CN1457358A - Aqueous heavy duty liquid detergent compositions comprising modified alkylbenzene sulfonates - Google Patents
Aqueous heavy duty liquid detergent compositions comprising modified alkylbenzene sulfonates Download PDFInfo
- Publication number
- CN1457358A CN1457358A CN 99815763 CN99815763A CN1457358A CN 1457358 A CN1457358 A CN 1457358A CN 99815763 CN99815763 CN 99815763 CN 99815763 A CN99815763 A CN 99815763A CN 1457358 A CN1457358 A CN 1457358A
- Authority
- CN
- China
- Prior art keywords
- mixture
- carbon atoms
- alkylbenzene sulfonate
- surfactant
- composition
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
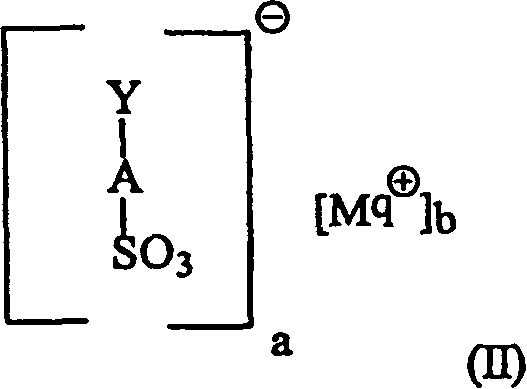
- 239000000203 mixture Substances 0.000 title claims abstract description 686
- -1 alkylbenzene sulfonates Chemical class 0.000 title claims abstract description 171
- 239000003599 detergent Substances 0.000 title claims description 141
- 239000007788 liquid Substances 0.000 title claims description 60
- 239000004094 surface-active agent Substances 0.000 claims abstract description 205
- 125000004432 carbon atom Chemical group C* 0.000 claims description 131
- 229910052799 carbon Inorganic materials 0.000 claims description 112
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 85
- 125000000217 alkyl group Chemical group 0.000 claims description 84
- 239000003795 chemical substances by application Substances 0.000 claims description 77
- 238000000034 method Methods 0.000 claims description 77
- 238000005804 alkylation reaction Methods 0.000 claims description 76
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 claims description 74
- 239000003054 catalyst Substances 0.000 claims description 73
- 239000010457 zeolite Substances 0.000 claims description 72
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 68
- 229910052739 hydrogen Inorganic materials 0.000 claims description 55
- 230000029936 alkylation Effects 0.000 claims description 54
- 239000001257 hydrogen Substances 0.000 claims description 47
- 150000001336 alkenes Chemical class 0.000 claims description 45
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 claims description 43
- 150000003839 salts Chemical class 0.000 claims description 43
- 150000001875 compounds Chemical class 0.000 claims description 32
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 31
- 229910052680 mordenite Inorganic materials 0.000 claims description 29
- 125000001931 aliphatic group Chemical group 0.000 claims description 27
- 239000012634 fragment Substances 0.000 claims description 26
- 229910052708 sodium Inorganic materials 0.000 claims description 26
- 239000002904 solvent Substances 0.000 claims description 26
- 230000002378 acidificating effect Effects 0.000 claims description 23
- 102000004190 Enzymes Human genes 0.000 claims description 22
- 108090000790 Enzymes Proteins 0.000 claims description 22
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 claims description 16
- 150000001768 cations Chemical class 0.000 claims description 15
- 235000014113 dietary fatty acids Nutrition 0.000 claims description 15
- 239000000194 fatty acid Substances 0.000 claims description 15
- 229930195729 fatty acid Natural products 0.000 claims description 15
- 150000002431 hydrogen Chemical class 0.000 claims description 15
- 239000002243 precursor Substances 0.000 claims description 15
- 238000004061 bleaching Methods 0.000 claims description 14
- AKEJUJNQAAGONA-UHFFFAOYSA-N sulfur trioxide Chemical compound O=S(=O)=O AKEJUJNQAAGONA-UHFFFAOYSA-N 0.000 claims description 14
- 150000004665 fatty acids Chemical class 0.000 claims description 13
- 239000004064 cosurfactant Substances 0.000 claims description 12
- 230000008569 process Effects 0.000 claims description 12
- 239000003945 anionic surfactant Substances 0.000 claims description 11
- 125000001183 hydrocarbyl group Chemical group 0.000 claims description 11
- 229910052700 potassium Inorganic materials 0.000 claims description 11
- 239000007787 solid Substances 0.000 claims description 11
- 239000012190 activator Substances 0.000 claims description 10
- 239000007844 bleaching agent Substances 0.000 claims description 10
- JRZJOMJEPLMPRA-UHFFFAOYSA-N olefin Natural products CCCCCCCC=C JRZJOMJEPLMPRA-UHFFFAOYSA-N 0.000 claims description 10
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims description 8
- 229910052760 oxygen Inorganic materials 0.000 claims description 8
- 239000001301 oxygen Substances 0.000 claims description 8
- SRSXLGNVWSONIS-UHFFFAOYSA-M benzenesulfonate Chemical compound [O-]S(=O)(=O)C1=CC=CC=C1 SRSXLGNVWSONIS-UHFFFAOYSA-M 0.000 claims description 7
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 7
- 125000003158 alcohol group Chemical group 0.000 claims description 6
- 150000001335 aliphatic alkanes Chemical class 0.000 claims description 6
- 239000002270 dispersing agent Substances 0.000 claims description 6
- QUCDWLYKDRVKMI-UHFFFAOYSA-M sodium;3,4-dimethylbenzenesulfonate Chemical compound [Na+].CC1=CC=C(S([O-])(=O)=O)C=C1C QUCDWLYKDRVKMI-UHFFFAOYSA-M 0.000 claims description 6
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 claims description 5
- 150000008051 alkyl sulfates Chemical class 0.000 claims description 5
- 239000004711 α-olefin Substances 0.000 claims description 5
- VSCWAEJMTAWNJL-UHFFFAOYSA-K aluminium trichloride Chemical compound Cl[Al](Cl)Cl VSCWAEJMTAWNJL-UHFFFAOYSA-K 0.000 claims description 4
- 125000004106 butoxy group Chemical group [*]OC([H])([H])C([H])([H])C(C([H])([H])[H])([H])[H] 0.000 claims description 4
- 150000003138 primary alcohols Chemical class 0.000 claims description 4
- 230000035484 reaction time Effects 0.000 claims description 4
- 230000003472 neutralizing effect Effects 0.000 claims description 3
- 150000001298 alcohols Chemical class 0.000 claims description 2
- 150000005673 monoalkenes Chemical class 0.000 claims 7
- 239000002168 alkylating agent Substances 0.000 claims 5
- 229940100198 alkylating agent Drugs 0.000 claims 5
- 239000003752 hydrotrope Substances 0.000 claims 5
- 230000007935 neutral effect Effects 0.000 claims 4
- 125000000129 anionic group Chemical group 0.000 claims 3
- 239000002736 nonionic surfactant Substances 0.000 claims 3
- 125000001424 substituent group Chemical group 0.000 claims 3
- 125000002015 acyclic group Chemical group 0.000 claims 2
- 125000001570 methylene group Chemical group [H]C([H])([*:1])[*:2] 0.000 claims 2
- GOKVKLCCWGRQJV-UHFFFAOYSA-N 2-[6-(decanoylamino)hexanoyloxy]benzenesulfonic acid Chemical compound CCCCCCCCCC(=O)NCCCCCC(=O)OC1=CC=CC=C1S(O)(=O)=O GOKVKLCCWGRQJV-UHFFFAOYSA-N 0.000 claims 1
- ISBYGXCCBJIBCG-UHFFFAOYSA-N 2-[6-(nonanoylamino)hexanoyloxy]benzenesulfonic acid Chemical compound CCCCCCCCC(=O)NCCCCCC(=O)OC1=CC=CC=C1S(O)(=O)=O ISBYGXCCBJIBCG-UHFFFAOYSA-N 0.000 claims 1
- JKZLOWDYIRTRJZ-UHFFFAOYSA-N 2-[6-(octanoylamino)hexanoyloxy]benzenesulfonic acid Chemical group CCCCCCCC(=O)NCCCCCC(=O)OC1=CC=CC=C1S(O)(=O)=O JKZLOWDYIRTRJZ-UHFFFAOYSA-N 0.000 claims 1
- AVLQNPBLHZMWFC-UHFFFAOYSA-N 6-(nonylamino)-6-oxohexaneperoxoic acid Chemical compound CCCCCCCCCNC(=O)CCCCC(=O)OO AVLQNPBLHZMWFC-UHFFFAOYSA-N 0.000 claims 1
- 239000000654 additive Substances 0.000 claims 1
- 239000002280 amphoteric surfactant Substances 0.000 claims 1
- 125000004429 atom Chemical group 0.000 claims 1
- 239000012876 carrier material Substances 0.000 claims 1
- 239000003093 cationic surfactant Substances 0.000 claims 1
- 239000007809 chemical reaction catalyst Substances 0.000 claims 1
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 claims 1
- 150000002191 fatty alcohols Chemical class 0.000 claims 1
- 125000001165 hydrophobic group Chemical group 0.000 claims 1
- 125000002572 propoxy group Chemical group [*]OC([H])([H])C(C([H])([H])[H])([H])[H] 0.000 claims 1
- 150000003467 sulfuric acid derivatives Chemical class 0.000 claims 1
- 239000002888 zwitterionic surfactant Substances 0.000 claims 1
- 238000004140 cleaning Methods 0.000 abstract description 17
- 150000004996 alkyl benzenes Chemical class 0.000 description 151
- 239000002253 acid Substances 0.000 description 89
- HNPSIPDUKPIQMN-UHFFFAOYSA-N dioxosilane;oxo(oxoalumanyloxy)alumane Chemical compound O=[Si]=O.O=[Al]O[Al]=O HNPSIPDUKPIQMN-UHFFFAOYSA-N 0.000 description 74
- 239000000463 material Substances 0.000 description 74
- 238000005406 washing Methods 0.000 description 73
- 229910021536 Zeolite Inorganic materials 0.000 description 68
- 229940077388 benzenesulfonate Drugs 0.000 description 56
- 150000001721 carbon Chemical group 0.000 description 50
- 239000000047 product Substances 0.000 description 48
- 239000000243 solution Substances 0.000 description 41
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 39
- 239000013543 active substance Substances 0.000 description 37
- 239000000126 substance Substances 0.000 description 37
- 239000011734 sodium Substances 0.000 description 33
- 238000002360 preparation method Methods 0.000 description 32
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 31
- 150000001412 amines Chemical class 0.000 description 31
- 238000004128 high performance liquid chromatography Methods 0.000 description 31
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 30
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 30
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 29
- 229920000642 polymer Polymers 0.000 description 28
- 239000000523 sample Substances 0.000 description 28
- 238000006277 sulfonation reaction Methods 0.000 description 28
- 239000002585 base Substances 0.000 description 27
- 239000000975 dye Substances 0.000 description 27
- 150000002500 ions Chemical class 0.000 description 26
- 238000006243 chemical reaction Methods 0.000 description 25
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 23
- 229940088598 enzyme Drugs 0.000 description 21
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 20
- 238000005516 engineering process Methods 0.000 description 20
- 230000000694 effects Effects 0.000 description 19
- 239000003352 sequestering agent Substances 0.000 description 19
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 18
- 230000003287 optical effect Effects 0.000 description 18
- KHBQMWCZKVMBLN-UHFFFAOYSA-N Benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1 KHBQMWCZKVMBLN-UHFFFAOYSA-N 0.000 description 17
- 101000605014 Homo sapiens Putative L-type amino acid transporter 1-like protein MLAS Proteins 0.000 description 17
- 238000005481 NMR spectroscopy Methods 0.000 description 17
- 102100038206 Putative L-type amino acid transporter 1-like protein MLAS Human genes 0.000 description 17
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical group [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 17
- 239000012535 impurity Substances 0.000 description 17
- 239000012071 phase Substances 0.000 description 17
- BPQQTUXANYXVAA-UHFFFAOYSA-N Orthosilicate Chemical compound [O-][Si]([O-])([O-])[O-] BPQQTUXANYXVAA-UHFFFAOYSA-N 0.000 description 15
- 239000011541 reaction mixture Substances 0.000 description 14
- 230000002087 whitening effect Effects 0.000 description 14
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 13
- 238000004821 distillation Methods 0.000 description 13
- 239000004744 fabric Substances 0.000 description 13
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 12
- LSNNMFCWUKXFEE-UHFFFAOYSA-M Bisulfite Chemical compound OS([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-M 0.000 description 12
- 239000011572 manganese Substances 0.000 description 12
- 238000004519 manufacturing process Methods 0.000 description 12
- 238000003756 stirring Methods 0.000 description 12
- 238000012546 transfer Methods 0.000 description 12
- NIXOWILDQLNWCW-UHFFFAOYSA-N Acrylic acid Chemical compound OC(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 11
- 150000002632 lipids Chemical class 0.000 description 11
- 159000000000 sodium salts Chemical class 0.000 description 11
- 238000012360 testing method Methods 0.000 description 11
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 10
- 229910052728 basic metal Inorganic materials 0.000 description 10
- 150000003818 basic metals Chemical group 0.000 description 10
- 229960004106 citric acid Drugs 0.000 description 10
- 239000011521 glass Substances 0.000 description 10
- 239000000178 monomer Substances 0.000 description 10
- 238000012545 processing Methods 0.000 description 10
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 9
- 101150117004 atg18 gene Proteins 0.000 description 9
- 239000006185 dispersion Substances 0.000 description 9
- DECIPOUIJURFOJ-UHFFFAOYSA-N ethoxyquin Chemical compound N1C(C)(C)C=C(C)C2=CC(OCC)=CC=C21 DECIPOUIJURFOJ-UHFFFAOYSA-N 0.000 description 9
- 238000010438 heat treatment Methods 0.000 description 9
- 238000005342 ion exchange Methods 0.000 description 9
- 238000002156 mixing Methods 0.000 description 9
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 9
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 9
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 9
- 239000000377 silicon dioxide Substances 0.000 description 9
- BDHFUVZGWQCTTF-UHFFFAOYSA-M sulfonate Chemical compound [O-]S(=O)=O BDHFUVZGWQCTTF-UHFFFAOYSA-M 0.000 description 9
- 239000003643 water by type Substances 0.000 description 9
- 238000001644 13C nuclear magnetic resonance spectroscopy Methods 0.000 description 8
- 239000004258 Ethoxyquin Substances 0.000 description 8
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 8
- WQDUMFSSJAZKTM-UHFFFAOYSA-N Sodium methoxide Chemical compound [Na+].[O-]C WQDUMFSSJAZKTM-UHFFFAOYSA-N 0.000 description 8
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 8
- 239000003513 alkali Substances 0.000 description 8
- 229910052782 aluminium Inorganic materials 0.000 description 8
- 238000004458 analytical method Methods 0.000 description 8
- 150000001555 benzenes Chemical class 0.000 description 8
- 229940093500 ethoxyquin Drugs 0.000 description 8
- 235000019285 ethoxyquin Nutrition 0.000 description 8
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 8
- HZVOZRGWRWCICA-UHFFFAOYSA-N methanediyl Chemical compound [CH2] HZVOZRGWRWCICA-UHFFFAOYSA-N 0.000 description 8
- 239000011833 salt mixture Substances 0.000 description 8
- 235000012239 silicon dioxide Nutrition 0.000 description 8
- 229960001866 silicon dioxide Drugs 0.000 description 8
- 108010059892 Cellulase Proteins 0.000 description 7
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical group C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 7
- 230000015572 biosynthetic process Effects 0.000 description 7
- 230000008859 change Effects 0.000 description 7
- 239000003153 chemical reaction reagent Substances 0.000 description 7
- 239000004927 clay Substances 0.000 description 7
- 230000010354 integration Effects 0.000 description 7
- 230000014759 maintenance of location Effects 0.000 description 7
- 229920000768 polyamine Polymers 0.000 description 7
- 238000006116 polymerization reaction Methods 0.000 description 7
- 229910001220 stainless steel Inorganic materials 0.000 description 7
- 239000010935 stainless steel Substances 0.000 description 7
- 238000005303 weighing Methods 0.000 description 7
- ZXVONLUNISGICL-UHFFFAOYSA-N 4,6-dinitro-o-cresol Chemical compound CC1=CC([N+]([O-])=O)=CC([N+]([O-])=O)=C1O ZXVONLUNISGICL-UHFFFAOYSA-N 0.000 description 6
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 6
- 239000004382 Amylase Substances 0.000 description 6
- 108010065511 Amylases Proteins 0.000 description 6
- 102000013142 Amylases Human genes 0.000 description 6
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 6
- 108090001060 Lipase Proteins 0.000 description 6
- 102000004882 Lipase Human genes 0.000 description 6
- PWHULOQIROXLJO-UHFFFAOYSA-N Manganese Chemical compound [Mn] PWHULOQIROXLJO-UHFFFAOYSA-N 0.000 description 6
- QPCDCPDFJACHGM-UHFFFAOYSA-N N,N-bis{2-[bis(carboxymethyl)amino]ethyl}glycine Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(=O)O)CCN(CC(O)=O)CC(O)=O QPCDCPDFJACHGM-UHFFFAOYSA-N 0.000 description 6
- BCXBKOQDEOJNRH-UHFFFAOYSA-N NOP(O)=O Chemical class NOP(O)=O BCXBKOQDEOJNRH-UHFFFAOYSA-N 0.000 description 6
- UFWIBTONFRDIAS-UHFFFAOYSA-N Naphthalene Chemical class C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 6
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical class CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 6
- 108091005804 Peptidases Proteins 0.000 description 6
- 102000035195 Peptidases Human genes 0.000 description 6
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 6
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 6
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 6
- 239000004411 aluminium Substances 0.000 description 6
- 125000003368 amide group Chemical group 0.000 description 6
- 235000019418 amylase Nutrition 0.000 description 6
- 229940106157 cellulase Drugs 0.000 description 6
- RWGFKTVRMDUZSP-UHFFFAOYSA-N cumene Chemical class CC(C)C1=CC=CC=C1 RWGFKTVRMDUZSP-UHFFFAOYSA-N 0.000 description 6
- 239000000706 filtrate Substances 0.000 description 6
- 229910052748 manganese Inorganic materials 0.000 description 6
- 239000002245 particle Substances 0.000 description 6
- 125000000864 peroxy group Chemical group O(O*)* 0.000 description 6
- 239000011148 porous material Substances 0.000 description 6
- 238000007789 sealing Methods 0.000 description 6
- MWNQXXOSWHCCOZ-UHFFFAOYSA-L sodium;oxido carbonate Chemical compound [Na+].[O-]OC([O-])=O MWNQXXOSWHCCOZ-UHFFFAOYSA-L 0.000 description 6
- XTHPWXDJESJLNJ-UHFFFAOYSA-N sulfurochloridic acid Chemical compound OS(Cl)(=O)=O XTHPWXDJESJLNJ-UHFFFAOYSA-N 0.000 description 6
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 6
- 238000011282 treatment Methods 0.000 description 6
- AVWQQPYHYQKEIZ-UHFFFAOYSA-K trisodium;2-dodecylbenzenesulfonate;3-dodecylbenzenesulfonate;4-dodecylbenzenesulfonate Chemical compound [Na+].[Na+].[Na+].CCCCCCCCCCCCC1=CC=C(S([O-])(=O)=O)C=C1.CCCCCCCCCCCCC1=CC=CC(S([O-])(=O)=O)=C1.CCCCCCCCCCCCC1=CC=CC=C1S([O-])(=O)=O AVWQQPYHYQKEIZ-UHFFFAOYSA-K 0.000 description 6
- GAWIXWVDTYZWAW-UHFFFAOYSA-N C[CH]O Chemical group C[CH]O GAWIXWVDTYZWAW-UHFFFAOYSA-N 0.000 description 5
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 5
- 239000004367 Lipase Substances 0.000 description 5
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 5
- 229910000323 aluminium silicate Inorganic materials 0.000 description 5
- 125000003118 aryl group Chemical group 0.000 description 5
- 239000011324 bead Substances 0.000 description 5
- YUUVOSXMIOYUGG-UHFFFAOYSA-N benzenesulfonic acid;hydrate Chemical compound O.OS(=O)(=O)C1=CC=CC=C1.OS(=O)(=O)C1=CC=CC=C1 YUUVOSXMIOYUGG-UHFFFAOYSA-N 0.000 description 5
- 239000012141 concentrate Substances 0.000 description 5
- 230000006872 improvement Effects 0.000 description 5
- XEEYBQQBJWHFJM-UHFFFAOYSA-N iron Substances [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 5
- 235000019421 lipase Nutrition 0.000 description 5
- 239000011777 magnesium Substances 0.000 description 5
- 229910052749 magnesium Inorganic materials 0.000 description 5
- ZUHZZVMEUAUWHY-UHFFFAOYSA-N n,n-dimethylpropan-1-amine Chemical compound CCCN(C)C ZUHZZVMEUAUWHY-UHFFFAOYSA-N 0.000 description 5
- 229910052757 nitrogen Inorganic materials 0.000 description 5
- TWNQGVIAIRXVLR-UHFFFAOYSA-N oxo(oxoalumanyloxy)alumane Chemical compound O=[Al]O[Al]=O TWNQGVIAIRXVLR-UHFFFAOYSA-N 0.000 description 5
- 238000010992 reflux Methods 0.000 description 5
- 229910021653 sulphate ion Inorganic materials 0.000 description 5
- BCAUVGPOEXLTJD-UHFFFAOYSA-N (2-cyclohexyl-4,6-dinitrophenyl) acetate Chemical compound C1=C([N+]([O-])=O)C=C([N+]([O-])=O)C(OC(=O)C)=C1C1CCCCC1 BCAUVGPOEXLTJD-UHFFFAOYSA-N 0.000 description 4
- 125000000954 2-hydroxyethyl group Chemical group [H]C([*])([H])C([H])([H])O[H] 0.000 description 4
- YGUMVDWOQQJBGA-VAWYXSNFSA-N 5-[(4-anilino-6-morpholin-4-yl-1,3,5-triazin-2-yl)amino]-2-[(e)-2-[4-[(4-anilino-6-morpholin-4-yl-1,3,5-triazin-2-yl)amino]-2-sulfophenyl]ethenyl]benzenesulfonic acid Chemical compound C=1C=C(\C=C\C=2C(=CC(NC=3N=C(N=C(NC=4C=CC=CC=4)N=3)N3CCOCC3)=CC=2)S(O)(=O)=O)C(S(=O)(=O)O)=CC=1NC(N=C(N=1)N2CCOCC2)=NC=1NC1=CC=CC=C1 YGUMVDWOQQJBGA-VAWYXSNFSA-N 0.000 description 4
- KXDHJXZQYSOELW-UHFFFAOYSA-N Carbamic acid Chemical compound NC(O)=O KXDHJXZQYSOELW-UHFFFAOYSA-N 0.000 description 4
- 239000004215 Carbon black (E152) Substances 0.000 description 4
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 4
- 235000008733 Citrus aurantifolia Nutrition 0.000 description 4
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 4
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 4
- MHAJPDPJQMAIIY-UHFFFAOYSA-N Hydrogen peroxide Chemical compound OO MHAJPDPJQMAIIY-UHFFFAOYSA-N 0.000 description 4
- AVXURJPOCDRRFD-UHFFFAOYSA-N Hydroxylamine Chemical group ON AVXURJPOCDRRFD-UHFFFAOYSA-N 0.000 description 4
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 4
- 235000006508 Nelumbo nucifera Nutrition 0.000 description 4
- 240000002853 Nelumbo nucifera Species 0.000 description 4
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 4
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 4
- 229920002125 Sokalan® Polymers 0.000 description 4
- 235000011941 Tilia x europaea Nutrition 0.000 description 4
- 125000002490 anilino group Chemical group [H]N(*)C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 4
- 239000002738 chelating agent Substances 0.000 description 4
- 229910017052 cobalt Inorganic materials 0.000 description 4
- 239000010941 cobalt Substances 0.000 description 4
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 4
- 238000001816 cooling Methods 0.000 description 4
- 239000013078 crystal Substances 0.000 description 4
- 238000002425 crystallisation Methods 0.000 description 4
- 230000008025 crystallization Effects 0.000 description 4
- 230000001351 cycling effect Effects 0.000 description 4
- 150000002148 esters Chemical class 0.000 description 4
- 239000012065 filter cake Substances 0.000 description 4
- 238000011010 flushing procedure Methods 0.000 description 4
- 239000008187 granular material Substances 0.000 description 4
- 230000036571 hydration Effects 0.000 description 4
- 238000006703 hydration reaction Methods 0.000 description 4
- 229930195733 hydrocarbon Natural products 0.000 description 4
- 230000002209 hydrophobic effect Effects 0.000 description 4
- 239000004615 ingredient Substances 0.000 description 4
- 238000002347 injection Methods 0.000 description 4
- 239000007924 injection Substances 0.000 description 4
- LVHBHZANLOWSRM-UHFFFAOYSA-N itaconic acid Chemical compound OC(=O)CC(=C)C(O)=O LVHBHZANLOWSRM-UHFFFAOYSA-N 0.000 description 4
- 239000004571 lime Substances 0.000 description 4
- 239000007791 liquid phase Substances 0.000 description 4
- 239000011159 matrix material Substances 0.000 description 4
- 229910052751 metal Inorganic materials 0.000 description 4
- 239000002184 metal Substances 0.000 description 4
- 238000006386 neutralization reaction Methods 0.000 description 4
- 150000002892 organic cations Chemical class 0.000 description 4
- 238000012856 packing Methods 0.000 description 4
- 239000012188 paraffin wax Substances 0.000 description 4
- 229920001296 polysiloxane Polymers 0.000 description 4
- 239000011591 potassium Substances 0.000 description 4
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 4
- 239000002994 raw material Substances 0.000 description 4
- CXWXQJXEFPUFDZ-UHFFFAOYSA-N tetralin Chemical compound C1=CC=C2CCCCC2=C1 CXWXQJXEFPUFDZ-UHFFFAOYSA-N 0.000 description 4
- VKZRWSNIWNFCIQ-WDSKDSINSA-N (2s)-2-[2-[[(1s)-1,2-dicarboxyethyl]amino]ethylamino]butanedioic acid Chemical compound OC(=O)C[C@@H](C(O)=O)NCCN[C@H](C(O)=O)CC(O)=O VKZRWSNIWNFCIQ-WDSKDSINSA-N 0.000 description 3
- BZJTUOGZUKFLQT-UHFFFAOYSA-N 1,3,5,7-tetramethylcyclooctane Chemical group CC1CC(C)CC(C)CC(C)C1 BZJTUOGZUKFLQT-UHFFFAOYSA-N 0.000 description 3
- OSSNTDFYBPYIEC-UHFFFAOYSA-N 1-ethenylimidazole Chemical compound C=CN1C=CN=C1 OSSNTDFYBPYIEC-UHFFFAOYSA-N 0.000 description 3
- CNGYZEMWVAWWOB-VAWYXSNFSA-N 5-[[4-anilino-6-[bis(2-hydroxyethyl)amino]-1,3,5-triazin-2-yl]amino]-2-[(e)-2-[4-[[4-anilino-6-[bis(2-hydroxyethyl)amino]-1,3,5-triazin-2-yl]amino]-2-sulfophenyl]ethenyl]benzenesulfonic acid Chemical compound N=1C(NC=2C=C(C(\C=C\C=3C(=CC(NC=4N=C(N=C(NC=5C=CC=CC=5)N=4)N(CCO)CCO)=CC=3)S(O)(=O)=O)=CC=2)S(O)(=O)=O)=NC(N(CCO)CCO)=NC=1NC1=CC=CC=C1 CNGYZEMWVAWWOB-VAWYXSNFSA-N 0.000 description 3
- JBKVHLHDHHXQEQ-UHFFFAOYSA-N Caprolactam Natural products O=C1CCCCCN1 JBKVHLHDHHXQEQ-UHFFFAOYSA-N 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 3
- 229910020366 ClO 4 Inorganic materials 0.000 description 3
- QXNVGIXVLWOKEQ-UHFFFAOYSA-N Disodium Chemical class [Na][Na] QXNVGIXVLWOKEQ-UHFFFAOYSA-N 0.000 description 3
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 3
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical compound NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 description 3
- WHNWPMSKXPGLAX-UHFFFAOYSA-N N-Vinyl-2-pyrrolidone Chemical compound C=CN1CCCC1=O WHNWPMSKXPGLAX-UHFFFAOYSA-N 0.000 description 3
- 150000001204 N-oxides Chemical class 0.000 description 3
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 3
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 3
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 3
- 229910004298 SiO 2 Inorganic materials 0.000 description 3
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 3
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 3
- BGRWYDHXPHLNKA-UHFFFAOYSA-N Tetraacetylethylenediamine Chemical compound CC(=O)N(C(C)=O)CCN(C(C)=O)C(C)=O BGRWYDHXPHLNKA-UHFFFAOYSA-N 0.000 description 3
- 229910052783 alkali metal Inorganic materials 0.000 description 3
- 150000001447 alkali salts Chemical class 0.000 description 3
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 3
- 150000001342 alkaline earth metals Chemical class 0.000 description 3
- 125000003545 alkoxy group Chemical group 0.000 description 3
- 230000002152 alkylating effect Effects 0.000 description 3
- 229910021529 ammonia Inorganic materials 0.000 description 3
- 150000003863 ammonium salts Chemical class 0.000 description 3
- 239000012298 atmosphere Substances 0.000 description 3
- 229910021538 borax Inorganic materials 0.000 description 3
- 239000011575 calcium Substances 0.000 description 3
- 229910052791 calcium Inorganic materials 0.000 description 3
- 150000007942 carboxylates Chemical group 0.000 description 3
- 238000006555 catalytic reaction Methods 0.000 description 3
- 229920001577 copolymer Polymers 0.000 description 3
- 238000007334 copolymerization reaction Methods 0.000 description 3
- 238000005202 decontamination Methods 0.000 description 3
- 230000003588 decontaminative effect Effects 0.000 description 3
- 238000006356 dehydrogenation reaction Methods 0.000 description 3
- 238000001704 evaporation Methods 0.000 description 3
- 230000008020 evaporation Effects 0.000 description 3
- 235000019197 fats Nutrition 0.000 description 3
- 230000002349 favourable effect Effects 0.000 description 3
- 239000000835 fiber Substances 0.000 description 3
- 239000006260 foam Substances 0.000 description 3
- 235000011187 glycerol Nutrition 0.000 description 3
- 239000008233 hard water Substances 0.000 description 3
- 150000002430 hydrocarbons Chemical class 0.000 description 3
- GPRLSGONYQIRFK-UHFFFAOYSA-N hydron Chemical compound [H+] GPRLSGONYQIRFK-UHFFFAOYSA-N 0.000 description 3
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 3
- 239000003112 inhibitor Substances 0.000 description 3
- 229910052742 iron Inorganic materials 0.000 description 3
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical group O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 3
- FPYJFEHAWHCUMM-UHFFFAOYSA-N maleic anhydride Chemical compound O=C1OC(=O)C=C1 FPYJFEHAWHCUMM-UHFFFAOYSA-N 0.000 description 3
- 229910044991 metal oxide Inorganic materials 0.000 description 3
- 150000004706 metal oxides Chemical class 0.000 description 3
- XJRBAMWJDBPFIM-UHFFFAOYSA-N methyl vinyl ether Chemical group COC=C XJRBAMWJDBPFIM-UHFFFAOYSA-N 0.000 description 3
- MGFYIUFZLHCRTH-UHFFFAOYSA-N nitrilotriacetic acid Chemical compound OC(=O)CN(CC(O)=O)CC(O)=O MGFYIUFZLHCRTH-UHFFFAOYSA-N 0.000 description 3
- 239000002304 perfume Substances 0.000 description 3
- UEZVMMHDMIWARA-UHFFFAOYSA-M phosphonate Chemical compound [O-]P(=O)=O UEZVMMHDMIWARA-UHFFFAOYSA-M 0.000 description 3
- 229910052698 phosphorus Inorganic materials 0.000 description 3
- 239000011574 phosphorus Substances 0.000 description 3
- IEQIEDJGQAUEQZ-UHFFFAOYSA-N phthalocyanine Chemical compound N1C(N=C2C3=CC=CC=C3C(N=C3C4=CC=CC=C4C(=N4)N3)=N2)=C(C=CC=C2)C2=C1N=C1C2=CC=CC=C2C4=N1 IEQIEDJGQAUEQZ-UHFFFAOYSA-N 0.000 description 3
- 229920000728 polyester Polymers 0.000 description 3
- 238000005070 sampling Methods 0.000 description 3
- 239000011780 sodium chloride Substances 0.000 description 3
- NTHWMYGWWRZVTN-UHFFFAOYSA-N sodium silicate Chemical compound [Na+].[Na+].[O-][Si]([O-])=O NTHWMYGWWRZVTN-UHFFFAOYSA-N 0.000 description 3
- 239000004328 sodium tetraborate Substances 0.000 description 3
- 235000010339 sodium tetraborate Nutrition 0.000 description 3
- 235000019832 sodium triphosphate Nutrition 0.000 description 3
- YKLJGMBLPUQQOI-UHFFFAOYSA-M sodium;oxidooxy(oxo)borane Chemical compound [Na+].[O-]OB=O YKLJGMBLPUQQOI-UHFFFAOYSA-M 0.000 description 3
- 241000894007 species Species 0.000 description 3
- KDYFGRWQOYBRFD-UHFFFAOYSA-L succinate(2-) Chemical compound [O-]C(=O)CCC([O-])=O KDYFGRWQOYBRFD-UHFFFAOYSA-L 0.000 description 3
- FAGUFWYHJQFNRV-UHFFFAOYSA-N tetraethylenepentamine Chemical compound NCCNCCNCCNCCN FAGUFWYHJQFNRV-UHFFFAOYSA-N 0.000 description 3
- 230000008719 thickening Effects 0.000 description 3
- RYFMWSXOAZQYPI-UHFFFAOYSA-K trisodium phosphate Chemical compound [Na+].[Na+].[Na+].[O-]P([O-])([O-])=O RYFMWSXOAZQYPI-UHFFFAOYSA-K 0.000 description 3
- 238000009736 wetting Methods 0.000 description 3
- DNIAPMSPPWPWGF-GSVOUGTGSA-N (R)-(-)-Propylene glycol Chemical compound C[C@@H](O)CO DNIAPMSPPWPWGF-GSVOUGTGSA-N 0.000 description 2
- VQOXUMQBYILCKR-UHFFFAOYSA-N 1-Tridecene Chemical group CCCCCCCCCCCC=C VQOXUMQBYILCKR-UHFFFAOYSA-N 0.000 description 2
- MPJQXAIKMSKXBI-UHFFFAOYSA-N 2,7,9,14-tetraoxa-1,8-diazabicyclo[6.6.2]hexadecane-3,6,10,13-tetrone Chemical compound C1CN2OC(=O)CCC(=O)ON1OC(=O)CCC(=O)O2 MPJQXAIKMSKXBI-UHFFFAOYSA-N 0.000 description 2
- OSAPYGUYJFZUIZ-UHFFFAOYSA-N 2-(1,2-dicarboxyethoxy)propane-1,2,3-tricarboxylic acid Chemical compound OC(=O)CC(C(O)=O)OC(CC(O)=O)(CC(O)=O)C(O)=O OSAPYGUYJFZUIZ-UHFFFAOYSA-N 0.000 description 2
- SMZOUWXMTYCWNB-UHFFFAOYSA-N 2-(2-methoxy-5-methylphenyl)ethanamine Chemical compound COC1=CC=C(C)C=C1CCN SMZOUWXMTYCWNB-UHFFFAOYSA-N 0.000 description 2
- QQZOPKMRPOGIEB-UHFFFAOYSA-N 2-Oxohexane Chemical compound CCCCC(C)=O QQZOPKMRPOGIEB-UHFFFAOYSA-N 0.000 description 2
- JJJOZVFVARQUJV-UHFFFAOYSA-N 2-ethylhexylphosphonic acid Chemical compound CCCCC(CC)CP(O)(O)=O JJJOZVFVARQUJV-UHFFFAOYSA-N 0.000 description 2
- DRMYLINAGHHBNG-UHFFFAOYSA-N 2-oxo-3h-furan-4-carboxylic acid Chemical compound OC(=O)C1=COC(=O)C1 DRMYLINAGHHBNG-UHFFFAOYSA-N 0.000 description 2
- RUDWFJHKDHQWHO-UHFFFAOYSA-N 2-phenylpentadecane-1-sulfonic acid Chemical compound CCCCCCCCCCCCCC(CS(O)(=O)=O)C1=CC=CC=C1 RUDWFJHKDHQWHO-UHFFFAOYSA-N 0.000 description 2
- KWYJDIUEHHCHCZ-UHFFFAOYSA-N 3-[2-[bis(2-carboxyethyl)amino]ethyl-(2-carboxyethyl)amino]propanoic acid Chemical class OC(=O)CCN(CCC(O)=O)CCN(CCC(O)=O)CCC(O)=O KWYJDIUEHHCHCZ-UHFFFAOYSA-N 0.000 description 2
- FAGGUIDTQQXDSJ-UHFFFAOYSA-N 3-benzoylazepan-2-one Chemical compound C=1C=CC=CC=1C(=O)C1CCCCNC1=O FAGGUIDTQQXDSJ-UHFFFAOYSA-N 0.000 description 2
- GNJORAZPXCKMSY-UHFFFAOYSA-N 6,10-dimethylundecan-2-ol Chemical compound CC(C)CCCC(C)CCCC(C)O GNJORAZPXCKMSY-UHFFFAOYSA-N 0.000 description 2
- WZSBYAZGSDMUAR-UHFFFAOYSA-N 6-methyldodecan-6-ol Chemical compound CCCCCCC(C)(O)CCCCC WZSBYAZGSDMUAR-UHFFFAOYSA-N 0.000 description 2
- RMTFYBCGUKERHL-UHFFFAOYSA-N 6-methylundecan-6-ol Chemical compound CCCCCC(C)(O)CCCCC RMTFYBCGUKERHL-UHFFFAOYSA-N 0.000 description 2
- NIXOWILDQLNWCW-UHFFFAOYSA-M Acrylate Chemical compound [O-]C(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-M 0.000 description 2
- 229910017090 AlO 2 Inorganic materials 0.000 description 2
- 239000005995 Aluminium silicate Substances 0.000 description 2
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonia chloride Chemical compound [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 2
- NGGQSTYSOSIPIM-UHFFFAOYSA-L C(C)(=O)[O-].C(CCCC)N.[Co+2].C(C)(=O)[O-] Chemical compound C(C)(=O)[O-].C(CCCC)N.[Co+2].C(C)(=O)[O-] NGGQSTYSOSIPIM-UHFFFAOYSA-L 0.000 description 2
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 2
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 2
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 2
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 2
- WTEVQBCEXWBHNA-UHFFFAOYSA-N Citral Natural products CC(C)=CCCC(C)=CC=O WTEVQBCEXWBHNA-UHFFFAOYSA-N 0.000 description 2
- ROSDSFDQCJNGOL-UHFFFAOYSA-N Dimethylamine Chemical compound CNC ROSDSFDQCJNGOL-UHFFFAOYSA-N 0.000 description 2
- IMROMDMJAWUWLK-UHFFFAOYSA-N Ethenol Chemical compound OC=C IMROMDMJAWUWLK-UHFFFAOYSA-N 0.000 description 2
- 241000282326 Felis catus Species 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- IFVNATVJZVXOFC-UHFFFAOYSA-N OC(CCC(O)=O)=O.OC(CCC(O)=O)=O.O.O Chemical compound OC(CCC(O)=O)=O.OC(CCC(O)=O)=O.O.O IFVNATVJZVXOFC-UHFFFAOYSA-N 0.000 description 2
- 229920000805 Polyaspartic acid Polymers 0.000 description 2
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical group C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 2
- 239000004115 Sodium Silicate Substances 0.000 description 2
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 2
- PPBRXRYQALVLMV-UHFFFAOYSA-N Styrene Chemical compound C=CC1=CC=CC=C1 PPBRXRYQALVLMV-UHFFFAOYSA-N 0.000 description 2
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 2
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 2
- DPDMMXDBJGCCQC-UHFFFAOYSA-N [Na].[Cl] Chemical compound [Na].[Cl] DPDMMXDBJGCCQC-UHFFFAOYSA-N 0.000 description 2
- 125000000218 acetic acid group Chemical group C(C)(=O)* 0.000 description 2
- JTPLPDIKCDKODU-UHFFFAOYSA-N acetic acid;2-(2-aminoethylamino)ethanol Chemical compound CC(O)=O.CC(O)=O.CC(O)=O.NCCNCCO JTPLPDIKCDKODU-UHFFFAOYSA-N 0.000 description 2
- QTONSPKDOKVNBJ-UHFFFAOYSA-N acetic acid;n'-(2-aminoethyl)ethane-1,2-diamine Chemical compound CC(O)=O.CC(O)=O.CC(O)=O.CC(O)=O.CC(O)=O.NCCNCCN QTONSPKDOKVNBJ-UHFFFAOYSA-N 0.000 description 2
- 125000002252 acyl group Chemical group 0.000 description 2
- 238000013019 agitation Methods 0.000 description 2
- 150000001340 alkali metals Chemical group 0.000 description 2
- 125000003342 alkenyl group Chemical group 0.000 description 2
- 125000002877 alkyl aryl group Chemical group 0.000 description 2
- 235000012211 aluminium silicate Nutrition 0.000 description 2
- 239000011260 aqueous acid Substances 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- 239000012752 auxiliary agent Substances 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 238000006065 biodegradation reaction Methods 0.000 description 2
- 230000033228 biological regulation Effects 0.000 description 2
- KGBXLFKZBHKPEV-UHFFFAOYSA-N boric acid Chemical compound OB(O)O KGBXLFKZBHKPEV-UHFFFAOYSA-N 0.000 description 2
- 239000004327 boric acid Substances 0.000 description 2
- 238000001354 calcination Methods 0.000 description 2
- 125000002091 cationic group Chemical group 0.000 description 2
- 238000012512 characterization method Methods 0.000 description 2
- 239000007795 chemical reaction product Substances 0.000 description 2
- HNEGQIOMVPPMNR-IHWYPQMZSA-N citraconic acid Chemical compound OC(=O)C(/C)=C\C(O)=O HNEGQIOMVPPMNR-IHWYPQMZSA-N 0.000 description 2
- 229940018557 citraconic acid Drugs 0.000 description 2
- 229940043350 citral Drugs 0.000 description 2
- NSFKBZXCXCJZDQ-UHFFFAOYSA-N cumene;sodium Chemical compound [Na].CC(C)C1=CC=CC=C1 NSFKBZXCXCJZDQ-UHFFFAOYSA-N 0.000 description 2
- GHVNFZFCNZKVNT-UHFFFAOYSA-N decanoic acid Chemical compound CCCCCCCCCC(O)=O GHVNFZFCNZKVNT-UHFFFAOYSA-N 0.000 description 2
- 238000007872 degassing Methods 0.000 description 2
- 239000008367 deionised water Substances 0.000 description 2
- 229910021641 deionized water Inorganic materials 0.000 description 2
- 150000004683 dihydrates Chemical class 0.000 description 2
- 239000003085 diluting agent Substances 0.000 description 2
- 238000004090 dissolution Methods 0.000 description 2
- 238000009826 distribution Methods 0.000 description 2
- 229940075933 dithionate Drugs 0.000 description 2
- POULHZVOKOAJMA-UHFFFAOYSA-N dodecanoic acid Chemical compound CCCCCCCCCCCC(O)=O POULHZVOKOAJMA-UHFFFAOYSA-N 0.000 description 2
- DUYCTCQXNHFCSJ-UHFFFAOYSA-N dtpmp Chemical compound OP(=O)(O)CN(CP(O)(O)=O)CCN(CP(O)(=O)O)CCN(CP(O)(O)=O)CP(O)(O)=O DUYCTCQXNHFCSJ-UHFFFAOYSA-N 0.000 description 2
- 229940009662 edetate Drugs 0.000 description 2
- 238000010828 elution Methods 0.000 description 2
- 230000007613 environmental effect Effects 0.000 description 2
- 125000002573 ethenylidene group Chemical group [*]=C=C([H])[H] 0.000 description 2
- 238000007046 ethoxylation reaction Methods 0.000 description 2
- HCPOCMMGKBZWSJ-UHFFFAOYSA-N ethyl 3-hydrazinyl-3-oxopropanoate Chemical compound CCOC(=O)CC(=O)NN HCPOCMMGKBZWSJ-UHFFFAOYSA-N 0.000 description 2
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 2
- 229940071087 ethylenediamine disuccinate Drugs 0.000 description 2
- 238000001914 filtration Methods 0.000 description 2
- 239000000796 flavoring agent Substances 0.000 description 2
- 238000005187 foaming Methods 0.000 description 2
- 235000013355 food flavoring agent Nutrition 0.000 description 2
- 238000009472 formulation Methods 0.000 description 2
- 210000002196 fr. b Anatomy 0.000 description 2
- 210000003918 fraction a Anatomy 0.000 description 2
- 239000001530 fumaric acid Substances 0.000 description 2
- 238000004817 gas chromatography Methods 0.000 description 2
- WTEVQBCEXWBHNA-JXMROGBWSA-N geranial Chemical compound CC(C)=CCC\C(C)=C\C=O WTEVQBCEXWBHNA-JXMROGBWSA-N 0.000 description 2
- PSFDQSOCUJVVGF-UHFFFAOYSA-N harman Chemical compound C12=CC=CC=C2NC2=C1C=CN=C2C PSFDQSOCUJVVGF-UHFFFAOYSA-N 0.000 description 2
- CATSNJVOTSVZJV-UHFFFAOYSA-N heptan-2-one Chemical compound CCCCCC(C)=O CATSNJVOTSVZJV-UHFFFAOYSA-N 0.000 description 2
- FUZZWVXGSFPDMH-UHFFFAOYSA-N hexanoic acid Chemical compound CCCCCC(O)=O FUZZWVXGSFPDMH-UHFFFAOYSA-N 0.000 description 2
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 2
- ILHIHKRJJMKBEE-UHFFFAOYSA-N hydroperoxyethane Chemical compound CCOO ILHIHKRJJMKBEE-UHFFFAOYSA-N 0.000 description 2
- 239000005457 ice water Substances 0.000 description 2
- VKOBVWXKNCXXDE-UHFFFAOYSA-N icosanoic acid Chemical compound CCCCCCCCCCCCCCCCCCCC(O)=O VKOBVWXKNCXXDE-UHFFFAOYSA-N 0.000 description 2
- 229910010272 inorganic material Inorganic materials 0.000 description 2
- 229910052809 inorganic oxide Inorganic materials 0.000 description 2
- 238000004811 liquid chromatography Methods 0.000 description 2
- TWRXJAOTZQYOKJ-UHFFFAOYSA-L magnesium chloride Substances [Mg+2].[Cl-].[Cl-] TWRXJAOTZQYOKJ-UHFFFAOYSA-L 0.000 description 2
- 239000000395 magnesium oxide Substances 0.000 description 2
- CPLXHLVBOLITMK-UHFFFAOYSA-N magnesium oxide Inorganic materials [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 description 2
- 238000012423 maintenance Methods 0.000 description 2
- 239000011976 maleic acid Substances 0.000 description 2
- 238000001819 mass spectrum Methods 0.000 description 2
- 230000010534 mechanism of action Effects 0.000 description 2
- YDSWCNNOKPMOTP-UHFFFAOYSA-N mellitic acid Chemical compound OC(=O)C1=C(C(O)=O)C(C(O)=O)=C(C(O)=O)C(C(O)=O)=C1C(O)=O YDSWCNNOKPMOTP-UHFFFAOYSA-N 0.000 description 2
- HNEGQIOMVPPMNR-NSCUHMNNSA-N mesaconic acid Chemical compound OC(=O)C(/C)=C/C(O)=O HNEGQIOMVPPMNR-NSCUHMNNSA-N 0.000 description 2
- NCPHGZWGGANCAY-UHFFFAOYSA-N methane;ruthenium Chemical compound C.[Ru] NCPHGZWGGANCAY-UHFFFAOYSA-N 0.000 description 2
- HNEGQIOMVPPMNR-UHFFFAOYSA-N methylfumaric acid Natural products OC(=O)C(C)=CC(O)=O HNEGQIOMVPPMNR-UHFFFAOYSA-N 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 239000010813 municipal solid waste Substances 0.000 description 2
- 125000004433 nitrogen atom Chemical group N* 0.000 description 2
- 230000003647 oxidation Effects 0.000 description 2
- 238000007254 oxidation reaction Methods 0.000 description 2
- 125000000913 palmityl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- XNLICIUVMPYHGG-UHFFFAOYSA-N pentan-2-one Chemical compound CCCC(C)=O XNLICIUVMPYHGG-UHFFFAOYSA-N 0.000 description 2
- 229960003330 pentetic acid Drugs 0.000 description 2
- XUWHAWMETYGRKB-UHFFFAOYSA-N piperidin-2-one Chemical compound O=C1CCCCN1 XUWHAWMETYGRKB-UHFFFAOYSA-N 0.000 description 2
- 229920000058 polyacrylate Polymers 0.000 description 2
- 108010064470 polyaspartate Proteins 0.000 description 2
- 235000019353 potassium silicate Nutrition 0.000 description 2
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 2
- 238000000746 purification Methods 0.000 description 2
- 150000003254 radicals Chemical class 0.000 description 2
- 229910052761 rare earth metal Inorganic materials 0.000 description 2
- 150000002910 rare earth metals Chemical class 0.000 description 2
- 239000000376 reactant Substances 0.000 description 2
- 239000012925 reference material Substances 0.000 description 2
- 230000000452 restraining effect Effects 0.000 description 2
- 238000002390 rotary evaporation Methods 0.000 description 2
- 239000012488 sample solution Substances 0.000 description 2
- 229920006395 saturated elastomer Polymers 0.000 description 2
- 238000005204 segregation Methods 0.000 description 2
- 238000000926 separation method Methods 0.000 description 2
- 239000002002 slurry Substances 0.000 description 2
- 239000000344 soap Substances 0.000 description 2
- URGAHOPLAPQHLN-UHFFFAOYSA-N sodium aluminosilicate Chemical compound [Na+].[Al+3].[O-][Si]([O-])=O.[O-][Si]([O-])=O URGAHOPLAPQHLN-UHFFFAOYSA-N 0.000 description 2
- MSFGZHUJTJBYFA-UHFFFAOYSA-M sodium dichloroisocyanurate Chemical compound [Na+].ClN1C(=O)[N-]C(=O)N(Cl)C1=O MSFGZHUJTJBYFA-UHFFFAOYSA-M 0.000 description 2
- 239000001488 sodium phosphate Substances 0.000 description 2
- 229910052911 sodium silicate Inorganic materials 0.000 description 2
- 229910052938 sodium sulfate Inorganic materials 0.000 description 2
- 235000011152 sodium sulphate Nutrition 0.000 description 2
- MZSDGDXXBZSFTG-UHFFFAOYSA-M sodium;benzenesulfonate Chemical compound [Na+].[O-]S(=O)(=O)C1=CC=CC=C1 MZSDGDXXBZSFTG-UHFFFAOYSA-M 0.000 description 2
- 238000010561 standard procedure Methods 0.000 description 2
- 238000003860 storage Methods 0.000 description 2
- 229910052717 sulfur Inorganic materials 0.000 description 2
- 239000001117 sulphuric acid Substances 0.000 description 2
- 235000011149 sulphuric acid Nutrition 0.000 description 2
- 238000003786 synthesis reaction Methods 0.000 description 2
- 239000003760 tallow Substances 0.000 description 2
- KKEYFWRCBNTPAC-UHFFFAOYSA-L terephthalate(2-) Chemical compound [O-]C(=O)C1=CC=C(C([O-])=O)C=C1 KKEYFWRCBNTPAC-UHFFFAOYSA-L 0.000 description 2
- 229920001897 terpolymer Polymers 0.000 description 2
- 238000002411 thermogravimetry Methods 0.000 description 2
- QAEDZJGFFMLHHQ-UHFFFAOYSA-N trifluoroacetic anhydride Chemical compound FC(F)(F)C(=O)OC(=O)C(F)(F)F QAEDZJGFFMLHHQ-UHFFFAOYSA-N 0.000 description 2
- 235000019801 trisodium phosphate Nutrition 0.000 description 2
- 229910000406 trisodium phosphate Inorganic materials 0.000 description 2
- 229910052725 zinc Inorganic materials 0.000 description 2
- 239000011701 zinc Substances 0.000 description 2
- WRIDQFICGBMAFQ-UHFFFAOYSA-N (E)-8-Octadecenoic acid Natural products CCCCCCCCCC=CCCCCCCC(O)=O WRIDQFICGBMAFQ-UHFFFAOYSA-N 0.000 description 1
- PULUUPKYWQEYCF-UHFFFAOYSA-N 1,2,3,4-tetrapropylbenzene Chemical compound CCCC1=CC=C(CCC)C(CCC)=C1CCC PULUUPKYWQEYCF-UHFFFAOYSA-N 0.000 description 1
- YZUPZGFPHUVJKC-UHFFFAOYSA-N 1-bromo-2-methoxyethane Chemical compound COCCBr YZUPZGFPHUVJKC-UHFFFAOYSA-N 0.000 description 1
- CRSBERNSMYQZNG-UHFFFAOYSA-N 1-dodecene Chemical group CCCCCCCCCCC=C CRSBERNSMYQZNG-UHFFFAOYSA-N 0.000 description 1
- VJSWLXWONORKLD-UHFFFAOYSA-N 2,4,6-trihydroxybenzene-1,3,5-trisulfonic acid Chemical compound OC1=C(S(O)(=O)=O)C(O)=C(S(O)(=O)=O)C(O)=C1S(O)(=O)=O VJSWLXWONORKLD-UHFFFAOYSA-N 0.000 description 1
- QCYOIFVBYZNUNW-UHFFFAOYSA-N 2-(dimethylazaniumyl)propanoate Chemical compound CN(C)C(C)C(O)=O QCYOIFVBYZNUNW-UHFFFAOYSA-N 0.000 description 1
- HZAXFHJVJLSVMW-UHFFFAOYSA-N 2-Aminoethan-1-ol Chemical compound NCCO HZAXFHJVJLSVMW-UHFFFAOYSA-N 0.000 description 1
- UXFQFBNBSPQBJW-UHFFFAOYSA-N 2-amino-2-methylpropane-1,3-diol Chemical compound OCC(N)(C)CO UXFQFBNBSPQBJW-UHFFFAOYSA-N 0.000 description 1
- HQBYUMGOEJNFJB-UHFFFAOYSA-N 2-aminoethanol;azane Chemical compound N.NCCO HQBYUMGOEJNFJB-UHFFFAOYSA-N 0.000 description 1
- GLVYLTSKTCWWJR-UHFFFAOYSA-N 2-carbonoperoxoylbenzoic acid Chemical compound OOC(=O)C1=CC=CC=C1C(O)=O GLVYLTSKTCWWJR-UHFFFAOYSA-N 0.000 description 1
- ZPVFWPFBNIEHGJ-UHFFFAOYSA-N 2-octanone Chemical compound CCCCCCC(C)=O ZPVFWPFBNIEHGJ-UHFFFAOYSA-N 0.000 description 1
- LQJBNNIYVWPHFW-UHFFFAOYSA-N 20:1omega9c fatty acid Natural products CCCCCCCCCCC=CCCCCCCCC(O)=O LQJBNNIYVWPHFW-UHFFFAOYSA-N 0.000 description 1
- CJAZCKUGLFWINJ-UHFFFAOYSA-N 3,4-dihydroxybenzene-1,2-disulfonic acid Chemical compound OC1=CC=C(S(O)(=O)=O)C(S(O)(=O)=O)=C1O CJAZCKUGLFWINJ-UHFFFAOYSA-N 0.000 description 1
- CDWQJRGVYJQAIT-UHFFFAOYSA-N 3-benzoylpiperidin-2-one Chemical compound C=1C=CC=CC=1C(=O)C1CCCNC1=O CDWQJRGVYJQAIT-UHFFFAOYSA-N 0.000 description 1
- VFIUQNHHBQTNNM-UHFFFAOYSA-N 3-decanoylazepan-2-one Chemical compound CCCCCCCCCC(=O)C1CCCCNC1=O VFIUQNHHBQTNNM-UHFFFAOYSA-N 0.000 description 1
- OLDXODLIOAKDPY-UHFFFAOYSA-N 3-decanoylpiperidin-2-one Chemical compound CCCCCCCCCC(=O)C1CCCNC1=O OLDXODLIOAKDPY-UHFFFAOYSA-N 0.000 description 1
- QZPSOSOOLFHYRR-UHFFFAOYSA-N 3-hydroxypropyl prop-2-enoate Chemical compound OCCCOC(=O)C=C QZPSOSOOLFHYRR-UHFFFAOYSA-N 0.000 description 1
- CLSGAPMZXLVENM-UHFFFAOYSA-N 3-nonanoylazepan-2-one Chemical compound CCCCCCCCC(=O)C1CCCCNC1=O CLSGAPMZXLVENM-UHFFFAOYSA-N 0.000 description 1
- PUEBLFAPKBDUKE-UHFFFAOYSA-N 3-octanoylazepan-2-one Chemical compound CCCCCCCC(=O)C1CCCCNC1=O PUEBLFAPKBDUKE-UHFFFAOYSA-N 0.000 description 1
- YILDPURCUKWQHU-UHFFFAOYSA-N 3-octanoylpiperidin-2-one Chemical compound CCCCCCCC(=O)C1CCCNC1=O YILDPURCUKWQHU-UHFFFAOYSA-N 0.000 description 1
- OFFQNNRKQRWVLU-UHFFFAOYSA-N 3-undecanoylazepan-2-one Chemical compound CCCCCCCCCCC(=O)C1CCCCNC1=O OFFQNNRKQRWVLU-UHFFFAOYSA-N 0.000 description 1
- QBBADBFKAPIUGH-UHFFFAOYSA-N 3-undecanoylpiperidin-2-one Chemical compound CCCCCCCCCCC(=O)C1CCCNC1=O QBBADBFKAPIUGH-UHFFFAOYSA-N 0.000 description 1
- ILNGCUVXLQVDTC-UHFFFAOYSA-N 4-chloromorpholine Chemical compound ClN1CCOCC1 ILNGCUVXLQVDTC-UHFFFAOYSA-N 0.000 description 1
- LWYAUHJRUCQFCX-UHFFFAOYSA-N 4-dodecoxy-4-oxobutanoic acid Chemical class CCCCCCCCCCCCOC(=O)CCC(O)=O LWYAUHJRUCQFCX-UHFFFAOYSA-N 0.000 description 1
- XGIPGWJHNHEEAL-UHFFFAOYSA-N 4-hexadecoxy-4-oxobutanoic acid Chemical compound CCCCCCCCCCCCCCCCOC(=O)CCC(O)=O XGIPGWJHNHEEAL-UHFFFAOYSA-N 0.000 description 1
- LSWKXNPXIJXDHU-UHFFFAOYSA-N 4-oxo-4-tetradecoxybutanoic acid Chemical compound CCCCCCCCCCCCCCOC(=O)CCC(O)=O LSWKXNPXIJXDHU-UHFFFAOYSA-N 0.000 description 1
- AFHIIJICYLMCSH-VOTSOKGWSA-N 5-amino-2-[(e)-2-(4-benzamido-2-sulfophenyl)ethenyl]benzenesulfonic acid Chemical compound OS(=O)(=O)C1=CC(N)=CC=C1\C=C\C(C(=C1)S(O)(=O)=O)=CC=C1NC(=O)C1=CC=CC=C1 AFHIIJICYLMCSH-VOTSOKGWSA-N 0.000 description 1
- QSBYPNXLFMSGKH-UHFFFAOYSA-N 9-Heptadecensaeure Natural products CCCCCCCC=CCCCCCCCC(O)=O QSBYPNXLFMSGKH-UHFFFAOYSA-N 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical group CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 1
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 1
- BTBUEUYNUDRHOZ-UHFFFAOYSA-N Borate Chemical compound [O-]B([O-])[O-] BTBUEUYNUDRHOZ-UHFFFAOYSA-N 0.000 description 1
- ONYKNTRMIOQQBJ-UHFFFAOYSA-N C(CCC(=O)O)(=O)O.C(CCC(=O)O)(=O)O.[O] Chemical compound C(CCC(=O)O)(=O)O.C(CCC(=O)O)(=O)O.[O] ONYKNTRMIOQQBJ-UHFFFAOYSA-N 0.000 description 1
- XSNXNRNMDLLTRG-UHFFFAOYSA-M C1(=CC=CC=C1)S(=O)(=O)[O-].C(CCCCCCCC)(=O)[O+] Chemical compound C1(=CC=CC=C1)S(=O)(=O)[O-].C(CCCCCCCC)(=O)[O+] XSNXNRNMDLLTRG-UHFFFAOYSA-M 0.000 description 1
- 101100148128 Caenorhabditis elegans rsp-4 gene Proteins 0.000 description 1
- 101100201838 Caenorhabditis elegans rsp-6 gene Proteins 0.000 description 1
- UXVMQQNJUSDDNG-UHFFFAOYSA-L Calcium chloride Chemical compound [Cl-].[Cl-].[Ca+2] UXVMQQNJUSDDNG-UHFFFAOYSA-L 0.000 description 1
- 239000005632 Capric acid (CAS 334-48-5) Substances 0.000 description 1
- RKWGIWYCVPQPMF-UHFFFAOYSA-N Chloropropamide Chemical compound CCCNC(=O)NS(=O)(=O)C1=CC=C(Cl)C=C1 RKWGIWYCVPQPMF-UHFFFAOYSA-N 0.000 description 1
- 241000640882 Condea Species 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- 239000004375 Dextrin Substances 0.000 description 1
- 229920001353 Dextrin Polymers 0.000 description 1
- RPNUMPOLZDHAAY-UHFFFAOYSA-N Diethylenetriamine Chemical compound NCCNCCN RPNUMPOLZDHAAY-UHFFFAOYSA-N 0.000 description 1
- SNRUBQQJIBEYMU-UHFFFAOYSA-N Dodecane Natural products CCCCCCCCCCCC SNRUBQQJIBEYMU-UHFFFAOYSA-N 0.000 description 1
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 1
- 240000004859 Gamochaeta purpurea Species 0.000 description 1
- 101000643890 Homo sapiens Ubiquitin carboxyl-terminal hydrolase 5 Proteins 0.000 description 1
- OWYWGLHRNBIFJP-UHFFFAOYSA-N Ipazine Chemical compound CCN(CC)C1=NC(Cl)=NC(NC(C)C)=N1 OWYWGLHRNBIFJP-UHFFFAOYSA-N 0.000 description 1
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 1
- 239000005639 Lauric acid Substances 0.000 description 1
- OYHQOLUKZRVURQ-HZJYTTRNSA-N Linoleic acid Chemical compound CCCCC\C=C/C\C=C/CCCCCCCC(O)=O OYHQOLUKZRVURQ-HZJYTTRNSA-N 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- 244000261422 Lysimachia clethroides Species 0.000 description 1
- TUNFSRHWOTWDNC-UHFFFAOYSA-N Myristic acid Natural products CCCCCCCCCCCCCC(O)=O TUNFSRHWOTWDNC-UHFFFAOYSA-N 0.000 description 1
- IZWSFJTYBVKZNK-UHFFFAOYSA-O N-dodecyl-N,N-dimethyl-3-ammonio-1-propanesulfonic acid Chemical compound CCCCCCCCCCCC[N+](C)(C)CCCS(O)(=O)=O IZWSFJTYBVKZNK-UHFFFAOYSA-O 0.000 description 1
- MBBZMMPHUWSWHV-BDVNFPICSA-N N-methylglucamine Chemical compound CNC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO MBBZMMPHUWSWHV-BDVNFPICSA-N 0.000 description 1
- 229910000503 Na-aluminosilicate Inorganic materials 0.000 description 1
- 239000006057 Non-nutritive feed additive Substances 0.000 description 1
- 239000004677 Nylon Substances 0.000 description 1
- BFMRUPBDRCFGEB-UHFFFAOYSA-N O.O.[Na].[Na].[Na] Chemical compound O.O.[Na].[Na].[Na] BFMRUPBDRCFGEB-UHFFFAOYSA-N 0.000 description 1
- REYJJPSVUYRZGE-UHFFFAOYSA-N Octadecylamine Chemical compound CCCCCCCCCCCCCCCCCCN REYJJPSVUYRZGE-UHFFFAOYSA-N 0.000 description 1
- 239000005642 Oleic acid Substances 0.000 description 1
- ZQPPMHVWECSIRJ-UHFFFAOYSA-N Oleic acid Natural products CCCCCCCCC=CCCCCCCCC(O)=O ZQPPMHVWECSIRJ-UHFFFAOYSA-N 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 102000003992 Peroxidases Human genes 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 108010020346 Polyglutamic Acid Proteins 0.000 description 1
- 239000004642 Polyimide Substances 0.000 description 1
- WCUXLLCKKVVCTQ-UHFFFAOYSA-M Potassium chloride Chemical compound [Cl-].[K+] WCUXLLCKKVVCTQ-UHFFFAOYSA-M 0.000 description 1
- 239000004111 Potassium silicate Substances 0.000 description 1
- NBBJYMSMWIIQGU-UHFFFAOYSA-N Propionic aldehyde Chemical compound CCC=O NBBJYMSMWIIQGU-UHFFFAOYSA-N 0.000 description 1
- 101710194948 Protein phosphatase PhpP Proteins 0.000 description 1
- RWRDLPDLKQPQOW-UHFFFAOYSA-N Pyrrolidine Chemical compound C1CCNC1 RWRDLPDLKQPQOW-UHFFFAOYSA-N 0.000 description 1
- 235000019484 Rapeseed oil Nutrition 0.000 description 1
- 229910004283 SiO 4 Inorganic materials 0.000 description 1
- FKNQFGJONOIPTF-UHFFFAOYSA-N Sodium cation Chemical compound [Na+] FKNQFGJONOIPTF-UHFFFAOYSA-N 0.000 description 1
- 235000021355 Stearic acid Nutrition 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 1
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 1
- 102100021017 Ubiquitin carboxyl-terminal hydrolase 5 Human genes 0.000 description 1
- 238000002441 X-ray diffraction Methods 0.000 description 1
- 238000010306 acid treatment Methods 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 229920006243 acrylic copolymer Polymers 0.000 description 1
- 230000004913 activation Effects 0.000 description 1
- 125000004423 acyloxy group Chemical group 0.000 description 1
- 150000001334 alicyclic compounds Chemical class 0.000 description 1
- 125000002723 alicyclic group Chemical group 0.000 description 1
- 229910001413 alkali metal ion Inorganic materials 0.000 description 1
- 229910000318 alkali metal phosphate Inorganic materials 0.000 description 1
- 229910052910 alkali metal silicate Inorganic materials 0.000 description 1
- 150000008055 alkyl aryl sulfonates Chemical class 0.000 description 1
- 125000005037 alkyl phenyl group Chemical group 0.000 description 1
- 125000005466 alkylenyl group Chemical group 0.000 description 1
- 125000001118 alkylidene group Chemical group 0.000 description 1
- DTOSIQBPPRVQHS-PDBXOOCHSA-N alpha-linolenic acid Chemical compound CC\C=C/C\C=C/C\C=C/CCCCCCCC(O)=O DTOSIQBPPRVQHS-PDBXOOCHSA-N 0.000 description 1
- 235000020661 alpha-linolenic acid Nutrition 0.000 description 1
- 239000005030 aluminium foil Substances 0.000 description 1
- HPTYUNKZVDYXLP-UHFFFAOYSA-N aluminum;trihydroxy(trihydroxysilyloxy)silane;hydrate Chemical compound O.[Al].[Al].O[Si](O)(O)O[Si](O)(O)O HPTYUNKZVDYXLP-UHFFFAOYSA-N 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 235000019270 ammonium chloride Nutrition 0.000 description 1
- 239000000908 ammonium hydroxide Substances 0.000 description 1
- 230000003625 amylolytic effect Effects 0.000 description 1
- 229960004543 anhydrous citric acid Drugs 0.000 description 1
- 238000000149 argon plasma sintering Methods 0.000 description 1
- 150000001491 aromatic compounds Chemical class 0.000 description 1
- WXQWKYFPCLREEY-UHFFFAOYSA-N azane;ethanol Chemical class N.CCO.CCO.CCO WXQWKYFPCLREEY-UHFFFAOYSA-N 0.000 description 1
- 230000004888 barrier function Effects 0.000 description 1
- 235000015278 beef Nutrition 0.000 description 1
- SVPXDRXYRYOSEX-UHFFFAOYSA-N bentoquatam Chemical compound O.O=[Si]=O.O=[Al]O[Al]=O SVPXDRXYRYOSEX-UHFFFAOYSA-N 0.000 description 1
- LTPBRCUWZOMYOC-UHFFFAOYSA-N beryllium oxide Inorganic materials O=[Be] LTPBRCUWZOMYOC-UHFFFAOYSA-N 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- 238000009835 boiling Methods 0.000 description 1
- 230000031709 bromination Effects 0.000 description 1
- 238000005893 bromination reaction Methods 0.000 description 1
- 239000006227 byproduct Substances 0.000 description 1
- 239000001110 calcium chloride Substances 0.000 description 1
- 229910001628 calcium chloride Inorganic materials 0.000 description 1
- 239000004202 carbamide Substances 0.000 description 1
- 239000001569 carbon dioxide Substances 0.000 description 1
- 229910002092 carbon dioxide Inorganic materials 0.000 description 1
- 238000003763 carbonization Methods 0.000 description 1
- 239000012159 carrier gas Substances 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 230000003197 catalytic effect Effects 0.000 description 1
- 238000009614 chemical analysis method Methods 0.000 description 1
- 238000007385 chemical modification Methods 0.000 description 1
- 239000000460 chlorine Substances 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 239000002131 composite material Substances 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 238000013016 damping Methods 0.000 description 1
- 238000013523 data management Methods 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 230000018044 dehydration Effects 0.000 description 1
- 238000006297 dehydration reaction Methods 0.000 description 1
- 238000003795 desorption Methods 0.000 description 1
- 235000019425 dextrin Nutrition 0.000 description 1
- 229910001649 dickite Inorganic materials 0.000 description 1
- GSPKZYJPUDYKPI-UHFFFAOYSA-N diethoxy sulfate Chemical compound CCOOS(=O)(=O)OOCC GSPKZYJPUDYKPI-UHFFFAOYSA-N 0.000 description 1
- GPLRAVKSCUXZTP-UHFFFAOYSA-N diglycerol Chemical compound OCC(O)COCC(O)CO GPLRAVKSCUXZTP-UHFFFAOYSA-N 0.000 description 1
- 125000000118 dimethyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 239000004205 dimethyl polysiloxane Substances 0.000 description 1
- ZKRXZOLGLXXMEA-UHFFFAOYSA-N dioxosilane zirconium Chemical compound [Zr].[Si](=O)=O ZKRXZOLGLXXMEA-UHFFFAOYSA-N 0.000 description 1
- VZCXJNJJHBTBIM-UHFFFAOYSA-L disodium 2-[(4-anilino-6-morpholin-4-yl-1,3,5-triazin-2-yl)amino]-6-[2-(2-sulfonatophenyl)ethenyl]benzenesulfonate Chemical compound [Na+].[Na+].N(C1=CC=CC=C1)C1=NC(=NC(=N1)N1CCOCC1)NC1=C(C(=CC=C1)C=CC=1C(=CC=CC=1)S(=O)(=O)[O-])S(=O)(=O)[O-] VZCXJNJJHBTBIM-UHFFFAOYSA-L 0.000 description 1
- CDMADVZSLOHIFP-UHFFFAOYSA-N disodium;3,7-dioxido-2,4,6,8,9-pentaoxa-1,3,5,7-tetraborabicyclo[3.3.1]nonane;decahydrate Chemical compound O.O.O.O.O.O.O.O.O.O.[Na+].[Na+].O1B([O-])OB2OB([O-])OB1O2 CDMADVZSLOHIFP-UHFFFAOYSA-N 0.000 description 1
- 238000006073 displacement reaction Methods 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 125000005066 dodecenyl group Chemical group C(=CCCCCCCCCCC)* 0.000 description 1
- SYELZBGXAIXKHU-UHFFFAOYSA-N dodecyldimethylamine N-oxide Chemical compound CCCCCCCCCCCC[N+](C)(C)[O-] SYELZBGXAIXKHU-UHFFFAOYSA-N 0.000 description 1
- 229960001484 edetic acid Drugs 0.000 description 1
- 239000007938 effervescent tablet Substances 0.000 description 1
- 230000005611 electricity Effects 0.000 description 1
- 238000005538 encapsulation Methods 0.000 description 1
- 235000021321 essential mineral Nutrition 0.000 description 1
- 125000004185 ester group Chemical group 0.000 description 1
- OCLXJTCGWSSVOE-UHFFFAOYSA-N ethanol etoh Chemical compound CCO.CCO OCLXJTCGWSSVOE-UHFFFAOYSA-N 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000000284 extract Substances 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 150000002193 fatty amides Chemical class 0.000 description 1
- 238000011049 filling Methods 0.000 description 1
- 239000012467 final product Substances 0.000 description 1
- 239000010419 fine particle Substances 0.000 description 1
- 238000004401 flow injection analysis Methods 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 239000003205 fragrance Substances 0.000 description 1
- JPZROSNLRWHSQQ-UHFFFAOYSA-N furan-2,5-dione;prop-2-enoic acid Chemical compound OC(=O)C=C.O=C1OC(=O)C=C1 JPZROSNLRWHSQQ-UHFFFAOYSA-N 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- HNZUNIKWNYHEJJ-FMIVXFBMSA-N geranyl acetone Chemical compound CC(C)=CCC\C(C)=C\CCC(C)=O HNZUNIKWNYHEJJ-FMIVXFBMSA-N 0.000 description 1
- HNZUNIKWNYHEJJ-UHFFFAOYSA-N geranyl acetone Natural products CC(C)=CCCC(C)=CCCC(C)=O HNZUNIKWNYHEJJ-UHFFFAOYSA-N 0.000 description 1
- 229930182470 glycoside Natural products 0.000 description 1
- 230000005484 gravity Effects 0.000 description 1
- 229910052621 halloysite Inorganic materials 0.000 description 1
- 239000001307 helium Substances 0.000 description 1
- 229910052734 helium Inorganic materials 0.000 description 1
- SWQJXJOGLNCZEY-UHFFFAOYSA-N helium atom Chemical compound [He] SWQJXJOGLNCZEY-UHFFFAOYSA-N 0.000 description 1
- 125000000623 heterocyclic group Chemical group 0.000 description 1
- IPCSVZSSVZVIGE-UHFFFAOYSA-N hexadecanoic acid Chemical compound CCCCCCCCCCCCCCCC(O)=O IPCSVZSSVZVIGE-UHFFFAOYSA-N 0.000 description 1
- JISVIRFOSOKJIU-UHFFFAOYSA-N hexylidene Chemical group [CH2+]CCCC[CH-] JISVIRFOSOKJIU-UHFFFAOYSA-N 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- OUUQCZGPVNCOIJ-UHFFFAOYSA-N hydroperoxyl Chemical compound O[O] OUUQCZGPVNCOIJ-UHFFFAOYSA-N 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 150000002460 imidazoles Chemical class 0.000 description 1
- 230000002779 inactivation Effects 0.000 description 1
- 150000002484 inorganic compounds Chemical class 0.000 description 1
- 239000011147 inorganic material Substances 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- LVPMIMZXDYBCDF-UHFFFAOYSA-N isocinchomeronic acid Chemical compound OC(=O)C1=CC=C(C(O)=O)N=C1 LVPMIMZXDYBCDF-UHFFFAOYSA-N 0.000 description 1
- XMVBHZBLHNOQON-UHFFFAOYSA-N isolauryl alcohol Natural products CCCCCCC(CO)CCCC XMVBHZBLHNOQON-UHFFFAOYSA-N 0.000 description 1
- QXJSBBXBKPUZAA-UHFFFAOYSA-N isooleic acid Natural products CCCCCCCC=CCCCCCCCCC(O)=O QXJSBBXBKPUZAA-UHFFFAOYSA-N 0.000 description 1
- 150000002576 ketones Chemical class 0.000 description 1
- 238000004900 laundering Methods 0.000 description 1
- 229960004232 linoleic acid Drugs 0.000 description 1
- 229960004488 linolenic acid Drugs 0.000 description 1
- KQQKGWQCNNTQJW-UHFFFAOYSA-N linolenic acid Natural products CC=CCCC=CCC=CCCCCCCCC(O)=O KQQKGWQCNNTQJW-UHFFFAOYSA-N 0.000 description 1
- 230000002366 lipolytic effect Effects 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- 108010056929 lyticase Proteins 0.000 description 1
- 229910001629 magnesium chloride Inorganic materials 0.000 description 1
- VTHJTEIRLNZDEV-UHFFFAOYSA-L magnesium dihydroxide Chemical compound [OH-].[OH-].[Mg+2] VTHJTEIRLNZDEV-UHFFFAOYSA-L 0.000 description 1
- 239000000347 magnesium hydroxide Substances 0.000 description 1
- 229910001862 magnesium hydroxide Inorganic materials 0.000 description 1
- HCWCAKKEBCNQJP-UHFFFAOYSA-N magnesium orthosilicate Chemical compound [Mg+2].[Mg+2].[O-][Si]([O-])([O-])[O-] HCWCAKKEBCNQJP-UHFFFAOYSA-N 0.000 description 1
- 239000000391 magnesium silicate Substances 0.000 description 1
- 235000019792 magnesium silicate Nutrition 0.000 description 1
- 229910052919 magnesium silicate Inorganic materials 0.000 description 1
- NXPHGHWWQRMDIA-UHFFFAOYSA-M magnesium;carbanide;bromide Chemical compound [CH3-].[Mg+2].[Br-] NXPHGHWWQRMDIA-UHFFFAOYSA-M 0.000 description 1
- 238000010907 mechanical stirring Methods 0.000 description 1
- LULAYUGMBFYYEX-UHFFFAOYSA-N metachloroperbenzoic acid Natural products OC(=O)C1=CC=CC(Cl)=C1 LULAYUGMBFYYEX-UHFFFAOYSA-N 0.000 description 1
- 229910021645 metal ion Inorganic materials 0.000 description 1
- 108010020132 microbial serine proteinases Proteins 0.000 description 1
- 239000003094 microcapsule Substances 0.000 description 1
- 235000010755 mineral Nutrition 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 239000003607 modifier Substances 0.000 description 1
- 239000002808 molecular sieve Substances 0.000 description 1
- 238000012544 monitoring process Methods 0.000 description 1
- CQDGTJPVBWZJAZ-UHFFFAOYSA-N monoethyl carbonate Chemical compound CCOC(O)=O CQDGTJPVBWZJAZ-UHFFFAOYSA-N 0.000 description 1
- 125000004573 morpholin-4-yl group Chemical group N1(CCOCC1)* 0.000 description 1
- IBOBFGGLRNWLIL-UHFFFAOYSA-N n,n-dimethylhexadecan-1-amine oxide Chemical compound CCCCCCCCCCCCCCCC[N+](C)(C)[O-] IBOBFGGLRNWLIL-UHFFFAOYSA-N 0.000 description 1
- AFFLGGQVNFXPEV-UHFFFAOYSA-N n-decene Natural products CCCCCCCCC=C AFFLGGQVNFXPEV-UHFFFAOYSA-N 0.000 description 1
- ONLRKTIYOMZEJM-UHFFFAOYSA-N n-methylmethanamine oxide Chemical compound C[NH+](C)[O-] ONLRKTIYOMZEJM-UHFFFAOYSA-N 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 239000005445 natural material Substances 0.000 description 1
- 238000000655 nuclear magnetic resonance spectrum Methods 0.000 description 1
- 230000000269 nucleophilic effect Effects 0.000 description 1
- 229920001778 nylon Polymers 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 1
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 1
- GTYIGAUPHJCMLF-UHFFFAOYSA-N octan-2-ylbenzene Chemical compound CCCCCCC(C)C1=CC=CC=C1 GTYIGAUPHJCMLF-UHFFFAOYSA-N 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 235000019198 oils Nutrition 0.000 description 1
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 description 1
- 229960002969 oleic acid Drugs 0.000 description 1
- 125000001117 oleyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])/C([H])=C([H])\C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 150000002894 organic compounds Chemical class 0.000 description 1
- VGTPKLINSHNZRD-UHFFFAOYSA-N oxoborinic acid Chemical compound OB=O VGTPKLINSHNZRD-UHFFFAOYSA-N 0.000 description 1
- 125000006353 oxyethylene group Chemical group 0.000 description 1
- 230000020477 pH reduction Effects 0.000 description 1
- SECPZKHBENQXJG-FPLPWBNLSA-N palmitoleic acid Chemical compound CCCCCC\C=C/CCCCCCCC(O)=O SECPZKHBENQXJG-FPLPWBNLSA-N 0.000 description 1
- 239000002907 paramagnetic material Substances 0.000 description 1
- 238000005192 partition Methods 0.000 description 1
- GNTXATNEGVVGQJ-UHFFFAOYSA-N pentadecan-2-ylbenzene Chemical compound CCCCCCCCCCCCCC(C)C1=CC=CC=C1 GNTXATNEGVVGQJ-UHFFFAOYSA-N 0.000 description 1
- HWGNBUXHKFFFIH-UHFFFAOYSA-I pentasodium;[oxido(phosphonatooxy)phosphoryl] phosphate Chemical compound [Na+].[Na+].[Na+].[Na+].[Na+].[O-]P([O-])(=O)OP([O-])(=O)OP([O-])([O-])=O HWGNBUXHKFFFIH-UHFFFAOYSA-I 0.000 description 1
- 238000001935 peptisation Methods 0.000 description 1
- 230000010412 perfusion Effects 0.000 description 1
- 108040007629 peroxidase activity proteins Proteins 0.000 description 1
- 238000005502 peroxidation Methods 0.000 description 1
- 150000002978 peroxides Chemical class 0.000 description 1
- 150000004965 peroxy acids Chemical class 0.000 description 1
- JRKICGRDRMAZLK-UHFFFAOYSA-L persulfate group Chemical group S(=O)(=O)([O-])OOS(=O)(=O)[O-] JRKICGRDRMAZLK-UHFFFAOYSA-L 0.000 description 1
- 229940044652 phenolsulfonate Drugs 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 235000021317 phosphate Nutrition 0.000 description 1
- 230000002186 photoactivation Effects 0.000 description 1
- 150000003053 piperidines Chemical class 0.000 description 1
- 229920000435 poly(dimethylsiloxane) Polymers 0.000 description 1
- 239000004584 polyacrylic acid Substances 0.000 description 1
- 229920001281 polyalkylene Polymers 0.000 description 1
- 229920006122 polyamide resin Polymers 0.000 description 1
- 229920000570 polyether Polymers 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 229920002643 polyglutamic acid Polymers 0.000 description 1
- 229920001721 polyimide Polymers 0.000 description 1
- 229920001444 polymaleic acid Polymers 0.000 description 1
- 239000002861 polymer material Substances 0.000 description 1
- 229920001282 polysaccharide Polymers 0.000 description 1
- 239000005017 polysaccharide Substances 0.000 description 1
- 229920005996 polystyrene-poly(ethylene-butylene)-polystyrene Polymers 0.000 description 1
- 229920001343 polytetrafluoroethylene Polymers 0.000 description 1
- 239000004810 polytetrafluoroethylene Substances 0.000 description 1
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Substances [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 1
- 235000015320 potassium carbonate Nutrition 0.000 description 1
- 239000001103 potassium chloride Substances 0.000 description 1
- 235000011164 potassium chloride Nutrition 0.000 description 1
- NNHHDJVEYQHLHG-UHFFFAOYSA-N potassium silicate Chemical compound [K+].[K+].[O-][Si]([O-])=O NNHHDJVEYQHLHG-UHFFFAOYSA-N 0.000 description 1
- 229910052913 potassium silicate Inorganic materials 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 108010043393 protease N Proteins 0.000 description 1
- 230000002797 proteolythic effect Effects 0.000 description 1
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Chemical group COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 1
- GJAWHXHKYYXBSV-UHFFFAOYSA-N pyridinedicarboxylic acid Natural products OC(=O)C1=CC=CN=C1C(O)=O GJAWHXHKYYXBSV-UHFFFAOYSA-N 0.000 description 1
- 150000003233 pyrroles Chemical class 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 229920005989 resin Polymers 0.000 description 1
- 239000011347 resin Substances 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- WBHHMMIMDMUBKC-QJWNTBNXSA-N ricinoleic acid Chemical compound CCCCCC[C@@H](O)C\C=C/CCCCCCCC(O)=O WBHHMMIMDMUBKC-QJWNTBNXSA-N 0.000 description 1
- 229960003656 ricinoleic acid Drugs 0.000 description 1
- FEUQNCSVHBHROZ-UHFFFAOYSA-N ricinoleic acid Natural products CCCCCCC(O[Si](C)(C)C)CC=CCCCCCCCC(=O)OC FEUQNCSVHBHROZ-UHFFFAOYSA-N 0.000 description 1
- 239000012266 salt solution Substances 0.000 description 1
- 150000003333 secondary alcohols Chemical class 0.000 description 1
- 230000011218 segmentation Effects 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 150000004760 silicates Chemical class 0.000 description 1
- 229920002545 silicone oil Polymers 0.000 description 1
- 239000010454 slate Substances 0.000 description 1
- 239000000429 sodium aluminium silicate Substances 0.000 description 1
- 235000012217 sodium aluminium silicate Nutrition 0.000 description 1
- 235000017557 sodium bicarbonate Nutrition 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- 235000017550 sodium carbonate Nutrition 0.000 description 1
- 229910000029 sodium carbonate Inorganic materials 0.000 description 1
- HLBBKKJFGFRGMU-UHFFFAOYSA-M sodium formate Chemical compound [Na+].[O-]C=O HLBBKKJFGFRGMU-UHFFFAOYSA-M 0.000 description 1
- 229910001415 sodium ion Inorganic materials 0.000 description 1
- 229960001922 sodium perborate Drugs 0.000 description 1
- PFUVRDFDKPNGAV-UHFFFAOYSA-N sodium peroxide Chemical compound [Na+].[Na+].[O-][O-] PFUVRDFDKPNGAV-UHFFFAOYSA-N 0.000 description 1
- 229940048842 sodium xylenesulfonate Drugs 0.000 description 1
- KVCGISUBCHHTDD-UHFFFAOYSA-M sodium;4-methylbenzenesulfonate Chemical compound [Na+].CC1=CC=C(S([O-])(=O)=O)C=C1 KVCGISUBCHHTDD-UHFFFAOYSA-M 0.000 description 1
- WGRULTCAYDOGQK-UHFFFAOYSA-M sodium;sodium;hydroxide Chemical compound [OH-].[Na].[Na+] WGRULTCAYDOGQK-UHFFFAOYSA-M 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 239000008117 stearic acid Substances 0.000 description 1
- 125000004079 stearyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000001384 succinic acid Substances 0.000 description 1
- 150000003900 succinic acid esters Chemical class 0.000 description 1
- 150000005846 sugar alcohols Polymers 0.000 description 1
- 230000001180 sulfating effect Effects 0.000 description 1
- 230000019635 sulfation Effects 0.000 description 1
- 238000005670 sulfation reaction Methods 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 238000010189 synthetic method Methods 0.000 description 1
- 235000012976 tarts Nutrition 0.000 description 1
- 108010075550 termamyl Proteins 0.000 description 1
- 238000010998 test method Methods 0.000 description 1
- BFKJFAAPBSQJPD-UHFFFAOYSA-N tetrafluoroethene Chemical group FC(F)=C(F)F BFKJFAAPBSQJPD-UHFFFAOYSA-N 0.000 description 1
- 150000004685 tetrahydrates Chemical class 0.000 description 1
- 239000004753 textile Substances 0.000 description 1
- 238000007669 thermal treatment Methods 0.000 description 1
- 229910003452 thorium oxide Inorganic materials 0.000 description 1
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N titanium dioxide Inorganic materials O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 1
- 238000004448 titration Methods 0.000 description 1
- 150000003613 toluenes Chemical class 0.000 description 1
- 229910052723 transition metal Inorganic materials 0.000 description 1
- 150000003624 transition metals Chemical class 0.000 description 1
- UNXRWKVEANCORM-UHFFFAOYSA-I triphosphate(5-) Chemical compound [O-]P([O-])(=O)OP([O-])(=O)OP([O-])([O-])=O UNXRWKVEANCORM-UHFFFAOYSA-I 0.000 description 1
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical compound OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 1
- 229920002554 vinyl polymer Polymers 0.000 description 1
- 230000004304 visual acuity Effects 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
- 239000002759 woven fabric Substances 0.000 description 1
Landscapes
- Detergent Compositions (AREA)
Abstract
本发明涉及包括改性烷基苯磺酸盐表面活性剂混合物的硬表面清洗组合物。The present invention relates to hard surface cleaning compositions comprising a mixture of modified alkylbenzene sulfonate surfactants.
Description
Invention field
The present invention relates to clothing heavy duty liquid detergent product, this product in fact is aqueous, the improvement alkylbenzene sulfonate surfactant mixture that wherein contains particular type, in order to control combination thing parameter, 2/3-phenyl exponential sum 2-methyl-2-phenyl index particularly.
Background of invention
Use the alkyl benzene sulfonate surfactant of high side chain in the past washing composition, abbreviate the benzene sulfonate based on the tetrapropylene base of " ABS " or " TPBS " as those.Yet these tensio-active agents are difficult to biological degradation, thereby the alkyl benzene sulphonate (ABS) technology of threonates has been carried out secular improvement, so that production of linear alkylbenzene sulfonate (being abbreviated as " LAS ") as far as possible.The production technology of most alkyl benzene sulfonate surfactants improvement at present is for this purpose, and the relevant extensive alkylbenzene sulfonate production method during all use also just develops towards the production of linear alkylbenzene sulfonate.But linear alkyl benzene sulfonate neither for example, if improve its detergency ability in hard water or cold water, can be brought into play effect without limits better.If use its clean effect often bad in hard water areas.
Because the limitation of alkylbenzene sulfonate, institute is so that the washing prescription needs more high-load cosurfactant, washing assistant and other auxiliary agents when using super alkylbenzene sulfonate.
The reference of relevant Alkyl benzene sulfonate detergent technology is a lot, but support is arranged and oppose two kinds of viewpoints with regard to the everyway of these compositions.And, there is the people at present the explanation of the mechanism of action of LAS under working conditions to be incorrect or to misread to some extent, particularly aspect water hardness tolerable limit.These documents are belittled relevant technologies, make that people are difficult to obtain useful information from these documents that does not have repeated experiments to confirm.In order to understand relevant technologies better, should be realized that the relevant LAS technical problem that present people do not understand fully, and aspect the biological degradability of LAS and the mechanism of action of LAS in hard water, all have misunderstanding.
In the market, being the linear alkyl benzene sulfonate tensio-active agent basically is relative simple composition for analyzing with the explanation speech, just very complicated when containing two kinds of alkyl benzene sulfonate surfactants of side chain and straight chain in the composition simultaneously.Usually such composition varies, and can contain one or more different side chains in the composition, and any position that branch point may be on aliphatic chain.May contain many kinds of chemical substances in this mixture, nearly hundreds of unexpectedly sometimes.The clean effect that improves these detergent composition when therefore will keep biological degradability is a heavy task experimentally, and it is very important that formulating of recipe person's knowledge and experience here just seems.
Another problem that does not also solve in alkylbenzene sulfonate production is how to improve the efficient that LAS feeds intake at present.Utilize various branched hydrocarbyl mixtures not only meaningful better, and also be very useful economically product performance.
Present alkyl benzene sulfonate surfactant composition is not also really satisfied the demand, and requires further improvement, and especially need have characteristics such as ultra-clean, wide water hardness permission, favorable biological degradability and nominal price.
Background technology US 5,659, and 099; US 5,393, and 718; US 5,256, and 392; US 5,227, and 558; US 5,139, and 759; US 5,164, and 169; US 5,116, and 794; US 4,840, and 929; US 5,744, and 673; US 5,522, and 984; US 5,811, and 623; US 5,777, and 187; WO 9,729, and 064; WO 9,747, and 573; WO9,729,063; US 5,026, and 933; US 4,990, and 718; US 4,301, and 316; US 4,301, and 317; US 4,855, and 527; US 4,870, and 038; US 2,477, and 382; EP 466,558, and 1/15/92; EP469,940,2/5/92; FR 2,697, and 246,4/29/94; SU 793,972, and 1/7/81; US2,564,072; US 3,196, and 174; US 3,238, and 249; US 3,355, and 484; US 3,442, and 964; US 3,492, and 364; US 4,959, and 491; WO 88/07030,9/25/90; US 4,962, and 256; US5,196,624; US 5,196, and 625; EP 364,012B, 2/15/90; US 3,312, and 745; US3,341,614; US 3,442, and 965; US 3,674, and 885; US 4,447, and 664; US 4,533, and 651; US4,587,374; US 4,996, and 386; US 5,210, and 060; US 5,510, and 306; WO 95/17961,7/6/95; WO 95/18084; US 5,510, and 306; US 5,087, and 788; US 4,301, and 316; US4,301,317; US 4,855, and 527; US 4,870, and 038; US 5,026, and 933; US 5,625, and 105 and US 4,973,788.Consult " tensio-active agent science " book series (Marcel Dekker press, New York, 1996) the 56th volume chapter 2 " alkylbenzene sulfonate: history, production, analysis and environmental performance ", p39-108; " tensio-active agent science " book series (Marcel Dekker press, New York, 1998) the 73rd volume; " tensio-active agent science " book series (Marcel Dekker press, New York, 1992) the 40th volume.Also referring to U.S. Patent application 60/053,319, patent agent's tag number 6766P, on July 21st, 1997 submitted to; 60/053,318, patent agent's tag number 6767P, on July 21st, 1997 submitted to; 60/053,321, patent agency testimony of a witness 6768P, on July 21st, 1997 submitted to; 60/053,209, patent agency testimony of a witness 6769P, on July 21st, 1997 submitted to; 60/053,328, patent agency testimony of a witness 6770P, on July 21st, 1997 submitted to; 60/053,186, patent agency testimony of a witness 6771P, on July 21st, 1997.Above-mentioned document integral body is as reference of the present invention.
Summary of the invention
The invention provides a kind of water base heavy duty liquid detergent compositions that contains the alkylbenzenesulfonatsurfactants surfactants mixture.
Particularly the present invention contains a kind of water base heavy duty liquid detergent compositions.
Preferably contain surfactant system in the clothing water basic weight duty liquid detergent compositions of the present invention, the tensio-active agent that this system comprises is selected from non-ionic detergent tensio-active agent, anionic detergent tensio-active agent, zwitterionic detergent tensio-active agent, amine oxide type detergent use tensio-active agent and composition thereof.
Especially, first embodiment of the present invention comprises water base heavy duty type laundry detergent composition, comprising: (i) weight is the alkylbenzenesulfonatsurfactants surfactants mixture of whole composition about 5% to about 70%, comprising:
(a) weight is surfactant mixture about 60% to about 95%, preferred about 65% to about 90%, more preferably from about the mixture of 70% to about 85% the branch-alkylbenzene sulfonate with following general molecular formula (I):
The non-annularity aliphatic moiety be made up of carbon and hydrogen of L wherein, this fragment has two methyl ends, except A, R
1And R
2Outward without any other substituting group; Contain two or more in the described branch-alkylbenzene sulfonate, preferably at least three kinds, the optional different described branch-alkylbenzene sulfonate of multiple anionicsite (seeing formula (I)) molecular weight, in the wherein said branch-alkylbenzene sulfonate mixture:
-R
1, L and R
2The carbon atom summation be 9 to 15, preferred 10 to 14.Comprise R
1, L and R
2But the average aliphatic carbon content that does not comprise A is about 10.0 to about 14.0, and preferred about 11.0 to about 13.0, especially preferred about 11.5 to about 12.5 carbon atoms; M is positively charged ion or cation mixt, and preferred M is selected from H, Na, K, Ca, Mg or this several ion mixtures, and more preferably M is selected from H, Na, K and composition thereof, and the valency that especially preferred M is selected from H, Na and composition thereof M is q, is generally 1 to 2, is preferably 1; A and b can keep the electroneutral integer of described branch-alkylbenzene sulfonate (a is generally 1 or 2, is preferably 1, and b is 1); R
1Be the alkyl of 1 to 3 carbon atom, the alkyl of preferred 1 to 2 carbon atom, special preferable methyl; R
2Be alkyl and H (alkyl or the H of preferred 1 to 2 carbon atom that is selected from 1 to 3 carbon atom, more preferably methyl or H, more preferably the supplementary condition of methyl or H are at least about 0.50, preferably surpass 0.70, more preferably in the described branch-alkylbenzene sulfonate of 0.9 to 1.0 molar fraction, R
2Be H); A is that (general A is-C phenyl moiety
6H
4, with SO
3Group shown in general molecular formula (I), only is no more than 5%, the SO of preferred 0 to 5% (weight) usually in the contraposition of L
3Ortho position at L); With
(b) weight be surfactant mixture about 5% to about 40%, preferred about 10% to about 35%, more preferably from about the mixture of 15% to about 30% the non-branched-chain alkyl benzene sulfonate with following general molecular formula (II):
Wherein identical in the definition of a, b, M, A and q and the general formula (I), Y is the linear aliphatic family fragment of the non-replacement be made up of carbon and hydrogen, and this fragment has two methyl ends, and the carbon atom summation among the described Y is 9 to 15, preferred 10 to 14.Average aliphatic carbon atom content among the described Y is about 10.0 to about 14.0, preferred about 11.0 to about 13.0, and more preferably 11.5 to 12.5; And the feature of wherein said alkylbenzenesulfonatsurfactants surfactants mixture is that also its 2/3-phenyl index range is about 275 to about 10000, and preferred about 350 to about 1200, more preferably from about 500 to about 700; And the 2-of preferred wherein said alkylbenzenesulfonatsurfactants surfactants mixture methyl-2-phenyl index is less than about 0.3, preferably less than about 0.2, is more preferably less than about 0.1; Especially especially preferred 0 to 0.05; (ii) about 0.1% to about 8% cosurfactant mixture is selected from alkyl polyhydroxy fatty acid amide, alkyl amido propyl-dimethyl amine and composition thereof; (iii) about 30% to about 95% water liquid carrier; And the feature of wherein said composition is that also its 2/3-phenyl index range is about 275 to about 10000.
Second embodiment according to the present invention the present invention includes a lot of interchangeable schemes, for example wherein other alkyl benzene sulfonate surfactant blend of the new improved alkylbenzene sulfonate surfactant mixture of the present invention and one or more.Aborning, this blend is carried out before sulfonation reaction and washing composition preparation, but final product is the water base heavy duty type laundry detergent composition that contains new improved alkylbenzene sulfonate surfactant mixture and other common alkylbenzene sulfonates.This interchangeable embodiment includes but are not limited to so-called " medium 2/3-phemyol surface active agent system " among the present invention.Basically contain the improved alkyl benzene sulfonate surfactant of significant quantity and other common alkyl benzene sulfonate surfactants in this surfactant system, make whole composition/3-phenyl index reaches the requirement of regulation.This water base heavy duty type laundry detergent composition comprises: (i) weight is about 0.1% to about 95% medium 2/3-phemyol surface active agent system of whole composition, and this system is made up of following component basically:
(1) weight be surfactant system 1% (preferably at least about 5%, more preferably at least about 10%) (preferably be lower than about 50% to 60%, more preferably less than about 40%) first kind of alkyl benzene sulfonate surfactant, here so-called first kind of alkyl benzene sulfonate surfactant refers to the alkylbenzenesulfonatsurfactants surfactants mixture, and this surfactant mixture comprises:
(a) weight is the mixture of about 60% to about 95% the branch-alkylbenzene sulfonate with following general molecular formula (I) of surfactant mixture:
The non-annularity aliphatic moiety formed by carbon and hydrogen of L wherein, this fragment has two methyl ends, and described L is except A, R
1And R
2Outward without any other substituting group; Contain the different described branch-alkylbenzene sulfonate of two or more anionicsite (seeing formula (I)) molecular weight in the described branch-alkylbenzene sulfonate mixture, in the wherein said branch-alkylbenzene sulfonate mixture:
-R
1, L and R
2The carbon atom summation be 9 to 15;
-average aliphatic carbon atom content is about 10.0 to about 14.0; M is that valency is positively charged ion or the cation mixt of q; A and b can keep the electroneutral integer of described branch-alkylbenzene sulfonate; R
1It is the alkyl of 1 to 3 carbon atom; R
2Be selected from the alkyl and the H of 1 to 3 carbon atom; A is a phenyl moiety; With
(b) weight is about 5% to about 40% the non-branched-chain alkyl benzene sulfonate mixture with following general molecular formula (II) of surfactant mixture:
Wherein identical in the definition of a, b, M, A and q and the general formula (I), Y is the linear aliphatic family fragment of the non-replacement be made up of carbon and hydrogen, and this fragment has two methyl ends, and the carbon atom summation among the described Y is 9 to 15, preferred 10 to 14.Average aliphatic carbon atom content among the described Y is 10.0 to 14.0;
The feature of wherein said alkylbenzenesulfonatsurfactants surfactants mixture is that also its 2/3-phenyl index range is about 275 to about 10000; (2) weight be surfactant system 40% (preferably at least about 50%, more preferably at least about 60%) (preferably be lower than about 95% to about 99%, more preferably less than about 90%) second kind of alkyl benzene sulfonate surfactant, here so-called second kind of alkyl benzene sulfonate surfactant refers to the alkylbenzene sulfonate surfactant mixture except described alkylbenzenesulfonatsurfactants surfactants mixture (1) in the system (although commercially available linearity in common any market (LAS) or side chain (ABS, TPBS) the class alkyl benzene sulfonate surfactant all can use, but general said second kind of alkyl benzene sulfonate surfactant is the linear alkyl benzene sulfonate tensio-active agent with 10 to 14 carbon atoms that market buys, as DETAL technology LAS or HF technology LAS etc.); And the 2/3-phenyl index range of second kind of alkyl benzene sulfonate surfactant is about 75 to about 160;
Supplementary condition are that the 2/3-phenyl index range of described medium 2/3-phemyol surface active agent system is 160 to about 275, (preferred about 170 to about 265, more preferably from about 180 to about 255); (ii) about 0.1% to about 8% cosurfactant composition is selected from alkyl polyhydroxy fatty acid amide, alkyl amido propyl-dimethyl amine and composition thereof; (iii) about 30% to about 95% water liquid carrier;
Especially, the 3rd embodiment of the present invention comprises water base heavy duty type laundry detergent compositions, comprising:
(i) a kind of improvement alkylbenzene sulfonate surfactant mixture, contain the product that is made by the method that comprises the following steps:
(I) use the alkylation mixture alkylated benzenes;
(II) (I) product is carried out sulfonation; With
(III) product to (II) neutralizes; Comprise in the wherein said alkylation mixture:
(a) the side chain monoolefine of about 1% of alkylation mixture weight to about 99.9% 9 to 20 carbon atoms, the structure of described side chain monoolefine and structural formula are R
1LR
2The similar of the side chain monoolefine that obtains of branched paraffin dehydrogenation, the non-annularity aliphatic moiety that L is made up of carbon and hydrogen in the formula has two terminal methyls; R
1Be the alkyl of 1 to 3 carbon atom, R
2Be selected from the alkyl and the H of 1 to 3 carbon atom; With
(b) weight is the linear aliphatic family alkene of about 0.1% to about 85% 9-20 carbon atom of alkylation mixture; Wherein said alkylation mixture contains the side chain monoolefine of 9-20 carbon atom, has at least two kinds of different carbonatomss in 9-20 carbon atom scope, and average carbon atom number is about 9.0 to about 15.0; And wherein component (a) and weight ratio (b) were at least about 15: 85;
(ii) about 0.1% to about 8% cosurfactant composition is selected from alkyl polyhydroxy fatty acid amide, alkyl amido propyl-dimethyl amine and composition thereof;
(iii) about 30% to about 95% water liquid carrier; And the feature of wherein said composition is that also its 2/3-phenyl index range is about 275 to about 10000.
Especially, the 4th embodiment of the present invention comprises water base heavy duty type laundry detergent compositions, comprising:
(i) a kind of alkylbenzenesulfonatsurfactants surfactants mixture, basically by the product slate of the method that comprises the following steps successively:
(I) use the alkylation mixture alkylated benzenes;
(II) (I) product is carried out sulfonation; With
(III) (II) product is neutralized; Comprise in the wherein said alkylation mixture:
(a) about 1% of alkylation mixture weight to about 99.9% branched-chain alkyl agent is selected from following component:
(A) the inside monoolefine R of 9 to 20 carbon atoms
1LR
2, the non-cyclic olefin fragment formed by carbon and hydrogen of L wherein, and contain two terminal methyls;
(B) α of 9 to 20 carbon atoms-monoolefine R
1AR
2, the non-annularity alpha-olefin fragment formed by carbon and hydrogen of A wherein, and contain a terminal methyl and a terminal olefine methylene radical;
(C) the vinylidene monoolefine R of 9 to 20 carbon atoms
1BR
2, the non-annularity vinylidene alkene fragment formed by carbon and hydrogen of B wherein, and contain two terminal methyls and an inner alkene methylene radical;
(D) the primary alconol R of 9 to 20 carbon atoms
1QR
2, the non-annularity aliphatics one-level terminal alcohol fragment formed by carbon, hydrogen and oxygen of Q wherein, and contain a terminal methyl;
(E) the primary alconol R of 9 to 20 carbon atoms
1ZR
2, the non-terminal alcohol fragment of non-annularity aliphatics one-level formed by carbon, hydrogen and oxygen of Z wherein, and contain two terminal methyls;
(F) mixture of above-mentioned several materials; Wherein in any one of (A) to (F), described R
1Be the alkyl of 1 to 3 carbon atom, R
2Be selected from the alkyl and the H of 1 to 3 carbon atom; With
(b) weight is the linear alkylated agent of about 0.1% to about 85% 9-20 carbon atom of alkylation mixture, is selected from the linear aliphatic family alkene of 9-20 carbon atom, linear aliphatic alcohol of 9-20 carbon atom and composition thereof; Wherein said alkylation mixture contains described branched-chain alkyl agent, has at least two kinds of different carbonatomss in 9-20 carbon atom scope, but average carbon atom number is about 9.0 to about 15.0; And wherein component (a) and weight ratio (b) were at least about 15: 85;
(ii) about 0.1% to about 8% cosurfactant mixture is selected from alkyl polyhydroxy fatty acid amide, alkyl amido propyl-dimethyl amine and composition thereof;
(iii) about 30% to about 95% water liquid carrier; The feature of wherein said composition is that also 2/3-phenyl index range is about 275 to about 10000.
Especially, the 5th embodiment of the present invention comprises water base heavy duty type laundry detergent compositions, comprising:
(i) be about 0.01% to about 95% alkylbenzenesulfonatsurfactants surfactants mixture of composition weight, comprise:
(a) be about 60% to about 95% branch-alkylbenzene sulfonate mixture of surfactant mixture weight with following general molecular formula (I):
The non-annularity aliphatic moiety be made up of carbon and hydrogen of L wherein, this fragment has two methyl ends, and described L is except A, R
1And R
2Outward without any other substituting group; Contain the different described branch-alkylbenzene sulfonate of two or more anionicsite (seeing formula (I)) molecular weight in the described branch-alkylbenzene sulfonate, and in the wherein said branch-alkylbenzene sulfonate mixture:
-R
1, L and R
2The carbon atom summation be 9 to 15;
-average aliphatic carbon atom content is about 10.0 to about 14.0; M is that valency is positively charged ion or the cation mixt of q; A and b can keep the electroneutral integer of described branch-alkylbenzene sulfonate; R
1It is the alkyl of 1 to 3 carbon atom; R
2Be selected from the alkyl and the H of 1 to 3 carbon atom; A is a phenyl moiety; With
(b) weight is about 5% to about 40% the non-branched-chain alkyl benzene sulfonate with following general molecular formula (II) of surfactant mixture:
Wherein identical in the definition of a, b, M, A and q and the general formula (I), Y is the non-replacement linear aliphatic family fragment of being made up of carbon and hydrogen, this fragment has two terminal methyls, and the carbon atom summation of wherein said Y is 9 to 15, preferred 10 to 14, and the average aliphatic carbon atom content of described Y is about 10.0 to about 14.0; And the feature of wherein said alkylbenzenesulfonatsurfactants surfactants mixture is that also its 2/3-phenyl index range is about 275 to about 10000; The 2-methyl of described alkylbenzenesulfonatsurfactants surfactants mixture-2-phenyl index is less than about 0.3;
(ii) about 0.1% to about 8% cosurfactant mixture comprises alkyl polyhydroxy fatty acid amide, alkyl amido propyl-dimethyl amine and composition thereof;
(iii) about 30% to about 95% water liquid carrier; With
(iv) about 0.00001% to about 99.9% surfactant composition is selected from anion surfactant except that (i), nonionic surface active agent, zwitterionics, cats product, amphoterics and composition thereof;
The feature of wherein said composition is that also its 2/3-phenyl index range is about 275 to about 10000; Supplementary condition are when any alkyl benzene sulfonate surfactant that contains in the described composition except that described alkylbenzenesulfonatsurfactants surfactants mixture, and the feature of said composition is that also its integral body/3-phenyl index is at least about 200.Wherein said integral body/3-phenyl exponential is measured, be according to defined method herein, measure alkylbenzenesulfonatsurfactants surfactants mixture and the 2/3-phenyl index that joins the blend of any other alkylbenzene sulfonate in this detergent composition, in order to measure, this blend is by the alkylbenzenesulfonatsurfactants surfactants mixture, with the aliquots containig preparation of described other alkylbenzene sulfonate that does not contact with any other component of detergent composition as yet; And another supplementary condition are, when described detergent composition comprises any alkyl benzene sulfonate surfactant except that described alkylbenzenesulfonatsurfactants surfactants mixture, the feature of this detergent composition is that also its whole 2-methyl-2-phenyl index is lower than about 0.3, wherein said whole 2-methyl-2-phenyl exponential is measured, be according to defined method herein, measure alkylbenzenesulfonatsurfactants surfactants mixture and the 2-methyl-2-phenyl index that joins the blend of any other alkylbenzene sulfonate in this detergent mixture, in order to measure, this blend is by the alkylbenzenesulfonatsurfactants surfactants mixture, with the aliquots containig preparation of described other alkylbenzene sulfonate that does not contact with any other component of detergent composition as yet.
Detergent composition of the present invention also contains other detergent components of about 1% to about 80% of composition weight, as washing assistant, enzyme, tinting material, SYNTHETIC OPTICAL WHITNER, bleach-activating agent, color spot, organic detergent builder compound, inorganic alkali source and composition thereof.
Above-mentioned embodiment of the present invention and others will illustrate more fully and illustrate in detailed Description Of The Invention after this.
The per-cent that relates in this specification sheets, ratio, ratio etc. specify and all to represent with weight except that having, all temperature except that have specify all with degree centigrade (℃) represent that the relevant portion of all references file is incorporated herein by reference.Detailed Description Of The Invention
Aqueous liquid detergent compositions of the present invention contains the alkylbenzenesulfonatsurfactants surfactants mixture.To be described in detail form, preparation and the application etc. (all concentration and ratio are all represented with weight) of basic and optional alkylbenzenesulfonatsurfactants surfactants component of mixture, other optional, materials in this aqueous liquid detergent compositions, composition below except that special instruction is arranged.On the other hand, the present invention does not want to comprise the liquid detergent composition of any routine, for example exclusively based on the liquid detergent composition of linear alkyl benzene sulfonate, this linear alkyl benzene sulfonate can be any method preparation, or exclusively based on known unsafty branched-alkyl benzene sulfonate, for example liquid detergent composition of ABS or TPBS.
Preferably (it is industrialized for example to be blended into one or more in detergent composition when detergent composition of the present invention contains any alkyl benzene sulfonate surfactant except that described alkylbenzenesulfonatsurfactants surfactants mixture, particularly linear, representational is linear C
10-C
14Alkyl benzene sulfonate surfactant) time, it is about 200 that the feature of described composition is that also its whole 2/3-phenyl index is at least, and preferably is at least approximately 250, more preferably is at least approximately 350, especially preferably is at least about 500.Wherein said whole 2/3-phenyl exponential is measured, be according to defined method herein, measure described alkylbenzenesulfonatsurfactants surfactants mixture and the 2/3-phenyl index that joins the blend of any other alkylbenzene sulfonate in the described composition, in order to measure, this blend is by described alkylbenzenesulfonatsurfactants surfactants mixture, with the aliquots containig preparation of described other alkylbenzene sulfonate that does not contact with any other component of described composition as yet; And another supplementary condition are, (it is industrialized for example to be blended into one or more in detergent composition when described composition comprises any alkyl benzene sulfonate surfactant except that described alkylbenzenesulfonatsurfactants surfactants mixture, particularly linear, representational is linear C
10-C
14Alkyl benzene sulfonate surfactant), the feature of said composition is that also its whole 2-methyl-2-phenyl index is lower than about 0.3, be preferably 0~0.2, more preferably no more than about 0.1, be most preferably not exceeding about 0.05, wherein said whole 2-methyl-2-phenyl exponential is measured, be according to defined method herein, measure described alkylbenzenesulfonatsurfactants surfactants mixture and the 2-methyl-2-phenyl index that joins the blend of any other alkylbenzene sulfonate in the described composition, in order to measure, this blend is by described alkylbenzenesulfonatsurfactants surfactants mixture, with the aliquots containig preparation of described other alkylbenzene sulfonate that does not contact with any other component of detergent composition as yet.Some is unusual for these regulation possibilities, but they are consistent with the spirit and scope of the present invention, it comprises some economy, but to whole clean-up performance is not preferable methods, for example can be when synthetic, or when composite, detergent composition is advanced in alkylbenzenesulfonatsurfactants surfactants and conventional linear alkyl benzene sulfonate tensio-active agent blend.And as the manual well-known analysis of dealer of washing the dishes, some manual annexes that wash the dishes (paramagnetic material, sometimes or even water) can influence the method for after this disclosed mensuration alkylbenzene sulfonate surfactant mixture parameter.Therefore as possible, just should before material mixing is gone into composition, dry substance be analyzed.
Also contain hydrotrote precursor and/or hydrotrote in the present composition, alkylbenzene as 1 to 8 carbon atom, it more typically is the sulfonated derivative of toluene, isopropyl benzene, dimethylbenzene, naphthalene or these materials, and a spot of any other material, as three branch-alkylbenzene sulfonate tensio-active agents, dialkyl benzene and derivative thereof, dialkyl group tetraline, wetting agent, processing aid or the like.Should be understood that except that hydrotrote, do not comprise any such material among the present invention usually.They should be understood that equally and if when such material impact analysis method, will be not included in the composition sample that is used for analysis purposes.
Be selected from H, Na, K and composition thereof according to the M in the preferred alkylbenzenesulfonatsurfactants surfactants mixture of first embodiment of the present invention, described a=1, b=1, q=1, the 2-methyl of described alkylbenzenesulfonatsurfactants surfactants mixture-2-phenyl index is less than about 0.3, preferably less than about 0.2, more preferably 0 to about 0.1.
These alkylbenzenesulfonatsurfactants surfactants mixtures are products that the zeolite by being selected from the mordenite, offretite or the H-ZSM-12 that are at least the part acid form prepares as the method for catalyzer, catalyzer is preferably acidic mordenites (some zeolite beta also can be used as catalyzer generally speaking, but effect is not fine).After this embodiment for preparing them, and appropriate catalyst will be described in further detail.
Another preferred detergent composition according to first embodiment of the present invention, alkylbenzenesulfonatsurfactants surfactants mixture is wherein gone up by (a) substantially and (b) is mixed and to form, the 2-methyl of wherein said alkylbenzenesulfonatsurfactants surfactants mixture-2-phenyl index is less than about 0.1, and average aliphatic carbon atom is about 11.5 to about 12.5; R
1It is methyl; R
2Be selected from methyl or H, supplementary condition are in the described branched-alkyl benzene sulfonate at least about 0.7 molar fraction, R
2Be H; R
1, L and R
2Middle the total number of carbon atoms is 10 to 14; In described non-branched-chain alkyl benzene sulfonate mixture, the carbon atom summation of described Y is 10 to 14; The average aliphatic carbon atom content of described non-branched-chain alkyl benzene sulfonate is about 11.5 to about 12.5; Described M is univalent cation or the cation mixt that is selected from H, Na and composition thereof.
Definition:
Terminal methyl: term " methyl end " and/or " terminal methyl " are meant the alkyl fragment, and promptly L in formula (I) and formula (II) and/or the terminal carbon of Y always are connected with three hydrogen atoms respectively.That is to say that they form CH
3-group.For explaining this point better, following structure demonstrates two end capped methyl in alkylbenzene sulfonate.
The abbreviation of so-called " gravity die " or non-biodegradation type " alkylbenzene " represented in term " AB " when not having specified otherwise, its sulfonated products is " ABS ".Term " LAB " is the abbreviation of industrialized biodegradable " LINEAR ALKYL BENZENE (LAB) " at present, and its sulfonation forms linear alkyl benzene sulfonate, is abbreviated as " LAS ".Term " MLAS " is the abbreviation of modified alkylbenzene sulfonates mixture of the present invention.
Impurity: surfactant mixture described here does not preferably contain substantially and is selected from three side chain impurity, the impurity of dialkyl group tetraline impurity and composition thereof.So-called " being substantially free of " refers to that foreign matter content in the system is not enough to influence the cleaning performance of detergent composition.It is about 5% that foreign matter content is generally less than, and preferably less than about 1%, is more preferably less than about 0.1%.Various generally speaking impurity in fact all be detect less than.
Topology example
For the complicacy of better explanation alkylbenzenesulfonatsurfactants surfactants mixture of the present invention and the detergent composition that makes, below structural formula (a) to (v) example the part preferred compound of formula (I) expression.These structures only are some that can form in the possible preferred structure of the hundreds of of a large amount of compositions, and should not regard limitation of the present invention as.
The structure (w) and (x) compound of the not too preferred formula (I) of explanation without limitation, it can be to exist than the low level of above-mentioned illustrative preferred structure type in alkylbenzenesulfonatsurfactants surfactants mixture of the present invention and in the detergent composition that obtains.
Structure (y), (z) and (aa) popularity of compound in formula (I) scope without limitation, it is not preferred, but it may reside in the alkylbenzenesulfonatsurfactants surfactants mixture of the present invention and in the detergent composition that obtains.
Structure (bb) illustrates not three branched structures in formula (I) scope, but it can be used as the impurity existence.
Preferred alkylbenzenesulfonatsurfactants surfactants mixture is the sulfonated products of modified alkylbenzene (except tetrapropylene base class and AB class that everybody knows), modified alkylbenzene is the branched-chain alkene alkylating product of benzene except that the tetrapropylene base, more specifically be the slight branching type that hereinafter describes in detail, will be described in detail hereinafter about mixture and as the acidic mordenites of catalyzer and other suitable catalyzer.
Sometimes described alkylbenzenesulfonatsurfactants surfactants mixture also can prepare by mixed.Therefore, the present invention includes the detergent composition of use according to the alkylbenzenesulfonatsurfactants surfactants mixture of first embodiment, wherein said alkylbenzenesulfonatsurfactants surfactants mixture can be prepared from by comprising the method that is selected from following step: (i) be that 500 to 700 branching and linear alkyl benzene sulfonate surfactant mixtures and 2/3-phenyl index are 75 to 160 alkylbenzene sulfonate surfactant mixture blend with 2/3-phenyl index; (ii) be 500 to 700 branching and linear alkyl benzol mixture with 2/3-phenyl index with/3-phenyl index is 75 to 160 alkylbenzene mixture blend, then blend is carried out sulfonation.When the alkylbenzenesulfonatsurfactants surfactants mixture by present method preparation, the 2/3-phenyl index that obtains is about 275 to about 10000.
Put it briefly, the alkylbenzenesulfonatsurfactants surfactants mixture can prepare by following steps:
(I) use the alkylation mixture alkylated benzenes;
(II) (I) product is carried out sulfonation; (randomly, but most preferably)
(III) (II) product is neutralized;
If the suitable alkylation catalyst and the processing condition that adopt the present invention to require, the product of step (I) is the modification alkylated benzenes mixture that the present invention requires.If adopt the LAS manufacturer commonly used and repeatably condition carry out (relevant condition can be consulted the document that this patent is quoted), the product of step (II) is the modified alkylbenzene azochlorosulfonate acid mixture that the present invention requires.If require to carry out the neutralization reaction of step (III) according to the present invention, the product of that step (III) is the alkylbenzenesulfonatsurfactants surfactants mixture that the present invention requires.Because neutralizing effect may be incomplete, so with various ratios, the acid of the modified alkylbenzene sulfonates system of the present invention that the ratio of for example about 1000: 1~1: 1000 (weight) exists and the mixture of corrective also are parts of the present invention.Generally speaking, that most critical is step (I).
The preferred alkylbenzenesulfonatsurfactants surfactants mixture of the present invention contains the product by the method preparation that comprises the following steps: (I) use the alkylation mixture alkylated benzenes; (II) (I) product is carried out sulfonation; (randomly, but most preferably) (III) neutralizes to (II) product.Wherein said alkylation mixture comprises: (a) the side chain monoolefine of about 1% 9 to 20 carbon atoms to about 99.9% (weight) (preferred 9 to 15 carbon atoms, more preferably 10 to 14 carbon atoms), the structure of described side chain monoolefine and structural formula are R
1LR
2The side chain monoolefine similar that obtains of branched paraffin dehydrogenation, the non-annularity aliphatic moiety that L is made up of carbon and hydrogen in the formula, and contain two terminal methyls; R
1Be the alkyl of 1 to 3 carbon atom, R
2Be selected from the alkyl or the H of 1 to 3 carbon atom; (b) the linear aliphatic family alkene of about 0.1% 9-20 carbon atom (preferred 9 to 15 carbon atoms, more preferably 10 to 14 carbon atoms) to about 85% (weight); Wherein said alkylation mixture comprises described side chain C
9-C
20Monoolefine, it has at least two kinds of different carbonatomss in described 9-20 carbon atom scope, average carbon atom number be about 9.0 to about 15.0 (preferred about 10.0 to about 14.0 carbon atoms, more preferably from about 11.0 to about 13.0 carbon atoms, and preferred about 11.5 to about 12.5 carbon atoms especially); Wherein said component (a) and weight ratio (b) are at least about 15: 85, and (preferred branched fraction (a) surpasses linear component (b), is 51% or more as the weight percent of component (a), and component (b) weight percent is 49% or still less; More preferably component (a) weight percent is 60% to 95%, and component (b) weight percent is 5% to 40%; Special preferred ingredient (a) weight percent is 65% to 90%, component (b) weight percent is 10% to 35%, more special preferred ingredient (a) weight percent is 70% to 85%, component (b) weight percent is 15% to 30%, wherein these weight percents do not comprise any other material, for example the hydrocarbon diluent that can exist in this technology).
The present invention also comprises the alkylbenzenesulfonatsurfactants surfactants mixture of being made up of the product of following technology basically, and described processing step is in order: (I) use the alkylation mixture alkylated benzenes; (II) step (I) product being carried out sulfonation (III) neutralizes to step (II) product.Wherein said alkylation mixture comprises: (a) the about 1% branched-chain alkyl agent to about 99.9% (weight) is selected from: (A) the inside monoolefine R of 9 to 20 carbon atoms (preferred 9 to 15 carbon atoms, more preferably 10 to 14 carbon atoms)
1LR
2, wherein the non-cyclic olefin fragment be made up of carbon and hydrogen of L contains two terminal methyls; (B) α-monoolefine R of 9 to 20 carbon atoms (preferred 9 to 15 carbon atoms, more preferably 10 to 14 carbon atoms)
1AR
2, wherein the non-annularity alpha-olefin fragment be made up of carbon and hydrogen of A contains a terminal methyl and a terminal olefine methylene radical; (C) the vinylidene monoolefine R of 9 to 20 carbon atoms (preferred 9 to 15 carbon atoms, more preferably 10 to 14 carbon atoms)
1BR
2, wherein the non-annularity vinylidene alkene fragment be made up of carbon and hydrogen of B contains two terminal methyls and an inner alkene methylene radical; (D) the primary alconol R of 9 to 20 carbon atoms (preferred 9 to 15 carbon atoms, more preferably 10 to 14 carbon atoms)
1QR
2, wherein the non-annularity aliphatics one-level terminal alcohol fragment be made up of carbon, hydrogen and oxygen of Q contains a terminal methyl; (E) the primary alconol R of 9 to 20 carbon atoms (preferred 9 to 15 carbon atoms, more preferably 10 to 14 carbon atoms)
1ZR
2, wherein the non-terminal alcohol fragment of non-annularity aliphatics one-level be made up of carbon, hydrogen and oxygen of Z contains two terminal methyls; (F) mixture of above-mentioned several materials; Wherein in any one of (A) to (F), described R
1Be the alkyl of 1 to 3 carbon atom, R
2Be selected from the alkyl or the H of 1 to 3 carbon atom; (b) about 0.1% to about 85% 9-20 carbon atom (preferred 9 to 15 carbon atoms, more preferably 10 to 14 carbon atoms) linear alkylated agent, be selected from 9-20 carbon atom (preferred 9 to 15 carbon atoms, more preferably 10 to 14 carbon atoms) linear aliphatic family alkene, linear aliphatic alcohol of 9-20 carbon atom (preferred 9 to 15 carbon atoms, more preferably 10 to 14 carbon atoms) and composition thereof; Wherein said alkylation mixture contains described branched-chain alkyl agent, it is (preferred 9 to 15 carbon atoms in described 9-20 carbon atom scope, preferred especially 10 to 14 carbon atoms) have at least two kinds of different carbonatomss, average carbon atom number is about 9.0 to about 15.0 (preferred about 10.0 to about 14.0, more preferably from about 11.0 to about 13.0, preferred about 11.5 to about 12.5 especially); Component (a) and weight ratio (b) are at least about 15: 85, and (preferred branched fraction (a) surpasses linear component (b), is 51% or more as the weight percent of component (a), and component (b) weight percent is 49% or still less; More preferably component (a) weight percent is 60% to 95%, and component (b) weight percent is 5% to 40%; Special preferred ingredient (a) weight percent is 65% to 90%, and component (b) weight percent is 10% to 35%; Special preferred ingredient (a) weight percent is 70% to 85%, and component (b) weight percent is 15% to 30%, and wherein these weight percents do not comprise any other material, for example the hydrocarbon diluent that can exist in this technology).
In highly preferred embodiment more, the alkylbenzenesulfonatsurfactants surfactants mixture that the present invention includes is to be prepared from by above-mentioned processing step, wherein said alkylation mixture is made of following component basically: (a) the about 0.5% branched-chain alkyl mixture to about 47.5% (weight) is selected from: (G) the inside monoolefine R of 9 to 14 carbon atoms
1LR
2, wherein the non-cyclic olefin fragment be made up of carbon and hydrogen of L contains two terminal methyls; (H) α of 9 to 14 carbon atoms-monoolefine R
1AR
2, wherein the non-annularity alpha-olefin fragment be made up of carbon and hydrogen of A contains a terminal methyl and a terminal olefine methylene radical; (J) both mixtures; Wherein in any one of (G) to (H), described R
1Be methyl, R
2Be H or methyl, supplementary condition are at least about in the whole described monoolefine of 0.7mol fractional, R
2Be H; (b) about 0.1% to the linear aliphatic olefin hydrocarbon of 9 to 14 carbon atoms of about 25% (weight); (c) about 50% carrier substance to about 98.9% (weight) comprises the non-alkane solvent of alkane and inertia; Wherein said alkylation mixture contains described branched-chain alkyl agent, and it has at least two kinds of different carbonatomss in 9 to 14 carbon atom scopes, and average carbon atom number is about 11.5 to about 12.5; Wherein said component (a) and weight ratio (b) are about 51: 49 to about 90: 10.
Other alkylbenzenesulfonatsurfactants surfactants mixtures of the present invention also can form by above-mentioned prepared, wherein in the presence of alkylation catalyst, carry out in the alkylation described in the step (I), described catalyst for alkylation reaction is a kind of multi-hole type solid alkylation catalyst of moderate acid, and the product that step (II) is included in step (I) contacts preceding other components that need except that removing monoalkylated benzenes with sulfonated reagent.
Alkylbenzenesulfonatsurfactants surfactants mixture of the present invention also can be by above-mentioned prepared, and wherein catalyst for alkylation reaction is to be different to be selected from HF, AlCl
3, sulfuric acid and composition thereof catalyzer.Catalyst for alkylation reaction is selected from acidic mordenites type catalyzer, fluorizated acidic mordenites type catalyzer of nonfluorinated and composition thereof during this situation.Relevant catalyzer will be described in detail hereinafter.
Above-mentioned technology can have various changes, as can step (I) (II) and (III) before and after or add some conventional steps simultaneously.This technology is particularly suitable for using the situation that helps water solvent or its precursor.Therefore, alkylbenzenesulfonatsurfactants surfactants mixture of the present invention can form by above-mentioned prepared, wherein helps water solvent, helps water solvent precursor or its mixture to add in step (I) back; Or help water solvent, help water solvent precursor or its mixture can be in step (II) or after the step (II), and the preceding adding of step (III); Or help water solvent also can in step (III) or after the step (III), add.
Sulfonation and processing or neutralization (Step II/III)
Usually, the sulfonation of modified alkylbenzene of the present invention can use any well-known sulfonation system to finish, comprise the report in the various documents, as (Sitting edits in " containing the washing composition production of zeolite builders and other novel substances ", Noyes Data company publishes, 1979), and " tensio-active agent science " series of books (Marcel Dekker publication, New York, 1992) 56 volumes, particularly including chapter 2, title is " alkylaryl sulfonate: history, production, analysis and environmental performance ", the 39-108 page or leaf comprises 297 pieces of reference.Wherein a large amount of bibliographical informations various technologies and processing step, not only comprise sulfonation reaction, also comprise technologies such as dehydrogenation reaction, alkylated reaction, alkylbenzene distillation.Suitable sulfonation reaction system of the present invention generally comprises sulfuric acid, chlorsulfonic acid, oleum, sulphur trioxide or the like.Preferred especially sulphur trioxide/air system.United States Patent (USP) 3,427,342 (contriver Chmithon) have disclosed employing sulphur trioxide/air system in detail and have carried out the details of sulfonation reaction.The technology of relevant sulfonation reaction has detailed narration in the book of " sulfonation technology in the detergent industry " (W.H.de Groot, Kluwer academic press, Boston, 1991).Can adopt the treatment step of any routine in the methods of the invention.Adopt any suitable alkali to neutralize after the sulfonation reaction of being everlasting in the practice.Therefore can use the alkali of alkali of being selected from sodium, potassium, ammonia, magnesium and replacement ammonium and composition thereof to carry out neutralization procedure.Potassium helps to improve solvability, and magnesium can promote the performance of softening water, and the ammonium of replacement can help to prepare various special tensio-active agent of the present invention.The present invention includes any this class derivative form of the alkylbenzenesulfonatsurfactants surfactants of producing with present method, and their application in consumer product compositions.
Selectively, the tensio-active agent of the present invention of sour shape can directly join in the tart cleaning product, or can mix with cleaning component, neutralizes then.
What the present invention was used helps water solvent or helps the water solvent precursor can be selected from any suitable helping water solvent or help the water solvent precursor, comprise low alkyl group (1 to 8 carbon atom) aromatic compound and sulfonic acid and sulfonate, but more typical sulfonic acid and the sulfonate sodium derivative that is based on toluene, dimethylbenzene, isopropyl benzene, naphthalene or its mixture.Help the water solvent precursor to be selected from any suitable water solvent precursor that helps, be generally toluene, dimethylbenzene, isopropyl benzene, naphthalene or its mixture.Help the water solvent precursor be a kind of be to be converted into the compound that helps water solvent in the sulfonation procedure in step (III).
Processing condition according to alkylated reaction, in alkylbenzenesulfonatsurfactants surfactants mixture of the present invention, step (I) is to be about 125 ℃ to about 230 ℃ (preferred about 175 ℃ to about 215 ℃) in temperature, and pressure extremely carries out under about 1000psig (preferably about 100psig is about 250psig extremely) condition for about 50psig.The preferred processing condition of the alkylated reaction described in the step (I) is about 175 ℃ to about 215 ℃ an of temperature, the about 100psig of pressure is to about 250psig, reaction times is about 0.01 hour to about 18 hours (the preferred reaction time is short as far as possible, preferred about 0.1 to about 5 hours especially).If desired, this alkylated reaction can be finished in a step, also can proceed step by step.The different steps of this method can be carried out on different production units.In fact, step (I) is normally finished by the LAB manufacturer, and step (III) is normally finished by detergent manufacturers.Step (II) both can be finished by the LAB manufacturer, also can be finished by detergent manufacturers, even can also be finished by the third party.
In above-mentioned scope, step (I) is preferred usually to adopt relatively low temperature of reaction (as 175 ℃ to about 215 ℃) and the medium reaction times (1 to about 8 hours) to carry out.
By selecting relatively low temperature of reaction, 190 ℃ according to appointment, and use any method easily (as sampling and carry out NMR and analyze) monitoring reaction process, so that under the situation that makes 2-methyl-2-phenyl index maintenance minimum, guarantee to react completely, can realize the relatively low 2-methyl-2-phenyl index of the present composition like this.
And, can expect, can proceed step by step at this alkylated reaction of step (I), that is to say the two or more reactors that turn round under the differential responses condition that can use in institute's limited range.By in a plurality of reactors, reacting, can allow to have more preferred 2-methyl-2-phenyl exponential material in initial stage formation, and astoundingly, can more such material be converted into and have preferred 2-methyl-2-phenyl exponential material.
Aspect the selection of sulfonated reagent, the present invention includes the alkylbenzenesulfonatsurfactants surfactants mixture, wherein used sulphonating agent is selected from sulphur trioxide, sulphur trioxide/air mixture, sulfuric acid (comprising oleum) in the step (II).Though chlorsulfonic acid or other known sulphonating agents are directly bought difficulty from market, also be useful, and comprise in the present invention.
Although neutralization procedure (III) can adopt any suitable alkaline matter to finish usually, but the present invention includes a kind of alkylbenzenesulfonatsurfactants surfactants mixture, use basic salt to finish in the wherein said step (III), positively charged ion in this basic salt is selected from basic metal, alkaline-earth metal, ammonium, replacement ammonium and composition thereof, and negatively charged ion is selected from hydroxide radical, oxonium ion, carbonate, silicate, phosphate radical and composition thereof.Preferred basic salt is selected from sodium hydroxide, water glass, potassium hydroxide, potassium silicate, magnesium hydroxide, ammonium hydroxide and composition thereof.
Catalyst for alkylation reaction
In order to obtain alkylbenzenesulfonatsurfactants surfactants mixture of the present invention, the present invention adopts specific catalyst for alkylation reaction.Described catalyst for alkylation reaction is a kind of moderate acid multi-hole type solid alkylation catalysts as will be detailed later.Particularly preferred catalyst for alkylation reaction comprises the acid fluorine-containing mordenite to the small part dealuminzation, to acid floride-free mordenite of small part dealuminzation and composition thereof.
Most catalyst for alkylation reaction also are not suitable for preparation modified alkylbenzene mixture of the present invention and alkylbenzenesulfonatsurfactants surfactants mixture.The catalyst for alkylation reaction that is not suitable for the present invention's use comprises any following material: sulfuric acid, aluminum chloride and HF.Nonacid calcium mordenite and many other catalyzer are not suitable for the present invention yet.Other catalyzer also are unaccommodated as the catalyzer DETAL of UOP, and its catalyzer of commercially producing at present is unaccommodated at least.In fact, when the linear alkyl benzene sulfonate of suitability for industrialized production laundry detergent products C 10 to C14, there is not suitable catalyst for alkylation reaction to be suitable for it is carried out alkylated reaction at present.
On the contrary, the catalyst for alkylation reaction that suitable the present invention uses is selected from the catalyst for alkylation reaction of the moderate acid of selecting shape, preferred zeolite.It is mordenite, HZSM-12 and the offretite of acid form that the zeolite catalyst that suitable alkylated reaction step (I) is used is preferably selected to small part.Also can use these mixture of catalysts, also can be with catalyst junction altogether with described tackiness agent after this etc.More preferably, described zeolite is essentially acid form, and is included in the granules of catalyst that contains tackiness agent commonly used, comprise at least about 1% in the described granules of catalyst, and more preferably at least 5%, preferred especially 50% to 90% above-mentioned zeolite.
More generally, a kind of suitable alkylation catalyst to small part is a crystal, is crystal basically more preferably, does not form particle, aggregate or employed tackiness agent of mixture or other materials and do not contain.And described catalyzer is acid to small part usually.It is unaccommodated for example exchanging calcium type mordenite entirely, and h-mordenite is suitable for.
The hole of the zeolite that uses in the alkylated reaction of the present invention is circular basically, and the hole of the homogeneous of about 6.2 , or preferably slightly a little ellipticity are as mordenite.Should be understood that, under any circumstance, the pore size of majority of zeolite that is used as catalyzer in the alkylated reaction of the present invention between the aperture of large pore zeolite such as X zeolite and Y zeolite and relative pore zeolite ZSM-5 and ZSM-11, preferably between about 6 to about 7 .In fact, the present invention once tried out ZSM-5, but unworkable.The aperture and the crystalline texture of some zeolite is specified in " zeolite structure type handbook " (W.M.Meier and D.H.Olson, structure branch of international zeolite federation publish, 1978 or upgrade version, Polycrystal Book Service, Pittsburg, Pa distribution).
On the zeolite that uses in the inventive method alkylation step, there is at least 10% positively charged ion site occupied usually by the ion beyond basic metal and the alkaline-earth metal.Typical but the replacement ion of indefiniteness comprises ammonium, hydrogen, rare earth, zinc, copper and aluminium.In this group ion, particularly preferably be ammonium, hydrogen, rare earth or its combination.In a preferred embodiment, by replacing original alkalimetal ion or other ions with the hydrogen ion precursor, be converted into mainly is Hydrogen to zeolite usually, and described hydrogen ion precursor such as ammonium ion can obtain Hydrogen by calcining.Ion-exchange utilizes well-known ion exchange technique usually, contacts with ammonium salt solution such as ammonium chloride by zeolite and finishes.In certain preferred aspects, the degree of ion-exchange will reach in the zeolitic material at least 50% positively charged ion site and occupied by hydrogen ion.
Zeolite can pass through various chemical treatments, comprises aluminium extraction (dealuminzation) and and one or more metal components, particularly IIB, III, IV, VI, VII and VIII family metal mixed.Can expect that zeolite also may need thermal treatment sometimes, comprise steam treatment or in air, hydrogen or environment such as rare gas element such as nitrogen, helium, calcine.
Suitable modification is handled and need be made zeolite and the atmosphere that contains 5%~about 100% steam of having an appointment, and contacts under about 250 ℃~1000 ℃ temperature, carries out decatize.Decatize can continue about 0.25~about 100h, and can carry out being lower than under normal atmosphere~hundreds of atmospheric pressure.
When carrying out the required alkylation step of present method, make the crystalline zeolite of above-mentioned mesopore be combined in another kind of material, be useful in the tackiness agent of the temperature of using in this method of ability and other condition or the matrix for example.Such body material comprises synthetic or natural substance, and inorganic materials, clay for example, silicon-dioxide, and/or metal oxide.Body material can be the gel form that comprises the mixture of silicon-dioxide and metal oxide.Metal oxide can be natural, or the form of gel or gelatinous precipitate.The clay that can comprise polynite and kaolin group with zeolite compound natural clay, this family's clay comprises inferior wilkinite and is commonly referred to Dixie, the kaolin of McNamee-Georgia and Florida clay, or wherein essential mineral consists of halloysite, kaolin, dickite, other clay of nakrite or anauxite.This class clay can be calcined with state or elder generation's process that original exploitation is come out, uses after acid treatment or the chemical modification.
Except that above-mentioned materials, as used herein mesopore zeolite can with porous body material, for example aluminum oxide, silica-alumina, silica-magnesia, silicon-dioxide-zirconium white, silica-thorium oxide, silica-beryllia, and silica-titania, and ternary composition, for example silica-alumina-Thorotrast, silica-alumina-zirconium white, silica-alumina-magnesium oxide and silica-magnesia-zirconium white are used in combination.Matrix can be cogelled form.The relative proportion of the zeolite of fine segmentation and inorganic oxide gel matrix can change in very wide scope, zeolite content can be mixture weight about 1~about 99%, be more typically about 5%~about 80%.
Comprise one group of zeolite of the zeolite of the alkylation step that some are used in this, its silicon-dioxide: the ratio of aluminum oxide was at least 2: 1, preferably was at least 10: 1, more preferably was at least 20: 1.The silicon-dioxide of mentioning in this manual: alumina ration is structure or framework ratio, i.e. SiO
4With AlO
4Tetrahedral ratio.In fact, with the silicon-dioxide of various physics and chemical gauging: alumina ration is an acceptable for application herein.Should be understood that these methods may provide some acceptable differences.For example, a kind of complete chemical analysis method may comprise the aluminium of the cationic form existence of accompanying with the acid position of zeolite, can obtain the silicon-dioxide of low test determination thus: alumina ration.Similarly, if this ratio is to measure by the thermo-gravimetric analysis (TGA) of ammonia desorption,, can obtain low ammonia titration value if aluminium cations stops ammonium ion to exchange on the acid position.These differences are well-known in the art.When adopting some method to handle, for example following causing do not exist aluminum ions dealumination process to handle in zeolite structured, and these differences are special troubles.Therefore, should be careful especially to guarantee correctly to measure framework silicon-dioxide: the ratio of aluminum oxide, the precision of this mensuration should be that this area practitioner institute is receptible.
When zeolite be in the presence of organic cation when preparation, they normally do not have catalytic activity, this may be because intracrystalline freeboard is come the organic cation of solution of self-forming zeolite occupied.For example by under inert atmosphere,, carry out the alkali exchange with ammonium salt subsequently, then in air, calcine in 540 ℃, can activate these zeolites in 540 ℃ of heating 1h.For the formation of zeolite, not that absolute demand exists organic cation in the solution of formation zeolite; It is favourable that but organic cation demonstrates the zeolite that forms this special shape really.Some natural zeolites are by various activation procedure and other processing, and for example alkali exchanges, decatize, and aluminum oxide extracts and calcining, can change into the zeolite of required type sometimes.This zeolite is under exsiccant hydrogen shape, and preferred crystallization framework density is not less than about 1.6g/cm basically
3Dry density for known structure, can add that the number of aluminium atom calculates by the Siliciumatom of per 1000 cubic angstroms, see, for example " Proceedings of the Conference on Molecular Sieves, London; April1967 ", published by the Society of Chemical Industry, London is in 1968, W.M.Meier about zeolite structured article, the 19th page.Quote this piece article so that crystallization framework density to be discussed.U.S. Patent No. 4,016 has further been discussed crystallization framework density in 218 and to the evaluation of some typical zeolite, the document is also quoted at this as a reference.When the synthetic zeolite was alkali metal form, this zeolite was easy to change into hydrogen shape, usually by ammonium ion exchange to form ammonium shape, calcine ammonium shape again to obtain hydrogen shape.Have now found that, although hydrogen shape zeolite successfully catalysis reaction, this zeolite also can partly be basic metal shape and/or other metal-salt shapes.
EP466,558 have disclosed a kind of acidic mordenites class catalyst for alkylation reaction that also can be suitable in the present invention, the sial atomic ratio of this zeolite is 15 to 85 (15-60), the weight content of sodium is less than 1000ppm (preferably less than 250ppm), non-structure aluminium content is few or be zero, and according to EP466,558 is defined, basic sieve aperture volume is less than 2,760nm
3
USP 5,057, and 472 have disclosed the preparation method who is applicable to catalyst for alkylation reaction of the present invention, and this patent relates to the stable zeolite that contains sodium ion of acid, and preferred mordenite carries out dealuminzation and ion-exchange simultaneously.This processing is to make zeolite and contain enough NH
4NO
3The HNO of 0.5 to 3 (preferred 1 to 2.5) M
3Solution contacts, with Na
+Be exchanged for NH fully
4 +And H
+SiO in the zeolite that obtains
2: Al
2O
3Be 15: 1 to 26: 1, preferred 17: 1 to 23: 1, and preferably calcine so that near small part NH
4 +/ H
+Type is converted into H
+Type.Though be not essential especially in the present invention, randomly, described catalyst for alkylation reaction can contain the calcined zeolite and the VIII family metal (optional inorganic oxide in addition) of No. 472 patents.
USP4,861,935 have disclosed the another kind of acidic mordenite catalyst that is applicable to alkylation step of the present invention, and this patent relates to a kind of and aluminum oxide bonded h-mordenite, and the surface-area of this mixture is minimum to be 580m
2/ g.Other the acidic mordenite catalyst that is applicable to alkylation step of the present invention comprises and is disclosed in USP5, the catalyzer in 243,116 and USP5,198,595.USP5,175,135 have also disclosed another kind is applicable to acidic mordenites catalyst for alkylation reaction of the present invention, silica alumina ratio in this acidic mordenites was at least 50: 1, the symmetry index that records by the x-ray diffraction analysis is at least 1.0, total pore volume is that porosity is about 0.18cc/g to about 0.45cc/g, and is medium and be about 0.25 to about 0.75 than the ratio that the pore volume of macropore accounts for total pore volume.
The particularly preferred catalyst for alkylation reaction of the present invention comprises the acidic mordenite catalyst Zeocat of Zeochem company
TMThe CBV 90A of FM-8/25H Zeolyst International company; The LZM-8 of UOP chemical catalyst company and the fluorinated product of above-mentioned commercialization catalyzer.Fluoridizing mordenite can prepare by several different methods.USP5,777,187 have disclosed and a kind ofly can provide useful especially method of fluoridizing mordenite.The present invention includes the preferred embodiment that mordenite is fluoridized in wherein use, but also comprise other preferred embodiments of using nonfluorinated mordenites.
The most at large, as long as alkylation catalyst can (a) catalyzer in minimum hole also can hold branched-chain alkene of the present invention, (b) can with described branched-chain alkene and randomly with the mixture of non-branching alkene alkylated benzenes optionally, all alkylation catalysts all can use in the present invention.Acceptable selectivity is as defined in this, and 2/3-phenyl index is 275 to 10000.
On the other hand, catalyst selectivity partly is the formation (as 4-phenyl, 5-phenyl etc.) of alkylbenzene in reducing.The prescription personnel of this patent are surprised to find that, control interior alkyl benzene sulphonate (ABS) isomer in the surfactant mixture of the present invention, and introduce limited methyl branch, and be very useful for improving its performance.The present invention will find to link together with the discovery of synthetic chemistry of the present invention man.How synthetic chemistry man has determined the requirement according to the prescription personnel, in the alkylbenzenesulfonatsurfactants surfactants mixture, and when limited methyl branch is provided, the content of control internal (position) isomer.
The control degree of internal (position) isomer content, can according to the application of the consumer's goods and according to whether the outstanding optimal performance of needs, or require the factors such as balance of performance and cost to change.Say that utterly the content of internal (position) isomer such as interior alkylbenzene isomer preferably generally remains on below 25% (weight), but, should remain on 0 to 10%, preferably less than about 5% (weight) in order to reach best result.Comprise that at this defined " interior alkylbenzene " isomer phenyl is connected 4,5,6 or 7 alkylbenzene on the aliphatic chain.
Do not want to be limited by theory, but preferred alkylation catalyst is selected the above-mentioned shape zeolites catalyzer of selecting, especially mordenite has following two reasons: the one, can optionally synthesize preferred compound, as branching or non-branching 2-phenyl or 3-phenylalkyl benzene.This selectivity is by 2/3-phenyl assessment of indices.Second reason is to control the amount of quaternary alkylbenzene, thus the amount of control quaternary alkylbenzene sulfonate.
Document (consulting J.Org.Chem., 37 volumes, 25,1972) is pointed out, uses the catalyst for alkylation reaction as HF, will provide very high-load quaternary alkylbenzene.This is opposite with discovery of the present invention, in the present invention, by benzene and branched-chain alkene catalyzed reaction, can obtain the quaternary alkylbenzene (characterizing with 2-methyl-2-phenyl index) of low levels.In the present invention even use when being essentially the alkene of two branching, as this illustrational, also can obtain low 2-methyl-2-phenyl index less than 0.1.
Detergent composition of the present invention can also have many change examples, and these become example and comprise:
● detergent composition is substantially free of other alkyl benzene sulfonate surfactants except above-mentioned alkylbenzenesulfonatsurfactants surfactants mixture;
● detergent composition, contain composition weight at least about 0.1%, but preferably be no more than approximately 10%, more preferably no more than about 5%, be most preferably not exceeding the linear alkyl benzene sulfonate tensio-active agent of about 1% business-like C10 to C14;
● detergent composition, contain composition weight at least about 0.1%, but preferably be no more than approximately 10%,, be most preferably not exceeding about 1% business-like highly branched alkyl benzene sulfonate surfactant (as TPBS or tetrapropyl benzene sulfonate) more preferably no more than about 5%;
● detergent composition, about 0.5% to about 25% the nonionogenic tenside that contains composition weight, wherein said nonionogenic tenside is end-blocking or not end capped polyalkoxylated alcohol, have :-hydrophobic grouping, be selected from the alkyl of 10 to 16 carbon atoms of the alkyl of linear 10 to 16 carbon atoms, the alkyl that there is 10 to 16 carbon atoms of 1 to 3 carbon atom side chain at the chain middle part, Guerbet branching, and composition thereof; With-hydrophilic radical, be selected from end-blocking or not end capped 1-15 ethoxylate, 1-15 propoxylated glycerine, 1-15 butoxy thing, and composition thereof.(when end-blocking not, also have terminal one-level-OH fragment, when end-blocking, also have the fragment of end-OR group form, wherein R is the alkyl fragment of 1 to 6 carbon atom, can randomly contain primary alcohol, or preferably comprises secondary alcohol when existing.);
● detergent composition, about 0.5% to about 25% the alkyl sulfate surfactant that contains composition weight, the hydrophobic grouping that has in the wherein said alkyl sulfate surfactant are selected from the alkyl group and composition thereof of 10 to 18 carbon atoms of the alkyl group of linear 10 to 18 carbon atoms, the alkyl group that there is 10 to 18 carbon atoms of 1 to 3 carbon atom side chain at the chain middle part, Guerbet branching; The positively charged ion that has is selected from Na, K and composition thereof;
● detergent composition, about 0.5% to about 25% alkyl (many alkoxyl groups) the vitriol nonionogenic tenside that contains composition weight, have in wherein said alkyl (many alkoxyl groups) sulfate surfactant :-hydrophobic grouping, be selected from the alkyl group of linear 10 to 16 carbon atoms, there is the alkyl group of 10 to 16 carbon atoms of 1 to 3 carbon atom side chain at the chain middle part, alkyl group of 10 to 16 carbon atoms of Guerbet branching and composition thereof, with-(many alkoxyl groups) vitriol hydrophilic radical, be selected from the end-blocking or the 1-15 polyethoxye vitriol of end-blocking form not, 1-15 gathers propoxy-vitriol, 1-15 gathers butoxy vitriol, 1-15 mix poly-(oxyethyl group/propoxy-/butoxy) vitriol and composition thereof and-be selected from Na, the positively charged ion of K and composition thereof.
Further, the detergent composition that the present invention includes contains following component (preferably being made up of following component basically): (i) about 0.01% of composition weight to about 95% (preferred about 0.5% to about 50%, more preferably from about 1%, preferably at least 2%, more preferably at least 4%, more more preferably at least 6%, most preferably at least 8% to about 35%) alkylbenzenesulfonatsurfactants surfactants mixture of the present invention; (ii) about 0.00001% of composition weight to the craft of the routine of about 99.9% (preferred about 5% to about 98%, more preferably from about 50% to the about 95%) auxiliary agent that washes the dishes; (iii) about 0.00001% of composition weight to about 99.9% (preferred about 0.1% to about 50%, more preferably from about 0.2% to about 40%, most preferably from about 0.5% to about 30%) tensio-active agent, this tensio-active agent are selected from anion surfactant except alkylbenzenesulfonatsurfactants surfactants mixture of the present invention, nonionic surface active agent, cats product, amphoterics, zwitterionics and composition thereof; Supplementary condition are, during other alkylbenzene sulfonates outside containing alkylbenzenesulfonatsurfactants surfactants mixture of the present invention in the described detergent composition, the whole 2/3-phenyl index of the mixture of described alkylbenzenesulfonatsurfactants surfactants mixture and described other alkylbenzene sulfonates is about 275 to about 10,000 (preferred about 350 to about 1,200, more preferably from about 500 to about 700).
Like this, the present invention includes 2/3-phemyol surface active agent mixture, basically constitute by following component: surfactant system weight 1% (preferably at least about 5%, more preferably at least about 10%) to about 60% (in a kind of mode, preferably less than about 50%, be more preferably less than about 40%) first kind of alkyl benzene sulfonate surfactant, wherein said first kind of alkyl benzene sulfonate surfactant is the alkylbenzenesulfonatsurfactants surfactants mixture according to first embodiment; With surfactant system weight 40% (in a kind of mode, preferably at least about 50%, more preferably at least about 60%) to about 99% (preferably less than about 95%, be more preferably less than about 90%) second kind of alkyl benzene sulfonate surfactant, wherein said second kind of alkyl benzene sulfonate surfactant, it is the alkylbenzene sulfonate surfactant mixture except described alkylbenzenesulfonatsurfactants surfactants mixture according to first embodiment, and the 2/3-phenyl index range of wherein said second kind of alkyl benzene sulfonate surfactant is about 75 to about 160 (although any business-like linear class (LAS) or side chain class (ABS usually, TPBS) all can use, but typical described second kind of alkyl benzene sulfonate surfactant is business-like linear alkyl benzene sulfonate tensio-active agent with 10 to 14 carbon atoms, as the LAS of DETAL method or the LAS of HF method); Supplementary condition are, the 2/3-phenyl index of described medium 2/3-phemyol surface active agent mixture is about 160 to about 275 (preferred about 170 to about 265, more preferably from about 180 to about 255).(certainly, in principle of the present invention and scope, the blend for preparing alkylbenzenesulfonatsurfactants surfactants mixture of the present invention with any known business-like linearity or branch-alkylbenzene sulfonate tensio-active agent also is equivalent).
The technology for preparing medium 2/3-phemyol surface active agent mixture comprises the step that is selected from one of following steps: (i) described first kind of alkyl benzene sulfonate surfactant and described second kind of alkyl benzene sulfonate surfactant are mixed; (ii) the unsulfonated precursor of described first kind of alkyl benzene sulfonate surfactant and the unsulfonated precursor of described second kind of alkyl benzene sulfonate surfactant are mixed, then the sulfonation blend.
Preparation embodiment
Embodiment 14-methyl-4-nonyl alcohol, 5-methyl-5-decyl alcohol, the mixture (starting raw material of branched olefin) of 6-methyl-6-hendecanol and 6-methyl-6-dodecanol
In feed hopper, add 4.65g 2 pentanone, 20.7g methyl-n-butyl ketone, 51.0g 2-heptanone, the mixture of 36.7g methyln-hexyl ketone and 72.6g ether.Use 2.25h then, alcohol/ketone mixtures is added drop-wise in the three mouthfuls of round-bottomed flasks of 2L under the stirring of nitrogen protection, this round-bottomed flask is equipped with reflux exchanger, and contains the diethyl ether solution of n-pentyl bromination magnesium of 600mL 2.0M and the 400mL ether that adds in addition.After being added dropwise to complete, this reaction mixture is at 20 ℃ of restir 2.5h.Then under agitation, this reaction mixture is joined in the 1kg trash ice.In this mixture, add 393.3g 30% sulphuric acid soln.Emit aqueous acid layer, 750mL water washing 2 time of remaining ether layer.Vaporising under vacuum ether layer obtains 176.1g 4-methyl-4-nonyl alcohol then, 5-methyl-5-decyl alcohol, the mixture of 6-methyl-6-hendecanol and 6-methyl-6-dodecanol.
Embodiment 2 is randomly branched is the alkene mixture (a kind of branched olefin mixture that is used to prepare the alkylating reagent of modified alkylbenzene of the present invention) of monomethyl branching basically
A) the monomethyl branching alcohol blend sample of 174.9g embodiment 1 is joined in three mouthfuls of round-bottomed flasks of 500mL under the stirring of nitrogen protection, this round-bottomed flask is equipped with zeolite catalyst (the acidic mordenite catalyst Zeocat that Dean-Stark trap and reflux exchanger and 35.8g are selected shape
TMFM-8/25H).Mix, then this mixture heating up is arrived about 110-155 ℃, in 4-5h, in Dean-Stark trap, collect water and some alkene.The alcohol mixture of having finished embodiment 1 now to non-random be the conversion of the alkene mixture of methyl-branched basically.With remaining in non-random in the flask is the alkene mixture of methyl-branched basically, is that the alkene mixture of methyl-branched merges basically with being collected in non-random in the Dean-Stark trap, and filters to remove catalyzer.Twice of 100mL hexane wash of solid filter cake.Vaporising under vacuum hexane filtrate, the product that obtains and primary filtrate merge, obtain 148.2g non-random be the alkene mixture of methyl-branched basically.
B) with alkene mixture and 36g shape-selective zeolite catalysts (the acidic mordenite catalyst Zeocat of embodiment 2a
TMFM-8/25H) mix, and react according to the embodiment 2a that has made following change.Temperature of reaction is brought up to 190-200 ℃, react about 1-2h, make branch position randomization specific in the alkene mixture.With remaining in randomly branched in the flask is the alkene mixture of monomethyl branching basically, is that the alkene mixture of monomethyl branching merges basically with being collected in randomly branched in the Dean-Stark trap, and filters to remove catalyzer.Twice of 100mL hexane wash of solid filter cake.Vaporising under vacuum hexane filtrate, the product that obtains and primary filtrate merge, obtain 147.5g randomly branched be the alkene mixture of monomethyl branching basically.
Embodiment 32/3-phenyl index be about 550 and 2-methyl-2-phenyl index be about 0.02 be the alkylbenzene mixture (according to modified alkylbenzene mixture of the present invention) of monomethyl branching basically
With 147g embodiment 2 is the alkene mixture and 36g shape-selective zeolite catalysts (the acidic mordenite catalyst Zeocat of monomethyl branching basically
TMFM-8/25H) join in 2 gallons of stainless steel autoclaves under the stirring.Alkene and catalyzer remaining in container enter in the autoclave with the washing of 300mL normal hexane, and the sealing autoclave.From the outside of autoclave chamber, in autoclave, add 2000g benzene (be contained in the isolated container, and join in the isolated autoclave chamber) with the separate pump system.Autoclave 250psig N
2Clean 2 times, charge into 60psig N then
2Stir this mixture, and at about 200 ℃ of heating 4-5h.Autoclave is cooled to about 20 ℃ to spend the night.Open the valve that autoclave leads to benzene condenser and receiving tank.Autoclave is heated to about 120 ℃, collects benzene continuously.When temperature of reactor reaches 120 ℃, no longer collect benzene.Then with reactor cooling to 40 ℃, and in autoclave, pump into the 750g normal hexane and mix.From autoclave, emit reaction mixture then.Filter this reaction mixture removing catalyzer, and under vacuumizing, remove normal hexane.This product is then in vacuum (1-5mmHg) distillation down.Collect 76 ℃-130 ℃ cut, obtain 2/3-phenyl index be about 550 and 2-methyl-2-phenyl index be about 0.02 be the alkylbenzene mixture (167g) of monomethyl branching basically.
Embodiment 42/3-phenyl index be about 550 and 2-methyl-2-phenyl index be about 0.02 be the benzene sulfonamide acid mixture (according to modified alkylbenzene azochlorosulfonate acid mixture of the present invention) of monomethyl branching basically
Use methylene dichloride as solvent, the product of embodiment 3 is carried out sulfonation with equimolar chlorsulfonic acid.Remove methylene dichloride, obtain 210g 2/3-phenyl index be about 550 and 2-methyl-2-phenyl index be about 0.02 be the benzene sulfonamide acid mixture of monomethyl branching basically.
Embodiment 52/3-phenyl index be about 550 be the alkyl benzene sulphonate (ABS) sodium salt mixt (according to alkylbenzenesulfonatsurfactants surfactants mixture of the present invention) of monomethyl branching basically
With in the methanol solution of equimolar sodium methylate and the product of embodiment 4, methyl alcohol is removed in evaporation, obtain 225g 2/3-phenyl index be about 550 and 2-methyl-2-phenyl index be about 0.02 be the alkyl benzene sulphonate (ABS) sodium salt mixt of monomethyl branching basically.
Embodiment 62/3-phenyl index be about 550 and 2-methyl-2-phenyl index be about 0.02 be linear alkylbenzene mixture (as the alkylbenzene mixture of modified alkylbenzene component) basically
Use shape-selective zeolite catalysts (acidic mordenite catalyst Zeocat
TMFM-8/25H), preparation 2/3-phenyl index be about 550 and 2-methyl-2-phenyl index to be about 0.02 chain length be linear alkylbenzene mixture basically.To 70g shape-selective zeolite catalysts (acidic mordenite catalyst Zeocat is housed
TMFM-8/25H) in 2 gallons of stainless steel autoclaves under the stirring, add 15.1g Neodene (R) 10,136.6g Neodene (R) 1112,89.5g Neodene (R) 12 and 109.1g 1-tridecylene.Neodene is the trade(brand)name of the alkene of Shell Chemical Company.Alkene and catalyzer remaining in container enter in the autoclave with the washing of 200mL normal hexane, and the sealing autoclave.From the outside of autoclave chamber, in autoclave, add 2500g benzene (be contained in the isolated container, and join in the isolated autoclave chamber) with the separate pump system.Autoclave 250psig N
2Clean 2 times, be inflated to 60psig N then
2Stir this mixture, and, be cooled to 70~80 ℃ then at about 200 ℃~205 ℃ about 4~5h of heating.Open the valve that autoclave leads to benzene condenser and receiving tank.Autoclave is heated to about 120 ℃, collects benzene in receiving tank continuously.When temperature of reactor reaches 120 ℃, no longer collect benzene.Then with reactor cooling to 40 ℃, and in autoclave, pump into the 1kg normal hexane and mix.From autoclave, emit reaction mixture then.Filter this reaction mixture removing catalyzer, and under rough vacuum, evaporate normal hexane.This product is then in high vacuum (1-5mmHg) distillation down.Collect 85 ℃~150 ℃ cut, obtain 2/3-phenyl index be about 550 and 2-methyl-2-phenyl index be about 0.02 be linear alkylbenzene mixture (426.2g) basically.
Embodiment 72/3-phenyl index be about 550 and 2-methyl-2-phenyl index be about 0.02 be linear benzene sulfonamide acid mixture (as benzene sulfonamide acid mixture) basically according to modified alkylbenzene sulfonic acid component of the present invention
Use methylene dichloride as solvent, with the 422.45g product of equimolar chlorsulfonic acid sulfonation embodiment 6.Remove methylene dichloride, obtain 574g 2/3-phenyl index be about 550 and 2-methyl-2-phenyl index be about 0.02 be linear benzene sulfonamide acid mixture basically.
Embodiment 82/3-phenyl index be about 550 and 2-methyl-2-phenyl index be about 0.02 be linear alkyl benzene sulphonate (ABS) sodium salt mixt (as alkylbenzene sulfonate surfactant mixture) basically according to alkylbenzenesulfonatsurfactants surfactants component of mixture of the present invention
With in the methanol solution of equimolar sodium methylate and embodiment 7 be linear benzene sulfonamide acid mixture basically, evaporation methyl alcohol, obtain 613g 2/3-phenyl index be about 550 and 2-methyl-2-phenyl index be about 0.02 be linear alkyl benzene sulphonate (ABS) sodium salt mixt basically.
Embodiment 96,10-dimethyl-2-hendecanol (starting raw material of branched olefin)
In the glass autoclave inner bag, add 299g geranyl acetone, ruthenium-carbon of 3.8g 5% and 150ml methyl alcohol.Glass inner bag is sealed in the 3L stainless steel autoclave inside of shaking, autoclave 250psigN
2, 250psig H
2Each cleaning once charges into 1000psig H then
2Mix the reacting by heating mixture.At about 75 ℃, this reaction begins and begins to consume H
2, heat release makes temperature reach 170~180 ℃.In 10~15 minutes, temperature drops to 100~110 ℃, and pressure drops to 500psig.Autoclave H
2Be pressurized to 1000psig, and 100~110 ℃ of remix 1 hour 40 minutes, reaction consumed 160psig H again
2, but do not observe H this moment
2Consumption.Autoclave is cooled to 40 ℃, takes out reaction mixture, remove by filter catalyzer, and concentrate, obtain 297.75g 6,10-dimethyl-2-hendecanol by vaporising under vacuum methyl alcohol.
Embodiment 105,7-dimethyl-2-decyl alcohol (starting raw material of branched olefin)
Add 249g 5,7-dimethyl-3,5,9-triolefin in the last of the ten Heavenly stems-2-ketone, 2.2g 5% ruthenium-carbon and 200ml methyl alcohol to the glass autoclave inner bag.Glass inner bag is sealed in the 3L stainless steel autoclave inside of shaking, autoclave 250psig N
2, 250psig H
2Each cleaning once charges into 500psigH then
2Mix the reacting by heating mixture.At about 75 ℃, this reaction begins and begins to consume H
2, heat release makes temperature reach 170 ℃.In 10 minutes, temperature drops to 115~120 ℃, and pressure drops to 270psig.Autoclave H
2Be pressurized to 1000psig, and, be cooled to 30 ℃ then 110~115 ℃ of remix 7 hours 15 minutes.From autoclave, take out reaction mixture, remove by filter catalyzer, and concentrate, obtain 225.8g 5,7-dimethyl-2-decyl alcohol by vaporising under vacuum methyl alcohol.
Embodiment 114,8-dimethyl-2-nonyl alcohol (starting raw material of branched olefin)
The mixture that in feed hopper, adds 671.2g citral and 185.6g ether.Use 5h then, the citral mixture is added drop-wise in the three mouthfuls of round-bottomed flasks of 5L under the stirring of nitrogen protection, this round-bottomed flask is equipped with reflux exchanger, and contains 1.6L 3.0M methyl-magnesium-bromide solution and other 740ml ether.This reaction flask is placed ice-water bath, and the ether that causes with control heat release and heat release refluxes.After being added dropwise to complete, remove ice-water bath, and with reactant at 20~25 ℃ of remix 2h, under fine stirring, reaction mixture is joined in the 3.5Kg trash ice then.The sulphuric acid soln that in this mixture, adds 1570g 30%.Emit the aqueous acids layer, residual ether layer is washed twice with 2L.By the vaporising under vacuum ether ether layer is concentrated, obtain 720.6g 4,8-dimethyl-3,7-nonadiene-2-alcohol.In the glass autoclave inner bag, add 249.8g 4,8-dimethyl-3,7-nonadiene-2-alcohol, 5.8g 5% palladium-gac and 200ml normal hexane.Glass inner bag is sealed in the 3L stainless steel autoclave inside of shaking, autoclave 250psig N
2Cleaning twice, 250psig H
2Cleaning once charges into 100psig H then
2Firm hybrid reaction promptly begins, and begins to consume H
2, heat release makes temperature reach 75 ℃.Autoclave is heated to 80 ℃, uses H
2Be pressurized to 500psig, mix 3h, be cooled to 30 ℃ then.From autoclave, take out reaction mixture, remove by filter catalyzer, and concentrate, obtain 242g 4,8-dimethyl-2-nonyl alcohol by the vaporising under vacuum normal hexane.
What embodiment 12 was randomly branched is the alkene mixture (a kind of branched olefin mixture, it is the alkylating reagent of preparation modified alkylbenzene of the present invention) of dimethyl-branched basically
To thermometer is housed; in three mouthfuls of round-bottomed flasks of 2L of the mechanical stirrer and the nitrogen protection of the Dean-Stark trap that reflux exchanger is housed; add 225g 4; 8-dimethyl-2-nonyl alcohol (embodiment 11); 450g 5; 7-dimethyl-2-decyl alcohol (embodiment 10), 225g 6,10-dimethyl-2-hendecanol (embodiment 9) and 180g shape-selective zeolite catalysts (acidic mordenite catalyst Zeocat
TMFM-8/25H).Mix, mixture heating up to suitable temperature (135~160 ℃), flows out water and some alkene, and is collected in the Dean-Stark trap with the moderate speed.After several hours, the speed of collecting water slows down, and temperature rises to 180~195 ℃, and reactant is remix 2~4h under this temperature.
Remain in the alkene mixture of the dimethyl-branched in the flask,, filter to remove catalyzer with the alkene mixture merging of the dimethyl-branched that distills.Catalyzer filter cake and 500ml hexane form slurry and vacuumizing filtration.The catalyzer filter cake washes twice with the 100ml hexane, and filtrate concentrates by the vaporising under vacuum hexane.The product and the primary filtrate that obtain are merged, obtain the alkene mixture of the randomly branched dimethyl-branched of 820g.
Embodiment 132/3-phenyl index be about 600 and 2-methyl-2-phenyl index be about 0.04 randomly branched be the alkylbenzene mixture (according to modified alkylbenzene mixture of the present invention) of dimethyl-branched basically
Dimethyl-branched alkene mixture and 160g shape-selective zeolite catalysts (acidic mordenite catalyst Zeocat with 820g embodiment 12
TMFM-8/25H) join in 2 gallons of stainless steel autoclaves under the stirring, and the sealing autoclave.Autoclave 80psig N
2Cleaning twice charges into 60psig N then
2From the outside of autoclave chamber, 3000g benzene (be contained in the isolated container, and join isolated autoclave inside by the separate pump system) is joined in the autoclave.Stir this mixture, and be heated to 205 ℃~about 210 ℃.Reaction continues about 10 minutes, at this moment takes a sample from product mixture.This sample filtering of getting in 10 minutes to be removing catalyzer, and vacuumizes to remove any remaining trace benzene.This sample is in vacuum (1~5mmHg) time distillation.The cut of collecting 90~140 ℃ promptly obtains randomly branched dimethyl-branched alkylbenzene mixture, and its 2/3-phenyl index is about 600, and 2-methyl-2-phenyl index is about 0.26.Continuation is cooled to autoclave about 30 ℃ then and spends the night at 205~about 210 ℃ of about 8h of reaction.Open the valve that autoclave leads to benzene condenser and receiving tank.Autoclave is heated to about 120 ℃, collects benzene continuously.When temperature of reactor reaches 120 ℃, no longer collect benzene, then with reactor cooling to 40 ℃.From autoclave, emit reaction mixture then.Filter this reaction mixture to remove catalyzer, vacuumize and from mixture, remove remaining trace benzene.Vacuum distilling product (1~5mmHg).Collect 90 ℃~140 ℃ cuts, obtain the alkylbenzene mixture of randomly branched dimethyl-branched, its 2/3-phenyl index is about 600, and 2-methyl-2-phenyl index is about 0.04.
What embodiment 14 was randomly branched is the benzene sulfonamide acid mixture of dimethyl-branched basically, and its 2/3-phenyl index is about 600, and 2-methyl-2-phenyl index is about 0.04 (according to modified alkylbenzene azochlorosulfonate acid mixture of the present invention)
Use methylene dichloride as solvent, with the alkylbenzene product of the dimethyl-branched of equimolar chlorsulfonic acid sulfonation embodiment 13, HCl emits as by product.Concentrate by the vaporising under vacuum methylene dichloride and to obtain the sulfonic acid product.What obtain is that the 2/3-phenyl index of the benzene sulfonamide acid mixture of dimethyl-branched is about 600 basically, and 2-methyl-2-phenyl index is about 0.04.
What embodiment 15 was randomly branched is the alkyl benzene sulphonate (ABS) sodium salt mixt of dimethyl-branched basically, and its 2/3-phenyl index is about 600, and 2-methyl-2-phenyl index is about 0.04 (according to alkylbenzenesulfonatsurfactants surfactants mixture of the present invention)
With in the methanol solution of equimolar sodium methylate and the benzene sulfonamide acid mixture of the dimethyl-branched of embodiment 14, evaporate methyl alcohol, obtain the alkyl benzene sulphonate (ABS) sodium salt mixt solid of randomly branched dimethyl-branched, its 2/3-phenyl index is about 600, and 2-methyl-2-phenyl index is about 0.04.
Embodiment 16 is according to alkylbenzenesulfonatsurfactants surfactants mixture of the present invention (medium/3-phenyl index)
Make blend by following material:
I) 2/3-phenyl index is about 550 alkylbenzenesulfonatsurfactants surfactants mixture of the present invention (according to embodiment 5)
II) 2/3-phenyl index is about 100 commercialization C
11.7In following table, per-cent is based on weight to the LINER ALKYL BENZENE SULPHONIC ACID SOFT 96 sodium salt surface active agent of (on average) (HF type):
A??????????????B??????????CI?????????25%???????????15%????????38%II????????75%???????????85%????????62%
Above the 2/3-phenyl index of each blend about 160 to about 275 scope.
Embodiment 17 alkylbenzenesulfonatsurfactants surfactants mixtures of the present invention (medium 2/3-phenyl index)
Make blend by following material:
I) 2/3-phenyl index is about 550 alkylbenzenesulfonatsurfactants surfactants mixture of the present invention (according to embodiment 5)
II) 2/3-phenyl index is about 150 commercialization C
11.7In following table, per-cent is based on weight to the LINER ALKYL BENZENE SULPHONIC ACID SOFT 96 sodium salt surface active agent of (on average) (DETAL type):
A?????????????B?????????????CI?????????25%??????????15%??????????10%II????????75%??????????85%??????????90%
Above the 2/3-phenyl index of each blend about 160 to about 275 scope.
Embodiment 18 modified alkylbenzene azochlorosulfonate acid mixtures of the present invention (medium 2/3-phenyl type)
Make blend by following material:
I) 2/3-phenyl index is about 550 modified alkylbenzene sulfonic acid surfactant mixture of the present invention (according to embodiment 4)
II)/3-phenyl index is about 100 commercialization C
11.7The LINER ALKYL BENZENE SULPHONIC ACID SOFT 96 of (on average) (HF type).In following table, per-cent is based on weight:
A????????????B?????????????CI??????????25%?????????15%??????????38%II?????????75%?????????85%??????????62%
Above the 2/3-phenyl index of each blend about 160 to about 275 scope.
Embodiment 19 modified alkylbenzene azochlorosulfonate acid mixtures of the present invention (medium 2/3-phenyl type)
Make blend by following material:
I) 2/3-phenyl index is about 550 modified alkylbenzene azochlorosulfonate acid mixture of the present invention (according to embodiment 4)
II) 2/3-phenyl index is about 150 commercialization C
11.7The LINER ALKYL BENZENE SULPHONIC ACID SOFT 96 of (on average) (DETAL type).In following table, per-cent is based on weight:
A?????????????B????????????CI??????????25%?????????15%?????????10%II?????????75%?????????85%?????????90%
Above the 2/3-phenyl index of each blend about 160 to about 275 scope.
Embodiment 20 modified alkylbenzene mixtures of the present invention (medium 2/3-phenyl type)
Make blend by following material:
I) 2/3-phenyl index is about 550 modified alkylbenzene mixture of the present invention (according to embodiment 3)
II) 2/3-phenyl index is about 100 commercialization C
11.7The LINEAR ALKYL BENZENE (LAB) of (on average) (HF type).In following table, per-cent is based on weight:
A??????????B????????????CI??????????25%????????15%????????38%II?????????75%????????85%????????62%
Above the 2/3-phenyl index of each blend about 160 to about 275 scope.
Embodiment 21 modified alkylbenzene mixtures of the present invention (medium 2/3-phenyl type)
Make blend by following material:
I) 2/3-phenyl index is about 550 modified alkylbenzene mixture of the present invention (according to embodiment 3)
II) 2/3-phenyl index is about 150 commercialization C
11.7The LINEAR ALKYL BENZENE (LAB) of (on average) (DETAL type).In following table, per-cent is based on weight:
A????????????B?????????????CI???????????25%????????15%??????????10%II??????????75%????????85%??????????90%
Above the 2/3-phenyl index of each blend about 160 to about 275 scope.
Embodiment 222/3-phenyl index be about 550 and 2-methyl-2-phenyl index be about 0.02 modified alkylbenzene mixture of the present invention
With 110.25g embodiment 2 is the alkene mixture of monomethyl branching basically, 18) and 36g shape-selective zeolite catalysts (acidic mordenite catalyst Zeocat 36.75g (decene: undecylene: dodecylene: the ratio of tridecylene is 2: 9: 20 to the alkene mixture of non-branching:
TMFM-8/25H), join in 2 gallons of stainless steel autoclaves under the stirring.Alkene and catalyzer remaining in container enter in the autoclave with the washing of 300mL normal hexane, and the sealing autoclave.From the outside of autoclave chamber, in autoclave, add 2000g benzene (be contained in the isolated container, and join in the isolated autoclave chamber) with the separate pump system.Autoclave 250psig N
2Clean 2 times, charge into 60psig N then
2Stir this mixture, and at about 200 ℃ of about 4~5h of heating.Autoclave is cooled to about 20 ℃ to spend the night.Open the valve that autoclave leads to benzene condenser and receiving tank.Autoclave is heated to about 120 ℃, collects benzene continuously.When temperature of reactor reaches 120 ℃, no longer collect benzene.Then with reactor cooling to 40 ℃, and in autoclave, pump into the 750g normal hexane and mix.From autoclave, emit reaction mixture then.Filter this reaction mixture removing catalyzer, and under vacuum, remove normal hexane.At vacuum (1~5mmHg) time this product of distillation.Collect 76 ℃~130 ℃ cut, obtain 2/3-phenyl index be about 550 and 2-methyl-2-phenyl index be about 0.02 modified alkylbenzene mixture (167g).
Embodiment 232/3-phenyl index be about 550 and 2-methyl-2-phenyl index be about 0.02 modified alkylbenzene azochlorosulfonate acid mixture of the present invention (branching and non-branching benzene sulfonamide acid mixture)
Use methylene dichloride as solvent, with the modified alkylbenzene mixture of equimolar chlorsulfonic acid sulfonation embodiment 22.Remove methylene dichloride, obtain 210g 2/3-phenyl index be about 550 and 2-methyl-2-phenyl index be about 0.02 modified alkylbenzene azochlorosulfonate acid mixture.
Embodiment 242/3-phenyl index be about 550 and 2-methyl-2-phenyl index be about 0.02 modified alkylbenzene sulfonate sodium mixture of the present invention (branching and non-branching alkyl benzene sulphonate (ABS) sodium salt mixt)
With in the methanol solution of equimolar sodium methylate and the modified alkylbenzene sulfonic acid of embodiment 23, evaporation methyl alcohol, obtain 225g 2/3-phenyl index be about 550 and 2-methyl-2-phenyl index be about 0.02 modified alkylbenzene sulfonate sodium mixture.Measure the method for the complex parameter (2/3-phenyl index, 2-methyl-2-phenyl index) of blended alkylbenzene/alkylbenzene sulfonate/benzene sulfonamide acid system
(TPBS, the method for complex parameter ABS) is well known in the art to measure conventional LINEAR ALKYL BENZENE (LAB) and/or high degree of branching alkylbenzene sulfonate.Referring to, Surfactant ScienceSeries for example, 40 volumes, the 7th chapter and Surfactant Science Series, 73 volumes, the 7th chapter.Typically, measure alkylbenzene, measure alkylbenzene sulfonate or sulfonic acid by HPLC by GC and/or GC-mass spectrum;
13C nmr is also more general.Another kind of method in common is to take off sulfonation.Allow like this to use GC and/or GC-mass spectrum, sulfonate or sulfonic acid are changed into the alkylbenzene that is easy to these method processing because take off sulfonation.
In a word, the invention provides unique and the alkylbenzene mixture of relative complex and complicated similarly alkylbenzene sulfonate and/or the surfactant mixture of alkyl benzene sulphonate (ABS).Can use the change example of means known in the art and the complex parameter that this based composition is measured in combination.
The order of the method for using depends on the composition that will characterize, and is specific as follows:
In material, contain and surpass about 10% impurity, as dialkyl benzene, alkene, paraffin, when helping water solvent, dialkyl benzene sulfonate etc., normally preferred.GC
Device: the gas-chromatography HP5890 of Hewlett-Packard series II, be equipped with shunting/presumptuously flow injection head and FIDJ﹠amp; W Scientific capillary column DB-1HT, 30 meters, internal diameter 0.25mm, 0.1um film thickness, the red less salt partition of catalog number (Cat.No.) 1221131Restek 11mm, the gooseneck inlet cannula of catalog number (Cat.No.) 22306Restek 4mm carbonization glass, catalog number (Cat.No.) 20799-209.5 inlet liner O-type ring, the catalog number (Cat.No.) 5180-4182J.T.Baker HPLC of Hewlett-Packard level methylene dichloride, catalog number (Cat.No.) 9315-33, or the automatic sampling bottle of Equivalent 2ml GC band elbow, or Equivalent specimen preparation: weighing 4-5mg sample, the 2ml GC automatic sampling bottle of packing into adds 1ml J.T.Baker HPLC level methylene dichloride in the GC bottle, catalog number (Cat.No.) 9315-33 is with the sealing member (bottle cap) of the inner liner polytetrafluoroethylene of 11mm band elbow bottle, Part No. HP5181-1210, use the instrument of curling, Part No. HP8710-0979, sealing and fine mixing.Now sample is ready to, can injects and enter the GCGC parameter: carrier gas: hydrogen column head pressure: 9psi flow velocity: column flow rate.
| The composition that is characterized | Testing method order (with carrying out in order that comma separates, other can parallelly carry out between method) |
| Alkylbenzene mixture | GC,NMR1?NMR2 |
| The alkylbenzene mixture of band impurity * | GC,DIS,GC,NMR1?NMR2 |
| The benzene sulfonamide acid mixture | Select 1:HPLC, NMR3 NMR4 selects 2:HPLC, DE, NMR1 NMR2 |
| The alkyl benzene sulphonate (ABS) salt mixture | Select 1:HPLC, AC, NMR3 NMR4 selects 2:HPLC, DE, NMR1 NMR2 |
| The benzene sulfonamide acid mixture of band impurity | Select 1:HPLC, HPLC-P, HPLC, NMR3 NMR4 selects 2:HPLC, DE, DIS, GC, NMR1 NMR2 |
| The alkyl benzene sulphonate (ABS) salt mixture of band impurity | Select 1:HPLC, HPLC-P, HPLC, AC, NMR3 NMR4 selects 2:HPLC, DE, DIS, GC, NMR1 NMR2 |
Diffluence pass @~3ml/min.
Barrier film cleaning @1ml/min.Injection: HP 7673 self-actuated samplers, 10ul syringe, 1ul injection liquid injection head temperature: 350 ℃ of detector temperatures: 400 ℃ of program oven temperatures: keep 1min for initial 70 ℃.
1 ℃/min of speed.
Keep 10min for last 180 ℃.
The standard of this method is 2-phenyl octane and 2-phenyl pentadecane, and each all is new distillatory, and purity surpasses 98%.The condition that use describes in detail is above moved this two standards, to determine the retention time of each standard.The retention time scope of Que Dinging is the retention time scope that is used to characterize interior any alkylbenzene of the scope of the invention or alkylbenzene mixture (for example specimen) like this.Operation now needs to measure the specimen of complex parameter.If the GC area in the retention time scope that two standards are determined, thinks then that specimen tests by GC greater than 90% of total GC area.Specimen by the GC test can directly be used on NMR1 and the NMR2 test method., must not carry out purifying by distillation again, test by GC up to specimen by the specimen of GC test.
Take off sulfonation (DE)
Taking off sulfonation method is a kind of standard method, and " washing composition and cleaning product analysis " book 197-199 page or leaf outstanding at G.F.Longman is described in detail.230-231 page or leaf " tensio-active agent analysis " and John Cross compile in other two the 40th volumes of compiling about the article of this standard method such as T.M.Schmitt " tensio-active agent science book series " the 73rd rolls up " anion surfactant " in 272 pages in " tensio-active agent science book series ".This method disclosed herein is a kind of replacement method of HPLC method, can be used to estimate branching and non-branching alkyl benzene sulphonate (ABS) and/or salt mixture (modified alkylbenzene sulfonic acid and/or salt mixture).This method provides the mode that sulfonic acid and/or salt mixture is changed into the alkylbenzene mixture of branching and non-branching, and the latter can analyze with GC disclosed herein and NMR method NMR1 and NMR2.
HPLC
Consult L.R.Snyder and J.J.Kirkland, " modern liquid chromatography introduction ", second edition, Wiley, NY, 1979.
Instrument Appropriate H PLC systemWaters Division of Millipore, or Equivalent HPLC He squirt pump and temperature control are Waters, 600 types or Equivalent system self-actuated sampler/syringe Waters 717 or Equivalent self-actuated sampler 48 feed pallets Waters or Equivalent UV detector Waters PDA 996 or Equivalent fluorimetric detector Waters 740 or Equivalent data system/integrator Waters 860 or Equivalent self-actuated sampler bottle and bottle cap 4mL volume, Millipore#78514 and #78515HPLC post, X2 Supelcosil LC18,5 μ m, 4.6mm*25cm
Supelcosil#58298 column inlet strainer Rheodyne 0.5um * 3mm
Rheodyne#7335LC elutriant film filter Millipore SJHV M47 10 uses
0.45 the disposable filter funnel balance Sartorius or the Equivalent of μ m film; Tolerance range=±
0.0001g the sample purification bag of vacuum band pump and strainer,
Waters#WAT085113
ReagentC8 LAS reference material is right-and 2-octyl group benzene sulfonic acid sodium salt C15 LAS reference material is right-2-pentadecyl benzene sulfonic acid sodium salt
ProgramA.
The preparation of HPLC moving phase
1. mobile phase A
A) weighing 11.690g sodium-chlor, and transfer in the 2000mL volumetric flask. exists
Dissolve in the 200mL hplc grade water.
B) add 800mL acetonitrile and mixing.Solution is diluted to appearance after arriving room temperature
Long-pending.Configuration obtains 100mM NaCl/40%ACN solution like this.
C) filter by LC elutriant film filter, and the degassing before use.
2. 60% acetonitrile solution of Mobile phase B-preparation 2000mL in hplc grade water.Logical
Cross LC elutriant film filter and filter, and the degassing before use.B.C8 and C15 inner mark solution
1. weighing 0.050g 2-phenyl octyl group benzene sulfonate and 0.050g 2-phenyl pentadecane sulfonic acid
The salt standard substance, and quantitatively transfer in the 100mL volumetric flask.
2. be diluted to volume with 30mL ACN dissolving, and with hplc grade water.Configuration like this
Obtain the blended standard substance solution of about 1500ppm.C. sample solution
1.
Rinse solution-250 μ L standardized solution are transferred to 1mL self-actuated sampler bottle
In, and add 750 μ L rinse solutions.Covering bottle cap is placed in the self-actuated sampler dish.
2.
Alkyl benzene sulphonate (ABS) or alkylbenzene sulfonate-weighing 0.10g alkyl benzene sulphonate (ABS) or salt,
And quantitatively be transferred in the 100mL volumetric flask.With 30mL ACN dissolving, and use
Hplc grade water is diluted to volume.It is automatic that 250 μ L standardized solution are transferred to 1mL
In the sampler bottle, and add 750 μ L sample solutions.Covering bottle cap is placed on automatically
In the sampler dish.If solution is very muddy, transferring to the self-actuated sampler bottle
Before, the film by 0.45 μ m filters.Cover bottle cap and be placed on self-actuated sampler
In the dish.The D.HPLC system
1. with moving phase perfusion HPLC pump.Erection column and column inlet strainer, and use wash-out
Liquid carries out balance (0.3mL/min, 1h at least).
2. use following HPLC condition operation sample:
Mobile phase A 100mM NaCl/40%ACN
Mobile phase B 40%H
2O/60%ACN
Time 0min 100% mobile phase A 0% Mobile phase B
Time 75min 5% mobile phase A 95% Mobile phase B
Time 98min 5% mobile phase A 95% Mobile phase B
Time 110min 100% mobile phase A 0% Mobile phase B
Time 120min 100% mobile phase A 0% Mobile phase B
Annotate:, may need 5-10 minute gradient time of lag according to the difference of HPLC system dead volume.
Flow velocity 1.2mL/min
25 ℃ of temperature
He blasts speed 50mL/hr
UV detector 225nm
Fluorimetric detector λ=225nm, λ=295nm,
Sensitivity is amplified 10 times
Working time 120min
Volume injected 10 μ L
Duplicate injection 2
Data speed 0.45MB/Hr
Resolving power 4.8nm
3. pillar should then with the flushing of 100% acetonitrile, and be preserved earlier with the flushing of 100% water
In 80/20 ACN/ water.
For alkyl benzene sulphonate (ABS)/salt mixture of the present invention, the HPLC elution time of 2-phenyl octyl group benzene sulfonate has defined the lower limit that HPLC analyzes, and the elution time of 2-phenyl pentadecane sulfonic acid standard substance has defined the upper limit that HPLC analyzes.If the retention time of 90% alkyl benzene sulphonate (ABS)/salt mixture component is in the scope of above-mentioned standard substance, this sample can further be used NMR 3 and NMR4 methods analyst so.
If have in alkyl benzene sulphonate (ABS)/salt mixture 10% or more multi-component retention time not in retention time scope with standard substance definition, this mixture should be used the HPLC-P method so, or uses DE, the DIS method is further purified.
Preparation property HPLC (HPLC-P)
The alkyl benzene sulphonate (ABS) and/or the salt preparation property HPLC purifying that contain a large amount of impurity (10% or more).Related content is consulted L.R.Snyder and J.J.Kirkland, " modern liquid chromatography introduction ", the 2nd edition, Wiley press, New York, 1979.This is ordinary method to those skilled in the art.Should the enough amounts of purifying, to satisfy the requirement of NMR 3 and NMR 4.
Use the preparation LC method of Mega Bond EIut Seu Pak@ (HPLC-P)
Also can adopt LC method (being also referred to as HPLC-P herein) purifying to contain the alkyl benzene sulphonate (ABS) and/or the salt of a large amount of impurity (10% or more).This method in fact than preparation property HPLC column purification more preferably.
Nearly the unpurified MLAS salt of 500mg can load to 10g (60ml) Mega Bond ElutSep Pak
On, under the chromatographic condition of the best, the MLAS salt of purifying can be isolated, and in 2h, lyophilize can be ready for.100mg modified alkylbenzene sulfonates sample can load on 5g (20ml) the Bond Elut Sep Pak, and can be ready in the same time.A. instrument HPLC:Waters 600E type gradient pump, 717 type self-actuated samplers, Water ' s Millennium PDA, Millenium data management system (v.2.15) Mega Bond Elut:C18 is in conjunction with phase, Varian 5g or 10g have the PN:1225-6023 of adapter, 1225-6031HPLC post: Supelcosil LC-18 (X2), 250 * 4.6mm, 5mm; #58298 analytical balance: Mettler AE240 type, can arrive ± 0.01mgB. accessory capabilities bottle by the weighing sample: glass, 10mL graduated cylinder: 1LHPLC self-actuated sampler bottle: 4mL vial, have tetrafluoroethylene bottle cap and glass lower volume inset and can accurately pipette 1,2 and the transfer pipet C. reagent and the chemical water (DI-H of 5mL capacity
2O): derive from Millipore, the deionized-distilled water of Milli-Q system or equivalent acetonitrile (CH
3CN): derive from the HPLC level product of Baker, or the equivalent sodium chloride crystal: the preparation of Baker AG or equivalent D.HPLC condition water:
A: the 600mL DI-H in being contained in the 1L graduated cylinder
2Add 5.845 sodium-chlor among the O.
Thorough mixing also adds 400ml ACN.Thorough mixing.
B: the 400mL DI-H in being contained in the 1L graduated cylinder
2Add 600ml ACN also among the O
Thorough mixing.
Storage tank A:60/40, H
2O/CAN and salt, storage tank B:40/60, H
2O/ACN operational conditions: gradient: 100%A continues to 75 minutes, and 5%A/95%B continues to 98 minutes, and 5%A/95%B continues to 110 minutes, and 100%A continues to 125 minutes.Column temperature without constant temperature (promptly, room temperature) HPLC flow velocity 1.2mL/min volume injected 10mL working time 125 minutes UV detectors 225nm concentration>4mg/mLSEP PAK balance (BOND ELUT, 5G)
1. use the 10cc syringe to apply malleation,, make 10ml contain 25/75H with~40 droplets/minute speed
2The solution of O/ACN does not allow sep pak do by sep pak.
2. according to the mode identical, make 10ml (x3) contain 70/30 H at once with #1
2The solution of O/ACN passes through.Do not allow sep pak do.Keep the horizontal plane of solution at the top of sep pak. (~1mm).
3.sep pak is ready to add sample now.
Filling/separation of MLAS sample and segregation
4. weighing<200mg sample enters in the 1 dram bottle, and adds 2ml 70/30 H
2O/ACN.With sonic treatment and thorough mixing.
5. sample is installed among the Bond Elut, and apply malleation with the 10cc syringe and begin to separate.With 1ml (x2) 70/30 solution flushing bottle, and be added among the sep pak.Solution at the top of sep pak maintenance~1mm.
6. use the 10cc syringe to apply malleation,, make 10ml 70/30 solution by Bond Elut with~40 droplets/minute speed.
7., and collect effluent if to the interested words of impurity, repeat this process with 3ml and 4ml.MLAS separates and collects
1. use the 10cc syringe to apply malleation, make 10ml contain 25/75 H
2The solution of O/ACN passes through, and collects effluent.Repeat this process with other 10ml, and repeat once again with 5ml.The MLAS that separates carries out lyophilize, then characterizes.
2. rotary evaporation is till removing ACN, and the H of lyophilize remnants
2O.Sample is ready to now, can carry out chromatogram.
Attention: when being used in combination Mega Bond Elut Sep Pak (10g type), the 500mg sample of can packing in seppak at most, and the volume by regulator solution can be ready to effluent and carry out lyophilize in 2h.SEP PAK balance (BOND ELUT, 10G)
1. use the laboratory air or regulate the interior air of cylinder,, make 20ml contain 25/75 H with about 40 droplets/minute speed
2The solution of O/ACN is by sep pak.Can not use the malleation that produces by syringe, because it is not enough to promote solution by sep pak.Do not allow sep pak drain off.
2. according to the mode identical, make 20ml (x2) and other 10ml contain 70/30H at once with #1
2The solution of O/ACN passes through.Do not allow sep pak drain off.Keep the horizontal plane of solution at the top of seppak (~1mm).
3. this sep pak is ready to now, the sample of can packing into.The MLAS sample is packed into/is separated and emanates
1. weighing<500mg sample enters in the 2 dram bottles, and adds 5ml 70/30 H
2O/ACN.With sonic treatment and thorough mixing.
2. the sample of in Bond Elut, packing into, and apply malleation with air source and begin to separate.With 70/30 solution of 2ml (x2) flushing bottle and add among the sep pak.The solution that keeps about 1mm at the top of sep pak.
3. apply malleation with air source,, make 20ml 70/30 solution pass through BondElut with about 40 droplets/minute speed.If interested, repeat this process with 6ml and 8ml, and collect effluent impurity.MLAS separates and collects
1. apply malleation with air source, make 20ml contain 25/75 H
2The solution of O/ACN passes through, and collects effluent.
2. repeat this process with other 20ml, and repeat with 10ml again.Separate the component that obtains and contain purified MLAS.
3. separate the MLAS that obtains and be ready to now, can carry out lyophilize, then characterize.
4. rotary evaporation is till removing ACN, and the H of lyophilize remnants
2O.Sample is ready to now, can carry out chromatogram.Attention: need to regulate the concentration of organic modifiers, with optimized Separation and segregation.
Distillation (DIS)
In three mouthfuls of round-bottomed flasks of the 5L that has 24/40 interface, load onto the mechanical stirring rod.In flask, add several effervescent tablets (Hengar Granules, catalog number (Cat.No.) 136-C).The 91/2 inch long Wei Geluo condenser that has 24/40 interface is connected on the flask intermediary mouth.The Wei Geluo condenser is equipped with the thermometer through calibration, and its top is connected to water condenser.Vacuum-resistant receives the end that flask is connected on condenser.A glass stopper beyond the Great Wall on the arm of 5L flask one side is loaded onto thermometer through calibration at opposite side.With flask and Wei Geluo condenser with on the Aluminium Foil Package.Add the 2270g alkylbenzene mixture in the 5L flask, this mixture is measured with the GC method, contains 10% or more impurity.The valve tube of deriving from vacuum pump is connected on the receiving bottle.Be stirred in the alkylbenzene mixture in the 5L flask, and this system is vacuumized.In case reach maximum vacuum (scale is at least 1 inch Hg pressure or lower), the electricity consumption heating jacket heats alkylbenzene mixture.Collect the distillment of two kinds of cuts.Fraction A is collected from about 25 ℃~about 90 ℃, and described temperature is to measure with the thermometer through calibration of Wei Geluo column top.Fraction B is from about 90 ℃~about 155 ℃ of collections, and described temperature is to measure with the thermometer through calibration of Wei Geluo column top.Fraction A and bottle end residue (high boiling point) discard.Fraction B (1881g) contains interested alkylbenzene mixture.This method is according to professional's needs, can scaled down, and supplementary condition are alkylbenzene mixtures of the remaining q.s in distillation back, so that estimate with NMR method NMR1 and NMR2.
Acidifying (AC)
Use usual manner, for example in solvent with HCl or sulfuric acid reaction, or and acidic resins, for example Amberlyst 15 reaction can be with the salt acidifying of alkyl benzene sulphonate (ABS).To those skilled in the art, acidifying is an ordinary method.Remove all solvents after the acidifying, particularly any moisture, sample is anhydrous like this, and does not contain solvent.Attention: for following all NMR testing method, the chemical displacement value CDCl of NMR spectrum
3, i.e. chloroform external standard.
NMR1Alkylbenzene mixture
13C-NMR 2/3-phenyl index
In the anhydrous deuterochloroform of 1ml, described deuterochloroform contains 1%v/v TMS as object of reference with 400mg alkylbenzene mixture sample dissolution, and this solution is contained in the standard N MR test tube.Should
13C NMR carries out on 300MHz NMR instrument, uses 20 seconds cycling time, 40 ℃
13The C pulse width, and go lotus root with the gate heteronuclear.At least write down 2000 scanning.To about 145.00ppm~about 150.00ppm's
13Integration is carried out in C NMR spectrogram zone.2/3-phenyl index with following equation definition alkylbenzene mixture.2/3-phenyl index=(integrated value of about 147.65ppm~about 148.05ppm)/(integration of about 145.70ppm~about 146.15ppm) * 100
NMR 2 13C-NMR 2-methyl-2-phenyl index
In the anhydrous deuterochloroform of 1ml, described deuterochloroform contains 1%v/v TMS as object of reference with the anhydrous alkylbenzene mixture sample dissolution of 400mg, and this solution is contained in the standard N MR test tube.Should
13C NMR carries out on 300MHz NMR instrument, uses 20 seconds cycling time, 40 ℃
13The C pulse width, and go lotus root with the gate heteronuclear.At least write down 2000 scanning.To about 145.00ppm~about 150.00ppm's
13Integration is carried out in C NMR spectrogram zone.2-methyl-2-phenyl index with following equation definition alkylbenzene mixture.2-methyl-2-phenyl index=(integration of about 149.35ppm~about 149.80ppm)/(integration of about 145.00ppm~about 150.00ppm)
NMR3The benzene sulfonamide acid mixture
13C-NMR 2/3-phenyl index
The anhydrous alkyl benzene sulphonate (ABS) blend sample of 400mg is dissolved in the anhydrous deuterochloroform of 1ml, and described deuterochloroform contains 1%v/v TMS as object of reference, and this solution is contained in the standard N MR test tube.Should
13C NMR carries out on 300MHz NMR instrument, uses 20 seconds cycling time, 40 ℃
13The C pulse width, and go lotus root with the gate heteronuclear.At least write down 2000 scanning.To about 152.50ppm~about 156.90ppm's
13Integration is carried out in C NMR spectrogram zone.2/3-phenyl index with following equation definition benzene sulfonamide acid mixture.2/3-phenyl index=(integration of about 154.40ppm~about 154.80ppm)/(integration of about 152.70ppm~about 153.15ppm) * 100
NMR 4The benzene sulfonamide acid mixture
13C-NMR 2-methyl-2-phenyl index
The anhydrous alkyl benzene sulphonate (ABS) blend sample of 400mg is dissolved in the anhydrous deuterochloroform of 1ml, and described deuterochloroform contains 1%v/v TMS as object of reference, and this solution is contained in the standard N MR test tube.Should
13C NMR carries out on 300MHz NMR instrument, uses 20 seconds cycling time, 40 ℃
13The C pulse width, and go lotus root with the gate heteronuclear.At least write down 2000 scanning.To about 152.50ppm~about 156.90ppm's
13Integration is carried out in C NMR spectrogram zone.2-methyl-2-phenyl index with following equation definition benzene sulfonamide acid mixture.2-methyl-2-phenyl index=(integration of about 156.40ppm~about 156.65ppm)/(integration of about 152.50ppm~about 156.90ppm)
Water base heavy duty liquid detergent Tensio-active agent
The present invention also comprises aqueous liquid detergent compositions.The waterborne liquid detergent composition preferably includes about 10~about 98% (weight), preferred about water liquid carrier of 30%~about 95% (weight), and this carrier is preferably water.In addition, waterborne liquid detergent composition of the present invention comprises surfactant system, and this system preferably contains one or more washing cosurfactants except that top disclosed branched surfactants.Described other cosurfactant can be selected from the nonionic detergent surfactant, the anionic detergent tensio-active agent, and zwitierionic detergent surfactant, the amine oxide detergent surfactant, and composition thereof.Described surfactant system comprises about 5%~about 70% (weight) usually, preferred about detergent composition of 15%~about 30% (weight).
Anion surfactant
Anion surfactant comprises C
11-C
18Alkylbenzene sulfonate (LAS); And one-level, branched chain and random C
10-C
20Alkyl-sulphate (AS); C
10-C
18Secondary (2,3) alkyl-sulphate, its general formula is CH
3(CH
2)
x(CHOSO
3 -M
+) CH
3And CH
3(CH
2)
y(CHOSO
3 -M
+) CH
2CH
3, wherein x and (y+1) be to be at least about 7 integer preferably is at least about 9, and M is water-soluble cationic, particularly sodium; Unsaturated vitriol, for example oleyl vitriol; C
10-C
18Alkyl alkoxy sulfate (" AExS "; EO 1-7 ethoxy sulfate particularly); C
10-C
18Alkyl alkoxy carboxylate salt (particularly EO 1-5 ethoxy carboxylate); C
10-C
18Glyceryl ether; C
10-C
18APG and corresponding polysaccharide glycosides vitriol thereof, and C
12-C
18α-sulfonated fatty acid ester.
Generally speaking, be disclosed in U.S. Patent No. 4,285,841 at this used anion surfactant, Barrat etc., August 25 in 1981 announced and U.S. Patent No. 3,919,678 that Laughlin etc. announced on December 30th, 1975.
Useful anion surfactant comprises the water-soluble salt of organic sulfating reaction product, basic metal particularly, ammonium and pure ammonium are (for example, monoethanolamine ammonium or triethanol ammonium) salt, have the alkyl and sulfonic acid or the sulfate group (term " alkyl " has comprised the moieties of aryl) that contain 10~about 20 carbon atoms of having an appointment in the molecular structure of described organosulfur acidification reaction product.This example that is combined into tensio-active agent is an alkyl-sulphate, and particularly those are by higher alcohols (C
8-C
18Carbon atom), the product that obtains of those pure sulfations that obtain by the reduction of Tallow, beef or Oleum Cocois for example.
At this, other anion surfactant is water miscible alkyl phenol epoxy ethane ether salt, and per molecule contains 1~about 4 the ethylene oxide unit of having an appointment in the described vitriol, and the alkyl of about 8~about 12 carbon atoms.
At this, other useful anion surfactant comprises the water-soluble salt of non-sulfonated fatty acid ester, and described non-alpha-sulfonated fatty acid contains the fatty acid-based of 6~20 carbon atoms of having an appointment, and the ester group of about 1~10 carbon atom; The water-soluble salt that contains the segmental 2-acyloxy-alkyl of the alkane-1-sulfonic acid of the acyl group of 2~9 carbon atoms of having an appointment and about 9~about 23 carbon atoms; The water soluble alkene sulfonated hydrocarbon that contains 12~24 carbon atoms of having an appointment; The segmental b-alkoxy alkane of alkane sulfonate with the alkyl that contains 1~3 carbon atom of having an appointment and about 8~20 carbon atoms.
Particularly preferred herein anion surfactant is an alkyl polyethoxye vitriol, and general formula is RO (C
2H
4O)
xSO
3 -M
+, wherein R is the saturated or unsaturated alkyl chain with about 10~about 22 carbon atoms, M makes compound water-soluble positively charged ion, particularly basic metal, the ammonium cation of ammonium or replacement, and x average out to about 1~about 15.
Preferred alkyl sulfate surfactant is the C of non-ethoxylation
12-15The firsts and seconds alkyl-sulphate.Under the cold water washing condition, promptly be lower than about 65 °F (18.3 ℃), the preferred mixing used this ethoxylation and alkyl-sulphate non-ethoxylatedization.The example of lipid acid comprises capric acid, lauric acid, and tetradecanoic acid, palmitinic acid, stearic acid, eicosanoic acid is with docosoic.Other lipid acid comprises Zoomeric acid, oleic acid, linolic acid, linolenic acid and ricinolic acid.
Nonionogenic tenside
Conventional nonionic and amphoterics comprise C
12-C
18Alkylethoxylate (AE) comprises so-called narrow peak type alkylethoxylate and C
6-C
12Alkyl phenolic alkoxy thing (particularly ethoxylate and blended oxyethyl group/propoxylated glycerine).Also can use C
10-C
18N-alkyl polyhydroxy fatty amide.Typical example comprises C
12-C
18The N-methyl glucose amide.See WO9,206,154.Other sugared deutero-tensio-active agent comprises N-alkoxyl group polyhydroxy fatty acid amide, for example C
10-C
18N-(3-methoxy-propyl) glucamide.The N-propyl group is to the C of N-hexyl
12-C
18Glucamide can be used for low foaming.Also can use C
10-C
20Conventional soap.High if desired foaming can be used side chain C
10-C
16Soap.The example of nonionogenic tenside is disclosed in U.S. Patent No. 4,285,841, and Barrat etc. announced on August 25th, 1981.
The example of preferred these tensio-active agents comprises ethoxy alcohol and oxyethyl group alkyl phenol, and its general formula is R (OC
2H
4)
nOH, wherein R be selected from contain have an appointment the aliphatic group of 8~about 15 carbon atoms and wherein alkyl contain the alkyl phenyl of 8~about 12 carbon atoms of having an appointment, and the mean value of n is about 5~about 15.These tensio-active agents are disclosed in the U.S. Patent No. of announcing on August 18th, 1,981 4,284,532 more fully, among the Leikhim etc.Particularly preferably be ethoxy alcohol, it has average about 10~about 15 carbon atoms in alcohol, and average ethoxyquin degree is every mol of alcohol 6~about 12 moles of ethylene oxide of having an appointment.The mixture of negatively charged ion and nonionogenic tenside is useful especially.
The useful tensio-active agent of other routine is listed in the standard body, comprises C
12-C
18Trimethyl-glycine and sulphobetaine (sultaine).
Amine oxide surfactant
At this, described composition also contains the following amine oxide surfactant of general formula: R
1(EO)
x(PO)
y(BO)
zN (O) (CH
2R ')
2qH
2O (I)
Usually, as can be seen, structure (I) provides a long-chain fragment R
1(EO)
x(PO)
y(BO)
zWith two short pieces, CH
2R '.R ' preferentially is selected from hydrogen, methyl and-CH
2OH.Common R
1Being the alkyl fragment of one-level or branching, can be saturated or unsaturated, preferred R
1It is one-level alkyl fragment.When x+y+z=0, R
1Be that chain is about 8~about 18 alkyl fragment.When x+y+z is not 0, R
1Can be a little longer, the chain length scope is C
12-C
24This general formula also comprises amine oxide, x+y+z=0 wherein, R
1=C
8-C
18, R ' is H, and q is 0-2, is preferably 2.The example of these amine oxides is C
12-14Alkyl dimethyl amine oxide, cetyl dimethyl amine oxide, octadecyl amine oxide, and hydrate, particularly dihydrate, as be disclosed in United States Patent (USP) 5,075, the compound in 501 and 5,071,594 is combined in herein as a reference.
The present invention also comprises amine oxide, and wherein x+y+z is not 0, particularly x+y+z be about 1~about 10, R
1For containing 8~about 24 carbon atoms, the one-level alkyl of preferred about 12~about 16 carbon atoms; In these examples, y+z is preferably 0, and x is preferably about 1~about 6, more preferably about 2~about 4; EO represents ethyleneoxy group, and PO represents propylidene oxygen base, and BO represents the butylene oxide base.This amine oxide can for example by alkyl ethoxy sulfate and dimethyl amine reaction, prepare with the hydrogen peroxide oxidation amine ethoxylate subsequently by conventional synthetic method preparation.
Herein, highly preferred amine oxide is a solid in room temperature, and more preferably their fusing point is 30 ℃~90 ℃.Many suppliers comprise Akzo Chemie, Ethyl Corp. and Procter ﹠amp; Gamble, suitability for industrialized production is suitable for amine oxide herein.Selectable amine oxide manufacturer sees McCutcheon ' s compilation and Kirk-Othmer survey article.Preferred commercially available amine oxide is a solid, i.e. two hydration ADMOX 16 and the ADMOX 18 of Ethyl Corp, and ADMOX 12, and particularly ADMOX 14.
