FR2840806A1 - COLORED SELF-TANNING COMPOSITIONS COMPRISING AT LEAST ONE RED OR ORANGE DYE CHOSEN AMONG FLUORANES OR THEIR ALKALI METAL SALTS - Google Patents
COLORED SELF-TANNING COMPOSITIONS COMPRISING AT LEAST ONE RED OR ORANGE DYE CHOSEN AMONG FLUORANES OR THEIR ALKALI METAL SALTS Download PDFInfo
- Publication number
- FR2840806A1 FR2840806A1 FR0207289A FR0207289A FR2840806A1 FR 2840806 A1 FR2840806 A1 FR 2840806A1 FR 0207289 A FR0207289 A FR 0207289A FR 0207289 A FR0207289 A FR 0207289A FR 2840806 A1 FR2840806 A1 FR 2840806A1
- Authority
- FR
- France
- Prior art keywords
- composition according
- red
- derivatives
- weight
- chosen
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/49—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds
- A61K8/4973—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with oxygen as the only hetero atom
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
- A61Q19/04—Preparations for care of the skin for chemically tanning the skin
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Dermatology (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Birds (AREA)
- Epidemiology (AREA)
- Cosmetics (AREA)
Abstract
L'invention concerne des compositions cosmétiques et/ou dermatologiques colorées à usage topique destinées au bronzage etlou au brunissage artificiels de la peau, caractérisées par le fait qu'elle comprennent, dans un support cosmétiquement acceptable, au moins un agent autobronzant mono- oupolycarbonylé et au moins un colorant rouge ou orange choisi parmi les fluoranes ou leurs sels de métal alcalin. La composition colorée selon l'invention ne se décolore pas dans le temps et présente d'autre part une bonne efficacité auto-bronzante sur la peau (intensité et tenue de la coloration). L'invention concerne également l'utilisation de cette composition dans l'application cosmétique susmentionnée.The invention relates to colored cosmetic and / or dermatological compositions for topical use intended for artificial tanning and / or browning of the skin, characterized in that they comprise, in a cosmetically acceptable carrier, at least one mono- or polycarbonylated self-tanning agent and at least one red or orange dye chosen from fluoranes or their alkali metal salts. The colored composition according to the invention does not fade over time and also exhibits good self-tanning efficacy on the skin (intensity and durability of the coloring). The invention also relates to the use of this composition in the above-mentioned cosmetic application.
Description
<Desc/Clms Page number 1> <Desc / Clms Page number 1>
COMPOSITIONS AUTOBRONZANTES COLOREES COMPORTANT AU MOINS UN COLORANT ROUGE OU ORANGE CHOISI PARMI LES FLUORANES OU LEURS SELS DE METAL ALCALIN L'invention concerne des compositions cosmétiques et/ou dermatologiques colorées à usage topique destinées au bronzage et/ou au brunissage artificiels de la peau, caractérisées par le fait qu'elle comprennent, dans un support cosmétiquement acceptable, au moins un agent autobronzant mono- ou polycarbonylé et au moins un colorant rouge ou orange choisi parmi les fluoranes ou leurs sels de métal alcalin. COLORED SELF-TANNING COMPOSITIONS COMPRISING AT LEAST ONE RED OR ORANGE DYE CHOSEN AMONG FLUORANES OR THEIR ALKALI METAL SALTS The invention relates to colored cosmetic and / or dermatological compositions for topical use intended for artificial tanning and / or browning of the skin, characterized by the fact that they comprise, in a cosmetically acceptable carrier, at least one self-tanning agent mono- or polycarbonylated and at least one red or orange dye chosen from fluoranes or their alkali metal salts.
On sait que les composés mono- ou polycarbonylés tels que par exemple l'isatine, l'alloxane, la ninhydrine, le glycéraldéhyde, l'aldéhyde mésotartrique, la glutaraldéhyde, l'érythrulose, la dihydroxyacétone (DHA) sont des produits particulièrement intéressants qui sont couramment utilisés en cosmétique comme agents de bronzage artificiel de la peau. It is known that mono- or polycarbonylated compounds such as for example isatin, alloxane, ninhydrin, glyceraldehyde, mesotartaric aldehyde, glutaraldehyde, erythrulose, dihydroxyacetone (DHA) are particularly interesting products which are commonly used in cosmetics as artificial tanning agents for the skin.
Appliqués sur la peau, notamment sur le visage, ces composés permettent d'obtenir un effet de bronzage ou de brunissage d'apparence plus ou moins semblable à celui qui peut résulter d'une exposition prolongée au soleil (bronzage naturel) ou sous une lampe UV. Une telle utilisation présente en outre pour avantage d'éviter totalement les risques de réaction cutanée généralement attachés aux expositions prolongées précitées (érythèmes, brûlures, perte d'élasticité, apparition de rides, vieillissement prématuré de la peau, et autres). Applied to the skin, in particular to the face, these compounds make it possible to obtain a tanning or browning effect of appearance more or less similar to that which may result from prolonged exposure to the sun (natural tanning) or under a lamp. UV. Such use also has the advantage of completely avoiding the risks of skin reaction generally associated with the aforementioned prolonged exposures (erythema, burns, loss of elasticity, appearance of wrinkles, premature aging of the skin, and others).
Pour mieux visualiser l'application des formulations autobronzantes sur la peau ou pour les rendre plus esthétiques, on incorpore généralement dans la composition des colorants organiques hydrosolubles ou liposolubles rouges ou oranges ou bien des pigments minéraux rouges ou oranges comme les oxydes de fer. Afin d'obtenir une large gamme de nuances ou s'il on souhaite visualiser sur la peau l'application du produit autobronzant par une teinte proche de la peau, la présence des colorants rouges ou oranges dans la composition est nécessaire. To better visualize the application of self-tanning formulations on the skin or to make them more aesthetic, red or orange water-soluble or liposoluble organic dyes or red or orange mineral pigments such as iron oxides are generally incorporated into the composition. In order to obtain a wide range of shades or if it is desired to visualize on the skin the application of the self-tanning product with a shade close to the skin, the presence of red or orange dyes in the composition is necessary.
Les oxydes de fer sont généralement incompatibles avec les composés mono- ou polycarbonylés et plus particulièrement la dihydroxyacétone (DHA). Les colorants liposolubles ont pour inconvénients de teindre les articles de conditionnement et de tacher la peau et les vêtements. Les formulations autobronzantes colorées à base de colorants hydrosolubles organiques rouges ne présentent pas ces inconvénients mais ont généralement tendance à se décolorer dans le temps. Iron oxides are generally incompatible with mono- or polycarbonylated compounds and more particularly dihydroxyacetone (DHA). The drawbacks of fat-soluble dyes are the dyeing of packaging articles and the staining of skin and clothing. Colored self-tanning formulations based on red organic water-soluble dyes do not have these drawbacks but generally tend to discolour over time.
On connaît également dans les demandes de brevet EP1060738 et le brevet US 6,113,888 des compositions autobronzantes colorées à base de carmin de cochenille. Ce colorant particulier est une matière première d'origine animale, de qualité peu reproductible et dont les approvisionnements sont dépendants des conditions climatiques. Also known in patent applications EP1060738 and US patent 6,113,888 are colored self-tanning compositions based on cochineal carmine. This particular dye is a raw material of animal origin, of poorly reproducible quality and whose supplies are dependent on climatic conditions.
La présente invention recherche de nouvelles compositions autobronzantes colorées à base d'un composé mono- ou polycarbonylé et contenant au moins un colorant rouge qui ne présentent pas les inconvénients définis ci-dessus et notamment qui ne se décolorent pas dans le temps et présentent d'autre part une bonne efficacité autobronzante sur la peau (intensité et tenue de la coloration). The present invention seeks new colored self-tanning compositions based on a mono- or polycarbonylated compound and containing at least one red dye which do not have the drawbacks defined above and in particular which do not fade over time and exhibit on the other hand good self-tanning efficacy on the skin (intensity and color fastness).
<Desc/Clms Page number 2> <Desc / Clms Page number 2>
Ainsi, à la suite d'importantes recherches menées sur la question, il a maintenant été trouvé par la Demanderesse, et ceci de façon tout à fait inattendue et surprenante, qu'il est possible d'améliorer la stabilité dans le temps de la couleur de compositions autobronzantes colorées en utilisant un colorant rouge ou orange du type fluorane ou son sel de métal alcalin. Les compositions obtenues ne présentent pas les inconvénients indiqués ci-dessus propres à l'utilisation des colorants de l'art antérieur. Thus, as a result of important research carried out on the question, it has now been found by the Applicant, and this in a completely unexpected and surprising manner, that it is possible to improve the stability over time of the color. of self-tanning compositions colored using a red or orange dye of the fluorane type or its alkali metal salt. The compositions obtained do not have the drawbacks indicated above specific to the use of dyes of the prior art.
Cette découverte est à la base de la présente invention. This discovery is the basis of the present invention.
Conformément à l'un des objets de la présente invention, il est donc maintenant proposé de nouvelles compositions cosmétiques et/ou dermatologiques colorées destinées au bronzage artificiel de la peau, caractérisées par le fait qu'elles contiennent, dans un support cosmétiquement acceptable, au moins un agent autobronzant mono- ou polycarbonylé et au moins un colorant rouge ou orange choisi parmi les fluoranes et leurs sels de métal alcalin. In accordance with one of the objects of the present invention, it is therefore now proposed new cosmetic and / or dermatological colored compositions intended for artificial tanning of the skin, characterized in that they contain, in a cosmetically acceptable carrier, at at least one mono- or polycarbonylated self-tanning agent and at least one red or orange dye chosen from fluoranes and their alkali metal salts.
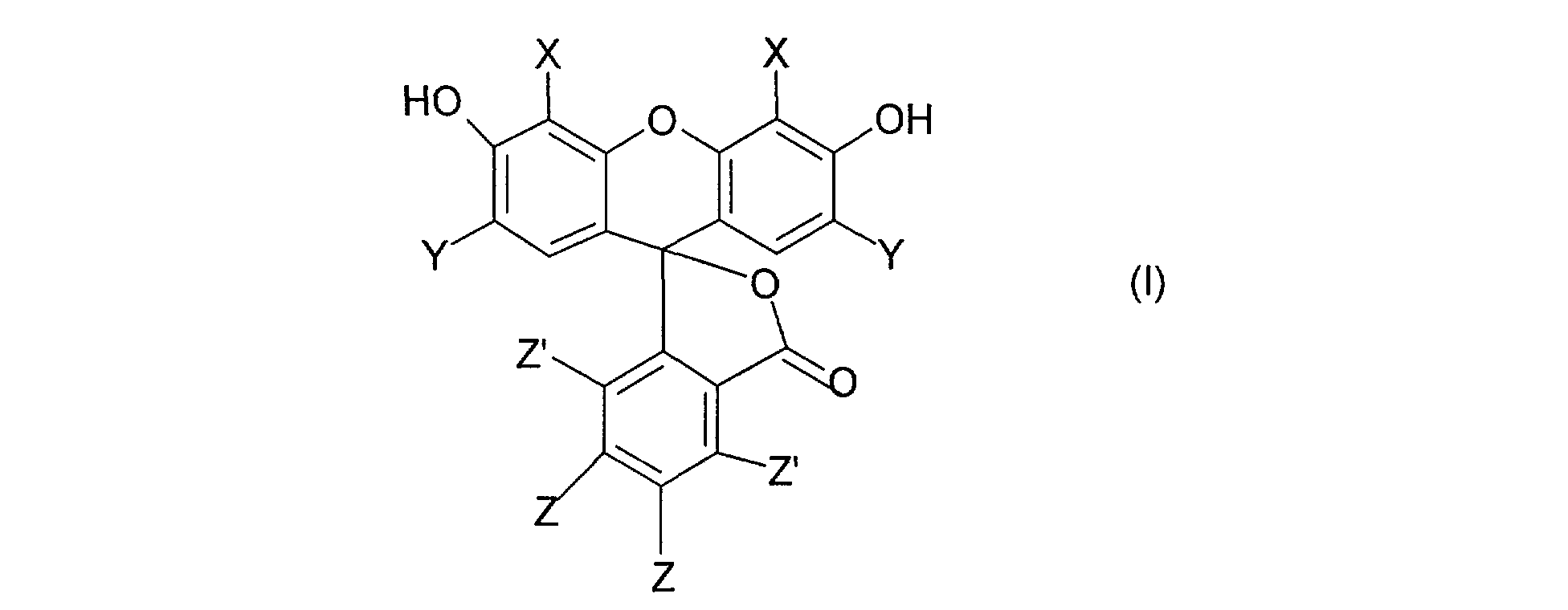
Les colorants rouges ou oranges du type fluorane conformes à l'invention répondent à la formule (1) suivante :
dans laquelle X, Y, Z et Z', indépendamment les uns des autres, désignent un atome d'hydrogène ou un atome d'halogène sous réserve qu'au moins un des groupes X, Y Z et Z' désigne un atome d'halogène. The red or orange dyes of the fluorane type in accordance with the invention correspond to the following formula (1):
in which X, Y, Z and Z ', independently of each other, denote a hydrogen atom or a halogen atom provided that at least one of the groups X, YZ and Z' denotes a halogen atom .
Parmi les colorants du type fluorane de formule (I) conformes à l'invention, on peut citer : - le tetrabromofluorosceine ou éosine (X=Y=Br et Z= Z'= H) connu sous le nom CTFA : CI 45380 ou Red 21 - la phloxine B (X=Y=Br et Z=Z'= CI) connu sous le nom CTFA : CI 45410 ou Red 27 - la diiodofluoresceine (X= I, Y=Z=Z'=H) connu sous le nom CTFA : CI 45425 ou Orange 10 ; - la dibromofluoresceine (X= Br, Y=Z=Z'= H) connu sous le nom CTFA : CI 45370 ou Orange 5. Among the fluorane type dyes of formula (I) in accordance with the invention, there may be mentioned: - tetrabromofluoroscein or eosin (X = Y = Br and Z = Z '= H) known under the name CTFA: CI 45380 or Red 21 - phloxine B (X = Y = Br and Z = Z '= CI) known as CTFA: CI 45410 or Red 27 - diiodofluorescein (X = I, Y = Z = Z' = H) known as CTFA name: CI 45425 or Orange 10; - dibromofluorescein (X = Br, Y = Z = Z '= H) known under the name CTFA: CI 45370 or Orange 5.
<Desc/Clms Page number 3><Desc / Clms Page number 3>
Leurs sels de métal alcalin correspondants répondent à la formule (II) suivante :
dans laquelle X, Y, Z et Z' ont les mêmes significations indiquées dans la formule (I) ci-dessus et M désigne un métal alcalin tel que Na et K . Their corresponding alkali metal salts correspond to the following formula (II):
in which X, Y, Z and Z 'have the same meanings indicated in formula (I) above and M denotes an alkali metal such as Na and K.
Parmi les colorants de formule (II) conformes à l'invention, on peut citer : - le sel de sodium de la tetrabromofluorosceine (X=Y= Br ; H ; = Na) connu sous le nom CTFA : CI 45380 (Na sait) ou Red 22 - le sel de sodium de la phloxine B (X=Y= Br ; Z= Z'= CI; M= Na) connu sous le nom CTFA : CI 45410 (Na salt) ou Red 28 - le sel de sodium de la diiodofluoresceine (X= # ; Y=Z=Z'= H ; M= Na) connu sous le nom CTFA : CI 45425 (Na sait) ou Orange 11 ; - l'érythrosine (X= Y= # ; Z= Z'= H ; M= Na) connu sous le nom CTFA : CI 45430 ou Acid Red 51. Among the dyes of formula (II) in accordance with the invention, there may be mentioned: - the sodium salt of tetrabromofluoroscein (X = Y = Br; H; = Na) known under the name CTFA: CI 45380 (Na knows) or Red 22 - the sodium salt of phloxin B (X = Y = Br; Z = Z '= CI; M = Na) known as CTFA: CI 45410 (Na salt) or Red 28 - the sodium salt diiodofluorescein (X = #; Y = Z = Z '= H; M = Na) known as CTFA: CI 45425 (Na knows) or Orange 11; - Erythrosine (X = Y = #; Z = Z '= H; M = Na) known as CTFA: CI 45430 or Acid Red 51.
- la phloxine (X= Y= Br ; Z= H ;Z' = CI ;M= K) connu sous le nom CTFA : CI 45405 ou Acid Red 98. - phloxine (X = Y = Br; Z = H; Z '= CI; M = K) known as CTFA: CI 45405 or Acid Red 98.
On utilisera plus particulièrement pour l'invention la tetrabromofluorosceine (Red 21) ou son sel de sodium (Red 22). More particularly, the tetrabromofluoroscein (Red 21) or its sodium salt (Red 22) will be used for the invention.
Les colorants fluoranes ou leurs sels de sodium conformes à l'invention sont de préférence présents dans les compositions de l'invention dans des concentrations allant de 0. 0001 à 0.5% en poids et plus préférentiellement de 0.001% à 0. 2 % en poids par rapport au poids total de la composition. The fluorane dyes or their sodium salts in accordance with the invention are preferably present in the compositions of the invention in concentrations ranging from 0.001% to 0.5% by weight and more preferably from 0.001% to 0.2% by weight relative to the total weight of the composition.
Les agents autobronzants mono ou polycarbonylés sont choisis par exemple parmi l'isatine, l'alloxane, la ninhydrine, le glycéraldéhyde, l'aldéhyde mésotartrique, la glutaraldéhyde, l'érythrulose, les dérivés de pyrazolin-4,5-diones telles que décrits dans la demande de brevet FR 2466492 et WO 9735842, la dihydroxyacétone (DHA), les dérivés de 4,4-dihydroxypyrazolin-5-ones telles que décrits dans la demande de brevet EP-A-0903342 . The mono or polycarbonylated self-tanning agents are chosen, for example, from isatin, alloxane, ninhydrin, glyceraldehyde, mesotartric aldehyde, glutaraldehyde, erythrulose, pyrazolin-4,5-diones derivatives as described in patent application FR 2466492 and WO 9735842, dihydroxyacetone (DHA), derivatives of 4,4-dihydroxypyrazolin-5-ones as described in patent application EP-A-0903342.
Dans un mode de réalisation particulièrement préféré de l'invention on utilisera plus particulièrement comme agent autobronzant la dihydroxyacétone (DHA). In a particularly preferred embodiment of the invention, use will more particularly be made as self-tanning agent of dihydroxyacetone (DHA).
Les compositions conformes à l'invention peuvent contenir en plus d'autres agents de coloration artificielle de la peau parmi lesquels on peut citer notamment : The compositions in accordance with the invention may also contain other artificial coloring agents for the skin, among which there may be mentioned in particular:
<Desc/Clms Page number 4><Desc / Clms Page number 4>
(i) les dérivés indoliques comme les monohydroxyindoles tels que décrits dans le brevet FR2651126 ( ie : 5-, 6- ou 7-hydroxyindole) ou les di-hydroxyindoles tels que décrits dans le brevet EP-B-0425324 (ie : 5,6-dihydroxyindole, 2-méthyl 5,6dihydroxyindole, 3-méthyl 5,6-dihydroxyindole, 2,3-diméthyl 5,6-dihydroxyindole) ; (ii) les extraits végétaux agents de coloration artificielle de la peau tels que : - les extraits de bois rouges "insolubles" du genre Pterocarpus et du genre Baphia comme le Pterocarpus santalinus, le Pterocarpus osun, le Pterocarpus soyauxii, le Pterocarpus erinaceus, le Pterocarpus indicus ou encore le Baphia nitida tels que ceux décrits dans la demande de brevet EP-A-0 971683 ; - les extraits végétaux du genre Saxifraga comme le Saxifraga cuneifolia, Saxifraga glaucescens, Saxifraga rotundifolia, Saxifraga granulata, Saxifraga bulbifera, Saxifraga umbrosa, Saxifraga tridactylites ; - les extraits végétaux du genre Sorgho comme le Sorghum caudatum , le
Sorghum Bicolor. (i) indole derivatives such as monohydroxyindoles as described in patent FR2651126 (ie: 5-, 6- or 7-hydroxyindole) or di-hydroxyindoles as described in patent EP-B-0425324 (ie: 5, 6-dihydroxyindole, 2-methyl 5,6dihydroxyindole, 3-methyl 5,6-dihydroxyindole, 2,3-dimethyl 5,6-dihydroxyindole); (ii) plant extracts for artificial coloring of the skin such as: - "insoluble" red wood extracts of the genus Pterocarpus and of the genus Baphia such as Pterocarpus santalinus, Pterocarpus osun, Pterocarpus soyauxii, Pterocarpus erinaceus, Pterocarpus indicus or even Baphia nitida such as those described in patent application EP-A-0 971683; - plant extracts of the genus Saxifraga such as Saxifraga cuneifolia, Saxifraga glaucescens, Saxifraga rotundifolia, Saxifraga granulata, Saxifraga bulbifera, Saxifraga umbrosa, Saxifraga tridactylites; - Sorghum plant extracts such as Sorghum caudatum,
Sorghum Bicolor.
