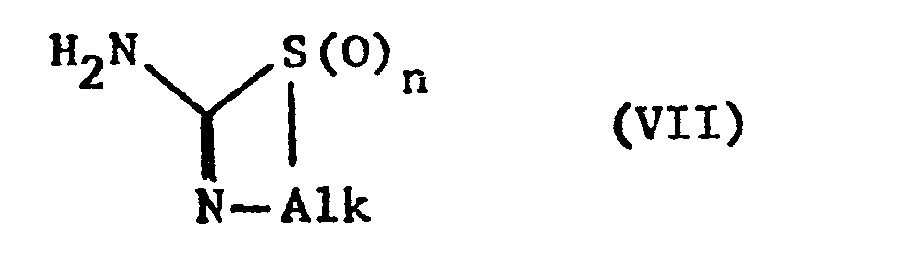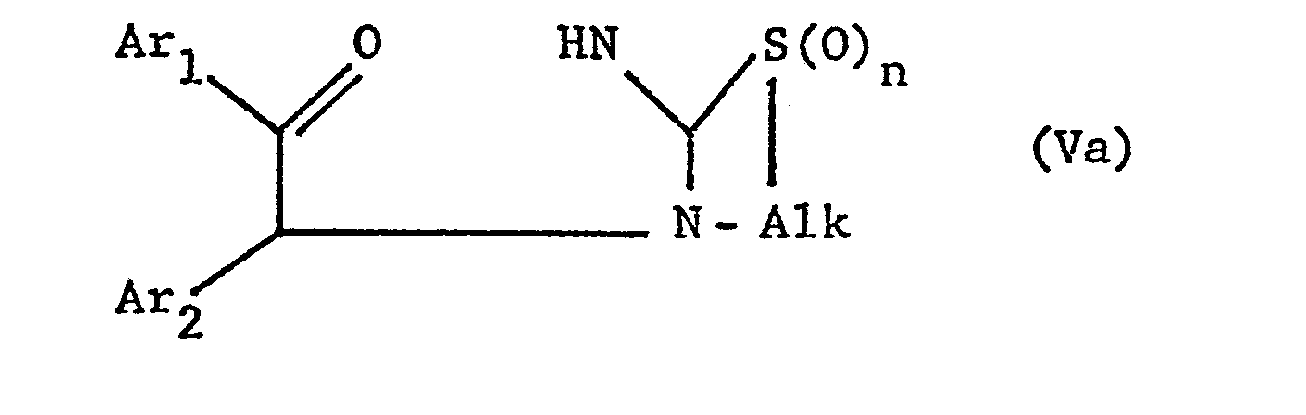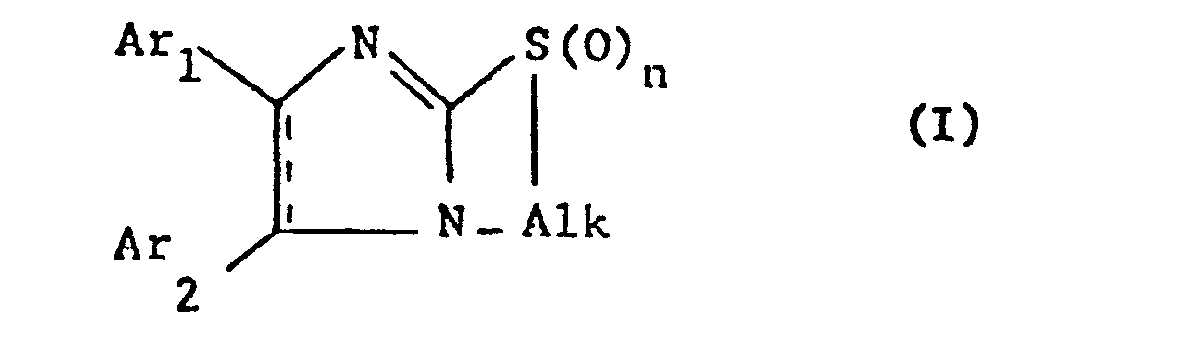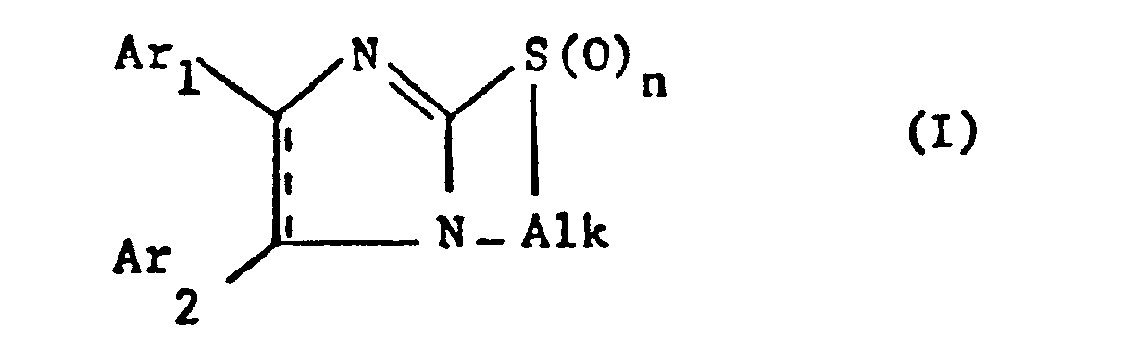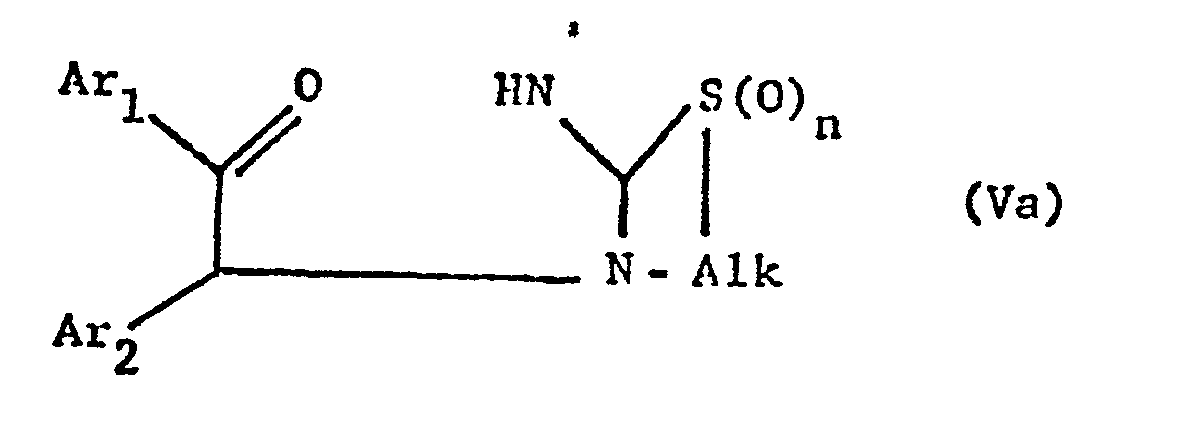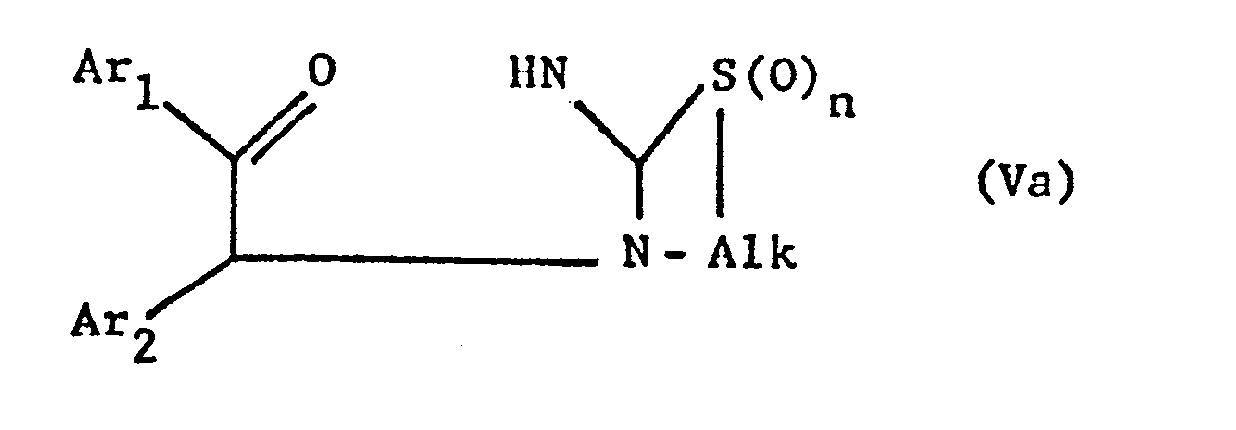EP0000353A2 - Bicyclic thiadiaza compounds, process and intermediates for their preparation, and medicaments containing these compounds or the intermediates - Google Patents
Bicyclic thiadiaza compounds, process and intermediates for their preparation, and medicaments containing these compounds or the intermediates Download PDFInfo
- Publication number
- EP0000353A2 EP0000353A2 EP78100272A EP78100272A EP0000353A2 EP 0000353 A2 EP0000353 A2 EP 0000353A2 EP 78100272 A EP78100272 A EP 78100272A EP 78100272 A EP78100272 A EP 78100272A EP 0000353 A2 EP0000353 A2 EP 0000353A2
- Authority
- EP
- European Patent Office
- Prior art keywords
- salts
- compounds
- phenyl
- thiazole
- imidazo
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
- 150000001875 compounds Chemical class 0.000 title claims abstract description 56
- 238000000034 method Methods 0.000 title claims abstract description 13
- 238000002360 preparation method Methods 0.000 title claims abstract description 7
- 239000003814 drug Substances 0.000 title claims 3
- 125000002619 bicyclic group Chemical group 0.000 title abstract description 3
- 239000000543 intermediate Substances 0.000 title description 4
- 150000003839 salts Chemical class 0.000 claims abstract description 40
- 125000004432 carbon atom Chemical group C* 0.000 claims abstract description 11
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims abstract description 11
- 125000004076 pyridyl group Chemical group 0.000 claims abstract description 11
- 241000534944 Thia Species 0.000 claims abstract description 10
- 125000002947 alkylene group Chemical group 0.000 claims abstract description 9
- 125000001544 thienyl group Chemical group 0.000 claims abstract description 9
- MTNDZQHUAFNZQY-UHFFFAOYSA-N imidazoline Chemical group C1CN=CN1 MTNDZQHUAFNZQY-UHFFFAOYSA-N 0.000 claims abstract description 7
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 claims abstract description 5
- 239000005977 Ethylene Substances 0.000 claims abstract description 4
- 125000002883 imidazolyl group Chemical group 0.000 claims abstract description 4
- -1 thienyl radical Chemical class 0.000 claims description 36
- 239000000203 mixture Substances 0.000 claims description 17
- 125000004172 4-methoxyphenyl group Chemical group [H]C1=C([H])C(OC([H])([H])[H])=C([H])C([H])=C1* 0.000 claims description 13
- 229910052736 halogen Inorganic materials 0.000 claims description 12
- 150000002367 halogens Chemical class 0.000 claims description 12
- OYJGEOAXBALSMM-UHFFFAOYSA-N 2,3-dihydro-1,3-thiazole Chemical compound C1NC=CS1 OYJGEOAXBALSMM-UHFFFAOYSA-N 0.000 claims description 11
- 125000003545 alkoxy group Chemical group 0.000 claims description 11
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 9
- CIUQDSCDWFSTQR-UHFFFAOYSA-N [C]1=CC=CC=C1 Chemical compound [C]1=CC=CC=C1 CIUQDSCDWFSTQR-UHFFFAOYSA-N 0.000 claims description 8
- 125000000217 alkyl group Chemical group 0.000 claims description 7
- 239000000825 pharmaceutical preparation Substances 0.000 claims description 6
- 125000000475 sulfinyl group Chemical group [*:2]S([*:1])=O 0.000 claims description 5
- 125000000472 sulfonyl group Chemical group *S(*)(=O)=O 0.000 claims description 5
- 125000003349 3-pyridyl group Chemical group N1=C([H])C([*])=C([H])C([H])=C1[H] 0.000 claims description 4
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 claims description 4
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 claims description 4
- 125000004429 atom Chemical group 0.000 claims description 4
- 239000000460 chlorine Substances 0.000 claims description 4
- 229910052801 chlorine Inorganic materials 0.000 claims description 4
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 claims description 4
- 229910052757 nitrogen Inorganic materials 0.000 claims description 4
- 125000004433 nitrogen atom Chemical group N* 0.000 claims description 4
- 229910052717 sulfur Inorganic materials 0.000 claims description 4
- 239000011593 sulfur Substances 0.000 claims description 4
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 claims description 4
- AGIJRRREJXSQJR-UHFFFAOYSA-N 2h-thiazine Chemical compound N1SC=CC=C1 AGIJRRREJXSQJR-UHFFFAOYSA-N 0.000 claims description 3
- JLBQKIKVJYJZME-UHFFFAOYSA-N 5,6-bis(3-chlorophenyl)-2,3,5,6-tetrahydroimidazo[2,1-b][1,3]thiazole Chemical compound ClC1=CC=CC(C2C(N3CCSC3=N2)C=2C=C(Cl)C=CC=2)=C1 JLBQKIKVJYJZME-UHFFFAOYSA-N 0.000 claims description 3
- CAUHXBKRLGVONM-UHFFFAOYSA-N 5,6-bis(4-chlorophenyl)-2,3,5,6-tetrahydroimidazo[2,1-b][1,3]thiazole Chemical compound C1=CC(Cl)=CC=C1C1C(C=2C=CC(Cl)=CC=2)N2CCSC2=N1 CAUHXBKRLGVONM-UHFFFAOYSA-N 0.000 claims description 3
- LSXOZUQLGVRCKP-UHFFFAOYSA-N 5,6-bis(4-methylphenyl)-2,3,5,6-tetrahydroimidazo[2,1-b][1,3]thiazole Chemical compound C1=CC(C)=CC=C1C1C(C=2C=CC(C)=CC=2)N2CCSC2=N1 LSXOZUQLGVRCKP-UHFFFAOYSA-N 0.000 claims description 3
- 150000003254 radicals Chemical class 0.000 claims description 3
- 239000000126 substance Substances 0.000 claims description 3
- CBDKQYKMCICBOF-UHFFFAOYSA-N thiazoline Chemical compound C1CN=CS1 CBDKQYKMCICBOF-UHFFFAOYSA-N 0.000 claims description 3
- QQGYDWWMCCPYNZ-UHFFFAOYSA-N 2-(2-imino-1,3-thiazol-3-yl)-1,2-diphenylethanone Chemical compound N=C1SC=CN1C(C=1C=CC=CC=1)C(=O)C1=CC=CC=C1 QQGYDWWMCCPYNZ-UHFFFAOYSA-N 0.000 claims description 2
- QXBWJQICVJKBQI-UHFFFAOYSA-N 5,6-dihydro-4h-thiazin-3-amine Chemical compound NC1=NSCCC1 QXBWJQICVJKBQI-UHFFFAOYSA-N 0.000 claims description 2
- 238000004519 manufacturing process Methods 0.000 claims description 2
- RVARLIQYXFXYNY-UHFFFAOYSA-N 5,6-bis(4-methoxyphenyl)-3-methyl-2,3,5,6-tetrahydroimidazo[2,1-b][1,3]thiazole Chemical compound C1=CC(OC)=CC=C1C1C(C=2C=CC(OC)=CC=2)N2C(C)CSC2=N1 RVARLIQYXFXYNY-UHFFFAOYSA-N 0.000 claims 1
- XRILYHALWKQWIC-UHFFFAOYSA-N 5,6-diphenyl-2,3,5,6-tetrahydroimidazo[2,1-b][1,3]thiazole Chemical compound N1=C2SCCN2C(C=2C=CC=CC=2)C1C1=CC=CC=C1 XRILYHALWKQWIC-UHFFFAOYSA-N 0.000 claims 1
- 230000000144 pharmacologic effect Effects 0.000 abstract description 2
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 48
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 38
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 25
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 18
- 239000000243 solution Substances 0.000 description 18
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 15
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 15
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 15
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 15
- 239000011541 reaction mixture Substances 0.000 description 14
- 239000000725 suspension Substances 0.000 description 13
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 12
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 12
- 239000007858 starting material Substances 0.000 description 12
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 10
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 8
- 238000006243 chemical reaction Methods 0.000 description 8
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 8
- CDBYLPFSWZWCQE-UHFFFAOYSA-L sodium carbonate Substances [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 8
- 229910052938 sodium sulfate Inorganic materials 0.000 description 8
- 235000011152 sodium sulphate Nutrition 0.000 description 8
- 239000002904 solvent Substances 0.000 description 8
- 238000001953 recrystallisation Methods 0.000 description 7
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 6
- MHAJPDPJQMAIIY-UHFFFAOYSA-N Hydrogen peroxide Chemical compound OO MHAJPDPJQMAIIY-UHFFFAOYSA-N 0.000 description 6
- 239000012074 organic phase Substances 0.000 description 6
- 238000010992 reflux Methods 0.000 description 6
- FYSNRJHAOHDILO-UHFFFAOYSA-N thionyl chloride Chemical compound ClS(Cl)=O FYSNRJHAOHDILO-UHFFFAOYSA-N 0.000 description 6
- RAIPHJJURHTUIC-UHFFFAOYSA-N 1,3-thiazol-2-amine Chemical compound NC1=NC=CS1 RAIPHJJURHTUIC-UHFFFAOYSA-N 0.000 description 5
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 5
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 5
- 239000002253 acid Substances 0.000 description 5
- 239000013078 crystal Substances 0.000 description 5
- 239000005457 ice water Substances 0.000 description 5
- 239000003208 petroleum Substances 0.000 description 5
- 238000003756 stirring Methods 0.000 description 5
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 4
- 229920002472 Starch Polymers 0.000 description 4
- 239000012267 brine Substances 0.000 description 4
- 239000012043 crude product Substances 0.000 description 4
- 239000000706 filtrate Substances 0.000 description 4
- 229910000029 sodium carbonate Inorganic materials 0.000 description 4
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical compound O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 4
- 235000019698 starch Nutrition 0.000 description 4
- SZIFAVKTNFCBPC-UHFFFAOYSA-N 2-chloroethanol Chemical compound OCCCl SZIFAVKTNFCBPC-UHFFFAOYSA-N 0.000 description 3
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 3
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 3
- CPELXLSAUQHCOX-UHFFFAOYSA-N Hydrogen bromide Chemical compound Br CPELXLSAUQHCOX-UHFFFAOYSA-N 0.000 description 3
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 3
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 3
- FZWLAAWBMGSTSO-UHFFFAOYSA-N Thiazole Chemical compound C1=CSC=N1 FZWLAAWBMGSTSO-UHFFFAOYSA-N 0.000 description 3
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 3
- 239000004480 active ingredient Substances 0.000 description 3
- 238000001816 cooling Methods 0.000 description 3
- 238000010438 heat treatment Methods 0.000 description 3
- 239000000741 silica gel Substances 0.000 description 3
- 229910002027 silica gel Inorganic materials 0.000 description 3
- 239000011734 sodium Substances 0.000 description 3
- 229910052708 sodium Inorganic materials 0.000 description 3
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 3
- APQIUTYORBAGEZ-UHFFFAOYSA-N 1,1-dibromoethane Chemical compound CC(Br)Br APQIUTYORBAGEZ-UHFFFAOYSA-N 0.000 description 2
- PAAZPARNPHGIKF-UHFFFAOYSA-N 1,2-dibromoethane Chemical compound BrCCBr PAAZPARNPHGIKF-UHFFFAOYSA-N 0.000 description 2
- 125000004105 2-pyridyl group Chemical group N1=C([*])C([H])=C([H])C([H])=C1[H] 0.000 description 2
- REGFWZVTTFGQOJ-UHFFFAOYSA-N 4,5-dihydro-1,3-thiazol-2-amine Chemical compound NC1=NCCS1 REGFWZVTTFGQOJ-UHFFFAOYSA-N 0.000 description 2
- 125000000339 4-pyridyl group Chemical group N1=C([H])C([H])=C([*])C([H])=C1[H] 0.000 description 2
- YYROPELSRYBVMQ-UHFFFAOYSA-N 4-toluenesulfonyl chloride Chemical compound CC1=CC=C(S(Cl)(=O)=O)C=C1 YYROPELSRYBVMQ-UHFFFAOYSA-N 0.000 description 2
- JLJDWUFGUNAJBW-UHFFFAOYSA-N 5,6-bis(4-methoxyphenyl)-2-methyl-2,3,5,6-tetrahydroimidazo[2,1-b][1,3]thiazole Chemical compound C1=CC(OC)=CC=C1C1C(C=2C=CC(OC)=CC=2)N2CC(C)SC2=N1 JLJDWUFGUNAJBW-UHFFFAOYSA-N 0.000 description 2
- AUNNPPFDQHDXHE-UHFFFAOYSA-N 5-(4-chlorophenyl)-6-phenyl-2,3,5,6-tetrahydroimidazo[2,1-b][1,3]thiazole Chemical compound C1=CC(Cl)=CC=C1C1N2CCSC2=NC1C1=CC=CC=C1 AUNNPPFDQHDXHE-UHFFFAOYSA-N 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 2
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 2
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 2
- 108010010803 Gelatin Proteins 0.000 description 2
- WTDHULULXKLSOZ-UHFFFAOYSA-N Hydroxylamine hydrochloride Chemical compound Cl.ON WTDHULULXKLSOZ-UHFFFAOYSA-N 0.000 description 2
- 229930195725 Mannitol Natural products 0.000 description 2
- 241001465754 Metazoa Species 0.000 description 2
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 2
- 206010030113 Oedema Diseases 0.000 description 2
- KFSLWBXXFJQRDL-UHFFFAOYSA-N Peracetic acid Chemical compound CC(=O)OO KFSLWBXXFJQRDL-UHFFFAOYSA-N 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 2
- 235000021355 Stearic acid Nutrition 0.000 description 2
- 229930006000 Sucrose Natural products 0.000 description 2
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 2
- 229960000583 acetic acid Drugs 0.000 description 2
- 239000013543 active substance Substances 0.000 description 2
- 125000004390 alkyl sulfonyl group Chemical group 0.000 description 2
- 239000002260 anti-inflammatory agent Substances 0.000 description 2
- 229940121363 anti-inflammatory agent Drugs 0.000 description 2
- 230000003110 anti-inflammatory effect Effects 0.000 description 2
- 239000002585 base Substances 0.000 description 2
- SRSXLGNVWSONIS-UHFFFAOYSA-N benzenesulfonic acid Chemical compound OS(=O)(=O)C1=CC=CC=C1 SRSXLGNVWSONIS-UHFFFAOYSA-N 0.000 description 2
- 239000012876 carrier material Substances 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 238000004587 chromatography analysis Methods 0.000 description 2
- 238000009833 condensation Methods 0.000 description 2
- 230000005494 condensation Effects 0.000 description 2
- 239000003085 diluting agent Substances 0.000 description 2
- CCIVGXIOQKPBKL-UHFFFAOYSA-M ethanesulfonate Chemical compound CCS([O-])(=O)=O CCIVGXIOQKPBKL-UHFFFAOYSA-M 0.000 description 2
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 2
- 238000001704 evaporation Methods 0.000 description 2
- 230000008020 evaporation Effects 0.000 description 2
- 239000008273 gelatin Substances 0.000 description 2
- 229920000159 gelatin Polymers 0.000 description 2
- 235000019322 gelatine Nutrition 0.000 description 2
- 235000011852 gelatine desserts Nutrition 0.000 description 2
- 239000012362 glacial acetic acid Substances 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-M hydroxide Chemical compound [OH-] XLYOFNOQVPJJNP-UHFFFAOYSA-M 0.000 description 2
- 238000011065 in-situ storage Methods 0.000 description 2
- 150000002576 ketones Chemical class 0.000 description 2
- 231100001231 less toxic Toxicity 0.000 description 2
- 239000000594 mannitol Substances 0.000 description 2
- 235000010355 mannitol Nutrition 0.000 description 2
- 239000000155 melt Substances 0.000 description 2
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 2
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 2
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 2
- 239000003921 oil Substances 0.000 description 2
- 150000007524 organic acids Chemical class 0.000 description 2
- 239000000047 product Substances 0.000 description 2
- 238000006798 ring closing metathesis reaction Methods 0.000 description 2
- JQWHASGSAFIOCM-UHFFFAOYSA-M sodium periodate Chemical compound [Na+].[O-]I(=O)(=O)=O JQWHASGSAFIOCM-UHFFFAOYSA-M 0.000 description 2
- 239000008107 starch Substances 0.000 description 2
- 239000008117 stearic acid Substances 0.000 description 2
- 125000001424 substituent group Chemical group 0.000 description 2
- 239000005720 sucrose Substances 0.000 description 2
- 239000000454 talc Substances 0.000 description 2
- 229910052623 talc Inorganic materials 0.000 description 2
- 235000012222 talc Nutrition 0.000 description 2
- 238000004809 thin layer chromatography Methods 0.000 description 2
- GETQZCLCWQTVFV-UHFFFAOYSA-N trimethylamine Chemical compound CN(C)C GETQZCLCWQTVFV-UHFFFAOYSA-N 0.000 description 2
- SZSPKBXYQNXXBH-UHFFFAOYSA-N 1-(2-bromo-4-methoxyphenyl)-2-(4-methoxyphenyl)ethanone Chemical compound C1=CC(OC)=CC=C1CC(=O)C1=CC=C(OC)C=C1Br SZSPKBXYQNXXBH-UHFFFAOYSA-N 0.000 description 1
- IXPNQXFRVYWDDI-UHFFFAOYSA-N 1-methyl-2,4-dioxo-1,3-diazinane-5-carboximidamide Chemical compound CN1CC(C(N)=N)C(=O)NC1=O IXPNQXFRVYWDDI-UHFFFAOYSA-N 0.000 description 1
- FZKCAHQKNJXICB-UHFFFAOYSA-N 2,1-benzoxazole Chemical compound C1=CC=CC2=CON=C21 FZKCAHQKNJXICB-UHFFFAOYSA-N 0.000 description 1
- JHXOAXMCYMGJLQ-UHFFFAOYSA-N 2,3-dihydro-1,3-thiazol-2-amine Chemical compound NC1NC=CS1 JHXOAXMCYMGJLQ-UHFFFAOYSA-N 0.000 description 1
- RLQZIECDMISZHS-UHFFFAOYSA-N 2-phenylcyclohexa-2,5-diene-1,4-dione Chemical compound O=C1C=CC(=O)C(C=2C=CC=CC=2)=C1 RLQZIECDMISZHS-UHFFFAOYSA-N 0.000 description 1
- 125000000175 2-thienyl group Chemical group S1C([*])=C([H])C([H])=C1[H] 0.000 description 1
- NTYABNDBNKVWOO-UHFFFAOYSA-N 2h-1,3-thiazine Chemical compound C1SC=CC=N1 NTYABNDBNKVWOO-UHFFFAOYSA-N 0.000 description 1
- BMYNFMYTOJXKLE-UHFFFAOYSA-N 3-azaniumyl-2-hydroxypropanoate Chemical compound NCC(O)C(O)=O BMYNFMYTOJXKLE-UHFFFAOYSA-N 0.000 description 1
- LAMUXTNQCICZQX-UHFFFAOYSA-N 3-chloropropan-1-ol Chemical compound OCCCCl LAMUXTNQCICZQX-UHFFFAOYSA-N 0.000 description 1
- IPVFCRUKTBYITA-UHFFFAOYSA-N 4,5-bis[(4-methoxyphenyl)methyl]-1,3-dihydroimidazole-2-thione Chemical compound C1=CC(OC)=CC=C1CC1=C(CC=2C=CC(OC)=CC=2)NC(S)=N1 IPVFCRUKTBYITA-UHFFFAOYSA-N 0.000 description 1
- DNQAQWCFLJOCNU-UHFFFAOYSA-N 4,5-bis[(4-methoxyphenyl)methyl]imidazolidine-2-thione Chemical compound C1=CC(OC)=CC=C1CC1C(CC=2C=CC(OC)=CC=2)NC(=S)N1 DNQAQWCFLJOCNU-UHFFFAOYSA-N 0.000 description 1
- HJYSGEJLUSZCTF-UHFFFAOYSA-N 4,5-diphenylimidazolidine-2-thione Chemical compound N1C(S)=NC(C=2C=CC=CC=2)C1C1=CC=CC=C1 HJYSGEJLUSZCTF-UHFFFAOYSA-N 0.000 description 1
- HVBSAKJJOYLTQU-UHFFFAOYSA-N 4-aminobenzenesulfonic acid Chemical compound NC1=CC=C(S(O)(=O)=O)C=C1 HVBSAKJJOYLTQU-UHFFFAOYSA-N 0.000 description 1
- WUBBRNOQWQTFEX-UHFFFAOYSA-N 4-aminosalicylic acid Chemical compound NC1=CC=C(C(O)=O)C(O)=C1 WUBBRNOQWQTFEX-UHFFFAOYSA-N 0.000 description 1
- PXACTUVBBMDKRW-UHFFFAOYSA-N 4-bromobenzenesulfonic acid Chemical compound OS(=O)(=O)C1=CC=C(Br)C=C1 PXACTUVBBMDKRW-UHFFFAOYSA-N 0.000 description 1
- 125000005274 4-hydroxybenzoic acid group Chemical group 0.000 description 1
- SILRNKZBMNSABG-UHFFFAOYSA-N 4-methyl-4,5-dihydro-1,3-thiazol-2-amine Chemical compound CC1CSC(N)=N1 SILRNKZBMNSABG-UHFFFAOYSA-N 0.000 description 1
- GMZXABSPDNLLLA-UHFFFAOYSA-N 5,6-bis[(4-methoxyphenyl)methyl]-2,3,5,6-tetrahydroimidazo[2,1-b][1,3]thiazole Chemical compound C1=CC(OC)=CC=C1CC1C(CC=2C=CC(OC)=CC=2)N2CCSC2=N1 GMZXABSPDNLLLA-UHFFFAOYSA-N 0.000 description 1
- IKFLLHVVKJTHAH-UHFFFAOYSA-N 5-methyl-4,5-dihydro-1,3-thiazol-2-amine Chemical compound CC1CN=C(N)S1 IKFLLHVVKJTHAH-UHFFFAOYSA-N 0.000 description 1
- 229920001817 Agar Polymers 0.000 description 1
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 1
- 239000005995 Aluminium silicate Substances 0.000 description 1
- 241000416162 Astragalus gummifer Species 0.000 description 1
- LSNNMFCWUKXFEE-UHFFFAOYSA-M Bisulfite Chemical compound OS([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-M 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- COVZYZSDYWQREU-UHFFFAOYSA-N Busulfan Chemical compound CS(=O)(=O)OCCCCOS(C)(=O)=O COVZYZSDYWQREU-UHFFFAOYSA-N 0.000 description 1
- 0 CC(*C(NN*)=C)C(I)=O Chemical compound CC(*C(NN*)=C)C(I)=O 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 1
- 235000005979 Citrus limon Nutrition 0.000 description 1
- 244000131522 Citrus pyriformis Species 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- 208000009386 Experimental Arthritis Diseases 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- 244000151018 Maranta arundinacea Species 0.000 description 1
- 235000010804 Maranta arundinacea Nutrition 0.000 description 1
- ARLZGEXVMUDUQZ-UHFFFAOYSA-N O.O.[Ca] Chemical compound O.O.[Ca] ARLZGEXVMUDUQZ-UHFFFAOYSA-N 0.000 description 1
- 241000283973 Oryctolagus cuniculus Species 0.000 description 1
- 240000007594 Oryza sativa Species 0.000 description 1
- 235000007164 Oryza sativa Nutrition 0.000 description 1
- SCKXCAADGDQQCS-UHFFFAOYSA-N Performic acid Chemical compound OOC=O SCKXCAADGDQQCS-UHFFFAOYSA-N 0.000 description 1
- 201000005702 Pertussis Diseases 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 102000004005 Prostaglandin-endoperoxide synthases Human genes 0.000 description 1
- 108090000459 Prostaglandin-endoperoxide synthases Proteins 0.000 description 1
- 208000010378 Pulmonary Embolism Diseases 0.000 description 1
- 208000025747 Rheumatic disease Diseases 0.000 description 1
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical compound [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 description 1
- 241000779819 Syncarpia glomulifera Species 0.000 description 1
- 235000012419 Thalia geniculata Nutrition 0.000 description 1
- 229920001615 Tragacanth Polymers 0.000 description 1
- 241000209140 Triticum Species 0.000 description 1
- 235000021307 Triticum Nutrition 0.000 description 1
- 240000008042 Zea mays Species 0.000 description 1
- 235000005824 Zea mays ssp. parviglumis Nutrition 0.000 description 1
- 235000002017 Zea mays subsp mays Nutrition 0.000 description 1
- AYLIEDQYYJIGDP-UHFFFAOYSA-N [C]1=CC=CS1 Chemical compound [C]1=CC=CS1 AYLIEDQYYJIGDP-UHFFFAOYSA-N 0.000 description 1
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 239000003463 adsorbent Substances 0.000 description 1
- 239000008272 agar Substances 0.000 description 1
- 235000010419 agar Nutrition 0.000 description 1
- 230000001476 alcoholic effect Effects 0.000 description 1
- 235000010443 alginic acid Nutrition 0.000 description 1
- 239000000783 alginic acid Substances 0.000 description 1
- 229920000615 alginic acid Polymers 0.000 description 1
- 229960001126 alginic acid Drugs 0.000 description 1
- 150000004781 alginic acids Chemical class 0.000 description 1
- 150000001335 aliphatic alkanes Chemical class 0.000 description 1
- 125000001931 aliphatic group Chemical group 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 150000003973 alkyl amines Chemical class 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 235000012211 aluminium silicate Nutrition 0.000 description 1
- SNAAJJQQZSMGQD-UHFFFAOYSA-N aluminum magnesium Chemical compound [Mg].[Al] SNAAJJQQZSMGQD-UHFFFAOYSA-N 0.000 description 1
- 230000000202 analgesic effect Effects 0.000 description 1
- 229940035676 analgesics Drugs 0.000 description 1
- 239000003957 anion exchange resin Substances 0.000 description 1
- 239000000730 antalgic agent Substances 0.000 description 1
- 230000002322 anti-exudative effect Effects 0.000 description 1
- 230000003356 anti-rheumatic effect Effects 0.000 description 1
- 230000002785 anti-thrombosis Effects 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 239000007900 aqueous suspension Substances 0.000 description 1
- 230000002917 arthritic effect Effects 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 229940092714 benzenesulfonic acid Drugs 0.000 description 1
- CSKNSYBAZOQPLR-UHFFFAOYSA-N benzenesulfonyl chloride Chemical compound ClS(=O)(=O)C1=CC=CC=C1 CSKNSYBAZOQPLR-UHFFFAOYSA-N 0.000 description 1
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid group Chemical group C(C1=CC=CC=C1)(=O)O WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 238000009835 boiling Methods 0.000 description 1
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- CJZGTCYPCWQAJB-UHFFFAOYSA-L calcium stearate Chemical compound [Ca+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O CJZGTCYPCWQAJB-UHFFFAOYSA-L 0.000 description 1
- 239000008116 calcium stearate Substances 0.000 description 1
- 235000013539 calcium stearate Nutrition 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 230000003197 catalytic effect Effects 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 239000012230 colorless oil Substances 0.000 description 1
- 235000005822 corn Nutrition 0.000 description 1
- HCAJEUSONLESMK-UHFFFAOYSA-N cyclohexylsulfamic acid Chemical compound OS(=O)(=O)NC1CCCCC1 HCAJEUSONLESMK-UHFFFAOYSA-N 0.000 description 1
- 239000008121 dextrose Substances 0.000 description 1
- 239000002027 dichloromethane extract Substances 0.000 description 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 1
- 239000007884 disintegrant Substances 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 239000012259 ether extract Substances 0.000 description 1
- 125000001033 ether group Chemical group 0.000 description 1
- 125000006125 ethylsulfonyl group Chemical group 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000000284 extract Substances 0.000 description 1
- 239000012065 filter cake Substances 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 235000019634 flavors Nutrition 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 235000003599 food sweetener Nutrition 0.000 description 1
- 238000001640 fractional crystallisation Methods 0.000 description 1
- 239000007903 gelatin capsule Substances 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 229940093915 gynecological organic acid Drugs 0.000 description 1
- 125000000623 heterocyclic group Chemical group 0.000 description 1
- 229910000042 hydrogen bromide Inorganic materials 0.000 description 1
- IXCSERBJSXMMFS-UHFFFAOYSA-N hydrogen chloride Substances Cl.Cl IXCSERBJSXMMFS-UHFFFAOYSA-N 0.000 description 1
- 229910000041 hydrogen chloride Inorganic materials 0.000 description 1
- 239000012433 hydrogen halide Substances 0.000 description 1
- 229910000039 hydrogen halide Inorganic materials 0.000 description 1
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 1
- UVNXNSUKKOLFBM-UHFFFAOYSA-N imidazo[2,1-b][1,3,4]thiadiazole Chemical compound N1=CSC2=NC=CN21 UVNXNSUKKOLFBM-UHFFFAOYSA-N 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 238000000338 in vitro Methods 0.000 description 1
- 239000011261 inert gas Substances 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 239000003978 infusion fluid Substances 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 150000007529 inorganic bases Chemical class 0.000 description 1
- CDAISMWEOUEBRE-GPIVLXJGSA-N inositol Chemical compound O[C@H]1[C@H](O)[C@@H](O)[C@H](O)[C@H](O)[C@@H]1O CDAISMWEOUEBRE-GPIVLXJGSA-N 0.000 description 1
- SUMDYPCJJOFFON-UHFFFAOYSA-N isethionic acid Chemical compound OCCS(O)(=O)=O SUMDYPCJJOFFON-UHFFFAOYSA-N 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 125000004491 isohexyl group Chemical group C(CCC(C)C)* 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Substances [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- 230000008018 melting Effects 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 235000010981 methylcellulose Nutrition 0.000 description 1
- 125000004170 methylsulfonyl group Chemical group [H]C([H])([H])S(*)(=O)=O 0.000 description 1
- 235000013336 milk Nutrition 0.000 description 1
- 239000008267 milk Substances 0.000 description 1
- 210000004080 milk Anatomy 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 239000012452 mother liquor Substances 0.000 description 1
- WHQSYGRFZMUQGQ-UHFFFAOYSA-N n,n-dimethylformamide;hydrate Chemical compound O.CN(C)C=O WHQSYGRFZMUQGQ-UHFFFAOYSA-N 0.000 description 1
- VMOWKUTXPNPTEN-UHFFFAOYSA-N n,n-dimethylpropan-2-amine Chemical compound CC(C)N(C)C VMOWKUTXPNPTEN-UHFFFAOYSA-N 0.000 description 1
- SYSQUGFVNFXIIT-UHFFFAOYSA-N n-[4-(1,3-benzoxazol-2-yl)phenyl]-4-nitrobenzenesulfonamide Chemical class C1=CC([N+](=O)[O-])=CC=C1S(=O)(=O)NC1=CC=C(C=2OC3=CC=CC=C3N=2)C=C1 SYSQUGFVNFXIIT-UHFFFAOYSA-N 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000003136 n-heptyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000006124 n-propyl sulfonyl group Chemical group 0.000 description 1
