JP2010000078A - Process for producing plant activator, activation method, activity promoter and method for applying the promoter - Google Patents
Process for producing plant activator, activation method, activity promoter and method for applying the promoter Download PDFInfo
- Publication number
- JP2010000078A JP2010000078A JP2009174204A JP2009174204A JP2010000078A JP 2010000078 A JP2010000078 A JP 2010000078A JP 2009174204 A JP2009174204 A JP 2009174204A JP 2009174204 A JP2009174204 A JP 2009174204A JP 2010000078 A JP2010000078 A JP 2010000078A
- Authority
- JP
- Japan
- Prior art keywords
- aib
- component
- molecular weight
- ala
- medium
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 230000000694 effects Effects 0.000 title claims abstract description 59
- 238000000034 method Methods 0.000 title claims abstract description 37
- 230000004913 activation Effects 0.000 title abstract description 4
- 230000008569 process Effects 0.000 title abstract description 4
- 239000005962 plant activator Substances 0.000 title abstract 2
- 241000223260 Trichoderma harzianum Species 0.000 claims abstract description 46
- 239000007787 solid Substances 0.000 claims abstract description 10
- 239000002689 soil Substances 0.000 claims description 50
- 239000000126 substance Substances 0.000 claims description 36
- 241000233866 Fungi Species 0.000 claims description 16
- 239000003795 chemical substances by application Substances 0.000 claims description 16
- 239000007788 liquid Substances 0.000 claims description 16
- 239000002245 particle Substances 0.000 claims description 13
- 238000004519 manufacturing process Methods 0.000 claims description 11
- 239000000463 material Substances 0.000 claims description 6
- 235000015097 nutrients Nutrition 0.000 claims description 4
- 238000005273 aeration Methods 0.000 claims description 2
- 230000035699 permeability Effects 0.000 claims description 2
- 238000012258 culturing Methods 0.000 abstract description 6
- 239000002609 medium Substances 0.000 description 96
- 241000196324 Embryophyta Species 0.000 description 69
- 241000894006 Bacteria Species 0.000 description 49
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 48
- 230000012010 growth Effects 0.000 description 41
- 230000000844 anti-bacterial effect Effects 0.000 description 24
- 238000012360 testing method Methods 0.000 description 21
- 240000007594 Oryza sativa Species 0.000 description 19
- 235000007164 Oryza sativa Nutrition 0.000 description 19
- 235000009566 rice Nutrition 0.000 description 19
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 18
- 238000010521 absorption reaction Methods 0.000 description 18
- 239000000047 product Substances 0.000 description 18
- 239000000284 extract Substances 0.000 description 17
- 238000004949 mass spectrometry Methods 0.000 description 17
- FUOOLUPWFVMBKG-UHFFFAOYSA-N 2-Aminoisobutyric acid Chemical compound CC(C)(N)C(O)=O FUOOLUPWFVMBKG-UHFFFAOYSA-N 0.000 description 15
- 238000000746 purification Methods 0.000 description 15
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 15
- 238000004128 high performance liquid chromatography Methods 0.000 description 14
- 241000813090 Rhizoctonia solani Species 0.000 description 12
- 239000001963 growth medium Substances 0.000 description 12
- 239000012264 purified product Substances 0.000 description 11
- 229920001817 Agar Polymers 0.000 description 10
- 239000008272 agar Substances 0.000 description 10
- 238000005259 measurement Methods 0.000 description 10
- 239000000575 pesticide Substances 0.000 description 10
- 238000009331 sowing Methods 0.000 description 10
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 9
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 9
- 241000191967 Staphylococcus aureus Species 0.000 description 9
- 125000003275 alpha amino acid group Chemical group 0.000 description 9
- 241000223259 Trichoderma Species 0.000 description 8
- 238000004458 analytical method Methods 0.000 description 8
- 201000010099 disease Diseases 0.000 description 8
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 8
- 238000002156 mixing Methods 0.000 description 8
- 241000234282 Allium Species 0.000 description 7
- 235000002732 Allium cepa var. cepa Nutrition 0.000 description 7
- 239000013543 active substance Substances 0.000 description 7
- 238000012925 biological evaluation Methods 0.000 description 7
- 239000002361 compost Substances 0.000 description 7
- 230000035784 germination Effects 0.000 description 7
- 238000003306 harvesting Methods 0.000 description 7
- 238000005507 spraying Methods 0.000 description 7
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 6
- 235000002017 Zea mays subsp mays Nutrition 0.000 description 6
- 239000000919 ceramic Substances 0.000 description 6
- 235000013399 edible fruits Nutrition 0.000 description 6
- 238000005160 1H NMR spectroscopy Methods 0.000 description 5
- 241000219112 Cucumis Species 0.000 description 5
- 235000015510 Cucumis melo subsp melo Nutrition 0.000 description 5
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 5
- 240000008042 Zea mays Species 0.000 description 5
- 235000005824 Zea mays ssp. parviglumis Nutrition 0.000 description 5
- FJJCIZWZNKZHII-UHFFFAOYSA-N [4,6-bis(cyanoamino)-1,3,5-triazin-2-yl]cyanamide Chemical compound N#CNC1=NC(NC#N)=NC(NC#N)=N1 FJJCIZWZNKZHII-UHFFFAOYSA-N 0.000 description 5
- 150000001413 amino acids Chemical class 0.000 description 5
- 239000003242 anti bacterial agent Substances 0.000 description 5
- 238000003556 assay Methods 0.000 description 5
- 235000005822 corn Nutrition 0.000 description 5
- 230000004720 fertilization Effects 0.000 description 5
- 238000003973 irrigation Methods 0.000 description 5
- 230000002262 irrigation Effects 0.000 description 5
- 238000002955 isolation Methods 0.000 description 5
- 239000000203 mixture Substances 0.000 description 5
- 230000008635 plant growth Effects 0.000 description 5
- 238000011160 research Methods 0.000 description 5
- 241000219310 Beta vulgaris subsp. vulgaris Species 0.000 description 4
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 4
- 241000228143 Penicillium Species 0.000 description 4
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 4
- 235000021536 Sugar beet Nutrition 0.000 description 4
- 229940088710 antibiotic agent Drugs 0.000 description 4
- 238000012136 culture method Methods 0.000 description 4
- 238000010586 diagram Methods 0.000 description 4
- 238000011156 evaluation Methods 0.000 description 4
- 239000008103 glucose Substances 0.000 description 4
- 230000009036 growth inhibition Effects 0.000 description 4
- 230000002401 inhibitory effect Effects 0.000 description 4
- 239000013028 medium composition Substances 0.000 description 4
- 230000000813 microbial effect Effects 0.000 description 4
- 230000002093 peripheral effect Effects 0.000 description 4
- 230000001737 promoting effect Effects 0.000 description 4
- 239000000243 solution Substances 0.000 description 4
- 238000001228 spectrum Methods 0.000 description 4
- 230000001954 sterilising effect Effects 0.000 description 4
- 238000004659 sterilization and disinfection Methods 0.000 description 4
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 3
- 240000007124 Brassica oleracea Species 0.000 description 3
- 235000003899 Brassica oleracea var acephala Nutrition 0.000 description 3
- 235000011301 Brassica oleracea var capitata Nutrition 0.000 description 3
- 235000001169 Brassica oleracea var oleracea Nutrition 0.000 description 3
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 3
- 229920001661 Chitosan Polymers 0.000 description 3
- 240000008067 Cucumis sativus Species 0.000 description 3
- 235000010799 Cucumis sativus var sativus Nutrition 0.000 description 3
- 241000588724 Escherichia coli Species 0.000 description 3
- 238000005481 NMR spectroscopy Methods 0.000 description 3
- 241000295644 Staphylococcaceae Species 0.000 description 3
- 229930006000 Sucrose Natural products 0.000 description 3
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 3
- 230000002776 aggregation Effects 0.000 description 3
- 238000004220 aggregation Methods 0.000 description 3
- 239000012911 assay medium Substances 0.000 description 3
- 244000052616 bacterial pathogen Species 0.000 description 3
- LFYJSSARVMHQJB-QIXNEVBVSA-N bakuchiol Chemical compound CC(C)=CCC[C@@](C)(C=C)\C=C\C1=CC=C(O)C=C1 LFYJSSARVMHQJB-QIXNEVBVSA-N 0.000 description 3
- 230000015572 biosynthetic process Effects 0.000 description 3
- 229910000019 calcium carbonate Inorganic materials 0.000 description 3
- AXCZMVOFGPJBDE-UHFFFAOYSA-L calcium dihydroxide Chemical compound [OH-].[OH-].[Ca+2] AXCZMVOFGPJBDE-UHFFFAOYSA-L 0.000 description 3
- 239000000920 calcium hydroxide Substances 0.000 description 3
- 229910001861 calcium hydroxide Inorganic materials 0.000 description 3
- 235000011116 calcium hydroxide Nutrition 0.000 description 3
- 229940041514 candida albicans extract Drugs 0.000 description 3
- 150000001875 compounds Chemical class 0.000 description 3
- 239000012043 crude product Substances 0.000 description 3
- 230000001086 cytosolic effect Effects 0.000 description 3
- 230000007613 environmental effect Effects 0.000 description 3
- 230000006872 improvement Effects 0.000 description 3
- 238000011835 investigation Methods 0.000 description 3
- 244000052769 pathogen Species 0.000 description 3
- 239000000741 silica gel Substances 0.000 description 3
- 229910002027 silica gel Inorganic materials 0.000 description 3
- 239000002904 solvent Substances 0.000 description 3
- 239000005720 sucrose Substances 0.000 description 3
- 239000012138 yeast extract Substances 0.000 description 3
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- 241000123650 Botrytis cinerea Species 0.000 description 2
- 244000017020 Ipomoea batatas Species 0.000 description 2
- 235000002678 Ipomoea batatas Nutrition 0.000 description 2
- 244000061176 Nicotiana tabacum Species 0.000 description 2
- 235000002637 Nicotiana tabacum Nutrition 0.000 description 2
- WONDPHGHRLIPCB-OWOJBTEDSA-N OC(C1)C(/C=C/C(O)=O)=CC1=O Chemical compound OC(C1)C(/C=C/C(O)=O)=CC1=O WONDPHGHRLIPCB-OWOJBTEDSA-N 0.000 description 2
- 241001361634 Rhizoctonia Species 0.000 description 2
- 235000019764 Soybean Meal Nutrition 0.000 description 2
- 238000000862 absorption spectrum Methods 0.000 description 2
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 2
- 230000002378 acidificating effect Effects 0.000 description 2
- 230000009471 action Effects 0.000 description 2
- 239000004480 active ingredient Substances 0.000 description 2
- 230000003042 antagnostic effect Effects 0.000 description 2
- 230000008485 antagonism Effects 0.000 description 2
- 230000001580 bacterial effect Effects 0.000 description 2
- 230000004071 biological effect Effects 0.000 description 2
- 210000004899 c-terminal region Anatomy 0.000 description 2
- 210000004027 cell Anatomy 0.000 description 2
- 238000007796 conventional method Methods 0.000 description 2
- 239000007799 cork Substances 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 230000018109 developmental process Effects 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- 239000003337 fertilizer Substances 0.000 description 2
- 230000036039 immunity Effects 0.000 description 2
- 238000000338 in vitro Methods 0.000 description 2
- 230000006698 induction Effects 0.000 description 2
- 239000010954 inorganic particle Substances 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 2
- 238000009335 monocropping Methods 0.000 description 2
- 238000010899 nucleation Methods 0.000 description 2
- 229920001184 polypeptide Polymers 0.000 description 2
- 238000002360 preparation method Methods 0.000 description 2
- 108090000765 processed proteins & peptides Proteins 0.000 description 2
- 102000004196 processed proteins & peptides Human genes 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- 239000004455 soybean meal Substances 0.000 description 2
- 239000008223 sterile water Substances 0.000 description 2
- 239000010902 straw Substances 0.000 description 2
- 239000002344 surface layer Substances 0.000 description 2
- 230000009897 systematic effect Effects 0.000 description 2
- 238000010998 test method Methods 0.000 description 2
- MYPYJXKWCTUITO-UHFFFAOYSA-N vancomycin Natural products O1C(C(=C2)Cl)=CC=C2C(O)C(C(NC(C2=CC(O)=CC(O)=C2C=2C(O)=CC=C3C=2)C(O)=O)=O)NC(=O)C3NC(=O)C2NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(CC(C)C)NC)C(O)C(C=C3Cl)=CC=C3OC3=CC2=CC1=C3OC1OC(CO)C(O)C(O)C1OC1CC(C)(N)C(O)C(C)O1 MYPYJXKWCTUITO-UHFFFAOYSA-N 0.000 description 2
- 230000000007 visual effect Effects 0.000 description 2
- 238000005406 washing Methods 0.000 description 2
- UHPMCKVQTMMPCG-UHFFFAOYSA-N 5,8-dihydroxy-2-methoxy-6-methyl-7-(2-oxopropyl)naphthalene-1,4-dione Chemical compound CC1=C(CC(C)=O)C(O)=C2C(=O)C(OC)=CC(=O)C2=C1O UHPMCKVQTMMPCG-UHFFFAOYSA-N 0.000 description 1
- 235000001270 Allium sibiricum Nutrition 0.000 description 1
- 229920000856 Amylose Polymers 0.000 description 1
- 101100298224 Arabidopsis thaliana POT2 gene Proteins 0.000 description 1
- 244000063299 Bacillus subtilis Species 0.000 description 1
- 235000014469 Bacillus subtilis Nutrition 0.000 description 1
- 241000255789 Bombyx mori Species 0.000 description 1
- 241000222122 Candida albicans Species 0.000 description 1
- 241000207199 Citrus Species 0.000 description 1
- 241000037488 Coccoloba pubescens Species 0.000 description 1
- 201000007336 Cryptococcosis Diseases 0.000 description 1
- 241000221204 Cryptococcus neoformans Species 0.000 description 1
- 241000235036 Debaryomyces hansenii Species 0.000 description 1
- 241000588698 Erwinia Species 0.000 description 1
- 235000016623 Fragaria vesca Nutrition 0.000 description 1
- 240000009088 Fragaria x ananassa Species 0.000 description 1
- 235000011363 Fragaria x ananassa Nutrition 0.000 description 1
- 229930091371 Fructose Natural products 0.000 description 1
- 239000005715 Fructose Substances 0.000 description 1
- RFSUNEUAIZKAJO-ARQDHWQXSA-N Fructose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O RFSUNEUAIZKAJO-ARQDHWQXSA-N 0.000 description 1
- 241000223218 Fusarium Species 0.000 description 1
- 235000011201 Ginkgo Nutrition 0.000 description 1
- 244000194101 Ginkgo biloba Species 0.000 description 1
- 235000008100 Ginkgo biloba Nutrition 0.000 description 1
- 244000068988 Glycine max Species 0.000 description 1
- 235000010469 Glycine max Nutrition 0.000 description 1
- 240000005979 Hordeum vulgare Species 0.000 description 1
- 235000007340 Hordeum vulgare Nutrition 0.000 description 1
- 244000285963 Kluyveromyces fragilis Species 0.000 description 1
- 235000014663 Kluyveromyces fragilis Nutrition 0.000 description 1
- 101000878457 Macrocallista nimbosa FMRFamide Proteins 0.000 description 1
- 241000192041 Micrococcus Species 0.000 description 1
- 241001503951 Phoma Species 0.000 description 1
- 241000235648 Pichia Species 0.000 description 1
- 241000589516 Pseudomonas Species 0.000 description 1
- 241000589157 Rhizobiales Species 0.000 description 1
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 1
- 235000014680 Saccharomyces cerevisiae Nutrition 0.000 description 1
- 240000000111 Saccharum officinarum Species 0.000 description 1
- 235000007201 Saccharum officinarum Nutrition 0.000 description 1
- 241000235347 Schizosaccharomyces pombe Species 0.000 description 1
- 229920005654 Sephadex Polymers 0.000 description 1
- 239000012507 Sephadex™ Substances 0.000 description 1
- KSQXVLVXUFHGJQ-UHFFFAOYSA-M Sodium ortho-phenylphenate Chemical compound [Na+].[O-]C1=CC=CC=C1C1=CC=CC=C1 KSQXVLVXUFHGJQ-UHFFFAOYSA-M 0.000 description 1
- 244000061456 Solanum tuberosum Species 0.000 description 1
- 235000002595 Solanum tuberosum Nutrition 0.000 description 1
- 206010041925 Staphylococcal infections Diseases 0.000 description 1
- 241001480015 Trigonopsis variabilis Species 0.000 description 1
- 108010059993 Vancomycin Proteins 0.000 description 1
- 241000607479 Yersinia pestis Species 0.000 description 1
- 241000482268 Zea mays subsp. mays Species 0.000 description 1
- 244000273928 Zingiber officinale Species 0.000 description 1
- 235000006886 Zingiber officinale Nutrition 0.000 description 1
- 241000981595 Zoysia japonica Species 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 230000004520 agglutination Effects 0.000 description 1
- 239000003905 agrochemical Substances 0.000 description 1
- 238000007605 air drying Methods 0.000 description 1
- 230000000845 anti-microbial effect Effects 0.000 description 1
- 229940124350 antibacterial drug Drugs 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 244000000005 bacterial plant pathogen Species 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- 238000004166 bioassay Methods 0.000 description 1
- 230000003115 biocidal effect Effects 0.000 description 1
- 230000000853 biopesticidal effect Effects 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- 239000002374 bone meal Substances 0.000 description 1
- 229940036811 bone meal Drugs 0.000 description 1
- 238000009395 breeding Methods 0.000 description 1
- 230000001488 breeding effect Effects 0.000 description 1
- 239000006227 byproduct Substances 0.000 description 1
- YYRMJZQKEFZXMX-UHFFFAOYSA-N calcium;phosphoric acid Chemical compound [Ca+2].OP(O)(O)=O.OP(O)(O)=O YYRMJZQKEFZXMX-UHFFFAOYSA-N 0.000 description 1
- 229940095731 candida albicans Drugs 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 230000034303 cell budding Effects 0.000 description 1
- 230000032823 cell division Effects 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 238000002512 chemotherapy Methods 0.000 description 1
- 229960005091 chloramphenicol Drugs 0.000 description 1
- WIIZWVCIJKGZOK-RKDXNWHRSA-N chloramphenicol Chemical compound ClC(Cl)C(=O)N[C@H](CO)[C@H](O)C1=CC=C([N+]([O-])=O)C=C1 WIIZWVCIJKGZOK-RKDXNWHRSA-N 0.000 description 1
- 235000020971 citrus fruits Nutrition 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 238000005336 cracking Methods 0.000 description 1
- VOXWVWGNZYVMQA-UHFFFAOYSA-N cycloprop-2-en-1-ol Chemical compound OC1C=C1 VOXWVWGNZYVMQA-UHFFFAOYSA-N 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 238000003113 dilution method Methods 0.000 description 1
- 230000006806 disease prevention Effects 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 239000003814 drug Substances 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 238000010828 elution Methods 0.000 description 1
- 230000001747 exhibiting effect Effects 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 230000004992 fission Effects 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 244000053095 fungal pathogen Species 0.000 description 1
- 235000008397 ginger Nutrition 0.000 description 1
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 1
- 239000010931 gold Substances 0.000 description 1
- 229910052737 gold Inorganic materials 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 210000004209 hair Anatomy 0.000 description 1
- 238000011534 incubation Methods 0.000 description 1
- 230000008595 infiltration Effects 0.000 description 1
- 238000001764 infiltration Methods 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 230000005764 inhibitory process Effects 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 239000010410 layer Substances 0.000 description 1
- 238000009630 liquid culture Methods 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 230000013011 mating Effects 0.000 description 1
- 238000003808 methanol extraction Methods 0.000 description 1
- 208000015688 methicillin-resistant staphylococcus aureus infectious disease Diseases 0.000 description 1
- 244000005700 microbiome Species 0.000 description 1
- ICYJJTNLBFMCOZ-UHFFFAOYSA-J molybdenum(4+);disulfate Chemical compound [Mo+4].[O-]S([O-])(=O)=O.[O-]S([O-])(=O)=O ICYJJTNLBFMCOZ-UHFFFAOYSA-J 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 231100000989 no adverse effect Toxicity 0.000 description 1
- 235000015816 nutrient absorption Nutrition 0.000 description 1
- 238000009329 organic farming Methods 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 230000000243 photosynthetic effect Effects 0.000 description 1
- 244000000003 plant pathogen Species 0.000 description 1
- OVARTBFNCCXQKS-UHFFFAOYSA-N propan-2-one;hydrate Chemical compound O.CC(C)=O OVARTBFNCCXQKS-UHFFFAOYSA-N 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 239000004576 sand Substances 0.000 description 1
- 238000007790 scraping Methods 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 238000010561 standard procedure Methods 0.000 description 1
- 230000000638 stimulation Effects 0.000 description 1
- 239000004575 stone Substances 0.000 description 1
- 239000002426 superphosphate Substances 0.000 description 1
- 239000008400 supply water Substances 0.000 description 1
- 230000004083 survival effect Effects 0.000 description 1
- 230000009885 systemic effect Effects 0.000 description 1
- 238000003971 tillage Methods 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 230000005945 translocation Effects 0.000 description 1
- 238000002211 ultraviolet spectrum Methods 0.000 description 1
- MYPYJXKWCTUITO-LYRMYLQWSA-N vancomycin Chemical compound O([C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1OC1=C2C=C3C=C1OC1=CC=C(C=C1Cl)[C@@H](O)[C@H](C(N[C@@H](CC(N)=O)C(=O)N[C@H]3C(=O)N[C@H]1C(=O)N[C@H](C(N[C@@H](C3=CC(O)=CC(O)=C3C=3C(O)=CC=C1C=3)C(O)=O)=O)[C@H](O)C1=CC=C(C(=C1)Cl)O2)=O)NC(=O)[C@@H](CC(C)C)NC)[C@H]1C[C@](C)(N)[C@H](O)[C@H](C)O1 MYPYJXKWCTUITO-LYRMYLQWSA-N 0.000 description 1
- 229960003165 vancomycin Drugs 0.000 description 1
- 235000013311 vegetables Nutrition 0.000 description 1
Images
Landscapes
- Preparation Of Compounds By Using Micro-Organisms (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Cultivation Of Plants (AREA)
Abstract
Description
この発明は、トリコデルマ ハルジアナム SK−5−5(Trichoderuma hurzianum SK-5-5)菌の培地から抽出した糸状菌及び細菌に抗菌性を有する物質を得ることを目的とした植物の活性付与剤の製造方法に関する。また他の発明は、トリコデルマ ハルジアナム SK−5−5菌の分生胞子の生成する物質によって植物の根、茎、葉等に病原菌に対する抵抗性を付与することを目的とした植物の活性付与方法及び植物の活性促進剤並びにその施用方法に関する。 The present invention relates to the production of a plant activity-imparting agent for the purpose of obtaining a filamentous fungus extracted from a medium of Trichoderuma hurzianum SK-5-5 (Trichoderuma hurzianum SK-5-5) and a substance having antibacterial properties against the bacterium. Regarding the method. Another invention relates to a method for imparting plant activity for the purpose of imparting resistance to pathogenic bacteria to the roots, stems, leaves, etc. of plants by substances produced by conidia of Trichoderma harzianum SK-5-5. The present invention relates to a plant activity promoter and a method for applying the same.
トリコデルマ菌は、微生物農薬として病害防除に実用化されている糸状菌であり、Trichoderuma lignourum がタバコの白絹病の防除用農薬として登録されている(農薬登録 No.7023)。また英国、フランスでもTrichoderuma hurzianumを有効成分とする野菜の立枯病、苗腐病に有効な微生物農薬(F-stop)が登録されている。 Trichoderma is a fungus that has been put to practical use as a microbial pesticide for disease control, and Trichoderuma lignourum has been registered as a pesticide for control of tobacco silkworm (Agricultural Chemical Registration No. 7023). In the UK and France, microbial pesticides (F-stop) are also registered, which are effective against vegetable wilt and seedling rot, which contain Trichoderuma hurzianum as an active ingredient.
近来減農薬・生態系保全型の病害防除の必要性が強く唱えられ、特に生物防除方法は環境負荷の影響が少ないと考えられるので、精力的に研究が進められている。植物の根や根圏から分離される細菌や、菌類の中には植物の生育を促進するものがあり、それぞれ植物生育促進根圏(plant growth-promoting rhizobacteria:PGPR)、植物生育促進菌類(plant growth-promoting fungi:PGPF)と呼ばれている。 Recently, the need for pesticide reduction and ecosystem conservation type disease control is strongly advocated, and the biological control method is considered to have little impact on the environment, so research is being conducted energetically. Some bacteria and fungi isolated from plant roots and rhizospheres promote plant growth. Plant growth-promoting rhizobacteria (PGPR) and plant growth-promoting fungi (plant) It is called growth-promoting fungi (PGPF).
前記有用根圏微生物であるPGPR、PGPFは植物の生育を促進するのみならず、各種の土壌病害を抑制することも知られている。また最近では、土壌病害のみならず、地上病害も抑制する事実も明らかにされ、前記PGPR、PGPFは、植物の全身抵抗性の誘導が関わっていることが見出されたと紹介されている(1999年6月号、今月の農業誌)。 The useful rhizosphere microorganisms PGPR and PGPF are known not only to promote plant growth but also to suppress various soil diseases. Recently, the fact that not only soil diseases but also ground diseases are also clarified, and it has been introduced that the PGPR and PGPF were found to be involved in the induction of systemic resistance of plants (1999). June issue, this month's agricultural magazine).
またコウライシバから分離選抜したPhoma,Trichoderma, Fusarium,Penicillium,Sterileなどの菌が、キュウリにおける炭そ病に対し、誘導抵抗性を示すことが報告されている(1999年6月号、今月の農業誌)。 In addition, it has been reported that fungi such as Phoma, Trichoderma, Fusarium, Penicillium, and Sterile isolated and selected from cucumber shiba show induction resistance against anthracnose in cucumber (June 1999 issue, this month's agricultural journal). ).
この発明は、植物病原菌に抗菌性を有する物質を得ることを課題として研究しており、北海道十勝地方の土壌からTrichoderuma hurzianum SK−5−5(トリコデルマ ハルジアナム SK−5−5)菌を分離した。この菌は、植物病原菌の80%を占めるとされる土壌伝染性病原糸状菌に対して広く拮抗性を示す菌であり、現在芝のブラウンバッチ、ラージバッチなどの Rhizoctonia類に微生物農薬(生菌製剤)として開発中のものである。前記トリコデルマ ハルジアナムSK−5−5菌の拮抗作用は、相手菌糸に接触し、コイリングをする事により、細胞質凝集を促し死滅させるものである。その外観的観察経過より見てペニシリアム(Penicillium sp)等が生産する抗生物質のような培地上で阻止円を示すほど活性が高いものではなく、菌糸同志の接触により発現するものと推定される。 The present invention has been studied as an object to obtain a substance having antibacterial properties against plant pathogenic bacteria, and Trichoderuma hurzianum SK-5-5 (Trichoderma harzianum SK-5-5) is isolated from soil in Tokachi region, Hokkaido. This bacterium is widely antagonizing against soil-borne pathogenic fungi, which accounts for 80% of plant pathogens. Currently, microbial pesticides (living bacteria) are present in Rhizoctonias such as turf brown and large batches. It is under development as a formulation. The antagonism of the Trichoderma haldianum SK-5-5 bacterium promotes cytoplasmic aggregation and kills by contacting the other hyphae and coiling. It is presumed from the appearance observation process that the activity is not so high as to show a circle of inhibition on a medium such as an antibiotic produced by penicillium (Penicillium sp) and the like, and is expressed by contact between mycelia.
前記細胞質凝集作用が、トリコデルマ ハルジアナム SK−5−5菌の生産する何らかの物質によってもたらされると考えられ、これを究明することを第一の課題としたものである。
なお、この発明で用いるトリコデルマ ハルジアナム SK−5−5菌は、日本国内工業技術院微生物工業研究所に受託番号「微工研菌寄第13327号」として寄託されている。微工研は、当該原寄託よりブダペスト条約に基づく寄託への移管請求を行ない、受託番号BP−4346が付与されている。
It is considered that the cytoplasmic aggregation action is brought about by some substance produced by Trichoderma harzianum SK-5-5, and it is a first object to investigate this.
The Trichoderma harzianum SK-5-5 bacterium used in the present invention has been deposited with the Japanese Industrial Technology Institute, Microbial Industrial Research Institute under the accession number “Seikenken No. 13327”. The Institute of Fine Arts and Technology has requested transfer from the original deposit to a deposit based on the Budapest Treaty and has been assigned a deposit number of BP-4346.
前記従来のPGPR、PGPFは、特定植物(例えばキュウリ)に対し、炭そ病についての有効性を示すもので、今後研究の結果、その使用方法の改善などにより、他の植物、病原菌に対して有効なことが判明する可能性はあるとしても、未だ具体的植物、病原菌に対しては今後の課題とされている。
然し乍ら単に病原菌を殺菌するという従来の生物農薬の思考形態が、植物の根茎に亘り抵抗性を付与する方向に変りつつあることは、今後の植物栽培上重要な示唆を含むものである。
The conventional PGPR and PGPF are effective against anthracnose for specific plants (for example, cucumber). As a result of future research, improvement of the method of use, etc., against other plants and pathogens Even though it may prove effective, it is still considered as a future problem for specific plants and pathogens.
However, the fact that the conventional form of biological pesticides, which simply sterilizes pathogenic bacteria, is changing in the direction of imparting resistance to the rhizomes of plants, includes important suggestions for future plant cultivation.
この発明は、トリコデルマ ハルジアナム SK-5−5菌について、各種実験を重ねている間に、前記生物農薬の将来性と合致することに想到し、更に使用方法、対象植物等を選定、研究の結果、この発明を完成したのである。将来の植物栽培に多大の影響を与えるものとして、この発明はきわめて有望であり、将来の農業等を支える重要な手段の1つとなることに疑はない。 The present invention was conceived to be consistent with the potential of the biopesticides during various experiments on Trichoderma harzianum SK-5-5, and further, the method of use, the target plant, etc. were selected, and the results of the research The present invention has been completed. The present invention is very promising as having a great influence on future plant cultivation, and there is no doubt that it will be one of the important means for supporting future agriculture and the like.
この発明は、トリコデルマ ハルジアナム SK−5−5菌の生産する物質により、細胞凝集作用(抗菌性)がもたらされるものと推定し、前記菌を培養、精製、同定した所、糸状菌又は細菌に抗菌性を有する物質を得たのである。 The present invention presumes that the substance produced by Trichoderma harzianum SK-5-5 bacterium causes cell agglutination (antibacterial activity). When the bacterium is cultured, purified and identified, it is antibacterial against filamentous fungi or bacteria. The substance with the property was obtained.
また他の発明は、植物栽培に際し、トリコデルマ ハルジアナム SK−5−5菌の分生胞子を覆土中に共存させて増殖させることにより、その生成物質により、各種植物の根茎活性を付与し、耐菌性を向上させることが判明し、前記従来の問題点を発展的に改善させて、実用性を確立したものである。 In another aspect of the invention, in the cultivation of plants, conidia of Trichoderma harzianum SK-5-5 are coexisted in the overburden and proliferated, thereby imparting rhizome activity of various plants with the product, As a result, it has been found that the conventional problems have been improved and the practical problems have been established.
即ち活性付与剤の発明は、トリコデルマ ハルジアナム SK−5−5菌を固体培地で培養し、抽出した物質であって、糸状菌に抗菌力を有することを特徴とした植物の活性付与剤である。また他の発明は、トリコデルマ ハルジアナムSK−5−5菌を液体培地で培養し、抽出した物質であって、細菌の一種(Staphylococcus aureus 209p)に抗菌力を有することを特徴とした植物の活性付与剤である。 That is, the invention of the activity-imparting agent is a plant activity-imparting agent characterized in that it is a substance obtained by culturing Trichoderma harzianum SK-5-5 bacteria in a solid medium and extracting them, and having antibacterial activity against filamentous fungi. Another invention is a substance obtained by culturing Trichoderma harzianum SK-5-5 bacteria in a liquid medium and extracting the plant, characterized by having antibacterial activity against a kind of bacteria (Staphylococcus aureus 209p) It is an agent.