Au sens de la présente invention, on entendra, par agent de coloration artificielle de la peau , toute substance ayant une affinité particulière pour la peau lui permettant de conférer à cette dernière une coloration durable qui ne s'élimine ni à l'eau ni à l'aide d'un solvant, et qui résiste à la fois au frottement et au lavage par une solution contenant des tensioactifs. Une telle coloration durable se distingue donc de la coloration superficielle et momentanée apportée par exemple par un produit de maquillage. For the purposes of the present invention, the term “artificial coloring agent for the skin” will be understood to mean any substance having a particular affinity for the skin allowing it to impart to the latter a lasting coloring which is not eliminated with water or using a solvent, and which resists both rubbing and washing with a solution containing surfactants. Such long-lasting coloring therefore differs from the surface and temporary coloring provided for example by a makeup product.
Les agents autobronzants conformes à l'invention sont généralement présents dans les compositions dans des proportions allant de 0,1 à 10% en poids par rapport au poids total de la composition, et de préférence de 0,2 à 8% en poids par rapport au poids total de la composition Les compositions peuvent également contenir en plus au moins un filtre UV organique et/ou au moins un filtre UV inorganique actifs dans l'UVA et/ou l'UVB ; lesdits filtres UV pouvant être hydrosolubles, liposolubles ou bien insolubles dans les solvants cosmétiques couramment utilisés. The self-tanning agents according to the invention are generally present in the compositions in proportions ranging from 0.1 to 10% by weight relative to the total weight of the composition, and preferably from 0.2 to 8% by weight relative to the total weight of the composition The compositions can also additionally contain at least one organic UV filter and / or at least one inorganic UV filter active in UVA and / or UVB; said UV filters may be water-soluble, liposoluble or even insoluble in the cosmetic solvents commonly used.
Les filtres UV organiques sont notamment choisis parmi les anthranilates ; les dérivés cinnamiques ; les dérivés salicyliques, les dérivés du camphre ; les dérivés de triazine tels que ceux décrits dans les demandes de brevet US 4367390, EP863145, EP517104, EP570838, EP796851, EP775698, EP878469 et EP933376 ; les dérivés de la benzophénone ; les dérivés de ss,ss'-diphénylacrylate ; les dérivés de benzotriazole ; les dérivés de benzalmalonate ; les dérivés de benzimidazole ; les imadazolines ; les dérivés bis-benzoazolyle tels que décrits dans les brevets EP669323 et US 2,463,264; les dérivés de l'acide p-aminobenzoïque (PABA) ; les dérivés de méthylène bis-(hydroxyphényl benzotriazole) tels que décrits dans s les demandes US5,237,071, US5,166,355, GB2303549, DE 197 26 184 et EP893119 ; les polymères filtres et silicones filtres tels que ceux décrits notamment dans la demande WO-93/04665 ; les dimères dérivés d'a-alkylstyrène tels que ceux décrits dans la demande de brevet DE19855649 ; les 4,4-diarylbutadiènes tels que décrits dans les demandes EP0967200, DE19746654, DE19755649, EP-A-1008586 ; les Organic UV filters are chosen in particular from anthranilates; cinnamic derivatives; salicylic derivatives, camphor derivatives; triazine derivatives such as those described in patent applications US 4367390, EP863145, EP517104, EP570838, EP796851, EP775698, EP878469 and EP933376; benzophenone derivatives; derivatives of ss, ss'-diphenylacrylate; benzotriazole derivatives; benzalmalonate derivatives; benzimidazole derivatives; imadazolines; bis-benzoazolyl derivatives as described in patents EP669323 and US 2,463,264; p-aminobenzoic acid derivatives (PABA); derivatives of methylene bis- (hydroxyphenyl benzotriazole) as described in s applications US5,237,071, US5,166,355, GB2303549, DE 197 26 184 and EP893119; filter polymers and filter silicones such as those described in particular in application WO-93/04665; dimers derived from α-alkylstyrene such as those described in patent application DE19855649; 4,4-diarylbutadienes as described in applications EP0967200, DE19746654, DE19755649, EP-A-1008586; the
<Desc/Clms Page number 5><Desc / Clms Page number 5>
dérivés de 2-hydroxybenzophénone aminosubstitués dans les demandes de brevet EP-A-1046391 et DE100 12 408 et leurs mélanges. derivatives of amino-substituted 2-hydroxybenzophenone in patent applications EP-A-1046391 and DE100 12 408 and their mixtures.
Comme exemples de filtres organiques actifs dans l'UV-A et/ou l'UV-B, on peut citer désignés ci-dessus sous leur nom INCI : Dérivés de l'acide para-aminobenzoique : - PABA, - Ethyl PABA, - Ethyl Dihydroxypropyl PABA, - Ethylhexyl Diméthyl PABA vendu notamment sous le nom ESCALOL 507 par
ISP, - Glyceryl PABA, - PEG-25 PABA vendu sous le nom UVINUL P25 par BASF, Dérivés salicyliques : - Homosalate vendu sous le nom EUSOLEX HMS par RONA/EM INDUSTRIES, - Ethylhexyl Salicylate vendu sous le nom NEO HELIOPAN OS par
HAARMANN et REIMER, - Dipropyleneglycol Salicylate vendu sous le nom DIPSAL par SCHER, - TEA Salicylate, vendu sous le nom NEO HELIOPAN TS par HAARMANN et
REIMER, Dérivés du dibenzovlméthane : - Butyl Methoxydibenzoylmethane vendu notamment sous le nom commercial PARSOL 1789 par HOFFMANN LA ROCHE, - Isopropyl Dibenzoylmethane, Dérivés cinnamiques: - Ethylhexyl Methoxycinnamate vendu notamment sous le nom commercial PARSOL MCX par HOFFMANN LA ROCHE, - Isopropyl Methoxy cinnamate, - Isoamyl Methoxy cinnamate vendu sous le nom commercial NEO HELIOPAN E
1000 par HAARMANN et REIMER, - Cinoxate, - DEA Methoxycinnamate, - - Diisopropyl Methylcinnamate, - Glyceryl Ethylhexanoate Dimethoxycinnamate Dérivés de ss,ss'-diphénylacrylate : - Octocrylene vendu notamment sous le nom commercial UVINUL N539 par
BASF, - Etocrylene, vendu notamment sous le nom commercial UVINUL N35 par
BASF, Dérivés de la benzophénone : - Benzophenone-1 vendu sous le nom commercial UVINUL 400 par BASF, - Benzophenone-2 vendu sous le nom commercial UVINUL D50 par BASF - Benzophenone-3 ou Oxybenzone, vendu sous le nom commercial UVINUL
M40 par BASF, As examples of organic filters active in UV-A and / or UV-B, there may be mentioned designated above under their name INCI: Derivatives of para-aminobenzoic acid: - PABA, - Ethyl PABA, - Ethyl Dihydroxypropyl PABA, - Ethylhexyl Dimethyl PABA sold in particular under the name ESCALOL 507 by
ISP, - Glyceryl PABA, - PEG-25 PABA sold under the name UVINUL P25 by BASF, Salicylic derivatives: - Homosalate sold under the name EUSOLEX HMS by RONA / EM INDUSTRIES, - Ethylhexyl Salicylate sold under the name NEO HELIOPAN OS by
HAARMANN and REIMER, - Dipropyleneglycol Salicylate sold under the name DIPSAL by SCHER, - TEA Salicylate, sold under the name NEO HELIOPAN TS by HAARMANN and
REIMER, Dibenzovlmethane derivatives: - Butyl Methoxydibenzoylmethane sold in particular under the trade name PARSOL 1789 by HOFFMANN LA ROCHE, - Isopropyl Dibenzoylmethane, Cinnamic derivatives: - Ethylhexyl Methoxycinnamate sold in particular under the trade name PARSOL MCXOCHEMANN, HOFFMANN, - Isoamyl Methoxy cinnamate sold under the trade name NEO HELIOPAN E
1000 by HAARMANN and REIMER, - Cinoxate, - DEA Methoxycinnamate, - - Diisopropyl Methylcinnamate, - Glyceryl Ethylhexanoate Dimethoxycinnamate Derivatives of ss, ss'-diphenylacrylate: - Octocrylene sold in particular under the trade name UVINUL N539
BASF, - Etocrylene, sold in particular under the trade name UVINUL N35 by
BASF, Benzophenone derivatives: - Benzophenone-1 sold under the trade name UVINUL 400 by BASF, - Benzophenone-2 sold under the trade name UVINUL D50 by BASF - Benzophenone-3 or Oxybenzone, sold under the trade name UVINUL
M40 by BASF,
<Desc/Clms Page number 6><Desc / Clms Page number 6>
- Benzophenone-4 vendu sous le nom commercial UVINUL MS40 par BASF, - Benzophenone-5 - Benzophenone-6 vendu sous le nom commercial HELISORB 11 par
NORQUAY - Benzophenone-8 vendu sous le nom commercial SPECTRA-SORB UV-24
PAR AMERICAN CYANAMID - Benzophenone-9 vendu sous le nom commercial UVINUL DS-49 par BASF, - Benzophenone-12 - le 2-(4-diéthylamino-2-hydroxybenzoyl)-benzoate de n-hexyle Dérivé du benzylidène camphre : - 3-Benzylidene camphor fabriqué sous le nom MEXORYL SD par CHIMEX, - 4-Methylbenzylidene camphor vendu sous le nom EUSOLEX 6300 par
MERCK, - Benzylidene Camphor Sulfonic Acid fabriqué sous le nom MEXORYL SL par
CHIMEX, - Camphor Benzalkonium Methosulfate fabriqué sous le nom MEXORYL SO par
CHIMEX, - -Terephthalylidene Dicamphor Sulfonic Acid fabriqué sous le nom MEXORYL
SX par CHIMEX, - Polyacrylamidomethyl Benzylidene Camphor fabriqué sous le nom MESORYL
SW par CHIMEX, Dérivés du phenyl benzimidazole : - Phenylbenzimidazole Sulfonic Acid vendu notamment sous le nom commercial EUSOLEX 232 par MERCK, - Benzimidazilate vendu sous le nom commercial commercial NEO HELIOPAN
AP par HAARMANN et REIMER, Dérivés de la triazine : - Anisotriazine vendu sous le nom commercial TINOSORB S par CIBA GEIGY, - Ethylhexyl triazone vendu notamment sous le nom commercial UVINUL T150 par BASF, - Diethylhexyl Butamido Triazone vendu sous le nom commercial UVASORB
HEB par SIGMA 3V, - la 2,4,6-tris-(4'-amino benzalmalonate de diisobutyle)-s- triazine. - Benzophenone-4 sold under the trade name UVINUL MS40 by BASF, - Benzophenone-5 - Benzophenone-6 sold under the trade name HELISORB 11 by
NORQUAY - Benzophenone-8 sold under the trade name SPECTRA-SORB UV-24
BY AMERICAN CYANAMID - Benzophenone-9 sold under the trade name UVINUL DS-49 by BASF, - Benzophenone-12 - 2- (4-diethylamino-2-hydroxybenzoyl) -n-hexyl benzoate Benzylidene camphor derivative: - 3- Benzylidene camphor manufactured under the name MEXORYL SD by CHIMEX, - 4-Methylbenzylidene camphor sold under the name EUSOLEX 6300 by
MERCK, - Benzylidene Camphor Sulfonic Acid manufactured under the name MEXORYL SL by
CHIMEX, - Camphor Benzalkonium Methosulfate manufactured under the name MEXORYL SO by
CHIMEX, - -Terephthalylidene Dicamphor Sulfonic Acid manufactured under the name MEXORYL
SX by CHIMEX, - Polyacrylamidomethyl Benzylidene Camphor manufactured under the name MESORYL
SW by CHIMEX, Phenyl benzimidazole derivatives: - Phenylbenzimidazole Sulfonic Acid sold in particular under the trade name EUSOLEX 232 by MERCK, - Benzimidazilate sold under the trade name NEO HELIOPAN
AP by HAARMANN and REIMER, Triazine derivatives: - Anisotriazine sold under the trade name TINOSORB S by CIBA GEIGY, - Ethylhexyl triazone sold in particular under the trade name UVINUL T150 by BASF, - Diethylhexyl Butamido Triazone sold under the trade name UVASORB
HEB by SIGMA 3V, - 2,4,6-tris- (4'-amino benzalmalonate diisobutyl) -s-triazine.
Dérivés duphenyl benzotriazole : - Drometrizole Trisiloxane vendu sous le nom SILATRIZOLE par RHODIA
CHIMIE , - Methylène bis-Benzotriazolyl Tetramethylbutylphénol, vendu sous forme solide sous le nom commercial MIXXIM BB/100 par FAIRMOUNT CHEMICAL ou sous forme micronisé en dispersion aqueuse sous le nom commercial TINOSORB M par CIBA SPECIALTY CHEMICALS, Dérivés anthraniliques : - Menthyl anthranilate vendu sous le nom commercial commercial NEO
HELIOPAN MA par HAARMANN et REIMER, Dérivés d'imidazolines : Dphenyl benzotriazole derivatives: - Drometrizole Trisiloxane sold under the name SILATRIZOLE by RHODIA
CHEMISTRY, - Methylene bis-Benzotriazolyl Tetramethylbutylphenol, sold in solid form under the trade name MIXXIM BB / 100 by FAIRMOUNT CHEMICAL or in micronized form in aqueous dispersion under the trade name TINOSORB M by CIBA SPECIALTY CHEMICALS, Anthranilic derivatives: - Menthyl anthranilate sold under the trade name NEO
HELIOPAN MA by HAARMANN and REIMER, Imidazoline derivatives:
<Desc/Clms Page number 7><Desc / Clms Page number 7>
- Ethylhexyl Dimethoxybenzylidene Dioxoimidazoline Propionate, Dérivés du benzalmalonate : - Polyorganosiloxane à fonctions benzalmalonate vendu sous la dénomination commerciale PARSOL SLX par HOFFMANN LA ROCHE et leurs mélanges. - Ethylhexyl Dimethoxybenzylidene Dioxoimidazoline Propionate, Benzalmalonate derivatives: - Polyorganosiloxane with benzalmalonate functions sold under the trade name PARSOL SLX by HOFFMANN LA ROCHE and their mixtures.
Les filtres UV organiques plus particulièrement préférés sont choisis parmi les composés suivants : - Ethylhexyl Salicylate, - Butyl Methoxydibenzoylmethane, - Ethylhexyl Methoxycinnamate, - Octocrylene, - Phenylbenzimidazole Sulfonic Acid, - Terephthalylidene Dicamphor Sulfonic,. The more particularly preferred organic UV filters are chosen from the following compounds: - Ethylhexyl Salicylate, - Butyl Methoxydibenzoylmethane, - Ethylhexyl Methoxycinnamate, - Octocrylene, - Phenylbenzimidazole Sulfonic Acid, - Terephthalylidene Dicamphor Sulfonic ,.
- Benzophenone-3, - Benzophenone-4, - Benzophenone-5, - 2-(4-diéthylamino-2-hydroxybenzoyl)-benzoate de n-hexyle - 4-Methylbenzylidene camphor, - Benzimidazilate, - Anisotriazine, - Ethylhexyl triazone, - Diethylhexyl Butamido Triazone, - la 2,4,6-tris-(4'-amino benzalmalonate de diisobutyle)-s- triazine. - Benzophenone-3, - Benzophenone-4, - Benzophenone-5, - 2- (4-diethylamino-2-hydroxybenzoyl) -n-hexyl benzoate - 4-Methylbenzylidene camphor, - Benzimidazilate, - Anisotriazine, - Ethylhexyl triazone, - Diethylhexyl Butamido Triazone, - 2,4,6-tris- (4'-amino diisobutyl benzalmalonate) -s- triazine.
- Methylene bis-Benzotriazolyl Tetramethylbutylphenol, - Drometrizole Trisiloxane, et leurs mélanges. - Methylene bis-Benzotriazolyl Tetramethylbutylphenol, - Drometrizole Trisiloxane, and mixtures thereof.
Les filtres UV inorganiques sont généralement choisis parmi des pigments ou bien encore des nanopigments (taille moyenne des particules primaires : entre 5 nm et 100 nm, de préférence entre 10 nm et 50 nm) d'oxydes métalliques enrobés ou non comme par exemple des nanopigments d'oxyde de titane (amorphe ou cristallisé sous forme rutile et/ou anatase), de fer, de zinc, de zirconium ou de cérium qui sont tous des agents photoprotecteurs UV bien connus en soi. Des agents d'enrobage classiques sont par ailleurs l'alumine et/ou le stéarate d'aluminium. De tels nanopigments d'oxydes métalliques, enrobés ou non enrobés, sont en particulier décrits dans les demandes de brevets EP-A-0518772 et EP-A-0518773. The inorganic UV filters are generally chosen from pigments or else nanopigments (average size of the primary particles: between 5 nm and 100 nm, preferably between 10 nm and 50 nm) of metallic oxides coated or not, such as nanopigments for example titanium oxide (amorphous or crystallized in rutile form and / or anatase), iron, zinc, zirconium or cerium which are all UV photoprotective agents well known per se. Conventional coating agents are moreover alumina and / or aluminum stearate. Such metal oxide nanopigments, coated or uncoated, are in particular described in patent applications EP-A-0518772 and EP-A-0518773.
Les filtres UV conformes à l'invention sont généralement présents dans les compositions selon l'invention dans des proportions allant de 0,1 à 20% en poids par rapport au poids total de la composition, et de préférence allant de 0,2 à 15% en poids par rapport au poids total de la composition. The UV filters in accordance with the invention are generally present in the compositions according to the invention in proportions ranging from 0.1 to 20% by weight relative to the total weight of the composition, and preferably ranging from 0.2 to 15 % by weight relative to the total weight of the composition.
Les compositions conformes à la présente invention peuvent comprendre en outre des adjuvants cosmétiques classiques notamment choisis parmi les corps gras, les solvants organiques, les épaississants ioniques ou non ioniques, les adoucissants, les antioxydants, les agents anti radicaux libres, les opacifiants, les stabilisants, les émollients, les silicones, les a-hydroxyacides, les agents anti-mousse, les agents hydratants, les vitamines, les agents répulsifs contre les insectes, les parfums, les The compositions in accordance with the present invention may also comprise conventional cosmetic adjuvants in particular chosen from fatty substances, organic solvents, ionic or nonionic thickeners, softeners, antioxidants, anti-free radical agents, opacifiers, stabilizers , emollients, silicones, α-hydroxy acids, defoamers, moisturizers, vitamins, insect repellents, perfumes,
<Desc/Clms Page number 8><Desc / Clms Page number 8>
conservateurs, les tensioactifs, les anti-inflammatoires, les antagonistes de substance P, les charges, les polymères, les propulseurs, les agents alcalinisants ou acidifiants, les colorants ou tout autre ingrédient habituellement utilisé en cosmétique et/ou dermatologique, en particulier pour la fabrication de compositions sous forme d'émulsions. preservatives, surfactants, anti-inflammatories, substance P antagonists, fillers, polymers, propellants, basifying or acidifying agents, dyes or any other ingredient usually used in cosmetics and / or dermatology, in particular for the manufacture of compositions in the form of emulsions.
Les corps gras peuvent être constitués par une huile ou une cire ou leurs mélanges. The fatty substances can consist of an oil or a wax or their mixtures.
Par huile, on entend un composé liquide à température ambiante. Par cire, on entend un composé solide ou substantiellement solide à température ambiante, et dont le point de fusion est généralement supérieur à 35 C. By oil is meant a compound which is liquid at room temperature. The term “wax” is intended to mean a compound which is solid or substantially solid at room temperature, and the melting point of which is generally greater than 35 C.
Comme huiles, on peut citer les huiles minérales (paraffine); végétales (huile d'amande douce, de macadamia, de pépin de cassis, de jojoba) ; synthétiques comme le perhydrosqualène, les alcools, les acides ou les esters gras (comme le benzoate d'alcools en C12-C15 vendu sous la dénomination commerciale Finsolv TN par la société Finetex, le palmitate d'octyle, le lanolate d'isopropyle, les triglycérides dont ceux des acides caprique/caprylique), les esters et éthers gras oxyéthylénés ou oxypropylénés; siliconées (cyclométhicone, polydiméthysiloxanes ou PDMS) ou fluorées, les polyalkylènes. As oils, mention may be made of mineral oils (paraffin); vegetable (sweet almond oil, macadamia oil, blackcurrant seed, jojoba); synthetics such as perhydrosqualene, alcohols, fatty acids or esters (such as the benzoate of C12-C15 alcohols sold under the trade name Finsolv TN by the company Finetex, octyl palmitate, isopropyl lanolate, triglycerides including those of capric / caprylic acids), oxyethylenated or oxypropylenated fatty esters and ethers; silicone (cyclomethicone, polydimethysiloxanes or PDMS) or fluorinated, polyalkylenes.
Comme composés cireux, on peut citer la paraffine, la cire de carnauba, la cire d'abeille, l'huile de ricin hydrogénée. As waxy compounds, there may be mentioned paraffin, carnauba wax, beeswax, hydrogenated castor oil.
Parmi les solvants organiques, on peut citer les alcools et polyols inférieurs. Among the organic solvents, mention may be made of lower alcohols and polyols.
Les épaississants peuvent être choisis notamment parmi les acides polyacryliques réticulés ; les gommes de xanthane ; les gommes de guar telles que la gomme de guar hydroxypropylée ; les celluloses modifiées ou non telles que la méthylhydroxyéthylcellulose et l'hydroxypropylméthyl cellulose ; les polyacrylamides comme le Polyacrylamide (and) C13-C14-Isoparaffine (and) Laureth-7 vendu sous le nom commercial SEPIGEL 305 par la Société SEPPIC ; les polymères d'acide acrylamidomethylpropane sulfonique (AMPS) comme le Sodium Polyacryloyldimthyl Taurate (and) Polysorbate 80 (and) Sorbitan Oleate vendu sous le nom commercial SIMULGEL 800 par la Société SEPPIC ou le Sodium Acrylate/Sodium Acryloyldiméthyl Taurate (and) Isohexadecane (and) Polysorbate 80 vendu sous le nom commercial SIMULGEL EG par la Société SEPPIC. The thickeners can be chosen in particular from crosslinked polyacrylic acids; xanthan gums; guar gums such as hydroxypropylated guar gum; modified or unmodified celluloses such as methylhydroxyethylcellulose and hydroxypropylmethyl cellulose; polyacrylamides such as Polyacrylamide (and) C13-C14-Isoparaffin (and) Laureth-7 sold under the trade name SEPIGEL 305 by the company SEPPIC; polymers of acrylamidomethylpropane sulfonic acid (AMPS) such as Sodium Polyacryloyldimthyl Taurate (and) Polysorbate 80 (and) Sorbitan Oleate sold under the trade name SIMULGEL 800 by the company SEPPIC or Sodium Acrylate / Sodium Acryloyldimethyl Taurate (and) Isohexadecane ) Polysorbate 80 sold under the trade name SIMULGEL EG by the company SEPPIC.
Parmi les colorants additionnels, on citera plus particulièrement le caramel. Among the additional dyes, mention will be made more particularly of caramel.
Bien entendu, l'homme de l'art veillera à choisir le ou les éventuels composés complémentaires cités ci-dessus et/ou leurs quantités de manière telle que les propriétés avantageuses attachées intrinsèquement à l'invention ne soient pas, ou substantiellement pas, altérées par la ou les adjonctions envisagées. Of course, those skilled in the art will take care to choose the optional compound (s) mentioned above and / or their quantities in such a way that the advantageous properties intrinsically attached to the invention are not, or not substantially, altered. by the addition (s) envisaged.
Les compositions selon l'invention peuvent être préparées selon les techniques bien connues de l'homme de l'art, en particulier celles destinées à la préparation d'émulsions de type huile-dans-eau ou eau-dans-huile. The compositions according to the invention can be prepared according to techniques well known to those skilled in the art, in particular those intended for the preparation of emulsions of the oil-in-water or water-in-oil type.
Cette composition peut se présenter en particulier sous forme d'émulsion, simple ou complexe (H/E, E/H, H/E/H ou E/H/E) telle qu'une crème, un lait, ou sous la forme This composition can be in particular in the form of an emulsion, simple or complex (O / W, W / O, O / W / O or W / O / W) such as a cream, a milk, or in the form
<Desc/Clms Page number 9><Desc / Clms Page number 9>
d'un gel ou d'un gel crème, sous la forme d'une lotion, de poudre, de bâtonnet solide et éventuellement être conditionnée en aérosol et se présenter sous forme de mousse ou de spray. a gel or a cream gel, in the form of a lotion, powder, solid stick and possibly be packaged in an aerosol and be in the form of foam or spray.
De préférence, les compositions selon l'invention se présentent sous la forme d'une émulsion huile-dans-eau ou eau-dans huile. Preferably, the compositions according to the invention are in the form of an oil-in-water or water-in-oil emulsion.
Lorsqu'il s'agit d'une émulsion, la phase aqueuse de celle-ci peut comprendre une dispersion vésiculaire non ionique préparée selon des procédés connus (Bangham, Standish and Watkins. J. Mol. Biol. 13, 238 (1965), FR 2 315 991 et FR 2 416 008). When it is an emulsion, the aqueous phase thereof may comprise a nonionic vesicular dispersion prepared according to known methods (Bangham, Standish and Watkins. J. Mol. Biol. 13, 238 (1965), FR 2 315 991 and FR 2 416 008).
L'invention concerne également un procédé de traitement cosmétique pour bronzer et/ou brunir artificiellement la peau, caractérisé par le fait qu'il consiste à appliquer sur celle-ci une quantité efficace d'une composition telle que définie ci-dessus. The invention also relates to a cosmetic treatment method for artificially tanning and / or browning the skin, characterized in that it consists in applying to the latter an effective amount of a composition as defined above.
Des exemples concrets, mais nullement limitatifs, illustrant l'invention, vont maintenant être donnés. Concrete, but in no way limiting, examples illustrating the invention will now be given.
EXEMPLES Exemple 1 : Crème autobronzante rouge - Mélange de monostéarate de glycéryle et de PEG-100-stéarate (SIMULSOL 165-SEPPIC) 1,75% en poids - DC 580 (Dow Corning) 1,0% en poids - Dimethicone 1 % en poids - Silicone volatile 8% en poids - Diméthiconol 0,7% en poids - Gomme de Xanthane 0,2% en poids - Glycérine 4% en poids - Propylène glycol 4% en poids - Dihydroxyacétone 5% en poids - CI 45380 / Red 21 (LCW) 0,01 % en poids - Sodium Acrylate/Sodium Acryloyldiméthyl taurate (and) Isohexadecane (and) Polysorbate 80 (Simulgel EG - SEPPIC) 2 % en poids - Conservateurs qs - Eau purifiée qsp 100% en poids EXAMPLES Example 1: Red self-tanning cream - Mixture of glyceryl monostearate and PEG-100-stearate (SIMULSOL 165-SEPPIC) 1.75% by weight - DC 580 (Dow Corning) 1.0% by weight - Dimethicone 1% by weight weight - Volatile silicone 8% by weight - Dimethiconol 0.7% by weight - Xanthan gum 0.2% by weight - Glycerin 4% by weight - Propylene glycol 4% by weight - Dihydroxyacetone 5% by weight - CI 45380 / Red 21 (LCW) 0.01% by weight - Sodium Acrylate / Sodium Acryloyldimethyl taurate (and) Isohexadecane (and) Polysorbate 80 (Simulgel EG - SEPPIC) 2% by weight - Preservatives qs - Purified water qs 100% by weight
<Desc/Clms Page number 10><Desc / Clms Page number 10>
Exemple 2 : Crème autobronzante rouge : - Mélange de monostéarate de glycéryle et de PEG-100-stéarate (SIMULSOL 165-SEPPIC) 1,75% en poids - DC 580 (Dow Corning) 1,0% en poids - Octocrylène 5% en poids - Octyl Méthoxycinnamate 5% en poids - Benzophénone-3 2% en poids - Diméthicone 1 % en poids - Silicone volatile 8% en poids - Diméthiconol 0,7% en poids - Gomme de Xanthane 0,2% en poids - Glycérine 4% en poids - Propylène glycol 4% en poids - Dihydroxyacétone 5% en poids - CI 45380 / Red 22 (LCW) 0,01 % en poids - Sodium Acrylate/Sodium Acryloyldiméthyl Taurate (and) Isohexadecane (and) Polysorbate 80 (Simulgel EG - SEPPIC) 2 % en poids - Conservateurs qs - Eau purifiée qsp 100% en poids Example 2: Red self-tanning cream: - Mixture of glyceryl monostearate and PEG-100-stearate (SIMULSOL 165-SEPPIC) 1.75% by weight - DC 580 (Dow Corning) 1.0% by weight - Octocrylene 5% weight - Octyl Methoxycinnamate 5% by weight - Benzophenone-3 2% by weight - Dimethicone 1% by weight - Volatile silicone 8% by weight - Dimethiconol 0.7% by weight - Xanthan gum 0.2% by weight - Glycerin 4 % by weight - Propylene glycol 4% by weight - Dihydroxyacetone 5% by weight - CI 45380 / Red 22 (LCW) 0.01% by weight - Sodium Acrylate / Sodium Acryloyldimethyl Taurate (and) Isohexadecane (and) Polysorbate 80 (Simulgel EG - SEPPIC) 2% by weight - Preservatives qs - Purified water qs 100% by weight
<Desc/Clms Page number 11><Desc / Clms Page number 11>
Exemple 3 : Crème autobronzante rouge : - Mélange d'alkyl polyglucosides et d'alcools gras (Montanov 202- SEPPIC) 3% en poids - Mélange de monostéarate de glycéryle et de PEG-100-stéarate (SIMULSOL 165-SEPPIC) 2% en poids - Isohexadecane 4% en poids - Benzoate d'alcools en C12-15 (Finsolv TN- Finetex) 4% en poids - Tri-Citrate d'alcool en C14-15 - (Cosmacol ECL- Condea Augusta) 3% en poids - Dimethicone 1% en poids - Octyl Methoxycinnamate 7.5% en poids - Drometrizole trisiloxane 1.5% en poids - CI 45380 / Red 21 (LCW) 0,05% en poids - Terephthalylidene dicamphor sulfonic acid 0.5% en poids - Glycerine 4% en poids - Propylène glycol 6% en poids - Dihydroxyacétone 5,0% en poids - Talc 1 % en poids - Amidon de maïs réticulé 1 % en poids - Ethanol 4,5% en poids - Polyacrylamide (and) C13-C14-Isoparaffine (and) Laureth-7 (Sepigel 305 - SEPPIC) 1,5% en poids - Soude qs - Conservateurs qs - Eau purifiée qsp 100% en poids Example 3: Red self-tanning cream: - Mixture of alkyl polyglucosides and fatty alcohols (Montanov 202- SEPPIC) 3% by weight - Mixture of glyceryl monostearate and PEG-100-stearate (SIMULSOL 165-SEPPIC) 2% weight - Isohexadecane 4% by weight - Benzoate of alcohols in C12-15 (Finsolv TN- Finetex) 4% by weight - Tri-Citrate of alcohol in C14-15 - (Cosmacol ECL- Condea Augusta) 3% by weight - Dimethicone 1% by weight - Octyl Methoxycinnamate 7.5% by weight - Drometrizole trisiloxane 1.5% by weight - CI 45380 / Red 21 (LCW) 0.05% by weight - Terephthalylidene dicamphor sulfonic acid 0.5% by weight - Glycerine 4% by weight - Propylene glycol 6% by weight - Dihydroxyacetone 5.0% by weight - Talc 1% by weight - Cross-linked corn starch 1% by weight - Ethanol 4.5% by weight - Polyacrylamide (and) C13-C14-Isoparaffin (and) Laureth-7 (Sepigel 305 - SEPPIC) 1.5% by weight - Soda qs - Preservatives qs - Purified water qs 100% by weight
<Desc/Clms Page number 12><Desc / Clms Page number 12>
Exemple 4 : Crème autobronzante rouge : - Mélange d'alkyl polyglucosides et d'alcools gras (Montanov 202- SEPPIC) 3% en poids - Mélange de monostéarate de glycéryle et de PEG-100-stéarate (SIMULSOL 165-SEPPIC) 2% en poids - Isohexadecane 4% en poids - Benzoate d'alcools en C 12-15 (Finsolv TN- Finetex) 4% en poids - Tri-Citrate d'alcool en C14-15 (Cosmacol ECL- Condea Augusta) 2% en poids - Dimethicone 1 % en poids - Octyl Methoxycinnamate 8.5% en poids - Drometrizole trisiloxane 1.5% en poids - CI 45380 / D & C Red 21 (LCW) 0,006% en poids - Mica enrobé d'oxyde de titane 0.5% en poids - Mélange tocophérols naturels /huile de soja 0.2% en poids - Caramel à 75% dans l'eau 1.7% en poids - Glycerine 4% en poids - Propylène glycol 4% en poids - Dihydroxyacétone 5,0% en poids - Ethanol 4,5% en poids - Sodium Polyacryloyldimethyl Taurate (and) Polysorbate 80 (and) Sorbitan Oleate (Simulgel 800 - SEPPIC) 2,7% en poids - Conservateurs qs - Eau purifiée qsp 100% en poids Exemple 5 : Crème autobronzante rouge : - Mélange d'alkyl polyglucosides et d'alcools gras (Montanov 68- SEPPIC) 7% en poids - Isohexadecane 4% en poids - Silicone volatile 2% en poids - Tri-Citrate d'alcool en C14-15 (Cosmacol ECL- Condea Augusta) 5% en poids - Dimethicone 1 % en poids - Octyl Methoxycinnamate 7.5% en poids - Benzophenone-3 4% en poids - CI 45380 / Red 21 (LCW) 0,005% en poids - Mica enrobé d'oxyde de titane 3% en poids - Mélange tocophérols naturels /huile de soja 0.2% en poids - Caramel à 75% dans l'eau 1.5% en poids - Glycerine 4% en poids - Propylène glycol 4% en poids - Dihydroxyacétone 5,0% en poids - parfum qs - Conservateurs qs - Eau purifiée qsp 100% en poids Example 4: Red self-tanning cream: - Mixture of alkyl polyglucosides and fatty alcohols (Montanov 202- SEPPIC) 3% by weight - Mixture of glyceryl monostearate and PEG-100-stearate (SIMULSOL 165-SEPPIC) 2% weight - Isohexadecane 4% by weight - Benzoate of C 12-15 alcohols (Finsolv TN- Finetex) 4% by weight - Tri-Citrate of C14-15 alcohol (Cosmacol ECL- Condea Augusta) 2% by weight - Dimethicone 1% by weight - Octyl Methoxycinnamate 8.5% by weight - Drometrizole trisiloxane 1.5% by weight - CI 45380 / D & C Red 21 (LCW) 0.006% by weight - Mica coated with titanium oxide 0.5% by weight - Tocopherol mixture natural / soybean oil 0.2% by weight - Caramel 75% in water 1.7% by weight - Glycerin 4% by weight - Propylene glycol 4% by weight - Dihydroxyacetone 5.0% by weight - Ethanol 4.5% by weight - Sodium Polyacryloyldimethyl Taurate (and) Polysorbate 80 (and) Sorbitan Oleate (Simulgel 800 - SEPPIC) 2.7% by weight - Preservatives qs - Purified water qs 100% by weight Example 5 : Red self-tanning cream: - Mixture of alkyl polyglucosides and fatty alcohols (Montanov 68- SEPPIC) 7% by weight - Isohexadecane 4% by weight - Volatile silicone 2% by weight - Tri-Citrate of alcohol in C14-15 (Cosmacol ECL- Condea Augusta) 5% by weight - Dimethicone 1% by weight - Octyl Methoxycinnamate 7.5% by weight - Benzophenone-3 4% by weight - CI 45380 / Red 21 (LCW) 0.005% by weight - Mica coated with titanium oxide 3% by weight - Natural tocopherol / soybean oil mixture 0.2% by weight - Caramel 75% in water 1.5% by weight - Glycerin 4% by weight - Propylene glycol 4% by weight - Dihydroxyacetone 5.0 % by weight - perfume qs - Preservatives qs - Purified water qs 100% by weight
<Desc/Clms Page number 13><Desc / Clms Page number 13>
Exemple 6 : Crème autobronzante rouge : - Mélange de monostéarate de glycéryle et de PEG-100-stéarate (SIMULSOL 165-SEPPIC) 1,75% en poids - DC 580 (Dow Corning) 1,0% en poids - Octocrylène 5% en poids - Octyl Méthoxycinnamate 5% en poids - Benzophénone-3 2% en poids - Diméthicone 1 % en poids - Silicone volatile 8% en poids - Diméthiconol 0,7% en poids - Gomme de Xanthane 0,2% en poids - Glycérine 4% en poids - Propylène glycol 4% en poids - Dihydroxyacétone 5% en poids - CI 45380 / Red 21 (LCW) 0,01 % en poids - Sodium acrylate/sodium Acryloyldiméthyl taurate et isohexadecane (Simulgel EG - SEPPIC) 2 % en poids - Conservateurs qs - Eau purifiée qsp 100% en poids La composition présente une couleur rouge. Après 1 mois de conservation à température ambiante, on n'observe aucun phénomène de décoloration. Example 6: Red self-tanning cream: - Mixture of glyceryl monostearate and PEG-100-stearate (SIMULSOL 165-SEPPIC) 1.75% by weight - DC 580 (Dow Corning) 1.0% by weight - Octocrylene 5% weight - Octyl Methoxycinnamate 5% by weight - Benzophenone-3 2% by weight - Dimethicone 1% by weight - Volatile silicone 8% by weight - Dimethiconol 0.7% by weight - Xanthan gum 0.2% by weight - Glycerin 4 % by weight - Propylene glycol 4% by weight - Dihydroxyacetone 5% by weight - CI 45380 / Red 21 (LCW) 0.01% by weight - Sodium acrylate / sodium Acryloyldimethyl taurate and isohexadecane (Simulgel EG - SEPPIC) 2% by weight - Preservatives qs - Purified water qs 100% by weight The composition has a red color. After 1 month of storage at room temperature, no discoloration phenomenon is observed.
Exemple 7 (comparatif): Identique à la composition de l'exemple 5 mais on remplace le colorant rouge RED 21 par le colorant hydrosoluble azoïque CI 15985/ YELLOW 6 dans la même concentration. Example 7 (comparative): Identical to the composition of Example 5, but the red dye RED 21 is replaced by the water-soluble azo dye CI 15985 / YELLOW 6 in the same concentration.
La composition présente une couleur orange. Elle se décolore complètement après 1 mois de conservation à température ambiante. The composition has an orange color. It completely discolours after 1 month of storage at room temperature.
Exemple 8 (comparatif): Identique à la composition de l'exemple 4 mais on remplace le colorant rouge RED 21 par le colorant hydrosoluble azoïque CI 16035/ RED 40 dans la même concentration. Example 8 (comparative): Identical to the composition of Example 4, but the red dye RED 21 is replaced by the water-soluble azo dye CI 16035 / RED 40 in the same concentration.
La composition présente une couleur rouge/rose. Elle se décolore complètement après 1 mois de conservation à température ambiante.The composition has a red / pink color. It completely discolours after 1 month of storage at room temperature.
Claims (26)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR0207289A FR2840806B1 (en) | 2002-06-13 | 2002-06-13 | COLOR-SELF-TONING COMPOSITIONS COMPRISING AT LEAST ONE RED OR ORANGE COLOR, CHOSEN FROM FLUORANES OR THEIR ALKALI METAL SALTS |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR0207289A FR2840806B1 (en) | 2002-06-13 | 2002-06-13 | COLOR-SELF-TONING COMPOSITIONS COMPRISING AT LEAST ONE RED OR ORANGE COLOR, CHOSEN FROM FLUORANES OR THEIR ALKALI METAL SALTS |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| FR2840806A1 true FR2840806A1 (en) | 2003-12-19 |
| FR2840806B1 FR2840806B1 (en) | 2005-02-11 |
Family
ID=29595208
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| FR0207289A Expired - Fee Related FR2840806B1 (en) | 2002-06-13 | 2002-06-13 | COLOR-SELF-TONING COMPOSITIONS COMPRISING AT LEAST ONE RED OR ORANGE COLOR, CHOSEN FROM FLUORANES OR THEIR ALKALI METAL SALTS |
Country Status (1)
| Country | Link |
|---|---|
| FR (1) | FR2840806B1 (en) |
Cited By (28)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1671680A1 (en) | 2004-12-20 | 2006-06-21 | L'oreal | Self-tanning composition having two components, multicompartment device and process for colouring the skin |
| EP1754513A2 (en) | 2005-08-17 | 2007-02-21 | L'oreal | Use of 8-hexadecene-1,16-dicarboxylic acid as a care agent for promoting the horny layer cohesion |
| WO2007060021A1 (en) * | 2005-11-28 | 2007-05-31 | Johnson & Johnson Consumer France S.A.S. | Self-tanning composition |
| WO2007099434A3 (en) * | 2006-02-28 | 2007-11-22 | Chanel Parfums Beaute | Autobronzing composition |
| EP2011481A2 (en) | 2007-07-06 | 2009-01-07 | L'Oreal | Sun-protection composition containing the association of a semi-crystalline polymer and hollow latex particles |
| EP2014277A1 (en) | 2007-07-12 | 2009-01-14 | L'Oreal | Aqueous liquid photoprotective composition based on a polyamide polymer with tertiary amide termination |
| EP2119429A1 (en) | 2008-05-14 | 2009-11-18 | L'Oréal | Cosmetic composition containing a dibenzoylmethane derivative and a pyrrolidinone derivative; method for photostabilising the dibenzoylmethane derivative |
| WO2009016017A3 (en) * | 2007-07-31 | 2010-02-25 | Henkel Ag & Co. Kgaa | Stable self-tanning compositions in emulsion form |
| EP2174644A1 (en) | 2008-10-08 | 2010-04-14 | L'oreal | Cosmetic composition comprising a dibenzoylmethane derivative and a dithiolane; Photostabilization of a dibenzoylmethane derivative |
| WO2011023886A2 (en) | 2009-08-28 | 2011-03-03 | L'oreal | Composition containing at least one lipophilic 2-hydroxybenzophenone filter and a silicon s-triazine substituted by at least two alkylaminobenzoate groups |
| WO2011042358A1 (en) | 2009-10-08 | 2011-04-14 | L'oreal | Photoprotective composition based on a 2-alkoxy-4-alkyl ketone phenol compound; use of said compound for increasing the sun protection factor |
| WO2011073279A2 (en) | 2009-12-18 | 2011-06-23 | L'oreal | Cosmetic treatment method involving a compound capable of condensing in situ |
| WO2011080043A1 (en) | 2010-01-04 | 2011-07-07 | L'oreal | Ureidopyrimidones; cosmetic use as uv-screening agents; antisun compositions containing them |
| WO2011113719A2 (en) | 2010-03-15 | 2011-09-22 | L'oreal | Composition comprising a dibenzoylmethane screening agent and a merocyanine dicyano or cyanoacetate derivative; method for the photostabilization of the dibenzoylmethane screening agent |
| WO2011113718A1 (en) | 2010-03-15 | 2011-09-22 | L'oreal | Composition containing a dibenzoylmethane screening agent and a hydrophilic or water-soluble merocyanin uv-screening agent; process for photostabilizing the dibenzoylmethane screening agent |
| WO2011151203A2 (en) | 2010-06-03 | 2011-12-08 | L'oreal | Cosmetic treatment process using a coating based on a copolymer containing polyamide blocks and polyether blocks |
| WO2012010554A1 (en) | 2010-07-20 | 2012-01-26 | L'oreal | Use of compounds resulting from a sugar polyol or from a dehydrogenated sugar polyol derivative as a uv-screening agent; antisun compositions containing same; and novel compounds |
| WO2012059348A1 (en) | 2010-11-05 | 2012-05-10 | L'oreal | Fluid aqueous antisun composition based on a superabsorbent polymer and a crosslinked copolymer of methacrylic acid and of a c1-c4 alkyl acrylate |
| WO2012105723A1 (en) | 2011-02-04 | 2012-08-09 | L'oreal | Composite pigment and method for preparation thereof |
| WO2012119860A1 (en) | 2011-03-09 | 2012-09-13 | L'oreal | Cosmetic composition containing a dibenzoylmethane derivative and a monoester-monoamide compound of methylsuccinic acid; process for photostabilizing a dibenzoylmethane derivative |
| WO2013004777A1 (en) | 2011-07-07 | 2013-01-10 | L'oreal | Photoprotective composition |
| WO2013010590A1 (en) | 2011-07-21 | 2013-01-24 | L'oreal | Cosmetic and/or dermatological composition containing a merocyanine derivative comprising specific polar groups consisting of hydroxyl- and ether-functionalities |
| CN102898858A (en) * | 2012-10-19 | 2013-01-30 | 中国乐凯胶片集团公司 | Preparation method of 3', 6'-dimethoxy fluorane yellow thermosensitive dye |
| WO2013068237A1 (en) | 2011-11-07 | 2013-05-16 | L'oreal | Composition with a continuous oil phase containing at least one lipophilic organic uv-screening agent and hydrophobic silica aerogel particles |
| US9452125B2 (en) | 2011-04-20 | 2016-09-27 | Kao Usa Inc. | Self-tanning compositions having reduced maillard reaction malodor |
| CN110603025A (en) * | 2017-08-30 | 2019-12-20 | 上海百雀羚生物科技有限公司 | Cosmetic composition with antioxidant properties |
| US11266584B2 (en) | 2012-07-13 | 2022-03-08 | L'oreal | Cosmetic composition comprising composite sunscreen particles |
| US11523976B2 (en) | 2012-07-13 | 2022-12-13 | L'oreal | Composite pigment and method for preparing the same |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0627214A1 (en) * | 1993-06-04 | 1994-12-07 | MERCK PATENT GmbH | Skin dyeing composition |
| FR2712804A1 (en) * | 1993-11-25 | 1995-06-02 | Merck Patent Gmbh | Skin colouring compsn. for durable, high intensity natural tan tones |
| WO1998023256A1 (en) * | 1996-11-25 | 1998-06-04 | Schering-Plough Healthcare Products, Inc. | Sunless tanning emulsions with disappearing color indicator |
| EP1060738A2 (en) * | 1999-06-15 | 2000-12-20 | Neutrogena Corporation | Self-tanning composition comprising carmine |
| WO2001012150A1 (en) * | 1999-08-13 | 2001-02-22 | Unilever Plc | Cosmetic compositions with thermochromic color change substances and an agent for inducing a temperature change |
| US6231837B1 (en) * | 1997-06-06 | 2001-05-15 | Schering-Plough Healthcare Products, Inc. | Self-tanning dihydroxyacetone formulations having improved stability and providing enhanced delivery |
| EP1277461A2 (en) * | 2001-07-20 | 2003-01-22 | Beiersdorf AG | Cosmetic compositions with triazines as sunscreening agents and dihydroxyacetone |
-
2002
- 2002-06-13 FR FR0207289A patent/FR2840806B1/en not_active Expired - Fee Related
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0627214A1 (en) * | 1993-06-04 | 1994-12-07 | MERCK PATENT GmbH | Skin dyeing composition |
| FR2712804A1 (en) * | 1993-11-25 | 1995-06-02 | Merck Patent Gmbh | Skin colouring compsn. for durable, high intensity natural tan tones |
| WO1998023256A1 (en) * | 1996-11-25 | 1998-06-04 | Schering-Plough Healthcare Products, Inc. | Sunless tanning emulsions with disappearing color indicator |
| US6231837B1 (en) * | 1997-06-06 | 2001-05-15 | Schering-Plough Healthcare Products, Inc. | Self-tanning dihydroxyacetone formulations having improved stability and providing enhanced delivery |
| EP1060738A2 (en) * | 1999-06-15 | 2000-12-20 | Neutrogena Corporation | Self-tanning composition comprising carmine |
| WO2001012150A1 (en) * | 1999-08-13 | 2001-02-22 | Unilever Plc | Cosmetic compositions with thermochromic color change substances and an agent for inducing a temperature change |
| EP1277461A2 (en) * | 2001-07-20 | 2003-01-22 | Beiersdorf AG | Cosmetic compositions with triazines as sunscreening agents and dihydroxyacetone |
Cited By (35)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1671680A1 (en) | 2004-12-20 | 2006-06-21 | L'oreal | Self-tanning composition having two components, multicompartment device and process for colouring the skin |
| EP1754513A2 (en) | 2005-08-17 | 2007-02-21 | L'oreal | Use of 8-hexadecene-1,16-dicarboxylic acid as a care agent for promoting the horny layer cohesion |
| WO2007060021A1 (en) * | 2005-11-28 | 2007-05-31 | Johnson & Johnson Consumer France S.A.S. | Self-tanning composition |
| EP1810659A1 (en) * | 2005-11-28 | 2007-07-25 | Johnson & Johnson Consumer France SAS | Self-tanning compositions |
| WO2007099434A3 (en) * | 2006-02-28 | 2007-11-22 | Chanel Parfums Beaute | Autobronzing composition |
| EP2011481A2 (en) | 2007-07-06 | 2009-01-07 | L'Oreal | Sun-protection composition containing the association of a semi-crystalline polymer and hollow latex particles |