- PSZYNBSKGUBXEH-UHFFFAOYSA-N naphthalene-1-sulfonic acid Chemical compound C1=CC=C2C(S(=O)(=O)O)=CC=CC2=C1 PSZYNBSKGUBXEH-UHFFFAOYSA-N 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 1
- 231100000252 nontoxic Toxicity 0.000 description 1
- 230000003000 nontoxic effect Effects 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 150000007530 organic bases Chemical class 0.000 description 1
- 230000003204 osmotic effect Effects 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 229940094443 oxytocics prostaglandins Drugs 0.000 description 1
- 238000007911 parenteral administration Methods 0.000 description 1
- KHIWWQKSHDUIBK-UHFFFAOYSA-N periodic acid Chemical compound OI(=O)(=O)=O KHIWWQKSHDUIBK-UHFFFAOYSA-N 0.000 description 1
- 150000002978 peroxides Chemical class 0.000 description 1
- 150000004965 peroxy acids Chemical class 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- WLJVXDMOQOGPHL-UHFFFAOYSA-N phenylacetic acid Chemical compound OC(=O)CC1=CC=CC=C1 WLJVXDMOQOGPHL-UHFFFAOYSA-N 0.000 description 1
- 239000001739 pinus spp. Substances 0.000 description 1
- 208000008423 pleurisy Diseases 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- LPNYRYFBWFDTMA-UHFFFAOYSA-N potassium tert-butoxide Chemical compound [K+].CC(C)(C)[O-] LPNYRYFBWFDTMA-UHFFFAOYSA-N 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 150000003180 prostaglandins Chemical class 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 230000003014 reinforcing effect Effects 0.000 description 1
- 230000000552 rheumatic effect Effects 0.000 description 1
- 201000003068 rheumatic fever Diseases 0.000 description 1
- 206010039073 rheumatoid arthritis Diseases 0.000 description 1
- 235000009566 rice Nutrition 0.000 description 1
- 238000007363 ring formation reaction Methods 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- 238000005245 sintering Methods 0.000 description 1
- 235000010413 sodium alginate Nutrition 0.000 description 1
- 239000000661 sodium alginate Substances 0.000 description 1
- 229940005550 sodium alginate Drugs 0.000 description 1
- 239000012279 sodium borohydride Substances 0.000 description 1
- 229910000033 sodium borohydride Inorganic materials 0.000 description 1
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 description 1
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 description 1
- 239000012312 sodium hydride Substances 0.000 description 1
- 229910000104 sodium hydride Inorganic materials 0.000 description 1
- VGTPCRGMBIAPIM-UHFFFAOYSA-M sodium thiocyanate Chemical compound [Na+].[S-]C#N VGTPCRGMBIAPIM-UHFFFAOYSA-M 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 239000003765 sweetening agent Substances 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 150000003549 thiazolines Chemical class 0.000 description 1
- 229950009928 thiazosulfone Drugs 0.000 description 1
- 125000003396 thiol group Chemical group [H]S* 0.000 description 1
- 239000012485 toluene extract Substances 0.000 description 1
- 235000010487 tragacanth Nutrition 0.000 description 1
- 239000000196 tragacanth Substances 0.000 description 1
- 229940116362 tragacanth Drugs 0.000 description 1
- 229940036248 turpentine Drugs 0.000 description 1
- 239000000052 vinegar Substances 0.000 description 1
- 235000021419 vinegar Nutrition 0.000 description 1
- NLVXSWCKKBEXTG-UHFFFAOYSA-N vinylsulfonic acid Chemical compound OS(=O)(=O)C=C NLVXSWCKKBEXTG-UHFFFAOYSA-N 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- 238000005303 weighing Methods 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/24—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with substituted hydrocarbon radicals attached to ring carbon atoms
- C07D213/44—Radicals substituted by doubly-bound oxygen, sulfur, or nitrogen atoms, or by two such atoms singly-bound to the same carbon atom
- C07D213/53—Nitrogen atoms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/06—Antiarrhythmics
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/24—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with substituted hydrocarbon radicals attached to ring carbon atoms
- C07D213/44—Radicals substituted by doubly-bound oxygen, sulfur, or nitrogen atoms, or by two such atoms singly-bound to the same carbon atom
- C07D213/46—Oxygen atoms
- C07D213/50—Ketonic radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D233/00—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings
- C07D233/54—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having two double bonds between ring members or between ring members and non-ring members
- C07D233/66—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having two double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D233/84—Sulfur atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D277/00—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings
- C07D277/02—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings
- C07D277/08—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member
- C07D277/12—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D277/18—Nitrogen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D277/00—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings
- C07D277/02—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings
- C07D277/20—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D277/32—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D277/38—Nitrogen atoms
- C07D277/40—Unsubstituted amino or imino radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D279/00—Heterocyclic compounds containing six-membered rings having one nitrogen atom and one sulfur atom as the only ring hetero atoms
- C07D279/04—1,3-Thiazines; Hydrogenated 1,3-thiazines
- C07D279/06—1,3-Thiazines; Hydrogenated 1,3-thiazines not condensed with other rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D513/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for in groups C07D463/00, C07D477/00 or C07D499/00 - C07D507/00
- C07D513/02—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for in groups C07D463/00, C07D477/00 or C07D499/00 - C07D507/00 in which the condensed system contains two hetero rings
- C07D513/04—Ortho-condensed systems
Definitions
- the invention relates to new bicyclic thia-diaza compounds, in particular 1,3-diaza-cyclopent-2-eno [2,1-b] (1-thia-3-aza-cycloalkanes) of the general formula (I) whose 1,3-diaza-cyclopent-2-ene ring may have a further double bond,
- Alk represents lower alkylene which separates the thia from the aza atom by 2-4 carbon atoms
- Ar 1 and Ar 2 independently of one another optionally substituted phenyl, Pyridyl or thienyl and n is 0, 1 or 2, with the proviso that at least one of the radicals Ar 1 and Ar 2 is different from phenyl when alk ethylene and the 1,3-diaza-cyclopent-2-ene ring represents an imidazole ring, and their salts, and processes for their preparation, furthermore pharmaceutical preparations containing these compounds and their use, preferably in the form of pharmaceutical preparations.
- radicals and compounds designated "lower" in connection with the present description preferably contain up to 7 and primarily up to 4 carbon atoms.
- Lower alkylene alk is preferably unbranched, but also branched lower alkylene with 2-4 carbon atoms in the chain between the sulfur and the nitrogen atom.
- Pyridyl is a 2-, 3- or 4-pyridyl and thienyl is a 3- or especially 2-thienyl.
- Substituted phenyl, pyridyl or thienyl is e.g. single, double or multiple substituted.
- Substituents, especially on the phenyl radical include Lower alkyl, lower alkoxy, halogen, trifluoromethyl, lower alkylsulfonyl or nitro.
- Substituents on the pyridyl or thienyl radical are preferably lower alkyl, halogen or trifluoromethyl.
- Lower alkyl is e.g. Methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl or tert-butyl, furthermore n-pentyl, n-hexyl, isohexyl or n-heptyl.
- Lower alkylene is ethylene, and also 1,3-propylene, 1,4-butylene, but can also be 1,2-propylene, 1,2- or 2,3-butylene, 1,3- or 2,4-pentylene or 1, Be 4-pentylene.
- Lower alkoxy is e.g. Methoxy, ethoxy, n-propyloxy, isopropyloxy, n-butyloxy or tert-butyloxy.
- Halogen is one with the atomic number up to and 35 and stands for fluorine or bromine, preferably for chlorine.
- Lower alkylsulfonyl is e.g. for methylsulfonyl, ethylsulfonyl or n-propylsulfonyl.
- the compounds according to the invention have valuable pharmacological properties, in particular anti-inflammatory and anti-rheumatic effects, as can be shown in animal experiments. e.g. in the kaolin paw edema test (Helv. Physiol. Acta 25 (1967) 156) on the rat at a dose given from around 10 mg / kg or in the turpentine pleurisy test [Helv. Physiol. Acta 26 (1969) 287) given orally to the rat at a dose of 30 to 100 mg / kg, they show an anti-inflammatory or anti-exudative effect. In particular, the unsaturated compounds also show an excellent effect in the adjuvant arthritis test [Pharmacology 2 (1969) 288] on the rat at a oral dose of 10-30 mg / kg.
- the new compounds are also analgesic, as shown in the phenyl p-benzoquinone test on the mouse (Proc. Soc. Exp. Biol. 95 (1957) 729) at doses of 30 to 100 mg / kg, given orally leaves.
- the tetrahydro compounds additionally show a reinforcing effect in the pertussis edema test (Agents and Actions, vol. 6, 613, 1976) at 5-50 mg / kg / rat.
- the new compounds can therefore be used as anti-inflammatory agents, for example for the treatment of rheumatic, arthritic and other inflammations Diseases in particular, rheumatoid arthritis or as analgesics, for example to treat painful conditions.
- the invention relates in particular to compounds of the formula I in which Ar 1 and Ar 2 independently of one another are a phenyl radical which is optionally substituted by lower alkyl, lower alkoxy, halogen or trifluoromethyl, a pyridyl such as a 2-, 3- or 4-pyridyl or thienyl , in particular represent a 2-thienyl radical, alk is a lower alkylene radical which connects the sulfur and nitrogen atoms via 2-3 carbon atoms, primarily an unbranched lower alkylene radical and n is especially 0, furthermore also 1 or 2, and their salts.
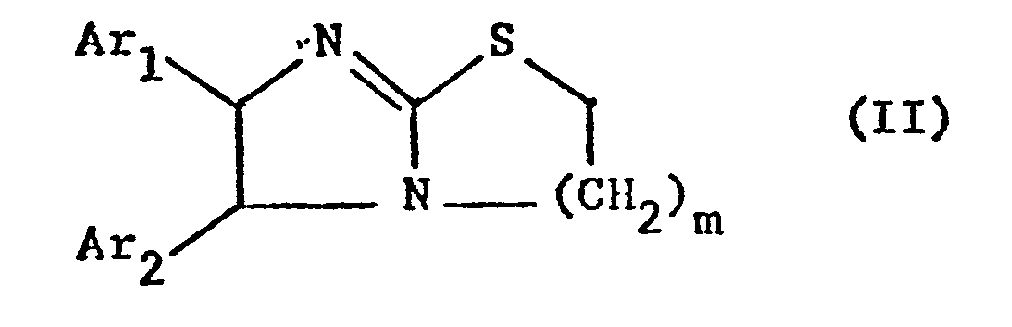
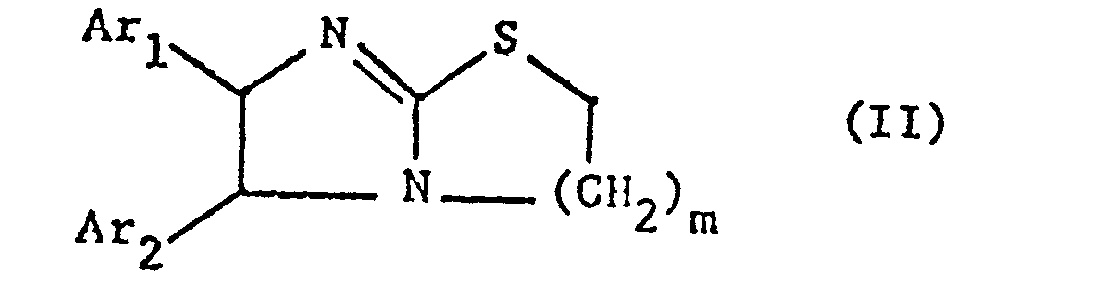
- the invention relates primarily to compounds of the formula II in which Ar 1 and Ar 2 independently of one another represent a phenyl radical which is optionally substituted by lower alkoxy, such as methoxy or halogen, in particular chlorine, and m is primarily 1, furthermore also 2, and their salts.
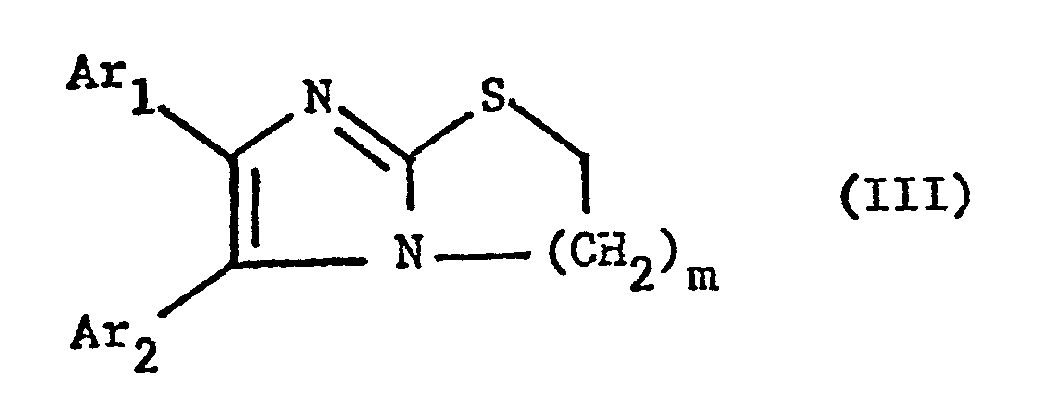
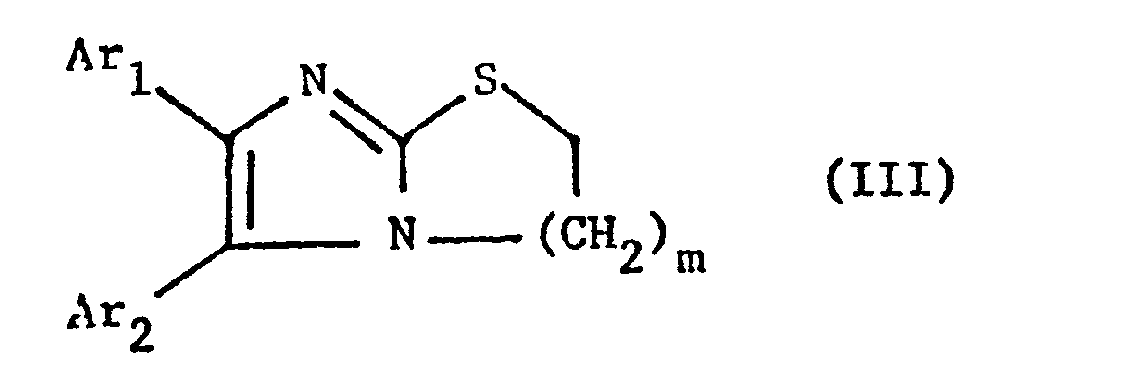
- the invention particularly relates to compounds of the formula III wherein Ar 1 and Ar 2 independently of one another, optionally by lower alkoxy, such as methoxy or halogen, ins special chlorine-substituted phenyl radical and m is primarily 1, furthermore also 2, and their salts.
- the invention relates to the new compounds described in the examples.
- the new compounds can be obtained by methods known per se.
- Reactively esterified hydroxy is especially one with a strong inorganic acid, e.g. Hydrogen halide, especially hydrogen chloride, or sulfuric acid, or with a strong organic acid, such as with a lower alkanesulfonic acid, e.g. Methane or ethanesulfonic acid or a benzenesulfonic acid optionally substituted by lower alkyl, lower alkoxy or halogen, e.g. p-Toluenesulfonic acid or p-bromobenzenesulfonic acid esterified hydroxy group.
- a strong inorganic acid e.g. Hydrogen halide, especially hydrogen chloride, or sulfuric acid
- a strong organic acid such as with a lower alkanesulfonic acid, e.g. Methane or ethanesulfonic acid or a benzenesulfonic acid optionally substituted by lower alkyl, lower alkoxy or halogen,
- the ring closure is preferably carried out under acid-releasing conditions. This is done primarily in a low-boiling solvent, such as dimethylformamide or acetone, an alcohol, e.g. Methanol or ethanol, if desired, in the presence of a base, e.g. an inorganic base, such as an alkali or alkaline earth hydroxide, hydroxide or carbonate, primarily sodium hydride, sodium hydroxide or sodium carbonate, or an organic base, preferably a nitrogen base, such as tri-lower alkylamine, e.g. Trimethylamine, triethylamine, dimethylisopropylamine, or pyridine.
- a base e.g. an inorganic base, such as an alkali or alkaline earth hydroxide, hydroxide or carbonate, primarily sodium hydride, sodium hydroxide or sodium carbonate, or an organic base, preferably a nitrogen base, such as tri-lower alkylamine, e.g. Trimethyl
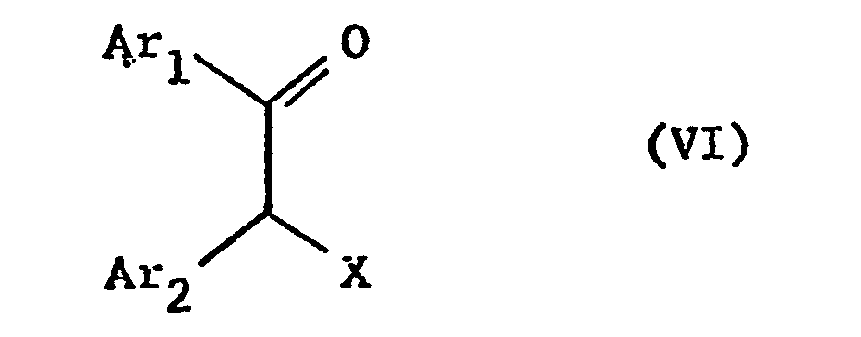
- the starting materials can be obtained if a corresponding diaza-2-mercapto compound is reacted with a dihydroxyalkylene in which at least one of the two hydroxyl groups has been reactively esterified, if appropriate the mercapto group is oxidized to the sulfinyl or sulfonyl group and then, if necessary, the second hydroxyl group reactively esterified.
- the reactive esterified hydroxyl groups correspond to the above-mentioned conditions as well as the condensation conditions. In this reaction, in particular if both hydroxyl groups are esterified in a reactive manner, the starting materials of the formula IV can be obtained in situ, which are closed to form a ring in the same reaction.
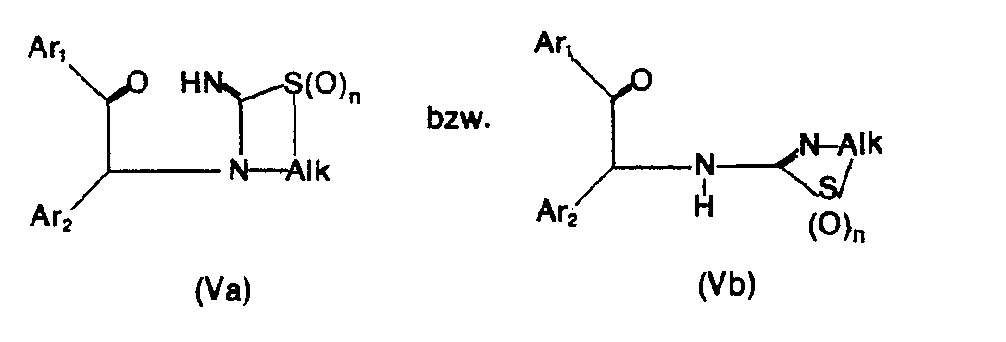
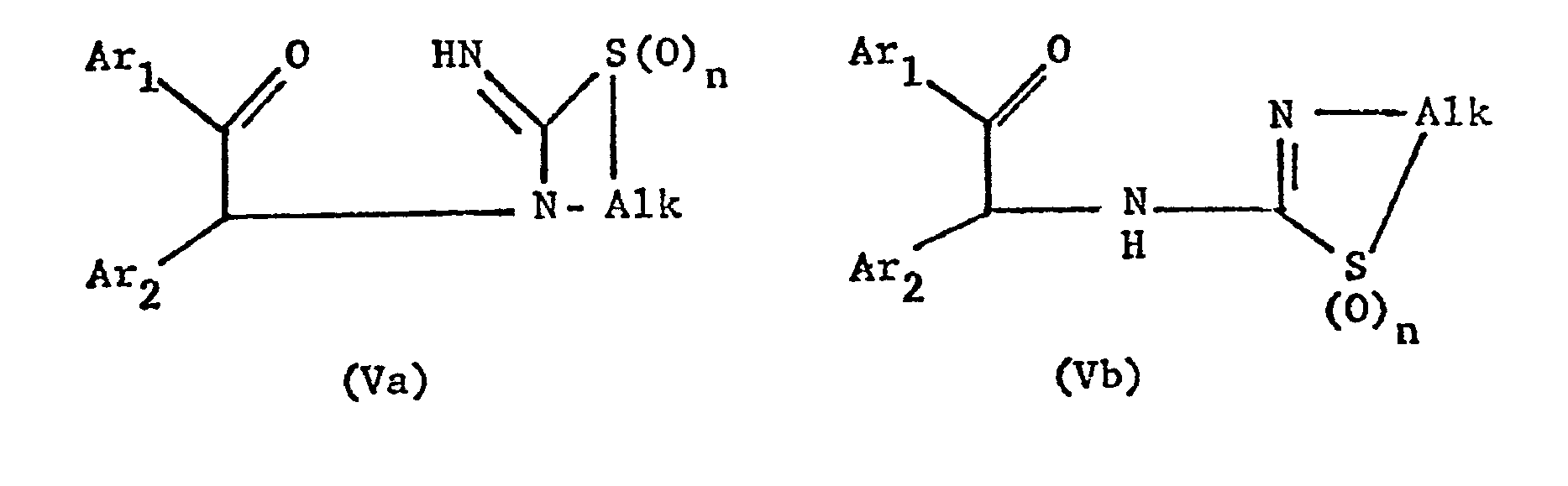
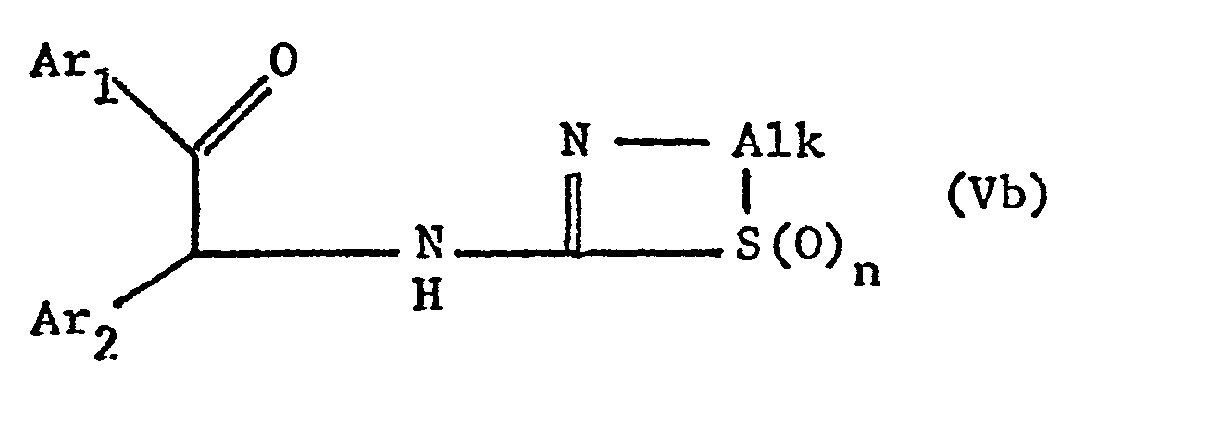
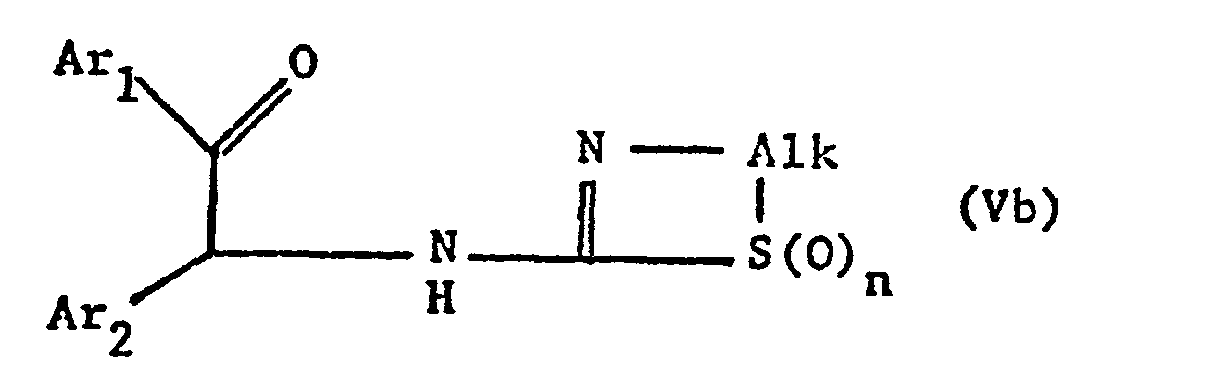
- the new compounds of the formula I which have a further double bond can also be obtained if compounds of the formula Va or Vb or ring-closes their tautomers and, if desired, in optionally obtained compounds in which n is 0, the thia atom is oxidized to the sulfinyl or sulfonyl group and / or, if desired, converts the free compounds obtained into their salts or salts obtained into the free ones Converting compounds and / or separating an isomer mixture obtained according to the process into the individual isomers.
- the ring closure takes place under water-releasing conditions, such as by heating, e.g. from about 50 ° to about 150 °, preferably in the presence of a solvent such as acetonitrile or an alcohol, e.g. Methanol or ethanol.
- a solvent such as acetonitrile or an alcohol, e.g. Methanol or ethanol.
- the starting materials can be obtained if a compound of the formula VI with a compound of formula VII or their tautomers.
- Reactively esterified hydroxy groups X are in particular the above.
- this reaction is also carried out under water-releasing conditions, for example with heating in a solvent such as acetonitrile or an alcohol, the starting material of the formula Va or Vb can be obtained in situ, which closes under these reaction conditions.
- a solvent such as acetonitrile or an alcohol
- the oxidation of the thia atom to the sulfinyl or sulfonyl group can be carried out in a manner known per se, e.g. with peroxides such as hydrogen peroxide or peracids e.g. carry out an optionally substituted by lower alkyl, lower alkoxy, halogen or another carboxyl group, such as benzoperic acid itself or phthalmonoperacid, or an alkane percarboxylic acid such as peracetic acid or a periodate such as sodium periodate.
- This reaction is usually carried out at low temperatures in a solvent such as glacial acetic acid or acetone.
- the new compounds can be in the form of acid addition salts, especially pharmaceutically acceptable, non-toxic salts, e.g. with organic acids, such as hydrochloric, hydrobromic, sulfuric or phosphoric acid, or with organic, such as aliphatic, cycloaliphatic, cycloaliphatic-aliphatic, aromatic, araliphatic, heterocyclic or heterocyclic-aliphatic carbon or sulfonic acid, e.g.
- organic acids such as hydrochloric, hydrobromic, sulfuric or phosphoric acid
- organic such as aliphatic, cycloaliphatic, cycloaliphatic-aliphatic, aromatic, araliphatic, heterocyclic or heterocyclic-aliphatic carbon or sulfonic acid, e.g.
- Salts of this type can e.g. can be obtained by treating the free compounds, with the acids or with suitable anion exchange resins.
- the free compounds or the salts are also to be understood as meaningful and expedient, if appropriate, the corresponding salts or free compounds.
- the new compounds can be present as isomer mixtures, such as racemates or diastereoisomer mixtures, or in the form of the pure isomers, such as optically active components.
- the separation of isomer mixtures obtained into the pure isomers can be carried out by the known methods. Racemates can e.g. due to physico-chemical differences, e.g. Separate those of solubility, their diastereomeric salts, or by fractional crystallization from an optically active solvent, or by chromatography, in particular thin-layer chromatography, on an optically active carrier material, into the optically active antipodes.
- the pharmacologically more effective or less toxic pure isomer is advantageously isolated, in particular the more effective or less toxic active antipode.
- the process also includes those embodiments according to which compounds obtained as intermediates are used as starting materials and the remaining process steps are carried out with them, or the process is terminated at any stage; furthermore, starting materials in the form of derivatives can be used or formed during the reaction.
- the invention also relates to the new compounds of the general formula Va or Vb obtainable as intermediates or. or their tautomers, where Alk is lower alkylene, which separates the thia from the aza atom by 2-4 carbon atoms, Ar 1 and Ar 2 independently of one another are optionally substituted phenyl, pyridyl or thienyl and n is 0, 1 or 2, and their Salts.
- Alk is lower alkylene, which separates the thia from the aza atom by 2-4 carbon atoms
- Ar 1 and Ar 2 independently of one another are optionally substituted phenyl, pyridyl or thienyl and n is 0, 1 or 2, and their Salts.
- they are approximately as effective as the dihydro compounds of the formula I and can be used as anti-inflammatory agents, for example for the treatment of rheumatic arthritis.
- the new compounds of the present invention can be used, for example, for the production of pharmaceutical preparations which contain an effective amount of the active substance together or in a mixture with inorganic or contain organic, solid or liquid, pharmaceutically usable excipients which are suitable for enteral or parenteral administration.
- tablets or gelatin capsules which contain the active ingredient together with diluents, for example lactose, dextrose, sucrose, mannitol, sorbitol, cellulose and / or glycerol, and lubricants, for example silica, talc, stearic acid or salts thereof, such as magnesium or calcium stearate. and / or polyethylene glycol; Tablets also contain binders, e.g.
- magnesium aluminum silicate starches such as corn, wheat, rice or arrowroot starch, gelatin, tragacanth, methyl cellulose, sodium carboxymethyl cellulose and / or polyvinylpyrrolidone, and, if desired, disintegrants, e.g. starches, agar, alginic acid or a salt thereof, such as sodium alginate, and / or effervescent mixtures, or adsorbents, colors, flavors and sweeteners.
- disintegrants e.g. starches, agar, alginic acid or a salt thereof, such as sodium alginate, and / or effervescent mixtures, or adsorbents, colors, flavors and sweeteners.
- the new pharmacologically active compounds can be used in the form of injectable, for example intravenously administrable, preparations or infusion solutions.
- Such solutions are preferably isotonic aqueous solutions or suspensions, these being able to be prepared, for example, from lyophilized preparations which contain the active substance on their own or together with a carrier material, for example mannitol.
- the pharmaceutical preparations can be sterilized and / or contain auxiliaries, for example preservatives, stabilizers, wetting agents and / or emulsifiers, solubilizers, salts for regulating the osmotic pressure and / or buffers.
- the present pharmaceutical preparations which, if desired, may contain other pharmacologically valuable substances, are produced in a manner known per se, for example by means of conventional mixing, granulating, confectioning, dissolving or lyophilizing processes and contain from about 0.1% to 100%, in particular from about 1% to about 50%, of lyophilisates up to 100% of the active ingredient.
- the single dose for a warm-blooded animal weighing approximately 70 kg is between 0.1 and 0.75 g, the daily dose between 0.2 and 1.0 g.
- a suspension of 7 ml of 1,2-dibromoethane, 7 g of sodium carbonate and 55 ml of isopropanol is stirred at room temperature and within one hour with the suspension of 4.7 g of 4,5-diphenyl-imidazolidin-2-thione in 110 ml of l, 5% sodium hydroxide solution added.
- the reaction mixture is refluxed for 7 hours, then the isopropanol and the dibromoethane are removed on a rotary evaporator and the remaining suspension is extracted with toluene.
- the toluene extract is washed with brine, dried over sodium sulfate and evaporated. The residue is chromatographed on silica gel.
- a suspension of 3.5 ml of 1,2-dibromoethane, 3.5 g of sodium carbonate and 30 ml of isopropanol is stirred in with the suspension of 3 g of 4,5-di-anisyl-2-mercapto-imidazole in at room temperature for one hour 50 ml of 1.5% sodium hydroxide solution are added.
- the reaction mixture is refluxed for 6 hours, then the isopropanol and the dibromoethane are removed on a rotary evaporator and the remaining suspension is mixed with 10 ml of 20% potassium hydroxide solution and extracted with ethyl acetate. The organic phases are washed with brine, dried with sodium sulfate and evaporated.
- the starting material can e.g. are prepared: 2.5 g of sodium are dissolved in 220 ml of ethanol. 35 g of 4,5-dianisyl-imidazolin-2-thione are added. A suspension is obtained. For this purpose, 15.7 ml of 2-chloroethanol are added dropwise at room temperature (stirring). The mixture is stirred for 1 hour at 60 ° and 4 hours at reflux. The weak suspension is then evaporated to dryness. The residue is purified from acetone / water.
- the starting material used can e.g. received as follows:
- the 2-mercapto-4,5-di-p-chlorophenylimidazole with 2-chloroethanol is the 2- (2-hydroxyethylthio) -4,5-di-p-chlorophenylimidazole from F. 197-199 ° and from it by cyclization with thionyl chloride the 5,6-di-p-chlorophenyl-imidazo [2,1-b] dihydro-thiazole of mp 199-204 °.
- the 5,6-di-p-methoxyphenyl-4H-imidazo [2,1-b] dihydro-thiazole can also be prepared analogously.
- the hydrobromide of 5,6-di- (p-methoxyphenyl) imidazo [2, l-b] dihydro-thiazole can be obtained from the mother liquor, mp. 200-210 °.
- the L-5,6-di-p-anisyl-2,3,5,6-tetrahydro-4H-imidazo [2,1-b] thiazole N-benzenesulfonyl L (+) - glutamate is filtered off with a little Washed water, suspended in water and decomposed with dilute sodium hydroxide solution. From this, the L-5,6-di-p-anisyl-2,3,5,6-tetrahydro-4H-imidazo [2,1-b] thiazole is extracted with ethyl acetate.
- sucrose, calcium dihydrate and the active ingredient are mixed and granulated with a 10% gelatin solution.
- the moist granules are sieved, dried, mixed with the starch, talc and stearic acid, sieved and compressed into tablets.
Landscapes
- Organic Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Veterinary Medicine (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Life Sciences & Earth Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Heart & Thoracic Surgery (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Cardiology (AREA)
- Pain & Pain Management (AREA)
- Rheumatology (AREA)
- Nitrogen And Oxygen Or Sulfur-Condensed Heterocyclic Ring Systems (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Thiazole And Isothizaole Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
Abstract
Bicyclische Thia-diaza-verbindungen, insbesondere 1,3-Diaza-cyclopent-2-eno[2,1-b]-(1-thia-3-aza-cycloal- kane) der allgemeinen Formel
Die neuen Verbindungen I und Zwischenverbindungen der Formel Va bzw. Vb
Description
Die Erfindung betrifft neue bicyclische Thia-diaza-verbindungen, insbesondere 1,3-Diaza-cyclopent-2-eno [2,1-b](1-thia-3-aza-cycloalkane) der allgemeinen Formel (I)
Die im Zusammenhang mit der vorliegenden Beschreibung mit "nieder" bezeichneten Reste und Verbindungen enthalten vorzugsweise bis 7 und in erster Linie bis 4 Kohlenstoffatome.The radicals and compounds designated "lower" in connection with the present description preferably contain up to 7 and primarily up to 4 carbon atoms.
Niederalkylen Alk ist vorzugsweise unverzweigtes, aber auch verzweigtes Niederalkylen mit 2-4 Kohlenstöffatomen in der Kette zwischen dem Schwefel- und dem Stickstoffatom.Lower alkylene alk is preferably unbranched, but also branched lower alkylene with 2-4 carbon atoms in the chain between the sulfur and the nitrogen atom.
Pyridyl ist ein 2-, 3- oder 4-Pyridyl und Thienyl ein 3- oder insbesondere 2-Thienyl.Pyridyl is a 2-, 3- or 4-pyridyl and thienyl is a 3- or especially 2-thienyl.
Substituiertes Phenyl, Pyridyl oder Thienyl ist z.B. einfach, zweifach oder auch mehrfach substituiert. Substituenten, insbesondere am Phenylrest sind u.a. Niederalkyl, Niederalkoxy, Halogen, Trifluormethyl, Niederalkylsulfonyl oder Nitro. Substituenten am Pyridyl- oder Thienyl-rest sind vorzugsweise Niederalkyl, Halogen oder Trifluormethyl.Substituted phenyl, pyridyl or thienyl is e.g. single, double or multiple substituted. Substituents, especially on the phenyl radical, include Lower alkyl, lower alkoxy, halogen, trifluoromethyl, lower alkylsulfonyl or nitro. Substituents on the pyridyl or thienyl radical are preferably lower alkyl, halogen or trifluoromethyl.
Vorstehend wie nachfolgend können die Allgemeinbegriffe folgende Bedeutung haben:Above and below, the general terms can have the following meanings:
Niederalkyl ist z.B. Methyl, Aethyl, n-Propyl, Isopropyl, n-Butyl, Isobutyl, sec.-Butyl oder tert.-Butyl, ferner n-Pentyl, n-Hexyl, Isohexyl oder n-Heptyl.Lower alkyl is e.g. Methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl or tert-butyl, furthermore n-pentyl, n-hexyl, isohexyl or n-heptyl.
Niederalkylen ist Aethylen, sowie 1,3-Propylen, 1,4-Butylen, kann aber auch 1,2-Propylen, 1,2- oder 2,3-Butylen, 1,3- oder 2,4-Pentylen oder 1,4-Pentylen sein.Lower alkylene is ethylene, and also 1,3-propylene, 1,4-butylene, but can also be 1,2-propylene, 1,2- or 2,3-butylene, 1,3- or 2,4-pentylene or 1, Be 4-pentylene.
Niederalkoxy ist z.B. Methoxy, Aethoxy, n-Propyloxy, Isopropyloxy, n-Butyloxy oder tert.-Butyloxy.Lower alkoxy is e.g. Methoxy, ethoxy, n-propyloxy, isopropyloxy, n-butyloxy or tert-butyloxy.
Halogen ist solches mit der Atomnummer bis und mit 35 und steht fUr Fluor oder Brom, vorzugsweise fUr Chlor.Halogen is one with the atomic number up to and 35 and stands for fluorine or bromine, preferably for chlorine.
Niederalkylsulfonyl steht z.B. für Methylsulfonyl, Aethylsulfonyl oder n-Propylsulfonyl.Lower alkylsulfonyl is e.g. for methylsulfonyl, ethylsulfonyl or n-propylsulfonyl.