Preferred examples comprises the dimethyl dodecyl amine oxide dihydrate, the cetyl dimethyl amine oxide dihydrate, octadecyl dimethyl amine oxide dihydrate, hexadecyl three (ethyleneoxy group) dimethyl oxidation amine, the CH3-(CH2)13N(CH3)2-O dihydrate, and composition thereof.
Although R ' is H in some preferred examples, wishing that in some cases R ' is a bit larger tham H.Especially, the present invention comprises that also wherein R ' is CH
2The example of OH, for example two (2-hydroxyethyl) amine oxides of hexadecyl, two (2-hydroxyethyl) amine oxides of tallow, two (2-hydroxyethyl) amine oxides of octadecyl and two (2-hydroxyethyl) amine oxides of oil base.
Washing assistant
At this, described composition also randomly, but preferably contain the highest about 50%, more preferably from about 1%~about 40%, the detergent builder compound material of 5%~about 30% (weight) most preferably from about.Yet this washing assistant that does not mean that lower or higher level is left out.Detergent builder compound can randomly be included in the compositions herein, to help the control hardness of minerals.Can use inorganic and organic washing-assisting detergent.In fabric cleaning composition, use washing assistant typically, to help to remove the particulate stain.Detergent builder compound is disclosed in the U.S. Patent No. of announcing March 23 nineteen eighty-two 4,321,165, among the Smith etc.The preferred washing assistant that uses is disclosed in the U.S. Patent No. of announcing on August 18th, 1,981 4,284,532 in the liquid washing agent herein, among the Leikhim etc.
The example of silicate-like builder is an alkalimetal silicate, particularly those SiO
2: Na
2The O ratio is 1.6: 1~3.2: 1 silicate and a lamellated silicate, for example is disclosed in the United States Patent (USP) of announcing on May 12nd, 1,987 4,664,839, the lamina sodium silicate among the H.P.Rieck.NaSKS-6 is the trade mark (generally abbreviating " SKS-6 " herein as) of the crystalline layered silicate of Hoechst sale.Different with zeolite builders, Na SKS-6 silicate-like builder does not contain aluminium.NaSKS-6 has the δ-Na of layered silicate
2SiO
5Form.It can be with for example being disclosed in German patent DE-A-3, the method preparation in 417,649 and DE-A-3,742,043.SKS-6 is the highly preferred layered silicate that uses herein, but other this class layered silicate, for example those general formulas are NaMSi
xO
2x+1yH
2O, wherein M is sodium or hydrogen, x is 1.9~4 numerical value, is preferably 2, and y is 0~20 numerical value, is preferably 0 layered silicate and also can be used for herein.Other layered silicate that Hoechst provides comprises NaSKS-5, and NaSKS-7 and NaSKS-11 is α, β and γ type.As noted above, δ-Na
2SiO
5(NaSKS-6 type) most preferably uses herein.Other silicate also can use, Magnesium Silicate q-agent for example, and it can be as the stablizer of oxygen bleaching agent, and as the component of foam control system.
The example of carbonate builders is alkaline-earth metal and alkaline carbonate, as is disclosed in the German patent application No.2 that announced on November 15th, 1973, the carbonate in 321,001.
The silico-aluminate washing assistant can be used for the present invention.The silico-aluminate washing assistant is a kind of important washing assistant composition in liquid cleaning composition.
The silico-aluminate washing assistant comprises the compound that empirical formula is following: M
z(zAlO
2)
y] xH
2O wherein z and y is at least 6 integer, and the mol ratio of z and y is 1.0~about 0.5, and x is about integer of 15~about 264.
Useful aluminosilicate ion exchange material is commercially available.These silico-aluminates structurally can be crystalline or unformed, and can be the natural silico-aluminate or the alumino-silicate derivatives of synthetic.A kind of method of producing aluminosilicate ion exchange material is disclosed in United States Patent (USP) 3,985,669, and Krummel etc. announced on October 12nd, 1976.The available name that can be used for preferred synthetic crystallization aluminosilicate ion exchange material herein is called zeolite A, zeolite P (B), zeolite MAP and X zeolite.In an especially preferred embodiment, the crystal aluminosilicate ion-exchange material has following general formula: Na
12[(AlO
2)
12(SiO
2)
12] xH
2O wherein x is about 20~about 30, particularly about 27.This material is called as zeolite A.Dehydration zeolite (x=0~10) also can be used for herein.Preferably, the granular size of silico-aluminate is about 0.1~10 micron an of diameter.
The organic washing-assisting detergent that is suitable for the object of the invention comprises various multi-carboxylate's compounds, but is not limited to this.When using herein, " multi-carboxylate " is meant the compound with a plurality of carboxylate group, preferably has 3 carboxylate group at least.The multi-carboxy acid salt washing agent can join with the form of acid in the described composition usually, but also can add with the form of neutralized salt.When using with the form of neutralized salt, basic metal, sodium for example, potassium, and lithium, or pure ammonium salt is preferred.
Comprise various useful materials among the multi-carboxy acid salt washing agent.The important multi-carboxy acid salt washing agent of one class comprises the ether multi-carboxylate, comprises the oxygen disuccinate, is disclosed in the United States Patent (USP) of announcing on April 7th, 1,964 3,128,287, the United States Patent (USP) 3,635,830 that Berg and on January 18th, 1972 announce, Lamberti etc.Also can be referring to the United States Patent (USP) of announcing on May 5th, 1,987 4,663,071, Bush etc., in disclosed " TMS/TDS " washing assistant.Suitable ether multi-carboxylate also comprises ring compound, particularly alicyclic compound, for example is disclosed in United States Patent (USP) 3,923,679; 3,835,163; 4,158,635; Compound in 4,120,874 and 4,102,903.
Other useful detergent builder compound comprises ether hydroxypolycarboxylic acid salt, the multipolymer of maleic anhydride and ethene or methoxy ethylene, 1,3,5-trihydroxybenzene-2,4,6-trisulfonic acid and carboxymethyl oxydisuccinic acid, polyacetic acid, the various basic metal of ethylenediamine tetraacetic acid (EDTA) and nitrilotriacetic acid(NTA) for example, ammonium and replace the salt of ammonium, and multi-carboxylate, mellitic acid for example, succsinic acid, oxygen disuccinic acid, polymaleic acid, 1,3, the 5-benzene tricarboxylic acid, carboxymethyl oxydisuccinic acid, and soluble salt.
The Citrate trianion washing assistant, for example citric acid and soluble salt thereof (particularly sodium salt) are the multi-carboxy acid salt washing agents of the particularly important of heavy duty liquid detergent compositions, because it can be obtained by renewable resources, and are biodegradable.In this based composition and mixture, the oxygen disuccinate also is useful especially.
Disclosed 3 in the United States Patent (USP) 4,566,984 of the Bush that announced on January 28th, 1986,3-dicarboxyl-4-oxa--1,6-hexanedione salt and related compound also are applicable to detergent composition of the present invention.Useful succsinic acid washing assistant comprises C
5-C
20Alkyl and alkenyl succinic acid and salt thereof.A kind of particularly preferred this compounds is the dodecenyl succinic succsinic acid.The specific example of succinate washing assistant comprises dodecyl succinate salt, tetradecyl succinate, hexadecyl succinate, 2-dodecenyl succinic succinate (preferably), 2-15 carbene base succinates etc.Dodecyl succinate salt is preferred washing assistant in this group washing assistant, and is disclosed in the european patent application of announcing on November 5th, 1,986 86200690.5/0,200,263.
Other suitable multi-carboxylate is disclosed in the United States Patent (USP) 3,308,067 of the Diehl that the United States Patent (USP) of the Crutchfield that announced on March 13rd, 1979 on March 7th, 4,144,226 and 1967 announced.Also can be referring to the United States Patent (USP) 3,723,322 of Diehl.
Lipid acid, for example C
12-C
18Monocarboxylic acid, also can be individually, or with aforementioned washing assistant, particularly Citrate trianion and/or succinate washing assistant in combination, be included in the described composition, wash activity so that additional helping to be provided.Use lipid acid can reduce lather usually like this, formulator should be considered this point.
Under the situation that can use phosphorus base washing assistant, can use various alkali metal phosphates, for example well-known tripoly phosphate sodium STPP, trisodium phosphate, and sodium orthophosphate.The phosphonate washing assistant, ethane-1-hydroxyl-1 for example, 1-diphosphonate and other known phosphonate (are seen for example United States Patent (USP) 3,159,581; 3,213,030; 3,422,021; 3,400,148 and 3,422,137) also can use.
The composition component that other is optional
Except that disclosed liquid phase and solid components before this, described aqueous detergent compositions can and preferably contain various other optional components.The optional component of this class can be the liquid or solid form.Described optional component can be dissolved in the liquid phase, perhaps can be dispersed in the liquid phase with the form of fine particle or small droplets.Can randomly be used for being described in detail as follows at this some other materials of composition:
Optional inorganic builders
The detergent composition here can also be chosen wantonly and contain one or more listed before this washing assistants inorganic builders in addition, and this inorganic builders also plays the effect of alkali source.The optional inorganic builders of this class for example comprises the silico-aluminate such as zeolite.Aluminosilicate zeolite, and they are as the application of washing assistant has detailed discussion in the U.S. Patent No. 4,605,509 of the Corkill that announces on August 12nd, 1986 etc., and disclosed content is incorporated herein by reference in this patent.The crystalline layered silicate, for example those layered silicates of discussing in these No. 509 United States Patent (USP)s also are applicable to the detergent composition at this.If use optional inorganic builders, it can constitute about 2%~15% (weight) of composition described herein.
Optional enzyme
Detergent composition described herein can also be chosen wantonly and contain one or more detergent enzymes.Such detergent enzyme can comprise proteolytic enzyme, amylase, cellulase and lipase.These materials are known in the art, and are commercially available.They also can be with suspension, and the form of " marumes " or " bead " is combined in the waterborne liquid detergent composition described herein.Another kind of suitable enzyme comprises the enzyme that exists with the enzyme slurries form in nonionogenic tenside, the enzyme of the commodity " SL " by name of for example Novo Nordisk sale, or the enzyme of the micro encapsulation of the commodity " LDP " by name of Novo Nordisk sale.
Preferred especially use form with the enzyme bead of routine to join enzyme in the composition described herein herein.The size of such bead is generally about 100~1000 microns, and more preferably about 200~800 microns, and be suspended in the whole non-water liquid phase of described composition.Keeping for a long time aspect the enzymic activity, having been found that with other the form of enzyme and comparing that the bead in the present composition has the enzyme stability of special hope.Like this, use the composition of enzyme bead not need to contain conventional enzyme stabilizers, for example institute's stablizer of necessary use usually when combine enzyme in the waterborne liquid washing composition.
If the use enzyme, then enzyme is combined in herein the non-aqueous liquid compositions with enough amounts usually, so that the weight of organized enzyme is up to about 10mg in every gram composition, is more typically about 0.01mg~about 5mg.Point out that in addition it is about 0.001%~5% that non-aqueous liquid detergent compositions described herein generally includes, the commercialization zymin of preferred about 0.01%~1% (weight).For example, the content of proteolytic enzyme in such commercialization preparation is enough to provide every gram composition 0.005~0.1Anson unit (AU) activity usually.
Optional sequestrant
Detergent composition described herein can also be chosen wantonly and contain a kind of sequestrant, and this sequestrant is used for being sequestered in the metal ion in this non-aqueous liquid detergent compositions, for example iron ion and/or mn ion.Like this, described sequestrant in order to composition in metallic impurity form title complex, otherwise these impurity make the component in the composition easily, for example the peroxygen bleach inactivation.Useful chelating agents comprises aminocarboxylate, phosphonate, amino phosphonates do, fragrant chelating agent of multifunctional replacement and composition thereof.
Useful aminocarboxylate as optional sequestrant comprises edetate, the N-hydroxyethyl-ethylenediamine triacetate, nitrilotriacetic acid(NTA) salt, ethylenediamine tetrapropionic acid(EDTP) salt, triethylenetetraaminehexaacetic acid salt, diethylentriamine pentacetate, ethylenediamine disuccinate and ethanol Diglycocol.An alkali metal salt of these materials is preferred.
When allow having at least low-level total phosphorus in the detergent composition, amino phosphonates do also is suitable as sequestrant and is used for composition of the present invention, and comprises satisfactory (DEQUEST) ethylenediamine tetraacetic (methylene phosphonic acid salt).These amino phosphonates do preferably do not contain the alkyl or alkenyl with 6 above carbon atoms.
Preferred sequestrant comprises hydroxyethyl di 2 ethylhexyl phosphonic acid (HEDP), diethylene triaminepentaacetic acid(DTPA) (DTPA), ethylenediamine disuccinic acid (EDDS) and pyridine dicarboxylic acid (DPA), and salt.Certainly, when composition described herein was used for fabric washing/bleaching process, described sequestrant was also as washing assistant.If use sequestrant, sequestrant can constitute about 0.1%~4% (weight) of composition.More preferably, sequestrant can be formed in about 0.2%~2% (weight) of this detergent composition.
Optional thickening material, viscosity control agent and/or dispersion agent
Can also choose wantonly at this detergent composition and to contain polymeric material, this polymeric material keeps its solid particulate components to be the ability of suspended state in order to improve said composition.Like this, these materials can play thickening material, the effect of viscosity control agent and/or dispersion agent.These materials often are the multi-carboxylate polymers, but also can comprise other polymeric material, for example polyvinylpyrrolidone (PVP) or polyamide resin.
The polymeric polycarboxylic acid salt material can be by suitable polymerization of unsaturated monomers or the incompatible preparation of copolymerization, and described monomer is preferably with the form polymerization of acid.Can polymerization comprise vinylformic acid, toxilic acid (or maleic anhydride), fumaric acid, methylene-succinic acid, aconic acid, methylfumaric acid, citraconic acid and methylene radical propanedioic acid with the unsaturated monomer acid that forms suitable multi-carboxylate polymer.In multi-carboxylate polymer described herein, suitably there are some not carboxylic monomer fragments, vinyl methyl ether for example, vinylbenzene, ethene etc., but these fragments surpass about 40% of polymer weight.
Specially suitable multi-carboxylate polymer can be derived by vinylformic acid and obtain.The acrylic acid based polymer that can be used for herein is polyacrylic water-soluble salt.It is about 2,000~100,000 that the molecular-weight average of polymkeric substance of acid form is preferably, more preferably about 2,000~10,000, more more preferably about 4,000~7,000, most preferably is about 4,000~5,000.Polyacrylic water-soluble salt like this can comprise for example an alkali metal salt.The soluble polymer of this class is known material.In detergent composition, use this class polyacrylate, be disclosed in the United States Patent (USP) 3,308,067 of the Diehl that for example announced on March 7th, 1967.Such material can also play the effect of washing assistant.
If use optional thickening material, viscosity control agent and/or dispersion agent, then its content in composition described herein should be about 0.1%~4% (weight).More preferably, such material constitutes about 0.5%~2% (weight) of detergent composition described herein.
Optional earth dirt remover/anti redeposition agent
The present composition can also be chosen wantonly to contain has water-soluble ethoxylated amine of removing earth dirt and antiredeposition performance.If use, then the decontamination material can be formed in about 0.01%~about 5% (weight) of this composition.
Most preferred decontamination and anti redeposition agent are the ethoxyquin tetrens.Typical ethoxylated amine is disclosed in the United States Patent (USP) 4,597,898 of the VanderMeer that announced on July 1st, 1986 in detail.Another organizes preferred earth dirt removal-anti redeposition agent is cation compound, is disclosed in the european patent application 111,965 of the Oh that announced on June 27th, 1984 and Gosselink.Other operable earth dirt removal/anti redeposition agent comprises the ethoxylated amine polymkeric substance in the european patent application 111,984 that is disclosed in the Gosselink that announced on June 27th, 1984; Be disclosed in the amphoteric ion polymer in the european patent application 112,592 of the Gosselink that announced on July 4th, 1984; With the amine oxide in the United States Patent (USP) 4,548,744 that is disclosed in the Connor that announced on October 22nd, 1985.Can also use other earth dirt remover known in the art and/or anti redeposition agent in the composition described herein.Another kind of preferred anti redeposition agent comprises carboxymethyl cellulose (CMC) material.These materials are well-known in the art.
The peroxygen bleach that uses with optional bleach-activating agent
Peroxygen bleach can be organic or inorganic compound in essence.The inorganic peroxy SYNTHETIC OPTICAL WHITNER often is used in combination with bleach-activating agent.
Useful organic peroxy SYNTHETIC OPTICAL WHITNER comprises percarboxylic acids SYNTHETIC OPTICAL WHITNER and salt thereof.The suitable example of this class SYNTHETIC OPTICAL WHITNER comprises six hydration monoperphthalic acid magnesium, metachloroperbenzoic acid magnesium salts, 4-nonyl amino-4-oxo Perbutyric Acid and diperoxy dodecandioic acid.Such SYNTHETIC OPTICAL WHITNER is disclosed in the United States Patent (USP) 4,483,781 of the Hartman that announced on November 20th, 1984; European patent application EP-A-133 of the Banks that on February 20th, 1985 announced etc., 354; United States Patent (USP) 4,412,934 with the Chung that announces November 1 nineteen eighty-three etc.Particularly preferred SYNTHETIC OPTICAL WHITNER also comprises disclosed 6-nonyl amino-6-oxo peroxidation caproic acid (NAPAA) in the United States Patent (USP) 4,634,551 of the Burns that announced on January 6th, 1987 etc.
The inorganic peroxy SYNTHETIC OPTICAL WHITNER also can be used for detergent composition described herein.In fact preferred inorganic SYNTHETIC OPTICAL WHITNER.This class inorganic peroxy compounds comprises alkali metal perborate and percarbonate material, most preferably percarbonate.For example, can use Sodium peroxoborate (for example a hydration or tetrahydrate).Suitable inorganic SYNTHETIC OPTICAL WHITNER can also comprise the peroxyhydrate and the percarbonate bleach of equal value of yellow soda ash or salt of wormwood, trisodium phosphate peroxyhydrate, peroxide hydration urea, and sodium peroxide.Also can use persulphate SYNTHETIC OPTICAL WHITNER (for example OXONE of DuPont company suitability for industrialized production).The inorganic peroxy SYNTHETIC OPTICAL WHITNER often is coated with a layer silicate, borate, vitriol or water miscible tensio-active agent.For example, the percarbonate particles of band coating can be by many commercial sources, for example FMC, Solvay Interox, Tokai Denka and Degussa acquisition.
The inorganic peroxy SYNTHETIC OPTICAL WHITNER, perborate for example, percarbonate etc. preferably are used in combination with bleach-activating agent, this will be in the aqueous solution original position (promptly in the process that composition described herein is used for fabric washing/bleaching) generate peroxy acid corresponding to bleach-activating agent.Various nonrestrictive activator examples are disclosed in the United States Patent (USP) 4,915,854 of the Mao that announces April 10 nineteen ninety etc.; In the United States Patent (USP) 4,412,934 of the Chung that announces November 1 nineteen eighty-three etc.Be typically nonanoyl oxygen benzene sulfonate (NOBS) and tetra acetyl ethylene diamine (TAED) activator.Also can use its mixture.Other can be as used herein typical SYNTHETIC OPTICAL WHITNER and activator are referring to mentioned before this United States Patent (USP) 4,634,551.
Other useful amido deutero-bleach-activating agent has following chemical formula:
R
1N (R
5) C (O) R
2C (O) L or R
1C (O) N (R
5) R
2C (O) L is R wherein
1Be the alkyl that contains 6~about 12 carbon atoms of having an appointment, R
2Be the alkylidene group that contains 1~about 6 carbon atoms, R
5Be hydrogen or the alkyl that contains 1~about 10 carbon atoms of having an appointment, aryl, or alkaryl, L are any suitable leavings groups, oxygen benzene sulfonate for example ,-OOH ,-OOM.Leavings group is substituted any group from the bleach-activating agent that hydroperoxyl radical negatively charged ion nucleophilic attack bleach-activating agent is caused.Preferred leavings group is a phenolsulfonate.
The example of the preferred bleach-activating agent in the above-mentioned chemical formula comprises (the amino caproyl of 6-decoyl) oxygen benzene sulfonate, (the amino caproyl of 6-nonanoyl) oxygen benzene sulfonate, (the amino caproyl of 6-caprinoyl) oxygen benzene sulfonate; and composition thereof; be disclosed in the United States Patent (USP) of mentioning before this 4,634,551.Such mixture is reduced (6-C at this
8-C
10The alkyl amido caproyl) oxygen benzene sulfonate.
Another kind of useful bleach-activating agent comprises the benzo oxazinyl activator, is disclosed in the United States Patent (USP) 4,966,723 of the Hodge that announces October 30 nineteen ninety etc., and it is combined in herein as a reference.Highly preferred benzo oxazinyl activator is:
Also have the useful bleach-activating agent of a class to comprise acyl caprolactam and the acyl group Valerolactim that acyl lactam activator, particularly general formula are following:
R wherein
6For H or contain the alkyl of 1~about 12 carbon atoms, aryl, alkoxy aromatic yl, or alkaryl.Highly preferred lactan activator comprises benzoyl caprolactam, capryloyl hexanolactam, 3; 5,5-trimethyl acetyl base hexanolactam, nonanoyl hexanolactam; the decanoyl hexanolactam, undecanoyl hexanolactam, benzoyl Valerolactim; the capryloyl Valerolactim, decanoyl Valerolactim, undecanoyl Valerolactim; 3; 5,5-trimethyl acetyl base Valerolactim, and composition thereof.Referring to the United States Patent (USP) 4,545,784 of the Sanderson that announced on October 8th, 1985, it is combined in herein as a reference, and it discloses acyl caprolactam, comprises the benzoyl caprolactam that is adsorbed on the Sodium peroxoborate.
If the use peroxygen bleach, its content is about 0.1%~30% of composition weight usually.More preferably, the content of peroxygen bleach is about 1%~20% of composition weight.Most preferably the content of peroxygen bleach is about 5%~20% of composition weight.If use, the content of bleach-activating agent is the about 0.5%~20% of composition weight, more preferably from about 3%~10%.Frequently, the mol ratio of SYNTHETIC OPTICAL WHITNER and activator is about 1: 1~10: 1, more preferably from about 1.5: 1~5: 1.
In addition, have been found that when for example citric acid flocked together, its chemical was more stable when bleach-activating agent and some acid.
Optional bleaching catalyst
If desired, bleaching compounds can carry out catalysis with manganic compound.This compound is well known in the art, and comprises and for example be disclosed in United States Patent (USP) 5,246,621, United States Patent (USP) 5,244,594; United States Patent (USP) 5,194,416; United States Patent (USP) 5,114,606; With the open Nos.549 of european patent application, 271A1,549,272A1,544,440A2 and 544, the manganese-based catalyst among the 490A1.The preferred embodiment of these catalyzer comprises: Mn
IV 2(u-O)
3(1,4,7-trimethylammonium-1,4,7-7-triazacyclononane)
2(PF
6)
2, Mn
III 2(u-O)
1(u-OAc)
2(1,4,7-trimethylammonium-1,4,7-7-triazacyclononane)
2(ClO
4)
2, Mn
IV 4(u-O)
6(1,4, the 7-7-triazacyclononane)
4(ClO
4)
4, Mn
IIIMn
IV 4(u-O)
1(u-OAc)
2-(1,4,7-trimethylammonium-1,4,7-7-triazacyclononane)
2(ClO
4)
3, Mn
IV(1,4,7-trimethylammonium-1,4,7-7-triazacyclononane)-(OCH
3)
3(PF
6), and composition thereof.Other metal matrix bleaching catalyst comprises and is disclosed in United States Patent (USP) 4,430,243 and United States Patent (USP) 5,114,611 in those catalyzer.Use improves bleaching effect with the manganese of various complicated parts, report is also arranged: 4,728,455 in following United States Patent (USP); 5,284,944; 5,246,612; 5,256,779; 5,280,117; 5,274,147; 5,153,161; With 5,227,084.
In fact, but without limitation, can regulate the compositions and methods of the invention, the order of magnitude of the active bleaching catalyst in the aqueous washing liq one of was at least 1,000 ten thousand fens, and preferably the laundry liquid in catalyst content be about 0.1ppm~about 700ppm, more preferably about 1ppm~about 500ppm.
Operable herein cobalt bleaching catalyst is known, and is disclosed in for example M.L.Tobe, " Base Hydrolysis of Transition-Metal Complexes ",
Adv.lnorg.Bioinorg. Mech., (1983), 2, in the 1-94 page or leaf.Operable herein most preferred cobalt catalyst is that general formula is [Co (NH
3)
5OAc] the cobalt amylamine acetate of Ty, wherein " OAc " represents the acetate moiety fragment, and " Ty " is negatively charged ion, cobalt amylamine acetate muriate particularly, [Co (NH
3)
5OAc] Cl
2And [Co (NH
3)
5OAc] (OAc)
2[Co (NH
3)
5OAc] (PF
6)
2[Co (NH
3)
5OAc] (SO
4); [Co (NH
3)
5OAc] (BF
4)
2[Co (NH
3)
5OAc] (NO
3)
2(being " PAC ") herein.
These cobalt catalyst prepare easily with currently known methods, for example in the article of Tobe and its reference of quoting, and the United States Patent (USP) 4,810,410 of the Diakun that on March 7th, 1989 announced etc.,
J. Chem.Ed.(1989),
66(12), 1043-45; The Synthesis and Characterization ofInorganic Compounds, W.L.Jolly (Prentice-Hall; 1970), pp.461-3;
Inorg. Chem.,
18, 1497-1502 (1979);
Lnorg.Chem., 21,2881-2885 (1982);
Inorg. Chem.,
18, 2023-2025 (1979); Inorg.Synthesis, 173-176 (1960); With
Journal. Of Physical Chemistry,
56, disclosed method among the 22-25 (1952).
In fact, but without limitation, can regulate compositions herein and cleaning method, make the order of magnitude of the active bleaching catalyst class material in the aqueous cleaning medium be at least 1/100000000th, and preferably to make the content of the bleaching catalyst class material in the washing liq be about 0.01ppm~about 25ppm, more preferably about 0.05ppm~about 10ppm, most preferably from about 0.1ppm~about 5ppm.For obtaining the content of this level in the washing liq of automatic washing process, typical herein composition will comprise about 0.0005%~about 0.2%, more preferably from about 0.004%~about 0.08% bleaching catalyst, particularly manganese or cobalt catalyst are in the weight of cleaning compositions.
Optional whitening agent, suds suppressor, dyestuff and/or perfume compound
Detergent composition of the present invention can also be chosen wantonly and contain conventional whitening agent, suds suppressor, dyestuff and/or fragrance materials.Certainly, these whitening agent, suds suppressor, silicone oil, dyestuff and perfume compound, compatible with other composition component under water-less environment, and do not react with them.If exist, whitening agent then, suds suppressor, the typical content of dyestuff and/or perfume compound is about 0.0001%~2% of a present composition weight.
The polymkeric substance stain remover
In the compositions and methods of the invention, can choose wantonly and use any polymkeric substance stain remover well known by persons skilled in the art.The feature of polymkeric substance stain remover is existing hydrophilic segment, makes hydrophobic fiber, as the surface hydrophilic of polyester and nylon, hydrophobic fragment is arranged again, being deposited on the hydrophobic fiber, and on whole washing and rinse cycle can both be attached to fibers, serve as the anchorage of hydrophilic segment.This just makes the dirt easier cleaning in the washing process of back after the detergent-treatment.
The example of the polymkeric substance stain remover that the present invention is used comprises: the United States Patent (USP) 4,721,580 of the Gosselink that on January 26th, 1988 announced; The United States Patent (USP) 4,000,093 of the Nicol that on December 28th, 1976 announced etc.; The european patent application 0 219 048 of the Kud that on April 22nd, 1987 announced etc.; The United States Patent (USP) 4,702,857 of the Gosselink that on October 27th, 1987 announced etc.; The United States Patent (USP) 4,968,451 of the J.J.Scheibel that announce November 6 nineteen ninety.Commercially available stain remover comprises SOKALAN type material, and SOKALAN HP-22 for example is available from BASF (West Germany).The United States Patent (USP) 3,893,929 of the Basadur that can on July 8th, 3,959,230 and 1975 announce referring to the United States Patent (USP) of the Hays of the announcement on May 25th, 1976 also.The example of this polymkeric substance comprises commercially available material ZELCON5126 (available from Dupont) and MILEASE T (available from ICI).Other suitable polymers stain remover comprises the United States Patent (USP) 4 of the Gosselink that announced on December 8th, 1987 etc., 711, disclosed polyterephthalate in 730, the United States Patent (USP) 4 of the Gosselink that on January 26th, 1988 announced, the end capped oligomer ester of disclosed negatively charged ion in 721,580, and the United States Patent (USP) 4 of the Gosselink that announced on October 27th, 1987, disclosed block polyester oligopolymer in 702,857.Preferred polymkeric substance stain remover also comprises disclosed stain remover in the United States Patent (USP) 4,877,896 of the Maldonado that on October 31st, 1989 announced etc.
If use, the content of stain remover be generally detergent composition weight about 0.01%~about 10.0%, typically be about 0.1%~about 5%, preferred about 0.2%~about 3.0%.
Sequestrant
Detergent composition of the present invention can also be chosen wantonly and contain one or more iron and/or manganese sequestrant.The defined aminocarboxylate in the optional back freely of this sequestrant, amino phosphonates do, fragrant chelating agent that polyfunctional group replaces and composition thereof.Be not to limit with theory intentionally, but it is believed that the benefit of using these materials, part is because they can be by forming the special ability that soluble inner complex removes iron and manganese from washing soln.
The aminocarboxylate that can be used as optional sequestrant comprises edetate, the N-hydroxyethyl-ethylenediamine triacetate, nitrilo-triacetate, ethylenediamine tetrapropionic acid(EDTP) salt, triethylenetetraaminehexaacetic acid salt, diethylentriamine pentacetate, with the ethanol Diglycocol, its basic metal, ammonium, with the salt that replaces ammonium, and composition thereof.
When allow having at least low-level total phosphorus in detergent composition, amino phosphonates do also is suitable as the sequestrant in the present composition, and comprises satisfactory ethylenediamine tetraacetic (methylene phosphonic acid salt).Preferably, these amino phosphonates do do not contain the alkyl or alkenyl that has above about 6 carbon atoms.
The fragrant chelating agent that polyfunctional group replaces also can be used in the composition of the present invention.United States Patent (USP) 3,812,044 referring to the Connor that announced on May 21st, 1974 etc.The preferred this compound that exists with sour shape is the dihydroxyl disulfobenzene, for example 1, and 2-dihydroxyl-3,5-disulfobenzene.
The preferred biodegradation type sequestrant that herein uses is ethylenediamine disuccinate (" EDDS "), disclosed [S, S] isomer particularly as in the United States Patent (USP) 4,704,233 of Harman that announced on November 3rd, 1987 and Perkins.
If use, the content of these sequestrants is generally about 0.1%~about 10% of detergent composition weight.More preferably, if use, the content of these sequestrants is about 0.1%~about 3.0% of this composition weight.
Earth dirt remover/anti redeposition agent
Composition of the present invention also can be chosen wantonly and contain water miscible ethoxylated amine, and it has the earth dirt and removes performance and antiredeposition performance.Typically, liquid detergent composition contains 0.01%~about 5% these compositions of having an appointment.
Most preferred decontamination and anti redeposition agent are the ethoxyquin tetren.Typical ethoxylated amine also further is disclosed in the United States Patent (USP) 4,597,898 of the VanderMeer that announced on July 1st, 1986.Another organizes preferred earth dirt remover-anti redeposition agent, is the cation compound that is disclosed in the european patent application 111,965 of the Oh that announced on June 27th, 1984 and Gosselink.Other operable earth dirt remover/anti redeposition agent comprises the ethoxylated amine polymkeric substance in the european patent application 111,984 that is disclosed in the Gosselink that announced on June 27th, 1984; Be disclosed in the amphoteric ion polymer in the european patent application 112,592 of the Gosselink that announced on July 4th, 1984; With the amine oxide in the United States Patent (USP) 4,548,744 that is disclosed in the Connor that announced on October 22nd, 1985.In composition of the present invention, also can use other earth dirt remover and/or anti redeposition agents known in the art.Another kind of preferred anti redeposition agent comprises carboxymethyl cellulose (CMC) material.These materials are known in this area.
Polymeric dispersant
In composition of the present invention, it is favourable using polymeric dispersant with the level of about 0.1%~7% (weight), particularly under the situation of zeolite and/or the existence of layered silicate washing assistant.The suitable polymers dispersion agent comprises multi-carboxylate polymer and polyoxyethylene glycol, although also can use other polymeric dispersant known in the art.Be not limited by theory although do not want, but it is believed that when polymeric dispersant and other washing assistant (comprising low-molecular-weight multi-carboxylate) when being used in combination, by suppressing crystalline growth, particle dirt peptization and anti-redeposition, it can improve the overall performance of washing assistant.
The polymeric polycarboxylic acid salt material can be by suitable polymerization of unsaturated monomers or the incompatible preparation of copolymerization, and described monomer is preferably with the form polymerization of acid.Can polymerization comprise vinylformic acid, toxilic acid (or maleic anhydride), fumaric acid, methylene-succinic acid, aconic acid, methylfumaric acid, citraconic acid and methylene radical propanedioic acid with the unsaturated monomer acid that forms suitable multi-carboxylate polymer.In multi-carboxylate polymer described herein, suitably there are some not monomer fragments of carboxylate-containing base, vinyl methyl ether for example, vinylbenzene, ethene etc., but these fragments surpass about 40% of polymer weight.
Specially suitable multi-carboxylate polymer can be derived by vinylformic acid and obtain.The acrylic acid based polymer that can be used for herein is polyacrylic water-soluble salt.It is about 2,000~10,000 that the molecular-weight average of polymkeric substance of acid form is preferably, more preferably about 4,000~7,000, most preferably is about 4,000~5,000.Polyacrylic water-soluble salt like this can comprise for example basic metal, the salt of ammonium and replacement ammonium.The soluble polymer of this class is known material.In detergent composition, use this class polyacrylate, be disclosed in the United States Patent (USP) 3,308,067 of the Diehl that for example announced on March 7th, 1967.
Vinylformic acid/maleic copolymer also can be as preferred dispersion/anti redeposition agent component.This material comprises the water-soluble salt of vinylformic acid and maleic acid.It is about 2,000~100,000 that the molecular-weight average of this multipolymer of acid form is preferably, more preferably about 5,000~75,000, most preferably is about 7,000~65,000.The segmental ratio of acrylate and maleate was generally about 30: 1~about 1: 1 in this multipolymer, more preferably from about 10: 1~2: 1.The water-soluble salt of this vinylformic acid/maleic acid can comprise, basic metal for example, ammonium and replace the salt of ammonium.Such water soluble acrylate/maleate copolymer is a known substances, it is disclosed in the european patent application No.66915 that announces December 15 nineteen eighty-two, and among the EP 193,360 of announcement on September 3rd, 1986, it also discloses this polymkeric substance that comprises the hydroxypropyl acrylate.Other useful dispersion agent also comprises toxilic acid/vinylformic acid/vinyl alcohol terpolymer.This material also is disclosed among the EP 193,360, comprises 45/45/10 terpolymer as vinylformic acid/toxilic acid/vinyl alcohol.
The another kind of polymkeric substance that can comprise is polyoxyethylene glycol (PEG).PEG demonstrates dispersing agent performance, and can be used as earth dirt remover-anti redeposition agent.The typical molecular weight ranges that uses as this purpose for about 500~about 100,000, preferred about 1,000~about 50,000, more preferably from about 1,500~about 10,000.
Also can use polyaspartic acid salts and polyglutamic acid dipersant, particularly unite use with zeolite builders.Dispersion agent, for example the molecular weight of polyaspartic acid salts (avg.) is preferably about 10,000.
Dye transfer inhibitor
Composition of the present invention can also comprise that one or more can effectively be suppressed at the material that dyestuff is shifted to another fabric by a fabric in the washing process.Usually, this dye transfer inhibitor comprises Polyvinylpyrolidone (PVP), polyamines N-oxide polymer, and the multipolymer of N-vinyl pyrrolidone and N-vinyl imidazole, manganese phthalocyanine, peroxidase, and composition thereof.If use, then the content of these reagent be generally composition weight about 0.01%~about 10%, preferred about 0.01%~about 5%, more preferably from about 0.05%~about 2%.
More clearly, contain the unit with following structural formula: R-A at this polyamines N-oxide polymer that preferably uses
x-P; Wherein P is a polymerizable unit, connects the N-O group above it, and perhaps the N-O group constitutes the part of polymerizable unit, and perhaps the N-O group is connected on two unit; A be down one of array structure :-NC (O)-,-C (O) O-,-S-,-O-,-N=; X is 0 or 1; R is an aliphatics, ethoxylated fat family, and aromatic series, heterocycle or alicyclic group, or its arbitrary combination can connect the N atom of N-O group above it, and perhaps the N-O group is the part of these groups.Preferred polyamines N-oxide compound is that wherein R is a heterocyclic radical, pyridine for example, pyrroles, imidazoles, tetramethyleneimine, piperidines, and those compounds of derivative.
The N-O group can be represented by following formula:
R wherein
1, R
2, R
3Be fatty group, aromatic base, heterocyclic radical or alicyclic radical, or its combination; X, y and z are 0 or 1; And the nitrogen-atoms of N-O group can be connected to any above-mentioned group or constitute the part of any above-mentioned group.The unitary pKa of amine oxide<10 of polyamines N-oxide compound, preferred pKa<7, more preferably pKa<6.
Can use any polymer backbone,, and have the dye transfer rejection as long as the amine oxide polymers that forms is water miscible.The example of suitable polymers skeleton is a polyvinyls, polyalkylene, polyester, polyethers, polymeric amide, polyimide, polyacrylic ester and composition thereof.These polymkeric substance comprise random or segmented copolymer, and wherein a kind of monomer is an amine n-oxide, and other monomer is the N-oxide compound.The amine of typical amine n-oxide polymkeric substance and the ratio of amine n-oxide are 10: 1~1: 1,000,000.Yet the quantity of the amine oxide group that exists in the polyamine oxide polymer can be regulated by suitable copolymerization or by suitable N-oxidisability.Almost can obtain the polyamine oxide compound of any polymerization degree.Typically, its average molecular weight range is 500~1,000,000; More preferably 1,000~500,000; Most preferably 5,000~100,000.The preferred material of this class can be described as " PVNO ".
The most preferred polyamine N-oxide of using in detergent composition of the present invention is poly-(4-vinylpridine-N-oxide compound), and its molecular-weight average is about 50,000, and the ratio of amine and amine n-oxide is about 1: 4.
The also preferred herein multipolymer (being called " PVPVI " class) that uses N-vinyl pyrrolidone and N-vinyl imidazole.The average molecular weight range of preferred PVPVI is 5,000~1,000,000, more preferably 5,000~200,000, most preferably 10,000~20,000 (average molecular weight range can pass through light scattering determining, and this method is disclosed in Barth etc.,
Chemical Analysis, 113 volumes, " ModernMethods of Polymer Characterization ", the content of this piece article is combined in herein as a reference).The mol ratio of N-vinyl imidazole and N-vinyl pyrrolidone is 1: 1~0.2: 1 in the typical PVP VI multipolymer, more preferably 0.8: 1~0.3: 1, and most preferably 0.6: 1~0.4: 1.These multipolymers can be linear, also can be branching.
Composition of the present invention can also use molecular-weight average for about 5,000~about 400,000, preferred about 5,000~about 200,000,5,000~about 50,000 Polyvinylpyrolidone (PVP) (" PVP ") more preferably from about.PVP is known to the technician of detergent applications; Referring to for example EP-A-262,897 and EP-A-256,696, it is combined in herein as a reference.The composition that contains PVP, also can contain molecular-weight average and be about 500~about 100,000, preferred about polyoxyethylene glycol of 1,000~about 10,000 (" PEG ").Preferably, PEG that discharges with the ppm level in washing soln and the ratio of PVP are about 2: 1~about 50: 1, more preferably from about 3: 1~about 10: 1.
Detergent composition of the present invention also can be chosen the wetting ability white dyes that contains 0.005%~about 5% (weight) some type of having an appointment wantonly, and it also can provide the dye transfer restraining effect.If use this white dyes, composition of the present invention preferably contains the white dyes of have an appointment 0.01%~1% (weight).
The wetting ability white dyes of Shi Yonging has following structural formula in the present invention:
R wherein
1Be selected from anilino, N-2-double hydroxyethyl and NH-2-hydroxyethyl; R
2Be selected from the N-2-double hydroxyethyl, N-2-hydroxyethyl-N-methylamino-, morpholino, chlorine and amino; M is the salifiable positively charged ion of shape, for example sodium or potassium.
When in the said structure formula, R
1Be anilino, R
2Be the N-2-double hydroxyethyl, and M is when being positively charged ion as sodium, this white dyes is 4,4 '-two [(4-anilino-6-(N-2-double hydroxyethyl)-s-triazine-2-yl) amino]-2,2 '-stilbene disulfonic acid and disodium salt.This specific brightener species can buy from Ciba-Geigy company, and its commodity are called Tinopal-UNPA-GX.Tinopal-UNPA-GX is the preferred wetting ability white dyes that uses in detergent composition of the present invention.
When in the said structure formula, R
1Be anilino, R
2Be N-2-hydroxyethyl-N-2-methylamino-, and M is when being positively charged ion as sodium, this white dyes is 4,4 '-two [(4-anilino-6-(N-2-hydroxyethyl-N-methylamino-)-s-triazine-2-yl) amino]-2,2 '-stilbene disulfonic acid disodium salt.This specific brightener species can buy from Ciba-Geigy company, and its commodity are called Tinopal 5BM-GX.
When in the said structure formula, R
1Be anilino, R
2Be morpholino, and M is when being positively charged ion as sodium, this white dyes is 4,4 '-two [(4-anilino-6-morpholino-s-triazine-2-yl) amino]-2,2 '-stilbene disulfonic acid sodium salt.This specific brightener species can buy from Ciba Geigy company, and its commodity are called Tinopal AMS-GX.
Select the specific white dyes kind of use among the present invention, when uniting use, have especially effectively dye transfer rejection with selected aforementioned polymer dye transfer inhibitor.The polymer materials of Xuan Zeing (for example PVNO and/or PVPVI) and the white dyes of selecting (Tinopal UNPA-GX for example like this, Tinopal 5BM-GX and/or Tinopal AMS-GX) unite use, compare when using separately with these two kinds of detergent composition components, it has significantly good dye transfer restraining effect in the water-based washing soln.Being not intended to be limited by theory, but it is believed that such whitening agent produces this effect, is because it has strong avidity to fabric in washing soln, therefore can deposit on these fabrics relatively soon.Whitening agent deposits to the degree on the fabric in washing soln, can determine by the parameter that is called " consumption coefficient ".Consumption coefficient normally a) deposits to whitening agent material and b on the fabric) ratio of the starting point concentration of whitening agent in the washing liq.Whitening agent with higher relatively consumption coefficient is suitable for suppressing dye transfer in the present invention most.
Certainly, should be realized that other conventional optical brightener compounds also can randomly be used in the present composition, " brighten " effect so that conventional fabric to be provided, rather than real dye transfer suppresses effect.This usage is conventional to detergent formulation, and is well-known.
Composition forms
Water base heavy duty liquid detergent compositions disclosed by the invention can contain water and other solvent as carrier.Low-molecular-weight firsts and seconds alcohol, methyl alcohol for example, ethanol, propyl alcohol and Virahol are fit to.The preferred monohydroxy-alcohol that uses dissolves tensio-active agent, but also can use polyvalent alcohol, for example contains the polyvalent alcohol (for example, 1, ammediol, ethylene glycol, glycerine and 1,2-propylene glycol) of 2~about 6 carbon atoms and 2~about 6 hydroxyls.Can contain 5%~90% in the described composition, typically be 10%~50% this carrier.
Detergent composition of the present invention is preferably filled a prescription and is, when using in the watersoluble cleaning operation, the pH value of washing water is about 6.5~about 11, preferred about 7.5~11.Be used to control the pH value and comprise the use damping fluid in the technology of the level of recommending to use, alkali, acid etc., and these technology are well known to those skilled in the art.
Preparation of compositions and application
Can prepare water base heavy duty liquid detergent compositions of the present invention by required component is mixed and blend with required solvent.In the typical method of this composition of preparation, necessary also mixes according to specific order under certain conditions with some preferred optional component.
Zhi Bei composition of the present invention as stated above can be used for forming the water-based washing soln that washing and bleached woven fabric are used.Usually, this composition of significant quantity is added to the water, preferably in the automatic washing machine of the laundering of textile fabrics of routine, forms such water-based washing/liquid lime chloride.Water-based washing/the liquid lime chloride that so makes then with to contact with the fabric of its washing and bleaching, preferably under agitation contact.
Herein the be added to the water significant quantity of the liquid detergent composition that forms water-based washing/liquid lime chloride can be that to be enough in the aqueous solution formation about 500~7, the amount of the composition of 000ppm.More preferably, in water-based washing/liquid lime chloride, will provide about 800~3, the detergent composition of 000ppm.
The following examples illustrate the preparation and the performance advantage of the waterborne liquid detergent composition of the present invention that contains the alkylbenzenesulfonatsurfactants surfactants mixture.Yet these embodiment are used for limiting or defining scope of the present invention.Unless otherwise noted, all umbers used herein, per-cent and ratio all are meant weight percent.In the following embodiments, the implication of the abbreviation of used various components is as follows in the composition.In these embodiments, following abbreviation MLAS is sodium salt or the sylvite that is used for representing according to the modified alkylbenzene sulfonic acid of any aforesaid method embodiment preparation.Abbreviation LAS LINER ALKYL BENZENE SULPHONIC ACID SOFT 96 sodium MBAS
x(the vitriol MBAE of average total carbon number=x) of the one-level alkyl of branching in the chain
xS
z(the ethoxyquin of average total carbon number=z) of the one-level alkyl of branching in the chain
(average EO=x) sodium sulfate salt MBAE
x(the b-oxide of average total carbon number=x) of the one-level alkyl of branching in the chain
The activity that (average EO=5) Endolase NOVO Industries A/S sells is 3000 CEVU/g's
Endoglucanase MEA monoethanolamine PG propylene glycol BPP butoxy-propoxy--propyl alcohol EtOH ethanol NaOH sodium hydroxide solution NaTS toluenesulfonic acid sodium salt anhydrous citric acid citric acid C
XyFA C
1x-C
1yLipid acid C
XyE
zContract with average z moles of ethylene oxide and C
1x-1yThe primary alcohol carbonate particle size of branching is that the Carbon Dioxide na citrate granular size of 200 μ m~900 μ m is that the activity of 425 μ m~850 μ m is 86.4% citric acid
Trisodium dihydrate TFAA C16-18 alkyl N-methyl glucose amide LMFAA C12-14 alkyl N-methyl glucose amide APA C8-C10 amido propyl dimethylamine aliphatic acid, (C12/14) C12-C14 aliphatic acid fat acid, (TPK) palm of topping nuclear aliphatic acid fat acid, (RPS) rapeseed oil aliphatic acid Borax sodium borate decahydrate PAA polyacrylic acid, (mw=4500) PEG polyethylene glycol, (mw=4600) MES alkyl methyl sulfonated ester SAS secondary alkyl sulphate salt NaPS paraffin sodium sulfonate C45AS C14-C
15Linear alkyl sodium sulfate CxyAS C
1x-C
1ySodium alkyl sulfate (or other salt, if specify) CxyEzS and the z moles of ethylene oxide contracts and C
1x-C
1ySodium alkyl sulfate (or other
Salt is if specify) CxyEz contract with average z moles of ethylene oxide and C
1x-1yThe primary alcohol AQA R of branching
2N
+(CH
3)
x((C
2H
4O)
yH)
z, R wherein
2=C
8-C
18, x+z=3,
X=0~3, z=0~3, y=1~15STPP anhydrous sodium tripolyphosphate zeolite A primary granule size is that the chemical formula of 0.1~10 μ m is
Na
12(AlO
2SiO
2)
1227H
2The hydrated sodium aluminosilicate NaSKS-6 chemical formula of O is δ-Na
2Si
2O
5Crystalline layered silicate carbonate particle size be that the anhydrous sodium carbonate bicarbonate particles size distribution of 200 μ m~900 μ m is the anhydrous sodium bicarbonate silicate amorphous sodium silicate (SiO of 400 μ m~1200 μ m
2: Na
2O=2.0) toxilic acid/acrylic copolymer of 1: 4 of vitriol anhydrous sodium sulphate PAE ethoxyquin (15-18) tetren PIE ethoxyquin polymine PAEC methyl quaternised ethoxyquin di hexylidene triamine MA/AA, molecular-weight average is about 70, the commodity that 000CMC Xylo-Mucine protease N OVO Industries A/S sells Savinase's by name
Activity is that the commodity that the proteolytic ferment cellulase NOVO Industries A/S of 4KNPU/g sells are called Carezyme's
Activity is the commodity Termamyl 60T by name that the lyticase amylase NOVO Industries A/S of 1000CEVU/g sells
Activity be the commodity Lipolase's by name that sells of the amylolytic enzyme lipase NOVO Industries A/S of 60KNU/g
Active lipolytic enzyme PB1 nominal formula for 100kLU/g is NaBO
2H
2O
2Anhydrous sodium perborate SYNTHETIC OPTICAL WHITNER percarbonate nominal formula be 2Na
2CO
33H
2O
2The tetra-acetylated ethylene diamine DTPMP of SPC-D NaDCC dichloroisocyanuric acid sodium NOBS acyloxy in the ninth of the ten Heavenly Stems Phenylsulfonic acid sodium salt TAED Monsanto commodity Dequest 2060 by name that sells
Diethylenetriamine five (methylene phosphonic acid salt)
The sulfonation phthalocyanine phthalocyanine zinc SYNTHETIC OPTICAL WHITNER of photoactivation bleaching is at the dextrin soluble poly
Form microcapsule whitening agent 14,4 '-two (2-sulfo group styryl) xenyl disodium salt whitening agent 24,4 '-two (4-anilino-6-morpholino-1.3.5-triazine-2-yl) in the compound
Amino) toluylene-2,2 '-disulfonic acid disodium salt HEDP 1,1-hydroxyl ethane di 2 ethylhexyl phosphonic acid SRP 1 have oxyethylene group oxygen base and terephthaloyl skeleton
The ethoxyquin terephthalate polymer silicone antifoams polydimethylsiloxane Foam Control of the end capped ester SRP 2 sulfonated ethoxyquin terephthalate polymer SRP of sulfo group benzoyl 3 methyl blockings has as dispersion agent
Siloxanes-oxyalkylene copolymers, described Foam Control and described
The ratio of dispersion agent is 10: 1~100: the Condea trade mark CaCl of 1Isofol 16 C16 (on average) Guerbet alcohol
2Calcium chloride MgCl
2Magnesium chloride DTPA diethylene triaminepentaacetic acid(DTPA)
Embodiment 25 is according to following formulation liquid detergent composition.
| A | B | ?C | ?D | |
| ?C25AE3S | 2 | 8 | ?7 | ?5 |
| ?MLAS | 15 | 12 | ?10 | ?8 |
| The C12-C14 alkyl dimethyl amine oxide | - | - | ?- | ?2 |
| ?C25AS | 6 | 4 | ?6 | ?8 |
| The C24N-methyl glucose amide | 5 | 4 | ?3 | ?3 |
| ?C24AE5 | 6 | 1 | ?1 | ?1 |
| C12-C18 lipid acid | 11 | 4 | ?4 | ?3 |
| Citric acid | 1 | 3 | ?3 | ?2 |
| ?DTPMP | 1 | 1 | ?1 | ?0.5 |
| ?MEA | 8 | 5 | ?5 | ?2 |
| ?NaOH | 1 | 2.5 | ?1 | ?1.5 |
| ?PG | 14.5 | 13.1 | ?10.0 | ?8 |
| ?EtOH | 1.8 | 4.7 | ?5.4 | ?1 |
| Amylase (300KNU/g) | 0.1 | 0.1 | ?0.1 | ?0.1 |
| Lipase D96/L (100KNU/g) | 0.15 | 0.15 | ?0.15 | ?0.15 |
| Proteolytic enzyme (35g/l) | 0.5 | 0.5 | ?0.5 | ?0.5 |
| ?Endolase | 0.05 | 0.05 | ?0.05 | ?0.05 |
| Cellulase | 0.09 | 0.09 | ?0.09 | ?0.09 |
| The terephthalate based polyalcohol | 0.5 | - | ?0.3 | ?0.3 |
| Boric acid | 2.4 | 2.8 | ?2.8 | ?2.4 |
| Sodium xylene sulfonate | - | 3 | ?- | ?- |
| 2-butyl-octanol | 1 | 1 | ?1 | ?1 |
| Branched silicone | 0.3 | 0.3 | ?0.3 | ?0.3 |
| Water and minor materials | Reach 100% | |||
(A~D) is very effective to remove various dirts from fabric under various working conditionss to the aforesaid liquid detergent composition.
Embodiment 26
(E~J) is according to heavy duty type liquid laundry detergent composition of the present invention to following composition.
| Embodiment #: | ?E | ?F | ?G | ?H | ?I | ?J |
| ?MLAS | ?17 | ?15 | ?7.0 | ?7.0 | ?12 | ?12 |
| ?C35AE3S/C25AE3S | ?2.0 | ?9.0 | ?- | ?- | ?7.0 | ?7.0 |
| ?C25AE2.5S | ?- | ?- | ?12.0 | ?12.0 | ?- | ?- |
| C24 N-methyl glucose amide | ?6.0 | ?5.0 | ?4.5 | ?3.7 | ?4.0 | ?4.0 |
| ?C35E7 | ?6.0 | ?1.0 | ?- | ?- | ?- | ?- |
| ?C23E9 | ?- | ?- | ?2.0 | ?1.0 | ?5.0 | ?5.0 |
| ?C10?APA | ?- | ?1.5 | ?- | ?2.0 | ?- | ?2.5 |
| C24 lipid acid | ?7.5 | ?1.1 | ?2.0 | ?4.0 | ?5.0 | ?5.0 |
| C48 lipid acid | ?3.0 | ?3.5 | ?- | ?- | ?- | ?- |
| Citric acid | ?1.0 | ?3.5 | ?3.0 | ?3.0 | ?3.0 | ?3.0 |
| Proteolytic enzyme (34g/#) | ?0.6 | ?0.6 | ?0.9 | ?0.9 | ?1.2 | ?1.2 |
| Lipase | ?0.1 | ?0.1 | ?0.1 | ?0.1 | ?0.2 | ?0.2 |
| Amylase (300KMU/g) | ?0.1 | ?0.1 | ?0.1 | ?0.1 | ?- | ?0.1 |
| Cellulase | ?0.03 | ?0.03 | ?0.05 | ?0.05 | ?0.2 | ?0.2 |
| ?Endolase | ?0.1 | ?0.1 | ?- | ?- | ?- | ?- |
| Whitening agent 2 | ?0.1 | ?0.1 | ?- | ?- | ?- | ?- |
| Boric acid | ?3.0 | ?3.0 | ?3.5 | ?3.5 | ?4.0 | ?4.0 |
| ?MEA | ?8.0 | ?4.0 | ?1.0 | ?1.5 | ?7.0 | ?7.0 |
| ?NaOH | ?1.0 | ?4.0 | ?3.0 | ?2.5 | ?1.0 | ?1.0 |
| ?PG | ?12.0 | ?12.0 | ?7.5 | ?7.5 | ?7.0 | ?7.0 |
| ?EtOH | ?1.0 | ?1.0 | ?3.5 | ?3.5 | ?6.0 | ?6.0 |
| ?NaTS | ?- | ?- | ?2.5 | ?2.5 | ?- | ?- |
| Minor materials | Surplus | Surplus | Surplus | Surplus | Surplus | Surplus |
The following example illustrates according to aqueous liquid detergent compositions of the present invention.
Embodiment 27
Listed water base heavy duty type liquid laundry detergent composition F~J below, described composition comprises the tensio-active agent of branching in the chain of the present invention.
Embodiment 28
| Component | F | ?G | ?H | ?I | ?J |
| ?MBAE1.8S14.4 | 10 | ?12 | ?14 | ?16 | ?20 |
| ?MLAS | 10 | ?8 | ?6 | ?4 | ?0 |
| ?C23E9 | 2 | ?2 | ?2 | ?2 | ?2 |
| ?LMFAA | 5 | ?5 | ?5 | ?5 | ?0 |
| The citric acid washing assistant | 3 | ?3 | ?3 | ?3 | ?5 |
| The lipid acid washing assistant | 2 | ?2 | ?2 | ?2 | ?0 |
| ?PAE | 1 | ?1 | ?1.2 | ?1.2 | ?0.5 |
| ?PG | 8 | ?8 | ?8 | ?8 | ?4.5 |
| ?EtOH | 4 | ?4 | ?4 | ?4 | ?2 |
| Boric acid | 3.5 | ?3.5 | ?3.5 | ?3.5 | ?2 |
| The isopropyl benzene sodium sulfonate | 3 | ?3 | ?3 | ?3 | ?0 |
| ?PH= | 8.0 | ?8.0 | ?8.0 | ?8.0 | ?7.0 |
| Enzyme, dyestuff, water | Surplus | Surplus | Surplus | Surplus | Surplus |
| 100% | ?100% | ?100% | ?100% | ?100% |
Following waterborne liquid laundry detergent composition K~O prepares according to the present invention.
Embodiment 29
| K | ?L | ?M | ?N | ?O | |
| ?MLAS | 1-7 | ?7-12 | ?12-17 | ?17-22 | ?1-35 |
| The arbitrary combination of following component: C25AExS*Na (x=1.0-2.5) C25AS (linear) to senior 2-alkyl | 15-21 | ?10-15 | ?5-10 | ?0-5 | ?0-25 |
| C14-17 NaPS C12-16 SAS C18 1,4-dithionate LAS C12-16 MES MBAE1.8S14.4 and/or MBAS14.4 | |||||
| ?LMFAA | ?0-3.5 | ?0-3.5 | ?0-3.5 | ?0-3.5 | ?0-8 |
| C23E9 or C23E6.5 | ?0-2 | ?0-2 | ?0-2 | ?0-2 | ?0-8 |
| ?APA | ?0.5 | ?0.5 | ?0.5 | ?0.5 | ?0.5-2 |
| Citric acid | ?5 | ?5 | ?5 | ?5 | ?0-8 |
| Lipid acid (TPK or C12/14) | ?2 | ?2 | ?2 | ?2 | ?0-14 |
| ?EtOH | ?4 | ?4 | ?4 | ?4 | ?0-8 |
| ?PG | ?6 | ?6 | ?6 | ?6 | ?0-10 |
| ?MEA | ?1 | ?1 | ?1 | ?1 | ?0-3 |
| ?NaOH | ?3 | ?3 | ?3 | ?3 | ?0-7 |
| ?NaTS | ?2.3 | ?2.3 | ?2.3 | ?2.3 | ?0-4 |
| Sodium formiate | ?0.1 | ?0.1 | ?0.1 | ?0.1 | ?0-1 |
| ?Borax | ?2.5 | ?2.5 | ?2.5 | ?2.5 | ?0-5 |
| Proteolytic enzyme | ?0.9 | ?0.9 | ?0.9 | ?0.9 | ?0-1.3 |
| Lipase | ?0.06 | ?0.06 | ?0.06 | ?0.06 | ?0-0.3 |
| Amylase | ?0.15 | ?0.15 | ?0.15 | ?0.15 | ?0-0.4 |
| Cellulase | ?0.05 | ?0.05 | ?0.05 | ?0.05 | ?0-0.2 |
| ?PAE | ?0-0.6 | ?0-0.6 | ?0-0.6 | ?0-0.6 | ?0-2.5 |
| ?PIE | ?1.2 | ?1.2 | ?1.2 | ?1.2 | ?0-2.5 |
| ?PAEC | ?0-0.4 | ?0-0.4 | ?0-0.4 | ?0-0.4 | ?0-2 |
| ?SRP?2 | ?0.2 | ?0.2 | ?0.2 | ?0.2 | ?0-0.5 |
| Whitening agent 1 or 2 | ?0.15 | ?0.15 | ?0.15 | ?0.15 | ?0-0.5 |
| The silicone antifoams | ?0.12 | ?0.12 | ?0.12 | ?0.12 | ?0-0.3 |
| The silicon-dioxide of fumigating | 0.0015 | ?0.0015 | ?0.0015 | ?0.0015 | ?0-0.003 |
| Flavouring agent | 0.3 | ?0.3 | ?0.3 | ?0.3 | ?0-0.6 |
| Dyestuff | 0.0013 | ?0.0013 | ?0.0013 | ?0.0013 | ?0-0.003 |
| Moisture/minor materials | Surplus | Surplus | Surplus | Surplus | Surplus |
| Product pH (10% deionized water solution) | 7.7 | ?7.7 | ?7.7 | ?7.7 | ?6-9.5 |
Following waterborne liquid laundry detergent composition P~T prepares according to the present invention.
Embodiment 30
| ?P | ?Q | ?R | ?S | ?T | |
| ?MLAS | ?2 | ?6.25 | ?10.5 | ?14.75 | ?19 |
| Any combination of following component: C25E1-3S C25AS C14-17 NaPS C12-16 SAS C18 Isosorbide-5-Nitrae-dithionate LAS C12-16 MES MBAE1S14.4 and/or MBAS14.4 | ?17 | ?12.75 | ?8.5 | ?4.25 | ?0 |
| ?LMFAA | ?1-5.5 | ?1-5.5 | ?1-5.5 | ?1-5.5 | ?1-5.5 |
| ?C23E9 | ?4-6 | ?4-6 | ?4-6 | ?4-6 | ?4-6 |
| ?APA | ?0-1.5 | ?0-1.5 | ?0-1.5 | ?0-1.5 | ?0-1.5 |
| Citric acid | ?1 | ?1 | ?1 | ?1 | ?1 |
| Lipid acid (TPK, C12/14) | ?7.5 | ?77.5 | ?7.5 | ?7.5 | ?7.5 |
| Lipid acid (RPS) | ?3.1 | ?3.1 | ?3.1 | ?3.1 | ?3.1 |
| ?EtOH | ?0-6 | ?0-6 | ?0-6 | ?0-6 | ?0-6 |
| ?PG | ?4-10 | ?4-10 | ?4-10 | ?4-10 | ?4-10 |
| ?MEA | ?3-8 | ?3-8 | ?3-8 | ?3-8 | ?3-8 |
| ?NaOH | ?1.5 | ?1.5 | ?1.5 | ?1.5 | ?1.5 |
| ?NaTS | ?0-2 | ?0-2 | ?0-2 | ?0-2 | ?0-2 |
| ?Borax | ?2-2.5 | ?2-2.5 | ?2-2.5 | ?2-2.5 | ?2-2.5 |
| ?CaCl 2 | ?0.02 | ?0.02 | ?0.02 | ?0.02 | ?0.02 |
| Proteolytic enzyme | ?0.3-1 | ?0.3-1 | ?0.3-1 | ?0.3-1 | ?0.3-1 |
| Lipase | ?0.05-0.3 | ?0.05-0.3 | ?0.05-0.3 | ?0.05-0.3 | ?0.05-0.3 |
| Amylase | ?0.05-0.5 | ?0.05-0.5 | ?0.05-0.5 | ?0.05-0.5 | ?0.05-0.5 |
| Cellulase | ?0.3 | ?0.3 | ?0.3 | ?0.3 | ?0.3 |
| ?PIE | ?1.2 | ?1.2 | ?1.2 | ?1.2 | ?0-2.5 |
| ?PAEC | ?0-0.4 | ?0-0.4 | ?0-0.4 | ?0-0.4 | ?0-2 |
| ?PAE | ?0.2-0.7 | ?0.2-0.7 | ?0.2-0.7 | ?0.2-0.7 | ?0.2-0.7 |
| ?SRP?3 | ?0.1-0.2 | ?0.1-0.2 | ?0.1-0.2 | ?0.1-0.2 | ?0.1-0.2 |
| Whitening agent 1 or 2 | ?0.15 | ?0.15 | ?0.15 | ?0.15 | ?0.15 |
| The silicone antifoams | ?0.2-0.25 | ?0.2-0.25 | ?0.2-0.25 | ?0.2-0.25 | ?0.2-0.25 |
| ?Isofol?16 | ?0-2 | ?0-2 | ?0-2 | ?0-2 | ?0-2 |
| Flavouring agent | ?0.5 | ?0.5 | ?0.5 | ?0.5 | ?0.5 |
| Moisture/minor materials | Surplus | Surplus | Surplus | Surplus | Surplus |
| PH (10% deionized water solution) | ?7.6 | ?7.6 | ?7.6 | ?7.6 | ?7.6 |
Listed water base heavy duty type liquid laundry detergent composition below, described composition comprises the tensio-active agent of branching in the chain of the present invention.
| Component | ??A | ?B | ?C | ??D | ??E |
| MLAS | ??10 | ?12 | ?14 | ??16 | ??20 |
| Na C25AES tensio-active agent | ??10 | ?8 | ?6 | ??4 | ??0 |
| The C23EO9 tensio-active agent | ??2 | ?2 | ?2 | ??2 | ??2 |
| The C12 methyl glucamine | ??5 | ?5 | ?5 | ??5 | ??0 |
| The citric acid washing assistant | ??3 | ?3 | ?3 | ??3 | ??5 |
| The lipid acid washing assistant | ??2 | ?2 | ?2 | ??2 | ??0 |
| Tetren b-oxide (15-18) | ??1 | ?1 | ?1.2 | ??1.2 | ??0.5 |
| Propylene glycol | ??8 | ?8 | ?8 | ??8 | ??4.5 |
| Ethanol | ??4 | ?4 | ?4 | ??4 | ??2 |
| Boric acid | ??3.5 | ?3.5 | ?3.5 | ??3.5 | ??2 |
| The isopropyl benzene sodium sulfonate | ??3 | ?3 | ?3 | ??3 | ??0 |
| PH= | ??8.0 | ?8.0 | ?8.0 | ??8.0 | ??7.0 |
| Enzyme, dyestuff, water | Surplus | Surplus | Surplus | Surplus | Surplus |
| ??100% | ?100% | ?100% | ??100% | ??100% |
Claims (29)
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP99815763 | 1999-12-15 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN1457358A true CN1457358A (en) | 2003-11-19 |
Family
ID=34178742
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN 99815763 Pending CN1457358A (en) | 1999-12-15 | 1999-12-15 | Aqueous heavy duty liquid detergent compositions comprising modified alkylbenzene sulfonates |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN1457358A (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102533462A (en) * | 2011-12-14 | 2012-07-04 | 中国日用化学工业研究院 | Low-temperature efficient liquid detergent composition and preparation process thereof |
| CN110903912A (en) * | 2019-11-29 | 2020-03-24 | 华东理工大学 | Aryl grease and preparation method and application thereof |
-
1999
- 1999-12-15 CN CN 99815763 patent/CN1457358A/en active Pending
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102533462A (en) * | 2011-12-14 | 2012-07-04 | 中国日用化学工业研究院 | Low-temperature efficient liquid detergent composition and preparation process thereof |
| CN102533462B (en) * | 2011-12-14 | 2013-10-30 | 中国日用化学工业研究院 | Low-temperature efficient liquid detergent composition and preparation process thereof |
| CN110903912A (en) * | 2019-11-29 | 2020-03-24 | 华东理工大学 | Aryl grease and preparation method and application thereof |
| CN110903912B (en) * | 2019-11-29 | 2022-01-28 | 华东理工大学 | Aryl grease and preparation method and application thereof |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN1168807C (en) | Improved alkylbenzenesulfonate surfactant compositions | |
| CN1211474C (en) | Deteragent composition containing mixtures of crystallinity-disrupted surfactants | |
| CN1165604C (en) | Mixed Surfactant System | |
| CN1331737A (en) | Laundry detergents comprising modified alkylbenzene sulfonates | |
| CN1411501A (en) | Laundry detergents comprising modified alkylbenzene sulfonates | |
| CN1120226C (en) | Polyoxyalkylene surfactants | |
| CN1101461C (en) | Mid-chain branched surfactants | |
| CN1243729C (en) | Medium-chain branched primary alkyl sulfates used as surfactants | |
| CN1270622A (en) | Improved alkylbenzenesulfonate surfactants | |
| CN1087341C (en) | Detergent compsns. containing selected mid-chain branched surfactants | |
| CN1177917C (en) | Medium chain branched primary alkyl alkoxylated sulfate surfactant | |
| CN1161448C (en) | Cleaning products containing improved alkylaryl sulfonate surfactants prepared from vinylidene olefins and methods for their preparation | |
| US6596680B2 (en) | Enhanced alkylbenzene surfactant mixture | |
| CN1225681A (en) | Detergent composition | |
| CN1281499A (en) | Granular detergent compositions comprising mid-chain branched surfactants | |
| CN1247561A (en) | Color-safe bleach boosters, compsns. and laundry methods employing same | |
| CN1192085C (en) | Oxoalcohol-based detergent | |
| CN1259992A (en) | Detergent particle | |
| CN1344310A (en) | Aqueous heavy duty liquid detergent composns comprising modified alkylbenzene sulfonates | |
| CN1322242A (en) | Detergent compositions | |
| CN1457358A (en) | Aqueous heavy duty liquid detergent compositions comprising modified alkylbenzene sulfonates | |
| CN1276004A (en) | Mid-chain branched surfactands with cellulose derivatives | |
| CA2358856A1 (en) | Aqueous heavy duty liquid detergent compositions comprising modified alkylbenzene sulfonates | |
| CN1069763A (en) | detergent composition | |
| CN1239986A (en) | Detergent composition containing isopeptidase |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C02 | Deemed withdrawal of patent application after publication (patent law 2001) | ||
| WD01 | Invention patent application deemed withdrawn after publication |