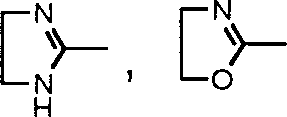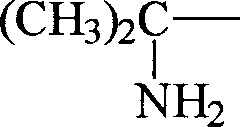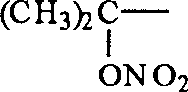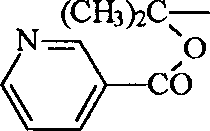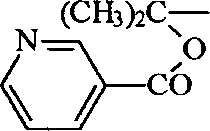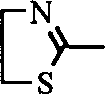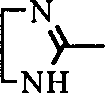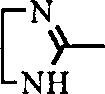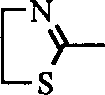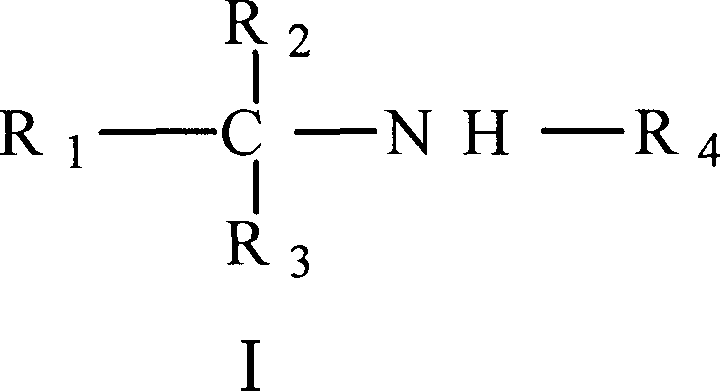CN1415596A - Amine derivative possessing the function of anti-pulmonary hypertension and its application in pharmacological science - Google Patents
Amine derivative possessing the function of anti-pulmonary hypertension and its application in pharmacological science Download PDFInfo
- Publication number
- CN1415596A CN1415596A CN 01137272 CN01137272A CN1415596A CN 1415596 A CN1415596 A CN 1415596A CN 01137272 CN01137272 CN 01137272 CN 01137272 A CN01137272 A CN 01137272A CN 1415596 A CN1415596 A CN 1415596A
- Authority
- CN
- China
- Prior art keywords
- alkyl
- dimethyl
- butylamine
- group
- methyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 150000001412 amines Chemical class 0.000 title claims abstract description 15
- 208000002815 pulmonary hypertension Diseases 0.000 title claims description 56
- 230000000144 pharmacologic effect Effects 0.000 title description 2
- 238000000034 method Methods 0.000 claims abstract description 43
- 150000003839 salts Chemical class 0.000 claims abstract description 29
- 150000001408 amides Chemical class 0.000 claims abstract description 28
- 239000003814 drug Substances 0.000 claims abstract description 26
- 230000003287 optical effect Effects 0.000 claims abstract description 16
- 239000000203 mixture Substances 0.000 claims abstract description 15
- 150000002148 esters Chemical class 0.000 claims abstract description 13
- 210000004072 lung Anatomy 0.000 claims abstract description 11
- 206010006458 Bronchitis chronic Diseases 0.000 claims abstract description 9
- 206010006451 bronchitis Diseases 0.000 claims abstract description 9
- 208000007451 chronic bronchitis Diseases 0.000 claims abstract description 9
- 208000008128 pulmonary tuberculosis Diseases 0.000 claims abstract description 8
- -1 isobutyl- Chemical group 0.000 claims description 77
- 239000002585 base Substances 0.000 claims description 69
- 150000001875 compounds Chemical class 0.000 claims description 53
- 238000002360 preparation method Methods 0.000 claims description 52
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Natural products CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 claims description 50
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 46
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 claims description 38
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical group OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 claims description 36
- 125000000217 alkyl group Chemical group 0.000 claims description 31
- 238000006467 substitution reaction Methods 0.000 claims description 28
- 238000006243 chemical reaction Methods 0.000 claims description 27
- 239000002253 acid Substances 0.000 claims description 26
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 24
- 229940124530 sulfonamide Drugs 0.000 claims description 22
- 150000003456 sulfonamides Chemical class 0.000 claims description 21
- 229920002554 vinyl polymer Polymers 0.000 claims description 21
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 claims description 15
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 claims description 15
- 239000003795 chemical substances by application Substances 0.000 claims description 15
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 claims description 15
- 230000002265 prevention Effects 0.000 claims description 15
- 150000003141 primary amines Chemical class 0.000 claims description 15
- 230000002685 pulmonary effect Effects 0.000 claims description 15
- 230000000694 effects Effects 0.000 claims description 14
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 14
- 125000006651 (C3-C20) cycloalkyl group Chemical group 0.000 claims description 12
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 claims description 12
- 239000004202 carbamide Substances 0.000 claims description 12
- 150000007524 organic acids Chemical class 0.000 claims description 11
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 claims description 9
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 claims description 9
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 claims description 9
- 239000003960 organic solvent Substances 0.000 claims description 9
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 9
- 229950004288 tosilate Drugs 0.000 claims description 9
- 208000004248 Familial Primary Pulmonary Hypertension Diseases 0.000 claims description 8
- AVXURJPOCDRRFD-UHFFFAOYSA-N Hydroxylamine Chemical compound ON AVXURJPOCDRRFD-UHFFFAOYSA-N 0.000 claims description 8
- 229920003171 Poly (ethylene oxide) Polymers 0.000 claims description 8
- 206010064911 Pulmonary arterial hypertension Diseases 0.000 claims description 8
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 claims description 8
- 208000006673 asthma Diseases 0.000 claims description 8
- 125000001924 fatty-acyl group Chemical group 0.000 claims description 8
- 125000000623 heterocyclic group Chemical group 0.000 claims description 8
- 208000030603 inherited susceptibility to asthma Diseases 0.000 claims description 8
- 201000008312 primary pulmonary hypertension Diseases 0.000 claims description 8
- RGWFHCMKBAMWDX-UHFFFAOYSA-N 2,3-dimethyl-n-propan-2-ylbutan-2-amine Chemical compound CC(C)NC(C)(C)C(C)C RGWFHCMKBAMWDX-UHFFFAOYSA-N 0.000 claims description 7
- 239000004215 Carbon black (E152) Substances 0.000 claims description 7
- 206010014561 Emphysema Diseases 0.000 claims description 7
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 claims description 7
- 125000000539 amino acid group Chemical group 0.000 claims description 7
- 201000009267 bronchiectasis Diseases 0.000 claims description 7
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 claims description 7
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 claims description 7
- 229930195733 hydrocarbon Natural products 0.000 claims description 7
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 claims description 7
- 230000000414 obstructive effect Effects 0.000 claims description 7
- 206010035653 pneumoconiosis Diseases 0.000 claims description 7
- 208000005069 pulmonary fibrosis Diseases 0.000 claims description 7
- 125000004423 acyloxy group Chemical group 0.000 claims description 6
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 6
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 claims description 6
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 6
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 claims description 6
- 150000002576 ketones Chemical class 0.000 claims description 6
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical class CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 claims description 6
- 239000008194 pharmaceutical composition Substances 0.000 claims description 6
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 6
- WSLDOOZREJYCGB-UHFFFAOYSA-N 1,2-Dichloroethane Chemical compound ClCCCl WSLDOOZREJYCGB-UHFFFAOYSA-N 0.000 claims description 5
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 claims description 5
- 235000005979 Citrus limon Nutrition 0.000 claims description 5
- XTHFKEDIFFGKHM-UHFFFAOYSA-N Dimethoxyethane Chemical compound COCCOC XTHFKEDIFFGKHM-UHFFFAOYSA-N 0.000 claims description 5
- FEWJPZIEWOKRBE-JCYAYHJZSA-L L-tartrate(2-) Chemical compound [O-]C(=O)[C@H](O)[C@@H](O)C([O-])=O FEWJPZIEWOKRBE-JCYAYHJZSA-L 0.000 claims description 5
- 150000001338 aliphatic hydrocarbons Chemical class 0.000 claims description 5
- 150000001336 alkenes Chemical class 0.000 claims description 5
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 claims description 5
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 5
- 125000002485 formyl group Chemical group [H]C(*)=O 0.000 claims description 5
- 229910052736 halogen Inorganic materials 0.000 claims description 5
- 150000002367 halogens Chemical class 0.000 claims description 5
- 230000007062 hydrolysis Effects 0.000 claims description 5
- 238000006460 hydrolysis reaction Methods 0.000 claims description 5
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 5
- 229940095064 tartrate Drugs 0.000 claims description 5
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 claims description 5
- 125000006700 (C1-C6) alkylthio group Chemical group 0.000 claims description 4
- RGHNJXZEOKUKBD-SQOUGZDYSA-M D-gluconate Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C([O-])=O RGHNJXZEOKUKBD-SQOUGZDYSA-M 0.000 claims description 4
- 125000003368 amide group Chemical group 0.000 claims description 4
- 239000003937 drug carrier Substances 0.000 claims description 4
- 229940050410 gluconate Drugs 0.000 claims description 4
- 238000005984 hydrogenation reaction Methods 0.000 claims description 4
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 4
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 claims description 4
- 239000003444 phase transfer catalyst Substances 0.000 claims description 4
- 150000003016 phosphoric acids Chemical class 0.000 claims description 4
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims description 4
- 108090000765 processed proteins & peptides Proteins 0.000 claims description 4
- 125000001424 substituent group Chemical group 0.000 claims description 4
- 229940086735 succinate Drugs 0.000 claims description 4
- KDYFGRWQOYBRFD-UHFFFAOYSA-L succinate(2-) Chemical compound [O-]C(=O)CCC([O-])=O KDYFGRWQOYBRFD-UHFFFAOYSA-L 0.000 claims description 4
- FMQLGQMULAQWOQ-AWEZNQCLSA-N (2s)-2-amino-n-(2,3-dimethylbutan-2-yl)-3-(1h-indol-3-yl)propanamide Chemical compound C1=CC=C2C(C[C@H](N)C(=O)NC(C)(C)C(C)C)=CNC2=C1 FMQLGQMULAQWOQ-AWEZNQCLSA-N 0.000 claims description 3
- UCYVIFOAPHCKDB-ZDUSSCGKSA-N (2s)-2-amino-n-(2,3-dimethylbutan-2-yl)-3-phenylpropanamide Chemical compound CC(C)C(C)(C)NC(=O)[C@@H](N)CC1=CC=CC=C1 UCYVIFOAPHCKDB-ZDUSSCGKSA-N 0.000 claims description 3
- DSGPOUCYSBVYPY-JTQLQIEISA-N (2s)-2-amino-n-(2,3-dimethylbutan-2-yl)-4-methylpentanamide Chemical compound CC(C)C[C@H](N)C(=O)NC(C)(C)C(C)C DSGPOUCYSBVYPY-JTQLQIEISA-N 0.000 claims description 3
- JTCBEHNQCCIATK-UWVGGRQHSA-N (2s,3s)-2-amino-n-(2,3-dimethylbutan-2-yl)-3-methylpentanamide Chemical compound CC[C@H](C)[C@H](N)C(=O)NC(C)(C)C(C)C JTCBEHNQCCIATK-UWVGGRQHSA-N 0.000 claims description 3
- KSGCCALQBFADCL-UHFFFAOYSA-N 2,3-dimethyl-3-(propan-2-ylamino)butan-2-ol Chemical compound CC(C)NC(C)(C)C(C)(C)O KSGCCALQBFADCL-UHFFFAOYSA-N 0.000 claims description 3
- VKGPKHSHOIYTCM-UHFFFAOYSA-N 2,3-dimethyl-N-prop-1-enylbutan-2-amine Chemical compound C(=CC)NC(C)(C(C)C)C VKGPKHSHOIYTCM-UHFFFAOYSA-N 0.000 claims description 3
- VZRJHJWQXVTUDR-UHFFFAOYSA-N 2,3-dimethyl-n-(2-methylpropyl)butan-2-amine Chemical compound CC(C)CNC(C)(C)C(C)C VZRJHJWQXVTUDR-UHFFFAOYSA-N 0.000 claims description 3
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 claims description 3
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 claims description 3
- 229910000564 Raney nickel Inorganic materials 0.000 claims description 3
- 125000002947 alkylene group Chemical group 0.000 claims description 3
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 claims description 3
- 239000003054 catalyst Substances 0.000 claims description 3
- YOCUPQPZWBBYIX-UHFFFAOYSA-N copper nickel Chemical compound [Ni].[Cu] YOCUPQPZWBBYIX-UHFFFAOYSA-N 0.000 claims description 3
- 125000006202 diisopropylaminoethyl group Chemical group [H]C([H])([H])C([H])(N(C([H])([H])C([H])([H])*)C([H])(C([H])([H])[H])C([H])([H])[H])C([H])([H])[H] 0.000 claims description 3
- 229940043278 dimethylamphetamine Drugs 0.000 claims description 3
- 239000012634 fragment Substances 0.000 claims description 3
- 229960001252 methamphetamine Drugs 0.000 claims description 3
- UKVIEHSSVKSQBA-UHFFFAOYSA-N methane;palladium Chemical group C.[Pd] UKVIEHSSVKSQBA-UHFFFAOYSA-N 0.000 claims description 3
- UVXNOYVRAWBANB-UHFFFAOYSA-N n,2,3-trimethylbutan-2-amine Chemical compound CNC(C)(C)C(C)C UVXNOYVRAWBANB-UHFFFAOYSA-N 0.000 claims description 3
- OQELTOXRXRBYOG-UHFFFAOYSA-N n-(2,3-dimethylbutan-2-yl)-3-phenyl-n-propan-2-ylprop-2-enamide Chemical compound CC(C)C(C)(C)N(C(C)C)C(=O)C=CC1=CC=CC=C1 OQELTOXRXRBYOG-UHFFFAOYSA-N 0.000 claims description 3
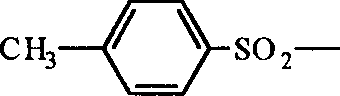
- YCUWQPPTJSOLPX-UHFFFAOYSA-N n-(2,3-dimethylbutan-2-yl)-4-methylbenzenesulfonamide Chemical compound CC(C)C(C)(C)NS(=O)(=O)C1=CC=C(C)C=C1 YCUWQPPTJSOLPX-UHFFFAOYSA-N 0.000 claims description 3
- TYNQIUJISCDNII-UHFFFAOYSA-N n-butyl-2,3-dimethylbutan-2-amine Chemical compound CCCCNC(C)(C)C(C)C TYNQIUJISCDNII-UHFFFAOYSA-N 0.000 claims description 3
- MUMZUERVLWJKNR-UHFFFAOYSA-N oxoplatinum Chemical compound [Pt]=O MUMZUERVLWJKNR-UHFFFAOYSA-N 0.000 claims description 3
- 229910003446 platinum oxide Inorganic materials 0.000 claims description 3
- 229920001184 polypeptide Polymers 0.000 claims description 3
- 102000004196 processed proteins & peptides Human genes 0.000 claims description 3
- 239000002879 Lewis base Substances 0.000 claims description 2
- FXHOOIRPVKKKFG-UHFFFAOYSA-N N,N-Dimethylacetamide Chemical compound CN(C)C(C)=O FXHOOIRPVKKKFG-UHFFFAOYSA-N 0.000 claims description 2
- CMEWLCATCRTSGF-UHFFFAOYSA-N N,N-dimethyl-4-nitrosoaniline Chemical compound CN(C)C1=CC=C(N=O)C=C1 CMEWLCATCRTSGF-UHFFFAOYSA-N 0.000 claims description 2
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 claims description 2
- 230000009471 action Effects 0.000 claims description 2
- 150000007527 lewis bases Chemical group 0.000 claims description 2
- 125000003944 tolyl group Chemical group 0.000 claims description 2
- 244000248349 Citrus limon Species 0.000 claims 2
- 125000001475 halogen functional group Chemical group 0.000 claims 2
- JISVIRFOSOKJIU-UHFFFAOYSA-N hexylidene Chemical group [CH2+]CCCC[CH-] JISVIRFOSOKJIU-UHFFFAOYSA-N 0.000 claims 1
- 125000004491 isohexyl group Chemical group C(CCC(C)C)* 0.000 claims 1
- 206010021143 Hypoxia Diseases 0.000 abstract description 19
- 229940079593 drug Drugs 0.000 abstract description 4
- 230000008569 process Effects 0.000 abstract description 2
- 206010002660 Anoxia Diseases 0.000 abstract 1
- 241000976983 Anoxia Species 0.000 abstract 1
- 230000007953 anoxia Effects 0.000 abstract 1
- 210000001367 artery Anatomy 0.000 abstract 1
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 68
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 34
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 33
- 235000002639 sodium chloride Nutrition 0.000 description 27
- 102400000686 Endothelin-1 Human genes 0.000 description 25
- 101800004490 Endothelin-1 Proteins 0.000 description 25
- 239000000243 solution Substances 0.000 description 25
- 150000003254 radicals Chemical class 0.000 description 20
- 238000005160 1H NMR spectroscopy Methods 0.000 description 18
- 210000001147 pulmonary artery Anatomy 0.000 description 18
- 239000007787 solid Substances 0.000 description 18
- 241000700159 Rattus Species 0.000 description 16
- 238000004458 analytical method Methods 0.000 description 16
- 230000004872 arterial blood pressure Effects 0.000 description 16
- 239000007788 liquid Substances 0.000 description 16
- YQNVPNRHJFZONU-UHFFFAOYSA-N 2,3-dimethylbutan-2-amine Chemical compound CC(C)C(C)(C)N YQNVPNRHJFZONU-UHFFFAOYSA-N 0.000 description 15
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 15
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 15
- 238000003756 stirring Methods 0.000 description 15
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 14
- 210000005241 right ventricle Anatomy 0.000 description 14
- 239000003921 oil Substances 0.000 description 13
- 235000019198 oils Nutrition 0.000 description 13
- CFHIDWOYWUOIHU-UHFFFAOYSA-N oxomethyl Chemical compound O=[CH] CFHIDWOYWUOIHU-UHFFFAOYSA-N 0.000 description 13
- 238000001035 drying Methods 0.000 description 12
- 238000000605 extraction Methods 0.000 description 12
- 229940125904 compound 1 Drugs 0.000 description 11
- OKHUDYOUNFDWEI-BGERDNNASA-N (2s)-2-amino-1-(2-diphenoxyphosphorylpyrrolidin-1-yl)-3-(1h-imidazol-5-yl)propan-1-one Chemical compound C([C@H](N)C(=O)N1C(CCC1)P(=O)(OC=1C=CC=CC=1)OC=1C=CC=CC=1)C1=CN=CN1 OKHUDYOUNFDWEI-BGERDNNASA-N 0.000 description 10
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 10
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 10
- 230000004071 biological effect Effects 0.000 description 9
- 229960000935 dehydrated alcohol Drugs 0.000 description 9
- 229960004756 ethanol Drugs 0.000 description 9
- AICOOMRHRUFYCM-ZRRPKQBOSA-N oxazine, 1 Chemical compound C([C@@H]1[C@H](C(C[C@]2(C)[C@@H]([C@H](C)N(C)C)[C@H](O)C[C@]21C)=O)CC1=CC2)C[C@H]1[C@@]1(C)[C@H]2N=C(C(C)C)OC1 AICOOMRHRUFYCM-ZRRPKQBOSA-N 0.000 description 9
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 8
- 230000001684 chronic effect Effects 0.000 description 8
- 238000001816 cooling Methods 0.000 description 8
- 230000007954 hypoxia Effects 0.000 description 8
- 239000002504 physiological saline solution Substances 0.000 description 8
- 239000003513 alkali Substances 0.000 description 7
- 208000018875 hypoxemia Diseases 0.000 description 7
- 229910000027 potassium carbonate Inorganic materials 0.000 description 7
- 239000002904 solvent Substances 0.000 description 7
- 239000003826 tablet Substances 0.000 description 7
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 6
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 6
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 6
- SMWDFEZZVXVKRB-UHFFFAOYSA-N Quinoline Chemical compound N1=CC=CC2=CC=CC=C21 SMWDFEZZVXVKRB-UHFFFAOYSA-N 0.000 description 6
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 6
- 229910052799 carbon Inorganic materials 0.000 description 6
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 6
- 238000004821 distillation Methods 0.000 description 6
- 229910052500 inorganic mineral Inorganic materials 0.000 description 6
- 239000002609 medium Substances 0.000 description 6
- 235000010755 mineral Nutrition 0.000 description 6
- 239000011707 mineral Substances 0.000 description 6
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 6
- 230000000050 nutritive effect Effects 0.000 description 6
- 238000001953 recrystallisation Methods 0.000 description 6
- 238000005406 washing Methods 0.000 description 6
- YLQBMQCUIZJEEH-UHFFFAOYSA-N Furan Chemical compound C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 5
- 238000006434 Ritter amidation reaction Methods 0.000 description 5
- 229920002472 Starch Polymers 0.000 description 5
- 238000009833 condensation Methods 0.000 description 5
- 239000007924 injection Substances 0.000 description 5
- 238000002347 injection Methods 0.000 description 5
- 239000012044 organic layer Substances 0.000 description 5
- 229910052760 oxygen Inorganic materials 0.000 description 5
- 239000000843 powder Substances 0.000 description 5
- 230000000069 prophylactic effect Effects 0.000 description 5
- 125000001501 propionyl group Chemical group O=C([*])C([H])([H])C([H])([H])[H] 0.000 description 5
- 235000019698 starch Nutrition 0.000 description 5
- 239000008107 starch Substances 0.000 description 5
- 230000001225 therapeutic effect Effects 0.000 description 5
- DSSYKIVIOFKYAU-XCBNKYQSSA-N (R)-camphor Chemical compound C1C[C@@]2(C)C(=O)C[C@@H]1C2(C)C DSSYKIVIOFKYAU-XCBNKYQSSA-N 0.000 description 4
- WNWHHMBRJJOGFJ-UHFFFAOYSA-N 16-methylheptadecan-1-ol Chemical compound CC(C)CCCCCCCCCCCCCCCO WNWHHMBRJJOGFJ-UHFFFAOYSA-N 0.000 description 4
- NPOAOTPXWNWTSH-UHFFFAOYSA-N 3-hydroxy-3-methylglutaric acid Chemical class OC(=O)CC(O)(C)CC(O)=O NPOAOTPXWNWTSH-UHFFFAOYSA-N 0.000 description 4
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 4
- NQRYJNQNLNOLGT-UHFFFAOYSA-N Piperidine Chemical compound C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 description 4
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 4
- 238000010521 absorption reaction Methods 0.000 description 4
- 150000001299 aldehydes Chemical class 0.000 description 4
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 4
- 210000004204 blood vessel Anatomy 0.000 description 4
- 229960000846 camphor Drugs 0.000 description 4
- 230000005494 condensation Effects 0.000 description 4
- 125000000753 cycloalkyl group Chemical group 0.000 description 4
- VYFYYTLLBUKUHU-UHFFFAOYSA-N dopamine Chemical compound NCCC1=CC=C(O)C(O)=C1 VYFYYTLLBUKUHU-UHFFFAOYSA-N 0.000 description 4
- 239000007789 gas Substances 0.000 description 4
- 239000008103 glucose Substances 0.000 description 4
- 235000001727 glucose Nutrition 0.000 description 4
- 235000011187 glycerol Nutrition 0.000 description 4
- 238000010438 heat treatment Methods 0.000 description 4
- 239000012280 lithium aluminium hydride Substances 0.000 description 4
- 239000001301 oxygen Substances 0.000 description 4
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 4
- 238000011160 research Methods 0.000 description 4
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 4
- 230000002792 vascular Effects 0.000 description 4
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 3
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 3
- 244000131522 Citrus pyriformis Species 0.000 description 3
- PVNIIMVLHYAWGP-UHFFFAOYSA-N Niacin Chemical compound OC(=O)C1=CC=CN=C1 PVNIIMVLHYAWGP-UHFFFAOYSA-N 0.000 description 3
- 241000219161 Theobroma Species 0.000 description 3
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 3
- 229960000583 acetic acid Drugs 0.000 description 3
- 150000001298 alcohols Chemical class 0.000 description 3
- 125000001118 alkylidene group Chemical group 0.000 description 3
- 125000003435 aroyl group Chemical group 0.000 description 3
- 239000011575 calcium Substances 0.000 description 3
- 235000001465 calcium Nutrition 0.000 description 3
- 229910052791 calcium Inorganic materials 0.000 description 3
- 239000002775 capsule Substances 0.000 description 3
- 230000006378 damage Effects 0.000 description 3
- 239000002552 dosage form Substances 0.000 description 3
- 150000002081 enamines Chemical class 0.000 description 3
- 238000002474 experimental method Methods 0.000 description 3
- 229920000159 gelatin Polymers 0.000 description 3
- 235000019322 gelatine Nutrition 0.000 description 3
- 239000012362 glacial acetic acid Substances 0.000 description 3
- 239000010410 layer Substances 0.000 description 3
- 229920000609 methyl cellulose Polymers 0.000 description 3
- 239000001923 methylcellulose Substances 0.000 description 3
- 229940023488 pill Drugs 0.000 description 3
- 239000006187 pill Substances 0.000 description 3
- 239000002994 raw material Substances 0.000 description 3
- 230000009467 reduction Effects 0.000 description 3
- 238000010992 reflux Methods 0.000 description 3
- 238000010025 steaming Methods 0.000 description 3
- 210000002784 stomach Anatomy 0.000 description 3
- RWRDLPDLKQPQOW-UHFFFAOYSA-N tetrahydropyrrole Natural products C1CCNC1 RWRDLPDLKQPQOW-UHFFFAOYSA-N 0.000 description 3
- SMYMJHWAQXWPDB-UHFFFAOYSA-N (2,4,5-trichlorophenoxy)acetic acid Chemical compound OC(=O)COC1=CC(Cl)=C(Cl)C=C1Cl SMYMJHWAQXWPDB-UHFFFAOYSA-N 0.000 description 2
- IKECULIHBUCAKR-UHFFFAOYSA-N 2,3-dimethylbutan-2-ol Chemical class CC(C)C(C)(C)O IKECULIHBUCAKR-UHFFFAOYSA-N 0.000 description 2
- MRWHUNCOQKBNKR-UHFFFAOYSA-N 2,3-dimethylbutan-2-ylurea Chemical compound CC(C)C(C)(C)NC(N)=O MRWHUNCOQKBNKR-UHFFFAOYSA-N 0.000 description 2
- 239000003559 2,4,5-trichlorophenoxyacetic acid Substances 0.000 description 2
- PKUPAJQAJXVUEK-UHFFFAOYSA-N 2-phenoxyacetyl chloride Chemical compound ClC(=O)COC1=CC=CC=C1 PKUPAJQAJXVUEK-UHFFFAOYSA-N 0.000 description 2
- SYBYTAAJFKOIEJ-UHFFFAOYSA-N 3-Methylbutan-2-one Chemical compound CC(C)C(C)=O SYBYTAAJFKOIEJ-UHFFFAOYSA-N 0.000 description 2
- RBOBXILUTBVQOQ-UHFFFAOYSA-N 3-methylbutan-2-ylazanium;chloride Chemical compound Cl.CC(C)C(C)N RBOBXILUTBVQOQ-UHFFFAOYSA-N 0.000 description 2
- VHYFNPMBLIVWCW-UHFFFAOYSA-N 4-Dimethylaminopyridine Chemical compound CN(C)C1=CC=NC=C1 VHYFNPMBLIVWCW-UHFFFAOYSA-N 0.000 description 2
- 229920001817 Agar Polymers 0.000 description 2
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 2
- 239000005995 Aluminium silicate Substances 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 2
- RGHNJXZEOKUKBD-SQOUGZDYSA-N D-gluconic acid Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C(O)=O RGHNJXZEOKUKBD-SQOUGZDYSA-N 0.000 description 2
- 239000004375 Dextrin Substances 0.000 description 2
- 229920001353 Dextrin Polymers 0.000 description 2
- 239000001856 Ethyl cellulose Substances 0.000 description 2
- ZZSNKZQZMQGXPY-UHFFFAOYSA-N Ethyl cellulose Chemical compound CCOCC1OC(OC)C(OCC)C(OCC)C1OC1C(O)C(O)C(OC)C(CO)O1 ZZSNKZQZMQGXPY-UHFFFAOYSA-N 0.000 description 2
- JOYRKODLDBILNP-UHFFFAOYSA-N Ethyl urethane Chemical compound CCOC(N)=O JOYRKODLDBILNP-UHFFFAOYSA-N 0.000 description 2
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 2
- 108010010803 Gelatin Proteins 0.000 description 2
- 206010020880 Hypertrophy Diseases 0.000 description 2
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 2
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 2
- 241001465754 Metazoa Species 0.000 description 2
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 description 2
- 241000283973 Oryctolagus cuniculus Species 0.000 description 2
- OFBQJSOFQDEBGM-UHFFFAOYSA-N Pentane Chemical compound CCCCC OFBQJSOFQDEBGM-UHFFFAOYSA-N 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 2
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 2
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 2
- 229930006000 Sucrose Natural products 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- 125000002252 acyl group Chemical group 0.000 description 2
- OIRDTQYFTABQOQ-KQYNXXCUSA-N adenosine Chemical compound C1=NC=2C(N)=NC=NC=2N1[C@@H]1O[C@H](CO)[C@@H](O)[C@H]1O OIRDTQYFTABQOQ-KQYNXXCUSA-N 0.000 description 2
- 239000008272 agar Substances 0.000 description 2
- 235000010419 agar Nutrition 0.000 description 2
- 238000013019 agitation Methods 0.000 description 2
- 235000010443 alginic acid Nutrition 0.000 description 2
- 229920000615 alginic acid Polymers 0.000 description 2
- 150000001335 aliphatic alkanes Chemical class 0.000 description 2
- 125000005360 alkyl sulfoxide group Chemical group 0.000 description 2
- 235000012211 aluminium silicate Nutrition 0.000 description 2
- 230000008485 antagonism Effects 0.000 description 2
- 210000000709 aorta Anatomy 0.000 description 2
- 125000003710 aryl alkyl group Chemical group 0.000 description 2
- 125000003118 aryl group Chemical group 0.000 description 2
- 239000012298 atmosphere Substances 0.000 description 2
- 150000001541 aziridines Chemical class 0.000 description 2
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 2
- 230000037396 body weight Effects 0.000 description 2
- 125000001246 bromo group Chemical group Br* 0.000 description 2
- HQABUPZFAYXKJW-UHFFFAOYSA-N butan-1-amine Chemical compound CCCCN HQABUPZFAYXKJW-UHFFFAOYSA-N 0.000 description 2
- OSGAYBCDTDRGGQ-UHFFFAOYSA-L calcium sulfate Chemical compound [Ca+2].[O-]S([O-])(=O)=O OSGAYBCDTDRGGQ-UHFFFAOYSA-L 0.000 description 2
- 238000009903 catalytic hydrogenation reaction Methods 0.000 description 2
- 239000003153 chemical reaction reagent Substances 0.000 description 2
- 210000000038 chest Anatomy 0.000 description 2
- 229940114081 cinnamate Drugs 0.000 description 2
- 235000015165 citric acid Nutrition 0.000 description 2
- 239000000470 constituent Substances 0.000 description 2
- 239000013078 crystal Substances 0.000 description 2
- 238000002425 crystallisation Methods 0.000 description 2
- 230000008025 crystallization Effects 0.000 description 2
- 230000006837 decompression Effects 0.000 description 2
- 239000003405 delayed action preparation Substances 0.000 description 2
- 235000019425 dextrin Nutrition 0.000 description 2
- 230000003205 diastolic effect Effects 0.000 description 2
- 235000014113 dietary fatty acids Nutrition 0.000 description 2
- CCAFPWNGIUBUSD-UHFFFAOYSA-N diethyl sulfoxide Chemical compound CCS(=O)CC CCAFPWNGIUBUSD-UHFFFAOYSA-N 0.000 description 2
- GGSUCNLOZRCGPQ-UHFFFAOYSA-N diethylaniline Chemical compound CCN(CC)C1=CC=CC=C1 GGSUCNLOZRCGPQ-UHFFFAOYSA-N 0.000 description 2
- FPAFDBFIGPHWGO-UHFFFAOYSA-N dioxosilane;oxomagnesium;hydrate Chemical compound O.[Mg]=O.[Mg]=O.[Mg]=O.O=[Si]=O.O=[Si]=O.O=[Si]=O.O=[Si]=O FPAFDBFIGPHWGO-UHFFFAOYSA-N 0.000 description 2
- 201000010099 disease Diseases 0.000 description 2
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 2
- SNRUBQQJIBEYMU-UHFFFAOYSA-N dodecane Chemical compound CCCCCCCCCCCC SNRUBQQJIBEYMU-UHFFFAOYSA-N 0.000 description 2
- 229960003638 dopamine Drugs 0.000 description 2
- 239000000839 emulsion Substances 0.000 description 2
- 235000019325 ethyl cellulose Nutrition 0.000 description 2
- 229920001249 ethyl cellulose Polymers 0.000 description 2
- 230000003203 everyday effect Effects 0.000 description 2
- 239000000194 fatty acid Substances 0.000 description 2
- 229930195729 fatty acid Natural products 0.000 description 2
- 239000008273 gelatin Substances 0.000 description 2
- 235000011852 gelatine desserts Nutrition 0.000 description 2
- 125000005843 halogen group Chemical group 0.000 description 2
- 235000012907 honey Nutrition 0.000 description 2
- IXCSERBJSXMMFS-UHFFFAOYSA-N hydrogen chloride Substances Cl.Cl IXCSERBJSXMMFS-UHFFFAOYSA-N 0.000 description 2
- 229910000041 hydrogen chloride Inorganic materials 0.000 description 2
- 230000001146 hypoxic effect Effects 0.000 description 2
- RAXXELZNTBOGNW-UHFFFAOYSA-N imidazole Natural products C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 description 2
- 150000002466 imines Chemical class 0.000 description 2
- 230000000622 irritating effect Effects 0.000 description 2
- NNPPMTNAJDCUHE-UHFFFAOYSA-N isobutane Chemical compound CC(C)C NNPPMTNAJDCUHE-UHFFFAOYSA-N 0.000 description 2
- JJWLVOIRVHMVIS-UHFFFAOYSA-N isopropylamine Chemical compound CC(C)N JJWLVOIRVHMVIS-UHFFFAOYSA-N 0.000 description 2
- 210000004731 jugular vein Anatomy 0.000 description 2
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 2
- 239000008101 lactose Substances 0.000 description 2
- 210000005240 left ventricle Anatomy 0.000 description 2
- 235000012204 lemonade/lime carbonate Nutrition 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Natural products C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 2
- 230000007935 neutral effect Effects 0.000 description 2
- 235000001968 nicotinic acid Nutrition 0.000 description 2
- 239000011664 nicotinic acid Substances 0.000 description 2
- 235000016709 nutrition Nutrition 0.000 description 2
- 230000035764 nutrition Effects 0.000 description 2
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 2
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 2
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 2
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 2
- FVSKHRXBFJPNKK-UHFFFAOYSA-N propionitrile Chemical compound CCC#N FVSKHRXBFJPNKK-UHFFFAOYSA-N 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 229910052708 sodium Inorganic materials 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- DAJSVUQLFFJUSX-UHFFFAOYSA-M sodium;dodecane-1-sulfonate Chemical compound [Na+].CCCCCCCCCCCCS([O-])(=O)=O DAJSVUQLFFJUSX-UHFFFAOYSA-M 0.000 description 2
- 239000000600 sorbitol Substances 0.000 description 2
- 239000005720 sucrose Substances 0.000 description 2
- 239000000829 suppository Substances 0.000 description 2
- CBDKQYKMCICBOF-UHFFFAOYSA-N thiazoline Chemical compound C1CN=CS1 CBDKQYKMCICBOF-UHFFFAOYSA-N 0.000 description 2
- FYSNRJHAOHDILO-UHFFFAOYSA-N thionyl chloride Chemical compound ClS(Cl)=O FYSNRJHAOHDILO-UHFFFAOYSA-N 0.000 description 2
- WBYWAXJHAXSJNI-VOTSOKGWSA-M trans-cinnamate Chemical compound [O-]C(=O)\C=C\C1=CC=CC=C1 WBYWAXJHAXSJNI-VOTSOKGWSA-M 0.000 description 2
- LWIHDJKSTIGBAC-UHFFFAOYSA-K tripotassium phosphate Chemical compound [K+].[K+].[K+].[O-]P([O-])([O-])=O LWIHDJKSTIGBAC-UHFFFAOYSA-K 0.000 description 2
- 230000002227 vasoactive effect Effects 0.000 description 2
- 229940124549 vasodilator Drugs 0.000 description 2
- 239000003071 vasodilator agent Substances 0.000 description 2
- 239000003981 vehicle Substances 0.000 description 2
- LNAZSHAWQACDHT-XIYTZBAFSA-N (2r,3r,4s,5r,6s)-4,5-dimethoxy-2-(methoxymethyl)-3-[(2s,3r,4s,5r,6r)-3,4,5-trimethoxy-6-(methoxymethyl)oxan-2-yl]oxy-6-[(2r,3r,4s,5r,6r)-4,5,6-trimethoxy-2-(methoxymethyl)oxan-3-yl]oxyoxane Chemical compound CO[C@@H]1[C@@H](OC)[C@H](OC)[C@@H](COC)O[C@H]1O[C@H]1[C@H](OC)[C@@H](OC)[C@H](O[C@H]2[C@@H]([C@@H](OC)[C@H](OC)O[C@@H]2COC)OC)O[C@@H]1COC LNAZSHAWQACDHT-XIYTZBAFSA-N 0.000 description 1
- NQBWNECTZUOWID-UHFFFAOYSA-N (E)-cinnamyl (E)-cinnamate Natural products C=1C=CC=CC=1C=CC(=O)OCC=CC1=CC=CC=C1 NQBWNECTZUOWID-UHFFFAOYSA-N 0.000 description 1
- DYLIWHYUXAJDOJ-OWOJBTEDSA-N (e)-4-(6-aminopurin-9-yl)but-2-en-1-ol Chemical compound NC1=NC=NC2=C1N=CN2C\C=C\CO DYLIWHYUXAJDOJ-OWOJBTEDSA-N 0.000 description 1
- IVVNZDGDKPTYHK-JTQLQIEISA-N 1-cyano-2-[(2s)-3,3-dimethylbutan-2-yl]-3-pyridin-4-ylguanidine Chemical compound CC(C)(C)[C@H](C)N=C(NC#N)NC1=CC=NC=C1 IVVNZDGDKPTYHK-JTQLQIEISA-N 0.000 description 1
- SWMMSULAOPNBRA-UHFFFAOYSA-N 2,2,3,5-tetramethylpyrrolidine Chemical compound CC1CC(C)C(C)(C)N1 SWMMSULAOPNBRA-UHFFFAOYSA-N 0.000 description 1
- PWOKJVJRVIFKLP-UHFFFAOYSA-N 2,2,3,6-tetramethylpiperidine Chemical compound CC1CCC(C)C(C)(C)N1 PWOKJVJRVIFKLP-UHFFFAOYSA-N 0.000 description 1
- IBQKGQDIZBMBGZ-UHFFFAOYSA-N 2,2,3,7-tetramethylazepane Chemical compound CC1(NC(CCCC1C)C)C IBQKGQDIZBMBGZ-UHFFFAOYSA-N 0.000 description 1
- SRQJMYDUKYXWSL-UHFFFAOYSA-N 2,3-dihydro-1h-pyrazole;quinoline Chemical compound C1NNC=C1.N1=CC=CC2=CC=CC=C21 SRQJMYDUKYXWSL-UHFFFAOYSA-N 0.000 description 1
- 125000001917 2,4-dinitrophenyl group Chemical group [H]C1=C([H])C(=C([H])C(=C1*)[N+]([O-])=O)[N+]([O-])=O 0.000 description 1
- 125000003456 2,6-dinitrophenyl group Chemical group [H]C1=C([H])C(=C(*)C(=C1[H])[N+]([O-])=O)[N+]([O-])=O 0.000 description 1
- UXFQFBNBSPQBJW-UHFFFAOYSA-N 2-amino-2-methylpropane-1,3-diol Chemical compound OCC(N)(C)CO UXFQFBNBSPQBJW-UHFFFAOYSA-N 0.000 description 1
- XGLHYBVJPSZXIF-UHFFFAOYSA-N 2-phenylbutan-2-ol Chemical class CCC(C)(O)C1=CC=CC=C1 XGLHYBVJPSZXIF-UHFFFAOYSA-N 0.000 description 1
- 125000003682 3-furyl group Chemical group O1C([H])=C([*])C([H])=C1[H] 0.000 description 1
- KZVSJCRPDWUPEP-UHFFFAOYSA-N 3-methyl-2-phenylbutan-2-ol Chemical class CC(C)C(C)(O)C1=CC=CC=C1 KZVSJCRPDWUPEP-UHFFFAOYSA-N 0.000 description 1
- JOZZAIIGWFLONA-UHFFFAOYSA-N 3-methylbutan-2-amine Chemical compound CC(C)C(C)N JOZZAIIGWFLONA-UHFFFAOYSA-N 0.000 description 1
- GZPHSAQLYPIAIN-UHFFFAOYSA-N 3-pyridinecarbonitrile Chemical compound N#CC1=CC=CN=C1 GZPHSAQLYPIAIN-UHFFFAOYSA-N 0.000 description 1
- 125000003349 3-pyridyl group Chemical group N1=C([H])C([*])=C([H])C([H])=C1[H] 0.000 description 1
- 125000001541 3-thienyl group Chemical group S1C([H])=C([*])C([H])=C1[H] 0.000 description 1
- 125000000339 4-pyridyl group Chemical group N1=C([H])C([H])=C([*])C([H])=C1[H] 0.000 description 1
- 244000215068 Acacia senegal Species 0.000 description 1
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical class N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 1
- XPSGESXVBSQZPL-SRVKXCTJSA-N Arg-Arg-Arg Chemical compound NC(N)=NCCC[C@H](N)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CCCN=C(N)N)C(O)=O XPSGESXVBSQZPL-SRVKXCTJSA-N 0.000 description 1
- NOWKCMXCCJGMRR-UHFFFAOYSA-N Aziridine Chemical compound C1CN1 NOWKCMXCCJGMRR-UHFFFAOYSA-N 0.000 description 1
- 239000002126 C01EB10 - Adenosine Substances 0.000 description 1
- 229940127291 Calcium channel antagonist Drugs 0.000 description 1
- ODINCKMPIJJUCX-UHFFFAOYSA-N Calcium oxide Chemical compound [Ca]=O ODINCKMPIJJUCX-UHFFFAOYSA-N 0.000 description 1
- JZUFKLXOESDKRF-UHFFFAOYSA-N Chlorothiazide Chemical compound C1=C(Cl)C(S(=O)(=O)N)=CC2=C1NCNS2(=O)=O JZUFKLXOESDKRF-UHFFFAOYSA-N 0.000 description 1
- FITPCXSHEGAMCJ-JJKGCWMISA-N ClC(=O)[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO.[Na] Chemical compound ClC(=O)[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO.[Na] FITPCXSHEGAMCJ-JJKGCWMISA-N 0.000 description 1
- 229940127007 Compound 39 Drugs 0.000 description 1
- XDTMQSROBMDMFD-UHFFFAOYSA-N Cyclohexane Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 description 1
- LVZWSLJZHVFIQJ-UHFFFAOYSA-N Cyclopropane Chemical compound C1CC1 LVZWSLJZHVFIQJ-UHFFFAOYSA-N 0.000 description 1
- OABOXRPGTFRBFZ-IMJSIDKUSA-N Cys-Cys Chemical compound SC[C@H](N)C(=O)N[C@@H](CS)C(O)=O OABOXRPGTFRBFZ-IMJSIDKUSA-N 0.000 description 1
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 1
- RGHNJXZEOKUKBD-UHFFFAOYSA-N D-gluconic acid Natural products OCC(O)C(O)C(O)C(O)C(O)=O RGHNJXZEOKUKBD-UHFFFAOYSA-N 0.000 description 1
- LTMHDMANZUZIPE-AMTYYWEZSA-N Digoxin Natural products O([C@H]1[C@H](C)O[C@H](O[C@@H]2C[C@@H]3[C@@](C)([C@@H]4[C@H]([C@]5(O)[C@](C)([C@H](O)C4)[C@H](C4=CC(=O)OC4)CC5)CC3)CC2)C[C@@H]1O)[C@H]1O[C@H](C)[C@@H](O[C@H]2O[C@@H](C)[C@H](O)[C@@H](O)C2)[C@@H](O)C1 LTMHDMANZUZIPE-AMTYYWEZSA-N 0.000 description 1
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 1
- 101100136092 Drosophila melanogaster peng gene Proteins 0.000 description 1
- 102000002045 Endothelin Human genes 0.000 description 1
- 108050009340 Endothelin Proteins 0.000 description 1
- 239000004593 Epoxy Substances 0.000 description 1
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical class C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 1
- 239000005977 Ethylene Substances 0.000 description 1
- 239000001828 Gelatine Substances 0.000 description 1
- 229920000084 Gum arabic Polymers 0.000 description 1
- 206010019280 Heart failures Diseases 0.000 description 1
- HTTJABKRGRZYRN-UHFFFAOYSA-N Heparin Chemical compound OC1C(NC(=O)C)C(O)OC(COS(O)(=O)=O)C1OC1C(OS(O)(=O)=O)C(O)C(OC2C(C(OS(O)(=O)=O)C(OC3C(C(O)C(O)C(O3)C(O)=O)OS(O)(=O)=O)C(CO)O2)NS(O)(=O)=O)C(C(O)=O)O1 HTTJABKRGRZYRN-UHFFFAOYSA-N 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 229920001543 Laminarin Polymers 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- 229930195725 Mannitol Natural products 0.000 description 1
- 229920000168 Microcrystalline cellulose Polymers 0.000 description 1
- DHVIZBBCXGKXDW-UHFFFAOYSA-N N-(1-bromoethyl)-N-propan-2-ylpropan-2-amine Chemical compound BrC(C)N(C(C)C)C(C)C DHVIZBBCXGKXDW-UHFFFAOYSA-N 0.000 description 1
- 240000007594 Oryza sativa Species 0.000 description 1
- 235000007164 Oryza sativa Nutrition 0.000 description 1
- 102000007079 Peptide Fragments Human genes 0.000 description 1
- 108010033276 Peptide Fragments Proteins 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- SSSFPISOZOLQNP-GUBZILKMSA-N Pro-Arg-Asp Chemical compound [H]N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(O)=O)C(O)=O SSSFPISOZOLQNP-GUBZILKMSA-N 0.000 description 1
- 241000700157 Rattus norvegicus Species 0.000 description 1
- 206010039163 Right ventricular failure Diseases 0.000 description 1
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 1
- 239000004141 Sodium laurylsulphate Substances 0.000 description 1
- 229920001615 Tragacanth Polymers 0.000 description 1
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical compound OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 1
- 206010047139 Vasoconstriction Diseases 0.000 description 1
- 208000027418 Wounds and injury Diseases 0.000 description 1
- 235000010489 acacia gum Nutrition 0.000 description 1
- 239000000205 acacia gum Substances 0.000 description 1
- 239000004480 active ingredient Substances 0.000 description 1
- 238000007259 addition reaction Methods 0.000 description 1
- 229960005305 adenosine Drugs 0.000 description 1
- 210000000577 adipose tissue Anatomy 0.000 description 1
- 239000000443 aerosol Substances 0.000 description 1
- 229940008126 aerosol Drugs 0.000 description 1
- 230000001476 alcoholic effect Effects 0.000 description 1
- 125000004390 alkyl sulfonyl group Chemical group 0.000 description 1
- BHELZAPQIKSEDF-UHFFFAOYSA-N allyl bromide Chemical compound BrCC=C BHELZAPQIKSEDF-UHFFFAOYSA-N 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- PZZYQPZGQPZBDN-UHFFFAOYSA-N aluminium silicate Chemical compound O=[Al]O[Si](=O)O[Al]=O PZZYQPZGQPZBDN-UHFFFAOYSA-N 0.000 description 1
- 229910000323 aluminium silicate Inorganic materials 0.000 description 1
- 230000002785 anti-thrombosis Effects 0.000 description 1
- 229960004676 antithrombotic agent Drugs 0.000 description 1
- 210000002376 aorta thoracic Anatomy 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 239000011260 aqueous acid Substances 0.000 description 1
- 239000012736 aqueous medium Substances 0.000 description 1
- 239000002635 aromatic organic solvent Substances 0.000 description 1
- 125000005002 aryl methyl group Chemical group 0.000 description 1
- 125000005361 aryl sulfoxide group Chemical group 0.000 description 1
- 239000000305 astragalus gummifer gum Substances 0.000 description 1
- 230000001746 atrial effect Effects 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- KGBXLFKZBHKPEV-UHFFFAOYSA-N boric acid Chemical compound OB(O)O KGBXLFKZBHKPEV-UHFFFAOYSA-N 0.000 description 1
- 239000004327 boric acid Substances 0.000 description 1
- 210000004556 brain Anatomy 0.000 description 1
- 230000031709 bromination Effects 0.000 description 1
- 238000005893 bromination reaction Methods 0.000 description 1
- 239000006189 buccal tablet Substances 0.000 description 1
- 229940046011 buccal tablet Drugs 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 239000000480 calcium channel blocker Substances 0.000 description 1
- 235000011132 calcium sulphate Nutrition 0.000 description 1
- 235000013877 carbamide Nutrition 0.000 description 1
- 150000004649 carbonic acid derivatives Chemical class 0.000 description 1
- 230000000747 cardiac effect Effects 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 230000003197 catalytic effect Effects 0.000 description 1
- 150000001805 chlorine compounds Chemical class 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- NQBWNECTZUOWID-QSYVVUFSSA-N cinnamyl cinnamate Chemical compound C=1C=CC=CC=1\C=C/C(=O)OC\C=C\C1=CC=CC=C1 NQBWNECTZUOWID-QSYVVUFSSA-N 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 229940125758 compound 15 Drugs 0.000 description 1
- 229940126086 compound 21 Drugs 0.000 description 1
- 229940125846 compound 25 Drugs 0.000 description 1
- 229940125851 compound 27 Drugs 0.000 description 1
- 229940125807 compound 37 Drugs 0.000 description 1
- 229940127573 compound 38 Drugs 0.000 description 1
- 229940126540 compound 41 Drugs 0.000 description 1
- 229940125936 compound 42 Drugs 0.000 description 1
- 229940125898 compound 5 Drugs 0.000 description 1
- 239000012141 concentrate Substances 0.000 description 1
- 235000008504 concentrate Nutrition 0.000 description 1
- 230000008602 contraction Effects 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 125000004122 cyclic group Chemical group 0.000 description 1
- 125000004367 cycloalkylaryl group Chemical group 0.000 description 1
- 125000000596 cyclohexenyl group Chemical group C1(=CCCCC1)* 0.000 description 1
- 125000004210 cyclohexylmethyl group Chemical group [H]C([H])(*)C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 125000002433 cyclopentenyl group Chemical group C1(=CCCC1)* 0.000 description 1
- 125000004851 cyclopentylmethyl group Chemical group C1(CCCC1)C* 0.000 description 1
- 125000004186 cyclopropylmethyl group Chemical group [H]C([H])(*)C1([H])C([H])([H])C1([H])[H] 0.000 description 1
- 108010004073 cysteinylcysteine Proteins 0.000 description 1
- DIOQZVSQGTUSAI-NJFSPNSNSA-N decane Chemical compound CCCCCCCCC[14CH3] DIOQZVSQGTUSAI-NJFSPNSNSA-N 0.000 description 1
- 230000002950 deficient Effects 0.000 description 1
- 230000018044 dehydration Effects 0.000 description 1
- 238000006297 dehydration reaction Methods 0.000 description 1
- 238000006356 dehydrogenation reaction Methods 0.000 description 1
- 238000010511 deprotection reaction Methods 0.000 description 1
- LTMHDMANZUZIPE-PUGKRICDSA-N digoxin Chemical compound C1[C@H](O)[C@H](O)[C@@H](C)O[C@H]1O[C@@H]1[C@@H](C)O[C@@H](O[C@@H]2[C@H](O[C@@H](O[C@@H]3C[C@@H]4[C@]([C@@H]5[C@H]([C@]6(CC[C@@H]([C@@]6(C)[C@H](O)C5)C=5COC(=O)C=5)O)CC4)(C)CC3)C[C@@H]2O)C)C[C@@H]1O LTMHDMANZUZIPE-PUGKRICDSA-N 0.000 description 1
- 229960005156 digoxin Drugs 0.000 description 1
- LTMHDMANZUZIPE-UHFFFAOYSA-N digoxine Natural products C1C(O)C(O)C(C)OC1OC1C(C)OC(OC2C(OC(OC3CC4C(C5C(C6(CCC(C6(C)C(O)C5)C=5COC(=O)C=5)O)CC4)(C)CC3)CC2O)C)CC1O LTMHDMANZUZIPE-UHFFFAOYSA-N 0.000 description 1
- 229960001760 dimethyl sulfoxide Drugs 0.000 description 1
- XHFGWHUWQXTGAT-UHFFFAOYSA-N dimethylamine hydrochloride Natural products CNC(C)C XHFGWHUWQXTGAT-UHFFFAOYSA-N 0.000 description 1
- XEYBHCRIKKKOSS-UHFFFAOYSA-N disodium;azanylidyneoxidanium;iron(2+);pentacyanide Chemical compound [Na+].[Na+].[Fe+2].N#[C-].N#[C-].N#[C-].N#[C-].N#[C-].[O+]#N XEYBHCRIKKKOSS-UHFFFAOYSA-N 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 239000002934 diuretic Substances 0.000 description 1
- 230000001882 diuretic effect Effects 0.000 description 1
- 239000008298 dragée Substances 0.000 description 1
- ZUBDGKVDJUIMQQ-UBFCDGJISA-N endothelin-1 Chemical compound C([C@@H](C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(O)=O)NC(=O)[C@H]1NC(=O)[C@H](CC=2C=CC=CC=2)NC(=O)[C@@H](CC=2C=CC(O)=CC=2)NC(=O)[C@H](C(C)C)NC(=O)[C@H]2CSSC[C@@H](C(N[C@H](CO)C(=O)N[C@@H](CO)C(=O)N[C@H](CC(C)C)C(=O)N[C@@H](CCSC)C(=O)N[C@H](CC(O)=O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(O)=O)C(=O)N2)=O)NC(=O)[C@@H](CO)NC(=O)[C@H](N)CSSC1)C1=CNC=N1 ZUBDGKVDJUIMQQ-UBFCDGJISA-N 0.000 description 1
- 239000003623 enhancer Substances 0.000 description 1
- KAQKFAOMNZTLHT-VVUHWYTRSA-N epoprostenol Chemical compound O1C(=CCCCC(O)=O)C[C@@H]2[C@@H](/C=C/[C@@H](O)CCCCC)[C@H](O)C[C@@H]21 KAQKFAOMNZTLHT-VVUHWYTRSA-N 0.000 description 1
- 229960001123 epoprostenol Drugs 0.000 description 1
- 235000019441 ethanol Nutrition 0.000 description 1
- LBAQSKZHMLAFHH-UHFFFAOYSA-N ethoxyethane;hydron;chloride Chemical compound Cl.CCOCC LBAQSKZHMLAFHH-UHFFFAOYSA-N 0.000 description 1
- 238000007046 ethoxylation reaction Methods 0.000 description 1
- IDNUEBSJWINEMI-UHFFFAOYSA-N ethyl nitrate Chemical compound CCO[N+]([O-])=O IDNUEBSJWINEMI-UHFFFAOYSA-N 0.000 description 1
- DEFVIWRASFVYLL-UHFFFAOYSA-N ethylene glycol bis(2-aminoethyl)tetraacetic acid Chemical compound OC(=O)CN(CC(O)=O)CCOCCOCCN(CC(O)=O)CC(O)=O DEFVIWRASFVYLL-UHFFFAOYSA-N 0.000 description 1
- 210000001105 femoral artery Anatomy 0.000 description 1
- 239000000174 gluconic acid Substances 0.000 description 1
- 235000012208 gluconic acid Nutrition 0.000 description 1
- 125000005908 glyceryl ester group Chemical group 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 230000001435 haemodynamic effect Effects 0.000 description 1
- 239000007902 hard capsule Substances 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 230000000004 hemodynamic effect Effects 0.000 description 1
- 229960002897 heparin Drugs 0.000 description 1
- 229920000669 heparin Polymers 0.000 description 1
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 150000002430 hydrocarbons Chemical class 0.000 description 1
- 229960002003 hydrochlorothiazide Drugs 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 239000008172 hydrogenated vegetable oil Substances 0.000 description 1
- NPZTUJOABDZTLV-UHFFFAOYSA-N hydroxybenzotriazole Substances O=C1C=CC=C2NNN=C12 NPZTUJOABDZTLV-UHFFFAOYSA-N 0.000 description 1
- 239000005457 ice water Substances 0.000 description 1
- 150000002460 imidazoles Chemical class 0.000 description 1
- 150000002462 imidazolines Chemical class 0.000 description 1
- 125000002636 imidazolinyl group Chemical group 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- 150000002475 indoles Chemical class 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 208000014674 injury Diseases 0.000 description 1
- 238000007912 intraperitoneal administration Methods 0.000 description 1
- 239000007928 intraperitoneal injection Substances 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 239000004310 lactic acid Substances 0.000 description 1
- 235000014655 lactic acid Nutrition 0.000 description 1
- DBTMGCOVALSLOR-VPNXCSTESA-N laminarin Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)OC1O[C@@H]1[C@@H](O)C(O[C@H]2[C@@H]([C@@H](CO)OC(O)[C@@H]2O)O)O[C@H](CO)[C@H]1O DBTMGCOVALSLOR-VPNXCSTESA-N 0.000 description 1
- 210000005246 left atrium Anatomy 0.000 description 1
- 239000002502 liposome Substances 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- IHLVCKWPAMTVTG-UHFFFAOYSA-N lithium;carbanide Chemical compound [Li+].[CH3-] IHLVCKWPAMTVTG-UHFFFAOYSA-N 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- 239000000594 mannitol Substances 0.000 description 1
- 235000010355 mannitol Nutrition 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 210000004379 membrane Anatomy 0.000 description 1
- 239000003094 microcapsule Substances 0.000 description 1
- 229940016286 microcrystalline cellulose Drugs 0.000 description 1
- 235000019813 microcrystalline cellulose Nutrition 0.000 description 1
- 239000008108 microcrystalline cellulose Substances 0.000 description 1
- 239000011259 mixed solution Substances 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 239000000178 monomer Substances 0.000 description 1
- 230000002969 morbid Effects 0.000 description 1
- 210000002200 mouth mucosa Anatomy 0.000 description 1
- 210000003205 muscle Anatomy 0.000 description 1
- DIOQZVSQGTUSAI-UHFFFAOYSA-N n-butylhexane Natural products CCCCCCCCCC DIOQZVSQGTUSAI-UHFFFAOYSA-N 0.000 description 1
- SQDFHQJTAWCFIB-UHFFFAOYSA-N n-methylidenehydroxylamine Chemical class ON=C SQDFHQJTAWCFIB-UHFFFAOYSA-N 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 210000003928 nasal cavity Anatomy 0.000 description 1
- 230000001537 neural effect Effects 0.000 description 1
- 229960003512 nicotinic acid Drugs 0.000 description 1
- HYIMSNHJOBLJNT-UHFFFAOYSA-N nifedipine Chemical compound COC(=O)C1=C(C)NC(C)=C(C(=O)OC)C1C1=CC=CC=C1[N+]([O-])=O HYIMSNHJOBLJNT-UHFFFAOYSA-N 0.000 description 1
- 229960001597 nifedipine Drugs 0.000 description 1
- 239000002840 nitric oxide donor Substances 0.000 description 1
- 150000002825 nitriles Chemical class 0.000 description 1
- LQNUZADURLCDLV-UHFFFAOYSA-N nitrobenzene Chemical compound [O-][N+](=O)C1=CC=CC=C1 LQNUZADURLCDLV-UHFFFAOYSA-N 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- ZCYXXKJEDCHMGH-UHFFFAOYSA-N nonane Chemical compound CCCC[CH]CCCC ZCYXXKJEDCHMGH-UHFFFAOYSA-N 0.000 description 1
- BKIMMITUMNQMOS-UHFFFAOYSA-N normal nonane Natural products CCCCCCCCC BKIMMITUMNQMOS-UHFFFAOYSA-N 0.000 description 1
- 230000000269 nucleophilic effect Effects 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 1
- TVMXDCGIABBOFY-UHFFFAOYSA-N octane Chemical compound CCCCCCCC TVMXDCGIABBOFY-UHFFFAOYSA-N 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 125000002524 organometallic group Chemical group 0.000 description 1
- 235000006408 oxalic acid Nutrition 0.000 description 1
- 125000000636 p-nitrophenyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)[N+]([O-])=O 0.000 description 1
- 238000012856 packing Methods 0.000 description 1
- 230000003950 pathogenic mechanism Effects 0.000 description 1
- 125000006340 pentafluoro ethyl group Chemical group FC(F)(F)C(F)(F)* 0.000 description 1
- DPBLXKKOBLCELK-UHFFFAOYSA-N pentan-1-amine Chemical compound CCCCCN DPBLXKKOBLCELK-UHFFFAOYSA-N 0.000 description 1
- MONRWRVYLOHUFA-UHFFFAOYSA-N pentylurea Chemical group CCCCCNC(N)=O MONRWRVYLOHUFA-UHFFFAOYSA-N 0.000 description 1
- 230000002085 persistent effect Effects 0.000 description 1
- 239000000825 pharmaceutical preparation Substances 0.000 description 1
- MRBDMNSDAVCSSF-UHFFFAOYSA-N phentolamine Chemical compound C1=CC(C)=CC=C1N(C=1C=C(O)C=CC=1)CC1=NCCN1 MRBDMNSDAVCSSF-UHFFFAOYSA-N 0.000 description 1
- 229960001999 phentolamine Drugs 0.000 description 1
- 229960002310 pinacidil Drugs 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- NNFCIKHAZHQZJG-UHFFFAOYSA-N potassium cyanide Chemical compound [K+].N#[C-] NNFCIKHAZHQZJG-UHFFFAOYSA-N 0.000 description 1
- 229940093916 potassium phosphate Drugs 0.000 description 1
- 229910000160 potassium phosphate Inorganic materials 0.000 description 1
- 235000011009 potassium phosphates Nutrition 0.000 description 1
- 230000036593 pulmonary vascular resistance Effects 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- 125000002755 pyrazolinyl group Chemical group 0.000 description 1
- 150000003242 quaternary ammonium salts Chemical class 0.000 description 1
- 230000000384 rearing effect Effects 0.000 description 1
- 239000003087 receptor blocking agent Substances 0.000 description 1
- 210000000664 rectum Anatomy 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 238000012827 research and development Methods 0.000 description 1
- 230000000284 resting effect Effects 0.000 description 1
- 235000009566 rice Nutrition 0.000 description 1
- 238000007142 ring opening reaction Methods 0.000 description 1
- 230000000630 rising effect Effects 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 150000003334 secondary amides Chemical class 0.000 description 1
- 150000003335 secondary amines Chemical class 0.000 description 1
- 238000010898 silica gel chromatography Methods 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- 235000012239 silicon dioxide Nutrition 0.000 description 1
- 229960001866 silicon dioxide Drugs 0.000 description 1
- 210000003491 skin Anatomy 0.000 description 1
- 239000002002 slurry Substances 0.000 description 1
- 150000003384 small molecules Chemical class 0.000 description 1
- 235000017557 sodium bicarbonate Nutrition 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- 235000019333 sodium laurylsulphate Nutrition 0.000 description 1
- 229940083618 sodium nitroprusside Drugs 0.000 description 1
- KKCBUQHMOMHUOY-UHFFFAOYSA-N sodium oxide Chemical compound [O-2].[Na+].[Na+] KKCBUQHMOMHUOY-UHFFFAOYSA-N 0.000 description 1
- 229910001948 sodium oxide Inorganic materials 0.000 description 1
- 239000007901 soft capsule Substances 0.000 description 1
- 235000013599 spices Nutrition 0.000 description 1
- 210000000278 spinal cord Anatomy 0.000 description 1
- 238000007920 subcutaneous administration Methods 0.000 description 1
- 125000003107 substituted aryl group Chemical group 0.000 description 1
- 235000000346 sugar Nutrition 0.000 description 1
- 239000007940 sugar coated tablet Substances 0.000 description 1
- 125000000565 sulfonamide group Chemical group 0.000 description 1
- 125000000472 sulfonyl group Chemical group *S(*)(=O)=O 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 125000002769 thiazolinyl group Chemical group 0.000 description 1
- 210000001519 tissue Anatomy 0.000 description 1
- 230000001256 tonic effect Effects 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 description 1
- DCXXMTOCNZCJGO-UHFFFAOYSA-N tristearoylglycerol Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCC(OC(=O)CCCCCCCCCCCCCCCCC)COC(=O)CCCCCCCCCCCCCCCCC DCXXMTOCNZCJGO-UHFFFAOYSA-N 0.000 description 1
- 229960004418 trolamine Drugs 0.000 description 1
- 230000025033 vasoconstriction Effects 0.000 description 1
- 230000002861 ventricular Effects 0.000 description 1
- 239000000052 vinegar Substances 0.000 description 1
- 235000021419 vinegar Nutrition 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- PJVWKTKQMONHTI-UHFFFAOYSA-N warfarin Chemical compound OC=1C2=CC=CC=C2OC(=O)C=1C(CC(=O)C)C1=CC=CC=C1 PJVWKTKQMONHTI-UHFFFAOYSA-N 0.000 description 1
- 229960005080 warfarin Drugs 0.000 description 1
- 230000003313 weakening effect Effects 0.000 description 1
- 230000003442 weekly effect Effects 0.000 description 1
- 238000005303 weighing Methods 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
Images
Landscapes
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
An amine derivative for resisting high pressure of lung artery, its isomer, racemate, optical isomer and salt for medicines, its amide or ester, the medical composition containing them, its preparing process, and its application in preparing medicines to prevent and cure anoxia, chronic bronchitis, pneumonectasis, pulmonary tuberculosis, etc are disclosed.
Description
The present invention relates to have sulfonamide derivatives, its steric isomer, its pharmaceutical salts of prevention or treatment pulmonary hypertension, its preparation method and in prevention or treatment by pulmonary hypertension due to anoxic, chronic bronchitis, obstructive emphysema, bronchiectasis, bronchial asthma, pulmonary tuberculosis, pneumoconiosis, pulmonary fibrosis, multiple pulmonary microembolization and the lung arteriolitis or the application in the agnogenic primary pulmonary hypertension.
Pulmonary hypertension is the major disease of serious harm human health, and along with the continuous progress of the state of an illness, pulmonary hypertension can cause lung, cardiac insufficiency and other organ injury, and final the appearance breathes and right heart failure.Pulmonary hypertension can significantly reduce patient's work capacity, and its disability rate is very high, brings heavy economical load for family and society.
In the modern medicine field, pulmonary hypertension still is a problem of failing to solve fully.Pulmonary hypertension mostly is nonspecific to patient's influence and the clinical symptom that pulmonary hypertension showed, thereby can not cause enough attention.People lack perfect understanding for the generation evolution and the fundamental mechanism thereof of pulmonary hypertension.The pathogeny complexity of pulmonary hypertension is not illustrated at present as yet fully, and existing pathomechanism comprises: (1) left atrium blood drainage is not smooth, makes the venous pressure rising and causes the Ppa pulmonary artery pressure passivity to raise; (2) with or can cause the lung vasoconstriction without acidosic alveolar anoxic, cause Ppa pulmonary artery pressure to raise.Persistent alveolar anoxic can cause the pulmonary artery pressure long term maintenance in high level, becomes pulmonary hypertension.(3) area of section of pulmonary vascular bed dwindles, and pulmonary vascular resistance increases, and causes pulmonary hypertension.The medicine for the treatment of pulmonary hypertension clinically comprises: cardiac tonic such as digoxin, diuretic(s) such as hydrochlorothiazide, alpha receptor blocking agent phentolamine, calcium antagonist, prostacyclin, NO donor sodium nitroprusside, adenosine and antithrombotics warfarin etc.All there is defective in various degree in these medicines, as: the toxicity that has is big; The general untoward reaction that has is many; The no definite clinical efficacy that has etc.In a word, there is not ideal to have the medicine of the accepted treatment pulmonary hypertension of definite curative effect as yet.Therefore, the newtype drug of research and development anti-pulmonary hypertension has crucial meaning.
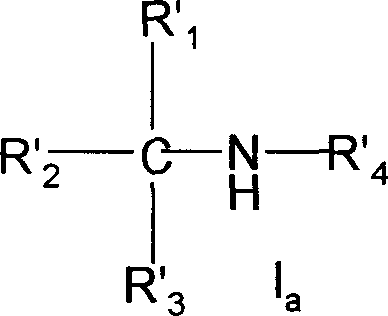
The application's general formula I
aSulfonamide derivatives and general formula I sulfonamide derivatives do not appear in the newspapers as yet in the purposes aspect prevention or the treatment pulmonary hypertension.
The objective of the invention is to seek and develop medicine, the especially prevention of new prevention or treatment pulmonary hypertension or treat by pulmonary hypertension or agnogenic primary pulmonary hypertension due to anoxic, chronic bronchitis, obstructive emphysema, bronchiectasis, bronchial asthma, pulmonary tuberculosis, pneumoconiosis, pulmonary fibrosis, multiple pulmonary microembolization and the lung arteriolitis.
The inventor has now found that the formula I with good potassium road regulating effect through research extensively and profoundly
aOr the sulfonamide derivatives shown in the formula I, this compounds can be used for prevention or treatment pulmonary hypertension.Studies show that formula I
aOr the ammonia derivative shown in the formula I has the effect of anti-pulmonary hypertension.Further synthetic with studies show that the pharmaceutical salts of the derivative that the present invention is included and suitable mineral acid or organic acid formation has the effect of anti-pulmonary hypertension equally.The present invention is based on above-mentioned discovery is accomplished.
First aspect present invention relates to the sulfonamide derivatives shown in the general formula I, its isomer, raceme or optical isomer, its medicinal acid addition salt, its acid amides or its ester and can be used for preventing or treat purposes in the medicine of pulmonary hypertension in preparation,
Wherein:
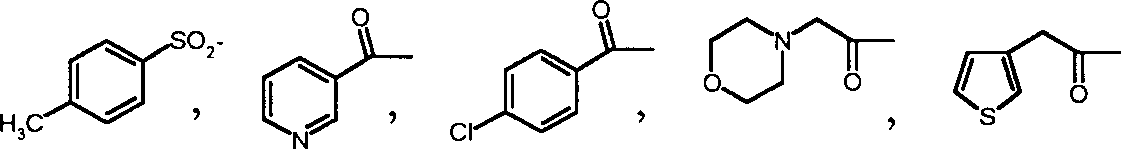
R
1, R
2, R
3Represent hydrogen atom, C respectively
1-20Saturated or undersaturated straight or branched aliphatic hydrocarbon, C
3-20Cycloalkyl group, replace C
3-20Cycloalkyl group, C
5-20Aryl radical, replace C
5-20Aryl radical, C
5-20Heterocycle alkyl, replacement C
5-20Heterocycle alkyl, Alpha-hydroxy C
2-20Alkyl, α-C
1-10Alkane carbonyl oxygen C
1-10Alkyl, C
6-14α-Fang carbonyl oxygen base C
1-10Alkyl, alpha-substitution C
6-14Virtue carbonyl oxygen base C
1-10Alkyl, α-C
1-10Alkoxy C
1-10Alkyl, alpha-substitution C
5-10Aryloxy C
1-10Alkyl, alpha-amino group C
1-20Alkyl, α-C
1-10Alkylamino radical C
1-10Alkyl, α-C
5-14Aryl amine C
1-10Alkyl, alpha-substitution C
5-14Aryl amine C
1-10Alkyl, α-C
1-10Alkyl amide C
1-10Alkyl, α-C
6-14Aromatic amide C
1-10Alkyl, alpha-substitution C
6-14Aromatic amide C
1-10Alkyl;
R
4Represent hydrogen atom, C
1-20Saturated fatty alkyl, C
5-20Aryl radical, replacement C
5-20Aryl radical, C
3-20Heterocycle alkyl, replacement C
3-20Heterocycle alkyl, C
3-20Heterocyclic radical, replacement C
3-20Heterocyclic radical, C
1-20Straight chain fatty acyl group, C
4-20Side chain fatty acyl group or and R
1, R
2, R
3The C that forms
3-20Cyclic hydrocarbon radical, C
3-20Heterocyclic radical, wherein said heterocycle refer to contain 1-3 and are selected from N, O or heteroatomic list of S or annelated heterocycles, and each substituting group that has substituent group is selected from: halogen, hydroxyl, cyano group, nitro, C
1-6Alkyl, C
1-6Alkoxyl group, C
1-6Alkylthio, one, two or three halo C
1-6Alkyl, amino, C
1-10The hydroxylamine base, C
1-10Alkylacyloxy, C
6-10Aryl acyloxy or C
1-10Amide group.
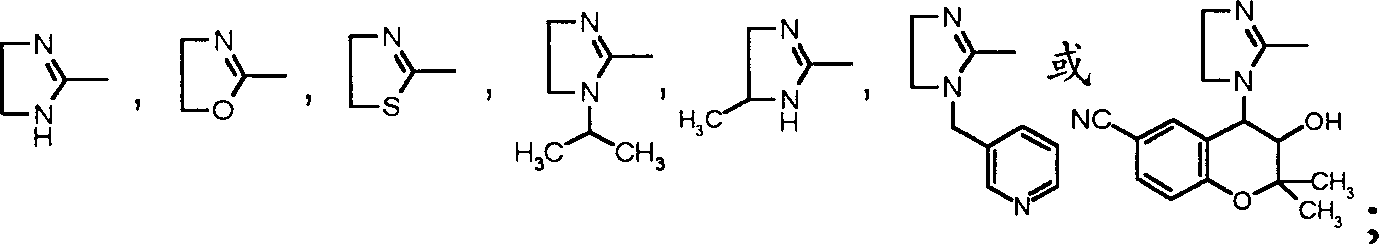
Second aspect present invention relates to the general formula I that is used to prevent or treat pulmonary hypertension
aShown sulfonamide derivatives, its isomer, raceme or optical isomer, medicinal acid addition salt, its acid amides or its ester,
Wherein:
(1). work as R '
1Be sec.-propyl, R '
2, R '
3During for methyl, R '
4Can be sec.-propyl, normal-butyl, isobutyl-, tertiary butyl, ring third methyl, dimethylamino ethyl, allyl group, diisopropylamino ethyl; Or
(2). work as R '
1, R '
2Be methyl, R '
3-C-NH-R '
4Can be the sulfonamide derivatives shown in the following formula, its
Isomer, raceme or optical isomer,
Wherein R and R ' are C
1-5Alkyl, n are the integer of 1-8; Or
(3). work as R '
1Be phenyl, R '
2During for methyl, R '
3Can be methyl, ethyl, sec.-propyl, R '
4Can be propyl group, methoxy carbonyl methyl; Or
(4). work as R '
1Be (CH
3)
2C (NH
2)-, R '
2, R '
3Be CH
3-time, R '
4Be (CH
3)
2CH-; Or
Work as R '
1Be (CH
3)
2C (OH)-, R '
2, R '
3Be CH
3-time, R '
4Be (CH
3)
2CH-or (CH
3)
2CH (CH
3)-; Or
Work as R '
1For
R '
2, R '
3Be CH
3-or R '
2With R '
3Be together-(CH
2)
4-or-(CH
2)
5-time, R '
4Be (CH
3)
2CH-; Or
Work as R '
1Be (CH
3)
2C (ONO
2)-, R '
2, R '
3Be CH
3-time, R '
4Be (CH
3)
2CH-; Or
Work as R '
1For
R '
2, R '
3Be CH
3-or R '
2With R '
3Be together-(CH
2)
4-or-(CH
2)
5-time, R '
4Be (CH
3)
2CH-; Or
Work as R '
1For
R '
2, R '
3Be CH
3-time, R '
4Be (CH
3)
2CH-or (CH
3)
2CH (CH
3)-; Or
Work as R '
1For
R '
2, R '
3For-(CH
2)
5-time, R '
4Be (CH
3)
2CCH (CH
3)-; Or
(6). work as R '
1Be (CH
3)
2CH-, R '
2, R '
3Be CH
3-time, R '
4Be Val-, Trp-, Ile-, Leu-, Phe-, O
2N-Arg-, Pro-, Leu-Val-, Trp-Trp-Trp-, (CH
3)
2CH-SO
2-, perhaps R '
4Be one of following group:
Or
Or
Work as R '
1Be cyclopropyl, R '
2And R '
3For-CH
2-CH
2-time, R '
4Be Val-; Or
Work as R '
1Be cyclohexyl, R '
2And R '
3Be CH
3-time, R '
4Be Pro-; Or
Work as R '
1Be cyclohexyl, R '
2And R '
3For-CH
2-CH
2-time, R '
4For Pro-or
Further aspect of the present invention relates to and is used to prevent or treats amine biology, its isomer, raceme or optical isomer, its medicinal acid addition salt, its acid amides or its ester shown in the general formula I of pulmonary hypertension
Wherein:
R
1, R
2, R
3Represent hydrogen atom, C respectively
1-20Saturated or undersaturated straight or branched aliphatic hydrocarbon, C
3-20Cycloalkyl group, replace C
3-20Cycloalkyl group, C
5-20Aryl radical, replace C
5-20Aryl radical, C
5-20Heterocycle alkyl, replacement C
5-20Heterocycle alkyl, Alpha-hydroxy C
2-20Alkyl, α-C
1-10Alkane carbonyl oxygen C
1-10Alkyl, C
6-14α-Fang carbonyl oxygen base C
1-10Alkyl, alpha-substitution C
6-14Virtue carbonyl oxygen base C
1-10Alkyl, α-C
1-10Alkoxy C
1-10Alkyl, alpha-substitution C
5-10Aryloxy C
1-10Alkyl, alpha-amino group C
1-20Alkyl, α-C
1-10Alkylamino radical C
1-10Alkyl, α-C
5-14Aryl amine C
1-10Alkyl, alpha-substitution C
5-14Aryl amine C
1-10Alkyl, α-C
1-10Alkyl amide C
1-10Alkyl, α-C
6-14Aromatic amide C
1-10Alkyl, alpha-substitution C
6-14Aromatic amide C
1-10Alkyl;
R
4Represent hydrogen atom, C
1-20Saturated fatty alkyl, C
5-20Aryl radical, replacement C
5-20Aryl radical, C
3-20Heterocycle alkyl, replacement C
3-20Heterocycle alkyl, C
3-20Heterocyclic radical, replacement C
3-20Heterocyclic radical, C
1-20Straight chain fatty acyl group, C
4-20Side chain fatty acyl group or and R
1, R
2, R
3The C that forms
3-20Cyclic hydrocarbon radical, C
3-20Heterocyclic radical, wherein said heterocycle refer to contain 1-3 and are selected from N, O or heteroatomic list of S or annelated heterocycles, and each substituting group that has substituent group is selected from: halogen, hydroxyl, cyano group, nitro, C
1-6Alkyl, C
1-6Alkoxyl group, C
1-6Alkylthio, one, two or three halo C
1-6Alkyl, amino, C
1-10The hydroxylamine base, C
1-10Alkylacyloxy, C
6-10Aryl acyloxy or C
1-10Amide group.
Further aspect of the present invention relates to pharmaceutical composition, and it contains at least a formula I
aOr formula I sulfonamide derivatives, its isomer, raceme or optical isomer, its medicinal acid addition salt, and pharmaceutical carrier or vehicle.
The invention still further relates to the method for prevention or treatment pulmonary hypertension, it comprises formula I or the formula I that the patient who suffers from pulmonary hypertension is treated significant quantity
aSulfonamide derivatives, its isomer, raceme or optical isomer, its medicinal acid addition salt.
The invention still further relates to the above-mentioned formula I of preparation
aThe method of compound is with primary amine R
1' R
2' R
3' CNH
2With R
4' X in organic solvent, through being heated to 50-300 ℃ and/or be pressurized to 0.1-20MPa and react, R wherein
1', R
2', R
3' and R
4' with as defined above, X is easy leavings group such as halogen, sulfonyloxy; Described being reflected under the catalyst action carried out, and described catalyzer is disacidify agent and/or phase-transfer catalyst, and wherein, described disacidify agent is that Lewis base comprises organic bases tertiary amine and mineral alkali, and phase-transfer catalyst is ethylene glycol or polyoxyethylene glycol; Described organic solvent is toluene, dimethylbenzene, 1,2-ethylene dichloride, 1,4-dioxane, glycol dimethyl ether, N, dinethylformamide, N,N-dimethylacetamide, N-Methyl pyrrolidone, N, accelerine or N, N-Diethyl Aniline.And primary amine R wherein
1' R
2' R
3' CNH
2Be with the same R of urea
1' R
2' R
3' the corresponding alkene of C or alcohol or the two mixture be heated to 20-200 ℃ of prepared in reaction alkyl urea in organic acid under vitriol oil effect; Then, hydrolysis prepares.Wherein, described organic acid is selected from acetate, trifluoroacetic acid or methylsulfonic acid.
The present invention also provides another kind of preparation described formula I
aThe method of compounds is with primary amine R
1' R
2' R
3' CNH
2With R
4' corresponding aldehydes or ketones is at catalyzer and have/organic solvent-free in the presence of, through being heated to 30-300 ℃ and/or be pressurized to 0.1-20MPa and carry out hydrogenation, R wherein
1', R
2', R
3' and R
4' as defined above; Be reflected under the catalyzer and carried out, wherein, described catalyzer is palladium-carbon, Raney's nickel, platinum oxide or nickel-copper etc., and described organic solvent is R
4' corresponding excessive aldehydes or ketones, toluene, dimethylbenzene, 1,2-ethylene dichloride, 1,4-dioxane, glycol dimethyl ether, methyl alcohol or ethanol.
The present invention also provides a kind of new preparation formula I
aThe method of amine compound, be with R
1' R
2' CNHR
4' corresponding enamine or Schiff alkali or its nitrone derivative and organometallic reagent R
3' M reacts; Perhaps, will with R
1' R
2' R
3' CNHR
4' corresponding enamine or Schiff alkali is through reduction or catalytic hydrogenation; Wherein, M is selected from the group that Li, Na, Mg, Al and Zn form.
Aforesaid method comprises that also the product that described reaction is obtained is through asymmetric reaction or the further step of making isomers or optical isomer that splits; Also comprise the resulting product of reaction and mineral acid or organic acid reaction, form pharmacy acceptable salt, i.e. mineral acid example hydrochloric acid, sulfuric acid, phosphoric acid and hydrobromic salt; Or organic acid salt, the i.e. salt of acetate, oxalic acid, citric acid, gluconic acid, succsinic acid, tartrate, tosic acid, methylsulfonic acid, phenylformic acid, lactic acid and toxilic acid.
According to the present invention, used term " pulmonary hypertension " is meant under the morbid state pulmonary hypertension or the agnogenic primary pulmonary hypertension due to for example anoxic, chronic bronchitis, obstructive emphysema, bronchiectasis, bronchial asthma, pulmonary tuberculosis, pneumoconiosis, pulmonary fibrosis, multiple pulmonary microembolization and the lung arteriolitis among the present invention.The compound of general formula I according to the present invention, R
1Be preferably the tertiary butyl, cyclohexyl, cyclopentyl, cyclobutyl, cyclopropyl, isopentyl, cyclobutyl; Perhaps, R
1Be cyclohexyl, cyclopentyl, cyclobutyl, cyclopropyl, dissident's base, isopentyl, isobutyl-, the sec.-propyl of alpha-substitution, wherein said substituting group is amino, hydroxyl, C
1-10Hydroxylamine base, C
1-6-oxyl, C
1-10Alkylacyloxy, C
6-10Aryl acyloxy, C
1-10Amide group;
R
2Or R
3Preferably be respectively hydrogen atom, C
1-12Chain alkylene or C
3-8Cyclic hydrocarbon radical;
R
4Be preferably hydrogen atom, C
1-20Saturated fatty alkyl, C
3-20Cycloalkyl, C
1-10Acyl group, C
1-10Hydroxylamine base C
1-10Alkyl, C
1-20Sulfoxide group, Amino acid residue and the micromolecule polypeptide fragment that combines thereof, β-nitroethylene base, beta-cyano vinyl, replacement carbonyl imido grpup, C
3-20Heterocyclic radical, and C
4-20Heterocyclic acyl.
The compound of general formula I according to the present invention, R
2, R
3More preferably be respectively methyl, ethyl, propyl group, perhaps, R
2And R
3Be propylidene, butylidene, pentylidene and inferior hexyl.
In a preferred embodiment of the invention, described R
1Be sec.-propyl, R
2Be methyl, R
3Be methyl.
According to the present invention, formula I
aCompound can be selected from a kind of in the group that following compound forms:
N-(1-methylethyl)-2,3-dimethyl-2-butylamine;
N-propyl group-2,3-dimethyl-2-butylamine;
N-(2-methyl-propyl)-2,3-dimethyl-2-butylamine;
N-encircles third methyl-2,3-dimethyl-2-butylamine;
N-propenyl-2,3-dimethyl-2-butylamine;
N-(2-(two-(1-methylethyl) amidos) ethyl)-2,3-dimethyl-2-butylamine;
N-butyl-2,3-dimethyl-2-butylamine;
N-propyl group-Alpha-Methyl amphetamine;
N-propyl group-α, the beta-dimethyl-amphetamine;
N-(3-pyridyl) formyl radical-2,3-dimethyl-2-butylamine;
N-is valyl-2,3-dimethyl-2-butylamine;
N-tryptophyl-2,3-dimethyl-2-butylamine;
N-(N-nitro) arginyl-2,3-dimethyl-2-butylamine;
N-phenylalanyl-2,3-dimethyl-2-butylamine;
N-leucyl-2,3-dimethyl-2-butylamine;
N-isoleucyl-2,3-dimethyl-2-butylamine;
N-p-toluenesulfonyl-2,3-dimethyl-2-butylamine;
N-(1-methylethyl)-2,3-dimethyl-3-hydroxyl-2-butylamine;
N-cinnamoyl-N-(1-methylethyl)-2,3-dimethyl-2-butylamine;
N-(1-methylethyl)-N-(2,4,5-trichlorobenzene oxygen ethanoyl)-2,3-dimethyl-2-butylamine.
According to the present invention, the acid salt of formula I compound says it is inorganic acid salt example hydrochloric acid salt, vitriol, phosphoric acid salt, hydrobromate for example; Or organic acid salt such as acetate, oxalate, lemon salt, gluconate, succinate, tartrate, tosilate, mesylate, benzoate, lactic acid salt and maleate; As N-(1-methylethyl)-2,3-dimethyl-2-butylamine tosilate.
The invention further relates to new being used to and prevent or treat the general formula I of pulmonary hypertension
aShown sulfonamide derivatives, its isomer, raceme or optical isomer, medicinal acid addition salt,
Wherein:
(1). work as R '
1Be sec.-propyl, R '
2, R '
3During for methyl, R '
4Can be sec.-propyl, normal-butyl, isobutyl-, tertiary butyl, ring third methyl, dimethylamino ethyl, allyl group, diisopropylamino ethyl; Or
(2). work as R '
1, R '
2Be methyl, R '
3-C-NH-R '
4Can be the sulfonamide derivatives shown in the following formula, its
Isomer, raceme or optical isomer,
Wherein R and R ' are C
1-5Alkyl, n are the integer of 1-8; Or
(3). work as R '
1Be phenyl, R '
2During for methyl, R '
3Can be methyl, ethyl, sec.-propyl, R '
4Can be propyl group, methoxy carbonyl methyl; Or
(4). work as R '
1Be (CH
3)
2C (NH
2)-, R '
2, R '
3Be CH
3-time, R '
4Be (CH
3)
2CH-; Or
Work as R '
1Be (CH
3)
2C (OH)-, R '
2, R '
3Be CH
3-time, R '
4Be (CH
3)
2CH-or (CH
3)
2CH (CH
3)-; Or
Work as R '
1For
R '
2, R '
3Be CH
3-or R '
2With R '
3Be together-(CH
2)
4-or-(CH
2)
5-time, R '
4Be (CH
3)
2CH-; Or
Work as R '
1Be (CH
3)
2C (ONO
2)-, R '
2, R '
3Be CH
3-time, R '
4Be (CH
3)
2CH-; Or
Work as R '
1For
R '
2, R '
3Be CH
3-or R '
2With R '
3Be together-(CH
2)
4-or-(CH
2)
5-time, R '
4Be (CH
3)
2CH-; Or
Work as R '
1For
R '
2, R '
3Be CH
3-time, R '
4Be (CH
3)
2CH-or (CH
3)
2CH (CH
3)-; Or
Work as R '
1For
R '
2, R '
3For-(CH
2)
5-time, R '
4Be (CH
3)
2CCH (CH
3)-; Or
(5). work as R '
1Be cyclohexyl, R '
2, R '
3Be CH
3-time, R '
4For
Or
Or
Work as R '
1Be (CH
3)
2CH-, R '
2, R '
3Be CH
3-time, R '
4For
Or
(6). work as R '
1Be (CH
3)
2CH-, R '
2, R '
3Be CH
3-time, R '
4Be Val-, Trp-, Ile-, Leu-, Phe-, O
2N-Arg-, Pro-, Leu-Val-, Trp-Trp-Trp-, (CH
3)
2CH-SO
2-, perhaps R '
4Be one of following group:
Or
Or
Work as R '
1Be cyclopropyl, R '
2And R '
3For-CH
2-CH
2-time, R '
4Be Val-; Or
Work as R '
1Be cyclohexyl, R '
2And R '
3Be CH
3-time, R '
4Be Pro-; Or
According to the present invention, formula I
aShown sulfonamide derivatives, its isomer, raceme or optical isomer, medicinal acid addition salt also have the effect that prevention or neural hypoxic-ischemic such as treatment brain and spinal cord damage.Its Chinese style I
aThe acid salt of sulfonamide derivatives says it is inorganic acid salt example hydrochloric acid salt, vitriol, phosphoric acid salt, hydrobromate for example; Or organic acid salt such as acetate, oxalate, lemon salt, gluconate, succinate, tartrate, tosilate, mesylate, benzoate, lactic acid salt, maleate, nicotinate, cinnamate or 3-hydroxy-3-methylglutaric acid salt.Preferred formula I
aThe hydrochloride of sulfonamide derivatives, maleate, tosilate, cinnamate and 3-hydroxy-3-methylglutaric acid salt.
More specifically say, in the general formula I derivative of the present invention, R
1, R
2And R
3Can be identical or different, represent hydrogen atom, C respectively
1-20Saturated or undersaturated straight or branched aliphatic hydrocarbon, C
3-20Cycloalkyl group, replace C
3-20Cycloalkyl group, C
5-20Aryl radical, replace C
5-20Aryl radical, C
5-20Heterocycle alkyl, replacement C
5-20Heterocycle alkyl, Alpha-hydroxy C
2-20Alkyl, α-C
1-10Alkane carbonyl oxygen C
1-10Alkyl, C
6-14α-Fang carbonyl oxygen base C
1-10Alkyl, alpha-substitution C
6-14Virtue carbonyl oxygen base C
1-10Alkyl, α-C
1-10Alkoxy C
1-10Alkyl, alpha-substitution C
5-10Aryloxy C
1-10Alkyl, alpha-amino group C
1-20Alkyl, α-C
1-10Alkylamino radical C
1-10Alkyl, α-C
5-14Aryl amine C
1-10Alkyl, alpha-substitution C
5-14Aryl amine C
1-10Alkyl, α-C
1-10Alkyl amide C
1-10Alkyl, α-C
6-14Aromatic amide C
1-10Alkyl, alpha-substitution C
6-14Aromatic amide C
1-10Alkyl;
R
4Representative: hydrogen atom, methyl, ethyl, n-propyl, sec.-propyl, substituted ring propyl group, substituted ring butyl, substituted ring amyl group, substituted cyclohexyl, cyclopropyl methyl, cyclopentyl-methyl, cyclohexyl methyl, α-(1-propenyl), 2-(1-butylene base), 3-(1-butylene base), cyclopentenyl, cyclohexenyl; Or
R
4For: aryl and substituted aryl such as phenyl, substituted-phenyl, ortho-nitrophenyl base, m-nitro base, p-nitrophenyl, 2,4-dinitrophenyl, 3,5-dinitrophenyl, 2,6-dinitrophenyl; Heterocyclic radical and substituted heterocyclic radical are as 4-pyridyl, 3-pyridyl, 3-furyl, 3-thienyl, 3-pyrryl, imidazolinyl, thiazolinyl, pyrazolinyl, dihydro miaow quinoline base, thiazoline quinoline base, pyrazoline quinoline base, N-substituted acyl pyrazolinyl, N-substituted acyl glyoxalidine quinoline base, N-substituted acyl pyrrolin quinoline base, imidazolyl, substituted imidazole base; The alpha-substitution arylalkyl is as alpha-substitution arylmethyl, alpha-substitution aryl ethyl, alpha-substitution arylpropyl, alpha-substitution aryl butyl and alpha-substitution cycloalkyl aryl; Or
R
4For: natural or alpha-non-natural amino acid residue replaces natural or alpha-non-natural amino acid residue, as Ala, Val, Leu, Ile, Phe, Asn, Glu, Trp, Tyr, Pro, Ser, Thr, Hyp, Cys, Met, Asp, Lys, Arg, His, O
2N-Arg; Natural or alpha-non-natural amino acid residue replaces little peptide fragment natural or that the alpha-non-natural amino acid residue is formed, as Cys-Cys, Arg-Arg-Arg, Pro-Arg-Asp etc.; Or
R
4For: aroyl, replacement aroyl; Sulfuryl is as alkyl sulfoxide base, aryl sulfoxide group, alkyl sulfuryl, aryl sulfuryl; The substituted ethylene base is as α-aryl amine-β-nitroethylene base, alpha-aromatic-beta-cyano vinyl; Replace the carbonyl imido grpup, as N-aryl-N '-nitro-carbonyl imido grpup, N-aryl-N '-nitro methyl-carbonyl imido grpup; Or
R
4For: alkyloyl, as propionyl, butyryl radicals and isobutyryl etc.; 4-hetaroylpyrazol, as 4-pyridine formyl radical, 3-pyrroles's ethanoyl etc., 3-indoles formyl radical, 3-indyl ethanoyl, 2-pyrroyl group and 3-pyrroles's ethanoyl etc.; Replace 4-hetaroylpyrazol, as 4-(2-nitro)-pyridine formyl radical, 3-(5-nitro) pyridine formyl radical, 3-(5-hydroxyl) indoles formyl radical, 3-(5-methoxyl group) indoles ethanoyl etc.; The alkylamino radical alkyloyl, as the dimethylin ethanoyl, the dimethylin propionyl, the diethylamine ethanoyl, the diethylin propionyl, two-(sec.-propyl) amido propionyls, 2-tropyl formyl radical, 2-tropine alkene formyl radical, 3-tropyl formyl radical, 3-tropine thiazolinyl formyl radical, N-piperazine formyl radical, N-benzoyl-1-piperazine formyl radical, 1-Pyrrolidine base formyl radical, 1-Pyrrolidine base ethanoyl, 1-Pyrrolidine base propionyl, 1-hexahydropyridine base formyl radical, 1-hexahydropyridine base ethanoyl, 1-tetrahydro pyridyl propionyl, 1-hexahydropyridine base formyl radical, 1-hexahydropyridine base ethanoyl, 1-hexahydropyridine base propionyl, two-(cyclohexyl) amido ethanoyl, two-(cyclohexyl) amido propionyls, 1-(4-hydroxyl) hexahydropyridine base ethanoyl, 1-(4-hydroxyl) hexahydropyridine base propionyl etc.; The alkyl sulfoxide base is as methyl sulfoxide base, ethyl-sulfoxide base, sec.-propyl sulfoxide group and N-morpholine ethyl-sulfoxide base etc.; The alkyl sulfuryl is as methyl sulfuryl, ethyl sulfuryl, sec.-propyl sulfone and N-morpholine ethyl sulfuryl etc.; The aryl sulfuryl is as phenyl sulfuryl-3-pyridine sulfuryl, 4-pyridyl ethyl sulfuryl and p-methylphenyl sulfuryl etc.; α-aryl amine-β-nitroethylene base, as α-(3-pyridyl) amido-β-nitroethylene base, α-(4-pyridyl) amido-β-nitroethylene base, α-(6-amino-3-pyridyl) amido-β-nitroethylene base, α-(3-oil of mirbane) amido-β-nitroethylene base, α-(3-carboxyl phenyl) amido-β-nitroethylene base, α-(3-cyano-phenyl) amido-β-nitroethylene base, α-(3-trifluoromethyl) amido-β-nitroethylene base, α-(3, the 4-dihalogenated phenyl) amido-β-nitroethylene base etc.; α-aryl amine-beta-cyano vinyl, as α-(3-pyridyl) amido-beta-cyano vinyl, α-(4-pyridyl) amido-beta-cyano vinyl, α-(6-amino-3-pyridyl) amido-beta-cyano vinyl, α-(3-oil of mirbane) amido-beta-cyano vinyl, α-(3-carboxyl phenyl) amido-beta-cyano vinyl, α-(3-cyano-phenyl) amido-beta-cyano vinyl, α-(3-trifluoromethyl) amido-beta-cyano vinyl and α-(3, the 4-dihalogenated phenyl) amido-beta-cyano vinyl etc.; N-aryl-N '-nitro carbon list imido grpup is as N-(3-pyridyl)-N '-nitro-N '-carbon list imido grpup, N-(3-nitrophenyl)-N '-nitro-N '-carbon list imido grpup and N-(3-halogenophenyl)-N '-nitro-N '-carbon list imido grpup etc.; N-aryl-N '-nitro methyl-carbon list imido grpup is as N-(3-pyridyl)-N '-nitro methyl-N '-carbon list imido grpup etc.; Alpha-aromatic-β-nitroethylene base, as α-(3-pyridyl)-β-nitroethylene base, α-(4-pyridyl)-β-nitroethylene base, α-(6-amino-3-pyridyl)-β-nitroethylene base, α-(4-nitropyridine base)-β-nitroethylene base, α-(3-cyano-phenyl)-β-nitroethylene base, α-(3, the 4-dihalogenated phenyl)-β-nitroethylene base etc., alpha-aromatic-beta-cyano vinyl, as α-(3-pyridyl)-beta-cyano vinyl, α-(4-pyridyl)-beta-cyano vinyl, α-(6-amino-3-pyridyl)-beta-cyano vinyl, α-(3-nitropyridine base)-beta-cyano vinyl, α-(3-cyano-phenyl)-beta-cyano vinyl, α-(3-trifluoromethyl)-beta-cyano vinyl, and α-(3, the 4-dihalogenated phenyl)-beta-cyano vinyl etc.; Or
R
4Be α-heterocyclic substituted-β-nitroethylene substituting group and α-heterocyclic substituted-beta-cyano ethene substituting group, wherein, heterocyclic substituent can for: the 2-position is 2,2-dimethyl, spirocyclopentyl or spiral shell cyclohexyl, 3, the 4-position is dehydrogenation or 3-hydroxyl, and the 6-position is for inhaling electric substituent 4-benzopyranyl, 4-pyrido pyranyl or 4-thieno-pyranyl; Wherein, 6-inhales electric substituting group and can be nitro, cyano group, trifluoromethyl, pentafluoroethyl group and alkylsulfonyl etc.
Work as R
1Be alkyl, cycloalkyl, alpha-aminoalkyl, alpha-amino group cycloalkyl or aryl, R
2And R
3All be alkyl or alkylidene group, R
4During for alkyl, alkylamino radical alkyl, alkylene, cycloalkyl, alkanoic acid ester or arylalkyl, preferred formula I compound-base is shown in Table 1.
The preferred formula I compound of table 1, in its Chinese style, each substituting group is alkyl compound R1 R
2 R
3 R
4Molecular formula 1 (CH3)
2CH- CH
3- CH
3- (CH
3)
2CH- C
9H
21N
2 (CH
3)
2CH- CH
3- CH
3- H- C
6H
15N
3 (CH
3)
2CH- CH
3- CH
3- CH
3- C
7H
17N
4 (CH
3)
2CH- CH
3- CH
3- C
2H
5- C
8H
19N
5 (CH
3)
2CH- CH
3- CH
3- CH
3CH
2CH
2- C
9H
21N
6 (CH
3)
2CH- CH
3- CH
3- n-C
4H
9- C
10H
23N
7 (CH
3)
2CH- CH
3- H- (CH
3)
2CH- C
8H
19N
8 H- H- H- (CH
3)
2CH- C
4H
11N
9 C
2H
5- CH
3- CH
3- (CH
3)
2CH- C
6H
15N
10 C
2H
5- CH
3- H- H- C
4H
11N
11 C
2H
5- CH
3- CH
3- CH
3- C
6H
15N
12 C
2H
5- CH
3- H- H- C
4H
11N
13 (CH
3)
2CH- CH
3- CH
3- (CH
3)
2CHCH
2- C
10H
23N
14 (CH
3)
2CH- CH
3- CH
3- t-C
4H
9- C
10H
23N
15 (CH
3)
2CH- CH
3- CH
3-
C
10H
21N
16 (CH
3)
2CH- CH
3- CH
3- (CH
3)
2NCH
2CH
2- C
10H
24N
2
17 Ph- CH
3- CH
3- CH
3OCOCH
2- C
12H
17NO
2
18 Ph- CH
3- C
2H
5- (CH
3)
2NCH
2CH
2- C
14H
24N
2
19 Ph- CH
3- (CH
3)
2CH- (CH
3)
2CH- C
14H
23N
20 Ph- CH
3- CH
3- CH
3CH
2CH
2- C
12H
19N
21 Ph- C
2H
5- CH
3- CH
3CH
2CH
2- C
13H
21N
22 Ph- (CH
3)
2CH- CH
3- CH
3CH
2CH
2- C
14H
23N
23 (CH
3)
2CH- CH
3- CH
3- CH
3OCOCH
2- C
9H
19NO
2
24 (CH
3)
2CH- CH
3- CH
3- PhCH
2- C
13H
21N
25 (CH
3)
2CH- CH
3- CH
3- CH
2=CH-CH
2- C
9H
19N
26 (CH
3)
2CH- CH
3- CH
3- (CH
3)
2CH-CH
2- C
10H
23N
27 (CH
3)
2CH- CH
3- CH
3- ((CH
3)
2CH)
2NCH
2CH
2- C
14H
32N
2
28
CH
3- CH
3- (CH
3)
2CH- C
12H
25N
29
CH
3- CH
3- (CH
3)
2NCH
2CH
2- C
13H
28N
2
30
CH
3- CH
3- PhCH
2- C
16H
25N
31
CH
3- CH
3- CH
3OCOCH
2- C
12H
23NO
2
32
-CH
2-CH
2- (CH
3)
2CH- C
9H
17N
33
-CH
2-CH
2- (CH
3)
2NCH
2CH
2- C
10H
20N
2
34
-CH
2-CH
2- PhCH
2- C
13H
17N
35
-CH
2-CH
2- CH
3OCOCH
2- C
9H
15NO
2
36
CH
3- CH
3- (CH
3)
2CH- C
9H
22N
2
Work as R
1Be alkyl, cycloalkyl, α-amido alkyl or α-amide group cycloalkyl, R
2And R
3All be alkyl and alkylidene group, R
4During for amino acid acyl, small molecules peptide chain, sulfonic acid acyl group, aroyl or heterocyclic acyl, preferred formula I compound sees Table 2.
The preferred formula I amine derivative of table 2, have the acyl substituent compound R in its Chinese style1 R
2 R
3 R
4Molecular formula 37 (CH3)
2CH- CH
3- CH
3- Val- C
11H
24N
2O
38 (CH
3)
2CH- CH
3- CH
3-
C
13H
21N
2OS
39 (CH
3)
2CH- CH
3- CH
3-
C
12H
28N
2O
40 (CH
3)
2CH- CH
3- CH
3- Trp- C
17H
25N
3O
41 (CH
3)
2CH- CH
3- CH
3- Ile- C
12H
26N
2O
42 (CH
3)
2CH- CH
3- CH
3- Leu- C
12H
26N
2O
43 (CH
3)
2CH- CH
3- CH
3- Phe- C
15H
24N
2O
44 (CH
3)
2CH- CH
3- CH
3- O
2N-Arg- C
12H
26N
6O
3
45 (CH
3)
2CH- CH
3- CH
3- Pro- C
11H
22N
2O
46 (CH
3)
2CH- CH
3- CH
3- Leu-Val- C
17H
35N
3O
2
47
-CH
2-CH
2- Val- C
11H
20N
2O
48 (CH
3)
2CH- CH
3- CH
3- Trp-Trp-Trp- C
39H
45N
7O
3
49
CH
3- CH
3- Pro- C
14H
26N
2O
50
-CH
2-CH
2- Pro- C
14H
24N
2O
51 (CH
3)
2CH- CH
3- CH
3- (CH
3)
2CHSO
2- C
9H
21N
2OS
52 (CH
3)
2CH- CH
3- CH
3-
C
13H
18ClNO
53 (CH
3)
2CH- CH
3- CH
3-
C
12H
24N
2O
2
54 (CH
3)
2CH- CH
3- CH
3-
C
12H
19NOS
55 (CH
3)
2CH- CH
3- CH
3-
C
16H
22N
2O
56
-CH
2-CH
2-
C
15H
20N
2O
57
CH
3- CH
3 (CH
3)
2CH- C
16H
26ClN
2O
R
2, R
3And R
4All be that alkyl or alkylidene group replace R
1When replacing for Alpha-hydroxy alkyl, Alpha-hydroxy cycloalkyl or its corresponding nitric ether, preferred substituted sees Table 3 for the formula I compound of alcohol or its ester.
The formula I compound compound R that alcohol or its ester are arranged in table 3 substituting group
1R
2R
3R
4Molecular formula 58
CH
3-CH
3-(CH
3)
2CH-C
9H
21NO59
CH
3-CH
3-
C
12H
27NO60
CH
3-CH
3-(CH
3)
2CH-C
12H
25NO61
-(CH
2)
5-(CH
3)
2CH-C
15H
29NO62
CH
3-CH
3-(CH
3)
2CH-C
11H
23NO63
-(CH
2)
4-(CH
3)
2CH-C
13H
25NO64
CH
3-CH
3-(CH
3)
2CH-C
9H
20N
2O
365
CH
3-CH
3-(CH
3)
2CH-C
12H
24N
2O
366
CH
3-CH
3-(CH
3)
2CH-C
11H
22N
2O
367
-(CH
2)
5-(CH
3)
2CH-C
15H
28N
2O
368
-(CH
2)
4-(CH
3)
2CH-C
13H
24N
2O
369
CH
3-CH
3-(CH
3)
2CH-C
15H
24N
2O
270
CH
3-CH
3-
C
18H
30N
2O
271
-(CH
2)
5-
C
24H
38N
2O
2
Work as R
1, R
2And R
3All be alkyl or cycloalkyl, R
4During for tetrahydroglyoxaline, thiazoline or substituted imidazoline, R
4For the formula I compound of heterocyclic radical sees Table 4.
Table 4 R
4Formula I compound compound R for heterocyclic substituent
1R
2R
3R
4Molecular formula 72 (CH
3)
2CH-CH
3-CH
3-
C
9H
19N
373 (CH
3)
2CH-CH
3-CH
3-
C
9H
18N
2O74 (CH
3)
2CH-CH
3-CH
3-
C
9H
18N
2S75
CH
3-CH
3-
C
12H
23N
376
CH
3-CH
3-
C
11H
21N
377
CH
3-CH
3-
C
12H
22N
2S78
CH
3-CH
3-
C
11H
20N
2S79
CH
3-CH
3-
C
12H
22N
2O80
CH
3-CH
3-
C
11H
20N
2O81 (CH
3)
2CH-CH
3-CH
3-
C
15H
22N
4O82 (CH
3)
2CH-CH
3-CH
3-
C
21H
30N
4O
283 (CH
3)
2CH-CH
3-CH
3-
C
12H
25N
384 (CH
3)
2CH-CH
3-CH
3-
C
10H
21N
3
Further, R
4Also can with R
1, R
2And R
3Constitute ring compound, such as the corresponding cyclic analogs of compound in table 1, table 2 and the table 3.As 2,2,3,5-tetramethyl-Pyrrolidine, 2,2,3,6-tetramethyl piperidine and 2,2,3,7-tetramethyl-azepan etc.
Further, formula I
aCompound also can with C
1-10Organic acid generates derivatives such as acid amides or ester, and preferred acid is nicotinic acid, styracin, toxilic acid, 2,4,5-trichlorophenoxyacetic acid and 3-hydroxy-3-methylglutaric acid.
According to the present invention, formula I
aThe preparation method's 1 of compound general conditions is generally: with suitable primary amine (R
1') (R
2') (R
3') CNH
2Same R
4' X is solvent-free or in containing hydrocarbon organic solvent, or aromatic organic solvents or alcoholic solvent, as cyclohexane, pentane, hexane, heptane, octane, nonane, decane, dodecane etc., benzene, toluene, dimethylbenzene, oil of mirbane and ethylene glycol, propylene glycol, glycerol etc., in the organic solvent, at catalyst-free or there is catalyzer to comprise various organic basess or mineral alkali and organic alcohols, as KOH, NaOH, pyridine, ethylene glycol, propylene glycol, glycerol, effect such as oligomeric ethylene glycol down, through heating and or pressurization to carry out the Heating temperature scope be 50~400 ℃, preferred range is 110 ℃~250 ℃.Be reflected in the autoclave and carry out, pressure is decided with adding solvent types and Heating temperature, perhaps charges into rare gas element, as N
2, gas such as He, Ar gas, pressure is generally at 0.1~20MPa in the reaction process, the preferred pressure scope is 0.5~15MPa.The purification process of target compound can be by general recrystallization method, and perhaps chromatography is separated.As needs, can be with general formula I
aCompound and mineral acid or organic acid reaction form suitable pharmaceutical salts.
According to the present invention, formula I
aThe preparation method's 2 of compound general condition is, with primary amine R
1' R
2' R
3' CNH
2With R
4' corresponding aldehydes or ketones is at catalyzer and have/organic solvent-free in the presence of, through being heated to 30-300 ℃ and/or be pressurized to 0.1-20MPa and carry out hydrogenation, R wherein
1', R
2', R
3' and R
4' as defined above; Be reflected under the catalyzer and carried out, wherein, described catalyzer is palladium-carbon, Raney's nickel, platinum oxide and nickel-copper, and described organic solvent is and R
4' corresponding excessive aldehydes or ketones, toluene, dimethylbenzene, 1,2-ethylene dichloride, 1,4-dioxane, glycol dimethyl ether, methyl alcohol and ethanol.
The preparation method of primary amine is with the same R of urea in aforesaid method
1' R
2' R
3' the corresponding alkene of C or alcohol or the two mixture be heated to 20-200 ℃ of reaction and form alkyl urea R in organic acid under vitriol oil effect
1' R
2' R
3' CNHCONH
2Then, this alkyl urea of hydrolysis prepares.Wherein, described organic acid is selected from acetate, trifluoroacetic acid or methylsulfonic acid.
In aforesaid method, when the compound of general formula I representative is an alkane secondary amine, secondary amide, and during secondary sulphonamide, can prepare by methods known in the art, referring to M.S.Dunn, B.M.Smart., Org.Synth., Coll.Vol.IV, 55 (1963); Houben-Weyl., XI/2,482; J.B.Hendrickson, R.Bergeron, Tetrahedron lett, 1973,3839; Also can again through reduction or catalytic hydrogenation, prepare secondary amine compound by imines or the Schiff alkali of forming earlier known in the art, referring to D.M.Balcom, C.R.Noller, Org.Synth., Coll Vol.IV, 603 (1963); Cesare Ferri, " Reaktionen der organischen Synthese ", Stuttgart, 1978, p85.The preparation of secondary amine compound still has special method, as with Schiff alkali or imines through the Grignard addition reaction, the secondary amine that preparation replaces, referring to Klusener P.A.A, Tipl and Brandsma, Tetrahedron 1991,2041; Klusener P.A.A, J.Chem.Soc., Chem.Commun, 1985,1677.
Secondary amine compound when the general formula I representative, mean that Alpha-hydroxy or alpha-amino group replace, or during the derivative of its replacement, can be raw material with epoxy compounds or ring tetrahydroform (Aziridine) derivative, with full amine through the nucleophilic ring opening prepared in reaction, referring to O.C.Dermer, G.E.Ham, Ethylenimine and other Aziridines, Academic Press, New York, 1969; L.B.Clapp, J.Amer.Chem.Soc 70,184 (1948).
In the compound of general formula I representative, R
4When representing the micromolecule polypeptide fragment that various Amino acid residues or its combine, can be by protection-condensation known in the art-deprotection or the preparation of its circulation method, referring to Ming Zhao, Chinese J.Med.Chem., 1995; 5 (2): 91, GuMingdi, Peng Shipi, Yu Xuemin, J.Chin.Pharm.Sci., 1993; 2 (2): 102.
R in the compound of general formula I representative
4During=H, be the primary amine raw material, its preparation method is generally: through the synthetic corresponding amide of Ritter reaction, hydrolysis gets then earlier.F.M.Furan and N.J.Somerville prepare the tertiary amyl urea method (US1972,3673249) of synthetic special amylamine then from corresponding alcohol.The present invention and then a kind of method for preparing the primary amine raw material is provided, that is, earlier with the same R of urea
1R
2R
3Corresponding alkene of C or alcohol or the mixture of the two are heated to 20-200 ℃ of prepared in reaction alkyl urea R in organic acid under vitriol oil effect
1R
2R
3CNHCONH
2Wherein, described organic acid is selected from acetate, trifluoroacetic acid or methylsulfonic acid etc.Then, hydrolysis makes corresponding primary amines under acid or alkali or other catalyst.
According to the present invention, formula I or formula I
aCompound can exist by stereoisomer form.The asymmetric center that exists in formula (I) compound can have S configuration or R configuration.The present invention includes all possible steric isomer such as enantiomorph or diastereomer, and the mixture of two or more steric isomers, for example mixture of any required ratio of enantiomorph and/or diastereomer.Therefore, the present invention relates to enantiomorph, for example left-handed-and the mixture or the racemoid of two kinds of enantiomorphs existing of dextrorotation-enantiomorph and different ratios that exists with enantiopure form.If there is suitable/trans isomer, the present invention relates to the mixture of cis form and trans forms and these forms.If desired, the preparation of single stereoisomers can split mixture according to conventional methods, or by for example synthetic preparation of three-dimensional selection.If there is the mobile hydrogen atom, the present invention also relates to the tautomeric form of formula I compound.
According to the present invention, formula I compound and steric isomer thereof show excellent results in prevention or treatment pulmonary hypertension.Therefore can be used as prevention or treatment primary pulmonary hypertension or, be preferred for Mammals, particularly the people because the medicine of the pulmonary hypertension that anoxic, chronic bronchitis, bronchial asthma etc. cause is used for animal.
Therefore the present invention also relates at least a formula I or the formula I that contains as the effective dose of active ingredient
aCompound, or the pharmaceutical composition of its pharmaceutical salts and/or its steric isomer and conventional medicine vehicle or assistant agent.Usually pharmaceutical composition of the present invention contains formula I or the formula I of 0.1-90 weight %
aCompound and/or its physiologically acceptable salt.Pharmaceutical composition can prepare according to methods known in the art.When being used for this purpose, if desired, can be with formula I or formula I
aCompound and/or steric isomer and one or more solids or liquid medicine vehicle and/or assistant agent combine, and make the suitable administration form or the dosage form that can be used as human.
Formula I of the present invention or formula I
aCompound or contain its pharmaceutical composition can the unit dosage form administration, route of administration can be enteron aisle or non-enteron aisle, as oral, muscle, subcutaneous, nasal cavity, oral mucosa, skin, peritonaeum or rectum etc.Form of administration is tablet, capsule, dripping pill, aerosol, pill, pulvis, solution, suspensoid, emulsion, granule, liposome, transdermal agent, buccal tablet, suppository, lyophilized injectable powder etc. for example.Can be ordinary preparation, sustained release preparation, controlled release preparation and various particulate delivery system.For the unit form of administration is made tablet, can be extensive use of various carrier well known in the art.Example about carrier is, for example thinner and absorption agent are as starch, dextrin, calcium sulfate, lactose, N.F,USP MANNITOL, sucrose, sodium-chlor, glucose, urea, lime carbonate, white bole, Microcrystalline Cellulose, pure aluminium silicate etc.; Wetting agent and tackiness agent are as water, glycerine, polyoxyethylene glycol, ethanol, propyl alcohol, starch slurry, dextrin, syrup, honey, glucose solution, mucialga of arabic gummy, gelatine size, Xylo-Mucine, lac, methylcellulose gum, potassiumphosphate, polyvinylpyrrolidone etc.; Disintegrating agent, for example dry starch, alginates, agar powder, laminaran, sodium bicarbonate and Citric Acid, lime carbonate, polyoxyethylene sorbitol fatty acid ester, sodium laurylsulfonate, methylcellulose gum, ethyl cellulose etc.; Disintegration inhibitor, for example sucrose, Tristearoylglycerol, theobroma oil, hydrogenation wet goods; Absorption enhancer, for example quaternary ammonium salt, sodium lauryl sulphate etc.; Lubricant, for example talcum powder, silicon-dioxide, W-Gum, stearate, boric acid, whiteruss, polyoxyethylene glycol etc.Tablet further can also be made coating tablet, for example sugar coated tablet, thin membrane coated tablet, ECT, or double-layer tablets and multilayer tablet.For pill is made in the administration unit, can be extensive use of various carrier well known in the art.Example about carrier is, for example thinner and absorption agent are as glucose, lactose, starch, theobroma oil, hydrogenated vegetable oil, polyvinylpyrrolidone, Gelucire, kaolin, talcum powder etc.; Tackiness agent such as gum arabic, tragacanth gum, gelatin, ethanol, honey, liquid sugar, rice paste or batter etc.; Disintegrating agent is as agar powder, dry starch, alginates, sodium laurylsulfonate, methylcellulose gum, ethyl cellulose etc.For suppository is made in the administration unit, can be extensive use of various carrier well known in the art.Example about carrier is, for example the ester of polyoxyethylene glycol, Yelkin TTS, theobroma oil, higher alcohols, higher alcohols, gelatin, semi-synthetic glyceryl ester etc.For capsule is made in the administration unit, with effective constituent formula I or formula I
aCompound or its steric isomer mix with above-mentioned various carriers, and the mixture that will obtain thus places hard obviously capsule or soft capsule.Also can be with effective constituent formula I or formula I
aCompound or its steric isomer are made microcapsule, are suspended in and form suspensoid in the aqueous medium, in the hard capsule of also can packing into or make injection and use.For injection preparation is made in the administration unit, as solution, emulsion, lyophilized injectable powder and suspensoid, can use this area all thinners commonly used, for example, water, ethanol, polyoxyethylene glycol, 1, the isooctadecanol of ammediol, ethoxylation, the isooctadecanol of polyoxyization, Polyoxyethylene Sorbitol Fatty Acid Esters etc.In addition, ooze injection liquid, can in injection preparation, add proper amount of sodium chloride, glucose or glycerine, in addition, can also add conventional solubility promoter, buffer reagent, pH regulator agent etc. in order to prepare etc.
In addition, as needs, also can in pharmaceutical preparation, add tinting material, sanitas, spices, correctives, sweeting agent or other material.
Formula I of the present invention or formula I
aCompound, or the dosage of its pharmaceutical salts or its steric isomer depends on many factors, for example will prevent or treat the character and the severity of disease, the sex of patient or animal, age, body weight and individual reaction, used particular compound, route of administration and administration number of times etc.Above-mentioned dosage can the single dose form or be divided into several, for example two, three or four dosage form administrations.
Embodiment
The present invention can be further specified by the following example, but these examples of implementation do not mean that any limitation of the invention.
Embodiment 1 N-(1-methylethyl)-2, the preparation of 3-dimethyl-2-butylamine-compound 1
Method one:
Get 7.6g (0.0745mol) 2,3-dimethyl-2-butanols, 32.4ml Glacial acetic acid, mixing.Bathe control with cryosel and make Nei Wenda-5 to-8 ℃, gradation adds powder solid potassium cyanide 7.3g (0.49mol), stirs.Slowly drip vitriol oil 32.4ml, temperature is below 20 ℃ in the control.Finish, in stirring 3.5 hours below 20 ℃, stirred 6 hours in room temperature again, room temperature is placed and is spent the night.Pour among the frozen water.Be neutralized to pH10 with 20% sodium hydroxide solution, use ether extraction 4 times, spend the night with anhydrous sodium sulfate drying.Filter next day, removes siccative, steams except that behind the ether, and the decompression base leaks, and collects bp105-108 ℃/5mmHg fraction, gets 8.8g, and productive rate 91.6% is N-(2-(2, the 3-dimethylbutyl) methane amide.
(2-(2, the 3-dimethylbutyl) methane amide after ethanol 6.2ml and water 51.6ml mix, adds concentrated hydrochloric acid 17.4ml, refluxes 4 hours in the oil bath to get 7.7g (0.0597mol) N-.After the decompression base removed ethanol, the ice bath cooling was alkalized to more than the pH12 with 40% sodium hydroxide solution down.Use ether extraction, the Anhydrous potassium carbonate drying.After reclaiming ether, air distillation is collected bp97-104 ℃ and is leaked branch, is total to 3.75g, productive rate 62.2%.Be 2,3-dimethyl-2-butylamine.
Get 10.6g (0.15mol) 2,3-dimethyl-2-butylamine, alkane in 6.45g (0.0524mol) bromo is different, 3.0ml ethylene glycol and 22.0ml toluene place the 50.0ml autoclave, and heat is 17 hours in 170 ℃ of stirrings.Finish, tell organic layer.Divide the extraction organic layer four times with 6N hydrochloric acid 60.0ml, merge acid solution, once with toluene wash.Ice bath down with 4% sodium hydroxide alkalize to pH be 12-13, use ether extraction, the Anhydrous potassium carbonate drying.After reclaiming ether, distillation is collected bp135-145 ℃ of fraction, productive rate 68.8%.Hydrochloride mp228-230 ℃ of (i-PrOH: Et
2O).Ultimate analysis C
9H
22ClN (%) calculated value C60.14, H12.34, N7.79, Cl19.73, measured value C60.14, H12.48, N7.31, Cl19.67.
1H-NMR(D
2O,ppm)0.98(d,J=6.75H,6H),1.33(S,6H),1.37(d,J=6.46,6H),2.10(m,1H),3.70(m,1H)。MS(m/z)143(M
+),100(B)。
Method two:
Getting 288ml glacial acetic acid and 412g (6.86mol) urea mixes, add 288g (3.43mol) 2,3-dimethyl-2-butylene, stir and drip the 412ml vitriol oil and 412ml glacial acetic acid mixed solution down, interior warm 45-50 ℃ of control is dripped and is finished, and stirs 5h in 50-55 ℃ again, placement is spent the night, and continues at 50-55 ℃ of reaction 7h.To the 8000ml frozen water that is dissolved with 1200g (30mol) sodium hydroxide, filter collection solid, 5 * 200ml water washing, drying gets the 404g white solid, mp175-176 ℃, is N-(2,3-dimethyl-2-butyl) urea, productive rate 81.8%.
1H-NMR(CDCl
3,ppm)0.88-0.91(d,6H,2×CH
3),1.26(s,6H,2×CH
3),2.20-2.26(m,1H,CH),4.45(br,2H),4.65(br,1H).MS(m/z)145.0?144.0(M
+)143.0,129.1,101.0,86.1,69.1,58.0(B)。Ultimate analysis C
7H
16N
2O (%) theoretical value C 58.30, H11.18, N 19.42; Measured value C 58.70, H 11.54, and N 19.25.
Get 118g (2.95mol) sodium hydroxide, be dissolved in 118ml water, cold slightly, be added in 196g (1.36mol) N-(2,3-dimethyl-2-butyl) urea and 392ml ethylene glycol or the trolamine, in 120 ℃ of oil baths, react 8h, 95-102 ℃ of fraction collected in air distillation, adds 75g Anhydrous potassium carbonate, 40g sodium hydroxide, redistillation, collecting 99-101 ℃ of fraction, get the 88.5g colourless liquid, is 2,3-dimethyl-2-butylamine, productive rate 64.3%.
1H-NMR(CDCl
3,ppm)0.88-0.91(d,6H,2×CH
3),1.04(s,6H,2×CH
3),1.53(m,1H,CH)。
Get 10.6g (0.15mol) 2,3-dimethyl-2-butylamine, alkane in 6.45g (0.0524mol) bromo is different, 3.0ml ethylene glycol and 22.0ml toluene place the 50.0ml autoclave, and heat is 17 hours in 170 ℃ of stirrings.Finish, tell organic layer.Divide the extraction organic layer four times with 6N hydrochloric acid 60.0ml, merge acid solution, once with toluene wash.Ice bath down with 4% sodium hydroxide alkalize to pH be 12-13, use ether extraction, the Anhydrous potassium carbonate drying.After reclaiming ether, distillation is collected bp135-145 ℃ of fraction, productive rate 68.8%.Hydrochloride mp228-230 ℃ of (i-PrOH: Et
2O).Ultimate analysis C
9H
22ClN (%) calculated value C 60.14, H 12.34, and N 7.79, and Cl 19.73, measured value C 60.14, H 12.48, and N 7.31, and Cl 19.67.
1H-NMR(D
2O,ppm)0.98(d,J=6.75H,6H),1.33(S,6H),1.37(d,J=6.46,6H),2.10(m,1H),3.70(m,1H)。MS(m/z)143(M
+),100(B)。
Method three:
Get the enamine that 0.1mol is made by methyl isopropyl Ketone and Isopropylamine dehydration, be dissolved in the 20ml hexane, logical N
2Under low temperature this solution is splashed into to containing in the 0.1mol lithium methide solution, ice bath stirs, and reaction is finished, and content is poured in the 0.5Kg frozen water, stirs.Water layer ether extraction secondary.Concentrate organic layer, add 3N hydrochloric acid, make pH value be lower than 1, reacted 10 minutes, with 10% sodium hydroxide alkalize to pH greater than 11.Ether extraction three times is used the Anhydrous potassium carbonate drying.Filter, underpressure distillation is collected bp140-145 ℃ of cut, productive rate 80%.
Embodiment 2 N-propyl group-2, the preparation of 3-dimethyl-2-butylamine-compound 5
Get the third clear 8.25g (0.15mol) and be dissolved among the ice vinegar liquid 25ml, slowly drip vitriol oil 15g for about 38 ℃, drip 2 again in interior temperature, 3-dimethyl-2-butanols 5.2g (0.051mol), interior temperature is no more than 40 ℃, the constant temperature whipping process.Pour into to frozen water,, use ether extraction with the alkalization of 40% sodium oxide.The combined ether layer washes with water once, uses anhydrous magnesium sulfate drying.Get light yellow liquid 6.2g after reclaiming ether, be dissolved among the anhydrous diethyl ether 80ml.This drips of solution is added among the anhydrous diethyl ether 80ml that contains lithium aluminum hydride 3.04g (0.08mol), refluxed again 10 hours.Cryosel cooling down, slowly drip 40% sodium hydroxide solution suitable in, carefully inclining upper strata ether liquid, with ether washing lower floor solid three times.Combined ether liquid is used the Anhydrous potassium carbonate drying.Filter, the cooling of external application ice bath, it is acid dripping anhydrous diethyl ether hydrogen chloride solution to solution.Filter collection solid is used Virahol and acetone recrystallization three times, gets white plates crystallization 3.33g, productive rate 46.33%, mp183-185 ℃.Ultimate analysis C
9H
22NCl (%) calculated value C 10.15, H 12.34, and N 7.79; Measured value C 60.20, H 13.80, and N 7.85; MS (m/z) 143 (M
+);
1H-NMR (D
2O, ppm) 0.98 (d, 6H), 1.00 (t, 3H), 1.24 (s, 6H), 1.63 (m, 2H), 2.05 (m, 1H), 2.98 (t, 2H).
Embodiment 3 N-(1-methyl-propyl)-2, the preparation of 3-dimethyl-2-butylamine-compound 13
Press the method for example example 1.Get 2,3-dimethyl-2-butylamine and bromination Trimethylmethane prepared in reaction.Productive rate 17.1%, hydrochloride mp203-204 ℃.Ultimate analysis C
10H
14NCl (%) calculated value C61.99, H1249, N7.23, measured value C62.17, H13.18, N7.27; MS (m/z) 157 (M
+);
1H-NMR (D
2O, ppm) 0.90 (d, 6H), 1.18 (t, 3H), 1.26 (d, 3H), 1.28-3.43 (m, 19H).
Embodiment 4 N-encircle third methyl-2, the preparation of 3-dimethyl-2-butylamine-compound 15
Press the method for embodiment 1, get 2,3-dimethyl-2-butylamine and brooethyl cyclopropane prepared in reaction.Productive rate 27.6%, hydrochloride mp176-178 ℃.Ultimate analysis C
10H
11NCl (%) calculated value C 62.64, H 11.57, and N 7.31, measured value C 62.69, H 11.82, N 7.01.MS (m/z) 155 (M
+), 112 (M
+-43);
1H-NMR (D
2O, ppm) 0.95 (d, 6H), 1.30-3.10 (m, 14H), 5.20 (m, 1H).
Embodiment 5 N-allyl groups-2, the preparation of 3-dimethyl-2-butylamine-compound 25
Press the method for embodiment 1, get 2,3-dimethyl-2-butylamine and allyl bromide 98 prepared in reaction.Productive rate 79.3%, hydrochloride mp173-175 ℃.Ultimate analysis C9H20NCl (%) calculated value C 60.80, H 11.34, and N 7.88, measured value C 60.68, H 11.43, N 7.94.MS (m/z) 141 (M
+);
1H-NMR (D
2O) 0.98 (d, 6H), 1.31 (s, 6H), 2.20 (m, 1H), 3.66 (d, 2H), 5.87 (m, 2H), 5.95 (m, 1H).
Embodiment 6 N-(2-(two (1-methylethyl) amido) ethyl)-2, the preparation of 3-dimethyl-2-butylamine-compound 27
Press the method for embodiment 1, get 2,3-dimethyl-2-butylamine and 2-(diisopropylamino) monobromoethane prepared in reaction.Productive rate 31.8%, hydrochloride mp176-178 ℃.Ultimate analysis C
14H
34N
2Cl
2(%) calculated value C55.8, H 11.37, and N 9.3, measured value C55.90, H11.68, N9.21.MS(m/z)229(M
+);
1H-NMR(D
2O,ppm)1.01(d,6H),1.38(s,6H),1.40(d,12H),2.04(m,1H),3.39-3.83(m,6H)。
Embodiment 7 N-butyl-2, the preparation of 3-dimethyl-2-butylamine-compound 6
Press the method for embodiment 2, get positive fourth table nitrile and 2,3-dimethyl-2-butanols carries out the Ritter reaction, and the preparation amide intermediate is again with lithium aluminium hydride reduction preparation, productive rate 26.1%.Hydrochloride mp140-142 ℃, ultimate analysis C
10H
24NCl (%) calculated value C 61.99, H 12.49, and N 7.23, measured value C 62.06, H 12.73, N 5.90.MS (m/z) 157 (M
+).
1H-NMR (D
2O, ppm) 0.98 (d, 6H), 1.42 (s, 6H), 1.45 (t, 3H), 1.65 (m, 6H), 2.31 (m, 1H).
The preparation of embodiment 8 N-propyl group-Alpha-Methyl amphetamine-compound 21
Press the method for embodiment 2, get propionitrile and 2-phenyl-2-butanols and carry out the Ritter reaction, the preparation amide intermediate is reduced with lithium aluminium hydride more earlier.Mp159-161 ℃ of preparation hydrochloride.Ultimate analysis C
13H
22NCl (%) calculated value C 68.55, H 9.73, and N 6.15, measured value C 68.59, H 10.22, and N 5.86.MS(m/z)192(M
+),133(M
+-C
3H
8N);
1H-NMR(D
2O,ppm)0.83(m,6H,2CH
3),1.58(m,2H,CH
2),1.78(s,3H,CH
3),2.05(m,1H,CH),2.29(m,1H,CH),2.53(m,1H,CH),2.85(m,1H,CH),7.54(m,5H,Ar-H)
Embodiment 9 N-propyl group-α, the preparation of beta-dimethyl-amphetamine-compound 19
Press the method for embodiment 2, get propionitrile and 2-phenyl-3-methyl-2-butanols and carry out the Ritter reaction, the preparation amide intermediate is reduced with lithium aluminium hydride more earlier.Mp190-192 ℃ of preparation hydrochloride.Ultimate analysis C
14H
24NCl (%) calculated value C 69.54, H 10.0, and N 5.79, measured value C 69.43, H 10.40, N 5.41.MS (m/z) 206 (M
+), 147 (M
+-C
3H
8N);
1H-NMR (D
2O, ppm) 0.98 (t, 3H, CH
3), 1.28 (s, 3H, CH
3), 1.39 (t, 6H, 2CH
3), 1.63 (m, 2H, CH
2), 3.98 (m, 1H, CH), 3.12-3.28 (m, 2H, CH
2), 7.43 (m, 5H, Ar-H).
Embodiment 10 N-(3-pyridyl) methyl-2, the preparation of 3-dimethyl-2-butylamine-compound 39
Press the method for embodiment 2, get 3-pyridine carbonitrile and 2,3-dimethyl-2-butylene carries out the Ritter reaction.Mp166-168 ℃ of preparation hydrochloride, ultimate analysis C
12H
19ClN
2O (%) calculated value C59.36, H7.89, N11.54, measured value C59.33, H7.98, N11.45.MS(m/z)207(M
+)106(M
+-C
6H
14N);
1H-NMR(D
2O,ppm)0.92(d,6H,2CH
3),1.42(s,6H,2CH
3),2.42(m,1H,CH),8.13(q,1H,ArH),8.86(m,2H,ArH),9.08(s,1H,ArH).
Embodiment 11 N-are valyl-2, the preparation of 3-dimethyl-2-butylamine-compound 37
Get Val-Boc 0.434g (2mmol), be dissolved among the anhydrous tetrahydro furan 2.5ml, add 2,3-dimethyl-2-butylamine 0.200g (2mmol), HOBt 0.135g (1mmol), it is molten entirely to be stirred to solid.Ice bath cooling is dissolved in the solution of tetrahydrofuran (THF) 2.5ml with DCC 0.412g (2mmol) down, drops among the above-mentioned stirring liquid, stirs 4h, and placement is spent the night.The decompressing and extracting solvent gets white solid, adds ethyl acetate 7.5ml, makes solid molten entirely.With the aqueous acid washing secondary of unsaturated carbonate hydracid sodium water solution and saturated lemon, wash secondary again with water successively to neutral, with anhydrous magnesium sulfate drying.Filter,, drip the about 5ml of anhydrous ether solution of hydrogenchloride, shake up the back and place 5h, jolting therebetween 4 times in room temperature with ethyl acetate 7ml washing secondary.Take out ether in room temperature, remove ethyl acetate under reduced pressure with warm water bath again, get the white plates solid, add anhydrous diethyl ether 10ml, stir, remove ether again under reduced pressure, get the 0.324g solid.With dehydrated alcohol and vinyl acetic monomer recrystallization, get 0.165g, productive rate 35%, hydrochloride mp240-241 ℃ after the drying.Ultimate analysis C
11H
25ClN
2O (%) calculated value C55.80, H10.64, N11.83, example value C55.85, H10.71, N11.63; MS (m/z) 201.0 (M
+).
Embodiment 12 N-tryptophyl-2, the preparation of 3-diformazan-2-butylamine-compound 40
By implementing just 11 method, get Trp-Boc under the DCC effect with 2,3-dimethyl-2-butylamine condensation prepared.Productive rate 17.5%, hydrochloride mp135-137 ℃ (dehydrated alcohol/ethyl acetate/anhydrous diethyl ether).MS(m/z)287(M
+)。
Embodiment 13 N-(N-nitro arginyl)-2, the preparation of 3-dimethyl-2-butylamine-compound 44
Press the method for embodiment 11, get Arg (NO
2)-Boc is in the presence of DCC, and with 2,3-dimethyl-2-closes preparation in butylamine side.Productive rate 36.4%, hydrochloride mp175 (d) (dehydrated alcohol/ethyl acetate); MS (m/z) 302 (M
+).
Embodiment 14 N-phenylalanyls-2, the preparation of 3-dimethyl-2-butylamine-compound 43
Press the method for embodiment 11, get Pha-Boc in the presence of DCC, with 2,3-dimethyl-2-butylamine condensation prepared.Productive rate 21.8%, hydrochloride mp232-233 ℃ (dehydrated alcohol/anhydrous diethyl ether).MS(m/z)248(M
+)。
Embodiment 15 N-leucyls-2, the preparation of 3-dimethyl-2-butylamine-compound 42
Press the method for embodiment 11, get Leu-Boc in the presence of DCC, with 2,3-dimethyl-2-butylamine condensation prepared.Hydrochloride mp250-252 ℃ (dehydrated alcohol/anhydrous diethyl ether).MS(m/z)214(M
+)。
Embodiment 16 N-isoleucyl-2, the preparation of 3-dimethyl-2-butylamine-compound 41
Press the method for embodiment 11, get Ile-Boc in the presence of DCC, with 2,3-dimethyl-2-butylamine condensation prepared.Hydrochloride mp246-248 ℃ (dehydrated alcohol/anhydrous diethyl ether).MS(m/z)214(M
+)。
Embodiment 17 N-p-toluenesulfonyls-2, the preparation of 3-dimethyl-2-butylamine-compound 38
With 2,3-dimethyl-2-butylamine 1.2 grams (0.012mol), reach the 20ml pyridine and place reaction flask, the ice-water bath cooling slowly is added dropwise to the 20ml pyridine solution of 1.9 gram (0.01mol) Tosyl chlorides down, reaction solution is a yellowly, frozen water stirs 3h, stirring at room reaction 1 hour, heating, stirring reaction 2h under the temperature outside 95~100 ℃, reaction solution still is pale brown look transparence, removes solvent under reduced pressure, adds toluene, the evaporated under reduced pressure solvent, add suitable quantity of water, can all dissolve methylbenzene extraction 4 times, water is counter carries benzene liquid once, anhydrous MgSO
4Dry benzene liquid filters, and steaming desolventizes, recrystallization in Virahol, colourless column crystallization 1.1 grams, mp80-89 ℃, recrystallization in Virahol again, white crystals shape solid 0.8 gram, mp88-89 ℃.Ultimate analysis C
13H
21NO
2S (%): theoretical value C61.14, H8.29, N5.48, experimental value C61.11, H8.37, N5.55; MS (m/z) 255 (M
+), 172 (B);
1H-NMR (CDCl
3, ppm) 0.91 (d, J=6.8Hz, 6H, 2CH
3), 1.17 (S, 6H, 2CH
3), 1.82 (M, 1H, CH), 2.47 (S, 3H, CH
3), 7.32 (d, J=8.0,2H, 2Ar-H), 7.82 (d, J=8.0,2H, 2Ar-H ').
Embodiment 18 N-(1-methylethyl)-2, the preparation of 3-dimethyl-3-hydroxyl-2-butylamine-compound 58
Get 2,3-dimethyl-2,3-oxyethane 41.8g (0.418mol), 2-propylamine 20.0g (0.33mol) and toluene 50ml placed autoclave, in 170 ℃ of heated and stirred 48 hours.After being chilled to room temperature, open still.With 6N hydrochloric acid soln 100ml, divide three extractions.Merge acid solution, wash in right amount with toluene, again with 40% oxychlorination sodaization to pH be 12.Oil float is arranged in liquid level.Use ether 150ml, divide three extractions.Combined ether liquid is used the Anhydrous potassium carbonate drying.Filter, reclaim ether as for.Bp60-65/10mm Hg cut is collected in underpressure distillation, gets 2-(N-2-methylethyl)-3-hydroxyl-2, the 3-dimethyl butylamine.Hydrochloride mp156-158 ℃ (dehydrated alcohol/anhydrous diethyl ether).Ultimate analysis C
9H
22ClNO (%) calculated value C55.23, H11.33, N7.16, measured value C55.23, H11.65, N6.95; MS (m/z) 160 (M
+);
1H-NMR (CDCl
3, ppm), 1.33 (s, 6H, 2CH
3), 1.41 (d, 6H, 2CH
3), 1.42 (s, 6H, 2CH
3), 3.81 (m, 1H, CH).
Embodiment 19 N-(1-methylethyl)-2, the preparation of 3-methyl-2-butylamine tosilate
Anhydrous tosic acid: get the tosic acid that contains crystal water, in 110 ℃ of heat fused, simultaneously water is steamed, be heated to till anhydrous the steaming, cooling is placed standby in moisture eliminator.
Get the anhydrous tosic acid of 0.60g (3.5mmol), be dissolved in few dehydrated alcohol of trying one's best, under agitation splash into and be dissolved with 0.55g (0.38mmol) N-(1-methylethyl)-2, the 10mL anhydrous ether solution of 3-dimethyl-2-butylamine drips and finishes, and placement is spent the night; Then, boil off solvent, solid gets colorless solid 1.07g, mp119-120 ℃ with the dehydrated alcohol thorough washing.
1H-NMR(D
2O,ppm):0.96(d,6H,2CH
3),1.303(s,6H,2CH
3),1.36(d,6H,2CH
3),2.02-2.15(m,1H,CH),2.401(s,3H,CH
3),3.62-3.73(m,1H,CH),7.38(d,2H,2Ar-H),7.70(d,2H,2Ar-H’)。
Embodiment 20 N-(1-methylethyl)-2, the preparation of 3-methyl-2-butylamine hydrochloride
Take by weighing N-(1-methylethyl)-2,3-dimethyl-2-butylamine 100.0g, be dissolved in 200mL ethanol, frozen water cools off and shakes down and adds 100mL hydrochloric acid, and the pressure reducing and steaming solvent is added ethanol to doing then, evaporated under reduced pressure again, then with 1: 1 Virahol-cyclohexane recrystallization, get colorless solid 130.5g (95.5%), mp228-230 ℃ of (i-PrOH: Et
2O).Ultimate analysis C
9H
22ClN (%) calculated value C60.14, H12.34, N7.79, Cl19.73, measured value C60.14, H12.48, N7.31, Cl19.67.
1H-NMR(D
2O,ppm)0.98(d,J=6.75H,6H),1.33(S,6H),1.37(d,J=6.46,6H),2.10(m,1H),3.70(m,1H)。MS(m/z)143(M
+),100(B)。
Embodiment 21 N-(1-methylethyl)-N-(2,4,5-trichlorobenzene oxygen ethanoyl)-2, the preparation of 3-dimethyl-2-butylamine
2,4,5-trichlorine phenoxyacetyl chloride: 2,4,5-trichlorophenoxyacetic acid 51.3g and sulfur oxychloride 18mL, stirring and refluxing reaction 2.5h adds a small amount of dry benzene, changes reflux into water distilling apparatus, remove excessive sulfur oxychloride and benzene under reduced pressure, solid is promptly separated out in cooling, places standby.
Add N-(1-methylethyl)-2 in the reaction flask, 3-methyl-2-butylamine hydrochloride 1.81g, triethylamine 3.30g, the 4-dimethylamino pyridine of catalytic amount and toluene 50mL, under agitation splash into and be dissolved with 2,4, the 20mL toluene solution of 5-trichlorine phenoxyacetyl chloride 5.50g, drip and finish, heated and stirred reaction 14h in 80 ℃ of oil baths, cooling then, filter, the toluene wash solid, combining methylbenzene solution is successively with water 50mL, 1N NaOH (50mL * 2), water 50mL, 1N HCl (50mL * 2) and water 50mL washing, anhydrous sodium sulfate drying, get brown thick liquid, silica gel column chromatography gets light yellow semi-solid 3.30g (86.8%).
1H-NMR(CDCl
3,ppm)0.865(d,J=6.75Hz,6H),1.401(S,6H),1.448(d,J=6.75Hz,6H),2.85(m,1H),3.98(m,1H),4.740(s,2H),6.928(s,1H),7.441(s,1H)。MS (m/z) EI
+: 338/336 (1: 1, (M-i-Pr)
+), 298/296 (M-thexyl)
+/ 294,84 (B, C
6H
12 +.); FAB
+: 378.2/380.2 (M+H)
+/ 382.2, and 336.2/338.2 (1: 1, (M-i-Pr)
+), 296.1/298.1 (B, M+H-C
6H
12), 100.1 (thexylamine)
+, 85.1 (C
6H
13 +). following biological activity test is used for further specifying the present invention.
1 pair of endothelin-1 of biological effect EXPERIMENTAL EXAMPLE 1 formula I representative compounds of the present invention causes the influence of SD pulmonary arterial pressure in rats highly compressed
As shown in table 1, the SD rat is injected endothelin-1 1.5 μ g/kg through pulmonary artery can bring out pulmonary hypertension, and 5-30min after the administration, mean pulmonary arterial pressure (mPAP) are significantly higher than the preceding level of administration; Compare with the physiological saline group, compound 10.5 and 1.0mg/kg do not have obvious influence (P>0.05) to normal Ppa pulmonary artery pressure.Show that 1 pair of normal Ppa pulmonary artery pressure of compound do not have influence.
1 pair of normal mean pulmonary arterial pressure of table 1. compound, (mPAP) influence before the medicine n administration different time after the administration, (min) mPAP, (mmHg), (mg/kg) 5 10 20 30 60 physiological saline group 7 20.6 ± 1.8 20.1 ± 1.6 20.3 ± 1.5 20.4 ± 1.1 20.6 ± 1.7 21.0 ± 2.1 endothelin-1 group 13 21.4 ± 2.6 26.2 ± 3.9
*26.1 ± 4.2
*25.8 ± 4.3
*24.8 ± 3.6
*22.0 ± 2.5 compound 1 0.5 7 20.9 ± 1.1 20.7 ± 1.2
##20.6 ± 2.2
##19.9 ± 1.6
##20.3 ± 1.1
##20.9 ± 1.1 compound 1 1.0 8 20.0 ± 2.4 20.0 ± 2.9
##20.4 ± 2.4
##20.0 ± 1.8
##20.4 ± 2.6
##19.6. ± 1.7
#Compare * * P<0.01 with the physiological saline group; Compare with the endothelin-1 group,
#P<0.05,
##P<0.01.
As shown in table 2, the SD rat is injected compound 10.5 or 1.0mg/kg earlier through pulmonary artery, injects 1.5 μ g/kg endothelin-1s then, observes medicine causes pulmonary hypertension to endothelin-1 prophylactic effect.The pulmonary hypertension that compound 10.5 and 1.0mg/kg can prevent endothelin-1 to bring out approaches the level of physiological saline group all significantly being lower than the endothelin-1 group with 5,10,15,30 and 60 minutes mPAP behind the compound 11.0mg/kg; Compound 10.5mg/kg is only to 7,8, and 9,15,25 and 35 minutes Ppa pulmonary artery pressures increase prophylactic effect.Prompting compound 1 has the effect of the pulmonary hypertension that significant anti-endothelin-1 causes.
1 pair of endothelin-1 of table 2. compound, (ET) cause before the prophylactic effect medicine n administration of pulmonary hypertension different time after the administration, (min) mPAP, (mmHg), (mg/kg) 5 10 20 30 60 physiological saline group 7 20.6 ± 1.8 20.1 ± 1.6 20.3 ± 1.5 20.4 ± 1.1 20.6 ± 1.7 21.0 ± 2.1 endothelin-1 group 13 21.4 ± 2.6 26.2 ± 3.9
*26.1 ± 4.2
*25.8 ± 4.3
*24.8 ± 3.6
*22.0 ± 2.5 compounds 1 0.5+ET 7 18.0 ± 2.6 20.8 ± 5.3 20.1 ± 4.2 20.1 ± 4.3 19.5 ± 3.3 19.3 ± 2.3 compounds, 1 1.0+ET 7 19.1 ± 1.5 20.9 ± 2.4
##21.2 ± 2.3
##20.6 ± 2.6
##19.9 ± 1.7
##18.7 ± 1.5
##Compare * * P<0.01 with the physiological saline group; Compare with the endothelin-1 group,
#P<0.05,
##P<0.01.
As shown in table 3, the SD rat is injected 1.5 μ g/kg endothelin-1s earlier through pulmonary artery, injects compound 10.5 or 1.0mg/kg then, observes medicine causes pulmonary hypertension to endothelin-1 therapeutic action.Compound 10.5 and 1.0mg/kg are relatively poor to the pulmonary hypertension therapeutic action that endothelin-1 brings out.
1 pair of endothelin-1 of table 3. compound, (ET) cause before the therapeutic action medicine n administration of pulmonary hypertension different time after the administration, (min) mPAP, (mmHg), (mg/kg) 5 10 20 30 60 physiological saline group 7 20.6 ± 1.8 20.1 ± 1.6 20.3 ± 1.5 20.4 ± 1.1 20.6 ± 1.7 21.0 ± 2.1 endothelin-1 group 13 21.4 ± 2.6 26.2 ± 3.9
*26.1 ± 4.2
*25.8 ± 4.3
*24.8 ± 3.6
*22.0 ± 2.5ET+ compound 1 0.5 7 22.4 ± 1.4 24.3 ± 4.4
*24.3 ± 3.8
*25.7 ± 4.7
*25.3 ± 4.2
*23.4 ± 3.4ET+ compound 1 1.0 7 22.0 ± 2.1 23.4 ± 2.7
*22.1 ± 2.0 21.7 ± 1.5 22.3 ± 1.8 22.3 ± 1.2 with the physiological saline group relatively, * P<0.05, * * P<0.01.
Above biological effect EXPERIMENTAL EXAMPLE 1 prompting: inject endothelin-1 through pulmonary artery and can bring out pulmonary hypertension, compound 10.5 and 1.0mg/kg do not have obvious influence to normal Ppa pulmonary artery pressure, but the pulmonary hypertension that can prevent endothelin-1 to bring out, and the pulmonary hypertension therapeutic action that endothelin-1 is brought out is relatively poor.
The research method of biological effect EXPERIMENTAL EXAMPLE 1: SD rat 280-320g, ♂.Anaesthetize through 20% urethane intraperitoneal injection, the microtubular that inserts full heparin solution through the right side external jugular vein reaches pulmonary artery, connect pressure transducer, with four road physiograph record rat mean pulmonary arterial pressures (mPAP), the while recorded heart rate, and, stablize and measure the basic hemodynamic index of record after 30 minutes by femoral artery record body circulation pressure.Inject compound 10.5 and 1.0mg/kg through pulmonary artery, observing medicine influences normal mean pulmonary arterial pressure (mPAP); Inject compound 10.5 or 1.0mg/kg earlier through pulmonary artery, inject 1.5 μ g/kg endothelin-1s then, observe medicine causes pulmonary hypertension to endothelin-1 prophylactic effect; Inject 1.5 μ g/kg endothelin-1s earlier through pulmonary artery, inject compound 10.5 and 1.0mg/kg then, observe medicine causes pulmonary hypertension to endothelin-1 therapeutic action.
Experimental technique sees reference for details: Guan Zhanjun etc.The relation of Chronic Hypoxic Rats pulmonary hypertension and hypertrophy of right heart and DOPAMINE CONTENT IN RABBIT.Chinese Medical Journal 1990; 70 (10): 582
The influence of the aorta contraction that 1 pair of endothelin-1 of biological effect EXPERIMENTAL EXAMPLE 2 formula I representative compounds of the present invention causes
As shown in Figure 1, in the calcic nutritive medium, compound 10.5-100 μ mol/L can partly resist the blood vessel function that contracts of endothelin-1, and the maximum diastolic rate of compound 1100 μ mol/L is 65.4 ± 14.6% (n=6), the EC of its vasodilator effect
50± L
95Be 5.5 ± 4.0 μ mol/L, b ± S
bBe 0.29 ± 0.05, r=0.94 (P<0.05).In no calcium nutritive medium, compound 10.5-100 μ mol/L antagonism endothelin-1 contracts vasoactive effect than obviously weakening in the calcic nutritive medium, and the maximum diastolic rate of compound 1100 μ mol/L only is 36.4 ± 11.0% (n=6).Prompting compound 1 antagonism endothelin-1 contract flow in blood vessel function and its prevention extracellular Ca2 relevant.
The research method of biological effect EXPERIMENTAL EXAMPLE 2: with the male Wistar rat sacrificed by decapitation, open chest taking-up thoracic aorta rapidly and fall branch, place cold (4 ℃) vascular nutrition liquid, the composition of nutritive medium (mmol/L): NaCl 117, and KCl 5, CaCl
21.5, NaH
2PO
41.0, NaHCO
325, MgSO
47H
2O 1.2, and glucose 11.5 is with distilled water preparation, pH7.4.Carefully peel off blood vessel fatty tissue on every side, the flush away clot is cut into the long vascular circle of 3mm, hangs in the bath that the 10ml nutritive medium is housed, and passes to 95%O
2And CO
25% gas mixture, vascular circle one end is fixed, and the other end is connected in tonotransducer, by the tension variation of self-balancing recorder record blood vessel.Resting tension 0.5g, 37 ℃ of constant temperature.Sample is beginning administration behind the balance 45min (every 10min changes one time of nutrition liquid) under certain tension force.(the former forms the same, the CaCl during the latter will form with no calcium nutritive medium at calcic respectively earlier
2Change 5mmol/L EGTA into) in 1nmol/L endothelin-1 preshrinking aortic annulus, observe the vasodilator effect of compound 10.5~100 μ mol/L then.
Experimental technique sees reference for details: what is magnificent, Wang Hai, Xiao Wenbin.Pinacidil and nifedipine are to the endothelin vasoactive influence of contracting.Chinese Pharmacological circular 1997; 13 (6): 502-505.
The influence of pulmonary hypertension and right ventricle reconstruct due to 1 pair of chronic hypoxia of biological effect EXPERIMENTAL EXAMPLE 3 formula I representative compounds of the present invention
As shown in table 4, chronic hypoxia can make SD rat mean pulmonary arterial pressure (MPAP) significantly increase, and right ventricle weight (RV) enlarges markedly with the ratio of left ventricle and interventricular septum weight sum (LV+S), compares P<0.01 with normal group.Show that chronic hypoxia can make Ppa pulmonary artery pressure increase, right ventricle generation reconstruct.Preceding 30 minutes of treatment group rat anoxic every day gives compound 10.75mg/kg or 1.5mg/kg by irritating stomach, and the MPAP that can obviously suppress due to the hypoxemia raises, and makes it to maintain normal level.But 1 couple of RV/ of compound (LV+S) does not have obviously influence.
Above biological effect EXPERIMENTAL EXAMPLE 3 promptings: the property anoxic can make Ppa pulmonary artery pressure increase, right ventricle generation reconstruct.Preceding 30 minutes of anoxic is irritated stomach and is given the MPAP that SD rat compound 10.75mg/kg or 1.5mg/kg can obviously suppress due to the hypoxemia and raise, but reconstruct does not have obvious influence to right ventricle.1 pair of chronic hypoxia of table 4. compound causes prophylactic effect medicine (mg/kg) n MPAP (mmHg) RV/ (LV+S) normal group 7 20.29 ± 0.76 0.26 ± 0.06 chronic hypoxia group 7 44.29 ± 7.61** 0.39 ± 0.07** compound 1 0.75 9 20.24 ± 2.40 of pulmonary hypertension
##0.37 ± 0.07** compound 1 1.5 7 20.57 ± 1.90
##0.38 ± 0.07** and normal group are relatively, * * P<0.01; Compare with the chronic hypoxia group,
##P<0.01.
The research method of biological effect EXPERIMENTAL EXAMPLE 3: 30 of male SD rats, purchase in the school Experimental Animal Center, 2~3 months ages of mouse, body weight 200~220g, sub-cage rearing is freely drunk water.Be divided into 4 groups at random: 7 of normal control groups; 7 of hypoxia model groups; 0.75mg/kg 9 of treatment groups (hypoxemia+compound 1); 1.5mg/kg 7 of treatment groups (hypoxemia+compound 1).Preceding 30 minutes of treatment group anoxic every day gives compound 10.75mg/kg or 1.5mg/kg by irritating stomach.Hypoxemia is respectively organized rat to be positioned in the normal pressure low oxygen cabin, when hypoxemia begins, elder generation's nitrogen injection in the cabin, reduce oxygen concentration in the cabin, with oxygen measuring instrument and magnetic valve feedback assembly, make that oxygen concn is stable at 10 ± 0.5% in the cabin, the hypoxemia cabin has aperture to communicate with ambient atmosphere, cabin atmosphere is pressed with the external world be consistent.CO in the cabin
2With water vapour respectively with sodica calx and anhydrous chlorides of rase calcium absorption.Carry out intermittent hypoxia, 6 hours every days, 6 days weekly, the 7th day not anoxic, totally 4 weeks.The induced lung haemodynamics detects: rat is anaesthetized with 20% urethane intraperitoneal.With microguide through the external jugular vein intubate to pulmonary artery, adopt four road physiographs (RM-6200C, Chengdu Instruement Factory) records mean pulmonary arterial pressure (mPAP) and heart rate.Ventricular weight is measured: deep cutting chest after above mensuration, taking out rat heart places 10% neutral formalin solution to fix for 1 week, cut off atrial tissue, separate right ventricle (RV) and left ventricle+interventricular septum (LV+S), with the weight of dividing another name RV and LV+S behind the filter paper suck dry moisture, calculate the variation of RV/ (LV+S) reflection right ventricle weight then, to determine to have or not the right ventricle plumpness.
Experimental technique sees reference for details: Guan Zhanjun etc.The relation of Chronic Hypoxic Rats pulmonary hypertension and hypertrophy of right heart and DOPAMINE CONTENT IN RABBIT.Chinese Medical Journal 1990; 70 (10): 582.
Description of drawings:
The effect of the isolated rat aorta vessel that Fig. 1 causes for 1 pair of endothelin-1 of compound
Claims (21)
1, prevention or treatment are by the pulmonary hypertension due to anoxic, chronic bronchitis, obstructive emphysema, bronchiectasis, bronchial asthma, pulmonary tuberculosis, pneumoconiosis, pulmonary fibrosis, multiple pulmonary microembolization and the lung arteriolitis or the general formula I of agnogenic primary pulmonary hypertension
aShown sulfonamide derivatives, its isomer, raceme or optical isomer, medicinal acid addition salt, its acid amides or its ester,
Wherein:
(1). work as R '
1Be sec.-propyl, R '
2, R '
3During for methyl, R '
4Can be sec.-propyl, normal-butyl, isobutyl-, tertiary butyl, ring third methyl, dimethylamino ethyl, allyl group, diisopropylamino ethyl; Or
(2). work as R '
1, R '
2Be methyl, R '
3-C-NH-R '
4Can be the sulfonamide derivatives shown in the following formula, its
Isomer, raceme or optical isomer,
Wherein R and R ' are C
1-5Alkyl, n are the integer of 1-8; Or
(3). work as R '
1Be phenyl, R '
2During for methyl, R '
3Can be methyl, ethyl, sec.-propyl, R '
4Can be propyl group, methoxy carbonyl methyl; Or
(4). work as R '
1Be (CH
3)
2C (NH
2)-, R '
2, R '
3Be CH
3-time, R '
4Be (CH
3)
2CH-; Or
Work as R '
1Be (CH
3)
2C (OH)-, R '
2, R '
3Be CH
3-time, R '
4Be (CH
3)
2CH-or (CH
3)
2CH (CH
3)-; Or
Work as R '
1For
R '
2, R '
3Be CH
3-or R '
2With R '
3Be together-(CH
2)
4-or-(CH
2)
5-time, R '
4Be (CH
3)
2CH-; Or
Work as R '
1Be (CH
3)
2C (ONO
2)-, R '
2, R '
3Be CH
3-time, R '
4Be (CH
3)
2CH-; Or
Work as R '
1For
R '
2, R '
3Be CH
3-or R '
2With R '
3Be together-(CH
2)
4-or-(CH
2)
5-time, R '
4Be (CH
3)
2CH-; Or
Work as R '
1For
R '
2, R '
3For-(CH
2)
5-time, R '
4Be (CH
3)
2CCH (CH
3)-; Or
(6). work as R '
1Be (CH
3)
2CH-, R '
2, R '
3Be CH
3-time, R '
4Be Val-, Trp-, Ile-, Leu-, Phe-, O
2N-Arg-, Pro-, Leu-Val-, Trp-Trp-Trp-, (CH
3)
2CH-SO
2-, perhaps R '
4Be one of following group:
Or
Or
Work as R '
1Be cyclopropyl, R '
2And R '
3For-CH
2-CH
2-time, R '
4Be Val-; Or
Work as R '
1Be cyclohexyl, R '
2And R '
3Be CH
3-time, R '
4Be Pro-; Or
2, the sulfonamide derivatives of claim 1, wherein, described compound is selected from a kind of in the group that following compound forms:
N-(1-methylethyl)-2,3-dimethyl-2-butylamine;
N-propyl group-2,3-dimethyl-2-butylamine;
N-(2-methyl-propyl)-2,3-dimethyl-2-butylamine;
N-encircles third methyl-2,3-dimethyl-2-butylamine;
N-propenyl-2,3-dimethyl-2-butylamine;
N-(2-(two-(1-methylethyl) amidos) ethyl)-2,3-dimethyl-2-butylamine;
N-butyl-2,3-dimethyl-2-butylamine;
N-propyl group-Alpha-Methyl amphetamine;
N-propyl group-α, the beta-dimethyl-amphetamine;
N-(3-pyridyl) formyl radical-2,3-dimethyl-2-butylamine;
N-is valyl-2,3-dimethyl-2-butylamine;
N-tryptophyl-2,3-dimethyl-2-butylamine;
N-(N-nitro) arginyl-2,3-dimethyl-2-butylamine;
N-phenylalanyl-2,3-dimethyl-2-butylamine;
N-leucyl-2,3-dimethyl-2-butylamine;
N-isoleucyl-2,3-dimethyl-2-butylamine;
N-p-toluenesulfonyl-2,3-dimethyl-2-butylamine;
N-(1-methylethyl)-2,3-dimethyl-3-hydroxyl-2-butylamine;
N-cinnamoyl-N-(1-methylethyl)-2,3-dimethyl-2-butylamine;
N-(1-methylethyl)-N-(2,4,5-trichlorobenzene oxygen ethanoyl)-2,3-dimethyl-2-butylamine.
3, the sulfonamide derivatives of claim 1, wherein, the salt of described medicinal sour addition is selected from hydrochloride, vitriol, phosphoric acid salt, hydrobromate; Or acetate, oxalate, lemon salt, gluconate, succinate, tartrate, tosilate, mesylate, benzoate, lactic acid salt or maleate.
4, the sulfonamide derivatives of claim 1, wherein, described compound is N-(1-methylethyl)-2,3-dimethyl-2-butylamine tosilate.
5, a kind of preparation is as the defined formula I of claim 1
aThe method of aminated compounds, it comprises primary amine R
1' R
2' R
3' CNH
2With R
4' X in organic solvent, through being heated to 50-300 ℃ and/or be pressurized to 0.1-20MPa and react, R wherein
1', R
2', R
3' and R
4' according to claim 1, X is easy leavings group; It is characterized in that described primary amine R
1' R
2' R
3' CNH
2Prepare by the following method:
At first, with the same R of urea
1' R
2' R
3' the corresponding alkene of C or alcohol or the two mixture be heated to 20-200 ℃ of prepared in reaction formula R in organic acid under vitriol oil effect
1' R
2' R
3' CNHCONH
2The alkyl urea; Then, this alkyl urea of hydrolysis prepares corresponding primary amines.Wherein, described organic acid is selected from acetate, trifluoroacetic acid or methylsulfonic acid.
6, the described method of claim 5, wherein, described primary amine and R
4' X has been reflected under the catalyst action and carried out, described catalyzer is disacidify agent and/or phase-transfer catalyst.
7, the described method of claim 6, wherein, described disacidify agent is a Lewis base, phase-transfer catalyst is ethylene glycol or polyoxyethylene glycol.
8, the described method of claim 5, wherein, described organic solvent is toluene, dimethylbenzene, 1,2-ethylene dichloride, 1,4-dioxane, glycol dimethyl ether, N, dinethylformamide, N,N-dimethylacetamide, N-Methyl pyrrolidone, N, accelerine or N, the N-Diethyl Aniline.
9, a kind of preparation I as claimed in claim 1
aThe method of aminated compounds, it comprises primary amine R
1' R
2' R
3' CNH
2With R
4' corresponding aldehydes or ketones is at catalyzer and have/organic solvent-free in the presence of, through being heated to 30-300 ℃ and/or be pressurized to 0.1-20MPa and carry out hydrogenation, R wherein
1', R
2', R
3' and R
4' according to claim 1; It is characterized in that described primary amine R
1' R
2 'R
3' CNH
2The preparation method as described in the claim 2.
10, the described method of claim 9, wherein, described catalyzer is palladium-carbon, Raney's nickel, platinum oxide or nickel-copper etc.
11, the described method of claim 9, wherein, described organic solvent is corresponding excessive aldehydes or ketones, toluene, dimethylbenzene, 1,2-ethylene dichloride, 1,4-dioxane, glycol dimethyl ether, methyl alcohol or ethanol.
12, the sulfonamide derivatives shown in the general formula I, its isomer, raceme or optical isomer, its medicinal acid addition salt, its acid amides or its ester can be used for preventing or treating by the application in the medicine of pulmonary hypertension due to anoxic, chronic bronchitis, obstructive emphysema, bronchiectasis, bronchial asthma, pulmonary tuberculosis, pneumoconiosis, pulmonary fibrosis, multiple pulmonary microembolization and the lung arteriolitis or agnogenic primary pulmonary hypertension in preparation
Wherein:
R
1, R
2, R
3Represent hydrogen atom, C respectively
1-20Saturated or undersaturated straight or branched aliphatic hydrocarbon, C
3-20Cycloalkyl group, replace C
3-20Cycloalkyl group, C
5-20Aryl radical, replace C
5-20Aryl radical, C
5-20Heterocycle alkyl, replacement C
5-20Heterocycle alkyl, Alpha-hydroxy C
2-20Alkyl, α-C
1-10Alkane carbonyl oxygen C
1-10Alkyl, C
6-14α-Fang carbonyl oxygen base C
1-10Alkyl, alpha-substitution C
6-14Virtue carbonyl oxygen base C
1-10Alkyl, α-C
1-10Alkoxy C
1-10Alkyl, alpha-substitution C
5-10Aryloxy C
1-10Alkyl, alpha-amino group C
1-20Alkyl, α-C
1-10Alkylamino radical C
1-10Alkyl, α-C
5-14Aryl amine C
1-10Alkyl, alpha-substitution C
5-14Aryl amine C
1-10Alkyl, α-C
1-10Alkyl amide C
1-10Alkyl, α-C
6-14Aromatic amide C
1-10Alkyl, alpha-substitution C
6-14Aromatic amide C
1-10Alkyl;
R
4Represent hydrogen atom, C
1-20Saturated fatty alkyl, C
5-20Aryl radical, replacement C
5-20Aryl radical, C
3-20Heterocycle alkyl, replacement C
3-20Heterocycle alkyl, C
3-20Heterocyclic radical, replacement C
3-20Heterocyclic radical, C
1-20Straight chain fatty acyl group, C
4-20Side chain fatty acyl group or and R
1, R
2, R
3The C that forms
3-20Cyclic hydrocarbon radical, C
3-20Heterocyclic radical, wherein said heterocycle refer to contain 1-3 and are selected from N, O or heteroatomic list of S or annelated heterocycles, and each substituting group that has substituent group is selected from: halogen, hydroxyl, cyano group, nitro, C
1-6Alkyl, C
1-6Alkoxyl group, C
1-6Alkylthio, one, two or three halo C
1-6Alkyl, amino, C
1-10The hydroxylamine base, C
1-10Alkylacyloxy, C
6-10Aryl acyloxy or C
1-10Amide group.
13, as the application of claim 12, wherein:
Described R
1Be the tertiary butyl, cyclohexyl, cyclopentyl, cyclobutyl, cyclopropyl, isopentyl, cyclobutyl; Perhaps, R
1Be cyclohexyl, cyclopentyl, cyclobutyl, cyclopropyl, isohexyl, isopentyl, isobutyl-, the sec.-propyl of alpha-substitution, wherein said substituting group is amino, hydroxyl, C
1-10Hydroxylamine base, C
1-6-oxyl, C
1-10Alkylacyloxy, C
6-10Aryl acyloxy, C
1-10Amide group;
Described R
2Or R
3Be respectively hydrogen atom, C
1-12Chain alkylene or C
3-20Cyclic hydrocarbon radical;
R
4Be hydrogen atom, C
1-20Saturated fatty alkyl, C
3-20Cycloalkyl, C
1-20Acyl group, C
1-10Hydroxylamine base C
1-10Alkyl, C
1-20Sulfoxide group, amino-acid residue and the micromolecule polypeptide fragment that combines thereof, β-nitroethylene base, beta-cyano vinyl, replacement carbonyl imido grpup, C
3-20Heterocyclic radical, and C
4-20The heterocyclic acyl substituting group.
14, the application of claim 12, wherein, described R
2, R
3Be respectively methyl, ethyl, propyl group, perhaps, R
2And R
3Be propylidene, butylidene, pentylidene and hexylidene.
15, claim 12 or 13 application, wherein, described R
1Be sec.-propyl, R
2Be methyl, R
3Be methyl.
16, as the application of claim 12, wherein, described compound is selected from a kind of in the group that following compound forms:
N-(1-methylethyl)-2,3-dimethyl-2-butylamine;
N-propyl group-2,3-dimethyl-2-butylamine;
N-(2-methyl-propyl)-2,3-dimethyl-2-butylamine;
N-encircles third methyl-2,3-dimethyl-2-butylamine;
N-propenyl-2,3-dimethyl-2-butylamine;
N-(2-(two-(1-methylethyl) amidos) ethyl)-2,3-dimethyl-2-butylamine;
N-butyl-2,3-dimethyl-2-butylamine;
N-propyl group-Alpha-Methyl amphetamine;
N-propyl group-α, the beta-dimethyl-amphetamine;
N-(3-pyridyl) formyl radical-2,3-dimethyl-2-butylamine;
N-is valyl-2,3-dimethyl-2-butylamine;
N-tryptophyl-2,3-dimethyl-2-butylamine;
N-(N-nitro) arginyl-2,3-dimethyl-2-butylamine;
N-phenylalanyl-2,3-dimethyl-2-butylamine;
N-leucyl-2,3-dimethyl-2-butylamine;
N-isoleucyl-2,3-dimethyl-2-butylamine;
N-p-toluenesulfonyl-2,3-dimethyl-2-butylamine;
N-(1-methylethyl)-2,3-dimethyl-3-hydroxyl-2-butylamine;
N-cinnamoyl-N-(1-methylethyl)-2,3-dimethyl-2-butylamine;
N-(1-methylethyl)-N-(2,4,5-trichlorobenzene oxygen ethanoyl)-2,3-dimethyl-2-butylamine.
17, as the application of claim 12, wherein, the salt of described medicinal sour addition is selected from hydrochloride, vitriol, phosphoric acid salt, hydrobromate; Or acetate, oxalate, lemon salt, gluconate, succinate, tartrate, tosilate, mesylate, benzoate, lactic acid salt or maleate.
18, as the application of claim 16, wherein, described compound is N-(1-methylethyl)-2,3-dimethyl-2-butylamine tosilate.
19, as the application of claim 12, wherein, described application is meant in medicine, the especially prevention of preparation prevention or treatment pulmonary hypertension or treats by the application in the medicine of pulmonary hypertension due to anoxic, chronic bronchitis, obstructive emphysema, bronchiectasis, bronchial asthma, pulmonary tuberculosis, pneumoconiosis, pulmonary fibrosis, multiple pulmonary microembolization and the lung arteriolitis or agnogenic primary pulmonary hypertension.
20, be used to prevent or treat the medicine of pulmonary hypertension, especially the prevention or the treatment by pulmonary hypertension due to anoxic, chronic bronchitis, obstructive emphysema, bronchiectasis, bronchial asthma, pulmonary tuberculosis, pneumoconiosis, pulmonary fibrosis, multiple pulmonary microembolization and the lung arteriolitis or agnogenic primary pulmonary hypertension general formula I shown in sulfonamide derivatives, its isomer, raceme or optical isomer, its medicinal acid addition salt, its acid amides or its ester
Wherein:
R
1, R
2, R
3Represent hydrogen atom, C respectively
1-20Saturated or undersaturated straight or branched aliphatic hydrocarbon, C
3-20Cycloalkyl group, replace C
3-20Cycloalkyl group, C
5-20Aryl radical, replace C
5-20Aryl radical, C
5-20Heterocycle alkyl, replacement C
5-20Heterocycle alkyl, Alpha-hydroxy C
2-20Alkyl, α-C
1-10Alkane carbonyl oxygen C
1-10Alkyl, C
6-14α-Fang carbonyl oxygen base C
1-10Alkyl, alpha-substitution C
6-14Virtue carbonyl oxygen base C
1-10Alkyl, α-C
1-10Alkoxy C
1-10Alkyl, alpha-substitution C
5-10Aryloxy C
1-10Alkyl, alpha-amino group C
1-20Alkyl, α-C
1-10Alkylamino radical C
1-10Alkyl, α-C
5-14Aryl amine C
1-10Alkyl, alpha-substitution C
5-14Aryl amine C
1-10Alkyl, α-C
1-10Alkyl amide C
1-10Alkyl, α-C
6-14Aromatic amide C
1-10Alkyl, alpha-substitution C
6-14Aromatic amide C
1-10Alkyl;
R
4Represent hydrogen atom, C
1-20Saturated fatty alkyl, C
5-20Aryl radical, replacement C
5-20Aryl radical, C
3-20Heterocycle alkyl, replacement C
3-20Heterocycle alkyl, C
3-20Heterocyclic radical, replacement C
3-20Heterocyclic radical, C
1-20Straight chain fatty acyl group, C
4-20Side chain fatty acyl group or and R
1, R
2, R
3The C that forms
3-20Cyclic hydrocarbon radical, C
3-20Heterocyclic radical, wherein said heterocycle refer to contain 1-3 and are selected from N, O or heteroatomic list of S or annelated heterocycles, and each substituting group that has substituent group is selected from: halogen, hydroxyl, cyano group, nitro, C
1-6Alkyl, C
1-6Alkoxyl group, C
1-6Alkylthio, one, two or three halo C
1-6Alkyl, amino, C
1-10The hydroxylamine base, C
1-10Alkylacyloxy, C
6-10Aryl acyloxy or C
1-10Amide group.
21, pharmaceutical composition, it contains the formula I arbitrary requirement of at least a claim 1-4 or that claim 20 requires
aOr formula I sulfonamide derivatives, its isomer, raceme or optical isomer, its medicinal acid addition salt, acid amides or its ester and pharmaceutical carrier or vehicle.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 01137272 CN1205175C (en) | 2001-11-02 | 2001-11-02 | Amine derivative possessing the function of anti-pulmonary hypertension and its application in pharmacological science |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 01137272 CN1205175C (en) | 2001-11-02 | 2001-11-02 | Amine derivative possessing the function of anti-pulmonary hypertension and its application in pharmacological science |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN1415596A true CN1415596A (en) | 2003-05-07 |
| CN1205175C CN1205175C (en) | 2005-06-08 |
Family
ID=4674109
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN 01137272 Expired - Lifetime CN1205175C (en) | 2001-11-02 | 2001-11-02 | Amine derivative possessing the function of anti-pulmonary hypertension and its application in pharmacological science |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN1205175C (en) |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7794965B2 (en) | 2002-03-13 | 2010-09-14 | Signum Biosciences, Inc. | Method of identifying modulators of PP2A methylase |
| US7923041B2 (en) | 2005-02-03 | 2011-04-12 | Signum Biosciences, Inc. | Compositions and methods for enhancing cognitive function |
| RU2419602C2 (en) * | 2003-08-06 | 2011-05-27 | Синомикс Инк. | Novel aromatising substances, taste modifiers, flavouring compounds, taste boosters, umami or sweet taste endowing compounds, and/or boosters and use thereof |
| CN102276698A (en) * | 2010-06-10 | 2011-12-14 | 中国人民解放军军事医学科学院毒物药物研究所 | MC4-R cyclopeptide agonist and application thereof |
| US8221804B2 (en) | 2005-02-03 | 2012-07-17 | Signum Biosciences, Inc. | Compositions and methods for enhancing cognitive function |
| US9486441B2 (en) | 2008-04-21 | 2016-11-08 | Signum Biosciences, Inc. | Compounds, compositions and methods for making the same |
| CN107436333A (en) * | 2017-09-05 | 2017-12-05 | 中国医学科学院阜外医院 | Spermine is as application of the mark in the diagnostic products of pulmonary hypertension are prepared and medicine equipment |
-
2001
- 2001-11-02 CN CN 01137272 patent/CN1205175C/en not_active Expired - Lifetime
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7794965B2 (en) | 2002-03-13 | 2010-09-14 | Signum Biosciences, Inc. | Method of identifying modulators of PP2A methylase |
| RU2419602C2 (en) * | 2003-08-06 | 2011-05-27 | Синомикс Инк. | Novel aromatising substances, taste modifiers, flavouring compounds, taste boosters, umami or sweet taste endowing compounds, and/or boosters and use thereof |
| US7923041B2 (en) | 2005-02-03 | 2011-04-12 | Signum Biosciences, Inc. | Compositions and methods for enhancing cognitive function |
| US8221804B2 (en) | 2005-02-03 | 2012-07-17 | Signum Biosciences, Inc. | Compositions and methods for enhancing cognitive function |
| US9486441B2 (en) | 2008-04-21 | 2016-11-08 | Signum Biosciences, Inc. | Compounds, compositions and methods for making the same |
| US10583119B2 (en) | 2008-04-21 | 2020-03-10 | Signum Biosciences, Inc. | Compounds, compositions and methods for making the same |
| CN102276698A (en) * | 2010-06-10 | 2011-12-14 | 中国人民解放军军事医学科学院毒物药物研究所 | MC4-R cyclopeptide agonist and application thereof |
| CN102276698B (en) * | 2010-06-10 | 2014-02-26 | 中国人民解放军军事医学科学院毒物药物研究所 | MC4-R cyclopeptide agonist and application thereof |
| CN107436333A (en) * | 2017-09-05 | 2017-12-05 | 中国医学科学院阜外医院 | Spermine is as application of the mark in the diagnostic products of pulmonary hypertension are prepared and medicine equipment |
| CN107436333B (en) * | 2017-09-05 | 2019-12-20 | 中国医学科学院阜外医院 | Application of spermine as a marker in the preparation of diagnostic products for pulmonary hypertension and medical devices |
Also Published As
| Publication number | Publication date |
|---|---|
| CN1205175C (en) | 2005-06-08 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| RU2095360C1 (en) | Indole derivatives, their optical isomers and pharmaceutically acceptable salts | |
| JP4091648B2 (en) | 2- (1H-Indolylsulfanyl) -benzylamine derivative | |
| US20030105132A1 (en) | N-phenpropylcuclopentyl-substituted glutaramide derivatives as inhibitors of neutral endopeptidase | |
| JP5663657B2 (en) | 1-[(4-Hydroxypiperidin-4-yl) methyl] pyridin-2 (1H) -one derivative, process for its preparation and use thereof | |
| MXPA03006597A (en) | N-phenpropylcyclopentyl-substituted glutaramide derivatives as nep inhibitors for fsad. | |
| CN85107020A (en) | The preparation method of alkyl sulfinamido phenyl alkylamine | |
| JPH01283220A (en) | Hypotensive action enhancer | |
| EP1527048A2 (en) | N- phenyl(piperidin-2-yl)methyl]benzamide derivatives, preparation thereof, and use thereof in therapy | |
| JP2012509879A (en) | Adamantylbenzamide compounds | |
| EP2238105B1 (en) | Novel compounds as vanilloid receptor antagonists | |
| JP4279685B2 (en) | Amine derivatives that regulate the function of potassium channels, methods for their preparation and their use | |
| CN1250536C (en) | 4-aminobenzopyran derivatives | |
| NO174959B (en) | Analogous Process for Preparing Therapeutically Active N-Aminobutyl-N-Phenyl-Arylamide Derivatives | |
| CN1415596A (en) | Amine derivative possessing the function of anti-pulmonary hypertension and its application in pharmacological science | |
| JP2569088B2 (en) | Basic substituted phenylacetonitrile and therapeutic agent for cardiovascular diseases having calcium antagonism | |
| JPS626545B2 (en) | ||
| JP2005538047A (en) | Bombesin antagonist | |
| JPH06184140A (en) | 1,4-benzodioxane derivative | |
| CN1250519C (en) | Amine derivative with K channel regulation function and its preparing process and application | |
| TW201024286A (en) | Novel compounds | |
| FR2845384A1 (en) | New 2-thiohydantoin derivatives used for treating diabetes, hyperglycemic disorders, obesity, cerebral ischemia and cerebral vascular accidents | |
| CN101084205A (en) | N-biaryl and n-arylheteroaryl piperazine derivatives as modulators of the 5ht2c receptor useful for the treatment of disorders related thereto | |
| US5296485A (en) | Substituted N-phenylpiperidines and drugs therefrom | |
| US4661513A (en) | 3-aminopropoxyphenyl derivatives, their preparation and pharmaceutical compositions containing them | |
| CN1250518C (en) | Amine derivative for preventing ischemic and anoxia damage of nerve and its medical application |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| CX01 | Expiry of patent term |
Granted publication date: 20050608 |
|
| CX01 | Expiry of patent term |