Die erfindungsgemässen Verbindungen besitzen wertvolle pharmakologische Eigenschaften, insbesondere entzündungshemmende und antirheumatische Wirkungen, wie sich in Tierversuchen zeigen lässt. z.B. im Kaolin-Pfotenoedem-Test (Helv. Physiol. Acta 25 (1967) 156) an der Ratte bei einer peroral gegebenen Dosis ab etwa 10 mg/kg oder im Terpentin-Pleuritis-Test [Helv. Physiol. Acta 26 (1969) 287) an der Ratte peroral gegeben bei einer Dosis von 30 bis 100 mg/kg zeigen sie eine antiinflammatorische bzw. anti-exsudative Wirkung. Insbesondere die ungesättigten Verbindungen zeigen auch im AdjuvansArthritis-Test [Pharmacology 2 (1969) 288] an der Ratte bei einer peroralen Dosis von 10-30 mg/kg eine ausgezeichnete Wirkung.The compounds according to the invention have valuable pharmacological properties, in particular anti-inflammatory and anti-rheumatic effects, as can be shown in animal experiments. e.g. in the kaolin paw edema test (Helv. Physiol. Acta 25 (1967) 156) on the rat at a dose given from around 10 mg / kg or in the turpentine pleurisy test [Helv. Physiol. Acta 26 (1969) 287) given orally to the rat at a dose of 30 to 100 mg / kg, they show an anti-inflammatory or anti-exudative effect. In particular, the unsaturated compounds also show an excellent effect in the adjuvant arthritis test [Pharmacology 2 (1969) 288] on the rat at a oral dose of 10-30 mg / kg.
Die neuen Verbindungen sind auch analgetisch wirksam, wie sich im Phenyl-p-benzochinon-Test an der Maus (Proc. Soc. Exp. Biol. 95 (1957) 729) bei Dosen von 30 bis 100 mg/kg, peroral gegeben, zeigen lässt.The new compounds are also analgesic, as shown in the phenyl p-benzoquinone test on the mouse (Proc. Soc. Exp. Biol. 95 (1957) 729) at doses of 30 to 100 mg / kg, given orally leaves.
Ferner ist die Hemmwirkung der neuen Präparate auf die Prostaglandin-Synthetase in vitro [Prostaglandins, 7 (1974) 123] in Konzentrationen von 0.05-20 µg/ml zu nennen. Ausserdem zeigen sie einen wertvollen antithrombotischen Effekt, nämlich einen Schutz vor tödlicher Lungenembolie am Kaninchen [Pharmacology 14 (1976) 522] in peroralen Dosen von 0.03-3 mg/kg.Furthermore, the inhibitory effect of the new preparations on prostaglandin synthetase in vitro [Prostaglandins, 7 (1974) 123] in concentrations of 0.05-20 µg / ml should be mentioned. In addition, they show a valuable antithrombotic effect, namely protection against fatal pulmonary embolism in rabbits [Pharmacology 14 (1976) 522] in oral doses of 0.03-3 mg / kg.
Die Tetrahydro-verbindungen zeigen zusätzlich dazu im Pertussisoedem-Test (Agents and Actions, vol. 6, 613, 1976) bei 5-50 mg/kg/Ratte eine verstärkende Wirkung.The tetrahydro compounds additionally show a reinforcing effect in the pertussis edema test (Agents and Actions, vol. 6, 613, 1976) at 5-50 mg / kg / rat.
Die neuen Verbindungen können desshalb als Antiphlogistika z.B. zur Behandlung von rheumatischen, arthritischen und anderen, mit EntzUndungen verbundenen Erkrankungen insbesondere, rheumatischer Arthritis oder als Analgetika, z.B. zur Behandlung von Schmerzzuständen, verwendet werden.The new compounds can therefore be used as anti-inflammatory agents, for example for the treatment of rheumatic, arthritic and other inflammations Diseases in particular, rheumatoid arthritis or as analgesics, for example to treat painful conditions.
Die Erfindung betrifft insbesondere Verbindungen der Formel I, worin Ar1 und Ar2 unabhängig voneinander einen gegebenenfalls durch Niederalkyl, Niederalkoxy, Halogen oder Trifluormethyl substituierten Phenylrest, einen Pyridyl-, wie einen 2-, 3- oder 4-Pyridyl-,oder Thienyl-, ins besondere einen 2-Thienylrest darstellen, Alk einen Niederalkylenrest, der das Schwefel- und Stickstoffatom Uber 2-3 Kohlenstoffatome miteinander verbindet, in erster Linie einen unverzweigten Niederalkylenrest und n besonders 0, ferner auch 1 oder 2 bedeuten, und deren Salze.The invention relates in particular to compounds of the formula I in which Ar 1 and Ar 2 independently of one another are a phenyl radical which is optionally substituted by lower alkyl, lower alkoxy, halogen or trifluoromethyl, a pyridyl such as a 2-, 3- or 4-pyridyl or thienyl , in particular represent a 2-thienyl radical, alk is a lower alkylene radical which connects the sulfur and nitrogen atoms via 2-3 carbon atoms, primarily an unbranched lower alkylene radical and n is especially 0, furthermore also 1 or 2, and their salts.
Die Erfindung betrifft in erster Linie Verbindungen der Formel II
Die Erfindung betrifft aber besonders auch Verbindungen der Formel III
Insbesondere betrifft uie Erfindung die neuen in den Beispielen beschriebenen Verbindungen.In particular, the invention relates to the new compounds described in the examples.
Die neuen Verbindungen lassen sich nach an sich bekannten Methoden gewinnen.The new compounds can be obtained by methods known per se.
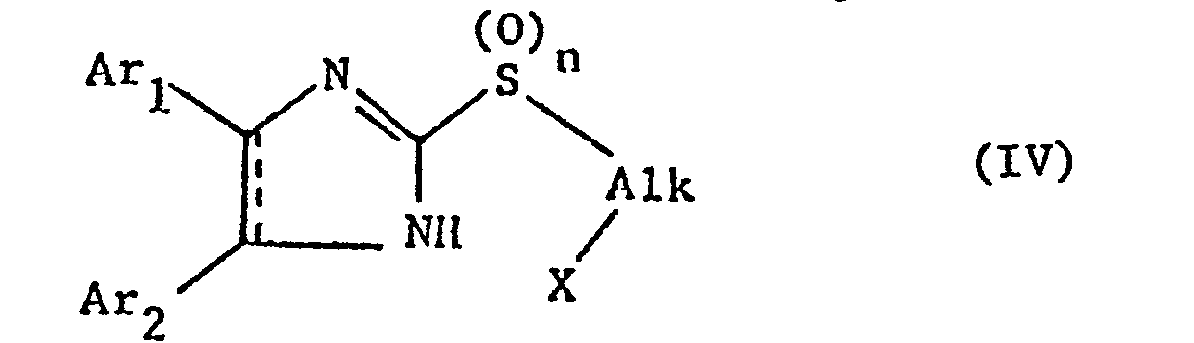
So kann man z.B. Verbindungen der Formel IV
Reaktionsfähig verestertes Hydroxy ist insbesondere eine mit einer starken anorganischen Säure, z.B. Halogenwasserstoff, insbesondere Chlorwasserstoff, oder Schwefelsäure, oder mit einer starken organischen Säure, wie mit einer Niederalkansulfonsäure, z.B. Methan- oder Aethansulfonsäure oder einer gegebenenfalls durch Niederalkyl, Niederalkoxy oder Halogen substituierten Benzolsulfonsäure, z.B. p-Toluolsulfonsäure oder p-Brombenzolsulfonsäure veresterte Hydroxygruppe.Reactively esterified hydroxy is especially one with a strong inorganic acid, e.g. Hydrogen halide, especially hydrogen chloride, or sulfuric acid, or with a strong organic acid, such as with a lower alkanesulfonic acid, e.g. Methane or ethanesulfonic acid or a benzenesulfonic acid optionally substituted by lower alkyl, lower alkoxy or halogen, e.g. p-Toluenesulfonic acid or p-bromobenzenesulfonic acid esterified hydroxy group.
Der Ringsschluss wird vorzugsweise unter Säureabspaltenden Bedingungen durchgeführt. Dabei arbeitet man in erster Linie in einem niedersiedenden Lösungsmittel, wie Dimethylformamid oder Aceton, einem Alkohol, z.B. Methanol oder Aethanol, wenn erwünscht, in Gegenwart einer Base, z.B. einer anorganischen Base, wie einem Alkali-oder Erdalkalihydrid, -hydroxyd oder -carbonat, in erster Linie Natriumhydrid, Natriumhydroxyd oder Natriumcarbonat, oder einer organischen Base, vorzugsweise einer Stickstoffbase, wie Triniederalkylamin, z.B. Trimethylamin, Triäthylamin, Dimethyl-isopropylamin, oder Pyridin.The ring closure is preferably carried out under acid-releasing conditions. This is done primarily in a low-boiling solvent, such as dimethylformamide or acetone, an alcohol, e.g. Methanol or ethanol, if desired, in the presence of a base, e.g. an inorganic base, such as an alkali or alkaline earth hydroxide, hydroxide or carbonate, primarily sodium hydride, sodium hydroxide or sodium carbonate, or an organic base, preferably a nitrogen base, such as tri-lower alkylamine, e.g. Trimethylamine, triethylamine, dimethylisopropylamine, or pyridine.
Die Ausgangsstoffe lassen sich erhalten, wenn man eine ensprechende Diaza-2-mercapto-verbindung mit einem Dihydroxyalkylen, worin mindestens eine der beiden Hydroxygruppen reaktionsfähig verestert ist, miteinander umsetzt, gegebenenfalls die Mercaptogruppe zur Sulfinyl-oder Sulfonylgruppe oxidiert und anschliessend, falls notwendig, die zweite Hydroxygruppe reaktionsfähig verestert. Die reaktionsfähig veresterten Hydroxygruppen entsprechen den obgenannten wie auch die Bedingungen der Kondensation. Bei dieser Reaktion können, insbesondere wenn beide Hydroxylgruppen reaktionsfähig verestert sind, die Ausgangsstoffe der Formel IV in situ erhalten werden, welche in der gleichen Reaktion zum Ring geschlossen werden.The starting materials can be obtained if a corresponding diaza-2-mercapto compound is reacted with a dihydroxyalkylene in which at least one of the two hydroxyl groups has been reactively esterified, if appropriate the mercapto group is oxidized to the sulfinyl or sulfonyl group and then, if necessary, the second hydroxyl group reactively esterified. The reactive esterified hydroxyl groups correspond to the above-mentioned conditions as well as the condensation conditions. In this reaction, in particular if both hydroxyl groups are esterified in a reactive manner, the starting materials of the formula IV can be obtained in situ, which are closed to form a ring in the same reaction.
Die neuen Verbindungen der Formel I, die eine weitere Doppelbindung aufweisen, können auch erhalten werden, wenn man Verbindungen der Formel Va bzw. Vb
Der Ringschluss erfolgt unter wasserabspaltenden Bedingungen, wie durch Erwärmen, z.B. von etwa 50° bis etwa 150°, vorzugsweise in Gegenwart eines Lösungsmittels, wie Acetonitril oder einem Alkohol, z.B. Methanol oder Aethanol.The ring closure takes place under water-releasing conditions, such as by heating, e.g. from about 50 ° to about 150 °, preferably in the presence of a solvent such as acetonitrile or an alcohol, e.g. Methanol or ethanol.
Die Ausgangsstoffe lassen sich erhalten, wenn man in an sich bekannter Weise eine Verbindung der Formel VI
Reaktionsfähig veresterte Hydroxygruppen X sind insbesondere die obgenannten.Reactively esterified hydroxy groups X are in particular the above.
Wird diese Umsetzung auch unter wasserabspaltenden Bedingungen vorgenommen, z.B. unter Erwärmen in einem Lösungsmittel wie Acetonitril oder einem Alkohol, kann der Ausgangsstoff der Formel Va bzw. Vb in situ erhalten werden, der sich unter diesen Reaktionsbedingungen ringschliesst.If this reaction is also carried out under water-releasing conditions, for example with heating in a solvent such as acetonitrile or an alcohol, the starting material of the formula Va or Vb can be obtained in situ, which closes under these reaction conditions.
Die Oxydation des Thia-atoms zur Sulfinyl- oder Sulfonylgruppe lässt sich in an sich bekannter Weise z.B. mit Peroxyden, wie Wasserstoffperoxyd, oder Persäuren, z.B. einer gegebenenfalls durch Niederalkyl, Niederalkoxy, Halogen oder einer weiteren Carboxylgruppe substituierten Benzoepersäure, wie Benzoepersäure selbst oder Phthalmono- persäure,oder einer Alkanpercarbonsäure, wie Peressigsäure oder einem Perjodat, wie Natriumperjodat durchführen. Diese Reaktion wird meist bei tiefen Temperaturen in einem Lösungsmittel, wie Eisessig oder Aceton vorgenommen.The oxidation of the thia atom to the sulfinyl or sulfonyl group can be carried out in a manner known per se, e.g. with peroxides such as hydrogen peroxide or peracids e.g. carry out an optionally substituted by lower alkyl, lower alkoxy, halogen or another carboxyl group, such as benzoperic acid itself or phthalmonoperacid, or an alkane percarboxylic acid such as peracetic acid or a periodate such as sodium periodate. This reaction is usually carried out at low temperatures in a solvent such as glacial acetic acid or acetone.
Die neuen Verbindungen können in Form von Säureadditionssalzen, insbesondere pharmazeutisch verwendbaren, nicht-toxischen Salzen, z.B. mit organischen Säuren, wie Chlorwasserstoff-, Bromwasserstoff-, Schwefel- oder Phosphorsäure, oder mit organischen, wie aliphatischen, cycloaliphatischen, cycloaliphatisch-aliphatischen, aromatischen, araliphatischen, heterocyclischen oder heterocyclisch-aliphatischen Carbon- oder Sulfonsäure, z.B. Essig-, Propion-, Bernstein-, Glykol-, Milch-, Aepfel-, 'Wein-, Zitronen-, Ascorbin-, Malein-, Phenylessig-, Benzoe-, 4-Aminobenzoe-, Anthranil-, 4-Hydroxybenzoe-, Salicyl-, Aminosalicyl-, Embon- oder Nicotin-, sowie Methansulfon-, Aethansulfon-, 2-Hydroxyäthansulfon-, Aethylensulfon-, Benzolsulfon-, p-Toluolsulfon-, Naphthalinsulfon-, Sulfanil- oder Cyclohexylsulfaminsäure, vorliegen. Salze dieser Art können z.B. durch Behandeln der freien Verbindungen, mit den Säuren oder mit geeigneten Anionenaustauscherharzen erhalten werden.The new compounds can be in the form of acid addition salts, especially pharmaceutically acceptable, non-toxic salts, e.g. with organic acids, such as hydrochloric, hydrobromic, sulfuric or phosphoric acid, or with organic, such as aliphatic, cycloaliphatic, cycloaliphatic-aliphatic, aromatic, araliphatic, heterocyclic or heterocyclic-aliphatic carbon or sulfonic acid, e.g. Vinegar, propion, amber, glycol, milk, apple, 'wine, lemon, ascorbic, maleic, phenylacetic, benzoic, 4-aminobenzoic, anthranil, 4-hydroxybenzoic, Salicylic, aminosalicylic, embon or nicotinic, as well as methanesulfonic, ethanesulfonic, 2-hydroxyethanesulfonic, ethylenesulfonic, benzenesulfonic, p-toluenesulfonic, naphthalenesulfonic, sulfanilic or cyclohexylsulfamic acid are present. Salts of this type can e.g. can be obtained by treating the free compounds, with the acids or with suitable anion exchange resins.
Infolge der engen Beziehungen zwischen den neuen Verbindungen in freier Form und in Form ihrer Salze sind im Vorausgegangenen und nachfolgend unter den freien Verbindungen oder den Salzen sinn- und zweckmässig gegebenenfalls auch die entsprechenden Salze bzw. freien Verbindungen zu verstehen.As a result of the close relationships between the new compounds in free form and in the form of their salts, in the preceding and the following, the free compounds or the salts are also to be understood as meaningful and expedient, if appropriate, the corresponding salts or free compounds.
Die neuen Verbindungen können als Isomerengemische, wie Racemate oder Diastereoisomerengemische, oder in Form der reinen Isomeren, wie optisch aktiven Komponenten, vorliegen. Die Auftrennung von erhaltenen Isomerengemischen in die reinen Isomeren kann nach den bekannten Methoden geschehen. Racemate lassen sich z.B. auf Grund physikalisch-chemischer Unterschide, wie z.B. solchen der Löslichkeit, ihrer diastereomeren Salze, oder durch fraktioniertes Kristallisieren aus einem optisch aktiven Lösungsmittel, oder durch Chromatographie, insbesondere DUnnschichtchromatographle, an einem optisch aktiven Trägermaterial, in die optisch aktiven Antipoden auftrennen. Dabei isoliert man vorteilhafterweise das pharmakologisch wirksamere oder weniger toxische reine Isomere, insbesondere den wirksameren oder weniger toxischen aktiven Antipoden.The new compounds can be present as isomer mixtures, such as racemates or diastereoisomer mixtures, or in the form of the pure isomers, such as optically active components. The separation of isomer mixtures obtained into the pure isomers can be carried out by the known methods. Racemates can e.g. due to physico-chemical differences, e.g. Separate those of solubility, their diastereomeric salts, or by fractional crystallization from an optically active solvent, or by chromatography, in particular thin-layer chromatography, on an optically active carrier material, into the optically active antipodes. The pharmacologically more effective or less toxic pure isomer is advantageously isolated, in particular the more effective or less toxic active antipode.
Die obigen Reaktionen werden in üblicher Weise in An- oder Abwesenheit von VerdUnnungs-, Kondensations-und/oder katalytischen Mitteln, falls notwendig, bei erniedrigter oder erhöhter Temperatur, im geschlossenen Gefäss und/oder in einer Inertgasatmosphäre durchgeführt.The above reactions are carried out in the customary manner in the presence or absence of diluent, condensation and / or catalytic agents, if necessary at reduced or elevated temperature, in a closed vessel and / or in an inert gas atmosphere.
Das Verfahren umfasst auch diejenigen AusfUhrungsformen, wonach als Zwischenprodukte anfallende Verbindungen als Ausgangsstoffe verwendet und die restlichen Verfahrensschritte mit diesen durchgeführt werden, oder das Verfahren auf irgendeiner Stufe abgebrochen wird; ferner können Ausgangsstoffe in Form von Derivaten verwendet oder während der Reaktion gebildet werden.The process also includes those embodiments according to which compounds obtained as intermediates are used as starting materials and the remaining process steps are carried out with them, or the process is terminated at any stage; furthermore, starting materials in the form of derivatives can be used or formed during the reaction.
Vorzugsweise werden solche Ausgangsstoffe verwendet und die Reaktionsbedingungen so gewählt, dass man zu den eingangs als besonders bevorzugt aufgeführten Verbindungen gelangt.Such starting materials are preferably used and the reaction conditions are selected so that the compounds listed at the outset as particularly preferred are obtained.
