CN116407554A - Drugs for reducing intestinal damage caused by targeted antineoplastic drugs and their application - Google Patents
Drugs for reducing intestinal damage caused by targeted antineoplastic drugs and their application Download PDFInfo
- Publication number
- CN116407554A CN116407554A CN202111671059.XA CN202111671059A CN116407554A CN 116407554 A CN116407554 A CN 116407554A CN 202111671059 A CN202111671059 A CN 202111671059A CN 116407554 A CN116407554 A CN 116407554A
- Authority
- CN
- China
- Prior art keywords
- compound
- pharmaceutical composition
- pharmaceutically acceptable
- formula
- group
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 230000000968 intestinal effect Effects 0.000 title claims abstract description 61
- 239000002246 antineoplastic agent Substances 0.000 title claims abstract description 34
- 230000006378 damage Effects 0.000 title claims abstract description 33
- 239000003814 drug Substances 0.000 title claims description 26
- 229940079593 drug Drugs 0.000 title claims description 23
- 229940127072 targeted antineoplastic agent Drugs 0.000 title 1
- 150000001875 compounds Chemical class 0.000 claims abstract description 171
- 229940041181 antineoplastic drug Drugs 0.000 claims abstract description 33
- 206010012735 Diarrhoea Diseases 0.000 claims abstract description 27
- 239000008194 pharmaceutical composition Substances 0.000 claims description 45
- 238000002360 preparation method Methods 0.000 claims description 24
- 150000003839 salts Chemical class 0.000 claims description 23
- 239000000825 pharmaceutical preparation Substances 0.000 claims description 21
- 239000004480 active ingredient Substances 0.000 claims description 17
- 239000003937 drug carrier Substances 0.000 claims description 14
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 12
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 claims description 11
- 229910052799 carbon Inorganic materials 0.000 claims description 11
- 230000001225 therapeutic effect Effects 0.000 claims description 10
- 125000001072 heteroaryl group Chemical group 0.000 claims description 9
- 125000000882 C2-C6 alkenyl group Chemical group 0.000 claims description 8
- 125000003601 C2-C6 alkynyl group Chemical group 0.000 claims description 8
- 239000008187 granular material Substances 0.000 claims description 8
- 125000006552 (C3-C8) cycloalkyl group Chemical group 0.000 claims description 7
- 239000002775 capsule Substances 0.000 claims description 7
- 238000000034 method Methods 0.000 claims description 7
- 239000006187 pill Substances 0.000 claims description 7
- 239000003826 tablet Substances 0.000 claims description 7
- 206010028980 Neoplasm Diseases 0.000 claims description 6
- 125000003118 aryl group Chemical group 0.000 claims description 6
- 239000000843 powder Substances 0.000 claims description 6
- 238000006467 substitution reaction Methods 0.000 claims description 6
- 125000004191 (C1-C6) alkoxy group Chemical group 0.000 claims description 5
- 230000000259 anti-tumor effect Effects 0.000 claims description 5
- 229910052736 halogen Inorganic materials 0.000 claims description 5
- 150000002367 halogens Chemical class 0.000 claims description 5
- 210000000936 intestine Anatomy 0.000 claims description 5
- 201000010099 disease Diseases 0.000 claims description 4
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims description 4
- 239000002552 dosage form Substances 0.000 claims description 4
- 229910052760 oxygen Inorganic materials 0.000 claims description 4
- 229910052708 sodium Inorganic materials 0.000 claims description 4
- LSNNMFCWUKXFEE-UHFFFAOYSA-M Bisulfite Chemical group OS([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-M 0.000 claims description 3
- 239000013543 active substance Substances 0.000 claims description 3
- 125000002947 alkylene group Chemical group 0.000 claims description 3
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 claims description 3
- 125000002768 hydroxyalkyl group Chemical group 0.000 claims description 3
- 229910021645 metal ion Inorganic materials 0.000 claims description 3
- 229910052717 sulfur Inorganic materials 0.000 claims description 3
- 239000003405 delayed action preparation Substances 0.000 claims description 2
- 150000001721 carbon Chemical group 0.000 claims 3
- 230000000694 effects Effects 0.000 abstract description 25
- 238000011282 treatment Methods 0.000 abstract description 12
- 241000700159 Rattus Species 0.000 description 50
- 239000005411 L01XE02 - Gefitinib Substances 0.000 description 33
- 229960002584 gefitinib Drugs 0.000 description 33
- XGALLCVXEZPNRQ-UHFFFAOYSA-N gefitinib Chemical compound C=12C=C(OCCCN3CCOCC3)C(OC)=CC2=NC=NC=1NC1=CC=C(F)C(Cl)=C1 XGALLCVXEZPNRQ-UHFFFAOYSA-N 0.000 description 33
- -1 hydroxypropyl Chemical group 0.000 description 25
- 241000252212 Danio rerio Species 0.000 description 23
- 229940125904 compound 1 Drugs 0.000 description 21
- DUYJMQONPNNFPI-UHFFFAOYSA-N osimertinib Chemical compound COC1=CC(N(C)CCN(C)C)=C(NC(=O)C=C)C=C1NC1=NC=CC(C=2C3=CC=CC=C3N(C)C=2)=N1 DUYJMQONPNNFPI-UHFFFAOYSA-N 0.000 description 20
- 229960003278 osimertinib Drugs 0.000 description 19
- 239000005551 L01XE03 - Erlotinib Substances 0.000 description 18
- 229960001433 erlotinib Drugs 0.000 description 18
- AAKJLRGGTJKAMG-UHFFFAOYSA-N erlotinib Chemical compound C=12C=C(OCCOC)C(OCCOC)=CC2=NC=NC=1NC1=CC=CC(C#C)=C1 AAKJLRGGTJKAMG-UHFFFAOYSA-N 0.000 description 18
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 18
- 125000004432 carbon atom Chemical group C* 0.000 description 15
- 239000008280 blood Substances 0.000 description 11
- 210000004369 blood Anatomy 0.000 description 11
- 239000000203 mixture Substances 0.000 description 11
- 230000037396 body weight Effects 0.000 description 10
- 230000001681 protective effect Effects 0.000 description 10
- 210000001072 colon Anatomy 0.000 description 9
- 238000002474 experimental method Methods 0.000 description 9
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 8
- 210000002919 epithelial cell Anatomy 0.000 description 8
- 230000002496 gastric effect Effects 0.000 description 8
- 229920000858 Cyclodextrin Polymers 0.000 description 7
- 206010070834 Sensitisation Diseases 0.000 description 7
- 230000002550 fecal effect Effects 0.000 description 7
- 210000003405 ileum Anatomy 0.000 description 7
- 210000004877 mucosa Anatomy 0.000 description 7
- 230000008313 sensitization Effects 0.000 description 7
- 239000000243 solution Substances 0.000 description 7
- 239000002904 solvent Substances 0.000 description 7
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 6
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 6
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 6
- 238000003304 gavage Methods 0.000 description 6
- 210000002175 goblet cell Anatomy 0.000 description 6
- 208000037817 intestinal injury Diseases 0.000 description 6
- 241001465754 Metazoa Species 0.000 description 5
- 102000015728 Mucins Human genes 0.000 description 5
- 108010063954 Mucins Proteins 0.000 description 5
- 235000010355 mannitol Nutrition 0.000 description 5
- 230000028327 secretion Effects 0.000 description 5
- 239000011734 sodium Substances 0.000 description 5
- 239000000126 substance Substances 0.000 description 5
- 208000024891 symptom Diseases 0.000 description 5
- 206010059024 Gastrointestinal toxicity Diseases 0.000 description 4
- 238000010521 absorption reaction Methods 0.000 description 4
- 229940044683 chemotherapy drug Drugs 0.000 description 4
- 239000000945 filler Substances 0.000 description 4
- 206010016766 flatulence Diseases 0.000 description 4
- 231100000414 gastrointestinal toxicity Toxicity 0.000 description 4
- 210000001035 gastrointestinal tract Anatomy 0.000 description 4
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 4
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 4
- 239000000546 pharmaceutical excipient Substances 0.000 description 4
- 238000007619 statistical method Methods 0.000 description 4
- 238000005303 weighing Methods 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- GAWIXWVDTYZWAW-UHFFFAOYSA-N C[CH]O Chemical group C[CH]O GAWIXWVDTYZWAW-UHFFFAOYSA-N 0.000 description 3
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 3
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 3
- OFOBLEOULBTSOW-UHFFFAOYSA-N Malonic acid Chemical compound OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 3
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 3
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 3
- 230000005856 abnormality Effects 0.000 description 3
- 239000002253 acid Substances 0.000 description 3
- 125000002057 carboxymethyl group Chemical group [H]OC(=O)C([H])([H])[*] 0.000 description 3
- 210000004534 cecum Anatomy 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 229940125898 compound 5 Drugs 0.000 description 3
- 230000007850 degeneration Effects 0.000 description 3
- 238000001514 detection method Methods 0.000 description 3
- 238000001035 drying Methods 0.000 description 3
- 239000003995 emulsifying agent Substances 0.000 description 3
- 230000003203 everyday effect Effects 0.000 description 3
- 210000003608 fece Anatomy 0.000 description 3
- 238000005469 granulation Methods 0.000 description 3
- 230000003179 granulation Effects 0.000 description 3
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 3
- 238000003305 oral gavage Methods 0.000 description 3
- 230000001575 pathological effect Effects 0.000 description 3
- HFHDHCJBZVLPGP-UHFFFAOYSA-N schardinger α-dextrin Chemical compound O1C(C(C2O)O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC(C(O)C2O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC2C(O)C(O)C1OC2CO HFHDHCJBZVLPGP-UHFFFAOYSA-N 0.000 description 3
- 239000007787 solid Substances 0.000 description 3
- 239000007909 solid dosage form Substances 0.000 description 3
- 239000000725 suspension Substances 0.000 description 3
- 229940121358 tyrosine kinase inhibitor Drugs 0.000 description 3
- 239000000080 wetting agent Substances 0.000 description 3
- PUPZLCDOIYMWBV-UHFFFAOYSA-N (+/-)-1,3-Butanediol Chemical compound CC(O)CCO PUPZLCDOIYMWBV-UHFFFAOYSA-N 0.000 description 2
- PCWPQSDFNIFUPO-VDQKLNDWSA-N (1S,3R,5R,6S,8R,10R,11S,13R,15R,16S,18R,20R,21S,23R,25R,26S,28R,30R,31S,33R,35R,36R,37S,38R,39S,40R,41S,42R,43S,44R,45S,46R,47S,48R,49S)-37,39,41,43,45,47,49-heptakis(2-hydroxyethoxy)-5,10,15,20,25,30,35-heptakis(hydroxymethyl)-2,4,7,9,12,14,17,19,22,24,27,29,32,34-tetradecaoxaoctacyclo[31.2.2.23,6.28,11.213,16.218,21.223,26.228,31]nonatetracontane-36,38,40,42,44,46,48-heptol Chemical compound OCCO[C@H]1[C@H](O)[C@@H]2O[C@H]3O[C@H](CO)[C@@H](O[C@H]4O[C@H](CO)[C@@H](O[C@H]5O[C@H](CO)[C@@H](O[C@H]6O[C@H](CO)[C@@H](O[C@H]7O[C@H](CO)[C@@H](O[C@H]8O[C@H](CO)[C@@H](O[C@H]1O[C@@H]2CO)[C@@H](O)[C@@H]8OCCO)[C@@H](O)[C@@H]7OCCO)[C@@H](O)[C@@H]6OCCO)[C@@H](O)[C@@H]5OCCO)[C@@H](O)[C@@H]4OCCO)[C@@H](O)[C@@H]3OCCO PCWPQSDFNIFUPO-VDQKLNDWSA-N 0.000 description 2
- QFLWZFQWSBQYPS-AWRAUJHKSA-N (3S)-3-[[(2S)-2-[[(2S)-2-[5-[(3aS,6aR)-2-oxo-1,3,3a,4,6,6a-hexahydrothieno[3,4-d]imidazol-4-yl]pentanoylamino]-3-methylbutanoyl]amino]-3-(4-hydroxyphenyl)propanoyl]amino]-4-[1-bis(4-chlorophenoxy)phosphorylbutylamino]-4-oxobutanoic acid Chemical compound CCCC(NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@@H](NC(=O)CCCCC1SC[C@@H]2NC(=O)N[C@H]12)C(C)C)P(=O)(Oc1ccc(Cl)cc1)Oc1ccc(Cl)cc1 QFLWZFQWSBQYPS-AWRAUJHKSA-N 0.000 description 2
- DEQANNDTNATYII-OULOTJBUSA-N (4r,7s,10s,13r,16s,19r)-10-(4-aminobutyl)-19-[[(2r)-2-amino-3-phenylpropanoyl]amino]-16-benzyl-n-[(2r,3r)-1,3-dihydroxybutan-2-yl]-7-[(1r)-1-hydroxyethyl]-13-(1h-indol-3-ylmethyl)-6,9,12,15,18-pentaoxo-1,2-dithia-5,8,11,14,17-pentazacycloicosane-4-carboxa Chemical compound C([C@@H](N)C(=O)N[C@H]1CSSC[C@H](NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CCCCN)NC(=O)[C@@H](CC=2C3=CC=CC=C3NC=2)NC(=O)[C@H](CC=2C=CC=CC=2)NC1=O)C(=O)N[C@H](CO)[C@H](O)C)C1=CC=CC=C1 DEQANNDTNATYII-OULOTJBUSA-N 0.000 description 2
- VBICKXHEKHSIBG-UHFFFAOYSA-N 1-monostearoylglycerol Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCC(O)CO VBICKXHEKHSIBG-UHFFFAOYSA-N 0.000 description 2
- 229920001817 Agar Polymers 0.000 description 2
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 2
- WSVLPVUVIUVCRA-KPKNDVKVSA-N Alpha-lactose monohydrate Chemical compound O.O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O WSVLPVUVIUVCRA-KPKNDVKVSA-N 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- 125000000041 C6-C10 aryl group Chemical group 0.000 description 2
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 2
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 2
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 2
- KRHYYFGTRYWZRS-UHFFFAOYSA-N Fluorane Chemical compound F KRHYYFGTRYWZRS-UHFFFAOYSA-N 0.000 description 2
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 2
- 108010010803 Gelatin Proteins 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 2
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 2
- 241000124008 Mammalia Species 0.000 description 2
- 229930195725 Mannitol Natural products 0.000 description 2
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 2
- 108010016076 Octreotide Proteins 0.000 description 2
- 235000019483 Peanut oil Nutrition 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 2
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 2
- 229920002472 Starch Polymers 0.000 description 2
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 2
- 229930006000 Sucrose Natural products 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- 239000008272 agar Substances 0.000 description 2
- 235000010443 alginic acid Nutrition 0.000 description 2
- 229920000615 alginic acid Polymers 0.000 description 2
- 125000000217 alkyl group Chemical group 0.000 description 2
- 230000001142 anti-diarrhea Effects 0.000 description 2
- SRSXLGNVWSONIS-UHFFFAOYSA-N benzenesulfonic acid Chemical compound OS(=O)(=O)C1=CC=CC=C1 SRSXLGNVWSONIS-UHFFFAOYSA-N 0.000 description 2
- 229940092714 benzenesulfonic acid Drugs 0.000 description 2
- 239000011230 binding agent Substances 0.000 description 2
- OSGAYBCDTDRGGQ-UHFFFAOYSA-L calcium sulfate Chemical compound [Ca+2].[O-]S([O-])(=O)=O OSGAYBCDTDRGGQ-UHFFFAOYSA-L 0.000 description 2
- 238000004364 calculation method Methods 0.000 description 2
- 238000002512 chemotherapy Methods 0.000 description 2
- 229940125773 compound 10 Drugs 0.000 description 2
- 229940126214 compound 3 Drugs 0.000 description 2
- 230000013872 defecation Effects 0.000 description 2
- XBDQKXXYIPTUBI-UHFFFAOYSA-N dimethylselenoniopropionate Natural products CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 2
- 239000006185 dispersion Substances 0.000 description 2
- 239000012153 distilled water Substances 0.000 description 2
- 239000000839 emulsion Substances 0.000 description 2
- 238000011156 evaluation Methods 0.000 description 2
- 239000000796 flavoring agent Substances 0.000 description 2
- 239000008273 gelatin Substances 0.000 description 2
- 229920000159 gelatin Polymers 0.000 description 2
- 235000019322 gelatine Nutrition 0.000 description 2
- 235000011852 gelatine desserts Nutrition 0.000 description 2
- 125000005842 heteroatom Chemical group 0.000 description 2
- 239000003701 inert diluent Substances 0.000 description 2