| EP2014277A1 (en) | 2007-07-12 | 2009-01-14 | L'Oreal | Aqueous liquid photoprotective composition based on a polyamide polymer with tertiary amide termination |
| WO2009016017A3 (en) * | 2007-07-31 | 2010-02-25 | Henkel Ag & Co. Kgaa | Stable self-tanning compositions in emulsion form |
| EP2119429A1 (en) | 2008-05-14 | 2009-11-18 | L'Oréal | Cosmetic composition containing a dibenzoylmethane derivative and a pyrrolidinone derivative; method for photostabilising the dibenzoylmethane derivative |
| EP2174644A1 (en) | 2008-10-08 | 2010-04-14 | L'oreal | Cosmetic composition comprising a dibenzoylmethane derivative and a dithiolane; Photostabilization of a dibenzoylmethane derivative |
| WO2011023886A2 (en) | 2009-08-28 | 2011-03-03 | L'oreal | Composition containing at least one lipophilic 2-hydroxybenzophenone filter and a silicon s-triazine substituted by at least two alkylaminobenzoate groups |
| EP3281620A1 (en) | 2009-10-08 | 2018-02-14 | L'oreal | Photoprotective composition based on a 2-alkoxy-4-alkyl ketone phenol compound; use of said compound for increasing the sun protection factor |
| WO2011042358A1 (en) | 2009-10-08 | 2011-04-14 | L'oreal | Photoprotective composition based on a 2-alkoxy-4-alkyl ketone phenol compound; use of said compound for increasing the sun protection factor |
| WO2011073279A2 (en) | 2009-12-18 | 2011-06-23 | L'oreal | Cosmetic treatment method involving a compound capable of condensing in situ |
| WO2011073578A2 (en) | 2009-12-18 | 2011-06-23 | L'oreal | Cosmetic treatment method using a compound that can be condensed in situ and a uv-radiation-filtering agent |
| WO2011073576A2 (en) | 2009-12-18 | 2011-06-23 | L'oreal | Skin-colouring method using a compound that can be condensed in situ and a skin-colouring agent |
| WO2011073280A2 (en) | 2009-12-18 | 2011-06-23 | L'oreal | Cosmetic treatment method involving a durable compound capable of condensing in situ |
| WO2011080043A1 (en) | 2010-01-04 | 2011-07-07 | L'oreal | Ureidopyrimidones; cosmetic use as uv-screening agents; antisun compositions containing them |
| WO2011113718A1 (en) | 2010-03-15 | 2011-09-22 | L'oreal | Composition containing a dibenzoylmethane screening agent and a hydrophilic or water-soluble merocyanin uv-screening agent; process for photostabilizing the dibenzoylmethane screening agent |
| WO2011113719A2 (en) | 2010-03-15 | 2011-09-22 | L'oreal | Composition comprising a dibenzoylmethane screening agent and a merocyanine dicyano or cyanoacetate derivative; method for the photostabilization of the dibenzoylmethane screening agent |
| WO2011151203A2 (en) | 2010-06-03 | 2011-12-08 | L'oreal | Cosmetic treatment process using a coating based on a copolymer containing polyamide blocks and polyether blocks |
| WO2012010554A1 (en) | 2010-07-20 | 2012-01-26 | L'oreal | Use of compounds resulting from a sugar polyol or from a dehydrogenated sugar polyol derivative as a uv-screening agent; antisun compositions containing same; and novel compounds |
| WO2012059348A1 (en) | 2010-11-05 | 2012-05-10 | L'oreal | Fluid aqueous antisun composition based on a superabsorbent polymer and a crosslinked copolymer of methacrylic acid and of a c1-c4 alkyl acrylate |
| WO2012105723A1 (en) | 2011-02-04 | 2012-08-09 | L'oreal | Composite pigment and method for preparation thereof |
| WO2012119860A1 (en) | 2011-03-09 | 2012-09-13 | L'oreal | Cosmetic composition containing a dibenzoylmethane derivative and a monoester-monoamide compound of methylsuccinic acid; process for photostabilizing a dibenzoylmethane derivative |
| US9452125B2 (en) | 2011-04-20 | 2016-09-27 | Kao Usa Inc. | Self-tanning compositions having reduced maillard reaction malodor |
| WO2013004777A1 (en) | 2011-07-07 | 2013-01-10 | L'oreal | Photoprotective composition |
| WO2013011094A2 (en) | 2011-07-21 | 2013-01-24 | L'oreal | Cosmetic and/or dermatological composition containing a merocyanine derivative comprising specific polar groups consisting of hydroxyl- and ether-functionalities |
| WO2013010590A1 (en) | 2011-07-21 | 2013-01-24 | L'oreal | Cosmetic and/or dermatological composition containing a merocyanine derivative comprising specific polar groups consisting of hydroxyl- and ether-functionalities |
| WO2013068237A1 (en) | 2011-11-07 | 2013-05-16 | L'oreal | Composition with a continuous oil phase containing at least one lipophilic organic uv-screening agent and hydrophobic silica aerogel particles |
| US11266584B2 (en) | 2012-07-13 | 2022-03-08 | L'oreal | Cosmetic composition comprising composite sunscreen particles |
| US11523976B2 (en) | 2012-07-13 | 2022-12-13 | L'oreal | Composite pigment and method for preparing the same |
| CN102898858A (en) * | 2012-10-19 | 2013-01-30 | 中国乐凯胶片集团公司 | Preparation method of 3', 6'-dimethoxy fluorane yellow thermosensitive dye |
| CN102898858B (en) * | 2012-10-19 | 2014-10-01 | 中国乐凯集团有限公司 | A kind of preparation method of 3',6'-dimethoxyfluorane yellow thermosensitive dye |
| CN110603025A (en) * | 2017-08-30 | 2019-12-20 | 上海百雀羚生物科技有限公司 | Cosmetic composition with antioxidant properties |
Also Published As
| Publication number | Publication date |
|---|---|
| FR2840806B1 (en) | 2005-02-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| FR2840806A1 (en) | COLORED SELF-TANNING COMPOSITIONS COMPRISING AT LEAST ONE RED OR ORANGE DYE CHOSEN AMONG FLUORANES OR THEIR ALKALI METAL SALTS | |
| EP1277462B1 (en) | Self- tanning composition containing an ester of an N-acyl amino acid | |
| EP2196189B1 (en) | Cosmetic sunscreen compositions based on synergistic filter mixture and their use | |
| EP1441692B1 (en) | Composition containing an amino acid n-acylated ester and a polyamide-structured uv filter | |
| FR2939315A1 (en) | COSMETIC COMPOSITION CONTAINING DIBENZOYLMETHANE DERIVATIVE AND ESTER COMPOUND OF 2-PYRROLIDINONE 4-CARBOXY; METHOD FOR PHOTOSTABILIZATION OF THE DIBENZOYLMETHANE DERIVATIVE | |
| EP1172092B1 (en) | Compositions for coloring the skin containing at least one UV filter and flavylium salt compounds unsubstituted in position 3 | |
| WO2002041867A1 (en) | Coloured self-tanning compositions comprising cochineal carmine | |
| EP1471059B1 (en) | 2,4,6-tris(dineopentyl 4'-aminobenzalmalonate)-s-triazin, sunscreen compositions containing this compound and its use | |
| EP1430881B1 (en) | Compositions comprising a triazine derivative solubilized in a eutectic mixture of n-butylphthalimide/isopropylphthalimide and cosmetic use | |
| EP1815884A2 (en) | Compositions containing an UV-B filter of the cinnamic acid ester type, a UV-A filter of the dibenzoylmethane type and a derivative of s-triazine; process of photostabilisation | |
| EP1327437B1 (en) | Skin dyeing compositions containing a sorghum extract and use | |
| EP1302199A2 (en) | Compositions for giving the skin a natural sun tan colouration based on a monascus type pigment and its uses | |
| EP1370233B1 (en) | Filtering composition containing a 1,3,5-triazine derivative, a dibenzoylmethane derivative, and a 4,4-diarylbutadiene compound | |
| EP1317920A1 (en) | Self-tanning composition comprising an amino-2-hydroxybenzophenone derivative and a self-tanning agent | |
| EP1568700B1 (en) | S-triazine derivatives containing at least two silated p-aminobenzalmalonate groups, sunscreen compositions containing these derivatives as well as the use of the derivatives | |
| WO2002049594A2 (en) | Cosmetic sunscreen compositions based on a synergic mixture of filters and uses | |
| EP1370231A2 (en) | Filtering composition containing a filter of the type derived from dibenzoylmethane and a 4,4-diarylbutadiene type filter | |
| EP1348424A1 (en) | Self-tanning composition containing a tetrahydrocurcumine derivative and a self-tanning agent | |
| EP1454896A1 (en) | Amine, amide, sulfonamide and carbamate derivatives of benzylidenemalonoc acid salts photoprotective cosmetic compositions containing them | |
| FR2867188A1 (en) | S-TRIAZINE DERIVATIVES HAVING PARA-ALKOXYPHENYL GROUP AND AT LEAST ONE GRAINED AMINOBENZALMALONIC GROUP; COSMETIC COMPOSITIONS CONTAINING THESE DERIVATIVES; USES OF SAID S-TRI DERIVATIVES | |
| FR2842418A1 (en) | PHOTOSTABILIZED MATERIAL CONTAINING SOLAR-GEL-PRODUCED DIBENZOYLMETHANE SOLAR FILTER AND COSMETIC AND / OR DERMOTOLOGICAL COMPOSITION CONTAINING SUCH MATERIAL | |
| FR2847811A1 (en) | FILTERING COMPOSITION CONTAINING AT LEAST ONE DIBENZOYLMETHANE DERIVATIVE AND AT LEAST ONE 3- (2-AZACYCLOALKYLIDENE) -1,3-DIHYDRO-INDOL-2-ONE DERIVATIVE, PHOTOSTABILIZATION METHOD | |
| FR2834456A1 (en) | Composition for artificial skin tanning comprises an ultraviolet filter and a sorghum extract | |
| FR2847813A1 (en) | PHOTOPROTECTIVE COSMETIC COMPOSITIONS CONTAINING 3- (2-AZACYCLOALKYLIDENE) -1,3-DIHYDRO-INDOL-2-ONE DERIVATIVES AND USES THEREOF | |
| WO2002049598A2 (en) | Cosmetic solar protection compositions based on a synergic mixture of filters and uses |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ST | Notification of lapse |
Effective date: 20160229 |