Die Erfindung betrifft auch die als Zwischenprodukte erhältlichen neuen Verbindungen der allgemeinen Formel Va bzw. Vb
Die neuen Verbindungen der vorliegenden Erfindung können z.B. zur Herstellung von pharmazeutischen Präparaten verwendet werden, welche eine wirksame Menge der Aktivsubstanz zusammen oder im Gemisch mit anorganischen oder organischen, festen oder flüssigen, pharmazeutisch verwendbaren Trägerstoffen enthalten, die sich zur enteralen oder parenteralen Verabreichung eignen. So verwendet man Tabletten oder Gelatinekapseln, welche den Wirkstoff zusammen mit Verdünnungsmitteln, z.B. Laktose, Dextrose, Sukrose, Mannitol, Sorbitol, Cellulose und/oder Glycerin, und Schmiermitteln, z.B. Kieselerde, Talk, Stearinsäure oder Salze davon, wie Magnesium- oder Calciumstearat, und/oder Polyäthylenglykol, aufweisen; Tabletten enthalten ebenfalls Bindemittel, z.B. Magnesiumaluminiumsilikat, Stärken, wie Mais-, Weizen-, Reis- oder Pfeilwurzstärke, Gelatine, Traganth, Methylcellulose, Natriumcarboxymethylcellulose und/oder Polyvinylpyrrolidon, und, wenn erwUnscht, Sprengmittel, z.B. Stärken, Agar, Alginsäure oder ein Salz davon, wie Natriumalginat, und/oder Brausemischungen, oder Adsorptionsmittel, Farbstoffe, Geschmackstoffe und SUssmittel. Ferner kann man die neuen pharmakologisch wirksamen Verbindungen in Form von injizierbaren, z.B. intravenös verabreichbaren Präparaten oder von Infusionslösungen verwenden. Solche Lösungen sind vorzugsweise isotonische wässrige Lösungen oder Suspensionen, wobei diese z.B. aus lyophilisierten Präparaten, welche die Wirksubstanz allein oder zusammen mit einem Trägermaterial, z.B. Mannit, enthalten, vor Gebrauch hergestellt werden können. Die pharmazeutischen Präparate können sterilisiert sein und/oder Hilfsstoffe, z.B. Konservier-, Stabilisier-, Netz- und/oder Emulgiermittel, Löslichkeitsvermittler, Salze zur Regulierung des osmotischen Druckes und/oder Puffer enthalten. Die vorliegenden pharmazeutischen Präparate, die, wenn erwUnscht, weitere pharmakologisch wertvolle Stoffe enthalten können, werden in an sich bekannter Weise, z.B. mittels konventioneller Misch-, Granulier-, Dragier-, Lösungs- oder Lyophilisierungsverfahrcn, hergestellt und enthalten von etwa 0,1% bis 100%, insbesondere von etwa 1% bis etwa 50%, Lyophilisate bis zu 100% des Aktivstoffes. Die Einzeldosis fUr einen Warmblüter von etwa 70 kg Gewicht beträgt zwischen 0,1 und 0,75 g, die Tagesdosis zwischen 0,2 und 1,0 g.The new compounds of the present invention can be used, for example, for the production of pharmaceutical preparations which contain an effective amount of the active substance together or in a mixture with inorganic or contain organic, solid or liquid, pharmaceutically usable excipients which are suitable for enteral or parenteral administration. Thus, tablets or gelatin capsules are used which contain the active ingredient together with diluents, for example lactose, dextrose, sucrose, mannitol, sorbitol, cellulose and / or glycerol, and lubricants, for example silica, talc, stearic acid or salts thereof, such as magnesium or calcium stearate. and / or polyethylene glycol; Tablets also contain binders, e.g. magnesium aluminum silicate, starches such as corn, wheat, rice or arrowroot starch, gelatin, tragacanth, methyl cellulose, sodium carboxymethyl cellulose and / or polyvinylpyrrolidone, and, if desired, disintegrants, e.g. starches, agar, alginic acid or a salt thereof, such as sodium alginate, and / or effervescent mixtures, or adsorbents, colors, flavors and sweeteners. Furthermore, the new pharmacologically active compounds can be used in the form of injectable, for example intravenously administrable, preparations or infusion solutions. Such solutions are preferably isotonic aqueous solutions or suspensions, these being able to be prepared, for example, from lyophilized preparations which contain the active substance on their own or together with a carrier material, for example mannitol. The pharmaceutical preparations can be sterilized and / or contain auxiliaries, for example preservatives, stabilizers, wetting agents and / or emulsifiers, solubilizers, salts for regulating the osmotic pressure and / or buffers. The present pharmaceutical preparations, which, if desired, may contain other pharmacologically valuable substances, are produced in a manner known per se, for example by means of conventional mixing, granulating, confectioning, dissolving or lyophilizing processes and contain from about 0.1% to 100%, in particular from about 1% to about 50%, of lyophilisates up to 100% of the active ingredient. The single dose for a warm-blooded animal weighing approximately 70 kg is between 0.1 and 0.75 g, the daily dose between 0.2 and 1.0 g.
Die folgenden Beispiele dienen zur Illustration der Erfindung; Temperaturen werden in Celsiusgraden angegeben.The following examples serve to illustrate the invention; Temperatures are given in degrees Celsius.
Eine Mischung aus 5 g a-Brom-desoxyanisoin, 3 g 2-Aminothiazolin und 30 ml Aethanol wird 4 Stunden bei 60°, danach 2 Stunden am RUckfluss und anschliessend noch 12 Stunden bei Raumtemperatur gerührt. Die dabei ausgefallenen Kristalle werden abgenutscht und mit Aethanol und Diäthyläther nachgewaschen. Man erhält so rohes 5,6-Di-(p-methoxy-phenyl)-imidazo[2,1-b]dihydro-thiazol. F. nach dem Umkristallisieren aus Toluol-Petroläther 152-1540 A mixture of 5 g of a-bromo-deoxyanisoin, 3 g of 2-aminothiazoline and 30 ml of ethanol is stirred for 4 hours at 60 °, then for 2 hours at reflux and then for a further 12 hours at room temperature. The crystals which have precipitated are filtered off with suction and washed with ethanol and diethyl ether. This gives crude 5,6-di- (p-methoxyphenyl) imidazo [2,1-b] dihydro-thiazole. F. after recrystallization from toluene-petroleum ether 152-154 0
Eine Suspension von 7 ml 1,2-Dibromäthan, 7 g Natriumcarbonat und 55 ml Isopropanol wird bei Raumtemperatur gerührt und innert einer Stunde mit der Suspension von 4,7 g 4,5-Diphenyl-imidazolidin-2-thion in 110 ml l,5%-iger Natronlauge versetzt. Das Reaktionsgemisch wird 7 Stunden am RUckfluss gekocht, dann werden das Isopropanol und das Dibromäthan am Rotationsverdampfer entfernt und die verbleibende Suspension wird mit Toluol extrahiert. Der Toluolextrakt wird mit Sole gewaschen, über Natriumsulfat getrocknet und eingedampft. Der Rückstand wird auf Silicagel chromatographiert. Nach dem Abtrennen unpolarer Verunreinigungen mit Aethylacetat wird das cis-5,6-Dipheny1-2,3,5,6-tetrahydro-imidazo[2,1-b]thiazol mit einem Gemisch aus Aethylacetat:Methanol = 99:1 als farbloses Oel eluiert. Es kristallisiert spontan zu weissen Kristallen vom Smp. 110-113° (Sinterpunkt 103°)A suspension of 7 ml of 1,2-dibromoethane, 7 g of sodium carbonate and 55 ml of isopropanol is stirred at room temperature and within one hour with the suspension of 4.7 g of 4,5-diphenyl-imidazolidin-2-thione in 110 ml of l, 5% sodium hydroxide solution added. The reaction mixture is refluxed for 7 hours, then the isopropanol and the dibromoethane are removed on a rotary evaporator and the remaining suspension is extracted with toluene. The toluene extract is washed with brine, dried over sodium sulfate and evaporated. The residue is chromatographed on silica gel. After separating non-polar impurities with ethyl acetate, the cis-5,6-dipheny1-2,3,5,6-tetrahydro-imidazo [2,1-b] thiazole is mixed with a mixture of ethyl acetate: methanol = 99: 1 as a colorless oil eluted. It spontaneously crystallizes to white crystals of mp 110-113 ° (sintering point 103 °)
Eine Suspension von 3,5 ml 1,2-Dibromäthan, 3,5 g Natriumcarbonat und 30 ml Isopropanol wird unter Rühren bei Raumtemperatur während einer Stunde mit der Suspension von 3 g 4,5-Di-anisyl-2-mercapto-imidazol in 50 ml 1,5%-iger Natronlauge versetzt. Das Reaktionsgemisch wird 6 Stunden am RUckfluss gekocht, dann werden das Isopropanol und das Dibromäthan am Rotationsverdampfer entfernt und die verbleibende Suspension wird mit 10 ml 20%-iger Kalilauge versetzt und mit Aethylacetat extrahiert. Die organischen Phasen werden mit Sole gewaschen, mit Natriumsulfat getrocknet und eingedampft. Aus dem Rückstand erhält man nach dem Umkristallisieren aus Toluol-Petroläther das 5,6-Di-(p-methoxy-phenyl)-imidazo[2,1-b]dihydro-thiazol vom F. 152-154°.A suspension of 3.5 ml of 1,2-dibromoethane, 3.5 g of sodium carbonate and 30 ml of isopropanol is stirred in with the suspension of 3 g of 4,5-di-anisyl-2-mercapto-imidazole in at room temperature for one hour 50 ml of 1.5% sodium hydroxide solution are added. The reaction mixture is refluxed for 6 hours, then the isopropanol and the dibromoethane are removed on a rotary evaporator and the remaining suspension is mixed with 10 ml of 20% potassium hydroxide solution and extracted with ethyl acetate. The organic phases are washed with brine, dried with sodium sulfate and evaporated. After recrystallization from toluene-petroleum ether, the 5,6-di- (p-methoxy-phenyl) -imidazo [2,1-b] dihydro-thiazole of mp 152-154 ° is obtained from the residue.
14,8 g 4,5-Di-(p-Methoxy-phenyl)-2-ß-hydroxy- äthylmercapto-imidazol werden in 50 ml absolutem Pyridin gelöst und unter Rühren bei ca -5° Innentemperatur zu einer Lösung von 15,8 g Benzolsulfochlorid in 60 ml absolutem Pyridin getropft. Anschliessend rührt man 60 Stunden bei 0° aus. Das Reaktionsgemisch wird auf Eiswasser gegossen und mit Dichlormethan extrahiert. Die mit Wasser 3-mal gewaschenen und vereinigten Dichlormethanextrakte werden über Natriumsulfat getrocknet und zur Trockne eingedampft. Das viskose Oel wird aus Toluol-Petroläther kristallisiert. Das so erhaltene 5,6-Di-(p-methoxyphenyl)-imidazo(2,1-b]dihydro-thiazol vom F. 152-154°.14.8 g of 4,5-di- (p-methoxy-phenyl) -2-ß-hydroxy-ethylmercapto-imidazole are dissolved in 50 ml of absolute pyridine and with stirring at about -5 ° internal temperature to a solution of 15.8 g of benzenesulfonyl chloride dropwise in 60 ml of absolute pyridine. The mixture is then stirred at 0 ° for 60 hours. The reaction mixture is poured onto ice water and extracted with dichloromethane. The dichloromethane extracts washed and combined 3 times with water are dried over sodium sulfate and evaporated to dryness. The viscous oil is crystallized from toluene-petroleum ether. The 5,6-di- (p-methoxyphenyl) imidazo (2,1-b] dihydro-thiazole of mp 152-154 ° thus obtained.
Das Ausgangsmaterial kann z,B. hergestellt werden: 2,5 g Natrium werden in 220 ml Aethanol gelöst. Dazu gibt man 35 g 4,5-Dianisyl-imidazolin-2-thion. Man erhält eine Suspension. Innerhalb 2 Minuten werden dazu bei Raumtemperatur (RUhren) 15,7 ml 2-Chloräthanol getropft. Man rührt 1 Stunde bei 60° und 4 Stunden am RUckfluss weiter. Anschliessend wird die schwache Suspension zur Trockne eingedampft. Der Rückstand wird aus Aceton/ Wasser gereinigt.The starting material can e.g. are prepared: 2.5 g of sodium are dissolved in 220 ml of ethanol. 35 g of 4,5-dianisyl-imidazolin-2-thione are added. A suspension is obtained. For this purpose, 15.7 ml of 2-chloroethanol are added dropwise at room temperature (stirring). The mixture is stirred for 1 hour at 60 ° and 4 hours at reflux. The weak suspension is then evaporated to dryness. The residue is purified from acetone / water.
Ein Gemisch aus 60 g N-Aethyl-diisopropylamin, 70 g 2-Aminodihydrothiazol, 150 g a-Brom-desoxyanisoin und 800 ml Acetonitril wird 48 Stunden bei Raumtemperatur gerUhrt. Das dabei entstandene 5,6-Di-(p-methoxyphenyl)-imidazo[2,1-b)dihydro-thiazolin wird abgenutscht und mit Acetonitril nachgewaschen. Durch Eindampfen des Filtrates und Verteilen des so vom Lösungsmittel befreiten Rückstandes zwischen Aethylacetat und Wasser erhält man weitere 50 g Rohprodukt. Durch Umkristallisieren des Rohproduktes aus Toluol erhält man 5,6-Di-(p-methoxyphenyl)-imidazo[2.1-b]dihydro-thiazol vom F. 152-154°.A mixture of 60 g of N-ethyl-diisopropylamine, 70 g of 2-aminodihydrothiazole, 150 g of a-bromo-deoxyanisoin and 800 ml of acetonitrile is stirred for 48 hours at room temperature. The resulting 5,6-di- (p-methoxyphenyl) imidazo [2,1-b) dihydro-thiazoline is filtered off and washed with acetonitrile. Evaporation of the filtrate and distribution of the residue thus freed from solvent between ethyl acetate and water gives a further 50 g of crude product. By recrystallizing the crude product from toluene, 5,6-di- (p-methoxyphenyl) imidazo [2.1-b] dihydro-thiazole of mp 152-154 ° is obtained.
Zu einer Lösung von 23 g p-Toluolsulfochlorid in 80 ml Pyridin wird bei 0° unter Rühren die Lösung von 20 g 2-(2-Hydroxy-äthyl-thio)-4(5)-3'-pyridyl-5(4)-phenyl-imidazol zugegeben. Das Reaktionsgemisch wird 16 Stunden bei 0° gerührt, auf Eis-Wasser gegossen und mit Aether extrahiert. Aus den Aetherextrakten erhält man nach dem Waschen mit Wasser und Trocknen über Natriumsulfat einen Eindampfrückstand der nach dem Chromatographieren auf Silicagel mit Toluol und Essigester und nach dem Umkristallisieren der entsprechenden Fraktionen aus Essigester das Isomerengemisch 5(6)-(3-Pyridyl)-6(5)-phenyl-imidazo-[2,1-b]-dihydro-thiazol als weisse Kristalle vom F. 150-151° ergibt.The solution of 20 g of 2- (2-hydroxyethylthio) -4 (5) -3'-pyridyl-5 (4) is added to a solution of 23 g of p-toluenesulfochloride in 80 ml of pyridine at 0 ° with stirring. -phenyl imidazole added. The reaction mixture is stirred at 0 ° for 16 hours, poured onto ice-water and extracted with ether. After washing with water and drying over sodium sulfate, an ether residue is obtained from the ether extracts, which after chromatography on silica gel with toluene and ethyl acetate and after recrystallization install the appropriate fractions from ethyl acetate, the isomer mixture 5 (6) - (3-pyridyl) -6 (5) -phenyl-imidazo- [2,1-b] -dihydro-thiazole as white crystals of mp 150-151 ° .
Das Ausgangsmaterial lässt sich wie folgt erhalten:
- 10,8 g Benzyl-pyridyl(3)-keton werden zusammen
mit 40 ml Pyridin und einer Lösung von 8 g Hydroxylaminhydorchlorid in 15 ml Pyridin während 6 Stunden bei 100° gerührt. Das Reaktionsgemisch wird auf Eis/Wasser gegossen und 15 Minuten weitergerührt. Die ausgefallenen Kristalle werden abgenutscht, mit Wasser gewaschen und am Hochvakuum getrocknet. Man erhält das Benzyl-pyridyl(3)-keton-oxim vom F. 122-126°.The starting material can be obtained as follows:
- 10.8 g of benzyl pyridyl (3) ketone are combined
stirred with 40 ml of pyridine and a solution of 8 g of hydroxylamine hydrochloride in 15 ml of pyridine at 100 ° for 6 hours. The reaction mixture is poured onto ice / water and stirred for a further 15 minutes. The precipitated crystals are filtered off, washed with water and dried in a high vacuum. The benzyl-pyridyl (3) -ketone-oxime of mp 122-126 ° is obtained.
Zu einer bei -10° gerührten Lösung von 8,5 g Benzyl-pyridyl(3)-keton-oxim in 20 ml Pyridin wird innert 5 Minuten die Lösung von 7,7 g p-Toluolsulfochlorid in 15 ml Pyridin zugetropft. Das Reaktionsgemisch wird 24 Stunden im Eisschrank aufbewahrt und dann auf Eis/Wasser gegossen. Nach längerem Rühren und Verreiben erstarrt das ausgefallene Oel zu Kristallen. Diese werden abgenutscht, mit Wasser gewaschen und am Hochvakuum getrocknet. Man erhält 11,6 g eines Rohproduktes vom F. 87-92°, welches nach Dünnschichtchromatographie jedoch noch Edukt enthält. Es wird direkt in der nächsten Stufe eingesetzt.The solution of 7.7 g of p-toluenesulfochloride in 15 ml of pyridine is added dropwise to a solution of 8.5 g of benzylpyridyl (3) ketone oxime in 20 ml of pyridine which is stirred at -10 ° within 5 minutes. The reaction mixture is kept in the refrigerator for 24 hours and then poured onto ice / water. After stirring and rubbing for a long time, the precipitated oil solidifies to crystals. These are filtered off, washed with water and dried in a high vacuum. 11.6 g of a crude product having a melting point of 87-92 ° are obtained, which however still contains starting material after thin layer chromatography. It is used directly in the next stage.
11,6 g Rohprodukt (Benzyl-pyridyl(3)-keton-oxim- p-toluolsulfoester) werden in 90 ml absolutem Aethanol suspendiert und bei 0° unter Rühren die Lösung von 3,7 g Kalium-tert.-butylat in 30 ml absolutem Aethanol zugetropft. Das Reaktionsgemisch wird 2 Stunden bei 0° gerührt. Die Suspension wird abgenutscht und das Filtrat sofort in der nächsten Stufe eingesetzt.11.6 g of crude product (benzyl-pyridyl (3) -ketone-oxime-p-toluenesulfoester) are suspended in 90 ml of absolute ethanol and at 0 ° with stirring the solution of 3.7 g of potassium tert-butoxide in 30 ml dropped in absolute ethanol. The reaction mixture is stirred at 0 ° for 2 hours. The suspension is filtered off and the filtrate is immediately used in the next stage.
3,6 g Natriumthiocyanat werden in 60 ml Aethanol gelöst und mit 4,5 ml konzentrierter Salzsäure versetzt. Die Suspension wird abgenutscht und das Filtrat wird zusammen mit der alkoholischen Lösung des erhaltenen a-Amino- benzyl-pyridyl(3)-keton während 18 Stunden am RUckfluss gehalten. Nach dem Abkühlen lässt sich aus dem Reaktionsgemisch 2,8 g rohes 4-Phenyl-5-pyridyl(3)-2-mercapto- imidazol abnutschen. Das Filtrat enthält weitere Mengen des Produktes. Nach dem Umkristallisieren aus Dimethylformamid-Wasser schmilzt es bei 290-300°.3.6 g of sodium thiocyanate are dissolved in 60 ml of ethanol and 4.5 ml of concentrated hydrochloric acid are added. The suspension is filtered off with suction and the filtrate, together with the alcoholic solution of the a-aminobenzyl-pyridyl (3) ketone obtained, is refluxed for 18 hours. After cooling, 2.8 g of crude 4-phenyl-5-pyridyl (3) -2-mercapto-imidazole can be sucked off from the reaction mixture. The filtrate contains further amounts of the product. After recrystallization from dimethylformamide water, it melts at 290-300 °.
0,8 g Natrium werden in 200 ml Aethanol gelöst. Die Lösung wird mit 8 g l-Phenyl-5-pyridyl(3)-2-mercapto- imidazol versetzt und zum Rückfluss erhitzt. Nachdem eine klare Lösung entstanden ist, werden 2,3 ml 2-Chloräthanol zugetropft und das Reaktionsgemisch wird 16 Stunden am RUckfluss gekocht. Das Lösungsmittel wird am Vakuum abgedampft und der Rückstand zwischen Wasser und Essigester verteilt. Die organischen Phasen werden mit Wasser und Sole gewaschen, mit Natriumsulfat getrocknet und eingedampft. Als Rückstand erhält man kristallines 2-(2-Hydroxy-äthyl-thio)-4(5)-3'-pyridyl-5(4)-phenyl-imidazol. Es schmilzt nach dem Umkristallisieren aus Essigester/Aethanol bei 157-159°C.0.8 g of sodium are dissolved in 200 ml of ethanol. The solution is mixed with 8 g of l-phenyl-5-pyridyl (3) -2-mercapto-imidazole and heated to reflux. After a clear solution has formed, 2.3 ml of 2-chloroethanol are added dropwise and the reaction mixture is boiled under reflux for 16 hours. The solvent is evaporated off in vacuo and the residue is partitioned between water and ethyl acetate. The organic phases are washed with water and brine, dried with sodium sulfate and evaporated. The residue obtained is crystalline 2- (2-hydroxyethylthio) -4 (5) -3'-pyridyl-5 (4) -phenyl-imidazole. It melts after recrystallization from ethyl acetate / ethanol at 157-159 ° C.
In analoger Weise, ausgehend von den entsprechenden Ausgangsstoffe lassen sich die folgenden Verbindungen herstellen:
- das 5,6-Di-(p-methyl-phenyl)-2,3,5,6-tetrahydro- imidazo[2,1-b]thiazol,
- das 5,6-Di-(p-chlor-phenyl)-2,3,5,6-tetrahydro- imidazo[2,1-b]thiazol,
- das 5,6-Di-(m-chlor-phenyl)-2,3,5,6-tetrahydro- imidazo[2,1-b]thiazol,
- das 5,6-Di-(p-methoxy-phenyl)-3-methyl-2,3,5,6-tetrahydro-imidazo(2,1-b]thiazol oder
- das 6,7-Di-(p-methoxy-phenyl)-2,3,4,5,6,7-hexa- hydro-imidazo(2,1-b]thiazin.
- 5,6-di- (p-methylphenyl) -2,3,5,6-tetrahydroimidazo [2,1-b] thiazole,
- the 5,6-di- (p-chlorophenyl) -2,3,5,6-tetrahydroimidazo [2,1-b] thiazole,
- the 5,6-di- (m-chlorophenyl) -2,3,5,6-tetrahydroimidazo [2,1-b] thiazole,
- the 5,6-di- (p-methoxy-phenyl) -3-methyl-2,3,5,6-tetrahydro-imidazo (2,1-b] thiazole or
- the 6,7-di- (p-methoxy-phenyl) -2,3,4,5,6,7-hexa- hydroimidazo (2,1-b] thiazine.
14,3 g 2-[N-(1,2-Bis-p-methoxy-pheny1-2-hydroxyäthyl]-amino-thiazolin werden in 24,3 ml konzentrierter Schwefelsäure gelöst und eine Stunde bei Raumtemperatur gerührt. Das Reaktionsgemisch wird auf Eis gegossen, mit 2-n Sodalösung alkalisch gestellt und mit Essigester extrahiert Die organischen Phasen werden mit Wasser gewaschen, über Natriumsulfat getrocknet und eingedampft. Aus dem Rückstand erhält man durch Umkristallisieren aus Toluol-Petroläther das 5,6-trans-Di-(p-methoxyphenyl)-2,3,5,6-tetrahydro-4H-imidazol[2,1-b]thiazol vom F. 125-126°.14.3 g of 2- [N- (1,2-bis-p-methoxy-pheny1-2-hydroxyethyl] amino thiazoline are dissolved in 24.3 ml of concentrated sulfuric acid and stirred for one hour at room temperature. The reaction mixture is stirred up Poured ice, made alkaline with 2N sodium carbonate solution and extracted with ethyl acetate. The organic phases are washed with water, dried over sodium sulfate and evaporated. The residue obtained by recrystallization from toluene-petroleum ether gives 5,6-trans-di- (p -methoxyphenyl) -2,3,5,6-tetrahydro-4H-imidazole [2,1-b] thiazole, mp 125-126 °.
Das verwendete Ausgangsmaterial lässt sich z.B. wie folgt erhalten:The starting material used can e.g. received as follows:
45 g Brom-desoxy-anisoin werden in 170 ml Acetonitril suspendiert. Die Suspension wird mit 23 ml N-Aethyl-diisopropylamin und mit 15,4 g 2-Aminothiazolin versetzt, 2 Stunden bei Raumtemperatur gerührt, abgenutscht und das Nutschgut mit wenig Essigester nachgewaschen. Kan erhält so das 2-[N-(1,2-Bis-p-methoxyphenyl-2-oxo-äthyl)-aminothiazolin bzw. das 2-Imino-3-(l,2-bis-p-methoxy-phenyl-2-oxo-äthyl)-thiazolin vom F. = 95-98°.45 g of bromo-deoxy-anisoin are suspended in 170 ml of acetonitrile. The suspension is mixed with 23 ml of N-ethyl-diisopropylamine and with 15.4 g of 2-aminothiazoline, stirred for 2 hours at room temperature, suction filtered and the filter cake washed with a little ethyl acetate. Kan thus receives the 2- [N- (1,2-bis-p-methoxyphenyl-2-oxo-ethyl) aminothiazoline or the 2-imino-3- (1,2-bis-p-methoxy-phenyl- 2-oxo-ethyl) -thiazoline with mp = 95-98 °.
In analoger Weise erhält man
- das 2-(N-(1,2-Bis-p-methoxyphenyl-2-oxo-äthyl)-amino-5-methyl-thiazolin bzw. 2-Imino-5-methyl-3-(1,2-Bis-p-methoxyphenyl-2-oxo-äthyl)-thiazolin oder deren Tautomeren,
- das 2-[N-(1,2-Bis-p-methoxyphenyl-2-oxo-äthyl)-amino-4-methyl-thiazolin bzw: 2-Imino-4-methyl-3-(1,2-bis- p-methoxyphenyl-2-oxo-äthyl)-thiazolin oder deren Tautomeren,
- das 2-[N-(1,2-Bis-phenyl-2-oxo-äthyl)-aminothiazolin bzw. 2-Imino-3-(1,2-bis-phenyl-2-oxo-äthyl)-thiazolin oder deren Tautomeren vom F. 121-123°, oder
- das 2-[N-(1,2-Bis-p-methoxyphenyl-2-oxo-äthyl)-amino-5,6-dihydro-4H-thiazin bzw. 2-Imino-3-(1,2-bis-p-methoxyphenyl-2-oxo-äthyl)-5,6-dihydro-4H-thiazin oder deren Tautomeren.
- the 2- (N- (1,2-bis-p-methoxyphenyl-2-oxo-ethyl) amino-5-methylthiazoline or 2-imino-5-methyl-3- (1,2-bis- p-methoxyphenyl-2-oxo-ethyl) thiazoline or its tautomers,
- the 2- [N- (1,2-bis-p-methoxyphenyl-2-oxo-ethyl) amino-4-methylthiazoline or: 2-imino-4-methyl-3- (1,2-bis- p-methoxyphenyl-2-oxo-ethyl) thiazoline or its tautomers,
- the 2- [N- (1,2-bis-phenyl-2-oxo-ethyl) aminothiazoline or 2-imino-3- (1,2-bis-phenyl-2-oxo-ethyl) thiazoline or their Tautomers of F. 121-123 °, or
- the 2- [N- (1,2-bis-p-methoxyphenyl-2-oxo-ethyl) amino-5,6-dihydro-4H-thiazine or 2-imino-3- (1,2-bis- p-methoxyphenyl-2-oxo-ethyl) -5,6-dihydro-4H-thiazine or its tautomers.
31,5 g 2-[N-(1,2-Bis-p-methoxyphenyl-2-oxo- äthyl)-amino-thiazolin oder dessen Isomeren werden in 350 ml Methanol gelöst und portionsweise mit 4,26 g Natriumborhydrid versetzt. Die Suspension wird 2 Stunden bei Raumtemperatur gerührt, dann abgenutscht und mit Methanol nachgewaschen. Man erhält so das 2-[N-(l,2-Bis-p-methoxyphenyl-2-hydroxy-äthyl)-amino-thiazolin vom F. 185-187°.31.5 g of 2- [N- (1,2-bis-p-methoxyphenyl-2-oxoethyl) amino thiazoline or its isomers are dissolved in 350 ml of methanol, and 4.26 g of sodium borohydride are added in portions. The suspension is stirred for 2 hours at room temperature, then suction filtered and washed with methanol. This gives the 2- [N- (1,2-bis-p-methoxyphenyl-2-hydroxy-ethyl) amino thiazoline of mp 185-187 °.
Ausgehend von den entsprechenden Thiazolinverbindungen lassen sich in analoger Weise die folgenden Verbindungen erhalten:
- das 5-Phenyl-6-p-chlorphenyl-2,3,5,6-tetrahydro- imidazo[2,1-b]thiazol,
- das 6-Phenyl-5-p-chlorphenyl-2,3,5,6-tetrahydro- imidazo[2,1-b]thiazol,
- das 5,6-Di-(p-methoxyphenyl)-2-methyl-2,3,5,6-tetrahydro-imidazo[2,1-b]thiazol oder
- das 5,6-Di-(p-methoxyphenyl-3-methyl-2,3,5,6-tetrahydro-imidazo(2,1-b]thiazol.
- 5-phenyl-6-p-chlorophenyl-2,3,5,6-tetrahydroimidazo [2,1-b] thiazole,
- 6-phenyl-5-p-chlorophenyl-2,3,5,6-tetrahydroimidazo [2,1-b] thiazole,
- the 5,6-di- (p-methoxyphenyl) -2-methyl-2,3,5,6-tetrahydro-imidazo [2,1-b] thiazole or
- the 5,6-di- (p-methoxyphenyl-3-methyl-2,3,5,6-tetrahydro-imidazo (2,1-b) thiazole.
2 g 5,6-Di-p-methoxyphenyl-2,3-dihydro-4H-imida- zo[2,1-b] thiazol werden in 33 ml Aethanol suspendiert und innert 5 Minuten mit 7,91 ml 30%-igem Wasserstoffperoxyd versetzt. Das Reaktionsgemisch wird 90 Minuten am RUckfluss gehalten, abgekühlt, auf 200 ml Eiswasser gegossen und mit Essigester extrahiert. Aus dem EindampfrUckstand der mit Wasser gewaschenen und über Natriumsulfat getrockneten organischen Phasen erhält man durch Umkristallisieren das 5,6-Di-p-methoxyphenyl-imidazo[2,1-b]2,3-dihydro-thiazol-sulfoxid vom F. 176-177°.2 g of 5,6-di-p-methoxyphenyl-2,3-dihydro-4H-imidazo [2,1-b] thiazole are suspended in 33 ml of ethanol and within 5 minutes with 7.91 ml of 30% strength Hydrogen peroxide added. The reaction mixture is refluxed for 90 minutes, cooled, poured onto 200 ml of ice water and extracted with ethyl acetate. From the evaporation residue the organic phases, washed with water and dried over sodium sulfate, are obtained by recrystallization from the 5,6-di-p-methoxyphenyl-imidazo [2,1-b] 2,3-dihydro-thiazole sulfoxide of mp 176-177 °.
0,69 g Natrium werden in 60 ml Aethanol gelöst und mit 9,36 g 2-Mercapto-4,5-di-p-methoxyphenyl-imidazol versetzt. Das Reaktionsgemisch wird 30 Minuten gerührt, mit 3,1 g 3-Chlor-l-propanol versetzt, 2 Stunden am Rückfluss gehalten, abgekühlt, klar-filtriert und zur Trockne eingedampft. Der Rückstand, das rohe 2-(3-Hydroxy-propyl- thio)-4,5-di-p-methoxyphenyl-imidazol wird in 50 m1 Thionylchlorid 30 Minuten am RUckfluss gehalten. Das Uberschüssige Thionylchlorid wird abgedampft, durch Abdestillieren von 100 ml noch zugesetztem Chloroform abgetrieben und der Rückstand zusammen mit 100 ml Aethanol und 50 ml 40%-iger Kalilauge 4 Stunden am RUckfluss gehalten. Danach wird das Reaktionsgemisch am Rotationsverdampfer eingedampft, mit Eiswasser versetzt und mit Essigester extrahiert. Die organischen Phasen werden mit Wasser gewaschen, getrocknet und eingedampft. Der Rückstand wird mit Toluol:Essigester 1:1 auf Silicagel chromatrographiert. Durch Umkristallisieren der entsprechenden Fraktionen aus Aceton erhält man das 6,7-Di-p-methoxyphenyl-2,3,4,5-tetrahydro-imidazo[2,1-b] (1,3)-thiazin vom 189-191°.0.69 g of sodium are dissolved in 60 ml of ethanol and 9.36 g of 2-mercapto-4,5-di-p-methoxyphenyl-imidazole are added. The reaction mixture is stirred for 30 minutes, 3.1 g of 3-chloro-l-propanol are added, the mixture is kept under reflux for 2 hours, cooled, filtered clear and evaporated to dryness. The residue, the crude 2- (3-hydroxy-propyl-thio) -4,5-di-p-methoxyphenyl-imidazole, is refluxed in 50 ml of thionyl chloride for 30 minutes. The excess thionyl chloride is evaporated, stripped off by distilling off 100 ml of chloroform still added, and the residue is refluxed for 4 hours together with 100 ml of ethanol and 50 ml of 40% potassium hydroxide solution. The reaction mixture is then evaporated on a rotary evaporator, mixed with ice water and extracted with ethyl acetate. The organic phases are washed with water, dried and evaporated. The residue is chromatographed on silica gel with toluene: ethyl acetate 1: 1. By recrystallizing the corresponding fractions from acetone, the 6,7-di-p-methoxyphenyl-2,3,4,5-tetrahydro-imidazo [2,1-b] (1,3) -thiazine at 189-191 ° is obtained .
Analog Beispiel 9 wird aus dem 2-Mercapto-4,5-di-p-chlorphenyl-imidazol mit 2-Chloräthanol das 2-(2-Hydroxy-äthyl-thio)-4,5-di-p-chlorphenyl-imidazol vom F. 197-199° hergestellt und daraus durch Cyclisation mit Thionylchlorid das 5,6-Di-p-chlorphenyl-imidazo[2,1-b]dihydro-thiazol vom F. 199-204°.Analogously to Example 9, the 2-mercapto-4,5-di-p-chlorophenylimidazole with 2-chloroethanol is the 2- (2-hydroxyethylthio) -4,5-di-p-chlorophenylimidazole from F. 197-199 ° and from it by cyclization with thionyl chloride the 5,6-di-p-chlorophenyl-imidazo [2,1-b] dihydro-thiazole of mp 199-204 °.
Analog kann auch das 5,6-di-p-Methoxyphenyl-4H-imidazo[2,1-b]dihydro-thiazol hergestellt werden.The 5,6-di-p-methoxyphenyl-4H-imidazo [2,1-b] dihydro-thiazole can also be prepared analogously.
0,5 g 5,6-Di-p-Methoxyphenyl-2,3-dihydro-4H-imidazo[2,1-b]thiazol werden in 1,5 ml Eisessig suspendiert und mit 0,9 ml 30%-igem Wasserstoffperoxid versetzt. Das Reaktionsgemisch wird 7 Stunden bei 50° gerührt, dann auf Eis-Wasser gegossen und mit Essigester extrahiert. Die organischen Phasen werden mit 1-n Natronlauge gewaschen bis die Extrakte basisch bleiben, dann mit Wasser und Sole neutralgewaschen, über Natriumsulfat getrocknet und eingedampft. Der RUckstand wird aus Essigester kristallisiert, und aus Essigester-Petroläther umkristallisiert. Man erhält so das 5,6-di-p-Methoxyphenyl-imidazo[2,1-b]dihydro-thiazol-sulfon vom F. 186-187°.0.5 g of 5,6-di-p-methoxyphenyl-2,3-dihydro-4H-imidazo [2,1-b] thiazole are suspended in 1.5 ml of glacial acetic acid and with 0.9 ml of 30% hydrogen peroxide transferred. The reaction mixture is stirred at 50 ° for 7 hours, then poured onto ice-water and extracted with ethyl acetate. The organic phases are washed with 1N sodium hydroxide solution until the extracts remain basic, then washed neutral with water and brine, dried over sodium sulfate and evaporated. The residue is crystallized from ethyl acetate and recrystallized from ethyl acetate-petroleum ether. This gives the 5,6-di-p-methoxyphenylimidazo [2,1-b] dihydro-thiazole sulfone of mp 186-187 °.
35 g 2-[N-(1,2-Bis-p-methoxypheny 1-2-oxo-äthyl)-aminothiazolin (vgl. Beispiel 7) werden zusammen mit 250 ml Isopropanol und 1 ml Bromwasserstoff (48%-ig) 2 Stunden am Rückfluss gehalten. Durch Abkühlen im Eisbad und Abnutschen des Produktes erhält man das 5,6-Di-(p-methoxyphenyl)-imidazol [2,1-b]dihydro-thiazol vom F. 155-156°.35 g of 2- [N- (1,2-bis-p-methoxypheny 1-2-oxo-ethyl) aminothiazoline (see Example 7) together with 250 ml of isopropanol and 1 ml of hydrogen bromide (48%) 2 Held at reflux for hours. Cooling in an ice bath and filtering off the product gives the 5,6-di- (p-methoxyphenyl) imidazole [2,1-b] dihydro-thiazole of mp 155-156 °.
Aus der Mutterlauge lässt sich das Hydrobromid des 5,6-Di-(p-methoxyphenyl)-imidazo[2,l-b]dihydro-thia- zols gewinnen, Smp. 200-210°.The hydrobromide of 5,6-di- (p-methoxyphenyl) imidazo [2, l-b] dihydro-thiazole can be obtained from the mother liquor, mp. 200-210 °.
Zu einer Suspension von 11,8 g 5,6-di-p-Anisyl-2,3,5,6-tetrahydro-4H-imidazo [2,1-b]-thiazol in 30 ml Wasser werden 17 ml 1-n Salzsäure und 5 g N-Benzolsulfo- nyl-L(+)-glutaminsäure zugegeben. Das Gemischwird durch Erwärmen gelöst und durch Abkühlen zur Kristallisation gebracht. Das L-5,6-di-p-Anisyl-2,3,5,6-tetrahydro-4H-imidazo [2,1-b]thiazol N-benzol-sulfonyl L(+)-glutamat wird abgenutscht, mit wenig Wasser gewaschen, in Wasser suspendiert und mit verdünnter Natronlauge zerlegt. Daraus wird das L-5,6-di-p-Anisyl-2,3,5,6-tetrahydro-4H-imidazo[2,1-b]thiazol mit Aethylacetat extrahiert.To a suspension of 11.8 g of 5,6-di-p-anisyl-2,3,5,6-tetrahydro-4H-imidazo [2,1-b] thiazole in 30 ml of water, 17 ml of 1-n Hydrochloric acid and 5 g N-benzenesulfo- nyl-L (+) - glutamic acid added. The mixture is dissolved by heating and crystallized by cooling. The L-5,6-di-p-anisyl-2,3,5,6-tetrahydro-4H-imidazo [2,1-b] thiazole N-benzenesulfonyl L (+) - glutamate is filtered off with a little Washed water, suspended in water and decomposed with dilute sodium hydroxide solution. From this, the L-5,6-di-p-anisyl-2,3,5,6-tetrahydro-4H-imidazo [2,1-b] thiazole is extracted with ethyl acetate.
Diese Bestandteile werden gesiebt, gut gemischt und die Mischung in Hargelatinekapseln gefüllt.These ingredients are sieved, mixed well and the mixture is filled into Hargelatine capsules.
Die Saccharose, das Calzium-dihydrat und der Wirkstoff werden gemischt und mit einer 10%-ige Gelatinelösung granuliert. Das feuchte Granulat wird gesiebt, getrocknet, mit der Stärke, dem Talkum und der Stearinsäure vermischt, gesiebt und zu Tabletten verpresst.The sucrose, calcium dihydrate and the active ingredient are mixed and granulated with a 10% gelatin solution. The moist granules are sieved, dried, mixed with the starch, talc and stearic acid, sieved and compressed into tablets.
Claims (15)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| LU77703 | 1977-07-07 | ||
| LU77703A LU77703A1 (en) | 1977-07-07 | 1977-07-07 | METHOD FOR PRODUCING BICYCLIC THIA-DIAZA COMPOUNDS |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP80101323A Division EP0019688A1 (en) | 1977-07-07 | 1978-06-29 | Diaza compounds and process for their preparation |
| EP80101323.6 Division-Into | 1980-03-13 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0000353A2 true EP0000353A2 (en) | 1979-01-24 |
| EP0000353A3 EP0000353A3 (en) | 1979-06-13 |
Family
ID=19728633
Family Applications (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP80101323A Ceased EP0019688A1 (en) | 1977-07-07 | 1978-06-29 | Diaza compounds and process for their preparation |
| EP80101322A Withdrawn EP0020858A1 (en) | 1977-07-07 | 1978-06-29 | Cyclic thiaaza compounds, process for their preparation, pharmaceutical preparations containing them and their use |
| EP78100272A Withdrawn EP0000353A3 (en) | 1977-07-07 | 1978-06-29 | Bicyclic thiadiaza compounds, process and intermediates for their preparation, and medicaments containing these compounds or the intermediates |
Family Applications Before (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP80101323A Ceased EP0019688A1 (en) | 1977-07-07 | 1978-06-29 | Diaza compounds and process for their preparation |
| EP80101322A Withdrawn EP0020858A1 (en) | 1977-07-07 | 1978-06-29 | Cyclic thiaaza compounds, process for their preparation, pharmaceutical preparations containing them and their use |
Country Status (18)
| Country | Link |
|---|---|
| EP (3) | EP0019688A1 (en) |
| JP (1) | JPS5416470A (en) |
| AR (2) | AR222309A1 (en) |
| AU (1) | AU3778878A (en) |
| DD (2) | DD145538A5 (en) |
| DK (1) | DK305578A (en) |
| ES (4) | ES471458A1 (en) |
| FI (1) | FI782132A7 (en) |
| GR (1) | GR64978B (en) |
| IL (1) | IL55086A0 (en) |
| IT (1) | IT8149647A0 (en) |
| LU (1) | LU77703A1 (en) |
| NO (1) | NO782357L (en) |
| NZ (1) | NZ187789A (en) |
| PL (1) | PL116596B1 (en) |
| PT (1) | PT68246A (en) |
| SU (4) | SU873886A3 (en) |
| ZA (1) | ZA783898B (en) |
Cited By (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0011111A1 (en) * | 1978-09-27 | 1980-05-28 | Smithkline Beckman Corporation | 5-(4-Pyridyl)-6-(4-fluorophenyl)-2,3-dihydroimidazo-(2,1-b)-thiazole, process for its preparation and medical composition |
| EP0013561A1 (en) * | 1979-01-05 | 1980-07-23 | Ciba-Geigy Ag | Levorotatory thia-diaza bicyclic derivative, process for its preparation and pharmaceutical compositions containing it |
| EP0004648B1 (en) * | 1978-04-11 | 1982-08-25 | Ciba-Geigy Ag | Mercapto-imidazole derivatives, their preparation, mercapto-imidazole derivatives for the treatment of inflammatory diseases and their pharmaceutical compositions |
| EP0005545B1 (en) * | 1978-05-24 | 1985-09-18 | Schering Aktiengesellschaft | Imidazole derivatives, process for their preparation and pharmaceutical compositions containing them |
| EP0164765A1 (en) * | 1981-02-19 | 1985-12-18 | Yamanouchi Pharmaceutical Co. Ltd. | 3,5-Di-Tert-Butyl-4-hydroxyphenyl-substituted heterocyclic compounds |
| EP0203787A3 (en) * | 1985-05-23 | 1988-05-25 | Smithkline Beckman Corporation | Process for preparing pyridyl-substituted imidazoû2,1-b¨thiazoles and thiazines |
| US4803279A (en) * | 1985-05-23 | 1989-02-07 | Smithkline Beckman Corporation | 1.4-dihydro-4-pyridyl-substituted imidazo (2,1-b) thiazoles and the corresponding thiazines. |
| EP0231622A3 (en) * | 1985-12-12 | 1989-05-24 | Smithkline Beckman Corporation | Inhibition of the 5-lipoxygenase pathway |
| EP0411754A3 (en) * | 1989-06-13 | 1991-03-13 | Smithkline Beecham Corporation | Medicament for the inhibition of interleukin-1 or tumor necrosis factor production by monocytes and/or macrophages |
| US5008390A (en) * | 1985-05-23 | 1991-04-16 | Smithkline Beckman Corporation | Compounds for preparing 6-phenyl-2,3-dihydroimidazo[2,1-b]-thiazoles and corresponding thiazines |
| WO1991013876A1 (en) * | 1990-03-16 | 1991-09-19 | Rhone-Poulenc Rorer Limited | Imidazoles |
| US5552422A (en) * | 1995-01-11 | 1996-09-03 | Merck Frosst Canada, Inc. | Aryl substituted 5,5 fused aromatic nitrogen compounds as anti-inflammatory agents |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4719218A (en) * | 1985-12-12 | 1988-01-12 | Smithkline Beckman Corporation | Pyrrolo[1,2-a]imidazole and pyrrolo[1,2-a]pyridine derivatives and their use as 5-lipoxygenase pathway inhibitor |
| GB9019838D0 (en) * | 1990-09-11 | 1990-10-24 | Smith Kline French Lab | Compounds |
| CA2063865A1 (en) * | 1992-03-24 | 1993-09-25 | Karoly Lempert | 2-(substituted imino)-thiazolidines and process for the preparation thereof |
| RU2405788C1 (en) * | 2009-07-14 | 2010-12-10 | Государственное образовательное учреждение высшего профессионального образования "БАШКИРСКИЙ ГОСУДАРСТВЕННЫЙ МЕДИЦИНСКИЙ УНИВЕРСИТЕТ ФЕДЕРАЛЬНОГО АГЕНТСТВА ПО ЗДРАВООХРАНЕНИЮ И СОЦИАЛЬНОМУ РАЗВИТИЮ" | 3-CYCLOHEXYLAMINOMETHYLTHIAZOLE[3,2-a]BENZIMIDAZOLE DIHYDROCHLORIDE HAVING IMMUNOTROPIC AND ANTI-AGGREGATION ACTIVITY |
Family Cites Families (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2981739A (en) * | 1958-01-27 | 1961-04-25 | Diamond Alkali Co | Certain alpha-haloaldehyde addition products of ethylenethioureas and process |
| NL131034C (en) * | 1964-05-11 | |||
| US3297708A (en) * | 1965-10-06 | 1967-01-10 | American Cyanamid Co | Method of preparing thiazolidines |
| US3455924A (en) * | 1967-02-08 | 1969-07-15 | Upjohn Co | Dianisylimidazoles |
| NL164285C (en) * | 1968-01-09 | 1980-12-15 | Rhone Poulenc Sa | PROCESS FOR THE PREPARATION OF A PHARMACEUTICAL PREPARATION CONTAINING AN IMIDAZOŸ2.1-BŸTHIAZOLE DERIVATIVE, THE PREPARED PHARMACEUTICAL PREPARATIONS AND A METHOD FOR PREPARING THIS IMIDAZOŸ2.1-BŸTHIAZOLDER |
| BE788792A (en) * | 1971-09-30 | 1973-03-13 | Ici Ltd | METHODS AND COMPOSITIONS FOR ANTI-BACTERIAL USE |
| US3842097A (en) * | 1973-01-22 | 1974-10-15 | Searle & Co | 2-(phenoxyalkylthio)imidazoles and congeners |
| SE428686B (en) * | 1975-08-11 | 1983-07-18 | Du Pont | PROCEDURE FOR PREPARING ANTI-INFLAMMATORALLY ACTIVE IMIDAZOLS |
| GB1541321A (en) * | 1976-03-10 | 1979-02-28 | Metabio | 2,3-dihydroimidazo(2,1-b)thiazole derivatives and process for their preparation |
| US4064260A (en) * | 1976-07-29 | 1977-12-20 | E. I. Du Pont De Nemours And Company | Anti-inflammatory diarylimidazothiazoles and their corresponding S-oxides |
| US4263311A (en) * | 1976-09-27 | 1981-04-21 | Smithkline Corporation | 5,6-Phenyl-2,3-dihydroimidazo [2,1-b] thiazoles |
-
1977
- 1977-07-07 LU LU77703A patent/LU77703A1/en unknown
-
1978
- 1978-06-29 EP EP80101323A patent/EP0019688A1/en not_active Ceased
- 1978-06-29 EP EP80101322A patent/EP0020858A1/en not_active Withdrawn
- 1978-06-29 EP EP78100272A patent/EP0000353A3/en not_active Withdrawn
- 1978-07-03 PT PT68246A patent/PT68246A/en unknown
- 1978-07-03 FI FI782132A patent/FI782132A7/en not_active Application Discontinuation
- 1978-07-05 ES ES471458A patent/ES471458A1/en not_active Expired
- 1978-07-05 AU AU37788/78A patent/AU3778878A/en active Pending
- 1978-07-05 IL IL55086A patent/IL55086A0/en unknown
- 1978-07-05 GR GR56695A patent/GR64978B/en unknown
- 1978-07-05 AR AR272843A patent/AR222309A1/en active
- 1978-07-06 NO NO782357A patent/NO782357L/en unknown
- 1978-07-06 DK DK783055A patent/DK305578A/en unknown
- 1978-07-06 ZA ZA00783898A patent/ZA783898B/en unknown
- 1978-07-06 SU SU782632647A patent/SU873886A3/en active
- 1978-07-06 NZ NZ187789A patent/NZ187789A/en unknown
- 1978-07-06 JP JP8153378A patent/JPS5416470A/en active Pending
- 1978-07-06 DD DD78214990A patent/DD145538A5/en unknown
- 1978-07-06 DD DD78206565A patent/DD138212A5/en unknown
- 1978-07-07 PL PL1978208253A patent/PL116596B1/en unknown
-
1979
- 1979-03-05 ES ES478296A patent/ES478296A1/en not_active Expired
- 1979-05-18 SU SU792763599A patent/SU893134A3/en active
- 1979-09-10 ES ES484033A patent/ES484033A1/en not_active Expired
- 1979-09-20 ES ES484327A patent/ES484327A1/en not_active Expired
- 1979-10-23 SU SU792831085A patent/SU850007A3/en active
- 1979-12-03 AR AR279136A patent/AR224381A1/en active
- 1979-12-20 SU SU792855458A patent/SU873887A3/en active
-
1981
- 1981-11-06 IT IT8149647A patent/IT8149647A0/en unknown
Cited By (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0004648B1 (en) * | 1978-04-11 | 1982-08-25 | Ciba-Geigy Ag | Mercapto-imidazole derivatives, their preparation, mercapto-imidazole derivatives for the treatment of inflammatory diseases and their pharmaceutical compositions |
| EP0005545B1 (en) * | 1978-05-24 | 1985-09-18 | Schering Aktiengesellschaft | Imidazole derivatives, process for their preparation and pharmaceutical compositions containing them |
| EP0011111A1 (en) * | 1978-09-27 | 1980-05-28 | Smithkline Beckman Corporation | 5-(4-Pyridyl)-6-(4-fluorophenyl)-2,3-dihydroimidazo-(2,1-b)-thiazole, process for its preparation and medical composition |
| EP0013561A1 (en) * | 1979-01-05 | 1980-07-23 | Ciba-Geigy Ag | Levorotatory thia-diaza bicyclic derivative, process for its preparation and pharmaceutical compositions containing it |
| EP0013560A1 (en) * | 1979-01-05 | 1980-07-23 | Ciba-Geigy Ag | Bicyclic dextrorotatory thiadiaza compound, process for its preparation, pharmaceutical compositions containing it and its use |
| EP0059090B1 (en) * | 1981-02-19 | 1986-01-29 | Yamanouchi Pharmaceutical Co., Ltd. | 3,5-di-tert-butyl-4-hydroxyphenyl-substituted heterocyclic compounds |
| EP0164765A1 (en) * | 1981-02-19 | 1985-12-18 | Yamanouchi Pharmaceutical Co. Ltd. | 3,5-Di-Tert-Butyl-4-hydroxyphenyl-substituted heterocyclic compounds |
| EP0203787A3 (en) * | 1985-05-23 | 1988-05-25 | Smithkline Beckman Corporation | Process for preparing pyridyl-substituted imidazoû2,1-b¨thiazoles and thiazines |
| US4803279A (en) * | 1985-05-23 | 1989-02-07 | Smithkline Beckman Corporation | 1.4-dihydro-4-pyridyl-substituted imidazo (2,1-b) thiazoles and the corresponding thiazines. |
| US5008390A (en) * | 1985-05-23 | 1991-04-16 | Smithkline Beckman Corporation | Compounds for preparing 6-phenyl-2,3-dihydroimidazo[2,1-b]-thiazoles and corresponding thiazines |
| EP0231622A3 (en) * | 1985-12-12 | 1989-05-24 | Smithkline Beckman Corporation | Inhibition of the 5-lipoxygenase pathway |
| EP0411754A3 (en) * | 1989-06-13 | 1991-03-13 | Smithkline Beecham Corporation | Medicament for the inhibition of interleukin-1 or tumor necrosis factor production by monocytes and/or macrophages |
| WO1991013876A1 (en) * | 1990-03-16 | 1991-09-19 | Rhone-Poulenc Rorer Limited | Imidazoles |
| US5552422A (en) * | 1995-01-11 | 1996-09-03 | Merck Frosst Canada, Inc. | Aryl substituted 5,5 fused aromatic nitrogen compounds as anti-inflammatory agents |
Also Published As
| Publication number | Publication date |
|---|---|
| ZA783898B (en) | 1979-07-25 |
| ES471458A1 (en) | 1979-10-01 |
| NZ187789A (en) | 1981-03-16 |
| EP0000353A3 (en) | 1979-06-13 |
| PL116596B1 (en) | 1981-06-30 |
| PT68246A (en) | 1978-08-01 |
| SU850007A3 (en) | 1981-07-23 |
| ES478296A1 (en) | 1979-05-16 |
| SU873886A3 (en) | 1981-10-15 |
| NO782357L (en) | 1979-01-09 |
| DD138212A5 (en) | 1979-10-17 |
| IL55086A0 (en) | 1978-09-29 |
| DK305578A (en) | 1979-01-08 |
| FI782132A7 (en) | 1979-01-08 |
| EP0019688A1 (en) | 1980-12-10 |
| EP0020858A1 (en) | 1981-01-07 |
| ES484327A1 (en) | 1980-05-16 |
| JPS5416470A (en) | 1979-02-07 |
| AU3778878A (en) | 1980-01-10 |
| ES484033A1 (en) | 1980-04-16 |
| LU77703A1 (en) | 1979-03-26 |
| SU873887A3 (en) | 1981-10-15 |
| PL208253A1 (en) | 1979-06-04 |
| DD145538A5 (en) | 1980-12-17 |
| SU893134A3 (en) | 1981-12-23 |
| AR222309A1 (en) | 1981-05-15 |
| AR224381A1 (en) | 1981-11-30 |
| GR64978B (en) | 1980-06-11 |
| IT8149647A0 (en) | 1981-11-06 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0000353A2 (en) | Bicyclic thiadiaza compounds, process and intermediates for their preparation, and medicaments containing these compounds or the intermediates | |
| EP0353557A1 (en) | Tetrahydro-1-benz-[c,d]-indolepropionic-acid sulfon amides | |
| EP0001279A1 (en) | Imidazole derivatives their synthesis and their pharmaceutical formulations | |
| EP0004648B1 (en) | Mercapto-imidazole derivatives, their preparation, mercapto-imidazole derivatives for the treatment of inflammatory diseases and their pharmaceutical compositions | |
| EP0114270B1 (en) | Acyl derivatives of 1,4:3,6-dianhydro-hexitoles, method for their preparation and their pharmazeutical use | |
| EP0005545B1 (en) | Imidazole derivatives, process for their preparation and pharmaceutical compositions containing them | |
| DE69006872T2 (en) | 1H, 3H, 7-pyrrolo [1,2-c] thiazolecarboxamide derivatives, their preparation and pharmaceutical preparations containing them. | |
| EP0053767B1 (en) | Tricyclic cytosine derivatives for use in pharmaceutical preparations and process for their preparation | |
| EP0008652B1 (en) | New intermediates and their use for the preparation of new oxadiazolo-pyrimidine derivatives | |
| US4134900A (en) | 5,6-Dihydro-4-oxo-4H-thieno[2,3-b]thiopyran-5-carboxamides, and process for the preparation thereof | |
| CS256400B2 (en) | Process for preparing novel pyridylmethylsulfinylthieno (2,3-d) imidazole derivatives | |
| FR2539417A1 (en) | NEW PYRROLO-1, 2 HETEROCYCLES, THEIR PREPARATION AND THE MEDICINAL PRODUCTS CONTAINING THEM | |
| GB2039882A (en) | Bicyclic thiadiaza compounds and their use as medicaments | |
| EP0163842A1 (en) | Tricyclic imidazole derivatives | |
| EP0211157B1 (en) | Isoxazole derivatives, process for their preparation and phamaceutical compositions containing them | |
| EP0242767B1 (en) | N-dihydroindolyl ethyl-sulfon amides | |
| US4164579A (en) | Hydroxythiazolidine-2-thiones | |
| AT360526B (en) | METHOD FOR PRODUCING NEW BICYCLIC THIA-DIAZA COMPOUNDS AND THEIR SALTS AND OPT. ECT. ISOMERS | |
| DD201788A5 (en) | PROCESS FOR PREPARING NEW DIARYL-IMIDAZOLE COMPOUNDS | |
| DE60013607T2 (en) | BEMZIMIDAZOL COMPOUNDS AND MEDICAMENTS CONTAINING THEM | |
| CZ695188A3 (en) | ENOL ETHER OF 1,1-DIOXIDE OF 6-CHLORO-4-HYDROXY-2-METHYL-N- (2-PYRIDYL-2H-THIENO(2,3-c)-1,2-THIAZINECARBOXYLIC ACID AMIDE, PROCESS OF ITS PREPARATION AND ITS USE | |
| DE69117578T2 (en) | Delta-2 cephem sulfones | |
| US4353918A (en) | Dextrorotatory bicyclic thiadiaza compounds and pharmaceutical preparations containing them | |
| EP0371342A1 (en) | 2-Halogen-substituted N-indolyl sulfonic-acid amides, process for their preparation and their pharmaceutical use | |
| NO783529L (en) | NEW TIAZOLIN DERIVATIVES. |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Designated state(s): BE CH DE FR GB NL SE |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Designated state(s): BE CH DE FR GB NL SE |
|
| 17P | Request for examination filed | ||
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE APPLICATION IS DEEMED TO BE WITHDRAWN |
|
| 18D | Application deemed to be withdrawn |
Effective date: 19821126 |
|
| RIN1 | Information on inventor provided before grant (corrected) |
Inventor name: FERRINI, PIER GIORGIO, DR. Inventor name: GOESCHKE, RICHARD, DR. |