- 238000002347 injection Methods 0.000 description 2
- 239000007924 injection Substances 0.000 description 2
- 238000001990 intravenous administration Methods 0.000 description 2
- ZLVXBBHTMQJRSX-VMGNSXQWSA-N jdtic Chemical compound C1([C@]2(C)CCN(C[C@@H]2C)C[C@H](C(C)C)NC(=O)[C@@H]2NCC3=CC(O)=CC=C3C2)=CC=CC(O)=C1 ZLVXBBHTMQJRSX-VMGNSXQWSA-N 0.000 description 2
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 2
- 239000008101 lactose Substances 0.000 description 2
- 229960001375 lactose Drugs 0.000 description 2
- 229960001021 lactose monohydrate Drugs 0.000 description 2
- 239000008297 liquid dosage form Substances 0.000 description 2
- RDOIQAHITMMDAJ-UHFFFAOYSA-N loperamide Chemical compound C=1C=CC=CC=1C(C=1C=CC=CC=1)(C(=O)N(C)C)CCN(CC1)CCC1(O)C1=CC=C(Cl)C=C1 RDOIQAHITMMDAJ-UHFFFAOYSA-N 0.000 description 2
- 229960001571 loperamide Drugs 0.000 description 2
- 239000000314 lubricant Substances 0.000 description 2
- 235000019359 magnesium stearate Nutrition 0.000 description 2
- 239000000594 mannitol Substances 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 2
- 125000001624 naphthyl group Chemical group 0.000 description 2
- 229960002700 octreotide Drugs 0.000 description 2
- 239000003921 oil Substances 0.000 description 2
- 235000019198 oils Nutrition 0.000 description 2
- 239000004006 olive oil Substances 0.000 description 2
- 235000008390 olive oil Nutrition 0.000 description 2
- 239000000312 peanut oil Substances 0.000 description 2
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 2
- 229920005862 polyol Polymers 0.000 description 2
- 150000003077 polyols Chemical class 0.000 description 2
- 239000003755 preservative agent Substances 0.000 description 2
- 125000006413 ring segment Chemical group 0.000 description 2
- 239000008159 sesame oil Substances 0.000 description 2
- 235000011803 sesame oil Nutrition 0.000 description 2
- 235000019333 sodium laurylsulphate Nutrition 0.000 description 2
- 239000000600 sorbitol Substances 0.000 description 2
- 235000019698 starch Nutrition 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 239000005720 sucrose Substances 0.000 description 2
- 229940097346 sulfobutylether-beta-cyclodextrin Drugs 0.000 description 2
- 239000000375 suspending agent Substances 0.000 description 2
- 239000000454 talc Substances 0.000 description 2
- 229910052623 talc Inorganic materials 0.000 description 2
- 235000012222 talc Nutrition 0.000 description 2
- 238000011200 topical administration Methods 0.000 description 2
- 231100000419 toxicity Toxicity 0.000 description 2
- 230000001988 toxicity Effects 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- ODLHGICHYURWBS-LKONHMLTSA-N trappsol cyclo Chemical compound CC(O)COC[C@H]([C@H]([C@@H]([C@H]1O)O)O[C@H]2O[C@@H]([C@@H](O[C@H]3O[C@H](COCC(C)O)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](COCC(C)O)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](COCC(C)O)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](COCC(C)O)[C@H]([C@@H]([C@H]3O)O)O3)[C@H](O)[C@H]2O)COCC(O)C)O[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@@H]3O[C@@H]1COCC(C)O ODLHGICHYURWBS-LKONHMLTSA-N 0.000 description 2
- JNYAEWCLZODPBN-JGWLITMVSA-N (2r,3r,4s)-2-[(1r)-1,2-dihydroxyethyl]oxolane-3,4-diol Chemical class OC[C@@H](O)[C@H]1OC[C@H](O)[C@H]1O JNYAEWCLZODPBN-JGWLITMVSA-N 0.000 description 1
- 125000006699 (C1-C3) hydroxyalkyl group Chemical group 0.000 description 1
- 125000000229 (C1-C4)alkoxy group Chemical group 0.000 description 1
- 125000000171 (C1-C6) haloalkyl group Chemical group 0.000 description 1
- 125000006643 (C2-C6) haloalkenyl group Chemical group 0.000 description 1
- 125000005913 (C3-C6) cycloalkyl group Chemical group 0.000 description 1
- BJEPYKJPYRNKOW-REOHCLBHSA-N (S)-malic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O BJEPYKJPYRNKOW-REOHCLBHSA-N 0.000 description 1
- 229940058015 1,3-butylene glycol Drugs 0.000 description 1
- MEKOFIRRDATTAG-UHFFFAOYSA-N 2,2,5,8-tetramethyl-3,4-dihydrochromen-6-ol Chemical compound C1CC(C)(C)OC2=C1C(C)=C(O)C=C2C MEKOFIRRDATTAG-UHFFFAOYSA-N 0.000 description 1
- WEVYNIUIFUYDGI-UHFFFAOYSA-N 3-[6-[4-(trifluoromethoxy)anilino]-4-pyrimidinyl]benzamide Chemical compound NC(=O)C1=CC=CC(C=2N=CN=C(NC=3C=CC(OC(F)(F)F)=CC=3)C=2)=C1 WEVYNIUIFUYDGI-UHFFFAOYSA-N 0.000 description 1
- BMYNFMYTOJXKLE-UHFFFAOYSA-N 3-azaniumyl-2-hydroxypropanoate Chemical compound NCC(O)C(O)=O BMYNFMYTOJXKLE-UHFFFAOYSA-N 0.000 description 1
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 1
- VVIAGPKUTFNRDU-UHFFFAOYSA-N 6S-folinic acid Natural products C1NC=2NC(N)=NC(=O)C=2N(C=O)C1CNC1=CC=C(C(=O)NC(CCC(O)=O)C(O)=O)C=C1 VVIAGPKUTFNRDU-UHFFFAOYSA-N 0.000 description 1
- 206010000060 Abdominal distension Diseases 0.000 description 1
- 239000005995 Aluminium silicate Substances 0.000 description 1
- 206010002198 Anaphylactic reaction Diseases 0.000 description 1
- MLDQJTXFUGDVEO-UHFFFAOYSA-N BAY-43-9006 Chemical compound C1=NC(C(=O)NC)=CC(OC=2C=CC(NC(=O)NC=3C=C(C(Cl)=CC=3)C(F)(F)F)=CC=2)=C1 MLDQJTXFUGDVEO-UHFFFAOYSA-N 0.000 description 1
- 229940124291 BTK inhibitor Drugs 0.000 description 1
- 125000006577 C1-C6 hydroxyalkyl group Chemical group 0.000 description 1
- GAGWJHPBXLXJQN-UORFTKCHSA-N Capecitabine Chemical compound C1=C(F)C(NC(=O)OCCCCC)=NC(=O)N1[C@H]1[C@H](O)[C@H](O)[C@@H](C)O1 GAGWJHPBXLXJQN-UORFTKCHSA-N 0.000 description 1
- GAGWJHPBXLXJQN-UHFFFAOYSA-N Capecitabine Natural products C1=C(F)C(NC(=O)OCCCCC)=NC(=O)N1C1C(O)C(O)C(C)O1 GAGWJHPBXLXJQN-UHFFFAOYSA-N 0.000 description 1
- 206010010774 Constipation Diseases 0.000 description 1
- ZBNZXTGUTAYRHI-UHFFFAOYSA-N Dasatinib Chemical compound C=1C(N2CCN(CCO)CC2)=NC(C)=NC=1NC(S1)=NC=C1C(=O)NC1=C(C)C=CC=C1Cl ZBNZXTGUTAYRHI-UHFFFAOYSA-N 0.000 description 1
- 208000005156 Dehydration Diseases 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- 235000019739 Dicalciumphosphate Nutrition 0.000 description 1
- 208000002699 Digestive System Neoplasms Diseases 0.000 description 1
- 206010059866 Drug resistance Diseases 0.000 description 1
- 206010014418 Electrolyte imbalance Diseases 0.000 description 1
- 239000001856 Ethyl cellulose Substances 0.000 description 1
- ZZSNKZQZMQGXPY-UHFFFAOYSA-N Ethyl cellulose Chemical compound CCOCC1OC(OC)C(OCC)C(OCC)C1OC1C(O)C(O)C(OC)C(CO)O1 ZZSNKZQZMQGXPY-UHFFFAOYSA-N 0.000 description 1
- 239000004606 Fillers/Extenders Substances 0.000 description 1
- MPJKWIXIYCLVCU-UHFFFAOYSA-N Folinic acid Natural products NC1=NC2=C(N(C=O)C(CNc3ccc(cc3)C(=O)NC(CCC(=O)O)CC(=O)O)CN2)C(=O)N1 MPJKWIXIYCLVCU-UHFFFAOYSA-N 0.000 description 1
- 206010061172 Gastrointestinal injury Diseases 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 206010022678 Intestinal infections Diseases 0.000 description 1
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 1
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 1
- FBOZXECLQNJBKD-ZDUSSCGKSA-N L-methotrexate Chemical compound C=1N=C2N=C(N)N=C(N)C2=NC=1CN(C)C1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 FBOZXECLQNJBKD-ZDUSSCGKSA-N 0.000 description 1
- 239000005517 L01XE01 - Imatinib Substances 0.000 description 1
- 239000002147 L01XE04 - Sunitinib Substances 0.000 description 1
- 239000005511 L01XE05 - Sorafenib Substances 0.000 description 1
- 239000002067 L01XE06 - Dasatinib Substances 0.000 description 1
- 239000002136 L01XE07 - Lapatinib Substances 0.000 description 1
- 239000005536 L01XE08 - Nilotinib Substances 0.000 description 1
- 239000003798 L01XE11 - Pazopanib Substances 0.000 description 1
- 239000002177 L01XE27 - Ibrutinib Substances 0.000 description 1
- XNRVGTHNYCNCFF-UHFFFAOYSA-N Lapatinib ditosylate monohydrate Chemical compound O.CC1=CC=C(S(O)(=O)=O)C=C1.CC1=CC=C(S(O)(=O)=O)C=C1.O1C(CNCCS(=O)(=O)C)=CC=C1C1=CC=C(N=CN=C2NC=3C=C(Cl)C(OCC=4C=C(F)C=CC=4)=CC=3)C2=C1 XNRVGTHNYCNCFF-UHFFFAOYSA-N 0.000 description 1
- 240000007472 Leucaena leucocephala Species 0.000 description 1
- 235000010643 Leucaena leucocephala Nutrition 0.000 description 1
- 206010067125 Liver injury Diseases 0.000 description 1
- 208000002720 Malnutrition Diseases 0.000 description 1
- 240000003183 Manihot esculenta Species 0.000 description 1
- 235000016735 Manihot esculenta subsp esculenta Nutrition 0.000 description 1
- 229920000168 Microcrystalline cellulose Polymers 0.000 description 1
- ZDZOTLJHXYCWBA-VCVYQWHSSA-N N-debenzoyl-N-(tert-butoxycarbonyl)-10-deacetyltaxol Chemical compound O([C@H]1[C@H]2[C@@](C([C@H](O)C3=C(C)[C@@H](OC(=O)[C@H](O)[C@@H](NC(=O)OC(C)(C)C)C=4C=CC=CC=4)C[C@]1(O)C3(C)C)=O)(C)[C@@H](O)C[C@H]1OC[C@]12OC(=O)C)C(=O)C1=CC=CC=C1 ZDZOTLJHXYCWBA-VCVYQWHSSA-N 0.000 description 1
- 206010060860 Neurological symptom Diseases 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 1
- 229910020008 S(O) Inorganic materials 0.000 description 1
- 235000021355 Stearic acid Nutrition 0.000 description 1
- SSZBUIDZHHWXNJ-UHFFFAOYSA-N Stearinsaeure-hexadecylester Natural products CCCCCCCCCCCCCCCCCC(=O)OCCCCCCCCCCCCCCCC SSZBUIDZHHWXNJ-UHFFFAOYSA-N 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- 102000001400 Tryptase Human genes 0.000 description 1
- 108060005989 Tryptase Proteins 0.000 description 1
- 240000008042 Zea mays Species 0.000 description 1
- 235000005824 Zea mays ssp. parviglumis Nutrition 0.000 description 1
- 235000002017 Zea mays subsp mays Nutrition 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- 239000003655 absorption accelerator Substances 0.000 description 1
- 235000011054 acetic acid Nutrition 0.000 description 1
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 239000008186 active pharmaceutical agent Substances 0.000 description 1
- 125000004423 acyloxy group Chemical group 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- 239000002671 adjuvant Substances 0.000 description 1
- 239000003463 adsorbent Substances 0.000 description 1
- 229960001686 afatinib Drugs 0.000 description 1
- ULXXDDBFHOBEHA-CWDCEQMOSA-N afatinib Chemical compound N1=CN=C2C=C(O[C@@H]3COCC3)C(NC(=O)/C=C/CN(C)C)=CC2=C1NC1=CC=C(F)C(Cl)=C1 ULXXDDBFHOBEHA-CWDCEQMOSA-N 0.000 description 1
- 239000000783 alginic acid Substances 0.000 description 1
- 229960001126 alginic acid Drugs 0.000 description 1
- 150000004781 alginic acids Chemical class 0.000 description 1
- 125000003342 alkenyl group Chemical group 0.000 description 1
- 125000003545 alkoxy group Chemical group 0.000 description 1
- 125000000304 alkynyl group Chemical group 0.000 description 1
- 230000002009 allergenic effect Effects 0.000 description 1
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 description 1
- BJEPYKJPYRNKOW-UHFFFAOYSA-N alpha-hydroxysuccinic acid Natural products OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 235000012211 aluminium silicate Nutrition 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 235000001014 amino acid Nutrition 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- 208000003455 anaphylaxis Diseases 0.000 description 1
- 239000005557 antagonist Substances 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- 229940088710 antibiotic agent Drugs 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 239000008365 aqueous carrier Substances 0.000 description 1
- 235000003704 aspartic acid Nutrition 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 125000003785 benzimidazolyl group Chemical group N1=C(NC2=C1C=CC=C2)* 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- OQFSQFPPLPISGP-UHFFFAOYSA-N beta-carboxyaspartic acid Natural products OC(=O)C(N)C(C(O)=O)C(O)=O OQFSQFPPLPISGP-UHFFFAOYSA-N 0.000 description 1
- 210000000941 bile Anatomy 0.000 description 1
- 230000000740 bleeding effect Effects 0.000 description 1
- 208000024330 bloating Diseases 0.000 description 1
- 238000009534 blood test Methods 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 239000006172 buffering agent Substances 0.000 description 1
- 235000019437 butane-1,3-diol Nutrition 0.000 description 1
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical compound O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 description 1
- 125000004369 butenyl group Chemical group C(=CCC)* 0.000 description 1
- 125000004106 butoxy group Chemical group [*]OC([H])([H])C([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000000480 butynyl group Chemical group [*]C#CC([H])([H])C([H])([H])[H] 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- 239000001506 calcium phosphate Substances 0.000 description 1
- CJZGTCYPCWQAJB-UHFFFAOYSA-L calcium stearate Chemical compound [Ca+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O CJZGTCYPCWQAJB-UHFFFAOYSA-L 0.000 description 1
- 235000013539 calcium stearate Nutrition 0.000 description 1
- 239000008116 calcium stearate Substances 0.000 description 1
- 201000011510 cancer Diseases 0.000 description 1
- 229960004117 capecitabine Drugs 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 239000004359 castor oil Substances 0.000 description 1
- 235000019438 castor oil Nutrition 0.000 description 1
- 210000004027 cell Anatomy 0.000 description 1
- 230000005779 cell damage Effects 0.000 description 1
- 208000037887 cell injury Diseases 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 235000010980 cellulose Nutrition 0.000 description 1
- 229920002301 cellulose acetate Polymers 0.000 description 1
- 229960001602 ceritinib Drugs 0.000 description 1
- WRXDGGCKOUEOPW-UHFFFAOYSA-N ceritinib Chemical compound CC=1C=C(NC=2N=C(NC=3C(=CC=CC=3)NS(=O)(=O)C(C)C)C(Cl)=CN=2)C(OC(C)C)=CC=1C1CCNCC1 WRXDGGCKOUEOPW-UHFFFAOYSA-N 0.000 description 1
- 229960000541 cetyl alcohol Drugs 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- 201000001883 cholelithiasis Diseases 0.000 description 1
- 235000015165 citric acid Nutrition 0.000 description 1
- 229960004106 citric acid Drugs 0.000 description 1
- 229940121657 clinical drug Drugs 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 229940125782 compound 2 Drugs 0.000 description 1
- 235000005822 corn Nutrition 0.000 description 1
- 235000012343 cottonseed oil Nutrition 0.000 description 1
- 239000002385 cottonseed oil Substances 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 125000006165 cyclic alkyl group Chemical group 0.000 description 1
- 125000000392 cycloalkenyl group Chemical group 0.000 description 1
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000000640 cyclooctyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- 231100000433 cytotoxic Toxicity 0.000 description 1
- 230000001472 cytotoxic effect Effects 0.000 description 1
- 229950002205 dacomitinib Drugs 0.000 description 1
- LVXJQMNHJWSHET-AATRIKPKSA-N dacomitinib Chemical compound C=12C=C(NC(=O)\C=C\CN3CCCCC3)C(OC)=CC2=NC=NC=1NC1=CC=C(F)C(Cl)=C1 LVXJQMNHJWSHET-AATRIKPKSA-N 0.000 description 1
- 230000002354 daily effect Effects 0.000 description 1
- 229960002448 dasatinib Drugs 0.000 description 1
- 230000007423 decrease Effects 0.000 description 1
- 230000018044 dehydration Effects 0.000 description 1
- 238000006297 dehydration reaction Methods 0.000 description 1
- 230000003111 delayed effect Effects 0.000 description 1
- NEFBYIFKOOEVPA-UHFFFAOYSA-K dicalcium phosphate Chemical compound [Ca+2].[Ca+2].[O-]P([O-])([O-])=O NEFBYIFKOOEVPA-UHFFFAOYSA-K 0.000 description 1
- 229940038472 dicalcium phosphate Drugs 0.000 description 1
- 229910000390 dicalcium phosphate Inorganic materials 0.000 description 1
- 208000010643 digestive system disease Diseases 0.000 description 1
- 230000010339 dilation Effects 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 239000007884 disintegrant Substances 0.000 description 1
- 229960003668 docetaxel Drugs 0.000 description 1
- 231100000673 dose–response relationship Toxicity 0.000 description 1
- 238000001647 drug administration Methods 0.000 description 1
- 230000001804 emulsifying effect Effects 0.000 description 1
- 239000002702 enteric coating Substances 0.000 description 1
- 238000009505 enteric coating Methods 0.000 description 1
- 210000000981 epithelium Anatomy 0.000 description 1
- 235000019325 ethyl cellulose Nutrition 0.000 description 1
- 229920001249 ethyl cellulose Polymers 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 239000011888 foil Substances 0.000 description 1
- VVIAGPKUTFNRDU-ABLWVSNPSA-N folinic acid Chemical compound C1NC=2NC(N)=NC(=O)C=2N(C=O)C1CNC1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 VVIAGPKUTFNRDU-ABLWVSNPSA-N 0.000 description 1
- 235000008191 folinic acid Nutrition 0.000 description 1
- 239000011672 folinic acid Substances 0.000 description 1
- 235000013355 food flavoring agent Nutrition 0.000 description 1
- 235000019253 formic acid Nutrition 0.000 description 1
- 239000001530 fumaric acid Substances 0.000 description 1
- 229960002598 fumaric acid Drugs 0.000 description 1
- 125000002541 furyl group Chemical group 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 230000000762 glandular Effects 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 235000001727 glucose Nutrition 0.000 description 1
- 235000013922 glutamic acid Nutrition 0.000 description 1
- 239000004220 glutamic acid Substances 0.000 description 1
- ZEMPKEQAKRGZGQ-XOQCFJPHSA-N glycerol triricinoleate Natural products CCCCCC[C@@H](O)CC=CCCCCCCCC(=O)OC[C@@H](COC(=O)CCCCCCCC=CC[C@@H](O)CCCCCC)OC(=O)CCCCCCCC=CC[C@H](O)CCCCCC ZEMPKEQAKRGZGQ-XOQCFJPHSA-N 0.000 description 1
- 229940075507 glyceryl monostearate Drugs 0.000 description 1
- 125000000232 haloalkynyl group Chemical group 0.000 description 1
- 125000005347 halocycloalkyl group Chemical group 0.000 description 1
- 230000003862 health status Effects 0.000 description 1
- 231100000234 hepatic damage Toxicity 0.000 description 1
- BXWNKGSJHAJOGX-UHFFFAOYSA-N hexadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCO BXWNKGSJHAJOGX-UHFFFAOYSA-N 0.000 description 1
- 125000006038 hexenyl group Chemical group 0.000 description 1
- 125000005980 hexynyl group Chemical group 0.000 description 1
- 210000003630 histaminocyte Anatomy 0.000 description 1
- 239000003906 humectant Substances 0.000 description 1
- 150000004677 hydrates Chemical class 0.000 description 1
- 229920003063 hydroxymethyl cellulose Polymers 0.000 description 1
- 229940031574 hydroxymethyl cellulose Drugs 0.000 description 1
- 125000004029 hydroxymethyl group Chemical group [H]OC([H])([H])* 0.000 description 1
- 229960001507 ibrutinib Drugs 0.000 description 1
- XYFPWWZEPKGCCK-GOSISDBHSA-N ibrutinib Chemical compound C1=2C(N)=NC=NC=2N([C@H]2CN(CCC2)C(=O)C=C)N=C1C(C=C1)=CC=C1OC1=CC=CC=C1 XYFPWWZEPKGCCK-GOSISDBHSA-N 0.000 description 1
- 229960002411 imatinib Drugs 0.000 description 1
- KTUFNOKKBVMGRW-UHFFFAOYSA-N imatinib Chemical compound C1CN(C)CCN1CC1=CC=C(C(=O)NC=2C=C(NC=3N=C(C=CN=3)C=3C=NC=CC=3)C(C)=CC=2)C=C1 KTUFNOKKBVMGRW-UHFFFAOYSA-N 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- 238000011534 incubation Methods 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 230000008595 infiltration Effects 0.000 description 1
- 238000001764 infiltration Methods 0.000 description 1
- 210000004969 inflammatory cell Anatomy 0.000 description 1
- 230000028709 inflammatory response Effects 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 208000003243 intestinal obstruction Diseases 0.000 description 1
- 238000007918 intramuscular administration Methods 0.000 description 1
- 230000002601 intratumoral effect Effects 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- UWKQSNNFCGGAFS-XIFFEERXSA-N irinotecan Chemical compound C1=C2C(CC)=C3CN(C(C4=C([C@@](C(=O)OC4)(O)CC)C=4)=O)C=4C3=NC2=CC=C1OC(=O)N(CC1)CCC1N1CCCCC1 UWKQSNNFCGGAFS-XIFFEERXSA-N 0.000 description 1
- 229960004768 irinotecan Drugs 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 125000000842 isoxazolyl group Chemical group 0.000 description 1
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 1
- 230000002147 killing effect Effects 0.000 description 1
- 239000004310 lactic acid Substances 0.000 description 1
- 235000014655 lactic acid Nutrition 0.000 description 1
- 229960000448 lactic acid Drugs 0.000 description 1
- 238000002350 laparotomy Methods 0.000 description 1
- 229960004891 lapatinib Drugs 0.000 description 1
- 229960003784 lenvatinib Drugs 0.000 description 1
- WOSKHXYHFSIKNG-UHFFFAOYSA-N lenvatinib Chemical compound C=12C=C(C(N)=O)C(OC)=CC2=NC=CC=1OC(C=C1Cl)=CC=C1NC(=O)NC1CC1 WOSKHXYHFSIKNG-UHFFFAOYSA-N 0.000 description 1
- 229960001691 leucovorin Drugs 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 230000008818 liver damage Effects 0.000 description 1
- 231100000053 low toxicity Toxicity 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000011976 maleic acid Substances 0.000 description 1
- 229940098895 maleic acid Drugs 0.000 description 1
- 239000001630 malic acid Substances 0.000 description 1
- 235000011090 malic acid Nutrition 0.000 description 1
- 229940099690 malic acid Drugs 0.000 description 1
- 230000001071 malnutrition Effects 0.000 description 1
- 235000000824 malnutrition Nutrition 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 208000030159 metabolic disease Diseases 0.000 description 1
- 230000002503 metabolic effect Effects 0.000 description 1
- 229940098779 methanesulfonic acid Drugs 0.000 description 1
- 229960000485 methotrexate Drugs 0.000 description 1
- 229940016286 microcrystalline cellulose Drugs 0.000 description 1
- 235000019813 microcrystalline cellulose Nutrition 0.000 description 1
- 239000008108 microcrystalline cellulose Substances 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 238000010369 molecular cloning Methods 0.000 description 1
- 239000001788 mono and diglycerides of fatty acids Substances 0.000 description 1
- CQDGTJPVBWZJAZ-UHFFFAOYSA-N monoethyl carbonate Chemical compound CCOC(O)=O CQDGTJPVBWZJAZ-UHFFFAOYSA-N 0.000 description 1
- 210000004400 mucous membrane Anatomy 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 229960001346 nilotinib Drugs 0.000 description 1
- HHZIURLSWUIHRB-UHFFFAOYSA-N nilotinib Chemical compound C1=NC(C)=CN1C1=CC(NC(=O)C=2C=C(NC=3N=C(C=CN=3)C=3C=NC=CC=3)C(C)=CC=2)=CC(C(F)(F)F)=C1 HHZIURLSWUIHRB-UHFFFAOYSA-N 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 208000030212 nutrition disease Diseases 0.000 description 1
- 208000015380 nutritional deficiency disease Diseases 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 1
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 1
- 239000002674 ointment Substances 0.000 description 1
- 239000003605 opacifier Substances 0.000 description 1
- 229940126701 oral medication Drugs 0.000 description 1
- 235000006408 oxalic acid Nutrition 0.000 description 1
- AICOOMRHRUFYCM-ZRRPKQBOSA-N oxazine, 1 Chemical compound C([C@@H]1[C@H](C(C[C@]2(C)[C@@H]([C@H](C)N(C)C)[C@H](O)C[C@]21C)=O)CC1=CC2)C[C@H]1[C@@]1(C)[C@H]2N=C(C(C)C)OC1 AICOOMRHRUFYCM-ZRRPKQBOSA-N 0.000 description 1
- 125000002971 oxazolyl group Chemical group 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 239000012188 paraffin wax Substances 0.000 description 1
- 230000007170 pathology Effects 0.000 description 1
- 229960000639 pazopanib Drugs 0.000 description 1
- CUIHSIWYWATEQL-UHFFFAOYSA-N pazopanib Chemical compound C1=CC2=C(C)N(C)N=C2C=C1N(C)C(N=1)=CC=NC=1NC1=CC=C(C)C(S(N)(=O)=O)=C1 CUIHSIWYWATEQL-UHFFFAOYSA-N 0.000 description 1
- 125000002255 pentenyl group Chemical group C(=CCCC)* 0.000 description 1
- 125000005981 pentynyl group Chemical group 0.000 description 1
- 239000002304 perfume Substances 0.000 description 1
- 230000002572 peristaltic effect Effects 0.000 description 1
- 235000011007 phosphoric acid Nutrition 0.000 description 1
- OXNIZHLAWKMVMX-UHFFFAOYSA-N picric acid Chemical compound OC1=C([N+]([O-])=O)C=C([N+]([O-])=O)C=C1[N+]([O-])=O OXNIZHLAWKMVMX-UHFFFAOYSA-N 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- 239000013641 positive control Substances 0.000 description 1
- 229920001592 potato starch Polymers 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 239000003380 propellant Substances 0.000 description 1
- 125000004368 propenyl group Chemical group C(=CC)* 0.000 description 1
- 235000019260 propionic acid Nutrition 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000002568 propynyl group Chemical group [*]C#CC([H])([H])[H] 0.000 description 1
- 239000008213 purified water Substances 0.000 description 1
- 125000004309 pyranyl group Chemical group O1C(C=CC=C1)* 0.000 description 1
- 125000003373 pyrazinyl group Chemical group 0.000 description 1
- 125000003226 pyrazolyl group Chemical group 0.000 description 1
- 125000002098 pyridazinyl group Chemical group 0.000 description 1
- 125000004076 pyridyl group Chemical group 0.000 description 1
- 125000000714 pyrimidinyl group Chemical group 0.000 description 1
- 125000000168 pyrrolyl group Chemical group 0.000 description 1
- 150000003856 quaternary ammonium compounds Chemical class 0.000 description 1
- IUVKMZGDUIUOCP-BTNSXGMBSA-N quinbolone Chemical compound O([C@H]1CC[C@H]2[C@H]3[C@@H]([C@]4(C=CC(=O)C=C4CC3)C)CC[C@@]21C)C1=CCCC1 IUVKMZGDUIUOCP-BTNSXGMBSA-N 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 238000012502 risk assessment Methods 0.000 description 1
- 208000026775 severe diarrhea Diseases 0.000 description 1
- 239000011257 shell material Substances 0.000 description 1
- 150000004760 silicates Chemical class 0.000 description 1
- RMAQACBXLXPBSY-UHFFFAOYSA-N silicic acid Chemical compound O[Si](O)(O)O RMAQACBXLXPBSY-UHFFFAOYSA-N 0.000 description 1
- 235000012239 silicon dioxide Nutrition 0.000 description 1
- 150000003384 small molecules Chemical class 0.000 description 1
- 229910000029 sodium carbonate Inorganic materials 0.000 description 1
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 description 1
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 description 1
- 239000001509 sodium citrate Substances 0.000 description 1
- NLJMYIDDQXHKNR-UHFFFAOYSA-K sodium citrate Chemical compound O.O.[Na+].[Na+].[Na+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O NLJMYIDDQXHKNR-UHFFFAOYSA-K 0.000 description 1
- 239000012453 solvate Substances 0.000 description 1
- 229960003787 sorafenib Drugs 0.000 description 1
- 239000003549 soybean oil Substances 0.000 description 1
- 235000012424 soybean oil Nutrition 0.000 description 1
- 238000011895 specific detection Methods 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 239000008117 stearic acid Substances 0.000 description 1
- 210000002784 stomach Anatomy 0.000 description 1
- 238000007920 subcutaneous administration Methods 0.000 description 1
- 125000001424 substituent group Chemical group 0.000 description 1
- 125000000475 sulfinyl group Chemical group [*:2]S([*:1])=O 0.000 description 1
- 229960001796 sunitinib Drugs 0.000 description 1
- WINHZLLDWRZWRT-ATVHPVEESA-N sunitinib Chemical compound CCN(CC)CCNC(=O)C1=C(C)NC(\C=C/2C3=CC(F)=CC=C3NC\2=O)=C1C WINHZLLDWRZWRT-ATVHPVEESA-N 0.000 description 1
- 239000003765 sweetening agent Substances 0.000 description 1
- 238000002636 symptomatic treatment Methods 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 239000011975 tartaric acid Substances 0.000 description 1
- 235000002906 tartaric acid Nutrition 0.000 description 1
- 229960001367 tartaric acid Drugs 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000001973 tert-pentyl group Chemical group [H]C([H])([H])C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 239000012085 test solution Substances 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 229940126585 therapeutic drug Drugs 0.000 description 1
- 125000000335 thiazolyl group Chemical group 0.000 description 1
- 125000001544 thienyl group Chemical group 0.000 description 1
- 210000001519 tissue Anatomy 0.000 description 1
- 125000001425 triazolyl group Chemical group 0.000 description 1
- 229950002929 trinitrophenol Drugs 0.000 description 1
- 210000004881 tumor cell Anatomy 0.000 description 1
- 230000002792 vascular Effects 0.000 description 1
- 235000015112 vegetable and seed oil Nutrition 0.000 description 1
- 239000008158 vegetable oil Substances 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 239000001993 wax Substances 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/715—Polysaccharides, i.e. having more than five saccharide radicals attached to each other by glycosidic linkages; Derivatives thereof, e.g. ethers, esters
- A61K31/716—Glucans
- A61K31/724—Cyclodextrins
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/12—Antidiarrhoeals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02A—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE
- Y02A50/00—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE in human health protection, e.g. against extreme weather
- Y02A50/30—Against vector-borne diseases, e.g. mosquito-borne, fly-borne, tick-borne or waterborne diseases whose impact is exacerbated by climate change
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Chemical & Material Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Veterinary Medicine (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Molecular Biology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
技术领域Technical Field
本发明属于化工制药领域,具体地,涉及一种降低靶向抗肿瘤药对肠道损伤的药物的用途。The invention belongs to the field of chemical pharmacy, and in particular relates to the use of a drug for reducing intestinal damage caused by targeted anti-tumor drugs.
背景技术Background Art
许多化疗药物都是细胞毒药物,可快速靶向分裂肿瘤细胞,同时对正常细胞也具有杀伤作用。可导致胃肠道上皮细胞损伤,引起各种胃肠道症状。在临床上表现为化疗相关性腹泻(CID),在随机临床试验中被报道为3–4级,严重腹泻的频率为5–47%。除了作用于胃肠道肿瘤的化疗药物外,治疗其他肿瘤的化疗药如:多西他赛、卡培他滨或亚叶酸拮抗剂(如:甲氨蝶呤)都会增加化疗引起的腹泻风险。小分子靶向药物尤其是EFGR-TKI,VEGFR-TKI,BCR-ABL TKI,BTK抑制剂可引起较高的腹泻发生率,并且可能会造成脱水,营养不良,电解质紊乱和免疫力衰减等严重后果,危及生命。很多药物需要调整剂量或中断给药,同样也需要进行抗腹泻的干预措施。Many chemotherapy drugs are cytotoxic drugs that can quickly target dividing tumor cells and also have a killing effect on normal cells. They can cause gastrointestinal epithelial cell damage and cause various gastrointestinal symptoms. Clinically, it manifests as chemotherapy-related diarrhea (CID), which has been reported as grade 3–4 in randomized clinical trials, with a frequency of severe diarrhea of 5–47%. In addition to chemotherapy drugs that act on gastrointestinal tumors, chemotherapy drugs for other tumors such as docetaxel, capecitabine, or folinic acid antagonists (such as methotrexate) increase the risk of chemotherapy-induced diarrhea. Small molecule targeted drugs, especially EFGR-TKI, VEGFR-TKI, BCR-ABL TKI, and BTK inhibitors, can cause a high incidence of diarrhea and may cause serious consequences such as dehydration, malnutrition, electrolyte imbalance, and immune decline, which are life-threatening. Many drugs require dose adjustment or interruption of administration, and anti-diarrhea intervention measures are also required.
目前对抗癌药物引起的相关腹泻病理机制研究较少,尚不明确。化疗药物引起的腹泻可能与药物对小肠上皮组织的损伤、炎症反应、感染及使用抗生素等有关。At present, there are few studies on the pathological mechanism of diarrhea caused by anticancer drugs, and it is still unclear. Diarrhea caused by chemotherapy drugs may be related to drug damage to the small intestinal epithelial tissue, inflammatory response, infection and the use of antibiotics.
由于肿瘤患者口服靶向抗肿瘤药物导致肠道受损,腹泻次数大幅提升,严重影响治疗过程,目前临床上缺乏有效治疗肿瘤药物引起腹泻的药物,多数药物均为对症治疗。常用的药物只有口服制剂哌洛丁胺和注射制剂奥曲肽。洛哌丁胺对3-4级腹泻的止泻效果不理想,持续高密度的使用高剂量的会导致患者接受伊立替康时出现耐药。还会引起便秘、肠梗阻、影响药物吸收、加重肠道感染、肝功能损害等副作用,不能长期使用。奥曲肽存在胃肠道症状、神经系统症状、肝胆疾病(胆石症发生率约为15-30%)、代谢和营养机能紊乱。注射给药患者,不方便,患者依从性差。肿瘤靶向药需要长期使用至疾病进展,故这类减少靶向药物毒性反应的辅助临床用药受限,急需开发一种更加安全、有效、使用方便的新型口服预防及治疗腹泻的药物。Because oral targeted anti-tumor drugs cause intestinal damage in cancer patients, the frequency of diarrhea increases significantly, which seriously affects the treatment process. At present, there is a lack of effective drugs for treating diarrhea caused by tumor drugs in clinical practice, and most drugs are symptomatic treatments. The commonly used drugs are only oral preparations of loperamide and injectable preparations of octreotide. The antidiarrheal effect of loperamide on grade 3-4 diarrhea is not ideal. Continuous high-density use of high doses will lead to drug resistance when patients receive irinotecan. It can also cause side effects such as constipation, intestinal obstruction, affect drug absorption, aggravate intestinal infection, and liver damage, and cannot be used for a long time. Octreotide has gastrointestinal symptoms, neurological symptoms, hepatobiliary diseases (the incidence of cholelithiasis is about 15-30%), and metabolic and nutritional disorders. It is inconvenient for patients to be injected and the patient compliance is poor. Tumor targeted drugs need to be used for a long time until the disease progresses, so this type of auxiliary clinical drug that reduces the toxicity of targeted drugs is limited. It is urgent to develop a new oral drug for the prevention and treatment of diarrhea that is safer, more effective, and easier to use.
发明内容Summary of the invention
本发明的目的在于提供一种降低靶向抗肿瘤药物对肠道损伤的药物及其应用。The purpose of the present invention is to provide a drug for reducing the damage of targeted anti-tumor drugs to the intestine and its application.
本发明的第一方面,提供了一种式I化合物或其药学上可接受的盐的用途,用于制备一药物组合物或制剂,所述药物组合物或制剂用于:In a first aspect of the present invention, there is provided a use of a compound of formula I or a pharmaceutically acceptable salt thereof for preparing a pharmaceutical composition or preparation, wherein the pharmaceutical composition or preparation is used for:
(a)提高靶向抗肿瘤药物的治疗效果;和/或(a) improving the therapeutic effect of targeted anti-tumor drugs; and/or
(b)降低靶向抗肿瘤药物对肠道的伤损,减少腹泻;(b) Reduce the damage of targeted anti-tumor drugs to the intestine and reduce diarrhea;
式中,In the formula,
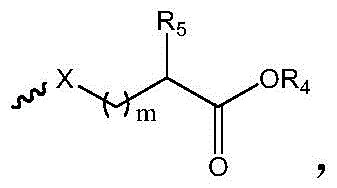
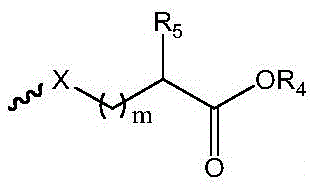
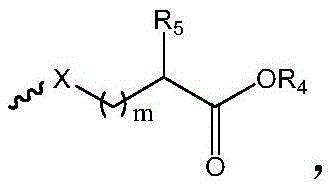
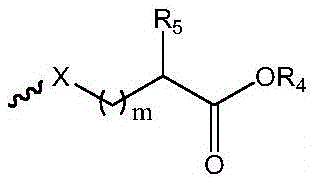
R1选自下组:羟基、C1-C6烷氧基、卤素、取代或未取代的-O-C1-C6烷基、其中所述的取代为磺酸基或羟基取代; R1 is selected from the group consisting of hydroxy, C1-C6 alkoxy, halogen, substituted or unsubstituted -O-C1-C6 alkyl, The substitution is sulfonic acid or hydroxyl substitution;
其中,in,
中的碳原子为手性碳原子,所述的手性碳原子选自下组: middle The carbon atom is a chiral carbon atom, and the chiral carbon atom is selected from the following group:
m为1-6的正整数;m is a positive integer from 1 to 6;
X为S或O;X is S or O;
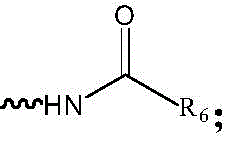
R5为H或 R5 is H or
R4为选自下组的金属离子:Na+、K+、Li+或Cs+;R 4 is a metal ion selected from the group consisting of Na + , K + , Li + or Cs + ;
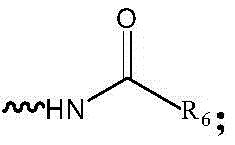
R6为C1-C6烷基、C3-C8环烷基、C2-C6烯基、C2-C6炔基、芳基或杂芳基; R6 is C1-C6 alkyl, C3-C8 cycloalkyl, C2-C6 alkenyl, C2-C6 alkynyl, aryl or heteroaryl;
R2和R3各自独立地为H、C1-C6烷基、C2-C6烯基、C2-C6炔基、C3-C8环烷基、C2-C6羟烷基或-(C1-C3亚烷基)-COOH; R2 and R3 are each independently H, C1-C6 alkyl, C2-C6 alkenyl, C2-C6 alkynyl, C3-C8 cycloalkyl, C2-C6 hydroxyalkyl or -(C1-C3 alkylene)-COOH;
n为6-12的正整数。n is a positive integer from 6 to 12.
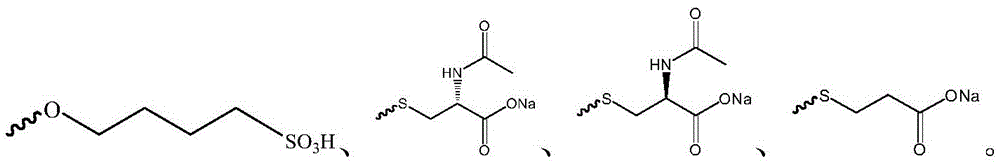
在另一优选例中,R1选自下组:羟基、甲氧基、 In another preferred embodiment, R1 is selected from the following group: hydroxyl, methoxy,
在另一优选例中,m=1。In another preferred embodiment, m=1.
在另一优选例中,X为S。In another preferred embodiment, X is S.
在另一优选例中,R4为Na+。In another preferred embodiment, R 4 is Na + .
在另一优选例中,R6为CH3。In another preferred embodiment, R 6 is CH 3 .
在另一优选例中,n为6、7或8。In another preferred embodiment, n is 6, 7 or 8.
在另一优选例中,R1选自下组:羟基、甲氧基、 In another preferred embodiment, R1 is selected from the following group: hydroxyl, methoxy,
在另一优选例中,R2和R3各自独立地为H、甲基、羟丙基、羟乙基或羧甲基。In another preferred embodiment, R 2 and R 3 are each independently H, methyl, hydroxypropyl, hydroxyethyl or carboxymethyl.
在另一优选例中,所述的式I化合物选自下组:甲基环糊精、羧甲基环糊精、羟乙基-β-乙环糊精、羟丙基-β-环糊精或磺丁基醚-β-环糊精。In another preferred embodiment, the compound of formula I is selected from the following group: methyl cyclodextrin, carboxymethyl cyclodextrin, hydroxyethyl-β-cyclodextrin, hydroxypropyl-β-cyclodextrin or sulfobutyl ether-β-cyclodextrin.
在另一优选例中,所述的式I化合物母核为环糊精。In another preferred embodiment, the core of the compound of formula I is cyclodextrin.
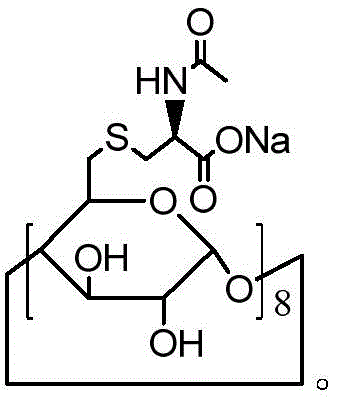
在另一优选例中,所述的式I化合物为 In another preferred embodiment, the compound of formula I is
在另一优选例中,所述的式I化合物选自下组:In another preferred embodiment, the compound of formula I is selected from the following group:
在另一优选例中,所述的药物组合物还含有额外的活性成分。In another preferred embodiment, the pharmaceutical composition further contains additional active ingredients.
在另一优选例中,所述药物组合物或制剂含有(a)作为活性成分的式(I)化合物或其药学上可接受的盐和(b)药学上可接受的载体。In another preferred embodiment, the pharmaceutical composition or preparation contains (a) a compound of formula (I) or a pharmaceutically acceptable salt thereof as an active ingredient and (b) a pharmaceutically acceptable carrier.
在另一优选例中,在所述的药物组合物中,组分(a)的总量为0.001-99wt%,较佳地0.1-90wt%,更佳地1-80wt%,按组合物的总重量计。In another preferred embodiment, in the pharmaceutical composition, the total amount of component (a) is 0.001-99wt%, preferably 0.1-90wt%, more preferably 1-80wt%, based on the total weight of the composition.
在另一优选例中,所述药物组合物或制剂用于提高靶向抗肿瘤药物的治疗效果。In another preferred embodiment, the pharmaceutical composition or preparation is used to improve the therapeutic effect of targeted anti-tumor drugs.
在另一优选例中,所述药物组合物或制剂用于降低服用靶向抗肿瘤药物导致的肠道损伤,减少腹泻。In another preferred embodiment, the pharmaceutical composition or preparation is used to reduce intestinal damage caused by taking targeted anti-tumor drugs and reduce diarrhea.
在另一优选例中,所述的药物组合物或制剂用于减少肠道损伤所导致的肠胀气。In another preferred embodiment, the pharmaceutical composition or preparation is used to reduce intestinal flatulence caused by intestinal damage.
在另一优选例中,所述的药物组合物或制剂用于保护肠壁绒毛的完整性以及减少肠道绒毛破坏。In another preferred embodiment, the pharmaceutical composition or preparation is used to protect the integrity of intestinal villi and reduce the destruction of intestinal villi.
在另一优选例中,所述的药物组合物或制剂不影响口服型靶向抗肿瘤药物的吸收。In another preferred embodiment, the pharmaceutical composition or preparation does not affect the absorption of oral targeted anti-tumor drugs.
在另一优选例中,所述药物组合物或制剂的剂型选自下组:片剂、胶囊剂、散剂、丸剂、颗粒剂或缓控释制剂。In another preferred embodiment, the dosage form of the pharmaceutical composition or preparation is selected from the following group: tablets, capsules, powders, pills, granules or sustained-release preparations.
本发明的第二方面,提供了一种药物组合物或制剂,所述的药物组合物或制剂含有(a)活性成分,所述活性成分包括式I化合物或其药学上可接受的盐;以及(b)药学上可接受的载体;所述药物组合物或制剂用于:The second aspect of the present invention provides a pharmaceutical composition or preparation, which contains (a) an active ingredient, wherein the active ingredient includes a compound of formula I or a pharmaceutically acceptable salt thereof; and (b) a pharmaceutically acceptable carrier; the pharmaceutical composition or preparation is used for:
(1)提高靶向抗肿瘤药物的治疗效果;和/或(1) Improving the therapeutic effect of targeted anti-tumor drugs; and/or
(2)降低靶向抗肿瘤药物对肠道的损伤,减少腹泻。(2) Reduce the damage of targeted anti-tumor drugs to the intestine and reduce diarrhea.
在另一优选例中,所述的药物组合物或制剂还可含有其他药物活性成分或药学上可接受的载体。In another preferred embodiment, the pharmaceutical composition or preparation may further contain other pharmaceutically active ingredients or pharmaceutically acceptable carriers.
在另一优选例中,当所述活性成分含有两种组分时,两种组分的重量比为1:20至20:1,较佳地1:10至10:1,更佳地1:5至5:1。In another preferred embodiment, when the active ingredient contains two components, the weight ratio of the two components is 1:20 to 20:1, preferably 1:10 to 10:1, and more preferably 1:5 to 5:1.
在另一优选例中,所述的其他药物活性成分包括靶向抗肿瘤药物。In another preferred embodiment, the other active pharmaceutical ingredients include targeted anti-tumor drugs.
在另一优选例中,所述的靶向抗肿瘤药物选自下组:吉非替尼,厄洛替尼,奥希替尼、达克替尼、阿法替尼、尼洛替尼、仑伐替尼、色瑞替尼、伊布替尼、达沙替尼、伊马替尼、舒尼替尼、帕唑帕尼、索拉非尼或拉帕替尼。In another preferred embodiment, the targeted anti-tumor drug is selected from the following group: gefitinib, erlotinib, osimertinib, dacomitinib, afatinib, nilotinib, lenvatinib, ceritinib, ibrutinib, dasatinib, imatinib, sunitinib, pazopanib, sorafenib or lapatinib.
在另一优选例中,当所述活性成分含有两种组分时,两种组分的重量比为1:20至20:1,较佳地1:10至10:1,更佳地1:5至5:1。In another preferred embodiment, when the active ingredient contains two components, the weight ratio of the two components is 1:20 to 20:1, preferably 1:10 to 10:1, and more preferably 1:5 to 5:1.
本发明的第三方面,提供了一种药盒,所述的药盒包括:The third aspect of the present invention provides a medicine kit, comprising:
(1)第一容器,以及位于所述容器内的第一药物组合物,所述的第一药物组合物含有第一化合物或其药学上可接受的盐,以及药学上可接受的载体;(1) a first container, and a first pharmaceutical composition in the container, wherein the first pharmaceutical composition comprises a first compound or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable carrier;
(2)第n容器,以及位于所述容器内的第n药物组合物,所述的第n药物组合物含有第n化合物或其药学上可接受的盐;以及药学上可接受的载体;其中,n为2-8中任一正整数;(2) an nth container, and an nth pharmaceutical composition in the container, wherein the nth pharmaceutical composition comprises an nth compound or a pharmaceutically acceptable salt thereof; and a pharmaceutically acceptable carrier; wherein n is any positive integer from 2 to 8;
其中,第一化合物和第n化合物均为选自权利要求1所述的化合物;或第一化合物为选自权利要求1所述的化合物和第n化合物中至少一个为靶向抗肿瘤活性物质;Wherein, the first compound and the nth compound are both compounds selected from the compounds described in claim 1; or at least one of the first compound selected from the compounds described in claim 1 and the nth compound is a targeted anti-tumor active substance;
和/或(3)任选的使用说明书。and/or (3) optional instructions for use.
本发明的第四方面,提供了一种疾病的治疗方法,所述的疾病如权利要求1所述,其特征在于,包括步骤:给需要对象施用式I化合物或其药学上可接受的盐,其中,式I化合物如权利要求1中所定义。The fourth aspect of the present invention provides a method for treating a disease, wherein the disease is as described in claim 1, characterized in that it comprises the step of administering a compound of formula I or a pharmaceutically acceptable salt thereof to a subject in need thereof, wherein the compound of formula I is as defined in claim 1.
在另一优选例中,所述的对象为哺乳动物。In another preferred embodiment, the subject is a mammal.
在另一优选例中,所述的对象为人。In another preferred embodiment, the subject is a human.
