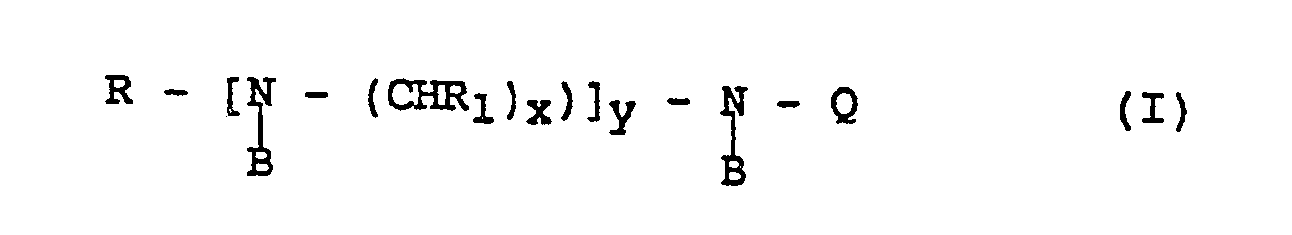EP0433257A1 - A process for enhancing the bleaching effect at washing and use of certain amphoteric compounds in a detergent composition for enhancing the bleaching effect - Google Patents
A process for enhancing the bleaching effect at washing and use of certain amphoteric compounds in a detergent composition for enhancing the bleaching effect Download PDFInfo
- Publication number
- EP0433257A1 EP0433257A1 EP90850401A EP90850401A EP0433257A1 EP 0433257 A1 EP0433257 A1 EP 0433257A1 EP 90850401 A EP90850401 A EP 90850401A EP 90850401 A EP90850401 A EP 90850401A EP 0433257 A1 EP0433257 A1 EP 0433257A1
- Authority
- EP
- European Patent Office
- Prior art keywords
- weight
- per cent
- carbon atoms
- amphoteric compound
- washing
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 150000001875 compounds Chemical class 0.000 title claims abstract description 53
- 239000000203 mixture Substances 0.000 title claims abstract description 42
- 239000003599 detergent Substances 0.000 title claims abstract description 30
- 230000000694 effects Effects 0.000 title claims abstract description 17
- 238000005406 washing Methods 0.000 title claims abstract description 16
- 238000004061 bleaching Methods 0.000 title claims abstract description 14
- 238000000034 method Methods 0.000 title claims description 9
- 230000002708 enhancing effect Effects 0.000 title claims description 5
- 150000001412 amines Chemical class 0.000 claims abstract description 22
- 125000004432 carbon atom Chemical group C* 0.000 claims abstract description 16
- 239000001257 hydrogen Substances 0.000 claims abstract description 14
- 229910052739 hydrogen Inorganic materials 0.000 claims abstract description 14
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims abstract description 13
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 claims abstract description 9
- 125000000217 alkyl group Chemical group 0.000 claims abstract description 8
- 229910052783 alkali metal Inorganic materials 0.000 claims abstract description 7
- 150000001340 alkali metals Chemical class 0.000 claims abstract description 7
- 125000001183 hydrocarbyl group Chemical group 0.000 claims abstract description 7
- 229910052784 alkaline earth metal Inorganic materials 0.000 claims abstract description 5
- 150000001342 alkaline earth metals Chemical class 0.000 claims abstract description 5
- 125000002947 alkylene group Chemical group 0.000 claims abstract description 5
- 125000000129 anionic group Chemical group 0.000 claims description 4
- 125000000816 ethylene group Chemical group [H]C([H])([*:1])C([H])([H])[*:2] 0.000 claims description 2
- MHAJPDPJQMAIIY-UHFFFAOYSA-N Hydrogen peroxide Chemical compound OO MHAJPDPJQMAIIY-UHFFFAOYSA-N 0.000 description 13
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 7
- 239000004094 surface-active agent Substances 0.000 description 6
- 235000014113 dietary fatty acids Nutrition 0.000 description 4
- 239000000194 fatty acid Substances 0.000 description 4
- 229930195729 fatty acid Natural products 0.000 description 4
- 150000004665 fatty acids Chemical class 0.000 description 4
- -1 for example mono- Chemical class 0.000 description 4
- 239000003760 tallow Substances 0.000 description 4
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 3
- 239000008139 complexing agent Substances 0.000 description 3
- 239000010457 zeolite Substances 0.000 description 3
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 2
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 2
- 229910019142 PO4 Inorganic materials 0.000 description 2
- BGRWYDHXPHLNKA-UHFFFAOYSA-N Tetraacetylethylenediamine Chemical compound CC(=O)N(C(C)=O)CCN(C(C)=O)C(C)=O BGRWYDHXPHLNKA-UHFFFAOYSA-N 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 238000007792 addition Methods 0.000 description 2
- 125000003342 alkenyl group Chemical group 0.000 description 2
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 2
- 239000007844 bleaching agent Substances 0.000 description 2
- 239000011575 calcium Substances 0.000 description 2
- 238000000354 decomposition reaction Methods 0.000 description 2
- 150000002191 fatty alcohols Chemical class 0.000 description 2
- 238000009472 formulation Methods 0.000 description 2
- 150000002500 ions Chemical group 0.000 description 2
- 239000011777 magnesium Substances 0.000 description 2
- 229910021645 metal ion Inorganic materials 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- ZBJVLWIYKOAYQH-UHFFFAOYSA-N naphthalen-2-yl 2-hydroxybenzoate Chemical compound OC1=CC=CC=C1C(=O)OC1=CC=C(C=CC=C2)C2=C1 ZBJVLWIYKOAYQH-UHFFFAOYSA-N 0.000 description 2
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 2
- 239000001301 oxygen Substances 0.000 description 2
- 229910052760 oxygen Inorganic materials 0.000 description 2
- 235000021317 phosphate Nutrition 0.000 description 2
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 2
- 239000000344 soap Substances 0.000 description 2
- 229910052708 sodium Inorganic materials 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 229910052938 sodium sulfate Inorganic materials 0.000 description 2
- 235000011152 sodium sulphate Nutrition 0.000 description 2
- BDHFUVZGWQCTTF-UHFFFAOYSA-M sulfonate Chemical compound [O-]S(=O)=O BDHFUVZGWQCTTF-UHFFFAOYSA-M 0.000 description 2
- HKLYDUXIXBVZOQ-UHFFFAOYSA-N 2-aminoethane-1,1,1-triol Chemical compound NCC(O)(O)O HKLYDUXIXBVZOQ-UHFFFAOYSA-N 0.000 description 1
- OCKGFTQIICXDQW-ZEQRLZLVSA-N 5-[(1r)-1-hydroxy-2-[4-[(2r)-2-hydroxy-2-(4-methyl-1-oxo-3h-2-benzofuran-5-yl)ethyl]piperazin-1-yl]ethyl]-4-methyl-3h-2-benzofuran-1-one Chemical compound C1=C2C(=O)OCC2=C(C)C([C@@H](O)CN2CCN(CC2)C[C@H](O)C2=CC=C3C(=O)OCC3=C2C)=C1 OCKGFTQIICXDQW-ZEQRLZLVSA-N 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-M Bicarbonate Chemical class OC([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-M 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- PWHULOQIROXLJO-UHFFFAOYSA-N Manganese Chemical compound [Mn] PWHULOQIROXLJO-UHFFFAOYSA-N 0.000 description 1
- ABLZXFCXXLZCGV-UHFFFAOYSA-N Phosphorous acid Chemical class OP(O)=O ABLZXFCXXLZCGV-UHFFFAOYSA-N 0.000 description 1
- 241001122767 Theaceae Species 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 239000012190 activator Substances 0.000 description 1
- 238000005054 agglomeration Methods 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 150000001335 aliphatic alkanes Chemical group 0.000 description 1
- 150000003973 alkyl amines Chemical class 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- 230000000845 anti-microbial effect Effects 0.000 description 1
- 239000004599 antimicrobial Substances 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 125000003710 aryl alkyl group Chemical group 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 150000004649 carbonic acid derivatives Chemical class 0.000 description 1
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 1
- 150000001768 cations Chemical class 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 230000000536 complexating effect Effects 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 125000001316 cycloalkyl alkyl group Chemical group 0.000 description 1
- 239000004744 fabric Substances 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 239000006260 foam Substances 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- 238000004900 laundering Methods 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 229910052748 manganese Inorganic materials 0.000 description 1
- 239000011572 manganese Substances 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 125000001570 methylene group Chemical group [H]C([H])([*:1])[*:2] 0.000 description 1
- 125000000325 methylidene group Chemical group [H]C([H])=* 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 239000002304 perfume Substances 0.000 description 1
- 150000004965 peroxy acids Chemical class 0.000 description 1
- 229920005646 polycarboxylate Polymers 0.000 description 1
- 159000000001 potassium salts Chemical class 0.000 description 1
- 235000019353 potassium silicate Nutrition 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 235000020095 red wine Nutrition 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 239000002453 shampoo Substances 0.000 description 1
- 239000002002 slurry Substances 0.000 description 1
- 235000017557 sodium bicarbonate Nutrition 0.000 description 1
- 235000011182 sodium carbonates Nutrition 0.000 description 1
- 239000012418 sodium perborate tetrahydrate Substances 0.000 description 1
- 159000000000 sodium salts Chemical class 0.000 description 1
- NTHWMYGWWRZVTN-UHFFFAOYSA-N sodium silicate Chemical compound [Na+].[Na+].[O-][Si]([O-])=O NTHWMYGWWRZVTN-UHFFFAOYSA-N 0.000 description 1
- AKHNMLFCWUSKQB-UHFFFAOYSA-L sodium thiosulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=S AKHNMLFCWUSKQB-UHFFFAOYSA-L 0.000 description 1
- 235000019345 sodium thiosulphate Nutrition 0.000 description 1
- 235000019832 sodium triphosphate Nutrition 0.000 description 1
- IBDSNZLUHYKHQP-UHFFFAOYSA-N sodium;3-oxidodioxaborirane;tetrahydrate Chemical compound O.O.O.O.[Na+].[O-]B1OO1 IBDSNZLUHYKHQP-UHFFFAOYSA-N 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 238000001694 spray drying Methods 0.000 description 1
- 125000001424 substituent group Chemical group 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 150000004685 tetrahydrates Chemical class 0.000 description 1
- 239000004711 α-olefin Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/39—Organic or inorganic per-compounds
- C11D3/3902—Organic or inorganic per-compounds combined with specific additives
- C11D3/3905—Bleach activators or bleach catalysts
- C11D3/3907—Organic compounds
- C11D3/3917—Nitrogen-containing compounds
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D1/00—Detergent compositions based essentially on surface-active compounds; Use of these compounds as a detergent
- C11D1/88—Ampholytes; Electroneutral compounds
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D1/00—Detergent compositions based essentially on surface-active compounds; Use of these compounds as a detergent
- C11D1/88—Ampholytes; Electroneutral compounds
- C11D1/94—Mixtures with anionic, cationic or non-ionic compounds
Definitions
- the invention relates to a method for enhancing the bleaching effect at washing in the presence of perborates.
- the bleaching effect is enhanced by using certain amphoteric compounds, or amine oxides of these, and the invention also relates to the use of such compounds in detergent compositions containing perborates.
- R is a higher hydrocarbon group
- R1 is hydrogen or a lower alkyl group
- x 2 or 3
- y is 0 to 4
- Q is a group ⁇ R2COOM wherein R2 is an alkylene group and M is hydrogen or another cation and B is hydrogen or a group Q are per se known.
- Compounds of this type are disclosed in the European patent applications 160507, 162600 and 214868. The compounds are used in detergent and shampoo compositions for surface activity and for their antimicrobial properties.
- amphoteric compounds of this type in combination with zeolites in detergent compositions since it has been found that they show a synergistic effect at softening of water. It is also evident from this application that the detergent compositions may contain perborates as bleaching agents. However, the amphoteric compound should be present in fairly high amounts to give the desired effect. Further, it is advised against the use of the amphoteric compounds in detergent compositions containing anionic tensides.
- amphoteric compounds or amine oxides of these, enhance the bleaching effect at washing in the presence of perborates.
- the amphoteric compounds and their amine oxides can inhibit the decomposition of hydrogen peroxide to free radicals at washing, which probably depends on their complexing ability.
- the invention thus relates to a process for enhancing the bleaching effect at washing under alkaline conditions in the presence of perborates as defined in the patent claims.
- the washing is carried out under alkaline conditions using a detergent composition containing perborate and up to 4.5 per cent by weight of at least one amphoteric compound of the general formula (I) wherein R is a hydrocarbon group having 7 to 22 carbon atoms, R1 is hydrogen or alkyl with 1 to 6 carbon atoms, x is 2 or 3, y is 0 or an integer of 1 to 4 and Q is (R2-COOM) where R2 is alkylene with 1 to 6 carbon atoms, M is H, an alkali metal, an alkaline earth metal, an ammonium or a substituted ammonium ion, and B is hydrogen or is defined according to Q, or a detergent composition containing perborate and up to 4.5 per cent by weight of at least one amine oxide of the defined amphoteric compound.
- R is a hydrocarbon group having 7 to 22
- the composition can thus contain corresponding amounts of at least one amine oxide of amphoteric compound as above defined.
- One or several of the nitrogen groups in the amphoteric compounds can be oxidized.
- Useful compounds can be defined by the general formula (II) wherein R, R1, x, y, Q, R2, M and B have the above given definitions.
- the term amine oxide is used to include also compounds wherein only some of the amine groups have been oxidized.
- the amine oxides of the defined amphoteric compounds are new compounds and can also be used for the same purposes as the previously known amphoteric compounds of formula (I), ie as tensides, antimicrobial agents or take part in softening of water.
- the hydrocarbon group R can be straight or branched, saturated or unsaturated, and may contain substituents such as hydroxyl groups or carbonyl groups.
- R is preferably an alkyl or alkenyl group, but can also be a cycloalkyl-alkyl group, an aralkyl or aralkenyl group where the alkyl or alkenyl group contains at least 6 carbon atoms.
- R suitably contains 12 to 22 carbon atoms, preferably 12 to 20 carbon atoms.
- R is a hydrocarbon group originating from natural fatty acids and particularly from tallow or coco fatty acid.
- R1 is hydrogen or a lower alkyl group, suitably with 1 to 6 carbon atoms and preferably hydrogen or a methyl group.
- x is 2 or 3 and y is suitably 1, 2, 3 or 4 and preferably 1, 2 or 3.
- the group R2 is suitably a methylene or ethylene group, preferably a methylene group.
- M is hydrogen or an ion from the groups alkali metals, alkaline earth metals, ammonium or substituted ammonium such as for example mono-, di- or trihydroxyethylammonium.
- M is preferably alkali metal, especially sodium. It is particularly preferred that all groups B in the compounds of the above given general formulae are groups Q.
- Amphoteric compounds which can be used according to the present invention are per se previously known, for example from the earlier mentioned patent applications. They are also commercially available under the trade name Ampholak (R) . Amine oxides can be prepared by allowing one of the earlier known amphoteric compounds to react with a per-compound, such as peracid or hydrogen peroxide, under acid conditions.
- the detergent composition suitably contains from 0.5 per cent by weight and preferably from 0.5 to 3 per cent by weight of the amphoteric compound and/or amine oxide of this.
- the perborate can be present as mono- or tetrahydrate with an alkali metal, such as for example sodium, as positive counter ion. Suitable amounts in the composition is within the range of from 6 to 25 per cent by weight, preferably from 10 to 20 per cent by weight.
- the invention also relates to the use of amphoteric compounds as above defined or amine oxides of these in detergent compositions containing perborates to enhance the bleaching effect at washing with the compositions.
- the compositions contain up to 4.5 per cent by weight, suitably from 0.5 per cent by weight and preferably from 0.5 to 3 per cent by weight of at least one of the defined amphoteric compounds, or an amine oxide of this.
- Suitable amounts of perborates in the compositions are within the range of from 6 to 25 per cent by weight, preferably from 10 to 20 per cent by weight.
- the combination according to the invention of perborate and amphoteric compound, or amine oxide of the amphoteric compound, can be used in otherwise conventional detergent compositions intended at first hand for laundering.
- the detergent compositions are suitably in powder form and can be prepared in a conventional manner, such as by dry blending, agglomeration or spray drying a slurry of the components.
- tensides are also present, preferably anionic and/or nonionic tensides and/or soaps.
- the anionic tensides are suitably present in an amount of from 6 to 20 per cent by weight and can for example comprise linear alkylbenzene sulfonate, secondary alkane sulfonate, alcohol-ethoxy sulfate or alpha-olefin sulfonate.
- the nonionic tensides are suitably present in an amount of from 2 to 8 per cent by weight and can for example comprise alkoxylated compounds, such as fatty alcohols, alkyl phenols and alkyl amines.
- the soaps are suitably present in amounts of from 2 to 4 per cent by weight and can for example comprise sodium ⁇ or potassium salts of tallow.
- the compositions can moreover comprise per se known components such as water glass, enzymes, builders such as phosphates or zeolites, fillers such as sodium sulfate, carbonates and bicarbonates, bleach activators such as TAED (tetraacetylethylene diamine) or TAGU (tetraacetylglucoluril), diperoxy acids, foam regulators, perfumes and colorants.
- a detergent composition containing perborate and amphoteric compounds, or amine oxides can for example contain the following main components up to 100%.
- phosphates 10 to 30 per cent by weight of zeolites and 3 to 10 per cent by weight of polycarboxylates can be used.
- An amine oxide according to the invention was prepared by mixing 100 g of Ampholak 7TX (R) (corresponds to formula (III) above and is a mixture of compounds with different values for y from 1 to 3 and wherein R originates from tallow fatty acid) with 10 g of H2O2 (35%) and heating at 70°C for 1 hour. The mixture was allowed to cool and was then freeze dried.
- R Ampholak 7TX
- R corresponds to formula (III) above and is a mixture of compounds with different values for y from 1 to 3 and wherein R originates from tallow fatty acid
- tests A and B give a corresponding yield of 79% and 94% respectively.
- the better results for the detergent with amine oxide might be due to the fact that part of the hydrogen peroxide in the tests according to A is consumed in the oxidation of nitrogen groups in the amphoteric compound.
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Wood Science & Technology (AREA)
- Organic Chemistry (AREA)
- Inorganic Chemistry (AREA)
- Detergent Compositions (AREA)
Abstract
wherein R is a hydrocarbon group having 7 to 22 carbon atoms, R₁ is hydrogen or alkyl with 1 to 6 carbon atoms, x is 2 or 3, y is 0 or an integer of 1 to 4 and Q is (R₂-COOM) where R₂ is alkylene with 1 to 6 carbon atoms, M is H, an alkali metal, an alkaline earth metal, an ammonium or a substituted ammonium ion, and B is hydrogen or is defined according to Q. Alternatively the composition contains at least one amine oxide of a compound of the above definition. Compounds as defined are used in detergent compositions containing perborate in order to enhance the bleaching effect at washing with the compositions.
Description
- The invention relates to a method for enhancing the bleaching effect at washing in the presence of perborates. The bleaching effect is enhanced by using certain amphoteric compounds, or amine oxides of these, and the invention also relates to the use of such compounds in detergent compositions containing perborates.
- Commercial detergent compositions usually contain perborates which in alkaline aqueous solutions release hydrogen peroxide. This is decomposed into H⁺ and HO₂⁻, which has a bleaching effect at washing. However, the hydrogen peroxide is to a high degree decomposed into free radicals, which do not have a bleaching effect, in the presence of metal ions which are commonly present in the wash water such as calcium, magnesium, copper, iron and manganese. A common method for preventing the decomposition into free radicals is the addition of complexing agents, such as phosphonates and EDTA, to the detergent composition. Despite additions of complexing agents part of the hydrogen peroxide is anyhow decomposed to free radicals at the washing. Several of these complexing agents are also doubtful with regard to environment.
- Amphoteric compounds of the type which can be characterized by the general formula
wherein R is a higher hydrocarbon group, R₁ is hydrogen or a lower alkyl group, x is 2 or 3, y is 0 to 4, Q is a group ―R₂COOM wherein R₂ is an alkylene group and M is hydrogen or another cation and B is hydrogen or a group Q are per se known. Compounds of this type are disclosed in the European patent applications 160507, 162600 and 214868. The compounds are used in detergent and shampoo compositions for surface activity and for their antimicrobial properties. From the European patent application 314648 it is also known to use amphoteric compounds of this type in combination with zeolites in detergent compositions since it has been found that they show a synergistic effect at softening of water. It is also evident from this application that the detergent compositions may contain perborates as bleaching agents. However, the amphoteric compound should be present in fairly high amounts to give the desired effect. Further, it is advised against the use of the amphoteric compounds in detergent compositions containing anionic tensides. - It has now surprisingly been found that certain amphoteric compounds, or amine oxides of these, enhance the bleaching effect at washing in the presence of perborates. The amphoteric compounds and their amine oxides can inhibit the decomposition of hydrogen peroxide to free radicals at washing, which probably depends on their complexing ability.
- The invention thus relates to a process for enhancing the bleaching effect at washing under alkaline conditions in the presence of perborates as defined in the patent claims. The washing is carried out under alkaline conditions using a detergent composition containing perborate and up to 4.5 per cent by weight of at least one amphoteric compound of the general formula (I)
wherein R is a hydrocarbon group having 7 to 22 carbon atoms, R₁ is hydrogen or alkyl with 1 to 6 carbon atoms, x is 2 or 3, y is 0 or an integer of 1 to 4 and Q is (R₂-COOM) where R₂ is alkylene with 1 to 6 carbon atoms, M is H, an alkali metal, an alkaline earth metal, an ammonium or a substituted ammonium ion, and B is hydrogen or is defined according to Q, or a detergent composition containing perborate and up to 4.5 per cent by weight of at least one amine oxide of the defined amphoteric compound. - The composition can thus contain corresponding amounts of at least one amine oxide of amphoteric compound as above defined. One or several of the nitrogen groups in the amphoteric compounds can be oxidized. Useful compounds can be defined by the general formula (II)
wherein R, R₁, x, y, Q, R₂, M and B have the above given definitions. However, the term amine oxide is used to include also compounds wherein only some of the amine groups have been oxidized. The amine oxides of the defined amphoteric compounds are new compounds and can also be used for the same purposes as the previously known amphoteric compounds of formula (I), ie as tensides, antimicrobial agents or take part in softening of water. - In the above given formulae the hydrocarbon group R can be straight or branched, saturated or unsaturated, and may contain substituents such as hydroxyl groups or carbonyl groups. R is preferably an alkyl or alkenyl group, but can also be a cycloalkyl-alkyl group, an aralkyl or aralkenyl group where the alkyl or alkenyl group contains at least 6 carbon atoms. R suitably contains 12 to 22 carbon atoms, preferably 12 to 20 carbon atoms. In particular it is preferred that R is a hydrocarbon group originating from natural fatty acids and particularly from tallow or coco fatty acid. R₁ is hydrogen or a lower alkyl group, suitably with 1 to 6 carbon atoms and preferably hydrogen or a methyl group. x is 2 or 3 and y is suitably 1, 2, 3 or 4 and preferably 1, 2 or 3. The group R₂ is suitably a methylene or ethylene group, preferably a methylene group. M is hydrogen or an ion from the groups alkali metals, alkaline earth metals, ammonium or substituted ammonium such as for example mono-, di- or trihydroxyethylammonium. M is preferably alkali metal, especially sodium. It is particularly preferred that all groups B in the compounds of the above given general formulae are groups Q.
-
- In particular compounds according to formula (III) included in formula (IV) are preferred
wherein y is 1, 2 or 3 and R is as earlier defined or an amine oxide of a compound of this formula. Mixtures of compounds with different values for y can also be used. It is especially preferred that R originates from tallow fatty acid. - Amphoteric compounds which can be used according to the present invention are per se previously known, for example from the earlier mentioned patent applications. They are also commercially available under the trade name Ampholak(R). Amine oxides can be prepared by allowing one of the earlier known amphoteric compounds to react with a per-compound, such as peracid or hydrogen peroxide, under acid conditions. The detergent composition suitably contains from 0.5 per cent by weight and preferably from 0.5 to 3 per cent by weight of the amphoteric compound and/or amine oxide of this.
- The perborate can be present as mono- or tetrahydrate with an alkali metal, such as for example sodium, as positive counter ion. Suitable amounts in the composition is within the range of from 6 to 25 per cent by weight, preferably from 10 to 20 per cent by weight.
- The invention also relates to the use of amphoteric compounds as above defined or amine oxides of these in detergent compositions containing perborates to enhance the bleaching effect at washing with the compositions. The compositions contain up to 4.5 per cent by weight, suitably from 0.5 per cent by weight and preferably from 0.5 to 3 per cent by weight of at least one of the defined amphoteric compounds, or an amine oxide of this. Suitable amounts of perborates in the compositions are within the range of from 6 to 25 per cent by weight, preferably from 10 to 20 per cent by weight.
- The combination according to the invention of perborate and amphoteric compound, or amine oxide of the amphoteric compound, can be used in otherwise conventional detergent compositions intended at first hand for laundering. The detergent compositions are suitably in powder form and can be prepared in a conventional manner, such as by dry blending, agglomeration or spray drying a slurry of the components.
- In the detergent compositions tensides are also present, preferably anionic and/or nonionic tensides and/or soaps. The anionic tensides are suitably present in an amount of from 6 to 20 per cent by weight and can for example comprise linear alkylbenzene sulfonate, secondary alkane sulfonate, alcohol-ethoxy sulfate or alpha-olefin sulfonate. The nonionic tensides are suitably present in an amount of from 2 to 8 per cent by weight and can for example comprise alkoxylated compounds, such as fatty alcohols, alkyl phenols and alkyl amines. The soaps are suitably present in amounts of from 2 to 4 per cent by weight and can for example comprise sodium― or potassium salts of tallow. The compositions can moreover comprise per se known components such as water glass, enzymes, builders such as phosphates or zeolites, fillers such as sodium sulfate, carbonates and bicarbonates, bleach activators such as TAED (tetraacetylethylene diamine) or TAGU (tetraacetylglucoluril), diperoxy acids, foam regulators, perfumes and colorants.
-
- As alternative to the phosphates 10 to 30 per cent by weight of zeolites and 3 to 10 per cent by weight of polycarboxylates can be used.
- The invention is further illustrated in the following examples which, however, are not intended to limit the same. All amounts are given in per cent by weight, unless otherwise stated.
- An amine oxide according to the invention was prepared by mixing 100 g of Ampholak 7TX(R) (corresponds to formula (III) above and is a mixture of compounds with different values for y from 1 to 3 and wherein R originates from tallow fatty acid) with 10 g of H₂O₂ (35%) and heating at 70°C for 1 hour. The mixture was allowed to cool and was then freeze dried.
- In order to investigate the effect of an amphoteric compound according to the invention the following detergent compositions were used;
- A:
- 15% LAS sulfosoft(R) (linear alkylbenzenesulfonate)
- 2% Berol 25 - 3(R) (nonionic ethoxylate)
- 6% Berol 25 - 7(R) (nonionic fatty alcohol ethoxylate)
- 6% disilicate
- 20% sodium perborate tetrahydrate
- 20% sodium tripolyphosphate
- 30% sodium sulphate
- 1% Ampholak 7TX(R)
- B:
- The same formulation as in A, with the exception that the detergent composition contained 1% of the amine oxide of Example 1 instead of Ampholak 7TX.
- C.
- The same formulation as in A, with the exception that the detergent did not contain any amphoteric compound or amine oxide as according to the invention.
- In all tests water with a hardness of 25° dH with regard to Ca/Mg in the ratio 3:1 was used. Further 3 ppm metal ions were added according to the following:
- 2.5 ppm Fe³⁺
- 0.25 ppm Cu²⁺
-
- The tests show that if no compound as according to the invention is used (according to tests C) on an average about 68% of the active oxygen remain after 60 minutes at 60°C. The compositions according to the invention (tests A and B) give a corresponding yield of 79% and 94% respectively. The better results for the detergent with amine oxide might be due to the fact that part of the hydrogen peroxide in the tests according to A is consumed in the oxidation of nitrogen groups in the amphoteric compound.
- Pieces of cloth were immersed in tea and red wine respectively and then allowed to dry. The reflectance was then measured at 460 nm, and then the pieces were washed in a washing machine at 60°C for about 60 minutes in the same type of water as used in Example 2. 10 g detergent, formulated according to Example 2A and 2C respectively, were added per liter of water. The reflectance was then measured again and the difference in relation to the reflectance before the washing was calculated. The results are shown in Table 2.
- The tests show a considerable improvement in the bleaching effect with the detergent composition according to the invention.
Claims (11)
- A process for enhancing the bleaching effect at washing under alkaline conditions in the presence of perborates, characterized in that the washing is carried out using a detergent composition containing perborate and up to 4.5 per cent by weight of at least one amphoteric compound of the general formula (I)
- A process according to any of claims 1 to 3, characterized in that the amphoteric compound is present in an amount of from 0.5 to 3 per cent by weight.
- A process according to any of the preceding claims, characterized in that the perborate is present in an amount of from 6 to 25 per cent by weight.
- Use of up to 4.5 per cent by weight of at least one amphoteric compound of the general formula (I)
- Use according to any of claims 6 to 8, whereby the amphoteric compound is present in an amount of from 0.5 to 3 per cent by weight.
- Use according to any of claims 6 to 9, whereby the detergent compositions contain from 6 to 25 per cent by weight of perborate.
- Use according to any of claims 6 to 11, whereby the detergent compositions contain at least one anionic tenside.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| SE8904177A SE8904177D0 (en) | 1989-12-11 | 1989-12-11 | TVAETTMEDELSKOMPOSITION |
| SE8904177 | 1989-12-11 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0433257A1 true EP0433257A1 (en) | 1991-06-19 |
| EP0433257B1 EP0433257B1 (en) | 1996-03-06 |
Family
ID=20377741
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP91900978A Withdrawn EP0505427A1 (en) | 1989-12-11 | 1990-07-12 | Amine oxides of amphoteric compounds, their preparation and their use |
| EP90850401A Expired - Lifetime EP0433257B1 (en) | 1989-12-11 | 1990-12-07 | A process for enhancing the bleaching effect at washing and use of certain amphoteric compounds in a detergent composition for enhancing the bleaching effect |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP91900978A Withdrawn EP0505427A1 (en) | 1989-12-11 | 1990-07-12 | Amine oxides of amphoteric compounds, their preparation and their use |
Country Status (6)
| Country | Link |
|---|---|
| EP (2) | EP0505427A1 (en) |
| AT (1) | ATE135037T1 (en) |
| DE (1) | DE69025728T2 (en) |
| DK (1) | DK0433257T3 (en) |
| SE (1) | SE8904177D0 (en) |
| WO (1) | WO1991009100A1 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0856577A1 (en) * | 1997-02-03 | 1998-08-05 | The Procter & Gamble Company | Liquid aqueous cleaning compositions |
| WO2017129424A1 (en) * | 2016-01-28 | 2017-08-03 | Henkel Ag & Co. Kgaa | Amphiphilic, carboxyalkylated polyamines as active anticrease ingredient |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8361946B2 (en) | 2004-04-08 | 2013-01-29 | Akzo Nobel N.V. | Detergent composition |
| DE202016101250U1 (en) | 2016-03-08 | 2016-04-27 | Klaus Lokietz | Pet crate |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0160507A2 (en) * | 1984-04-25 | 1985-11-06 | Eric Graham Fishlock-Lomax | Shampoo compositions |
| EP0162600A1 (en) * | 1984-04-25 | 1985-11-27 | Eric Graham Fishlock-Lomax | Cleaning compositions |
| EP0314648A2 (en) * | 1987-10-26 | 1989-05-03 | Berol Nobel Ab | Detergent compositions |
| EP0325124A2 (en) * | 1988-01-21 | 1989-07-26 | Colgate-Palmolive Company | Sugar ethers as bleach stable detergency boosters |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3712918A (en) * | 1972-05-31 | 1973-01-23 | Millmaster Onyx Corp | Amphoteric surface-active agents |
| GB8522413D0 (en) * | 1985-09-10 | 1985-10-16 | Amphoterics International Ltd | Surfactants |
-
1989
- 1989-12-11 SE SE8904177A patent/SE8904177D0/en unknown
-
1990
- 1990-07-12 WO PCT/SE1990/000813 patent/WO1991009100A1/en not_active Application Discontinuation
- 1990-07-12 EP EP91900978A patent/EP0505427A1/en not_active Withdrawn
- 1990-12-07 AT AT90850401T patent/ATE135037T1/en not_active IP Right Cessation
- 1990-12-07 EP EP90850401A patent/EP0433257B1/en not_active Expired - Lifetime
- 1990-12-07 DE DE69025728T patent/DE69025728T2/en not_active Expired - Fee Related
- 1990-12-07 DK DK90850401.2T patent/DK0433257T3/en active
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0160507A2 (en) * | 1984-04-25 | 1985-11-06 | Eric Graham Fishlock-Lomax | Shampoo compositions |
| EP0162600A1 (en) * | 1984-04-25 | 1985-11-27 | Eric Graham Fishlock-Lomax | Cleaning compositions |
| EP0314648A2 (en) * | 1987-10-26 | 1989-05-03 | Berol Nobel Ab | Detergent compositions |
| EP0325124A2 (en) * | 1988-01-21 | 1989-07-26 | Colgate-Palmolive Company | Sugar ethers as bleach stable detergency boosters |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0856577A1 (en) * | 1997-02-03 | 1998-08-05 | The Procter & Gamble Company | Liquid aqueous cleaning compositions |
| WO2017129424A1 (en) * | 2016-01-28 | 2017-08-03 | Henkel Ag & Co. Kgaa | Amphiphilic, carboxyalkylated polyamines as active anticrease ingredient |
Also Published As
| Publication number | Publication date |
|---|---|
| EP0433257B1 (en) | 1996-03-06 |
| DK0433257T3 (en) | 1996-06-17 |
| DE69025728D1 (en) | 1996-04-11 |
| DE69025728T2 (en) | 1996-07-25 |
| WO1991009100A1 (en) | 1991-06-27 |
| ATE135037T1 (en) | 1996-03-15 |
| EP0505427A1 (en) | 1992-09-30 |
| SE8904177D0 (en) | 1989-12-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0086511B1 (en) | Oxygen-bleach-containing liquid detergent compositions | |
| EP0293040B1 (en) | Liquid detergent containing solid peroxygen bleach | |
| US3001945A (en) | Liquid detergent composition | |
| EP0111963B1 (en) | Detergent compositions | |
| US4119557A (en) | Bleaching compositions and process for cleaning fabrics | |
| AU662501B2 (en) | Liquid detergent compositions containing a suspended peroxygen bleach | |
| CA2010036C (en) | Stabilized bleach containing liquid detergent compositions | |
| AU624328B2 (en) | Liquid detergent containing perborate bleach | |
| EP0145090A2 (en) | Detergent bleach compositions | |
| NZ202823A (en) | Bleach composition | |
| DE69004809T2 (en) | Liquid detergent containing enzyme and enzyme stabilization system. | |
| US5250212A (en) | Liquid detergent containing solid peroxygen bleach and solvent system comprising water and lower aliphatic monoalcohol | |
| WO1995033816A1 (en) | Aqueous bleaching compositions comprising peroxy carboxylic acids | |
| JPS6126958B2 (en) | ||
| PL179665B1 (en) | Whitening agents | |
| US4163732A (en) | Detergent composition containing water-insoluble phosphorus-containing aluminosilicate builders | |
| EP0433257B1 (en) | A process for enhancing the bleaching effect at washing and use of certain amphoteric compounds in a detergent composition for enhancing the bleaching effect | |
| CA1207956A (en) | Peroxyacid bleaching and laundering composition | |
| JPS636100A (en) | Softening bleaching detergent composition containing amide softener | |
| US3991000A (en) | Built bleaching detergent | |
| JPH0662992B2 (en) | Detergent composition | |
| EP0182411A2 (en) | Detergent compositions containing polymers | |
| WO1991009103A1 (en) | Concentrated aqueous liquid bleach compositions | |
| EP0463801A2 (en) | Composition and method for fabric encrustation prevention | |
| JP3246843B2 (en) | Nonionic powder detergent composition |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FR GB GR IT LI LU NL SE |
|
| 17P | Request for examination filed |
Effective date: 19911028 |
|
| 17Q | First examination report despatched |
Effective date: 19940919 |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: AKZO NOBEL SURFACE CHEMISTRY AKTIEBOLAG |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE CH DE DK ES FR GB GR IT LI LU NL SE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 19960306 Ref country code: ES Free format text: THE PATENT HAS BEEN ANNULLED BY A DECISION OF A NATIONAL AUTHORITY Effective date: 19960306 |
|
| REF | Corresponds to: |
Ref document number: 135037 Country of ref document: AT Date of ref document: 19960315 Kind code of ref document: T |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: NV Representative=s name: E. BLUM & CO. PATENTANWAELTE |
|
| REF | Corresponds to: |
Ref document number: 69025728 Country of ref document: DE Date of ref document: 19960411 |
|
| ET | Fr: translation filed | ||
| ITF | It: translation for a ep patent filed | ||
| REG | Reference to a national code |
Ref country code: DK Ref legal event code: T3 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Effective date: 19961207 Ref country code: DK Effective date: 19961207 Ref country code: AT Effective date: 19961207 |
|
| REG | Reference to a national code |
Ref country code: DK Ref legal event code: EBP |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Effective date: 19961208 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19961231 Ref country code: LI Effective date: 19961231 Ref country code: CH Effective date: 19961231 Ref country code: BE Effective date: 19961231 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| BERE | Be: lapsed |
Owner name: AKZO NOBEL SURFACE CHEMISTRY A.B. Effective date: 19961231 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Effective date: 19970701 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 19961207 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Effective date: 19970829 |
|
| NLV4 | Nl: lapsed or anulled due to non-payment of the annual fee |
Effective date: 19970701 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Effective date: 19970902 |
|
| EUG | Se: european patent has lapsed |
Ref document number: 90850401.2 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES;WARNING: LAPSES OF ITALIAN PATENTS WITH EFFECTIVE DATE BEFORE 2007 MAY HAVE OCCURRED AT ANY TIME BEFORE 2007. THE CORRECT EFFECTIVE DATE MAY BE DIFFERENT FROM THE ONE RECORDED. Effective date: 20051207 |