CN116270982A - Use of recombinant type XVII humanized collagen in the treatment of breast cancer - Google Patents
Use of recombinant type XVII humanized collagen in the treatment of breast cancer Download PDFInfo
- Publication number
- CN116270982A CN116270982A CN202310107604.5A CN202310107604A CN116270982A CN 116270982 A CN116270982 A CN 116270982A CN 202310107604 A CN202310107604 A CN 202310107604A CN 116270982 A CN116270982 A CN 116270982A
- Authority
- CN
- China
- Prior art keywords
- breast cancer
- seq
- collagen
- amino acid
- recombinant type
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/39—Connective tissue peptides, e.g. collagen, elastin, laminin, fibronectin, vitronectin, cold insoluble globulin [CIG]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P15/00—Drugs for genital or sexual disorders; Contraceptives
- A61P15/14—Drugs for genital or sexual disorders; Contraceptives for lactation disorders, e.g. galactorrhoea
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/04—Antineoplastic agents specific for metastasis
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/78—Connective tissue peptides, e.g. collagen, elastin, laminin, fibronectin, vitronectin or cold insoluble globulin [CIG]
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02A—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE
- Y02A50/00—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE in human health protection, e.g. against extreme weather
- Y02A50/30—Against vector-borne diseases, e.g. mosquito-borne, fly-borne, tick-borne or waterborne diseases whose impact is exacerbated by climate change
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Gastroenterology & Hepatology (AREA)
- Zoology (AREA)
- Immunology (AREA)
- Biophysics (AREA)
- Genetics & Genomics (AREA)
- Molecular Biology (AREA)
- Biochemistry (AREA)
- Gynecology & Obstetrics (AREA)
- Pregnancy & Childbirth (AREA)
- Endocrinology (AREA)
- Reproductive Health (AREA)
- Toxicology (AREA)
- Epidemiology (AREA)
- Biomedical Technology (AREA)
- Oncology (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
Abstract
本发明属于生物医药领域,具体涉及重组XVII型人源化胶原蛋白在乳腺癌治疗中的用途。本发明提供了重组XVII型人源化胶原蛋白在制备用于预防和/或治疗乳腺疾病的药物中的用途;所述重组XVII型人源化胶原蛋白包含(A)m所示的序列或者由(A)m所示的序列组成,其中每个A为选自SEQ IDNo.1、SEQ ID No.2和SEQ ID No.3任一个所示的氨基酸序列;m为1‑10之间的整数。研究结果表明,重组XVII型人源化胶原蛋白可以明显抑制乳腺癌细胞的生长增殖能力及迁移特性,同时还可以促进乳腺癌细胞休眠,从而为治疗乳腺癌的药物制备奠定了基础。
The invention belongs to the field of biomedicine, and in particular relates to the application of recombinant type XVII humanized collagen in the treatment of breast cancer. The present invention provides the use of recombinant type XVII humanized collagen in the preparation of medicaments for preventing and/or treating breast diseases; the recombinant type XVII humanized collagen comprises the sequence shown in (A)m or consists of (A) The sequence composition shown in m, wherein each A is selected from the amino acid sequence shown in any one of SEQ ID No.1, SEQ ID No.2 and SEQ ID No.3; m is an integer between 1-10 . The research results show that the recombinant type XVII humanized collagen can significantly inhibit the growth, proliferation and migration characteristics of breast cancer cells, and can also promote the dormancy of breast cancer cells, thus laying the foundation for the preparation of drugs for the treatment of breast cancer.
Description
技术领域technical field
本发明属于生物医药领域,具体涉及重组XVII型人源化胶原蛋白在乳腺癌治疗中的用途。The invention belongs to the field of biomedicine, and in particular relates to the application of recombinant type XVII humanized collagen in the treatment of breast cancer.
背景技术Background technique
目前,乳腺癌(breast carcinoma,BC)在全世界癌症发病率中位居第一。乳腺癌的治疗方式有限,主要包括外科手术切除、放射治疗、化学治疗和激素治疗等。Currently, breast cancer (BC) ranks first in the incidence of cancer in the world. There are limited treatment options for breast cancer, mainly including surgical resection, radiation therapy, chemotherapy and hormone therapy.
三阴性乳腺癌(triple negative breast cancer,TNBC)是乳腺癌中的一种亚型,是指癌组织免疫组织化学检查结果为雌激素受体(estrogen receptor,ER)、孕激素受体(progesterone receptor,PR)和人表皮生长因子受体(epidermal growth factorreceptor 2,HER2)均为阴性的乳腺癌。其具有易复发、转移率高的特点,并且由于缺少有效的治疗靶点,导致其治疗手段较为单一,目前主要为化学治疗,因此急需对其更有效和安全的治疗手段。Triple negative breast cancer (TNBC) is a subtype of breast cancer. , PR) and human epidermal growth factor receptor (epidermal growth factor receptor 2, HER2) negative breast cancer. It has the characteristics of easy recurrence and high metastasis rate, and due to the lack of effective therapeutic targets, its treatment method is relatively single. At present, chemotherapy is the main treatment method, so more effective and safe treatment methods are urgently needed.
胶原蛋白是动物体内含量最丰富的蛋白质,约占总蛋白质的30%,在细胞粘附等生理过程中发挥着重要作用。由于胶原蛋白具有良好的生物相容性和生物降解安全性,其已被广泛的应用于医药领域。直至目前,各种研究使用的胶原蛋白绝大多数来自于动物组织、皮肤提取物。从动物体提取胶原蛋白水溶性差,可加工性很弱,直接限制了许多潜在用途的开发。而利用基因工程技术生产的胶原蛋白,可以有效克服上述缺点。Collagen is the most abundant protein in animals, accounting for about 30% of the total protein, and plays an important role in physiological processes such as cell adhesion. Because collagen has good biocompatibility and biodegradation safety, it has been widely used in the field of medicine. Until now, most of the collagen used in various researches comes from animal tissue and skin extracts. Collagen extracted from animal bodies has poor water solubility and weak processability, which directly limits the development of many potential uses. The collagen produced by genetic engineering technology can effectively overcome the above shortcomings.
申请号为201911051106.3、发明名称为“人胶原蛋白17型多肽、其生产方法和用途”的中国专利申请记载了一种重组XVII型人源化胶原蛋白,其具有高水溶性,高生物活性的特点。目前尚未有报道提出重组XVII型人源化胶原蛋白与乳腺癌治疗之间的联系。The Chinese patent application with the application number 201911051106.3 and the invention title "Human Collagen Type 17 Polypeptide, Its Production Method and Use" records a recombinant type XVII humanized collagen, which has the characteristics of high water solubility and high biological activity . No reports have suggested a link between recombinant type XVII humanized collagen and breast cancer treatment.
发明内容Contents of the invention
发明要解决的问题The problem to be solved by the invention
本发明拟应用申请号为201911051106.3的中国专利申请中的重组XVII型人源化胶原蛋白作为原材料,评价其在乳腺癌等乳腺疾病治疗中的有效性。具体而言,本发明拟利用重组XVII型人源化胶原蛋白抑制乳腺癌细胞的增殖活性和/或迁移能力,并促进乳腺癌细胞休眠从而达到预防和/或治疗乳腺癌的效果。This invention intends to use the recombinant humanized collagen type XVII in the Chinese patent application with application number 201911051106.3 as a raw material to evaluate its effectiveness in the treatment of breast cancer and other breast diseases. Specifically, the present invention intends to use recombinant type XVII humanized collagen to inhibit the proliferation activity and/or migration ability of breast cancer cells, and promote the dormancy of breast cancer cells so as to achieve the effect of preventing and/or treating breast cancer.
用于解决问题的方案solutions to problems
本发明提供了重组XVII型人源化胶原蛋白在制备用于预防和/或治疗乳腺疾病的药物中的用途;The present invention provides the use of recombinant type XVII humanized collagen in the preparation of medicines for preventing and/or treating breast diseases;
其中,所述重组XVII型人源化胶原蛋白包含(A)m所示的序列或者由(A)m所示的序列组成,其中每个A为选自SEQ ID No.1、SEQ ID No.2和SEQ ID No.3任一个所示的氨基酸序列或SEQ ID No.1、SEQ ID No.2和SEQ IDNo.3任一个取代、添加、插入或缺失了1或多个氨基酸残基的氨基酸序列或者与SEQ ID No.1、SEQ ID No.2和SEQ ID No.3任一个所示的氨基酸序列具有至少83%序列同一性的序列;m为1-10之间的整数;其中每个A相同或不同并且相邻两个A之间直接通过肽键连接或者通过1个以上的氨基酸残基连接。Wherein, the recombinant type XVII humanized collagen comprises the sequence shown in (A)m or consists of the sequence shown in (A)m, wherein each A is selected from SEQ ID No.1, SEQ ID No. 2 and the amino acid sequence shown in any one of SEQ ID No.3 or any one of SEQ ID No.1, SEQ ID No.2 and SEQ ID No.3 is substituted, added, inserted or deleted with 1 or more amino acid residues sequence or a sequence having at least 83% sequence identity with the amino acid sequence shown in any one of SEQ ID No.1, SEQ ID No.2 and SEQ ID No.3; m is an integer between 1-10; wherein each The A's are the same or different, and two adjacent A's are directly linked by a peptide bond or linked by more than one amino acid residue.
进一步地,所述重组XVII型人源化胶原蛋白包含SEQ ID No.1、SEQ IDNo.2、SEQID No.3、SEQ ID No.4或SEQ ID No.5所示的氨基酸序列或由其组成。Further, the recombinant type XVII humanized collagen comprises or consists of the amino acid sequence shown in SEQ ID No.1, SEQ ID No.2, SEQ ID No.3, SEQ ID No.4 or SEQ ID No.5 .
进一步地,所述乳腺疾病为乳腺癌。Further, the breast disease is breast cancer.
进一步地,所述乳腺癌包括管腔A型乳腺癌、管腔B型乳腺癌、HER-2过表达型乳腺癌、基底细胞样型乳腺癌、三阴性乳腺癌和正常乳腺样型乳腺癌。Further, the breast cancer includes luminal A breast cancer, luminal B breast cancer, HER-2 overexpression breast cancer, basal cell-like breast cancer, triple-negative breast cancer and normal breast-like breast cancer.
进一步地,所述乳腺癌为管腔A型乳腺癌、HER-2过表达型乳腺癌、基底细胞样型乳腺癌或三阴性乳腺癌。Further, the breast cancer is luminal A breast cancer, HER-2 overexpression breast cancer, basal cell-like breast cancer or triple negative breast cancer.
进一步地,所述重组XVII型人源化胶原蛋白通过抑制乳腺癌细胞的增殖能力、抑制乳腺癌细胞的迁移能力和/或促进乳腺癌细胞进入休眠来预防和/或治疗乳腺癌。Further, the recombinant type XVII humanized collagen prevents and/or treats breast cancer by inhibiting the proliferation ability of breast cancer cells, inhibiting the migration ability of breast cancer cells and/or promoting the breast cancer cells to enter dormancy.
进一步地,所述抑制乳腺癌细胞的增殖能力包括抑制乳腺癌细胞的DNA复制活性。Further, said inhibiting the proliferation ability of breast cancer cells includes inhibiting the DNA replication activity of breast cancer cells.
发明的效果The effect of the invention
本发明的研究结果表明,重组XVII型人源化胶原蛋白可以明显抑制乳腺癌细胞(既包括非三阴性乳腺癌细胞,还包括三阴性乳腺癌细胞)的生长增殖能力(包括DNA复制活性),并可以抑制肿瘤细胞(既包括非三阴性乳腺癌细胞,还包括三阴性乳腺癌细胞)的迁移特性,同时还可以促进乳腺癌细胞休眠,从而为治疗乳腺癌的药物制备奠定了基础,也为重组XVII型人源化胶原蛋白在临床上用于乳腺癌患者的安全性和有效性提供了一定的理论基础。The research results of the present invention show that the recombinant type XVII humanized collagen can significantly inhibit the growth and proliferation ability (including DNA replication activity) of breast cancer cells (including non-triple-negative breast cancer cells and triple-negative breast cancer cells), And it can inhibit the migration characteristics of tumor cells (including both non-triple-negative breast cancer cells and triple-negative breast cancer cells), and can also promote the dormancy of breast cancer cells, thereby laying the foundation for the preparation of drugs for the treatment of breast cancer, and also for the The safety and effectiveness of recombinant type XVII humanized collagen in breast cancer patients provide a certain theoretical basis.
附图说明Description of drawings
图1为不同浓度重组XVII型人源化胶原蛋白在不同时间对MCF-7细胞增殖活性影响统计结果。Figure 1 is the statistical result of the effect of different concentrations of recombinant humanized collagen type XVII on the proliferation activity of MCF-7 cells at different times.
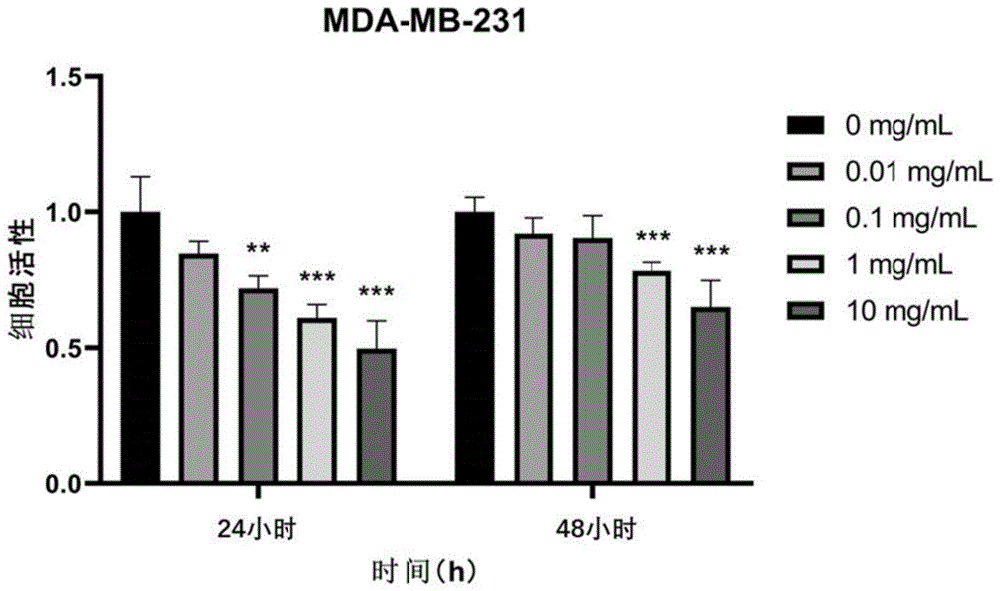
图2为不同浓度重组XVII型人源化胶原蛋白在不同时间对MDA-MB-231细胞增殖活性影响统计结果。Figure 2 is the statistical result of the effect of different concentrations of recombinant XVII humanized collagen on the proliferation activity of MDA-MB-231 cells at different times.
图3为不同浓度重组XVII型人源化胶原蛋白在不同时间对SK-BR-3细胞增殖活性影响统计结果。Figure 3 is the statistical results of the effects of different concentrations of recombinant XVII humanized collagen on the proliferation activity of SK-BR-3 cells at different times.
图4为本发明提供的重组XVII型人源化胶原蛋白在不同时间对MCF-7细胞迁移能力影响统计结果。Fig. 4 is the statistical result of the effect of recombinant type XVII humanized collagen provided by the present invention on the migration ability of MCF-7 cells at different times.
图5为本发明提供的重组XVII型人源化胶原蛋白在不同时间对MDA-MB-231细胞迁移能力影响统计结果。Fig. 5 is the statistical result of the effect of recombinant type XVII humanized collagen provided by the present invention on the migration ability of MDA-MB-231 cells at different times.
图6为本发明提供的重组XVII型人源化胶原蛋白在不同时间对SK-BR-3细胞迁移能力影响统计结果。Fig. 6 is the statistical result of the effect of recombinant type XVII humanized collagen provided by the present invention on the migration ability of SK-BR-3 cells at different times.
图7为EDU细胞增殖实验中本发明提供的重组XVII型人源化胶原蛋白对MCF-7细胞DNA复制活性的影响的检测结果。Fig. 7 is the detection result of the effect of the recombinant type XVII humanized collagen provided by the present invention on the DNA replication activity of MCF-7 cells in the EDU cell proliferation experiment.
图8为EDU细胞增殖实验中本发明提供的重组XVII型人源化胶原蛋白对MDA-MB-231细胞DNA复制活性的影响的检测结果。Fig. 8 is the detection result of the effect of the recombinant type XVII humanized collagen provided by the present invention on the DNA replication activity of MDA-MB-231 cells in the EDU cell proliferation experiment.
图9为EDU细胞增殖实验中本发明提供的重组XVII型人源化胶原蛋白对SK-BR-3细胞DNA复制活性的影响的检测结果。Fig. 9 is the detection result of the effect of the recombinant type XVII humanized collagen provided by the present invention on the DNA replication activity of SK-BR-3 cells in the EDU cell proliferation experiment.
图10为本发明提供的重组XVII型人源化胶原蛋白对MDA-MB-231细胞细胞周期的影响的检测结果。Fig. 10 is the detection result of the effect of recombinant type XVII humanized collagen provided by the present invention on the cell cycle of MDA-MB-231 cells.
具体实施方式Detailed ways
以下对本发明的实施方式进行说明,但本发明不限定于此。Embodiments of the present invention will be described below, but the present invention is not limited thereto.
在本发明中,使用“可以”表示的含义包括了进行某种处理以及不进行某种处理两方面的含义。In the present invention, the meaning expressed by "may" includes both meanings of performing certain processing and not performing certain processing.
在本发明中,“任选的”或“任选地”是指接下来描述的事件或情况可发生或可不发生,并且该描述包括该事件发生的情况和该事件不发生的情况。In the present invention, "optional" or "optionally" means that the next described event or circumstance may or may not occur, and that the description includes instances where the event occurs and instances where it does not.
在本发明中,术语“包含”、“具有”、“包括”或“含有”可以指包括在内的或开放式的,并不排除额外的、未引述的元件或方法步骤。与此同时,“包含”、“具有”、“包括”或“含有”也可以表示封闭式的,排除额外的、未引述的元件或方法步骤。In the present invention, the terms "comprising", "having", "comprising" or "containing" may mean inclusive or open-ended and do not exclude additional, unrecited elements or method steps. At the same time, "comprising", "having", "including" or "comprising" can also mean enclosing, excluding additional, unrecited elements or method steps.
在本发明中,所提及的“一些具体/优选的实施方案”、“另一些具体/优选的实施方案”、“实施方案”等是指所描述的与该实施方式有关的特定要素(例如,特征、结构、性质和/或特性)包括在此处所述的至少一种实施方案中,并且可存在于其它实施方案中或者可不存在于其它实施方案中。另外,应理解,所述要素可以任何合适的方式组合在各种实施方案中。In the present invention, references to "some specific/preferred embodiments", "other specific/preferred embodiments", "embodiments" and the like refer to specific elements described in relation to the embodiments (such as , feature, structure, property and/or characteristic) are included in at least one embodiment described herein and may or may not be present in other embodiments. In addition, it is to be understood that the described elements may be combined in any suitable manner in the various embodiments.
在本发明中,使用“数值A~数值B”或“数值A-数值B”表示的数值范围是指包含端点数值A、B的范围。In the present invention, the numerical range represented by "numerical value A - numerical value B" or "numerical value A-numerical value B" refers to a range including numerical values A and B at the endpoints.
在本发明中,术语“一(a)”或“一(an)”或“一(the)”可以指“一个”,也可以指“一个或多个”、“至少一个”以及“一个或多于一个”。In the present invention, the term "one (a)" or "one (an)" or "one (the)" may refer to "one", and may also refer to "one or more", "at least one" and "one or more than one".
在本发明中,术语“个体”、“患者”或“受试者”包括哺乳动物。哺乳动物包括但不限于,家养动物(例如,牛,羊,猫,狗和马),灵长类动物(例如,人和非人灵长类动物如猴),兔,以及啮齿类动物(例如,小鼠和大鼠)。In the present invention, the term "individual", "patient" or "subject" includes mammals. Mammals include, but are not limited to, domesticated animals (e.g., cattle, sheep, cats, dogs, and horses), primates (e.g., humans and non-human primates such as monkeys), rabbits, and rodents (e.g., , mice and rats).
在本发明中,“治疗”是指:在罹患疾病之后,使受试者接触(例如给药)本发明中所述的重组XVII型人源化胶原蛋白,从而与不接触时相比使该疾病的症状减轻,并不意味着必需完全抑制疾病的症状。罹患疾病是指:身体出现了疾病症状。In the present invention, "treatment" refers to: after suffering from a disease, subject is contacted (for example, administered) with the recombinant type XVII humanized collagen described in the present invention, thereby reducing the Alleviation of disease symptoms does not necessarily mean complete suppression of disease symptoms. Suffering from a disease means that the body has symptoms of a disease.
在本发明中,“预防”是指:在罹患疾病之前,通过使受试者接触本发明中所述的重组XVII型人源化胶原蛋白,从而与不接触时相比降低罹患疾病的概率和/或减轻罹患疾病后的症状,并不意味着必需完全抑制患病。In the present invention, "prevention" refers to: before suffering from a disease, by exposing the subject to the recombinant type XVII humanized collagen described in the present invention, thereby reducing the probability of suffering from the disease and the And/or alleviating the symptoms of a disease does not necessarily mean completely suppressing the disease.
在本发明中,所述重组XVII型人源化胶原蛋白为申请号为201911051106.3、发明名称为“人胶原蛋白17型多肽、其生产方法和用途”的中国专利申请记载。In the present invention, the recombinant type XVII humanized collagen is described in a Chinese patent application with the application number 201911051106.3 and the title of the invention "Human Collagen Type 17 Polypeptide, Its Production Method and Use".
在一些实施方案中,本发明所述的重组XVII型人源化胶原蛋白包含(A)m所示的序列或者由(A)m所示的序列组成,其中每个A为选自SEQ IDNo.1、SEQ ID No.2和SEQ ID No.3任一个所示的氨基酸序列或SEQ ID No.1、SEQ ID No.2和SEQ ID No.3任一个取代、添加、或缺失了1或多个氨基酸残基的氨基酸序列或者与SEQ ID No.1、SEQ ID No.2和SEQ IDNo.3任一个所示的氨基酸序列具有至少83%序列同一性的序列;m为1-10之间的整数;其中每个A相同或不同并且相邻两个A之间直接通过肽键连接或者通过1个以上的氨基酸残基连接。In some embodiments, the recombinant type XVII humanized collagen of the present invention comprises or consists of the sequence shown in (A)m, wherein each A is selected from SEQ ID No. 1. The amino acid sequence shown in any one of SEQ ID No.2 and SEQ ID No.3 or any one of SEQ ID No.1, SEQ ID No.2 and SEQ ID No.3 has 1 or more substitutions, additions, or deletions Amino acid sequence of amino acid residues or a sequence having at least 83% sequence identity with the amino acid sequence shown in any one of SEQ ID No.1, SEQ ID No.2 and SEQ ID No.3; m is between 1-10 Integer; where each A is the same or different, and two adjacent A are directly linked by a peptide bond or linked by more than one amino acid residue.
本发明所述的SEQ ID No.1、SEQ ID No.2或SEQ ID No.3来源于人17型胶原蛋白COL17A1序列,人17型胶原蛋白COL17A1序列参见https://www.ncbi.nlm.nih.gov/protein/Q9UMD9.3。The SEQ ID No.1, SEQ ID No.2 or SEQ ID No.3 of the present invention is derived from the human type 17 collagen COL17A1 sequence, and the human type 17 collagen COL17A1 sequence can be found at https://www.ncbi.nlm. nih.gov/protein/Q9UMD9.3.
SEQ ID No.1:GSPGPKGDMGSPGPKGDRGFPGTPGIPGPLGHPGPQG PKGQKGSVGDPGMEGPMGQRGREGPMGPRGEA。SEQ ID No. 1: GSPGPKGDMGSPGPKGDRGFPGTPPGIPGPLGHPGPQGPKGQKGSVGDPPGMEGPMGQRGREGPMGPRGEA.
SEQ ID No.2:GLQGLRGEVGLPGVKGDKGPMGPPGPKGDQGEKGPRGLTGEPGMRGLPGAVGEPGAKGAMGPA。SEQ ID No. 2: GLQGLRGEVGLPGVKGDKGPMGPPGPKGDQGEKGPRGLTGEPGMRGLPGAVGEPGAKGAMGPA.
SEQ ID No.3:GADFAGDLDYNELAVRVSESMQRQGLLQGMAYTVQ GPPGQPGPQGPPGISKVFSAYSNVTADLMDFFQTYGAIQGPPGQKGEMGT PGPKGDRGPAGPPGHPGPPGPRGHKGEKGDKGDQ。SEQ ID No. 3: GADFAGDLDYNELAVRVSESMQRQGLLQGMAYTVQGPPGQPGPQGPPGISKVFSAYSNVTADLMDFFQTYGAIQGPPGQKGEMGTPGPKGDRGPAGPGPGHPGPPGPRGHKGEKGDKGDQ.
在一些优选的实施方案中,本发明所述的重组XVII型人源化胶原蛋白包含如SEQID No.4所示的序列或由SEQ ID No.4所示的序列组成,其以SEQ ID No.1作为重复单元,重复了3次。In some preferred embodiments, the recombinant type XVII humanized collagen of the present invention comprises or consists of the sequence shown in SEQ ID No.4, which is represented by SEQ ID No. 1 was used as a repeating unit and was repeated 3 times.
SEQ ID No.4:GSPGPKGDMGSPGPKGDRGFPGTPGIPGPLGHPGPQG PKGQKGSVGDPGMEGPMGQRGREGPMGPRGEAGSPGPKGDMGSPGPKGDRGFPGTPGIPGPLGHPGPQGPKGQKGSVGDPGMEGPMGQRGREGPMGPRGEAGSPGPKGDMGSPGPKGDRGFPGTPGIPGPLGHPGPQGPKGQKGSVGDPGMEGPMGQRGREGPMGPRGEA。SEQ ID No. 4: GSPGPKGDMGSPGPKGDRGFPGTPPGIPGPLGHPGPQG PKGQKGSVGDPGMEGPMGQRGREGPMGPRGEAGSPGPKGDMGSPGPKGDRGFPGTPPGIPGPLGHPGPQGPKGQKGSVGDPGMEGPMGQRGREGPMGPRGEAGSPGPKGDMGSPGPKGDRGFPGTPPGIPGPLGHPGPQGPKG QKGSVGDPGMEGPMGQRGREGPMGPRGEA.
在另一些优选的实施方案中,本发明所述的重组XVII型人源化胶原蛋白包含如SEQ ID No.5所示的序列或由SEQ ID No.5所示的序列组成,其以SEQ ID No.2作为重复单元,重复了3次。In some other preferred embodiments, the recombinant type XVII humanized collagen of the present invention comprises or consists of the sequence shown in SEQ ID No.5, which is represented by SEQ ID No.2 was repeated 3 times as a repeating unit.
SEQ ID No.5:GLQGLRGEVGLPGVKGDKGPMGPPGPKGDQGEKGP RGLTGEPGMRGLPGAVGEPGAKGAMGPAGLQGLRGEVGLPGVKGDKGPMGPPGPKGDQGEKGPRGLTGEPGMRGLPGAVGEPGAKGAMGPAGLQGLRGEVGLPGVKGDKGPMGPPGPKGDQGEKGPRGLTGEPGMRGLPGAVGEPGAKGAMGPA。SEQ ID No. 5: GLQGLRGEVGLPGVKGDKGPMGPPGPKGDQGEKGPRGLTGEPGMRGLPGAVGEPGAKGAMGPAGLQGLRGEVGLPGVKGDKGPMGPPGPKGDQGEKGPRGLTGEPGMRGLPGAVGEPGAKGAMGPAGLQGLRGEVGLPGVKGDKGPMGPPGPKGDQGEKGPRGLTGEPGMRGL PGAVGEPGAKGAMGPA.
在本发明中,氨基酸添加指在氨基酸序列,例如SEQ ID No.4的C端或N端添加氨基酸,只要本发明的重组XVII型人源化胶原蛋白保留SEQ ID No.4的氨基酸序列的活性。In the present invention, amino acid addition refers to adding amino acids to the amino acid sequence, such as the C-terminal or N-terminal of SEQ ID No.4, as long as the recombinant type XVII humanized collagen of the present invention retains the activity of the amino acid sequence of SEQ ID No.4 .
在本发明中,氨基酸取代指在氨基酸序列,例如SEQ ID No.4的序列的某个位置的某个氨基酸残基被其他氨基酸残基替代,只要本发明的重组XVII型人源化胶原蛋白保留SEQ ID No.4的氨基酸序列的活性。In the present invention, amino acid substitution refers to the substitution of an amino acid residue at a certain position in an amino acid sequence, such as a sequence of SEQ ID No.4, by other amino acid residues, as long as the recombinant humanized collagen type XVII of the present invention remains Activity of the amino acid sequence of SEQ ID No.4.
在本发明中,氨基酸取代可以是保守氨基酸取代,指与SEQ ID No.4的氨基酸序列相比,有3个,更佳地2个氨基酸或1个氨基酸被性质相似或相近的氨基酸所替换而形成肽。这些保守性变异肽可以根据下列氨基酸替换而产生:Val、Leu或Ile对Ala的取代,Lys、Gln、Asn或His对Arg的取代,Gln、His、Lys或Arg对Asn的取代,Glu或Asn对Asp的取代,Ser或Ala对Cys的取代,Asn或Glu对Gln的取代,Asp或Gln对Glu的取代,Ala对Gly的取代,Asn、Lys、Gln或Arg对His的取代,Leu、Met、Ala、Val、Phe或正亮氨酸对Ile的取代,Ile、Met、Ala、Val、Phe或正亮氨酸对Leu的取代,Asn、Gln或Arg对Lys的取代,Ile、Leu或Phe对Met的取代,Leu、Val、Ile、Ala或Tyr对Phe的取代,Ala对Pro的取代,Thr对Ser的取代,Ser或Val对Thr的取代,Phe或Tyr对Trp的取代,Trp、Phe、Thr或Ser对Tyr的取代,及Phe、Ala、Met、Ile、Leu或正亮氨酸对Val的取代。氨基酸替换也可以是非保守氨基酸替换。In the present invention, the amino acid substitution can be a conservative amino acid substitution, which means that compared with the amino acid sequence of SEQ ID No.4, there are 3, more preferably 2 amino acids or 1 amino acid are replaced by amino acids with similar or similar properties. form peptides. These conservative variant peptides can be produced according to the following amino acid substitutions: substitution of Val, Leu or Ile for Ala, substitution of Lys, Gln, Asn or His for Arg, substitution of Gln, His, Lys or Arg for Asn, Glu or Asn Substitution of Asp, substitution of Cys by Ser or Ala, substitution of Gln by Asn or Glu, substitution of Glu by Asp or Gln, substitution of Gly by Ala, substitution of His by Asn, Lys, Gln or Arg, Leu, Met , Ala, Val, Phe or Norleucine for Ile, Ile, Met, Ala, Val, Phe or Norleucine for Leu, Asn, Gln or Arg for Lys, Ile, Leu or Phe Substitution of Met, substitution of Leu, Val, Ile, Ala or Tyr to Phe, substitution of Ala to Pro, substitution of Thr to Ser, substitution of Ser or Val to Thr, substitution of Phe or Tyr to Trp, Trp, Phe , Thr or Ser for Tyr substitution, and Phe, Ala, Met, Ile, Leu or norleucine for Val substitution. Amino acid substitutions can also be non-conservative amino acid substitutions.
在本发明中,氨基酸插入指在氨基酸序列,例如SEQ ID No.4的序列的适当位置插入氨基酸残基,插入的氨基酸残基也可以全部或部分彼此相邻,或插入的氨基酸之间都不彼此相邻,只要本发明的重组XVII型人源化胶原蛋白保留SEQ ID No.4的氨基酸序列的活性。在本文中氨基酸的插入位置不在各重复序列之间。In the present invention, amino acid insertion refers to the insertion of amino acid residues at an appropriate position in an amino acid sequence, such as the sequence of SEQ ID No.4, and the inserted amino acid residues can also be all or partly adjacent to each other, or there is no gap between the inserted amino acids. adjacent to each other as long as the recombinant type XVII humanized collagen of the present invention retains the activity of the amino acid sequence of SEQ ID No.4. The insertion position of the amino acid here is not between the individual repeat sequences.
在本发明中,氨基酸缺失指可以从氨基酸序列,例如SEQ ID No.4的序列中删除1、2或3个以上氨基酸,只要本发明的重组XVII型人源化胶原蛋白保留SEQ ID No.4的氨基酸序列的活性。In the present invention, amino acid deletion means that 1, 2 or more than 3 amino acids can be deleted from the amino acid sequence, such as the sequence of SEQ ID No.4, as long as the recombinant type XVII humanized collagen of the present invention retains SEQ ID No.4 activity of the amino acid sequence.
除非另有定义,本发明所用的其他技术和科学术语具有与本发明所属技术领域中的普通技术人员所通常理解的相同含义。Unless otherwise defined, other technical and scientific terms used in the present invention have the same meaning as commonly understood by one of ordinary skill in the art to which this invention belongs.
实施例Example
本发明通过下述实施例进一步阐明,但任何实施例或其组合不应当理解为对本发明的范围或实施方式的限制。本发明的范围由所附权利要求书限定,结合本说明书和本领域一般常识,本领域普通技术人员可以清楚地明白权利要求书所限定的范围。在不偏离本发明的精神和范围的前提下,本领域技术人员可以对本发明的技术方案进行任何修改或改变,这种修改和改变也包含在本发明的范围内。The present invention is further illustrated by the following examples, but any example or combination thereof should not be construed as limiting the scope or implementation of the present invention. The scope of the present invention is defined by the appended claims, and those skilled in the art can clearly understand the scope defined by the claims in combination with the description and common knowledge in the field. Without departing from the spirit and scope of the present invention, those skilled in the art may make any modifications or changes to the technical solutions of the present invention, and such modifications and changes are also included in the scope of the present invention.
实施例中未注明具体条件者,按照常规条件或制造商建议的条件进行。所有试剂或仪器未注明生产厂商者,均为可以通过市购的常规产品。为了更好地说明本发明,在下文的具体实施方式中给出了众多的具体细节。本领域技术人员应当理解,没有某些具体细节,本发明同样可以实施。在另外一些实施例中,对于本领域技术人员熟知的方法、手段、器材和步骤未作详细描述,以便凸显本发明的主旨。Those who do not indicate the specific conditions in the examples are carried out according to the conventional conditions or the conditions suggested by the manufacturer. All reagents or instruments that did not indicate the manufacturer are all conventional products that can be purchased from the market. In order to better illustrate the present invention, numerous specific details are given in the following specific embodiments. It will be understood by those skilled in the art that the present invention may be practiced without certain of the specific details. In some other embodiments, methods, means, devices and steps well known to those skilled in the art are not described in detail in order to highlight the gist of the present invention.
实验材料:Experimental Materials:
乳腺癌细胞:MCF-7细胞、MDA-MB-231细胞、SK-BR-3细胞来源于ATCC;Breast cancer cells: MCF-7 cells, MDA-MB-231 cells, SK-BR-3 cells are from ATCC;
FBS来源于:Sigma;FBS source: Sigma;
无血清培养基来源于:Sigma;Serum-free medium was sourced from: Sigma;
重组XVII型人源化胶原蛋白的氨基酸序列如SEQ ID No.4所示;The amino acid sequence of the recombinant type XVII humanized collagen is shown in SEQ ID No.4;
CCK-8试剂盒来源于:GLPbio;CCK-8 kit comes from: GLPbio;
EDU及细胞周期检测试剂盒来源于:碧云天。EDU and cell cycle detection kits are from: Biyuntian.
实施例1:重组XVII型人源化胶原蛋白对细胞增殖的影响Example 1: Effect of Recombinant Type XVII Humanized Collagen on Cell Proliferation
实验方法:experimental method:
实验组:乳腺癌细胞+10%FBS培养基+重组XVII型人源化胶原蛋白共培养。Experimental group: breast cancer cells + 10% FBS medium + recombinant type XVII humanized collagen co-cultured.
阴性对照组:乳腺癌细胞+10%FBS培养基,无胶原蛋白共培养。Negative control group: breast cancer cells + 10% FBS medium, co-cultured without collagen.
空白对照组:无细胞,10%FBS培养基,无胶原蛋白共培养。Blank control group: no cells, 10% FBS medium, co-culture without collagen.
设置实验分组后根据需要接种合适密度的三种乳腺癌细胞(MCF-7细胞、MDA-MB-231细胞和SK-BR-3细胞)于96孔板中,体积为100μL/孔,待细胞贴壁后加入重组XVII型人源化胶原蛋白,终浓度分别为0.01mg/mL、0.1mg/mL、1mg/mL、10mg/mL(图中标记为0.01mg/mL、0.1mg/mL、1mg/mL、10mg/mL),阴性对照组不加胶原蛋白(图中标记为0mg/mL);细胞与胶原蛋白共培养24h及48h后,将96孔板转移至细胞房超净台,弃去培养基,吸净残留液体,避光条件下每孔加入100μL无血清培养基及10μL CCK-8,并在96孔板外围加入PBS防止试剂挥发;将96孔板放于37℃孵箱中避光培养1小时,使用酶标仪测吸光度值OD450nm,重复测量3次取均值。依据CCK-8试剂盒说明书公式计算:细胞存活率=(实验组OD值-空白对照组OD值)/(阴性对照组OD值-空白对照组OD值)×100%。After setting up the experimental groups, inoculate three kinds of breast cancer cells (MCF-7 cells, MDA-MB-231 cells and SK-BR-3 cells) at the appropriate density in a 96-well plate according to the needs, with a volume of 100 μL/well. Recombinant type XVII humanized collagen was added behind the wall, and the final concentrations were 0.01mg/mL, 0.1mg/mL, 1mg/mL, and 10mg/mL (marked as 0.01mg/mL, 0.1mg/mL, and 1mg/mL in the figure). mL , 10mg/mL), the negative control group did not add collagen (marked as 0mg/mL in the figure); after 24h and 48h of co-culture of cells and collagen, the 96-well plate was transferred to the ultra-clean bench of the cell room, and the culture was discarded. Add 100 μL serum-free medium and 10 μL CCK-8 to each well in the dark, and add PBS to the periphery of the 96-well plate to prevent reagent volatilization; place the 96-well plate in a 37°C incubator to avoid light After incubation for 1 hour, the absorbance value OD 450nm was measured with a microplate reader, and the measurement was repeated three times to obtain the average value. Calculated according to the formula in the CCK-8 kit manual: cell survival rate = (OD value of the experimental group - OD value of the blank control group) / (OD value of the negative control group - OD value of the blank control group) × 100%.
实验结果:Experimental results:
结果如图1~3所示,重组XVII型人源化胶原蛋白可以抑制乳腺癌细胞的增殖活性。The results are shown in Figures 1-3, the recombinant type XVII humanized collagen can inhibit the proliferative activity of breast cancer cells.
分组培养后,和对照组相比,重组XVII型人源化胶原蛋白处理后的三种乳腺癌细胞的增殖活性均显著降低,说明重组XVII型人源化胶原蛋白对乳腺癌细胞的增殖能力有抑制作用。After group culture, compared with the control group, the proliferation activities of the three types of breast cancer cells treated with recombinant XVII humanized collagen were significantly reduced, indicating that recombinant XVII humanized collagen has an effect on the proliferation ability of breast cancer cells. inhibition.
实施例2:重组XVII型人源化胶原蛋白对细胞迁移的影响Example 2: Effect of Recombinant Type XVII Humanized Collagen on Cell Migration
实验方法:experimental method:
实验分组同实施例1。The experimental grouping is the same as in Example 1.
将乳腺癌细胞接种于六孔板中,体积为2mL/孔,待细胞贴壁后于实验组加入利用10%FBS培养基配制的重组XVII型人源化胶原蛋白进行共培养(图中标记为实验组),胶原蛋白终浓度为10mg/mL,对照组则加入10%FBS培养基(图中标记为对照组)。当每孔细胞铺满95%形成单层细胞时,用200μL移液枪枪头在单层细胞上呈“一”字划痕,用PBS清洗3次,继续于实验组加入利用无血清培养基配制的重组XVII型人源化胶原蛋白培养,胶原蛋白终浓度为10mg/mL,对照组则加入无血清培养基,培养箱孵育24h后吸去培养液,用PBS清洗3次后,在倒置荧光显微镜下观察并拍照,同时统计细胞相对迁移率。Breast cancer cells were inoculated in six-well plates with a volume of 2 mL/well. After the cells adhered to the wall, the recombinant XVII humanized collagen prepared with 10% FBS medium was added to the experimental group for co-culture (marked in the figure as experimental group), the final concentration of collagen was 10 mg/mL, and 10% FBS medium was added to the control group (marked as the control group in the figure). When the cells in each well are 95% full and form a single layer of cells, use a 200 μL pipette tip to make a "one" scratch on the monolayer of cells, wash with PBS for 3 times, and continue to add serum-free medium to the experimental group The prepared recombinant type XVII humanized collagen was cultured, the final concentration of collagen was 10 mg/mL, and the control group was added with serum-free medium. After incubation in the incubator for 24 hours, the culture medium was sucked off, washed with PBS for 3 times, and then placed in an inverted fluorescent tube. Observe and take pictures under a microscope, and count the relative migration rate of cells at the same time.
实验结果:Experimental results:
结果如图4~6所示,重组XVII型人源化胶原蛋白可以抑制乳腺癌细胞的迁移能力。The results are shown in Figures 4-6, the recombinant type XVII humanized collagen can inhibit the migration ability of breast cancer cells.
分组培养后,和对照组相比,重组XVII型人源化胶原蛋白处理后的三种乳腺癌细胞的迁移率均显著下降,说明重组XVII型人源化胶原蛋白对乳腺癌细胞的迁移能力有抑制作用。After group culture, compared with the control group, the migration rates of the three types of breast cancer cells treated with recombinant XVII humanized collagen decreased significantly, indicating that recombinant XVII humanized collagen has an effect on the migration ability of breast cancer cells. inhibition.
实施例3:重组XVII型人源化胶原蛋白对细胞DNA复制活性的影响实验方法:Example 3: Effect of recombinant type XVII humanized collagen on cellular DNA replication activity Experimental method:
实验分组同实施例1,胶原蛋白共培养终浓度为10mg/mL。The experimental grouping was the same as in Example 1, and the final concentration of collagen co-culture was 10 mg/mL.
取对数生长期的乳腺癌细胞,以每孔8×103个细胞接种于96孔板中。待第二天贴壁培养至正常生长阶段,使用EDU试剂盒进行染色。首先,将37℃预热的1×的EdU工作液(10μM),等体积加入6孔板中,置于37℃孵箱孵育2h;EdU标记细胞完成后,去除培养液,并加入0.1mL 4%(W/V)的多聚甲醛进行固定,常温固定15min后,去除固定液,每孔用0.1mL洗涤液洗涤细胞3次,每次3-5分钟。去除洗涤液后,每孔用0.1mL通透液(0.3%(V/V)Triton X-100的PBS),室温孵育10-15分钟。接着,去除通透液,每孔用0.1mL洗涤液洗涤细胞1-2次,每次3-5分钟,再每孔加入0.05mL Click反应液,室温避光孵育30分钟后吸除Click反应液,用洗涤液洗涤3次,每次3-5分钟。随后,用Hoechst 33342进行细胞核染色,室温避光孵育10分钟。随后即可进行荧光检测。Breast cancer cells in the logarithmic growth phase were seeded in 96-well plates at 8×10 3 cells per well. After adherent culture to the normal growth stage on the second day, the EDU kit was used for staining. First, add an equal volume of 1× EdU working solution (10 μM) preheated at 37°C to a 6-well plate, and incubate in a 37°C incubator for 2 hours; after EdU labeling of cells, remove the culture medium and add 0.1mL 4 % (W/V) paraformaldehyde for fixation. After fixing at room temperature for 15 minutes, remove the fixative, and wash the cells with 0.1 mL washing solution for each well 3 times, 3-5 minutes each time. After removing the washing solution, each well was incubated with 0.1 mL permeabilization solution (0.3% (V/V) Triton X-100 in PBS) at room temperature for 10-15 minutes. Next, remove the permeabilization solution, wash the cells 1-2 times with 0.1mL washing solution per well, 3-5 minutes each time, then add 0.05mL Click reaction solution to each well, incubate at room temperature in the dark for 30 minutes, then aspirate the Click reaction solution ,
实验结果:Experimental results:
结果如图7~9所示,重组XVII型人源化胶原蛋白可以抑制乳腺癌细胞的DNA复制活性等增殖能力。The results are shown in Figures 7-9, the recombinant type XVII humanized collagen can inhibit the proliferation ability of breast cancer cells such as DNA replication activity.
分组培养后,和对照组相比,重组XVII型人源化胶原蛋白处理后处于增殖活性的乳腺癌细胞(红色荧光)比例显著下降,说明重组XVII型人源化胶原蛋白对乳腺癌细胞的增殖有抑制作用。After group culture, compared with the control group, the proportion of breast cancer cells with proliferative activity (red fluorescence) decreased significantly after the treatment with recombinant XVII humanized collagen, indicating that the recombinant XVII humanized collagen has an effect on the proliferation of breast cancer cells. Has an inhibitory effect.
实施例4:重组XVII型人源化胶原蛋白对细胞周期的影响Example 4: Effect of Recombinant Type XVII Humanized Collagen on Cell Cycle
实验方法:experimental method:
实验分组同实施例1,胶原蛋白共培养终浓度为10mg/mL。The experimental grouping was the same as in Example 1, and the final concentration of collagen co-culture was 10 mg/mL.
将MDA-MB-231乳腺癌细胞用胰酶消化后,加入1ml冰浴预冷70%乙醇中,轻轻吹打混匀,4℃固定30分钟。以1000g离心3-5分钟,沉淀细胞。加入1ml冰浴预冷的PBS,重悬细胞。再次离心沉淀细胞,吸除上清,每管细胞样品中加入0.5mL碘化丙啶染色液,缓慢并充分重悬细胞沉淀,37℃避光温浴30分钟后完成流式检测。MDA-MB-231 breast cancer cells were digested with trypsin, added to 1ml ice-bath pre-cooled 70% ethanol, mixed gently by pipetting, and fixed at 4°C for 30 minutes. Centrifuge at 1000g for 3-5 minutes to pellet the cells. Add 1ml ice-bath pre-cooled PBS to resuspend the cells. Centrifuge again to pellet the cells, aspirate the supernatant, add 0.5mL propidium iodide staining solution to each tube of cell samples, slowly and fully resuspend the cell pellet, and incubate at 37°C for 30 minutes in the dark to complete the flow detection.
实验结果:Experimental results:
结果如图10所示,重组XVII型人源化胶原蛋白可以影响乳腺癌细胞的分裂周期。The results are shown in FIG. 10 , the recombinant type XVII humanized collagen can affect the division cycle of breast cancer cells.
分组培养后,和对照组相比,重组XVII型人源化胶原蛋白处理后,处于休眠周期(G1/G0期)的乳腺癌细胞比例显著上升,处于增殖活性周期(G2/M期)的乳腺癌细胞比例显著下降,说明重组XVII型人源化胶原蛋白能够促进乳腺癌细胞进入休眠周期(G1/G0期)。After group culture, compared with the control group, the proportion of breast cancer cells in the dormant cycle (G1/G0 phase) increased significantly after treatment with recombinant XVII humanized collagen, and the breast cancer cells in the proliferative active cycle (G2/M phase) The proportion of cancer cells decreased significantly, indicating that the recombinant type XVII humanized collagen can promote breast cancer cells to enter the dormant cycle (G1/G0 phase).
总结:Summarize:
乳腺癌目前在全世界癌症发病率中位居第一,治疗方式有限,尤其是三阴性乳腺癌,治疗手段单一,复发转移率高,急需更有效安全的治疗手段。重组XVII型人源化胶原蛋白具有高水溶性,高生物活性的特点。现有实验证实,重组XVII型人源化胶原蛋白可以抑制乳腺癌细胞的生长增殖能力,并且可以抑制肿瘤细胞的迁移特性,且其机制可能依赖于促进肿瘤细胞进入休眠周期。这将为重组XVII型人源化胶原蛋白在临床上应用于乳腺癌患者的安全性和有效性提供一定的理论基础。Breast cancer currently ranks first in the incidence of cancer in the world. There are limited treatment options, especially for triple-negative breast cancer. The treatment method is single, and the recurrence and metastasis rate is high. More effective and safe treatment methods are urgently needed. Recombinant type XVII humanized collagen has the characteristics of high water solubility and high biological activity. Existing experiments have confirmed that recombinant type XVII humanized collagen can inhibit the growth and proliferation of breast cancer cells, and can inhibit the migration characteristics of tumor cells, and its mechanism may depend on promoting tumor cells to enter the dormant cycle. This will provide a certain theoretical basis for the safety and effectiveness of recombinant XVII humanized collagen in clinical application to breast cancer patients.
Claims (7)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202310107604.5A CN116270982A (en) | 2023-02-09 | 2023-02-09 | Use of recombinant type XVII humanized collagen in the treatment of breast cancer |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202310107604.5A CN116270982A (en) | 2023-02-09 | 2023-02-09 | Use of recombinant type XVII humanized collagen in the treatment of breast cancer |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN116270982A true CN116270982A (en) | 2023-06-23 |
Family
ID=86827816
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202310107604.5A Pending CN116270982A (en) | 2023-02-09 | 2023-02-09 | Use of recombinant type XVII humanized collagen in the treatment of breast cancer |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN116270982A (en) |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN117843763A (en) * | 2024-01-08 | 2024-04-09 | 山西锦波生物医药股份有限公司 | Method for biosynthesizing type XVII collagen, a structural material for the human body |
| CN118903389A (en) * | 2023-08-01 | 2024-11-08 | 山西锦波生物医药股份有限公司 | Application of recombinant XVII type humanized collagen in cervical cancer treatment |
| CN119264242A (en) * | 2024-10-11 | 2025-01-07 | 常州泰美瑞生物科技有限公司 | A recombinant type XVII collagen and its preparation method and application |
| CN119462898A (en) * | 2023-12-05 | 2025-02-18 | 北京华熙荣熙生物技术研究有限公司 | Recombinant type XVII collagen with triple helix structure and its application |
| CN119751646A (en) * | 2024-12-31 | 2025-04-04 | 江苏创健医疗科技股份有限公司 | Highly stable recombinant type ⅩⅦ collagen and its construction method and application |
| CN120248093A (en) * | 2025-06-04 | 2025-07-04 | 浙江暨北生物科技有限公司 | Recombinant humanized type A collagen and type XVII collagen and its construction and application |
| CN120284771A (en) * | 2025-03-12 | 2025-07-11 | 山西锦波生物医药股份有限公司 | Application of recombinant humanized type XVII collagen in preventing hair loss and promoting hair growth |
| CN120309715A (en) * | 2025-06-16 | 2025-07-15 | 杭州凯曼健康科技有限公司 | Recombinant humanized type XVII collagen and its preparation method and application |
| CN120818046A (en) * | 2025-09-19 | 2025-10-21 | 浙江崇山生物制品有限公司 | A synthetic type XVII humanized collagen and its preparation method and application |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20190040473A1 (en) * | 2013-04-18 | 2019-02-07 | Gencurix Inc. | Genetic marker for early breast cancer prognosis prediction and diagnosis, and use thereof |
| CN110845603A (en) * | 2019-10-31 | 2020-02-28 | 中国科学院生物物理研究所 | Human collagen type 17 polypeptide, production method and use thereof |
| US20210199660A1 (en) * | 2019-11-22 | 2021-07-01 | 10X Genomics, Inc. | Biomarkers of breast cancer |
| CN113185604A (en) * | 2021-05-13 | 2021-07-30 | 江苏创健医疗科技有限公司 | Recombinant human XVII type collagen, preparation method and application |
-
2023
- 2023-02-09 CN CN202310107604.5A patent/CN116270982A/en active Pending
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20190040473A1 (en) * | 2013-04-18 | 2019-02-07 | Gencurix Inc. | Genetic marker for early breast cancer prognosis prediction and diagnosis, and use thereof |
| CN110845603A (en) * | 2019-10-31 | 2020-02-28 | 中国科学院生物物理研究所 | Human collagen type 17 polypeptide, production method and use thereof |
| US20210199660A1 (en) * | 2019-11-22 | 2021-07-01 | 10X Genomics, Inc. | Biomarkers of breast cancer |
| CN113185604A (en) * | 2021-05-13 | 2021-07-30 | 江苏创健医疗科技有限公司 | Recombinant human XVII type collagen, preparation method and application |
Non-Patent Citations (1)
| Title |
|---|
| MUTTARIN LOTHONG ET AL.: "Collagen XVII inhibits breast cancer cell proliferation and growth through deactivation of the AKT/mTOR signaling pathway", 《PLOS ONE》, vol. 16, no. 7, 22 July 2021 (2021-07-22) * |
Cited By (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN118903389A (en) * | 2023-08-01 | 2024-11-08 | 山西锦波生物医药股份有限公司 | Application of recombinant XVII type humanized collagen in cervical cancer treatment |
| CN118903389B (en) * | 2023-08-01 | 2026-02-03 | 山西锦波生物医药股份有限公司 | Application of recombinant XVII type humanized collagen in cervical cancer treatment |
| CN119462898A (en) * | 2023-12-05 | 2025-02-18 | 北京华熙荣熙生物技术研究有限公司 | Recombinant type XVII collagen with triple helix structure and its application |
| WO2025148254A1 (en) * | 2024-01-08 | 2025-07-17 | 山西锦波生物医药股份有限公司 | Method for biosynthesis of human structural material type xvii collagen |
| CN117843763A (en) * | 2024-01-08 | 2024-04-09 | 山西锦波生物医药股份有限公司 | Method for biosynthesizing type XVII collagen, a structural material for the human body |
| CN119264242A (en) * | 2024-10-11 | 2025-01-07 | 常州泰美瑞生物科技有限公司 | A recombinant type XVII collagen and its preparation method and application |
| CN119751646A (en) * | 2024-12-31 | 2025-04-04 | 江苏创健医疗科技股份有限公司 | Highly stable recombinant type ⅩⅦ collagen and its construction method and application |
| CN120284771A (en) * | 2025-03-12 | 2025-07-11 | 山西锦波生物医药股份有限公司 | Application of recombinant humanized type XVII collagen in preventing hair loss and promoting hair growth |
| CN120284771B (en) * | 2025-03-12 | 2025-12-23 | 山西锦波生物医药股份有限公司 | Application of recombinant XVII type humanized collagen in hair loss prevention and hair growth |
| CN120248093A (en) * | 2025-06-04 | 2025-07-04 | 浙江暨北生物科技有限公司 | Recombinant humanized type A collagen and type XVII collagen and its construction and application |
| CN120309715A (en) * | 2025-06-16 | 2025-07-15 | 杭州凯曼健康科技有限公司 | Recombinant humanized type XVII collagen and its preparation method and application |
| CN120818046A (en) * | 2025-09-19 | 2025-10-21 | 浙江崇山生物制品有限公司 | A synthetic type XVII humanized collagen and its preparation method and application |
| CN120818046B (en) * | 2025-09-19 | 2025-12-05 | 浙江崇山生物制品有限公司 | Synthetic XVII type humanized collagen and preparation method and application thereof |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN116270982A (en) | Use of recombinant type XVII humanized collagen in the treatment of breast cancer | |
| JP2806634B2 (en) | Pharmaceutical formulations for controlling tumors associated with prostate, gastric and breast cancer | |
| CN118903389B (en) | Application of recombinant XVII type humanized collagen in cervical cancer treatment | |
| CN115957306B (en) | Application of a Caerin1.1/1.9 combined with anti-CD47 antibody in the preparation of drugs for the treatment of melanoma | |
| CN112641775B (en) | Application of brucea javanica picrol and analogues thereof in treatment of pituitary adenoma | |
| CN116407620A (en) | Use of recombinant type III humanized collagen in breast cancer treatment | |
| CN116617371A (en) | Use of Recombinant Type III Humanized Collagen in the Treatment of Ovarian Cancer | |
| CN108329381B (en) | A kind of hexadeceptide derived from Kirin cabbage and its application in the preparation of drugs for preventing and treating malignant tumor metastasis | |
| CN116196397A (en) | Application of recombinant type I humanized collagen in breast cancer treatment | |
| CN102068448A (en) | Application of icariside II in preparation of anti-melanoma medicament | |
| JP6872713B2 (en) | Synthetic peptides that increase the radiosensitivity of tumor cells and their use | |
| US5024833A (en) | Combined interferon/antiestrogen therapy for treatment of breast cancer | |
| CN108379578B (en) | Application of interleukin-23 neutralizing antibody in preparation of endometriosis medicine | |
| EP1013281A1 (en) | hCG therapy for the treatment of breast cancer | |
| US6096301A (en) | Combined interferon/antiestrogen therapy for treatment of breast cancer | |
| CN118634310A (en) | A cordycepin preparation and its application in tumor immunotherapy | |
| CN108272810A (en) | Application of the Rhizoma Picrorhizae glucoside II in anti-breast cancer medicines | |
| CN106924271A (en) | Ginseng saponin C-K is used to prepare treatment psoriasis | |
| EP4458367A1 (en) | Application of polypeptide in preparation of product for preventing or treating skin injury diseases | |
| CN113528520B (en) | Circular RNAhas_circ_0000591 and its applications | |
| CN113813403B (en) | Use of METTL3 in preparation of medicines for repairing dental pulp injury | |
| CN102805768A (en) | Application of catclaw buttercup root extract to preparation of anti-lung cancer medicines | |
| Nie et al. | Genetically engineered MSCs overexpressing hepatocyte growth factor for the treatment of human refractory wounds | |
| CN116747311A (en) | Application of PPARδ antagonists combined with SIRPα antibody drugs in the preparation of tumor immunotherapy drugs | |
| CN113244236A (en) | Application of ceritinib in preparation of medicine for treating thyroid-associated ophthalmopathy |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination |