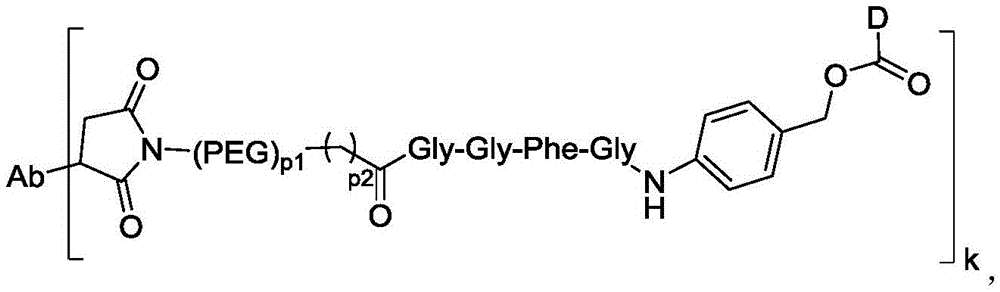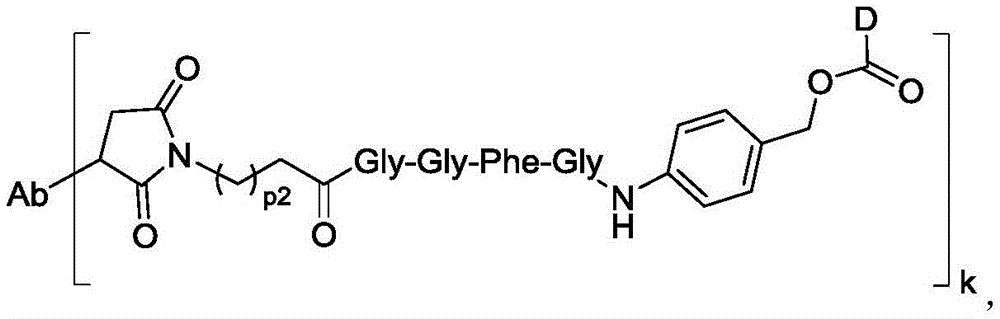CN115845080A - Eribulin derivative-anti-folate receptor antibody conjugate - Google Patents
Eribulin derivative-anti-folate receptor antibody conjugate Download PDFInfo
- Publication number
- CN115845080A CN115845080A CN202210867359.3A CN202210867359A CN115845080A CN 115845080 A CN115845080 A CN 115845080A CN 202210867359 A CN202210867359 A CN 202210867359A CN 115845080 A CN115845080 A CN 115845080A
- Authority
- CN
- China
- Prior art keywords
- antibody
- cancer
- drug conjugate
- peg
- integer
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Images
Landscapes
- Peptides Or Proteins (AREA)
Abstract
本公开涉及艾日布林衍生物‑抗叶酸受体抗体偶联物。艾日布林衍生物的药物偶联物具体而言,提供了艾日布林衍生物‑抗叶酸受体α(FRA)抗体或其抗原结合片段的抗体偶联物、其制备方法及其在医药上的应用。本公开进一步涉及通过施用本文提供的抗体‑药物偶联物,用于治疗癌症的方法和组合物。The present disclosure relates to eribulin derivative-anti-folate receptor antibody conjugates. Drug conjugates of eribulin derivatives Specifically, antibody conjugates of eribulin derivatives-anti-folate receptor α (FRA) antibodies or antigen-binding fragments thereof, their preparation methods and their use in Medical applications. The present disclosure further relates to methods and compositions for treating cancer by administering the antibody-drug conjugates provided herein.
Description
技术领域Technical Field
本公开涉及艾日布林衍生物-抗叶酸受体抗体偶联物。The present disclosure relates to an eribulin derivative-anti-folate receptor antibody conjugate.
背景技术Background Art
抗体药物偶联物(antibody drug conjugate,ADC)把单克隆抗体或者抗体片段通过稳定的化学接头化合物与具有生物活性的药物相连,充分利用了抗体对正常细胞和肿瘤细胞表面抗原结合的特异性和药物的高效性,同时又避免了前者疗效偏低和后者毒副作用过大等缺陷。这也就意味着,与以往传统的化疗药物相比,抗体药物偶联物能精准地结合肿瘤细胞并降低将对正常细胞的影响(Mullard A,(2013)Nature Reviews Drug Discovery,12:329–332;DiJoseph JF,Armellino DC,(2004)Blood,103:1807-1814)。Antibody drug conjugates (ADCs) connect monoclonal antibodies or antibody fragments to biologically active drugs through stable chemical linker compounds, making full use of the specificity of antibodies binding to surface antigens of normal cells and tumor cells and the high efficiency of drugs, while avoiding the defects of the former's low efficacy and the latter's excessive toxic side effects. This means that compared with traditional chemotherapy drugs, antibody drug conjugates can accurately bind to tumor cells and reduce the impact on normal cells (Mullard A, (2013) Nature Reviews Drug Discovery, 12: 329-332; DiJoseph JF, Armellino DC, (2004) Blood, 103: 1807-1814).
至2000年第一个抗体药物偶联物Mylotarg(吉妥珠单抗奥唑米星(gemtuzumabozogamicin),惠氏制药有限公司)被美国FDA批准上市用于治疗急性髓细胞白血病开始,进入临床阶段的ADC药物共有164项,大多数(n=100)处于临床一期,46项进入临床二期,7项进入临床三期,BLA申请药物有3项。三代上市的代表药物分别是基因泰克和Genetics公司合作开发的Polatuzumab vedotin(商品名,Polivy,2019年6月获批上市),Agensys(安斯泰来的子公司)和Seattle Genetics合作开发的Enfortumab vedotin(商品名,Padcev,2019年12月)以及第一三共开发的Fam-trastuzumab deruxtecan(商品名,Enhertu)。Since the first antibody-drug conjugate Mylotarg (gemtuzumab ozogamicin, Wyeth Pharmaceuticals Co., Ltd.) was approved by the U.S. FDA for the treatment of acute myeloid leukemia in 2000, there have been 164 ADC drugs in clinical trials, most of which (n=100) are in Phase I clinical trials, 46 are in Phase II clinical trials, 7 are in Phase III clinical trials, and 3 have applied for BLA. The representative drugs of the third generation are Polatuzumab vedotin (trade name, Polivy, approved for marketing in June 2019) jointly developed by Genentech and Genetics, Enfortumab vedotin (trade name, Padcev, December 2019) jointly developed by Agensys (a subsidiary of Astellas) and Seattle Genetics, and Fam-trastuzumab deruxtecan (trade name, Enhertu) developed by Daiichi Sankyo.
微管为与包括细胞内迁移和转运、细胞信号传导和维持细胞形状的多种细胞功能相关的有力的细丝状细胞骨架蛋白。微管也在有丝分裂细胞分裂中通过形成染色体分成两个子细胞所需的有丝分裂纺锤体而起到关键作用。所有细胞中微管的生物功能大部分由其聚合动力学调节,这通过α和β微管蛋白二聚物可逆、非共价地加在微管两端进行。这种动力学行为和所产生的对微管长度的控制为有丝分裂纺锤体的适当功能所不可缺少的。甚至微管动力学的微小改变也会牵涉轴检查点,抑制有丝分裂时细胞周期进展,且随后引起细胞死亡(Mukhtar等人(2014)Mol.Cancer Ther.13:275-84)。由于癌细胞的细胞分裂快速,所以与正常细胞相比,其一般对结合于微管蛋白且破坏其正常功能的化合物更加敏感。因此,微管蛋白抑制剂和其它靶向微管剂有望成为一类治疗癌症的药物(Dumontet和Jordan(2010)Nat.Rev.Drug Discov.9:790-803)。Microtubules are powerful, filamentous cytoskeletal proteins associated with a variety of cellular functions including intracellular migration and transport, cell signaling, and maintenance of cell shape. Microtubules also play a key role in mitotic cell division by forming the mitotic spindle required for the separation of chromosomes into two daughter cells. The biological functions of microtubules in all cells are largely regulated by their polymerization dynamics, which is reversibly and non-covalently attached to the ends of microtubules by α and β tubulin dimers. This dynamic behavior and the resulting control of microtubule length are indispensable for the proper function of the mitotic spindle. Even minor changes in microtubule dynamics can involve the axis checkpoint, inhibit cell cycle progression during mitosis, and subsequently cause cell death (Mukhtar et al. (2014) Mol. Cancer Ther. 13: 275-84). Due to the rapid cell division of cancer cells, they are generally more sensitive to compounds that bind to tubulin and disrupt its normal function than normal cells. Therefore, tubulin inhibitors and other agents targeting microtubules are expected to become a class of drugs for treating cancer (Dumontet and Jordan (2010) Nat. Rev. Drug Discov. 9: 790-803).
另一方面,叶酸受体α(FRA)为一种结合叶酸盐的经甘油磷脂酰肌醇(GPI)连接的膜蛋白。虽然未充分了解FRA在正常和癌组织的生物学中的作用,但其在高百分比的上皮细胞来源的卵巢癌上(O′Shannessy等人,Int.J.Gynecol.Pathol.2003,32(3):258-68),以及在一定百分比的非小细胞肺癌中(Christoph等人Clin.Lung Cancer 2014 15(5):320-30)过度表达。FRA在正常组织中也具有有限表达。这些特性使得FRA成为癌症免疫疗法的吸引人的标靶。On the other hand, folate receptor alpha (FRA) is a glycerophosphatidylinositol (GPI)-linked membrane protein that binds folate. Although the role of FRA in the biology of normal and cancerous tissues is not fully understood, it is overexpressed in a high percentage of ovarian cancers of epithelial origin (O'Shannessy et al., Int. J. Gynecol. Pathol. 2003, 32(3): 258-68), and in a certain percentage of non-small cell lung cancers (Christoph et al. Clin. Lung Cancer 2014 15(5): 320-30). FRA also has limited expression in normal tissues. These properties make FRA an attractive target for cancer immunotherapy.
发明内容Summary of the invention
本公开提供了一种抗体-药物偶联物(ADC),其具有式I所示结构或其药学上可接受的盐或溶剂化物:The present disclosure provides an antibody-drug conjugate (ADC) having a structure as shown in Formula I or a pharmaceutically acceptable salt or solvate thereof:
Ab-(L-D)k (I)Ab-(LD) k (I)
其中,Ab为抗叶酸受体α(FRA)抗体或其抗原结合片段,Wherein, Ab is an anti-folate receptor α (FRA) antibody or an antigen-binding fragment thereof,
L为将Ab共价连接于D的连接子,且k为1至20(包括1、2、3、4、5、6、7、8、9、10、11、12、13、14、15、16、17、18、19、20或任意两数值之间任意数值),L is a linker that covalently links Ab to D, and k is 1 to 20 (including 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20 or any number between any two numbers),
-D如式所示:-D is as follows:
在一些实施方案中,抗体-药物偶联物Ab-(L-D)k中k选自1至10,可以为整数,也可以为小数。In some embodiments, k in the antibody-drug conjugate Ab-(LD) k is selected from 1 to 10 and can be an integer or a decimal.
在一些实施方案中,连接子在细胞外是稳定的,使得ADC在存在于细胞外环境中时保持完整,但在例如癌细胞的细胞中内化时能够裂解。在一些实施方案中,当ADC进入表达对ADC的抗体部分具有特异性的抗原的细胞时,艾日布林衍生物药物部分从抗体部分裂解,且裂解释放艾日布林衍生物的未修饰形式。在一些实施方案中,连接子包含被定位成使得所述连接子或所述抗体部分中没有部分在裂解后保持结合于艾日布林衍生物的可裂解部分。In some embodiments, the linker is stable outside of the cell, so that the ADC remains intact when present in the extracellular environment, but is capable of cleavage when internalized in a cell, such as a cancer cell. In some embodiments, when the ADC enters a cell expressing an antigen specific for the antibody portion of the ADC, the eribulin derivative drug portion is cleaved from the antibody portion, and the cleavage releases the unmodified form of the eribulin derivative. In some embodiments, the linker comprises a cleavable portion positioned so that no portion of the linker or the antibody portion remains bound to the eribulin derivative after cleavage.
在一些实施方案中,连接子中的可裂解部分为可裂解肽部分。在一些实施方案中,相对于包含替代可裂解部分的ADC,包含可裂解肽部分的ADC显示较低的聚集水平,改善的抗体:药物比率,增加的癌细胞的靶向杀死,减少的非癌细胞的脱靶杀死,和/或较高的药物负载。在一些实施方案中,相对于不可裂解的连接子,添加可裂解部分增加细胞毒性和/或效力。在一些实施方案中,增加的效力和/或细胞毒性是在表达中等水平的由ADC的抗体部分所靶向的抗原(例如中等FRA表达)的癌症中。在一些实施方案中,可裂解肽部分能够由酶裂解,且连接子为酶能够裂解的连接子。在一些实施方案中,酶为组织蛋白酶,且连接子为组织蛋白酶能够裂解的连接子。在某些实施方案中,与替代分裂机制相比,酶能够裂解的连接子(例如组织蛋白酶能够裂解的连接子)显示上述改善特性中的一种或多种。In some embodiments, the cleavable portion in the linker is a cleavable peptide portion. In some embodiments, relative to an ADC comprising an alternative cleavable portion, an ADC comprising a cleavable peptide portion shows a lower aggregation level, an improved antibody: drug ratio, an increased targeted killing of cancer cells, a reduced off-target killing of non-cancerous cells, and/or a higher drug load. In some embodiments, relative to a non-cleavable linker, the addition of a cleavable portion increases cytotoxicity and/or efficacy. In some embodiments, the increased efficacy and/or cytotoxicity is in cancers that express a moderate level of an antigen targeted by the antibody portion of the ADC (e.g., moderate FRA expression). In some embodiments, the cleavable peptide portion can be cleaved by an enzyme, and the linker is a linker that the enzyme can cleave. In some embodiments, the enzyme is a cathepsin, and the linker is a linker that the cathepsin can cleave. In certain embodiments, compared to alternative cleavage mechanisms, linkers that the enzyme can cleave (e.g., linkers that the cathepsin can cleave) show one or more of the above-mentioned improved properties.
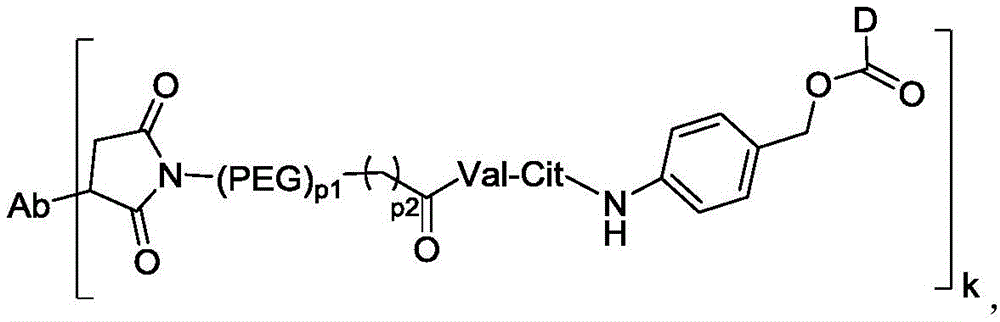
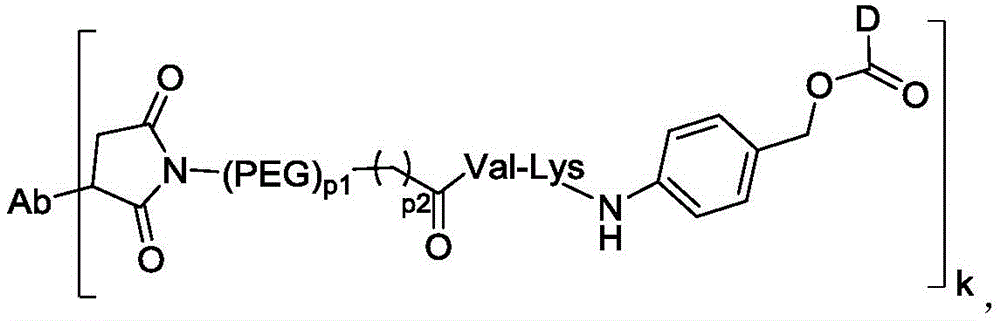
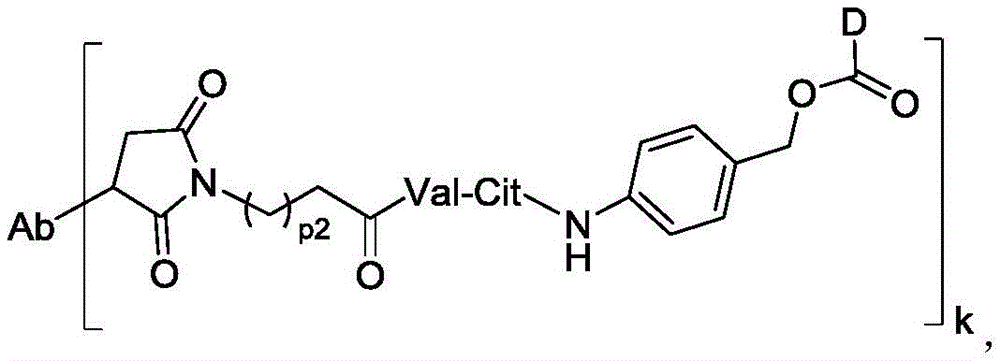
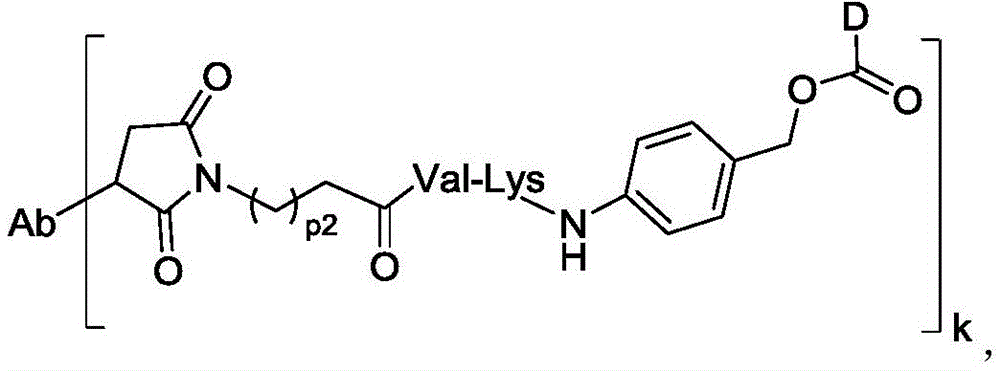
在一些实施方案中,连接子包含氨基酸单元,所述氨基酸单元优选包含由2至7个选自苯丙氨酸、甘氨酸、缬氨酸、赖氨酸、瓜氨酸、丝氨酸、谷氨酸、天冬氨酸的氨基酸构成的肽残基,更优选缬氨酸-瓜氨酸(Val-Cit)、丙氨酸-丙氨酸-天冬酰胺(Ala-Ala-Asn)、甘氨酸-甘氨酸-赖氨酸(Gly-Gly-lys)、缬氨酸-赖氨酸(Val-lys)、缬氨酸-丙氨酸(Val-Ala)、缬氨酸-苯丙氨酸(Val-Phe)或甘氨酸-甘氨酸-苯丙氨酸-甘氨酸(Gly-Gly-Phe-Gly)。In some embodiments, the linker comprises an amino acid unit, which preferably comprises a peptide residue consisting of 2 to 7 amino acids selected from phenylalanine, glycine, valine, lysine, citrulline, serine, glutamic acid, and aspartic acid, more preferably valine-citrulline (Val-Cit), alanine-alanine-asparagine (Ala-Ala-Asn), glycine-glycine-lysine (Gly-Gly-lys), valine-lysine (Val-lys), valine-alanine (Val-Ala), valine-phenylalanine (Val-Phe) or glycine-glycine-phenylalanine-glycine (Gly-Gly-Phe-Gly).
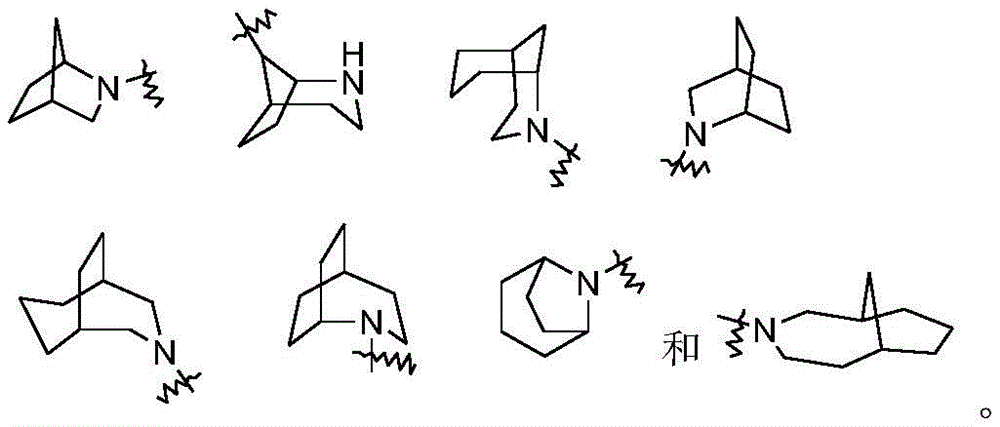
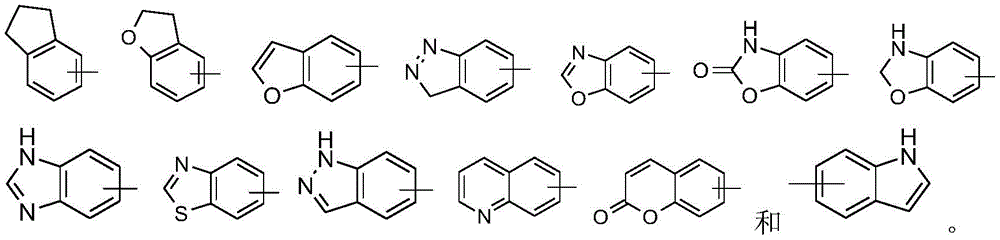
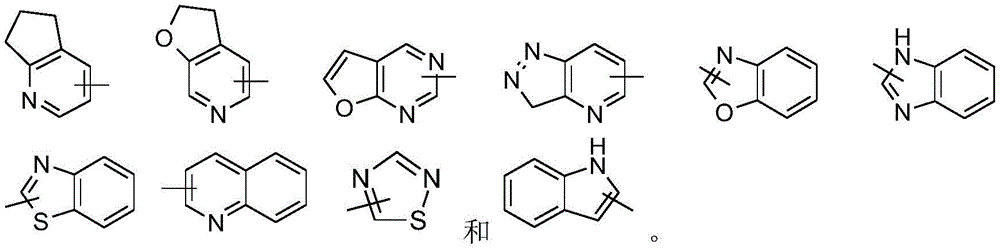
在一些实施方案中,本公开包括氨基酸单元的连接子选自:In some embodiments, the disclosure includes a linker of an amino acid unit selected from the group consisting of:
在一些实施方案中,氨基酸单元包含缬氨酸-瓜氨酸(Val-Cit)。在一些实施方案中,相对于包含替代氨基酸单元或替代可裂解部分的ADC,包含Val-Cit的ADC显示增加的稳定性,减少的脱靶细胞杀死,增加的靶向细胞杀死,较低的聚集水平,和/或较高的药物负载。In some embodiments, the amino acid unit comprises valine-citrulline (Val-Cit). In some embodiments, the ADC comprising Val-Cit exhibits increased stability, reduced off-target cell killing, increased on-target cell killing, lower aggregation levels, and/or higher drug loading relative to ADCs comprising alternative amino acid units or alternative cleavable moieties.
另一方面,一些实施方案提供的连接子包含可裂解磺酰胺部分,所述连接子在还原条件下能够裂解。In another aspect, some embodiments provide a linker comprising a cleavable sulfonamide moiety, wherein the linker is capable of being cleaved under reducing conditions.
在一些实施方案中,所述连接子包含可裂解二硫化物部分,所述连接子在还原条件下能够裂解。In some embodiments, the linker comprises a cleavable disulfide moiety, and the linker is capable of being cleaved under reducing conditions.
另一方面,本公开抗体偶联物中连接子包含至少一种将艾日布林衍生物D附接于可裂解部分的间隔单元。在一些实施方案中,所述连接子包含附接于D的间隔单元。In another aspect, in the antibody conjugates of the present disclosure, the linker comprises at least one spacer unit that attaches the eribulin derivative D to the cleavable moiety. In some embodiments, the linker comprises a spacer unit attached to D.
在一些实施方案中,所述间隔单元包含对氨基苯甲氧基羰基(pAB),In some embodiments, the spacer unit comprises p-aminobenzyloxycarbonyl (pAB),
在一些实施方案中,所述间隔单元包含:In some embodiments, the spacer unit comprises:
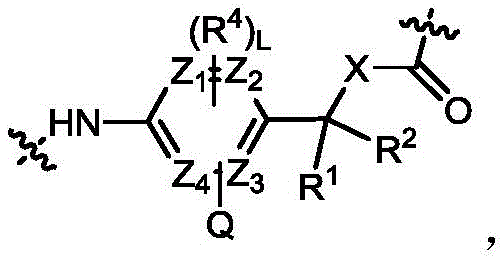
其中,Z1~Z4任选自碳原子或氮原子;R4选自烷基、环烷基、芳基和杂芳基,所述的烷基、环烷基、芳基和杂芳基各自独立地任选被选自烷基、烷氧基、卤素、氨基、氰基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基中的一个或多个取代基所取代;R1和R2各自独立选自氢、C1-6烷基、卤代烷基或C3-6环烷基,优选氢;或者,R1与R2与其相连接的碳原子一起形成C3-6环烷基;X选自-O-或-NH-;L选自1-4之间整数;Wherein, Z 1 to Z 4 are selected from carbon atoms or nitrogen atoms; R 4 is selected from alkyl, cycloalkyl, aryl and heteroaryl, and the alkyl, cycloalkyl, aryl and heteroaryl are each independently substituted by one or more substituents selected from alkyl, alkoxy, halogen, amino, cyano, nitro, hydroxyl, hydroxyalkyl, cycloalkyl, heterocyclyl, aryl and heteroaryl; R 1 and R 2 are each independently selected from hydrogen, C 1-6 alkyl, haloalkyl or C 3-6 cycloalkyl, preferably hydrogen; or, R 1 and R 2 together with the carbon atom to which they are connected form a C 3-6 cycloalkyl; X is selected from -O- or -NH-; L is selected from an integer between 1 and 4 ;
Q为V-E,V-E提供了可被位于胞内的糖苷酶切割的糖苷键,E选自-O-、-S-或-NR3-,R3选自氢或甲基,进一步地,V选自其中R5选自-COOH或CH2OH。在一些实施方案中,V选自-COOH。Q is VE, VE provides a glycosidic bond that can be cleaved by intracellular glycosidases, E is selected from -O-, -S- or -NR 3 -, R 3 is selected from hydrogen or methyl, and further, V is selected from wherein R 5 is selected from -COOH or CH 2 OH. In some embodiments, V is selected from -COOH.
在一些实施方案中,所述间隔单元包含:In some embodiments, the spacer unit comprises:
另一方面,本公开抗体偶联物(ADC)中L-D是由下式表示的化学部分:In another aspect, in the antibody conjugate (ADC) of the present disclosure, L-D is a chemical moiety represented by the following formula:
-Str-(Pep)-Sp-D-Str-(Pep)-Sp-D
Str是与Ab共价连接的伸展基单元,Str is a stretching unit covalently linked to Ab.
Sp为间隔单元,Sp is a spacer unit,
Pep选自氨基酸单元。Pep is selected from amino acid units.
另一方面,ADC中Str选自下式表示的化学部分:In another aspect, the ADC wherein Str is selected from a chemical moiety represented by the following formula:
其中R6选自-W-C(O)-、-C(O)-W-C(O)-、(CH2CH2O)p1C(O)-、(CH2CH2O)p1CH2C(O)-、(CH2CH2O)p1CH2CH2C(O)-,其中W选自C1-8亚烷基、C1-8亚烷基-环烷基或1至8个原子的直链杂亚烷基,所述杂亚烷基包含1至3个选自N、O或S的杂原子,其中所述的C1-8亚烷基、C1-8亚烷基-环烷基和直链杂亚烷基各自独立地任选进一步被选自卤素、羟基、氰基、氨基、烷基、氯代烷基、烷氧基和环烷基的一个或多个取代基所取代; wherein R 6 is selected from -WC(O)-, -C(O)-WC(O)-, (CH 2 CH 2 O) p1 C(O)-, (CH 2 CH 2 O) p1 CH 2 C(O)-, (CH 2 CH 2 O) p1 CH 2 CH 2 C(O)-, wherein W is selected from C 1-8 alkylene, C 1-8 alkylene-cycloalkyl or a straight-chain heteroalkylene of 1 to 8 atoms, wherein the heteroalkylene contains 1 to 3 heteroatoms selected from N, O or S, wherein the C 1-8 alkylene, C 1-8 alkylene-cycloalkyl and straight-chain heteroalkylene are each independently optionally further substituted with one or more substituents selected from halogen, hydroxy, cyano, amino, alkyl, chloroalkyl, alkoxy and cycloalkyl;
L1选自-NR7(CH2CH2O)p1CH2CH2C(O)-、-NR7(CH2CH2O)p1CH2C(O)-、-S(CH2)p1C(O)-、-(CH2)p1C(O)-或化学键,其中p1为1至20的整数,优选化学键;p1为1至20的整数,R7选自氢原子、烷基、卤代烷基和羟烷基。L 1 is selected from -NR 7 (CH 2 CH 2 O) p1 CH 2 CH 2 C(O)-, -NR 7 (CH 2 CH 2 O) p1 CH 2 C(O)-, -S(CH 2 ) p1 C(O)-, -(CH 2 ) p1 C(O)- or a chemical bond, wherein p1 is an integer from 1 to 20, preferably a chemical bond; p1 is an integer from 1 to 20, and R 7 is selected from a hydrogen atom, an alkyl group, a halogenated alkyl group and a hydroxyalkyl group.
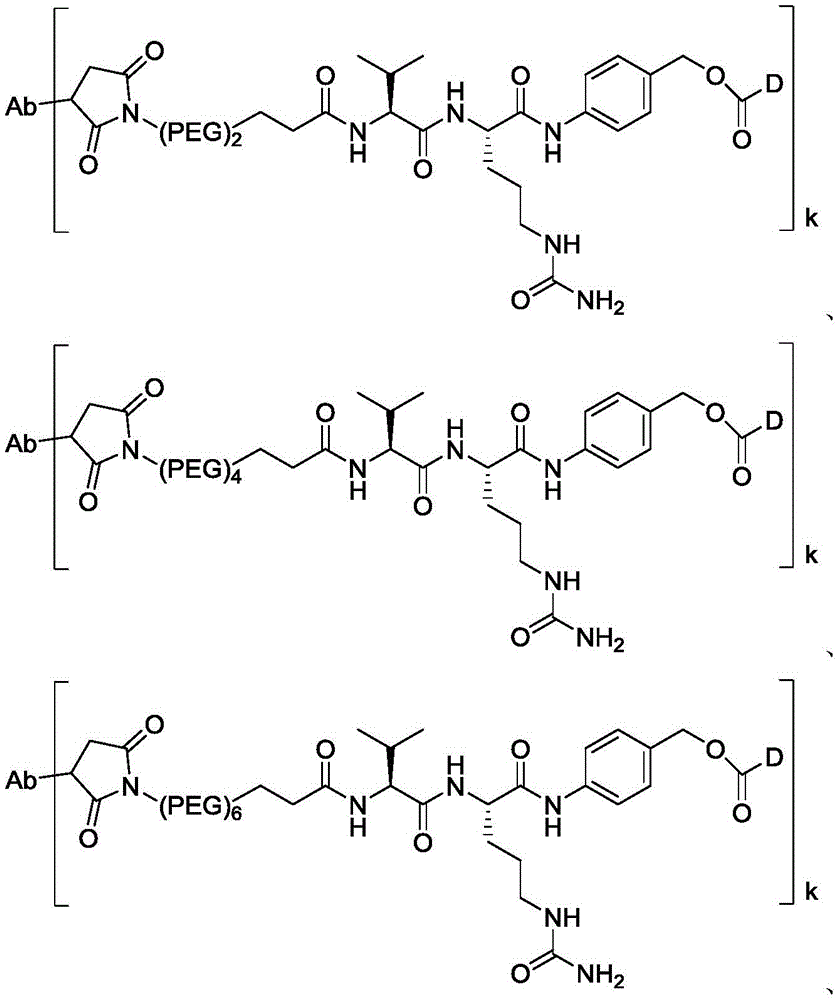
在一些实施方案中,连接子可以包含至少一种聚乙二醇(PEG)部分。PEG部分可以例如包含-(PEG)p1-,其中p1为整数1至20,例如(PEG)2;(PEG)4;In some embodiments, the linker can include at least one polyethylene glycol (PEG) moiety. The PEG moiety can, for example, include -(PEG) p1 -, Where p1 is an integer from 1 to 20, for example (PEG) 2 ; (PEG) 4 ;
(PEG)5。 (PEG) 5 .
在一些实施方案中,连接子中的间隔子单元包含(PEG)2。在一些实施方案中,尽管连接子长度较短,但相对于包含较长间隔子单元(例如(PEG)8)的ADC,包含较短间隔子单元(例如(PEG)2)的ADC显示较低的聚集水平和/或较高的药物负载。In some embodiments, the spacer unit in the linker comprises (PEG) 2. In some embodiments, despite the shorter linker length, ADCs comprising shorter spacer units (e.g., (PEG) 2 ) exhibit lower aggregation levels and/or higher drug loading relative to ADCs comprising longer spacer units (e.g., (PEG) 8 ).
在一些实施方案中,所述抗体-药物偶联物中R7选自C1-6亚烷基C(O)-、-(CH2-CH2O)2C(O)-、-(CH2-CH2O)2CH2C(O)-、-(CH2-CH2O)2CH2CH2C(O)-、-(CH2-CH2O)3C(O)-和-(CH2-CH2O)4C(O)-。In some embodiments, R7 in the antibody-drug conjugate is selected from C1-6 alkylene C(O)-, -( CH2 - CH2O ) 2C (O)-, -(CH2-CH2O) 2CH2C ( O )-, -( CH2 -CH2O) 2CH2CH2C (O)- , - (CH2-CH2O) 2CH2CH2C (O)-, -( CH2 - CH2O ) 3C (O ) -, and -( CH2 - CH2O ) 4C (O)-.
在一些实施方案中,所述抗体-药物偶联物中连接子L包含:顺丁烯二酰亚胺-(PEG)2-Val-Cit、顺丁烯二酰亚胺-(PEG)6-Val-Cit、顺丁烯二酰亚胺-(PEG)8-Val-Cit、顺丁烯二酰亚胺-(PEG)4-CH2CH2C(O)-Val-lys、顺丁烯二酰亚胺-(CH2)5-Val-Cit、顺丁烯二酰亚胺-(CH2)5-Val-lys、顺丁烯二酰亚胺-(CH2)5-Gly-Gly-Phe-Gly、顺丁烯二酰亚胺-(PEG)2-Ala-Ala-Asn、顺丁烯二酰亚胺-(PEG)6-Ala-Ala-Asn、顺丁烯二酰亚胺-(PEG)8-Ala-Ala-Asn、顺丁烯二酰亚胺-(PEG)4-三唑-(PEG)3-磺酰胺、顺丁烯二酰亚胺-(PEG)2-CH2CH2C(O)-Val-lys、顺丁烯二酰亚胺-(PEG)4-三唑-(PEG)3-磺酰胺或Mal-(PEG)4-三唑-(PEG)3-二硫化物。In some embodiments, the linker L in the antibody-drug conjugate comprises: maleimide-(PEG) 2 -Val-Cit, maleimide-(PEG) 6 -Val-Cit, maleimide-(PEG) 8 -Val-Cit, maleimide-(PEG) 4 -CH 2 CH 2 C(O)-Val-lys, maleimide-(CH 2 ) 5 -Val-Cit, maleimide-(CH 2 ) 5 -Val-lys, maleimide-(CH 2 ) 5 -Gly-Gly-Phe-Gly, maleimide-(PEG) 2 -Ala-Ala-Asn, maleimide-(PEG) 6 -Ala-Ala-Asn, maleimide-(PEG) 8 -Val-Cit, -Ala-Ala-Asn, maleimide-(PEG) 4 -triazole-(PEG) 3 -sulfonamide, maleimide-(PEG) 2- CH 2 CH 2 C(O)-Val-lys, maleimide-(PEG) 4 -triazole-(PEG) 3 -sulfonamide or Mal-(PEG) 4 -triazole-(PEG) 3 -disulfide.
另一方面,一些实施方案提供所述抗体-药物偶联物中Str选自下式表示的化学部分:On the other hand, some embodiments provide the antibody-drug conjugate wherein Str is selected from a chemical moiety represented by the following formula:
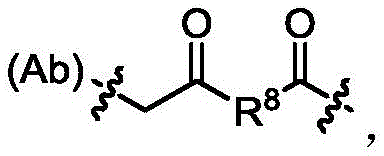
其中R8选自C1-10亚烷基、C2-10烯基、(C1-10亚烷基)O-、N(Rd)-(C2-6亚烷基)-N(Rd)和N(Rd)-(C2-6亚烷基);且每个Rd独立为H或C1-C6烷基。 wherein R 8 is selected from C 1-10 alkylene, C 2-10 alkenyl, (C 1-10 alkylene)O—, N(R d )-(C 2-6 alkylene)-N(R d ) and N(R d )-(C 2-6 alkylene); and each R d is independently H or C 1 -C 6 alkyl.
在一些实施方案中,所述抗体-药物偶联物,其由下式表示:In some embodiments, the antibody-drug conjugate is represented by the following formula:
k选自1至10,可以为整数,也可以为小数;P1选自2、4、6或8;P2选自0、1或2; k is selected from 1 to 10, and can be an integer or a decimal; P1 is selected from 2, 4, 6 or 8; P2 is selected from 0, 1 or 2;
k选自1至10,可以为整数,也可以为小数;P1选自2、4、6或8;P2选自0、1或2; k is selected from 1 to 10, and can be an integer or a decimal; P1 is selected from 2, 4, 6 or 8; P2 is selected from 0, 1 or 2;
k选自1至10,可以为整数,也可以为小数;P1选自2、4、6或8;P2选自0、1或2; k is selected from 1 to 10, and can be an integer or a decimal; P1 is selected from 2, 4, 6 or 8; P2 is selected from 0, 1 or 2;
k选自1至10,可以为整数,也可以为小数;P2选自1-6之间整数; k is selected from 1 to 10, and can be an integer or a decimal; P2 is selected from an integer between 1 and 6;
k选自1至10,可以为整数,也可以为小数;P2选自1-6之间整数; k is selected from 1 to 10, and can be an integer or a decimal; P2 is selected from an integer between 1 and 6;
k选自1至10,可以为整数,也可以为小数;P2选自1-6之间整数。 k is selected from 1 to 10 and can be an integer or a decimal; P2 is selected from an integer between 1 and 6.
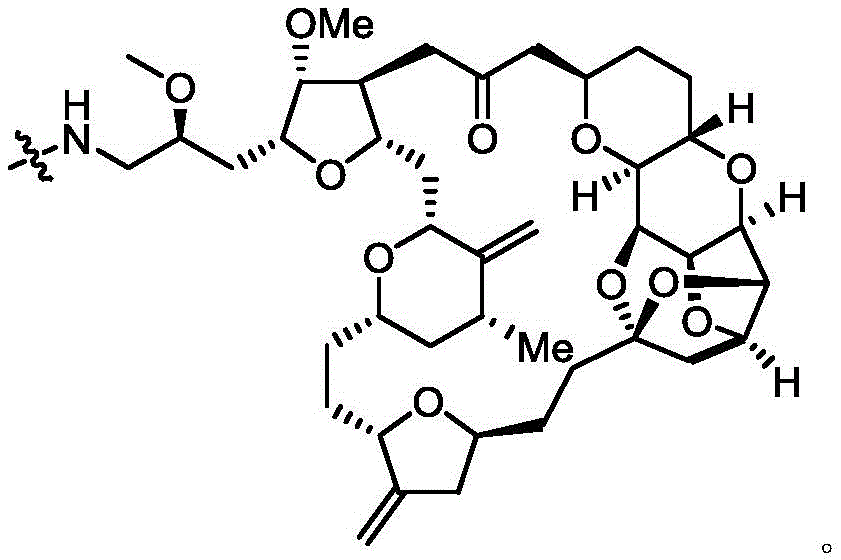
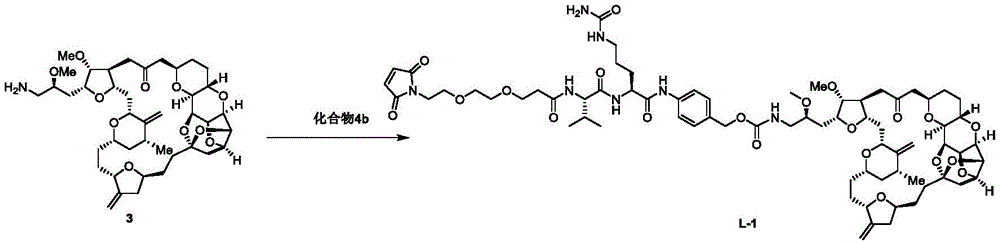
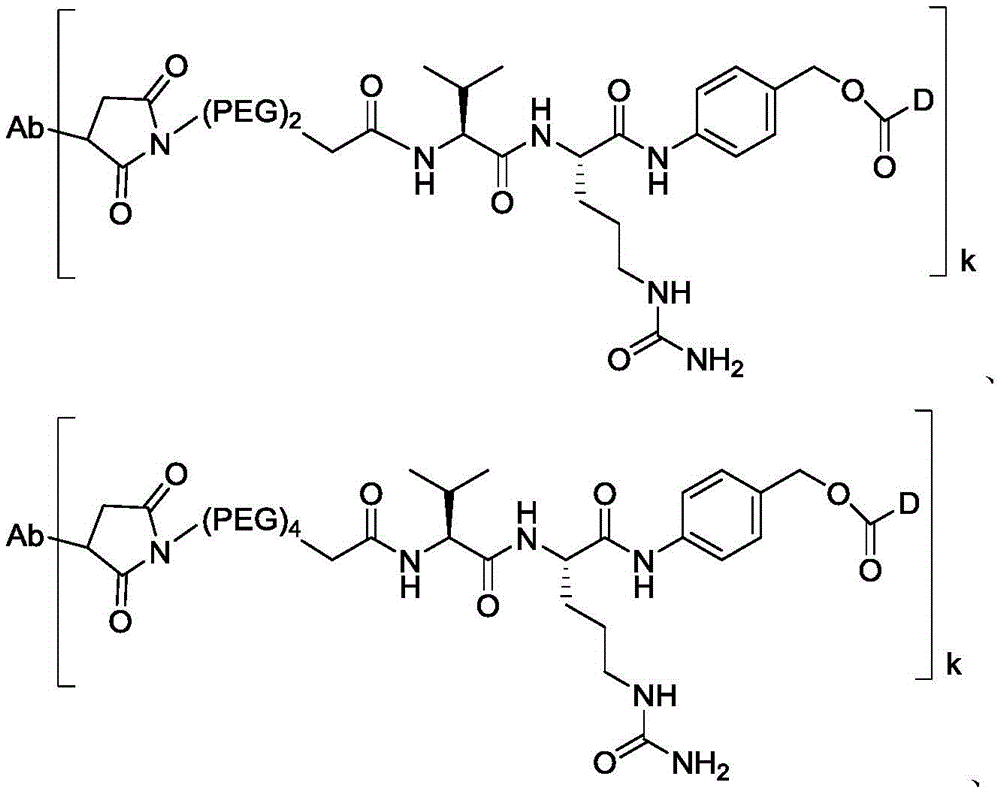
进一步地,本公开所述抗体-药物偶联物(ADC)选自:Further, the antibody-drug conjugate (ADC) of the present disclosure is selected from:
其中,k选自1至10,可以为整数,也可以为小数。 Wherein, k is selected from 1 to 10 and can be an integer or a decimal.
在一些实施方案中,所述抗体或其抗原结合片段为抗叶酸受体α(FRA)抗体抗体或其抗原结合片段。In some embodiments, the antibody or antigen-binding fragment thereof is an anti-folate receptor alpha (FRA) antibody or antigen-binding fragment thereof.
在一些实施方案中,所述抗FRA抗体或其抗原结合片段包含:与如SEQ ID NO:7所示的重链可变区具有相同序列的HCDR1、HCDR2和HCDR3,和与如SEQ ID NO:8所示的轻链可变区具有相同序列的LCDR1、LCDR2和LCDR3。In some embodiments, the anti-FRA antibody or antigen-binding fragment thereof comprises: HCDR1, HCDR2 and HCDR3 having the same sequence as the heavy chain variable region shown in SEQ ID NO:7, and LCDR1, LCDR2 and LCDR3 having the same sequence as the light chain variable region shown in SEQ ID NO:8.
在一些实施方案中,所述抗FRA抗体或其抗原结合片段包含:如SEQ ID NO:1、SEQID NO:2和SEQ ID NO:3所示的HCDR1、HCDR2和HCDR3;和如SEQ ID NO:4、SEQ ID NO:5和SEQID NO:6所示的LCDR1、LCDR2和LCDR3。In some embodiments, the anti-FRA antibody or antigen-binding fragment thereof comprises: HCDR1, HCDR2, and HCDR3 as shown in SEQ ID NO:1, SEQ ID NO:2, and SEQ ID NO:3; and LCDR1, LCDR2, and LCDR3 as shown in SEQ ID NO:4, SEQ ID NO:5, and SEQ ID NO:6.
其中,按照Kabat编码的抗FRA抗体或其抗原结合片段的各CDR序列如下表所示:The CDR sequences of the anti-FRA antibody or antigen-binding fragment thereof encoded according to Kabat are shown in the following table:
在一些实施方案中,所述抗叶酸受体α(FRA)抗体为人源化抗体或其片段。In some embodiments, the anti-folate receptor alpha (FRA) antibody is a humanized antibody or a fragment thereof.
在可选实施方案中,本公开中所述抗叶酸受体α(FRA)抗体或其抗原结合片段选自由Fab,Fab’-SH,Fv,scFv,和(Fab’)2片段组成的组的抗体片段。在一些实施方案中,内化抗体或内化抗原结合片段包含人类IgG1重链恒定域和Igκ轻链恒定域。In an alternative embodiment, the anti-folate receptor alpha (FRA) antibody or antigen-binding fragment thereof described in the present disclosure is an antibody fragment selected from the group consisting of Fab, Fab'-SH, Fv, scFv, and (Fab')2 fragments. In some embodiments, the internalizing antibody or internalizing antigen-binding fragment comprises a human IgG1 heavy chain constant domain and an Igκ light chain constant domain.
进一步地,在一些实施方案中,所述抗叶酸受体α(FRA)抗体或其抗原结合片段包含SEQ ID NO:7的氨基酸序列的重链可变区和包含SEQ ID NO:8的氨基酸序列的轻链可变区。Further, in some embodiments, the anti-folate receptor alpha (FRA) antibody or antigen-binding fragment thereof comprises a heavy chain variable region comprising the amino acid sequence of SEQ ID NO:7 and a light chain variable region comprising the amino acid sequence of SEQ ID NO:8.
重链可变区:Heavy chain variable region:
EVQLVESGGGVVQPGRSLRLSCSASGFTFSGYGLSWVRQAPGKGLEWVAMISSGGSYTYYADSVKGRFAISRDNAKNTLFLQMDSLRPEDTGVYFCARHGDDPAWFAYWGQGTPVTVSSEVQLVESGGGVVQPGRSLRLSCSASGFTFSGYGLSWVRQAPGKGLEWVAMISSGGSYTYYADSVKGRFAISRDNAKNTLFLQMDSLRPEDTGVYFCARHGDDPAWFAYWGQGTPVTVSS
SEQID NO:7SEQ ID NO: 7
轻链可变区:Light chain variable region:
DIQLTQSPSSLSASVGDRVTITCSVSSSISSNNLHWYQQKPGKAPKPWIYGTSNLASGVPSRFSGSGSGTDYTFTISSLQPEDIATYYCQQWSSYPYMYTFGQGTKVEIKDIQLTQSPSSSLSASVGDRVTITCSVSSSISSNNLHWYQQKPGKAPKPWIYGTSNLASGVPSRFSGSGSGTDYTFTISSLQPEDIATYYCQQWSSYPYMYTFGQGTKVEIK
SEQID NO:8SEQ ID NO: 8
在本公开实施方案中,所述抗叶酸受体α(FRA)抗体包含SEQ ID NO:9的氨基酸序列的重链和包含SEQ ID NO:10的氨基酸序列的轻链。In an embodiment of the present disclosure, the anti-folate receptor alpha (FRA) antibody comprises a heavy chain having an amino acid sequence of SEQ ID NO:9 and a light chain having an amino acid sequence of SEQ ID NO:10.
重链:Heavy Chain:
EVQLVESGGGVVQPGRSLRLSCSASGFTFSGYGLSWVRQAPGKGLEWVAMISSGGSYTYYADSVKGRFAISRDNAKNTLFLQMDSLRPEDTGVYFCARHGDDPAWFAYWGQGTPVTVSSASTKGPSVFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKALPAPIEKTISKAKGQPREPQVYTLPPSRDELTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGKEVQLVESGGGVVQPGRSLRLSCSASGFTFSGYGLSWVRQAPGKGLEWVAMISSGGSYTYYADSVKGRFAISRDNAKNTLFLQMDSLRPEDTGVYFCARHGDDPAWFAYWGQGTPVTVSSASTKGPSVFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQTYICNVNHKPSNTKVD KKVEPKSCDK THTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVLTVLHQDWLNGKEYKCKVSNKALPAPIEKTISKAKGQPREPQVYTLPPSRDELTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDGSFFLYSKLTVDKSRWQ QGNVFSCSVMHEALHNHYTQKSLSLSPGK
SEQID NO:9SEQ ID NO: 9
轻链:Light chain:
DIQLTQSPSSLSASVGDRVTITCSVSSSISSNNLHWYQQKPGKAPKPWIYGTSNLASGVPSRFSGSGSGTDYTFTISSLQPEDIATYYCQQWSSYPYMYTFGQGTKVEIKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFYPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSLSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFNRGECDIQLTQSPSSSLSASVGDRVTITCSVSSSISSNNLHWYQQKPGKAPKPWIYGTSNLASGVPSRFSGSGSGTDYTFTISSLQPEDIATYYCQQWSSYPYMYTFGQGTKVEIKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFYPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSLSSTLTLSKADYEKHKVYACEVTHQGLS SPVTKSFNRGEC
SEQID NO:10SEQ ID NO: 10
进一步地,前述抗体-偶联物采用异质化连接技术,即基于胺的赖氨酸结合(amine-based lysine conjugation)和基于巯基的半胱氨酸结合(thiol-based cysteineconjugation)。在一些实施方案中,前述抗体-偶联物中与Ab共价连接的伸展基单元Str与如抗叶酸受体α(FRA)抗体上半胱氨基酸相连接。Further, the aforementioned antibody-conjugate uses heterogeneous connection technology, i.e., amine-based lysine conjugation and thiol-based cysteine conjugation. In some embodiments, the stretching unit Str covalently linked to Ab in the aforementioned antibody-conjugate is linked to a cysteine amino acid on an anti-folate receptor α (FRA) antibody.
另一方面,前述抗体-偶联物可采用位点特异性(site-specific,homogeneousconjugation chemistries)的结合方法可以有效地控制DAR值,获得ADC稳定性和聚集性均较佳,在一些实施方案中,前述抗体-偶联物中与Ab共价连接的伸展基单元Str与如抗叶酸受体α(FRA)抗体上On the other hand, the aforementioned antibody-conjugate can be conjugated by site-specific (homogeneous conjugation chemistries) to effectively control the DAR value, thereby obtaining ADC with better stability and aggregation. In some embodiments, the stretching unit Str covalently linked to Ab in the aforementioned antibody-conjugate is linked to, for example, an anti-folate receptor α (FRA) antibody.
如同质(homogeneous)DAR值为2的ADC比随机结合方法得到平均DAR值为3.4-4的ADC的疏水性和聚集效应均低,但TI(therapeuticindex,治疗指数)可能有限(如PBD和Aur0101)。ADCs with homogeneous DAR values of 2 have lower hydrophobicity and aggregation effects than ADCs with average DAR values of 3.4-4 obtained by random binding methods, but the TI (therapeutic index) may be limited (such as PBD and Aur0101).
在一些实施方案中,本公开抗体偶联物中k选自2.0~2.5,包括2.0、2.1、2.2、2.3、2.4、2.5或任意两数之间值。In some embodiments, k in the antibody conjugates of the present disclosure is selected from 2.0 to 2.5, including 2.0, 2.1, 2.2, 2.3, 2.4, 2.5 or any value between two numbers.
在另一些实施方案中,本公开抗体偶联物中k选自2.5~3.5,包括2.5、2.6、2.7、2.8、2.9、3.0、3.1、3.2、3.3、3.4、3.6、3.7、3.8、3.9、4.0或任意两数之间值。In other embodiments, k in the antibody conjugates of the present disclosure is selected from 2.5 to 3.5, including 2.5, 2.6, 2.7, 2.8, 2.9, 3.0, 3.1, 3.2, 3.3, 3.4, 3.6, 3.7, 3.8, 3.9, 4.0 or any value between two numbers.
在另一些实施方案中,与Ab共价连接的伸展基单元Str通过糖基酶催化与如抗叶酸受体α(FRA)抗体连接。In other embodiments, the stretcher unit Str covalently linked to Ab is linked to, for example, an anti-folate receptor alpha (FRA) antibody by glycosylase catalysis.
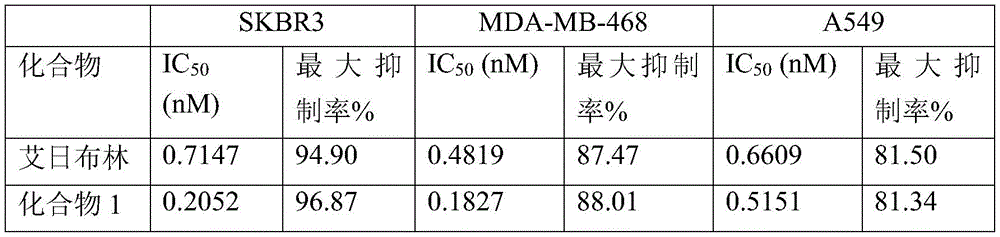
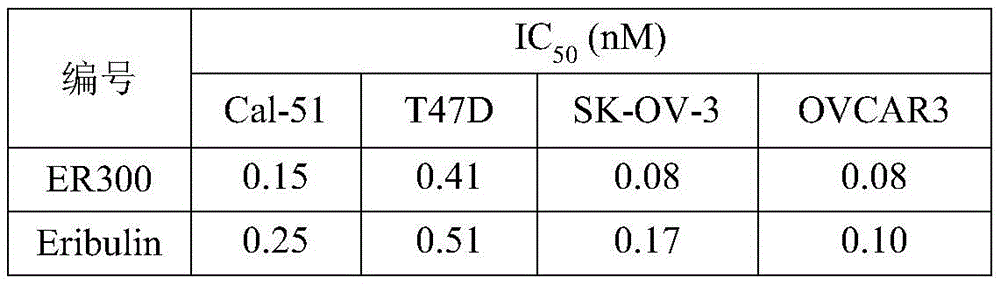
另一方面,本公开抗叶酸抗体偶联物对FOLR1阳性肿瘤细胞增殖具有较好的抑制作用。例如,在一些实施方案中,本公开抗体偶联物对KB(Cervix)细胞增殖抑制IC50在0.01至1000ng/mL。在一些实施方案中,本公开抗体偶联物对KB(Cervix)细胞增殖抑制IC50在0.01至500ng/mL。在一些实施方案中,本公开抗体偶联物对KB(Cervix)细胞增殖抑制IC50在<300ng/mL。在一些实施方案中,本公开抗体偶联物对KB(Cervix)细胞增殖抑制IC50在<200ng/mL。在一些实施方案中,本公开抗体偶联物对KB(Cervix)细胞增殖抑制IC50在<50ng/mL。On the other hand, the anti-folate antibody conjugates disclosed herein have a good inhibitory effect on the proliferation of FOLR1-positive tumor cells. For example, in some embodiments, the antibody conjugates disclosed herein inhibit KB (Cervix) cell proliferation with an IC 50 of 0.01 to 1000 ng/mL. In some embodiments, the antibody conjugates disclosed herein inhibit KB (Cervix) cell proliferation with an IC 50 of 0.01 to 500 ng/mL. In some embodiments, the antibody conjugates disclosed herein inhibit KB (Cervix) cell proliferation with an IC 50 of <300 ng/mL. In some embodiments, the antibody conjugates disclosed herein inhibit KB (Cervix) cell proliferation with an IC 50 of <200 ng/mL. In some embodiments, the antibody conjugates disclosed herein inhibit KB (Cervix) cell proliferation with an IC 50 of <50 ng/mL.
在另一些实施方案中,本公开抗体偶联物对SK-OV-3(Ovarian)细胞增殖抑制IC50在0.01至500ng/mL。在一些实施方案中,本公开抗体偶联物对SK-OV-3(Ovarian)细胞增殖抑制IC50在0.01至300ng/mL。在一些实施方案中,本公开抗体偶联物对SK-OV-3(Ovarian)细胞增殖抑制IC50在<200ng/mL。在一些实施方案中,本公开抗体偶联物对SK-OV-3(Ovarian)细胞增殖抑制IC50在<100ng/mL。在一些实施方案中,本公开抗体偶联物对SK-OV-3(Ovarian)细胞增殖抑制IC50在<50ng/mL。In other embodiments, the antibody conjugates of the present disclosure inhibit SK-OV-3 (Ovarian) cell proliferation at an IC 50 of 0.01 to 500 ng/mL. In some embodiments, the antibody conjugates of the present disclosure inhibit SK-OV-3 (Ovarian) cell proliferation at an IC 50 of 0.01 to 300 ng/mL. In some embodiments, the antibody conjugates of the present disclosure inhibit SK-OV-3 (Ovarian) cell proliferation at an IC 50 of <200 ng/mL. In some embodiments, the antibody conjugates of the present disclosure inhibit SK-OV-3 (Ovarian) cell proliferation at an IC 50 of <100 ng/mL. In some embodiments, the antibody conjugates of the present disclosure inhibit SK-OV-3 (Ovarian) cell proliferation at an IC 50 of <50 ng/mL.
在另一些实施方案中,本公开抗体偶联物对T47D细胞增殖抑制IC50在0.01至500ng/mL。在一些实施方案中,本公开抗体偶联物对T47D细胞增殖抑制IC50在0.01至300ng/mL。在一些实施方案中,本公开抗体偶联物对T47D细胞增殖抑制IC50在<200ng/mL。在一些实施方案中,本公开抗体偶联物对T47D细胞增殖抑制IC50在<100ng/mL。在一些实施方案中,本公开抗体偶联物对T47D细胞增殖抑制IC50在<50ng/mL。In other embodiments, the antibody conjugates disclosed herein inhibit T47D cell proliferation at an IC50 of 0.01 to 500 ng/mL. In some embodiments, the antibody conjugates disclosed herein inhibit T47D cell proliferation at an IC50 of 0.01 to 300 ng/mL. In some embodiments, the antibody conjugates disclosed herein inhibit T47D cell proliferation at an IC50 of <200 ng/mL. In some embodiments, the antibody conjugates disclosed herein inhibit T47D cell proliferation at an IC50 of <100 ng/mL. In some embodiments, the antibody conjugates disclosed herein inhibit T47D cell proliferation at an IC50 of <50 ng/mL.
另一方面,本公开还提供前述抗体-偶联物的同位素取代物。在一些实施方案中,所述同位素取代物为氘原子取代的。On the other hand, the present disclosure also provides isotope substitutions of the aforementioned antibody-conjugates. In some embodiments, the isotope substitutions are substituted with deuterium atoms.
另一方面,本公开还提供一种药物组合物,其含有治疗有效量的前述抗体-药物偶联物,或其同位素取代物,以及药学上可接受的载体、稀释剂或赋形剂。On the other hand, the present disclosure also provides a pharmaceutical composition, which contains a therapeutically effective amount of the aforementioned antibody-drug conjugate, or an isotope substituted product thereof, and a pharmaceutically acceptable carrier, diluent or excipient.
在一些实施方案中,所述的药物组合物的单位剂量为0.001mg-1000mg。In some embodiments, the unit dose of the pharmaceutical composition is 0.001 mg-1000 mg.
在某些实施方案中,基于组合物的总重量,所述的药物组合物含有0.01-99.99%的前述抗体-药物偶联物,或其同位素取代物。在某些实施方案中,所述的药物组合物含有0.1-99.9%的前述抗体-药物偶联物,或其同位素取代物。在某些实施方案中,所述的药物组合物含有0.5%-99.5%的前述抗体-药物偶联物,或其同位素取代物。在某些实施方案中,所述的药物组合物含有1%-99%的前述抗体-药物偶联物,或其同位素取代物。在某些实施方案中,所述的药物组合物含有2%-98%的前述抗体-药物偶联物,或其同位素取代物。In certain embodiments, the pharmaceutical composition contains 0.01-99.99% of the aforementioned antibody-drug conjugate, or its isotope substitution, based on the total weight of the composition. In certain embodiments, the pharmaceutical composition contains 0.1-99.9% of the aforementioned antibody-drug conjugate, or its isotope substitution. In certain embodiments, the pharmaceutical composition contains 0.5%-99.5% of the aforementioned antibody-drug conjugate, or its isotope substitution. In certain embodiments, the pharmaceutical composition contains 1%-99% of the aforementioned antibody-drug conjugate, or its isotope substitution. In certain embodiments, the pharmaceutical composition contains 2%-98% of the aforementioned antibody-drug conjugate, or its isotope substitution.
在某些实施方案中,基于组合物的总重量,所述的药物组合物含有0.01%-99.99%的药学上可接受的赋形剂。在某些实施方案中,所述的药物组合物含有0.1%-99.9%的药学上可接受的赋形剂。在某些实施方案中,所述的药物组合物含有0.5%-99.5%的药学上可接受的赋形剂。在某些实施方案中,所述的药物组合物含有1%-99%的药学上可接受的赋形剂。在某些实施方案中,所述的药物组合物含有2%-98%的药学上可接受的赋形剂。In certain embodiments, the pharmaceutical composition contains 0.01%-99.99% of a pharmaceutically acceptable excipient based on the total weight of the composition. In certain embodiments, the pharmaceutical composition contains 0.1%-99.9% of a pharmaceutically acceptable excipient. In certain embodiments, the pharmaceutical composition contains 0.5%-99.5% of a pharmaceutically acceptable excipient. In certain embodiments, the pharmaceutical composition contains 1%-99% of a pharmaceutically acceptable excipient. In certain embodiments, the pharmaceutical composition contains 2%-98% of a pharmaceutically acceptable excipient.
本公开还提供一种前述抗体-药物偶联物或前述药物组合物在制备用于治疗或预防肿瘤的药物中的用途。在一些实施方案中,所述的肿瘤为与叶酸受体α表达相关的。The present disclosure also provides a use of the aforementioned antibody-drug conjugate or the aforementioned pharmaceutical composition in the preparation of a drug for treating or preventing a tumor. In some embodiments, the tumor is associated with the expression of folate receptor α.
本公开还提供一种前述抗体-药物偶联物或前述药物组合物在制备治疗和/或预防癌症的药物中的用途。在一些实施方案中,所述癌症选自乳腺癌、卵巢癌、宫颈癌、子宫癌、前列腺癌、肾癌、尿道癌、膀胱癌、肝癌、胃癌、子宫内膜癌、唾液腺癌、食道癌、黑色素瘤、神经胶质瘤、神经母细胞瘤、肉瘤、肺癌、结肠癌、直肠癌、结直肠癌、白血病、骨癌、皮肤癌、甲状腺癌、胰腺癌和淋巴瘤。The present disclosure also provides a use of the aforementioned antibody-drug conjugate or the aforementioned pharmaceutical composition in the preparation of a drug for treating and/or preventing cancer. In some embodiments, the cancer is selected from breast cancer, ovarian cancer, cervical cancer, uterine cancer, prostate cancer, kidney cancer, urethral cancer, bladder cancer, liver cancer, gastric cancer, endometrial cancer, salivary gland cancer, esophageal cancer, melanoma, glioma, neuroblastoma, sarcoma, lung cancer, colon cancer, rectal cancer, colorectal cancer, leukemia, bone cancer, skin cancer, thyroid cancer, pancreatic cancer and lymphoma.
本公开还提供一种治疗或预防与叶酸受体α表达相关的的癌症患者的方法,其通过向所述患者施用治疗有效量的如前述抗体-药物偶联物,或其同位素取代物,或前述药物组合物。The present disclosure also provides a method for treating or preventing cancer patients associated with folate receptor α expression, by administering to the patient a therapeutically effective amount of the aforementioned antibody-drug conjugate, or an isotope substituted product thereof, or the aforementioned pharmaceutical composition.
本公开还提供了用于治疗或预防与叶酸受体α表达相关的的癌症的前述抗体-药物偶联物,或其同位素取代物,或前述药物组合物。The present disclosure also provides the aforementioned antibody-drug conjugate, or an isotope substitution thereof, or the aforementioned pharmaceutical composition for treating or preventing cancer associated with folate receptor α expression.
可将活性化合物制成适合于通过任何适当途径给药的形式,活性化合物优选是以单位剂量的方式,或者是以患者可以以单剂自我给药的方式。本公开化合物或组合物的单位剂量的表达方式可以是片剂、胶囊、扁囊剂、瓶装药水、药粉、颗粒剂、锭剂、栓剂、再生药粉或液体制剂。The active compound may be formulated for administration by any appropriate route, preferably in a unit dosage form, or in a form in which a patient can self-administer a single dose. The unit dosage form of the disclosed compound or composition may be tablets, capsules, cachets, bottled liquids, powders, granules, lozenges, suppositories, reconstituted powders or liquid preparations.
发明的详细说明Detailed description of the invention
除非另有限定,本公开所用的所有技术和科学术语均与本公开所属领域普通技术人员的通常理解一致。虽然也可采用与本公开所述相似或等同的任何方法和材料实施或测试本公开,但本公开描述了优选的方法和材料。描述和要求保护本公开时,依据以下定义使用下列术语。Unless otherwise defined, all technical and scientific terms used in the present disclosure are consistent with the common understanding of those of ordinary skill in the art to which the present disclosure belongs. Although any methods and materials similar or equivalent to those described in the present disclosure may be used to implement or test the present disclosure, the present disclosure describes preferred methods and materials. When describing and claiming the present disclosure, the following terms are used in accordance with the following definitions.
当本公开中使用商品名时,申请人旨在包括该商品名产品的制剂、该商品名产品的非专利药和活性药物部分。When a trade name is used in this disclosure, Applicants intend to include formulations of the trade name product, generic drugs, and the active drug portion of the trade name product.
除非有相反陈述,在说明书和权利要求书中使用的术语具有下述含义。Unless stated otherwise, the terms used in the specification and claims have the following meanings.
本公开化合物可以存在特定的几何或立体异构体形式。本公开设想所有的这类化合物,包括顺式和反式异构体、(-)-和(+)-对对映体、(R)-和(S)-对映体、非对映异构体、(D)-异构体、(L)-异构体,及其外消旋混合物和其他混合物,例如对映异构体或非对映体富集的混合物,所有这些混合物都属于本公开的范围之内。烷基等取代基中可存在另外的不对称碳原子。所有这些异构体以及它们的混合物,均包括在本公开的范围之内。本公开的含有不对称碳原子的化合物可以以光学活性纯的形式或外消旋形式被分离出来。光学活性纯的形式可以从外消旋混合物拆分,或通过使用手性原料或手性试剂合成。The compounds of the present disclosure may exist in specific geometric or stereoisomeric forms. The present disclosure contemplates all such compounds, including cis and trans isomers, (-)- and (+)-enantiomers, (R)- and (S)-enantiomers, diastereomers, (D)-isomers, (L)-isomers, and racemic mixtures and other mixtures thereof, such as mixtures enriched in enantiomers or diastereomers, all of which are within the scope of the present disclosure. Additional asymmetric carbon atoms may be present in substituents such as alkyl. All of these isomers and their mixtures are included within the scope of the present disclosure. The compounds of the present disclosure containing asymmetric carbon atoms can be isolated in optically pure form or in racemic form. Optically pure forms can be resolved from racemic mixtures or synthesized by using chiral raw materials or chiral reagents.
可以通过的手性合成或手性试剂或者其他常规技术制备光学活性的(R)-和(S)-异构体以及D和L异构体。如果想得到本公开某化合物的一种对映体,可以通过不对称合成或者具有手性助剂的衍生作用来制备,其中将所得非对映体混合物分离,并且辅助基团裂开以提供纯的所需对映异构体。或者,当分子中含有碱性官能团(如氨基)或酸性官能团(如羧基)时,与适当的光学活性的酸或碱形成非对映异构体的盐,然后通过本领域所公知的常规方法进行非对映异构体拆分,然后回收得到纯的对映体。此外,对映异构体和非对映异构体的分离通常是通过使用色谱法完成的,所述色谱法采用手性固定相,并任选地与化学衍生法相结合(例如由胺生成氨基甲酸盐)。Optically active (R)- and (S)-isomers as well as D and L isomers can be prepared by chiral synthesis or chiral reagents or other conventional techniques. If one enantiomer of a compound of the present disclosure is desired, it can be prepared by asymmetric synthesis or derivatization with a chiral auxiliary, wherein the resulting diastereomeric mixture is separated and the auxiliary group is cleaved to provide the pure desired enantiomer. Alternatively, when the molecule contains a basic functional group (such as an amino group) or an acidic functional group (such as a carboxyl group), a diastereomeric salt is formed with an appropriate optically active acid or base, and then the diastereoisomers are separated by conventional methods known in the art, and then the pure enantiomer is recovered. In addition, the separation of enantiomers and diastereomers is usually accomplished by using chromatography, which uses a chiral stationary phase and is optionally combined with a chemical derivatization method (e.g., carbamates are generated from amines).
本公开所述化合物的化学结构中,键表示未指定构型,即如果化学结构中存在手性异构体,键可以为或或者同时包含和两种构型。键表示未指定构型,包括顺式(E)或反式(Z)构型。或者本公开所描述的是指双键,以该键键合的结构可为“顺式异构体”或“反式异构体”或者“顺式异构体和反式异构体以任何比例形成的混合物”,例如式E代表E-1、式E-2或两者以任何比例形成的混合物:In the chemical structures of the compounds disclosed herein, the bond Indicates that the configuration is not specified, that is, if there are chiral isomers in the chemical structure, the bond Can be or or include both and Two configurations. Key indicates an unspecified configuration, including cis (E) or trans (Z) configurations. Refers to a double bond, and the structure bonded by this bond can be a "cis isomer" or a "trans isomer" or a "mixture of cis isomers and trans isomers in any proportion". For example, Formula E represents E-1, Formula E-2 or a mixture of the two in any proportion:
本公开所述化合物的化学结构中,键并未指定构型,即可以为Z构型或E构型,或者同时包含两种构型。In the chemical structures of the compounds disclosed herein, the bond The configuration is not specified, that is, it can be Z configuration or E configuration, or contain both configurations.
本公开的化合物和中间体还可以以不同的互变异构体形式存在,并且所有这样的形式包含于本公开的范围内。术语“互变异构体”或“互变异构体形式”是指可经由低能垒互变的不同能量的结构异构体。例如,质子互变异构体(也称为质子转移互变异构体)包括经由质子迁移的互变,如酮-烯醇及亚胺-烯胺、内酰胺-内酰亚胺异构化。内酰胺-内酰亚胺平衡实例是在如下所示的A和B之间。Compounds and intermediates of the present disclosure may also exist in different tautomeric forms, and all such forms are included within the scope of the present disclosure. The term "tautomer" or "tautomeric form" refers to structural isomers of different energies that can interconvert via a low energy barrier. For example, proton tautomers (also referred to as prototransfer tautomers) include interconversion via proton migration, such as keto-enol and imine-enamine, lactam-lactim isomerization. Lactam-lactim equilibrium examples are between A and B as shown below.
本公开中的所有化合物可以被画成A型或B型。所有的互变异构形式在本公开的范围内。化合物的命名不排除任何互变异构体。All compounds in the present disclosure can be drawn as either Form A or Form B. All tautomeric forms are within the scope of the present disclosure. The naming of the compounds does not exclude any tautomers.
本公开还包括一些与本文中记载的那些相同的,但一个或多个原子被原子量或质量数不同于自然中通常发现的原子量或质量数的原子置换的同位素标记的本公开化合物。可结合到本公开化合物的同位素的实例包括氢、碳、氮、氧、磷、硫、氟、碘和氯的同位素,诸如分别为2H、3H、11C、13C、14C、13N、15N、15O、17O、18O、31P、32P、35S、18F、123I、125I和36Cl等。The present disclosure also includes isotopically labeled compounds of the present disclosure that are identical to those described herein, but in which one or more atoms are replaced by atoms having an atomic mass or mass number different from that normally found in nature. Examples of isotopes that can be incorporated into compounds of the present disclosure include isotopes of hydrogen, carbon, nitrogen, oxygen, phosphorus, sulfur, fluorine, iodine, and chlorine, such as 2 H, 3 H, 11 C, 13 C, 14 C, 13 N, 15 N, 15 O, 17 O, 18 O, 31 P, 32 P, 35 S, 18 F, 123 I, 125 I, and 36 Cl, respectively .
除另有说明,当一个位置被特别地指定为氘(D)时,该位置应理解为具有大于氘的天然丰度(其为0.015%)至少1000倍的丰度的氘(即,至少10%的氘掺入)。示例中化合物的具有大于氘的天然丰度可以是至少1000倍的丰度的氘、至少2000倍的丰度的氘、至少3000倍的丰度的氘、至少4000倍的丰度的氘、至少5000倍的丰度的氘、至少6000倍的丰度的氘或更高丰度的氘。本公开还包括各种氘化形式的式(I)化合物。与碳原子连接的各个可用的氢原子可独立地被氘原子替换。本领域技术人员能够参考相关文献合成氘化形式的式(I)化合物。在制备氘代形式的式(I)化合物时可使用市售的氘代起始物质,或它们可使用常规技术采用氘代试剂合成,氘代试剂包括但不限于氘代硼烷、三氘代硼烷四氢呋喃溶液、氘代氢化锂铝、氘代碘乙烷和氘代碘甲烷等。Unless otherwise specified, when a position is specifically designated as deuterium (D), the position is understood to have deuterium (i.e., at least 10% deuterium incorporation) at least 1000 times greater than the natural abundance of deuterium (which is 0.015%). The natural abundance of the compound in the example may be at least 1000 times greater than deuterium, at least 2000 times greater than deuterium, at least 3000 times greater than deuterium, at least 4000 times greater than deuterium, at least 5000 times greater than deuterium, at least 6000 times greater than deuterium or more. The present disclosure also includes various deuterated forms of the formula (I) compound. Each available hydrogen atom connected to a carbon atom may be independently replaced by a deuterium atom. Those skilled in the art can synthesize deuterated forms of the formula (I) compound with reference to the relevant literature. In preparing deuterated forms of compounds of formula (I), commercially available deuterated starting materials may be used, or they may be synthesized using conventional techniques using deuterated reagents, including but not limited to deuterated borane, trideuterated borane in tetrahydrofuran, deuterated lithium aluminum hydride, deuterated iodoethane and deuterated iodomethane, and the like.
“任选地”或“任选”是指意味着随后所描述的事件或环境可以但不必发生,该说明包括该事件或环境发生或不发生的场合。例如“任选的被卤素或者氰基取代的C1-6烷基”是指卤素或者氰基可以但不必须存在,该说明包括烷基被卤素或者氰基取代的情形和烷基不被卤素和氰基取代的情形。"Optionally" or "optionally" means that the subsequently described event or circumstance may but need not occur, and the description includes instances where the event or circumstance occurs or does not occur. For example, "C 1-6 alkyl optionally substituted with halogen or cyano" means that halogen or cyano may but need not be present, and the description includes instances where the alkyl is substituted with halogen or cyano and instances where the alkyl is not substituted with halogen and cyano.
“药物组合物”表示含有一种或多种本文所述化合物或其生理学上可药用盐或前体药物与其他化学组分的混合物,以及其他组分例如生理学可药用的载体和赋形剂。药物组合物的目的是促进对生物体的给药,利于活性成分的吸收进而发挥生物活性。"Pharmaceutical composition" means a mixture containing one or more compounds described herein or their physiologically pharmaceutically acceptable salts or prodrugs and other chemical components, as well as other components such as physiologically pharmaceutically acceptable carriers and excipients. The purpose of a pharmaceutical composition is to facilitate administration to an organism, facilitate the absorption of the active ingredients, and thus exert biological activity.
“可药用赋形剂”包括但不限于任何已经被美国食品和药物管理局批准对于人类或家畜动物使用可接受的任何助剂、载体、赋形剂、助流剂、甜味剂、稀释剂、防腐剂、染料/着色剂、增香剂、表面活性剂、润湿剂、分散剂、助悬剂、稳定剂、等渗剂、溶剂或乳化剂。"Pharmaceutically acceptable excipients" include, but are not limited to, any adjuvant, carrier, excipient, glidant, sweetener, diluent, preservative, dye/colorant, flavoring agent, surfactant, wetting agent, dispersant, suspending agent, stabilizer, isotonic agent, solvent or emulsifier approved by the U.S. Food and Drug Administration for use in humans or domestic animals.
本公开中所述“有效量”或“有效治疗量”包含足以改善或预防医学病症的症状或病症的量。有效量还意指足以允许或促进诊断的量。用于特定患者或兽医学受试者的有效量可依据以下因素而变化:如待治疗的病症、患者的总体健康情况、给药的方法途径和剂量以及副作用严重性。有效量可以是避免显著副作用或毒性作用的最大剂量或给药方案。As used herein, an "effective amount" or "therapeutically effective amount" includes an amount sufficient to improve or prevent the symptoms or symptoms of a medical condition. An effective amount also means an amount sufficient to allow or facilitate diagnosis. The effective amount for a particular patient or veterinary subject may vary depending on factors such as the condition to be treated, the patient's overall health, the method, route and dosage of administration, and the severity of side effects. An effective amount may be the maximum dose or dosing regimen that avoids significant side effects or toxic effects.
术语“药物”是指细胞毒性药物或免疫调节剂。细胞毒性药物能在肿瘤细胞内具有较强破坏其正常生长的化学分子。细胞毒性药物原则上在足够高的浓度下都可以杀死肿瘤细胞,但是由于缺乏特异性,在杀伤肿瘤细胞的同时,也会导致正常细胞的凋亡,导致严重的副作用。该术语包括毒素,如细菌、真菌、植物或动物来源的小分子毒素或酶活性毒素,放射性同位素(例如At211、I131、I125、Y90、Re186、Re188、Sm153、Bi212、P32和Lu的放射性同位素),毒性药物,化疗药物,抗生素和核溶酶。免疫调节剂是免疫关卡分子的抑制剂。The term "drug" refers to a cytotoxic drug or an immunomodulator. Cytotoxic drugs are chemical molecules that have strong ability to destroy the normal growth of tumor cells. In principle, cytotoxic drugs can kill tumor cells at sufficiently high concentrations, but due to the lack of specificity, while killing tumor cells, they can also cause apoptosis of normal cells, leading to serious side effects. The term includes toxins, such as small molecule toxins or enzyme-active toxins from bacteria, fungi, plants or animals, radioactive isotopes (e.g., At 211 , I 131 , I 125 , Y 90 , Re 186 , Re 188 , Sm 153 , Bi 212 , P 32 and radioactive isotopes of Lu), toxic drugs, chemotherapeutic drugs, antibiotics and nucleolytic enzymes. Immunomodulators are inhibitors of immune checkpoint molecules.
术语“连接子”、“连接单元”、“接头单元”、“接头”或“连接片段”是指一端与配体连接而另一端与药物相连的化学结构片段或键,也可以连接其他接头后再与药物相连。The terms "linker", "linking unit", "linker unit", "linker" or "linking fragment" refer to a chemical structure fragment or bond that is connected to a ligand at one end and to a drug at the other end, and can also be connected to other linkers before being connected to the drug.
接头可以包含一种或多种接头构件。例示性的接头构件包括6-马来酰亚氨基己酰基(MC)、马来酰亚氨基丙酰基(MP)、缬氨酸-瓜氨酸(Val-Cit或vc)、丙氨酸-苯丙氨酸(ala-phe)、对氨基苄氧羰基(PAB),及那些源自与接头试剂的偶联的:N-琥珀酰亚氨基4-(2-吡啶基硫代)戊酸酯(SPP)、N-琥珀酰亚氨基4-(N-马来酰亚氨基甲基)环己烷-1羧酸酯(SMCC,在本文中也称作MCC)和N-琥珀酰亚氨基(4-碘-乙酰基)氨基苯甲酸酯(SIAB)。接头可以包括拉伸单元、间隔单元、氨基酸单元和延伸单元。可以通过本领域已知方法合成,诸如US2005-0238649A1中所记载的。接头可以是便于在细胞中释放药物的“可切割接头”。例如,可使用酸不稳定接头(例如腙)、蛋白酶敏感(例如肽酶敏感)接头、光不稳定接头、二甲基接头、或含二硫化物接头(Chari等,Cancer Research 52:127-131(1992);美国专利No.5,208,020)。The joint can include one or more joint components. Exemplary joint components include 6-maleimidocaproyl (MC), maleimidopropionyl (MP), valine-citrulline (Val-Cit or vc), alanine-phenylalanine (ala-phe), p-aminobenzyloxycarbonyl (PAB), and those derived from coupling with joint reagents: N-succinimidyl 4-(2-pyridylthio) valerate (SPP), N-succinimidyl 4-(N-maleimidomethyl) cyclohexane-1 carboxylate (SMCC, also referred to as MCC in this article) and N-succinimidyl (4-iodo-acetyl) aminobenzoate (SIAB). The joint can include a stretching unit, a spacer unit, an amino acid unit and an extension unit. It can be synthesized by methods known in the art, such as described in US2005-0238649A1. The joint can be a "cleavable joint" that is convenient for releasing drugs in cells. For example, an acid-labile linker (eg, hydrazone), a protease-sensitive (eg, peptidase-sensitive) linker, a photolabile linker, a dimethyl linker, or a disulfide-containing linker (Chari et al., Cancer Research 52:127-131 (1992); U.S. Pat. No. 5,208,020) can be used.
术语“拉伸单元”指一端通过碳原子与抗体共价连接而另一端与氨基酸单元、二硫化物部分、磺酰胺部分或非肽化学部分相连的化学结构片段。The term "stretcher" refers to a chemical structural fragment that is covalently linked to an antibody via a carbon atom at one end and is linked to an amino acid unit, a disulfide moiety, a sulfonamide moiety, or a non-peptide chemical moiety at the other end.
术语“间隔单元”是一种双功能化合结构片段,可用于偶联氨基酸单元和细胞毒性药物最终形成抗体-药物偶联物,这种偶联方式可以将细胞毒性药物选择性的连接到氨基酸单元上。The term "spacer unit" refers to a bifunctional compound structural fragment that can be used to couple an amino acid unit and a cytotoxic drug to ultimately form an antibody-drug conjugate. This coupling method can selectively link the cytotoxic drug to the amino acid unit.
术语“氨基酸”是指分子结构中含有氨基和羧基,并且氨基和羧基都直接连接在-CH-结构上的有机化合物。通式是H2NCHRCOOH,R为H、取代或未取代烷基等。根据氨基连结在羧酸中碳原子的位置,可分为α、β、γ、δ、ε……-氨基酸。在生物界中,构成天然蛋白质的氨基酸具有其特定的结构特点,即其氨基直接连接在α-碳原子上,即α-氨基酸,包括甘氨酸(Glycine)、丙氨酸(Alanine)、缬氨酸(Valine)、亮氨酸(Leucine)、异亮氨酸(Isoleucine)、苯丙氨酸(Phenylalanine)、色氨酸(Tryptophan)、酪氨酸(Tyrosine)、天冬氨酸(Aspartic acid)、组氨酸(Histidine)、天冬酰胺(Asparagine)、谷氨酸(Glutamicacid)、赖氨酸(Lysine)、谷氨酰胺(Glutamine)、甲硫氨酸(Methionine)、精氨酸(Arginine)、丝氨酸(Serine)、苏氨酸(Threonine)、半胱氨酸(Cysteine)、脯氨酸(Proline)等。非天然氨基酸如瓜氨酸。如本领域技术人员所公知的,非天然氨基酸并不构成天然蛋白质,因此也不参与本公开中抗体的合成。本公开所用氨基酸三字母代码和单字母代码如J.biol.chem,243,p3558(1968)中所述。The term "amino acid" refers to an organic compound that contains an amino group and a carboxyl group in its molecular structure, and both the amino group and the carboxyl group are directly connected to the -CH- structure. The general formula is H 2 NCHRCOOH, where R is H, substituted or unsubstituted alkyl, etc. Depending on the position of the amino group attached to the carbon atom in the carboxylic acid, it can be divided into α, β, γ, δ, ε...-amino acids. In the biological world, the amino acids constituting natural proteins have their specific structural characteristics, that is, their amino groups are directly connected to the α-carbon atom, i.e., α-amino acids, including glycine, alanine, valine, leucine, isoleucine, phenylalanine, tryptophan, tyrosine, aspartic acid, histidine, asparagine, glutamic acid, lysine, glutamine, methionine, arginine, serine, threonine, cysteine, proline, etc. Non-natural amino acids such as citrulline. As known to those skilled in the art, non-natural amino acids do not constitute natural proteins and therefore do not participate in the synthesis of antibodies in the present disclosure. The three letter codes and one letter codes for amino acids used in this disclosure are as described in J. biol. chem, 243, p3558 (1968).
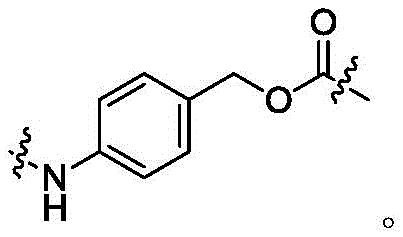
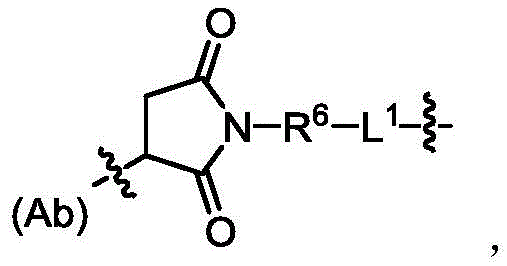
本公开中间隔单元为PAB,结构如对氨基苯甲氧羰基片段,其结构如式(VI)所示,连接在D上,The spacer unit in the present disclosure is PAB, the structure of which is a p-aminobenzyloxycarbonyl fragment, the structure of which is shown in formula (VI), connected to D,
接头组件包括但不限于:Joint components include but are not limited to:
MC=6-马来酰亚氨基己酰基,结构如下:MC = 6-maleimidocaproyl, the structure is as follows:
Val-Cit或“vc”=缬氨酸-瓜氨酸(蛋白酶可切割接头中的例示二肽)Val-Cit or "vc" = valine-citrulline (an exemplary dipeptide in a protease cleavable linker)
瓜氨酸=2-氨基-5-脲基戊酸Citrulline = 2-amino-5-ureidopentanoic acid
Me-Val-Cit=N-甲基-缬氨酸-瓜氨酸(其中接头肽键已经修饰以防止其受到组织蛋白酶B的切割)Me-Val-Cit = N-methyl-valine-citrulline (wherein the linker peptide bond has been modified to protect it from cleavage by cathepsin B)
MC(PEG)6-OH=马来酰亚氨基己酰基-聚乙二醇(可附着于抗体半胱氨酸)MC(PEG) 6 -OH = Maleimidocaproyl-polyethylene glycol (can be attached to antibody cysteine)
SPP=N-琥珀酰亚氨基4-(2-吡啶基硫代)戊酸酯SPP = N-succinimidyl 4-(2-pyridylthio)pentanoate
SPDP=N-琥珀酰亚氨基3-(2-吡啶基二硫代)丙酸酯SPDP = N-succinimidyl 3-(2-pyridyldithio) propionate
SMCC=琥珀酰亚氨基-4-(N-马来酰亚氨基甲基)环己烷-1-羧酸酯SMCC = succinimidyl-4-(N-maleimidomethyl)cyclohexane-1-carboxylate
IT=亚氨基硫烷IT = Iminothiolane
PBS=磷酸缓冲盐溶液。PBS = phosphate buffered saline.
术语“抗体-药物偶联物”,指配体通过稳定的连接单元与具有生物活性的药物相连。在本公开中“抗体-药物偶联物”(antibody drug conjugate,ADC),指把单克隆抗体或者抗体片段通过稳定的连接单元与具有生物活性的毒性药物相连。The term "antibody-drug conjugate" refers to a ligand connected to a biologically active drug via a stable linker. In the present disclosure, "antibody-drug conjugate" (ADC) refers to a monoclonal antibody or antibody fragment connected to a biologically active toxic drug via a stable linker.
术语“载药量”可以表示为药物量和抗体量的比值,即ADC中每个抗体所偶联的药物的平均数量。载药量的范围可以是每个抗体(Ab)连接1-20个,优选1-10个细胞毒性药物(D)。在本公开的实施方式中,载药量表示为k,示例性的可以为1、2、3、4、5、6、7、8、9、10、11、12、13、14、15、16、17、18、19、20或任意两数值之间数值的均值。优选1-10,更优选1-8,或2-8,或2-7,或3-8,或3-7,或3-6,或4-7,或4-6,或4-5的均值。可用常规方法如UV/可见光光谱法、质谱、ELISA试验、单抗分子大小变异体测定法(CE-SDS)和HPLC特征鉴定偶联反应后每个ADC分子的药物平均数量。The term "drug loading" can be expressed as the ratio of the amount of drug to the amount of antibody, i.e., the average number of drugs coupled to each antibody in the ADC. The range of drug loading can be 1-20, preferably 1-10 cytotoxic drugs (D) connected to each antibody (Ab). In the embodiments of the present disclosure, drug loading is expressed as k, which can be 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20 or the mean of any two values. Preferably 1-10, more preferably 1-8, or 2-8, or 2-7, or 3-8, or 3-7, or 3-6, or 4-7, or 4-6, or 4-5 mean. Conventional methods such as UV/visible light spectroscopy, mass spectrometry, ELISA test, monoclonal antibody molecular size variant assay (CE-SDS) and HPLC feature identification of the average number of drugs per ADC molecule after the coupling reaction.
本公开单抗分子大小变异体测定法(CE-SDS)可采用十二烷基硫酸钠毛细管电泳(CE-SDS)紫外检测方法,在还原和非还原条件下,依据分子量大小,按毛电泳法(2015年版《中国药典》0542),定量测定重组单克隆抗体产品的纯度。The monoclonal antibody molecular size variant determination method (CE-SDS) disclosed in the present invention can adopt the sodium dodecyl sulfate capillary electrophoresis (CE-SDS) ultraviolet detection method to quantitatively determine the purity of the recombinant monoclonal antibody product according to the capillary electrophoresis method (2015 edition of the "Chinese Pharmacopoeia" 0542) based on the molecular weight under reducing and non-reducing conditions.
可以用以下非限制性方法控制抗体-药物偶联物的载量,包括:The loading capacity of the antibody-drug conjugate can be controlled by the following non-limiting methods, including:
(1)控制连接试剂和单抗的摩尔比,(1) Control the molar ratio of the linking reagent and the monoclonal antibody,
(2)控制反应时间和温度,(2) Control the reaction time and temperature,
(3)选择不同的反应试剂。(3) Select different reaction reagents.
术语“抗体”指免疫球蛋白,是由两条相同的重链和两条相同的轻链通过链间二硫键连接而成的四肽链结构。免疫球蛋白重链恒定区的氨基酸组成和排列顺序不同,故其抗原性也不同。据此,可将免疫球蛋白分为五类,或称为免疫球蛋白的同种型,即IgM、IgD、IgG、IgA和IgE,其相应的重链分别为μ链、δ链、γ链、α链、和ε链。同一类Ig根据其铰链区氨基酸组成和重链二硫键的数目和位置的差别,又可分为不同的亚类,如IgG可分为IgG1、IgG2、IgG3、IgG4。轻链通过恒定区的不同分为κ链或λ链。五类Ig中每类Ig都可以有κ链或λ链。本公开所述的抗体优选为针对靶细胞上细胞表面抗原的特异性抗体,抗体重链和轻链靠近N端的约110个氨基酸的序列变化很大,为可变区(Fv区);靠近C端的其余氨基酸序列相对稳定,为恒定区。可变区包括3个高变区(HVR)和4个序列相对保守的骨架区(FR)。3个高变区决定抗体的特异性,又称为互补性决定区(CDR)。每条轻链可变区(LCVR)和重链可变区(HCVR)由3个CDR区4个FR区组成,从氨基端到羧基端依次排列的顺序为:FR1,CDR1,FR2,CDR2,FR3,CDR3,FR4。轻链的3个CDR区指LCDR1、LCDR2、和LCDR3;重链的3个CDR区指HCDR1、HCDR2和HCDR3。The term "antibody" refers to immunoglobulin, which is a tetrapeptide chain structure composed of two identical heavy chains and two identical light chains connected by interchain disulfide bonds. The amino acid composition and arrangement order of the constant region of the immunoglobulin heavy chain are different, so their antigenicity is also different. Based on this, immunoglobulins can be divided into five categories, or isotypes of immunoglobulins, namely IgM, IgD, IgG, IgA and IgE, and their corresponding heavy chains are μ chain, δ chain, γ chain, α chain, and ε chain respectively. The same class of Ig can be divided into different subclasses according to the difference in the amino acid composition of its hinge region and the number and position of disulfide bonds in the heavy chain, such as IgG can be divided into IgG1, IgG2, IgG3, and IgG4. Light chains are divided into κ chains or λ chains according to the difference in constant regions. Each of the five classes of Ig can have κ chains or λ chains. The antibodies disclosed herein are preferably specific antibodies against cell surface antigens on target cells. The sequences of about 110 amino acids near the N-terminus of the antibody heavy chain and light chain vary greatly, which is the variable region (Fv region); the remaining amino acid sequences near the C-terminus are relatively stable, which is the constant region. The variable region includes three hypervariable regions (HVRs) and four relatively conservative framework regions (FRs). The three hypervariable regions determine the specificity of the antibody, also known as complementarity determining regions (CDRs). Each light chain variable region (LCVR) and heavy chain variable region (HCVR) consists of three CDR regions and four FR regions, arranged in the order from the amino terminus to the carboxyl terminus: FR1, CDR1, FR2, CDR2, FR3, CDR3, FR4. The three CDR regions of the light chain refer to LCDR1, LCDR2, and LCDR3; the three CDR regions of the heavy chain refer to HCDR1, HCDR2, and HCDR3.
本公开的抗体包括鼠源抗体、嵌合抗体、人源化抗体和全人源抗体,优选人源化抗体和全人源抗体。The antibodies disclosed herein include murine antibodies, chimeric antibodies, humanized antibodies and fully human antibodies, with humanized antibodies and fully human antibodies being preferred.
术语“鼠源抗体”在本公开中为根据本领域知识和技能用鼠制备抗体。制备时用特定抗原注射试验对象,然后分离表达具有所需序列或功能特性的抗体的杂交瘤。The term "murine antibody" in this disclosure refers to antibodies prepared in mice according to the knowledge and skills in the art. When preparing, a test subject is injected with a specific antigen, and then a hybridoma expressing an antibody with the desired sequence or functional characteristics is isolated.
术语“嵌合抗体(chimeric antibody)”,是将鼠源性抗体的可变区与人抗体的恒定区融合而成的抗体,可以减轻鼠源性抗体诱发的免疫应答反应。建立嵌合抗体,要先建立分泌鼠源性特异性单抗的杂交瘤,然后从鼠杂交瘤细胞中克隆可变区基因,再根据需要克隆人抗体的恒定区基因,将鼠可变区基因与人恒定区基因连接成嵌合基因后插入表达载体中,最后在真核系统或原核系统中表达嵌合抗体分子。The term "chimeric antibody" refers to an antibody formed by fusing the variable region of a mouse antibody with the constant region of a human antibody, which can reduce the immune response induced by the mouse antibody. To establish a chimeric antibody, it is necessary to first establish a hybridoma that secretes mouse-specific monoclonal antibodies, then clone the variable region gene from the mouse hybridoma cells, and then clone the constant region gene of the human antibody as needed, connect the mouse variable region gene with the human constant region gene into a chimeric gene, and then insert it into an expression vector, and finally express the chimeric antibody molecule in a eukaryotic system or a prokaryotic system.
术语“人源化抗体(humanized antibody)”,也称为CDR移植抗体(CDR-graftedantibody),是指将鼠的CDR序列移植到人的抗体可变区框架,即不同类型的人种系抗体框架序列中产生的抗体。可以克服嵌合抗体由于携带大量鼠蛋白成分,从而诱导的异源性反应。此类构架序列可以从包括种系抗体基因序列的公共DNA数据库或公开的参考文献获得。如人重链和轻链可变区基因的种系DNA序列可以在“VBase”人种系序列数据库(在因特网www.mrccpe.com.ac.uk/vbase可获得),以及在Kabat,E.A.等人,1991Sequences ofProteins of Immunological Interest,第5版中找到。为避免免疫原性下降的同时,引起的活性下降,可对所述的人抗体可变区框架序列进行最少反向突变或回复突变,以保持活性。本公开的人源化抗体也包括进一步由噬菌体展示对CDR进行亲和力成熟后的人源化抗体。进一步描述参与人源化可使用小鼠抗体的方法的文献包括,例如Queen等,Proc.,Natl.Acad.Sci.USA,88,2869,1991和Winter及其同事的方法[Jones等,Nature,321,522(1986),Riechmann,等,Nature,332,323-327(1988),Verhoeyen,等,Science,239,1534(1988)]。The term "humanized antibody", also known as CDR-grafted antibody, refers to an antibody produced by transplanting the mouse CDR sequence into the human antibody variable region framework, that is, different types of human germline antibody framework sequences. The heterologous reaction induced by chimeric antibodies due to carrying a large amount of mouse protein components can be overcome. Such framework sequences can be obtained from public DNA databases including germline antibody gene sequences or published references. For example, the germline DNA sequences of human heavy chain and light chain variable region genes can be found in the "VBase" human germline sequence database (available on the Internet www.mrccpe.com.ac.uk/vbase), and in Kabat, E.A. et al., 1991 Sequences of Proteins of Immunological Interest, 5th edition. In order to avoid the decrease in activity caused by the decrease in immunogenicity, the human antibody variable region framework sequence can be subjected to minimal reverse mutation or back mutation to maintain activity. The humanized antibodies disclosed in the present invention also include humanized antibodies after further affinity maturation of CDR by phage display. References further describing methods for humanizing mouse antibodies include, for example, Queen et al., Proc., Natl. Acad. Sci. USA, 88, 2869, 1991 and the methods of Winter and colleagues [Jones et al., Nature, 321, 522 (1986), Riechmann, et al., Nature, 332, 323-327 (1988), Verhoeyen, et al., Science, 239, 1534 (1988)].
术语“全人源抗体”、“全人抗体”或“完全人源抗体”,也称“全人源单克隆抗体”,其抗体的可变区和恒定区都是人源的,去除免疫原性和毒副作用。单克隆抗体的发展经历了四个阶段,分别为:鼠源性单克隆抗体、嵌合性单克隆抗体、人源化单克隆抗体和全人源单克隆抗体。本公开为全人源单克隆抗体。全人抗体制备的相关技术主要有:人杂交瘤技术、EBV转化B淋巴细胞技术、噬菌体显示技术(phage display)、转基因小鼠抗体制备技术(transgenic mouse)和单个B细胞抗体制备技术等。The terms "fully human antibody", "fully human antibody" or "completely human antibody", also known as "fully human monoclonal antibody", are antibodies whose variable and constant regions are both human, eliminating immunogenicity and toxic side effects. The development of monoclonal antibodies has gone through four stages, namely: murine monoclonal antibodies, chimeric monoclonal antibodies, humanized monoclonal antibodies and fully human monoclonal antibodies. The present disclosure is a fully human monoclonal antibody. The relevant technologies for the preparation of fully human antibodies mainly include: human hybridoma technology, EBV-transformed B lymphocyte technology, phage display technology (phage display), transgenic mouse antibody preparation technology (transgenic mouse) and single B cell antibody preparation technology, etc.
术语“抗原结合片段”是指抗体的保持特异性结合抗原的能力的一个或多个片段。已显示可利用全长抗体的片段来进行抗体的抗原结合功能。“抗原结合片段”中包含的结合片段的实例包括(i)Fab片段,由VL、VH、CL和CH1结构域组成的单价片段;(ii)F(ab')2片段,包含通过铰链区上的二硫桥连接的两个Fab片段的二价片段;(iii)由VH和CH1结构域组成的Fd片段;(iv)由抗体的单臂的VH和VL结构域组成的Fv片段;(v)单结构域或dAb片段(Ward等人,(1989)Nature341:544-546),其由VH结构域组成;和(vi)分离的互补决定区(CDR)或(vii)可任选地通过合成的接头连接的两个或更多个分离的CDR的组合。此外,虽然Fv片段的两个结构域VL和VH由分开的基因编码,但可使用重组方法,通过合成的接头连接它们,从而使得其能够产生为其中VL和VH区配对形成单价分子的单个蛋白质链(称为单链Fv(scFv);参见,例如,Bird等人(1988)Science242:423-426;和Huston等人(1988)Proc.Natl.Acad.Sci USA85:5879-5883)。此类单链抗体也意欲包括在术语抗体的“抗原结合片段”中。使用本领域技术人员已知的常规技术获得此类抗体片段,并且以与对于完整抗体的方式相同的方式就功用性筛选片段。可通过重组DNA技术或通过酶促或化学断裂完整免疫球蛋白来产生抗原结合部分。抗体可以是不同同种型的抗体,例如,IgG(例如,IgG1、IgG2、IgG3或IgG4亚型)、IgA1、IgA2、IgD、IgE或IgM抗体。The term "antigen-binding fragment" refers to one or more fragments of an antibody that retain the ability to specifically bind to an antigen. It has been shown that fragments of full-length antibodies can be used to perform the antigen-binding function of an antibody. Examples of binding fragments included in "antigen-binding fragments" include (i) Fab fragments, monovalent fragments consisting of VL, VH, CL and CH1 domains; (ii) F(ab') 2 fragments, bivalent fragments comprising two Fab fragments connected by a disulfide bridge on the hinge region; (iii) Fd fragments consisting of VH and CH1 domains; (iv) Fv fragments consisting of the VH and VL domains of a single arm of an antibody; (v) single domain or dAb fragments (Ward et al., (1989) Nature 341: 544-546), which consist of a VH domain; and (vi) isolated complementarity determining regions (CDRs) or (vii) combinations of two or more isolated CDRs, optionally connected by synthetic linkers. In addition, although the two domains VL and VH of the Fv fragment are encoded by separate genes, they can be connected by synthetic linkers using recombinant methods, so that they can be produced as a single protein chain in which the VL and VH regions are paired to form a monovalent molecule (called single-chain Fv (scFv); see, for example, Bird et al. (1988) Science 242: 423-426; and Huston et al. (1988) Proc. Natl. Acad. Sci USA 85: 5879-5883). Such single-chain antibodies are also intended to be included in the term "antigen-binding fragment" of an antibody. Such antibody fragments are obtained using conventional techniques known to those skilled in the art, and the fragments are screened for functionality in the same manner as for intact antibodies. Antigen-binding portions can be produced by recombinant DNA technology or by enzymatic or chemical cleavage of intact immunoglobulins. The antibodies can be of different isotypes, for example, IgG (eg, IgG1, IgG2, IgG3, or IgG4 subtype), IgA1, IgA2, IgD, IgE, or IgM antibodies.
Fab是通过用蛋白酶木瓜蛋白酶(切割H链的224位的氨基酸残基)处理IgG抗体分子所获得的片段中的具有约50,000的分子量并具有抗原结合活性的抗体片段,其中H链N端侧的约一半和整个L链通过二硫键结合在一起。Fab is an antibody fragment having a molecular weight of about 50,000 and antigen-binding activity among fragments obtained by treating IgG antibody molecules with the protease papain (cleaving the amino acid residue at position 224 of the H chain), in which about half of the N-terminal side of the H chain and the entire L chain are bound together by a disulfide bond.
F(ab')2是通过用酶胃蛋白酶消化IgG铰链区中两个二硫键的下方部分而获得的分子量为约100,000并具有抗原结合活性并包含在铰链位置相连的两个Fab区的抗体片段。F(ab')2 is an antibody fragment having a molecular weight of about 100,000 and antigen-binding activity, obtained by digesting the portion below two disulfide bonds in the hinge region of IgG with the enzyme pepsin, and comprising two Fab regions linked at the hinge position.
Fab'是通过切割上述F(ab')2的铰链区的二硫键而获得的分子量为约50,000并具有抗原结合活性的抗体片段。Fab' is an antibody fragment having a molecular weight of about 50,000 and having antigen-binding activity obtained by cleaving the disulfide bond of the hinge region of the above-mentioned F(ab')2.
此外,可以通过将编码抗体的Fab'片段的DNA插入到原核生物表达载体或真核生物表达载体中并将载体导入到原核生物或真核生物中以表达Fab'来生产所述Fab'。Furthermore, the Fab' fragment of the antibody can be produced by inserting a DNA encoding the Fab' fragment into a prokaryotic expression vector or a eukaryotic expression vector and introducing the vector into a prokaryotic organism or a eukaryotic organism to express the Fab'.
术语“单链抗体”、“单链Fv”或“scFv”意指包含通过接头连接的抗体重链可变结构域(或区域;VH)和抗体轻链可变结构域(或区域;VL)的分子。此类scFv分子可具有一般结构:NH2-VL-接头-VH-COOH或NH2-VH-接头-VL-COOH。合适的现有技术接头由重复的GGGGS氨基酸序列或其变体组成,例如使用1-4个重复的变体(Holliger等人(1993),Proc.Natl.Acad.Sci.USA90:6444-6448)。可用于本公开的其他接头由Alfthan等人(1995),Protein Eng.8:725-731,Choi等人(2001),Eur.J.Immuno l.31:94-106,Hu等人(1996),Cancer Res.56:3055-3061,Kipriyanov等人(1999),J.Mol.Biol.293:41-56和Roovers等人(2001),Cancer Immunol.描述。The term "single-chain antibody", "single-chain Fv" or "scFv" means a molecule comprising an antibody heavy chain variable domain (or region; VH) and an antibody light chain variable domain (or region; VL) connected by a linker. Such scFv molecules may have the general structure: NH2 - VL-linker-VH-COOH or NH2 - VH-linker-VL-COOH. Suitable prior art linkers consist of repeated GGGGS amino acid sequences or variants thereof, for example variants using 1-4 repeats (Holliger et al. (1993), Proc. Natl. Acad. Sci. USA 90: 6444-6448). Other linkers useful in the present disclosure are described by Alfthan et al. (1995), Protein Eng. 8:725-731, Choi et al. (2001), Eur. J. Immunol. 31:94-106, Hu et al. (1996), Cancer Res. 56:3055-3061, Kipriyanov et al. (1999), J. Mol. Biol. 293:41-56 and Roovers et al. (2001), Cancer Immunol.
术语“CDR”是指抗体的可变结构域内主要促成抗原结合的6个高变区之一。所述6个CDR的最常用的定义之一由Kabat E.A.等人,(1991)Sequences of proteins ofimmunological interest.NIH Publication91-3242)提供。如本文中使用的,CDR的Kabat定义只应用于轻链可变结构域的CDR1、CDR2和CDR3(CDR L1、CDR L2、CDR L3或L1、L2、L3),以及重链可变结构域的CDR2和CDR3(CDR H2、CDR H3或H2、H3)。通常,每个重链可变区中存在三个CDR(HCDR1、HCDR2、HCDR3),每个轻链可变区中存在三个CDR(LCDR1、LCDR2、LCDR3)。可以使用各种公知方案中的任何一种来确定CDR的氨基酸序列边界,包括“Kabat”编号规则(参见Kabat等(1991),“Sequences of Proteins of Immunological Interest”,第5版,Public Health Service,National Institutes of Health,Bethesda,MD)、“Chothia”编号规则(参见Al-Lazikani等人,(1997)JMB 273:927-948)和ImMunoGenTics(IMGT)编号规则(参见Lefranc M.P.,Immunologist,7,132-136(1999);Lefranc,M.P.等,Dev.Comp.Immunol.,27,55-77(2003))等。遵循IMGT规则,抗体的CDR区可以使用程序IMGT/DomainGap Align确定。The term "CDR" refers to one of the six hypervariable regions in the variable domain of an antibody that primarily contribute to antigen binding. One of the most commonly used definitions of the six CDRs is provided by Kabat E.A. et al. (1991) Sequences of proteins of immunological interest. NIH Publication 91-3242). As used herein, the Kabat definition of CDR applies only to CDR1, CDR2 and CDR3 (CDR L1, CDR L2, CDR L3 or L1, L2, L3) of the light chain variable domain, and CDR2 and CDR3 (CDR H2, CDR H3 or H2, H3) of the heavy chain variable domain. Typically, there are three CDRs (HCDR1, HCDR2, HCDR3) in each heavy chain variable region, and three CDRs (LCDR1, LCDR2, LCDR3) in each light chain variable region. The amino acid sequence boundaries of CDRs can be determined using any of a variety of well-known schemes, including the "Kabat" numbering convention (see Kabat et al. (1991), "Sequences of Proteins of Immunological Interest", 5th Edition, Public Health Service, National Institutes of Health, Bethesda, MD), the "Chothia" numbering convention (see Al-Lazikani et al., (1997) JMB 273: 927-948), and the ImMunoGenTics (IMGT) numbering convention (see Lefranc M.P., Immunologist, 7, 132-136 (1999); Lefranc, M.P. et al., Dev. Comp. Immunol., 27, 55-77 (2003)), etc. Following the IMGT convention, the CDR regions of antibodies can be determined using the program IMGT/DomainGap Align.
术语“抗体框架”,是指可变结构域VL或VH的一部分,其用作该可变结构域的抗原结合环(CDR)的支架。从本质上讲,其是不具有CDR的可变结构域。The term "antibody framework" refers to a portion of a variable domain VL or VH that serves as a scaffold for the antigen binding loops (CDRs) of the variable domain. Essentially, it is a variable domain without CDRs.
术语“表位”或“抗原决定簇”是指抗原上免疫球蛋白或抗体特异性结合的部位。表位通常以独特的空间构象包括至少3、4、5、6、7、8、9、10、11、12、13、14或15个连续或非连续的氨基酸(参见,例如,Epitope Mapping Protocols in Methods in Molecular Biology,第66卷,G.E.Morris,Ed.(1996))。The term "epitope" or "antigenic determinant" refers to the site on an antigen to which an immunoglobulin or antibody specifically binds. An epitope typically includes at least 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14 or 15 consecutive or non-continuous amino acids in a unique spatial conformation (see, e.g., Epitope Mapping Protocols in Methods in Molecular Biology, Vol. 66, G.E.Morris, Ed. (1996)).
术语“特异性结合”、“选择性结合”、“选择性地结合”和“特异性地结合”是指抗体对预先确定的抗原上的表位的结合。通常,抗体以大约小于10-7M,例如:大约小于10-8M、10- 9M或10-10M或更小的亲和力(KD)结合。The terms "specific binding", "selective binding", "selectively binds" and "specifically binds" refer to the binding of an antibody to a predetermined epitope on an antigen. Typically, the antibody binds with an affinity (KD) of less than about 10-7 M, for example, less than about 10-8 M, 10-9 M or 10-10 M or less.
术语“核酸分子”是指DNA分子和RNA分子。核酸分子可以是单链或双链的,但优选是双链DNA。当将核酸与另一个核酸序列置于功能关系中时,核酸是“有效连接的”。例如,如果启动子或增强子影响编码序列的转录,那么启动子或增强子有效地连接至所述编码序列。The term "nucleic acid molecule" refers to DNA molecules and RNA molecules. Nucleic acid molecules can be single-stranded or double-stranded, but are preferably double-stranded DNA. A nucleic acid is "operably linked" when it is placed in a functional relationship with another nucleic acid sequence. For example, if a promoter or enhancer affects the transcription of a coding sequence, then the promoter or enhancer is operably linked to the coding sequence.
术语“载体”是指能够运输已与其连接的另一个核酸的核酸分子。在一个实施方案中,载体是“质粒”,其是指可将另外的DNA区段连接至其中的环状双链DNA环。在另一个实施方案中,载体是病毒载体,其中可将另外的DNA区段连接至病毒基因组中。本文中公开的载体能够在已引入它们的宿主细胞中自主复制(例如,具有细菌的复制起点的细菌载体和附加型哺乳动物载体)或可在引入宿主细胞后整合入宿主细胞的基因组,从而随宿主基因组一起复制(例如,非附加型哺乳动物载体)。The term "vector" refers to a nucleic acid molecule capable of transporting another nucleic acid connected thereto. In one embodiment, the vector is a "plasmid", which refers to a circular double-stranded DNA loop into which other DNA segments can be connected. In another embodiment, the vector is a viral vector, in which other DNA segments can be connected to the viral genome. The vector disclosed herein can be autonomously replicated in the host cell introduced therein (e.g., bacterial vectors and additional mammalian vectors with a bacterial origin of replication) or can be integrated into the genome of the host cell after being introduced into the host cell, thereby replicating with the host genome (e.g., non-additional mammalian vectors).
现有技术中熟知生产和纯化抗体和抗原结合片段的方法,如冷泉港的抗体实验技术指南,5-8章和15章。抗原结合片段同样可以用常规方法制备。发明所述的抗体或抗原结合片段用基因工程方法在非人源的CDR区加上一个或多个人源FR区。人FR种系序列可以通过比对IMGT人类抗体可变区种系基因数据库和MOE软件,从ImMunoGeneTics(IMGT)的网站http://imgt.cines.fr得到,或者从免疫球蛋白杂志,2001ISBN012441351上获得。Methods for producing and purifying antibodies and antigen-binding fragments are well known in the prior art, such as the Cold Spring Harbor Guide to Antibody Experimental Techniques, Chapters 5-8 and 15. Antigen-binding fragments can also be prepared by conventional methods. The antibodies or antigen-binding fragments of the invention are genetically engineered to add one or more human FR regions to the non-human CDR region. Human FR germline sequences can be obtained from the ImMunoGeneTics (IMGT) website http://imgt.cines.fr by comparing the IMGT human antibody variable region germline gene database and MOE software, or from the Journal of Immunoglobulins, 2001 ISBN012441351.
术语“宿主细胞”是指已向其中引入了表达载体的细胞。宿主细胞可包括细菌、微生物、植物或动物细胞。易于转化的细菌包括肠杆菌科(enterobacteriaceae)的成员,例如大肠杆菌(Escherichia coli)或沙门氏菌(Salmonella)的菌株;芽孢杆菌科(Bacillaceae)例如枯草芽孢杆菌(Bacillus subtilis);肺炎球菌(Pneumococcus);链球菌(Streptococcus)和流感嗜血菌(Haemophilus influenzae)。适当的微生物包括酿酒酵母(Saccharomyces cerevisiae)和毕赤酵母(Pichia pastoris)。适当的动物宿主细胞系包括CHO(中国仓鼠卵巢细胞系)和NS0细胞。The term "host cell" refers to a cell into which an expression vector has been introduced. Host cells may include bacteria, microorganisms, plants or animal cells. Easily transformed bacteria include members of the family Enterobacteriaceae, such as strains of Escherichia coli or Salmonella; Bacillaceae, such as Bacillus subtilis; Pneumococcus; Streptococcus and Haemophilus influenzae. Suitable microorganisms include Saccharomyces cerevisiae and Pichia pastoris. Suitable animal host cell lines include CHO (Chinese Hamster Ovary Cell Line) and NS0 cells.