次に製造方法の発明は、トリコデルマ ハルジアナム SK−5−5菌を固体培地に植菌し、25℃〜30℃で7日〜15日間静置培養した後、抽出して、前記の活性付与剤を得ることを特徴とした植物の活性付与剤の製造方法であり、またトリコデルマ ハルジアナム SK−5−5菌を液体培地に植菌し、25℃〜30℃で4日〜10日間振盪培養した後、抽出して、前記の活性付与剤を得ることを特徴とした植物の活性付与剤の製造方法である。 Next, the invention of the production method is such that Trichoderma harzianum SK-5-5 is inoculated into a solid medium, left to stand at 25 ° C. to 30 ° C. for 7 to 15 days, extracted, and extracted. And after inoculating Trichoderma harzianum SK-5-5 in a liquid medium and shaking culture at 25 ° C. to 30 ° C. for 4 to 10 days. Extracting to obtain the above-mentioned activity imparting agent, a method for producing a plant activity imparting agent.
この発明において、使用する固体培地として、米培地を用いた。米培地は、米100%、大豆かす3%、滅菌水10%よりなる固定培地であって、培養中に培地の表面が乾燥しないよう、滅菌水を添加した。 In this invention, a rice medium was used as the solid medium to be used. The rice medium was a fixed medium composed of 100% rice, 3% soybean meal, and 10% sterilized water, and sterilized water was added so that the surface of the medium was not dried during the cultivation.
この発明において、使用する液体培地は、グルコース3.0%〜5.0%、ポリペプトン0.5%、Nacl0.8%、酵母エキス0.2%、炭酸カルシウム1.0%である。 In this invention, the liquid medium to be used is glucose 3.0% to 5.0%, polypeptone 0.5%, Nacl 0.8%, yeast extract 0.2%, calcium carbonate 1.0%.
前記のように固定培地と、液体培地によれば、夫々の特性により、異なる物質を生成することが明らかとなった。このような結果についてのメカニズムは明らかでないが、トリコデルマ ハルジアナム SK−5−5菌に対する刺激その他の作用の相違、かつ培地構成物質に関する特性により、異なる物質を生成するものであって、前記米培地及び液体培地以外の成分の培地であっても同様の物質を生成することは十分考えられ、かつ生成効率向上等今後の研究課題は多大である。 As described above, it has been clarified that the fixed medium and the liquid medium generate different substances depending on the respective characteristics. Although the mechanism for such a result is not clear, it produces different substances depending on differences in stimulation and other actions on Trichoderma harzianum SK-5-5, and on the characteristics of the medium constituents. Even if it is a culture medium of components other than a liquid culture medium, producing | generating the same substance is considered enough, and future research subjects, such as improvement of production efficiency, are great.
次に活性付与方法の発明は、植物栽培に際し、トリコデルマ ハルジアナムSK−5−5菌の分生胞子に施用手段を付加し、前記植物に活性を付与する覆土中に共存させることを特徴とした植物の活性付与方法であり、植物栽培に際し、トリコデルマ ハルジアナム SK−5−5菌の分生胞子に施用手段を付加し、前記植物に活性を付与する覆土中に共存させて増殖させ、糸条菌に抗菌力を有する物質の生産を持続させることを特徴とした植物の活性付与方法である。また植物栽培に際し、トリコデルマ ハルジアナム SK−5−5菌の分生胞子に施用手段を付加し、前記植物に活性を付与する覆土中に共存させると共に、前記分生胞子の増殖促進条件を付与することを特徴とした植物の活性付与方法であり、分生胞子の施用手段は、植物の種子処理、覆土と分生胞子との混和、分生胞子の散布、潅水又は埋設したものである。更に増殖促進条件は、土壌温度を15℃〜30℃とし、水分を30%以上としたものであり、他の増殖促進条件は、土壌に増殖可能な通気性又は含気性を付与するものである。 Next, the invention of the activity imparting method is a plant characterized in that, in plant cultivation, an application means is added to the conidia of Trichoderma harzianum SK-5-5 and coexisted in the covering soil imparting activity to the plant. In the plant cultivation, an application means is added to the conidia of Trichoderma harzianum SK-5-5, and it is allowed to coexist in the covering soil that imparts the activity to the plant. A plant activity imparting method characterized by sustaining production of a substance having antibacterial activity. In addition, during plant cultivation, an application means is added to the conidia of Trichoderma harzianum SK-5-5, and it is allowed to coexist in the cover soil that imparts activity to the plant, and conditions for promoting the growth of the conidia are provided. The method for imparting plant activity characterized by the above, wherein the conidia application means are plant seed treatment, mixing of the cover soil with the conidia, spraying of the conidia, irrigation or embedding. Further, the growth promotion condition is that the soil temperature is 15 ° C. to 30 ° C. and the water content is 30% or more, and the other growth promotion condition is to impart air permeability or aeration that can grow on the soil. .
次に他の発明は、多孔性セラミックス粒子その他の担体に、トリコデルマ ハルジアナム SK−5−5菌の分生胞子を付着させたことを特徴とする植物の活性促進剤であり、無菌処理した無機質粒子に、トリコデルマ ハルジアナム SK−5−5菌の分生胞子を混合し、又はトリコデルマ ハルジアナム SK−5−5菌を付着させ、前記無機質粒子と他の粒子とを混合したことを特徴とする植物の活性促進剤である。またトリコデルマ ハルジアナム SK−5−5菌の分生胞子を培養し、これを培地と共に適量宛分離し、無菌の多孔質粒子に栄養分(例えばキトサン)と共に付着させた後、所定量宛包装したことを特徴とする植物の活性促進剤であり、トリコデルマ ハルジアナム SK−5−5菌の分生胞子を液体培地で増殖させ、これを無菌の多孔質粒子に栄養分と共に付着させた後、多孔質粒子を所定量宛包装したことを特徴とする植物の活性促進剤である。 Next, another invention is a plant activity promoter characterized in that conidia of Trichoderma harzianum SK-5-5 are attached to porous ceramic particles and other carriers, and is an aseptically treated inorganic particle Plant activity characterized by mixing conidia of Trichoderma harzianum SK-5-5, or adhering Trichoderma harzianum SK-5-5, and mixing the inorganic particles with other particles It is an accelerator. In addition, the conidia of Trichoderma harzianum SK-5-5 were cultured, separated to an appropriate amount together with the medium, attached to a sterile porous particle together with nutrients (for example, chitosan), and then packaged for the predetermined amount. A plant activity promoter characterized by the growth of Trichoderma harzianum SK-5-5 conidia in a liquid medium, which is attached to a sterile porous particle with nutrients, and then the porous particle is placed in place. It is a plant activity promoter characterized by being packaged for fixed quantity.
また他の発明は、前記記載の植物の活性促進剤を、育苗土壌に混入し、又は育苗時に土壌に散布或いは舗場に散布することを特徴とした植物の活性促進剤の施用方法である。この場合の活性促進剤の使用量は、トリコデルマ ハルジアナムSK−5−5菌5×104/g〜5×109/gを1m2当り5g〜100g散布又は栽培土1m3当り5g〜100g混入するものである。前記発明における植物は、てん菜、メロン、トウモロコシ、水稲、キャベツ及びタマネギなどであり、根、茎、葉及び実を採取する植物について、何れも有効であることが確認され、その効果も収量増加、糖度増加その他の有為性が明確になった。 Another aspect of the invention is a method for applying a plant activity promoter, characterized in that the plant activity promoter described above is mixed into seedling soil, or is sprayed on soil or on a pavement at the time of seedling. In this case, the amount of the activity promoter used is 5 to 10 g of 5 × 10 4 / g to 5 × 10 9 / g of Trichoderma haldianum SK-5-5 per 1 m 2 or 5 to 100 g mixed per 1 m 3 of cultivated soil. To do. Plants in the invention are sugar beet, melon, corn, paddy rice, cabbage and onion, etc., and it is confirmed that all of the plants from which roots, stems, leaves and fruits are collected are effective, and the effect is also increased in yield. Increased sugar content and other potentials became clear.
前記の発明におけるトリコデルマ ハルジアナム SK−5−5菌の分生胞子を培養し、その単離精製物を検討した所、UV吸収スペクトラム、質量分析及びNMR測定結果より、Aib(α-Aminoisobutyric acid)を含むPolypeptideの、Peptibols系と判断した。Peptibols系抗 生物質はアミノ酸配列の中に α-Aminoisobutyric acid(Aib)を含んでおり、 N末端がAcetyl基で、C末端が Amino alcohol結合であることが特徴である(多くは Phenylalaniol基でおわる)。なお今回の精製物、A成分、B成分、D成分は質量分析より一部のアミノ酸配列を下記のように推定し、その部分構造を既存のPeptibols のアミノ酸配列と検索した結果、一致する物が無く、新規 Peptibols系であると推定した。Eはアミノ酸配列の推定が困難であるため判断できなかった。 The conidia of Trichoderma harzianum SK-5-5 in the above invention were cultured, and the isolated and purified product was examined. From the results of UV absorption spectrum, mass spectrometry and NMR measurement, Aib (α-Aminoisobutyric acid) was obtained. The polypeptide contained was determined to be Peptibols. Peptibols antibiotics contain α-Aminoisobutyric acid (Aib) in the amino acid sequence, and are characterized by an N-terminal Acetyl group and a C-terminal Amino alcohol bond (mostly Phenylalaniol groups). ). As for the purified product, A component, B component, and D component, the partial amino acid sequence was estimated by mass spectrometry as follows, and the partial structure was searched with the existing amino acid sequence of Peptibols. There was no new Peptibols system. E could not be determined because it was difficult to estimate the amino acid sequence.
UV吸収波長:4成分とも末端吸収
A成分=1933
B成分=1949または1964
D成分=1810
E成分=1829
推定構造:
A成分:Ac-Aib-Ala-Aib-Aib-Aib-Aib-Gln-Aib-Aib-......
B成分:Ac-Aib-Ala-Aib-Aib-Val-Aib-Gln-Aib-Aib-......
D成分:Ac-Aib-Ala-Aib-Aib-Aib-.....
前記のように生成物質は新規配列のアミノ酸と認められるが、植物の根より吸収されて、茎及び葉に至り、ついで消失する。従って収穫時の植物には、根、茎、葉共に残留の有無は不明であるけれども、当初(発芽以後の幼茎等)植物に吸収された段階で、植物のDNAに何等かの変化を与えるものか、残留するものと推定される。何故ならば、一旦抗菌性を取得した植物は、根茎等の分裂生成に拘らず抗菌性の持続が認められるからである。従って免疫性付与(活性付与)と類似であり、育苗時又は比較的若い植物(実質的に植物の増殖時)に施用すれば、再使用の必要性が認められないのは、一旦活性ができると、その植物の一生に亘り効力が持続されるからである。
UV absorption wavelength: terminal absorption of all four components A component = 1933
B component = 1949 or 1964
D component = 1810
E component = 1829
Estimated structure:
A component: Ac-Aib-Ala-Aib-Aib-Aib-Aib-Gln-Aib-Aib -......
B component: Ac-Aib-Ala-Aib-Aib-Val-Aib-Gln-Aib-Aib -......
D component: Ac-Aib-Ala-Aib-Aib-Aib -.....
As described above, the product is recognized as an amino acid having a novel sequence, but is absorbed from the roots of the plant, reaches the stem and leaves, and then disappears. Therefore, it is unknown whether the roots, stems, and leaves remain in the plants at the time of harvest, but some changes are made to the DNA of the plants when they are absorbed by the plants (eg, young shoots after germination). It is presumed that there is something or remains. This is because plants that have once obtained antibacterial properties are recognized as having antibacterial properties regardless of fission formation such as rhizomes. Therefore, it is similar to immunity imparting (activity imparting), and if applied to seedlings or relatively young plants (substantially during plant growth), there is no need for re-use, once it can be activated This is because the efficacy is maintained throughout the life of the plant.
この発明のトリコデルマ ハルジアナム SK−5−5菌の分生胞子は、植物の苗の時、又は播種時に覆土中に混入すると、前記分生胞子の増殖に伴って生成物質も増加し、これが根から吸収されて茎葉に至り、全体に活性が付与されるので(恰も免疫性付与の如く)、植物が生長して根又は実などを採取する時期(例えば播種後1ヶ月〜6ヶ月後)になっても、前記抗菌性は保たれることが確認された。従って多くの植物は、一回の処理(散布その他の手段)によって活性が付与されてその目的を十分達成することができた。 When the conidia of Trichoderma harzianum SK-5-5 of this invention are mixed in the cover soil at the time of plant seedling or sowing, the amount of product increases with the growth of the conidia, Since it is absorbed and reaches the foliage, and the activity is imparted to the whole (as is the case with immunity), it is time for the plant to grow and collect roots or fruits (for example, 1 to 6 months after sowing). However, it was confirmed that the antibacterial property was maintained. Therefore, many plants were imparted with activity by a single treatment (dispersion or other means), and their objectives could be fully achieved.
この発明における分生胞子の担体は、多孔質セラミックス粒子(例えば麦飯石の粒子)であって、これをキトサン液に浸漬し、水分を蒸発したものである。前記粒子の大きさについて特定はないが、取扱いの容易性から直径0.5mm〜5mm位が好ましい。前記担体は、多孔質セラミックス粒子(天然又は人工)に限定されることなく、分生胞子に悪影響がない物は使用することができる。 The conidia carrier in the present invention is porous ceramic particles (for example, barley stone particles), which are immersed in chitosan liquid and water is evaporated. Although the size of the particles is not specified, a diameter of about 0.5 mm to 5 mm is preferable because of easy handling. The carrier is not limited to porous ceramic particles (natural or artificial), and those that do not adversely affect the conidia can be used.
前記セラミックス粒子に予め培養したトリコデルマ ハルジアナム SK−5−5菌を5×104/g〜5×109/g宛付着させ、これを1m2当り5g〜100g宛散布する。この場合に、水と共に散布する場合もあるが、潅水するのは、水分付与を目的とする場合と、分生胞子を均等に浸透させて、土壌中に均等に分布させる為に用いる場合とがある。 Trichoderma harzianum SK-5-5 bacteria previously cultured on the ceramic particles are attached to 5 × 10 4 / g to 5 × 10 9 / g and sprayed to 5 to 100 g per 1 m 2 . In this case, it may be sprayed together with water, but irrigation may be used for the purpose of watering or when it is used to uniformly infiltrate the conidia and distribute it in the soil. is there.
前記トリコデルマ ハルジアナム SK−5−5菌の量を5×104/g未満にした場合に、前記菌の繁殖が阻害されなければ、増殖されるが、何等かの理由により増殖しない場合があるので、一応の目途とした。然し乍ら菌の増殖は環境によって著しく相違するので、施用後の環境整備によっては更に少量(例えば5g未満)の菌でも植物の生育に合せて増殖し、必要量の生産物質を得ることは可能と考えられる。 When the amount of Trichoderma harzianum SK-5-5 bacterium is less than 5 × 10 4 / g, it is proliferated if the bacterium is not inhibited from breeding, but may not grow for some reason. , For the time being. However, since the growth of bacteria varies significantly depending on the environment, it is considered that even if a small amount (for example, less than 5 g) of bacteria grows along with the growth of the plant, it is possible to obtain the required amount of product by environmental preparation after application. It is done.
一方菌量の上限を5×109/g以下としたのは、前記生産物質の関係から不必要と考えたからである。菌の増殖条件が悪い場合には、比較的高濃度の施用を要するが、5×109/gを越える必要はないと考えられる。この発明におけるトリコデルマ ハルジアナム SK−5−5菌は通常分生胞子として与えるが、施用時の環境が厳しい場合(例えば高温時、寒冷時)には厚膜胞子として与える方が好ましい場合もある。 On the other hand, the reason why the upper limit of the amount of bacteria was set to 5 × 10 9 / g or less was that it was considered unnecessary because of the relationship between the production substances. When the growth conditions of the fungus are poor, it is necessary to apply a relatively high concentration, but it is not considered necessary to exceed 5 × 10 9 / g. The Trichoderma harzianum SK-5-5 bacterium in the present invention is usually given as conidia, but it may be preferable to give it as a thick film spore when the environment during application is severe (for example, at high temperature or cold).
前記発明において、トリコデルマ ハルジアナム SK−5−5菌を固体培地又は液体培地又はその他の培地で培養し、その生成物を抽出して、これを植物の活性付与剤として施用する場合には、例えば0.1〜1g/m2必要となる。 In the above invention, when Trichoderma harzianum SK-5-5 bacterium is cultured in a solid medium, liquid medium or other medium, and the product is extracted and applied as a plant activity-imparting agent, for example, 0 .1 to 1 g / m 2 is required.
また分生胞子を散布する場合には、当該分生胞子の増殖下限菌数(例えば5×109/g)が必要である。 In addition, when spraying conidia, the lower limit number of bacteria (for example, 5 × 10 9 / g) of the conidia is necessary.
この発明の植物の活性付与剤は、菌の時に1回施用するだけで、爾後収穫まで施用する必要はない。この点は一般農薬と異なり、そのメカニズムは不明であるが、植物のDNAに何等かの影響を与え、又は生成物質が植物の根、茎、葉に微量残留して、前記特性を発揮するものと推定される。 The plant activity-imparting agent of the present invention is applied only once at the time of fungi, and does not need to be applied until after harvesting. This is different from general pesticides, and its mechanism is unknown, but it has some effect on the DNA of the plant, or the produced substance remains in trace amounts in the roots, stems and leaves of the plant and exhibits the above characteristics It is estimated to be.
前記において、分生胞子を施用する場合には、舗場などで、分生胞子が十分増殖し、飽和に達したならば、分生胞子は急速に活力を失い、遂には消滅することが確認された。尤も一部分分生胞子が残留していることも考えられ、増殖条件が良好になれば再び増殖する場合も有り得る。 In the above, when conidia are applied, it is confirmed that the conidia rapidly loses vitality and eventually disappears if the conidia grows sufficiently on the pavement and reaches saturation. It was done. However, conidial spores may partially remain, and if the growth conditions are improved, they may proliferate again.
前記発明においては、トリコデルマ ハルジアナム SK−5−5を固体培地又は液体培地で培養し、その生成物質中A、B、C、D、E成分について単離精製したが、活性物質は、新規アミノ酸配列をもつ新規物質で、その分子量は、A成分=192,B成分=206,C成分=168,D1=154,D2=220であって、当該アミノ酸又はその周辺物が植物の活性化に有効であろうと推定される。 In the above invention, Trichoderma haldianum SK-5-5 was cultured in a solid medium or a liquid medium, and the components A, B, C, D, and E in the product were isolated and purified. The molecular weight is A component = 192, B component = 206, C component = 168, D 1 = 154, D 2 = 220, and the amino acid or its peripherals are used for plant activation. Presumed to be effective.
そこでトリコデルマ ハルジアナム SK−5−5菌の分生胞子を所定濃度(例えば1g中5×105以上)以上で施用(土壌に混用、又は根物の根圏に散布)することにより、所期の目的を達成することが確認されている。恐らく、植物により最適の成分又は混用比があるに違いない。 Therefore, by applying conidia of Trichoderma harzianum SK-5-5 at a predetermined concentration (for example, 5 × 10 5 or more in 1 g) or more (mixed in soil or sprayed in root rhizosphere of root), It has been confirmed that the objective is achieved. Perhaps there must be an optimal component or mixing ratio depending on the plant.
そこで前記トリコデルマ ハルジアナム SK−5−5菌を従来法によって多量培養し、その生成物質を抽出精製して植物の活性付与剤を得ることができる。前記生成物質は、前記A成分、B成分、C成分、D成分及びE成分が判明している。 Accordingly, the Trichoderma harzianum SK-5-5 bacterium can be cultured in a large amount by a conventional method, and the product can be extracted and purified to obtain a plant activity-imparting agent. The product A, B component, C component, D component, and E component are known.
前記発明は、各種植物について有効であるが、実験の結果(実験中も含む)によれば、水稲、トウモロコシ、てん菜、メロン、馬鈴薯、さつまいも、いちご、玉ねぎ、キャベツなどに有効なことが確認された。 The invention is effective for various plants, but according to experimental results (including during the experiment), it has been confirmed that it is effective for rice, corn, sugar beet, melon, potato, sweet potato, strawberry, onion, cabbage and the like. It was.
この発明によれば、トリコデルマ ハルジアナム SK−5−5菌を固形培地に培養した場合には、表2のように、糸状菌について抗菌性を示すことが認められた。
またトリコデルマ ハルジアナム SK−5−5菌を液体培地で培養した場合には表2のようにバクテリアに有効であるものと認められた。
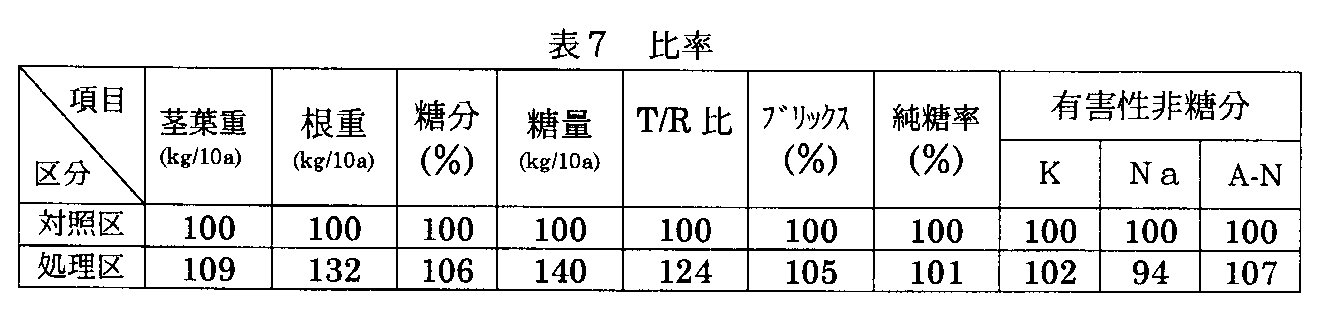
この発明によれば、植物の生育初期に、床土等にトリコデルマ ハルジアナムSK−5−5菌を適量宛混入することにより、又は生育時の散布など根圏に施用することにより、植物を活性化してその根・茎・葉を改善し、罹病防止、病原菌に対する耐性を付与することができる。これにより生育時の発病を激減させるのみならず、植物の生育を促進し、増収を図り、糖度を向上させるなどの諸効果がある。
然して前記効果は、前記トリコデルマ ハルジアナム SK−5−5菌の分生胞子の生成物質(例えば新規アミノ酸)によるものと推定されるが、前記分生胞子は増殖後、また前記生成物質は、植物の成長終期には何れも消失するものと認められるので、如何なる意味の影響もなく、かつ連続使用についても何等の悪影響も見られない効果がある。更に前記生成物質は新規アミノ酸及びその周辺物質と認められるから、植物の葉、茎などに微量残留しても無害である。
According to this invention, when Trichoderma harzianum SK-5-5 bacterium was cultured in a solid medium, as shown in Table 2, it was confirmed that the fungus exhibited antibacterial properties.
When Trichoderma harzianum SK-5-5 was cultured in a liquid medium, it was recognized that it was effective against bacteria as shown in Table 2.
According to the present invention, at the initial stage of plant growth, the plant is activated by mixing an appropriate amount of Trichoderma harzianum SK-5-5 bacteria into the floor soil or by applying to the rhizosphere such as spraying during growth. The roots, stems and leaves can be improved, disease prevention and resistance against pathogenic bacteria can be imparted. This not only drastically reduces the occurrence of disease at the time of growth, but also has various effects such as promoting the growth of plants, increasing the yield, and improving the sugar content.
However, the effect is presumed to be caused by a conidial spore product of Trichoderma harzianum SK-5-5 (for example, a novel amino acid). Since it is recognized that all disappear at the end of growth, there is no effect of any meaning, and there is an effect that no adverse effect is seen even in continuous use. Furthermore, since the product is recognized as a novel amino acid and its peripheral substances, it is harmless even if it remains in a small amount on the leaves and stems of plants.
またこの発明のトリコデルマ ハルジアナム SK−5−5菌は、土壌中で増殖するので、環境条件が増殖に適する場合には、当初の散布濃度が不十分の場合であっても、増殖により必要量の生成物質を補給し、所期の目的を達成した後、自然消滅する特性が認められ、人体への影響はもとより、土壌、植物その他環境破壊などのおそれは皆無である。更に連作不良の作物についても、連作の不利は解消されるものと推定された。 The Trichoderma harzianum SK-5-5 bacterium of the present invention grows in soil, so that if the environmental conditions are suitable for growth, even if the initial spraying concentration is insufficient, the necessary amount is increased by the growth. After replenishing the product and achieving the intended purpose, it has a characteristic of self-destruction, and there is no risk of soil, plant or other environmental damage, as well as impact on the human body. Furthermore, it was estimated that the disadvantages of continuous cropping would be resolved for crops with poor continuous cropping.
この発明は、トリコデルマ ハルジアナム SK−5−5菌を固体培地又は液体培地で培養して抗生物質を生成することを特徴とした植物の活性付与剤である。また製造方法の発明は、トリコデルマ ハルジアナム SK−5−5菌を米培地などの固体培地又は液体培地に植菌し、所定温度(例えば25℃〜30℃)で、所定時間(例えば4日〜30日)間培養した後、精製、抽出することを特徴とした植物の活性付与剤の製造方法である。 This invention is a plant activity-imparting agent characterized by culturing Trichoderma harzianum SK-5-5 bacteria in a solid medium or a liquid medium to produce antibiotics. In addition, the invention of the production method inoculates Trichoderma harzianum SK-5-5 in a solid medium or liquid medium such as a rice medium, and at a predetermined temperature (for example, 25 ° C. to 30 ° C.) for a predetermined time (for example, 4 days to 30 days). This is a method for producing a plant activity-imparting agent, characterized by purifying and extracting after culturing for 2 days).
また活性付与方法の発明はトリコデルマ ハルジアナム SK−5−5菌の分生胞子を根圏付近の覆土中に共存させるものである。この場合に前記分生胞子は多孔性セラミックス粒子を担体とする。このようにすれば、散布の容易性と、十分な酸素の補給性を確保することができるので好ましく、前記多孔性セラミックス粒子にキトサンその他、分生胞子の増殖時に必要な栄養分を補給することが一層好ましい。前記のようにすれば、分生胞子の増殖を円滑かつ確実にすることができるので、必然的に有用物質の単離生成物も多くなる。 The invention of the activity imparting method is one in which conidia of Trichoderma harzianum SK-5-5 are coexisted in the soil covering near the rhizosphere. In this case, the conidia have porous ceramic particles as a carrier. In this way, it is preferable because it is easy to spray and sufficient oxygen replenishment can be ensured, and the porous ceramic particles can be supplemented with chitosan and other nutrients necessary for the growth of conidia. Even more preferred. By doing so, the growth of conidia can be made smooth and reliable, and the number of isolated products of useful substances inevitably increases.
次に活性付与剤の発明は、土壌中の増殖環境を良好にする為に、土壌温度を15℃〜30℃とし、水分を30%以上とするが、その他の雑菌処理については必要性がない場合もある。更に施用方法の発明は、種子への付着、散布、土壌との混和、散布潅水による浸透などがある。 Next, the invention of the activity imparting agent has a soil temperature of 15 ° C. to 30 ° C. and a water content of 30% or more in order to improve the growth environment in the soil, but there is no need for other miscellaneous bacteria treatment. In some cases. Further, the invention of the application method includes adhesion to seeds, spraying, mixing with soil, and infiltration by spraying watering.
(供試菌名称)
Trichoderuma hurzianum SK-5-5(トリコテ゛ルマ ハルシ゛アナム SK-5-5)
供試菌は97年1月北海道グリーン興産より寒天培地のスラント1本を受領した。供試菌は下記の斜面培地に植菌し、28℃で5日間培養後、冷蔵にて保存した。
(Test bacteria name)
Trichoderuma hurzianum SK-5-5 (Trichoderma harzianum SK-5-5)
The test bacterium received a slant of agar medium from Hokkaido Green Kosan in January 1997. The test bacteria were inoculated into the following slant culture medium, cultured at 28 ° C. for 5 days, and stored in a refrigerator.
寒天斜面培地組成
オートミール 5.0%
シュクロース 5.0%
寒天 1.0%
28℃、5日間培養後冷蔵保存
Agar slope medium composition Oatmeal 5.0%
Sucrose 5.0%
Agar 1.0%
Refrigerated after incubation at 28 ° C for 5 days
(培養)
培養は500ml三角フラスコを用い、米培地(A培地)と液体培地(B培地I)の2種類で行った。A培地の条件は、28℃で静置培養(培養途中に滅菌水10mlを添加)とし、B培地Iの培養は、28℃で270rpmの振盪培養をした。
培地組成:A培地
米 100%
大豆かす 3%
滅菌水 10%
培地組成:B培地I
グルコース 5.0%
ポリペプトン 0.5%
NaCl 0.8%
酵母エキス 0.2%
炭酸カルシウム 1.0%
(culture)
Cultivation was performed in two types of rice medium (A medium) and liquid medium (B medium I) using a 500 ml Erlenmeyer flask. The condition of the medium A was stationary culture at 28 ° C. (10 ml of sterilized water was added during the culture), and the culture of the medium B was shake culture at 28 ° C. at 270 rpm.
Medium composition: A medium 100% rice
Soybean ground 3%
Medium composition: B medium I
Glucose 5.0%
Polypeptone 0.5%
NaCl 0.8%
Yeast extract 0.2%
Calcium carbonate 1.0%
(検定法)
検定は、すべてペーパーディスク平板法によるin vitroで行い、供試菌には下記の菌を用いた。
(Test method)
All tests were performed in vitro by the paper disk plate method, and the following bacteria were used as test bacteria.
(1) Rhizoctonia solani(AG-1IA)
検定用培地にはPDA(Nissui)1.3%、Chloramphenicol 0.002%の組成からなる培地を用いた。Rhizoctonia の菌糸の先端をコルクボーラーで抜いたものを平板培地の中央にのせ、ブロスを染み込ませたペーパーディスクを置き菌糸の伸長の様子を観察した。
(1) Rhizoctonia solani (AG-1IA)
A medium having a composition of PDA (Nissui) 1.3% and Chloramphenicol 0.002% was used as an assay medium. The tip of Rhizoctonia hyphae was removed with a cork borer and placed in the center of the plate medium, and a paper disk soaked with broth was placed to observe the growth of the hyphae.
(2) Botrytis cinerea
検定用培地にはPotato-extract 20.0%、Sucrose 2.0%、寒天1.5%の組成からなる培地を用いた。検定はペーパーディスクに試料をのせ風乾後シャーレにのせた。シャーレをインキュベーターに入れて培養し、ペーパーディスクの周りに阻止円が形成されているか否かを観察した。
(2) Botrytis cinerea
A medium comprising a composition of Potato-extract 20.0%, Sucrose 2.0%, and agar 1.5% was used as the assay medium. The test was carried out by placing the sample on a paper disc, air-drying and then placing on a petri dish. The petri dish was placed in an incubator and cultured, and it was observed whether or not a blocking circle was formed around the paper disk.
(3) 抗菌スペクトラム検定菌
抗菌スペクトラムを下記の検定菌を用いて測定した。
Bacillus subtilis ATCC6633
Micrococcus luters ATCC6633
Staphylococcus aureus 209P
Escherichia coli NIHJ
Saccharomyces cerevisiae SHY3
Candida albicans M9001
Candida pseudotropicalis M9035
Cryptococcus neoformans M9010
Debaryomyces hansenii M9011
Trigonopsis variabilis M9031
Schizosaccharomyces pombe M9025
Hansenula schneggi IAM4269
(3) Antibacterial spectrum test bacteria The antibacterial spectrum was measured using the following test bacteria.
Bacillus subtilis ATCC6633
Micrococcus luters ATCC6633
Staphylococcus aureus 209P
Escherichia coli NIHJ
Saccharomyces cerevisiae SHY3
Candida albicans M9001
Candida pseudotropicalis M9035
Cryptococcus neoformans M9010
Debaryomyces hansenii M9011
Trigonopsis variabilis M9031
Schizosaccharomyces pombe M9025
Hansenula schneggi IAM4269
(培養法)
(1) A培地培養法
Rhizoctonia solani(AG-1IA) 活性成物
糸状菌Rhizoctonia solani(AG-1IA)に対するA培地の培養抽出液活性物の単離精製。
A培地(米培地1kg)500mlの三角フラスコ1本に米100gと大豆かす3gを加え、同じ条件のもの10本分の培地を調製した。滅菌後エーゼで植菌して、28℃10日間(菌がよく増殖するように途中何度かフラスコを振る。また培地の表面が乾いてきたら滅菌水を添加する。)静置培養後、50%アセトン水を2リットル加え抽出をした。ここで培養物50%アセトン抽出液2リットルを得た。
(Culture method)
(1) A medium culture method
Rhizoctonia solani (AG-1IA) active product Isolation and purification of the culture extract actives of medium A against the filamentous fungus Rhizoctonia solani (AG-1IA).
100 g of rice and 3 g of soybean meal were added to a 500 ml Erlenmeyer flask of A medium (1 kg of rice medium) to prepare 10 mediums under the same conditions. After sterilization, inoculate with ase and shake at 280 ° C for 10 days (shake the flask several times in the middle so that the bacteria can grow well. Add sterile water when the surface of the medium has dried.) Extraction was performed by adding 2 liters of% acetone water. Here, 2 liters of 50% acetone extract of the culture was obtained.
(2) B培地培養法
Staphylococcus aureus 209P 活性成物
細菌Staphylococcus aureus 209Pに対するB培地培養抽出液中活性物の単離精製。
(2) B medium culture method
Staphylococcus aureus 209P active product Isolation and purification of active product in B medium culture extract against bacterium Staphylococcus aureus 209P.
液体培地であるB培地Iの抗細菌活性成分をより多く生産する目的で培地組成の窒素源と炭素源の割合が異なるI〜IVの4種を比較検討した。各培地500ml三角フラスコ2本を用い、培養5日間後培養液を5倍濃縮に調製し、検定に用いた。 For the purpose of producing more antibacterial active ingredients of B medium I, which is a liquid medium, four types I to IV having different ratios of nitrogen source and carbon source in the medium composition were compared. Two 500 ml Erlenmeyer flasks were used for each medium, and after 5 days of culture, the culture solution was concentrated to 5 times and used for the assay.
(3) B培地I培養法培地
検討の結果よりB培地Iで精製用培養を行ったB培地(グルコース 5.0%、ポリペプトン 0.5%、NaCl 0.8%、酵母エキス 0.2%、炭酸カルシウム 1.0%)500ml三角フラスコに、100mlの培地を調製した。滅菌後植菌し、28℃、5日間、振盪培養を行った。計1.5リットルの培養液と等量のアセトンを加え、50%アセトン抽出液3リットルを得た。
(3) B medium I culture method medium B medium (Glucose 5.0%, Polypeptone 0.5%, NaCl 0.8%, Yeast extract 0.2%) , Calcium carbonate 1.0%) In a 500 ml Erlenmeyer flask, 100 ml of medium was prepared. After sterilization, the cells were inoculated and cultured at 28 ° C. for 5 days with shaking. A total of 1.5 liters of the culture solution and an equal amount of acetone were added to obtain 3 liters of 50% acetone extract.
(精製法)
(1) A培地培養抽出物精製法A培地培養50%アセトン抽出液2リットルのアセトン留去後、1リットルをHP−20−Sephadex カラム(60.0cm×4.5cm φ)に全量を通過吸着させた。等量の水で洗浄後、等量の50%アセトン、次いで等量の100%アセトンで溶離させた。それぞれの分画を検定菌Rhizoctonia solani(AG-1IA)で検定後、活性物質が100%アセトン溶離部に溶離されていることを確認した。続いて100%アセトン溶離部1リットルを濃縮し、1098mgの粗精製物を得て、その中の100mgを展開溶媒メタノール系のLH−20カラムにかけた(100.0cm×2.0cmφ)。分画をアッセイし、活性分画を確認した。これら活性分画を回収後シリカゲルTLC(酢酸エチル:酢酸:水=5:1:1)で展開し、モリブデン硫酸呈色反応をしたところ、Rf=0.3付近にスポットが検出された。同時に不活性分画の展開部にはこれらのスポットは検出されなかった。このことから活性物はRf=0.3付近に検出されるスポットと推定し、さらに精製を進めた。シリカゲルPTLC展開(酢酸エチル:酢酸:水=5:1:1)、かき取りメタノール抽出後、HPLCを用いて単離精製をした。単離精製物は器機分析を実施し同定をした(図31)。
(Purification method)
(1) A medium culture extract purification method A
(2) B培地I培養抽出物精製法
B培地培養50%アセトン抽出液3リットルのアセトン留去後(1.5リットル)、活性炭吸着カラム(60.0cm×2.0cm φ)に全量1.5リットルを通過、吸着させた。等量の水で洗浄後、等量の50%アセトン、続いて等量の100%アセトンで溶離させた。各分画を検定菌Staphylococcus aureus 209Pを用いて検定した結果、100%アセトン溶離部に活性物質が溶離されていることが確認された。100%アセトン溶離部1.5リットルを濃縮し、酢酸エチルで分配抽出を行い、次いでメタノールを展開溶媒とするLH−20を実施し、HPLCで単離精製を行った。単離精製物は機器分析を実施し同定をした(図32)。
(2) B medium I culture extract purification method
(単離精製物生物活性法)
(1) A培地単離精製物評価法
4成分の生物評価法
検定用培地にはPDA(Nissui) 1.3%、Choloramphenicol 0.002%の組成からなる培地を用いた。Rhizoctonia solani(AG-1IA)の菌糸の先端をコルクボーラーで抜いたものを平板培地の中央に乗せ、サイドに調製した単離物質を染み込ませたペーパーディスクを置き菌糸の伸長の様子を観察した。
(Isolated and purified biological activity method)
(1) A medium isolated and purified product evaluation method 4-component biological evaluation method A medium comprising PDA (Nissui) 1.3% and Choloramphenicol 0.002% was used as the assay medium. The hyphae of Rhizoctonia solani (AG-1IA), which had been extracted with a cork borer, was placed on the center of the plate medium, and a paper disk impregnated with the isolated substance was placed on the side to observe the state of hyphal elongation.
(2) B培地I単離精製物評価物
C成分の生物評価(MW168)
単離精製したC成分(MW:168)は新規物であり、きわめて単純な構造を保持している。母核としての興味が持たれた為生物評価を拡大して実施した。
(2) B medium I isolated and purified product evaluation biological evaluation of C component (MW168)
The isolated and purified component C (MW: 168) is novel and retains a very simple structure. The biological evaluation was expanded and carried out because of interest as a mother nucleus.
(3) バクテリア属の評価
Bioassay
試験菌を加えた寒天培地をシャーレに入れ固める。その上に試料を含むペーパーディスクをのせ、37℃で18時間培養した後、発育阻止円の形成を確認する。
(3) Evaluation of bacteria
Bioassay
Put the agar medium with the test bacteria in a petri dish and harden. A paper disk containing the sample is placed on it and cultured at 37 ° C. for 18 hours, and then the formation of a growth inhibition circle is confirmed.
(a) 使用菌株
Staphylococcus aureus 209P、Pseudomonas syringal(タバコ野火病菌)、Xanthomonoas Campestris pv.citri(カンキツかいよう病)、Erwinia sp(ウメかいよう病)の4菌株を用いた。
(a) Strains used
Four strains were used: Staphylococcus aureus 209P, Pseudomonas syringal (tobacco wildfire fungus), Xanthomonoas Campestris pv.citri (citrus canker), Erwinia sp (plum canker).
(b) 使用培地
前培養にはブイヨン培地 (DIFCO)を用い、一晩培養後、×102 に希釈し、これを上層の培地0.5%に混ぜ、菌測定培地には MYCIN AGAR (ミクニ化学)を用いた。
上層 − MYCIN AGAR 1.5%(ミクニ化学)+ブイヨン2%培地 (DIFCO)
にて一晩培養した培養液×102 に希釈したものを0.5%加える。
下層 − MYCIN AGAR 2.0%(ミクニ化学)
(b) using the spent medium before bouillon medium for culturing (DIFCO), cultured overnight, diluted × 10 2, which mixed with 0.5% top layer of the medium, the bacteria measurement medium MYCIN AGAR (MIKUNI Chemistry) was used.
Upper layer-MYCIN AGAR 1.5% (Mikuni Chemical) +
Add 0.5% of the diluted culture broth overnight at 102.
Lower layer-MYCIN AGAR 2.0% (Mikuni Chemical)
(c) 抗菌測定
ペーパーディスクに1000pppm,500ppm,250ppmに調製した単離精製物を染み込ませ、風乾後シャーレに乗せ、37℃で18時間培養し、発育阻止円の形成の有無を観察した。
(c) Antibacterial measurement A paper disk was impregnated with an isolated and purified product prepared at 1000 pppm, 500 ppm, and 250 ppm, air-dried, placed on a petri dish, cultured at 37 ° C. for 18 hours, and observed for the presence of growth-inhibiting circles.
(4) ブドウ球菌に対する抗菌力(MIC)の測定
(a) 使用菌株
S.aureusu 209P JC−1,第1G保存の臨床分離黄色ブドウ球菌20株(MSSA,MRSA 各10株)及び基準菌株である E.coli NIHJ JC−2の計22株を用いた。
(4) Measurement of antimicrobial activity (MIC) against staphylococci
(a) Strains used
A total of 22 strains of S. aureusu 209P JC-1, 20 clinical isolates of Staphylococcus aureus (MSSA, MRSA each 10 strains) preserved in the 1G and the reference strain E. coli NIHJ JC-2 were used.
(b) 使用抗菌薬
MW:168、methicllin(DMPPC,注射用スタフシリン、Lot.No.FSB 19,900μg/mg、萬有製薬)、vancomycin (VCM,Lot.No.41H0457,10750SIGMA)
(b) Antibacterial drugs used
MW: 168, methicllin (DMPPC, Stafcillin for injection, Lot.No.FSB 19,900μg / mg, Ariyu Pharmaceutical), vancomycin (VCM, Lot.No.41H0457,10750SIGMA)
(c) 使用培地
抗菌力測定にはMueller-Hinton agar (MIA:Difco)を用い前培養には、Mueller Hinton broth(MHB:Difco)を用いた。
(c) Medium used Mueller-Hinton agar (MIA: Difco) was used for antibacterial activity measurement, and Mueller Hinton broth (MHB: Difco) was used for preculture.
(d) 抗菌力測定
使用菌株に対する各薬剤の最小発育阻止濃度(MIC)は日本化学療法学会標準法に準拠し、寒天平板希釈法にて測定した。菌株はMHAに塗抹し、37℃で一晩培養して生育したコロニーをMHBにて37℃で一晩培養し、その菌液を100倍希釈(E.Coliのみ1000倍希釈)したものを接種菌液とした。
(d) Antibacterial activity measurement The minimum inhibitory concentration (MIC) of each drug against the strain used was measured by the agar plate dilution method in accordance with the standard method of the Japanese Society of Chemotherapy. The strain is smeared on MHA, and the colony grown by overnight culture at 37 ° C is cultured overnight at 37 ° C in MHB, and the bacterial solution is diluted 100 times (only E. Coli is diluted 1000 times). Bacterial fluid was used.
(結果)
(1) 検定菌の結果A培地とB培地Iの培養液活性は表2の通りである。
抗菌スペクトラムを下記の検定菌を用いて測定した。
(result)
(1) As a result of the test bacteria, the culture medium activities of A medium and B medium I are as shown in Table 2.
The antibacterial spectrum was measured using the following test bacteria.
A培地の培養抽出液中の検定の結果、Rhizoctonia solani(AG-1IA)の菌糸がA培地培養抽出物のペーパーディスクを避けている様子が観察された。本菌が生産するA培地培養液中の活性物が、菌糸の成長を妨げる物質を生産していると推察した。このためA培地培養液中より、Rhizoctonia solani(AG-1IA)に阻害活性を示す物質を指標に単離精製を試みた。 As a result of the assay in the culture medium A extract, it was observed that the mycelium of Rhizoctonia solani (AG-1IA) was avoiding the paper disk of the medium A culture extract. It was inferred that the active substance in the medium A culture medium produced by this bacterium produced a substance that hinders mycelial growth. For this reason, isolation and purification were attempted from the medium A culture medium using as an index a substance exhibiting inhibitory activity against Rhizoctonia solani (AG-1IA).
またB培地Iの培養抽出液中の検定の結果、Botrytis cinerea、Rhizoctoniasolaniに活性は確認されず、バクテリアにのみ発育阻止円の形成が確認された。このことは、本菌は拮抗作用以外の抗菌力があると見られ、抗生物質を生産している可能性が考えられた。特にStaphylococcus aureus 209Pに形成された発育阻止円はクリアーであった為、B培地IではバクテリアStaphylococcus aureus 209Pに対する活性物質を指標に単離精製を実施することにした。A培地培養物とB培地I培養物の抗菌スペクトラムは異なっており、それぞれ系統の異なるものを生産していると思われた。それぞれの活性物を単離精製することとした。 Further, as a result of the assay in the culture extract of B medium I, activity was not confirmed in Botrytis cinerea and Rhizoctoniasolani, and formation of growth inhibition circles was confirmed only in bacteria. This indicates that this bacterium has antibacterial activity other than antagonism and may produce antibiotics. In particular, since the growth inhibition circle formed in Staphylococcus aureus 209P was clear, it was decided to carry out isolation and purification in B medium I using the active substance against bacteria Staphylococcus aureus 209P as an index. The antibacterial spectrum of the A medium culture and the B medium I culture were different, and it seemed that they produced different strains. Each active product was isolated and purified.
(2) 培養の結果
(a) A培地培養結果
A培地(米培地 1kg)500mlの三角フラスコ10本分を28℃で10日間、静置培養し、培養物50%アセトン抽出液2リットルを得た。
(2) Culture results
(a) Culture result of A medium Ten medium culture flasks (rice medium 1 kg) of 500 ml Erlenmeyer flasks were statically cultured at 28 ° C. for 10 days to obtain 2 liters of 50% acetone extract of the culture.
(b) B培地検討と培養結果
表1に表したB培地IとB培地IIに活性が認められ、B培地Iにより強い活性が認められた本菌は、窒素源が少ない条件で活性物を生産することが確認された。この結果よりB培地Iを用いて28℃で5日間、振盪培養をした。
(b) B medium examination and culture results The activity of B medium I and B medium II shown in Table 1 and the strong activity of B medium I were confirmed. Production was confirmed. From this result, shaking culture was performed using B medium I at 28 ° C. for 5 days.
(3) 精製結果
(a) A培地培養精製結果
シリカゲルPTLC処理後の活性分画を粗精製物として、HPLC(0.05%TFA含 MeOH/H2O)で純度の確認を行った。分析では0分から30分にかけてメタノール0%から100%のグラジエントをかけ、さらに30分から100%メタノールで流速1ml/minで分析したところ、RT(リテションタイム)32.5分(Fr65)で検出された(図1)。一見、単一物質のピークが検出されたかのように思われたが、拡大することにより複数ピークの存在が明らかとなった(図2)。RTの速い方からA,B,C,D,Eと称し(図2)、そのうちの4成分を単離精製した。単離精製物の成分と量は、A成分 7.2mg,B成分12.1mg,D成分 3.5mg,E成分 4.5mgであった。ピークCは量が少量であり、精製は断念した。
(3) Purification results
(a) Results of culture and purification of medium A Using the active fraction after silica gel PTLC treatment as a crude product, purity was confirmed by HPLC (0.05% TFA-containing MeOH / H 2 O). In the analysis, a gradient from 0% to 100% methanol was applied from 0 to 30 minutes, and an analysis was performed at a flow rate of 1 ml / min from 30 minutes to 100% methanol. RT (retention time) was detected at 32.5 minutes (Fr65) ( FIG. 1). At first glance, it seemed as if a single substance peak was detected, but the presence of multiple peaks became apparent by enlargement (FIG. 2). From the fastest RT, they were called A, B, C, D, and E (FIG. 2), and four of them were isolated and purified. The components and amount of the isolated and purified product were A component 7.2 mg, B component 12.1 mg, D component 3.5 mg, and E component 4.5 mg. Peak C was a small amount and purification was abandoned.
(b) B培地I培養精製結果
それぞれの分画のアッセイより活性Fr41〜54に活性があることを確認し回収した。HPLC(0.05%TFA含MeOH/H2O)分取、分析を行い検出されたピークのRTの速い方からA,B,C,D,Eと数え、5成分の存在を確認した。このうちC成分 8.1mg単離精製することが出来た。
(b) B medium I culture purification result It was confirmed that the active Fr41 to 54 had activity from the assay of each fraction and recovered. HPLC (0.05% TFA-containing MeOH / H 2 O) was collected and analyzed, and the detected peaks were counted as A, B, C, D, E from the fastest RT, and the presence of 5 components was confirmed. Of these, 8.1 mg of component C was isolated and purified.
(4) 機器分析の結果
(a) A培地単離精製物UV吸収スペクトラム、質量分析及びNMR測定結果(図3〜14)より、Aib(α-Aminoisobutyric acid)を含むPolypeptideの、Peptibols系と判断した。Peptibols系抗生物質はアミノ酸配列の中に α-Aminoisobutyric acid(Aib)を含んでおり、N末端がAcetyl基で、C末端が Amino alcohol結合であることが特徴である(多くは Phenylalaniol基でおわる)。なお今回の精製物、A成分、B成分、D成分は質量分析より一部のアミノ酸配列を下記のように推定し、その部分構造を既存のPeptibols のアミノ酸配列と検索した結果、一致する物が無く、新規 Peptibols系であると推定した。Eはアミノ酸配列の推定が困難であるため判断できなかった。
(4) Results of instrument analysis
(a) Based on the UV absorption spectrum, mass spectrometry and NMR measurement results (FIGS. 3 to 14) of the A medium isolated and purified product, it was determined that the polypeptide containing Aib (α-Aminoisobutyric acid) was a Peptibols system. Peptibols antibiotics contain α-Aminoisobutyric acid (Aib) in the amino acid sequence, and are characterized by an N-terminal Acetyl group and a C-terminal Amino alcohol bond (mostly Phenylalaniol groups) . As for the purified product, A component, B component, and D component, the partial amino acid sequence was estimated by mass spectrometry as follows, and the partial structure was searched with the existing amino acid sequence of Peptibols. There was no new Peptibols system. E could not be determined because it was difficult to estimate the amino acid sequence.
UV吸収波長:4成分とも末端吸収(図3、図6、図9、図12)
A成分=1933(図5)
B成分=1949または1964(図8)
D成分=1810(図11)
E成分=1829(図14)
推定構造
A成分:Ac-Aib-Ala-Aib-Aib-Aib-Aib-Gln-Aib-Aib-...
B成分:Ac-Aib-Ala-Aib-Aib-Val-Aib-Gln-Aib-Aib-...
D成分:Ac-Aib-Ala-Aib-Aib-Aib-...
UV absorption wavelength: terminal absorption of all four components (FIGS. 3, 6, 9, and 12)
A component = 1933 (FIG. 5)
B component = 1949 or 1964 (FIG. 8)
D component = 1810 (FIG. 11)
E component = 1829 (Fig. 14)
Estimated structure A component: Ac-Aib-Ala-Aib-Aib-Aib-Aib-Gln-Aib-Aib -...
B component: Ac-Aib-Ala-Aib-Aib-Val-Aib-Gln-Aib-Aib -...
D component: Ac-Aib-Ala-Aib-Aib-Aib -...
(b) B培地I(液体培地)活性物
機器分析の結果C成分は、分子量168(C8H8O4)の新規物質であることが推定された。他の成分について単離精製は非常に困難であり、不可能と判断したが、LC/MS を実施した。A成分のUV吸収波長は末端であった(図15)。HPLCの条件はカラムCapcel pac C18 UG120 (2×150mm)を用い、1%酢酸:アセトニトリル=98:2、流速0.2ml/min、温室の条件で分析した。A成分は、RT7.48分に検出された(図16)。A成分が検出された分画の質量分析の結果、分子量は192であることを推定した(図17)。同じくB成分についても同様の分析をしたところB成分もUV吸収波長が末端(図18)で、HPLCのRT 12、45分に検出された(図19)。質量分析の結果、分子量は206であることを推定した(図20)。C成分は単離精製が可能であった分画である。C成分のUV吸収波長は270nm(図21)で、HPLCのRTは15.92分に検出された(図22)。質量分析の結果、分子量は168であることを推定した(図23、24)。また溶媒CD3ODで溶解し、NMRを測定(図25)、解析したところ五員環を含み、共役結合をもつ新規物質であることが推定された。推定構造を図26に示した。D成分はUV吸収波長が230nm付近にあり(図27)、HPLCのRTは30.28分に検出された(図28)。しかし再度HPLCを実施し、UVスペクトラム検出器の波長を末端と230nmで分析した結果、RT0.6分の差で2成分(D1,D2)存在していることが確認された(図29)。それぞれの質量分析の結果、RTの速いUV吸収が230nmのものは分子量154であった(D1)。たまRTが0.6分遅れて検出される成分の分子量は220であることを推定した(D2)(図30)。これらの分子量の明らかとなった成分を分子量の少ない順にならべると154(D1),168(C),192(A),206(B),220(D2)であることが確認された。
(b) B medium I (liquid medium) active substance As a result of instrumental analysis, it was estimated that component C is a novel substance having a molecular weight of 168 (C 8 H 8 O 4 ). Although it was judged that isolation and purification of other components were very difficult and impossible, LC / MS was performed. The UV absorption wavelength of component A was terminal (FIG. 15). The HPLC was performed using a column Capcel pac C18 UG120 (2 × 150 mm), 1% acetic acid: acetonitrile = 98: 2, flow rate 0.2 ml / min, and greenhouse conditions. A component was detected at RT 7.48 minutes (FIG. 16). As a result of mass spectrometry of the fraction in which the A component was detected, it was estimated that the molecular weight was 192 (FIG. 17). Similarly, the same analysis was performed on the B component, and the B component was detected at
(5) 生物評価結果
(a) A培地培養単離精製物生物評価結果
4成分の生物評価結果
A,B,C,D成分について5000ppmから希釈し、5000ppm,2500ppm,1250ppm,625ppmで検定した。結果A成分、E成分は1250ppmまで Rhizoctonia solaniの菌糸の伸長を阻害しおり、B成分、D成分は625ppmまでRhizoctonia solaniの菌糸の伸長を阻害しているのが確認された。
(5) Biological evaluation results
(a) Biological evaluation results of culture medium isolated and purified from A medium Biological evaluation results of four components A, B, C, and D components were diluted from 5000 ppm and tested at 5000 ppm, 2500 ppm, 1250 ppm, and 625 ppm. As a result, it was confirmed that the A component and the E component inhibited the mycelial elongation of Rhizoctonia solani up to 1250 ppm, and the B component and the D component inhibited the mycelial elongation of Rhizoctonia solani up to 625 ppm.
(b) B培地I培養単離精製物生物評価結果
(イ) バクテリア属の評価結果
B培養物(MW168) のバクテリアに対する生物活性は発育阻止円が形成されていなかったことから抗菌力は無いか、弱いものと考えられた。
(b) B medium I culture isolated and purified product biological evaluation results
(B) Bacteria genus evaluation results The biological activity of B culture (MW168) against bacteria was considered to have no antibacterial activity or weakness because no growth inhibition circle was formed.
(ロ) ブドウ球菌に対する抗菌力(MIC)の測定結果は表3の通りである。 (B) Table 3 shows the measurement results of antibacterial activity (MIC) against staphylococci.
表3に示したようにB培養物(MW168) は今回測定した濃度において抗菌力を示さず、ブドウ球菌に対する抗菌力は無いか極めて弱いものと考えられた。 As shown in Table 3, the B culture (MW168) did not show antibacterial activity at the concentration measured this time, and was considered to have no or extremely weak antibacterial activity against staphylococci.
考察A培地培養物より単離精製した成分からは、Aib(α-Aminoisobutyric acid)を含むPeptibols系のもの4成分を単離することが出来た。Trichodermaが生産する Peptibols系のものは幾つかすでに報告されているが、今回単離精製したものは、それらのものとは一致しなかった。また、それぞれの成分のアミノ酸配列の結果、3成分が新規の配列であることが推定された。(E成分のみ配列を測定するのが不可能だった)さらにin vitro試験ではRhizoctonia solani(AG-1IA)の菌糸を阻害している様子が観察された。この結果から本菌の米培養抽出物中にRhizoctonia solani(AG-1IA)に対する菌糸伸長阻害物質が生産されていることが明らかとなった。本菌の拮抗作用である相手菌糸との接触により発現する細胞質凝集を促し死滅させる物質そのものであるという断言はまだ出来ないが、本菌の米培養抽出物中に菌糸成長伸長抑制物が生産されていることは明らかである。 Discussion From the components isolated and purified from the culture medium A, 4 components of Peptibols containing Aib (α-Aminoisobutyric acid) could be isolated. Several Peptibols strains produced by Trichoderma have already been reported, but those isolated and purified this time did not match those. Moreover, as a result of the amino acid sequence of each component, it was estimated that three components are novel sequences. (It was impossible to measure the sequence of only the E component) Further, in vitro tests showed that the mycelium of Rhizoctonia solani (AG-1IA) was inhibited. From these results, it was revealed that a mycelial elongation inhibitory substance against Rhizoctonia solani (AG-1IA) was produced in the rice culture extract of this bacterium. Although it is not yet possible to assert that the substance itself is a substance that promotes and kills cytoplasmic aggregation that is expressed by contact with the partner mycelium, which is an antagonistic action of this fungus, a mycelium growth elongation inhibitor is produced in the rice culture extract of this fungus. It is clear that
またB培地I培養より単離精製した成分はMW:168の新規(3(3-hydroxy-cyclopropene 5-one-2yl)2-propenoic asid)であり、Bioassay を幾つか実施したが活性は無いか、きわめて弱いものと判断した。おそらく活性本体はMW:168周辺化合物であり、MW:168は活性物の副成物であると推測している。またMW:168周辺化合物の分子量はMW:154、MW:192、MW:206、MW:220であり、分子量の差がそれぞれ14であることがわかる。これはメチル基一個分と同量であり、メチル基の増減による活性の違いも考えられる。またMW:168は新規な構造を保持しているが、構造検索より類似していた化合物が幾つか報告されていた。その中でMW:168に一番近い構造であったもの(図33)は、鳥取大学のPGR活性スクリーニングで見出されたものであり、糸状菌Penicillium valiabile SOPPより代謝され単離精製されたと報告され、C8H8O5 (MW:198)の組成式で表されている新規物質であるが、活性は弱いものと報告されている。 In addition, the component isolated and purified from B medium I culture is a new MW: 168 (3 (3-hydroxy-cyclopropene 5-one-2yl) 2-propenoic asid). Judged that it was extremely weak. Presumably, the active body is a peripheral compound of MW: 168, and MW: 168 is a by-product of the active substance. The molecular weights of the MW: 168 peripheral compounds are MW: 154, MW: 192, MW: 206, and MW: 220, and the difference in molecular weight is 14 respectively. This is the same amount as that of one methyl group, and a difference in activity due to increase or decrease of the methyl group is also considered. In addition, although MW: 168 has a new structure, several similar compounds have been reported by structure search. Among them, the structure closest to MW: 168 (Fig. 33) was found by PGR activity screening at Tottori University, and was reported to be isolated and purified from the filamentous fungus Penicillium valiabile SOPP. Although it is a novel substance represented by the composition formula of C 8 H 8 O 5 (MW: 198), its activity is reported to be weak.
この発明の植物の活性促進剤をぎんがメロンについて農地で施用した所、次の結果を得た。 The following results were obtained when the plant activity promoter of the present invention was applied to farmland for Ginkgo melon.
実施場所 北海道常呂郡訓子府町弥生
田川農園
実施期間 平成11年4月23日〜同年7月29日
実施条件 アグロミックSK−10(トリコデルマ ハルジアナム SK−5−5菌の商標、以下同じ)を50g/m2の割合でメロン床土に散布した。
直径1〜2mmの麦飯石に、5×107/gの分生胞子を付着させてあった。試験区、対照区共に200m2で、両区の相違は、アグロミックSK−10の散布の有無だけであった。農薬、追肥は一切しなかった。
Location Yayoi Kunikofu Town, Tokoro-gun, Hokkaido
Tagawa Farm Implementation period April 23, 1999-July 29, 1999 Implementation conditions Agromic SK-10 (Tricoderma harzianum SK-5-5 trademark, hereinafter the same) is applied to melon bed soil at a rate of 50 g / m 2 Scattered.
5 × 10 7 / g conidia were attached to barleystone with a diameter of 1 to 2 mm. Both the test group and the control group were 200 m 2 , and the difference between the two groups was only whether or not the agromic SK-10 was sprayed. There was no pesticide or topdressing.
前記アグロミックSK−10を平成11年4月23日散布、平成11年4月26日定植、平成11年7月29日収穫を開始した。ぎんがメロンの平均重量と糖度は表4の通りである。 The agromic SK-10 was sprayed on April 23, 1999, planted on April 26, 1999, and harvested on July 29, 1999. Table 4 shows the average weight and sugar content of Ginger melon.
調査経過の特徴
(1) 初期段階の特徴として、根の活着が顕著で葉形が大きく茎が太い上に花めが大きい。
(2) 中間階段の特徴は、つる(ヘタ)の状態がガッチリして対照区と大きな格差があり、勿論農薬の使用は一切無く苗の立枯れ、つる割れ病、つる枯れ病の兆候も無かった。
(3) 収穫時の特徴としては、果実が大きく形が揃っており、ネットは太くて、張りは抜群であった。
(4) 食味検査について、果実全体に糖度が等しく、外皮ぎりぎりまで果肉が柔らかく、糖分が高く、しかもさらっとして上品な味覚である(対照区のメロンとは確実に格差を生じた)。
(5) 果実がしまっており、収穫後相当長期(対照区の1.5倍以上)の保存が可能であった。
Characteristics of the survey process
(1) The characteristics of the initial stage are remarkable root survival, large leaf shapes, thick stems and large flowers.
(2) The characteristic of the middle staircase is that the state of the vine is sharp and there is a big difference from the control zone. Of course, there is no use of pesticides, and there are no signs of seedling dying, vine cracking disease, vine wilt disease. It was.
(3) As for the characteristics at the time of harvest, the fruits were large and shaped, the net was thick, and the tension was outstanding.
(4) Regarding the taste test, the sugar content is the same throughout the fruit, the flesh is soft up to the bare skin, the sugar content is high, and the taste is smooth and refined.
(5) Fruits were trapped and could be preserved for a considerable period (more than 1.5 times that of the control plot) after harvesting.
実施場所 明治製菓株式会社生物科学研究所
実施期間 平成11年4月〜同年8月
実施条件 4月6日播種
4月8日〜22日発芽、育苗
5月7日定植、アグロミックSK−10の散布
2×107/g、30〜40g/m2
6月4日〜7日交配
7月12日〜23日収穫
前記中適宜施肥、潅水した。前記実施により、表5の結果を得た。
Place of Implementation Meiji Seika Co., Ltd. Biological Science Laboratory Implementation period April 1999-August August Implementation conditions Seeding April 6
April 8-22, germination, seedling
May 7 fixed planting, spraying of agromic SK-10
2 × 10 7 / g, 30-40 g / m 2
June 4-7 mating
Harvest from July 12 to 23 Fertilization and irrigation were conducted as needed during the above period. According to the above implementation, the results shown in Table 5 were obtained.
結果
糖度は施用区の方が1.5%高かった。SK−10施用によりショ糖量が上昇し、ブドウ糖、果糖が抑制された。SK−10の一度の施用により糖度が向上したことは、根圏における相互作用により光合成産物の転流その他活性が付与された結果である。
Results The sugar content was 1.5% higher in the application area. Application of SK-10 increased the amount of sucrose and suppressed glucose and fructose. The improvement in sugar content by a single application of SK-10 is a result of the translocation of the photosynthetic product and other activities provided by the interaction in the rhizosphere.
この発明の植物の活性促進剤をてん菜について施用した所、次の結果を得た。 When the plant activity promoter of this invention was applied to sugar beet, the following results were obtained.
実施場所 北海道斜里町朱丹
実施期間 平成11年3月12日〜同年10月7日
実施条件 ポット上部土約30kgに対し、アグロミックSK−55(トリコデルマ ハルジアナム SK−5−5菌の商標)相当量を混和処理した。分生胞子は2×108/g以上であった。覆土に対し殺菌処理なし。菌定着の為に3月下旬〜4月中旬、週一回程度潅水を実施した。7月30日アグロミックSK−55の100倍液を株元と葉面に2リットル/m2噴霧散布した。処理区と対照区との相違は、殺菌処理しないことと、アグロミックSK−55の処理をしないことであり、施肥量、施肥方法その他一切同一とした。
Implementation place Shadan, Shari-cho, Hokkaido Implementation period March 12, 1999 to October 7, 1999 Implementation conditions Amount of equivalent to Agromic SK-55 (Tricoderma harzianam SK-5-5) for about 30 kg of pot soil Blended. Conidia were 2 × 10 8 / g or more. No sterilization treatment for soil covering. Water irrigation was carried out once a week from late March to mid April for colonization. On July 30, 100 liters of Agromic SK-55 was sprayed onto the strain and the leaf surface by 2 liters / m 2 . The difference between the treatment group and the control group is that no sterilization treatment and no treatment of Agromic SK-55 were made, and the amount of fertilization, the method of fertilization, etc. were completely the same.
結果
上記実施について、平成10年7月29日収穫した所、表6の実数、表7の比率の結果を得た。
Results Regarding the above implementation, the result of harvesting on July 29, 1998, the real numbers in Table 6, and the ratios in Table 7 were obtained.
前記実施例によれば、根重が32%増加し、糖量が40%増加している。従って同一面積で40%増収したことになり、大変な成果である。 According to the example, the root weight is increased by 32% and the sugar amount is increased by 40%. Therefore, the same area increased by 40%, which is a great achievement.
前記実施例によれば、発芽時の混和と、7月30日にアグロミック SK−55の散布で、収量の著しい増加が認められたことは、アグロミック SK−55により、てん菜の根茎を活性化し、これにより養分吸収その他が合理的、かつ強力になり、細胞分裂も順調になったものと推定される。 According to the above example, a remarkable increase in yield was observed by mixing at germination and application of Agromic SK-55 on July 30. Agromic SK-55 activated sugar beet roots. This suggests that nutrient absorption and other factors are rational and powerful, and that cell division is also smooth.
てん菜の場合にも種子の処理、土壌混和、中間散布など、今後は施用量と時機を研究することにより、更なる利点が浮上する可能性がある。 In the case of sugar beet, further benefits may emerge in the future by studying the application rate and timing, such as seed treatment, soil mixing, and intermediate application.
この発明の植物の活性促進剤を水稲について施用した所、次の結果を得た。 When the plant activity promoter of this invention was applied to paddy rice, the following results were obtained.
実施場所 北海道札幌市清田(佐々木農園)
北海道夕張郡田仁町(樋山農園)
北海道雨竜郡北竜町(高橋農園)
実施期間 平成11年5月6日〜同年9月16日
実施条件 品種名 ほしのゆめ、きらら397
アグロミックSK−10(トリコデルマ ハルジアナム SK−5−5菌の商標)散布量
A… 50g/m2、分生子2×108/g
B…100g/m2、分生子2×108/g
対照区には、タチガレンエース液を散布し、追肥した(従来法)以外は同一である。
Location: Kiyota (Sasaki Farm), Sapporo, Hokkaido
Tani-cho, Yubari-gun, Hokkaido (Kashiyama Farm)
Hokuryu-cho, Uryu-gun, Hokkaido (Takahashi Farm)
Implementation period May 6, 1999-September 16, 1999 Implementation conditions Variety name Hoshi no Yume, Kirara 397
Agromic SK-10 (Tricoderma harzianum SK-5-5 trademark)
A ... 50 g / m 2 ,
B ... 100 g / m 2 ,
The control group is the same except that the Tachigalen ace solution was sprayed and topdressed (conventional method).
前記の条件で、水稲栽培した所、表8(佐々木農園)、表9(樋山農園)、表10(高橋農園)の結果を得た。 Under the above-mentioned conditions, the results of paddy rice cultivation, Table 8 (Sasaki Farm), Table 9 (Kashiyama Farm), and Table 10 (Takahashi Farm) were obtained.
前記実施例によれば、きらら397の場合に、蛋白質で、慣行区に比べて4〜6ポイント下がり、アミロースで1〜3ポイントの差が確認された(1ポイントは0.5とされる)。 According to the above example, in the case of Kirara 397, the protein was 4 to 6 points lower than the conventional section, and the difference of 1 to 3 points was confirmed for amylose (1 point is 0.5). .
そこで味度を測定した所、平均91.0であった。一方市販の味が良いと言われる米について測定した所、88.0が最高であったから、現時点で最高の味度を示したものということができる。 Therefore, when the taste was measured, the average was 91.0. On the other hand, when it was measured for rice that is said to have a good commercial taste, 88.0 was the highest, so it can be said that it showed the highest taste at the present time.
前記結果より判断するに、水稲の活性化により、病原菌、害虫を忌避した低農薬有機農法であり、植物本来の力を引出して味覚を改善するのみならず収穫量も増加(20〜50%)するなどの特徴が確認された。 Judging from the above results, it is a low pesticide organic farming method that repels pathogens and pests by activating paddy rice, and it not only improves the taste by drawing out the original power of plants but also increases the yield (20-50%) The characteristics such as being confirmed were confirmed.
この発明の植物の活性促進剤をトウモロコシについて施用した所、次の結果を得た。 When the plant activity promoter of this invention was applied to corn, the following results were obtained.
実施場所 明治製菓株式会社生物科学研究所
実施期間 平成11年4月〜同年8月30日
使用菌:アグロミックSK−10(2×107/g)
施用法:発芽第2週目に、土壌散布、潅水150ml/pot
施用説明 前記条件のもとに、表11の施用設計に基づき施用した。
Place of implementation Meiji Seika Co., Ltd. Biological Sciences Laboratory Period of implementation April 1999-August 30, 2005 Bacteria used: Agromic SK-10 (2 × 10 7 / g)
Application method: Soil application, irrigation 150ml / pot in the second week of germination
Application description Based on the above conditions, application was performed based on the application design of Table 11.
前記により栽培し、収穫した所、表12の結果を得た。 The place cultivated and harvested as described above, the results shown in Table 12 were obtained.
前記のように、果実の肥大効果と、糖度の向上が認められた。施用量の多少による有為差は殆ど認められなかった。即ち20g/m2の施用量と、200g/m2の施用量との間に差がないということになり、トウモロコシの場合には、発芽後1回の施用で活性付与が行われることにより収穫まで継続するものと推定される。 As described above, fruit enlargement effect and improved sugar content were observed. There was almost no significant difference depending on the amount of application. That is, there is no difference between the application rate of 20 g / m 2 and the application rate of 200 g / m 2 , and in the case of corn, harvesting is performed by applying the activity once after germination. It is estimated that it will continue until.
従ってアグロミックSK−10により、生成される新アミノ酸の量が活性を付与するのに十分であるならば、施用量は20g/m2以下でも十分効力を発揮し得ることになる。 Therefore, if the amount of the new amino acid produced by the agromic SK-10 is sufficient to confer activity, the application rate can be sufficiently effective even at a dose of 20 g / m 2 or less.
前記理由により、土壌がアグロミックSK−10の増殖に必要な条件を有する場合には、20g/m2以下でも十分効力を有するものと認められる。恐らく増殖可能な条件の下限によって定まるであろう。そこで発芽後一定期間、アグロミックSK−10の増殖条件のもとにトウモロコシを育成する方法も考えられる。 For the above reasons, if the soil has the conditions necessary for the growth of Agromic SK-10, it is recognized that even if it is 20 g / m 2 or less, it is sufficiently effective. Perhaps determined by the lower limit of conditions that allow growth. Therefore, a method of growing corn under the growth conditions of agromic SK-10 for a certain period after germination can be considered.
アグロミックSK−10(トリコデルマ ハルジアナム SK−5−5の商標)をたまねぎ育苗床土に施用し、その苗及び定植後の生育促進効果を以下のように調査した。 Agromic SK-10 (Tricoderma harzianum SK-5-5 trademark) was applied to an onion nursery bed soil, and the growth promotion effect after the seedling and planting was investigated as follows.
(1) 試験材料:
(a) 供試種子:スーパーハイゴールドの薬剤処理していない種子
(b) 供試資材:アグロミックSK−10(Trichoderma harzianum SK−5−5の5×108CFUの生菌剤、北海道グリーン興産株式会社提供)
(c) 供試場所:千葉県木更津市下郡今間
(2) 試験方法:
(a) 苗床の準備:育苗床は千葉県下の山砂土で、前作はスイートコーンを栽培した圃場を用いた。pHは6.0前後の弱酸性土壌であったので、pHの調整を兼ねて消石灰1m2あたり50g、完熟堆肥(牛糞主体のモミガラ入り堆肥)1kg、CDU20gをアグロミックSK−10散布施用15日前に施肥し、耕耘した。
(1) Test material:
(a) Test seeds: Super High Gold seeds not treated with chemicals
(b) Materials to be tested: Agromic SK-10 (Trichoderma harzianum SK-5-5 5 × 10 8 CFU viable agent, provided by Hokkaido Green Kosan Co., Ltd.)
(c) Test location: Imuma, Shimo-gun, Kisarazu City, Chiba Prefecture
(2) Test method:
(a) Preparation of nursery bed: The nursery bed was mountain sand soil under Chiba Prefecture, and the previous crop was a field where sweet corn was grown. Since the pH was weakly acidic soil of around 6.0, 50 g per 1 m 2 of slaked lime, 1 kg of fully-ripened compost (compost containing chives), and 20 g of CDU were applied 15 days before application of Agromic SK-10. Fertilized and plowed.
(b) アグロミックSK−10施用および方法:
施用日:平成11年9月11日
準備した育苗床に、供試資材アグロミックSK−10粒剤を播種4日前に1m2あたり50g散布し、表層3−5cmの深さによく混和し、水道水を用いて、土壌水分が多湿にならないように(手で握りしめて団子になる程度の湿り程度、即ち糸状菌を増殖培養のための最適湿度条件)に留意して散水を行なった。
(b) Agromic SK-10 application and method:
Application Date: March 11 September 11 the prepared nursery floor, test records and material Agromic SK-10 Granules to 50g sprayed per 1m 2 in sowing four days ago, and mix well to the depth of the surface layer 3-5cm, water supply Water was used for watering so that the soil moisture did not become humid (weighed by hand to form dumplings, that is, optimum humidity conditions for growing fungi).
(c) 播種およびアグロミックSK−10の施用後の管理:
播種日:平成11年9月15日
残暑の厳しい、高温・乾燥であるので、トリコデルマ菌が育苗床土に増殖・定着を良くし、たまねぎの出芽を良くするために、できるだけ地温を20℃〜25℃程度に保ち、且つ水分条件の安定を図るように、両区ともライ麦ワラを2〜3cmの厚さに敷き、その上に寒冷紗をかけた。
(c) Management after sowing and application of Agromic SK-10:
Sowing date: September 15, 1999 Due to the high heat and dryness of the remaining heat, the soil temperature should be as low as 20 ° C in order to improve the growth and colonization of the Trichoderma fungus in the nursery bed soil and to improve the germination of the onion. In order to keep the temperature at about 25 ° C. and to stabilize the moisture condition, rye straw was laid in a thickness of 2 to 3 cm in both sections, and a cold koji was applied thereon.
たまねぎの種子は、すじまきで50cm間に70粒程度を播種し、覆土後、適度の散水を行なって、出芽が始まるまで、上記方法で被覆した。 About 70 seeds of onion seeds were sown in 50 cm between 50 cm, and after covering the soil, moderate watering was performed, and the seeds were covered by the above method until budding started.
出芽後は敷きワラを除去し、寒冷紗トンネル被覆を1ヶ月程度行なった。定植に備えるために、前記トンネル被覆を取り除き、堆肥と混合したCDU化成肥料を1m2あたり20g程度、条間に浅く中耕して施肥した。苗の大きさは径0.5〜0.6cm×葉丈5〜6cm程度の充熟した苗を作ることを目標にして育苗した。 After emergence, the straw was removed and the cold-cold tunnel was covered for about one month. In order to prepare for the fixed planting, the tunnel covering was removed, and the CDU chemical fertilizer mixed with compost was fertilized by shallowly plowing about 20 g per m 2 between the strips. The seedlings were grown for the purpose of producing mature seedlings having a diameter of 0.5 to 0.6 cm and a leaf length of about 5 to 6 cm.
(d) 定植:
定植日:平成11年11月7日(播種後53日目)
本圃の土壌は沖積土、pHは6.0程度の弱酸性土壌で、さつまいもの後作。消石灰10aあたり60kg、完熟堆肥500kg、骨粉60kg、米粕60kgを堆肥とよく混和して施用し、耕耘した。一週間後に畦幅1m・3条植の畦を作り、CDU20kgと過燐酸石灰10kgを完熟堆肥300kgによく混和して、溝施肥した。苗は大きさの順に株間15cmに施肥溝に定植した。
(d) Planting:
Planting date: November 7, 1999 (53 days after sowing)
The soil in this farm is alluvial soil, pH is weakly acidic soil of about 6.0, and the subsequent crop of sweet potatoes. 60 kg per slaked lime, 500 kg of fully-ripened compost, 60 kg of bone meal, and 60 kg of rice bran were mixed well with compost and applied. One week later, a cocoon with a ridge width of 1 m and a three-row planting was made, and 20 kg of CDU and 10 kg of superphosphate were mixed well with 300 kg of fully-ripened compost, and fertilized with a groove. The seedlings were planted in the fertilization groove at 15 cm between the plants in order of size.
(3) 調査結果:
(a) 出芽および生育状況:出芽では無処理区、処理区とも、ほとんど差は無かったが、一週間後にわずかながら無処理区で苗立ち枯れが発生した。その後の生育では、25日目ごろまで差は認められなかったが、トンネル除去・追肥後(播種後35日目)より、処理区で葉色が濃くなり、目視でも生育が旺盛となり、差が認められるようになった。
(3) Survey results:
(a) Emergence and growth status: There was almost no difference in emergence between the untreated and treated groups, but seedling wilt occurred in the untreated group slightly after one week. In the subsequent growth, there was no difference until around day 25, but after removal of the tunnel and after fertilization (35 days after sowing), the leaf color became darker in the treated area, and the growth became vigorous by visual observation. It came to be able to.
(b) 苗の生育調査:
(イ) 定植前の苗の生育状況:調査日平成11年11月7日(播種後53日目)詳細なデータは表13に示した。
(b) Seedling growth survey:
(I) Growth of seedlings before planting: Survey date November 7, 1999 (53 days after sowing) Detailed data are shown in Table 13.
良苗(大および中苗本数)は処理区78%で、無処理区では65%であった。アグロミックSK−10の施用によって良苗比率が13%増加した。苗径が0.7mm以上の苗はなかった。促進効果について、表13で示すとおり、アグロミックSK−10処理区では生育指数が大きくなり、生育が旺盛で、根量も多くなった。生育促進率を比較すると、SK−10処理区は、無処理区より27%の生育増加が認められた。 The good seedlings (large and medium seedlings) were 78% in the treated group and 65% in the untreated group. The application of Agromic SK-10 increased the percentage of good seedlings by 13%. There were no seedlings having a seedling diameter of 0.7 mm or more. Regarding the promoting effect, as shown in Table 13, the growth index increased, the growth was vigorous, and the root mass increased in the agromic SK-10 treated section. When the growth promotion rate was compared, a 27% increase in growth was observed in the SK-10 treated group than in the untreated group.
(ロ) 定植後の生育調査:越冬前の生育状況について調査した。 (B) Growth investigation after planting: The growth situation before wintering was investigated.
調査方法:図34の方法で調査した。 Investigation method: Investigation was carried out by the method shown in FIG.
結果:詳細な結果は表14のとおりである。 Results: Detailed results are shown in Table 14.
アグロミックSK−10処理区は、無処理区と比較して、出葉枚数で0.4枚多く、生育促進率は152.5%、バルブ径154.7%の増加率となった。目視による観察でも、明らかに生育促進効果が認められた。アグロミックSK−10処理区は、無処理区と比較して、根量が多く、根張りも良く、根に良く土が付着し、根毛も多かった。またアグロミックSK−10処理区において、葉面積とバルブ径は相関関係が認められた(図35、表15、表16)。 In the agromic SK-10 treated group, the number of leaves emerged 0.4 more than the untreated group, the growth promotion rate was 152.5%, and the bulb diameter was 154.7%. A growth promoting effect was clearly observed by visual observation. The agromic SK-10 treatment group had a larger amount of roots, better rooting, better soil adherence to the roots, and more root hairs than the non-treatment group. Further, in the agromic SK-10 treatment group, there was a correlation between the leaf area and the bulb diameter (FIG. 35, Table 15, Table 16).
アグロミックSK−10をキャベツ育苗床土に施用し、その苗及び定植後の生育促進効果を以下のように調査した。 Agromic SK-10 was applied to cabbage nursery bed soil, and the growth promotion effect after the seedling and planting was investigated as follows.
(1) 使用材料:
(a) 供試種子:薬剤処理していない種子
(b) 供試資材:アグロミックSK−10(トリコデルマ ハルジアナム SK−5−5の5×108CFU、北海道グリーン興産株式会社提供)
(2) 実施場所:千葉県成田市吉田農園
(3) 施用概要:
(a)施用日:平成11年9月15日
(b) 播種日:平成11年9月19日
(c) 床 土:pH6.0前後の砂壌土を中性に調整する為、消石灰を1m2あたり50g位散布した。これに完熟堆肥を1m2当り1kg、2週間前に施肥し、耕耘した。
(1) Materials used:
(a) Test seeds: seeds not treated with chemicals
(b) Test material: Agromic SK-10 (Trichoderma Halzianam SK-5-5 5 × 10 8 CFU, provided by Hokkaido Green Kosan Co., Ltd.)
(2) Location: Yoshida Farm, Narita City, Chiba Prefecture
(3) Application overview:
(a) Application date: September 15, 1999
(b) Sowing date: September 19, 1999
(c) Floor soil: In order to adjust the sandy loam soil of pH around 6.0 to neutral, about 50 g of slaked lime was sprayed per 1 m 2 . This mature compost and fertilizer to 1m 2 per 1kg, 2 weeks ago, and tillage.
播種前にアグロミックSK−10を、1m2あたり50g位散布した。ついで表層5cm位を土壌混和させ、1m2あたり3リットル位散水したが、土壌水分は多湿にならない程度とした(手で握りしめて団子になる程度)。 Before seeding, about 50 g of agromic SK-10 was sprayed per 1 m 2 . Next, about 5 cm of the surface layer was mixed with the soil and sprinkled with about 3 liters per 1 m 2 , but the soil moisture did not become so humid (to the extent that it became a dumpling when grasped by hand).
(4) 調査日:平成11年12月23日(播種後94日)
前記調査日において、処理区は葉色濃く、厚葉でしまりがよく、結球性が速いことが認められた。
(4) Survey date: December 23, 1999 (94 days after sowing)
On the day of the survey, it was confirmed that the treated area was dark in leaf color, thick and tight, and fast in heading.
Claims (7)
A成分:Ac-Aib-Ala-Aib-Aib-Aib-Aib-Gln-Aib-Aib-...であって分子量192
B成分:Ac-Aib-Ala-Aib-Aib-Val-Aib-Gln-Aib-Aib-...であって分子量206
C成分:
...であって分子量168
D成分:Ac-Aib-Ala-Aib-Aib-Aib-...であって分子量154又は220 Plant activity characterized by inoculating Trichoderma harzianum SK-55 in a solid medium, standing at 25 to 30 ° C. for 7 to 15 days, and then extracting the product to obtain the following substance: A method for producing an imparting agent.
A component: Ac-Aib-Ala-Aib-Aib-Aib-Aib-Gln-Aib-Aib-. . . With a molecular weight of 192
B component: Ac-Aib-Ala-Aib-Aib-Val-Aib-Gln-Aib-Aib-. . . With a molecular weight of 206
C component:
. . . With a molecular weight of 168
D component: Ac-Aib-Ala-Aib-Aib-Aib-. . . And having a molecular weight of 154 or 220
A成分:Ac-Aib-Ala-Aib-Aib-Aib-Aib-Gln-Aib-Aib-...であって分子量192
B成分:Ac-Aib-Ala-Aib-Aib-Val-Aib-Gln-Aib-Aib-...であって分子量206
C成分:
...であって分子量168
D成分:Ac-Aib-Ala-Aib-Aib-Aib-...であって分子量154又は220 Trichoderma harzianum SK-55 fungus is inoculated in a liquid medium, shaken at 25 ° C. to 30 ° C. for 4 days to 10 days, and then the product is extracted to obtain the following substance. Manufacturing method.
A component: Ac-Aib-Ala-Aib-Aib-Aib-Aib-Gln-Aib-Aib-. . . With a molecular weight of 192
B component: Ac-Aib-Ala-Aib-Aib-Val-Aib-Gln-Aib-Aib-. . . With a molecular weight of 206
C component:
. . . With a molecular weight of 168
D component: Ac-Aib-Ala-Aib-Aib-Aib-. . . And having a molecular weight of 154 or 220
A成分:Ac-Aib-Ala-Aib-Aib-Aib-Aib-Gln-Aib-Aib-...であって分子量192
B成分:Ac-Aib-Ala-Aib-Aib-Val-Aib-Gln-Aib-Aib-...であって分子量206
C成分:
...であって分子量168
D成分:Ac-Aib-Ala-Aib-Aib-Aib-...であって分子量154又は220 In plant cultivation, the conidia of Trichoderma harzianum SK-55 is attached to the seeds of the plant, the cover soil and the conidia are mixed, or the appropriate amount of the conidia is sprayed, irrigated or embedded, A method for imparting plant activity, characterized by producing the following substances:
A component: Ac-Aib-Ala-Aib-Aib-Aib-Aib-Gln-Aib-Aib-. . . With a molecular weight of 192
B component: Ac-Aib-Ala-Aib-Aib-Val-Aib-Gln-Aib-Aib-. . . With a molecular weight of 206
C component:
. . . With a molecular weight of 168
D component: Ac-Aib-Ala-Aib-Aib-Aib-. . . And having a molecular weight of 154 or 220
A成分:Ac-Aib-Ala-Aib-Aib-Aib-Aib-Gln-Aib-Aib-...であって分子量192
B成分:Ac-Aib-Ala-Aib-Aib-Val-Aib-Gln-Aib-Aib-...であって分子量206
C成分:
...であって分子量168
D成分:Ac-Aib-Ala-Aib-Aib-Aib-...であって分子量154又は220 In plant cultivation, the conidia of Trichoderma harzianum SK-55 is attached to the seeds of the plant, the cover soil and the conidia are mixed, or the appropriate amount of the conidia is sprayed, irrigated or embedded, The following substances are produced, the temperature of the soil where the conidia are placed is 15 ° C. to 30 ° C., the moisture is 30% or more, and air permeability or aeration that can grow on the soil is imparted. A method for imparting plant activity.
A component: Ac-Aib-Ala-Aib-Aib-Aib-Aib-Gln-Aib-Aib-. . . With a molecular weight of 192
B component: Ac-Aib-Ala-Aib-Aib-Val-Aib-Gln-Aib-Aib-. . . With a molecular weight of 206
C component:
. . . With a molecular weight of 168
D component: Ac-Aib-Ala-Aib-Aib-Aib-. . . And having a molecular weight of 154 or 220
A成分:Ac-Aib-Ala-Aib-Aib-Aib-Aib-Gln-Aib-Aib-...であって分子量192
B成分:Ac-Aib-Ala-Aib-Aib-Val-Aib-Gln-Aib-Aib-...であって分子量206
C成分:
...であって分子量168
D成分:Ac-Aib-Ala-Aib-Aib-Aib-...であって分子量154又は220 After cultivating conidia of Trichoderma harzianum SK-55 to produce the following activity-imparting agent, it is separated into an appropriate amount together with a medium, and mixed with a bulking material composed of sterile porous particles and nutrients A plant activity promoter characterized by being packaged for a predetermined amount.
A component: Ac-Aib-Ala-Aib-Aib-Aib-Aib-Gln-Aib-Aib-. . . With a molecular weight of 192
B component: Ac-Aib-Ala-Aib-Aib-Val-Aib-Gln-Aib-Aib-. . . With a molecular weight of 206
C component:
. . . With a molecular weight of 168
D component: Ac-Aib-Ala-Aib-Aib-Aib-. . . And having a molecular weight of 154 or 220
A成分:Ac-Aib-Ala-Aib-Aib-Aib-Aib-Gln-Aib-Aib-...であって分子量192
B成分:Ac-Aib-Ala-Aib-Aib-Val-Aib-Gln-Aib-Aib-...であって分子量206
C成分:
...であって分子量168
D成分:Ac-Aib-Ala-Aib-Aib-Aib-...であって分子量154又は220 A plant activity promoter characterized in that conidia of Trichoderma harzianum SK-55 are grown in a liquid medium to produce the following activity-imparting agent and then packaged to a predetermined amount.
A component: Ac-Aib-Ala-Aib-Aib-Aib-Aib-Gln-Aib-Aib-. . . With a molecular weight of 192
B component: Ac-Aib-Ala-Aib-Aib-Val-Aib-Gln-Aib-Aib-. . . With a molecular weight of 206
C component:
. . . With a molecular weight of 168
D component: Ac-Aib-Ala-Aib-Aib-Aib-. . . And having a molecular weight of 154 or 220
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2009174204A JP2010000078A (en) | 1999-04-08 | 2009-07-27 | Process for producing plant activator, activation method, activity promoter and method for applying the promoter |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP10139899 | 1999-04-08 | ||
| JP28473499 | 1999-10-05 | ||
| JP2009174204A JP2010000078A (en) | 1999-04-08 | 2009-07-27 | Process for producing plant activator, activation method, activity promoter and method for applying the promoter |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2000015680A Division JP2001172112A (en) | 1999-04-08 | 2000-01-25 | Activator for plant, method for producing the activator, method for imparting activity, activity promoter and method for applying the promoter |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| JP2010000078A true JP2010000078A (en) | 2010-01-07 |
Family
ID=41582316
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2009174204A Pending JP2010000078A (en) | 1999-04-08 | 2009-07-27 | Process for producing plant activator, activation method, activity promoter and method for applying the promoter |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP2010000078A (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2015030716A (en) * | 2013-08-06 | 2015-02-16 | 公立大学法人大阪府立大学 | Anti-pythium agent |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH06192028A (en) * | 1992-12-25 | 1994-07-12 | Hokkaido Green Kosan:Kk | Plant disease controlling bacterium, control agent using the same, method for producing control agent and method of using the same |
-
2009
- 2009-07-27 JP JP2009174204A patent/JP2010000078A/en active Pending
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH06192028A (en) * | 1992-12-25 | 1994-07-12 | Hokkaido Green Kosan:Kk | Plant disease controlling bacterium, control agent using the same, method for producing control agent and method of using the same |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2015030716A (en) * | 2013-08-06 | 2015-02-16 | 公立大学法人大阪府立大学 | Anti-pythium agent |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US12239132B2 (en) | Compounds and methods for increasing soil nutrient availability | |
| JP5714603B2 (en) | Novel fluorescent Pseudomonas species of the Pseudomonas azotoformans species for enhancing budding and growth of plants | |
| KR101199931B1 (en) | Promotion of nitrogen mineralization of organic fertilizers and control of plant diseases using bacillus velezensis KRICT934 | |
| CN107846838A (en) | Isolated complex endophyte compositions and methods for improved plant traits | |
| BG112709A (en) | BACTERIAL STRAIN BACILLUS AMYLOLIQUEFACIENS SUBSP. PLANTARUM BS89 AS A MEANS FOR INCREASING PLANT PRODUCTIVITY AND THEIR PROTECTION AGAINST DISEASES 1 claim | |
| TW201428097A (en) | Bacillus sp. strain with antifungal, antibacterial and growth promotion activity | |
| KR102604427B1 (en) | Composition for controlling plant diseases comprising Trichoderma longibrachiatum strain and method to control plant diseases | |
| US6753295B1 (en) | Plant activator, process for producing the same, activation method, activity promoter and method for applying the promoter | |
| EP2735607A1 (en) | Strain of Trichoderma harzianum and controlled release composition which contains said strain | |
| CN117757657B (en) | A strain of Methylobacterium tortuosus and its application in microbial agents | |
| KR20140071145A (en) | Novel Paenibacillus polymyxa AB-15 strain and use the same | |
| US11674118B2 (en) | PGPR compositions and methods for improved cultivation of tomato and potato species | |
| KR20230001353A (en) | Composition for controlling plant diseases comprising compound lsolated from Trichoderma longibrachiatum as an active ingredient and method of controlling plant diseases using the same | |
| KR102068090B1 (en) | Commposition for accelerating growth of plants using bacillus amyloliquefaciens and use thereof | |
| El Aymani et al. | on the Development of | |
| WO2025017538A1 (en) | Compositions comprising a mixture of lipo-chitooligosaccharide(s) and plant biostimulant(s), and uses thereof for increasing plant growth and crop yields | |
| Al-Masri et al. | Ethylene production by Sclerotinia sclerotiorum and influence of exogenously applied hormone and its inhibitor aminoethoxyvinylglycine on white mold | |
| JP2010000078A (en) | Process for producing plant activator, activation method, activity promoter and method for applying the promoter | |
| JP2007008960A (en) | Antibacterial agent and method for producing the same | |
| Jain et al. | Identification of etiological agent of Telya disease of Pomegranate, its pathogenesis and control using integrated management approach. Res J Biotechnol 18 | |
| KR102158226B1 (en) | Hypoxylon rubiginosum KDC0605 strain having antifungal activity against pathogens and plant growth promoting effect, and uses thereof | |
| WO2021133218A1 (en) | Means for stimulating crop growth | |
| KR100612041B1 (en) | Novel Streptomyces catensis VK-A60 strain having antibacterial activity against phytopathogens and fungicides produced from the strain | |
| RU2823059C1 (en) | Bacillus pumilus bis88 strain as universal agent for improving nutrition, accelerating growth and increasing productivity of agricultural grain, vegetable and industrial crops, as well as their protection against diseases and stress factors of chloride salinization | |
| JP2005289943A (en) | Agent and method for controlling phytophthora rot and downy mildew by using pseudomonas fluorescens |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20120110 |
|
| A521 | Written amendment |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20120308 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20120515 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20120918 |