FR2470767A1 - N-Acyl-2-carboxy fused ring heterocyclic cpds. - useful for inhibiting angiotensin converting enzyme and enkephalin(s) - Google Patents
N-Acyl-2-carboxy fused ring heterocyclic cpds. - useful for inhibiting angiotensin converting enzyme and enkephalin(s) Download PDFInfo
- Publication number
- FR2470767A1 FR2470767A1 FR7930046A FR7930046A FR2470767A1 FR 2470767 A1 FR2470767 A1 FR 2470767A1 FR 7930046 A FR7930046 A FR 7930046A FR 7930046 A FR7930046 A FR 7930046A FR 2470767 A1 FR2470767 A1 FR 2470767A1
- Authority
- FR
- France
- Prior art keywords
- radical
- general formula
- substituents
- hydrogen
- acid
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- UUUHXMGGBIUAPW-UHFFFAOYSA-N 1-[1-[2-[[5-amino-2-[[1-[5-(diaminomethylideneamino)-2-[[1-[3-(1h-indol-3-yl)-2-[(5-oxopyrrolidine-2-carbonyl)amino]propanoyl]pyrrolidine-2-carbonyl]amino]pentanoyl]pyrrolidine-2-carbonyl]amino]-5-oxopentanoyl]amino]-3-methylpentanoyl]pyrrolidine-2-carbon Chemical compound C1CCC(C(=O)N2C(CCC2)C(O)=O)N1C(=O)C(C(C)CC)NC(=O)C(CCC(N)=O)NC(=O)C1CCCN1C(=O)C(CCCN=C(N)N)NC(=O)C1CCCN1C(=O)C(CC=1C2=CC=CC=C2NC=1)NC(=O)C1CCC(=O)N1 UUUHXMGGBIUAPW-UHFFFAOYSA-N 0.000 title abstract 2
- 102000004270 Peptidyl-Dipeptidase A Human genes 0.000 title abstract 2
- 108090000882 Peptidyl-Dipeptidase A Proteins 0.000 title abstract 2
- 125000000623 heterocyclic group Chemical group 0.000 title description 2
- 230000002401 inhibitory effect Effects 0.000 title description 2
- URLZCHNOLZSCCA-VABKMULXSA-N Leu-enkephalin Chemical compound C([C@@H](C(=O)N[C@@H](CC(C)C)C(O)=O)NC(=O)CNC(=O)CNC(=O)[C@@H](N)CC=1C=CC(O)=CC=1)C1=CC=CC=C1 URLZCHNOLZSCCA-VABKMULXSA-N 0.000 title 1
- -1 carboxy, cyano, hydroxy, mercapto Chemical class 0.000 claims abstract description 52
- 239000002253 acid Substances 0.000 claims abstract description 23
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims abstract description 16
- 125000000217 alkyl group Chemical group 0.000 claims abstract description 10
- 125000003545 alkoxy group Chemical group 0.000 claims abstract description 9
- 125000004122 cyclic group Chemical group 0.000 claims abstract description 6
- 125000005842 heteroatom Chemical group 0.000 claims abstract description 5
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims abstract description 4
- ORTFAQDWJHRMNX-UHFFFAOYSA-N hydroxidooxidocarbon(.) Chemical compound O[C]=O ORTFAQDWJHRMNX-UHFFFAOYSA-N 0.000 claims abstract description 3
- 150000001875 compounds Chemical class 0.000 claims description 31
- 125000001424 substituent group Chemical group 0.000 claims description 29
- 239000001257 hydrogen Substances 0.000 claims description 18
- 229910052739 hydrogen Inorganic materials 0.000 claims description 18
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 18
- 238000000034 method Methods 0.000 claims description 16
- XBDQKXXYIPTUBI-UHFFFAOYSA-N dimethylselenoniopropionate Natural products CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 claims description 12
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 claims description 8
- 150000001732 carboxylic acid derivatives Chemical class 0.000 claims description 8
- 239000008194 pharmaceutical composition Substances 0.000 claims description 8
- 125000003118 aryl group Chemical group 0.000 claims description 7
- IUVKMZGDUIUOCP-BTNSXGMBSA-N quinbolone Chemical class O([C@H]1CC[C@H]2[C@H]3[C@@H]([C@]4(C=CC(=O)C=C4CC3)C)CC[C@@]21C)C1=CCCC1 IUVKMZGDUIUOCP-BTNSXGMBSA-N 0.000 claims description 7
- 150000003839 salts Chemical class 0.000 claims description 7
- 229910052736 halogen Chemical group 0.000 claims description 6
- 150000002367 halogens Chemical group 0.000 claims description 6
- 235000019260 propionic acid Nutrition 0.000 claims description 6
- 125000002252 acyl group Chemical group 0.000 claims description 5
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 5
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 claims description 5
- RWSOTUBLDIXVET-UHFFFAOYSA-N Dihydrogen sulfide Chemical compound S RWSOTUBLDIXVET-UHFFFAOYSA-N 0.000 claims description 4
- 125000003277 amino group Chemical group 0.000 claims description 4
- MDFFNEOEWAXZRQ-UHFFFAOYSA-N aminyl Chemical compound [NH2] MDFFNEOEWAXZRQ-UHFFFAOYSA-N 0.000 claims description 4
- 229910021529 ammonia Inorganic materials 0.000 claims description 4
- 125000002619 bicyclic group Chemical group 0.000 claims description 4
- 150000003857 carboxamides Chemical class 0.000 claims description 4
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 claims description 4
- 230000000295 complement effect Effects 0.000 claims description 4
- 125000002950 monocyclic group Chemical group 0.000 claims description 4
- 150000007530 organic bases Chemical class 0.000 claims description 4
- 239000000546 pharmaceutical excipient Substances 0.000 claims description 4
- 229920006395 saturated elastomer Polymers 0.000 claims description 4
- 230000002195 synergetic effect Effects 0.000 claims description 4
- 150000003573 thiols Chemical class 0.000 claims description 4
- 125000005907 alkyl ester group Chemical group 0.000 claims description 3
- 125000003710 aryl alkyl group Chemical group 0.000 claims description 3
- YDVJBLJCSLVMSY-UHFFFAOYSA-N carbamoyl cyanide Chemical compound NC(=O)C#N YDVJBLJCSLVMSY-UHFFFAOYSA-N 0.000 claims description 3
- 150000002148 esters Chemical class 0.000 claims description 3
- CYEBJEDOHLIWNP-UHFFFAOYSA-N methanethioamide Chemical compound NC=S CYEBJEDOHLIWNP-UHFFFAOYSA-N 0.000 claims description 3
- 231100000252 nontoxic Toxicity 0.000 claims description 3
- 230000003000 nontoxic effect Effects 0.000 claims description 3
- 239000000126 substance Substances 0.000 claims description 3
- 125000005530 alkylenedioxy group Chemical group 0.000 claims description 2
- 230000009435 amidation Effects 0.000 claims description 2
- 238000007112 amidation reaction Methods 0.000 claims description 2
- 150000001408 amides Chemical class 0.000 claims description 2
- JAKDMTBLDOEDGS-UHFFFAOYSA-N carbonocyanidoylcarbamic acid Chemical compound OC(=O)NC(=O)C#N JAKDMTBLDOEDGS-UHFFFAOYSA-N 0.000 claims description 2
- 230000001882 diuretic effect Effects 0.000 claims description 2
- 125000005843 halogen group Chemical group 0.000 claims description 2
- 125000004029 hydroxymethyl group Chemical group [H]OC([H])([H])* 0.000 claims description 2
- 150000007529 inorganic bases Chemical class 0.000 claims description 2
- HZVOZRGWRWCICA-UHFFFAOYSA-N methanediyl Chemical group [CH2] HZVOZRGWRWCICA-UHFFFAOYSA-N 0.000 claims description 2
- 125000006340 pentafluoro ethyl group Chemical group FC(F)(F)C(F)(F)* 0.000 claims description 2
- 125000005034 trifluormethylthio group Chemical group FC(S*)(F)F 0.000 claims description 2
- 125000000876 trifluoromethoxy group Chemical group FC(F)(F)O* 0.000 claims description 2
- HAVZTGSQJIEKPI-UHFFFAOYSA-N benzothiadiazine Chemical compound C1=CC=C2C=NNSC2=C1 HAVZTGSQJIEKPI-UHFFFAOYSA-N 0.000 claims 1
- 150000002460 imidazoles Chemical class 0.000 claims 1
- 125000000446 sulfanediyl group Chemical group *S* 0.000 claims 1
- 229940124530 sulfonamide Drugs 0.000 claims 1
- 150000003456 sulfonamides Chemical class 0.000 claims 1
- 125000003039 tetrahydroisoquinolinyl group Chemical class C1(NCCC2=CC=CC=C12)* 0.000 claims 1
- 208000037849 arterial hypertension Diseases 0.000 abstract description 3
- 206010019280 Heart failures Diseases 0.000 abstract description 2
- 238000006243 chemical reaction Methods 0.000 abstract description 2
- 125000001841 imino group Chemical group [H]N=* 0.000 abstract description 2
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 abstract description 2
- 125000005605 benzo group Chemical group 0.000 abstract 1
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 abstract 1
- 238000010511 deprotection reaction Methods 0.000 abstract 1
- 239000003112 inhibitor Substances 0.000 abstract 1
- 125000003396 thiol group Chemical group [H]S* 0.000 abstract 1
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 18
- 239000000243 solution Substances 0.000 description 16
- 239000000203 mixture Substances 0.000 description 11
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 10
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 9
- 239000000047 product Substances 0.000 description 9
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 8
- CUKWUWBLQQDQAC-VEQWQPCFSA-N (3s)-3-amino-4-[[(2s)-1-[[(2s)-1-[[(2s)-1-[[(2s,3s)-1-[[(2s)-1-[(2s)-2-[[(1s)-1-carboxyethyl]carbamoyl]pyrrolidin-1-yl]-3-(1h-imidazol-5-yl)-1-oxopropan-2-yl]amino]-3-methyl-1-oxopentan-2-yl]amino]-3-(4-hydroxyphenyl)-1-oxopropan-2-yl]amino]-3-methyl-1-ox Chemical compound C([C@@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CC=1NC=NC=1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](C)C(O)=O)NC(=O)[C@@H](NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@@H](N)CC(O)=O)C(C)C)C1=CC=C(O)C=C1 CUKWUWBLQQDQAC-VEQWQPCFSA-N 0.000 description 7
- 102000005862 Angiotensin II Human genes 0.000 description 7
- 101800000733 Angiotensin-2 Proteins 0.000 description 7
- 238000004458 analytical method Methods 0.000 description 7
- 229950006323 angiotensin ii Drugs 0.000 description 7
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 7
- 101800000734 Angiotensin-1 Proteins 0.000 description 6
- 102400000344 Angiotensin-1 Human genes 0.000 description 6
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 6
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 6
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 6
- ORWYRWWVDCYOMK-HBZPZAIKSA-N angiotensin I Chemical compound C([C@@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CC=1NC=NC=1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](CC=1NC=NC=1)C(=O)N[C@@H](CC(C)C)C(O)=O)NC(=O)[C@@H](NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@@H](N)CC(O)=O)C(C)C)C1=CC=C(O)C=C1 ORWYRWWVDCYOMK-HBZPZAIKSA-N 0.000 description 6
- NENLYAQPNATJSU-DTWKUNHWSA-N (4as,8as)-1,2,3,4,4a,5,6,7,8,8a-decahydroisoquinoline Chemical compound C1NCC[C@@H]2CCCC[C@@H]21 NENLYAQPNATJSU-DTWKUNHWSA-N 0.000 description 5
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical class [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 5
- UWYZHKAOTLEWKK-UHFFFAOYSA-N 1,2,3,4-tetrahydroisoquinoline Chemical compound C1=CC=C2CNCCC2=C1 UWYZHKAOTLEWKK-UHFFFAOYSA-N 0.000 description 4
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 4
- KWGKDLIKAYFUFQ-UHFFFAOYSA-M lithium chloride Chemical compound [Li+].[Cl-] KWGKDLIKAYFUFQ-UHFFFAOYSA-M 0.000 description 4
- 150000004702 methyl esters Chemical class 0.000 description 4
- 230000000144 pharmacologic effect Effects 0.000 description 4
- 239000002244 precipitate Substances 0.000 description 4
- YGSDEFSMJLZEOE-UHFFFAOYSA-M salicylate Chemical compound OC1=CC=CC=C1C([O-])=O YGSDEFSMJLZEOE-UHFFFAOYSA-M 0.000 description 4
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 3
- XBPCUCUWBYBCDP-UHFFFAOYSA-N Dicyclohexylamine Chemical compound C1CCCCC1NC1CCCCC1 XBPCUCUWBYBCDP-UHFFFAOYSA-N 0.000 description 3
- 206010020772 Hypertension Diseases 0.000 description 3
- SIKJAQJRHWYJAI-UHFFFAOYSA-N Indole Chemical compound C1=CC=C2NC=CC2=C1 SIKJAQJRHWYJAI-UHFFFAOYSA-N 0.000 description 3
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 229920002472 Starch Polymers 0.000 description 3
- 229940024606 amino acid Drugs 0.000 description 3
- 150000001413 amino acids Chemical class 0.000 description 3
- FAKRSMQSSFJEIM-RQJHMYQMSA-N captopril Chemical compound SC[C@@H](C)C(=O)N1CCC[C@H]1C(O)=O FAKRSMQSSFJEIM-RQJHMYQMSA-N 0.000 description 3
- 229960000830 captopril Drugs 0.000 description 3
- 150000001721 carbon Chemical group 0.000 description 3
- 238000004587 chromatography analysis Methods 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 238000009833 condensation Methods 0.000 description 3
- 230000005494 condensation Effects 0.000 description 3
- 239000013078 crystal Substances 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 239000000706 filtrate Substances 0.000 description 3
- 238000001914 filtration Methods 0.000 description 3
- 125000002883 imidazolyl group Chemical group 0.000 description 3
- 238000002329 infrared spectrum Methods 0.000 description 3
- 238000001802 infusion Methods 0.000 description 3
- 238000002347 injection Methods 0.000 description 3
- 239000007924 injection Substances 0.000 description 3
- 239000002609 medium Substances 0.000 description 3
- 239000002904 solvent Substances 0.000 description 3
- 238000003756 stirring Methods 0.000 description 3
- 230000001225 therapeutic effect Effects 0.000 description 3
- 230000009466 transformation Effects 0.000 description 3
- KWGRBVOPPLSCSI-WPRPVWTQSA-N (-)-ephedrine Chemical compound CN[C@@H](C)[C@H](O)C1=CC=CC=C1 KWGRBVOPPLSCSI-WPRPVWTQSA-N 0.000 description 2
- OFFSPAZVIVZPHU-UHFFFAOYSA-N 1-benzofuran-2-carboxylic acid Chemical compound C1=CC=C2OC(C(=O)O)=CC2=C1 OFFSPAZVIVZPHU-UHFFFAOYSA-N 0.000 description 2
- CFEGVJOLMOPMHU-UHFFFAOYSA-N 1-methyl-1,2,3,4-tetrahydroisoquinolin-2-ium;chloride Chemical compound Cl.C1=CC=C2C(C)NCCC2=C1 CFEGVJOLMOPMHU-UHFFFAOYSA-N 0.000 description 2
- YBYIRNPNPLQARY-UHFFFAOYSA-N 1H-indene Chemical compound C1=CC=C2CC=CC2=C1 YBYIRNPNPLQARY-UHFFFAOYSA-N 0.000 description 2
- HBEDSQVIWPRPAY-UHFFFAOYSA-N 2,3-dihydrobenzofuran Chemical compound C1=CC=C2OCCC2=C1 HBEDSQVIWPRPAY-UHFFFAOYSA-N 0.000 description 2
- VFVHNRJEYQGRGE-UHFFFAOYSA-N 3-acetylsulfanyl-2-methylpropanoic acid Chemical compound OC(=O)C(C)CSC(C)=O VFVHNRJEYQGRGE-UHFFFAOYSA-N 0.000 description 2
- 241000282472 Canis lupus familiaris Species 0.000 description 2
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 2
- ROSDSFDQCJNGOL-UHFFFAOYSA-N Dimethylamine Chemical compound CNC ROSDSFDQCJNGOL-UHFFFAOYSA-N 0.000 description 2
- 108090000790 Enzymes Proteins 0.000 description 2
- 102000004190 Enzymes Human genes 0.000 description 2
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 2
- COLNVLDHVKWLRT-QMMMGPOBSA-N L-phenylalanine Chemical compound OC(=O)[C@@H](N)CC1=CC=CC=C1 COLNVLDHVKWLRT-QMMMGPOBSA-N 0.000 description 2
- 241001465754 Metazoa Species 0.000 description 2
- BAVYZALUXZFZLV-UHFFFAOYSA-N Methylamine Chemical compound NC BAVYZALUXZFZLV-UHFFFAOYSA-N 0.000 description 2
- UFWIBTONFRDIAS-UHFFFAOYSA-N Naphthalene Chemical compound C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 2
- KYQCOXFCLRTKLS-UHFFFAOYSA-N Pyrazine Chemical compound C1=CN=CC=N1 KYQCOXFCLRTKLS-UHFFFAOYSA-N 0.000 description 2
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 2
- KAESVJOAVNADME-UHFFFAOYSA-N Pyrrole Chemical compound C=1C=CNC=1 KAESVJOAVNADME-UHFFFAOYSA-N 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Thiophene Chemical compound C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 description 2
- 239000004480 active ingredient Substances 0.000 description 2
- 150000003973 alkyl amines Chemical class 0.000 description 2
- 150000008064 anhydrides Chemical class 0.000 description 2
- MSWZFWKMSRAUBD-UHFFFAOYSA-N beta-D-galactosamine Natural products NC1C(O)OC(CO)C(O)C1O MSWZFWKMSRAUBD-UHFFFAOYSA-N 0.000 description 2
- 230000036772 blood pressure Effects 0.000 description 2
- 238000009835 boiling Methods 0.000 description 2
- OSGAYBCDTDRGGQ-UHFFFAOYSA-L calcium sulfate Chemical compound [Ca+2].[O-]S([O-])(=O)=O OSGAYBCDTDRGGQ-UHFFFAOYSA-L 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 2
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 2
- 150000001735 carboxylic acids Chemical class 0.000 description 2
- 239000005018 casein Substances 0.000 description 2
- BECPQYXYKAMYBN-UHFFFAOYSA-N casein, tech. Chemical compound NCCCCC(C(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(CC(C)C)N=C(O)C(CCC(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(C(C)O)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(COP(O)(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(N)CC1=CC=CC=C1 BECPQYXYKAMYBN-UHFFFAOYSA-N 0.000 description 2
- 235000021240 caseins Nutrition 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 239000012153 distilled water Substances 0.000 description 2
- LIWAQLJGPBVORC-UHFFFAOYSA-N ethylmethylamine Chemical compound CCNC LIWAQLJGPBVORC-UHFFFAOYSA-N 0.000 description 2
- 238000001704 evaporation Methods 0.000 description 2
- 230000008020 evaporation Effects 0.000 description 2
- RERZNCLIYCABFS-UHFFFAOYSA-N harmaline Chemical compound C1CN=C(C)C2=C1C1=CC=C(OC)C=C1N2 RERZNCLIYCABFS-UHFFFAOYSA-N 0.000 description 2
- 238000005984 hydrogenation reaction Methods 0.000 description 2
- NDDAHWYSQHTHNT-UHFFFAOYSA-N indapamide Chemical compound CC1CC2=CC=CC=C2N1NC(=O)C1=CC=C(Cl)C(S(N)(=O)=O)=C1 NDDAHWYSQHTHNT-UHFFFAOYSA-N 0.000 description 2
- 230000005764 inhibitory process Effects 0.000 description 2
- 229910052500 inorganic mineral Inorganic materials 0.000 description 2
- 239000002198 insoluble material Substances 0.000 description 2
- LOIDKAGDLUKYNE-UHFFFAOYSA-N isoquinoline-6,7-diol Chemical compound C1=NC=C2C=C(O)C(O)=CC2=C1 LOIDKAGDLUKYNE-UHFFFAOYSA-N 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 239000011707 mineral Substances 0.000 description 2
- 235000010755 mineral Nutrition 0.000 description 2
- 125000004433 nitrogen atom Chemical group N* 0.000 description 2
- VLTRZXGMWDSKGL-UHFFFAOYSA-N perchloric acid Chemical compound OCl(=O)(=O)=O VLTRZXGMWDSKGL-UHFFFAOYSA-N 0.000 description 2
- XSCHRSMBECNVNS-UHFFFAOYSA-N quinoxaline Chemical compound N1=CC=NC2=CC=CC=C21 XSCHRSMBECNVNS-UHFFFAOYSA-N 0.000 description 2
- 150000003254 radicals Chemical class 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- 239000000454 talc Substances 0.000 description 2
- 229910052623 talc Inorganic materials 0.000 description 2
- FYSNRJHAOHDILO-UHFFFAOYSA-N thionyl chloride Chemical compound ClS(Cl)=O FYSNRJHAOHDILO-UHFFFAOYSA-N 0.000 description 2
- 229940086542 triethylamine Drugs 0.000 description 2
- 229940100445 wheat starch Drugs 0.000 description 2
- AIFRHYZBTHREPW-UHFFFAOYSA-N β-carboline Chemical compound N1=CC=C2C3=CC=CC=C3NC2=C1 AIFRHYZBTHREPW-UHFFFAOYSA-N 0.000 description 2
- SUFGJGROOFDYRU-UHFFFAOYSA-N (cyanocarbothioyl)carbamic acid Chemical compound OC(=O)NC(=S)C#N SUFGJGROOFDYRU-UHFFFAOYSA-N 0.000 description 1
- MGFREDWKELGWML-UHFFFAOYSA-N 1,2,3,4-tetrahydroisoquinolin-2-ium;chloride Chemical compound [Cl-].C1=CC=C2C[NH2+]CCC2=C1 MGFREDWKELGWML-UHFFFAOYSA-N 0.000 description 1
- BWKMGYQJPOAASG-UHFFFAOYSA-N 1,2,3,4-tetrahydroisoquinoline-3-carboxylic acid Chemical compound C1=CC=C2CNC(C(=O)O)CC2=C1 BWKMGYQJPOAASG-UHFFFAOYSA-N 0.000 description 1
- OTPDWCMLUKMQNO-UHFFFAOYSA-N 1,2,3,4-tetrahydropyrimidine Chemical compound C1NCC=CN1 OTPDWCMLUKMQNO-UHFFFAOYSA-N 0.000 description 1
- ADFXKUOMJKEIND-UHFFFAOYSA-N 1,3-dicyclohexylurea Chemical compound C1CCCCC1NC(=O)NC1CCCCC1 ADFXKUOMJKEIND-UHFFFAOYSA-N 0.000 description 1
- TYLXHTVATOWOEK-UHFFFAOYSA-N 1-(3,4-dihydro-1h-isoquinolin-2-yl)-2-methyl-3-sulfanylpropan-1-one Chemical compound C1=CC=C2CN(C(=O)C(CS)C)CCC2=C1 TYLXHTVATOWOEK-UHFFFAOYSA-N 0.000 description 1
- CFTOTSJVQRFXOF-UHFFFAOYSA-N 2,3,4,9-tetrahydro-1H-pyrido[3,4-b]indole Chemical compound N1C2=CC=CC=C2C2=C1CNCC2 CFTOTSJVQRFXOF-UHFFFAOYSA-N 0.000 description 1
- 125000004201 2,4-dichlorophenyl group Chemical group [H]C1=C([H])C(*)=C(Cl)C([H])=C1Cl 0.000 description 1
- GDGPVPYTXHNOSO-UHFFFAOYSA-N 2-(2-methyl-3-sulfanylpropanoyl)-3,4-dihydro-1h-isoquinoline-3-carboxylic acid Chemical compound C1=CC=C2CC(C(O)=O)N(C(=O)C(CS)C)CC2=C1 GDGPVPYTXHNOSO-UHFFFAOYSA-N 0.000 description 1
- CALGIEBSJLYYOS-UHFFFAOYSA-N 2-(acetylsulfanylmethyl)-3,3,3-trifluoropropanoic acid Chemical compound CC(=O)SCC(C(O)=O)C(F)(F)F CALGIEBSJLYYOS-UHFFFAOYSA-N 0.000 description 1
- WKFXOUSFZMOKOH-UHFFFAOYSA-N 2-(dimethylamino)-1-(4-nitrophenyl)propane-1,3-diol Chemical compound CN(C)C(CO)C(O)C1=CC=C([N+]([O-])=O)C=C1 WKFXOUSFZMOKOH-UHFFFAOYSA-N 0.000 description 1
- XNMYNYSCEJBRPZ-UHFFFAOYSA-N 2-[(3-butyl-1-isoquinolinyl)oxy]-N,N-dimethylethanamine Chemical compound C1=CC=C2C(OCCN(C)C)=NC(CCCC)=CC2=C1 XNMYNYSCEJBRPZ-UHFFFAOYSA-N 0.000 description 1
- MSWZFWKMSRAUBD-IVMDWMLBSA-N 2-amino-2-deoxy-D-glucopyranose Chemical compound N[C@H]1C(O)O[C@H](CO)[C@@H](O)[C@@H]1O MSWZFWKMSRAUBD-IVMDWMLBSA-N 0.000 description 1
- MSWZFWKMSRAUBD-CBPJZXOFSA-N 2-amino-2-deoxy-D-mannopyranose Chemical compound N[C@@H]1C(O)O[C@H](CO)[C@@H](O)[C@@H]1O MSWZFWKMSRAUBD-CBPJZXOFSA-N 0.000 description 1
- QXODWULWEVPKFS-UHFFFAOYSA-N 2-methyl-4-oxopentanethioic s-acid Chemical compound SC(=O)C(C)CC(C)=O QXODWULWEVPKFS-UHFFFAOYSA-N 0.000 description 1
- VSWICNJIUPRZIK-UHFFFAOYSA-N 2-piperideine Chemical compound C1CNC=CC1 VSWICNJIUPRZIK-UHFFFAOYSA-N 0.000 description 1
- JMTMSDXUXJISAY-UHFFFAOYSA-N 2H-benzotriazol-4-ol Chemical compound OC1=CC=CC2=C1N=NN2 JMTMSDXUXJISAY-UHFFFAOYSA-N 0.000 description 1
- 125000005809 3,4,5-trimethoxyphenyl group Chemical group [H]C1=C(OC([H])([H])[H])C(OC([H])([H])[H])=C(OC([H])([H])[H])C([H])=C1* 0.000 description 1
- 125000002774 3,4-dimethoxybenzyl group Chemical group [H]C1=C([H])C(=C([H])C(OC([H])([H])[H])=C1OC([H])([H])[H])C([H])([H])* 0.000 description 1
- 125000003762 3,4-dimethoxyphenyl group Chemical group [H]C1=C([H])C(OC([H])([H])[H])=C(OC([H])([H])[H])C([H])=C1* 0.000 description 1
- RJSBWCNRDKOMPO-UHFFFAOYSA-N 3-chloro-n-(2-methyl-2,3-dihydroindol-1-yl)-4-sulfamoylbenzamide Chemical compound CC1CC2=CC=CC=C2N1NC(=O)C1=CC=C(S(N)(=O)=O)C(Cl)=C1 RJSBWCNRDKOMPO-UHFFFAOYSA-N 0.000 description 1
- LMEKMWZJKGZZLR-UHFFFAOYSA-N 3-cyano-2-methylpropanoic acid Chemical compound OC(=O)C(C)CC#N LMEKMWZJKGZZLR-UHFFFAOYSA-N 0.000 description 1
- 125000004203 4-hydroxyphenyl group Chemical group [H]OC1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 1
- UIPNPFUNPXYKGP-UHFFFAOYSA-N 6,7-dihydroxy-1,2,3,4-tetrahydroisoquinoline-3-carboxylic acid Chemical compound OC1=C(O)C=C2CNC(C(=O)O)CC2=C1 UIPNPFUNPXYKGP-UHFFFAOYSA-N 0.000 description 1
- KYARBIJYVGJZLB-UHFFFAOYSA-N 7-amino-4-hydroxy-2-naphthalenesulfonic acid Chemical compound OC1=CC(S(O)(=O)=O)=CC2=CC(N)=CC=C21 KYARBIJYVGJZLB-UHFFFAOYSA-N 0.000 description 1
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 1
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 1
- 102000004881 Angiotensinogen Human genes 0.000 description 1
- 108090001067 Angiotensinogen Proteins 0.000 description 1
- 108010064733 Angiotensins Proteins 0.000 description 1
- 102000015427 Angiotensins Human genes 0.000 description 1
- 239000004475 Arginine Substances 0.000 description 1
- 229930008564 C01BA04 - Sparteine Natural products 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- 229920002261 Corn starch Polymers 0.000 description 1
- HTJDQJBWANPRPF-UHFFFAOYSA-N Cyclopropylamine Chemical compound NC1CC1 HTJDQJBWANPRPF-UHFFFAOYSA-N 0.000 description 1
- QOSSAOTZNIDXMA-UHFFFAOYSA-N Dicylcohexylcarbodiimide Chemical compound C1CCCCC1N=C=NC1CCCCC1 QOSSAOTZNIDXMA-UHFFFAOYSA-N 0.000 description 1
- ZAFNJMIOTHYJRJ-UHFFFAOYSA-N Diisopropyl ether Chemical compound CC(C)OC(C)C ZAFNJMIOTHYJRJ-UHFFFAOYSA-N 0.000 description 1
- 208000007530 Essential hypertension Diseases 0.000 description 1
- 239000004471 Glycine Substances 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- ODKSFYDXXFIFQN-BYPYZUCNSA-P L-argininium(2+) Chemical compound NC(=[NH2+])NCCC[C@H]([NH3+])C(O)=O ODKSFYDXXFIFQN-BYPYZUCNSA-P 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- MBBZMMPHUWSWHV-BDVNFPICSA-N N-methylglucamine Chemical compound CNC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO MBBZMMPHUWSWHV-BDVNFPICSA-N 0.000 description 1
- FUVGZDDOHNQZEO-UHFFFAOYSA-N NS(=O)(=O)NCl Chemical compound NS(=O)(=O)NCl FUVGZDDOHNQZEO-UHFFFAOYSA-N 0.000 description 1
- QMGVPVSNSZLJIA-UHFFFAOYSA-N Nux Vomica Natural products C1C2C3C4N(C=5C6=CC=CC=5)C(=O)CC3OCC=C2CN2C1C46CC2 QMGVPVSNSZLJIA-UHFFFAOYSA-N 0.000 description 1
- PCNDJXKNXGMECE-UHFFFAOYSA-N Phenazine Natural products C1=CC=CC2=NC3=CC=CC=C3N=C21 PCNDJXKNXGMECE-UHFFFAOYSA-N 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 1
- CZPWVGJYEJSRLH-UHFFFAOYSA-N Pyrimidine Chemical compound C1=CN=CN=C1 CZPWVGJYEJSRLH-UHFFFAOYSA-N 0.000 description 1
- 108090000783 Renin Proteins 0.000 description 1
- 102100028255 Renin Human genes 0.000 description 1
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- 241000534944 Thia Species 0.000 description 1
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 239000012190 activator Substances 0.000 description 1
- 125000004849 alkoxymethyl group Chemical group 0.000 description 1
- SLRCCWJSBJZJBV-UHFFFAOYSA-N alpha-isosparteine Natural products C1N2CCCCC2C2CN3CCCCC3C1C2 SLRCCWJSBJZJBV-UHFFFAOYSA-N 0.000 description 1
- AZDRQVAHHNSJOQ-UHFFFAOYSA-N alumane Chemical class [AlH3] AZDRQVAHHNSJOQ-UHFFFAOYSA-N 0.000 description 1
- 125000004397 aminosulfonyl group Chemical group NS(=O)(=O)* 0.000 description 1
- 210000000709 aorta Anatomy 0.000 description 1
- 239000012736 aqueous medium Substances 0.000 description 1
- 239000008346 aqueous phase Substances 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- XYOVOXDWRFGKEX-UHFFFAOYSA-N azepine Chemical compound N1C=CC=CC=C1 XYOVOXDWRFGKEX-UHFFFAOYSA-N 0.000 description 1
- RQPZNWPYLFFXCP-UHFFFAOYSA-L barium dihydroxide Chemical compound [OH-].[OH-].[Ba+2] RQPZNWPYLFFXCP-UHFFFAOYSA-L 0.000 description 1
- 229910001863 barium hydroxide Inorganic materials 0.000 description 1
- TZCXTZWJZNENPQ-UHFFFAOYSA-L barium sulfate Chemical compound [Ba+2].[O-]S([O-])(=O)=O TZCXTZWJZNENPQ-UHFFFAOYSA-L 0.000 description 1
- 239000010428 baryte Substances 0.000 description 1
- 229910052601 baryte Inorganic materials 0.000 description 1
- RFRXIWQYSOIBDI-UHFFFAOYSA-N benzarone Chemical compound CCC=1OC2=CC=CC=C2C=1C(=O)C1=CC=C(O)C=C1 RFRXIWQYSOIBDI-UHFFFAOYSA-N 0.000 description 1
- RTEXIPZMMDUXMR-UHFFFAOYSA-N benzene;ethyl acetate Chemical compound CCOC(C)=O.C1=CC=CC=C1 RTEXIPZMMDUXMR-UHFFFAOYSA-N 0.000 description 1
- QOCVOOWVZVIZEO-UHFFFAOYSA-N benzene;sulfamide Chemical compound NS(N)(=O)=O.C1=CC=CC=C1 QOCVOOWVZVIZEO-UHFFFAOYSA-N 0.000 description 1
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid group Chemical group C(C1=CC=CC=C1)(=O)O WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 1
- MSWZFWKMSRAUBD-QZABAPFNSA-N beta-D-glucosamine Chemical compound N[C@H]1[C@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O MSWZFWKMSRAUBD-QZABAPFNSA-N 0.000 description 1
- MDHYEMXUFSJLGV-UHFFFAOYSA-N beta-phenethyl acetate Natural products CC(=O)OCCC1=CC=CC=C1 MDHYEMXUFSJLGV-UHFFFAOYSA-N 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- GRSTVVGJSKHCCS-UHFFFAOYSA-N bis(1h-imidazol-2-yl)methanone Chemical compound N=1C=CNC=1C(=O)C1=NC=CN1 GRSTVVGJSKHCCS-UHFFFAOYSA-N 0.000 description 1
- RRKTZKIUPZVBMF-IBTVXLQLSA-N brucine Chemical compound O([C@@H]1[C@H]([C@H]2C3)[C@@H]4N(C(C1)=O)C=1C=C(C(=CC=11)OC)OC)CC=C2CN2[C@@H]3[C@]41CC2 RRKTZKIUPZVBMF-IBTVXLQLSA-N 0.000 description 1
- RRKTZKIUPZVBMF-UHFFFAOYSA-N brucine Natural products C1=2C=C(OC)C(OC)=CC=2N(C(C2)=O)C3C(C4C5)C2OCC=C4CN2C5C31CC2 RRKTZKIUPZVBMF-UHFFFAOYSA-N 0.000 description 1
- 230000005587 bubbling Effects 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- AXCZMVOFGPJBDE-UHFFFAOYSA-L calcium dihydroxide Chemical compound [OH-].[OH-].[Ca+2] AXCZMVOFGPJBDE-UHFFFAOYSA-L 0.000 description 1
- 239000000920 calcium hydroxide Substances 0.000 description 1
- 229910001861 calcium hydroxide Inorganic materials 0.000 description 1
- 239000001506 calcium phosphate Substances 0.000 description 1
- 239000001175 calcium sulphate Substances 0.000 description 1
- 235000011132 calcium sulphate Nutrition 0.000 description 1
- 238000004364 calculation method Methods 0.000 description 1
- 125000004432 carbon atom Chemical group C* 0.000 description 1
- 239000001569 carbon dioxide Substances 0.000 description 1
- 229910002092 carbon dioxide Inorganic materials 0.000 description 1
- 150000001733 carboxylic acid esters Chemical class 0.000 description 1
- 125000002057 carboxymethyl group Chemical group [H]OC(=O)C([H])([H])[*] 0.000 description 1
- 125000004803 chlorobenzyl group Chemical group 0.000 description 1
- 239000008119 colloidal silica Substances 0.000 description 1
- 239000008120 corn starch Substances 0.000 description 1
- 125000000753 cycloalkyl group Chemical group 0.000 description 1
- KWGRBVOPPLSCSI-UHFFFAOYSA-N d-ephedrine Natural products CNC(C)C(O)C1=CC=CC=C1 KWGRBVOPPLSCSI-UHFFFAOYSA-N 0.000 description 1
- UAOMVDZJSHZZME-UHFFFAOYSA-N diisopropylamine Chemical compound CC(C)NC(C)C UAOMVDZJSHZZME-UHFFFAOYSA-N 0.000 description 1
- 239000002934 diuretic Substances 0.000 description 1
- 238000010828 elution Methods 0.000 description 1
- 229960002179 ephedrine Drugs 0.000 description 1
- AVOLMBLBETYQHX-UHFFFAOYSA-N etacrynic acid Chemical compound CCC(=C)C(=O)C1=CC=C(OCC(O)=O)C(Cl)=C1Cl AVOLMBLBETYQHX-UHFFFAOYSA-N 0.000 description 1
- 229960003199 etacrynic acid Drugs 0.000 description 1
- RIFGWPKJUGCATF-UHFFFAOYSA-N ethyl chloroformate Chemical compound CCOC(Cl)=O RIFGWPKJUGCATF-UHFFFAOYSA-N 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- WMYNMYVRWWCRPS-UHFFFAOYSA-N ethynoxyethane Chemical group CCOC#C WMYNMYVRWWCRPS-UHFFFAOYSA-N 0.000 description 1
- 210000001105 femoral artery Anatomy 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 235000013355 food flavoring agent Nutrition 0.000 description 1
- 235000003599 food sweetener Nutrition 0.000 description 1
- ZZUFCTLCJUWOSV-UHFFFAOYSA-N furosemide Chemical compound C1=C(Cl)C(S(=O)(=O)N)=CC(C(O)=O)=C1NCC1=CC=CO1 ZZUFCTLCJUWOSV-UHFFFAOYSA-N 0.000 description 1
- 229960003883 furosemide Drugs 0.000 description 1
- 229960002442 glucosamine Drugs 0.000 description 1
- 150000002337 glycosamines Chemical class 0.000 description 1
- 150000004820 halides Chemical class 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 239000005556 hormone Substances 0.000 description 1
- 229940088597 hormone Drugs 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- NPZTUJOABDZTLV-UHFFFAOYSA-N hydroxybenzotriazole Substances O=C1C=CC=C2NNN=C12 NPZTUJOABDZTLV-UHFFFAOYSA-N 0.000 description 1
- 239000005457 ice water Substances 0.000 description 1
- MTNDZQHUAFNZQY-UHFFFAOYSA-N imidazoline Chemical compound C1CN=CN1 MTNDZQHUAFNZQY-UHFFFAOYSA-N 0.000 description 1
- PZOUSPYUWWUPPK-UHFFFAOYSA-N indole Natural products CC1=CC=CC2=C1C=CN2 PZOUSPYUWWUPPK-UHFFFAOYSA-N 0.000 description 1
- RKJUIXBNRJVNHR-UHFFFAOYSA-N indolenine Natural products C1=CC=C2CC=NC2=C1 RKJUIXBNRJVNHR-UHFFFAOYSA-N 0.000 description 1
- 150000002476 indolines Chemical class 0.000 description 1
- 239000011261 inert gas Substances 0.000 description 1
- 230000010354 integration Effects 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 239000007791 liquid phase Substances 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- HCWCAKKEBCNQJP-UHFFFAOYSA-N magnesium orthosilicate Chemical compound [Mg+2].[Mg+2].[O-][Si]([O-])([O-])[O-] HCWCAKKEBCNQJP-UHFFFAOYSA-N 0.000 description 1
- 239000000391 magnesium silicate Substances 0.000 description 1
- 229910052919 magnesium silicate Inorganic materials 0.000 description 1
- 235000019792 magnesium silicate Nutrition 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 238000003760 magnetic stirring Methods 0.000 description 1
- 238000010907 mechanical stirring Methods 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- ULEZWUGQDAQWPT-UHFFFAOYSA-N n-ethylcyclopropanamine Chemical compound CCNC1CC1 ULEZWUGQDAQWPT-UHFFFAOYSA-N 0.000 description 1
- 229940075566 naphthalene Drugs 0.000 description 1
- 125000001624 naphthyl group Chemical group 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- MBFUSGLXKQWVDW-UHFFFAOYSA-N norsalsolinol Natural products C1CNCC2=C1C=C(O)C(O)=C2 MBFUSGLXKQWVDW-UHFFFAOYSA-N 0.000 description 1
- 238000000655 nuclear magnetic resonance spectrum Methods 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 125000005702 oxyalkylene group Chemical group 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 1
- 239000012071 phase Substances 0.000 description 1
- LCPDWSOZIOUXRV-UHFFFAOYSA-N phenoxyacetic acid Chemical class OC(=O)COC1=CC=CC=C1 LCPDWSOZIOUXRV-UHFFFAOYSA-N 0.000 description 1
- 229960005190 phenylalanine Drugs 0.000 description 1
- 125000000286 phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004344 phenylpropyl group Chemical group 0.000 description 1
- 238000000053 physical method Methods 0.000 description 1
- 229940072033 potash Drugs 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Substances [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 1
- 235000015320 potassium carbonate Nutrition 0.000 description 1
- IENZQIKPVFGBNW-UHFFFAOYSA-N prazosin Chemical compound N=1C(N)=C2C=C(OC)C(OC)=CC2=NC=1N(CC1)CCN1C(=O)C1=CC=CO1 IENZQIKPVFGBNW-UHFFFAOYSA-N 0.000 description 1
- 229960001289 prazosin Drugs 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 108090000765 processed proteins & peptides Proteins 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- QELSKZZBTMNZEB-UHFFFAOYSA-N propylparaben Chemical compound CCCOC(=O)C1=CC=C(O)C=C1 QELSKZZBTMNZEB-UHFFFAOYSA-N 0.000 description 1
- 229960003415 propylparaben Drugs 0.000 description 1
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 1
- 229960005038 quinisocaine Drugs 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 239000012429 reaction media Substances 0.000 description 1
- 238000010992 reflux Methods 0.000 description 1
- 230000000241 respiratory effect Effects 0.000 description 1
- 230000029058 respiratory gaseous exchange Effects 0.000 description 1
- 230000000894 saliuretic effect Effects 0.000 description 1
- 238000007127 saponification reaction Methods 0.000 description 1
- 229930195734 saturated hydrocarbon Natural products 0.000 description 1
- 239000012047 saturated solution Substances 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 159000000000 sodium salts Chemical class 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- SLRCCWJSBJZJBV-AJNGGQMLSA-N sparteine Chemical compound C1N2CCCC[C@H]2[C@@H]2CN3CCCC[C@H]3[C@H]1C2 SLRCCWJSBJZJBV-AJNGGQMLSA-N 0.000 description 1
- 229960001945 sparteine Drugs 0.000 description 1
- 238000001228 spectrum Methods 0.000 description 1
- 238000009987 spinning Methods 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 239000008223 sterile water Substances 0.000 description 1
- PXQLVRUNWNTZOS-UHFFFAOYSA-N sulfanyl Chemical compound [SH] PXQLVRUNWNTZOS-UHFFFAOYSA-N 0.000 description 1
- 125000005420 sulfonamido group Chemical group S(=O)(=O)(N*)* 0.000 description 1
- 229910052717 sulfur Inorganic materials 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 239000003765 sweetening agent Substances 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 238000004809 thin layer chromatography Methods 0.000 description 1
- 229930192474 thiophene Natural products 0.000 description 1
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 1
- 229940078499 tricalcium phosphate Drugs 0.000 description 1
- 229910000391 tricalcium phosphate Inorganic materials 0.000 description 1
- 235000019731 tricalcium phosphate Nutrition 0.000 description 1
- 125000006493 trifluoromethyl benzyl group Chemical group 0.000 description 1
- 238000001665 trituration Methods 0.000 description 1
- 239000012808 vapor phase Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/12—Antihypertensives
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D209/00—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D209/02—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom condensed with one carbocyclic ring
- C07D209/04—Indoles; Hydrogenated indoles
- C07D209/30—Indoles; Hydrogenated indoles with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, directly attached to carbon atoms of the hetero ring
- C07D209/42—Carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D209/00—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D209/02—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom condensed with one carbocyclic ring
- C07D209/44—Iso-indoles; Hydrogenated iso-indoles
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D217/00—Heterocyclic compounds containing isoquinoline or hydrogenated isoquinoline ring systems
- C07D217/22—Heterocyclic compounds containing isoquinoline or hydrogenated isoquinoline ring systems with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to carbon atoms of the nitrogen-containing ring
- C07D217/26—Carbon atoms having three bonds to hetero atoms with at the most one bond to halogen
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D495/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having sulfur atoms as the only ring hetero atoms
- C07D495/02—Heterocyclic compounds containing in the condensed system at least one hetero ring having sulfur atoms as the only ring hetero atoms in which the condensed system contains two hetero rings
- C07D495/04—Ortho-condensed systems
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Public Health (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Heart & Thoracic Surgery (AREA)
- Cardiology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Oxygen Or Sulfur (AREA)
- Indole Compounds (AREA)
- Quinoline Compounds (AREA)
- Other In-Based Heterocyclic Compounds (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
Abstract
Description
La présente invention se rapporte a de nouveaux imino acides et à leurs procédés de préparation. The present invention relates to new imino acids and to their methods of preparation.
Elle se rapporte plus particulièrement a des imino acides bi-cycliques dont l'atome d'azote est substitué par une chaîne propionylée. It relates more particularly to bi-cyclic imino acids whose nitrogen atom is substituted by a propionylated chain.
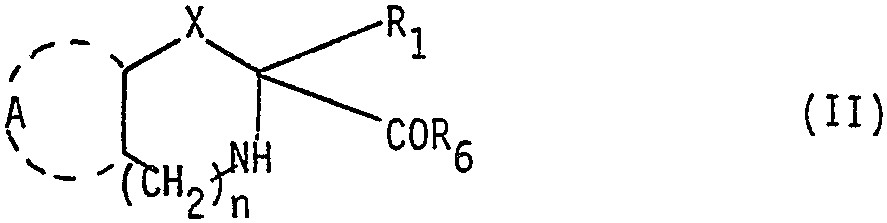
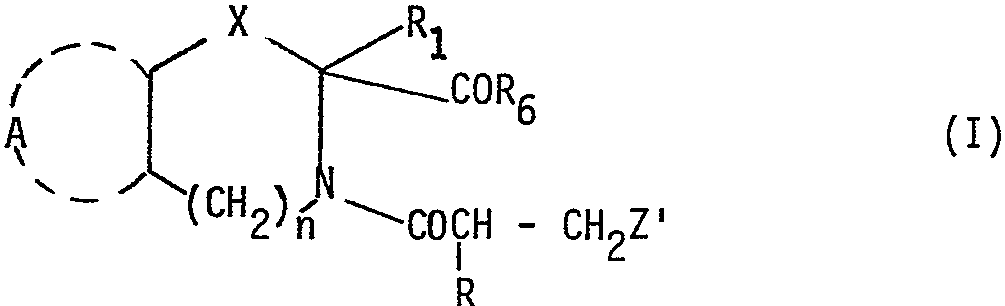
Spécifiquement elle concerne des acides Azabicycloalcane 2-carboxyliques de formule générale I
Specifically, it relates to Azabicycloalkane 2-carboxylic acids of general formula I
dans laquelle A représente une structure cyclique, mono ou bicyclique, saturée, insaturée ou à caractère aromatique, pouvant incorporer un ou plusieurs hétéroatomes, non substituée ou substituée par un ou plusieurs substituants. in which A represents a cyclic, mono or bicyclic, saturated, unsaturated or aromatic structure, which can incorporate one or more heteroatoms, unsubstituted or substituted by one or more substituents.
n est egal à zéro ou est un nombre entier variant de 1 à 3
X est un radical méthylène de formule
n is equal to zero or is an integer varying from 1 to 3
X is a methylene radical of formula
(dans laquelle R8 est de l'hydrogène, un hydroxyle ou un ha- logent et rnest égal à zéro, 1, 2 ou 3), thioalcoylene de la forme -S-CH2,oxyalcoylène de la forme -O-CH2 ou imino alcoylène de la forme
dans laquelle R3 est de l'hydrogène, un radical alcoyle inférieur, phényle, phényl substitué, aralcoyle ou aralcoyle substitué
R1 est de l'hydrogène, un radical alcoyle inférieur, ou un atome d'halogène
R est de l'hydrogène, un radical méthyle, trifluorométhyle, pentafluoroéthyle, hydroxyméthyle ou alcoxy méthyle
Z est un radical carboxy, carbalcoxy, cyano, carboxamide, thiocarboxamide de la forme
(dans laquelle R5 représente de l'hydrogène, un radical alcoyle inférieur, un radical acy- le inférieur ou un radical aroyle) un radical thio de la forme -SR4 dans laquelle R4 est de l'hydrogène, un reste acyle d'acide alcoyl carboxylique, arylcarboxylique ou arylalcoyl carboxylique, ou un reste hydroxycarbamoyl
R6 est un hydroxyle, un alcoxy inférieur ou un groupe amino
L'invention se rapporte également aux sels de composes de formule générale I dans laquelle R6 est un hydroxyle. Ces compo sés peuvent être salifiés par une base minérale ou organique, de préférence thérapeutiquement compatible.(in which R8 is hydrogen, a hydroxyl or a host and rnest equal to zero, 1, 2 or 3), thioalkoylene of the form -S-CH2, oxyalkylene of the form -O-CH2 or iminoalkylene of shape
in which R3 is hydrogen, a lower alkyl, phenyl, substituted phenyl, aralkyl or substituted aralkyl radical
R1 is hydrogen, a lower alkyl radical, or a halogen atom
R is hydrogen, a methyl, trifluoromethyl, pentafluoroethyl, hydroxymethyl or alkoxy methyl radical
Z is a carboxy, carbalkoxy, cyano, carboxamide, thiocarboxamide radical of the form
(in which R5 represents hydrogen, a lower alkyl radical, a lower alkyl radical or an aroyl radical) a thio radical of the form -SR4 in which R4 is hydrogen, an acyl residue of alkyl acid carboxylic, arylcarboxylic or arylalkoyl carboxylic, or a hydroxycarbamoyl residue
R6 is a hydroxyl, lower alkoxy or an amino group
The invention also relates to the salts of compounds of general formula I in which R6 is a hydroxyl. These compounds can be salified with a mineral or organic base, preferably therapeutically compatible.
Parmi les composés de l'invention on pourra distinguer plus particulièrement les sous-groupes suivants, actuellement préférés
10) les acides tetrahydroisoquinoleines -carboxyliques de formule générale IA
Among the compounds of the invention, the following subgroups, which are currently preferred, can be distinguished more particularly
10) tetrahydroisoquinoleine -carboxylic acids of general formula IA
dans laquelle les substituants R6, R, Z et R1 on les significations fournies antérieurement
R2 représente un hydrogène, un halogène ou un hydroxyle
D représente de l'hydrogène, un radical alcoyle inférieur, un alcoxy inférieur, un trifluorométhyl, un trifluorométhoxy, un trifluorométhylthio, un groupe alcoylenedioxy ou un groupe sulfonamido
et p est égal a 1, 2, 3
2") les décahydroisoquinoléines de formule générale 1B
in which the substituents R6, R, Z and R1 have the meanings previously supplied
R2 represents hydrogen, halogen or hydroxyl
D represents hydrogen, a lower alkyl radical, a lower alkoxy, a trifluoromethyl, a trifluoromethoxy, a trifluoromethylthio, an alkylenedioxy group or a sulfonamido group
and p is 1, 2, 3
2 ") decahydroisoquinolines of general formula 1B
dans laquelle les substituants R2, D, R1, R6, R, Z, n et p ont les significations fournies antérieurement 3 ) les B -carbol ibes de formule générale I
in which the substituents R2, D, R1, R6, R, Z, n and p have the meanings provided previously 3) the B -carbol ibes of general formula I
dans laquelle la définition des substituants R, R1, R6, Z,
D et p demeure celle fournie précédemment
et R2 représente un hydroxyle, un alcoyle inférieur, un ou deux halogènes.in which the definition of the substituents R, R1, R6, Z,
D and p remains that previously provided
and R2 represents a hydroxyl, a lower alkyl, one or two halogens.
4 ) les imidazolo tétrahydropyridines de formule générale
4) the tetrahydropyridine imidazolo of general formula
dans laquelle la définition des substituants R, R1, R6, et
Z demeure celle fournie précédemment
et R7 représente de l'hydrogène ou un radical alcoyle inférieur.in which the definition of the substituents R, R1, R6, and
Z remains that previously provided
and R7 represents hydrogen or a lower alkyl radical.
5 ) les indolines de structure 1e
5) indolines of structure 1e
dans laquelle la définition des substituants R1, R2, R, Z,
D et p demeure inchangée.in which the definition of the substituents R1, R2, R, Z,
D and p remain unchanged.
Les composés de formule générale I comportent au moins un atome de Carbone asymétrique. Selon la position des substituants et le degré d'hydrogénation il existe de deux a sept centres d'asymétrie. Les composes racémiques peuvent être dédoublés en leurs mélanges de dîastèrèoisomères ou d'épimères, ou dédoublés en leur énantiomères. The compounds of general formula I contain at least one asymmetric carbon atom. Depending on the position of the substituents and the degree of hydrogenation there are two to seven centers of asymmetry. The racemic compounds can be split into their mixtures of diastereoisomers or epimers, or split into their enantiomers.
D'une maniere préférée on choisit comme matière première des acides (oxa) (Aza) (thia) Azacycloalcoyl carboxyliques ou des acides Azacycloalcoylcarboxyliques ayant la configuration des amino-acides du règne animal. Preferably, the raw material chosen is (oxa) (Aza) (thia) Azacycloalkyl carboxylic acids or Azacycloalkylcarboxylic acids having the amino acid configuration of the animal kingdom.
La chaîne propionique peut comporter un atome de Carbone asymétrique et on préfère utiliser les composés de configuration
D.The propionic chain can comprise an asymmetric carbon atom and it is preferred to use the configuration compounds
D.
On peut présumer que l'activité pharmacologique des composés de formule générale I dépend en partie de la stéréochimie de la molécule. Une seule forme optiquement-active porte l'activité pharmacologique. On peut'donc présumer que les mélanges non dédoublés ou les melanges de diastéréoisomêres n'ont que la moitié ou le quart de l'activité de la configuration moléculaire optiquement-active responsable de l'activité. It can be assumed that the pharmacological activity of the compounds of general formula I depends in part on the stereochemistry of the molecule. Only one optically active form carries the pharmacological activity. It can therefore be assumed that the non-split mixtures or the diastereoisomer mixtures have only half or a quarter of the activity of the optically-active molecular configuration responsible for the activity.
Les composés selon l'invention ainsi que leurs sels sont doués de propriétés pharmacologiques intéressantes. Ils inhibent la transformation du décapeptide Angiotensine I en ltoctapeptide angiotensine II par une inhibition sélective de l'enzyme responsable de cette conversion. The compounds according to the invention and their salts are endowed with interesting pharmacological properties. They inhibit the transformation of the angiotensin I decapeptide into angiotensin II ltoctapeptide by a selective inhibition of the enzyme responsible for this conversion.
Il est reconnu que l'angiotensine existe dans la circulation sous forme d'un précurseur l'Angiotensinogene qu'un enzyme spécifique, la Rénine, transforme en Angiotensine I inactive. La transformation de l'Angiotensine I en Angiotensine II donne naissance à une substance biologique très active sur la pression artérielle en entrainant une hypertension. C'est la raison pour laquelle on a consid-éré qu'un taux trop élevé d'Angiotensine II peut être responsable de diverses formes d'hypertension et notamment da-ns l'hypertension artérielle essentielle.Les composés selon 1 'in- vention ont une action inhibitrice sur la carboxypolypepti dase responsable de la transformation de l'angiotensine I en angiotensine II. Leur emploi en thérapeutique permet donc de réduire ou même de suprimer la formation d'angiotensine II en agissant sur un des mécanismes directement responsables de l'hypertension artérielle chez l'homme. It is recognized that angiotensin exists in the circulation in the form of a precursor the Angiotensinogen that a specific enzyme, Renin, transforms into inactive Angiotensin I. The transformation of Angiotensin I into Angiotensin II gives rise to a very active biological substance on blood pressure, leading to hypertension. This is the reason why it has been considered that an excessively high level of Angiotensin II can be responsible for various forms of hypertension and in particular in essential hypertension. The compounds according to the invention have an inhibitory action on the carboxypolypepti dase responsible for the transformation of angiotensin I into angiotensin II. Their use in therapy therefore makes it possible to reduce or even suppress the formation of angiotensin II by acting on one of the mechanisms directly responsible for arterial hypertension in humans.
L'invention se rapporte donc a l'emploi en thérapeutique des composés de formule générale I et de leurs sels, notamment pour le traitement de l'hypertension artérielle et l'insuffisance cardiaque. The invention therefore relates to the therapeutic use of the compounds of general formula I and their salts, in particular for the treatment of arterial hypertension and heart failure.
L'invention s'étend aussi aux compositions pharmaceutiques renfermant comme principe actif au moins un composé de formule générale I ou un de ses sels d'addition, avec une base minérale ou organique, en association avec un excipient inerte non-toxique pharmaceutiquement acceptable. The invention also extends to pharmaceutical compositions containing as active principle at least one compound of general formula I or one of its addition salts, with an inorganic or organic base, in association with an inert non-toxic excipient which is pharmaceutically acceptable.
En vue de l'emploi en thérapeutique3 les composé de formule générale I ou leurs sels sont présentés sous des formes pharmaceutiques convenant pour l'administration par voie parentérale, buccale, perlinguale ou rectale. Les compositions pharmaceutiques selon l'invention renferment outre le principe actif, un ou plusieurs excipients inertes, non-toxiques convenant pour l'usage pharmaceutique et/ou un agent liant, un agent aromatisant, un agent de délitement, un agent édulcorant, un agent lubrifiant ou bien encore un véhicule liquide ou solide adapté a l'administration par voie parentérale ou rectale. For the purpose of therapeutic use3 the compounds of general formula I or their salts are presented in pharmaceutical forms suitable for administration by the parenteral, buccal, perlingual or rectal route. The pharmaceutical compositions according to the invention contain, in addition to the active principle, one or more inert, non-toxic excipients suitable for pharmaceutical use and / or a binding agent, a flavoring agent, a disintegrating agent, a sweetening agent, an agent lubricant or else a liquid or solid vehicle suitable for parenteral or rectal administration.
Les compositions pharmaceutiques selon l'invention peuvent en outre contenir un autre principe actif d'action complémentaire ou synergistique. The pharmaceutical compositions according to the invention may also contain another active principle of complementary or synergistic action.
Parmi ces derniers principes actifs on pourra citer un diurétique et notamment un saliurétique, comme par exemple un th-ia- vide, un dihydrothiazide, un chlorosulfamide, un acide sulfamide benzolque, un acide benzo furan 2-carboxylique, un acide dihydrobenzofuran 2-carboxylique ou un dérivé de l'acide phénoxy acétique. Des exemples de tels composés sont la N(3'-chloro 4'sulfamyl benzamido)2-méthyl indoline, l'acide éthacrynique, le furosémide. Among these latter active principles, mention may be made of a diuretic and in particular a saliuretic, such as for example a th-ia-vacuum, a dihydrothiazide, a chlorosulfamide, a benzol sulfamide acid, a benzo furan 2-carboxylic acid, a dihydrobenzofuran 2-carboxylic acid. or a derivative of phenoxy acetic acid. Examples of such compounds are N (3'-chloro 4'sulfamyl benzamido) 2-methyl indoline, ethacrynic acid, furosemide.
On pourra également ajouter des substances a-adrénolytiques comme la Prazosine. We can also add a-adrenolytic substances like Prazosin.
La posologie utile peut varier largement en fonction de 1' âge, du poids du patient, de la sévérité de l'indication thérapeutique ainsi que la voie d'administration. La voie d'administration préférée est la voie buccale mais la voie rectale ou parentérale sont également parfaitement appropriées au traitement de l'hypertension. D'une manière générale la posologie unitaire s'échelonnera entre 25 et 250 mg et la posologie journalière entre 100 et 500 mg. The useful dosage can vary widely depending on the age, weight of the patient, the severity of the therapeutic indication and the route of administration. The preferred route of administration is the buccal route but the rectal or parenteral route are also perfectly suitable for the treatment of hypertension. In general, the unit dosage will range between 25 and 250 mg and the daily dosage between 100 and 500 mg.
L'invention comprend également un procédé d'obtention des composés de formule I
The invention also includes a process for obtaining the compounds of formula I
dans laquelle la définition des substituants X, R1, R, Z et n demeure celle fournie précédemment
A représente une structure cyclique, mono ou bicyclique, sa turée, non saturées ou à caractere aromatique, pouvant incorporer un ou plusieurs hétéroatomes, non substituée ou substituée par un ou plusieurs substituants
et R6 est un radical alcoxy inférieur ou amino
dans lequel on soumet un ester d'alcoyle d'acide Azabicycloalkane 2-carboxylique de formule générale II
in which the definition of the substituents X, R1, R, Z and n remains that provided previously
A represents a cyclic structure, mono or bicyclic, its size, unsaturated or of aromatic character, which can incorporate one or more heteroatoms, unsubstituted or substituted by one or more substituents
and R6 is a lower alkoxy or amino radical
in which an alkyl ester of Azabicycloalkane 2-carboxylic acid of general formula II is subjected
dans laquelle la définition des substituants demeure celle mentionnée ci-dessus
et R6 représente un alcoxy inférieur
à l'action d'un acide propionique substitué de formule générale III
dans laquelle Z' représente un radical carbalcoxy, cyano, carboxamide, thiocarboxamide de la forme
(dans laquelle
R5 représente un radical alcoyle inférieur , un radical acyle in rieur ou un radical aroyle) un radical thio de la forme -SR4 dans laquelle R4 est un reste acyle d'acide alcoyl carboxylique ou arylcarboxylique, ou un reste hydroxycarbamoyl
ou un de ses dérivés fonctionnels) pour obtenir un amide de formule générale IV
in which the definition of the substituents remains that mentioned above
and R6 represents a lower alkoxy
to the action of a substituted propionic acid of general formula III
in which Z 'represents a carbalkoxy, cyano, carboxamide, thiocarboxamide radical of the form
(in which
R5 represents a lower alkyl radical, an inner acyl radical or an aroyl radical) a thio radical of the form -SR4 in which R4 is an acyl residue of alkyl carboxylic or arylcarboxylic acid, or a hydroxycarbamoyl residue
or one of its functional derivatives) to obtain an amide of general formula IV
dans laquelle Z' possède les définitions fournies précédemment
R6 représente un radical alcoxy inférieur
et les substituants R1, X, A et n gardent les significations fournies antérieurement
que l'on peut, si désiré, hydrolyser partiellement en dérivé thiolique de formule V
in which Z 'has the definitions provided above
R6 represents a lower alkoxy radical
and the substituents R1, X, A and n keep the meanings provided previously
which can, if desired, be partially hydrolyzed to the thiol derivative of formula V
dans laquelle Z' représente un radical thiol
R6 représente un radical alcoxy inférieur
et les substituants R, X, A et n gardent les significations fournies antérieurement ou saponifier totalement pour-former un dérivé carboxylique de formule VI
in which Z 'represents a thiol radical
R6 represents a lower alkoxy radical
and the substituents R, X, A and n keep the meanings provided previously or completely saponify to form a carboxylic derivative of formula VI
dans laquelle Z' représente un groupe thiol, carboxy, cyano, carboxamide, thiocarboxamide
R6 représente un hydroxyle
et les substituants A, X, R1 et n ont les définitions fournies antérieurement.in which Z 'represents a thiol, carboxy, cyano, carboxamide, thiocarboxamide group
R6 represents a hydroxyl
and the substituents A, X, R1 and n have the definitions previously provided.
Le procédé selon l'invention comprend encore la phase éventuelle d'amidification qui consiste à soumettre un composé de formule IV à l'action de l'ammoniaque concentrée pour former un composé de formule I
The process according to the invention also comprises the optional amidation phase which consists in subjecting a compound of formula IV to the action of concentrated ammonia to form a compound of formula I
dans laquelle R6 est un groupe amino
Z est un radical thiol3 carboxy, cyanothiocarboxamide, hydroxycarbamoyl, carboxamide ou carboxy
et les substituants X, R1, R, A et n ont les mêmes significations que précédemment.in which R6 is an amino group
Z is a thiol3 carboxy, cyanothiocarboxamide, hydroxycarbamoyl, carboxamide or carboxy radical
and the substituents X, R1, R, A and n have the same meanings as above.
Les composés de formule générale I pour lesquels R6 est un hydroxyle, donnent des sels avec les bases minérale ou organiques comme par exemple les sels de sodium, de potassium, de lithium, d'ammonium, de calcium, de magnésium, d'aluminium ; d'alcoyl amine comme la méthylamine, la dimethylamine, la méthyle éthylamine, la di-iso propylamine ou la triéthylamine ; de cycloalcoyl amine comme par exemple la cyclopropylamine ou la dicyclohexyl amine ; de (cycloalcoyl) alcoyl amine comme par exemple la cyclopropyl ethyl amine ; d'amino sucres comme par exemple la glucosamine, la mannosamine, la N-méthylglucamine ; d'amino acides comme la glycine ou l'arginine. The compounds of general formula I for which R6 is a hydroxyl, give salts with mineral or organic bases such as for example the sodium, potassium, lithium, ammonium, calcium, magnesium, aluminum salts; alkyl amine such as methylamine, dimethylamine, methyl ethylamine, di-iso propylamine or triethylamine; cycloalkyl amine such as, for example, cyclopropylamine or dicyclohexyl amine; (cycloalkyl) alkyl amine such as, for example, cyclopropyl ethyl amine; amino sugars such as, for example, glucosamine, mannosamine, N-methylglucamine; amino acids like glycine or arginine.
Selon des modes opératoires préférés, le procédé peut être encore défini comme suit
- la condensation entre l'acide propionique substitué de formule générale III et l'ester d'acide Azabicycloalkane carboxylique de formule générale II s'effectue, en présence ou en 1 'ab- sence de solvant, a température voisine de 00. According to preferred procedures, the process can be further defined as follows
the condensation between the substituted propionic acid of general formula III and the ester of azabicycloalkane carboxylic acid of general formula II is carried out, in the presence or in the absence of solvent, at a temperature close to 00.
- la condensation entre l'acide propionique de formule générale III et l'ester d'acide Azacycloalkane carboxylique de formule générale II s'effectue en présence d'un activateur du carboxyle comme par exemple un di-alcoyl ou di-cycloalcoylcarbodi-imide, l'ethoxyacétylène ou le carbonyl di-imidazole. the condensation between the propionic acid of general formula III and the ester of Azacycloalkane carboxylic acid of general formula II takes place in the presence of a carboxyl activator such as for example a di-alkyl or di-cycloalkylcarbodi-imide, ethoxyacetylene or carbonyl di-imidazole.
- la condensation de l'ester d'acide Azabicycloalkane carboxylique de formule générale II avec l'acide propionique substitué de formule générale III est effectuée en utilisant un dérivé fonctionnel de l'acide propionique de formule générale III comme par exemple un halogénure d'acide, un anhydride ; un anhydride mixte avec le chloroformiate d'éthyle ou un ester d'alcoyle inférieur. the condensation of the azabicycloalkane carboxylic acid ester of general formula II with the substituted propionic acid of general formula III is carried out using a functional derivative of propionic acid of general formula III such as for example an acid halide , an anhydride; a mixed anhydride with ethyl chloroformate or a lower alkyl ester.
- la saponification partielle est effectuée en milieu basique faible comme par exemple en présence d'ammoniaque diluée, d' hydroxyde de calcium ou d'hydroxyde de baryum. - the partial saponification is carried out in a weak basic medium, for example in the presence of dilute ammonia, calcium hydroxide or barium hydroxide.
- l'hydrolyse totale est effectuée en milieu acide comme par exemple en présence d'acide chlorhydrique, d'acide sulfurique ou d'acide perchlorique en milieu aqueux. - total hydrolysis is carried out in an acid medium, for example in the presence of hydrochloric acid, sulfuric acid or perchloric acid in an aqueous medium.
- la séparation des diastéréoisomeres des composés de formule générale I s'effectue de préférence par des méthodes physiques en utilisant la chromatographie en phase vapeur, la chromatogra- phie en phase liquide haute pression ou l'entraînement par saturation réciproque. the separation of the diastereoisomers from the compounds of general formula I is preferably carried out by physical methods using chromatography in vapor phase, chromatography in high pressure liquid phase or entrainment by reciprocal saturation.
- le dédoublement des diastereoisomeres en leurs antipodes optiques s'effectue de préférence par salification des composés de formule générale I pour lesquels R6 est un hydroxyle a l'aide d'une base optiquement active comme par exemple la brucine, la strychnine, la spartéine, l'éphédrine, le d-threo 1-paranitrophényl 2-diméthylamino propane 1,3-diol, la d-glucosamine ou la 1streptamine. the splitting of the diastereoisomers into their optical antipodes is preferably carried out by salification of the compounds of general formula I for which R6 is a hydroxyl using an optically active base such as, for example, brucine, strychnine, sparteine, ephedrine, d-threo 1-paranitrophenyl 2-dimethylamino propane 1,3-diol, d-glucosamine or 1streptamine.
Pour autant que l'invention soit concernée, le terme alcoyle inférieur désigne un radical hydrocarboné saturé ayant 1 à 6 atomes de carbone en chaine droite ou ramifiée. De tels radicaux sont par exemple, le radical méthyle, éthyle, isopropyle, terbutyle, sec pentyle et n hexyle. As far as the invention is concerned, the term lower alkyl denotes a saturated hydrocarbon radical having 1 to 6 carbon atoms in a straight or branched chain. Such radicals are, for example, the methyl, ethyl, isopropyl, terbutyl, dry pentyl and n hexyl radicals.
Le terme aryle désigne un noyau benzénique ou naphtalénique non substitué ou substitué par un ou plusieurs substituants choi sis dans le groupe constitué par les halogènes, un radical alcoxy inférieur, comme par exemple, 3,4-diméthoxy phényl, 2,4-dichlorophényl, m.trifluorométhyl phényl , 3,4,5-triméthoxy phényl, 2,6diméthylphényl, 3,5-diméthoxy phényl, 6,7-diméthoxynaphtyl ou 4hydroxy phényl, il peut aussi être substitué par un alcoyle, un hydroxyle ou un trifluorométhyle
Le terme aryl (alcoyle inférieur) est défini de la même fa çon. Il inclut par exemple, les radicaux 3,4-diméthoxy benzyle, m.trifluorométhyl benzyle, -méthyl benzyl, phényl éthyle, phényl propyle, 8 -méthyl phényl-éthyle, p.chlorobenzyle, methylène dioxy benzyle et benzyle.The term aryl denotes a benzene or naphthalene ring which is unsubstituted or substituted by one or more substituents chosen from the group consisting of halogens, a lower alkoxy radical, such as, for example, 3,4-dimethoxy phenyl, 2,4-dichlorophenyl, m.trifluoromethyl phenyl, 3,4,5-trimethoxy phenyl, 2,6dimethylphenyl, 3,5-dimethoxy phenyl, 6,7-dimethoxynaphtyl or 4hydroxy phenyl, it can also be substituted by an alkyl, a hydroxyl or a trifluoromethyl
The term aryl (lower alkyl) is defined in the same way. It includes, for example, the 3,4-dimethoxy benzyl, m.trifluoromethyl benzyl, -methyl benzyl, phenyl ethyl, phenyl propyl, 8-methyl phenylethyl, p.chlorobenzyl, methylene dioxy benzyl and benzyl radicals.
L'invention comprend aussi un autre procédé d'obtention des acides Azabicycloalcane carboxyliques de formule générale I qui consiste à soumettre un amino acide de formule générale
The invention also includes another method for obtaining Azabicycloalkane carboxylic acids of general formula I which consists in subjecting an amino acid of general formula
dans laquelle A et R6 ont les définitions fournies antérieurement
à l'action d'acide propionique substitué de formule générale III
in which A and R6 have the definitions previously provided
to the action of substituted propionic acid of general formula III
dans laquelle Z' et R sont définis comme précédemment
ou un dérivé fonctionnel de cet acide pour obtenir un peptide de formule générale
in which Z 'and R are defined as above
or a functional derivative of this acid to obtain a peptide of general formula
dans laquelle la définition des substituants X, A, R6, R et
Z demeure inchangée
et soumet celui-ci à l'action du formol en milieu acide pour obtenir un dérivé d'acide bicycloalcane carboxylique de formule générale
in which the definition of the substituents X, A, R6, R and
Z remains unchanged
and subjects it to the action of formalin in an acid medium to obtain a bicycloalkane carboxylic acid derivative of general formula
dans laquelle la définition des substituants A, X, R6, R et
Z' demeure celle fournie precédemment. in which the definition of the substituents A, X, R6, R and
Z 'remains that provided previously.
Parmi les différentes significations de la structure cyclique A on pourra citer des homocycles comme le benzène, le naphta lène ou l'indène ; des hétérocycles incluant un ou plusieurs atomes d'oxygène, de soufre et/ou d'azote comme par exemple la pyridine, la pyrimidine, la tétrahydropyrimi dine, le pyrrole, l'i,mi- dazole, l'imidazoline, le thiophène, le thiachromanne, le benzofuranne, l'indole, 1 'indoline, ltoxazole, l'azepine, la pyrazine ou la quinoxaline. Among the different meanings of the cyclic structure A, mention may be made of homocycles such as benzene, naphtha lene or indene; heterocycles including one or more oxygen, sulfur and / or nitrogen atoms such as, for example, pyridine, pyrimidine, tetrahydropyrimi dine, pyrrole, i, midazole, imidazoline, thiophene, thiachromanne, benzofuran, indole, indoline, ltoxazole, azepine, pyrazine or quinoxaline.
Les exemples suivants illustrent l'invention. Ils ne la limitent en aucune façon. The following examples illustrate the invention. They do not limit it in any way.
EXEMPLE I
Acide 2-(3-mercapto 2-méthyl propionyl) 1,2,3,4-tetrahydro- isoquinoléine 3-carboxylique (mélange de diasteréoisomepes)
Stade A
Acide tetrahydroisoquinoléine (lévogyre) 3-carboxylique.EXAMPLE I
Acid 2- (3-mercapto 2-methyl propionyl) 1,2,3,4-tetrahydro-isoquinoline 3-carboxylic (mixture of diastereoisomepes)
Stage A
Tetrahydroisoquinoline (levorotatory) 3-carboxylic acid.
Dans un ballon a trois tubulures surmonté d'un réfrigérant on introduit 15 g de l-phénylalanine, puis 34 ml d'une solution de formol à 40 ,0 et 105 ml d'acide chlorhydrique concentré. 15 g of l-phenylalanine are introduced into a three-tube flask surmounted by a condenser, followed by 34 ml of a formalin solution containing 40.0 and 105 ml of concentrated hydrochloric acid.
On chauffe pendant 30 minutes au bain-marie bouillant. On obtient ainsi une solution claire, on laisse revenir le milieu réactionnel à température ambiante et on ajoute alors 15 ml de formol et 30 ml d'acide chlorhydrique concentré. On chauffe en suite 3 heures au reflux du solvant. On laisse refroidir puis sépare le précipité par filtration. Après essorage on le reprend par 200 ml d'eau bouillante et 400 ml d'éthanol chaud. On réunit les solutions qu'on neutralise par addition d'ammoniaque à 10 %. It is heated for 30 minutes in a boiling water bath. A clear solution is thus obtained, the reaction medium is allowed to return to ambient temperature and then 15 ml of formalin and 30 ml of concentrated hydrochloric acid are added. Then heated 3 hours at reflux of the solvent. The mixture is allowed to cool and then the precipitate is separated by filtration. After spinning, it is taken up in 200 ml of boiling water and 400 ml of hot ethanol. The solutions are combined and neutralized by adding 10% ammonia.
L'acide tétrahydroisoquinoléine 3~carboxylique cristallise. Tetrahydroisoquinoline 3 ~ carboxylic acid crystallizes.
On laisse reposer le mélange cristallin une nuit en glacière puis separe le précipité que l'on essore et lave à l'éthanol. On recueille ainsi 1793 de produit brut. On prélève un échantillon pour des fins analytiques. Le produit est séché sous vide phosphorique.The crystalline mixture is left to stand overnight in a cooler and then separates the precipitate which is drained and washed with ethanol. 1793 of raw product are thus collected. A sample is taken for analytical purposes. The product is dried under phosphoric vacuum.
Analyse : C10 H1l 2 N = 177
C H
Calculé 67.78 6.26 7.90
Trouvé 66.87 6.20 7.96
Spectre IR
NH2+ bande à 2800-2400 cm -1
COO bande carbonyle à 1630 cm 1
Pouvoir rotatoire
a D = - 108 (c = 2,2 NaOH normal)
Stade B
Chlorhydrate de tétrahydroisoquinoléine 3-carboxylate de méthyle.Analysis: C10 H1l 2 N = 177
CH
Calculated 67.78 6.26 7.90
Found 66.87 6.20 7.96
IR spectrum
NH2 + band at 2800-2400 cm -1
COO carbonyl strip at 1630 cm 1
Rotating power
a D = - 108 (c = 2.2 NaOH normal)
Stage B
Methyl tetrahydroisoquinoline hydrochloride 3-carboxylate.
Dans un ballon a trois tubulures on charge successivement 5 g d'acide tétrahydroisoquinoléine 3-carboxylique et 30 ml de méthanol . A cette suspension on ajoute par coulée tout en evitant que la température ne dépasse 0,+ 5 , 6g de chlorure de thionyle. 5 g of a tetrahydroisoquinoline 3-carboxylic acid and 30 ml of methanol are successively charged into a three-tube flask. To this suspension is added by pouring while avoiding that the temperature does not exceed 0, + 5.6 g of thionyl chloride.
L'addition dure environ 10 minutes. Après achevement de cette addition on maintient l'agitation pendant 2 heures à température ambiante, puis on porte au reflux-du solvant 1 heure et demie.The addition takes about 10 minutes. After this addition is complete, stirring is continued for 2 hours at ambient temperature, then the solvent is refluxed for 1 hour and a half.
Une fois le mélange complètement dissout, on arrète le chauffage puis évapore a sec. Le résidu est repris au méthanol à trois reprises et amené ensuite à sec. On recueille enfin 8 g de cristaux incolores que l'on purifie par trituration avec de l'éther. On sé pare les cristaux par filtration, les essore, les lave à l'éther et les sèche.On obtient ainsi 694 de Chlorhydrate de tétrahydro- isoquinoléine 3-carboxylate de méthyle
Analyse C10 H13 N02 ClH = 227.69
C H N Cl %
Calculé 58,03 6,20 6,15 15,57
Trouvé 57,79 6,46 6,38 15,67
Spectre IR
bande carbonyle à 1735 cm 1
bande NH2 à 2800-2400 cm 1
Stade C
2-(3 acetylthio 2-methyl propionyl) 3-methoxy carbonyl 1,2, 3,4-tétrahydroisouinoléine. Once the mixture is completely dissolved, the heating is stopped and then evaporated to dryness. The residue is taken up in methanol three times and then brought to dryness. Finally collected 8 g of colorless crystals which are purified by trituration with ether. The crystals are separated by filtration, drained, washed with ether and dried. This gives 694 of methyl tetrahydroisoquinoline hydrochloride 3-carboxylate.
Analysis C10 H13 N02 ClH = 227.69
CHN Cl%
Calculated 58.03 6.20 6.15 15.57
Found 57.79 6.46 6.38 15.67
IR spectrum
carbonyl band at 1735 cm 1
NH2 band at 2800-2400 cm 1
Stage C
2- (3 acetylthio 2-methyl propionyl) 3-methoxy carbonyl 1,2, 3,4-tetrahydroisouinoline.
On introduit dans un récipient pourvu d'une agitation mécanique 6 g de chlorhydrate de tétrahydroisoquinoléine 3-carboxylate de méthyle, 1,4 ml de triéthyl ami ne et 200 ml de chlorure de méthylène. On refroidit la solution au bain d'eau glacée puis on ajoute 3g96 de dicyclohexyl carbodimide, 1g47 d'hydroxybenzotriazole et 196 d'acide 2-méthyl 3-acétylthiopropionique dl. On maintient une agitation pendant 12 heures à temperature ambiante. On filtre le précipité de dicyclohexyl Urée puis on évapore a sec le filtrat. On reprend le résidu sec par 100 ml d'acétate d'éthyle et filtre l'insoluble puis lave le filtrat avec une solution saturée de chlorure de sodium, puis avec une solution d'acide citrique à 10 % et enfin avec une solution saturée de chlorure de sodium. On sèche l'acétate d'éthyle puis évapore a sec. On obtient ainsi 3 g d'un produit huileux, pratiquement homogene en chromatographie en couche mince. 6 g of tetrahydroisoquinoline hydrochloride 3-methyl carboxylate, 1.4 ml of triethyl amine and 200 ml of methylene chloride are introduced into a container provided with mechanical stirring. The solution is cooled in an ice-water bath and then 3g96 of dicyclohexyl carbodimide, 1g47 of hydroxybenzotriazole and 196 of 2-methyl 3-acetylthiopropionic acid dl are added. Stirring is continued for 12 hours at room temperature. The precipitate of dicyclohexyl urea is filtered and then the filtrate is evaporated to dryness. The dry residue is taken up in 100 ml of ethyl acetate and the insoluble material is filtered, then the filtrate is washed with a saturated sodium chloride solution, then with a 10% citric acid solution and finally with a saturated solution of sodium chloride. The ethyl acetate is dried and then evaporated to dryness. 3 g of an oily product are thus obtained, practically homogeneous in thin layer chromatography.
Le produit est purifié par chromatographie sur silice et élution par un mélange benzène-acétate d'éthyle. On élimine les fractions de tête. Les fractions médianes fournissent par évaporation 198 de 2-(3-acetylthio 2-méthylpropionyl) 3-méthoxy carbonyl 1,2,3,4-têtrahydroisoquinolène. The product is purified by chromatography on silica and elution with a benzene-ethyl acetate mixture. The head fractions are eliminated. The middle fractions provide by evaporation 198 of 2- (3-acetylthio 2-methylpropionyl) 3-methoxy carbonyl 1,2,3,4-tetrahydroisoquinolene.
Spectre RMN
compatible avec la structure
triplet 5,45 ppm
4 protons phényl à 7,25 ppm
3 protons acétyl à 3,65 ppm
5 protons de carbones saturés entre 3 et 3,5 ppm.NMR spectrum
compatible with structure
triplet 5.45 ppm
4 phenyl protons at 7.25 ppm
3 acetyl protons at 3.65 ppm
5 protons of saturated carbon between 3 and 3.5 ppm.
en CPV
présence d'un pic principal unique pur à 98 % environ dans les conditions opératoires utilisées.in CPV
presence of a single main peak approximately 98% pure under the operating conditions used.
Stade D
Acide 2-(3-mercapto 2-méthyl propionyl) 1,2,3,4-tetrahydroisoquinoléine 3-L carboxylique.Stage D
2- (3-Mercapto 2-methyl propionyl) 1,2,3,4-tetrahydroisoquinoline 3-L carboxylic acid.
Dans un ballon à trois tubulures pourvu d'une agitation magnétique et d'un barbottage de gaz inerte on introduit 1,7 g de 2-(3-acétylthio 2-méthyl propionyl) 3-methoxycarbonyl 1,2,3,4tetrahydro isoquinoléine, 11 ml de potasse normale et 30 ml d'é- thanol. On agite pendant 24 heures à temperature ambiante puis on neutralise exactement par addition d'acide chlorhydrique. On concentre la solution éthanolique que l'on epuise avec de l'acetate d'éthyle. La solution dans l'acétate d'éthyle. La solution dans l'acétate d'éthyle est lavée a l'eau, séchée sur sulfate de calcium et évaporée a sec. On reprend le résidu avec 20 ml d'éther isopropylique et filtre l'insoluble. On amène le filtrat à sec.On obtient ainsi 1g2 de produit pur, presentant en CCM une tache très largement dominante. 1.7 g of 2- (3-acetylthio 2-methyl propionyl) 3-methoxycarbonyl 1,2,3,4tetrahydro isoquinoline are introduced into a three-tube flask provided with magnetic stirring and a bubbling of inert gas. 11 ml of normal potash and 30 ml of ethanol. Stirred for 24 hours at room temperature and then neutralized exactly by adding hydrochloric acid. The ethanolic solution is concentrated which is exhausted with ethyl acetate. The solution in ethyl acetate. The ethyl acetate solution is washed with water, dried over calcium sulphate and evaporated to dryness. The residue is taken up in 20 ml of isopropyl ether and the insoluble material is filtered. The filtrate is brought to dryness, thus obtaining 1 g 2 of pure product, presenting in CCM a very largely dominant spot.
Analyse C14 H17 N03S L 279,36
C H N
Calcule 60,19 6,13 5,01 11,48
Trouvé 60,48 6,49 4,62 11,00
Cet acide est soluble dans les solutions diluées de soude.Analysis C14 H17 N03S L 279.36
CHN
Calculate 60.19 6.13 5.01 11.48
Found 60.48 6.49 4.62 11.00
This acid is soluble in dilute soda solutions.
Par evaporation sous pression réduite on isole le sel de sodium de l'acide 2(3-mercapto 2-méthyl propionyl) 1,2,3,4-tetrahydroisoquinoléine 3-L.carboxylique.By evaporation under reduced pressure, the sodium salt of 2 (3-mercapto 2-methyl propionyl) 1,2,3,4-tetrahydroisoquinoline 3-L.carboxylic acid is isolated.
EXEMPLE II
Acide 2-(3-mercapto 2-methylpropionyl) cis décahydroisoqui noléine 3-carboxylique.EXAMPLE II
2- (3-Mercapto 2-methylpropionyl) cis decahydroisoqui noline 3-carboxylic acid.
En utilisant le mode opératoire de l'exemple I au départ de l'acide cis décahydro isoquinoléine 3-carboxylique obtenu selon la méthode d'hydrogenation décrite par Rapala J.of Am. Chem. Soc. Using the procedure of Example I starting from cis decahydro-isoquinoline 3-carboxylic acid obtained according to the hydrogenation method described by Rapala J. of Am. Chem. Soc.
79 (1957) 3770, et de l'acide 3-acetylthio 2-méthyl propionique, on obtient successivement
- l'ester méthylique de l'acide cis décahydroisoquinoléine 3-carboxylique
- le 2-(3-acétylthio 2-méthyl propionyl) cis décahydroisoquinoléine 3-carboxylate de méthyle
- l'acide 2-(3-acétylthio 2-méthyl propionyl) cis decahydroisoquinoléine 3-carboxylique.79 (1957) 3770, and 3-acetylthio 2-methyl propionic acid, successively obtained
- the methyl ester of cis decahydroisoquinoline 3-carboxylic acid
- 2- (3-acetylthio 2-methyl propionyl) cis decahydroisoquinoline 3-methyl carboxylate
- 2- (3-acetylthio 2-methyl propionyl) cis decahydroisoquinoline 3-carboxylic acid.
Le produit est constitue d'un mélange de diastéréosomeres. The product consists of a mixture of diastereomers.
Spectre IR : conforme à la structure
Spectre RMV : intégration des protons en accord avec la structure.IR spectrum: conforms to the structure
RMV spectrum: integration of protons in agreement with the structure.
Analyse C13 H21 N03S =- 271,38
C H N
Calculé 57,78 7,80 5,16 11,82
Trouvé 57,94 7,12 5,24 11,66
EXEMPLE III
Acide 5'-(3"-mercapto 2"-methylpropionyl) 1',2',5',6' tetrahydro (pyridino 3',4'-4,5-imidazolyl) 6-carboxylique
En utilisant le même mode opératoire qu'a l'exemple I au dé- part de l'acide 1', 2', 5', 6' tétrahydro (pyridino 3', 4' - 4,5 imidazolyl) 6'-carboxylique préparé selon la méthode décrite par
Ackermann H.S. Z.f. Phys.Chem. 284 (1949) 131, et de l'acide 3-acétylthio 2-méthyl propionique, on obtient successivement
- le 1', 2', 5', 6' tétrahydro (pyridino 3', 4'-4,5 imidazolyl) 6'-carboxylate d'ethyle
- le 5'-(,3"-acéthylthio 2"-méthyl propionyl) 1', 2', 5', 6' tetrahydro (pyridino 3', 4' - 4,5 imidazolyl) 6'-carboxylate d' éthyle
- l'acide 5'-(3"-thio 2"-methylpropionyl) 1', 2', 5', 6' téerahydro (pyridino 3', 4'-4,5-imidazolyl) 6'-carboxylique
Analyse C10 H12 N3 03 S = 254,29
C H N
Cal cule 47,23 4,76 16,52 12,61
Trouvé 47,42 4,68 16,73 12,38
EXEMPLE IV
Acide 2- 0(3' mercapto 2'-trifluoromethylpropionyl) 1,2,3,4 tetrahydro 9H| pyrido- (3,4b) indole 3-carboxylique.Analysis C13 H21 N03S = - 271.38
CHN
Calculated 57.78 7.80 5.16 11.82
Found 57.94 7.12 5.24 11.66
EXAMPLE III
5 '- (3 "-mercapto 2" -methylpropionyl) acid 1', 2 ', 5', 6 'tetrahydro (pyridino 3', 4'-4,5-imidazolyl) 6-carboxylic
Using the same procedure as in Example I, starting with the acid 1 ', 2', 5 ', 6' tetrahydro (pyridino 3 ', 4' - 4,5 imidazolyl) 6'-carboxylic prepared according to the method described by
Ackermann HSZf Phys.Chem. 284 (1949) 131, and 3-acetylthio 2-methyl propionic acid, successively obtained
- 1 ', 2', 5 ', 6' tetrahydro (pyridino 3 ', 4'-4,5 imidazolyl) 6'-ethyl carboxylate
- 5 '- (, 3 "-acethylthio 2" -methyl propionyl) 1', 2 ', 5', 6 'tetrahydro (pyridino 3', 4 '- 4,5 imidazolyl) 6'-ethyl carboxylate
- 5 '- (3 "-thio 2" -methylpropionyl) acid 1', 2 ', 5', 6 'téerahydro (pyridino 3', 4'-4,5-imidazolyl) 6'-carboxylic
Analysis C10 H12 N3 03 S = 254.29
CHN
Calculation 47.23 4.76 16.52 12.61
Found 47.42 4.68 16.73 12.38
EXAMPLE IV
Acid 2- 0 (3 'mercapto 2'-trifluoromethylpropionyl) 1,2,3,4 tetrahydro 9H | pyrido- (3,4b) indole 3-carboxylic.
En utilisant le mode opératoire de l'exemple I au départ de l'acide(1,2,3,4-tetrahydro [9H] pyrido (3,4-b) indole)3-carboxylique, obtenu selon la méthode decrite par A. Brossi J. Med,
Chem. 16 (1973) 419 et de l'acide 3-acétylthio 2-trifluorométhyl propionique (obtenu selon le procédé décrit dans le brevet belge 872,972), on obtient successivement
- le (tetrahydro ho pyrido (3,4-b) indole) 3-carboxylate de méthyle
- le 2-(3'-acetylthio 2'-trifluoromethyl propionyl) 1,2,3,4tetrahydro [9H] pyrido (3,4-b) indole 3-carboxylate de méthyle
- l'acide 2-[(3'-mercapto 2'-trifluoromethylpropionyl) 1,2,3, 4-tetrahydro [9Hi pyrido (3,4-b) indoles 3-carboxylique.Using the procedure of Example I starting from the acid (1,2,3,4-tetrahydro [9H] pyrido (3,4-b) indole) 3-carboxylic, obtained according to the method described by A Brossi J. Med,
Chem. 16 (1973) 419 and 3-acetylthio 2-trifluoromethyl propionic acid (obtained according to the process described in Belgian patent 872,972), successively obtained
- methyl tetrahydro ho pyrido (3,4-b) indole) 3-carboxylate
- methyl 2- (3'-acetylthio 2'-trifluoromethyl propionyl) 1,2,3,4tetrahydro [9H] pyrido (3,4-b) indole 3-carboxylate
- 2 - [(3'-mercapto 2'-trifluoromethylpropionyl) 1,2,3,4-tetrahydro [9Hi pyrido (3,4-b) indoles 3-carboxylic acid.
- le sel de dicyclohexylamine de l'acide 2-3'-mercapto 2'trifluoromethyl propionyl) 1,2,3,4-tetrahydro [9H] pyrido (3,4-b) indole]3-carboxylique (mélange de diastéréoisomères). - the dicyclohexylamine salt of 2-3'-mercapto 2'trifluoromethyl propionyl) 1,2,3,4-tetrahydro [9H] pyrido (3,4-b) indole] 3-carboxylic acid (mixture of diastereoisomers) .
Analyse C16 H14 F3 N2 02S + C12 H23N = 533,69
C H N
Calculé 63,02 6,99 8,06 6,01
Trouvé 62,54 7,08 8,22 5,79
EXEMPLE V
Acide 2-(3'-cyano 2'-méthylpropionyl) 3 xmethyl 6,7-dihydroxy 1,2,3,4-tetrahydro isoquinoléine 3-carboxylique (mélange de diastéréoisomères). Analysis C16 H14 F3 N2 02S + C12 H23N = 533.69
CHN
Calculated 63.02 6.99 8.06 6.01
Found 62.54 7.08 8.22 5.79
EXAMPLE V
2- (3'-cyano 2'-methylpropionyl) 3 xmethyl 6,7-dihydroxy 1,2,3,4-tetrahydro isoquinoline 3-carboxylic acid (mixture of diastereoisomers).
En opérant selon le mode opératoire de l'-exemple I au départ de l'acide 6,7-dShydroxy 3 a-methyl 1,2,3,4-tétrahydroisoquinoleine 3(L)-carboxylique (décrit par Rachlin Scheur Tétrahedron Letters (1968), 39 4163), on prépare l'ester méthylique de l'acide 6,7-dihydroxy 3 -methyl 1,2,3,4-tétrahydro iso quinoleine 3(L)-carboxylique que l'on condense avec le chlorure de l'acide 3-cyano 2-methylpropionique en solution dans le dime- thyl formami de. By operating according to the procedure of Example I starting from 6,7-dShydroxy 3 a-methyl 1,2,3,4-tetrahydroisoquinoleine 3 (L) -carboxylic acid (described by Rachlin Scheur Tétrahedron Letters ( 1968), 39 4163), the methyl ester of 6,7-dihydroxy 3 -methyl 1,2,3,4-tetrahydro iso quinoleine 3 (L) -carboxylic acid is prepared which is condensed with the chloride of 3-cyano 2-methylpropionic acid in solution in dime-methyl formami.
On obtient ainsi l'ester méthylique de l'acide 2-(3'-cyano 2'-methylpropionyl) 3 a-méthyl 6,7-dihydroxy 1,2,3,4-tétrahydroisoquinoléine 3(L) carboxylique. This gives the methyl ester of 2- (3'-cyano 2'-methylpropionyl) 3 a-methyl 6,7-dihydroxy 1,2,3,4-tetrahydroisoquinoline 3 (L) carboxylic acid.
L'ester méthylique est saponifié par agitation avec une solution de baryte à temperature ordinaire. La solution est ensuite saturée par du gaz carbonique. On sépare le précipité par filtration, le lave à l'eau à plusieurs reprises, réunit les phases aqueuses et les amène à sec sous vide. The methyl ester is saponified by stirring with a baryte solution at ordinary temperature. The solution is then saturated with carbon dioxide. The precipitate is separated by filtration, washed with water several times, the aqueous phases are combined and brought to dryness under vacuum.
Le residu sec est repris dans l'acétate d'éthyle par chaud et froid. The dry residue is taken up in ethyl acetate by hot and cold.
L'acide 1,2,3,4-tétrahydro 2-(3'-cyano 2'-méthyl propionyl) 3 améthyl 6,7-dihydroxy isoquinoléine 3 (L)-carboxylique est obtenu sous forme de cristaux légèrement jaunâtres. 1,2,3,4-tetrahydro 2- (3'-cyano 2'-methyl propionyl) 3 amethyl 6,7-dihydroxy isoquinoline 3 (L) -carboxylic acid is obtained in the form of slightly yellowish crystals.
Analyse C16 H18 O, N2 = 318,17
C H
Calculé 60,35 5,70 8,80
Trouvé 59,84 5,79 8,97
EXEMPLE VI
Etude pharmacologique des composés de l'invention.Analysis C16 H18 O, N2 = 318.17
CH
Calculated 60.35 5.70 8.80
Found 59.84 5.79 8.97
EXAMPLE VI
Pharmacological study of the compounds of the invention.
Les composes selon l'invention sont testés par administration à des lots de chiens de race Mongrel, non anesthésiés, maintenus en respiration forcée à l'aide d'un appareil respiratoire
Mark VII Bird.The compounds according to the invention are tested by administration to batches of dogs of the Mongrel breed, not anesthetized, maintained in forced breathing using a respiratory device.
Mark VII Bird.
La pression artérielle des chiens a été mesuree par un capteur de pression Statham P23Db après cathéterisation de l'aorte par l'intermédiaire de l'artère fémorale. L'enregistrement est réalise par un appareil enregistreur Brush 400. The dogs' blood pressure was measured by a Statham P23Db pressure sensor after catheterization of the aorta via the femoral artery. The recording is carried out by a Brush 400 recording device.
L'angiotensine I et l'angiotensine Il sont injectées aux animaus par voie intra-veineuse à la dose de 0,3 1Kg. On établit une courbe dose/activé pour chacune de ces hormones. On administre ensuite les composés selon l'invention par voie buccale à une dose correspondant a 0,023 M/Kg de produit dédoublé. On établit ensuite une deuxieme courbe dose/activité pour l'angiotensine I et pour l'angiotensine II apres administration des produits es sayés. Angiotensin I and angiotensin II are injected into animals intravenously at a dose of 0.3 1Kg. A dose / activated curve is established for each of these hormones. The compounds according to the invention are then administered by the oral route at a dose corresponding to 0.023 M / Kg of split product. A second dose / activity curve is then established for angiotensin I and for angiotensin II after administration of the trial products.
Les résultats suivants ont éte obtenus en comparaison avec ceux obtenus avec le captopril administré dans les mêmes conditions. The following results were obtained in comparison with those obtained with captopril administered under the same conditions.
composé de l'exemple temps % d'inhibition
1 15 mn 80
30 mn 90
60 mn 60
90 44
2 heures 44
3 heures 29
4 heures 18
5 heures 16
6 heures 20
composés de l'exemple
2 15 mn 75
30 mn 89
60 mn 94
90 mn 58
2 heures 52
3 heures 41
4 heures 28
Captopril 15 mn 100 %
(0,023 M/kg = 5 mg/kg)
30 mn 92
60 mn 88
90 mn 80
2 heures 62
3 heures 51
4 heures 34
5 heures 28
6 heures 26
Les produits selon l'invention sont donc pratiquement aussi actifs que le captopril par voie buccale à dose équimoléculaires.composed of the example time% inhibition
1 15 min 80
30 min 90
60 mins 60
90 44
2 hours 44
3:29
4:18
5:16 a.m.
6:20 a.m.
compounds of the example
2 15 min 75
30 min 89
60 min 94
90 min 58
2 hours 52
3 hours 41
4:28 a.m.
Captopril 15 mins 100%
(0.023 M / kg = 5 mg / kg)
30 min 92
60 min 88
90 min 80
2 hours 62
3 hours 51
4 hours 34
5:28 a.m.
6:26 a.m.
The products according to the invention are therefore practically as active as captopril by the oral route in equimolecular doses.
EXEMPLE VII
Solution injectable pour perfusion veineuse
2-(3'-mercapto 2'-methylpropionyl) tétrahydroisoquinoléine 3-carboxylate de dicyclohexyl ami ne Logo15
N-(3'-sulfamoyl 4'-chlorobenzamido) 2-methyl indoline 09075 p.hydroxybenzoate de méthyle Og15 p.hydroxybenzoate de propyle Ogl5 chlorure de sodium eau distillée qsp 1000 ml
EXEMPLE VIII
Solution injectable pour perfusion veineuse
2-(3'-mercapto 2'-méthylpropionyl) tétrahydroisoquinoléine 3-carboxylate de dicyclohexylamine / Og015 acide 5-(aminosulfonyl) 4-chloro 2 (2-furanylmethyl) amino] benzolque 09045 p.hydroxybenzoate de méthyle Og15
P.hydroxybenzoate de propyle Og15 chlorure de lithium 6g5 eau distillee qsp 1000 ml
EXEMPLE IX
solution injectable pour perfusion intraveineuse
N-(3-mercapto 2-méthyl propionyl) décahydro isoquinoléine 3-carboxylate de di cyclohexyl amine 09015
N-(3'sulfamoyl 4'-chlorobenzamido) 2-méthyl indoline 09075 p.hydroxybenzoate de methyle Yoga5 p.hydroxybenzoate de propyle Og15 chlorure de lithium 695 eau stérile qsp 1000 ml
EXEMPLE X
comprimés à 50 mg de principe actif
Acide 2-(3'-mercapto 2-méthylpropionyl) tétrahydro isoquinoléine 3-carboxylique acide 5-aminosulfonyl 50 g 4-chloro 2-(2-furanyl méthylamino)benzoique 45 g amidon de blé 80 g amidon de mais 70 g caséine formolée 20 g stéarate de magnésium 15 g talc 20 g pour 1000 comprimés d'un poids moyen de 09300
EXEMPLE XI
comprimes à 100 mg de principe actif
Acide 2-(3mercapto 2-méthyl propionyl) décahydroisoquinoléine 3-carboxylique 100 g phosphate tricalcique 125 g amidon de blé 35 g carboxyméthylamidon 25 g caséine formolée 5g silice colloïdale 5g silicate de magnésium 25 g talc 20 g pour 1000 comprimés d'un poids moyen de 0g25 EXAMPLE VII
Solution for injection for venous infusion
2- (3'-mercapto 2'-methylpropionyl) tetrahydroisoquinoline 3-carboxylate dicyclohexyl ami ne Logo15
N- (3'-sulfamoyl 4'-chlorobenzamido) 2-methyl indoline 09075 p.hydroxybenzoate Og15 p.hydroxybenzoate propyl Ogl5 sodium chloride distilled water qs 1000 ml
EXAMPLE VIII
Solution for injection for venous infusion
2- (3'-mercapto 2'-methylpropionyl) tetrahydroisoquinoline dicyclohexylamine 3-carboxylate / Og015 acid 5- (aminosulfonyl) 4-chloro 2 (2-furanylmethyl) amino] benzolque 09045 p.hydroxybenzoate Og15
Propyl p. Hydroxybenzoate Og15 lithium chloride 6g5 distilled water qs 1000 ml
EXAMPLE IX
solution for injection for intravenous infusion
N- (3-mercapto 2-methyl propionyl) decahydro isoquinoline 3-carboxylate di cyclohexyl amine 09015
N- (3'sulfamoyl 4'-chlorobenzamido) 2-methyl indoline 09075 p.hydroxybenzoate Yoga5 p.propyl hydroxybenzoate Og15 lithium chloride 695 sterile water qs 1000 ml
EXAMPLE X
50 mg tablets of active ingredient
2- (3'-mercapto 2-methylpropionyl) tetrahydro isoquinoline 3-carboxylic acid 5-aminosulfonyl 50 g 4-chloro 2- (2-furanyl methylamino) benzoic 45 g wheat starch 80 g corn starch 70 g formalin casein 20 g magnesium stearate 15 g talc 20 g per 1000 tablets with an average weight of 09300
EXAMPLE XI
100 mg tablets of active ingredient
2- (3mercapto 2-methyl propionyl) decahydroisoquinoline 3-carboxylic acid 100 g tricalcium phosphate 125 g wheat starch 35 g carboxymethyl starch 25 g formalin casein 5g colloidal silica 5g magnesium silicate 25 g talc 20 g per 1000 tablets of average weight 0g25
Claims (2)
Priority Applications (14)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR7930046A FR2470767A1 (en) | 1979-12-07 | 1979-12-07 | N-Acyl-2-carboxy fused ring heterocyclic cpds. - useful for inhibiting angiotensin converting enzyme and enkephalin(s) |
| FR8016875A FR2487829A2 (en) | 1979-12-07 | 1980-07-31 | NOVEL SUBSTITUTED IMINO ACIDS, PROCESSES FOR THEIR PREPARATION AND THEIR USE AS AN ENZYME INHIBITOR |
| EP80401728A EP0031741A1 (en) | 1979-12-07 | 1980-12-03 | Substituted imino-acids, process for their preparation and their use as enzyme inhibitors |
| US06/212,607 US4404206A (en) | 1979-12-07 | 1980-12-03 | Substituted iminoacid derivatives, process for preparing them and their use as enzyme inhibitors |
| ES497517A ES497517A0 (en) | 1979-12-07 | 1980-12-05 | PROCEDURE FOR THE PREPARATION OF NEW IMINOACIDS AND THEIR DERIVATIVES. |
| AU65123/80A AU539414B2 (en) | 1979-12-07 | 1980-12-05 | 2-carboxylic azabicydoalkanes |
| JP17195780A JPS5692270A (en) | 1979-12-07 | 1980-12-05 | Novel bicyclic iminooacids* their manufacture and medicine containing them |
| CA000366326A CA1209985A (en) | 1979-12-07 | 1980-12-08 | Preparation of novel substituted imino-acids |
| US06/308,234 US4508729A (en) | 1979-12-07 | 1981-10-02 | Substituted iminodiacids, their preparation and pharmaceutical compositions containing them |
| US06/420,005 US4565819A (en) | 1979-12-07 | 1982-09-20 | Substituted imino diacids, their preparation and pharmaceutical compositions which contain them |
| US06/659,275 US4616029A (en) | 1979-12-07 | 1984-10-10 | Perhydromdole-2-carboxylic acids as antihypertensives |
| US06/659,276 US4616031A (en) | 1979-12-07 | 1984-10-10 | Perhydroindole-2-carboxylic acids as antihypertensives |
| US06/659,274 US4644008A (en) | 1979-12-07 | 1984-10-10 | Perhydroindole-2-carboxylic acids as antihypertensives |
| US06/679,320 US4616030A (en) | 1979-12-07 | 1984-12-06 | Perhydroindole-2-carboxylic acids as antihypertensives |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR7930046A FR2470767A1 (en) | 1979-12-07 | 1979-12-07 | N-Acyl-2-carboxy fused ring heterocyclic cpds. - useful for inhibiting angiotensin converting enzyme and enkephalin(s) |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| FR2470767A1 true FR2470767A1 (en) | 1981-06-12 |
| FR2470767B1 FR2470767B1 (en) | 1982-04-30 |
Family
ID=9232491
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| FR7930046A Granted FR2470767A1 (en) | 1979-12-07 | 1979-12-07 | N-Acyl-2-carboxy fused ring heterocyclic cpds. - useful for inhibiting angiotensin converting enzyme and enkephalin(s) |
Country Status (2)
| Country | Link |
|---|---|
| JP (1) | JPS5692270A (en) |
| FR (1) | FR2470767A1 (en) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2456733A1 (en) * | 1979-05-16 | 1980-12-12 | Morton Norwich Products Inc | NEW ACIDS 2- (MERCAPTO-1-OXO (LOWER ALKYL)) - 1,2,3,4-TETRAHYDROISOQUINOLEINE-3-CARBOXYLIQUES, ESPECIALLY USEFUL AS INHIBITORS OF THE ANGIOTENSIN I CONVERSION ENZYME |
| FR2503155A2 (en) * | 1980-10-02 | 1982-10-08 | Science Union & Cie | NOVEL SUBSTITUTED IMINO DIACIDES, PROCESSES FOR THEIR PREPARATION AND THEIR USE AS AN ENZYME INHIBITOR |
| US4460775A (en) * | 1979-08-09 | 1984-07-17 | Norwich Eaton Pharmaceuticals, Inc. | 2-(3-Mercapto-1-oxopropyl)-1,2,3,4-tetrahydroisoquinoline-1-carboxylic acid derivatives |
| US4479963A (en) * | 1981-02-17 | 1984-10-30 | Ciba-Geigy Corporation | 1-Carboxyalkanoylindoline-2-carboxylic acids |
| US4665087A (en) * | 1982-02-22 | 1987-05-12 | Ciba-Geigy Corporation | 1-(carbamyl, thiocarbamyl, and iminocarbamyl)-indoline derivatives |
| US5158959A (en) * | 1980-08-30 | 1992-10-27 | Hoechst Aktiengesellschaft | Decahydroisoquinoline carboxylic acids |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4350704A (en) | 1980-10-06 | 1982-09-21 | Warner-Lambert Company | Substituted acyl derivatives of octahydro-1H-indole-2-carboxylic acids |
| US4350633A (en) | 1980-07-01 | 1982-09-21 | American Home Products Corporation | N-(2-Substituted-1-oxoalkyl)-2,3-dihydro-1H-indole-2-carboxylic acid derivatives |
| CN104470930B (en) | 2012-01-25 | 2017-02-22 | 诺华股份有限公司 | Heterocyclic compounds and methods for their use |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1524481A (en) * | 1976-01-07 | 1978-09-13 | Farmaceutici Italia | Tetrahydo - imidazopyridine derivatives |
-
1979
- 1979-12-07 FR FR7930046A patent/FR2470767A1/en active Granted
-
1980
- 1980-12-05 JP JP17195780A patent/JPS5692270A/en active Granted
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1524481A (en) * | 1976-01-07 | 1978-09-13 | Farmaceutici Italia | Tetrahydo - imidazopyridine derivatives |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2456733A1 (en) * | 1979-05-16 | 1980-12-12 | Morton Norwich Products Inc | NEW ACIDS 2- (MERCAPTO-1-OXO (LOWER ALKYL)) - 1,2,3,4-TETRAHYDROISOQUINOLEINE-3-CARBOXYLIQUES, ESPECIALLY USEFUL AS INHIBITORS OF THE ANGIOTENSIN I CONVERSION ENZYME |
| US4460775A (en) * | 1979-08-09 | 1984-07-17 | Norwich Eaton Pharmaceuticals, Inc. | 2-(3-Mercapto-1-oxopropyl)-1,2,3,4-tetrahydroisoquinoline-1-carboxylic acid derivatives |
| US5158959A (en) * | 1980-08-30 | 1992-10-27 | Hoechst Aktiengesellschaft | Decahydroisoquinoline carboxylic acids |
| US5401766A (en) * | 1980-08-30 | 1995-03-28 | Hoechst Aktiengesellschaft | Aminoacid derivatives, a process for their preparation, agents containing these compounds, and the use thereof |
| FR2503155A2 (en) * | 1980-10-02 | 1982-10-08 | Science Union & Cie | NOVEL SUBSTITUTED IMINO DIACIDES, PROCESSES FOR THEIR PREPARATION AND THEIR USE AS AN ENZYME INHIBITOR |
| US4479963A (en) * | 1981-02-17 | 1984-10-30 | Ciba-Geigy Corporation | 1-Carboxyalkanoylindoline-2-carboxylic acids |
| US4665087A (en) * | 1982-02-22 | 1987-05-12 | Ciba-Geigy Corporation | 1-(carbamyl, thiocarbamyl, and iminocarbamyl)-indoline derivatives |
Also Published As
| Publication number | Publication date |
|---|---|
| FR2470767B1 (en) | 1982-04-30 |
| JPH0127061B2 (en) | 1989-05-26 |
| JPS5692270A (en) | 1981-07-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0049658B1 (en) | Substituted imino diacids, their preparation and pharmaceutical preparations containing them | |
| FR2487829A2 (en) | NOVEL SUBSTITUTED IMINO ACIDS, PROCESSES FOR THEIR PREPARATION AND THEIR USE AS AN ENZYME INHIBITOR | |
| EP0051020B1 (en) | Substituted azobicyclooctanecarboxylic acids, process for their preparation und pharmaceutical compositions containing them | |
| FR2491469A1 (en) | 2-Carboxy-per:hydro-indole(s) and per or tetra:hydro-isoquinoline(s) - having a carboxy-substd. amino-acyl N-gp., inhibit carboxy:poly:peptidase(s), and kininase II and control hypertension | |
| EP0167459B1 (en) | 2-amino oxazolines and process for their preparation | |
| FR2470767A1 (en) | N-Acyl-2-carboxy fused ring heterocyclic cpds. - useful for inhibiting angiotensin converting enzyme and enkephalin(s) | |
| CA1097647A (en) | Preparation process of novel imidazoquinoleins and salts thereof | |
| JPH10316662A (en) | Substituted 6-and 7-aminotetrahydroisoquinolinecarboxylic acid | |
| FR2724654A1 (en) | NOVEL DERIVATIVES OF GALLIC ACID, PROCESS FOR THE PREPARATION THEREOF, THE NEW INTERMEDIATES OBTAINED, THEIR APPLICATION AS MEDICAMENTS AND THE PHARMACEUTICAL COMPOSITIONS COMPRISING THEM | |
| US4134991A (en) | Derivatives of 2-(3-phenyl-2-aminopropionyloxy)-acetic acid | |
| EP0366511A1 (en) | Benzoxazolinone derivatives, processes for their preparation and pharmaceutical compositions containing them | |
| EP0463944B1 (en) | Acylbenzoxazolinones, process for their preparation and pharmaceutical compositions containing them | |
| CA2089349C (en) | New pyrolidine derivatives, process for their preparation and pharmaceutical composition holding same | |
| US4285935A (en) | Dehydropeptide compounds, their production and their medical use | |
| IE44588B1 (en) | Trans-4-heterocyclyl-3,4-dihidro-2h-benzo(b)pyran-3-ol esters | |
| FR2460934A1 (en) | ISOQUINOLINE DERIVATIVES CONTAINING SULFUR, PROCESSES FOR THEIR PREPARATION AND PHARMACEUTICAL COMPOSITIONS CONTAINING SAME | |
| CH634311A5 (en) | BENZO (D) THIAZOLE DERIVATIVES, THEIR PREPARATION PROCESS AND THEIR THERAPEUTIC APPLICATIONS. | |
| CA1262903A (en) | Process for preparing 2-dihydroxy substituted 4-hydroxy-quinoline carboxylic acid derivatives, eventually etherified or esterified | |
| CA2166032A1 (en) | 1-oxo-2-(phenylsulfonylamino)pentylpiperidine; process for preparing them and their use as therapeutic agents | |
| JP4658512B2 (en) | Coumarin derivative and Maillard reaction inhibitor containing the derivative | |
| EP0395526B1 (en) | Benzothiazolinone derivatives, process for their preparation and pharmaceutical compositions containing them | |
| CN113620887A (en) | A class of SIRT5 protein inhibitors and their uses | |
| FR2756825A1 (en) | NEW DERIVATIVES [3H] -BENZOXAZOLE-2-THIONES AND [3H] - BENZOTHIAZOLE-2-THIONES SUBSTITUTES, THEIR PREPARATION PROCESS AND THE PHARMACEUTICAL COMPOSITIONS CONTAINING THEM | |
| US4051251A (en) | Novel anti-hypertensive compositions | |
| CH658786A5 (en) | DRUGS THAT CONTAIN ALPHA- (N-PYRROLYL) -ACIDS OR THEIR SALTS OR ESTERS. |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ST | Notification of lapse |