本发明第五方面,提供了一种靶向抗肿瘤药物和式I化合物的组合的用途,用于制备治疗肿瘤的药物。In a fifth aspect, the present invention provides a use of a combination of a targeted anti-tumor drug and a compound of formula I for preparing a drug for treating tumors.
在另一优选例中,所述的靶向抗肿瘤药物和式I化合物的组合包含:In another preferred embodiment, the combination of the targeted anti-tumor drug and the compound of formula I comprises:
(a)第一药物组合物,所述的第一药物组合物包括:靶向抗肿瘤活性成分和药学上可接受的载体;和(a) a first pharmaceutical composition, the first pharmaceutical composition comprising: a targeted anti-tumor active ingredient and a pharmaceutically acceptable carrier; and
(b)第二药物组合物,所述的第二药物组合物包括:作为活性成分的式I化合物和药学上可接受的载体;(b) a second pharmaceutical composition, comprising: a compound of formula I as an active ingredient and a pharmaceutically acceptable carrier;
在另一优选例中,所述的第一药物组合物和第二药物组合物是各自独立的或者是同一药物组合物。In another preferred embodiment, the first pharmaceutical composition and the second pharmaceutical composition are independent of each other or are the same pharmaceutical composition.
在另一优选例中,所述的靶向抗肿瘤药物选自下组:吉非替尼、厄洛替尼、奥希替尼,或其组合。In another preferred embodiment, the targeted anti-tumor drug is selected from the following group: gefitinib, erlotinib, osimertinib, or a combination thereof.
应理解,在本发明范围内中,本发明的上述各技术特征和在下文(如实施例)中具体描述的各技术特征之间都可以互相组合,从而构成新的或优选的技术方案。限于篇幅,在此不再一一累述。It should be understood that within the scope of the present invention, the above-mentioned technical features of the present invention and the technical features specifically described below (such as embodiments) can be combined with each other to form a new or preferred technical solution. Due to space limitations, they will not be described one by one here.
具体实施方式DETAILED DESCRIPTION
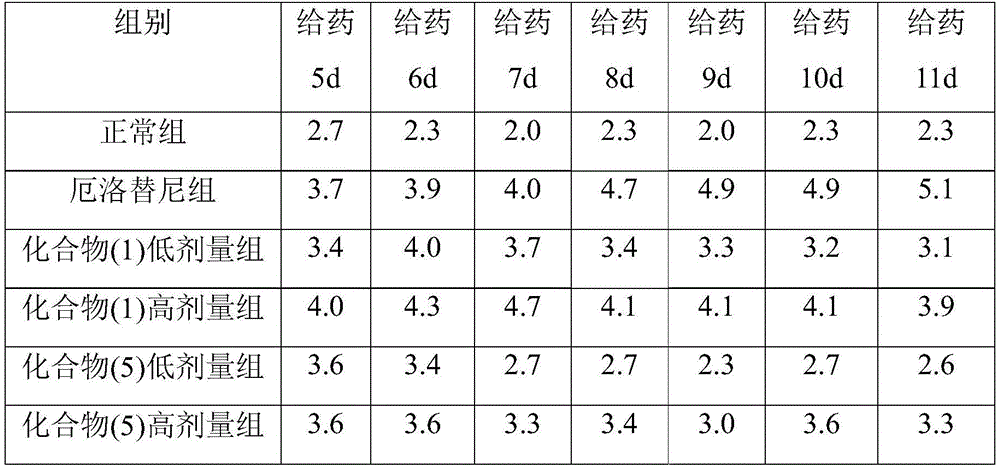
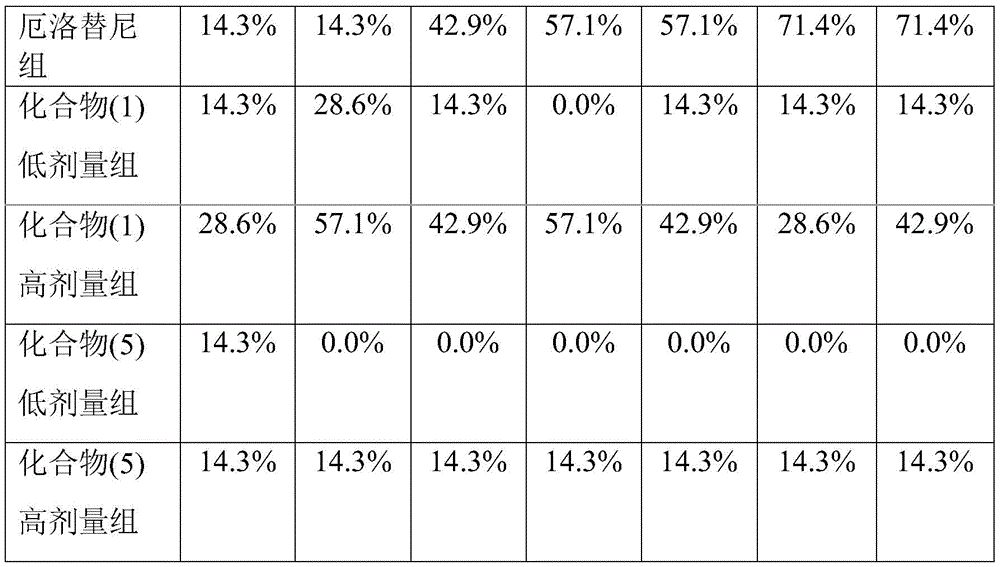
本发明人经过广泛而深入地研究,首次意外地发现一种母核为环糊精的式I所示的化合物对降低靶向抗肿瘤药物对肠道损伤的用途。进一步的实验表明,所述的式I化合物可以降低靶向抗肿瘤药物对肠道损伤,减少腹泻症状,同时提高靶向抗肿瘤药物的治疗效果,且安全性高。在此基础上,完成本发明。After extensive and in-depth research, the inventor unexpectedly discovered for the first time that a compound represented by Formula I with a cyclodextrin as a parent core can be used to reduce the intestinal damage caused by targeted anti-tumor drugs. Further experiments show that the compound represented by Formula I can reduce the intestinal damage caused by targeted anti-tumor drugs, reduce diarrhea symptoms, and improve the therapeutic effect of targeted anti-tumor drugs, and has high safety. On this basis, the present invention is completed.
术语:the term:
术语“卤素”指F、Cl、Br和I。The term "halogen" refers to F, Cl, Br and I.
术语“C1-C6烷基”是指包括1-6个碳原子的直链或支链的烷基,例如甲基、乙基、丙基、异丙基、丁基、异丁基、叔丁基、新戊基、特戊基、或类似基团。The term "C1-C6 alkyl" refers to a straight or branched chain alkyl group comprising 1 to 6 carbon atoms, such as methyl, ethyl, propyl, isopropyl, butyl, isobutyl, tert-butyl, neopentyl, tert-pentyl, or the like.
术语“C2-C6烯基”是指具有2-6个碳原子的含有一个双键的直链或支链烯基,非限制性地包括乙烯基、丙烯基、丁烯基、异丁烯基、戊烯基和己烯基等。The term "C2-C6 alkenyl" refers to a straight or branched alkenyl group having 2 to 6 carbon atoms and containing one double bond, including but not limited to ethenyl, propenyl, butenyl, isobutenyl, pentenyl, hexenyl and the like.
术语“C2-C6炔基”是指具有2-6个碳原子的含有一个三键的直链或支链炔基,非限制性地包括乙炔基、丙炔基、丁炔基、异丁炔基、戊炔基和己炔基等。The term "C2-C6 alkynyl" refers to a straight or branched alkynyl group having 2 to 6 carbon atoms and containing one triple bond, including but not limited to ethynyl, propynyl, butynyl, isobutynyl, pentynyl, hexynyl and the like.
术语“C1-C6羟烷基”是指具有1-6个碳原子的含有一个羟基的直链或支链烷基,非限制性地包括羟甲基、羟乙基、羟丙基、羟丁基等,优选为C1-C3羟烷基。The term "C1-C6 hydroxyalkyl" refers to a straight or branched chain alkyl group having 1 to 6 carbon atoms and containing one hydroxyl group, including but not limited to hydroxymethyl, hydroxyethyl, hydroxypropyl, hydroxybutyl, etc., preferably a C1-C3 hydroxyalkyl group.
术语“C3-C8环烷基”是指在环上具有3-8个碳原子的环状烷基,非限制性地包括环丙基、环丁基、环戊基、环己基、环庚基、环辛基等。The term "C3-C8 cycloalkyl" refers to a cyclic alkyl group having 3 to 8 carbon atoms in the ring, including but not limited to cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl and the like.
术语“C1-C6烷氧基”是指具有1-6个碳原子的直链或支链烷氧基,非限制性地包括甲氧基、乙氧基、丙氧基、异丙氧基和丁氧基等。优选为C1-C4烷氧基。The term "C1-C6 alkoxy" refers to a straight or branched alkoxy group having 1 to 6 carbon atoms, including but not limited to methoxy, ethoxy, propoxy, isopropoxy and butoxy, etc. Preferably, it is a C1-C4 alkoxy group.
术语“芳环”或“芳基”具有相同的含义,优选为“C6-C10芳基”。术语“C6-C10芳基”是指在环上不含杂原子的具有6-10个碳原子的芳香族环基,如苯基、萘基等。The term "aromatic ring" or "aryl" has the same meaning, preferably "C6-C10 aryl". The term "C6-C10 aryl" refers to an aromatic ring group having 6 to 10 carbon atoms without heteroatoms in the ring, such as phenyl, naphthyl, etc.
术语“杂芳基”表示包含1至4个杂原子的杂芳族体系,所述杂原子包括氮、氧和S(O)r(其中r是整数0、1、2)的杂原子,例如,4-8元杂芳基指含有4-8个环原子的杂芳族体系,4-10元杂芳基指含有4-10个环原子的杂芳族体系,包括但不限于吡咯基、呋喃基、噻吩基、吡唑基、噻唑基、咪唑基、噁唑基、异噁唑基、吡啶基、吡喃基、哒嗪基、嘧啶基、吡嗪基、苯并咪唑基、三唑基等。The term "heteroaryl" refers to a heteroaromatic system containing 1 to 4 heteroatoms, including nitrogen, oxygen and S(O)r (wherein r is an integer of 0, 1, or 2). For example, a 4-8 membered heteroaryl refers to a heteroaromatic system containing 4 to 8 ring atoms, and a 4-10 membered heteroaryl refers to a heteroaromatic system containing 4 to 10 ring atoms, including but not limited to pyrrolyl, furanyl, thienyl, pyrazolyl, thiazolyl, imidazolyl, oxazolyl, isoxazolyl, pyridinyl, pyranyl, pyridazinyl, pyrimidinyl, pyrazinyl, benzimidazolyl, triazolyl, and the like.
本发明所述的基团除非特别说明是“取代或未取代的”,否则本发明的基团均可被选自下组的取代基所取代:卤素、酰氧基、氰基、氨基、硝基、羧基、酰胺基、羧基甲基、C1-C6烷基、C1-C6烷氧基、C1-C6卤代烷基、C2-C6烯基、C2-C6卤代烯基、C2-C6炔基、C2-C6卤代炔基、羟基、C3-C6环烷基、C3-C6卤代环烷基、羟基C1-C4烷基、C5-C7环烯基、苯基、萘基等。Unless otherwise specified as being "substituted or unsubstituted", the groups described in the present invention may be substituted by substituents selected from the following groups: halogen, acyloxy, cyano, amino, nitro, carboxyl, amide, carboxymethyl, C1-C6 alkyl, C1-C6 alkoxy, C1-C6 haloalkyl, C2-C6 alkenyl, C2-C6 haloalkenyl, C2-C6 alkynyl, C2-C6 haloalkynyl, hydroxyl, C3-C6 cycloalkyl, C3-C6 halocycloalkyl, hydroxyl C1-C4 alkyl, C5-C7 cycloalkenyl, phenyl, naphthyl, etc.
表示基团的接合位置。 Indicates the bonding position of the group.
活性物质Active substances
如本文所用,术语“本发明化合物”和“本发明活性成分”可互换使用,指式I化合物。As used herein, the terms "compound of the present invention" and "active ingredient of the present invention" are used interchangeably to refer to a compound of Formula I.
在本发明中,还包括式I化合物的药学上可接受的盐。术语“药学上可接受的盐”指本发明化合物与酸或碱所形成的适合用作药物的盐。药学上可接受的盐包括无机盐和有机盐。一类优选的盐是本发明聚合物与酸形成的盐。适合形成盐的酸包括但并不限于:盐酸、氢溴酸、氢氟酸、硫酸、硝酸、磷酸等无机酸,甲酸、乙酸、丙酸、草酸、丙二酸、琥珀酸、富马酸、马来酸、乳酸、苹果酸、酒石酸、柠檬酸、苦味酸、甲磺酸、苯甲磺酸,苯磺酸等有机酸;以及天冬氨酸、谷氨酸等酸性氨基酸。In the present invention, pharmaceutically acceptable salts of the compounds of formula I are also included. The term "pharmaceutically acceptable salt" refers to a salt suitable for use as a drug formed by the compounds of the present invention and an acid or a base. Pharmaceutically acceptable salts include inorganic salts and organic salts. A preferred class of salts is a salt formed by the polymer of the present invention and an acid. Acids suitable for forming salts include but are not limited to: inorganic acids such as hydrochloric acid, hydrobromic acid, hydrofluoric acid, sulfuric acid, nitric acid, phosphoric acid, formic acid, acetic acid, propionic acid, oxalic acid, malonic acid, succinic acid, fumaric acid, maleic acid, lactic acid, malic acid, tartaric acid, citric acid, picric acid, methanesulfonic acid, benzenesulfonic acid, benzenesulfonic acid, and acidic amino acids such as aspartic acid and glutamic acid.
本发明的式I化合物可采用现有技术中本领域技术人员熟知的方法进行制备,对各个步骤的反应参数没有特别限制。此外,本发明的典型化合物也可通过市售方式获得。The compounds of formula I of the present invention can be prepared by methods well known to those skilled in the art, and there is no particular restriction on the reaction parameters of each step. In addition, the typical compounds of the present invention can also be obtained by commercial means.
如本文所用,在式I化合物中,如果存在手性碳原子,则手性碳原子可以为R构型,也可以为S构型,或二者的混合物。As used herein, in the compounds of Formula I, if a chiral carbon atom exists, the chiral carbon atom may be in the R configuration, the S configuration, or a mixture of the two.
在本发明中,所述的活性成分式I化合物具有如下所示结构:In the present invention, the active ingredient compound of formula I has the following structure:
式中:Where:
R1选自下组:羟基、C1-C6烷氧基、卤素、取代或未取代的-O-C1-C6烷基、其中所述的取代为磺酸基或羟基取代; R1 is selected from the following group: hydroxyl, C1-C6 alkoxy, halogen, substituted or unsubstituted -O-C1-C6 alkyl, The substitution is sulfonic acid or hydroxyl substitution;
其中,in,
中的碳原子为手性碳原子,所述的手性碳原子选自下组: middle The carbon atom is a chiral carbon atom, and the chiral carbon atom is selected from the following group:
X为S或O;m为1-6的正整数;X is S or O; m is a positive integer from 1 to 6;
R5为H或 R5 is H or
R4为选自下组的金属离子:Na+、K+、Li+或Cs+;R 4 is a metal ion selected from the group consisting of Na + , K + , Li + or Cs + ;
R6为C1-C6烷基、C3-C8环烷基、C2-C6烯基、C2-C6炔基、芳基或杂芳基; R6 is C1-C6 alkyl, C3-C8 cycloalkyl, C2-C6 alkenyl, C2-C6 alkynyl, aryl or heteroaryl;
R2和R3各自独立地为H、C1-C6烷基、C2-C6烯基、C2-C6炔基、C3-C8环烷基、C2-C6羟烷基或-(C1-C3亚烷基)-COOH; R2 and R3 are each independently H, C1-C6 alkyl, C2-C6 alkenyl, C2-C6 alkynyl, C3-C8 cycloalkyl, C2-C6 hydroxyalkyl or -(C1-C3 alkylene)-COOH;
n为6-12的正整数。n is a positive integer from 6 to 12.
一个实施方式中,R1选自下组:羟基、甲氧基、 In one embodiment, R1 is selected from the group consisting of hydroxyl, methoxy,
另一个实施方式中,m=1。In another embodiment, m=1.
另一个实施方式中,R4为Na+。In another embodiment, R 4 is Na + .
另一个实施方式中,R6为CH3。In another embodiment, R 6 is CH 3 .
另一个实施方式中,n为6、7或8。In another embodiment, n is 6, 7 or 8.
另一个实施方式中,R1选自下组:羟基、甲氧基、 In another embodiment, R1 is selected from the group consisting of hydroxyl, methoxy,
另一个实施方式中,R2和R3各自独立地为H、甲基、羟丙基、羟乙基或羧甲基。In another embodiment, R2 and R3 are each independently H, methyl, hydroxypropyl, hydroxyethyl or carboxymethyl.
另一个实施方式中,所述的式I化合物选自下组:甲基环糊精、羧甲基环糊精、羟乙基-β-乙环糊精、羟丙基-β-环糊精或磺丁基醚-β-环糊精。In another embodiment, the compound of formula I is selected from the group consisting of methyl cyclodextrin, carboxymethyl cyclodextrin, hydroxyethyl-β-cyclodextrin, hydroxypropyl-β-cyclodextrin or sulfobutyl ether-β-cyclodextrin.
另一个实施方式中,所述的式I化合物母核为环糊精。In another embodiment, the core of the compound of formula I is cyclodextrin.
另一个实施方式中,所述的式I化合物为 In another embodiment, the compound of formula I is
另一个实施方式中,所述的式I化合物选自下组:In another embodiment, the compound of formula I is selected from the following group:
药物组合物和施用方法Pharmaceutical compositions and methods of administration
本发明还提供了一种药物组合物,包含药学上可接受的载体和一种或多种安全有效量的本发明所述的化合物。The present invention also provides a pharmaceutical composition comprising a pharmaceutically acceptable carrier and one or more safe and effective amounts of the compounds of the present invention.
由于本发明化合物具有优异的肠道保护活性,因此本发明化合物及其各种晶型,药学上可接受的无机或有机盐,水合物或溶剂合物,以及含有本发明化合物为主要活性成分的药物组合物可用于降低靶向抗肿瘤药物对肠道损伤,减少腹泻症状。Since the compounds of the present invention have excellent intestinal protective activity, the compounds of the present invention and their various crystal forms, pharmaceutically acceptable inorganic or organic salts, hydrates or solvates, and pharmaceutical compositions containing the compounds of the present invention as the main active ingredient can be used to reduce intestinal damage caused by targeted anti-tumor drugs and reduce diarrhea symptoms.
本发明的药物组合物包含安全有效量范围内的本发明化合物或其药理上可接受的盐及药理上可以接受的赋形剂或载体。其中“安全有效量”指的是:化合物的量足以明显改善病情,而不至于产生严重的副作用。通常,药物组合物含有1-2000mg本发明化合物/剂,更佳地,含有10-1000mg本发明化合物/剂。较佳地,所述的“一剂”为一个胶囊或药片。The pharmaceutical composition of the present invention comprises a safe and effective amount of the compound of the present invention or a pharmacologically acceptable salt thereof and a pharmacologically acceptable excipient or carrier. Wherein "safe and effective amount" means: the amount of the compound is sufficient to significantly improve the condition without causing serious side effects. Usually, the pharmaceutical composition contains 1-2000 mg of the compound of the present invention per dose, and more preferably, contains 10-1000 mg of the compound of the present invention per dose. Preferably, the "one dose" is a capsule or tablet.
“药学上可以接受的载体”指的是:一种或多种相容性固体或液体填料或凝胶物质,它们适合于人使用,而且必须有足够的纯度和足够低的毒性。“相容性”在此指的是组合物中各组份能和本发明的化合物以及它们之间相互掺和,而不明显降低化合物的药效。药学上可以接受的载体部分例子有纤维素及其衍生物(如羧甲基纤维素钠、乙基纤维素钠、纤维素乙酸酯等)、明胶、滑石、固体润滑剂(如硬脂酸、硬脂酸镁)、硫酸钙、植物油(如豆油、芝麻油、花生油、橄榄油等)、多元醇(如丙二醇、甘油、甘露醇、山梨醇等)、乳化剂(如)、润湿剂(如十二烷基硫酸钠)、着色剂、调味剂、稳定剂、抗氧化剂、防腐剂、无热原水等。"Pharmaceutically acceptable carrier" refers to: one or more compatible solid or liquid fillers or gel substances, which are suitable for human use and must have sufficient purity and sufficiently low toxicity. "Compatibility" here means that the components in the composition can be mixed with the compounds of the present invention and with each other without significantly reducing the efficacy of the compounds. Some examples of pharmaceutically acceptable carriers include cellulose and its derivatives (such as sodium carboxymethyl cellulose, sodium ethyl cellulose, cellulose acetate, etc.), gelatin, talc, solid lubricants (such as stearic acid, magnesium stearate), calcium sulfate, vegetable oils (such as soybean oil, sesame oil, peanut oil, olive oil, etc.), polyols (such as propylene glycol, glycerol, mannitol, sorbitol, etc.), emulsifiers (such as ), wetting agents (such as sodium lauryl sulfate), colorants, flavoring agents, stabilizers, antioxidants, preservatives, pyrogen-free water, etc.
所述的药物组合物为注射剂、囊剂、片剂、丸剂、散剂或颗粒剂。The pharmaceutical composition is in the form of injection, capsule, tablet, pill, powder or granule.
本发明化合物或药物组合物的施用方式没有特别限制,代表性的施用方式包括(但并不限于):口服、瘤内、直肠、肠胃外(静脉内、肌肉内或皮下)、和局部给药。There is no particular limitation on the administration of the compound or pharmaceutical composition of the present invention. Representative administrations include, but are not limited to, oral, intratumoral, rectal, parenteral (intravenous, intramuscular or subcutaneous), and topical administration.
用于口服给药的固体剂型包括胶囊剂、片剂、丸剂、散剂和颗粒剂。在这些固体剂型中,活性化合物与至少一种常规惰性赋形剂(或载体)混合,如柠檬酸钠或磷酸二钙,或与下述成分混合:(a)填料或增容剂,例如,淀粉、乳糖、蔗糖、葡萄糖、甘露醇和硅酸;(b)粘合剂,例如,羟甲基纤维素、藻酸盐、明胶、聚乙烯基吡咯烷酮、蔗糖和阿拉伯胶;(c)保湿剂,例如,甘油;(d)崩解剂,例如,琼脂、碳酸钙、马铃薯淀粉或木薯淀粉、藻酸、某些复合硅酸盐、和碳酸钠;(e)缓溶剂,例如石蜡;(f)吸收加速剂,例如,季胺化合物;(g)润湿剂,例如鲸蜡醇和单硬脂酸甘油酯;(h)吸附剂,例如,高岭土;和(i)润滑剂,例如,滑石、硬脂酸钙、硬脂酸镁、固体聚乙二醇、十二烷基硫酸钠,或其混合物。胶囊剂、片剂和丸剂中,剂型也可包含缓冲剂。Solid dosage forms for oral administration include capsules, tablets, pills, powders and granules. In these solid dosage forms, the active compound is mixed with at least one conventional inert excipient (or carrier), such as sodium citrate or dicalcium phosphate, or with the following ingredients: (a) fillers or extenders, for example, starches, lactose, sucrose, glucose, mannitol, and silicic acid; (b) binders, for example, hydroxymethylcellulose, alginates, gelatin, polyvinyl pyrrolidone, sucrose, and acacia; (c) humectants, for example, glycerol; (d) disintegrants, for example, agar, calcium carbonate, potato starch or tapioca starch, alginic acid, certain complex silicates, and sodium carbonate; (e) solubilizers, for example, paraffin; (f) absorption accelerators, for example, quaternary ammonium compounds; (g) wetting agents, for example, cetyl alcohol and glyceryl monostearate; (h) adsorbents, for example, kaolin; and (i) lubricants, for example, talc, calcium stearate, magnesium stearate, solid polyethylene glycols, sodium lauryl sulfate, or mixtures thereof. In the case of capsules, tablets and pills, the dosage forms may also comprise buffering agents.
固体剂型如片剂、糖丸、胶囊剂、丸剂和颗粒剂可采用包衣和壳材制备,如肠衣和其它本领域公知的材料。它们可包含不透明剂,并且,这种组合物中活性化合物或化合物的释放可以延迟的方式在消化道内的某一部分中释放。可采用的包埋组分的实例是聚合物质和蜡类物质。必要时,活性化合物也可与上述赋形剂中的一种或多种形成微胶囊形式。Solid dosage forms such as tablets, pills, capsules, pills and granules can be prepared using coatings and shell materials, such as enteric coatings and other materials known in the art. They may contain opacifiers, and the release of the active compound or compounds in such compositions can be delayed in a certain part of the digestive tract. Examples of embedding components that can be used are polymeric substances and waxes. If necessary, the active compound can also be formed into microencapsulated form with one or more of the above-mentioned excipients.
用于口服给药的液体剂型包括药学上可接受的乳液、溶液、悬浮液、糖浆或酊剂。除了活性化合物外,液体剂型可包含本领域中常规采用的惰性稀释剂,如水或其它溶剂,增溶剂和乳化剂,例知,乙醇、异丙醇、碳酸乙酯、乙酸乙酯、丙二醇、1,3-丁二醇、二甲基甲酰胺以及油,特别是棉籽油、花生油、玉米胚油、橄榄油、蓖麻油和芝麻油或这些物质的混合物等。Liquid dosage forms for oral administration include pharmaceutically acceptable emulsions, solutions, suspensions, syrups or tinctures. In addition to the active compound, the liquid dosage form may contain an inert diluent conventionally used in the art, such as water or other solvents, solubilizers and emulsifiers, for example, ethanol, isopropanol, ethyl carbonate, ethyl acetate, propylene glycol, 1,3-butylene glycol, dimethylformamide and oils, in particular cottonseed oil, peanut oil, corn germ oil, olive oil, castor oil and sesame oil or mixtures of these substances.
除了这些惰性稀释剂外,组合物也可包含助剂,如润湿剂、乳化剂和悬浮剂、甜味剂、矫味剂和香料。Besides such inert diluents, the composition may also include adjuvants such as wetting agents, emulsifying and suspending agents, sweetening, flavoring, and perfuming agents.
除了活性化合物外,悬浮液可包含悬浮剂,例如,乙氧基化异十八烷醇、聚氧乙烯山梨醇和脱水山梨醇酯、微晶纤维素、甲醇铝和琼脂或这些物质的混合物等。Suspensions, in addition to the active compounds, may contain suspending agents such as, for example, ethoxylated isostearyl alcohols, polyoxyethylene sorbitol and sorbitan esters, microcrystalline cellulose, aluminum methanol and agar, or mixtures of these substances, and the like.
用于肠胃外注射的组合物可包含生理上可接受的无菌含水或无水溶液、分散液、悬浮液或乳液,和用于重新溶解成无菌的可注射溶液或分散液的无菌粉末。适宜的含水和非水载体、稀释剂、溶剂或赋形剂包括水、乙醇、多元醇及其适宜的混合物。Compositions for parenteral injection may include physiologically acceptable sterile aqueous or anhydrous solutions, dispersions, suspensions or emulsions, and sterile powders for reconstitution into sterile injectable solutions or dispersions. Suitable aqueous and non-aqueous carriers, diluents, solvents or excipients include water, ethanol, polyols and suitable mixtures thereof.
用于局部给药的本发明化合物的剂型包括软膏剂、散剂、贴剂、喷射剂和吸入剂。活性成分在无菌条件下与生理上可接受的载体及任何防腐剂、缓冲剂,或必要时可能需要的推进剂一起混合。Dosage forms for topical administration of the compounds of the invention include ointments, powders, patches, sprays and inhalants. The active ingredient is mixed under sterile conditions with a physiologically acceptable carrier and any preservatives, buffers, or propellants that may be required.
本发明化合物可以单独给药,或者与其他药学上可接受的其他化合物(如靶向抗肿瘤药物)联合给药。The compound of the present invention can be administered alone or in combination with other pharmaceutically acceptable compounds (such as targeted anti-tumor drugs).
本发明治疗方法可以单独施用,或者与其它治疗手段或者治疗药物联用。The treatment method of the present invention can be used alone or in combination with other treatment methods or therapeutic drugs.
使用药物组合物时,是将安全有效量的本发明化合物适用于需要治疗的哺乳动物(如人),其中施用时剂量为药学上认为的有效给药剂量,对于60kg体重的人而言,日给药剂量通常为1~2000mg,优选50~1000mg。当然,具体剂量还应考虑给药途径、病人健康状况等因素,这些都是熟练医师技能范围之内的。When using the pharmaceutical composition, a safe and effective amount of the compound of the present invention is applied to a mammal (such as a human) in need of treatment, wherein the dosage during administration is a pharmaceutically effective dosage, and for a person weighing 60 kg, the daily dosage is usually 1 to 2000 mg, preferably 50 to 1000 mg. Of course, the specific dosage should also take into account factors such as the route of administration and the health status of the patient, which are all within the skill of a skilled physician.
本发明的主要优点包括:The main advantages of the present invention include:
(1)本发明的式I化合物可用于提高靶向抗肿瘤药物的治疗效果;(1) The compounds of formula I of the present invention can be used to improve the therapeutic effect of targeted anti-tumor drugs;
(2)本发明的式I化合物可用于降低服用靶向抗肿瘤药物导致的肠道损伤,减少腹泻;(2) The compounds of formula I of the present invention can be used to reduce intestinal damage caused by taking targeted anti-tumor drugs and reduce diarrhea;
(3)本发明的式I化合物可用于减少肠道损伤所导致的肠胀气;(3) The compounds of formula I of the present invention can be used to reduce intestinal flatulence caused by intestinal damage;
(4)本发明的式I化合物可用于保护肠壁绒毛的完整性以及减少肠道绒毛破坏;(4) The compounds of formula I of the present invention can be used to protect the integrity of the intestinal villi and reduce the destruction of intestinal villi;
(5)本发明的式I化合物生物安全性高,疗效好。(5) The compound of formula I of the present invention has high biological safety and good therapeutic effect.
下面结合具体实施例,进一步阐述本发明。应理解,这些实施例仅用于说明本发明而不用于限制本发明的范围。下列实施例中未注明具体条件的实验方法,通常按照常规条件,例如Sambrook等人,分子克隆:实验室手册(New York:Cold Spring HarborLaboratory Press,1989)中所述的条件,或按照制造厂商所建议的条件。除非另外说明,否则百分比和份数是重量百分比和重量份数。The present invention will be further described below in conjunction with specific examples. It should be understood that these examples are intended to illustrate the present invention only and are not intended to limit the scope of the present invention. The experimental methods for which specific conditions are not specified in the following examples are generally performed under conventional conditions, such as those described in Sambrook et al., Molecular Cloning: A Laboratory Manual (New York: Cold Spring Harbor Laboratory Press, 1989), or under conditions recommended by the manufacturer. Unless otherwise indicated, percentages and parts are weight percentages and weight parts.
实施例1样品对胃肠道保护功效评价Example 1 Evaluation of the protective efficacy of samples on the gastrointestinal tract
实验方法:Experimental methods:
随机选取390尾4dpf野生型AB品系斑马鱼于6孔板中,每孔(实验组)均处理30尾斑马鱼。除正常对照组外,其余各实验组均水溶给予吉非替尼建立斑马鱼胃肠道损伤模型。分别水溶给予“化合物(1)”、“化合物(2)”、“化合物(3)”、“化合物(4)”、“化合物(5)”、“化合物(6)”、“化合物(7)”、“化合物(8)”、“化合物(9)”、“化合物(10)”、“化合物(11)”和“化合物(12)”均83.3μg/mL浓度,同时设置正常对照组和模型对照组,每孔容量为3mL。28L处理2天后,每个实验组在显微镜下观察,观察指标包括:①肠腔大小、②肠腔变性,统计发生率并拍摄典型图片,以各指标的统计学分析结果分别评价本发明化合物的胃肠道保护功效。390 4dpf wild-type AB strain zebrafish were randomly selected and placed in a 6-well plate, with 30 zebrafish treated in each well (experimental group). Except for the normal control group, all other experimental groups were given gefitinib in water to establish a zebrafish gastrointestinal injury model. "Compound (1)", "Compound (2)", "Compound (3)", "Compound (4)", "Compound (5)", "Compound (6)", "Compound (7)", "Compound (8)", "Compound (9)", "Compound (10)", "Compound (11)" and "Compound (12)" were given in water at a concentration of 83.3 μg/mL, and a normal control group and a model control group were set up at the same time, with a capacity of 3 mL per well. After 2 days of treatment with 28L, each experimental group was observed under a microscope, and the observation indicators included: ① intestinal cavity size, ② intestinal cavity degeneration, the incidence was counted and typical pictures were taken, and the gastrointestinal protective efficacy of the compounds of the present invention was evaluated based on the statistical analysis results of each indicator.
表1.本发明化合物处理后斑马鱼胃肠道表型(肠腔变小)及发生率(n=30)Table 1. Gastrointestinal phenotype (smaller intestinal cavity) and incidence of zebrafish after treatment with the compounds of the present invention (n=30)
与模型对照组比较,*p<0.05,**p<0.01Compared with the model control group, *p<0.05, **p<0.01
表2.本发明化合物处理后斑马鱼胃肠道表型(肠腔变性)及发生率(n=30)Table 2. Gastrointestinal phenotype (intestinal degeneration) and incidence of zebrafish after treatment with the compounds of the present invention (n=30)
与模型对照组比较,*p<0.05。Compared with the model control group, *p<0.05.
实验结果:本实验浓度下,以上12个化合物引起的肠道变小发生率均低于模型对照组。“化合物4”和“化合物9”的肠腔变性发生率高于模型对照组,“化合物3”、“化合物4”、“化合物5”、“化合物6”、“化合物8”、“化合物9”和“化合物10”的死亡率均高于模型对照组。Experimental results: At this experimental concentration, the incidence of intestinal shrinkage caused by the above 12 compounds was lower than that of the model control group. The incidence of intestinal degeneration caused by "Compound 4" and "Compound 9" was higher than that of the model control group, and the mortality rates of "Compound 3", "Compound 4", "Compound 5", "Compound 6", "Compound 8", "Compound 9" and "Compound 10" were all higher than those of the model control group.
实验结论:在本实验条件下,“化合物1”、“化合物2”、“化合物3”、“化合物5”、“化合物6、“化合物7”、“化合物8”、“化合物10”和“化合物11”,显示在83.3μg/mL实验条件下浓度下对斑马鱼胃肠道保护功效有改善趋势,其中化合物11对斑马鱼有明显的胃肠道保护功效。Experimental conclusion: Under the experimental conditions, "Compound 1", "Compound 2", "Compound 3", "Compound 5", "Compound 6", "Compound 7", "Compound 8", "Compound 10" and "Compound 11" showed an improving trend in gastrointestinal protective efficacy on zebrafish at a concentration of 83.3 μg/mL under the experimental conditions, among which Compound 11 had obvious gastrointestinal protective efficacy on zebrafish.
实施例2样品诱发致敏风险评价Example 2 Sample-induced sensitization risk assessment
实验方法:Experimental methods:
选野生型AB品系,每个实验组为30尾斑马鱼,在28马培养箱中孵育。实验组别共3组。每个样品1个检测剂量(500ng/尾)、1个溶剂对照组和1个正常对照组。样品以静脉注射给药方式处理正常斑马鱼。Wild-type AB strain was selected, and each experimental group consisted of 30 zebrafish, which were incubated in a 28-hour incubator. There were 3 experimental groups in total. Each sample had 1 test dose (500 ng/tail), 1 solvent control group, and 1 normal control group. The samples were treated with normal zebrafish by intravenous administration.
样品处理结束后,各实验组斑马鱼用类致敏反应(肥大细胞脱颗粒)特异性检测试剂检测各实验组斑马鱼类胰蛋白酶(Tryptase)表达水平。统计学处理结果以mean。统计学表示;样品致敏作用计算公式如下:After the sample treatment, the expression level of tryptase in zebrafish of each experimental group was detected by using a specific detection reagent for anaphylactic reaction (mast cell degranulation). The statistical processing results were expressed as mean. The calculation formula of sample sensitization is as follows:
用SPSS软件进行统计学分析,p<0.05表明具有显著性差异;SPSS software was used for statistical analysis, and p < 0.05 indicated a significant difference;
表3各样品的致敏风险Table 3 Allergenic risk of each sample
实验结果:在500ng/尾的剂量条件下,测试的本发明化合物的致敏风险较低。代表性地,如表3所示,测试的本发明化合物(1)、化合物(2)、化合物(7)、化合物(11)致敏风险均显著低于阳性对照的致敏风险。Experimental results: Under the dosage condition of 500 ng/tail, the tested compounds of the present invention have a low risk of sensitization. Representatively, as shown in Table 3, the sensitization risks of the tested compounds of the present invention (1), (2), (7) and (11) are all significantly lower than the sensitization risk of the positive control.
其中,“化合物(2)”和“化合物(1)”没有或基本上没有致敏风险,“化合物(7)”和“化合物(11)”均具有低致敏风险。Among them, "Compound (2)" and "Compound (1)" have no or substantially no sensitization risk, and "Compound (7)" and "Compound (11)" both have low sensitization risk.
实验结论:结合实施例1和2的实验结果,即综合考虑安全性及有效性,本发明选择化合物(1)作为优选化合物进行后续实验。Experimental conclusion: Combining the experimental results of Examples 1 and 2, that is, taking safety and effectiveness into consideration, the present invention selects compound (1) as the preferred compound for subsequent experiments.
实施例3化合物1胃肠道毒性评价Example 3 Gastrointestinal toxicity evaluation of compound 1
实验方法:Experimental methods:
随机选取180尾4dpf野生型AB品系斑马鱼于6孔板中,每孔30尾。分别水溶给予样品(浓度见表4),同时设置溶剂对照组(1%DMSO)和正常对照组(标准稀释水处理斑马鱼),每孔容量为3mL。斑马鱼28马孵育2天后:(1)每个实验组在显微镜下观察,观察指标包括:①肠腔大小、②肠腔颜色、③肠腔有无胆汁、④肠道褶皱、⑤出血状况,统计毒性发生率并拍摄典型图片,评价样品的胃肠道毒性;(2)每个浓度组随机选取10尾斑马鱼在解剖显微镜下拍照并采集数据,分析统计斑马鱼肠腔面积,以统计学分析结果评价样品的胃肠道毒性。样品对肠腔面积的影响计算公式如下:180 4dpf wild-type AB strain zebrafish were randomly selected and placed in a 6-well plate, with 30 in each well. The samples were dissolved in water (concentrations are shown in Table 4), and a solvent control group (1% DMSO) and a normal control group (zebrafish treated with standard diluted water) were set up at the same time, with a capacity of 3mL per well. After 2 days of incubation of zebrafish for 28 days: (1) Each experimental group was observed under a microscope, and the observation indicators included: ① intestinal cavity size, ② intestinal cavity color, ③ presence of bile in the intestinal cavity, ④ intestinal folds, and ⑤ bleeding conditions. The toxicity incidence was statistically analyzed and typical pictures were taken to evaluate the gastrointestinal toxicity of the samples; (2) 10 zebrafish were randomly selected from each concentration group and photographed and data was collected under a dissecting microscope. The intestinal cavity area of zebrafish was analyzed and statistically analyzed, and the gastrointestinal toxicity of the samples was evaluated based on the statistical analysis results. The calculation formula for the effect of the sample on the intestinal cavity area is as follows:
用SPSS26.0软件进行统计学分析,p<0.05表明差异具有统计学意义。SPSS26.0 software was used for statistical analysis, and p<0.05 indicated that the difference was statistically significant.
表4化合物(1)对斑马鱼胃肠道毒性反应情况(n=30)Table 4 Gastrointestinal toxicity of compound (1) to zebrafish (n=30)
备注:“注:表示未见明显异常Note: "Note: indicates no obvious abnormality
表5化合物(1)对斑马鱼胃肠道的影响(n=10)Table 5 Effects of compound (1) on the gastrointestinal tract of zebrafish (n=10)
与溶剂对照组比较,*p<0.05,***p<0.001。Compared with the solvent control group, *p<0.05, ***p<0.001.
与吉非替尼12.5μg/mL浓度比较,#p<0.05,###p<0.001。Compared with gefitinib 12.5 μg/mL concentration, # p<0.05, ### p<0.001.
实验结果:Experimental results:
(1)与吉非替尼12.5μg/mL浓度比较,吉非替尼12.5μg/mL+“化合物(1)”0.139μg/mL浓度、吉非替尼12.5μg/mL+“化合物(1)”0.416μg/mL浓度和吉非替尼12.5μg/mL+“化合物(1)”1.25μg/mL浓度斑马鱼肠腔变小发生率降低,且斑马鱼肠腔变黑发生率降低。(1) Compared with gefitinib at a concentration of 12.5 μg/mL, the incidence of zebrafish intestinal cavity shrinkage was reduced in the concentrations of gefitinib 12.5 μg/mL + "Compound (1)" 0.139 μg/mL, gefitinib 12.5 μg/mL + "Compound (1)" 0.416 μg/mL, and gefitinib 12.5 μg/mL + "Compound (1)" 1.25 μg/mL, and the incidence of zebrafish intestinal cavity darkening was reduced.
(2)吉非替尼12.5μg/mL浓度、吉非替尼12.5μg/mL+“化合物(1)”0.416μg/mL浓度和吉非替尼12.5μg/mL+“化合物(1)”1.25μg/mL诱发斑马鱼肠腔变小,其中吉非替尼12.5μg/mL+“化合物(1)”0.139μg/mL浓度对斑马鱼肠腔面积无影响作用。(2) Gefitinib at a concentration of 12.5 μg/mL, gefitinib at a concentration of 12.5 μg/mL + "Compound (1)" at a concentration of 0.416 μg/mL, and gefitinib at a concentration of 12.5 μg/mL + "Compound (1)" at a concentration of 1.25 μg/mL induced the zebrafish intestinal cavity to shrink. Among them, gefitinib at a concentration of 12.5 μg/mL + "Compound (1)" at a concentration of 0.139 μg/mL had no effect on the zebrafish intestinal cavity area.
(3)吉非替尼12.5μg/mL+“化合物(1)”0.139μg/mL浓度、吉非替尼12.5μg/mL+“化合物(1)”0.416μg/mL浓度和吉非替尼12.5μg/mL+“化合物(1)”1.25μg/mL浓度肠腔面积,与吉非替尼12.5μg/mL浓度比较,各浓度组p<0.001、p<0.05、p<0.05,表明相较于吉非替尼单独给药,“化合物(1)”+吉非替尼,可改善斑马鱼肠腔变小。(3) Compared with gefitinib 12.5 μg/mL + "Compound (1)" 0.139 μg/mL, gefitinib 12.5 μg/mL + "Compound (1)" 0.416 μg/mL, and gefitinib 12.5 μg/mL + "Compound (1)" 1.25 μg/mL, the intestinal area of each concentration group was p<0.001, p<0.05, and p<0.05, indicating that compared with gefitinib alone, "Compound (1)" + gefitinib can improve the shrinkage of the zebrafish intestinal cavity.
实验结论:在本实验条件下,相较于吉非替尼单独给药,“化合物(1)”+吉非替尼联合给药,可显著改善斑马鱼肠腔变小,且肠腔变小和变黑发生率均降低。Experimental conclusion: Under the experimental conditions, compared with the administration of gefitinib alone, the combined administration of "Compound (1)" + gefitinib can significantly improve the shrinkage of the zebrafish intestinal cavity, and the incidence of shrinkage and darkening of the intestinal cavity is reduced.
实施例4化合物1对吉非替尼致大鼠肠道损伤的保护作用及药代动力学检测Example 4 Protective effect of compound 1 on gefitinib-induced intestinal damage in rats and pharmacokinetic detection
实验方法:SD大鼠30只,雌雄各半,体重:175.9~212.0g,按性别体重随机分为5组,分别为:正常对照组、模型对照组、化合物1低、中、高剂量组(40mg/kg、120mg/kg、180mg/kg),每组6只。除正常对照组外,其余动物灌胃给予吉非替尼(22.5mg/kg),造模后正常对照组、模型对照组大鼠灌胃给予蒸馏水,化合物1低、中、高剂量组(40mg/kg、120mg/kg、180mg/kg)大鼠在给予吉非替尼3小时后,灌胃给予相应药液,给药体积为10mL/kg,1次/日,连续给药28天。给药期间每天观察动物排便情况,每周动物称重,末次给药结束后采血进行血药浓度检测,解剖取回肠和结肠进行组织病理学检查。Experimental methods: 30 SD rats, half male and half female, weighing 175.9-212.0g, were randomly divided into 5 groups according to sex and weight, namely: normal control group, model control group, low, medium and high dose groups of compound 1 (40mg/kg, 120mg/kg, 180mg/kg), 6 rats in each group. Except for the normal control group, the other animals were given gefitinib (22.5mg/kg) by gavage. After modeling, the rats in the normal control group and model control group were given distilled water by gavage. The rats in the low, medium and high dose groups of compound 1 (40mg/kg, 120mg/kg, 180mg/kg) were given the corresponding drug solution by gavage 3 hours after gefitinib administration. The administration volume was 10mL/kg, once a day, and the administration lasted for 28 days. During the administration period, the defecation of the animals was observed every day, the animals were weighed every week, blood was collected for blood drug concentration detection after the last administration, and the ileum and colon were dissected for histopathological examination.
实验结果:Experimental results:
4.1一般观察4.1 General Observations
模型对照组大鼠于给药D11时出现明显腹泻,化合物1各给药组均未见明显腹泻症状。各组大鼠粪便隐血试纸检测结果均为阴性,粪便无隐血。The rats in the model control group had obvious diarrhea when they were given D11, but no obvious diarrhea was observed in the groups given compound 1. The fecal occult blood test results of the rats in each group were negative, and there was no occult blood in the feces.
4.2化合物1对吉非替尼致肠道损伤大鼠体重的影响4.2 Effect of compound 1 on body weight of rats with gefitinib-induced intestinal injury
如表6所示,与正常对照组比较,模型对照组大鼠体重无明显差异。与模型对照组比较,化合物1低、中、高剂量组大鼠体重无明显差异。As shown in Table 6, compared with the normal control group, the body weight of the rats in the model control group was not significantly different. Compared with the model control group, the body weight of the rats in the low, medium and high dose groups of compound 1 was not significantly different.
表6化合物1对吉非替尼致肠道损伤大鼠体重的影响(n=6)Table 6 Effect of compound 1 on body weight of rats with intestinal injury induced by gefitinib ( n=6)
4.3化合物1对吉非替尼致肠道损伤大鼠血药浓度的影响4.3 Effect of compound 1 on blood concentration of gefitinib-induced intestinal injury in rats
与模型对照组比较,化合物1低、中、高剂量组大鼠在给予吉非替尼4h内血药浓度无明显差异。化合物1对吉非替尼的吸收没有明显影响,不影响吉非替尼的抗肿瘤效果。Compared with the model control group, there was no significant difference in the blood concentration of gefitinib in the low, medium and high dose groups of compound 1 within 4 hours of administration. Compound 1 had no significant effect on the absorption of gefitinib and did not affect the anti-tumor effect of gefitinib.
4.4化合物1对吉非替尼致肠道损伤大鼠肠道组织学的影响4.4 Effect of compound 1 on intestinal histology in rats with gefitinib-induced intestinal injury
回肠组织病理学检查结果:正常对照组大鼠回肠黏膜结构清晰,未见明显异常;模型对照组大鼠回肠黏膜固有层血管扩张,管腔内有少量炎性细胞浸润,杯状细胞数量增多,黏蛋白分泌增加,肠道柱状上皮细胞未见明显损伤。与模型对照组比较,化合物1低、中、高剂量组大鼠回肠黏膜杯状细胞数量明显减少,黏蛋白分泌明显减少,对肠道柱状上皮细胞未见明显效果或未引起损伤。即模型对照组和化合物1低、中、高剂量组大鼠回肠肠道柱状上皮细胞均正常。Results of ileum histopathological examination: The ileum mucosa structure of rats in the normal control group was clear, with no obvious abnormalities; the vascular dilation of the lamina propria of the ileum mucosa of rats in the model control group, a small amount of inflammatory cell infiltration in the lumen, an increase in the number of goblet cells, an increase in mucin secretion, and no obvious damage to intestinal columnar epithelial cells. Compared with the model control group, the number of goblet cells in the ileum mucosa of rats in the low, medium, and high dose groups of compound 1 was significantly reduced, and mucin secretion was significantly reduced, and no obvious effect was observed on intestinal columnar epithelial cells or caused no damage. That is, the intestinal columnar epithelial cells of the ileum of rats in the model control group and the low, medium, and high dose groups of compound 1 were normal.
结肠组织病理学检查结果:正常对照组大鼠结肠黏膜结构清晰,未见明显异常;模型对照组大鼠结肠黏膜内杯状细胞数量增多,黏蛋白分泌增加,肠道柱状上皮细胞未见明显损伤。与模型对照组比较,化合物1中、高剂量组大鼠结肠黏膜内杯状细胞数量明显减少,黏蛋白分泌明显减少,对肠道柱状上皮细胞未见明显效果或未引起损伤。即模型对照组和化合物1低、中、高剂量组大鼠结肠肠道柱状上皮细胞均正常。Results of colon histopathological examination: The colon mucosal structure of rats in the normal control group was clear, with no obvious abnormalities; the number of goblet cells in the colon mucosa of rats in the model control group increased, mucin secretion increased, and no obvious damage was observed in the intestinal columnar epithelial cells. Compared with the model control group, the number of goblet cells in the colon mucosa of rats in the medium and high dose groups of compound 1 was significantly reduced, mucin secretion was significantly reduced, and no obvious effect or damage was observed on the intestinal columnar epithelial cells. That is, the intestinal columnar epithelial cells of the colon of rats in the model control group and the low, medium, and high dose groups of compound 1 were normal.
表7化合物1对大鼠肠道杯状细胞数量的影响(n=6)Table 7 Effect of compound 1 on the number of intestinal goblet cells in rats ( n=6)
注:与正常对照组比较,++P<0.01;与模型对照组比较,*P<0.05,**P<0.01。Note: Compared with the normal control group, ++ P<0.01; compared with the model control group, *P<0.05, **P<0.01.
实验结果:化合物1能明显减少吉非替尼致大鼠腹泻症状,减少大鼠肠道组织杯状细胞数量和黏蛋白分泌,对柱状上皮细胞无明显效果或损伤,同时不影响吉非替尼血药浓度。Experimental results: Compound 1 can significantly reduce the diarrhea symptoms induced by gefitinib in rats, reduce the number of goblet cells and mucin secretion in the intestinal tissue of rats, have no obvious effect or damage to columnar epithelial cells, and do not affect the blood concentration of gefitinib.
实验结论:化合物1对吉非替尼致大鼠肠道损伤有明显的保护作用,且对吉非替尼血药浓度无影响。Experimental conclusion: Compound 1 has a significant protective effect on gefitinib-induced intestinal damage in rats and has no effect on the blood concentration of gefitinib.
实施例5化合物(1)和化合物(5)对厄洛替尼致大鼠肠道损伤的保护作用Example 5 Protective effects of compound (1) and compound (5) on erlotinib-induced intestinal damage in rats
实验方法:1)雄性SD大鼠38只,体重:200-220g,随机分为5组,分别为:正常对照组(3只)、模型对照组(7只)、化合物(1)低、高剂量组(1mg/kg、3mg/kg),化合物(5)低、高剂量组(1mg/kg、3mg/kg)。除正常对照组外,其余动物空腹灌胃给予厄洛替尼(48mg/kg),造模后正常对照组、模型对照组大鼠灌胃给予蒸馏水,化合物(1)和化合物(5)各剂量组大鼠在给予厄洛替尼后,同时灌胃给予相应受试药液,给药体积为10mL/kg,1次/日,连续给药28天。给药期间每天观察动物排便情况,并计算粪便含水量。2)雄性SD大鼠9只,体重:200-220g,随机分为3组,分别为:厄洛替尼组(48mg/kg)、厄洛替尼+化合物(5)低剂量组(7.5mg/kg)、厄洛替尼+化合物(5)高剂量组(30mg/kg)。两组分别灌胃给予前0h、给药后2、4、6、7、8、9、10、12、24h分别采血进行血药浓度检测。Experimental methods: 1) 38 male SD rats, weighing 200-220g, were randomly divided into 5 groups, namely: normal control group (3 rats), model control group (7 rats), low-dose and high-dose groups of compound (1) (1 mg/kg, 3 mg/kg), and low-dose and high-dose groups of compound (5) (1 mg/kg, 3 mg/kg). Except for the normal control group, the other animals were given erlotinib (48 mg/kg) by gavage on an empty stomach. After modeling, the rats in the normal control group and model control group were given distilled water by gavage. After erlotinib was given, the rats in each dose group of compound (1) and compound (5) were given the corresponding test solution by gavage at the same time. The administration volume was 10 mL/kg, once a day, and the administration was continued for 28 days. During the administration period, the defecation of the animals was observed every day, and the water content of the feces was calculated. 2) Nine male SD rats, weighing 200-220 g, were randomly divided into three groups: erlotinib group (48 mg/kg), erlotinib + compound (5) low-dose group (7.5 mg/kg), and erlotinib + compound (5) high-dose group (30 mg/kg). Blood samples were collected from the two groups at 0 h before oral administration and 2, 4, 6, 7, 8, 9, 10, 12, and 24 h after administration for blood drug concentration detection.
表8化合物(1)和化合物(5)对厄洛替尼致肠道损伤粪便评分的影响Table 8 Effects of Compound (1) and Compound (5) on Fecal Scores of Erlotinib-induced Intestinal Damage
表8化合物(1)和化合物(5)对厄洛替尼致肠道损伤腹泻率的影响Table 8 Effects of Compound (1) and Compound (5) on the Rate of Erlotinib-induced Intestinal Injury Diarrhea
表9化合物(1)和化合物(5)对厄洛替尼致肠道损伤粪便含水量的影响Table 9 Effects of Compound (1) and Compound (5) on Fecal Water Content in Intestinal Damage Induced by Erlotinib
实验结论:与厄洛替尼组比较,化合物(1)和(5)的各剂量组能够降低大鼠粪便评分和大鼠粪便含水量方面,且随着给药时间的延长,化合物1和化合物5均能够降低大鼠腹泻率。低剂量的化合物(1)和化合物(5)在降低大鼠粪便评分、大鼠粪便含水量和大鼠腹泻率方面优于化合物(1)和化合物(5)的高剂量组。Experimental conclusion: Compared with the erlotinib group, each dose group of compound (1) and (5) can reduce the fecal score and fecal water content of rats, and with the extension of administration time, compound 1 and compound 5 can reduce the diarrhea rate of rats. Low doses of compound (1) and compound (5) are superior to high doses of compound (1) and compound (5) in reducing fecal score, fecal water content and diarrhea rate of rats.
5.4单次给药化合物(1)和化合物(5)对厄洛替尼致肠道损伤大鼠血药浓度的影响5.4 Effects of single administration of compound (1) and compound (5) on blood concentrations of erlotinib in rats with intestinal injury
与模型对照组比较,化合物(1)低、高剂量组和化合物(5)低、高剂量组大鼠在给予厄洛替尼24h内血药浓度无明显差异。化合物(1)和(5)对厄洛替尼的吸收没有明显影响,因此不影响厄洛替尼的抗肿瘤效果。Compared with the model control group, there was no significant difference in the blood concentration of erlotinib in the low-dose and high-dose groups of compound (1) and the low-dose and high-dose groups of compound (5) within 24 hours after administration of erlotinib. Compounds (1) and (5) had no significant effect on the absorption of erlotinib, and therefore did not affect the anti-tumor effect of erlotinib.
实施例6化合物1对奥希替尼致大鼠肠道损伤的保护作用Example 6 Protective effect of compound 1 on osimertinib-induced intestinal damage in rats
实验方法:Experimental methods:
将SD大鼠随机分为5组,每组6-8只;空白组、模型组(奥希替尼30mg/kg,15mg/kg)、模型+治疗组(化合物(1)240mg/kg+奥希替尼30mg/kg;化合物(1)60mg/kg+奥希替尼30mg/kg);空白组大鼠每天经口灌胃给予纯水,奥希替尼30mg/kg组经口灌胃给予3mg/ml奥希替尼,奥希替尼15mg/kg组经口灌胃给予1.5mg/ml奥希替尼,化合物(1)240mg/kg组经口灌胃给予3mg/ml奥希替尼,化合物(1)60mg/kg组经口灌胃给予3mg/ml奥希替尼;12小时后,空白组大鼠经口灌胃给予纯水,奥希替尼30mg/kg组经口灌胃给予纯水,奥希替尼15mg/kg组经口灌胃给予纯水,化合物(1)240mg/kg组经口灌胃给予24mg/ml化合物(1),化合物(1)60mg/kg组经口灌胃给予6mg/ml化合物(1),连续给药7周。SD rats were randomly divided into 5 groups, with 6-8 rats in each group; blank group, model group (osimertinib 30 mg/kg, 15 mg/kg), model + treatment group (compound (1) 240 mg/kg + osimertinib 30 mg/kg; compound (1) 60 mg/kg + osimertinib 30 mg/kg); the blank group rats were given pure water by oral gavage every day, the osimertinib 30 mg/kg group was given 3 mg/ml osimertinib by oral gavage, the osimertinib 15 mg/kg group was given 1.5 mg/ml osimertinib, and compound (1) 240 mg/kg + osimertinib 30 mg/kg) by oral gavage. The 40 mg/kg group was orally gavaged with 3 mg/ml osimertinib, and the compound (1) 60 mg/kg group was orally gavaged with 3 mg/ml osimertinib; 12 hours later, the blank group rats were orally gavaged with pure water, the osimertinib 30 mg/kg group was orally gavaged with pure water, the osimertinib 15 mg/kg group was orally gavaged with pure water, the compound (1) 240 mg/kg group was orally gavaged with 24 mg/ml compound (1), and the compound (1) 60 mg/kg group was orally gavaged with 6 mg/ml compound (1), and the administration was continued for 7 weeks.
实验结果:Experimental results:
(1)对大鼠体重的影响:给予奥希替尼一周后,30mg/kg奥希替尼组的大鼠体重增长缓慢,和其他组别有显著性差异;第2、4、5、6、7、周开始和空白体重有显著差异;第4、5、6、7周给予低剂量奥希替尼组的大鼠体重和高剂量相比有显著差异;给予化合物(1)治疗的大鼠体重也增长较缓慢,但其平均体重高于同等奥希替尼剂量下不治疗的大鼠体重,且均数表现为有量效关系。(1) Effect on rat body weight: After one week of administration of osimertinib, the body weight of rats in the 30 mg/kg osimertinib group increased slowly, which was significantly different from that of other groups; there was a significant difference in body weight between the 2nd, 4th, 5th, 6th and 7th weeks and the blank control group; the body weight of rats in the low-dose osimertinib group at the 4th, 5th, 6th and 7th weeks was significantly different from that of the high-dose group; the body weight of rats treated with compound (1) also increased slowly, but their average body weight was higher than that of untreated rats at the same osimertinib dose, and the mean showed a dose-response relationship.
(2)对大鼠粪便的影响:大鼠麻醉后处死,剖腹探查,发现模型组的大鼠肠道回肠段有胀气,肠壁扩张且盲肠和近盲肠的结肠段内容物和空白相比异常;化合物(1)治疗组肠胀气发生率和盲肠结肠稀便发生率显著降低。(2) Effect on rat feces: Rats were anesthetized and then killed. Laparotomy was performed. It was found that the ileum of the model group rats had flatulence, intestinal wall expansion, and abnormal contents of the cecum and colon segments near the cecum compared with the control group. The incidence of intestinal flatulence and loose stools in the cecum and colon in the group treated with compound (1) was significantly reduced.
(3)回肠病理结果:空白组黏膜完整,绒毛排列整齐,腺体结构清晰,隐窝结构清晰完整;模型组绒毛排列较杂乱,绒毛较短,隐窝结构破坏,绒毛有缺失;化合物(1)治疗组绒毛结构基本完整,绒毛排列较整齐。(3) Ileal pathological results: The blank group had intact mucosa, neatly arranged villi, clear glandular structure, and clear and complete crypt structure; the model group had chaotically arranged villi, short villi, destroyed crypt structure, and missing villi; the compound (1) treatment group had basically intact villi structure, and neatly arranged villi.
(4)结肠病理结果:空白组黏膜完整,绒毛排列整齐;模型组绒毛较短,隐窝结构破坏,肠壁较薄;给予化合物(1)治疗组绒毛结构基本完整,绒毛排列较整齐。(4) Colon pathology results: The blank group had intact mucosa and neatly arranged villi; the model group had shorter villi, damaged crypt structure, and thinner intestinal wall; the group treated with compound (1) had basically intact villi structure and neatly arranged villi.
实验结论:化合物(1)治疗的大鼠的肠道胀气的情况较模型组要有所改善。从病理结果分析,化合物(1)对奥希替尼导致的消化道损伤有一定的保护作用,可以保护肠壁绒毛的完整性以及降低肠道绒毛破坏,对奥希替尼导致的腹泻的产生有一定的治疗作用。Experimental conclusion: The intestinal bloating of rats treated with compound (1) was improved compared with that of the model group. From the analysis of pathological results, compound (1) has a certain protective effect on the digestive tract damage caused by osimertinib, can protect the integrity of the intestinal wall villi and reduce the damage to the intestinal villi, and has a certain therapeutic effect on the diarrhea caused by osimertinib.
实施例7颗粒剂的制备Example 7 Preparation of granules
表10化合物(1)制备颗粒剂的配方表Table 10 Formula of compound (1) for preparing granules
按处方量称取化合物(1)、D-甘露糖醇、一水乳糖;将化合物(1)、D-甘露糖醇、一水乳糖加入湿法混合混合制粒机的锅体内,开启搅拌,混合5min;a.制粒:开启搅拌转速、切割转速,用蠕动泵加雾化形式入粘合剂溶液,制粒时间1min;b.整粒、干燥:制得的湿颗粒经摇摆式颗粒机过筛,置电热恒温干燥箱中70℃干燥(每5-10min翻动一次),水分≤0.5%);烘干的物料经摇摆式颗粒机过筛,采用铝箔袋包装。Weigh compound (1), D-mannitol and lactose monohydrate according to the prescription amount; add compound (1), D-mannitol and lactose monohydrate into the pot of a wet mixing granulator, start stirring and mix for 5 minutes; a. Granulation: start the stirring speed and cutting speed, use a peristaltic pump to add the binder solution in atomized form, and the granulation time is 1 minute; b. Granulation and drying: the obtained wet granules are sieved by a swing granulator, placed in an electric constant temperature drying oven at 70°C for drying (turned once every 5-10 minutes, moisture ≤0.5%); the dried material is sieved by a swing granulator and packaged in an aluminum foil bag.
在本发明提及的所有文献都在本申请中引用作为参考,就如同每一篇文献被单独引用作为参考那样。此外应理解,在阅读了本发明的上述讲授内容之后,本领域技术人员可以对本发明作各种改动或修改,这些等价形式同样落于本申请所附权利要求书所限定的范围。All documents mentioned in the present invention are cited as references in this application, just as each document is cited as reference individually. In addition, it should be understood that after reading the above teachings of the present invention, those skilled in the art can make various changes or modifications to the present invention, and these equivalent forms also fall within the scope defined by the claims attached to this application.
Claims (10)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202111671059.XA CN116407554A (en) | 2021-12-31 | 2021-12-31 | Drugs for reducing intestinal damage caused by targeted antineoplastic drugs and their application |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202111671059.XA CN116407554A (en) | 2021-12-31 | 2021-12-31 | Drugs for reducing intestinal damage caused by targeted antineoplastic drugs and their application |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN116407554A true CN116407554A (en) | 2023-07-11 |
Family
ID=87058433
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202111671059.XA Pending CN116407554A (en) | 2021-12-31 | 2021-12-31 | Drugs for reducing intestinal damage caused by targeted antineoplastic drugs and their application |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN116407554A (en) |
-
2021
- 2021-12-31 CN CN202111671059.XA patent/CN116407554A/en active Pending
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6612200B2 (en) | Choline salts of anti-inflammatory substituted cyclobutenedione compounds | |
| WO2009015560A1 (en) | Use of allylcysteine or its analogs and pharmaceutical composition threrof | |
| KR102790143B1 (en) | Antivirus or anticancer composition comprising niclosamide and preparing method for thereof | |
| CN119233966A (en) | Salt forms of dencichine | |
| JP2019503353A (en) | Amorphous onapristone composition and method of making the same | |
| TW202341973A (en) | Therapeutic compounds, formulations, and use thereof | |
| WO2023123468A1 (en) | Cardiovascular and cerebrovascular drug and use thereof | |
| CN116407554A (en) | Drugs for reducing intestinal damage caused by targeted antineoplastic drugs and their application | |
| TWI870751B (en) | Cardiovascular and cerebrovascular drugs and their applications | |
| CN112204025B (en) | Compounds for the treatment of pain, compositions comprising the same and methods of using the same | |
| CN116925015B (en) | Active substance for inhibiting premature ovarian failure and preparation method thereof | |
| CN116850289B (en) | Application of PLK4-targeted drugs in the treatment of platinum-resistant tumors | |
| TWI855481B (en) | Pharmaceutical composition comprising docetaxel or pharmaceutically acceptable salt thereof and preparing method for thereof | |
| CN114989138B (en) | Vonoprazan salt and crystal form, preparation method and application thereof | |
| WO2011095095A1 (en) | Medicinal composition comprising alcohol-soluble and water-insoluble licorice extract, pharmaceutical preparation, pharmaceutical application, therapeutic method, and preparative method thereof | |
| WO2006033505A1 (en) | Paroxetine cholate or cholic acid derivative salts, and composition comprising paroxetine and cholic acid or cholic acid derivatives | |
| US20250170065A1 (en) | Therapeutic compounds, formulations, and use thereof | |
| WO2013135187A1 (en) | Solid dispersion of improved adsorption performance and preparation thereof | |
| US11304930B2 (en) | Ammonium salts of 3-(3,5-dibromo-4-hydroxybenzyliden)-5-indo-1,3-dihydroindol-2-one and uses thereof | |
| CN113018268B (en) | Delaxacin meglumine freeze-dried preparation for injection and preparation method thereof | |
| CN101497605B (en) | Benzimidazole derivative containing alkoxy substituted pyridine | |
| CN109988216B (en) | Betulin crystal D-type substance, preparation method, composition and application thereof | |
| CN101039681B (en) | Intestinal Polyp Inhibitors | |
| WO2021083347A1 (en) | Use of quinazoline derivative or salt thereof or pharmaceutical composition thereof | |
| WO2015021922A1 (en) | Ginsenoside rd or enteric formulation thereof and use |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination |