JP2001003266A5 - - Google Patents
Download PDFInfo
- Publication number
- JP2001003266A5 JP2001003266A5 JP1999170622A JP17062299A JP2001003266A5 JP 2001003266 A5 JP2001003266 A5 JP 2001003266A5 JP 1999170622 A JP1999170622 A JP 1999170622A JP 17062299 A JP17062299 A JP 17062299A JP 2001003266 A5 JP2001003266 A5 JP 2001003266A5
- Authority
- JP
- Japan
- Prior art keywords
- group
- formula
- carbon fiber
- compound represented
- embedded image
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 229920000049 Carbon (fiber) Polymers 0.000 description 17
- 239000004917 carbon fiber Substances 0.000 description 17
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 12
- 150000001875 compounds Chemical class 0.000 description 11
- 238000004513 sizing Methods 0.000 description 9
- -1 methacryloyl group Chemical group 0.000 description 8
- 239000003795 chemical substances by application Substances 0.000 description 7
- 125000000524 functional group Chemical group 0.000 description 7
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 6
- 125000003700 epoxy group Chemical group 0.000 description 5
- 125000003647 acryloyl group Chemical group O=C([*])C([H])=C([H])[H] 0.000 description 3
- 230000001476 alcoholic effect Effects 0.000 description 3
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 3
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N phenol group Chemical group C1(=CC=CC=C1)O ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 3
- JOYRKODLDBILNP-UHFFFAOYSA-N Ethyl urethane Chemical compound CCOC(N)=O JOYRKODLDBILNP-UHFFFAOYSA-N 0.000 description 2
- 239000000835 fiber Substances 0.000 description 2
- 150000002430 hydrocarbons Chemical group 0.000 description 2
- 238000004519 manufacturing process Methods 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 238000000034 method Methods 0.000 description 2
- 239000000178 monomer Substances 0.000 description 2
- 239000000243 solution Substances 0.000 description 2
- 238000009987 spinning Methods 0.000 description 2
- FKTHNVSLHLHISI-UHFFFAOYSA-N 1,2-bis(isocyanatomethyl)benzene Chemical compound O=C=NCC1=CC=CC=C1CN=C=O FKTHNVSLHLHISI-UHFFFAOYSA-N 0.000 description 1
- ZONNLCHLAZWCEN-UHFFFAOYSA-N 3-(2-hydroxyethyl)-4-[2-(2-methylprop-2-enoyloxy)ethyl]phthalic acid Chemical compound CC(=C)C(=O)OCCC1=CC=C(C(O)=O)C(C(O)=O)=C1CCO ZONNLCHLAZWCEN-UHFFFAOYSA-N 0.000 description 1
- 229920002972 Acrylic fiber Polymers 0.000 description 1
- NLHHRLWOUZZQLW-UHFFFAOYSA-N Acrylonitrile Chemical group C=CC#N NLHHRLWOUZZQLW-UHFFFAOYSA-N 0.000 description 1
- 125000000217 alkyl group Chemical group 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 238000009835 boiling Methods 0.000 description 1
- 230000015271 coagulation Effects 0.000 description 1
- 238000005345 coagulation Methods 0.000 description 1
- 229920001577 copolymer Polymers 0.000 description 1
- 125000005442 diisocyanate group Chemical group 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 239000003733 fiber-reinforced composite Substances 0.000 description 1
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 1
- 150000002440 hydroxy compounds Chemical class 0.000 description 1
- 125000005395 methacrylic acid group Chemical group 0.000 description 1
- 239000000203 mixture Substances 0.000 description 1
- 238000000465 moulding Methods 0.000 description 1
- 125000000962 organic group Chemical group 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 239000012779 reinforcing material Substances 0.000 description 1
- 239000011342 resin composition Substances 0.000 description 1
- 238000010301 surface-oxidation reaction Methods 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 230000002194 synthesizing effect Effects 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
Description
【特許請求の範囲】
【請求項1】 下記式(1)で表わされる化合物を主成分とすることを特徴とする炭素繊維用サイズ剤。
【化1】
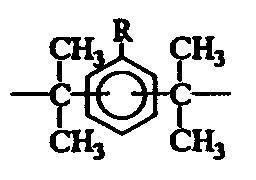
[式(1)において、A及びBは、ビニル基、アクリロイル基、メタクリロイル基、エポキシ基、環式脂肪族エポキシ基、カルボキシル基、及びフェノール性水酸基より選ばれる1個の官能基を具備する有機残基であり、AとBはそれぞれ異なる。
又、Xは下記の[化2]〜[化8]に示すいずれか1個の炭化水素基であり、該炭化水素基中のRは水素原子又はアルキル基であり、nは0〜12の整数であり、mは2〜12の整数である。]
【化2】
【化3】
【化4】
【化5】
【化6】
【化7】
【化8】
【請求項2】 請求項1に記載の炭素繊維用サイズ剤において、式(1)で表わされる化合物のA及びBのうちの少なくともいずれかが、ビニル基、アクリロイル基、メタクリロイル基、エポキシ基、環式脂肪族エポキシ基、カルボキシル基、及びフェノール性水酸基より選ばれる1個の官能基を末端基として具備する有機残基であることを特徴とする炭素繊維用サイズ剤。
【請求項3】 請求項1または2に記載の炭素繊維用サイズ剤において、式(1)で表わされる化合物が、1分子のジイソシアネートモノマーに対して、異なる官能基を有する2種類のアルコール性モノヒドロキシ化合物を反応させるウレタン化反応によって生成するウレタン化合物であることを特徴とする炭素繊維用サイズ剤。
【請求項4】 請求項1〜3のいずれかの1項に記載の炭素繊維用サイズ剤の水溶液又は水分散液からなるサイズ剤溶液によって炭素繊維を処理することを特徴とする炭素繊維のサイジング方法。
【請求項5】 請求項1〜3のいずれかの1項に記載の炭素繊維用サイズ剤がその表面に付着していることを特徴とするサイジング処理された炭素繊維。
【請求項6】 請求項5に記載の炭素繊維を使用したシート状物からなることを特徴とするサイジング処理された炭素繊維によるシート状物。
【請求項7】 請求項5に記載の炭素繊維或いは請求項6に記載のシート状物を強化材とする繊維強化樹脂組成物を成形してなることを特徴とする繊維強化複合材料。
[Claims]
1. A sizing agent for carbon fibers comprising a compound represented by the following formula (1) as a main component.
Embedded image
[In the formula (1), A and B represent an organic group having one functional group selected from a vinyl group, an acryloyl group, a methacryloyl group, an epoxy group, a cycloaliphatic epoxy group, a carboxyl group, and a phenolic hydroxyl group. A and B are different from each other.
X is any one of the following hydrocarbon groups represented by the following [Chemical Formula 2] to [Chemical Formula 8]; R in the hydrocarbon group is a hydrogen atom or an alkyl group; And m is an integer of 2 to 12. ]
Embedded image
Embedded image
Embedded image
Embedded image
Embedded image
Embedded image
Embedded image
2. The sizing agent for carbon fibers according to claim 1, wherein at least one of A and B of the compound represented by the formula (1) is a vinyl group, an acryloyl group, a methacryloyl group, an epoxy group, A sizing agent for carbon fibers, which is an organic residue having, as a terminal group, one functional group selected from a cyclic aliphatic epoxy group, a carboxyl group, and a phenolic hydroxyl group.
3. The sizing agent for carbon fibers according to claim 1, wherein the compound represented by the formula (1) has two kinds of alcoholic monomers having different functional groups with respect to one molecule of diisocyanate monomer. A sizing agent for carbon fibers, which is a urethane compound produced by a urethanization reaction of reacting a hydroxy compound.
4. A carbon fiber sizing characterized by treating a carbon fiber with a sizing solution comprising an aqueous solution or an aqueous dispersion of the sizing agent for a carbon fiber according to claim 1. Method.
5. A sizing-processed carbon fiber, characterized in that the carbon fiber sizing agent according to any one of claims 1 to 3 is adhered to the surface thereof.
6. A sheet made of a sizing-processed carbon fiber, comprising a sheet made of the carbon fiber according to claim 5. Description:
7. A fiber-reinforced composite material obtained by molding a fiber-reinforced resin composition using the carbon fiber according to claim 5 or the sheet-like material according to claim 6 as a reinforcing material.
又該式(1)で表わされる化合物は、異なる官能基を有する2種類のアルコール性モノヒドロキシ化合物を反応させるウレタン化反応によって生成するウレタン化合物であることが好ましい。 The compound represented by the formula (1) is preferably a urethane compound formed by a urethanization reaction in which two kinds of alcoholic monohydroxy compounds having different functional groups are reacted.
上記式(1)で表わされる化合物中のA及びBは、ビニル基、アクリロイル基、メタクリロイル基、エポキシ基、環式脂肪族エポキシ基、カルボキシル基、及びフェノール性水酸基より選ばれる1個の官能基を具備する有機残基であり、AとBはそれぞれ異なる。 A and B in the compound represented by the above formula (1) are one functional group selected from a vinyl group, an acryloyl group, a methacryloyl group, an epoxy group, a cycloaliphatic epoxy group, a carboxyl group, and a phenolic hydroxyl group. And A and B are different from each other.
更に式(1)で表わされる化合物を上記のウレタン化反応によって合成する際には、異なる官能基を有する2種類のアルコール性モノヒドロキシ化合物の混合物を用いる場合には、ジイソシアネート化合物に由来する分子構造を中心にしてその左,右が異なる分子構造のウレタン結合を有していて、しかもそれぞれの末端の官能基が異なる式(1)で表わされる化合物を得ることができる。 Further, when the compound represented by the formula (1) is synthesized by the above urethane reaction, when a mixture of two kinds of alcoholic monohydroxy compounds having different functional groups is used, the molecular structure derived from the diisocyanate compound is used. A compound represented by the formula (1) having urethane bonds having different molecular structures on the left and right sides of the compound represented by the formula (1) and having different functional groups at the respective ends can be obtained.
参考例1
[炭素繊維束の製造]
アクリロニトリル単位97重量%とメタクリル酸単位3重量%とのアクリロニトリル共重合体をジメチルホルムアミド(DMF)に溶解させた紡糸液を、紡糸ノズルを通して凝固浴中に吐出して紡糸した後、洗浄、及び沸水延伸に付し、更に沸水による洗浄、及び乾燥を施すことにより、単糸デニール1.2の炭素繊維用の前駆体繊維としてのアクリル系繊維を得た。
Reference Example 1
[Manufacture of carbon fiber bundles]
A spinning solution obtained by dissolving an acrylonitrile copolymer of 97% by weight of acrylonitrile unit and 3% by weight of methacrylic acid unit in dimethylformamide (DMF) is discharged through a spinning nozzle into a coagulation bath, spun, washed, and boiled. The fiber was drawn, washed with boiling water, and dried to obtain an acrylic fiber as a precursor fiber for carbon fiber having a single denier of 1.2.
参考例2
[式(1)で表されるウレタン化合物の合成]
参考例1のウレタン化合物の合成の手法と同様にして、2−メタクリロイルオキシエチル−2−ヒドロキシエチルフタル酸2.1モルと、キシリレンジイソシアネート1モルとから、下記の[化26]に示すウレタン化合物1モルを合成した。
Reference example 2
[Synthesis of urethane compound represented by formula (1)]
In the same manner as in the method for synthesizing the urethane compound of Reference Example 1, 2.1 mol of 2-methacryloyloxyethyl-2-hydroxyethylphthalic acid and 1 mol of xylylene diisocyanate were used to form the urethane represented by the following [Chemical Formula 26]. One mole of the compound was synthesized.
実施例3
[炭素繊維束の製造]
参考例1と同様にして、電気化学的な表面酸化処理を施してあるフィラメント数3,000本の炭素繊維束を得た。
Example 3
[Manufacture of carbon fiber bundles]
In the same manner as in Reference Example 1, a carbon fiber bundle having 3,000 filaments subjected to an electrochemical surface oxidation treatment was obtained.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP17062299A JP4216409B2 (en) | 1999-06-17 | 1999-06-17 | Carbon fiber sizing agent, carbon fiber sizing method, sized carbon fiber, sheet-like material using the carbon fiber, and fiber-reinforced composite material |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP17062299A JP4216409B2 (en) | 1999-06-17 | 1999-06-17 | Carbon fiber sizing agent, carbon fiber sizing method, sized carbon fiber, sheet-like material using the carbon fiber, and fiber-reinforced composite material |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2001003266A JP2001003266A (en) | 2001-01-09 |
| JP2001003266A5 true JP2001003266A5 (en) | 2006-07-27 |
| JP4216409B2 JP4216409B2 (en) | 2009-01-28 |
Family
ID=15908298
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP17062299A Expired - Lifetime JP4216409B2 (en) | 1999-06-17 | 1999-06-17 | Carbon fiber sizing agent, carbon fiber sizing method, sized carbon fiber, sheet-like material using the carbon fiber, and fiber-reinforced composite material |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP4216409B2 (en) |
Families Citing this family (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| TWI220147B (en) * | 2001-07-24 | 2004-08-11 | Mitsubishi Rayon Co | Sizing agent for carbon fibers and water dispersion thereof, sized carbon fibers, sheet-like articles using said carbon fibers, and carbon fiber enhanced composite material |
| JP2007070428A (en) * | 2005-09-06 | 2007-03-22 | Fujitsu Ltd | Vegetable resin-containing composition and vegetable resin-containing molded article |
| KR101602200B1 (en) | 2010-01-20 | 2016-03-10 | 도레이 카부시키가이샤 | Carbon fiber bundles |
| JP5872245B2 (en) * | 2011-10-24 | 2016-03-01 | 三菱レイヨン株式会社 | Oil agent composition for carbon fiber precursor acrylic fiber, and oil agent treatment liquid for carbon fiber precursor acrylic fiber |
| HUE035239T2 (en) | 2011-06-06 | 2018-05-02 | Mitsubishi Chem Corp | Oil agent for carbon fiber precursor acrylic fiber, processed-oil solution for carbon fiber precursor acrylic fibers, carbon fiber precursor acrylic fiber bundle, and method for producing carbon fiber bundle using carbon fiber precursor acrylic fiber ... |
| JP5831129B2 (en) * | 2011-10-24 | 2015-12-09 | 三菱レイヨン株式会社 | Carbon fiber precursor acrylic fiber bundle |
| JP6059903B2 (en) | 2012-08-06 | 2017-01-11 | ダイセルポリマー株式会社 | Thermoplastic resin composition |
| KR20150042829A (en) | 2012-08-15 | 2015-04-21 | 쓰리엠 이노베이티브 프로퍼티즈 컴파니 | Sized short alumina-based inorganic oxide fiber, method of making, and composition including the same |
| JP6884983B2 (en) * | 2016-02-01 | 2021-06-09 | 三菱瓦斯化学株式会社 | Fiber Reinforced Thermoplastic Resin Composition Composite Material and Mold |
| JP7463123B2 (en) * | 2019-02-22 | 2024-04-08 | 三洋化成工業株式会社 | Fiber sizing agent |
| EP4255967A1 (en) * | 2020-12-04 | 2023-10-11 | Toray Composite Materials America, Inc. | Prepreg and carbon fiber-reinforced composite material |
| CN113550148A (en) * | 2021-08-03 | 2021-10-26 | 长春工业大学 | High-surface-energy carbon fiber water-soluble sizing agent for carbon paper and preparation method thereof |
| CN113957718B (en) * | 2021-09-30 | 2024-10-01 | 青岛汇智领先新材料科技有限公司 | Preparation method of environment-friendly modified polyurethane vinyl carbon fiber sizing agent |
| CN118407253B (en) * | 2024-04-24 | 2025-07-25 | 华东理工大学华昌聚合物有限公司 | Carbon nanotube modified vinyl carbon fiber sizing agent composition and preparation method and application thereof |
-
1999
- 1999-06-17 JP JP17062299A patent/JP4216409B2/en not_active Expired - Lifetime
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP2001003266A5 (en) | ||
| US11332852B2 (en) | Carbon-fiber-precursor fiber bundle, carbon fiber bundle, and uses thereof | |
| US6517933B1 (en) | Hybrid polymer materials | |
| JP3438957B2 (en) | Aqueous surface treatment agent for reinforcement, reinforcement treated with the aqueous surface treatment, and composite reinforced with reinforcement | |
| CN105037667A (en) | Amphipathic polyvinylidene-fluoride-base block copolymer and application thereof | |
| JP4216409B2 (en) | Carbon fiber sizing agent, carbon fiber sizing method, sized carbon fiber, sheet-like material using the carbon fiber, and fiber-reinforced composite material | |
| CN116180296A (en) | Antibacterial fabric and preparation method thereof | |
| JPS61225373A (en) | Carbon fiber bundle | |
| JP2004238761A (en) | Carbon fiber strand and fiber-reinforced composite material | |
| CN110804161B (en) | Low-viscosity bio-based epoxy resin composition and carbon fiber composite material | |
| JPS63270863A (en) | Sizing agent for carbon fiber | |
| CN118562162B (en) | Preparation process of hyperbranched polyether amide modified glass fiber reinforced nylon composite material | |
| JP3194241B2 (en) | Method for modifying cellulose fiber | |
| JP3502962B2 (en) | Block copolymer oligomer, method for producing the same, antifouling agent and fiber product | |
| CN113480707B (en) | Aramid fiber water-soluble epoxy resin surface sizing agent and its preparation and application | |
| CN118421168A (en) | A method for preparing a wear-resistant water-based coating | |
| CN102102235A (en) | Spinning solution containing silicon acrylonitrile copolymer and preparation method thereof | |
| CN113736015B (en) | A kind of biomimetic polymer containing dopamine functional group and preparation method thereof | |
| JPH07101965A (en) | Alkoxysilane compound | |
| KR101254783B1 (en) | Fluorinated cellulose acetate, the fabrication method thereof, and the piezoelectric paper obtained therefrom | |
| CN1970877A (en) | Wool fabric soft finish and application therefor | |
| JP2657259B2 (en) | Modifier composition for composite material and composite material using the same | |
| JPH0325550B2 (en) | ||
| CN111909117A (en) | A kind of phthalic anhydride diisopropanolamide epoxy resin and preparation method and application thereof | |
| CN116355388B (en) | Polyurethane anticorrosion composite material bracket |