JP2004331939A - Anticorrosion coating composition for gas transport steel pipe and gas transport steel pipe - Google Patents
Anticorrosion coating composition for gas transport steel pipe and gas transport steel pipe Download PDFInfo
- Publication number
- JP2004331939A JP2004331939A JP2003328353A JP2003328353A JP2004331939A JP 2004331939 A JP2004331939 A JP 2004331939A JP 2003328353 A JP2003328353 A JP 2003328353A JP 2003328353 A JP2003328353 A JP 2003328353A JP 2004331939 A JP2004331939 A JP 2004331939A
- Authority
- JP
- Japan
- Prior art keywords
- steel pipe
- anticorrosion
- pigment
- coating
- gas transport
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Landscapes
- Application Of Or Painting With Fluid Materials (AREA)
- Paints Or Removers (AREA)
- Protection Of Pipes Against Damage, Friction, And Corrosion (AREA)
- Rigid Pipes And Flexible Pipes (AREA)
Abstract
【課題】 ガス輸送鋼管用防食塗料組成物、およびこれらの塗料組成物から得られる塗膜を有するガス輸送鋼管を提供すること。
【解決手段】 エポキシ樹脂、硬化剤および防食顔料を含有する、ガス輸送鋼管用防食塗料組成物であって、この防食顔料が、(i)ホウ酸亜鉛化合物および、(ii)リンモリブデン酸アルミニウムおよび/またはトリポリリン酸二水素アルミニウム、を含有する、ガス輸送鋼管用防食塗料組成物。
【選択図】 なし
PROBLEM TO BE SOLVED: To provide an anticorrosion paint composition for a gas transport steel pipe and a gas transport steel pipe having a coating film obtained from the paint composition.
SOLUTION: The anticorrosion paint composition for a gas transport steel pipe, comprising an epoxy resin, a curing agent and an anticorrosion pigment, wherein the anticorrosion pigment comprises: (i) a zinc borate compound; and (ii) aluminum phosphomolybdate; And / or an aluminum dihydrogen tripolyphosphate anticorrosion coating composition for gas-transporting steel pipes.
[Selection diagram] None
Description
本発明は、ガス輸送鋼管用防食塗料組成物、特に天然ガスや都市ガスを輸送する鋼管の内面に塗装する防食塗料組成物に関する。 TECHNICAL FIELD The present invention relates to an anticorrosion coating composition for gas-transporting steel pipes, particularly to an anticorrosion coating composition applied to the inner surface of a steel pipe for transporting natural gas or city gas.
天然ガスや都市ガスを輸送する鋼管の内面には、防食塗膜が形成されている。ガス輸送管で輸送されるガス中には、種々の腐食性イオンが高圧で含まれておりそれらが鋼管に到達すると腐食が進行するので、それを防止するために鋼管の内面にエポキシ塗料を塗布している。しかし、腐食性イオンはエポキシ塗膜を透過して、鋼管表面に達して腐食を引き起こす。そのような腐食を防止するために、従来、エポキシ塗料中には鉛系やクロム系の防錆顔料を配合していた。 An anticorrosion coating is formed on the inner surface of a steel pipe that transports natural gas and city gas. The gas transported by the gas transport pipe contains various corrosive ions at high pressure, and when they reach the steel pipe, corrosion progresses. To prevent this, apply an epoxy paint to the inner surface of the steel pipe. are doing. However, corrosive ions permeate the epoxy coating and reach the surface of the steel pipe causing corrosion. In order to prevent such corrosion, a lead-based or chromium-based rust-preventive pigment has conventionally been compounded in an epoxy paint.
鉛系やクロム系の防錆顔料は、防食効果が高く、有効な防錆剤として利用されてきたが、それらが人体に有害であることが確認されてからはそれら防錆顔料に代わるものを開発する必要が生じてきた。 Lead-based and chromium-based rust preventive pigments have a high anticorrosion effect and have been used as effective rust preventive agents.However, after they have been confirmed to be harmful to the human body, alternatives to these rust preventive pigments have been developed. The need to develop has arisen.
特許文献1(特開平6−100802号公報)には、エポキシなどの熱硬化性樹脂中にホウ酸亜鉛化合物を配合した防食塗料組成物が提案されている。この防食塗料は、特に鋼材に電気防食処理をする場合に起こる陰極剥離を防止する機能は高いが、高圧ガス輸送菅内面の防食性能については、余り高い性能が得られない。 Patent Document 1 (JP-A-6-100802) proposes an anticorrosion coating composition in which a zinc borate compound is blended in a thermosetting resin such as epoxy. This anticorrosion paint has a high function of preventing cathodic peeling, which occurs particularly when a steel material is subjected to an electrolytic anticorrosion treatment, but does not provide very high anticorrosion performance on the inner surface of the high-pressure gas transport tube.
本発明は上記従来の問題を解決するものであり、その目的とするところは、ガス輸送鋼管用防食塗料組成物、特に天然ガスや都市ガスを輸送する鋼管の内面に塗装する防食塗料組成物であって、鉛およびクロムの両物質を含まない防食塗料組成物を提供することにある。 The present invention has been made to solve the above-mentioned conventional problems, and an object thereof is to provide an anticorrosion paint composition for gas-transporting steel pipes, particularly an anticorrosion paint composition for coating the inner surface of steel pipes that transport natural gas and city gas. Accordingly, it is an object of the present invention to provide an anticorrosion coating composition containing neither lead nor chromium.
本発明は、エポキシ樹脂、硬化剤および防食顔料を含有する、ガス輸送鋼管用防食塗料組成物であって、この防食顔料が、(i)ホウ酸亜鉛化合物および、(ii)リンモリブデン酸アルミニウムおよび/またはトリポリリン酸二水素アルミニウム、を含有する、ガス輸送鋼管用防食塗料組成物を提供するものであり、そのことにより上記目的が達成される。 The present invention is an anticorrosion paint composition for a gas transporting steel pipe, comprising an epoxy resin, a curing agent and an anticorrosion pigment, wherein the anticorrosion pigment comprises (i) a zinc borate compound and (ii) aluminum phosphomolybdate and And / or aluminum dihydrogen tripolyphosphate, which provides an anticorrosion coating composition for gas-transporting steel pipes, whereby the above object is achieved.
上記防食顔料を含む顔料成分が、乾燥塗膜中に含まれる顔料成分の体積百分率が15〜20%となる量で塗料組成物中に含有されるのが好ましい。 It is preferable that the pigment component containing the anticorrosive pigment is contained in the coating composition in such an amount that the volume percentage of the pigment component contained in the dried coating film is 15 to 20%.
また、上記防食顔料は、全顔料成分100重量部に対して5〜40重量部で含有されるのが好ましい。 The anticorrosion pigment is preferably contained in an amount of 5 to 40 parts by weight based on 100 parts by weight of all the pigment components.
本発明のガス輸送鋼管用防食塗料組成物において、防食顔料として(i)ホウ酸亜鉛化合物と(ii)リンモリブデン酸アルミニウムとを使用するのが好ましい。 In the anticorrosion coating composition for gas transport steel pipes of the present invention, it is preferable to use (i) a zinc borate compound and (ii) aluminum phosphomolybdate as anticorrosion pigments.
さらに本発明は、鋼管の内面に上記のガス輸送鋼管用防食塗料組成物からなる塗膜を有するガス輸送鋼管を提供するものである。 Further, the present invention provides a gas transporting steel pipe having a coating made of the above-described anticorrosive coating composition for a gas transporting steel pipe on the inner surface of the steel pipe.
また本発明は、上記ガス輸送鋼管の外面にさらにポリオレフィン被覆を有するガス輸送鋼管を提供するものである。 The present invention also provides a gas transporting steel pipe having a polyolefin coating on the outer surface of the gas transporting steel pipe.
本明細書中における「顔料成分」とは、着色顔料および体質顔料などの通常使用される顔料と、上記防食顔料とを含む成分をいう。 The “pigment component” in the present specification refers to a component containing a commonly used pigment such as a coloring pigment and an extender pigment and the above-described anticorrosion pigment.
なお本明細書中、乾燥塗膜中に含まれる顔料成分の体積百分率(%)は、「PVC」で表わすこともある。 In addition, in this specification, the volume percentage (%) of the pigment component contained in the dried coating film may be represented by "PVC".
本明細書中「鉛およびクロムの両物質を含まない」とは、防食塗料組成物中に、環境に悪影響を与えるような量で、鉛および鉛化合物が含まれず、かつクロムが含まれないことを意味する。 In the present specification, "not containing both lead and chromium" means that the anticorrosion coating composition does not contain lead and a lead compound and does not contain chromium in such an amount as to have an adverse effect on the environment. Means
本発明の塗料組成物は、鉛およびクロムの両物質を含まない塗料組成物であるにもかかわらず、鉛含有塗料組成物に匹敵する防食性を、ガス輸送鋼管の内面に付与することができる。従って、本発明によって、安全性の高い防食塗料組成物を提供することができる。また、本発明の塗料組成物を用いることにより、内面の防食性に優れたガス輸送鋼管を得ることができる。 Although the coating composition of the present invention is a coating composition that does not contain both lead and chromium substances, it can impart corrosion protection comparable to that of a lead-containing coating composition to the inner surface of a gas transport steel pipe. . Therefore, according to the present invention, a highly safe anticorrosive coating composition can be provided. Further, by using the coating composition of the present invention, a gas-transporting steel pipe having excellent corrosion resistance on the inner surface can be obtained.
本発明のガス輸送鋼管用防食塗料組成物は、エポキシ樹脂、硬化剤および防食顔料を含有する、ガス輸送鋼管用防食塗料組成物であって、この防食顔料が、(i)ホウ酸亜鉛化合物と、(ii)リンモリブデン酸アルミニウムおよび/またはトリポリリン酸二水素アルミニウムとを含むものである。 The gas transport steel pipe anticorrosion paint composition of the present invention is a gas transport steel pipe anticorrosion paint composition containing an epoxy resin, a curing agent and an anticorrosion pigment, wherein the anticorrosion pigment comprises (i) a zinc borate compound. , (Ii) aluminum phosphomolybdate and / or aluminum dihydrogen tripolyphosphate.
エポキシ樹脂
本発明に好適なエポキシ樹脂は、ビスフェノールA、ビスフェノールF、ビスフェノールADといった4,4−(ビスヒドロキシフェニル)アルカン類にエピハロヒドリンを作用させて製造される4,4−(ビスヒドロキシフェニル)アルカンのジグリシジルエーテル類である。また、4,4−(ビスヒドロキシフェニル)アルカンのグリシジルエーテルを主成分としてフェノールノボラック型エポキシ樹脂やクレゾールノボラック型エポキシ樹脂を併用してもよい。これら4,4−(ビスヒドロキシフェニル)アルカンの種類に応じて無溶剤型、溶剤型、粉体型などの塗料形態とし、作業性に応じて任意に選択して使用することができる。なお、この種のエポキシ樹脂は、商品名「エピコート」(油化シェルエポキシ(株)社製)、「エポトート」(東都化成(株)社製)、「アラルダイト」(チバガイギー社製)、「エピクロン」(大日本インキ化学工業(株)社製)、「ダウエポキシ」(旭ダウ(株)社製)として市販されている。
Epoxy resin Epoxy resins suitable for the present invention are 4,4- (bishydroxyphenyl) alkanes produced by reacting 4,4- (bishydroxyphenyl) alkanes such as bisphenol A, bisphenol F and bisphenol AD with epihalohydrin. Diglycidyl ethers. Moreover, a phenol novolak type epoxy resin or a cresol novolak type epoxy resin may be used in combination with glycidyl ether of 4,4- (bishydroxyphenyl) alkane as a main component. Depending on the type of the 4,4- (bishydroxyphenyl) alkane, a paint form such as a non-solvent type, a solvent type or a powder type can be used, and can be arbitrarily selected and used according to workability. Epoxy resins of this type include “Epicoat” (product name of Yuka Shell Epoxy Co., Ltd.), “Epototo” (product of Toto Kasei Co., Ltd.), “Araldite” (product of Ciba Geigy), and “Epiclon”. (Manufactured by Dainippon Ink and Chemicals, Inc.) and "Dow Epoxy" (manufactured by Asahi Dow Co., Ltd.).
硬化剤
硬化剤は、エポキシ樹脂のエポキシ基に対して付加重合性の官能基を分子内に複数個有する一般のエポキシ硬化剤を用いることができる。使用できる硬化剤として、脂肪族ジアミン、芳香族ジアミン、複素環式ジアミンなどのジアミン類、ポリアミン類およびそれらの各種変性物、これらのジアミン類およびポリアミン類等と脂肪酸およびその2量体とを反応させたポリアミドアミン樹脂、酸無水物、チオール類、フェノール類等が挙げられる。本発明では、好ましくはポリアミドアミン樹脂を使用する。使用できる硬化剤として、例えば「エポメート」(油化シェルエポキシ(株)社製、各種複素環式ジアミン変性物)、「サンマイド」(三和化学工業(株)社製、各種アミンアダクト、ポリアミドアミン樹脂など)、「トーマイド」(富士化成(株)社製、各種ポリアミドアミン)、「エピキュア」(油化シェルエポキシ(株)社製、各種アミンアダクト、チオール類、フェノール類)、「リカシッド」(新日本理科(株)社製、酸無水物)等が挙げられる。また、付加反応型ではなく、付加反応性とエポキシ基間の自己重合触媒能を有するジシアンジアミドやその誘導体、イミダール類を、硬化剤とすることもできる。これら硬化剤は、塗料の形態や硬化条件(常温硬化、加熱硬化)等に応じて適宜に選択使用される。
As the curing agent, a general epoxy curing agent having a plurality of functional groups capable of addition polymerization with respect to the epoxy group of the epoxy resin in a molecule can be used. As usable curing agents, diamines such as aliphatic diamines, aromatic diamines, and heterocyclic diamines, polyamines and various modified products thereof, and reaction of these diamines and polyamines with fatty acids and dimers thereof Polyamidoamine resins, acid anhydrides, thiols, phenols and the like. In the present invention, a polyamidoamine resin is preferably used. Examples of usable curing agents include “Epomate” (manufactured by Yuka Shell Epoxy Co., Ltd., various modified heterocyclic diamines) and “Sunmide” (manufactured by Sanwa Chemical Industry Co., Ltd., various amine adducts, polyamideamines) Resin, etc.), "Tomide" (manufactured by Fuji Kasei Co., Ltd., various polyamidoamines), "Epicure" (manufactured by Yuka Shell Epoxy Co., Ltd., various amine adducts, thiols, phenols), "Ricacid" ( Acid anhydride, manufactured by Shin Nippon Science Co., Ltd.). In addition, dicyandiamide having an addition reactivity and a self-catalytic activity between epoxy groups, a derivative thereof, and imidals can be used as the curing agent instead of the addition reaction type. These curing agents are appropriately selected and used according to the form of the coating material and the curing conditions (room temperature curing, heat curing).
本発明の塗料組成物において、エポキシ樹脂および硬化剤の含有量は、好ましくは、エポキシ樹脂のエポキシ当量と硬化剤の活性水素当量との比で1:0.5〜1:2.0となる量であり、より好ましくは1:1〜1:1.5となる量である。硬化剤が、上記当量比の範囲外の量で用いられる場合、硬化性が不十分となり、硬度が弱く、防食性も不十分となる。 In the coating composition of the present invention, the content of the epoxy resin and the curing agent is preferably 1: 0.5 to 1: 2.0 in a ratio of the epoxy equivalent of the epoxy resin to the active hydrogen equivalent of the curing agent. And more preferably in the range of 1: 1 to 1: 1.5. If the curing agent is used in an amount outside the range of the equivalent ratio, the curability will be insufficient, the hardness will be weak, and the corrosion protection will also be insufficient.
本発明の塗料組成物ではエポキシ樹脂および硬化剤を樹脂成分として含むが、必要に応じて、エポキシ樹脂および硬化剤以外の樹脂を樹脂成分として含んでもよい。このような樹脂として、例えばアクリル樹脂、ウレタン樹脂、オレフィン樹脂、アルキッド樹脂、フェノキシ樹脂、石油樹脂等が挙げられる。 Although the coating composition of the present invention contains an epoxy resin and a curing agent as resin components, it may contain a resin other than the epoxy resin and the curing agent as a resin component, if necessary. Examples of such a resin include an acrylic resin, a urethane resin, an olefin resin, an alkyd resin, a phenoxy resin, and a petroleum resin.
防食顔料
本発明の塗料組成物は、防食顔料として(i)ホウ酸亜鉛化合物と、(ii)リンモリブデン酸アルミニウムおよび/またはトリポリリン酸二水素アルミニウムとを含有する。(i)ホウ酸亜鉛化合物として、メタホウ酸亜鉛(Zn(BO2)2)、塩基性ホウ酸亜鉛(ZnB4O7・2ZnO)またはホウ酸亜鉛(2ZnO・3B2O3・3.5H2O)の1種もしくは2種以上の混合物を使用することができる。本発明では(i)ホウ酸亜鉛化合物としてホウ酸亜鉛を使用するのが好ましい。ホウ酸亜鉛は、酸化亜鉛とホウ酸の混合原料を溶融する方法や混合原料の水溶液を複分解する方法によって得ることができる。
Anticorrosion pigment The coating composition of the present invention contains (i) a zinc borate compound and (ii) aluminum phosphomolybdate and / or aluminum dihydrogen tripolyphosphate as anticorrosion pigments. (i) a zinc borate compound, zinc metaborate (Zn (BO 2) 2) , basic zinc borate (ZnB 4 O 7 · 2ZnO) or zinc borate (2ZnO · 3B 2 O 3 · 3.5H 2 One or a mixture of two or more of O) can be used. In the present invention, it is preferable to use zinc borate as the zinc borate compound (i). Zinc borate can be obtained by a method of melting a mixed raw material of zinc oxide and boric acid or a method of double-decomposing an aqueous solution of the mixed raw material.
防食顔料として、(i)ホウ酸亜鉛化合物に加えて、(ii)リンモリブデン酸アルミニウムおよび/またはトリポリリン酸二水素アルミニウムが含まれる。ホウ酸亜鉛化合物と、リンモリブデン酸アルミニウムおよび/またはトリポリリン酸二水素アルミニウムとを併用することにより、防食性に優れた、鉛およびクロムの両物質を含まない塗料組成物を得ることができる。防食顔料として(i)ホウ酸亜鉛化合物と(ii)リンモリブデン酸アルミニウムとが含まれるのが好ましい。良好な防食性を有するガス輸送鋼管用の塗料組成物を得ることができるからである。上記化合物は市販されているものを使用することができる。例えば、リンモリブデン酸アルミニウムとして「LFボウセイ」(キクチカラー(株)社製)が市販されており、これらを使用することができる。 Anticorrosion pigments include (ii) aluminum phosphomolybdate and / or aluminum dihydrogen tripolyphosphate in addition to (i) the zinc borate compound. By using a zinc borate compound in combination with aluminum phosphomolybdate and / or aluminum dihydrogen tripolyphosphate, it is possible to obtain a coating composition excellent in anticorrosion properties and free of both lead and chromium substances. The anticorrosion pigment preferably contains (i) a zinc borate compound and (ii) aluminum phosphomolybdate. This is because it is possible to obtain a coating composition for gas transport steel pipes having good corrosion resistance. The said compound can use a commercially available thing. For example, “LF BOSEI” (manufactured by Kikuchi Color Co., Ltd.) is commercially available as aluminum phosphomolybdate, and these can be used.
防食顔料は、本発明の塗料組成物中、全顔料成分100重量部に対して5〜40固形分重量部、好ましくは10〜20固形分重量部で含有される。防食顔料が5固形分重量部未満では防食効果が有効に発現せず、40固形分重量部を超えると溶出成分が多すぎて耐水性が低下することとなり実用に好ましくない。 The anticorrosion pigment is contained in the coating composition of the present invention in an amount of 5 to 40 solid parts by weight, preferably 10 to 20 solid parts by weight, based on 100 parts by weight of all the pigment components. If the amount of the anticorrosive pigment is less than 5 parts by weight, the anticorrosive effect is not effectively exhibited. If the amount is more than 40 parts by weight, the eluted components are too large and the water resistance is lowered, which is not preferable for practical use.
本発明の塗料組成物において、防食顔料に含まれる(i)ホウ酸亜鉛化合物と、(ii)リンモリブデン酸アルミニウムおよび/またはトリポリリン酸二水素アルミニウム、との重量比は、好ましくは、(i)1重量部に対して(ii)1.5〜2.0重量部である。(ii)が上記重量比1:1.5より少ない量で含まれると、防食効果が有効に発揮されず、(ii)が上記重量比1:2.0を超える量で含まれると、耐水性が低下することとなる。 In the coating composition of the present invention, the weight ratio of (i) the zinc borate compound contained in the anticorrosion pigment to (ii) aluminum phosphomolybdate and / or aluminum dihydrogen tripolyphosphate is preferably (i) (Ii) 1.5 to 2.0 parts by weight with respect to 1 part by weight. When (ii) is contained in an amount less than the above weight ratio of 1: 1.5, the anticorrosion effect is not effectively exhibited, and when (ii) is contained in an amount exceeding the above weight ratio of 1: 2.0, water resistance is reduced. Will be reduced.
顔料
本発明の塗料組成物には通常用いられる顔料を含有させてもよい。但し、本明細書でいう「顔料」には、前述の防食顔料は含まれない。使用し得る顔料の例としては、チタンホワイト、カーボンブラック及びベンガラのような着色顔料;カオリン、タルク、ケイ酸アルミニウム、炭酸カルシウム、シリカ、マイカおよびクレーのような体質顔料等が挙げられる。
Pigment The coating composition of the present invention may contain a commonly used pigment. However, the “pigment” referred to in the present specification does not include the aforementioned anticorrosion pigment. Examples of pigments that can be used include coloring pigments such as titanium white, carbon black, and red iron oxide; extender pigments such as kaolin, talc, aluminum silicate, calcium carbonate, silica, mica, and clay.
本発明の塗料組成物において、上記樹脂成分に対して、(i)および(ii)を含む防食顔料、ならびに上記顔料(着色顔料および体質顔料等)を含む成分を、顔料成分という。そして、本発明の塗料組成物では、この顔料成分が、本発明の塗料組成物より得られる乾燥塗膜中に含まれる顔料成分の体積百分率(PVC)で、好ましくは15〜20%となる量で塗料組成物中に含まれる。上記顔料成分がPVCで15%より少なくなる量で塗料組成物中に含まれると、得られる塗膜が脆弱となるため実用に好ましくなく、PVCで20%を超える量で塗料組成物中に含まれると、耐アルカリ性が低下する。 In the coating composition of the present invention, an anticorrosive pigment containing (i) and (ii) and a component containing the pigment (color pigment, extender pigment, and the like) with respect to the resin component are referred to as a pigment component. In the coating composition of the present invention, the amount of the pigment component is preferably 15 to 20% by volume percentage (PVC) of the pigment component contained in the dried coating film obtained from the coating composition of the present invention. In the coating composition. If the pigment component is contained in the coating composition in an amount of less than 15% by PVC, the resulting coating film becomes brittle, which is not preferable for practical use, and is included in the coating composition in an amount of more than 20% by PVC. , The alkali resistance decreases.
本発明の塗料組成物は、上記した樹脂成分、防食顔料、顔料のほか、必要に応じて有機溶剤、表面調整剤等の当分野において公知の他の成分を添加することができる。本発明の塗料組成物の固形分濃度は10〜100%で使用する。 The coating composition of the present invention may contain, if necessary, other components known in the art, such as an organic solvent and a surface conditioner, in addition to the above-mentioned resin component, anticorrosive pigment, and pigment. The coating composition of the present invention has a solid content of 10 to 100%.
本発明の塗料組成物は、通常の塗料組成物の製造で用いられる一般的な製造方法を用いて製造することができる。すなわち、本発明の塗料組成物を液状無溶剤型塗料とする場合には、エポキシ樹脂に所定量の硬化剤、防食顔料および顔料等を加え、ロールミル、ディゾルバー等で分散処理することにより製造することができる。本発明の塗料組成物を有機溶剤型塗料とする場合にはロールミル、ディゾルバー、SGミル、ポットミル等で分散処理することにより製造することができる。本発明の塗料組成物を粉体塗料とする場合には、エポキシ樹脂に所定量の硬化剤、防食顔料および顔料等を加え、予備混合したのち加熱混練し、冷却後に粉砕、分級することにより製造することができる。 The coating composition of the present invention can be manufactured by using a general manufacturing method used for manufacturing a usual coating composition. That is, when the coating composition of the present invention is used as a liquid solventless coating, a predetermined amount of a curing agent, an anticorrosive pigment, a pigment, and the like are added to an epoxy resin, and the epoxy resin is dispersed and processed using a roll mill, a dissolver, or the like. Can be. When the coating composition of the present invention is used as an organic solvent type coating, it can be produced by subjecting it to a dispersion treatment using a roll mill, dissolver, SG mill, pot mill or the like. When the coating composition of the present invention is used as a powder coating, a predetermined amount of a curing agent, an anticorrosive pigment and a pigment are added to an epoxy resin, premixed, heated and kneaded, cooled, then pulverized and classified. can do.
本発明による防食塗料組成物を塗布するには、刷毛、ローラー、エアレススプレー、エアスプレー、粉体塗装機などを塗料の形態に合わせて選定し、常法に従って塗装される。塗装後の塗膜面には、ポリエチレンライニング、重防食ウレタン被覆、エポキシ樹脂塗料などの重防食塗膜や着色、美装のための上塗り層を形成してもよい。 To apply the anticorrosion coating composition according to the present invention, a brush, a roller, an airless spray, an air spray, a powder coating machine or the like is selected according to the form of the coating, and the coating is applied according to a conventional method. On the coated film surface after coating, a heavy-duty corrosion-resistant coating film such as polyethylene lining, heavy-duty urethane coating, epoxy resin paint, or an overcoat layer for coloring and dressing may be formed.
ガス輸送鋼管
上記した本発明の塗料組成物をガス輸送鋼管の内面に塗装することにより、鋼管の内面に防食性が付与されたガス輸送鋼管を提供することができる。防食性が付与される鋼管を構成する鋼材として、例えば炭素鋼、またはステンレス鋼等の合金鋼など、ラインパイプ等での使用に有用な鋼材が挙げられる。ガス輸送鋼管は、本発明の塗料組成物を塗装する前に、必要に応じて、公知の表面処理方法(例えば、ショットブラスト処理、グリッドブラスト処理、サンドブラスト処理等の物理的手段、または酸洗、アルカリ脱脂等の化学的手段、あるいはそれらの組み合わせ)を用いて表面を清浄化されていることが好ましい。ガス輸送鋼管の表面に、防食性の観点から、一般に密着性や防食性を高めるための下地処理として公知の化成処理(例えば、リン酸塩処理、クロメート処理、クロム酸処理など)が施されていているものも好ましい。化成処理とは、化学的または電気化学的処理により、金属である鋼材表面に非金属の皮膜を沈積させる処理法をいう。このような皮膜を沈積させる化合物としては、例えばリン酸塩、クロム酸塩、塩基性塩、酸化物、硫化物などが挙げられる。
Gas transport steel pipe By coating the above-described coating composition of the present invention on the inner surface of the gas transport steel pipe, a gas transport steel pipe having the inner surface of the steel pipe provided with anticorrosion properties can be provided. Examples of the steel material constituting the steel pipe provided with anticorrosion properties include a steel material useful for use in a line pipe or the like, for example, carbon steel or alloy steel such as stainless steel. Before applying the coating composition of the present invention, the gas-transporting steel pipe may be subjected to a known surface treatment method (for example, physical means such as shot blasting, grid blasting, or sandblasting, or pickling, The surface is preferably cleaned using chemical means such as alkali degreasing or a combination thereof. From the viewpoint of corrosion resistance, the surface of the gas transporting steel pipe is generally subjected to a chemical conversion treatment (for example, a phosphate treatment, a chromate treatment, a chromic acid treatment, etc.) as a base treatment for improving adhesion and corrosion resistance. Are preferred. The chemical conversion treatment is a treatment method in which a nonmetallic film is deposited on the surface of a steel material as a metal by chemical or electrochemical treatment. Compounds that deposit such coatings include, for example, phosphates, chromates, basic salts, oxides, sulfides, and the like.
上記した本発明の塗料組成物をガス輸送鋼管の内面に塗装する方法として、鋼管の表面に、スプレー塗装またはしごき塗り等の通常使用される塗装方法を用いることができる。塗装後、加熱して乾燥および硬化させることにより防食塗膜を形成する。本発明の塗料組成物から得られる乾燥させた塗膜の膜厚は、好ましくは10μm〜100μm、より好ましくは30〜100μmである。膜厚が10μm未満では、剥離が生じやすく、防食機能が十分に発現できない。また、膜厚が100μmを超えると衝撃耐性が低下することがある。 As a method for applying the above-described coating composition of the present invention to the inner surface of a gas transporting steel pipe, a commonly used coating method such as spray coating or ironing can be used on the surface of the steel pipe. After coating, the coating is heated and dried and cured to form an anticorrosion coating. The thickness of the dried coating film obtained from the coating composition of the present invention is preferably from 10 μm to 100 μm, more preferably from 30 to 100 μm. If the film thickness is less than 10 μm, peeling is likely to occur, and the anticorrosion function cannot be sufficiently exhibited. On the other hand, when the thickness exceeds 100 μm, the impact resistance may decrease.
埋設などのパイプラインの敷設環境に応じて、鋼管の外面に防食性を付与することができる。鋼管の外面に防食性を付与する方法として、例えば鋼管の外面にポリオレフィン被覆を設ける方法、またはエポキシ樹脂塗装を行なう方法などが挙げられる。上記のポリオレフィン被覆は、鋼管の外面にポリオレフィンライニングを施すことにより設けることができる。このポリオレフィン被覆を形成するのに使用するポリオレフィン樹脂は、鋼材の被覆に一般に用いられている任意のポリオレフィンを使用することができる。例えば、低密度ポリエチレン、中密度ポリエチレン、高密度ポリエチレン等、およびエチレンと少量の他のオレフィンもしくはビニルモノマー(例えば、プロピレン、酢酸ビニル、アクリル酸エステル等)との共重合体が挙げられる。これらは単独でまたは混合して用いてよい。前記被覆用ポリオレフィン樹脂には、防食塗膜に通常使用される酸化防止剤、紫外線吸収剤、顔料、充填剤等を添加してもよい。特に、酸化防止剤や紫外線吸収剤をポリオレフィン樹脂に添加することにより、得られるポリオレフィン被覆の耐久性を向上させることができる。また、ポリオレフィン被覆に、当該分野において公知の着色顔料を好ましくは5重量部以下の量で添加することもできる。これらのポリオレフィン樹脂を用いて、公知の方法(例えば、溶融丸ダイ共押出法または溶融Tダイ押出法)により、厚さ1〜6mmのポリオレフィン被覆を、防食塗膜が形成されたガス輸送鋼管の外面上に形成することができる。 Corrosion protection can be imparted to the outer surface of the steel pipe according to the laying environment of the pipeline such as burial. As a method for imparting corrosion resistance to the outer surface of the steel pipe, for example, a method of providing a polyolefin coating on the outer surface of the steel pipe, a method of performing epoxy resin coating, and the like are given. The above-mentioned polyolefin coating can be provided by applying a polyolefin lining to the outer surface of the steel pipe. As the polyolefin resin used for forming the polyolefin coating, any polyolefin generally used for coating steel materials can be used. For example, low-density polyethylene, medium-density polyethylene, high-density polyethylene, and the like, and a copolymer of ethylene with a small amount of another olefin or vinyl monomer (for example, propylene, vinyl acetate, acrylate, and the like) can be mentioned. These may be used alone or as a mixture. The coating polyolefin resin may contain an antioxidant, an ultraviolet absorber, a pigment, a filler, and the like, which are usually used for an anticorrosion coating. In particular, by adding an antioxidant or an ultraviolet absorber to the polyolefin resin, the durability of the resulting polyolefin coating can be improved. Further, a coloring pigment known in the art can be added to the polyolefin coating, preferably in an amount of 5 parts by weight or less. Using these polyolefin resins, a polyolefin coating having a thickness of 1 to 6 mm is formed by a known method (for example, a co-extrusion method using a molten round die or an extrusion method using a molten T-die) to form a gas transport steel pipe having a corrosion-resistant coating film formed thereon. It can be formed on the outer surface.
鋼管の外面にエポキシ樹脂塗装を行なう場合は、通常用いられる液状エポキシ樹脂塗料または粉体エポキシ樹脂塗料などを用いることができる。前記ポリオレフィン被膜を鋼管の外面に設ける前に、鋼管の外面にエポキシ樹脂塗装を行なうことができる。また、鋼管の外面にこのようなエポキシ樹脂塗装のみを行なうこともできる。 When the outer surface of the steel pipe is coated with epoxy resin, a commonly used liquid epoxy resin paint or powder epoxy resin paint can be used. Before providing the polyolefin coating on the outer surface of the steel pipe, the outer surface of the steel pipe may be coated with an epoxy resin. Also, such an epoxy resin coating alone can be applied to the outer surface of the steel pipe.
また、前記ポリオレフィン被覆を設ける前に、必要に応じて変性ポリオレフィン接着樹脂層を設けることができる。この変性ポリオレフィン接着樹脂層を設けることにより、鋼管とポリオレフィン被覆との、または樹脂塗装の塗膜とポリオレフィン被膜との間の密着性を高めることができる。このような変性ポリオレフィン接着樹脂として、例えば、ポリオレフィン樹脂を、マレイン酸もしくはアクリル酸等の不飽和カルボン酸またはこれらの無水物で変性して接着性を付与した無水マレイン酸もしくはカルボン酸変性ポリオレフィン樹脂、またはエチレン/メタクリル酸共重合体が挙げられる。特に、無水マレイン酸変性ポリオレフィン樹脂は、高い接着性を示すことから、この目的を達成する観点から好ましい。このような無水マレイン酸変性ポリオレフィン樹脂として、例えば、三井化学製接着性ポリオレフィン、製品名「アドマー(ADMER)」などが使用できる。必要に応じて設けられる変性ポリオレフィン接着樹脂層の厚さは、一般に0.1〜0.5mmであるが、この範囲に特に限定されるものではない。 Before providing the polyolefin coating, a modified polyolefin adhesive resin layer may be provided as necessary. By providing this modified polyolefin adhesive resin layer, the adhesion between the steel pipe and the polyolefin coating or between the resin-coated coating film and the polyolefin coating can be increased. As such a modified polyolefin adhesive resin, for example, a polyolefin resin, maleic anhydride or carboxylic acid-modified polyolefin resin modified with unsaturated carboxylic acid such as maleic acid or acrylic acid or an anhydride thereof to impart adhesiveness, Or an ethylene / methacrylic acid copolymer. In particular, a maleic anhydride-modified polyolefin resin is preferable from the viewpoint of achieving this object because it exhibits high adhesiveness. As such a maleic anhydride-modified polyolefin resin, for example, an adhesive polyolefin manufactured by Mitsui Chemicals, Inc., product name “ADMER” and the like can be used. The thickness of the modified polyolefin adhesive resin layer provided as needed is generally 0.1 to 0.5 mm, but is not particularly limited to this range.
本発明の防食性鋼管には、必要に応じて、上記の外面のポリオレフィン被覆上に、更に上塗り層を、当該分野において通常使用される方法により形成してもよい。このような上塗り層として、例えば粉体エポキシ塗装による塗膜が挙げられる。 In the anticorrosion steel pipe of the present invention, if necessary, an overcoat layer may be further formed on the above-mentioned polyolefin coating on the outer surface by a method generally used in the art. An example of such an overcoat layer is a coating film formed by powder epoxy coating.
以下に実施例を用いて本発明を説明するが、本発明は以下の実施例に限定されるものではない。ことわりのない限り各例中の「部」は「重量部」を表し、「%」は「重量%」を表す。 Hereinafter, the present invention will be described using examples, but the present invention is not limited to the following examples. Unless otherwise specified, “parts” in each example represents “parts by weight”, and “%” represents “% by weight”.
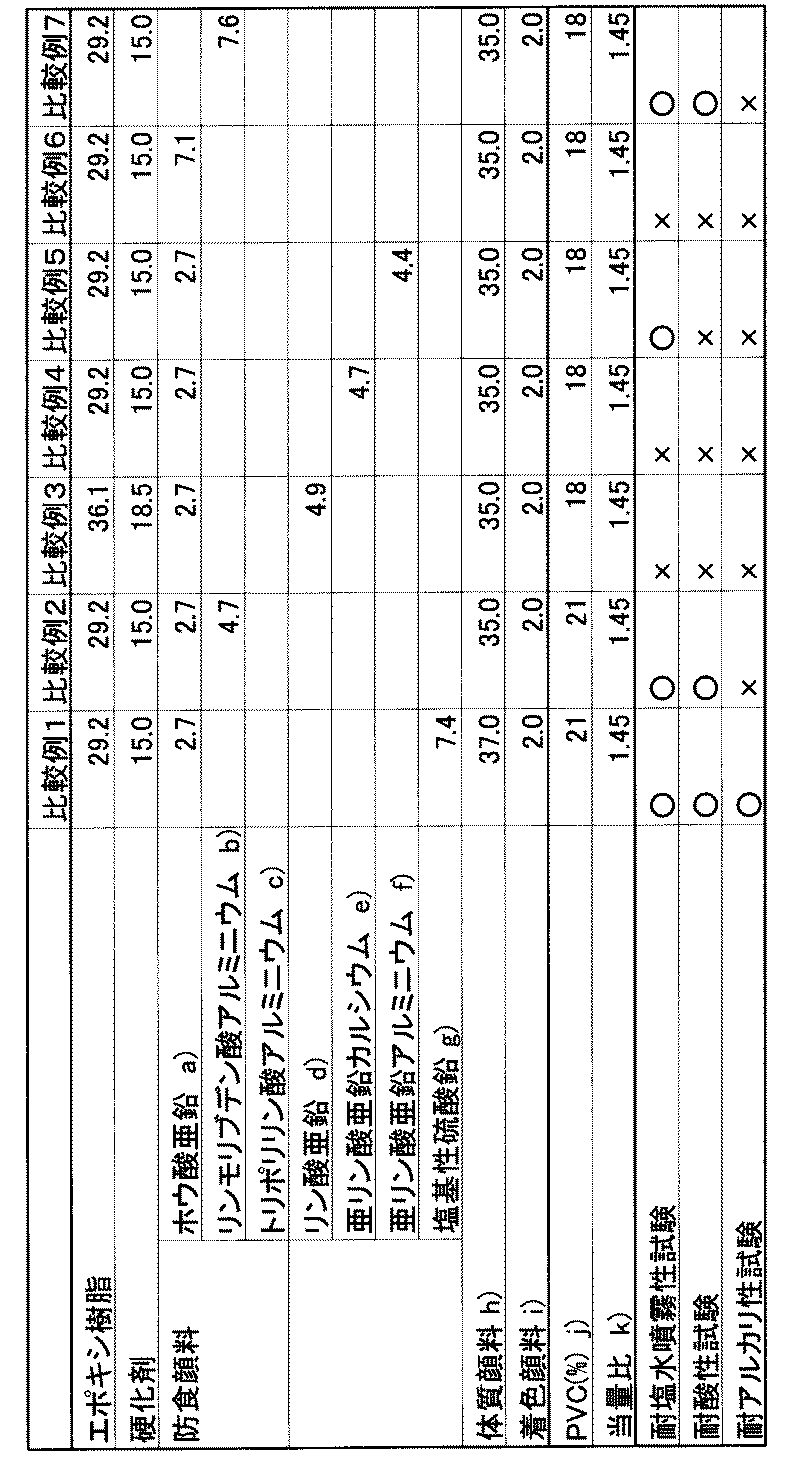
実施例1〜6、比較例1〜7
硬化剤(ポリアミドアミン樹脂、活性水素当量168:商品名サンマイド、三和化学工業(株)社製)、防食顔料および顔料それぞれを、表1、表2に記載の重量部で加えた。次いで、サンドグランドミルで1500rpm、20分間分散処理し、次いで400メッシュの金網で濾過した。得られた分散物に、エポキシ樹脂(ビスフェノールA型エポキシ樹脂、エポキシ当量475:商品名エポトートYD−011、東都化成(株)社製)を表1、表2に記載の重量部で加えて混合し、防食塗料組成物を調製した。
Examples 1 to 6, Comparative Examples 1 to 7
A curing agent (polyamide amine resin, active hydrogen equivalent 168: trade name Sunmide, manufactured by Sanwa Chemical Industry Co., Ltd.), an anticorrosive pigment and a pigment were added in parts by weight described in Tables 1 and 2. Next, the mixture was dispersed in a sand ground mill at 1500 rpm for 20 minutes, and then filtered through a 400-mesh wire net. To the obtained dispersion, an epoxy resin (bisphenol A type epoxy resin, epoxy equivalent: 475, trade name: Epototo YD-011, manufactured by Toto Kasei Co., Ltd.) is added in parts by weight described in Tables 1 and 2, and mixed. Then, an anticorrosion coating composition was prepared.
上記表1および表2中の各符号は、以下の意味を表わす。
a:ホウ酸亜鉛
b:リンモリブデン酸アルミニウム、LFボウセイ
(キクチカラー(株)社製)
c:トリポリリン酸アルミニウム、Kホワイト
(テイカ(株)社製)
d:リン酸亜鉛、LFボウセイ
(キクチカラー(株)社製)
e:亜リン酸亜鉛カルシウム、Expert
(東邦顔料(株)社製)
f:亜リン酸亜鉛アルミニウム、Expert
(東邦顔料(株)社製)
g:塩基性硫酸鉛、エンリュウN(キクチカラー(株)社製)
h:沈降性硫酸バリウム
i:三菱カーボンブラック、MA100(三菱化学(株)社製)
j:乾燥塗膜中に含まれる防食顔料および顔料の体積百分率
k:硬化剤の活性水素当量/エポキシ樹脂のエポキシ当量、を示す当量比
Each symbol in Tables 1 and 2 has the following meaning.
a: Zinc borate b: Aluminum phosphomolybdate, LF Bosei (manufactured by Kikuchi Color Co., Ltd.)
c: Aluminum tripolyphosphate, K white (manufactured by Teica Corporation)
d: Zinc phosphate, LF dye (Kikuchi Color Co., Ltd.)
e: zinc calcium phosphite, Expert
(Manufactured by Toho Pigment Co., Ltd.)
f: zinc aluminum phosphite, Expert
(Manufactured by Toho Pigment Co., Ltd.)
g: Basic lead sulfate, Enryu N (manufactured by Kikuchi Color Co., Ltd.)
h: precipitated barium sulfate i: Mitsubishi carbon black, MA100 (manufactured by Mitsubishi Chemical Corporation)
j: volume percentage of anticorrosive pigment and pigment contained in the dried coating film k: equivalent ratio indicating active hydrogen equivalent of curing agent / epoxy equivalent of epoxy resin
評価方法
試験材Aの作成
幅70mm、長さ150mm、厚さ1.6mmの鋼板(SS−400)をサンドブラストし、その後キシレンを用いて脱脂した。この鋼板に、実施例1〜6、比較例1〜7で得られた塗料組成物を、エアスプレーを用いて、乾燥膜厚が40〜60μmとなるように塗布した。50℃で1時間乾燥させた後、室温で7日間放置し、試験材Aとした。
Evaluation method
A steel sheet (SS-400) having a width of 70 mm, a length of 150 mm, and a thickness of 1.6 mm of the test material A was sandblasted, and then degreased using xylene. The coating compositions obtained in Examples 1 to 6 and Comparative Examples 1 to 7 were applied to this steel sheet using an air spray so that the dry film thickness became 40 to 60 µm. After drying at 50 ° C. for 1 hour, the resultant was left at room temperature for 7 days to obtain a test material A.
試験材Bの作成
幅70mm、長さ150mm、厚さ0.8mmの鋼板(JIS G3141)を#180の研磨紙で研磨し、その後キシレンを用いて脱脂した。この鋼板に、実施例1〜6、比較例1〜7で得られた塗料組成物を、エアスプレーを用いて、乾燥膜厚が40〜60μmとなるように塗布した。50℃で1時間乾燥させた後、室温で7日間放置し、試験材Bとした。
Preparation of Test Material B A steel plate (JIS G3141) having a width of 70 mm, a length of 150 mm, and a thickness of 0.8 mm was polished with # 180 abrasive paper, and then degreased using xylene. The coating compositions obtained in Examples 1 to 6 and Comparative Examples 1 to 7 were applied to this steel sheet using an air spray so that the dry film thickness became 40 to 60 µm. After drying at 50 ° C. for 1 hour, it was left at room temperature for 7 days to obtain a test material B.
耐塩水噴霧性試験
JIS K 5600 7−1に準拠して、耐塩水噴霧性試験を行なった。試験材A全体にクロスカットを入れて行なった。判定は以下の基準で行なった。
クロスカット部分から生じた錆または膨れの幅が3mm以内:○
クロスカット部分から生じた錆または膨れの幅が3mm以上:×
Salt water spray resistance test A salt water spray resistance test was performed in accordance with JIS K 5600 7-1. A cross cut was made on the entire test material A. The judgment was made based on the following criteria.
The width of the rust or blister generated from the cross cut portion is within 3 mm: ○
The width of the rust or blister generated from the cross cut portion is 3 mm or more: ×
耐酸性試験
JIS K 5600 6−1に準拠して、耐酸性試験を行なった。試験材Bを、5%塩酸水溶液中に23℃で30日間浸漬した。塗膜状態を目視評価した。判定は以下の基準で行なった。
錆、膨れ、剥がれ等が生じていない:○
錆、膨れ、剥がれ等が生じている :×
Acid resistance test An acid resistance test was performed according to JIS K 5600 6-1. Test material B was immersed in a 5% hydrochloric acid aqueous solution at 23 ° C. for 30 days. The state of the coating film was visually evaluated. The judgment was made based on the following criteria.
No rust, swelling, peeling, etc .: ○
Rust, swelling, peeling, etc .: ×
耐アルカリ性試験
JIS K 5600 6−1に準拠して、耐酸性試験を行なった。試験材Bを、5%水酸化ナトリウム水溶液中に23℃で30日間浸漬した。塗膜状態を目視評価した。判定は以下の基準で行なった。
錆、膨れ、剥がれ等が生じていない:○
錆、膨れ、剥がれ等が生じている :×
Alkali Resistance Test An acid resistance test was performed according to JIS K 5600 6-1. Test material B was immersed in a 5% aqueous sodium hydroxide solution at 23 ° C. for 30 days. The state of the coating film was visually evaluated. The judgment was made based on the following criteria.
No rust, swelling, peeling, etc .: ○
Rust, swelling, peeling, etc .: ×
上記試験の結果を表1、表2に示す。これらの結果より、本発明の塗料組成物は、鉛およびクロムの両物質を含まない塗料組成物であるにもかかわらず、鉛含有塗料組成物に匹敵する防食性を付与できることがわかる。
Tables 1 and 2 show the results of the above test. These results show that the coating composition of the present invention can impart corrosion protection comparable to that of a lead-containing coating composition, even though the coating composition does not contain both lead and chromium.
Claims (7)
該防食顔料が、(i)ホウ酸亜鉛化合物および、(ii)リンモリブデン酸アルミニウムおよび/またはトリポリリン酸二水素アルミニウム、を含有する、ガス輸送鋼管用防食塗料組成物。 An anticorrosion coating composition for a gas-transporting steel pipe, comprising an epoxy resin, a curing agent and an anticorrosion pigment,
An anticorrosion paint composition for gas transport steel pipes, wherein the anticorrosion pigment contains (i) a zinc borate compound and (ii) aluminum phosphomolybdate and / or aluminum dihydrogen tripolyphosphate.
The gas transport steel pipe according to claim 6, wherein the gas transport steel pipe further has a polyolefin coating on an outer surface of the steel pipe.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2003328353A JP4560284B2 (en) | 2003-04-15 | 2003-09-19 | Anticorrosion coating composition for gas transport steel pipe and gas transport steel pipe |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2003110549 | 2003-04-15 | ||
| JP2003328353A JP4560284B2 (en) | 2003-04-15 | 2003-09-19 | Anticorrosion coating composition for gas transport steel pipe and gas transport steel pipe |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2004331939A true JP2004331939A (en) | 2004-11-25 |
| JP4560284B2 JP4560284B2 (en) | 2010-10-13 |
Family
ID=33513171
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2003328353A Expired - Fee Related JP4560284B2 (en) | 2003-04-15 | 2003-09-19 | Anticorrosion coating composition for gas transport steel pipe and gas transport steel pipe |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP4560284B2 (en) |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN100424143C (en) * | 2006-05-18 | 2008-10-08 | 武汉科技大学 | A kind of corrosion-resistant zinc-based aluminum-magnesium rare earth alloy coating and preparation method thereof |
| CN100436910C (en) * | 2007-02-05 | 2008-11-26 | 伦慧东 | Antistatic flame-proof light high-intensity resin steel mesh composite tube and production technique |
| CN100473518C (en) * | 2005-11-07 | 2009-04-01 | 伦慧东 | Technique for steel-plastic composite tube enhanced by antistatic flame retardant Nano fibers and products therefrom |
| CN102618147A (en) * | 2012-04-10 | 2012-08-01 | 武汉铁神化工有限公司 | heat reflective heat insulation heavy anticorrosive paint and use method thereof |
| WO2013009452A1 (en) * | 2011-07-08 | 2013-01-17 | Dow Global Technologies Llc | Cured-in-place pipe rehabilitation process and such cured-in-place pipe |
| CN103497643A (en) * | 2013-09-18 | 2014-01-08 | 浙江绿色德化工有限公司 | Super-weatherproof heavy-duty high-solid thermal insulating electrostatic conducting coating system |
| JP2015527926A (en) * | 2012-07-06 | 2015-09-24 | オンデルゼックスケントルム フォー アーンヴェンディング ファン スタール エン フォーOnderzoekscentrum Voor Aanwending Van Staal N.V. | Pipe for pipelines with internal coating and method of applying the coating |
| CN106800861A (en) * | 2016-12-27 | 2017-06-06 | 天津灯塔涂料工业发展有限公司 | A kind of Chrome-free high solid bi-component epoxy priming paint and preparation method thereof |
| WO2023013482A1 (en) * | 2021-08-04 | 2023-02-09 | 日本ペイント・サーフケミカルズ株式会社 | Steel pipe and method for coating steel pipe |
Citations (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS53101025A (en) * | 1977-02-16 | 1978-09-04 | Dainippon Toryo Co Ltd | Corrosion protection primary paint composiion |
| JPS53104636A (en) * | 1976-04-19 | 1978-09-12 | Dainippon Toryo Co Ltd | Primary anticorrosive coating composition |
| JPS5815571A (en) * | 1981-07-22 | 1983-01-28 | Dainippon Toryo Co Ltd | Paint composition for power transmission towers |
| JPS5817176A (en) * | 1981-07-23 | 1983-02-01 | Dainippon Toryo Co Ltd | Paint composition for power transmission towers |
| JPH02151666A (en) * | 1988-12-05 | 1990-06-11 | Teika Corp | Antirust pigment composition |
| JPH02151664A (en) * | 1988-12-05 | 1990-06-11 | Teika Corp | Antirust pigment composition |
| JPH0373340A (en) * | 1989-08-14 | 1991-03-28 | Nippon Steel Corp | Polyolefin clad steel material excellent in resistance to hot salt water |
| JPH041035A (en) * | 1990-04-18 | 1992-01-06 | Nippon Steel Corp | Heavy anticorrosion coated steel material |
| JPH04110355A (en) * | 1990-08-30 | 1992-04-10 | Teika Corp | Antirust pigment composition |
| JPH04359973A (en) * | 1991-06-06 | 1992-12-14 | Teika Corp | Antirust pigment composition |
| JPH06100802A (en) * | 1992-09-17 | 1994-04-12 | Nippon Paint Co Ltd | Anticorrosion paint composition |
| JPH06340831A (en) * | 1993-05-28 | 1994-12-13 | Kansai Paint Co Ltd | Cationic electrodeposition coating method |
| JPH08259852A (en) * | 1995-03-24 | 1996-10-08 | Shinto Paint Co Ltd | Cold-drying rust-proofing coating composition |
| JPH11279481A (en) * | 1998-03-27 | 1999-10-12 | Sumitomo Metal Ind Ltd | Anticorrosion paint composition for steel |
| JP2001089702A (en) * | 1999-09-24 | 2001-04-03 | Sumitomo Metal Ind Ltd | Anticorrosion paint composition for steel |
-
2003
- 2003-09-19 JP JP2003328353A patent/JP4560284B2/en not_active Expired - Fee Related
Patent Citations (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS53104636A (en) * | 1976-04-19 | 1978-09-12 | Dainippon Toryo Co Ltd | Primary anticorrosive coating composition |
| JPS53101025A (en) * | 1977-02-16 | 1978-09-04 | Dainippon Toryo Co Ltd | Corrosion protection primary paint composiion |
| JPS5815571A (en) * | 1981-07-22 | 1983-01-28 | Dainippon Toryo Co Ltd | Paint composition for power transmission towers |
| JPS5817176A (en) * | 1981-07-23 | 1983-02-01 | Dainippon Toryo Co Ltd | Paint composition for power transmission towers |
| JPH02151666A (en) * | 1988-12-05 | 1990-06-11 | Teika Corp | Antirust pigment composition |
| JPH02151664A (en) * | 1988-12-05 | 1990-06-11 | Teika Corp | Antirust pigment composition |
| JPH0373340A (en) * | 1989-08-14 | 1991-03-28 | Nippon Steel Corp | Polyolefin clad steel material excellent in resistance to hot salt water |
| JPH041035A (en) * | 1990-04-18 | 1992-01-06 | Nippon Steel Corp | Heavy anticorrosion coated steel material |
| JPH04110355A (en) * | 1990-08-30 | 1992-04-10 | Teika Corp | Antirust pigment composition |
| JPH04359973A (en) * | 1991-06-06 | 1992-12-14 | Teika Corp | Antirust pigment composition |
| JPH06100802A (en) * | 1992-09-17 | 1994-04-12 | Nippon Paint Co Ltd | Anticorrosion paint composition |
| JPH06340831A (en) * | 1993-05-28 | 1994-12-13 | Kansai Paint Co Ltd | Cationic electrodeposition coating method |
| JPH08259852A (en) * | 1995-03-24 | 1996-10-08 | Shinto Paint Co Ltd | Cold-drying rust-proofing coating composition |
| JPH11279481A (en) * | 1998-03-27 | 1999-10-12 | Sumitomo Metal Ind Ltd | Anticorrosion paint composition for steel |
| JP2001089702A (en) * | 1999-09-24 | 2001-04-03 | Sumitomo Metal Ind Ltd | Anticorrosion paint composition for steel |
Cited By (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN100473518C (en) * | 2005-11-07 | 2009-04-01 | 伦慧东 | Technique for steel-plastic composite tube enhanced by antistatic flame retardant Nano fibers and products therefrom |
| CN100424143C (en) * | 2006-05-18 | 2008-10-08 | 武汉科技大学 | A kind of corrosion-resistant zinc-based aluminum-magnesium rare earth alloy coating and preparation method thereof |
| CN100436910C (en) * | 2007-02-05 | 2008-11-26 | 伦慧东 | Antistatic flame-proof light high-intensity resin steel mesh composite tube and production technique |
| WO2013009452A1 (en) * | 2011-07-08 | 2013-01-17 | Dow Global Technologies Llc | Cured-in-place pipe rehabilitation process and such cured-in-place pipe |
| US9651189B2 (en) | 2011-07-08 | 2017-05-16 | Blue Cube Ip Llc | Cured-in place pipe rehabilitation process |
| CN102618147A (en) * | 2012-04-10 | 2012-08-01 | 武汉铁神化工有限公司 | heat reflective heat insulation heavy anticorrosive paint and use method thereof |
| JP2015527926A (en) * | 2012-07-06 | 2015-09-24 | オンデルゼックスケントルム フォー アーンヴェンディング ファン スタール エン フォーOnderzoekscentrum Voor Aanwending Van Staal N.V. | Pipe for pipelines with internal coating and method of applying the coating |
| CN103497643A (en) * | 2013-09-18 | 2014-01-08 | 浙江绿色德化工有限公司 | Super-weatherproof heavy-duty high-solid thermal insulating electrostatic conducting coating system |
| CN106800861A (en) * | 2016-12-27 | 2017-06-06 | 天津灯塔涂料工业发展有限公司 | A kind of Chrome-free high solid bi-component epoxy priming paint and preparation method thereof |
| CN106800861B (en) * | 2016-12-27 | 2019-04-19 | 天津灯塔涂料工业发展有限公司 | A kind of Chrome-free high solid bi-component epoxy priming paint and preparation method thereof |
| WO2023013482A1 (en) * | 2021-08-04 | 2023-02-09 | 日本ペイント・サーフケミカルズ株式会社 | Steel pipe and method for coating steel pipe |
Also Published As
| Publication number | Publication date |
|---|---|
| JP4560284B2 (en) | 2010-10-13 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN102991023B (en) | The surface treated steel plate that corrosion stability, conductivity and coating appearance are excellent | |
| JP2004263252A (en) | Chrome-free chemical treated steel sheet with excellent white rust resistance | |
| JP3665046B2 (en) | Surface-treated steel sheet excellent in white rust resistance and method for producing the same | |
| JP2007314762A (en) | Powder coating composition and heavy anticorrosion coated steel material using the same | |
| JP3903739B2 (en) | Organic coated steel plate with excellent corrosion resistance | |
| JP5345874B2 (en) | High corrosion resistance surface-treated steel sheet | |
| JP4879793B2 (en) | High corrosion resistance surface-treated steel sheet | |
| JP4560284B2 (en) | Anticorrosion coating composition for gas transport steel pipe and gas transport steel pipe | |
| WO2021261071A1 (en) | Water-based anticorrosive coating composition | |
| JP4099218B2 (en) | High corrosion-resistant surface-treated steel sheet and manufacturing method thereof | |
| JPS61163971A (en) | Highly durable internally coated steel pipes for gas transportation | |
| JP4844128B2 (en) | High corrosion resistance surface-treated steel sheet and manufacturing method thereof | |
| JP6085932B2 (en) | Polyethylene-coated steel material and epoxy resin primer layer forming material | |
| JP4733874B2 (en) | Polyolefin coated steel | |
| JP5640960B2 (en) | Heavy duty anti-corrosion coated steel pipe | |
| JPS61162564A (en) | Corrosionproof coating composition | |
| JP5933095B1 (en) | Anticorrosion paint composition, paint film, ship and marine structure | |
| JP6398851B2 (en) | Polyolefin-coated steel with base conversion treatment | |
| JPH0373340A (en) | Polyolefin clad steel material excellent in resistance to hot salt water | |
| JP3787047B2 (en) | Anticorrosive paint composition for steel | |
| JP4665151B2 (en) | Anticorrosion paint composition for steel and steel coated with the same | |
| JP6623543B2 (en) | Organic resin coated steel | |
| JP6607265B2 (en) | Polyethylene-coated steel pipe and method for producing the same | |
| JPH0630726B2 (en) | Painted steel | |
| JP2000073003A (en) | Anticorrosion paint composition for steel |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20051122 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20090210 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20090414 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20090612 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A821 Effective date: 20090612 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20100427 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20100611 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20100629 |
|
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20100726 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 4560284 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20130730 Year of fee payment: 3 |
|
| S533 | Written request for registration of change of name |
Free format text: JAPANESE INTERMEDIATE CODE: R313533 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20130730 Year of fee payment: 3 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| S533 | Written request for registration of change of name |
Free format text: JAPANESE INTERMEDIATE CODE: R313533 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| LAPS | Cancellation because of no payment of annual fees |