本公开工程化的抗体或抗原结合片段可用常规方法制备和纯化。比如,编码重链和轻链的cDNA序列,可以克隆并重组至GS表达载体。重组的免疫球蛋白表达载体可以稳定地转染CHO细胞。作为一种更推荐的现有技术,哺乳动物类表达系统会导致抗体的糖基化,特别是在Fc区的高度保守N端位点。阳性的克隆在生物反应器的无血清培养基中扩大培养以生产抗体。分泌了抗体的培养液可以用常规技术纯化。比如,用含调整过的缓冲液的A或GSepharose FF柱进行纯化。洗去非特异性结合的组分。再用PH梯度法洗脱结合的抗体,用SDS-PAGE检测抗体片段,收集。抗体可用常规方法进行过滤浓缩。可溶的混合物和多聚体,也可以用常规方法去除,比如分子筛、离子交换。得到的产物需立即冷冻,如-70℃,或者冻干。The engineered antibodies or antigen-binding fragments disclosed herein can be prepared and purified by conventional methods. For example, cDNA sequences encoding heavy and light chains can be cloned and recombined into GS expression vectors. The recombinant immunoglobulin expression vector can stably transfect CHO cells. As a more recommended prior art, mammalian expression systems lead to glycosylation of antibodies, especially at the highly conserved N-terminal site of the Fc region. Positive clones are expanded in serum-free culture medium in a bioreactor to produce antibodies. The culture fluid that secretes antibodies can be purified by conventional techniques. For example, purification is performed using an A or G Sepharose FF column containing an adjusted buffer. Non-specifically bound components are washed away. The bound antibodies are then eluted using a pH gradient method, and the antibody fragments are detected by SDS-PAGE and collected. The antibodies can be filtered and concentrated using conventional methods. Soluble mixtures and polymers can also be removed using conventional methods, such as molecular sieves and ion exchange. The resulting product must be immediately frozen, such as -70°C, or lyophilized.
氨基酸序列“同一性”指在比对氨基酸序列及必要时引入间隙,以达成最大序列同一性百分比,且不将任何保守性取代视为序列同一性的一部分,第一序列中与第二序列中的氨基酸残基同一的氨基酸残基的百分比。为测定氨基酸序列同一性百分比的目的,比对可以通过属于本领域技术的范围内的多种方式来实现,例如使用公开可得到的计算机软件,诸如BLAST、BLAST-2、ALIGN、ALIGN-2或Megalign(DNASTAR)软件。本领域技术人员可确定适用于测量比对的参数,包括在所比较的序列全长上达成最大比对所需的任何算法。Amino acid sequence "identity" refers to the percentage of amino acid residues in a first sequence that are identical to the amino acid residues in a second sequence, while aligning the amino acid sequences and, if necessary, introducing gaps to achieve maximum sequence identity percentage, and not considering any conservative substitutions as part of the sequence identity. For the purpose of determining the percentage of amino acid sequence identity, alignment can be achieved in a variety of ways within the scope of the art, for example, using publicly available computer software such as BLAST, BLAST-2, ALIGN, ALIGN-2 or Megalign (DNASTAR) software. Those skilled in the art can determine the parameters suitable for measuring alignment, including any algorithm required to achieve maximum alignment over the full length of the compared sequences.
术语“肽”是指介于氨基酸和蛋白质之间的化合物片段,由2个或2个以上氨基酸分子通过肽键相互连接而成,是蛋白质的结构与功能片段,如激素、酶类等本质上都是肽。The term "peptide" refers to a compound fragment between amino acids and proteins. It is composed of two or more amino acid molecules connected to each other by peptide bonds. It is a structural and functional fragment of protein. Hormones, enzymes, etc. are essentially peptides.
术语“糖”是指由C、H、O三种元素组成的生物大分子,可分为单糖、二糖和多糖等。The term "sugar" refers to a biological macromolecule composed of three elements: C, H, and O, which can be divided into monosaccharides, disaccharides, and polysaccharides.
术语“荧光探针”是指在紫外-可见-近红外区有特征荧光,并且其荧光性质(激发和发射波长、强度、寿命和偏振等)可随所处环境的性质,如极性、折射率、粘度等改变而灵敏地改变的一类荧光性分子,其与核酸(DNA或RNA)、蛋白质或其他大分子结构非共价相互作用而使一种或几种荧光性质发生改变,可用于研究大分子物质的性质和行为。The term "fluorescent probe" refers to a class of fluorescent molecules that have characteristic fluorescence in the ultraviolet-visible-near infrared region, and whose fluorescence properties (excitation and emission wavelengths, intensity, lifetime and polarization, etc.) can sensitively change with the properties of the environment, such as polarity, refractive index, viscosity, etc. They non-covalently interact with nucleic acids (DNA or RNA), proteins or other macromolecular structures to change one or several fluorescence properties, and can be used to study the properties and behaviors of macromolecules.
术语“烷基”指饱和脂肪族烃基团,其为包含1至20个碳原子的直链或支链基团,优选含有1至12个碳原子的烷基,更优选含有1至10个碳原子的烷基,最优选含有1至6个碳原子的烷基。非限制性实例包括甲基、乙基、正丙基、异丙基、正丁基、异丁基、叔丁基、正己基、1-乙基-2-甲基丙基,及其各种支链异构体等。更优选的是含有1至6个碳原子的低级烷基,非限制性实施例包括甲基、乙基、正丙基、异丙基、正丁基、异丁基、叔丁基、仲丁基、正戊基、1,1-二甲基丙基、1,2-二甲基丙基、2,2-二甲基丙基、1-乙基丙基、2-甲基丁基等。烷基可以是取代的或非取代的,当被取代时,取代基可以在任何可使用的连接点上被取代,所述取代基优选为一个或多个以下基团,其独立地选自烷基、烷氧基、卤素、氨基、氰基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基。The term "alkyl" refers to a saturated aliphatic hydrocarbon group, which is a straight or branched chain group containing 1 to 20 carbon atoms, preferably an alkyl group containing 1 to 12 carbon atoms, more preferably an alkyl group containing 1 to 10 carbon atoms, and most preferably an alkyl group containing 1 to 6 carbon atoms. Non-limiting examples include methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, tert-butyl, n-hexyl, 1-ethyl-2-methylpropyl, and various branched isomers thereof. More preferred are lower alkyl groups containing 1 to 6 carbon atoms, and non-limiting examples include methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, tert-butyl, sec-butyl, n-pentyl, 1,1-dimethylpropyl, 1,2-dimethylpropyl, 2,2-dimethylpropyl, 1-ethylpropyl, 2-methylbutyl, and the like. The alkyl group may be substituted or unsubstituted. When substituted, the substituent may be substituted at any available point of attachment. The substituent is preferably one or more of the following groups independently selected from alkyl, alkoxy, halogen, amino, cyano, nitro, hydroxy, hydroxyalkyl, cycloalkyl, heterocyclyl, aryl and heteroaryl.
术语“杂烷基”指含有一个或多个选自N、O或S的杂原子的烷基,其中烷基如上所定义。The term "heteroalkyl" refers to an alkyl group containing one or more heteroatoms selected from N, O or S, wherein alkyl is as defined above.
“一价基团”是指一个化合物从“形式上”消除一个单价的原子或基团。“亚基”则是指化合物从“形式上”消除两个单价或一个双价形成的原子或原子团。示例“烷基”是指由烷烃分子中去除1个氢原子后余下的部分,包括1至20个碳原子的直链和支链一价基团。含有1至6个碳原子的烷基,非限制性实施例包括甲基、乙基、正丙基、异丙基、正丁基、异丁基、叔丁基、仲丁基、正戊基、1,1-二甲基丙基、1,2-二甲基丙基、2,2-二甲基丙基及其各种支链异构体等。"Monovalent group" refers to a compound that "formally" eliminates a monovalent atom or group. "Subunit" refers to a compound that "formally" eliminates two monovalent or one divalent atoms or groups. Example "alkyl" refers to the remaining part after removing one hydrogen atom from an alkane molecule, including straight-chain and branched monovalent groups of 1 to 20 carbon atoms. Alkyl groups containing 1 to 6 carbon atoms, non-limiting examples include methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, tert-butyl, sec-butyl, n-pentyl, 1,1-dimethylpropyl, 1,2-dimethylpropyl, 2,2-dimethylpropyl and various branched isomers thereof.
术语“亚烷基”指饱和的直链或支链脂肪族烃基,其具有2个从母体烷的相同碳原子或两个不同的碳原子上除去两个氢原子所衍生的残基,其为包含1至20个碳原子的直链或支链基团,优选含有1至12个碳原子,更优选含有1至6个碳原子的亚烷基。亚烷基的非限制性实例包括但不限于亚甲基(-CH2-)、1,1-亚乙基(-CH(CH3)-)、1,2-亚乙基(-CH2CH2)-、1,1-亚丙基(-CH(CH2CH3)-)、1,2-亚丙基(-CH2CH(CH3)-)、1,3-亚丙基(-CH2CH2CH2-)、1,4-亚丁基(-CH2CH2CH2CH2-)和1,5-亚丁基(-CH2CH2CH2CH2CH2-)等。亚烷基可以是取代的或非取代的,当被取代时,取代基可以在任何可使用的连接点上被取代,所述取代基优选独立地任选选自卤素、羟基、氰基、氨基、烷基、氯代烷基、烷氧基和环烷基中的一个或多个。同理,“亚烯基”如前所定义。The term "alkylene" refers to a saturated straight or branched aliphatic hydrocarbon group having two residues derived from the same carbon atom or two different carbon atoms of an alkane radical by removing two hydrogen atoms, and is a straight or branched group containing 1 to 20 carbon atoms, preferably an alkylene group containing 1 to 12 carbon atoms, and more preferably an alkylene group containing 1 to 6 carbon atoms. Non-limiting examples of alkylene groups include, but are not limited to, methylene (—CH 2 —), 1,1-ethylene (—CH(CH 3 )—), 1,2-ethylene (—CH 2 CH 2 )—, 1,1-propylene (—CH(CH 2 CH 3 )—), 1,2-propylene (—CH 2 CH(CH 3 )—), 1,3-propylene (—CH 2 CH 2 CH 2 —), 1,4-butylene (—CH 2 CH 2 CH 2 CH 2 —), and 1,5-butylene (—CH 2 CH 2 CH 2 CH 2 CH 2 —). Alkylene groups may be substituted or unsubstituted, and when substituted, the substituents may be substituted at any available attachment point, and the substituents are preferably independently selected from one or more of halogen, hydroxy, cyano, amino, alkyl, chloroalkyl, alkoxy, and cycloalkyl. Similarly, “alkenylene” is as defined above.
术语“烷氧基”指-O-(烷基)和-O-(非取代的环烷基),其中烷基或环烷基的定义如上所述。烷氧基的非限制性实例包括:甲氧基、乙氧基、丙氧基、丁氧基、环丙氧基、环丁氧基、环戊氧基、环己氧基。烷氧基可以是任选取代的或非取代的,当被取代时,取代基优选为一个或多个以下基团,其独立地选自烷基、烯基、炔基、烷氧基、烷硫基、烷基氨基、卤素、巯基、羟基、硝基、氰基、环烷基、杂环烷基、芳基、杂芳基、环烷氧基、杂环烷氧基、环烷硫基、杂环烷硫基。The term "alkoxy" refers to-O-(alkyl) and-O-(unsubstituted cycloalkyl), wherein the definition of alkyl or cycloalkyl is as described above. Non-limiting examples of alkoxy include: methoxy, ethoxy, propoxy, butoxy, cyclopropyloxy, cyclobutyloxy, cyclopentyloxy, cyclohexyloxy. Alkoxy can be optionally substituted or unsubstituted, and when substituted, substituents are preferably one or more of the following groups, which are independently selected from alkyl, alkenyl, alkynyl, alkoxy, alkylthio, alkylamino, halogen, sulfhydryl, hydroxyl, nitro, cyano, cycloalkyl, heterocycloalkyl, aryl, heteroaryl, cycloalkyloxy, heterocycloalkyloxy, cycloalkylthio, heterocycloalkylthio.
术语“环烷基”指饱和或部分不饱和单环或多环环状烃取代基,环烷基环包含3至20个碳原子,优选包含3至12个碳原子,更优选包含3至10个碳原子,最优选包含3至8个碳原子。单环环烷基的非限制性实例包括环丙基、环丁基、环戊基、环戊烯基、环己基、环己烯基、环己二烯基、环庚基、环庚三烯基、环辛基等;多环环烷基包括螺环、稠环和桥环的环烷基。The term "cycloalkyl" refers to a saturated or partially unsaturated monocyclic or polycyclic cyclic hydrocarbon substituent, wherein the cycloalkyl ring contains 3 to 20 carbon atoms, preferably 3 to 12 carbon atoms, more preferably 3 to 10 carbon atoms, and most preferably 3 to 8 carbon atoms. Non-limiting examples of monocyclic cycloalkyls include cyclopropyl, cyclobutyl, cyclopentyl, cyclopentenyl, cyclohexyl, cyclohexenyl, cyclohexadienyl, cycloheptyl, cycloheptatrienyl, cyclooctyl, etc.; polycyclic cycloalkyls include spirocyclic, fused and bridged cycloalkyls.
术语“杂环烷基”指饱和或部分不饱和单环或多环环状烃取代基,其包含3至20个环原子,其中一个或多个环原子为选自氮、氧或S(O)m(其中m是整数0至2)的杂原子,但不包括-O-O-、-O-S-或-S-S-的环部分,其余环原子为碳。优选包含3至12个环原子,其中1~4个是杂原子;更优选环烷基环包含3至10个环原子。单环杂环烷基的非限制性实例包括吡咯烷基、哌啶基、哌嗪基、吗啉基、硫代吗啉基、高哌嗪基等。多环杂环烷基包括螺环、稠环和桥环的杂环烷基。The term "heterocycloalkyl" refers to a saturated or partially unsaturated monocyclic or polycyclic cyclic hydrocarbon substituent containing 3 to 20 ring atoms, one or more of which are heteroatoms selected from nitrogen, oxygen or S(O) m (wherein m is an integer from 0 to 2), but excluding the ring portion of -OO-, -OS- or -SS-, and the remaining ring atoms are carbon. Preferably, it contains 3 to 12 ring atoms, of which 1 to 4 are heteroatoms; more preferably, the cycloalkyl ring contains 3 to 10 ring atoms. Non-limiting examples of monocyclic heterocycloalkyls include pyrrolidinyl, piperidinyl, piperazinyl, morpholinyl, thiomorpholinyl, homopiperazinyl, etc. Polycyclic heterocycloalkyls include spirocyclic, fused ring and bridged heterocycloalkyls.
术语“螺杂环烷基”指5至20元的单环之间共用一个原子(称螺原子)的多环杂环烷基团,其中一个或多个环原子为选自氮、氧或S(O)m(其中m是整数0至2)的杂原子,其余环原子为碳。其可以含有一个或多个双键,但没有一个环具有完全共轭的π电子系统。优选为6至14元,更优选为7至10元。根据环与环之间共用螺原子的数目将螺杂环烷基分为单螺杂环烷基、双螺杂环烷基或多螺杂环烷基,优选为单螺杂环烷基和双螺杂环烷基。更优选为4元/4元、4元/5元、4元/6元、5元/5元或5元/6元单螺杂环烷基。螺杂环烷基的非限制性实例包括:The term "spiro heterocycloalkyl" refers to a polycyclic heterocycloalkyl group that shares one atom (called spiro atom) between 5 to 20 monocyclic rings, wherein one or more ring atoms are heteroatoms selected from nitrogen, oxygen or S(O) m (wherein m is an integer from 0 to 2), and the remaining ring atoms are carbon. It may contain one or more double bonds, but no ring has a completely conjugated π electron system. It is preferably 6 to 14 yuan, more preferably 7 to 10 yuan. According to the number of spiro atoms shared between rings, spiro heterocycloalkyl is divided into single spiro heterocycloalkyl, double spiro heterocycloalkyl or multi-spiro heterocycloalkyl, preferably single spiro heterocycloalkyl and double spiro heterocycloalkyl. More preferably 4 yuan/4 yuan, 4 yuan/5 yuan, 4 yuan/6 yuan, 5 yuan/5 yuan or 5 yuan/6 yuan single spiro heterocycloalkyl. Non-limiting examples of spiro heterocycloalkyl include:
术语“稠杂环烷基”指5至20元,系统中的每个环与体系中的其他环共享毗邻的一对原子的多环杂环烷基团,一个或多个环可以含有一个或多个双键,但没有一个环具有完全共轭的π电子系统,其中一个或多个环原子为选自氮、氧或S(O)m(其中m是整数0至2)的杂原子,其余环原子为碳。优选为6至14元,更优选为7至10元。根据组成环的数目可以分为双环、三环、四环或多环稠杂环烷基,优选为双环或三环,更优选为5元/5元或5元/6元双环稠杂环烷基。稠杂环烷基的非限制性实例包括:The term "fused heterocycloalkyl" refers to a polycyclic heterocycloalkyl group of 5 to 20 yuan, each ring in the system shares a pair of adjacent atoms with other rings in the system, one or more rings may contain one or more double bonds, but no ring has a completely conjugated π electron system, wherein one or more ring atoms are selected from nitrogen, oxygen or S (O) m (wherein m is an
术语“桥杂环烷基”指5至14元,任意两个环共用两个不直接连接的原子的多环杂环烷基团,其可以含有一个或多个双键,但没有一个环具有完全共轭的π电子系统,其中一个或多个环原子为选自氮、氧或S(O)m(其中m是整数0至2)的杂原子,其余环原子为碳。优选为6至14元,更优选为7至10元。根据组成环的数目可以分为双环、三环、四环或多环桥杂环烷基,优选为双环、三环或四环,更优选为双环或三环。桥杂环烷基的非限制性实例包括:The term "bridged heterocycloalkyl" refers to a 5 to 14-membered polycyclic heterocycloalkyl group in which any two rings share two atoms that are not directly connected, which may contain one or more double bonds, but no ring has a completely conjugated π electron system, wherein one or more ring atoms are heteroatoms selected from nitrogen, oxygen or S(O) m (wherein m is an integer from 0 to 2), and the remaining ring atoms are carbon. Preferably, it is 6 to 14 members, more preferably 7 to 10 members. According to the number of constituent rings, it can be divided into bicyclic, tricyclic, tetracyclic or polycyclic bridged heterocycloalkyl, preferably bicyclic, tricyclic or tetracyclic, more preferably bicyclic or tricyclic. Non-limiting examples of bridged heterocycloalkyl include:
所述杂环烷基环可以稠合于芳基、杂芳基或环烷基环上,其中与母体结构连接在一起的环为杂环烷基,其非限制性实例包括:The heterocycloalkyl ring may be fused to an aryl, heteroaryl or cycloalkyl ring, wherein the ring attached to the parent structure is a heterocycloalkyl, non-limiting examples of which include:
等。 wait.
杂环烷基可以是任选取代的或非取代的,当被取代时,取代基优选为一个或多个以下基团,其独立地选自烷基、烯基、炔基、烷氧基、烷硫基、烷基氨基、卤素、巯基、羟基、硝基、氰基、环烷基、杂环烷基、芳基、杂芳基、环烷氧基、杂环烷氧基、环烷硫基、杂环烷硫基、氧代基。The heterocycloalkyl radical may be optionally substituted or unsubstituted, and when substituted, the substituents are preferably one or more of the following groups independently selected from alkyl, alkenyl, alkynyl, alkoxy, alkylthio, alkylamino, halogen, mercapto, hydroxy, nitro, cyano, cycloalkyl, heterocycloalkyl, aryl, heteroaryl, cycloalkyloxy, heterocycloalkyloxy, cycloalkylthio, heterocycloalkylthio, and oxo.
术语“芳基”指具有共轭的π电子体系的6至14元全碳单环或稠合多环(也就是共享毗邻碳原子对的环)基团,优选为6至10元,例如苯基和萘基,优选苯基。所述芳基环可以稠合于杂芳基、杂环烷基或环烷基环上,其中与母体结构连接在一起的环为芳基环,其非限制性实例包括:The term "aryl" refers to a 6- to 14-membered all-carbon monocyclic or fused polycyclic (i.e., rings that share adjacent pairs of carbon atoms) group having a conjugated π electron system, preferably 6- to 10-membered, such as phenyl and naphthyl, preferably phenyl. The aryl ring may be fused to a heteroaryl, heterocycloalkyl or cycloalkyl ring, wherein the ring connected to the parent structure is an aryl ring, non-limiting examples of which include:
芳基可以是取代的或非取代的,当被取代时,取代基优选为一个或多个以下基团,其独立地选自烷基、烯基、炔基、烷氧基、烷硫基、烷基氨基、卤素、巯基、羟基、硝基、氰基、环烷基、杂环烷基、芳基、杂芳基、环烷氧基、杂环烷氧基、环烷硫基、杂环烷硫基。The aryl group may be substituted or unsubstituted. When substituted, the substituent is preferably one or more of the following groups independently selected from alkyl, alkenyl, alkynyl, alkoxy, alkylthio, alkylamino, halogen, mercapto, hydroxy, nitro, cyano, cycloalkyl, heterocycloalkyl, aryl, heteroaryl, cycloalkyloxy, heterocycloalkyloxy, cycloalkylthio, heterocycloalkylthio.
术语“杂芳基”指包含1至4个杂原子、5至14个环原子的杂芳族体系,其中杂原子选自氧、硫和氮。杂芳基优选为5至10元,更优选为5元或6元,例如呋喃基、噻吩基、吡啶基、吡咯基、N-烷基吡咯基、嘧啶基、吡嗪基、咪唑基、四唑基等。所述杂芳基环可以稠合于芳基、杂环烷基或环烷基环上,其中与母体结构连接在一起的环为杂芳基环,其非限制性实例包括:The term "heteroaryl" refers to a heteroaromatic system containing 1 to 4 heteroatoms, 5 to 14 ring atoms, wherein the heteroatoms are selected from oxygen, sulfur and nitrogen. The heteroaryl is preferably 5 to 10 yuan, more preferably 5 or 6 yuan, such as furanyl, thienyl, pyridyl, pyrrolyl, N-alkylpyrrolyl, pyrimidyl, pyrazinyl, imidazolyl, tetrazolyl, etc. The heteroaryl ring can be fused to an aryl, heterocycloalkyl or cycloalkyl ring, wherein the ring connected to the parent structure is a heteroaryl ring, non-limiting examples of which include:
杂芳基可以是任选取代的或非取代的,当被取代时,取代基优选为一个或多个以下基团,其独立地选自烷基、烯基、炔基、烷氧基、烷硫基、烷基氨基、卤素、巯基、羟基、硝基、氰基、环烷基、杂环烷基、芳基、杂芳基、环烷氧基、杂环烷氧基、环烷硫基、杂环烷硫基。The heteroaryl group may be optionally substituted or unsubstituted, and when substituted, the substituents are preferably one or more of the following groups independently selected from alkyl, alkenyl, alkynyl, alkoxy, alkylthio, alkylamino, halogen, mercapto, hydroxy, nitro, cyano, cycloalkyl, heterocycloalkyl, aryl, heteroaryl, cycloalkyloxy, heterocycloalkyloxy, cycloalkylthio, heterocycloalkylthio.
术语“氨基杂环烷基”指杂环烷基被一个或多个氨基取代,优选被一个氨基取代,其中杂环烷基如上所定义,其中“氨基”指-NH2。本公开的代表性实施例如下:The term "aminoheterocycloalkyl" refers to a heterocycloalkyl group substituted by one or more amino groups, preferably by one amino group, wherein heterocycloalkyl is as defined above, and wherein "amino" refers to -NH 2 . Representative embodiments of the present disclosure are as follows:
术语“杂环烷基氨基”指氨基被一个或多个杂环烷基取代,优选被一个杂环烷基取代,其中氨基如上所定义,其中杂环烷基如上所定义。本公开的代表性实施例如下:The term "heterocycloalkylamino" refers to an amino group substituted by one or more heterocycloalkyl groups, preferably by one heterocycloalkyl group, wherein the amino group is as defined above, and wherein the heterocycloalkyl group is as defined above. Representative embodiments of the present disclosure are as follows:
术语“环烷基氨基”指氨基被一个或多个环烷基取代,优选被一个环烷基取代,其中氨基如上所定义,其中环烷基如上所定义。本公开的代表性实施例如下:The term "cycloalkylamino" refers to an amino group substituted by one or more cycloalkyl groups, preferably by one cycloalkyl group, wherein the amino group is as defined above, and wherein the cycloalkyl group is as defined above. Representative embodiments of the present disclosure are as follows:
术语“环烷基烷基”指烷基被一个或多个环烷基取代,优选被一个环烷基取代,其中烷基如上所定义,其中环烷基如上所定义。The term "cycloalkylalkyl" refers to an alkyl group substituted by one or more cycloalkyl groups, preferably by one cycloalkyl group, wherein alkyl is as defined above and wherein cycloalkyl is as defined above.
术语“卤代烷基”指烷基被一个或多个卤素取代,其中烷基如上所定义。The term "haloalkyl" refers to an alkyl group substituted with one or more halogens, wherein alkyl is as defined above.
术语“氘代烷基”指烷基被一个或多个氘原子取代,其中烷基如上所定义。The term "deuterated alkyl" refers to an alkyl group substituted with one or more deuterium atoms, wherein alkyl is as defined above.
术语“羟基”指-OH基团。The term "hydroxy" refers to an -OH group.
术语“卤素”指氟、氯、溴或碘。The term "halogen" refers to fluorine, chlorine, bromine or iodine.
术语“氨基”指-NH2。The term "amino" refers to -NH2 .
术语“硝基”指-NO2。The term "nitro" refers to -NO2 .
化学式中简称“Me”为甲基。The abbreviation "Me" in the chemical formula is methyl.
“任选”或“任选地”意味着随后所描述的事件或环境可以但不必发生,该说明包括该事件或环境发生或不发生地场合。例如,“任选被烷基取代的杂环烷基团”意味着烷基可以但不必须存在,该说明包括杂环烷基团被烷基取代的情形和杂环烷基团不被烷基取代的情形。"Optional" or "optionally" means that the subsequently described event or circumstance may but need not occur, and the description includes instances where the event or circumstance occurs or does not occur. For example, "a heterocycloalkyl group optionally substituted with an alkyl group" means that an alkyl group may but need not be present, and the description includes instances where the heterocycloalkyl group is substituted with an alkyl group and instances where the heterocycloalkyl group is not substituted with an alkyl group.
“取代的”指基团中的一个或多个氢原子,优选为最多5个,更优选为1~3个氢原子彼此独立地被相应数目的取代基取代。不言而喻,取代基仅处在它们的可能的化学位置,本领域技术人员能够在不付出过多努力的情况下确定(通过实验或理论)可能或不可能的取代。例如,具有游离氢的氨基或羟基与具有不饱和(如烯属)键的碳原子结合时可能是不稳定的。"Substituted" means that one or more hydrogen atoms, preferably up to 5, more preferably 1 to 3 hydrogen atoms in the group are replaced independently of each other by a corresponding number of substituents. It goes without saying that the substituents are only in their possible chemical positions, and the skilled person can determine (by experiment or theory) possible or impossible substitutions without undue effort. For example, amino or hydroxy groups with free hydrogens may be unstable when combined with carbon atoms with unsaturated (e.g. olefinic) bonds.
术语“药学上可接受的盐”或“可药用盐”是指本公开抗体-药物偶联物的盐,或本公开中所述的化合物的盐,这类盐用于哺乳动物体内时具有安全性和有效性,且具有应有的生物活性。本公开抗体-药物偶联物至少含有一个氨基,因此可以与酸形成盐,可药用盐的非限制性实例包括:盐酸盐。The term "pharmaceutically acceptable salt" or "pharmaceutically acceptable salt" refers to a salt of the antibody-drug conjugate of the present disclosure, or a salt of a compound described in the present disclosure, which is safe and effective when used in mammals and has the desired biological activity. The antibody-drug conjugate of the present disclosure contains at least one amino group, and thus can form a salt with an acid, and non-limiting examples of pharmaceutically acceptable salts include: hydrochloride.
术语“溶剂合物”指本公开的配体-药物偶联化合物与一种或多种溶剂分子形成可药用的溶剂合物,溶剂分子的非限制性实例包括水、乙醇、乙腈、异丙醇、DMSO、乙酸乙酯。The term "solvate" refers to a pharmaceutically acceptable solvate formed by the ligand-drug conjugate compound of the present disclosure and one or more solvent molecules. Non-limiting examples of solvent molecules include water, ethanol, acetonitrile, isopropanol, DMSO, ethyl acetate.
术语“药物载体”用于本公开的药物,是指能改变药物进入人体的方式和在体内的分布、控制药物的释放速度并将药物输送到靶向器官的体系。药物载体释放和靶向系统能够减少药物降解及损失,降低副作用,提高生物利用度。如可作为载体的高分子表面活性剂由于其独特的两亲性结构,可以进行自组装,形成各种形式的聚集体,优选的实例如胶束、微乳液、凝胶、液晶、囊泡等。这些聚集体具有包载药物分子的能力,同时又对膜有良好的渗透性,可以作为优良的药物载体。The term "drug carrier" is used for the drugs disclosed herein and refers to a system that can change the way the drug enters the human body and its distribution in the body, control the release rate of the drug, and deliver the drug to the targeted organ. The drug carrier release and targeting system can reduce drug degradation and loss, reduce side effects, and improve bioavailability. For example, polymer surfactants that can be used as carriers can self-assemble to form various forms of aggregates due to their unique amphiphilic structure, and preferred examples are micelles, microemulsions, gels, liquid crystals, vesicles, etc. These aggregates have the ability to encapsulate drug molecules and have good permeability to membranes, and can be used as excellent drug carriers.
术语“赋形剂”是在药物制剂中除主药以外的附加物,也可称为辅料。如片剂中的粘合剂、填充剂、崩解剂、润滑剂;半固体制剂软膏剂、霜剂中的基质部分;液体制剂中的防腐剂、抗氧剂、矫味剂、芳香剂、助溶剂、乳化剂、增溶剂、渗透压调节剂、着色剂等均可称为赋形剂。The term "excipient" refers to the additives in pharmaceutical preparations other than the main drug, which can also be called auxiliary materials. For example, the binders, fillers, disintegrants, and lubricants in tablets; the matrix part in semi-solid preparations such as ointments and creams; the preservatives, antioxidants, flavoring agents, aromatics, cosolvents, emulsifiers, solubilizers, osmotic pressure regulators, colorants, etc. in liquid preparations can all be called excipients.
术语“稀释剂”又称填充剂,其主要用途是增加片剂的重量和体积。稀释剂的加入不仅保证一定的体积大小,而且减少主要成分的剂量偏差,改善药物的压缩成型性等。当片剂的药物含有油性组分时,需加入吸收剂吸收油性物,使保持“干燥”状态,以利于制成片剂。The term "diluent" is also called filler, and its main purpose is to increase the weight and volume of the tablet. The addition of diluent not only ensures a certain volume, but also reduces the dosage deviation of the main ingredients, improves the compression molding of the drug, etc. When the drug in the tablet contains oily components, an absorbent needs to be added to absorb the oily substance and keep it in a "dry" state to facilitate tableting.
在本文中抗叶酸受体α(FRA)抗体或其抗原结合片段的制备方法可参见WO2017151979并将相关内容引入本文以示说明。In this article, the preparation method of anti-folate receptor alpha (FRA) antibodies or antigen-binding fragments thereof can be found in WO2017151979 and the relevant content is introduced herein for illustration.
附图说明BRIEF DESCRIPTION OF THE DRAWINGS
图1:ADC-001在T47D/A549细胞系中旁杀效应比对图。Figure 1: Comparison of the bystander effect of ADC-001 in T47D/A549 cell lines.
具体实施方式DETAILED DESCRIPTION
以下结合实施例进一步描述本公开,但这些实施例并非限制本公开的范围。The present disclosure is further described below in conjunction with embodiments, but these embodiments are not intended to limit the scope of the present disclosure.
本公开实施例中未注明具体条件的实验方法,通常按照常规条件,或按照原料或商品制造厂商所建议的条件。未注明具体来源的试剂,为市场购买的常规试剂。The experimental methods in the examples disclosed herein that do not specify specific conditions are usually carried out under conventional conditions or under conditions recommended by raw material or product manufacturers. Reagents that do not specify specific sources are conventional reagents purchased from the market.
化合物的结构是通过核磁共振(NMR)或/和质谱(MS)来确定的。NMR位移(δ)以10-6(ppm)的单位给出。NMR的测定是用Bruker AVANCE-400核磁仪,测定溶剂为氘代二甲基亚砜(DMSO-d6)、氘代氯仿(CDCl3)、氘代甲醇(CD3OD),内标为四甲基硅烷(TMS)。The structures of the compounds were determined by nuclear magnetic resonance (NMR) and/or mass spectrometry (MS). NMR shifts (δ) are given in units of 10 -6 (ppm). NMR measurements were performed using a Bruker AVANCE-400 NMR spectrometer, with deuterated dimethyl sulfoxide (DMSO-d 6 ), deuterated chloroform (CDCl 3 ), deuterated methanol (CD 3 OD) as the measuring solvent, and tetramethylsilane (TMS) as the internal standard.
MS的测定用Agilent 1200/1290DAD-6110/6120Quadrupole MS液质联用仪(生产商:Agilent,MS型号:6110/6120Quadrupole MS)。MS was measured using Agilent 1200/1290 DAD-6110/6120 Quadrupole MS liquid spectrometer-mass spectrometer (manufacturer: Agilent, MS model: 6110/6120 Quadrupole MS).
高效液相色谱法(HPLC)分析使用Agilent HPLC 1200DAD、Agilent HPLC1200VWD和Waters HPLC e2695-2489高压液相色谱仪。High performance liquid chromatography (HPLC) analysis was performed using Agilent HPLC 1200DAD, Agilent HPLC1200VWD and Waters HPLC e2695-2489.
手性HPLC分析测定使用Agilent 1260DAD高效液相色谱仪。Chiral HPLC analysis was performed using an Agilent 1260DAD high performance liquid chromatograph.
高效液相制备使用Waters 2545-2767、Waters 2767-SQ Detecor2、Shimadzu LC-20AP和Gilson GX-281制备型色谱仪。High performance liquid chromatography was performed using Waters 2545-2767, Waters 2767-SQ Detecor2, Shimadzu LC-20AP and Gilson GX-281 preparative chromatographs.
RP-HPLC分析测定使用Agilent 1260InfinityⅡ液相色谱仪,色谱柱:岛津SEC-300 3um×7.8mm×300mm,流动相:0.1%三氟醋酸的水溶液/0.1%三氟醋酸的乙腈(ACN)溶液。RP-HPLC analysis was performed using an Agilent 1260 InfinityⅡ liquid chromatograph, chromatographic column: Shimadzu SEC-300 3um×7.8mm×300mm, mobile phase: 0.1% trifluoroacetic acid in water/0.1% trifluoroacetic acid in acetonitrile (ACN) solution.
手性制备使用Shimadzu LC-20AP制备型色谱仪。Chiral preparations were performed using a Shimadzu LC-20AP preparative chromatograph.
CombiFlash快速制备仪使用Combiflash Rf200(TELEDYNE ISCO)。The CombiFlash rapid preparation instrument used Combiflash Rf200 (TELEDYNE ISCO).
薄层层析硅胶板使用烟台黄海HSGF254或青岛GF254硅胶板,薄层色谱法(TLC)使用的硅胶板采用的规格是0.15mm~0.2mm,薄层层析分离纯化产品采用的规格是0.4mm~0.5mm。The thin layer chromatography silica gel plate uses Yantai Huanghai HSGF254 or Qingdao GF254 silica gel plate. The silica gel plate used in thin layer chromatography (TLC) adopts a specification of 0.15mm-0.2mm, and the specification used for thin layer chromatography separation and purification products is 0.4mm-0.5mm.
硅胶柱色谱法一般使用烟台黄海硅胶200~300目硅胶为载体。Silica gel column chromatography generally uses Yantai Huanghai Silica Gel 200-300 mesh silica gel as the carrier.
本公开的已知的起始原料可以采用或按照本领域已知的方法来合成,或可购买自ABCR GmbH&Co.KG,Acros Organics,Aldrich Chemical Company,韶远化学科技(AccelaChemBio Inc)、达瑞化学品等公司。The known starting materials disclosed in the present invention can be synthesized by methods known in the art, or can be purchased from ABCR GmbH & Co. KG, Acros Organics, Aldrich Chemical Company, AccelaChemBio Inc, Darui Chemicals and other companies.
实施例中无特殊说明,反应能够均在氩气氛或氮气氛下进行。Unless otherwise specified in the examples, the reactions can be carried out under an argon atmosphere or a nitrogen atmosphere.
氩气氛或氮气氛是指反应瓶连接一个约1L容积的氩气或氮气气球。Argon atmosphere or nitrogen atmosphere means that the reaction bottle is connected to an argon or nitrogen balloon with a volume of about 1L.
氢气氛是指反应瓶连接一个约1L容积的氢气气球。Hydrogen atmosphere means that the reaction bottle is connected to a hydrogen balloon with a capacity of about 1L.
实施例中无特殊说明,溶液是指水溶液。Unless otherwise specified in the examples, the solution refers to an aqueous solution.
实施例中无特殊说明,反应的温度为室温,为20℃~30℃。Unless otherwise specified in the examples, the reaction temperature is room temperature, 20°C to 30°C.
实施例中的反应进程的监测采用薄层色谱法(TLC),反应所使用的展开剂,纯化化合物采用的柱层析的洗脱剂的体系和薄层色谱法的展开剂体系,溶剂的体积比根据化合物的极性不同而进行调节,也可以加入少量的三乙胺和醋酸等碱性或酸性试剂进行调节。The reaction progress in the embodiment is monitored by thin layer chromatography (TLC), the developing solvent used in the reaction, the system of column chromatography eluent used for purifying the compound and the developing solvent system for thin layer chromatography, the volume ratio of the solvent is adjusted according to the polarity of the compound, and a small amount of alkaline or acidic reagents such as triethylamine and acetic acid can be added for adjustment.
实施例1Example 1
步骤1:化合物1b的制备Step 1: Preparation of compound 1b
冰水浴下,将化合物1a(艾日布林,参照ZL201010236637.2方法制备获得)(72.91mg,0.1mmol)溶于10mL的四氢呋喃中,加入Fmoc-OSu(芴甲氧羰酰琥珀酰亚胺,41mg,0.12mmol),然后在室温搅拌至反应完全。减压浓缩得粗品,直接用于下一步反应。Under ice water bath, compound 1a (eribulin, prepared according to ZL201010236637.2) (72.91 mg, 0.1 mmol) was dissolved in 10 mL of tetrahydrofuran, Fmoc-OSu (fluorenylmethoxycarbonyl succinimide, 41 mg, 0.12 mmol) was added, and then stirred at room temperature until the reaction was complete. The crude product was concentrated under reduced pressure and used directly in the next step.
步骤2:化合物1c的制备Step 2: Preparation of compound 1c
将上步所得化合物1b的粗品溶于10mL无水乙醚中,加入氧化银(34.8mg,0.15mmol),接着加入碘甲烷(28.4mg,0.2mmol),然后于室温搅拌至反应完全,过滤,然后减压浓缩得粗品,直接进行下一步反应。The crude compound 1b obtained in the previous step was dissolved in 10 mL of anhydrous ether, and silver oxide (34.8 mg, 0.15 mmol) was added, followed by iodomethane (28.4 mg, 0.2 mmol). The mixture was stirred at room temperature until the reaction was complete, filtered, and then concentrated under reduced pressure to obtain a crude product, which was directly subjected to the next step of reaction.
步骤3:化合物1的制备Step 3: Preparation of
将上步所得化合物1c粗品溶于10mL的四氢呋喃中,加入2mL的二乙胺,然后在室温搅拌至反应完全,减压浓缩得粗品,经硅胶柱层析色谱法(洗脱剂:二氯甲烷/乙酸乙酯/石油醚)纯化,得3mg目标产物化合物1。The crude compound 1c obtained in the previous step was dissolved in 10 mL of tetrahydrofuran, and 2 mL of diethylamine was added. The mixture was stirred at room temperature until the reaction was complete, and the crude product was concentrated under reduced pressure. The crude product was purified by silica gel column chromatography (eluent: dichloromethane/ethyl acetate/petroleum ether) to obtain 3 mg of the
LC/MS(ESI):m/z 744.2[M+H]+。LC/MS(ESI): m/z 744.2[M+H] + .
冰水浴下,将4a(50mg,0.08mmol,参照WO2017151979中方法制备)溶解到1.5mL的N,N-二甲基甲酰胺中,然后加入DIPEA(N,N-二异丙基乙胺,18mg,0.14mmol),接着加入二(对硝基苯)碳酸酯(49mg,0.16mmol),然后在室温下搅拌10加入20mL甲基叔丁基醚,搅拌20分钟,过滤,干燥得到固体36mg,LC/MS(ESI):m/z 784.1[M+H]+。Under ice-water bath, 4a (50 mg, 0.08 mmol, prepared according to the method in WO2017151979) was dissolved in 1.5 mL of N,N-dimethylformamide, and then DIPEA (N,N-diisopropylethylamine, 18 mg, 0.14 mmol) was added, followed by di(p-nitrobenzene) carbonate (49 mg, 0.16 mmol). After stirring at room temperature for 10 min, 20 mL of methyl tert-butyl ether was added, stirred for 20 min, filtered, and dried to obtain 36 mg of a solid, LC/MS (ESI): m/z 784.1 [M+H] + .
将化合物1(13.5mg,0.018mmol)溶于1.5mL的DMF中,加入DIPEA(7mg,0.054mmol),然后分批加入化合物4b(18mg,1.3mmol),搅拌至反应基本完全,浓缩得粗品,经HPLC制备分离得6.5mg化合物L-1,纯度96.95%,LC/MS(ESI):m/z 1388.3[M+H]+。Compound 1 (13.5 mg, 0.018 mmol) was dissolved in 1.5 mL of DMF, DIPEA (7 mg, 0.054 mmol) was added, and then compound 4b (18 mg, 1.3 mmol) was added in batches. The mixture was stirred until the reaction was almost complete, and the crude product was concentrated. 6.5 mg of compound L-1 was obtained by HPLC separation with a purity of 96.95%. LC/MS (ESI): m/z 1388.3 [M+H] + .
实施例2Example 2
在37℃条件下,向抗叶酸受体α(FRA)抗体的PBS缓冲水溶液(pH=6.5的0.05M的PBS缓冲水溶液;10.0mg/mL,1.43mL,97nmol)加入配置好的的三(2-羧乙基)膦盐酸盐(TCEP-HCl)的水溶液(10mM,21.3uL,21.34mmol),置于水浴振荡器,于37℃下振荡反应3小时,停止反应。将反应液用水浴降温至25℃。At 37°C, add the prepared tris(2-carboxyethyl)phosphine hydrochloride (TCEP-HCl) aqueous solution (10mM, 21.3uL, 21.34mmol) to the PBS buffered aqueous solution of anti-folate receptor α (FRA) antibody (pH=6.5 0.05M PBS buffered aqueous solution; 10.0mg/mL, 1.43mL, 97nmol), place in a water bath shaker, shake at 37°C for 3 hours, and stop the reaction. Cool the reaction solution to 25°C in a water bath.
将化合物L-1(1.07mg,771nmol)溶解于50uL二甲亚砜中,加入到上述反应液中,置于水浴振荡器,于25℃下振荡反应3小时,停止反应。将反应液用Sephadex G25凝胶柱脱盐纯化(洗脱相:pH=6.5的0.05M的PBS缓冲水溶液,含0.001M的EDTA),得到标题产物的PBS缓冲液(1.21mg/mL,10.8mL),于4℃冷藏储存。RP-HPLC检测方法计算平均DAR值:n=4。Compound L-1 (1.07 mg, 771 nmol) was dissolved in 50 uL dimethyl sulfoxide, added to the above reaction solution, placed in a water bath oscillator, and oscillated at 25°C for 3 hours to stop the reaction. The reaction solution was desalted and purified using a Sephadex G25 gel column (elution phase: 0.05 M PBS buffer solution at pH = 6.5, containing 0.001 M EDTA) to obtain the title product in PBS buffer (1.21 mg/mL, 10.8 mL), which was stored in a refrigerator at 4°C. The average DAR value was calculated by the RP-HPLC detection method: n = 4.
对比例1Comparative Example 1
参照CN201780022381.5方法制备获得,其中,RP-HPLC检测方法计算平均DAR值:n=4。Prepared according to the method of CN201780022381.5, wherein the average DAR value was calculated by the RP-HPLC detection method: n=4.
测试例1:化合物1的体外细胞毒活性筛选Test Example 1: In vitro cytotoxic activity screening of
1.1.实验原理及方法1.1. Experimental principles and methods
本实验利用CTG检测ATP含量,反映肿瘤细胞的存活情况。首先通过种植不同密度的细胞并培养3天和5天,根据IC50和最大抑制率确定最终培养条件。然后按照此条件检测毒素分子的杀伤作用。This experiment uses CTG to detect ATP content, reflecting the survival of tumor cells. First, by planting cells at different densities and culturing for 3 and 5 days, the final culture conditions are determined based on IC 50 and maximum inhibition rate. Then, the killing effect of the toxin molecules is tested under these conditions.
1.2.细胞株的选择1.2. Cell line selection
根据实验目的,选择乳腺癌、NSCLC两种疾病模型,选出SKBR3肿瘤细胞(HER2+,ATCC,货号HTB-30)、MDA-MB-468(HER2-,ATCC,货号HTB-132)和A549(人非小细胞肺癌细胞,ATCC,货号CCL-185)三株细胞用于筛选实验。According to the purpose of the experiment, two disease models of breast cancer and NSCLC were selected, and three cell lines, SKBR3 tumor cells (HER2+, ATCC, catalog number HTB-30), MDA-MB-468 (HER2-, ATCC, catalog number HTB-132) and A549 (human non-small cell lung cancer cells, ATCC, catalog number CCL-185), were selected for screening experiments.
1.3.细胞培养条件的确定1.3. Determination of cell culture conditions
1)细胞培养:A549、SK-BR-3和MDA-MB-468细胞分别用含10%FBS(Gibco,10099-141)的Ham’s F-12K(Kaighn’s)培养基(Gibco,21127030)和McCoy's 5A培养基(ThermoFisher,货号16600108)培养及Leibovitz's L-15培养基(ThermoFisher,货号11415-114)进行培养。1) Cell culture: A549, SK-BR-3 and MDA-MB-468 cells were cultured with Ham's F-12K (Kaighn's) medium (Gibco, 21127030) and McCoy's 5A medium (ThermoFisher, Catalog No. 16600108) containing 10% FBS (Gibco, 10099-141) and Leibovitz's L-15 medium (ThermoFisher, Catalog No. 11415-114), respectively.
2)细胞铺板:将A549用胰酶消化后,用上述培养基终止,计数后分别取4.3×105、7.2×105、11.5×105个细胞加培养液使终体积均为26ml。在96孔板(康宁,货号3903)第2列到第11列每孔加180ul细胞悬液,使得细胞密度分别为每孔3K、5K、8K。第12列为200ul培养液,剩余孔用PBS填充。SKBR3和MDA-MB-468细胞重复上述操作。平行两份。2) Cell plating: After A549 was digested with trypsin, it was terminated with the above culture medium. After counting, 4.3×10 5 , 7.2×10 5 , and 11.5×10 5 cells were taken and culture medium was added to make the final volume 26 ml. 180ul of cell suspension was added to each well of the 2nd to 11th columns of a 96-well plate (Corning, Cat. No. 3903), so that the cell density was 3K, 5K, and 8K per well, respectively. 200ul of culture medium was added to the 12th column, and the remaining wells were filled with PBS. Repeat the above operation for SKBR3 and MDA-MB-468 cells. Two parallel copies were made.
2)药物配制:在圆底96孔板(康宁,货号3788)中,用DMSO配制阳性对照艾日布林和本公开化合物储备液。在配药板1的第1列配制2mM(用DMSO将储液稀释10倍),此后到第10列梯度10倍稀释于DMSO中,第11列为DMSO。配药板2第2列至第11列每孔加入相应培养液95ul,从配药板1的第2列至第11列吸取5ul溶液至配药板2,混匀后,吸取20ul加入铺好的细胞中,继续培养3天和5天。2) Drug preparation: In a round-bottom 96-well plate (Corning, catalog number 3788), the positive control eribulin and the stock solution of the disclosed compound were prepared with DMSO. 2mM was prepared in the first column of the preparation plate 1 (the stock solution was diluted 10 times with DMSO), and then the gradient was diluted 10 times in DMSO to the 10th column, and the 11th column was DMSO. 95ul of the corresponding culture solution was added to each well of the 2nd to 11th columns of the preparation plate 2, and 5ul of the solution was drawn from the 2nd to 11th columns of the
3)CTG检测(Cell Titer-GloTM,发光细胞生存能力检测,Promega公司):分别在第3天和第5天取出细胞板,平衡至室温。每孔加入90ul CTG,避光室温反应10min,酶标仪读取发光值并计算IC50。3) CTG assay (Cell Titer-GloTM, luminescent cell viability assay, Promega): The cell plates were removed on the 3rd and 5th days, and equilibrated to room temperature. 90ul CTG was added to each well, and the cells were reacted at room temperature in the dark for 10 minutes. The luminescence value was read by a microplate reader and the IC 50 was calculated.
1.4.数据结果1.4. Data Results
表1Table 1
测试例2:化合物1药代动力学测试Test Example 2: Pharmacokinetic Test of
1、概述1. Overview
以Non-的比格犬为受试动物,应用LC/MS/MS法测定了比格犬静脉注射给予化合物D-1和艾日布林后不同时刻血浆中的药物浓度。研究本公开化合物在犬体内的药代动力学行为,评价其药动学特征。Non- Beagles were used as test animals, and the drug concentrations in the plasma of beagles at different times after intravenous injection of compound D-1 and eribulin were determined by LC/MS/MS. The pharmacokinetic behavior of the disclosed compound in dogs was studied, and its pharmacokinetic characteristics were evaluated.
2、试验方案2. Experimental plan
2.1试验药品2.1 Trial Drugs
化合物1和艾日布林
2.2试验动物2.2 Experimental animals
比格犬6只,雄性,平均分为2组,由美迪西普亚医药科技(上海)有限公司采购并进行动物给药实验。Six male beagle dogs were equally divided into two groups and purchased by Medicipia Pharmaceutical Technology (Shanghai) Co., Ltd. for animal drug administration experiments.
2.3药物配制2.3 Drug preparation
称取化合物D-1,加5%体积的DMSO,20%PG和20%PEG400使其溶解,然后加入55%生理盐水配制成0.25mg/ml无色澄明溶液。Compound D-1 was weighed, and 5% volume of DMSO, 20% PG and 20% PEG400 were added to dissolve it, and then 55% physiological saline was added to prepare a 0.25 mg/ml colorless clear solution.
称取艾日布林,加5%体积的DMSO,20%PG和20%PEG400使其溶解,然后加入55%生理盐水配制成0.25mg/ml无色澄明溶液。Eribulin was weighed, and 5% volume of DMSO, 20% PG and 20% PEG400 were added to dissolve it, and then 55% physiological saline was added to prepare a 0.25 mg/ml colorless clear solution.
2.4给药2.4 Administration
一组犬静脉注射给药化合物D-1,给药剂量均为0.5mg/kg,给药体积均为2ml/kg。A group of dogs were intravenously administered with compound D-1, the dosage was 0.5 mg/kg, and the administration volume was 2 ml/kg.
另一组犬静脉注射给药艾日布林,给药剂量均为0.5mg/kg,给药体积均为2ml/kg。Another group of dogs were given eribulin intravenously, with the dose of 0.5 mg/kg and the administration volume of 2 ml/kg.
3、操作3. Operation
犬注射给药化合物D-1,于给药前及给药后5分钟、0.25、0.5、1.0、2.0、4.0、8.0、12.0、24.0小时采血1ml,将收集的血液样本置于EDTA-K2抗凝型采血管中,将收集到的全血放在冰上,1小时内离心分离血浆(离心力2200g,离心10min,2-8℃)。血浆样本在检测前存放于-80℃冰箱内。Dogs were injected with compound D-1, and 1 ml of blood was collected before administration and at 5 minutes, 0.25, 0.5, 1.0, 2.0, 4.0, 8.0, 12.0, and 24.0 hours after administration. The collected blood samples were placed in EDTA-K2 anticoagulant blood collection tubes, and the collected whole blood was placed on ice and centrifuged within 1 hour to separate plasma (centrifugal force 2200g, centrifugation 10min, 2-8°C). The plasma samples were stored in a -80°C refrigerator before testing.
犬注射给药艾日布林化合物,于给药前及给药后5分钟、0.25、0.5、1.0、2.0、4.0、8.0、12.0、24.0小时采血1ml,将收集的血液样本置于EDTA-K2抗凝型采血管中,将收集到的全血放在冰上,1小时内离心分离血浆(离心力2200g,离心10min,2-8℃)。血浆样本在检测前存放于-80℃冰箱内。Dogs were injected with eribulin compounds, and 1 ml of blood was collected before administration and 5 minutes, 0.25, 0.5, 1.0, 2.0, 4.0, 8.0, 12.0, and 24.0 hours after administration. The collected blood samples were placed in EDTA-K2 anticoagulant blood collection tubes, and the collected whole blood was placed on ice and centrifuged to separate plasma within 1 hour (centrifugal force 2200g, centrifugation 10min, 2-8°C). Plasma samples were stored in a -80°C refrigerator before testing.
测定药物注射给药后犬血浆中的待测化合物含量:取给药后各时刻的犬血浆25μl,加入内标溶液喜树碱(中国生物制品检定所)50μl(100ng/mL)和乙腈200μl,涡旋混合5分钟,离心10分钟(3700转/分钟),血浆样品取上清液3-4μl进行LC/MS/MS(API4000三重四极杆串联质谱仪(No.2),美国Applied Biosystems公司;Shimadzu LC-30AD超高效液相色谱系统,日本Shimadzu公司)分析。Determination of the content of the test compound in dog plasma after drug injection: Take 25 μl of dog plasma at each time after administration, add 50 μl (100 ng/mL) of internal standard solution camptothecin (China National Institute for the Control of Biological Products) and 200 μl of acetonitrile, vortex mix for 5 minutes, centrifuge for 10 minutes (3700 rpm), and take 3-4 μl of the supernatant of the plasma sample for LC/MS/MS (API4000 triple quadrupole tandem mass spectrometer (No. 2), Applied Biosystems, USA; Shimadzu LC-30AD ultra high performance liquid chromatography system, Shimadzu, Japan) analysis.
4、药代动力学参数结果4. Pharmacokinetic parameter results
本公开化合物的药代动力学参数如下表2所示。The pharmacokinetic parameters of the disclosed compounds are shown in Table 2 below.
表2Table 2
测试例3:细胞增殖抑制活性Test Example 3: Cell proliferation inhibitory activity
1、供试样品1. Test samples
化合物1和艾日布林
2、实验方法2. Experimental methods
1)将细胞用胰酶消化后,计数,离心;1) Digest the cells with trypsin, count them, and centrifuge them;
2)按照需求调整细胞密度,SK-OV-3,Cal-51细胞用5%FBS的培养基混匀细胞并调整细胞密度,OVCAR3和T47D用10%的培养基混匀细胞并调整细胞密度;2) Adjust the cell density as needed. For SK-OV-3 and Cal-51 cells, mix the cells with 5% FBS medium and adjust the cell density. For OVCAR3 and T47D cells, mix the cells with 10% FBS medium and adjust the cell density.
3)将细胞按照75μL/孔接种到96孔板中,SK-OV-3,Cal-51细胞每孔接种750个细胞,OVCAR3和T47D细胞每孔接种1000个细胞,过夜,用2x药物稀释;3) Inoculate cells into 96-well plates at 75 μL/well. 750 cells per well for SK-OV-3 and Cal-51 cells, and 1000 cells per well for OVCAR3 and T47D cells, overnight, and dilute with 2x drug.
4)再在37℃孵育72h,更换新鲜培养基130μL,加入20μL 7.5x浓度药物,37℃孵育48h;4) Incubate at 37°C for another 72 h, replace with 130 μL of fresh culture medium, add 20 μL of 7.5x concentration drug, and incubate at 37°C for 48 h;
5)将细胞板与CellTiter Glo试剂平衡至室温;5) Equilibrate the cell plate and CellTiter Glo reagent to room temperature;
6)每个孔加入50μL CellTiter Glo试剂,室温震荡混匀10min。将板子置于多功能酶标仪上读取化学发光值。6) Add 50 μL of CellTiter Glo reagent to each well and mix at room temperature for 10 min. Place the plate on a multifunctional microplate reader to read the chemiluminescence value.
3、实验结果3. Experimental results
表3Table 3
测试例4:抗体依赖的细胞介导的细胞毒性作用Test Example 4: Antibody-dependent cell-mediated cytotoxicity
1、供试样品1. Test samples
抗体偶联物ADC-001和ADC-002;Antibody conjugates ADC-001 and ADC-002;
Farletuzumab来自上海迈晋生物医药科技有限公司。Farletuzumab comes from Shanghai Maijin Biopharmaceutical Technology Co., Ltd.
2、实验方法2. Experimental methods
1)用胰酶将Cal-51细胞消化成单个后离心;1) Cal-51 cells were digested into single cells using trypsin and centrifuged;
2)用培养基将细胞密度调整为1×105cells/mL,按照100μL/孔接种到96孔板,过夜,用含有20ng/mL IL-2的RPMI 1640培养基配置抗体,对应孔每孔加入50μL,每个浓度2个复孔;2) Adjust the cell density to 1×10 5 cells/mL with culture medium, inoculate into 96-well plates at 100 μL/well, incubate overnight, prepare antibodies with RPMI 1640 culture medium containing 20 ng/mL IL-2, add 50 μL to each well, and replicate for each concentration;
3)取出NK92-CD16A-176V细胞,用含有20ng/mL IL-2的RPMI 1640培养基将细胞密度调整为1×106cells/mL;3) Remove NK92-CD16A-176V cells and adjust the cell density to 1×10 6 cells/mL using RPMI 1640 medium containing 20 ng/mL IL-2;
4)将NK92-CD16A-176V细胞按照50μL/well接种到对应孔中,将细胞放置培养箱共孵育4h;4) NK92-CD16A-176V cells were seeded into the corresponding wells at 50 μL/well and the cells were placed in an incubator for 4 h;
5)取出板子,每孔取出100μL培养上清后平衡至室温;5) Take out the plate, take out 100 μL of culture supernatant from each well and equilibrate to room temperature;
6)每孔加入50μL CTG,室温震荡10min后,检测化学发光。6) Add 50 μL CTG to each well, shake at room temperature for 10 min, and then detect chemiluminescence.
计算特异性杀伤比例,计算公式:特异性杀伤%=(靶细胞+效应细胞对照组-靶细胞+效应细胞处理组)/靶细胞*100Calculate the specific killing ratio, the calculation formula is: specific killing % = (target cells + effector cell control group - target cells + effector cell treatment group) / target cells * 100
3、实验结果3. Experimental results
表4Table 4
测试例5:旁杀伤效应Test Case 5: Bystander Effect
1、供试样品1. Test samples
抗体偶联物ADC-001和ADC-002Antibody conjugates ADC-001 and ADC-002
2、实验方法2. Experimental methods
1)用胰酶将T47D和A549消化成单个后离心;1) Digest T47D and A549 into single cells with trypsin and centrifuge;
2)将细胞密度调整为1×106cells/mL,分别用CellTraceTM Violet(CTV)和CellTraceTM CFSE(CFSE)标记T47D和A549;2) The cell density was adjusted to 1×10 6 cells/mL, and T47D and A549 were labeled with CellTrace TM Violet (CTV) and CellTrace TM CFSE (CFSE), respectively;
3)标记后在24孔板中每孔接种6.5×104cells T47D和1.3×104cells A549,体系为500μL,每个浓度3个复孔,过夜;3) After labeling, 6.5×10 4 cells T47D and 1.3×10 4 cells A549 were inoculated into each well of a 24-well plate in 500 μL, with 3 replicates for each concentration, overnight;
4)每孔加入500μL2x含药培养基(终浓度20nM),处理48h;4) Add 500 μL of 2x drug-containing culture medium (final concentration 20 nM) to each well and treat for 48 h;
5)用胰酶消化并收集细胞,流式细胞仪分析CTV或CFSE阳性的细胞数目。5) The cells were digested with trypsin and collected, and the number of CTV or CFSE-positive cells was analyzed by flow cytometry.
4、实验结果4. Experimental results
实验结果表明,叶酸受体阴性的细胞A549在单独培养时,对ADC-001和ADC-002不敏感,当与叶酸受体阳性的细胞T47D共同培养时,由于受到旁杀伤作用,A549细胞对于ADC-001和ADC-002相对更敏感。The experimental results showed that folate receptor-negative cells A549 were insensitive to ADC-001 and ADC-002 when cultured alone. However, when co-cultured with folate receptor-positive cells T47D, A549 cells became more sensitive to ADC-001 and ADC-002 due to bystander killing.
测试例6:在Cal-51模型上的体内抗肿瘤抑制作用Test Example 6: In vivo anti-tumor inhibitory effect on Cal-51 model
1、供试样品1. Test samples
抗体偶联物ADC-001和ADC-002Antibody conjugates ADC-001 and ADC-002
2、实验方法2. Experimental methods
2.1实验动物2.1 Experimental animals
BALB/c-裸小鼠,6周龄,雌性,上海市计划生育科学研究所实验动物经营部。BALB/c-nude mice, 6 weeks old, female, obtained from the Experimental Animal Management Department of Shanghai Institute of Family Planning Sciences.
2.2实验步骤2.2 Experimental procedures
每只裸小鼠皮下接种5x106 Cal-51细胞(来自ATCC),待瘤体积达到150-200mm3后,根据肿瘤体积将动物分组,小鼠静脉注射(IV)给药,具体给药剂量和给药方案见表5。每周测2次肿瘤体积,称小鼠体重,记录数据。Each nude mouse was subcutaneously inoculated with 5x10 6 Cal-51 cells (from ATCC). When the tumor volume reached 150-200 mm 3 , the animals were divided into groups according to the tumor volume and the mice were intravenously injected (IV). The specific dosage and dosing schedule are shown in Table 5. The tumor volume was measured twice a week, the mice were weighed, and the data were recorded.
表5给药方案Table 5 Dosage regimen
2.3实验指标2.3 Experimental indicators
实验指标为考察药物对肿瘤生长的影响,具体指标为T/C%或抑瘤率TGI(%)。The experimental index is to examine the effect of the drug on tumor growth, and the specific index is T/C% or tumor inhibition rate TGI (%).
每周二次用游标卡尺测量肿瘤直径,肿瘤体积(V)计算公式为:Tumor diameter was measured with a vernier caliper twice a week, and tumor volume (V) was calculated as:
V=1/2×a×b2其中a、b分别表示长、宽。V = 1/2 × a × b 2 where a and b represent length and width respectively.
T/C(%)=(T-T0)/(C-C0)×100其中T、C为实验结束时的肿瘤体积;T0、C0为实验开始时的肿瘤体积。T/C (%) = (T-T0)/(C-C0) × 100, where T and C are the tumor volumes at the end of the experiment; T0 and C0 are the tumor volumes at the beginning of the experiment.
抑瘤率(TGI)(%)=100-T/C(%)。Tumor inhibition rate (TGI) (%) = 100-T/C (%).
当肿瘤出现消退时,抑瘤率(TGI)(%)=100-(T-T0)/T0×100When the tumor regresses, tumor inhibition rate (TGI) (%) = 100-(T-T0)/T0×100
如果肿瘤比起始体积缩小,即T<T0或C<C0时,即定义为肿瘤部分消退(PR);如果肿瘤完全消失,即定义为肿瘤完全消退(CR)。If the tumor is smaller than its initial volume, that is, T<T0 or C<C0, it is defined as partial tumor regression (PR); if the tumor disappears completely, it is defined as complete tumor regression (CR).
实验结束、达到实验终点、或溶剂组平均肿瘤体积达到1500mm3,CO2麻醉处死动物,随后解剖取瘤并拍照。At the end of the experiment, when the experimental endpoint was reached, or when the average tumor volume of the solvent group reached 1500 mm 3 , the animals were killed by CO 2 anesthesia, and then the tumors were dissected and photographed.
2.4统计学分析2.4 Statistical analysis
除非特别说明,两组肿瘤体积之间比较采用双尾学生t检验,P<0.05定义为有统计学显著性差异。Unless otherwise specified, the tumor volumes between the two groups were compared using the two-tailed Student's t test, and P < 0.05 was defined as a statistically significant difference.
3实验结果3 Experimental results
表6Table 6
在1.5cmpk时,抗体-药物偶联物ADC-001和ADC-002(阳性对照组)对人乳腺癌细胞抑瘤率分别为47%和0,ADC-001抗肿瘤活性优于阳性对照ADC-002,在5mpk时,抗体-药物偶联物ADC-001和ADC-002(阳性对照组)具有几乎相当的抗肿瘤活性。说明ADC-001药物起效浓度更低。同时荷瘤小鼠对以上药物均能很好耐受,体重没有明显下降。At 1.5 cmpk, the tumor inhibition rates of antibody-drug conjugates ADC-001 and ADC-002 (positive control group) on human breast cancer cells were 47% and 0, respectively. The anti-tumor activity of ADC-001 was better than that of the positive control ADC-002. At 5 mpk, the antibody-drug conjugates ADC-001 and ADC-002 (positive control group) had almost equivalent anti-tumor activity. This indicates that the effective concentration of ADC-001 is lower. At the same time, tumor-bearing mice can tolerate the above drugs well, and there is no significant weight loss.
Claims (24)
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110829951X | 2021-07-22 | ||
| CN202110829951 | 2021-07-22 | ||
| CN202111570686 | 2021-12-21 | ||
| CN2021115706864 | 2021-12-21 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN115845080A true CN115845080A (en) | 2023-03-28 |
Family
ID=85660349
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202210867359.3A Pending CN115845080A (en) | 2021-07-22 | 2022-07-22 | Eribulin derivative-anti-folate receptor antibody conjugate |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN115845080A (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2024199442A1 (en) * | 2023-03-30 | 2024-10-03 | Genequantum Healthcare (Suzhou) Co., Ltd. | Disaccharide linker, linker-payload containing the same and glycan chain-remodeled antibody-drug conjugate, preparation method and uses thereof |
| WO2025176180A1 (en) * | 2024-02-23 | 2025-08-28 | 江苏恒瑞医药股份有限公司 | Use of eribulin derivative drug conjugate in treatment of tumor |
-
2022
- 2022-07-22 CN CN202210867359.3A patent/CN115845080A/en active Pending
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2024199442A1 (en) * | 2023-03-30 | 2024-10-03 | Genequantum Healthcare (Suzhou) Co., Ltd. | Disaccharide linker, linker-payload containing the same and glycan chain-remodeled antibody-drug conjugate, preparation method and uses thereof |
| WO2025176180A1 (en) * | 2024-02-23 | 2025-08-28 | 江苏恒瑞医药股份有限公司 | Use of eribulin derivative drug conjugate in treatment of tumor |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US12377163B2 (en) | Anti-B7H3 antibody-exatecan analog conjugate and medicinal use thereof | |
| CN114845739B (en) | Drug conjugate of eribulin derivative, preparation method thereof and application thereof in medicine | |
| WO2021052402A1 (en) | Camptothecin derivative and conjugate thereof | |
| CN106687480A (en) | Anti-c-Met antibody and anti-c-Met antibody-cytotoxic drug conjugate and medical use thereof | |
| CN118403182A (en) | Anti-claudin antibody drug conjugate and medical application thereof | |
| CN110483639A (en) | Target antibody and the antibody-drug conjugates and its preparation method and application of AXL | |
| CN115298186B (en) | Anti-PSMA antibody-exitecan analog conjugate and its medical use | |
| CN113121639B (en) | Aureostatin analogue and conjugate thereof, preparation method and application thereof | |
| WO2018068758A1 (en) | Medical use of anti-c met antibody-cytotoxic drug conjugate | |
| CN115845080A (en) | Eribulin derivative-anti-folate receptor antibody conjugate | |
| US20240382607A1 (en) | Drug conjugate of eribulin derivative | |
| CN114729035B (en) | Anti-CEA antibody-exitecan analog conjugate and its medical use | |
| CN114652853B (en) | Anti-IL-4R antibody-drug conjugate and medical application thereof | |
| CN118079013A (en) | Condensed ring compound, conjugate and application thereof | |
| WO2024230742A1 (en) | Antibody or antigen-binding fragment thereof, antibody-drug conjugate, and use thereof | |
| CN120239617A (en) | Condensed ring compounds and conjugates thereof and uses thereof | |
| RU2830242C1 (en) | Eribulin derivative drug conjugate, method for preparation thereof and use thereof in medicine | |
| HK40082496B (en) | Anti-psma antibody-exatecan analogue conjugate and medical use thereof | |
| HK40082496A (en) | Anti-psma antibody-exatecan analogue conjugate and medical use thereof | |
| HK40047594B (en) | Anti-b7h3 antibody-exatecan analog conjugate and medicinal use thereof | |
| HK1232895B (en) | Anti-c-met antibody and anti-c-met antibody-cytotoxic drug conjugate and pharmaceutical use thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination |