C)N(C)C.CC[C@H]1CCC[C@@H]([C@H](C(=O)C2=C[C@H]3[C@@H]4C[C@@H](C[C@H]4C=C[C@H]3C2CC(=O)O1)O[C@H]5[C@@H]([C@@H]([C@H]([C@@H](O5)C)OC)OC)OC)C)O[C@H]6CC[C@@H]([C@H](O6)C)N(C)C JFLRKDZMHNBDQS-UCQUSYKYSA-N 0.000 description 1
- 241001182720 Cacopsylla pyrisuga Species 0.000 description 1
- 229910021532 Calcite Inorganic materials 0.000 description 1
- 241001313742 Callosobruchus chinensis Species 0.000 description 1
- 241000253350 Capillaria Species 0.000 description 1
- 239000005745 Captan Substances 0.000 description 1
- 241000252229 Carassius auratus Species 0.000 description 1
- MPKTWNYCNKUVKZ-UHFFFAOYSA-N Carbamorph Chemical compound CN(C)C(=S)SCN1CCOCC1 MPKTWNYCNKUVKZ-UHFFFAOYSA-N 0.000 description 1
- TWFZGCMQGLPBSX-UHFFFAOYSA-N Carbendazim Natural products C1=CC=C2NC(NC(=O)OC)=NC2=C1 TWFZGCMQGLPBSX-UHFFFAOYSA-N 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 1
- 239000005746 Carboxin Substances 0.000 description 1
- 241001347511 Carposina sasakii Species 0.000 description 1
- 239000005973 Carvone Substances 0.000 description 1
- 244000209117 Castanea crenata Species 0.000 description 1
- 235000003801 Castanea crenata Nutrition 0.000 description 1
- 241000781521 Cavelerius Species 0.000 description 1
- 241001303562 Centrolophus niger Species 0.000 description 1
- 241000255579 Ceratitis capitata Species 0.000 description 1
- 241000630083 Ceroplastes ceriferus Species 0.000 description 1
- 241001450756 Ceroplastes rubens Species 0.000 description 1
- 241000893172 Chabertia Species 0.000 description 1
- 241000738549 Chaetocnema concinna Species 0.000 description 1
- 241001147107 Chanos Species 0.000 description 1
- 241000270617 Cheloniidae Species 0.000 description 1
- 241000426497 Chilo suppressalis Species 0.000 description 1
- REEFSLKDEDEWAO-UHFFFAOYSA-N Chloraniformethan Chemical compound ClC1=CC=C(NC(NC=O)C(Cl)(Cl)Cl)C=C1Cl REEFSLKDEDEWAO-UHFFFAOYSA-N 0.000 description 1
- 239000005886 Chlorantraniliprole Substances 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- 239000005747 Chlorothalonil Substances 0.000 description 1
- 239000005944 Chlorpyrifos Substances 0.000 description 1
- 239000005945 Chlorpyrifos-methyl Substances 0.000 description 1
- 239000005887 Chromafenozide Substances 0.000 description 1
- 241000693598 Chromatomyia horticola Species 0.000 description 1
- 241001124179 Chrysops Species 0.000 description 1
- 239000005654 Clofentezine Substances 0.000 description 1
- 241001327965 Clonorchis sinensis Species 0.000 description 1
- 241000098289 Cnaphalocrocis medinalis Species 0.000 description 1
- 241000202814 Cochliomyia hominivorax Species 0.000 description 1
- JJLJMEJHUUYSSY-UHFFFAOYSA-L Copper hydroxide Chemical compound [OH-].[OH-].[Cu+2] JJLJMEJHUUYSSY-UHFFFAOYSA-L 0.000 description 1
- 239000005750 Copper hydroxide Substances 0.000 description 1
- 239000005752 Copper oxychloride Substances 0.000 description 1
- 241001509962 Coptotermes formosanus Species 0.000 description 1
- 229920002261 Corn starch Polymers 0.000 description 1
- 241001367013 Ctenoplusia agnata Species 0.000 description 1
- 240000008067 Cucumis sativus Species 0.000 description 1
- 235000010799 Cucumis sativus var sativus Nutrition 0.000 description 1
- 241000256054 Culex <genus> Species 0.000 description 1
- 241000144210 Culex pipiens pallens Species 0.000 description 1
- 241000692095 Cuterebra Species 0.000 description 1
- 239000005889 Cyantraniliprole Substances 0.000 description 1
- 239000005754 Cyazofamid Substances 0.000 description 1
- 241000612152 Cyclamen hederifolium Species 0.000 description 1
- 239000005755 Cyflufenamid Substances 0.000 description 1
- 241001503766 Cylas formicarius Species 0.000 description 1
- 239000005756 Cymoxanil Substances 0.000 description 1
- MKZGVGOBXZJKGV-UHFFFAOYSA-N Cypendazole Chemical compound C1=CC=C2N(C(=O)NCCCCCC#N)C(NC(=O)OC)=NC2=C1 MKZGVGOBXZJKGV-UHFFFAOYSA-N 0.000 description 1
- 239000005758 Cyprodinil Substances 0.000 description 1
- KRZUZYJEQBXUIN-UHFFFAOYSA-N Cyprofuram Chemical compound ClC1=CC=CC(N(C2C(OCC2)=O)C(=O)C2CC2)=C1 KRZUZYJEQBXUIN-UHFFFAOYSA-N 0.000 description 1
- 239000005891 Cyromazine Substances 0.000 description 1
- 201000003808 Cystic echinococcosis Diseases 0.000 description 1
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 1
- RGHNJXZEOKUKBD-UHFFFAOYSA-N D-gluconic acid Natural products OCC(O)C(O)C(O)C(O)C(O)=O RGHNJXZEOKUKBD-UHFFFAOYSA-N 0.000 description 1
- 239000005644 Dazomet Substances 0.000 description 1
- 239000004287 Dehydroacetic acid Substances 0.000 description 1
- 239000005892 Deltamethrin Substances 0.000 description 1
- 241001128002 Demodex canis Species 0.000 description 1
- 241000714959 Demodex cati Species 0.000 description 1
- 241001480824 Dermacentor Species 0.000 description 1
- 241001481695 Dermanyssus gallinae Species 0.000 description 1
- 229920001353 Dextrin Polymers 0.000 description 1
- 239000004375 Dextrin Substances 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- 241000489973 Diabrotica undecimpunctata Species 0.000 description 1
- 241000489976 Diabrotica undecimpunctata howardi Species 0.000 description 1
- 241000489977 Diabrotica virgifera Species 0.000 description 1
- 241000489947 Diabrotica virgifera virgifera Species 0.000 description 1
- 241001205778 Dialeurodes citri Species 0.000 description 1
- 241000526125 Diaphorina citri Species 0.000 description 1
- WGOWCPGHOCIHBW-UHFFFAOYSA-N Dichlofenthion Chemical compound CCOP(=S)(OCC)OC1=CC=C(Cl)C=C1Cl WGOWCPGHOCIHBW-UHFFFAOYSA-N 0.000 description 1
- JDZSMXLTQNHBRF-UHFFFAOYSA-N Dichlozoline Chemical compound O=C1C(C)(C)OC(=O)N1C1=CC(Cl)=CC(Cl)=C1 JDZSMXLTQNHBRF-UHFFFAOYSA-N 0.000 description 1
- 241001147667 Dictyocaulus Species 0.000 description 1
- LWLJUMBEZJHXHV-UHFFFAOYSA-N Dienochlor Chemical compound ClC1=C(Cl)C(Cl)=C(Cl)C1(Cl)C1(Cl)C(Cl)=C(Cl)C(Cl)=C1Cl LWLJUMBEZJHXHV-UHFFFAOYSA-N 0.000 description 1
- 239000005760 Difenoconazole Substances 0.000 description 1
- 239000005893 Diflubenzuron Substances 0.000 description 1
- IIUZTXTZRGLYTI-UHFFFAOYSA-N Dihydrogriseofulvin Natural products COC1CC(=O)CC(C)C11C(=O)C(C(OC)=CC(OC)=C2Cl)=C2O1 IIUZTXTZRGLYTI-UHFFFAOYSA-N 0.000 description 1
- ZAFNJMIOTHYJRJ-UHFFFAOYSA-N Diisopropyl ether Chemical compound CC(C)OC(C)C ZAFNJMIOTHYJRJ-UHFFFAOYSA-N 0.000 description 1
- 239000005947 Dimethoate Substances 0.000 description 1
- 239000005761 Dimethomorph Substances 0.000 description 1
- 239000005762 Dimoxystrobin Substances 0.000 description 1
- HDWLUGYOLUHEMN-UHFFFAOYSA-N Dinobuton Chemical compound CCC(C)C1=CC([N+]([O-])=O)=CC([N+]([O-])=O)=C1OC(=O)OC(C)C HDWLUGYOLUHEMN-UHFFFAOYSA-N 0.000 description 1
- ODOSVAWLEGXOPB-UHFFFAOYSA-N Dinocton 6 Chemical compound COC(=O)OC1=C(CCCCCC(C)C)C=C([N+]([O-])=O)C=C1[N+]([O-])=O ODOSVAWLEGXOPB-UHFFFAOYSA-N 0.000 description 1
- 241001137876 Diphyllobothrium Species 0.000 description 1
- 241000866683 Diphyllobothrium latum Species 0.000 description 1
- 241000935792 Dipylidium caninum Species 0.000 description 1
- 241000243990 Dirofilaria Species 0.000 description 1
- MTBZIGHNGSTDJV-UHFFFAOYSA-N Ditalimfos Chemical compound C1=CC=C2C(=O)N(P(=S)(OCC)OCC)C(=O)C2=C1 MTBZIGHNGSTDJV-UHFFFAOYSA-N 0.000 description 1
- 239000005764 Dithianon Substances 0.000 description 1
- 239000005766 Dodine Substances 0.000 description 1
- 241000544229 Dolycoris baccarum Species 0.000 description 1
- 241001319084 Dracunculidae Species 0.000 description 1
- OOTHTARUZHONSW-LCYFTJDESA-N Drazoxolon Chemical compound CC1=NOC(=O)\C1=N/NC1=CC=CC=C1Cl OOTHTARUZHONSW-LCYFTJDESA-N 0.000 description 1
- 241001446133 Drosicha corpulenta Species 0.000 description 1
- 206010059866 Drug resistance Diseases 0.000 description 1
- 241001220350 Dryocosmus kuriphilus Species 0.000 description 1
- AIGRXSNSLVJMEA-UHFFFAOYSA-N EPN Chemical compound C=1C=CC=CC=1P(=S)(OCC)OC1=CC=C([N+]([O-])=O)C=C1 AIGRXSNSLVJMEA-UHFFFAOYSA-N 0.000 description 1
- 239000001692 EU approved anti-caking agent Substances 0.000 description 1
- 241000244170 Echinococcus granulosus Species 0.000 description 1
- 241000244163 Echinococcus multilocularis Species 0.000 description 1
- 241001566624 Eciton Species 0.000 description 1
- 241001566614 Eciton burchellii Species 0.000 description 1
- 241000223931 Eimeria acervulina Species 0.000 description 1
- 241000499566 Eimeria brunetti Species 0.000 description 1
- 241000223934 Eimeria maxima Species 0.000 description 1
- 241000499563 Eimeria necatrix Species 0.000 description 1
- 241000146360 Eimeria ovinoidalis Species 0.000 description 1
- 241000223932 Eimeria tenella Species 0.000 description 1
- LVGKNOAMLMIIKO-UHFFFAOYSA-N Elaidinsaeure-aethylester Natural products CCCCCCCCC=CCCCCCCCC(=O)OCC LVGKNOAMLMIIKO-UHFFFAOYSA-N 0.000 description 1
- YUGWDVYLFSETPE-JLHYYAGUSA-N Empenthrin Chemical compound CC\C=C(/C)C(C#C)OC(=O)C1C(C=C(C)C)C1(C)C YUGWDVYLFSETPE-JLHYYAGUSA-N 0.000 description 1
- 241000995023 Empoasca Species 0.000 description 1
- 241001222563 Empoasca onukii Species 0.000 description 1
- 241000087267 Empoasca sakaii Species 0.000 description 1
- 241000224432 Entamoeba histolytica Species 0.000 description 1
- 241000336797 Eoeurysa flavocapitata Species 0.000 description 1
- 241001518474 Epiacanthus Species 0.000 description 1
- 241000462639 Epilachna varivestis Species 0.000 description 1
- 241000459130 Epirhynchites heros Species 0.000 description 1
- 239000005767 Epoxiconazole Substances 0.000 description 1
- 241000260853 Epuraea terminalis Species 0.000 description 1
- 241001558857 Eriophyes Species 0.000 description 1
- 239000005895 Esfenvalerate Substances 0.000 description 1
- FNELVJVBIYMIMC-UHFFFAOYSA-N Ethiprole Chemical compound N1=C(C#N)C(S(=O)CC)=C(N)N1C1=C(Cl)C=C(C(F)(F)F)C=C1Cl FNELVJVBIYMIMC-UHFFFAOYSA-N 0.000 description 1
- 239000004258 Ethoxyquin Substances 0.000 description 1
- 241001486250 Etiella zinckenella Species 0.000 description 1
- 239000005896 Etofenprox Substances 0.000 description 1
- 239000005897 Etoxazole Substances 0.000 description 1
- 239000005769 Etridiazole Substances 0.000 description 1
- FGIWFCGDPUIBEZ-UHFFFAOYSA-N Etrimfos Chemical compound CCOC1=CC(OP(=S)(OC)OC)=NC(CC)=N1 FGIWFCGDPUIBEZ-UHFFFAOYSA-N 0.000 description 1
- 241000020674 Euhadra peliomphala Species 0.000 description 1
- 241000416536 Euproctis pseudoconspersa Species 0.000 description 1
- 235000014066 European mistletoe Nutrition 0.000 description 1
- 241000825563 Eurydema Species 0.000 description 1
- 241000144291 Eurytrema pancreaticum Species 0.000 description 1
- 241000073845 Eysarcoris aeneus Species 0.000 description 1
- 241000283633 Eysarcoris lewisi Species 0.000 description 1
- 241000659205 Eysarcoris ventralis Species 0.000 description 1
- 241000204939 Fasciola gigantica Species 0.000 description 1
- 239000005774 Fenamidone Substances 0.000 description 1
- IWDQPCIQCXRBQP-UHFFFAOYSA-M Fenaminosulf Chemical compound [Na+].CN(C)C1=CC=C(N=NS([O-])(=O)=O)C=C1 IWDQPCIQCXRBQP-UHFFFAOYSA-M 0.000 description 1
- 239000005958 Fenamiphos (aka phenamiphos) Substances 0.000 description 1
- OQOULEWDDRNBSG-UHFFFAOYSA-N Fenapanil Chemical compound C=1C=CC=CC=1C(CCCC)(C#N)CN1C=CN=C1 OQOULEWDDRNBSG-UHFFFAOYSA-N 0.000 description 1
- 239000005656 Fenazaquin Substances 0.000 description 1
- 239000005775 Fenbuconazole Substances 0.000 description 1
- 239000005776 Fenhexamid Substances 0.000 description 1
- AYBALPYBYZFKDS-OLZOCXBDSA-N Fenitropan Chemical compound CC(=O)OC[C@@H]([N+]([O-])=O)[C@@H](OC(C)=O)C1=CC=CC=C1 AYBALPYBYZFKDS-OLZOCXBDSA-N 0.000 description 1
- 239000005777 Fenpropidin Substances 0.000 description 1
- 239000005778 Fenpropimorph Substances 0.000 description 1
- 239000005657 Fenpyroximate Substances 0.000 description 1
- PNVJTZOFSHSLTO-UHFFFAOYSA-N Fenthion Chemical compound COP(=S)(OC)OC1=CC=C(SC)C(C)=C1 PNVJTZOFSHSLTO-UHFFFAOYSA-N 0.000 description 1
- 241000567920 Filariidae Species 0.000 description 1
- 241000669058 Fiorinia theae Species 0.000 description 1
- 239000005899 Fipronil Substances 0.000 description 1
- 239000005900 Flonicamid Substances 0.000 description 1
- 239000005780 Fluazinam Substances 0.000 description 1
- 239000005901 Flubendiamide Substances 0.000 description 1
- 239000005781 Fludioxonil Substances 0.000 description 1
- 239000005782 Fluopicolide Substances 0.000 description 1
- 239000005783 Fluopyram Substances 0.000 description 1
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 1
- LXMQMMSGERCRSU-UHFFFAOYSA-N Fluotrimazole Chemical compound FC(F)(F)C1=CC=CC(C(C=2C=CC=CC=2)(C=2C=CC=CC=2)N2N=CN=C2)=C1 LXMQMMSGERCRSU-UHFFFAOYSA-N 0.000 description 1
- 239000005784 Fluoxastrobin Substances 0.000 description 1
- 239000005902 Flupyradifurone Substances 0.000 description 1
- 239000005785 Fluquinconazole Substances 0.000 description 1
- 239000005786 Flutolanil Substances 0.000 description 1
- 239000005787 Flutriafol Substances 0.000 description 1
- 239000005788 Fluxapyroxad Substances 0.000 description 1
- 239000005789 Folpet Substances 0.000 description 1
- 239000005948 Formetanate Substances 0.000 description 1
- AIKKULXCBHRFOS-UHFFFAOYSA-N Formothion Chemical compound COP(=S)(OC)SCC(=O)N(C)C=O AIKKULXCBHRFOS-UHFFFAOYSA-N 0.000 description 1
- 239000005959 Fosthiazate Substances 0.000 description 1
- RHJOIOVESMTJEK-UHFFFAOYSA-N Fosthietan Chemical compound CCOP(=O)(OCC)N=C1SCS1 RHJOIOVESMTJEK-UHFFFAOYSA-N 0.000 description 1
- 241000365767 Frankliniella intonsa Species 0.000 description 1
- 241000927584 Frankliniella occidentalis Species 0.000 description 1
- 239000005715 Fructose Substances 0.000 description 1
- 229930091371 Fructose Natural products 0.000 description 1
- RFSUNEUAIZKAJO-ARQDHWQXSA-N Fructose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O RFSUNEUAIZKAJO-ARQDHWQXSA-N 0.000 description 1
- 239000005791 Fuberidazole Substances 0.000 description 1
- QTDRLOKFLJJHTG-UHFFFAOYSA-N Furmecyclox Chemical compound C1=C(C)OC(C)=C1C(=O)N(OC)C1CCCCC1 QTDRLOKFLJJHTG-UHFFFAOYSA-N 0.000 description 1
- 241000982383 Gametis jucunda Species 0.000 description 1
- 239000005903 Gamma-cyhalothrin Substances 0.000 description 1
- 241001660203 Gasterophilus Species 0.000 description 1
- 241000282816 Giraffa camelopardalis Species 0.000 description 1
- 241000257323 Glossina morsitans Species 0.000 description 1
- 241000257334 Glossina palpalis Species 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- 244000068988 Glycine max Species 0.000 description 1
- 235000010469 Glycine max Nutrition 0.000 description 1
- DYMNZCGFRHLNMT-UHFFFAOYSA-N Glyodin Chemical compound CC(O)=O.CCCCCCCCCCCCCCCCCC1=NCCN1 DYMNZCGFRHLNMT-UHFFFAOYSA-N 0.000 description 1
- 241000751601 Glyphodes pyloalis Species 0.000 description 1
- 241001441330 Grapholita molesta Species 0.000 description 1
- 241000578422 Graphosoma lineatum Species 0.000 description 1
- UXWOXTQWVMFRSE-UHFFFAOYSA-N Griseoviridin Natural products O=C1OC(C)CC=C(C(NCC=CC=CC(O)CC(O)C2)=O)SCC1NC(=O)C1=COC2=N1 UXWOXTQWVMFRSE-UHFFFAOYSA-N 0.000 description 1
- 241001568505 Gryllotalpa orientalis Species 0.000 description 1
- 241001423309 Haemaphysalis campanulata Species 0.000 description 1
- 241001053172 Haemaphysalis flava Species 0.000 description 1
- 241000179420 Haemaphysalis longicornis Species 0.000 description 1
- 241000257232 Haematobia irritans Species 0.000 description 1
- 241000894055 Haematopinus eurysternus Species 0.000 description 1
- 241000670091 Haematopinus suis Species 0.000 description 1
- 241000243976 Haemonchus Species 0.000 description 1
- YDNLKBDXQCHOTH-UHFFFAOYSA-N Halacrinate Chemical compound C1=CC=C2C(Cl)=CC(Br)=C(OC(=O)C=C)C2=N1 YDNLKBDXQCHOTH-UHFFFAOYSA-N 0.000 description 1
- 241000663953 Halticus Species 0.000 description 1
- 241000896365 Haplothrips statices Species 0.000 description 1
- 241001369160 Helcystogramma Species 0.000 description 1
- 241000255990 Helicoverpa Species 0.000 description 1
- 241001147381 Helicoverpa armigera Species 0.000 description 1
- 241000256244 Heliothis virescens Species 0.000 description 1
- 241001581044 Hellula undalis Species 0.000 description 1
- 241001299253 Henosepilachna vigintioctopunctata Species 0.000 description 1
- 241000306498 Heptophylla picea Species 0.000 description 1
- 241000920462 Heterakis Species 0.000 description 1
- 241001480224 Heterodera Species 0.000 description 1
- 241000498254 Heterodera glycines Species 0.000 description 1
- CMBYOWLFQAFZCP-UHFFFAOYSA-N Hexyl dodecanoate Chemical compound CCCCCCCCCCCC(=O)OCCCCCC CMBYOWLFQAFZCP-UHFFFAOYSA-N 0.000 description 1
- 239000005661 Hexythiazox Substances 0.000 description 1
- 241000244156 Hydatigera taeniaeformis Species 0.000 description 1
- 239000004354 Hydroxyethyl cellulose Substances 0.000 description 1
- 229920000663 Hydroxyethyl cellulose Polymers 0.000 description 1
- 241000244166 Hymenolepis diminuta Species 0.000 description 1
- 239000005794 Hymexazol Substances 0.000 description 1
- 241001531327 Hyphantria cunea Species 0.000 description 1
- 241000257176 Hypoderma <fly> Species 0.000 description 1
- 241000543830 Hypoderma bovis Species 0.000 description 1
- 241000257174 Hypoderma lineatum Species 0.000 description 1
- 241001058150 Icerya purchasi Species 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 239000005795 Imazalil Substances 0.000 description 1
- 239000005906 Imidacloprid Substances 0.000 description 1
- FKWDSATZSMJRLC-UHFFFAOYSA-N Iminoctadine acetate Chemical compound CC([O-])=O.CC([O-])=O.CC([O-])=O.NC([NH3+])=NCCCCCCCC[NH2+]CCCCCCCCN=C(N)[NH3+] FKWDSATZSMJRLC-UHFFFAOYSA-N 0.000 description 1
- 239000005796 Ipconazole Substances 0.000 description 1
- 239000005867 Iprodione Substances 0.000 description 1
- 239000005797 Iprovalicarb Substances 0.000 description 1
- XRHGWAGWAHHFLF-UHFFFAOYSA-N Isazofos Chemical compound CCOP(=S)(OCC)OC=1N=C(Cl)N(C(C)C)N=1 XRHGWAGWAHHFLF-UHFFFAOYSA-N 0.000 description 1
- 239000005799 Isopyrazam Substances 0.000 description 1
- 240000007049 Juglans regia Species 0.000 description 1
- 235000009496 Juglans regia Nutrition 0.000 description 1
- 239000005909 Kieselgur Substances 0.000 description 1
- 241001467800 Knemidokoptes Species 0.000 description 1
- 239000005800 Kresoxim-methyl Substances 0.000 description 1
- NWUWYYSKZYIQAE-ZBFHGGJFSA-N L-(R)-iprovalicarb Chemical compound CC(C)OC(=O)N[C@@H](C(C)C)C(=O)N[C@H](C)C1=CC=C(C)C=C1 NWUWYYSKZYIQAE-ZBFHGGJFSA-N 0.000 description 1
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 1
- 239000005717 Laminarin Substances 0.000 description 1
- 229920001543 Laminarin Polymers 0.000 description 1
- 241000981121 Leguminivora glycinivorella Species 0.000 description 1
- 241000222722 Leishmania <genus> Species 0.000 description 1
- 241001029692 Lemyra imparilis Species 0.000 description 1
- 241001261797 Leptoconops Species 0.000 description 1
- 241000284249 Leptocorisa chinensis Species 0.000 description 1
- 241000006351 Leucophyllum frutescens Species 0.000 description 1
- 241000389333 Liceaceae Species 0.000 description 1
- 229920001732 Lignosulfonate Polymers 0.000 description 1
- 241001523405 Limax Species 0.000 description 1
- 241001113946 Linognathus vituli Species 0.000 description 1
- 241000562507 Liphyra brassolis Species 0.000 description 1
- 241000594036 Liriomyza Species 0.000 description 1
- 241000594033 Liriomyza bryoniae Species 0.000 description 1
- 241000675914 Liriomyza chinensis Species 0.000 description 1
- 241000594031 Liriomyza sativae Species 0.000 description 1
- 241000966204 Lissorhoptrus oryzophilus Species 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- 241000254022 Locusta migratoria Species 0.000 description 1
- 241000131091 Lucanus cervus Species 0.000 description 1
- 241000257162 Lucilia <blowfly> Species 0.000 description 1
- 241000501345 Lygus lineolaris Species 0.000 description 1
- 241001190211 Lyonetia Species 0.000 description 1
- 241001581015 Lyonetia clerkella Species 0.000 description 1
- 241001414659 Macrosteles Species 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- 239000005949 Malathion Substances 0.000 description 1
- 241000555303 Mamestra brassicae Species 0.000 description 1
- 239000005802 Mancozeb Substances 0.000 description 1
- 241001646125 Mandarina Species 0.000 description 1
- 239000005803 Mandestrobin Substances 0.000 description 1
- 239000005804 Mandipropamid Substances 0.000 description 1
- 229930195725 Mannitol Natural products 0.000 description 1
- 241001296368 Matsumuraeses phaseoli Species 0.000 description 1
- ZCAHBOURBFRXOT-UHFFFAOYSA-N Mecarbinzid Chemical compound C1=CC=C2N(C(=O)NCCSC)C(NC(=O)OC)=NC2=C1 ZCAHBOURBFRXOT-UHFFFAOYSA-N 0.000 description 1
- 241001318725 Meghimatium bilineatum Species 0.000 description 1
- 241001108871 Melanotus tamsuyensis Species 0.000 description 1
- 241000243786 Meloidogyne incognita Species 0.000 description 1
- 241000243785 Meloidogyne javanica Species 0.000 description 1
- 241000292449 Menacanthus stramineus Species 0.000 description 1
- 239000005805 Mepanipyrim Substances 0.000 description 1
- 239000005806 Meptyldinocap Substances 0.000 description 1
- 240000001463 Mertensia maritima Species 0.000 description 1
- 235000014272 Mertensia maritima var. asiatica Nutrition 0.000 description 1
- 235000003168 Mertensia maritima var. maritima Nutrition 0.000 description 1
- 241001481698 Mesostigmata Species 0.000 description 1
- 239000005914 Metaflumizone Substances 0.000 description 1
- MIFOMMKAVSCNKQ-HWIUFGAZSA-N Metaflumizone Chemical compound C1=CC(OC(F)(F)F)=CC=C1NC(=O)N\N=C(C=1C=C(C=CC=1)C(F)(F)F)\CC1=CC=C(C#N)C=C1 MIFOMMKAVSCNKQ-HWIUFGAZSA-N 0.000 description 1
- 239000005807 Metalaxyl Substances 0.000 description 1
- 239000002169 Metam Substances 0.000 description 1
- 239000005868 Metconazole Substances 0.000 description 1
- NTAHCMPOMKHKEU-AATRIKPKSA-N Methacrifos Chemical compound COC(=O)C(\C)=C\OP(=S)(OC)OC NTAHCMPOMKHKEU-AATRIKPKSA-N 0.000 description 1
- 239000005916 Methomyl Substances 0.000 description 1
- 239000005917 Methoxyfenozide Substances 0.000 description 1
- 239000005809 Metiram Substances 0.000 description 1
- 239000005810 Metrafenone Substances 0.000 description 1
- UDSJPFPDKCMYBD-UHFFFAOYSA-N Metsulfovax Chemical compound S1C(C)=NC(C)=C1C(=O)NC1=CC=CC=C1 UDSJPFPDKCMYBD-UHFFFAOYSA-N 0.000 description 1
- 239000005918 Milbemectin Substances 0.000 description 1
- 241000237852 Mollusca Species 0.000 description 1
- 241001626715 Moniezia benedeni Species 0.000 description 1
- 241001442207 Monochamus alternatus Species 0.000 description 1
- 241000736258 Myrmecia <insect> Species 0.000 description 1
- 241000409991 Mythimna separata Species 0.000 description 1
- 241000237536 Mytilus edulis Species 0.000 description 1
- IUOKJNROJISWRO-UHFFFAOYSA-N N-(2-cyano-3-methylbutan-2-yl)-2-(2,4-dichlorophenoxy)propanamide Chemical compound CC(C)C(C)(C#N)NC(=O)C(C)OC1=CC=C(Cl)C=C1Cl IUOKJNROJISWRO-UHFFFAOYSA-N 0.000 description 1
- XFOXDUJCOHBXRC-UHFFFAOYSA-N N-Ethyl-N-methyl-4-(trifluoromethyl)-2-(3,4-dimethoxyphenyl)benzamide Chemical compound CCN(C)C(=O)C1=CC=C(C(F)(F)F)C=C1C1=CC=C(OC)C(OC)=C1 XFOXDUJCOHBXRC-UHFFFAOYSA-N 0.000 description 1
- CCCGEKHKTPTUHJ-UHFFFAOYSA-N N-[9-(dichloromethylene)-1,2,3,4-tetrahydro-1,4-methanonaphthalen-5-yl]-3-(difluoromethyl)-1-methylpyrazole-4-carboxamide Chemical compound FC(F)C1=NN(C)C=C1C(=O)NC1=CC=CC2=C1C1CCC2C1=C(Cl)Cl CCCGEKHKTPTUHJ-UHFFFAOYSA-N 0.000 description 1
- NQRFDNJEBWAUBL-UHFFFAOYSA-N N-[cyano(2-thienyl)methyl]-4-ethyl-2-(ethylamino)-1,3-thiazole-5-carboxamide Chemical compound S1C(NCC)=NC(CC)=C1C(=O)NC(C#N)C1=CC=CS1 NQRFDNJEBWAUBL-UHFFFAOYSA-N 0.000 description 1
- SJRJJKPEHAURKC-UHFFFAOYSA-N N-methylmorpholine Substances CN1CCOCC1 SJRJJKPEHAURKC-UHFFFAOYSA-N 0.000 description 1
- XQJQCBDIXRIYRP-UHFFFAOYSA-N N-{2-[1,1'-bi(cyclopropyl)-2-yl]phenyl}-3-(difluoromethyl)-1-methyl-1pyrazole-4-carboxamide Chemical compound FC(F)C1=NN(C)C=C1C(=O)NC1=CC=CC=C1C1C(C2CC2)C1 XQJQCBDIXRIYRP-UHFFFAOYSA-N 0.000 description 1
- 238000005481 NMR spectroscopy Methods 0.000 description 1
- DDUHZTYCFQRHIY-UHFFFAOYSA-N Negwer: 6874 Natural products COC1=CC(=O)CC(C)C11C(=O)C(C(OC)=CC(OC)=C2Cl)=C2O1 DDUHZTYCFQRHIY-UHFFFAOYSA-N 0.000 description 1
- 241001137882 Nematodirus Species 0.000 description 1
- 241000407918 Neodiprion sertifer Species 0.000 description 1
- 241000358422 Nephotettix cincticeps Species 0.000 description 1
- 241001521166 Nezara antennata Species 0.000 description 1
- 241001671709 Nezara viridula Species 0.000 description 1
- 244000061176 Nicotiana tabacum Species 0.000 description 1
- 235000002637 Nicotiana tabacum Nutrition 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- VJAWBEFMCIINFU-UHFFFAOYSA-N Nitrothal-isopropyl Chemical compound CC(C)OC(=O)C1=CC(C(=O)OC(C)C)=CC([N+]([O-])=O)=C1 VJAWBEFMCIINFU-UHFFFAOYSA-N 0.000 description 1
- 241001036422 Nosopsyllus fasciatus Species 0.000 description 1
- 241000562094 Notoedres cati Species 0.000 description 1
- JKZRQYDDXKVCHF-UHFFFAOYSA-N O.O.[W+6].[Na+] Chemical compound O.O.[W+6].[Na+] JKZRQYDDXKVCHF-UHFFFAOYSA-N 0.000 description 1
- RYJURILISJOAHG-UHFFFAOYSA-N OC(=O)c1cn2ccc(I)cc2n1 Chemical compound OC(=O)c1cn2ccc(I)cc2n1 RYJURILISJOAHG-UHFFFAOYSA-N 0.000 description 1
- 241000238633 Odonata Species 0.000 description 1
- 241000866536 Odontotermes formosanus Species 0.000 description 1
- 241000510960 Oesophagostomum Species 0.000 description 1
- 241001634934 Oligonychus modestus Species 0.000 description 1
- 241000243981 Onchocerca Species 0.000 description 1
- 241000371089 Orgyia thyellina Species 0.000 description 1
- BPQQTUXANYXVAA-UHFFFAOYSA-N Orthosilicate Chemical compound [O-][Si]([O-])([O-])[O-] BPQQTUXANYXVAA-UHFFFAOYSA-N 0.000 description 1
- 241000243795 Ostertagia Species 0.000 description 1
- 241000346285 Ostrinia furnacalis Species 0.000 description 1
- 241000532856 Otiorhynchus sulcatus Species 0.000 description 1
- 239000005812 Oxathiapiprolin Substances 0.000 description 1
- YXLXNENXOJSQEI-UHFFFAOYSA-L Oxine-copper Chemical compound [Cu+2].C1=CN=C2C([O-])=CC=CC2=C1.C1=CN=C2C([O-])=CC=CC2=C1 YXLXNENXOJSQEI-UHFFFAOYSA-L 0.000 description 1
- KYGZCKSPAKDVKC-UHFFFAOYSA-N Oxolinic acid Chemical compound C1=C2N(CC)C=C(C(O)=O)C(=O)C2=CC2=C1OCO2 KYGZCKSPAKDVKC-UHFFFAOYSA-N 0.000 description 1
- 241000382928 Oxya Species 0.000 description 1
- JAYZFNIOOYPIAH-UHFFFAOYSA-N Oxydeprofos Chemical compound CCS(=O)CC(C)SP(=O)(OC)OC JAYZFNIOOYPIAH-UHFFFAOYSA-N 0.000 description 1
- 241000904715 Oxyuris Species 0.000 description 1
- 241000751898 Paederus fuscipes Species 0.000 description 1
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 1
- 241001441425 Pandemis heparana Species 0.000 description 1
- 241000488581 Panonychus citri Species 0.000 description 1
- 241001443862 Panonychus mori Species 0.000 description 1
- 241000488583 Panonychus ulmi Species 0.000 description 1
- 241000282376 Panthera tigris Species 0.000 description 1
- 239000005662 Paraffin oil Substances 0.000 description 1
- 241001480234 Paragonimus westermani Species 0.000 description 1
- 241000497111 Paralobesia viteana Species 0.000 description 1
- 241000459456 Parapediasia teterrellus Species 0.000 description 1
- 241000244187 Parascaris Species 0.000 description 1
- 241001622642 Parnara bada Species 0.000 description 1
- 241000237509 Patinopecten sp. Species 0.000 description 1
- 241000721451 Pectinophora gossypiella Species 0.000 description 1
- 241000562493 Pegomya Species 0.000 description 1
- 239000005813 Penconazole Substances 0.000 description 1
- 239000005815 Penflufen Substances 0.000 description 1
- 239000005816 Penthiopyrad Substances 0.000 description 1
- 241001510010 Periplaneta fuliginosa Species 0.000 description 1
- 241000048273 Periplaneta japonica Species 0.000 description 1
- 241000286209 Phasianidae Species 0.000 description 1
- 241001058021 Phenacoccus solani Species 0.000 description 1
- 241000255129 Phlebotominae Species 0.000 description 1
- 239000005921 Phosmet Substances 0.000 description 1
- 241001525654 Phyllocnistis citrella Species 0.000 description 1
- 241000437063 Phyllotreta striolata Species 0.000 description 1
- 241001396980 Phytonemus pallidus Species 0.000 description 1
- 239000005818 Picoxystrobin Substances 0.000 description 1
- 241000255969 Pieris brassicae Species 0.000 description 1
- 241000227425 Pieris rapae crucivora Species 0.000 description 1
- 241000476428 Piezodorus hybneri Species 0.000 description 1
- 239000005923 Pirimicarb Substances 0.000 description 1
- 239000005924 Pirimiphos-methyl Substances 0.000 description 1
- 241000224016 Plasmodium Species 0.000 description 1
- 241001089531 Plautia crossota Species 0.000 description 1
- 241000500437 Plutella xylostella Species 0.000 description 1
- 229930182764 Polyoxin Natural products 0.000 description 1
- JPFWJDMDPLEUBD-UHFFFAOYSA-N Polyoxin D Natural products OC1C(O)C(C(NC(=O)C(C(O)C(O)COC(N)=O)N)C(O)=O)OC1N1C(=O)NC(=O)C(C(O)=O)=C1 JPFWJDMDPLEUBD-UHFFFAOYSA-N 0.000 description 1
- 241000952063 Polyphagotarsonemus latus Species 0.000 description 1
- 229920001214 Polysorbate 60 Polymers 0.000 description 1
- 241000570011 Pomacea canaliculata Species 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 241000193977 Pratylenchus musicola Species 0.000 description 1
- 241000193940 Pratylenchus penetrans Species 0.000 description 1
- 241000193966 Pratylenchus vulnus Species 0.000 description 1
- ONIBWKKTOPOVIA-UHFFFAOYSA-N Proline Natural products OC(=O)C1CCCN1 ONIBWKKTOPOVIA-UHFFFAOYSA-N 0.000 description 1
- MKIMSXGUTQTKJU-UHFFFAOYSA-N Propamocarb hydrochloride Chemical compound [Cl-].CCCOC(=O)NCCC[NH+](C)C MKIMSXGUTQTKJU-UHFFFAOYSA-N 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 1
- 239000005822 Propiconazole Substances 0.000 description 1
- 239000005823 Propineb Substances 0.000 description 1
- 239000005824 Proquinazid Substances 0.000 description 1
- 241000736232 Prosimulium Species 0.000 description 1
- 239000005825 Prothioconazole Substances 0.000 description 1
- 241000027515 Psacothea hilaris Species 0.000 description 1
- 241000669297 Pseudaonidia Species 0.000 description 1
- 241000669298 Pseudaulacaspis pentagona Species 0.000 description 1
- 241000669319 Pseudaulacaspis prunicola Species 0.000 description 1
- 241000596535 Pseudococcus comstocki Species 0.000 description 1
- 241001649230 Psoroptes ovis Species 0.000 description 1
- 241001160824 Psylliodes Species 0.000 description 1
- 241000895239 Pterella Species 0.000 description 1
- 239000005925 Pymetrozine Substances 0.000 description 1
- YPCALTGLHFLNGA-UHFFFAOYSA-N Pyracarbolid Chemical compound C1CCOC(C)=C1C(=O)NC1=CC=CC=C1 YPCALTGLHFLNGA-UHFFFAOYSA-N 0.000 description 1
- 239000005869 Pyraclostrobin Substances 0.000 description 1
- 239000005926 Pyridalyl Substances 0.000 description 1
- OVZITGHGWBXFEA-UHFFFAOYSA-N Pyridinitril Chemical compound ClC1=NC(Cl)=C(C#N)C(C=2C=CC=CC=2)=C1C#N OVZITGHGWBXFEA-UHFFFAOYSA-N 0.000 description 1
- 239000005828 Pyrimethanil Substances 0.000 description 1
- 239000005829 Pyriofenone Substances 0.000 description 1
- 239000005927 Pyriproxyfen Substances 0.000 description 1
- CDEWHBGYSWJUFP-UHFFFAOYSA-N Quinacetol sulfate Chemical compound OS(O)(=O)=O.C1=CC=C2C(C(=O)C)=CC=C(O)C2=N1.C1=CC=C2C(C(=O)C)=CC=C(O)C2=N1 CDEWHBGYSWJUFP-UHFFFAOYSA-N 0.000 description 1
- 239000005831 Quinoxyfen Substances 0.000 description 1
- IWYDHOAUDWTVEP-UHFFFAOYSA-N R-2-phenyl-2-hydroxyacetic acid Natural products OC(=O)C(O)C1=CC=CC=C1 IWYDHOAUDWTVEP-UHFFFAOYSA-N 0.000 description 1
- 235000019484 Rapeseed oil Nutrition 0.000 description 1
- 241000866500 Reticulitermes speratus Species 0.000 description 1
- 241000238680 Rhipicephalus microplus Species 0.000 description 1
- 241001481696 Rhipicephalus sanguineus Species 0.000 description 1
- 244000152640 Rhipsalis cassutha Species 0.000 description 1
- 235000012300 Rhipsalis cassutha Nutrition 0.000 description 1
- 241000260845 Rhizoglyphus robini Species 0.000 description 1
- 241000982229 Rhopalus maculatus Species 0.000 description 1
- 241000133716 Riptortus clavatus Species 0.000 description 1
- 241000283984 Rodentia Species 0.000 description 1
- 241000509427 Sarcoptes scabiei Species 0.000 description 1
- 241000253973 Schistocerca gregaria Species 0.000 description 1
- 241000242678 Schistosoma Species 0.000 description 1
- 241000242683 Schistosoma haematobium Species 0.000 description 1
- 241000242680 Schistosoma mansoni Species 0.000 description 1
- 241000343234 Scirtothrips citri Species 0.000 description 1
- 239000005834 Sedaxane Substances 0.000 description 1
- 239000005835 Silthiofam Substances 0.000 description 1
- 241000256103 Simuliidae Species 0.000 description 1
- 241000256108 Simulium <genus> Species 0.000 description 1
- 241000068648 Sitodiplosis mosellana Species 0.000 description 1
- 241000254152 Sitophilus oryzae Species 0.000 description 1
- 241000254154 Sitophilus zeamais Species 0.000 description 1
- DWAQJAXMDSEUJJ-UHFFFAOYSA-M Sodium bisulfite Chemical compound [Na+].OS([O-])=O DWAQJAXMDSEUJJ-UHFFFAOYSA-M 0.000 description 1
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical class [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 1
- 239000005708 Sodium hypochlorite Substances 0.000 description 1
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 1
- 241000176086 Sogatella furcifera Species 0.000 description 1
- 244000061458 Solanum melongena Species 0.000 description 1
- 235000002597 Solanum melongena Nutrition 0.000 description 1
- 241000044147 Solenopotes capillatus Species 0.000 description 1
- 241001492664 Solenopsis <angiosperm> Species 0.000 description 1
- 241000517830 Solenopsis geminata Species 0.000 description 1
- HVUMOYIDDBPOLL-XWVZOOPGSA-N Sorbitan monostearate Chemical compound CCCCCCCCCCCCCCCCCC(=O)OC[C@@H](O)[C@H]1OC[C@H](O)[C@H]1O HVUMOYIDDBPOLL-XWVZOOPGSA-N 0.000 description 1
- 241000532887 Sphenophorus venatus Species 0.000 description 1
- 239000005929 Spinetoram Substances 0.000 description 1
- GOENIMGKWNZVDA-OAMCMWGQSA-N Spinetoram Chemical compound CO[C@@H]1[C@H](OCC)[C@@H](OC)[C@H](C)O[C@H]1OC1C[C@H]2[C@@H]3C=C4C(=O)[C@H](C)[C@@H](O[C@@H]5O[C@H](C)[C@H](CC5)N(C)C)CCC[C@H](CC)OC(=O)CC4[C@@H]3CC[C@@H]2C1 GOENIMGKWNZVDA-OAMCMWGQSA-N 0.000 description 1
- 239000005930 Spinosad Substances 0.000 description 1
- 239000005664 Spirodiclofen Substances 0.000 description 1
- 239000005665 Spiromesifen Substances 0.000 description 1
- 239000005931 Spirotetramat Substances 0.000 description 1
- 239000005837 Spiroxamine Substances 0.000 description 1
- 241000681499 Spodoptera depravata Species 0.000 description 1
- 241000256247 Spodoptera exigua Species 0.000 description 1
- 241000256251 Spodoptera frugiperda Species 0.000 description 1
- 241000405655 Stathmopoda masinissa Species 0.000 description 1
- 241000903611 Stenodema Species 0.000 description 1
- 241000775407 Stenotus rubrovittatus Species 0.000 description 1
- 241001508985 Stephanitis pyrioides Species 0.000 description 1
- 241000122932 Strongylus Species 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- 239000005934 Sulfoxaflor Substances 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- LSNNMFCWUKXFEE-UHFFFAOYSA-N Sulfurous acid Chemical compound OS(O)=O LSNNMFCWUKXFEE-UHFFFAOYSA-N 0.000 description 1
- 241001124066 Tachinidae Species 0.000 description 1
- 241000244159 Taenia saginata Species 0.000 description 1
- 241000244157 Taenia solium Species 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- 239000005839 Tebuconazole Substances 0.000 description 1
- 239000005658 Tebufenpyrad Substances 0.000 description 1
- 239000005938 Teflubenzuron Substances 0.000 description 1
- 241000896142 Teleogryllus emma Species 0.000 description 1
- 241000255588 Tephritidae Species 0.000 description 1
- 239000005840 Tetraconazole Substances 0.000 description 1
- 241001454293 Tetranychus urticae Species 0.000 description 1
- 241000223777 Theileria Species 0.000 description 1
- 239000005940 Thiacloprid Substances 0.000 description 1
- JZFICWYCTCCINF-UHFFFAOYSA-N Thiadiazin Chemical compound S=C1SC(C)NC(C)N1CCN1C(=S)SC(C)NC1C JZFICWYCTCCINF-UHFFFAOYSA-N 0.000 description 1
- 239000005941 Thiamethoxam Substances 0.000 description 1
- GNOOAFGERMHQJE-UHFFFAOYSA-N Thicyofen Chemical compound CCS(=O)C=1SC(C#N)=C(Cl)C=1C#N GNOOAFGERMHQJE-UHFFFAOYSA-N 0.000 description 1
- IRVDMKJLOCGUBJ-UHFFFAOYSA-N Thionazin Chemical compound CCOP(=S)(OCC)OC1=CN=CC=N1 IRVDMKJLOCGUBJ-UHFFFAOYSA-N 0.000 description 1
- 239000005842 Thiophanate-methyl Substances 0.000 description 1
- 239000005843 Thiram Substances 0.000 description 1
- 239000005845 Tolclofos-methyl Substances 0.000 description 1
- 241000244031 Toxocara Species 0.000 description 1
- 241000223996 Toxoplasma Species 0.000 description 1
- 239000005846 Triadimenol Substances 0.000 description 1
- 241000018137 Trialeurodes vaporariorum Species 0.000 description 1
- 239000005847 Triazoxide Substances 0.000 description 1
- 241000254113 Tribolium castaneum Species 0.000 description 1
- APQHKWPGGHMYKJ-UHFFFAOYSA-N Tributyltin oxide Chemical compound CCCC[Sn](CCCC)(CCCC)O[Sn](CCCC)(CCCC)CCCC APQHKWPGGHMYKJ-UHFFFAOYSA-N 0.000 description 1
- NHTFLYKPEGXOAN-UHFFFAOYSA-N Trichlamide Chemical compound CCCCOC(C(Cl)(Cl)Cl)NC(=O)C1=CC=CC=C1O NHTFLYKPEGXOAN-UHFFFAOYSA-N 0.000 description 1
- 241001061558 Trichomonadidae Species 0.000 description 1
- 241000255993 Trichoplusia ni Species 0.000 description 1
- 241000243797 Trichostrongylus Species 0.000 description 1
- 241001489151 Trichuris Species 0.000 description 1
- 239000005857 Trifloxystrobin Substances 0.000 description 1
- 239000005858 Triflumizole Substances 0.000 description 1
- 239000005942 Triflumuron Substances 0.000 description 1
- 241000693382 Trigonotylus caelestialium Species 0.000 description 1
- 239000007983 Tris buffer Substances 0.000 description 1
- 239000005859 Triticonazole Substances 0.000 description 1
- 235000021307 Triticum Nutrition 0.000 description 1
- 244000098338 Triticum aestivum Species 0.000 description 1
- 241000223109 Trypanosoma cruzi Species 0.000 description 1
- 241000722921 Tulipa gesneriana Species 0.000 description 1
- 241000611866 Tyrophagus putrescentiae Species 0.000 description 1
- 241000261596 Tyrophagus similis Species 0.000 description 1
- 241000669244 Unaspis euonymi Species 0.000 description 1
- 241001630065 Unaspis yanonensis Species 0.000 description 1
- 241000571986 Uncinaria Species 0.000 description 1
- 244000274883 Urtica dioica Species 0.000 description 1
- 235000009108 Urtica dioica Nutrition 0.000 description 1
- 229930195482 Validamycin Natural products 0.000 description 1
- 239000005860 Valifenalate Substances 0.000 description 1
- 241000895650 Varroa jacobsoni Species 0.000 description 1
- 241000256856 Vespidae Species 0.000 description 1
- 241001274787 Viteus Species 0.000 description 1
- 235000009754 Vitis X bourquina Nutrition 0.000 description 1
- 235000012333 Vitis X labruscana Nutrition 0.000 description 1
- 235000014787 Vitis vinifera Nutrition 0.000 description 1
- 240000006365 Vitis vinifera Species 0.000 description 1
- 241000244002 Wuchereria Species 0.000 description 1
- 241000353223 Xenopsylla cheopis Species 0.000 description 1
- 241001182329 Xylotrechus pyrrhoderus Species 0.000 description 1
- 240000008042 Zea mays Species 0.000 description 1
- 235000005824 Zea mays ssp. parviglumis Nutrition 0.000 description 1
- 235000002017 Zea mays subsp mays Nutrition 0.000 description 1
- 229920002494 Zein Polymers 0.000 description 1
- 229910021536 Zeolite Inorganic materials 0.000 description 1
- 241001136529 Zeugodacus cucurbitae Species 0.000 description 1
- 239000005870 Ziram Substances 0.000 description 1
- 239000005863 Zoxamide Substances 0.000 description 1
- VXSIXFKKSNGRRO-MXOVTSAMSA-N [(1s)-2-methyl-4-oxo-3-[(2z)-penta-2,4-dienyl]cyclopent-2-en-1-yl] (1r,3r)-2,2-dimethyl-3-(2-methylprop-1-enyl)cyclopropane-1-carboxylate;[(1s)-2-methyl-4-oxo-3-[(2z)-penta-2,4-dienyl]cyclopent-2-en-1-yl] (1r,3r)-3-[(e)-3-methoxy-2-methyl-3-oxoprop-1-enyl Chemical class CC1(C)[C@H](C=C(C)C)[C@H]1C(=O)O[C@@H]1C(C)=C(C\C=C/C=C)C(=O)C1.CC1(C)[C@H](/C=C(\C)C(=O)OC)[C@H]1C(=O)O[C@@H]1C(C)=C(C\C=C/C=C)C(=O)C1 VXSIXFKKSNGRRO-MXOVTSAMSA-N 0.000 description 1
- QQODLKZGRKWIFG-RUTXASTPSA-N [(R)-cyano-(4-fluoro-3-phenoxyphenyl)methyl] (1S)-3-(2,2-dichloroethenyl)-2,2-dimethylcyclopropane-1-carboxylate Chemical compound CC1(C)C(C=C(Cl)Cl)[C@@H]1C(=O)O[C@@H](C#N)C1=CC=C(F)C(OC=2C=CC=CC=2)=C1 QQODLKZGRKWIFG-RUTXASTPSA-N 0.000 description 1
- FZSVSABTBYGOQH-XFFZJAGNSA-N [(e)-(3,3-dimethyl-1-methylsulfanylbutan-2-ylidene)amino] n-methylcarbamate Chemical compound CNC(=O)O\N=C(C(C)(C)C)\CSC FZSVSABTBYGOQH-XFFZJAGNSA-N 0.000 description 1
- BZMIHNKNQJJVRO-LVZFUZTISA-N [(e)-c-(3-chloro-2,6-dimethoxyphenyl)-n-ethoxycarbonimidoyl] benzoate Chemical compound COC=1C=CC(Cl)=C(OC)C=1C(=N/OCC)\OC(=O)C1=CC=CC=C1 BZMIHNKNQJJVRO-LVZFUZTISA-N 0.000 description 1
- KAATUXNTWXVJKI-QPIRBTGLSA-N [(s)-cyano-(3-phenoxyphenyl)methyl] 3-(2,2-dichloroethenyl)-2,2-dimethylcyclopropane-1-carboxylate Chemical compound CC1(C)C(C=C(Cl)Cl)C1C(=O)O[C@H](C#N)C1=CC=CC(OC=2C=CC=CC=2)=C1 KAATUXNTWXVJKI-QPIRBTGLSA-N 0.000 description 1
- FSAVDKDHPDSCTO-WQLSENKSSA-N [(z)-2-chloro-1-(2,4-dichlorophenyl)ethenyl] diethyl phosphate Chemical compound CCOP(=O)(OCC)O\C(=C/Cl)C1=CC=C(Cl)C=C1Cl FSAVDKDHPDSCTO-WQLSENKSSA-N 0.000 description 1
- QSGNQELHULIMSJ-POHAHGRESA-N [(z)-2-chloro-1-(2,4-dichlorophenyl)ethenyl] dimethyl phosphate Chemical compound COP(=O)(OC)O\C(=C/Cl)C1=CC=C(Cl)C=C1Cl QSGNQELHULIMSJ-POHAHGRESA-N 0.000 description 1
- KVIZNNVXXNFLMU-AIIUZBJTSA-N [2,3,5,6-tetrafluoro-4-(methoxymethyl)phenyl]methyl (1r,3r)-2,2-dimethyl-3-[(e)-prop-1-enyl]cyclopropane-1-carboxylate Chemical compound FC1=C(F)C(COC)=C(F)C(F)=C1COC(=O)[C@H]1C(C)(C)[C@@H]1\C=C\C KVIZNNVXXNFLMU-AIIUZBJTSA-N 0.000 description 1
- DPJITPZADZSLBP-PIPQINALSA-N [2,3,5,6-tetrafluoro-4-(methoxymethyl)phenyl]methyl (1r,3r)-3-[(e)-2-cyanoprop-1-enyl]-2,2-dimethylcyclopropane-1-carboxylate Chemical compound FC1=C(F)C(COC)=C(F)C(F)=C1COC(=O)[C@H]1C(C)(C)[C@@H]1\C=C(/C)C#N DPJITPZADZSLBP-PIPQINALSA-N 0.000 description 1
- MWFQAAWRPDRKDG-KOLCDFICSA-N [2,3,5,6-tetrafluoro-4-(methoxymethyl)phenyl]methyl (1r,3s)-3-(2,2-dichloroethenyl)-2,2-dimethylcyclopropane-1-carboxylate Chemical compound FC1=C(F)C(COC)=C(F)C(F)=C1COC(=O)[C@H]1C(C)(C)[C@@H]1C=C(Cl)Cl MWFQAAWRPDRKDG-KOLCDFICSA-N 0.000 description 1
- OOWCJRMYMAMSOH-UHFFFAOYSA-N [2,3,5,6-tetrafluoro-4-(methoxymethyl)phenyl]methyl 2,2-dimethyl-3-(2-methylprop-1-enyl)cyclopropane-1-carboxylate Chemical compound FC1=C(F)C(COC)=C(F)C(F)=C1COC(=O)C1C(C)(C)C1C=C(C)C OOWCJRMYMAMSOH-UHFFFAOYSA-N 0.000 description 1
- INISTDXBRIBGOC-CGAIIQECSA-N [cyano-(3-phenoxyphenyl)methyl] (2s)-2-[2-chloro-4-(trifluoromethyl)anilino]-3-methylbutanoate Chemical compound N([C@@H](C(C)C)C(=O)OC(C#N)C=1C=C(OC=2C=CC=CC=2)C=CC=1)C1=CC=C(C(F)(F)F)C=C1Cl INISTDXBRIBGOC-CGAIIQECSA-N 0.000 description 1
- YXWCBRDRVXHABN-JCMHNJIXSA-N [cyano-(4-fluoro-3-phenoxyphenyl)methyl] 3-[(z)-2-chloro-2-(4-chlorophenyl)ethenyl]-2,2-dimethylcyclopropane-1-carboxylate Chemical compound C=1C=C(F)C(OC=2C=CC=CC=2)=CC=1C(C#N)OC(=O)C1C(C)(C)C1\C=C(/Cl)C1=CC=C(Cl)C=C1 YXWCBRDRVXHABN-JCMHNJIXSA-N 0.000 description 1
- ZJOCMGQZWCTWNX-UHFFFAOYSA-N [ethoxy(ethylsulfanyl)phosphoryl]oxycyclohexane Chemical compound CCOP(=O)(SCC)OC1CCCCC1 ZJOCMGQZWCTWNX-UHFFFAOYSA-N 0.000 description 1
- ZVQOOHYFBIDMTQ-UHFFFAOYSA-N [methyl(oxido){1-[6-(trifluoromethyl)pyridin-3-yl]ethyl}-lambda(6)-sulfanylidene]cyanamide Chemical compound N#CN=S(C)(=O)C(C)C1=CC=C(C(F)(F)F)N=C1 ZVQOOHYFBIDMTQ-UHFFFAOYSA-N 0.000 description 1
- 229950008167 abamectin Drugs 0.000 description 1
- 230000004308 accommodation Effects 0.000 description 1
- YASYVMFAVPKPKE-UHFFFAOYSA-N acephate Chemical compound COP(=O)(SC)NC(C)=O YASYVMFAVPKPKE-UHFFFAOYSA-N 0.000 description 1
- QDRXWCAVUNHOGA-UHFFFAOYSA-N acequinocyl Chemical group C1=CC=C2C(=O)C(CCCCCCCCCCCC)=C(OC(C)=O)C(=O)C2=C1 QDRXWCAVUNHOGA-UHFFFAOYSA-N 0.000 description 1
- 150000001242 acetic acid derivatives Chemical class 0.000 description 1
- YLJLLELGHSWIDU-OKZTUQRJSA-N acetic acid;(2s,6r)-4-cyclododecyl-2,6-dimethylmorpholine Chemical compound CC(O)=O.C1[C@@H](C)O[C@@H](C)CN1C1CCCCCCCCCCC1 YLJLLELGHSWIDU-OKZTUQRJSA-N 0.000 description 1
- UELITFHSCLAHKR-UHFFFAOYSA-N acibenzolar-S-methyl Chemical group CSC(=O)C1=CC=CC2=C1SN=N2 UELITFHSCLAHKR-UHFFFAOYSA-N 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 206010000496 acne Diseases 0.000 description 1
- YLFSVIMMRPNPFK-WEQBUNFVSA-N acrinathrin Chemical compound CC1(C)[C@@H](\C=C/C(=O)OC(C(F)(F)F)C(F)(F)F)[C@H]1C(=O)O[C@H](C#N)C1=CC=CC(OC=2C=CC=CC=2)=C1 YLFSVIMMRPNPFK-WEQBUNFVSA-N 0.000 description 1
- 239000012190 activator Substances 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- 239000001361 adipic acid Substances 0.000 description 1
- 235000011037 adipic acid Nutrition 0.000 description 1
- 239000002671 adjuvant Substances 0.000 description 1
- LRZWFURXIMFONG-HRSIRGMGSA-N afidopyropen Chemical compound C([C@@]1(C)[C@H]2[C@]([C@H]3[C@@H](O)C=4C(=O)OC(=CC=4O[C@]3(C)[C@@H](O)C2)C=2C=NC=CC=2)(C)CC[C@@H]1OC(=O)C1CC1)OC(=O)C1CC1 LRZWFURXIMFONG-HRSIRGMGSA-N 0.000 description 1
- 229960000982 afoxolaner Drugs 0.000 description 1
- 239000008272 agar Substances 0.000 description 1
- GMAUQNJOSOMMHI-JXAWBTAJSA-N alanycarb Chemical compound CSC(\C)=N/OC(=O)N(C)SN(CCC(=O)OCC)CC1=CC=CC=C1 GMAUQNJOSOMMHI-JXAWBTAJSA-N 0.000 description 1
- QGLZXHRNAYXIBU-WEVVVXLNSA-N aldicarb Chemical compound CNC(=O)O\N=C\C(C)(C)SC QGLZXHRNAYXIBU-WEVVVXLNSA-N 0.000 description 1
- 229940072056 alginate Drugs 0.000 description 1
- 239000000783 alginic acid Substances 0.000 description 1
- 229960001126 alginic acid Drugs 0.000 description 1
- 150000004781 alginic acids Chemical class 0.000 description 1
- 125000001931 aliphatic group Chemical group 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 150000001336 alkenes Chemical class 0.000 description 1
- 125000002877 alkyl aryl group Chemical group 0.000 description 1
- 150000005215 alkyl ethers Chemical class 0.000 description 1
- 125000005037 alkyl phenyl group Chemical group 0.000 description 1
- 125000004390 alkyl sulfonyl group Chemical group 0.000 description 1
- 239000011717 all-trans-retinol Substances 0.000 description 1
- 235000019169 all-trans-retinol Nutrition 0.000 description 1
- 229910001583 allophane Inorganic materials 0.000 description 1
- 239000008168 almond oil Substances 0.000 description 1
- BJEPYKJPYRNKOW-UHFFFAOYSA-N alpha-hydroxysuccinic acid Natural products OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 1
- AZDRQVAHHNSJOQ-UHFFFAOYSA-N alumane Chemical class [AlH3] AZDRQVAHHNSJOQ-UHFFFAOYSA-N 0.000 description 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 235000012211 aluminium silicate Nutrition 0.000 description 1
- GGKQIOFASHYUJZ-UHFFFAOYSA-N ametoctradin Chemical compound NC1=C(CCCCCCCC)C(CC)=NC2=NC=NN21 GGKQIOFASHYUJZ-UHFFFAOYSA-N 0.000 description 1
- BREATYVWRHIPIY-UHFFFAOYSA-N amisulbrom Chemical compound CN(C)S(=O)(=O)N1C=NC(S(=O)(=O)N2C3=CC(F)=CC=C3C(Br)=C2C)=N1 BREATYVWRHIPIY-UHFFFAOYSA-N 0.000 description 1
- 229960002587 amitraz Drugs 0.000 description 1
- QXAITBQSYVNQDR-ZIOPAAQOSA-N amitraz Chemical compound C=1C=C(C)C=C(C)C=1/N=C/N(C)\C=N\C1=CC=C(C)C=C1C QXAITBQSYVNQDR-ZIOPAAQOSA-N 0.000 description 1
- QGZKDVFQNNGYKY-UHFFFAOYSA-N ammonia Natural products N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 1
- 235000019270 ammonium chloride Nutrition 0.000 description 1
- LFVGISIMTYGQHF-UHFFFAOYSA-N ammonium dihydrogen phosphate Chemical compound [NH4+].OP(O)([O-])=O LFVGISIMTYGQHF-UHFFFAOYSA-N 0.000 description 1
- 229910000387 ammonium dihydrogen phosphate Inorganic materials 0.000 description 1
- BFNBIHQBYMNNAN-UHFFFAOYSA-N ammonium sulfate Chemical compound N.N.OS(O)(=O)=O BFNBIHQBYMNNAN-UHFFFAOYSA-N 0.000 description 1
- 229910052921 ammonium sulfate Inorganic materials 0.000 description 1
- 235000011130 ammonium sulphate Nutrition 0.000 description 1
- SOIFLUNRINLCBN-UHFFFAOYSA-N ammonium thiocyanate Chemical compound [NH4+].[S-]C#N SOIFLUNRINLCBN-UHFFFAOYSA-N 0.000 description 1
- FROZIYRKKUFAOC-UHFFFAOYSA-N amobam Chemical compound N.N.SC(=S)NCCNC(S)=S FROZIYRKKUFAOC-UHFFFAOYSA-N 0.000 description 1
- IMHBYKMAHXWHRP-UHFFFAOYSA-N anilazine Chemical compound ClC1=CC=CC=C1NC1=NC(Cl)=NC(Cl)=N1 IMHBYKMAHXWHRP-UHFFFAOYSA-N 0.000 description 1
- 150000001450 anions Chemical class 0.000 description 1
- 230000002421 anti-septic effect Effects 0.000 description 1
- 239000002518 antifoaming agent Substances 0.000 description 1
- 239000007798 antifreeze agent Substances 0.000 description 1
- 230000003078 antioxidant effect Effects 0.000 description 1
- 125000005228 aryl sulfonate group Chemical group 0.000 description 1
- 235000003704 aspartic acid Nutrition 0.000 description 1
- 125000004429 atom Chemical group 0.000 description 1
- 229960000892 attapulgite Drugs 0.000 description 1
- AKNQMEBLVAMSNZ-UHFFFAOYSA-N azaconazole Chemical compound ClC1=CC(Cl)=CC=C1C1(CN2N=CN=C2)OCCO1 AKNQMEBLVAMSNZ-UHFFFAOYSA-N 0.000 description 1
- 229950000294 azaconazole Drugs 0.000 description 1
- RQVGAIADHNPSME-UHFFFAOYSA-N azinphos-ethyl Chemical group C1=CC=C2C(=O)N(CSP(=S)(OCC)OCC)N=NC2=C1 RQVGAIADHNPSME-UHFFFAOYSA-N 0.000 description 1
- CJJOSEISRRTUQB-UHFFFAOYSA-N azinphos-methyl Chemical group C1=CC=C2C(=O)N(CSP(=S)(OC)OC)N=NC2=C1 CJJOSEISRRTUQB-UHFFFAOYSA-N 0.000 description 1
- ONHBDDJJTDTLIR-UHFFFAOYSA-N azocyclotin Chemical compound C1CCCCC1[Sn](N1N=CN=C1)(C1CCCCC1)C1CCCCC1 ONHBDDJJTDTLIR-UHFFFAOYSA-N 0.000 description 1
- WFDXOXNFNRHQEC-GHRIWEEISA-N azoxystrobin Chemical compound CO\C=C(\C(=O)OC)C1=CC=CC=C1OC1=CC(OC=2C(=CC=CC=2)C#N)=NC=N1 WFDXOXNFNRHQEC-GHRIWEEISA-N 0.000 description 1
- 229940097012 bacillus thuringiensis Drugs 0.000 description 1
- 235000015173 baked goods and baking mixes Nutrition 0.000 description 1
- XEGGRYVFLWGFHI-UHFFFAOYSA-N bendiocarb Chemical compound CNC(=O)OC1=CC=CC2=C1OC(C)(C)O2 XEGGRYVFLWGFHI-UHFFFAOYSA-N 0.000 description 1
- FYZBOYWSHKHDMT-UHFFFAOYSA-N benfuracarb Chemical compound CCOC(=O)CCN(C(C)C)SN(C)C(=O)OC1=CC=CC2=C1OC(C)(C)C2 FYZBOYWSHKHDMT-UHFFFAOYSA-N 0.000 description 1
- LJOZMWRYMKECFF-UHFFFAOYSA-N benodanil Chemical compound IC1=CC=CC=C1C(=O)NC1=CC=CC=C1 LJOZMWRYMKECFF-UHFFFAOYSA-N 0.000 description 1
- RIOXQFHNBCKOKP-UHFFFAOYSA-N benomyl Chemical compound C1=CC=C2N(C(=O)NCCCC)C(NC(=O)OC)=NC2=C1 RIOXQFHNBCKOKP-UHFFFAOYSA-N 0.000 description 1
- YFXPPSKYMBTNAV-UHFFFAOYSA-N bensultap Chemical compound C=1C=CC=CC=1S(=O)(=O)SCC(N(C)C)CSS(=O)(=O)C1=CC=CC=C1 YFXPPSKYMBTNAV-UHFFFAOYSA-N 0.000 description 1
- USRKFGIXLGKMKU-ABAIWWIYSA-N benthiavalicarb-isopropyl Chemical compound C1=C(F)C=C2SC([C@@H](C)NC(=O)[C@H](C(C)C)NC(=O)OC(C)C)=NC2=C1 USRKFGIXLGKMKU-ABAIWWIYSA-N 0.000 description 1
- 229960000686 benzalkonium chloride Drugs 0.000 description 1
- SRSXLGNVWSONIS-UHFFFAOYSA-N benzenesulfonic acid Chemical compound OS(=O)(=O)C1=CC=CC=C1 SRSXLGNVWSONIS-UHFFFAOYSA-N 0.000 description 1
- 229940092714 benzenesulfonic acid Drugs 0.000 description 1
- 150000001558 benzoic acid derivatives Chemical class 0.000 description 1
- MITFXPHMIHQXPI-UHFFFAOYSA-N benzoxaprofen Natural products N=1C2=CC(C(C(O)=O)C)=CC=C2OC=1C1=CC=C(Cl)C=C1 MITFXPHMIHQXPI-UHFFFAOYSA-N 0.000 description 1
- ZYXYTGQFPZEUFX-UHFFFAOYSA-N benzpyrimoxan Chemical compound O1C(OCCC1)C=1C(=NC=NC=1)OCC1=CC=C(C=C1)C(F)(F)F ZYXYTGQFPZEUFX-UHFFFAOYSA-N 0.000 description 1
- 235000019445 benzyl alcohol Nutrition 0.000 description 1
- 229960002903 benzyl benzoate Drugs 0.000 description 1
- CADWTSSKOVRVJC-UHFFFAOYSA-N benzyl(dimethyl)azanium;chloride Chemical compound [Cl-].C[NH+](C)CC1=CC=CC=C1 CADWTSSKOVRVJC-UHFFFAOYSA-N 0.000 description 1
- YOUGRGFIHBUKRS-UHFFFAOYSA-N benzyl(trimethyl)azanium Chemical class C[N+](C)(C)CC1=CC=CC=C1 YOUGRGFIHBUKRS-UHFFFAOYSA-N 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- OQFSQFPPLPISGP-UHFFFAOYSA-N beta-carboxyaspartic acid Natural products OC(=O)C(N)C(C(O)=O)C(O)=O OQFSQFPPLPISGP-UHFFFAOYSA-N 0.000 description 1
- 229960003237 betaine Drugs 0.000 description 1
- VHLKTXFWDRXILV-UHFFFAOYSA-N bifenazate Chemical compound C1=C(NNC(=O)OC(C)C)C(OC)=CC=C1C1=CC=CC=C1 VHLKTXFWDRXILV-UHFFFAOYSA-N 0.000 description 1
- 229960001901 bioallethrin Drugs 0.000 description 1
- 229950002373 bioresmethrin Drugs 0.000 description 1
- VEMKTZHHVJILDY-UXHICEINSA-N bioresmethrin Chemical compound CC1(C)[C@H](C=C(C)C)[C@H]1C(=O)OCC1=COC(CC=2C=CC=CC=2)=C1 VEMKTZHHVJILDY-UXHICEINSA-N 0.000 description 1
- 239000004305 biphenyl Substances 0.000 description 1
- 235000010290 biphenyl Nutrition 0.000 description 1
- OIPMQULDKWSNGX-UHFFFAOYSA-N bis[[ethoxy(oxo)phosphaniumyl]oxy]alumanyloxy-ethoxy-oxophosphanium Chemical compound [Al+3].CCO[P+]([O-])=O.CCO[P+]([O-])=O.CCO[P+]([O-])=O OIPMQULDKWSNGX-UHFFFAOYSA-N 0.000 description 1
- 229960002326 bithionol Drugs 0.000 description 1
- JFIOVJDNOJYLKP-UHFFFAOYSA-N bithionol Chemical compound OC1=C(Cl)C=C(Cl)C=C1SC1=CC(Cl)=CC(Cl)=C1O JFIOVJDNOJYLKP-UHFFFAOYSA-N 0.000 description 1
- LDLMOOXUCMHBMZ-UHFFFAOYSA-N bixafen Chemical compound FC(F)C1=NN(C)C=C1C(=O)NC1=CC=C(F)C=C1C1=CC=C(Cl)C(Cl)=C1 LDLMOOXUCMHBMZ-UHFFFAOYSA-N 0.000 description 1
- 229920001400 block copolymer Polymers 0.000 description 1
- 230000037396 body weight Effects 0.000 description 1
- 229940118790 boscalid Drugs 0.000 description 1
- WYEMLYFITZORAB-UHFFFAOYSA-N boscalid Chemical compound C1=CC(Cl)=CC=C1C1=CC=CC=C1NC(=O)C1=CC=CN=C1Cl WYEMLYFITZORAB-UHFFFAOYSA-N 0.000 description 1
- QSLZKWPYTWEWHC-UHFFFAOYSA-N broflanilide Chemical compound C=1C=CC(C(=O)NC=2C(=CC(=CC=2Br)C(F)(C(F)(F)F)C(F)(F)F)C(F)(F)F)=C(F)C=1N(C)C(=O)C1=CC=CC=C1 QSLZKWPYTWEWHC-UHFFFAOYSA-N 0.000 description 1
- 125000005997 bromomethyl group Chemical group 0.000 description 1
- FOANIXZHAMJWOI-UHFFFAOYSA-N bromopropylate Chemical compound C=1C=C(Br)C=CC=1C(O)(C(=O)OC(C)C)C1=CC=C(Br)C=C1 FOANIXZHAMJWOI-UHFFFAOYSA-N 0.000 description 1
- HJJVPARKXDDIQD-UHFFFAOYSA-N bromuconazole Chemical compound ClC1=CC(Cl)=CC=C1C1(CN2N=CN=C2)OCC(Br)C1 HJJVPARKXDDIQD-UHFFFAOYSA-N 0.000 description 1
- 229960003168 bronopol Drugs 0.000 description 1
- 239000000337 buffer salt Substances 0.000 description 1
- DSKJPMWIHSOYEA-UHFFFAOYSA-N bupirimate Chemical compound CCCCC1=C(C)N=C(NCC)N=C1OS(=O)(=O)N(C)C DSKJPMWIHSOYEA-UHFFFAOYSA-N 0.000 description 1
- PRLVTUNWOQKEAI-VKAVYKQESA-N buprofezin Chemical compound O=C1N(C(C)C)\C(=N\C(C)(C)C)SCN1C1=CC=CC=C1 PRLVTUNWOQKEAI-VKAVYKQESA-N 0.000 description 1
- 229940043232 butyl acetate Drugs 0.000 description 1
- CZBZUDVBLSSABA-UHFFFAOYSA-N butylated hydroxyanisole Chemical compound COC1=CC=C(O)C(C(C)(C)C)=C1.COC1=CC=C(O)C=C1C(C)(C)C CZBZUDVBLSSABA-UHFFFAOYSA-N 0.000 description 1
- 125000004063 butyryl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- KXRPCFINVWWFHQ-UHFFFAOYSA-N cadusafos Chemical compound CCC(C)SP(=O)(OCC)SC(C)CC KXRPCFINVWWFHQ-UHFFFAOYSA-N 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- 235000010216 calcium carbonate Nutrition 0.000 description 1
- HCWYXKWQOMTBKY-UHFFFAOYSA-N calcium;dodecyl benzenesulfonate Chemical compound [Ca].CCCCCCCCCCCCOS(=O)(=O)C1=CC=CC=C1 HCWYXKWQOMTBKY-UHFFFAOYSA-N 0.000 description 1
- JHRWWRDRBPCWTF-OLQVQODUSA-N captafol Chemical compound C1C=CC[C@H]2C(=O)N(SC(Cl)(Cl)C(Cl)Cl)C(=O)[C@H]21 JHRWWRDRBPCWTF-OLQVQODUSA-N 0.000 description 1
- 229940117949 captan Drugs 0.000 description 1
- 150000004657 carbamic acid derivatives Chemical class 0.000 description 1
- 239000004202 carbamide Substances 0.000 description 1
- 229960005286 carbaryl Drugs 0.000 description 1
- CVXBEEMKQHEXEN-UHFFFAOYSA-N carbaryl Chemical compound C1=CC=C2C(OC(=O)NC)=CC=CC2=C1 CVXBEEMKQHEXEN-UHFFFAOYSA-N 0.000 description 1
- 239000006013 carbendazim Substances 0.000 description 1
- JNPZQRQPIHJYNM-UHFFFAOYSA-N carbendazim Chemical compound C1=C[CH]C2=NC(NC(=O)OC)=NC2=C1 JNPZQRQPIHJYNM-UHFFFAOYSA-N 0.000 description 1
- 238000003763 carbonization Methods 0.000 description 1
- JLQUFIHWVLZVTJ-UHFFFAOYSA-N carbosulfan Chemical compound CCCCN(CCCC)SN(C)C(=O)OC1=CC=CC2=C1OC(C)(C)C2 JLQUFIHWVLZVTJ-UHFFFAOYSA-N 0.000 description 1
- GYSSRZJIHXQEHQ-UHFFFAOYSA-N carboxin Chemical compound S1CCOC(C)=C1C(=O)NC1=CC=CC=C1 GYSSRZJIHXQEHQ-UHFFFAOYSA-N 0.000 description 1
- 150000001734 carboxylic acid salts Chemical class 0.000 description 1
- RXDMAYSSBPYBFW-UHFFFAOYSA-N carpropamid Chemical compound C=1C=C(Cl)C=CC=1C(C)NC(=O)C1(CC)C(C)C1(Cl)Cl RXDMAYSSBPYBFW-UHFFFAOYSA-N 0.000 description 1
- IRUJZVNXZWPBMU-UHFFFAOYSA-N cartap Chemical compound NC(=O)SCC(N(C)C)CSC(N)=O IRUJZVNXZWPBMU-UHFFFAOYSA-N 0.000 description 1
- WOWHHFRSBJGXCM-UHFFFAOYSA-M cetyltrimethylammonium chloride Chemical compound [Cl-].CCCCCCCCCCCCCCCC[N+](C)(C)C WOWHHFRSBJGXCM-UHFFFAOYSA-M 0.000 description 1
- PSOVNZZNOMJUBI-UHFFFAOYSA-N chlorantraniliprole Chemical compound CNC(=O)C1=CC(Cl)=CC(C)=C1NC(=O)C1=CC(Br)=NN1C1=NC=CC=C1Cl PSOVNZZNOMJUBI-UHFFFAOYSA-N 0.000 description 1
- UISUNVFOGSJSKD-UHFFFAOYSA-N chlorfluazuron Chemical compound FC1=CC=CC(F)=C1C(=O)NC(=O)NC(C=C1Cl)=CC(Cl)=C1OC1=NC=C(C(F)(F)F)C=C1Cl UISUNVFOGSJSKD-UHFFFAOYSA-N 0.000 description 1
- XTEGARKTQYYJKE-UHFFFAOYSA-N chloric acid Chemical compound OCl(=O)=O XTEGARKTQYYJKE-UHFFFAOYSA-N 0.000 description 1
- 229940005991 chloric acid Drugs 0.000 description 1
- 239000012320 chlorinating reagent Substances 0.000 description 1
- QGTYWWGEWOBMAK-UHFFFAOYSA-N chlormephos Chemical compound CCOP(=S)(OCC)SCCl QGTYWWGEWOBMAK-UHFFFAOYSA-N 0.000 description 1
- HKMOPYJWSFRURD-UHFFFAOYSA-N chloro hypochlorite;copper Chemical compound [Cu].ClOCl HKMOPYJWSFRURD-UHFFFAOYSA-N 0.000 description 1
- 125000002668 chloroacetyl group Chemical group ClCC(=O)* 0.000 description 1
- 229960004926 chlorobutanol Drugs 0.000 description 1
- OSASVXMJTNOKOY-UHFFFAOYSA-N chlorobutanol Chemical compound CC(C)(O)C(Cl)(Cl)Cl OSASVXMJTNOKOY-UHFFFAOYSA-N 0.000 description 1
- 125000004789 chlorodifluoromethoxy group Chemical group ClC(O*)(F)F 0.000 description 1
- 125000004775 chlorodifluoromethyl group Chemical group FC(F)(Cl)* 0.000 description 1
- 125000004218 chloromethyl group Chemical group [H]C([H])(Cl)* 0.000 description 1
- PFIADAMVCJPXSF-UHFFFAOYSA-N chloroneb Chemical compound COC1=CC(Cl)=C(OC)C=C1Cl PFIADAMVCJPXSF-UHFFFAOYSA-N 0.000 description 1
- LFHISGNCFUNFFM-UHFFFAOYSA-N chloropicrin Chemical compound [O-][N+](=O)C(Cl)(Cl)Cl LFHISGNCFUNFFM-UHFFFAOYSA-N 0.000 description 1
- RQNZCZHWQZMZER-ZLDLUXBVSA-N chloroprallethrin Chemical compound CC1=C(CC#C)C(=O)C[C@@H]1OC(=O)[C@H]1C(C)(C)[C@@H]1C=C(Cl)Cl RQNZCZHWQZMZER-ZLDLUXBVSA-N 0.000 description 1
- CRQQGFGUEAVUIL-UHFFFAOYSA-N chlorothalonil Chemical compound ClC1=C(Cl)C(C#N)=C(Cl)C(C#N)=C1Cl CRQQGFGUEAVUIL-UHFFFAOYSA-N 0.000 description 1
- SBPBAQFWLVIOKP-UHFFFAOYSA-N chlorpyrifos Chemical compound CCOP(=S)(OCC)OC1=NC(Cl)=C(Cl)C=C1Cl SBPBAQFWLVIOKP-UHFFFAOYSA-N 0.000 description 1
- HPNSNYBUADCFDR-UHFFFAOYSA-N chromafenozide Chemical compound CC1=CC(C)=CC(C(=O)N(NC(=O)C=2C(=C3CCCOC3=CC=2)C)C(C)(C)C)=C1 HPNSNYBUADCFDR-UHFFFAOYSA-N 0.000 description 1
- 235000015165 citric acid Nutrition 0.000 description 1
- 229960004106 citric acid Drugs 0.000 description 1
- 229960003344 climbazole Drugs 0.000 description 1
- UXADOQPNKNTIHB-UHFFFAOYSA-N clofentezine Chemical compound ClC1=CC=CC=C1C1=NN=C(C=2C(=CC=CC=2)Cl)N=N1 UXADOQPNKNTIHB-UHFFFAOYSA-N 0.000 description 1
- VNFPBHJOKIVQEB-UHFFFAOYSA-N clotrimazole Chemical compound ClC1=CC=CC=C1C(N1C=NC=C1)(C=1C=CC=CC=1)C1=CC=CC=C1 VNFPBHJOKIVQEB-UHFFFAOYSA-N 0.000 description 1
- 229960004022 clotrimazole Drugs 0.000 description 1
- 239000003240 coconut oil Substances 0.000 description 1
- 235000019864 coconut oil Nutrition 0.000 description 1
- 239000008119 colloidal silica Substances 0.000 description 1
- 239000012230 colorless oil Substances 0.000 description 1
- 238000004440 column chromatography Methods 0.000 description 1
- 239000012141 concentrate Substances 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 229940116318 copper carbonate Drugs 0.000 description 1
- 229910001956 copper hydroxide Inorganic materials 0.000 description 1
- OPQARKPSCNTWTJ-UHFFFAOYSA-L copper(ii) acetate Chemical compound [Cu+2].CC([O-])=O.CC([O-])=O OPQARKPSCNTWTJ-UHFFFAOYSA-L 0.000 description 1
- SVOAENZIOKPANY-CVBJKYQLSA-L copper;(z)-octadec-9-enoate Chemical compound [Cu+2].CCCCCCCC\C=C/CCCCCCCC([O-])=O.CCCCCCCC\C=C/CCCCCCCC([O-])=O SVOAENZIOKPANY-CVBJKYQLSA-L 0.000 description 1
- GEZOTWYUIKXWOA-UHFFFAOYSA-L copper;carbonate Chemical compound [Cu+2].[O-]C([O-])=O GEZOTWYUIKXWOA-UHFFFAOYSA-L 0.000 description 1
- JOXAXMBQVHFGQT-UHFFFAOYSA-J copper;manganese(2+);n-[2-(sulfidocarbothioylamino)ethyl]carbamodithioate Chemical compound [Mn+2].[Cu+2].[S-]C(=S)NCCNC([S-])=S.[S-]C(=S)NCCNC([S-])=S JOXAXMBQVHFGQT-UHFFFAOYSA-J 0.000 description 1
- 239000007799 cork Substances 0.000 description 1
- 235000005822 corn Nutrition 0.000 description 1
- 239000008120 corn starch Substances 0.000 description 1
- 229940099112 cornstarch Drugs 0.000 description 1
- 235000012343 cottonseed oil Nutrition 0.000 description 1
- 239000002385 cottonseed oil Substances 0.000 description 1
- CWVRPJSBNHNJSI-XQNSMLJCSA-N coumoxystrobin Chemical compound C1=C2OC(=O)C(CCCC)=C(C)C2=CC=C1OCC1=CC=CC=C1\C(=C/OC)C(=O)OC CWVRPJSBNHNJSI-XQNSMLJCSA-N 0.000 description 1
- 230000009193 crawling Effects 0.000 description 1
- 239000006071 cream Substances 0.000 description 1
- 229930003836 cresol Natural products 0.000 description 1
- 229940013361 cresol Drugs 0.000 description 1
- BWPPQTSQDHTDQR-UHFFFAOYSA-N cyanatosulfanyl cyanate;potassium Chemical compound [K].N#COSOC#N BWPPQTSQDHTDQR-UHFFFAOYSA-N 0.000 description 1
- SCKHCCSZFPSHGR-UHFFFAOYSA-N cyanophos Chemical compound COP(=S)(OC)OC1=CC=C(C#N)C=C1 SCKHCCSZFPSHGR-UHFFFAOYSA-N 0.000 description 1
- DVBUIBGJRQBEDP-UHFFFAOYSA-N cyantraniliprole Chemical compound CNC(=O)C1=CC(C#N)=CC(C)=C1NC(=O)C1=CC(Br)=NN1C1=NC=CC=C1Cl DVBUIBGJRQBEDP-UHFFFAOYSA-N 0.000 description 1
- YXKMMRDKEKCERS-UHFFFAOYSA-N cyazofamid Chemical compound CN(C)S(=O)(=O)N1C(C#N)=NC(Cl)=C1C1=CC=C(C)C=C1 YXKMMRDKEKCERS-UHFFFAOYSA-N 0.000 description 1
- 229930186364 cyclamen Natural products 0.000 description 1
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- HPXRVTGHNJAIIH-UHFFFAOYSA-N cyclohexanol Chemical compound OC1CCCCC1 HPXRVTGHNJAIIH-UHFFFAOYSA-N 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- LSFUGNKKPMBOMG-UHFFFAOYSA-N cycloprothrin Chemical compound ClC1(Cl)CC1(C=1C=CC=CC=1)C(=O)OC(C#N)C1=CC=CC(OC=2C=CC=CC=2)=C1 LSFUGNKKPMBOMG-UHFFFAOYSA-N 0.000 description 1
- APJLTUBHYCOZJI-VZCXRCSSSA-N cyenopyrafen Chemical compound CC1=NN(C)C(\C(OC(=O)C(C)(C)C)=C(/C#N)C=2C=CC(=CC=2)C(C)(C)C)=C1C APJLTUBHYCOZJI-VZCXRCSSSA-N 0.000 description 1
- ACMXQHFNODYQAT-UHFFFAOYSA-N cyflufenamid Chemical compound FC1=CC=C(C(F)(F)F)C(C(NOCC2CC2)=NC(=O)CC=2C=CC=CC=2)=C1F ACMXQHFNODYQAT-UHFFFAOYSA-N 0.000 description 1
- 229960001591 cyfluthrin Drugs 0.000 description 1
- QQODLKZGRKWIFG-QSFXBCCZSA-N cyfluthrin Chemical compound CC1(C)[C@@H](C=C(Cl)Cl)[C@H]1C(=O)O[C@@H](C#N)C1=CC=C(F)C(OC=2C=CC=CC=2)=C1 QQODLKZGRKWIFG-QSFXBCCZSA-N 0.000 description 1
- NNRSYETYEADPBW-UHFFFAOYSA-N cyhalodiamide Chemical compound CC1=CC(C(F)(C(F)(F)F)C(F)(F)F)=CC=C1NC(=O)C1=CC=CC(Cl)=C1C(=O)NC(C)(C)C#N NNRSYETYEADPBW-UHFFFAOYSA-N 0.000 description 1
- ZXQYGBMAQZUVMI-UNOMPAQXSA-N cyhalothrin Chemical compound CC1(C)C(\C=C(/Cl)C(F)(F)F)C1C(=O)OC(C#N)C1=CC=CC(OC=2C=CC=CC=2)=C1 ZXQYGBMAQZUVMI-UNOMPAQXSA-N 0.000 description 1
- HAORKNGNJCEJBX-UHFFFAOYSA-N cyprodinil Chemical compound N=1C(C)=CC(C2CC2)=NC=1NC1=CC=CC=C1 HAORKNGNJCEJBX-UHFFFAOYSA-N 0.000 description 1
- LVQDKIWDGQRHTE-UHFFFAOYSA-N cyromazine Chemical compound NC1=NC(N)=NC(NC2CC2)=N1 LVQDKIWDGQRHTE-UHFFFAOYSA-N 0.000 description 1
- 229950000775 cyromazine Drugs 0.000 description 1
- GVJHHUAWPYXKBD-UHFFFAOYSA-N d-alpha-tocopherol Natural products OC1=C(C)C(C)=C2OC(CCCC(C)CCCC(C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-UHFFFAOYSA-N 0.000 description 1
- QAYICIQNSGETAS-UHFFFAOYSA-N dazomet Chemical compound CN1CSC(=S)N(C)C1 QAYICIQNSGETAS-UHFFFAOYSA-N 0.000 description 1
- 229960002483 decamethrin Drugs 0.000 description 1
- GHVNFZFCNZKVNT-UHFFFAOYSA-M decanoate Chemical compound CCCCCCCCCC([O-])=O GHVNFZFCNZKVNT-UHFFFAOYSA-M 0.000 description 1
- SASYSVUEVMOWPL-NXVVXOECSA-N decyl oleate Chemical compound CCCCCCCCCCOC(=O)CCCCCCC\C=C/CCCCCCCC SASYSVUEVMOWPL-NXVVXOECSA-N 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 235000019258 dehydroacetic acid Nutrition 0.000 description 1
- 229940061632 dehydroacetic acid Drugs 0.000 description 1
- PGRHXDWITVMQBC-UHFFFAOYSA-N dehydroacetic acid Natural products CC(=O)C1C(=O)OC(C)=CC1=O PGRHXDWITVMQBC-UHFFFAOYSA-N 0.000 description 1
- OWZREIFADZCYQD-NSHGMRRFSA-N deltamethrin Chemical compound CC1(C)[C@@H](C=C(Br)Br)[C@H]1C(=O)O[C@H](C#N)C1=CC=CC(OC=2C=CC=CC=2)=C1 OWZREIFADZCYQD-NSHGMRRFSA-N 0.000 description 1
- 235000019425 dextrin Nutrition 0.000 description 1
- WOWBFOBYOAGEEA-UHFFFAOYSA-N diafenthiuron Chemical compound CC(C)C1=C(NC(=S)NC(C)(C)C)C(C(C)C)=CC(OC=2C=CC=CC=2)=C1 WOWBFOBYOAGEEA-UHFFFAOYSA-N 0.000 description 1
- MNNHAPBLZZVQHP-UHFFFAOYSA-N diammonium hydrogen phosphate Chemical compound [NH4+].[NH4+].OP([O-])([O-])=O MNNHAPBLZZVQHP-UHFFFAOYSA-N 0.000 description 1
- 229910000388 diammonium phosphate Inorganic materials 0.000 description 1
- 235000019838 diammonium phosphate Nutrition 0.000 description 1
- FHIVAFMUCKRCQO-UHFFFAOYSA-N diazinon Chemical compound CCOP(=S)(OCC)OC1=CC(C)=NC(C(C)C)=N1 FHIVAFMUCKRCQO-UHFFFAOYSA-N 0.000 description 1
- WURGXGVFSMYFCG-UHFFFAOYSA-N dichlofluanid Chemical compound CN(C)S(=O)(=O)N(SC(F)(Cl)Cl)C1=CC=CC=C1 WURGXGVFSMYFCG-UHFFFAOYSA-N 0.000 description 1
- BIXZHMJUSMUDOQ-UHFFFAOYSA-N dichloran Chemical compound NC1=C(Cl)C=C([N+]([O-])=O)C=C1Cl BIXZHMJUSMUDOQ-UHFFFAOYSA-N 0.000 description 1
- 125000004772 dichloromethyl group Chemical group [H]C(Cl)(Cl)* 0.000 description 1
- OEBRKCOSUFCWJD-UHFFFAOYSA-N dichlorvos Chemical compound COP(=O)(OC)OC=C(Cl)Cl OEBRKCOSUFCWJD-UHFFFAOYSA-N 0.000 description 1
- 229950001327 dichlorvos Drugs 0.000 description 1
- YEJGPFZQLRMXOI-PKEIRNPWSA-N diclocymet Chemical compound N#CC(C(C)(C)C)C(=O)N[C@H](C)C1=CC=C(Cl)C=C1Cl YEJGPFZQLRMXOI-PKEIRNPWSA-N 0.000 description 1
- 229940004812 dicloran Drugs 0.000 description 1
- PVDQXPBKBSCNJZ-UHFFFAOYSA-N dicloromezotiaz Chemical compound CC1=CC=C[N+](C(C(C=2C=C(Cl)C=C(Cl)C=2)=C2[O-])=O)=C1N2CC1=CN=C(Cl)S1 PVDQXPBKBSCNJZ-UHFFFAOYSA-N 0.000 description 1
- PNKGIAQRMACYGQ-UHFFFAOYSA-N dicopper;dizinc;chromium(3+);oxygen(2-) Chemical compound [O-2].[O-2].[O-2].[O-2].[O-2].[O-2].[Cr+3].[Cr+3].[Cu+2].[Cu+2].[Zn+2].[Zn+2] PNKGIAQRMACYGQ-UHFFFAOYSA-N 0.000 description 1
- JXSJBGJIGXNWCI-UHFFFAOYSA-N diethyl 2-[(dimethoxyphosphorothioyl)thio]succinate Chemical compound CCOC(=O)CC(SP(=S)(OC)OC)C(=O)OCC JXSJBGJIGXNWCI-UHFFFAOYSA-N 0.000 description 1
- 229940028356 diethylene glycol monobutyl ether Drugs 0.000 description 1
- BQYJATMQXGBDHF-UHFFFAOYSA-N difenoconazole Chemical compound O1C(C)COC1(C=1C(=CC(OC=2C=CC(Cl)=CC=2)=CC=1)Cl)CN1N=CN=C1 BQYJATMQXGBDHF-UHFFFAOYSA-N 0.000 description 1
- 229940019503 diflubenzuron Drugs 0.000 description 1
- 125000004786 difluoromethoxy group Chemical group [H]C(F)(F)O* 0.000 description 1
- 125000001028 difluoromethyl group Chemical group [H]C(F)(F)* 0.000 description 1
- 229940031578 diisopropyl adipate Drugs 0.000 description 1
- WZPMZMCZAGFKOC-UHFFFAOYSA-N diisopropyl hydrogen phosphate Chemical compound CC(C)OP(O)(=O)OC(C)C WZPMZMCZAGFKOC-UHFFFAOYSA-N 0.000 description 1
- 238000010790 dilution Methods 0.000 description 1
- 239000012895 dilution Substances 0.000 description 1
- CJHXCRMKMMBYJQ-UHFFFAOYSA-N dimethirimol Chemical compound CCCCC1=C(C)NC(N(C)C)=NC1=O CJHXCRMKMMBYJQ-UHFFFAOYSA-N 0.000 description 1
- MCWXGJITAZMZEV-UHFFFAOYSA-N dimethoate Chemical compound CNC(=O)CSP(=S)(OC)OC MCWXGJITAZMZEV-UHFFFAOYSA-N 0.000 description 1
- 125000000118 dimethyl group Chemical group [H]C([H])([H])* 0.000 description 1
- ZHIKKPYAYGUYPV-UHFFFAOYSA-N dimethylaminocarbamothioylsulfanyl n-(dimethylamino)carbamodithioate Chemical compound CN(C)NC(=S)SSC(=S)NN(C)C ZHIKKPYAYGUYPV-UHFFFAOYSA-N 0.000 description 1
- MZGNSEAPZQGJRB-UHFFFAOYSA-N dimethyldithiocarbamic acid Chemical compound CN(C)C(S)=S MZGNSEAPZQGJRB-UHFFFAOYSA-N 0.000 description 1
- WXUZAHCNPWONDH-DYTRJAOYSA-N dimoxystrobin Chemical compound CNC(=O)C(=N\OC)\C1=CC=CC=C1COC1=CC(C)=CC=C1C WXUZAHCNPWONDH-DYTRJAOYSA-N 0.000 description 1
- YKBZOVFACRVRJN-UHFFFAOYSA-N dinotefuran Chemical compound [O-][N+](=O)\N=C(/NC)NCC1CCOC1 YKBZOVFACRVRJN-UHFFFAOYSA-N 0.000 description 1
- 235000019329 dioctyl sodium sulphosuccinate Nutrition 0.000 description 1
- HNPSIPDUKPIQMN-UHFFFAOYSA-N dioxosilane;oxo(oxoalumanyloxy)alumane Chemical compound O=[Si]=O.O=[Al]O[Al]=O HNPSIPDUKPIQMN-UHFFFAOYSA-N 0.000 description 1
- USIUVYZYUHIAEV-UHFFFAOYSA-N diphenyl ether Chemical compound C=1C=CC=CC=1OC1=CC=CC=C1 USIUVYZYUHIAEV-UHFFFAOYSA-N 0.000 description 1
- SZXQTJUDPRGNJN-UHFFFAOYSA-N dipropylene glycol Chemical compound OCCCOCCCO SZXQTJUDPRGNJN-UHFFFAOYSA-N 0.000 description 1
- FPKXBFWMIYHCID-UHFFFAOYSA-N dipymetitrone Chemical compound S1C=2C(=O)N(C)C(=O)C=2SC2=C1C(=O)N(C)C2=O FPKXBFWMIYHCID-UHFFFAOYSA-N 0.000 description 1
- ZHDBTKPXEJDTTQ-UHFFFAOYSA-N dipyrithione Chemical compound [O-][N+]1=CC=CC=C1SSC1=CC=CC=[N+]1[O-] ZHDBTKPXEJDTTQ-UHFFFAOYSA-N 0.000 description 1
- 235000021186 dishes Nutrition 0.000 description 1
- YHAIUSTWZPMYGG-UHFFFAOYSA-L disodium;2,2-dioctyl-3-sulfobutanedioate Chemical compound [Na+].[Na+].CCCCCCCCC(C([O-])=O)(C(C([O-])=O)S(O)(=O)=O)CCCCCCCC YHAIUSTWZPMYGG-UHFFFAOYSA-L 0.000 description 1
- KSDGSKVLUHKDAL-UHFFFAOYSA-L disodium;3-[2-carboxylatoethyl(dodecyl)amino]propanoate Chemical compound [Na+].[Na+].CCCCCCCCCCCCN(CCC([O-])=O)CCC([O-])=O KSDGSKVLUHKDAL-UHFFFAOYSA-L 0.000 description 1
- WBZKQQHYRPRKNJ-UHFFFAOYSA-L disulfite Chemical compound [O-]S(=O)S([O-])(=O)=O WBZKQQHYRPRKNJ-UHFFFAOYSA-L 0.000 description 1
- DOFZAZXDOSGAJZ-UHFFFAOYSA-N disulfoton Chemical compound CCOP(=S)(OCC)SCCSCC DOFZAZXDOSGAJZ-UHFFFAOYSA-N 0.000 description 1
- PYZSVQVRHDXQSL-UHFFFAOYSA-N dithianon Chemical compound S1C(C#N)=C(C#N)SC2=C1C(=O)C1=CC=CC=C1C2=O PYZSVQVRHDXQSL-UHFFFAOYSA-N 0.000 description 1
- 229910000514 dolomite Inorganic materials 0.000 description 1
- 239000010459 dolomite Substances 0.000 description 1
- GCKZANITAMOIAR-XWVCPFKXSA-N dsstox_cid_14566 Chemical compound [O-]C(=O)C1=CC=CC=C1.C1=C[C@H](C)[C@@H]([C@@H](C)CC)O[C@]11O[C@H](C\C=C(C)\[C@@H](O[C@@H]2O[C@@H](C)[C@H](O[C@@H]3O[C@@H](C)[C@H]([NH2+]C)[C@@H](OC)C3)[C@@H](OC)C2)[C@@H](C)\C=C\C=C/2[C@]3([C@H](C(=O)O4)C=C(C)[C@@H](O)[C@H]3OC\2)O)C[C@H]4C1 GCKZANITAMOIAR-XWVCPFKXSA-N 0.000 description 1
- 238000010410 dusting Methods 0.000 description 1
- AWZOLILCOUMRDG-UHFFFAOYSA-N edifenphos Chemical compound C=1C=CC=CC=1SP(=O)(OCC)SC1=CC=CC=C1 AWZOLILCOUMRDG-UHFFFAOYSA-N 0.000 description 1
- 239000004495 emulsifiable concentrate Substances 0.000 description 1
- 239000004516 emulsifiable gel Substances 0.000 description 1
- 230000001804 emulsifying effect Effects 0.000 description 1
- RDYMFSUJUZBWLH-SVWSLYAFSA-N endosulfan Chemical compound C([C@@H]12)OS(=O)OC[C@@H]1[C@]1(Cl)C(Cl)=C(Cl)[C@@]2(Cl)C1(Cl)Cl RDYMFSUJUZBWLH-SVWSLYAFSA-N 0.000 description 1
- 229960002125 enilconazole Drugs 0.000 description 1
- VMNULHCTRPXWFJ-UJSVPXBISA-N enoxastrobin Chemical compound CO\C=C(\C(=O)OC)C1=CC=CC=C1CO\N=C(/C)\C=C\C1=CC=C(Cl)C=C1 VMNULHCTRPXWFJ-UJSVPXBISA-N 0.000 description 1
- 229940007078 entamoeba histolytica Drugs 0.000 description 1
- KVIZNNVXXNFLMU-DUVUQDDDSA-N epsilon-metofluthrin Chemical compound FC1=C(F)C(COC)=C(F)C(F)=C1COC(=O)[C@H]1C(C)(C)[C@@H]1\C=C/C KVIZNNVXXNFLMU-DUVUQDDDSA-N 0.000 description 1
- DPJITPZADZSLBP-DQXQJKBJSA-N epsilon-momfluorothrin Chemical compound FC1=C(F)C(COC)=C(F)C(F)=C1COC(=O)[C@H]1C(C)(C)[C@@H]1\C=C(\C)C#N DPJITPZADZSLBP-DQXQJKBJSA-N 0.000 description 1
- NYPJDWWKZLNGGM-RPWUZVMVSA-N esfenvalerate Chemical compound C=1C([C@@H](C#N)OC(=O)[C@@H](C(C)C)C=2C=CC(Cl)=CC=2)=CC=CC=1OC1=CC=CC=C1 NYPJDWWKZLNGGM-RPWUZVMVSA-N 0.000 description 1
- DWRKFAJEBUWTQM-UHFFFAOYSA-N etaconazole Chemical compound O1C(CC)COC1(C=1C(=CC(Cl)=CC=1)Cl)CN1N=CN=C1 DWRKFAJEBUWTQM-UHFFFAOYSA-N 0.000 description 1
- CCIVGXIOQKPBKL-UHFFFAOYSA-M ethanesulfonate Chemical compound CCS([O-])(=O)=O CCIVGXIOQKPBKL-UHFFFAOYSA-M 0.000 description 1
- 150000002169 ethanolamines Chemical class 0.000 description 1
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 description 1
- 125000003754 ethoxycarbonyl group Chemical group C(=O)(OCC)* 0.000 description 1
- 229940093500 ethoxyquin Drugs 0.000 description 1
- DECIPOUIJURFOJ-UHFFFAOYSA-N ethoxyquin Chemical compound N1C(C)(C)C=C(C)C2=CC(OCC)=CC=C21 DECIPOUIJURFOJ-UHFFFAOYSA-N 0.000 description 1
- 235000019285 ethoxyquin Nutrition 0.000 description 1
- IGUYEXXAGBDLLX-UHFFFAOYSA-N ethyl 3-(3,5-dichlorophenyl)-5-methyl-2,4-dioxo-1,3-oxazolidine-5-carboxylate Chemical compound O=C1C(C(=O)OCC)(C)OC(=O)N1C1=CC(Cl)=CC(Cl)=C1 IGUYEXXAGBDLLX-UHFFFAOYSA-N 0.000 description 1
- VICYTAYPKBLQFB-UHFFFAOYSA-N ethyl 3-bromo-2-oxopropanoate Chemical compound CCOC(=O)C(=O)CBr VICYTAYPKBLQFB-UHFFFAOYSA-N 0.000 description 1
- KVIFAOLNQUIVLP-UHFFFAOYSA-N ethyl 7-(trifluoromethyl)imidazo[1,2-a]pyridine-2-carboxylate Chemical compound C1=CC(C(F)(F)F)=CC2=NC(C(=O)OCC)=CN21 KVIFAOLNQUIVLP-UHFFFAOYSA-N 0.000 description 1
- ZCEUTMCJAZNXDC-UHFFFAOYSA-N ethyl 7-iodoimidazo[1,2-a]pyridine-2-carboxylate Chemical compound CCOC(=O)c1cn2ccc(I)cc2n1 ZCEUTMCJAZNXDC-UHFFFAOYSA-N 0.000 description 1
- 229940093499 ethyl acetate Drugs 0.000 description 1
- 229940116333 ethyl lactate Drugs 0.000 description 1
- LVGKNOAMLMIIKO-QXMHVHEDSA-N ethyl oleate Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OCC LVGKNOAMLMIIKO-QXMHVHEDSA-N 0.000 description 1
- 229940093471 ethyl oleate Drugs 0.000 description 1
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 description 1
- YREQHYQNNWYQCJ-UHFFFAOYSA-N etofenprox Chemical compound C1=CC(OCC)=CC=C1C(C)(C)COCC1=CC=CC(OC=2C=CC=CC=2)=C1 YREQHYQNNWYQCJ-UHFFFAOYSA-N 0.000 description 1
- 229950005085 etofenprox Drugs 0.000 description 1
- IXSZQYVWNJNRAL-UHFFFAOYSA-N etoxazole Chemical compound CCOC1=CC(C(C)(C)C)=CC=C1C1N=C(C=2C(=CC=CC=2F)F)OC1 IXSZQYVWNJNRAL-UHFFFAOYSA-N 0.000 description 1
- KQTVWCSONPJJPE-UHFFFAOYSA-N etridiazole Chemical compound CCOC1=NC(C(Cl)(Cl)Cl)=NS1 KQTVWCSONPJJPE-UHFFFAOYSA-N 0.000 description 1
- 208000006275 fascioliasis Diseases 0.000 description 1
- 235000019387 fatty acid methyl ester Nutrition 0.000 description 1
- 150000004665 fatty acids Chemical class 0.000 description 1
- LMVPQMGRYSRMIW-KRWDZBQOSA-N fenamidone Chemical compound O=C([C@@](C)(N=C1SC)C=2C=CC=CC=2)N1NC1=CC=CC=C1 LMVPQMGRYSRMIW-KRWDZBQOSA-N 0.000 description 1
- ZCJPOPBZHLUFHF-UHFFFAOYSA-N fenamiphos Chemical compound CCOP(=O)(NC(C)C)OC1=CC=C(SC)C(C)=C1 ZCJPOPBZHLUFHF-UHFFFAOYSA-N 0.000 description 1
- DMYHGDXADUDKCQ-UHFFFAOYSA-N fenazaquin Chemical compound C1=CC(C(C)(C)C)=CC=C1CCOC1=NC=NC2=CC=CC=C12 DMYHGDXADUDKCQ-UHFFFAOYSA-N 0.000 description 1
- VDLGAVXLJYLFDH-UHFFFAOYSA-N fenhexamid Chemical compound C=1C=C(O)C(Cl)=C(Cl)C=1NC(=O)C1(C)CCCCC1 VDLGAVXLJYLFDH-UHFFFAOYSA-N 0.000 description 1
- ZNOLGFHPUIJIMJ-UHFFFAOYSA-N fenitrothion Chemical compound COP(=S)(OC)OC1=CC=C([N+]([O-])=O)C(C)=C1 ZNOLGFHPUIJIMJ-UHFFFAOYSA-N 0.000 description 1
- DIRFUJHNVNOBMY-UHFFFAOYSA-N fenobucarb Chemical compound CCC(C)C1=CC=CC=C1OC(=O)NC DIRFUJHNVNOBMY-UHFFFAOYSA-N 0.000 description 1
- HJUFTIJOISQSKQ-UHFFFAOYSA-N fenoxycarb Chemical compound C1=CC(OCCNC(=O)OCC)=CC=C1OC1=CC=CC=C1 HJUFTIJOISQSKQ-UHFFFAOYSA-N 0.000 description 1
- FKLFBQCQQYDUAM-UHFFFAOYSA-N fenpiclonil Chemical compound ClC1=CC=CC(C=2C(=CNC=2)C#N)=C1Cl FKLFBQCQQYDUAM-UHFFFAOYSA-N 0.000 description 1
- QGTOTYJSCYHYFK-RBODFLQRSA-N fenpicoxamid Chemical compound COC1=CC=NC(C(=O)N[C@@H]2C(O[C@@H](C)[C@H](OC(=O)C(C)C)[C@@H](CC=3C=CC=CC=3)C(=O)OC2)=O)=C1OCOC(=O)C(C)C QGTOTYJSCYHYFK-RBODFLQRSA-N 0.000 description 1
- YYJNOYZRYGDPNH-MFKUBSTISA-N fenpyroximate Chemical compound C=1C=C(C(=O)OC(C)(C)C)C=CC=1CO/N=C/C=1C(C)=NN(C)C=1OC1=CC=CC=C1 YYJNOYZRYGDPNH-MFKUBSTISA-N 0.000 description 1
- BFWMWWXRWVJXSE-UHFFFAOYSA-M fentin hydroxide Chemical compound C=1C=CC=CC=1[Sn](C=1C=CC=CC=1)(O)C1=CC=CC=C1 BFWMWWXRWVJXSE-UHFFFAOYSA-M 0.000 description 1
- WHDGWKAJBYRJJL-UHFFFAOYSA-K ferbam Chemical compound [Fe+3].CN(C)C([S-])=S.CN(C)C([S-])=S.CN(C)C([S-])=S WHDGWKAJBYRJJL-UHFFFAOYSA-K 0.000 description 1
- GOWLARCWZRESHU-AQTBWJFISA-N ferimzone Chemical compound C=1C=CC=C(C)C=1C(/C)=N\NC1=NC(C)=CC(C)=N1 GOWLARCWZRESHU-AQTBWJFISA-N 0.000 description 1
- 239000010408 film Substances 0.000 description 1
- 229940013764 fipronil Drugs 0.000 description 1
- RLQJEEJISHYWON-UHFFFAOYSA-N flonicamid Chemical compound FC(F)(F)C1=CC=NC=C1C(=O)NCC#N RLQJEEJISHYWON-UHFFFAOYSA-N 0.000 description 1
- PHCCDUCBMCYSNQ-UHFFFAOYSA-N fluazaindolizine Chemical compound COC1=CC=C(Cl)C(S(=O)(=O)NC(=O)C=2N=C3C(Cl)=CC(=CN3C=2)C(F)(F)F)=C1 PHCCDUCBMCYSNQ-UHFFFAOYSA-N 0.000 description 1
- UZCGKGPEKUCDTF-UHFFFAOYSA-N fluazinam Chemical compound [O-][N+](=O)C1=CC(C(F)(F)F)=C(Cl)C([N+]([O-])=O)=C1NC1=NC=C(C(F)(F)F)C=C1Cl UZCGKGPEKUCDTF-UHFFFAOYSA-N 0.000 description 1
- YOWNVPAUWYHLQX-UHFFFAOYSA-N fluazuron Chemical compound FC1=CC=CC(F)=C1C(=O)NC(=O)NC1=CC=C(Cl)C(OC=2C(=CC(=CN=2)C(F)(F)F)Cl)=C1 YOWNVPAUWYHLQX-UHFFFAOYSA-N 0.000 description 1
- 229950006719 fluazuron Drugs 0.000 description 1
- ZGNITFSDLCMLGI-UHFFFAOYSA-N flubendiamide Chemical compound CC1=CC(C(F)(C(F)(F)F)C(F)(F)F)=CC=C1NC(=O)C1=CC=CC(I)=C1C(=O)NC(C)(C)CS(C)(=O)=O ZGNITFSDLCMLGI-UHFFFAOYSA-N 0.000 description 1
- GBIHOLCMZGAKNG-CGAIIQECSA-N flucythrinate Chemical compound O=C([C@@H](C(C)C)C=1C=CC(OC(F)F)=CC=1)OC(C#N)C(C=1)=CC=CC=1OC1=CC=CC=C1 GBIHOLCMZGAKNG-CGAIIQECSA-N 0.000 description 1
- MUJOIMFVNIBMKC-UHFFFAOYSA-N fludioxonil Chemical compound C=12OC(F)(F)OC2=CC=CC=1C1=CNC=C1C#N MUJOIMFVNIBMKC-UHFFFAOYSA-N 0.000 description 1
- GJEREQYJIQASAW-UHFFFAOYSA-N flufenerim Chemical compound CC(F)C1=NC=NC(NCCC=2C=CC(OC(F)(F)F)=CC=2)=C1Cl GJEREQYJIQASAW-UHFFFAOYSA-N 0.000 description 1
- RYLHNOVXKPXDIP-UHFFFAOYSA-N flufenoxuron Chemical compound C=1C=C(NC(=O)NC(=O)C=2C(=CC=CC=2F)F)C(F)=CC=1OC1=CC=C(C(F)(F)F)C=C1Cl RYLHNOVXKPXDIP-UHFFFAOYSA-N 0.000 description 1
- MBHXIQDIVCJZTD-RVDMUPIBSA-N flufenoxystrobin Chemical compound CO\C=C(\C(=O)OC)C1=CC=CC=C1COC1=CC=C(C(F)(F)F)C=C1Cl MBHXIQDIVCJZTD-RVDMUPIBSA-N 0.000 description 1
- GBOYJIHYACSLGN-UHFFFAOYSA-N fluopicolide Chemical compound ClC1=CC(C(F)(F)F)=CN=C1CNC(=O)C1=C(Cl)C=CC=C1Cl GBOYJIHYACSLGN-UHFFFAOYSA-N 0.000 description 1
- KVDJTXBXMWJJEF-UHFFFAOYSA-N fluopyram Chemical compound ClC1=CC(C(F)(F)F)=CN=C1CCNC(=O)C1=CC=CC=C1C(F)(F)F KVDJTXBXMWJJEF-UHFFFAOYSA-N 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 125000001153 fluoro group Chemical group F* 0.000 description 1
- UFEODZBUAFNAEU-NLRVBDNBSA-N fluoxastrobin Chemical compound C=1C=CC=C(OC=2C(=C(OC=3C(=CC=CC=3)Cl)N=CN=2)F)C=1C(=N/OC)\C1=NOCCO1 UFEODZBUAFNAEU-NLRVBDNBSA-N 0.000 description 1
- QOIYTRGFOFZNKF-UHFFFAOYSA-N flupyradifurone Chemical compound C=1C(=O)OCC=1N(CC(F)F)CC1=CC=C(Cl)N=C1 QOIYTRGFOFZNKF-UHFFFAOYSA-N 0.000 description 1
- IJJVMEJXYNJXOJ-UHFFFAOYSA-N fluquinconazole Chemical compound C=1C=C(Cl)C=C(Cl)C=1N1C(=O)C2=CC(F)=CC=C2N=C1N1C=NC=N1 IJJVMEJXYNJXOJ-UHFFFAOYSA-N 0.000 description 1
- MLBZKOGAMRTSKP-UHFFFAOYSA-N fluralaner Chemical compound C1=C(C(=O)NCC(=O)NCC(F)(F)F)C(C)=CC(C=2CC(ON=2)(C=2C=C(Cl)C=C(Cl)C=2)C(F)(F)F)=C1 MLBZKOGAMRTSKP-UHFFFAOYSA-N 0.000 description 1
- 229960004498 fluralaner Drugs 0.000 description 1
- FQKUGOMFVDPBIZ-UHFFFAOYSA-N flusilazole Chemical compound C=1C=C(F)C=CC=1[Si](C=1C=CC(F)=CC=1)(C)CN1C=NC=N1 FQKUGOMFVDPBIZ-UHFFFAOYSA-N 0.000 description 1
- GNVDAZSPJWCIQZ-UHFFFAOYSA-N flusulfamide Chemical compound ClC1=CC([N+](=O)[O-])=CC=C1NS(=O)(=O)C1=CC=C(Cl)C(C(F)(F)F)=C1 GNVDAZSPJWCIQZ-UHFFFAOYSA-N 0.000 description 1
- KGXUEPOHGFWQKF-ZCXUNETKSA-N flutianil Chemical compound COC1=CC=CC=C1N(CCS\1)C/1=C(C#N)/SC1=CC(C(F)(F)F)=CC=C1F KGXUEPOHGFWQKF-ZCXUNETKSA-N 0.000 description 1
- PTCGDEVVHUXTMP-UHFFFAOYSA-N flutolanil Chemical compound CC(C)OC1=CC=CC(NC(=O)C=2C(=CC=CC=2)C(F)(F)F)=C1 PTCGDEVVHUXTMP-UHFFFAOYSA-N 0.000 description 1
- SXSGXWCSHSVPGB-UHFFFAOYSA-N fluxapyroxad Chemical compound FC(F)C1=NN(C)C=C1C(=O)NC1=CC=CC=C1C1=CC(F)=C(F)C(F)=C1 SXSGXWCSHSVPGB-UHFFFAOYSA-N 0.000 description 1
- 239000010881 fly ash Substances 0.000 description 1
- HKIOYBQGHSTUDB-UHFFFAOYSA-N folpet Chemical compound C1=CC=C2C(=O)N(SC(Cl)(Cl)Cl)C(=O)C2=C1 HKIOYBQGHSTUDB-UHFFFAOYSA-N 0.000 description 1
- KVGLBTYUCJYMND-UHFFFAOYSA-N fonofos Chemical compound CCOP(=S)(CC)SC1=CC=CC=C1 KVGLBTYUCJYMND-UHFFFAOYSA-N 0.000 description 1
- NVVZQXQBYZPMLJ-UHFFFAOYSA-N formaldehyde;naphthalene-1-sulfonic acid Chemical compound O=C.C1=CC=C2C(S(=O)(=O)O)=CC=CC2=C1 NVVZQXQBYZPMLJ-UHFFFAOYSA-N 0.000 description 1
- RMFNNCGOSPBBAD-MDWZMJQESA-N formetanate Chemical compound CNC(=O)OC1=CC=CC(\N=C\N(C)C)=C1 RMFNNCGOSPBBAD-MDWZMJQESA-N 0.000 description 1
- 235000019253 formic acid Nutrition 0.000 description 1
- DUFVKSUJRWYZQP-UHFFFAOYSA-N fosthiazate Chemical compound CCC(C)SP(=O)(OCC)N1CCSC1=O DUFVKSUJRWYZQP-UHFFFAOYSA-N 0.000 description 1
- 238000005194 fractionation Methods 0.000 description 1
- ZEYJIQLVKGBLEM-UHFFFAOYSA-N fuberidazole Chemical compound C1=COC(C=2N=C3[CH]C=CC=C3N=2)=C1 ZEYJIQLVKGBLEM-UHFFFAOYSA-N 0.000 description 1
- 239000001530 fumaric acid Substances 0.000 description 1
- 229960002598 fumaric acid Drugs 0.000 description 1
- 235000011087 fumaric acid Nutrition 0.000 description 1
- ZXQYGBMAQZUVMI-GCMPRSNUSA-N gamma-cyhalothrin Chemical compound CC1(C)[C@@H](\C=C(/Cl)C(F)(F)F)[C@H]1C(=O)O[C@H](C#N)C1=CC=CC(OC=2C=CC=CC=2)=C1 ZXQYGBMAQZUVMI-GCMPRSNUSA-N 0.000 description 1
- 239000000174 gluconic acid Substances 0.000 description 1
- 235000012208 gluconic acid Nutrition 0.000 description 1
- 229950006191 gluconic acid Drugs 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 239000003292 glue Substances 0.000 description 1
- 229940075507 glyceryl monostearate Drugs 0.000 description 1
- 229930182470 glycoside Natural products 0.000 description 1
- DDUHZTYCFQRHIY-RBHXEPJQSA-N griseofulvin Chemical compound COC1=CC(=O)C[C@@H](C)[C@@]11C(=O)C(C(OC)=CC(OC)=C2Cl)=C2O1 DDUHZTYCFQRHIY-RBHXEPJQSA-N 0.000 description 1
- 229960002867 griseofulvin Drugs 0.000 description 1
- 229910052602 gypsum Inorganic materials 0.000 description 1
- 239000010440 gypsum Substances 0.000 description 1
- WIFXJBMOTMKRMM-UHFFFAOYSA-N halfenprox Chemical compound C=1C=C(OC(F)(F)Br)C=CC=1C(C)(C)COCC(C=1)=CC=CC=1OC1=CC=CC=C1 WIFXJBMOTMKRMM-UHFFFAOYSA-N 0.000 description 1
- CNKHSLKYRMDDNQ-UHFFFAOYSA-N halofenozide Chemical compound C=1C=CC=CC=1C(=O)N(C(C)(C)C)NC(=O)C1=CC=C(Cl)C=C1 CNKHSLKYRMDDNQ-UHFFFAOYSA-N 0.000 description 1
- 125000006343 heptafluoro propyl group Chemical group 0.000 description 1
- BKACAEJQMLLGAV-PLNGDYQASA-N heptafluthrin Chemical compound FC1=C(F)C(COC)=C(F)C(F)=C1COC(=O)C1C(C)(C)C1\C=C/C(F)(F)F BKACAEJQMLLGAV-PLNGDYQASA-N 0.000 description 1
- 125000000268 heptanoyl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 230000002363 herbicidal effect Effects 0.000 description 1
- CKAPSXZOOQJIBF-UHFFFAOYSA-N hexachlorobenzene Chemical compound ClC1=C(Cl)C(Cl)=C(Cl)C(Cl)=C1Cl CKAPSXZOOQJIBF-UHFFFAOYSA-N 0.000 description 1
- RGNPBRKPHBKNKX-UHFFFAOYSA-N hexaflumuron Chemical compound C1=C(Cl)C(OC(F)(F)C(F)F)=C(Cl)C=C1NC(=O)NC(=O)C1=C(F)C=CC=C1F RGNPBRKPHBKNKX-UHFFFAOYSA-N 0.000 description 1
- 125000004836 hexamethylene group Chemical group [H]C([H])([*:2])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[*:1] 0.000 description 1
- FUZZWVXGSFPDMH-UHFFFAOYSA-N hexanoic acid Chemical compound CCCCCC(O)=O FUZZWVXGSFPDMH-UHFFFAOYSA-N 0.000 description 1
- 125000003104 hexanoyl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 229940100463 hexyl laurate Drugs 0.000 description 1
- 229940051250 hexylene glycol Drugs 0.000 description 1
- 235000012907 honey Nutrition 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- 229940071870 hydroiodic acid Drugs 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 235000019447 hydroxyethyl cellulose Nutrition 0.000 description 1
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 description 1
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 description 1
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 description 1
- UFVKGYZPFZQRLF-UHFFFAOYSA-N hydroxypropyl methyl cellulose Chemical class OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC2C(C(O)C(OC3C(C(O)C(O)C(CO)O3)O)C(CO)O2)O)C(CO)O1 UFVKGYZPFZQRLF-UHFFFAOYSA-N 0.000 description 1
- KGVPNLBXJKTABS-UHFFFAOYSA-N hymexazol Chemical compound CC1=CC(O)=NO1 KGVPNLBXJKTABS-UHFFFAOYSA-N 0.000 description 1
- 239000005457 ice water Substances 0.000 description 1
- AGKSTYPVMZODRV-UHFFFAOYSA-N imibenconazole Chemical compound C1=CC(Cl)=CC=C1CSC(CN1N=CN=C1)=NC1=CC=C(Cl)C=C1Cl AGKSTYPVMZODRV-UHFFFAOYSA-N 0.000 description 1
- 229940056881 imidacloprid Drugs 0.000 description 1
- YWTYJOPNNQFBPC-UHFFFAOYSA-N imidacloprid Chemical compound [O-][N+](=O)\N=C1/NCCN1CC1=CC=C(Cl)N=C1 YWTYJOPNNQFBPC-UHFFFAOYSA-N 0.000 description 1
- WQLJLPDGSLZYEP-UHFFFAOYSA-N imidazo[1,2-a]pyridine-2-carboxylic acid Chemical compound C1=CC=CC2=NC(C(=O)O)=CN21 WQLJLPDGSLZYEP-UHFFFAOYSA-N 0.000 description 1
- RONFGUROBZGJKP-UHFFFAOYSA-N iminoctadine Chemical compound NC(N)=NCCCCCCCCNCCCCCCCCN=C(N)N RONFGUROBZGJKP-UHFFFAOYSA-N 0.000 description 1
- VPRAQYXPZIFIOH-UHFFFAOYSA-N imiprothrin Chemical compound CC1(C)C(C=C(C)C)C1C(=O)OCN1C(=O)N(CC#C)CC1=O VPRAQYXPZIFIOH-UHFFFAOYSA-N 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 229940102223 injectable solution Drugs 0.000 description 1
- YTCIYOXHHQLDEI-SNVBAGLBSA-N inpyrfluxam Chemical compound C([C@H](C=12)C)C(C)(C)C2=CC=CC=1NC(=O)C1=CN(C)N=C1C(F)F YTCIYOXHHQLDEI-SNVBAGLBSA-N 0.000 description 1
- 238000007912 intraperitoneal administration Methods 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- HVTICUPFWKNHNG-UHFFFAOYSA-N iodoethane Chemical compound CCI HVTICUPFWKNHNG-UHFFFAOYSA-N 0.000 description 1
- INQOMBQAUSQDDS-UHFFFAOYSA-N iodomethane Chemical compound IC INQOMBQAUSQDDS-UHFFFAOYSA-N 0.000 description 1
- QTYCMDBMOLSEAM-UHFFFAOYSA-N ipconazole Chemical compound C1=NC=NN1CC1(O)C(C(C)C)CCC1CC1=CC=C(Cl)C=C1 QTYCMDBMOLSEAM-UHFFFAOYSA-N 0.000 description 1
- FCOAHACKGGIURQ-UHFFFAOYSA-N iprobenfos Chemical compound CC(C)OP(=O)(OC(C)C)SCC1=CC=CC=C1 FCOAHACKGGIURQ-UHFFFAOYSA-N 0.000 description 1
- ONUFESLQCSAYKA-UHFFFAOYSA-N iprodione Chemical compound O=C1N(C(=O)NC(C)C)CC(=O)N1C1=CC(Cl)=CC(Cl)=C1 ONUFESLQCSAYKA-UHFFFAOYSA-N 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- HOQADATXFBOEGG-UHFFFAOYSA-N isofenphos Chemical compound CCOP(=S)(NC(C)C)OC1=CC=CC=C1C(=O)OC(C)C HOQADATXFBOEGG-UHFFFAOYSA-N 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- XUGNVMKQXJXZCD-UHFFFAOYSA-N isopropyl palmitate Chemical compound CCCCCCCCCCCCCCCC(=O)OC(C)C XUGNVMKQXJXZCD-UHFFFAOYSA-N 0.000 description 1
- 229940089456 isopropyl stearate Drugs 0.000 description 1
- UFHLMYOGRXOCSL-UHFFFAOYSA-N isoprothiolane Chemical compound CC(C)OC(=O)C(C(=O)OC(C)C)=C1SCCS1 UFHLMYOGRXOCSL-UHFFFAOYSA-N 0.000 description 1
- PVTHJAPFENJVNC-MHRBZPPQSA-N kasugamycin Chemical compound N[C@H]1C[C@H](NC(=N)C(O)=O)[C@@H](C)O[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@@H](O)[C@H](O)[C@@H]1O PVTHJAPFENJVNC-MHRBZPPQSA-N 0.000 description 1
- 239000003350 kerosene Substances 0.000 description 1
- 150000002576 ketones Chemical class 0.000 description 1
- ZOTBXTZVPHCKPN-HTXNQAPBSA-N kresoxim-methyl Chemical compound CO\N=C(\C(=O)OC)C1=CC=CC=C1COC1=CC=CC=C1C ZOTBXTZVPHCKPN-HTXNQAPBSA-N 0.000 description 1
- 238000002372 labelling Methods 0.000 description 1
- 235000014655 lactic acid Nutrition 0.000 description 1
- 239000004310 lactic acid Substances 0.000 description 1
- 229960000448 lactic acid Drugs 0.000 description 1
- 239000005910 lambda-Cyhalothrin Substances 0.000 description 1
- 239000000787 lecithin Substances 0.000 description 1
- 235000010445 lecithin Nutrition 0.000 description 1
- 229940067606 lecithin Drugs 0.000 description 1
- QDLAGTHXVHQKRE-UHFFFAOYSA-N lichenxanthone Natural products COC1=CC(O)=C2C(=O)C3=C(C)C=C(OC)C=C3OC2=C1 QDLAGTHXVHQKRE-UHFFFAOYSA-N 0.000 description 1
- 239000004611 light stabiliser Substances 0.000 description 1
- 239000000944 linseed oil Substances 0.000 description 1
- 235000021388 linseed oil Nutrition 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 210000004072 lung Anatomy 0.000 description 1
- 239000010721 machine oil Substances 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- ZLNQQNXFFQJAID-UHFFFAOYSA-L magnesium carbonate Chemical compound [Mg+2].[O-]C([O-])=O ZLNQQNXFFQJAID-UHFFFAOYSA-L 0.000 description 1
- 239000001095 magnesium carbonate Substances 0.000 description 1
- 229910000021 magnesium carbonate Inorganic materials 0.000 description 1
- 235000014380 magnesium carbonate Nutrition 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 1
- 235000019341 magnesium sulphate Nutrition 0.000 description 1
- 229960000453 malathion Drugs 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000011976 maleic acid Substances 0.000 description 1
- 229940098895 maleic acid Drugs 0.000 description 1
- 239000001630 malic acid Substances 0.000 description 1
- 235000011090 malic acid Nutrition 0.000 description 1
- 229940099690 malic acid Drugs 0.000 description 1
- 229960002510 mandelic acid Drugs 0.000 description 1
- YKSNLCVSTHTHJA-UHFFFAOYSA-L maneb Chemical compound [Mn+2].[S-]C(=S)NCCNC([S-])=S YKSNLCVSTHTHJA-UHFFFAOYSA-L 0.000 description 1
- 229920000940 maneb Polymers 0.000 description 1
- 239000000594 mannitol Substances 0.000 description 1
- 235000010355 mannitol Nutrition 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 235000012054 meals Nutrition 0.000 description 1
- CIFWZNRJIBNXRE-UHFFFAOYSA-N mepanipyrim Chemical compound CC#CC1=CC(C)=NC(NC=2C=CC=CC=2)=N1 CIFWZNRJIBNXRE-UHFFFAOYSA-N 0.000 description 1
- BCTQJXQXJVLSIG-UHFFFAOYSA-N mepronil Chemical compound CC(C)OC1=CC=CC(NC(=O)C=2C(=CC=CC=2)C)=C1 BCTQJXQXJVLSIG-UHFFFAOYSA-N 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- HYVVJDQGXFXBRZ-UHFFFAOYSA-N metam Chemical compound CNC(S)=S HYVVJDQGXFXBRZ-UHFFFAOYSA-N 0.000 description 1
- XWPZUHJBOLQNMN-UHFFFAOYSA-N metconazole Chemical compound C1=NC=NN1CC1(O)C(C)(C)CCC1CC1=CC=C(Cl)C=C1 XWPZUHJBOLQNMN-UHFFFAOYSA-N 0.000 description 1
- NNKVPIKMPCQWCG-UHFFFAOYSA-N methamidophos Chemical compound COP(N)(=O)SC NNKVPIKMPCQWCG-UHFFFAOYSA-N 0.000 description 1
- 229940098779 methanesulfonic acid Drugs 0.000 description 1
- 125000005948 methanesulfonyloxy group Chemical group 0.000 description 1
- WSFSSNUMVMOOMR-NJFSPNSNSA-N methanone Chemical compound O=[14CH2] WSFSSNUMVMOOMR-NJFSPNSNSA-N 0.000 description 1
- IXJOSTZEBSTPAG-UHFFFAOYSA-N methasulfocarb Chemical compound CNC(=O)SC1=CC=C(OS(C)(=O)=O)C=C1 IXJOSTZEBSTPAG-UHFFFAOYSA-N 0.000 description 1
- MEBQXILRKZHVCX-UHFFFAOYSA-N methidathion Chemical compound COC1=NN(CSP(=S)(OC)OC)C(=O)S1 MEBQXILRKZHVCX-UHFFFAOYSA-N 0.000 description 1
- UHXUZOCRWCRNSJ-QPJJXVBHSA-N methomyl Chemical compound CNC(=O)O\N=C(/C)SC UHXUZOCRWCRNSJ-QPJJXVBHSA-N 0.000 description 1
- 229950003442 methoprene Drugs 0.000 description 1
- 229930002897 methoprene Natural products 0.000 description 1
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 1
- 125000001160 methoxycarbonyl group Chemical group [H]C([H])([H])OC(*)=O 0.000 description 1
- QCAWEPFNJXQPAN-UHFFFAOYSA-N methoxyfenozide Chemical compound COC1=CC=CC(C(=O)NN(C(=O)C=2C=C(C)C=C(C)C=2)C(C)(C)C)=C1C QCAWEPFNJXQPAN-UHFFFAOYSA-N 0.000 description 1
- KBHDSWIXRODKSZ-UHFFFAOYSA-N methyl 5-chloro-2-(trifluoromethylsulfonylamino)benzoate Chemical compound COC(=O)C1=CC(Cl)=CC=C1NS(=O)(=O)C(F)(F)F KBHDSWIXRODKSZ-UHFFFAOYSA-N 0.000 description 1
- ZQEIXNIJLIKNTD-UHFFFAOYSA-N methyl N-(2,6-dimethylphenyl)-N-(methoxyacetyl)alaninate Chemical compound COCC(=O)N(C(C)C(=O)OC)C1=C(C)C=CC=C1C ZQEIXNIJLIKNTD-UHFFFAOYSA-N 0.000 description 1
- CJPQIRJHIZUAQP-UHFFFAOYSA-N methyl N-(2,6-dimethylphenyl)-N-(phenylacetyl)alaninate Chemical compound CC=1C=CC=C(C)C=1N(C(C)C(=O)OC)C(=O)CC1=CC=CC=C1 CJPQIRJHIZUAQP-UHFFFAOYSA-N 0.000 description 1
- 229940102396 methyl bromide Drugs 0.000 description 1
- OIRDBPQYVWXNSJ-UHFFFAOYSA-N methyl trifluoromethansulfonate Chemical compound COS(=O)(=O)C(F)(F)F OIRDBPQYVWXNSJ-UHFFFAOYSA-N 0.000 description 1
- 125000006216 methylsulfinyl group Chemical group [H]C([H])([H])S(*)=O 0.000 description 1
- 125000004170 methylsulfonyl group Chemical group [H]C([H])([H])S(*)(=O)=O 0.000 description 1
- 229920000257 metiram Polymers 0.000 description 1
- AMSPWOYQQAWRRM-UHFFFAOYSA-N metrafenone Chemical compound COC1=CC=C(Br)C(C)=C1C(=O)C1=C(C)C=C(OC)C(OC)=C1OC AMSPWOYQQAWRRM-UHFFFAOYSA-N 0.000 description 1
- 229960001952 metrifonate Drugs 0.000 description 1
- 239000004530 micro-emulsion Substances 0.000 description 1
- ZLBGSRMUSVULIE-GSMJGMFJSA-N milbemycin A3 Chemical compound O1[C@H](C)[C@@H](C)CC[C@@]11O[C@H](C\C=C(C)\C[C@@H](C)\C=C\C=C/2[C@]3([C@H](C(=O)O4)C=C(C)[C@@H](O)[C@H]3OC\2)O)C[C@H]4C1 ZLBGSRMUSVULIE-GSMJGMFJSA-N 0.000 description 1
- 239000002480 mineral oil Substances 0.000 description 1
- 235000010446 mineral oil Nutrition 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- 230000003129 miticidal effect Effects 0.000 description 1
- 239000001788 mono and diglycerides of fatty acids Substances 0.000 description 1
- 150000005673 monoalkenes Chemical class 0.000 description 1
- 235000019837 monoammonium phosphate Nutrition 0.000 description 1
- KRTSDMXIXPKRQR-AATRIKPKSA-N monocrotophos Chemical compound CNC(=O)\C=C(/C)OP(=O)(OC)OC KRTSDMXIXPKRQR-AATRIKPKSA-N 0.000 description 1
- 238000000465 moulding Methods 0.000 description 1
- 235000020638 mussel Nutrition 0.000 description 1
- JEFUQUGZXLEHLD-UHFFFAOYSA-N n-[(5-chloro-2-propan-2-ylphenyl)methyl]-n-cyclopropyl-3-(difluoromethyl)-5-fluoro-1-methylpyrazole-4-carboxamide Chemical compound CC(C)C1=CC=C(Cl)C=C1CN(C(=O)C=1C(=NN(C)C=1F)C(F)F)C1CC1 JEFUQUGZXLEHLD-UHFFFAOYSA-N 0.000 description 1
- YNKFZRGTXAPYFD-UHFFFAOYSA-N n-[[2-chloro-3,5-bis(trifluoromethyl)phenyl]carbamoyl]-2,6-difluorobenzamide Chemical compound FC1=CC=CC(F)=C1C(=O)NC(=O)NC1=CC(C(F)(F)F)=CC(C(F)(F)F)=C1Cl YNKFZRGTXAPYFD-UHFFFAOYSA-N 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000006126 n-butyl sulfonyl group Chemical group 0.000 description 1
- OYRIKLVYHTWHCZ-UHFFFAOYSA-N n-cyclohexyl-2,5-dimethylfuran-3-carboxamide Chemical compound O1C(C)=CC(C(=O)NC2CCCCC2)=C1C OYRIKLVYHTWHCZ-UHFFFAOYSA-N 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- ARAFQFMXQDBEPU-UHFFFAOYSA-N n-methyl-5-(trifluoromethyl)-1,3,4-thiadiazol-2-amine Chemical compound CNC1=NN=C(C(F)(F)F)S1 ARAFQFMXQDBEPU-UHFFFAOYSA-N 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000006124 n-propyl sulfonyl group Chemical group 0.000 description 1
- UQJQVUOTMVCFHX-UHFFFAOYSA-L nabam Chemical compound [Na+].[Na+].[S-]C(=S)NCCNC([S-])=S UQJQVUOTMVCFHX-UHFFFAOYSA-L 0.000 description 1
- PSZYNBSKGUBXEH-UHFFFAOYSA-M naphthalene-1-sulfonate Chemical compound C1=CC=C2C(S(=O)(=O)[O-])=CC=CC2=C1 PSZYNBSKGUBXEH-UHFFFAOYSA-M 0.000 description 1
- PSZYNBSKGUBXEH-UHFFFAOYSA-N naphthalene-1-sulfonic acid Chemical compound C1=CC=C2C(S(=O)(=O)O)=CC=CC2=C1 PSZYNBSKGUBXEH-UHFFFAOYSA-N 0.000 description 1
- 235000010298 natamycin Nutrition 0.000 description 1
- 239000004311 natamycin Substances 0.000 description 1
- 229960003255 natamycin Drugs 0.000 description 1
- NCXMLFZGDNKEPB-FFPOYIOWSA-N natamycin Chemical compound O[C@H]1[C@@H](N)[C@H](O)[C@@H](C)O[C@H]1O[C@H]1/C=C/C=C/C=C/C=C/C[C@@H](C)OC(=O)/C=C/[C@H]2O[C@@H]2C[C@H](O)C[C@](O)(C[C@H](O)[C@H]2C(O)=O)O[C@H]2C1 NCXMLFZGDNKEPB-FFPOYIOWSA-N 0.000 description 1
- 229940079888 nitenpyram Drugs 0.000 description 1
- DCUJJWWUNKIJPH-UHFFFAOYSA-N nitrapyrin Chemical compound ClC1=CC=CC(C(Cl)(Cl)Cl)=N1 DCUJJWWUNKIJPH-UHFFFAOYSA-N 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- 150000002825 nitriles Chemical class 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 125000004433 nitrogen atom Chemical group N* 0.000 description 1
- 125000005246 nonafluorobutyl group Chemical group FC(F)(F)C(F)(F)C(F)(F)C(F)(F)* 0.000 description 1
- NJPPVKZQTLUDBO-UHFFFAOYSA-N novaluron Chemical compound C1=C(Cl)C(OC(F)(F)C(OC(F)(F)F)F)=CC=C1NC(=O)NC(=O)C1=C(F)C=CC=C1F NJPPVKZQTLUDBO-UHFFFAOYSA-N 0.000 description 1
- YTYGAJLZOJPJGH-UHFFFAOYSA-N noviflumuron Chemical compound FC1=C(Cl)C(OC(F)(F)C(C(F)(F)F)F)=C(Cl)C=C1NC(=O)NC(=O)C1=C(F)C=CC=C1F YTYGAJLZOJPJGH-UHFFFAOYSA-N 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 1
- JRZJOMJEPLMPRA-UHFFFAOYSA-N olefin Natural products CCCCCCCC=C JRZJOMJEPLMPRA-UHFFFAOYSA-N 0.000 description 1
- BARWIPMJPCRCTP-UHFFFAOYSA-N oleic acid oleyl ester Natural products CCCCCCCCC=CCCCCCCCCOC(=O)CCCCCCCC=CCCCCCCCC BARWIPMJPCRCTP-UHFFFAOYSA-N 0.000 description 1
- 229940055577 oleyl alcohol Drugs 0.000 description 1
- XMLQWXUVTXCDDL-UHFFFAOYSA-N oleyl alcohol Natural products CCCCCCC=CCCCCCCCCCCO XMLQWXUVTXCDDL-UHFFFAOYSA-N 0.000 description 1
- BARWIPMJPCRCTP-CLFAGFIQSA-N oleyl oleate Chemical compound CCCCCCCC\C=C/CCCCCCCCOC(=O)CCCCCCC\C=C/CCCCCCCC BARWIPMJPCRCTP-CLFAGFIQSA-N 0.000 description 1
- 239000004006 olive oil Substances 0.000 description 1
- 235000008390 olive oil Nutrition 0.000 description 1
- PZXOQEXFMJCDPG-UHFFFAOYSA-N omethoate Chemical compound CNC(=O)CSP(=O)(OC)OC PZXOQEXFMJCDPG-UHFFFAOYSA-N 0.000 description 1
- 150000002903 organophosphorus compounds Chemical class 0.000 description 1
- 235000010292 orthophenyl phenol Nutrition 0.000 description 1
- 239000004306 orthophenyl phenol Substances 0.000 description 1
- JHIPUJPTQJYEQK-ZLHHXESBSA-N orysastrobin Chemical compound CNC(=O)C(=N\OC)\C1=CC=CC=C1CO\N=C(/C)\C(=N\OC)\C(\C)=N\OC JHIPUJPTQJYEQK-ZLHHXESBSA-N 0.000 description 1
- 230000003204 osmotic effect Effects 0.000 description 1
- UWVQIROCRJWDKL-UHFFFAOYSA-N oxadixyl Chemical compound CC=1C=CC=C(C)C=1N(C(=O)COC)N1CCOC1=O UWVQIROCRJWDKL-UHFFFAOYSA-N 0.000 description 1
- 230000001590 oxidative effect Effects 0.000 description 1
- JCGNDDUYTRNOFT-UHFFFAOYSA-N oxolane-2,4-dione Chemical compound O=C1COC(=O)C1 JCGNDDUYTRNOFT-UHFFFAOYSA-N 0.000 description 1
- 229960000321 oxolinic acid Drugs 0.000 description 1
- AMEKQAFGQBKLKX-UHFFFAOYSA-N oxycarboxin Chemical compound O=S1(=O)CCOC(C)=C1C(=O)NC1=CC=CC=C1 AMEKQAFGQBKLKX-UHFFFAOYSA-N 0.000 description 1
- NFHFRUOZVGFOOS-UHFFFAOYSA-N palladium;triphenylphosphane Chemical compound [Pd].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 NFHFRUOZVGFOOS-UHFFFAOYSA-N 0.000 description 1
- 229910052625 palygorskite Inorganic materials 0.000 description 1
- FJKROLUGYXJWQN-UHFFFAOYSA-N papa-hydroxy-benzoic acid Natural products OC(=O)C1=CC=C(O)C=C1 FJKROLUGYXJWQN-UHFFFAOYSA-N 0.000 description 1
- 239000012188 paraffin wax Substances 0.000 description 1
- LCCNCVORNKJIRZ-UHFFFAOYSA-N parathion Chemical compound CCOP(=S)(OCC)OC1=CC=C([N+]([O-])=O)C=C1 LCCNCVORNKJIRZ-UHFFFAOYSA-N 0.000 description 1
- RLBIQVVOMOPOHC-UHFFFAOYSA-N parathion-methyl Chemical group COP(=S)(OC)OC1=CC=C([N+]([O-])=O)C=C1 RLBIQVVOMOPOHC-UHFFFAOYSA-N 0.000 description 1
- 238000007911 parenteral administration Methods 0.000 description 1
- 239000008188 pellet Substances 0.000 description 1
- LKPLKUMXSAEKID-UHFFFAOYSA-N pentachloronitrobenzene Chemical compound [O-][N+](=O)C1=C(Cl)C(Cl)=C(Cl)C(Cl)=C1Cl LKPLKUMXSAEKID-UHFFFAOYSA-N 0.000 description 1
- 125000006340 pentafluoro ethyl group Chemical group FC(F)(F)C(F)(F)* 0.000 description 1
- 239000010451 perlite Substances 0.000 description 1
- 235000019362 perlite Nutrition 0.000 description 1
- 229960000490 permethrin Drugs 0.000 description 1
- RLLPVAHGXHCWKJ-UHFFFAOYSA-N permethrin Chemical compound CC1(C)C(C=C(Cl)Cl)C1C(=O)OCC1=CC=CC(OC=2C=CC=CC=2)=C1 RLLPVAHGXHCWKJ-UHFFFAOYSA-N 0.000 description 1
- 150000004965 peroxy acids Chemical class 0.000 description 1
- 239000000575 pesticide Substances 0.000 description 1
- XAMUDJHXFNRLCY-UHFFFAOYSA-N phenthoate Chemical compound CCOC(=O)C(SP(=S)(OC)OC)C1=CC=CC=C1 XAMUDJHXFNRLCY-UHFFFAOYSA-N 0.000 description 1
- BULVZWIRKLYCBC-UHFFFAOYSA-N phorate Chemical compound CCOP(=S)(OCC)SCSCC BULVZWIRKLYCBC-UHFFFAOYSA-N 0.000 description 1
- IOUNQDKNJZEDEP-UHFFFAOYSA-N phosalone Chemical compound C1=C(Cl)C=C2OC(=O)N(CSP(=S)(OCC)OCC)C2=C1 IOUNQDKNJZEDEP-UHFFFAOYSA-N 0.000 description 1
- LMNZTLDVJIUSHT-UHFFFAOYSA-N phosmet Chemical compound C1=CC=C2C(=O)N(CSP(=S)(OC)OC)C(=O)C2=C1 LMNZTLDVJIUSHT-UHFFFAOYSA-N 0.000 description 1
- RGCLLPNLLBQHPF-HJWRWDBZSA-N phosphamidon Chemical compound CCN(CC)C(=O)C(\Cl)=C(/C)OP(=O)(OC)OC RGCLLPNLLBQHPF-HJWRWDBZSA-N 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- ATROHALUCMTWTB-OWBHPGMISA-N phoxim Chemical compound CCOP(=S)(OCC)O\N=C(\C#N)C1=CC=CC=C1 ATROHALUCMTWTB-OWBHPGMISA-N 0.000 description 1
- 229950001664 phoxim Drugs 0.000 description 1
- 125000005498 phthalate group Chemical class 0.000 description 1
- URHWNXDZOULUHC-ULJHMMPZSA-N picarbutrazox Chemical compound CN1N=NN=C1\C(C=1C=CC=CC=1)=N/OCC1=CC=CC(NC(=O)OC(C)(C)C)=N1 URHWNXDZOULUHC-ULJHMMPZSA-N 0.000 description 1
- SIOXPEMLGUPBBT-UHFFFAOYSA-M picolinate Chemical compound [O-]C(=O)C1=CC=CC=N1 SIOXPEMLGUPBBT-UHFFFAOYSA-M 0.000 description 1
- IBSNKSODLGJUMQ-SDNWHVSQSA-N picoxystrobin Chemical compound CO\C=C(\C(=O)OC)C1=CC=CC=C1COC1=CC=CC(C(F)(F)F)=N1 IBSNKSODLGJUMQ-SDNWHVSQSA-N 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- YFGYUFNIOHWBOB-UHFFFAOYSA-N pirimicarb Chemical compound CN(C)C(=O)OC1=NC(N(C)C)=NC(C)=C1C YFGYUFNIOHWBOB-UHFFFAOYSA-N 0.000 description 1
- QHOQHJPRIBSPCY-UHFFFAOYSA-N pirimiphos-methyl Chemical group CCN(CC)C1=NC(C)=CC(OP(=S)(OC)OC)=N1 QHOQHJPRIBSPCY-UHFFFAOYSA-N 0.000 description 1
- 239000005648 plant growth regulator Substances 0.000 description 1
- 239000003375 plant hormone Substances 0.000 description 1
- 229920001467 poly(styrenesulfonates) Polymers 0.000 description 1
- 229920005646 polycarboxylate Polymers 0.000 description 1
- 229920000259 polyoxyethylene lauryl ether Polymers 0.000 description 1
- 229920002503 polyoxyethylene-polyoxypropylene Polymers 0.000 description 1
- 229920001282 polysaccharide Polymers 0.000 description 1
- 239000005017 polysaccharide Substances 0.000 description 1
- 150000004804 polysaccharides Chemical class 0.000 description 1
- 239000011970 polystyrene sulfonate Substances 0.000 description 1
- 229960002796 polystyrene sulfonate Drugs 0.000 description 1
- 229920002635 polyurethane Polymers 0.000 description 1
- 229910052573 porcelain Inorganic materials 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229960003975 potassium Drugs 0.000 description 1
- TZLVRPLSVNESQC-UHFFFAOYSA-N potassium azide Chemical compound [K+].[N-]=[N+]=[N-] TZLVRPLSVNESQC-UHFFFAOYSA-N 0.000 description 1
- 239000011736 potassium bicarbonate Substances 0.000 description 1
- 235000015497 potassium bicarbonate Nutrition 0.000 description 1
- 229910000028 potassium bicarbonate Inorganic materials 0.000 description 1
- 239000001103 potassium chloride Substances 0.000 description 1
- 235000011164 potassium chloride Nutrition 0.000 description 1
- TYJJADVDDVDEDZ-UHFFFAOYSA-M potassium hydrogencarbonate Chemical compound [K+].OC([O-])=O TYJJADVDDVDEDZ-UHFFFAOYSA-M 0.000 description 1
- 229940086066 potassium hydrogencarbonate Drugs 0.000 description 1
- ZNNZYHKDIALBAK-UHFFFAOYSA-M potassium thiocyanate Chemical compound [K+].[S-]C#N ZNNZYHKDIALBAK-UHFFFAOYSA-M 0.000 description 1
- 229940116357 potassium thiocyanate Drugs 0.000 description 1
- HDMGAZBPFLDBCX-UHFFFAOYSA-N potassium;sulfooxy hydrogen sulfate Chemical compound [K+].OS(=O)(=O)OOS(O)(=O)=O HDMGAZBPFLDBCX-UHFFFAOYSA-N 0.000 description 1
- 229920001592 potato starch Polymers 0.000 description 1
- 229940116317 potato starch Drugs 0.000 description 1
- 244000144977 poultry Species 0.000 description 1
- 125000001844 prenyl group Chemical group [H]C([*])([H])C([H])=C(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- WHHIPMZEDGBUCC-UHFFFAOYSA-N probenazole Chemical compound C1=CC=C2C(OCC=C)=NS(=O)(=O)C2=C1 WHHIPMZEDGBUCC-UHFFFAOYSA-N 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- QXJKBPAVAHBARF-BETUJISGSA-N procymidone Chemical compound O=C([C@]1(C)C[C@@]1(C1=O)C)N1C1=CC(Cl)=CC(Cl)=C1 QXJKBPAVAHBARF-BETUJISGSA-N 0.000 description 1
- QYMMJNLHFKGANY-UHFFFAOYSA-N profenofos Chemical compound CCCSP(=O)(OCC)OC1=CC=C(Br)C=C1Cl QYMMJNLHFKGANY-UHFFFAOYSA-N 0.000 description 1
- ZPWFUIUNWDIYCJ-UHFFFAOYSA-N propan-2-yl octadecanoate Chemical compound CCCCCCCCCCCCCCCCCC(=O)OC(C)C ZPWFUIUNWDIYCJ-UHFFFAOYSA-N 0.000 description 1
- 125000001325 propanoyl group Chemical group O=C([*])C([H])([H])C([H])([H])[H] 0.000 description 1
- PWYIUEFFPNVCMW-UHFFFAOYSA-N propaphos Chemical compound CCCOP(=O)(OCCC)OC1=CC=C(SC)C=C1 PWYIUEFFPNVCMW-UHFFFAOYSA-N 0.000 description 1
- ZYHMJXZULPZUED-UHFFFAOYSA-N propargite Chemical compound C1=CC(C(C)(C)C)=CC=C1OC1C(OS(=O)OCC#C)CCCC1 ZYHMJXZULPZUED-UHFFFAOYSA-N 0.000 description 1
- STJLVHWMYQXCPB-UHFFFAOYSA-N propiconazole Chemical compound O1C(CCC)COC1(C=1C(=CC(Cl)=CC=1)Cl)CN1N=CN=C1 STJLVHWMYQXCPB-UHFFFAOYSA-N 0.000 description 1
- KKMLIVYBGSAJPM-UHFFFAOYSA-L propineb Chemical compound [Zn+2].[S-]C(=S)NC(C)CNC([S-])=S KKMLIVYBGSAJPM-UHFFFAOYSA-L 0.000 description 1
- 235000019260 propionic acid Nutrition 0.000 description 1
- 125000001501 propionyl group Chemical group O=C([*])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000002572 propoxy group Chemical group [*]OC([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 1
- 235000013772 propylene glycol Nutrition 0.000 description 1
- 125000004742 propyloxycarbonyl group Chemical group 0.000 description 1
- FLVBXVXXXMLMOX-UHFFFAOYSA-N proquinazid Chemical compound C1=C(I)C=C2C(=O)N(CCC)C(OCCC)=NC2=C1 FLVBXVXXXMLMOX-UHFFFAOYSA-N 0.000 description 1
- YRRBXJLFCBCKNW-UHFFFAOYSA-N prothiocarb Chemical compound CCSC(=O)NCCCN(C)C YRRBXJLFCBCKNW-UHFFFAOYSA-N 0.000 description 1
- FITIWKDOCAUBQD-UHFFFAOYSA-N prothiofos Chemical compound CCCSP(=S)(OCC)OC1=CC=C(Cl)C=C1Cl FITIWKDOCAUBQD-UHFFFAOYSA-N 0.000 description 1
- 238000000425 proton nuclear magnetic resonance spectrum Methods 0.000 description 1
- 239000008262 pumice Substances 0.000 description 1
- QHMTXANCGGJZRX-WUXMJOGZSA-N pymetrozine Chemical compound C1C(C)=NNC(=O)N1\N=C\C1=CC=CN=C1 QHMTXANCGGJZRX-WUXMJOGZSA-N 0.000 description 1
- QHGVXILFMXYDRS-UHFFFAOYSA-N pyraclofos Chemical compound C1=C(OP(=O)(OCC)SCCC)C=NN1C1=CC=C(Cl)C=C1 QHGVXILFMXYDRS-UHFFFAOYSA-N 0.000 description 1
- HZRSNVGNWUDEFX-UHFFFAOYSA-N pyraclostrobin Chemical compound COC(=O)N(OC)C1=CC=CC=C1COC1=NN(C=2C=CC(Cl)=CC=2)C=C1 HZRSNVGNWUDEFX-UHFFFAOYSA-N 0.000 description 1
- DDIQWGKUSJOETH-UHFFFAOYSA-N pyrafluprole Chemical compound ClC=1C=C(C(F)(F)F)C=C(Cl)C=1N1N=C(C#N)C(SCF)=C1NCC1=CN=CC=N1 DDIQWGKUSJOETH-UHFFFAOYSA-N 0.000 description 1
- DWTVBEZBWMDXIY-UHFFFAOYSA-N pyrametostrobin Chemical compound COC(=O)N(OC)C1=CC=CC=C1COC1=C(C)C(C=2C=CC=CC=2)=NN1C DWTVBEZBWMDXIY-UHFFFAOYSA-N 0.000 description 1
- URXNNPCNKVAQRA-XMHGGMMESA-N pyraoxystrobin Chemical compound CO\C=C(\C(=O)OC)C1=CC=CC=C1COC1=CC(C=2C=CC(Cl)=CC=2)=NN1C URXNNPCNKVAQRA-XMHGGMMESA-N 0.000 description 1
- KKEJMLAPZVXPOF-UHFFFAOYSA-N pyraziflumid Chemical compound C1=C(F)C(F)=CC=C1C1=CC=CC=C1NC(=O)C1=NC=CN=C1C(F)(F)F KKEJMLAPZVXPOF-UHFFFAOYSA-N 0.000 description 1
- JOOMJVFZQRQWKR-UHFFFAOYSA-N pyrazophos Chemical compound N1=C(C)C(C(=O)OCC)=CN2N=C(OP(=S)(OCC)OCC)C=C21 JOOMJVFZQRQWKR-UHFFFAOYSA-N 0.000 description 1
- HYJYGLGUBUDSLJ-UHFFFAOYSA-N pyrethrin Natural products CCC(=O)OC1CC(=C)C2CC3OC3(C)C2C2OC(=O)C(=C)C12 HYJYGLGUBUDSLJ-UHFFFAOYSA-N 0.000 description 1
- 229940070846 pyrethrins Drugs 0.000 description 1
- AEHJMNVBLRLZKK-UHFFFAOYSA-N pyridalyl Chemical group N1=CC(C(F)(F)F)=CC=C1OCCCOC1=C(Cl)C=C(OCC=C(Cl)Cl)C=C1Cl AEHJMNVBLRLZKK-UHFFFAOYSA-N 0.000 description 1
- GPHQHTOMRSGBNZ-UHFFFAOYSA-N pyridine-4-carbonitrile Chemical compound N#CC1=CC=NC=C1 GPHQHTOMRSGBNZ-UHFFFAOYSA-N 0.000 description 1
- ZLIBICFPKPWGIZ-UHFFFAOYSA-N pyrimethanil Chemical compound CC1=CC(C)=NC(NC=2C=CC=CC=2)=N1 ZLIBICFPKPWGIZ-UHFFFAOYSA-N 0.000 description 1
- ITKAIUGKVKDENI-UHFFFAOYSA-N pyrimidifen Chemical compound CC1=C(C)C(CCOCC)=CC=C1OCCNC1=NC=NC(CC)=C1Cl ITKAIUGKVKDENI-UHFFFAOYSA-N 0.000 description 1
- YYXSCUSVVALMNW-FOWTUZBSSA-N pyriminostrobin Chemical compound CO\C=C(\C(=O)OC)C1=CC=CC=C1COC1=CC(C(F)(F)F)=NC(NC=2C(=CC(Cl)=CC=2)Cl)=N1 YYXSCUSVVALMNW-FOWTUZBSSA-N 0.000 description 1
- BAUQXSYUDSNRHL-UHFFFAOYSA-N pyrimorph Chemical compound C1=CC(C(C)(C)C)=CC=C1C(C=1C=NC(Cl)=CC=1)=CC(=O)N1CCOCC1 BAUQXSYUDSNRHL-UHFFFAOYSA-N 0.000 description 1
- NMVCBWZLCXANER-UHFFFAOYSA-N pyriofenone Chemical compound COC1=C(OC)C(OC)=CC(C)=C1C(=O)C1=C(C)C(Cl)=CN=C1OC NMVCBWZLCXANER-UHFFFAOYSA-N 0.000 description 1
- NHDHVHZZCFYRSB-UHFFFAOYSA-N pyriproxyfen Chemical compound C=1C=CC=NC=1OC(C)COC(C=C1)=CC=C1OC1=CC=CC=C1 NHDHVHZZCFYRSB-UHFFFAOYSA-N 0.000 description 1
- 229960003811 pyrithione disulfide Drugs 0.000 description 1
- 239000010453 quartz Substances 0.000 description 1
- 150000003242 quaternary ammonium salts Chemical class 0.000 description 1
- IUVKMZGDUIUOCP-BTNSXGMBSA-N quinbolone Chemical compound O([C@H]1CC[C@H]2[C@H]3[C@@H]([C@]4(C=CC(=O)C=C4CC3)C)CC[C@@]21C)C1=CCCC1 IUVKMZGDUIUOCP-BTNSXGMBSA-N 0.000 description 1
- HSNIRAAZKNNQDA-UHFFFAOYSA-N quinolin-8-ol 1H-quinolin-2-one sulfuric acid Chemical compound OS(O)(=O)=O.Oc1ccc2ccccc2n1.Oc1cccc2cccnc12 HSNIRAAZKNNQDA-UHFFFAOYSA-N 0.000 description 1
- WRPIRSINYZBGPK-UHFFFAOYSA-N quinoxyfen Chemical compound C1=CC(F)=CC=C1OC1=CC=NC2=CC(Cl)=CC(Cl)=C12 WRPIRSINYZBGPK-UHFFFAOYSA-N 0.000 description 1
- RDYMFSUJUZBWLH-AZVNHNRSSA-N qy5y9r7g0e Chemical compound C([C@H]12)OS(=O)OC[C@@H]1[C@]1(Cl)C(Cl)=C(Cl)[C@@]2(Cl)C1(Cl)Cl RDYMFSUJUZBWLH-AZVNHNRSSA-N 0.000 description 1
- 238000001953 recrystallisation Methods 0.000 description 1
- 239000013558 reference substance Substances 0.000 description 1
- 229940108410 resmethrin Drugs 0.000 description 1
- VEMKTZHHVJILDY-FIWHBWSRSA-N resmethrin Chemical compound CC1(C)[C@H](C=C(C)C)C1C(=O)OCC1=COC(CC=2C=CC=CC=2)=C1 VEMKTZHHVJILDY-FIWHBWSRSA-N 0.000 description 1
- 229940100486 rice starch Drugs 0.000 description 1
- 229940080817 rotenone Drugs 0.000 description 1
- JUVIOZPCNVVQFO-UHFFFAOYSA-N rotenone Natural products O1C2=C3CC(C(C)=C)OC3=CC=C2C(=O)C2C1COC1=C2C=C(OC)C(OC)=C1 JUVIOZPCNVVQFO-UHFFFAOYSA-N 0.000 description 1
- 239000010979 ruby Substances 0.000 description 1
- 229910001750 ruby Inorganic materials 0.000 description 1
- WKEDVNSFRWHDNR-UHFFFAOYSA-N salicylanilide Chemical compound OC1=CC=CC=C1C(=O)NC1=CC=CC=C1 WKEDVNSFRWHDNR-UHFFFAOYSA-N 0.000 description 1
- 229950000975 salicylanilide Drugs 0.000 description 1
- 150000004671 saturated fatty acids Chemical class 0.000 description 1
- 235000020637 scallop Nutrition 0.000 description 1
- 201000004409 schistosomiasis Diseases 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 239000008159 sesame oil Substances 0.000 description 1
- 235000011803 sesame oil Nutrition 0.000 description 1
- HPYNBECUCCGGPA-UHFFFAOYSA-N silafluofen Chemical compound C1=CC(OCC)=CC=C1[Si](C)(C)CCCC1=CC=C(F)C(OC=2C=CC=CC=2)=C1 HPYNBECUCCGGPA-UHFFFAOYSA-N 0.000 description 1
- 239000000741 silica gel Substances 0.000 description 1
- 229910002027 silica gel Inorganic materials 0.000 description 1
- MXMXHPPIGKYTAR-UHFFFAOYSA-N silthiofam Chemical compound CC=1SC([Si](C)(C)C)=C(C(=O)NCC=C)C=1C MXMXHPPIGKYTAR-UHFFFAOYSA-N 0.000 description 1
- 235000015424 sodium Nutrition 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910000029 sodium carbonate Inorganic materials 0.000 description 1
- 235000017550 sodium carbonate Nutrition 0.000 description 1
- 229940079827 sodium hydrogen sulfite Drugs 0.000 description 1
- 235000010267 sodium hydrogen sulphite Nutrition 0.000 description 1
- SUKJFIGYRHOWBL-UHFFFAOYSA-N sodium hypochlorite Chemical compound [Na+].Cl[O-] SUKJFIGYRHOWBL-UHFFFAOYSA-N 0.000 description 1
- 235000019333 sodium laurylsulphate Nutrition 0.000 description 1
- 159000000000 sodium salts Chemical class 0.000 description 1
- 229910052938 sodium sulfate Inorganic materials 0.000 description 1
- 235000011152 sodium sulphate Nutrition 0.000 description 1
- VGTPCRGMBIAPIM-UHFFFAOYSA-M sodium thiocyanate Chemical compound [Na+].[S-]C#N VGTPCRGMBIAPIM-UHFFFAOYSA-M 0.000 description 1
- XMVONEAAOPAGAO-UHFFFAOYSA-N sodium tungstate Chemical compound [Na+].[Na+].[O-][W]([O-])(=O)=O XMVONEAAOPAGAO-UHFFFAOYSA-N 0.000 description 1
- PYODKQIVQIVELM-UHFFFAOYSA-M sodium;2,3-bis(2-methylpropyl)naphthalene-1-sulfonate Chemical compound [Na+].C1=CC=C2C(S([O-])(=O)=O)=C(CC(C)C)C(CC(C)C)=CC2=C1 PYODKQIVQIVELM-UHFFFAOYSA-M 0.000 description 1
- 239000003516 soil conditioner Substances 0.000 description 1
- 239000007909 solid dosage form Substances 0.000 description 1
- 239000004550 soluble concentrate Substances 0.000 description 1
- 239000011877 solvent mixture Substances 0.000 description 1
- 239000001587 sorbitan monostearate Substances 0.000 description 1
- 235000011076 sorbitan monostearate Nutrition 0.000 description 1
- 229940035048 sorbitan monostearate Drugs 0.000 description 1
- 239000003549 soybean oil Substances 0.000 description 1
- 235000012424 soybean oil Nutrition 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 238000001228 spectrum Methods 0.000 description 1
- 229940014213 spinosad Drugs 0.000 description 1
- DTDSAWVUFPGDMX-UHFFFAOYSA-N spirodiclofen Chemical compound CCC(C)(C)C(=O)OC1=C(C=2C(=CC(Cl)=CC=2)Cl)C(=O)OC11CCCCC1 DTDSAWVUFPGDMX-UHFFFAOYSA-N 0.000 description 1
- GOLXNESZZPUPJE-UHFFFAOYSA-N spiromesifen Chemical compound CC1=CC(C)=CC(C)=C1C(C(O1)=O)=C(OC(=O)CC(C)(C)C)C11CCCC1 GOLXNESZZPUPJE-UHFFFAOYSA-N 0.000 description 1
- WOPFPAIGRGHWAQ-UHFFFAOYSA-N spiropidion Chemical compound CCOC(=O)OC1=C(C=2C(=CC(Cl)=CC=2C)C)C(=O)N(C)C11CCN(OC)CC1 WOPFPAIGRGHWAQ-UHFFFAOYSA-N 0.000 description 1
- CLSVJBIHYWPGQY-GGYDESQDSA-N spirotetramat Chemical compound CCOC(=O)OC1=C(C=2C(=CC=C(C)C=2)C)C(=O)N[C@@]11CC[C@H](OC)CC1 CLSVJBIHYWPGQY-GGYDESQDSA-N 0.000 description 1
- PUYXTUJWRLOUCW-UHFFFAOYSA-N spiroxamine Chemical compound O1C(CN(CC)CCC)COC11CCC(C(C)(C)C)CC1 PUYXTUJWRLOUCW-UHFFFAOYSA-N 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 239000004575 stone Substances 0.000 description 1
- 229960005322 streptomycin Drugs 0.000 description 1
- 238000007920 subcutaneous administration Methods 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-L succinate(2-) Chemical compound [O-]C(=O)CCC([O-])=O KDYFGRWQOYBRFD-UHFFFAOYSA-L 0.000 description 1
- 239000001384 succinic acid Substances 0.000 description 1
- 235000011044 succinic acid Nutrition 0.000 description 1
- 229960005137 succinic acid Drugs 0.000 description 1
- 150000005846 sugar alcohols Polymers 0.000 description 1
- 150000003460 sulfonic acids Chemical class 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- 229910052717 sulfur Inorganic materials 0.000 description 1
- 125000004434 sulfur atom Chemical group 0.000 description 1
- JXHJNEJVUNHLKO-UHFFFAOYSA-N sulprofos Chemical compound CCCSP(=S)(OCC)OC1=CC=C(SC)C=C1 JXHJNEJVUNHLKO-UHFFFAOYSA-N 0.000 description 1
- 239000000375 suspending agent Substances 0.000 description 1
- 239000004548 suspo-emulsion Substances 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 229920001059 synthetic polymer Polymers 0.000 description 1
- 239000008399 tap water Substances 0.000 description 1
- 235000020679 tap water Nutrition 0.000 description 1
- 239000011975 tartaric acid Substances 0.000 description 1
- 229960001367 tartaric acid Drugs 0.000 description 1
- 235000002906 tartaric acid Nutrition 0.000 description 1
- 239000005936 tau-Fluvalinate Substances 0.000 description 1
- INISTDXBRIBGOC-XMMISQBUSA-N tau-fluvalinate Chemical compound N([C@H](C(C)C)C(=O)OC(C#N)C=1C=C(OC=2C=CC=CC=2)C=CC=1)C1=CC=C(C(F)(F)F)C=C1Cl INISTDXBRIBGOC-XMMISQBUSA-N 0.000 description 1
- ZZYSLNWGKKDOML-UHFFFAOYSA-N tebufenpyrad Chemical compound CCC1=NN(C)C(C(=O)NCC=2C=CC(=CC=2)C(C)(C)C)=C1Cl ZZYSLNWGKKDOML-UHFFFAOYSA-N 0.000 description 1
- LWLJEQHTPVPKSJ-UHFFFAOYSA-N tebufloquin Chemical compound C1=C(C(C)(C)C)C=C2C(OC(=O)C)=C(C)C(C)=NC2=C1F LWLJEQHTPVPKSJ-UHFFFAOYSA-N 0.000 description 1
- ROZUQUDEWZIBHV-UHFFFAOYSA-N tecloftalam Chemical compound OC(=O)C1=C(Cl)C(Cl)=C(Cl)C(Cl)=C1C(=O)NC1=CC=CC(Cl)=C1Cl ROZUQUDEWZIBHV-UHFFFAOYSA-N 0.000 description 1
- XQTLDIFVVHJORV-UHFFFAOYSA-N tecnazene Chemical compound [O-][N+](=O)C1=C(Cl)C(Cl)=CC(Cl)=C1Cl XQTLDIFVVHJORV-UHFFFAOYSA-N 0.000 description 1
- CJDWRQLODFKPEL-UHFFFAOYSA-N teflubenzuron Chemical compound FC1=CC=CC(F)=C1C(=O)NC(=O)NC1=CC(Cl)=C(F)C(Cl)=C1F CJDWRQLODFKPEL-UHFFFAOYSA-N 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000005931 tert-butyloxycarbonyl group Chemical group [H]C([H])([H])C(OC(*)=O)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000001973 tert-pentyl group Chemical group [H]C([H])([H])C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- DZLFLBLQUQXARW-UHFFFAOYSA-N tetrabutylammonium Chemical class CCCC[N+](CCCC)(CCCC)CCCC DZLFLBLQUQXARW-UHFFFAOYSA-N 0.000 description 1
- UGNWTBMOAKPKBL-UHFFFAOYSA-N tetrachloro-1,4-benzoquinone Chemical compound ClC1=C(Cl)C(=O)C(Cl)=C(Cl)C1=O UGNWTBMOAKPKBL-UHFFFAOYSA-N 0.000 description 1
- UBCKGWBNUIFUST-YHYXMXQVSA-N tetrachlorvinphos Chemical compound COP(=O)(OC)O\C(=C/Cl)C1=CC(Cl)=C(Cl)C=C1Cl UBCKGWBNUIFUST-YHYXMXQVSA-N 0.000 description 1
- QEMXHQIAXOOASZ-UHFFFAOYSA-N tetramethylammonium Chemical class C[N+](C)(C)C QEMXHQIAXOOASZ-UHFFFAOYSA-N 0.000 description 1
- CZDYPVPMEAXLPK-UHFFFAOYSA-N tetramethylsilane Chemical compound C[Si](C)(C)C CZDYPVPMEAXLPK-UHFFFAOYSA-N 0.000 description 1
- KNDVJPKNBVIKML-UHFFFAOYSA-N tetraniliprole Chemical compound CNC(=O)C1=CC(C#N)=CC(C)=C1NC(=O)C1=CC(CN2N=C(N=N2)C(F)(F)F)=NN1C1=NC=CC=C1Cl KNDVJPKNBVIKML-UHFFFAOYSA-N 0.000 description 1
- OULAJFUGPPVRBK-UHFFFAOYSA-N tetratriacontan-1-ol Chemical compound CCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCO OULAJFUGPPVRBK-UHFFFAOYSA-N 0.000 description 1
- 230000008719 thickening Effects 0.000 description 1
- WOSNCVAPUOFXEH-UHFFFAOYSA-N thifluzamide Chemical compound S1C(C)=NC(C(F)(F)F)=C1C(=O)NC1=C(Br)C=C(OC(F)(F)F)C=C1Br WOSNCVAPUOFXEH-UHFFFAOYSA-N 0.000 description 1
- DNVLJEWNNDHELH-UHFFFAOYSA-N thiocyclam Chemical compound CN(C)C1CSSSC1 DNVLJEWNNDHELH-UHFFFAOYSA-N 0.000 description 1
- BAKXBZPQTXCKRR-UHFFFAOYSA-N thiodicarb Chemical compound CSC(C)=NOC(=O)NSNC(=O)ON=C(C)SC BAKXBZPQTXCKRR-UHFFFAOYSA-N 0.000 description 1
- OPASCBHCTNRLRM-UHFFFAOYSA-N thiometon Chemical compound CCSCCSP(=S)(OC)OC OPASCBHCTNRLRM-UHFFFAOYSA-N 0.000 description 1
- YFNCATAIYKQPOO-UHFFFAOYSA-N thiophanate Chemical compound CCOC(=O)NC(=S)NC1=CC=CC=C1NC(=S)NC(=O)OCC YFNCATAIYKQPOO-UHFFFAOYSA-N 0.000 description 1
- QGHREAKMXXNCOA-UHFFFAOYSA-N thiophanate-methyl Chemical compound COC(=O)NC(=S)NC1=CC=CC=C1NC(=S)NC(=O)OC QGHREAKMXXNCOA-UHFFFAOYSA-N 0.000 description 1
- ILERPRJWJPJZDN-UHFFFAOYSA-N thioquinox Chemical compound C1=CC=C2N=C(SC(=S)S3)C3=NC2=C1 ILERPRJWJPJZDN-UHFFFAOYSA-N 0.000 description 1
- 229960002447 thiram Drugs 0.000 description 1
- KUAZQDVKQLNFPE-UHFFFAOYSA-N thiram Chemical compound CN(C)C(=S)SSC(=S)N(C)C KUAZQDVKQLNFPE-UHFFFAOYSA-N 0.000 description 1
- VJQYLJSMBWXGDV-UHFFFAOYSA-N tiadinil Chemical compound N1=NSC(C(=O)NC=2C=C(Cl)C(C)=CC=2)=C1C VJQYLJSMBWXGDV-UHFFFAOYSA-N 0.000 description 1
- IHNSIFFSNUQGQN-UHFFFAOYSA-N tioxazafen Chemical compound C1=CSC(C=2ON=C(N=2)C=2C=CC=CC=2)=C1 IHNSIFFSNUQGQN-UHFFFAOYSA-N 0.000 description 1
- 235000010384 tocopherol Nutrition 0.000 description 1
- 229960001295 tocopherol Drugs 0.000 description 1
- 229930003799 tocopherol Natural products 0.000 description 1
- 239000011732 tocopherol Substances 0.000 description 1
- OBZIQQJJIKNWNO-UHFFFAOYSA-N tolclofos-methyl Chemical compound COP(=S)(OC)OC1=C(Cl)C=C(C)C=C1Cl OBZIQQJJIKNWNO-UHFFFAOYSA-N 0.000 description 1
- WPALTCMYPARVNV-UHFFFAOYSA-N tolfenpyrad Chemical compound CCC1=NN(C)C(C(=O)NCC=2C=CC(OC=3C=CC(C)=CC=3)=CC=2)=C1Cl WPALTCMYPARVNV-UHFFFAOYSA-N 0.000 description 1
- RSOBJVBYZCMJOS-CYBMUJFWSA-N tolprocarb Chemical compound FC(F)(F)COC(=O)N[C@@H](C(C)C)CNC(=O)C1=CC=C(C)C=C1 RSOBJVBYZCMJOS-CYBMUJFWSA-N 0.000 description 1
- HYVWIQDYBVKITD-UHFFFAOYSA-N tolylfluanid Chemical compound CN(C)S(=O)(=O)N(SC(F)(Cl)Cl)C1=CC=C(C)C=C1 HYVWIQDYBVKITD-UHFFFAOYSA-N 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 238000012549 training Methods 0.000 description 1
- YWSCPYYRJXKUDB-KAKFPZCNSA-N tralomethrin Chemical compound CC1(C)[C@@H](C(Br)C(Br)(Br)Br)[C@H]1C(=O)O[C@H](C#N)C1=CC=CC(OC=2C=CC=CC=2)=C1 YWSCPYYRJXKUDB-KAKFPZCNSA-N 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 150000003626 triacylglycerols Chemical class 0.000 description 1
- BAZVSMNPJJMILC-UHFFFAOYSA-N triadimenol Chemical compound C1=NC=NN1C(C(O)C(C)(C)C)OC1=CC=C(Cl)C=C1 BAZVSMNPJJMILC-UHFFFAOYSA-N 0.000 description 1
- IQGKIPDJXCAMSM-UHFFFAOYSA-N triazoxide Chemical compound N=1C2=CC=C(Cl)C=C2[N+]([O-])=NC=1N1C=CN=C1 IQGKIPDJXCAMSM-UHFFFAOYSA-N 0.000 description 1
- NFACJZMKEDPNKN-UHFFFAOYSA-N trichlorfon Chemical compound COP(=O)(OC)C(O)C(Cl)(Cl)Cl NFACJZMKEDPNKN-UHFFFAOYSA-N 0.000 description 1
- 125000003866 trichloromethyl group Chemical group ClC(Cl)(Cl)* 0.000 description 1
- QFNFRZHOXWNWAQ-UHFFFAOYSA-N triclopyricarb Chemical compound COC(=O)N(OC)C1=CC=CC=C1COC1=NC(Cl)=C(Cl)C=C1Cl QFNFRZHOXWNWAQ-UHFFFAOYSA-N 0.000 description 1
- IGOWHGRNPLFNDJ-UHFFFAOYSA-N tricos-9t-ene Natural products CCCCCCCCCCCCCC=CCCCCCCCC IGOWHGRNPLFNDJ-UHFFFAOYSA-N 0.000 description 1
- DQJCHOQLCLEDLL-UHFFFAOYSA-N tricyclazole Chemical compound CC1=CC=CC2=C1N1C=NN=C1S2 DQJCHOQLCLEDLL-UHFFFAOYSA-N 0.000 description 1
- ITMCEJHCFYSIIV-UHFFFAOYSA-N triflic acid Chemical compound OS(=O)(=O)C(F)(F)F ITMCEJHCFYSIIV-UHFFFAOYSA-N 0.000 description 1
- ONCZDRURRATYFI-TVJDWZFNSA-N trifloxystrobin Chemical compound CO\N=C(\C(=O)OC)C1=CC=CC=C1CO\N=C(/C)C1=CC=CC(C(F)(F)F)=C1 ONCZDRURRATYFI-TVJDWZFNSA-N 0.000 description 1
- HSMVPDGQOIQYSR-KGENOOAVSA-N triflumizole Chemical compound C1=CN=CN1C(/COCCC)=N/C1=CC=C(Cl)C=C1C(F)(F)F HSMVPDGQOIQYSR-KGENOOAVSA-N 0.000 description 1
- XAIPTRIXGHTTNT-UHFFFAOYSA-N triflumuron Chemical compound C1=CC(OC(F)(F)F)=CC=C1NC(=O)NC(=O)C1=CC=CC=C1Cl XAIPTRIXGHTTNT-UHFFFAOYSA-N 0.000 description 1
- 125000004044 trifluoroacetyl group Chemical group FC(C(=O)*)(F)F 0.000 description 1
- 125000005951 trifluoromethanesulfonyloxy group Chemical group 0.000 description 1
- 125000000876 trifluoromethoxy group Chemical group FC(F)(F)O* 0.000 description 1
- RROQIUMZODEXOR-UHFFFAOYSA-N triforine Chemical compound O=CNC(C(Cl)(Cl)Cl)N1CCN(C(NC=O)C(Cl)(Cl)Cl)CC1 RROQIUMZODEXOR-UHFFFAOYSA-N 0.000 description 1
- UFTFJSFQGQCHQW-UHFFFAOYSA-N triformin Chemical compound O=COCC(OC=O)COC=O UFTFJSFQGQCHQW-UHFFFAOYSA-N 0.000 description 1
- 150000003672 ureas Chemical class 0.000 description 1
- 125000003774 valeryl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- JARYYMUOCXVXNK-IMTORBKUSA-N validamycin Chemical compound N([C@H]1C[C@@H]([C@H]([C@H](O)[C@H]1O)O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)CO)[C@H]1C=C(CO)[C@H](O)[C@H](O)[C@H]1O JARYYMUOCXVXNK-IMTORBKUSA-N 0.000 description 1
- DBXFMOWZRXXBRN-LWKPJOBUSA-N valifenalate Chemical compound CC(C)OC(=O)N[C@@H](C(C)C)C(=O)NC(CC(=O)OC)C1=CC=C(Cl)C=C1 DBXFMOWZRXXBRN-LWKPJOBUSA-N 0.000 description 1
- 235000015112 vegetable and seed oil Nutrition 0.000 description 1
- 239000008158 vegetable oil Substances 0.000 description 1
- 235000013311 vegetables Nutrition 0.000 description 1
- 239000010455 vermiculite Substances 0.000 description 1
- 229910052902 vermiculite Inorganic materials 0.000 description 1
- 235000019354 vermiculite Nutrition 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 229920002554 vinyl polymer Polymers 0.000 description 1
- 235000020234 walnut Nutrition 0.000 description 1
- 239000004562 water dispersible granule Substances 0.000 description 1
- 239000004549 water soluble granule Substances 0.000 description 1
- 239000004552 water soluble powder Substances 0.000 description 1
- 239000001993 wax Substances 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
- 229940100445 wheat starch Drugs 0.000 description 1
- 239000002023 wood Substances 0.000 description 1
- 239000000230 xanthan gum Substances 0.000 description 1
- 229920001285 xanthan gum Polymers 0.000 description 1
- 235000010493 xanthan gum Nutrition 0.000 description 1
- 229940082509 xanthan gum Drugs 0.000 description 1
- 239000005019 zein Substances 0.000 description 1
- 229940093612 zein Drugs 0.000 description 1
- 239000010457 zeolite Substances 0.000 description 1
- 239000005943 zeta-Cypermethrin Substances 0.000 description 1
- GTLDTDOJJJZVBW-UHFFFAOYSA-N zinc cyanide Chemical compound [Zn+2].N#[C-].N#[C-] GTLDTDOJJJZVBW-UHFFFAOYSA-N 0.000 description 1
- NWONKYPBYAMBJT-UHFFFAOYSA-L zinc sulfate Chemical compound [Zn+2].[O-]S([O-])(=O)=O NWONKYPBYAMBJT-UHFFFAOYSA-L 0.000 description 1
- 229910000368 zinc sulfate Inorganic materials 0.000 description 1
- 229960001763 zinc sulfate Drugs 0.000 description 1
- DUBNHZYBDBBJHD-UHFFFAOYSA-L ziram Chemical compound [Zn+2].CN(C)C([S-])=S.CN(C)C([S-])=S DUBNHZYBDBBJHD-UHFFFAOYSA-L 0.000 description 1
- GVJHHUAWPYXKBD-IEOSBIPESA-N α-tocopherol Chemical compound OC1=C(C)C(C)=C2O[C@@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-IEOSBIPESA-N 0.000 description 1
Landscapes
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
Abstract
Description
本発明は、新規なアミド化合物及びそれらの塩、並びに該化合物を有効成分として含有することを特徴とする有害生物防除剤に関するものである。 The present invention relates to a novel amide compound and a salt thereof, and a pest control agent comprising the compound as an active ingredient.
例えば、特許文献1乃至特許文献7には、アミド化合物が開示されているが、本発明に係るアミド化合物に関しては何ら開示されていない。さらに、その有害生物防除剤、特に、殺虫・殺ダニ剤、及び哺乳動物又は鳥類の内部若しくは外部寄生虫防除剤としての有用性は、全く知られていない。 For example, Patent Documents 1 to 7 disclose amide compounds, but do not disclose any amide compounds according to the present invention. Furthermore, its usefulness as a pest control agent, in particular, an insecticide / acaricide, and an internal or ectoparasite control agent for mammals or birds is not known at all.
農園芸病害虫、森林病害虫、或いは衛生病害虫等、各種病害虫の防除を目的とする有害生物防除剤の開発が進み、多種多様な薬剤が今日まで実用に供されてきた。 The development of pest control agents for the purpose of controlling various pests such as agricultural and horticultural pests, forest pests, and hygiene pests has progressed, and a wide variety of drugs have been put to practical use to date.
しかしながら、こうした薬剤の長年にわたる使用により、近年、病害虫が薬剤抵抗性を獲得し、従来用いられてきた既存の殺虫剤や殺菌剤による防除が困難となる場面が増えてきている。また、既存の有害生物防除剤の一部のものは毒性が高く、或いはあるものは環境中に長期間残留することにより、生態系を攪乱するという問題も顕在化しつつある。このような状況下、高度な有害生物防除活性を有するのみならず、低毒性且つ低残留性の新規な有害生物防除剤の開発が常に期待されている。 However, due to the long-term use of such drugs, in recent years, pests have acquired drug resistance, making it difficult to control with existing insecticides and fungicides that have been used. In addition, some existing pest control agents are highly toxic, or some of them remain in the environment for a long time, and the problem of disturbing the ecosystem is becoming apparent. Under such circumstances, development of a novel pest control agent having not only high pest control activity but also low toxicity and low persistence is always expected.
本発明の目的は、優れた有害生物防除活性を示し、ホ乳動物、魚類及び益虫等の非標的生物に対してほとんど悪影響が無いなどの低毒性で、且つ低残留性の新規な有害生物防除剤の提供することである。 The object of the present invention is to provide a novel pest control which exhibits excellent pest control activity, has low toxicity and has little adverse effect on non-target organisms such as mammals, fish and beneficial insects. It is to provide an agent.
本発明者らは、上記の課題解決を目標に鋭意研究を重ねた結果、本発明に係る下記式(1)で表される新規な縮合複素環化合物が優れた有害生物防除活性、特に殺虫・殺ダニ活性を示し、且つ、ホ乳動物、魚類及び益虫等の非標的生物に対しては、ほとんど悪影響の無い、極めて有用な化合物であることを見い出し、本発明を完成した。 As a result of intensive research aimed at solving the above-mentioned problems, the present inventors have found that the novel condensed heterocyclic compound represented by the following formula (1) according to the present invention has excellent pest control activity, particularly insecticide / The present invention was completed by discovering that it is an extremely useful compound that exhibits acaricidal activity and has almost no adverse effect on non-target organisms such as mammals, fish and beneficial insects.
すなわち、本発明は下記〔1〕〜〔14〕に関するものである。 That is, the present invention relates to the following [1] to [14].
〔1〕
式(1):
[1]
Formula (1):
[式中、R1は、水素原子、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルケニル、ハロ(C1〜C6)アルケニル、C1〜C6アルキニル、ハロ(C1〜C6)アルキニル、C3〜C6シクロアルキル、ハロ(C3〜C6)シクロアルキル、C3〜C6シクロアルケニル又はハロ(C3〜C6)シクロアルケニルを表し、
R2は、水素原子、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルケニル、ハロ(C1〜C6)アルケニル、C1〜C6アルキニル、ハロ(C1〜C6)アルキニル、C3〜C6シクロアルキル、ハロ(C3〜C6)シクロアルキル、C3〜C6シクロアルケニル又はハロ(C3〜C6)シクロアルケニルを表し、
Gは、G1〜G6で表される構造を表し、
[Wherein, R 1 is a hydrogen atom, C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkenyl, halo (C 1 -C 6 ) alkenyl, C 1 -C 6 Alkynyl, halo (C 1 -C 6 ) alkynyl, C 3 -C 6 cycloalkyl, halo (C 3 -C 6 ) cycloalkyl, C 3 -C 6 cycloalkenyl or halo (C 3 -C 6 ) cycloalkenyl Represent,
R 2 is a hydrogen atom, C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkenyl, halo (C 1 -C 6 ) alkenyl, C 1 -C 6 alkynyl, halo ( C 1 -C 6 ) alkynyl, C 3 -C 6 cycloalkyl, halo (C 3 -C 6 ) cycloalkyl, C 3 -C 6 cycloalkenyl or halo (C 3 -C 6 ) cycloalkenyl,
G represents a structure represented by G 1 to G 6 ;
Y2及びY4は、各々独立して水素原子、ハロゲン原子、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルケニル、ハロ(C1〜C6)アルケニル、C1〜C6アルキニル、ハロ(C1〜C6)アルキニル、C3〜C6シクロアルキル、ハロ(C3〜C6)シクロアルキル、C3〜C6シクロアルケニル、ハロ(C3〜C6)シクロアルケニル、−OR40、−N(R80)R81、−C(O)OR70、−S(O)mR50、シアノ又はニトロを表し、
Y3は、水素原子、ハロゲン原子、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルケニル、ハロ(C1〜C6)アルケニル、C1〜C6アルキニル、ハロ(C1〜C6)アルキニル、C3〜C6シクロアルキル、ハロ(C3〜C6)シクロアルキル、C3〜C6シクロアルケニル、ハロ(C3〜C6)シクロアルケニル、−OR40、−N(R60)R61、−C(O)OR70、−S(O)mR50、シアノ又はニトロを表し、
X1、X2、X3、X4及びX6は、各々独立して水素原子、ハロゲン原子、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルケニル、ハロ(C1〜C6)アルケニル、C1〜C6アルキニル、ハロ(C1〜C6)アルキニル、C3〜C6シクロアルキル、ハロ(C3〜C6)シクロアルキル、C3〜C6シクロアルケニル、ハロ(C3〜C6)シクロアルケニル、−OR90、−N(R10)R11、−C(O)OR70、−S(O)pR30、シアノ又はニトロを表し、
R10は、水素原子、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルケニル、ハロ(C1〜C6)アルケニル、C1〜C6アルキニル、ハロ(C1〜C6)アルキニル、C3〜C6シクロアルキル、ハロ(C3〜C6)シクロアルキル、C3〜C6シクロアルケニル、ハロ(C3〜C6)シクロアルケニル、C1〜C6アルキルカルボニル、ハロ(C1〜C6)アルキルカルボニル、C1〜C6アルコキシ、ハロ(C1〜C6)アルコキシ、C1〜C6アルコキシカルボニル、ハロ(C1〜C6)アルコキシカルボニル又はC1〜C6アルキルスルホニルを表し、
R11は、水素原子、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルキルカルボニル、ハロ(C1〜C6)アルキルカルボニル、C1〜C6アルコキシカルボニル、ハロ(C1〜C6)アルコキシカルボニル又はC1〜C6アルキルスルホニルを表し、
R30は、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルケニル、ハロ(C1〜C6)アルケニル、C1〜C6アルキニル、ハロ(C1〜C6)アルキニル
C3〜C6シクロアルキル、ハロ(C3〜C6)シクロアルキル、C3〜C6シクロアルケニル又はハロ(C3〜C6)シクロアルケニルを表し、
R40は、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルケニル、ハロ(C1〜C6)アルケニル、C1〜C6アルキニル、ハロ(C1〜C6)アルキニル、C3〜C6シクロアルキル、ハロ(C3〜C6)シクロアルキル、C3〜C6シクロアルケニル又はハロ(C3〜C6)シクロアルケニルを表し、
R50は、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルケニル、ハロ(C1〜C6)アルケニル、C1〜C6アルキニル、ハロ(C1〜C6)アルキニル、C3〜C6シクロアルキル、ハロ(C3〜C6)シクロアルキル、C3〜C6シクロアルケニル、ハロ(C3〜C6)シクロアルケニル又はC1〜C10アルコキシカルボニル(C1〜C6)アルキルを表し、
R60は、水素原子、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルケニル、ハロ(C1〜C6)アルケニル、C1〜C6アルキニル、ハロ(C1〜C6)アルキニル、C3〜C6シクロアルキル、ハロ(C3〜C6)シクロアルキル、C3〜C6シクロアルケニル、ハロ(C3〜C6)シクロアルケニル、C1〜C6アルキルカルボニル、ハロ(C1〜C6)アルキルカルボニル、C1〜C6アルコキシ、ハロ(C1〜C6)アルコキシ、C1〜C6アルコキシカルボニル、ハロ(C1〜C6)アルコキシカルボニル又はC1〜C6アルキルスルホニルを表し、
R61は、水素原子、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルキルカルボニル、ハロ(C1〜C6)アルキルカルボニル、C1〜C6アルコキシカルボニル、ハロ(C1〜C6)アルコキシカルボニル又はC1〜C6アルキルスルホニルを表し、
R70は、水素原子、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルケニル、ハロ(C1〜C6)アルケニル、C1〜C6アルキニル、ハロ(C1〜C6)アルキニル、C3〜C6シクロアルキル、ハロ(C3〜C6)シクロアルキル、C3〜C6シクロアルケニル又はハロ(C3〜C6)シクロアルケニルを表し、
R80は、水素原子、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルケニル、ハロ(C1〜C6)アルケニル、C1〜C6アルキニル、ハロ(C1〜C6)アルキニル、C3〜C6シクロアルキル、ハロ(C3〜C6)シクロアルキル、C3〜C6シクロアルケニル、ハロ(C3〜C6)シクロアルケニル、C1〜C6アルキルカルボニル、ハロ(C1〜C6)アルキルカルボニル、C1〜C6アルコキシ、ハロ(C1〜C6)アルコキシ、C1〜C6アルコキシカルボニル、ハロ(C1〜C6)アルコキシカルボニル又はC1〜C6アルキルスルホニルを表し、
R81は、水素原子、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルキルカルボニル、ハロ(C1〜C6)アルキルカルボニル、C1〜C6アルコキシカルボニル、ハロ(C1〜C6)アルコキシカルボニル又はC1〜C6アルキルスルホニルを表し、
R90は、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルケニル、ハロ(C1〜C6)アルケニル、C1〜C6アルキニル、ハロ(C1〜C6)アルキニル、C3〜C6シクロアルキル、ハロ(C3〜C6)シクロアルキル、C3〜C6シクロアルケニル又はハロ(C3〜C6)シクロアルケニルを表し、
nは、0、1又は2の整数を表し、
mは、0、1又は2の整数を表し、
pは、0、1又は2の整数を表す。]で表される縮合複素環化合物若しくはその塩又はそれらのN−オキシド。
Y2 and Y4 are each independently a hydrogen atom, a halogen atom, C 1 -C 6 alkyl, halo (C 1 ~C 6) alkyl, C 1 -C 6 alkenyl, halo (C 1 ~C 6) alkenyl, C 1 -C 6 alkynyl, halo (C 1 ~C 6) alkynyl, C 3 -C 6 cycloalkyl, halo (C 3 ~C 6) cycloalkyl, C 3 -C 6 cycloalkenyl, halo (C 3 -C 6 ) Represents cycloalkenyl, —OR 40 , —N (R 80 ) R 81 , —C (O) OR 70 , —S (O) m R 50 , cyano or nitro;
Y3 is hydrogen atom, halogen atom, C 1 -C 6 alkyl, halo (C 1 ~C 6) alkyl, C 1 -C 6 alkenyl, halo (C 1 ~C 6) alkenyl, C 1 -C 6 alkynyl, Halo (C 1 -C 6 ) alkynyl, C 3 -C 6 cycloalkyl, halo (C 3 -C 6 ) cycloalkyl, C 3 -C 6 cycloalkenyl, halo (C 3 -C 6 ) cycloalkenyl, -OR 40, -N (R 60) R 61, -C (O) oR 70, -S (O) m R 50, cyano or nitro,
X 1 , X 2 , X 3 , X 4 and X 6 are each independently a hydrogen atom, a halogen atom, C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkenyl, Halo (C 1 -C 6 ) alkenyl, C 1 -C 6 alkynyl, halo (C 1 -C 6 ) alkynyl, C 3 -C 6 cycloalkyl, halo (C 3 -C 6 ) cycloalkyl, C 3 -C Represents 6 cycloalkenyl, halo (C 3 -C 6 ) cycloalkenyl, —OR 90 , —N (R 10 ) R 11 , —C (O) OR 70 , —S (O) p R 30 , cyano or nitro. ,
R 10 is a hydrogen atom, C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkenyl, halo (C 1 -C 6 ) alkenyl, C 1 -C 6 alkynyl, halo ( C 1 -C 6) alkynyl, C 3 -C 6 cycloalkyl, halo (C 3 ~C 6) cycloalkyl, C 3 -C 6 cycloalkenyl, halo (C 3 ~C 6) cycloalkenyl, C 1 -C 6 alkylcarbonyl, halo (C 1 -C 6 ) alkylcarbonyl, C 1 -C 6 alkoxy, halo (C 1 -C 6 ) alkoxy, C 1 -C 6 alkoxycarbonyl, halo (C 1 -C 6 ) alkoxycarbonyl or represents C 1 -C 6 alkylsulfonyl,
R 11 is a hydrogen atom, C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkylcarbonyl, halo (C 1 -C 6 ) alkylcarbonyl, C 1 -C 6 alkoxycarbonyl , Halo (C 1 -C 6 ) alkoxycarbonyl or C 1 -C 6 alkylsulfonyl,
R 30 is C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkenyl, halo (C 1 -C 6 ) alkenyl, C 1 -C 6 alkynyl, halo (C 1- C 6 ) represents alkynyl C 3 -C 6 cycloalkyl, halo (C 3 -C 6 ) cycloalkyl, C 3 -C 6 cycloalkenyl or halo (C 3 -C 6 ) cycloalkenyl,
R 40 is C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkenyl, halo (C 1 -C 6 ) alkenyl, C 1 -C 6 alkynyl, halo (C 1- C 6 ) represents alkynyl, C 3 -C 6 cycloalkyl, halo (C 3 -C 6 ) cycloalkyl, C 3 -C 6 cycloalkenyl or halo (C 3 -C 6 ) cycloalkenyl,
R 50 is C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkenyl, halo (C 1 -C 6 ) alkenyl, C 1 -C 6 alkynyl, halo (C 1- C 6) alkynyl, C 3 -C 6 cycloalkyl, halo (C 3 ~C 6) cycloalkyl, C 3 -C 6 cycloalkenyl, halo (C 3 ~C 6) cycloalkenyl or C 1 -C 10 alkoxycarbonyl (C 1 ~C 6) alkyl,
R 60 is a hydrogen atom, C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkenyl, halo (C 1 -C 6 ) alkenyl, C 1 -C 6 alkynyl, halo ( C 1 -C 6) alkynyl, C 3 -C 6 cycloalkyl, halo (C 3 ~C 6) cycloalkyl, C 3 -C 6 cycloalkenyl, halo (C 3 ~C 6) cycloalkenyl, C 1 -C 6 alkylcarbonyl, halo (C 1 -C 6 ) alkylcarbonyl, C 1 -C 6 alkoxy, halo (C 1 -C 6 ) alkoxy, C 1 -C 6 alkoxycarbonyl, halo (C 1 -C 6 ) alkoxycarbonyl or represents C 1 -C 6 alkylsulfonyl,
R 61 is a hydrogen atom, C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkylcarbonyl, halo (C 1 -C 6 ) alkylcarbonyl, C 1 -C 6 alkoxycarbonyl , Halo (C 1 -C 6 ) alkoxycarbonyl or C 1 -C 6 alkylsulfonyl,
R 70 represents a hydrogen atom, C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkenyl, halo (C 1 -C 6 ) alkenyl, C 1 -C 6 alkynyl, halo ( C 1 -C 6 ) alkynyl, C 3 -C 6 cycloalkyl, halo (C 3 -C 6 ) cycloalkyl, C 3 -C 6 cycloalkenyl or halo (C 3 -C 6 ) cycloalkenyl,
R 80 is a hydrogen atom, C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkenyl, halo (C 1 -C 6 ) alkenyl, C 1 -C 6 alkynyl, halo ( C 1 -C 6) alkynyl, C 3 -C 6 cycloalkyl, halo (C 3 ~C 6) cycloalkyl, C 3 -C 6 cycloalkenyl, halo (C 3 ~C 6) cycloalkenyl, C 1 -C 6 alkylcarbonyl, halo (C 1 -C 6 ) alkylcarbonyl, C 1 -C 6 alkoxy, halo (C 1 -C 6 ) alkoxy, C 1 -C 6 alkoxycarbonyl, halo (C 1 -C 6 ) alkoxycarbonyl or represents C 1 -C 6 alkylsulfonyl,
R 81 is a hydrogen atom, C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkylcarbonyl, halo (C 1 -C 6 ) alkylcarbonyl, C 1 -C 6 alkoxycarbonyl , Halo (C 1 -C 6 ) alkoxycarbonyl or C 1 -C 6 alkylsulfonyl,
R 90 is C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkenyl, halo (C 1 -C 6 ) alkenyl, C 1 -C 6 alkynyl, halo (C 1- C 6 ) represents alkynyl, C 3 -C 6 cycloalkyl, halo (C 3 -C 6 ) cycloalkyl, C 3 -C 6 cycloalkenyl or halo (C 3 -C 6 ) cycloalkenyl,
n represents an integer of 0, 1 or 2;
m represents an integer of 0, 1 or 2;
p represents an integer of 0, 1 or 2. ] The condensed heterocyclic compound or its salt represented by these, or those N-oxides.
〔2〕
R1は、C1〜C6アルキルを表し、
R2は、水素原子又はC1〜C6アルキルを表し、
Y2及びY4は、各々独立して水素原子、ハロゲン原子、ハロ(C1〜C6)アルキル、C3〜C6シクロアルキル、−N(R80)R81、−C(O)OR70、−S(O)mR50、シアノ又はニトロを表し、
Y3は、水素原子、ハロゲン原子、ハロ(C1〜C6)アルキル、C3〜C6シクロアルキル、−N(R60)R61、−C(O)OR70、−S(O)mR50、シアノ又はニトロを表し、
X1、X2、X3、X4及びX6は、各々独立して水素原子、ハロゲン原子、ハロ(C1〜C6)アルキル、−N(R10)R11又はニトロを表し、
R10は、水素原子又はC1〜C6アルキルを表し、
R11は、水素原子を表し、
R50は、C1〜C6アルキル又はC1〜C10アルコキシカルボニル(C1〜C6)アルキルを表し、
R60は、水素原子、C1〜C6アルキルカルボニル、C1〜C6アルコキシカルボニル又はC1〜C6アルキルスルホニルを表し、
R61は、水素原子、C1〜C6アルキルカルボニル、C1〜C6アルコキシカルボニル又はC1〜C6アルキルスルホニルを表し、
R70は、水素原子又はC1〜C6アルキルを表し、
R80は、水素原子、C1〜C6アルキルカルボニル、C1〜C6アルコキシカルボニル又はC1〜C6アルキルスルホニルを表し、
R81は、水素原子、C1〜C6アルキルカルボニル、C1〜C6アルコキシカルボニル又はC1〜C6アルキルスルホニルを表す上記〔1〕に記載の縮合複素環化合物若しくはその塩又はそれらのN−オキシド。
[2]
R 1 represents C 1 -C 6 alkyl;
R 2 represents a hydrogen atom or C 1 -C 6 alkyl,
Y2 and Y4 are each independently a hydrogen atom, a halogen atom, halo (C 1 ~C 6) alkyl, C 3 -C 6 cycloalkyl, -N (R 80) R 81 , -C (O) OR 70, Represents —S (O) m R 50 , cyano or nitro,
Y3 represents a hydrogen atom, a halogen atom, halo (C 1 ~C 6) alkyl, C 3 -C 6 cycloalkyl, -N (R 60) R 61 , -C (O) OR 70, -S (O) m R 50 represents cyano or nitro,
X 1 , X 2 , X 3 , X 4 and X 6 each independently represent a hydrogen atom, a halogen atom, halo (C 1 -C 6 ) alkyl, —N (R 10 ) R 11 or nitro,
R 10 represents a hydrogen atom or C 1 -C 6 alkyl,
R 11 represents a hydrogen atom,
R 50 represents C 1 -C 6 alkyl or C 1 -C 10 alkoxycarbonyl (C 1 -C 6 ) alkyl;
R 60 represents a hydrogen atom, C 1 -C 6 alkylcarbonyl, C 1 -C 6 alkoxycarbonyl or C 1 -C 6 alkylsulfonyl,
R 61 represents a hydrogen atom, C 1 -C 6 alkylcarbonyl, C 1 -C 6 alkoxycarbonyl or C 1 -C 6 alkylsulfonyl,
R 70 represents a hydrogen atom or C 1 -C 6 alkyl,
R 80 represents a hydrogen atom, C 1 -C 6 alkylcarbonyl, C 1 -C 6 alkoxycarbonyl or C 1 -C 6 alkylsulfonyl,
R 81 represents a hydrogen atom, a C 1 -C 6 alkylcarbonyl, a C 1 -C 6 alkoxycarbonyl, or a C 1 -C 6 alkylsulfonyl, the condensed heterocyclic compound or a salt thereof according to the above [1], or their N -Oxides.
〔3〕
Y2は、水素原子、ハロゲン原子、ハロ(C1〜C6)アルキル、C3〜C6シクロアルキル又は−N(R80)R81を表し、
Y3は、水素原子、ハロゲン原子、ハロ(C1〜C6)アルキル、C3〜C6シクロアルキル、−N(R60)R61、−C(O)OR70、−S(O)mR50、シアノ又はニトロを表し、
Y4は、水素原子又は−N(R80)R81を表し、
X1は、水素原子、ハロゲン原子、−N(R10)R11又はニトロを表し、
X2は、水素原子又はハロ(C1〜C6)アルキルを表し、
X3は、水素原子又はハロ(C1〜C6)アルキルを表し、
X4は、水素原子又はハロ(C1〜C6)アルキルを表し、
X6は、ハロゲン原子を表し、
R60は、水素原子又はC1〜C6アルキルカルボニルを表し、
R61は、水素原子を表し、
R80は、水素原子を表し、
R81は、水素原子を表し、
mは、0又は1の整数を表す上記〔2〕に記載の縮合複素環化合物若しくはその塩又はそれらのN−オキシド。
[3]
Y2 represents a hydrogen atom, a halogen atom, halo (C 1 ~C 6) alkyl, a C 3 -C 6 cycloalkyl or -N (R 80) R 81,
Y3 represents a hydrogen atom, a halogen atom, halo (C 1 ~C 6) alkyl, C 3 -C 6 cycloalkyl, -N (R 60) R 61 , -C (O) OR 70, -S (O) m R 50 represents cyano or nitro,
Y4 represents a hydrogen atom or -N (R 80) R 81,
X 1 represents a hydrogen atom, a halogen atom, —N (R 10 ) R 11 or nitro,
X 2 represents a hydrogen atom or halo (C 1 -C 6 ) alkyl,
X 3 represents a hydrogen atom or halo (C 1 -C 6 ) alkyl,
X 4 represents a hydrogen atom or halo (C 1 -C 6 ) alkyl,
X 6 represents a halogen atom,
R 60 represents a hydrogen atom or a C 1 -C 6 alkylcarbonyl,
R 61 represents a hydrogen atom,
R 80 represents a hydrogen atom,
R 81 represents a hydrogen atom,
m represents the integer of 0 or 1, the fused heterocyclic compound according to the above [2], a salt thereof, or an N-oxide thereof.
〔4〕
Y2は、水素原子、ハロゲン原子又はハロ(C1〜C6)アルキルを表し、
Y3は、水素原子、ハロゲン原子、ハロ(C1〜C6)アルキル、−S(O)mR50又はシアノを表し、
Y4は、水素原子を表し、
X1は、水素原子又は−N(R10)R11を表し、
R10は、C1〜C6アルキルを表し、
R50は、C1〜C6アルキルを表す上記〔3〕に記載の縮合複素環化合物若しくはその塩又はそれらのN−オキシド。
[4]
Y2 represents a hydrogen atom, a halogen atom or a halo (C 1 ~C 6) alkyl,
Y3 represents a hydrogen atom, a halogen atom, halo (C 1 ~C 6) alkyl, -S and (O) m R 50 or cyano,
Y4 represents a hydrogen atom,
X 1 represents a hydrogen atom or —N (R 10 ) R 11 ,
R 10 represents C 1 -C 6 alkyl,
R 50 represents the condensed heterocyclic compound or a salt thereof or the N-oxide thereof according to the above [3], wherein C 50 represents C 1 -C 6 alkyl.
〔5〕
Y3は、水素原子、ハロゲン原子又はハロ(C1〜C6)アルキルを表し、
X4は、ハロ(C1〜C6)アルキルを表し、
nは、0又は2の整数を表す上記〔4〕に記載の縮合複素環化合物若しくはその塩又はそれらのN−オキシド。
[5]
Y3 represents a hydrogen atom, a halogen atom or a halo (C 1 ~C 6) alkyl,
X 4 represents halo (C 1 -C 6 ) alkyl;
n is the condensed heterocyclic compound or a salt thereof or the N-oxide thereof according to the above [4], wherein n represents an integer of 0 or 2.
〔6〕
上記〔1〕〜〔5〕のいずれか1項に記載の縮合複素環化合物及びその塩から選ばれる1種又は2種以上を有効成分として含有する有害生物防除剤。
[6]
The pest control agent which contains 1 type, or 2 or more types chosen from the condensed heterocyclic compound and its salt of any one of said [1]-[5] as an active ingredient.
〔7〕
上記〔1〕〜〔5〕のいずれか1項に記載の縮合複素環化合物及びその塩から選ばれる1種又は2種以上を有効成分として含有する農薬。
[7]
An agrochemical containing one or more selected from the condensed heterocyclic compound according to any one of [1] to [5] above and a salt thereof as an active ingredient.
〔8〕
上記〔1〕〜〔5〕のいずれか1項に記載の縮合複素環化合物及びその塩から選ばれる1種又は2種以上を有効成分として含有する哺乳動物又は鳥類の内部若しくは外部寄生虫の防除剤。
[8]
Control of internal or ectoparasites of mammals or birds containing, as an active ingredient, one or more selected from the fused heterocyclic compounds according to any one of [1] to [5] above and salts thereof Agent.
〔9〕
外部寄生虫がノミ目類又はマダニ類である上記〔8〕に記載の防除剤。
[9]
The control agent according to [8] above, wherein the ectoparasite is a flea or ticks.
〔10〕
上記〔1〕〜〔5〕のいずれか1項に記載の縮合複素環化合物及びその塩から選ばれる1種又は2種以上を有効成分として含有する殺虫剤又は殺ダニ剤。
[10]
An insecticide or acaricide containing, as an active ingredient, one or more selected from the condensed heterocyclic compound according to any one of [1] to [5] above and a salt thereof.
〔11〕
上記〔1〕〜〔5〕のいずれか1項に記載の縮合複素環化合物及びその塩から選ばれる1種又は2種以上を有効成分として含有する種子処理剤。
[11]
A seed treatment agent containing one or more selected from the condensed heterocyclic compound according to any one of [1] to [5] above and a salt thereof as an active ingredient.
〔12〕
種子処理が浸漬処理によって行われる上記〔11〕に記載の種子処理剤。
[12]
The seed treatment agent according to [11] above, wherein the seed treatment is performed by dipping treatment.
〔13〕
上記〔1〕〜〔5〕のいずれか1項に記載の縮合複素環化合物及びその塩から選ばれる1種又は2種以上を有効成分として含有する土壌処理剤。
[13]
The soil treatment agent which contains 1 type, or 2 or more types chosen from the condensed heterocyclic compound and its salt of any one of said [1]-[5] as an active ingredient.
〔14〕
土壌処理が土壌潅注処理によって行われる上記〔13〕に記載の土壌処理剤。
[14]
The soil treatment agent according to [13], wherein the soil treatment is performed by soil irrigation treatment.
本発明化合物は多くの農業害虫、ハダニ類、哺乳動物又は鳥類の内部もしくは外部寄生虫に対して優れた殺虫・殺ダニ活性を有し、既存の殺虫剤に対して抵抗性を獲得した害虫に対しても十分な防除効果を発揮する。さらに、ホ乳類、魚類及び益虫に対してほとんど悪影響を及ぼさず、低残留性で環境に対する負荷も軽い。従って、本発明は有用な新規有害生物防除剤を提供することができる。 The compound of the present invention has excellent insecticidal / miticidal activity against many agricultural pests, spider mites, mammals or birds, or against insects that have acquired resistance to existing insecticides. Even against this, it exhibits a sufficient control effect. Furthermore, it has little adverse effect on mammals, fish and beneficial insects, has low persistence, and has a light environmental impact. Therefore, the present invention can provide a useful novel pest control agent.
本発明に包含される化合物には、置換基の種類によってはE−体及びZ−体の幾何異性体が存在する場合があるが、本発明はこれらE−体、Z−体又はE−体及びZ−体を任意の割合で含む混合物を包含するものである。 The compounds included in the present invention may have geometrical isomers of E-form and Z-form depending on the type of substituent, and the present invention is not limited to these E-form, Z-form or E-form. And a mixture containing the Z-form in an arbitrary ratio.
また、本発明に包含される化合物は、1個又は2個以上の不斉炭素原子又は不斉硫黄原子の存在に起因する光学活性体が存在するが、本発明は全ての光学活性体又はラセミ体を包含する。 Further, the compounds included in the present invention include optically active substances resulting from the presence of one or more asymmetric carbon atoms or asymmetric sulfur atoms, but the present invention includes all optically active substances or racemic compounds. Includes the body.
また、本発明に包含される化合物には、置換基の種類によって互変異性体が存在する場合があるが、本発明は全ての互変異性体又は任意の割合で含む互変異性体の混合物を包含するものである。 In addition, the compounds included in the present invention may have tautomers depending on the type of substituent, but the present invention includes all tautomers or a mixture of tautomers included in an arbitrary ratio. Is included.
本発明に包含される化合物のうちで、常法に従って塩にすることができるものは、例えば、フッ化水素酸、塩酸、臭化水素酸、ヨウ化水素酸等のハロゲン化水素酸の塩、硝酸、硫酸、燐酸、塩素酸、過塩素酸等の無機酸の塩、メタンスルホン酸、エタンスルホン酸、トリフルオロメタンスルホン酸、ベンゼンスルホン酸、p−トルエンスルホン酸等のスルホン酸の塩、ギ酸、酢酸、プロピオン酸、トリフルオロ酢酸、フマール酸、酒石酸、蓚酸、マレイン酸、リンゴ酸、コハク酸、安息香酸、マンデル酸、アスコルビン酸、乳酸、グルコン酸、クエン酸等のカルボン酸の塩、グルタミン酸、アスパラギン酸等のアミノ酸の塩、リチウム、ナトリウム、カリウムといったアルカリ金属の塩、カルシウム、バリウム、マグネシウムといったアルカリ土類金属の塩、アルミニウムの塩、テトラメチルアンモニウム塩、テトラブチルアンモニウム塩、ベンジルトリメチルアンモニウム塩等の四級アンモニウム塩とすることができる。 Among the compounds included in the present invention, those that can be converted into a salt according to a conventional method are, for example, hydrohalic acid salts such as hydrofluoric acid, hydrochloric acid, hydrobromic acid, hydroiodic acid, Salts of inorganic acids such as nitric acid, sulfuric acid, phosphoric acid, chloric acid, perchloric acid, salts of sulfonic acids such as methanesulfonic acid, ethanesulfonic acid, trifluoromethanesulfonic acid, benzenesulfonic acid, p-toluenesulfonic acid, formic acid, Acetic acid, propionic acid, trifluoroacetic acid, fumaric acid, tartaric acid, succinic acid, maleic acid, malic acid, succinic acid, benzoic acid, mandelic acid, ascorbic acid, lactic acid, gluconic acid, citric acid and other carboxylic acid salts, glutamic acid, Salts of amino acids such as aspartic acid, alkali metal salts such as lithium, sodium and potassium, alkalis such as calcium, barium and magnesium Earth metal salts, aluminum salts, tetramethylammonium salts, tetrabutylammonium salts, may be quaternary ammonium salts such as benzyltrimethylammonium salts.
本発明化合物において、N−オキシドとは、複素環上の環を構成する窒素原子が酸化された化合物である。N−オキシドを形成しうる複素環としは、例えばピリジン環を含む縮合環が挙げられる。 In the compound of the present invention, N-oxide is a compound in which a nitrogen atom constituting a ring on a heterocyclic ring is oxidized. Examples of the heterocyclic ring that can form an N-oxide include condensed rings including a pyridine ring.
「式(1)で表される本発明化合物」は、「本発明化合物(1)」とも記載され、また、「式(1−A)で表される化合物」は、「化合物(1−A)」とも記載される。他の化合物についてもこれに準じて同様に記載される。 “The compound of the present invention represented by formula (1)” is also described as “the compound of the present invention (1)”, and “the compound represented by formula (1-A)” is “compound (1-A ) ". The other compounds are similarly described.
次に、本明細書において示した各置換基の具体例を以下に示す。ここで、n−はノルマル、i−はイソ、s−はセカンダリー及びtert−はターシャリーを各々意味する。 Next, specific examples of each substituent shown in the present specification are shown below. Here, n- means normal, i- means iso, s- means secondary, and tert- means tertiary.
本明細書における「ハロゲン原子」としては、フッ素原子、塩素原子、臭素原子及びヨウ素原子が挙げられる。尚、本明細書中「ハロ」の表記もこれらのハロゲン原子を表す。 Examples of the “halogen atom” in the present specification include a fluorine atom, a chlorine atom, a bromine atom and an iodine atom. In the present specification, the notation “halo” also represents these halogen atoms.
本明細書における「Ca〜Cbアルキル」の表記は、炭素原子数がa〜b個よりなる直鎖状又は分岐鎖状の炭化水素基を表し、例えばメチル基、エチル基、n−プロピル基、i−プロピル基、n−ブチル基、i−ブチル基、s−ブチル基、tert−ブチル基、n−ペンチル基、1,1−ジメチルプロピル基、n−ヘキシル基等が具体例として挙げられ、各々の指定の炭素原子数の範囲で選択される。 In the present specification, the expression “C a -C b alkyl” represents a linear or branched hydrocarbon group having a carbon number of a to b , such as a methyl group, an ethyl group, or n-propyl. Specific examples include a group, i-propyl group, n-butyl group, i-butyl group, s-butyl group, tert-butyl group, n-pentyl group, 1,1-dimethylpropyl group, n-hexyl group and the like. Selected within a range of each specified number of carbon atoms.
本明細書における「ハロ(Ca〜Cb)アルキル」の表記は、炭素原子に結合した水素原子がハロゲン原子によって任意に置換された、炭素原子数がa〜b個よりなる直鎖状又は分岐鎖状の炭化水素基を表し、このとき、2個以上のハロゲン原子によって置換されている場合、それらのハロゲン原子は互いに同一でも、または互いに相異なっていてもよい。例えばフルオロメチル基、クロロメチル基、ブロモメチル基、ヨードメチル基、ジフルオロメチル基、ジクロロメチル基、トリフルオロメチル基、クロロジフルオロメチル基、トリクロロメチル基、ブロモジフルオロメチル基、1−フルオロエチル基、2−フルオロエチル基、2−クロロエチル基、2−ブロモエチル基、2,2−ジフルオロエチル基、2,2,2−トリフルオロエチル基、2−クロロ−2,2−ジフルオロエチル基、2,2,2−トリクロロエチル基、2−ブロモ−2,2−ジフルオロエチル基、1,1,2,2−テトラフルオロエチル基、2−クロロ−1,1,2−トリフルオロエチル基、2−クロロ−1,1,2,2−テトラフルオロエチル基、ペンタフルオロエチル基、2,2−ジフルオロプロピル基、3,3,3−トリフルオロプロピル基、3−ブロモ−3,3−ジフルオロプロピル基、2,2,3,3−テトラフルオロプロピル基、2,2,3,3,3−ペンタフルオロプロピル基、1,1,2,3,3,3−ヘキサフルオロプロピル基、ヘプタフルオロプロピル基、2,2,2−トリフルオロ−1−(メチル)エチル基、2,2,2−トリフルオロ−1−(トリフルオロメチル)エチル基、1,2,2,2−テトラフルオロ−1−(トリフルオロメチル)エチル基、2,2,3,4,4,4−ヘキサフルオロブチル基、2,2,3,3,4,4,4−ヘプタフルオロブチル基、ノナフルオロブチル基等が具体例として挙げられ、各々の指定の炭素原子数の範囲で選択される。 In the present specification, the expression “halo (C a -C b ) alkyl” refers to a straight chain composed of a to b carbon atoms in which a hydrogen atom bonded to a carbon atom is optionally substituted with a halogen atom, or This represents a branched hydrocarbon group, and when substituted with two or more halogen atoms, these halogen atoms may be the same as or different from each other. For example, fluoromethyl group, chloromethyl group, bromomethyl group, iodomethyl group, difluoromethyl group, dichloromethyl group, trifluoromethyl group, chlorodifluoromethyl group, trichloromethyl group, bromodifluoromethyl group, 1-fluoroethyl group, 2- Fluoroethyl group, 2-chloroethyl group, 2-bromoethyl group, 2,2-difluoroethyl group, 2,2,2-trifluoroethyl group, 2-chloro-2,2-difluoroethyl group, 2,2,2 -Trichloroethyl group, 2-bromo-2,2-difluoroethyl group, 1,1,2,2-tetrafluoroethyl group, 2-chloro-1,1,2-trifluoroethyl group, 2-chloro-1 , 1,2,2-tetrafluoroethyl group, pentafluoroethyl group, 2,2-difluoropropyl group, 3,3,3- Lifluoropropyl group, 3-bromo-3,3-difluoropropyl group, 2,2,3,3-tetrafluoropropyl group, 2,2,3,3,3-pentafluoropropyl group, 1,1,2 , 3,3,3-hexafluoropropyl group, heptafluoropropyl group, 2,2,2-trifluoro-1- (methyl) ethyl group, 2,2,2-trifluoro-1- (trifluoromethyl) Ethyl group, 1,2,2,2-tetrafluoro-1- (trifluoromethyl) ethyl group, 2,2,3,4,4,4-hexafluorobutyl group, 2,2,3,3,4 Specific examples include, 4,4-heptafluorobutyl group, nonafluorobutyl group and the like, and each is selected within the range of the designated number of carbon atoms.
「Ca〜Cbアルケニル」の表記は、炭素原子数がa〜b個よりなる直鎖状又は分岐鎖状で、且つ分子内に1個又は2個以上の二重結合を有する不飽和炭化水素を表し、例えばビニル基、1−プロペニル基、2−プロペニル基、1−メチルエテニル基、2−ブテニル基、2−メチル−2−プロペニル基、3−メチル−2−ブテニル基、1,1−ジメチル−2−プロペニル基等が具体例として挙げられ、各々の指定の炭素原子数の範囲で選択される。 The expression “C a -C b alkenyl” is a linear or branched chain having a carbon number of a to b and an unsaturated carbonization having one or more double bonds in the molecule. Represents hydrogen, for example, vinyl group, 1-propenyl group, 2-propenyl group, 1-methylethenyl group, 2-butenyl group, 2-methyl-2-propenyl group, 3-methyl-2-butenyl group, 1,1- A dimethyl-2-propenyl group etc. are mentioned as a specific example, and it selects in the range of each designated number of carbon atoms.
「ハロ(Ca〜Cb)アルケニル」の表記は、炭素原子に結合した水素原子が、ハロゲン原子によって任意に置換された、炭素原子数がa〜b個よりなる直鎖状又は分岐鎖状で、且つ分子内に1個又は2個以上の二重結合を有する不飽和炭化水素を表す。このとき、2個以上のハロゲン原子によって置換されている場合、それらのハロゲン原子は互いに同一でも、又は互いに相異なっていてもよい。例えば2,2−ジクロロビニル基、2−フルオロ−2−プロペニル基、2−クロロ−2−プロペニル基、3−クロロ−2−プロペニル基、2−ブロモ−2−プロペニル基、3,3−ジフルオロ−2−プロペニル基、2,3−ジクロロ−2−プロペニル基、3,3−ジクロロ−2−プロペニル基、2,3,3−トリフルオロ−2−プロペニル基、2,3,3−トリクロロ−2−プロペニル基、1−(トリフルオロメチル)エテニル基、4,4−ジフルオロ−3−ブテニル基、3,4,4−トリフルオロ−3−ブテニル基、3−クロロ−4,4,4−トリフルオロ−2−ブテニル基等が具体例として挙げられ、各々の指定の炭素原子数の範囲で選択される。 The expression “halo (C a -C b ) alkenyl” is a linear or branched chain having a carbon number of a to b in which a hydrogen atom bonded to a carbon atom is optionally substituted with a halogen atom And an unsaturated hydrocarbon having one or more double bonds in the molecule. At this time, when substituted by two or more halogen atoms, these halogen atoms may be the same as or different from each other. For example, 2,2-dichlorovinyl group, 2-fluoro-2-propenyl group, 2-chloro-2-propenyl group, 3-chloro-2-propenyl group, 2-bromo-2-propenyl group, 3,3-difluoro -2-propenyl group, 2,3-dichloro-2-propenyl group, 3,3-dichloro-2-propenyl group, 2,3,3-trifluoro-2-propenyl group, 2,3,3-trichloro- 2-propenyl group, 1- (trifluoromethyl) ethenyl group, 4,4-difluoro-3-butenyl group, 3,4,4-trifluoro-3-butenyl group, 3-chloro-4,4,4- Specific examples include a trifluoro-2-butenyl group and the like, and each is selected within the range of the designated number of carbon atoms.
「Ca〜Cbアルキニル」の表記は、炭素原子数がa〜b個よりなる直鎖状又は分岐鎖状で、且つ分子内に1個又は2個以上の三重結合を有する不飽和炭化水素を表し、例えばエチニル基、プロパルギル基、2−ブチニル基、3−ブチニル基、1−ペンチニル基、1−ヘキシニル基、4,4,4−トリフルオロ−2−ブチニル基等が具体例として挙げられ、各々の指定の炭素原子数の範囲で選択される。 The expression “C a -C b alkynyl” refers to an unsaturated hydrocarbon having a linear or branched chain of a to b carbon atoms and having one or more triple bonds in the molecule. Specific examples include ethynyl group, propargyl group, 2-butynyl group, 3-butynyl group, 1-pentynyl group, 1-hexynyl group, 4,4,4-trifluoro-2-butynyl group and the like. , Each selected range of carbon atoms.
「ハロ(Ca〜Cb)アルキニル」の表記は、炭素原子に結合した水素原子が、ハロゲン原子によって任意に置換された、炭素原子数がa〜b個よりなる直鎖状又は分岐鎖状で、且つ分子内に1個又は2個以上の三重結合を有する不飽和炭化水素を表す。このとき、2個以上のハロゲン原子によって置換されている場合、それらのハロゲン原子は互いに同一でも、又は互いに相異なっていてもよい。例えば2−クロロエチニル基、2−ブロモエチニル基、2−ヨードエチニル基、3−クロロ−2−プロピニル基、3−ブロモ−2−プロピニル基、3−ヨード−2−プロピニル基等が具体例として挙げられ、各々の指定の炭素原子数の範囲で選択される。 The expression “halo (C a -C b ) alkynyl” is a linear or branched chain comprising a to b carbon atoms, in which a hydrogen atom bonded to a carbon atom is optionally substituted with a halogen atom And an unsaturated hydrocarbon having one or more triple bonds in the molecule. At this time, when substituted by two or more halogen atoms, these halogen atoms may be the same as or different from each other. Specific examples include 2-chloroethynyl group, 2-bromoethynyl group, 2-iodoethynyl group, 3-chloro-2-propynyl group, 3-bromo-2-propynyl group, 3-iodo-2-propynyl group and the like. Each of which is selected for each specified number of carbon atoms.
「Ca〜Cbアルコキシ」の表記は、炭素原子数がa〜b個よりなる前記の意味であるアルキル−O−基を表し、例えばメトキシ基、エトキシ基、n−プロピルオキシ基、i−プロピルオキシ基、n−ブチルオキシ基、i−ブチルオキシ基、s−ブチルオキシ基、tert−ブチルオキシ基、2−エチルへキシルオキシ基等が具体例として挙げられ、各々の指定の炭素原子数の範囲で選択される。 The expression “C a -C b alkoxy” represents an alkyl-O— group having the above-mentioned meaning consisting of a to b carbon atoms, for example, methoxy group, ethoxy group, n-propyloxy group, i- Specific examples include propyloxy group, n-butyloxy group, i-butyloxy group, s-butyloxy group, tert-butyloxy group, 2-ethylhexyloxy group, etc., and each is selected within the range of the designated number of carbon atoms. The
「ハロ(Ca〜Cb)アルコキシ」の表記は、炭素原子数がa〜b個よりなる前記の意味であるハロアルキル−O−基を表し、例えばジフルオロメトキシ基、トリフルオロメトキシ基、クロロジフルオロメトキシ基、ブロモジフルオロメトキシ基、2−フルオロエトキシ基、2−クロロエトキシ基、2,2,2−トリフルオロエトキシ基、1,1,2,2,−テトラフルオロエトキシ基、2−クロロ−1,1,2−トリフルオロエトキシ基、1,1,2,3,3,3−ヘキサフルオロプロピルオキシ基等が具体例として挙げられ、各々の指定の炭素原子数の範囲で選択される。 The expression “halo (C a -C b ) alkoxy” represents a haloalkyl-O— group having the above-mentioned meaning consisting of a to b carbon atoms, for example, difluoromethoxy group, trifluoromethoxy group, chlorodifluoro Methoxy group, bromodifluoromethoxy group, 2-fluoroethoxy group, 2-chloroethoxy group, 2,2,2-trifluoroethoxy group, 1,1,2,2, -tetrafluoroethoxy group, 2-chloro-1 Specific examples include 1,2,2-trifluoroethoxy group, 1,1,2,3,3,3-hexafluoropropyloxy group and the like, and each is selected within the range of the designated number of carbon atoms.
「Ca〜Cbアルキルスルホニル」の表記は、炭素原子数がa〜b個よりなる前記の意味であるアルキル−SO2−基を表し、例えばメチルスルホニル基、エチルスルホニル基、n−プロピルスルホニル基、i−プロピルスルホニル基、n−ブチルスルホニル基、i−ブチルスルホニル基、s−ブチルスルホニル基、tert−ブチルスルホニル基等が具体例として挙げられ、各々の指定の炭素原子数の範囲で選択される。 The notation “C a -C b alkylsulfonyl” represents an alkyl-SO 2 -group having the above-mentioned meaning consisting of a to b carbon atoms, for example, methylsulfonyl group, ethylsulfonyl group, n-propylsulfonyl. Group, i-propylsulfonyl group, n-butylsulfonyl group, i-butylsulfonyl group, s-butylsulfonyl group, tert-butylsulfonyl group and the like are mentioned as specific examples, and each selected within the range of the designated number of carbon atoms. Is done.
「Ca〜Cbアルキルカルボニル」の表記は、炭素原子数がa〜b個よりなる前記の意味であるアルキル−C(O)−基を表し、例えば、アセチル基、プロピオニル基、ブチリル基、イソブチリル基、バレリル基、イソバレリル基、2−メチルブタノイル基、ピバロイル基、ヘキサノイル基、ヘプタノイル基等が具体例として挙げられ、各々の指定の炭素原子数の範囲で選択される。 The expression “C a -C b alkylcarbonyl” represents an alkyl-C (O) — group having the above-mentioned meaning consisting of a to b carbon atoms, for example, an acetyl group, a propionyl group, a butyryl group, Specific examples include isobutyryl group, valeryl group, isovaleryl group, 2-methylbutanoyl group, pivaloyl group, hexanoyl group, heptanoyl group and the like, and each is selected in the range of the designated number of carbon atoms.
「ハロ(Ca〜Cb)アルキルカルボニル」の表記は、炭素原子数がa〜b個よりなる前記の意味であるハロアルキル−C(O)−基を表し、例えば、フルオロアセチル基、クロロアセチル基、ジフルオロアセチル基、ジクロロアセチル基、トリフルオロアセチル基、クロロジフルオロアセチル基、ブロモジフルオロアセチル基、トリクロロアセチル基、ペンタフルオロプロピオニル基、ヘプタフルオロブタノイル基、3−クロロ−2,2,−ジメチルプロパノイル基等が具体例として挙げられ、各々の指定の炭素原子数の範囲で選択される。 The expression “halo (C a -C b ) alkylcarbonyl” represents a haloalkyl-C (O) — group as defined above consisting of a to b carbon atoms, for example, a fluoroacetyl group, a chloroacetyl group, Group, difluoroacetyl group, dichloroacetyl group, trifluoroacetyl group, chlorodifluoroacetyl group, bromodifluoroacetyl group, trichloroacetyl group, pentafluoropropionyl group, heptafluorobutanoyl group, 3-chloro-2,2, -dimethyl Specific examples include a propanoyl group and the like, and each is selected within the range of the designated number of carbon atoms.
「Ca〜Cbアルコキシカルボニル」の表記は、炭素原子数がa〜b個よりなる前記の意味であるアルキル−OC(O)−基を表し、例えばメトキシカルボニル基、エトキシカルボニル基、n−プロピルオキシカルボニル基、i−プロピルオキシカルボニル基、n−ブトキシカルボニル基、i−ブトキシカルボニル基、s−ブトキシカルボニル基、tert−ブトキシカルボニル基、2−エチルへキシルオキシカルボニル基等が具体例として挙げられ、各々の指定の炭素原子数の範囲で選択される。 The expression “C a -C b alkoxycarbonyl” represents an alkyl-OC (O) — group having the above-mentioned meaning consisting of a to b carbon atoms, such as a methoxycarbonyl group, an ethoxycarbonyl group, n- Specific examples include propyloxycarbonyl group, i-propyloxycarbonyl group, n-butoxycarbonyl group, i-butoxycarbonyl group, s-butoxycarbonyl group, tert-butoxycarbonyl group, 2-ethylhexyloxycarbonyl group and the like. Selected within a range of each specified number of carbon atoms.
「Ca〜Cbアルコキシカルボニル(Cd〜Ce)アルキル」の表記は、炭素原子数がa〜b個よりなる前記の意味であるCa〜Cbアルコキシカルボニルによって、炭素原子に結合した水素原子が任意に置換された、炭素原子数がd〜e個よりなる前記の意味であるアルキル基を表し、例えば、2−メトキシカルボニルエチル基、2−エトキシカルボニルエチル基、2−(2−オクチルオキシカルボニル)エチル基等が具体例として挙げられ、各々の指定の炭素原子数の範囲で選択される。 The notation “C a -C b alkoxycarbonyl (C d -C e ) alkyl” is attached to a carbon atom by C a -C b alkoxycarbonyl which is the above-mentioned meaning consisting of a to b carbon atoms. It represents an alkyl group having the above-mentioned meaning consisting of de to e carbon atoms, optionally substituted with hydrogen atoms, such as 2-methoxycarbonylethyl group, 2-ethoxycarbonylethyl group, 2- (2- Specific examples include an octyloxycarbonyl) ethyl group and the like, and each is selected within the range of the designated number of carbon atoms.
「ハロ(Ca〜Cb)アルコキシカルボニル」の表記は、炭素原子数がa〜b個よりなる前記の意味であるハロアルキル−OC(O)−基を表し、例えば、クロロメトキシカルボニル基、2−クロロエトキシカルボニル基、2,2−ジフルオロエトキシカルボニル基、2,2,2−トリフルオロエトキシカルボニル基、2,2,2−トリクロロエトキシカルボニル基等が具体例として挙げられ、各々の指定の炭素原子数の範囲で選択される。 The expression “halo (C a -C b ) alkoxycarbonyl” represents a haloalkyl-OC (O) — group as defined above consisting of a to b carbon atoms, such as a chloromethoxycarbonyl group, 2 Specific examples include -chloroethoxycarbonyl group, 2,2-difluoroethoxycarbonyl group, 2,2,2-trifluoroethoxycarbonyl group, 2,2,2-trichloroethoxycarbonyl group and the like. It is selected in the range of the number of atoms.
「Ca〜Cbシクロアルキル」の表記は、炭素原子数がa〜b個よりなる環状の炭化水素を表し、3員環から6員環までの単環又は複合環構造を形成することが出来る。また、各々の環は指定の炭素原子数の範囲でアルキル基によって任意に置換されていてもよい。例えばシクロプロピル基、1−メチルシクロプロピル基、2−メチルシクロプロピル基、2,2−ジメチルシクロプロピル基、シクロブチル基、シクロペンチル基、シクロヘキシル基等が具体例として挙げられ、各々の指定の炭素原子数の範囲で選択される。 The expression “C a -C b cycloalkyl” represents a cyclic hydrocarbon composed of a to b carbon atoms, and may form a monocyclic or complex ring structure having 3 to 6 members. I can do it. Each ring may be optionally substituted with an alkyl group within the range of the specified number of carbon atoms. Specific examples include cyclopropyl group, 1-methylcyclopropyl group, 2-methylcyclopropyl group, 2,2-dimethylcyclopropyl group, cyclobutyl group, cyclopentyl group, cyclohexyl group, etc. Selected in a range of numbers.
「ハロ(Ca〜Cb)シクロアルキル」の表記は、炭素原子に結合した水素原子が、ハロゲン原子によって任意に置換された、炭素原子数がa〜b個よりなる環状の炭化水素を表し、3員環から6員環までの単環又は複合環構造を形成することが出来る。また、各々の環は指定の炭素原子数の範囲でアルキル基によって任意に置換されていてもよく、ハロゲン原子による置換は環構造部分であっても、側鎖部分であっても、或いはそれらの両方であってもよく、さらに、2個以上のハロゲン原子によって置換されている場合、それらのハロゲン原子は互いに同一でも、又は互いに相異なっていてもよい。例えば2,2−ジフルオロシクロプロピル基、2,2−ジクロロシクロプロピル基、2,2−ジブロモシクロプロピル基、2,2−ジフルオロ−1−メチルシクロプロピル基、2,2−ジクロロ−1−メチルシクロプロピル基、2,2−ジブロモ−1−メチルシクロプロピル基、2,2,3,3−テトラフルオロシクロブチル基等が具体例として挙げられ、各々の指定の炭素原子数の範囲で選択される。 The expression “halo (C a -C b ) cycloalkyl” represents a cyclic hydrocarbon composed of a to b carbon atoms in which a hydrogen atom bonded to a carbon atom is optionally substituted with a halogen atom. A monocyclic or complex ring structure from 3 to 6-membered ring can be formed. Each ring may be optionally substituted with an alkyl group within the range of the specified number of carbon atoms, and the substitution with a halogen atom may be a ring structure part, a side chain part, They may be both, and when they are substituted by two or more halogen atoms, the halogen atoms may be the same as or different from each other. For example, 2,2-difluorocyclopropyl group, 2,2-dichlorocyclopropyl group, 2,2-dibromocyclopropyl group, 2,2-difluoro-1-methylcyclopropyl group, 2,2-dichloro-1-methyl Specific examples include cyclopropyl group, 2,2-dibromo-1-methylcyclopropyl group, 2,2,3,3-tetrafluorocyclobutyl group, and the like, and each is selected within the range of the designated number of carbon atoms. The
「Ca〜Cbシクロアルケニル」の表記は、炭素原子数がa〜b個よりなる環状の、且つ1個又は2個以上の二重結合を有する不飽和炭化水素を表し、3員環から6員環までの単環又は複合環構造を形成することが出来る。また、各々の環は指定の炭素原子数の範囲でアルキル基によって任意に置換されていてもよく、さらに、二重結合はendo−又はexo−のどちらの形式であってもよい。例えば1−シクロペンテン−1−イル基、2−シクロペンテン−1−イル基、1−シクロヘキセン−1−イル基、2−シクロヘキセン−1−イル基等が具体例として挙げられ、各々の指定の炭素原子数の範囲で選択される。 The expression “C a -C b cycloalkenyl” represents a cyclic unsaturated hydrocarbon having 1 to 2 carbon atoms and having 1 or 2 or more double bonds. Monocyclic or complex ring structures up to 6-membered rings can be formed. Each ring may be optionally substituted with an alkyl group within the range of the specified number of carbon atoms, and the double bond may be either endo- or exo-. Specific examples include 1-cyclopenten-1-yl group, 2-cyclopenten-1-yl group, 1-cyclohexen-1-yl group, 2-cyclohexen-1-yl group, and the like. Selected in a range of numbers.
「ハロ(Ca〜Cb)シクロアルケニル」の表記は、炭素原子に結合した水素原子がハロゲン原子によって任意に置換された、炭素原子数がa〜b個よりなる環状の、且つ1個又は2個以上の二重結合を有する不飽和炭化水素基を表し、3員環から6員環までの単環又は複合環構造を形成することが出来る。また、各々の環は指定の炭素原子数の範囲でアルキル基によって任意に置換されていてもよく、さらに、二重結合はendo−又はexo−のどちらの形式であってもよい。また、ハロゲン原子による置換は環構造部分であっても、側鎖部分であっても、或いはそれらの両方であってもよく、2個以上のハロゲン原子によって置換されている場合、それらのハロゲン原子は互いに同一でも、又は互いに相異なっていても良い。例えば2-フルオロ-1-シクロペンテニル基、2-クロロ-1-シクロペンテニル基、3-クロロ-2-シクロペンテニル基、2-フルオロ-1-シクロヘキセニル基等が具体例として挙げられ、各々の指定の炭素原子数の範囲で選択される。 The notation “halo (C a -C b ) cycloalkenyl” is cyclic, consisting of a to b carbon atoms, in which a hydrogen atom bonded to a carbon atom is optionally substituted by a halogen atom, It represents an unsaturated hydrocarbon group having two or more double bonds, and can form a monocyclic or complex ring structure having 3 to 6 members. Each ring may be optionally substituted with an alkyl group within the range of the specified number of carbon atoms, and the double bond may be either endo- or exo-. In addition, the substitution by a halogen atom may be a ring structure part, a side chain part or both of them, and when substituted by two or more halogen atoms, those halogen atoms May be the same as or different from each other. For example, 2-fluoro-1-cyclopentenyl group, 2-chloro-1-cyclopentenyl group, 3-chloro-2-cyclopentenyl group, 2-fluoro-1-cyclohexenyl group and the like are listed as specific examples. Selected within the specified number of carbon atoms.
式(1)で表される本発明化合物の具体的な例を第1表及び第2表に示す。尚、表中「Me」の記載はメチル基、「Et」の記載はエチル基、「n−Pr」の記載はノルマルプロピル基、「c−Pr」の記載はシクロプロピル基、「Ac」の表記はアセチル基を表す。 Specific examples of the compound of the present invention represented by the formula (1) are shown in Tables 1 and 2. In the table, "Me" is a methyl group, "Et" is an ethyl group, "n-Pr" is a normal propyl group, "c-Pr" is a cyclopropyl group, and "Ac" The notation represents an acetyl group.
[第1表] [Table 1]
―――――――――――――――――――――――――――
n Y2 Y3 Y4 R2 X1 X2 X3
―――――――――――――――――――――――――――
0 CF3 H H Me H H CF3
2 CF3 H H Me H H CF3
2 H CF3 H H H CF3 H
2 H CF3 H n-Pr H CF3 H
0 CF3 H H Me NH2 H CF3
1 CF3 H H Me NH2 H CF3
2 CF3 H H Me NH2 H CF3
0 I H H Me NH2 H CF3
1 I H H Me NH2 H CF3
2 I H H Me NH2 H CF3
0 Br H H Me NH2 H CF3
1 Br H H Me NH2 H CF3
2 Br H H Me NH2 H CF3
0 Cl H H Me NH2 H CF3
1 Cl H H Me NH2 H CF3
2 Cl H H Me NH2 H CF3
0 c-Pr H H Me NH2 H CF3
1 c-Pr H H Me NH2 H CF3
2 c-Pr H H Me NH2 H CF3
0 NH2 H H Me NH2 H CF3
1 NH2 H H Me NH2 H CF3
2 NH2 H H Me NH2 H CF3
0 H CF3 H Me NH2 H CF3
1 H CF3 H Me NH2 H CF3
2 H CF3 H Me NH2 H CF3
0 H I H Me NH2 H CF3
1 H I H Me NH2 H CF3
2 H I H Me NH2 H CF3
0 H Br H Me NH2 H CF3
1 H Br H Me NH2 H CF3
2 H Br H Me NH2 H CF3
0 H Cl H Me NH2 H CF3
1 H Cl H Me NH2 H CF3
2 H Cl H Me NH2 H CF3
0 H c-Pr H Me NH2 H CF3
1 H c-Pr H Me NH2 H CF3
2 H c-Pr H Me NH2 H CF3
0 H NH2 H Me NH2 H CF3
1 H NH2 H Me NH2 H CF3
2 H NH2 H Me NH2 H CF3
0 H NHAc H Me NH2 H CF3
1 H NHAc H Me NH2 H CF3
2 H NHAc H Me NH2 H CF3
0 H NO2 H Me NH2 H CF3
1 H NO2 H Me NH2 H CF3
2 H NO2 H Me NH2 H CF3
0 H C(O)OH H Me NH2 H CF3
1 H C(O)OH H Me NH2 H CF3
2 H C(O)OH H Me NH2 H CF3
0 H C(O)OEt H Me NH2 H CF3
1 H C(O)OEt H Me NH2 H CF3
2 H C(O)OEt H Me NH2 H CF3
0 H CN H Me NH2 H CF3
1 H CN H Me NH2 H CF3
2 H CN H Me NH2 H CF3
0 H H NH2 Me NH2 H CF3
1 H H NH2 Me NH2 H CF3
2 H H NH2 Me NH2 H CF3
0 H H H Me NH2 H CF3
1 H H H Me NH2 H CF3
2 H H H Me NH2 H CF3
0 CF3 H H Me NO2 H CF3
1 CF3 H H Me NO2 H CF3
2 CF3 H H Me NO2 H CF3
0 I H H Me NO2 H CF3
1 I H H Me NO2 H CF3
2 I H H Me NO2 H CF3
0 Br H H Me NO2 H CF3
1 Br H H Me NO2 H CF3
2 Br H H Me NO2 H CF3
0 Cl H H Me NO2 H CF3
1 Cl H H Me NO2 H CF3
2 Cl H H Me NO2 H CF3
0 c-Pr H H Me NO2 H CF3
1 c-Pr H H Me NO2 H CF3
2 c-Pr H H Me NO2 H CF3
0 NH2 H H Me NO2 H CF3
1 NH2 H H Me NO2 H CF3
2 NH2 H H Me NO2 H CF3
0 H CF3 H Me NO2 H CF3
1 H CF3 H Me NO2 H CF3
2 H CF3 H Me NO2 H CF3
0 H I H Me NO2 H CF3
1 H I H Me NO2 H CF3
2 H I H Me NO2 H CF3
0 H Br H Me NO2 H CF3
1 H Br H Me NO2 H CF3
2 H Br H Me NO2 H CF3
0 H Cl H Me NO2 H CF3
1 H Cl H Me NO2 H CF3
2 H Cl H Me NO2 H CF3
0 H c-Pr H Me NO2 H CF3
1 H c-Pr H Me NO2 H CF3
2 H c-Pr H Me NO2 H CF3
0 H NH2 H Me NO2 H CF3
1 H NH2 H Me NO2 H CF3
2 H NH2 H Me NO2 H CF3
0 H NHAc H Me NO2 H CF3
1 H NHAc H Me NO2 H CF3
2 H NHAc H Me NO2 H CF3
0 H NO2 H Me NO2 H CF3
1 H NO2 H Me NO2 H CF3
2 H NO2 H Me NO2 H CF3
0 H C(O)OH H Me NO2 H CF3
1 H C(O)OH H Me NO2 H CF3
2 H C(O)OH H Me NO2 H CF3
0 H C(O)OEt H Me NO2 H CF3
1 H C(O)OEt H Me NO2 H CF3
2 H C(O)OEt H Me NO2 H CF3
0 H CN H Me NO2 H CF3
1 H CN H Me NO2 H CF3
2 H CN H Me NO2 H CF3
0 H H NH2 Me NO2 H CF3
1 H H NH2 Me NO2 H CF3
2 H H NH2 Me NO2 H CF3
0 H H H Me NO2 H CF3
1 H H H Me NO2 H CF3
2 H H H Me NO2 H CF3
0 CF3 H H Me Cl H CF3
1 CF3 H H Me Cl H CF3
2 CF3 H H Me Cl H CF3
0 I H H Me Cl H CF3
1 I H H Me Cl H CF3
2 I H H Me Cl H CF3
0 Br H H Me Cl H CF3
1 Br H H Me Cl H CF3
2 Br H H Me Cl H CF3
0 Cl H H Me Cl H CF3
1 Cl H H Me Cl H CF3
2 Cl H H Me Cl H CF3
0 c-Pr H H Me Cl H CF3
1 c-Pr H H Me Cl H CF3
2 c-Pr H H Me Cl H CF3
0 NH2 H H Me Cl H CF3
1 NH2 H H Me Cl H CF3
2 NH2 H H Me Cl H CF3
0 H CF3 H Me Cl H CF3
1 H CF3 H Me Cl H CF3
2 H CF3 H Me Cl H CF3
0 H I H Me Cl H CF3
1 H I H Me Cl H CF3
2 H I H Me Cl H CF3
0 H Br H Me Cl H CF3
1 H Br H Me Cl H CF3
2 H Br H Me Cl H CF3
0 H Cl H Me Cl H CF3
1 H Cl H Me Cl H CF3
2 H Cl H Me Cl H CF3
0 H c-Pr H Me Cl H CF3
1 H c-Pr H Me Cl H CF3
2 H c-Pr H Me Cl H CF3
0 H NH2 H Me Cl H CF3
1 H NH2 H Me Cl H CF3
2 H NH2 H Me Cl H CF3
0 H NHAc H Me Cl H CF3
1 H NHAc H Me Cl H CF3
2 H NHAc H Me Cl H CF3
0 H NO2 H Me Cl H CF3
1 H NO2 H Me Cl H CF3
2 H NO2 H Me Cl H CF3
0 H C(O)OH H Me Cl H CF3
1 H C(O)OH H Me Cl H CF3
2 H C(O)OH H Me Cl H CF3
0 H C(O)OEt H Me Cl H CF3
1 H C(O)OEt H Me Cl H CF3
2 H C(O)OEt H Me Cl H CF3
0 H CN H Me Cl H CF3
1 H CN H Me Cl H CF3
2 H CN H Me Cl H CF3
0 H H NH2 Me Cl H CF3
1 H H NH2 Me Cl H CF3
2 H H NH2 Me Cl H CF3
0 H H H Me Cl H CF3
1 H H H Me Cl H CF3
2 H H H Me Cl H CF3
―――――――――――――――――――――――――――
[第2表]
―――――――――――――――――――――――――――
n Y2 Y3 Y4 R 2 X 1 X 2 X 3
―――――――――――――――――――――――――――
0 CF 3 HH Me HH CF 3
2 CF 3 HH Me HH CF 3
2 H CF 3 HHH CF 3 H
2 H CF 3 H n-Pr H CF 3 H
0 CF 3 HH Me NH 2 H CF 3
1 CF 3 HH Me NH 2 H CF 3
2 CF 3 HH Me NH 2 H CF 3
0 IHH Me NH 2 H CF 3
1 IHH Me NH 2 H CF 3
2 IHH Me NH 2 H CF 3
0 Br HH Me NH 2 H CF 3
1 Br HH Me NH 2 H CF 3
2 Br HH Me NH 2 H CF 3
0 Cl HH Me NH 2 H CF 3
1 Cl HH Me NH 2 H CF 3
2 Cl HH Me NH 2 H CF 3
0 c-Pr HH Me NH 2 H CF 3
1 c-Pr HH Me NH 2 H CF 3
2 c-Pr HH Me NH 2 H CF 3
0 NH 2 HH Me NH 2 H CF 3
1 NH 2 HH Me NH 2 H CF 3
2 NH 2 HH Me NH 2 H CF 3
0 H CF 3 H Me NH 2 H CF 3
1 H CF 3 H Me NH 2 H CF 3
2 H CF 3 H Me NH 2 H CF 3
0 HIH Me NH 2 H CF 3
1 HIH Me NH 2 H CF 3
2 HIH Me NH 2 H CF 3
0 H Br H Me NH 2 H CF 3
1 H Br H Me NH 2 H CF 3
2 H Br H Me NH 2 H CF 3
0 H Cl H Me NH 2 H CF 3
1 H Cl H Me NH 2 H CF 3
2 H Cl H Me NH 2 H CF 3
0 H c-Pr H Me NH 2 H CF 3
1 H c-Pr H Me NH 2 H CF 3
2 H c-Pr H Me NH 2 H CF 3
0 H NH 2 H Me NH 2 H CF 3
1 H NH 2 H Me NH 2 H CF 3
2 H NH 2 H Me NH 2 H CF 3
0 H NHAc H Me NH 2 H CF 3
1 H NHAc H Me NH 2 H CF 3
2 H NHAc H Me NH 2 H CF 3
0 H NO 2 H Me NH 2 H CF 3
1 H NO 2 H Me NH 2 H CF 3
2 H NO 2 H Me NH 2 H CF 3
0 HC (O) OH H Me NH 2 H CF 3
1 HC (O) OH H Me NH 2 H CF 3
2 HC (O) OH H Me NH 2 H CF 3
0 HC (O) OEt H Me NH 2 H CF 3
1 HC (O) OEt H Me NH 2 H CF 3
2 HC (O) OEt H Me NH 2 H CF 3
0 H CN H Me NH 2 H CF 3
1 H CN H Me NH 2 H CF 3
2 H CN H Me NH 2 H CF 3
0 HH NH 2 Me NH 2 H CF 3
1 HH NH 2 Me NH 2 H CF 3
2 HH NH 2 Me NH 2 H CF 3
0 HHH Me NH 2 H CF 3
1 HHH Me NH 2 H CF 3
2 HHH Me NH 2 H CF 3
0 CF 3 HH Me NO 2 H CF 3
1 CF 3 HH Me NO 2 H CF 3
2 CF 3 HH Me NO 2 H CF 3
0 IHH Me NO 2 H CF 3
1 IHH Me NO 2 H CF 3
2 IHH Me NO 2 H CF 3
0 Br HH Me NO 2 H CF 3
1 Br HH Me NO 2 H CF 3
2 Br HH Me NO 2 H CF 3
0 Cl HH Me NO 2 H CF 3
1 Cl HH Me NO 2 H CF 3
2 Cl HH Me NO 2 H CF 3
0 c-Pr HH Me NO 2 H CF 3
1 c-Pr HH Me NO 2 H CF 3
2 c-Pr HH Me NO 2 H CF 3
0 NH 2 HH Me NO 2 H CF 3
1 NH 2 HH Me NO 2 H CF 3
2 NH 2 HH Me NO 2 H CF 3
0 H CF 3 H Me NO 2 H CF 3
1 H CF 3 H Me NO 2 H CF 3
2 H CF 3 H Me NO 2 H CF 3
0 HIH Me NO 2 H CF 3
1 HIH Me NO 2 H CF 3
2 HIH Me NO 2 H CF 3
0 H Br H Me NO 2 H CF 3
1 H Br H Me NO 2 H CF 3
2 H Br H Me NO 2 H CF 3
0 H Cl H Me NO 2 H CF 3
1 H Cl H Me NO 2 H CF 3
2 H Cl H Me NO 2 H CF 3
0 H c-Pr H Me NO 2 H CF 3
1 H c-Pr H Me NO 2 H CF 3
2 H c-Pr H Me NO 2 H CF 3
0 H NH 2 H Me NO 2 H CF 3
1 H NH 2 H Me NO 2 H CF 3
2 H NH 2 H Me NO 2 H CF 3
0 H NHAc H Me NO 2 H CF 3
1 H NHAc H Me NO 2 H CF 3
2 H NHAc H Me NO 2 H CF 3
0 H NO 2 H Me NO 2 H CF 3
1 H NO 2 H Me NO 2 H CF 3
2 H NO 2 H Me NO 2 H CF 3
0 HC (O) OH H Me NO 2 H CF 3
1 HC (O) OH H Me NO 2 H CF 3
2 HC (O) OH H Me NO 2 H CF 3
0 HC (O) OEt H Me NO 2 H CF 3
1 HC (O) OEt H Me NO 2 H CF 3
2 HC (O) OEt H Me NO 2 H CF 3
0 H CN H Me NO 2 H CF 3
1 H CN H Me NO 2 H CF 3
2 H CN H Me NO 2 H CF 3
0 HH NH 2 Me NO 2 H CF 3
1 HH NH 2 Me NO 2 H CF 3
2 HH NH 2 Me NO 2 H CF 3
0 HHH Me NO 2 H CF 3
1 HHH Me NO 2 H CF 3
2 HHH Me NO 2 H CF 3
0 CF 3 HH Me Cl H CF 3
1 CF 3 HH Me Cl H CF 3
2 CF 3 HH Me Cl H CF 3
0 IHH Me Cl H CF 3
1 IHH Me Cl H CF 3
2 IHH Me Cl H CF 3
0 Br HH Me Cl H CF 3
1 Br HH Me Cl H CF 3
2 Br HH Me Cl H CF 3
0 Cl HH Me Cl H CF 3
1 Cl HH Me Cl H CF 3
2 Cl HH Me Cl H CF 3
0 c-Pr HH Me Cl H CF 3
1 c-Pr HH Me Cl H CF 3
2 c-Pr HH Me Cl H CF 3
0 NH 2 HH Me Cl H CF 3
1 NH 2 HH Me Cl H CF 3
2 NH 2 HH Me Cl H CF 3
0 H CF 3 H Me Cl H CF 3
1 H CF 3 H Me Cl H CF 3
2 H CF 3 H Me Cl H CF 3
0 HIH Me Cl H CF 3
1 HIH Me Cl H CF 3
2 HIH Me Cl H CF 3
0 H Br H Me Cl H CF 3
1 H Br H Me Cl H CF 3
2 H Br H Me Cl H CF 3
0 H Cl H Me Cl H CF 3
1 H Cl H Me Cl H CF 3
2 H Cl H Me Cl H CF 3
0 H c-Pr H Me Cl H CF 3
1 H c-Pr H Me Cl H CF 3
2 H c-Pr H Me Cl H CF 3
0 H NH 2 H Me Cl H CF 3
1 H NH 2 H Me Cl H CF 3
2 H NH 2 H Me Cl H CF 3
0 H NHAc H Me Cl H CF 3
1 H NHAc H Me Cl H CF 3
2 H NHAc H Me Cl H CF 3
0 H NO 2 H Me Cl H CF 3
1 H NO 2 H Me Cl H CF 3
2 H NO 2 H Me Cl H CF 3
0 HC (O) OH H Me Cl H CF 3
1 HC (O) OH H Me Cl H CF 3
2 HC (O) OH H Me Cl H CF 3
0 HC (O) OEt H Me Cl H CF 3
1 HC (O) OEt H Me Cl H CF 3
2 HC (O) OEt H Me Cl H CF 3
0 H CN H Me Cl H CF 3
1 H CN H Me Cl H CF 3
2 H CN H Me Cl H CF 3
0 HH NH 2 Me Cl H CF 3
1 HH NH 2 Me Cl H CF 3
2 HH NH 2 Me Cl H CF 3
0 HHH Me Cl H CF 3
1 HHH Me Cl H CF 3
2 HHH Me Cl H CF 3
―――――――――――――――――――――――――――
[Table 2]
―――――――――――――――――― ――――――――――――――――――
n Y2 Y3 Y4 X1 X4 n Y2 Y3 Y4 X1 X4
―――――――――――――――――― ――――――――――――――――――
0 CF3 H H Cl CF3 0 CF3 H H NHMe CF3
1 CF3 H H Cl CF3 1 CF3 H H NHMe CF3
2 CF3 H H Cl CF3 2 CF3 H H NHMe CF3
0 I H H Cl CF3 0 I H H NHMe CF3
1 I H H Cl CF3 1 I H H NHMe CF3
2 I H H Cl CF3 2 I H H NHMe CF3
0 Br H H Cl CF3 0 Br H H NHMe CF3
1 Br H H Cl CF3 1 Br H H NHMe CF3
2 Br H H Cl CF3 2 Br H H NHMe CF3
0 Cl H H Cl CF3 0 Cl H H NHMe CF3
1 Cl H H Cl CF3 1 Cl H H NHMe CF3
2 Cl H H Cl CF3 2 Cl H H NHMe CF3
0 c-Pr H H Cl CF3 0 c-Pr H H NHMe CF3
1 c-Pr H H Cl CF3 1 c-Pr H H NHMe CF3
2 c-Pr H H Cl CF3 2 c-Pr H H NHMe CF3
0 NH2 H H Cl CF3 0 NH2 H H NHMe CF3
1 NH2 H H Cl CF3 1 NH2 H H NHMe CF3
2 NH2 H H Cl CF3 2 NH2 H H NHMe CF3
0 H CF3 H Cl CF3 0 H CF3 H NHMe CF3
1 H CF3 H Cl CF3 1 H CF3 H NHMe CF3
2 H CF3 H Cl CF3 2 H CF3 H NHMe CF3
0 H I H Cl CF3 0 H I H NHMe CF3
1 H I H Cl CF3 1 H I H NHMe CF3
2 H I H Cl CF3 2 H I H NHMe CF3
0 H Br H Cl CF3 0 H Br H NHMe CF3
1 H Br H Cl CF3 1 H Br H NHMe CF3
2 H Br H Cl CF3 2 H Br H NHMe CF3
0 H Cl H Cl CF3 0 H Cl H NHMe CF3
1 H Cl H Cl CF3 1 H Cl H NHMe CF3
2 H Cl H Cl CF3 2 H Cl H NHMe CF3
0 H c-Pr H Cl CF3 0 H c-Pr H NHMe CF3
1 H c-Pr H Cl CF3 1 H c-Pr H NHMe CF3
2 H c-Pr H Cl CF3 2 H c-Pr H NHMe CF3
0 H NH2 H Cl CF3 0 H NH2 H NHMe CF3
1 H NH2 H Cl CF3 1 H NH2 H NHMe CF3
2 H NH2 H Cl CF3 2 H NH2 H NHMe CF3
0 H NHAc H Cl CF3 0 H NHAc H NHMe CF3
1 H NHAc H Cl CF3 1 H NHAc H NHMe CF3
2 H NHAc H Cl CF3 2 H NHAc H NHMe CF3
0 H NO2 H Cl CF3 0 H NO2 H NHMe CF3
1 H NO2 H Cl CF3 1 H NO2 H NHMe CF3
2 H NO2 H Cl CF3 2 H NO2 H NHMe CF3
0 H C(O)OH H Cl CF3 0 H C(O)OH H NHMe CF3
1 H C(O)OH H Cl CF3 1 H C(O)OH H NHMe CF3
2 H C(O)OH H Cl CF3 2 H C(O)OH H NHMe CF3
0 H C(O)OEt H Cl CF3 0 H C(O)OEt H NHMe CF3
1 H C(O)OEt H Cl CF3 1 H C(O)OEt H NHMe CF3
2 H C(O)OEt H Cl CF3 2 H C(O)OEt H NHMe CF3
0 H CN H Cl CF3 0 H CN H NHMe CF3
1 H CN H Cl CF3 1 H CN H NHMe CF3
2 H CN H Cl CF3 2 H CN H NHMe CF3
0 H H NH2 Cl CF3 0 H H NH2 NHMe CF3
1 H H NH2 Cl CF3 1 H H NH2 NHMe CF3
2 H H NH2 Cl CF3 2 H H NH2 NHMe CF3
0 H H H Cl CF3 0 H H H NHMe CF3
1 H H H Cl CF3 1 H H H NHMe CF3
2 H H H Cl CF3 2 H H H NHMe CF3
―――――――――――――――――― ――――――――――――――――――
次に、前記式(1)で表される本発明化合物の製造法を以下に説明する。本発明化合物は、例えば、以下の製造法1〜製造法4により製造することができる。
―――――――――――――――――― ――――――――――――――――――
n Y2 Y3 Y4 X 1 X 4 n Y2 Y3 Y4 X 1 X 4
―――――――――――――――――― ――――――――――――――――――
0 CF 3 HH Cl CF 3 0 CF 3 HH NHMe CF 3
1 CF 3 HH Cl CF 3 1 CF 3 HH NHMe CF 3
2 CF 3 HH Cl CF 3 2 CF 3 HH NHMe CF 3
0 IHH Cl CF 3 0 IHH NHMe CF 3
1 IHH Cl CF 3 1 IHH NHMe CF 3
2 IHH Cl CF 3 2 IHH NHMe CF 3
0 Br HH Cl CF 3 0 Br HH NHMe CF 3
1 Br HH Cl CF 3 1 Br HH NHMe CF 3
2 Br HH Cl CF 3 2 Br HH NHMe CF 3
0 Cl HH Cl CF 3 0 Cl HH NHMe CF 3
1 Cl HH Cl CF 3 1 Cl HH NHMe CF 3
2 Cl HH Cl CF 3 2 Cl HH NHMe CF 3
0 c-Pr HH Cl CF 3 0 c-Pr HH NHMe CF 3
1 c-Pr HH Cl CF 3 1 c-Pr HH NHMe CF 3
2 c-Pr HH Cl CF 3 2 c-Pr HH NHMe CF 3
0 NH 2 HH Cl CF 3 0 NH 2 HH NHMe CF 3
1 NH 2 HH Cl CF 3 1 NH 2 HH NHMe CF 3
2 NH 2 HH Cl CF 3 2 NH 2 HH NHMe CF 3
0 H CF 3 H Cl CF 3 0 H CF 3 H NHMe CF 3
1 H CF 3 H Cl CF 3 1 H CF 3 H NHMe CF 3
2 H CF 3 H Cl CF 3 2 H CF 3 H NHMe CF 3
0 HIH Cl CF 3 0 HIH NHMe CF 3
1 HIH Cl CF 3 1 HIH NHMe CF 3
2 HIH Cl CF 3 2 HIH NHMe CF 3
0 H Br H Cl CF 3 0 H Br H NHMe CF 3
1 H Br H Cl CF 3 1 H Br H NHMe CF 3
2 H Br H Cl CF 3 2 H Br H NHMe CF 3
0 H Cl H Cl CF 3 0 H Cl H NHMe CF 3
1 H Cl H Cl CF 3 1 H Cl H NHMe CF 3
2 H Cl H Cl CF 3 2 H Cl H NHMe CF 3
0 H c-Pr H Cl CF 3 0 H c-Pr H NHMe CF 3
1 H c-Pr H Cl CF 3 1 H c-Pr H NHMe CF 3
2 H c-Pr H Cl CF 3 2 H c-Pr H NHMe CF 3
0 H NH 2 H Cl CF 3 0 H NH 2 H NHMe CF 3
1 H NH 2 H Cl CF 3 1 H NH 2 H NHMe CF 3
2 H NH 2 H Cl CF 3 2 H NH 2 H NHMe CF 3
0 H NHAc H Cl CF 3 0 H NHAc H NHMe CF 3
1 H NHAc H Cl CF 3 1 H NHAc H NHMe CF 3
2 H NHAc H Cl CF 3 2 H NHAc H NHMe CF 3
0 H NO 2 H Cl CF 3 0 H NO 2 H NHMe CF 3
1 H NO 2 H Cl CF 3 1 H NO 2 H NHMe CF 3
2 H NO 2 H Cl CF 3 2 H NO 2 H NHMe CF 3
0 HC (O) OH H Cl CF 3 0 HC (O) OH H NHMe CF 3
1 HC (O) OH H Cl CF 3 1 HC (O) OH H NHMe CF 3
2 HC (O) OH H Cl CF 3 2 HC (O) OH H NHMe CF 3
0 HC (O) OEt H Cl CF 3 0 HC (O) OEt H NHMe CF 3
1 HC (O) OEt H Cl CF 3 1 HC (O) OEt H NHMe CF 3
2 HC (O) OEt H Cl CF 3 2 HC (O) OEt H NHMe CF 3
0 H CN H Cl CF 3 0 H CN H NHMe CF 3
1 H CN H Cl CF 3 1 H CN H NHMe CF 3
2 H CN H Cl CF 3 2 H CN H NHMe CF 3
0 HH NH 2 Cl CF 3 0 HH NH 2 NHMe CF 3
1 HH NH 2 Cl CF 3 1 HH NH 2 NHMe CF 3
2 HH NH 2 Cl CF 3 2 HH NH 2 NHMe CF 3
0 HHH Cl CF 3 0 HHH NHMe CF 3
1 HHH Cl CF 3 1 HHH NHMe CF 3
2 HHH Cl CF 3 2 HHH NHMe CF 3
―――――――――――――――――― ――――――――――――――――――
Next, the manufacturing method of this invention compound represented by the said Formula (1) is demonstrated below. The compound of the present invention can be produced, for example, by the following production methods 1 to 4.
[製造法1]
前記式(1)で表される本発明化合物のうち、nが1又は2の整数である式(1−A)で表される化合物[以下、化合物(1−A)と略称する。]は、例えば、本発明化合物のうち、nが0の整数である式(1−B)で表される化合物[以下、化合物(1−B)と略称する。]と、酸化剤とを反応させることにより製造することができる。
[Production Method 1]
Among the compounds of the present invention represented by the formula (1), a compound represented by the formula (1-A) wherein n is an integer of 1 or 2 [hereinafter abbreviated as compound (1-A). ] Is, for example, a compound represented by the formula (1-B) in which n is an integer of 0 among the compounds of the present invention [hereinafter abbreviated as compound (1-B). And an oxidizing agent.
(式中、G、R1、R2、Y2、Y3及びY4は前記と同じ意味を表し、mは1又は2の整数を表す。)
化合物(1−A)は、化合物(1−B)と酸化剤とを、溶媒中又は無溶媒で、場合によっては触媒存在下、反応させることにより製造することができる。
(In the formula, G, R 1 , R 2 , Y 2 , Y 3 and Y 4 represent the same meaning as described above, and m represents an integer of 1 or 2.)
Compound (1-A) can be produced by reacting compound (1-B) with an oxidizing agent in a solvent or without a solvent, optionally in the presence of a catalyst.
溶媒を用いる場合、用いられる溶媒としては反応に不活性であればよく、例えば水、メタノール、エタノール等の低級アルコール類、ジエチルエーテル、テトラヒドロフラン、1,4−ジオキサン、1,2−ジメトキシエタン等のエーテル類、ベンゼン、クロロベンゼン、ブロモベンゼン、キシレン、トルエン等の芳香族炭化水素類、ペンタン、ヘキサン、シクロヘキサン等の脂肪族炭化水素類、ジクロロメタン、クロロホルム、1,2−ジクロロエタン等のハロゲン化炭化水素類、アセトニトリル、プロピオニトリル等のニトリル類、N,N−ジメチルホルムアミド、N,N−ジメチルアセトアミド、N−メチルピロリドン、N,N’−ジメチルイミダゾリジノン等のアミド類、ジメチルスルホキシド等のスルホキシド類、酢酸等の低級脂肪酸類又はこれらの混合溶媒等が挙げられる。 When using a solvent, the solvent used may be inert to the reaction, such as water, lower alcohols such as methanol and ethanol, diethyl ether, tetrahydrofuran, 1,4-dioxane, 1,2-dimethoxyethane and the like. Ethers, aromatic hydrocarbons such as benzene, chlorobenzene, bromobenzene, xylene, and toluene, aliphatic hydrocarbons such as pentane, hexane, and cyclohexane, and halogenated hydrocarbons such as dichloromethane, chloroform, and 1,2-dichloroethane Nitriles such as acetonitrile and propionitrile, amides such as N, N-dimethylformamide, N, N-dimethylacetamide, N-methylpyrrolidone and N, N′-dimethylimidazolidinone, and sulfoxides such as dimethyl sulfoxide , Lower fats such as acetic acid Acids or mixed solvents thereof.
酸化剤としては、例えばメタクロロ過安息香酸、過酢酸等の過酸、過酸化水素、オキソン(OXONE、イー・アイ・デュポン社登録商標;ぺルオキソ硫酸水素カリウム含有物)が挙げられる。酸化剤の当量としては、化合物(1−B)1当量に対して0.1から100当量の範囲で用いることができ、1から20当量の範囲が好ましい。 Examples of the oxidizing agent include peracids such as metachloroperbenzoic acid and peracetic acid, hydrogen peroxide, and oxone (OXONE, registered trademark of EI Dupont; containing potassium peroxosulfate). The equivalent of the oxidizing agent can be used in the range of 0.1 to 100 equivalents relative to 1 equivalent of compound (1-B), and preferably in the range of 1 to 20 equivalents.
該反応は触媒の存在下に行うことができる。用いることのできる触媒としては、例えばタングステン酸ナトリウム等が挙げられる。触媒の当量としては、化合物(1−B)1当量に対して0.005から20当量の範囲で用いることができ、0.1から5当量の範囲が好ましい。 The reaction can be carried out in the presence of a catalyst. Examples of the catalyst that can be used include sodium tungstate. As an equivalent of a catalyst, it can use in 0.005 to 20 equivalent with respect to 1 equivalent of compound (1-B), and the range of 0.1 to 5 equivalent is preferable.
反応温度は、−80℃から反応混合物の還流温度まで任意の温度で設定することができ、0℃から反応混合物の還流温度の範囲が好ましい。 The reaction temperature can be set at any temperature from −80 ° C. to the reflux temperature of the reaction mixture, and the range of 0 ° C. to the reflux temperature of the reaction mixture is preferred.
反応時間は、反応基質の濃度、反応温度によって変化するが、通常5分から100時間の範囲で任意に設定でき、1時間から48時間の範囲が好ましい。 The reaction time varies depending on the concentration of the reaction substrate and the reaction temperature, but can usually be arbitrarily set in the range of 5 minutes to 100 hours, and preferably in the range of 1 hour to 48 hours.
[製造法2]
前記式(1)で表される本発明化合物のうち、式(1−D)で表される化合物[以下、化合物(1−D)と略称する。]は、例えば、本発明化合物のうち、式(1−C)で表される化合物[以下、化合物(1−C)と略称する。]と、式(50)で表される化合物[以下、化合物(50)と略称する。]とを反応させることにより製造することができる。
[Production Method 2]
Among the compounds of the present invention represented by the formula (1), a compound represented by the formula (1-D) [hereinafter abbreviated as compound (1-D). ] Is a compound represented by the formula (1-C) among the compounds of the present invention [hereinafter abbreviated as compound (1-C). ] And a compound represented by formula (50) [hereinafter abbreviated as compound (50). ] Can be made to react.
[式中、G、R1、Y2、Y3、Y4及びnは前記と同じ意味を表し、nは0、1又は2の整数を表し、R2はC1〜C6アルキルを表し、X1はハロゲン原子、C1〜C4アルキルスルホネート(例えば、メタンスルホニルオキシ等)、C1〜C4ハロアルキルスルホネート(例えば、トリフルオロメタンスルホニルオキシ等)又はアリールスルホネート(例えば、ベンゼンスルホニルオキシ、p-トルエンスルホニルオキシ等)等の脱離基を表す。]
化合物(1−D)は、化合物(1−C)と化合物(50)とを、溶媒中又は無溶媒で、場合によっては塩基存在下反応させることで製造することができる。
[Wherein, G, R 1 , Y 2, Y 3, Y 4 and n represent the same meaning as described above, n represents an integer of 0, 1 or 2, R 2 represents C 1 -C 6 alkyl, and X 1 Is a halogen atom, C 1 -C 4 alkyl sulfonate (eg methanesulfonyloxy etc.), C 1 -C 4 haloalkyl sulfonate (eg trifluoromethanesulfonyloxy etc.) or aryl sulfonate (eg benzenesulfonyloxy, p-toluenesulfonyl etc.) Represents a leaving group such as oxy. ]
Compound (1-D) can be produced by reacting compound (1-C) and compound (50) in a solvent or without a solvent, and optionally in the presence of a base.
用いる化合物(50)の当量としては、化合物(1−C)1当量に対して、0.5から50当量の範囲で用いることができ、1から20当量の範囲が好ましい。 As an equivalent of the compound (50) to be used, it can be used in the range of 0.5-50 equivalent with respect to 1 equivalent of compound (1-C), and the range of 1-20 equivalent is preferable.
化合物(50)の或るものは公知化合物であり、一部は市販品として入手できる。 Some of the compounds (50) are known compounds, and some of them are commercially available.
溶媒を用いる場合、用いられる溶媒としては反応に不活性であればよく、例えば水、メタノール、エタノール等の低級アルコール類、ジエチルエーテル、テトラヒドロフラン、1,4−ジオキサン、1,2−ジメトキシエタン等のエーテル類、ベンゼン、クロロベンゼン、ブロモベンゼン、キシレン、トルエン等の芳香族炭化水素類、ペンタン、ヘキサン、シクロヘキサン等の脂肪族炭化水素類、ジクロロメタン、クロロホルム、1,2−ジクロロエタン等のハロゲン化炭化水素類、アセトニトリル、プロピオニトリル等のニトリル類、N,N−ジメチルホルムアミド、N,N−ジメチルアセトアミド、N−メチルピロリドン、N,N’−ジメチルイミダゾリジノン等のアミド類、ジメチルスルホキシド等のスルホキシド類、ピリジン、キノリン等の含窒素芳香族化合物類又はこれらの混合溶媒等が挙げられる。 When using a solvent, the solvent used may be inert to the reaction, such as water, lower alcohols such as methanol and ethanol, diethyl ether, tetrahydrofuran, 1,4-dioxane, 1,2-dimethoxyethane and the like. Ethers, aromatic hydrocarbons such as benzene, chlorobenzene, bromobenzene, xylene, and toluene, aliphatic hydrocarbons such as pentane, hexane, and cyclohexane, and halogenated hydrocarbons such as dichloromethane, chloroform, and 1,2-dichloroethane Nitriles such as acetonitrile and propionitrile, amides such as N, N-dimethylformamide, N, N-dimethylacetamide, N-methylpyrrolidone and N, N′-dimethylimidazolidinone, and sulfoxides such as dimethyl sulfoxide , Pyridine, quino Nitrogen-containing aromatic compounds or a mixture of these solvents, such as emissions and the like.
該反応は塩基の存在下に行うことができる。用いることのできる塩基としては、例えばピリジン、2,6−ルチジン、4−ジメチルアミノピリジン、トリエチルアミン、ジイソプロピルエチルアミン、トリブチルアミン、4-(ジメチルアミノ)ピリジン、1,4−ジアザビシクロ[2.2.2]オクタン(DABCO)、1,8−ジアザビシクロ[5.4.0]−7−ウンデセン(DBU)又は1,5−ジアザビシクロ[4.3.0]−5−ノネン(DBN)等の有機塩基類、水酸化ナトリウム、水酸化カリウム、水素化ナトリウム、炭酸水素ナトリウム、炭酸カリウム、炭酸セシウム等の無機塩基類が挙げられる。塩基の当量としては、化合物(1−C)1当量に対して0.1から100当量の範囲で用いることができ、1から20当量の範囲が好ましい。 The reaction can be performed in the presence of a base. Examples of the base that can be used include pyridine, 2,6-lutidine, 4-dimethylaminopyridine, triethylamine, diisopropylethylamine, tributylamine, 4- (dimethylamino) pyridine, 1,4-diazabicyclo [2.2.2. ] Organic bases such as octane (DABCO), 1,8-diazabicyclo [5.4.0] -7-undecene (DBU) or 1,5-diazabicyclo [4.3.0] -5-nonene (DBN) And inorganic bases such as sodium hydroxide, potassium hydroxide, sodium hydride, sodium hydrogen carbonate, potassium carbonate, cesium carbonate and the like. The equivalent of the base can be used in the range of 0.1 to 100 equivalents relative to 1 equivalent of the compound (1-C), and preferably in the range of 1 to 20 equivalents.
反応温度は、−80℃から反応混合物の還流温度まで任意の温度で設定することができ、0℃から反応混合物の還流温度の範囲が好ましい。 The reaction temperature can be set at any temperature from −80 ° C. to the reflux temperature of the reaction mixture, and the range of 0 ° C. to the reflux temperature of the reaction mixture is preferred.
反応時間は、反応基質の濃度、反応温度によって変化するが、通常5分から100時間の範囲で任意に設定でき、1時間から48時間の範囲が好ましい。 The reaction time varies depending on the concentration of the reaction substrate and the reaction temperature, but can usually be arbitrarily set in the range of 5 minutes to 100 hours, and preferably in the range of 1 hour to 48 hours.
[製造法3]
前記式(1)で表される本発明化合物は、例えば、式(2)で表される化合物[以下、化合物(2)と略称する。]と式(51)で表される化合物[以下、化合物(51)と略称する。]とを反応させることにより製造することができる。
[Production Method 3]
The compound of the present invention represented by the formula (1) is, for example, a compound represented by the formula (2) [hereinafter abbreviated as compound (2). ] And a compound represented by formula (51) [hereinafter abbreviated as compound (51). ] Can be made to react.
(式中、R1、R2、Y2、Y3、Y4、G及びnは前記と同じ意味を表す。)
化合物(1)は、化合物(2)と化合物(51)とを、溶媒中又は無溶媒で、場合によっては塩基存在下反応させることで製造することができる。
(Wherein R 1 , R 2 , Y 2 , Y 3, Y 4, G and n represent the same meaning as described above.)
Compound (1) can be produced by reacting compound (2) and compound (51) in a solvent or without a solvent, optionally in the presence of a base.
用いる化合物(51)の当量としては、化合物(2)1当量に対して、0.5から50当量の範囲で用いることができ、1から20当量の範囲が好ましい。 The equivalent of the compound (51) to be used can be used in the range of 0.5 to 50 equivalents relative to 1 equivalent of the compound (2), and preferably in the range of 1 to 20 equivalents.
化合物(51)の或るものは公知化合物であり、一部は市販品として入手できる。 Some of the compounds (51) are known compounds, and some of them are commercially available.
溶媒を用いる場合、用いられる溶媒としては反応に不活性であればよく、例えば水、メタノール、エタノール等の低級アルコール類、ジエチルエーテル、テトラヒドロフラン、1,4−ジオキサン、1,2−ジメトキシエタン等のエーテル類、ベンゼン、クロロベンゼン、ブロモベンゼン、キシレン、トルエン等の芳香族炭化水素類、ペンタン、ヘキサン、シクロヘキサン等の脂肪族炭化水素類、ジクロロメタン、クロロホルム、1,2−ジクロロエタン等のハロゲン化炭化水素類、アセトニトリル、プロピオニトリル等のニトリル類、N,N−ジメチルホルムアミド、N,N−ジメチルアセトアミド、N−メチルピロリドン、N,N’−ジメチルイミダゾリジノン等のアミド類、ジメチルスルホキシド等のスルホキシド類、ピリジン、キノリン等の含窒素芳香族化合物類又はこれらの混合溶媒等が挙げられる。 When using a solvent, the solvent used may be inert to the reaction, such as water, lower alcohols such as methanol and ethanol, diethyl ether, tetrahydrofuran, 1,4-dioxane, 1,2-dimethoxyethane and the like. Ethers, aromatic hydrocarbons such as benzene, chlorobenzene, bromobenzene, xylene, and toluene, aliphatic hydrocarbons such as pentane, hexane, and cyclohexane, and halogenated hydrocarbons such as dichloromethane, chloroform, and 1,2-dichloroethane Nitriles such as acetonitrile and propionitrile, amides such as N, N-dimethylformamide, N, N-dimethylacetamide, N-methylpyrrolidone and N, N′-dimethylimidazolidinone, and sulfoxides such as dimethyl sulfoxide , Pyridine, quino Nitrogen-containing aromatic compounds or a mixture of these solvents, such as emissions and the like.
該反応は塩基の存在下に行うことができる。用いることのできる塩基としては、例えばピリジン、2,6−ルチジン、4−ジメチルアミノピリジン、トリエチルアミン、ジイソプロピルエチルアミン、トリブチルアミン、4-(ジメチルアミノ)ピリジン、1,4−ジアザビシクロ[2.2.2]オクタン(DABCO)、1,8−ジアザビシクロ[5.4.0]−7−ウンデセン(DBU)又は1,5−ジアザビシクロ[4.3.0]−5−ノネン(DBN)等の有機塩基類、水酸化ナトリウム、水酸化カリウム、水素化ナトリウム、炭酸水素ナトリウム、炭酸カリウム、炭酸セシウム等の無機塩基類が挙げられる。塩基の当量としては、化合物(2)に対して0.1から100当量の範囲で用いることができ、1から20当量の範囲が好ましい。 The reaction can be performed in the presence of a base. Examples of the base that can be used include pyridine, 2,6-lutidine, 4-dimethylaminopyridine, triethylamine, diisopropylethylamine, tributylamine, 4- (dimethylamino) pyridine, 1,4-diazabicyclo [2.2.2. ] Organic bases such as octane (DABCO), 1,8-diazabicyclo [5.4.0] -7-undecene (DBU) or 1,5-diazabicyclo [4.3.0] -5-nonene (DBN) And inorganic bases such as sodium hydroxide, potassium hydroxide, sodium hydride, sodium hydrogen carbonate, potassium carbonate, cesium carbonate and the like. The equivalent of the base can be used in the range of 0.1 to 100 equivalents relative to the compound (2), and preferably in the range of 1 to 20 equivalents.
反応温度は、−80℃から反応混合物の還流温度まで任意の温度で設定することができ、0℃から反応混合物の還流温度の範囲が好ましい。 The reaction temperature can be set at any temperature from −80 ° C. to the reflux temperature of the reaction mixture, and the range of 0 ° C. to the reflux temperature of the reaction mixture is preferred.
反応時間は、反応基質の濃度、反応温度によって変化するが、通常5分から100時間の範囲で任意に設定でき、1時間から48時間の範囲が好ましい。 The reaction time varies depending on the concentration of the reaction substrate and the reaction temperature, but can usually be arbitrarily set in the range of 5 minutes to 100 hours, and preferably in the range of 1 hour to 48 hours.
[製造法4]
前記式(1)で表される本発明化合物は、例えば化合物(3)と化合物(51)とを反応させることにより製造することができる。
[Production Method 4]
The compound of the present invention represented by the formula (1) can be produced, for example, by reacting the compound (3) with the compound (51).
(式中、R1、R2、Y2、Y3、Y4、G及びnは前記と同じ意味を表す。)
化合物(1)は、化合物(3)と化合物(51)とを、溶媒中又は無溶媒で、場合によっては塩基及び縮合剤の存在下反応させることで製造することができる。
(Wherein R 1 , R 2 , Y 2 , Y 3, Y 4, G and n represent the same meaning as described above.)
Compound (1) can be produced by reacting compound (3) and compound (51) in a solvent or without a solvent, optionally in the presence of a base and a condensing agent.
用いる化合物(51)の当量としては、化合物(3)1当量に対して、0.5〜50当量で用いることができ、1〜20当量が好ましい。化合物(51)の或るものは公知化合物であり、一部は市販品として入手できる。 As an equivalent of the compound (51) to be used, it can be used by 0.5-50 equivalent with respect to 1 equivalent of compound (3), and 1-20 equivalent is preferable. Some of the compounds (51) are known compounds, and some of them are commercially available.
溶媒を用いる場合、用いられる溶媒としては反応に不活性であればよく、例えばメタノール、エタノール等の低級アルコール類、ジエチルエーテル、テトラヒドロフラン、1,4−ジオキサン、1,2−ジメトキシエタン等のエーテル類、ベンゼン、クロロベンゼン、ブロモベンゼン、キシレン、トルエン等の芳香族炭化水素類、ペンタン、ヘキサン、シクロヘキサン等の脂肪族炭化水素類、ジクロロメタン、クロロホルム、1,2−ジクロロエタン等のハロゲン化炭化水素類、アセトニトリル、プロピオニトリル等のニトリル類、N,N−ジメチルホルムアミド、N,N−ジメチルアセトアミド、N−メチルピロリドン、N,N’−ジメチルイミダゾリジノン等のアミド類、ジメチルスルホキシド等のスルホキシド類又はこれらの混合溶媒等が挙げられる。 When a solvent is used, the solvent used may be inert to the reaction. For example, lower alcohols such as methanol and ethanol, ethers such as diethyl ether, tetrahydrofuran, 1,4-dioxane, and 1,2-dimethoxyethane , Aromatic hydrocarbons such as benzene, chlorobenzene, bromobenzene, xylene and toluene, aliphatic hydrocarbons such as pentane, hexane and cyclohexane, halogenated hydrocarbons such as dichloromethane, chloroform and 1,2-dichloroethane, acetonitrile , Nitriles such as propionitrile, amides such as N, N-dimethylformamide, N, N-dimethylacetamide, N-methylpyrrolidone, N, N′-dimethylimidazolidinone, sulfoxides such as dimethyl sulfoxide or the like Mixed solvent And the like.
塩基及び縮合剤を用いる場合、用いることのできる塩基としては、例えばピリジン、2,6−ルチジン、4−ジメチルアミノピリジン、トリエチルアミン、ジイソプロピルエチルアミン、トリブチルアミン、4-(ジメチルアミノ)ピリジン、1,4−ジアザビシクロ[2.2.2]オクタン(DABCO)、1,8−ジアザビシクロ[5.4.0]−7−ウンデセン(DBU)又は1,5−ジアザビシクロ[4.3.0]−5−ノネン(DBN)等の有機塩基類、水酸化ナトリウム、水酸化カリウム、水素化ナトリウム、炭酸水素ナトリウム、炭酸カリウム、炭酸セシウム等の無機塩基類が挙げられる。塩基の当量としては、化合物(3)1当量に対して0.1〜100当量で用いることができ、1〜20当量が好ましい。 When a base and a condensing agent are used, examples of bases that can be used include pyridine, 2,6-lutidine, 4-dimethylaminopyridine, triethylamine, diisopropylethylamine, tributylamine, 4- (dimethylamino) pyridine, 1,4 -Diazabicyclo [2.2.2] octane (DABCO), 1,8-diazabicyclo [5.4.0] -7-undecene (DBU) or 1,5-diazabicyclo [4.3.0] -5-nonene Organic bases such as (DBN), and inorganic bases such as sodium hydroxide, potassium hydroxide, sodium hydride, sodium hydrogen carbonate, potassium carbonate, cesium carbonate and the like. As an equivalent of a base, it can be used in 0.1-100 equivalent with respect to 1 equivalent of compound (3), and 1-20 equivalent is preferable.
用いることのできる縮合剤としては、例えば1−エチル−(3−ジメチルアミノプロピル)カルボジイミド、4−(4,6−ジメトキシ−1,3,5−トリアンジン−2−イル)−4−メチルモルホリニウムクロリド、無水酢酸、ピバル酸無水物又は2−メチル−6−ニトロ安息香酸無水物などが挙げられる。縮合剤の当量としては、化合物(3)1当量に対して0.1〜100当量で用いることができ、1〜20当量が好ましい。 Examples of the condensing agent that can be used include 1-ethyl- (3-dimethylaminopropyl) carbodiimide, 4- (4,6-dimethoxy-1,3,5-triangin-2-yl) -4-methylmorpholine. Examples thereof include nium chloride, acetic anhydride, pivalic anhydride, and 2-methyl-6-nitrobenzoic anhydride. As an equivalent of a condensing agent, it can be used by 0.1-100 equivalent with respect to 1 equivalent of compound (3), and 1-20 equivalent is preferable.
反応温度は、−80℃から反応混合物の還流温度まで任意の温度で設定することができ、0℃から反応混合物の還流温度の範囲が好ましい。 The reaction temperature can be set at any temperature from −80 ° C. to the reflux temperature of the reaction mixture, and the range of 0 ° C. to the reflux temperature of the reaction mixture is preferred.
反応時間は、反応基質の濃度、反応温度によって変化するが、通常5分〜100時間の範囲で任意に設定でき、1〜48時間が好ましい。 The reaction time varies depending on the concentration of the reaction substrate and the reaction temperature, but can usually be arbitrarily set in the range of 5 minutes to 100 hours, and preferably 1 to 48 hours.
製造法3に用いられる化合物(2)のうち、nが1又は2の整数である式(2−A)で表される化合物[以下、化合物(2−A)と略称する。]、並びに製造法4に用いられる化合物(3)のうち、nが1又は2の整数である化合物(3−A)及びnが0の整数である化合物(3−B)は、下記の反応式で示される製造ルートにしたがって製造することができる。 Of the compounds (2) used in production method 3, a compound represented by formula (2-A) wherein n is an integer of 1 or 2 [hereinafter abbreviated as compound (2-A). And compound (3-A) in which n is an integer of 1 or 2 and compound (3-B) in which n is an integer of 0 among compounds (3) used in production method 4 are the following reactions: It can manufacture according to the manufacturing route shown by a formula.
[反応式1] [Reaction Formula 1]
(式中、R1、Y2、Y3、Y4及びmは前記と同じ意味を表し、L1は塩素原子、臭素原子、ヨウ素原子等のハロゲン原子を表し、RaはC1〜C6アルキルを表す。)
[製造工程1]
式(6−1)で表される化合物[以下、化合物(6−1)と略称する。]を文献既知の公知の方法、例えば、国際公開第2013/018899号等に記載の方法に準じて、ハロゲン化剤と反応させることにより、式(5−1)で表される化合物[以下、化合物(5−1)と略称する。]を製造することができる。化合物(6−1)の或るものは公知化合物であり、一部は市販品として入手できる。
(Wherein R 1 , Y 2, Y 3, Y 4 and m represent the same meaning as described above, L 1 represents a halogen atom such as a chlorine atom, a bromine atom or an iodine atom, and R a represents C 1 to C 6 alkyl. Represents.)
[Manufacturing process 1]
Compound represented by formula (6-1) [hereinafter abbreviated as compound (6-1) ] In accordance with a known method known in the literature, for example, the method described in International Publication No. 2013/018899, etc., to give a compound represented by the formula (5-1) [hereinafter, Abbreviated as compound (5-1). Can be manufactured. Some of the compounds (6-1) are known compounds, and some of them are commercially available.
[製造工程2]
化合物(5−1)を文献既知の公知の方法、例えば、オーガニック・レターズ(Organic Letters)2007年,9巻,3687頁等に記載の方法に準じて、式(52)で表される化合物[以下、化合物(52)と略称する。]と反応させることにより、式(4−1)で表される化合物[以下、化合物(4−1)と略称する。]を製造することができる。化合物(52)の或るものは公知化合物であり、一部は市販品として入手できる。
[Manufacturing process 2]
Compound (5-1) is represented by formula (52) according to a known method known in the literature, for example, the method described in Organic Letters 2007, Vol. 9, page 3687, etc. [ Hereafter, it abbreviates as a compound (52). ] Is reacted with a compound represented by formula (4-1) [hereinafter abbreviated as compound (4-1). Can be manufactured. Some of the compounds (52) are known compounds, and some of them are commercially available.
[製造工程1−2]
化合物(4−1)は、公知の方法、例えばオーガニック・レターズ(Organic Letters)2014年,16巻,2978頁等に記載のに準じた方法により、化合物(6−1)と化合物(52)とをハロゲン化剤存在下で反応させることにより、化合物(6−1)より1工程で製造することができる。
[Manufacturing process 1-2]
Compound (4-1) can be obtained by a known method, for example, a method according to Organic Letters 2014, Vol. 16, page 2978, etc. Can be produced in one step from compound (6-1) by reacting in the presence of a halogenating agent.
[製造工程3]
化合物(4−1)を文献既知の公知の方法、例えば、バイオオーガニック・アンド・メディシナル・ケミストリー・レターズ(Bioorganic & Medicinal Chemistry Letters)2012年、22巻、1870頁等に記載の方法に準じて、加水分解反応を行うことにより、式(3−B)で表される化合物[以下、化合物(3−B)と略称する。]を製造することができる。
[Manufacturing process 3]
According to a known method known in the literature for compound (4-1), for example, the method described in Bioorganic & Medicinal Chemistry Letters 2012, Vol. 22, p. 1870, etc. By performing a hydrolysis reaction, the compound represented by the formula (3-B) [hereinafter abbreviated as compound (3-B). Can be manufactured.
[製造工程4]
化合物(3−B)を製造法1に記載の方法に準じて、酸化剤と反応させることにより、式(3−A)で表される化合物[以下、化合物(3−A)と略称する。]を製造することができる。
[Manufacturing process 4]
The compound (3-B) is reacted with an oxidant according to the method described in Production Method 1 to give a compound represented by the formula (3-A) [hereinafter abbreviated as compound (3-A). Can be manufactured.
[製造工程5]
化合物(3−A)を文献既知の公知の方法、例えば、国際公開第2011/041713号等に記載の方法に準じて、ハロゲン化剤と反応させることにより、化合物(2−A)を製造することができる。
[Manufacturing process 5]
Compound (2-A) is produced by reacting compound (3-A) with a halogenating agent according to a known method known in the literature, for example, the method described in International Publication No. 2011/041713. be able to.
製造法3に用いられる化合物(2)のうち、nが0の整数である式(2−B)で表される化合物[以下、化合物(2−B)と略称する。]は、下記の反応式2で示される製造方法にしたがって製造することができる。 Among the compounds (2) used in production method 3, a compound represented by formula (2-B) wherein n is an integer of 0 [hereinafter abbreviated as compound (2-B). ] Can be produced according to the production method shown in Reaction Scheme 2 below.
[反応式2] [Reaction Formula 2]
(式中、R1、Y2、Y3及びY4は前記と同じ意味を表す。)
化合物(3−B)を反応式1の製造工程5に記載の方法に準じて、塩素化剤と反応させることにより、化合物(2−B)を製造することができる。
(In the formula, R 1 , Y 2, Y 3 and Y 4 represent the same meaning as described above.)
The compound (2-B) can be produced by reacting the compound (3-B) with a chlorinating agent according to the method described in Production Process 5 of Reaction Scheme 1.
反応式1に用いられる化合物(6−1)は、化合物(7−1)と化合物(53)とを反応させることにより製造することができる。 Compound (6-1) used in Reaction Scheme 1 can be produced by reacting compound (7-1) with compound (53).
[反応式3] [Reaction Formula 3]
(式中、R1、Y2、Y3、Y4、X1及びRaは前記と同じ意味を表す。)
化合物(7−1)を公知の方法、例えば国際公開第2013/087792号、国際公開第2012/096929号等に記載の方法に準じて、化合物(53)と反応させることにより、化合物(6−1)を製造することができる。
(In the formula, R 1 , Y 2, Y 3, Y 4, X 1 and R a represent the same meaning as described above.)
The compound (7-1) is reacted with the compound (53) according to a known method, for example, the method described in International Publication No. 2013/087772, International Publication No. 2012/096929, etc., to give the compound (6- 1) can be manufactured.
反応式1に用いられる化合物(3−B)は、下記の反応式で示される製造ルートにしたがって製造することができる。 Compound (3-B) used in Reaction Scheme 1 can be produced according to the production route represented by the following reaction formula.
[反応式4] [Reaction Formula 4]
(式中、R1、Y2、Y3、Y4、X1及びRaは前記と同じ意味を表す。)
[製造工程1]
化合物(6−1)を公知の方法、例えば、オーガニック・プロセス・リサーチ・アンド・ディベロップメント(Organic Process Research & Development)2008年,12巻,1060頁等に記載の方法に準じてチオシアン酸ナトリウム、チオンシアン酸カリウム又は、チオシアン酸アンモニウムと反応させることにより、化合物(8−1)を製造することができる。
(In the formula, R 1 , Y 2, Y 3, Y 4, X 1 and R a represent the same meaning as described above.)
[Manufacturing process 1]
Compound (6-1) is prepared by a known method, for example, sodium thiocyanate according to the method described in Organic Process Research & Development 2008, Vol. 12, page 1060, etc. Compound (8-1) can be produced by reacting with potassium thionocyanate or ammonium thiocyanate.
[製造工程2]
化合物(8−1)を公知の方法、例えば、ペスト・マネジメント・サイエンス(Pest Management Science)2002年,58巻,1175頁等に記載の方法に準じて化合物(54)と反応させることにより、化合物(3−B)を製造することができる。
[Manufacturing process 2]
Compound (8-1) is reacted with Compound (54) according to a known method, for example, the method described in Pest Management Science 2002, 58, 1175, etc. (3-B) can be manufactured.
製造法1〜4、並びに反応式1〜4の反応において、反応終了後の反応混合物は、直接濃縮又は有機溶媒に溶解し、水洗後濃縮又は氷水に投入、有機溶媒抽出後濃縮といった通常の後処理を行ない、目的の本発明化合物を得ることができる。また、精製の必要が生じたときには、再結晶、カラムクロマトグラフ、薄層クロマトグラフ、液体クロマトグラフ分取等の任意の精製方法によって分離、精製することができる。 In the production methods 1 to 4 and the reactions of reaction formulas 1 to 4, the reaction mixture after completion of the reaction is directly concentrated or dissolved in an organic solvent, washed and then concentrated in water or poured into ice water, and concentrated after extraction with an organic solvent. By performing the treatment, the target compound of the present invention can be obtained. Moreover, when the necessity for purification arises, it can be separated and purified by any purification method such as recrystallization, column chromatograph, thin layer chromatograph, liquid chromatographic fractionation and the like.
式(1)で表される本発明化合物の製造中間体の具体的な例を、第3表及び第4表に示す。 Specific examples of the production intermediate of the compound of the present invention represented by the formula (1) are shown in Tables 3 and 4.
[第3表] [Table 3]
―――――――――――― ―――――――――――― ――――――――――――
n Y2 Y3 Y4 Z1 n Y2 Y3 Y4 Z1 n Y2 Y3 Y4 Z1
―――――――――――― ―――――――――――― ――――――――――――
0 CF3 H H OEt 0 CF3 H H OH 0 CF3 H H Cl
1 CF3 H H OEt 1 CF3 H H OH 1 CF3 H H Cl
2 CF3 H H OEt 2 CF3 H H OH 2 CF3 H H Cl
0 I H H OEt 0 I H H OH 0 I H H Cl
1 I H H OEt 1 I H H OH 1 I H H Cl
2 I H H OEt 2 I H H OH 2 I H H Cl
0 Br H H OEt 0 Br H H OH 0 Br H H Cl
1 Br H H OEt 1 Br H H OH 1 Br H H Cl
2 Br H H OEt 2 Br H H OH 2 Br H H Cl
0 Cl H H OEt 0 Cl H H OH 0 Cl H H Cl
1 Cl H H OEt 1 Cl H H OH 1 Cl H H Cl
2 Cl H H OEt 2 Cl H H OH 2 Cl H H Cl
0 c-Pr H H OEt 0 c-Pr H H OH 0 c-Pr H H Cl
1 c-Pr H H OEt 1 c-Pr H H OH 1 c-Pr H H Cl
2 c-Pr H H OEt 2 c-Pr H H OH 2 c-Pr H H Cl
0 NH2 H H OEt 0 NH2 H H OH 0 NH2 H H Cl
1 NH2 H H OEt 1 NH2 H H OH 1 NH2 H H Cl
2 NH2 H H OEt 2 NH2 H H OH 2 NH2 H H Cl
0 H CF3 H OEt 0 H CF3 H OH 0 H CF3 H Cl
1 H CF3 H OEt 1 H CF3 H OH 1 H CF3 H Cl
2 H CF3 H OEt 2 H CF3 H OH 2 H CF3 H Cl
0 H I H OEt 0 H I H OH 0 H I H Cl
1 H I H OEt 1 H I H OH 1 H I H Cl
2 H I H OEt 2 H I H OH 2 H I H Cl
0 H Br H OEt 0 H Br H OH 0 H Br H Cl
1 H Br H OEt 1 H Br H OH 1 H Br H Cl
2 H Br H OEt 2 H Br H OH 2 H Br H Cl
0 H Cl H OEt 0 H Cl H OH 0 H Cl H Cl
1 H Cl H OEt 1 H Cl H OH 1 H Cl H Cl
2 H Cl H OEt 2 H Cl H OH 2 H Cl H Cl
0 H c-Pr H OEt 0 H c-Pr H OH 0 H c-Pr H Cl
1 H c-Pr H OEt 1 H c-Pr H OH 1 H c-Pr H Cl
2 H c-Pr H OEt 2 H c-Pr H OH 2 H c-Pr H Cl
0 H NH2 H OEt 0 H NH2 H OH 0 H NH2 H Cl
1 H NH2 H OEt 1 H NH2 H OH 1 H NH2 H Cl
2 H NH2 H OEt 2 H NH2 H OH 2 H NH2 H Cl
0 H NHAc H OEt 0 H NHAc H OH 0 H NHAc H Cl
1 H NHAc H OEt 1 H NHAc H OH 1 H NHAc H Cl
2 H NHAc H OEt 2 H NHAc H OH 2 H NHAc H Cl
0 H NO2 H OEt 0 H NO2 H OH 0 H NO2 H Cl
1 H NO2 H OEt 1 H NO2 H OH 1 H NO2 H Cl
2 H NO2 H OEt 2 H NO2 H OH 2 H NO2 H Cl
0 H C(O)OH H OEt 0 H C(O)OH H OH 0 H C(O)OH H Cl
1 H C(O)OH H OEt 1 H C(O)OH H OH 1 H C(O)OH H Cl
2 H C(O)OH H OEt 2 H C(O)OH H OH 2 H C(O)OH H Cl
0 H C(O)OEt H OEt 0 H C(O)OEt H OH 0 H C(O)OEt H Cl
1 H C(O)OEt H OEt 1 H C(O)OEt H OH 1 H C(O)OEt H Cl
2 H C(O)OEt H OEt 2 H C(O)OEt H OH 2 H C(O)OEt H Cl
0 H CN H OEt 0 H CN H OH 0 H CN H Cl
1 H CN H OEt 1 H CN H OH 1 H CN H Cl
2 H CN H OEt 2 H CN H OH 2 H CN H Cl
0 H H NH2 OEt 0 H H NH2 OH 0 H H NH2 Cl
1 H H NH2 OEt 1 H H NH2 OH 1 H H NH2 Cl
2 H H NH2 OEt 2 H H NH2 OH 2 H H NH2 Cl
0 H H H OEt 0 H H H OH 0 H H H Cl
1 H H H OEt 1 H H H OH 1 H H H Cl
2 H H H OEt 2 H H H OH 2 H H H Cl
―――――――――――― ―――――――――――― ―――――――――――
[第4表]
―――――――――――― ―――――――――――― ――――――――――――――
n Y2 Y3 Y4 Z1 n Y2 Y3 Y4 Z1 n Y2 Y3 Y4 Z1
―――――――――――― ―――――――――――― ――――――――――――――
0 CF 3 HH OEt 0 CF 3 HH OH 0 CF 3 HH Cl
1 CF 3 HH OEt 1 CF 3 HH OH 1 CF 3 HH Cl
2 CF 3 HH OEt 2 CF 3 HH OH 2 CF 3 HH Cl
0 IHH OEt 0 IHH OH 0 IHH Cl
1 IHH OEt 1 IHH OH 1 IHH Cl
2 IHH OEt 2 IHH OH 2 IHH Cl
0 Br HH OEt 0 Br HH OH 0 Br HH Cl
1 Br HH OEt 1 Br HH OH 1 Br HH Cl
2 Br HH OEt 2 Br HH OH 2 Br HH Cl
0 Cl HH OEt 0 Cl HH OH 0 Cl HH Cl
1 Cl HH OEt 1 Cl HH OH 1 Cl HH Cl
2 Cl HH OEt 2 Cl HH OH 2 Cl HH Cl
0 c-Pr HH OEt 0 c-Pr HH OH 0 c-Pr HH Cl
1 c-Pr HH OEt 1 c-Pr HH OH 1 c-Pr HH Cl
2 c-Pr HH OEt 2 c-Pr HH OH 2 c-Pr HH Cl
0 NH 2 HH OEt 0 NH 2 HH OH 0 NH 2 HH Cl
1 NH 2 HH OEt 1 NH 2 HH OH 1 NH 2 HH Cl
2 NH 2 HH OEt 2 NH 2 HH OH 2 NH 2 HH Cl
0 H CF 3 H OEt 0 H CF 3 H OH 0 H CF 3 H Cl
1 H CF 3 H OEt 1 H CF 3 H OH 1 H CF 3 H Cl
2 H CF 3 H OEt 2 H CF 3 H OH 2 H CF 3 H Cl
0 HIH OEt 0 HIH OH 0 HIH Cl
1 HIH OEt 1 HIH OH 1 HIH Cl
2 HIH OEt 2 HIH OH 2 HIH Cl
0 H Br H OEt 0 H Br H OH 0 H Br H Cl
1 H Br H OEt 1 H Br H OH 1 H Br H Cl
2 H Br H OEt 2 H Br H OH 2 H Br H Cl
0 H Cl H OEt 0 H Cl H OH 0 H Cl H Cl
1 H Cl H OEt 1 H Cl H OH 1 H Cl H Cl
2 H Cl H OEt 2 H Cl H OH 2 H Cl H Cl
0 H c-Pr H OEt 0 H c-Pr H OH 0 H c-Pr H Cl
1 H c-Pr H OEt 1 H c-Pr H OH 1 H c-Pr H Cl
2 H c-Pr H OEt 2 H c-Pr H OH 2 H c-Pr H Cl
0 H NH 2 H OEt 0 H NH 2 H OH 0 H NH 2 H Cl
1 H NH 2 H OEt 1 H NH 2 H OH 1 H NH 2 H Cl
2 H NH 2 H OEt 2 H NH 2 H OH 2 H NH 2 H Cl
0 H NHAc H OEt 0 H NHAc H OH 0 H NHAc H Cl
1 H NHAc H OEt 1 H NHAc H OH 1 H NHAc H Cl
2 H NHAc H OEt 2 H NHAc H OH 2 H NHAc H Cl
0 H NO 2 H OEt 0 H NO 2 H OH 0 H NO 2 H Cl
1 H NO 2 H OEt 1 H NO 2 H OH 1 H NO 2 H Cl
2 H NO 2 H OEt 2 H NO 2 H OH 2 H NO 2 H Cl
0 HC (O) OH H OEt 0 HC (O) OH H OH 0 HC (O) OH H Cl
1 HC (O) OH H OEt 1 HC (O) OH H OH 1 HC (O) OH H Cl
2 HC (O) OH H OEt 2 HC (O) OH H OH 2 HC (O) OH H Cl
0 HC (O) OEt H OEt 0 HC (O) OEt H OH 0 HC (O) OEt H Cl
1 HC (O) OEt H OEt 1 HC (O) OEt H OH 1 HC (O) OEt H Cl
2 HC (O) OEt H OEt 2 HC (O) OEt H OH 2 HC (O) OEt H Cl
0 H CN H OEt 0 H CN H OH 0 H CN H Cl
1 H CN H OEt 1 H CN H OH 1 H CN H Cl
2 H CN H OEt 2 H CN H OH 2 H CN H Cl
0 HH NH 2 OEt 0 HH NH 2 OH 0 HH NH 2 Cl
1 HH NH 2 OEt 1 HH NH 2 OH 1 HH NH 2 Cl
2 HH NH 2 OEt 2 HH NH 2 OH 2 HH NH 2 Cl
0 HHH OEt 0 HHH OH 0 HHH Cl
1 HHH OEt 1 HHH OH 1 HHH Cl
2 HHH OEt 2 HHH OH 2 HHH Cl
―――――――――――― ―――――――――――― ―――――――――――
[Table 4]
――――――――――――――――― ―――――――――――――――――
Y2 Y3 Y4 Z1 Z2 Y2 Y3 Y4 Z1 Z2
――――――――――――――――― ―――――――――――――――――
CF3 H H OEt H CF3 H H OEt Br
I H H OEt H I H H OEt Br
Br H H OEt H Br H H OEt Br
Cl H H OEt H Cl H H OEt Br
c-Pr H H OEt H c-Pr H H OEt Br
NH2 H H OEt H NH2 H H OEt Br
H CF3 H OEt H H CF3 H OEt Br
H I H OEt H H I H OEt Br
H Br H OEt H H Br H OEt Br
H Cl H OEt H H Cl H OEt Br
H c-Pr H OEt H H c-Pr H OEt Br
H NH2 H OEt H H NH2 H OEt Br
H NHAc H OEt H H NHAc H OEt Br
H NO2 H OEt H H NO2 H OEt Br
H C(O)OH H OEt H H C(O)OH H OEt Br
H C(O)OEt H OEt H H C(O)OEt H OEt Br
H CN H OEt H H CN H OEt Br
H H NH2 OEt H H H NH2 OEt Br
H H H OEt H H H H OEt Br
CF3 H H OH H CF3 H H OH Br
I H H OH H I H H OH Br
Br H H OH H Br H H OH Br
Cl H H OH H Cl H H OH Br
c-Pr H H OH H c-Pr H H OH Br
NH2 H H OH H NH2 H H OH Br
H CF3 H OH H H CF3 H OH Br
H I H OH H H I H OH Br
H Br H OH H H Br H OH Br
H Cl H OH H H Cl H OH Br
H c-Pr H OH H H c-Pr H OH Br
H NH2 H OH H H NH2 H OH Br
H NHAc H OH H H NHAc H OH Br
H NO2 H OH H H NO2 H OH Br
H C(O)OH H OH H H C(O)OH H OH Br
H C(O)OEt H OH H H C(O)OEt H OH Br
H CN H OH H H CN H OH Br
H H NH2 OH H H H NH2 OH Br
H H H OH H H H H OH Br
CF3 H H Cl H CF3 H H Cl Br
I H H Cl H I H H Cl Br
Br H H Cl H Br H H Cl Br
Cl H H Cl H Cl H H Cl Br
c-Pr H H Cl H c-Pr H H Cl Br
NH2 H H Cl H NH2 H H Cl Br
H CF3 H Cl H H CF3 H Cl Br
H I H Cl H H I H Cl Br
H Br H Cl H H Br H Cl Br
H Cl H Cl H H Cl H Cl Br
H c-Pr H Cl H H c-Pr H Cl Br
H NH2 H Cl H H NH2 H Cl Br
H NHAc H Cl H H NHAc H Cl Br
H NO2 H Cl H H NO2 H Cl Br
H C(O)OH H Cl H H C(O)OH H Cl Br
H C(O)OEt H Cl H H C(O)OEt H Cl Br
H CN H Cl H H CN H Cl Br
H H NH2 Cl H H H NH2 Cl Br
H H H Cl H H H H Cl Br
CF3 H H OEt Cl CF3 H H OEt I
I H H OEt Cl I H H OEt I
Br H H OEt Cl Br H H OEt I
Cl H H OEt Cl Cl H H OEt I
c-Pr H H OEt Cl c-Pr H H OEt I
NH2 H H OEt Cl NH2 H H Oet I
H CF3 H OEt Cl H CF3 H OEt I
H I H OEt Cl H I H OEt I
H Br H OEt Cl H Br H OEt I
H Cl H OEt Cl H Cl H OEt I
H c-Pr H OEt Cl H c-Pr H OEt I
H NH2 H OEt Cl H NH2 H OEt I
H NHAc H OEt Cl H NHAc H OEt I
H NO2 H OEt Cl H NO2 H OEt I
H C(O)OH H OEt Cl H C(O)OH H OEt I
H C(O)OEt H OEt Cl H C(O)OEt H OEt I
H CN H OEt Cl H CN H OEt I
H H NH2 OEt Cl H H NH2 OEt I
H H H OEt Cl H H H OEt I
CF3 H H OH Cl CF3 H H OH I
I H H OH Cl I H H OH I
Br H H OH Cl Br H H OH I
Cl H H OH Cl Cl H H OH I
c-Pr H H OH Cl c-Pr H H OH I
NH2 H H OH Cl NH2 H H OH I
H CF3 H OH Cl H CF3 H OH I
H I H OH Cl H I H OH I
H Br H OH Cl H Br H OH I
H Cl H OH Cl H Cl H OH I
H c-Pr H OH Cl H c-Pr H OH I
H NH2 H OH Cl H NH2 H OH I
H NHAc H OH Cl H NHAc H OH I
H NO2 H OH Cl H NO2 H OH I
H C(O)OH H OH Cl H C(O)OH H OH I
H C(O)OEt H OH Cl H C(O)OEt H OH I
H CN H OH Cl H CN H OH I
H H NH2 OH Cl H H NH2 OH I
H H H OH Cl H H H OH I
CF3 H H Cl Cl CF3 H H Cl I
I H H Cl Cl I H H Cl I
Br H H Cl Cl Br H H Cl I
Cl H H Cl Cl Cl H H Cl I
c-Pr H H Cl Cl c-Pr H H Cl I
NH2 H H Cl Cl NH2 H H Cl I
H CF3 H Cl Cl H CF3 H Cl I
H I H Cl Cl H I H Cl I
H Br H Cl Cl H Br H Cl I
H Cl H Cl Cl H Cl H Cl I
H c-Pr H Cl Cl H c-Pr H Cl I
H NH2 H Cl Cl H NH2 H Cl I
H NHAc H Cl Cl H NHAc H Cl I
H NO2 H Cl Cl H NO2 H Cl I
H C(O)OH H Cl Cl H C(O)OH H Cl I
H C(O)OEt H Cl Cl H C(O)OEt H Cl I
H CN H Cl Cl H CN H Cl I
H H NH2 Cl Cl H H NH2 Cl I
H H H Cl Cl H H H Cl I
――――――――――――――――― ―――――――――――――――――
本発明における有害生物防除剤とは、農園芸分野又は畜産・衛生分野(家畜や愛玩動物としての哺乳動物又は鳥類に対する内部寄生虫・外部寄生虫や家庭用及び業務用の衛生害虫・不快害虫)等における有害な節足動物を対象とした害虫防除剤を意味する。また、本発明における農薬とは、農園芸分野における殺虫・殺ダニ剤、殺線虫剤、除草剤及び殺菌剤等を意味する。
――――――――――――――――― ―――――――――――――――――
Y2 Y3 Y4 Z1 Z2 Y2 Y3 Y4 Z1 Z2
――――――――――――――――― ―――――――――――――――――
CF 3 HH OEt H CF 3 HH OEt Br
IHH OEt HIHH OEt Br
Br HH OEt H Br HH OEt Br
Cl HH OEt H Cl HH OEt Br
c-Pr HH OEt H c-Pr HH OEt Br
NH 2 HH OEt H NH 2 HH OEt Br
H CF 3 H OEt HH CF 3 H OEt Br
HIH OEt HHIH OEt Br
H Br H OEt HH Br H OEt Br
H Cl H OEt HH Cl H OEt Br
H c-Pr H OEt HH c-Pr H OEt Br
H NH 2 H OEt HH NH 2 H OEt Br
H NHAc H OEt HH NHAc H OEt Br
H NO 2 H OEt HH NO 2 H OEt Br
HC (O) OH H OEt HHC (O) OH H OEt Br
HC (O) OEt H OEt HHC (O) OEt H OEt Br
H CN H OEt HH CN H OEt Br
HH NH 2 OEt HHH NH 2 OEt Br
HHH OEt HHHH OEt Br
CF 3 HH OH H CF 3 HH OH Br
IHH OH HIHH OH Br
Br HH OH H Br HH OH Br
Cl HH OH H Cl HH OH Br
c-Pr HH OH H c-Pr HH OH Br
NH 2 HH OH H NH 2 HH OH Br
H CF 3 H OH HH CF 3 H OH Br
HIH OH HHIH OH Br
H Br H OH HH Br H OH Br
H Cl H OH HH Cl H OH Br
H c-Pr H OH HH c-Pr H OH Br
H NH 2 H OH HH NH 2 H OH Br
H NHAc H OH HH NHAc H OH Br
H NO 2 H OH HH NO 2 H OH Br
HC (O) OH H OH HHC (O) OH H OH Br
HC (O) OEt H OH HHC (O) OEt H OH Br
H CN H OH HH CN H OH Br
HH NH 2 OH HHH NH 2 OH Br
HHH OH HHHH OH Br
CF 3 HH Cl H CF 3 HH Cl Br
IHH Cl HIHH Cl Br
Br HH Cl H Br HH Cl Br
Cl HH Cl H Cl HH Cl Br
c-Pr HH Cl H c-Pr HH Cl Br
NH 2 HH Cl H NH 2 HH Cl Br
H CF 3 H Cl HH CF 3 H Cl Br
HIH Cl HHIH Cl Br
H Br H Cl HH Br H Cl Br
H Cl H Cl HH Cl H Cl Br
H c-Pr H Cl HH c-Pr H Cl Br
H NH 2 H Cl HH NH 2 H Cl Br
H NHAc H Cl HH NHAc H Cl Br
H NO 2 H Cl HH NO 2 H Cl Br
HC (O) OH H Cl HHC (O) OH H Cl Br
HC (O) OEt H Cl HHC (O) OEt H Cl Br
H CN H Cl HH CN H Cl Br
HH NH 2 Cl HHH NH 2 Cl Br
HHH Cl HHHH Cl Br
CF 3 HH OEt Cl CF 3 HH OEt I
IHH OEt Cl IHH OEt I
Br HH OEt Cl Br HH OEt I
Cl HH OEt Cl Cl HH OEt I
c-Pr HH OEt Cl c-Pr HH OEt I
NH 2 HH OEt Cl NH 2 HH Oet I
H CF 3 H OEt Cl H CF 3 H OEt I
HIH OEt Cl HIH OEt I
H Br H OEt Cl H Br H OEt I
H Cl H OEt Cl H Cl H OEt I
H c-Pr H OEt Cl H c-Pr H OEt I
H NH 2 H OEt Cl H NH 2 H OEt I
H NHAc H OEt Cl H NHAc H OEt I
H NO 2 H OEt Cl H NO 2 H OEt I
HC (O) OH H OEt Cl HC (O) OH H OEt I
HC (O) OEt H OEt Cl HC (O) OEt H OEt I
H CN H OEt Cl H CN H OEt I
HH NH 2 OEt Cl HH NH 2 OEt I
HHH OEt Cl HHH OEt I
CF 3 HH OH Cl CF 3 HH OH I
IHH OH Cl IHH OH I
Br HH OH Cl Br HH OH I
Cl HH OH Cl Cl HH OH I
c-Pr HH OH Cl c-Pr HH OH I
NH 2 HH OH Cl NH 2 HH OH I
H CF 3 H OH Cl H CF 3 H OH I
HIH OH Cl HIH OH I
H Br H OH Cl H Br H OH I
H Cl H OH Cl H Cl H OH I
H c-Pr H OH Cl H c-Pr H OH I
H NH 2 H OH Cl H NH 2 H OH I
H NHAc H OH Cl H NHAc H OH I
H NO 2 H OH Cl H NO 2 H OH I
HC (O) OH H OH Cl HC (O) OH H OH I
HC (O) OEt H OH Cl HC (O) OEt H OH I
H CN H OH Cl H CN H OH I
HH NH 2 OH Cl HH NH 2 OH I
HHH OH Cl HHH OH I
CF 3 HH Cl Cl CF 3 HH Cl I
IHH Cl Cl IHH Cl I
Br HH Cl Cl Br HH Cl I
Cl HH Cl Cl Cl HH Cl I
c-Pr HH Cl Cl c-Pr HH Cl I
NH 2 HH Cl Cl NH 2 HH Cl I
H CF 3 H Cl Cl H CF 3 H Cl I
HIH Cl Cl HIH Cl I
H Br H Cl Cl H Br H Cl I
H Cl H Cl Cl H Cl H Cl I
H c-Pr H Cl Cl H c-Pr H Cl I
H NH 2 H Cl Cl H NH 2 H Cl I
H NHAc H Cl Cl H NHAc H Cl I
H NO 2 H Cl Cl H NO 2 H Cl I
HC (O) OH H Cl Cl HC (O) OH H Cl I
HC (O) OEt H Cl Cl HC (O) OEt H Cl I
H CN H Cl Cl H CN H Cl I
HH NH 2 Cl Cl HH NH 2 Cl I
HHH Cl Cl HHH Cl I
――――――――――――――――― ―――――――――――――――――
The pest control agent in the present invention is the field of agriculture and horticulture or the field of livestock and hygiene (internal parasites / external parasites for domestic animals and pets, hygiene pests and unpleasant pests for domestic and commercial use) It means a pest control agent for harmful arthropods such as The agrochemical in the present invention means an insecticide / acaricide, nematicide, herbicide, fungicide, etc. in the field of agriculture and horticulture.
本発明化合物を用いて防除しうる昆虫類、ダニ類、甲殻類、軟体動物及び線虫類としては、具体的には、例えば、以下の生物等が挙げられが、本発明はこれらのみに限定されるものではない。 Specific examples of insects, mites, crustaceans, molluscs and nematodes that can be controlled using the compounds of the present invention include the following organisms, but the present invention is not limited to these. Is not to be done.
チャノコカクモンハマキ(Adoxophyes honmai)、リンゴコカクモンハマキ(Adoxophyes orana fascita)、リンゴモンハマキ(Archips breviplicanus)、ミダレカクモンハマキ(Archips fuscocupreanus)、ナシヒメシンクイ(Grapholita molesta)、チャハマキ(Homona magnanima)、マメシンクイガ(Leguminivora glycinivorella)、マメヒメサヤムシガ(Matsumuraeses phaseoli)、トビハマキ(Pandemis heparana)、ナシチビガ(Bucculatrix pyrivorella)、モモハモグリガ(Lyonetia clerkella)、ギンモンハモグリガ(Lyonetia prunifoliella malinella)、チャノホソガ(Caloptilia theivora)、キンモンホソガ(Phyllonorycter ringoniella)、ミカンハモグリガ(Phyllocnistis citrella)、ネギコガ(Acrolepiopsis sapporensis)、ヤマノイモコガ(Acrolepiopsis suzukiella)、コナガ(Plutella xylostella)、カキノヘタムシガ(Stathmopoda masinissa)、イモキバガ(Helcystogramma triannulella)、ワタアカミムシ(Pectinophora gossypiella)、モモシンクイガ(Carposina sasakii)、コドリンガ(Cydla pomonella)、ニカメイガ(Chilo suppressalis)、コブノメイガ(Cnaphalocrocis medinalis)、モモノゴマダラノメイガ(Conogethes punctiferalis)、ワタヘリクロノメイガ(Diaphania indica)、シロイチモジマダラメイガ(Etiella zinckenella)、クワノメイガ(Glyphodes pyloalis)、ハイマダラノメイガ(Hellula undalis)、アワノメイガ(Ostrinia furnacalis)、アズキノメイガ(Ostrinia scapulalis)、ヨーロピアンコーンボーラー(Ostrinia nubilalis)、シバツトガ(Parapediasia teterrella)、イチモンジセセリ(Parnara guttata)、オオモンシロチョウ(Pieris brassicae)、モンシロチョウ(Pieris rapae crucivora)、ヨモギエダシャク(Ascotis selenaria)、ソイビーンルーパー(Pseudoplusia includens)、チャドクガ(Euproctis pseudoconspersa)、マイマイガ(Lymantria dispar)、ヒメシロモンドクガ(Orgyia thyellina)、アメリカシロヒトリ(Hyphantria cunea)、クワゴマダラヒトリ(Lemyra imparilis)、アケビコノハ(Adris tyrannus)、ナカジロシタバ(Aedia leucomelas)、タマナヤガ(Agrotis ipsilon)、カブラヤガ(Agrotis segetum)、タマナギンウワバ(Autographa nigrisigna)、ミツモンキンウワバ(Ctenoplusia agnata)、オオタバコガ(Helicoverpa armigera)、タバコガ(Helicoverpa assulta)、コットンボールワーム(Helicoverpa zea)、タバコバッドワーム(Heliothis virescens)、ヨトウガ(Mamestra brassicae)、アワヨトウ(Mythimna separata)、フタオビコヤガ(Naranga aenescens)、サザンアーミーワーム(Spodoptera eridania)、シロイチモジヨトウ(Spodoptera exigua)、フォールアーミーワーム(Spodoptera frugiperda)、コットンリーフワーム(Spodoptera littoralis)、ハスモンヨトウ(Spodoptera litura)、スジキリヨトウ(Spodoptera depravata)、イラクサギンウワバ(Trichoplusia ni)、グレープベリーモス(Endopiza viteana)、トマトホーンワーム(Manduca quinquemaculata)、タバコホーンワーム(Manduca sexta)等の鱗翅目昆虫。 Adoxophyes honmai, Adoxophyes orana fascita, Archips breviplicanus, Archips fuscocupreanus, Grapholita molesta, Chamani nan Leguminivora glycinivorella, Matsumuraeses phaseoli, Pandemis heparana, Bucculatrix pyrivorella, Lyonetia clerkella, Lyonetia prunifal malp ringoniella), citrus leafworm (Phyllocnistis citrella), green moth (Acrolepiopsis sapporensis), yamanoimokoga (Acrolepiopsis suzukiella), konaga (Plutella xylostella), oyster leaf moth (Stathmopoda masinissa), Hemogram (Helcystogramma triannulella), Cotton beetle (Pectinophora gossypiella), Peachinosigaiga (Carposina sasakii), Codlinga (Cydla pomonella), Green moth (Chilo suppressalis), Cnaphalocrocis medinalis, Hemagogai indica), Etiella zinckenella, Glyphodes pyloalis, Hellula undalis, Ostrinia furnacalis, Austinia scapulalinia, biloba tris, European tribe Parapediasia teterrella, Parnara guttata, Pieris brassicae, Pieris rapae crucivora, Ascotis selenaria, Soybean looper (Pseudoplusia includens), Chadokuga (Euproctis pseudoconspersa), Japanese gypsy moth (Lymantria dispar), Japanese white squirrel (Orgyia thyellina), American white starfish (Hyphantria cunea), Kuwagomadara hitori (Lemyra imparilis), Akebiko ty ), Agrotis ipsilon, Agrotis segetum, Autographa nigrisigna, Ctenoplusia agnata, Helicoverpa armigera, Helicoverpa assultaH, coverball worm, coverball worm Bad worm (Heliothis virescens), Mamestra brassicae, Mythimna separata, Naranga aenescens, Southern army worm (Spodoptera eridania), Spodoptera exigua, Falla Mewworm (Spodoptera frugiperda), Cotton leaf worm (Spodoptera littoralis), Spodoptera litura, Spodoptera depravata, Nettle quail (Trichoplusia ni), Grapeberry moss (Endopiza viteana), Tomato horn Lepidopterous insects such as tobacco hornworm (Manduca sexta).
ヒラズハナアザミウマ(Frankliniella intonsa)、ミカンキイロアザミウマ(Frankliniella occidentalis)、クロトンアザミウマ(Heliothrips haemorrhoidalis)、チャノキイロアザミウマ(Scirtothrips dorsalis)、ミナミキイロアザミウマ(Thrips palmi)、ネギアザミウマ(Thrips tabaci)、カキクダアザミウマ(Ponticulothrips diospyrosi)等の総翅目昆虫。 Thrips thrips (Frankliniella intonsa), Thrips thrips (Frankliniella occidentalis), Black thrips (Trips thrips) Moths in total.
ブチヒゲカメムシ(Dolycoris baccarum)、ナガメ(Eurydema rugosum)、トゲシラホシカメムシ(Eysarcoris aeneus)、オオトゲシラホシカメムシ(Eysarcoris lewisi)、シラホシカメムシ(Eysarcoris ventralis)、ツヤアオカメムシ(Glaucias subpunctatus)、クサギカメムシ(Halyomorpha halys)、アオクサカメムシ(Nezara antennata)、ミナミアオカメムシ(Nezara viridula)、イチモンジカメムシ(Piezodorus hybneri)、チャバネアオカメムシ(Plautia crossota)、イネクロカメムシ(Scotinophora lurida)、ホソハリカメムシ(Cletus punctiger)、クモヘリカメムシ(Leptocorisa chinensis)、ホソヘリカメムシ(Riptortus clavatus)、アカヒメヘリカメムシ(Rhopalus maculatus)、カンシャコバネナガカメムシ(Cavelerius saccharivorus)、コバネヒョウタンナガカメムシ(Togo hemipterus)、アカホシカメムシ(Dysdercus cingulatus)、ツツジグンバイ(Stephanitis pyrioides)、クロトビカスミカメ(Halticus insularis)、ターニッシュドプラントバグ(Lygus lineolaris)、ナガムギカスミカメ(Stenodema sibiricum)、アカスジカスミカメ(Stenotus rubrovittatus)、イネホソミドリカスミカメ(Trigonotylus caelestialium)、フタテンヒメヨコバイ(Arboridia apicalis)、ミドリナガヨコバイ(Balclutha saltuella)、フタテンオオヨコバイ(Epiacanthus stramineus)、ポテトリーフホッパー(Empoasca fabae)、カキノヒメヨコバイ(Empoasca nipponica)、チャノミドリヒメヨコバイ(Empoasca onukii)、マメノミドリヒメヨコバイ(Empoasca sakaii)、ヒメフタテンヨコバイ(Macrosteles striifrons)、ツマグロヨコバイ(Nephotettix cincticeps)、コットンフリーホッパー(Psuedatomoscelis seriatus)、ヒメトビウンカ(Laodelphax striatella)、トビイロウンカ(Nilaparvata lugens)、セジロウンカ(Sogatella furcifera)、ミカンキジラミ(Diaphorina citri)、ナシキジラミ(Psylla pyrisuga)、ミカントゲコナジラミ(Aleurocanthus spiniferus)、シルバーリーフコナジラミ(Bemisia argentifolii)、タバココナジラミ(Bemisia tabaci)、ミカンコナジラミ(Dialeurodes citri)、オンシツコナジラミ(Trialeurodes vaporariorum)、ブドウネアブラムシ(Viteus vitifolii)、ワタアブラムシ(Aphis gossypii)、ユキヤナギアブラムシ(Aphis spiraecola)、モモアカアブラムシ(Myzus persicae)、コミカンアブラムシ(Toxoptera aurantii)、オオワラジカイガラムシ(Drosicha corpulenta)、イセリアカイガラムシ(Icerya purchasi)、ナスコナカイガラムシ(Phenacoccus solani)、ミカンコナカイガラムシ(Planococcus citri)、フジコナカイガラムシ(Planococcus kraunhiae)、クワコナカイガラムシ(Pseudococcus comstocki)、ツノロウムシ(Ceroplastes ceriferus)、ルビーロウムシ(Ceroplastes rubens)、アカマルカイガラムシ(Aonidiella aurantii)、ナシマルカイガラムシ(Comstockaspis perniciosa)、ティースケール(Fiorinia theae)、チャノマルカイガラムシ(Pseudaonidia paeoniae)、クワシロカイガラムシ(Pseudaulacaspis pentagona)、ウメシロカイガラムシ(Pseudaulacaspis prunicola)、マサキナガカイガラムシ(Unaspis euonymi)、ヤノネカイガラムシ(Unaspis yanonensis)、トコジラミ(Cimex lectularius)等の半翅目昆虫。 Spotted beetle (Dolycoris baccarum), sea turtle (Eurydema rugosum), bark beetle (Eysarcoris aeneus), bark beetle (Eysarcoris lewisi), bark beetle (Eysarcoris ventralis), subtilus , Nezara antennata, Nezara viridula, Piezodorus hybneri, Plautia crossota, Scotinophora lurida, tiger p Stink bug (Leptocorisa chinensis), Hoso-heli beetle (Riptortus clavatus), Red-headed beetle (Rhopalus maculatus), Cavelerius saccharivorus, Red-footed beetle (Togo hemipterus), Red-billed bug (Togo hemipterus) cus cingulatus, Stephanitis pyrioides, Black beetle turtle (Halticus insularis), Turned plant bug (Lygus lineolaris), Nugget beetle turtle (Stenodema sibiricum), Akazika damselfly (Stenotus rubrovittatus), Mine hawk beetle Trigonotylus caelestialium), Arboridia apicalis, Balclutha saltuella, Epiacanthus stramineus, Potato leaf hopper (Empoasca fabae), Empoasca nipp (Empoasca onukii), green bean leaf (Empoasca sakaii), yellow leaf butterfly (Macrosteles striifrons), black leafhopper (Nephotettix cincticeps), cotton free hopper (Psuedatomoscelis seriatus), Himetolluna x a), white planthopper (Nilaparvata lugens), white planthopper (Sogatella furcifera), citrus lice (Diaphorina citri), pear lice (Psylla pyrisuga), citrus white lice (Aleurocanthus spiniferus), silver leaf white genta (Boli gentia) Japanese whitefly (Dialeurodes citri), Whitefly (Trialeurodes vaporariorum), Grape aphid (Viteus vitifolii), Cotton aphid (Aphis gossypii), Snowy aphid (Aphis spiraecola), Peach aphid (Myzus persicae) , Drosicha corpulenta, Icerya purchasi, Phenacoccus solani, Planococcus citri, Planococcus citri, Planococci occus kraunhiae), stag beetle (Pseudococcus comstocki), horned beetle (Ceroplastes ceriferus), ruby weevil (Ceroplastes rubens), red beetle (Aonidiella aurantii), pear beetle (Comstockaspis perniciosa), tea scale (Comstockaspis perniciosa), scale Pseudaonidia paeoniae), Pseudaulacaspis pentagona, Pseudaulacaspis prunicola, Pepperula scales (Unaspis euonymi), Pteridae scales (Unaspis yanonensis), Insect lecti
ドウガネブイブイ(Anomala cuprea)、ヒメコガネ(Anomala rufocuprea)、コアオハナムグリ(Gametis jucunda)、ナガチャコガネ(Heptophylla picea)、マメコガネ(Popillia japonica)、コロラドポテトビートル(Leptinotarsa decemlineata)、マルクビクシコメツキ(Melanotus fortnumi)、カンシャクシコメツキ(Melanotus tamsuyensis)、タバコシバンムシ(Lasioderma serricorne)、ヒメヒラタケシキスイ(Epuraea domina)、インゲンテントウ(Epilachna varivestis)、ニジュウヤホシテントウ(Epilachna vigintioctopunctata)、チャイロコメノゴミムシダマシ(Tenebrio molitor)、コクヌストモドキ(Tribolium castaneum)、ゴマダラカミキリ(Anoplophora malasiaca)、マツノマダラカミキリ(Monochamus alternatus)、キボシカミキリ(Psacothea hilaris)、ブドウトラカミキリ(Xylotrechus pyrrhoderus)、アズキゾウムシ(Callosobruchus chinensis)、ウリハムシ(Aulacophora femoralis)、テンサイトビハムシ(Chaetocnema concinna)、サザンコーンルートワーム(Diabrotica undecimpunctata)、ウエスタンコーンルートワーム(Diabrotica virgifera)、ノーザンコーンルートワーム(Diabrotica barberi)、イネドロオイムシ(Oulema oryzae)、キスジノミハムシ(Phyllotreta striolata)、ナスナガスネトビハムシ(Psylliodes angusticollis)、モモチョッキリゾウムシ(Rhynchites heros)、アリモドキゾウムシ(Cylas formicarius)、ワタミゾウムシ(Anthonomus grandis)、イネゾウムシ(Echinocnemus squameus)、イモゾウムシ(Euscepes postfasciatus)、アルファルファタコゾウムシ(Hypera postica)、イネミズゾウムシ(Lissorhoptrus oryzophilus)、キンケクチブトゾウムシ(Otiorhynchus sulcatus)、グラナリーウィービル(Sitophilus granarius)、コクゾウムシ(Sitophilus zeamais)、シバオサゾウムシ(Sphenophorus venatus vestitus)、アオバアリガタハネカクシ(Paederus fuscipes)等の鞘翅目昆虫。 Douganebubui (Anomala cuprea), Japanese common squirrel (Anomala rufocuprea), Coreo hanamuri (Gametis jucunda), Nagachakogane (Heptophylla picea), Japanese beetle (Popillia japonica), Colorado potato beetle (Leptinotarsa decemlineata) White-footed beetle (Melanotus tamsuyensis), Tobacco beetle (Lasioderma serricorne), Himetaketakekisui (Epuraea domina), Common beetle (Epilachna varivestis), Nyungyahoshi-tento (Epilachna vigintioctopunctata), Tenebromometomu-to Tribolium castaneum, Anoplophora malasiaca, Monochamus alternatus, Psacothea hilaris, Xylotrechus pyrrhoderus, Azuki beetle (Callosobruchus chinensis), cucumber beetle (Aulacophora femoralis), beetle beetle (Chaetocnema concinna), southern corn root worm (Diabrotica undecimpunctata), western corn root worm (Diabrotica virgifera), northern corn root worm (Diabrotica barberi), rice plant oryzae, Phyllotreta striolata, Psylliodes angusticollis, Rhynchites heros, Cylas formicarius, Anthonomus grandis, cezo, ce postfasciatus), alfalfa weevil (Hypera postica), rice weevil (Lissorhoptrus oryzophilus), horned weevil (Otiorhynchus sulcatus), granary weevil (Sitophilus granarius) ), Sitophilus zeamais, Sphenophorus venatus vestitus, and Coleoptera (Paederus fuscipes).
ダイズサヤタマバエ(Asphondylia yushimai)、ムギアカタマバエ(Sitodiplosis mosellana)、ウリミバエ(Bactrocera cucurbitae)、ミカンコミバエ(Bactrocera dorsalis)、チチュウカイミバエ(Ceratitis capitata)、イネヒメハモグリバエ(Hydrellia griseola)、オウトウショウジョウバエ(Drosophila suzukii)、イネハモグリバエ(Agromyza oryzae)、ナモグリバエ(Chromatomyia horticola)、ナスハモグリバエ(Liriomyza bryoniae)、ネギハモグリバエ(Liriomyza chinensis)、トマトハモグリバエ(Liriomyza sativae)、マメハモグリバエ(Liriomyza trifolii)、タネバエ(Delia platura)、テンサイモグリハナバエ(Pegomya cunicularia)、アップルマゴット(Rhagoletis pomonella)、ヘシアンフライ(Mayetiola destructor)、イエバエ(Musca domestica)、サシバエ(Stomoxys calcitrans)、ヒツジシラミバエ(Melophagus ovinus)、ウシバエ(Hypoderma bovis)、キスジウシバエ(Hypoderma lineatum)、ヒツジバエ(Oestrus ovis)、ツェツェバエ(Glossina palpalis, Glossina morsitans)、キアシオオブユ(Prosimulium yezoensis)、ウシアブ(Tabanus trigonus)、オオチョウバエ(Telmatoscopus albipunctatus)、トクナガヌカカ(Leptoconops nipponensis)、アカイエカ(Culex pipiens pallens)、ネッタイシマカ(Aedes aegypti)、ヒトスジシマカ(Aedes albopictus)、シナハマダラカ(Anopheles hyracanus sinesis)等の双翅目昆虫。 Soybean flies (Asphondylia yushimai), mud wings (Sitodiplosis mosellana), sand flies (Bactrocera cucurbitae), citrus fruit flies (Bactrocera dorsalis), citrus fruit ross (Ceratitis capitata), rice larvae rel Leafhopper (Agromyza oryzae), Leafworm (Chromatomyia horticola), Eggplant leaffly (Liriomyza bryoniae), Leafhopper fly (Liriomyza chinensis), Tomato leaffly (Liriomyza sativae), Beanworm fly (Liriomyza trifolia) (Pegomya cunicularia), Apple Maggot (Rhagoletis pomonella), Hessian Fly (Mayetiola destructor), Housefly (Musca domestica), Stomoxys calcitrans, Sheep Flies (Melophagus ovinu) s), bullflies (Hypoderma bovis), bullflies (Hypoderma lineatum), sheep flies (Oestrus ovis), tsetse flies (Glossina palpalis, Glossina morsitans), bark fly (Prosimulium yezoensis), bullfly (Tabanus trigonco), moth butterfly Diptera such as Leptoconops nipponensis, Culex pipiens pallens, Aedes aegypti, Aedes albopictus, Anopheles hyracanus sinesis.
クリハバチ(Apethymus kuri)、カブラハバチ(Athalia rosae)、チュウレンジハバチ(Arge pagana)、マツノキハバチ(Neodiprion sertifer)、クリタマバチ(Dryocosmus kuriphilus)、グンタイアリ(Eciton burchelli, Eciton schmitti)、クロオオアリ(Camponotus japonicus)、オオスズメバチ(Vespa mandarina)、ブルドックアント(Myrmecia spp.)、ファイヤーアント類(Solenopsis spp.)、ファラオアント(Monomorium pharaonis)等の膜翅目昆虫。 Bumblebee (Apethymus kuri), Athalia rosae, Bumblebee (Arge pagana), Bumblebee (Neodiprion sertifer), Bumblebee (Dryocosmus kuriphilus), Gunciari (Eciton burchelli, Eciton schmitus) Hymenoptera insects such as mandarina, bulldog ant (Myrmecia spp.), fire ant (Solenopsis spp.), pharaoh ant (Monomorium pharaonis).
エンマコオロギ(Teleogryllus emma)、ケラ(Gryllotalpa orientalis)、トノサマバッタ(Locusta migratoria)、コバネイナゴ(Oxya yezoensis)、サバクワタリバッタ(Schistocerca gregaria)等の直翅目昆虫。 Direct insects such as Teleogryllus emma, Gryllotalpa orientalis, Locusta migratoria, Oxya yezoensis, and Schistocerca gregaria.
トゲナシシロトビムシ(Onychiurus folsomi)、シベリアシロトビムシ(Onychiurus sibiricus)、キボシマルトビムシ(Bourletiella hortensis)等の粘管目昆虫。 Coleoptera insects such as Onychiurus folsomi, Onychiurus sibiricus and Bourletiella hortensis.
クロゴキブリ(Periplaneta fuliginosa)、ヤマトゴキブリ(Periplaneta japonica)、チャバネゴキブリ(Blattella germanica)等の網翅目昆虫。 Reticulate insects such as the black cockroach (Periplaneta fuliginosa), the cockroach (Periplaneta japonica) and the German cockroach (Blattella germanica).
イエシロアリ(Coptotermes formosanus)、ヤマトシロアリ(Reticulitermes speratus)、タイワンシロアリ(Odontotermes formosanus)等のシロアリ目昆虫。 Termite insects such as termites (Coptotermes formosanus), Yamato termites (Reticulitermes speratus), and Thai termites (Odontotermes formosanus).
ネコノミ(Ctenocephalides felis)、イヌノミ(Ctenocephalides canis)、ニワトリノミ(Echidnophaga gallinacea)、ヒトノミ(Pulex irritans)、ケオプスネズミノミ(Xenopsylla cheopis)等のノミ目類。 Fleas such as cat fleas (Ctenocephalides felis), dog fleas (Ctenocephalides canis), chicken fleas (Echidnophaga gallinacea), human fleas (Pulex irritans), keops rat fleas (Xenopsylla cheopis).
ニワトリオオハジラミ(Menacanthus stramineus)、ウシハジラミ(Bovicola bovis)等のハジラミ目昆虫。 Insects such as chicken lice (Menacanthus stramineus) and bovine lice (Bovicola bovis).
ウシジラミ(Haematopinus eurysternus)、ブタジラミ(Haematopinus suis)、ウシホソジラミ(Linognathus vituli)、ケブカウシジラミ(Solenopotes capillatus)等のシラミ目昆虫。 Lice insects such as cattle lice (Haematopinus eurysternus), pig lice (Haematopinus suis), cattle white lice (Linognathus vituli), solenopotes capillatus and the like.
シクラメンホコリダニ(Phytonemus pallidus)、チャノホコリダニ(Polyphagotarsonemus latus)、スジブトホコリダニ(Tarsonemus bilobatus)等のホコリダニ類。 Dust mites such as cyclamen dust mites (Phytonemus pallidus), chano dust mites (Polyphagotarsonemus latus), and mites (Tarsonemus bilobatus).
ハクサイダニ(Penthaleus erythrocephalus)、ムギダニ(Penthaleus major)等のハシリダニ類。 Spider mites such as cabbage mite (Penthaleus erythrocephalus) and wheat mite (Penthaleus major).
イネハダニ(Oligonychus shinkajii)、ミカンハダニ(Panonychus citri)、クワオオハダニ(Panonychus mori)、リンゴハダニ(Panonychus ulmi)、カンザワハダニ(Tetranychus kanzawai)、ナミハダニ(Tetranychus urticae)等のハダニ類。 Spider mites such as rice spider mite (Oligonychus shinkajii), citrus spider mite (Panonychus citri), swan spider mite (Panonychus mori), apple spider mite (Panonychus ulmi), Kanzawa spider mite (Tetranychus kanzawai), spider mite (Tetranychus urticae), etc.
チャノナガサビダニ(Acaphylla theavagrans)、チューリップサビダニ(Aceria tulipae)、トマトサビダニ(Aculops lycopersici)、ミカンサビダニ(Aculops pelekassi)、リンゴサビダニ(Aculus schlechtendali)、ニセナシサビダニ(Eriophyes chibaensis)、シトラスラストマイト(Phyllocoptruta oleivora)等のフシダニ類。 Chinese cabbage mite (Acaphylla theavagrans), Tulip rust mite (Aceria tulipae), Tomato rust mite (Aculops lycopersici), Citrus radix (Aculops pelekassi), Apple rust mite (Aculus schlechtendali), Green rust mite (Eriophyes chitrastrostrum) ) Etc.
ロビンネダニ(Rhizoglyphus robini)、ケナガコナダニ(Tyrophagus putrescentiae)、ホウレンソウケナガコナダニ(Tyrophagus similis)等のコナダニ類。 Acarids such as Robin tick (Rhizoglyphus robini), Tyrophagus putrescentiae, Tyrophagus similis.
ミツバチヘギイタダニ(Varroa jacobsoni)等のハチダニ類。 Spider mites such as honeybees (Varroa jacobsoni).
オウシマダニ(Boophilus microplus)、クリイロコイタマダニ(Rhipicephalus sanguineus)、フタトゲチマダニ(Haemaphysalis longicornis)、キチマダニ(Haemaphysalis flava)、ツリガネチマダニ(Haemaphysalis campanulata)、ヤマトマダニ(Ixodes ovatus)、シュルツェマダニ(Ixodes persulcatus)、オオマダニ(Amblyomma spp.)、アミメマダニ(Dermacentor spp.)等のマダニ類。 Tick tick (Boophilus microplus), Rhipicephalus sanguineus, Tick tick (Haemaphysalis longicornis), Tick tick (Haemaphysalis flava), Tick tick (Haemaphysalis campanulata), Tick tick (Ixo tus tick (Ixo tus tick) Tick such as Amblyomma spp.), Dermacentor spp.
ワクモRed mite(Dermanyssus gallinae)、イエダニTropical rat mite(Ornithonyssus bacoti)、トリサシダニNorthern fowl mite(Ornithonyssus sylviarum)等のトゲダニ亜目(Mesostigmata)ダニ類。 Mesostigmata ticks such as red mite (Dermanyssus gallinae), house dust mite Tropical rat mite (Ornithonyssus bacoti), avian mite Northern fowl mite (Ornithonyssus sylviarum).
イヌツメダニ(Cheyletiella yasguri)、ネコツメダニ(Cheyletiella blakei)等のツメダニ類。 Crawling ticks, such as the crayfish tick (Cheyletiella yasguri) and the cat crawfish tick (Cheyletiella blakei).
イヌニキビダニ(Demodex canis)、ネコニキビダニ(Demodex cati)などのニキビダニ類。 Acne mites, such as the Inode mite (Demodex canis) and the Caterpillar mite (Demodex cati).
ヒツジキュウセンダニ(Psoroptes ovis)等のキュウセンダニ類。 Cucumber mites such as sheep cucumber mites (Psoroptes ovis).
センコウヒゼンダニ(Sarcoptes scabiei)、ネコショウセンコウヒゼンダニ(Notoedres cati)、ニワトリヒゼンダニ(Knemidocoptes spp.)等のヒゼンダニ類。 Spider mites, such as Sarcoptes scabiei, cattle spider mite (Notoedres cati), chicken spider mite (Knemidocoptes spp.).
オカダンゴムシ(Armadillidium vulgare)等の甲殻類。 Crustaceans such as Armadillidium vulgare.
スクミリンゴガイ(Pomacea canaliculata)、アフリカマイマイ(Achatina fulica)、ナメクジ(Meghimatium bilineatum)、チャコウラナメクジ(Limax Valentiana)、ウスカワマイマイ(Acusta despecta sieboldiana)、ミスジマイマイ(Euhadra peliomphala)等の腹足類。 Gastropods such as Pomacea canaliculata, African mussel (Achatina fulica), Slug (Meghimatium bilineatum), Chinese scallop (Limax Valentiana), Uskawamai (Acusta despecta sieboldiana), Mishamai mai (Euhadra peliomphala).
ミナミネグサレセンチュウ(Pratylenchus coffeae)、キタネグサレセンチュウ(Pratylenchus penetrans)、クルミネグサレセンチュウ(Pratylenchus vulnus)、ジャガイモシストセンチュウ(Globodera rostochiensis)、ダイズシストセンチュウ(Heterodera glycines)、キタネコブセンチュウ(Meloidogyne hapla)、サツマイモネコブセンチュウ(Meloidogyne incognita)、イネシンガレセンチュウ(Aphelenchoides besseyi)、マツノザイセンチュウ(Bursaphelenchus xylophilus)等の線虫類。 Southern nematode nematode (Pratylenchus coffeae), Kitane equestrian nematode (Pratylenchus penetrans), Klemine easter nematode (Pratylenchus vulnus), potato cyst nematode (Globodera rostochiensis), soybean cyst nematode (Heterodera hemopus) Nematodes such as root-knot nematodes (Meloidogyne incognita), rice-spotted nematodes (Aphelenchoides besseyi), and pine wood nematodes (Bursaphelenchus xylophilus).
ノサシバエ又はHaematobia irritans、アブ又はTabanus spp.、サシバエ又はStomoxys calcitrans、ブユ又はSimulium spp.、メクラアブ又はChrysops spp.、シラミバエ又はMelophagus ovinus、及びツェツェバエ又はGlossina spp.等の成虫のハエ。 Wild flies or Haematobia irritans, Abu or Tabanus spp. , Flies or Stomoxys calcitrans, blackfish or Simulium spp. , Mekurabu or Chrysops spp. , Lice flies or Melophagus ovinus, and tsetse flies or Glossina spp. Adult flies such as.
ヒツジバエ(Oestrus ovisおよびCuterebra spp.)、クロバエ又はPhaenicia spp.、ラセンウジバエ又はCochliomyia hominivorax、ウシバエ又はHypoderma spp.、ウマのフリースワーム(fleeceworm)及びGastrophilus等の寄生性のハエウジ。 Sheep flies (Oestrus ovis and Cuterebra spp.), Black flies or Phaenicia spp. , Flyfly or Cochliomyia hominivorax, cowfly or Hypoderma spp. Parasitic flies such as horse fleeceworm and Gastrophilus.
Culex spp.、Anopheles spp.及びAedes spp.等の蚊。 Culex spp. Anopheles spp. And Aedes spp. Mosquitoes etc.
また、本発明化合物を用いて防除しうる家畜、家禽、愛玩動物等の内部寄生虫としては具体的には、例えば、下記の内部寄生虫等が挙げられるが、本発明はこれらのみに限定されるものではない。 Specific examples of endoparasites such as livestock, poultry, and pet animals that can be controlled using the compounds of the present invention include the following endoparasites, but the present invention is not limited to these. It is not something.
ヘモンクス属(Haemonchus)、トリコストロンギルス属(Trichostrongylus)、オステルターギヤ属(Ostertagia)、ネマトディルス属(Nematodirus)、クーペリア属(Cooperia)、アスカリス属(Ascaris)、ブノストムーム属(Bunostomum)、エスファゴストムーム属(Oesophagostomum)、チャベルチア属(Chabertia)、トリキュリス属(Trichuris)、ストロンギルス属(Strongylus)、トリコネマ属(Trichonema)、ディクチオカウルス属(Dictyocaulus)、キャピラリア属(Capillaria)、ヘテラキス属(Heterakis)、トキソカラ属(Toxocara)、アスカリディア属(Ascaridia)、オキシウリス属(Oxyuris)、アンキロストーマ属(Ancylostoma)、ウンシナリア属(Uncinaria)、トキサスカリス属(Toxascaris)、パラスカリス属(Parascaris)などの線虫類。 Haemonchus, Trichostrongylus, Ostertagia, Nematodirus, Couperia, Ascaris, Bunostomum, Esphagotome Genus (Oesophagostomum), Chabertia, Trichuris, Strongylus, Trichonema, Dictyocaulus, Capillaria, Heterakis ), Toxocara, Ascaridia, Oxyuris, Ancylostoma, Uncinaria, Toxacaris, Parascaris and other nematodes .
ブツヘレリア属(Wuchereria)、ブルージア属(Brugia)、オンコセルカ属(Onchocerca)、ディロフィラリア属(Dirofilaria)、ロア糸状虫属(Loa)などのフィラリア科(Filariidae)線虫類。 Filariidae nematodes such as Wuchereria, Bruga, Onchocerca, Dirofilaria, and Loa.
ドラクンクルス属(Deacunculus)などの蛇状線虫科(Dracunculidae)線虫類。 Dracunculidae nematodes, such as the genus Deacunculus.
犬条虫(Dipylidium caninum)、猫条虫(Taenia taeniaeformis)、有鉤条虫(Taenia solium)、無鉤条虫(Taenia saginata)、縮小条虫(Hymenolepis diminuta)、ベネデン条虫(Moniezia benedeni)、広節裂頭条虫(Diphyllobothrium latum)、マンソン裂頭条虫(Diphyllobothrium erinacei)、単包条虫(Echinococcus granulosus)、多包条虫(Echinococcus multilocularis)などの条虫類。 Dogworms (Dipylidium caninum), cats (Taenia taeniaeformis), rodents (Taenia solium), striped tapeworms (Taenia saginata), contracted tapeworms (Hymenolepis diminuta), Beneden tapeworms (Moniezia benedeni), Tapeworms such as Diphyllobothrium latum, Diphyllobothrium erinacei, Echinococcus granulosus, Echinococcus multilocularis.
肝蛭(Fasciola hepatica,F.gigantica)、ウエステルマン肺吸虫(Paragonimus westermani)、肥大吸虫(Fasciolopsic buski)、膵吸虫(Eurytrema pancreaticum,E.coelomaticum)、肝吸虫(Clonorchis sinensis)、日本住血吸虫(Schistosoma japonicum)、ビルハルツ住血吸虫(Schistosoma haematobium)、マンソン住血吸虫(Schistosoma mansoni)などの吸虫類。 Fasciola hepatica (F. gigantica), Westermann lung fluke (Paragonimus westermani), hypertrophied fluke (Fasciolopsic buski), pancreatic fluke (Eurytrema pancreaticum, E. coelomaticum), liver fluke (Clonorchis sinensis), Japanese schistosomiasis (Schistosoma) japonicum), Schistosoma haematobium, Schistosoma mansoni, etc.
エイメリア・テネラ(Eimeria tenella)、エイメリア・アセルブリナ(Eimeria acervulina)、エイメリア・ブルネッチ(Eimeria brunetti)、エイメリア・マクシマ(Eimeria maxima)、エイメリア・ネカトリクス(Eimeria necatrix)、エイメリア・ボビス(Eimeria bovis)、エイメリア・オビノイダリス(Eimeria ovinoidalis)のようなエイメリア類(Eimeria spp.)。 Eimeria tenella, Eimeria acervulina, Eimeria brunetti, Eimeria maxima, Eimeria necatrix, Eimeria bovis, Eimeria bovis Eimeria spp., Such as Eimeria ovinoidalis.
クルーズトリパノソーマ(Trypanosoma cruzi)、リーシュマニア類(Leishmania spp.)、マラリア原虫(Plasmodium spp.)、バベシア類(Babesis spp.)、トリコモナス類(Trichomonadidae spp.)、ヒストモナス類(Histomanas spp.)、ジアルディア類(Giardia spp.)、トキソプラズマ類(Toxoplasma spp.)、赤痢アメーバ(Entamoeba histolytica)、タイレリア類(Theileria spp.)。 Trypanosoma cruzi, Leishmania spp., Plasmodium spp., Babesis spp., Trichomonadidae spp., Histomanas spp., Giardia (Giardia spp.), Toxoplasma spp., Entamoeba histolytica, Theileria spp.
さらに、本発明化合物は、有機燐系化合物、カーバメート系化合物、ピレスロイド系化合物等の既存の殺虫剤に対して抵抗性の発達した有害生物に対しても有効である。 Furthermore, the compound of the present invention is also effective against pests having developed resistance against existing insecticides such as organophosphorus compounds, carbamate compounds, pyrethroid compounds and the like.
すなわち、本発明化合物は、粘管目(トビムシ目)、網翅目(ゴキブリ目)、直翅目(バッタ目)、シロアリ目、総翅目(アザミウマ目)、半翅目(カメムシ目及びヨコバイ目)、鱗翅目(チョウ目)、鞘翅目(コウチュウ目)、膜翅目(ハチ目)、双翅目(ハエ目)、等翅目(ノミ目)、シラミ目等の昆虫類、ダニ類、腹足類、線虫類等に属する有害生物を、低濃度で有効に防除することが出来る。 That is, the compound of the present invention is composed of the order of slime (Coleoptera), reticulate (Roaches), straight (Plicata), termites, common moths (Thrips), hemimorphs (stink bugs and leafhoppers). Eyes), Lepidoptera (Lepidoptera), Coleoptera (Coleoptera), Hymenoptera (Hymenoptera), Diptera (Flyidae), Isoleoptera (Flea), Liceidae, and other insects, ticks In addition, pests belonging to gastropods, nematodes and the like can be effectively controlled at low concentrations.
一方、本発明化合物はホ乳類、魚類、甲殻類及び益虫(ミツバチ、マルハナバチ等の有用昆虫やツヤコバチ、アブラバチ、ヤドリバエ、ヒメハナカメムシ、カブリダニ等の天敵)に対してほとんど悪影響の無い、極めて有用な特長を有している。 On the other hand, the compound of the present invention has very little adverse effect on mammals, fish, crustaceans and beneficial insects (useful insects such as honeybees and bumblebees, natural enemies such as honeybees, wasps, mistletoe flies, stink bugs, and tick mites) and is extremely useful. It has various features.
本発明化合物を使用するにあたっては、通常、適当な固体担体又は液体担体と混合し、更に所望により界面活性剤、浸透剤、展着剤、増粘剤、凍結防止剤、結合剤、固結防止剤、崩壊剤、消泡剤、防腐剤、分解防止剤等を添加して、液剤(soluble concentrate)、乳剤(emulsifiable concentrate)、水和剤(wettable powder)、水溶剤(water soluble powder)、顆粒水和剤(water dispersible granule)、顆粒水溶剤(water soluble granule)、懸濁剤(suspension concentrate)、乳濁剤(concentrated emulsion)、サスポエマルジョン(suspoemulsion)、マイクロエマルジョン(microemulsion)、粉剤(dustable powder)、粒剤(granule)錠剤(tablet)、乳化性ゲル剤(emulsifiable gel)等の任意の剤型の製剤として実用に供することができる。また、省力化及び安全性向上の観点から、上記任意の剤型の製剤を、水溶性カプセル、水溶性フィルムの袋等の水溶性包装体に封入して供することもできる。 When using the compound of the present invention, it is usually mixed with an appropriate solid carrier or liquid carrier, and further, if desired, a surfactant, penetrant, spreading agent, thickener, antifreeze agent, binder, anti-caking agent. Agent, disintegrating agent, antifoaming agent, antiseptic, anti-degradation agent, etc. added, soluble concentrate, emulsion (emulsifiable concentrate), wettable powder, water soluble powder, granule Water dispersible granule, water soluble granule, suspension concentrate, concentrated emulsion, suspoemulsion, microemulsion, dustable It can be put to practical use as a preparation of any dosage form such as powder, granule tablet, and emulsifiable gel. In addition, from the viewpoint of labor saving and safety improvement, the preparations of any of the above dosage forms can be provided by being enclosed in a water-soluble package such as a water-soluble capsule or a water-soluble film bag.
固体担体としては、例えば、石英、方解石、海泡石、ドロマイト、チョーク、カオリナイト、パイロフィライト、セリサイト、ハロサイト、メタハロサイト、木節粘土、蛙目粘土、陶石、ジークライト、アロフェン、シラス、きら、タルク、ベントナイト、活性白土、酸性白土、軽石、アタパルジャイト、ゼオライト、珪藻土等の天然鉱物質;焼成クレー、パーライト、シラスバルーン、バーミキュライト、アタパルガスクレー、焼成珪藻土等の天然鉱物質の焼成品;炭酸マグネシウム、炭酸カルシウム、炭酸ナトリウム、炭酸水素ナトリウム、硫酸アンモニウム、硫酸ナトリウム、硫酸マグネシウム、リン酸水素二アンモニウム、リン酸二水素アンモニウム、塩化カリウム等の無機塩類;ブドウ糖、果糖、しょ糖、乳糖等の糖類;澱粉、粉末セルロース、デキストリン等の多糖類;尿素、尿素誘導体、安息香酸、安息香酸の塩等の有機物;木粉、コルク粉、トウモロコシ穂軸、クルミ殻、タバコ茎等の植物類;フライアッシュ、ホワイトカーボン(例えば、含水合成シリカ、無水合成シリカ、含水合成シリケート等)、肥料;等が挙げられる。 Examples of the solid support include quartz, calcite, gypsum, dolomite, chalk, kaolinite, pyrophyllite, sericite, halosite, metahalosite, kibushi clay, glazed clay, porcelain stone, dikelite, Natural minerals such as allophane, shirasu, giraffe, talc, bentonite, activated clay, acid clay, pumice, attapulgite, zeolite, diatomaceous earth, etc .; natural minerals such as calcined clay, perlite, shirasu balloon, vermiculite, attapalgus clay, calcined diatomaceous earth Quality baked goods; inorganic salts such as magnesium carbonate, calcium carbonate, sodium carbonate, sodium hydrogen carbonate, ammonium sulfate, sodium sulfate, magnesium sulfate, diammonium hydrogen phosphate, ammonium dihydrogen phosphate, potassium chloride; glucose, fructose, sucrose , Sugars such as lactose; starch, powder Polysaccharides such as cellulose and dextrin; Organic substances such as urea, urea derivatives, benzoic acid, benzoic acid salts; plants such as wood flour, cork flour, corn cobs, walnut shells, tobacco stems; fly ash, white carbon ( For example, hydrous synthetic silica, anhydrous synthetic silica, hydrous synthetic silicate, etc.), fertilizer;
液体担体としては、例えば、キシレン、アルキル(C9又はC10等)ベンゼン、フェニルキシリルエタン、アルキル(C1又はC3等)ナフタレン等の芳香族炭化水素類;マシン油、ノルマルパラフィン、イソパラフィン、ナフテン等の脂肪族炭化水素類;ケロシン等の芳香族炭化水素と脂肪族炭化水素の混合物;エタノール、イソプロパノール、シクロヘキサノール、フェノキシエタノール、ベンジルアルコール等のアルコール;エチレングリコール、プロピレングリコール、ジエチレングリコール、ヘキシレングリコール、ポリエチレングリコール、ポリプロピレングリコール等の多価アルコール;プロピルセロソルブ、ブチルセロソルブ、フェニルセロソルブ、プロピレングリコールモノメチルエーテル、プロピレングリコールモノエチルエーテル、プロピレングリコールモノプロピルエーテル、プロピレングリコールモノブチルエーテル、プロピレングリコールモノフェニルエーテル等のエーテル;アセトフェノン、シクロヘキサノン、γ−ブチロラクトン等のケトン;脂肪酸メチルエステル、コハク酸ジアルキルエステル、グルタミン酸ジアルキルエステル、アジピン酸ジアルキルエステル、フタル酸ジアルキルエステル等のエステル;N−アルキル(C1、C8又はC12等)ピロリドン等の酸アミド;大豆油、アマニ油、ナタネ油、ヤシ油、綿実油、ヒマシ油等の油脂;ジメチルスルホキシド、水;等が挙げられる。 Examples of liquid carriers include aromatic hydrocarbons such as xylene, alkyl (C 9 or C 10 etc.) benzene, phenyl xylyl ethane, alkyl (C 1 or C 3 etc.) naphthalene; machine oil, normal paraffin, isoparaffin Aliphatic hydrocarbons such as naphthene, aromatic hydrocarbons such as kerosene and aliphatic hydrocarbons; alcohols such as ethanol, isopropanol, cyclohexanol, phenoxyethanol, benzyl alcohol; ethylene glycol, propylene glycol, diethylene glycol, hexylene Polyhydric alcohols such as glycol, polyethylene glycol, polypropylene glycol; propyl cellosolve, butyl cellosolve, phenyl cellosolve, propylene glycol monomethyl ether, propylene glycol monoethyl Ethers such as ether, propylene glycol monopropyl ether, propylene glycol monobutyl ether, propylene glycol monophenyl ether; ketones such as acetophenone, cyclohexanone, and γ-butyrolactone; fatty acid methyl esters, succinic acid dialkyl esters, glutamic acid dialkyl esters, adipic acid dialkyl esters , Esters such as dialkyl phthalates; acid amides such as N-alkyl (C 1 , C 8 or C 12 ) pyrrolidone; fats and oils such as soybean oil, linseed oil, rapeseed oil, coconut oil, cottonseed oil, castor oil; dimethyl And sulfoxide, water; and the like.
これら固体担体及び液体担体は、単独で用いても2種以上を併用してもよい。 These solid carriers and liquid carriers may be used alone or in combination of two or more.
界面活性剤としては、例えば、ポリオキシエチレンアルキルエーテル、ポリオキシエチレンアルキル(モノ又はジ)フェニルエーテル、ポリオキシエチレン(モノ、ジ又はトリ)スチリルフェニルエーテル、ポリオキシエチレンポリオキシプロピレンブロックコポリマー、ポリオキシエチレン脂肪酸(モノ又はジ)エステル、ソルビタン脂肪酸エステル、ポリオキシエチレンソルビタン脂肪酸エステル、ヒマシ油エチレンオキサイド付加物、アセチレングリコール、アセチレンアルコール、アセチレングリコールのエチレンオキサイド付加物、アセチレンアルコールのエチレンオキサイド付加物、アルキルグリコシド等のノニオン性界面活性剤;アルキル硫酸エステル塩、アルキルベンゼンスルホン酸塩、リグニンスルホン酸塩、アルキルスルホコハク酸塩、ナフタレンスルホン酸塩、アルキルナフタレンスルホン酸塩、ナフタレンスルホン酸のホルマリン縮合物の塩、アルキルナフタレンスルホン酸のホルマリン縮合物の塩、ポリオキシエチレンアルキルエーテル硫酸又は燐酸エステル塩、ポリオキシエチレン(モノ又はジ)アルキルフェニルエーテル硫酸又は燐酸エステル塩、ポリオキシエチレン(モノ、ジ又はトリ)スチリルフェニルエーテル硫酸又は燐酸エステル塩、ポリカルボン酸塩(例えば、ポリアクリル酸塩、ポリマレイン酸塩、マレイン酸とオレフィンとの共重合物等)、ポリスチレンスルホン酸塩等のアニオン性界面活性剤;アルキルアミン塩、アルキル4級アンモニウム塩等のカチオン性界面活性剤;アミノ酸型、ベタイン型等の両性界面活性剤;シリコーン系界面活性剤、フッ素系界面活性剤;等が挙げられる。 Examples of the surfactant include polyoxyethylene alkyl ether, polyoxyethylene alkyl (mono or di) phenyl ether, polyoxyethylene (mono, di or tri) styryl phenyl ether, polyoxyethylene polyoxypropylene block copolymer, polyoxyethylene Oxyethylene fatty acid (mono or di) ester, sorbitan fatty acid ester, polyoxyethylene sorbitan fatty acid ester, castor oil ethylene oxide adduct, acetylene glycol, acetylene alcohol, ethylene oxide adduct of acetylene glycol, ethylene oxide adduct of acetylene alcohol, Nonionic surfactants such as alkyl glycosides; alkyl sulfate salts, alkylbenzene sulfonates, lignin sulfonates, alkyls Succinate, naphthalenesulfonate, alkylnaphthalenesulfonate, salt of formalin condensate of naphthalenesulfonic acid, salt of formalin condensate of alkylnaphthalenesulfonic acid, polyoxyethylene alkyl ether sulfate or phosphate ester salt, polyoxyethylene (Mono or di) alkyl phenyl ether sulfate or phosphate ester salt, polyoxyethylene (mono, di or tri) styryl phenyl ether sulfate or phosphate ester salt, polycarboxylate (eg, polyacrylate, polymaleate, malee) Copolymer of acid and olefin), anionic surfactant such as polystyrene sulfonate; cationic surfactant such as alkylamine salt and alkyl quaternary ammonium salt; amphoteric surfactant such as amino acid type and betaine type Agent; silicone Surfactants, fluorine-based surfactants; and the like.
これら界面活性剤の含有量は、特に限定されるものではないが、本発明の製剤100質量部に対し、通常0.05〜20質量部の範囲が望ましい。また、これら界面活性剤は、単独で用いても2種以上を併用してもよい。 Although content of these surfactants is not specifically limited, The range of 0.05-20 mass parts is desirable normally with respect to 100 mass parts of preparations of this invention. These surfactants may be used alone or in combination of two or more.
本発明化合物の施用薬量は、適用場面、施用時期、施用方法、栽培作物等により差異は有るが、通常は、有効成分量として、ヘクタール(ha)当たり0.005〜50kg程度が適当である。 The application amount of the compound of the present invention varies depending on the application scene, application time, application method, cultivated crops, etc., but usually about 0.005 to 50 kg per hectare (ha) is appropriate as the amount of active ingredient. .
一方、家畜及び愛玩動物としての哺乳動物及び鳥類の外部又は内部寄生虫の防除に、本発明化合物を使用するにあたっては、本発明化合物の有効量を製剤用添加物とともに、経口投与;注射(筋肉内、皮下、静脈内、腹腔内等)などの非経口投与;浸漬、スプレー、入浴、洗浄、滴下(pouring-on)及びスポッティング(spotting-on)、ダスティング(dusting)などの経皮投与;経鼻投与;等により投与することができる。本発明化合物はまた、細片、プレート、バンド、カラー、イヤー・マーク(ear mark)、リム(limb)・バンド、標識装置などを用いた成形製品により投与することができる。 On the other hand, when using the compound of the present invention for the control of the ectoparasites of mammals and birds as domestic animals and pets, an effective amount of the compound of the present invention is administered orally together with a pharmaceutical additive; Parenteral administration such as internal, subcutaneous, intravenous, intraperitoneal, etc .; transdermal administration such as immersion, spraying, bathing, washing, pouring-on and spotting-on, dusting; It can be administered by nasal administration or the like. The compounds of the present invention can also be administered by molded products using strips, plates, bands, collars, ear marks, limb bands, labeling devices and the like.
投与にあたっては、本発明化合物を投与経路に適した任意の剤型とすることができる。 In administration, the compound of the present invention can be in any dosage form suitable for the administration route.
本発明化合物を用いて外部又は内部寄生虫を駆除する場合、有効成分である式(1)で表される本発明化合物の好ましい投与量は、防除すべき対象寄生虫の種類、投与される対象動物の種又は投与方法などにより左右されるが、通常は、投与する対象動物の体重当たり、0.01〜100mg/kg、好ましくは0.01〜50mg/kgである。特にイヌへの投与量は、対象となるイヌの種類又は年齢、あるいは防除する外部寄生虫によっても変わり得るが、対象となるイヌの生体重1kg当たり、通常1〜5000mg/kgであり、好ましくは1〜100mg/kgである。 When exterminating ectoparasites or endoparasites using the compound of the present invention, the preferred dosage of the compound of the present invention represented by the formula (1), which is the active ingredient, is the type of the target parasite to be controlled and the subject to be administered. Although it depends on the species of animal or administration method, it is generally 0.01-100 mg / kg, preferably 0.01-50 mg / kg, based on the body weight of the subject animal to be administered. In particular, the dose to the dog may vary depending on the type or age of the target dog or the ectoparasite to be controlled, but is usually 1 to 5000 mg / kg per kg of the living weight of the target dog, preferably 1-100 mg / kg.
本発明化合物の投与により外部又は内部寄生虫を駆除する場合、その投与間隔は、防除すべき対象寄生虫の種類、投与される対象動物の種又は投与方法などにより左右されるが、通常は、毎日〜年1回の範囲で任意に設定することができる。好ましくは週1回〜6ヶ月に1回であり、より好ましくは毎日(24時間)、毎月、月1回、2か月に1回又は3ヶ月に1回である。 When exterminating ectoparasites or endoparasites by administration of the compound of the present invention, the administration interval depends on the type of target parasite to be controlled, the type of target animal to be administered or the administration method, etc. It can be arbitrarily set in a range from daily to once a year. It is preferably once a week to once every 6 months, more preferably daily (24 hours), monthly, once a month, once every two months, or once every three months.
また、本発明化合物をイヌの外部寄生虫の防除に使用する場合、本発明化合物をイヌに投与するタイミングとして、例えば食餌開始30分直前や食餌終了120分後のタイミングにてイヌに経口投与することが挙げられる。ここでいう食餌開始30分前および食餌終了120分後とは、栄養摂取を目的にイヌに与えられた餌を摂食する行為を基準とする。例えばイヌの食餌時間が20分である場合は、規定される時間は食事行為を基準として、食餌開始30分前から食餌終了120分後の合計170分間となる。食餌中に一旦食餌を中断し、本発明化合物を経口投与した後に食餌を再開する場合も含む。尚、本明細書において食餌とは動物が餌を食する行為を意味する。 In addition, when the compound of the present invention is used for controlling ectoparasites in dogs, the compound of the present invention is orally administered to the dog, for example, at a timing just before 30 minutes after the start of food or 120 minutes after the end of the diet. Can be mentioned. Here, 30 minutes before the start of the diet and 120 minutes after the end of the diet are based on the act of eating the food given to the dog for the purpose of nutrition intake. For example, when the dog's diet time is 20 minutes, the prescribed time is a total of 170 minutes from 30 minutes before the start of the diet to 120 minutes after the end of the diet, based on the meal act. This includes the case where the diet is interrupted once during the diet, and the diet is resumed after oral administration of the compound of the present invention. In addition, in this specification, a diet means the act which an animal eats.
一般にイヌの一日の食餌回数は犬種や年齢や習慣によって異なるが、通常は生後半年未満のイヌで1日3〜4回、生後半年から1年未満のイヌで1日2〜3回、1歳〜5歳程度の成犬で1日2回、6歳以上の老犬で1日2〜3回程度とされている。本発明において、食餌とは栄養を摂取することを目的とした摂食行為を意味し、所謂イヌの躾や訓練を目的に与える餌等を与える行為は含まない。 In general, the number of daily diets for dogs varies depending on the breed, age and custom, but usually 3-4 times a day for dogs younger than 2 years, 2 to 3 times a day for dogs less than 1 year from the second half of life, It is considered to be twice a day for adult dogs of about 1 to 5 years old and about 2 to 3 times a day for elderly dogs over 6 years old. In the present invention, diet means an act of feeding for the purpose of ingesting nutrition, and does not include an act of feeding so-called dogs and food for the purpose of training.
調製される任意の剤型としては、粉剤、粒剤、水和剤、ペレット、錠剤、大丸薬、カプセル剤、活性化合物を含む成形製品等の固体調製物;注射用液剤、経口用液剤、皮膚上又は体腔中に用いる液剤等の液剤調製物;滴下(Pour-on)剤、点下(Spot-on)剤、フロアブル剤、乳剤等の溶液調製物;軟膏剤、ゲル等の半固体調製物;などが挙げられる。 Arbitrary dosage forms to be prepared include powders, granules, wettable powders, pellets, tablets, large pills, capsules, solid preparations such as molded products containing active compounds; injection solutions, oral solutions, skin Liquid preparations such as liquids used above or in body cavities; Solution preparations such as Pour-on, Spot-on, flowable and emulsion; Semi-solid preparations such as ointments and gels And so on.
本発明化合物を経口投与する場合の剤型としては、例えば、錠剤(Tablets)、固型剤(Chewables)、カプセル剤(Capsules)、丸剤(Pills)、大丸剤(Boluses)、顆粒剤(Granules)、散剤(Powders)等の固体調製物;ペースト剤(Pastes)、ゲル剤(Gels)等の半固体調製物;ドリンク剤(Drinks)等の液体調製物;などが挙げられる。 Examples of the dosage form for oral administration of the compound of the present invention include tablets (Tablets), solid dosage forms (Chewables), capsules (Capsules), pills (Pills), large pills (Boluses), and granules (Granules). ), Solid preparations such as powders; semi-solid preparations such as pastes and gels; liquid preparations such as drinks; and the like.
また、経皮投与する場合の剤型としては、例えば、粉剤(Powders)等の固体調製物;クリーム剤(Cream)、軟膏剤(Salve and Ointment)、ペースト剤(Pastes)、ゲル剤(Gels)等の半固体調製物;スプレー剤(Spray)、エアゾール剤(Aerosols)、液剤(Solutions and Emulsions)、懸濁剤(Suspensions)、ローション剤(Lotions)等の液体調製物;などが挙げられる。 The dosage form for transdermal administration includes, for example, solid preparations such as powders (Cream), ointments (Salve and Ointment), pastes (Pastes), gels (Gels) Semi-solid preparations such as; sprays (Spray), aerosols (Aerosols), solutions (Solutions and Emulsions), suspensions (Suspensions), liquid preparations such as lotions (Lotions); and the like.
さらに、注射により投与する場合の剤型としては、例えば、液剤(Solutions and Emulsions)、懸濁剤(Suspensions)等の液体調製物などが挙げられ、経鼻投与する場合の剤型としては、例えば、エアゾール剤(Aerosols)等の液体調製物などが挙げられる。 Furthermore, examples of the dosage form for administration by injection include liquid preparations such as solutions (Solutions and Emulsions) and suspensions (Suspensions). Examples of dosage forms for nasal administration include And liquid preparations such as aerosols (Aerosols).
また、畜舎等の動物の飼育環境に散布処理する場合の剤型としては、例えば、水和剤(Wettable powders)、粉剤(Dusts)、粒剤(Granules)などの固体調製物;乳剤(Emulsions)、フロアブル剤(Suspension concentrates)などの液体調製物;などが挙げられる。 In addition, as a dosage form in the case of spraying treatment in an animal breeding environment such as a barn, for example, solid preparations such as wettable powders, powders (Dusts), granules (Emulsions); emulsions (Emulsions) And liquid preparations such as flowable concentrates (Suspension concentrates).
なお、本発明の寄生虫防除剤に用いられる製剤は、これらの剤型のみに限定されるものではない。 In addition, the formulation used for the parasite control agent of this invention is not limited only to these dosage forms.
固体調製物は、そのまま経口投与するか、或いは水で希釈して、経皮投与、畜舎等の動物の飼育環境に散布処理するなどして用いることができる。 The solid preparation can be administered orally as it is, or diluted with water and used by transdermal administration or spraying in an animal breeding environment such as a barn.
経口投与で用いられる固体調製物は、式(1)で表される化合物又はその塩と、1種又は2種以上の経口投与に適した賦形剤及び結合剤、さらに、必要ならば滑沢剤、崩壊剤、染料、顔料などの生理学的に許容しうる添加剤とを混合し、所望の形状に成形することにより調製できる。 The solid preparation used for oral administration comprises a compound represented by formula (1) or a salt thereof, one or more excipients and binders suitable for oral administration, and if necessary, a lubricant. It can be prepared by mixing with a physiologically acceptable additive such as an agent, a disintegrant, a dye, and a pigment and molding the mixture into a desired shape.
賦形剤及び結合剤としては、例えば、ラクトース、シュクロース、マンニトール、ソルビトール等の糖又は糖誘導体;トウモロコシ澱粉、コムギ澱粉、コメ澱粉、ジャガイモ澱粉等の澱粉;メチルセルロース、カルボキシメチルセルロース、ヒドロキシエチルセルロース、ヒドロキシプロピルセルロース、ヒドロキシプロピルメチルセルロース等のセルロース又はセルロース誘導体;ゼイン、ゼラチン等のタンパク質又はタンパク質誘導体;ハチミツ、アラビアゴム糊、ポリビニルアルコール、ポリビニルピロリドン等の合成高分子化合物;などが挙げられる。 Examples of the excipient and binder include sugars or sugar derivatives such as lactose, sucrose, mannitol, and sorbitol; starches such as corn starch, wheat starch, rice starch, and potato starch; methylcellulose, carboxymethylcellulose, hydroxyethylcellulose, hydroxy Examples thereof include cellulose or cellulose derivatives such as propylcellulose and hydroxypropylmethylcellulose; proteins or protein derivatives such as zein and gelatin; synthetic polymer compounds such as honey, gum arabic glue, polyvinyl alcohol, and polyvinylpyrrolidone.
滑沢剤としては、例えば、ステアリン酸マグネシウムなどが挙げられ、崩壊剤としては、例えば、セルロース、寒天、アルギン酸、架橋ポリビニルピロリジノン、炭酸塩などが挙げられる。 Examples of the lubricant include magnesium stearate, and examples of the disintegrant include cellulose, agar, alginic acid, cross-linked polyvinyl pyrrolidinone, and carbonate.
さらに、経口投与で用いられる固体調製物のうち、特にチュアブル剤などの固型剤の場合、投与する動物の好む味、食感、風味を付与する添加物も用いられるが、本発明の寄生虫防除剤組成物の固体調製物に用いられる担体及び添加剤は、これらのみに限定されるものではない。 Furthermore, among solid preparations used for oral administration, especially in the case of solid preparations such as chewable agents, additives that give taste, texture, and taste preferred by the animals to be administered can also be used. The carriers and additives used in the solid preparation of the control agent composition are not limited to these.
液体調製物は、そのまま経皮又は注射によって投与するか、或いは飼料に混和して、経口投与、水で希釈して経皮投与、畜舎等の動物の飼育環境に散布処理する等して用いることができる。 Liquid preparations should be administered as they are, transdermally or by injection, or mixed with feed, orally administered, diluted with water, transdermally administered, and sprayed into animal breeding environments such as barns. Can do.
注射用液剤は、静脈内、筋肉内及び皮下に投与できる。注射用液剤は、活性化合物を適当な溶媒に溶解させ、必要ならば可溶化剤、酸、塩基、緩衝用塩、酸化防止剤、保護剤等の添加剤を加えることにより調製できる。 Injection solutions can be administered intravenously, intramuscularly and subcutaneously. Injection solutions can be prepared by dissolving the active compound in a suitable solvent and adding additives such as solubilizers, acids, bases, buffer salts, antioxidants, protective agents, etc., if necessary.
適当な溶媒としては、水、エタノール、ブタノール、ベンジルアルコール、グリセリン、プロピレングリコール、ポリエチレングリコール、N−メチルピロリドン及びこれらの混合物、生理学的に許容しうる植物油、注射に適する合成油等が挙げられる。 Suitable solvents include water, ethanol, butanol, benzyl alcohol, glycerin, propylene glycol, polyethylene glycol, N-methylpyrrolidone and mixtures thereof, physiologically acceptable vegetable oils, synthetic oils suitable for injection, and the like.
可溶化剤としては、ポリビニルピロリドン、ポリオキシエチル化されたヒマシ油、ポリオキシエチル化されたソルビタンエステル等が挙げられる。 Examples of the solubilizer include polyvinyl pyrrolidone, polyoxyethylated castor oil, polyoxyethylated sorbitan ester, and the like.
保護剤としては、ベンジルアルコール、トリクロロブタノール、p−ヒドロキシ安息香酸エステル、n−ブタノール等が挙げられる。 Examples of the protective agent include benzyl alcohol, trichlorobutanol, p-hydroxybenzoic acid ester, n-butanol and the like.
経口液剤は、直接又は希釈して投与することができる。注射用液剤と同様に調製することができる。 Oral solutions can be administered directly or diluted. It can be prepared in the same manner as an injectable solution.
フロアブル剤、乳剤等は、直接又は希釈して、経皮的に、又は環境処理にて投与できる。 Flowable agents, emulsions and the like can be administered directly or diluted, transdermally, or by environmental treatment.
皮膚上で用いる液剤は、滴下し、広げ、すり込み、噴霧し、散布するか、又は浸漬(浸漬、入浴又は洗浄)により塗布することにより投与できる。これらの液剤は、注射用液剤と同様に調製できる。 Solutions used on the skin can be administered by dripping, spreading, rubbing, spraying, spraying or applying by dipping (immersion, bathing or washing). These solutions can be prepared in the same manner as injection solutions.
滴下(Pour-on)剤及び点下(Spot-on)剤は、皮膚の限定された場所に滴下するか、又は噴霧し、これにより活性化合物を皮膚に浸漬させ、全身的に作用させることができる。 Pour-on and spot-on agents can be dripped or sprayed onto a limited area of the skin, so that the active compound is immersed in the skin and acts systemically. it can.
滴下剤及び点下剤は、有効成分を適当な皮膚適合性溶媒又は溶媒混合物に溶解するか、懸濁させるか、又は乳化することにより調製できる。必要ならば、界面活性剤、着色剤、吸収促進物質、酸化防止剤、光安定剤、接着剤等の補助剤を加えてもよい。 Drops and drop preparations can be prepared by dissolving, suspending or emulsifying the active ingredient in suitable skin-compatible solvents or solvent mixtures. If necessary, auxiliary agents such as surfactants, colorants, absorption promoting substances, antioxidants, light stabilizers, and adhesives may be added.
適当な溶媒としては、水、アルカノール、グリコール、ポリエチレングリコール、ポリプロピレングリコール、グリセリン、ベンジルアルコール、フェニルエタノール、フェノキシエタノール、酢酸エチル、酢酸ブチル、安息香酸ベンジル、ジプロピレングリコールモノメチルエーテル、ジエチレングリコールモノブチルエーテル、アセトン、メチルエチルケトン、芳香族及び/又は脂肪族炭化水素、植物又は合成油、DMF(N,N−ジメチルホルムアミド)、流動パラフィン、軽質流動パラフィン、シリコーン、ジメチルアセトアミド、N−メチルピロリドン又は2,2−ジメチル−4−オキシ−メチレン−1,3−ジオキソランが挙げられる。 Suitable solvents include water, alkanol, glycol, polyethylene glycol, polypropylene glycol, glycerin, benzyl alcohol, phenylethanol, phenoxyethanol, ethyl acetate, butyl acetate, benzyl benzoate, dipropylene glycol monomethyl ether, diethylene glycol monobutyl ether, acetone, Methyl ethyl ketone, aromatic and / or aliphatic hydrocarbons, vegetable or synthetic oil, DMF (N, N-dimethylformamide), liquid paraffin, light liquid paraffin, silicone, dimethylacetamide, N-methylpyrrolidone or 2,2-dimethyl- 4-oxy-methylene-1,3-dioxolane is mentioned.
吸収促進物質としては、DMSO(ジメチルスルホキシド)、ミリスチン酸イソプロピル、ペラルゴン酸ジプロピレングリコール、シリコーン油、脂肪族エステル、トリグリセリド又は脂肪アルコールが挙げられる。 Absorption promoting substances include DMSO (dimethyl sulfoxide), isopropyl myristate, dipropylene glycol pelargonate, silicone oil, aliphatic esters, triglycerides or fatty alcohols.
酸化防止剤には、亜硫酸塩、メタ重亜硫酸塩、アスコルビン酸、ブチルヒドロキシトルエン、ブチルヒドロキシアニソール又はトコフェロールが挙げられる。 Antioxidants include sulfite, metabisulfite, ascorbic acid, butylhydroxytoluene, butylhydroxyanisole or tocopherol.
乳剤は、経口投与、経皮投与又は注射として投与できる。乳剤は、有効成分を疎水性相又は親水性相に溶解させ、このものを適当な乳化剤により、必要ならば、さらに着色剤、吸収促進物質、保護剤、酸化防止剤、遮光剤、増粘物質等の補助剤と共に、他の相の溶媒と均質化することにより調製できる。 The emulsion can be administered orally, transdermally or as an injection. An emulsion is prepared by dissolving an active ingredient in a hydrophobic phase or a hydrophilic phase, and adding this to a suitable emulsifier, if necessary, a colorant, an absorption promoting substance, a protective agent, an antioxidant, a light-shielding agent, a thickening substance. It can be prepared by homogenizing with other phase solvents together with other auxiliary agents.
疎水性相(油)としては、パラフィン油、シリコーン油、ゴマ油、アーモンド油、ヒマシ油、合成トリグリセリド、ステアリン酸エチル、アジピン酸ジーn−ブチリル、ラウリル酸ヘキシル、ペラルゴン酸ジプロピレングリコール、分枝鎖状の短鎖長脂肪酸と鎖長C16〜C18の飽和脂肪酸とのエステル、ミリスチン酸イソプロピル、パルミチン酸イソプロピル、鎖長C12〜C18の飽和脂肪アルコールのカプリル/カプリン酸エステル、ステアリン酸イソプロピル、オレイン酸オレイル、オレイン酸デシル、オレイン酸エチル、乳酸エチル、ワックス状脂肪酸エステル、フタル酸ジブチル、アジピン酸ジイソプロピル、イソトリデシルアルコール、2−オクチルドデカノール、セチルステアリルアルコール、オレイルアルコール等が挙げられる。 As hydrophobic phase (oil), paraffin oil, silicone oil, sesame oil, almond oil, castor oil, synthetic triglyceride, ethyl stearate, di-n-butyryl adipate, hexyl laurate, dipropylene glycol pelargonate, branched chain Of a short fatty acid having a chain length and a saturated fatty acid having a chain length of C16 to C18, isopropyl myristate, isopropyl palmitate, capryl / caprate of a saturated fatty alcohol having a chain length of C12 to C18, isopropyl stearate, oleyl oleate Decyl oleate, ethyl oleate, ethyl lactate, waxy fatty acid ester, dibutyl phthalate, diisopropyl adipate, isotridecyl alcohol, 2-octyldodecanol, cetyl stearyl alcohol, oleyl alcohol, etc. It is.
親水性相としては、水、プロピレングリコール、グリセリン、ソルビトール等が挙げられる。 Examples of the hydrophilic phase include water, propylene glycol, glycerin, sorbitol and the like.
乳化剤としては、ポリオキシエチル化されたヒマシ油、ポリオキシエチル化されたモノオレフィン酸ソルビタン、モノステアリン酸ソルビタン、モノステアリン酸グリセリン、ステアリン酸ポリオキシエチル、アルキルフェノールポリグリコールエーテルなどの非イオン性界面活性剤;N−ラウリル−β−イミノジプロピオン酸二ナトリウム、レシチンなどの両性界面活性剤;ラウリル硫酸ナトリウム、脂肪アルコール硫酸エーテル、モノ/ジアルキルポリグリコールオルトリン酸エステルのモノエタノールアミン塩などの陰イオン性界面活性剤;塩化セチルトリメチルアンモニウム等の陽イオン性界面活性剤;などが挙げられる。 Emulsifiers include nonionic interfaces such as polyoxyethylated castor oil, polyoxyethylated sorbitan monoolefin acid, sorbitan monostearate, glyceryl monostearate, polyoxyethyl stearate, alkylphenol polyglycol ether, etc. Activators; amphoteric surfactants such as disodium N-lauryl-β-iminodipropionate and lecithin; anions such as sodium lauryl sulfate, fatty alcohol sulfate ether, monoethanolamine salt of mono / dialkyl polyglycol orthophosphate Surfactants; cationic surfactants such as cetyltrimethylammonium chloride; and the like.
他の補助剤としては、カルボキシメチルセルロース、メチルセルロース、ポリアクリレート、アルギネート、ゼラチン、アラビアゴム、ポリビニルピロリドン、ポリビニルアルコール、メチルビニルエーテル、無水マレイン酸の共重合体、ポリエチレングリコール、ワックス、コロイド状シリカ等が挙げられる。 Other adjuvants include carboxymethyl cellulose, methyl cellulose, polyacrylate, alginate, gelatin, gum arabic, polyvinyl pyrrolidone, polyvinyl alcohol, methyl vinyl ether, maleic anhydride copolymer, polyethylene glycol, wax, colloidal silica and the like. It is done.
半固体調製物は、皮膚上に塗布するか、若しくは広げるか、又は体腔中に導入することにより投与できる。ゲルは、注射用液剤について上記したように調製した溶液に、軟膏状の粘稠性を有する透明な物質を生じさせるに十分なシックナーを加えることにより調製できる。 Semi-solid preparations can be administered by application on the skin or spreading or introduction into body cavities. Gels can be prepared by adding sufficient thickener to a solution prepared as described above for injectable solutions to produce a clear material having an ointment-like consistency.
次に、本発明化合物を用いる場合の製剤の配合例を示す。但し、本発明の配合例は、これらに限定されるものではない。なお、以下の配合例において、「部」は質量部を意味する。 Next, formulation examples of the preparation when the compound of the present invention is used are shown. However, the formulation examples of the present invention are not limited to these. In the following formulation examples, “parts” means parts by mass.
〔水和剤〕
本発明化合物 0.1〜80部
固体担体 5〜98.9部
界面活性剤 1〜10部
その他 0〜5部
その他として、例えば、固結防止剤、分解防止剤等が挙げられる。
[Wettable powder]
Compound of the present invention 0.1-80 parts Solid carrier 5-98.9 parts Surfactant 1-10 parts Others 0-5 parts Others include, for example, anti-caking agents and decomposition inhibitors.
〔乳剤〕
本発明化合物 0.1〜30部
液体担体 45〜95部
界面活性剤 4.9〜15部
その他 0〜10部
その他として、例えば、展着剤、分解防止剤等が挙げられる。
〔emulsion〕
Compound of the present invention 0.1-30 parts Liquid carrier 45-95 parts Surfactant 4.9-15 parts Others 0-10 parts Others include, for example, spreading agents, decomposition inhibitors and the like.
〔懸濁剤〕
本発明化合物 0.1〜70部
液体担体 15〜98.89部
界面活性剤 1〜12部
その他 0.01〜30部
その他として、例え、ば凍結防止剤、増粘剤等が挙げられる。
[Suspension]
Compounds of the present invention 0.1 to 70 parts Liquid carrier 15 to 98.89 parts Surfactant 1 to 12 parts Others 0.01 to 30 parts Others include, for example, antifreezing agents and thickeners.
〔顆粒水和剤〕
本発明化合物 0.1〜90部
固体担体 0〜98.9部
界面活性剤 1〜20部
その他 0〜10部
その他として、例えば、結合剤、分解防止剤等が挙げられる。
(Granule wettable powder)
Compound of the present invention 0.1 to 90 parts Solid carrier 0 to 98.9 parts Surfactant 1 to 20 parts Others 0 to 10 parts Others include, for example, binders and decomposition inhibitors.
〔液剤〕
本発明化合物 0.01〜70部
液体担体 20〜99.99部
その他 0〜10部
その他として、例えば、凍結防止剤、展着剤等が挙げられる。
[Liquid]
Compound of the present invention 0.01 to 70 parts Liquid carrier 20 to 99.99 parts Others 0 to 10 parts Others include, for example, antifreezing agents and spreading agents.
〔粒剤〕
本発明化合物 0.01〜80部
固体担体 10〜99.99部
その他 0〜10部
その他として、例えば、結合剤、分解防止剤等が挙げられる。
[Granule]
Compound of the present invention 0.01 to 80 parts Solid carrier 10 to 99.99 parts Others 0 to 10 parts Others include, for example, binders, decomposition inhibitors and the like.
〔粉剤〕
本発明化合物 0.01〜30部
固体担体 65〜99.99部
その他 0〜5部
その他として、例えば、ドリフト防止剤、分解防止剤等が挙げられる。
[Dust]
Compound of the present invention 0.01 to 30 parts Solid support 65 to 99.99 parts Others 0 to 5 parts Other examples include a drift inhibitor and a decomposition inhibitor.
次に、本発明化合物を有効成分とする製剤例をより具体的に示すが、本発明はこれらに限定されるものではない。 Next, although the formulation example which uses this invention compound as an active ingredient is shown more concretely, this invention is not limited to these.
なお、以下の配合例において、「部」は質量部を意味する。 In the following formulation examples, “parts” means parts by mass.
〔配合例1〕水和剤
本発明化合物No.1−002a 20部
パイロフィライト 74部
ソルポール5039 4部
(商品名、非イオン性界面活性剤とアニオン性界面活性剤との混合物:東邦化学工業社製)
カープレックス#80D 2部
(商品名、合成含水珪酸:塩野義製薬社製)
以上を均一に混合粉砕して水和剤とする。
[Formulation Example 1] Wetting Agent Compound No. 1-002a 20 parts Pyrophyllite 74 parts Solpol 5039 4 parts (trade name, mixture of nonionic surfactant and anionic surfactant: manufactured by Toho Chemical Industry Co., Ltd.)
Carplex # 80D 2 parts (trade name, synthetic hydrous silicic acid: manufactured by Shionogi & Co.)
The above is uniformly mixed and ground to obtain a wettable powder.
〔配合例2〕乳剤
本発明化合物No.1−002a 5部
キシレン 75部
N−メチルピロリドン 15部
ソルポール2680 5部
(商品名、非イオン性界面活性剤とアニオン性界面活性剤との混合物:東邦化学工業社製)
以上を均一に混合して乳剤とする。
[Formulation Example 2] Emulsion Compound No. 1-002a 5 parts xylene 75 parts N-methylpyrrolidone 15 parts Solpol 2680 5 parts (trade name, mixture of nonionic surfactant and anionic surfactant: manufactured by Toho Chemical Industry Co., Ltd.)
The above is uniformly mixed to obtain an emulsion.
〔配合例3〕懸濁剤
本発明化合物No.1−002a 25部
アグリゾールS−710 10部
(商品名、非イオン性界面活性剤:花王社製)
ルノックス1000C 0.5部
(商品名、アニオン性界面活性剤:東邦化学工業社製)
キサンタンガム 0.2部
水 64.3部
以上を均一に混合した後、湿式粉砕して懸濁剤とする。
[Formulation Example 3] Suspending Agent Compound No. 1-002a 25 parts Agrisol S-710 10 parts (trade name, nonionic surfactant: manufactured by Kao Corporation)
Lnox 1000C 0.5 part (trade name, anionic surfactant: manufactured by Toho Chemical Industry Co., Ltd.)
Xanthan gum 0.2 parts Water 64.3 parts After uniformly mixing the above, wet pulverize to make a suspension.
〔配合例4〕顆粒水和剤
本発明化合物No.1−002a 75部
ハイテノールNE−15 5部
(商品名、アニオン性界面活性剤:第一工業製薬社製)
バニレックスN 10部
(商品名、アニオン性界面活性剤:日本製紙社製)
カープレックス#80D 10部
(商品名、合成含水珪酸:塩野義製薬社製)
以上を均一に混合粉砕した後、少量の水を加えて撹拌混合し、押出式造粒機で造粒し、乾燥して、顆粒水和剤とする。
[Formulation Example 4] Granule wettable powder of the present compound No. 1-002a 75 parts Hytenol NE-15 5 parts (trade name, anionic surfactant: manufactured by Daiichi Kogyo Seiyaku Co., Ltd.)
Vanillex N 10 parts (trade name, anionic surfactant: manufactured by Nippon Paper Industries Co., Ltd.)
Carplex # 80D 10 parts (trade name, synthetic hydrous silicic acid: manufactured by Shionogi & Co.)
After uniformly mixing and pulverizing the above, a small amount of water is added, stirred and mixed, granulated with an extrusion granulator, and dried to obtain a granulated wettable powder.
〔配合例5〕粒剤
本発明化合物No.1−002a 5部
ベントナイト 50部
タルク 45部
以上を均一に混合粉砕した後、少量の水を加えて撹拌混合し、押出式造粒機で造粒し、乾燥して粒剤とする。
[Formulation Example 5] Granules of the present compound No. 1-002a 5 parts Bentonite 50 parts Talc 45 parts After uniformly mixing and pulverizing the above, a small amount of water is added, stirred and mixed, granulated with an extrusion granulator, and dried to form granules.
〔配合例6〕粉剤
本発明化合物No.1−002a 3部
カープレックス#80D 0.5部
(商品名、合成含水珪酸:塩野義製薬社製)
カオリナイト 95部
リン酸ジイソプロピル 1.5部
以上を均一に混合粉砕して粉剤とする。
[Composition Example 6] Powder This invention compound No. 1-002a 3 parts Carplex # 80D 0.5 part (trade name, synthetic hydrous silicic acid: manufactured by Shionogi & Co.)
Kaolinite 95 parts Diisopropyl phosphate 1.5 parts or more uniformly mixed and pulverized to obtain a powder.
使用に際しては、上記製剤を、水で1〜10000倍に希釈、又は希釈せずに直接散布する。 In use, the above preparation is diluted 1 to 10,000 times with water or sprayed directly without dilution.
〔配合例7〕水和剤調製物
本発明化合物No.1−002a 25部
ジイソブチルナフタレンスルホン酸ナトリウム 1部
n−ドデシルベンゼンスルホン酸カルシウム 10部
アルキルアリール ポリグリコールエーテル 12部
ナフタレンスルホン酸ホルマリン縮合物のナトリウム塩 3部
エマルジョン型シリコーン 1部
二酸化ケイ素 3部
カオリン 45部
〔配合例8〕水溶性濃厚剤調製物
本発明化合物No.1−002a 20部
ポリオキシエチレンラウリルエーテル 3部
ジオクチルスルホコハク酸ナトリウム 3.5部
ジメチルスルホキシド 37部
2−プロパノール 36.5部
〔配合例9〕噴霧用液剤
本発明化合物No.1−002a 2部
ジメチルスルホキシド 10部
2−プロパノール 35部
アセトン 53部
〔配合例10〕経皮投与用液剤
本発明化合物No.1−002a 5部
ヘキシレングリコール 50部
イソプロパノール 45部
〔配合例11〕経皮投与用液剤
本発明化合物No.1−002a 5部
プロピレングリコールモノメチルエーテル 50部
ジプロピレングリコール 45部
〔配合例12〕経皮投与(滴下)用液剤
本発明化合物No.1−002a 2部
軽質流動パラフィン 98部
〔配合例13〕経皮投与(滴下)用液剤
本発明化合物No.1−002a 2部
軽質流動パラフィン 58部
オリーブ油 30部
ODO−H 9部
信越シリコーン 1部
また、本発明化合物を農薬として使用する場合には、必要に応じて製剤時又は散布時に、他種の除草剤、各種殺虫剤、殺ダニ剤、殺線虫剤、殺菌剤、植物生長調節剤、共力剤、肥料、土壌改良剤等と混合施用しても良い。
[Formulation 7] wettable powder preparation Compound No. 1-002a 25 parts sodium diisobutyl naphthalenesulfonate 1 part calcium n-dodecylbenzenesulfonate 10 parts alkylaryl polyglycol ether 12 parts sodium salt of naphthalenesulfonic acid formalin condensate 3 parts emulsion type silicone 1 part silicon dioxide 3 parts kaolin 45 Part [Formulation Example 8] Water-soluble thickener preparation 1-002a 20 parts polyoxyethylene lauryl ether 3 parts sodium dioctylsulfosuccinate 3.5 parts dimethyl sulfoxide 37 parts 2-propanol 36.5 parts [Formulation Example 9] Spraying solution 1-002a 2 parts dimethyl sulfoxide 10 parts 2-propanol 35 parts acetone 53 parts [Formulation Example 10] Solution for transdermal administration Compound No. of the present invention 1-002a 5 parts Hexylene glycol 50 parts Isopropanol 45 parts [Formulation Example 11] Solution for transdermal administration Compound No. of the present invention 1-002a 5 parts propylene glycol monomethyl ether 50 parts dipropylene glycol 45 parts [Formulation Example 12] Solution for transdermal administration (dropping) The present compound No. 1 1-002a 2 parts light liquid paraffin 98 parts [Formulation Example 13] Solution for transdermal administration (dropping) The present compound No. 1 1-002a 2 parts light liquid paraffin 58 parts olive oil 30 parts ODO-H 9 parts Shin-Etsu silicone 1 part In addition, when the compound of the present invention is used as an agrochemical, other types of herbicides are used at the time of formulation or spraying as necessary. You may mix and apply with an agent, various insecticides, an acaricide, a nematicide, a fungicide, a plant growth regulator, a synergist, a fertilizer, a soil conditioner, etc.
特に、他の農薬或いは植物ホルモンと混合施用することにより、施用薬量の低減による低コスト化、混合薬剤の相乗作用による殺虫スペクトラムの拡大やより高い有害生物防除効果が期待できる。この際、同時に複数の公知農薬との組み合わせも可能である。 In particular, by applying a mixture with other agricultural chemicals or plant hormones, it is possible to reduce the cost by reducing the amount of applied medicine, to expand the insecticidal spectrum due to the synergistic action of the mixed drugs, and to achieve a higher pest control effect. At this time, a combination with a plurality of known agricultural chemicals is also possible.
本発明化合物と混合使用する農薬の種類としては、例えば、ザ・ペスティサイド・マニュアル(The Pesticide Manual)15版、2009年に記載されている化合物等が挙げられる。具体的にその一般名を例示すれば、次の通りであるが、必ずしもこれらのみに限定されるものではない。 Examples of the type of agricultural chemical used in combination with the compound of the present invention include compounds described in The Pesticide Manual 15th edition, 2009, and the like. Specific examples of the general names are as follows, but are not necessarily limited to these.
殺菌剤:アシベンゾラルーS−メチル(acibenzolar-S-methyl)、アシルアミノベンザミド(acylaminobenzamide)、アシペタックス(acypetacs)、アルジモルフ(aldimorph)、アメトクトラジン(ametoctradin)、アミノピリフェン(aminopyrifen)、アミスルブロム(amisulbrom)、アンバム(amobam)、アムプロピルホス(ampropyfos)、アニラジン(anilazine)、アザコナゾール(azaconazole)、アジチラム(azithiram)、アゾキシストロビン(azoxystrobin)、バリウムポリサルファイド(barium polysulfide)、ベナラキシル(benalaxyl)、ベナラキシル−M(benalaxyl-M)、ベノダニル(benodanil)、ベノミル(benomyl)、ベンキノックス(benquinox)、ベンタルロン(bentaluron)、ベンチアバリカルブ−イソプロピル(benthiavalicarb-isopropyl)、ベンチアゾール(benthiazole)、ベンザマクリル(benzamacril)、ベンズアモルフ(benzamorf)、ベンゾビンジフルピル(benzovindiflupyr)、ベトキサジン(bethoxazine)、ビナパクリル(binapacryl)、ビフェニル(biphenyl)、ビテルタノール(bitertanol)、ブラストサイジン−S(blasticidin-S)、ビキサフェン(bixafen)、ボルドー液(bordeaux mixture)、ボスカリド(boscalid)、ブロムコナゾール(bromuconazole)、ブピリメート(bupirimate)、ブチオベート(buthiobate)、石灰硫黄合剤(calcium polysulfide)、カルシウムポリスルフィド(calcium polysulfide)、キャプタフォール(captafol)、キャプタン(captan)、カルプロパミド(carpropamid)、カルバモルフ(carbamorph)、カルベンダジム(carbendazim)、カルボキシン(carboxin)、カルボン(carvone)、チェシュントミクスチャ(cheshunt mixture)、キノメチオネート(chinomethionat)、クロベンチアゾン(chlobenthiazone)、クロラニフォルメタン(chloraniformethane)、クロラニル(chloranil)、クロルフェナゾール(chlorfenazol)、クロロネブ(chloroneb)、クロロピクリン(chloropicrin)、クロロタロニル(chlorothalonil)、クロロキノックス(chlorquinox)、クロゾリネート(chlozolinate)、クリムバゾール(climbazole)、クロトリマゾール(clotrimazole)、カッパーアセテイト(copper acetate)、塩基性炭酸銅(copper carbonate, basic)、水酸化第二銅(copper hydroxide)、ナフテン酸銅(copper naphthenate)、オレイン酸銅(copper oleate)、塩基性塩化銅(copper oxychloride)、硫酸銅(copper sulfate)、塩基性硫酸銅(copper sulfate, basic)、カッパージンククロメイト(copper zinc chromate)、クフラネブ(cufraneb)、クモキシストロビン(coumoxystrobin)、クプロバム(cuprobam)、シアゾファミド(cyazofamid)、シクラフルアミド(cyclafuramid)、シクロヘキシミド(cycloheximide)、シフルフェナミド(cyflufenamid)、シモキサニル(cymoxanil)、サイペンダゾール(cypendazole)、シプロコナゾール(cyproconazol)、シプロジニル(cyprodinil)、シプロフラム(cyprofuram)、ダゾメット(dazomet)、デバカルブ(debacarb)、デカフェンチン(decafentin)、デハイドロアセテイト(dehydroacetic acid)、ジクロベンチアゾクス(dichlobentiazox)、ジクロフルアニド(dichlofluanid)、ジクロン(dichlone)、ジクロロフェン(dichlorophen)、ジクロゾリン(dichlozoline)、ジクロブトラゾール(diclobutrazol)、ジクロシメット(diclocymet)、ジクロメジン(diclomedine)、ジクロラン(dicloran)など。 Bactericides: acibenzolar-S-methyl, acylaminobenzamide, acypetacs, aldimorph, ametoctradin, aminopyrifen, amisulbrom, Amobam, ampropyfos, anilazine, azaconazole, azithiram, azoxystrobin, barium polysulfide, benalaxyl, benalaxyl-M (Benalaxyl-M), benodanil, benomyl, benquinox, bentaluron, benthiavalicarb-isopropyl, benthiazole, benzacryl zamacril), benzamorf, benzovindiflupyr, bethoxazine, binapacryl, biphenyl, bitertanol, blasticidin-S (blasticidin-S), bixaphene ( bixafen, bordeaux mixture, boscalid, bromuconazole, bupirimate, buthiobate, calcium polysulfide, calcium polysulfide, captaphor (Captafol), captan, carpropamid, carbamorph, carbendazim, carboxin, carvone, cheshunt mixture, chinomethionat Chlobenthiazone, chloraniformethane, chloranil, chlorfenazol, chloroneb, chloropicrin, chlorothalonil, chloroquinox, Chlozolinate, climbazole, clotrimazole, copper acetate, copper carbonate, basic, copper hydroxide, copper naphthenate ( copper naphthenate, copper oleate, basic copper oxychloride, copper sulfate, copper sulfate, basic, copper zinc chromate, cupraneb (Cufraneb), coumoxystrobin, cuprobum (cupro) bam), cyazofamid, cyclafuramid, cycloheximide, cyflufenamid, cymoxanil, cypendazole, cyproconazol, cyprodinil, propromil (Cyprofuram), dazomet, debacarb, decafentin, dehydroacetic acid, diclobentiazox, dichlofluanid, dichlone, dichloro Fen (dichlorophen), dichlozoline, diclobutrazol, diclocymet, diclomedine, dicloran, etc.
殺菌剤(続き):ジエトフェンカルブ(diethofencarb)、ジフェノコナゾール(difenoconazole)、ジフルメトリム(diflumetorim)、ジメチリモール(dimethirimol)、ジメトモルフ(dimethomorph)、ジモキシストロビン(dimoxystrobin)、ジニコナゾール(diniconazole)、ジニコナゾール−M(diniconazole-M)、ジノブトン(dinobuton)、ジノカップ(dinocap)、ジノカップ−4(dinocap-4)、ジノカップ−6(dinocap-6)、ジノクトン(dinocton)、ジノスルフォン(dinosulfon)、ジノテルボン(dinoterbon)、ジフェニルアミン(diphenylamine)、ジピメチトロネ(dipymetitrone)、ジピリチオン(dipyrithione)、ジタリムホス(ditalimfos)、ジチアノン(dithianon)、ドデモルフ酢酸塩(dodemorph-acetate)、ドジン(dodine)、ドラゾクソロン(drazoxolon)、エジフェンホス(edifenphos)、エネストロビン(enestrobin)、エノキサストロビン(enoxastrobin)、エポキシコナゾール(epoxiconazole)、エタコナゾール(etaconazole)、エタボキサム(ethaboxam)、エテム(etem)、エチリモル(ethirimol)、エトキシキン(ethoxyquin)、エトリジアゾール(etridiazole)、ファモキサドン(famoxadone)、フェナリモル(fenarimol)、フェンブコナゾール(fenbuconazole)、フェナミドン(fenamidone)、フェナミノスルフ(fenaminosulf)、フェナミンストロビン(fenaminstrobin)、フェナパニル(fenapanil)、フェンダゾスラム(fendazosulam)、フェンフラム(fenfuram)、フェンヘキサミド(fenhexamid)、フェニトロパン(fenitropan)、フェノキサニル(fenoxanil)、フェンピクロニル(fenpiclonil)、フェンピコキサミド(fenpicoxamid)、フェンプロピジン(fenpropidin)、フェンピラザミン(fenpyrazamine)、フェンプロピモルフ(fenpropimorph)、フェンチン(fentin)、ファーバム(ferbam)、フェリムゾン(ferimzone)、フルアジナム(fluazinam)、フルジオキソニル(fludioxonil)、フルフェノキシストロビン(flufenoxystrobin)、フルインダピル(fluindapyr)、フルメトベル(flumetover)、フルモルフ(flumorph)、フルオピコリド(fluopicolide)、フルオピラム(fluopyram)、フルオルイミド(fluoroimide)、フルオトリマゾール(fluotrimazole)、フルオキサストロビン(fluoxastrobin)、フルキンコナゾール(fluquinconazole)、フルシラゾール(flusilazole)、フルスルファミド(flusulfamide)、フルチアニル(flutianil)、フルトラニル(flutolanil)、フルトリアフォール(flutriafol)、フルキサピロキサド(fluxapyroxad)、フォルペット(folpet)、ホセチル−アルミニウム(fosetyl-aluminium)、フサライド(fthalide)、フベリダゾール(fuberidazole)、フララキシル(furalaxyl)、フラメトピル(furametpyr)、フルカルバニル(furcarbanil)、フルコナゾール(furconazole)、フルコナゾール−シス(furconazole-cis)、フルメシクロックス(furmecyclox)、フルファネート(furphanate)、グリオジン(glyodin)、グリセオフルビン(griseofulvin)、グアザチン(guazatine)、ハラクリネイト(halacrinate)、ヘキサクロロベンゼン(hexachlorobenzene)、ヘキサコナゾール(hexaconazole)、ヘキシルチオフォス(hexylthiofos)、ハイドロキシキノリン サルフェイト(8-hydroxyquinoline sulfate)、ヒメキサゾール(hymexazol)、イマザリル(imazalil)、イミベンコナゾール(imibenconazole)、イミノクタジン−アルベシル酸塩(iminoctadine-albesilate)、イミノクタジン酢酸塩(iminoctadine-triacetate)、インピルフルキサム(inpyrfluxam)、イプコナゾール(ipconazole)、イプフェントリフルコナゾール(ipfentrifluconazole)、イプロベンホス(iprobenfos)、イプロジオン(iprodione)、イプロバリカルブ(iprovalicarb)、イソフェタミド(isofetamid)、イソフルシプラム(isoflucypram)、イソプロチオラン(isoprothiolane)、イソピラザム(isopyrazam)、イソチアニル(isotianil)、イソバレジオン(isovaledione)など。 Bactericides (continued): dietofencarb, difenoconazole, diflumetorim, dimethirimol, dimethomorph, dimoxystrobin, diiconazole-diniconazole-diniconazole-M M), dinobuton, dinocap, dinocap-4, dinocap-6, dinocton, dinosulfon, dinoterbon, diphenylamine ), Dipymetitrone, dipyrithione, ditalimfos, dithianon, dodemorph-acetate, dodine, drazoxolon, edifenphos, nesifenphos Robin (enestrobin), enoxastrobin, epoxiconazole, etaconazole, ethaboxam, etem, etirimol, ethoxyquin, etridiazole, famoxadone (Famoxadone), fenarimol, fenbuconazole, fenamidone, fenaminosulf, fenaminoinstrobin, fenapanil, fendazosulam, fendazosulam, fenfuram, fenfuram Fenhexamid, fenitropan, fenoxanil, fenpiclonil, fenpicoxamid, fenpropidin, fenpyrazamine fenpyrazamine, fenpropimorph, fentin, ferbam, ferimzone, fluazinam, fludioxonil, flufenoxystrobin, fluindapyr, fluindapyr flumetover, flumorph, fluopicolide, fluopyram, fluorimide, fluotrimazole, fluoxastrobin, fluquinconazole, flusilazole, Flusulfamide, flutianil, flutolanil, flutriafol, fluxapyroxad, folpet, fosetyl-alumini Fosetyl-aluminium, fthalide, fuberidazole, furaxaxyl, furametpyr, furcarbanil, furconazole, furconazole, fluconazole-cis, flumeciclos ( furmecyclox, furphanate, glyodin, griseofulvin, guazatine, halacrinate, hexachlorobenzene, hexaconazole, hexylthiofos, hydroxyquinoline 8-hydroxyquinoline sulfate, hymexazol, imazalil, imibenconazole, iminoctadine-albesilate, iminocta Acetate (iminoctadine-triacetate), impylfluxam (inpyrfluxam), ipconazole, ipfentrifluconazole, iprobenfos, iprodione, iprovalicarb (is), isetamid, isetamid Isoflucypram, isoprothiolane, isopyrazam, isothianil, isovaledione and the like.
殺菌剤(続き):カスガマイシン(kasugamycin)、クレソキシム−メチル(kresoxim-methyl)、ラミナリン(laminarin)、マンカッパー(mancopper)、マンコゼブ(mancozeb)、マンデストロビン(mandestrobin)、マンジプロパミド(mandipropamid)、マンネブ(maneb)、メベニル(mebenil)、メカルビンジド(mecarbinzid)、メフェントリフルコナゾール(mefentrifluconazole)、メパニピリム(mepanipyrim)、メプチルジノカップ(meptyldinocap)、メプロニル(mepronil)、メタラキシル(metalaxyl)、メタラキシル−M(metalaxyl-M)、メタム(metam)、メタゾキソロン(metazoxolon)、メトコナゾール(metconazole)、メタスルホカルブ(methasulfocarb)、メトフロキサム(methfuroxam)、メチルイソチオシアネイト(methyl isothiocyanate)、メチラム(metiram)、メトミノストロビン(metominostrobin)、メトラフェノン(metrafenone)、メトスルフォバックス(metsulfovax)、ミルネブ(milneb)、ミクロブタニル(myclobutanil)、ミクロゾリン(myclozolin)、ナバム(nabam)、ナタマイシン(natamycin)、ニッケルビス(ジメチルジチオカーバメート)(nickel bis(dimethyldithiocarbamate))、ニトロスチレン(nitrostyrene)、ニトロタール−イソプロピル(nitrothal-isopropyl)、ヌアリモル(nuarimol)、オーシーエイチ(OCH)、オクチリノン(octhilinone)、オフレース(ofurace)、オリサストロビン(orysastrobin)、オキサチアピプロリン(oxathiapiprolin)、オキサジキシル(oxadixyl)、8−ヒドロキシキノリン銅(oxine copper)、オキシカルボキシン(oxycarboxin)、オキスポコナゾールフマル酸塩(oxpoconazole fumarate)、ペフラゾエート(pefurzoate)、ペンコナゾール(penconazole)、ペンフルフェン(penflufen)、ペンシクロン(pencycuron)、ペンチオピラド(penthiopyrad)、オルソフェニルフェノール(o-phenylphenol)、フォスジフェン(phosdiphen)、ピカルブトラゾクス(picarbutrazox)、ピコキシストロビン(picoxystrobin)、ピペラリン(piperalin)、ポリカーバメート(polycarbamate)、ポリオキシン(polyoxins)、ポリオキシン−D(polyoxorim)、ポタシウムアザイド(potassium azide)、炭酸水素カリウム(potassium hydrogen carbonate)、プロキナジド(proquinazid)、プロベナゾール(probenazole)、プロクロラズ(prochloraz)、プロシミドン(procymidone)、プロパモカルブ塩酸塩(propamocarb hydrochloride)、プロピコナゾール(propiconazole)、プロピネブ(propineb)、プロチオカルブ(prothiocarb)、プロチオコナゾール(prothioconazole)、ピジフルメトフェン(pydiflumetofen)、ピラカルボリド(pyracarbolid)、ピラクロストロビン(pyraclostrobin)、ピラメトストロビン(pyrametostrobin)、ピラオキシストロビン(pyraoxystrobin)、ピラジフルミド(pyraziflumid)、ピラゾホス(pyrazophos)、ピリベンカルブメチル(pyribencarb-methyl)、ピリジニトリル(pyridinitril)、ピリフェノックス(pyrifenox)、ピリメタニル(pyrimethanil)、ピリミノストロビン(pyriminostrobin)、ピリモルフ(pyrimorph)、ピリオフェノン(pyriofenone)、ピリソキサゾール(pyrisoxazole)、ピロキロン(pyroquilon)、ピロキシクロル(pyroxychlor)、ピロキシフル(pyroxyfur)、キノメチオネート(quinomethionate)、キノフメリン(quinofumelin)、キノキシフェン(quinoxyfen)、キントゼン(quintozene)、キナセトール・スルフェート(quinacetol-sulfate)、キナザミド(quinazamid)、キンコナゾール(quinconazole)、ラベンザゾール(rabenzazole)及びバチルス ズブチリス(Bacillus subtilis, Strain:D747, FZB24, GBO3, HAI0404, MBI600, QST713, Y1336等)など。 Bactericides (continued): kasugamycin, kresoxim-methyl, laminarin, mancopper, mancozeb, mandestrobin, mandipropamid, mannepropamid maneb, mebenil, mecarbinzid, mefentrifluconazole, mepanipyrim, meptyldinocap, mepronil, metalaxyl, metalaxyl-M (metalaxyl-M) M), metam, metazoxolon, metconazole, methasulfocarb, metfuroxam, methyl isothiocyanate, metiram, metinominostrone obin, metrafenone, metsulfovax, milneb, microbutanil, microzoline, nabam, natamycin, nickel bis (dimethyldithiocarbamate) (Dimethyldithiocarbamate), nitrostyrene, nitrothal-isopropyl, nuarimol, OCH, octhilinone, offurace, orysastrobin, oxathapi Proline (oxathiapiprolin), oxadixyl, oxine copper, oxycarboxin, oxpoconazole fumarate, pepozoate, penconazole nazole), penflufen, penencycuron, penthiopyrad, orthophenylphenol, phosdiphen, phosdiphen, picarbutrazox, picoxystrobin, piperaline ( piperalin, polycarbamate, polyoxins, polyoxin-D (polyoxorim), potassium azide, potassium hydrogen carbonate, proquinazid, probenazole, prochloraz ( prochloraz, procymidone, propamocarb hydrochloride, propiconazole, propineb, prothiocarb, prothioconazole, pidiflume Pydiflumetofen, pyracarbolid, pyraclostrobin, pyrametostrobin, pyraoxystrobin, pyraziflumid, pyrazophos, pyribencarb-methyl ), Pyridinitril, Pyrifenox, Pyrimethanil, Pyriminostrobin, Pyrimorph, Pyriofenone, Pyroxoxazole, Pyroxilone, roxychlor ), Pyroxyfur, quinomethionate, quinofumelin, quinoxyfen, quintozene, quinacetol-sulfate, ki Zamido (quinazamid), Kinkonazoru (quinconazole), Rabenzazoru (Rabenzazole) and Bacillus subtilis (Bacillus subtilis, Strain: D747, FZB24, GBO3, HAI0404, MBI600, QST713, Y1336, or the like).
殺菌剤(続き):セダキサン(sedaxane)、アジ化ナトリウム(sodium azide)、炭酸水素ナトリウム(sodium hydrogen carbonate)、次亜塩素酸ナトリウム(sodium hypochlorite)、硫黄(sulfur)、スピロキサミン(spiroxamine)、サリチルアニリド(salycylanilide)、シルチオファム(silthiofam)、シメコナゾール(simeconazole)、テブコナゾール(tebuconazole)、テブフロキン(tebufloquin)、テクナゼン(tecnazene)、テコラム(tecoram)、テトラコナゾール(tetraconazole)、チアベンダゾール(thiabendazole)、チアジフルオール(thiadifluor)、チシオフェン(thicyofen)、チフルザミド(thifluzamide)、チオクロルフェンフィム(thiochlorfenphim)、チオファネート(thiophanate)、チオファネート−メチル(thiophanate-methyl)、チオキノックス(thioquinox)、チウラム(thiram)、チアジニル(tiadinil)、チオキシミド(tioxymid)、トルクロホス−メチル(tolclofos-methyl)、トルプロカルブ(tolprocarb)、トリルフルアニド(tolylfluanid)、トリアジメホン(triadimefon)、トリアジメノール(toriadimenol)、トリアミフォス(triamiphos)、トリアリモル(triarimol)、トリアゾキシド(triazoxide)、トリアズブチル(triazbutil)、トリブチルチンオキサイド(tributyltin oxide)、トリクラミド(trichlamide)、トリシクラゾール(tricyclazole)、トリデモルフ(tridemorph)、トリフロキシストロビン(trifloxystrobin)、トリフルミゾール(triflumizole)、トリホリン(triforine)、トリクロピリカルブ(triclopyricarb)、トリチコナゾール(triticonazole)、バリダマイシン(validamycin)、バリフェナレート(valifenalate)、ビンクロゾリン(vinclozolin)、ザリルアミド(zarilamide)、硫酸亜鉛(zinc sulfate)、ジネブ(zineb)、ジラム(ziram)、ゾキサミド(zoxamide)、シイタケ菌糸体抽出物、シイタケ子実体抽出物、NF−180(試験名)、MIF−1002(試験名)、S-2399(試験名)及びAKD−5195(試験名)など。 Bactericides (continued): sedaxane, sodium azide, sodium hydrogen carbonate, sodium hypochlorite, sulfur, spiroxamine, salicylanilide (Salycylanilide), silthiofam, simeconazole, tebuconazole, tebufloquin, tecnazene, tecoram, tetraconazole, thiabendazole, thiabendazole thiadifluor, thicyofen, thifluzamide, thiochlorfenphim, thiophanate, thiophanate-methyl, thioquinox, thiram, thiadi Tiadinil, tioxymid, tolclofos-methyl, tolprocarb, tolylfluanid, triadimefon, triadimenol, triamiphos, triamiphos (triamiphos) triarimol, triazoxide, triazbutil, tributyltin oxide, trichlamide, tricyclazole, triridemorph, trifloxystrobin, triflumizole , Triforine, triclopyricarb, triticonazole, validamycin, valifenalate, vinclozolin, zarylamide (z) arilamide, zinc sulfate, zineb, ziram, zoxamide, shiitake mycelium extract, shiitake fruiting body extract, NF-180 (test name), MIF-1002 (test) Name), S-2399 (test name) and AKD-5195 (test name).
殺バクテリア剤:ベンザルコニウムクロライド(benzalkonium chloride)、ビチオノール(bithionol)、ブロノポール(bronopol)、クレゾール(cresol)、ホルムアルデヒド(formaldehyde)、ニトラピリン(nitrapyrin)、オキソリニックアシド(oxolinic acid)、オキシテトラサイクリン(oxyterracycline)、ストレプトマイシン(streptomycin)及びテクロフタラム(tecloftalam)など。 Bactericides: benzalkonium chloride, bithionol, bronopol, cresol, formaldehyde, nitrapyrin, oxolinic acid, oxyterracycline , Streptomycin and tecloftalam.
殺線虫剤:アルドキシカルブ(aldoxycarb)、ベンクロチアズ(benclothiaz)、カズサホス(cadusafos)、デービーシーピー(DBCP)、ジクロフェンチオン(dichlofenthion)、デーエスピー(DSP)、エトプロホス(ethoprophos)、フェナミホス(fenamiphos)、フェンスルホチオン(fensulfothion)、フルアザインドリジン(fluazaindolizine)、フルエンスルホン(fluensulfone)、ホスチアゼート(fosthiazate)、フォスチエタン(fosthietan)、イミシアホス(imicyafos)、イサミドホス(isamidofos)、イサゾホス(isazofos)、オキサミル(oxamyl)、チオキサザフェン(thiaxazafen)、チオナジン(thionazin)、チオキサザフェン(tioxazafen)、BYI−1921(試験名)及びMAI−08015(試験名)など。 Nematicides: aldoxycarb, benclothiaz, cadusafos, davylope (DBCP), dichlofenthion, dsp (DSP), etoprophos, fenamiphos, fensulfothion (Fensulfothion), fluazaindolizine, fluenesulfone, fosthiazate, fosthietan, imisiafos, isamidofos, isazofos, oxamyl, oxamyl, thiaxazafen), thionazin, tioxazafen, BYI-1921 (test name) and MAI-08015 (test name).
殺ダニ剤:アセキノシル(acequinocyl)、アクリナトリン(acrinathrin)、アシノナピル(acynonapyr)、アミドフルメット(amidoflumet)、アミトラズ(amitraz)、アゾシクロチン(azocyclotin)、BCI−033(試験名)、ベンゾキシメート(benzoximate)、ビフェナゼート(bifenazate)、ブロモプロピレート(bromopropylate)、チノメチオネート(chinomethionat)、クロロベンジラート(chlorobezilate)、クロフェンテジン(clofentezine)、シエノピラフェン(cyenopyrafen)、シフルメトフェン(cyflumetofen)、サイヘキサチン(cyhexatine)、ジコホール(dicofol)、ジエノクロル(dienochlor)、ジフロビダジン(diflovidazin)、デーエヌオーシー(DNOC)、エトキサゾール(etoxazole)、フェナザキン(fenazaquin)、フェンブタチンオキシド(fenbutatin oxide)、フェノチオカルブ(fenothiocarb)、フェンプロパトリン(fenpropathrin)、フェンピロキシメート(fenpyroximate)、フルアクリピリム(fluacrypyrim)、ハルフェンプロックス(halfenprox)、ヘキシチアゾクス(hexythiazox)、ミルベメクチン(milbemectin)、プロパルギット(propargite)、ピフルブミド(pyflubumide)、ピリダベン(pyridaben)、ピリミジフェン(pyrimidifen)、S−1870(試験名)、スピロジクロフェン(spirodiclofen)、スピロメシフェン(spyromesifen)、CL900167(試験名)、テブフェンピラド(tebufenpyrad)及びNA−89(試験名)など。 Acaricide: acequinocyl, acrinathrin, acynonapyr, amidoflumet, amitraz, azocyclotin, BCI-033 (study name), benzoximate, Bifenazate, bromopropylate, chinomethionat, chlorobezilate, clofentezine, cyenopyrafen, cyflumetofen, cyhexatine, olhol , Dienochlor, diflovidazin, DNOC, etoxazole, fenazaquin, fenbutatin oxide, phenothiocarb ( fenothiocarb, fenpropathrin, fenpyroximate, fluacrypyrim, fluacrypyrim, halfenprox, hexythiazox, milbemectin, propargite, piflumide (pyflubum) ), Pyrimidifen, S-1870 (test name), spirodiclofen, spyromesifen, CL900187 (test name), tebufenpyrad and NA-89 (test name).
殺虫剤:アバメクチン(abamectin)、アセフェート(acephate)、アセタミピリド(acetamipirid)、アフィドピロペン(afidopyropen)、アフォクソラネル(afoxolaner)、アラニカルブ(alanycarb)、アルジカルブ(aldicarb)、アレスリン(allethrin)、アザメチホス(azamethiphos)、アジンホス−エチル(azinphos-ethyl)、アジンホス−メチル(azinphos-methyl)、バチルスチューリンゲンシス(bacillus thuringiensis)、ベンダイオカルブ(bendiocarb)、ベンフルトリン(benfluthrin)、ベンフラカルブ(benfuracarb)、ベンスルタップ(bensultap)、、ベンズピリモキサン(benzpyrimoxan)、ビフェントリン(bifenthrin)、ビオアレスリン(bioallethrin)、ビオレスメトリン(bioresmethrin)、ビストリフルロン(bistrifluron)、ブロフラニリド(broflanilide)、ブプロフェジン(buprofezin)、ブトカルボキシム(butocarboxim)、カルバリル(carbaryl)、カルボフラン(carbofuran)、カルボスルファン(carbosulfan)、カルタップ(cartap)、クロルアントラニリプロール(chlorantraniliprole)、クロルエトキシホス(chlorethxyfos)、クロルフェナピル(chlorfenapyr)、クロルフェンビンホス(chlorfenvinphos)、クロルフルアズロン(chlorfluazuron)、クロルメホス(chlormephos)、クロロプラレスリン(chloroprallethrin)、クロルピリホス(chlorpyrifos)、クロピリホス−メチル(chlorpyrifos-methyl)、クロマフェノジド(chromafenozide)、クロチアニジン(clothianidin)、シアノホス(cyanophos)、シアントラニリプロール(cyantraniliprole)、シクラニリプロール(cyclaniliprole)、シクロプロトリン(cycloprothrin)、シフルメトフェン(cyflumetofen)、シフルトリン(cyfluthrin)、ベータ−シフルトリン(beta-cyfluthrin)、シハロジアミド(cyhalodiamide)、シハロトリン(cyhalothrin)、ガンマ−シハロトリン(gamma-cyhalothrin)、ラムダ−シハロトリン(lambda-cyhalothrin)、シペルメトリン(cypermethrin)、アルファ−シペルメトリン(alpha-cypermethrin)、ベータ−シペルメトリン(beta-cypermethrin)、ゼタ−シペルメトリン(zeta-cypermethrin)、シフェノトリン(cyphenothrin)、シロマジン(cyromazine)、デルタメトリン(deltamethrin)、ジアクロデン(diacloden)、ジアフェンチウロン(diafenthiuron)、ダイアジノン(diazinon)、ジクロロメゾチアズ(dicloromezotiaz)、ジクロルボス(dichlorvos)、ジフルベンズロン(diflubenzuron)、ジメフルトリン(dimefluthrin)、ジメチルビンホス(dimethylvinphos)、ジノテフラン(dinotefuran)、ジオフェノラン(diofenolan)、ジスルフォトン(disulfoton)、ジメトエート(dimethoate)、エマメクチンベンゾエート(emamectin-benzoate)、エンペントリン(empenthrin)、エンドスルファン(endosulfan)、アルファ−エンドスルファン(alpha-endosulfan)、イーピーエヌ(EPN)、エスフェンバレレート(esfenvalerate)、エチオフェンカルブ(ethiofencarb)、エチプロール(ethiprole)、エトフェンプロックス(etofenprox)、エトリムホス(etrimfos)、フェニトロチオン(fenitrothion)、フェノブカルブ(fenobucarb)、フェノキシカルブ(fenoxycarb)、フェンプロパトリン(fenpropathrin)、フェンチオン(fenthion)、フェンバレレート(fenvalerate)、フィプロニル(fipronil)、フロニカミド(flonicamid)、フルアズロン(fluazuron)、フルベンジアミド(flubendiamide)、フルシクロクスロン(flucycloxuron)、フルシトリネート(flucythrinate)、フルフェネリム(flufenerim)、フルフェノクスロン(flufenoxuron)、フルフェンプロックス(flufenprox)、フルメトリン(flumethrin)、フルピリミン(flupyrimin)、フルララネル(fluralaner)、フルバリネート(fluvalinate)、タウ−フルバリネート(tau-fluvalinate)、ホノホス(fonophos)、ホルメタネート(formetanate)、フォルモチオン(formothion)、フラチオカルブ(furathiocarb)、フルフィプロール(flufiprole)、フルヘキサホン(fluhexafon)、フルピラジフロン(flupyradifurone)、フロメトキン(flometoquin)など。 Insecticides: abamectin, acephate, acetamipirid, afidopyropen, afoxolaner, alanycarb, aldicarb, alesphos, amethphos, azaphos Ethyl (azinphos-ethyl), azinphos-methyl, bacillus thuringiensis, bendiocarb, benfluthrin, benfuracarb, bensultap, benzpyrimoxy Sun (benzpyrimoxan), bifenthrin, bioallethrin, bioresmethrin, bistrifluron, broflanilide, buprofezin, Tocarboxim, carbaryl, carbofuran, carbofuran, carbosulfan, cartap, chlorantraniliprole, chlorethxyfos, chlorfenapyr, chlorfenapyr Chlorfenvinphos, chlorfluazuron, chlormephos, chloroprallethrin, chlorpyrifos, chlorpyrifos-methyl, chromafenozide, clothianidin, clothianidin Cyanophos, cyantraniliprole, cyclaniliprole, cycloprothrin, cyflumetofen, cyfluthrin, Beta-cyfluthrin, cyhalodiamide, cyhalothrin, gamma-cyhalothrin, lambda-cyhalothrin, cypermethrin, alpha-cypermethrin (alpha-) cypermethrin), beta-cypermethrin, beta-cypermethrin, zeta-cypermethrin, cyphenothrin, cyromazine, deltamethrin, diacloden, diafenthiuron ), Diazinon, dicloromezotiaz, dichlorvos, diflubenzuron, dimefluthrin, dimethylvinphos, dinotefuran, diofenol an), disulfoton, dimethoate, emamectin-benzoate, empenthrin, endosulfan, alpha-endosulfan, EPN, esfenvalerate ), Etiofencarb, ethiprole, etofenprox, etrimfos, fenitrothion, fenobucarb, phenoxycarb, fenpropathrin, fenthion ), Fenvalerate, fipronil, flonicamid, fluazuron, flubendiamide, flucycloxuron , Flucythrinate, flufenerim, flufenoxuron, flufenprox, flumethrin, flupyrinin, fluralaner, fluvalinate, tau Fluvalinate, tono-fluvalinate, fonophos, formetanate, formothion, furthiothiocarb, flufiprole, fluhexafon, flupyradifurone, flometoquin, flometoquin, etc. .
殺虫剤(続き):ハロフェノジド(halofenozide)、ヘプタフルトリン(heptafluthrin)、ヘキサフルムロン(hexaflumuron)、ヒドラメチルノン(hydramethylnon)、イミダクロプリド(imidacloprid)、イミプロトリン(imiprothrin)、イソフェンホス(isofenphos)、インドキサカルブ(indoxacarb)、インドキサカルブ−MP(indoxacarb-MP)、イソプロカルブ(isoprocarb)、イソキサチオン(isoxathion)、カッパ−ビフェントリン(kappa-bifenthrin)、カッパ−テフルトリン(kappa-tefluthrin)、レピメクチン(lepimectin)、ルフェヌロン(lufenuron)、マラチオン(malathion)、メペルフルスリン(meperfluthrin)、メタフルミゾン(metaflumizone)、メタルデヒド(metaldehyde)、メタミドホス(methamidophos)、メチダチオン(methidathion)、メタクリホス(methacrifos)、メタルカルブ(metalcarb)、メソミル(methomyl)、メソプレン(methoprene)、メトキシクロル(methoxychlor)、メトキシフェノジド(methoxyfenozide)、臭化メチル(methyl bromide)、メトフルトリン(metofluthrin)、イプシロン−メトフルトリン(epsilon-metofluthrin)、モムフルオロトリン(momfluorothrin)、イプシロン−モムフルオロトリン(epsilon-momfluorothrin)、モノクロトホス(monocrotophos)、ムスカルア(muscalure)、ニテンピラム(nitenpyram)、ノバルロン(novaluron)、ノビフルムロン(noviflumuron)、オメトエート(omethoate)、オキシデメトン−メチル(oxydemeton-methyl)、オキシデプロホス(oxydeprofos)、パラチオン(parathion)、パラチオン−メチル(parathion-methyl)、ペンタクロロフェノール(pentachlorophenol)、ペルメトリン(permethrin)、フェノトリン(phenothrin)、フェントエート(phenthoate)、ホキシム(phoxim)、ホレート(phorate)、ホサロン(phosalone)、ホスメット(phosmet)、ホスファミドン(phosphamidon)、ピリミカーブ(pirimicarb)、ピリミホス−メチル(pirimiphos-methyl)、プロフェノホス(profenofos)、プロフルトリン(profluthrin)、プロチオホス(prothiofos)、プロパホス(propaphos)、プロトリフェンブト(protrifenbute)、ピメトロジン(pymetrozine)、ピラクロホス(pyraclofos)、ピレトリン(pyrethrins)、ピリダリル(pyridalyl)、ピリフルキナゾン(pyrifluquinazon)、ピリプロール(pyriprole)、ピラフルプロール(pyrafluprole)、ピリプロキシフェン(pyriproxyfen)、レスメトリン(resmethrin)、ロテノン(rotenone)、SI−0405(試験名)、スルプロホス(sulprofos)、シラフルオフェン(silafluofen)、スピネトラム(spinetoram)、スピノサド(spinosad)、スピロメシフェン(spiromesifen)、スピロテトラマート(spirotetramat)、スピロピジオン(spiropidion)、スルホキサフロル(sulfoxaflor)、スルホテップ(sulfotep)、SYJ−159(試験名)、テブフェノジド(tebfenozide)、テフルベンズロン(teflubenzuron)、テフルトリン(tefluthorin)、テルブホス(terbufos)、テトラクロロビンホス(tetrachlorvinphos)、テトラメトリン(tetramethrin)、d−T−80−フタルスリン(d-tetramethrin)、テトラメチルフルスリン(tetramethylfluthrin)、テトラニリプロール(tetraniliprole)、チアクロプリド(thiacloprid)、チオシクラム(thiocyclam)、チオジカルブ(thiodicarb)、チアメトキサム(thiamethoxam)、チオファノックス(thiofanox)、チオメトン(thiometon)、トルフェンピラド(tolfenpyrad)、トラロメトリン(tralomethrin)、トランスフルトリン(transfluthrin)、トリアザメート(triazamate)、トリクロルホン(trichlorfon)、トリアズロン(triazuron)、トリフルメゾピリム(triflumezopyrim)、トリフルムロン(triflumuron)、バミドチオン(vamidothion)、フルキサメタミド(fluxametamide)、MIE−1209(試験名)及びME5382(試験名)など。 Insecticides (continued): halofenozide, heptafluthrin, hexaflumuron, hydramethylnon, imidacloprid, imiprothrin, isofenphos, indoxacarb (Indoxacarb), indoxacarb-MP (indoxacarb-MP), isoprocarb (isoprocarb), isoxathion (isoxathion), kappa-bifenthrin, kappa-tefluthrin, lepimectin (lepimectin), lufenuron ( lufenuron, malathion, meperfluthrin, metaflumizone, metalaldehyde, metamidophos, methidathion, methacrifos, metalcarb b), methomyl, methoprene, methoxychlor, methoxyfenozide, methyl bromide, metofluthrin, epsilon-metofluthrin, momfluorothrin ), Epsilon-momfluorothrin, monocrotophos, muscalure, nitenpyram, novaluron, noviflumuron, omethoate, oxydemeton-methyl (oxydemeton-methyl) methyl), oxydeprofos, parathion, parathion-methyl, pentachlorophenol, permethrin, phenothrin, phenothrin Phenthoate, phoxim, phorate, phosalone, phosmet, phosphamidon, pirimicarb, pirimiphos-methyl, profenofos, profluthrin (Profluthrin), prothiofos, propaphos, protrifenbute, pymetrozine, pyraclofos, pyrethrins, pyridalyl, pyrifluquinapy, prorifenquine ), Pyrafluprole, pyriproxyfen, resmethrin, rotenone, SI-0405 (study name), sulprofos, silafluofen, spinetoram (spine) toram), spinosad, spiromesifen, spirotetramat, spiropidion, sulfoxaflor, sulfoxap, SYJ-159 (test name), tebfenozide, Teflubenzuron, tefluthorin, terbufos, tetrachlorovinphos, tetramethrin, d-T-80-phthalthrin, tetramethylfluthrin, tetramethylfluthrin, tetramethylfluthrin, tetramethylfluthrin, tetramethylfluthrin, tetramethylfluthrin, tetramethylfluthrin, tetramethylfluthrin Niliprol (tetraniliprole), thiacloprid, thiocyclam, thiodicarb, thiamethoxam, thiofanox, thiometon, tolfenpyrad, Tralomethrin, transfluthrin, triazamate, trichlorfon, triazuron, triflumezopyrim, triflumuron, vamidion, MI -1209 (test name) and ME5382 (test name).
以下に、本発明化合物の合成例、試験例を実施例として述べることにより、本発明をさらに詳しく説明するが、本発明はこれらによって限定されるものではない。 Hereinafter, the present invention will be described in more detail by describing synthesis examples and test examples of the compounds of the present invention as examples, but the present invention is not limited thereto.
合成例に記載した中圧分取液体クロマトグラフィーは、山善株式会社中圧分取装置;YFLC−Wprep(流速18ml/min、シリカゲル40μmのカラム)を使用した。 The medium pressure preparative liquid chromatography described in the synthesis example used an intermediate pressure preparative apparatus (Yamazen Co., Ltd.); YFLC-Wprep (flow rate 18 ml / min, silica gel 40 μm column).
また、以下に記載のプロトン核磁気共鳴スペクトル(以下、1H−NMRと記載する。)のケミカルシフト値は、基準物質としてMe4Si(テトラメチルシラン)を用い、重クロロホルム溶媒中で、300MHz(機種;JNM−ECX300又はJNM−ECP300、JEOL社製)にて測定した。尚、1H−NMRのケミカルシフト値における記号は、下記の意味を表す。
s:シングレット、d:ダブレット、dd:ダブルダブレット、t:トリプレット、q:カルテット、m:マルチプレット、brs:ブロードシングレット
合成例1:N−(2,2−ジフルオロベンゾ[d][1,3]ジオキソール−5−イル)−3−(エチルスルホニル)−7−(トリフルオロメチル)イミダゾ[1,2−a]ピリジン−2−カルボキサミド(本発明化合物No.5-001a)の合成
5−アミノ−2,2−ジフルオロ−1,3−ベンゾ[d][1,3]ジオキソール83mg、トリエチルアミン391mg及びジクロロメタン1.0mlの混合溶液に、室温にて、3−(エチルスルホニル)−7−(トリフルオロメチル)イミダゾ[1,2−a]ピリジン−2−カルボニルクロリド110mgとジクロロメタン1.0mlとの混合溶液を添加した。添加終了後、該反応混合物を室温にて18時間撹拌した。反応終了後、該反応混合物に飽和炭酸水素ナトリウム5mlを加え、クロロホルムにて抽出(5ml×2)した。得られた有機層を無水硫酸ナトリウムで脱水・乾燥した後、減圧下にて溶媒を留去した。得られた残留物をn−ヘキサン−酢酸エチル[90:10〜20:80(体積比、以下同じである。)のグラジエント]にて溶出する中圧分取液体クロマトグラフィーにて精製し、目的物120mgを淡黄色固体として得た。
融点:197〜200℃
1H-NMR(CDCl3) :δ9.55(d, J=7.5Hz, 1H), 9.35(s, 1H), 8.05(s, 1H), 7.85(d, J=1.8Hz, 1H), 7.30-7.20(m, 2H), 7.05(d, J=8.7Hz, 1H), 3.96(q, J=7.2Hz, 2H), 1.37(t, J=7.2Hz, 3H).
合成例2:N−(2,2−ジフルオロベンゾ[d][1,3]ジオキソール−5−イル)−3−(エチルスルホニル)−N−メチル−7−(トリフルオロメチル)イミダゾ[1,2−a]ピリジン−2−カルボキサミド(本発明化合物No.5-002a)の合成
60質量%水素化ナトリウム(鉱油中に分散)10mg及びTHF0.5mlの混合溶液に、室温にて上記合成例1に記載の方法により合成したN−(2,2−ジフルオロベンゾ[d][1,3]ジオキソール−5−イル)−3−(エチルスルホニル)−7−(トリフルオロメチル)イミダゾ[1,2−a]ピリジン−2−カルボキサミド108mgのTHF1.2ml溶液を添加した。添加終了後、該反応混合物を室温にて1時間撹拌した。撹拌終了後、該反応混合物に室温にてヨウ化メチル35mgを添加した。添加終了後、該反応混合物を室温にて1時間撹拌した。反応終了後、該反応混合物に水5mlを添加し、酢酸エチルにて抽出(5ml×3)した。得られた有機層を、飽和食塩水、次いで無水硫酸ナトリウムの順で脱水・乾燥した後、減圧下にて溶媒を留去した。得られた残留物を、n−ヘキサン−酢酸エチル(95:5〜30:70のグラジエント)にて溶出する中圧分取液体クロマトグラフィーにて精製し、目的物42mgを無色油状物として得た。
1H-NMR(CDCl3) :δ9.03(d, J=7.2Hz, 1H), 7.86(s, 1H), 7.25-7.05(m, 3H), 6.86(d, J=8.4Hz, 1H), 3.60(q, J=7.5Hz, 2H), 3.49(s, 3H), 1.36(t, J=7.5Hz, 3H).
合成例3:3−(エチルチオ)−N−メチル−7−(トリフルオロメチル)−N−[5−(トリフルオロメチル)−1,3,4−チアジアゾール−2−イル]イミダゾ[1,2−a]ピリジン−2−カルボキサミド(本発明化合物No.4-001b)の合成
3−(エチルチオ)−7−(トリフルオロメチル)イミダゾ[1,2−a]ピリジン−2−カルボン酸300mg及びジクロロメタン4mlの混合溶液に、氷冷下にてオキサリルクロリド377mg及びN,N−ジメチルホルムアミド20mgを順次添加した。添加終了後、該反応混合物を室温にて30分間撹拌した。反応終了後、減圧下にて該反応混合物から溶媒を留去して、3−(エチルスルホニル)−7−(トリフルオロメチル)イミダゾ[1,2−a]ピリジン−2−カルボニルクロリドを得た。ジクロロメタン3mlに溶解した3−(エチルスルホニル)−7−(トリフルオロメチル)イミダゾ[1,2−a]ピリジン−2−カルボニルクロリドを、2−メチルアミノ−5−(トリフルオロメチル)−1,3,4−チアジアゾール208mg及びピリジン3mlの混合溶液に、氷冷下にて添加した。添加終了後、該反応混合物を室温にて1時間撹拌した。反応終了後、減圧下にて該反応混合物の溶媒を留去した。得られた残留物に水5mlを添加し、酢酸エチルにて抽出(5ml×3)した。得られた有機層を1mol/L塩酸5mlで洗浄し、飽和食塩水、次いで無水硫酸ナトリウムの順で脱水・乾燥した後、減圧下にて溶媒を留去した。得られた残留物を、n−ヘキサン−酢酸エチル(95:5〜30:70のグラジエント)にて溶出するアルミナカラムクロマトグラフィーにて精製した。更に得られた固体をn−ヘキサン−酢酸エチル(95:5〜30:70のグラジエント)にて溶出する中圧分取液体クロマトグラフィーにて精製し、目的物525mgを淡黄色固体として得た。
融点:84〜86℃
1H-NMR(CDCl3) :δ8.70(d, J=7.5Hz, 1H), 8.03(s, 1H), 7.23(d, J=7.5Hz, 1H), 4.04(s, 3H), 2.98(q, J=7.5Hz, 2H), 1.23(t, J=7.5Hz, 3H).
合成例4:3−(エチルスルフィニル)−N−メチル−7−(トリフルオロメチル)−N−[5−(トリフルオロメチル)−1,3,4−チアジアゾール−2−イル]イミダゾ[1,2−a]ピリジン−2−カルボキサミド(本発明化合物No.4-001c)の合成
上記合成例3に記載の方法により合成した3−(エチルチオ)−N−メチル−7−(トリフルオロメチル)−N−[5−(トリフルオロメチル)−1,3,4−チアジアゾール−2−イル]イミダゾ[1,2−a]ピリジン−2−カルボキサミド200mg及びクロロホルム3mlの混合溶液に、氷冷下にて、65質量%メタクロロ過安息香酸(東京化成工業社製:約30質量%の水を含む。以下同様である。)35mgを添加した。添加終了後、該反応混合物を室温にて1時間撹拌した。反応終了後、該反応混合物に飽和チオ硫酸ナトリウム水溶液5mlを加え、クロロホルムにて抽出(5ml×3)した。得られた有機層を1mol/L水酸化ナトリウム水溶液5mlで洗浄し、無水硫酸ナトリウムで脱水・乾燥した後、減圧下にて溶媒を留去した。得られた残留物をn−ヘキサン−酢酸エチル(95:5〜5:95のグラジエント)にて溶出する中圧分取液体クロマトグラフィーにて精製し、目的物75mgを淡黄色固体として得た。
融点:153〜154℃
1H-NMR(CDCl3) :δ9.56(d, J=7.2Hz, 1H), 8.09(s, 1H), 7.19(d, J=7.2Hz, 1H), 4.43(s, 3H), 3.60-3.30(m, 2H), 1.50(t, J=7.5Hz, 3H).
合成例5:3−(エチルスルホニル)−N−メチル−7−(トリフルオロメチル)−N−[5−(トリフルオロメチル)−1,3,4−チアジアゾール−2−イル]イミダゾ[1,2−a]ピリジン−2−カルボキサミド(本発明化合物No.4-001a)の合成
上記合成例3に記載の方法により合成した3−(エチルチオ)−N−メチル−7−(トリフルオロメチル)−N−[5−(トリフルオロメチル)−1,3,4−チアジアゾール−2−イル]イミダゾ[1,2−a]ピリジン−2−カルボキサミド150mg及びクロロホルム3mlの混合溶液に、氷冷下にて65質量%メタクロロ過安息香酸(約30質量%の水を含む)193mgを添加した。添加終了後、該反応混合物を室温にて3時間撹拌した。反応終了後、該反応混合物に飽和チオ硫酸ナトリウム水溶液5mlを加え、クロロホルムにて抽出(5ml×3)した。得られた有機層を1mol/L水酸化ナトリウム水溶液5mlで洗浄し、無水硫酸ナトリウムで脱水・乾燥した後、減圧下にて溶媒を留去した。得られた残留物をn−ヘキサン−酢酸エチル(95:5〜5:95のグラジエント)にて溶出する中圧分取液体クロマトグラフィーにて精製し、目的物183mgを淡黄色油状物として得た。1H-NMR(CDCl3) :δ9.16(d, J=7.5Hz, 1H), 8.15(s, 1H), 7.38(d, J=7.5Hz, 1H), 3.87(s, 3H), 3.57(q, J=7.5Hz, 2H), 1.42(t, J=7.5Hz, 3H).
合成例6:3−(エチルチオ)−7−ヨード−N−メチル−N−[5−(トリフルオロメチル)−1,3,4−チアジアゾール−2−イル]イミダゾ[1,2−a]ピリジン−2−カルボキサミド(本発明化合物No.4-002c)の合成
3−(エチルチオ)−7−ヨードイミダゾ[1,2−a]ピリジン−2−カルボン酸624mg、1,2−ジクロロエタン10ml及びテトラヒドロフラン1mlの混合溶液に、室温にて4−ジメチルアミノピリジン765mg及び2−メチル−6−ニトロ安息香酸無水物741mgを順次添加した。添加終了後、室温にて20分撹拌した。該反応混合物にN−メチル−5−(トリフルオロメチル)−1,3,4−チアジアゾール−2−アミン344mgを添加し、室温にて21時間撹拌した。反応終了後、該反応混合物に飽和炭酸水素ナトリウム水溶液10mlを加え、クロロホルムにて抽出(10ml×3)した。得られた有機層を無水硫酸ナトリウムで脱水・乾燥した後、減圧下にて溶媒を留去した。得られた残留物を中圧分取液体クロマトグラフィー(n−ヘキサン:酢酸エチル=100:0〜50:50のグラジエント)にて精製し、目的物554mgを淡黄色固体として得た。
1H-NMR(CDCl3) :δ8.32(d, J=7.5Hz, 1H), 8.13(s, 1H), 7.35-7.25(m, 1H), 4.03(s, 3H), 2.94(q, J=7.2Hz, 2H), 1.27(t, J=7.2Hz, 3H).
融点:184〜186℃
合成例7:7−シアノ−3−(エチルチオ)−N−メチル−N−[5−(トリフルオロメチル)−1,3,4−チアジアゾール−2イル]イミダゾ[1,2−a]ピリジン−2−カルボキサミド(本発明化合物No.4-003c)の合成
合成例6に記載の方法により合成した3−(エチルチオ)−7−ヨード−N−メチル−N−[5−(トリフルオロメチル)−1,3,4−チアジアゾール−2−イル]イミダゾ[1,2−a]ピリジン−2−カルボキサミド350mg、シアン化亜鉛120mg及びN−メチル−2−ピロリドン3.4mlの混合溶液に、テトラキス(トリエニルホスフィン)パラジウム(0)79mgを添加した。反応容器内を窒素ガスで置換した後、120℃にて2時間撹拌した。反応終了後、該反応混合物に28質量%アンモニア水0.2mlを添加し、クロロホルムにて抽出(10ml×3)した。得られた有機層を無水硫酸ナトリウムで脱水・乾燥した後、減圧下にて溶媒を留去した。得られた残留物を中圧分取液体クロマトグラフィー(n−ヘキサン:酢酸エチル=100:0〜50:50のグラジエント)にて精製し、目的物109mgを白色固体として得た。
1H-NMR(CDCl3) :δ8.67(d, J=7.2Hz, 1H), 8.15-8.10(m, 1H), 7.25-7.15(m, 1H), 4.03(s, 3H), 2.99(q, J=7.5Hz, 2H), 1.23(t, J=7.5Hz, 3H).
融点:194〜195℃
合成例8:7−シアノ−3−(エチルスルホニル)−N−メチル−N−[5−(トリフルオロメチル)−1,3,4−チアジアゾール−2−イル]イミダゾ[1,2−a]ピリジン−2−カルボキサミド(本発明化合物No.4-003a)の合成
合成例7に記載の方法により合成した7−シアノ−3−(エチルチオ)−N−メチル−N−[5−(トリフルオロメチル)−1,3,4−チアジアゾール−2イル]イミダゾ[1,2−a]ピリジン−2−カルボキサミド82mg及びクロロホルム2mlの混合溶液に、氷冷下にて65質量%メタクロロ過安息香酸111mgを添加した。添加終了後、該反応混合物を室温にて3時間撹拌した。反応終了後、該反応混合物に飽和チオ硫酸ナトリウム水溶液5mlを加え、クロロホルムにて抽出(5ml×3)した。得られた有機層を飽和炭酸水素ナトリウム水溶液5mlで洗浄した。該有機層を無水硫酸ナトリウムで脱水・乾燥した後、減圧下にて溶媒を留去した。得られた残留物を中圧分取液体クロマトグラフィー(n−ヘキサン:酢酸エチル=100:0〜50:50のグラジエント)にて精製し、目的物34mgを白色固体として得た。
1H-NMR(CDCl3) :δ9.15-9.10(m, 1H), 8.25-8.20(m, 1H), 7.40-7.30(m, 1H), 3.87(s, 3H), 3.57(q, J=7.5Hz, 2H), 1.41(t, J=7.5Hz, 3H).
融点:161〜165℃
合成例9:3−(エチルスルホニル)−7−ヨード−N−メチル−N−[5−(トリフルオロメチル)−1,3,4−チアジアゾール−2−イル]イミダゾ[1,2−a]ピリジン−2−カルボキサミド(本発明化合物No.4-002a)の合成
合成例6に記載の方法により合成した3−(エチルチオ)−7−ヨード−N−メチル−N−[5−(トリフルオロメチル)−1,3,4−チアジアゾール−2−イル]イミダゾ[1,2−a]ピリジン−2−カルボキサミド91mg及びクロロホルム2mlの混合溶液に、0℃にて65質量%メタクロロ過安息香酸99mgを添加した。該反応混合物を室温にて14時間撹拌した。該反応混合物に飽和チオ硫酸ナトリウム水溶液5mlを加え、クロロホルムにて抽出(5ml×3)した。得られた有機層を飽和炭酸水素ナトリウム水溶液5mlで洗浄した。該有機層を無水硫酸ナトリウムで脱水・乾燥した後、減圧下にて溶媒を留去した。得られた残留物を中圧分取液体クロマトグラフィー(n−ヘキサン:酢酸エチル=100:0〜50:50のグラジエント)にて精製し、目的物89mgを淡黄色固体として得た。
1H-NMR(CDCl3) :δ8.75-8.70(m, 1H), 8.25(s, 1H), 7.50-7.40(m, 1H), 3.85(s, 3H), 3.51(q, J=7.5Hz, 2H), 1.39(t, J=7.5Hz, 3H).
融点:185〜188℃
合成例10:3−(エチルスルホニル)−N−メチル−7−(メチルチオ)−N−[5−(トリフルオロメチル)−1,3,4−チアジアゾール−2−イル]イミダゾ[1,2−a]ピリジン−2−カルボキサミド(本発明化合物No.4-004a)の合成
工程1:2−エチルヘキシル 3−[(3−[エチルスルホニル]−2−(メチル[5−(トリフルオロメチル)−1,3,4−チアジアゾール−2−イル]カルバモイル)イミダゾ[1,2−a]ピリジン−7−イル)チオ]プロパノエートの合成
合成例9に記載の方法により合成した3−(エチルスルホニル)−7−ヨード−N−メチル−N−[5−(トリフルオロメチル)−1,3,4−チアジアゾール−2−イル]イミダゾ[1,2−a]ピリジン−2−カルボキサミド90mg、及び1,4−ジオキサン1mlの混合溶液に、室温にて、ジイソプロピルエチルアミン85μl、4,5−ビス(ジフェニルホスフィノ)−9,9−ジメチルキサンテン10mg、トリス(ジベンジリデンアセトン)ジパラジウム(0)10mg及び2−エチルヘキシル 3−メルカプトプロパノエート54mgを順次添加した。反応容器内を窒素ガスで置換した後、加熱還流下にて2時間撹拌した。該反応混合物に水5mlを加え、クロロホルムにて抽出(5ml×3)した。得られた有機層を無水硫酸ナトリウムで脱水・乾燥した後、減圧下にて溶媒を留去した。得られた残留物を、中圧分取液体クロマトグラフィー(n−ヘキサン:酢酸エチル=95:5〜60:40のグラジエント)にて精製し、目的物95mgを黄色油状物として得た。
1H-NMR(CDCl3) : δ8.80-8.75(m, 1H), 7.55-7.45(m, 1H), 7.05-7.00(m, 1H), 4.10-4.00(m, 2H), 3.86(s, 3H), 3.50(q, J=7.2Hz, 2H), 3.35(t, J=7.2Hz, 2H), 2.79(t, J=7.2Hz, 2H), 1.45-1.20(m, 12H), 0.95-0.85(m, 6H).
工程2:3−(エチルスルホニル)−N−メチル−7−(メチルチオ)−N−[5−(トリフルオロメチル)−1,3,4−チアジアゾール−2−イル]イミダゾ[1,2−a]ピリジン−2−カルボキサミド(本発明化合物No.4-004a)の合成
工程1に記載の方法により合成した2−エチルヘキシル 3−[(3−[エチルスルホニル]−2−(メチル[5−(トリフルオロメチル)−1,3,4−チアジアゾール−2−イル]カルバモイル)イミダゾ[1,2−a]ピリジン−7−イル)チオ]プロパノエート179mg及びテトラヒドロフラン2.8mlの混合溶液に、室温にて1,8−ジアザビシクロ[5.4.0]−7−ウンデセン129mgを添加した。該反応混合物を4時間撹拌した。該反応混合物にトリフルオロメタンスルホン酸メチル139mgを添加し、室温にて1.5時間撹拌した。反応終了後、該反応混合物に飽和塩化アンモニウム水溶液5mlを加え、クロロホルムにて抽出(5ml×3)した。得られた有機層を無水硫酸ナトリウムで脱水・乾燥した後、減圧下にて溶媒を留去した。得られた残留物を中圧分取液体クロマトグラフィー(n−ヘキサン:酢酸エチル=95:5〜50:50のグラジエント)にて精製し、目的物40mgを黄色固体として得た。
1H-NMR(CDCl3) : δ8.75-8.70(m, 1H), 7.40-7.35(m, 1H), 7.05-6.95(m, 1H), 3.86(s, 3H), 3.49(q, J=7.5Hz, 2H), 2.60(s, 3H), 1.39(t, J=7.5Hz, 3H).
合成例11:3−(エチルスルホニル)−N−メチル−7−(メチルスルフィニル)−N−[5−(トリフルオロメチル)−1,3,4−チアジアゾール−2−イル]イミダゾ[1,2−a]ピリジン−2−カルボキサミド(本発明化合物No.4-005a)の合成
上記合成例10に記載の方法により合成した3−(エチルスルホニル)−N−メチル−7−(メチルチオ)−N−[5−(トリフルオロメチル)−1,3,4−チアジアゾール−2−イル]イミダゾ[1,2−a]ピリジン−2−カルボキサミド40mg及び酢酸0.3mlの混合溶液に、室温にて30重量%過酸化水素水10mgを添加した。該反応混合物を室温にて19時間撹拌した。該反応混合物を、n−ヘキサン−酢酸エチル(100:0〜0:100のグラジエント)にて溶出する中圧分取液体クロマトグラフィーにて精製し、目的物26mgを白色固体として得た。
融点:174〜177℃
1H-NMR(CDCl3) : δ9.15(d, J=7.5Hz, 1H), 8.18(s, 1H), 7.40-7.30(m, 1H), 3.87(s, 3H), 3.56(q, J=7.5Hz, 2H), 2.87(s, 3H), 1.42(t, J=7.5Hz, 3H).
以下に、本発明化合物の合成中間体の合成例を記載する。
The chemical shift value of the proton nuclear magnetic resonance spectrum described below (hereinafter referred to as 1 H-NMR) is 300 MHz in deuterated chloroform solvent using Me 4 Si (tetramethylsilane) as a reference substance. (Model: JNM-ECX300 or JNM-ECP300, manufactured by JEOL). In addition, the symbol in the chemical shift value of 1 H-NMR represents the following meaning.
s: singlet, d: doublet, dd: double doublet, t: triplet, q: quartet, m: multiplet, brs: broad singlet Synthesis Example 1: N- (2,2-difluorobenzo [d] [1,3 Synthesis of dioxole-5-yl) -3- (ethylsulfonyl) -7- (trifluoromethyl) imidazo [1,2-a] pyridine-2-carboxamide (the present compound No. 5-001a) 5-amino To a mixed solution of 83 mg of 2,2-difluoro-1,3-benzo [d] [1,3] dioxole, 391 mg of triethylamine and 1.0 ml of dichloromethane at room temperature, 3- (ethylsulfonyl) -7- (tri Fluoromethyl) imidazo [1,2-a] pyridine-2-carbonyl chloride 110 mg and dichloromethane 1.0 ml If the solution was added. After the addition was complete, the reaction mixture was stirred at room temperature for 18 hours. After completion of the reaction, 5 ml of saturated sodium bicarbonate was added to the reaction mixture, and the mixture was extracted with chloroform (5 ml × 2). The obtained organic layer was dehydrated and dried over anhydrous sodium sulfate, and then the solvent was distilled off under reduced pressure. The obtained residue was purified by medium pressure preparative liquid chromatography eluting with n-hexane-ethyl acetate [gradient of 90:10 to 20:80 (volume ratio, the same applies hereinafter)]. 120 mg of the product was obtained as a pale yellow solid.
Melting point: 197-200 ° C
1 H-NMR (CDCl 3 ): δ9.55 (d, J = 7.5Hz, 1H), 9.35 (s, 1H), 8.05 (s, 1H), 7.85 (d, J = 1.8Hz, 1H), 7.30 -7.20 (m, 2H), 7.05 (d, J = 8.7Hz, 1H), 3.96 (q, J = 7.2Hz, 2H), 1.37 (t, J = 7.2Hz, 3H).
Synthesis Example 2: N- (2,2-difluorobenzo [d] [1,3] dioxol-5-yl) -3- (ethylsulfonyl) -N-methyl-7- (trifluoromethyl) imidazo [1, 2-a] Synthesis of pyridine-2-carboxamide (Compound No. 5-002a of the present invention) In a mixed solution of 60% by weight sodium hydride (dispersed in mineral oil) 10 mg and THF 0.5 ml, the above Synthesis Example 1 was performed at room temperature. N- (2,2-difluorobenzo [d] [1,3] dioxol-5-yl) -3- (ethylsulfonyl) -7- (trifluoromethyl) imidazo [1,2 -A] A solution of 108 mg of pyridine-2-carboxamide in 1.2 ml of THF was added. After the addition was complete, the reaction mixture was stirred at room temperature for 1 hour. After completion of stirring, 35 mg of methyl iodide was added to the reaction mixture at room temperature. After the addition was complete, the reaction mixture was stirred at room temperature for 1 hour. After completion of the reaction, 5 ml of water was added to the reaction mixture and extracted with ethyl acetate (5 ml × 3). The obtained organic layer was dehydrated and dried in the order of saturated saline and then anhydrous sodium sulfate, and then the solvent was distilled off under reduced pressure. The obtained residue was purified by medium pressure preparative liquid chromatography eluting with n-hexane-ethyl acetate (gradient from 95: 5 to 30:70) to obtain 42 mg of the desired product as a colorless oil. .
1 H-NMR (CDCl 3 ): δ9.03 (d, J = 7.2Hz, 1H), 7.86 (s, 1H), 7.25-7.05 (m, 3H), 6.86 (d, J = 8.4Hz, 1H) , 3.60 (q, J = 7.5Hz, 2H), 3.49 (s, 3H), 1.36 (t, J = 7.5Hz, 3H).
Synthesis Example 3: 3- (Ethylthio) -N-methyl-7- (trifluoromethyl) -N- [5- (trifluoromethyl) -1,3,4-thiadiazol-2-yl] imidazo [1,2 -A] Synthesis of pyridine-2-carboxamide (the present compound No. 4-001b) 3- (Ethylthio) -7- (trifluoromethyl) imidazo [1,2-a] pyridine-2-carboxylic acid 300 mg and dichloromethane To 4 ml of the mixed solution, 377 mg of oxalyl chloride and 20 mg of N, N-dimethylformamide were sequentially added under ice cooling. After the addition was complete, the reaction mixture was stirred at room temperature for 30 minutes. After completion of the reaction, the solvent was distilled off from the reaction mixture under reduced pressure to obtain 3- (ethylsulfonyl) -7- (trifluoromethyl) imidazo [1,2-a] pyridine-2-carbonyl chloride. . 3- (Ethylsulfonyl) -7- (trifluoromethyl) imidazo [1,2-a] pyridine-2-carbonyl chloride dissolved in 3 ml of dichloromethane was converted to 2-methylamino-5- (trifluoromethyl) -1, To a mixed solution of 208 mg of 3,4-thiadiazole and 3 ml of pyridine, it was added under ice cooling. After the addition was complete, the reaction mixture was stirred at room temperature for 1 hour. After completion of the reaction, the solvent of the reaction mixture was distilled off under reduced pressure. To the obtained residue, 5 ml of water was added and extracted with ethyl acetate (5 ml × 3). The obtained organic layer was washed with 5 ml of 1 mol / L hydrochloric acid, dehydrated and dried in the order of saturated brine and then anhydrous sodium sulfate, and then the solvent was distilled off under reduced pressure. The resulting residue was purified by alumina column chromatography eluting with n-hexane-ethyl acetate (95: 5 to 30:70 gradient). Further, the obtained solid was purified by medium pressure preparative liquid chromatography eluting with n-hexane-ethyl acetate (gradient of 95: 5 to 30:70) to obtain 525 mg of the desired product as a pale yellow solid.
Melting point: 84-86 ° C
1 H-NMR (CDCl 3 ): δ8.70 (d, J = 7.5Hz, 1H), 8.03 (s, 1H), 7.23 (d, J = 7.5Hz, 1H), 4.04 (s, 3H), 2.98 (q, J = 7.5Hz, 2H), 1.23 (t, J = 7.5Hz, 3H).
Synthesis Example 4: 3- (Ethylsulfinyl) -N-methyl-7- (trifluoromethyl) -N- [5- (trifluoromethyl) -1,3,4-thiadiazol-2-yl] imidazo [1, 2-a] Synthesis of pyridine-2-carboxamide (Compound No. 4-001c of the present invention) 3- (ethylthio) -N-methyl-7- (trifluoromethyl)-synthesized by the method described in Synthesis Example 3 above To a mixed solution of N- [5- (trifluoromethyl) -1,3,4-thiadiazol-2-yl] imidazo [1,2-a] pyridine-2-carboxamide and 3 ml of chloroform under ice-cooling. 35 mg of 65% by mass metachloroperbenzoic acid (manufactured by Tokyo Chemical Industry Co., Ltd .: containing about 30% by mass of water; the same applies hereinafter) was added. After the addition was complete, the reaction mixture was stirred at room temperature for 1 hour. After completion of the reaction, 5 ml of a saturated aqueous sodium thiosulfate solution was added to the reaction mixture, followed by extraction with chloroform (5 ml × 3). The obtained organic layer was washed with 5 ml of 1 mol / L sodium hydroxide aqueous solution, dehydrated and dried over anhydrous sodium sulfate, and then the solvent was distilled off under reduced pressure. The obtained residue was purified by medium pressure preparative liquid chromatography eluting with n-hexane-ethyl acetate (gradient of 95: 5 to 5:95) to obtain 75 mg of the desired product as a pale yellow solid.
Melting point: 153-154 ° C
1 H-NMR (CDCl 3 ): δ9.56 (d, J = 7.2Hz, 1H), 8.09 (s, 1H), 7.19 (d, J = 7.2Hz, 1H), 4.43 (s, 3H), 3.60 -3.30 (m, 2H), 1.50 (t, J = 7.5Hz, 3H).
Synthesis Example 5: 3- (Ethylsulfonyl) -N-methyl-7- (trifluoromethyl) -N- [5- (trifluoromethyl) -1,3,4-thiadiazol-2-yl] imidazo [1, 2-a] Synthesis of pyridine-2-carboxamide (Compound No. 4-001a of the present invention) 3- (ethylthio) -N-methyl-7- (trifluoromethyl)-synthesized by the method described in Synthesis Example 3 above To a mixed solution of 150 mg of N- [5- (trifluoromethyl) -1,3,4-thiadiazol-2-yl] imidazo [1,2-a] pyridine-2-carboxamide and 3 ml of chloroform under ice cooling. 193 mg of 65 wt% metachloroperbenzoic acid (containing about 30 wt% water) was added. After the addition was complete, the reaction mixture was stirred at room temperature for 3 hours. After completion of the reaction, 5 ml of a saturated aqueous sodium thiosulfate solution was added to the reaction mixture, followed by extraction with chloroform (5 ml × 3). The obtained organic layer was washed with 5 ml of 1 mol / L sodium hydroxide aqueous solution, dehydrated and dried over anhydrous sodium sulfate, and then the solvent was distilled off under reduced pressure. The obtained residue was purified by medium pressure preparative liquid chromatography eluting with n-hexane-ethyl acetate (gradient of 95: 5 to 5:95) to obtain 183 mg of the desired product as a pale yellow oil. . 1 H-NMR (CDCl 3 ): δ 9.16 (d, J = 7.5Hz, 1H), 8.15 (s, 1H), 7.38 (d, J = 7.5Hz, 1H), 3.87 (s, 3H), 3.57 (q, J = 7.5Hz, 2H), 1.42 (t, J = 7.5Hz, 3H).
Synthesis Example 6: 3- (Ethylthio) -7-iodo-N-methyl-N- [5- (trifluoromethyl) -1,3,4-thiadiazol-2-yl] imidazo [1,2-a] pyridine Synthesis of 2-carboxamide (Compound No. 4-002c of the present invention) 3- (ethylthio) -7-iodoimidazo [1,2-a] pyridine-2-carboxylic acid 624 mg, 1,2-dichloroethane 10 ml and tetrahydrofuran 1 ml To this mixed solution, 765 mg of 4-dimethylaminopyridine and 741 mg of 2-methyl-6-nitrobenzoic anhydride were sequentially added at room temperature. After completion of the addition, the mixture was stirred at room temperature for 20 minutes. 344 mg of N-methyl-5- (trifluoromethyl) -1,3,4-thiadiazol-2-amine was added to the reaction mixture, and the mixture was stirred at room temperature for 21 hours. After completion of the reaction, 10 ml of a saturated aqueous sodium hydrogen carbonate solution was added to the reaction mixture, and the mixture was extracted with chloroform (10 ml × 3). The obtained organic layer was dehydrated and dried over anhydrous sodium sulfate, and then the solvent was distilled off under reduced pressure. The obtained residue was purified by medium pressure preparative liquid chromatography (n-hexane: ethyl acetate = 100: 0 to 50:50 gradient) to obtain 554 mg of the desired product as a pale yellow solid.
1 H-NMR (CDCl 3 ): δ 8.32 (d, J = 7.5Hz, 1H), 8.13 (s, 1H), 7.35-7.25 (m, 1H), 4.03 (s, 3H), 2.94 (q, J = 7.2Hz, 2H), 1.27 (t, J = 7.2Hz, 3H).
Melting point: 184-186 ° C
Synthesis Example 7: 7-Cyano-3- (ethylthio) -N-methyl-N- [5- (trifluoromethyl) -1,3,4-thiadiazol-2-yl] imidazo [1,2-a] pyridine- Synthesis of 2-carboxamide (Compound No. 4-003c of the present invention) 3- (ethylthio) -7-iodo-N-methyl-N- [5- (trifluoromethyl)-synthesized by the method described in Synthesis Example 6 To a mixed solution of 350 mg of 1,3,4-thiadiazol-2-yl] imidazo [1,2-a] pyridine-2-carboxamide, 120 mg of zinc cyanide and 3.4 ml of N-methyl-2-pyrrolidone, tetrakis (tri 79 mg of enylphosphine) palladium (0) was added. After replacing the inside of the reaction vessel with nitrogen gas, the mixture was stirred at 120 ° C. for 2 hours. After completion of the reaction, 0.2 ml of 28% by mass aqueous ammonia was added to the reaction mixture, followed by extraction with chloroform (10 ml × 3). The obtained organic layer was dehydrated and dried over anhydrous sodium sulfate, and then the solvent was distilled off under reduced pressure. The obtained residue was purified by medium pressure preparative liquid chromatography (gradient of n-hexane: ethyl acetate = 100: 0 to 50:50) to obtain 109 mg of the desired product as a white solid.
1 H-NMR (CDCl 3 ): δ8.67 (d, J = 7.2Hz, 1H), 8.15-8.10 (m, 1H), 7.25-7.15 (m, 1H), 4.03 (s, 3H), 2.99 ( q, J = 7.5Hz, 2H), 1.23 (t, J = 7.5Hz, 3H).
Melting point: 194-195 ° C
Synthesis Example 8 7-Cyano-3- (ethylsulfonyl) -N-methyl-N- [5- (trifluoromethyl) -1,3,4-thiadiazol-2-yl] imidazo [1,2-a] Synthesis of pyridine-2-carboxamide (the present compound No. 4-003a) 7-Cyano-3- (ethylthio) -N-methyl-N- [5- (trifluoromethyl) synthesized by the method described in Synthesis Example 7 ) -1,3,4-thiadiazol-2yl] imidazo [1,2-a] pyridine-2-carboxamide in a mixed solution of 82 mg and 2 ml of chloroform was added 111 mg of 65% by mass metachloroperbenzoic acid under ice-cooling. did. After the addition was complete, the reaction mixture was stirred at room temperature for 3 hours. After completion of the reaction, 5 ml of a saturated aqueous sodium thiosulfate solution was added to the reaction mixture, followed by extraction with chloroform (5 ml × 3). The obtained organic layer was washed with 5 ml of a saturated aqueous sodium hydrogen carbonate solution. The organic layer was dehydrated and dried over anhydrous sodium sulfate, and the solvent was distilled off under reduced pressure. The obtained residue was purified by medium pressure preparative liquid chromatography (gradient of n-hexane: ethyl acetate = 100: 0-50: 50) to obtain 34 mg of the desired product as a white solid.
1 H-NMR (CDCl 3 ): δ 9.15-9.10 (m, 1H), 8.25-8.20 (m, 1H), 7.40-7.30 (m, 1H), 3.87 (s, 3H), 3.57 (q, J = 7.5Hz, 2H), 1.41 (t, J = 7.5Hz, 3H).
Melting point: 161-165 ° C
Synthesis Example 9: 3- (Ethylsulfonyl) -7-iodo-N-methyl-N- [5- (trifluoromethyl) -1,3,4-thiadiazol-2-yl] imidazo [1,2-a] Synthesis of Pyridine-2-carboxamide (Compound No. 4-002a of the Present Invention) 3- (Ethylthio) -7-iodo-N-methyl-N- [5- (trifluoromethyl) synthesized by the method described in Synthesis Example 6 ) -1,3,4-thiadiazol-2-yl] imidazo [1,2-a] pyridine-2-carboxamide 91 mg and chloroform 2 mass 99 ml were added 99% 65% metachloroperbenzoic acid at 0 ° C. did. The reaction mixture was stirred at room temperature for 14 hours. To the reaction mixture was added 5 ml of a saturated aqueous sodium thiosulfate solution, and the mixture was extracted with chloroform (5 ml × 3). The obtained organic layer was washed with 5 ml of a saturated aqueous sodium hydrogen carbonate solution. The organic layer was dehydrated and dried over anhydrous sodium sulfate, and the solvent was distilled off under reduced pressure. The obtained residue was purified by medium pressure preparative liquid chromatography (n-hexane: ethyl acetate = 100: 0 to 50:50 gradient) to obtain 89 mg of the desired product as a pale yellow solid.
1 H-NMR (CDCl 3 ): δ8.75-8.70 (m, 1H), 8.25 (s, 1H), 7.50-7.40 (m, 1H), 3.85 (s, 3H), 3.51 (q, J = 7.5 Hz, 2H), 1.39 (t, J = 7.5Hz, 3H).
Melting point: 185-188 ° C
Synthesis Example 10: 3- (Ethylsulfonyl) -N-methyl-7- (methylthio) -N- [5- (trifluoromethyl) -1,3,4-thiadiazol-2-yl] imidazo [1,2- a] Synthesis of pyridine-2-carboxamide (Compound No. 4-004a of the present invention) Step 1: 2-ethylhexyl 3-[(3- [ethylsulfonyl] -2- (methyl [5- (trifluoromethyl) -1 , 3,4-thiadiazol-2-yl] carbamoyl) imidazo [1,2-a] pyridin-7-yl) thio] propanoate 3- (ethylsulfonyl) -7 synthesized by the method described in Synthesis Example 9 90 mg of iodo-N-methyl-N- [5- (trifluoromethyl) -1,3,4-thiadiazol-2-yl] imidazo [1,2-a] pyridine-2-carboxamide; To a mixed solution of 1 ml of 1,4-dioxane, 85 μl of diisopropylethylamine, 10 mg of 4,5-bis (diphenylphosphino) -9,9-dimethylxanthene, 10 mg of tris (dibenzylideneacetone) dipalladium (0) at room temperature. And 54 mg of 2-ethylhexyl 3-mercaptopropanoate were added sequentially. The inside of the reaction vessel was replaced with nitrogen gas, and then stirred for 2 hours under heating and reflux. 5 ml of water was added to the reaction mixture, and the mixture was extracted with chloroform (5 ml × 3). The obtained organic layer was dehydrated and dried over anhydrous sodium sulfate, and then the solvent was distilled off under reduced pressure. The obtained residue was purified by medium pressure preparative liquid chromatography (n-hexane: ethyl acetate = 95: 5 to 60:40 gradient) to obtain the desired product (95 mg) as a yellow oil.
1 H-NMR (CDCl 3 ): δ8.80-8.75 (m, 1H), 7.55-7.45 (m, 1H), 7.05-7.00 (m, 1H), 4.10-4.00 (m, 2H), 3.86 (s , 3H), 3.50 (q, J = 7.2Hz, 2H), 3.35 (t, J = 7.2Hz, 2H), 2.79 (t, J = 7.2Hz, 2H), 1.45-1.20 (m, 12H), 0.95 -0.85 (m, 6H).
Step 2: 3- (Ethylsulfonyl) -N-methyl-7- (methylthio) -N- [5- (trifluoromethyl) -1,3,4-thiadiazol-2-yl] imidazo [1,2-a ] Synthesis of pyridine-2-carboxamide (Compound No. 4-004a of the present invention) 2-ethylhexyl 3-[(3- [ethylsulfonyl] -2- (methyl [5- (tri Fluoromethyl) -1,3,4-thiadiazol-2-yl] carbamoyl) imidazo [1,2-a] pyridin-7-yl) thio] propanoate in a mixed solution of 179 mg and tetrahydrofuran 2.8 ml at room temperature , 8-diazabicyclo [5.4.0] -7-undecene 129 mg was added. The reaction mixture was stirred for 4 hours. To the reaction mixture, 139 mg of methyl trifluoromethanesulfonate was added and stirred at room temperature for 1.5 hours. After completion of the reaction, 5 ml of a saturated aqueous ammonium chloride solution was added to the reaction mixture, and the mixture was extracted with chloroform (5 ml × 3). The obtained organic layer was dehydrated and dried over anhydrous sodium sulfate, and then the solvent was distilled off under reduced pressure. The obtained residue was purified by medium pressure preparative liquid chromatography (n-hexane: ethyl acetate = 95: 5 to 50:50 gradient) to obtain 40 mg of the desired product as a yellow solid.
1 H-NMR (CDCl 3 ): δ8.75-8.70 (m, 1H), 7.40-7.35 (m, 1H), 7.05-6.95 (m, 1H), 3.86 (s, 3H), 3.49 (q, J = 7.5Hz, 2H), 2.60 (s, 3H), 1.39 (t, J = 7.5Hz, 3H).
Synthesis Example 11 3- (Ethylsulfonyl) -N-methyl-7- (methylsulfinyl) -N- [5- (trifluoromethyl) -1,3,4-thiadiazol-2-yl] imidazo [1,2 -A] Synthesis of pyridine-2-carboxamide (Compound No. 4-005a of the present invention) 3- (ethylsulfonyl) -N-methyl-7- (methylthio) -N- synthesized by the method described in Synthesis Example 10 above 30 weights at room temperature in a mixed solution of 40 mg of [5- (trifluoromethyl) -1,3,4-thiadiazol-2-yl] imidazo [1,2-a] pyridine-2-carboxamide and 0.3 ml of acetic acid 10% aqueous hydrogen peroxide was added. The reaction mixture was stirred at room temperature for 19 hours. The reaction mixture was purified by medium pressure preparative liquid chromatography eluting with n-hexane-ethyl acetate (gradient of 100: 0 to 0: 100) to obtain 26 mg of the desired product as a white solid.
Melting point: 174-177 ° C
1 H-NMR (CDCl 3 ): δ9.15 (d, J = 7.5Hz, 1H), 8.18 (s, 1H), 7.40-7.30 (m, 1H), 3.87 (s, 3H), 3.56 (q, J = 7.5Hz, 2H), 2.87 (s, 3H), 1.42 (t, J = 7.5Hz, 3H).
Below, the synthesis example of the synthetic intermediate of this invention compound is described.
中間体合成例1:3−(エチルチオ)−7−(トリフルオロメチル)イミダゾ[1,2−a]ピリジン−2−カルボン酸の合成
工程1:3−ヨード−7−(トリフルオロメチル)イミダゾ[1,2−a]ピリジン−2−カルボン酸エチルの合成
7−(トリフルオロメチル)イミダゾ[1,2−a]ピリジン−2−カルボン酸エチル3.73g及びN,N−ジメチルホルムアミド20mlの混合溶液に、室温にて、N−ヨードスクシンイミド6.5gを添加した。添加終了後、該反応混合物を80℃にて、5時間攪拌した。反応終了後、該反応混合物に水を添加し、析出した固体を、ろ過によりろ別した。得られた固体をクロロホルム20mlに溶解させ、飽和チオ硫酸ナトリウム水溶液、次いで飽和炭酸水素ナトリウムの順で洗浄した。得られた有機層を無水硫酸ナトリウムで脱水・乾燥後、減圧下にて溶媒を留去することで、目的物5.08gを淡黄色固体として得た。
融点:183〜185℃
1H-NMR(CDCl3) : δ8.40(d, J=7.2Hz, 1H), 7.99(s, 1H), 7.17(dd, J=7.2, 1.7Hz, 1H), 4.53(q, J=7.2Hz, 2H), 1.50(t, J=7.2Hz, 3H).
工程2:3−(エチルチオ)−7−(トリフルオロメチル)イミダゾ[1,2−a]ピリジン−2−カルボン酸エチルの合成
3−ヨード−7−(トリフルオロメチル)イミダゾ[1,2−a]ピリジン−2−カルボン酸エチル5.73g及び1,4−ジオキサン40mlの混合溶液に、室温にて、ジイソプロピルエチルアミン5.79g、4,5−ビス(ジフェニルホスフィノ)−9,9−ジメチルキサンテン862mg、トリス(ジベンジリデンアセトン)ジパラジウム(0価)682mg、エタンチオール1.85gを順次添加した。添加終了後、反応容器内を窒素ガスで置換した後、加熱還流下にて2時間撹拌した。反応終了後、該反応混合物を、セライトを用いてろ過し、セライトをクロロホルム30mlで洗浄した。得られたろ液と洗浄液を合わせ、減圧下にて溶媒を留去した。得られた残留物を、n−ヘキサン−酢酸エチル(100:0〜50:50のグラジエント)にて溶出する中圧分取液体クロマトグラフィーにて精製し、目的物5.58gを茶色固体として得た。
融点:50〜52℃
1H-NMR(CDCl3) : δ8.67(d, J=7.4Hz, 1H), 8.02-8.00(m, 1H), 7.15(dd, J=7.4, 1.8Hz, 1H), 4.52(q, J=7.0Hz, 2H), 2.98(q, J=7.5Hz, 2H), 1.48(t, J=7.0Hz, 3H), 1.20(t, J=7.5Hz, 3H).
工程3:3−(エチルチオ)−7−(トリフルオロメチル)イミダゾ[1,2−a]ピリジン−2−カルボン酸の合成
3−(エチルチオ)−7−(トリフルオロメチル)イミダゾ[1,2−a]ピリジン−2−カルボン酸エチル5.58g、エタノール60ml及びテトラヒドロフラン30mlの混合溶液に、室温にて、3mol/L水酸化ナトリウム水溶液10mlを添加した。添加終了後、該反応混合物を室温にて5時間撹拌した。反応終了後、減圧下にて該反応混合物の溶媒を留去した。得られた残留物に、1mol/L塩酸水溶液を添加し、水層のpHを2とした後、酢酸エチル(20ml×2)にて抽出した。得られた有機層を、飽和食塩水、次いで無水硫酸ナトリウムの順で脱水・乾燥した後、減圧下にて溶媒を留去し、目的物3.40gを黄色固体として得た。
融点:163〜171℃
1H-NMR(CDCl3) : δ8.69(d, J=7.2Hz, 1H), 8.19(s, 1H), 7.22(dd, J=7.2, 1.4Hz, 1H), 3.06(q, J=7.4Hz, 2H), 1.23(t, J=7.4Hz, 3H)(CO2Hのプロトンのシグナルは観測できなかった).
中間体合成例2:3−(エチルスルホニル)−7−(トリフルオロメチル)イミダゾ[1,2−a]ピリジン−2−カルボニルクロリドの合成
工程1:3−(エチルスルホニル)−7−(トリフルオロメチル)イミダゾ[1,2−a]ピリジン−2−カルボン酸の合成
3−(エチルチオ)−7−(トリフルオロメチル)イミダゾ[1,2−a]ピリジン−2−カルボン酸5.00g及び酢酸43mlの混合溶液に、室温にて、30重量%過酸化水素水4.88g、タングステン(VI)酸ナトリウム二水和物10mgを順次添加した。添加終了後、該反応混合物を50℃にて6時間撹拌した。反応終了後、減圧下にて該反応混合物の溶媒を留去した。析出した固体をヘキサン30mlで洗浄し、ろ過によりろ別することで、目的物4.39gを淡黄色固体として得た。
1H-NMR(CDCl3) :δ9.41(d, J=7.2Hz, 1H), 8.08(s, 1H), 7.30-7.20(m, 1H), 3.78(q, J=7.2Hz, 2H), 1.34(t, J=7.2Hz, 3H) (CO2Hのプロトンのシグナルは観測できなかった).
工程2:3−(エチルスルホニル)−7−(トリフルオロメチル)イミダゾ[1,2−a]ピリジン−2−カルボニルクロリドの合成
3−(エチルスルホニル)−7−(トリフルオロメチル)イミダゾ[1,2−a]ピリジン−2−カルボン酸4.39g及びジクロロメタン13.6mlの混合溶液に、室温にて、オキサリルクロリド2.3ml、N,N−ジメチルホルムアミド1滴を順次添加した。添加終了後、該反応混合物を室温にて1時間撹拌した。反応終了後、減圧下にて該反応混合物の溶媒を留去した。析出した固体をヘキサン30mlで洗浄し、ろ過によりろ別することで、目的物4.52gを白色固体として得た。
1H-NMR(CDCl3) : δ9.39(d, J=7.5Hz, 1H), 8.20(brs, 1H), 7.40-7.30(m, 1H), 3.64(q, J=7.5Hz, 2H), 1.38(t, J=7.5Hz, 3H).
中間体合成例3:3−(エチルチオ)−7−ヨードイミダゾ[1,2−a]ピリジン−2−カルボン酸の合成
工程1:7−ヨードイミダゾ[1,2−a]ピリジン−2−カルボン酸エチルの合成
2−アミノ−4−ヨードピリジン19.0g及びエタノール100mlの混合溶液に、室温にて、炭酸水素ナトリウム9.4g、ブロモピルビン酸エチル22.0gを順次添加した。該反応混合物を加熱還流下にて2時間攪拌した。反応終了後、減圧下にて溶媒を留去した。得られた残留物に水100ml及びジイソプロピルエーテル100mlを加え、析出した固体をろ過により取り出した。得られた固体にエタノール100mlを加え、減圧下で溶媒を留去した。得られた残留物にトルエン100mlを加え、減圧下で溶媒を留去することで目的物23.4gを橙色固体として得た。
1H-NMR(CDCl3) : δ8.15-8.05(m, 2H), 7.90-7.85(m, 1H), 7.10-7.05(m, 1H), 4.45(q, J=7.2Hz, 2H), 1.43(t, J=7.2Hz, 3H).
工程2:3−(エチルチオ)−7−ヨードイミダゾ[1,2−a]ピリジン−2−カルボン酸エチルの合成
N−クロロスクシンイミド3.38g及び1,2−ジクロロエタン31mlの混合溶液に、−20℃にてエタンチオール2.28mlを添加した。該反応混合物を室温にて4時間撹拌した。該反応混合物に、室温にて7−ヨードイミダゾ[1,2−a]ピリジン−2−カルボン酸エチル1.00gを添加した。該反応混合物を加熱還流下にて6時間攪拌した。該反応混合物に、別容器で調製したN−クロロスクシンイミド3.38g及びエタンチオール2.28mlの1,2−ジクロロエタン31ml溶液を、室温にて添加した。該反応混合物を加熱還流下にて5時間攪拌した。反応終了後、該反応混合物に飽和炭酸水素ナトリウム水溶液100mlを添加し、クロロホルム(100ml×2)にて抽出した。得られた有機層を無水硫酸ナトリウムで脱水・乾燥後、減圧下にて溶媒を留去した。得られた残留物を中圧分取液体クロマトグラフィー(n−ヘキサン−酢酸エチル=98:2〜90:10のグラジエント)にて精製し、目的物880mgを茶色固体として得た。
1H-NMR(CDCl3) : δ8.29(d, J=6.6Hz, 1H), 8.10(d, J=0.9Hz, 1H), 7.21(dd, J=7.2, 1.5Hz, 1H), 4.49(q, J=7.2Hz, 2H), 2.93(q, J=7.2Hz, 2H), 1.47(t, J=7.5Hz, 3H), 1.18(t, J=7.5Hz, 3H).
工程3:3−(エチルチオ)−7−ヨードイミダゾ[1,2−a]ピリジン−2−カルボン酸の合成
3−(エチルチオ)−7−ヨードイミダゾ[1,2−a]ピリジン−2−カルボン酸エチル880mg及びテトラヒドロフラン9mlの混合溶液に、0℃にて0.6mol/L水酸化リチウム水溶液3mlを添加した。該反応混合物を室温にて3時間撹拌した。反応終了後、減圧下にて該反応混合物の溶媒を留去した。得られた残留物に水10mlを加え、水層をジエチルエーテル10mlで洗浄した。得られた水層に2mol/L塩酸を添加して水層のpHを2とした後、クロロホルム(10ml×2)にて抽出した。得られた有機層を無水硫酸ナトリウムで脱水・乾燥した後、減圧下にて溶媒を留去することにより目的物654mgを黄色固体として得た。
融点:175〜178℃
中間体合成例4:3−(エチルチオ)−7−ヨードイミダゾ[1,2−a]ピリジン−2−カルボン酸の合成
工程1:7−ヨード−3−チオシアナトイミダゾ[1,2−a]ピリジン−2−カルボン酸エチルの合成
チオシアン酸カリウム184mg及びアセトニトリル3mlの混合溶液に、0℃にて、臭素182mg及び中間体合成例3の工程1の方法により合成した7−ヨードイミダゾ[1,2−a]ピリジン−2−カルボン酸エチル300mgを順次添加した。該反応混合物を室温にて1時間撹拌した。該反応混合物を60℃にて2時間撹拌した。反応終了後、該反応混合物に10質量%亜硫酸水素ナトリウム水溶液5ml及びクロロホルム10mlを順次添加した。該反応混合物をセライトを用いてろ過し、セライトをクロロホルムで洗浄した。得られたろ液と洗浄液を合わせ、クロロホルム10mlにて抽出した。得られた有機層を無水硫酸ナトリウムで脱水・乾燥した後、減圧下にて溶媒を留去した。得られた残留物を中圧分取液体クロマトグラフィー(n−ヘキサン−酢酸エチル=100:5〜50:50のグラジエント)にて精製することにより目的物161mgを白色固体として得た。
1H-NMR(CDCl3) : δ8.30-8.25(m, 1H), 8.17(d, J=6.9Hz, 1H), 7.47(dd, J=6.9, 1.5Hz, 1H), 4.54(q, J=6.9Hz, 2H), 1.49(t, J=6.9Hz, 3H).
工程2:3−(エチルチオ)−7−ヨードイミダゾ[1,2−a]ピリジン−2−カルボン酸の合成
工程1で得られた7−ヨード−3−チオシアナトイミダゾ[1,2−a]ピリジン−2−カルボン酸エチル106mg及びエタノール2mlの混合溶液に、室温にて21質量%ナトリウムエトキシドのエタノール溶液(シグマ・アルドリッチ社製)193mgを添加した。該反応混合物を60℃にて30分間撹拌した。該反応混合物に、室温にて21質量%ナトリウムエトキシドのエタノール溶液109mg及びヨードエタン109mgを順次添加した。該反応混合物を室温にて30分間撹拌した。反応終了後、減圧下にて溶媒を留去した。得られた残留物に1.0mol/L水酸化ナトリウム水溶液10mlを添加し、水層をクロロホルム10mlにて洗浄した。得られた水層に濃塩酸を添加して水層のpHを3とした後、クロロホルム10mlにて抽出した。得られた有機層を無水硫酸ナトリウムで脱水・乾燥した後、減圧下にて溶媒を留去することにより目的物33mgを薄茶色固体として得た。
1H-NMR(CDCl3) : δ8.50-8.20(m, 2H), 7.30-7.20(m, 1H), 3.00(q, J=7.2Hz, 2H), 1.18(t, J=7.2Hz, 3H) (CO2Hのプロトンのシグナルは観測できなかった).
中間体合成例5:7−シアノ−3−(エチルチオ)イミダゾ[1,2−a]ピリジン−2−カルボン酸エチルの合成
工程1:3−(エチルチオ)−2−(メトキシイミノ)プロピオン酸エチルの合成
3−ブロモ−2−(メトキシイミノ)プロピオン酸エチル5.5g及びN,N−ジメチルホルムアミド20mlの混合溶液に、0℃にて、エチルメルカプタン2.2ml及び炭酸カリウム4.1gを順次添加した。該反応混合物を室温にて2時間撹拌した。反応終了後、該反応混合物に水100mlを加え、ジエチルエーテル(100ml×2)にて抽出した。得られた有機層を無水硫酸ナトリウムで脱水・乾燥した後、減圧下にて溶媒を留去することにより目的物5.6gを黄色油状物として得た。
1H-NMR(CDCl3) : δ4.35(q, J=7.2Hz, 2H), 4.06(s, 3H), 3.58(s, 2H), 2.54(q, J=7.5Hz, 2H), 1.36(t, J=7.2Hz, 3H), 1.26(t, J=7.5Hz, 3H).
工程2:3−(エチルスルフィニル)−2−(メトキシイミノ)プロピオン酸エチルの合成
工程1で得られた3−(エチルチオ)−2−(メトキシイミノ)プロピオン酸エチル2.0g及びクロロホルム20mlの混合溶液に、0℃にて、65質量%メタクロロ過安息香酸2.6gを添加した。該反応混合物を0℃にて30分間撹拌した。反応終了後、該反応混合物に10質量%飽和チオ硫酸ナトリウム水溶液15mlを加え、クロロホルムにて抽出(20ml×3)した。得られた有機層を飽和炭酸水素ナトリウム水溶液100mlで洗浄した。該有機層を無水硫酸ナトリウムで脱水・乾燥した後、減圧下にて溶媒を留去した。得られた残留物を中圧分取液体クロマトグラフィー(n−ヘキサン:酢酸エチル=100:0〜0:100のグラジエント)にて精製することにより目的物1.7gを淡黄色油状物として得た。
1H-NMR(CDCl3) :δ4.37(q, J=7.2Hz, 2H), 4.15(s, 3H), 4.10-4.00(m, 2H), 2.85-2.65(m, 2H), 1.45-1.30(m, 6H).
目的物とは別に3−(エチルスルホニル)−2−(メトキシイミノ)プロピオン酸エチル351mgを淡黄色固体として得た。
1H-NMR(CDCl3) :δ4.39(s, 2H), 4.38(q, J=7.5Hz, 2H), 3.06(s, 3H), 3.06(q, J=7.5Hz, 2H), 1.42(t, J=7.5Hz, 3H), 1.38(t, J=7.5Hz, 3H).
工程3:3−クロロ−3−(エチルチオ)−2−(メトキシイミノ)プロピオン酸エチルの合成
工程2で得られた3−(エチルスルフィニル)−2−(メトキシイミノ)プロピオン酸エチル200mg及びジクロロメタン3mlの混合溶液に、0℃にて塩化チオニル108mgを添加した。該反応混合物を室温にて10分間撹拌した。反応終了後、減圧下にて溶媒を留去することで目的物250mgを薄茶色油状物として得た。
1H-NMR(CDCl3) :δ6.31(s, 1H), 4.38(q, J=7.2Hz, 2H), 4.16(s, 3H), 2.82(q, J=7.5Hz, 2H), 1.38(t, J=7.2Hz, 3H), 1.35(t, J=7.5Hz, 3H).
工程4:7−シアノ−3−(エチルチオ)イミダゾ[1,2−a]ピリジン−2−カルボン酸エチルの合成
工程3で得られた3−クロロ−3−(エチルチオ)−2−(メトキシイミノ)プロピオン酸エチル250mg及びアセトニトリル2mlの混合溶媒に、室温にて4−シアノピリジン94mgを添加した。該反応混合物を加熱還流下にて2時間攪拌した。該反応混合物にN,N−ジイソプロピルエチルアミン116mgを添加し、加熱還流下にて1時間攪拌した。反応終了後、該反応混合物に1mol/L塩酸5mlを加え、クロロホルムにて抽出(5ml×3)した。得られた有機層を無水硫酸ナトリウムで脱水・乾燥した後、減圧下にて溶媒を留去した。得られた残留物を中圧分取液体クロマトグラフィー(n−ヘキサン:酢酸エチル=100:0〜0:100のグラジエント)にて精製することにより目的物50mgを淡黄色油状物として得た。
1H-NMR(CDCl3) :δ8.66(dd, J=7.2, 0.9Hz, 1H), 8.15-8.10(m, 1H), 7.12(dd, J=7.2, 1.7Hz, 1H), 4.52(q, J=7.2Hz, 2H), 3.00(q, J=7.4Hz, 2H), 1.48(t, J=7.2Hz, 3H), 1.20(q, J=7.4Hz, 3H).
本発明化合物及び合成中間体は、前記製造法及び合成例に準じて合成することができる。合成例1〜合成例11及び中間体合成例1〜中間体合成例5と同様に製造した縮合複素環化合物の例を、第5表〜第10表に示すが、本発明に包含される縮合複素環化合物はこれらのみに限定されるものではない。
Intermediate Synthesis Example 1: Synthesis of 3- (ethylthio) -7- (trifluoromethyl) imidazo [1,2-a] pyridine-2-carboxylic acid Step 1: 3-iodo-7- (trifluoromethyl) imidazo Synthesis of ethyl [1,2-a] pyridine-2-carboxylate of 3.73 g of ethyl 7- (trifluoromethyl) imidazo [1,2-a] pyridine-2-carboxylate and 20 ml of N, N-dimethylformamide. To the mixed solution, 6.5 g of N-iodosuccinimide was added at room temperature. After completion of the addition, the reaction mixture was stirred at 80 ° C. for 5 hours. After completion of the reaction, water was added to the reaction mixture, and the precipitated solid was separated by filtration. The obtained solid was dissolved in 20 ml of chloroform, and washed with a saturated aqueous sodium thiosulfate solution and then with saturated sodium hydrogen carbonate. The obtained organic layer was dehydrated and dried over anhydrous sodium sulfate, and then the solvent was distilled off under reduced pressure to obtain 5.08 g of the desired product as a pale yellow solid.
Melting point: 183-185 ° C
1 H-NMR (CDCl 3 ): δ8.40 (d, J = 7.2Hz, 1H), 7.99 (s, 1H), 7.17 (dd, J = 7.2, 1.7Hz, 1H), 4.53 (q, J = 7.2Hz, 2H), 1.50 (t, J = 7.2Hz, 3H).
Step 2: Synthesis of ethyl 3- (ethylthio) -7- (trifluoromethyl) imidazo [1,2-a] pyridine-2-carboxylate 3-Iodo-7- (trifluoromethyl) imidazo [1,2- a] To a mixed solution of 5.73 g of ethyl pyridine-2-carboxylate and 40 ml of 1,4-dioxane at room temperature, 5.79 g of diisopropylethylamine, 4,5-bis (diphenylphosphino) -9,9-dimethyl 862 mg of xanthene, 682 mg of tris (dibenzylideneacetone) dipalladium (zero valent), and 1.85 g of ethanethiol were sequentially added. After completion of the addition, the inside of the reaction vessel was replaced with nitrogen gas, and then stirred for 2 hours under heating and reflux. After completion of the reaction, the reaction mixture was filtered using celite, and the celite was washed with 30 ml of chloroform. The obtained filtrate and washing solution were combined and the solvent was distilled off under reduced pressure. The obtained residue was purified by medium pressure preparative liquid chromatography eluting with n-hexane-ethyl acetate (gradient of 100: 0 to 50:50) to obtain 5.58 g of the objective product as a brown solid. It was.
Melting point: 50-52 ° C
1 H-NMR (CDCl 3 ): δ 8.67 (d, J = 7.4Hz, 1H), 8.02-8.00 (m, 1H), 7.15 (dd, J = 7.4, 1.8Hz, 1H), 4.52 (q, J = 7.0Hz, 2H), 2.98 (q, J = 7.5Hz, 2H), 1.48 (t, J = 7.0Hz, 3H), 1.20 (t, J = 7.5Hz, 3H).
Step 3: Synthesis of 3- (ethylthio) -7- (trifluoromethyl) imidazo [1,2-a] pyridine-2-carboxylic acid 3- (ethylthio) -7- (trifluoromethyl) imidazo [1,2 -A] To a mixed solution of 5.58 g of ethyl pyridine-2-carboxylate, 60 ml of ethanol and 30 ml of tetrahydrofuran, 10 ml of a 3 mol / L aqueous sodium hydroxide solution was added at room temperature. After the addition was complete, the reaction mixture was stirred at room temperature for 5 hours. After completion of the reaction, the solvent of the reaction mixture was distilled off under reduced pressure. A 1 mol / L hydrochloric acid aqueous solution was added to the obtained residue to adjust the pH of the aqueous layer to 2, followed by extraction with ethyl acetate (20 ml × 2). The obtained organic layer was dehydrated and dried in the order of saturated brine and then anhydrous sodium sulfate, and then the solvent was distilled off under reduced pressure to obtain 3.40 g of the desired product as a yellow solid.
Melting point: 163-171 ° C
1 H-NMR (CDCl 3 ): δ 8.69 (d, J = 7.2Hz, 1H), 8.19 (s, 1H), 7.22 (dd, J = 7.2, 1.4Hz, 1H), 3.06 (q, J = (7.4Hz, 2H), 1.23 (t, J = 7.4Hz, 3H) (CO 2 H proton signal could not be observed).
Intermediate Synthesis Example 2: Synthesis of 3- (ethylsulfonyl) -7- (trifluoromethyl) imidazo [1,2-a] pyridine-2-carbonyl chloride Step 1: 3- (ethylsulfonyl) -7- (tri Synthesis of fluoromethyl) imidazo [1,2-a] pyridine-2-carboxylic acid 5.00 g of 3- (ethylthio) -7- (trifluoromethyl) imidazo [1,2-a] pyridine-2-carboxylic acid and To a mixed solution of 43 ml of acetic acid, 4.88 g of 30% by weight hydrogen peroxide and 10 mg of sodium tungsten (VI) dihydrate were sequentially added at room temperature. After completion of the addition, the reaction mixture was stirred at 50 ° C. for 6 hours. After completion of the reaction, the solvent of the reaction mixture was distilled off under reduced pressure. The precipitated solid was washed with 30 ml of hexane and filtered off by filtration to obtain 4.39 g of the desired product as a pale yellow solid.
1 H-NMR (CDCl 3 ): δ9.41 (d, J = 7.2Hz, 1H), 8.08 (s, 1H), 7.30-7.20 (m, 1H), 3.78 (q, J = 7.2Hz, 2H) , 1.34 (t, J = 7.2Hz, 3H) (The proton signal of CO 2 H could not be observed).
Step 2: Synthesis of 3- (ethylsulfonyl) -7- (trifluoromethyl) imidazo [1,2-a] pyridine-2-carbonyl chloride 3- (ethylsulfonyl) -7- (trifluoromethyl) imidazo [1 , 2-a] To a mixed solution of 4.39 g of pyridine-2-carboxylic acid and 13.6 ml of dichloromethane, 2.3 ml of oxalyl chloride and 1 drop of N, N-dimethylformamide were sequentially added at room temperature. After the addition was complete, the reaction mixture was stirred at room temperature for 1 hour. After completion of the reaction, the solvent of the reaction mixture was distilled off under reduced pressure. The precipitated solid was washed with 30 ml of hexane and filtered off by filtration to obtain 4.52 g of the desired product as a white solid.
1 H-NMR (CDCl 3 ): δ9.39 (d, J = 7.5Hz, 1H), 8.20 (brs, 1H), 7.40-7.30 (m, 1H), 3.64 (q, J = 7.5Hz, 2H) , 1.38 (t, J = 7.5Hz, 3H).
Intermediate Synthesis Example 3: Synthesis of 3- (ethylthio) -7-iodoimidazo [1,2-a] pyridine-2-carboxylic acid Step 1: 7-iodoimidazo [1,2-a] pyridine-2-carboxylic acid Synthesis of ethyl acid To a mixed solution of 19.0 g of 2-amino-4-iodopyridine and 100 ml of ethanol, 9.4 g of sodium bicarbonate and 22.0 g of ethyl bromopyruvate were sequentially added at room temperature. The reaction mixture was stirred for 2 hours under heating to reflux. After completion of the reaction, the solvent was distilled off under reduced pressure. 100 ml of water and 100 ml of diisopropyl ether were added to the obtained residue, and the precipitated solid was removed by filtration. 100 ml of ethanol was added to the obtained solid, and the solvent was distilled off under reduced pressure. 100 ml of toluene was added to the obtained residue, and the solvent was distilled off under reduced pressure to obtain 23.4 g of the desired product as an orange solid.
1 H-NMR (CDCl 3 ): δ8.15-8.05 (m, 2H), 7.90-7.85 (m, 1H), 7.10-7.05 (m, 1H), 4.45 (q, J = 7.2Hz, 2H), 1.43 (t, J = 7.2Hz, 3H).
Step 2: Synthesis of ethyl 3- (ethylthio) -7-iodoimidazo [1,2-a] pyridine-2-carboxylate To a mixed solution of 3.38 g of N-chlorosuccinimide and 31 ml of 1,2-dichloroethane, -20 At 28 ° C., 2.28 ml of ethanethiol was added. The reaction mixture was stirred at room temperature for 4 hours. To the reaction mixture, 1.00 g of ethyl 7-iodoimidazo [1,2-a] pyridine-2-carboxylate was added at room temperature. The reaction mixture was stirred for 6 hours under heating to reflux. To the reaction mixture, a solution of 3.38 g of N-chlorosuccinimide prepared in a separate container and 2.28 ml of ethanethiol in 31 ml of 1,2-dichloroethane was added at room temperature. The reaction mixture was stirred for 5 hours under heating to reflux. After completion of the reaction, 100 ml of a saturated aqueous sodium hydrogen carbonate solution was added to the reaction mixture, and the mixture was extracted with chloroform (100 ml × 2). The obtained organic layer was dehydrated and dried over anhydrous sodium sulfate, and the solvent was distilled off under reduced pressure. The obtained residue was purified by medium pressure preparative liquid chromatography (gradient of n-hexane-ethyl acetate = 98: 2-90: 10) to obtain 880 mg of the desired product as a brown solid.
1 H-NMR (CDCl 3 ): δ 8.29 (d, J = 6.6Hz, 1H), 8.10 (d, J = 0.9Hz, 1H), 7.21 (dd, J = 7.2, 1.5Hz, 1H), 4.49 (q, J = 7.2Hz, 2H), 2.93 (q, J = 7.2Hz, 2H), 1.47 (t, J = 7.5Hz, 3H), 1.18 (t, J = 7.5Hz, 3H).
Step 3: Synthesis of 3- (ethylthio) -7-iodoimidazo [1,2-a] pyridine-2-carboxylic acid 3- (ethylthio) -7-iodoimidazo [1,2-a] pyridine-2-carboxylic acid To a mixed solution of 880 mg of ethyl acid and 9 ml of tetrahydrofuran, 3 ml of 0.6 mol / L lithium hydroxide aqueous solution was added at 0 ° C. The reaction mixture was stirred at room temperature for 3 hours. After completion of the reaction, the solvent of the reaction mixture was distilled off under reduced pressure. 10 ml of water was added to the obtained residue, and the aqueous layer was washed with 10 ml of diethyl ether. 2 mol / L hydrochloric acid was added to the obtained aqueous layer to adjust the pH of the aqueous layer to 2, followed by extraction with chloroform (10 ml × 2). The obtained organic layer was dehydrated and dried over anhydrous sodium sulfate, and the solvent was evaporated under reduced pressure to obtain 654 mg of the desired product as a yellow solid.
Melting point: 175-178 ° C
Intermediate Synthesis Example 4: Synthesis of 3- (ethylthio) -7-iodoimidazo [1,2-a] pyridine-2-carboxylic acid Step 1: 7-iodo-3-thiocyanatoimidazo [1,2-a Synthesis of ethyl pyridine-2-carboxylate 7-iodoimidazo [1,1] synthesized in the mixed solution of 184 mg of potassium thiocyanate and 3 ml of acetonitrile at 0 ° C. by the method of Step 1 of Intermediate Synthesis Example 3 2-a] 300 mg of ethyl pyridine-2-carboxylate was added sequentially. The reaction mixture was stirred at room temperature for 1 hour. The reaction mixture was stirred at 60 ° C. for 2 hours. After completion of the reaction, 5 ml of a 10% by mass aqueous sodium hydrogen sulfite solution and 10 ml of chloroform were sequentially added to the reaction mixture. The reaction mixture was filtered using celite, and the celite was washed with chloroform. The obtained filtrate and washings were combined and extracted with 10 ml of chloroform. The obtained organic layer was dehydrated and dried over anhydrous sodium sulfate, and then the solvent was distilled off under reduced pressure. The obtained residue was purified by medium pressure preparative liquid chromatography (gradient of n-hexane-ethyl acetate = 100: 5 to 50:50) to obtain 161 mg of the desired product as a white solid.
1 H-NMR (CDCl 3 ): δ 8.30-8.25 (m, 1H), 8.17 (d, J = 6.9Hz, 1H), 7.47 (dd, J = 6.9, 1.5Hz, 1H), 4.54 (q, J = 6.9Hz, 2H), 1.49 (t, J = 6.9Hz, 3H).
Step 2: Synthesis of 3- (ethylthio) -7-iodoimidazo [1,2-a] pyridine-2-carboxylic acid 7-iodo-3-thiocyanatoimidazo [1,2-a obtained in Step 1 ] To a mixed solution of 106 mg of ethyl pyridine-2-carboxylate and 2 ml of ethanol, 193 mg of an ethanol solution of 21% by mass sodium ethoxide (manufactured by Sigma-Aldrich) was added at room temperature. The reaction mixture was stirred at 60 ° C. for 30 minutes. To the reaction mixture, 109 mg of an ethanol solution of 21% by mass sodium ethoxide and 109 mg of iodoethane were sequentially added at room temperature. The reaction mixture was stirred at room temperature for 30 minutes. After completion of the reaction, the solvent was distilled off under reduced pressure. To the obtained residue, 10 ml of a 1.0 mol / L aqueous sodium hydroxide solution was added, and the aqueous layer was washed with 10 ml of chloroform. Concentrated hydrochloric acid was added to the obtained aqueous layer to adjust the pH of the aqueous layer to 3, followed by extraction with 10 ml of chloroform. The obtained organic layer was dehydrated and dried over anhydrous sodium sulfate, and then the solvent was distilled off under reduced pressure to obtain 33 mg of the desired product as a light brown solid.
1 H-NMR (CDCl 3 ): δ8.50-8.20 (m, 2H), 7.30-7.20 (m, 1H), 3.00 (q, J = 7.2Hz, 2H), 1.18 (t, J = 7.2Hz, 3H) (Proton signal of CO 2 H could not be observed).
Intermediate Synthesis Example 5: Synthesis of ethyl 7-cyano-3- (ethylthio) imidazo [1,2-a] pyridine-2-carboxylate Step 1: Ethyl 3- (ethylthio) -2- (methoxyimino) propionate Synthesis of ethyl 3-bromo-2- (methoxyimino) propionate (5.5 g) and N, N-dimethylformamide (20 ml) at 0 ° C., ethyl mercaptan (2.2 ml) and potassium carbonate (4.1 g) were sequentially added. did. The reaction mixture was stirred at room temperature for 2 hours. After completion of the reaction, 100 ml of water was added to the reaction mixture and extracted with diethyl ether (100 ml × 2). The obtained organic layer was dehydrated and dried over anhydrous sodium sulfate, and then the solvent was distilled off under reduced pressure to obtain 5.6 g of the desired product as a yellow oil.
1 H-NMR (CDCl 3 ): δ4.35 (q, J = 7.2Hz, 2H), 4.06 (s, 3H), 3.58 (s, 2H), 2.54 (q, J = 7.5Hz, 2H), 1.36 (t, J = 7.2Hz, 3H), 1.26 (t, J = 7.5Hz, 3H).
Step 2: Synthesis of ethyl 3- (ethylsulfinyl) -2- (methoxyimino) propionate Mixing of 2.0 g of ethyl 3- (ethylthio) -2- (methoxyimino) propionate obtained in Step 1 and 20 ml of chloroform To the solution, 2.6 g of 65% by mass metachloroperbenzoic acid was added at 0 ° C. The reaction mixture was stirred at 0 ° C. for 30 minutes. After completion of the reaction, 15 ml of a 10% by weight saturated aqueous sodium thiosulfate solution was added to the reaction mixture, followed by extraction with chloroform (20 ml × 3). The obtained organic layer was washed with 100 ml of saturated aqueous sodium hydrogen carbonate solution. The organic layer was dehydrated and dried over anhydrous sodium sulfate, and the solvent was distilled off under reduced pressure. The obtained residue was purified by medium pressure preparative liquid chromatography (gradient of n-hexane: ethyl acetate = 100: 0 to 0: 100) to obtain 1.7 g of the desired product as a pale yellow oil. .
1 H-NMR (CDCl 3 ): δ 4.37 (q, J = 7.2Hz, 2H), 4.15 (s, 3H), 4.10-4.00 (m, 2H), 2.85-2.65 (m, 2H), 1.45- 1.30 (m, 6H).
Separately from the desired product, 351 mg of ethyl 3- (ethylsulfonyl) -2- (methoxyimino) propionate was obtained as a pale yellow solid.
1 H-NMR (CDCl 3 ): δ 4.39 (s, 2H), 4.38 (q, J = 7.5Hz, 2H), 3.06 (s, 3H), 3.06 (q, J = 7.5Hz, 2H), 1.42 (t, J = 7.5Hz, 3H), 1.38 (t, J = 7.5Hz, 3H).
Step 3: Synthesis of ethyl 3-chloro-3- (ethylthio) -2- (methoxyimino) propionate 200 mg of ethyl 3- (ethylsulfinyl) -2- (methoxyimino) propionate obtained in Step 2 and 3 ml of dichloromethane 108 mg of thionyl chloride was added to the mixed solution at 0 ° C. The reaction mixture was stirred at room temperature for 10 minutes. After completion of the reaction, the solvent was distilled off under reduced pressure to obtain 250 mg of the desired product as a light brown oil.
1 H-NMR (CDCl 3 ): δ6.31 (s, 1H), 4.38 (q, J = 7.2Hz, 2H), 4.16 (s, 3H), 2.82 (q, J = 7.5Hz, 2H), 1.38 (t, J = 7.2Hz, 3H), 1.35 (t, J = 7.5Hz, 3H).
Step 4: Synthesis of ethyl 7-cyano-3- (ethylthio) imidazo [1,2-a] pyridine-2-carboxylate 3-Chloro-3- (ethylthio) -2- (methoxyimino obtained in Step 3 ) 94 mg of 4-cyanopyridine was added to a mixed solvent of 250 mg of ethyl propionate and 2 ml of acetonitrile at room temperature. The reaction mixture was stirred for 2 hours under heating to reflux. 116 mg of N, N-diisopropylethylamine was added to the reaction mixture, and the mixture was stirred for 1 hour with heating under reflux. After completion of the reaction, 5 ml of 1 mol / L hydrochloric acid was added to the reaction mixture, followed by extraction with chloroform (5 ml × 3). The obtained organic layer was dehydrated and dried over anhydrous sodium sulfate, and then the solvent was distilled off under reduced pressure. The obtained residue was purified by medium pressure preparative liquid chromatography (gradient of n-hexane: ethyl acetate = 100: 0 to 0: 100) to obtain 50 mg of the desired product as a pale yellow oil.
1 H-NMR (CDCl 3 ): δ 8.66 (dd, J = 7.2, 0.9Hz, 1H), 8.15-8.10 (m, 1H), 7.12 (dd, J = 7.2, 1.7Hz, 1H), 4.52 ( q, J = 7.2Hz, 2H), 3.00 (q, J = 7.4Hz, 2H), 1.48 (t, J = 7.2Hz, 3H), 1.20 (q, J = 7.4Hz, 3H).
The compounds of the present invention and synthetic intermediates can be synthesized according to the above production method and synthesis examples. Examples of the condensed heterocyclic compounds produced in the same manner as in Synthesis Examples 1 to 11 and Intermediate Synthesis Examples 1 to 5 are shown in Tables 5 to 10, but are included in the present invention. The heterocyclic compound is not limited to these.
尚、表中「Me」の記載はメチル基、「Et」の記載はエチル基、「n−Pr」の記載はノルマルプロピル基、「c−Pr」の記載はシクロプロピル基、「Ac」の表記はアセチル基を表す。また、「*」の表記は、化合物が融点のない油状物又は樹脂状の化合物であることを表す。 In the table, "Me" is a methyl group, "Et" is an ethyl group, "n-Pr" is a normal propyl group, "c-Pr" is a cyclopropyl group, and "Ac" The notation represents an acetyl group. The notation “*” represents that the compound is an oily or resinous compound having no melting point.
[第5表] [Table 5]
――――――――――――――――――――――――――――――――
No. n Y2 Y3 R2 X2 X3 X4 m.p.(℃)
――――――――――――――――――――――――――――――――
1-001a 2 H CF3 H H CF3 H 213-215
1-002a 2 H CF3 Me H CF3 H *
1-003a 2 H CF3 H CF3 H CF3 205-208
1-004a 2 H CF3 Me CF3 H CF3 *
――――――――――――――――――――――――――――――――
[第6表]
――――――――――――――――――――――――――――――――
No. n Y2 Y3 R 2 X 2 X 3 X 4 mp (℃)
――――――――――――――――――――――――――――――――
1-001a 2 H CF 3 HH CF 3 H 213-215
1-002a 2 H CF 3 Me H CF 3 H *
1-003a 2 H CF 3 H CF 3 H CF 3 205-208
1-004a 2 H CF 3 Me CF 3 H CF 3 *
――――――――――――――――――――――――――――――――
[Table 6]
――――――――――――――――――――――――――――――――――――
No. n Y2 Y3 Y4 R2 X1 X2 X3 m.p.(℃)
――――――――――――――――――――――――――――――――――――
2-001b 0 CF3 H H Me H H CF3 *
2-001a 2 CF3 H H Me H H CF3 168-170
2-002a 2 H CF3 H H H CF3 H 194-199
2-003a 2 H CF3 H n-Pr H CF3 H *
――――――――――――――――――――――――――――――――――――
[第7表]
――――――――――――――――――――――――――――――――――――
No. n Y2 Y3 Y4 R 2 X 1 X 2 X 3 mp (℃)
――――――――――――――――――――――――――――――――――――
2-001b 0 CF 3 HH Me HH CF 3 *
2-001a 2 CF 3 HH Me HH CF 3 168-170
2-002a 2 H CF 3 HHH CF 3 H 194-199
2-003a 2 H CF 3 H n-Pr H CF 3 H *
――――――――――――――――――――――――――――――――――――
[Table 7]
――――――――――――――――――――――――――――――
No. n Y2 Y3 R2 X3 m.p.(℃)
――――――――――――――――――――――――――――――
3-001a 2 H CF3 Me CF3 224-226
――――――――――――――――――――――――――――――
[第8表]
――――――――――――――――――――――――――――――
No. n Y2 Y3 R 2 X 3 mp (℃)
――――――――――――――――――――――――――――――
3-001a 2 H CF 3 Me CF 3 224-226
――――――――――――――――――――――――――――――
[Table 8]
――――――――――――――――――――――――――――――
No. n Y2 Y3 R2 X3 m.p.(℃)
――――――――――――――――――――――――――――――
4-001b 0 H CF3 Me CF3 84-86
4-001c 1 H CF3 Me CF3 153-154
4-001a 2 H CF3 Me CF3 *
4-002c 0 H I Me CF3 184-186
4-002a 2 H I Me CF3 185-188
4-003c 0 H CN Me CF3 194-195
4-003a 2 H CN Me CF3 161-165
4-004a 2 H SMe Me CF3 *
4-005a 2 H S(O)Me Me CF3 174-177
――――――――――――――――――――――――――――――
[第9表]
――――――――――――――――――――――――――――――
No. n Y2 Y3 R 2 X 3 mp (℃)
――――――――――――――――――――――――――――――
4-001b 0 H CF 3 Me CF 3 84-86
4-001c 1 H CF 3 Me CF 3 153-154
4-001a 2 H CF 3 Me CF 3 *
4-002c 0 HI Me CF 3 184-186
4-002a 2 HI Me CF 3 185-188
4-003c 0 H CN Me CF 3 194-195
4-003a 2 H CN Me CF 3 161-165
4-004a 2 H SMe Me CF 3 *
4-005a 2 HS (O) Me Me CF 3 174-177
――――――――――――――――――――――――――――――
[Table 9]
――――――――――――――――――――――――――――――
No. n Y2 Y3 R2 X6 m.p.(℃)
――――――――――――――――――――――――――――――
5-001a 2 H CF3 H F 197-200
5-002a 2 H CF3 Me F *
――――――――――――――――――――――――――――――
[第10表]
――――――――――――――――――――――――――――――
No. n Y2 Y3 R 2 X 6 mp (℃)
――――――――――――――――――――――――――――――
5-001a 2 H CF 3 HF 197-200
5-002a 2 H CF 3 Me F *
――――――――――――――――――――――――――――――
[Table 10]
――――――――――――――――――――――――――――――
No. n Y2 Y3 Y4 X1 X4 m.p.(℃)
――――――――――――――――――――――――――――――
5-021c 0 I H H NHMe CF3 90-92
5-026c 0 H CF3 H NHMe CF3 135-140
5-027c 0 H I H NHMe CF3 *
――――――――――――――――――――――――――――――
以下に本発明化合物の合成中間体の例を示す
[第11表]
――――――――――――――――――――――――――――――
No. n Y2 Y3 Y4 X 1 X 4 mp (℃)
――――――――――――――――――――――――――――――
5-021c 0 IHH NHMe CF 3 90-92
5-026c 0 H CF 3 H NHMe CF 3 135-140
5-027c 0 HIH NHMe CF 3 *
――――――――――――――――――――――――――――――
Examples of synthetic intermediates of the compounds of the present invention are shown below [Table 11]
―――――――――――――――――――――――――――
No. n Y2 Y3 Y4 Z1 m.p.(℃)
―――――――――――――――――――――――――――
i1-001c 0 CF3 H H OEt 76-78
i1-002c 0 I H H OEt 110-112
i1-002a 2 I H H OEt 202-204
i1-004c 0 Cl H H OEt 63-64
i1-007c 0 H CF3 H OEt 50-52
i1-007a 2 H CF3 H OEt 129-131
i1-008a 2 H I H OEt 124-126
i1-010c 0 H Cl H OEt 70-71
i1-012c 0 H NH2 H OEt *
i1-012a 2 H NH2 H OEt *
i1-013a 2 H NHAc H OEt *
i1-014c 0 H NO2 H OEt 234-235
i1-017c 0 H CN H OEt *
i1-020c 0 CF3 H H OH 200-201
i1-021c 0 I H H OH 214-215
i1-021a 2 I H H OH 164-166
i1-022c 0 Br H H OH 170-173
i1-023c 0 Cl H H OH 178-180
i1-026c 0 H CF3 H OH 163-171
i1-026a 2 H CF3 H OH 158-160
i1-027c 0 H I H OH 175-178
i1-027a 2 H I H OH 148-150
i1-028c 0 H Br H OH 150-152
i1-029c 0 H Cl H OH 210-212
i1-031a 2 H NH2 H OH *
―――――――――――――――――――――――――――
[第12表]
―――――――――――――――――――――――――――
No. n Y2 Y3 Y4 Z1 mp (℃)
―――――――――――――――――――――――――――
i1-001c 0 CF 3 HH OEt 76-78
i1-002c 0 IHH OEt 110-112
i1-002a 2 IHH OEt 202-204
i1-004c 0 Cl HH OEt 63-64
i1-007c 0 H CF 3 H OEt 50-52
i1-007a 2 H CF 3 H OEt 129-131
i1-008a 2 HIH OEt 124-126
i1-010c 0 H Cl H OEt 70-71
i1-012c 0 H NH 2 H OEt *
i1-012a 2 H NH 2 H OEt *
i1-013a 2 H NHAc H OEt *
i1-014c 0 H NO 2 H OEt 234-235
i1-017c 0 H CN H OEt *
i1-020c 0 CF 3 HH OH 200-201
i1-021c 0 IHH OH 214-215
i1-021a 2 IHH OH 164-166
i1-022c 0 Br HH OH 170-173
i1-023c 0 Cl HH OH 178-180
i1-026c 0 H CF 3 H OH 163-171
i1-026a 2 H CF 3 H OH 158-160
i1-027c 0 HIH OH 175-178
i1-027a 2 HIH OH 148-150
i1-028c 0 H Br H OH 150-152
i1-029c 0 H Cl H OH 210-212
i1-031a 2 H NH 2 H OH *
―――――――――――――――――――――――――――
[Table 12]
―――――――――――――――――――――――――――――
No. Y2 Y3 Y4 Z1 Z2 m.p.(℃)
―――――――――――――――――――――――――――――
i2-001 CF3 H H OEt H 84-88
i2-003 Br H H OEt H 103-104
i2-007 H CF3 H OEt H 180-182
i2-009 H Br H OEt H 154-157
i2-010 H Cl H OEt H 170-174
i2-020 CF3 H H OH H 245-250
i2-065 H I H OEt Cl 149-151
i2-115 CF3 H H OEt Br 114-116
i2-175 Cl H H OEt I 168-169
i2-178 H CF3 H OEt I 183-185
i2-181 H Cl H OEt I 160-162
i2-190 H H H OEt I 133-135
―――――――――――――――――――――――――――――
第5表〜第12表に示す本発明化合物及び合成中間体のうち、融点の記載のない化合物の1H−NMRデータを第13表に示す。尚、No.i1-031aは重水溶媒中、それ以外は重クロロホルム中で測定した。
―――――――――――――――――――――――――――――
No. Y2 Y3 Y4 Z1 Z2 mp (℃)
―――――――――――――――――――――――――――――
i2-001 CF 3 HH OEt H 84-88
i2-003 Br HH OEt H 103-104
i2-007 H CF 3 H OEt H 180-182
i2-009 H Br H OEt H 154-157
i2-010 H Cl H OEt H 170-174
i2-020 CF 3 HH OH H 245-250
i2-065 HIH OEt Cl 149-151
i2-115 CF 3 HH OEt Br 114-116
i2-175 Cl HH OEt I 168-169
i2-178 H CF 3 H OEt I 183-185
i2-181 H Cl H OEt I 160-162
i2-190 HHH OEt I 133-135
―――――――――――――――――――――――――――――
Of the compounds of the present invention and synthetic intermediates shown in Tables 5 to 12, 1 H-NMR data of compounds having no melting point are shown in Table 13. In addition, No.i1-031a was measured in a heavy water solvent, and other than that was measured in deuterated chloroform.
[第13表]
―――――――――――――――――――――――――――――――――――――
No. 1H NMR (CDCl3, Me4Si, 300MHz).
―――――――――――――――――――――――――――――――――――――
1-002a
δ9.04 (d, J=7.2Hz, 1H), 7.82 (s, 1H), 7.55-7.40 (m, 4H), 7.25-7.10 (m, 1H), 3.62 (q, J=7.5Hz, 2H), 3.55 (s, 3H), 1.37 (t, J=7.5Hz, 3H).
1-004a
δ9.02 (brs, 1H), 8.10-7.50 (m, 4H), 7.20 (brs, 1H), 3.70-3.35 (m, 5H), 1.37 (t, J=7.8Hz, 3H).
2-001b
δ8.83 (s, 1H), 8.62 (s, 1H), 7.77 (dd, J=8.7, 1.8Hz, 1H), 7.64 (d, J=9.6Hz, 1H), 7.55-7.40 (m, 2H), 3.67 (s, 3H), 2.92 (q, J=7.2Hz, 2H), 1.24 (t, J=7.2Hz, 3H).
2-003a
δ9.14 (d, J=7.5Hz, 1H), 8.59 (d, J=5.1Hz, 1H), 7.87 (brs, 1H), 7.30-7.15 (m, 3H), 4.12 (t, J=7.2Hz, 2H), 3.64 (q, J=6.9Hz, 2H), 1.85-1.65 (m, 2H), 1.38 (t, J=7.2Hz, 3H), 1.00-0.85 (m, 3H).
4-001a
δ9.16 (d, J=7.5Hz, 1H), 8.15 (s, 1H), 7.38 (d, J=7.5Hz, 1H), 3.87 (s, 3H), 3.57 (q, J=7.5Hz, 2H), 1.42 (t, J=7.5Hz, 3H).5-002a δ9.03 (d, J=7.2Hz, 1H), 7.86 (s, 1H), 7.25-7.05 (m, 3H), 6.86 (d, J=8.4Hz, 1H), 3.60 (q, J=7.5Hz, 2H), 3.49 (s, 3H), 1.36 (t, J=7.5Hz, 3H).
5-027c
δ9.00-8.90 (brs, 1H), 8.35-8.30 (m, 2H), 8.10-8.00 (m, 1H), 7.92 (d, J=1.8Hz, 1H), 7.30-7.20 (m, 1H), 5.30-5.20 (brs, 1H), 3.09 (d, J=5.1Hz, 3H), 3.01 (q, J=7.5Hz, 2H), 1.19 (t, J=7.4Hz, 3H).
i1-012c
δ8.27 (d, J=7.4Hz, 1H), 6.75-6.65 (m, 1H), 6.46 (dd, J=7.4, 1.6Hz, 1H), 4.46 (q, J=7.0Hz, 2H), 4.20-4.05 (brs, 2H), 2.87 (q, J=7.4Hz, 2H), 1.44 (t, J=7.0Hz, 3H), 1.17 (t, J=7.4Hz, 3H).
i1-012a
δ8.90 (d, J=7.2Hz, 1H), 6.76 (s, 1H), 6.58-6.48 (m, 1H), 4.48 (q, J=7.2Hz, 2H), 4.45 (brs, 2H), 3.65 (q, J=7.2Hz, 2H), 1.45 (t, J=7.2Hz, 3H), 1.33 (t, J=7.2Hz, 3H).
i1-013a
δ9.12 (d, J=8.1Hz, 1H), 8.06 (s, 1H), 7.63-7.58 (m, 1H), 7.34 (d, J=8.1Hz, 1H), 4.51 (q, J=7.2Hz, 2H), 3.70 (q, J=7.2Hz, 2H), 2.25 (s, 3H), 1.47 (t, J=7.2Hz, 3H), 1.34 (t, J=7.2Hz, 3H).
i1-017c
δ8.66 (dd, J=7.2, 0.9Hz, 1H), 8.15-8.10 (m, 1H), 7.12 (dd, J=7.2, 1.7Hz, 1H), 4.52 (q, J=7.2Hz, 2H), 3.00 (q, J=7.4Hz, 2H), 1.48 (t, J=7.2Hz, 3H), 1.20 (q, J=7.4Hz, 3H).
i1-031a
δ8.35 (d, J=6.6Hz, 1H), 6.62 (d, J=6.6Hz, 1H), 6.61 (s, H), 3.43 (q, J=7.5Hz, 2H), 1.91 (t, J=7.5Hz, 3H) (NH2及びCO2Hのプロトンのシグナルは観測できなかった).
―――――――――――――――――――――――――――――――――――――
次に、本発明化合物の有害生物防除剤としての有用性について、以下の試験例において具体的に説明するが、本発明はこれらに限定されるものではない。
[Table 13]
―――――――――――――――――――――――――――――――――――――
No. 1 H NMR (CDCl 3 , Me 4 Si, 300 MHz).
―――――――――――――――――――――――――――――――――――――
1-002a
δ9.04 (d, J = 7.2Hz, 1H), 7.82 (s, 1H), 7.55-7.40 (m, 4H), 7.25-7.10 (m, 1H), 3.62 (q, J = 7.5Hz, 2H) , 3.55 (s, 3H), 1.37 (t, J = 7.5Hz, 3H).
1-004a
δ9.02 (brs, 1H), 8.10-7.50 (m, 4H), 7.20 (brs, 1H), 3.70-3.35 (m, 5H), 1.37 (t, J = 7.8Hz, 3H).
2-001b
δ8.83 (s, 1H), 8.62 (s, 1H), 7.77 (dd, J = 8.7, 1.8Hz, 1H), 7.64 (d, J = 9.6Hz, 1H), 7.55-7.40 (m, 2H) , 3.67 (s, 3H), 2.92 (q, J = 7.2Hz, 2H), 1.24 (t, J = 7.2Hz, 3H).
2-003a
δ9.14 (d, J = 7.5Hz, 1H), 8.59 (d, J = 5.1Hz, 1H), 7.87 (brs, 1H), 7.30-7.15 (m, 3H), 4.12 (t, J = 7.2Hz , 2H), 3.64 (q, J = 6.9Hz, 2H), 1.85-1.65 (m, 2H), 1.38 (t, J = 7.2Hz, 3H), 1.00-0.85 (m, 3H).
4-001a
δ9.16 (d, J = 7.5Hz, 1H), 8.15 (s, 1H), 7.38 (d, J = 7.5Hz, 1H), 3.87 (s, 3H), 3.57 (q, J = 7.5Hz, 2H ), 1.42 (t, J = 7.5Hz, 3H) .5-002a δ9.03 (d, J = 7.2Hz, 1H), 7.86 (s, 1H), 7.25-7.05 (m, 3H), 6.86 (d , J = 8.4Hz, 1H), 3.60 (q, J = 7.5Hz, 2H), 3.49 (s, 3H), 1.36 (t, J = 7.5Hz, 3H).
5-027c
δ9.00-8.90 (brs, 1H), 8.35-8.30 (m, 2H), 8.10-8.00 (m, 1H), 7.92 (d, J = 1.8Hz, 1H), 7.30-7.20 (m, 1H), 5.30-5.20 (brs, 1H), 3.09 (d, J = 5.1Hz, 3H), 3.01 (q, J = 7.5Hz, 2H), 1.19 (t, J = 7.4Hz, 3H).
i1-012c
δ8.27 (d, J = 7.4Hz, 1H), 6.75-6.65 (m, 1H), 6.46 (dd, J = 7.4, 1.6Hz, 1H), 4.46 (q, J = 7.0Hz, 2H), 4.20 -4.05 (brs, 2H), 2.87 (q, J = 7.4Hz, 2H), 1.44 (t, J = 7.0Hz, 3H), 1.17 (t, J = 7.4Hz, 3H).
i1-012a
δ8.90 (d, J = 7.2Hz, 1H), 6.76 (s, 1H), 6.58-6.48 (m, 1H), 4.48 (q, J = 7.2Hz, 2H), 4.45 (brs, 2H), 3.65 (q, J = 7.2Hz, 2H), 1.45 (t, J = 7.2Hz, 3H), 1.33 (t, J = 7.2Hz, 3H).
i1-013a
δ9.12 (d, J = 8.1Hz, 1H), 8.06 (s, 1H), 7.63-7.58 (m, 1H), 7.34 (d, J = 8.1Hz, 1H), 4.51 (q, J = 7.2Hz , 2H), 3.70 (q, J = 7.2Hz, 2H), 2.25 (s, 3H), 1.47 (t, J = 7.2Hz, 3H), 1.34 (t, J = 7.2Hz, 3H).
i1-017c
δ8.66 (dd, J = 7.2, 0.9Hz, 1H), 8.15-8.10 (m, 1H), 7.12 (dd, J = 7.2, 1.7Hz, 1H), 4.52 (q, J = 7.2Hz, 2H) , 3.00 (q, J = 7.4Hz, 2H), 1.48 (t, J = 7.2Hz, 3H), 1.20 (q, J = 7.4Hz, 3H).
i1-031a
δ8.35 (d, J = 6.6Hz, 1H), 6.62 (d, J = 6.6Hz, 1H), 6.61 (s, H), 3.43 (q, J = 7.5Hz, 2H), 1.91 (t, J = 7.5Hz, 3H) (NH 2 and CO 2 H proton signals could not be observed).
―――――――――――――――――――――――――――――――――――――
Next, the usefulness of the compound of the present invention as a pest control agent will be specifically described in the following test examples, but the present invention is not limited thereto.
試験例1:トビイロウンカに対する殺虫試験
本発明化合物の10%乳剤(化合物によっては10%水和剤を供試)を、展着剤の入った水で希釈して、500ppm濃度の薬液を調製した。該薬液中にイネの葉鞘を約10秒間浸漬した。浸漬操作が終了した後、薬液処理された稲の葉鞘を風乾した後、試験管に入れた。この中にトビイロウンカ(Nilaparvata lugens)の3齢幼虫を、試験管当たり5頭放虫し、スポンジで蓋をして、25℃恒温室に収容した。収容してから6日後に、試験管中のトビイロウンカの死虫数を調査し、下記の計算式より死虫率を算出した。尚、試験は2連制で行なった。
Test Example 1: Insecticidal test against brown planthopper A 10% emulsion of the compound of the present invention (depending on the compound, 10% wettable powder) was diluted with water containing a spreading agent to prepare a chemical solution having a concentration of 500 ppm. Rice leaf sheaths were immersed in the chemical solution for about 10 seconds. After the dipping operation was finished, the leaf sheath of the rice treated with the chemical solution was air-dried and then placed in a test tube. In this, 5 third-instar larvae of the green planthopper (Nilaparvatta lugens) were released per test tube, covered with a sponge, and housed in a constant temperature room at 25 ° C. Six days after accommodation, the number of dead planthoppers in the test tube was examined, and the death rate was calculated from the following formula. In addition, the test was performed by 2 continuous systems.
(計算式)死虫率(%)=(死虫数/供試虫数)×100
その結果、供試した化合物の内、下記の化合物が90%以上の死虫率を示した。
本発明化合物:1-002a、4-001a、4-002a、4-003a、4-005a。
(Calculation formula) Death rate (%) = (Number of dead insects / Number of test insects) × 100
As a result, among the compounds tested, the following compounds showed a death rate of 90% or more.
The present compound: 1-002a, 4-001a, 4-002a, 4-003a, 4-005a.
試験例2:コナガに対する殺虫試験
本発明化合物の10%乳剤(化合物によっては10%水和剤を供試)を、展着剤の入った水で希釈して、500ppm濃度の薬液を調製した。該薬液中に、カンランの葉を約10秒間浸漬した。浸漬操作が終了した後、薬液処理されたカンランの葉を風乾した後、シャーレに入れた。この中にコナガ(Plutella xylostella)の3齢幼虫を、シャーレ当たり5頭放虫し、蓋をして25℃の恒温室に収容した。収容してから6日後に、シャーレ中のコナガの死虫数を調査し、試験例1と同様の計算式から死虫率を算出した。尚、試験は2連制で行なった。
Test Example 2: Insecticidal test for goldfish 10% emulsion of the compound of the present invention (depending on the compound, 10% wettable powder was tested) was diluted with water containing a spreading agent to prepare a chemical solution having a concentration of 500 ppm. In the chemical solution, kanran leaves were immersed for about 10 seconds. After the dipping operation was completed, the kanran leaves treated with the chemical solution were air-dried and then placed in a petri dish. In this, 5 third-instar larvae of Pterella xylostella were released per petri dish, covered, and housed in a thermostatic chamber at 25 ° C. Six days after housing, the number of dead moths in petri dishes was examined, and the mortality rate was calculated from the same calculation formula as in Test Example 1. In addition, the test was performed by 2 continuous systems.
その結果、供試した化合物の内、下記の化合物が90%以上の死虫率を示した。
本発明化合物:1-001a、1-002a、1-004a、2-001b、2-001a、2-002a、2-003a、4-001b、4-001c、4-001a、4-002c、4-002a、4-003a、4-005a、5-002a、5-021c、5-026c。
As a result, among the compounds tested, the following compounds showed a death rate of 90% or more.
Compounds of the present invention: 1-001a, 1-002a, 1-004a, 2-001b, 2-001a, 2-002a, 2-003a, 4-001b, 4-001c, 4-001a, 4-002c, 4- 002a, 4-003a, 4-005a, 5-002a, 5-021c, 5-026c.
試験例3:ハスモンヨトウに対する殺虫試験
本発明化合物の10%乳剤(化合物によっては10%水和剤を供試)を、展着剤の入った水で希釈して、500ppm濃度の薬液を調製した。該薬液中に、カンランの葉を約10秒間浸漬した。浸漬操作が終了した後、薬液処理されたカンランの葉を風乾した後、シャーレに入れた。この中にハスモンヨトウ(Spodoptera litura)の3齢幼虫を、シャーレ当たり5頭放虫し、蓋をして25℃の恒温室に収容した。収容してから6日後に、シャーレ中のハスモンヨトウの死虫数を調査し、試験例1と同様の計算式から死虫率を算出した。尚、試験は2連制で行なった。
Test Example 3: Insecticidal test against Spodoptera litura 10% emulsion of the compound of the present invention (depending on the compound, 10% wettable powder was tested) was diluted with water containing a spreading agent to prepare a chemical solution having a concentration of 500 ppm. In the chemical solution, kanran leaves were immersed for about 10 seconds. After the dipping operation was completed, the kanran leaves treated with the chemical solution were air-dried and then placed in a petri dish. In this, 5 third-instar larvae of Spodoptera litura were released per petri dish, covered, and housed in a thermostatic chamber at 25 ° C. Six days after being housed, the number of dead insects of Spodoptera litura in the petri dish was examined, and the death rate was calculated from the same formula as in Test Example 1. In addition, the test was performed by 2 continuous systems.
その結果、供試した化合物の内、下記の化合物が90%以上の死虫率を示した。
本発明化合物:1-001a、1-002a、1-003a、1-004a、2-001b、2-001a、2-002a、4-001a、4-002c、4-002a、5-002a。
As a result, among the compounds tested, the following compounds showed a death rate of 90% or more.
Compounds of the present invention: 1-001a, 1-002a, 1-003a, 1-004a, 2-001b, 2-001a, 2-002a, 4-001a, 4-002c, 4-002a, 5-002a.
試験例4:ミカンキイロアザミウマに対する殺虫活性
内径7cmのスチロールカップに湿った濾紙を敷き、その上に3cm角に切り取ったインゲンの葉を置き、ミカンキイロアザミウマ(Frankliniella occidentalis)の幼虫を1葉あたり20頭接種した。本発明化合物の10%乳剤(化合物によっては10%水和剤を供試)を、展着剤の入った水で希釈して、500ppm濃度の薬液を調製した。調製後の薬液を、回転式散布塔を用いてスチロールカップ当たり2.5mlずつ散布した(2.5mg/cm2)。2日後に摂取したミカンキイロアザミウマの死虫数と、インゲンの食害を調査した。死虫率は、試験例1と同様の計算式から算出した。食害度は以下のように判定した。1:0〜20%の食害、2:20〜50%の食害、3:50〜70%の食害、4:70%以上の食害。尚、試験は2連制で行なった。
Test Example 4: Insecticidal activity against citrus white thrips A wet filter paper was laid on a 7 cm inner diameter styrol cup, and 3 mm square green beans were placed on it, and 20 larvae of the citrus white thrips (Franklinella occentalis) per leaf were placed. Head inoculated. A 10% emulsion of the compound of the present invention (a 10% wettable powder was tested depending on the compound) was diluted with water containing a spreading agent to prepare a chemical solution having a concentration of 500 ppm. The prepared chemical solution was sprayed by 2.5 ml per styrene cup using a rotary spray tower (2.5 mg / cm 2 ). The number of dead citrus thrips ingested two days later and the damage of green beans were investigated. The mortality was calculated from the same calculation formula as in Test Example 1. The damage degree was determined as follows. 1: 0-20% damage, 2: 20-50% damage, 3: 50-70% damage, 4: 70% or more damage. In addition, the test was performed by 2 continuous systems.
その結果、供試した化合物の内、下記の化合物が50%以上の死虫率及び2以上の食害度を示した。
本発明化合物:1-002a、1-003a、1-004a、2-001b、2-001a、4-003c。
As a result, among the compounds tested, the following compounds showed a mortality rate of 50% or more and a food damage level of 2 or more.
The compound of the present invention: 1-002a, 1-003a, 1-004a, 2-001b, 2-001a, 4-003c.
試験例5:モモアカアブラムシに対する殺虫試験
内径3cmのガラスシャーレに湿った脱脂綿を敷き、その上に同径に切り取ったカンランの葉を置き、モモアカアブラムシ(Myzus persicae)無翅成虫を4頭放虫した。1日後、本発明化合物の10%乳剤(化合物によっては10%水和剤を供試)を、展着剤の入った水で希釈して、500ppm濃度の薬液を調製した。調製後の薬液を、回転式散布塔にて散布(2.5mg/cm2)し、蓋をして25℃恒温室に収容した。収容してから6日後に、シャーレ中のモモアカアブラムシの死虫数を調査し、試験例1と同様の計算式から死虫率を算出した。尚、試験は2連制で行なった。
Test Example 5: Insecticidal test against peach aphid Placed wet cotton wool on a glass petri dish with an inner diameter of 3 cm, placed kanran leaves cut out on the same diameter, and released four adult mosquito aphids (Myzus persicae) Insects. One day later, a 10% emulsion of the compound of the present invention (depending on the compound, 10% wettable powder was tested) was diluted with water containing a spreading agent to prepare a chemical solution having a concentration of 500 ppm. The prepared chemical was sprayed (2.5 mg / cm 2 ) in a rotary spray tower, covered, and stored in a 25 ° C. constant temperature room. Six days after housing, the number of dead peach aphids in the petri dish was examined, and the mortality rate was calculated from the same formula as in Test Example 1. In addition, the test was performed by 2 continuous systems.
その結果、供試した化合物の内、下記の化合物が90%以上の死虫率を示した。
本発明化合物:1-002a、1-004a、2-001b、2-001a、4-001b、4-001c、4-001a、4-002a、4-003a、4-005a、5-002a。
As a result, among the compounds tested, the following compounds showed a death rate of 90% or more.
Compounds of the present invention: 1-002a, 1-004a, 2-001b, 2-001a, 4-001b, 4-001c, 4-001a, 4-002a, 4-003a, 4-005a, 5-002a.
試験例6:トビイロウンカに対する浸透移行試験
本発明化合物の10%乳剤を水道水で希釈して、濃度が20ppmの薬液を調製し、イネセル苗(2本葉期)の根を浸した。7日後にイネを切り出して、試験管に入れ、この中にトビイロウンカ(Nilaparvata lugens)の3齢幼虫を、試験管当たり5頭放虫し、スポンジで蓋をして25℃恒温室に収容した。放虫してから6日後に、トビイロウンカの死虫数を調査し、試験例1と同様の計算式から死虫率を算出した。
Test Example 6: Osmotic transfer test against brown planthopper A 10% emulsion of the compound of the present invention was diluted with tap water to prepare a chemical solution having a concentration of 20 ppm, and the roots of rice cell seedlings (two leaves) were immersed. Seven days later, the rice was cut out and placed in a test tube, in which five third-instar larvae of the green planthopper (Nilaparvata lugens) were released per test tube, covered with a sponge, and housed in a thermostatic chamber at 25 ° C. Six days after the release, the number of dead planthoppers was examined, and the death rate was calculated from the same calculation formula as in Test Example 1.
その結果、供試した化合物の内、下記の化合物が90%以上の死虫率を示した。
本発明化合物:1-001a、1-002a、2-001a、4-001a、4-002a、4-003a、4-005a。
As a result, among the compounds tested, the following compounds showed a death rate of 90% or more.
The compound of the present invention: 1-001a, 1-002a, 2-001a, 4-001a, 4-002a, 4-003a, 4-005a.
試験例7:クリイロコイタマダニに対する効果試験
本発明化合物3.5mgを3.5mlのアセトンに溶解し、1000ppm濃度の薬液を調製した。該薬液350μlを、内壁表面積35cm2のガラス製容器の底面と側面とに塗布した後、アセトンを揮発させて、化合物の薄膜をガラス容器内壁に作製した。用いたガラス容器の内壁は35cm2であり、処理薬量は10μg/cm2となる。上記処理したガラス容器内に、クリイロコイタマダニ(Rhipicephalus sanguineus)の第1若虫(雌雄混合)を5頭放虫し、蓋をして25℃恒温室に収容した。放虫してから4日後に、マダニの死虫数を調査し、試験例1と同様の計算式から死虫率を算出した。
Test Example 7: Effect test on mung beetle mite 3.5 mg of the compound of the present invention was dissolved in 3.5 ml of acetone to prepare a chemical solution having a concentration of 1000 ppm. After 350 μl of the chemical solution was applied to the bottom and side surfaces of a glass container having an inner wall surface area of 35 cm 2, acetone was volatilized to produce a thin film of the compound on the inner wall of the glass container. The inner wall of the glass container used is 35 cm2, and the amount of treatment drug is 10 μg / cm2. In the treated glass container, 5 first nymphs (mixed male and female) of Rhipicophalus sanguineus were released, capped and housed in a constant temperature room at 25 ° C. Four days after the release, the number of ticks was examined, and the death rate was calculated from the same calculation formula as in Test Example 1.
その結果、供試した化合物の内、下記の化合物が50%以上の死虫率を示した。
本発明化合物:1-002a、2-002a、4-001b、4-003c、4-003a、、4-005a、5-001a、5-002a。
As a result, among the compounds tested, the following compounds showed a death rate of 50% or more.
The compounds of the present invention: 1-002a, 2-002a, 4-001b, 4-003c, 4-003a, 4-005a, 5-001a, 5-002a.
試験例8:ネコノミに対する効果試験
本発明化合物3.5mgを3.5mlのアセトンに溶解し、1000ppm濃度の薬液を調製した。該薬液350μlを、内壁表面積35cm2のガラス製容器の底面と側面とに塗布した後、アセトンを揮発させて、化合物の薄膜をガラス容器内壁に作製した。用いたガラス容器の内壁は35cm2なので、処理薬量は10μg/cm2となる。上記処理したガラス容器内に、ネコノミ(Ctenocephalides felis)の成虫(雌雄混合)を5頭放虫し、蓋をして25℃の恒温室に収容した。放虫してから4日後に、ノミの死虫数を調査し、試験例1と同様の計算式から死虫率を算出した。
Test Example 8: Effect test on cat fleas 3.5 mg of the present compound was dissolved in 3.5 ml of acetone to prepare a chemical solution having a concentration of 1000 ppm. After 350 μl of the chemical solution was applied to the bottom and side surfaces of a glass container having an inner wall surface area of 35 cm 2, acetone was volatilized to produce a thin film of the compound on the inner wall of the glass container. Since the inner wall of the used glass container is 35 cm 2, the treatment dose is 10 μg / cm 2. In the treated glass container, 5 adults (mixed male and female) of cat flea (Ctenocephalides felis) were released, covered, and housed in a thermostatic chamber at 25 ° C. Four days after releasing, the number of dead fleas was investigated, and the death rate was calculated from the same calculation formula as in Test Example 1.
その結果、供試した化合物の内、下記の化合物が50%以上の死虫率を示した。
本発明化合物:1-002a、1-004a、2-001b、4-001b、4-001c、4-001a、4-003a。
As a result, among the compounds tested, the following compounds showed a death rate of 50% or more.
The compounds of the present invention: 1-002a, 1-004a, 2-001b, 4-001b, 4-001c, 4-001a, 4-003a.
本発明における化合物は、優れた有害生物防除活性を示し、且つ、ホ乳動物、魚類及び益虫等の非標的生物に対してほとんど悪影響の無い、極めて有用な化合物である。 The compound in the present invention is an extremely useful compound that exhibits excellent pest control activity and has almost no adverse effect on non-target organisms such as mammals, fish and beneficial insects.
Claims (14)
[式中、R1は、水素原子、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルケニル、ハロ(C1〜C6)アルケニル、C1〜C6アルキニル、ハロ(C1〜C6)アルキニル、C3〜C6シクロアルキル、ハロ(C3〜C6)シクロアルキル、C3〜C6シクロアルケニル又はハロ(C3〜C6)シクロアルケニルを表し、
R2は、水素原子、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルケニル、ハロ(C1〜C6)アルケニル、C1〜C6アルキニル、ハロ(C1〜C6)アルキニル、C3〜C6シクロアルキル、ハロ(C3〜C6)シクロアルキル、C3〜C6シクロアルケニル又はハロ(C3〜C6)シクロアルケニルを表し、
Gは、G1〜G6で表される構造を表し、
Y2及びY4は、各々独立して水素原子、ハロゲン原子、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルケニル、ハロ(C1〜C6)アルケニル、C1〜C6アルキニル、ハロ(C1〜C6)アルキニル、C3〜C6シクロアルキル、ハロ(C3〜C6)シクロアルキル、C3〜C6シクロアルケニル、ハロ(C3〜C6)シクロアルケニル、−OR40、−N(R80)R81、−C(O)OR70、−S(O)mR50、シアノ又はニトロを表し、
Y3は、水素原子、ハロゲン原子、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルケニル、ハロ(C1〜C6)アルケニル、C1〜C6アルキニル、ハロ(C1〜C6)アルキニル、C3〜C6シクロアルキル、ハロ(C3〜C6)シクロアルキル、C3〜C6シクロアルケニル、ハロ(C3〜C6)シクロアルケニル、−OR40、−N(R60)R61、−C(O)OR70、−S(O)mR50、シアノ又はニトロを表し、
X1、X2、X3、X4及びX6は、各々独立して水素原子、ハロゲン原子、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルケニル、ハロ(C1〜C6)アルケニル、C1〜C6アルキニル、ハロ(C1〜C6)アルキニル、C3〜C6シクロアルキル、ハロ(C3〜C6)シクロアルキル、C3〜C6シクロアルケニル、ハロ(C3〜C6)シクロアルケニル、−OR90、−N(R10)R11、−C(O)OR70、−S(O)pR30、シアノ又はニトロを表し、
R10は、水素原子、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルケニル、ハロ(C1〜C6)アルケニル、C1〜C6アルキニル、ハロ(C1〜C6)アルキニル、C3〜C6シクロアルキル、ハロ(C3〜C6)シクロアルキル、C3〜C6シクロアルケニル、ハロ(C3〜C6)シクロアルケニル、C1〜C6アルキルカルボニル、ハロ(C1〜C6)アルキルカルボニル、C1〜C6アルコキシ、ハロ(C1〜C6)アルコキシ、C1〜C6アルコキシカルボニル、ハロ(C1〜C6)アルコキシカルボニル又はC1〜C6アルキルスルホニルを表し、
R11は、水素原子、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルキルカルボニル、ハロ(C1〜C6)アルキルカルボニル、C1〜C6アルコキシカルボニル、ハロ(C1〜C6)アルコキシカルボニル又はC1〜C6アルキルスルホニルを表し、
R30は、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルケニル、ハロ(C1〜C6)アルケニル、C1〜C6アルキニル、ハロ(C1〜C6)アルキニル
C3〜C6シクロアルキル、ハロ(C3〜C6)シクロアルキル、C3〜C6シクロアルケニル又はハロ(C3〜C6)シクロアルケニルを表し、
R40は、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルケニル、ハロ(C1〜C6)アルケニル、C1〜C6アルキニル、ハロ(C1〜C6)アルキニル、C3〜C6シクロアルキル、ハロ(C3〜C6)シクロアルキル、C3〜C6シクロアルケニル又はハロ(C3〜C6)シクロアルケニルを表し、
R50は、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルケニル、ハロ(C1〜C6)アルケニル、C1〜C6アルキニル、ハロ(C1〜C6)アルキニル、C3〜C6シクロアルキル、ハロ(C3〜C6)シクロアルキル、C3〜C6シクロアルケニル、ハロ(C3〜C6)シクロアルケニル又はC1〜C10アルコキシカルボニル(C1〜C6)アルキルを表し、
R60は、水素原子、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルケニル、ハロ(C1〜C6)アルケニル、C1〜C6アルキニル、ハロ(C1〜C6)アルキニル、C3〜C6シクロアルキル、ハロ(C3〜C6)シクロアルキル、C3〜C6シクロアルケニル、ハロ(C3〜C6)シクロアルケニル、C1〜C6アルキルカルボニル、ハロ(C1〜C6)アルキルカルボニル、C1〜C6アルコキシ、ハロ(C1〜C6)アルコキシ、C1〜C6アルコキシカルボニル、ハロ(C1〜C6)アルコキシカルボニル又はC1〜C6アルキルスルホニルを表し、
R61は、水素原子、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルキルカルボニル、ハロ(C1〜C6)アルキルカルボニル、C1〜C6アルコキシカルボニル、ハロ(C1〜C6)アルコキシカルボニル又はC1〜C6アルキルスルホニルを表し、
R70は、水素原子、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルケニル、ハロ(C1〜C6)アルケニル、C1〜C6アルキニル、ハロ(C1〜C6)アルキニル、C3〜C6シクロアルキル、ハロ(C3〜C6)シクロアルキル、C3〜C6シクロアルケニル又はハロ(C3〜C6)シクロアルケニルを表し、
R80は、水素原子、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルケニル、ハロ(C1〜C6)アルケニル、C1〜C6アルキニル、ハロ(C1〜C6)アルキニル、C3〜C6シクロアルキル、ハロ(C3〜C6)シクロアルキル、C3〜C6シクロアルケニル、ハロ(C3〜C6)シクロアルケニル、C1〜C6アルキルカルボニル、ハロ(C1〜C6)アルキルカルボニル、C1〜C6アルコキシ、ハロ(C1〜C6)アルコキシ、C1〜C6アルコキシカルボニル、ハロ(C1〜C6)アルコキシカルボニル又はC1〜C6アルキルスルホニルを表し、
R81は、水素原子、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルキルカルボニル、ハロ(C1〜C6)アルキルカルボニル、C1〜C6アルコキシカルボニル、ハロ(C1〜C6)アルコキシカルボニル又はC1〜C6アルキルスルホニルを表し、
R90は、C1〜C6アルキル、ハロ(C1〜C6)アルキル、C1〜C6アルケニル、ハロ(C1〜C6)アルケニル、C1〜C6アルキニル、ハロ(C1〜C6)アルキニル、C3〜C6シクロアルキル、ハロ(C3〜C6)シクロアルキル、C3〜C6シクロアルケニル又はハロ(C3〜C6)シクロアルケニルを表し、
nは、0、1又は2の整数を表し、
mは、0、1又は2の整数を表し、
pは、0、1又は2の整数を表す。]で表される縮合複素環化合物若しくはその塩又はそれらのN−オキシド。 Formula (1):
[Wherein, R 1 is a hydrogen atom, C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkenyl, halo (C 1 -C 6 ) alkenyl, C 1 -C 6 Alkynyl, halo (C 1 -C 6 ) alkynyl, C 3 -C 6 cycloalkyl, halo (C 3 -C 6 ) cycloalkyl, C 3 -C 6 cycloalkenyl or halo (C 3 -C 6 ) cycloalkenyl Represent,
R 2 is a hydrogen atom, C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkenyl, halo (C 1 -C 6 ) alkenyl, C 1 -C 6 alkynyl, halo ( C 1 -C 6 ) alkynyl, C 3 -C 6 cycloalkyl, halo (C 3 -C 6 ) cycloalkyl, C 3 -C 6 cycloalkenyl or halo (C 3 -C 6 ) cycloalkenyl,
G represents a structure represented by G 1 to G 6 ;
Y2 and Y4 are each independently a hydrogen atom, a halogen atom, C 1 -C 6 alkyl, halo (C 1 ~C 6) alkyl, C 1 -C 6 alkenyl, halo (C 1 ~C 6) alkenyl, C 1 -C 6 alkynyl, halo (C 1 ~C 6) alkynyl, C 3 -C 6 cycloalkyl, halo (C 3 ~C 6) cycloalkyl, C 3 -C 6 cycloalkenyl, halo (C 3 -C 6 ) Represents cycloalkenyl, —OR 40 , —N (R 80 ) R 81 , —C (O) OR 70 , —S (O) m R 50 , cyano or nitro;
Y3 is hydrogen atom, halogen atom, C 1 -C 6 alkyl, halo (C 1 ~C 6) alkyl, C 1 -C 6 alkenyl, halo (C 1 ~C 6) alkenyl, C 1 -C 6 alkynyl, Halo (C 1 -C 6 ) alkynyl, C 3 -C 6 cycloalkyl, halo (C 3 -C 6 ) cycloalkyl, C 3 -C 6 cycloalkenyl, halo (C 3 -C 6 ) cycloalkenyl, -OR 40, -N (R 60) R 61, -C (O) oR 70, -S (O) m R 50, cyano or nitro,
X 1 , X 2 , X 3 , X 4 and X 6 are each independently a hydrogen atom, a halogen atom, C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkenyl, Halo (C 1 -C 6 ) alkenyl, C 1 -C 6 alkynyl, halo (C 1 -C 6 ) alkynyl, C 3 -C 6 cycloalkyl, halo (C 3 -C 6 ) cycloalkyl, C 3 -C Represents 6 cycloalkenyl, halo (C 3 -C 6 ) cycloalkenyl, —OR 90 , —N (R 10 ) R 11 , —C (O) OR 70 , —S (O) p R 30 , cyano or nitro. ,
R 10 is a hydrogen atom, C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkenyl, halo (C 1 -C 6 ) alkenyl, C 1 -C 6 alkynyl, halo ( C 1 -C 6) alkynyl, C 3 -C 6 cycloalkyl, halo (C 3 ~C 6) cycloalkyl, C 3 -C 6 cycloalkenyl, halo (C 3 ~C 6) cycloalkenyl, C 1 -C 6 alkylcarbonyl, halo (C 1 -C 6 ) alkylcarbonyl, C 1 -C 6 alkoxy, halo (C 1 -C 6 ) alkoxy, C 1 -C 6 alkoxycarbonyl, halo (C 1 -C 6 ) alkoxycarbonyl or represents C 1 -C 6 alkylsulfonyl,
R 11 is a hydrogen atom, C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkylcarbonyl, halo (C 1 -C 6 ) alkylcarbonyl, C 1 -C 6 alkoxycarbonyl , Halo (C 1 -C 6 ) alkoxycarbonyl or C 1 -C 6 alkylsulfonyl,
R 30 is C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkenyl, halo (C 1 -C 6 ) alkenyl, C 1 -C 6 alkynyl, halo (C 1- C 6 ) represents alkynyl C 3 -C 6 cycloalkyl, halo (C 3 -C 6 ) cycloalkyl, C 3 -C 6 cycloalkenyl or halo (C 3 -C 6 ) cycloalkenyl,
R 40 is C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkenyl, halo (C 1 -C 6 ) alkenyl, C 1 -C 6 alkynyl, halo (C 1- C 6 ) represents alkynyl, C 3 -C 6 cycloalkyl, halo (C 3 -C 6 ) cycloalkyl, C 3 -C 6 cycloalkenyl or halo (C 3 -C 6 ) cycloalkenyl,
R 50 is C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkenyl, halo (C 1 -C 6 ) alkenyl, C 1 -C 6 alkynyl, halo (C 1- C 6) alkynyl, C 3 -C 6 cycloalkyl, halo (C 3 ~C 6) cycloalkyl, C 3 -C 6 cycloalkenyl, halo (C 3 ~C 6) cycloalkenyl or C 1 -C 10 alkoxycarbonyl (C 1 ~C 6) alkyl,
R 60 is a hydrogen atom, C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkenyl, halo (C 1 -C 6 ) alkenyl, C 1 -C 6 alkynyl, halo ( C 1 -C 6) alkynyl, C 3 -C 6 cycloalkyl, halo (C 3 ~C 6) cycloalkyl, C 3 -C 6 cycloalkenyl, halo (C 3 ~C 6) cycloalkenyl, C 1 -C 6 alkylcarbonyl, halo (C 1 -C 6 ) alkylcarbonyl, C 1 -C 6 alkoxy, halo (C 1 -C 6 ) alkoxy, C 1 -C 6 alkoxycarbonyl, halo (C 1 -C 6 ) alkoxycarbonyl or represents C 1 -C 6 alkylsulfonyl,
R 61 is a hydrogen atom, C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkylcarbonyl, halo (C 1 -C 6 ) alkylcarbonyl, C 1 -C 6 alkoxycarbonyl , Halo (C 1 -C 6 ) alkoxycarbonyl or C 1 -C 6 alkylsulfonyl,
R 70 represents a hydrogen atom, C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkenyl, halo (C 1 -C 6 ) alkenyl, C 1 -C 6 alkynyl, halo ( C 1 -C 6 ) alkynyl, C 3 -C 6 cycloalkyl, halo (C 3 -C 6 ) cycloalkyl, C 3 -C 6 cycloalkenyl or halo (C 3 -C 6 ) cycloalkenyl,
R 80 is a hydrogen atom, C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkenyl, halo (C 1 -C 6 ) alkenyl, C 1 -C 6 alkynyl, halo ( C 1 -C 6) alkynyl, C 3 -C 6 cycloalkyl, halo (C 3 ~C 6) cycloalkyl, C 3 -C 6 cycloalkenyl, halo (C 3 ~C 6) cycloalkenyl, C 1 -C 6 alkylcarbonyl, halo (C 1 -C 6 ) alkylcarbonyl, C 1 -C 6 alkoxy, halo (C 1 -C 6 ) alkoxy, C 1 -C 6 alkoxycarbonyl, halo (C 1 -C 6 ) alkoxycarbonyl or represents C 1 -C 6 alkylsulfonyl,
R 81 is a hydrogen atom, C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkylcarbonyl, halo (C 1 -C 6 ) alkylcarbonyl, C 1 -C 6 alkoxycarbonyl , Halo (C 1 -C 6 ) alkoxycarbonyl or C 1 -C 6 alkylsulfonyl,
R 90 is C 1 -C 6 alkyl, halo (C 1 -C 6 ) alkyl, C 1 -C 6 alkenyl, halo (C 1 -C 6 ) alkenyl, C 1 -C 6 alkynyl, halo (C 1- C 6 ) represents alkynyl, C 3 -C 6 cycloalkyl, halo (C 3 -C 6 ) cycloalkyl, C 3 -C 6 cycloalkenyl or halo (C 3 -C 6 ) cycloalkenyl,
n represents an integer of 0, 1 or 2;
m represents an integer of 0, 1 or 2;
p represents an integer of 0, 1 or 2. ] The condensed heterocyclic compound or its salt represented by these, or those N-oxides.
R2は、水素原子又はC1〜C6アルキルを表し、
Y2及びY4は、各々独立して水素原子、ハロゲン原子、ハロ(C1〜C6)アルキル、C3〜C6シクロアルキル、−N(R80)R81、−C(O)OR70、−S(O)mR50、シアノ又はニトロを表し、
Y3は、水素原子、ハロゲン原子、ハロ(C1〜C6)アルキル、C3〜C6シクロアルキル、−N(R60)R61、−C(O)OR70、−S(O)mR50、シアノ又はニトロを表し、
X1、X2、X3、X4及びX6は、各々独立して水素原子、ハロゲン原子、ハロ(C1〜C6)アルキル、−N(R10)R11又はニトロを表し、
R10は、水素原子又はC1〜C6アルキルを表し、
R11は、水素原子を表し、
R50は、C1〜C6アルキル又はC1〜C10アルコキシカルボニル(C1〜C6)アルキルを表し、
R60は、水素原子、C1〜C6アルキルカルボニル、C1〜C6アルコキシカルボニル又はC1〜C6アルキルスルホニルを表し、
R61は、水素原子、C1〜C6アルキルカルボニル、C1〜C6アルコキシカルボニル又はC1〜C6アルキルスルホニルを表し、
R70は、水素原子又はC1〜C6アルキルを表し、
R80は、水素原子、C1〜C6アルキルカルボニル、C1〜C6アルコキシカルボニル又はC1〜C6アルキルスルホニルを表し、
R81は、水素原子、C1〜C6アルキルカルボニル、C1〜C6アルコキシカルボニル又はC1〜C6アルキルスルホニルを表す請求項1記載の縮合複素環化合物若しくはその塩又はそれらのN−オキシド。 R 1 represents C 1 -C 6 alkyl;
R 2 represents a hydrogen atom or C 1 -C 6 alkyl,
Y2 and Y4 are each independently a hydrogen atom, a halogen atom, halo (C 1 ~C 6) alkyl, C 3 -C 6 cycloalkyl, -N (R 80) R 81 , -C (O) OR 70, Represents —S (O) m R 50 , cyano or nitro,
Y3 represents a hydrogen atom, a halogen atom, halo (C 1 ~C 6) alkyl, C 3 -C 6 cycloalkyl, -N (R 60) R 61 , -C (O) OR 70, -S (O) m R 50 represents cyano or nitro,
X 1 , X 2 , X 3 , X 4 and X 6 each independently represent a hydrogen atom, a halogen atom, halo (C 1 -C 6 ) alkyl, —N (R 10 ) R 11 or nitro,
R 10 represents a hydrogen atom or C 1 -C 6 alkyl,
R 11 represents a hydrogen atom,
R 50 represents C 1 -C 6 alkyl or C 1 -C 10 alkoxycarbonyl (C 1 -C 6 ) alkyl;
R 60 represents a hydrogen atom, C 1 -C 6 alkylcarbonyl, C 1 -C 6 alkoxycarbonyl or C 1 -C 6 alkylsulfonyl,
R 61 represents a hydrogen atom, C 1 -C 6 alkylcarbonyl, C 1 -C 6 alkoxycarbonyl or C 1 -C 6 alkylsulfonyl,
R 70 represents a hydrogen atom or C 1 -C 6 alkyl,
R 80 represents a hydrogen atom, C 1 -C 6 alkylcarbonyl, C 1 -C 6 alkoxycarbonyl or C 1 -C 6 alkylsulfonyl,
The condensed heterocyclic compound or a salt thereof or an N-oxide thereof according to claim 1, wherein R 81 represents a hydrogen atom, C 1 -C 6 alkylcarbonyl, C 1 -C 6 alkoxycarbonyl, or C 1 -C 6 alkylsulfonyl. .
Y3は、水素原子、ハロゲン原子、ハロ(C1〜C6)アルキル、C3〜C6シクロアルキル、−N(R60)R61、−C(O)OR70、−S(O)mR50、シアノ又はニトロを表し、
Y4は、水素原子又は−N(R80)R81を表し、
X1は、水素原子、ハロゲン原子、−N(R10)R11又はニトロを表し、
X2は、水素原子又はハロ(C1〜C6)アルキルを表し、
X3は、水素原子又はハロ(C1〜C6)アルキルを表し、
X4は、水素原子又はハロ(C1〜C6)アルキルを表し、
X6は、ハロゲン原子を表し、
R60は、水素原子又はC1〜C6アルキルカルボニルを表し、
R61は、水素原子を表し、
R80は、水素原子を表し、
R81は、水素原子を表し、
mは、0又は1の整数を表す請求項2記載の縮合複素環化合物若しくはその塩又はそれらのN−オキシド。 Y2 represents a hydrogen atom, a halogen atom, halo (C 1 ~C 6) alkyl, a C 3 -C 6 cycloalkyl or -N (R 80) R 81,
Y3 represents a hydrogen atom, a halogen atom, halo (C 1 ~C 6) alkyl, C 3 -C 6 cycloalkyl, -N (R 60) R 61 , -C (O) OR 70, -S (O) m R 50 represents cyano or nitro,
Y4 represents a hydrogen atom or -N (R 80) R 81,
X 1 represents a hydrogen atom, a halogen atom, —N (R 10 ) R 11 or nitro,
X 2 represents a hydrogen atom or halo (C 1 -C 6 ) alkyl,
X 3 represents a hydrogen atom or halo (C 1 -C 6 ) alkyl,
X 4 represents a hydrogen atom or halo (C 1 -C 6 ) alkyl,
X 6 represents a halogen atom,
R 60 represents a hydrogen atom or a C 1 -C 6 alkylcarbonyl,
R 61 represents a hydrogen atom,
R 80 represents a hydrogen atom,
R 81 represents a hydrogen atom,
The fused heterocyclic compound or a salt thereof, or an N-oxide thereof according to claim 2, wherein m represents an integer of 0 or 1.
Y3は、水素原子、ハロゲン原子、ハロ(C1〜C6)アルキル、−S(O)mR50又はシアノを表し、
Y4は、水素原子を表し、
X1は、水素原子又は−N(R10)R11を表し、
R10は、C1〜C6アルキルを表し、
R50は、C1〜C6アルキルを表す請求項3記載の縮合複素環化合物若しくはその塩又はそれらのN−オキシド。 Y2 represents a hydrogen atom, a halogen atom or a halo (C 1 ~C 6) alkyl,
Y3 represents a hydrogen atom, a halogen atom, halo (C 1 ~C 6) alkyl, -S and (O) m R 50 or cyano,
Y4 represents a hydrogen atom,
X 1 represents a hydrogen atom or —N (R 10 ) R 11 ,
R 10 represents C 1 -C 6 alkyl,
The condensed heterocyclic compound or a salt thereof, or an N-oxide thereof according to claim 3, wherein R 50 represents C 1 to C 6 alkyl.
X4は、ハロ(C1〜C6)アルキルを表し、
nは、0又は2の整数を表す請求項4記載の縮合複素環化合物若しくはその塩又はそれらのN−オキシド。 Y3 represents a hydrogen atom, a halogen atom or a halo (C 1 ~C 6) alkyl,
X 4 represents halo (C 1 -C 6 ) alkyl;
5. The condensed heterocyclic compound or a salt thereof or an N-oxide thereof according to claim 4, wherein n represents 0 or an integer of 2.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2016154907 | 2016-08-05 | ||
| JP2016154907 | 2016-08-05 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| JP2018024660A true JP2018024660A (en) | 2018-02-15 |
Family
ID=61193953
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2017151963A Pending JP2018024660A (en) | 2016-08-05 | 2017-08-04 | Amide compound and pest control agent |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP2018024660A (en) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPWO2020251013A1 (en) * | 2019-06-13 | 2020-12-17 | ||
| WO2022071434A1 (en) * | 2020-09-30 | 2022-04-07 | 住友化学株式会社 | Heterocyclic compound and harmful arthropod-controlling composition containing same |
| WO2024071393A1 (en) * | 2022-09-30 | 2024-04-04 | 住友化学株式会社 | Heterocyclic compound and harmful arthropod-controlling composition containing same |
| WO2024080330A1 (en) | 2022-10-13 | 2024-04-18 | 住友化学株式会社 | Sulfonamide compound and harmful-arthropod controlling composition containing same |
-
2017
- 2017-08-04 JP JP2017151963A patent/JP2018024660A/en active Pending
Cited By (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP7543264B2 (en) | 2019-06-13 | 2024-09-02 | アグロカネショウ株式会社 | Agricultural and horticultural agents containing novel imidazopyridine-2-carboxamide derivatives as active ingredients |
| US12279617B2 (en) | 2019-06-13 | 2025-04-22 | Agro-Kanesho Co., Ltd. | Agricultural and horticultural chemical agent containing new imidazopyridine-2-carboxamide derivative as active ingredient |
| CN114008045A (en) * | 2019-06-13 | 2022-02-01 | 阿格罗-金正株式会社 | Agrohorticultural agent containing novel imidazopyridine-2-carboxamide derivative as active ingredient |
| KR20220024013A (en) | 2019-06-13 | 2022-03-03 | 아구로카네쇼 가부시키가이샤 | Agro-horticultural drug containing novel imidazopyridine-2-carboxamide derivative as an active ingredient |
| JPWO2020251013A1 (en) * | 2019-06-13 | 2020-12-17 | ||
| WO2020251013A1 (en) | 2019-06-13 | 2020-12-17 | アグロカネショウ株式会社 | Agricultural and horticultural chemical agent having novel imidazopyridine-2-carboxamide derivative as active ingredient |
| AU2020290268B2 (en) * | 2019-06-13 | 2025-07-17 | Agro-Kanesho Co., Ltd. | Agricultural and horticultural chemical agent having novel imidazopyridine-2-carboxamide derivative as active ingredient |
| JPWO2022071434A1 (en) * | 2020-09-30 | 2022-04-07 | ||
| WO2022071434A1 (en) * | 2020-09-30 | 2022-04-07 | 住友化学株式会社 | Heterocyclic compound and harmful arthropod-controlling composition containing same |
| CN116456818A (en) * | 2020-09-30 | 2023-07-18 | 住友化学株式会社 | Heterocyclic compound and harmful arthropod control composition containing same |
| JP7704770B2 (en) | 2020-09-30 | 2025-07-08 | 住友化学株式会社 | Heterocyclic compound and harmful arthropod control composition containing the same |
| CN116456818B (en) * | 2020-09-30 | 2025-12-05 | 住友化学株式会社 | Heterocyclic compounds and harmful arthropod control compositions containing such heterocyclic compounds |
| WO2024071393A1 (en) * | 2022-09-30 | 2024-04-04 | 住友化学株式会社 | Heterocyclic compound and harmful arthropod-controlling composition containing same |
| WO2024080330A1 (en) | 2022-10-13 | 2024-04-18 | 住友化学株式会社 | Sulfonamide compound and harmful-arthropod controlling composition containing same |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US10759815B2 (en) | Condensed heterocyclic compounds and pesticides | |
| JP5206993B2 (en) | Isoxazoline-substituted benzamide compounds and pest control agents | |
| JP6781404B2 (en) | Condensed heterocyclic compounds and pest control agents | |
| JP2012188418A (en) | Triazole derivative, and pest control agent | |
| JP2013075871A (en) | Thiazole derivative and pest controller | |
| JP2013107867A (en) | Pyrazole derivative and pest controlling agent | |
| JP2013082699A (en) | Pyrazole derivative and pest control agent | |
| JP2010083883A (en) | Isoxazoline-substituted benzamide compound and harmful organism-controlling agent | |
| JP2018024672A (en) | Condensed heterocyclic compound and pest control agent | |
| JP2014208631A (en) | Pyridine derivative and pest control agent | |
| TW202124378A (en) | Condensed heterocyclic compound and noxious organism control agent capable of effectively eliminating insects or mites | |
| JP2018024658A (en) | Condensed heterocyclic compound and pest control agent | |
| JP2018024660A (en) | Amide compound and pest control agent | |
| JP2019043944A (en) | Condensed heterocyclic compound and pest control agent | |
| JP2018024657A (en) | Condensed heterocyclic compound and pest control agent | |
| JP2018070585A (en) | Condensed heterocyclic compound and pest control agent | |
| JP2015147757A (en) | Heterocyclic derivative and pest control agent | |
| TWI705066B (en) | Condensed heterocyclic compounds and pest control agents | |
| JP2018027943A (en) | Condensed heterocyclic compound and pest controlling agent | |
| JP2018090578A (en) | Condensed heterocyclic compound and pest control agent | |
| JP2015003895A (en) | Thiazole derivative and pest control agent | |
| JP2019182855A (en) | Condensed heterocyclic compound and pest control agent | |
| JP2018030834A (en) | Condensed heterocyclic compound and pest control agent | |
| JP2017186327A (en) | Condensed heterocyclic compound and pest control agent | |
| JP2014034539A (en) | Optically active pyrazole amide derivative and pest control agent |