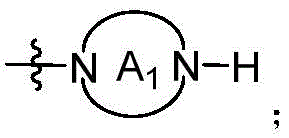
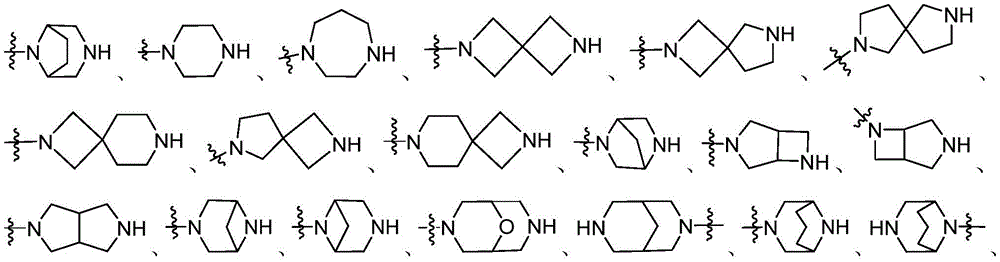
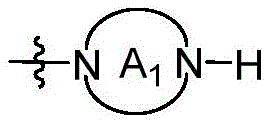
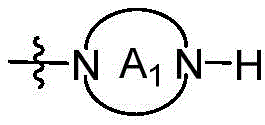
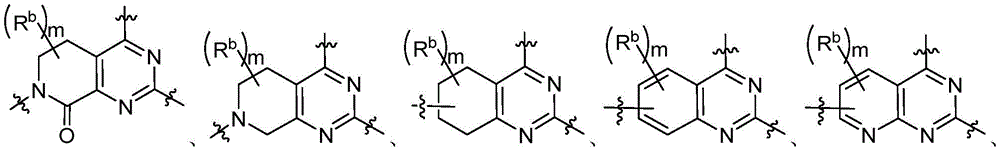
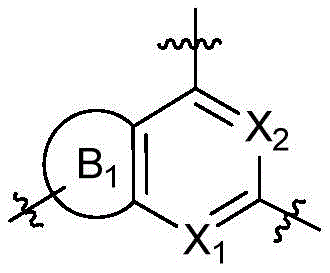
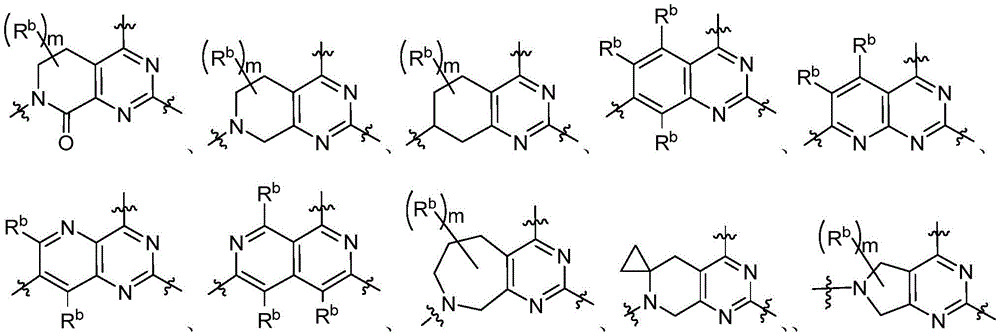
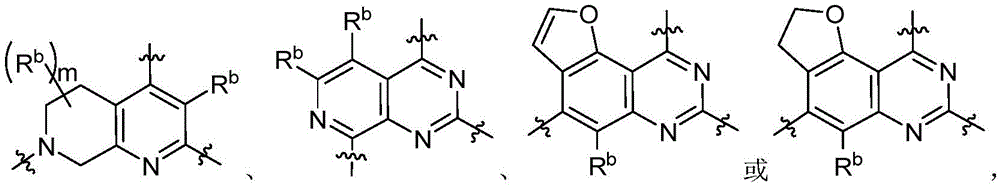
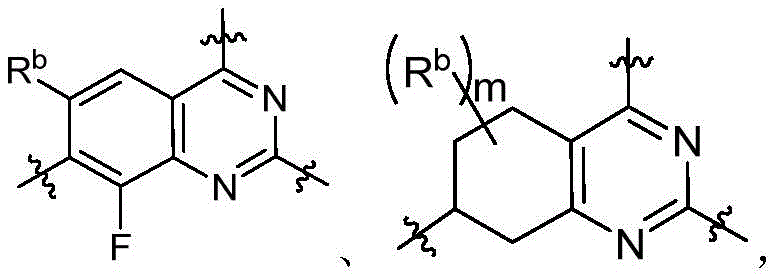
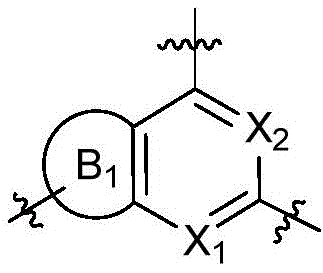
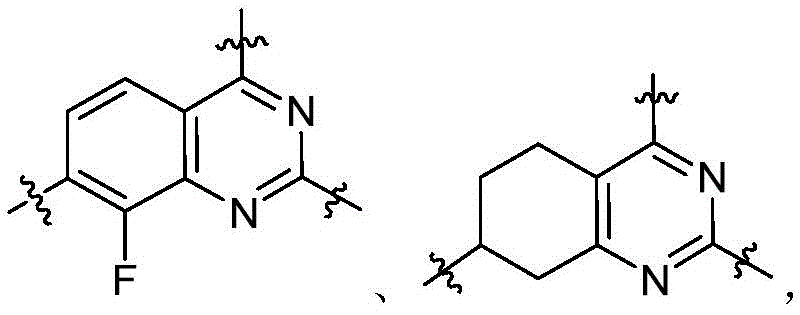
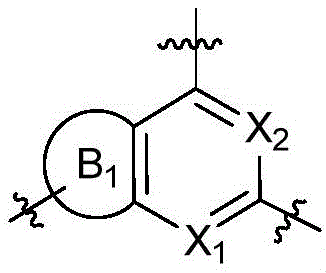
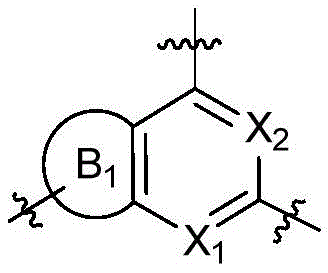
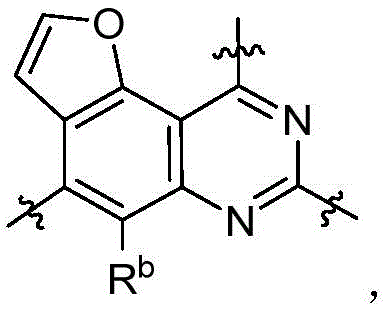
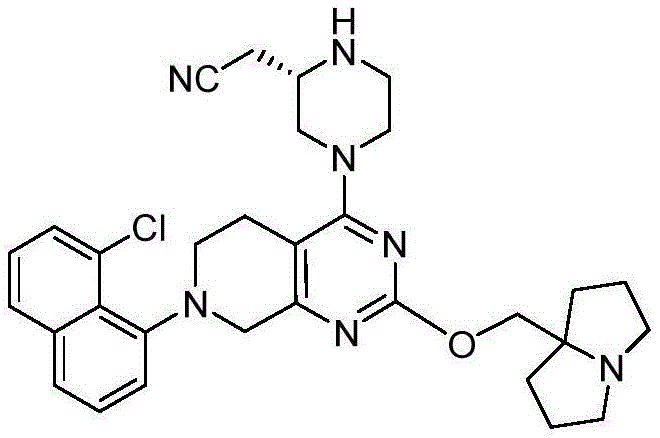
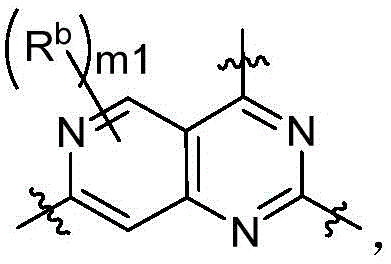
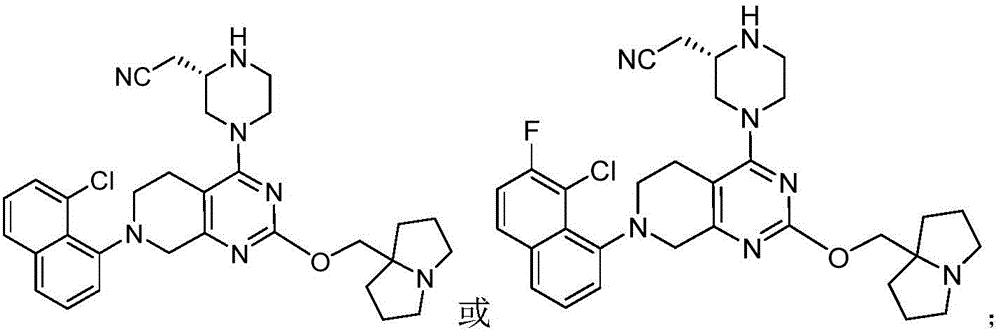
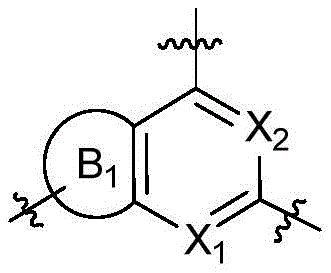
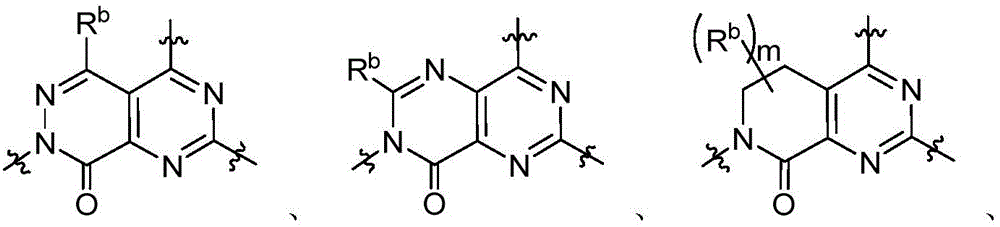
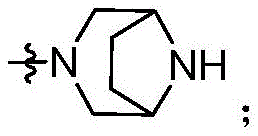
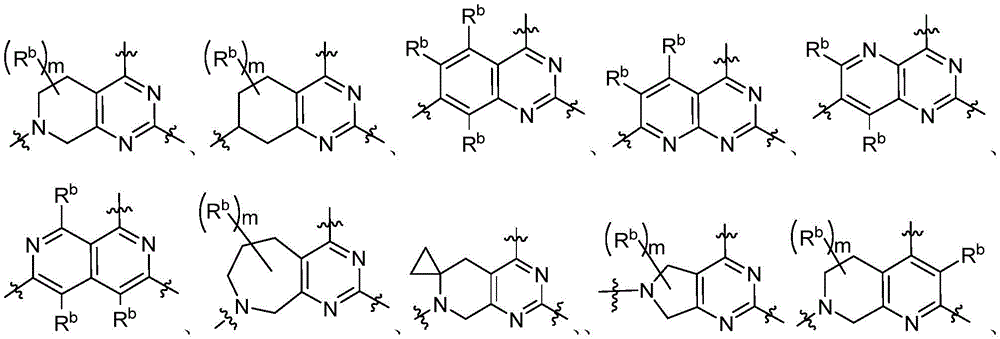
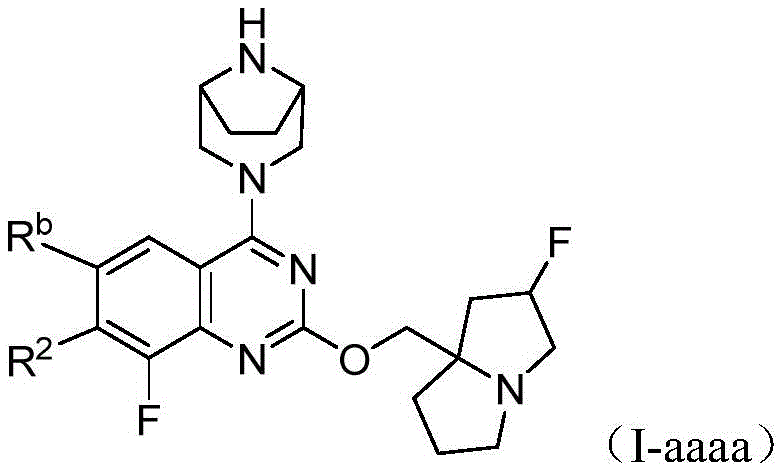
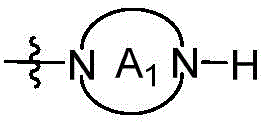
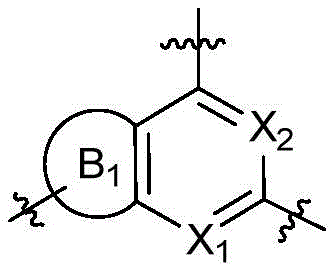
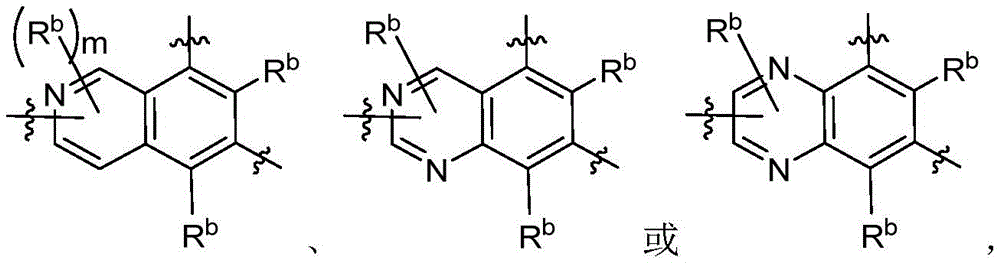
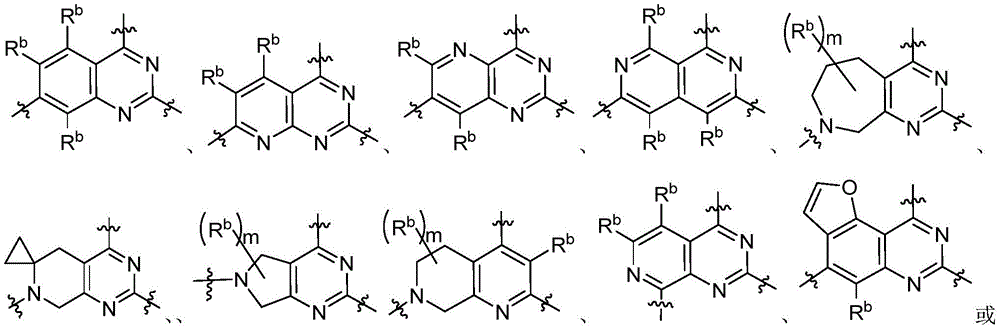
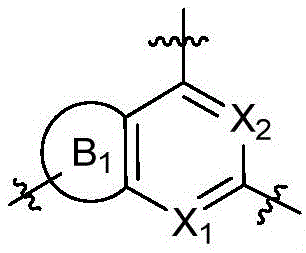
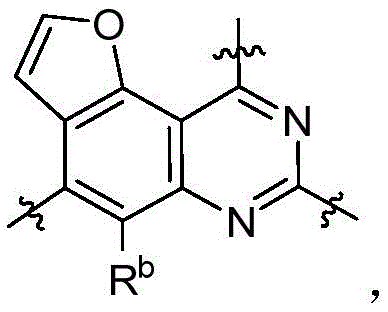
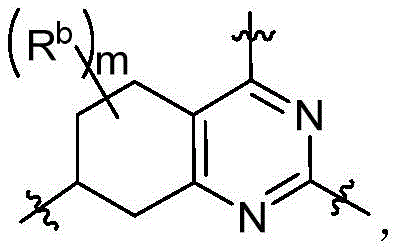
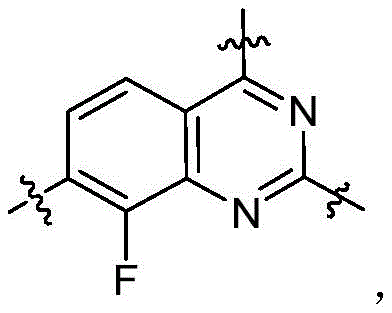
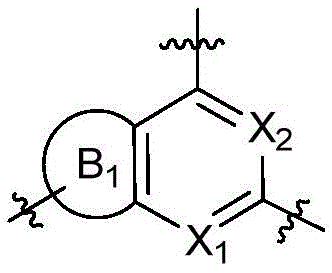
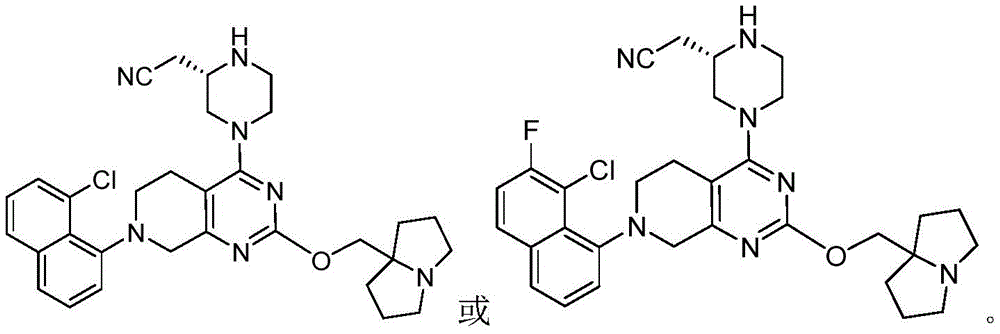
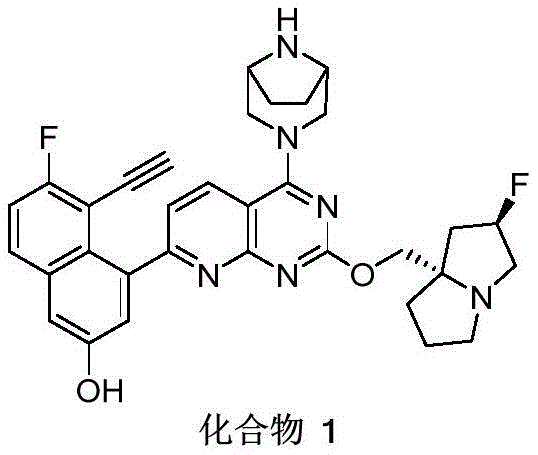
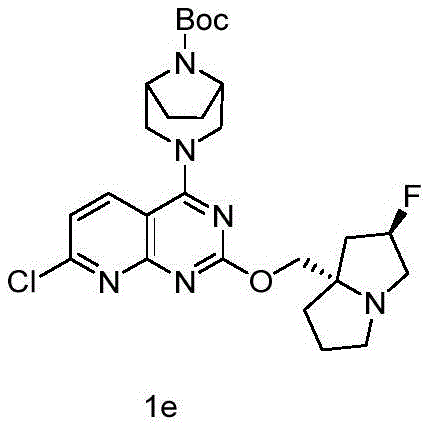
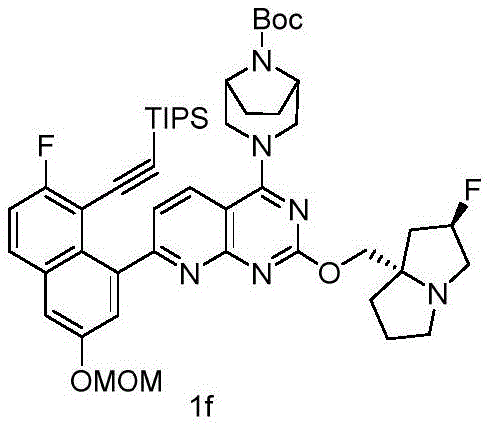
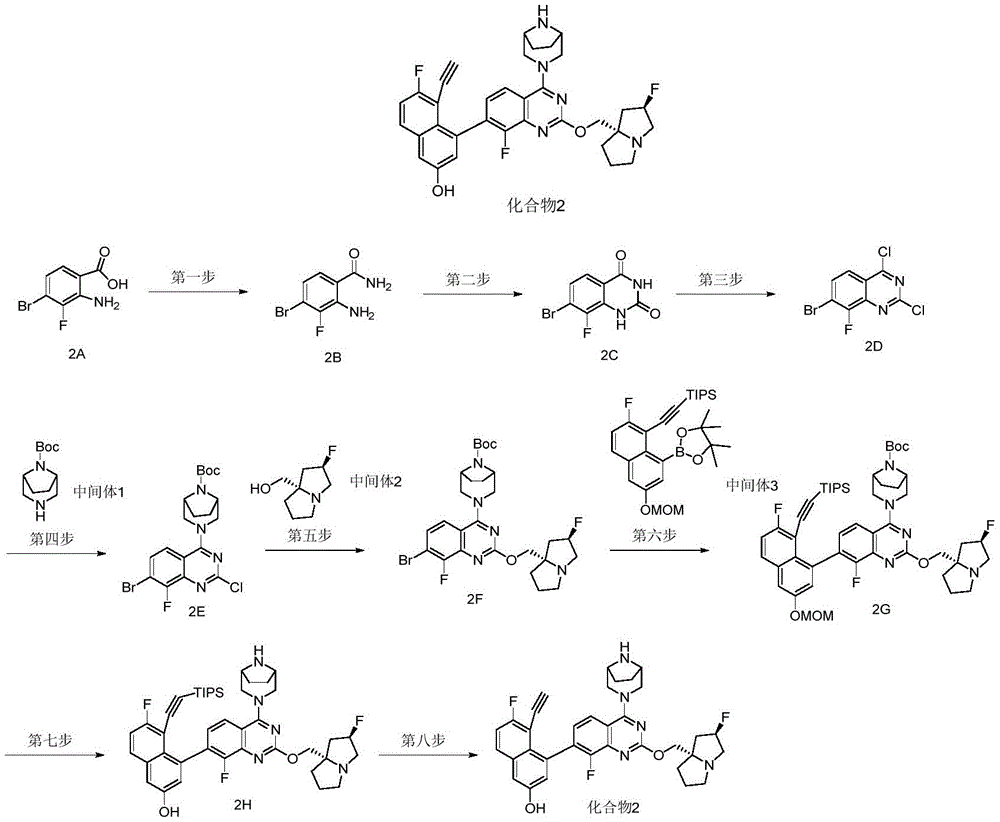
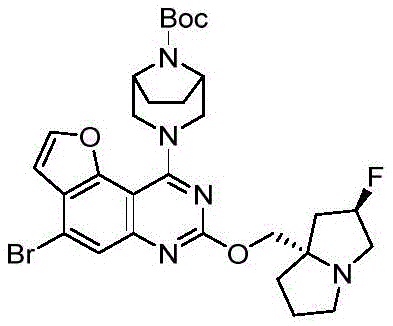
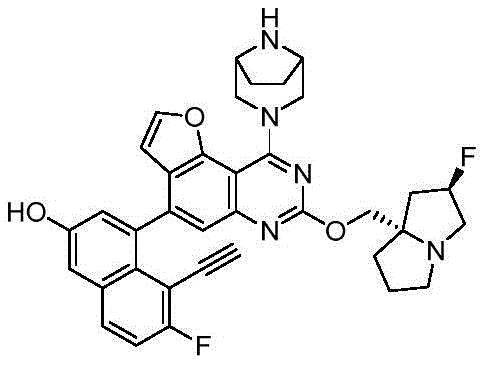
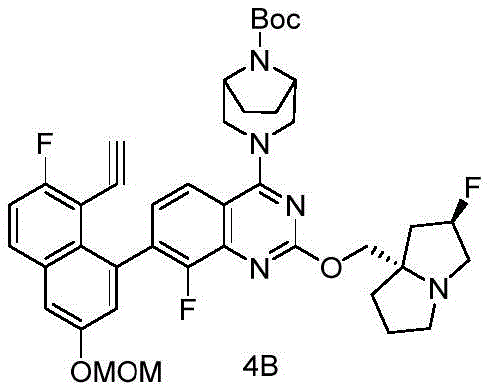
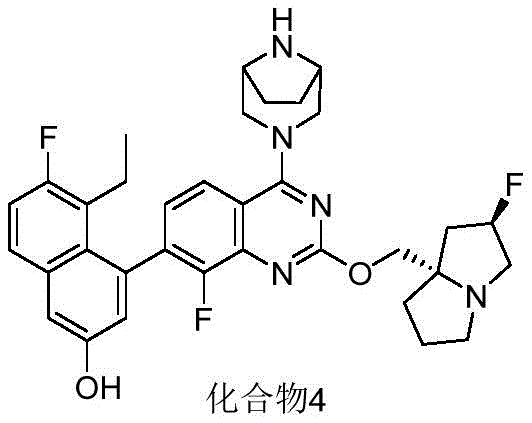
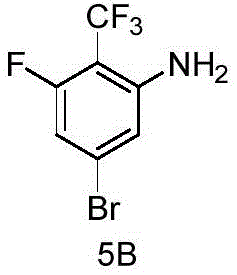
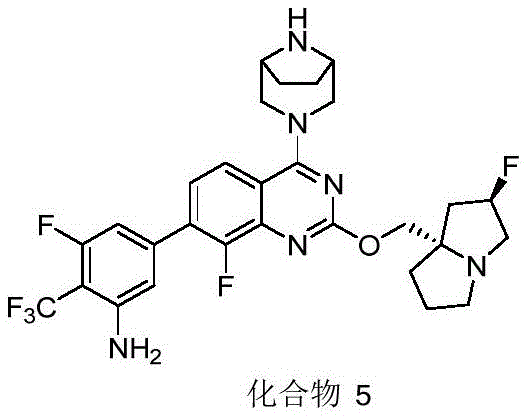
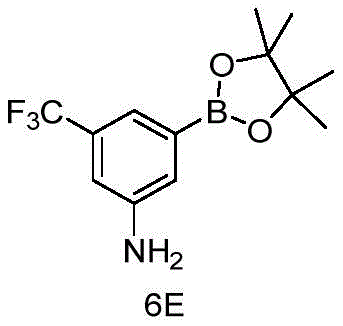
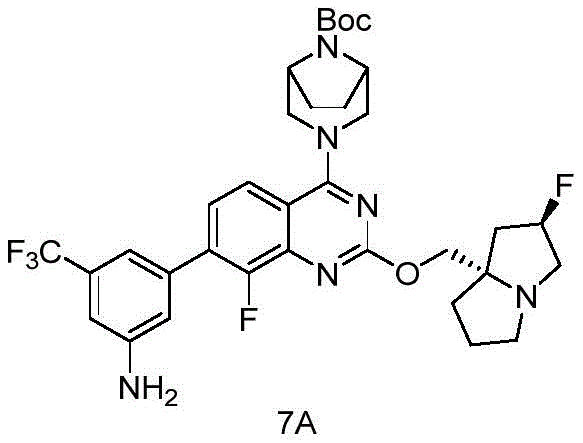
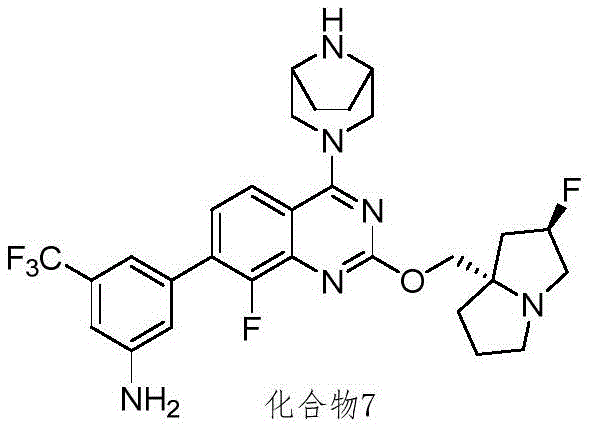
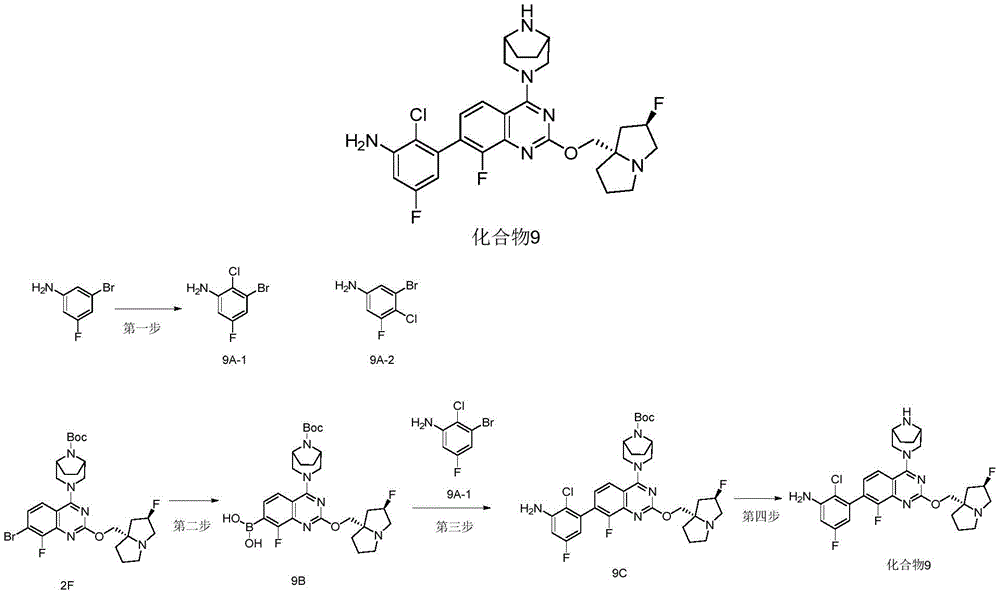
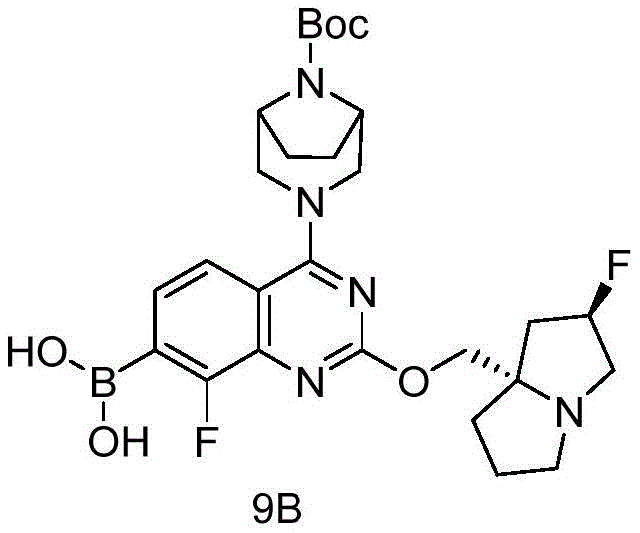
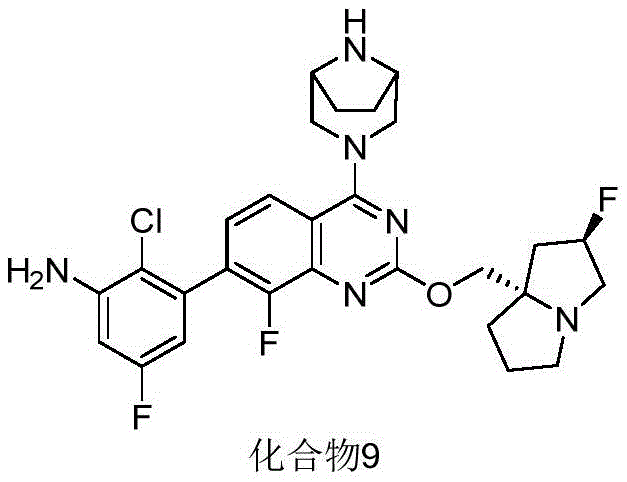
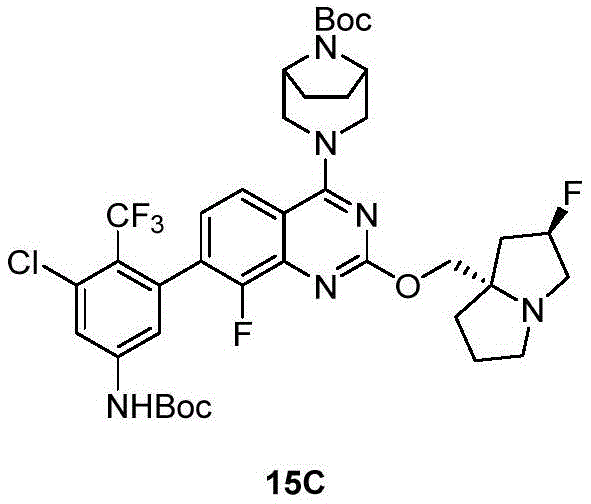
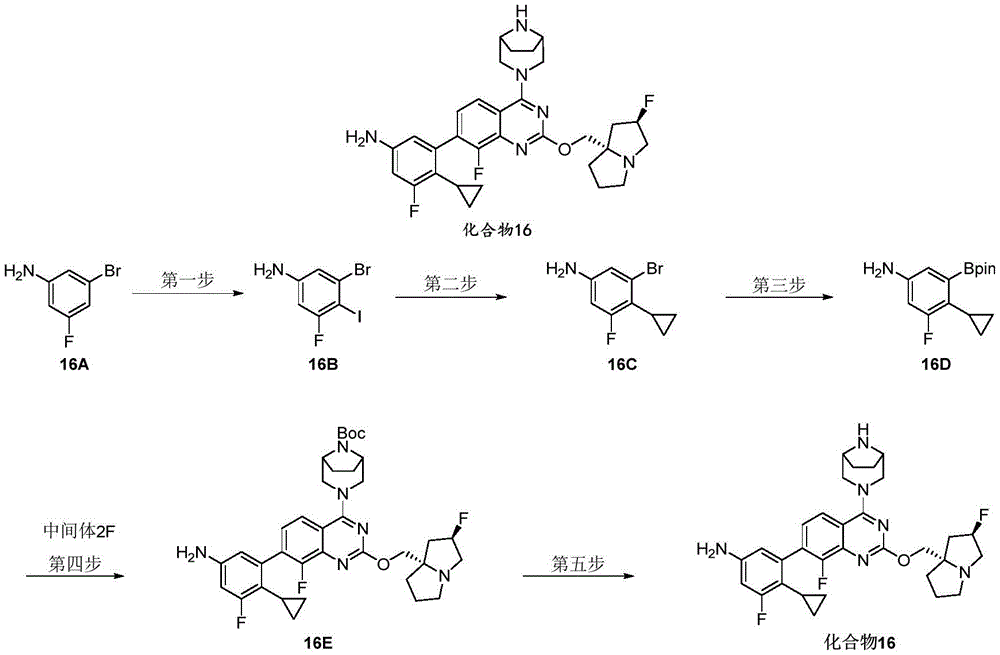
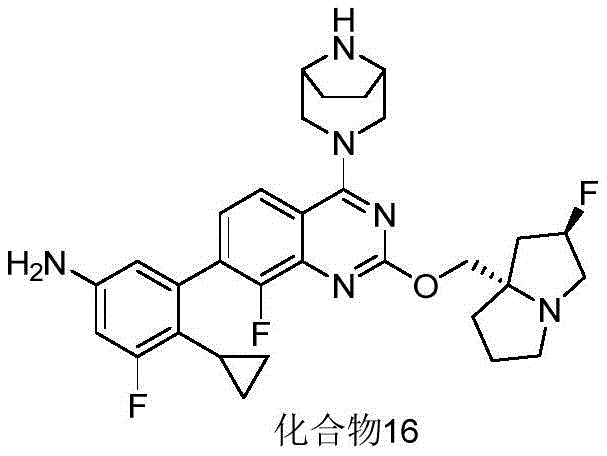
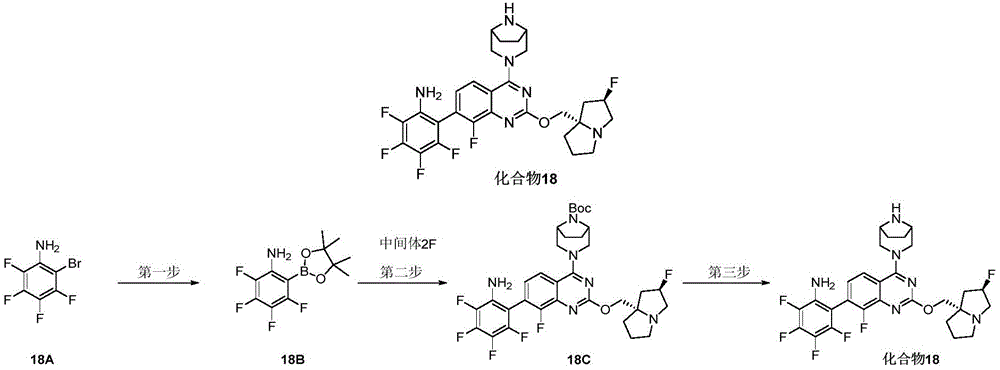
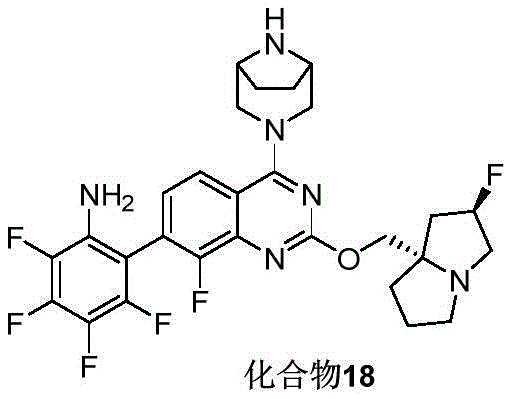
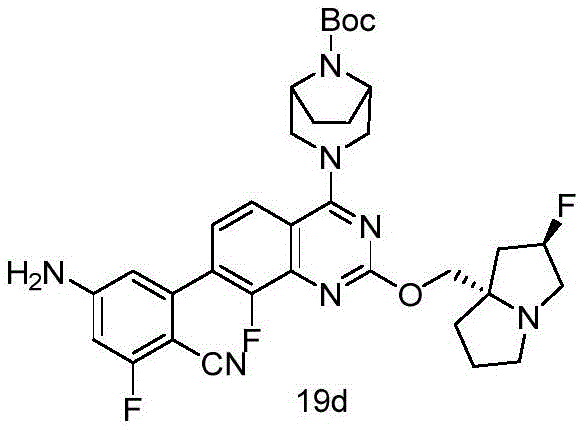
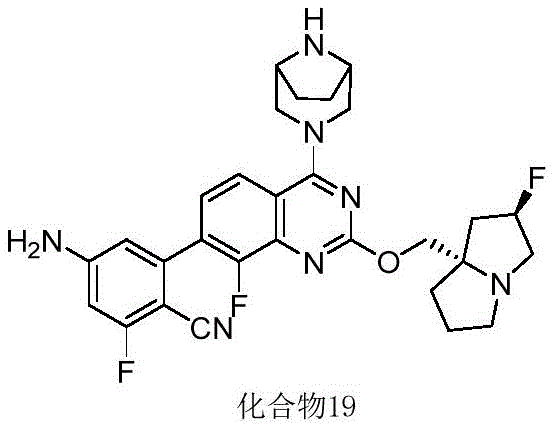
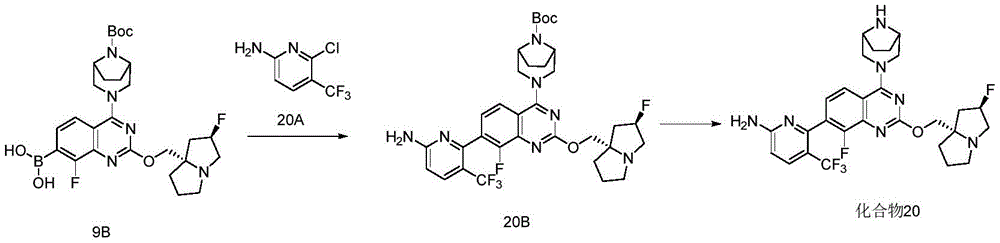
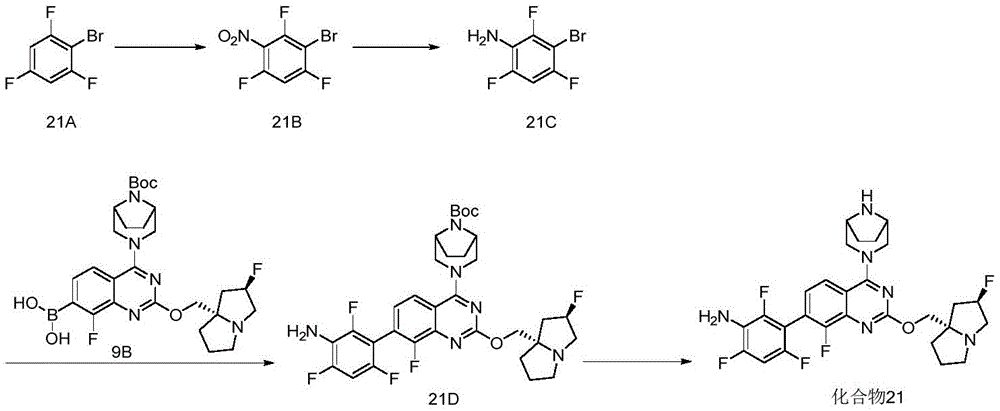
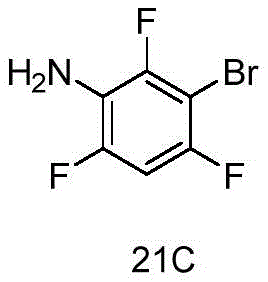
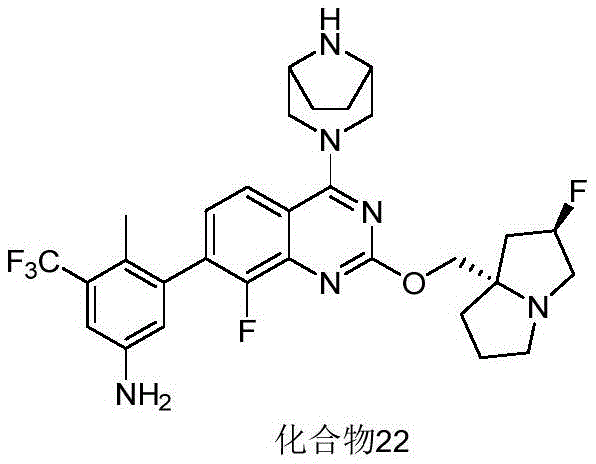
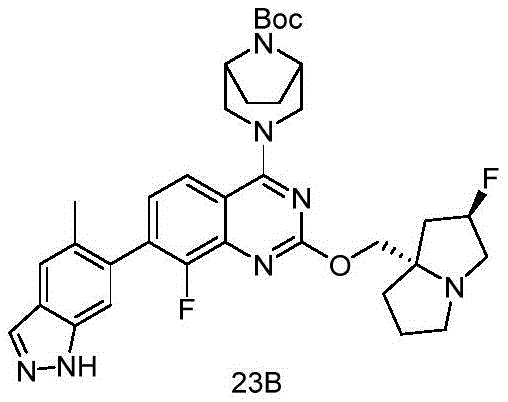
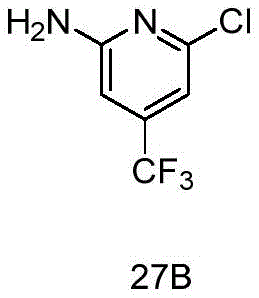
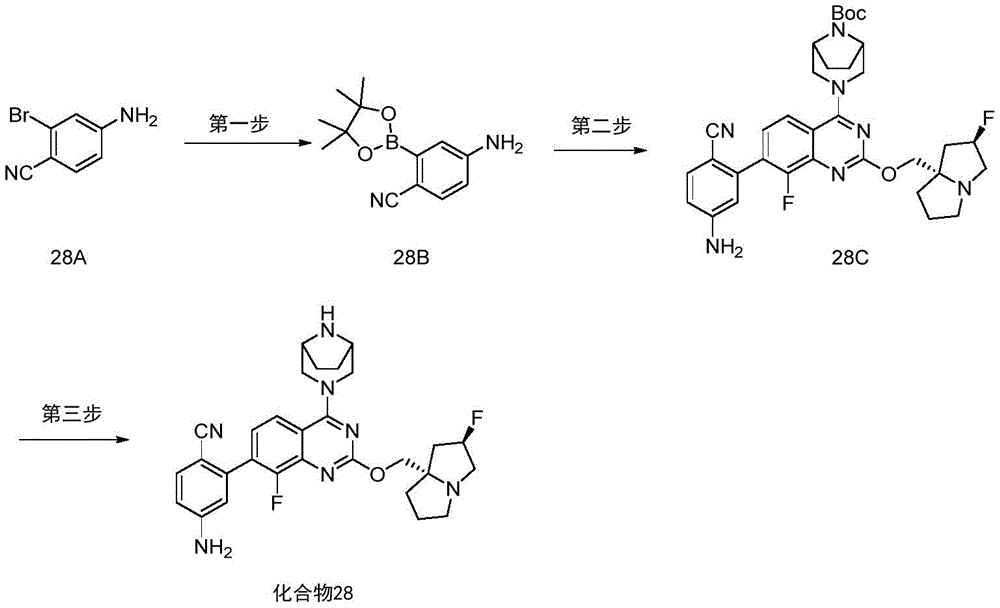
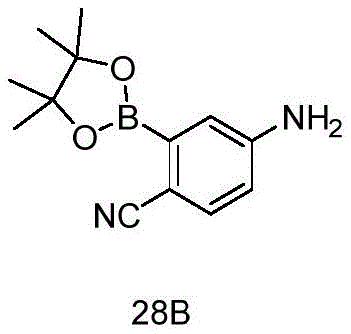
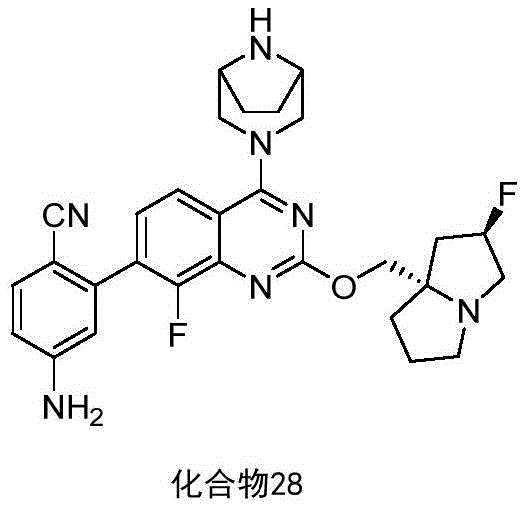
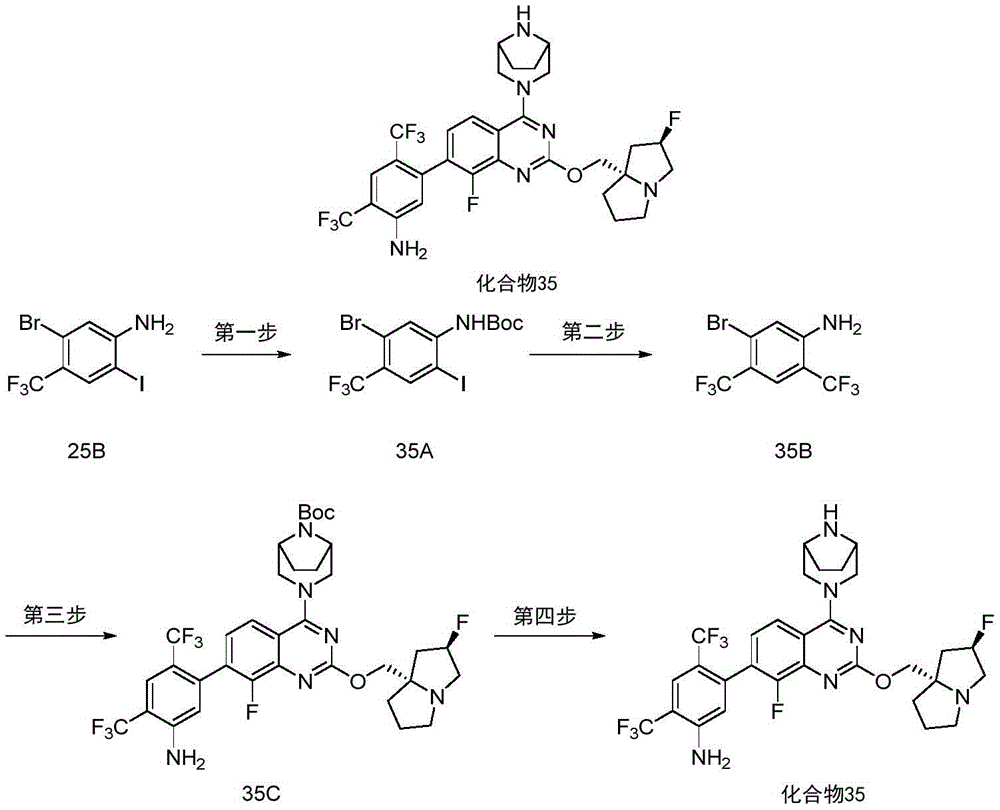
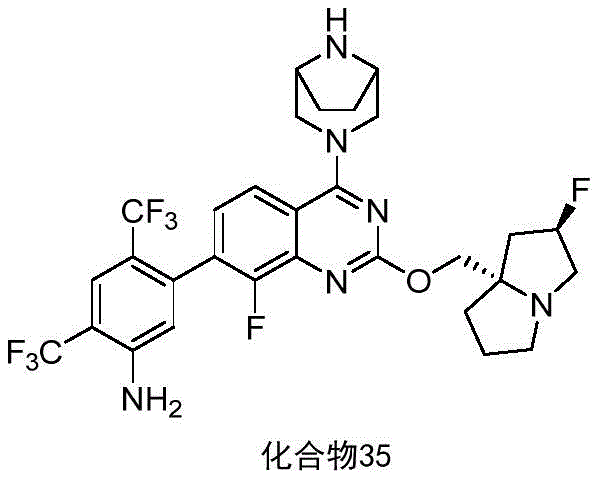
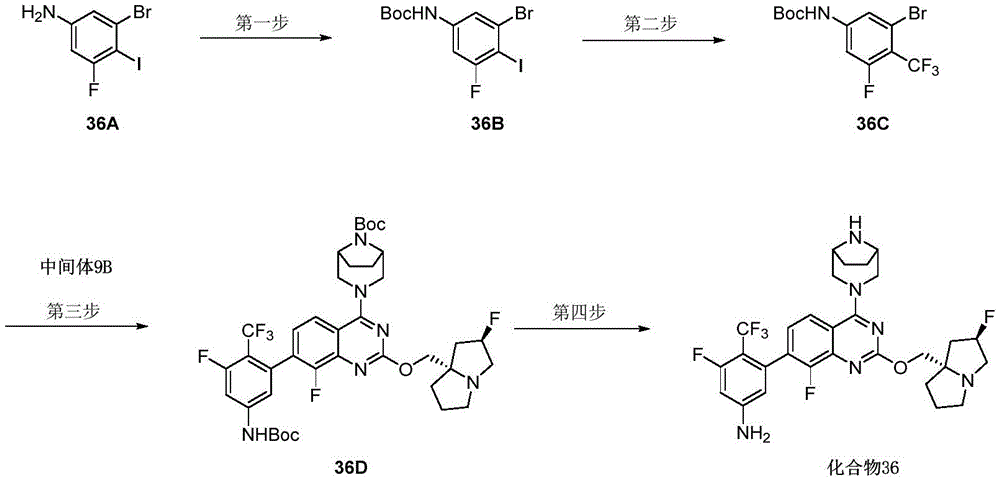
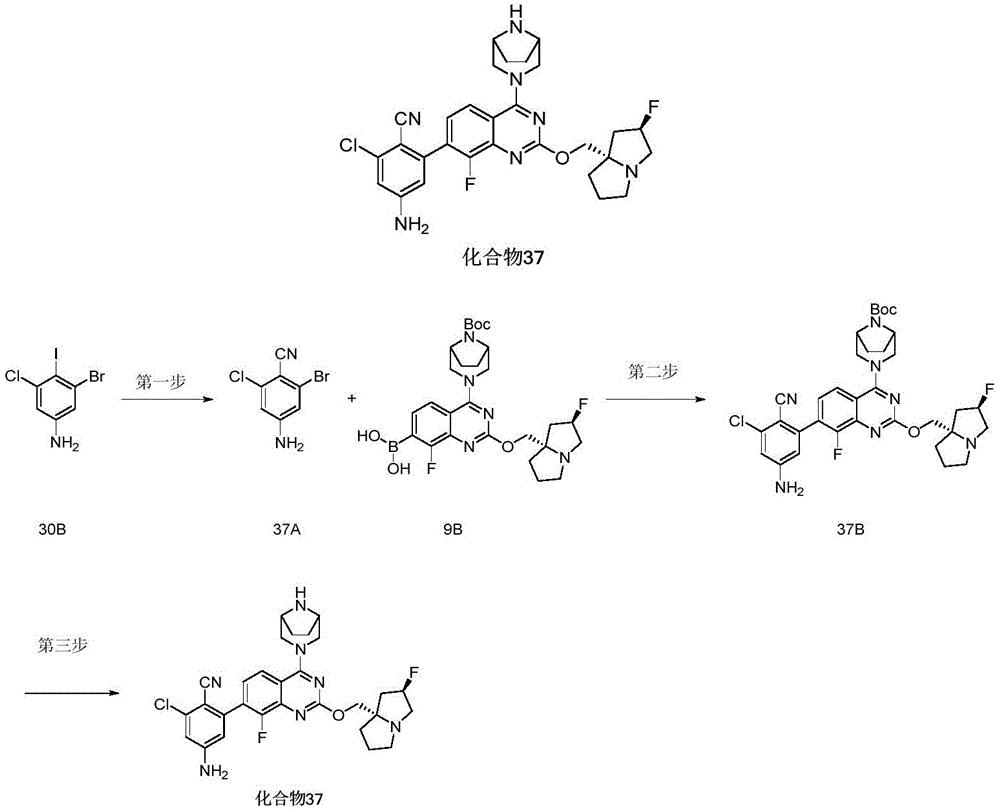
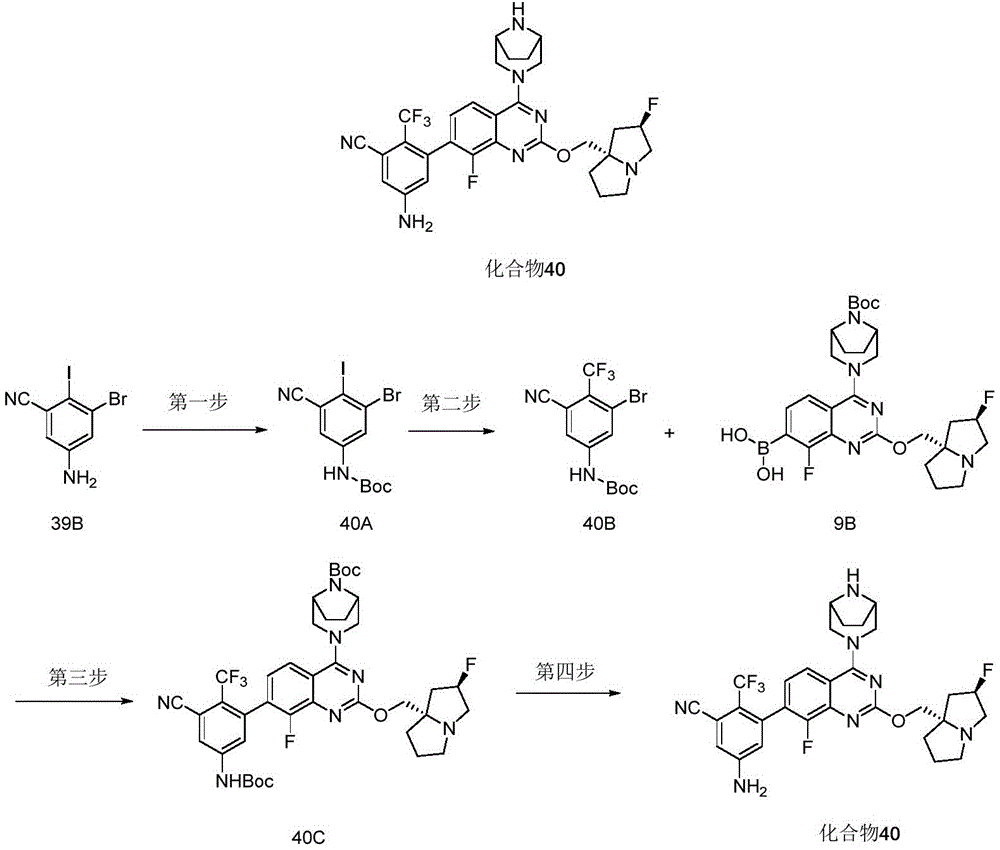
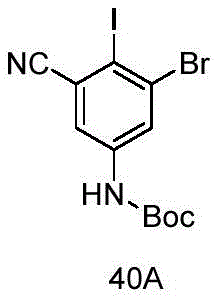
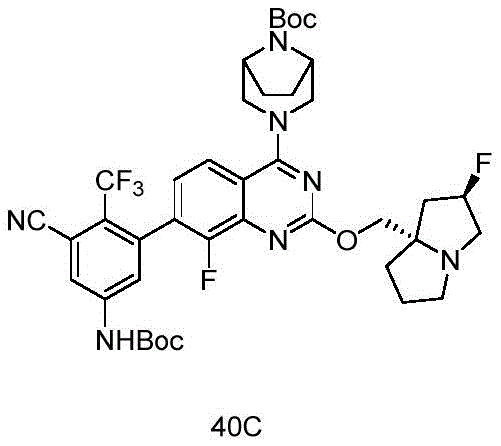
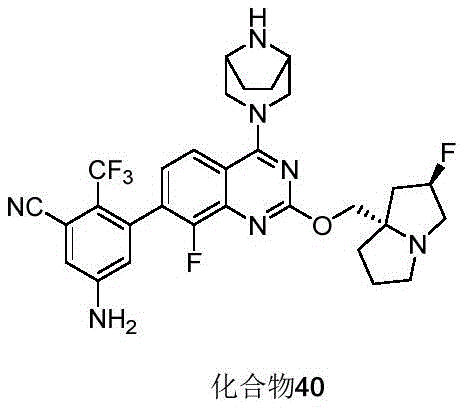
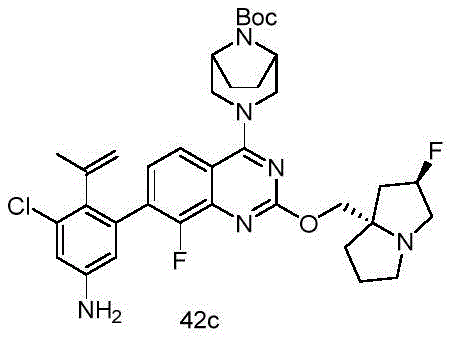
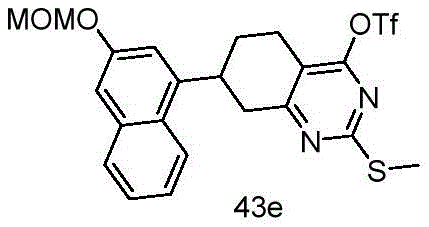
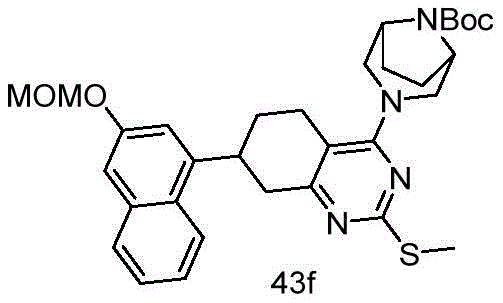
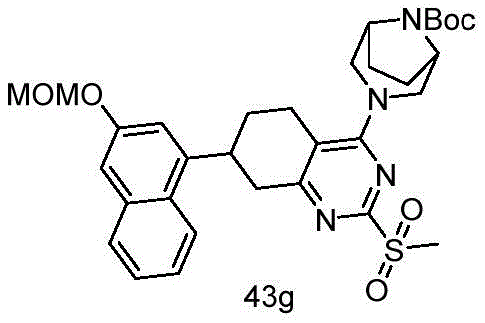
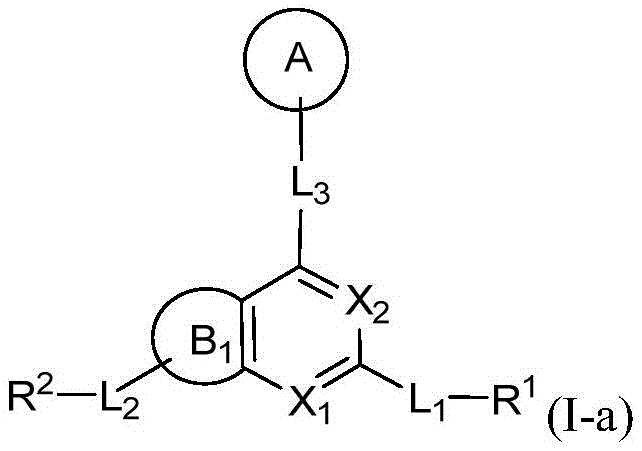
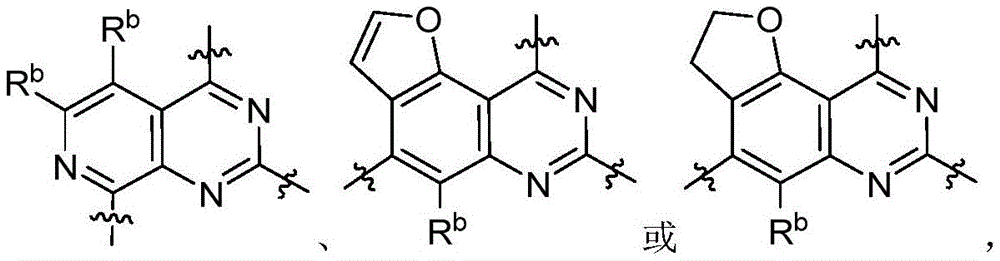
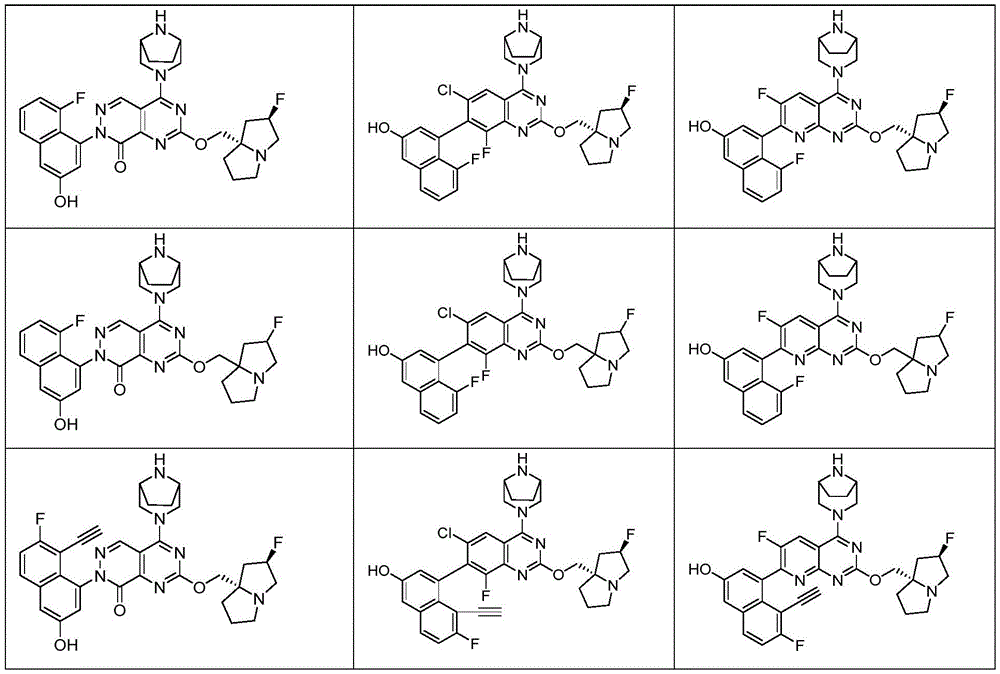
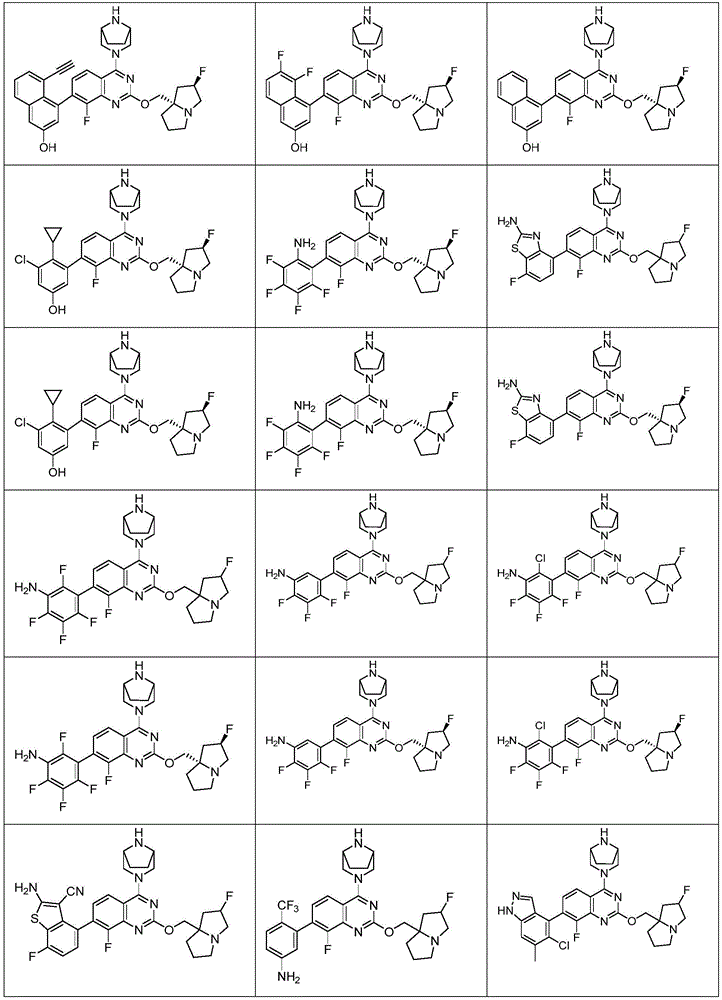
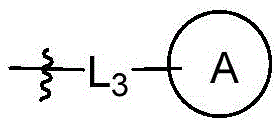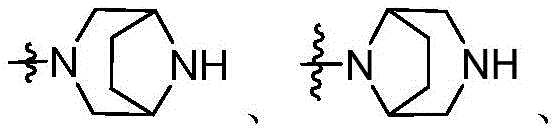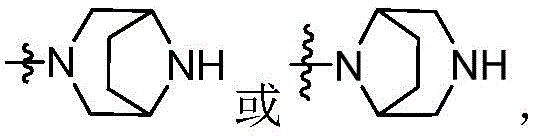CN115490709A - A KRASG12D inhibitor and its application in medicine - Google Patents
A KRASG12D inhibitor and its application in medicine Download PDFInfo
- Publication number
- CN115490709A CN115490709A CN202210444808.3A CN202210444808A CN115490709A CN 115490709 A CN115490709 A CN 115490709A CN 202210444808 A CN202210444808 A CN 202210444808A CN 115490709 A CN115490709 A CN 115490709A
- Authority
- CN
- China
- Prior art keywords
- alkyl
- membered
- methoxy
- cyano
- compound
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
- 239000003814 drug Substances 0.000 title claims abstract description 13
- 239000003112 inhibitor Substances 0.000 title abstract description 4
- 150000001875 compounds Chemical class 0.000 claims abstract description 239
- 239000000651 prodrug Substances 0.000 claims abstract description 39
- 229940002612 prodrug Drugs 0.000 claims abstract description 39
- 150000003839 salts Chemical class 0.000 claims abstract description 39
- 239000013078 crystal Substances 0.000 claims abstract description 37
- 239000012453 solvate Substances 0.000 claims abstract description 36
- 239000002207 metabolite Substances 0.000 claims abstract description 35
- 150000001204 N-oxides Chemical class 0.000 claims abstract description 23
- 201000010099 disease Diseases 0.000 claims abstract description 10
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims abstract description 10
- 230000000694 effects Effects 0.000 claims abstract description 9
- -1 Alkyl radical Chemical class 0.000 claims description 542
- 125000000623 heterocyclic group Chemical group 0.000 claims description 247
- 125000000217 alkyl group Chemical group 0.000 claims description 244
- 229910052757 nitrogen Inorganic materials 0.000 claims description 165
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 163
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 123
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 121
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 claims description 117
- 125000003545 alkoxy group Chemical group 0.000 claims description 110
- 125000005842 heteroatom Chemical group 0.000 claims description 108
- 229910052717 sulfur Inorganic materials 0.000 claims description 101
- 229910052731 fluorine Inorganic materials 0.000 claims description 98
- 229910052736 halogen Inorganic materials 0.000 claims description 98
- 150000002367 halogens Chemical class 0.000 claims description 98
- 229910052760 oxygen Inorganic materials 0.000 claims description 98
- 125000001424 substituent group Chemical group 0.000 claims description 95
- 125000000592 heterocycloalkyl group Chemical group 0.000 claims description 94
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 88
- 229910052739 hydrogen Inorganic materials 0.000 claims description 82
- 125000000753 cycloalkyl group Chemical group 0.000 claims description 70
- 125000004452 carbocyclyl group Chemical group 0.000 claims description 66
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 59
- 125000003118 aryl group Chemical group 0.000 claims description 55
- 125000001072 heteroaryl group Chemical group 0.000 claims description 55
- 229910052740 iodine Inorganic materials 0.000 claims description 55
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 claims description 52
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 claims description 48
- 125000002568 propynyl group Chemical group [*]C#CC([H])([H])[H] 0.000 claims description 47
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 claims description 45
- 125000003342 alkenyl group Chemical group 0.000 claims description 42
- 125000004368 propenyl group Chemical group C(=CC)* 0.000 claims description 42
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 claims description 41
- 125000004043 oxo group Chemical group O=* 0.000 claims description 40
- 125000005913 (C3-C6) cycloalkyl group Chemical group 0.000 claims description 39
- 125000000304 alkynyl group Chemical group 0.000 claims description 39
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 claims description 38
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 claims description 38
- 125000003226 pyrazolyl group Chemical group 0.000 claims description 31
- 125000002837 carbocyclic group Chemical group 0.000 claims description 29
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 claims description 29
- 125000002433 cyclopentenyl group Chemical group C1(=CCCC1)* 0.000 claims description 28
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 claims description 24
- 229920002554 vinyl polymer Polymers 0.000 claims description 23
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 claims description 22
- 125000003453 indazolyl group Chemical group N1N=C(C2=C1C=CC=C2)* 0.000 claims description 22
- 125000003601 C2-C6 alkynyl group Chemical group 0.000 claims description 20
- 125000004414 alkyl thio group Chemical group 0.000 claims description 20
- 125000003253 isopropoxy group Chemical group [H]C([H])([H])C([H])(O*)C([H])([H])[H] 0.000 claims description 20
- 125000004429 atom Chemical group 0.000 claims description 19
- 125000004618 benzofuryl group Chemical group O1C(=CC2=C1C=CC=C2)* 0.000 claims description 19
- 125000004076 pyridyl group Chemical group 0.000 claims description 17
- 125000001313 C5-C10 heteroaryl group Chemical group 0.000 claims description 16
- 125000001164 benzothiazolyl group Chemical group S1C(=NC2=C1C=CC=C2)* 0.000 claims description 15
- 125000004196 benzothienyl group Chemical group S1C(=CC2=C1C=CC=C2)* 0.000 claims description 15
- 125000001624 naphthyl group Chemical group 0.000 claims description 14
- 229920006395 saturated elastomer Polymers 0.000 claims description 14
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 claims description 13
- 229910052799 carbon Inorganic materials 0.000 claims description 12
- 125000002947 alkylene group Chemical group 0.000 claims description 11
- 125000002950 monocyclic group Chemical group 0.000 claims description 10
- 102200006539 rs121913529 Human genes 0.000 claims description 9
- 238000006467 substitution reaction Methods 0.000 claims description 9
- 125000006661 (C4-C6) heterocyclic group Chemical group 0.000 claims description 7
- 206010028980 Neoplasm Diseases 0.000 claims description 7
- 125000002572 propoxy group Chemical group [*]OC([H])([H])C(C([H])([H])[H])([H])[H] 0.000 claims description 7
- 125000005330 8 membered heterocyclic group Chemical group 0.000 claims description 6
- 125000003785 benzimidazolyl group Chemical group N1=C(NC2=C1C=CC=C2)* 0.000 claims description 6
- 125000002541 furyl group Chemical group 0.000 claims description 6
- 125000002883 imidazolyl group Chemical group 0.000 claims description 6
- 125000001544 thienyl group Chemical group 0.000 claims description 6
- BAXOFTOLAUCFNW-UHFFFAOYSA-N 1H-indazole Chemical compound C1=CC=C2C=NNC2=C1 BAXOFTOLAUCFNW-UHFFFAOYSA-N 0.000 claims description 5
- 102100030708 GTPase KRas Human genes 0.000 claims description 5
- 101000584612 Homo sapiens GTPase KRas Proteins 0.000 claims description 5
- 125000002971 oxazolyl group Chemical group 0.000 claims description 5
- 125000000168 pyrrolyl group Chemical group 0.000 claims description 5
- 125000000335 thiazolyl group Chemical group 0.000 claims description 5
- 125000001786 isothiazolyl group Chemical group 0.000 claims description 4
- 239000008194 pharmaceutical composition Substances 0.000 claims description 4
- 125000006570 (C5-C6) heteroaryl group Chemical group 0.000 claims description 3
- 125000000842 isoxazolyl group Chemical group 0.000 claims description 3
- 239000003937 drug carrier Substances 0.000 claims description 2
- 125000004008 6 membered carbocyclic group Chemical group 0.000 claims 3
- 125000000499 benzofuranyl group Chemical group O1C(=CC2=C1C=CC=C2)* 0.000 claims 3
- 238000004519 manufacturing process Methods 0.000 claims 2
- 238000002360 preparation method Methods 0.000 abstract description 61
- YZCKVEUIGOORGS-OUBTZVSYSA-N Deuterium Chemical compound [2H] YZCKVEUIGOORGS-OUBTZVSYSA-N 0.000 abstract description 12
- 229910052805 deuterium Inorganic materials 0.000 abstract description 10
- 229940079593 drug Drugs 0.000 abstract description 6
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 405
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 245
- 230000002829 reductive effect Effects 0.000 description 169
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 161
- 238000006243 chemical reaction Methods 0.000 description 159
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 126
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 113
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 111
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 107
- 239000000460 chlorine Substances 0.000 description 103
- 239000000243 solution Substances 0.000 description 78
- DTQVDTLACAAQTR-UHFFFAOYSA-N trifluoroacetic acid Substances OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 78
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 77
- 229910052801 chlorine Inorganic materials 0.000 description 76
- 239000012141 concentrate Substances 0.000 description 76
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 76
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 75
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 70
- 239000012071 phase Substances 0.000 description 69
- 239000012074 organic phase Substances 0.000 description 62
- 238000010828 elution Methods 0.000 description 60
- 238000004440 column chromatography Methods 0.000 description 57
- 238000004128 high performance liquid chromatography Methods 0.000 description 55
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 48
- 229910052794 bromium Inorganic materials 0.000 description 46
- 239000010410 layer Substances 0.000 description 41
- FJDQFPXHSGXQBY-UHFFFAOYSA-L caesium carbonate Chemical compound [Cs+].[Cs+].[O-]C([O-])=O FJDQFPXHSGXQBY-UHFFFAOYSA-L 0.000 description 37
- 229910000024 caesium carbonate Inorganic materials 0.000 description 37
- 239000002904 solvent Substances 0.000 description 32
- 239000007791 liquid phase Substances 0.000 description 28
- 239000008213 purified water Substances 0.000 description 28
- 239000000523 sample Substances 0.000 description 28
- 239000012488 sample solution Substances 0.000 description 28
- 239000007787 solid Substances 0.000 description 28
- 239000003643 water by type Substances 0.000 description 28
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 27
- KDLHZDBZIXYQEI-UHFFFAOYSA-N palladium Substances [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 27
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 26
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 25
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical class O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 24
- KZPYGQFFRCFCPP-UHFFFAOYSA-N 1,1'-bis(diphenylphosphino)ferrocene Chemical compound [Fe+2].C1=CC=C[C-]1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=C[C-]1P(C=1C=CC=CC=1)C1=CC=CC=C1 KZPYGQFFRCFCPP-UHFFFAOYSA-N 0.000 description 23
- 238000005481 NMR spectroscopy Methods 0.000 description 23
- 239000000843 powder Substances 0.000 description 23
- 239000000741 silica gel Substances 0.000 description 23
- 229910002027 silica gel Inorganic materials 0.000 description 23
- 238000010898 silica gel chromatography Methods 0.000 description 22
- 239000002585 base Substances 0.000 description 21
- 238000003756 stirring Methods 0.000 description 21
- PAYRUJLWNCNPSJ-UHFFFAOYSA-N Aniline Chemical compound NC1=CC=CC=C1 PAYRUJLWNCNPSJ-UHFFFAOYSA-N 0.000 description 18
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 16
- 239000008346 aqueous phase Substances 0.000 description 16
- 239000011259 mixed solution Substances 0.000 description 16
- 238000000746 purification Methods 0.000 description 16
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 15
- 125000004432 carbon atom Chemical group C* 0.000 description 15
- SCVFZCLFOSHCOH-UHFFFAOYSA-M potassium acetate Chemical compound [K+].CC([O-])=O SCVFZCLFOSHCOH-UHFFFAOYSA-M 0.000 description 15
- SNRCKKQHDUIRIY-UHFFFAOYSA-L cyclopenta-1,4-dien-1-yl(diphenyl)phosphane;dichloromethane;dichloropalladium;iron(2+) Chemical compound [Fe+2].ClCCl.Cl[Pd]Cl.C1=C[CH-]C(P(C=2C=CC=CC=2)C=2C=CC=CC=2)=C1.C1=C[CH-]C(P(C=2C=CC=CC=2)C=2C=CC=CC=2)=C1 SNRCKKQHDUIRIY-UHFFFAOYSA-L 0.000 description 14
- 125000004122 cyclic group Chemical group 0.000 description 13
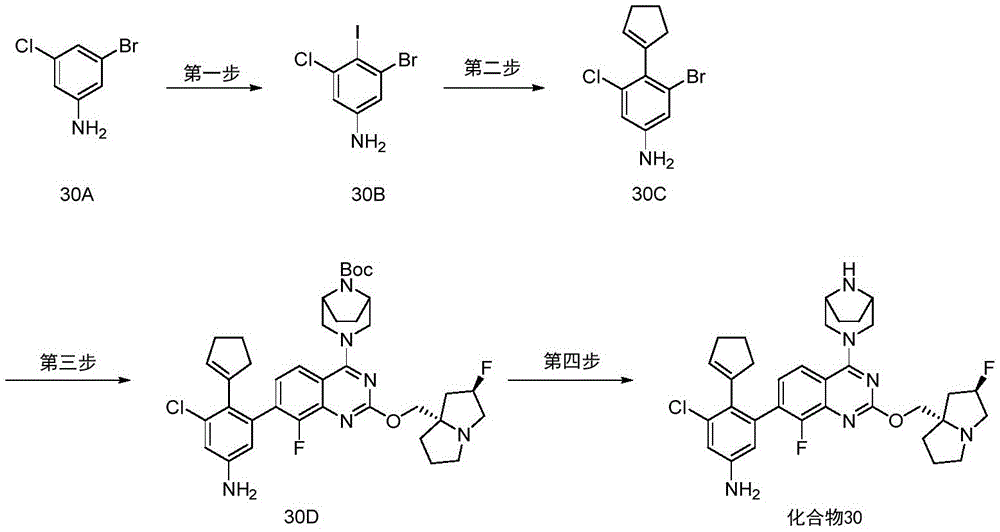
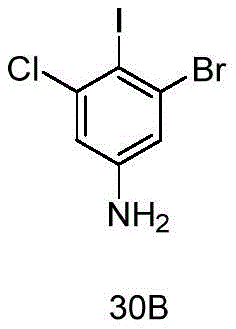
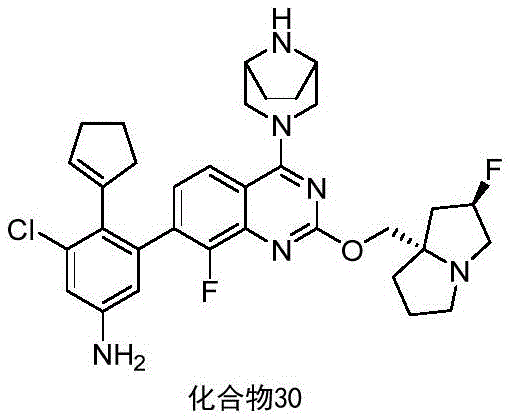
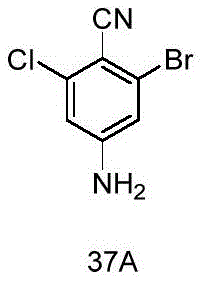
- DWAXZLJZJKIQNN-UHFFFAOYSA-N 3-bromo-5-chloro-4-iodoaniline Chemical compound Nc1cc(Cl)c(I)c(Br)c1 DWAXZLJZJKIQNN-UHFFFAOYSA-N 0.000 description 12
- 239000000706 filtrate Substances 0.000 description 12
- 125000000882 C2-C6 alkenyl group Chemical group 0.000 description 11
- 230000035772 mutation Effects 0.000 description 11
- FBUKVWPVBMHYJY-UHFFFAOYSA-N nonanoic acid Chemical compound CCCCCCCCC(O)=O FBUKVWPVBMHYJY-UHFFFAOYSA-N 0.000 description 11
- 125000003003 spiro group Chemical group 0.000 description 11
- LQZMLBORDGWNPD-UHFFFAOYSA-N N-iodosuccinimide Substances IN1C(=O)CCC1=O LQZMLBORDGWNPD-UHFFFAOYSA-N 0.000 description 10
- 239000012046 mixed solvent Substances 0.000 description 10
- NROKBHXJSPEDAR-UHFFFAOYSA-M potassium fluoride Chemical compound [F-].[K+] NROKBHXJSPEDAR-UHFFFAOYSA-M 0.000 description 10
- 102000016914 ras Proteins Human genes 0.000 description 10
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonia chloride Chemical compound [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 9
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 9
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 9
- 239000004698 Polyethylene Substances 0.000 description 9
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 9
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 9
- 239000002253 acid Substances 0.000 description 9
- 239000007788 liquid Substances 0.000 description 9
- 239000000047 product Substances 0.000 description 9
- 239000002994 raw material Substances 0.000 description 9
- BMIBJCFFZPYJHF-UHFFFAOYSA-N 2-methoxy-5-methyl-3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)pyridine Chemical compound COC1=NC=C(C)C=C1B1OC(C)(C)C(C)(C)O1 BMIBJCFFZPYJHF-UHFFFAOYSA-N 0.000 description 8
- PCLIMKBDDGJMGD-UHFFFAOYSA-N N-bromosuccinimide Chemical compound BrN1C(=O)CCC1=O PCLIMKBDDGJMGD-UHFFFAOYSA-N 0.000 description 8
- 235000012538 ammonium bicarbonate Nutrition 0.000 description 8
- XJHCXCQVJFPJIK-UHFFFAOYSA-M caesium fluoride Chemical compound [F-].[Cs+] XJHCXCQVJFPJIK-UHFFFAOYSA-M 0.000 description 8
- 125000006574 non-aromatic ring group Chemical group 0.000 description 8
- 239000000126 substance Substances 0.000 description 8
- XBTXLOYTLZINLR-UHFFFAOYSA-N 5-bromo-2-iodo-4-(trifluoromethyl)aniline Chemical compound NC1=CC(Br)=C(C(F)(F)F)C=C1I XBTXLOYTLZINLR-UHFFFAOYSA-N 0.000 description 7
- 101150040459 RAS gene Proteins 0.000 description 7
- 238000002953 preparative HPLC Methods 0.000 description 7
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 7
- 125000006700 (C1-C6) alkylthio group Chemical group 0.000 description 6
- FSYYMVYBUKORNS-UHFFFAOYSA-N 5-bromo-2-methyl-4-(trifluoromethyl)aniline Chemical compound Cc1cc(c(Br)cc1N)C(F)(F)F FSYYMVYBUKORNS-UHFFFAOYSA-N 0.000 description 6
- NXQGGXCHGDYOHB-UHFFFAOYSA-L cyclopenta-1,4-dien-1-yl(diphenyl)phosphane;dichloropalladium;iron(2+) Chemical compound [Fe+2].Cl[Pd]Cl.[CH-]1C=CC(P(C=2C=CC=CC=2)C=2C=CC=CC=2)=C1.[CH-]1C=CC(P(C=2C=CC=CC=2)C=2C=CC=CC=2)=C1 NXQGGXCHGDYOHB-UHFFFAOYSA-L 0.000 description 6
- VBXDEEVJTYBRJJ-UHFFFAOYSA-N diboronic acid Chemical compound OBOBO VBXDEEVJTYBRJJ-UHFFFAOYSA-N 0.000 description 6
- 125000002911 monocyclic heterocycle group Chemical group 0.000 description 6
- 239000012044 organic layer Substances 0.000 description 6
- PIBWKRNGBLPSSY-UHFFFAOYSA-L palladium(II) chloride Chemical compound Cl[Pd]Cl PIBWKRNGBLPSSY-UHFFFAOYSA-L 0.000 description 6
- XHXFXVLFKHQFAL-UHFFFAOYSA-N phosphoryl trichloride Chemical compound ClP(Cl)(Cl)=O XHXFXVLFKHQFAL-UHFFFAOYSA-N 0.000 description 6
- 239000011698 potassium fluoride Substances 0.000 description 6
- 238000000926 separation method Methods 0.000 description 6
- DYHSDKLCOJIUFX-UHFFFAOYSA-N tert-butoxycarbonyl anhydride Chemical compound CC(C)(C)OC(=O)OC(=O)OC(C)(C)C DYHSDKLCOJIUFX-UHFFFAOYSA-N 0.000 description 6
- IMNIMPAHZVJRPE-UHFFFAOYSA-N triethylenediamine Chemical compound C1CN2CCN1CC2 IMNIMPAHZVJRPE-UHFFFAOYSA-N 0.000 description 6
- HRAQPRIXJFDOAK-UHFFFAOYSA-N 2,3,4-trifluoro-5-iodoaniline Chemical compound FC1=C(N)C=C(C(=C1F)F)I HRAQPRIXJFDOAK-UHFFFAOYSA-N 0.000 description 5
- MBZAVZRGXBFAPQ-UHFFFAOYSA-N 3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-5-(trifluoromethyl)aniline Chemical compound O1C(C)(C)C(C)(C)OB1C1=CC(N)=CC(C(F)(F)F)=C1 MBZAVZRGXBFAPQ-UHFFFAOYSA-N 0.000 description 5
- WXLAKXBQJATZKF-UHFFFAOYSA-N CC(C)(C)OC(NC1=CC(Cl)=C(C(F)(F)F)C(Br)=C1)=O Chemical compound CC(C)(C)OC(NC1=CC(Cl)=C(C(F)(F)F)C(Br)=C1)=O WXLAKXBQJATZKF-UHFFFAOYSA-N 0.000 description 5
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 5
- KTGSPBURVFUONI-UHFFFAOYSA-N FC(F)(F)c1cc2cn[nH]c2cc1Br Chemical compound FC(F)(F)c1cc2cn[nH]c2cc1Br KTGSPBURVFUONI-UHFFFAOYSA-N 0.000 description 5
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical class [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 5
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical class [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 5
- 150000001335 aliphatic alkanes Chemical class 0.000 description 5
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 5
- 230000005496 eutectics Effects 0.000 description 5
- 239000001257 hydrogen Substances 0.000 description 5
- 230000000670 limiting effect Effects 0.000 description 5
- YNESATAKKCNGOF-UHFFFAOYSA-N lithium bis(trimethylsilyl)amide Chemical compound [Li+].C[Si](C)(C)[N-][Si](C)(C)C YNESATAKKCNGOF-UHFFFAOYSA-N 0.000 description 5
- 239000001301 oxygen Substances 0.000 description 5
- 235000011056 potassium acetate Nutrition 0.000 description 5
- 235000003270 potassium fluoride Nutrition 0.000 description 5
- JWVCLYRUEFBMGU-UHFFFAOYSA-N quinazoline Chemical compound N1=CN=CC2=CC=CC=C21 JWVCLYRUEFBMGU-UHFFFAOYSA-N 0.000 description 5
- 150000003254 radicals Chemical group 0.000 description 5
- 108010014186 ras Proteins Proteins 0.000 description 5
- 125000006413 ring segment Chemical group 0.000 description 5
- 239000007858 starting material Substances 0.000 description 5
- OEQJOYQHIGIVTN-UHFFFAOYSA-N tert-butyl nonanoate Chemical compound CCCCCCCCC(=O)OC(C)(C)C OEQJOYQHIGIVTN-UHFFFAOYSA-N 0.000 description 5
- WZZBNLYBHUDSHF-DHLKQENFSA-N 1-[(3s,4s)-4-[8-(2-chloro-4-pyrimidin-2-yloxyphenyl)-7-fluoro-2-methylimidazo[4,5-c]quinolin-1-yl]-3-fluoropiperidin-1-yl]-2-hydroxyethanone Chemical compound CC1=NC2=CN=C3C=C(F)C(C=4C(=CC(OC=5N=CC=CN=5)=CC=4)Cl)=CC3=C2N1[C@H]1CCN(C(=O)CO)C[C@@H]1F WZZBNLYBHUDSHF-DHLKQENFSA-N 0.000 description 4
- GQHTUMJGOHRCHB-UHFFFAOYSA-N 2,3,4,6,7,8,9,10-octahydropyrimido[1,2-a]azepine Chemical compound C1CCCCN2CCCN=C21 GQHTUMJGOHRCHB-UHFFFAOYSA-N 0.000 description 4
- SYTFXOAIAHTGBO-UHFFFAOYSA-N 2-bromo-1,3,5-trifluoro-4-nitrobenzene Chemical compound [O-][N+](=O)C1=C(F)C=C(F)C(Br)=C1F SYTFXOAIAHTGBO-UHFFFAOYSA-N 0.000 description 4
- MTFDNVGNGPISNO-UHFFFAOYSA-N 3-bromo-2,4,6-trifluoroaniline Chemical compound NC1=C(F)C=C(F)C(Br)=C1F MTFDNVGNGPISNO-UHFFFAOYSA-N 0.000 description 4
- YZGMWJRUAGKYER-UHFFFAOYSA-N 3-bromo-2-chloro-5-fluoroaniline Chemical compound NC1=CC(F)=CC(Br)=C1Cl YZGMWJRUAGKYER-UHFFFAOYSA-N 0.000 description 4
- VMERAUXEEDONDK-UHFFFAOYSA-N 3-bromo-4-(trifluoromethyl)aniline Chemical compound NC1=CC=C(C(F)(F)F)C(Br)=C1 VMERAUXEEDONDK-UHFFFAOYSA-N 0.000 description 4
- XPKPUVCHXFMSBC-UHFFFAOYSA-N 3-bromo-4-chloro-5-fluoroaniline Chemical compound BrC=1C=C(N)C=C(C1Cl)F XPKPUVCHXFMSBC-UHFFFAOYSA-N 0.000 description 4
- YYBRLVWWPRAQDX-UHFFFAOYSA-N 3-bromo-5-chloroaniline Chemical compound NC1=CC(Cl)=CC(Br)=C1 YYBRLVWWPRAQDX-UHFFFAOYSA-N 0.000 description 4
- QBZSDQPAVVBXBH-UHFFFAOYSA-N 3-bromo-5-fluoro-4-iodoaniline Chemical compound Nc1cc(F)c(I)c(Br)c1 QBZSDQPAVVBXBH-UHFFFAOYSA-N 0.000 description 4
- MWRPSTIQIFJYMI-UHFFFAOYSA-N 5-bromo-3-fluoro-2-(trifluoromethyl)aniline Chemical compound Nc1cc(Br)cc(F)c1C(F)(F)F MWRPSTIQIFJYMI-UHFFFAOYSA-N 0.000 description 4
- CREQAXQASINAFR-UHFFFAOYSA-N 6-chloro-4-(trifluoromethyl)pyridin-2-amine Chemical compound NC1=CC(C(F)(F)F)=CC(Cl)=N1 CREQAXQASINAFR-UHFFFAOYSA-N 0.000 description 4
- MZPLLPUWIXCTEI-UHFFFAOYSA-N 7-bromo-2,4,6-trichloro-8-fluoroquinazoline Chemical compound Fc1c(Br)c(Cl)cc2c(Cl)nc(Cl)nc12 MZPLLPUWIXCTEI-UHFFFAOYSA-N 0.000 description 4
- CJXZYJMCMOYEKA-UHFFFAOYSA-N 7-bromo-8-fluoro-1H-quinazoline-2,4-dione Chemical compound OC(C1=CC=C2Br)=NC(O)=NC1=C2F CJXZYJMCMOYEKA-UHFFFAOYSA-N 0.000 description 4
- UFQNRHVPOVZVKT-UHFFFAOYSA-N BrC1=CC=C2C(=NC(=NC2=C1F)Cl)N1CC2CCC(C1)N2C(=O)OC(C)(C)C Chemical compound BrC1=CC=C2C(=NC(=NC2=C1F)Cl)N1CC2CCC(C1)N2C(=O)OC(C)(C)C UFQNRHVPOVZVKT-UHFFFAOYSA-N 0.000 description 4
- QBERSHBNSRUGHS-UHFFFAOYSA-N BrC=1C=C(N)C=C(C1C1CC1)F Chemical compound BrC=1C=C(N)C=C(C1C1CC1)F QBERSHBNSRUGHS-UHFFFAOYSA-N 0.000 description 4
- OZMMOVNOZKRCII-UHFFFAOYSA-N CC1(C)OB(C2=CC(N)=CC(Cl)=C2C2CC2)OC1(C)C Chemical compound CC1(C)OB(C2=CC(N)=CC(Cl)=C2C2CC2)OC1(C)C OZMMOVNOZKRCII-UHFFFAOYSA-N 0.000 description 4
- OKKJLVBELUTLKV-MZCSYVLQSA-N Deuterated methanol Chemical compound [2H]OC([2H])([2H])[2H] OKKJLVBELUTLKV-MZCSYVLQSA-N 0.000 description 4
- RMTQLOZHXIEDGZ-UHFFFAOYSA-N Fc1c(Br)ccc2c(Cl)nc(Cl)nc12 Chemical compound Fc1c(Br)ccc2c(Cl)nc(Cl)nc12 RMTQLOZHXIEDGZ-UHFFFAOYSA-N 0.000 description 4
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 4
- 241001465754 Metazoa Species 0.000 description 4
- JRNVZBWKYDBUCA-UHFFFAOYSA-N N-chlorosuccinimide Chemical compound ClN1C(=O)CCC1=O JRNVZBWKYDBUCA-UHFFFAOYSA-N 0.000 description 4
- RQQPKTJGAJJMCT-UHFFFAOYSA-N NC(C(C=CC(Br)=C1F)=C1N)=O Chemical compound NC(C(C=CC(Br)=C1F)=C1N)=O RQQPKTJGAJJMCT-UHFFFAOYSA-N 0.000 description 4
- NVVFZZOLQSJEFU-UHFFFAOYSA-N NC1=CC(Br)=C(C2CC2)C(Cl)=C1 Chemical compound NC1=CC(Br)=C(C2CC2)C(Cl)=C1 NVVFZZOLQSJEFU-UHFFFAOYSA-N 0.000 description 4
- UFWIBTONFRDIAS-UHFFFAOYSA-N Naphthalene Chemical compound C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 4
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 4
- 235000019270 ammonium chloride Nutrition 0.000 description 4
- 201000011510 cancer Diseases 0.000 description 4
- WLVKDFJTYKELLQ-UHFFFAOYSA-N cyclopropylboronic acid Chemical compound OB(O)C1CC1 WLVKDFJTYKELLQ-UHFFFAOYSA-N 0.000 description 4
- ATUOYWHBWRKTHZ-UHFFFAOYSA-N dimethylmethane Natural products CCC ATUOYWHBWRKTHZ-UHFFFAOYSA-N 0.000 description 4
- 238000001035 drying Methods 0.000 description 4
- 238000000034 method Methods 0.000 description 4
- 239000002808 molecular sieve Substances 0.000 description 4
- RJTDBHBDKXIZJM-UHFFFAOYSA-N n-[3-bromo-4-(trifluoromethyl)phenyl]acetamide Chemical compound CC(=O)NC1=CC=C(C(F)(F)F)C(Br)=C1 RJTDBHBDKXIZJM-UHFFFAOYSA-N 0.000 description 4
- XBXCNNQPRYLIDE-UHFFFAOYSA-M n-tert-butylcarbamate Chemical compound CC(C)(C)NC([O-])=O XBXCNNQPRYLIDE-UHFFFAOYSA-M 0.000 description 4
- IVDFJHOHABJVEH-UHFFFAOYSA-N pinacol Chemical compound CC(C)(O)C(C)(C)O IVDFJHOHABJVEH-UHFFFAOYSA-N 0.000 description 4
- URGAHOPLAPQHLN-UHFFFAOYSA-N sodium aluminosilicate Chemical compound [Na+].[Al+3].[O-][Si]([O-])=O.[O-][Si]([O-])=O URGAHOPLAPQHLN-UHFFFAOYSA-N 0.000 description 4
- FEKPJTKLMHEVJH-UHFFFAOYSA-N tert-butyl N-[3-bromo-4-methyl-5-(trifluoromethyl)phenyl]carbamate Chemical compound BrC=1C=C(C=C(C=1C)C(F)(F)F)NC(OC(C)(C)C)=O FEKPJTKLMHEVJH-UHFFFAOYSA-N 0.000 description 4
- 238000004809 thin layer chromatography Methods 0.000 description 4
- 125000000025 triisopropylsilyl group Chemical group C(C)(C)[Si](C(C)C)(C(C)C)* 0.000 description 4
- UAOUIVVJBYDFKD-XKCDOFEDSA-N (1R,9R,10S,11R,12R,15S,18S,21R)-10,11,21-trihydroxy-8,8-dimethyl-14-methylidene-4-(prop-2-enylamino)-20-oxa-5-thia-3-azahexacyclo[9.7.2.112,15.01,9.02,6.012,18]henicosa-2(6),3-dien-13-one Chemical compound C([C@@H]1[C@@H](O)[C@@]23C(C1=C)=O)C[C@H]2[C@]12C(N=C(NCC=C)S4)=C4CC(C)(C)[C@H]1[C@H](O)[C@]3(O)OC2 UAOUIVVJBYDFKD-XKCDOFEDSA-N 0.000 description 3
- AOSZTAHDEDLTLQ-AZKQZHLXSA-N (1S,2S,4R,8S,9S,11S,12R,13S,19S)-6-[(3-chlorophenyl)methyl]-12,19-difluoro-11-hydroxy-8-(2-hydroxyacetyl)-9,13-dimethyl-6-azapentacyclo[10.8.0.02,9.04,8.013,18]icosa-14,17-dien-16-one Chemical compound C([C@@H]1C[C@H]2[C@H]3[C@]([C@]4(C=CC(=O)C=C4[C@@H](F)C3)C)(F)[C@@H](O)C[C@@]2([C@@]1(C1)C(=O)CO)C)N1CC1=CC=CC(Cl)=C1 AOSZTAHDEDLTLQ-AZKQZHLXSA-N 0.000 description 3
- GLGNXYJARSMNGJ-VKTIVEEGSA-N (1s,2s,3r,4r)-3-[[5-chloro-2-[(1-ethyl-6-methoxy-2-oxo-4,5-dihydro-3h-1-benzazepin-7-yl)amino]pyrimidin-4-yl]amino]bicyclo[2.2.1]hept-5-ene-2-carboxamide Chemical compound CCN1C(=O)CCCC2=C(OC)C(NC=3N=C(C(=CN=3)Cl)N[C@H]3[C@H]([C@@]4([H])C[C@@]3(C=C4)[H])C(N)=O)=CC=C21 GLGNXYJARSMNGJ-VKTIVEEGSA-N 0.000 description 3
- SZUVGFMDDVSKSI-WIFOCOSTSA-N (1s,2s,3s,5r)-1-(carboxymethyl)-3,5-bis[(4-phenoxyphenyl)methyl-propylcarbamoyl]cyclopentane-1,2-dicarboxylic acid Chemical compound O=C([C@@H]1[C@@H]([C@](CC(O)=O)([C@H](C(=O)N(CCC)CC=2C=CC(OC=3C=CC=CC=3)=CC=2)C1)C(O)=O)C(O)=O)N(CCC)CC(C=C1)=CC=C1OC1=CC=CC=C1 SZUVGFMDDVSKSI-WIFOCOSTSA-N 0.000 description 3
- GHYOCDFICYLMRF-UTIIJYGPSA-N (2S,3R)-N-[(2S)-3-(cyclopenten-1-yl)-1-[(2R)-2-methyloxiran-2-yl]-1-oxopropan-2-yl]-3-hydroxy-3-(4-methoxyphenyl)-2-[[(2S)-2-[(2-morpholin-4-ylacetyl)amino]propanoyl]amino]propanamide Chemical compound C1(=CCCC1)C[C@@H](C(=O)[C@@]1(OC1)C)NC([C@H]([C@@H](C1=CC=C(C=C1)OC)O)NC([C@H](C)NC(CN1CCOCC1)=O)=O)=O GHYOCDFICYLMRF-UTIIJYGPSA-N 0.000 description 3
- IUSARDYWEPUTPN-OZBXUNDUSA-N (2r)-n-[(2s,3r)-4-[[(4s)-6-(2,2-dimethylpropyl)spiro[3,4-dihydropyrano[2,3-b]pyridine-2,1'-cyclobutane]-4-yl]amino]-3-hydroxy-1-[3-(1,3-thiazol-2-yl)phenyl]butan-2-yl]-2-methoxypropanamide Chemical compound C([C@H](NC(=O)[C@@H](C)OC)[C@H](O)CN[C@@H]1C2=CC(CC(C)(C)C)=CN=C2OC2(CCC2)C1)C(C=1)=CC=CC=1C1=NC=CS1 IUSARDYWEPUTPN-OZBXUNDUSA-N 0.000 description 3
- ITOFPJRDSCGOSA-KZLRUDJFSA-N (2s)-2-[[(4r)-4-[(3r,5r,8r,9s,10s,13r,14s,17r)-3-hydroxy-10,13-dimethyl-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1h-cyclopenta[a]phenanthren-17-yl]pentanoyl]amino]-3-(1h-indol-3-yl)propanoic acid Chemical compound C([C@H]1CC2)[C@H](O)CC[C@]1(C)[C@@H](CC[C@]13C)[C@@H]2[C@@H]3CC[C@@H]1[C@H](C)CCC(=O)N[C@H](C(O)=O)CC1=CNC2=CC=CC=C12 ITOFPJRDSCGOSA-KZLRUDJFSA-N 0.000 description 3
- WWTBZEKOSBFBEM-SPWPXUSOSA-N (2s)-2-[[2-benzyl-3-[hydroxy-[(1r)-2-phenyl-1-(phenylmethoxycarbonylamino)ethyl]phosphoryl]propanoyl]amino]-3-(1h-indol-3-yl)propanoic acid Chemical compound N([C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)O)C(=O)C(CP(O)(=O)[C@H](CC=1C=CC=CC=1)NC(=O)OCC=1C=CC=CC=1)CC1=CC=CC=C1 WWTBZEKOSBFBEM-SPWPXUSOSA-N 0.000 description 3
- STBLNCCBQMHSRC-BATDWUPUSA-N (2s)-n-[(3s,4s)-5-acetyl-7-cyano-4-methyl-1-[(2-methylnaphthalen-1-yl)methyl]-2-oxo-3,4-dihydro-1,5-benzodiazepin-3-yl]-2-(methylamino)propanamide Chemical compound O=C1[C@@H](NC(=O)[C@H](C)NC)[C@H](C)N(C(C)=O)C2=CC(C#N)=CC=C2N1CC1=C(C)C=CC2=CC=CC=C12 STBLNCCBQMHSRC-BATDWUPUSA-N 0.000 description 3
- QFLWZFQWSBQYPS-AWRAUJHKSA-N (3S)-3-[[(2S)-2-[[(2S)-2-[5-[(3aS,6aR)-2-oxo-1,3,3a,4,6,6a-hexahydrothieno[3,4-d]imidazol-4-yl]pentanoylamino]-3-methylbutanoyl]amino]-3-(4-hydroxyphenyl)propanoyl]amino]-4-[1-bis(4-chlorophenoxy)phosphorylbutylamino]-4-oxobutanoic acid Chemical compound CCCC(NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@@H](NC(=O)CCCCC1SC[C@@H]2NC(=O)N[C@H]12)C(C)C)P(=O)(Oc1ccc(Cl)cc1)Oc1ccc(Cl)cc1 QFLWZFQWSBQYPS-AWRAUJHKSA-N 0.000 description 3
- KQZLRWGGWXJPOS-NLFPWZOASA-N 1-[(1R)-1-(2,4-dichlorophenyl)ethyl]-6-[(4S,5R)-4-[(2S)-2-(hydroxymethyl)pyrrolidin-1-yl]-5-methylcyclohexen-1-yl]pyrazolo[3,4-b]pyrazine-3-carbonitrile Chemical compound ClC1=C(C=CC(=C1)Cl)[C@@H](C)N1N=C(C=2C1=NC(=CN=2)C1=CC[C@@H]([C@@H](C1)C)N1[C@@H](CCC1)CO)C#N KQZLRWGGWXJPOS-NLFPWZOASA-N 0.000 description 3
- ONBQEOIKXPHGMB-VBSBHUPXSA-N 1-[2-[(2s,3r,4s,5r)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]oxy-4,6-dihydroxyphenyl]-3-(4-hydroxyphenyl)propan-1-one Chemical compound O[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1OC1=CC(O)=CC(O)=C1C(=O)CCC1=CC=C(O)C=C1 ONBQEOIKXPHGMB-VBSBHUPXSA-N 0.000 description 3
- UNILWMWFPHPYOR-KXEYIPSPSA-M 1-[6-[2-[3-[3-[3-[2-[2-[3-[[2-[2-[[(2r)-1-[[2-[[(2r)-1-[3-[2-[2-[3-[[2-(2-amino-2-oxoethoxy)acetyl]amino]propoxy]ethoxy]ethoxy]propylamino]-3-hydroxy-1-oxopropan-2-yl]amino]-2-oxoethyl]amino]-3-[(2r)-2,3-di(hexadecanoyloxy)propyl]sulfanyl-1-oxopropan-2-yl Chemical compound O=C1C(SCCC(=O)NCCCOCCOCCOCCCNC(=O)COCC(=O)N[C@@H](CSC[C@@H](COC(=O)CCCCCCCCCCCCCCC)OC(=O)CCCCCCCCCCCCCCC)C(=O)NCC(=O)N[C@H](CO)C(=O)NCCCOCCOCCOCCCNC(=O)COCC(N)=O)CC(=O)N1CCNC(=O)CCCCCN\1C2=CC=C(S([O-])(=O)=O)C=C2CC/1=C/C=C/C=C/C1=[N+](CC)C2=CC=C(S([O-])(=O)=O)C=C2C1 UNILWMWFPHPYOR-KXEYIPSPSA-M 0.000 description 3
- YSUIQYOGTINQIN-UZFYAQMZSA-N 2-amino-9-[(1S,6R,8R,9S,10R,15R,17R,18R)-8-(6-aminopurin-9-yl)-9,18-difluoro-3,12-dihydroxy-3,12-bis(sulfanylidene)-2,4,7,11,13,16-hexaoxa-3lambda5,12lambda5-diphosphatricyclo[13.2.1.06,10]octadecan-17-yl]-1H-purin-6-one Chemical compound NC1=NC2=C(N=CN2[C@@H]2O[C@@H]3COP(S)(=O)O[C@@H]4[C@@H](COP(S)(=O)O[C@@H]2[C@@H]3F)O[C@H]([C@H]4F)N2C=NC3=C2N=CN=C3N)C(=O)N1 YSUIQYOGTINQIN-UZFYAQMZSA-N 0.000 description 3
- TVTJUIAKQFIXCE-HUKYDQBMSA-N 2-amino-9-[(2R,3S,4S,5R)-4-fluoro-3-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-7-prop-2-ynyl-1H-purine-6,8-dione Chemical compound NC=1NC(C=2N(C(N(C=2N=1)[C@@H]1O[C@@H]([C@H]([C@H]1O)F)CO)=O)CC#C)=O TVTJUIAKQFIXCE-HUKYDQBMSA-N 0.000 description 3
- NPRYCHLHHVWLQZ-TURQNECASA-N 2-amino-9-[(2R,3S,4S,5R)-4-fluoro-3-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-7-prop-2-ynylpurin-8-one Chemical compound NC1=NC=C2N(C(N(C2=N1)[C@@H]1O[C@@H]([C@H]([C@H]1O)F)CO)=O)CC#C NPRYCHLHHVWLQZ-TURQNECASA-N 0.000 description 3
- QBWKPGNFQQJGFY-QLFBSQMISA-N 3-[(1r)-1-[(2r,6s)-2,6-dimethylmorpholin-4-yl]ethyl]-n-[6-methyl-3-(1h-pyrazol-4-yl)imidazo[1,2-a]pyrazin-8-yl]-1,2-thiazol-5-amine Chemical compound N1([C@H](C)C2=NSC(NC=3C4=NC=C(N4C=C(C)N=3)C3=CNN=C3)=C2)C[C@H](C)O[C@H](C)C1 QBWKPGNFQQJGFY-QLFBSQMISA-N 0.000 description 3
- FUKITJMBFMUDGQ-UHFFFAOYSA-N 3-bromo-4-methyl-5-(trifluoromethyl)benzoic acid Chemical compound BrC=1C=C(C(=O)O)C=C(C=1C)C(F)(F)F FUKITJMBFMUDGQ-UHFFFAOYSA-N 0.000 description 3
- NGZAVSDIXFIWHJ-UHFFFAOYSA-N 3-bromo-5-fluoroaniline Chemical compound NC1=CC(F)=CC(Br)=C1 NGZAVSDIXFIWHJ-UHFFFAOYSA-N 0.000 description 3
- 229940126657 Compound 17 Drugs 0.000 description 3
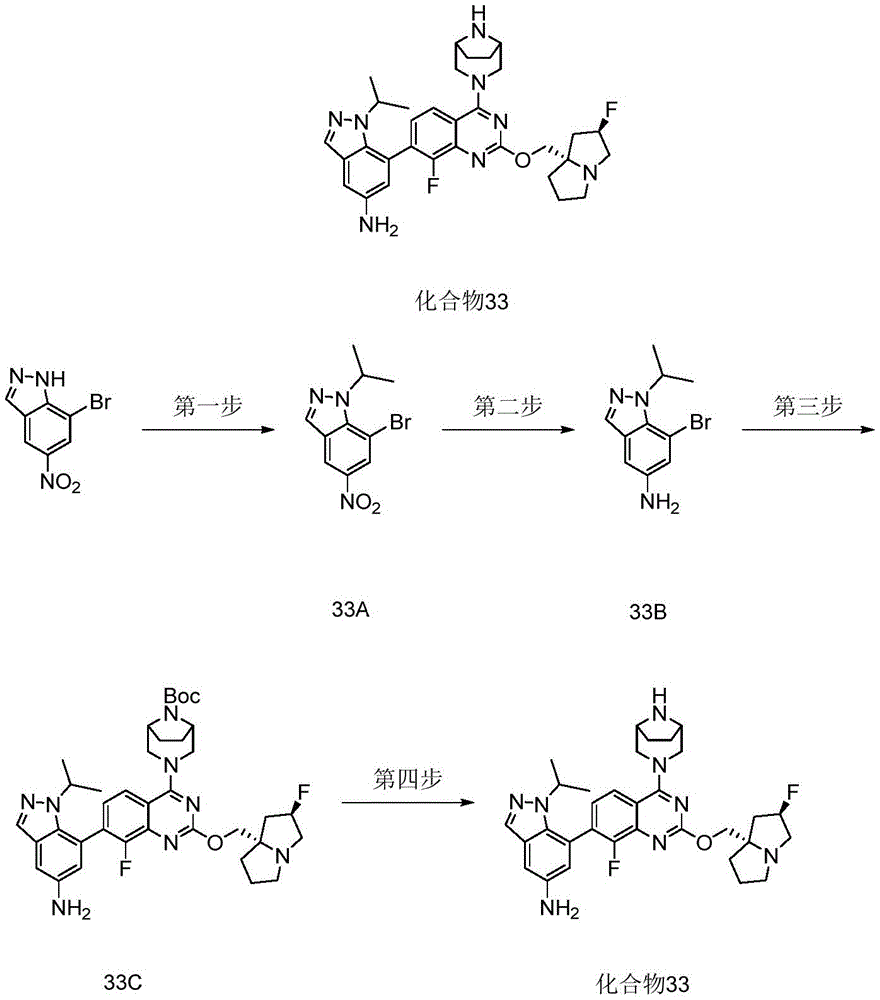
- 229940126639 Compound 33 Drugs 0.000 description 3
- 229910021595 Copper(I) iodide Inorganic materials 0.000 description 3
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 3
- 102000008300 Mutant Proteins Human genes 0.000 description 3
- 108010021466 Mutant Proteins Proteins 0.000 description 3
- OPFJDXRVMFKJJO-ZHHKINOHSA-N N-{[3-(2-benzamido-4-methyl-1,3-thiazol-5-yl)-pyrazol-5-yl]carbonyl}-G-dR-G-dD-dD-dD-NH2 Chemical compound S1C(C=2NN=C(C=2)C(=O)NCC(=O)N[C@H](CCCN=C(N)N)C(=O)NCC(=O)N[C@H](CC(O)=O)C(=O)N[C@H](CC(O)=O)C(=O)N[C@H](CC(O)=O)C(N)=O)=C(C)N=C1NC(=O)C1=CC=CC=C1 OPFJDXRVMFKJJO-ZHHKINOHSA-N 0.000 description 3
- PNUZDKCDAWUEGK-CYZMBNFOSA-N Sitafloxacin Chemical compound C([C@H]1N)N(C=2C(=C3C(C(C(C(O)=O)=CN3[C@H]3[C@H](C3)F)=O)=CC=2F)Cl)CC11CC1 PNUZDKCDAWUEGK-CYZMBNFOSA-N 0.000 description 3
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 3
- LJOOWESTVASNOG-UFJKPHDISA-N [(1s,3r,4ar,7s,8s,8as)-3-hydroxy-8-[2-[(4r)-4-hydroxy-6-oxooxan-2-yl]ethyl]-7-methyl-1,2,3,4,4a,7,8,8a-octahydronaphthalen-1-yl] (2s)-2-methylbutanoate Chemical compound C([C@H]1[C@@H](C)C=C[C@H]2C[C@@H](O)C[C@@H]([C@H]12)OC(=O)[C@@H](C)CC)CC1C[C@@H](O)CC(=O)O1 LJOOWESTVASNOG-UFJKPHDISA-N 0.000 description 3
- LNUFLCYMSVYYNW-ZPJMAFJPSA-N [(2r,3r,4s,5r,6r)-2-[(2r,3r,4s,5r,6r)-6-[(2r,3r,4s,5r,6r)-6-[(2r,3r,4s,5r,6r)-6-[[(3s,5s,8r,9s,10s,13r,14s,17r)-10,13-dimethyl-17-[(2r)-6-methylheptan-2-yl]-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1h-cyclopenta[a]phenanthren-3-yl]oxy]-4,5-disulfo Chemical compound O([C@@H]1[C@@H](COS(O)(=O)=O)O[C@@H]([C@@H]([C@H]1OS(O)(=O)=O)OS(O)(=O)=O)O[C@@H]1[C@@H](COS(O)(=O)=O)O[C@@H]([C@@H]([C@H]1OS(O)(=O)=O)OS(O)(=O)=O)O[C@@H]1[C@@H](COS(O)(=O)=O)O[C@H]([C@@H]([C@H]1OS(O)(=O)=O)OS(O)(=O)=O)O[C@@H]1C[C@@H]2CC[C@H]3[C@@H]4CC[C@@H]([C@]4(CC[C@@H]3[C@@]2(C)CC1)C)[C@H](C)CCCC(C)C)[C@H]1O[C@H](COS(O)(=O)=O)[C@@H](OS(O)(=O)=O)[C@H](OS(O)(=O)=O)[C@H]1OS(O)(=O)=O LNUFLCYMSVYYNW-ZPJMAFJPSA-N 0.000 description 3
- SMNRFWMNPDABKZ-WVALLCKVSA-N [[(2R,3S,4R,5S)-5-(2,6-dioxo-3H-pyridin-3-yl)-3,4-dihydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl] [[[(2R,3S,4S,5R,6R)-4-fluoro-3,5-dihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-hydroxyphosphoryl]oxy-hydroxyphosphoryl] hydrogen phosphate Chemical compound OC[C@H]1O[C@H](OP(O)(=O)OP(O)(=O)OP(O)(=O)OP(O)(=O)OC[C@H]2O[C@H]([C@H](O)[C@@H]2O)C2C=CC(=O)NC2=O)[C@H](O)[C@@H](F)[C@@H]1O SMNRFWMNPDABKZ-WVALLCKVSA-N 0.000 description 3
- 239000008186 active pharmaceutical agent Substances 0.000 description 3
- 239000007864 aqueous solution Substances 0.000 description 3
- XRWSZZJLZRKHHD-WVWIJVSJSA-N asunaprevir Chemical compound O=C([C@@H]1C[C@H](CN1C(=O)[C@@H](NC(=O)OC(C)(C)C)C(C)(C)C)OC1=NC=C(C2=CC=C(Cl)C=C21)OC)N[C@]1(C(=O)NS(=O)(=O)C2CC2)C[C@H]1C=C XRWSZZJLZRKHHD-WVWIJVSJSA-N 0.000 description 3
- KGNDCEVUMONOKF-UGPLYTSKSA-N benzyl n-[(2r)-1-[(2s,4r)-2-[[(2s)-6-amino-1-(1,3-benzoxazol-2-yl)-1,1-dihydroxyhexan-2-yl]carbamoyl]-4-[(4-methylphenyl)methoxy]pyrrolidin-1-yl]-1-oxo-4-phenylbutan-2-yl]carbamate Chemical compound C1=CC(C)=CC=C1CO[C@H]1CN(C(=O)[C@@H](CCC=2C=CC=CC=2)NC(=O)OCC=2C=CC=CC=2)[C@H](C(=O)N[C@@H](CCCCN)C(O)(O)C=2OC3=CC=CC=C3N=2)C1 KGNDCEVUMONOKF-UGPLYTSKSA-N 0.000 description 3
- 238000013375 chromatographic separation Methods 0.000 description 3
- 238000011097 chromatography purification Methods 0.000 description 3
- 229940125904 compound 1 Drugs 0.000 description 3
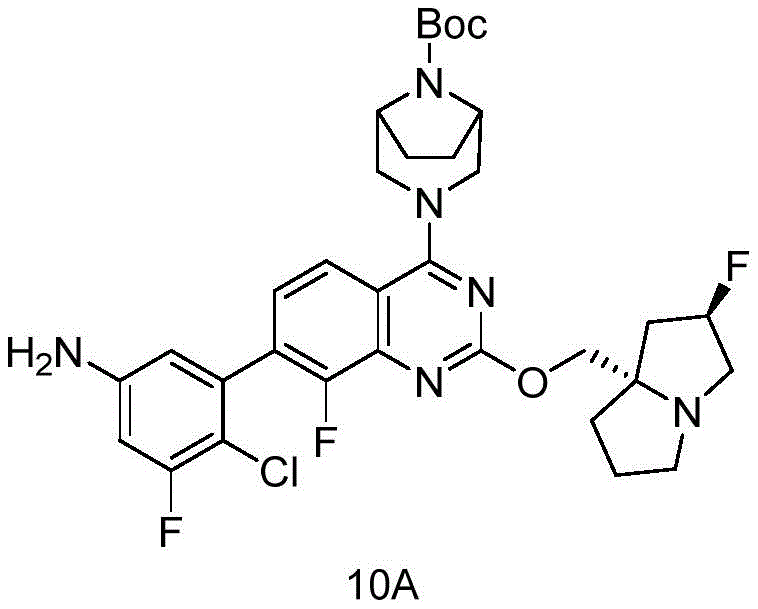
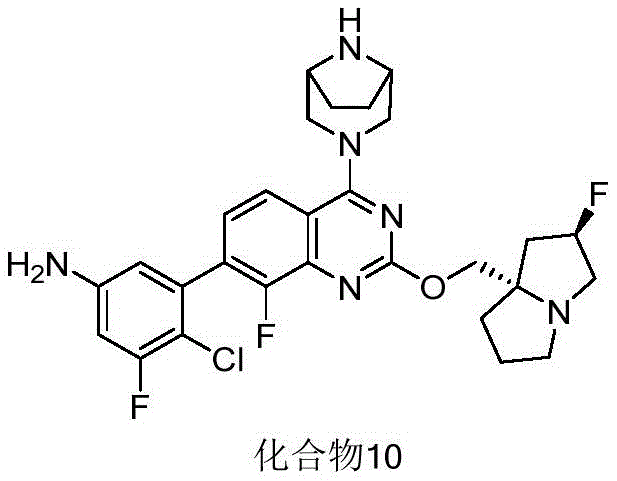
- 229940125773 compound 10 Drugs 0.000 description 3
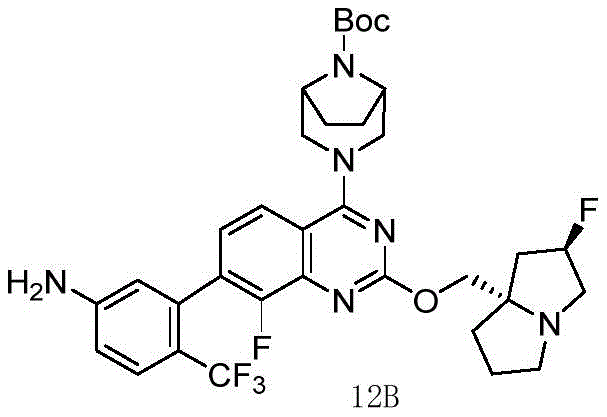
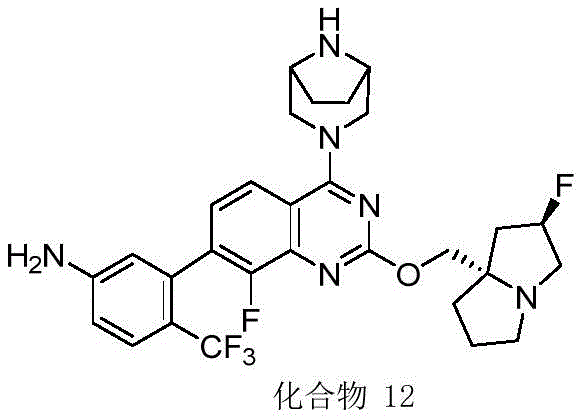
- 229940125797 compound 12 Drugs 0.000 description 3
- 229940126543 compound 14 Drugs 0.000 description 3
- 229940125758 compound 15 Drugs 0.000 description 3
- 229940126142 compound 16 Drugs 0.000 description 3
- 229940125782 compound 2 Drugs 0.000 description 3
- 229940125810 compound 20 Drugs 0.000 description 3
- 229940126086 compound 21 Drugs 0.000 description 3
- 229940126208 compound 22 Drugs 0.000 description 3
- 229940125833 compound 23 Drugs 0.000 description 3
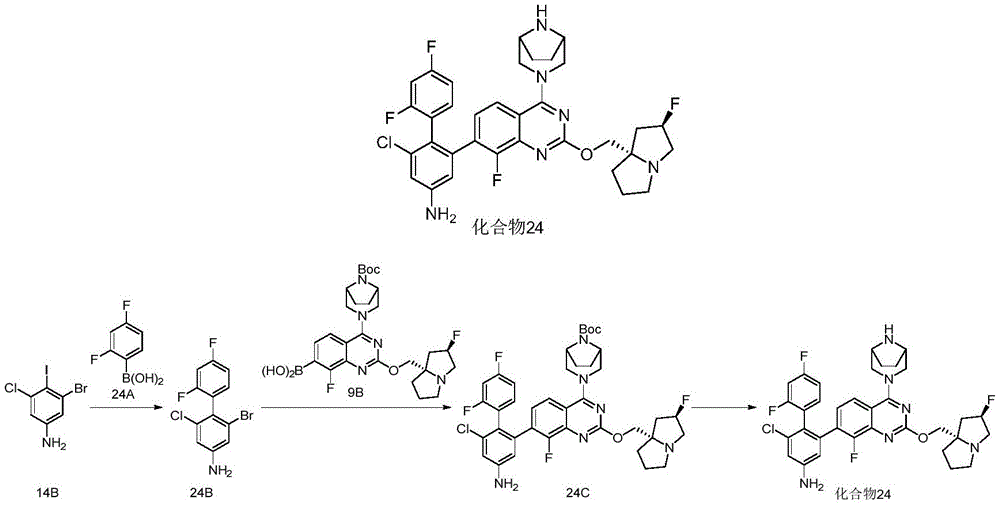
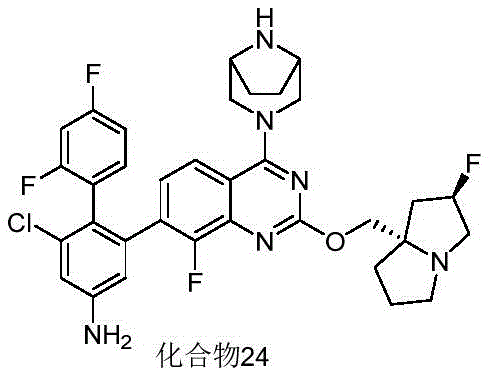
- 229940125961 compound 24 Drugs 0.000 description 3
- 229940125846 compound 25 Drugs 0.000 description 3
- 229940125851 compound 27 Drugs 0.000 description 3
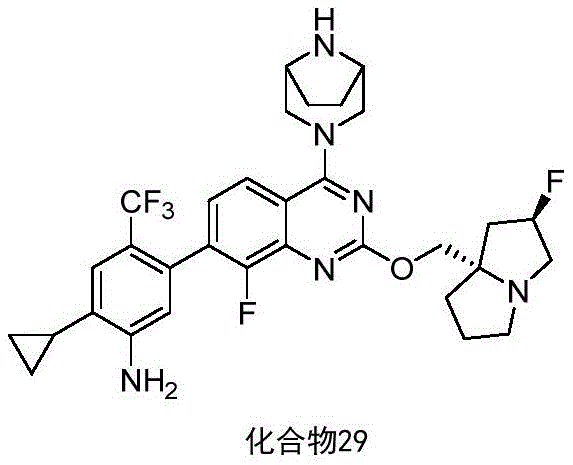
- 229940127204 compound 29 Drugs 0.000 description 3
- 229940125877 compound 31 Drugs 0.000 description 3
- 229940125878 compound 36 Drugs 0.000 description 3
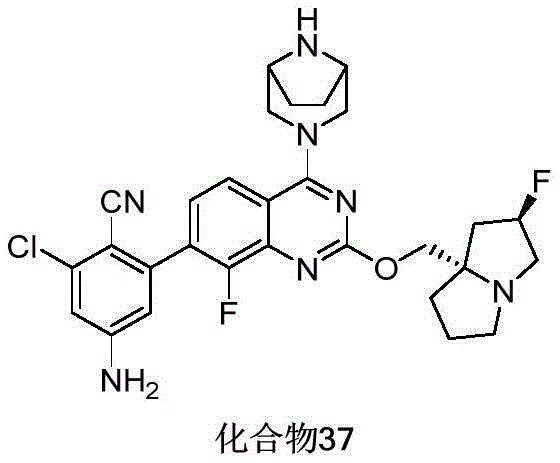
- 229940125807 compound 37 Drugs 0.000 description 3
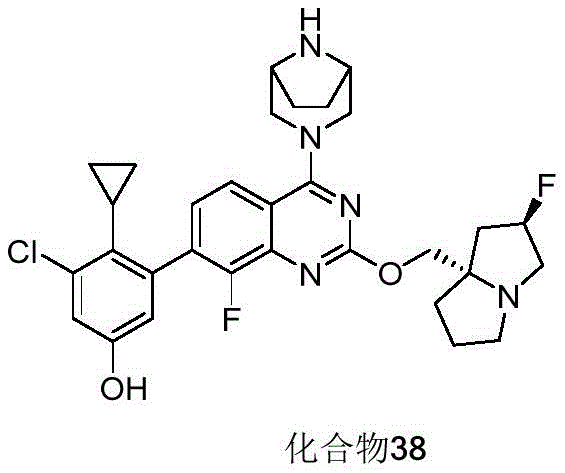
- 229940127573 compound 38 Drugs 0.000 description 3
- 229940125898 compound 5 Drugs 0.000 description 3
- 238000001816 cooling Methods 0.000 description 3
- 239000012043 crude product Substances 0.000 description 3
- GPAYUJZHTULNBE-UHFFFAOYSA-N diphenylphosphine Chemical compound C=1C=CC=CC=1PC1=CC=CC=C1 GPAYUJZHTULNBE-UHFFFAOYSA-N 0.000 description 3
- RTZKZFJDLAIYFH-UHFFFAOYSA-N ether Substances CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 3
- VFRSADQPWYCXDG-LEUCUCNGSA-N ethyl (2s,5s)-5-methylpyrrolidine-2-carboxylate;2,2,2-trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F.CCOC(=O)[C@@H]1CC[C@H](C)N1 VFRSADQPWYCXDG-LEUCUCNGSA-N 0.000 description 3
- KTWOOEGAPBSYNW-UHFFFAOYSA-N ferrocene Chemical compound [Fe+2].C=1C=C[CH-]C=1.C=1C=C[CH-]C=1 KTWOOEGAPBSYNW-UHFFFAOYSA-N 0.000 description 3
- DMEGYFMYUHOHGS-UHFFFAOYSA-N heptamethylene Natural products C1CCCCCC1 DMEGYFMYUHOHGS-UHFFFAOYSA-N 0.000 description 3
- 150000002430 hydrocarbons Chemical group 0.000 description 3
- 239000005457 ice water Substances 0.000 description 3
- 230000002401 inhibitory effect Effects 0.000 description 3
- ZLVXBBHTMQJRSX-VMGNSXQWSA-N jdtic Chemical compound C1([C@]2(C)CCN(C[C@@H]2C)C[C@H](C(C)C)NC(=O)[C@@H]2NCC3=CC(O)=CC=C3C2)=CC=CC(O)=C1 ZLVXBBHTMQJRSX-VMGNSXQWSA-N 0.000 description 3
- 238000004949 mass spectrometry Methods 0.000 description 3
- 239000003921 oil Substances 0.000 description 3
- 235000019198 oils Nutrition 0.000 description 3
- PIDFDZJZLOTZTM-KHVQSSSXSA-N ombitasvir Chemical compound COC(=O)N[C@@H](C(C)C)C(=O)N1CCC[C@H]1C(=O)NC1=CC=C([C@H]2N([C@@H](CC2)C=2C=CC(NC(=O)[C@H]3N(CCC3)C(=O)[C@@H](NC(=O)OC)C(C)C)=CC=2)C=2C=CC(=CC=2)C(C)(C)C)C=C1 PIDFDZJZLOTZTM-KHVQSSSXSA-N 0.000 description 3
- 230000003647 oxidation Effects 0.000 description 3
- 238000007254 oxidation reaction Methods 0.000 description 3
- 125000003367 polycyclic group Chemical group 0.000 description 3
- 239000001294 propane Substances 0.000 description 3
- 108090000623 proteins and genes Proteins 0.000 description 3
- 102000004169 proteins and genes Human genes 0.000 description 3
- 125000003373 pyrazinyl group Chemical group 0.000 description 3
- 238000010791 quenching Methods 0.000 description 3
- 239000011593 sulfur Substances 0.000 description 3
- QAOWNCQODCNURD-UHFFFAOYSA-N sulfuric acid Substances OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 3
- 238000012360 testing method Methods 0.000 description 3
- 238000005406 washing Methods 0.000 description 3
- ASGMFNBUXDJWJJ-JLCFBVMHSA-N (1R,3R)-3-[[3-bromo-1-[4-(5-methyl-1,3,4-thiadiazol-2-yl)phenyl]pyrazolo[3,4-d]pyrimidin-6-yl]amino]-N,1-dimethylcyclopentane-1-carboxamide Chemical compound BrC1=NN(C2=NC(=NC=C21)N[C@H]1C[C@@](CC1)(C(=O)NC)C)C1=CC=C(C=C1)C=1SC(=NN=1)C ASGMFNBUXDJWJJ-JLCFBVMHSA-N 0.000 description 2
- ABJSOROVZZKJGI-OCYUSGCXSA-N (1r,2r,4r)-2-(4-bromophenyl)-n-[(4-chlorophenyl)-(2-fluoropyridin-4-yl)methyl]-4-morpholin-4-ylcyclohexane-1-carboxamide Chemical compound C1=NC(F)=CC(C(NC(=O)[C@H]2[C@@H](C[C@@H](CC2)N2CCOCC2)C=2C=CC(Br)=CC=2)C=2C=CC(Cl)=CC=2)=C1 ABJSOROVZZKJGI-OCYUSGCXSA-N 0.000 description 2
- OWOHLURDBZHNGG-YFKPBYRVSA-N (8ar)-hexahydropyrrolo[1,2-a]pyrazine-1,4-dione Chemical group O=C1CNC(=O)[C@@H]2CCCN12 OWOHLURDBZHNGG-YFKPBYRVSA-N 0.000 description 2
- WORJRXHJTUTINR-UHFFFAOYSA-N 1,4-dioxane;hydron;chloride Chemical compound Cl.C1COCCO1 WORJRXHJTUTINR-UHFFFAOYSA-N 0.000 description 2
- BJAHHTUQMZCNPO-UHFFFAOYSA-N 2-[2-fluoro-6-(methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)naphthalen-1-yl]ethynyl-tri(propan-2-yl)silane Chemical compound B1(OC(C(O1)(C)C)(C)C)C2=CC(=CC3=C2C(=C(C=C3)F)C#C[Si](C(C)C)(C(C)C)C(C)C)OCOC BJAHHTUQMZCNPO-UHFFFAOYSA-N 0.000 description 2
- SFRJQFICUQFDFU-UHFFFAOYSA-N 2-amino-4-bromo-3-fluorobenzoic acid Chemical compound NC1=C(F)C(Br)=CC=C1C(O)=O SFRJQFICUQFDFU-UHFFFAOYSA-N 0.000 description 2
- SCVJRXQHFJXZFZ-KVQBGUIXSA-N 2-amino-9-[(2r,4s,5r)-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-3h-purine-6-thione Chemical compound C1=2NC(N)=NC(=S)C=2N=CN1[C@H]1C[C@H](O)[C@@H](CO)O1 SCVJRXQHFJXZFZ-KVQBGUIXSA-N 0.000 description 2
- JWAZRIHNYRIHIV-UHFFFAOYSA-N 2-naphthol Chemical compound C1=CC=CC2=CC(O)=CC=C21 JWAZRIHNYRIHIV-UHFFFAOYSA-N 0.000 description 2
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 2
- 125000001494 2-propynyl group Chemical group [H]C#CC([H])([H])* 0.000 description 2
- JHUUPUMBZGWODW-UHFFFAOYSA-N 3,6-dihydro-1,2-dioxine Chemical compound C1OOCC=C1 JHUUPUMBZGWODW-UHFFFAOYSA-N 0.000 description 2
- HJTLKVYOWNTDPF-UHFFFAOYSA-N 3-bromo-5-(trifluoromethyl)aniline Chemical compound NC1=CC(Br)=CC(C(F)(F)F)=C1 HJTLKVYOWNTDPF-UHFFFAOYSA-N 0.000 description 2
- SQLHPBJYHIXFHZ-OHAKLNMTSA-N 4-[4-(3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-[[(2R,8S)-2-fluoro-1,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]quinazolin-7-yl]-5-ethynyl-6-fluoronaphthalen-2-ol Chemical compound C#CC(C(C(C(C=CC(C1=N2)=C(N3CC(CC4)NC4C3)N=C2OC[C@](CCC2)(C3)N2C[C@@H]3F)=C1F)=C1)=C(C=C2)C=C1O)=C2F SQLHPBJYHIXFHZ-OHAKLNMTSA-N 0.000 description 2
- OSYGPHDNNWRFJO-UHFFFAOYSA-N 4-amino-2-bromobenzonitrile Chemical compound NC1=CC=C(C#N)C(Br)=C1 OSYGPHDNNWRFJO-UHFFFAOYSA-N 0.000 description 2
- QFGUYMHEJVAZEZ-UHFFFAOYSA-N 4-amino-2-chloro-6-fluorobenzonitrile Chemical compound NC1=CC(F)=C(C#N)C(Cl)=C1 QFGUYMHEJVAZEZ-UHFFFAOYSA-N 0.000 description 2
- VOPLRYDSRVEQDI-UHFFFAOYSA-N 4-bromo-3-chloro-5-fluoroaniline Chemical compound NC1=CC(F)=C(Br)C(Cl)=C1 VOPLRYDSRVEQDI-UHFFFAOYSA-N 0.000 description 2
- QPJKIRNIIXIPIE-UHFFFAOYSA-N 5-bromo-1,3-difluoro-2-(trifluoromethyl)benzene Chemical compound FC1=CC(Br)=CC(F)=C1C(F)(F)F QPJKIRNIIXIPIE-UHFFFAOYSA-N 0.000 description 2
- FSHPSRISCPOKKQ-UHFFFAOYSA-N 7-bromo-5-nitro-1h-indazole Chemical compound [O-][N+](=O)C1=CC(Br)=C2NN=CC2=C1 FSHPSRISCPOKKQ-UHFFFAOYSA-N 0.000 description 2
- KAKZBPTYRLMSJV-UHFFFAOYSA-N Butadiene Chemical compound C=CC=C KAKZBPTYRLMSJV-UHFFFAOYSA-N 0.000 description 2
- DOIHVLZRKCCFDO-YQDLUDSKSA-N C#CC(C(C(C1=NC2=NC(OC[C@](CCC3)(C4)N3C[C@@H]4F)=NC(N3CC(CC4)NC4C3)=C2C=C1)=C1)=C(C=C2)C=C1O)=C2F Chemical compound C#CC(C(C(C1=NC2=NC(OC[C@](CCC3)(C4)N3C[C@@H]4F)=NC(N3CC(CC4)NC4C3)=C2C=C1)=C1)=C(C=C2)C=C1O)=C2F DOIHVLZRKCCFDO-YQDLUDSKSA-N 0.000 description 2
- GPPATPDJEXSEHA-UHFFFAOYSA-N CC1(C)OB(C2=CC(N)=C(C(F)(F)F)C(F)=C2)OC1(C)C Chemical compound CC1(C)OB(C2=CC(N)=C(C(F)(F)F)C(F)=C2)OC1(C)C GPPATPDJEXSEHA-UHFFFAOYSA-N 0.000 description 2
- RTZRCYXHWAMBLD-GUEIHZCPSA-N CCC1=C2C(C3=CC=C4C(N5C[C@H](CC6)N[C@H]6C5)=NC(OC[C@](CCC5)(C6)N5C[C@@H]6F)=NC4=C3F)=CC(O)=CC2=CC=C1F Chemical compound CCC1=C2C(C3=CC=C4C(N5C[C@H](CC6)N[C@H]6C5)=NC(OC[C@](CCC5)(C6)N5C[C@@H]6F)=NC4=C3F)=CC(O)=CC2=CC=C1F RTZRCYXHWAMBLD-GUEIHZCPSA-N 0.000 description 2
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical class [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 2
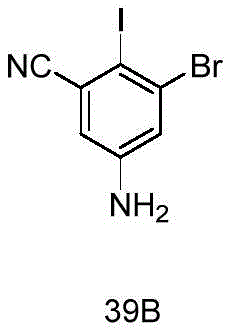
- 229940127007 Compound 39 Drugs 0.000 description 2
- RGSFGYAAUTVSQA-UHFFFAOYSA-N Cyclopentane Chemical compound C1CCCC1 RGSFGYAAUTVSQA-UHFFFAOYSA-N 0.000 description 2
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 2
- 206010064571 Gene mutation Diseases 0.000 description 2
- 108010067218 Guanine Nucleotide Exchange Factors Proteins 0.000 description 2
- 102000016285 Guanine Nucleotide Exchange Factors Human genes 0.000 description 2
- 239000007821 HATU Substances 0.000 description 2
- 241000282412 Homo Species 0.000 description 2
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 2
- 239000005909 Kieselgur Substances 0.000 description 2
- BZLVMXJERCGZMT-UHFFFAOYSA-N Methyl tert-butyl ether Chemical compound COC(C)(C)C BZLVMXJERCGZMT-UHFFFAOYSA-N 0.000 description 2
- QMEWGPDOWWAQFF-QFVACWOZSA-N OC1=CC(C2=CC=C3C(N4C[C@H](CC5)N[C@H]5C4)=NC(OC[C@](CCC4)(C5)N4C[C@@H]5F)=NC3=C2F)=C(C2CC2)C(Cl)=C1 Chemical compound OC1=CC(C2=CC=C3C(N4C[C@H](CC5)N[C@H]5C4)=NC(OC[C@](CCC4)(C5)N4C[C@@H]5F)=NC3=C2F)=C(C2CC2)C(Cl)=C1 QMEWGPDOWWAQFF-QFVACWOZSA-N 0.000 description 2
- 108700020796 Oncogene Proteins 0.000 description 2
- SMWDFEZZVXVKRB-UHFFFAOYSA-N Quinoline Chemical compound N1=CC=CC2=CC=CC=C21 SMWDFEZZVXVKRB-UHFFFAOYSA-N 0.000 description 2
- 206010039491 Sarcoma Diseases 0.000 description 2
- DKGAVHZHDRPRBM-UHFFFAOYSA-N Tert-Butanol Chemical compound CC(C)(C)O DKGAVHZHDRPRBM-UHFFFAOYSA-N 0.000 description 2
- 230000004913 activation Effects 0.000 description 2
- ORILYTVJVMAKLC-UHFFFAOYSA-N adamantane Chemical compound C1C(C2)CC3CC1CC2C3 ORILYTVJVMAKLC-UHFFFAOYSA-N 0.000 description 2
- 125000002029 aromatic hydrocarbon group Chemical group 0.000 description 2
- 125000002393 azetidinyl group Chemical group 0.000 description 2
- 125000001584 benzyloxycarbonyl group Chemical group C(=O)(OCC1=CC=CC=C1)* 0.000 description 2
- 125000002619 bicyclic group Chemical group 0.000 description 2
- 230000004071 biological effect Effects 0.000 description 2
- ZADPBFCGQRWHPN-UHFFFAOYSA-N boronic acid Chemical compound OBO ZADPBFCGQRWHPN-UHFFFAOYSA-N 0.000 description 2
- 150000001721 carbon Chemical group 0.000 description 2
- 239000011203 carbon fibre reinforced carbon Substances 0.000 description 2
- 230000010261 cell growth Effects 0.000 description 2
- 229940126214 compound 3 Drugs 0.000 description 2
- DOBRDRYODQBAMW-UHFFFAOYSA-N copper(i) cyanide Chemical compound [Cu+].N#[C-] DOBRDRYODQBAMW-UHFFFAOYSA-N 0.000 description 2
- 125000000596 cyclohexenyl group Chemical group C1(=CCCCC1)* 0.000 description 2
- 238000001514 detection method Methods 0.000 description 2
- 150000002148 esters Chemical class 0.000 description 2
- 238000000605 extraction Methods 0.000 description 2
- 239000012458 free base Substances 0.000 description 2
- 125000004404 heteroalkyl group Chemical group 0.000 description 2
- 150000002431 hydrogen Chemical class 0.000 description 2
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 2
- 238000001727 in vivo Methods 0.000 description 2
- 238000006317 isomerization reaction Methods 0.000 description 2
- HNQIVZYLYMDVSB-UHFFFAOYSA-N methanesulfonimidic acid Chemical compound CS(N)(=O)=O HNQIVZYLYMDVSB-UHFFFAOYSA-N 0.000 description 2
- GQJCAQADCPTHKN-UHFFFAOYSA-N methyl 2,2-difluoro-2-fluorosulfonylacetate Chemical compound COC(=O)C(F)(F)S(F)(=O)=O GQJCAQADCPTHKN-UHFFFAOYSA-N 0.000 description 2
- 239000000203 mixture Substances 0.000 description 2
- 125000002757 morpholinyl group Chemical group 0.000 description 2
- VMGAPWLDMVPYIA-HIDZBRGKSA-N n'-amino-n-iminomethanimidamide Chemical group N\N=C\N=N VMGAPWLDMVPYIA-HIDZBRGKSA-N 0.000 description 2
- 230000007935 neutral effect Effects 0.000 description 2
- 231100000252 nontoxic Toxicity 0.000 description 2
- 230000003000 nontoxic effect Effects 0.000 description 2
- 125000003566 oxetanyl group Chemical group 0.000 description 2
- 239000000546 pharmaceutical excipient Substances 0.000 description 2
- 125000004193 piperazinyl group Chemical group 0.000 description 2
- 125000003386 piperidinyl group Chemical group 0.000 description 2
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 2
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 2
- 125000002098 pyridazinyl group Chemical group 0.000 description 2
- 125000000714 pyrimidinyl group Chemical group 0.000 description 2
- RQGPLDBZHMVWCH-UHFFFAOYSA-N pyrrolo[3,2-b]pyrrole Chemical group C1=NC2=CC=NC2=C1 RQGPLDBZHMVWCH-UHFFFAOYSA-N 0.000 description 2
- 230000002441 reversible effect Effects 0.000 description 2
- 229910000029 sodium carbonate Inorganic materials 0.000 description 2
- LPXPTNMVRIOKMN-UHFFFAOYSA-M sodium nitrite Chemical compound [Na+].[O-]N=O LPXPTNMVRIOKMN-UHFFFAOYSA-M 0.000 description 2
- HNINFCBLGHCFOJ-UHFFFAOYSA-N tert-butyl 3,8-diazabicyclo[3.2.1]octane-8-carboxylate Chemical compound C1NCC2CCC1N2C(=O)OC(C)(C)C HNINFCBLGHCFOJ-UHFFFAOYSA-N 0.000 description 2
- 125000003718 tetrahydrofuranyl group Chemical group 0.000 description 2
- CZDYPVPMEAXLPK-UHFFFAOYSA-N tetramethylsilane Chemical group C[Si](C)(C)C CZDYPVPMEAXLPK-UHFFFAOYSA-N 0.000 description 2
- 229930195735 unsaturated hydrocarbon Natural products 0.000 description 2
- 230000003612 virological effect Effects 0.000 description 2
- CYPYTURSJDMMMP-WVCUSYJESA-N (1e,4e)-1,5-diphenylpenta-1,4-dien-3-one;palladium Chemical compound [Pd].[Pd].C=1C=CC=CC=1\C=C\C(=O)\C=C\C1=CC=CC=C1.C=1C=CC=CC=1\C=C\C(=O)\C=C\C1=CC=CC=C1.C=1C=CC=CC=1\C=C\C(=O)\C=C\C1=CC=CC=C1 CYPYTURSJDMMMP-WVCUSYJESA-N 0.000 description 1
- QQLRSCZSKQTFGY-UHFFFAOYSA-N (2,4-difluorophenyl)boronic acid Chemical compound OB(O)C1=CC=C(F)C=C1F QQLRSCZSKQTFGY-UHFFFAOYSA-N 0.000 description 1
- IWZSHWBGHQBIML-ZGGLMWTQSA-N (3S,8S,10R,13S,14S,17S)-17-isoquinolin-7-yl-N,N,10,13-tetramethyl-2,3,4,7,8,9,11,12,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-3-amine Chemical compound CN(C)[C@H]1CC[C@]2(C)C3CC[C@@]4(C)[C@@H](CC[C@@H]4c4ccc5ccncc5c4)[C@@H]3CC=C2C1 IWZSHWBGHQBIML-ZGGLMWTQSA-N 0.000 description 1
- PRBHEGAFLDMLAL-GQCTYLIASA-N (4e)-hexa-1,4-diene Chemical compound C\C=C\CC=C PRBHEGAFLDMLAL-GQCTYLIASA-N 0.000 description 1
- 125000006714 (C3-C10) heterocyclyl group Chemical group 0.000 description 1
- PMJHHCWVYXUKFD-SNAWJCMRSA-N (E)-1,3-pentadiene Chemical compound C\C=C\C=C PMJHHCWVYXUKFD-SNAWJCMRSA-N 0.000 description 1
- YCTDZYMMFQCTEO-FNORWQNLSA-N (E)-3-octene Chemical compound CCCC\C=C\CC YCTDZYMMFQCTEO-FNORWQNLSA-N 0.000 description 1
- IGERFAHWSHDDHX-UHFFFAOYSA-N 1,3-dioxanyl Chemical group [CH]1OCCCO1 IGERFAHWSHDDHX-UHFFFAOYSA-N 0.000 description 1
- JPRPJUMQRZTTED-UHFFFAOYSA-N 1,3-dioxolanyl Chemical group [CH]1OCCO1 JPRPJUMQRZTTED-UHFFFAOYSA-N 0.000 description 1
- ILWJAOPQHOZXAN-UHFFFAOYSA-N 1,3-dithianyl Chemical group [CH]1SCCCS1 ILWJAOPQHOZXAN-UHFFFAOYSA-N 0.000 description 1
- HAICRZPDDBKXPQ-UHFFFAOYSA-N 1-benzofuran-7-carbonitrile Chemical compound N#CC1=CC=CC2=C1OC=C2 HAICRZPDDBKXPQ-UHFFFAOYSA-N 0.000 description 1
- 125000004973 1-butenyl group Chemical group C(=CCC)* 0.000 description 1
- 125000004972 1-butynyl group Chemical group [H]C([H])([H])C([H])([H])C#C* 0.000 description 1
- 125000006039 1-hexenyl group Chemical group 0.000 description 1
- 125000006025 1-methyl-1-butenyl group Chemical group 0.000 description 1
- 125000006044 1-methyl-1-pentenyl group Chemical group 0.000 description 1
- 125000006023 1-pentenyl group Chemical group 0.000 description 1
- 125000006017 1-propenyl group Chemical group 0.000 description 1
- 125000000530 1-propynyl group Chemical group [H]C([H])([H])C#C* 0.000 description 1
- HYZJCKYKOHLVJF-UHFFFAOYSA-N 1H-benzimidazole Chemical compound C1=CC=C2NC=NC2=C1 HYZJCKYKOHLVJF-UHFFFAOYSA-N 0.000 description 1
- XWIYUCRMWCHYJR-UHFFFAOYSA-N 1h-pyrrolo[3,2-b]pyridine Chemical compound C1=CC=C2NC=CC2=N1 XWIYUCRMWCHYJR-UHFFFAOYSA-N 0.000 description 1
- ATFYJCGWEXYXNI-UHFFFAOYSA-N 2,3,4,5-tetrafluoro-6-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)aniline Chemical compound FC1=C(N)C(=C(C(=C1F)F)F)B1OC(C(O1)(C)C)(C)C ATFYJCGWEXYXNI-UHFFFAOYSA-N 0.000 description 1
- WRDGNXCXTDDYBZ-UHFFFAOYSA-N 2,3,4-trifluoroaniline Chemical compound NC1=CC=C(F)C(F)=C1F WRDGNXCXTDDYBZ-UHFFFAOYSA-N 0.000 description 1
- DNFDLCRLLQVUQK-UHFFFAOYSA-N 2,4,7-trichloropyrido[2,3-d]pyrimidine Chemical compound ClC1=NC(Cl)=NC2=NC(Cl)=CC=C21 DNFDLCRLLQVUQK-UHFFFAOYSA-N 0.000 description 1
- JECYNCQXXKQDJN-UHFFFAOYSA-N 2-(2-methylhexan-2-yloxymethyl)oxirane Chemical compound CCCCC(C)(C)OCC1CO1 JECYNCQXXKQDJN-UHFFFAOYSA-N 0.000 description 1
- QLVGHFBUSGYCCG-UHFFFAOYSA-N 2-amino-n-(1-cyano-2-phenylethyl)acetamide Chemical compound NCC(=O)NC(C#N)CC1=CC=CC=C1 QLVGHFBUSGYCCG-UHFFFAOYSA-N 0.000 description 1
- 125000004974 2-butenyl group Chemical group C(C=CC)* 0.000 description 1
- 125000000069 2-butynyl group Chemical group [H]C([H])([H])C#CC([H])([H])* 0.000 description 1
- 125000006040 2-hexenyl group Chemical group 0.000 description 1
- 125000006045 2-methyl-1-pentenyl group Chemical group 0.000 description 1
- 125000006031 2-methyl-3-butenyl group Chemical group 0.000 description 1
- 125000006024 2-pentenyl group Chemical group 0.000 description 1
- ASYONLUGMHMMDA-UHFFFAOYSA-N 2h-thieno[3,2-b]pyrrole Chemical group C1=NC2=CCSC2=C1 ASYONLUGMHMMDA-UHFFFAOYSA-N 0.000 description 1
- 125000004975 3-butenyl group Chemical group C(CC=C)* 0.000 description 1
- 125000000474 3-butynyl group Chemical group [H]C#CC([H])([H])C([H])([H])* 0.000 description 1
- 125000006041 3-hexenyl group Chemical group 0.000 description 1
- YLEJPSKVIOETSC-UHFFFAOYSA-N 4,4,5,5-tetramethyl-2-[2-methyl-3-(trifluoromethyl)phenyl]-1,3,2-dioxaborolane Chemical compound C1=CC=C(C(F)(F)F)C(C)=C1B1OC(C)(C)C(C)(C)O1 YLEJPSKVIOETSC-UHFFFAOYSA-N 0.000 description 1
- KGDMVKJUKFLWST-UHFFFAOYSA-N 4-amino-2-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzonitrile Chemical compound O1C(C)(C)C(C)(C)OB1C1=CC(N)=CC=C1C#N KGDMVKJUKFLWST-UHFFFAOYSA-N 0.000 description 1
- 125000006042 4-hexenyl group Chemical group 0.000 description 1
- 125000002373 5 membered heterocyclic group Chemical group 0.000 description 1
- 125000006043 5-hexenyl group Chemical group 0.000 description 1
- 125000006163 5-membered heteroaryl group Chemical group 0.000 description 1
- BSVYLJCOTFYPSN-UHFFFAOYSA-N 5h-furo[3,2-c]pyrazole Chemical group N1=NC2=CCOC2=C1 BSVYLJCOTFYPSN-UHFFFAOYSA-N 0.000 description 1
- RDHOEMPRFDWIHL-UHFFFAOYSA-N 5h-thieno[3,2-c]pyrazole Chemical group N1=NC2=CCSC2=C1 RDHOEMPRFDWIHL-UHFFFAOYSA-N 0.000 description 1
- 125000004070 6 membered heterocyclic group Chemical group 0.000 description 1
- YLLZDOJZLKUKEA-UHFFFAOYSA-N 6-bromo-5-methyl-1h-indazole Chemical compound C1=C(Br)C(C)=CC2=C1NN=C2 YLLZDOJZLKUKEA-UHFFFAOYSA-N 0.000 description 1
- 125000003341 7 membered heterocyclic group Chemical group 0.000 description 1
- QFMMOOSPRNOBFO-UHFFFAOYSA-N 7-bromo-6-chloro-8-fluoro-1H-quinazoline-2,4-dione Chemical compound Fc1c(Br)c(Cl)cc2c1[nH]c(=O)[nH]c2=O QFMMOOSPRNOBFO-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 1
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 1
- BTBUEUYNUDRHOZ-UHFFFAOYSA-N Borate Chemical compound [O-]B([O-])[O-] BTBUEUYNUDRHOZ-UHFFFAOYSA-N 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- 239000005489 Bromoxynil Substances 0.000 description 1
- QDWMWTOQRFNADL-UHFFFAOYSA-N CC(C)(C)OC(NC(C=C1Cl)=CC(Br)=C1I)=O Chemical compound CC(C)(C)OC(NC(C=C1Cl)=CC(Br)=C1I)=O QDWMWTOQRFNADL-UHFFFAOYSA-N 0.000 description 1
- KOWFLPHPPTWXGY-UHFFFAOYSA-N COC(CCCCCOOC1(C(C1(OCCCC)OC(C)C)(OC(C)(C)C)OC(C)CC)OCCCCC)(OCCC)OCC Chemical compound COC(CCCCCOOC1(C(C1(OCCCC)OC(C)C)(OC(C)(C)C)OC(C)CC)OCCCCC)(OCCC)OCC KOWFLPHPPTWXGY-UHFFFAOYSA-N 0.000 description 1
- 241000282472 Canis lupus familiaris Species 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- 108020004705 Codon Proteins 0.000 description 1
- 206010009944 Colon cancer Diseases 0.000 description 1
- XFXPMWWXUTWYJX-UHFFFAOYSA-N Cyanide Chemical compound N#[C-] XFXPMWWXUTWYJX-UHFFFAOYSA-N 0.000 description 1
- PMPVIKIVABFJJI-UHFFFAOYSA-N Cyclobutane Chemical compound C1CCC1 PMPVIKIVABFJJI-UHFFFAOYSA-N 0.000 description 1
- XDTMQSROBMDMFD-UHFFFAOYSA-N Cyclohexane Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 description 1
- LVZWSLJZHVFIQJ-UHFFFAOYSA-N Cyclopropane Chemical compound C1CC1 LVZWSLJZHVFIQJ-UHFFFAOYSA-N 0.000 description 1
- IAZDPXIOMUYVGZ-WFGJKAKNSA-N Dimethyl sulfoxide Chemical group [2H]C([2H])([2H])S(=O)C([2H])([2H])[2H] IAZDPXIOMUYVGZ-WFGJKAKNSA-N 0.000 description 1
- SNRUBQQJIBEYMU-UHFFFAOYSA-N Dodecane Natural products CCCCCCCCCCCC SNRUBQQJIBEYMU-UHFFFAOYSA-N 0.000 description 1
- 206010014733 Endometrial cancer Diseases 0.000 description 1
- 206010014759 Endometrial neoplasm Diseases 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 241000283086 Equidae Species 0.000 description 1
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 1
- BDAGIHXWWSANSR-UHFFFAOYSA-M Formate Chemical compound [O-]C=O BDAGIHXWWSANSR-UHFFFAOYSA-M 0.000 description 1
- 102000034286 G proteins Human genes 0.000 description 1
- 108091006027 G proteins Proteins 0.000 description 1
- 102000013446 GTP Phosphohydrolases Human genes 0.000 description 1
- 102000018898 GTPase-Activating Proteins Human genes 0.000 description 1
- 108091006094 GTPase-accelerating proteins Proteins 0.000 description 1
- 108091006109 GTPases Proteins 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- 206010069755 K-ras gene mutation Diseases 0.000 description 1
- 108091007984 KARS Proteins 0.000 description 1
- 229940126204 KRAS G12D inhibitor Drugs 0.000 description 1
- 101150105104 Kras gene Proteins 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- 239000012359 Methanesulfonyl chloride Substances 0.000 description 1
- 229920000168 Microcrystalline cellulose Polymers 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- 206010061902 Pancreatic neoplasm Diseases 0.000 description 1
- YNHIGQDRGKUECZ-UHFFFAOYSA-L PdCl2(PPh3)2 Substances [Cl-].[Cl-].[Pd+2].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 YNHIGQDRGKUECZ-UHFFFAOYSA-L 0.000 description 1
- YZCKVEUIGOORGS-IGMARMGPSA-N Protium Chemical compound [1H] YZCKVEUIGOORGS-IGMARMGPSA-N 0.000 description 1
- 241000720974 Protium Species 0.000 description 1
- UIIMBOGNXHQVGW-DEQYMQKBSA-M Sodium bicarbonate-14C Chemical class [Na+].O[14C]([O-])=O UIIMBOGNXHQVGW-DEQYMQKBSA-M 0.000 description 1
- 229920002472 Starch Chemical class 0.000 description 1
- OJFKUJDRGJSAQB-UHFFFAOYSA-N TAK-632 Chemical compound C1=C(NC(=O)CC=2C=C(C=CC=2)C(F)(F)F)C(F)=CC=C1OC(C(=C1S2)C#N)=CC=C1N=C2NC(=O)C1CC1 OJFKUJDRGJSAQB-UHFFFAOYSA-N 0.000 description 1
- DHXVGJBLRPWPCS-UHFFFAOYSA-N Tetrahydropyran Chemical compound C1CCOCC1 DHXVGJBLRPWPCS-UHFFFAOYSA-N 0.000 description 1
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical class [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 1
- 239000007983 Tris buffer Substances 0.000 description 1
- YZCKVEUIGOORGS-NJFSPNSNSA-N Tritium Chemical compound [3H] YZCKVEUIGOORGS-NJFSPNSNSA-N 0.000 description 1
- WETWJCDKMRHUPV-UHFFFAOYSA-N acetyl chloride Chemical compound CC(Cl)=O WETWJCDKMRHUPV-UHFFFAOYSA-N 0.000 description 1
- 239000012346 acetyl chloride Substances 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 125000002252 acyl group Chemical group 0.000 description 1
- 208000009956 adenocarcinoma Diseases 0.000 description 1
- 150000001299 aldehydes Chemical class 0.000 description 1
- 125000001931 aliphatic group Chemical group 0.000 description 1
- 150000001336 alkenes Chemical class 0.000 description 1
- MDFFNEOEWAXZRQ-UHFFFAOYSA-N aminyl Chemical compound [NH2] MDFFNEOEWAXZRQ-UHFFFAOYSA-N 0.000 description 1
- 235000011114 ammonium hydroxide Nutrition 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 125000005110 aryl thio group Chemical group 0.000 description 1
- METKIMKYRPQLGS-UHFFFAOYSA-N atenolol Chemical compound CC(C)NCC(O)COC1=CC=C(CC(N)=O)C=C1 METKIMKYRPQLGS-UHFFFAOYSA-N 0.000 description 1
- CBHOOMGKXCMKIR-UHFFFAOYSA-N azane;methanol Chemical compound N.OC CBHOOMGKXCMKIR-UHFFFAOYSA-N 0.000 description 1
- 125000004541 benzoxazolyl group Chemical group O1C(=NC2=C1C=CC=C2)* 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 230000031018 biological processes and functions Effects 0.000 description 1
- 239000004305 biphenyl Substances 0.000 description 1
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 1
- OSVHLUXLWQLPIY-KBAYOESNSA-N butyl 2-[(6aR,9R,10aR)-1-hydroxy-9-(hydroxymethyl)-6,6-dimethyl-6a,7,8,9,10,10a-hexahydrobenzo[c]chromen-3-yl]-2-methylpropanoate Chemical compound C(CCC)OC(C(C)(C)C1=CC(=C2[C@H]3[C@H](C(OC2=C1)(C)C)CC[C@H](C3)CO)O)=O OSVHLUXLWQLPIY-KBAYOESNSA-N 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- 235000010216 calcium carbonate Nutrition 0.000 description 1
- 239000001506 calcium phosphate Chemical class 0.000 description 1
- 229910000389 calcium phosphate Inorganic materials 0.000 description 1
- 235000011010 calcium phosphates Nutrition 0.000 description 1
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 1
- 150000001732 carboxylic acid derivatives Chemical class 0.000 description 1
- 210000004027 cell Anatomy 0.000 description 1
- 239000001913 cellulose Chemical class 0.000 description 1
- 229920002678 cellulose Chemical class 0.000 description 1
- 235000010980 cellulose Nutrition 0.000 description 1
- 239000012295 chemical reaction liquid Substances 0.000 description 1
- 239000007810 chemical reaction solvent Substances 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 208000029742 colonic neoplasm Diseases 0.000 description 1
- 239000012230 colorless oil Substances 0.000 description 1
- 229940125796 compound 3d Drugs 0.000 description 1
- LSXDOTMGLUJQCM-UHFFFAOYSA-M copper(i) iodide Chemical compound I[Cu] LSXDOTMGLUJQCM-UHFFFAOYSA-M 0.000 description 1
- TXWRERCHRDBNLG-UHFFFAOYSA-N cubane Chemical compound C12C3C4C1C1C4C3C12 TXWRERCHRDBNLG-UHFFFAOYSA-N 0.000 description 1
- 150000001924 cycloalkanes Chemical class 0.000 description 1
- 125000001352 cyclobutyloxy group Chemical group C1(CCC1)O* 0.000 description 1
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- UZBHNSVUMGIKLU-UHFFFAOYSA-N cyclopenten-1-ylboronic acid Chemical compound OB(O)C1=CCCC1 UZBHNSVUMGIKLU-UHFFFAOYSA-N 0.000 description 1
- 210000004292 cytoskeleton Anatomy 0.000 description 1
- 230000004069 differentiation Effects 0.000 description 1
- 125000004852 dihydrofuranyl group Chemical group O1C(CC=C1)* 0.000 description 1
- 125000005043 dihydropyranyl group Chemical group O1C(CCC=C1)* 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 125000000532 dioxanyl group Chemical group 0.000 description 1
- 125000005879 dioxolanyl group Chemical group 0.000 description 1
- MKRTXPORKIRPDG-UHFFFAOYSA-N diphenylphosphoryl azide Chemical compound C=1C=CC=CC=1P(=O)(N=[N+]=[N-])C1=CC=CC=C1 MKRTXPORKIRPDG-UHFFFAOYSA-N 0.000 description 1
- 239000007884 disintegrant Substances 0.000 description 1
- 125000003438 dodecyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- QHQKEFQTKKSZEH-GTFNEXEASA-N e-8-carboxylate Chemical compound O=C1[C@H](C(C)(C)C)NC(=O)O[C@@H]2C[C@H]2CCCCCC2=NC3=CC=C(OC)C=C3N=C2O[C@@H]2C[C@@H](C(=O)OC)N1C2 QHQKEFQTKKSZEH-GTFNEXEASA-N 0.000 description 1
- 239000012636 effector Substances 0.000 description 1
- 125000004494 ethyl ester group Chemical group 0.000 description 1
- OJCSPXHYDFONPU-UHFFFAOYSA-N etoac etoac Chemical compound CCOC(C)=O.CCOC(C)=O OJCSPXHYDFONPU-UHFFFAOYSA-N 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 125000000524 functional group Chemical group 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 239000003979 granulating agent Substances 0.000 description 1
- 125000001188 haloalkyl group Chemical group 0.000 description 1
- 125000005843 halogen group Chemical group 0.000 description 1
- 125000004474 heteroalkylene group Chemical group 0.000 description 1
- 125000005343 heterocyclic alkyl group Chemical group 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- 125000002768 hydroxyalkyl group Chemical group 0.000 description 1
- 125000002632 imidazolidinyl group Chemical group 0.000 description 1
- 150000007529 inorganic bases Chemical class 0.000 description 1
- 230000003834 intracellular effect Effects 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- HVTICUPFWKNHNG-UHFFFAOYSA-N iodoethane Chemical compound CCI HVTICUPFWKNHNG-UHFFFAOYSA-N 0.000 description 1
- 230000007794 irritation Effects 0.000 description 1
- 125000002462 isocyano group Chemical group *[N+]#[C-] 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- FMKOJHQHASLBPH-UHFFFAOYSA-N isopropyl iodide Chemical compound CC(C)I FMKOJHQHASLBPH-UHFFFAOYSA-N 0.000 description 1
- 239000003446 ligand Substances 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 208000015486 malignant pancreatic neoplasm Diseases 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- QARBMVPHQWIHKH-UHFFFAOYSA-N methanesulfonyl chloride Chemical compound CS(Cl)(=O)=O QARBMVPHQWIHKH-UHFFFAOYSA-N 0.000 description 1
- KTMKRRPZPWUYKK-UHFFFAOYSA-N methylboronic acid Chemical compound CB(O)O KTMKRRPZPWUYKK-UHFFFAOYSA-N 0.000 description 1
- GRVDJDISBSALJP-UHFFFAOYSA-N methyloxidanyl Chemical compound [O]C GRVDJDISBSALJP-UHFFFAOYSA-N 0.000 description 1
- 235000019813 microcrystalline cellulose Nutrition 0.000 description 1
- 239000008108 microcrystalline cellulose Substances 0.000 description 1
- 229940016286 microcrystalline cellulose Drugs 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- NKCCODPFBDGPRJ-UHFFFAOYSA-N nitridocarbon(1+) Chemical compound N#[C+] NKCCODPFBDGPRJ-UHFFFAOYSA-N 0.000 description 1
- 208000002154 non-small cell lung carcinoma Diseases 0.000 description 1
- 125000001400 nonyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 150000007530 organic bases Chemical class 0.000 description 1
- 125000001181 organosilyl group Chemical group [SiH3]* 0.000 description 1
- 125000000160 oxazolidinyl group Chemical group 0.000 description 1
- NFHFRUOZVGFOOS-UHFFFAOYSA-N palladium;triphenylphosphane Chemical compound [Pd].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 NFHFRUOZVGFOOS-UHFFFAOYSA-N 0.000 description 1
- 201000002528 pancreatic cancer Diseases 0.000 description 1
- 208000008443 pancreatic carcinoma Diseases 0.000 description 1
- QYZLKGVUSQXAMU-UHFFFAOYSA-N penta-1,4-diene Chemical compound C=CCC=C QYZLKGVUSQXAMU-UHFFFAOYSA-N 0.000 description 1
- 239000003208 petroleum Substances 0.000 description 1
- DHHVAGZRUROJKS-UHFFFAOYSA-N phentermine Chemical compound CC(C)(N)CC1=CC=CC=C1 DHHVAGZRUROJKS-UHFFFAOYSA-N 0.000 description 1
- UYWQUFXKFGHYNT-UHFFFAOYSA-N phenylmethyl ester of formic acid Natural products O=COCC1=CC=CC=C1 UYWQUFXKFGHYNT-UHFFFAOYSA-N 0.000 description 1
- FAIAAWCVCHQXDN-UHFFFAOYSA-N phosphorus trichloride Chemical compound ClP(Cl)Cl FAIAAWCVCHQXDN-UHFFFAOYSA-N 0.000 description 1
- PMJHHCWVYXUKFD-UHFFFAOYSA-N piperylene Natural products CC=CC=C PMJHHCWVYXUKFD-UHFFFAOYSA-N 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 229910000027 potassium carbonate Inorganic materials 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 230000035755 proliferation Effects 0.000 description 1
- QQONPFPTGQHPMA-UHFFFAOYSA-N propylene Natural products CC=C QQONPFPTGQHPMA-UHFFFAOYSA-N 0.000 description 1
- 229940121649 protein inhibitor Drugs 0.000 description 1
- 239000012268 protein inhibitor Substances 0.000 description 1
- 125000004309 pyranyl group Chemical group O1C(C=CC=C1)* 0.000 description 1
- VNAUDIIOSMNXBA-UHFFFAOYSA-N pyrazolo[4,3-c]pyrazole Chemical group N1=NC=C2N=NC=C21 VNAUDIIOSMNXBA-UHFFFAOYSA-N 0.000 description 1
- 125000001725 pyrenyl group Chemical group 0.000 description 1
- 125000000719 pyrrolidinyl group Chemical group 0.000 description 1
- GZTPJDLYPMPRDF-UHFFFAOYSA-N pyrrolo[3,2-c]pyrazole Chemical group N1=NC2=CC=NC2=C1 GZTPJDLYPMPRDF-UHFFFAOYSA-N 0.000 description 1
- 125000006085 pyrrolopyridyl group Chemical group 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- BOLDJAUMGUJJKM-LSDHHAIUSA-N renifolin D Natural products CC(=C)[C@@H]1Cc2c(O)c(O)ccc2[C@H]1CC(=O)c3ccc(O)cc3O BOLDJAUMGUJJKM-LSDHHAIUSA-N 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 239000013557 residual solvent Substances 0.000 description 1
- 102200006531 rs121913529 Human genes 0.000 description 1
- 102200006538 rs121913530 Human genes 0.000 description 1
- 229930195734 saturated hydrocarbon Natural products 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 230000028327 secretion Effects 0.000 description 1
- 230000019491 signal transduction Effects 0.000 description 1
- 230000011664 signaling Effects 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- 235000017557 sodium bicarbonate Nutrition 0.000 description 1
- 235000010288 sodium nitrite Nutrition 0.000 description 1
- 229910052938 sodium sulfate Inorganic materials 0.000 description 1
- 235000011152 sodium sulphate Nutrition 0.000 description 1
- AKHNMLFCWUSKQB-UHFFFAOYSA-L sodium thiosulfate Chemical class [Na+].[Na+].[O-]S([O-])(=O)=S AKHNMLFCWUSKQB-UHFFFAOYSA-L 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 235000000346 sugar Nutrition 0.000 description 1
- 150000008163 sugars Chemical class 0.000 description 1
- 125000000472 sulfonyl group Chemical group *S(*)(=O)=O 0.000 description 1
- 125000005931 tert-butyloxycarbonyl group Chemical group [H]C([H])([H])C(OC(*)=O)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- JRMUNVKIHCOMHV-UHFFFAOYSA-M tetrabutylammonium bromide Chemical compound [Br-].CCCC[N+](CCCC)(CCCC)CCCC JRMUNVKIHCOMHV-UHFFFAOYSA-M 0.000 description 1
- 125000001412 tetrahydropyranyl group Chemical group 0.000 description 1
- 125000003554 tetrahydropyrrolyl group Chemical group 0.000 description 1
- 125000000383 tetramethylene group Chemical group [H]C([H])([*:1])C([H])([H])C([H])([H])C([H])([H])[*:2] 0.000 description 1
- 229940124597 therapeutic agent Drugs 0.000 description 1
- 125000002813 thiocarbonyl group Chemical group *C(*)=S 0.000 description 1
- 150000003573 thiols Chemical class 0.000 description 1
- 210000001519 tissue Anatomy 0.000 description 1
- 230000032258 transport Effects 0.000 description 1
- ZGYICYBLPGRURT-UHFFFAOYSA-N tri(propan-2-yl)silicon Chemical compound CC(C)[Si](C(C)C)C(C)C ZGYICYBLPGRURT-UHFFFAOYSA-N 0.000 description 1
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical class [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 1
- ITMCEJHCFYSIIV-UHFFFAOYSA-N triflic acid Chemical compound OS(=O)(=O)C(F)(F)F ITMCEJHCFYSIIV-UHFFFAOYSA-N 0.000 description 1
- 125000001889 triflyl group Chemical group FC(F)(F)S(*)(=O)=O 0.000 description 1
- 125000000026 trimethylsilyl group Chemical group [H]C([H])([H])[Si]([*])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- UCPYLLCMEDAXFR-UHFFFAOYSA-N triphosgene Chemical compound ClC(Cl)(Cl)OC(=O)OC(Cl)(Cl)Cl UCPYLLCMEDAXFR-UHFFFAOYSA-N 0.000 description 1
- 229910052722 tritium Inorganic materials 0.000 description 1
- 235000015112 vegetable and seed oil Nutrition 0.000 description 1
- 239000008158 vegetable oil Substances 0.000 description 1
- LQUPKVMEAATBSL-UHFFFAOYSA-L zinc;2,3,4-trichlorophenolate Chemical compound [Zn+2].[O-]C1=CC=C(Cl)C(Cl)=C1Cl.[O-]C1=CC=C(Cl)C(Cl)=C1Cl LQUPKVMEAATBSL-UHFFFAOYSA-L 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D519/00—Heterocyclic compounds containing more than one system of two or more relevant hetero rings condensed among themselves or condensed with a common carbocyclic ring system not provided for in groups C07D453/00 or C07D455/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
一种KRASG12D抑制剂及其在医药上的应用。本发明涉及一种通式(I‑a)所述的化合物或者其立体异构体、氘代物、N‑氧化物、溶剂化物、前药、代谢产物、药学上可接受的盐或共晶,及其中间体和制备方法,以及在制备治疗与KRASG12D活性或表达量相关疾病的药物中的应用。 A KRASG12D inhibitor and its application in medicine. The present invention relates to a compound described in general formula (I-a) or its stereoisomer, deuterium, N-oxide, solvate, prodrug, metabolite, pharmaceutically acceptable salt or co-crystal, Its intermediate and preparation method, as well as its application in the preparation of medicines for treating diseases related to KRASG12D activity or expression.
Description
技术领域technical field
本发明涉及一种通式(I)所述的化合物或者其立体异构体、氘代物、溶剂化物、前药、代谢产物、药学上可接受的盐或共晶,及其中间体和制备方法,以及在制备治疗与KRASG12D活性或表达量相关疾病的药物中的应用。The present invention relates to a compound described in general formula (I) or its stereoisomer, deuterium, solvate, prodrug, metabolite, pharmaceutically acceptable salt or co-crystal, and its intermediate and preparation method , and the application in the preparation of medicines for treating diseases related to KRASG12D activity or expression.
背景技术Background technique
RAS蛋白由RAS基因(Rat Sarcoma viral oncogene)表达,是细胞内鸟嘌呤核苷酸结合蛋白,属于GTP酶(水解活性较弱)。RAS蛋白存在于两种不同的状态:非活性的GDP结合状态和活性的GTP结合状态。激活态的RAS蛋白通过与不同下游效应器相互作用进行信号传导,对细胞的生长、分化、细胞骨架、蛋白质运输和分泌等都具有影响。RAS信号传导的激活受鸟嘌呤核苷酸交换因子(GEF,可导致GDP-GTP交换)或者GTP酶活化蛋白(GAP,可导致RAS蛋白由活化态转变为失活态)的调节,突变型RAS蛋白可致对GAP的抵抗导致RAS 蛋白处于持续激活状态,引起细胞不受控制的生长,最终发展为癌变组织(Molecular Cancer,2018,17:33)。RAS protein is expressed by RAS gene (Rat Sarcoma viral oncogene), which is an intracellular guanine nucleotide binding protein and belongs to GTPase (weak hydrolysis activity). RAS proteins exist in two distinct states: an inactive GDP-bound state and an active GTP-bound state. The activated RAS protein conducts signal transduction by interacting with different downstream effectors, and has effects on cell growth, differentiation, cytoskeleton, protein transport and secretion. Activation of RAS signaling is regulated by guanine nucleotide exchange factor (GEF, which causes GDP-GTP exchange) or GTPase activating protein (GAP, which causes the RAS protein to switch from an active to an inactive state), and mutant RAS The protein can cause resistance to GAP and lead to the continuous activation of RAS protein, causing uncontrolled cell growth and eventually developing into cancerous tissue (Molecular Cancer, 2018, 17:33).
RAS基因突变是癌症患者中常见的基因突变类型(Nat.Rev.Drug Discov.2014,13, 828-851),例如RAS基因突变在胰腺癌、结直肠癌、多发性骨髓瘤及NSCLC中分别占到了97.7%,52.2%,42.6%及32.2%。KARS基因(Kristen Rat Sarcoma viral oncogene)突变是RAS 突变中影响最大的突变,占到所有RAS突变的86%。KRAS基因被激活最常见的方式是点突变,95%的KRAS突变主要发生在2号外显子的第12号密码子和13号密码子上,常见的突变形式有KRAS G12C突变(39%),KRAS G12V(18-21%)和KRAS G12D(17-18%)突变。RAS gene mutation is a common type of gene mutation in cancer patients (Nat.Rev.Drug Discov.2014, 13, 828-851). To 97.7%, 52.2%, 42.6% and 32.2%. KARS gene (Kristen Rat Sarcoma viral oncogene) mutation is the most influential mutation among RAS mutations, accounting for 86% of all RAS mutations. The most common way for KRAS gene to be activated is point mutation. 95% of KRAS mutations mainly occur in codons 12 and 13 of exon 2. Common mutations include KRAS G12C mutation (39%), KRAS G12V (18-21%) and KRAS G12D (17-18%) mutations.
自发现癌症中的KRAS突变蛋白并且观察到抑制这些突变蛋白能抑制肿瘤增殖以来, KRAS突变蛋白抑制剂就受到了广泛关注。KRAS长期以来被认为是一个“不可成药靶点”:RAS 对GTP/GDP具有很高的亲和力(皮摩尔级别),整个蛋白也缺少其他的“配体结合口袋”(Clin. Cancer Res.2015,21,1810–1818)。KRAS mutant protein inhibitors have received a lot of attention since the discovery of KRAS mutant proteins in cancer and the observation that inhibiting these mutant proteins can inhibit tumor proliferation. KRAS has long been considered a "non-druggable target": RAS has a high affinity (picomolar level) for GTP/GDP, and the entire protein lacks other "ligand binding pockets" (Clin. Cancer Res.2015, 21, 1810–1818).
KRAS G12D突变影响欧美国家约18万患者。KRAS G12D在胰腺癌患者中占36%,在结肠癌患者中占12%,在NSCLC腺癌患者中占4%,在子宫内膜癌患者中占6%。目前,针对KRAS G12D突变的抑制剂仍处于早期阶段,未有化合物进入临床研究,仅少数公司有该领域的相关专利公开,例如Mirati Therapeutics公司申请了一篇关于KRAS G12D的抑制剂专利(WO2021041671)。因此,有必要开发能够抑制KRAS G12D蛋白的化合物,用于治疗因KRASG12D突变引起的相关疾病。The KRAS G12D mutation affects about 180,000 patients in Europe and the United States. KRAS G12D was found in 36% of pancreatic cancer patients, 12% of colon cancer patients, 4% of NSCLC adenocarcinoma patients, and 6% of endometrial cancer patients. At present, inhibitors against KRAS G12D mutation are still in the early stage, no compound has entered clinical research, and only a few companies have published related patents in this field. For example, Mirati Therapeutics has applied for a KRAS G12D inhibitor patent (WO2021041671) . Therefore, it is necessary to develop compounds capable of inhibiting KRAS G12D protein for the treatment of related diseases caused by KRASG12D mutations.
发明内容Contents of the invention
本发明的目的是提供能够抑制KRAS G12D蛋白的化合物或者其立体异构体、氘代物、溶剂化物、前药、代谢产物、药学上可接受的盐或共晶,及其中间体和制备方法,以及在制备治疗与KRAS G12D活性或表达量相关疾病的药物中的应用。The object of the present invention is to provide a compound capable of inhibiting KRAS G12D protein or its stereoisomer, deuterium, solvate, prodrug, metabolite, pharmaceutically acceptable salt or co-crystal, and its intermediate and preparation method, And the application in preparation of medicines for treating diseases related to KRAS G12D activity or expression.
本发明提供一种通式(I-a)所述的化合物或者其立体异构体、氘代物、N-氧化物、溶剂化物、前药、代谢产物、药学上可接受的盐或共晶,其中The present invention provides a compound described in general formula (I-a) or its stereoisomer, deuterated substance, N-oxide, solvate, prodrug, metabolite, pharmaceutically acceptable salt or co-crystal, wherein
在一些实施方案中,X1或X2各自独立地选自N或CRb; In some embodiments, X or X are each independently selected from N or CR b ;
在一些实施方案中,X1、X2选自N;In some embodiments, X 1 , X 2 are selected from N;
在一些实施方案中,X1、X2选自CRb;In some embodiments, X 1 , X 2 are selected from CR b ;
在一些实施方案中,X1选自N,X2选自CRb;In some embodiments, X 1 is selected from N, and X 2 is selected from CR b ;
在一些实施方案中,X1选自CRb,X2选自N;In some embodiments, X 1 is selected from CR b and X 2 is selected from N;
在一些实施方案中,环A选自3至12元碳环基或4至12元杂环基,所述的碳环基或杂环基任选进一步被0至6个Ra取代,所述的杂环基含有1至4个(例如1、2、3或4个)选自O、S、N的杂原子;In some embodiments, Ring A is selected from a 3- to 12-membered carbocyclyl or a 4- to 12-membered heterocyclyl, and said carbocyclyl or heterocyclyl is optionally further substituted by 0 to 6 R a , said The heterocyclyl group contains 1 to 4 (eg 1, 2, 3 or 4) heteroatoms selected from O, S, N;
在一些实施方案中,L1、L2或L3各自独立地选自键、O、-CH2-、-OCH2-、-CH2O-、-C(=O)-、 S或NRx;In some embodiments, each of L 1 , L 2 or L 3 is independently selected from a bond, O, -CH 2 -, -OCH 2 -, -CH 2 O-, -C(=O)-, S, or NR x ;
在一些实施方案中,Rx选自H、C1-6烷基或C3-6环烷基,所述的烷基或环烷基任选进一步被0至4个(例如0、1、2、3或4个)选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、 C1-4烷基或C1-4烷氧基的取代基所取代;In some embodiments, R x is selected from H, C 1-6 alkyl or C 3-6 cycloalkyl, and said alkyl or cycloalkyl is optionally further replaced by 0 to 4 (for example 0, 1, 2, 3 or 4) are substituted by substituents selected from H, halogen, OH, cyano, oxo, CF 3 , COOH, NH 2 , C 1-4 alkyl or C 1-4 alkoxy;
在一些实施方案中,L3选自键;In some embodiments, L is selected from a bond;
在一些实施方案中,选自 In some embodiments, selected from
在一些实施方案中,选自4至12元含氮杂环,所述的含氮杂环选自饱和或部分饱和的如下基团之一:单环、并环、桥环或螺环,所述的含氮杂环、单环、并环、桥环或螺环任选进一步被0至4个(例如0、1、2、3或4个)Ra取代,环A1中N-H不被取代;In some embodiments, Selected from 4 to 12 membered nitrogen-containing heterocyclic rings, the nitrogen-containing heterocyclic rings are selected from one of the following saturated or partially saturated groups: monocyclic, parallel rings, bridged rings or spiro rings, and the nitrogen-containing heterocyclic rings , monocyclic ring, parallel ring, bridging ring or spiro ring are optionally further substituted by 0 to 4 (for example 0, 1, 2, 3 or 4) R a , and NH in ring A is not substituted;
在一些实施方案中,选自未被取代的或者取代的如下基团之一: 当被取代时,任选进一步被0至4个(例如0、1、2、 3或4个)Ra取代基所取代,环A1中N-H不被取代;In some embodiments, One of the following unsubstituted or substituted groups: When substituted, it is optionally further substituted by 0 to 4 (eg 0, 1 , 2, 3 or 4) substituents R, and NH in ring A is not substituted;
在一些实施方案中,选自未被取代的或者取代的当被取代时,任选进一步被0至4个(例如0、1、2、3或4个)Ra取代基所取代,环A1中N-H 不被取代;In some embodiments, selected from unsubstituted or substituted When substituted, it is optionally further substituted by 0 to 4 (eg 0, 1, 2, 3 or 4) R substituents, and NH in ring A 1 is not substituted;
在一些实施方案中,选自 In some embodiments, selected from
在一些实施方案中,Ra各自独立的选自H、oxo、卤素、氰基、OH、NH2、-NH(C1-6烷基)、-N(C1-6烷基)2、C1-6烷基、C1-6烷氧基、3至8元环烷基、4至8元杂环基、-O-3至8元环烷基、-O-4至8元杂环烷基,所述的烷基、烷氧基、环烷基、杂环基任选进一步被0至4个(例如0、 1、2、3或4个)选自H、卤素、OH、氰基、CF3、COOH、NH2、C1-4烷基或C1-4烷氧基的取代基所取代,所述的杂环基含有1至4个选自O、S、N的杂原子;In some embodiments, each R a is independently selected from H, oxo, halogen, cyano, OH, NH 2 , -NH(C 1-6 alkyl), -N(C 1-6 alkyl) 2 , C 1-6 alkyl, C 1-6 alkoxy, 3 to 8 membered cycloalkyl, 4 to 8 membered heterocyclyl, -O-3 to 8 membered cycloalkyl, -O-4 to 8 membered heterocyclic Cycloalkyl, the alkyl, alkoxy, cycloalkyl, and heterocyclic groups are optionally further selected from 0 to 4 (for example, 0, 1, 2, 3 or 4) selected from H, halogen, OH, Substituted by cyano, CF 3 , COOH, NH 2 , C 1-4 alkyl or C 1-4 alkoxy, the heterocyclic group contains 1 to 4 selected from O, S, N heteroatom;
在一些实施方案中,Ra各自独立的选自H、oxo、卤素、氰基、C1-4烷基、C1-4烷氧基、3至6元环烷基、4至6元杂环基、-O-3至6元环烷基、-O-4至6元杂环烷基,所述的烷基、烷氧基、环烷基、杂环基任选进一步被0至4个(例如0、1、2、3或4个)选自H、卤素、OH、氰基、CF3、COOH、NH2、C1-4烷基或C1-4烷氧基的取代基所取代,所述的杂环基含有1至4 个选自O、S、N的杂原子;In some embodiments, each R a is independently selected from H, oxo, halogen, cyano, C 1-4 alkyl, C 1-4 alkoxy, 3 to 6 membered cycloalkyl, 4 to 6 membered hetero Cyclic group, -O-3 to 6-membered cycloalkyl group, -O-4 to 6-membered heterocycloalkyl group, the alkyl group, alkoxy group, cycloalkyl group and heterocyclyl group are optionally further replaced by 0 to 4 substituents (for example 0, 1, 2, 3 or 4) selected from H, halogen, OH, cyano, CF 3 , COOH, NH 2 , C 1-4 alkyl or C 1-4 alkoxy Substituted, the heterocyclic group contains 1 to 4 heteroatoms selected from O, S, N;
在一些实施方案中,Ra各自独立的选自H、oxo、F、Cl、Br、I、氰基、OH、NH2、甲基、乙基、丙基、异丙基、甲氧基、乙氧基、丙氧基、异丙氧基、环丙基、环丁基、环戊基、环己基、-O-环丙基、-O-环丁基、-O-环戊基、-O-环己基,所述的甲基、乙基、丙基、甲氧基、乙氧基、丙氧基、异丙氧基任选进一步被0至4个(例如0、1、2、3或4个)选自H、F、Cl、 Br、I、OH、氰基、CF3、COOH、NH2、甲基、乙基、丙基、甲氧基、乙氧基、丙氧基或异丙氧基的取代基所取代;In some embodiments, each R a is independently selected from H, oxo, F, Cl, Br, I, cyano, OH, NH 2 , methyl, ethyl, propyl, isopropyl, methoxy, Ethoxy, propoxy, isopropoxy, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, -O-cyclopropyl, -O-cyclobutyl, -O-cyclopentyl, - O-cyclohexyl, the methyl, ethyl, propyl, methoxy, ethoxy, propoxy, isopropoxy are optionally further replaced by 0 to 4 (eg 0, 1, 2, 3 or 4) selected from H, F, Cl, Br, I, OH, cyano, CF 3 , COOH, NH 2 , methyl, ethyl, propyl, methoxy, ethoxy, propoxy or Substituents of isopropoxy are substituted;
在一些实施方案中,Ra各自独立的选自H、oxo、F、Cl、Br、I、氰基、OH、NH2、甲基、乙基、甲氧基、乙氧基、丙氧基、异丙氧基、环丙基、-O-环丙基,所述的甲基、乙基、丙基、甲氧基、乙氧基、丙氧基、丙氧基、异丙氧基、环丙基任选进一步被0至4个(例如0、1、2、 3或4个)选自H、F、Cl、Br、I、OH、氰基、CF3、COOH、NH2、甲基、乙基、丙基、甲氧基、乙氧基或丙氧基所取代;In some embodiments, each R a is independently selected from H, oxo, F, Cl, Br, I, cyano, OH, NH 2 , methyl, ethyl, methoxy, ethoxy, propoxy , isopropoxy, cyclopropyl, -O-cyclopropyl, the methyl, ethyl, propyl, methoxy, ethoxy, propoxy, propoxy, isopropoxy, Cyclopropyl is optionally further 0 to 4 (eg 0, 1, 2, 3 or 4) selected from H, F, Cl, Br, I, OH, cyano, CF 3 , COOH, NH 2 , formazan Substituted by radical, ethyl, propyl, methoxy, ethoxy or propoxy;
在一些实施方案中,Ra各自独立的选自H;In some embodiments, each R a is independently selected from H;
在一些实施方案中,两个Ra与其相连接的原子共同形成3至12元碳环基或4至12元杂环基,所述的碳环基或杂环基任选进一步被0至4个(例如0、1、2、3或4个)选自H、卤素、OH、氰基、CF3、COOH、NH2、-NH(C1-6烷基)、-N(C1-6烷基)2、C1-4烷基或C1-4烷氧基的取代基所取代,所述的杂环基含有1至4个(例如1、2、3或4个)选自O、S、N的杂原子;In some embodiments, two R a together form a 3 to 12 membered carbocyclyl or a 4 to 12 membered heterocyclyl, and the carbocyclyl or heterocyclyl is optionally further replaced by 0 to 4 (eg 0, 1, 2, 3 or 4) selected from H, halogen, OH, cyano, CF 3 , COOH, NH 2 , -NH(C 1-6 alkyl), -N(C 1- 6 alkyl) 2 , C 1-4 alkyl or C 1-4 alkoxy substituents, the heterocyclic group contains 1 to 4 (such as 1, 2, 3 or 4) selected from Heteroatoms of O, S, N;
在一些实施方案中,两个Ra与其相连接的原子共同形成3至6元碳环基或4至6元杂环基,所述的碳环基或杂环基任选进一步被0至4个选自H、卤素、OH、氰基、CF3、COOH、 NH2、C1-4烷基或C1-4烷氧基的取代基所取代,所述的杂环基含有1至4个(例如1、2、3或 4个)选自O、S、N的杂原子;In some embodiments, two R a together form a 3-6 membered carbocyclyl or a 4-6 membered heterocyclyl, and the carbocyclyl or heterocyclyl is optionally further replaced by 0 to 4 Substituents selected from H, halogen, OH, cyano, CF 3 , COOH, NH 2 , C 1-4 alkyl or C 1-4 alkoxy, the heterocyclic group contains 1 to 4 (eg 1, 2, 3 or 4) heteroatoms selected from O, S, N;
在一些实施方案中,环B1选自3至8元碳环基、5至7元杂环基、5至6元杂芳基或苯基,所述碳环基、杂环基、杂芳基或苯基任选进一步被0至7个(例如0、1、2、3、4、5、6、7 个)Rb取代,所述的杂环基或杂芳基含有1至4个选自O、S、N的杂原子;In some embodiments, Ring B is selected from 3 to 8 membered carbocyclyl, 5 to 7 membered heterocyclyl, 5 to 6 membered heteroaryl or phenyl, said carbocyclyl, heterocyclyl, heteroaryl The base or phenyl group is optionally further substituted by 0 to 7 (eg 0, 1, 2, 3, 4, 5, 6, 7) R b , and the heterocyclic or heteroaryl group contains 1 to 4 Heteroatoms selected from O, S, N;
在一些实施方案中,选自 In some embodiments, selected from
其左边与L2连接,上方与环A1连接; Its left side is connected with L 2 , and its upper part is connected with ring A 1 ;
在一些实施方案中,选自 其左边与L2连接,上方与环A1连接;In some embodiments, selected from Its left side is connected with L 2 , and its upper part is connected with ring A 1 ;
在一些实施方案中,选自其左边与L2连接,上方与环A1连接;In some embodiments, selected from Its left side is connected with L 2 , and its upper part is connected with ring A 1 ;
在一些实施方案中,选自其左边与 L2连接,上方与环A1连接;In some embodiments, selected from Its left side is connected with L 2 , and its upper part is connected with ring A 1 ;
在一些实施方案中,选自其左边与L2连接,上方与环 A1连接;In some embodiments, selected from Its left side is connected with L 2 , and its upper part is connected with ring A 1 ;
在一些实施方案中,选自其左边与L2连接,上方与环 A1连接;In some embodiments, selected from Its left side is connected with L 2 , and its upper part is connected with ring A 1 ;
在一些实施方案中,Rb各自独立的选自H、oxo、卤素、氰基、OH、NH2、-NH(C1-6烷基)、-N(C1-6烷基)2、C1-6烷基、C1-6烷硫基、C2-6烯基、C2-6炔基、C1-6烷氧基、3至8元环烷基、4 至8元杂环基、-O-3至8元环烷基、-O-4至8元杂环烷基,所述的烷基、烯基、炔基、烷氧基、烷硫基、环烷基、杂环基或杂环烷基任选进一步被0至4个(例如0、1、2、3或4个) 选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、C1-4烷基、C1-4烷氧基、4-7元杂环基或C3-6环烷基的取代基所取代,所述的杂环基或杂环烷基含有1至4个选自O、S、N的杂原子;In some embodiments, each R b is independently selected from H, oxo, halogen, cyano, OH, NH 2 , -NH(C 1-6 alkyl), -N(C 1-6 alkyl) 2 , C 1-6 alkyl, C 1-6 alkylthio, C 2-6 alkenyl, C 2-6 alkynyl, C 1-6 alkoxy, 3 to 8 membered cycloalkyl, 4 to 8 membered hetero Cyclic group, -O-3 to 8-membered cycloalkyl group, -O-4 to 8-membered heterocycloalkyl group, said alkyl group, alkenyl group, alkynyl group, alkoxy group, alkylthio group, cycloalkyl group, Heterocyclyl or heterocycloalkyl is optionally further selected from 0 to 4 (eg 0, 1, 2, 3 or 4) selected from H, halogen, OH, cyano, oxo, CF 3 , COOH, NH 2 , Substituents of C 1-4 alkyl, C 1-4 alkoxy, 4-7 membered heterocyclyl or C 3-6 cycloalkyl, the heterocyclyl or heterocycloalkyl containing 1 to 4 heteroatoms selected from O, S, N;
在一些实施方案中,两个Rb与其相连接的原子共同形成3至12元碳环基或4至12元杂环基,所述的碳环基或杂环基任选进一步被0至4个(例如0、1、2、3或4个)选自H、卤素、OH、氰基、CF3、COOH、NH2、-NH(C1-6烷基)、-N(C1-6烷基)2、C1-4烷基或C1-4烷氧基的取代基所取代,所述的杂环基含有1至4个(例如1、2、3或4个)选自O、S、N的杂原子;In some embodiments, two R b together form a 3- to 12-membered carbocyclyl or a 4- to 12-membered heterocyclyl, and the carbocyclyl or heterocyclyl is optionally further replaced by 0 to 4 (eg 0, 1, 2, 3 or 4) selected from H, halogen, OH, cyano, CF 3 , COOH, NH 2 , -NH(C 1-6 alkyl), -N(C 1- 6 alkyl) 2 , C 1-4 alkyl or C 1-4 alkoxy substituents, the heterocyclic group contains 1 to 4 (such as 1, 2, 3 or 4) selected from Heteroatoms of O, S, N;
在一些实施方案中,Rb各自独立的选自H、oxo、卤素、氰基、C1-4烷基、C1-4烷硫基、C2-4烯基、C2-4炔基、C1-4烷氧基、3至6元环烷基、4至6元杂环基、-O-3至6元环烷基、-O-4 至6元杂环烷基,所述的烷基、烯基、炔基、烷氧基、烷硫基、环烷基、杂环基或杂环烷基任选进一步被0至4个(例如0、1、2、3或4个)选自H、卤素、OH、氰基、oxo、CF3、COOH、 NH2、C1-4烷基或C1-4烷氧基的取代基所取代,所述的杂环基或杂环烷基含有1至4个选自O、 S、N的杂原子;In some embodiments, each R b is independently selected from H, oxo, halogen, cyano, C 1-4 alkyl, C 1-4 alkylthio, C 2-4 alkenyl, C 2-4 alkynyl , C 1-4 alkoxy, 3 to 6 membered cycloalkyl, 4 to 6 membered heterocyclyl, -O-3 to 6 membered cycloalkyl, -O-4 to 6 membered heterocyclyl, said The alkyl, alkenyl, alkynyl, alkoxy, alkylthio, cycloalkyl, heterocyclyl or heterocycloalkyl groups are optionally further replaced by 0 to 4 (for example 0, 1, 2, 3 or 4 ) is substituted by a substituent selected from H, halogen, OH, cyano, oxo, CF 3 , COOH, NH 2 , C 1-4 alkyl or C 1-4 alkoxy, and the heterocyclic or hetero Cycloalkyl contains 1 to 4 heteroatoms selected from O, S, N;
在一些实施方案中,两个Rb与其相连接的原子共同形成3至6元碳环基或4至6元杂环基,所述的碳环基或杂环基任选进一步被0至4个(例如0、1、2、3或4个)选自H、卤素、 OH、氰基、CF3、COOH、NH2、C1-4烷基或C1-4烷氧基的取代基所取代,所述的杂环基含有1 至4个(例如1、2、3或4个)选自O、S、N的杂原子;In some embodiments, two R b together form a 3-6 membered carbocyclyl or a 4-6 membered heterocyclyl, and the carbocyclyl or heterocyclyl is optionally further replaced by 0 to 4 one (eg 0, 1, 2, 3 or 4) substituents selected from H, halogen, OH, cyano, CF 3 , COOH, NH 2 , C 1-4 alkyl or C 1-4 alkoxy Substituted, the heterocyclic group contains 1 to 4 (eg 1, 2, 3 or 4) heteroatoms selected from O, S, N;
在一些实施方案中,Rb各自独立的选自H、oxo、F、Cl、Br、I、氰基、OH、NH2、甲基、乙基、丙基、异丙基、乙烯基、丙烯基、乙炔基、丙炔基、甲氧基、乙氧基、丙氧基、异丙氧基、环丙基、环丁基、环戊基、环己基、-O-环丙基、-O-环丁基、-O-环戊基、-O-环己基,所述的甲基、乙基、丙基、乙烯基、丙烯基、乙炔基、丙炔基、甲氧基、乙氧基、丙氧基、异丙氧基任选进一步被0至4个(例如0、1、2、3或4个)选自H、F、Cl、Br、I、OH、氰基、 oxo、CF3、COOH、NH2、甲基、乙基、丙基、甲氧基、乙氧基、丙氧基或异丙氧基的取代基所取代;In some embodiments, each R b is independently selected from H, oxo, F, Cl, Br, I, cyano, OH, NH 2 , methyl, ethyl, propyl, isopropyl, vinyl, propylene radical, ethynyl, propynyl, methoxy, ethoxy, propoxy, isopropoxy, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, -O-cyclopropyl, -O -Cyclobutyl, -O-cyclopentyl, -O-cyclohexyl, the methyl, ethyl, propyl, vinyl, propenyl, ethynyl, propynyl, methoxy, ethoxy , propoxy, isopropoxy are optionally further selected from 0 to 4 (for example 0, 1, 2, 3 or 4) selected from H, F, Cl, Br, I, OH, cyano, oxo, CF 3. Substituents of COOH, NH 2 , methyl, ethyl, propyl, methoxy, ethoxy, propoxy or isopropoxy;
在一些实施方案中,Rb各自独立的选自H、oxo、F、Cl、Br、I、氰基、OH、NH2、甲基、乙基、甲氧基、乙氧基、丙氧基、异丙氧基、环丙基、-O-环丙基、乙烯基、乙炔基,所述的甲基、乙基、丙基、甲氧基、乙氧基、丙氧基、丙氧基、异丙氧基、乙烯基、乙炔基、环丙基任选进一步被0至4个(例如0、1、2、3或4个)选自H、F、Cl、Br、I、OH、氰基、oxo、 CF3、COOH、NH2、甲基、乙基、丙基、甲氧基、乙氧基或丙氧基所取代;In some embodiments, each R b is independently selected from H, oxo, F, Cl, Br, I, cyano, OH, NH 2 , methyl, ethyl, methoxy, ethoxy, propoxy , isopropoxy, cyclopropyl, -O-cyclopropyl, vinyl, ethynyl, the methyl, ethyl, propyl, methoxy, ethoxy, propoxy, propoxy , isopropoxy, vinyl, ethynyl, cyclopropyl are optionally further selected from 0 to 4 (for example 0, 1, 2, 3 or 4) selected from H, F, Cl, Br, I, OH, Cyano, oxo, CF 3 , COOH, NH 2 , methyl, ethyl, propyl, methoxy, ethoxy or propoxy;
在一些实施方案中,Rb选自H、F、Cl、氰基、OH、NH2、甲基、乙基、甲氧基、乙氧基、环丙基、-O-环丙基;In some embodiments, R is selected from H, F, Cl, cyano, OH, NH2 , methyl, ethyl, methoxy, ethoxy, cyclopropyl, -O-cyclopropyl;
在一些实施方案中,L1选自-O-和-O-CH2-;In some embodiments, L 1 is selected from -O- and -O-CH 2 -;
在一些实施方案中,R1选自卤素、OH、氰基、COOH、NH2、C1-6烷基、C2-6烯基、C2-6炔基、C1-6烷氧基、NH2、-(CH2)q-C(=O)C1-6烷基、-(CH2)q-C(=O)-3至12元杂环、 -(CH2)q-C(=O)-C3-10碳环、-(CH2)q-3至12元杂环或-(CH2)q-C3-10碳环,所述的CH2、烷基、烯基、炔基、烷氧基、碳环或杂环任选进一步被0至4个(例如0、1、2、3或4个)R1a取代,所述的杂环含有1至4个(例如1、2、3或4个)选自O、S、N的杂原子;In some embodiments, R 1 is selected from halogen, OH, cyano, COOH, NH 2 , C 1-6 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, C 1-6 alkoxy , NH 2 , -(CH 2 ) q -C(=O)C 1-6 alkyl, -(CH 2 ) q -C(=O)-3 to 12-membered heterocycle, -(CH 2 ) q - C(=O)-C 3-10 carbocycle, -(CH 2 ) q -3 to 12 membered heterocycle or -(CH 2 ) q -C 3-10 carbocycle, the CH 2 , alkyl, Alkenyl, alkynyl, alkoxy, carbocycle or heterocycle are optionally further substituted by 0 to 4 (eg 0, 1, 2, 3 or 4) R 1a , said heterocycle containing 1 to 4 (eg 1, 2, 3 or 4) heteroatoms selected from O, S, N;
在一些实施方案中,-L1-R1选自在一些实施方案中,R1a各自独立地选自H、卤素、oxo、OH、氰基、CF3、COOH、NH2、-NH(C1-6烷基)、-N(C1-6烷基)2、C1-6烷基、 C2-6烯基、C2-6炔基、-C1-6亚烷基-OH、-(CH2)q-C3-10碳环、-(CH2)q-3至12元杂环、-O-C3-10碳环、-O-3至12元杂环、C1-6烷氧基、-N(C1-6烷基)C(=O)-3至12元杂环的取代基所取代,所述的烷基、CH2、烷氧基、烯基、炔基、亚烷基、碳环或杂环任选进一步被0至4个(例如0、1、 2、3或4个)选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、C1-6烷基、C1-6烷氧基、 C3-6环烷基、3至6元杂环烷基的取代基所取代,所述的杂环或杂环烷基含有1至4个(例如1、2、3或4个)选自O、S、N的杂原子;In some embodiments, -L 1 -R 1 is selected from In some embodiments, each R 1a is independently selected from H, halogen, oxo, OH, cyano, CF 3 , COOH, NH 2 , -NH(C 1-6 alkyl), -N(C 1-6 Alkyl) 2 , C 1-6 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, -C 1-6 alkylene-OH, -(CH 2 ) q -C 3-10 carbocycle , -(CH 2 ) q -3 to 12-membered heterocycle, -OC 3-10 carbocycle, -O-3 to 12-membered heterocycle, C 1-6 alkoxy, -N(C 1-6 alkyl )C(=O)-substituted by a substituent of a 3- to 12-membered heterocycle, said alkyl, CH 2 , alkoxy, alkenyl, alkynyl, alkylene, carbocycle or heterocycle are optionally further 0 to 4 (eg 0, 1, 2, 3 or 4) selected from H, halogen, OH, cyano, oxo, CF 3 , COOH, NH 2 , C 1-6 alkyl, C 1-6 Substituents of alkoxy, C 3-6 cycloalkyl, 3 to 6 membered heterocycloalkyl, said heterocycle or heterocycloalkyl contain 1 to 4 (for example 1, 2, 3 or 4 a) a heteroatom selected from O, S, N;
在一些实施方案中,R1a各自独立地选自H、卤素、oxo、OH、氰基、CF3、COOH、NH2、 -NH(C1-4烷基)、-N(C1-4烷基)2、C1-4烷基、C2-4烯基、C2-4炔基、-C1-4亚烷基-OH、-(CH2)q-C3-8碳环、-(CH2)q-3至8元杂环、-O-C3-8碳环、-O-3至8元杂环、C1-4烷氧基、-N(C1-4烷基)C(=O)-3 至8元杂环的取代基所取代,所述的烷基、CH2、烷氧基、烯基、炔基、亚烷基、碳环或杂环任选进一步被0至4个(例如0、1、2、3或4个)选自H、卤素、OH、氰基、oxo、CF3、 COOH、NH2、C1-6烷基、C1-6烷氧基、C3-6环烷基、3至6元杂环烷基的取代基所取代,所述的杂环或杂环烷基含有1至4个(例如1、2、3或4个)选自O、S、N的杂原子;In some embodiments, each R 1a is independently selected from H, halogen, oxo, OH, cyano, CF 3 , COOH, NH 2 , -NH(C 1-4 alkyl), -N(C 1-4 Alkyl) 2 , C 1-4 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, -C 1-4 alkylene-OH, -(CH 2 ) q -C 3-8 carbocycle , -(CH 2 ) q -3 to 8-membered heterocycle, -OC 3-8 carbocycle, -O-3 to 8-membered heterocycle, C 1-4 alkoxy, -N(C 1-4 alkyl )C(=O)-3 to 8-membered heterocyclic substituents, said alkyl, CH 2 , alkoxy, alkenyl, alkynyl, alkylene, carbocycle or heterocycle are optionally further 0 to 4 (eg 0, 1, 2, 3 or 4) selected from H, halogen, OH, cyano, oxo, CF 3 , COOH, NH 2 , C 1-6 alkyl, C 1-6 Substituents of alkoxy, C 3-6 cycloalkyl, 3 to 6 membered heterocycloalkyl, said heterocycle or heterocycloalkyl contain 1 to 4 (for example 1, 2, 3 or 4 a) a heteroatom selected from O, S, N;
在一些实施方案中,R1a各自独立地选自H、F、Cl、Br、I、oxo、OH、氰基、CF3、COOH、NH2、甲基、乙基、丙基、乙烯基、丙烯基、乙炔基、丙炔基、甲氧基、乙氧基、丙氧基、异丙氧基、环丙基、环丁基、环戊基、环己基、-O-环丙基、-O-环丁基、-O-环戊基、-O-环己基,所述的甲基、乙基、丙基、乙烯基、丙烯基、乙炔基、丙炔基、甲氧基、乙氧基、丙氧基、异丙氧基任选进一步被0至4个(例如0、1、2、3或4个)选自H、F、Cl、Br、I、OH、氰基、 oxo、CF3、COOH、NH2、甲基、乙基、丙基、甲氧基、乙氧基、丙氧基或异丙氧基的取代基所取代;In some embodiments, each R 1a is independently selected from H, F, Cl, Br, I, oxo, OH, cyano, CF 3 , COOH, NH 2 , methyl, ethyl, propyl, vinyl, propenyl, ethynyl, propynyl, methoxy, ethoxy, propoxy, isopropoxy, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, -O-cyclopropyl, - O-cyclobutyl, -O-cyclopentyl, -O-cyclohexyl, the methyl, ethyl, propyl, vinyl, propenyl, ethynyl, propynyl, methoxy, ethoxy Base, propoxy, isopropoxy are optionally further selected from 0 to 4 (for example 0, 1, 2, 3 or 4) selected from H, F, Cl, Br, I, OH, cyano, oxo, Substituents of CF 3 , COOH, NH 2 , methyl, ethyl, propyl, methoxy, ethoxy, propoxy or isopropoxy;
在一些实施方案中,在一些实施方案中,R1a选自F;In some embodiments, R 1a is selected from F;
在一些实施方案中,在一些实施方案中,-L1-R1选自 In some embodiments, -L 1 -R 1 is selected from
在一些实施方案中,L2选自键、O或-C(=O)-;In some embodiments, L is selected from a bond, O, or -C (=O)-;
在一些实施方案中,L2选自键或-C(=O)-;In some embodiments, L is selected from a bond or -C (=O)-;
在一些实施方案中,L2选自键; In some embodiments, L is selected from a bond;
在一些实施方案中,R2选自C1-6烷基、C3-10碳环基、3至10元杂环基、C6-10芳基或5至10元杂芳基,所述的烷基、碳环基、杂环基、芳基或杂芳基任选进一步被0至7个(例如0、 1、2、3、4、5、6或7个)R2a取代,所述的杂环基或杂芳基含有1至4个(例如1、2、3或 4个)选自O、S、N的杂原子;In some embodiments, R is selected from C 1-6 alkyl, C 3-10 carbocyclyl, 3 to 10 membered heterocyclyl, C 6-10 aryl or 5 to 10 membered heteroaryl, said The alkyl, carbocyclyl, heterocyclyl, aryl or heteroaryl of is optionally further substituted by 0 to 7 (eg 0, 1, 2, 3, 4, 5, 6 or 7) R 2a , the The heterocyclic group or heteroaryl group contains 1 to 4 (eg 1, 2, 3 or 4) heteroatoms selected from O, S, N;
在一些实施方案中,R2选自C3-10碳环基、3至10元杂环基、C6-10芳基或5至10元杂芳基,所述的碳环基、杂环基、芳基或杂芳基任选进一步被0至5个(例如0、1、2、3、4或5 个)R2a取代,所述的杂环基或杂芳基含有1至4个(例如1、2、3或4个)选自O、S、N的杂原子; In some embodiments, R is selected from C 3-10 carbocyclyl, 3-10 membered heterocyclyl, C 6-10 aryl or 5-10 membered heteroaryl, the carbocyclyl, heterocyclic The radical, aryl or heteroaryl is optionally further substituted by 0 to 5 (eg 0, 1, 2, 3, 4 or 5) R 2a , and the heterocyclyl or heteroaryl contains 1 to 4 (eg 1, 2, 3 or 4) heteroatoms selected from O, S, N;
在一些实施方案中,R2选自取代的或者未被取代的如下基团之一:苯基、萘基、噻吩基、呋喃基、吡咯基、噻唑基、异噻唑基、噁唑基、异噁唑基、咪唑基、吡唑基、吲唑、苯并咪唑基、苯并吡唑基、苯并噻唑基、苯并噻吩基、苯并呋喃基、苯并嘧啶基、苯并吡啶基或吡啶基,当被取代时,任选被0至5个(例如0、1、2、3、4或5个)R2a取代; In some embodiments, R is selected from substituted or unsubstituted one of the following groups: phenyl, naphthyl, thienyl, furyl, pyrrolyl, thiazolyl, isothiazolyl, oxazolyl, iso Oxazolyl, imidazolyl, pyrazolyl, indazole, benzimidazolyl, benzopyrazolyl, benzothiazolyl, benzothienyl, benzofuryl, benzopyrimidinyl, benzopyridyl or Pyridyl, when substituted, is optionally substituted by 0 to 5 (eg 0, 1, 2, 3, 4 or 5) R 2a ;
在一些实施方案中,R2选自取代的或者未被取代的如下基团之一:苯基、吡啶、苯并噻唑基、苯并噻吩基、苯并呋喃基、苯并吡唑基或萘基,当被取代时,任选被0至5个(例如0、 1、2、3、4或5个)R2a取代; In some embodiments, R is selected from one of substituted or unsubstituted phenyl, pyridine, benzothiazolyl, benzothienyl, benzofuryl, benzopyrazolyl, or naphthalene The group, when substituted, is optionally substituted by 0 to 5 (eg 0, 1, 2, 3, 4 or 5) R 2a ;
在一些实施方案中,R2选自取代的如下基团之一:苯基、吡啶、苯并噻唑基、苯并噻吩基、苯并呋喃基、苯并吡唑基或萘基,当被取代时,被1至5个(例如1、2、3、4或5个) R2a取代,R2a各自独立的选自H、F、Cl、OH、氰基、NH2、CF3、OCF3、-SCH3、甲基、乙基、丙基、异丙基、甲氧基、乙氧基、乙炔基、丙炔基、环丙基、环丁基或环戊基,所述甲基、乙基、丙基、异丙基、甲氧基、乙氧基、乙炔基、丙炔基、环丙基、环丁基或环戊基任选进一步被0至4个选自H、F、Cl、OH、甲基、乙基或环丙基的取代基所取代,条件是至少有一个 R2a选自环丙基、环丁基或环戊基; In some embodiments, R is selected from one of the following substituted groups: phenyl, pyridine, benzothiazolyl, benzothienyl, benzofuryl, benzopyrazolyl, or naphthyl, when substituted , substituted by 1 to 5 (eg 1, 2, 3, 4 or 5) R 2a , each R 2a independently selected from H, F, Cl, OH, cyano, NH 2 , CF 3 , OCF 3 , -SCH 3 , methyl, ethyl, propyl, isopropyl, methoxy, ethoxy, ethynyl, propynyl, cyclopropyl, cyclobutyl or cyclopentyl, the methyl, Ethyl, propyl, isopropyl, methoxy, ethoxy, ethynyl, propynyl, cyclopropyl, cyclobutyl or cyclopentyl are optionally further replaced by 0 to 4 selected from H, F, Substituents of Cl, OH, methyl, ethyl or cyclopropyl, provided that at least one R 2a is selected from cyclopropyl, cyclobutyl or cyclopentyl;
在一些实施方案中,R2选自苯基、苯并吡唑基、苯并呋喃基、吡啶基,所述的苯基被1 至4个R2a取代,所述的苯并吡唑基、苯并呋喃基、吡啶基被0至4个R2a取代; In some embodiments, R is selected from phenyl, benzopyrazolyl, benzofuryl, pyridyl, said phenyl is substituted by 1 to 4 R 2a , said benzopyrazolyl, Benzofuryl and pyridyl are substituted by 0 to 4 R 2a ;
在一些实施方案中,R2a各自独立的选自H、卤素、oxo、OH、氰基、CF3、COOH、NH2、 -NH(C1-6烷基)、-N(C1-6烷基)2、C1-6烷基、C1-6烷氧基、C1-6烷硫基、C2-6烯基、C2-6炔基、C3-6环烷基、3至6元杂环烷基、-O-C3-6环烷基、-O-3至6元杂环烷基、C6-10芳基或5至10元杂芳基,所述的烷基、烷氧基、烷硫基、烯基、炔基、环烷基、杂环烷基、芳基、杂芳基任选进一步被0至4个(例如0、1、2、3或4个)选自H、卤素、OH、氰基、oxo、CF3、COOH、 NH2、C1-6烷基、C1-6烷氧基、C3-6环烷基、3至6元杂环烷基的取代基所取代,所述的杂环烷基、杂环基或杂芳基含有1至4个(例如1、2、3或4个)选自O、S、N的杂原子;In some embodiments, each R 2a is independently selected from H, halogen, oxo, OH, cyano, CF 3 , COOH, NH 2 , -NH(C 1-6 alkyl), -N(C 1-6 Alkyl) 2 , C 1-6 alkyl, C 1-6 alkoxy, C 1-6 alkylthio, C 2-6 alkenyl, C 2-6 alkynyl, C 3-6 cycloalkyl, 3 to 6 membered heterocycloalkyl, -OC 3-6 cycloalkyl, -O-3 to 6 membered heterocycloalkyl, C 6-10 aryl or 5 to 10 membered heteroaryl, the alkyl , alkoxy, alkylthio, alkenyl, alkynyl, cycloalkyl, heterocycloalkyl, aryl, heteroaryl are optionally further replaced by 0 to 4 (for example 0, 1, 2, 3 or 4 ) is selected from H, halogen, OH, cyano, oxo, CF 3 , COOH, NH 2 , C 1-6 alkyl, C 1-6 alkoxy, C 3-6 cycloalkyl, 3-6 membered hetero Substituents of cycloalkyl, said heterocycloalkyl, heterocyclyl or heteroaryl contain 1 to 4 (for example 1, 2, 3 or 4) heteroatoms selected from O, S, N ;
在一些实施方案中,R2a各自独立的选自-NHC(O)C1-6烷基、-NHS(O)2C1-6烷基、-P(O)(C1-6烷基)2,所述的烷基任选进一步被0至4个(例如0、1、2、3或4个)选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、C1-6烷基、C1-6烷氧基、C3-6环烷基、3至6元杂环烷基的取代基所取代,所述的杂环烷基含有1至4个(例如1、2、3或4个)选自O、S、N的杂原子;In some embodiments, each R 2a is independently selected from -NHC(O)C 1-6 alkyl, -NHS(O) 2 C 1-6 alkyl, -P(O)(C 1-6 alkyl ) 2 , the alkyl group is optionally further selected from 0 to 4 (eg 0, 1, 2, 3 or 4) selected from H, halogen, OH, cyano, oxo, CF 3 , COOH, NH 2 , Substituents of C 1-6 alkyl, C 1-6 alkoxy, C 3-6 cycloalkyl, and 3 to 6 membered heterocycloalkyl, wherein the heterocycloalkyl contains 1 to 4 ( For example 1, 2, 3 or 4) heteroatoms selected from O, S, N;
在一些实施方案中,两个R2a与其直接相连的原子共同形成3至12元碳环基或4至12元杂环基,所述的碳环基或杂环基任选进一步被0至4个(例如0、1、2、3或4个)选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、C1-4烷基或C1-4烷氧基的取代基所取代,所述的杂环基含有1至4个(例如1、2、3或4个)选自O、S、N的杂原子;In some embodiments, two R 2a together form a 3- to 12-membered carbocyclyl or a 4- to 12-membered heterocyclyl, which is optionally further replaced by 0 to 4 one (eg 0, 1, 2, 3 or 4) selected from H, halogen, OH, cyano, oxo, CF 3 , COOH, NH 2 , C 1-4 alkyl or C 1-4 alkoxy Substituents are substituted, and the heterocyclic group contains 1 to 4 (eg 1, 2, 3 or 4) heteroatoms selected from O, S, N;
在一些实施方案中,R2a各自独立的选自H、卤素、oxo、OH、氰基、CF3、COOH、NH2、 -NH(C1-4烷基)、-N(C1-4烷基)2、C1-4烷基、C1-4烷氧基、C1-4烷硫基、C2-4烯基、C2-6炔基、C3-6环烷基、3至6元杂环烷基、-O-C3-6环烷基、-O-3至6元杂环烷基、C6-10芳基或5至10元杂芳基,所述的烷基、烷氧基、烷硫基、烯基、炔基、环烷基、杂环烷基、芳基、杂芳基任选进一步被0至4个(例如0、1、2、3或4个)选自H、卤素、OH、氰基、oxo、CF3、COOH、 NH2、C1-4烷基、C1-4烷氧基、C3-6环烷基、3至6元杂环烷基的取代基所取代,所述的杂环烷基、杂环基或杂芳基含有1至4个(例如1、2、3或4个)选自O、S、N的杂原子;In some embodiments, each R 2a is independently selected from H, halogen, oxo, OH, cyano, CF 3 , COOH, NH 2 , -NH(C 1-4 alkyl), -N(C 1-4 Alkyl) 2 , C 1-4 alkyl, C 1-4 alkoxy, C 1-4 alkylthio, C 2-4 alkenyl, C 2-6 alkynyl, C 3-6 cycloalkyl, 3 to 6 membered heterocycloalkyl, -OC 3-6 cycloalkyl, -O-3 to 6 membered heterocycloalkyl, C 6-10 aryl or 5 to 10 membered heteroaryl, the alkyl , alkoxy, alkylthio, alkenyl, alkynyl, cycloalkyl, heterocycloalkyl, aryl, heteroaryl are optionally further replaced by 0 to 4 (for example 0, 1, 2, 3 or 4 ) is selected from H, halogen, OH, cyano, oxo, CF 3 , COOH, NH 2 , C 1-4 alkyl, C 1-4 alkoxy, C 3-6 cycloalkyl, 3 to 6 membered hetero Substituents of cycloalkyl, said heterocycloalkyl, heterocyclyl or heteroaryl contain 1 to 4 (for example 1, 2, 3 or 4) heteroatoms selected from O, S, N ;
在一些实施方案中,R2a各自独立的选自-NHC(O)C1-4烷基、-NHS(O)2C1-4烷基、-P(O)(C1-4烷基)2,所述的烷基任选进一步被0至4个(例如0、1、2、3或4个)选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、C1-6烷基、C1-6烷氧基、C3-6环烷基、3至6元杂环烷基的取代基所取代,所述的杂环烷基含有1至4个(例如1、2、3或4个)选自O、S、N的杂原子;In some embodiments, each R 2a is independently selected from -NHC(O)C 1-4 alkyl, -NHS(O) 2 C 1-4 alkyl, -P(O)(C 1-4 alkyl ) 2 , the alkyl group is optionally further selected from 0 to 4 (eg 0, 1, 2, 3 or 4) selected from H, halogen, OH, cyano, oxo, CF 3 , COOH, NH 2 , Substituents of C 1-6 alkyl, C 1-6 alkoxy, C 3-6 cycloalkyl, and 3 to 6 membered heterocycloalkyl, wherein the heterocycloalkyl contains 1 to 4 ( For example 1, 2, 3 or 4) heteroatoms selected from O, S, N;
在一些实施方案中,两个R2a与其相连接的原子共同形成3至6元碳环基或4至6元杂环基,所述的碳环基或杂环基任选进一步被0至4个(例如0、1、2、3或4个)选自H、卤素、 OH、氰基、oxo、CF3、COOH、NH2、C1-4烷基或C1-4烷氧基的取代基所取代,所述的杂环基含有1至4个(例如1、2、3或4个)选自O、S、N的杂原子;In some embodiments, two R 2a together form a 3 to 6 membered carbocyclyl or a 4 to 6 membered heterocyclyl, which is optionally further replaced by 0 to 4 one (eg 0, 1, 2, 3 or 4) selected from H, halogen, OH, cyano, oxo, CF 3 , COOH, NH 2 , C 1-4 alkyl or C 1-4 alkoxy Substituents are substituted, and the heterocyclic group contains 1 to 4 (eg 1, 2, 3 or 4) heteroatoms selected from O, S, N;
在一些实施方案中,R2a各自独立的选自H、F、Cl、Br、I、oxo、OH、NH2、氰基、CF3、 -SCH3、甲基、乙基、丙基、异丙基、乙烯基、丙烯基、乙炔基、丙炔基、甲氧基、乙氧基、丙氧基、异丙氧基、环丙基、环丁基、环戊基、环己基、-O-环丙基、-O-环丁基、-O-环戊基、 -O-环己基,所述的甲基、乙基、丙基、异丙基、乙烯基、丙烯基、乙炔基、丙炔基、甲氧基、乙氧基、丙氧基、异丙氧基、环丙基、环丁基、环戊基、环己基任选进一步被0至4个(例如 0、1、2、3或4个)选自H、F、Cl、Br、I、OH、氰基、oxo、CF3、COOH、NH2、甲基、乙基、丙基、甲氧基、乙氧基、丙氧基或异丙氧基的取代基所取代;In some embodiments, each R 2a is independently selected from H, F, Cl, Br, I, oxo, OH, NH 2 , cyano, CF 3 , -SCH 3 , methyl, ethyl, propyl, iso Propyl, vinyl, propenyl, ethynyl, propynyl, methoxy, ethoxy, propoxy, isopropoxy, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, -O -cyclopropyl, -O-cyclobutyl, -O-cyclopentyl, -O-cyclohexyl, the methyl, ethyl, propyl, isopropyl, vinyl, propenyl, ethynyl, Proynyl, methoxy, ethoxy, propoxy, isopropoxy, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl are optionally further replaced by 0 to 4 (for example 0, 1, 2 , 3 or 4) selected from H, F, Cl, Br, I, OH, cyano, oxo, CF 3 , COOH, NH 2 , methyl, ethyl, propyl, methoxy, ethoxy, Substituents of propoxy or isopropoxy;
在一些实施方案中,R2a各自独立的选自-NHC(O)CH3、-NHS(O)2CH3、-P(O)(CH3)2、环戊烯基、苯基、吡唑基,所述的-CH3、环戊烯基、苯基、吡唑基任选进一步被0至4个(例如0、1、2、3或4个)选自H、F、Cl、Br、I、OH、氰基、oxo、CF3、COOH、NH2、甲基、乙基、丙基、甲氧基、乙氧基、丙氧基或异丙氧基的取代基所取代;In some embodiments, each R 2a is independently selected from -NHC(O)CH 3 , -NHS(O) 2 CH 3 , -P(O)(CH 3 ) 2 , cyclopentenyl, phenyl, pyridine Azolyl, the -CH 3 , cyclopentenyl, phenyl, pyrazolyl are optionally further selected from 0 to 4 (for example 0, 1, 2, 3 or 4) selected from H, F, Cl, Substituents of Br, I, OH, cyano, oxo, CF 3 , COOH, NH 2 , methyl, ethyl, propyl, methoxy, ethoxy, propoxy or isopropoxy;
在一些实施方案中,两个R2a与其相连接的原子共同形成3至6元碳环基或4至6元杂环基,所述的碳环基或杂环基任选进一步被0至4个(例如0、1、2、3或4个)选自H、F、 Cl、Br、I、OH、氰基、oxo、CF3、COOH、NH2、甲基、乙基、丙基、甲氧基、乙氧基、丙氧基或异丙氧基的取代基所取代,所述的杂环基含有1至4个选自O、S、N的杂原子;In some embodiments, two R 2a together form a 3 to 6 membered carbocyclyl or a 4 to 6 membered heterocyclyl, which is optionally further replaced by 0 to 4 (for example 0, 1, 2, 3 or 4) selected from H, F, Cl, Br, I, OH, cyano, oxo, CF 3 , COOH, NH 2 , methyl, ethyl, propyl, Substituted by a substituent of methoxy, ethoxy, propoxy or isopropoxy, the heterocyclic group contains 1 to 4 heteroatoms selected from O, S, N;
在一些实施方案中,R2a各自独立的选自H、F、Cl、OH、氰基、NH2、CF3、OCF3、-SCH3、甲基、乙基、丙基、异丙基、甲氧基、乙氧基、乙炔基、丙炔基、环丙基、环丁基或环戊基,所述甲基、乙基、丙基、异丙基、甲氧基、乙氧基、乙炔基、丙炔基、环丙基、环丁基或环戊基任选进一步被0至4个选自H、F、Cl、OH、甲基、乙基或环丙基的取代基所取代;In some embodiments, each R 2a is independently selected from H, F, Cl, OH, cyano, NH 2 , CF 3 , OCF 3 , —SCH 3 , methyl, ethyl, propyl, isopropyl, Methoxy, ethoxy, ethynyl, propynyl, cyclopropyl, cyclobutyl or cyclopentyl, the methyl, ethyl, propyl, isopropyl, methoxy, ethoxy, Ethynyl, propynyl, cyclopropyl, cyclobutyl or cyclopentyl is optionally further substituted with 0 to 4 substituents selected from H, F, Cl, OH, methyl, ethyl or cyclopropyl ;
在一些实施方案中,R2a各自独立的选自丙烯基、-NHC(O)CH3、-NHS(O)2CH3、-P(O)(CH3)2、环戊烯基、苯基、吡唑基,所述-CH3、丙烯基、环戊烯基、苯基、吡唑基任选进一步被0至4 个选自H、F、Cl、OH、甲基、乙基或环丙基的取代基所取代;In some embodiments, each R 2a is independently selected from propenyl, -NHC(O)CH 3 , -NHS(O) 2 CH 3 , -P(O)(CH 3 ) 2 , cyclopentenyl, benzene Base, pyrazolyl, the -CH 3 , propenyl, cyclopentenyl, phenyl, pyrazolyl are optionally further selected from 0 to 4 selected from H, F, Cl, OH, methyl, ethyl or Cyclopropyl substituents are substituted;
在一些实施方案中,R2a各自独立的选自F、Cl、OH、氰基、NH2、CF3、OCF3、-SCH3、甲基、乙基、丙基、异丙基、甲氧基、乙氧基、乙炔基、丙炔基、环丙基、环丁基、环戊基、丙烯基、-NHC(O)CH3、-NHS(O)2CH3、-P(O)(CH3)2、环戊烯基、苯基、吡唑基,所述甲基、乙基、丙基、异丙基、甲氧基、乙氧基、乙炔基、丙炔基、环丙基、环丁基、环戊基、丙烯基、 -NHC(O)CH3、-NHS(O)2CH3、-P(O)(CH3)2、环戊烯基、苯基、吡唑基任选进一步被0至4个选自H、F、Cl、OH、甲基、乙基或环丙基的取代基所取代;In some embodiments, each R 2a is independently selected from F, Cl, OH, cyano, NH 2 , CF 3 , OCF 3 , -SCH 3 , methyl, ethyl, propyl, isopropyl, methoxy ethoxy, ethynyl, propynyl, cyclopropyl, cyclobutyl, cyclopentyl, propenyl, -NHC(O)CH 3 , -NHS(O) 2 CH 3 , -P(O) (CH 3 ) 2 , cyclopentenyl, phenyl, pyrazolyl, the methyl, ethyl, propyl, isopropyl, methoxy, ethoxy, ethynyl, propynyl, cyclopropanyl Cyclobutyl, cyclopentyl, propenyl, -NHC(O)CH 3 , -NHS(O) 2 CH 3 , -P(O)(CH 3 ) 2 , cyclopentenyl, phenyl, pyridine Azolyl is optionally further substituted by 0 to 4 substituents selected from H, F, Cl, OH, methyl, ethyl or cyclopropyl;
在一些实施方案中,q各自独立的选自0、1、2、3或4;In some embodiments, each q is independently selected from 0, 1, 2, 3 or 4;
在一些实施方案中,n选自0、1、2、3或4;In some embodiments, n is selected from 0, 1, 2, 3 or 4;
在一些实施方案中,m各自独立地选自0、1、2或3;In some embodiments, each m is independently selected from 0, 1, 2, or 3;
任选地,条件是不为m1选自0、1或2;Optionally, the condition is not for m1 is selected from 0, 1 or 2;
任选地,条件是化合物不为或 Optionally, with the proviso that the compound is not or
作为本发明的第一种实施方案,前述通式(I-a)所示的化合物或者其立体异构体、氘代物、 N-氧化物、溶剂化物、前药、代谢产物、药学上可接受的盐或共晶,As the first embodiment of the present invention, the compound represented by the aforementioned general formula (I-a) or its stereoisomer, deuterated substance, N-oxide, solvate, prodrug, metabolite, pharmaceutically acceptable salt or eutectic,
X1或X2各自独立地选自N或CRb;X 1 or X 2 are each independently selected from N or CR b ;
环A选自3至12元碳环基或4至12元杂环基,所述的碳环基或杂环基任选进一步被0至6个Ra取代,所述的杂环基含有1至4个选自O、S、N的杂原子;Ring A is selected from 3 to 12 membered carbocyclic groups or 4 to 12 membered heterocyclic groups, said carbocyclic group or heterocyclic group is optionally further substituted by 0 to 6 R a , and said heterocyclic group contains 1 to 4 heteroatoms selected from O, S, N;
环B1选自3至8元碳环基、5至7元杂环基、5至6元杂芳基或苯基,所述碳环基、杂环基、杂芳基或苯基任选进一步被0至7个Rb取代,所述的杂环基或杂芳基含有1至4个选自 O、S、N的杂原子;Ring B is selected from 3 to 8 membered carbocyclyl, 5 to 7 membered heterocyclyl, 5 to 6 membered heteroaryl or phenyl, said carbocyclyl, heterocyclyl, heteroaryl or phenyl optionally Further substituted by 0 to 7 R b , said heterocyclyl or heteroaryl contains 1 to 4 heteroatoms selected from O, S, N;
Ra各自独立的选自H、oxo、卤素、氰基、OH、NH2、-NH(C1-6烷基)、-N(C1-6烷基)2、C1-6烷基、C1-6烷氧基、3至8元环烷基、4至8元杂环基、-O-3至8元环烷基、-O-4至8元杂环烷基,所述的烷基、烷氧基、环烷基、杂环基任选进一步被0至4个选自H、卤素、OH、氰基、CF3、COOH、NH2、C1-4烷基或C1-4烷氧基的取代基所取代,所述的杂环基含有1至4个选自O、S、N的杂原子;R a is each independently selected from H, oxo, halogen, cyano, OH, NH 2 , -NH(C 1-6 alkyl), -N(C 1-6 alkyl) 2 , C 1-6 alkyl , C 1-6 alkoxy, 3 to 8 membered cycloalkyl, 4 to 8 membered heterocyclyl, -O-3 to 8 membered cycloalkyl, -O-4 to 8 membered heterocycloalkyl, said The alkyl, alkoxy, cycloalkyl, and heterocyclic groups are optionally further replaced by 0 to 4 selected from H, halogen, OH, cyano, CF 3 , COOH, NH 2 , C 1-4 alkyl or C Substituted by 1-4 alkoxy substituents, the heterocyclic group contains 1 to 4 heteroatoms selected from O, S, N;
Rb各自独立的选自H、oxo、卤素、氰基、OH、NH2、-NH(C1-6烷基)、-N(C1-6烷基)2、C1-6烷基、C1-6烷硫基、C2-6烯基、C2-6炔基、C1-6烷氧基、3至8元环烷基、4至8元杂环基、-O-3 至8元环烷基、-O-4至8元杂环烷基,所述的烷基、烯基、炔基、烷氧基、烷硫基、环烷基、杂环基或杂环烷基任选进一步被0至4个选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、 C1-4烷基、C1-4烷氧基、4-7元杂环基或C3-6环烷基的取代基所取代,所述的杂环基或杂环烷基含有1至4个选自O、S、N的杂原子;R b are each independently selected from H, oxo, halogen, cyano, OH, NH 2 , -NH(C 1-6 alkyl), -N(C 1-6 alkyl) 2 , C 1-6 alkyl , C 1-6 alkylthio, C 2-6 alkenyl, C 2-6 alkynyl, C 1-6 alkoxy, 3 to 8 membered cycloalkyl, 4 to 8 membered heterocyclic group, -O- 3 to 8-membered cycloalkyl, -O-4 to 8-membered heterocycloalkyl, said alkyl, alkenyl, alkynyl, alkoxy, alkylthio, cycloalkyl, heterocyclyl or heterocyclic Alkyl is optionally further replaced by 0 to 4 members selected from H, halogen, OH, cyano, oxo, CF 3 , COOH, NH 2 , C 1-4 alkyl, C 1-4 alkoxy, 4-7 member Substituents of heterocyclyl or C 3-6 cycloalkyl, said heterocyclyl or heterocycloalkyl containing 1 to 4 heteroatoms selected from O, S, N;
或者两个Ra或两个Rb与其相连接的原子共同形成3至12元碳环基或4至12元杂环基,所述的碳环基或杂环基任选进一步被0至4个选自H、卤素、OH、氰基、CF3、COOH、NH2、 -NH(C1-6烷基)、-N(C1-6烷基)2、C1-4烷基或C1-4烷氧基的取代基所取代,所述的杂环基含有1 至4个选自O、S、N的杂原子;Or two R a or two R b together form a 3 to 12 membered carbocyclic group or a 4 to 12 membered heterocyclic group, and the carbocyclic group or heterocyclic group is optionally further replaced by 0 to 4 one selected from H, halogen, OH, cyano, CF 3 , COOH, NH 2 , -NH(C 1-6 alkyl), -N(C 1-6 alkyl) 2 , C 1-4 alkyl or Substituted by C 1-4 alkoxy substituents, the heterocyclic group contains 1 to 4 heteroatoms selected from O, S, N;
L1、L2或L3各自独立地选自键、O、-CH2-、-OCH2-、-CH2O-、-C(=O)-、S或NRx;L 1 , L 2 or L 3 are each independently selected from a bond, O, -CH 2 -, -OCH 2 -, -CH 2 O-, -C(=O)-, S or NR x ;
Rx选自H、C1-6烷基或C3-6环烷基,所述的烷基或环烷基任选进一步被0至4个选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、C1-4烷基或C1-4烷氧基的取代基所取代;R x is selected from H, C 1-6 alkyl or C 3-6 cycloalkyl, and the alkyl or cycloalkyl is optionally further replaced by 0 to 4 selected from H, halogen, OH, cyano, oxo , CF 3 , COOH, NH 2 , C 1-4 alkyl or C 1-4 alkoxy substituents;
R1选自卤素、OH、氰基、COOH、NH2、C1-6烷基、C2-6烯基、C2-6炔基、C1-6烷氧基、 NH2、-(CH2)q-C(=O)C1-6烷基、-(CH2)q-C(=O)-3至12元杂环、-(CH2)q-C(=O)-C3-10碳环、-(CH2)q-3 至12元杂环或-(CH2)q-C3-10碳环,所述的CH2、烷基、烯基、炔基、烷氧基、碳环或杂环任选进一步被0至4个R1a取代,所述的杂环含有1至4个选自O、S、N的杂原子;R 1 is selected from halogen, OH, cyano, COOH, NH 2 , C 1-6 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, C 1-6 alkoxy, NH 2 , -( CH 2 ) q -C(=O)C 1-6 alkyl, -(CH 2 ) q -C(=O)-3 to 12-membered heterocycle, -(CH 2 ) q -C(=O)- C 3-10 carbocycle, -(CH 2 ) q -3 to 12-membered heterocycle or -(CH 2 ) q -C 3-10 carbocycle, the CH 2 , alkyl, alkenyl, alkynyl, Alkoxy, carbocycle or heterocycle are optionally further substituted by 0 to 4 R 1a , said heterocycle containing 1 to 4 heteroatoms selected from O, S, N;
R1a各自独立地选自H、卤素、oxo、OH、氰基、CF3、COOH、NH2、-NH(C1-6烷基)、-N(C1-6烷基)2、C1-6烷基、C2-6烯基、C2-6炔基、-C1-6亚烷基-OH、-(CH2)q-C3-10碳环、-(CH2)q-3至12 元杂环、-O-C3-10碳环、-O-3至12元杂环、C1-6烷氧基、-N(C1-6烷基)C(=O)-3至12元杂环的取代基所取代,所述的烷基、CH2、烷氧基、烯基、炔基、亚烷基、碳环或杂环任选进一步被 0至4个选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、C1-6烷基、C1-6烷氧基、C3-6环烷基、3至6元杂环烷基的取代基所取代,所述的杂环或杂环烷基含有1至4个选自O、S、 N的杂原子;Each R 1a is independently selected from H, halogen, oxo, OH, cyano, CF 3 , COOH, NH 2 , -NH(C 1-6 alkyl), -N(C 1-6 alkyl) 2 , C 1-6 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, -C 1-6 alkylene-OH, -(CH 2 ) q -C 3-10 carbocycle, -(CH 2 ) q -3 to 12-membered heterocycle, -OC 3-10 carbocycle, -O-3 to 12-membered heterocycle, C 1-6 alkoxy, -N(C 1-6 alkyl)C(=O) -substituted by a substituent of a 3- to 12-membered heterocycle, said alkyl, CH 2 , alkoxy, alkenyl, alkynyl, alkylene, carbocycle or heterocycle is optionally further selected from 0 to 4 From H, halogen, OH, cyano, oxo, CF 3 , COOH, NH 2 , C 1-6 alkyl, C 1-6 alkoxy, C 3-6 cycloalkyl, 3 to 6 membered heterocycloalkane The substituent of the group is substituted, and the heterocycle or heterocycloalkyl group contains 1 to 4 heteroatoms selected from O, S, N;
R2选自C1-6烷基、C3-10碳环基、3至10元杂环基、C6-10芳基或5至10元杂芳基,所述的烷基、碳环基、杂环基、芳基或杂芳基任选进一步被0至7个R2a取代,所述的杂环基或杂芳基含有1至4个选自O、S、N的杂原子;R is selected from C 1-6 alkyl , C 3-10 carbocyclyl, 3-10 membered heterocyclic group, C 6-10 aryl or 5-10 membered heteroaryl, the alkyl, carbocyclic Base, heterocyclyl, aryl or heteroaryl are optionally further substituted by 0 to 7 R 2a , said heterocyclyl or heteroaryl containing 1 to 4 heteroatoms selected from O, S, N;
R2a各自独立的选自H、卤素、oxo、OH、氰基、CF3、COOH、NH2、-NH(C1-6烷基)、-N(C1-6烷基)2、C1-6烷基、C1-6烷氧基、C1-6烷硫基、C2-6烯基、C2-6炔基、C3-6环烷基、3至6元杂环烷基、-O-C3-6环烷基、-O-3至6元杂环烷基、C6-10芳基或5至10元杂芳基,所述的烷基、烷氧基、烷硫基、烯基、炔基、环烷基、杂环烷基、芳基、杂芳基任选进一步被0至4个选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、C1-6烷基、C1-6烷氧基、C3-6环烷基、3至6元杂环烷基的取代基所取代,所述的杂环烷基、杂环基或杂芳基含有1至4个选自O、S、N的杂原子;或R 2a are each independently selected from H, halogen, oxo, OH, cyano, CF 3 , COOH, NH 2 , -NH(C 1-6 alkyl), -N(C 1-6 alkyl) 2 , C 1-6 alkyl, C 1-6 alkoxy, C 1-6 alkylthio, C 2-6 alkenyl, C 2-6 alkynyl, C 3-6 cycloalkyl, 3-6 membered heterocycle Alkyl, -OC 3-6 cycloalkyl, -O-3 to 6 membered heterocycloalkyl, C 6-10 aryl or 5 to 10 membered heteroaryl, said alkyl, alkoxy, alkane Thio, alkenyl, alkynyl, cycloalkyl, heterocycloalkyl, aryl, heteroaryl are optionally further selected from 0 to 4 selected from H, halogen, OH, cyano, oxo, CF 3 , COOH, Substituents of NH 2 , C 1-6 alkyl, C 1-6 alkoxy, C 3-6 cycloalkyl, 3 to 6 membered heterocycloalkyl, said heterocycloalkyl, heterocycle radical or heteroaryl containing 1 to 4 heteroatoms selected from O, S, N; or
R2a各自独立的选自-NHC(O)C1-6烷基、-NHS(O)2C1-6烷基、-P(O)(C1-6烷基)2,所述的烷基任选进一步被0至4个选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、C1-6烷基、C1-6烷氧基、C3-6环烷基、3至6元杂环烷基的取代基所取代,所述的杂环烷基含有1至4个选自 O、S、N的杂原子;R 2a are each independently selected from -NHC(O)C 1-6 alkyl, -NHS(O) 2 C 1-6 alkyl, -P(O)(C 1-6 alkyl) 2 , the Alkyl is optionally further replaced by 0 to 4 members selected from H, halogen, OH, cyano, oxo, CF 3 , COOH, NH 2 , C 1-6 alkyl, C 1-6 alkoxy, C 3-6 Cycloalkyl, 3 to 6-membered heterocycloalkyl substituents, the heterocycloalkyl contains 1 to 4 heteroatoms selected from O, S, N;
或者两个R2a与其直接相连的原子共同形成3至12元碳环基或4至12元杂环基,所述的碳环基或杂环基任选进一步被0至4个选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、 C1-4烷基或C1-4烷氧基的取代基所取代,所述的杂环基含有1至4个选自O、S、N的杂原子;Or two R 2a and the atoms directly connected together form a 3 to 12-membered carbocyclic group or a 4 to 12-membered heterocyclic group, and the described carbocyclic group or heterocyclic group is optionally further replaced by 0 to 4 members selected from H, Halogen, OH, cyano, oxo, CF 3 , COOH, NH 2 , C 1-4 alkyl or C 1-4 alkoxy substituents, the heterocyclic group contains 1 to 4 selected from Heteroatoms of O, S, N;
q各自独立的选自0、1、2、3或4。q are each independently selected from 0, 1, 2, 3 or 4.
作为本发明的第二种实施方案,下述通式(I-aa)所示的化合物或者其立体异构体、氘代物、 N-氧化物、溶剂化物、前药、代谢产物、药学上可接受的盐或共晶,As the second embodiment of the present invention, the compound represented by the following general formula (I-aa) or its stereoisomer, deuterium, N-oxide, solvate, prodrug, metabolite, pharmaceutically available Accepted salts or co-crystals,
选自4至12元含氮杂环,所述的含氮杂环选自饱和或部分饱和的如下基团之一:单环、并环、桥环或螺环,所述的含氮杂环、单环、并环、桥环或螺环任选进一步被0 至4个Ra取代,环A1中N-H不被取代; Selected from 4 to 12 membered nitrogen-containing heterocyclic rings, the nitrogen-containing heterocyclic rings are selected from one of the following saturated or partially saturated groups: monocyclic, parallel rings, bridged rings or spiro rings, and the nitrogen-containing heterocyclic rings , monocyclic ring, parallel ring, bridging ring or spiro ring are optionally further substituted by 0 to 4 R, and NH in ring A is not substituted;
n选自0、1、2、3或4;n is selected from 0, 1, 2, 3 or 4;
条件是不为m1选自0、1或2;requirement is not for m1 is selected from 0, 1 or 2;
条件是化合物不为 provided that the compound is not
其余定义与本发明第一种实施方案相同。The rest of the definitions are the same as the first embodiment of the present invention.
作为本发明的第三种实施方案,前述通式(I-aa)所示的化合物或者其立体异构体、氘代物、 N-氧化物、溶剂化物、前药、代谢产物、药学上可接受的盐或共晶,As the third embodiment of the present invention, the compound represented by the aforementioned general formula (I-aa) or its stereoisomer, deuterated substance, N-oxide, solvate, prodrug, metabolite, pharmaceutically acceptable salt or eutectic,
选自未被取代的或者取代的如下基团之一: 当被取代时,任选进一步被0至4个Ra取代基所取代,环A1中N-H不被取代; One of the following unsubstituted or substituted groups: When substituted, optionally further substituted by 0 to 4 R substituents, NH in ring A is not substituted;
选自 其左边与L2连接,上方与环A1连接; selected from Its left side is connected with L 2 , and its upper part is connected with ring A 1 ;
m各自独立地选自0、1、2或3;each m is independently selected from 0, 1, 2 or 3;
其余定义与本发明第一种或第二种实施方案相同。The remaining definitions are the same as in the first or second embodiment of the present invention.
作为本发明的第四种实施方案,前述通式(I-aa)所示的化合物或者其立体异构体、氘代物、 N-氧化物、溶剂化物、前药、代谢产物、药学上可接受的盐或共晶,As the fourth embodiment of the present invention, the compound represented by the aforementioned general formula (I-aa) or its stereoisomer, deuterated substance, N-oxide, solvate, prodrug, metabolite, pharmaceutically acceptable salt or eutectic,
选自 selected from
选自 其左边与L2连接,上方与环A1连接; selected from Its left side is connected with L 2 , and its upper part is connected with ring A 1 ;
m各自独立地选自0、1、2或3;each m is independently selected from 0, 1, 2 or 3;
其余定义与本发明第一种、第二种或第三种实施方案相同。The remaining definitions are the same as the first, second or third embodiment of the present invention.
作为本发明的第五种实施方案,前述通式(I-aa)所示的化合物或者其立体异构体、氘代物、溶剂化物、前药、代谢产物、药学上可接受的盐或共晶,As the fifth embodiment of the present invention, the compound represented by the aforementioned general formula (I-aa) or its stereoisomer, deuterated product, solvate, prodrug, metabolite, pharmaceutically acceptable salt or co-crystal ,
L2选自键、O或-C(=O)-;L is selected from a bond, O or -C (=O)-;
R2选自C3-10碳环基、3至10元杂环基、C6-10芳基或5至10元杂芳基,所述的碳环基、杂环基、芳基或杂芳基任选进一步被0至5个R2a取代,所述的杂环基或杂芳基含有1至4个选自O、S、N的杂原子;R 2 is selected from C 3-10 carbocyclyl, 3 to 10 membered heterocyclyl, C 6-10 aryl or 5 to 10 membered heteroaryl, the carbocyclyl, heterocyclyl, aryl or heterocyclyl The aryl is optionally further substituted by 0 to 5 R 2a , and the heterocyclyl or heteroaryl contains 1 to 4 heteroatoms selected from O, S, N;
R2a各自独立的选自H、卤素、oxo、OH、氰基、CF3、COOH、NH2、-NH(C1-4烷基)、-N(C1-4烷基)2、C1-4烷基、C1-4烷氧基、C1-4烷硫基、C2-4烯基、C2-6炔基、C3-6环烷基、3至6元杂环烷基、-O-C3-6环烷基、-O-3至6元杂环烷基、C6-10芳基或5至10元杂芳基,所述的烷基、烷氧基、烷硫基、烯基、炔基、环烷基、杂环烷基、芳基、杂芳基任选进一步被0至4个选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、C1-4烷基、C1-4烷氧基、C3-6环烷基、3至6元杂环烷基的取代基所取代,所述的杂环烷基、杂环基或杂芳基含有1至4个选自O、S、N的杂原子;或R 2a are each independently selected from H, halogen, oxo, OH, cyano, CF 3 , COOH, NH 2 , -NH(C 1-4 alkyl), -N(C 1-4 alkyl) 2 , C 1-4 alkyl, C 1-4 alkoxy, C 1-4 alkylthio, C 2-4 alkenyl, C 2-6 alkynyl, C 3-6 cycloalkyl, 3 to 6 membered heterocycle Alkyl, -OC 3-6 cycloalkyl, -O-3 to 6 membered heterocycloalkyl, C 6-10 aryl or 5 to 10 membered heteroaryl, said alkyl, alkoxy, alkane Thio, alkenyl, alkynyl, cycloalkyl, heterocycloalkyl, aryl, heteroaryl are optionally further selected from 0 to 4 selected from H, halogen, OH, cyano, oxo, CF 3 , COOH, Substituents of NH 2 , C 1-4 alkyl, C 1-4 alkoxy, C 3-6 cycloalkyl, 3 to 6 membered heterocycloalkyl, the heterocycloalkyl, heterocycle radical or heteroaryl containing 1 to 4 heteroatoms selected from O, S, N; or
R2a各自独立的选自-NHC(O)C1-4烷基、-NHS(O)2C1-4烷基、-P(O)(C1-4烷基)2,所述的烷基任选进一步被0至4个选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、C1-6烷基、C1-6烷氧基、C3-6环烷基、3至6元杂环烷基的取代基所取代,所述的杂环烷基含有1至4个选自 O、S、N的杂原子;R 2a are each independently selected from -NHC(O)C 1-4 alkyl, -NHS(O) 2 C 1-4 alkyl, -P(O)(C 1-4 alkyl) 2 , the Alkyl is optionally further replaced by 0 to 4 members selected from H, halogen, OH, cyano, oxo, CF 3 , COOH, NH 2 , C 1-6 alkyl, C 1-6 alkoxy, C 3-6 Cycloalkyl, 3 to 6-membered heterocycloalkyl substituents, the heterocycloalkyl contains 1 to 4 heteroatoms selected from O, S, N;
或者两个R2a与其相连接的原子共同形成3至6元碳环基或4至6元杂环基,所述的碳环基或杂环基任选进一步被0至4个选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、C1-4烷基或C1-4烷氧基的取代基所取代,所述的杂环基含有1至4个选自O、S、N的杂原子;Or two R 2a together form a 3 to 6-membered carbocyclyl or a 4 to 6-membered heterocyclic group, and the carbocyclyl or heterocyclic group is optionally further replaced by 0 to 4 members selected from H, Halogen, OH, cyano, oxo, CF 3 , COOH, NH 2 , C 1-4 alkyl or C 1-4 alkoxy substituents, the heterocyclic group contains 1 to 4 selected from Heteroatoms of O, S, N;
其余定义与本发明第一种、第二种、第三种或第四种实施方案相同。The remaining definitions are the same as those of the first, second, third or fourth embodiment of the present invention.
作为本发明的第六种实施方案,前述通式(I-aa)所示的化合物或者其立体异构体、氘代物、溶剂化物、前药、代谢产物、药学上可接受的盐或共晶,As the sixth embodiment of the present invention, the compound represented by the aforementioned general formula (I-aa) or its stereoisomer, deuterated product, solvate, prodrug, metabolite, pharmaceutically acceptable salt or co-crystal ,
Rb各自独立的选自H、oxo、卤素、氰基、C1-4烷基、C1-4烷硫基、C2-4烯基、C2-4炔基、C1-4烷氧基、3至6元环烷基、4至6元杂环基、-O-3至6元环烷基、-O-4至6元杂环烷基,所述的烷基、烯基、炔基、烷氧基、烷硫基、环烷基、杂环基、杂环烷基任选进一步被0至4 个选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、C1-4烷基或C1-4烷氧基的取代基所取代,所述的杂环基或杂环烷基含有1至4个选自O、S、N的杂原子;R b are each independently selected from H, oxo, halogen, cyano, C 1-4 alkyl, C 1-4 alkylthio, C 2-4 alkenyl, C 2-4 alkynyl, C 1-4 alkane Oxygen, 3 to 6-membered cycloalkyl, 4 to 6-membered heterocyclyl, -O-3 to 6-membered cycloalkyl, -O-4 to 6-membered heterocycloalkyl, said alkyl, alkenyl , alkynyl, alkoxy, alkylthio, cycloalkyl, heterocyclyl, heterocycloalkyl are optionally further selected from 0 to 4 selected from H, halogen, OH, cyano, oxo, CF 3 , COOH, Substituents of NH 2 , C 1-4 alkyl or C 1-4 alkoxy, the heterocyclyl or heterocycloalkyl containing 1 to 4 heteroatoms selected from O, S, N;
或者两个Rb与其相连接的原子共同形成3至6元碳环基或4至6元杂环基,所述的碳环基或杂环基任选进一步被0至4个选自H、卤素、OH、氰基、CF3、COOH、NH2、C1-4烷基或C1-4烷氧基的取代基所取代,所述的杂环基含有1至4个选自O、S、N的杂原子;Or two R b and the atoms to which they are connected jointly form a 3-6 membered carbocyclic group or a 4-6 membered heterocyclic group, and the described carbocyclic group or heterocyclic group is optionally further replaced by 0 to 4 members selected from H, Halogen, OH, cyano, CF 3 , COOH, NH 2 , C 1-4 alkyl or C 1-4 alkoxy substituents, the heterocyclic group contains 1 to 4 selected from O, S, N heteroatoms;
R1a各自独立地选自H、卤素、oxo、OH、氰基、CF3、COOH、NH2、-NH(C1-4烷基)、-N(C1-4烷基)2、C1-4烷基、C2-4烯基、C2-4炔基、-C1-4亚烷基-OH、-(CH2)q-C3-8碳环、-(CH2)q-3至8元杂环、-O-C3-8碳环、-O-3至8元杂环、C1-4烷氧基、-N(C1-4烷基)C(=O)-3至8元杂环的取代基所取代,所述的烷基、CH2、烷氧基、烯基、炔基、亚烷基、碳环或杂环任选进一步被0至 4个选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、C1-6烷基、C1-6烷氧基、C3-6环烷基、 3至6元杂环烷基的取代基所取代,所述的杂环或杂环烷基含有1至4个选自O、S、N的杂原子;Each R 1a is independently selected from H, halogen, oxo, OH, cyano, CF 3 , COOH, NH 2 , -NH(C 1-4 alkyl), -N(C 1-4 alkyl) 2 , C 1-4 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, -C 1-4 alkylene-OH, -(CH 2 ) q -C 3-8 carbocycle, -(CH 2 ) q -3 to 8-membered heterocycle, -OC 3-8 carbocycle, -O-3 to 8-membered heterocycle, C 1-4 alkoxy, -N(C 1-4 alkyl)C(=O) -substituted by a substituent of a 3- to 8-membered heterocycle, said alkyl, CH 2 , alkoxy, alkenyl, alkynyl, alkylene, carbocycle or heterocycle is optionally further selected from 0 to 4 From H, halogen, OH, cyano, oxo, CF 3 , COOH, NH 2 , C 1-6 alkyl, C 1-6 alkoxy, C 3-6 cycloalkyl, 3 to 6 membered heterocycloalkane The substituent of the group is substituted, and the heterocycle or heterocycloalkyl group contains 1 to 4 heteroatoms selected from O, S, N;
q各自独立地选自0、1、2、3或4;q is each independently selected from 0, 1, 2, 3 or 4;
其余定义与本发明第一种、第二种、第三种、第四种或第五种实施方案相同。The remaining definitions are the same as the first, second, third, fourth or fifth embodiment of the present invention.
作为本发明的第七种实施方案,前述通式(I-aa)所示的化合物或者其立体异构体、氘代物、溶剂化物、前药、代谢产物、药学上可接受的盐或共晶,As the seventh embodiment of the present invention, the compound represented by the aforementioned general formula (I-aa) or its stereoisomer, deuterated product, solvate, prodrug, metabolite, pharmaceutically acceptable salt or co-crystal ,
R2选自取代的或者未被取代的如下基团之一:苯基、萘基、噻吩基、呋喃基、吡咯基、噻唑基、异噻唑基、噁唑基、异噁唑基、咪唑基、吡唑基、吲唑、苯并咪唑基、苯并吡唑基、苯并噻唑基、苯并噻吩基、苯并呋喃基、苯并嘧啶基、苯并吡啶基或吡啶基,当被取代时,任选被0至5个R2a取代;R is selected from one of substituted or unsubstituted following groups : phenyl, naphthyl, thienyl, furyl, pyrrolyl, thiazolyl, isothiazolyl, oxazolyl, isoxazolyl, imidazolyl , pyrazolyl, indazole, benzimidazolyl, benzopyrazolyl, benzothiazolyl, benzothienyl, benzofuryl, benzopyrimidinyl, benzopyridyl or pyridyl, when substituted , optionally substituted by 0 to 5 R 2a ;
R2a各自独立的选自H、F、Cl、Br、I、oxo、OH、NH2、氰基、CF3、-SCH3、甲基、乙基、丙基、异丙基、乙烯基、丙烯基、乙炔基、丙炔基、甲氧基、乙氧基、丙氧基、异丙氧基、环丙基、环丁基、环戊基、环己基、-O-环丙基、-O-环丁基、-O-环戊基、-O-环己基,所述的甲基、乙基、丙基、异丙基、乙烯基、丙烯基、乙炔基、丙炔基、甲氧基、乙氧基、丙氧基、异丙氧基、环丙基、环丁基、环戊基、环己基任选进一步被0至4个选自H、F、Cl、Br、I、 OH、氰基、oxo、CF3、COOH、NH2、甲基、乙基、丙基、甲氧基、乙氧基、丙氧基或异丙氧基的取代基所取代;或R 2a are each independently selected from H, F, Cl, Br, I, oxo, OH, NH 2 , cyano, CF 3 , -SCH 3 , methyl, ethyl, propyl, isopropyl, vinyl, propenyl, ethynyl, propynyl, methoxy, ethoxy, propoxy, isopropoxy, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, -O-cyclopropyl, - O-cyclobutyl, -O-cyclopentyl, -O-cyclohexyl, methyl, ethyl, propyl, isopropyl, vinyl, propenyl, ethynyl, propynyl, methoxy Base, ethoxy, propoxy, isopropoxy, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl are optionally further selected from 0 to 4 selected from H, F, Cl, Br, I, OH , cyano, oxo, CF 3 , COOH, NH 2 , methyl, ethyl, propyl, methoxy, ethoxy, propoxy or isopropoxy; or
R2a各自独立的选自-NHC(O)CH3、-NHS(O)2CH3、-P(O)(CH3)2、环戊烯基、苯基、吡唑基,所述的-CH3、环戊烯基、苯基、吡唑基任选进一步被0至4个选自H、F、Cl、Br、I、OH、氰基、oxo、CF3、COOH、NH2、甲基、乙基、丙基、甲氧基、乙氧基、丙氧基或异丙氧基的取代基所取代;R 2a are each independently selected from -NHC(O)CH 3 , -NHS(O) 2 CH 3 , -P(O)(CH 3 ) 2 , cyclopentenyl, phenyl, pyrazolyl, and the -CH 3 , cyclopentenyl, phenyl, pyrazolyl are optionally further replaced by 0 to 4 selected from H, F, Cl, Br, I, OH, cyano, oxo, CF 3 , COOH, NH 2 , Substituents of methyl, ethyl, propyl, methoxy, ethoxy, propoxy or isopropoxy;
或者两个R2a与其相连接的原子共同形成3至6元碳环基或4至6元杂环基,所述的碳环基或杂环基任选进一步被0至4个选自H、F、Cl、Br、I、OH、氰基、oxo、CF3、COOH、 NH2、甲基、乙基、丙基、甲氧基、乙氧基、丙氧基或异丙氧基的取代基所取代,所述的杂环基含有1至4个选自O、S、N的杂原子;Or two R 2a together form a 3 to 6-membered carbocyclyl or a 4 to 6-membered heterocyclic group, and the carbocyclyl or heterocyclic group is optionally further replaced by 0 to 4 members selected from H, Substitution by F, Cl, Br, I, OH, cyano, oxo, CF 3 , COOH, NH 2 , methyl, ethyl, propyl, methoxy, ethoxy, propoxy or isopropoxy Substituted by a group, the heterocyclic group contains 1 to 4 heteroatoms selected from O, S, N;
Rb各自独立的选自H、oxo、F、Cl、Br、I、氰基、OH、NH2、甲基、乙基、丙基、异丙基、乙烯基、丙烯基、乙炔基、丙炔基、甲氧基、乙氧基、丙氧基、异丙氧基、环丙基、环丁基、环戊基、环己基、-O-环丙基、-O-环丁基、-O-环戊基、-O-环己基,所述的甲基、乙基、丙基、乙烯基、丙烯基、乙炔基、丙炔基、甲氧基、乙氧基、丙氧基、异丙氧基任选进一步被 0至4个选自H、F、Cl、Br、I、OH、氰基、oxo、CF3、COOH、NH2、甲基、乙基、丙基、甲氧基、乙氧基、丙氧基或异丙氧基的取代基所取代;R b are each independently selected from H, oxo, F, Cl, Br, I, cyano, OH, NH 2 , methyl, ethyl, propyl, isopropyl, vinyl, propenyl, ethynyl, propane Alkynyl, methoxy, ethoxy, propoxy, isopropoxy, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, -O-cyclopropyl, -O-cyclobutyl, - O-cyclopentyl, -O-cyclohexyl, the methyl, ethyl, propyl, vinyl, propenyl, ethynyl, propynyl, methoxy, ethoxy, propoxy, iso Propoxy is optionally further replaced by 0 to 4 members selected from H, F, Cl, Br, I, OH, cyano, oxo, CF 3 , COOH, NH 2 , methyl, ethyl, propyl, methoxy , ethoxy, propoxy or isopropoxy substituents;
R1a各自独立地选自H、F、Cl、Br、I、oxo、OH、氰基、CF3、COOH、NH2、甲基、乙基、丙基、乙烯基、丙烯基、乙炔基、丙炔基、甲氧基、乙氧基、丙氧基、异丙氧基、环丙基、环丁基、环戊基、环己基、-O-环丙基、-O-环丁基、-O-环戊基、-O-环己基,所述的甲基、乙基、丙基、乙烯基、丙烯基、乙炔基、丙炔基、甲氧基、乙氧基、丙氧基、异丙氧基任选进一步被0至4个选自H、F、Cl、Br、I、OH、氰基、oxo、CF3、COOH、NH2、甲基、乙基、丙基、甲氧基、乙氧基、丙氧基或异丙氧基的取代基所取代;Each R 1a is independently selected from H, F, Cl, Br, I, oxo, OH, cyano, CF 3 , COOH, NH 2 , methyl, ethyl, propyl, vinyl, propenyl, ethynyl, propynyl, methoxy, ethoxy, propoxy, isopropoxy, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, -O-cyclopropyl, -O-cyclobutyl, -O-cyclopentyl, -O-cyclohexyl, the methyl, ethyl, propyl, vinyl, propenyl, ethynyl, propynyl, methoxy, ethoxy, propoxy, Isopropoxy is optionally further replaced by 0 to 4 members selected from H, F, Cl, Br, I, OH, cyano, oxo, CF 3 , COOH, NH 2 , methyl, ethyl, propyl, methoxy Substituents of ethoxy, propoxy or isopropoxy;
其余定义与本发明第一种、第二种、第三种、第四种、第五种或第六种实施方案相同。The remaining definitions are the same as those of the first, second, third, fourth, fifth or sixth embodiment of the present invention.
作为本发明的第八种实施方案,下述通式(I-aaa)所示的化合物或者其立体异构体、氘代物、溶剂化物、前药、代谢产物、药学上可接受的盐或共晶,As the eighth embodiment of the present invention, the compound represented by the following general formula (I-aaa) or its stereoisomer, deuterated product, solvate, prodrug, metabolite, pharmaceutically acceptable salt or co- crystal,
L2选自键或-C(=O)-;L is selected from a bond or -C (=O)-;
Rb各自独立的选自H、oxo、F、Cl、Br、I、氰基、OH、NH2、甲基、乙基、甲氧基、乙氧基、丙氧基、异丙氧基、环丙基、-O-环丙基、乙烯基、乙炔基,所述的甲基、乙基、丙基、甲氧基、乙氧基、丙氧基、丙氧基、异丙氧基、乙烯基、乙炔基、环丙基任选进一步被0至4 个选自H、F、Cl、Br、I、OH、氰基、oxo、CF3、COOH、NH2、甲基、乙基、丙基、甲氧基、乙氧基或丙氧基所取代;R b are each independently selected from H, oxo, F, Cl, Br, I, cyano, OH, NH 2 , methyl, ethyl, methoxy, ethoxy, propoxy, isopropoxy, Cyclopropyl, -O-cyclopropyl, vinyl, ethynyl, the methyl, ethyl, propyl, methoxy, ethoxy, propoxy, propoxy, isopropoxy, Vinyl, ethynyl, and cyclopropyl are optionally further replaced by 0 to 4 selected from H, F, Cl, Br, I, OH, cyano, oxo, CF 3 , COOH, NH 2 , methyl, ethyl, Propyl, methoxy, ethoxy or propoxy;
R2选自取代的或者未被取代的如下基团之一:苯基、吡啶、苯并噻唑基、苯并噻吩基、苯并呋喃基、苯并吡唑基或萘基,当被取代时,任选被0至5个R2a取代;R is selected from one of substituted or unsubstituted phenyl, pyridine, benzothiazolyl, benzothienyl, benzofuryl, benzopyrazolyl or naphthyl, when substituted , optionally substituted by 0 to 5 R 2a ;
R2a各自独立的选自H、F、Cl、OH、氰基、NH2、CF3、OCF3、-SCH3、甲基、乙基、丙基、异丙基、甲氧基、乙氧基、乙炔基、丙炔基、环丙基、环丁基或环戊基,所述甲基、乙基、丙基、异丙基、甲氧基、乙氧基、乙炔基、丙炔基、环丙基、环丁基或环戊基任选进一步被0 至4个选自H、F、Cl、OH、甲基、乙基或环丙基的取代基所取代;或R 2a are each independently selected from H, F, Cl, OH, cyano, NH 2 , CF 3 , OCF 3 , -SCH 3 , methyl, ethyl, propyl, isopropyl, methoxy, ethoxy base, ethynyl, propynyl, cyclopropyl, cyclobutyl or cyclopentyl, the methyl, ethyl, propyl, isopropyl, methoxy, ethoxy, ethynyl, propynyl , cyclopropyl, cyclobutyl or cyclopentyl are optionally further substituted by 0 to 4 substituents selected from H, F, Cl, OH, methyl, ethyl or cyclopropyl; or
R2a各自独立的选自丙烯基、-NHC(O)CH3、-NHS(O)2CH3、-P(O)(CH3)2、环戊烯基、苯基、吡唑基,所述-CH3、丙烯基、环戊烯基、苯基、吡唑基任选进一步被0至4个选自H、F、Cl、OH、甲基、乙基或环丙基的取代基所取代;R 2a are each independently selected from propenyl, -NHC(O)CH 3 , -NHS(O) 2 CH 3 , -P(O)(CH 3 ) 2 , cyclopentenyl, phenyl, pyrazolyl, The -CH 3 , propenyl, cyclopentenyl, phenyl, pyrazolyl are optionally further substituted by 0 to 4 substituents selected from H, F, Cl, OH, methyl, ethyl or cyclopropyl replaced by
其余定义与本发明第一种、第二种、第三种、第四种、第五种或第六种实施方案相同。The remaining definitions are the same as those of the first, second, third, fourth, fifth or sixth embodiment of the present invention.
作为本发明的第九种实施方案,下述通式(I-aaaa)所示的化合物或者其立体异构体、氘代物、溶剂化物、前药、代谢产物、药学上可接受的盐或共晶,As the ninth embodiment of the present invention, the compound represented by the following general formula (I-aaaa) or its stereoisomer, deuterated product, solvate, prodrug, metabolite, pharmaceutically acceptable salt or co- crystal,
Rb选自H、F、Cl、氰基、OH、NH2、甲基、乙基、甲氧基、乙氧基、环丙基、-O-环丙基;R b is selected from H, F, Cl, cyano, OH, NH 2 , methyl, ethyl, methoxy, ethoxy, cyclopropyl, -O-cyclopropyl;
R2选自取代的或者未被取代的如下基团之一:苯基、吡啶、苯并噻唑基、苯并噻吩基、苯并呋喃基、苯并吡唑基或萘基,当被取代时,任选被0至5个R2a取代;R is selected from one of substituted or unsubstituted phenyl, pyridine, benzothiazolyl, benzothienyl, benzofuryl, benzopyrazolyl or naphthyl, when substituted , optionally substituted by 0 to 5 R 2a ;
R2a各自独立的选自H、F、Cl、OH、氰基、NH2、CF3、OCF3、-SCH3、甲基、乙基、丙基、异丙基、甲氧基、乙氧基、乙炔基、丙炔基、环丙基、环丁基或环戊基,所述甲基、乙基、丙基、异丙基、甲氧基、乙氧基、乙炔基、丙炔基、环丙基、环丁基或环戊基任选进一步被0 至4个选自H、F、Cl、OH、甲基、乙基或环丙基的取代基所取代;或R 2a are each independently selected from H, F, Cl, OH, cyano, NH 2 , CF 3 , OCF 3 , -SCH 3 , methyl, ethyl, propyl, isopropyl, methoxy, ethoxy base, ethynyl, propynyl, cyclopropyl, cyclobutyl or cyclopentyl, the methyl, ethyl, propyl, isopropyl, methoxy, ethoxy, ethynyl, propynyl , cyclopropyl, cyclobutyl or cyclopentyl are optionally further substituted by 0 to 4 substituents selected from H, F, Cl, OH, methyl, ethyl or cyclopropyl; or
R2a各自独立的选自丙烯基、-NHC(O)CH3、-NHS(O)2CH3、-P(O)(CH3)2、环戊烯基、苯基、吡唑基,所述-CH3、丙烯基、环戊烯基、苯基、吡唑基任选进一步被0至4个选自H、F、Cl、OH、甲基、乙基或环丙基的取代基所取代。R 2a are each independently selected from propenyl, -NHC(O)CH 3 , -NHS(O) 2 CH 3 , -P(O)(CH 3 ) 2 , cyclopentenyl, phenyl, pyrazolyl, The -CH 3 , propenyl, cyclopentenyl, phenyl, pyrazolyl are optionally further replaced by 0 to 4 substituents selected from H, F, Cl, OH, methyl, ethyl or cyclopropyl replaced.
作为本发明的第十种实施方案,下述通式(I-b)所示的化合物或者其立体异构体、氘代物、溶剂化物、前药、代谢产物、药学上可接受的盐或共晶,As the tenth embodiment of the present invention, the compound represented by the following general formula (I-b) or its stereoisomer, deuterium, solvate, prodrug, metabolite, pharmaceutically acceptable salt or co-crystal,
R2选自苯基、苯并吡唑基、苯并呋喃基、吡啶基,所述的苯基被1至4个R2a取代,所述的苯并吡唑基、苯并呋喃基、吡啶基被0至4个R2a取代;R 2 is selected from phenyl, benzopyrazolyl, benzofuryl, pyridyl, the phenyl is substituted by 1 to 4 R 2a , and the benzopyrazolyl, benzofuryl, pyridine The group is substituted by 0 to 4 R 2a ;
R2a各自独立的选自F、Cl、OH、氰基、NH2、CF3、OCF3、-SCH3、甲基、乙基、丙基、异丙基、甲氧基、乙氧基、乙炔基、丙炔基、环丙基、环丁基、环戊基、丙烯基、-NHC(O)CH3、 -NHS(O)2CH3、-P(O)(CH3)2、环戊烯基、苯基、吡唑基,所述甲基、乙基、丙基、异丙基、甲氧基、乙氧基、乙炔基、丙炔基、环丙基、环丁基、环戊基、丙烯基、-NHC(O)CH3、-NHS(O)2CH3、 -P(O)(CH3)2、环戊烯基、苯基、吡唑基任选进一步被0至4个选自H、F、Cl、OH、甲基、乙基或环丙基的取代基所取代。R 2a are each independently selected from F, Cl, OH, cyano, NH 2 , CF 3 , OCF 3 , -SCH 3 , methyl, ethyl, propyl, isopropyl, methoxy, ethoxy, Ethynyl, propynyl, cyclopropyl, cyclobutyl, cyclopentyl, propenyl, -NHC(O)CH 3 , -NHS(O) 2 CH 3 , -P(O)(CH 3 ) 2 , Cyclopentenyl, phenyl, pyrazolyl, methyl, ethyl, propyl, isopropyl, methoxy, ethoxy, ethynyl, propynyl, cyclopropyl, cyclobutyl, Cyclopentyl, propenyl, -NHC(O)CH 3 , -NHS(O) 2 CH 3 , -P(O)(CH 3 ) 2 , cyclopentenyl, phenyl, pyrazolyl are optionally further 0 to 4 substituents selected from H, F, Cl, OH, methyl, ethyl or cyclopropyl.
作为本发明的第十一种实施方案,下述通式(I-aaaa)所示的化合物或者其立体异构体、氘代物、溶剂化物、前药、代谢产物、药学上可接受的盐或共晶,As the eleventh embodiment of the present invention, the compound represented by the following general formula (I-aaaa) or its stereoisomer, deuterated product, solvate, prodrug, metabolite, pharmaceutically acceptable salt or eutectic,
Rb选自H、F、Cl、氰基、OH、NH2、甲基、乙基、甲氧基、乙氧基、环丙基、-O-环丙基;R b is selected from H, F, Cl, cyano, OH, NH 2 , methyl, ethyl, methoxy, ethoxy, cyclopropyl, -O-cyclopropyl;
R2选自取代的如下基团之一:苯基、吡啶、苯并噻唑基、苯并噻吩基、苯并呋喃基、苯并吡唑基或萘基,当被取代时,被1至5个R2a取代;R is selected from one of the following substituted groups: phenyl, pyridine, benzothiazolyl, benzothienyl, benzofuryl, benzopyrazolyl or naphthyl, when substituted, by 1 to 5 R 2a replaces;
R2a各自独立的选自H、F、Cl、OH、氰基、NH2、CF3、OCF3、-SCH3、甲基、乙基、丙基、异丙基、甲氧基、乙氧基、乙炔基、丙炔基、环丙基、环丁基或环戊基,所述甲基、乙基、丙基、异丙基、甲氧基、乙氧基、乙炔基、丙炔基、环丙基、环丁基或环戊基任选进一步被0 至4个选自H、F、Cl、OH、甲基、乙基或环丙基的取代基所取代;R 2a are each independently selected from H, F, Cl, OH, cyano, NH 2 , CF 3 , OCF 3 , -SCH 3 , methyl, ethyl, propyl, isopropyl, methoxy, ethoxy base, ethynyl, propynyl, cyclopropyl, cyclobutyl or cyclopentyl, the methyl, ethyl, propyl, isopropyl, methoxy, ethoxy, ethynyl, propynyl , cyclopropyl, cyclobutyl or cyclopentyl are optionally further substituted by 0 to 4 substituents selected from H, F, Cl, OH, methyl, ethyl or cyclopropyl;
条件是至少有一个R2a选自环丙基、环丁基或环戊基。with the proviso that at least one R2a is selected from cyclopropyl, cyclobutyl or cyclopentyl.
作为本发明的第十二种实施方案,下述通式(I-c)或(I-d)所示的化合物或者其立体异构体、氘代物、溶剂化物、前药、代谢产物、药学上可接受的盐或共晶,As the twelfth embodiment of the present invention, the compound represented by the following general formula (I-c) or (I-d) or its stereoisomer, deuterium, solvate, prodrug, metabolite, pharmaceutically acceptable salt or eutectic,
Rb选自H、F、Cl、氰基、OH、NH2、甲基、乙基、甲氧基、乙氧基、环丙基、-O-环丙基;R b is selected from H, F, Cl, cyano, OH, NH 2 , methyl, ethyl, methoxy, ethoxy, cyclopropyl, -O-cyclopropyl;
R2选自取代的或者未被取代的如下基团之一:苯基、吡啶、苯并噻唑基、苯并噻吩基、苯并呋喃基、苯并吡唑基或萘基,当被取代时,任选被0至5个R2a取代;R is selected from one of substituted or unsubstituted phenyl, pyridine, benzothiazolyl, benzothienyl, benzofuryl, benzopyrazolyl or naphthyl, when substituted , optionally substituted by 0 to 5 R 2a ;
R2a各自独立的选自H、F、Cl、OH、氰基、NH2、CF3、OCF3、-SCH3、甲基、乙基、丙基、异丙基、甲氧基、乙氧基、乙炔基、丙炔基、环丙基、环丁基或环戊基,所述甲基、乙基、丙基、异丙基、甲氧基、乙氧基、乙炔基、丙炔基、环丙基、环丁基或环戊基任选进一步被0 至4个选自H、F、Cl、OH、甲基、乙基或环丙基的取代基所取代;R 2a are each independently selected from H, F, Cl, OH, cyano, NH 2 , CF 3 , OCF 3 , -SCH 3 , methyl, ethyl, propyl, isopropyl, methoxy, ethoxy base, ethynyl, propynyl, cyclopropyl, cyclobutyl or cyclopentyl, the methyl, ethyl, propyl, isopropyl, methoxy, ethoxy, ethynyl, propynyl , cyclopropyl, cyclobutyl or cyclopentyl are optionally further substituted by 0 to 4 substituents selected from H, F, Cl, OH, methyl, ethyl or cyclopropyl;
m选自0、1、2或3。本发明涉及一种下述化合物或者其立体异构体、氘代物、溶剂化物、前药、代谢产物、药学上可接受的盐或共晶,其中该化合物选自如下结构之一:m is selected from 0, 1, 2 or 3. The present invention relates to a compound or its stereoisomer, deuterium, solvate, prodrug, metabolite, pharmaceutically acceptable salt or co-crystal, wherein the compound is selected from one of the following structures:
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,X1或X2各自独立地选自N或CRb。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), X 1 or X 2 are each independently selected from N or CR b .
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,X1、X2选自N。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), X 1 , X 2 are selected from N.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,X1、X2选自CRb。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), X 1 , X 2 are selected from CR b .
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,X1选自N,X2选自CRb。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), X 1 is selected from N, X 2 is selected from CR b .
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,X1选自CRb,X2选自N。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), X 1 is selected from CR b , and X 2 is selected from N.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,环A选自3至12元碳环基或4 至12元杂环基,所述的碳环基或杂环基任选进一步被0至6个Ra取代,所述的杂环基含有1 至4个(例如1、2、3或4个)选自O、S、N的杂原子。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), ring A is selected from 3 to 12 membered carbocyclic groups or 4 to 12 membered heterocyclic groups, and the carbon The cyclic or heterocyclic group is optionally further substituted by 0 to 6 R a , and the heterocyclic group contains 1 to 4 (eg 1, 2, 3 or 4) heteroatoms selected from O, S, N .
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,L1、L2或L3各自独立地选自键、 O、-CH2-、-OCH2-、-CH2O-、-C(=O)-、S或NRx。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), L 1 , L 2 or L 3 are each independently selected from a bond, O, -CH 2 -, -OCH 2 -, -CH 2 O-, -C(=O)-, S or NR x .
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,Rx选自H、C1-6烷基或C3-6环烷基,所述的烷基或环烷基任选进一步被0至4个(例如0、1、2、3或4个)选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、C1-4烷基或C1-4烷氧基的取代基所取代。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), R x is selected from H, C 1-6 alkyl or C 3-6 cycloalkyl, said Alkyl or cycloalkyl is optionally further selected from 0 to 4 (eg 0, 1, 2, 3 or 4) selected from H, halogen, OH, cyano, oxo, CF 3 , COOH, NH 2 , C 1 -4 alkyl or C 1-4 alkoxy substituents.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,L3选自键。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), L 3 is selected from a bond.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,选自 In some embodiments of the present invention related to general formula (Ia), (I-aa), (I-aaa), selected from
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,选自4至12元含氮杂环,所述的含氮杂环选自饱和或部分饱和的如下基团之一:单环、并环、桥环或螺环,所述的含氮杂环、单环、并环、桥环或螺环任选进一步被0至4个(例如0、1、2、3或4个)Ra取代,环A1中N-H不被取代。In some embodiments of the present invention related to general formula (Ia), (I-aa), (I-aaa), Selected from 4 to 12 membered nitrogen-containing heterocyclic rings, the nitrogen-containing heterocyclic rings are selected from one of the following saturated or partially saturated groups: monocyclic, parallel rings, bridged rings or spiro rings, and the nitrogen-containing heterocyclic rings , monocyclic ring, parallel ring, bridging ring or spiro ring are optionally further substituted by 0 to 4 (for example 0, 1, 2, 3 or 4) R a , and NH in ring A is not substituted.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,选自未被取代的或者取代的如下基团之一: 当被取代时,任选进一步被0至4个(例如0、1、2、3或4个)Ra取代基所取代,环A1中N-H不被取代。In some embodiments of the present invention related to general formula (Ia), (I-aa), (I-aaa), One of the following unsubstituted or substituted groups: When substituted, optionally further substituted by 0 to 4 (eg 0, 1 , 2, 3 or 4) R substituents, NH in ring A is unsubstituted.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,选自未被取代的或者取代的当被取代时,任选进一步被0至4个(例如0、1、2、3或4 个)Ra取代基所取代,环A1中N-H不被取代。In some embodiments of the present invention related to general formula (Ia), (I-aa), (I-aaa), selected from unsubstituted or substituted When substituted, optionally further substituted by 0 to 4 (eg 0, 1 , 2, 3 or 4) R substituents, NH in ring A is unsubstituted.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,选自 In some embodiments of the present invention related to general formula (Ia), (I-aa), (I-aaa), selected from
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,Ra各自独立的选自H、oxo、卤素、氰基、OH、NH2、-NH(C1-6烷基)、-N(C1-6烷基)2、C1-6烷基、C1-6烷氧基、3至8元环烷基、4至8元杂环基、-O-3至8元环烷基、-O-4至8元杂环烷基,所述的烷基、烷氧基、环烷基、杂环基任选进一步被0至4个(例如0、1、2、3或4个)选自H、卤素、OH、氰基、CF3、 COOH、NH2、C1-4烷基或C1-4烷氧基的取代基所取代,所述的杂环基含有1至4个选自O、S、 N的杂原子。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), R a each independently selected from H, oxo, halogen, cyano, OH, NH 2 , -NH( C 1-6 alkyl), -N(C 1-6 alkyl) 2 , C 1-6 alkyl, C 1-6 alkoxy, 3 to 8 membered cycloalkyl, 4 to 8 membered heterocyclic group , -O-3 to 8-membered cycloalkyl, -O-4 to 8-membered heterocycloalkyl, said alkyl, alkoxy, cycloalkyl, and heterocyclyl are optionally further replaced by 0 to 4 ( For example, 0, 1, 2, 3 or 4) are substituted by substituents selected from H, halogen, OH, cyano, CF 3 , COOH, NH 2 , C 1-4 alkyl or C 1-4 alkoxy , the heterocyclic group contains 1 to 4 heteroatoms selected from O, S, N.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,Ra各自独立的选自H、oxo、卤素、氰基、C1-4烷基、C1-4烷氧基、3至6元环烷基、4至6元杂环基、-O-3至6元环烷基、-O-4 至6元杂环烷基,所述的烷基、烷氧基、环烷基、杂环基任选进一步被0至4个(例如0、1、 2、3或4个)选自H、卤素、OH、氰基、CF3、COOH、NH2、C1-4烷基或C1-4烷氧基的取代基所取代,所述的杂环基含有1至4个选自O、S、N的杂原子。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), R a each independently selected from H, oxo, halogen, cyano, C 1-4 alkyl, C 1-4 alkoxy, 3 to 6-membered cycloalkyl, 4 to 6-membered heterocyclyl, -O-3 to 6-membered cycloalkyl, -O-4 to 6-membered heterocycloalkyl, said alkane radical, alkoxy, cycloalkyl, heterocyclyl are optionally further selected from 0 to 4 (for example 0, 1, 2, 3 or 4) selected from H, halogen, OH, cyano, CF 3 , COOH, Substituted by NH 2 , C 1-4 alkyl or C 1-4 alkoxy, the heterocyclic group contains 1 to 4 heteroatoms selected from O, S, and N.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,Ra各自独立的选自H、oxo、F、 Cl、Br、I、氰基、OH、NH2、甲基、乙基、丙基、异丙基、甲氧基、乙氧基、丙氧基、异丙氧基、环丙基、环丁基、环戊基、环己基、-O-环丙基、-O-环丁基、-O-环戊基、-O-环己基,所述的甲基、乙基、丙基、甲氧基、乙氧基、丙氧基、异丙氧基任选进一步被0至4个(例如 0、1、2、3或4个)选自H、F、Cl、Br、I、OH、氰基、CF3、COOH、NH2、甲基、乙基、丙基、甲氧基、乙氧基、丙氧基或异丙氧基的取代基所取代。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), R a each independently selected from H, oxo, F, Cl, Br, I, cyano, OH, NH 2 , methyl, ethyl, propyl, isopropyl, methoxy, ethoxy, propoxy, isopropoxy, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, -O -Cyclopropyl, -O-cyclobutyl, -O-cyclopentyl, -O-cyclohexyl, the methyl, ethyl, propyl, methoxy, ethoxy, propoxy, iso Propoxy is optionally further replaced by 0 to 4 (for example 0, 1, 2, 3 or 4) selected from H, F, Cl, Br, I, OH, cyano, CF 3 , COOH, NH 2 , formazan Substituents of radical, ethyl, propyl, methoxy, ethoxy, propoxy or isopropoxy.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,Ra各自独立的选自H、oxo、F、 Cl、Br、I、氰基、OH、NH2、甲基、乙基、甲氧基、乙氧基、丙氧基、异丙氧基、环丙基、 -O-环丙基,所述的甲基、乙基、丙基、甲氧基、乙氧基、丙氧基、丙氧基、异丙氧基、环丙基任选进一步被0至4个(例如0、1、2、3或4个)选自H、F、Cl、Br、I、OH、氰基、CF3、 COOH、NH2、甲基、乙基、丙基、甲氧基、乙氧基或丙氧基所取代。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), R a each independently selected from H, oxo, F, Cl, Br, I, cyano, OH, NH 2 , methyl, ethyl, methoxy, ethoxy, propoxy, isopropoxy, cyclopropyl, -O-cyclopropyl, the methyl, ethyl, propyl, methyl Oxygen, ethoxy, propoxy, propoxy, isopropoxy, cyclopropyl are optionally further selected from 0 to 4 (for example 0, 1, 2, 3 or 4) selected from H, F, Cl, Br, I, OH, cyano, CF 3 , COOH, NH 2 , methyl, ethyl, propyl, methoxy, ethoxy or propoxy.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,Ra各自独立的选自H。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), R a is each independently selected from H.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,两个Ra与其相连接的原子共同形成3至12元碳环基或4至12元杂环基,所述的碳环基或杂环基任选进一步被0至4个(例如0、1、2、3或4个)选自H、卤素、OH、氰基、CF3、COOH、NH2、-NH(C1-6烷基)、-N(C1-6烷基)2、C1-4烷基或C1-4烷氧基的取代基所取代,所述的杂环基含有1至4个(例如1、2、3 或4个)选自O、S、N的杂原子。The present invention relates to some embodiments of the general formula (Ia), (I-aa), (I-aaa), two R a together form a 3 to 12 membered carbocyclyl or a 4 to 12 membered hetero Cyclic group, the carbocyclyl or heterocyclic group is optionally further selected from 0 to 4 (eg 0, 1, 2, 3 or 4) selected from H, halogen, OH, cyano, CF 3 , COOH, Substituents of NH 2 , -NH(C 1-6 alkyl), -N(C 1-6 alkyl) 2 , C 1-4 alkyl or C 1-4 alkoxy, the hetero Cyclic groups contain 1 to 4 (eg 1, 2, 3 or 4) heteroatoms selected from O, S, N.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,两个Ra与其相连接的原子共同形成3至6元碳环基或4至6元杂环基,所述的碳环基或杂环基任选进一步被0至4个选自H、卤素、OH、氰基、CF3、COOH、NH2、C1-4烷基或C1-4烷氧基的取代基所取代,所述的杂环基含有1至4个(例如1、2、3或4个)选自O、S、N的杂原子。The present invention relates to some embodiments of the general formula (Ia), (I-aa), (I-aaa), two R a and the atoms to which they are connected together form a 3 to 6 membered carbocyclyl or a 4 to 6 membered heterocyclyl Cyclic group, the carbocyclyl or heterocyclic group is optionally further replaced by 0 to 4 selected from H, halogen, OH, cyano, CF 3 , COOH, NH 2 , C 1-4 alkyl or C 1- 4 alkoxy substituents, the heterocyclic group contains 1 to 4 (eg 1, 2, 3 or 4) heteroatoms selected from O, S, N.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,环B1选自3至8元碳环基、5至 7元杂环基、5至6元杂芳基或苯基,所述碳环基、杂环基、杂芳基或苯基任选进一步被0至 7个(例如0、1、2、3、4、5、6、7个)Rb取代,所述的杂环基或杂芳基含有1至4个选自 O、S、N的杂原子。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), ring B 1 is selected from 3 to 8 membered carbocyclyl, 5 to 7 membered heterocyclyl, 5 to 6 Member heteroaryl or phenyl, said carbocyclyl, heterocyclyl, heteroaryl or phenyl is optionally further replaced by 0 to 7 (such as 0, 1, 2, 3, 4, 5, 6, 7 ) R b is substituted, and the heterocyclic group or heteroaryl group contains 1 to 4 heteroatoms selected from O, S, and N.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,选自 其左边与L2连接,上方与环A1连接。In some embodiments of the present invention related to general formula (Ia), (I-aa), (I-aaa), selected from It is connected to L 2 on the left and ring A 1 above.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,选自 其左边与L2连接,上方与环A1连接。In some embodiments of the present invention related to general formula (Ia), (I-aa), (I-aaa), selected from It is connected to L 2 on the left and ring A 1 above.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,选自其左边与L2连接,上方与环A1连接。In some embodiments of the present invention related to general formula (Ia), (I-aa), (I-aaa), selected from It is connected to L 2 on the left and ring A 1 above.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,选自其左边与L2连接,上方与环A1连接。In some embodiments of the present invention related to general formula (Ia), (I-aa), (I-aaa), selected from It is connected to L 2 on the left and ring A 1 above.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,选自其左边与L2连接,上方与环A1连接;In some embodiments of the present invention related to general formula (Ia), (I-aa), (I-aaa), selected from Its left side is connected with L 2 , and its upper part is connected with ring A 1 ;
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,选自其左边与L2连接,上方与环A1连接。In some embodiments of the present invention related to general formula (Ia), (I-aa), (I-aaa), selected from It is connected to L 2 on the left and ring A 1 above.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,选自其左边与L2连接,上方与环A1连接。In some embodiments of the present invention related to general formula (Ia), (I-aa), (I-aaa), selected from It is connected to L 2 on the left and ring A 1 above.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,选自其左边与L2连接,上方与环A1连接。本发明涉及通式(I-a)、(I-aa)、(I-aaa)、(I-aaaa)、(I-c)的一些实施方案中,Rb各自独立的选自H、oxo、卤素、氰基、OH、NH2、-NH(C1-6烷基)、-N(C1-6烷基)2、C1-6烷基、C1-6烷硫基、C2-6烯基、C2-6炔基、C1-6烷氧基、3至8元环烷基、4至8元杂环基、-O-3至8元环烷基、-O-4至8元杂环烷基,所述的烷基、烯基、炔基、烷氧基、烷硫基、环烷基、杂环基或杂环烷基任选进一步被0至4个(例如0、1、2、3或4个)选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、C1-4烷基、C1-4烷氧基、4-7元杂环基或C3-6环烷基的取代基所取代,所述的杂环基或杂环烷基含有1至4个选自O、S、N的杂原子。In some embodiments of the present invention related to general formula (Ia), (I-aa), (I-aaa), selected from It is connected to L 2 on the left and ring A 1 above. The present invention relates to some embodiments of the general formula (Ia), (I-aa), (I-aaa), (I-aaaa), (Ic), R b each independently selected from H, oxo, halogen, cyanide radical, OH, NH 2 , -NH(C 1-6 alkyl), -N(C 1-6 alkyl) 2 , C 1-6 alkyl, C 1-6 alkylthio, C 2-6 alkene Base, C 2-6 alkynyl, C 1-6 alkoxy, 3 to 8 membered cycloalkyl, 4 to 8 membered heterocyclyl, -O-3 to 8 membered cycloalkyl, -O-4 to 8 membered heterocycloalkyl, said alkyl, alkenyl, alkynyl, alkoxy, alkylthio, cycloalkyl, heterocyclyl or heterocycloalkyl are optionally further replaced by 0 to 4 (such as 0, 1, 2, 3 or 4) selected from H, halogen, OH, cyano, oxo, CF 3 , COOH, NH 2 , C 1-4 alkyl, C 1-4 alkoxy, 4-7 membered hetero Substituents of cyclic group or C 3-6 cycloalkyl group, said heterocyclic group or heterocycloalkyl group contain 1 to 4 heteroatoms selected from O, S, N.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)、(I-aaaa)、(I-c)的一些实施方案中,两个Rb与其相连接的原子共同形成3至12元碳环基或4至12元杂环基,所述的碳环基或杂环基任选进一步被0至4个(例如0、1、2、3或4个)选自H、卤素、OH、氰基、CF3、COOH、NH2、-NH(C1-6烷基)、-N(C1-6烷基)2、C1-4烷基或C1-4烷氧基的取代基所取代,所述的杂环基含有1至4个(例如1、2、3或4个)选自O、S、N的杂原子。The present invention relates to some embodiments of the general formula (Ia), (I-aa), (I-aaa), (I-aaaa), (Ic), two R b together form 3 to 12 A membered carbocyclic group or a 4- to 12-membered heterocyclic group, said carbocyclic group or heterocyclic group is optionally further selected from 0 to 4 (for example 0, 1, 2, 3 or 4) selected from H, halogen, OH, cyano, CF 3 , COOH, NH 2 , -NH(C 1-6 alkyl), -N(C 1-6 alkyl) 2 , C 1-4 alkyl or C 1-4 alkoxy The substituent is substituted, and the heterocyclic group contains 1 to 4 (eg 1, 2, 3 or 4) heteroatoms selected from O, S, N.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)、(I-aaaa)、(I-c)的一些实施方案中,Rb各自独立的选自H、oxo、卤素、氰基、C1-4烷基、C1-4烷硫基、C2-4烯基、C2-4炔基、C1-4烷氧基、3至6 元环烷基、4至6元杂环基、-O-3至6元环烷基、-O-4至6元杂环烷基,所述的烷基、烯基、炔基、烷氧基、烷硫基、环烷基、杂环基或杂环烷基任选进一步被0至4个(例如0、1、2、 3或4个)选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、C1-4烷基或C1-4烷氧基的取代基所取代。The present invention relates to some embodiments of the general formula (Ia), (I-aa), (I-aaa), (I-aaaa), (Ic), R b each independently selected from H, oxo, halogen, cyanide C 1-4 alkyl, C 1-4 alkylthio, C 2-4 alkenyl, C 2-4 alkynyl, C 1-4 alkoxy, 3 to 6 membered cycloalkyl, 4 to 6 Heterocyclyl, -O-3 to 6-membered cycloalkyl, -O-4 to 6-membered heterocycloalkyl, the alkyl, alkenyl, alkynyl, alkoxy, alkylthio, cycloalkane The group, heterocyclyl or heterocycloalkyl is optionally further selected from 0 to 4 (for example 0, 1, 2, 3 or 4) selected from H, halogen, OH, cyano, oxo, CF 3 , COOH, NH 2. Substituents of C 1-4 alkyl or C 1-4 alkoxy.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)、(I-aaaa)、(I-c)的一些实施方案中,两个Rb与其相连接的原子共同形成3至6元碳环基或4至6元杂环基,所述的碳环基或杂环基任选进一步被0 至4个(例如0、1、2、3或4个)选自H、卤素、OH、氰基、CF3、COOH、NH2、C1-4烷基或C1-4烷氧基的取代基所取代,所述的杂环基含有1至4个(例如1、2、3或4个)选自O、 S、N的杂原子,所述的杂环基或杂环烷基含有1至4个选自O、S、N的杂原子。The present invention relates to some embodiments of the general formula (Ia), (I-aa), (I-aaa), (I-aaaa), (Ic), two R b together form 3 to 6 A membered carbocyclyl or a 4 to 6 membered heterocyclic group, said carbocyclyl or heterocyclic group is optionally further selected from 0 to 4 (such as 0, 1, 2, 3 or 4) selected from H, halogen, OH, cyano, CF 3 , COOH, NH 2 , C 1-4 alkyl or C 1-4 alkoxy substituents, the heterocyclic group contains 1 to 4 (such as 1, 2, 3 or 4) heteroatoms selected from O, S, and N, and the heterocyclic group or heterocycloalkyl group contains 1 to 4 heteroatoms selected from O, S, and N.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)、(I-aaaa)、(I-c)的一些实施方案中,Rb各自独立的选自H、oxo、F、Cl、Br、I、氰基、OH、NH2、甲基、乙基、丙基、异丙基、乙烯基、丙烯基、乙炔基、丙炔基、甲氧基、乙氧基、丙氧基、异丙氧基、环丙基、环丁基、环戊基、环己基、-O-环丙基、-O-环丁基、-O-环戊基、-O-环己基,所述的甲基、乙基、丙基、乙烯基、丙烯基、乙炔基、丙炔基、甲氧基、乙氧基、丙氧基、异丙氧基任选进一步被0至4个(例如0、 1、2、3或4个)选自H、F、Cl、Br、I、OH、氰基、oxo、CF3、COOH、NH2、甲基、乙基、丙基、甲氧基、乙氧基、丙氧基或异丙氧基的取代基所取代。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), (I-aaaa), (Ic), R b each independently selected from H, oxo, F, Cl , Br, I, cyano, OH, NH 2 , methyl, ethyl, propyl, isopropyl, vinyl, propenyl, ethynyl, propynyl, methoxy, ethoxy, propoxy , isopropoxy, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, -O-cyclopropyl, -O-cyclobutyl, -O-cyclopentyl, -O-cyclohexyl, the The methyl, ethyl, propyl, vinyl, propenyl, ethynyl, propynyl, methoxy, ethoxy, propoxy, isopropoxy groups are optionally further replaced by 0 to 4 (for example 0 , 1, 2, 3 or 4) selected from H, F, Cl, Br, I, OH, cyano, oxo, CF 3 , COOH, NH 2 , methyl, ethyl, propyl, methoxy, Substituents of ethoxy, propoxy or isopropoxy.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)、(I-aaaa)、(I-c)的一些实施方案中,Rb各自独立的选自H、oxo、F、Cl、Br、I、氰基、OH、NH2、甲基、乙基、甲氧基、乙氧基、丙氧基、异丙氧基、环丙基、-O-环丙基、乙烯基、乙炔基,所述的甲基、乙基、丙基、甲氧基、乙氧基、丙氧基、丙氧基、异丙氧基、乙烯基、乙炔基、环丙基任选进一步被0至4个(例如0、1、2、 3或4个)选自H、F、Cl、Br、I、OH、氰基、oxo、CF3、COOH、NH2、甲基、乙基、丙基、甲氧基、乙氧基或丙氧基所取代。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), (I-aaaa), (Ic), R b each independently selected from H, oxo, F, Cl , Br, I, cyano, OH, NH 2 , methyl, ethyl, methoxy, ethoxy, propoxy, isopropoxy, cyclopropyl, -O-cyclopropyl, vinyl, Ethynyl, the methyl, ethyl, propyl, methoxy, ethoxy, propoxy, propoxy, isopropoxy, vinyl, ethynyl, cyclopropyl are optionally further replaced by 0 to 4 (eg 0, 1, 2, 3 or 4) selected from H, F, Cl, Br, I, OH, cyano, oxo, CF 3 , COOH, NH 2 , methyl, ethyl, propane Substituted by radical, methoxy, ethoxy or propoxy.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)、(I-aaaa)、(I-c)的一些实施方案中,Rb选自H、F、 Cl、氰基、OH、NH2、甲基、乙基、甲氧基、乙氧基、环丙基、-O-环丙基。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), (I-aaaa), (Ic), R b is selected from H, F, Cl, cyano, OH , NH 2 , methyl, ethyl, methoxy, ethoxy, cyclopropyl, -O-cyclopropyl.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,L1选自-O-和-O-CH2-。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), L 1 is selected from -O- and -O-CH 2 -.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,R1选自卤素、OH、氰基、COOH、 NH2、C1-6烷基、C2-6烯基、C2-6炔基、C1-6烷氧基、NH2、-(CH2)q-C(=O)C1-6烷基、-(CH2)q-C(=O)-3 至12元杂环、-(CH2)q-C(=O)-C3-10碳环、-(CH2)q-3至12元杂环或-(CH2)q-C3-10碳环,所述的 CH2、烷基、烯基、炔基、烷氧基、碳环或杂环任选进一步被0至4个(例如0、1、2、3或4 个)R1a取代,所述的杂环含有1至4个(例如1、2、3或4个)选自O、S、N的杂原子。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), R 1 is selected from halogen, OH, cyano, COOH, NH 2 , C 1-6 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, C 1-6 alkoxy, NH 2 , -(CH 2 ) q -C(=O)C 1-6 alkyl, -(CH 2 ) q - C(=O)-3 to 12-membered heterocycle, -(CH 2 ) q -C(=O)-C 3-10 carbocycle, -(CH 2 ) q -3 to 12-membered heterocycle or -(CH 2 ) q -C 3-10 carbocycle, said CH 2 , alkyl, alkenyl, alkynyl, alkoxy, carbocycle or heterocycle is optionally further replaced by 0 to 4 (for example 0, 1, 2 , 3 or 4) R 1a is substituted, and the heterocyclic ring contains 1 to 4 (eg 1, 2, 3 or 4) heteroatoms selected from O, S, N.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,-L1-R1选自 In some embodiments of the present invention related to general formula (Ia), (I-aa), (I-aaa), -L 1 -R 1 is selected from
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,R1a各自独立地选自H、卤素、oxo、 OH、氰基、CF3、COOH、NH2、-NH(C1-6烷基)、-N(C1-6烷基)2、C1-6烷基、C2-6烯基、C2-6炔基、-C1-6亚烷基-OH、-(CH2)q-C3-10碳环、-(CH2)q-3至12元杂环、-O-C3-10碳环、-O-3至12 元杂环、C1-6烷氧基、-N(C1-6烷基)C(=O)-3至12元杂环的取代基所取代,所述的烷基、CH2、烷氧基、烯基、炔基、亚烷基、碳环或杂环任选进一步被0至4个(例如0、1、2、3或4个) 选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、C1-6烷基、C1-6烷氧基、C3-6环烷基、3 至6元杂环烷基的取代基所取代,所述的杂环或杂环烷基含有1至4个(例如1、2、3或4 个)选自O、S、N的杂原子。The present invention relates to some embodiments of the general formula (Ia), (I-aa), (I-aaa), each R 1a is independently selected from H, halogen, oxo, OH, cyano, CF 3 , COOH, NH 2. -NH(C 1-6 alkyl), -N(C 1-6 alkyl) 2 , C 1-6 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, -C 1- 6 alkylene-OH, -(CH 2 ) q -C 3-10 carbocycle, -(CH 2 ) q -3 to 12 membered heterocycle, -OC 3-10 carbocycle, -O-3 to 12 membered Heterocyclic, C 1-6 alkoxy, -N(C 1-6 alkyl)C(=O)-3 to 12-membered heterocyclic substituents, the alkyl, CH 2 , alkoxy Base, alkenyl, alkynyl, alkylene, carbocycle or heterocycle are optionally further selected from 0 to 4 (for example 0, 1, 2, 3 or 4) selected from H, halogen, OH, cyano, oxo , CF 3 , COOH, NH 2 , C 1-6 alkyl, C 1-6 alkoxy, C 3-6 cycloalkyl, 3 to 6 membered heterocycloalkyl, the hetero A ring or heterocycloalkyl group contains 1 to 4 (eg 1 , 2, 3 or 4) heteroatoms selected from O, S, N.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,R1a各自独立地选自H、卤素、oxo、 OH、氰基、CF3、COOH、NH2、-NH(C1-4烷基)、-N(C1-4烷基)2、C1-4烷基、C2-4烯基、C2-4炔基、-C1-4亚烷基-OH、-(CH2)q-C3-8碳环、-(CH2)q-3至8元杂环、-O-C3-8碳环、-O-3至8元杂环、C1-4烷氧基、-N(C1-4烷基)C(=O)-3至8元杂环的取代基所取代,所述的烷基、CH2、烷氧基、烯基、炔基、亚烷基、碳环或杂环任选进一步被0至4个(例如0、1、2、3或4个)选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、C1-6烷基、C1-6烷氧基、C3-6环烷基、3 至6元杂环烷基的取代基所取代,所述的杂环或杂环烷基含有1至4个(例如1、2、3或4 个)选自O、S、N的杂原子。The present invention relates to some embodiments of the general formula (Ia), (I-aa), (I-aaa), each R 1a is independently selected from H, halogen, oxo, OH, cyano, CF 3 , COOH, NH 2. -NH(C 1-4 alkyl), -N(C 1-4 alkyl) 2 , C 1-4 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, -C 1- 4 alkylene-OH, -(CH 2 ) q -C 3-8 carbocycle, -(CH 2 ) q -3 to 8 membered heterocycle, -OC 3-8 carbocycle, -O-3 to 8 membered Heterocyclic, C 1-4 alkoxy, -N(C 1-4 alkyl)C(=O)-3 to 8-membered heterocyclic substituents, the alkyl, CH 2 , alkoxy Base, alkenyl, alkynyl, alkylene, carbocycle or heterocycle are optionally further selected from 0 to 4 (for example 0, 1, 2, 3 or 4) selected from H, halogen, OH, cyano, oxo , CF 3 , COOH, NH 2 , C 1-6 alkyl, C 1-6 alkoxy, C 3-6 cycloalkyl, 3 to 6 membered heterocycloalkyl, the hetero A ring or heterocycloalkyl group contains 1 to 4 (eg 1 , 2, 3 or 4) heteroatoms selected from O, S, N.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,R1a各自独立地选自H、F、Cl、Br、I、oxo、OH、氰基、CF3、COOH、NH2、甲基、乙基、丙基、乙烯基、丙烯基、乙炔基、丙炔基、甲氧基、乙氧基、丙氧基、异丙氧基、环丙基、环丁基、环戊基、环己基、-O-环丙基、-O-环丁基、-O-环戊基、-O-环己基,所述的甲基、乙基、丙基、乙烯基、丙烯基、乙炔基、丙炔基、甲氧基、乙氧基、丙氧基、异丙氧基任选进一步被0至4个(例如0、1、2、3 或4个)选自H、F、Cl、Br、I、OH、氰基、oxo、CF3、COOH、NH2、甲基、乙基、丙基、甲氧基、乙氧基、丙氧基或异丙氧基的取代基所取代。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), R 1a each independently selected from H, F, Cl, Br, I, oxo, OH, cyano, CF 3 , COOH, NH 2 , methyl, ethyl, propyl, vinyl, propenyl, ethynyl, propynyl, methoxy, ethoxy, propoxy, isopropoxy, cyclopropyl , cyclobutyl, cyclopentyl, cyclohexyl, -O-cyclopropyl, -O-cyclobutyl, -O-cyclopentyl, -O-cyclohexyl, the methyl, ethyl, propyl , vinyl, propenyl, ethynyl, propynyl, methoxy, ethoxy, propoxy, isopropoxy are optionally further replaced by 0 to 4 (for example 0, 1, 2, 3 or 4 ) is selected from H, F, Cl, Br, I, OH, cyano, oxo, CF 3 , COOH, NH 2 , methyl, ethyl, propyl, methoxy, ethoxy, propoxy or iso Propoxy substituents are substituted.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,本发明涉及通式(I-a)、(I-aa)、(I-aaa) 的一些实施方案中,R1a选自F。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), the present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa) Among them, R 1a is selected from F.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,本发明涉及通式(I-a)、(I-aa)、(I-aaa) 的一些实施方案中,-L1-R1选自 The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), the present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa) Among them, -L 1 -R 1 is selected from
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,L2选自键、O或-C(=O)-。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), L2 is selected from a bond, O or -C (=O)-.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,L2选自键或-C(=O)-。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), L2 is selected from a bond or -C (=O)-.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,L2选自键。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I - aaa), L2 is selected from a bond.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)、(I-aaaa)、(I-b)、(I-c)、(I-d)的一些实施方案中,R2选自C1-6烷基、C3-10碳环基、3至10元杂环基、C6-10芳基或5至10元杂芳基,所述的烷基、碳环基、杂环基、芳基或杂芳基任选进一步被0至4个(例如0、1、2、3或4个)R2a取代,所述的杂环基或杂芳基含有1至4个(例如1、2、3或4个)选自O、S、N的杂原子。In some embodiments of the present invention related to general formula (Ia), (I-aa), (I-aaa), (I-aaaa), (Ib), (Ic), (Id), R 2 is selected from C 1 -6 alkyl, C 3-10 carbocyclyl, 3 to 10 membered heterocyclic group, C 6-10 aryl or 5 to 10 membered heteroaryl, the alkyl, carbocyclyl, heterocyclic group, Aryl or heteroaryl is optionally further substituted by 0 to 4 (for example 0, 1, 2, 3 or 4) R 2a , and said heterocyclyl or heteroaryl contains 1 to 4 (for example 1, 2, 3 or 4) heteroatoms selected from O, S, N.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)、(I-b)、(I-c)、(I-d)的一些实施方案中,R2选自C3-10碳环基、3至10元杂环基、C6-10芳基或5至10元杂芳基,所述的碳环基、杂环基、芳基或杂芳基任选进一步被0至7个(例如0、1、2、3、4、5、6或7个)R2a取代,所述的杂环基或杂芳基含有1至4个(例如1、2、3或4个)选自O、S、N的杂原子。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), (Ib), (Ic), (Id), R 2 is selected from C 3-10 carbocyclyl, 3 to 10 membered heterocyclic group, C 6-10 aryl group or 5 to 10 membered heteroaryl group, said carbocyclic group, heterocyclic group, aryl group or heteroaryl group are optionally further replaced by 0 to 7 (for example 0, 1, 2, 3, 4, 5, 6 or 7) R 2a is substituted, and the heterocyclyl or heteroaryl contains 1 to 4 (such as 1, 2, 3 or 4) selected from O , S, N heteroatoms.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)、(I-b)、(I-c)、(I-d)的一些实施方案中,R2选自取代的或者未被取代的如下基团之一:苯基、萘基、噻吩基、呋喃基、吡咯基、噻唑基、异噻唑基、噁唑基、异噁唑基、咪唑基、吡唑基、吲唑、苯并咪唑基、苯并吡唑基、苯并噻唑基、苯并噻吩基、苯并呋喃基、苯并嘧啶基、苯并吡啶基或吡啶基,当被取代时,任选被0至5个(例如 0、1、2、3、4或5个)R2a取代。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), (Ib), (Ic), (Id), R 2 is selected from substituted or unsubstituted as follows One of the groups: phenyl, naphthyl, thienyl, furyl, pyrrolyl, thiazolyl, isothiazolyl, oxazolyl, isoxazolyl, imidazolyl, pyrazolyl, indazole, benzimidazolyl , benzopyrazolyl, benzothiazolyl, benzothienyl, benzofuryl, benzopyrimidinyl, benzopyridyl or pyridyl, when substituted, optionally replaced by 0 to 5 (eg 0 , 1, 2, 3, 4 or 5) R 2a is substituted.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)、(I-aaaa)、(I-b)、(I-c)、(I-d)的一些实施方案中, R2选自取代的或者未被取代的如下基团之一:苯基、吡啶、苯并噻唑基、苯并噻吩基、苯并呋喃基、苯并吡唑基或萘基,当被取代时,任选被0至5个(例如0、1、2、3、4或5个)R2a取代。In some embodiments of the present invention related to general formula (Ia), (I-aa), (I-aaa), (I-aaaa), (Ib), (Ic), (Id ) , R is selected from substituted Or unsubstituted one of the following groups: phenyl, pyridine, benzothiazolyl, benzothienyl, benzofuryl, benzopyrazolyl or naphthyl, when substituted, optionally by 0 to 5 (eg 0, 1, 2, 3, 4 or 5) R 2a substitutions.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)、(I-aaaa、(I-b)、(I-c)、(I-d)的一些实施方案中,R2选自苯基、苯并吡唑基、苯并呋喃基、吡啶基,所述的苯基被1至4个R2a取代,所述的苯并吡唑基、苯并呋喃基、吡啶基被0至4个R2a取代;In some embodiments of the present invention related to general formula (Ia), (I-aa), (I-aaa), (I - aaaa, (Ib), (Ic), (Id), R is selected from phenyl, Benzopyrazolyl, benzofuryl, pyridyl, the phenyl is substituted by 1 to 4 R 2a , and the benzopyrazolyl, benzofuryl, pyridyl are 0 to 4 R 2a replaces;
本发明涉及通式(I-a)、(I-aa)、(I-aaa)、(I-aaaa、(I-b)、(I-c)、(I-d)的一些实施方案中,R2选自取代的如下基团之一:苯基、吡啶、苯并噻唑基、苯并噻吩基、苯并呋喃基、苯并吡唑基或萘基,当被取代时,被1至5个R2a取代,R2a各自独立的选自H、F、Cl、OH、氰基、NH2、 CF3、OCF3、-SCH3、甲基、乙基、丙基、异丙基、甲氧基、乙氧基、乙炔基、丙炔基、环丙基、环丁基或环戊基,所述甲基、乙基、丙基、异丙基、甲氧基、乙氧基、乙炔基、丙炔基、环丙基、环丁基或环戊基任选进一步被0至4个选自H、F、Cl、OH、甲基、乙基或环丙基的取代基所取代,条件是至少有一个R2a选自环丙基、环丁基或环戊基。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), (I-aaaa, (Ib), (Ic), (Id), R is selected from the following substituted One of the groups: phenyl, pyridine, benzothiazolyl, benzothienyl, benzofuryl, benzopyrazolyl or naphthyl, when substituted, is substituted by 1 to 5 R 2a , R 2a independently selected from H, F, Cl, OH, cyano, NH 2 , CF 3 , OCF 3 , -SCH 3 , methyl, ethyl, propyl, isopropyl, methoxy, ethoxy, Ethynyl, propynyl, cyclopropyl, cyclobutyl or cyclopentyl, the methyl, ethyl, propyl, isopropyl, methoxy, ethoxy, ethynyl, propynyl, cyclo Propyl, cyclobutyl or cyclopentyl is optionally further substituted with 0 to 4 substituents selected from H, F, Cl, OH, methyl, ethyl or cyclopropyl, provided that there is at least one R selected from cyclopropyl, cyclobutyl or cyclopentyl.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)、(I-aaaa)、(I-b)、(I-c)、(I-d)的一些实施方案中,R2a各自独立的选自H、卤素、oxo、OH、氰基、CF3、COOH、NH2、-NH(C1-6烷基)、-N(C1-6烷基)2、C1-6烷基、C1-6烷氧基、C1-6烷硫基、C2-6烯基、C2-6炔基、C3-6环烷基、3至6元杂环烷基、-O-C3-6环烷基、-O-3至6元杂环烷基、C6-10芳基或5至10元杂芳基,所述的烷基、烷氧基、烷硫基、烯基、炔基、环烷基、杂环烷基、芳基、杂芳基任选进一步被0至4个(例如0、 1、2、3或4个)选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、C1-6烷基、C1-6烷氧基、C3-6环烷基、3至6元杂环烷基的取代基所取代,所述的杂环烷基、杂环基或杂芳基含有 1至4个(例如1、2、3或4个)选自O、S、N的杂原子。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), (I-aaaa), (Ib), (Ic), (Id), R 2a each independently selected from from H, halogen, oxo, OH, cyano, CF 3 , COOH, NH 2 , -NH(C 1-6 alkyl), -N(C 1-6 alkyl) 2 , C 1-6 alkyl, C 1-6 alkoxy, C 1-6 alkylthio, C 2-6 alkenyl, C 2-6 alkynyl, C 3-6 cycloalkyl, 3 to 6 membered heterocycloalkyl, -OC 3 -6 cycloalkyl, -O-3 to 6-membered heterocycloalkyl, C 6-10 aryl or 5 to 10-membered heteroaryl, the alkyl, alkoxy, alkylthio, alkenyl, Alkynyl, cycloalkyl, heterocycloalkyl, aryl, heteroaryl are optionally further selected from 0 to 4 (for example 0, 1, 2, 3 or 4) selected from H, halogen, OH, cyano, oxo, CF 3 , COOH, NH 2 , C 1-6 alkyl, C 1-6 alkoxy, C 3-6 cycloalkyl, 3 to 6 membered heterocycloalkyl, said A heterocycloalkyl, heterocyclyl or heteroaryl group contains 1 to 4 (eg 1 , 2, 3 or 4) heteroatoms selected from O, S, N.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)、(I-aaaa)、(I-b)、(I-c)、(I-d)的一些实施方案中,R2a各自独立的选自-NHC(O)C1-6烷基、-NHS(O)2C1-6烷基、-P(O)(C1-6烷基)2,所述的烷基任选进一步被0至4个(例如0、1、2、3或4个)选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、C1-6烷基、C1-6烷氧基、C3-6环烷基、3至6元杂环烷基的取代基所取代,所述的杂环烷基含有1至4个(例如1、2、3或4个)选自O、S、N的杂原子。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), (I-aaaa), (Ib), (Ic), (Id), R 2a each independently selected from From -NHC(O)C 1-6 alkyl, -NHS(O) 2 C 1-6 alkyl, -P(O)(C 1-6 alkyl) 2 , the alkyl is optionally further 0 to 4 (eg 0, 1, 2, 3 or 4) selected from H, halogen, OH, cyano, oxo, CF 3 , COOH, NH 2 , C 1-6 alkyl, C 1-6 alkane Oxygen, C 3-6 cycloalkyl, 3 to 6 membered heterocycloalkyl substituents, the heterocycloalkyl contains 1 to 4 (such as 1, 2, 3 or 4) selected from O, S, N heteroatoms.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)、(I-aaaa)、(I-b)、(I-c)、(I-d)的一些实施方案中,R2a各自独立的选自F、Cl、OH、氰基、NH2、CF3、OCF3、-SCH3、甲基、乙基、丙基、异丙基、甲氧基、乙氧基、乙炔基、丙炔基、环丙基、环丁基、环戊基、丙烯基、-NHC(O)CH3、-NHS(O)2CH3、 -P(O)(CH3)2、环戊烯基、苯基、吡唑基,所述甲基、乙基、丙基、异丙基、甲氧基、乙氧基、乙炔基、丙炔基、环丙基、环丁基、环戊基、丙烯基、-NHC(O)CH3、-NHS(O)2CH3、-P(O)(CH3)2、环戊烯基、苯基、吡唑基任选进一步被0至4个选自H、F、Cl、OH、甲基、乙基或环丙基的取代基所取代。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), (I-aaaa), (Ib), (Ic), (Id), R 2a each independently selected from From F, Cl, OH, cyano, NH 2 , CF 3 , OCF 3 , -SCH 3 , methyl, ethyl, propyl, isopropyl, methoxy, ethoxy, ethynyl, propynyl , cyclopropyl, cyclobutyl, cyclopentyl, propenyl, -NHC(O)CH 3 , -NHS(O) 2 CH 3 , -P(O)(CH 3 ) 2 , cyclopentenyl, benzene Base, pyrazolyl, the methyl, ethyl, propyl, isopropyl, methoxy, ethoxy, ethynyl, propynyl, cyclopropyl, cyclobutyl, cyclopentyl, propenyl , -NHC(O)CH 3 , -NHS(O) 2 CH 3 , -P(O)(CH 3 ) 2 , cyclopentenyl, phenyl, pyrazolyl are optionally further selected from 0 to 4 Substituents of H, F, Cl, OH, methyl, ethyl or cyclopropyl.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)、(I-aaaa)、(I-b)、(I-c)、(I-d)的一些实施方案中,两个R2a与其直接相连的原子共同形成3至12元碳环基或4至12元杂环基,所述的碳环基或杂环基任选进一步被0至4个(例如0、1、2、3或4个)选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、C1-4烷基或C1-4烷氧基的取代基所取代,所述的杂环基含有1至4个(例如1、 2、3或4个)选自O、S、N的杂原子。The present invention relates to some embodiments of the general formula (Ia), (I-aa), (I-aaa), (I-aaaa), (Ib), (Ic), (Id), two R 2a are directly related to The atoms connected together form 3 to 12 membered carbocyclic groups or 4 to 12 membered heterocyclic groups, and the described carbocyclic group or heterocyclic group is optionally further replaced by 0 to 4 (such as 0, 1, 2, 3 or 4 Each) is substituted by a substituent selected from H, halogen, OH, cyano, oxo, CF 3 , COOH, NH 2 , C 1-4 alkyl or C 1-4 alkoxy, and the heterocyclic group contains 1 to 4 (eg 1, 2, 3 or 4) heteroatoms selected from O, S, N.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)、(I-aaaa)、(I-b)、(I-c)、(I-d)的一些实施方案中,R2a各自独立的选自H、卤素、oxo、OH、氰基、CF3、COOH、NH2、-NH(C1-4烷基)、-N(C1-4烷基)2、C1-4烷基、C1-4烷氧基、C1-4烷硫基、C2-4烯基、C2-6炔基、C3-6环烷基、3至6元杂环烷基、-O-C3-6环烷基、-O-3至6元杂环烷基、C6-10芳基或5至10元杂芳基,所述的烷基、烷氧基、烷硫基、烯基、炔基、环烷基、杂环烷基、芳基、杂芳基任选进一步被0至4个(例如0、 1、2、3或4个)选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、C1-4烷基、C1-4烷氧基、C3-6环烷基、3至6元杂环烷基的取代基所取代,所述的杂环烷基、杂环基或杂芳基含有 1至4个(例如1、2、3或4个)选自O、S、N的杂原子。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), (I-aaaa), (Ib), (Ic), (Id), R 2a each independently selected from from H, halogen, oxo, OH, cyano, CF 3 , COOH, NH 2 , -NH(C 1-4 alkyl), -N(C 1-4 alkyl) 2 , C 1-4 alkyl, C 1-4 alkoxy, C 1-4 alkylthio, C 2-4 alkenyl, C 2-6 alkynyl, C 3-6 cycloalkyl, 3 to 6 membered heterocycloalkyl, -OC 3 -6 cycloalkyl, -O-3 to 6-membered heterocycloalkyl, C 6-10 aryl or 5 to 10-membered heteroaryl, the alkyl, alkoxy, alkylthio, alkenyl, Alkynyl, cycloalkyl, heterocycloalkyl, aryl, heteroaryl are optionally further selected from 0 to 4 (for example 0, 1, 2, 3 or 4) selected from H, halogen, OH, cyano, oxo, CF 3 , COOH, NH 2 , C 1-4 alkyl, C 1-4 alkoxy, C 3-6 cycloalkyl, 3-6 membered heterocycloalkyl, said A heterocycloalkyl, heterocyclyl or heteroaryl group contains 1 to 4 (eg 1 , 2, 3 or 4) heteroatoms selected from O, S, N.
本发明涉及通式(I-a)、(I-aa)的一些实施方案中,R2a各自独立的选自-NHC(O)C1-4烷基、 -NHS(O)2C1-4烷基、-P(O)(C1-4烷基)2,所述的烷基任选进一步被0至4个(例如0、1、2、3或4个)选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、C1-6烷基、C1-6烷氧基、C3-6环烷基、3至6元杂环烷基的取代基所取代,所述的杂环烷基含有1至4个(例如1、2、3或 4个)选自O、S、N的杂原子。The present invention relates to some embodiments of general formula (Ia) and (I-aa), R 2a are each independently selected from -NHC(O)C 1-4 alkyl, -NHS(O) 2 C 1-4 alkane group, -P(O)(C 1-4 alkyl) 2 , the alkyl group is optionally further selected from 0 to 4 (eg 0, 1, 2, 3 or 4) selected from H, halogen, OH , cyano, oxo, CF 3 , COOH, NH 2 , C 1-6 alkyl, C 1-6 alkoxy, C 3-6 cycloalkyl, 3-6 membered heterocycloalkyl , the heterocycloalkyl group contains 1 to 4 (eg 1, 2, 3 or 4) heteroatoms selected from O, S, N.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)、(I-aaaa)、(I-b)、(I-c)、(I-d)的一些实施方案中,两个R2a与其相连接的原子共同形成3至6元碳环基或4至6元杂环基,所述的碳环基或杂环基任选进一步被0至4个(例如0、1、2、3或4个)选自H、卤素、OH、氰基、oxo、CF3、COOH、NH2、C1-4烷基或C1-4烷氧基的取代基所取代,所述的杂环基含有1至4个(例如1、 2、3或4个)选自O、S、N的杂原子。The present invention relates to some embodiments of the general formulas (Ia), (I-aa), (I-aaa), (I-aaaa), (Ib), (Ic), (Id), two R 2a corresponding to The atoms connected together form a 3-6 membered carbocyclyl or a 4-6 membered heterocyclyl, and said carbocyclyl or heterocyclyl is optionally further replaced by 0 to 4 (for example 0, 1, 2, 3 or 4 Each) is substituted by a substituent selected from H, halogen, OH, cyano, oxo, CF 3 , COOH, NH 2 , C 1-4 alkyl or C 1-4 alkoxy, and the heterocyclic group contains 1 to 4 (eg 1, 2, 3 or 4) heteroatoms selected from O, S, N.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)、(I-aaaa)、(I-b)、(I-c)、(I-d)的一些实施方案中,R2a各自独立的选自H、F、Cl、Br、I、oxo、OH、NH2、氰基、CF3、-SCH3、甲基、乙基、丙基、异丙基、乙烯基、丙烯基、乙炔基、丙炔基、甲氧基、乙氧基、丙氧基、异丙氧基、环丙基、环丁基、环戊基、环己基、-O-环丙基、-O-环丁基、-O-环戊基、-O-环己基,所述的甲基、乙基、丙基、异丙基、乙烯基、丙烯基、乙炔基、丙炔基、甲氧基、乙氧基、丙氧基、异丙氧基、环丙基、环丁基、环戊基、环己基任选进一步被0至4个(例如0、1、2、3或4个)选自H、 F、Cl、Br、I、OH、氰基、oxo、CF3、COOH、NH2、甲基、乙基、丙基、甲氧基、乙氧基、丙氧基或异丙氧基的取代基所取代。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), (I-aaaa), (Ib), (Ic), (Id), R 2a each independently selected from From H, F, Cl, Br, I, oxo, OH, NH 2 , cyano, CF 3 , -SCH 3 , methyl, ethyl, propyl, isopropyl, vinyl, propenyl, ethynyl, propynyl, methoxy, ethoxy, propoxy, isopropoxy, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, -O-cyclopropyl, -O-cyclobutyl, -O-cyclopentyl, -O-cyclohexyl, the methyl, ethyl, propyl, isopropyl, vinyl, propenyl, ethynyl, propynyl, methoxy, ethoxy, Propoxy, isopropoxy, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl are optionally further selected from 0 to 4 (for example 0, 1, 2, 3 or 4) selected from H, F, Substituents of Cl, Br, I, OH, cyano, oxo, CF 3 , COOH, NH 2 , methyl, ethyl, propyl, methoxy, ethoxy, propoxy or isopropoxy replace.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)、(I-aaaa)、(I-b)、(I-c)、(I-d)的一些实施方案中,R2a各自独立的选自-NHC(O)CH3、-NHS(O)2CH3、-P(O)(CH3)2、环戊烯基、苯基、吡唑基,所述的-CH3、环戊烯基、苯基、吡唑基任选进一步被0至4个(例如0、1、2、3或4个)选自H、F、Cl、Br、I、OH、氰基、oxo、CF3、COOH、NH2、甲基、乙基、丙基、甲氧基、乙氧基、丙氧基或异丙氧基的取代基所取代。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), (I-aaaa), (Ib), (Ic), (Id), R 2a each independently selected from From -NHC(O)CH 3 , -NHS(O) 2 CH 3 , -P(O)(CH 3 ) 2 , cyclopentenyl, phenyl, pyrazolyl, the -CH 3 , cyclopentyl Alkenyl, phenyl, pyrazolyl are optionally further selected from 0 to 4 (for example 0, 1, 2, 3 or 4) selected from H, F, Cl, Br, I, OH, cyano, oxo, CF 3. Substituents of COOH, NH 2 , methyl, ethyl, propyl, methoxy, ethoxy, propoxy or isopropoxy.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)、(I-b)、(I-c)、(I-d)的一些实施方案中,两个R2a与其相连接的原子共同形成3至6元碳环基或4至6元杂环基,所述的碳环基或杂环基任选进一步被0至4个(例如0、1、2、3或4个)选自H、F、Cl、Br、I、OH、氰基、oxo、CF3、COOH、 NH2、甲基、乙基、丙基、甲氧基、乙氧基、丙氧基或异丙氧基的取代基所取代。The present invention relates to some embodiments of the general formulas (Ia), (I-aa), (I-aaa), (Ib), (Ic), (Id), two R 2a and the atoms to which they are connected together form 3 to 6-membered carbocyclyl or 4 to 6-membered heterocyclyl, and said carbocyclyl or heterocyclyl is optionally further selected from 0 to 4 (such as 0, 1, 2, 3 or 4) selected from H, Substitution by F, Cl, Br, I, OH, cyano, oxo, CF 3 , COOH, NH 2 , methyl, ethyl, propyl, methoxy, ethoxy, propoxy or isopropoxy base replaced.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)、(I-aaaa)、(I-b)、(I-c)、(I-d)的一些实施方案中,R2a各自独立的选自H、F、Cl、OH、氰基、NH2、CF3、OCF3、-SCH3、甲基、乙基、丙基、异丙基、甲氧基、乙氧基、乙炔基、丙炔基、环丙基、环戊基或环己基,所述甲基、乙基、丙基、异丙基、甲氧基、乙氧基、乙炔基、丙炔基、环丙基、环戊基或环己基任选进一步被0至4 个选自H、F、Cl、OH、甲基、乙基或环丙基的取代基所取代。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), (I-aaaa), (Ib), (Ic), (Id), R 2a each independently selected from From H, F, Cl, OH, cyano, NH 2 , CF 3 , OCF 3 , -SCH 3 , methyl, ethyl, propyl, isopropyl, methoxy, ethoxy, ethynyl, propane Alkynyl, cyclopropyl, cyclopentyl or cyclohexyl, the methyl, ethyl, propyl, isopropyl, methoxy, ethoxy, ethynyl, propynyl, cyclopropyl, cyclopentyl The radical or cyclohexyl is optionally further substituted with 0 to 4 substituents selected from H, F, Cl, OH, methyl, ethyl or cyclopropyl.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)、(I-aaaa)、(I-b)、(I-c)、(I-d)的一些实施方案中,R2a各自独立的选自丙烯基、-NHC(O)CH3、-NHS(O)2CH3、-P(O)(CH3)2、环戊烯基、苯基、吡唑基,所述-CH3、丙烯基、环戊烯基、苯基、吡唑基任选进一步被0至4个(例如0、1、2、3或4个)选自H、F、Cl、OH、甲基、乙基或环丙基的取代基所取代。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), (I-aaaa), (Ib), (Ic), (Id), R 2a each independently selected from From propenyl, -NHC(O)CH 3 , -NHS(O) 2 CH 3 , -P(O)(CH 3 ) 2 , cyclopentenyl, phenyl, pyrazolyl, the -CH 3 , Propyl, cyclopentenyl, phenyl, pyrazolyl are optionally further selected from 0 to 4 (for example 0, 1, 2, 3 or 4) selected from H, F, Cl, OH, methyl, ethyl Or substituted by a cyclopropyl substituent.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,q各自独立的选自0、1、2、3或 4。The present invention relates to some embodiments of general formula (I-a), (I-aa), (I-aaa), q is each independently selected from 0, 1, 2, 3 or 4.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,n选自0、1、2、3或4。The present invention relates to some embodiments of general formula (I-a), (I-aa), (I-aaa), n is selected from 0, 1, 2, 3 or 4.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)、(I-c)的一些实施方案中,m各自独立地选自0、1、 2或3。The present invention relates to some embodiments of general formula (I-a), (I-aa), (I-aaa), (I-c), m is each independently selected from 0, 1, 2 or 3.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,条件是不为m1选自0、1或2。The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), with the proviso that not for m1 is selected from 0, 1 or 2.
本发明涉及通式(I-a)、(I-aa)、(I-aaa)的一些实施方案中,条件是化合物不为 The present invention relates to some embodiments of general formula (Ia), (I-aa), (I-aaa), with the proviso that the compound is not
本发明涉及一种药物组合物,包括本发明所述的化合物或者其立体异构体、氘代物、N- 氧化物、溶剂化物、前药、代谢产物、药学上可接受的盐或共晶,以及要学上可接受的载体。The present invention relates to a pharmaceutical composition, comprising the compound of the present invention or its stereoisomer, deuterated substance, N-oxide, solvate, prodrug, metabolite, pharmaceutically acceptable salt or co-crystal, And to be academically acceptable carrier.
本发明涉及一种化合物或者其立体异构体、氘代物、N-氧化物、溶剂化物、前药、代谢产物、药学上可接受的盐或共晶在用于制备治疗与KRAS活性或表达量相关疾病的药物中的应用。The present invention relates to a compound or its stereoisomer, deuterium, N-oxide, solvate, prodrug, metabolite, pharmaceutically acceptable salt or co-crystal used in the preparation of therapeutically related KRAS activity or expression level Drug application in related diseases.
本发明涉及一种合物或者其立体异构体、氘代物、N-氧化物、溶剂化物、前药、代谢产物、药学上可接受的盐或共晶在用于制备治疗与KRAS G12D活性或表达量相关疾病的药物中的应用,其中,所述的疾病选自肿瘤。The present invention relates to a compound or its stereoisomer, deuterium, N-oxide, solvate, prodrug, metabolite, pharmaceutically acceptable salt or co-crystal for use in the preparation of a treatment with KRAS G12D activity or Application in medicine for diseases related to expression level, wherein the diseases are selected from tumors.
除非有相反的陈述,在本申请说明书和权利要求书中使用的术语具有下述含义。Unless stated otherwise, the terms used in the specification and claims of this application have the following meanings.
本发明所述基团和化合物中所涉及的碳、氢、氧、硫、氮或F、Cl、Br、I均包括它们的同位素情况,及本发明所述基团和化合物中所涉及的碳、氢、氧、硫或氮任选进一步被一个或多个它们对应的同位素所替代,其中碳的同位素包括12C、13C和14C,氢的同位素包括氕(H)、氘(D,又叫重氢)、氚(T,又叫超重氢),氧的同位素包括16O、17O和18O,硫的同位素包括32S、33S、34S和36S,氮的同位素包括14N和15N,氟的同位素包括17F和19F,氯的同位素包括35Cl和37Cl,溴的同位素包括79Br和81Br。The carbon, hydrogen, oxygen, sulfur, nitrogen or F, Cl, Br, and I involved in the groups and compounds of the present invention include their isotopes, and the carbon involved in the groups and compounds of the present invention , hydrogen, oxygen, sulfur or nitrogen are optionally further replaced by one or more of their corresponding isotopes, wherein the isotopes of carbon include 12 C, 13 C and 14 C, and the isotopes of hydrogen include protium (H), deuterium (D, Also called heavy hydrogen), tritium (T, also called super heavy hydrogen), oxygen isotopes include 16 O, 17 O and 18 O, sulfur isotopes include 32 S, 33 S, 34 S and 36 S, nitrogen isotopes include 14 N and 15 N, the isotopes of fluorine include 17 F and 19 F, the isotopes of chlorine include 35 Cl and 37 Cl, and the isotopes of bromine include 79 Br and 81 Br.
“卤素”是指F、Cl、Br或I。"Halogen" means F, Cl, Br or I.
“卤素取代的”是指F、Cl、Br或I取代,包括但不限于1至10个选自F、Cl、Br或I的取代基所取代,1至6个选自F、Cl、Br或I的取代基所取代,为1至4个选自F、Cl、Br或I 的取代基所取代。“卤素取代的”简称为“卤代”。"Halogen substituted" refers to F, Cl, Br or I substitution, including but not limited to 1 to 10 substituents selected from F, Cl, Br or I, 1 to 6 substituents selected from F, Cl, Br Or substituted by a substituent of I, substituted by 1 to 4 substituents selected from F, Cl, Br or I. "Halo-substituted" is simply referred to as "halo".
“烷基”是指取代的或者未取代的直链或支链饱和脂肪族烃基,包括但不限于1至20个碳原子的烷基、1至8个碳原子的烷基、1至6个碳原子的烷基、1至4个碳原子的烷基。非限制性实施例包括甲基、乙基、正丙基、异丙基、正丁基、仲丁基、新丁基、叔丁基、正戊基、异戊基、新戊基、正己基及其各种支链异构体;本文中出现的烷基,其定义与本定义一致。烷基可以是一价、二价、三价或四价。"Alkyl" refers to a substituted or unsubstituted linear or branched saturated aliphatic hydrocarbon group, including but not limited to an alkyl group of 1 to 20 carbon atoms, an alkyl group of 1 to 8 carbon atoms, an alkyl group of 1 to 6 An alkyl group of carbon atoms, an alkyl group of 1 to 4 carbon atoms. Non-limiting examples include methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, neobutyl, tert-butyl, n-pentyl, isopentyl, neopentyl, n-hexyl And its various branched chain isomers; Alkyl group appearing in this article, its definition is consistent with this definition. Alkyl groups can be monovalent, divalent, trivalent or tetravalent.
“杂烷基”指取代的或者未取代的烷基中的1个或多个(包括但不限于2、3、4、5或6个) 碳原子被杂原子(包括但不限于N、O或S)替换。非限制性实施例包括 -X(CH2)v-X(CH2)v-X(CH2)v-H(v为1至5的整数,X各自独立的选自键或杂原子,杂原子包括但不限于N、O或S,且至少有1个X选自杂原子,且杂原子中的N或S可被氧化成各种氧化态)。杂烷基可以是一价、二价、三价或四价。"Heteroalkyl" refers to a substituted or unsubstituted alkyl group in which one or more (including but not limited to 2, 3, 4, 5 or 6) carbon atoms are replaced by heteroatoms (including but not limited to N, O or S) replace. Non-limiting examples include -X(CH 2 )vX(CH 2 )vX(CH 2 )vH (v is an integer from 1 to 5, each of X is independently selected from a bond or a heteroatom, and the heteroatom includes but is not limited to N , O or S, and at least one X is selected from heteroatoms, and N or S in heteroatoms can be oxidized into various oxidation states). Heteroalkyl groups can be monovalent, divalent, trivalent or tetravalent.
“亚烷基”是指取代的或者未取代的直链和支链的二价饱和烃基,包括-(CH2)v-(v为1至10 的整数),亚烷基实施例包括但不限于亚甲基、亚乙基、亚丙基和亚丁基等。"Alkylene" refers to substituted or unsubstituted linear and branched divalent saturated hydrocarbon groups, including -(CH 2 ) v - (v is an integer from 1 to 10), examples of alkylene include but not Limited to methylene, ethylene, propylene and butylene, etc.
“亚杂烷基”是指取代的或者未取代的亚烷基中的1个或多个(包括但不限于2、3、4、5或 6个)碳原子被杂原子(包括但不限于N、O或S)替换。非限制性实施例包括 -X(CH2)v-X(CH2)v-X(CH2)v-,v为1至5的整数,X各自独立的选自键、N、O或S,且至少有1个X选自N、O或S。"Heteroalkylene" means a substituted or unsubstituted alkylene group in which one or more (including but not limited to 2, 3, 4, 5 or 6) carbon atoms are replaced by heteroatoms (including but not limited to N, O or S) substitutions. Non-limiting examples include -X(CH 2 )vX(CH 2 )vX(CH 2 )v-, v is an integer from 1 to 5, each of X is independently selected from a bond, N, O or S, and at least 1 X is selected from N, O or S.
“环烷基”是指取代的或者未取代的饱和或不饱和非芳香的碳环烃基,通常有3至10个碳原子,非限制性实施例包括环丙基、环丁基、环戊基、环戊烯基、环己烯基、环己基或环庚基等。本文中出现的环烷基,其定义如上所述。环烷基可以是一价、二价、三价或四价。"Cycloalkyl" means a substituted or unsubstituted saturated or unsaturated non-aromatic carbocyclic hydrocarbon group, usually having 3 to 10 carbon atoms, non-limiting examples include cyclopropyl, cyclobutyl, cyclopentyl , cyclopentenyl, cyclohexenyl, cyclohexyl or cycloheptyl, etc. As used herein, cycloalkyl is as defined above. Cycloalkyl groups can be monovalent, divalent, trivalent or tetravalent.
“杂环烷基”是指取代的或者未取代的饱和的含有杂原子的环烃基,包括但不限于3至10 个原子、3至8个原子,包含1至3个选自N、O或S的杂原子,杂环烷基的环中选择性取代的N、S可被氧化成各种氧化态。杂环烷基可以连接在杂原子或者碳原子上,杂环烷基可以连接在芳香环上或者非芳香环上,杂环烷基可以连接有桥环或者螺环,非限制性实施例包括环氧乙基、氮杂环丙基、氧杂环丁基、氮杂环丁基、四氢呋喃基、四氢-2H-吡喃基、二氧戊环基、二氧六环基、吡咯烷基、哌啶基、咪唑烷基、噁唑烷基、噁嗪烷基、吗啉基、六氢嘧啶基、哌嗪基。杂环烷基可以是一价、二价、三价或四价"Heterocycloalkyl" refers to a substituted or unsubstituted saturated heteroatom-containing cyclic hydrocarbon group, including but not limited to 3 to 10 atoms, 3 to 8 atoms, containing 1 to 3 atoms selected from N, O or The heteroatoms of S, the selectively substituted N and S in the ring of heterocycloalkyl can be oxidized into various oxidation states. The heterocycloalkyl group can be connected to a heteroatom or a carbon atom, the heterocycloalkyl group can be connected to an aromatic ring or a non-aromatic ring, and the heterocycloalkyl group can be connected to a bridged ring or a spiro ring. Non-limiting examples include ring Oxyethyl, aziridyl, oxetanyl, azetidinyl, tetrahydrofuryl, tetrahydro-2H-pyranyl, dioxolanyl, dioxanyl, pyrrolidinyl, Piperidinyl, imidazolidinyl, oxazolidinyl, oxazinyl, morpholinyl, hexahydropyrimidinyl, piperazinyl. Heterocycloalkyl can be monovalent, divalent, trivalent or tetravalent
“烯基”是指取代的或者未取代的直链和支链的不饱和烃基,其具有至少1个,通常有1、 2或3个碳碳双键,主链包括但不限于2至10个、2至6个或2至4个碳原子,烯基实施例包括但不限于乙烯基、烯丙基、1-丙烯基、2-丙烯基、1-丁烯基、2-丁烯基、3-丁烯基、1-戊烯基、2-戊烯基、3-戊烯基、4-戊烯基、1-甲基-1-丁烯基、2-甲基-1-丁烯基、2-甲基-3-丁烯基、1-己烯基、2-己烯基、3-己烯基、4-己烯基、5-己烯基、1-甲基-1-戊烯基、2-甲基-1- 戊烯基、1-庚烯基、2-庚烯基、3-庚烯基、4-庚烯基、1-辛烯基、3-辛烯基、1-壬烯基、3- 壬烯基、1-癸烯基、4-癸烯基、1,3-丁二烯、1,3-戊二烯、1,4-戊二烯和1,4-己二烯等;本文中出现的烯基,其定义与本定义一致。烯基可以是一价、二价、三价或四价。"Alkenyl" refers to a substituted or unsubstituted straight chain and branched unsaturated hydrocarbon group, which has at least 1, usually 1, 2 or 3 carbon-carbon double bonds, the main chain includes but is not limited to 2 to 10 1, 2 to 6, or 2 to 4 carbon atoms, alkenyl examples include but are not limited to vinyl, allyl, 1-propenyl, 2-propenyl, 1-butenyl, 2-butenyl , 3-butenyl, 1-pentenyl, 2-pentenyl, 3-pentenyl, 4-pentenyl, 1-methyl-1-butenyl, 2-methyl-1-but Alkenyl, 2-methyl-3-butenyl, 1-hexenyl, 2-hexenyl, 3-hexenyl, 4-hexenyl, 5-hexenyl, 1-methyl-1 -pentenyl, 2-methyl-1-pentenyl, 1-heptenyl, 2-heptenyl, 3-heptenyl, 4-heptenyl, 1-octenyl, 3-octene base, 1-nonenyl, 3-nonenyl, 1-decenyl, 4-decenyl, 1,3-butadiene, 1,3-pentadiene, 1,4-pentadiene and 1,4-Hexadiene, etc.; alkenyl groups appearing in this text have the same definition as this one. Alkenyl groups can be monovalent, divalent, trivalent or tetravalent.
“炔基”是指取代的或者未取代的直链和支链的一价不饱和烃基,其具有至少1个,通常有 1、2或3个碳碳三键,主链包括2至10个碳原子,包括但不限于在主链上有2至6个碳原子,主链上有2至4个碳原子,炔基实施例包括但不限于乙炔基、炔丙基、1-丙炔基、2-丙炔基、 1-丁炔基、2-丁炔基、3-丁炔基、1-戊炔基、2-戊炔基、3-戊炔基、4-戊炔基、1-甲基-1-丁炔基、2-甲基-1-丁炔基、2-甲基-3-丁炔基、1-己炔基、2-己炔基、3-己炔基、4-己炔基、5-己炔基、1-甲基-1-戊炔基、2-甲基-1-戊炔基、1-庚炔基、2-庚炔基、3-庚炔基、4-庚炔基、1-辛炔基、3-辛炔基、1-壬炔基、3-壬炔基、1-癸炔基、4-癸炔基等;炔基可以是一价、二价、三价或四价。"Alkynyl" means a substituted or unsubstituted straight and branched monovalent unsaturated hydrocarbon group having at least 1, usually 1, 2 or 3 carbon-carbon triple bonds, the main chain comprising 2 to 10 Carbon atoms, including but not limited to 2 to 6 carbon atoms in the main chain, 2 to 4 carbon atoms in the main chain, alkynyl examples include but not limited to ethynyl, propargyl, 1-propynyl , 2-propynyl, 1-butynyl, 2-butynyl, 3-butynyl, 1-pentynyl, 2-pentynyl, 3-pentynyl, 4-pentynyl, 1 -Methyl-1-butynyl, 2-methyl-1-butynyl, 2-methyl-3-butynyl, 1-hexynyl, 2-hexynyl, 3-hexynyl, 4-hexynyl, 5-hexynyl, 1-methyl-1-pentynyl, 2-methyl-1-pentynyl, 1-heptynyl, 2-heptynyl, 3-heptynyl Base, 4-heptynyl, 1-octynyl, 3-octynyl, 1-nonynyl, 3-nonynyl, 1-decynyl, 4-decynyl, etc.; alkynyl can be a valence, divalent, trivalent or quadrivalent.
“烷氧基”是指取代的或者未取代的-O-烷基。非限制性实施例包括甲氧基、乙氧基、正丙氧基、异丙氧基、正丁氧基、仲丁氧基、叔丁氧基、正戊氧基、正己氧基、环丙氧基和环丁氧基。"Alkoxy" means a substituted or unsubstituted -O-alkyl group. Non-limiting examples include methoxy, ethoxy, n-propoxy, isopropoxy, n-butoxy, sec-butoxy, tert-butoxy, n-pentoxy, n-hexyloxy, cyclopropyl Oxygen and Cyclobutoxy.
“碳环基”或“碳环”是指取代的或未取代的饱和或不饱和的芳香环或者非芳香环,芳香环或者非芳香环可以是3至8元的单环、4至12元双环或者10至15元三环体系,碳环基可以连接在芳香环上或者非芳香环上,芳香环或者非芳香环任选为单环、桥环或者螺环。非限制性实施例包括环丙烷、环丁烷、环戊烷、环己烷、环庚烷、1-环戊基-1-烯基、1-环戊基-2-烯基、 1-环戊基-3-烯基、环己基、1-环己基-2-烯基、1-环己基-3-烯基、环己烯基、苯环、萘环、 “碳环基”或“碳环”可以是一价、二价、三价或四价。"Carbocyclyl" or "carbocycle" refers to a substituted or unsubstituted saturated or unsaturated aromatic ring or non-aromatic ring, the aromatic ring or non-aromatic ring can be 3 to 8 membered single ring, 4 to 12 membered Bicyclic or 10- to 15-membered tricyclic ring system, the carbocyclic group can be connected to an aromatic ring or a non-aromatic ring, and the aromatic ring or non-aromatic ring is optionally a monocyclic, bridged or spiro ring. Non-limiting examples include cyclopropane, cyclobutane, cyclopentane, cyclohexane, cycloheptane, 1-cyclopentyl-1-enyl, 1-cyclopentyl-2-enyl, 1-cyclo Pentyl-3-enyl, cyclohexyl, 1-cyclohexyl-2-enyl, 1-cyclohexyl-3-enyl, cyclohexenyl, benzene ring, naphthalene ring, A "carbocyclyl" or "carbocycle" can be monovalent, divalent, trivalent or tetravalent.
“杂环基”或“杂环”是指取代的或未取代的饱和或不饱和的芳香环或者非芳香环,芳香环或者非芳香环可以是3至8元的单环、4至12元双环或者10至15元三环体系,且包含1个或多个(包括但不限于2、3、4或5个)个选自N、O或S的杂原子,杂环基的环中选择性取代的N、S可被氧化成各种氧化态。杂环基可以连接在杂原子或者碳原子上,杂环基可以连接在芳香环上或者非芳香环上,杂环基可以连接有桥环或者螺环,非限制性实施例包括环氧乙基、氮杂环丙基、氧杂环丁基、氮杂环丁基、1,3-二氧戊环基、1,4-二氧戊环基、1,3-二氧六环基、氮杂环庚基、吡啶基、呋喃基、噻吩基、吡喃基、N-烷基吡咯基、嘧啶基、吡嗪基、哒嗪基、咪唑基、哌啶基、吗啉基、硫代吗啉基、1,3-二噻基、二氢呋喃基、二氢吡喃基、二噻戊环基、四氢呋喃基、四氢吡咯基、四氢咪唑基、四氢噻唑基、四氢吡喃基、苯并咪唑基、苯并吡啶基、吡咯并吡啶基、苯并二氢呋喃基、吡咯基、吡唑基、噻唑基、噁唑基、吡嗪基、吲唑基、苯并噻吩基、苯并呋喃基、苯并吡咯基、苯并咪唑基、苯并噻唑基、苯并噁唑基、苯并吡啶基、苯并嘧啶基、苯并吡嗪基、哌嗪基、氮杂二环[3.2.1]辛烷基、氮杂二环[5.2.0]壬烷基、氧杂三环[5.3.1.1]十二烷基、氮杂金刚烷基、氧杂螺[3.3]庚烷基、 “杂环基”或“杂环”可以是一价、二价、三价或四价。"Heterocyclic group" or "heterocyclic ring" refers to a substituted or unsubstituted saturated or unsaturated aromatic ring or non-aromatic ring, the aromatic ring or non-aromatic ring can be 3 to 8 membered single ring, 4 to 12 membered A bicyclic ring or a 10- to 15-membered tricyclic ring system, and contains 1 or more (including but not limited to 2, 3, 4 or 5) heteroatoms selected from N, O or S, and the ring of the heterocyclyl group is selected from Sexually substituted N and S can be oxidized into various oxidation states. The heterocyclic group can be connected to a heteroatom or a carbon atom, the heterocyclic group can be connected to an aromatic ring or a non-aromatic ring, and the heterocyclic group can be connected to a bridged ring or a spiro ring. Non-limiting examples include oxirane , aziridyl, oxetanyl, azetidinyl, 1,3-dioxolanyl, 1,4-dioxolanyl, 1,3-dioxanyl, nitrogen Heterocycloheptyl, pyridyl, furyl, thienyl, pyryl, N-alkylpyrrolyl, pyrimidinyl, pyrazinyl, pyridazinyl, imidazolyl, piperidinyl, morpholinyl, thiomorph Linyl, 1,3-dithianyl, dihydrofuranyl, dihydropyranyl, dithiapentanyl, tetrahydrofuranyl, tetrahydropyrrolyl, tetrahydroimidazolyl, tetrahydrothiazolyl, tetrahydropyranyl Base, benzimidazolyl, benzopyridyl, pyrrolopyridyl, benzodihydrofuryl, pyrrolyl, pyrazolyl, thiazolyl, oxazolyl, pyrazinyl, indazolyl, benzothienyl , benzofuryl, benzopyrrolyl, benzimidazolyl, benzothiazolyl, benzoxazolyl, benzopyridyl, benzopyrimidinyl, benzopyrazinyl, piperazinyl, azadi Cyclo[3.2.1]octyl, azabicyclo[5.2.0]nonyl, oxatricyclo[5.3.1.1]dodecyl, azaadamantyl, oxaspiro[3.3]heptanyl alkyl, A "heterocyclyl" or "heterocycle" may be monovalent, divalent, trivalent or tetravalent.
“螺环”或“螺环基”是指取代的或未取代的单环之间共用一个原子(称螺原子)的多环基团,螺环体系中环原子的个数包括但不限于含有5至20个、6至14个、6至12个、6至10个,其中一个或多个环可以含有0个或多个(包括但不限于1、2、3或4)双键,且任选可以含有0 至5个选自N、O或S(=O)n的杂原子。"Spiro ring" or "spirocyclic group" refers to a polycyclic group that shares one atom (called spiro atom) between substituted or unsubstituted monocyclic rings. The number of ring atoms in the spiro ring system includes but is not limited to 5 to 20, 6 to 14, 6 to 12, 6 to 10, wherein one or more rings may contain 0 or more (including but not limited to 1, 2, 3 or 4) double bonds, and any Optional may contain 0 to 5 heteroatoms selected from N, O or S(=O) n .
“螺环”或“螺环基”可以是一价、二价、三价或四价。 A "spirocycle" or "spirocyclyl" can be monovalent, divalent, trivalent or tetravalent.
“并环”或“并环基”是指系统中的每个环与体系中的其他环共享毗邻的一对原子的多环基团,其中一个或多个环可以含有0个或多个(包括但不限于1、2、3或4)双键,且可以是取代的或未取代,并环体系中的各个环可以含0至5个杂原子或含有杂原子的基团(包括但不限于选自N、S(=O)n或O,n为0、1或2)。并环体系中环原子的个数包括但不限于5至20个,5 至14个,5至12个,5至10个。非限定性实例包括: “并环”或“并环基”可以是一价、二价、三价或四价。"Acyl ring" or "annyl ring group" refers to a polycyclic group in which each ring in the system shares an adjacent pair of atoms with other rings in the system, wherein one or more rings may contain 0 or more ( including but not limited to 1, 2, 3 or 4) double bonds, and may be substituted or unsubstituted, and each ring in the ring system may contain from 0 to 5 heteroatoms or groups containing heteroatoms (including but not limited to be selected from N, S(=O) n or O, n being 0, 1 or 2). The number of ring atoms in the double ring system includes but is not limited to 5 to 20, 5 to 14, 5 to 12, and 5 to 10. Non-limiting examples include: "Alkyl" or "alkyl" may be monovalent, divalent, trivalent or tetravalent.
“桥环”或“桥环基”是指取代的或未取代的含有任意两个不直接连接的原子的多环基团,可以含有0个或多个双键,并环体系中的任意环可以含0至5个选自杂原子或含有杂原子的基团(包括但不限于N、S(=O)n或O,其中n为0、1、2)。环原子个数包括但不限于5至20个、5 至14个、5至12个或5至10个。非限定性实例包括 立方烷、金刚烷。“桥环”或“桥环基”可以是一价、二价、三价或四价。"Bridged ring" or "bridged ring group" refers to a substituted or unsubstituted polycyclic group containing any two atoms that are not directly connected, may contain 0 or more double bonds, and any ring in the ring system It may contain 0 to 5 groups selected from heteroatoms or groups containing heteroatoms (including but not limited to N, S(=O)n or O, wherein n is 0, 1, 2). The number of ring atoms includes, but is not limited to, 5 to 20, 5 to 14, 5 to 12 or 5 to 10. Non-limiting examples include Cubane, adamantane. A "bridged ring" or "bridged ring group" may be monovalent, divalent, trivalent or tetravalent.
“碳螺环”、“螺环碳环基”、“螺碳环基”或者“碳螺环基”是指环体系仅有碳原子组成的“螺环”。本文中出现的“碳螺环”、“螺环碳环基”、“螺碳环基”或者“碳螺环基”,其定义与螺环一致。"Carbospiro", "spirocarbocyclyl", "spirocarbocyclyl" or "carbospirocyclyl" refers to a "spirocycle" whose ring system consists only of carbon atoms. The definitions of "carbospirocycle", "spirocyclic carbocyclyl", "spirocarbocyclyl" or "carbospirocyclyl" appearing herein are consistent with spirocycle.
“碳并环”、“并环碳环基”、“并碳环基”或者“碳并环基”是指环体系仅有碳原子组成的“并环”。本文中出现的“碳并环”、“并环碳环基”、“并碳环基”或者“碳并环基”,其定义与并环一致。"Carbocyclic", "paracyclic carbocyclyl", "paracarbocyclyl" or "carbocyclyl" refers to a "carbocyclyl" whose ring system consists only of carbon atoms. The definition of "carbocyclyl", "paracyclic carbocyclyl", "paracarbocyclyl" or "carbocyclyl" used herein is consistent with that of paracyclyl.
“碳桥环”、“桥环碳环基”、“桥碳环基”或者“碳桥环基”是指环体系仅有碳原子组成的“桥环”。本文中出现的“碳桥环”、“桥环碳环基”、“桥碳环基”或者“碳桥环基”,其定义与桥环一致。"Carbobridged ring", "bridged ring carbocyclyl", "bridged carbocyclyl" or "carbobridged cycloyl" refers to a "bridged ring" whose ring system consists only of carbon atoms. The definitions of "carbon bridged ring", "bridged ring carbocyclyl", "bridged carbocyclyl" or "carbobridged ring" appearing in this article are consistent with those of bridged ring.
“杂单环”、“单环杂环基”或“杂单环基”是指单环体系的“杂环基”或“杂环”,本文中出现的杂环基、“单环杂环基”或“杂单环基”,其定义与杂环一致。"Heterocyclic group", "monocyclic heterocyclic group" or "heteromonocyclic group" refers to the "heterocyclic group" or "heterocyclic group" of a monocyclic ring system, and the heterocyclic group, "monocyclic heterocyclic group" appearing herein group" or "heteromonocyclic group", the definition of which is consistent with that of heterocycle.
“杂并环”、“杂并环基”“并环杂环基”或“杂并环基”是指含有杂原子的“并环”。本文中出现的杂并环、“杂并环基”“并环杂环基”或“杂并环基”,其定义与并环一致。"Heterocyclyl", "heterocyclyl", "heterocyclic heterocyclyl" or "heterocyclyl" refers to "heterocycles" that contain heteroatoms. The definition of heterocyclic ring, "heterocyclic group", "heterocyclic heterocyclic group" or "heterocyclic group" used herein is consistent with that of parallel ring.
“杂螺环”、“杂螺环基”、“螺环杂环基”或“杂螺环基”是指含有杂原子的“螺环”。本文中出现的杂螺环、“杂螺环基”、“螺环杂环基”或“杂螺环基”,其定义与螺环一致。"Heterospiro", "heterospirocyclyl", "spiroheterocyclyl" or "heterospirocyclyl" refers to a "spirocycle" that contains heteroatoms. As used herein, heterospirocycle, "heterospirocyclyl", "spiroheterocyclyl" or "heterospirocyclyl" have the same definition as spirocycle.
“杂桥环”、“杂桥环基”、“桥环杂环基”或“杂桥环基”是指含有杂原子的“桥环”。本文中出现的杂桥环、“杂桥环基”、“桥环杂环基”或“杂桥环基”,其定义与桥环一致。"Heterobridged ring", "heterobridged ring group", "bridged ring heterocyclyl" or "heterobridged ring group" refers to a "bridged ring" that contains heteroatoms. The definition of heterobridged ring, "heterobridged ring group", "bridged ring heterocyclyl group" or "heterobridged ring group" used herein is consistent with bridged ring.
“芳基”或“芳环”是指取代的或者未取代的具有单环或稠合环的芳香族烃基,芳香环中环原子个数包括但不限于6至18、6至12或6至10个碳原子。芳基环可以稠合于饱和或不饱和的碳环或杂环上,其中与母体结构连接在一起的环为芳基环,非限制性实施例包含苯环、萘环、“芳基”或“芳环”可以是一价、二价、三价或四价。当为二价、三价或四价时,连接位点位于芳基环上。"Aryl" or "aromatic ring" refers to a substituted or unsubstituted aromatic hydrocarbon group with a single ring or a condensed ring, and the number of ring atoms in the aromatic ring includes but is not limited to 6 to 18, 6 to 12 or 6 to 10 carbon atoms. The aryl ring may be fused to a saturated or unsaturated carbocyclic or heterocyclic ring, wherein the ring bonded to the parent structure is an aryl ring, non-limiting examples include benzene, naphthalene, "Aryl" or "aromatic ring" may be monovalent, divalent, trivalent or tetravalent. When divalent, trivalent or tetravalent, the point of attachment is on the aryl ring.
“杂芳基”或“杂芳环”是指取代或未取代的芳香族烃基,且含有1至5个选杂原子或含有杂原子的基团(包括但不限于N、O或S(=O)n,n为0、1、2),杂芳香环中环原子个数包括但不限于5至15、5至10或5至6个。杂芳基的非限制性实施例包括但不限于吡啶基、呋喃基、噻吩基、吡啶基、吡喃基、N-烷基吡咯基、嘧啶基、吡嗪基、哒嗪基、咪唑基、苯并吡唑、苯并咪唑、苯并吡啶、吡咯并吡啶等。所述杂芳基环可以稠合于饱和或不饱和的碳环或杂环上,其中与母体结构连接在一起的环为杂芳基环,非限制性实施例包含本文中出现的杂芳基,其定义与本定义一致。杂芳基可以是一价、二价、三价或四价。当为二价、三价或四价时,连接位点位于杂芳基环上。"Heteroaryl" or "heteroaromatic ring" refers to a substituted or unsubstituted aromatic hydrocarbon group containing 1 to 5 selected heteroatoms or groups containing heteroatoms (including but not limited to N, O or S (= O) n, n is 0, 1, 2), and the number of ring atoms in the heteroaromatic ring includes but is not limited to 5 to 15, 5 to 10 or 5 to 6. Non-limiting examples of heteroaryl include, but are not limited to, pyridyl, furyl, thienyl, pyridyl, pyranyl, N-alkylpyrrolyl, pyrimidinyl, pyrazinyl, pyridazinyl, imidazolyl, Benzopyrazole, benzimidazole, benzopyridine, pyrrolopyridine, etc. The heteroaryl ring may be fused to a saturated or unsaturated carbocyclic or heterocyclic ring, wherein the ring bonded to the parent structure is a heteroaryl ring, non-limiting examples include Where heteroaryl appears herein, its definition is consistent with this definition. Heteroaryl groups can be monovalent, divalent, trivalent or tetravalent. When divalent, trivalent or tetravalent, the point of attachment is on the heteroaryl ring.
“5元环并5元杂芳环”是指5并5元的稠合杂芳环,2个并环中至少有1个环含有1个以上的杂原子(包括但不限于O、S或N),整个基团具有芳香性,非限制实施例包括了吡咯并吡咯环、吡唑并吡咯环、吡唑并吡唑环、吡咯并呋喃环、吡唑并呋喃环、吡咯并噻吩环、吡唑并噻吩环。"5-membered ring and 5-membered heteroaromatic ring" refers to a 5-membered condensed heteroaromatic ring, at least one of the two rings contains more than one heteroatom (including but not limited to O, S or N), the entire group is aromatic, and non-limiting examples include pyrrolopyrrole ring, pyrazolopyrrole ring, pyrazolopyrazole ring, pyrrolopyrrole ring, pyrazolofuran ring, pyrrolothiophene ring, pyrazolothiophene ring.
“5并6元杂芳环”是指5并6元的稠合杂芳环,2个并环中至少有1个环含有1个以上的杂原子(包括但不限于O、S或N),整个基团具有芳香性,非限制实施例包括了苯并5元杂芳基、6元杂芳环并5元杂芳环。"5 and 6-membered heteroaryl ring" refers to a 5-6 membered fused heteroaryl ring, at least one of the two rings contains more than one heteroatom (including but not limited to O, S or N) , the entire group is aromatic, non-limiting examples include benzo 5-membered heteroaryl, 6-membered heteroaryl and 5-membered heteroaryl.
“取代”或“取代的”是指被1个或多个(包括但不限于2、3、4或5个)取代基所取代,取代基包括但不限于H、F、Cl、Br、I、烷基、环烷基、烷氧基、卤代烷基、硫醇、羟基、硝基、巯基、氨基、氰基、异氰基、芳基、杂芳基、杂环基、桥环基、螺环基、并环基、羟基烷基、=O、羰基、醛、羧酸、甲酸酯、-(CH2)m-C(=O)-Ra、-O-(CH2)m-C(=O)-Ra、-(CH2)m-C(=O)-NRbRc、 -(CH2)mS(=O)nRa、-(CH2)m-烯基-Ra、ORd或-(CH2)m-炔基-Ra(其中m、n为0、1或2)、芳基硫基、硫代羰基、硅烷基或-NRbRc等基团,其中Rb与Rc独立选自包括H、羟基、氨基、羰基、烷基、烷氧基、环烷基、杂环基、芳基、杂芳基、磺酰基、三氟甲磺酰基,作为选择, Rb与Rc可形成五或六元环烷基或杂环基。"Substituted" or "substituted" means substituted by 1 or more (including but not limited to 2, 3, 4 or 5) substituents including but not limited to H, F, Cl, Br, I , Alkyl, cycloalkyl, alkoxy, haloalkyl, thiol, hydroxyl, nitro, mercapto, amino, cyano, isocyano, aryl, heteroaryl, heterocyclyl, bridged ring, spiro Cyclic group, cyclic group, hydroxyalkyl group, =O, carbonyl, aldehyde, carboxylic acid, formate, -(CH 2 ) m -C(=O)-R a , -O-(CH 2 ) m - C(=O)-R a , -(CH 2 ) m -C(=O)-NR b R c , -(CH 2 ) m S(=O) n R a , -(CH 2 ) m -ene -R a , OR d or -(CH 2 ) m -alkynyl-R a (where m, n are 0, 1 or 2), arylthio, thiocarbonyl, silyl or -NR b R c Such groups, wherein R b and R c are independently selected from H, hydroxyl, amino, carbonyl, alkyl, alkoxy, cycloalkyl, heterocyclyl, aryl, heteroaryl, sulfonyl, trifluoromethane Sulfonyl, as an option, R b and R c can form a five- or six-membered cycloalkyl or heterocyclic group.
“含有1至5个选自O、S、N的杂原子”是指含有1、2、3、4或5个选自O、S、N的杂原子。"Containing 1 to 5 heteroatoms selected from O, S, N" means containing 1, 2, 3, 4 or 5 heteroatoms selected from O, S, N.
“0至X个取代基所取代”是指被0、1、2、3…X个取代基所取代,X选自1至10之间的任意整数。如“0至4个取代基所取代”是指被0、1、2、3或4个取代基所取代。如“0至5个取代基所取代”是指被0、1、2、3、4或5个取代基所取代。如“杂桥环任选进一步被0至4个选自H或F的取代基所取代”是指杂桥环任选进一步被0、1、2、3或4个选自H或F的取代基所取代。"Substituted by 0 to X substituents" means substituted by 0, 1, 2, 3...X substituents, X is selected from any integer between 1 and 10. For example, "substituted by 0 to 4 substituents" means substituted by 0, 1, 2, 3 or 4 substituents. For example, "substituted by 0 to 5 substituents" means substituted by 0, 1, 2, 3, 4 or 5 substituents. For example, "the heterobridged ring is optionally further substituted by 0 to 4 substituents selected from H or F" means that the heterobridged ring is optionally further substituted by 0, 1, 2, 3 or 4 substituents selected from H or F base replaced.
X-Y元的环(X选自小于Y大于3的整数,Y选自4至12之间的任意整数)包括了X+1、 X+2、X+3、X+4…Y元的环。环包括了杂环、碳环、芳环、芳基、杂芳基、环烷基、杂单环、杂并环、杂螺环或杂桥环。如“4-7元杂单环”是指4元、5元、6元或7元的杂单环,“5-10元杂并环”是指5元、6元、7元、8元、9元或10元的杂并环。X-Y-membered rings (X is selected from integers less than Y and greater than 3, Y is selected from any integer between 4 and 12) include X+1, X+2, X+3, X+4...Y-membered rings. Rings include heterocycles, carbocycles, aryls, aryls, heteroaryls, cycloalkyls, heteromonocycles, heteroheterocycles, heterospirocycles or heterobridged rings. For example, "4-7 membered heteromonocyclic ring" refers to 4-membered, 5-membered, 6-membered or 7-membered heteromonocyclic ring, and "5-10-membered heterocyclic ring" refers to 5-, 6-, 7-, and 8-membered heterocyclic rings. , 9- or 10-membered heterocyclic rings.
“任选”或“任选地”是指随后所描述的事件或环境可以但不必须发生,该说明包括该事件或环境发生或不发生的场合。如:“任选被F取代的烷基”指烷基可以但不必须被F取代,说明包括烷基被F取代的情形和烷基不被F取代的情形。"Optional" or "optionally" means that the subsequently described event or circumstance can but need not occur, and that the description includes instances where the event or circumstance occurs or does not occur. For example: "Alkyl optionally substituted by F" means that the alkyl group may but not necessarily be substituted by F, and the description includes the case where the alkyl group is substituted by F and the case where the alkyl group is not substituted by F.
“药学上可接受的盐”或者“其药学上可接受的盐”是指本发明化合物保持游离酸或者游离碱的生物有效性和特性,且所述的游离酸通过与无毒的无机碱或者有机碱,所述的游离碱通过与无毒的无机酸或者有机酸反应获得的盐。"Pharmaceutically acceptable salt" or "pharmaceutically acceptable salt thereof" means that the compound of the present invention maintains the biological effectiveness and characteristics of free acid or free base, and the free acid is mixed with a non-toxic inorganic base or Organic base, the salt obtained by reacting the free base with a non-toxic inorganic acid or organic acid.
“药物组合物”是指一种或多种本发明所述化合物、或者其立体异构体、互变异构体、氘代物、溶剂化物、前药、代谢产物、药学上可接受的盐或共晶和其它化学组分形成的混合物,其中,“其它化学组分”是指药学上可接受的载体、赋形剂和/或一种或多种其它治疗剂。"Pharmaceutical composition" refers to one or more compounds of the present invention, or their stereoisomers, tautomers, deuterated products, solvates, prodrugs, metabolites, pharmaceutically acceptable salts or A mixture of co-crystals and other chemical components, wherein "other chemical components" refer to pharmaceutically acceptable carriers, excipients and/or one or more other therapeutic agents.
“载体”是指不会对生物体产生明显刺激且不会消除所给予化合物的生物活性和特性的材料。"Carrier" refers to a material that does not produce significant irritation to an organism and that does not abrogate the biological activity and properties of the administered compound.
“赋形剂”是指加入到药物组合物中以促进化合物给药的惰性物质。非限制性实施例包括碳酸钙、磷酸钙、糖、淀粉、纤维素衍生物(包括微晶纤维素)、明胶、植物油、聚乙二醇类、稀释剂、成粒剂、润滑剂、粘合剂和崩解剂。"Excipient" refers to an inert substance added to a pharmaceutical composition to facilitate administration of the compound. Non-limiting examples include calcium carbonate, calcium phosphate, sugars, starches, cellulose derivatives (including microcrystalline cellulose), gelatin, vegetable oils, polyethylene glycols, diluents, granulating agents, lubricants, binders agents and disintegrants.
“前药”是指可经体内代谢转化为具有生物活性的本发明化合物。本发明的前药通过修饰本发明化合物中的氨基或者羧基来制备,该修饰可以通过常规的操作或者在体内被除去,而得到母体化合物。当本发明的前药被施予哺乳动物个体时,前药被割裂形成游离的氨基或者羧基。"Prodrug" refers to a compound of the present invention that can be transformed into a biologically active compound through in vivo metabolism. The prodrugs of the present invention are prepared by modifying the amino or carboxyl groups of the compounds of the present invention, which modifications can be removed by routine manipulation or in vivo to obtain the parent compound. When the prodrugs of the present invention are administered to a mammalian subject, the prodrugs are cleaved to form free amino or carboxyl groups.
“共晶”是指活性药物成分(API)和共晶形成物(CCF)在氢键或其他非共价键的作用下结合而成的晶体,其中API和CCF的纯态在室温下均为固体,并且各组分间存在固定的化学计量比。共晶是一种多组分晶体,既包含两种中性固体之间形成的二元共晶,也包含中性固体与盐或溶剂化物形成的多元共晶。"Co-crystal" refers to the crystal formed by the combination of active pharmaceutical ingredient (API) and co-crystal former (CCF) under the action of hydrogen bonding or other non-covalent bonds, wherein the pure state of API and CCF are both solid, and there is a fixed stoichiometric ratio between the components. A co-crystal is a multi-component crystal, including both a binary co-crystal formed between two neutral solids and a multi-element co-crystal formed between a neutral solid and a salt or solvate.
“动物”是指包括哺乳动物,例如人、陪伴动物、动物园动物和家畜,优选人、马或者犬。"Animal" is meant to include mammals such as humans, companion animals, zoo animals and domestic animals, preferably humans, horses or dogs.
“立体异构体”是指由分子中原子在空间上排列方式不同所产生的异构体,包括顺反异构体、对映异构体和构象异构体。"Stereoisomer" refers to isomers produced by different arrangements of atoms in a molecule in space, including cis-trans isomers, enantiomers and conformational isomers.
“互变异构体”是指分子中某一原子在两个位置迅速移动而产生的官能团异构体,如酮式- 烯醇式异构和酰胺-亚胺醇式异构等。"Tautomer" refers to a functional group isomer produced by a certain atom in a molecule moving rapidly at two positions, such as keto-enol isomerization and amide-imino alcohol isomerization.
“任选”或“任选地”或“选择性的”或“选择性地”是指随后所述的事件或状况可以但未必发生,该描述包括其中发生该事件或状况的情况及其中未发生的情况。例如,“选择性地被烷基取代的杂环基”是指该烷基可以但未必存在,该描述包括其中杂环基被烷基取代的情况,及其中杂环基未被烷基取代的情况。"Optional" or "optionally" or "optional" or "optionally" means that the subsequently described event or circumstance may, but need not, occur, and that the description includes the circumstances in which the event or circumstance occurs and the circumstances in which it is not what happened. For example, "heterocyclyl optionally substituted with an alkyl group" means that the alkyl group may but need not be present, and the description includes cases where the heterocyclyl group is substituted with an alkyl group, and cases where the heterocyclyl group is not substituted with an alkyl group. Condition.
“IC50”是对指定的生物过程(或该过程中的某个组分比如酶、受体、细胞等)抑制一半时所需的药物或者抑制剂的浓度。" IC50 " is the concentration of drug or inhibitor required to inhibit a given biological process (or some component of the process such as an enzyme, receptor, cell, etc.) by half.
具体实施方式detailed description
以下实施例详细说明本发明的技术方案,但本发明的保护范围包括但是不限于此。The following examples illustrate the technical solution of the present invention in detail, but the protection scope of the present invention includes but is not limited thereto.
化合物的结构是通过核磁共振(NMR)或(和)质谱(MS)来确定的。NMR位移(δ) 以10-6(ppm)的单位给出。NMR的测定是用(Bruker Avance III 400和Bruker Avance 300)核磁仪,测定溶剂为氘代二甲基亚砜(DMSO-d6),氘代氯仿(CDCl3),氘代甲醇(CD3OD),内标为四甲基硅烷(TMS);Compound structures were determined by nuclear magnetic resonance (NMR) or/and mass spectroscopy (MS). NMR shifts (δ) are given in units of 10 -6 (ppm). The determination of NMR is to use (Bruker Avance III 400 and Bruker Avance 300) nuclear magnetic instrument, and measuring solvent is deuterated dimethyl sulfoxide (DMSO-d 6 ), deuterated chloroform (CDCl 3 ), deuterated methanol (CD 3 OD ), internal standard is tetramethylsilane (TMS);
MS的测定用(Agilent 6120B(ESI)和Agilent 6120B(APCI));For MS determination (Agilent 6120B (ESI) and Agilent 6120B (APCI));
HPLC的测定使用Agilent 1260DAD高压液相色谱仪(Zorbax SB-C18 100×4.6mm,3.5 μM);The determination of HPLC uses Agilent 1260DAD high pressure liquid chromatography (Zorbax SB-C18 100×4.6mm, 3.5 μM);
薄层层析硅胶板使用烟台黄海HSGF254或青岛GF254硅胶板,薄层色谱法(TLC)使用的硅胶板采用的规格是0.15mm-0.20mm,薄层层析分离纯化产品采用的规格是0.4mm-0.5mm;The thin-layer chromatography silica gel plate uses Yantai Huanghai HSGF 254 or Qingdao GF 254 silica gel plate, the specification of the silica gel plate used in thin-layer chromatography (TLC) is 0.15mm-0.20mm, and the specification of thin-layer chromatography separation and purification products is 0.4mm-0.5mm;
柱层析一般使用烟台黄海硅胶200-300目硅胶为载体;Column chromatography generally uses Yantai Huanghai silica gel 200-300 mesh silica gel as the carrier;
Boc:叔丁氧基羰基;Boc: tert-butoxycarbonyl;
Ts:对甲苯磺酰基;Ts: p-toluenesulfonyl;
Cbz:苄氧羰基;Cbz: benzyloxycarbonyl;
TMS:三甲基硅基;TMS: Trimethylsilyl;
TIPS:三异丙基硅基;TIPS: Triisopropylsilyl;
Bpin:嚬哪醇硼酸酯;Bpin: Nalcohol borate;
MOM:亚甲基甲醚;MOM: methylene methyl ether;
HATU:2-(7-氮杂苯并三氮唑)-N,N,N',N'-四甲基脲六氟磷酸盐;HATU: 2-(7-azabenzotriazole)-N,N,N',N'-tetramethyluronium hexafluorophosphate;
LiHMDS:双三甲基硅基胺基锂;LiHMDS: lithium bistrimethylsilylamide;
EA:乙酸乙酯;EA: ethyl acetate;
MFSDA:氟磺酰基二氟乙酸甲酯;MFSDA: methyl fluorosulfonyl difluoroacetate;
DIPEA:N,N-二异丙基乙胺;DIPEA: N,N-Diisopropylethylamine;
DMF:N,N-二甲基甲酰胺;DMF: N,N-dimethylformamide;
PE:石油醚;PE: petroleum ether;
DCM:二氯甲烷;DCM: dichloromethane;
NIS:N-碘代丁二酰亚胺;NIS: N-iodosuccinimide;
Pd(dppf)Cl2:1,1'-双二苯基膦二茂铁二氯化钯;Pd(dppf)Cl 2 : 1,1'-bisdiphenylphosphinoferrocenepalladium dichloride;
NBS:N-溴代琥珀酰亚胺;NBS: N-bromosuccinimide;
ACN:乙腈;ACN: acetonitrile;
TFA:三氟乙酸;TFA: trifluoroacetic acid;
THF:四氢呋喃。THF: Tetrahydrofuran.
实施例1:4-(2-{[((2R,7aS)-2-氟-六氢-1H-吡咯嗪-7a-基]甲氧基]-4-(3,8-二氮杂双环 [3.2.1]辛-3-基)吡啶基[2,3-d]嘧啶-7-基)-5-乙炔基-6-氟萘-2-醇(化合物1);Example 1: 4-(2-{[((2R,7aS)-2-fluoro-hexahydro-1H-pyrrolidin-7a-yl]methoxy]-4-(3,8-diazabicyclo [3.2.1] Oct-3-yl)pyridyl[2,3-d]pyrimidin-7-yl)-5-ethynyl-6-fluoronaphthalen-2-ol (compound 1);
4-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolizin-7a-yl]methoxy}-4-(3,8-diazabicyclo[3.2.1]o ctan-3-yl)pyrido[2,3-d]pyrimidin-7-yl)-5-ethynyl-6-fluoronaphthalen-2-ol.4-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolizin-7a-yl]methoxy}-4-(3,8-diazabicyclo[3.2.1]octan-3-yl) pyrido[2,3-d]pyrimidin-7-yl)-5-ethynyl-6-fluoronaphthalen-2-ol.
第一步:3-(2,7-二氯吡啶并[2,3-d]嘧啶-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸叔丁酯(1b);The first step: tertiary 3-(2,7-dichloropyrido[2,3-d]pyrimidin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate Butyl ester (1b);
tert-butyl 3-(2,7-dichloropyrido[2,3-d]pyrimidin-4-yl)-3,8-diazabicyclo[3.2.1] octane-8-carboxylate.tert-butyl 3-(2,7-dichloropyrido[2,3-d]pyrimidin-4-yl)-3,8-diazabicyclo[3.2.1] octane-8-carboxylate.
将化合物2,4,7-三氯吡啶并[2,3-d]嘧啶(300mg,1.279mmol)溶于10mL二氯甲烷中,氮气保护下,将体系置于冰浴下搅拌5min,然后在此条件下依次加入N,N-二异丙基乙胺(0.42 mL,2.558mmol)、3,8-二氮杂双环[3.2.1]辛烷-8-羧酸叔丁酯(300mg,1.41mmol),缓慢升至室温反应2h。减压除去溶剂,残余物硅胶柱层析(PE:EA=3:1)得到目标化合物(1b)(520 mg,收率:99%)。The compound 2,4,7-trichloropyrido[2,3-d]pyrimidine (300mg, 1.279mmol) was dissolved in 10mL of dichloromethane, under the protection of nitrogen, the system was placed in an ice bath and stirred for 5min, then in Under this condition, N,N-diisopropylethylamine (0.42 mL, 2.558 mmol), 3,8-diazabicyclo[3.2.1]octane-8-carboxylic acid tert-butyl ester (300 mg, 1.41 mmol), slowly rise to room temperature for 2h. The solvent was removed under reduced pressure, and the residue was subjected to silica gel column chromatography (PE:EA=3:1) to obtain the target compound (1b) (520 mg, yield: 99%).
Ms m/z(ESI):410.1[M+H]+ Ms m/z(ESI):410.1[M+H] +
第二步:3-(7-氯-2-((((2R,7aS)-2-氟四氢-1H-吡咯嗪7a(5H)-基)甲氧基)吡啶基[2,3-d] 嘧啶-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸叔丁酯(1e);The second step: 3-(7-chloro-2-((((2R,7aS)-2-fluorotetrahydro-1H-pyrrolazin7a(5H)-yl)methoxy)pyridyl[2,3- d] tert-butyl pyrimidin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate (1e);
tert-butyl 3-(7-chloro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl) methoxy)pyrido[2,3-d]pyrimidin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate.tert-butyl 3-(7-chloro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl) methoxy)pyrido[2,3-d]pyrimidin-4-yl )-3,8-diazabicyclo[3.2.1]octane-8-carboxylate.
将化合物(1b)(240mg,0.585mmol)和化合物((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲醇(1c-1)(186mg,1.17mmol)溶于10mL超干1,4-二氧六环溶液中,然后加入N,N-二异丙基乙胺(0.29mL,1.755mmol)和4A分子筛(300mg)。氮气保护下,升至100℃反应24h,减压除去溶剂,残余物经硅胶柱层析(DCM:MeOH=20:1)得到目标化合物(1e)(210mg,收率:67%)。Compound (1b) (240mg, 0.585mmol) and compound ((2R, 7aS)-2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methanol (1c-1) (186mg, 1.17mmol) Dissolve in 10 mL of ultra-dry 1,4-dioxane solution, then add N,N-diisopropylethylamine (0.29 mL, 1.755 mmol) and 4A molecular sieves (300 mg). Under the protection of nitrogen, the temperature was raised to 100°C for 24 h, the solvent was removed under reduced pressure, and the residue was subjected to silica gel column chromatography (DCM:MeOH=20:1) to obtain the target compound (1e) (210 mg, yield: 67%).
Ms m/z(ESI):533.2[M+H]+ Ms m/z(ESI):533.2[M+H] +
第三步:3-(7-(7-氟-3-(甲氧基甲氧基)-8-((三异丙基甲硅烷基)乙炔基)萘-1-基) -2-(((((2R,7aS)-2-氟四氢-1H-吡咯嗪7a(5H)-基)甲氧基)吡啶基[2,3-d]嘧啶-4-基)-3,8- 二氮杂双环[3.2.1]辛烷-8-羧酸叔丁酯(1f);The third step: 3-(7-(7-fluoro-3-(methoxymethoxy)-8-((triisopropylsilyl)ethynyl)naphthalene-1-yl)-2-( ((((2R,7aS)-2-fluorotetrahydro-1H-pyrrolazin7a(5H)-yl)methoxy)pyridyl[2,3-d]pyrimidin-4-yl)-3,8- tert-butyl diazabicyclo[3.2.1]octane-8-carboxylate (1f);
tert-butyl 3-(7-(7-fluoro-3-(methoxymethoxy)-8-((triisopropylsilyl)ethynyl) naphthalen-1-yl)-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)pyrido[2,3-d]py rimidin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate.tert-butyl 3-(7-(7-fluoro-3-(methoxymethoxy)-8-((triisopropylsilyl)ethylnyl) naphthalen-1-yl)-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)pyrido[2,3-d]py rimidin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate.
将化合物(1e)(210mg,0.394mmol)和化合物((2-氟-6-(甲氧基甲氧基)-8-(4,4,5,5- 四甲基-1,3,2-二氧杂硼硼烷-2-基)萘-1-基)乙炔基)三异丙基硅烷(1d-1)(404mg,0.788mmol) 置于封管中,加入15mL 1,4-二氧六环溶液溶解,然后依次加入[1,1'-双(二苯基膦基)二茂铁] 二氯化钯(32mg,0.04mmol)、碳酸铯(385mg,1.18mmol)和1.5mL水。在氮气保护下,升至100℃反应3h,减压除去溶剂,加入15mL水和30mL乙酸乙酯溶解,水相用20mL×2 乙酸乙酯洗,合并有机相,有机相用10mL饱和食盐水洗一次,然后有机相用无水硫酸钠干燥,旋干,柱层析(DCM:MeOH=15:1)得到目标化合物(1f),黄色固体(230mg,收率:66%)。Compound (1e) (210 mg, 0.394 mmol) and compound ((2-fluoro-6-(methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2 -Dioxaborolan-2-yl)naphthalen-1-yl)ethynyl)triisopropylsilane (1d-1) (404mg, 0.788mmol) was placed in a sealed tube, and 15mL of 1,4-bis The oxane solution was dissolved, and then [1,1'-bis(diphenylphosphino)ferrocene]palladium dichloride (32mg, 0.04mmol), cesium carbonate (385mg, 1.18mmol) and 1.5mL of water were added successively . Under nitrogen protection, rise to 100°C for 3 hours, remove the solvent under reduced pressure, add 15mL water and 30mL ethyl acetate to dissolve, wash the aqueous phase with 20mL×2 ethyl acetate, combine the organic phases, and wash the organic phase once with 10mL saturated brine , and then the organic phase was dried with anhydrous sodium sulfate, spin-dried, and column chromatography (DCM:MeOH=15:1) gave the target compound (1f) as a yellow solid (230 mg, yield: 66%).
Ms m/z(ESI):442.3[(M+2H)/2]+ Ms m/z(ESI):442.3[(M+2H)/2] +
第四步:4-(4-(3,8-二氮杂双环[3.2.1]辛烷-3-基)-2-(((((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)吡啶并[2,3-d]嘧啶-7-基)-6-氟-5-((三异丙基甲硅烷基)乙炔基) 萘-2-醇(1g);The fourth step: 4-(4-(3,8-diazabicyclo[3.2.1]octane-3-yl)-2-(((((2R,7aS)-2-fluorotetrahydro-1H -pyrrolazin-7a(5H)-yl)methoxy)pyrido[2,3-d]pyrimidin-7-yl)-6-fluoro-5-((triisopropylsilyl)ethynyl) Naphthalene-2-ol (1 g);
4-(4-(3,8-diazabicyclo[3.2.1]octan-3-yl)-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)pyrido[2,3-d]pyrimidin-7-yl)-6-fluoro-5-((triisopropylsilyl)ethynyl)naphthalen-2-ol.4-(4-(3,8-diazabicyclo[3.2.1]octan-3-yl)-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy) pyrido[2,3-d]pyrimidin-7-yl)-6-fluoro-5-((triisopropylsilyl)ethynyl)naphthalen-2-ol.
将化合物(1f)(230mg,0.26mmol)溶于5mL乙腈中,然后于室温下滴加2mL盐酸1,4-二氧六环溶液,体系继续反应3h,减压除去多余的酸和溶剂,得粗品化合物(1g),(240mg),不需纯化直接用于下一步反应。Compound (1f) (230mg, 0.26mmol) was dissolved in 5mL of acetonitrile, then 2mL of hydrochloric acid 1,4-dioxane solution was added dropwise at room temperature, the system continued to react for 3h, and the excess acid and solvent were removed under reduced pressure to obtain The crude compound (1 g), (240 mg) was directly used in the next reaction without purification.
Ms m/z(ESI):739.3[M+H]+ Ms m/z(ESI):739.3[M+H] +
第五步:4-(2-{[((2R,7aS)-2-氟-六氢-1H-吡咯嗪-7a-基]甲氧基]-4-(3,8-二氮杂双环 [3.2.1]辛-3-基)吡啶基[2,3-d]嘧啶-7-基)-5-乙炔基-6-氟萘-2-醇(化合物1);The fifth step: 4-(2-{[((2R,7aS)-2-fluoro-hexahydro-1H-pyrrolazin-7a-yl]methoxy]-4-(3,8-diazabicyclo [3.2.1] Oct-3-yl)pyridyl[2,3-d]pyrimidin-7-yl)-5-ethynyl-6-fluoronaphthalen-2-ol (compound 1);
4-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolizin-7a-yl]methoxy}-4-(3,8-diazabicyclo[3.2.1]o ctan-3-yl)pyrido[2,3-d]pyrimidin-7-yl)-5-ethynyl-6-fluoronaphthalen-2-ol.4-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolizin-7a-yl]methoxy}-4-(3,8-diazabicyclo[3.2.1]octan-3-yl) pyrido[2,3-d]pyrimidin-7-yl)-5-ethynyl-6-fluoronaphthalen-2-ol.
将上一步粗品化合物(1g)(240mg)溶于2mL N,N-二甲基甲酰胺中,随后于室温下加入氟化铯(200mg,1.3mmol)。加完后,氮气保护下继续反应12h,向体系中加入30mL乙酸乙酯稀释,然后依次用10mL饱和碳酸氢钠、10mL×3水、10mL饱和食盐水洗有机相,有机相用无水硫酸钠干燥,浓缩,残余物经硅胶柱层析(DCM:MeOH=5:1)得到目标化合物(化合物1),黄色固体(30mg,两步收率:20%)。Crude compound (1 g) (240 mg) from the previous step was dissolved in 2 mL of N,N-dimethylformamide, then cesium fluoride (200 mg, 1.3 mmol) was added at room temperature. After the addition, continue to react for 12 h under the protection of nitrogen, add 30 mL of ethyl acetate to the system to dilute, then wash the organic phase with 10 mL of saturated sodium bicarbonate, 10 mL×3 water, and 10 mL of saturated saline, and dry the organic phase with anhydrous sodium sulfate , concentrated, and the residue was subjected to silica gel column chromatography (DCM:MeOH=5:1) to obtain the target compound (compound 1) as a yellow solid (30 mg, two-step yield: 20%).
Ms m/z(ESI):583.3[M+H]+ Ms m/z(ESI):583.3[M+H] +
实施例2:4-(2-{[(2R,7aS)-2-氟-六氢-1H-吡咯嗪-7a-基]甲氧基}-4-[3,8-二氮杂双环[3.2.1]辛烷-3-基]-8-氟喹唑啉-7-基)-5-乙炔基-6-氟萘-2-醇(化合物2)的三氟乙酸盐Example 2: 4-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolidin-7a-yl]methoxy}-4-[3,8-diazabicyclo[ 3.2.1] Octane-3-yl]-8-fluoroquinazolin-7-yl)-5-ethynyl-6-fluoronaphthalen-2-ol (compound 2) trifluoroacetate salt
4-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolizin-7a-yl]methoxy}-4-[3,8-diazabicyclo[3.2.1]o ctan-3-yl]-8-fluoroquinazolin-7-yl)-5-ethynyl-6-fluoronaphthalen-2-ol;trifluoroacetic acid4-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolizin-7a-yl]methoxy}-4-[3,8-diazabicyclo[3.2.1]octan-3-yl] -8-fluoroquinazolin-7-yl)-5-ethynyl-6-fluoronaphthalen-2-ol; trifluoroacetic acid
第一步:2-氨基-4-溴-3-氟苯甲酰胺(2B)The first step: 2-amino-4-bromo-3-fluorobenzamide (2B)
2-amino-4-bromo-3-fluorobenzamide2-amino-4-bromo-3-fluorobenzamide
将2-氨基-4-溴-3氟苯甲酸(2A)(5g,21.46mmol)溶于60mL N,N-二甲基甲酰胺中,分别加入HATU(12.24g,32.18mmol),N,N-二异丙基乙胺(5.53g,42.92mmol)和氯化铵(11.48 g,214.60mmol),并在室温下继续反应3h。将反应液缓慢倒入120mL冰水中,并在室温下搅拌10min,将析出的固体过滤,干燥,减压浓缩后即得到化合物(2B)(4.9g,产率98.39%)。Dissolve 2-amino-4-bromo-3fluorobenzoic acid (2A) (5g, 21.46mmol) in 60mL of N,N-dimethylformamide, add HATU (12.24g, 32.18mmol), N,N -Diisopropylethylamine (5.53g, 42.92mmol) and ammonium chloride (11.48g, 214.60mmol), and continue the reaction at room temperature for 3h. The reaction solution was slowly poured into 120 mL of ice water and stirred at room temperature for 10 min. The precipitated solid was filtered, dried, and concentrated under reduced pressure to obtain compound (2B) (4.9 g, yield 98.39%).
LCMS m/z=233.1[M+H]+ LCMS m/z=233.1[M+H] +
第二步:7-溴-8-氟-1,2,3,4-四氢喹唑啉-2,4-二酮(2C)The second step: 7-bromo-8-fluoro-1,2,3,4-tetrahydroquinazoline-2,4-dione (2C)
7-bromo-8-fluoro-1,2,3,4-tetrahydroquinazoline-2,4-dione7-bromo-8-fluoro-1,2,3,4-tetrahydroquinazoline-2,4-dione
将(2B)(4.9g,21.12mmol)溶解于四氢呋喃100mL中,加入三光气(9.4g,31.68mmol),加完后65℃下反应3h。将反应冷却至室温,减压浓缩后即得到化合物(2C)(4.7g,产率86.23%)Dissolve (2B) (4.9 g, 21.12 mmol) in 100 mL of tetrahydrofuran, add triphosgene (9.4 g, 31.68 mmol), and react at 65° C. for 3 h after the addition. The reaction was cooled to room temperature and concentrated under reduced pressure to obtain compound (2C) (4.7 g, yield 86.23%)
LCMS m/z=259.1[M+H]+ LCMS m/z=259.1[M+H] +
第三步:7-溴-2,4-二氯-8-氟喹唑啉(2D)The third step: 7-bromo-2,4-dichloro-8-fluoroquinazoline (2D)
7-bromo-2,4-dichloro-8-fluoroquinazoline7-bromo-2,4-dichloro-8-fluoroquinazoline
将(2C)(1.5g,5.81mmol)溶解于甲苯30mL中,分别加入三氯氧磷(4.45g,29.07mmol), N,N-二异丙基乙胺(2.25g,17.44mmol),加完后110℃下反应5h。将反应冷却至室温,减压浓缩后直接得到化合物(2D)棕色液体并直接用于下一步反应。Dissolve (2C) (1.5g, 5.81mmol) in 30mL of toluene, add phosphorus oxychloride (4.45g, 29.07mmol), N,N-diisopropylethylamine (2.25g, 17.44mmol), and After completion, react at 110°C for 5h. The reaction was cooled to room temperature and concentrated under reduced pressure to directly obtain compound (2D) as a brown liquid, which was directly used in the next reaction.
第四步:the fourth step:
3-(7-溴-2-氯-8-氟喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸叔丁酯(2E)tert-butyl 3-(7-bromo-2-chloro-8-fluoroquinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate (2E)
tert-butyltert-butyl
3-(7-bromo-2-chloro-8-fluoroquinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate3-(7-bromo-2-chloro-8-fluoroquinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
将(2D)溶解于干燥二氯甲烷30mL中,冰浴下缓慢加入N,N-二异丙基乙胺(2.25g,17.44 mmol),中间体1(1.23g,5.81mmol),加完后缓慢升至室温反应2h,减压浓缩后,残留物用硅胶色谱柱柱层析得到化合物(2E)(0.71g,两步产率26.00%)。Dissolve (2D) in 30 mL of dry dichloromethane, slowly add N,N-diisopropylethylamine (2.25 g, 17.44 mmol), intermediate 1 (1.23 g, 5.81 mmol) under ice-cooling, after the addition is complete Slowly rise to room temperature and react for 2 h. After concentration under reduced pressure, the residue was chromatographed on a silica gel column to obtain compound (2E) (0.71 g, two-step yield 26.00%).
LCMS m/z=471.1[M+H]+ LCMS m/z=471.1[M+H] +
第五步:the fifth step:
3-(2-{[(2R,7aS)-2-氟-六氢-1H-吡咯嗪-7a-基]甲氧基}-7-溴-8-氟喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸叔丁酯(2F)3-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolidin-7a-yl]methoxy}-7-bromo-8-fluoroquinazolin-4-yl)- tert-butyl 3,8-diazabicyclo[3.2.1]octane-8-carboxylate (2F)
tert-butyltert-butyl
3-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolizin-7a-yl]methoxy}-7-bromo-8-fluoroquinazolin-4-y l)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate3-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolizin-7a-yl]methoxy}-7-bromo-8-fluoroquinazolin-4-y l)-3,8-diazabicyclo[3.2 .1] octane-8-carboxylate
将中间体2(0.4g,2.51mmol)溶解于N,N-二甲基甲酰胺15mL中,加入碳酸铯(0.55g, 1.70mmol),三乙烯二胺(0.03g,0.26mmol),并加入分子筛500mg,氮气保护下室温下反应10min,将化合物2E(0.4g,0.85mmol)溶解于N,N-二甲基甲酰胺2mL中,并将其缓慢加入反应液中,加完后室温反应4h,缓慢加入水40mL,乙酸乙酯(40mL×2)萃取两次,合并乙酸乙酯层,乙酸乙酯层水(40mL×2)洗涤两次,并饱和食盐水洗涤一次(40mL×1),无水硫酸钠干燥,减压浓缩后,残留物用硅胶色谱柱柱层析得到化合物(2F)(0.36g,产率71.42%)Intermediate 2 (0.4g, 2.51mmol) was dissolved in N,N-dimethylformamide 15mL, cesium carbonate (0.55g, 1.70mmol), triethylenediamine (0.03g, 0.26mmol) were added, and Molecular sieves 500mg, reaction at room temperature for 10min under nitrogen protection, dissolving compound 2E (0.4g, 0.85mmol) in 2mL of N,N-dimethylformamide, and slowly adding it to the reaction solution, and reacting at room temperature for 4h after the addition , slowly add 40 mL of water, extract twice with ethyl acetate (40 mL×2), combine the ethyl acetate layers, wash the ethyl acetate layer with water (40 mL×2) twice, and wash once with saturated brine (40 mL×1), After drying over anhydrous sodium sulfate and concentrating under reduced pressure, the residue was chromatographed on a silica gel column to obtain compound (2F) (0.36 g, yield 71.42%)
LCMS m/z=594.1[M+H]+ LCMS m/z=594.1[M+H] +
第六步:Step six:
3-(2-{[(2R,7aS)-2-氟-六氢-1H-吡咯嗪-7a-基]甲氧基}-8-氟-7-(7-氟-3-(甲氧甲基)-8-(2-(三 (丙烷-2-基)硅基)乙炔基)萘-1-基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸叔丁酯(2G)3-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolidin-7a-yl]methoxy}-8-fluoro-7-(7-fluoro-3-(methoxy Methyl)-8-(2-(tri(propan-2-yl)silyl)ethynyl)naphthalene-1-yl)quinazolin-4-yl)-3,8-diazabicyclo[3.2. 1] tert-butyl octane-8-carboxylate (2G)
tert-butyltert-butyl
3-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolizin-7a-yl]methoxy}-8-fluoro-7-(7-fluoro-3-(methoxy methoxy)-8-(2-(tris(propan-2-yl)silyl)ethynyl)naphthalen-1-yl)quinazolin-4-yl)-3,8-diazabicyclo[3.2. 1]octane-8-carboxylate3-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolizin-7a-yl]methoxy}-8-fluoro-7-(7-fluoro-3-(methoxy methoxy)-8- (2-(tris(propan-2-yl)silyl)ethynyl)naphthalen-1-yl)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
将(2F)(0.36g,0.60mmol)溶解于1,4-二氧六环15mL和水3mL的混合溶液中,加入中间体3(0.62g,1.20mmol),碳酸铯(0.39g,1.20mmol),Pd(dppf)Cl2的二氯甲烷复合物(0.10g, 0.12mmol),氮气保护下100℃下反应5h,将反应冷却至室温,加入乙酸乙酯40mL和水25mL, 水相再用乙酸乙酯(40mL×1)萃取一次,合并乙酸乙酯层,乙酸乙酯层用饱和食盐水洗涤一次 (40mL×1),无水硫酸钠干燥,减压浓缩后,残留物用硅胶色谱柱柱层析得到化合物(2G)(0.24g,产率44.03%)Dissolve (2F) (0.36g, 0.60mmol) in a mixed solution of 1,4-dioxane 15mL and water 3mL, add intermediate 3 (0.62g, 1.20mmol), cesium carbonate (0.39g, 1.20mmol ), Pd(dppf)Cl 2 dichloromethane complex (0.10g, 0.12mmol), reacted at 100°C for 5h under nitrogen protection, cooled the reaction to room temperature, added 40mL of ethyl acetate and 25mL of water, and reused the aqueous phase Extract with ethyl acetate (40mL×1) once, combine the ethyl acetate layers, wash the ethyl acetate layer with saturated brine once (40mL×1), dry over anhydrous sodium sulfate, concentrate under reduced pressure, the residue is chromatographed on a silica gel column Column chromatography obtained compound (2G) (0.24g, yield 44.03%)
LCMS m/z=900.1[M+H]+ LCMS m/z=900.1[M+H] +
第七步:Step seven:
4-(2-{[(2R,7aS)-2-氟-六氢-1H-吡咯嗪-7a-基]甲氧基}-4-[3,8-二氮杂双环[3.2.1]辛烷-3- 基]-8-氟喹唑啉-7-基)-6-氟-5-(2-(三(丙烷-2-基)硅基)乙炔基)萘-2-醇(2H)4-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolazin-7a-yl]methoxy}-4-[3,8-diazabicyclo[3.2.1] Octane-3-yl]-8-fluoroquinazolin-7-yl)-6-fluoro-5-(2-(tri(propane-2-yl)silyl)ethynyl)naphthalene-2-alcohol ( 2H)
4-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolizin-7a-yl]methoxy}-4-[3,8-diazabicyclo[3.2.1]o ctan-3-yl]-8-fluoroquinazolin-7-yl)-6-fluoro-5-(2-(tris(propan-2-yl)silyl)ethynyl)naphthalen-2-ol4-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolizin-7a-yl]methoxy}-4-[3,8-diazabicyclo[3.2.1]octan-3-yl] -8-fluoroquinazolin-7-yl)-6-fluoro-5-(2-(tris(propan-2-yl)silyl)ethynyl)naphthalen-2-ol
将化合物(2G)(0.15g,0.16mmol)溶解于二氯甲烷10mL中,加入三氟乙酸0.5mL,加完后室温反应3h,减压浓缩后直接得到化合物(2H)直接用于下一步反应。Dissolve compound (2G) (0.15g, 0.16mmol) in 10mL of dichloromethane, add 0.5mL of trifluoroacetic acid, react at room temperature for 3h after addition, and concentrate under reduced pressure to obtain compound (2H) directly for the next reaction .
LCMS m/z=756.1[M+H]+ LCMS m/z=756.1[M+H] +
第八步:Step Eight:
4-(2-{[(2R,7aS)-2-氟-六氢-1H-吡咯嗪-7a-基]甲氧基}-4-[3,8-二氮杂双环[3.2.1]辛烷-3- 基]-8-氟喹唑啉-7-基)-5-乙炔基-6-氟萘-2-醇(化合物2)的三氟乙酸盐4-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolizin-7a-yl]methoxy}-4-[3,8-diazabicyclo[3.2.1] Trifluoroacetate salt of octane-3-yl]-8-fluoroquinazolin-7-yl)-5-ethynyl-6-fluoronaphthalen-2-ol (compound 2)
4-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolizin-7a-yl]methoxy}-4-[3,8-diazabicyclo[3.2.1]o ctan-3-yl]-8-fluoroquinazolin-7-yl)-5-ethynyl-6-fluoronaphthalen-2-ol;trifluoroacetic acid4-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolizin-7a-yl]methoxy}-4-[3,8-diazabicyclo[3.2.1]octan-3-yl] -8-fluoroquinazolin-7-yl)-5-ethynyl-6-fluoronaphthalen-2-ol; trifluoroacetic acid
将化合物(2H)溶解于N,N-二甲基甲酰胺6mL中,加入氟化铯(0.25g,0.16mmol),室温下反应16h,减压浓缩除去反应溶剂,残留物经HPLC制备纯化,制备液浓缩后得到化合物2的三氟乙酸盐(7mg,两步产率7.1%).Compound (2H) was dissolved in 6 mL of N,N-dimethylformamide, cesium fluoride (0.25 g, 0.16 mmol) was added, reacted at room temperature for 16 h, concentrated under reduced pressure to remove the reaction solvent, and the residue was prepared and purified by HPLC. The preparation was concentrated to give the trifluoroacetate salt of compound 2 (7 mg, 7.1% yield in two steps).
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%TFA);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% TFA); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min.
1H NMR(400MHz,CD3OD)δ7.91–7.82(m,2H),7.46–7.39(m,1H),7.36–7.28(m,2H),7.09(d,1H),5.62–5.44(m,1H),4.77–4.62(m,4H),4.32–4.20(m,2H),4.06–3.75(m,5H),3.50–3.40(m,1H),3.22(s,1H),2.75–2.56(m,2H),2.50–2.28(m,3H),2.26–2.05(m,5H). 1 H NMR (400MHz, CD 3 OD) δ7.91–7.82(m,2H),7.46–7.39(m,1H),7.36–7.28(m,2H),7.09(d,1H),5.62–5.44( m,1H),4.77–4.62(m,4H),4.32–4.20(m,2H),4.06–3.75(m,5H),3.50–3.40(m,1H),3.22(s,1H),2.75– 2.56(m,2H),2.50–2.28(m,3H),2.26–2.05(m,5H).
LCMS m/z=600.1[M+H]+ LCMS m/z=600.1[M+H] +
实施例3:4-(9-(3,8-二氮杂双环[3.2.1]辛-3-基)-7-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)- 基)甲氧基)呋喃[2,3-f]喹唑啉-4-基)-5-乙炔基-6-氟萘-2-醇(化合物3)Example 3: 4-(9-(3,8-diazabicyclo[3.2.1]oct-3-yl)-7-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolazine -7a(5H)-yl)methoxy)furo[2,3-f]quinazolin-4-yl)-5-ethynyl-6-fluoronaphthalen-2-ol (compound 3)
4-(9-(3,8-diazabicyclo[3.2.1]octan-3-yl)-7-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)m ethoxy)furo[2,3-f]quinazolin-4-yl)-5-ethynyl-6-fluoronaphthalen-2-ol4-(9-(3,8-diazabicyclo[3.2.1]octan-3-yl)-7-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)m ethoxy )furo[2,3-f]quinazolin-4-yl)-5-ethynyl-6-fluoronaphthalen-2-ol
第一步:3-(4-溴-7-氯呋喃[2,3-f]喹唑啉-9-基)-3,8-二氮杂双环[3.2.1]辛-8-羧酸叔丁酯 tert-butylThe first step: 3-(4-bromo-7-chlorofuro[2,3-f]quinazolin-9-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylic acid Tert-butyl ester tert-butyl
3-(4-bromo-7-chlorofuro[2,3-f]quinazolin-9-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate3-(4-bromo-7-chlorofuro[2,3-f]quinazolin-9-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
将原料3a(1.2g,4.27mmol)溶于25mL 1,4二氧六环中,加入N,N-二异丙基乙胺(2.76g,21.35mmol),随后于室温下缓慢滴加三氯氧磷(6.55g,42.7mmol)。滴加完毕后,体系在氮气保护下加热至100℃反应3小时。冷却至室温,减压除去多余的三氯氧磷和溶剂,得到粗产品为褐色油状液体,将残留物溶于10mL二氯甲烷中,氮气保护下,将体系置于冰浴下依次加入N,N-二异丙基乙胺(2.76g,21.35mmol)、3,8-二氮杂双环[3.2.1]辛烷-8-羧酸叔丁酯(0.91g, 4.27mmol),加完缓慢升至室温反应2h。减压除去溶剂,残余物硅胶柱层析(PE:EA=3:1) 得到目标化合物3b(0.16g,产率7.6%)Starting material 3a (1.2g, 4.27mmol) was dissolved in 25mL of 1,4-dioxane, N,N-diisopropylethylamine (2.76g, 21.35mmol) was added, followed by slowly dropwise addition of trichloro Phosphorus (6.55 g, 42.7 mmol). After the dropwise addition, the system was heated to 100° C. for 3 hours under the protection of nitrogen. Cool to room temperature, remove excess phosphorus oxychloride and solvent under reduced pressure, and obtain the crude product as a brown oily liquid. Dissolve the residue in 10 mL of dichloromethane. Under nitrogen protection, place the system in an ice bath and add N successively. N-diisopropylethylamine (2.76g, 21.35mmol), 3,8-diazabicyclo[3.2.1]octane-8-carboxylic acid tert-butyl ester (0.91g, 4.27mmol), added slowly Raised to room temperature for 2h. The solvent was removed under reduced pressure, and the residue was subjected to silica gel column chromatography (PE:EA=3:1) to obtain the target compound 3b (0.16 g, yield 7.6%)
LCMS m/z=493.0[M+1]+ LCMS m/z=493.0[M+1] +
第二步:3-(4-溴-7-((((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)呋喃[2,3-f] 喹唑啉-9-基)-3,8-二氮杂双环[3.2.1]辛-8-羧酸叔丁酯The second step: 3-(4-bromo-7-((((2R,7aS)-2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)furan[2,3- f] quinazoline-9-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylic acid tert-butyl ester
tert-butyltert-butyl
3-(4-bromo-7-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)furo[2,3-f]quinazolin -9-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate3-(4-bromo-7-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)furo[2,3-f]quinazolin-9-yl)-3 ,8-diazabicyclo[3.2.1]octane-8-carboxylate
将原料3b(140mg,0.28mmol),((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲醇(89mg,0.56mmol),碳酸铯(270mg,0.84mmol),三乙烯二胺(6mg,0.056mmol)溶于 10mL DMF中,加入分子筛,氮气保护室温反应6h。反应完全后,加入15mL水,用乙酸乙酯萃取2次,合并有机相,有机相用10mL饱和食盐水洗一次,然后有机相用无水硫酸钠干燥,浓缩,残余物经硅胶柱层析分离纯化得到目标化合物3c(140mg,产率81%)Starting material 3b (140 mg, 0.28 mmol), ((2R, 7aS)-2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methanol (89 mg, 0.56 mmol), cesium carbonate (270 mg, 0.84 mmol) ), triethylenediamine (6mg, 0.056mmol) was dissolved in 10mL DMF, added Molecular sieves, under nitrogen protection, reacted at room temperature for 6h. After the reaction is complete, add 15 mL of water, extract twice with ethyl acetate, combine the organic phases, wash the organic phase with 10 mL of saturated brine once, then dry the organic phase with anhydrous sodium sulfate, concentrate, and the residue is separated and purified by silica gel column chromatography Obtain target compound 3c (140mg, yield 81%)
LCMS m/z=616.2[M+1]+ LCMS m/z=616.2[M+1] +
第三步:3-(4-(7-氟-3-(甲氧基甲氧基)-8-((三异丙基甲硅烷基)乙炔基)萘-1-基) -7-((((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)呋喃[2,3-f]喹唑啉-9-基) -3,8-二氮杂双环[3.2.1]辛-8-羧酸叔丁酯tert-butylThe third step: 3-(4-(7-fluoro-3-(methoxymethoxy)-8-((triisopropylsilyl)ethynyl)naphthalene-1-yl)-7-( (((2R,7aS)-2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)furo[2,3-f]quinazolin-9-yl)-3,8 - tert-butyl diazabicyclo[3.2.1]oct-8-carboxylate
3-(4-(7-fluoro-3-(methoxymethoxy)-8-((triisopropylsilyl)ethynyl)naphthalen-1-yl)-7-(((2R,7aS)-2-fl uorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)furo[2,3-f]quinazolin-9-yl)-3,8-diazabicyclo[3.2.1] octane-8-carboxylate3-(4-(7-fluoro-3-(methoxymethoxy)-8-((triisopropylsilyl)ethylnyl)naphthalen-1-yl)-7-(((2R,7aS)-2-fl uorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)furo[2,3-f]quinazolin-9-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
将3c(140mg,0.23mmol)和化合物((2-氟-6-(甲氧基甲氧基)-8-(4,4,5,5-四甲基-1,3,2- 二氧杂硼硼烷-2-基)萘-1-基)乙炔基)三异丙基硅烷(240mg,0.46mmol)置于封管中,加入10mL 1,4-二氧六环溶液溶解,然后依次加入[1,1'-双(二苯基膦基)二茂铁]二氯化钯(50mg, 0.069mmol)、碳酸铯(220mg,0.69mmol)和2mL水。在氮气保护下,升至100℃反应3h,冷却至室温,减压除去溶剂,加入15mL水和30mL乙酸乙酯溶解,水相用20mL×2乙酸乙酯洗,合并有机相,有机相用10mL饱和食盐水洗一次,然后有机相用无水硫酸钠干燥,浓缩,柱层析得到目标化合物3d(150mg,产率70.7%)。3c (140 mg, 0.23 mmol) and compound ((2-fluoro-6-(methoxymethoxy)-8-(4,4,5,5-tetramethyl-1,3,2-dioxo Boroboran-2-yl)naphthalene-1-yl)ethynyl)triisopropylsilane (240mg, 0.46mmol) was placed in a sealed tube, 10mL of 1,4-dioxane solution was added to dissolve, and then [1,1'-Bis(diphenylphosphino)ferrocene]palladium dichloride (50 mg, 0.069 mmol), cesium carbonate (220 mg, 0.69 mmol) and 2 mL of water were added. Under the protection of nitrogen, rise to 100°C for 3 hours, cool to room temperature, remove the solvent under reduced pressure, add 15mL water and 30mL ethyl acetate to dissolve, wash the aqueous phase with 20mL×2 ethyl acetate, combine the organic phase, and use 10mL Washed once with saturated brine, then the organic phase was dried over anhydrous sodium sulfate, concentrated, and column chromatography gave the target compound 3d (150 mg, yield 70.7%).
LCMS m/z=922.60[M+1]+ LCMS m/z=922.60[M+1] +
第四步:4-(9-((3,8-二氮杂双环[3.2.1]辛-3-基)-7-((((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)呋喃[2,3-f]喹唑啉-4-基)-6-氟-5-((三异丙基甲硅烷基)乙炔基) 萘-2-醇The fourth step: 4-(9-((3,8-diazabicyclo[3.2.1]oct-3-yl)-7-((((2R,7aS)-2-fluorotetrahydro-1H- Pyrrolazin-7a(5H)-yl)methoxy)furo[2,3-f]quinazolin-4-yl)-6-fluoro-5-((triisopropylsilyl)ethynyl) Naphthalene-2-ol
4-(9-(3,8-diazabicyclo[3.2.1]octan-3-yl)-7-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)m ethoxy)furo[2,3-f]quinazolin-4-yl)-6-fluoro-5-((triisopropylsilyl)ethynyl)naphthalen-2-ol4-(9-(3,8-diazabicyclo[3.2.1]octan-3-yl)-7-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)m ethoxy )furo[2,3-f]quinazolin-4-yl)-6-fluoro-5-((triisopropylsilyl)ethynyl)naphthalen-2-ol
将原料3d(150mg,0.16mmol)溶于10mL乙腈中,然后于室温下滴加2mL盐酸/1, 4-二氧六环溶液,体系继续反应1h,减压除去多余的盐酸和溶剂,得粗品化合物3e,直接用于下一步反应。The raw material 3d (150 mg, 0.16 mmol) was dissolved in 10 mL of acetonitrile, and then 2 mL of hydrochloric acid/1,4-dioxane solution was added dropwise at room temperature. The system continued to react for 1 h, and the excess hydrochloric acid and solvent were removed under reduced pressure to obtain the crude product Compound 3e was directly used in the next reaction.
LCMS m/z=778.3[M+1]+ LCMS m/z=778.3[M+1] +
第五步:4-(9-(3,8-二氮杂双环[3.2.1]辛-3-基)-7-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)呋喃[2,3-f]喹唑啉-4-基)-5-乙炔基-6-氟萘-2-醇The fifth step: 4-(9-(3,8-diazabicyclo[3.2.1]oct-3-yl)-7-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolazine -7a(5H)-yl)methoxy)furo[2,3-f]quinazolin-4-yl)-5-ethynyl-6-fluoronaphthalen-2-ol
4-(9-(3,8-diazabicyclo[3.2.1]octan-3-yl)-7-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)m ethoxy)furo[2,3-f]quinazolin-4-yl)-5-ethynyl-6-fluoronaphthalen-2-ol4-(9-(3,8-diazabicyclo[3.2.1]octan-3-yl)-7-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)m ethoxy )furo[2,3-f]quinazolin-4-yl)-5-ethynyl-6-fluoronaphthalen-2-ol
将原料3e(100mg,0.13mmol)溶于3mL DMF,加入氟化铯(103mg,0.65mmol),室温搅拌16h,直接反相C18柱层析分离纯化得化合物3(40mg,产率49.5%)。The raw material 3e (100mg, 0.13mmol) was dissolved in 3mL DMF, cesium fluoride (103mg, 0.65mmol) was added, stirred at room temperature for 16h, and compound 3 (40mg, yield 49.5%) was obtained by separation and purification by reverse phase C18 column chromatography.
1H NMR(400MHz,CD3OD)δ7.87–7.80(m,2H),7.43(s,1H),7.32–7.25(m,2H),7.14(d, 1H),6.39–6.35(m,1H),5.39–5.21(m,1H),4.33–4.14(m,4H),3.65–3.58(m,2H),3.57–3.46 (m,2H),3.30–3.15(m,3H),3.05–2.96(m,1H),2.95–2.90(m,1H),2.40–2.18(m,2H),2.18– 1.95(m,2H),2.08–1.95(m,3H),1.94–1.82(m,3H). 1 H NMR (400MHz, CD 3 OD) δ7.87–7.80(m,2H),7.43(s,1H),7.32–7.25(m,2H),7.14(d,1H),6.39–6.35(m, 1H),5.39–5.21(m,1H),4.33–4.14(m,4H),3.65–3.58(m,2H),3.57–3.46 (m,2H),3.30–3.15(m,3H),3.05– 2.96(m,1H),2.95–2.90(m,1H),2.40–2.18(m,2H),2.18–1.95(m,2H),2.08–1.95(m,3H),1.94–1.82(m,3H ).
LCMS m/z=622.4[M+1]+.LCMS m/z = 622.4[M+1] + .
实施例4:4-(2-{[(2R,7aS)-2-氟-六氢-1H-吡咯嗪-7a-基]甲氧基}-4-[(1R,5S)-3,8-二氮杂双环 [3.2.1]辛烷-3-基]-8-氟喹唑啉-7-基)-5-乙基-6-氟萘-2-醇(化合物4)Example 4: 4-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolazin-7a-yl]methoxy}-4-[(1R,5S)-3,8 -Diazabicyclo[3.2.1]octane-3-yl]-8-fluoroquinazolin-7-yl)-5-ethyl-6-fluoronaphthalen-2-ol (compound 4)
4-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolizin-7a-yl]methoxy}-4-[(1R,5S)-3,8-diazabicycl o[3.2.1]octan-3-yl]-8-fluoroquinazolin-7-yl)-5-ethyl-6-fluoronaphthalen-2-ol4-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolizin-7a-yl]methoxy}-4-[(1R,5S)-3,8-diazabicyclo[3.2.1] octan-3-yl]-8-fluoroquinazolin-7-yl)-5-ethyl-6-fluoronaphthalen-2-ol
第一步:(1R,5S)-3-(2-{[(2R,7aS)-2-氟-六氢-1H-吡咯嗪-7a-基]甲氧基}-7-(8-乙炔基-7-氟 -3-(甲氧甲基)萘-1-基)-8-氟喹唑啉-4-yl)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸叔丁酯(4B);The first step: (1R,5S)-3-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolazin-7a-yl]methoxy}-7-(8-acetylene Base-7-fluoro-3-(methoxymethyl)naphthalen-1-yl)-8-fluoroquinazoline-4-yl)-3,8-diazabicyclo[3.2.1]octane-8 - tert-butyl carboxylate (4B);
tert-butyltert-butyl
(1R,5S)-3-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolizin-7a-yl]methoxy}-7-(8-ethynyl-7-fluoro-3 -(methoxymethoxy)naphthalen-1-yl)-8-fluoroquinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carbo xylate(1R,5S)-3-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolizin-7a-yl]methoxy}-7-(8-ethynyl-7-fluoro-3 -( methoxymethoxy)naphthalen-1-yl)-8-fluoroquinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carbo xylate
将化合物2G(0.35g,0.39mmol)溶于8mL N,N-二甲基甲酰胺中,加入氟化铯(0.295mg,1.95mmol),室温下搅拌3小时。缓慢加入水20mL,并用乙酸乙酯(20mL×2)萃取两次,合并乙酸乙酯层,乙酸乙酯层用水(20mL×2)洗两次,饱和氯化钠的水溶液20mL洗一次,最后用无水硫酸钠干燥,过滤,减压浓缩后残留物用硅胶色谱柱柱层析分离纯化得到化合物 4B(230mg,收率79.58%)Compound 2G (0.35 g, 0.39 mmol) was dissolved in 8 mL of N,N-dimethylformamide, cesium fluoride (0.295 mg, 1.95 mmol) was added, and stirred at room temperature for 3 hours. Slowly add 20 mL of water, and extract twice with ethyl acetate (20 mL×2), combine the ethyl acetate layers, wash the ethyl acetate layer twice with water (20 mL×2), once with 20 mL of saturated sodium chloride solution, and finally wash with Dry over anhydrous sodium sulfate, filter, and concentrate under reduced pressure, the residue is separated and purified by silica gel column chromatography to obtain compound 4B (230 mg, yield 79.58%)
Ms m/z(ESI):744.3[M+H]+ Ms m/z(ESI):744.3[M+H] +
第二步:(1R,5S)-3-(2-{[(2R,7aS)-2-氟-六氢-1H-吡咯嗪-7a-基]甲氧基}-7-(8-乙基-7-氟-3-(甲氧基甲氧基)萘-1-基)-8-氟喹唑啉-4-yl)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸叔丁酯(4C);The second step: (1R,5S)-3-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolazin-7a-yl]methoxy}-7-(8-ethane Base-7-fluoro-3-(methoxymethoxy)naphthalen-1-yl)-8-fluoroquinazoline-4-yl)-3,8-diazabicyclo[3.2.1]octane - tert-butyl 8-carboxylate (4C);
tert-butyltert-butyl
(1R,5S)-3-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolizin-7a-yl]methoxy}-7-(8-ethyl-7-fluoro-3-( methoxymethoxy)naphthalen-1-yl)-8-fluoroquinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carbox ylate(1R,5S)-3-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolizin-7a-yl]methoxy}-7-(8-ethyl-7-fluoro-3-( methoxymethoxy)naphthalen-1-yl)-8-fluoroquinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
将4B(230mg,0.31mmol)溶解于5mL甲醇中,加入230mg钯碳,氢气条件下室温反应2小时。将反应液过滤,滤液减压浓缩后残留物用硅胶色谱柱柱层析得到化合物4C(160 mg,收率69.26%)4B (230 mg, 0.31 mmol) was dissolved in 5 mL of methanol, 230 mg of palladium on carbon was added, and the reaction was carried out at room temperature under hydrogen for 2 hours. The reaction solution was filtered, the filtrate was concentrated under reduced pressure, and the residue was chromatographed on a silica gel column to obtain compound 4C (160 mg, yield 69.26%)
Ms m/z(ESI):748.1[M+H]+ Ms m/z(ESI):748.1[M+H] +
第三步:4-(2-{[(2R,7aS)-2-氟-六氢-1H-吡咯嗪-7a-基]甲氧基}-4-[(1R,5S)-3,8-二氮杂双环 [3.2.1]辛烷-3-基]-8-氟喹唑啉-7-基)-5-乙基-6-氟萘-2-醇(化合物4)The third step: 4-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolazin-7a-yl]methoxy}-4-[(1R,5S)-3,8 -Diazabicyclo[3.2.1]octane-3-yl]-8-fluoroquinazolin-7-yl)-5-ethyl-6-fluoronaphthalen-2-ol (compound 4)
4-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolizin-7a-yl]methoxy}-4-[(1R,5S)-3,8-diazabicycl o[3.2.1]octan-3-yl]-8-fluoroquinazolin-7-yl)-5-ethyl-6-fluoronaphthalen-2-ol4-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolizin-7a-yl]methoxy}-4-[(1R,5S)-3,8-diazabicyclo[3.2.1] octan-3-yl]-8-fluoroquinazolin-7-yl)-5-ethyl-6-fluoronaphthalen-2-ol
将4C(0.16mg,0.21mmol)溶解于二氯甲烷8mL中,加入三氟乙酸0.5mL,室温下搅拌1小时,减压浓缩除去残留溶剂,残留物进行制备得化合物4(8mg,收率6.2%)Dissolve 4C (0.16 mg, 0.21 mmol) in 8 mL of dichloromethane, add 0.5 mL of trifluoroacetic acid, stir at room temperature for 1 hour, concentrate under reduced pressure to remove residual solvent, and prepare the residue to obtain compound 4 (8 mg, yield 6.2 %)
制备方法:Preparation:
仪器:SHIMADZU LC-20AP;Instrument: SHIMADZU LC-20AP;
制备柱:Phenomenex C18;Preparative column: Phenomenex C18;
流动相:A为10mmol/L NH4HCO3水溶液;B为乙腈;Mobile phase: A is 10mmol/L NH 4 HCO 3 aqueous solution; B is acetonitrile;
洗脱方法:B从32%to 62%梯度洗脱12分钟;Elution method: B gradient elution from 32% to 62% for 12 minutes;
流速:25mL/min;Flow rate: 25mL/min;
柱温:室温;Column temperature: room temperature;
检测波长:220nm;Detection wavelength: 220nm;
Ms m/z(ESI):604.1[M+1]+ Ms m/z(ESI):604.1[M+1] +
实施例5:5-(4-(1R,5S)-3,8-重氮环氧[3.2.1]-辛烷-3-基)-8-氟-2-(2R,7aS)-2-氟四氢-1H-吡咯烷酮-7a(5H)-甲基)-喹唑啉-7-甲基)-3-氟-2-(三氟甲基)苯胺(化合物5)Example 5: 5-(4-(1R,5S)-3,8-diazoepoxy[3.2.1]-octane-3-yl)-8-fluoro-2-(2R,7aS)-2 -Fluorotetrahydro-1H-pyrrolidone-7a(5H)-methyl)-quinazoline-7-methyl)-3-fluoro-2-(trifluoromethyl)aniline (compound 5)
5-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-3-fluoro-2-(trifluoromethyl)aniline5-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-3-fluoro-2-(trifluoromethyl)aniline
第一步:5-溴-3-氟-2-(三氟甲基)苯胺(5B)The first step: 5-bromo-3-fluoro-2-(trifluoromethyl)aniline (5B)
5-bromo-3-fluoro-2-(trifluoromethyl)aniline5-bromo-3-fluoro-2-(trifluoromethyl)aniline
将5-溴-1,3-二氟-2-(三氟甲基)苯(5A)(2.00g,7.66mmol)和7N的氨甲醇溶液(10mL)依次加入到120mL的压力容器中,在80℃的条件下持续搅拌8h。将反应冷却至室温。减压浓缩除去溶剂,再用水(30mL)/二氯甲烷(40mL)进行萃取并分液,有机层用无水硫酸钠干燥后过滤。减压浓缩有机层得到粗产品(5B)(1.80g,91.04%),无需进一步纯化可直接用于下一步反应。5-Bromo-1,3-difluoro-2-(trifluoromethyl)benzene (5A) (2.00g, 7.66mmol) and 7N ammonia methanol solution (10mL) were sequentially added to a 120mL pressure vessel, and Stirring was continued for 8 h at 80°C. The reaction was cooled to room temperature. Concentrate under reduced pressure to remove the solvent, then extract with water (30 mL)/dichloromethane (40 mL) and separate the layers, and the organic layer is dried over anhydrous sodium sulfate and filtered. The organic layer was concentrated under reduced pressure to obtain the crude product (5B) (1.80 g, 91.04%), which was directly used in the next reaction without further purification.
Ms m/z(ESI):258.1[M+H]+ Ms m/z(ESI):258.1[M+H] +
第二步:3-氟-5-(4,4,5,5-四甲基-1,3,2-二氧硼烷-2-基)-2-(三氟甲基)苯胺(5C)The second step: 3-fluoro-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-2-(trifluoromethyl)aniline (5C )
3-fluoro-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-2-(trifluoromethyl)aniline3-fluoro-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-2-(trifluoromethyl)aniline
在氮气保护下,依次将5B(1.80g,6.98mmol)、联硼酸频那醇酯(2.66g,10.46mmol)、 Pd(dppf)Cl2(0.51g,0.70mmol)和KOAc(2.05g,20.93mmol)加入到50mL的圆底烧瓶中,再加入 1,4-二氧六环(15mL)和水(3mL)的混合溶剂,反应于90℃持续搅拌4h。冷却至室温,减压除去溶剂,加入30mL水,再用乙酸乙酯(20mL×3)萃取,合并有机相后用无水硫酸钠干燥后过滤,减压浓缩滤液,残余物经硅胶柱层析纯化,获得目标化合物5C(1.50g,70.48%)。Under nitrogen protection, 5B (1.80g, 6.98mmol), diboronic acid pinacol ester (2.66g, 10.46mmol), Pd(dppf)Cl 2 (0.51g, 0.70mmol) and KOAc (2.05g, 20.93 mmol) was added to a 50 mL round bottom flask, and then a mixed solvent of 1,4-dioxane (15 mL) and water (3 mL) was added, and the reaction was continuously stirred at 90° C. for 4 h. Cool to room temperature, remove the solvent under reduced pressure, add 30 mL of water, then extract with ethyl acetate (20 mL×3), combine the organic phases, dry with anhydrous sodium sulfate and filter, concentrate the filtrate under reduced pressure, and the residue is subjected to silica gel column chromatography After purification, the target compound 5C (1.50 g, 70.48%) was obtained.
Ms m/z(ESI):306.1[M+H]+ Ms m/z(ESI):306.1[M+H] +
第三步:(1R,5S)-3-(7-(3-氨基-5-氟-4-(三氟甲基)苯基)-8-氟-2-(2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸叔丁酯(5D)The third step: (1R,5S)-3-(7-(3-amino-5-fluoro-4-(trifluoromethyl)phenyl)-8-fluoro-2-(2R,7aS)-2- Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylic acid tert Butyl ester (5D)
tert-butyltert-butyl
(1R,5S)-3-(7-(3-amino-5-fluoro-4-(trifluoromethyl)phenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro -1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate(1R,5S)-3-(7-(3-amino-5-fluoro-4-(trifluoromethyl)phenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
在氮气保护下,将(5C)(0.5g,1.64mmol)溶解于1,4-二氧六环(10mL)和水(2mL)的混合溶液中,然后再依次加入中间体2F(0.65g,1.09mmol)、碳酸铯(0.71g,2.19mmol)和Pd(dppf)Cl2 (0.12g,0.11mmol),110℃的温度反应5h,将反应冷却至室温,加入乙酸乙酯40mL和水25 mL,水相再用乙酸乙酯(40mL×1)萃取一次,合并乙酸乙酯层,乙酸乙酯层用饱和食盐水洗涤一次(40mL×1),无水硫酸钠干燥,减压浓缩后,残余物用硅胶色谱柱柱层析得到化合物(5E) (0.61g,79.27%)Under nitrogen protection, (5C) (0.5g, 1.64mmol) was dissolved in a mixed solution of 1,4-dioxane (10mL) and water (2mL), and then intermediate 2F (0.65g, 1.09mmol), cesium carbonate (0.71g, 2.19mmol) and Pd(dppf)Cl 2 (0.12g, 0.11mmol), react at a temperature of 110°C for 5h, cool the reaction to room temperature, add ethyl acetate 40mL and water 25 mL , the aqueous phase was extracted once more with ethyl acetate (40mL×1), the ethyl acetate layers were combined, the ethyl acetate layer was washed once with saturated brine (40mL×1), dried over anhydrous sodium sulfate, and concentrated under reduced pressure, the residual The compound (5E) (0.61g, 79.27%) was obtained by silica gel column chromatography
Ms m/z(ESI):693.3[M+H]+ Ms m/z(ESI):693.3[M+H] +
第四步:5-(4-(1R,5S)-3,8-二氮杂双环[3.2.1]辛烷-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-3-氟-2-(三氟甲基)苯胺(化合物5)The fourth step: 5-(4-(1R,5S)-3,8-diazabicyclo[3.2.1]octane-3-yl)-8-fluoro-2-(((2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-3-fluoro-2-(trifluoromethyl)aniline (compound 5)
5-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-3-fluoro-2-(trifluoromethyl)aniline5-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-3-fluoro-2-(trifluoromethyl)aniline
将化合物(5D)(0.60g,0.87mmol)溶解于二氯甲烷10mL中,加入三氟乙酸0.5mL,加完后室温反应3h,然后将反应直接减压浓缩除去溶剂后,残余物经过反相柱层析分离纯化得目标化合物5(0.4g,77.93%%)。Dissolve compound (5D) (0.60g, 0.87mmol) in 10mL of dichloromethane, add 0.5mL of trifluoroacetic acid, react at room temperature for 3h after the addition, and then directly concentrate the reaction under reduced pressure to remove the solvent, and the residue is reversed phase The target compound 5 (0.4 g, 77.93%) was obtained by separation and purification by column chromatography.
Ms m/z(ESI):593.3[M+H]+ Ms m/z(ESI):593.3[M+H] +
实施例6:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3-基)-6-氯-8-氟-2-(((2R,7aS)-2-氟四氢-1H- 吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-5-(三氟甲基)苯胺(化合物6)Example 6: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3-yl)-6-chloro-8-fluoro-2-(((2R ,7aS)-2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-(trifluoromethyl)aniline (compound 6)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-6-chloro-8-fluoro-2-(((2R,7aS)-2-fluorotetrah ydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-(trifluoromethyl)aniline3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-6-chloro-8-fluoro-2-(((2R,7aS)-2-fluorotetrah ydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-(trifluoromethyl)aniline
第一步:7-溴-2,4,6-三氯-8-氟喹唑啉(6A)The first step: 7-bromo-2,4,6-trichloro-8-fluoroquinazoline (6A)
7-bromo-2,4,6-trichloro-8-fluoroquinazoline7-bromo-2,4,6-trichloro-8-fluoroquinazoline
将7-溴-6-氯-8-氟喹唑啉-2,4(1H,3H)-二酮(2.0g,6.85mmol)溶解于甲苯30mL中,分别加入三氯氧磷(5.25g,34.24mmol),N,N-二异丙基乙胺(2.65g,20.54mmol),110℃下反应 5h。将反应冷却至室温,减压浓缩后直接得到粗品化合物(6A)并直接用于下一步反应。Dissolve 7-bromo-6-chloro-8-fluoroquinazoline-2,4(1H,3H)-dione (2.0g, 6.85mmol) in toluene 30mL, add phosphorus oxychloride (5.25g, 34.24mmol), N,N-diisopropylethylamine (2.65g, 20.54mmol), reacted at 110°C for 5h. The reaction was cooled to room temperature and concentrated under reduced pressure to directly obtain the crude compound (6A) which was directly used in the next reaction.
第二步:(1R,5S)-3-(7-溴-2,6-二氯-8-氟喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸叔丁酯(6B)The second step: (1R,5S)-3-(7-bromo-2,6-dichloro-8-fluoroquinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane tert-butyl alkane-8-carboxylate (6B)
tert-butyltert-butyl
(1R,5S)-3-(7-bromo-2,6-dichloro-8-fluoroquinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxyl ate(1R,5S)-3-(7-bromo-2,6-dichloro-8-fluoroquinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
将化合物6A溶解于干燥二氯甲烷30mL中,冰浴下缓慢加入N,N-二异丙基乙胺(2.65g, 20.54mmol)和中间体1(1.45g,6.85mmol),加完后缓慢恢复至室温反应2h。减压浓缩后,残留物用硅胶色谱柱柱层析分离纯化得到化合物(6B)(0.88g,两步产率25.50%)。Dissolve compound 6A in 30 mL of dry dichloromethane, slowly add N,N-diisopropylethylamine (2.65 g, 20.54 mmol) and intermediate 1 (1.45 g, 6.85 mmol) under ice-cooling, and slowly add Return to room temperature and react for 2h. After concentration under reduced pressure, the residue was separated and purified by silica gel column chromatography to obtain compound (6B) (0.88 g, two-step yield 25.50%).
LCMS m/z=505.1[M+H]+ LCMS m/z=505.1[M+H] +
第三步:(1R,5S)-3-(2-{[(2R,7aS)-2-氟-六氢-1H-吡咯嗪-7a-基]甲氧基}-7-溴-6-氯-8-氟喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸叔丁酯(6C)The third step: (1R,5S)-3-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolazin-7a-yl]methoxy}-7-bromo-6- Chloro-8-fluoroquinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylic acid tert-butyl ester (6C)
tert-butyltert-butyl
(1R,5S)-3-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolizin-7a-yl]methoxy}-7-bromo-6-chloro-8-flu oroquinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate(1R,5S)-3-(2-{[(2R,7aS)-2-fluoro-hexahydro-1H-pyrrolizin-7a-yl]methoxy}-7-bromo-6-chloro-8-flu oroquinazolin-4 -yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
氮气保护下,将中间体2(0.5g,3.14mmol)溶解于N,N-二甲基甲酰胺15mL中,加入碳酸铯(0.64g,1.98mmol),三乙烯二胺(0.04g,0.35mmol),并加入分子筛500mg,室温搅拌10 min后,缓慢将化合物6B(0.5g,0.99mmol)的N,N-二甲基甲酰胺2mL加入至反应液中,加完后室温反应4h。缓慢加入水40mL,乙酸乙酯(40mL×2)萃取两次,合并乙酸乙酯层,乙酸乙酯层水(40mL×2)洗涤两次,并饱和食盐水洗涤一次(40mL×1),无水硫酸钠干燥,减压浓缩,残留物用硅胶色谱柱柱层析分离纯化得到化合物(6C)(0.46g,产率74.19%)Under nitrogen protection, Intermediate 2 (0.5g, 3.14mmol) was dissolved in N,N-dimethylformamide 15mL, cesium carbonate (0.64g, 1.98mmol), triethylenediamine (0.04g, 0.35mmol) were added ), and 500 mg of molecular sieves were added, and after stirring at room temperature for 10 min, 2 mL of N,N-dimethylformamide of compound 6B (0.5 g, 0.99 mmol) was slowly added to the reaction liquid, and reacted at room temperature for 4 h after the addition. Slowly add 40 mL of water, extract twice with ethyl acetate (40 mL×2), combine the ethyl acetate layers, wash the ethyl acetate layer with water (40 mL×2) twice, and wash once with saturated brine (40 mL×1). Dry over sodium sulfate, concentrate under reduced pressure, and the residue is separated and purified by silica gel column chromatography to obtain compound (6C) (0.46g, yield 74.19%)
LCMS m/z=628.1[M+H]+ LCMS m/z=628.1[M+H] +
第一步:3-(4,4,5,5-四甲基-1,3,2-dioxaborolan-2-yl)-5-(三氟甲基)苯胺(6E)The first step: 3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-5-(trifluoromethyl)aniline (6E)
3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-5-(trifluoromethyl)aniline3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-5-(trifluoromethyl)aniline
氮气保护下,将3-溴-5-(三氟甲基)苯胺(6D)(2g,8.33mmol),联硼酸频那醇酯(3.17 g,12.50mmol),醋酸钾(3.27g,33.32mmol),[1,1'-双(二苯基膦)二茂铁]二氯化钯二氯甲烷络合物(680mg,0.83mmol)置于封管中,加入15mL 1,4-二氧六环,升温至90℃,反应5小时。将反应冷却至室温,然后加入水,乙酸乙酯萃取三次。合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物(6E)(2g,产率84%)。Under nitrogen protection, 3-bromo-5-(trifluoromethyl)aniline (6D) (2g, 8.33mmol), pinacol diboronate (3.17 g, 12.50mmol), potassium acetate (3.27g, 33.32mmol ), [1,1'-bis(diphenylphosphino)ferrocene]dichloropalladium dichloromethane complex (680mg, 0.83mmol) was placed in a sealed tube, and 15mL of 1,4-dioxane was added ring, heated to 90°C, and reacted for 5 hours. The reaction was cooled to room temperature, then water was added and extracted three times with ethyl acetate. The organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound (6E) (2 g, yield 84%).
LCMS m/z=288.1[M+H]+ LCMS m/z=288.1[M+H] +
第二步:(1R,5S)-3-(7-(3-氨基-5-(三氟甲基)苯基)-6-氯-8-氟-2-(((2R,7aS)-2-氟四氢-1H)-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸叔丁酯(6F)The second step: (1R,5S)-3-(7-(3-amino-5-(trifluoromethyl)phenyl)-6-chloro-8-fluoro-2-(((2R,7aS)- 2-Fluorotetrahydro-1H)-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8- tert-butyl carboxylate (6F)
tert-butyltert-butyl
(1R,5S)-3-(7-(3-amino-5-(trifluoromethyl)phenyl)-6-chloro-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro -1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate(1R,5S)-3-(7-(3-amino-5-(trifluoromethyl)phenyl)-6-chloro-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
氮气保护下,将中间体6C(150mg,0.24mmol),6E(76mg,0.26mmol),[1,1'-双 (二苯基膦)二茂铁]二氯化钯二氯甲烷络合物(21mg,0.024mmol)和碳酸铯(160mg,0.48mmol)置于封管中,加入4.5mL1,4-二氧六环和1mL水。升温至105℃反应3小时。将反应冷却至室温,加水稀释,乙酸乙酯萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后残余物经过柱层析纯化,即得到化合物(6F)(118mg,产率69%)Under nitrogen protection, the intermediate 6C (150mg, 0.24mmol), 6E (76mg, 0.26mmol), [1,1'-bis(diphenylphosphino)ferrocene]dichloropalladium dichloromethane complex (21mg, 0.024mmol) and cesium carbonate (160mg, 0.48mmol) were placed in a sealed tube, and 4.5mL of 1,4-dioxane and 1mL of water were added. The temperature was raised to 105° C. for 3 hours. The reaction was cooled to room temperature, diluted with water, extracted three times with ethyl acetate, the organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure, and the residue was purified by column chromatography to obtain compound (6F) (118 mg, yield 69%)
LCMS m/z=709.3[M+H]+ LCMS m/z=709.3[M+H] +
第三步:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3-基)-6-氯-8-氟-2-(((2R,7aS)-2-氟四氢-1H- 吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-5-(三氟甲基)苯胺(化合物6)The third step: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3-yl)-6-chloro-8-fluoro-2-(((2R ,7aS)-2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-(trifluoromethyl)aniline (compound 6)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-6-chloro-8-fluoro-2-(((2R,7aS)-2-fluorotetrah ydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-(trifluoromethyl)aniline3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-6-chloro-8-fluoro-2-(((2R,7aS)-2-fluorotetrah ydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-(trifluoromethyl)aniline
将化合物(6F)(118mg,0.17mmol)溶解于二氯甲烷2.5mL中,然后滴入0.5mL三氟乙酸,室温下反应40分钟。减压浓缩反应液,然后加入二氯甲烷溶解,滴入0.5mL N,N-二异丙基乙胺后,减压浓缩,残留物经制备HPLC纯化,制备液浓缩后冻干得到化合物6(55mg,产率53%)。Compound (6F) (118 mg, 0.17 mmol) was dissolved in 2.5 mL of dichloromethane, then 0.5 mL of trifluoroacetic acid was added dropwise, and reacted at room temperature for 40 minutes. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 0.5mL N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, the residue is purified by preparative HPLC, and the preparation solution is concentrated and freeze-dried to obtain compound 6 ( 55 mg, yield 53%).
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH 4 HCO 3 ); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min .
LCMS m/z=609.2[M+H]+ LCMS m/z=609.2[M+H] +
实施例7:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-5-(三氟甲基)苯胺(化合物7)Example 7: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3-yl)-8-fluoro-2-(((2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-(trifluoromethyl)aniline (compound 7)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-(trifluoromethyl)aniline3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-5-(trifluoromethyl)aniline
第一步:(1R,5S)-3-(7-(3-氨-5-(三氟甲基)苯基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a) (5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸叔丁酯(7A)The first step: (1R,5S)-3-(7-(3-amino-5-(trifluoromethyl)phenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetra Hydrogen-1H-pyrrolazin-7a) (5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylic acid tert-butyl Esters (7A)
tert-butyltert-butyl
(1R,5S)-3-(7-(3-amino-5-(trifluoromethyl)phenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrr olizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate(1R,5S)-3-(7-(3-amino-5-(trifluoromethyl)phenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrr olizin-7a(5H )-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
氮气保护下,将中间体2F(110mg,0.19mmol),化合物6E(107mg,0.37mmol),[1,1'-双(二苯基膦)二茂铁]二氯化钯二氯甲烷络合物(16mg,0.019mmol)和碳酸铯(124mg,0.38mmol)置于封管中,加入3mL1,4-二氧六环和0.6mL水,升温至105℃反应3小时。将反应冷却至室温,加水稀释,乙酸乙酯萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后,残余物经过柱层析纯化,即得到化合物(7A)(115mg,产率90%)Under nitrogen protection, intermediate 2F (110mg, 0.19mmol), compound 6E (107mg, 0.37mmol), [1,1'-bis(diphenylphosphino)ferrocene]dichloropalladium dichloromethane complexed (16mg, 0.019mmol) and cesium carbonate (124mg, 0.38mmol) were placed in a sealed tube, 3mL of 1,4-dioxane and 0.6mL of water were added, and the temperature was raised to 105°C for 3 hours. The reaction was cooled to room temperature, diluted with water, extracted three times with ethyl acetate, the organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure, and the residue was purified by column chromatography to obtain compound (7A) (115mg, product rate 90%)
LCMS m/z=675.1[M+H]+ LCMS m/z=675.1[M+H] +
第二步:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-5-(三氟甲基)苯胺(化合物7)The second step: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3-yl)-8-fluoro-2-(((2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-(trifluoromethyl)aniline (compound 7)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-(trifluoromethyl)aniline3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-5-(trifluoromethyl)aniline
将化合物7A(115mg,0.17mmol)溶解于二氯甲烷2.5mL中,然后滴入0.5mL三氟乙酸,室温下反应40分钟。减压浓缩反应液,然后加入二氯甲烷溶解,滴入0.5mL N,N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物7(45mg,产率46%),为白色粉末。Compound 7A (115 mg, 0.17 mmol) was dissolved in 2.5 mL of dichloromethane, then 0.5 mL of trifluoroacetic acid was added dropwise, and reacted at room temperature for 40 minutes. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 0.5mL N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, the residue is prepared and purified by HPLC, the preparation solution is concentrated and freeze-dried to obtain compound 7 ( 45 mg, yield 46%), as a white powder.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH 4 HCO 3 ); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min .
LCMS m/z=575.3[M+H]+ LCMS m/z=575.3[M+H] +
实施例8:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3基)-6-氯-8-氟-2-(((2R,7aS)-2-氟四氢-1H- 吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-4-(三氟甲基)苯胺(化合物8)Example 8: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3yl)-6-chloro-8-fluoro-2-(((2R, 7aS)-2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)aniline (compound 8)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-6-chloro-8-fluoro-2-(((2R,7aS)-2-fluorotetrah ydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)aniline3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-6-chloro-8-fluoro-2-(((2R,7aS)-2-fluorotetrah ydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)aniline
第一步:(1R,5S)-3-(7-(5-胺基-2-(三氟甲基)苯基)-6-氯-8-氟-2-(((2R,7aS)-2-氟四氢-1H)-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸叔丁酯(8B)The first step: (1R,5S)-3-(7-(5-amino-2-(trifluoromethyl)phenyl)-6-chloro-8-fluoro-2-(((2R,7aS) -2-Fluorotetrahydro-1H)-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8 - tert-butyl carboxylate (8B)
tert-butyltert-butyl
(1R,5S)-3-(7-(5-amino-2-(trifluoromethyl)phenyl)-6-chloro-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro -1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate(1R,5S)-3-(7-(5-amino-2-(trifluoromethyl)phenyl)-6-chloro-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
氮气保护下,将化合物6C(246mg,0.39mmol),化合物5-氨基-2-(三氟甲基)苯基)硼酸(8A) (89mg,0.43mmol),三(二亚苄基丙酮)二钯(36mg,0.039mmol),2-二环己基磷-2',6'-二异丙氧基-1,1'-联苯(37mg,0.08mmol),四丁基溴化铵(13mg,0.039mmol)和碳酸钠(83mg, 0.78mmol)置于封管中,加入4mL 1,4-二氧六环和1mL水,升温至105℃反应3小时。将反应冷却至室温,加水稀释,乙酸乙酯萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后,残余物经过柱层析纯化,即得到化合物(8B)(76mg,产率27%)Under nitrogen protection, compound 6C (246mg, 0.39mmol), compound 5-amino-2-(trifluoromethyl)phenyl)boronic acid (8A) (89mg, 0.43mmol), tris(dibenzylideneacetone) di Palladium (36mg, 0.039mmol), 2-dicyclohexylphosphonium-2',6'-diisopropoxy-1,1'-biphenyl (37mg, 0.08mmol), tetrabutylammonium bromide (13mg, 0.039mmol) and sodium carbonate (83mg, 0.78mmol) were placed in a sealed tube, 4mL 1,4-dioxane and 1mL water were added, and the temperature was raised to 105°C for 3 hours. The reaction was cooled to room temperature, diluted with water, extracted three times with ethyl acetate, the organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure, and the residue was purified by column chromatography to obtain compound (8B) (76 mg, yield rate 27%)
LCMS m/z=709.3[M+H]+ LCMS m/z=709.3[M+H] +
第三步:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3基)-6-氯-8-氟-2-(((2R,7aS)-2-氟四氢-1H- 吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-4-(三氟甲基)苯胺(化合物8)The third step: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3 base)-6-chloro-8-fluoro-2-(((2R, 7aS)-2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)aniline (compound 8)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-6-chloro-8-fluoro-2-(((2R,7aS)-2-fluorotetrah ydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)aniline3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-6-chloro-8-fluoro-2-(((2R,7aS)-2-fluorotetrah ydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)aniline
将化合物8B(76mg,0.11mmol)溶解于二氯甲烷2.5mL中,然后滴入0.5mL三氟乙酸,室温下反应40分钟。减压浓缩反应液,然后加入二氯甲烷溶解,滴入0.5mL N,N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物8(12mg,产率17.9%)。Compound 8B (76 mg, 0.11 mmol) was dissolved in 2.5 mL of dichloromethane, then 0.5 mL of trifluoroacetic acid was added dropwise, and reacted at room temperature for 40 minutes. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 0.5mL N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, the residue is prepared and purified by HPLC, the preparation solution is concentrated and freeze-dried to obtain compound 8 ( 12 mg, yield 17.9%).
HPLC制备方法:HPLC preparation method:
仪器:SHIMADZU LC-20AP;Instrument: SHIMADZU LC-20AP;
色谱柱:Phenomenex C18;Chromatographic column: Phenomenex C18;
流动相:A为10mmol NH4HCO3水溶液;B为乙腈;Mobile phase: A is 10mmol NH 4 HCO 3 aqueous solution; B is acetonitrile;
洗脱方法:B的30%至60%A溶液梯度洗脱12分钟;Elution method: gradient elution of 30% to 60% A solution of B for 12 minutes;
流速:25mL/分钟;Flow rate: 25mL/min;
柱温:室温;Column temperature: room temperature;
检测波长:220nm;Detection wavelength: 220nm;
LCMS m/z=609.2[M+H]+ LCMS m/z=609.2[M+H] +
实施例9:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛基-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H- 吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-2-氯-5-氟苯胺(化合物9)Example 9: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octyl-3-yl)-8-fluoro-2-(((2R,7aS) -2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-2-chloro-5-fluoroaniline (compound 9)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-2-chloro-5-fluoroaniline3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-2-chloro-5-fluoroaniline
第一步:3-溴-2-氯-5-氟苯胺(9A-1)The first step: 3-bromo-2-chloro-5-fluoroaniline (9A-1)
3-bromo-2-chloro-5-fluoroaniline3-bromo-2-chloro-5-fluoroaniline
3-溴-4-氯-5-氟苯胺(9A-2)3-Bromo-4-chloro-5-fluoroaniline (9A-2)
3-bromo-4-chloro-5-fluoroaniline3-bromo-4-chloro-5-fluoroaniline
将原料3-氟-5-溴苯胺(15g,78.94mmol)溶于200ml N,N-二甲基甲酰胺中,冰浴下分批加入N-氯代丁二酰亚胺(11g,82.89mmol),室温搅拌16h,加水淬灭反应,加入200ml乙酸乙酯萃取一次,有机相用水洗涤2次(150ml×2),用150ml饱和食盐水洗涤一次,有机相用无水硫酸钠干燥,减压浓缩,残余物经柱层析分析纯化得到化合物9A-1(5g,产率28%),化合物9A-2(8g,产率45%)The raw material 3-fluoro-5-bromoaniline (15g, 78.94mmol) was dissolved in 200ml N,N-dimethylformamide, and N-chlorosuccinimide (11g, 82.89mmol ), stirred at room temperature for 16 hours, added water to quench the reaction, added 200ml ethyl acetate to extract once, the organic phase was washed twice with water (150ml×2), washed once with 150ml saturated saline, the organic phase was dried with anhydrous sodium sulfate, and reduced pressure Concentration, the residue was analyzed and purified by column chromatography to obtain compound 9A-1 (5g, yield 28%), compound 9A-2 (8g, yield 45%)
LCMS m/z=224.0[M+H]+ LCMS m/z=224.0[M+H] +
1H NMR(400MHz,DMSO-d6)δ6.79(dd,1H),6.61(dd,1H),5.99(br,2H).(9A-1) 1 H NMR (400MHz,DMSO-d 6 )δ6.79(dd,1H),6.61(dd,1H),5.99(br,2H).(9A-1)
1H NMR(400MHz,DMSO-d6)δ6.78(dd,1H),6.52(dd,1H),5.81(br,2H).(9A-2) 1 H NMR (400MHz,DMSO-d 6 )δ6.78(dd,1H),6.52(dd,1H),5.81(br,2H).(9A-2)
第二步:(4-((1R,5S)-8-(叔丁氧基羰基)-3,8-二氮杂双环[3.2.1]辛烷-3-基)-8-氟-2-(((2R,7aS)-2 -氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)硼酸(9B)The second step: (4-((1R,5S)-8-(tert-butoxycarbonyl)-3,8-diazabicyclo[3.2.1]octane-3-yl)-8-fluoro-2 -(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)boronic acid (9B)
(4-((1R,5S)-8-(tert-butoxycarbonyl)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2 -fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)boronic acid(4-((1R,5S)-8-(tert-butoxycarbonyl)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2 -fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)boronic acid
将(2F)(1.2g,2.02mmol)溶解于1,4-二氧六环15mL和水3mL的混合溶液中,加入联硼酸频那醇酯(0.62g,2.42mmol),乙酸钾(0.4g,4.04mmol),Pd(dppf)Cl2(0.15g,0.2mmol),氮气保护下100℃下反应3h,将反应冷却至室温,垫硅藻土过滤,滤液减压浓缩,反相柱层析得到化合物9B(1g,产率88.5%)Dissolve (2F) (1.2g, 2.02mmol) in a mixed solution of 1,4-dioxane 15mL and water 3mL, add biboronic acid pinacol ester (0.62g, 2.42mmol), potassium acetate (0.4g , 4.04mmol), Pd(dppf)Cl 2 (0.15g, 0.2mmol), reacted at 100°C for 3h under the protection of nitrogen, cooled the reaction to room temperature, filtered it with diatomaceous earth, concentrated the filtrate under reduced pressure, reversed phase column chromatography Obtained compound 9B (1 g, yield 88.5%)
LCMS m/z=560.2[M+H]+ LCMS m/z=560.2[M+H] +
第三步:叔丁基(1R,5S)-3-(7-(3-氨基-2-氯-5-氟苯基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸酯(9C)The third step: tert-butyl (1R,5S)-3-(7-(3-amino-2-chloro-5-fluorophenyl)-8-fluoro-2-(((2R,7aS)-2- Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate (9C)
tert-butyltert-butyl
(1R,5S)-3-(7-(3-amino-2-chloro-5-fluorophenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrol izin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate(1R,5S)-3-(7-(3-amino-2-chloro-5-fluorophenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrol izin-7a( 5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
将(9A-1)(0.11g,0.49mmol)溶解于1,4-二氧六环5mL和水1mL的混合溶液中,加入9B(0.22g,0.39mmol),碳酸铯(0.32g,0.98mmol),Pd(dppf)Cl2(0.036g,0.049mmol),氮气保护下100℃下反应2h,将反应冷却至室温,加入乙酸乙酯40mL和水25mL,水相再用乙酸乙酯(40mL×1)萃取一次,合并乙酸乙酯层,乙酸乙酯层用饱和食盐水洗涤一次(40mL×1),无水硫酸钠干燥,减压浓缩后,残留物用硅胶色谱柱柱层析得到化合物(9C)(0.13g,产率40.25%)Dissolve (9A-1) (0.11g, 0.49mmol) in a mixed solution of 1,4-dioxane 5mL and water 1mL, add 9B (0.22g, 0.39mmol), cesium carbonate (0.32g, 0.98mmol ), Pd(dppf)Cl 2 (0.036g, 0.049mmol), reacted at 100°C under nitrogen protection for 2h, cooled the reaction to room temperature, added 40mL of ethyl acetate and 25mL of water, and then used ethyl acetate (40mL× 1) Extract once, combine the ethyl acetate layers, wash the ethyl acetate layer once with saturated brine (40mL×1), dry over anhydrous sodium sulfate, and concentrate under reduced pressure, the residue is chromatographed on a silica gel column to obtain the compound ( 9C) (0.13g, 40.25% yield)
LCMS m/z=659.2[M+H]+ LCMS m/z=659.2[M+H] +
第四步:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛基-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-2-氯-5-氟苯胺(化合物9)The fourth step: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octyl-3-yl)-8-fluoro-2-(((2R,7aS) -2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-2-chloro-5-fluoroaniline (compound 9)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-2-chloro-5-fluoroaniline3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-2-chloro-5-fluoroaniline
将化合物(9C)(0.13g,0.87mmol)(0.13g,0.87mmol)溶解于二氯甲烷5mL中,加入三氟乙酸0.5mL,室温下反应40分钟。减压浓缩反应液,然后加入二氯甲烷溶解,滴入0.5mLN, N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物9(20 mg,产率20%),为白色粉末。Compound (9C) (0.13 g, 0.87 mmol) (0.13 g, 0.87 mmol) was dissolved in 5 mL of dichloromethane, 0.5 mL of trifluoroacetic acid was added, and the reaction was carried out at room temperature for 40 minutes. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve, add dropwise 0.5mL N, N-diisopropylethylamine, concentrate under reduced pressure, the residue is prepared and purified by HPLC, the preparation solution is concentrated and freeze-dried to obtain compound 9 (20 mg, yield 20%), white powder.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH 4 HCO 3 ); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min
Ms m/z(ESI):559.2[M+H]+ Ms m/z(ESI):559.2[M+H] +
实施例10:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛基-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H- 吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-4-氯-5-氟苯胺(化合物10)Example 10: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octyl-3-yl)-8-fluoro-2-(((2R,7aS) -2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-chloro-5-fluoroaniline (compound 10)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-chloro-5-fluoroaniline3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-4-chloro-5-fluoroaniline
第一步:叔丁基(1R,5S)-3-(7-(5-氨基-2-氯-3-氟苯基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-)7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸酯(10A)The first step: tert-butyl (1R,5S)-3-(7-(5-amino-2-chloro-3-fluorophenyl)-8-fluoro-2-(((2R,7aS)-2- Fluorotetrahydro-1H-pyrrolizine-)7a(5H-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylic acid Esters (10A)
tert-butyltert-butyl
(1R,5S)-3-(7-(5-amino-2-chloro-3-fluorophenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrol izin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate(1R,5S)-3-(7-(5-amino-2-chloro-3-fluorophenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrol izin-7a( 5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
将(9A-2)(0.11g,0.49mmol)溶解于1,4-二氧六环5mL和水1mL的混合溶液中,加入9B(0.22g,0.39mmol),碳酸铯(0.32g,0.98mmol),Pd(dppf)Cl2(0.036g,0.049mmol),氮气保护下100℃下反应2h,将反应冷却至室温,加入乙酸乙酯40mL和水25mL搅拌后分液, 水相再用乙酸乙酯(40mL×1)萃取一次,合并乙酸乙酯层,乙酸乙酯层用饱和食盐水洗涤一次(40mL×1),无水硫酸钠干燥,减压浓缩后,残留物用硅胶色谱柱柱层析得到化合物(10A)(0.13 g,产率40.25%)Dissolve (9A-2) (0.11g, 0.49mmol) in a mixed solution of 1,4-dioxane 5mL and water 1mL, add 9B (0.22g, 0.39mmol), cesium carbonate (0.32g, 0.98mmol ), Pd(dppf)Cl 2 (0.036g, 0.049mmol), react at 100°C for 2h under the protection of nitrogen, cool the reaction to room temperature, add 40mL of ethyl acetate and 25mL of water and stir to separate the liquids, and the aqueous phase is then washed with ethyl acetate Ethyl acetate (40mL×1) was extracted once, the ethyl acetate layers were combined, the ethyl acetate layer was washed once with saturated brine (40mL×1), dried over anhydrous sodium sulfate, concentrated under reduced pressure, and the residue was subjected to silica gel column chromatography Compound (10A) (0.13 g, yield 40.25%) was obtained by analysis
LCMS m/z=659.2[M+H]+LCMS m/z=659.2[M+H]+
第二步:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛基-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-4-氯-5-氟苯胺(化合物10)The second step: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octyl-3-yl)-8-fluoro-2-(((2R,7aS) -2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-chloro-5-fluoroaniline (compound 10)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-chloro-5-fluoroaniline3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-4-chloro-5-fluoroaniline
将化合物(10A)(0.13g,0.87mmol)溶解于二氯甲烷5mL中,加入三氟乙酸0.5mL,室温下反应40分钟。减压浓缩反应液,然后加入二氯甲烷溶解,滴入0.5mL N,N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物10(20mg,产率20%),为白色粉末。Compound (10A) (0.13 g, 0.87 mmol) was dissolved in 5 mL of dichloromethane, 0.5 mL of trifluoroacetic acid was added, and the reaction was carried out at room temperature for 40 minutes. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 0.5mL N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, the residue is prepared and purified by HPLC, the preparation solution is concentrated and freeze-dried to obtain compound 10( 20mg, yield 20%), as a white powder.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH 4 HCO 3 ); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min .
Ms m/z(ESI):559.2[M+H]+ Ms m/z(ESI):559.2[M+H] +
实施例11:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛基-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H- 吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-2-氯-4,5,6-三氟苯胺(化合物11)Example 11: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octyl-3-yl)-8-fluoro-2-(((2R,7aS) -2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-2-chloro-4,5,6-trifluoroaniline (compound 11)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-2-chloro-4,5,6-trifluoroaniline3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-2-chloro-4,5,6-trifluoroaniline
第一步:2,3,4-三氟-5-碘苯胺(11A)The first step: 2,3,4-trifluoro-5-iodoaniline (11A)
2,3,4-trifluoro-5-iodoaniline2,3,4-trifluoro-5-iodoaniline
将原料2,3,4-三氟苯胺(1.0g,6.8mmol)溶于5ml三氟甲磺酸中,加入NIS(2.3g,10.2mmol), 室温搅拌5h,加入30ml水稀释,用30ml乙酸乙酯萃取一次,无水硫酸钠干燥,减压浓缩,柱层析得到化合物11A(1.2g,产率64.6%)。Dissolve the raw material 2,3,4-trifluoroaniline (1.0g, 6.8mmol) in 5ml of trifluoromethanesulfonic acid, add NIS (2.3g, 10.2mmol), stir at room temperature for 5h, add 30ml of water to dilute, and dilute with 30ml of acetic acid Extracted once with ethyl ester, dried over anhydrous sodium sulfate, concentrated under reduced pressure, and column chromatography gave compound 11A (1.2 g, yield 64.6%).
第二步:2-氯-4,5,6-三氟-3-碘苯胺(11B)The second step: 2-chloro-4,5,6-trifluoro-3-iodoaniline (11B)
2-chloro-4,5,6-trifluoro-3-iodoaniline2-chloro-4,5,6-trifluoro-3-iodoaniline
将原料11A(0.3g,1.1mmol)溶于5ml乙酸中,加入NCS(0.16g,1.21mmol),室温搅拌5h,加入30ml水稀释,用30ml乙酸乙酯萃取一次,无水硫酸钠干燥,减压浓缩,柱层析得到化合物11B(0.16g,产率47.3%)。Dissolve raw material 11A (0.3g, 1.1mmol) in 5ml of acetic acid, add NCS (0.16g, 1.21mmol), stir at room temperature for 5h, add 30ml of water to dilute, extract once with 30ml of ethyl acetate, dry over anhydrous sodium sulfate, reduce Concentrate under reduced pressure, and obtain compound 11B (0.16 g, yield 47.3%) by column chromatography.
第三步:叔丁基(1R,5S)-3-(7-(3-氨基-2-氯-4,5,6-三氟苯基)-8-氟-2-((((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸酯(11C)The third step: tert-butyl (1R,5S)-3-(7-(3-amino-2-chloro-4,5,6-trifluorophenyl)-8-fluoro-2-((((2R ,7aS)-2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane -8-Carboxylate (11C)
tert-butyltert-butyl
(1R,5S)-3-(7-(3-amino-2-chloro-4,5,6-trifluorophenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate(1R,5S)-3-(7-(3-amino-2-chloro-4,5,6-trifluorophenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
将(11B)(0.16g,0.52mmol)溶解于1,4-二氧六环5mL和水1mL的混合溶液中,加入9B(0.23g,0.42mmol),碳酸铯(0.3g,1.04mmol),Pd(dppf)Cl2(0.038g,0.052mmol),氮气保护下100℃下反应2h,将反应冷却至室温,加入乙酸乙酯40mL和水25mL,水相再用乙酸乙酯(40mL×1)萃取一次,合并乙酸乙酯层,乙酸乙酯层用饱和食盐水洗涤一次(40mL×1),无水硫酸钠干燥,减压浓缩后,残留物用硅胶色谱柱柱层析得到化合物(11C)(0.11g,产率30.4%)Dissolve (11B) (0.16g, 0.52mmol) in a mixed solution of 1,4-dioxane 5mL and water 1mL, add 9B (0.23g, 0.42mmol), cesium carbonate (0.3g, 1.04mmol), Pd(dppf)Cl 2 (0.038g, 0.052mmol), reacted at 100°C under nitrogen protection for 2h, cooled the reaction to room temperature, added 40mL of ethyl acetate and 25mL of water, and then used ethyl acetate (40mL×1) for the aqueous phase Extract once, combine the ethyl acetate layers, wash the ethyl acetate layer once with saturated brine (40mL×1), dry over anhydrous sodium sulfate, concentrate under reduced pressure, and the residue is chromatographed on a silica gel column to obtain compound (11C) (0.11g, yield 30.4%)
LCMS m/z=695.2[M+H]+ LCMS m/z=695.2[M+H] +
第四步:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛基-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-2-氯-4,5,6-三氟苯胺(化合物11)The fourth step: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octyl-3-yl)-8-fluoro-2-(((2R,7aS) -2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-2-chloro-4,5,6-trifluoroaniline (compound 11)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-2-chloro-4,5,6-trifluoroaniline3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-2-chloro-4,5,6-trifluoroaniline
将化合物(11C)(0.11g,0.16mmol)溶解于二氯甲烷5mL中,加入三氟乙酸0.5mL,室温下反应40分钟。减压浓缩反应液,然后加入二氯甲烷溶解,滴入0.5mL N,N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物11(20mg,产率21%),为白色粉末。Compound (11C) (0.11 g, 0.16 mmol) was dissolved in 5 mL of dichloromethane, 0.5 mL of trifluoroacetic acid was added, and the reaction was carried out at room temperature for 40 minutes. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 0.5mL N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, the residue is prepared and purified by HPLC, the preparation solution is concentrated and freeze-dried to obtain compound 11( 20 mg, yield 21%), as a white powder.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH 4 HCO 3 ); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min .
Ms m/z(ESI):595.2[M+H]+ Ms m/z(ESI):595.2[M+H] +
实施例12:3-(4-((1R,5S)-3,8-二氮杂环[3.2.1]辛基-3-基)-8-氟-2-((2R,7aS)-2- 氟四氢-1H-吡咯啉-7a(5H)-基甲氧基)喹唑啉-7-基)-4-(三氟甲基)苯胺(化合物12)Example 12: 3-(4-((1R,5S)-3,8-diazacyclo[3.2.1]octyl-3-yl)-8-fluoro-2-((2R,7aS)- 2-Fluorotetrahydro-1H-pyrroline-7a(5H)-ylmethoxy)quinazolin-7-yl)-4-(trifluoromethyl)aniline (compound 12)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)aniline3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)aniline
第一步:叔丁基(1R,5S)-3-(7-(5-氨基-2-(三氟甲基)苯基)-8-氟-2-((2R,7aS)-2-氟四氢-1H-吡咯里嗪-7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8- 羧酸酯(12B)The first step: tert-butyl (1R, 5S)-3-(7-(5-amino-2-(trifluoromethyl)phenyl)-8-fluoro-2-((2R, 7aS)-2- Fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylic acid Esters (12B)
tert-butyl(1R,5S)-3-(7-(5-amino-2-(trifluoromethyl)phenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetra hydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxyla tetert-butyl(1R,5S)-3-(7-(5-amino-2-(trifluoromethyl)phenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetra hydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxyla te
将中间体2F(160mg,0.27mmol),12A(120mg,0.41mmol),Pd(dppf)Cl2·DCM(22mg,0.03mmol)和碳酸铯(260mg,0.81mmol)加入到1,4-二氧六环(8mL)和水(2mL)的混合溶剂中。在氮气保护下,反应于105℃搅拌5小时。待反应也冷却至室温后,减压除去溶剂,加入100mL水,再用乙酸乙酯(50mL×3)萃取,合并有机相后用无水硫酸钠干燥后过滤,旋干,残余物经硅胶柱层析分离纯化得到12B(140mg,收率:76.85%)。Intermediate 2F (160 mg, 0.27 mmol), 12A (120 mg, 0.41 mmol), Pd(dppf)Cl 2 ·DCM (22 mg, 0.03 mmol) and cesium carbonate (260 mg, 0.81 mmol) were added to 1,4-dioxo In a mixed solvent of hexacyclic (8mL) and water (2mL). Under a nitrogen blanket, the reaction was stirred at 105°C for 5 hours. After the reaction was cooled to room temperature, the solvent was removed under reduced pressure, 100mL of water was added, and then extracted with ethyl acetate (50mL×3). Chromatographic separation and purification gave 12B (140 mg, yield: 76.85%).
Ms m/z(ESI):675.4[M+H]+ Ms m/z(ESI):675.4[M+H] +
第二步:3-(4-((1R,5S)-3,8-二氮杂环[3.2.1]辛基-3-基)-8-氟-2-((2R,7aS)-2-氟四氢-1H-吡咯啉-7a(5H)-基甲氧基)喹唑啉-7-基)-4-(三氟甲基)苯胺(化合物12)The second step: 3-(4-((1R, 5S)-3,8-diazacyclo[3.2.1]octyl-3-yl)-8-fluoro-2-((2R, 7aS)- 2-Fluorotetrahydro-1H-pyrroline-7a(5H)-ylmethoxy)quinazolin-7-yl)-4-(trifluoromethyl)aniline (compound 12)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)anne3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)anne
将12B(140mg,0.21mmol)溶于二氯甲烷(10mL)中,然后于室温加入3mL TFA,反应继续再室温搅拌1小时。减压除去多余的酸和溶剂,经过制备HPLC纯化得目标化合物 12(40mg,收率:33.15%)。12B (140 mg, 0.21 mmol) was dissolved in dichloromethane (10 mL), then 3 mL of TFA was added at room temperature, and the reaction was continued to stir at room temperature for another 1 hour. The excess acid and solvent were removed under reduced pressure, and the target compound 12 (40 mg, yield: 33.15%) was purified by preparative HPLC.
Ms m/z(ESI):575.2[M+H]+ Ms m/z(ESI):575.2[M+H] +
实施例13:3-(4-((1R,5S)-3,8-二氮杂环[3.2.1]辛基-3-基)-8-氟-2-((2R,7aS)-2- 氟四氢-1H-吡咯啉-7a(5H)-基)甲氧基)喹唑啉-7-基)-4-(三氟甲氧基)苯胺(化合物13)Example 13: 3-(4-((1R,5S)-3,8-diazacyclo[3.2.1]octyl-3-yl)-8-fluoro-2-((2R,7aS)- 2-Fluorotetrahydro-1H-pyrroline-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethoxy)aniline (compound 13)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethoxy)aniline3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethoxy)aniline
第一步:叔丁基(1R,5S)-3-(7-(5-氨基-2-(三氟甲氧基)苯基)-8-氟-2-((2R,7aS) -2-氟四氢-1H-吡咯里嗪-7a(5H)-基甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸酯(13B)The first step: tert-butyl (1R, 5S)-3-(7-(5-amino-2-(trifluoromethoxy)phenyl)-8-fluoro-2-((2R, 7aS)-2 -Fluorotetrahydro-1H-pyrrolizin-7a(5H)-ylmethoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylic acid Esters (13B)
tert-butyl(1R,5S)-3-(7-(5-amino-2-(trifluoromethoxy)phenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetr ahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxyl atetert-butyl(1R,5S)-3-(7-(5-amino-2-(trifluoromethoxy)phenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetr ahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
将中间体2F(150mg,0.25mmol),13A(110mg,0.38mmol),Pd(dppf)Cl2·DCM(20mg,0.025mmol)和碳酸铯(240mg,0.75mmol)加入到1,4-二氧六环(10mL)和水(2mL) 的混合溶剂中。在氮气保护下,反应于105℃搅拌3小时。待反应也冷却至室温后,减压除去溶剂,加入100mL水,再用乙酸乙酯(50mL×3)萃取,合并有机相后用无水硫酸钠干燥后过滤,旋干,残余物经硅胶柱层析分离纯化得到13B(140mg,收率:81.08%)。Intermediate 2F (150 mg, 0.25 mmol), 13A (110 mg, 0.38 mmol), Pd(dppf)Cl 2 ·DCM (20 mg, 0.025 mmol) and cesium carbonate (240 mg, 0.75 mmol) were added to 1,4-dioxo Hexacyclic (10mL) and water (2mL) mixed solvent. Under a nitrogen blanket, the reaction was stirred at 105°C for 3 hours. After the reaction was cooled to room temperature, the solvent was removed under reduced pressure, 100mL of water was added, and then extracted with ethyl acetate (50mL×3). Chromatographic separation and purification gave 13B (140 mg, yield: 81.08%).
Ms m/z(ESI):691.3[M+H]+ Ms m/z(ESI):691.3[M+H] +
第二步:3-(4-((1R,5S)-3,8-二氮杂环[3.2.1]辛基-3-基)-8-氟-2-((2R,7aS)-2-氟四氢-1H-吡咯啉-7a(5H)-基)甲氧基)喹唑啉-7-基)-4-(三氟甲氧基)苯胺(化合物13)The second step: 3-(4-((1R, 5S)-3,8-diazacyclo[3.2.1]octyl-3-yl)-8-fluoro-2-((2R, 7aS)- 2-Fluorotetrahydro-1H-pyrroline-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethoxy)aniline (compound 13)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethoxy)aniline3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethoxy)aniline
将13b(140mg,0.20mmol)溶于二氯甲烷(8mL)中,然后于室温加入2mL TFA,反应继续再室温搅拌1小时。减压除去多余的酸和溶剂,经过制备HPLC纯化得目标化合物13(20mg,收率:16.93%)。13b (140 mg, 0.20 mmol) was dissolved in dichloromethane (8 mL), then 2 mL of TFA was added at room temperature, and the reaction was continued to stir at room temperature for another 1 hour. The excess acid and solvent were removed under reduced pressure, and the target compound 13 (20 mg, yield: 16.93%) was purified by preparative HPLC.
Ms m/z(ESI):591.2[M+H]+ Ms m/z(ESI):591.2[M+H] +
实施例14:3-(1R,5S)-3,8-重氮环氧[3.2.1]-辛烷-3-基)-5-氟-3-(2R,7aS)-2-氟四氢-1H-吡咯烷酮-7a(5H)-基)甲氧基)-异喹啉-6-基-5-氯-4-环丙基苯胺(化合物14)Example 14: 3-(1R,5S)-3,8-diazoepoxy[3.2.1]-octane-3-yl)-5-fluoro-3-(2R,7aS)-2-fluorotetra Hydrogen-1H-pyrrolidone-7a(5H-yl)methoxy)-isoquinolin-6-yl-5-chloro-4-cyclopropylaniline (compound 14)
3-(1-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-5-fluoro-3-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)isoquinolin-6-yl)-5-chloro-4-cyclopropylaniline3-(1-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-5-fluoro-3-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)isoquinolin-6-yl)-5-chloro-4-cyclopropylaniline
第一步:3-溴-5-氯-4-碘苯胺(14B)The first step: 3-bromo-5-chloro-4-iodoaniline (14B)
3-bromo-5-chloro-4-iodoaniline3-bromo-5-chloro-4-iodoaniline
将3-溴-5-氯-苯胺(14A)(1.00g,4.84mmol)和NIS(1.31g,5.81mmol)依次加入到50mL 的圆底烧瓶中,在常温条件下持续搅拌3h。待反应完全结束后,用水(30mL)/乙酸乙酯(40mL) 进行萃取,无水硫酸钠干燥后过滤。减压浓缩滤液后经硅胶柱层析纯化,获得目标化合物 (14B)(1.30g,80.76%)。3-Bromo-5-chloro-aniline (14A) (1.00g, 4.84mmol) and NIS (1.31g, 5.81mmol) were successively added into a 50mL round bottom flask, and the stirring was continued at room temperature for 3h. After the reaction is complete, extract with water (30mL)/ethyl acetate (40mL), dry over anhydrous sodium sulfate and filter. The filtrate was concentrated under reduced pressure and purified by silica gel column chromatography to obtain the target compound (14B) (1.30 g, 80.76%).
第二步:3-溴-5-氯-4-环丙基苯胺(14C)The second step: 3-bromo-5-chloro-4-cyclopropylaniline (14C)
3-bromo-5-chloro-4-cyclopropylaniline3-bromo-5-chloro-4-cyclopropylaniline
在氮气保护下,依次将(14B)(1.30g,3.91mmol)、环丙基硼酸(0.40g,4.69mmol)、Pd(dppf)Cl2(0.28g,0.39mmol)和Cs2CO3(2.55g,7.82mmol)加入到50mL的圆底烧瓶中,再加入 1,4-二氧六环(15mL)和水(3mL)的混合溶剂,反应于110℃持续搅拌4h。待反应完全结束后,冷却至室温,减压除去溶剂,加入30mL水,再用乙酸乙酯(20mL×3)萃取,合并有机相后用无水硫酸钠干燥后过滤,减压浓缩滤液后经硅胶柱层析纯化(PE:EA=10:1),获得目标化合物14C(0.80g,82.96%产率)。Under nitrogen protection, (14B) (1.30g, 3.91mmol), cyclopropylboronic acid (0.40g, 4.69mmol), Pd(dppf)Cl 2 (0.28g, 0.39mmol) and Cs 2 CO 3 (2.55 g, 7.82mmol) was added to a 50mL round bottom flask, and then a mixed solvent of 1,4-dioxane (15mL) and water (3mL) was added, and the reaction was continued to stir at 110°C for 4h. After the reaction is complete, cool to room temperature, remove the solvent under reduced pressure, add 30 mL of water, then extract with ethyl acetate (20 mL×3), combine the organic phases, dry with anhydrous sodium sulfate and filter, concentrate the filtrate under reduced pressure, and Purified by silica gel column chromatography (PE:EA=10:1), the target compound 14C was obtained (0.80 g, 82.96% yield).
第三步:3-氯-4-环丙基-5-(4,4,5,5-四甲基-1,3,2-二氧硼烷-2-基)苯胺(14D)The third step: 3-chloro-4-cyclopropyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)aniline (14D)
3-chloro-4-cyclopropyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)aniline3-chloro-4-cyclopropyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)aniline
在氮气保护下,依次将(14C)(0.80g,3.25mmol)、联硼酸频那醇酯(0.99g,3.89mmol)、 Pd(dppf)Cl2(0.23g,0.33mmol)和KOAc(0.96g,9.74mmol)加入到50mL的圆底烧瓶中,再加入 1,4-二氧六环(15mL)和水(3mL)的混合溶剂,反应于90℃持续搅拌4h。冷却至室温,减压除去溶剂,加入30mL水,再用乙酸乙酯(20mL×3)萃取,合并有机相后用无水硫酸钠干燥后过滤,减压浓缩滤液,残余物经硅胶柱层析纯化,获得目标化合物(14D)(0.8g,83.97%)。Under nitrogen protection, (14C) (0.80g, 3.25mmol), diboronic acid pinacol ester (0.99g, 3.89mmol), Pd(dppf)Cl 2 (0.23g, 0.33mmol) and KOAc (0.96g , 9.74mmol) into a 50mL round bottom flask, and then a mixed solvent of 1,4-dioxane (15mL) and water (3mL), and the reaction was stirred at 90°C for 4h. Cool to room temperature, remove the solvent under reduced pressure, add 30 mL of water, then extract with ethyl acetate (20 mL×3), combine the organic phases, dry with anhydrous sodium sulfate and filter, concentrate the filtrate under reduced pressure, and the residue is subjected to silica gel column chromatography After purification, the target compound (14D) (0.8 g, 83.97%) was obtained.
Ms m/z(ESI):294.2[M+H]+ Ms m/z(ESI):294.2[M+H] +
第四步:叔丁基(1R,5S)-3-(5-氨基-3-氯-2-环丙基苯)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯烷酮-7a(5H)-基)甲氧基)-喹唑啉-4-基)-3,8-重氮环[3.2.1]辛烷-8-羧酸酯(14E)The fourth step: tert-butyl (1R,5S)-3-(5-amino-3-chloro-2-cyclopropylbenzene)-8-fluoro-2-(((2R,7aS)-2-fluorotetra Hydrogen-1H-pyrrolidone-7a(5H)-yl)methoxy)-quinazolin-4-yl)-3,8-diazocyclo[3.2.1]octane-8-carboxylate (14E)
tert-butyl(1R,5S)-3-(7-(5-amino-3-chloro-2-cyclopropylphenyl)-8-fluoro-2-(((2R,7aS)-2-fluorot etrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carbo xylatetert-butyl(1R,5S)-3-(7-(5-amino-3-chloro-2-cyclopropylphenyl)-8-fluoro-2-(((2R,7aS)-2-fluorot etrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carbo xylate
在氮气保护下,将(14D)(0.12g,0.40mmol)溶解于1,4-二氧六环(5mL)和水(1mL)的混合溶液中,然后再依次加入中间体2F(0.20g,0.33mmol)、碳酸铯(0.22g,0.67mmol)和Pd(dppf)Cl2 (0.02g,0.03mmol),110℃的温度反应5h,将反应冷却至室温,加入乙酸乙酯40mL和水25 mL,搅拌并分液,水相再用乙酸乙酯(40mL×1)萃取一次,合并乙酸乙酯层,乙酸乙酯层用饱和食盐水洗涤一次(40mL×1),无水硫酸钠干燥,减压浓缩后,残余物用硅胶色谱柱柱层析得到化合物(14E)(0.20g,87.27%)Under nitrogen protection, (14D) (0.12g, 0.40mmol) was dissolved in a mixed solution of 1,4-dioxane (5mL) and water (1mL), and then intermediate 2F (0.20g, 0.33mmol), cesium carbonate (0.22g, 0.67mmol) and Pd(dppf)Cl 2 (0.02g, 0.03mmol), react at a temperature of 110°C for 5h, cool the reaction to room temperature, add ethyl acetate 40mL and water 25 mL , stirred and separated, the aqueous phase was extracted once with ethyl acetate (40mL×1), the ethyl acetate layers were combined, the ethyl acetate layer was washed once with saturated brine (40mL×1), dried over anhydrous sodium sulfate, and reduced After concentrated under reduced pressure, the residue was chromatographed on a silica gel column to obtain compound (14E) (0.20 g, 87.27%)
Ms m/z(ESI):681.3[M+H]+ Ms m/z(ESI):681.3[M+H] +
第五步:3-(4-((1R,5S)-3,8-重氮环氧[3.2.1]-辛烷-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯烷酮-7a(5H)-基)甲基)-5-氯-4-环丙基苯胺(化合物14)The fifth step: 3-(4-((1R,5S)-3,8-diazoepoxy[3.2.1]-octane-3-yl)-8-fluoro-2-(((2R,7aS )-2-fluorotetrahydro-1H-pyrrolidone-7a(5H)-yl)methyl)-5-chloro-4-cyclopropylaniline (compound 14)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-chloro-4-cyclopropylaniline3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-5-chloro-4-cyclopropylaniline
将化合物(14E)(0.20g,0.29mmol)溶解于二氯甲烷10mL中,加入三氟乙酸0.3mL,加完后室温反应3h,然后将反应直接减压浓缩除去溶剂后,残余物经过反相柱层析分离纯化得目标化合物14(0.18g,90.00%)。Dissolve compound (14E) (0.20g, 0.29mmol) in 10mL of dichloromethane, add 0.3mL of trifluoroacetic acid, react at room temperature for 3h after the addition, and then directly concentrate the reaction under reduced pressure to remove the solvent, and the residue is reversed phase The target compound 14 (0.18 g, 90.00%) was obtained by separation and purification by column chromatography.
Ms m/z(ESI):581.3[M+H]+ Ms m/z(ESI):581.3[M+H] +
1H NMR(400MHz,CDCl3)δ7.57(d,1H),7.09–6.99(m,1H),6.78(d,1H),6.50(d,1H),5.38 –5.17(m,1H),4.48–4.33(m,2H),4.26(d,1H),4.13(d,1H),3.80-3.59(m,4H),3.58–3.46(m, 2H),3.31–3.11(m,3H),3.03–2.92(dm,1H),2.35–2.14(m,3H),2.07–1.72(m,11H),0.68– 0.49(m,2H). 1 H NMR (400MHz, CDCl 3 )δ7.57(d,1H),7.09–6.99(m,1H),6.78(d,1H),6.50(d,1H),5.38–5.17(m,1H), 4.48–4.33(m,2H),4.26(d,1H),4.13(d,1H),3.80-3.59(m,4H),3.58–3.46(m,2H),3.31–3.11(m,3H), 3.03–2.92(dm,1H),2.35–2.14(m,3H),2.07–1.72(m,11H),0.68–0.49(m,2H).
实施例15:3-(4-(1R,5S)-3,8-二氮杂双环[3.2.1]-辛烷-3-基)-8-氟-2-(2R,7aS)-2-氟四氢-1H-吡咯烷酮-7a(5H)-基)甲基)喹唑啉-7-基)-5-氯-4-(三氟甲基)苯胺(化合物15)Example 15: 3-(4-(1R,5S)-3,8-diazabicyclo[3.2.1]-octane-3-yl)-8-fluoro-2-(2R,7aS)-2 -Fluorotetrahydro-1H-pyrrolidone-7a(5H)-yl)methyl)quinazolin-7-yl)-5-chloro-4-(trifluoromethyl)aniline (compound 15)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-chloro-4-(trifluoromethyl)aniline3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-5-chloro-4-(trifluoromethyl)aniline
第一步:(3-溴-5-氯-4-碘苯基)氨基甲酸叔丁酯(15A)The first step: (3-bromo-5-chloro-4-iodophenyl) tert-butyl carbamate (15A)
tert-butyl(3-bromo-5-chloro-4-iodophenyl)carbamatetert-butyl(3-bromo-5-chloro-4-iodophenyl)carbamate
在N2的保护下,先将化合物14B(0.5g,1.5mmol)、二碳酸二叔丁酯(0.36g,1.65mmol) 溶解在2mL的THF中,然后在冰浴的条件下,缓慢滴加LiHMDS(0.55g,3.30mL),滴加完毕后,将反应液移至常温条件下反应3h,待反应完全结束后,EA/H2O(150mL/100mL)进行萃取洗涤,浓缩有机层,经层析柱纯化得到化合物15A(0.45g,产率:69.37%)。Under the protection of N 2 , first dissolve compound 14B (0.5g, 1.5mmol), di-tert-butyl dicarbonate (0.36g, 1.65mmol) in 2mL of THF, and then slowly dropwise add LiHMDS (0.55g, 3.30mL), after the dropwise addition, the reaction solution was moved to normal temperature for 3h reaction, after the reaction was completely completed, EA/H 2 O (150mL/100mL) was extracted and washed, the organic layer was concentrated, and the The compound 15A (0.45 g, yield: 69.37%) was obtained by chromatography column purification.
Ms m/z(ESI):431.9[M-H]+ Ms m/z(ESI):431.9[MH] +
第二步:(3-溴-5-氯-4-(三氟甲基)苯基)氨基甲酸叔丁酯(15B)The second step: tert-butyl (3-bromo-5-chloro-4-(trifluoromethyl)phenyl)carbamate (15B)
tert-butyl(3-bromo-5-chloro-4-(trifluoromethyl)phenyl)carbamatetert-butyl(3-bromo-5-chloro-4-(trifluoromethyl)phenyl)carbamate
在N2的保护下,依次将化合物15A(0.1g,0.23mmol)、MFSDA(0.31g,1.61mmol)和CuI(53.0mg,0.28mmol)加入到反应试管中,然后再将DIPEA(0.21g,1.61mmol)溶解在5mL的DMF中并缓慢滴加到反应试管中。在75℃的油浴锅中持续反应16h,待反应结束后, EA/H2O(200mL/150mL*2)进行萃取洗涤。最后将有机层浓缩经层析柱纯化(PE:EA=10:1),获得化合物15B(80.0mg,产率:92.86%yield)。Under the protection of N2 , compound 15A (0.1g, 0.23mmol), MFSDA (0.31g, 1.61mmol) and CuI (53.0mg, 0.28mmol) were added to the reaction test tube in turn, and then DIPEA (0.21g, 1.61 mmol) was dissolved in 5 mL of DMF and slowly added dropwise to the reaction tube. The reaction was continued in an oil bath at 75° C. for 16 hours. After the reaction was completed, EA/H 2 O (200 mL/150 mL*2) was used for extraction and washing. Finally, the organic layer was concentrated and purified by column chromatography (PE:EA=10:1) to obtain compound 15B (80.0 mg, yield: 92.86% yield).
Ms m/z(ESI):373.9[M-H]+ Ms m/z(ESI):373.9[MH] +
第三步:叔丁基(1R,5S)-3-(7-(5-((叔丁氧羰基)氨基)-3-氯-2-(三氟甲基)苯基)-8-氟-2-((2R,7aS)-2- 氟四氢-1H-吡咯啉-7a(5H)-甲氧基)喹唑啉-4-基)-3,8-二氮杂环[3.2.1]辛烷-8-羧酸酯(15C)。 tert-butyl(1R,5S)-3-(7-(5-((tert-butoxycarbonyl)amino)-3-chloro-2-(trifluoromethyl)phenyl)-8-fluoro -2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicycl o[3.2.1]octane-8-carboxylateThe third step: tert-butyl (1R,5S)-3-(7-(5-((tert-butoxycarbonyl)amino)-3-chloro-2-(trifluoromethyl)phenyl)-8-fluoro -2-((2R,7aS)-2-fluorotetrahydro-1H-pyrroline-7a(5H)-methoxy)quinazolin-4-yl)-3,8-diazacyclo[3.2. 1] Octane-8-carboxylate (15C). tert-butyl(1R,5S)-3-(7-(5-((tert-butoxycarbonyl)amino)-3-chloro-2-(trifluoromethyl)phenyl)-8-fluoro-2-(((2R,7aS )-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
将中间体9B(200mg,0.36mmol)和化合物15B(0.16mg,0.43mmol)置于封管中,加入5mL 1,4-二氧六环溶液溶解,然后依次加入[1,1'-双(二苯基膦基)二茂铁]二氯化钯(26.0mg, 0.036mmol)、碳酸铯(230.0mg,0.72mmol)。在氮气保护下,升至110℃反应5h,减压除去溶剂,加入50mL水和100mL乙酸乙酯溶解,水相用50mL×2乙酸乙酯洗,合并有机相,有机相用40mL饱和食盐水洗一次,然后有机相用无水硫酸钠干燥,旋干,柱层析(DCM: MeOH=15:1)得到化合物15C(270.0mg,产率:92.68%)。Intermediate 9B (200 mg, 0.36 mmol) and compound 15B (0.16 mg, 0.43 mmol) were placed in a sealed tube, 5 mL of 1,4-dioxane solution was added to dissolve, and then [1,1'-bis( Diphenylphosphino)ferrocene]palladium dichloride (26.0 mg, 0.036 mmol), cesium carbonate (230.0 mg, 0.72 mmol). Under the protection of nitrogen, rise to 110 ° C for 5 h, remove the solvent under reduced pressure, add 50 mL of water and 100 mL of ethyl acetate to dissolve, wash the water phase with 50 mL×2 ethyl acetate, combine the organic phase, and wash the organic phase once with 40 mL of saturated saline , and then the organic phase was dried with anhydrous sodium sulfate, spin-dried, and column chromatography (DCM:MeOH=15:1) gave compound 15C (270.0 mg, yield: 92.68%).
Ms m/z(ESI):809.3[M+H]+ Ms m/z(ESI):809.3[M+H] +
第四步:3-(4-(1R,5S)-3,8-二氮杂双环[3.2.1]-辛烷-3-基)-8-氟-2-(2R,7aS)-2-氟四氢-1H-吡咯烷酮-7a(5H)-基)甲基)喹唑啉-7-基)-5-氯-4-(三氟甲基)苯胺(化合物15)The fourth step: 3-(4-(1R,5S)-3,8-diazabicyclo[3.2.1]-octane-3-yl)-8-fluoro-2-(2R,7aS)-2 -Fluorotetrahydro-1H-pyrrolidone-7a(5H)-yl)methyl)quinazolin-7-yl)-5-chloro-4-(trifluoromethyl)aniline (compound 15)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-chloro-4-(trifluoromethyl)aniline3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-chloro-4-(trifluoromethyl)aniline
将化合物15C(270mg,0.33mmol)溶于10mL的DCM中,然后于室温下滴加1mL三氟乙酸,体系继续反应3h,减压浓缩反应液,残留物经制备HPLC纯化,制备液浓缩后冻干得到目标化合物15(150mg,产率:74.63%)。Compound 15C (270 mg, 0.33 mmol) was dissolved in 10 mL of DCM, then 1 mL of trifluoroacetic acid was added dropwise at room temperature, the system continued to react for 3 h, the reaction solution was concentrated under reduced pressure, the residue was purified by preparative HPLC, and the preparative solution was concentrated and frozen Drying gave the target compound 15 (150 mg, yield: 74.63%).
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH 4 HCO 3 ); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min .
Ms m/z(ESI):609.2[M+H]+ Ms m/z(ESI):609.2[M+H] +
实施例16:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]-辛烷-3-基)-8-氟-2-(2R,7aS)-2-氟四氢-1H- 吡咯烷酮-7a(5H)-基)甲氧基)喹唑啉-7-基)-5-氟-4-环丙基苯胺(化合物16)Example 16: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]-octane-3-yl)-8-fluoro-2-(2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolidone-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-fluoro-4-cyclopropylaniline (compound 16)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-cyclopropyl-5-fluoroaniline3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-4-cyclopropyl-5-fluoroaniline
第一步:3-溴-5-氟-4-碘苯胺(16B)The first step: 3-bromo-5-fluoro-4-iodoaniline (16B)
3-bromo-5-fluoro-4-iodoaniline3-bromo-5-fluoro-4-iodoaniline
将3-溴-5-氟-苯胺(16A)(2.00g,10.53mmol)和NIS(2.61g,11.58mmol)依次加入到100mL 的圆底烧瓶中,在常温条件下持续搅拌3h。待反应完全结束后,用水(100mL)/乙酸乙酯(150 mL)进行萃取,无水硫酸钠干燥后过滤。减压浓缩滤液后经硅胶柱层析纯化,获得目标化合物 (16B)(3.10g,产率:97.19%)。3-Bromo-5-fluoro-aniline (16A) (2.00g, 10.53mmol) and NIS (2.61g, 11.58mmol) were successively added into a 100mL round bottom flask, and the stirring was continued at room temperature for 3h. After the reaction is complete, extract with water (100 mL)/ethyl acetate (150 mL), dry over anhydrous sodium sulfate and filter. The filtrate was concentrated under reduced pressure and purified by silica gel column chromatography to obtain the target compound (16B) (3.10 g, yield: 97.19%).
第二步:3-溴-5-氟-4-环丙基苯胺(16C)The second step: 3-bromo-5-fluoro-4-cyclopropylaniline (16C)
3-bromo-5-fluoro-4-cyclopropylaniline3-bromo-5-fluoro-4-cyclopropylaniline
在氮气保护下,依次将(16B)(3.0g,9.50mmol)、环丙基硼酸(0.98g,11.40mmol)、Pd(dppf)Cl2(0.70g,0.95mmol)和Cs2CO3(6.19g,19.00mmol)加入到100mL的圆底烧瓶中,再加入1,4-二氧六环(15mL)和水(3mL)的混合溶剂,反应于110℃持续搅拌4h。待反应完全结束后,冷却至室温,减压除去溶剂,加入100mL水,再用乙酸乙酯(100mL×3)萃取,合并有机相后用无水硫酸钠干燥后过滤,减压浓缩滤液后经硅胶柱层析纯化(PE:EA=10:1),获得目标化合物16C(1.40g,产率:64.05%)。Under nitrogen protection, (16B) (3.0g, 9.50mmol), cyclopropylboronic acid (0.98g, 11.40mmol), Pd(dppf)Cl 2 (0.70g, 0.95mmol) and Cs 2 CO 3 (6.19 g, 19.00mmol) was added to a 100mL round bottom flask, and then a mixed solvent of 1,4-dioxane (15mL) and water (3mL) was added, and the reaction was stirred continuously at 110°C for 4h. After the reaction is complete, cool to room temperature, remove the solvent under reduced pressure, add 100mL of water, and then extract with ethyl acetate (100mL×3), combine the organic phases, dry with anhydrous sodium sulfate and filter, concentrate the filtrate under reduced pressure, and Purified by silica gel column chromatography (PE:EA=10:1), the target compound 16C (1.40 g, yield: 64.05%) was obtained.
第三步:3-氟-4-环丙基-5-(4,4,5,5-四甲基-1,3,2-二氧硼烷-2-基)苯胺(16D)The third step: 3-fluoro-4-cyclopropyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)aniline (16D)
3-fluoro-4-cyclopropyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)aniline3-fluoro-4-cyclopropyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)aniline
在氮气保护下,依次将(16C)(1.40g,6.08mmol)、联硼酸频那醇酯(1.85g,7.30mmol)、 Pd(dppf)Cl2(0.45g,0.61mmol)和KOAc(1.79g,18.24mmol)加入到50mL的圆底烧瓶中,再加入1,4-二氧六环(15mL)和水(3mL)的混合溶剂,反应于90℃持续搅拌4h。冷却至室温,减压除去溶剂,加入30mL水,再用乙酸乙酯(40mL×3)萃取,合并有机相后用无水硫酸钠干燥后过滤,减压浓缩滤液,残余物经硅胶柱层析纯化,获得目标化合物(16D)(1.4g,产率: 83.09%)。Under nitrogen protection, (16C) (1.40g, 6.08mmol), diboronic acid pinacol ester (1.85g, 7.30mmol), Pd(dppf)Cl 2 (0.45g, 0.61mmol) and KOAc (1.79g , 18.24mmol) into a 50mL round bottom flask, and then a mixed solvent of 1,4-dioxane (15mL) and water (3mL), and the reaction was stirred at 90°C for 4h. Cool to room temperature, remove the solvent under reduced pressure, add 30 mL of water, then extract with ethyl acetate (40 mL×3), combine the organic phases, dry with anhydrous sodium sulfate and filter, concentrate the filtrate under reduced pressure, and the residue is subjected to silica gel column chromatography Purified to obtain the target compound (16D) (1.4 g, yield: 83.09%).
Ms m/z(ESI):278.3[M+H]+ Ms m/z(ESI):278.3[M+H] +
第四步:叔丁基(1R,5S)-3-(5-氨基-3-氟-2-环丙基苯)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯烷酮 -7a(5H)-基)甲氧基)-喹唑啉-4-基)-3,8-重氮环[3.2.1]辛烷-8-羧酸酯(16E)The fourth step: tert-butyl (1R,5S)-3-(5-amino-3-fluoro-2-cyclopropylphenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetra Hydrogen-1H-pyrrolidone-7a(5H-yl)methoxy)-quinazolin-4-yl)-3,8-diazocyclo[3.2.1]octane-8-carboxylate (16E)
tert-butyl(1R,5S)-3-(7-(5-amino-3-fluoro-2-cyclopropylphenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrah ydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylat etert-butyl(1R,5S)-3-(7-(5-amino-3-fluoro-2-cyclopropylphenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrah ydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
在氮气保护下,将(16D)(0.11g,0.41mmol)溶解于1,4-二氧六环(5mL)和水(1mL)的混合溶液中,然后再依次加入中间体2F(0.20g,0.34mmol)、碳酸铯(0.10g,1.02mmol)和Pd(dppf)Cl2 (25.0mg,0.03mmol),110℃的温度反应5h,将反应冷却至室温,加入乙酸乙酯40mL和水 25mL,搅拌并分液,水相再用乙酸乙酯(40mL×1)萃取一次,合并乙酸乙酯层,乙酸乙酯层用饱和食盐水洗涤一次(40mL×1),无水硫酸钠干燥,减压浓缩后,残余物用硅胶色谱柱柱层析得到化合物16E)(0.18g,产率:79.64%)Under nitrogen protection, (16D) (0.11g, 0.41mmol) was dissolved in a mixed solution of 1,4-dioxane (5mL) and water (1mL), and then intermediate 2F (0.20g, 0.34mmol), cesium carbonate (0.10g, 1.02mmol) and Pd(dppf)Cl 2 (25.0mg, 0.03mmol), reacted at a temperature of 110°C for 5h, cooled the reaction to room temperature, added 40mL of ethyl acetate and 25mL of water, Stir and separate the liquids, extract the aqueous phase once more with ethyl acetate (40mL×1), combine the ethyl acetate layers, wash the ethyl acetate layer once with saturated brine (40mL×1), dry over anhydrous sodium sulfate, and reduce pressure After concentration, the residue was chromatographed on a silica gel column to obtain compound 16E) (0.18g, yield: 79.64%)
Ms m/z(ESI):665.3[M+H]+ Ms m/z(ESI):665.3[M+H] +
第五步:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]-辛烷-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯烷酮-7a(5H)-基)甲基)-5-氟-4-环丙基苯胺(化合物16)The fifth step: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]-octane-3-yl)-8-fluoro-2-(((2R,7aS )-2-fluorotetrahydro-1H-pyrrolidone-7a(5H)-yl)methyl)-5-fluoro-4-cyclopropylaniline (compound 16)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-fluoro-4-cyclopropylaniline3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-5-fluoro-4-cyclopropylaniline
将化合物(14E)(0.18g,0.27mmol)溶解于二氯甲烷2.0mL中,加入三氟乙酸0.2mL,加完后室温反应3h,然后将反应直接减压浓缩除去溶剂后,残余物经过反相柱层析分离纯化得目标化合物16(0.10g,产率:65.59%)。Dissolve compound (14E) (0.18g, 0.27mmol) in 2.0mL of dichloromethane, add 0.2mL of trifluoroacetic acid, react at room temperature for 3h after the addition is complete, then directly concentrate the reaction under reduced pressure to remove the solvent, and the residue is subjected to reaction The target compound 16 (0.10 g, yield: 65.59%) was obtained by separation and purification by column chromatography.
Ms m/z(ESI):565.3[M+H]+ Ms m/z(ESI):565.3[M+H] +
实施例17:4-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛烷-3-基)-8-氟-2-((2R,7aS)-2-氟四氢呋喃-1H- 吡咯啉-7a(5H)-基)甲氧基)喹唑啉-7-基)-6-氨基苯并呋喃-7-碳腈(化合物17)。Example 17: 4-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octane-3-yl)-8-fluoro-2-((2R,7aS)- 2-Fluorotetrahydrofuran-1H-pyrrolin-7a(5H)-yl)methoxy)quinazolin-7-yl)-6-aminobenzofuran-7-carbonitrile (compound 17).
4-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-6-aminobenzofuran-7-carbonitrile4-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-6-aminobenzofuran-7-carbonitrile
第一步:6-氨基-4-(4,4,5,5-四甲基-1,3,2-二氧苯甲醛-2-基)苯并呋喃-7-碳腈6-amino-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzofuran-7-carbonitrileStep 1: 6-Amino-4-(4,4,5,5-Tetramethyl-1,3,2-Dioxybenzaldehyde-2-yl)benzofuran-7-carbonitrile 6-amino- 4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzofuran-7-carbonitrile
在氮气保护下,依次将(17A)(0.2g,0.84mmol)、联硼酸频那醇酯(10.26g,1.01mmol)、 Pd(dppf)Cl2(0.06g,0.084mmol)和KOAc(0.16g,1.68mmol)加入到50mL的圆底烧瓶中,再加入 1,4-二氧六环(3mL)溶剂,反应于90℃持续搅拌6h。冷却至室温,减压除去溶剂,加入 30mL水,再用乙酸乙酯(300mL×3)萃取,合并有机相后用无水硫酸钠干燥后过滤,减压浓缩滤液,残余物经硅胶柱层析纯化,获得目标化合物17B(0.2g,产率:83.8%)。Under nitrogen protection, (17A) (0.2g, 0.84mmol), diboronic acid pinacol ester (10.26g, 1.01mmol), Pd(dppf)Cl 2 (0.06g, 0.084mmol) and KOAc (0.16g , 1.68mmol) was added to a 50mL round bottom flask, and then 1,4-dioxane (3mL) solvent was added, and the reaction was continuously stirred at 90°C for 6h. Cool to room temperature, remove the solvent under reduced pressure, add 30 mL of water, then extract with ethyl acetate (300 mL×3), combine the organic phases, dry with anhydrous sodium sulfate and filter, concentrate the filtrate under reduced pressure, and the residue is subjected to silica gel column chromatography After purification, the target compound 17B was obtained (0.2 g, yield: 83.8%).
Ms m/z(ESI):285.1[M+H]+ Ms m/z(ESI):285.1[M+H] +
第二步:叔丁基(1R,5S)-3-(7-(6-氨基-7-氰基苯并呋喃-4-基)-8-氟-2-((2R,7aS)-2-氟四氢-1H- 吡咯啉-7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂环[3.2.1]辛烷-8-羧酸酯(17C)。The second step: tert-butyl (1R, 5S)-3-(7-(6-amino-7-cyanobenzofuran-4-yl)-8-fluoro-2-((2R, 7aS)-2 -Fluorotetrahydro-1H-pyrroline-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazacyclo[3.2.1]octane-8-carboxylic acid Ester (17C).
tert-butyl(1R,5S)-3-(7-(6-amino-7-cyanobenzofuran-4-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro- 1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylatetert-butyl(1R,5S)-3-(7-(6-amino-7-cyanobenzofuran-4-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro- 1H-pyrrolizin- 7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
将中间体2F(130.0mg,0.22mmol)和化合物17B(75.0mg,0.26mmol)置于封管中,加入2mL 1,4-二氧六环溶液溶解,然后依次加入[1,1'-双(二苯基膦基)二茂铁]二氯化钯(16.0 mg,0.022mmol)、碳酸铯(140.0mg,0.44mmol)。在氮气保护下,升至110℃反应6h,减压除去溶剂,加入30mL水和60mL乙酸乙酯溶解,水相用100mL×2乙酸乙酯洗,合并有机相,有机相用80mL饱和食盐水洗一次,然后有机相用无水硫酸钠干燥,旋干,柱层析(DCM: MeOH=15:1)得到目标化合物(100.0mg,收率:67.67%)。Intermediate 2F (130.0 mg, 0.22 mmol) and compound 17B (75.0 mg, 0.26 mmol) were placed in a sealed tube, 2 mL of 1,4-dioxane solution was added to dissolve, and then [1,1'-bis (Diphenylphosphino)ferrocene]palladium dichloride (16.0 mg, 0.022 mmol), cesium carbonate (140.0 mg, 0.44 mmol). Under the protection of nitrogen, rise to 110°C to react for 6h, remove the solvent under reduced pressure, add 30mL water and 60mL ethyl acetate to dissolve, wash the aqueous phase with 100mL×2 ethyl acetate, combine the organic phase, and wash the organic phase once with 80mL saturated brine , and then the organic phase was dried with anhydrous sodium sulfate, spin-dried, and column chromatography (DCM:MeOH=15:1) gave the target compound (100.0 mg, yield: 67.67%).
Ms m/z(ESI):672.3[M+H]+ Ms m/z(ESI):672.3[M+H] +
第三步:4-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛烷-3-基)-8-氟-2-((2R,7aS)-2-氟四氢呋喃-1H- 吡咯啉-7a(5H)-基)甲氧基)喹唑啉-7-基)-6-氨基苯并呋喃-7-碳腈(化合物17)。The third step: 4-(4-((1R, 5S)-3,8-diazabicyclo[3.2.1]octane-3-yl)-8-fluoro-2-((2R, 7aS)- 2-Fluorotetrahydrofuran-1H-pyrrolin-7a(5H)-yl)methoxy)quinazolin-7-yl)-6-aminobenzofuran-7-carbonitrile (compound 17).
4-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-6-aminobenzofuran-7-carbonitrile4-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-6-aminobenzofuran-7-carbonitrile
将化合物17C(100mg,0.15mmol)溶于1mLDCM中,然后于室温下滴加0.1mL三氟乙酸,体系继续反应3h,减压浓缩反应液,残留物经制备HPLC纯化,制备液浓缩后冻干得到目标化合物17(50.0mg,产率:58.31%)。Compound 17C (100mg, 0.15mmol) was dissolved in 1mL DCM, then 0.1mL trifluoroacetic acid was added dropwise at room temperature, the system continued to react for 3h, the reaction solution was concentrated under reduced pressure, the residue was purified by preparative HPLC, the preparative solution was concentrated and lyophilized The target compound 17 (50.0 mg, yield: 58.31%) was obtained.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH 4 HCO 3 ); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min .
Ms m/z(ESI):572.3[M+H]+ Ms m/z(ESI):572.3[M+H] +
实施例18:2-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛基-3-基)-8-氟-2-((2R,7aS)-2-氟四氢-1H-吡咯啉-7a(5H)-基)甲氧基)喹唑啉-7-基)-3,4,5,6-四氟苯胺(化合物18)。Example 18: 2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octyl-3-yl)-8-fluoro-2-((2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolin-7a(5H)-yl)methoxy)quinazolin-7-yl)-3,4,5,6-tetrafluoroaniline (Compound 18).
2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-3,4,5,6-tetrafluoroaniline2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-3,4,5,6-tetrafluoroaniline
第一步:2,3,4,5-四氟-6-(4,4,5,5-四甲基-1,3,2-二氧苯甲醛-2-基)苯胺(18B)The first step: 2,3,4,5-tetrafluoro-6-(4,4,5,5-tetramethyl-1,3,2-dioxobenzaldehyde-2-yl)aniline (18B)
2,3,4,5-tetrafluoro-6-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)aniline2,3,4,5-tetrafluoro-6-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)aniline
在氮气保护下,依次将(18A)(2.28g,9.35mmol)、联硼酸频那醇酯(3.97g,15.61mmol)、 Pd(dppf)Cl2(1.37g,1.87mmol)和KOAc(1.84g,18.7mmol)加入到100mL的圆底烧瓶中,再加入1,4-二氧六环(30mL)溶剂,反应于90℃持续搅拌6h。冷却至室温,减压除去溶剂,加入100mL水,再用乙酸乙酯(300mL×3)萃取,合并有机相后用无水硫酸钠干燥后过滤,减压浓缩滤液,残余物经硅胶柱层析纯化,获得目标化合物18B(1.4g,产率:83.6%)。Under nitrogen protection, (18A) (2.28g, 9.35mmol), diboronic acid pinacol ester (3.97g, 15.61mmol), Pd(dppf)Cl 2 (1.37g, 1.87mmol) and KOAc (1.84g , 18.7mmol) was added to a 100mL round bottom flask, and then 1,4-dioxane (30mL) solvent was added, and the reaction was continuously stirred at 90°C for 6h. Cool to room temperature, remove the solvent under reduced pressure, add 100mL of water, then extract with ethyl acetate (300mL×3), combine the organic phases, dry with anhydrous sodium sulfate and filter, concentrate the filtrate under reduced pressure, and the residue is subjected to silica gel column chromatography After purification, the target compound 18B was obtained (1.4 g, yield: 83.6%).
Ms m/z(ESI):292.1[M+H]+ Ms m/z(ESI):292.1[M+H] +
第二步:叔丁基(1R,5S)-3-(7-(2-氨基-3,4,5,6-四氟苯基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯利嗪-7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂环[3.2.1]辛烷-8-羧酸酯(18C)。The second step: tert-butyl (1R, 5S)-3-(7-(2-amino-3,4,5,6-tetrafluorophenyl)-8-fluoro-2-(((2R, 7aS) -2-Fluorotetrahydro-1H-pyrrolizine-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazacyclo[3.2.1]octane-8 -Carboxylate (18C).
tert-butyl(1R,5S)-3-(7-(2-amino-3,4,5,6-tetrafluorophenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro -1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylatetert-butyl(1R,5S)-3-(7-(2-amino-3,4,5,6-tetrafluorophenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
在氮气保护下,将中间体2F(0.12g,0.40mmol)溶解于1,4-二氧六环(5mL)和水(1mL)的混合溶液中,然后再依次加入18B(0.20g,0.33mmol)、碳酸铯(0.22g,0.67mmol)和Pd(dppf)Cl2 (0.02g,0.03mmol),110℃的温度反应5h,将反应冷却至室温,加入乙酸乙酯40mL和水25mL,水相再用乙酸乙酯(40mL×1)萃取一次,合并乙酸乙酯层,乙酸乙酯层用饱和食盐水洗涤一次 (40mL×1),无水硫酸钠干燥,减压浓缩后,残余物用硅胶色谱柱柱层析得到化合物18C(0.20 g,产率:86.67%)。Under nitrogen protection, the intermediate 2F (0.12g, 0.40mmol) was dissolved in a mixed solution of 1,4-dioxane (5mL) and water (1mL), and then 18B (0.20g, 0.33mmol) was added successively ), cesium carbonate (0.22g, 0.67mmol) and Pd(dppf)Cl 2 (0.02g, 0.03mmol), reacted at a temperature of 110°C for 5h, cooled the reaction to room temperature, added 40mL of ethyl acetate and 25mL of water, and the aqueous phase Extract once more with ethyl acetate (40mL×1), combine the ethyl acetate layers, wash the ethyl acetate layer once with saturated brine (40mL×1), dry over anhydrous sodium sulfate, concentrate under reduced pressure, and wash the residue with silica gel The compound 18C (0.20 g, yield: 86.67%) was obtained by column chromatography.
Ms m/z(ESI):679.3[M+H]+ Ms m/z(ESI):679.3[M+H] +
第三步:2-(4-((1R,5S)-3,8-二氮杂环[3.2.1]辛基-3-基)-8-氟-2-((2R,7aS)-2-氟四氢-1H-吡咯啉 -7a(5H)-基)甲氧基)喹唑啉-7-基)-3,4,5,6-四氟苯胺(化合物18)。The third step: 2-(4-((1R, 5S)-3,8-diazacyclo[3.2.1]octyl-3-yl)-8-fluoro-2-((2R, 7aS)- 2-Fluorotetrahydro-1H-pyrrolin-7a(5H)-yl)methoxy)quinazolin-7-yl)-3,4,5,6-tetrafluoroaniline (Compound 18).
2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-3,4,5,6-tetrafluoroaniline2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-3,4,5,6-tetrafluoroaniline
将化合物18C(0.20g,0.29mmol)溶解于二氯甲烷2.0mL中,加入三氟乙酸0.2mL,加完后室温反应3h,然后将反应直接减压浓缩除去溶剂后,经过反相柱层析分离纯化得目标化合物18(0.1g,产率:59.6%)。Dissolve compound 18C (0.20g, 0.29mmol) in 2.0mL of dichloromethane, add 0.2mL of trifluoroacetic acid, react at room temperature for 3h after the addition, and then directly concentrate the reaction under reduced pressure to remove the solvent, and perform reverse phase column chromatography The target compound 18 (0.1 g, yield: 59.6%) was obtained by separation and purification.
实施例19:2-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛烷-3-基)-8-氟-2-((2R,7aS)-2-氟四氢-1H-吡咯啉-7a(5H)-基)甲氧基)喹唑啉-7-基)-4-氨基-6-氟苯腈 2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-amino-6-fluorobenzonitrileExample 19: 2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octane-3-yl)-8-fluoro-2-((2R,7aS)- 2-Fluorotetrahydro-1H-pyrroline-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-amino-6-fluorobenzonitrile 2-(4-((1R,5S )-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy )quinazolin-7-yl)-4-amino-6-fluorobenzonitrile
第一步:4-溴-3-氯-5-氟苯胺The first step: 4-bromo-3-chloro-5-fluoroaniline
4-bromo-3-chloro-5-fluoroaniline4-bromo-3-chloro-5-fluoroaniline
将19a(1g,6.87mmol)溶于乙酸(12mL)中,然后于室温加入NBS(1.34g,7.56mmol),反应继续再室温搅拌4小时。减压除去多余的酸剂,经过40g正相柱纯化(PE:EA=50:1~10:1) 得目标化合物19b(750mg,收率:48.64%)。19a (1 g, 6.87 mmol) was dissolved in acetic acid (12 mL), then NBS (1.34 g, 7.56 mmol) was added at room temperature, and the reaction was continued to stir at room temperature for another 4 hours. The excess acid was removed under reduced pressure, and the target compound 19b (750 mg, yield: 48.64%) was obtained after 40 g of normal phase column purification (PE:EA=50:1~10:1).
Ms m/z(ESI):224.1[M+H]+ Ms m/z(ESI):224.1[M+H] +
第二步:4-氨基-2-氯-6-氟苯甲腈The second step: 4-amino-2-chloro-6-fluorobenzonitrile
4-amino-2-chloro-6-fluorobenzonitrile4-amino-2-chloro-6-fluorobenzonitrile
将19b(0.5g,2.23mmol)和CuCN(0.4g,4.46mmol)溶于DMF(5mL)中,在微波 190℃下反应搅拌1小时。加入100mL水,再用乙酸乙酯(50mL×3)萃取,合并有机相后用无水硫酸钠干燥后过滤,旋干,残余物经硅胶柱层析(PE:EA=50:1~5:1)得到19c(156mg,收率:41.01%)。19b (0.5g, 2.23mmol) and CuCN (0.4g, 4.46mmol) were dissolved in DMF (5mL), and the reaction was stirred under microwave at 190°C for 1 hour. Add 100mL of water, then extract with ethyl acetate (50mL×3), combine the organic phases, dry with anhydrous sodium sulfate, filter, spin dry, and the residue is subjected to silica gel column chromatography (PE:EA=50:1~5: 1) Obtain 19c (156 mg, yield: 41.01%).
Ms m/z(ESI):169.1[M-H]-Ms m/z(ESI):169.1[M-H]-
第三步:叔丁基(1R,5S)-3-(7-(5-氨基-2-氰基-3-氟苯基)-8-氟-2-((2R,7aS)-2-氟四氢 -1H-吡咯里嗪-7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂环[3.2.1]辛烷-8-羧酸酯 tert-butyl(1R,5S)-3-(7-(5-amino-2-cyano-3-fluorophenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro- 1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylateThe third step: tert-butyl (1R, 5S)-3-(7-(5-amino-2-cyano-3-fluorophenyl)-8-fluoro-2-((2R, 7aS)-2- Fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazacyclo[3.2.1]octane-8-carboxylic acid Ester tert-butyl(1R,5S)-3-(7-(5-amino-2-cyano-3-fluorophenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro- 1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
将中间体9B(150mg,0.28mmol),19c(72mg,0.42mmol),PdCl2(PPh3)2(20mg,0.028mmol)和KF(49mg,0.84mmol)加入到ACN(8mL)和水(2mL)的混合溶剂中。在氮气保护下,反应于封管中100℃搅拌12小时。待反应也冷却至室温后,减压除去溶剂,加入100mL水,再用乙酸乙酯(50mL×3)萃取,合并有机相后用无水硫酸钠干燥后过滤,旋干,残余物经硅胶柱层析分离纯化得到19d(81mg,收率:44.53%)。Intermediate 9B (150 mg, 0.28 mmol), 19c (72 mg, 0.42 mmol), PdCl2(PPh3)2 (20 mg, 0.028 mmol) and KF (49 mg, 0.84 mmol) were added to a mixture of ACN (8 mL) and water (2 mL) in a mixed solvent. Under nitrogen protection, the reaction was stirred at 100°C for 12 hours in a sealed tube. After the reaction was cooled to room temperature, the solvent was removed under reduced pressure, 100mL of water was added, and then extracted with ethyl acetate (50mL×3). Chromatographic separation and purification gave 19d (81 mg, yield: 44.53%).
Ms m/z(ESI):650.3[M+H]+ Ms m/z(ESI):650.3[M+H] +
第四步:2-(4-((1R,5S)-3,8-二氮杂环[3.2.1]辛烷-3-基)-8-氟-2-((2R,7aS)-2-氟四氢-1H- 吡咯啉-7a(5H)-基)甲氧基)喹唑啉-7-基)-4-氨基-6-氟苯腈 2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-amino-6-fluorobenzonitrileThe fourth step: 2-(4-((1R, 5S)-3,8-diazacyclo[3.2.1]octane-3-yl)-8-fluoro-2-((2R, 7aS)- 2-Fluorotetrahydro-1H-pyrroline-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-amino-6-fluorobenzonitrile 2-(4-((1R,5S )-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy )quinazolin-7-yl)-4-amino-6-fluorobenzonitrile
将19d(80mg,0.12mmol)溶于二氯甲烷(8mL)中,然后于室温加入2mL TFA,反应继续再室温搅拌1小时。减压除去多余的酸和溶剂,经过制备HPLC纯化得目标化合物19(18mg,收率:27.29%)。19d (80 mg, 0.12 mmol) was dissolved in dichloromethane (8 mL), then 2 mL of TFA was added at room temperature, and the reaction was continued to stir at room temperature for another 1 hour. The excess acid and solvent were removed under reduced pressure, and the target compound 19 (18 mg, yield: 27.29%) was obtained after purification by preparative HPLC.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH 4 HCO 3 ); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min .
Ms m/z(ESI):550.2[M+H]+ Ms m/z(ESI):550.2[M+H] +
1H NMR(400MHz,DMSO-d6)δ7.83(d,1H),7.24–7.18(m,1H),6.73(s,2H),6.59–6.49(m,2H),5.38–5.16(m,1H),4.26(d,2H),4.06(dd,2H),3.55–3.42(m,4H),3.16–2.98(m,4H), 2.86–2.78(m,1H),2.20–1.98(m,3H),1.88–1.73(m,3H),1.68–1.58(m,4H). 1 H NMR (400MHz,DMSO-d 6 )δ7.83(d,1H),7.24–7.18(m,1H),6.73(s,2H),6.59–6.49(m,2H),5.38–5.16(m ,1H),4.26(d,2H),4.06(dd,2H),3.55–3.42(m,4H),3.16–2.98(m,4H), 2.86–2.78(m,1H),2.20–1.98(m ,3H),1.88–1.73(m,3H),1.68–1.58(m,4H).
实施例20:6-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛基-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H- 吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-5-(三氟甲基)吡啶-2-胺(化合物20)Example 20: 6-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octyl-3-yl)-8-fluoro-2-(((2R,7aS) -2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-(trifluoromethyl)pyridin-2-amine (compound 20)
6-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-(trifluoromethyl)pyridin-2-amine.6-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-5-(trifluoromethyl)pyridin-2-amine.
第一步:叔丁基(1R,5S)-3-(7-(6-胺基-3-(三氟甲基)吡啶-2-基)-8-氟-2-(((2R,7aS)-2-氟四氢 -1H)-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸酯(20B)The first step: tert-butyl (1R,5S)-3-(7-(6-amino-3-(trifluoromethyl)pyridin-2-yl)-8-fluoro-2-(((2R, 7aS)-2-fluorotetrahydro-1H)-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane -8-Carboxylate (20B)
tert-butyl(1R,5S)-3-(7-(6-amino-3-(trifluoromethyl)pyridin-2-yl)-8-fluoro-2-(((2R,7aS)-2-fluoro tetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carbo xylate.tert-butyl(1R,5S)-3-(7-(6-amino-3-(trifluoromethyl)pyridin-2-yl)-8-fluoro-2-(((2R,7aS)-2-fluoro tetrahydro- 1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carbo xylate.
将(20A)(0.132g,0.672mmol)溶解于乙腈10mL和水1mL的混合溶液中,加入9B(0.188 g,0.336mmol),氟化钾(0.073g,1.257mmol),Pd(PPh)2Cl2(0.023g,0.034mmol),氮气保护下100℃下反应12h,将反应冷却至室温,加入乙酸乙酯40mL和水25mL搅拌后分液,水相再用乙酸乙酯(40mL×1)萃取一次,合并乙酸乙酯层,乙酸乙酯层用饱和食盐水洗涤一次(40 mL×1),无水硫酸钠干燥,减压浓缩后,残留物用硅胶色谱柱柱层析(DCM:MeOH=24:1)得到化合物(20B)(0.15g,产率66.07%)Dissolve (20A) (0.132g, 0.672mmol) in a mixed solution of 10mL of acetonitrile and 1mL of water, add 9B (0.188g, 0.336mmol), potassium fluoride (0.073g, 1.257mmol), Pd(PPh) 2 Cl 2 (0.023g, 0.034mmol), reacted at 100°C for 12h under nitrogen protection, cooled the reaction to room temperature, added 40mL of ethyl acetate and 25mL of water and stirred, then separated the liquid, and extracted the aqueous phase with ethyl acetate (40mL×1) Once, the ethyl acetate layers were combined, and the ethyl acetate layer was washed once with saturated brine (40 mL×1), dried over anhydrous sodium sulfate, concentrated under reduced pressure, and the residue was subjected to silica gel column chromatography (DCM:MeOH= 24:1) to obtain compound (20B) (0.15g, yield 66.07%)
LCMS m/z=676.3[M+H]+ LCMS m/z=676.3[M+H] +
第二步:6-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛基-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-5-(三氟甲基)吡啶-2-胺(化合物20)The second step: 6-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octyl-3-yl)-8-fluoro-2-(((2R,7aS) -2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-(trifluoromethyl)pyridin-2-amine (compound 20)
6-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-(trifluoromethyl)pyridin-2-amine.6-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-5-(trifluoromethyl)pyridin-2-amine.
将化合物(20B)(0.15g,0.22mmol)溶解于二氯甲烷5mL中,加入三氟乙酸0.5mL,室温下反应40分钟。减压浓缩反应液,然后加入二氯甲烷溶解,滴入0.5mL N,N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物20(40mg,产率31.3%),为白色粉末。Compound (20B) (0.15 g, 0.22 mmol) was dissolved in 5 mL of dichloromethane, 0.5 mL of trifluoroacetic acid was added, and the reaction was carried out at room temperature for 40 minutes. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 0.5mL N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, the residue is prepared and purified by HPLC, the preparation solution is concentrated and freeze-dried to obtain compound 20( 40 mg, yield 31.3%), as a white powder.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH 4 HCO 3 ); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min .
Ms m/z(ESI):576.2[M+H]+ Ms m/z(ESI):576.2[M+H] +
1H NMR(400MHz,CD3OD)δ7.79(d,2H),7.19–7.11(m,1H),6.69(d,1H),5.41–5.21(m, 1H),4.47(t,2H),4.24(dd,2H),3.66–3.53(m,4H),3.36–3.32(m,1H),3.26–3.15(m,2H),3.06 –2.96(m,1H),2.41–2.10(m,3H),2.05–1.87(m,3H),1.86–1.77(m,4H). 1 H NMR (400MHz, CD 3 OD) δ7.79(d,2H),7.19–7.11(m,1H),6.69(d,1H),5.41–5.21(m,1H),4.47(t,2H) ,4.24(dd,2H),3.66–3.53(m,4H),3.36–3.32(m,1H),3.26–3.15(m,2H),3.06–2.96(m,1H),2.41–2.10(m, 3H),2.05–1.87(m,3H),1.86–1.77(m,4H).
实施例21:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛基-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H- 吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-2,4,6-三氟苯胺(化合物21)Example 21: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octyl-3-yl)-8-fluoro-2-(((2R,7aS) -2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-2,4,6-trifluoroaniline (compound 21)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-2,4,6-trifluoroaniline.3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-2,4,6-trifluoroaniline.
第一步:2-溴-1,3,5-三氟-4-硝基苯(21B)The first step: 2-bromo-1,3,5-trifluoro-4-nitrobenzene (21B)
2-bromo-1,3,5-trifluoro-4-nitrobenzene.2-bromo-1,3,5-trifluoro-4-nitrobenzene.
将(21A)(5.0g,23.7mmol)溶解于浓硫酸5mL中,体系置于冰浴下10min。然后缓慢滴加5mL浓硝酸和15mL浓硫酸的混合溶剂,滴加完毕后,体系自动升至室温反应3h。待原料反应完全后,将体系倾倒至100mL冰水中淬灭反应,加入乙酸乙酯100mL搅拌后分液,水相再用乙酸乙酯(40mL×2)萃取一次,合并乙酸乙酯层,乙酸乙酯层分别用饱和碳酸氢钠溶液(40mL×1)和饱和食盐水(40mL×1)洗涤一次,无水硫酸钠干燥,减压浓缩后,残留物用硅胶色谱柱柱层析得到化合物(21B)(5.7g,产率94%)(21A) (5.0 g, 23.7 mmol) was dissolved in 5 mL of concentrated sulfuric acid, and the system was placed in an ice bath for 10 min. Then, a mixed solvent of 5 mL concentrated nitric acid and 15 mL concentrated sulfuric acid was slowly added dropwise. After the dropwise addition, the system automatically rose to room temperature and reacted for 3 hours. After the reaction of the raw materials is complete, pour the system into 100 mL of ice water to quench the reaction, add 100 mL of ethyl acetate and stir, then separate the liquids, extract the water phase with ethyl acetate (40 mL×2) once more, combine the ethyl acetate layers, and The ester layer was washed once with saturated sodium bicarbonate solution (40mL×1) and saturated brine (40mL×1), dried over anhydrous sodium sulfate, concentrated under reduced pressure, and the residue was chromatographed on a silica gel column to obtain compound (21B ) (5.7g, yield 94%)
第二步:3-溴-2,4,6-三氟苯胺(21C)The second step: 3-bromo-2,4,6-trifluoroaniline (21C)
3-bromo-2,4,6-trifluoroaniline.3-bromo-2,4,6-trifluoroaniline.
将化合物(21B)(5.7g,22.23mmol)溶解于50mL乙醇和10mL水的混合溶剂中,加入氯化铵(5.9g,111mmol)和铁粉(12.4g,222mmol),氮气保护下加热至85度反应1h。待原料反应完全后,体系冷却至室温,垫硅藻土滤掉不溶物,滤液减压浓缩,然后加入100mL 乙酸乙酯溶解,有机相分别用(50mL×1)水和饱和食盐水(40mL×1)洗涤一次,无水硫酸钠干燥,减压浓缩后,残留物用硅胶色谱柱柱层析得到化合物21C(3.7g,产率73.5%)。Compound (21B) (5.7g, 22.23mmol) was dissolved in a mixed solvent of 50mL ethanol and 10mL water, ammonium chloride (5.9g, 111mmol) and iron powder (12.4g, 222mmol) were added, and heated to 85 The degree of reaction is 1h. After the reaction of the raw materials is complete, the system is cooled to room temperature, and the insoluble matter is filtered out with a pad of diatomaceous earth. The filtrate is concentrated under reduced pressure, and then 100 mL of ethyl acetate is added to dissolve it. 1) Washing once, drying over anhydrous sodium sulfate, and concentrating under reduced pressure, the residue was chromatographed on a silica gel column to obtain compound 21C (3.7 g, yield 73.5%).
第三步:叔丁基(1R,5S)-3-(7-(3-氨基-2,4,6-三氟苯基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H)-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸酯(21D)The third step: tert-butyl (1R,5S)-3-(7-(3-amino-2,4,6-trifluorophenyl)-8-fluoro-2-(((2R,7aS)-2 -Fluorotetrahydro-1H)-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxy Ester (21D)
tert-butyl(1R,5S)-3-(7-(3-amino-2,4,6-trifluorophenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydr o-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate.tert-butyl(1R,5S)-3-(7-(3-amino-2,4,6-trifluorophenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydr o-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate.
将(21C)(0.072g,0.32mmol)溶解于乙腈10mL和水1mL的混合溶液中,加入9B(0.09g, 0.16mmol),氟化钾(0.035g,0.6mmol),Pd(PPh)2Cl2(0.011g,0.016mmol),氮气保护下100℃下反应12h,将反应冷却至室温,加入乙酸乙酯40mL和水25mL搅拌后分液,水相再用乙酸乙酯(40mL×1)萃取一次,合并乙酸乙酯层,乙酸乙酯层用饱和食盐水洗涤一次(40mL×1),无水硫酸钠干燥,减压浓缩后,残留物用硅胶色谱柱柱层析(DCM:MeOH=24:1)得到化合物 (21D)(0.063g,产率59%)Dissolve (21C) (0.072g, 0.32mmol) in a mixed solution of acetonitrile 10mL and water 1mL, add 9B (0.09g, 0.16mmol), potassium fluoride (0.035g, 0.6mmol), Pd(PPh) 2 Cl 2 (0.011g, 0.016mmol), reacted at 100°C for 12h under nitrogen protection, cooled the reaction to room temperature, added 40mL of ethyl acetate and 25mL of water and stirred, then separated the liquid, and extracted the aqueous phase with ethyl acetate (40mL×1) Once, the ethyl acetate layers were combined, and the ethyl acetate layer was washed once with saturated brine (40 mL×1), dried over anhydrous sodium sulfate, concentrated under reduced pressure, and the residue was chromatographed on a silica gel column (DCM: MeOH=24 :1) Obtain compound (21D) (0.063g, yield 59%)
LCMS m/z=661.2[M+H]+ LCMS m/z=661.2[M+H] +
第四步:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛基-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-2,4,6-三氟苯胺(化合物21)The fourth step: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octyl-3-yl)-8-fluoro-2-(((2R,7aS) -2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-2,4,6-trifluoroaniline (compound 21)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-2,4,6-trifluoroaniline.3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-2,4,6-trifluoroaniline.
将化合物(21D)(0.063g,0.095mmol)溶解于二氯甲烷5mL中,加入三氟乙酸0.5mL,室温下反应40分钟。减压浓缩反应液,然后加入二氯甲烷溶解,滴入0.5mL N,N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物21(13mg,产率24.3%),为白色粉末。Compound (21D) (0.063 g, 0.095 mmol) was dissolved in 5 mL of dichloromethane, 0.5 mL of trifluoroacetic acid was added, and the reaction was carried out at room temperature for 40 minutes. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 0.5mL N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, the residue is prepared and purified by HPLC, the preparation solution is concentrated and freeze-dried to obtain compound 21( 13 mg, yield 24.3%), as a white powder.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH 4 HCO 3 ); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min .
Ms m/z(ESI):561.2[M+H]+ Ms m/z(ESI):561.2[M+H] +
1H NMR(400MHz,CD3OD)δ7.80(d,1H),7.25–7.18(m,1H),6.96–6.87(m,1H),5.43–5.19(m,1H),4.52–4.44(m,2H),4.24(dd,2H),3.65–3.53(m,4H),3.27–3.11(m,3H),3.06–2.96(m,1H),2.42–2.09(m,3H),2.06–1.89(m,3H),1.87–1.75(m,4H). 1 H NMR (400MHz, CD 3 OD) δ7.80(d,1H),7.25–7.18(m,1H),6.96–6.87(m,1H),5.43–5.19(m,1H),4.52–4.44( m,2H),4.24(dd,2H),3.65–3.53(m,4H),3.27–3.11(m,3H),3.06–2.96(m,1H),2.42–2.09(m,3H),2.06– 1.89(m,3H),1.87–1.75(m,4H).
实施例22:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛基-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H- 吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-4-甲基-5-(三氟甲基)苯胺(化合物22)Example 22: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octyl-3-yl)-8-fluoro-2-(((2R,7aS) -2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-methyl-5-(trifluoromethyl)aniline (compound 22)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-methyl-5-(trifluoromethyl)aniline.3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-4-methyl-5-(trifluoromethyl)aniline.
第一步:3-溴-4-甲基-5-(三氟甲基)苯甲酸(22B)The first step: 3-bromo-4-methyl-5-(trifluoromethyl)benzoic acid (22B)
3-bromo-4-methyl-5-(trifluoromethyl)benzoic acid.3-bromo-4-methyl-5-(trifluoromethyl)benzoic acid.
将(22A)(5.0g,24.49mmol)溶解于浓硫酸50mL中,室温下加入N-溴代丁二酰亚胺(4.36 g,24.49mmol),然后室温反应3h。待原料反应完全后,将体系倾倒至100mL冰水中猝灭反应,加入乙酸乙酯100mL搅拌后分液,水相再用乙酸乙酯(40mL×2)萃取一次,合并乙酸乙酯层,乙酸乙酯层分别用饱和碳酸氢钠溶液(40mL×1)和饱和食盐水(40mL×1)洗涤一次,无水硫酸钠干燥,减压浓缩后,残留物用硅胶色谱柱柱层析得到化合物(22B)(7.1g,产率99%)Dissolve (22A) (5.0 g, 24.49 mmol) in 50 mL of concentrated sulfuric acid, add N-bromosuccinimide (4.36 g, 24.49 mmol) at room temperature, and react at room temperature for 3 h. After the reaction of the raw materials is complete, pour the system into 100 mL of ice water to quench the reaction, add 100 mL of ethyl acetate and stir, then separate the liquids, extract the water phase with ethyl acetate (40 mL×2), combine the ethyl acetate layers, and The ester layer was washed once with saturated sodium bicarbonate solution (40mL×1) and saturated brine (40mL×1), dried over anhydrous sodium sulfate, concentrated under reduced pressure, and the residue was chromatographed on a silica gel column to obtain compound (22B ) (7.1g, yield 99%)
LCMS m/z=283.1[M+H]+ LCMS m/z=283.1[M+H] +
第二步:(3-溴-4-甲基-5-(三氟甲基)苯基)氨基甲酸叔丁酯(22C)The second step: tert-butyl (3-bromo-4-methyl-5-(trifluoromethyl)phenyl)carbamate (22C)
tert-butyl(3-bromo-4-methyl-5-(trifluoromethyl)phenyl)carbamate.tert-butyl(3-bromo-4-methyl-5-(trifluoromethyl)phenyl)carbamate.
将(22B)(7.1g,24.49mmol)溶于甲苯(70mL)中,再加入三乙胺(10.5mL,73.47mmol) 和叠氮磷酸二苯酯(8.16mL,36.7mmol),体系置于室温搅拌30分钟,再加入叔丁醇(70mL),然后氮气保护,反应置于110℃搅拌2小时。将反应冷却至室温,减压浓缩后加入水(50mL),再用乙酸乙酯萃取(70mL×3),合并有机相后用饱和食盐水(40mL×1)洗涤一次,无水硫酸钠干燥,减压浓缩后,残留物用硅胶色谱柱柱层析得到化合物(22C)(7.9g,产率89%)Dissolve (22B) (7.1g, 24.49mmol) in toluene (70mL), then add triethylamine (10.5mL, 73.47mmol) and diphenylphosphoryl azide (8.16mL, 36.7mmol), and place the system at room temperature After stirring for 30 minutes, tert-butanol (70 mL) was added, and then under nitrogen protection, the reaction was stirred at 110° C. for 2 hours. The reaction was cooled to room temperature, concentrated under reduced pressure, added water (50 mL), and extracted with ethyl acetate (70 mL×3), combined the organic phases, washed once with saturated brine (40 mL×1), dried over anhydrous sodium sulfate, After concentration under reduced pressure, the residue was chromatographed on a silica gel column to obtain compound (22C) (7.9 g, yield 89%)
LCMS m/z=354.1[M+H]+ LCMS m/z=354.1[M+H] +
第三步:(4-甲基-3-(4,4,5,5-四甲基-1,3,2-二氧硼烷-2-基)-5-(三氟甲基)苯基)氨基甲酸叔丁酯 (22D)The third step: (4-methyl-3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-5-(trifluoromethyl)benzene base) tert-butyl carbamate (22D)
tert-butyl(4-methyl-3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-5-(trifluoromethyl)phenyl)c arbamate.tert-butyl(4-methyl-3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-5-(trifluoromethyl)phenyl)c arbamate.
氮气保护下,将(3-溴-4-甲基-5-(三氟甲基)苯基)氨基甲酸叔丁酯(22C)(2g,5.65mmol),联硼酸频那醇酯(1.6g,6.21mmol),醋酸钾(1.1g,11.3mmol),[1,1'-双(二苯基膦)二茂铁] 二氯化钯二氯甲烷络合物(461mg,0.57mmol)置于封管中,加入30mL 1,4-二氧六环,升温至90℃,反应5小时。将反应冷却至室温,然后加入水,乙酸乙酯萃取三次。合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物(22D)(1.8g,产率78%)。Under nitrogen protection, (3-bromo-4-methyl-5-(trifluoromethyl)phenyl) tert-butyl carbamate (22C) (2g, 5.65mmol), diboronic acid pinacol ester (1.6g , 6.21mmol), potassium acetate (1.1g, 11.3mmol), [1,1'-bis (diphenylphosphino) ferrocene] dichloropalladium dichloromethane complex (461mg, 0.57mmol) placed in In the sealed tube, 30 mL of 1,4-dioxane was added, the temperature was raised to 90°C, and the reaction was carried out for 5 hours. The reaction was cooled to room temperature, then water was added and extracted three times with ethyl acetate. The organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound (22D) (1.8 g, yield 78%).
Ms m/z(ESI):402.2[M+H]+ Ms m/z(ESI):402.2[M+H] +
第四步:叔丁基(1R,5S)-3-(7-(5-((叔丁氧基羰基)氨基)-2-甲基-3-(三氟甲基)苯基)-8-氟 -2-(((2R,7aS))-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷 -8-羧酸酯(22E)The fourth step: tert-butyl (1R,5S)-3-(7-(5-((tert-butoxycarbonyl)amino)-2-methyl-3-(trifluoromethyl)phenyl)-8 -Fluoro-2-(((2R,7aS))-2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-di Azabicyclo[3.2.1]octane-8-carboxylate (22E)
tert-butyl(1R,5S)-3-(7-(5-((tert-butoxycarbonyl)amino)-2-methyl-3-(trifluoromethyl)phenyl)-8-f luoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazab icyclo[3.2.1]octane-8-carboxylate.tert-butyl(1R,5S)-3-(7-(5-((tert-butoxycarbonyl)amino)-2-methyl-3-(trifluoromethyl)phenyl)-8-fluoro-2-(((2R, 7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazab icyclo[3.2.1]octane-8-carboxylate.
氮气保护下,将中间体22D(270mg,0.674mmol),2F(200mg,0.337mmol),[1,1'-双(二苯基膦)二茂铁]二氯化钯二氯甲烷络合物(55mg,0.067mmol)和碳酸铯(220mg,0.67mmol) 置于封管中,加入15mL1,4-二氧六环和1.5mL水。升温至105℃反应3小时。将反应冷却至室温,加水稀释,乙酸乙酯萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后残余物经过柱层析纯化,即得到化合物(22E)(170mg,产率64%)Under nitrogen protection, intermediate 22D (270mg, 0.674mmol), 2F (200mg, 0.337mmol), [1,1'-bis(diphenylphosphino)ferrocene]dichloropalladium dichloromethane complex (55mg, 0.067mmol) and cesium carbonate (220mg, 0.67mmol) were placed in a sealed tube, and 15mL of 1,4-dioxane and 1.5mL of water were added. The temperature was raised to 105° C. for 3 hours. The reaction was cooled to room temperature, diluted with water, extracted three times with ethyl acetate, the organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure, and the residue was purified by column chromatography to obtain compound (22E) (170 mg, yield 64%)
LCMS m/z=789.3[M+H]+ LCMS m/z=789.3[M+H] +
第五步:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛基-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-4-甲基-5-(三氟甲基)苯胺(化合物22)The fifth step: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octyl-3-yl)-8-fluoro-2-(((2R,7aS) -2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-methyl-5-(trifluoromethyl)aniline (compound 22)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-methyl-5-(trifluoromethyl)aniline.3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-4-methyl-5-(trifluoromethyl)aniline.
将化合物(22E)(0.17g,0.216mmol)溶解于二氯甲烷10mL中,加入三氟乙酸1mL,室温下反应40分钟。减压浓缩反应液,然后加入二氯甲烷溶解,滴入1mL N,N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物22(96mg,产率75.6%),为白色粉末。Compound (22E) (0.17 g, 0.216 mmol) was dissolved in 10 mL of dichloromethane, 1 mL of trifluoroacetic acid was added, and the reaction was carried out at room temperature for 40 minutes. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 1 mL of N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, prepare and purify the residue by HPLC, and freeze-dry the preparation solution to obtain compound 22 (96 mg , Yield 75.6%), white powder.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH 4 HCO 3 ); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min .
Ms m/z(ESI):589.3[M+H]+ Ms m/z(ESI):589.3[M+H] +
1H NMR(400MHz,CD3OD)δ7.80(d,1H),7.14–7.06(m,2H),6.78(d,1H),5.40–5.21(m,1H), 4.58–4.39(m,2H),4.24(dd,2H),3.67–3.53(m,4H),3.36–3.32(m,1H),3.27–3.17(m,2H), 3.06–2.96(m,1H),2.41–2.13(m,3H),2.10(s,3H),2.06–1.88(m,3H),1.87–1.76(m,4H). 1 H NMR (400MHz, CD 3 OD) δ7.80(d,1H),7.14–7.06(m,2H),6.78(d,1H),5.40–5.21(m,1H), 4.58–4.39(m, 2H),4.24(dd,2H),3.67–3.53(m,4H),3.36–3.32(m,1H),3.27–3.17(m,2H), 3.06–2.96(m,1H),2.41–2.13( m,3H),2.10(s,3H),2.06–1.88(m,3H),1.87–1.76(m,4H).
实施例23:4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛基-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)-7-(5-甲基-1H-吲唑-6-基)喹唑啉(化合物23)Example 23: 4-((1R,5S)-3,8-diazabicyclo[3.2.1]octyl-3-yl)-8-fluoro-2-(((2R,7aS)-2- Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)-7-(5-methyl-1H-indazol-6-yl)quinazoline (compound 23)
4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyr rolizin-7a(5H)-yl)methoxy)-7-(5-methyl-1H-indazol-6-yl)quinazoline4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyr rolizin-7a (5H)-yl)methoxy)-7-(5-methyl-1H-indazol-6-yl)quinazoline
第一步:叔丁基(1R,5S)-3-(8-氟-2-(((2R,7aS)-2-氟四氢-1H)-吡咯嗪-7a(5H)-yl)甲氧基)-7-(5-甲基-1H-吲唑-6-基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸酯(23B)The first step: tert-butyl (1R,5S)-3-(8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H)-pyrrolizine-7a(5H)-yl)methanol Oxy)-7-(5-methyl-1H-indazol-6-yl)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylic acid Esters (23B)
tert-butyl(1R,5S)-3-(8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methox y)-7-(5-methyl-1H-indazol-6-yl)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylatetert-butyl(1R,5S)-3-(8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methox y)-7-(5- methyl-1H-indazol-6-yl)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
氮气保护下,将6-溴-5-甲基-1H-吲唑23A(113mg,0.537mmol),9B(150mg,0.268mmol),Under nitrogen protection, 6-bromo-5-methyl-1H-indazole 23A (113mg, 0.537mmol), 9B (150mg, 0.268mmol),
[1,1'-双(二苯基膦)二茂铁]二氯化钯二氯甲烷络合物(22mg,0.027mmol)和碳酸铯(175mg, 0.537mmol)置于封管中,加入15mL1,4-二氧六环和1.5mL水。升温至105℃反应3小时。将反应冷却至室温,加水稀释,乙酸乙酯萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后残余物经过柱层析纯化,即得到化合物(23B)(150mg,产率90%)[1,1'-bis(diphenylphosphino)ferrocene]dichloropalladium dichloromethane complex (22mg, 0.027mmol) and cesium carbonate (175mg, 0.537mmol) were placed in a sealed tube, and 15mL1 , 4-dioxane and 1.5 mL of water. The temperature was raised to 105° C. for 3 hours. The reaction was cooled to room temperature, diluted with water, extracted three times with ethyl acetate, the organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure, and the residue was purified by column chromatography to obtain compound (23B) (150 mg, yield 90%)
LCMS m/z=646.3[M+H]+ LCMS m/z=646.3[M+H] +
第二步:4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛基-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪 -7a(5H)-基)甲氧基)-7-(5-甲基-1H-吲唑-6-基)喹唑啉(化合物23)The second step: 4-((1R,5S)-3,8-diazabicyclo[3.2.1]octyl-3-yl)-8-fluoro-2-(((2R,7aS)-2- Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)-7-(5-methyl-1H-indazol-6-yl)quinazoline (compound 23)
4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyr rolizin-7a(5H)-yl)methoxy)-7-(5-methyl-1H-indazol-6-yl)quinazoline4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyr rolizin-7a (5H)-yl)methoxy)-7-(5-methyl-1H-indazol-6-yl)quinazoline
将化合物(23B)(0.15g,0.23mmol)溶解于二氯甲烷10mL中,加入三氟乙酸1mL,室温下反应40分钟。减压浓缩反应液,然后加入二氯甲烷溶解,滴入1mL N,N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物23(73mg,产率58.2%),为白色粉末。Compound (23B) (0.15 g, 0.23 mmol) was dissolved in 10 mL of dichloromethane, 1 mL of trifluoroacetic acid was added, and the reaction was carried out at room temperature for 40 minutes. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 1 mL of N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, prepare and purify the residue by HPLC, and freeze-dry the preparation solution to obtain compound 23 (73 mg , yield 58.2%), white powder.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%CF3COOH);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% CF 3 COOH); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min.
Ms m/z(ESI):546.3[M+H]+ Ms m/z(ESI):546.3[M+H] +
1H NMR(400MHz,CD3OD)δ8.05(s,1H),7.93(d,1H),7.75(s,1H),7.43(s,1H),7.38(dd, 1H),5.67–5.48(m,1H),4.78–4.67(m,4H),4.25(s,2H),4.09–3.83(m,5H),3.53–3.44(m,1H), 2.77–2.54(m,2H),2.49–2.31(m,3H),2.26(s,3H),2.23–2.10(m,5H). 1 H NMR (400MHz, CD 3 OD) δ8.05(s,1H),7.93(d,1H),7.75(s,1H),7.43(s,1H),7.38(dd,1H),5.67–5.48 (m,1H),4.78–4.67(m,4H),4.25(s,2H),4.09–3.83(m,5H),3.53–3.44(m,1H), 2.77–2.54(m,2H),2.49 –2.31(m,3H),2.26(s,3H),2.23–2.10(m,5H).
实施例24:2-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛基-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H- 吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-6-氯-2',4'-二氟-[1,1'-联苯]-4-胺(化合物24)Example 24: 2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octyl-3-yl)-8-fluoro-2-(((2R,7aS) -2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-6-chloro-2',4'-difluoro-[1,1' -biphenyl]-4-amine (compound 24)
2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-6-chloro-2',4'-difluoro-[1,1'-biphenyl]-4-amine.2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-((((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-6-chloro-2',4'-difluoro-[1,1'-biphenyl]-4-amine.
第一步:2-溴-6-氯-2',4'-二氟-[1,1'-联苯]-4-胺(24B)The first step: 2-bromo-6-chloro-2',4'-difluoro-[1,1'-biphenyl]-4-amine (24B)
2-bromo-6-chloro-2',4'-difluoro-[1,1'-biphenyl]-4-amine.2-bromo-6-chloro-2',4'-difluoro-[1,1'-biphenyl]-4-amine.
氮气保护下,将中间体14B(500mg,1.5mmol),(2,4-二氟苯基)硼酸24A(285mg,1.8mmol),四三苯基膦钯(173mg,0.15mmol)和碳酸钾(414.6mg,3.0mmol)置于封管中,加入30mL1,4-二氧六环和6mL水。升温至100℃反应12小时。将反应冷却至室温,加水稀释,乙酸乙酯萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后残余物经过柱层析纯化,即得到化合物(24B)(507mg,产率88%)Under nitrogen protection, intermediate 14B (500mg, 1.5mmol), (2,4-difluorophenyl)boronic acid 24A (285mg, 1.8mmol), tetrakistriphenylphosphine palladium (173mg, 0.15mmol) and potassium carbonate ( 414.6mg, 3.0mmol) was placed in a sealed tube, and 30mL of 1,4-dioxane and 6mL of water were added. The temperature was raised to 100° C. for 12 hours. The reaction was cooled to room temperature, diluted with water, extracted three times with ethyl acetate, the organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure, and the residue was purified by column chromatography to obtain compound (24B) (507 mg, yield 88%)
LCMS m/z=317.9[M+H]+ LCMS m/z=317.9[M+H] +
第二步:叔丁基(1R,5S)-3-(7-(4-氨基-6-氯-2',4'-二氟-[1,1'-联苯]-2-基)-8-氟-2-(((2R,7aS)-2- 氟四氢-1H-pyrrolizin-7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸酯(24C)The second step: tert-butyl (1R,5S)-3-(7-(4-amino-6-chloro-2',4'-difluoro-[1,1'-biphenyl]-2-yl) -8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-di Azabicyclo[3.2.1]octane-8-carboxylate (24C)
tert-butyl(1R,5S)-3-(7-(4-amino-6-chloro-2',4'-difluoro-[1,1'-biphenyl]-2-yl)-8-fluoro-2-(((2R,7 aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]oc tane-8-carboxylate.tert-butyl(1R,5S)-3-(7-(4-amino-6-chloro-2',4'-difluoro-[1,1'-biphenyl]-2-yl)-8-fluoro-2 -(((2R,7 aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate .
将(24B)(0.145g,0.456mmol)溶解于乙腈10mL和水1mL的混合溶液中,加入9B(0.17 g,0.304mmol),氟化钾(0.066g,1.14mmol),Pd(PPh)2Cl2(0.025g,0.03mmol),氮气保护下100℃下反应12h,将反应冷却至室温,加入乙酸乙酯40mL和水25mL搅拌后分液,水相再用乙酸乙酯(40mL×1)萃取一次,合并乙酸乙酯层,乙酸乙酯层用饱和食盐水洗涤一次(40 mL×1),无水硫酸钠干燥,减压浓缩后,残留物用硅胶色谱柱柱层析分离纯化得到化合物(24C) (0.13g,产率74%)Dissolve (24B) (0.145g, 0.456mmol) in a mixed solution of acetonitrile 10mL and water 1mL, add 9B (0.17 g, 0.304mmol), potassium fluoride (0.066g, 1.14mmol), Pd(PPh)2Cl2( 0.025g, 0.03mmol), reacted at 100°C for 12h under the protection of nitrogen, cooled the reaction to room temperature, added 40mL of ethyl acetate and 25mL of water, stirred and separated the liquid, and extracted the water phase with ethyl acetate (40mL×1) once more, The ethyl acetate layers were combined, and the ethyl acetate layer was washed once with saturated brine (40 mL×1), dried over anhydrous sodium sulfate, concentrated under reduced pressure, and the residue was separated and purified by silica gel column chromatography to obtain compound (24C) (0.13g, yield 74%)
LCMS m/z=753.2[M+H]+ LCMS m/z=753.2[M+H] +
第三步:2-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛基-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-6-氯-2',4'-二氟-[1,1'-联苯]-4-胺(化合物24)The third step: 2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octyl-3-yl)-8-fluoro-2-(((2R,7aS) -2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-6-chloro-2',4'-difluoro-[1,1' -biphenyl]-4-amine (compound 24)
2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-6-chloro-2',4'-difluoro-[1,1'-biphenyl]-4-amine.2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-((((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-6-chloro-2',4'-difluoro-[1,1'-biphenyl]-4-amine.
将化合物(24C)(0.13g,0.17mmol)溶解于二氯甲烷10mL中,加入三氟乙酸1mL,室温下反应40分钟。减压浓缩反应液,然后加入二氯甲烷溶解,滴入1mL N,N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物24(30mg,产率27%),为白色粉末。Compound (24C) (0.13 g, 0.17 mmol) was dissolved in 10 mL of dichloromethane, 1 mL of trifluoroacetic acid was added, and the reaction was carried out at room temperature for 40 minutes. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 1 mL of N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, prepare and purify the residue by HPLC, and freeze-dry the prepared solution to obtain compound 24 (30 mg , yield 27%), as a white powder.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%CF3COOH);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% CF3COOH); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min.
Ms m/z(ESI):653.2[M+H]+ Ms m/z(ESI):653.2[M+H] +
1H NMR(400MHz,CD3OD)δ7.69(d,1H),7.20–7.14(m,1H),7.11–7.01(m,1H),6.96(d, 1H),6.79–6.69(m,2H),6.67(d,1H),5.65–5.46(m,1H),4.68–4.53(m,4H),4.20(s,2H),4.05– 3.71(m,5H),3.52–3.43(m,1H),2.75–2.51(m,2H),2.46–2.28(m,3H),2.20–2.06(m,5H). 1 H NMR (400MHz, CD 3 OD) δ7.69(d,1H),7.20–7.14(m,1H),7.11–7.01(m,1H),6.96(d,1H),6.79–6.69(m, 2H),6.67(d,1H),5.65–5.46(m,1H),4.68–4.53(m,4H),4.20(s,2H),4.05–3.71(m,5H),3.52–3.43(m, 1H),2.75–2.51(m,2H),2.46–2.28(m,3H),2.20–2.06(m,5H).
实施例25:5-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H- 吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-2-甲基-4-(三氟甲基)苯胺(化合物25)Example 25: 5-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3-yl)-8-fluoro-2-(((2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-2-methyl-4-(trifluoromethyl)aniline (compound 25)
5-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-2-methyl-4-(trifluoromethyl)aniline5-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-2-methyl-4-(trifluoromethyl)aniline
第一步:5-溴-2-碘-4-(三氟甲基)苯胺(25B)The first step: 5-bromo-2-iodo-4-(trifluoromethyl)aniline (25B)
5-bromo-2-iodo-4-(trifluoromethyl)aniline5-bromo-2-iodo-4-(trifluoromethyl)aniline
将3-溴-4-(三氟甲基)苯胺(25A)(4g,16.67mmol)溶于25mL乙酸,加入N-碘代丁二酰亚胺(4.12g,18.33mmol),室温下搅拌反应3小时。然后加入饱和碳酸氢钠水溶液调节pH至7,乙酸乙酯萃取三次。合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物(25B),为红褐色固体(5.77g,产率95%)Dissolve 3-bromo-4-(trifluoromethyl)aniline (25A) (4g, 16.67mmol) in 25mL of acetic acid, add N-iodosuccinimide (4.12g, 18.33mmol), and stir the reaction at room temperature 3 hours. Then, saturated aqueous sodium bicarbonate solution was added to adjust the pH to 7, and extracted three times with ethyl acetate. The organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound (25B) as a reddish-brown solid (5.77 g, yield 95%)
LCMS m/z=365.9[M+H]+ LCMS m/z=365.9[M+H] +
第二步:5-溴-2-甲基-4-(三氟甲基)苯胺(25C)The second step: 5-bromo-2-methyl-4-(trifluoromethyl)aniline (25C)
5-bromo-2-methyl-4-(trifluoromethyl)aniline5-bromo-2-methyl-4-(trifluoromethyl)aniline
将化合物25B(3g,8.22mmol),甲基硼酸(541mg,9.04mmol),[1,1'-双(二苯基膦)二茂铁]二氯化钯二氯甲烷络合物(671mg,0.82mmol)和碳酸铯(5.36g,16.44mmol)置于封管中,加入20mL1,4-二氧六环和5mL水,置换氮气保护。升温至60℃反应18小时。将反应冷却至室温,加水稀释,乙酸乙酯萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物(25C),为棕褐色固体(611mg,产率29%)Compound 25B (3g, 8.22mmol), methylboronic acid (541mg, 9.04mmol), [1,1'-bis(diphenylphosphino)ferrocene]dichloropalladium dichloromethane complex (671mg, 0.82mmol) and cesium carbonate (5.36g, 16.44mmol) were placed in a sealed tube, and 20mL of 1,4-dioxane and 5mL of water were added to replace the nitrogen protection. The temperature was raised to 60° C. for 18 hours. The reaction was cooled to room temperature, diluted with water, extracted three times with ethyl acetate, the organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound (25C) as a tan solid (611mg , yield 29%)
LCMS m/z=254.0[M+H]+ LCMS m/z=254.0[M+H] +
第三步:2-甲基-5-(4,4,5,5-四甲基-1,3,2-二氧杂硼-2-基)-4-(三氟甲基)苯胺(25D)The third step: 2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxabor-2-yl)-4-(trifluoromethyl)aniline ( 25D)
2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-4-(trifluoromethyl)aniline2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-4-(trifluoromethyl)aniline
将化合物25C(345mg,1.36mmol),联硼酸频那醇酯(517mg,2.03mmol),醋酸钾(400mg,4.08mmol),[1,1'-双(二苯基膦)二茂铁]二氯化钯二氯甲烷络合物(114mg,0.14mmol)置于封管中,加入5mL 1,4-二氧六环,置换氮气保护,升至90摄氏度下,反应6小时。将反应冷却至室温,然后加入水,乙酸乙酯萃取三次。合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物(25D),为棕色固体(232mg,产率56%)。Compound 25C (345mg, 1.36mmol), pinacol diboronate (517mg, 2.03mmol), potassium acetate (400mg, 4.08mmol), [1,1'-bis(diphenylphosphino)ferrocene] di Palladium chloride dichloromethane complex (114 mg, 0.14 mmol) was placed in a sealed tube, 5 mL of 1,4-dioxane was added, nitrogen was replaced, and the temperature was raised to 90 degrees Celsius for 6 hours of reaction. The reaction was cooled to room temperature, then water was added and extracted three times with ethyl acetate. The organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound (25D) as a brown solid (232 mg, yield 56%).
LCMS m/z=302.1[M+H]+ LCMS m/z=302.1[M+H] +
第四步:叔丁基(1R,5S)-3-(7-(5-氨基-4-甲基-2-(三氟甲基)苯基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)-喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸酯(25E) tert-butyl(1R,5S)-3-(7-(5-amino-4-methyl-2-(trifluoromethyl)phenyl)-8-fluoro-2-(((2R,7aS)-2-fluoro tetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carbo xylateThe fourth step: tert-butyl (1R,5S)-3-(7-(5-amino-4-methyl-2-(trifluoromethyl)phenyl)-8-fluoro-2-(((2R ,7aS)-2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)-quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane Alkane-8-carboxylate (25E) tert-butyl(1R,5S)-3-(7-(5-amino-4-methyl-2-(trifluoromethyl)phenyl)-8-fluoro-2-((( 2R,7aS)-2-fluoro tetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carbo xylate
将中间体2F(120mg,0.20mmol),化合物25D(117mg,0.39mmol),[1,1'-双(二苯基膦) 二茂铁]二氯化钯二氯甲烷络合物(17mg,0.02mmol)和碳酸铯(127mg,0.39mmol)置于封管中,加入2mL1,4-二氧六环和0.5mL水,置换氮气保护。升温至105℃反应3小时。将反应冷却至室温,加水稀释,乙酸乙酯萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物(25E),为棕褐色固体(123mg,产率90%)Intermediate 2F (120mg, 0.20mmol), compound 25D (117mg, 0.39mmol), [1,1'-bis(diphenylphosphine)ferrocene]dichloropalladium dichloromethane complex (17mg, 0.02mmol) and cesium carbonate (127mg, 0.39mmol) were placed in a sealed tube, and 2mL of 1,4-dioxane and 0.5mL of water were added to replace the nitrogen protection. The temperature was raised to 105° C. for 3 hours. The reaction was cooled to room temperature, diluted with water, extracted three times with ethyl acetate, the organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound (25E) as a tan solid (123mg , yield 90%)
LCMS m/z=689.3[M+H]+ LCMS m/z=689.3[M+H] +
第五步:5-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-2-甲基-4-(三氟甲基)苯胺(化合物25)The fifth step: 5-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3-yl)-8-fluoro-2-(((2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-2-methyl-4-(trifluoromethyl)aniline (compound 25)
5-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-2-methyl-4-(trifluoromethyl)aniline5-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-2-methyl-4-(trifluoromethyl)aniline
将化合物25E(123mg,0.18mmol)溶解于二氯甲烷2.5mL中,然后滴入0.5mL三氟乙酸,室温下反应3小时。减压浓缩反应液,然后加入二氯甲烷溶解,滴入0.5mL N,N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物25(45mg,产率42%),为白色粉末。Compound 25E (123 mg, 0.18 mmol) was dissolved in 2.5 mL of dichloromethane, then 0.5 mL of trifluoroacetic acid was added dropwise, and reacted at room temperature for 3 hours. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 0.5mL N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, and prepare and purify the residue by HPLC. The preparation solution is concentrated and freeze-dried to obtain compound 25( 45 mg, yield 42%), as a white powder.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH 4 HCO 3 ); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min .
LCMS m/z=589.3[M+H]+ LCMS m/z=589.3[M+H] +
1H NMR(400MHz,CD3OD)δ7.73(d,1H),7.38(s,1H),7.10(dd,1H),6.59(s,1H),5.40– 5.22(m,1H),4.55–4.40(m,2H),4.24(dd,2H),3.66–3.52(m,4H),3.26–3.13(m,3H),3.05– 2.96(m,1H),2.40–2.26(m,1H),2.23(s,3H),2.21–2.09(m,2H),2.05–1.94(m,2H),1.94– 1.75(m,5H). 1 H NMR (400MHz, CD 3 OD) δ7.73(d,1H),7.38(s,1H),7.10(dd,1H),6.59(s,1H),5.40–5.22(m,1H),4.55 –4.40(m,2H),4.24(dd,2H),3.66–3.52(m,4H),3.26–3.13(m,3H),3.05–2.96(m,1H),2.40–2.26(m,1H) ,2.23(s,3H),2.21–2.09(m,2H),2.05–1.94(m,2H),1.94–1.75(m,5H).
实施例26:4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)-7-(5-(三氟甲基)-1H-吲唑-6-基)喹唑啉(化合物26)Example 26: 4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluoro Tetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)-7-(5-(trifluoromethyl)-1H-indazol-6-yl)quinazoline (compound 26)
4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizi n-7a(5H)-yl)methoxy)-7-(5-(trifluoromethyl)-1H-indazol-6-yl)quinazoline4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-((((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizi n-7a (5H)-yl)methoxy)-7-(5-(trifluoromethyl)-1H-indazol-6-yl)quinazoline
第一步:6-溴-5-(三氟甲基)-1H-吲唑(26A)The first step: 6-bromo-5-(trifluoromethyl)-1H-indazole (26A)
6-bromo-5-(trifluoromethyl)-1H-indazole6-bromo-5-(trifluoromethyl)-1H-indazole
将化合物25C(616mg,2.44mmol)溶于6mL乙酸,置换氮气保护,置于冰浴下,缓慢加入亚硝酸钠(169mg,2.44mmol)的水溶液(1mL),然后置于室温下搅拌反应12小时。饱和碳酸氢钠调节pH至7,乙酸乙酯萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物26A,为棕褐色固体(292mg,产率45%)Compound 25C (616mg, 2.44mmol) was dissolved in 6mL of acetic acid, replaced with nitrogen, placed in an ice bath, slowly added an aqueous solution (1mL) of sodium nitrite (169mg, 2.44mmol), and then stirred at room temperature for 12 hours . Saturated sodium bicarbonate adjusted the pH to 7, extracted three times with ethyl acetate, combined the organic phases, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound 26A as a tan solid (292 mg, produced rate 45%)
LCMS m/z=265.0[M+H]+ LCMS m/z=265.0[M+H] +
第二步:叔丁基(1R,5S)-3-(8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)-7-(5-(三氟甲基)-1H-吲唑-6-基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸酯(26B)The second step: tert-butyl (1R,5S)-3-(8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy Base)-7-(5-(trifluoromethyl)-1H-indazol-6-yl)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8 -Carboxylate (26B)
tert-butyl(1R,5S)-3-(8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methox y)-7-(5-(trifluoromethyl)-1H-indazol-6-yl)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxyl atetert-butyl(1R,5S)-3-(8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methox y)-7-(5- (trifluoromethyl)-1H-indazol-6-yl)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
将中间体9B(190mg,0.37mmol),化合物26A(95mg,0.36mmol),三(二亚苄-BASE丙酮)二钯(37mg,0.04mmol),2-(二环己基膦)-3,6-二甲氧基-2'-4'-6'-三-I-丙基-11'-联苯(43mg, 0.08mmol)和碳酸钠(76mg,0.72mmol)置于封管中,加入2mL1,4-二氧六环和0.5mL水,置换氮气保护。升温至120℃反应3小时。将反应冷却至室温,加水稀释,乙酸乙酯萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物26B,为棕褐色固体(58mg,产率23%)Intermediate 9B (190mg, 0.37mmol), compound 26A (95mg, 0.36mmol), tris(dibenzylidene-BASEacetone)dipalladium (37mg, 0.04mmol), 2-(dicyclohexylphosphine)-3,6 -Dimethoxy-2'-4'-6'-tri-I-propyl-11'-biphenyl (43mg, 0.08mmol) and sodium carbonate (76mg, 0.72mmol) were placed in a sealed tube, and 2mL1 , 4-dioxane and 0.5mL of water, replace the nitrogen protection. The temperature was raised to 120° C. for 3 hours to react. The reaction was cooled to room temperature, diluted with water, extracted three times with ethyl acetate, the organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound 26B as a tan solid (58 mg, produced rate 23%)
LCMS m/z=700.3[M+H]+ LCMS m/z=700.3[M+H] +
第三步:4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)-7-(5-(三氟甲基)-1H-吲唑-6-基)喹唑啉(化合物26)The third step: 4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluoro Tetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)-7-(5-(trifluoromethyl)-1H-indazol-6-yl)quinazoline (compound 26)
4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyr rolizin-7a(5H)-yl)methoxy)-7-(5-(trifluoromethyl)-1H-indazol-6-yl)quinazoline4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyr rolizin-7a (5H)-yl)methoxy)-7-(5-(trifluoromethyl)-1H-indazol-6-yl)quinazoline
将化合物26B(58mg,0.083mmol)溶解于二氯甲烷2.5mL中,然后滴入0.5mL三氟乙酸,室温下反应60分钟。减压浓缩反应液,然后加入二氯甲烷溶解,滴入0.5mL N,N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物26(20mg,产率40%)。Compound 26B (58 mg, 0.083 mmol) was dissolved in 2.5 mL of dichloromethane, then 0.5 mL of trifluoroacetic acid was added dropwise, and reacted at room temperature for 60 minutes. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 0.5mL N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, the residue is prepared and purified by HPLC, the preparation solution is concentrated and freeze-dried to obtain compound 26( 20 mg, yield 40%).
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH4HCO3); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min.
LCMS m/z=600.2[M+H]+ LCMS m/z=600.2[M+H] +
实施例27:6-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H- 吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-4-(三氟甲基)吡啶-2-胺(化合物27)Example 27: 6-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3-yl)-8-fluoro-2-(((2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)pyridin-2-amine (compound 27)
6-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)pyridin-2-amine6-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)pyridin-2-amine
第一步:6-氯-4-(三氟甲基)吡啶-2-胺(27B)The first step: 6-chloro-4-(trifluoromethyl)pyridin-2-amine (27B)
6-chloro-4-(trifluoromethyl)pyridin-2-amine6-chloro-4-(trifluoromethyl)pyridin-2-amine
将化合物27A(2.5g,11.58mmol),置于高压釜中,加入70mL氨水,升温至100℃反应24小时。将反应冷却至室温,加水稀释,二氯甲烷萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物(27A),为无色油状(585mg,产率26%)Compound 27A (2.5 g, 11.58 mmol) was placed in an autoclave, 70 mL of ammonia water was added, and the temperature was raised to 100° C. for 24 hours. The reaction was cooled to room temperature, diluted with water, extracted three times with dichloromethane, the organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound (27A) as a colorless oil (585mg , yield 26%)
LCMS m/z=197.1[M+H]+ LCMS m/z=197.1[M+H] +
第二步:叔丁基(1R,5S)-3-(7-(6-氨基-4-(三氟甲基)吡啶-2-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸酯(27C)The second step: tert-butyl (1R, 5S)-3-(7-(6-amino-4-(trifluoromethyl)pyridin-2-yl)-8-fluoro-2-(((2R,7aS )-2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8 - Carboxylate (27C)
tert-butyl(1R,5S)-3-(7-(6-amino-4-(trifluoromethyl)pyridin-2-yl)-8-fluoro-2-(((2R,7aS)-2-fluoro tetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carbo xylatetert-butyl(1R,5S)-3-(7-(6-amino-4-(trifluoromethyl)pyridin-2-yl)-8-fluoro-2-(((2R,7aS)-2-fluoro tetrahydro- 1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carbo xylate
将中间体9B(160mg,0.29mmol),化合物27B(69mg,0.35mmol),双三苯基磷二氯化钯(21mg,0.03mmol),和氟化钾(63mg,1.08mmol)置于封管中,加入2mL乙腈和0.5mL 水,置换氮气保护。升温至100℃反应18小时。将反应冷却至室温,加水稀释,乙酸乙酯萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物 27C,为黄色固体(92mg,产率48%)Intermediate 9B (160 mg, 0.29 mmol), compound 27B (69 mg, 0.35 mmol), bistriphenylphosphine palladium dichloride (21 mg, 0.03 mmol), and potassium fluoride (63 mg, 1.08 mmol) were placed in a locked tube In, add 2mL of acetonitrile and 0.5mL of water to replace the nitrogen protection. The temperature was raised to 100° C. for 18 hours. The reaction was cooled to room temperature, diluted with water, extracted three times with ethyl acetate, the organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound 27C as a yellow solid (92mg, yield 48%)
LCMS m/z=676.3[M+H]+ LCMS m/z=676.3[M+H] +
第三步:6-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-4-(三氟甲基)吡啶-2-胺(化合物27)The third step: 6-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3-yl)-8-fluoro-2-(((2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)pyridin-2-amine (compound 27)
6-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)pyridin-2-amine6-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)pyridin-2-amine
将化合物27C(92mg,0.136mmol)溶解于二氯甲烷2.5mL中,然后滴入0.5mL三氟乙酸,室温下反应2小时。减压浓缩反应液,然后加入二氯甲烷溶解,滴入0.5mL N,N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物27(17mg,产率22%),为白色粉末。Compound 27C (92 mg, 0.136 mmol) was dissolved in 2.5 mL of dichloromethane, then 0.5 mL of trifluoroacetic acid was added dropwise, and reacted at room temperature for 2 hours. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 0.5mL N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, the residue is prepared and purified by HPLC, the preparation solution is concentrated and freeze-dried to obtain compound 27( 17 mg, yield 22%), as a white powder.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH 4 HCO 3 ); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min .
LCMS m/z=576.3[M+H]+ LCMS m/z=576.3[M+H] +
1H NMR(400MHz,CD3OD)δ7.83–7.73(m,2H),7.29(s,1H),6.82(s,1H),5.40–5.21(m,1H), 4.52–4.42(m,2H),4.24(dd,2H),3.63–3.54(m,4H),3.27–3.12(m,3H),3.05–2.96(m,1H), 2.40–2.12(m,3H),2.05–1.94(m,2H),1.94–1.75(m,5H). 1 H NMR (400MHz, CD 3 OD) δ7.83–7.73(m,2H),7.29(s,1H),6.82(s,1H),5.40–5.21(m,1H), 4.52–4.42(m, 2H),4.24(dd,2H),3.63–3.54(m,4H),3.27–3.12(m,3H),3.05–2.96(m,1H), 2.40–2.12(m,3H),2.05–1.94( m,2H),1.94–1.75(m,5H).
实施例28:2-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H- 吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-4-氨基苄腈(化合物28)Example 28: 2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3-yl)-8-fluoro-2-(((2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-aminobenzonitrile (compound 28)
2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-aminobenzonitrile2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-((((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-4-aminobenzonitrile
第一步:4-氨基-2-(4,4,5,5-四甲基-1,3,2-二氧杂硼-2-基)苄腈(28B)The first step: 4-amino-2-(4,4,5,5-tetramethyl-1,3,2-dioxaborin-2-yl)benzonitrile (28B)
4-amino-2-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzonitrile4-amino-2-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzonitrile
将4-氨基-2-溴苄腈(28A)(1.65g,8.33mmol),联硼酸频那醇酯(3.17g,12.50mmol),醋酸钾(3.27g,33.32mmol),[1,1'-双(二苯基膦)二茂铁]二氯化钯二氯甲烷络合物(680mg, 0.83mmol)置于封管中,加入15mL 1,4-二氧六环,置换氮气保护,升至90摄氏度下,反应 5小时。将反应冷却至室温,然后加入水,乙酸乙酯萃取三次。合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物28B,为黄色油状(1.6g,产率79%)。4-amino-2-bromobenzonitrile (28A) (1.65g, 8.33mmol), biboronic acid pinacol ester (3.17g, 12.50mmol), potassium acetate (3.27g, 33.32mmol), [1,1' -Bis(diphenylphosphino)ferrocene]palladium dichloride dichloromethane complex (680mg, 0.83mmol) was placed in a sealed tube, and 15mL 1,4-dioxane was added to replace the nitrogen protection. To 90 degrees Celsius, react for 5 hours. The reaction was cooled to room temperature, then water was added and extracted three times with ethyl acetate. The organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound 28B as a yellow oil (1.6 g, yield 79%).
LCMS m/z=245.2[M+H]+ LCMS m/z=245.2[M+H] +
第二步:叔丁基(1R,5S)-3-(7-(5-氨基-2-氰基苯基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H)-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸酯(28C)The second step: tert-butyl (1R,5S)-3-(7-(5-amino-2-cyanophenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro -1H)-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate (28C )
tert-butyl(1R,5S)-3-(7-(5-amino-2-cyanophenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrol izin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylatetert-butyl(1R,5S)-3-(7-(5-amino-2-cyanophenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrol izin-7a(5H )-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
将中间体2F(120mg,0.20mmol),化合物28B(98mg,0.40mmol),[1,1'-双(二苯基膦) 二茂铁]二氯化钯二氯甲烷络合物(16mg,0.02mmol)和碳酸铯(130mg,0.40mmol)置于封管中,加入2.5mL1,4-二氧六环和0.5mL水,置换氮气保护。升温至105℃反应3小时。将反应冷却至室温,加水稀释,乙酸乙酯萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物28C,为棕色固体(116mg,产率92%)Intermediate 2F (120mg, 0.20mmol), compound 28B (98mg, 0.40mmol), [1,1'-bis(diphenylphosphino)ferrocene]dichloropalladium dichloromethane complex (16mg, 0.02mmol) and cesium carbonate (130mg, 0.40mmol) were placed in a sealed tube, and 2.5mL of 1,4-dioxane and 0.5mL of water were added to replace the nitrogen protection. The temperature was raised to 105° C. for 3 hours. The reaction was cooled to room temperature, diluted with water, extracted three times with ethyl acetate, the organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound 28C as a brown solid (116 mg, yield 92%)
LCMS m/z=632.3[M+H]+ LCMS m/z=632.3[M+H] +
第三步:2-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-4-氨基苄腈(化合物28)The third step: 2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3-yl)-8-fluoro-2-(((2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-aminobenzonitrile (compound 28)
2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-aminobenzonitrile2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-((((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-4-aminobenzonitrile
将化合物28C(116mg,0.18mmol)溶解于二氯甲烷2.5mL中,然后滴入0.5mL三氟乙酸,室温下反应2小时。减压浓缩反应液,然后加入二氯甲烷溶解,滴入0.5mL N,N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物28(39mg,产率41%),为白色粉末。Compound 28C (116 mg, 0.18 mmol) was dissolved in 2.5 mL of dichloromethane, then 0.5 mL of trifluoroacetic acid was added dropwise, and reacted at room temperature for 2 hours. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 0.5mL N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, the residue is prepared and purified by HPLC, the preparation solution is concentrated and freeze-dried to obtain compound 28 ( 39 mg, yield 41%), as a white powder.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH4HCO3); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min.
LCMS m/z=532.3[M+H]+ LCMS m/z=532.3[M+H] +
1H NMR(400MHz,CD3OD)δ7.81(d,1H),7.50(d,1H),7.22(dd,1H),6.78–6.70(m,2H), 5.40–5.22(m,1H),4.53–4.44(m,2H),4.24(dd,2H),3.64–3.52(m,4H),3.27–3.11(m,3H), 3.06–2.96(m,1H),2.40–2.10(m,3H),2.05–1.94(m,2H),1.93–1.76(m,5H). 1 H NMR (400MHz, CD 3 OD) δ7.81(d,1H),7.50(d,1H),7.22(dd,1H),6.78–6.70(m,2H), 5.40–5.22(m,1H) ,4.53–4.44(m,2H),4.24(dd,2H),3.64–3.52(m,4H),3.27–3.11(m,3H), 3.06–2.96(m,1H),2.40–2.10(m, 3H),2.05–1.94(m,2H),1.93–1.76(m,5H).
实施例29:5-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H- 吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-2-环丙基-4-(三氟甲基)苯胺(化合物29)Example 29: 5-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3-yl)-8-fluoro-2-(((2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-2-cyclopropyl-4-(trifluoromethyl)aniline (compound 29)
5-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-2-cyclopropyl-4-(trifluoromethyl)aniline5-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-2-cyclopropyl-4-(trifluoromethyl)aniline
第一步:5-溴-2-环丙基-4-(三氟甲基)苯胺(29A)The first step: 5-bromo-2-cyclopropyl-4-(trifluoromethyl)aniline (29A)
5-bromo-2-cyclopropyl-4-(trifluoromethyl)aniline5-bromo-2-cyclopropyl-4-(trifluoromethyl)aniline
将化合物25B(1g,2.73mmol),环丙基硼酸(352mg,4.10mmol),[1,1'-双(二苯基膦)二茂铁]二氯化钯二氯甲烷络合物(221mg,0.27mmol)和碳酸铯(1.34g,4.10mmol)置于封管中,加入15mL1,4-二氧六环和4mL水,置换氮气保护。升温至80℃反应18小时。将反应冷却至室温,加水稀释,乙酸乙酯萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物29A,为黄色固体(181mg,产率24%)Compound 25B (1g, 2.73mmol), cyclopropylboronic acid (352mg, 4.10mmol), [1,1'-bis(diphenylphosphino)ferrocene]dichloropalladium dichloromethane complex (221mg , 0.27mmol) and cesium carbonate (1.34g, 4.10mmol) were placed in a sealed tube, and 15mL of 1,4-dioxane and 4mL of water were added to replace the nitrogen protection. The temperature was raised to 80° C. for 18 hours. The reaction was cooled to room temperature, diluted with water, extracted three times with ethyl acetate, the organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound 29A as a yellow solid (181 mg, yield twenty four%)
LCMS m/z=280.0[M+H]+ LCMS m/z=280.0[M+H] +
第二步:叔丁基(1R,5S)-3-(7-(5-氨基-4-环丙基-2-(三氟甲基)苯基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸酯(29B)The second step: tert-butyl (1R, 5S)-3-(7-(5-amino-4-cyclopropyl-2-(trifluoromethyl)phenyl)-8-fluoro-2-((( 2R,7aS)-2-fluorotetrahydro-1H-pyrrolidin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane Alkane-8-carboxylate (29B)
tert-butyl(1R,5S)-3-(7-(5-amino-4-cyclopropyl-2-(trifluoromethyl)phenyl)-8-fluoro-2-(((2R,7aS )-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octa ne-8-carboxylatetert-butyl(1R,5S)-3-(7-(5-amino-4-cyclopropyl-2-(trifluoromethyl)phenyl)-8-fluoro-2-(((2R,7aS )-2-fluorotetrahydro-1H -pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octa ne-8-carboxylate
将中间体9B(185mg,0.33mmol),化合物29A(62mg,0.22mmol),[1,1'-双(二苯基膦) 二茂铁]二氯化钯二氯甲烷络合物(16mg,0.02mmol)和碳酸铯(143mg,0.44mmol)置于封管中,加入2mL1,4-二氧六环和0.5mL水,置换氮气保护。升温至110℃反应3小时。将反应冷却至室温,加水稀释,乙酸乙酯萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物29B,为棕褐色固体(133mg,产率83%)Intermediate 9B (185mg, 0.33mmol), compound 29A (62mg, 0.22mmol), [1,1'-bis(diphenylphosphino)ferrocene]dichloropalladium dichloromethane complex (16mg, 0.02mmol) and cesium carbonate (143mg, 0.44mmol) were placed in a sealed tube, and 2mL of 1,4-dioxane and 0.5mL of water were added to replace the nitrogen protection. The temperature was raised to 110° C. for 3 hours. The reaction was cooled to room temperature, diluted with water, extracted three times with ethyl acetate, the organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound 29B as a tan solid (133 mg, produced rate 83%)
LCMS m/z=715.3[M+H]+ LCMS m/z=715.3[M+H] +
第三步:5-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-2-环丙基-4-(三氟甲基)苯胺(化合物29)The third step: 5-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3-yl)-8-fluoro-2-(((2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-2-cyclopropyl-4-(trifluoromethyl)aniline (compound 29)
5-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-2-cyclopropyl-4-(trifluoromethyl)aniline5-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-2-cyclopropyl-4-(trifluoromethyl)aniline
将化合物29B(133mg,0.19mmol)溶解于二氯甲烷2.5mL中,然后滴入0.5mL三氟乙酸,室温下反应2小时。减压浓缩反应液,然后加入二氯甲烷溶解,滴入0.5mL N,N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物29(48mg,产率41%),为白色粉末。Compound 29B (133 mg, 0.19 mmol) was dissolved in 2.5 mL of dichloromethane, then 0.5 mL of trifluoroacetic acid was added dropwise, and reacted at room temperature for 2 hours. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 0.5mL N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, the residue is prepared and purified by HPLC, the preparation solution is concentrated and freeze-dried to obtain compound 29( 48 mg, yield 41%), as a white powder.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH4HCO3); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min.
LCMS m/z=615.2[M+H]+ LCMS m/z=615.2[M+H] +
1H NMR(400MHz,CD3OD)δ7.84(d,1H),7.33–7.25(m,2H),6.61(s,1H),5.65–5.47(m, 1H),4.75–4.63(m,4H),4.24(s,2H),4.08–3.81(m,5H),3.52–3.43(m,1H),2.78–2.53(m,2H), 2.48–2.30(m,3H),2.23–2.10(m,5H),1.82–1.72(m,1H),1.05–0.95(m,2H),0.68–0.58(m, 2H). 1 H NMR (400MHz, CD 3 OD) δ7.84(d,1H),7.33–7.25(m,2H),6.61(s,1H),5.65–5.47(m,1H),4.75–4.63(m, 4H),4.24(s,2H),4.08–3.81(m,5H),3.52–3.43(m,1H),2.78–2.53(m,2H), 2.48–2.30(m,3H),2.23–2.10( m,5H),1.82–1.72(m,1H),1.05–0.95(m,2H),0.68–0.58(m,2H).
实施例30:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H- 吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-5-氯-4-(环戊-1-烯-1-基)苯胺(化合物30)Example 30: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3-yl)-8-fluoro-2-(((2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-chloro-4-(cyclopent-1-en-1-yl)aniline (compound 30)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-chloro-4-(cyclopent-1-en-1-yl)aniline3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-5-chloro-4-(cyclopent-1-en-1-yl)aniline
第一步:3-溴-5-氯-4-碘苯胺(30B)The first step: 3-bromo-5-chloro-4-iodoaniline (30B)
3-bromo-5-chloro-4-iodoaniline3-bromo-5-chloro-4-iodoaniline
将3-溴-5-氯苯胺(30A)(5g,24.22mmol)溶于25mL N,N-二甲基甲酰胺,加入N-碘代丁二酰亚胺(5.99g,26.64mmol),室温下搅拌反应3小时。然后加入饱和硫代硫酸钠溶液,乙酸乙酯萃取三次。合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物(30B),为红褐色固体(7.49g,产率93%)Dissolve 3-bromo-5-chloroaniline (30A) (5g, 24.22mmol) in 25mL of N,N-dimethylformamide, add N-iodosuccinimide (5.99g, 26.64mmol), room temperature The reaction was stirred for 3 hours. Then a saturated sodium thiosulfate solution was added and extracted three times with ethyl acetate. The organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound (30B) as a reddish-brown solid (7.49 g, yield 93%)
LCMS m/z=331.8[M+H]+ LCMS m/z=331.8[M+H] +
第二步:3-溴-5-氯-4-(环戊-1-烯-1-基)苯胺(30C)The second step: 3-bromo-5-chloro-4-(cyclopent-1-en-1-yl)aniline (30C)
3-bromo-5-chloro-4-(cyclopent-1-en-1-yl)aniline3-bromo-5-chloro-4-(cyclopent-1-en-1-yl)aniline
将化合物30B(1g,3.01mmol),环戊烯-1-基硼酸(438mg,3.91mmol),[1,1'-双(二苯基膦)二茂铁]二氯化钯二氯甲烷络合物(245mg,0.30mmol)和碳酸铯(1.96g,6.02mmol)置于封管中,加入15mL1,4-二氧六环和4mL水,置换氮气保护。升温至80℃反应8小时。将反应冷却至室温,加水稀释,乙酸乙酯萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物30C,为褐色固体(780mg,产率95%)Compound 30B (1g, 3.01mmol), cyclopenten-1-ylboronic acid (438mg, 3.91mmol), [1,1'-bis(diphenylphosphino)ferrocene]dichloropalladium dichloromethane Compound (245mg, 0.30mmol) and cesium carbonate (1.96g, 6.02mmol) were placed in a sealed tube, and 15mL of 1,4-dioxane and 4mL of water were added to replace the nitrogen protection. The temperature was raised to 80° C. for 8 hours. The reaction was cooled to room temperature, diluted with water, extracted three times with ethyl acetate, the organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound 30C as a brown solid (780 mg, yield 95%)
LCMS m/z=272.0[M+H]+ LCMS m/z=272.0[M+H] +
第三步:叔丁基(1R,5S)-3-(7-(5-氨基-3-氯-2-(环戊-1-烯-1-基)苯基)-8-氟-2-(((2R,7aS)-2- 氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸酯(30D)The third step: tert-butyl (1R,5S)-3-(7-(5-amino-3-chloro-2-(cyclopent-1-en-1-yl)phenyl)-8-fluoro-2 -(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2 .1] Octane-8-carboxylate (30D)
tert-butyl(1R,5S)-3-(7-(5-amino-3-chloro-2-(cyclopent-1-en-1-yl)phenyl)-8-fluoro-2-(((2R,7aS) -2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octan e-8-carboxylatetert-butyl(1R,5S)-3-(7-(5-amino-3-chloro-2-(cyclopent-1-en-1-yl)phenyl)-8-fluoro-2-(((2R, 7aS) -2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octan e-8-carboxylate
将中间体9B(185mg,0.33mmol),化合物30C(60mg,0.22mmol),[1,1'-双(二苯基膦) 二茂铁]二氯化钯二氯甲烷络合物(16mg,0.02mmol)和碳酸铯(143mg,0.44mmol)置于封管中,加入2mL1,4-二氧六环和0.5mL水,置换氮气保护。升温至110℃反应3小时。将反应冷却至室温,加水稀释,乙酸乙酯萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物30D,为棕褐色固体(112mg,产率72%)Intermediate 9B (185mg, 0.33mmol), compound 30C (60mg, 0.22mmol), [1,1'-bis(diphenylphosphino)ferrocene]dichloropalladium dichloromethane complex (16mg, 0.02mmol) and cesium carbonate (143mg, 0.44mmol) were placed in a sealed tube, and 2mL of 1,4-dioxane and 0.5mL of water were added to replace the nitrogen protection. The temperature was raised to 110° C. for 3 hours. The reaction was cooled to room temperature, diluted with water, extracted three times with ethyl acetate, the organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound 30D as a tan solid (112 mg, produced rate 72%)
LCMS m/z=707.3[M+H]+ LCMS m/z=707.3[M+H] +
第四步:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-5-氯-4-(环戊-1-烯-1-基)苯胺(化合物30)The fourth step: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3-yl)-8-fluoro-2-(((2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-chloro-4-(cyclopent-1-en-1-yl)aniline (compound 30)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-chloro-4-(cyclopent-1-en-1-yl)aniline3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-5-chloro-4-(cyclopent-1-en-1-yl)aniline
将化合物30D(112mg,0.16mmol)溶解于二氯甲烷2.5mL中,然后滴入0.5mL三氟乙酸,室温下反应2小时。减压浓缩反应液,然后加入二氯甲烷溶解,滴入0.5mL N,N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物30(30mg,产率31%),为白色粉末。Compound 30D (112 mg, 0.16 mmol) was dissolved in 2.5 mL of dichloromethane, then 0.5 mL of trifluoroacetic acid was added dropwise, and reacted at room temperature for 2 hours. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 0.5mL N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, the residue is prepared and purified by HPLC, the preparation solution is concentrated and freeze-dried to obtain compound 30( 30 mg, yield 31%), as a white powder.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH4HCO3); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min.
LCMS m/z=607.3[M+H]+ LCMS m/z=607.3[M+H] +
实施例31:N-(3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢 -1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-4-(三氟甲基)苯基)乙酰胺(化合物31)Example 31: N-(3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3-yl)-8-fluoro-2-(((2R, 7aS)-2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)phenyl)acetamide (compound 31 )
N-(3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1 H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)phenyl)acetamideN-(3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro- 1 H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)phenyl)acetamide
第一步:N-[3-溴-4-(三氟甲基)苯基]乙酰胺(31A)The first step: N-[3-bromo-4-(trifluoromethyl)phenyl]acetamide (31A)
N-[3-bromo-4-(trifluoromethyl)phenyl]acetamideN-[3-bromo-4-(trifluoromethyl)phenyl]acetamide
将原料3-溴-4-三氟甲基苯胺(1.0g,4.2mmol)溶于5ml二氯甲烷中,加入三乙胺(0.848 g,8.4mmol)乙酰氯(0.494g,6.3mmol),室温搅拌2h,反应完成后,减压浓缩,柱层析得到化合物31A(1.0g,产率85.12%)。Dissolve the raw material 3-bromo-4-trifluoromethylaniline (1.0g, 4.2mmol) in 5ml of dichloromethane, add triethylamine (0.848g, 8.4mmol) acetyl chloride (0.494g, 6.3mmol), room temperature After stirring for 2 h, after the reaction was completed, it was concentrated under reduced pressure, and column chromatography gave compound 31A (1.0 g, yield 85.12%).
LCMS m/z=282.1[M+H]+ LCMS m/z=282.1[M+H] +
第二步:叔丁基(1R,5S)-3-(7-(5-乙酰氨基-2-(三氟甲基)苯基]-8-氟-2-(((2R,7aS)-2-氟四氢 -1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸酯(31B)The second step: tert-butyl (1R,5S)-3-(7-(5-acetylamino-2-(trifluoromethyl)phenyl]-8-fluoro-2-(((2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxy Ester (31B)
tert-butyl(1R,5S)-3-(7-(5-acetamido-2-(trifluoromethyl)phenyl)-8-fluoro-2-(((2R,7aS)-2-fluorot etrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carbo xylatetert-butyl(1R,5S)-3-(7-(5-acetamido-2-(trifluoromethyl)phenyl)-8-fluoro-2-(((2R,7aS)-2-fluorot etrahydro-1H-pyrrolizin- 7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carbo xylate
将(31A)(0.15g,0.52mmol)溶解于1,4-二氧六环5mL和水1mL的混合溶液中,加入9B(0.23g,0.42mmol),碳酸铯(0.3g,1.04mmol),Pd(dppf)Cl2(0.038g,0.052mmol),氮气保护下100℃下反应2h,将反应冷却至室温,加入乙酸乙酯40mL和水25mL,水相再用乙酸乙酯(40mL×1)萃取一次,合并乙酸乙酯层,乙酸乙酯层用饱和食盐水洗涤一次(40mL×1),无水硫酸钠干燥,减压浓缩后,残留物用硅胶色谱柱柱层析得到化合物(31B)(0.11g,产率36.51%)Dissolve (31A) (0.15g, 0.52mmol) in a mixed solution of 1,4-dioxane 5mL and water 1mL, add 9B (0.23g, 0.42mmol), cesium carbonate (0.3g, 1.04mmol), Pd(dppf)Cl 2 (0.038g, 0.052mmol), reacted at 100°C under nitrogen protection for 2h, cooled the reaction to room temperature, added 40mL of ethyl acetate and 25mL of water, and then used ethyl acetate (40mL×1) for the aqueous phase Extract once, combine the ethyl acetate layers, wash the ethyl acetate layer once with saturated brine (40mL×1), dry over anhydrous sodium sulfate, concentrate under reduced pressure, and the residue is chromatographed on a silica gel column to obtain compound (31B) (0.11g, yield 36.51%)
LCMS m/z=717.3[M+H]+ LCMS m/z=717.3[M+H] +
第三步:N-(3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H- 吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-4-(三氟甲基)苯基)乙酰胺(化合物31)The third step: N-(3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3-yl)-8-fluoro-2-(((2R, 7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)phenyl)acetamide (compound 31 )
N-(3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1 H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)phenyl)acetamideN-(3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro- 1 H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)phenyl)acetamide
将化合物(31B)(0.11g,0.15mmol)溶解于二氯甲烷5mL中,加入三氟乙酸0.5mL,室温下反应40分钟。减压浓缩反应液,然后加入二氯甲烷溶解,滴入0.5mL N,N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物31(20mg,产率21.64%),为白色粉末。Compound (31B) (0.11 g, 0.15 mmol) was dissolved in 5 mL of dichloromethane, 0.5 mL of trifluoroacetic acid was added, and the reaction was carried out at room temperature for 40 minutes. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 0.5mL N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, prepare and purify the residue by HPLC, and freeze-dry the prepared solution to obtain compound 31( 20 mg, yield 21.64%), as a white powder.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH 4 HCO 3 ); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min .
Ms m/z(ESI):617.2[M+H]+ Ms m/z(ESI):617.2[M+H] +
1H NMR(400MHz,DMSO-d6)δ10.39(s,1H),7.85–7.73(m,4H),7.15–7.07(m,1H),5.38 –5.16(m,1H),4.26(d,2H),4.04(dd,2H),3.54–3.40(m,4H),3.16–2.98(m,3H),2.87–2.78(m, 1H),2.57–2.52(m,1H),2.19–2.10(m,1H),2.09(s,3H),2.06–1.97(m,2H),1.90–1.73(m,3H), 1.71–1.58(m,4H). 1 H NMR (400MHz,DMSO-d 6 )δ10.39(s,1H),7.85–7.73(m,4H),7.15–7.07(m,1H),5.38–5.16(m,1H),4.26(d ,2H),4.04(dd,2H),3.54–3.40(m,4H),3.16–2.98(m,3H),2.87–2.78(m, 1H),2.57–2.52(m,1H),2.19–2.10 (m,1H),2.09(s,3H),2.06–1.97(m,2H),1.90–1.73(m,3H), 1.71–1.58(m,4H).
实施例32:N-(3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H- 吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-4-(三氟甲基)苯基)甲磺酰胺(化合物32)Example 32: N-(3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3-yl)-8-fluoro-2-(((2R, 7aS)-2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)phenyl)methanesulfonamide (compound 32)
N-(3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1 H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)phenyl)methanesulfonamideN-(3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro- 1 H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)phenyl)methanesulfonamide
第一步:N-[3-溴-4-(三氟甲基)苯基]甲烷磺酰胺(32A)The first step: N-[3-bromo-4-(trifluoromethyl)phenyl]methanesulfonamide (32A)
N-[3-bromo-4-(trifluoromethyl)phenyl]methanesulfonamideN-[3-bromo-4-(trifluoromethyl)phenyl]methanesulfonamide
将原料3-溴-4-三氟甲基苯胺(1.0g,4.2mmol)溶于5mL二氯甲烷中,加入三乙胺(0.848 g,8.4mmol)和甲磺酰氯(0.718g,6.3mmol),室温搅拌2h,反应完成后,减压浓缩,柱层析得到化合物32A(1.0g,产率74.87%)。The starting material 3-bromo-4-trifluoromethylaniline (1.0 g, 4.2 mmol) was dissolved in 5 mL of dichloromethane, triethylamine (0.848 g, 8.4 mmol) and methanesulfonyl chloride (0.718 g, 6.3 mmol) were added , stirred at room temperature for 2 h, after the completion of the reaction, concentrated under reduced pressure and column chromatography to obtain compound 32A (1.0 g, yield 74.87%).
LCMS m/z=318.0[M+H]+ LCMS m/z=318.0[M+H] +
第二步:叔丁基(1R,5S)-3-(8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)-7-(5-(甲基磺酰氨基)-2-(三氟甲基)苯基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸酯 (32B)The second step: tert-butyl (1R,5S)-3-(8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy Base)-7-(5-(methylsulfonylamino)-2-(trifluoromethyl)phenyl)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane Alkane-8-carboxylate (32B)
tert-butyl(1R,5S)-3-(8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methox y)-7-(5-(methylsulfonamido)-2-(trifluoromethyl)phenyl)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octa ne-8-carboxylatetert-butyl(1R,5S)-3-(8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methox y)-7-(5- (methylsulfonamido)-2-(trifluoromethyl)phenyl)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octa ne-8-carboxylate
将(32A)(0.16g,0.52mmol)溶解于1,4-二氧六环5mL和水1mL的混合溶液中,加入9B(0.23g,0.42mmol),碳酸铯(0.3g,1.04mmol),Pd(dppf)Cl2(0.038g,0.052mmol),氮气保护下100℃下反应2h,将反应冷却至室温,加入乙酸乙酯40mL和水25mL,水相再用乙酸乙酯(40mL×1)萃取一次,合并乙酸乙酯层,乙酸乙酯层用饱和食盐水洗涤一次(40mL×1),无水硫酸钠干燥,减压浓缩后,残留物用硅胶色谱柱柱层析得到化合物(32B)(0.11g,产率34.82%)Dissolve (32A) (0.16g, 0.52mmol) in a mixed solution of 1,4-dioxane 5mL and water 1mL, add 9B (0.23g, 0.42mmol), cesium carbonate (0.3g, 1.04mmol), Pd(dppf)Cl 2 (0.038g, 0.052mmol), reacted at 100°C under nitrogen protection for 2h, cooled the reaction to room temperature, added 40mL of ethyl acetate and 25mL of water, and then used ethyl acetate (40mL×1) for the aqueous phase Extract once, combine the ethyl acetate layers, wash the ethyl acetate layer once with saturated brine (40mL×1), dry over anhydrous sodium sulfate, concentrate under reduced pressure, and the residue is chromatographed on a silica gel column to obtain compound (32B) (0.11g, yield 34.82%)
LCMS m/z=753.3[M+H]+ LCMS m/z=753.3[M+H] +
第三步:N-(3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-4-(三氟甲基)苯基)甲磺酰胺(化合物32)The third step: N-(3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3-yl)-8-fluoro-2-(((2R, 7aS)-2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)phenyl)methanesulfonamide (compound 32)
N-(3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1 H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)phenyl)methanesulfonamideN-(3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro- 1 H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-(trifluoromethyl)phenyl)methanesulfonamide
将化合物(32B)(0.11g,0.15mmol)溶解于二氯甲烷5mL中,加入三氟乙酸0.5mL,室温下反应40分钟。减压浓缩反应液,然后加入二氯甲烷溶解,滴入0.5mL N,N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物32(20mg,产率20.45%),为白色粉末。Compound (32B) (0.11 g, 0.15 mmol) was dissolved in 5 mL of dichloromethane, 0.5 mL of trifluoroacetic acid was added, and the reaction was carried out at room temperature for 40 minutes. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 0.5mL N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, the residue is prepared and purified by HPLC, the preparation solution is concentrated and freeze-dried to obtain compound 32( 20 mg, yield 20.45%), as a white powder.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH 4 HCO 3 ); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min .
Ms m/z(ESI):653.2[M+H]+ Ms m/z(ESI):653.2[M+H] +
实施例33:7-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H- 吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-1-异丙基-1H-吲唑-5-胺(化合物33)Example 33: 7-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3-yl)-8-fluoro-2-(((2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-1-isopropyl-1H-indazol-5-amine (compound 33)
7-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-1-isopropyl-1H-indazol-5-amine7-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-1-isopropyl-1H-indazol-5-amine
第一步:7-溴-1-异丙基-5-硝基吲唑(33A)The first step: 7-bromo-1-isopropyl-5-nitroindazole (33A)
7-bromo-1-isopropyl-5-nitro-indazole7-bromo-1-isopropyl-5-nitro-indazole
将原料7-溴-5-硝基-1H-吲唑(2.0g,8.3mmol)溶于20mL四氢呋喃中,加入1,8-二氮杂二环[5.4.0]十一碳-7-烯(2.52g,16.6mmol)和2-碘丙烷(1.7g,10mmol),升温至60℃搅拌 2h,反应完成后,减压浓缩,柱层析得到化合物33A(0.9g,产率38.10%)。The starting material 7-bromo-5-nitro-1H-indazole (2.0 g, 8.3 mmol) was dissolved in 20 mL of tetrahydrofuran, and 1,8-diazabicyclo[5.4.0]undec-7-ene was added (2.52g, 16.6mmol) and 2-iodopropane (1.7g, 10mmol), heated to 60°C and stirred for 2h, after the reaction was completed, concentrated under reduced pressure, and column chromatography gave compound 33A (0.9g, yield 38.10%).
LCMS m/z=284.1[M+H]+ LCMS m/z=284.1[M+H] +
第二步:7-溴-1-异丙基-吲唑-5-胺(33B)The second step: 7-bromo-1-isopropyl-indazol-5-amine (33B)
7-bromo-1-isopropyl-indazol-5-amine7-bromo-1-isopropyl-indazol-5-amine
将33A(0.90g,3.17mmol)溶于9mL乙醇和3mL水中,加入还原铁粉(0.90g,16.0mmol) 氯化铵(1.74g,31.7mmol),升温至80℃搅拌2h,反应完成后,减压浓缩,柱层析得到化合物 33B(0.56g,产率69.82%)。Dissolve 33A (0.90g, 3.17mmol) in 9mL ethanol and 3mL water, add reduced iron powder (0.90g, 16.0mmol) ammonium chloride (1.74g, 31.7mmol), heat up to 80°C and stir for 2h, after the reaction is complete, Concentration under reduced pressure and column chromatography gave compound 33B (0.56 g, yield 69.82%).
LCMS m/z=254.1[M+H]+ LCMS m/z=254.1[M+H] +
第三步:叔丁基(1R,5S)-3-(7-(5-氨基-1-异丙基-1H-吲唑-7-基)-8-氟-2-(((2R,7aS)-2-氟四氢 -1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸酯(33C)The third step: tert-butyl (1R,5S)-3-(7-(5-amino-1-isopropyl-1H-indazol-7-yl)-8-fluoro-2-(((2R, 7aS)-2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane- 8-Carboxylate (33C)
tert-butyl(1R,5S)-3-(7-(5-amino-1-isopropyl-1H-indazol-7-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotet rahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxy latetert-butyl(1R,5S)-3-(7-(5-amino-1-isopropyl-1H-indazol-7-yl)-8-fluoro-2-(((2R,7aS)-2-fluororotet rahydro -1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxy late
将(33B)(0.13g,0.52mmol)溶解于1,4-二氧六环5mL和水1mL的混合溶液中,加入9B(0.23g,0.42mmol),碳酸铯(0.3g,1.04mmol),Pd(dppf)Cl2(0.038g,0.052mmol),氮气保护下100℃下反应2h,将反应冷却至室温,加入乙酸乙酯40mL和水25mL,水相再用乙酸乙酯(40mL×1)萃取一次,合并乙酸乙酯层,乙酸乙酯层用饱和食盐水洗涤一次(40mL×1),无水硫酸钠干燥,减压浓缩后,残留物用硅胶色谱柱柱层析得到化合物(33C)(0.11g,产率38.06%)Dissolve (33B) (0.13g, 0.52mmol) in a mixed solution of 1,4-dioxane 5mL and water 1mL, add 9B (0.23g, 0.42mmol), cesium carbonate (0.3g, 1.04mmol), Pd(dppf)Cl 2 (0.038g, 0.052mmol), reacted at 100°C under nitrogen protection for 2h, cooled the reaction to room temperature, added 40mL of ethyl acetate and 25mL of water, and then used ethyl acetate (40mL×1) for the aqueous phase Extract once, combine the ethyl acetate layers, wash the ethyl acetate layer once with saturated brine (40mL×1), dry over anhydrous sodium sulfate, concentrate under reduced pressure, and the residue is chromatographed on a silica gel column to obtain compound (33C) (0.11g, yield 38.06%)
LCMS m/z=689.4[M+H]+ LCMS m/z=689.4[M+H] +
第四步:7-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-1-异丙基-1H-吲唑-5-胺(化合物33)The fourth step: 7-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3-yl)-8-fluoro-2-(((2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-1-isopropyl-1H-indazol-5-amine (compound 33)
7-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-1-isopropyl-1H-indazol-5-amine7-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-1-isopropyl-1H-indazol-5-amine
将化合物(33C)(0.11g,0.16mmol)溶解于二氯甲烷5mL中,加入三氟乙酸0.5mL,室温下反应40分钟。减压浓缩反应液,然后加入二氯甲烷溶解,滴入0.5mL N,N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物33(20mg,产率21.25%),为白色粉末。Compound (33C) (0.11 g, 0.16 mmol) was dissolved in 5 mL of dichloromethane, 0.5 mL of trifluoroacetic acid was added, and the reaction was carried out at room temperature for 40 minutes. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 0.5mL N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, the residue is prepared and purified by HPLC, the preparation solution is concentrated and freeze-dried to obtain compound 33( 20 mg, yield 21.25%), as a white powder.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH 4 HCO 3 ); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min .
Ms m/z(ESI):589.3[M+H]+ Ms m/z(ESI):589.3[M+H] +
实施例34:7-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-1-乙基-1H-吲唑-5-胺(化合物34)Example 34: 7-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3-yl)-8-fluoro-2-(((2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-1-ethyl-1H-indazol-5-amine (compound 34)
7-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-1-ethyl-1H-indazol-5-amine7-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-1-ethyl-1H-indazol-5-amine
第一步:7-溴-1-乙基-5-硝基吲唑(34A)The first step: 7-bromo-1-ethyl-5-nitroindazole (34A)
7-bromo-1-ethyl-5-nitro-indazole7-bromo-1-ethyl-5-nitro-indazole
将原料7-溴-5-硝基-1H-吲唑(2.0g,8.3mmol)溶于20mL四氢呋喃中,加入1,8-二氮杂二环[5.4.0]十一碳-7-烯(2.52g,16.6mmol),碘乙烷(1.56g,10mmol),升温至60℃搅拌2h,反应完成后,减压浓缩,柱层析得到化合物34A(0.9g,产率40.30%)。The starting material 7-bromo-5-nitro-1H-indazole (2.0 g, 8.3 mmol) was dissolved in 20 mL of tetrahydrofuran, and 1,8-diazabicyclo[5.4.0]undec-7-ene was added (2.52g, 16.6mmol), ethyl iodide (1.56g, 10mmol), heated to 60°C and stirred for 2h, after the reaction was completed, concentrated under reduced pressure, and column chromatography gave compound 34A (0.9g, yield 40.30%).
LCMS m/z=270.1[M+H]+ LCMS m/z=270.1[M+H] +
第二步:7-溴-1-乙基吲唑-5-胺(34B)The second step: 7-bromo-1-ethylindazol-5-amine (34B)
7-bromo-1-ethyl-indazol-5-amine7-bromo-1-ethyl-indazol-5-amine
将34A(0.90g,3.34mmol)溶于9mL乙醇和3mL水中,加入还原铁粉(0.90g,16.0mmol) 氯化铵(1.74g,31.7mmol),升温至80℃搅拌2h,反应完成后,减压浓缩,柱层析得到化合物 34B(0.56g,产率69.86%)。Dissolve 34A (0.90g, 3.34mmol) in 9mL ethanol and 3mL water, add reduced iron powder (0.90g, 16.0mmol) ammonium chloride (1.74g, 31.7mmol), heat up to 80°C and stir for 2h, after the reaction is complete, Concentration under reduced pressure and column chromatography gave compound 34B (0.56 g, yield 69.86%).
LCMS m/z=240.1[M+H]+ LCMS m/z=240.1[M+H] +
第三步:叔丁基(1R,5S)-3-(7-(5-氨基-1-乙基-1H-吲唑-7-基)-8-氟-2-(((2R,7aS)-2-氟四氢 -1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸酯(34C)The third step: tert-butyl (1R,5S)-3-(7-(5-amino-1-ethyl-1H-indazol-7-yl)-8-fluoro-2-(((2R,7aS )-2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8 - Carboxylate (34C)
tert-butyl(1R,5S)-3-(7-(5-amino-1-ethyl-1H-indazol-7-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahy dro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylatetert-butyl(1R,5S)-3-(7-(5-amino-1-ethyl-1H-indazol-7-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahy dro -1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
将(34B)(0.12g,0.52mmol)溶解于1,4-二氧六环5mL和水1mL的混合溶液中,加入9B(0.23g,0.42mmol),碳酸铯(0.3g,1.04mmol),Pd(dppf)Cl2(0.038g,0.052mmol),氮气保护下100℃下反应2h,将反应冷却至室温,加入乙酸乙酯40mL和水25mL,水相再用乙酸乙酯(40mL×1)萃取一次,合并乙酸乙酯层,乙酸乙酯层用饱和食盐水洗涤一次(40mL×1),无水硫酸钠干燥,减压浓缩后,残留物用硅胶色谱柱柱层析得到化合物(34C)(0.11g,产率38.85%)Dissolve (34B) (0.12g, 0.52mmol) in a mixed solution of 1,4-dioxane 5mL and water 1mL, add 9B (0.23g, 0.42mmol), cesium carbonate (0.3g, 1.04mmol), Pd(dppf)Cl 2 (0.038g, 0.052mmol), reacted at 100°C under nitrogen protection for 2h, cooled the reaction to room temperature, added 40mL of ethyl acetate and 25mL of water, and then used ethyl acetate (40mL×1) for the aqueous phase Extract once, combine the ethyl acetate layers, wash the ethyl acetate layer once with saturated brine (40mL×1), dry over anhydrous sodium sulfate, concentrate under reduced pressure, the residue is chromatographed on a silica gel column to obtain compound (34C) (0.11g, yield 38.85%)
LCMS m/z=675.3[M+H]+ LCMS m/z=675.3[M+H] +
第四步:7-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-1-乙基-1H-吲唑-5-胺(化合物34)The fourth step: 7-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3-yl)-8-fluoro-2-(((2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-1-ethyl-1H-indazol-5-amine (compound 34)
7-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-1-ethyl-1H-indazol-5-amine7-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-1-ethyl-1H-indazol-5-amine
将化合物(34C)(0.11g,0.16mmol)溶解于二氯甲烷5mL中,加入三氟乙酸0.5mL,室温下反应40分钟。减压浓缩反应液,然后加入二氯甲烷溶解,滴入0.5mL N,N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物34(20mg,产率21.77%),为白色粉末。Compound (34C) (0.11 g, 0.16 mmol) was dissolved in 5 mL of dichloromethane, 0.5 mL of trifluoroacetic acid was added, and the reaction was carried out at room temperature for 40 minutes. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 0.5mL N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, the residue is prepared and purified by HPLC, the preparation solution is concentrated and freeze-dried to obtain compound 34( 20 mg, yield 21.77%), as a white powder.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH 4 HCO 3 ); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min .
Ms m/z(ESI):575.3[M+H]+ Ms m/z(ESI):575.3[M+H] +
1H NMR(400MHz,DMSO-d6)δ7.88–7.81(m,2H),7.26(dd,1H),6.87(d,1H),6.70(d,1H),5.38–5.18(m,1H),4.95(s,2H),4.36–4.22(m,2H),4.16–3.97(m,2H),3.91–3.70(m,2H), 3.57–3.41(m,4H),3.19–2.96(m,4H),2.88–2.77(m,1H),2.21–1.97(m,3H),1.89–1.72(m, 3H),1.72–1.59(m,4H),0.91(t,3H). 1 H NMR (400MHz,DMSO-d 6 )δ7.88–7.81(m,2H),7.26(dd,1H),6.87(d,1H),6.70(d,1H),5.38–5.18(m,1H ),4.95(s,2H),4.36–4.22(m,2H),4.16–3.97(m,2H),3.91–3.70(m,2H), 3.57–3.41(m,4H),3.19–2.96(m ,4H),2.88–2.77(m,1H),2.21–1.97(m,3H),1.89–1.72(m, 3H),1.72–1.59(m,4H),0.91(t,3H).
实施例35:5-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H- 吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-2,4-双(三氟甲基)苯胺(化合物35)Example 35: 5-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3-yl)-8-fluoro-2-(((2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-2,4-bis(trifluoromethyl)aniline (compound 35)
5-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-2,4-bis(trifluoromethyl)aniline5-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-2,4-bis(trifluoromethyl)aniline
第一步:(5-溴-2-碘-4-(三氟甲基)苯基)氨基甲酸叔丁酯(35A)The first step: tert-butyl (5-bromo-2-iodo-4-(trifluoromethyl)phenyl)carbamate (35A)
tert-butyl(5-bromo-2-iodo-4-(trifluoromethyl)phenyl)carbamatetert-butyl(5-bromo-2-iodo-4-(trifluoromethyl)phenyl)carbamate
将化合物25B(1g,2.73mmol)溶于12mL四氢呋喃,冰浴下,加入双三甲基硅基胺基锂(6.0mL,1M),搅拌反应0.5小时,加入二碳酸二叔丁酯(656mg,3.00mmol),继续反应3 小时。然后加入饱和氯化铵溶液,乙酸乙酯萃取三次。合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物(35A),为白色固体(572mg,产率45%)Compound 25B (1g, 2.73mmol) was dissolved in 12mL tetrahydrofuran, under ice-cooling, lithium bistrimethylsilylamide (6.0mL, 1M) was added, the reaction was stirred for 0.5 hours, and di-tert-butyl dicarbonate (656mg, 3.00mmol), continue to react for 3 hours. Then a saturated ammonium chloride solution was added and extracted three times with ethyl acetate. The organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound (35A) as a white solid (572mg, yield 45%)
LCMS m/z=463.9[M-H]- LCMS m/z=463.9[MH] -
第二步:5-溴-2,4-双(三氟甲基)苯胺(35B)The second step: 5-bromo-2,4-bis(trifluoromethyl)aniline (35B)
5-bromo-2,4-bis(trifluoromethyl)aniline5-bromo-2,4-bis(trifluoromethyl)aniline
将化合物35A(0.572g,1.22mmol),氟磺酰基二氟乙酸甲酯(1.65g,8.59mmol),溶于 N,N-二甲基甲酰胺,然后加入碘化亚铜(465mg,2.44mmol)和N,N-二异丙基乙胺(1.11g,8.59 mmol),置于封管中,置换氮气保护。升温至75℃反应24小时。将反应冷却至室温,加水稀释,乙酸乙酯萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物35B,为褐色固体(132mg,产率35%)Compound 35A (0.572g, 1.22mmol), methyl fluorosulfonyl difluoroacetate (1.65g, 8.59mmol), dissolved in N,N-dimethylformamide, then added cuprous iodide (465mg, 2.44mmol ) and N,N-diisopropylethylamine (1.11g, 8.59 mmol), placed in a sealed tube, replaced by nitrogen protection. Raise the temperature to 75°C for 24 hours. The reaction was cooled to room temperature, diluted with water, extracted three times with ethyl acetate, the organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound 35B as a brown solid (132 mg, yield 35%)
LCMS m/z=306.0[M-H]- LCMS m/z=306.0[MH] -
第三步:叔丁基(1R,5S)-3-(7-(5-氨基-2,4-双(三氟甲基)苯基)-8-氟-2-(((2R,7aS)-2-氟四氢 -1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸酯(35C)The third step: tert-butyl (1R,5S)-3-(7-(5-amino-2,4-bis(trifluoromethyl)phenyl)-8-fluoro-2-(((2R,7aS )-2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8 -Carboxylate (35C)
tert-butyltert-butyl
(1R,5S)-3-(7-(5-amino-2,4-bis(trifluoromethyl)phenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H -pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate(1R,5S)-3-(7-(5-amino-2,4-bis(trifluoromethyl)phenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a (5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
将中间体9B(159mg,0.29mmol),化合物35B(60mg,0.19mmol),双三苯基磷二氯化钯(14mg,0.02mmol),和氟化钾(41mg,0.71mmol)置于封管中,加入2mL1,4-二氧六环和0.5mL水,置换氮气保护。升温至100℃反应18小时。将反应冷却至室温,加水稀释,乙酸乙酯萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物35C,为棕色固体(70mg,产率50%)Intermediate 9B (159 mg, 0.29 mmol), compound 35B (60 mg, 0.19 mmol), bistriphenylphosphine palladium dichloride (14 mg, 0.02 mmol), and potassium fluoride (41 mg, 0.71 mmol) were placed in a locked tube In, add 2mL of 1,4-dioxane and 0.5mL of water to replace the nitrogen protection. The temperature was raised to 100° C. for 18 hours. The reaction was cooled to room temperature, diluted with water, extracted three times with ethyl acetate, the organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound 35C as a brown solid (70mg, yield 50%)
LCMS m/z=743.3[M+H]+ LCMS m/z=743.3[M+H] +
第四步:5-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-2,4-双(三氟甲基)苯胺(化合物35)The fourth step: 5-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]oct-3-yl)-8-fluoro-2-(((2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-2,4-bis(trifluoromethyl)aniline (compound 35)
5-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-2,4-bis(trifluoromethyl)aniline5-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-2,4-bis(trifluoromethyl)aniline
将化合物35C(70mg,0.094mmol)溶解于二氯甲烷2.5mL中,然后滴入0.5mL三氟乙酸,室温下反应2小时。减压浓缩反应液,然后加入二氯甲烷溶解,滴入0.5mL N,N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物30(26mg,产率43%),为白色粉末。Compound 35C (70 mg, 0.094 mmol) was dissolved in 2.5 mL of dichloromethane, then 0.5 mL of trifluoroacetic acid was added dropwise, and reacted at room temperature for 2 hours. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 0.5mL N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, the residue is prepared and purified by HPLC, the preparation solution is concentrated and freeze-dried to obtain compound 30( 26 mg, yield 43%), as a white powder.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH4HCO3); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min.
LCMS m/z=643.2[M+H]+ LCMS m/z=643.2[M+H] +
1H NMR(400MHz,CD3OD)δ7.77(d,1H),7.73(s,1H),7.12(dd,1H),6.80(s,1H),5.38–5.22 (m,1H),4.55–4.40(m,2H),4.24(dd,2H),3.65–3.52(m,4H),3.27–3.11(m,3H),3.05–2.96 (m,1H),2.40–2.07(m,4H),2.04–1.94(m,2H),1.92–1.78(m,5H). 1 H NMR (400MHz, CD3OD) δ7.77(d,1H),7.73(s,1H),7.12(dd,1H),6.80(s,1H),5.38–5.22 (m,1H),4.55–4.40 (m,2H),4.24(dd,2H),3.65–3.52(m,4H),3.27–3.11(m,3H),3.05–2.96 (m,1H),2.40–2.07(m,4H),2.04 –1.94(m,2H),1.92–1.78(m,5H).
实施例36:3-(4-(1R,5S)-3,8-二氮杂双环[3.2.1]-辛烷-3-基)-8-氟-2-(2R,7aS)-2-氟四氢-1H-吡咯嗪 -7a(5H)-基)甲基)喹唑啉-7-基)-5-氟-4-(三氟甲基)苯胺(化合物36)Example 36: 3-(4-(1R,5S)-3,8-diazabicyclo[3.2.1]-octane-3-yl)-8-fluoro-2-(2R,7aS)-2 -Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methyl)quinazolin-7-yl)-5-fluoro-4-(trifluoromethyl)aniline (compound 36)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-fluoro-4-(trifluoromethyl)aniline3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-5-fluoro-4-(trifluoromethyl)aniline
第一步:(3-溴-5-氟-4-碘苯基)氨基甲酸叔丁酯(36B)The first step: (3-bromo-5-fluoro-4-iodophenyl) tert-butyl carbamate (36B)
tert-butyl(3-bromo-5-fluoro-4-iodophenyl)carbamatetert-butyl(3-bromo-5-fluoro-4-iodophenyl)carbamate
在N2的保护下,先将化合物36A(5.0g,15.83mmol)、二碳酸二叔丁酯(4.15g,19.0mmol) 溶解在20mL的THF中,然后在冰浴的条件下,缓慢滴加LiHMDS(6.55mL),滴加完毕会后,将反应液移至常温条件下反应8h,待反应完全结束后,EA/H2O(150mL/100mL)进行萃取洗涤,浓缩有机层,经层析柱纯化得到化合物36B(4.50g,产率:68.33%)。Under the protection of N2 , compound 36A (5.0g, 15.83mmol), di-tert-butyl dicarbonate (4.15g, 19.0mmol) were first dissolved in 20mL of THF, and then slowly dropwise added LiHMDS (6.55mL), after the dropwise addition, the reaction solution was moved to normal temperature for 8h, after the reaction was complete, EA/H 2 O (150mL/100mL) was extracted and washed, the organic layer was concentrated, and chromatographed Column purification gave compound 36B (4.50 g, yield: 68.33%).
Ms m/z(ESI):413.9[M-H]+ Ms m/z(ESI):413.9[MH] +
第二步:(3-溴-5-氟-4-(三氟甲基)苯基)氨基甲酸叔丁酯(36C)The second step: tert-butyl (3-bromo-5-fluoro-4-(trifluoromethyl)phenyl)carbamate (36C)
tert-butyl(3-bromo-5-fluoro-4-(trifluoromethyl)phenyl)carbamatetert-butyl(3-bromo-5-fluoro-4-(trifluoromethyl)phenyl)carbamate
在N2的保护下,依次将化合物36B(2.0g,4.81mmol)、MFSDA(6.47g,33.67mmol) 和CuI(1.37g,7.21mmol)加入到反应试管中,然后再将DIPEA(4.35g,33.67mmol)溶解在20mL的DMF中并缓慢滴加到反应试管中。在75℃的油浴锅中持续反应16h,待反应结束后, EA/H2O(200mL/150mL*2)进行萃取洗涤。最后将有机层浓缩经层析柱纯化(PE:EA=10:1),获得化合物36C(1.20g,产率:69.66%yield)。Under the protection of N2 , compound 36B (2.0g, 4.81mmol), MFSDA (6.47g, 33.67mmol) and CuI (1.37g, 7.21mmol) were added to the reaction test tube in turn, and then DIPEA (4.35g, 33.67mmol) was dissolved in 20mL of DMF and slowly added dropwise to the reaction test tube. The reaction was continued in an oil bath at 75° C. for 16 hours. After the reaction was completed, EA/H 2 O (200 mL/150 mL*2) was used for extraction and washing. Finally, the organic layer was concentrated and purified by column chromatography (PE:EA=10:1) to obtain compound 36C (1.20 g, yield: 69.66% yield).
Ms m/z(ESI):356.0[M-H]+ Ms m/z(ESI):356.0[MH] +
第三步:叔丁基(1R,5S)-3-(7-(5-((叔丁氧羰基)氨基)-3-氟-2-(三氟甲基)苯基)-8-氟-2-((2R,7aS)-2- 氟四氢-1H-吡咯嗪-7a(5H)-甲氧基)喹唑啉-4-基)-3,8-二氮杂环[3.2.1]辛烷-8-羧酸酯(36D)。The third step: tert-butyl(1R,5S)-3-(7-(5-((tert-butoxycarbonyl)amino)-3-fluoro-2-(trifluoromethyl)phenyl)-8-fluoro -2-((2R,7aS)-2-fluorotetrahydro-1H-pyrrolazine-7a(5H)-methoxy)quinazolin-4-yl)-3,8-diazacyclo[3.2. 1] Octane-8-carboxylate (36D).
tert-butyl(1R,5S)-3-(7-(5-((tert-butoxycarbonyl)amino)-3-fluoro-2-(trifluoromethyl)phenyl)-8-fluoro -2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicycl o[3.2.1]octane-8-carboxylatetert-butyl(1R,5S)-3-(7-(5-((tert-butoxycarbonyl)amino)-3-fluoro-2-(trifluoromethyl)phenyl)-8-fluoro-2-(((2R,7aS )-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
将中间体9B(200mg,0.36mmol)和化合物36C(160.0mg,0.43mmol)置于封管中,加入2mL 1,4-二氧六环溶液溶解,然后依次加入[1,1'-双(二苯基膦基)二茂铁]二氯化钯(26.0 mg,0.036mmol)、碳酸铯(230.0mg,0.72mmol)。在氮气保护下,升至110℃反应5h,减压除去溶剂,加入50mL水和100mL乙酸乙酯溶解,水相用50mL×2乙酸乙酯洗,合并有机相,有机相用40mL饱和食盐水洗一次,然后有机相用无水硫酸钠干燥,旋干,柱层析(DCM: MeOH=15:1)得到化合物36D(200.0mg,产率:70.07%)。Intermediate 9B (200 mg, 0.36 mmol) and compound 36C (160.0 mg, 0.43 mmol) were placed in a sealed tube, 2 mL of 1,4-dioxane solution was added to dissolve, and then [1,1'-bis( Diphenylphosphino)ferrocene]palladium dichloride (26.0 mg, 0.036 mmol), cesium carbonate (230.0 mg, 0.72 mmol). Under the protection of nitrogen, rise to 110 ° C for 5 h, remove the solvent under reduced pressure, add 50 mL of water and 100 mL of ethyl acetate to dissolve, wash the water phase with 50 mL×2 ethyl acetate, combine the organic phase, and wash the organic phase once with 40 mL of saturated saline , and then the organic phase was dried with anhydrous sodium sulfate, spin-dried, and column chromatography (DCM:MeOH=15:1) gave compound 36D (200.0 mg, yield: 70.07%).
Ms m/z(ESI):793.3[M+H]+ Ms m/z(ESI):793.3[M+H] +
第四步:3-(4-(1R,5S)-3,8-二氮杂双环[3.2.1]-辛烷-3-基)-8-氟-2-(2R,7aS)-2-氟四氢-1H-吡咯嗪-7a (5H)-基)甲基)喹唑啉-7-基)-5-氟-4-(三氟甲基)苯胺(化合物36)The fourth step: 3-(4-(1R,5S)-3,8-diazabicyclo[3.2.1]-octane-3-yl)-8-fluoro-2-(2R,7aS)-2 -Fluorotetrahydro-1H-pyrrolazin-7a (5H)-yl)methyl)quinazolin-7-yl)-5-fluoro-4-(trifluoromethyl)aniline (compound 36)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-fluoro-4-(trifluoromethyl)aniline3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-fluoro-4-(trifluoromethyl)aniline
将化合物36D(200mg,0.25mmol)溶于10mL的DCM中,然后于室温下滴加1mL三氟乙酸,体系继续反应3h,减压浓缩反应液,残留物经制备HPLC纯化,制备液浓缩后冻干得到目标化合物36(100mg,产率:67.5%yield)。Compound 36D (200 mg, 0.25 mmol) was dissolved in 10 mL of DCM, then 1 mL of trifluoroacetic acid was added dropwise at room temperature, the system continued to react for 3 h, the reaction solution was concentrated under reduced pressure, the residue was purified by preparative HPLC, and the preparative solution was concentrated and frozen The target compound 36 was obtained by drying (100 mg, yield: 67.5% yield).
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH4HCO3); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min.
Ms m/z(ESI):593.2[M+H]+ Ms m/z(ESI):593.2[M+H] +
1H NMR(400MHz,CD3OD)δ7.86(d,1H),7.30(dd,1H),6.54(d,1H),6.33(s,1H),5.67– 5.47(m,1H),4.76–4.59(m,4H),4.24(s,2H),4.09–3.78(m,5H),3.55–3.44(m,1H),2.82– 2.52(m,3H),2.48–2.30(m,3H),2.23–2.10(m,4H). 1 H NMR (400MHz, CD 3 OD) δ7.86(d,1H),7.30(dd,1H),6.54(d,1H),6.33(s,1H),5.67– 5.47(m,1H),4.76 –4.59(m,4H),4.24(s,2H),4.09–3.78(m,5H),3.55–3.44(m,1H),2.82–2.52(m,3H),2.48–2.30(m,3H) ,2.23–2.10(m,4H).
实施例37:2-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]-辛烷-3-基)-8-氟-2-(2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-4-氨基-6-氯苄腈(化合物37)Example 37: 2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]-octane-3-yl)-8-fluoro-2-(2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-amino-6-chlorobenzonitrile (compound 37)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-fluoro-4-(trifluoromethyl)aniline3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-5-fluoro-4-(trifluoromethyl)aniline
第一步:4-氨基-2-溴-5-氯苯腈(37A)The first step: 4-amino-2-bromo-5-chlorobenzonitrile (37A)
4-amino-2-bromo-6-chlorobenzonitrile4-amino-2-bromo-6-chlorobenzonitrile
将中间体30B(500mg,1.50mmol)、溶解在4mL的N,N-二甲基甲酰胺中,氮气吹三分钟,然后加入氰化亚铜(130mg,1.50mmol),随后在微波100℃下,反应1小时。加水稀释,乙酸乙酯萃取三次。合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物(37A),为淡黄色固体(259mg,产率75%)Intermediate 30B (500mg, 1.50mmol) was dissolved in 4mL of N,N-dimethylformamide, blown with nitrogen for three minutes, then added cuprous cyanide (130mg, 1.50mmol), followed by microwave at 100°C , reacted for 1 hour. Dilute with water and extract three times with ethyl acetate. The organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound (37A) as a light yellow solid (259 mg, yield 75%)
LCMS m/z=230.9[M+H]+ LCMS m/z=230.9[M+H] +
第二步:叔丁基(1R,5S)-3-(7-(5-氨基-3-氯-2-氰基苯基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸酯(37B)The second step: tert-butyl (1R,5S)-3-(7-(5-amino-3-chloro-2-cyanophenyl)-8-fluoro-2-(((2R,7aS)-2 -Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylic acid Esters (37B)
tert-butyl(1R,5S)-3-(7-(5-amino-3-chloro-2-cyanophenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro- 1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylatetert-butyl(1R,5S)-3-(7-(5-amino-3-chloro-2-cyanophenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro- 1H-pyrrolizin- 7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
将中间体9B(150mg,0.27mmol),化合物37A(62mg,0.27mmol),[1,1'-双(二苯基膦) 二茂铁]二氯化钯二氯甲烷络合物(22mg,0.03mmol)和碳酸铯(180mg,0.54mmol)置于封管中,加入2.5mL1,4-二氧六环和0.5mL水,置换氮气保护。升温至110℃反应3小时。将反应冷却至室温,加水稀释,乙酸乙酯萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物37B,为褐色固体(83mg,产率46%)Intermediate 9B (150mg, 0.27mmol), compound 37A (62mg, 0.27mmol), [1,1'-bis(diphenylphosphino)ferrocene]dichloropalladium dichloromethane complex (22mg, 0.03mmol) and cesium carbonate (180mg, 0.54mmol) were placed in a sealed tube, and 2.5mL of 1,4-dioxane and 0.5mL of water were added to replace the nitrogen protection. The temperature was raised to 110° C. for 3 hours. The reaction was cooled to room temperature, diluted with water, extracted three times with ethyl acetate, the organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound 37B as a brown solid (83mg, yield 46%)
LCMS m/z=666.3[M+H]+ LCMS m/z=666.3[M+H] +
第三步:2-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]-辛烷-3-基)-8-氟-2-(2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-4-氨基-6-氯苄腈(化合物37)The third step: 2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]-octane-3-yl)-8-fluoro-2-(2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-amino-6-chlorobenzonitrile (compound 37)
2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-amino-6-chlorobenzonitrile2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-((((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-4-amino-6-chlorobenzonitrile
将化合物37B(80mg,0.12mmol)溶解于二氯甲烷3mL中,然后滴入0.5mL三氟乙酸,室温下反应2小时。减压浓缩反应液,然后加入二氯甲烷溶解,滴入0.5mL N,N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物37(34mg,产率50%),为白色粉末。Compound 37B (80 mg, 0.12 mmol) was dissolved in 3 mL of dichloromethane, then 0.5 mL of trifluoroacetic acid was added dropwise, and reacted at room temperature for 2 hours. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 0.5mL N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, the residue is prepared and purified by HPLC, the preparation solution is concentrated and freeze-dried to obtain compound 37 ( 34 mg, yield 50%), as a white powder.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH4HCO3); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min.
LCMS m/z=566.2[M+H]+ LCMS m/z=566.2[M+H] +
实施例38:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]-辛烷-3-基)-8-氟-2-(2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-5-氯-4-环丙基苯酚(化合物38)Example 38: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]-octane-3-yl)-8-fluoro-2-(2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-chloro-4-cyclopropylphenol (compound 38)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-chloro-4-cyclopropylphenol3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-chloro-4-cyclopropylphenol
第一步:叔丁基(1R,5S)-3-(7-(3-氯-2-环丙基-5-(甲氧基甲氧基)苯基)-8-氟-2-((((2R, 7aS)-2-氟四氢-1H)-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸酯(38B)The first step: tert-butyl (1R, 5S)-3-(7-(3-chloro-2-cyclopropyl-5-(methoxymethoxy)phenyl)-8-fluoro-2-( (((2R, 7aS)-2-fluorotetrahydro-1H)-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2 .1] Octane-8-carboxylate (38B)
tert-butyl(1R,5S)-3-(7-(3-chloro-2-cyclopropyl-5-(methoxymethoxy)phenyl)-8-fluoro-2-(((2R,7aS)-2 -fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane- 8-carboxylatetert-butyl(1R,5S)-3-(7-(3-chloro-2-cyclopropyl-5-(methoxymethoxy)phenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H -pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
将中间体9B(200mg,0.36mmol),化合物38A(100mg,0.34mmol),[1,1'-双(二苯基膦) 二茂铁]二氯化钯二氯甲烷络合物(28mg,0.03mmol)和碳酸铯(220mg,0.68mmol)置于封管中,加入3mL1,4-二氧六环和0.8mL水,置换氮气保护。升温至110℃反应3小时。将反应冷却至室温,加水稀释,乙酸乙酯萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物38B,为褐色固体(112mg,产率45%)Intermediate 9B (200mg, 0.36mmol), compound 38A (100mg, 0.34mmol), [1,1'-bis(diphenylphosphine)ferrocene]dichloropalladium dichloromethane complex (28mg, 0.03mmol) and cesium carbonate (220mg, 0.68mmol) were placed in a sealed tube, and 3mL of 1,4-dioxane and 0.8mL of water were added to replace the nitrogen protection. The temperature was raised to 110° C. for 3 hours. The reaction was cooled to room temperature, diluted with water, extracted three times with ethyl acetate, the organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound 38B as a brown solid (112 mg, yield 45%)
LCMS m/z=726.3[M+H]+ LCMS m/z=726.3[M+H] +
第二步:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]-辛烷-3-基)-8-氟-2-(2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-5-氯-4-环丙基苯酚(化合物38)The second step: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]-octane-3-yl)-8-fluoro-2-(2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-chloro-4-cyclopropylphenol (compound 38)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-chloro-4-cyclopropylphenol3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-chloro-4-cyclopropylphenol
将化合物38B(112mg,0.15mmol)溶解于二氯甲烷2mL中,然后滴入2.65mL4N盐酸-二氧六环溶液,室温下反应1小时。减压浓缩反应液,然后加入二氯甲烷溶解,滴入0.8mL N,N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物38(57mg,产率65%),为白色粉末。Compound 38B (112 mg, 0.15 mmol) was dissolved in 2 mL of dichloromethane, then dropped into 2.65 mL of 4N hydrochloric acid-dioxane solution, and reacted at room temperature for 1 hour. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 0.8mL N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, the residue is prepared and purified by HPLC, the preparation solution is concentrated and freeze-dried to obtain compound 38 ( 57 mg, yield 65%), as a white powder.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH4HCO3); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min.
LCMS m/z=582.2[M+H]+ LCMS m/z=582.2[M+H] +
实施例39:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]-辛烷-3-基)-8-氟-2-(2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-5-氨基-2-环丙基苄腈(化合物39)Example 39: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]-octane-3-yl)-8-fluoro-2-(2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-amino-2-cyclopropylbenzonitrile (compound 39)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-amino-2-cyclopropylbenzonitrile3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-amino-2-cyclopropylbenzonitrile
第一步:5-氨基-3溴-2碘苯腈(39B)The first step: 5-amino-3 bromo-2 iodobenzonitrile (39B)
5-amino-3-bromo-2-iodobenzonitrile5-amino-3-bromo-2-iodobenzonitrile
加入2-氨基-5-溴苯腈39A(2.50g,12.69mmol),用甲基叔丁基醚溶解,然后加入碘单质 (6.44g,25.38mmol),二甲亚砜(3.15mL,44.41mmol)N2保护,室温下搅拌反应3小时,加水稀释,乙酸乙酯萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物39B,为黄色固体(1.276g,产率31%)Add 2-amino-5-bromoxynil 39A (2.50g, 12.69mmol), dissolve with methyl tert-butyl ether, then add iodine (6.44g, 25.38mmol), dimethyl sulfoxide (3.15mL, 44.41mmol ) under N2 protection, stirred at room temperature for 3 hours, diluted with water, extracted three times with ethyl acetate, combined organic phases, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound 39B as yellow Solid (1.276g, 31% yield)
第二步:5-氨基-3-溴-2-环丙基苯腈(化合物39C)The second step: 5-amino-3-bromo-2-cyclopropylbenzonitrile (compound 39C)
5-amino-3-bromo-2-cyclopropylbenzonitrile5-amino-3-bromo-2-cyclopropylbenzonitrile
将化合物38B(580mg,1.80mmol),环丙基硼酸(200mg,2.34mmol),[1,1'-双(二苯基膦) 二茂铁]二氯化钯二氯甲烷络合物(150mg,0.23mmol)和碳酸铯(750mg,5.4mmol)置于封管中,加入9mL1,4-二氧六环和3mL水,置换氮气保护。升温至80℃反应8小时。将反应冷却至室温,加水稀释,乙酸乙酯萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物39C,为黄色固体(163mg,产率38%)Compound 38B (580mg, 1.80mmol), cyclopropylboronic acid (200mg, 2.34mmol), [1,1'-bis(diphenylphosphine)ferrocene]dichloropalladium dichloromethane complex (150mg , 0.23mmol) and cesium carbonate (750mg, 5.4mmol) were placed in a sealed tube, and 9mL of 1,4-dioxane and 3mL of water were added to replace the nitrogen protection. The temperature was raised to 80° C. for 8 hours. The reaction was cooled to room temperature, diluted with water, extracted three times with ethyl acetate, the organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound 39C as a yellow solid (163 mg, yield 38%)
LCMS m/z=237.1[M+H]+ LCMS m/z=237.1[M+H] +
第三步:叔丁基(1R,5S)-3-(7-(5-氨基-3-氰基-2-环丙基苯基)-8-氟-2-(((2R,7aS) -2-氟四氢-1H-吡咯烷-7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸酯 (39D)The third step: tert-butyl (1R, 5S)-3-(7-(5-amino-3-cyano-2-cyclopropylphenyl)-8-fluoro-2-(((2R, 7aS) -2-Fluorotetrahydro-1H-pyrrolidin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8- Carboxylate (39D)
tert-butyl(1R,5S)-3-(7-(5-amino-3-cyano-2-cyclopropylphenyl)-8-fluoro-2-(((2R,7aS)-2-fluorot etrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carbo xylatetert-butyl(1R,5S)-3-(7-(5-amino-3-cyano-2-cyclopropylphenyl)-8-fluoro-2-(((2R,7aS)-2-fluorot etrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carbo xylate
将中间体9B(170mg,0.30mmol),化合物39C(71mg,0.30mmol),[1,1'-双(二苯基膦) 二茂铁]二氯化钯二氯甲烷络合物(25mg,0.03mmol)和碳酸铯(200mg,0.60mmol)置于封管中,加入4mL1,4-二氧六环和1mL水,置换氮气保护。升温至110℃反应3小时。将反应冷却至室温,加水稀释,乙酸乙酯萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物39D,为棕黑色固体(123mg,产率61%)Intermediate 9B (170mg, 0.30mmol), compound 39C (71mg, 0.30mmol), [1,1'-bis(diphenylphosphino)ferrocene]dichloropalladium dichloromethane complex (25mg, 0.03mmol) and cesium carbonate (200mg, 0.60mmol) were placed in a sealed tube, and 4mL of 1,4-dioxane and 1mL of water were added to replace the nitrogen protection. The temperature was raised to 110° C. for 3 hours. The reaction was cooled to room temperature, diluted with water, extracted three times with ethyl acetate, the organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound 39D as a brown-black solid (123 mg, produced rate 61%)
LCMS m/z=672.3[M+H]+ LCMS m/z=672.3[M+H] +
第四步:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]-辛烷-3-基)-8-氟-2-(2R,7aS)-2-氟四氢-1H-吡咯嗪 -7a(5H)-基)甲氧基)喹唑啉-7-基)-5-氨基-2-环丙基苄腈(化合物39)The fourth step: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]-octane-3-yl)-8-fluoro-2-(2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-amino-2-cyclopropylbenzonitrile (compound 39)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-amino-2-cyclopropylbenzonitrile3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-amino-2-cyclopropylbenzonitrile
将化合物39D(120mg,0.18mmol)溶解于二氯甲烷3mL中,然后滴入0.5mL三氟乙酸,室温下反应2小时。减压浓缩反应液,然后加入二氯甲烷溶解,滴入0.5mL N,N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物39(58mg,产率56%),为白色粉末。Compound 39D (120 mg, 0.18 mmol) was dissolved in 3 mL of dichloromethane, then 0.5 mL of trifluoroacetic acid was added dropwise, and reacted at room temperature for 2 hours. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 0.5mL N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, prepare and purify the residue by HPLC, and freeze-dry the prepared solution to obtain compound 39( 58 mg, yield 56%), as a white powder.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH4HCO3); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min.
LCMS m/z=572.3[M+H]+ LCMS m/z=572.3[M+H] +
实施例40:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]-辛烷-3-基)-8-氟-2-(2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-5-氨基-2-(三氟甲基)苄腈(化合物40)Example 40: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]-octane-3-yl)-8-fluoro-2-(2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-amino-2-(trifluoromethyl)benzonitrile (compound 40)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-amino-2-(trifluoromethyl)benzonitrile3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-amino-2-(trifluoromethyl)benzonitrole
第一步:(3-溴-5-氰基-4-碘苯基)氨基甲酸叔丁酯(40A)The first step: (3-bromo-5-cyano-4-iodophenyl) tert-butyl carbamate (40A)
tert-butyl(3-bromo-5-cyano-4-iodophenyl)carbamatetert-butyl(3-bromo-5-cyano-4-iodophenyl)carbamate
在N2的保护下,先将化合物39B(1.0g,3.10mmol)、二碳酸二叔丁酯(740mg,3.41mmol) 溶解在20mL的THF中,然后在冰浴的条件下,缓慢滴加1N LiHMDS(6.79mL),滴加完毕后,将反应液移至常温条件下反应2小时,加入饱和氯化铵溶液稀释,乙酸乙酯萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物40A,为黄色油状(1.01g,产率77%)Under the protection of N 2 , first dissolve compound 39B (1.0g, 3.10mmol), di-tert-butyl dicarbonate (740mg, 3.41mmol) in 20mL of THF, and then slowly dropwise add 1N LiHMDS (6.79mL), after the dropwise addition, the reaction solution was moved to normal temperature and reacted for 2 hours, diluted with saturated ammonium chloride solution, extracted three times with ethyl acetate, combined organic phases, dried over anhydrous sodium sulfate, filtered, and Purified by column chromatography after concentration under reduced pressure to obtain compound 40A as a yellow oil (1.01 g, yield 77%)
LCMS m/z=422.9[M+H]+ LCMS m/z=422.9[M+H] +
第二步:(3-溴-5-氰基-4-(三氟甲基)苯基)氨基甲酸叔丁酯(40B)The second step: tert-butyl (3-bromo-5-cyano-4-(trifluoromethyl)phenyl)carbamate (40B)
tert-butyl(3-bromo-5-cyano-4-(trifluoromethyl)phenyl)carbamatetert-butyl(3-bromo-5-cyano-4-(trifluoromethyl)phenyl)carbamate
在N2的保护下,依次将化合物40A(980mg,2.32mmol)、氟磺酰基二氟乙酸甲酯(3.12g, 16.24mmol)和碘化亚铜(660mg,3.48mmol)加入到反应试管中,然后再将N,N-二异丙基乙胺 (2.69mL,16.24mmol)溶解在10mL的DMF中并缓慢滴加到反应试管中。在75℃的油浴锅中持续反应24小时,将反应冷却至室温,加入水稀释,然后用乙酸乙酯萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物40B,为黄色固体(754 mg,产率89%)Under the protection of N2 , compound 40A (980mg, 2.32mmol), methyl fluorosulfonyl difluoroacetate (3.12g, 16.24mmol) and cuprous iodide (660mg, 3.48mmol) were added to the reaction test tube in sequence, Then N,N-diisopropylethylamine (2.69 mL, 16.24 mmol) was dissolved in 10 mL of DMF and slowly added dropwise into the reaction test tube. Continue the reaction in an oil bath at 75°C for 24 hours, cool the reaction to room temperature, add water to dilute, then extract three times with ethyl acetate, combine the organic phases, dry over anhydrous sodium sulfate, filter, concentrate under reduced pressure and pass through the column layer Purified by analysis to obtain compound 40B as a yellow solid (754 mg, yield 89%)
LCMS m/z=363.0[M-H]- LCMS m/z=363.0[MH] -
第三步:叔丁基(1R,5S)-3-(7-(5-((叔丁氧羰基)氨基)-3-氰基-2-(三氟甲基)苯基)-8-氟 -2-(((2R,7aS))-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷 -8-羧酸酯(40C)The third step: tert-butyl (1R,5S)-3-(7-(5-((tert-butoxycarbonyl)amino)-3-cyano-2-(trifluoromethyl)phenyl)-8- Fluoro-2-(((2R,7aS))-2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazepine Heterobicyclo[3.2.1]octane-8-carboxylate (40C)
tert-butyl(1R,5S)-3-(7-(5-((tert-butoxycarbonyl)amino)-3-cyano-2-(trifluoromethyl)phenyl)-8-fluoro -2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicycl o[3.2.1]octane-8-carboxylatetert-butyl(1R,5S)-3-(7-(5-((tert-butoxycarbonyl)amino)-3-cyano-2-(trifluoromethyl)phenyl)-8-fluoro-2-(((2R,7aS )-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
将中间体9B(180mg,0.32mmol),化合物40B(120mg,0.32mmol),[1,1'-双(二苯基膦) 二茂铁]二氯化钯二氯甲烷络合物(26mg,0.03mmol)和碳酸铯(210mg,0.64mmol)置于封管中,加入4mL1,4-二氧六环和1mL水,置换氮气保护。升温至110℃反应3小时。将反应冷却至室温,加水稀释,乙酸乙酯萃取三次,合并有机相,无水硫酸钠干燥后过滤,减压浓缩后经过柱层析纯化,即得到化合物40C,为红褐色固体(66mg,产率26%)Intermediate 9B (180mg, 0.32mmol), compound 40B (120mg, 0.32mmol), [1,1'-bis(diphenylphosphine)ferrocene]dichloropalladium dichloromethane complex (26mg, 0.03mmol) and cesium carbonate (210mg, 0.64mmol) were placed in a sealed tube, and 4mL of 1,4-dioxane and 1mL of water were added to replace the nitrogen protection. The temperature was raised to 110° C. for 3 hours. The reaction was cooled to room temperature, diluted with water, extracted three times with ethyl acetate, the organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to obtain compound 40C as a reddish-brown solid (66 mg, yield rate 26%)
LCMS m/z=800.3[M+H]+ LCMS m/z=800.3[M+H] +
第四步:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]-辛烷-3-基)-8-氟-2-(2R,7aS)-2-氟四氢-1H-吡咯嗪 -7a(5H)-基)甲氧基)喹唑啉-7-基)-5-氨基-2-(三氟甲基)苄腈(化合物40)The fourth step: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]-octane-3-yl)-8-fluoro-2-(2R,7aS)- 2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-amino-2-(trifluoromethyl)benzonitrile (compound 40)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-amino-2-(trifluoromethyl)benzonitrile3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrro lizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-amino-2-(trifluoromethyl)benzonitrole
将化合物40C(66mg,0.08mmol)溶解于二氯甲烷3mL中,然后滴入0.5mL三氟乙酸,室温下反应2小时。减压浓缩反应液,然后加入二氯甲烷溶解,滴入0.5mL N,N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物40(30mg,产率60%),为白色粉末。Compound 40C (66 mg, 0.08 mmol) was dissolved in 3 mL of dichloromethane, then 0.5 mL of trifluoroacetic acid was added dropwise, and reacted at room temperature for 2 hours. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 0.5mL N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, the residue is prepared and purified by HPLC, the preparation solution is concentrated and freeze-dried to obtain compound 40( 30mg, yield 60%), as a white powder.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH4HCO3); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min.
LCMS m/z=600.2[M+H]+ LCMS m/z=600.2[M+H] +
实施例41:(2-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛基-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H- 吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-4-氨基苯基)二甲基氧化膦(化合物41)Example 41: (2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octyl-3-yl)-8-fluoro-2-(((2R,7aS )-2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-aminophenyl)dimethylphosphine oxide (compound 41)
(2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-aminophenyl)dimethylphosphine oxide.(2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-aminophenyl)dimethylphosphine oxide.
第一步:(4-氨基-2-溴苯基)二甲基氧化膦(41c)The first step: (4-amino-2-bromophenyl) dimethylphosphine oxide (41c)
(4-amino-2-bromophenyl)dimethylphosphine oxide.(4-amino-2-bromophenyl)dimethylphosphine oxide.
将(41a)(2.86g,9.6mmol)置于封管中,加入50mL1,4-二氧六环溶解,然后依次加入 (41b)(500mg,6.4mmol)、四三苯基膦钯(1.1g,0.96mmol)、碳酸钾(1.77g,12.8mmol)和5mL 水。氮气置换气三次,加热至80℃反应12h,待反应完成后,将反应冷却至室温,加入乙酸乙酯40mL和水25mL搅拌后分液,水相再用乙酸乙酯(40mL×1)萃取一次,合并乙酸乙酯层,乙酸乙酯层用饱和食盐水洗涤一次(40mL×1),无水硫酸钠干燥,减压浓缩后,残留物用硅胶色谱柱柱层析(PE:EA=5:1)得到化合物(41c)(1.07g,产率:67%)Put (41a) (2.86g, 9.6mmol) in a sealed tube, add 50mL of 1,4-dioxane to dissolve, then add (41b) (500mg, 6.4mmol), tetrakistriphenylphosphine palladium (1.1g , 0.96mmol), potassium carbonate (1.77g, 12.8mmol) and 5mL of water. Replace the gas with nitrogen three times, heat to 80°C and react for 12 hours. After the reaction is completed, cool the reaction to room temperature, add 40 mL of ethyl acetate and 25 mL of water, stir and separate the liquids, and extract the water phase with ethyl acetate (40 mL×1) once more , the ethyl acetate layers were combined, and the ethyl acetate layer was washed once with saturated brine (40mL×1), dried over anhydrous sodium sulfate, concentrated under reduced pressure, and the residue was chromatographed on a silica gel column (PE:EA=5: 1) Obtain compound (41c) (1.07g, yield: 67%)
LCMS m/z=248.0[M+H]+LCMS m/z=248.0[M+H]+
第二步:叔丁基(1R,5S)-3-(7-(5-氨基-2-(二甲基磷酰基)苯基)-8-氟-2-((((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a)(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸酯(41d)The second step: tert-butyl (1R, 5S)-3-(7-(5-amino-2-(dimethylphosphoryl) phenyl)-8-fluoro-2-((((2R, 7aS) -2-Fluorotetrahydro-1H-pyrrolazin-7a)(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8 -Carboxylate (41d)
tert-butyltert-butyl
(1R,5S)-3-(7-(5-amino-2-(dimethylphosphoryl)phenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H -pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate.(1R,5S)-3-(7-(5-amino-2-(dimethylphosphoryl)phenyl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H) -yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate.
将(41c)(0.16g,0.646mmol)溶解于乙腈15mL和水1.5mL的混合溶液中,加入9B(180mg, 0.323mmol),氟化钾(0.070g,1.21mmol),双三苯基膦二氯化钯(0.023g,0.032mmol),氮气保护下100℃下反应12h,将反应冷却至室温,加入乙酸乙酯40mL和水25mL搅拌后分液, 水相再用乙酸乙酯(40mL×1)萃取一次,合并乙酸乙酯层,乙酸乙酯层用饱和食盐水洗涤一次 (40mL×1),无水硫酸钠干燥,减压浓缩后,残留物用硅胶色谱柱柱层析(DCM:MeOH=96:4) 得到化合物(41d)(0.15g,产率:68%)Dissolve (41c) (0.16g, 0.646mmol) in a mixed solution of 15mL of acetonitrile and 1.5mL of water, add 9B (180mg, 0.323mmol), potassium fluoride (0.070g, 1.21mmol), bistriphenylphosphine di Palladium chloride (0.023g, 0.032mmol), reacted at 100°C for 12h under the protection of nitrogen, cooled the reaction to room temperature, added 40mL of ethyl acetate and 25mL of water, stirred and separated the liquids, and then used ethyl acetate (40mL×1 ) was extracted once, the ethyl acetate layers were combined, and the ethyl acetate layer was washed once with saturated brine (40mL×1), dried over anhydrous sodium sulfate, concentrated under reduced pressure, and the residue was subjected to silica gel column chromatography (DCM: MeOH =96:4) Compound (41d) was obtained (0.15g, yield: 68%)
LCMS m/z=683.3[M+H]+ LCMS m/z=683.3[M+H] +
第三步:(2-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛基-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H- 吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-4-氨基苯基)二甲基氧化膦(化合物41)The third step: (2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octyl-3-yl)-8-fluoro-2-(((2R,7aS )-2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-aminophenyl)dimethylphosphine oxide (compound 41)
(2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-aminophenyl)dimethylphosphine oxide.(2-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-4-aminophenyl)dimethylphosphine oxide.
将化合物(41d)(0.15g,0.22mmol)溶解于二氯甲烷5mL中,加入三氟乙酸0.5mL,室温下反应40分钟。减压浓缩反应液,然后加入二氯甲烷溶解,滴入0.5mL N,N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物41(50mg,产率:39%),为白色粉末。Compound (41d) (0.15 g, 0.22 mmol) was dissolved in 5 mL of dichloromethane, 0.5 mL of trifluoroacetic acid was added, and the reaction was carried out at room temperature for 40 minutes. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 0.5mL N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, the residue is prepared and purified by HPLC, the preparation solution is concentrated and freeze-dried to obtain compound 41( 50 mg, yield: 39%), as a white powder.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH 4 HCO 3 ); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min .
Ms m/z(ESI):583.2[M+H]+ Ms m/z(ESI):583.2[M+H] +
实施例42:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛基-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H- 吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-5-氯-4-(丙-1-烯-2-基)苯胺(化合物42)Example 42: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octyl-3-yl)-8-fluoro-2-(((2R,7aS) -2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-chloro-4-(prop-1-en-2-yl)aniline (Compound 42)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-chloro-4-(prop-1-en-2-yl)aniline.3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-5-chloro-4-(prop-1-en-2-yl)aniline.
第一步:3-溴-5-氯-4-(丙-1-烯-2-基)苯胺(42b)The first step: 3-bromo-5-chloro-4-(prop-1-en-2-yl)aniline (42b)
3-bromo-5-chloro-4-(prop-1-en-2-yl)aniline.3-bromo-5-chloro-4-(prop-1-en-2-yl)aniline.
将(14b)(0.628g,1.89mmol)溶解于乙腈30mL和水3mL的混合溶液中,加入42a(265mg, 1.58mmol),氟化钾(0.343g,5.9mmol),双三苯基膦二氯化钯(0.11g,0.158mmol),氮气保护下100℃下反应12h,将反应冷却至室温,加入乙酸乙酯40mL和水25mL搅拌后分液,水相再用乙酸乙酯(40mL×1)萃取一次,合并乙酸乙酯层,乙酸乙酯层用饱和食盐水洗涤一次(40 mL×1),无水硫酸钠干燥,减压浓缩后,残留物用硅胶色谱柱柱层析(PE:EA=5:1)得到化合物(42b)(0.29g,产率:75%)Dissolve (14b) (0.628g, 1.89mmol) in a mixed solution of 30mL of acetonitrile and 3mL of water, add 42a (265mg, 1.58mmol), potassium fluoride (0.343g, 5.9mmol), bistriphenylphosphine dichloro Palladium chloride (0.11g, 0.158mmol), reacted at 100°C for 12h under the protection of nitrogen, cooled the reaction to room temperature, added 40mL of ethyl acetate and 25mL of water, stirred and separated the liquids, and then used ethyl acetate (40mL×1) Extract once, combine the ethyl acetate layers, wash the ethyl acetate layer once with saturated brine (40 mL×1), dry over anhydrous sodium sulfate, and concentrate under reduced pressure, the residue is chromatographed on a silica gel column (PE: EA =5:1) to obtain compound (42b) (0.29g, yield: 75%)
LCMS m/z=246.0[M+H]+ LCMS m/z=246.0[M+H] +
第二步:叔丁基(1R,5S)-3-(7-(5-氨基-3-氯-2-(丙-1-烯-2-基)苯基)-8-氟-2-(((2R,7aS)-2- 氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸酯(42c)The second step: tert-butyl (1R,5S)-3-(7-(5-amino-3-chloro-2-(prop-1-en-2-yl)phenyl)-8-fluoro-2- (((2R,7aS)-2-fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2. 1] Octane-8-carboxylate (42c)
tert-butyl(1R,5S)-3-(7-(5-amino-3-chloro-2-(prop-1-en-2-yl)phenyl)-8-fluoro-2-(((2R,7aS)-2-fl uorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-c arboxylate.tert-butyl(1R,5S)-3-(7-(5-amino-3-chloro-2-(prop-1-en-2-yl)phenyl)-8-fluoro-2-(((2R, 7aS)-2-fl uorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy)quinazolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-c arboxylate.
将(42b)(0.09g,0.365mmol)溶解于乙腈15mL和水1.5mL的混合溶液中,加入9B(170mg, 0.304mmol),氟化钾(0.066g,1.137mmol),双三苯基膦二氯化钯(0.021g,0.03mmol),氮气保护下100℃下反应12h,将反应冷却至室温,加入乙酸乙酯40mL和水25mL搅拌后分液, 水相再用乙酸乙酯(40mL×1)萃取一次,合并乙酸乙酯层,乙酸乙酯层用饱和食盐水洗涤一次 (40mL×1),无水硫酸钠干燥,减压浓缩后,残留物用硅胶色谱柱柱层析(DCM:MeOH=96:4) 得到化合物(42c)(0.11g,产率:53%)Dissolve (42b) (0.09g, 0.365mmol) in a mixed solution of 15mL of acetonitrile and 1.5mL of water, add 9B (170mg, 0.304mmol), potassium fluoride (0.066g, 1.137mmol), bistriphenylphosphine di Palladium chloride (0.021g, 0.03mmol), reacted at 100°C for 12h under the protection of nitrogen, cooled the reaction to room temperature, added 40mL of ethyl acetate and 25mL of water, stirred and separated the liquids, and then used ethyl acetate (40mL×1 ) was extracted once, the ethyl acetate layers were combined, and the ethyl acetate layer was washed once with saturated brine (40mL×1), dried over anhydrous sodium sulfate, concentrated under reduced pressure, and the residue was subjected to silica gel column chromatography (DCM: MeOH =96:4) Compound (42c) was obtained (0.11 g, yield: 53%)
LCMS m/z=681.3[M+H]+ LCMS m/z=681.3[M+H] +
第三步:3-(4-((1R,5S)-3,8-二氮杂双环[3.2.1]辛基-3-基)-8-氟-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基)甲氧基)喹唑啉-7-基)-5-氯-4-(丙-1-烯-2-基)苯胺(化合物42)The third step: 3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octyl-3-yl)-8-fluoro-2-(((2R,7aS) -2-Fluorotetrahydro-1H-pyrrolazin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-chloro-4-(prop-1-en-2-yl)aniline (Compound 42)
3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H- pyrrolizin-7a(5H)-yl)methoxy)quinazolin-7-yl)-5-chloro-4-(prop-1-en-2-yl)aniline.3-(4-((1R,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl)-8-fluoro-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin -7a(5H)-yl)methoxy)quinazolin-7-yl)-5-chloro-4-(prop-1-en-2-yl)aniline.
将化合物(42c)(0.11g,0.26mmol)溶解于二氯甲烷5mL中,加入三氟乙酸0.5mL,室温下反应40分钟。减压浓缩反应液,然后加入二氯甲烷溶解,滴入0.5mL N,N-二异丙基乙胺后,减压浓缩,残留物经HPLC制备纯化,制备液浓缩后冻干得到化合物42(51mg,产率:54%),为白色粉末。Compound (42c) (0.11 g, 0.26 mmol) was dissolved in 5 mL of dichloromethane, 0.5 mL of trifluoroacetic acid was added, and the reaction was carried out at room temperature for 40 minutes. Concentrate the reaction solution under reduced pressure, then add dichloromethane to dissolve it, add 0.5mL N,N-diisopropylethylamine dropwise, concentrate under reduced pressure, the residue is prepared and purified by HPLC, the preparation solution is concentrated and freeze-dried to obtain compound 42( 51 mg, yield: 54%), as a white powder.
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH 4 HCO 3 ); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min .
Ms m/z(ESI):581.3[M+H]+ Ms m/z(ESI):581.3[M+H] +
实施例43:4-(4-(3,8-二氮杂双环[3.2.1]辛烷-3-基)-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪 -7a(5H)-基)甲氧基)-5,6,7,8-四氢喹唑啉-7-基)萘-2-醇(化合物43)Example 43: 4-(4-(3,8-Diazabicyclo[3.2.1]octan-3-yl)-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrole Oxyzin-7a(5H)-yl)methoxy)-5,6,7,8-tetrahydroquinazolin-7-yl)naphthalen-2-ol (compound 43)
4-(4-(3,8-diazabicyclo[3.2.1]octan-3-yl)-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-5,6,7,8-tetrahydroquinazolin-7-yl)naphthalen-2-ol.4-(4-(3,8-diazabicyclo[3.2.1]octan-3-yl)-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy) -5,6,7,8-tetrahydroquinazolin-7-yl)naphthalen-2-ol.
第一步:3-(3-(甲氧基甲氧基)萘-1-基)环己-1-酮(43b)The first step: 3-(3-(methoxymethoxy)naphthalene-1-yl)cyclohexan-1-one (43b)
3-(3-(methoxymethoxy)naphthalen-1-yl)cyclohexan-1-one.3-(3-(methoxymethoxy)naphthalen-1-yl)cyclohexan-1-one.
将(43a)(2.0g,6.37mmol)置于封管中,加入40mL1,4-二氧六环溶解,然后依次加环己烯酮(0.92mL,9.56mmol)、(1,5-环辛二烯)氯钌(I)二聚体(314mg,0.637mmol)、磷酸钾(4.0g,19.11mmol)和8mL水。氮气置换气三次,加热至50℃反应12h,将反应冷却至室温,加入乙酸乙酯40mL和水25mL搅拌后分液,水相再用乙酸乙酯(40mL×2)萃取一次,合并乙酸乙酯层,乙酸乙酯层用饱和食盐水洗涤一次(40mL×1),无水硫酸钠干燥,减压浓缩后,残留物用硅胶色谱柱柱层析分离纯化得到化合物(43b)(1.7g,产率:94%)Put (43a) (2.0g, 6.37mmol) in a sealed tube, add 40mL of 1,4-dioxane to dissolve, then add cyclohexenone (0.92mL, 9.56mmol), (1,5-cyclooct Diene) ruthenium(I) chloride dimer (314 mg, 0.637 mmol), potassium phosphate (4.0 g, 19.11 mmol) and 8 mL of water. Replace the gas with nitrogen three times, heat to 50°C for 12 hours, cool the reaction to room temperature, add 40 mL of ethyl acetate and 25 mL of water and stir, then separate the liquids, extract the water phase with ethyl acetate (40 mL×2) once more, and combine the ethyl acetate layer, the ethyl acetate layer was washed once with saturated brine (40mL×1), dried over anhydrous sodium sulfate, and concentrated under reduced pressure. The residue was separated and purified by silica gel column chromatography to obtain compound (43b) (1.7g, yield Rate: 94%)
LCMS m/z=285.1[M+H]+ LCMS m/z=285.1[M+H] +
第二步:4-(3-(甲氧基甲氧基)萘-1-基)-2-氧代环己烷-1-羧酸乙酯(43c)The second step: ethyl 4-(3-(methoxymethoxy)naphthalen-1-yl)-2-oxocyclohexane-1-carboxylate (43c)
ethyl 4-(3-(methoxymethoxy)naphthalen-1-yl)-2-oxocyclohexane-1-carboxylate.ethyl 4-(3-(methoxymethoxy)naphthalen-1-yl)-2-oxocyclohexane-1-carboxylate.
将化合物43b(1.2g,4.22mmol)悬浮于30mL干燥的四氢呋喃中,氮气保护降温至-78℃,缓慢滴加LiHMDS的四氢呋喃溶液(6.33mL,1mol/L,6.33mmol),体系保持-78℃搅拌30min。然后滴加氰基甲酸乙酯(627mg,6.33mmol),滴加完后体系自动升至室温反应2h。冷却至0℃,滴加50mL冰水淬灭反应。然后用乙酸乙酯50mL萃取三次,合并有机相,用无水硫酸钠干燥,减压浓缩得到化合物43c(1.6g粗品),无需纯化直接用于下一步反应。Compound 43b (1.2g, 4.22mmol) was suspended in 30mL of dry tetrahydrofuran, cooled to -78°C under nitrogen protection, and a solution of LiHMDS in tetrahydrofuran (6.33mL, 1mol/L, 6.33mmol) was slowly added dropwise, and the system was maintained at -78°C Stir for 30min. Then ethyl cyanoformate (627mg, 6.33mmol) was added dropwise, and after the dropwise addition, the system automatically rose to room temperature and reacted for 2h. After cooling to 0°C, 50 mL of ice water was added dropwise to quench the reaction. Then it was extracted three times with 50 mL of ethyl acetate, the organic phases were combined, dried over anhydrous sodium sulfate, and concentrated under reduced pressure to obtain compound 43c (1.6 g crude product), which was directly used in the next reaction without purification.
LCMS m/z=357.2[M+H]+ LCMS m/z=357.2[M+H] +
第三步:7-(3-(甲氧基甲氧基)萘-1-基)-2-(甲硫基)-5,6,7,8-四氢喹唑啉-4-醇(43d)The third step: 7-(3-(methoxymethoxy)naphthalene-1-yl)-2-(methylthio)-5,6,7,8-tetrahydroquinazolin-4-alcohol ( 43d)
7-(3-(methoxymethoxy)naphthalen-1-yl)-2-(methylthio)-5,6,7,8-tetrahydroquinazolin-4-ol.7-(3-(methoxymethoxy)naphthalen-1-yl)-2-(methylthio)-5,6,7,8-tetrahydroquinazolin-4-ol.
将化合物43c(1.6g粗品)溶于100mL乙醇和20mL水的混合溶液中,加入S-甲基异硫脲硫酸盐(11.75g,42.2mmol),碳酸氢钠(15.95g,189.9mmol)。氮气保护下加热至50℃反应12h。减压浓缩除去溶剂后加入50mL水稀释,水相用50mL乙酸乙酯萃取三次,合并有机相,用无水硫酸钠干燥,减压浓缩。柱层析分离纯化得到目标产物43d(950mg,两步收率:59%)。Compound 43c (1.6g crude product) was dissolved in a mixed solution of 100mL ethanol and 20mL water, and S-methylisothiourea sulfate (11.75g, 42.2mmol) and sodium bicarbonate (15.95g, 189.9mmol) were added. Under the protection of nitrogen, it was heated to 50° C. for 12 h. Concentrate under reduced pressure to remove the solvent, add 50 mL of water to dilute, extract the aqueous phase with 50 mL of ethyl acetate three times, combine the organic phases, dry over anhydrous sodium sulfate, and concentrate under reduced pressure. Separation and purification by column chromatography gave the target product 43d (950 mg, two-step yield: 59%).
LCMS m/z=383.1[M+H]+ LCMS m/z=383.1[M+H] +
第四步:7-(3-(甲氧基甲氧基)萘-1-基)-2-(甲硫基)-5,6,7,8-四氢喹唑啉-4-基三氟甲磺酸酯(43e)The fourth step: 7-(3-(methoxymethoxy)naphthalen-1-yl)-2-(methylthio)-5,6,7,8-tetrahydroquinazolin-4-yltri Flate (43e)
7-(3-(methoxymethoxy)naphthalen-1-yl)-2-(methylthio)-5,6,7,8-tetrahydroquinazolin-4-yl trifluoromethanesulfonate.7-(3-(methoxymethoxy)naphthalen-1-yl)-2-(methylthio)-5,6,7,8-tetrahydroquinazolin-4-yl trifluoromethanesulfonate.
将化合物43d(950mg,2.49mmol)溶于30mL二氯甲烷中,加入N,N-二异丙基乙胺(1.65 mL,9.96mmol),冰浴下加入三氟甲磺酸酐(0.84mL,4.98mmol),冰浴下搅拌1h。加入20mL二氯甲烷稀释,后用20mL水洗涤反应,再用20mL饱和碳酸氢钠水溶液洗涤有机相,有机层用无水硫酸钠干燥,减压浓缩,得到目标粗产物43e(940mg粗品)。Compound 43d (950 mg, 2.49 mmol) was dissolved in 30 mL of dichloromethane, N,N-diisopropylethylamine (1.65 mL, 9.96 mmol) was added, trifluoromethanesulfonic anhydride (0.84 mL, 4.98 mmol), stirred in ice bath for 1h. Add 20 mL of dichloromethane to dilute, then wash the reaction with 20 mL of water, then wash the organic phase with 20 mL of saturated aqueous sodium bicarbonate solution, dry the organic layer over anhydrous sodium sulfate, and concentrate under reduced pressure to obtain the target crude product 43e (940 mg of crude product).
LCMS m/z=515.1[M+H]+ LCMS m/z=515.1[M+H] +
第五步:3-(7-(3-(甲氧基甲氧基)萘-1-基)-2-(甲硫基)-5,6,7,8-四氢喹唑啉-4-基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸叔丁酯(43f)The fifth step: 3-(7-(3-(methoxymethoxy)naphthalen-1-yl)-2-(methylthio)-5,6,7,8-tetrahydroquinazoline-4 -yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylic acid tert-butyl ester (43f)
tert-butyl 3-(7-(3-(methoxymethoxy)naphthalen-1-yl)-2-(methylthio)-5,6,7,8-tetrahydroquinazolin-4-yl)-3,8-di azabicyclo[3.2.1]octane-8-carboxylate.tert-butyl 3-(7-(3-(methoxymethoxy)naphthalen-1-yl)-2-(methylthio)-5,6,7,8-tetrahydroquinazolin-4-yl)-3,8-di azabicyclo[3.2 .1] octane-8-carboxylate.
将化合物43e(940mg粗品)溶于20mL N,N-二甲基乙酰胺中,依次加入N,N-二异丙基乙胺(1.65mL,9.96mmol)和3,8-二氮杂双环[3.2.1]辛烷-8-羧酸叔丁酯(793mg,3.74mmol),然后于室温下搅拌3h。加入100mL乙酸乙酯稀释,用20mL水洗有机相三次,20mL饱和食盐水洗一次,有机相用无水硫酸钠干燥,减压浓缩。柱层析后得到目标粗产物43f(920mg 粗品)。Compound 43e (940 mg crude product) was dissolved in 20 mL N,N-dimethylacetamide, N, N-diisopropylethylamine (1.65 mL, 9.96 mmol) and 3,8-diazabicyclo[ 3.2.1] tert-butyl octane-8-carboxylate (793mg, 3.74mmol), then stirred at room temperature for 3h. Add 100 mL of ethyl acetate for dilution, wash the organic phase three times with 20 mL of water and once with 20 mL of saturated brine, dry the organic phase over anhydrous sodium sulfate, and concentrate under reduced pressure. The target crude product 43f (920 mg crude) was obtained after column chromatography.
LCMS m/z=577.2[M+H]+ LCMS m/z=577.2[M+H] +
第六步:叔丁基3-(7-(3-(甲氧基甲氧基)萘-1-基)-2-(甲基磺酰基)-5,6,7,8-四氢喹唑啉-4- 基)-3,8-二氮杂双环[3.2.1]辛烷-8-羧酸酯(43g)The sixth step: tert-butyl 3-(7-(3-(methoxymethoxy)naphthalen-1-yl)-2-(methylsulfonyl)-5,6,7,8-tetrahydroquinone Azolin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate (43g)
tert-butyl 3-(7-(3-(methoxymethoxy)naphthalen-1-yl)-2-(methylsulfonyl)-5,6,7,8-tetrahydroquinazolin-4-yl)-3, 8-diazabicyclo[3.2.1]octane-8-carboxylate.tert-butyl 3-(7-(3-(methoxymethoxy)naphthalen-1-yl)-2-(methylsulfonyl)-5,6,7,8-tetrahydroquinazolin-4-yl)-3, 8-diazabicyclo[3.2. 1] octane-8-carboxylate.
将化合物43f(920mg粗品)溶于20mL四氢呋喃中,室温下加入间氯过氧苯甲酸(1.72 g,9.96mmol),室温搅拌2h。加入20mL饱和硫代硫酸钠水溶液,搅拌20min,加入50mL乙酸乙酯萃取,有机层用20mL饱和碳酸氢钠水溶液洗涤,减压浓缩,柱层析得到目标化合物43g(459mg,三步收率:30%)。Compound 43f (920 mg crude product) was dissolved in 20 mL of tetrahydrofuran, m-chloroperoxybenzoic acid (1.72 g, 9.96 mmol) was added at room temperature, and stirred at room temperature for 2 h. Add 20mL of saturated aqueous sodium thiosulfate solution, stir for 20min, add 50mL of ethyl acetate for extraction, the organic layer is washed with 20mL of saturated aqueous sodium bicarbonate solution, concentrated under reduced pressure, column chromatography to obtain the target compound 43g (459mg, three-step yield: 30 %).
LCMS m/z=609.2[M+H]+ LCMS m/z=609.2[M+H] +
第七步:4-(3,8-二氮杂双环[3.2.1]辛烷-3-基)-2-((((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)-基) 甲氧基)-7-(3-(甲氧基甲氧基)萘-1-基)-5,6,7,8-四氢喹唑啉(43h)The seventh step: 4-(3,8-diazabicyclo[3.2.1]octane-3-yl)-2-((((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizine- 7a(5H)-yl)methoxy)-7-(3-(methoxymethoxy)naphthalene-1-yl)-5,6,7,8-tetrahydroquinazoline (43h)
4-(3,8-diazabicyclo[3.2.1]octan-3-yl)-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl) methoxy)-7-(3-(methoxymethoxy)naphthalen-1-yl)-5,6,7,8-tetrahydroquinazoline.4-(3,8-diazabicyclo[3.2.1]octan-3-yl)-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl) methoxy)-7- (3-(methoxymethoxy)naphthalen-1-yl)-5,6,7,8-tetrahydroquinazoline.
将化合物43g(459mg,0.755mmol)和中间体1c-1(240mg,1.51mmol)溶于20mL 甲苯中,冰浴下加入叔丁醇钠(109mg,1.13mmol),冰浴下搅拌1h。加入20mL水淬灭反应,加入50mL乙酸乙酯萃取三次,合并有机相,用无水硫酸钠干燥,减压浓缩。柱层析分离纯化得到目标产物43h(520mg,收率:95%)。Compound 43g (459mg, 0.755mmol) and intermediate 1c-1 (240mg, 1.51mmol) were dissolved in 20mL toluene, sodium tert-butoxide (109mg, 1.13mmol) was added under ice-cooling, and stirred for 1h under ice-cooling. Add 20 mL of water to quench the reaction, add 50 mL of ethyl acetate to extract three times, combine the organic phases, dry over anhydrous sodium sulfate, and concentrate under reduced pressure. Separation and purification by column chromatography gave the target product 43h (520 mg, yield: 95%).
LCMS m/z=588.3[M+H]+ LCMS m/z=588.3[M+H] +
第八步:4-(4-(3,8-二氮杂双环[3.2.1]辛烷-3-基)-2-(((2R,7aS)-2-氟四氢-1H-吡咯嗪-7a(5H)- 基)甲氧基)-5,6,7,8-四氢喹唑啉-7-基)萘-2-醇(化合物43)The eighth step: 4-(4-(3,8-diazabicyclo[3.2.1]octane-3-yl)-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrole Azin-7a(5H)-yl)methoxy)-5,6,7,8-tetrahydroquinazolin-7-yl)naphthalene-2-ol (compound 43)
4-(4-(3,8-diazabicyclo[3.2.1]octan-3-yl)-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)- yl)methoxy)-5,6,7,8-tetrahydroquinazolin-7-yl)naphthalen-2-ol4-(4-(3,8-diazabicyclo[3.2.1]octan-3-yl)-2-(((2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl)methoxy) -5,6,7,8-tetrahydroquinazolin-7-yl)naphthalen-2-ol
将化合物(43h)(0.52g,0.88mmol)溶解于二氯甲烷5mL中,加入三氟乙酸0.5mL,室温下反应10分钟,立即加入N,N-二异丙基乙胺调节pH至碱性。减压浓缩,残留物经制备HPLC纯化,制备液浓缩后冻干得到化合物43(23mg,产率:4.8%)。Dissolve compound (43h) (0.52g, 0.88mmol) in 5mL of dichloromethane, add 0.5mL of trifluoroacetic acid, react at room temperature for 10 minutes, immediately add N,N-diisopropylethylamine to adjust the pH to alkaline . Concentrate under reduced pressure, and the residue is purified by preparative HPLC. The preparation solution is concentrated and then lyophilized to obtain compound 43 (23 mg, yield: 4.8%).
HPLC制备方法:HPLC preparation method:
1.仪器:waters 2767制备液相;色谱柱:SunFire@Prep C18(19mm*250mm).1. Instrument: waters 2767 preparative liquid phase; chromatographic column: SunFire@Prep C 18 (19mm*250mm).
2.样品用DMF溶解,用0.45μM滤头过滤,制成样品液。2. Dissolve the sample in DMF and filter it with a 0.45 μM filter head to make a sample solution.
3.流动相A:乙腈;B:纯化水(含1%NH4HCO3);梯度洗脱,流动相A含量从5%-50%洗脱;流量:12mL/min;洗脱时间:18min。3. Mobile phase A: acetonitrile; B: purified water (containing 1% NH 4 HCO 3 ); gradient elution, the content of mobile phase A is eluted from 5% to 50%; flow rate: 12mL/min; elution time: 18min .
Ms m/z(ESI):544.2[M+H]+ Ms m/z(ESI):544.2[M+H] +
1H NMR(400MHz,CD3OD)δ8.04(d,1H),7.66(d,1H),7.41–7.27(m,2H),7.03–6.96(m, 2H),5.38–5.18(m,1H),4.23–4.40(m,3H),3.95–3.85(m,1H),3.76–3.66(m,1H),3.58–3.51 (m,2H),3.39–3.33(m,1H),3.26–3.11(m,4H),3.08–2.74(m,4H),2.67–2.57(m,1H),2.33– 2.12(m,3H),2.11–1.74(m,9H). 1 H NMR (400MHz, CD 3 OD) δ8.04(d,1H),7.66(d,1H),7.41–7.27(m,2H),7.03–6.96(m,2H),5.38–5.18(m, 1H),4.23–4.40(m,3H),3.95–3.85(m,1H),3.76–3.66(m,1H),3.58–3.51 (m,2H),3.39–3.33(m,1H),3.26– 3.11(m,4H),3.08–2.74(m,4H),2.67–2.57(m,1H),2.33–2.12(m,3H),2.11–1.74(m,9H).
生物测试例biological test case
生物测试1:体外KRAS G12D结合检测实验Biological test 1: In vitro KRAS G12D binding assay
方法一:method one:
人重组蛋白Tag1-RAF1与Tag2-KRAS-G12D配制成2.5×的蛋白溶液。在384孔板中加入 2μL不同浓度的化合物,加入4μL 2.5×的Tag1-RAF1与4μL 2.5×的Tag2-KRAS-G12D蛋白,室温孵育60分钟。孵育结束后,加入10μL预先混合的抗体Tag1-Eu和抗体Tag2-XL665到微孔板中,常温孵育2小时后,使用多功能酶标仪设置激发光为320nM,发射光为615nM 和665nM,读615nm(Eu)和665nm(d2)的荧光信号。根据式(1)计算信号比值Ratio,运用 PrismGraphPad 7.0软件计算IC50值。Human recombinant protein Tag1-RAF1 and Tag2-KRAS-G12D were prepared into a 2.5× protein solution. Add 2 μL of different concentrations of compounds to a 384-well plate, add 4 μL of 2.5× Tag1-RAF1 and 4 μL of 2.5× Tag2-KRAS-G12D protein, and incubate at room temperature for 60 minutes. After the incubation, add 10 μL pre-mixed antibody Tag1-Eu and antibody Tag2-XL665 to the microwell plate, incubate at room temperature for 2 hours, use a multi-functional microplate reader to set the excitation light to 320nM, the emission light to 615nM and 665nM, read Fluorescent signals at 615nm (Eu) and 665nm (d2). Calculate the signal ratio Ratio according to formula (1), and use PrismGraphPad 7.0 software to calculate the IC 50 value.
Ratio=[Signal 665]/[Signal 615]*104 (式1)Ratio=[Signal 665]/[Signal 615]*10 4 (Formula 1)
方法二:Method Two:
向384孔白板中加入2μL稀释缓冲液稀释后的不同浓度的化合物。用稀释缓冲液对250X Tag2-KRAS-G12D蛋白和10mM GTP分别进行50倍和200倍稀释后,取混合液4μL加入测试板中。用稀释缓冲液对500X Tag1-RAF进行100倍稀释并取4μL加入测试板中;测试板在25℃孵育15分钟。孵育结束后,使用检测缓冲液对anti-Tag1-Eu和anti-Tag2-XL665分别进行 100倍和50倍稀释后,取10μL混合液加入测试板中,4℃孵育2小时。反应结束后采用多标记分析仪读取信号值,excitation:320nm,emission:615nm,665nm。Add 2 μL of the compound at different concentrations diluted in dilution buffer to the 384-well white plate. 250X Tag2-KRAS-G12D protein and 10mM GTP were diluted 50-fold and 200-fold with dilution buffer, respectively, and 4 μL of the mixture was added to the test plate. 500X Tag1-RAF was diluted 100 times with dilution buffer and 4 μL was added to the test plate; the test plate was incubated at 25°C for 15 minutes. After incubation, anti-Tag1-Eu and anti-Tag2-XL665 were diluted 100-fold and 50-fold with detection buffer, respectively, and 10 μL of the mixture was added to the test plate, and incubated at 4°C for 2 hours. After the reaction, use a multi-label analyzer to read the signal value, excitation: 320nm, emission: 615nm, 665nm.
采用公式Ratio=[Signal 665]/[Signal 615]×104计算信号比值,并使用式2计算抑制率,其中Rsample为化合物孔信号比值,Rmin为无KRAS G12D蛋白对照孔信号比值,Rmax为含有 1%DMSO溶剂对照孔信号比值,使用GraphPad Prism软件通过四参数进行曲线拟合并计算IC50值。试验结果见表1。Use the formula Ratio=[Signal 665]/[Signal 615]×10 4 to calculate the signal ratio, and use the formula 2 to calculate the inhibition rate, where R sample is the signal ratio of compound wells, R min is the signal ratio of control wells without KRAS G12D protein, R max is the signal ratio of the control well containing 1% DMSO solvent, and the GraphPad Prism software is used to perform curve fitting with four parameters and calculate the IC 50 value. The test results are shown in Table 1.
Inhibition%=(1-(Rsample-Rmin)/(Rmax-Rmin))×100%(式2)Inhibition%=(1-(R sample -R min )/(R max -R min ))×100% (Formula 2)
表1本发明化合物的IC50值Table 1 IC 50 values of compounds of the present invention
A≤100nM;100nM<B≤1000nMA≤100nM; 100nM<B≤1000nM
结论:本发明的化合物对KRAS G12D与RAF1的蛋白相互作用具有良好的抑制活性。Conclusion: the compound of the present invention has good inhibitory activity on the protein interaction between KRAS G12D and RAF1.
生物测试2:细胞增殖抑制实验Biological Test 2: Cell Proliferation Inhibition Experiment
ASPC-1细胞培养条件:RPMI-1640+10%FBS+1%双抗,培养于37℃,5%CO2孵箱中。第一天收集指数生长期的ASPC-1细胞铺板96孔培养板,每孔80μL,铺板密度为1000个/ 孔,于37℃,5%CO2孵箱中培养过夜,铺板同时铺T0孔。第二天每孔加入20μL不同浓度化合物,使每孔DMSO终浓度均为0.5%,于37℃,5%CO2孵箱中培养6天。第二天加药同时使用CellTiter-Glo试剂盒检测T0板,记为RLU0。培养结束后,每孔加入25μL检测液(Cell4Viability Assay,Promega),混匀2分钟,室温孵育10分钟,使用Nivo多标记分析仪(PerkinElmer) 检测化学发光读数。结果按照式(3)处理,计算出化合物各个浓度的增殖抑制率,并使用GraphPad Prism软件,采用四参数进行曲线拟合,计算增殖抑制率为50%时化合物的浓度GI50值。RLUcompound为药物处理组的读数,RLUcontrol为溶剂对照组的平均值。试验结果见表2。ASPC-1 cell culture conditions: RPMI-1640+10% FBS+1% double antibody, cultured at 37°C, 5% CO 2 incubator. On the first day, collect ASPC-1 cells in the exponential growth phase and plate them on 96-well culture plates, 80 μL per well, at a plate density of 1000 cells/well, and culture overnight in a 5% CO 2 incubator at 37°C, and plate T 0 wells at the same time . On the second day, 20 μL of different concentrations of compounds were added to each well so that the final concentration of DMSO in each well was 0.5%, and cultured at 37° C. in a 5% CO 2 incubator for 6 days. The next day, add the drug and use the CellTiter-Glo kit to detect the T 0 plate, which is recorded as RLU 0 . After the incubation, 25 μL of detection solution (Cell4Viability Assay, Promega) was added to each well, mixed for 2 minutes, incubated at room temperature for 10 minutes, and the chemiluminescence reading was detected using a Nivo multilabel analyzer (PerkinElmer). The results were processed according to formula (3), and the growth inhibition rate of each concentration of the compound was calculated, and the GraphPad Prism software was used to carry out curve fitting with four parameters, and the concentration GI 50 value of the compound was calculated when the growth inhibition rate was 50%. RLU compound is the reading of the drug treatment group, and RLU control is the mean value of the solvent control group. The test results are shown in Table 2.
Inhibition%=(1-(RLUcompound-RLU0)/(RLUcontrol-RLU0))×100% 式(3)Inhibition%=(1-(RLU compound -RLU 0 )/(RLU control -RLU 0 ))×100% Formula (3)
表2本发明化合物的增殖抑制活性GI50值Table 2 The growth inhibitory activity GI 50 value of the compound of the present invention
A≤100nM;100nM<B≤1000nM.A≤100nM; 100nM<B≤1000nM.
结论:本发明的化合物对ASPC-1细胞具有良好的增殖抑制活性。Conclusion: the compound of the present invention has good growth inhibitory activity on ASPC-1 cells.
Claims (15)
Applications Claiming Priority (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110470763 | 2021-04-30 | ||
| CN2021104707632 | 2021-04-30 | ||
| CN2021108408606 | 2021-07-27 | ||
| CN202110840860 | 2021-07-27 | ||
| CN2021110920217 | 2021-09-17 | ||
| CN202111092021 | 2021-09-17 | ||
| CN202210090554 | 2022-01-27 | ||
| CN202210090554X | 2022-01-27 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN115490709A true CN115490709A (en) | 2022-12-20 |
Family
ID=84464386
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202210444808.3A Withdrawn CN115490709A (en) | 2021-04-30 | 2022-04-27 | A KRASG12D inhibitor and its application in medicine |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN115490709A (en) |
Cited By (29)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN115141215A (en) * | 2021-03-30 | 2022-10-04 | 上海德琪医药科技有限公司 | KRAS G12D protein inhibitors and uses thereof |
| CN115836072A (en) * | 2020-06-04 | 2023-03-21 | 上海德琪医药科技有限公司 | Inhibitors of KRAS G12C protein and uses thereof |
| CN116120315A (en) * | 2023-04-19 | 2023-05-16 | 山东绿叶制药有限公司 | KRAS G12C inhibitor and application thereof |
| CN116262759A (en) * | 2021-12-31 | 2023-06-16 | 华润医药研究院(深圳)有限公司 | Pyrimidine tricyclic compound, and preparation method and medical application thereof |
| WO2023125627A1 (en) * | 2021-12-28 | 2023-07-06 | Lynk Pharmaceuticals Co., Ltd. | Nitrogen-containing heterocyclic compound and application thereof |
| WO2023138583A1 (en) * | 2022-01-21 | 2023-07-27 | 上海湃隆生物科技有限公司 | Heterocyclic compound, pharmaceutical composition and use thereof |
| WO2023198191A1 (en) * | 2022-04-15 | 2023-10-19 | 杭州多域生物技术有限公司 | Six- and six-membered compound, preparation method, pharmaceutical composition, and application |
| WO2024008179A1 (en) * | 2022-07-07 | 2024-01-11 | Beigene, Ltd. | Heterocyclic compounds, compositions thereof, and methods of treatment therewith |
| WO2024012456A1 (en) * | 2022-07-12 | 2024-01-18 | 南京明德新药研发有限公司 | Piperazine bridge-substituted heterocyclic pyrimidine compound |
| CN117440953A (en) * | 2021-12-09 | 2024-01-23 | 苏州浦合医药科技有限公司 | Substituted bicyclic heteroaryls as KRAS G12D inhibitors |
| WO2024206858A1 (en) | 2023-03-30 | 2024-10-03 | Revolution Medicines, Inc. | Compositions for inducing ras gtp hydrolysis and uses thereof |
| WO2024229406A1 (en) | 2023-05-04 | 2024-11-07 | Revolution Medicines, Inc. | Combination therapy for a ras related disease or disorder |
| CN118955610A (en) * | 2024-10-15 | 2024-11-15 | 正大天晴药业集团股份有限公司 | Benzopyrimidine compounds |
| WO2025034702A1 (en) | 2023-08-07 | 2025-02-13 | Revolution Medicines, Inc. | Rmc-6291 for use in the treatment of ras protein-related disease or disorder |
| WO2025080946A2 (en) | 2023-10-12 | 2025-04-17 | Revolution Medicines, Inc. | Ras inhibitors |
| WO2025077770A1 (en) * | 2023-10-10 | 2025-04-17 | 成都海博为药业有限公司 | Fused ring compound and use thereof in kras inhibitor |
| WO2025077663A1 (en) * | 2023-10-08 | 2025-04-17 | 成都海博为药业有限公司 | Fused ring compound and use |
| US12286427B2 (en) | 2020-08-28 | 2025-04-29 | Kumquat Biosciences Inc. | Heterocyclic compounds and uses thereof |
| WO2025162091A1 (en) * | 2024-01-30 | 2025-08-07 | 药雅科技(上海)有限公司 | Preparation and use of pyrimidothiopyranone kras mutant protein inhibitor |
| WO2025171296A1 (en) | 2024-02-09 | 2025-08-14 | Revolution Medicines, Inc. | Ras inhibitors |
| US12448400B2 (en) | 2023-09-08 | 2025-10-21 | Gilead Sciences, Inc. | KRAS G12D modulating compounds |
| WO2025240847A1 (en) | 2024-05-17 | 2025-11-20 | Revolution Medicines, Inc. | Ras inhibitors |
| WO2025255438A1 (en) | 2024-06-07 | 2025-12-11 | Revolution Medicines, Inc. | Methods of treating a ras protein-related disease or disorder |
| WO2025265060A1 (en) | 2024-06-21 | 2025-12-26 | Revolution Medicines, Inc. | Therapeutic compositions and methods for managing treatment-related effects |
| WO2026006747A1 (en) | 2024-06-28 | 2026-01-02 | Revolution Medicines, Inc. | Ras inhibitors |
| WO2026015825A1 (en) | 2024-07-12 | 2026-01-15 | Revolution Medicines, Inc. | Use of ras inhibitor for treating pancreatic cancer |
| WO2026015801A1 (en) | 2024-07-12 | 2026-01-15 | Revolution Medicines, Inc. | Methods of treating a ras related disease or disorder |
| WO2026015790A1 (en) | 2024-07-12 | 2026-01-15 | Revolution Medicines, Inc. | Methods of treating a ras related disease or disorder |
| WO2026015796A1 (en) | 2024-07-12 | 2026-01-15 | Revolution Medicines, Inc. | Methods of treating a ras related disease or disorder |
-
2022
- 2022-04-27 CN CN202210444808.3A patent/CN115490709A/en not_active Withdrawn
Cited By (37)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN115836072A (en) * | 2020-06-04 | 2023-03-21 | 上海德琪医药科技有限公司 | Inhibitors of KRAS G12C protein and uses thereof |
| CN115836072B (en) * | 2020-06-04 | 2025-10-14 | 上海德琪医药科技有限公司 | KRAS G12C protein inhibitors and uses thereof |
| US12365676B2 (en) | 2020-08-28 | 2025-07-22 | Kumquat Biosciences Inc. | Heterocyclic compounds and uses thereof |
| US12286427B2 (en) | 2020-08-28 | 2025-04-29 | Kumquat Biosciences Inc. | Heterocyclic compounds and uses thereof |
| CN115141215B (en) * | 2021-03-30 | 2023-09-15 | 上海德琪医药科技有限公司 | KRAS G12D protein inhibitors and their uses |
| CN115141215A (en) * | 2021-03-30 | 2022-10-04 | 上海德琪医药科技有限公司 | KRAS G12D protein inhibitors and uses thereof |
| CN117440953A (en) * | 2021-12-09 | 2024-01-23 | 苏州浦合医药科技有限公司 | Substituted bicyclic heteroaryls as KRAS G12D inhibitors |
| CN117440953B (en) * | 2021-12-09 | 2025-11-21 | 苏州浦合医药科技有限公司 | Substituted bicyclic heteroaryl compounds as KRAS G12D inhibitors |
| WO2023125627A1 (en) * | 2021-12-28 | 2023-07-06 | Lynk Pharmaceuticals Co., Ltd. | Nitrogen-containing heterocyclic compound and application thereof |
| CN118510785B (en) * | 2021-12-28 | 2025-03-18 | 凌科药业(杭州)有限公司 | A nitrogen-containing heterocyclic compound and its application |
| CN118510785A (en) * | 2021-12-28 | 2024-08-16 | 凌科药业(杭州)有限公司 | Nitrogen-containing heterocyclic compound and application thereof |
| CN116262759B (en) * | 2021-12-31 | 2023-12-08 | 华润医药研究院(深圳)有限公司 | Pyrimidine tricyclic compound, and preparation method and medical application thereof |
| CN116262759A (en) * | 2021-12-31 | 2023-06-16 | 华润医药研究院(深圳)有限公司 | Pyrimidine tricyclic compound, and preparation method and medical application thereof |
| WO2023138583A1 (en) * | 2022-01-21 | 2023-07-27 | 上海湃隆生物科技有限公司 | Heterocyclic compound, pharmaceutical composition and use thereof |
| WO2023198191A1 (en) * | 2022-04-15 | 2023-10-19 | 杭州多域生物技术有限公司 | Six- and six-membered compound, preparation method, pharmaceutical composition, and application |
| WO2024008179A1 (en) * | 2022-07-07 | 2024-01-11 | Beigene, Ltd. | Heterocyclic compounds, compositions thereof, and methods of treatment therewith |
| WO2024012456A1 (en) * | 2022-07-12 | 2024-01-18 | 南京明德新药研发有限公司 | Piperazine bridge-substituted heterocyclic pyrimidine compound |
| WO2024206858A1 (en) | 2023-03-30 | 2024-10-03 | Revolution Medicines, Inc. | Compositions for inducing ras gtp hydrolysis and uses thereof |
| CN116120315B (en) * | 2023-04-19 | 2023-06-09 | 山东绿叶制药有限公司 | KRAS G12C inhibitor and application thereof |
| CN116120315A (en) * | 2023-04-19 | 2023-05-16 | 山东绿叶制药有限公司 | KRAS G12C inhibitor and application thereof |
| WO2024229406A1 (en) | 2023-05-04 | 2024-11-07 | Revolution Medicines, Inc. | Combination therapy for a ras related disease or disorder |
| WO2025034702A1 (en) | 2023-08-07 | 2025-02-13 | Revolution Medicines, Inc. | Rmc-6291 for use in the treatment of ras protein-related disease or disorder |
| US12448400B2 (en) | 2023-09-08 | 2025-10-21 | Gilead Sciences, Inc. | KRAS G12D modulating compounds |
| WO2025077663A1 (en) * | 2023-10-08 | 2025-04-17 | 成都海博为药业有限公司 | Fused ring compound and use |
| WO2025077770A1 (en) * | 2023-10-10 | 2025-04-17 | 成都海博为药业有限公司 | Fused ring compound and use thereof in kras inhibitor |
| WO2025080946A2 (en) | 2023-10-12 | 2025-04-17 | Revolution Medicines, Inc. | Ras inhibitors |
| WO2025162091A1 (en) * | 2024-01-30 | 2025-08-07 | 药雅科技(上海)有限公司 | Preparation and use of pyrimidothiopyranone kras mutant protein inhibitor |
| WO2025171296A1 (en) | 2024-02-09 | 2025-08-14 | Revolution Medicines, Inc. | Ras inhibitors |
| WO2025240847A1 (en) | 2024-05-17 | 2025-11-20 | Revolution Medicines, Inc. | Ras inhibitors |
| WO2025255438A1 (en) | 2024-06-07 | 2025-12-11 | Revolution Medicines, Inc. | Methods of treating a ras protein-related disease or disorder |
| WO2025265060A1 (en) | 2024-06-21 | 2025-12-26 | Revolution Medicines, Inc. | Therapeutic compositions and methods for managing treatment-related effects |
| WO2026006747A1 (en) | 2024-06-28 | 2026-01-02 | Revolution Medicines, Inc. | Ras inhibitors |
| WO2026015825A1 (en) | 2024-07-12 | 2026-01-15 | Revolution Medicines, Inc. | Use of ras inhibitor for treating pancreatic cancer |
| WO2026015801A1 (en) | 2024-07-12 | 2026-01-15 | Revolution Medicines, Inc. | Methods of treating a ras related disease or disorder |
| WO2026015790A1 (en) | 2024-07-12 | 2026-01-15 | Revolution Medicines, Inc. | Methods of treating a ras related disease or disorder |
| WO2026015796A1 (en) | 2024-07-12 | 2026-01-15 | Revolution Medicines, Inc. | Methods of treating a ras related disease or disorder |
| CN118955610A (en) * | 2024-10-15 | 2024-11-15 | 正大天晴药业集团股份有限公司 | Benzopyrimidine compounds |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN115490709A (en) | A KRASG12D inhibitor and its application in medicine | |
| CN115304623A (en) | A kind of pyrimidocyclic derivative and its application in medicine | |
| CN112368283B (en) | Bicyclic derivative-containing inhibitor, preparation method and application thereof | |
| ES2742409T3 (en) | Pyrazolylquinoxaline Kinase Inhibitors | |
| WO2023051716A1 (en) | Heteroaryl derivative parp inhibitor and use thereof | |
| TWI238163B (en) | Pyrrolotriazine inhibitors of kinases | |
| WO2023280237A1 (en) | Synthesis and application of phosphatase degrader | |
| CN113072551B (en) | Nitrogen-containing biphenyl derivative inhibitor, preparation method and application thereof | |
| WO2021249057A1 (en) | Heterocyclic compound and use thereof | |
| CN116375742A (en) | Azaaromatic ring derivative, composition and pharmaceutical application thereof | |
| WO2023041049A1 (en) | Heterocyclic compound as sos1 inhibitor and uses thereof | |
| JP2020525522A (en) | RHO-related protein kinase inhibitor, pharmaceutical composition containing the same, preparation method and use thereof | |
| KR102373577B1 (en) | Multikinase inhibitor compounds, crystalline forms and uses thereof | |
| BRPI0708615A2 (en) | heterobicyclic pyrazole compounds and methods of use | |
| TW202016102A (en) | ERK inhibitor and use thereof | |
| CN101918403A (en) | Pyrazolopyridines as Tyrosine Kinase Inhibitors | |
| JP2016512815A (en) | Condensed pyrazolopyrimidine derivatives useful as inhibitors of ATR kinase | |
| KR20150129741A (en) | Chemical entities | |
| JP2021536436A (en) | A novel inhibitor prepared from quinoline derivatives | |
| WO2020233669A1 (en) | Indole derivative-containing inhibitor, preparation method therefor and application thereof | |
| CN116848119A (en) | A kind of pyrrolopyridazine compound and its preparation method and use | |
| JP2023538091A (en) | Heterocyclic compounds as BTK inhibitors | |
| WO2019076336A1 (en) | Pyrazolyl-containing tricyclic derivative, preparation method therefor and use thereof | |
| CN115768757A (en) | CD206 regulator and its use and preparation method | |
| CN113727984B (en) | Macrocyclic derivatives, preparation method and application thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| TA01 | Transfer of patent application right |
Effective date of registration: 20231225 Address after: 856099 Xingfu Jiayuan Economic Development Zone, Gyerba, Nedong District, Shannan City, Tibet Autonomous Region Applicant after: Tibet Haisike Pharmaceutical Co.,Ltd. Address before: 611130 No.136 Baili Road, Wenjiang cross strait science and Technology Park, Chengdu, Sichuan Applicant before: SICHUAN HAISCO PHARMACEUTICAL Co.,Ltd. |
|
| TA01 | Transfer of patent application right | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| WW01 | Invention patent application withdrawn after publication |
Application publication date: 20221220 |
|
| WW01 | Invention patent application withdrawn after publication |