EP0737738A2 - Bleach tablets - Google Patents
Bleach tablets Download PDFInfo
- Publication number
- EP0737738A2 EP0737738A2 EP96610014A EP96610014A EP0737738A2 EP 0737738 A2 EP0737738 A2 EP 0737738A2 EP 96610014 A EP96610014 A EP 96610014A EP 96610014 A EP96610014 A EP 96610014A EP 0737738 A2 EP0737738 A2 EP 0737738A2
- Authority
- EP
- European Patent Office
- Prior art keywords
- weight
- bleach
- tablets
- bleach tablets
- sodium
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 239000007844 bleaching agent Substances 0.000 title claims abstract description 68
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 claims abstract description 27
- MWNQXXOSWHCCOZ-UHFFFAOYSA-L sodium;oxido carbonate Chemical compound [Na+].[O-]OC([O-])=O MWNQXXOSWHCCOZ-UHFFFAOYSA-L 0.000 claims abstract description 14
- 239000003795 chemical substances by application Substances 0.000 claims abstract description 11
- 239000012190 activator Substances 0.000 claims abstract description 10
- CDBYLPFSWZWCQE-UHFFFAOYSA-L sodium carbonate Substances [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 claims abstract description 10
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 claims abstract description 9
- 229920000642 polymer Polymers 0.000 claims abstract description 9
- 229910052910 alkali metal silicate Inorganic materials 0.000 claims abstract description 8
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 claims abstract description 8
- 102000004190 Enzymes Human genes 0.000 claims abstract description 7
- 108090000790 Enzymes Proteins 0.000 claims abstract description 7
- BPQQTUXANYXVAA-UHFFFAOYSA-N Orthosilicate Chemical compound [O-][Si]([O-])([O-])[O-] BPQQTUXANYXVAA-UHFFFAOYSA-N 0.000 claims abstract description 7
- 239000002202 Polyethylene glycol Substances 0.000 claims abstract description 7
- 229920001223 polyethylene glycol Polymers 0.000 claims abstract description 7
- 229910000029 sodium carbonate Inorganic materials 0.000 claims abstract description 7
- 239000005662 Paraffin oil Substances 0.000 claims abstract description 6
- 239000002671 adjuvant Substances 0.000 claims abstract description 6
- 239000011230 binding agent Substances 0.000 claims abstract description 6
- 239000000654 additive Substances 0.000 claims abstract description 5
- 229910000028 potassium bicarbonate Inorganic materials 0.000 claims abstract description 4
- 235000015497 potassium bicarbonate Nutrition 0.000 claims abstract description 4
- 239000011736 potassium bicarbonate Substances 0.000 claims abstract description 4
- 229910000027 potassium carbonate Inorganic materials 0.000 claims abstract description 4
- 235000011181 potassium carbonates Nutrition 0.000 claims abstract description 4
- TYJJADVDDVDEDZ-UHFFFAOYSA-M potassium hydrogencarbonate Chemical compound [K+].OC([O-])=O TYJJADVDDVDEDZ-UHFFFAOYSA-M 0.000 claims abstract description 4
- 229910000030 sodium bicarbonate Inorganic materials 0.000 claims abstract description 4
- UIIMBOGNXHQVGW-UHFFFAOYSA-M sodium bicarbonate Substances [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 claims abstract description 4
- 235000017557 sodium bicarbonate Nutrition 0.000 claims abstract description 4
- BGRWYDHXPHLNKA-UHFFFAOYSA-N Tetraacetylethylenediamine Chemical compound CC(=O)N(C(C)=O)CCN(C(C)=O)C(C)=O BGRWYDHXPHLNKA-UHFFFAOYSA-N 0.000 claims abstract 5
- VTIIJXUACCWYHX-UHFFFAOYSA-L disodium;carboxylatooxy carbonate Chemical compound [Na+].[Na+].[O-]C(=O)OOC([O-])=O VTIIJXUACCWYHX-UHFFFAOYSA-L 0.000 claims description 7
- 229940045872 sodium percarbonate Drugs 0.000 claims description 7
- 150000001875 compounds Chemical class 0.000 claims description 6
- 229910052783 alkali metal Inorganic materials 0.000 claims description 5
- 150000001732 carboxylic acid derivatives Chemical class 0.000 claims description 5
- -1 alkali metal percarbonate Chemical class 0.000 claims description 3
- 229940001593 sodium carbonate Drugs 0.000 claims description 3
- 239000001913 cellulose Substances 0.000 claims description 2
- 229920002678 cellulose Polymers 0.000 claims description 2
- PMYUVOOOQDGQNW-UHFFFAOYSA-N hexasodium;trioxido(trioxidosilyloxy)silane Chemical group [Na+].[Na+].[Na+].[Na+].[Na+].[Na+].[O-][Si]([O-])([O-])O[Si]([O-])([O-])[O-] PMYUVOOOQDGQNW-UHFFFAOYSA-N 0.000 claims description 2
- 239000000203 mixture Substances 0.000 abstract description 18
- 238000004090 dissolution Methods 0.000 abstract description 17
- 239000003599 detergent Substances 0.000 abstract description 10
- 238000003860 storage Methods 0.000 abstract description 9
- 150000001735 carboxylic acids Chemical class 0.000 abstract description 4
- 239000004744 fabric Substances 0.000 abstract description 3
- 230000000694 effects Effects 0.000 description 18
- 238000005406 washing Methods 0.000 description 11
- 238000004061 bleaching Methods 0.000 description 7
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 6
- 238000009472 formulation Methods 0.000 description 6
- 239000004094 surface-active agent Substances 0.000 description 6
- FRPJTGXMTIIFIT-UHFFFAOYSA-N tetraacetylethylenediamine Chemical compound CC(=O)C(N)(C(C)=O)C(N)(C(C)=O)C(C)=O FRPJTGXMTIIFIT-UHFFFAOYSA-N 0.000 description 6
- 238000004140 cleaning Methods 0.000 description 5
- 239000011248 coating agent Substances 0.000 description 5
- 238000000576 coating method Methods 0.000 description 5
- 229940088598 enzyme Drugs 0.000 description 5
- 238000002360 preparation method Methods 0.000 description 5
- DBVJJBKOTRCVKF-UHFFFAOYSA-N Etidronic acid Chemical compound OP(=O)(O)C(O)(C)P(O)(O)=O DBVJJBKOTRCVKF-UHFFFAOYSA-N 0.000 description 4
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 4
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 3
- ABLZXFCXXLZCGV-UHFFFAOYSA-N Phosphorous acid Chemical compound OP(O)=O ABLZXFCXXLZCGV-UHFFFAOYSA-N 0.000 description 3
- 239000002253 acid Substances 0.000 description 3
- 230000032683 aging Effects 0.000 description 3
- 150000001340 alkali metals Chemical class 0.000 description 3
- 238000007906 compression Methods 0.000 description 3
- 230000006835 compression Effects 0.000 description 3
- 238000000354 decomposition reaction Methods 0.000 description 3
- 230000007613 environmental effect Effects 0.000 description 3
- 239000004615 ingredient Substances 0.000 description 3
- 150000003839 salts Chemical class 0.000 description 3
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 3
- URDCARMUOSMFFI-UHFFFAOYSA-N 2-[2-[bis(carboxymethyl)amino]ethyl-(2-hydroxyethyl)amino]acetic acid Chemical compound OCCN(CC(O)=O)CCN(CC(O)=O)CC(O)=O URDCARMUOSMFFI-UHFFFAOYSA-N 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- ZOXJGFHDIHLPTG-UHFFFAOYSA-N Boron Chemical compound [B] ZOXJGFHDIHLPTG-UHFFFAOYSA-N 0.000 description 2
- QXNVGIXVLWOKEQ-UHFFFAOYSA-N Disodium Chemical compound [Na][Na] QXNVGIXVLWOKEQ-UHFFFAOYSA-N 0.000 description 2
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 2
- 102000004317 Lyases Human genes 0.000 description 2
- 108090000856 Lyases Proteins 0.000 description 2
- QPCDCPDFJACHGM-UHFFFAOYSA-N N,N-bis{2-[bis(carboxymethyl)amino]ethyl}glycine Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(=O)O)CCN(CC(O)=O)CC(O)=O QPCDCPDFJACHGM-UHFFFAOYSA-N 0.000 description 2
- KKCBUQHMOMHUOY-UHFFFAOYSA-N Na2O Inorganic materials [O-2].[Na+].[Na+] KKCBUQHMOMHUOY-UHFFFAOYSA-N 0.000 description 2
- 108091005804 Peptidases Proteins 0.000 description 2
- 102000035195 Peptidases Human genes 0.000 description 2
- 150000007513 acids Chemical class 0.000 description 2
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 2
- 229910052796 boron Inorganic materials 0.000 description 2
- 229910052681 coesite Inorganic materials 0.000 description 2
- 229910052906 cristobalite Inorganic materials 0.000 description 2
- 230000018109 developmental process Effects 0.000 description 2
- 235000014113 dietary fatty acids Nutrition 0.000 description 2
- JZBWUTVDIDNCMW-UHFFFAOYSA-L dipotassium;oxido sulfate Chemical class [K+].[K+].[O-]OS([O-])(=O)=O JZBWUTVDIDNCMW-UHFFFAOYSA-L 0.000 description 2
- DUYCTCQXNHFCSJ-UHFFFAOYSA-N dtpmp Chemical compound OP(=O)(O)CN(CP(O)(O)=O)CCN(CP(O)(=O)O)CCN(CP(O)(O)=O)CP(O)(O)=O DUYCTCQXNHFCSJ-UHFFFAOYSA-N 0.000 description 2
- NFDRPXJGHKJRLJ-UHFFFAOYSA-N edtmp Chemical compound OP(O)(=O)CN(CP(O)(O)=O)CCN(CP(O)(O)=O)CP(O)(O)=O NFDRPXJGHKJRLJ-UHFFFAOYSA-N 0.000 description 2
- 239000000194 fatty acid Substances 0.000 description 2
- 229930195729 fatty acid Natural products 0.000 description 2
- 150000004665 fatty acids Chemical class 0.000 description 2
- 150000002191 fatty alcohols Chemical group 0.000 description 2
- 230000002209 hydrophobic effect Effects 0.000 description 2
- 239000003112 inhibitor Substances 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 229910052760 oxygen Inorganic materials 0.000 description 2
- 239000001301 oxygen Substances 0.000 description 2
- 239000001253 polyvinylpolypyrrolidone Substances 0.000 description 2
- 229920000523 polyvinylpolypyrrolidone Polymers 0.000 description 2
- 235000013809 polyvinylpolypyrrolidone Nutrition 0.000 description 2
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 2
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 2
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 2
- 230000002633 protecting effect Effects 0.000 description 2
- 239000000377 silicon dioxide Substances 0.000 description 2
- 229910052682 stishovite Inorganic materials 0.000 description 2
- 229910052905 tridymite Inorganic materials 0.000 description 2
- ODAKQJVOEZMLOD-UHFFFAOYSA-N 3-[bis(carboxymethyl)amino]-2-hydroxypropanoic acid Chemical compound OC(=O)C(O)CN(CC(O)=O)CC(O)=O ODAKQJVOEZMLOD-UHFFFAOYSA-N 0.000 description 1
- MHKLKWCYGIBEQF-UHFFFAOYSA-N 4-(1,3-benzothiazol-2-ylsulfanyl)morpholine Chemical compound C1COCCN1SC1=NC2=CC=CC=C2S1 MHKLKWCYGIBEQF-UHFFFAOYSA-N 0.000 description 1
- FEPBITJSIHRMRT-UHFFFAOYSA-N 4-hydroxybenzenesulfonic acid Chemical class OC1=CC=C(S(O)(=O)=O)C=C1 FEPBITJSIHRMRT-UHFFFAOYSA-N 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 1
- 108010053835 Catalase Proteins 0.000 description 1
- 102000016938 Catalase Human genes 0.000 description 1
- 108090000371 Esterases Proteins 0.000 description 1
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- MHAJPDPJQMAIIY-UHFFFAOYSA-N Hydrogen peroxide Chemical compound OO MHAJPDPJQMAIIY-UHFFFAOYSA-N 0.000 description 1
- 102000004195 Isomerases Human genes 0.000 description 1
- 108090000769 Isomerases Proteins 0.000 description 1
- 108090001060 Lipase Proteins 0.000 description 1
- 102000004882 Lipase Human genes 0.000 description 1
- 239000004367 Lipase Substances 0.000 description 1
- PWHULOQIROXLJO-UHFFFAOYSA-N Manganese Chemical compound [Mn] PWHULOQIROXLJO-UHFFFAOYSA-N 0.000 description 1
- 101710163270 Nuclease Proteins 0.000 description 1
- 102000004316 Oxidoreductases Human genes 0.000 description 1
- 108090000854 Oxidoreductases Proteins 0.000 description 1
- 108700020962 Peroxidase Proteins 0.000 description 1
- 102000003992 Peroxidases Human genes 0.000 description 1
- 229920002257 Plurafac® Polymers 0.000 description 1
- GOOHAUXETOMSMM-UHFFFAOYSA-N Propylene oxide Chemical compound CC1CO1 GOOHAUXETOMSMM-UHFFFAOYSA-N 0.000 description 1
- 239000004365 Protease Substances 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- 108010056079 Subtilisins Proteins 0.000 description 1
- 102000005158 Subtilisins Human genes 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 1
- 241001122767 Theaceae Species 0.000 description 1
- 102000004357 Transferases Human genes 0.000 description 1
- 108090000992 Transferases Proteins 0.000 description 1
- 108010046334 Urease Proteins 0.000 description 1
- UAOKXEHOENRFMP-ZJIFWQFVSA-N [(2r,3r,4s,5r)-2,3,4,5-tetraacetyloxy-6-oxohexyl] acetate Chemical compound CC(=O)OC[C@@H](OC(C)=O)[C@@H](OC(C)=O)[C@H](OC(C)=O)[C@@H](OC(C)=O)C=O UAOKXEHOENRFMP-ZJIFWQFVSA-N 0.000 description 1
- 239000004480 active ingredient Substances 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 125000003545 alkoxy group Chemical group 0.000 description 1
- 150000003973 alkyl amines Chemical class 0.000 description 1
- 125000002947 alkylene group Chemical group 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 239000002280 amphoteric surfactant Substances 0.000 description 1
- 230000003625 amylolytic effect Effects 0.000 description 1
- 125000000129 anionic group Chemical group 0.000 description 1
- 230000000844 anti-bacterial effect Effects 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 239000003899 bactericide agent Substances 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 108010089934 carbohydrase Proteins 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 125000002843 carboxylic acid group Chemical group 0.000 description 1
- 238000009833 condensation Methods 0.000 description 1
- 230000005494 condensation Effects 0.000 description 1
- 229920001577 copolymer Polymers 0.000 description 1
- 230000001461 cytolytic effect Effects 0.000 description 1
- 238000004851 dishwashing Methods 0.000 description 1
- 239000000975 dye Substances 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 239000006260 foam Substances 0.000 description 1
- 235000011389 fruit/vegetable juice Nutrition 0.000 description 1
- 125000000524 functional group Chemical group 0.000 description 1
- 239000000417 fungicide Substances 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 235000001727 glucose Nutrition 0.000 description 1
- 235000011187 glycerol Nutrition 0.000 description 1
- VPVSTMAPERLKKM-UHFFFAOYSA-N glycoluril Chemical compound N1C(=O)NC2NC(=O)NC21 VPVSTMAPERLKKM-UHFFFAOYSA-N 0.000 description 1
- 229920001519 homopolymer Polymers 0.000 description 1
- 230000003165 hydrotropic effect Effects 0.000 description 1
- 125000004356 hydroxy functional group Chemical group O* 0.000 description 1
- 230000000977 initiatory effect Effects 0.000 description 1
- 229910017053 inorganic salt Inorganic materials 0.000 description 1
- 239000002563 ionic surfactant Substances 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- 235000019421 lipase Nutrition 0.000 description 1
- 230000002366 lipolytic effect Effects 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 239000000391 magnesium silicate Substances 0.000 description 1
- 235000012243 magnesium silicates Nutrition 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 229910052748 manganese Inorganic materials 0.000 description 1
- 239000011572 manganese Substances 0.000 description 1
- 108010003855 mesentericopeptidase Proteins 0.000 description 1
- 229910021645 metal ion Inorganic materials 0.000 description 1
- 238000000034 method Methods 0.000 description 1
- 108010020132 microbial serine proteinases Proteins 0.000 description 1
- MGFYIUFZLHCRTH-UHFFFAOYSA-N nitrilotriacetic acid Chemical compound OC(=O)CN(CC(O)=O)CC(O)=O MGFYIUFZLHCRTH-UHFFFAOYSA-N 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 239000002736 nonionic surfactant Substances 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 238000004806 packaging method and process Methods 0.000 description 1
- 229960003330 pentetic acid Drugs 0.000 description 1
- 239000002304 perfume Substances 0.000 description 1
- UEZVMMHDMIWARA-UHFFFAOYSA-M phosphonate Chemical compound [O-]P(=O)=O UEZVMMHDMIWARA-UHFFFAOYSA-M 0.000 description 1
- 150000003009 phosphonic acids Chemical class 0.000 description 1
- 229920000447 polyanionic polymer Polymers 0.000 description 1
- 159000000001 potassium salts Chemical class 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 229940024999 proteolytic enzymes for treatment of wounds and ulcers Drugs 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 238000007789 sealing Methods 0.000 description 1
- 150000004760 silicates Chemical class 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- QSKQNALVHFTOQX-UHFFFAOYSA-M sodium nonanoyloxybenzenesulfonate Chemical compound [Na+].CCCCCCCCC(=O)OC1=CC=CC=C1S([O-])(=O)=O QSKQNALVHFTOQX-UHFFFAOYSA-M 0.000 description 1
- 229960001922 sodium perborate Drugs 0.000 description 1
- PXDLHKPVKLUIJV-UHFFFAOYSA-M sodium;2-octanoyloxybenzenesulfonate Chemical compound [Na+].CCCCCCCC(=O)OC1=CC=CC=C1S([O-])(=O)=O PXDLHKPVKLUIJV-UHFFFAOYSA-M 0.000 description 1
- YKLJGMBLPUQQOI-UHFFFAOYSA-M sodium;oxidooxy(oxo)borane Chemical compound [Na+].[O-]OB=O YKLJGMBLPUQQOI-UHFFFAOYSA-M 0.000 description 1
- 239000002689 soil Substances 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 230000003019 stabilising effect Effects 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 229910021653 sulphate ion Inorganic materials 0.000 description 1
- 239000007916 tablet composition Substances 0.000 description 1
- 108010075550 termamyl Proteins 0.000 description 1
- MSLRPWGRFCKNIZ-UHFFFAOYSA-J tetrasodium;hydrogen peroxide;dicarbonate Chemical compound [Na+].[Na+].[Na+].[Na+].OO.OO.OO.[O-]C([O-])=O.[O-]C([O-])=O MSLRPWGRFCKNIZ-UHFFFAOYSA-J 0.000 description 1
- 239000004753 textile Substances 0.000 description 1
- 238000005303 weighing Methods 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
- 239000002888 zwitterionic surfactant Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D17/00—Detergent materials or soaps characterised by their shape or physical properties
- C11D17/0047—Detergents in the form of bars or tablets
- C11D17/0065—Solid detergents containing builders
- C11D17/0073—Tablets
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D17/00—Detergent materials or soaps characterised by their shape or physical properties
- C11D17/0039—Coated compositions or coated components in the compositions, (micro)capsules
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/02—Inorganic compounds ; Elemental compounds
- C11D3/04—Water-soluble compounds
- C11D3/08—Silicates
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/02—Inorganic compounds ; Elemental compounds
- C11D3/12—Water-insoluble compounds
- C11D3/124—Silicon containing, e.g. silica, silex, quartz or glass beads
- C11D3/1246—Silicates, e.g. diatomaceous earth
- C11D3/1253—Layer silicates, e.g. talcum, kaolin, clay, bentonite, smectite, montmorillonite, hectorite or attapulgite
- C11D3/1273—Crystalline layered silicates of type NaMeSixO2x+1YH2O
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/16—Organic compounds
- C11D3/20—Organic compounds containing oxygen
- C11D3/2075—Carboxylic acids-salts thereof
- C11D3/2086—Hydroxy carboxylic acids-salts thereof
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/39—Organic or inorganic per-compounds
- C11D3/3902—Organic or inorganic per-compounds combined with specific additives
- C11D3/3905—Bleach activators or bleach catalysts
- C11D3/3907—Organic compounds
- C11D3/3917—Nitrogen-containing compounds
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/39—Organic or inorganic per-compounds
- C11D3/3942—Inorganic per-compounds
Definitions
- the present invention relates to bleach tablets intended to be used together with usual textile detergent compositions so as to obtain an improved washing effect and stain removal of bleach-sensitive stains.
- oxygen-based bleaches such as sodium perborate
- An increasing awareness of the potential environmental hazard of these boron-containing bleaches has, however, intensified the interest in other oxygen-based bleaches.
- potassium peroxomonosulphates and alkali metal perhydrates such as sodium percarbonate which are dissolved quickly in water and which in addition to their bleaching effect also have an alkaline effect supporting and intensifying the washing effect.
- a drawback of sodium percarbonate is, however, its poor stability which presents a limiting factor for its use in detergent compositions.
- Factors adversely affecting the stability are especially the presence of moisture, temperatures exceeding 28°C to 30°C, as well as the presence of metal ions, such as iron, manganese etc.
- sodium percarbonate is particularly difficult with respect to the formulation of the tablets because the compression pressure necessary for the tabletting results in heat development which in turn implies that the tablets reach critical temperatures initiating the decomposition of sodium percarbonate.
- EP publication No. 0 481 792 discloses bleach tablets comprising a bleach activator having a pseudo - first order perhydrolysis rate constant (K obs ) of from 1.5 x 10 -4 to 350 x 10 -4 sec -1 as well as a percarbonate being protected against decomposition by means of an inorganic salt, especially sodium carbonate.
- K obs pseudo - first order perhydrolysis rate constant
- Example 4 of the publication describes a bleach tablet composition containing 12.5 parts by weight of sodium percarbonate, 10.25 parts by weight of spray-dried sodium carbonate containing a small amount of polymer, and 2.25 parts by weight of TAED.
- the very high amount of carbonate in these bleach tablets may, however, cause problems of formation of incrustations in the laundry.
- the bleach tablets known from EP publication No. 0 481 792 have a very long rate of dissolution, and when used in the short washing programmes increasingly for environmental reason, this very long rate of dissolution may have the result that the tablets are not dissolved before the main washing is terminated. As a result, the cleaning and bleaching effect is considerably reduced. In addition, the tablets may be caught in a pocket, thereby causing a miscolouring due to the long contact period.
- bleach tablets according to the invention which are characterised in that in addition to optional, usual adjuvants and additives they comprise coated percarbonate 45 to 85 % by weight bleach activator, calculated as TAED 0 to 20 % by weight layered silicate and/or alkali metal silicate 1 to 50 % by weight sodium/potassium carbonate 0 to 25 % by weight sodium/potassium bicarbonate 0 to 25 % by weight polyfunctional carboxylic acid, calculated as citric acid 5 to 35 % by weight complex-binder 0 to 5 % by weight paraffin oil 0 to 2 % by weight disintegrating agent 0 to 5 % by weight enzymes 0 to 10 % by weight polymer 0 to 10 % by weight
- the bleach tablets are intended for use in ordinary household washing machines together with conventional detergent compositions so as to obtain an improved washing effect and stain removal of bleach-sensitive stains, such as juice, wine, tea as well as some protein-based stains.
- bleach-sensitive stains such as juice, wine, tea
- some protein-based stains such as juice, wine, tea
- an improved washing effect is also obtained at low temperatures, such as at 40°C and therebelow.
- the bleach tablets are highly practical, as the use thereof can be adapted to the washing habits prevailing in both Europe and in Asia without any problems.
- the bleach tablets according to the invention are advantageous in possessing a surprisingly good storage stability.
- the particular composition of the tablets has the effect that the coated percarbonate is not seriously deteriorated during the tabletting, and that the heat generated during said tabletting does not initiate the decomposition of the percarbonate to a harmful degree as is the case in conventional bleach-containing tablets.
- the amount of layered silicate and/or alkali metal silicate is in the range of from 1 to 50 % by weight, preferably from 1 to 25 % by weight, more preferred from 2 to 10 % by weight, and most preferred from 4 to 8 % by weight.
- the layered silicate is commercially available under the trade name SKS-6 supplied by Hoechst AG, Frankfurt, Germany.
- coated percarbonate being an ingredient of the bleach tablets according to the invention is of a completely conventional nature.
- An alkali metal percarbonate is advantageously used as it is easily soluble in water and in addition to the bleaching effect has an alkaline effect supporting and intensifying the washing effect.
- suitable coated percarbonates are two commercially available products, viz. sodium percarbonate coated-Q10 (which is coated with a boron containing coating) supplied by DEGUSSA AG, Frankfurt, Germany, and PCS-C Sodium Carbonate Peroxyhydrate (coating based on a sulphate and sodium carbonate supplied by Solway Interox GmbH, Pullach, Germany.
- the coated percarbonate is used in an amount of from 45 to 85 % by weight, such as from 50 to 65 % by weight.
- a small amount of a further bleach such as potassium peroxomonosulphate, may optionally be incorporated.
- the bleach tablets may furthermore, if desired, comprise a bleach activator.
- bleach activators are tetraacetylmethylenediamine, TAED (tetraacetylethylenediamine), TAGU (tetraacetyl glycoluril), PAGE (pentaacetyl glucose), p-hydroxybenzene sulphonates, NOBS (sodium nonanoyloxybenzene sulphonate) and sodium octanoyloxybenzene sulphonate.
- TAED tetraacetylmethylenediamine
- TAED tetraacetylethylenediamine
- TAGU tetraacetyl glycoluril
- PAGE pentaacetyl glucose
- NOBS sodium nonanoyloxybenzene sulphonate
- sodium octanoyloxybenzene sulphonate Most preferred is TAED, which may optionally be present in coated form
- the bleach tablets contain a coated percarbonate and a bleach activator, calculated as TAED, in a weight ratio ranging from 2: 1 to 20:1, such as from 3:1 1 to 15:1, e.g. from 3.5:1 1 to 12:1.
- the bleach tablets contain, furthermore, a polyfunctional carboxylic acid.
- polyfunctional carboxylic acids can be defined as such acids which in addition to the one obligatory carboxylic acid group also contain at least one further functional group selected from carboxyl and hydroxy. Furthermore, the polyfunctional acids can also contain nitrogen.
- Non-limiting examples of such polyfunctional carboxylic acids are citric acid, nitrilotriacetic acid (NTA), EDTA and isoserine diacetic acid, of which preference is given to citric acid.
- the polyfunctional carboxylic acid forms part of the bleach tablets in an amount of from 5 to 35 % by weight, preferably from 14 to 22 % by weight, such as from 19 to 21 % by weight, calculated as citric acid.
- ingredients advantageously forming part of the bleach tablets according to the invention are for instance sodium/potassium carbonate and sodium/potassium bicarbonate which in combination with citric acid improve the solubility.
- a binder may advantageously be incorporated, such as polyethylene glycol, e.g. polyethylene glycol of a molecular weight of from 200 to 8,000.
- a complex-binder and a precipitation inhibitor may advantageously be incorporated, such as a phosphonate, i.e. a phosphonic acid or a salt thereof.
- a phosphonate i.e. a phosphonic acid or a salt thereof.
- suitable phosphonates and phosphonic acids are for instance ethane-1-hydroxy-1,1-diphosphonic acid (HEDP), ethylene diaminetetra(methylene phosphonic acid) (EDTMP), diethylene triaminepenta(methylene phosphonic acid) (DETPMP), aminotrismethylene phosphonic acid (ATMP) as well as salts thereof.
- HEDP ethane-1-hydroxy-1,1-diphosphonic acid
- ETMP ethylene diaminetetra(methylene phosphonic acid)
- DETPMP diethylene triaminepenta(methylene phosphonic acid)
- ATMP aminotrismethylene phosphonic acid
- disodium and the tetrasodium salt respectively,
- Sequion 10 Na PDR supplied by G. Bozzetto S.p.A., Bergamo, Italy, and which is the disodium salt of ethane-1-hydroxy-1,1-diphosphonic acid.
- EDTA ethylenediaminetetraacetic acid
- DETPA diethylenetriaminepentaacetic acid
- HEEDTA hydroxyethylethylenediaminetriacetic acid
- ISDA isoserinediacetic acid
- enzymes may advantageously form part of the bleach tablets according to the invention.
- examples thereof are for instance mixtures of specifically acting types, such as proteases, carbohydrases, esterases, lipases, oxidoreductases, catalases, peroxidases, ureases, isomerases, lyases, transferases, desmolases or nucleases.
- Esperase®, Savinase® and Alcalase® proteolytic enzymes
- Termamyl® asmylolytic enzymes
- Lipolase lipolytic enzymes
- Celluzyme celluzyme
- polymers may form part of the bleach tablets.
- such polymers may be selected among all conventional polymers utilized within the detergent industry, such as the polyanionic polymers as described in US patent Nos. 3,308,067, 3,723,322, 4,144,226 and 4,146,495 and in GB patent No. 1.596.756. Among these homo- or copolymeric carboxylic acids and sodium and potassium salts thereof are preferred. If polymers are incorporated, the amount hereof typically ranges from 1 to 10 % by weight.
- Examples of further conventional laundry detergent ingredients are agents preventing the running of colour, antiagglutinants, dyes, deodorising agents, antioxidants, solvents, foam inhibitors, perfume, optical brighteners, antistatics, nonionic tensides, bactericides, fungicides, hydrotropic agents, wetting agents and fillers.
- the nonionic tensides used can in principle be selected from all conventional nonionic tensides usually prepared by condensation of a hydrophilic alkylene oxide with a hydrophobic compound.
- suitable nonionic tensides are alkoxylated nonionic surfactants, where the alkoxy moiety contains an ethylene oxide, propylene oxide and/or butylene oxide as well as mixtures thereof.
- Suitable hydrophobic compounds are alcohols, thioalcohols, dioles, fatty acids, fatty acid amides, alkanesulphone amides, alkyl amines as well as alkyl phenols.
- nonionic tensides especially ethoxylated compounds of primary and secondary fatty alcohols are preferred.
- Plurafac LF 403 is a fatty alcohol alkoxylate supplied by BASF, Ludwigshafen, Germany.
- the tenside component may also include other tensides of a non-anionic nature, such as all known types of amphoteric, ampholytic and zwitterionic surfactants.
- conventional tabletting adjuvants are usually incorporated, such as starch, magnesium stearate, magnesium silicates, glycerol, glucose, paraffin oil, polyethylene glycol and disintegrating agents, such as polyvinyl pyrrolidone (PVP), polyvinyl polypyrrolidone (PVPP) and cellulose-based or cellulose-modified compounds.
- PVP polyvinyl pyrrolidone
- PVPP polyvinyl polypyrrolidone
- cellulose-based or cellulose-modified compounds such as starch, magnesium stearate, magnesium silicates, glycerol, glucose, paraffin oil, polyethylene glycol and disintegrating agents, such as polyvinyl pyrrolidone (PVP), polyvinyl polypyrrolidone (PVPP) and cellulose-based or cellulose-modified compounds.
- Paraffin oil functions as a lubricant and facilitates the tabletting, and it is advantageously included in the bleach tablets according to the invention.
- corresponding compounds having the same or a similar function such as for instance glycerol.
- the formulation of a washing-intensifying bleach tablet requires fulfilment of a wide range of parameters in order to obtain the desired functional and performance characteristics.
- the tablet is to be placed directly in the drum of the washing machine and may not be resident in fabric creases, pockets etc. or in the washing machine window/sealing ring for a long period of time. Consequently, the rate of dissolution should be as short as possible, preferably of no more than 30 to 180 sec.
- rate of dissolution Apart from a minor acceptable increase of the rate of dissolution compared to the initial rate of dissolution measured within 5 to 24 hours after the preparation, said rate of dissolution must not, of course, be increased appreciably when stored for a long period of time.
- the tablet strength presents a vital parameter for the processes following the tabletting, i.e. the packaging and transport, and in order to obtain a good durability this parameter must not be noticeably reduced over time.
- the bleach tablets are also very advantageous in being environmentally desirable and by displaying good tablet-technical properties, such as a fast rate of dissolution and a high tablet strength, and these properties do not change to a noticeable extent during storage. In addition to ensuring the correct function and performance of the tablets, such properties are also important for the durability of the tablets.
- bleach tablets were prepared having a weight of 30 g, a diameter of 40 mm and a height of 17 mm.
- the present accelerated aging tests were performed in order to illustrate the good stability and tablet-technical properties of the tablets.
- the tablets were stored at 40°C and a relative humidity of 75%.
- the bleach tablets according to the invention show a good stability.
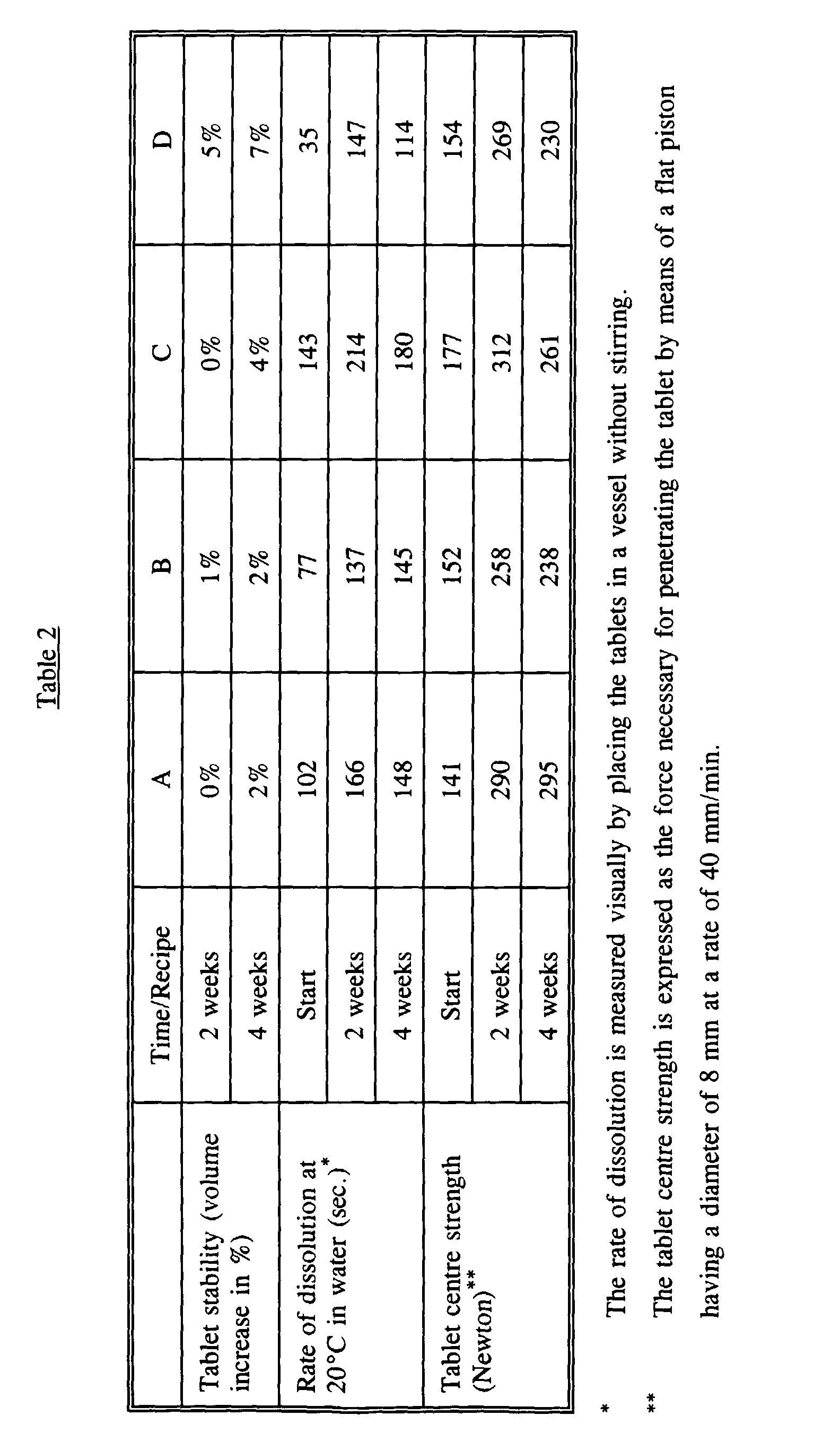
- the volume is not noticeably increased after storage for 4 weeks under accelerated aging conditions. It should be noted that for tablets of such a size, a volume increase of up to 15% is acceptable under the stated conditions.
- the bleach tablets have a satisfactory rate of dissolution which is not increased considerably during storage.
- the bleach tablets display a satisfactory tablet strength which is not reduced considerably during storage. It should be noted that compared to the initially values measured within 5 to 24 hours after the preparation, the said storage under extreme conditions causes the rate of dissolution as well as the tablet strength to increase due to the curing of the tablets.
- Example 2 As described above in Example 2, accelerated aging test were performed to illustrate the good stability and tablet-technical properties of the tablets stated in Example 3.
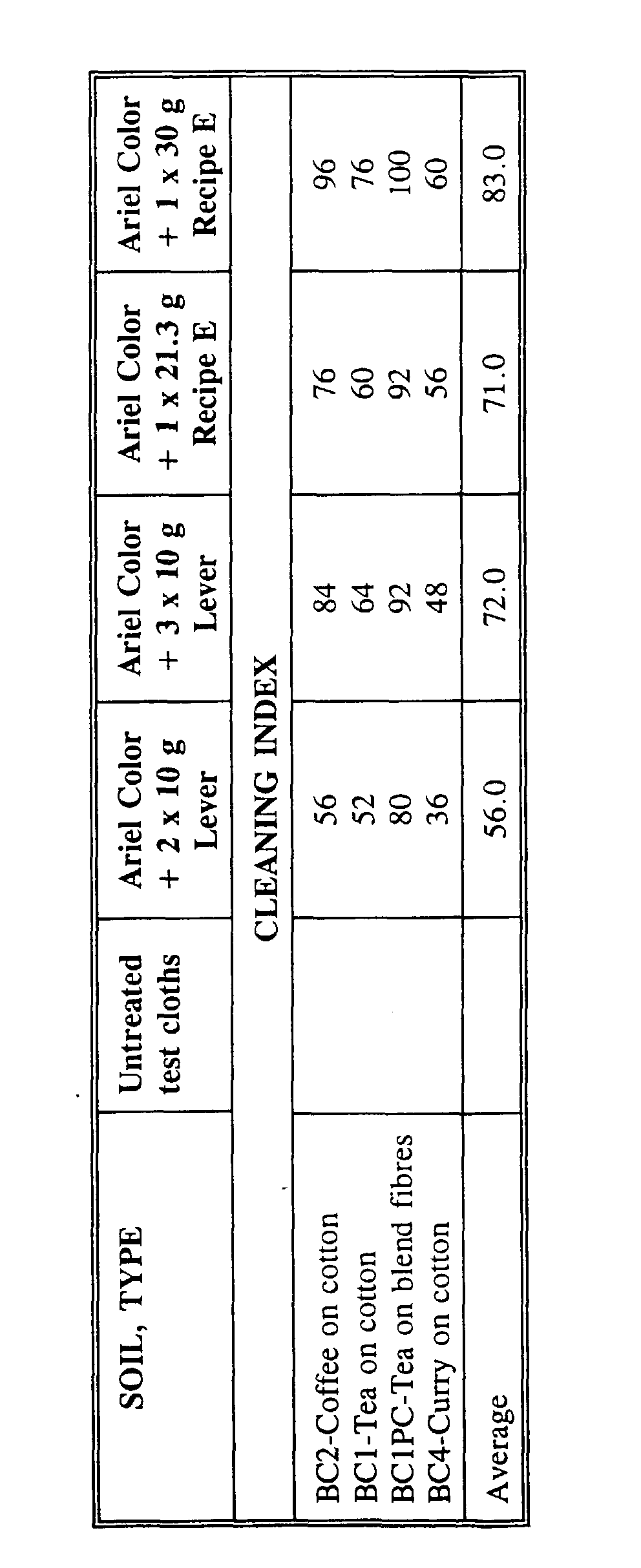
- the cleaning effect is calculated as the percentage increase of the reflectance value compared to an untreated test cloth.
- the cleaning index is calculated on the basis of the cleaning effect, the highest value of the cleaning effect (Ariel Color + 1 x 30 g recipe E on soil of the type BC1PC-Tea on blend fibres, 25) being set to 100.
- the bleach tablets according to the invention show an improved bleaching effect over the bleach tablets known from EP publication No. 0 481 792.
- the bleach tablets according to the invention clearly display a superior rate of dissolution in relation to the bleach tablets known from EP publication No. 0 481 792.
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Wood Science & Technology (AREA)
- Organic Chemistry (AREA)
- Inorganic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Emergency Medicine (AREA)
- Crystallography & Structural Chemistry (AREA)
- Detergent Compositions (AREA)
Abstract
Description
- The present invention relates to bleach tablets intended to be used together with usual textile detergent compositions so as to obtain an improved washing effect and stain removal of bleach-sensitive stains.
- During recent years, the detergent composition market has focused on developing more and more concentrated products which also take into account the environmental aspects. Accordingly, the tendency has been to develop the so-called compact formulations based on environmentally desirable and easily biodegradable raw materials, said compact formulations having bulk densities in the range of from approximately 0.6 to 1.0 kg/litre. A natural continuation of this development has been the introduction of detergent tablets, especially for use in dish washing machines, and having bulk densities of from approximately 1.4 to 1.7 kg/litre.
- However, the preparation of such highly concentrated detergent tablets involves formulation problems, because the active ingredients of the tablets are very tightly compressed, often at compression pressures of from 20 to 180 KN.
- For a number of years, oxygen-based bleaches, such as sodium perborate, have mainly been used. An increasing awareness of the potential environmental hazard of these boron-containing bleaches has, however, intensified the interest in other oxygen-based bleaches.
- Particular attention is here attached to potassium peroxomonosulphates and alkali metal perhydrates, such as sodium percarbonate which are dissolved quickly in water and which in addition to their bleaching effect also have an alkaline effect supporting and intensifying the washing effect. A drawback of sodium percarbonate is, however, its poor stability which presents a limiting factor for its use in detergent compositions.
- Factors adversely affecting the stability are especially the presence of moisture, temperatures exceeding 28°C to 30°C, as well as the presence of metal ions, such as iron, manganese etc.
- The use of sodium percarbonate is particularly difficult with respect to the formulation of the tablets because the compression pressure necessary for the tabletting results in heat development which in turn implies that the tablets reach critical temperatures initiating the decomposition of sodium percarbonate.
- Attempts have therefore been made to stabilise alkali metal percarbonates by incorporating stabilising components and/or by protecting the alkali metal percarbonates by means of a coating. Such a coating is, however, completely or partially destroyed during the high compression pressures used for the tabletting with the result that the protecting effect of the coating is reduced heavily.
- EP publication No. 0 481 792 discloses bleach tablets comprising a bleach activator having a pseudo - first order perhydrolysis rate constant (Kobs) of from 1.5 x 10-4 to 350 x 10-4 sec-1 as well as a percarbonate being protected against decomposition by means of an inorganic salt, especially sodium carbonate.
- Example 4 of the publication describes a bleach tablet composition containing 12.5 parts by weight of sodium percarbonate, 10.25 parts by weight of spray-dried sodium carbonate containing a small amount of polymer, and 2.25 parts by weight of TAED.
- The very high amount of carbonate in these bleach tablets may, however, cause problems of formation of incrustations in the laundry.
- Furthermore, the bleach tablets known from EP publication No. 0 481 792 have a very long rate of dissolution, and when used in the short washing programmes increasingly for environmental reason, this very long rate of dissolution may have the result that the tablets are not dissolved before the main washing is terminated. As a result, the cleaning and bleaching effect is considerably reduced. In addition, the tablets may be caught in a pocket, thereby causing a miscolouring due to the long contact period.
- Thus, it is the object of the present invention to provide storage stable bleach tablets based on a percarbonate with a good bleaching effect, a satisfactory rate of dissolution, as well as a low tendency to form incrustations in the laundry.
- Such a storage stability is surprisingly obtained by the bleach tablets according to the invention, which are characterised in that in addition to optional, usual adjuvants and additives they comprise
coated percarbonate 45 to 85 % by weight bleach activator, calculated as TAED 0 to 20 % by weight layered silicate and/or alkali metal silicate 1 to 50 % by weight sodium/potassium carbonate 0 to 25 % by weight sodium/potassium bicarbonate 0 to 25 % by weight polyfunctional carboxylic acid, calculated as citric acid 5 to 35 % by weight complex-binder 0 to 5 % by weight paraffin oil 0 to 2 % by weight disintegrating agent 0 to 5 % by weight enzymes 0 to 10 % by weight polymer 0 to 10 % by weight - Further scope of applicability of the present invention will become apparent from the detailed description given hereinafter. However, it should be understood that the detailed description and specific examples, while indicating preferred embodiments of the invention, are given by way of illustration only, since various changes and modifications within the spirit and scope of the invention will become apparent to those skilled in the art from this detailed description.
- The bleach tablets are intended for use in ordinary household washing machines together with conventional detergent compositions so as to obtain an improved washing effect and stain removal of bleach-sensitive stains, such as juice, wine, tea as well as some protein-based stains. Thus, an improved washing effect is also obtained at low temperatures, such as at 40°C and therebelow. The bleach tablets are highly practical, as the use thereof can be adapted to the washing habits prevailing in both Europe and in Asia without any problems.
- The bleach tablets according to the invention are advantageous in possessing a surprisingly good storage stability. Thus, the particular composition of the tablets has the effect that the coated percarbonate is not seriously deteriorated during the tabletting, and that the heat generated during said tabletting does not initiate the decomposition of the percarbonate to a harmful degree as is the case in conventional bleach-containing tablets.
- The content of layered silicate (in Germany known as "Schichtsilikat") and/or alkali metal silicate appears to be of great importance to obtain the good stability of the bleach tablets, but it is not known exactly how these silicates may protect the percarbonate bleach.
- The amount of layered silicate and/or alkali metal silicate is in the range of from 1 to 50 % by weight, preferably from 1 to 25 % by weight, more preferred from 2 to 10 % by weight, and most preferred from 4 to 8 % by weight.
- The layered silicate is commercially available under the trade name SKS-6 supplied by Hoechst AG, Frankfurt, Germany.
- The alkali metal silicate used has the composition of Na2O : SiO2 = 1: 1 to 1 : 3.5, preferably 1 : 1.6 to 1 : 2.6. A preferred alkali metal silicate is sodium disilicate with the composition of Na2O : SiO2 = 1 : 2 e.g. supplied by Crossfield Chemie, Eijsden, The Netherlands under the trade name Pyramid P50.
- The coated percarbonate being an ingredient of the bleach tablets according to the invention is of a completely conventional nature.
- An alkali metal percarbonate is advantageously used as it is easily soluble in water and in addition to the bleaching effect has an alkaline effect supporting and intensifying the washing effect. Examples of suitable coated percarbonates are two commercially available products, viz. sodium percarbonate coated-Q10 (which is coated with a boron containing coating) supplied by DEGUSSA AG, Frankfurt, Germany, and PCS-C Sodium Carbonate Peroxyhydrate (coating based on a sulphate and sodium carbonate supplied by Solway Interox GmbH, Pullach, Germany.
- The coated percarbonate is used in an amount of from 45 to 85 % by weight, such as from 50 to 65 % by weight.
- Furthermore, in addition to the coated percarbonate, a small amount of a further bleach, such as potassium peroxomonosulphate, may optionally be incorporated.
- The bleach tablets may furthermore, if desired, comprise a bleach activator. Non-limiting examples of bleach activators are tetraacetylmethylenediamine, TAED (tetraacetylethylenediamine), TAGU (tetraacetyl glycoluril), PAGE (pentaacetyl glucose), p-hydroxybenzene sulphonates, NOBS (sodium nonanoyloxybenzene sulphonate) and sodium octanoyloxybenzene sulphonate. Most preferred is TAED, which may optionally be present in coated form. The bleach activator is used in an amount ranging from 0 to 20 % by weight, preferably from 2 to 15 % by weight, and more preferred from 4 to 10 % by weight, such as from 5 to 6 % by weight.
- In a preferred embodiment of the invention the bleach tablets contain a coated percarbonate and a bleach activator, calculated as TAED, in a weight ratio ranging from 2: 1 to 20:1, such as from 3:1 1 to 15:1, e.g. from 3.5:1 1 to 12:1.
- The bleach tablets contain, furthermore, a polyfunctional carboxylic acid. As a general rule, polyfunctional carboxylic acids can be defined as such acids which in addition to the one obligatory carboxylic acid group also contain at least one further functional group selected from carboxyl and hydroxy. Furthermore, the polyfunctional acids can also contain nitrogen. Non-limiting examples of such polyfunctional carboxylic acids are citric acid, nitrilotriacetic acid (NTA), EDTA and isoserine diacetic acid, of which preference is given to citric acid.
- The polyfunctional carboxylic acid forms part of the bleach tablets in an amount of from 5 to 35 % by weight, preferably from 14 to 22 % by weight, such as from 19 to 21 % by weight, calculated as citric acid.
- Other ingredients advantageously forming part of the bleach tablets according to the invention are for instance sodium/potassium carbonate and sodium/potassium bicarbonate which in combination with citric acid improve the solubility.
- Furthermore, a binder may advantageously be incorporated, such as polyethylene glycol, e.g. polyethylene glycol of a molecular weight of from 200 to 8,000.
- Moreover, a complex-binder and a precipitation inhibitor may advantageously be incorporated, such as a phosphonate, i.e. a phosphonic acid or a salt thereof. Examples of suitable phosphonates and phosphonic acids are for instance ethane-1-hydroxy-1,1-diphosphonic acid (HEDP), ethylene diaminetetra(methylene phosphonic acid) (EDTMP), diethylene triaminepenta(methylene phosphonic acid) (DETPMP), aminotrismethylene phosphonic acid (ATMP) as well as salts thereof. Among these the disodium and the tetrasodium salt, respectively, of ethane-1-hydroxy-1,1-diphosphonic acid are particularly preferred. An example of a commercially available product is Sequion 10 Na PDR, supplied by G. Bozzetto S.p.A., Bergamo, Italy, and which is the disodium salt of ethane-1-hydroxy-1,1-diphosphonic acid.
- Among other complex-binders EDTA (ethylenediaminetetraacetic acid), DETPA (diethylenetriaminepentaacetic acid), HEEDTA (hydroxyethylethylenediaminetriacetic acid) and ISDA (isoserinediacetic acid).
- Moreover, enzymes may advantageously form part of the bleach tablets according to the invention. Examples thereof are for instance mixtures of specifically acting types, such as proteases, carbohydrases, esterases, lipases, oxidoreductases, catalases, peroxidases, ureases, isomerases, lyases, transferases, desmolases or nucleases. Among commercially available products Esperase®, Savinase® and Alcalase® (proteolytic enzymes), Termamyl® (amylolytic enzymes), Lipolase (lipolytic enzymes) and Celluzyme (cellulytic enzymes) can be mentioned all supplied by Novo Nordisk A/S, Bagsværd, Denmark.
- Moreover, if desired, polymers may form part of the bleach tablets. In principle such polymers may be selected among all conventional polymers utilized within the detergent industry, such as the polyanionic polymers as described in US patent Nos. 3,308,067, 3,723,322, 4,144,226 and 4,146,495 and in GB patent No. 1.596.756. Among these homo- or copolymeric carboxylic acids and sodium and potassium salts thereof are preferred. If polymers are incorporated, the amount hereof typically ranges from 1 to 10 % by weight.
- Examples of further conventional laundry detergent ingredients are agents preventing the running of colour, antiagglutinants, dyes, deodorising agents, antioxidants, solvents, foam inhibitors, perfume, optical brighteners, antistatics, nonionic tensides, bactericides, fungicides, hydrotropic agents, wetting agents and fillers.
- The nonionic tensides used can in principle be selected from all conventional nonionic tensides usually prepared by condensation of a hydrophilic alkylene oxide with a hydrophobic compound. Examples of suitable nonionic tensides are alkoxylated nonionic surfactants, where the alkoxy moiety contains an ethylene oxide, propylene oxide and/or butylene oxide as well as mixtures thereof. Suitable hydrophobic compounds are alcohols, thioalcohols, dioles, fatty acids, fatty acid amides, alkanesulphone amides, alkyl amines as well as alkyl phenols.
- Among the nonionic tensides, especially ethoxylated compounds of primary and secondary fatty alcohols are preferred.
- Among commercially available products, Plurafac LF 403 can be mentioned which is a fatty alcohol alkoxylate supplied by BASF, Ludwigshafen, Germany.
- In addition to the nonionic tenside, the tenside component may also include other tensides of a non-anionic nature, such as all known types of amphoteric, ampholytic and zwitterionic surfactants.
- By the preparation of the bleach tablets according to the invention conventional tabletting adjuvants are usually incorporated, such as starch, magnesium stearate, magnesium silicates, glycerol, glucose, paraffin oil, polyethylene glycol and disintegrating agents, such as polyvinyl pyrrolidone (PVP), polyvinyl polypyrrolidone (PVPP) and cellulose-based or cellulose-modified compounds.
- Paraffin oil functions as a lubricant and facilitates the tabletting, and it is advantageously included in the bleach tablets according to the invention. As an alternative, it is possible to use corresponding compounds having the same or a similar function, such as for instance glycerol.
- The preparation and the tabletting of the bleach tablets are carried out in a manner known per se, and a more detailed description can for instance be found in the US patent Nos. 4,099,912, 4,828,749 and 4,913,832 as well as the DE patent Nos. 355,626 and 3,827,895.
- The formulation of a washing-intensifying bleach tablet requires fulfilment of a wide range of parameters in order to obtain the desired functional and performance characteristics.
- The tablet is to be placed directly in the drum of the washing machine and may not be resident in fabric creases, pockets etc. or in the washing machine window/sealing ring for a long period of time. Consequently, the rate of dissolution should be as short as possible, preferably of no more than 30 to 180 sec.
- Apart from a minor acceptable increase of the rate of dissolution compared to the initial rate of dissolution measured within 5 to 24 hours after the preparation, said rate of dissolution must not, of course, be increased appreciably when stored for a long period of time.
- The tablet strength presents a vital parameter for the processes following the tabletting, i.e. the packaging and transport, and in order to obtain a good durability this parameter must not be noticeably reduced over time.
- In addition to being very stable as regards the percarbonate, the bleach tablets are also very advantageous in being environmentally desirable and by displaying good tablet-technical properties, such as a fast rate of dissolution and a high tablet strength, and these properties do not change to a noticeable extent during storage. In addition to ensuring the correct function and performance of the tablets, such properties are also important for the durability of the tablets.
- The following examples illustrate the bleach tablets according to the invention in greater details.
-
- The present accelerated aging tests were performed in order to illustrate the good stability and tablet-technical properties of the tablets.
- By the tests performed, the tablets were stored at 40°C and a relative humidity of 75%.
-
- As it appears from Table 2, the bleach tablets according to the invention show a good stability. Thus, the volume is not noticeably increased after storage for 4 weeks under accelerated aging conditions. It should be noted that for tablets of such a size, a volume increase of up to 15% is acceptable under the stated conditions.
- Moreover, it appears from Table 2 that the bleach tablets have a satisfactory rate of dissolution which is not increased considerably during storage. In addition, the bleach tablets display a satisfactory tablet strength which is not reduced considerably during storage. It should be noted that compared to the initially values measured within 5 to 24 hours after the preparation, the said storage under extreme conditions causes the rate of dissolution as well as the tablet strength to increase due to the curing of the tablets.
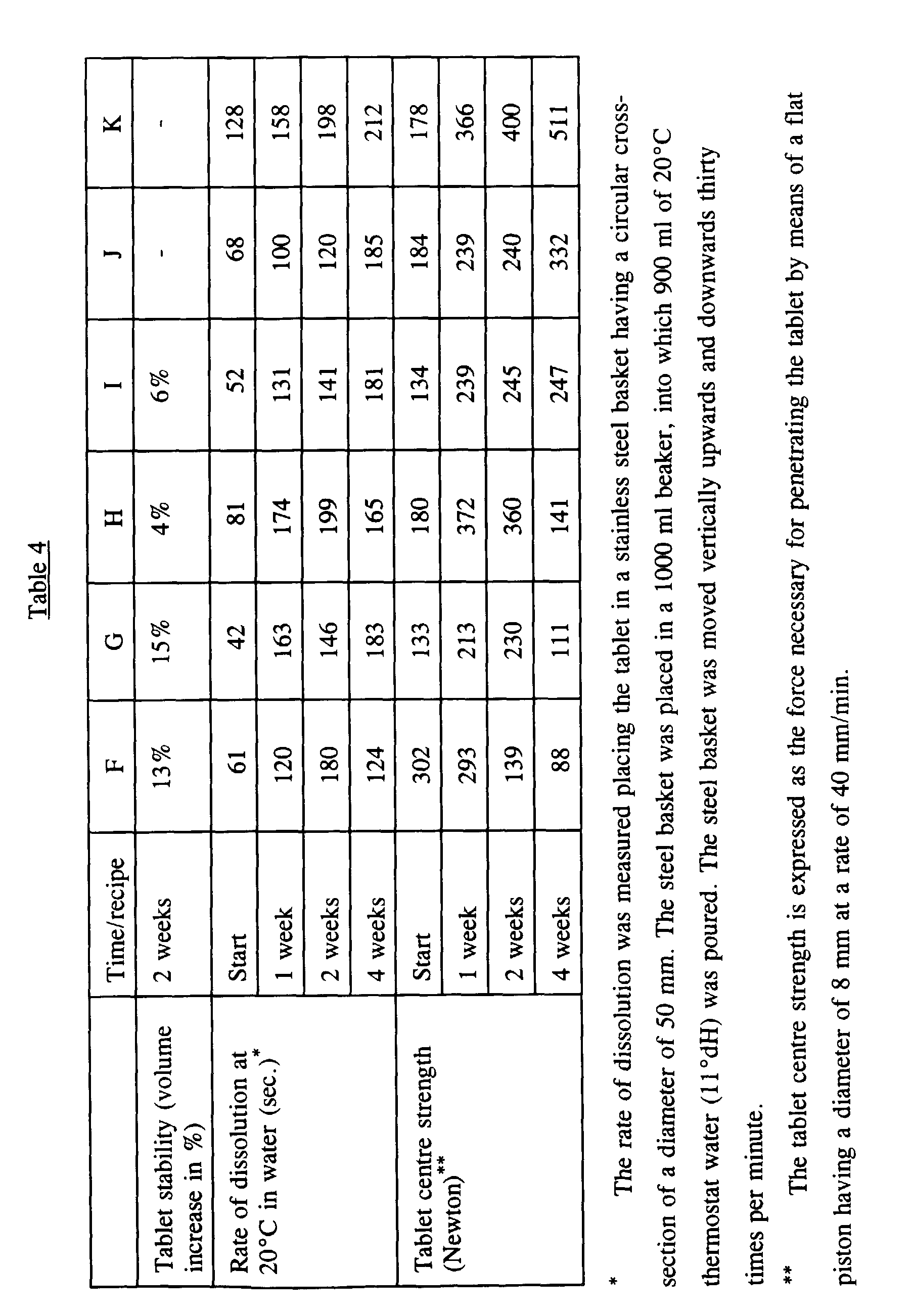
- Based on the following recipes bleach tablets each weighing 20 g were prepared. The tablets according to recipes F, J and K all had square cross-sections of 33 x 33 mm and heights of approximately 14 to 16 mm. The tablets according to recipes G, H and I all had square cross-sections of 25 mm x 35 mm and heights of approximately 17 to 19 mm.
- As described above in Example 2, accelerated aging test were performed to illustrate the good stability and tablet-technical properties of the tablets stated in Example 3.
-
- The present tests were carried out so as to compare the bleaching effect of the bleach tablets known from EP publication No. 0 481 792 with the bleaching effect of the bleach tablets according to the invention.
-
-
- The results appear from the following Table 5. The reflectance values stated in the table indicate the effeciency of the bleach together with the basic detergent composition used to remove the impregnated stains. A high value means a good efficiency, the theoretically highest possible value being 100 corresponding to a completely white surface.
- The cleaning effect is calculated as the percentage increase of the reflectance value compared to an untreated test cloth. The cleaning index is calculated on the basis of the cleaning effect, the highest value of the cleaning effect (Ariel Color + 1 x 30 g recipe E on soil of the type BC1PC-Tea on blend fibres, 25) being set to 100.
- As it appears from the results, the bleach tablets according to the invention show an improved bleaching effect over the bleach tablets known from EP publication No. 0 481 792.
- The present tests were carried out so as to compare the rate of dissolution of the bleach tablets known from EP publication No. 0 481 792 with the rate of dissolution of the bleach tablets according to the invention. The rate of dissolution were determined as stated in Example 4.
- At the tests the recipe according to EP publication No. 481 792, Ex. 4, stated in Example 5 above was used.
- The results appear from the following Table 6.
Table 6 Rate of dissolution at 20°C in water (sec.) Start 2020 2 weeks 1838 4 weeks 1725 - When comparing the results obtained by the bleach tablets as stated in Tables 2 and 4 with the results stated in Table 6, the bleach tablets according to the invention clearly display a superior rate of dissolution in relation to the bleach tablets known from EP publication No. 0 481 792.
- The invention being thus described, it will be obvious that the same may be varied in many ways. Such variations are not to be regarded as a departure from the spirit and scope of the invention, and all such modifications as would be obvious to a person skilled in the art are intended to be included within the scope of the following claims.
Claims (10)
- Bleach tablets, characterised in that in addition to optional usual adjuvants and additives they comprise
coated percarbonate 45 to 85 % by weight bleach activator, calculated as TAED 0 to 20 % by weight layered silicate and/or alkali metal silicate 1 to 50 % by weight sodium/potassium carbonate 0 to 25 % by weight sodium/potassium bicarbonate 0 to 25 % by weight polyfunctional carboxylic acid, calculated as citric acid 5 to 35 % by weight polyethylene glycol 0 to 8 % by weight complex-binder 0 to 5 % by weight paraffin oil 0 to 2 % by weight disintegrating agent 0 to 5 % by weight enzymes 0 to 10 % by weight polymer 0 to 10 % by weight - Bleach tablets as as claimed in claim 1, characterised in that the coated percarbonate is an alkali metal percarbonate.
- Bleach tablets as claimed in claim 1, characterised in that as bleach activator they contain TAED in an amount of from 4 to 15 % by weight.
- Bleach tablets as claimed in claim 3, characterised in that as bleach activator they contain TAED in an amount of from 4 to 10 % by weight.
- Bleach tablets as claimed in claim 1, characterised in that the alkali metal silicate is sodium disilicate.
- Bleach tablets as claimed in claim 1, characterised in that as polyfunctional carboxylic acid they contain citric acid.
- Bleach tablets as claimed in claim 1, characterised in that the polyethylene glycol has a molecular weight of from 200 to 8,000.
- Bleach tablets as claimed in claim 1, characterised in that the disintegrating agent is a cellulose-modified or cellulose-based compound.
- Bleach tablets as claimed in claim 1, characterised in that in addition to optional usual adjuvants and additives they comprise
coated sodium percarbonate 55 to 65 % by weight TAED 4 to 12 % by weight layered silicate 3 to 6 % by weight sodium carbonate 3 to 6 % by weight citric acid 18 to 22 % by weight polyethylene glycol 2 to 5 % by weight paraffin oil 0.1 to 1.0 % by weight disintegrating agent 0 to 4 % by weight polymer 0 to 10 % by weight
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DK446/95 | 1995-04-12 | ||
| DK44695 | 1995-04-12 | ||
| DK44695 | 1995-04-12 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP0737738A2 true EP0737738A2 (en) | 1996-10-16 |
| EP0737738A3 EP0737738A3 (en) | 1998-04-01 |
| EP0737738B1 EP0737738B1 (en) | 2003-06-25 |
Family
ID=8093639
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP19960610014 Expired - Lifetime EP0737738B1 (en) | 1995-04-12 | 1996-04-11 | Bleach tablets |
Country Status (4)
| Country | Link |
|---|---|
| EP (1) | EP0737738B1 (en) |
| DE (1) | DE69628784T2 (en) |
| DK (1) | DK0737738T3 (en) |
| ES (1) | ES2201162T3 (en) |
Cited By (40)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE19709991A1 (en) * | 1997-03-11 | 1998-09-17 | Herzog Stefan | Liquid disintegrating compact |
| EP0799886A3 (en) * | 1996-04-03 | 1998-11-18 | Cleantabs A/S | Laundry detergent tablets |
| US5854189A (en) * | 1994-03-15 | 1998-12-29 | Kruse; Hans | Process for the production of break-resistant, storable multifunctional detergent tablets |
| GB2327949A (en) * | 1997-08-02 | 1999-02-10 | Procter & Gamble | Detergent tablet |
| US5900399A (en) * | 1994-02-10 | 1999-05-04 | Henkel Kommanditgesellschaft Auf Aktien | Tablet containing builders |
| WO1999024547A1 (en) * | 1997-11-10 | 1999-05-20 | The Procter & Gamble Company | Detergent compositions |
| WO1999055818A1 (en) * | 1998-04-27 | 1999-11-04 | The Procter & Gamble Company | Non-particulate detergent product containing bleach activator |
| GB2340840A (en) * | 1998-08-28 | 2000-03-01 | Procter & Gamble | Detergent tablet |
| WO2000017307A1 (en) * | 1998-09-24 | 2000-03-30 | Henkel Kommanditgesellschaft Auf Aktien | Washing and cleaning agent shaped bodies comprising sodium percarbonate |
| GB2349390A (en) * | 1999-04-30 | 2000-11-01 | Procter & Gamble | Cleaning compositions comprising compressed clay |
| WO2000071666A1 (en) * | 1999-05-21 | 2000-11-30 | Unilever Plc | Detergent compositions |
| DE19937428A1 (en) * | 1999-08-07 | 2001-02-08 | Henkel Kgaa | Detergent tablets |
| GB2353800A (en) * | 1999-09-02 | 2001-03-07 | Procter & Gamble | Antibacterial detergent compositions |
| US6274538B1 (en) | 1997-11-10 | 2001-08-14 | The Procter & Gamble Company | Detergent compositions |
| US6329335B1 (en) * | 1997-03-07 | 2001-12-11 | Henkel Kommanditgesellschaft Auf Aktien | Detergent tablets |
| WO2002024848A3 (en) * | 2000-09-20 | 2002-08-08 | Reckitt Benckiser Inc | Improvements in enzyme containing tablets |
| US6462006B1 (en) * | 1998-04-30 | 2002-10-08 | Henkel Kommanditgesellschaft Auf Aktien | Solid machine dishwashing detergent with phosphate and crystalline lamellar silicates |
| GB2375543A (en) * | 2001-05-18 | 2002-11-20 | Reckitt Benckiser Inc | Laundry additive compositions |
| US6506720B1 (en) | 1997-03-13 | 2003-01-14 | Henkel Kommanditgesellschaft Auf Aktien | Process for preparing household detergent or cleaner shapes |
| WO2003062360A1 (en) * | 2002-01-23 | 2003-07-31 | The Procter & Gamble Company | Improved solubility aids for use in detergent compositions |
| DE10358827A1 (en) * | 2003-12-16 | 2005-07-28 | Henkel Kgaa | Bleaching detergent or cleaner |
| WO2006063668A1 (en) * | 2004-12-14 | 2006-06-22 | Degussa Gmbh | Compressed molded bodies containing coated sodium percarbonate particles |
| FR2885302A1 (en) * | 2005-05-09 | 2006-11-10 | Hypred Sa | Use of a composition in preparation of cleaning and/or disinfecting solution of mops for cleaning cow milk or bundle milk |
| EP1633847A4 (en) * | 2003-05-30 | 2007-04-11 | Church & Dwight Co Inc | Detergent formulations containing alkaline peroxide salts and organic acids |
| US7435714B2 (en) | 2002-12-20 | 2008-10-14 | Evonik Degussa Gmbh | Liquid detergent and cleaning agent composition comprising a multi-coated bleach particle |
| WO2009017748A1 (en) * | 2007-07-30 | 2009-02-05 | Milliken & Company | Solid cleaning composition and method of use |
| US7588697B2 (en) | 2003-05-07 | 2009-09-15 | Evonik Degussa Gmbh | Coated sodium percarbonate granules with improved storage stability |
| US7718592B2 (en) | 2004-11-11 | 2010-05-18 | Degussa, Gmbh | Sodium percarbonate particles having a shell layer comprising thiosulfate |
| WO2010094924A1 (en) * | 2009-02-23 | 2010-08-26 | Reckitt Benckiser N.V. | Percarbonate containing detergent product |
| WO2011134972A1 (en) * | 2010-04-27 | 2011-11-03 | Evonik Degussa Gmbh | Bleaching agent particles comprising sodium percarbonate and a bleach activator |
| US8658590B2 (en) | 2006-07-27 | 2014-02-25 | Evonik Degussa Gmbh | Coated sodium percarbonate particles |
| US8945671B2 (en) | 2007-12-19 | 2015-02-03 | Evonik Treibacher Gmbh | Method for producing encapsulated sodium percarbonate particles |
| FR3011006A1 (en) * | 2013-09-25 | 2015-03-27 | Guillaume Laurent Georges Ropars | MULTI-PURPOSE DETERGENT FOR TEXTILES |
| DE102014218951A1 (en) * | 2014-09-19 | 2016-03-24 | Henkel Ag & Co. Kgaa | Solid composition for textile treatment |
| DE102014218950A1 (en) * | 2014-09-19 | 2016-03-24 | Henkel Ag & Co. Kgaa | Solid composition for textile treatment |
| WO2016085687A1 (en) * | 2014-11-25 | 2016-06-02 | Milliken & Company | Cleaning composition |
| DE19919444B4 (en) * | 1999-04-29 | 2016-06-30 | Henkel Ag & Co. Kgaa | Detergent tablets with binder compound, process for its preparation and use of binder compounds |
| US9783766B2 (en) | 2015-04-03 | 2017-10-10 | Ecolab Usa Inc. | Enhanced peroxygen stability using anionic surfactant in TAED-containing peroxygen solid |
| US10280386B2 (en) | 2015-04-03 | 2019-05-07 | Ecolab Usa Inc. | Enhanced peroxygen stability in multi-dispense TAED-containing peroxygen solid |
| US10870818B2 (en) | 2018-06-15 | 2020-12-22 | Ecolab Usa Inc. | Enhanced peroxygen stability using fatty acid in bleach activating agent containing peroxygen solid |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11659838B2 (en) | 2021-04-01 | 2023-05-30 | Sterilex, Llc | Quat-free powdered disinfectant/sanitizer |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0550077A1 (en) | 1992-01-03 | 1993-07-07 | The Procter & Gamble Company | Granular laundry bleaching composition |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB9022724D0 (en) * | 1990-10-19 | 1990-12-05 | Unilever Plc | Detergent compositions |
| DE4424005A1 (en) * | 1994-07-07 | 1996-01-11 | Henkel Kgaa | Powdery bleach and detergent |
-
1996
- 1996-04-11 ES ES96610014T patent/ES2201162T3/en not_active Expired - Lifetime
- 1996-04-11 DK DK96610014T patent/DK0737738T3/en active
- 1996-04-11 EP EP19960610014 patent/EP0737738B1/en not_active Expired - Lifetime
- 1996-04-11 DE DE69628784T patent/DE69628784T2/en not_active Expired - Fee Related
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0550077A1 (en) | 1992-01-03 | 1993-07-07 | The Procter & Gamble Company | Granular laundry bleaching composition |
Cited By (53)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5900399A (en) * | 1994-02-10 | 1999-05-04 | Henkel Kommanditgesellschaft Auf Aktien | Tablet containing builders |
| US5854189A (en) * | 1994-03-15 | 1998-12-29 | Kruse; Hans | Process for the production of break-resistant, storable multifunctional detergent tablets |
| EP0799886A3 (en) * | 1996-04-03 | 1998-11-18 | Cleantabs A/S | Laundry detergent tablets |
| US6329335B1 (en) * | 1997-03-07 | 2001-12-11 | Henkel Kommanditgesellschaft Auf Aktien | Detergent tablets |
| DE19709991C2 (en) * | 1997-03-11 | 1999-12-23 | Rettenmaier & Soehne Gmbh & Co | Detergent compact and process for its manufacture |
| DE19709991A1 (en) * | 1997-03-11 | 1998-09-17 | Herzog Stefan | Liquid disintegrating compact |
| US7008912B1 (en) | 1997-03-11 | 2006-03-07 | Henkel Kgaa | Pressed piece which disintegrates in liquids |
| US6506720B1 (en) | 1997-03-13 | 2003-01-14 | Henkel Kommanditgesellschaft Auf Aktien | Process for preparing household detergent or cleaner shapes |
| USRE39139E1 (en) * | 1997-03-13 | 2006-06-20 | Henkel Kgaa | Process for preparing household detergent or cleaner shapes |
| GB2327949A (en) * | 1997-08-02 | 1999-02-10 | Procter & Gamble | Detergent tablet |
| WO1999024547A1 (en) * | 1997-11-10 | 1999-05-20 | The Procter & Gamble Company | Detergent compositions |
| US6274538B1 (en) | 1997-11-10 | 2001-08-14 | The Procter & Gamble Company | Detergent compositions |
| WO1999055818A1 (en) * | 1998-04-27 | 1999-11-04 | The Procter & Gamble Company | Non-particulate detergent product containing bleach activator |
| US6358902B1 (en) | 1998-04-27 | 2002-03-19 | The Procter & Gamble Company | Detergent tablet containing bleach activator of specific particle size |
| US6462006B1 (en) * | 1998-04-30 | 2002-10-08 | Henkel Kommanditgesellschaft Auf Aktien | Solid machine dishwashing detergent with phosphate and crystalline lamellar silicates |
| GB2340840A (en) * | 1998-08-28 | 2000-03-01 | Procter & Gamble | Detergent tablet |
| WO2000017307A1 (en) * | 1998-09-24 | 2000-03-30 | Henkel Kommanditgesellschaft Auf Aktien | Washing and cleaning agent shaped bodies comprising sodium percarbonate |
| DE19919444B4 (en) * | 1999-04-29 | 2016-06-30 | Henkel Ag & Co. Kgaa | Detergent tablets with binder compound, process for its preparation and use of binder compounds |
| GB2349390A (en) * | 1999-04-30 | 2000-11-01 | Procter & Gamble | Cleaning compositions comprising compressed clay |
| WO2000071666A1 (en) * | 1999-05-21 | 2000-11-30 | Unilever Plc | Detergent compositions |
| US6387861B1 (en) | 1999-05-21 | 2002-05-14 | Unilever Home & Personal Care Usa Division Of Conopco, Inc. | Detergent compositions |
| DE19937428A1 (en) * | 1999-08-07 | 2001-02-08 | Henkel Kgaa | Detergent tablets |
| GB2353800A (en) * | 1999-09-02 | 2001-03-07 | Procter & Gamble | Antibacterial detergent compositions |
| WO2002024848A3 (en) * | 2000-09-20 | 2002-08-08 | Reckitt Benckiser Inc | Improvements in enzyme containing tablets |
| GB2375543A (en) * | 2001-05-18 | 2002-11-20 | Reckitt Benckiser Inc | Laundry additive compositions |
| WO2003062360A1 (en) * | 2002-01-23 | 2003-07-31 | The Procter & Gamble Company | Improved solubility aids for use in detergent compositions |
| US7435714B2 (en) | 2002-12-20 | 2008-10-14 | Evonik Degussa Gmbh | Liquid detergent and cleaning agent composition comprising a multi-coated bleach particle |
| US7588697B2 (en) | 2003-05-07 | 2009-09-15 | Evonik Degussa Gmbh | Coated sodium percarbonate granules with improved storage stability |
| EP1633847A4 (en) * | 2003-05-30 | 2007-04-11 | Church & Dwight Co Inc | Detergent formulations containing alkaline peroxide salts and organic acids |
| DE10358827A1 (en) * | 2003-12-16 | 2005-07-28 | Henkel Kgaa | Bleaching detergent or cleaner |
| US7718592B2 (en) | 2004-11-11 | 2010-05-18 | Degussa, Gmbh | Sodium percarbonate particles having a shell layer comprising thiosulfate |
| WO2006063668A1 (en) * | 2004-12-14 | 2006-06-22 | Degussa Gmbh | Compressed molded bodies containing coated sodium percarbonate particles |
| EP1741451A1 (en) * | 2005-05-09 | 2007-01-10 | Societe Hypred | Cleaning and desinfecting product and method in the field of milk production hygiene |
| FR2885302A1 (en) * | 2005-05-09 | 2006-11-10 | Hypred Sa | Use of a composition in preparation of cleaning and/or disinfecting solution of mops for cleaning cow milk or bundle milk |
| US8658590B2 (en) | 2006-07-27 | 2014-02-25 | Evonik Degussa Gmbh | Coated sodium percarbonate particles |
| WO2009017748A1 (en) * | 2007-07-30 | 2009-02-05 | Milliken & Company | Solid cleaning composition and method of use |
| US8945671B2 (en) | 2007-12-19 | 2015-02-03 | Evonik Treibacher Gmbh | Method for producing encapsulated sodium percarbonate particles |
| WO2010094924A1 (en) * | 2009-02-23 | 2010-08-26 | Reckitt Benckiser N.V. | Percarbonate containing detergent product |
| WO2011134972A1 (en) * | 2010-04-27 | 2011-11-03 | Evonik Degussa Gmbh | Bleaching agent particles comprising sodium percarbonate and a bleach activator |
| FR3011006A1 (en) * | 2013-09-25 | 2015-03-27 | Guillaume Laurent Georges Ropars | MULTI-PURPOSE DETERGENT FOR TEXTILES |
| DE102014218951A1 (en) * | 2014-09-19 | 2016-03-24 | Henkel Ag & Co. Kgaa | Solid composition for textile treatment |
| DE102014218950A1 (en) * | 2014-09-19 | 2016-03-24 | Henkel Ag & Co. Kgaa | Solid composition for textile treatment |
| CN107148466B (en) * | 2014-11-25 | 2021-05-18 | 美利肯公司 | cleaning composition |
| WO2016085687A1 (en) * | 2014-11-25 | 2016-06-02 | Milliken & Company | Cleaning composition |
| CN107148466A (en) * | 2014-11-25 | 2017-09-08 | 美利肯公司 | cleaning composition |
| CN113214924A (en) * | 2014-11-25 | 2021-08-06 | 美利肯公司 | Cleaning composition |
| US10280386B2 (en) | 2015-04-03 | 2019-05-07 | Ecolab Usa Inc. | Enhanced peroxygen stability in multi-dispense TAED-containing peroxygen solid |
| US10557106B2 (en) | 2015-04-03 | 2020-02-11 | Ecolab Usa Inc. | Enhanced peroxygen stability using anionic surfactant in TAED-containing peroxygen solid |
| US11053459B2 (en) | 2015-04-03 | 2021-07-06 | Ecolab Usa Inc. | Enhanced peroxygen stability in multi-dispense TAED-containing peroxygen solid |
| US9783766B2 (en) | 2015-04-03 | 2017-10-10 | Ecolab Usa Inc. | Enhanced peroxygen stability using anionic surfactant in TAED-containing peroxygen solid |
| US11731889B2 (en) | 2015-04-03 | 2023-08-22 | Ecolab Usa Inc. | Enhanced peroxygen stability in multi-dispense TAED-containing peroxygen solid |
| US10870818B2 (en) | 2018-06-15 | 2020-12-22 | Ecolab Usa Inc. | Enhanced peroxygen stability using fatty acid in bleach activating agent containing peroxygen solid |
| US11193093B2 (en) | 2018-06-15 | 2021-12-07 | Ecolab Usa Inc. | Enhanced peroxygen stability using fatty acid in bleach activating agent containing peroxygen solid |
Also Published As
| Publication number | Publication date |
|---|---|
| EP0737738A3 (en) | 1998-04-01 |
| DK0737738T3 (en) | 2003-10-20 |
| ES2201162T3 (en) | 2004-03-16 |
| EP0737738B1 (en) | 2003-06-25 |
| DE69628784T2 (en) | 2004-02-12 |
| DE69628784D1 (en) | 2003-07-31 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0737738B1 (en) | Bleach tablets | |
| US5898025A (en) | Mildly alkaline dishwashing detergents | |
| EP0504091B1 (en) | A phosphate-free automatic dishwashing composition | |
| JP2941430B2 (en) | Bleaching composition containing a metal-containing bleaching catalyst | |
| JP3009471B2 (en) | Bleaching composition comprising a metal-containing bleaching catalyst and an antioxidant | |
| US5246612A (en) | Machine dishwashing composition containing peroxygen bleach, manganese complex and enzymes | |
| US3637339A (en) | Stain removal | |
| JP2628812B2 (en) | Detergent composition | |
| CA2145663A1 (en) | Mildly alkaline dishwashing detergents | |
| EP0799886B1 (en) | Laundry detergent tablets | |
| JPH05112799A (en) | Concentrated detergent powder composition | |
| EP1741774A1 (en) | Machine dishwashing compositions and their use | |
| MXPA02002396A (en) | Bleaching detergent compositions. | |
| US4079016A (en) | Solid compositions for washing, cleaning and bleaching and processes for their manufacture and their use | |
| JP2016529338A (en) | A composition comprising glutamic acid-N, N-diacetate (GLDA), water and an enzyme | |
| JP2611071B2 (en) | Detergent composition | |
| US6174852B1 (en) | High-density powdered detergent composition | |
| JP2010516832A (en) | Dish detergent | |
| JPH0317199A (en) | Detergent compounds | |
| US3725289A (en) | Stain removing composition | |
| AU635381B2 (en) | Bleaching process and bleach compositions | |
| JPH08245985A (en) | Washing and bleaching composition | |
| CN100390258C (en) | detergent composition | |
| CA2166277A1 (en) | Dishwashing detergents containing a biologically degradable builder component | |
| CN115151629A (en) | Dishwashing detergent products |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): DE DK ES FR GB IT |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): DE DK ES FR GB IT |
|
| 17P | Request for examination filed |
Effective date: 19980907 |
|
| 17Q | First examination report despatched |
Effective date: 20020117 |
|
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Designated state(s): DE DK ES FR GB IT |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D |
|
| REF | Corresponds to: |
Ref document number: 69628784 Country of ref document: DE Date of ref document: 20030731 Kind code of ref document: P |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2201162 Country of ref document: ES Kind code of ref document: T3 |
|
| ET | Fr: translation filed | ||
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed |
Effective date: 20040326 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: ES Payment date: 20090422 Year of fee payment: 14 Ref country code: DK Payment date: 20090423 Year of fee payment: 14 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IT Payment date: 20090424 Year of fee payment: 14 Ref country code: FR Payment date: 20090430 Year of fee payment: 14 Ref country code: DE Payment date: 20090429 Year of fee payment: 14 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20090429 Year of fee payment: 14 |
|
| REG | Reference to a national code |
Ref country code: DK Ref legal event code: EBP |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20100411 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST Effective date: 20101230 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20101103 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100411 Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100411 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DK Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100503 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FD2A Effective date: 20110715 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20110705 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100412 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100430 |