CN116196286A - Sodium mycophenolate gastric floating tablet - Google Patents
Sodium mycophenolate gastric floating tablet Download PDFInfo
- Publication number
- CN116196286A CN116196286A CN202310129298.5A CN202310129298A CN116196286A CN 116196286 A CN116196286 A CN 116196286A CN 202310129298 A CN202310129298 A CN 202310129298A CN 116196286 A CN116196286 A CN 116196286A
- Authority
- CN
- China
- Prior art keywords
- gastric
- sodium mycophenolate
- mycophenolate
- sodium
- floating tablet
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0053—Mouth and digestive tract, i.e. intraoral and peroral administration
- A61K9/0065—Forms with gastric retention, e.g. floating on gastric juice, adhering to gastric mucosa, expanding to prevent passage through the pylorus
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/335—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin
- A61K31/365—Lactones
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2013—Organic compounds, e.g. phospholipids, fats
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2013—Organic compounds, e.g. phospholipids, fats
- A61K9/2018—Sugars, or sugar alcohols, e.g. lactose, mannitol; Derivatives thereof, e.g. polysorbates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/2027—Organic macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyvinyl pyrrolidone, poly(meth)acrylates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/2031—Organic macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyethylene glycol, polyethylene oxide, poloxamers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/205—Polysaccharides, e.g. alginate, gums; Cyclodextrin
- A61K9/2054—Cellulose; Cellulose derivatives, e.g. hydroxypropyl methylcellulose
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/06—Immunosuppressants, e.g. drugs for graft rejection
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Epidemiology (AREA)
- Immunology (AREA)
- Biophysics (AREA)
- Molecular Biology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Physiology (AREA)
- Nutrition Science (AREA)
- Transplantation (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Medicinal Preparation (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
技术领域technical field
本发明属于医药技术领域,更具体涉及一种麦考酚钠胃漂浮片。The invention belongs to the technical field of medicine, and more specifically relates to a mycophenolate sodium gastric floating tablet.
背景技术Background technique
麦考酚钠是高效、选择性、非竞争性、可逆性的次黄嘌呤单核苷酸脱氢酶(IMPDH)抑制剂,可抑制鸟嘌呤核苷酸的经典合成途径。霉酚酸(MPA)对淋巴细胞具有高度选择作用。抑制淋巴细胞增殖所需的MPA浓度,对大多数淋巴细胞无抑制作用,MPA还能通过直接抑制B细胞的增殖来抑制抗体的形成。治疗量的麦考酚钠并不抑制多糖激活人外周血淋巴细胞产生白介素-I,也不抑制有丝分裂原激活的外周淋巴细胞合成白介素-Ⅱ和其受体表达,这点也不同于环孢霉素、FK-506。此外,MPA介导的体外淋巴细胞三磷酸鸟苷的耗竭可抑制甘露糖和岩藻糖转化成糖蛋白。通过这种机制,MPA可降低淋巴细胞和单核细胞在慢性炎症部位的聚集。Mycophenolate Sodium is a highly efficient, selective, non-competitive and reversible inhibitor of hypoxanthine mononucleotide dehydrogenase (IMPDH), which can inhibit the classical synthesis pathway of guanine nucleotides. Mycophenolic acid (MPA) has a highly selective effect on lymphocytes. The concentration of MPA required to inhibit the proliferation of lymphocytes has no inhibitory effect on most lymphocytes. MPA can also inhibit the formation of antibodies by directly inhibiting the proliferation of B cells. Therapeutic doses of mycophenolate sodium did not inhibit the polysaccharide-activated human peripheral blood lymphocytes to produce interleukin-I, nor inhibited the synthesis of interleukin-II and its receptor expression by mitogen-activated peripheral lymphocytes, which is also different from cyclosporine Su, FK-506. Furthermore, MPA-mediated depletion of guanosine triphosphate in lymphocytes in vitro inhibited the conversion of mannose and fucose into glycoproteins. Through this mechanism, MPA reduces the accumulation of lymphocytes and monocytes at sites of chronic inflammation.
2009年4月25日,北京诺华制药有限公司在京隆重召开了器官移植抑制排斥反应创新药物麦考酚钠肠溶片(米芙)上市会。米芙是美国食品药品管理局(FDA)批准的唯一一个麦考酚钠肠溶剂,并于2008年12月18日获得我国国家食品药品监督管理局(SFDA)批准,与环孢素A和皮质类固醇合用,预防接受同种异体肾移植成年患者术后急性排斥反应。麦考酚钠肠溶片以其良好的抑制排斥反应疗效和药物耐受性,提高肾移植患者接受免疫抑制剂治疗的依从性,减少因药物副作用而导致的移植物功能丧失。此前,米芙已在包括瑞士和美国在内的91个国家获得了上市批准。On April 25, 2009, Beijing Novartis Pharmaceuticals Co., Ltd. grandly held a launch meeting for the innovative drug Mycophenolate Sodium Enteric-Coated Tablets (Miff) for suppressing rejection in organ transplantation in Beijing. Mifu is the only mycophenolate sodium enteric solution approved by the US Food and Drug Administration (FDA), and it was approved by my country's State Food and Drug Administration (SFDA) on December 18, 2008. It is compatible with cyclosporine A and corticosteroids Combination steroids for prophylaxis of acute postoperative rejection in adult patients undergoing renal allograft transplantation. Mycophenolate Sodium Enteric-coated Tablets can improve the compliance of kidney transplant patients receiving immunosuppressant therapy and reduce the loss of graft function caused by drug side effects due to its good rejection suppression effect and drug tolerance. Previously, Mifu had been approved for marketing in 91 countries including Switzerland and the United States.
但是市售的肠溶片采用的肠溶材料包衣技术,制备过程中使用的有机溶剂,对环境影响较大,且设备成本较高;鉴于此,特提出本发明。However, the enteric-coated material coating technology used in commercially available enteric-coated tablets and the organic solvent used in the preparation process have a greater impact on the environment, and the equipment cost is higher; in view of this, the present invention is proposed.
发明内容Contents of the invention
本发明的目的在于公开一种口服的麦考酚钠胃漂浮片。该口服制剂生产工艺简单,对设备需求较低,服用方便,且个体差异小。The purpose of the present invention is to disclose an oral mycophenolate sodium gastric floating tablet. The oral preparation has a simple production process, requires less equipment, is convenient to take, and has little individual difference.
为实现上述目的,本发明采用以下技术方案:To achieve the above object, the present invention adopts the following technical solutions:
本发明公开了一种麦考酚钠胃漂浮片,所述麦考酚钠胃漂浮片的原料的用量为:每制备1000片麦考酚钠胃漂浮片,所用原料包括麦考酚钠180g、羟丙基甲基纤维素K4M 130~160g、卡波姆934 20~40g、乳糖50~65g、PEG4000 25~40g、硬脂酸镁2.7g;The invention discloses a mycophenolate sodium gastric-floating tablet. The dosage of the raw materials of the mycophenolate sodium gastric-floating tablet is as follows: for every 1000 mycophenolate sodium gastric-floating tablets, the raw materials used include 180g of mycophenolate sodium, Hydroxypropyl methylcellulose K4M 130~160g, Carbomer 934 20~40g, lactose 50~65g, PEG4000 25~40g, magnesium stearate 2.7g;
按照所述麦考酚钠胃漂浮片的原料用量制备麦考酚钠胃漂浮片。Mycophenolate Sodium Gastric Floating Tablets are prepared according to the raw material dosage of the Mycophenolate Sodium Gastric Floating Tablets.
在一些实施方式中,制备所述麦考酚钠胃漂浮片过程中,流化床制粒时不加入液体物料,且在流化床状态下冷却后得到颗粒。In some embodiments, during the preparation of the mycophenolate sodium gastric floating tablet, no liquid material is added during fluidized bed granulation, and the granules are obtained after cooling in a fluidized bed state.
在一些实施方式中,麦考酚钠胃漂浮片的硬度为5-7KN。In some embodiments, the hardness of mycophenolate sodium gastric floating tablet is 5-7KN.
与现有技术相比,本发明的有益效果是:Compared with prior art, the beneficial effect of the present invention is:
本发明所公开的麦考酚钠胃漂浮片,利用合理的原料,处方中无有机溶剂;采用现有技术中简单的工艺制备而成,其设备普及度高,适合企业规模化生产,极具市场开发前景。利用药物本身的溶解特点,达到肠溶片一样的疗效。The mycophenolate sodium gastric floating tablet disclosed by the present invention utilizes reasonable raw materials, and there is no organic solvent in the prescription; it is prepared by a simple process in the prior art, and its equipment is highly popularized, suitable for large-scale production of enterprises, and extremely market development prospects. Utilize the dissolution characteristics of the drug itself to achieve the same curative effect as enteric-coated tablets.
具体实施方式Detailed ways
下面结合各实施方式对本发明进行详细说明,但应当说明的是,这些实施方式并非对本发明的限制,本领域普通技术人员根据这些实施方式所作的功能、方法、或者结构上的等效变换或替代,均属于本发明的保护范围之内。The present invention will be described in detail below in conjunction with various embodiments, but it should be noted that these embodiments are not limitations of the present invention, and those of ordinary skill in the art can make functional, method, or structural equivalent transformations or replacements based on these embodiments. , all fall within the protection scope of the present invention.
实施例一Embodiment one
处方:(制备1000片)Prescription: (prepare 1000 tablets)
实施例二Embodiment two
处方:(制备1000片)Prescription: (prepare 1000 tablets)
实施例一、二按照以下方法制备麦考酚钠胃漂浮片:Embodiments 1 and 2 Mycophenolate Sodium Gastric Floating Tablets were prepared according to the following method:
将麦考酚钠、羟丙基甲基纤维素,卡波姆934,乳糖,PEG4000分别过80目筛;Pass mycophenolate sodium, hydroxypropyl methylcellulose, carbomer 934, lactose, and PEG4000 through an 80-mesh sieve;
称取处方量麦考酚钠,羟丙基甲基纤维素,卡波姆934,乳糖,PEG4000于混合机中混合10min;Weigh the prescription amount of mycophenolate sodium, hydroxypropyl methylcellulose, carbomer 934, lactose, PEG4000 and mix them in a mixer for 10 minutes;
将混合物料转移至流化床中,制粒,设定物料温度为60摄氏度,风机频率设置为15HZ;出风温度设置为40℃;制粒完成后,关闭加热,设定风机频率18Hz,冷却,当物料温度降至30℃时,收集物料。Transfer the mixed material to the fluidized bed, granulate, set the material temperature to 60 degrees Celsius, set the fan frequency to 15HZ; set the outlet temperature to 40°C; after the granulation is completed, turn off the heating, set the fan frequency to 18Hz, and cool , when the temperature of the material dropped to 30°C, the material was collected.
将所得物料与硬脂酸镁混合后压片,压片后制得的麦考酚钠胃漂浮片片剂硬度为5-7KN。The obtained material is mixed with the magnesium stearate and then compressed into tablets, and the hardness of the mycophenolate sodium gastric floating tablets obtained after the compression is 5-7KN.
实施例三、体内药动学实验:Embodiment three, in vivo pharmacokinetic experiment:
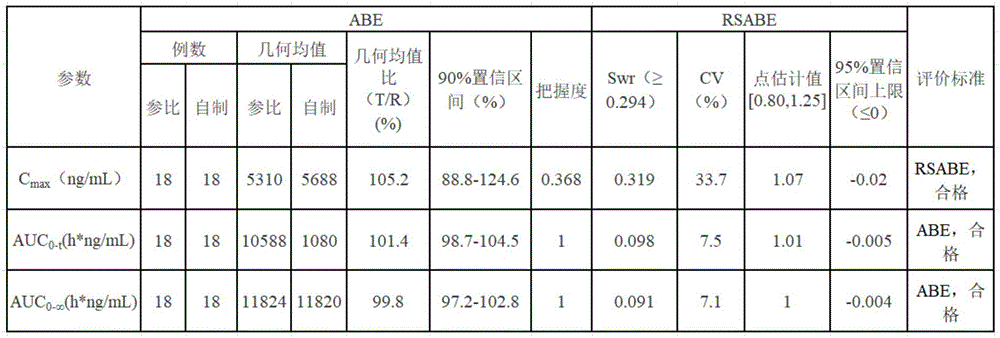
取实验家兔18只(体重范围1.5Kg-2.0Kg),随机分成2组,每组9只,分别灌服实施例二制得的麦考酚钠胃漂浮片和市售肠溶片(米芙),按250mg/Kg剂量进行给药,分别于给药15min,30min,45min,1h,2h,3h,4h,6h,8h,10h后于家兔股静脉取血1.0mL,加入甲醇超声20min,离心,取上清液20μL,HPLC检测含量,采用两制剂、四周期、两序列重复交叉设计,清洗周期7d。Get 18 experimental rabbits (body weight range 1.5Kg-2.0Kg), be divided into 2 groups at random, every group of 9, respectively gavage the mycophenolate sodium gastric floating tablet that embodiment two makes and commercially available enteric-coated tablet (rice Fu), according to the dosage of 250mg/Kg, after 15min, 30min, 45min, 1h, 2h, 3h, 4h, 6h, 8h, 10h, take 1.0mL of blood from the femoral vein of rabbits, and add methanol for 20min , centrifuged, and 20 μL of the supernatant was taken, and the content was detected by HPLC. Two preparations, four cycles, and two sequences were used to repeat the crossover design, and the cleaning cycle was 7 days.
各制剂的药代动力学参数如下表示:The pharmacokinetic parameters of each preparation are as follows:
上表的结果表明,按照市实施例二制备得到的麦考酚钠漂浮片,与市售制剂相比,可认为疗效一致。The results in the above table show that compared with the commercially available preparations, the mycophenolate sodium floating tablets prepared according to Commercial Example 2 can be considered to have the same curative effect.
本发明所公开的的麦考酚钠口服制剂具有生产工艺简单,解决了麦考酚钠与生产过程中遇水形成水合物的风险,生产设备通用,成本低,患者携带和吞服方便。与现有剂型具有一样的疗效。The oral preparation of mycophenolate sodium disclosed in the present invention has a simple production process, solves the risk of forming hydrates between mycophenolate sodium and water in the production process, has common production equipment, low cost, and is convenient for patients to carry and swallow. It has the same curative effect as the existing dosage form.
以上所述的仅是本发明的一些实施方式,应当指出,对于本领域的普通技术人员来说,在不脱离本发明的创造构思的前提下,还可以做出其它变形和改进,这些都属于本发明的保护范围。What have been described above are only some embodiments of the present invention, and it should be pointed out that for those of ordinary skill in the art, without departing from the creative concept of the present invention, other deformations and improvements can also be made, and these all belong to protection scope of the present invention.
Claims (3)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202310129298.5A CN116196286A (en) | 2023-02-17 | 2023-02-17 | Sodium mycophenolate gastric floating tablet |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202310129298.5A CN116196286A (en) | 2023-02-17 | 2023-02-17 | Sodium mycophenolate gastric floating tablet |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN116196286A true CN116196286A (en) | 2023-06-02 |
Family
ID=86514256
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202310129298.5A Pending CN116196286A (en) | 2023-02-17 | 2023-02-17 | Sodium mycophenolate gastric floating tablet |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN116196286A (en) |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20110223249A1 (en) * | 2007-08-13 | 2011-09-15 | Panacea Biotec Ltd. | Extended release compositions comprising mycophenolate sodium and processes thereof |
| CN110960499A (en) * | 2019-12-31 | 2020-04-07 | 卓和药业集团有限公司 | Posaconazole gastric floating tablet and preparation method thereof |
| US20210128479A1 (en) * | 2017-01-26 | 2021-05-06 | Triastek, Inc. | Dosage forms of controlled release at specific gastrointestinal sites |
-
2023
- 2023-02-17 CN CN202310129298.5A patent/CN116196286A/en active Pending
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20110223249A1 (en) * | 2007-08-13 | 2011-09-15 | Panacea Biotec Ltd. | Extended release compositions comprising mycophenolate sodium and processes thereof |
| US20210128479A1 (en) * | 2017-01-26 | 2021-05-06 | Triastek, Inc. | Dosage forms of controlled release at specific gastrointestinal sites |
| CN110960499A (en) * | 2019-12-31 | 2020-04-07 | 卓和药业集团有限公司 | Posaconazole gastric floating tablet and preparation method thereof |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN103550242B (en) | Pharmaceutical composition for treating hepatic fibrosis and preparation method thereof | |
| CN103893247A (en) | Pharmaceutical composition as well as preparation method and application thereof | |
| CN104887641A (en) | Palbociclib gastric-floating tablet and preparation method thereof | |
| CN103393617A (en) | Febuxostat tablet and preparation method thereof | |
| CN106265581B (en) | Tranexamic acid tablet and preparation method thereof | |
| CN103610658B (en) | Immunomodulator slow-release preparation and preparation method thereof | |
| CN101862333B (en) | Stable sodium levofolinate oral preparation and preparation method thereof | |
| CN110960499A (en) | Posaconazole gastric floating tablet and preparation method thereof | |
| CN116196286A (en) | Sodium mycophenolate gastric floating tablet | |
| CN105412023A (en) | Frovatriptan succinate controlled-release granule and preparation method thereof | |
| CN117298085A (en) | Medical application of tolypic acid and derivative thereof in preparation of medicines for treating immune and inflammation related diseases | |
| CN104650034A (en) | Stable axitinib compound | |
| CN103463554B (en) | A kind of treat malignant tumor pharmaceutical composition and preparation method and purposes | |
| CN106389376A (en) | Norfloxacin capsules and preparation method thereof | |
| CN107080741A (en) | Pirfenidone sustained release preparation and preparation method | |
| CN102727455A (en) | Tadalafil oral disintegrating tablet and preparation method thereof | |
| CN107982240B (en) | Potassium sodium dehydroandroan drographolide succinate enteric coated granules capable of being accurately dissolved out and preparation method thereof | |
| WO2017004733A1 (en) | Ginsenoside c-k oral solid preparation and preparation method thereof | |
| CN101023943A (en) | Myricetin dispersion tablets for treating cardio-cerebral blood vessel diseases | |
| CN101416950B (en) | Preparation method of camellia oleifera saponin controlled-release tablet | |
| CN100546578C (en) | A kind of anticancer drug composition and its application in the preparation of anticancer drug | |
| CN103393604B (en) | Tamoxifen citrate enteric coated particles | |
| CN104857180B (en) | Composition for resisting fatigue and improving immunity and preparation method and application thereof | |
| CN115245495A (en) | A kind of sitagliptin and metformin tablet and preparation method thereof | |
| CN103142544B (en) | Ubenimex capsule composition and preparation method thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| RJ01 | Rejection of invention patent application after publication | ||
| RJ01 | Rejection of invention patent application after publication |
Application publication date: 20230602 |