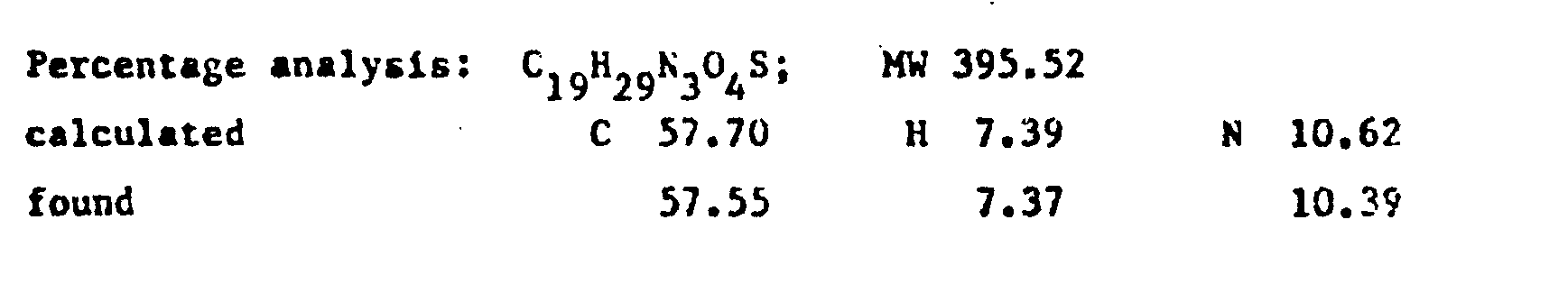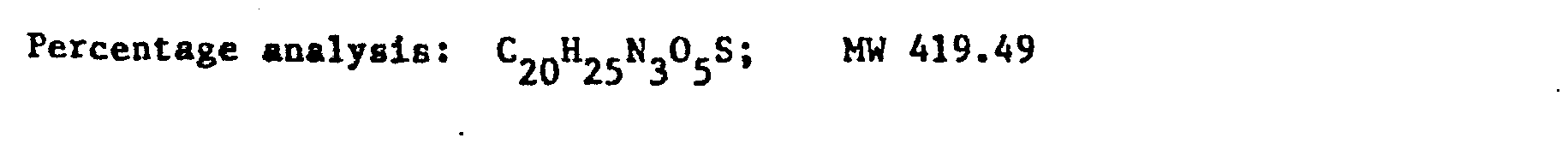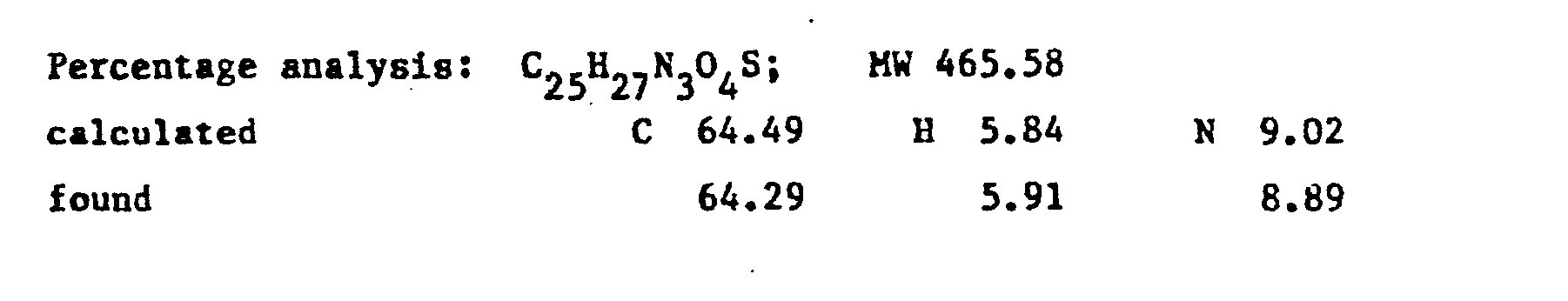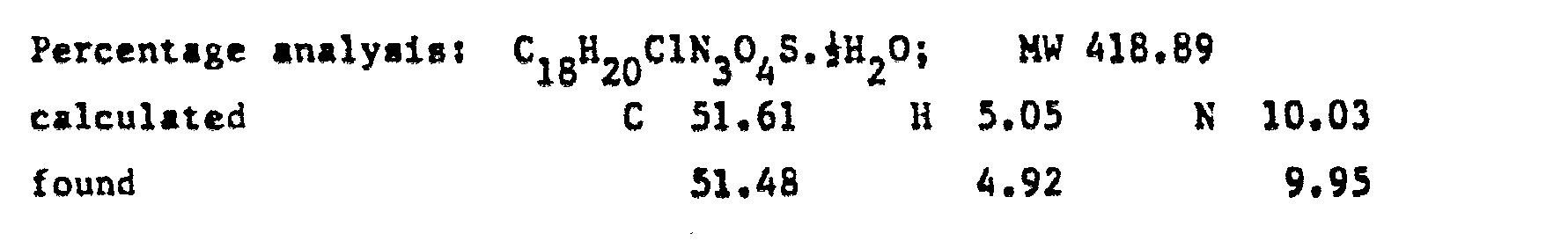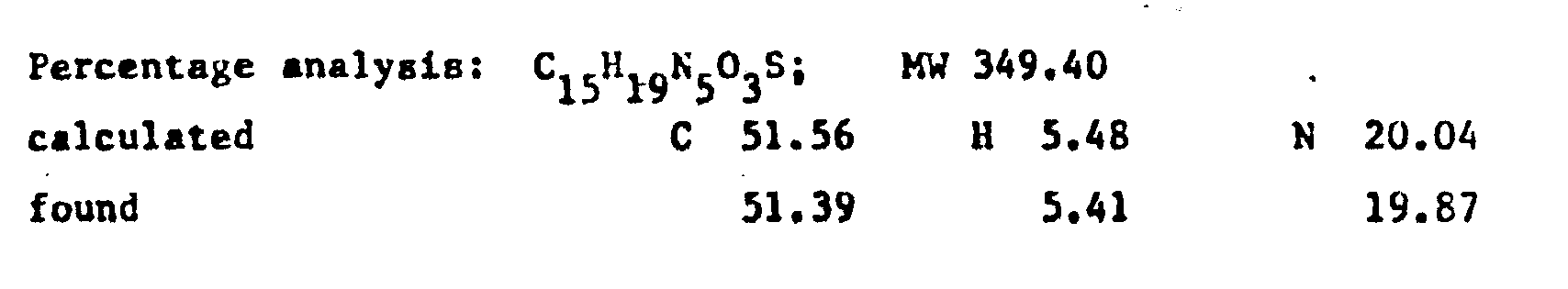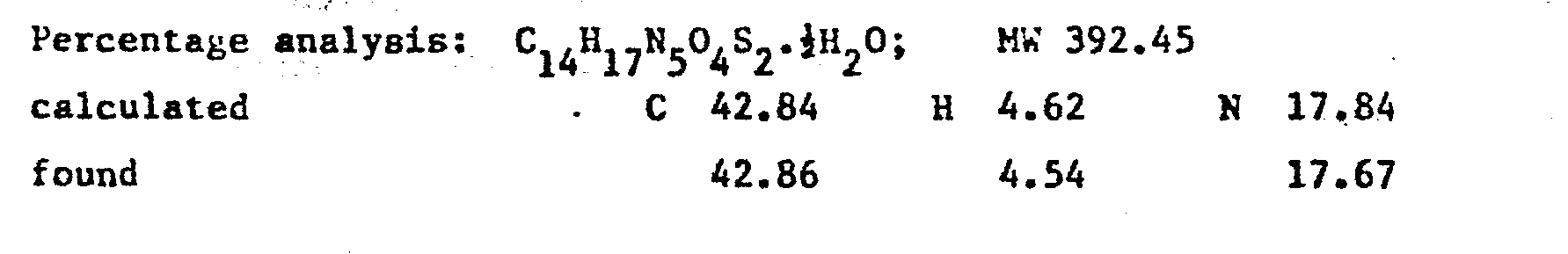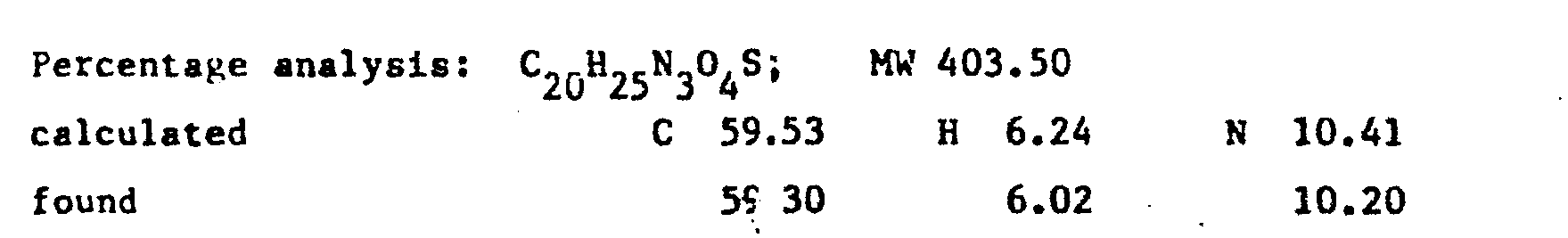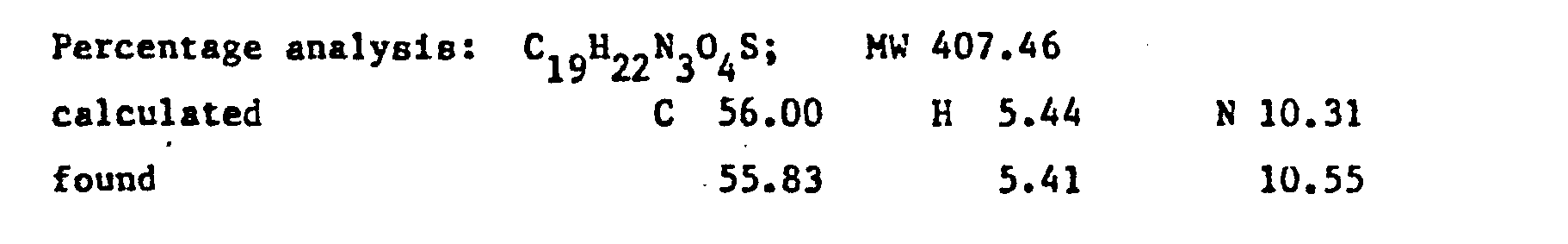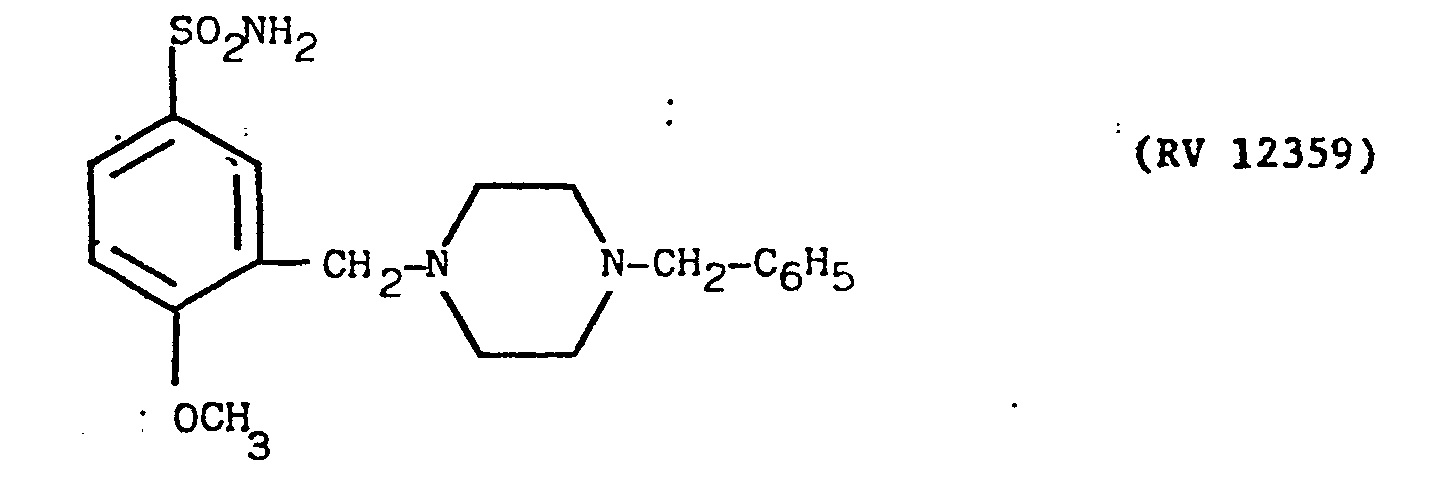EP0171636A1 - Pharmacologically active piperazino derivatives and the process for their preparation - Google Patents
Pharmacologically active piperazino derivatives and the process for their preparation Download PDFInfo
- Publication number
- EP0171636A1 EP0171636A1 EP85108942A EP85108942A EP0171636A1 EP 0171636 A1 EP0171636 A1 EP 0171636A1 EP 85108942 A EP85108942 A EP 85108942A EP 85108942 A EP85108942 A EP 85108942A EP 0171636 A1 EP0171636 A1 EP 0171636A1
- Authority
- EP
- European Patent Office
- Prior art keywords
- alkyl
- group
- alkenyl
- formula
- phenyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
- -1 piperazino Chemical class 0.000 title claims abstract description 25
- 238000000034 method Methods 0.000 title claims description 13
- 238000002360 preparation method Methods 0.000 title description 26
- 150000001875 compounds Chemical class 0.000 claims abstract description 39
- 238000006243 chemical reaction Methods 0.000 claims abstract description 13
- CIUQDSCDWFSTQR-UHFFFAOYSA-N [C]1=CC=CC=C1 Chemical group [C]1=CC=CC=C1 CIUQDSCDWFSTQR-UHFFFAOYSA-N 0.000 claims abstract description 7
- 229910052736 halogen Inorganic materials 0.000 claims abstract description 7
- 229910052757 nitrogen Inorganic materials 0.000 claims abstract description 5
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims abstract description 5
- 150000002367 halogens Chemical class 0.000 claims abstract description 4
- 125000006239 protecting group Chemical group 0.000 claims abstract description 4
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims abstract description 3
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims abstract description 3
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims abstract 6
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 claims description 44
- 150000001412 amines Chemical class 0.000 claims description 37
- FAIAAWCVCHQXDN-UHFFFAOYSA-N phosphorus trichloride Chemical compound ClP(Cl)Cl FAIAAWCVCHQXDN-UHFFFAOYSA-N 0.000 claims description 23
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 claims description 22
- 239000002253 acid Substances 0.000 claims description 21
- 239000002904 solvent Substances 0.000 claims description 18
- GLUUGHFHXGJENI-UHFFFAOYSA-N Piperazine Chemical compound C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 claims description 8
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 claims description 7
- 125000000623 heterocyclic group Chemical group 0.000 claims description 6
- 125000001424 substituent group Chemical group 0.000 claims description 6
- 125000004178 (C1-C4) alkyl group Chemical group 0.000 claims description 5
- 125000000217 alkyl group Chemical group 0.000 claims description 5
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 claims description 4
- 125000002252 acyl group Chemical group 0.000 claims description 4
- 125000003118 aryl group Chemical group 0.000 claims description 4
- 125000005843 halogen group Chemical group 0.000 claims description 3
- YOGFGFKRNRQDMF-UHFFFAOYSA-N n-(1,3,4-thiadiazol-2-yl)acetamide Chemical group CC(=O)NC1=NN=CS1 YOGFGFKRNRQDMF-UHFFFAOYSA-N 0.000 claims description 3
- 125000000229 (C1-C4)alkoxy group Chemical group 0.000 claims description 2
- 125000006727 (C1-C6) alkenyl group Chemical group 0.000 claims description 2
- 125000004191 (C1-C6) alkoxy group Chemical group 0.000 claims description 2
- GOJUJUVQIVIZAV-UHFFFAOYSA-N 2-amino-4,6-dichloropyrimidine-5-carbaldehyde Chemical group NC1=NC(Cl)=C(C=O)C(Cl)=N1 GOJUJUVQIVIZAV-UHFFFAOYSA-N 0.000 claims description 2
- 125000005024 alkenyl aryl group Chemical group 0.000 claims description 2
- 125000003342 alkenyl group Chemical group 0.000 claims description 2
- 125000005257 alkyl acyl group Chemical group 0.000 claims description 2
- 125000005741 alkyl alkenyl group Chemical group 0.000 claims description 2
- 125000002877 alkyl aryl group Chemical group 0.000 claims description 2
- 125000005119 alkyl cycloalkyl group Chemical group 0.000 claims description 2
- 150000005215 alkyl ethers Chemical class 0.000 claims description 2
- 125000005037 alkyl phenyl group Chemical group 0.000 claims description 2
- 125000003368 amide group Chemical group 0.000 claims description 2
- 239000003795 chemical substances by application Substances 0.000 claims description 2
- 229910052801 chlorine Inorganic materials 0.000 claims description 2
- 229910052739 hydrogen Inorganic materials 0.000 claims description 2
- 239000001257 hydrogen Substances 0.000 claims description 2
- 229910052740 iodine Inorganic materials 0.000 claims description 2
- 125000004433 nitrogen atom Chemical group N* 0.000 claims description 2
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims description 2
- 125000004953 trihalomethyl group Chemical group 0.000 claims description 2
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims 1
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 claims 1
- 125000001931 aliphatic group Chemical group 0.000 claims 1
- 229910052794 bromium Inorganic materials 0.000 claims 1
- 239000003054 catalyst Substances 0.000 claims 1
- 238000007796 conventional method Methods 0.000 claims 1
- 230000008030 elimination Effects 0.000 claims 1
- 238000003379 elimination reaction Methods 0.000 claims 1
- 238000005984 hydrogenation reaction Methods 0.000 claims 1
- 150000007529 inorganic bases Chemical class 0.000 claims 1
- 238000004519 manufacturing process Methods 0.000 claims 1
- 150000007530 organic bases Chemical class 0.000 claims 1
- 239000003960 organic solvent Substances 0.000 claims 1
- 102000003846 Carbonic anhydrases Human genes 0.000 abstract description 3
- 108090000209 Carbonic anhydrases Proteins 0.000 abstract description 3
- 230000002401 inhibitory effect Effects 0.000 abstract description 2
- 239000000243 solution Substances 0.000 description 78
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 72
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 51
- 239000000203 mixture Substances 0.000 description 41
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 39
- 238000013019 agitation Methods 0.000 description 39
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 22
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 20
- 238000010992 reflux Methods 0.000 description 20
- 239000002244 precipitate Substances 0.000 description 19
- 239000011541 reaction mixture Substances 0.000 description 19
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 18
- 229910052799 carbon Inorganic materials 0.000 description 18
- XVMSFILGAMDHEY-UHFFFAOYSA-N 6-(4-aminophenyl)sulfonylpyridin-3-amine Chemical compound C1=CC(N)=CC=C1S(=O)(=O)C1=CC=C(N)C=N1 XVMSFILGAMDHEY-UHFFFAOYSA-N 0.000 description 17
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 17
- SQAILWDRVDGLGY-UHFFFAOYSA-N 2-methoxy-5-sulfamoylbenzoic acid Chemical compound COC1=CC=C(S(N)(=O)=O)C=C1C(O)=O SQAILWDRVDGLGY-UHFFFAOYSA-N 0.000 description 16
- 235000019441 ethanol Nutrition 0.000 description 14
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 12
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 12
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 9
- 239000000047 product Substances 0.000 description 9
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 8
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 6
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 6
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 5
- 229910017974 NH40H Inorganic materials 0.000 description 4
- 238000001816 cooling Methods 0.000 description 4
- 238000002425 crystallisation Methods 0.000 description 4
- VUNXBQRNMNVUMV-UHFFFAOYSA-N phenyl(piperazin-1-yl)methanone Chemical compound C=1C=CC=CC=1C(=O)N1CCNCC1 VUNXBQRNMNVUMV-UHFFFAOYSA-N 0.000 description 4
- 0 **N1CC(*)N(*)CC1 Chemical compound **N1CC(*)N(*)CC1 0.000 description 3
- IQXXEPZFOOTTBA-UHFFFAOYSA-N 1-benzylpiperazine Chemical compound C=1C=CC=CC=1CN1CCNCC1 IQXXEPZFOOTTBA-UHFFFAOYSA-N 0.000 description 3
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- 229910021529 ammonia Inorganic materials 0.000 description 3
- NBOOZXVYXHATOW-UHFFFAOYSA-N 1-(1,3-benzodioxol-5-ylmethyl)piperazine Chemical compound C=1C=C2OCOC2=CC=1CN1CCNCC1 NBOOZXVYXHATOW-UHFFFAOYSA-N 0.000 description 2
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 2
- BMDFKRNKFIWLGH-UHFFFAOYSA-N [N]N1CCNCC1 Chemical compound [N]N1CCNCC1 BMDFKRNKFIWLGH-UHFFFAOYSA-N 0.000 description 2
- 229940073608 benzyl chloride Drugs 0.000 description 2
- SSOLNOMRVKKSON-UHFFFAOYSA-N proguanil Chemical compound CC(C)\N=C(/N)N=C(N)NC1=CC=C(Cl)C=C1 SSOLNOMRVKKSON-UHFFFAOYSA-N 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- KOHZQJWOAVVSSL-UHFFFAOYSA-N (1-methylpiperazin-2-yl)-pyrrolidin-1-ylmethanone Chemical compound CN1CCNCC1C(=O)N1CCCC1 KOHZQJWOAVVSSL-UHFFFAOYSA-N 0.000 description 1
- YBNZVBUWVIIJND-UHFFFAOYSA-N (2z)-1-cyclononyl-2-diazocyclononane Chemical compound [N-]=[N+]=C1CCCCCCCC1C1CCCCCCCC1 YBNZVBUWVIIJND-UHFFFAOYSA-N 0.000 description 1
- AVJKDKWRVSSJPK-UHFFFAOYSA-N 1-(4-fluorophenyl)piperazine Chemical compound C1=CC(F)=CC=C1N1CCNCC1 AVJKDKWRVSSJPK-UHFFFAOYSA-N 0.000 description 1
- LZBOHNCMCCSTJX-UHFFFAOYSA-N 1-(chloromethyl)-3-methylbenzene Chemical compound CC1=CC=CC(CCl)=C1 LZBOHNCMCCSTJX-UHFFFAOYSA-N 0.000 description 1
- LRPGNFROBDUREU-UHFFFAOYSA-N 1-(cyclohexylmethyl)piperazine Chemical compound C1CNCCN1CC1CCCCC1 LRPGNFROBDUREU-UHFFFAOYSA-N 0.000 description 1
- BYWNAISVQHRFEE-UHFFFAOYSA-N 1-[(3-methoxyphenyl)methyl]piperazine Chemical compound COC1=CC=CC(CN2CCNCC2)=C1 BYWNAISVQHRFEE-UHFFFAOYSA-N 0.000 description 1
- MGLUVVBFISROAH-UHFFFAOYSA-N 1-[(4-methoxyphenyl)methyl]piperazine Chemical compound C1=CC(OC)=CC=C1CN1CCNCC1 MGLUVVBFISROAH-UHFFFAOYSA-N 0.000 description 1
- WGEIOMTZIIOUMA-QPJJXVBHSA-N 1-[(e)-3-phenylprop-2-enyl]piperazine Chemical compound C1CNCCN1C\C=C\C1=CC=CC=C1 WGEIOMTZIIOUMA-QPJJXVBHSA-N 0.000 description 1
- XCJPYYOSESMDSP-UHFFFAOYSA-N 1-methyl-2-propan-2-ylpiperazine-2-carboxamide Chemical compound CC(C)C1(C(N)=O)CNCCN1C XCJPYYOSESMDSP-UHFFFAOYSA-N 0.000 description 1
- ZWAQJGHGPPDZSF-UHFFFAOYSA-N 1-prop-2-enylpiperazine Chemical compound C=CCN1CCNCC1 ZWAQJGHGPPDZSF-UHFFFAOYSA-N 0.000 description 1
- XRQNHQCPCJANBN-UHFFFAOYSA-N 2-sulfamoyl-1h-imidazole-5-carboxylic acid Chemical compound NS(=O)(=O)C1=NC(C(O)=O)=CN1 XRQNHQCPCJANBN-UHFFFAOYSA-N 0.000 description 1
- SDERAVMLJNQCLJ-UHFFFAOYSA-N 4-methoxy-3-(piperazine-1-carbonyl)benzenesulfonamide Chemical compound COC1=CC=C(S(N)(=O)=O)C=C1C(=O)N1CCNCC1 SDERAVMLJNQCLJ-UHFFFAOYSA-N 0.000 description 1
- XXRUQNNAKXZSOS-UHFFFAOYSA-N 5-(chloromethyl)-1,2,3-trimethoxybenzene Chemical compound COC1=CC(CCl)=CC(OC)=C1OC XXRUQNNAKXZSOS-UHFFFAOYSA-N 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 208000037273 Pathologic Processes Diseases 0.000 description 1
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 1
- WETWJCDKMRHUPV-UHFFFAOYSA-N acetyl chloride Chemical compound CC(Cl)=O WETWJCDKMRHUPV-UHFFFAOYSA-N 0.000 description 1
- 239000012346 acetyl chloride Substances 0.000 description 1
- 230000001476 alcoholic effect Effects 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 238000009903 catalytic hydrogenation reaction Methods 0.000 description 1
- 238000010531 catalytic reduction reaction Methods 0.000 description 1
- 230000007812 deficiency Effects 0.000 description 1
- NWVNXDKZIQLBNM-UHFFFAOYSA-N diphenylmethylpiperazine Chemical compound C1CNCCN1C(C=1C=CC=CC=1)C1=CC=CC=C1 NWVNXDKZIQLBNM-UHFFFAOYSA-N 0.000 description 1
- GCDDETLSKIAHHR-UHFFFAOYSA-N ethyl 1-methylpiperazine-2-carboxylate Chemical compound CCOC(=O)C1CNCCN1C GCDDETLSKIAHHR-UHFFFAOYSA-N 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 1
- 230000005764 inhibitory process Effects 0.000 description 1
- YMWUJEATGCHHMB-UHFFFAOYSA-N methylene chloride Substances ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 1
- 229940073584 methylene chloride Drugs 0.000 description 1
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 1
- 239000012074 organic phase Substances 0.000 description 1
- 230000009054 pathological process Effects 0.000 description 1
- 230000035790 physiological processes and functions Effects 0.000 description 1
- 239000000741 silica gel Substances 0.000 description 1
- 229910002027 silica gel Inorganic materials 0.000 description 1
- 229910000029 sodium carbonate Inorganic materials 0.000 description 1
- 230000001225 therapeutic effect Effects 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D285/00—Heterocyclic compounds containing rings having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by groups C07D275/00 - C07D283/00
- C07D285/01—Five-membered rings
- C07D285/02—Thiadiazoles; Hydrogenated thiadiazoles
- C07D285/04—Thiadiazoles; Hydrogenated thiadiazoles not condensed with other rings
- C07D285/12—1,3,4-Thiadiazoles; Hydrogenated 1,3,4-thiadiazoles
- C07D285/125—1,3,4-Thiadiazoles; Hydrogenated 1,3,4-thiadiazoles with oxygen, sulfur or nitrogen atoms, directly attached to ring carbon atoms, the nitrogen atoms not forming part of a nitro radical
- C07D285/135—Nitrogen atoms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D233/00—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings
- C07D233/04—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member
- C07D233/28—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D233/30—Oxygen or sulfur atoms
- C07D233/42—Sulfur atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D295/00—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms
- C07D295/16—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms acylated on ring nitrogen atoms
- C07D295/18—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms acylated on ring nitrogen atoms by radicals derived from carboxylic acids, or sulfur or nitrogen analogues thereof
- C07D295/182—Radicals derived from carboxylic acids
- C07D295/192—Radicals derived from carboxylic acids from aromatic carboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D317/00—Heterocyclic compounds containing five-membered rings having two oxygen atoms as the only ring hetero atoms
- C07D317/08—Heterocyclic compounds containing five-membered rings having two oxygen atoms as the only ring hetero atoms having the hetero atoms in positions 1 and 3
- C07D317/44—Heterocyclic compounds containing five-membered rings having two oxygen atoms as the only ring hetero atoms having the hetero atoms in positions 1 and 3 ortho- or peri-condensed with carbocyclic rings or ring systems
- C07D317/46—Heterocyclic compounds containing five-membered rings having two oxygen atoms as the only ring hetero atoms having the hetero atoms in positions 1 and 3 ortho- or peri-condensed with carbocyclic rings or ring systems condensed with one six-membered ring
- C07D317/48—Methylenedioxybenzenes or hydrogenated methylenedioxybenzenes, unsubstituted on the hetero ring
- C07D317/50—Methylenedioxybenzenes or hydrogenated methylenedioxybenzenes, unsubstituted on the hetero ring with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to atoms of the carbocyclic ring
- C07D317/58—Radicals substituted by nitrogen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/04—Ortho-condensed systems
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P20/00—Technologies relating to chemical industry
- Y02P20/50—Improvements relating to the production of bulk chemicals
- Y02P20/55—Design of synthesis routes, e.g. reducing the use of auxiliary or protecting groups
Definitions
- This invention relates to a new class of pharmacologically active piperazino derivatives, and the process for their preparation.
- the invention relates to a new class of piperazino derivatives possessing inhibiting activity towards carbonic anhydrase.
- Carbonic anhydrase is known to be one of the most widespread enzymes in human tissues, and plays a determining role in many physiological and pathological processes.
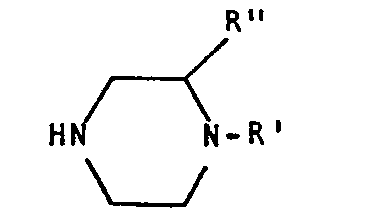
- R is a phenyl radical containing at least one sulphonamido group and possibly various substituents chosen from the group consisting of halogen, C 1 -C 4 alkyl, C 1 -C 4 alkoxy; or a nitrogenated heterocyclic ring containing as substituent at least one amino, amido or sulphonamido group;
- A is CU, CH 2 , SO 2 ;
- k' is H, alkyl, alkenyl, alkyl-aryl, alkyl-cycloalkyl, alkyl-acyl, alkyl-carboxyalkoxy, alkyl-carboxyamino, alkenyl-aryl, alkenyl- cycloalkyl, alkenyl-acyl, alkenyl-carboxyalkoxy, alkenyl-carboxyamino, acyl, aryl;
- R" is H or together with R' and with the nitrogen
- R is 2-methoxy-5-sulphamoyl-phenyl, 2-sulphamoyl-imidazyl, 2-amino or 2-acetamido-1,3,4-thiadiazole;
- R' is H, linear or branched C 1 -C 6 alkyl, linear or branched C 1 -C 6 alkenyl, alkyl-phenyl or alkenyl-phenyl in which the phenyl group is either simple or substituted with halogens, trihalomethyls, C 1 -C 6 alkyls, C 1 -C 6 alkoxys, C 1 -C 6 alkylethers, sulphonamido groups; C 2 -C 6 aliphatic acyl; alkyl-acyl radical in which the acyl is C 1 -C 6 aliphatic, aromatic or nitrogenated heterocyclic; C 3 -C 6 alkyl-carboxyalkoxy; alkyl
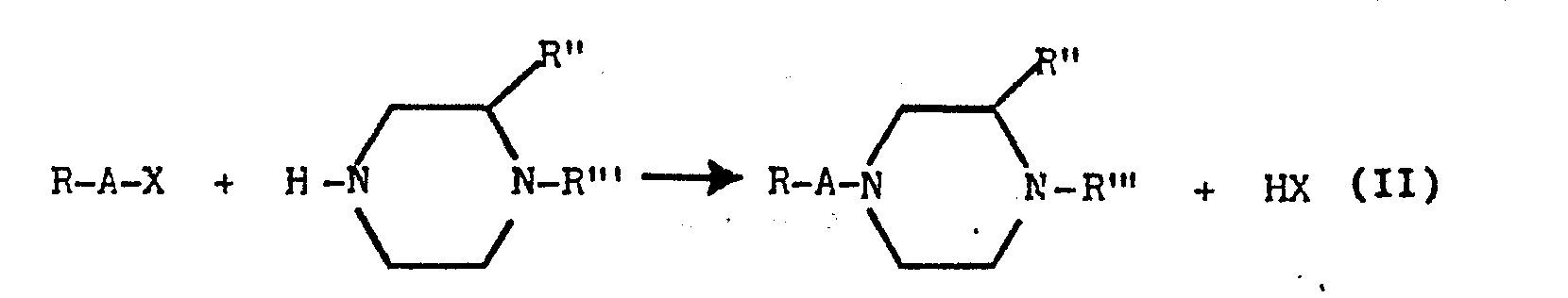
- the new products according to the present invention are prepared essentially in accordance with the following reaction: where R, R', R" are as heretofore defined, X is OH, SH, Cl, br, I, OR"", SR"" in which R"" is methyl, ethyl, phenyl, carbomethoxy, carboethoxy, in the presence of a base or a condensing agent.
- R' substituents can be introduced to the piperazino nitrogen either before or after reaction (II) is effected, using known methods comprising reacting the piperazino nitrogen bonded to the hydrogen with the required compound containing a reactive group.
- reaction (II) is effected beforehand using benzyl-piperazine
- the benzyl derivative thus obtained is debenzylated by catalytic hydrogenation with Pd on C, and the compound obtained . is reacted with a suitable compound YR', in which Y is a reactive group, preferably halogen.
- the R'-R" cyclisation can also be effected before or after effecting reaction (II).
- Reaction (II) is preferably effected by reacting, with the compound R-A-OH and PCl 3 or POC1 5 in a basic medium, the piperazino derivative containing the group R' or with one of the nitrogens suitably protected.
- the amine:R-A-OH:PC1 3 ratio is generally stoichiometric, or has a slight deficiency (10-20X) of the compound R-A-OH relative to the stoichiometric.
- the basic medium is preferably pyridine, or any organic basic solvent able to block the HC1 which forms during the reaction.
- the reaction takes place under hot conditions, preferably at the mixture reflux temperature.
- the product obtained is separated and purified by normal methods.
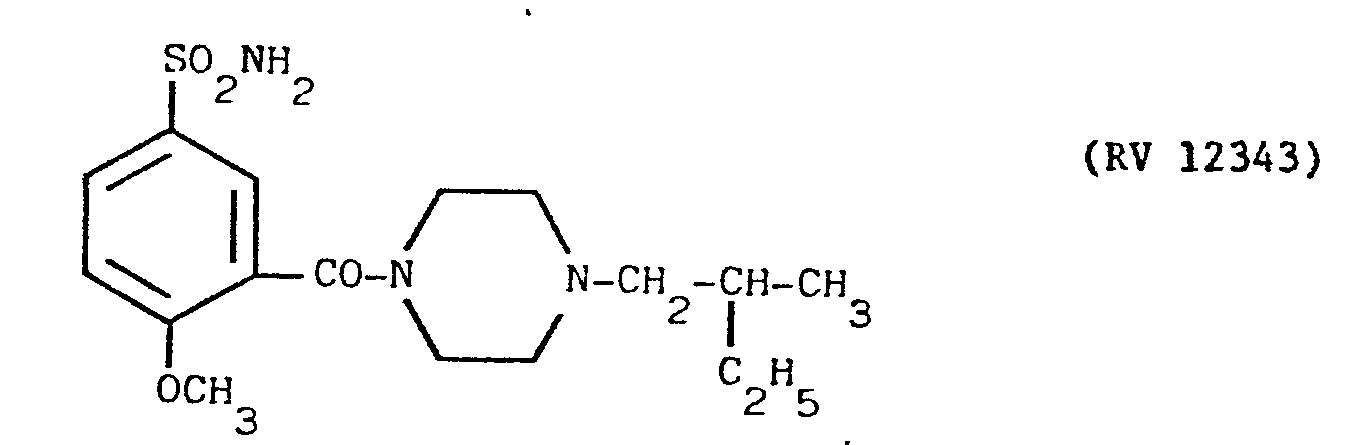
- a solution of N(2-methyl-l-butyl)piperazine in pyridine with an amine:pyridine weight ratio of 1:10 was fed into a suitable reactor, and a cold solution of PC1 3 in pyridine in a weight ratio of 1:10 was dripped in.
- the mixture was kept under agitation for one hour at ambient temperature, after which 2-methoxy-5-sulphamoyl-benzoic acid was added, and the mixture heated under reflux for 4 hours.
- reaction mixture was evaporated under vacuum, taken up in dilute 10% sodium hydroxide solution, washed with ether, acidified with acetic acid and filtered over carbon.
- the clear solution was alkalised with concentrated ammonia under agitation.
- the precipitate was filtered off after some hours and washed with water.
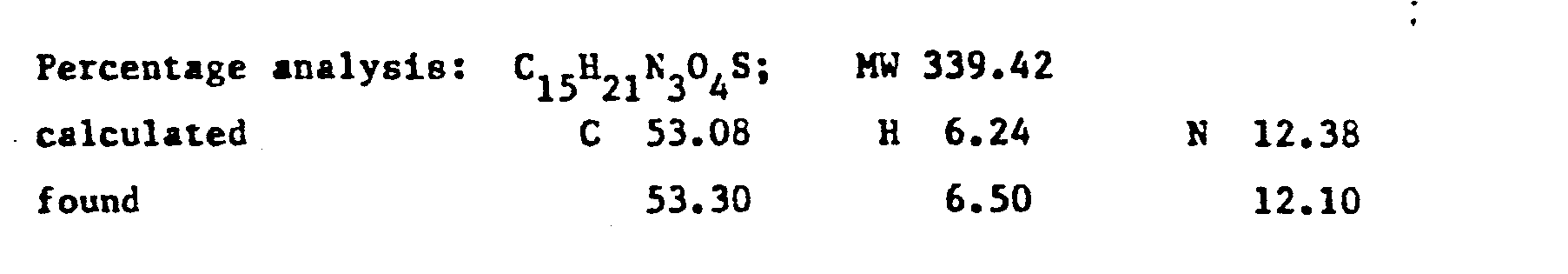
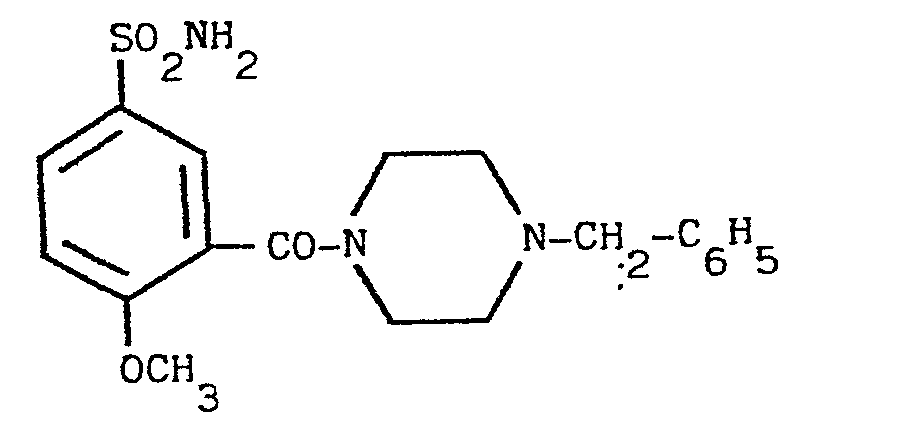
- the product RV 12343 in the form of the hydrochloride was purified by repeated crystallisations from n.butanol. Yield 65X with respect to the acid.
- a solution of N-allyl-piperazine in pyridine with an amine:solvent weight ratio of 1:15 was fed into the reactor, and a cold solution of PC1 3 in pyridine in a weight ratio of 1:10 was dripped in.
- the mixture was kept under agitation for one hour at ambient temperature, after which 2-methoxy-5-sulphamoyl-benzoic acid was added, and the mixture heated under reflux for 4 hours.
- reaction mixture was evaporated to dryness under vacuum, taken up in dilute 20% sodium hydroxide solution, washed with chloroform, acidified with acetic acid and filtered over carbon.
- the clear solution was alkalised with IN NaOH and left for some hours under agitation.
- a solution of diazobicyclononane in pyridine with an amine:solvent weight ratio of 1:10 was fed into the reactor, and a cold solution of PC1 3 in pyridine in a weight ratio of 1:10 was dripped in.
- reaction mixture was evaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washed with ether, acidified with acetic acid and filtered over carbon.
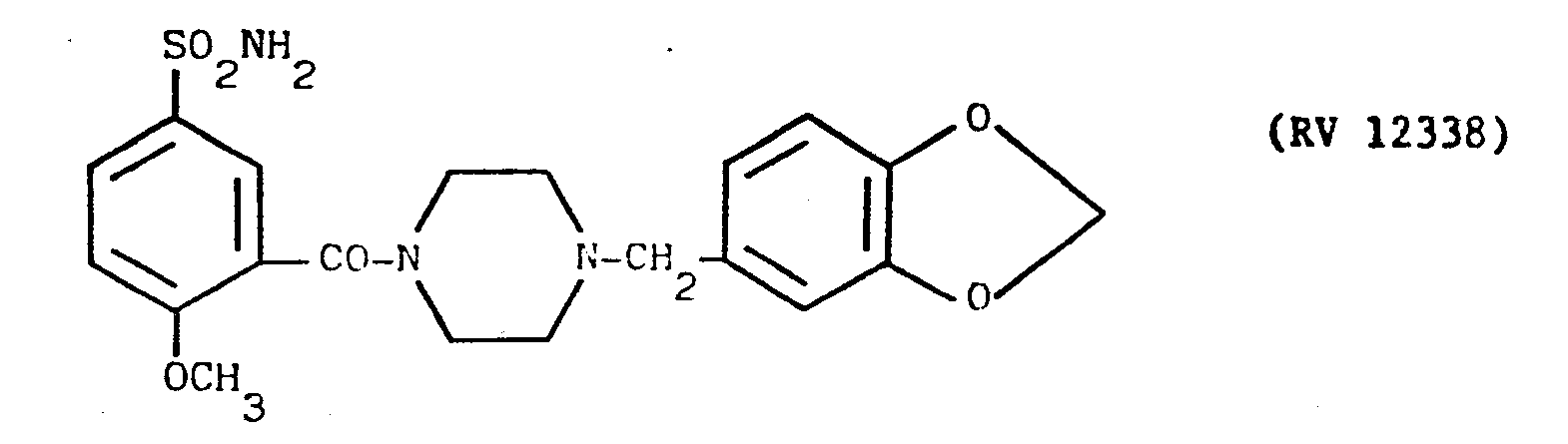
- a solution of piperonylpiperazine in pyridine with an amine:solvent weight ratio of 1:12 was fed into the reactor, and a cold solution of PCl 3 in pyridine in a weight ratio of 1:10 was dripped in.
- reaction mixture was evaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washed with CC1 4 , acidified with dilute hydrochloric acid and filtered over carbon.
- the clear solution was alkalised with concentrated NH 4 OH under agitation.
- a solution of 1-cyclohexyl-methyl-piperazine in pyridine with an amine:solvent weight ratio of 1:10 was fed into the reactor, and a cold solution of PC1 3 in pyridine in a weight ratio of 1:10 was dripped in.
- reaction mixture was evaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washed with ether, acidified with acetic acid and filtered over carbon.
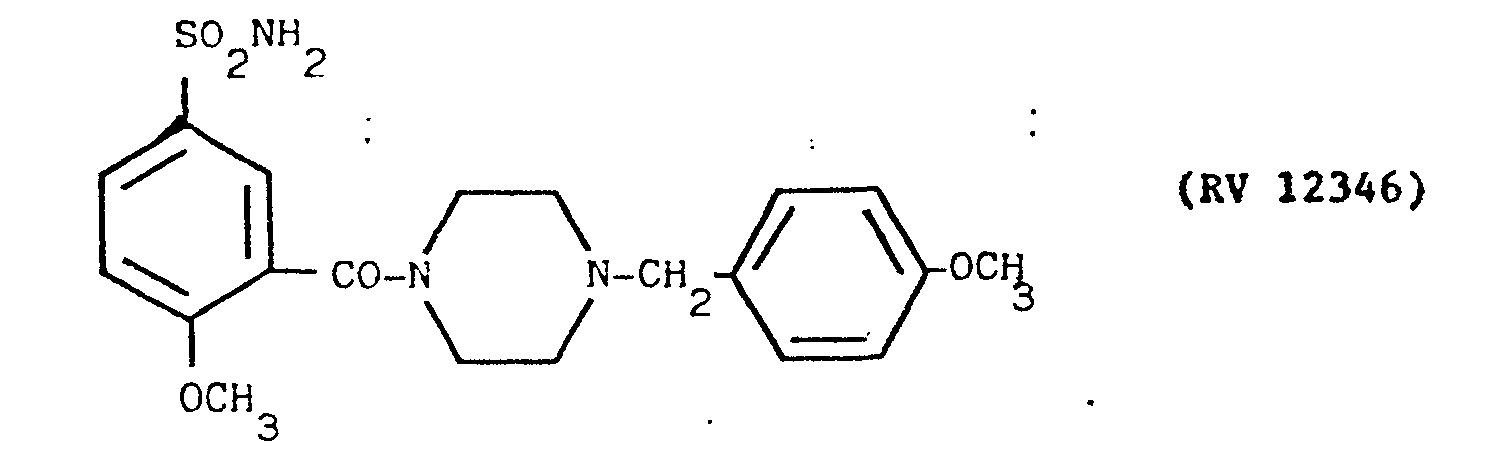
- a solution of 4-methoxy-benzylpiperazine in pyridine with an amine: solvent weight ratio of 1:15 was fed into the reactor, and a cold solution of PCI 3 in pyridine in a weight ratio of 1:10 was dripped in.
- reaction mixture was evaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washed with chloroform, acidified with acetic acid and filtered over carbon.
- the clear solution was alkalised with concentrated NH 4 OH under agitation.
- the precipitate was filtered off and washed with water.
- the acid-base treatment was repeated and the solid obtained was crystallised from isopropanol. Yield 65X.
- a solution of 3-methoxy-benzyl-piperazine in pyridine with an amine:solvent weight ratio of 1:10 was fed into the reactor, and a cold solution of PC1 3 in pyridine in a weight ratio of 1:10 was dripped in.
- reaction mixture was evaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washed with CH 2 C1 2 , acidified with acetic acid and filtered over carbon.
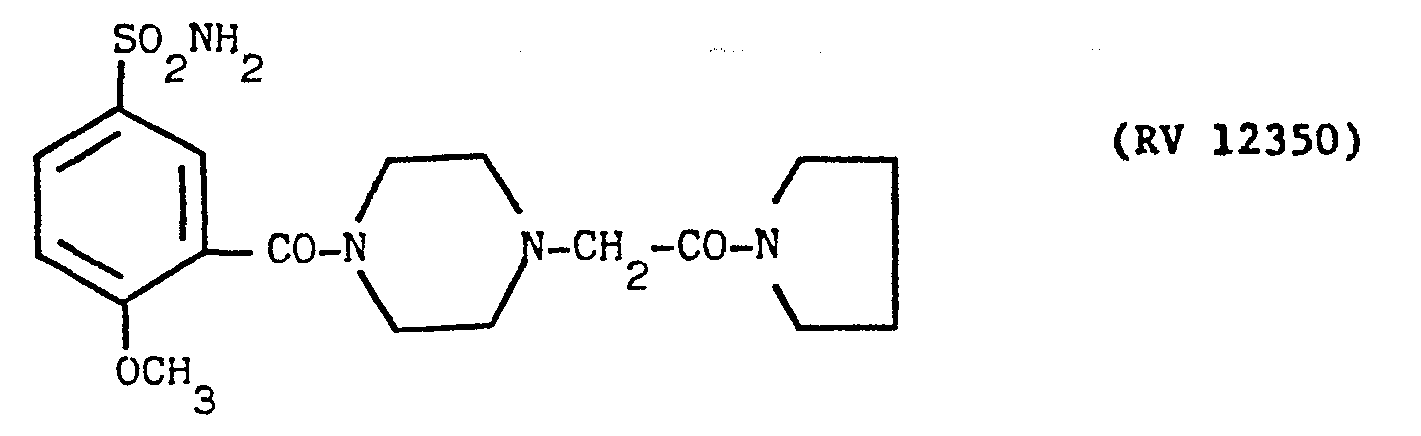
- a solution of pyrrolidino-carbonyl-N-methyl-piperazine in pyridine with an amine:solvent weight ratio of 1:10 was fed into the reactor, and a cold solution of PC1 3 in pyridine in a weight ratio of 1:10 was dripped in.
- the reaction mixture was evaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washed with ether, acidified with acetic acid and filtered over carbon.
- the clear solution was alkalised with concentrated NH 4 0H under agitation.
- the tacky precipitate was extracted with ethyl acetate, washed with water and evaporated to dryness.
- the residue was taken up in slightly alkaline water and solidified under agitation. It was filtered off and crystallised from aqueous ethyl alcohol. Yield 60X. M.P. - 205°-206°C.
- a solution of ethoxycarbonyl-methyl-piperazine in pyridine with an amine:solvent weight ratio of 1:10 was fed into the reactor, and a cold solution of PCl 3 in pyridine in a weight ratio of 1:10 was dripped in.
- reaction mixture was evaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washed with ether, acidified with acetic acid and filtered over carbon.
- the clear solution was alkalised with concentrated NH 4 0H under agitation.
- the tacky precipitate was extracted with ethyl acetate, precipitated with alcoholic HC1, filtered off and crystallised from ethanol. Yield 40%.
- a solution of cinnamyl-piperazine in pyridine with an amine:solvent weight ratio of 1:12 was fed into the reactor, and a cold solution of PCl 3 in pyridine in a weight ratio of 1:10 was dripped in..
- reaction mixture was evaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washed with ether, acidified with acetic acid and filtered over carbon.
- reaction mixture was evaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washed with CC1 4 , acidified with acetic acid and filtered over carbon.
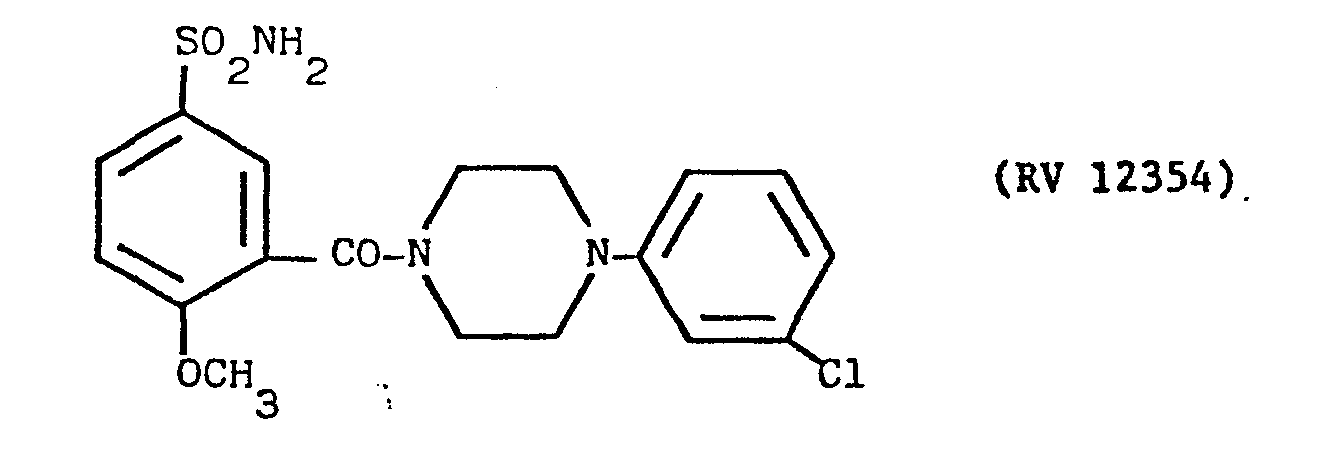
- a solution of N(3Cl-phenyl)piperazine in pyridine with an amine:solvent weight ratio of 1:10 was fed into the reactor, and a cold solution of PCl 3 in pyridine in a weight ratio of 1:10 was dripped in.
- reaction mixture was evaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washad with other, acidified with acetic acid and filtered over carbon.
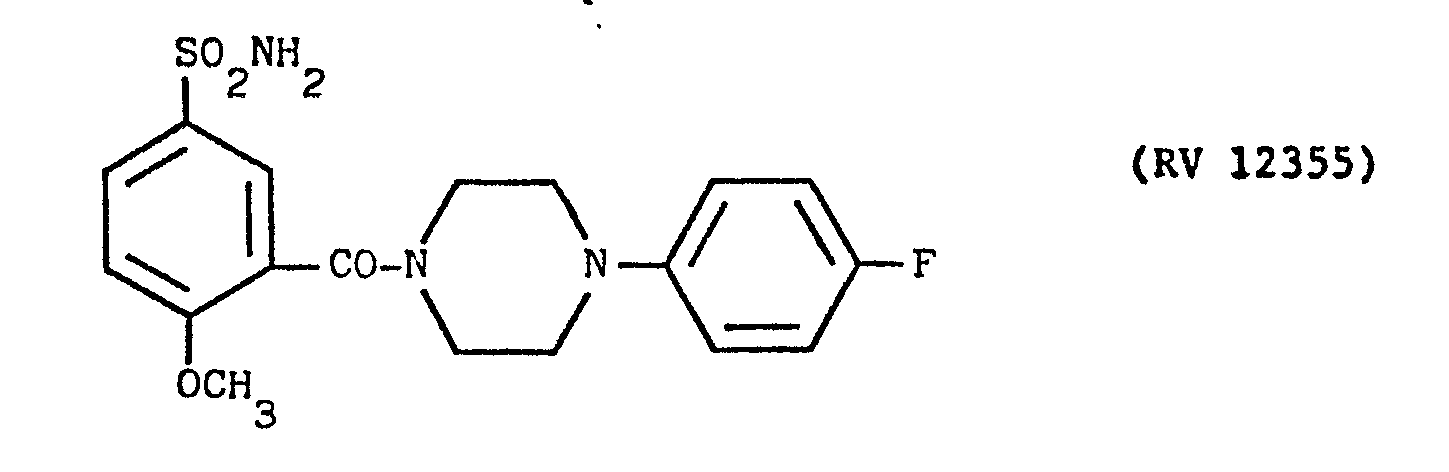
- a solution of N(4fluoro-phenyl)-piperazine in pyridine with an amine:solvent weight ratio of 1:12 was fed into the reactor, and a cold solution of PCl 3 in pyridine in a weight ratio of 1:10 was dripped in.
- reaction mixture was avaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washed with other, acidified with acetie acid and filtered over earbon.
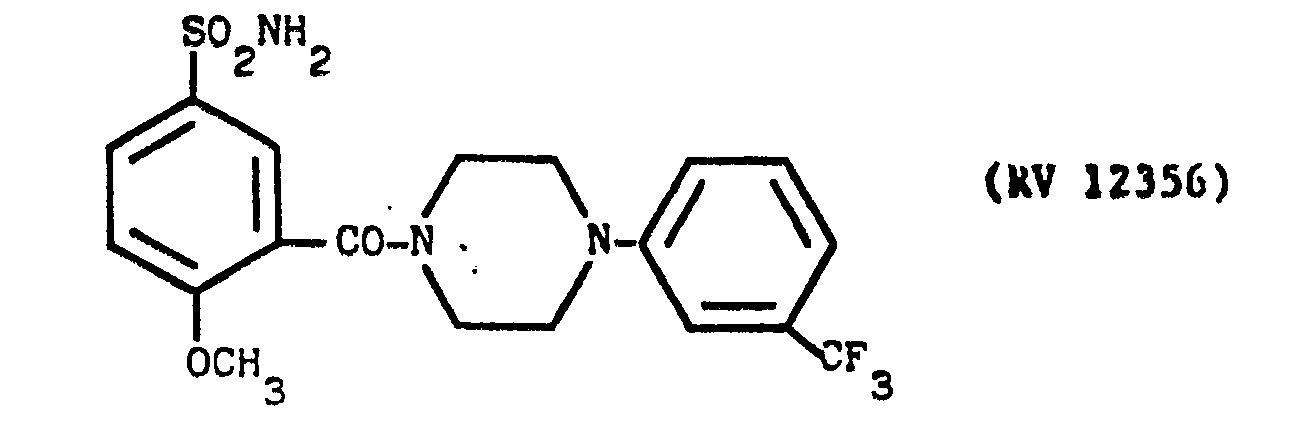
- a solution of N(3-trifluoromethyl-phenyl)piperasine in pyridine with an amine:solvent weight ratio of 1:10 was fed into the reactor, and a solution of PC1 3 in pyridine in a weight ratio of 1:10 was dripped in.
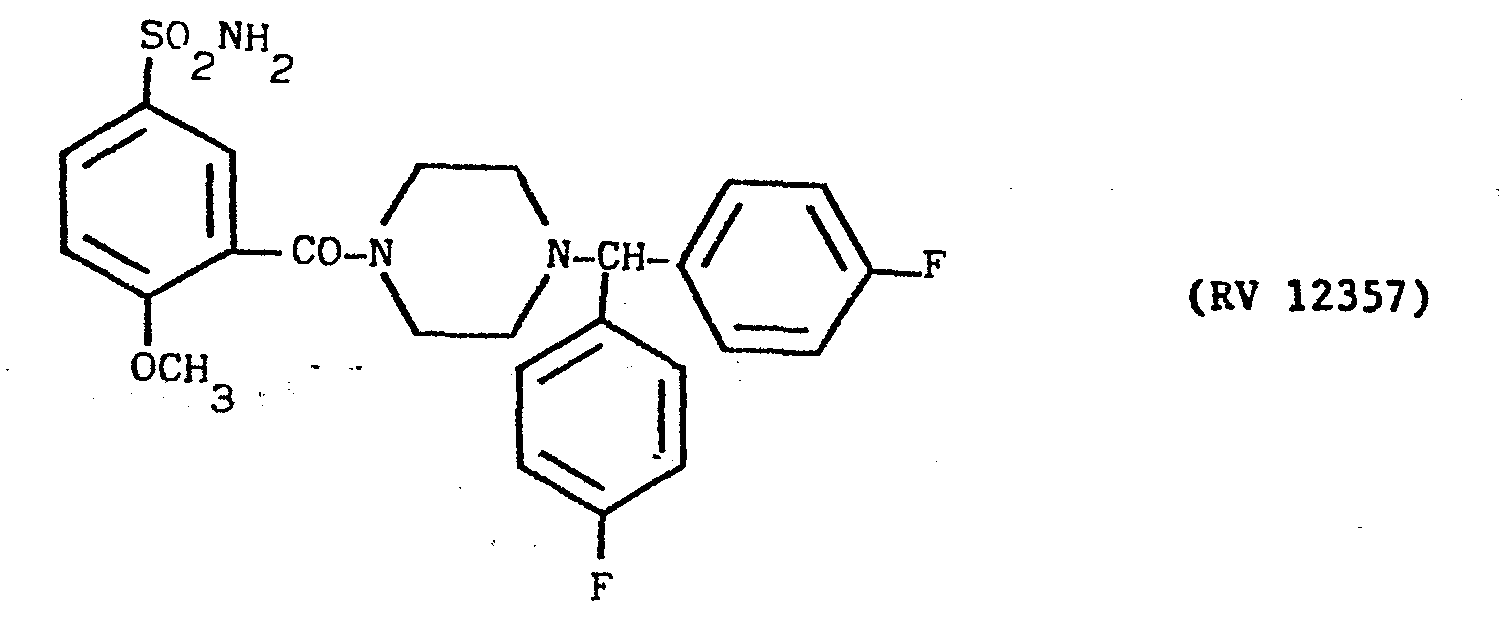
- a solution of bis(4F-phenyl)-methyl-piperazine in pyridine with an amine:solvent weight ratio of 1:10 was fed into the reactor, and a cold solution of PCl 3 in pyridine in a weight ratio of 1:10 was dripped in.
- a solution of isopropyl-amino-carbonyl-methyl-piperazine in pyridine with an amine:solvent weight ratio of 1:10 was fed into the reactor, and a cold solution of PCl 3 in pyridine in a weight ratio of 1:10 was dripped in.
- reaction mixture was evaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washed with ether, acidified with acetic acid and filtered over carbon.
- a solution of benzyl-plperazine in pyridine with an amine:solvent weight ratio of 1:15 was fed into the reactor, and a cold solution of PCl 3 in pyridine in a weight ratio of 1:10 was dripped in.
- reaction mixture was evaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washed with ether, acidified with acetic acid and filtered over carbon.
- the mixture was kept under agitation for 4-5 hours, cooling it with ice, after which it was evaporated to dryness under vacuum and the residue was taken up in dilute sodium hydroxide solution, washed with ether, acidified with acetic acid and filtered over carbon.
- Example 19 The compound obtained in Example 19 was hydrolysed in ethanol containing HC1 in a weight ratio of 20:3 by heating to a temperature of between 50° and 80°C for some hours.
- the mixture was kept under agitation for 3 hours at a temperature of 80°-100°C.
- reaction mixture was evaporated under vacuum, taken up in dilute sodium hydroxide solution, washed with chloroform, acidified with acetic acid and filtered over carbon.
- Tne mixture was heated to a temperature of 50°C for 2 hours.
- N-(2-methoxy-5-sulphamyl)benzoyl-piperazine obtained as described in Example 9 was reacted with 3F-benzylchloride in stoichiometric ratio in an aqueous alkal.ne solution at a temperature of 80°-100°C for 2 hours.
- the precipitated hydrochloride was purified by crystallisation from ethanol. It was dissolved in water and precipitated with ammonia. It was filtered off, washed with water and crystallised from ethanol. Yield 47X.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Pharmacology & Pharmacy (AREA)
- Engineering & Computer Science (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Heterocyclic Compounds That Contain Two Or More Ring Oxygen Atoms (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
- Plural Heterocyclic Compounds (AREA)
Abstract
New piperazino derivatives of formula
in which R is a phenyl radical substituted with at least one sulphonamido group or a substituted nitrogenated heterocyclit ring, and A is C0, CH2' SO2' possessing inhibiting activity towards carbonic anhydrase.
The new products are prepared from compounds of formula R-A-X in which X is OH, SH, halogen, OR"" or SR"" in which R'''' is methyl, ethyl, phenyl, carbomethoxy, carboethoxy, and from a piperazino derivative of formula
The R' group can also be introduced on termination of the reaction, by removing and substituting a protective group present at the nitrogen.
Description
- This invention relates to a new class of pharmacologically active piperazino derivatives, and the process for their preparation.
- More precisely, the invention relates to a new class of piperazino derivatives possessing inhibiting activity towards carbonic anhydrase. Carbonic anhydrase is known to be one of the most widespread enzymes in human tissues, and plays a determining role in many physiological and pathological processes.
- Consequently its inhibition can be a factor in the solution of therapeutical problems of considerable importance.
- The new compounds according to the present invention are those of general formula:
- Preferred meanings of the indicated radicals are as follows: R is 2-methoxy-5-sulphamoyl-phenyl, 2-sulphamoyl-imidazyl, 2-amino or 2-acetamido-1,3,4-thiadiazole; R' is H, linear or branched C1-C6 alkyl, linear or branched C1-C6 alkenyl, alkyl-phenyl or alkenyl-phenyl in which the phenyl group is either simple or substituted with halogens, trihalomethyls, C1-C6 alkyls, C1-C6 alkoxys, C1-C6 alkylethers, sulphonamido groups; C2-C6 aliphatic acyl; alkyl-acyl radical in which the acyl is C1-C6aliphatic, aromatic or nitrogenated heterocyclic; C3-C6 alkyl-carboxyalkoxy; alkyl-carboxyamino in which the alkyl is C1-C3 and the amine is mono or disubstituted with linear or branched C1-C6 alkyl radicals; R" is H or together with R' forms a 5 or 6 term ring condensed with the piperazine, but again taking into account said limitation that when R is a substituted phenyl radical and A is CO, R' is other than H, C1-C4 alkyl, benzyl.
- The new products according to the present invention are prepared essentially in accordance with the following reaction:
- The R' substituents can be introduced to the piperazino nitrogen either before or after reaction (II) is effected, using known methods comprising reacting the piperazino nitrogen bonded to the hydrogen with the required compound containing a reactive group.
-
-
- It is in any case apparent that instead of the benzyl group, use can be made of any other suitable protective group of one of the piperazino nitrogens, which can be eliminated when reaction (II) is terminated.
- The R'-R" cyclisation can also be effected before or after effecting reaction (II).
- Reaction (II) is preferably effected by reacting, with the compound R-A-OH and PCl3 or POC15 in a basic medium, the piperazino derivative containing the group R' or with one of the nitrogens suitably protected.
- The amine:R-A-OH:PC13 ratio is generally stoichiometric, or has a slight deficiency (10-20X) of the compound R-A-OH relative to the stoichiometric.
- The basic medium is preferably pyridine, or any organic basic solvent able to block the HC1 which forms during the reaction. The reaction takes place under hot conditions, preferably at the mixture reflux temperature.
- The product obtained is separated and purified by normal methods.
- In particular, it is purified by repeated crystallisations either as such or in the form of the hydrochloride. The compounds in which A is CH2 can be prepared by reducing the corresponding compounds in which A is C0, obtained by process (II).
- Some practical examples are described hereinafter in order to better clarify the process according to the present invention and make it more easily reproducible, but these are to be considered as purely illustrative and in no case limitative of the process according to the present invention.
-
- Amine:PCl3:acid molar ratio = 3:1:2.5
- The cooled reaction mixture was evaporated under vacuum, taken up in dilute 10% sodium hydroxide solution, washed with ether, acidified with acetic acid and filtered over carbon.
- The clear solution was alkalised with concentrated ammonia under agitation.
- The precipitate was filtered off after some hours and washed with water.
- The product RV 12343 in the form of the hydrochloride was purified by repeated crystallisations from n.butanol. Yield 65X with respect to the acid.
-
-
- Amine:PCl3:acid molar ratio - 3:1:3
- The cooled reaction mixture was evaporated to dryness under vacuum, taken up in dilute 20% sodium hydroxide solution, washed with chloroform, acidified with acetic acid and filtered over carbon.
- The clear solution was alkalised with IN NaOH and left for some hours under agitation.
- The precipitate was then filtered off, washed with water and recrystallised from ethyl acetate. Yield 35X.
-
-
- The mixture was kept under agitation for one hour at ambient temperature, after which 2-methoxy-5-sulphamoyl-benzoic acid was added, and the mixture heated under reflux for 4 hours. Amine:PC13:acid ratio = 3:1:3.
- The reaction mixture was evaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washed with ether, acidified with acetic acid and filtered over carbon.
- The clear solution was alkalised with concentrated NH40H under agitation. The product does not precipitate, and it is therefore extracted with chloroform, washed with water, the organic phase evaporated to dryness, and crystallised from isopropyl alcohol. Yield 68X.
- M. P. = 123°-125°C
-
-
- The mixture was kept under agitation for one hour at ambient temperature, after which 2-methoxy-5-sulphamoyl-benzoic acid was added and the mixture heated under reflux for 4 hours. Amine:PC13:acid ratio - 3:1:2.7
- The reaction mixture was evaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washed with CC14, acidified with dilute hydrochloric acid and filtered over carbon. The clear solution was alkalised with concentrated NH4OH under agitation.
- The precipitate was filtered off, washed with water and recrystallised from ethyl alcohol. Yield 40X.
-
-
- The mixture was kept under agitation for one hour at ambient temperature, after which 2-methoxy-5-sulphamoyl-benzoic acid was added and the mixture heated under reflux for 4 hours. Amine:PCl3:acid ratio - 3:1:2.5
- The reaction mixture was evaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washed with ether, acidified with acetic acid and filtered over carbon.
- The clear solution was alkalised with concentrated Na2CO3 under agitation. The slightly tacky precipitate was filtered off, washed with water, dissolved in acidified water and reprecipitated with dilute ammonia. Yield 40%.
-
-
- The mixture was kept under agitation for 2 hours at ambient temperature, after which 2-methoxy-5-sulphamoyl-benzoic acid was added and the mixture heated under reflux for 3 hours. Amine:PCl3:acid ratio - 3:1:3
- The reaction mixture was evaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washed with chloroform, acidified with acetic acid and filtered over carbon. The clear solution was alkalised with concentrated NH4OH under agitation. The precipitate was filtered off and washed with water. The acid-base treatment was repeated and the solid obtained was crystallised from isopropanol. Yield 65X.
- M.P. = 160°-162°C
-
-
- The mixture was kept under agitation for 1.5 hours at ambient temperature, after which 2-methoxy-5-sulphamoyl-benzoic acid was added and the mixture heated under reflux for 4 hours. Amine:PC13:acid ratio - 3:1:3
- The reaction mixture was evaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washed with CH2C12, acidified with acetic acid and filtered over carbon.
- The clear solution was alkalised with IN KOH under agitation. The precipitate was filtered off, washed with water and recrystallised from isopropyl alcohol. Yield 67X.
-
-
- The mixture was kept under agitation for one hour at ambient temperature, after which 2-methoxy-5-sulphamoyl-benzoic acid was added and the mixture heated under reflux for 5 hours. Amine:PCl3:acid ratio - 3:1:3.5
- The reaction mixture was evaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washed with ether, acidified with acetic acid and filtered over carbon. The clear solution was alkalised with concentrated NH40H under agitation. The tacky precipitate was extracted with ethyl acetate, washed with water and evaporated to dryness. The residue was taken up in slightly alkaline water and solidified under agitation. It was filtered off and crystallised from aqueous ethyl alcohol. Yield 60X. M.P. - 205°-206°C.
-
-
- The 2-methoxy-5-sulphamoyl-benzoylpiperazine obtained in this manner was reacted with acetyl chloride in a molar ratio of 1:1, in benzene in the presence of pyridine, was filtered off, washed and crystallised from a mixture of methanol and ethyl acetate. Yield 70%.
-
-
- A solution of ethoxycarbonyl-methyl-piperazine in pyridine with an amine:solvent weight ratio of 1:10 was fed into the reactor, and a cold solution of PCl3 in pyridine in a weight ratio of 1:10 was dripped in.
- The mixture was kept under agitation for one hour at ambient temperature, after which 2-methoxy-5-sulphamoyl-benzoic acid was added and the mixture heated under reflux for 3 hours. Amine:PC13:acid ratio = 3:1:3.
- The reaction mixture was evaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washed with ether, acidified with acetic acid and filtered over carbon.
- The clear solution was alkalised with concentrated NH40H under agitation. The tacky precipitate was extracted with ethyl acetate, precipitated with alcoholic HC1, filtered off and crystallised from ethanol. Yield 40%.
-
-
- A solution of cinnamyl-piperazine in pyridine with an amine:solvent weight ratio of 1:12 was fed into the reactor, and a cold solution of PCl3 in pyridine in a weight ratio of 1:10 was dripped in..
- The mixture was kept under agitation for 2 hours at ambient temperature, after which 2-methoxy-5-sulphamoyl-benzoic acid was added and the mixture heated under reflux for 4 hours. Amine:PC13:acid ratio = 3:1:2.5
- The reaction mixture was evaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washed with ether, acidified with acetic acid and filtered over carbon.
-
-
- The mixture was kept under agitation for one hour at ambient temperature, after which 2-methoxy-5-sulphamoyl-benzoic acid was added and the mixture heated under reflux for 4 hours. Amine:PCl3:acid ratio = 3:1:3.
- -The reaction mixture was evaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washed with CC14, acidified with acetic acid and filtered over carbon.
- The clear solution was alkalised with concentrated NH40H under agitation. The precipitate was filtered off, washed with water and recrystallised from ethanol. Yield 61X.
-
- Preparation of the compound of formula
-
- The mixture was kept under agitation for 2 hours at ambient temperature, after which 2-methoxy-5-sulphnmoyl-henzoic acid was added and the mixture heated under reflux for 2 hours, Amine:PCl3:acid ratio = 3:1:2.7
- The reaction mixture was evaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washad with other, acidified with acetic acid and filtered over carbon.
- The clear solution was alkalised with concentrated NH4OH under agitation. The precipitate was filtered off and washed with water. The acid-base treatment was repeated, and the solid obtained was crystallised from aqueous methanol. Yield 60%.
-
-
- A solution of N(4fluoro-phenyl)-piperazine in pyridine with an amine:solvent weight ratio of 1:12 was fed into the reactor, and a cold solution of PCl3 in pyridine in a weight ratio of 1:10 was dripped in.
- The mixture was kept under agitation for one hour at ambient temperature, after which 2-methoxy-5-sulphamoyl-benzoic acid was added and the mixtrue hheated under reflux for 3 hours, Anine:PCl3:acid ratio = 3:1:3
- The reaction mixture was avaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washed with other, acidified with acetie acid and filtered over earbon.
- The clear solution was alkalised with concentrated NH4OH under agitation. The precipitate was filtered off, washed with water and recrystallised from ethanol. Yield 30%.
- M.P. = 195°-197°C
-
-
- A solution of N(3-trifluoromethyl-phenyl)piperasine in pyridine with an amine:solvent weight ratio of 1:10 was fed into the reactor, and a solution of PC13 in pyridine in a weight ratio of 1:10 was dripped in.
- The mixture was kept under agitation for 1.5 hours at ambient temperature, after which 2-methoxy-5-sulphamoyl-benzoic acid was added and the mixture heated under reflux for 4 hours. Amine:PCl3:acid ratio = 3:1:2.5 The reaction mixture was evaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washed with CHC13, acidified with acetic acid and filtered over carbon.
- The clear solution was alkalised with concentrated NaOH under agitation. The precipitate was filtered off, washed with water and recrystallised from ethanol. Yield 32X.
- M.P. - 125°-127°C
-
-
- A solution of bis(4F-phenyl)-methyl-piperazine in pyridine with an amine:solvent weight ratio of 1:10 was fed into the reactor, and a cold solution of PCl3 in pyridine in a weight ratio of 1:10 was dripped in.
- The mixture was kept under agitation for 2 hours at ambient temperature, after which 2-methoxy-5-sulphamoyl-benzoic acid was added, and the mixture heated under reflux for 4 hours. Amine:PCl3:acid ratio - 3:1:3 the reaction mixture was evaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washed with ether, acidified with acetic acid and filtered over carbon.
- The clear solution was alkalised with concentrated NH4OH under agitation. The precipitate was filtered off, washed with water and recrystallised from n.butanol. Yield 56X.
- M.P. = 238°-240°C
-
- Preparation of the compound of formula
-
- Tne mixture was kept under agitation for one hour at ambient temperature, after which 2-methoxy-5-sulphamoyl-benzoic acid was added and the mixture heated under reflux for 2 hours. Amine:PCl3:acid ratio = 3:1:2.5
- The reaction mixture was evaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washed with ether, acidified with acetic acid and filtered over carbon.
- The clear solution was alkalised with concentrated NH4OH under agitation. The precipitate was filtered off, washed with water and recrystallised from ethanol. Yleld 48%.
- M.P. - 228°-230°C
-
- Preparation of the compound of formula
-
- The mixture was kept under agitation for one hour at ambient temperature, after which 2-sulphamoyl-4-imidazole carboxylic acid was added and the mixture heated under reflux for 3 hours. Amine:PCl3:acid ratio - 3:1:3.
- The reaction mixture was evaporated to dryness under vacuum, taken up in dilute sodium hydroxide solution, washed with ether, acidified with acetic acid and filtered over carbon.
- The clear solution was alkalised with concentrated NH4OH under agitation. The precipitate was filtered off, washed with water and recrystallised from n.butanol. Yield 30X.
- M.P. = 224°-225°C
-
- Preparation of the compound of formula
-
- The mixture was kept under agitation for 4-5 hours, cooling it with ice, after which it was evaporated to dryness under vacuum and the residue was taken up in dilute sodium hydroxide solution, washed with ether, acidified with acetic acid and filtered over carbon.
- The clear solution was alkalised with concentrated NH4OH under agitation. The precipitate was filtered off, washed with water and recrystallised from ethanol. Yield 70%.
- M.P. - 238°-240°C
-
-
- The compound obtained in Example 19 was hydrolysed in ethanol containing HC1 in a weight ratio of 20:3 by heating to a temperature of between 50° and 80°C for some hours.
- The product obtained was isolated from the reaction mixture as described in the preceding examples, and recrystallised from ethanol. Yield 93%.
-
-
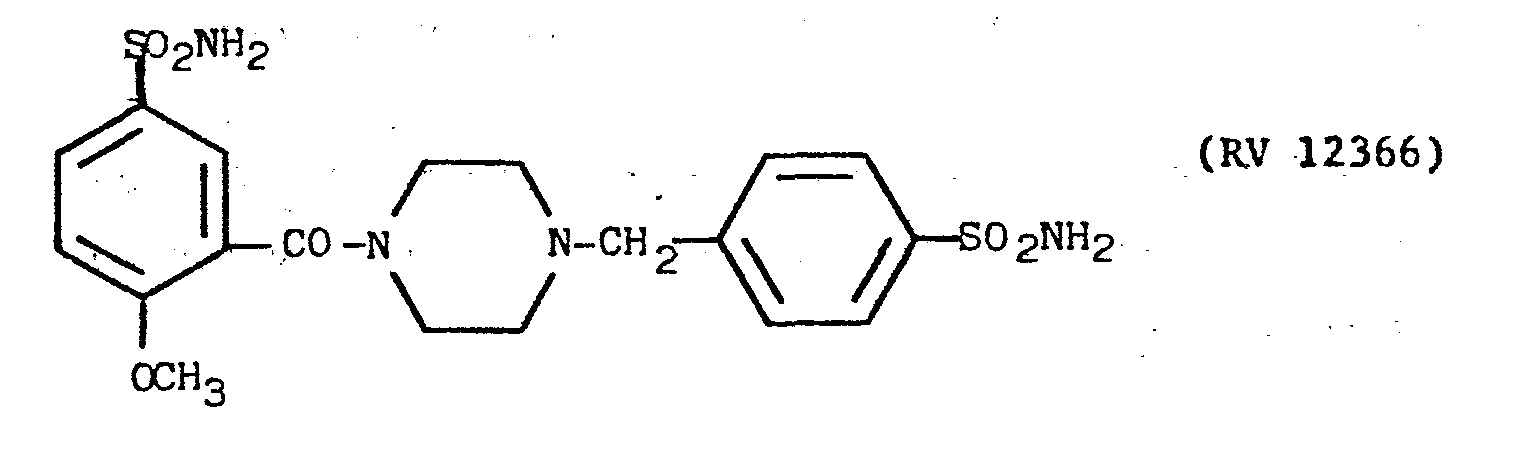
- The N(2-methoxy-5-sulphamoyl)benzoyl-piperazine obtained as described in Example 9 was reacted with 4-sulphamido-benzyl chloride in a 1.1 N aqueous NaOH solution.
- The mixture was kept under agitation for 3 hours at a temperature of 80°-100°C.
- On cooling the aqueous solution, the desired compound precipitated and was purified by crystallisation from methanol. Yield 691.
-
-
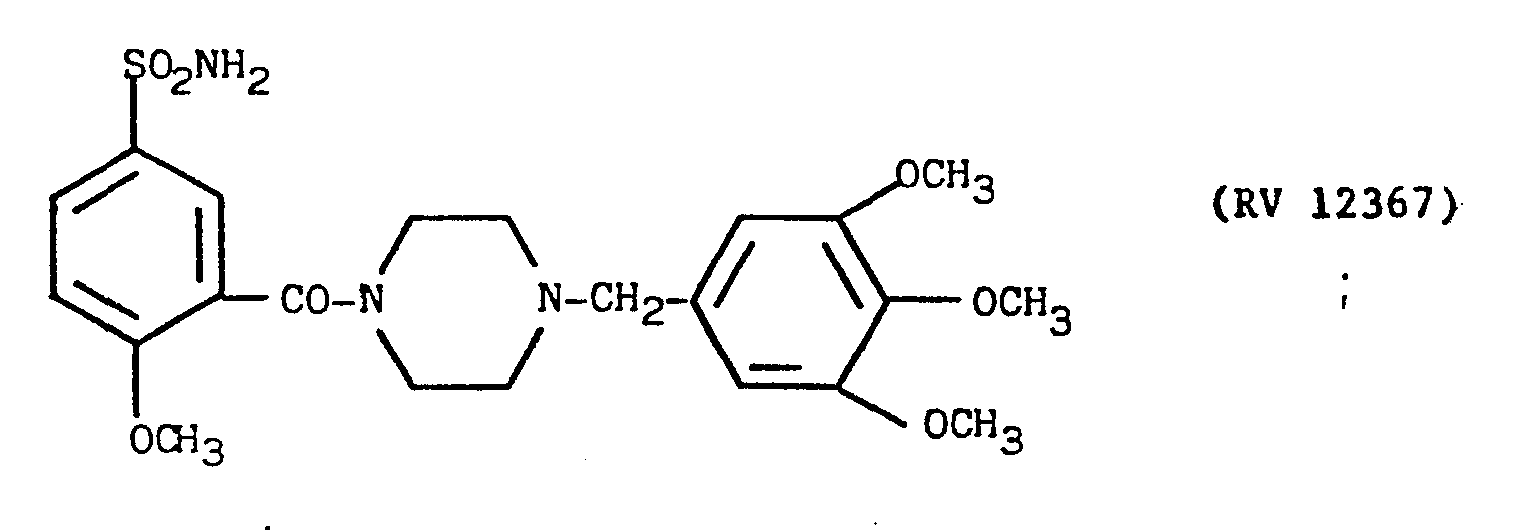
- The N-(2-methoxy-5-sulphamoyl)benzoyl-piperazine obtained as described in Example 9 was reacted under reflux for some hours with 3,4,5-trimethoxy-benzylchloride in stoichiometric ratio in a 1:1 chloroform: ethanol mixture in the presence of pyridine.
- The reaction mixture was evaporated under vacuum, taken up in dilute sodium hydroxide solution, washed with chloroform, acidified with acetic acid and filtered over carbon.
- The clear solution was alkalised with concentrated KOH under agitation. The precipitate was filtered off, washed with water and purified with silica gel. Yield 45X.
-
-
- The N(2-methoxy-5-sulphamoyl)benzoyl-piperazine obtained as described in Example 9 was reacted with 3-methyl-benzylchloride in stoichiometric ratio in an aqueous dilute NaOH solution.
- Tne mixture was heated to a temperature of 50°C for 2 hours.
- The desired product precipitated on cooling, and was recrystallised from isopropyl alcohol. Yield 50%.
-
-
- The N-(2-methoxy-5-sulphamyl)benzoyl-piperazine obtained as described in Example 9 was reacted with 3F-benzylchloride in stoichiometric ratio in an aqueous alkal.ne solution at a temperature of 80°-100°C for 2 hours.
- The desired product precipitated on cooling, and was recrystallised from isopropyl alcohol. Yield 35X.
-
-
- Benzyl-piperazine and 2-methoxy-5-sulphamoyl-methylenechloride in stoichiometric ratio were fed in chloroform solution into a reactor.
- The mixture was kept under reflux for 4 hours.
- When the reaction terminated, the mixture was poured into ice and acidified with HCl.
- The precipitated hydrochloride was purified by crystallisation from ethanol. It was dissolved in water and precipitated with ammonia. It was filtered off, washed with water and crystallised from ethanol. Yield 47X.
-
Claims (7)
1. Piperazino derivatives of formula
in which:
R is a phenyl radical containing at least one sulphonamido group and possibly other substituents chosen from the group consisting of halogen, C1-C4 alkyl, and C1-C4 alkoxy; or a nitrogenated heterocyclic ring containing as substituent at least one amino, amido or sulphonamido group;
A is CO, CH2, SO2;
R' is H, alkyl, alkenyl, alkyl-aryl, alkyl-cycloalkyl, alkyl-acyl, alkyl-carboxyalkoxy, alkyl-carboxyamino, alkenyl-aryl, alkenyl- cycloalkyl, alkenyl-acyl, alkenyl-carboxyalkoxy, alkenyl-carboxyamino, acyl, aryl;
R" is H or together with R' and with the nitrogen atom to which this latter is bonded forms a heterocyclic ring, provided that when R is a substituted phenyl radical and A is CO, R' is other than H, C1-C4 alkyl, or benzyl.
2. Piperazino derivatives as claimed in claim I, wherein
K is 2-methoxy-5-sulphamoyl-phenyl, 2-sulphamoyl-imidazyl, 2-amino or 2-acetamido-1,3,4-thiadiazole;
R' is H, linear or branched C1-C6 alkyl, linear or branched C1-C6 alkenyl, alkyl-phenyl or alkenyl-phenyl in which the phenyl group is either simple or substituted with halogens, trihalomethyls, C1-C6 alkyls, C1-C6 alkoxys, C1-C6 alkylethers, sulphonamido groups; C2-C6 aliphatic acyl; alkylacyl radical in which the acyl is C1-C6 aliphatic, aromatic or nitrogenated heterocyclic; C3-C6 alkylcarboxyalkoxy; alkyl-carboxyamino in which the alkyl is C1-C3 and the amine is mono or disubstituted with linear or branched C1-C6 alkyl radicals; R" is H or together with R' forms a 5 or 6 term ring condensed with the piperazine, provided that when R is a substituted phenyl radical and A is CO, R' is other than H, C1-C4 alkyl, or benzyl.
3. A process for preparing piperazino derivatives of formula
in which A, R, R', R" are as defined in claim 1, characterised by effecting the following reaction
in which X is OH, SH, Cl, Br, I, OR"", SR"" in which R"" is methyl, ethyl, phenyl, carbomethoxy, carboethoxy; AR, R, R" are as defined defined in claim 1, R"' is R' or. a protective group at the nitrogen, in the presence of a base or condensing agent, and the product obtained can be subjected to elimination of the protective group at the nitrogen and to introduction of a R' radical as defined in claim 1, or, when A is CO, can be subjected to reduction in order to obtain compounds in which A is CH2.
4. A process as claimed in claim 3, wherein the compound of formula R-A-OH is reacted with the piperazino derivative in the presence of PC13 or POCl5 in a basic solvent, and the product obtained is isolated by conventional methods.
5. A process as claimed in claim 4, wherein the reaction is conducted with an amine:PCl3:acid ratio of 3:1:2.5-3, in pyridine.
6. A process as claimed in claim 3, wherein R"' is benzyl and is eliminated by reduction with hydrogen using Pd on carbon as catalyst, before introducing the R' group by reaction with a compound of formula YR', in which Y is halogen, in an organic solvent in the presence of a strong inorganic or organic base.
7. A process as claimed in claim 3, wherein the reduction of the group A = CO to the group CH2 is effected by hydrogenation.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| IT2233484 | 1984-08-14 | ||
| IT22334/84A IT1176613B (en) | 1984-08-14 | 1984-08-14 | PHARMACOLOGICALLY ACTIVE PIPERAZINIC DERIVATIVES AND PROCESS FOR THEIR PREPARATION |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| EP0171636A1 true EP0171636A1 (en) | 1986-02-19 |
Family
ID=11194823
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP85108942A Withdrawn EP0171636A1 (en) | 1984-08-14 | 1985-07-17 | Pharmacologically active piperazino derivatives and the process for their preparation |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US4940793A (en) |
| EP (1) | EP0171636A1 (en) |
| JP (1) | JPS6156167A (en) |
| IT (1) | IT1176613B (en) |
Cited By (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0284359A1 (en) * | 1987-03-24 | 1988-09-28 | Takeda Chemical Industries, Ltd. | 1,4-Disubstituted piperazine compounds, their production and use |
| US4804661A (en) * | 1986-06-16 | 1989-02-14 | Ciba-Geigy Corporation | Disubstituted piperazines |
| EP0548798A1 (en) * | 1991-12-18 | 1993-06-30 | Sanwa Kagaku Kenkyusho Co., Ltd. | Antiviral agent |
| WO2005014563A1 (en) * | 2003-08-11 | 2005-02-17 | F. Hoffmann-La Roche Ag | Piperazine with or-substituted phenyl group and their use as glyt1 inhibitors |
| WO2005023260A1 (en) * | 2003-09-09 | 2005-03-17 | F. Hoffmann-La Roche Ag | 1- (2-amino-benzol) -piperazine derivatives as glycine uptake inhibitors for the treatment of psychoses |
| WO2005023261A1 (en) * | 2003-09-09 | 2005-03-17 | F. Hoffmann-La Roche Ag | 1-benzoyl-piperazine derivatives as glycine uptake inhibitors for the treatment of psychoses |
| US7046924B2 (en) | 2002-11-25 | 2006-05-16 | Eastman Kodak Company | Method and computer program product for determining an area of importance in an image using eye monitoring information |
| US7220744B2 (en) | 2005-01-07 | 2007-05-22 | Hoffman-La Roche Inc. | Monocyclic substituted phenyl methanones |
| US7241761B2 (en) | 2004-12-09 | 2007-07-10 | Hoffmann-La Roche Inc. | Phenyl-piperazine methanone derivatives, substituted by heterocyclic groups |
| US7327890B2 (en) | 2002-12-20 | 2008-02-05 | Eastman Kodak Company | Imaging method and system for determining an area of importance in an archival image |
| US7375226B2 (en) | 2004-12-15 | 2008-05-20 | Hoffman-La Roche Inc. | Bi- and tricyclic substituted phenyl methanones |
| US7442710B2 (en) | 2005-01-26 | 2008-10-28 | Hoffman-La Roche Inc. | Substituted phenyl methanones |
| US7485637B2 (en) | 2005-01-04 | 2009-02-03 | Hoffmann-La Roche Inc. | Benzoyl-tetrahydropiperidine derivatives |
| US7557114B2 (en) | 2005-02-07 | 2009-07-07 | Hoffman-La Roche Inc. | Heterocyclic-substituted phenyl methanones |
| US7790763B2 (en) | 2005-01-18 | 2010-09-07 | Hoffmann-La Roche Inc. | Substituted phenyl methanone derivatives |
Families Citing this family (40)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| IT1251144B (en) * | 1991-07-30 | 1995-05-04 | Boehringer Ingelheim Italia | BENZIMIDAZOLONE DERIVATIVES |
| US5565567A (en) * | 1993-06-04 | 1996-10-15 | Henkel Corporation. | Polymerizable N,N'-substituted piperazine acrylamide compounds |
| CN1126474A (en) * | 1993-06-04 | 1996-07-10 | 亨凯尔公司 | polymerized compound |
| US5922820A (en) * | 1993-06-04 | 1999-07-13 | Henkel Corporation | Polymerizable compounds |
| US5856326A (en) * | 1995-03-29 | 1999-01-05 | Merck & Co., Inc. | Inhibitors of farnesyl-protein transferase |
| US5756742A (en) * | 1995-12-22 | 1998-05-26 | Henkel Corporation | Polymerizable compounds |
| JP2000507579A (en) * | 1996-04-03 | 2000-06-20 | メルク エンド カンパニー インコーポレーテッド | Farnesyl-protein transferase inhibitor |
| US5885995A (en) * | 1996-04-03 | 1999-03-23 | Merck & Co., Inc. | Inhibitors of farnesyl-protein transferase |
| US5972942A (en) * | 1997-03-27 | 1999-10-26 | Merck & Co., Inc. | Inhibitors of farnesyl-protein transferase |
| JP2002538121A (en) * | 1999-03-03 | 2002-11-12 | メルク エンド カムパニー インコーポレーテッド | Inhibitors of prenyl protein transferase |
| US7183410B2 (en) * | 2001-08-02 | 2007-02-27 | Bidachem S.P.A. | Stable polymorph of flibanserin |
| US20030060475A1 (en) * | 2001-08-10 | 2003-03-27 | Boehringer Ingelheim Pharma Kg | Method of using flibanserin for neuroprotection |
| US10675280B2 (en) | 2001-10-20 | 2020-06-09 | Sprout Pharmaceuticals, Inc. | Treating sexual desire disorders with flibanserin |
| UA78974C2 (en) * | 2001-10-20 | 2007-05-10 | Boehringer Ingelheim Pharma | Use of flibanserin for treating disorders of sexual desire |
| US20050239798A1 (en) * | 2004-04-22 | 2005-10-27 | Boehringer Ingelheim Pharmaceuticals, Inc. | Method for the treatment of premenstrual and other female sexual disorders |
| AU2005235422B2 (en) * | 2004-04-22 | 2011-08-11 | Boehringer Ingelheim International Gmbh | New pharmaceutical compositions for the treatment of sexual disorders II |
| US20060025420A1 (en) * | 2004-07-30 | 2006-02-02 | Boehringer Ingelheimn International GmbH | Pharmaceutical compositions for the treatment of female sexual disorders |
| JP2008511569A (en) * | 2004-09-03 | 2008-04-17 | ベーリンガー インゲルハイム インターナショナル ゲゼルシャフト ミット ベシュレンクテル ハフツング | How to treat attention-deficit hyperactivity disorder |
| WO2006096439A2 (en) * | 2005-03-04 | 2006-09-14 | Boehringer Ingelheim International Gmbh | Pharmaceutical compositions for the treatment and/or prevention of schizophrenia and related diseases |
| EP1858515A2 (en) * | 2005-03-04 | 2007-11-28 | Boehringer Ingelheim International Gmbh | Pharmaceutical compositions for the treatment and/or prevention of anxiety disorders |
| CA2599721A1 (en) * | 2005-03-04 | 2006-09-14 | Boehringer Ingelheim International Gmbh | Pharmaceutical compositions for the treatment and/or prevention of depression |
| WO2006119884A2 (en) * | 2005-05-06 | 2006-11-16 | Boehringer Ingelheim International Gmbh | Method for the treatment of drug abuse with flibanserin |
| CA2608713A1 (en) * | 2005-05-19 | 2006-11-23 | Boehringer Ingelheim International Gmbh | Method for the treatment of sexual dysfunctions due to medical conditions |
| EP1888071A1 (en) * | 2005-05-19 | 2008-02-20 | Boehringer Ingelheim International GmbH | Method for the treatment of drug-induced sexual dysfunction |
| PL1912650T3 (en) | 2005-08-03 | 2018-01-31 | Sprout Pharmaceuticals Inc | Use of flibanserin in the treatment of obesity |
| US20070123540A1 (en) * | 2005-10-29 | 2007-05-31 | Angelo Ceci | Sexual desire enhancing medicaments comprising benzimidazolone derivatives |
| WO2007048803A1 (en) * | 2005-10-29 | 2007-05-03 | Boehringer Ingelheim International Gmbh | Benzimidazolone derivatives for the treatment of premenstrual and other female sexual disorders |
| US20070105869A1 (en) * | 2005-11-08 | 2007-05-10 | Stephane Pollentier | Use of flibanserin for the treatment of pre-menopausal sexual desire disorders |
| WO2007103456A2 (en) * | 2006-03-06 | 2007-09-13 | Trimeris, Inc. | Piperazine and piperidine biaryl derivatives |
| RS54398B1 (en) * | 2006-05-09 | 2016-04-28 | Sprout Pharmaceuticals, Inc. | THE USE OF FLIBANSERINE FOR THE TREATMENT OF SEXUAL DESIRE DISORDERS IN POSTMENOPAUSE |
| JP2009541443A (en) | 2006-06-30 | 2009-11-26 | ベーリンガー インゲルハイム インターナショナル ゲゼルシャフト ミット ベシュレンクテル ハフツング | Flibanserin for the treatment of urinary incontinence and related diseases |
| US20090318469A1 (en) * | 2006-07-14 | 2009-12-24 | Boehringer Ingelheim International Gmbh | Use of Flibanserin for the Treatment of Sexual Disorders in Females |
| CA2660476C (en) * | 2006-08-14 | 2015-11-17 | Boehringer Ingelheim International Gmbh | Formulations of flibanserin and method for manufacturing the same |
| CL2007002214A1 (en) * | 2006-08-14 | 2008-03-07 | Boehringer Ingelheim Int | PHARMACEUTICAL COMPOSITION IN THE FORM OF COMPRESSED, WHERE AT LEAST THE LENGTH OF THE COMPRESSED IN THE PREVIOUS STATE OF THE APPLICATION IS AT LEAST 7/12 OF THE PILOR DIAMETER OF THE PATIENT AND AFTER INGERING IT IN THE FOOD STATE, THE LENGTH OF THE COMP |
| KR20090045945A (en) * | 2006-08-25 | 2009-05-08 | 베링거 인겔하임 인터내셔날 게엠베하 | Controlled Release System and Manufacturing Method Thereof |
| CL2008002693A1 (en) * | 2007-09-12 | 2009-10-16 | Boehringer Ingelheim Int | Use of flibanserin for the treatment of selected vasomotor symptoms of hot flashes, night sweats, mood swings, and irritability |
| WO2009118292A1 (en) | 2008-03-24 | 2009-10-01 | Novartis Ag | Arylsulfonamide-based matrix metalloprotease inhibitors |
| CA2937616A1 (en) | 2014-03-26 | 2015-10-01 | F. Hoffmann-La Roche Ag | Bicyclic compounds as autotaxin (atx) and lysophosphatidic acid (lpa) production inhibitors |
| PE20180479A1 (en) | 2015-09-04 | 2018-03-07 | Hoffmann La Roche | NEW DERIVATIVES OF PHENOXYMETHYL |
| JP7090099B2 (en) | 2017-03-16 | 2022-06-23 | エフ.ホフマン-ラ ロシュ アーゲー | A novel bicyclic compound as an ATX inhibitor |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR1034958A (en) * | 1950-04-04 | 1953-08-10 | American Cyanamid Co | Improvements in the preparation of heterocyclic sulfonamides |
| FR1267777A (en) * | 1959-03-03 | 1961-07-28 | Geigy Ag J R | New sulfamyl-benzamides and their preparation |
| DE2414498A1 (en) * | 1974-03-26 | 1975-10-02 | Heumann Ludwig & Co Gmbh | N-Aminoalkyl-2-alkoxy-5-sulphamoyl-benzamides prepn - from 2-alkoxy-benzoic acids via 5-fluorosulphonyl cpds |
| US4097263A (en) * | 1976-11-22 | 1978-06-27 | Gulf Oil Corporation | 5-Acylamino-1,3,4-thiadiazole-2-sulfonamides and use as herbicides |
| FR2396757A2 (en) * | 1977-07-08 | 1979-02-02 | Ile De France | Psycho-functional alkylene bis:oxy benzamide cpds. - useful in gastroenterology, cardiology, urology, rheumatology and gynaecology |
| GB1561243A (en) * | 1976-03-18 | 1980-02-13 | Richter Gedeon Vegyeszet | Sulphonamides |
| EP0097340A2 (en) * | 1982-06-17 | 1984-01-04 | Ferrer Internacional, S.A. | Piperazine derivatives, a process for preparing them and pharmaceutical compositions |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| IL33030A0 (en) * | 1968-10-03 | 1969-11-30 | Du Pont | 1-carbamoylpyrazole-4-sulfonamides and pharmaceutical compositions containing them |
| BE791501A (en) * | 1971-11-19 | 1973-05-17 | Albert Ag Chem Werke | N, N'-DISUBSTITUTED CYCLIC DIAMINES AND THEIR PREPARATION PROCESS |
| US3865828A (en) * | 1972-03-22 | 1975-02-11 | Egyt Gyogyszervegyeszeti Gyar | Pyridine derivatives having antidepressant activity |
| DE2442851A1 (en) * | 1974-09-06 | 1976-03-18 | Hoechst Ag | BASIC SUBSTITUTED 3-SULPHAMOYLBENZOESIC ACID DERIVATIVES AND PROCESS FOR THEIR PRODUCTION |
| US4386098A (en) * | 1981-11-03 | 1983-05-31 | Merck & Co., Inc. | 6-Hydroxy-2-benzothiazolesulfonamide for the topical treatment of elevated intraocular pressure |
-
1984
- 1984-08-14 IT IT22334/84A patent/IT1176613B/en active
-
1985
- 1985-07-17 EP EP85108942A patent/EP0171636A1/en not_active Withdrawn
- 1985-07-18 US US06/756,257 patent/US4940793A/en not_active Expired - Fee Related
- 1985-08-14 JP JP60177809A patent/JPS6156167A/en active Pending
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR1034958A (en) * | 1950-04-04 | 1953-08-10 | American Cyanamid Co | Improvements in the preparation of heterocyclic sulfonamides |
| FR1267777A (en) * | 1959-03-03 | 1961-07-28 | Geigy Ag J R | New sulfamyl-benzamides and their preparation |
| DE2414498A1 (en) * | 1974-03-26 | 1975-10-02 | Heumann Ludwig & Co Gmbh | N-Aminoalkyl-2-alkoxy-5-sulphamoyl-benzamides prepn - from 2-alkoxy-benzoic acids via 5-fluorosulphonyl cpds |
| GB1561243A (en) * | 1976-03-18 | 1980-02-13 | Richter Gedeon Vegyeszet | Sulphonamides |
| US4097263A (en) * | 1976-11-22 | 1978-06-27 | Gulf Oil Corporation | 5-Acylamino-1,3,4-thiadiazole-2-sulfonamides and use as herbicides |
| FR2396757A2 (en) * | 1977-07-08 | 1979-02-02 | Ile De France | Psycho-functional alkylene bis:oxy benzamide cpds. - useful in gastroenterology, cardiology, urology, rheumatology and gynaecology |
| EP0097340A2 (en) * | 1982-06-17 | 1984-01-04 | Ferrer Internacional, S.A. | Piperazine derivatives, a process for preparing them and pharmaceutical compositions |
Cited By (31)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4804661A (en) * | 1986-06-16 | 1989-02-14 | Ciba-Geigy Corporation | Disubstituted piperazines |
| EP0250361A3 (en) * | 1986-06-16 | 1990-01-31 | Ciba-Geigy Ag | Disubstituted piperazines |
| EP0284359A1 (en) * | 1987-03-24 | 1988-09-28 | Takeda Chemical Industries, Ltd. | 1,4-Disubstituted piperazine compounds, their production and use |
| US4880809A (en) * | 1987-03-24 | 1989-11-14 | Takeda Chemical Industries, Ltd. | 1,4-disubstituted piperazines (or homopiperazines) as platelet-activating factor antagonists |
| AU608580B2 (en) * | 1987-03-24 | 1991-04-11 | Takeda Chemical Industries Ltd. | 1,4-disubstituted piperazine compounds, their production and use |
| EP0548798A1 (en) * | 1991-12-18 | 1993-06-30 | Sanwa Kagaku Kenkyusho Co., Ltd. | Antiviral agent |
| US7046924B2 (en) | 2002-11-25 | 2006-05-16 | Eastman Kodak Company | Method and computer program product for determining an area of importance in an image using eye monitoring information |
| US7327890B2 (en) | 2002-12-20 | 2008-02-05 | Eastman Kodak Company | Imaging method and system for determining an area of importance in an archival image |
| WO2005014563A1 (en) * | 2003-08-11 | 2005-02-17 | F. Hoffmann-La Roche Ag | Piperazine with or-substituted phenyl group and their use as glyt1 inhibitors |
| US7605163B2 (en) | 2003-08-11 | 2009-10-20 | Hoffmann-La Roche Inc. | Benzoyl-piperazine derivatives |
| US7319099B2 (en) | 2003-08-11 | 2008-01-15 | Hoffmann-La Roche Inc. | Benzoyl-piperazine derivatives |
| EA009986B1 (en) * | 2003-08-11 | 2008-04-28 | Ф. Хоффманн-Ля Рош Аг | Piperazine with or-substituted phenyl group and their use as glyt1 inhibitors |
| KR100774622B1 (en) * | 2003-08-11 | 2007-11-08 | 에프. 호프만-라 로슈 아게 | Piperazine with or-substituted phenyl group and their use as glyt1 inhibitors |
| WO2005023260A1 (en) * | 2003-09-09 | 2005-03-17 | F. Hoffmann-La Roche Ag | 1- (2-amino-benzol) -piperazine derivatives as glycine uptake inhibitors for the treatment of psychoses |
| US7595314B2 (en) | 2003-09-09 | 2009-09-29 | Hoffmann-La Roche Inc. | Benzoyl-piperazine derivatives |
| CN1874777B (en) * | 2003-09-09 | 2012-07-04 | 弗·哈夫曼-拉罗切有限公司 | 1-(2-amino-benzoyl)-piperazine derivatives as glycine uptake inhibitors for the treatment of psychoses |
| WO2005023261A1 (en) * | 2003-09-09 | 2005-03-17 | F. Hoffmann-La Roche Ag | 1-benzoyl-piperazine derivatives as glycine uptake inhibitors for the treatment of psychoses |
| KR100774630B1 (en) * | 2003-09-09 | 2007-11-08 | 에프. 호프만-라 로슈 아게 | 1-2-amino-benzol-piperazine derivatives as glycine uptake inhibitors for the treatment of psychoses |
| US7427612B2 (en) | 2003-09-09 | 2008-09-23 | Hoffmann-La Roche Inc. | Benzoyl-piperazine derivatives |
| CN100522171C (en) * | 2003-09-09 | 2009-08-05 | 弗·哈夫曼-拉罗切有限公司 | 1-Benzoylpiperazine derivatives as glycine uptake inhibitors for the treatment of psychosis |
| RU2354653C2 (en) * | 2003-09-09 | 2009-05-10 | Ф.Хоффманн-Ля Рош Аг | 1-(2-aminobenzol)piperazine derivatives as glycin uptake inhibitors to be used for psychosis treatment |
| US7462617B2 (en) | 2003-09-09 | 2008-12-09 | Hoffmann-La Roche Inc. | Substituted acylpiperazine derivatives |
| US7429585B2 (en) | 2004-12-09 | 2008-09-30 | Hoffmann-La Roche | Phenyl-piperazine methanone derivatives, substituted by heterocyclic groups |
| US7241761B2 (en) | 2004-12-09 | 2007-07-10 | Hoffmann-La Roche Inc. | Phenyl-piperazine methanone derivatives, substituted by heterocyclic groups |
| US7375226B2 (en) | 2004-12-15 | 2008-05-20 | Hoffman-La Roche Inc. | Bi- and tricyclic substituted phenyl methanones |
| US7485637B2 (en) | 2005-01-04 | 2009-02-03 | Hoffmann-La Roche Inc. | Benzoyl-tetrahydropiperidine derivatives |
| US7220744B2 (en) | 2005-01-07 | 2007-05-22 | Hoffman-La Roche Inc. | Monocyclic substituted phenyl methanones |
| US7790763B2 (en) | 2005-01-18 | 2010-09-07 | Hoffmann-La Roche Inc. | Substituted phenyl methanone derivatives |
| US7442710B2 (en) | 2005-01-26 | 2008-10-28 | Hoffman-La Roche Inc. | Substituted phenyl methanones |
| US7557114B2 (en) | 2005-02-07 | 2009-07-07 | Hoffman-La Roche Inc. | Heterocyclic-substituted phenyl methanones |
| US8188139B2 (en) | 2005-02-07 | 2012-05-29 | Hoffman-La Roche Inc. | Heterocyclic-substituted phenyl methanones |
Also Published As
| Publication number | Publication date |
|---|---|
| IT1176613B (en) | 1987-08-18 |
| US4940793A (en) | 1990-07-10 |
| IT8422334A0 (en) | 1984-08-14 |
| JPS6156167A (en) | 1986-03-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0171636A1 (en) | Pharmacologically active piperazino derivatives and the process for their preparation | |
| KR100467898B1 (en) | Method for the production of thiazolidin | |
| CA1092111A (en) | Sulfonamido-benzoic acid derivatives and process for the preparation thereof | |
| EP0128007A2 (en) | Phenyl tetrahydronaphthylcarboxylate derivatives | |
| EP0213850B1 (en) | Decyanation of pergolide intermediate | |
| US5084456A (en) | Oxazolopyridine compounds | |
| US3682957A (en) | Method of preparing morpholino cyano-acrylamides | |
| EP0126326B1 (en) | Process for preparing 1,2,4-triazole-3-carboxamides | |
| US3078275A (en) | N, n-disubstituted-alpha-(tertiaryaminoalkyl)-alpha, alpha-diphenylacetamides | |
| EP0644192A1 (en) | A process for making a benzothiadiazole derivative | |
| CA2050492C (en) | Piperazine compounds, processes for preparation thereof and medical uses thereof | |
| SU498907A3 (en) | The method of obtaining phenylimidazolidinone | |
| US4091095A (en) | Phosphinyl compounds | |
| EP0159112A1 (en) | Process for the preparation of diaminopyridines | |
| EP0664799A1 (en) | Amide derivatives | |
| US2753345A (en) | Substituted mercaptobenzoic acids and methods of preparing the same | |
| US3864341A (en) | 3-Morpholino-2-cyanoacrylamide | |
| US4118501A (en) | Thiazolidine derivatives | |
| JPS6355512B2 (en) | ||
| US3305549A (en) | Production of substituted biurets | |
| US4156735A (en) | Thiazolidine derivatives | |
| CS198157B2 (en) | Process for preparing 2-hydroxymethyl-3-hydoxy-6-/1-hydroxy-2-terc.butylaminoethyl/pyridine and additive salt with acid thereof | |
| US4221814A (en) | Terephthalic acid monoamide derivatives, process for preparing the same, and an anti-allergic agent prepared from the same | |
| US3252989A (en) | Pyrazole-1-carboxamidines | |
| US4945161A (en) | Process for the preparation of N,N'-bis-(2-hydroxy-ethyl) piperazine |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Designated state(s): AT BE CH DE FR GB LI LU NL SE |
|
| 17P | Request for examination filed |
Effective date: 19860502 |
|
| 17Q | First examination report despatched |
Effective date: 19871127 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE APPLICATION HAS BEEN WITHDRAWN |
|
| 18W | Application withdrawn |
Withdrawal date: 19901010 |
|
| R18W | Application withdrawn (corrected) |
Effective date: 19901010 |
|
| RIN1 | Information on inventor provided before grant (corrected) |
Inventor name: SIGNORINI, ROBERTO Inventor name: BOTRE, FRANCESCO |