CN115340551A - Amino cyclic compound, organic electroluminescent material, and organic electroluminescent device - Google Patents
Amino cyclic compound, organic electroluminescent material, and organic electroluminescent device Download PDFInfo
- Publication number
- CN115340551A CN115340551A CN202110530783.4A CN202110530783A CN115340551A CN 115340551 A CN115340551 A CN 115340551A CN 202110530783 A CN202110530783 A CN 202110530783A CN 115340551 A CN115340551 A CN 115340551A
- Authority
- CN
- China
- Prior art keywords
- group
- substituted
- groups
- atoms
- compound
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/04—Ortho-condensed systems
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E10/00—Energy generation through renewable energy sources
- Y02E10/50—Photovoltaic [PV] energy
- Y02E10/549—Organic PV cells
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Electroluminescent Light Sources (AREA)
Abstract
本发明涉及一种氨基环状化合物、有机电致发光材料和有机电致发光装置,特别是所述氨基环状化合物的结构如式1所示,提供了一种新颖的具有非常高抗三线态解离能的空穴传输材料,具有三线态解离能(TN‑BDE)大于0.2ev的空穴传输材料,且具有良好的空穴传输特性和电子耐受性;进一步地,提供了具有三线态解离能TN‑BDE>=0.4ev的空穴传输材料。
The present invention relates to an amino cyclic compound, an organic electroluminescent material and an organic electroluminescent device. In particular, the structure of the amino cyclic compound is shown in formula 1, which provides a novel anti-triplet The hole-transporting material of dissociation energy has a triplet dissociation energy (TN-BDE) greater than 0.2eV hole-transporting material, and has good hole-transporting characteristics and electron tolerance; A hole transport material with state dissociation energy TN‑BDE>=0.4eV.
Description
技术领域technical field
本发明涉及电致发光器件材料技术领域,尤其涉及一种具有三线态稳定基团的特定排列的氨基环状化合物及包含该氨基环状化合物的有机电致发光材料和有机电致发光装置。The invention relates to the technical field of electroluminescent device materials, in particular to an amino cyclic compound with a specific arrangement of triplet stable groups, an organic electroluminescent material and an organic electroluminescent device containing the amino cyclic compound.
背景技术Background technique
含有有机、有机金属和/或聚合物半导体的电子器件正变得越来越重要,并且出于成本考虑和由于其性能而被用于许多商业产品中。此处的实例包括复印机、有机或聚合物发光二极管(OLED或PLED)中以及读出和显示器件中的有机类电荷传输材料(例如三芳基胺类空穴传输体)或复印机中的有机感光体。有机太阳能电池(O-SC)、有机场效应晶体管(O-FET)、有机薄膜晶体管(O-TFT)、有机集成电路(O-IC)、有机光放大器和有机激光二极管(O-laser)正处于开发的后期并且可具有重大的未来意义。Electronic devices containing organic, organometallic and/or polymeric semiconductors are becoming increasingly important and are used in many commercial products due to cost considerations and due to their performance. Examples here include organic-based charge-transport materials (e.g. triarylamine-based hole transporters) in copiers, organic or polymer light-emitting diodes (OLEDs or PLEDs) and in readout and display devices, or organic photoreceptors in copiers . Organic solar cell (O-SC), organic field effect transistor (O-FET), organic thin film transistor (O-TFT), organic integrated circuit (O-IC), organic optical amplifier and organic laser diode (O-laser) In a later stage of development and could be of great future significance.
一般来说,OLED包含阳极、阴极和有机发光单元。最后一者包含数个功能层,如空穴或电子注入层、空穴或电子传输层和有机发光层。蒸发技术是用于制造OLED器件的最常用技术。然而,其显示了显著的成本缺点,特别是对于大面积器件更是如此,因为在多种不同的室中进行多级真空工艺非常昂贵并且必须被非常精确地控制。具体地,所述工艺应该能以低成本进行生产制造。同时,该工艺应适用于制造非常小的单元结构,从而能够通过该工艺获得高分辨率屏幕。In general, an OLED includes an anode, a cathode, and an organic light emitting unit. The last one contains several functional layers, such as hole or electron injection layers, hole or electron transport layers and organic light-emitting layers. Evaporation technology is the most commonly used technology for fabricating OLED devices. However, it presents a significant cost disadvantage, especially for large area devices, since the multi-stage vacuum process in a variety of different chambers is very expensive and must be controlled very precisely. In particular, the process should be able to be manufactured at low cost. At the same time, the process should be suitable for making very small cell structures, so that high-resolution screens can be obtained through this process.
另一个问题特别是有机电致发光器件需达到指定目的的能量效率。在基于一种或多种OLED材料的有机发光二极管的情况下,其外量子效率应该足够高。此外,还应需要尽可能低的电压来达到指定发光密度。更具体地,直接连接到发光层、特别是空穴传输层和过渡层对相邻发光层的特性具有很大的影响。直接连接到空穴传输层的空穴注入层的质量在OLED的性能中也起着重要的作用。Another problem is in particular the energy efficiency of organic electroluminescent devices to achieve the intended purpose. In the case of organic light-emitting diodes based on one or more OLED materials, their external quantum efficiency should be sufficiently high. In addition, the lowest possible voltage should be required to achieve the specified luminous density. More specifically, the direct connection to the emitting layer, especially the hole transport layer and the transition layer, has a great influence on the properties of the adjacent emitting layer. The quality of the hole injection layer directly connected to the hole transport layer also plays an important role in the performance of the OLED.
目前,为了进一步提升器件的寿命和效率,还会在空穴传输层和发光层之间加入辅助层,使得从阳极转移的空穴能够平稳地移动到发光层,并且可以阻挡从阴极转移的电子,以将电子限制在发光层内,减少空穴传输层与发光层之间的势垒,降低有机电致发光器件的驱动电压,进一步增加空穴的利用率,从而改善器件的发光效率和寿命。目前作为发光辅助层的材料有限,同时发光辅助层的参数要求尚未明确。同时,目前现有的双咔唑类单体,在合成和使用过程中,具有合成收率低,溶解性差等情况。At present, in order to further improve the life and efficiency of the device, an auxiliary layer is added between the hole transport layer and the light-emitting layer, so that the holes transferred from the anode can move smoothly to the light-emitting layer, and can block the electrons transferred from the cathode. , to confine electrons in the light-emitting layer, reduce the potential barrier between the hole transport layer and the light-emitting layer, reduce the driving voltage of the organic electroluminescent device, and further increase the utilization rate of holes, thereby improving the luminous efficiency and life of the device . At present, the materials used as the luminescence auxiliary layer are limited, and the parameter requirements of the luminescence auxiliary layer have not yet been clarified. At the same time, the existing biscarbazole monomers have problems such as low synthesis yield and poor solubility during synthesis and use.
发明内容Contents of the invention
为解决现有技术的不足,本发明提出TSM-三线态稳定体triplet-statestability matrix的概念,提高整个分子的抗三线态破坏能力,同时使得处于激发态的电荷载流子稳定化(Triplet-states Bond dissociation energy(TN-BDE)>0.2ev),同时该三线态稳定体的加入显著改善了化合物理化性能,有利于提升器件效率,延长器件寿命。In order to solve the deficiencies in the prior art, the present invention proposes the concept of the triplet-state stability matrix of TSM-triplet state, which improves the anti-triplet state damage ability of the whole molecule, and simultaneously stabilizes the charge carriers in the excited state (Triplet-states Bond dissociation energy (TN-BDE)>0.2ev), and the addition of the triplet stabilizer significantly improves the physical and chemical properties of the compound, which is conducive to improving device efficiency and prolonging device life.
本发明的一个目的是提供一个新颖的含胺化合物。尤其是,提供一种新颖的具有非常高抗三线态解离能的空穴传输材料。更进一步,提供一种具有三线态解离能(TN-BDE)大于0.2ev的空穴传输材料,且具有良好的空穴传输特性和电子耐受性。更进一步,优选提供一种具有三线态解离能TN-BDE>=0.4ev的空穴传输材料。An object of the present invention is to provide a novel amine-containing compound. In particular, a novel hole transport material with very high resistance to triplet dissociation energy is provided. Furthermore, a hole-transporting material with a triplet dissociation energy (TN-BDE) greater than 0.2 eV is provided, and has good hole-transporting properties and electron tolerance. Furthermore, it is preferred to provide a hole transport material having a triplet dissociation energy TN-BDE>=0.4 eV.
具体的,本发明涉及一种氨基环状化合物,其结构可以用式1表示:Specifically, the present invention relates to an amino cyclic compound, the structure of which can be represented by Formula 1:
在上述式1中,In the
N1和N2彼此相同或不同的表示含氮环状结构;N1 and N2 are the same or different from each other and represent nitrogen-containing ring structures;
o/q独立的表示0、1、2、3或4;o/q independently represent 0, 1, 2, 3 or 4;
p表示0、1或2;p represents 0, 1 or 2;
R1、R2和R3彼此独立的表示H、D、F、Cl、Br、I、N(R)2、CN、NO2、Si(R)3、B(OR)2、C(=O)R、P(=O)(R)2、S(=O)R、S(=O)2R、OSO2R,具有1至40个C原子的直链烷基、烷氧基或硫代烷氧基基团,具有2至40个C原子的直链烯基或炔基基团,具有3至40个C原子的支链或环状的烷基、烯基、炔基、烷氧基、烷基烷氧基或硫代烷氧基基团,具有5至60个C原子的芳基或杂芳基,上述基团中的每个氢可以被一个或多个基团R取代,其中一个或多个非相邻的CH2基团可以被RC=CR、C≡C、Si(R)2、Ge(R)2、Sn(R)2、C=O、C=S、C=Se、C=NR、P(=O)(R)、SO、SO2、NR、O、S或CONR代替;R 1 , R 2 and R 3 independently represent H, D, F, Cl, Br, I, N(R) 2 , CN, NO 2 , Si(R) 3 , B(OR) 2 , C(= O)R, P(=O)(R) 2 , S(=O)R, S(=O) 2 R, OSO 2 R, straight-chain alkyl, alkoxy or Thioalkoxy groups, straight-chain alkenyl or alkynyl groups having 2 to 40 C atoms, branched or cyclic alkyl, alkenyl, alkynyl, alkane groups having 3 to 40 C atoms Oxy, alkylalkoxy or thioalkoxy groups, aryl or heteroaryl groups having 5 to 60 C atoms, each hydrogen of which may be replaced by one or more radicals R , where one or more non-adjacent CH2 groups can be represented by RC=CR, C≡C, Si(R) 2 , Ge(R) 2 , Sn(R) 2 , C=O, C=S, C =Se, C=NR, P(=O)(R), SO, SO 2 , NR, O, S or CONR instead;
L1和L2彼此相同或不同的表示为单键或二价基团,其中所述二价基团选自具有1~10个碳原子的直链亚烷基基团、具有3~10个碳原子的支链或环状的亚烷基基团、具有5~60芳族环原子并且可以被一个或多个非芳族基团取代的芳族或杂芳族环系、NR、O或S;L 1 and L 2 that are the same or different from each other represent a single bond or a divalent group, wherein the divalent group is selected from linear alkylene groups having 1 to 10 carbon atoms, linear alkylene groups having 3 to 10 A branched or cyclic alkylene group of carbon atoms, an aromatic or heteroaromatic ring system having 5 to 60 aromatic ring atoms and which may be substituted by one or more non-aromatic groups, NR, O or S;
Ar1选自C6-C60芳基基团、C3-C60脂环族基团或C1-C50烷基基团,其中上述基团中的每个氢可以被一个或多个基团R取代,其中一个或多个非相邻的CH2基团可以被RC=CR、C≡C、Si(R)2、Ge(R)2、Sn(R)2、C=O、C=S、C=Se、C=NR、P(=O)(R)、SO、SO2、NR、O、S或CONR代替;Ar is selected from a C6-C60 aryl group, a C3-C60 alicyclic group or a C1-C50 alkyl group, wherein each hydrogen in the above-mentioned groups can be replaced by one or more groups R, wherein One or more non-adjacent CH 2 groups can be represented by RC=CR, C≡C, Si(R) 2 , Ge(R) 2 , Sn(R) 2 , C=O, C=S, C= Se, C=NR, P(=O)(R), SO, SO 2 , NR, O, S or CONR instead;
Ar2选自C6-C60芳基基团、包含至少一个杂原子的C5-C60芳杂环基团、C3-C60脂环族基团或C1-C50烷基基团,其中上述基团中的每个氢可以被一个或多个基团R取代,其中一个或多个非相邻的CH2基团可以被RC=CR、C≡C、Si(R)2、Ge(R)2、Sn(R)2、C=O、C=S、C=Se、C=NR、P(=O)(R)、SO、SO2、NR、O、S或CONR代替;Ar 2 is selected from a C6-C60 aryl group, a C5-C60 aromatic heterocyclic group containing at least one heteroatom, a C3-C60 alicyclic group or a C1-C50 alkyl group, wherein the above-mentioned groups Each hydrogen can be replaced by one or more groups R, where one or more non-adjacent CH 2 groups can be replaced by RC=CR, C≡C, Si(R) 2 , Ge(R) 2 , Sn (R) 2 , C=O, C=S, C=Se, C=NR, P(=O)(R), SO, SO 2 , NR, O, S or CONR instead;
其中环A、B、C相同或不同地代表具有5至60个芳族环原子的芳族或杂芳族环系,所述芳族或杂芳族环系在可被一个或多个基团R取代;Wherein the rings A, B, C identically or differently represent an aromatic or heteroaromatic ring system having 5 to 60 aromatic ring atoms, said aromatic or heteroaromatic ring system can be surrounded by one or more groups R replaces;
基团R相同或不同地标识H、D、F,具有1至20个C原子的脂族、芳族和/或杂芳族烃基团,其中,一个或多个H原子可以被F代替;The radical R identifies H, D, F identically or differently, an aliphatic, aromatic and/or heteroaromatic hydrocarbon radical having 1 to 20 C atoms, wherein one or more H atoms may be replaced by F;
两个或更多个基团R可以彼此形成单环或多环的脂族或芳族环系。Two or more radicals R may form with one another a monocyclic or polycyclic aliphatic or aromatic ring system.
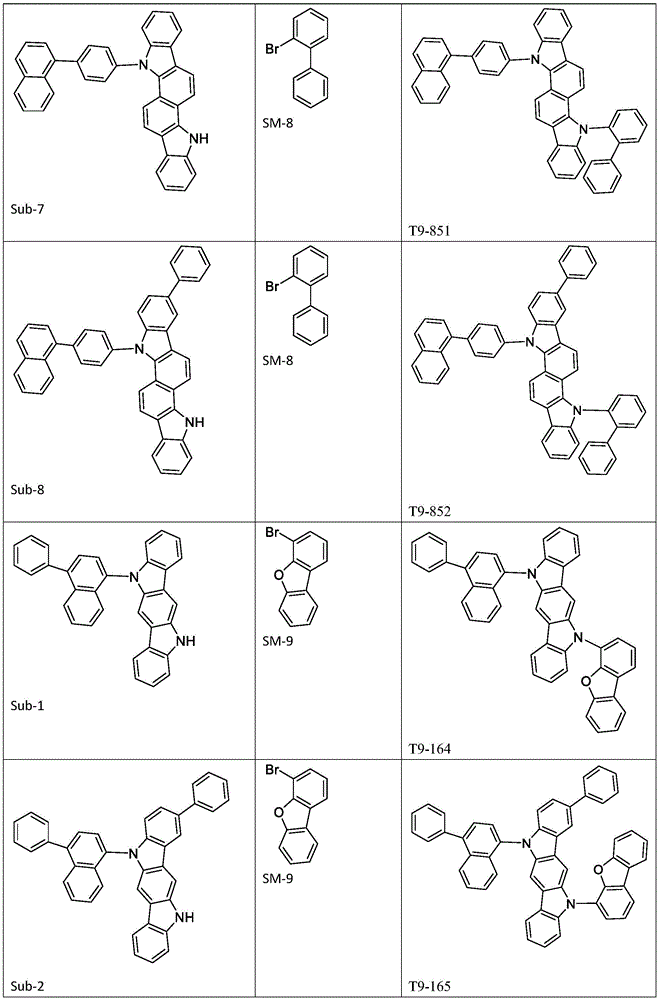
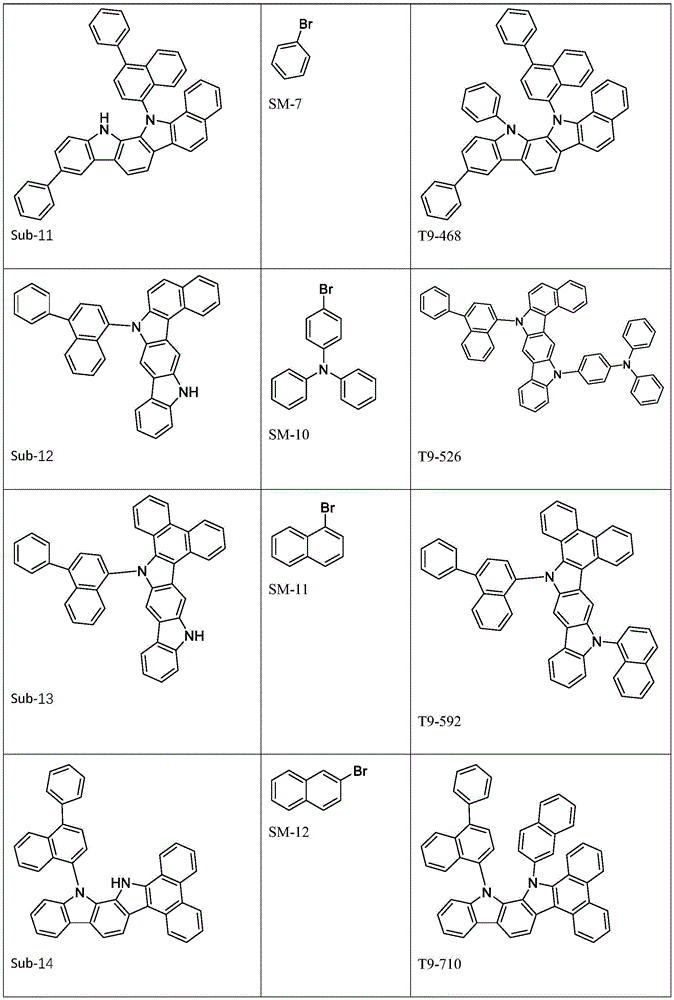
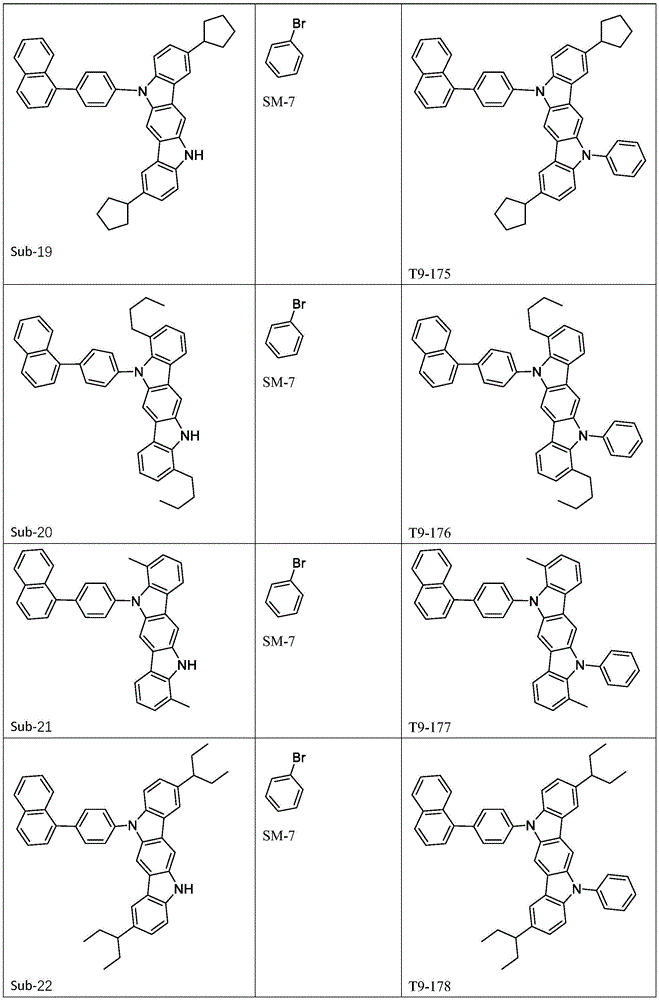
进一步地,所述式1所示结构的氨基环状化合物可由以下式1-1至式1-19中的任一个表示:Further, the amino cyclic compound with the structure shown in
Ar1、Ar2各自选自式3-1至式3-9所表示的结构;Ar 1 and Ar 2 are each selected from the structures represented by formula 3-1 to formula 3-9;
其中,环F、G、H、I各自独立地表示碳原子数为C6~C60的稠环芳基、R取代的碳原子数为C6~C60的稠环芳基、碳原子数为C5~C60的杂稠环芳基、R取代的碳原子数为C5~C60的杂稠环芳基、碳原子数为C3~C30的环烷基或R取代的碳原子数为C3~C30的环烷基,所述环烷基中任意一个或多个不相连的-CH2-任选被-RC=CR-、-C≡C-、-Si(R)2-、-Ge(R)2-、-Sn(R)2-、-C=O-、-C=S-、-C=Se-、-C=NR-、-P(=O)(R)-、-SO-、-SO2-、-NR-、-O-、S-或-CONR-取代;Among them, the rings F, G, H, and I each independently represent a condensed ring aryl group with a carbon number of C6 to C60, a condensed ring aryl group with a carbon number of C6 to C60 substituted by R, and a condensed ring aryl group with a carbon number of C5 to C60 Heterocondensed ring aryl group, heterofused ring aryl group with carbon atoms of C5-C60 substituted by R, cycloalkyl group with carbon atoms of C3-C30 or cycloalkyl group with carbon atoms of C3-C30 substituted by R , any one or more unconnected -CH2- in the cycloalkyl group is optionally replaced by -RC=CR-, -C≡C-, -Si(R)2-, -Ge(R)2-, - Sn(R)2-, -C=O-, -C=S-, -C=Se-, -C=NR-, -P(=O)(R)-, -SO-, -SO2-, -NR-, -O-, S- or -CONR-substitution;
Rc、Rd各自独立的代表H、D、F、Cl、Br、I、N(R)2、CN、NO2、Si(R)3、B(OR)2、C(=O)R、P(=O)(R)2、S(=O)R、S(=O)2R、OSO2R、碳原子数为C1~C40的烷基、R取代的碳原子数为C1~C40的烷基、碳原子数为C1~C40的烷氧基、R取代的碳原子数为C1~C40的烷氧基、硫取代的碳原子数为C1~C40的烷氧基、碳原子数为C2~C40的烯基、R取代的碳原子数为C2~C40的烯基、碳原子数为C2~C40的炔基、R取代的碳原子数为C2~C40的炔基、碳原子数为C6~C60的稠环芳基、R取代的碳原子数为C6~C60的稠环芳基、碳原子数为C5~C60的杂稠环芳基或R取代的碳原子数为C5~C60的杂稠环芳基,其中任意一个或多个不相连的-CH2-任选被-RC=CR-、-C≡C-、-Si(R)2-、-Ge(R)2-、-Sn(R)2-、-C=O-、-C=S-、-C=Se-、-C=NR-、-P(=O)(R)-、-SO-、-SO2-、-NR-、-O-、S-或-CONR-取代。R c and R d independently represent H, D, F, Cl, Br, I, N(R)2, CN, NO2, Si(R)3, B(OR)2, C(=O)R, P(=O)(R)2, S(=O)R, S(=O)2R, OSO2R, C1-C40 alkyl, R-substituted C1-C40 alkyl , an alkoxy group with C1-C40 carbon atoms, an alkoxy group with C1-C40 carbon atoms substituted by R, an alkoxy group with C1-C40 carbon atoms substituted by sulfur, and a C2-C40 carbon atom group Alkenyl, R-substituted alkenyl with C2-C40 carbons, C2-C40 alkynyl, R-substituted C2-C40 alkynyl, C6-C60 carbons fused ring aryl group, R-substituted fused ring aryl group with carbon atoms of C6-C60, hetero-fused ring aryl group with carbon atoms of C5-C60, or hetero-fused ring with carbon atoms of C5-C60 substituted by R Aryl, wherein any one or more unconnected -CH2- is optionally replaced by -RC=CR-, -C≡C-, -Si(R)2-, -Ge(R)2-, -Sn(R )2-, -C=O-, -C=S-, -C=Se-, -C=NR-, -P(=O)(R)-, -SO-, -SO2-, -NR- , -O-, S-, or -CONR-substitution.
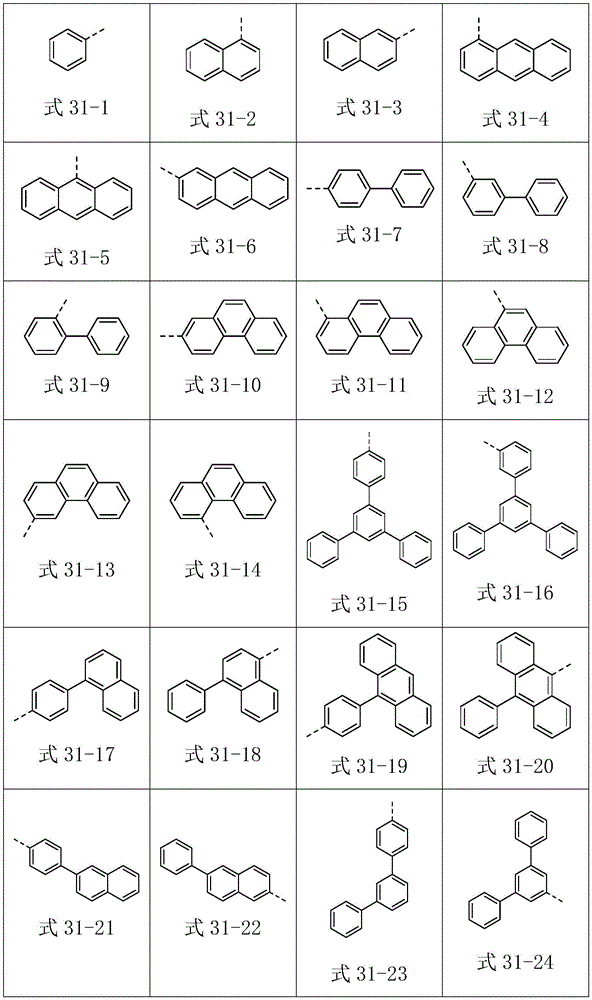
进一步地,Ar1选自以下式31-1至式31-88表示的基团:Further, Ar 1 is selected from groups represented by the following formula 31-1 to formula 31-88:
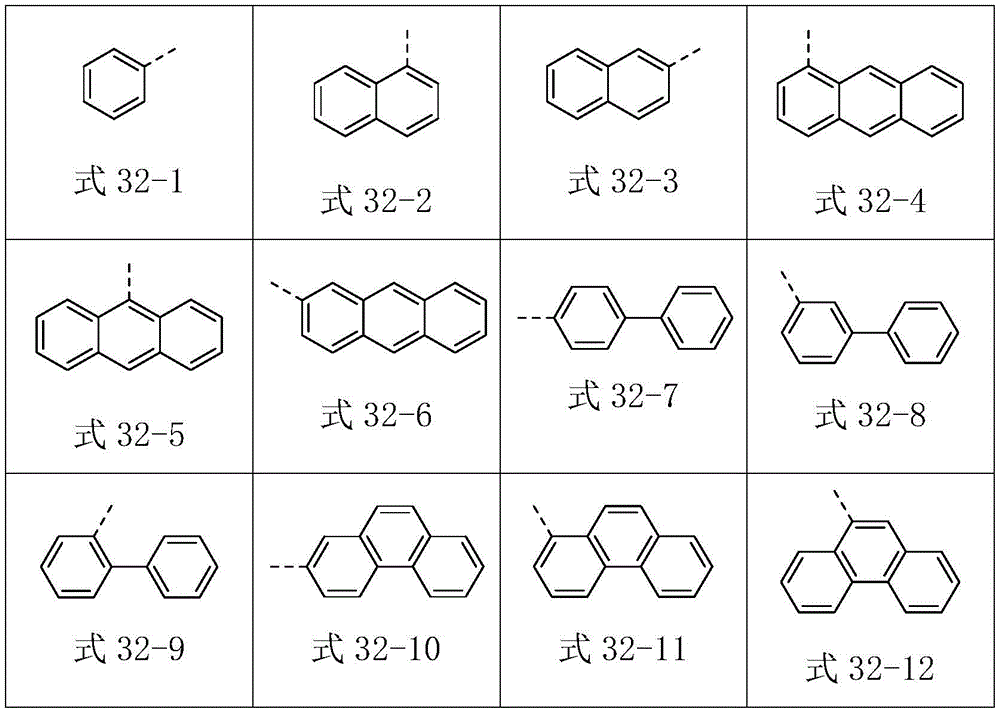
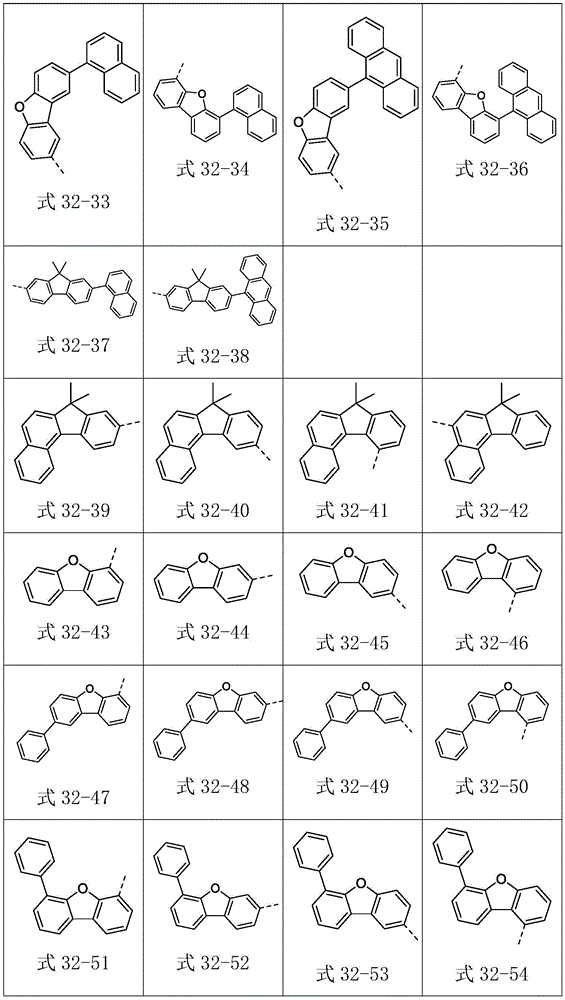
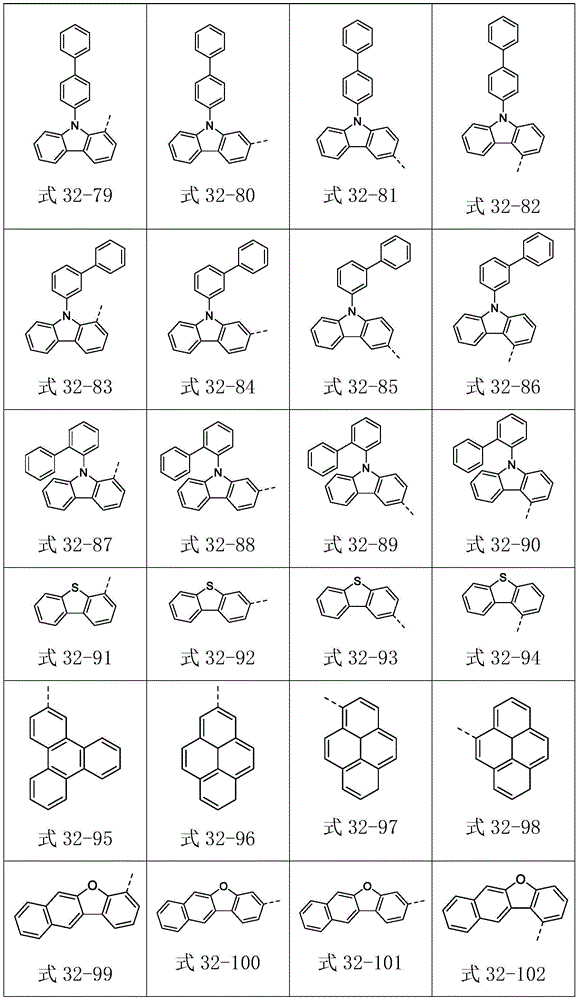
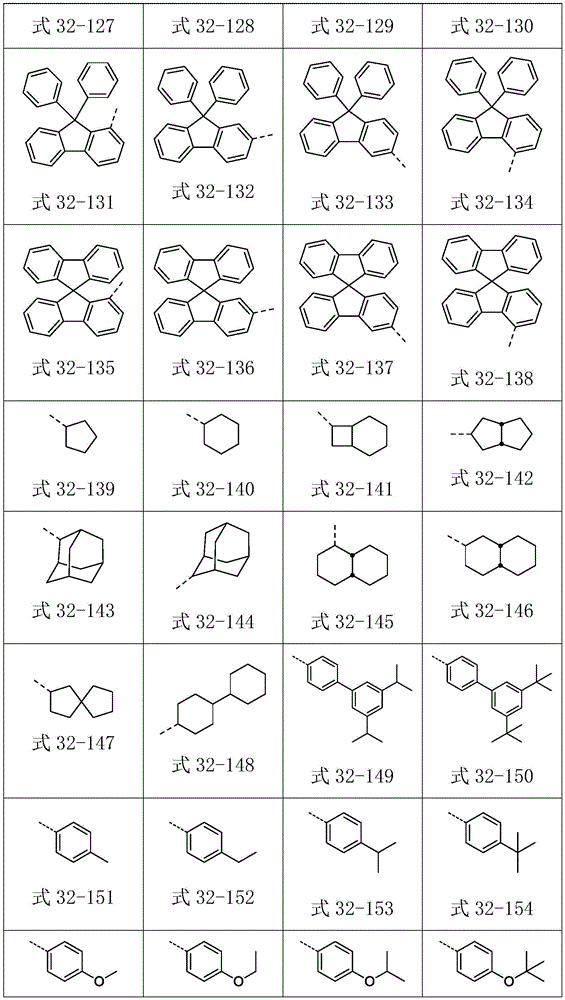
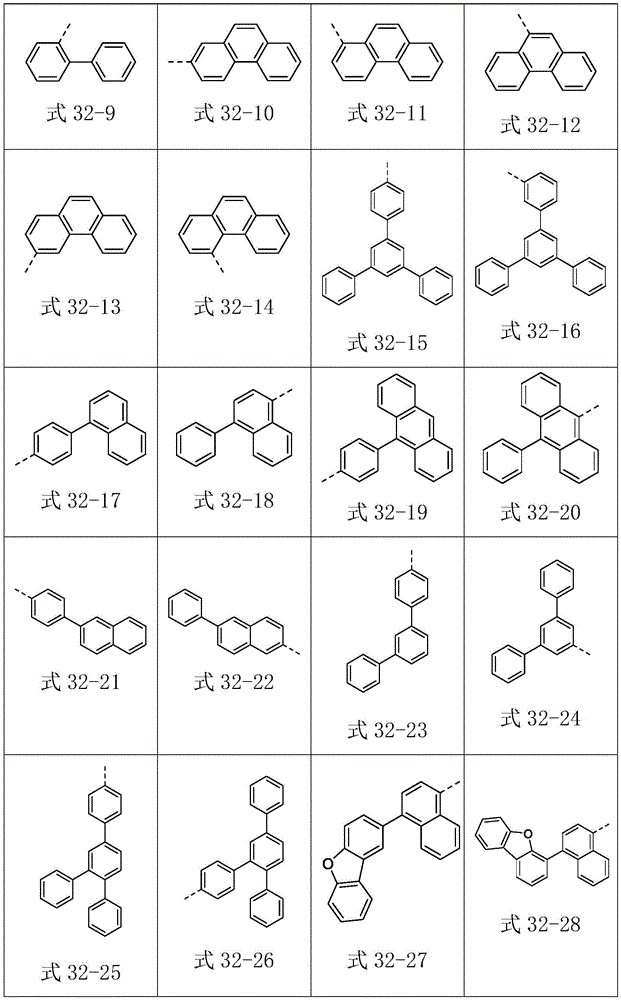
Ar2选自式32-1至式32-178表示的基团:Ar 2 is selected from groups represented by formula 32-1 to formula 32-178:
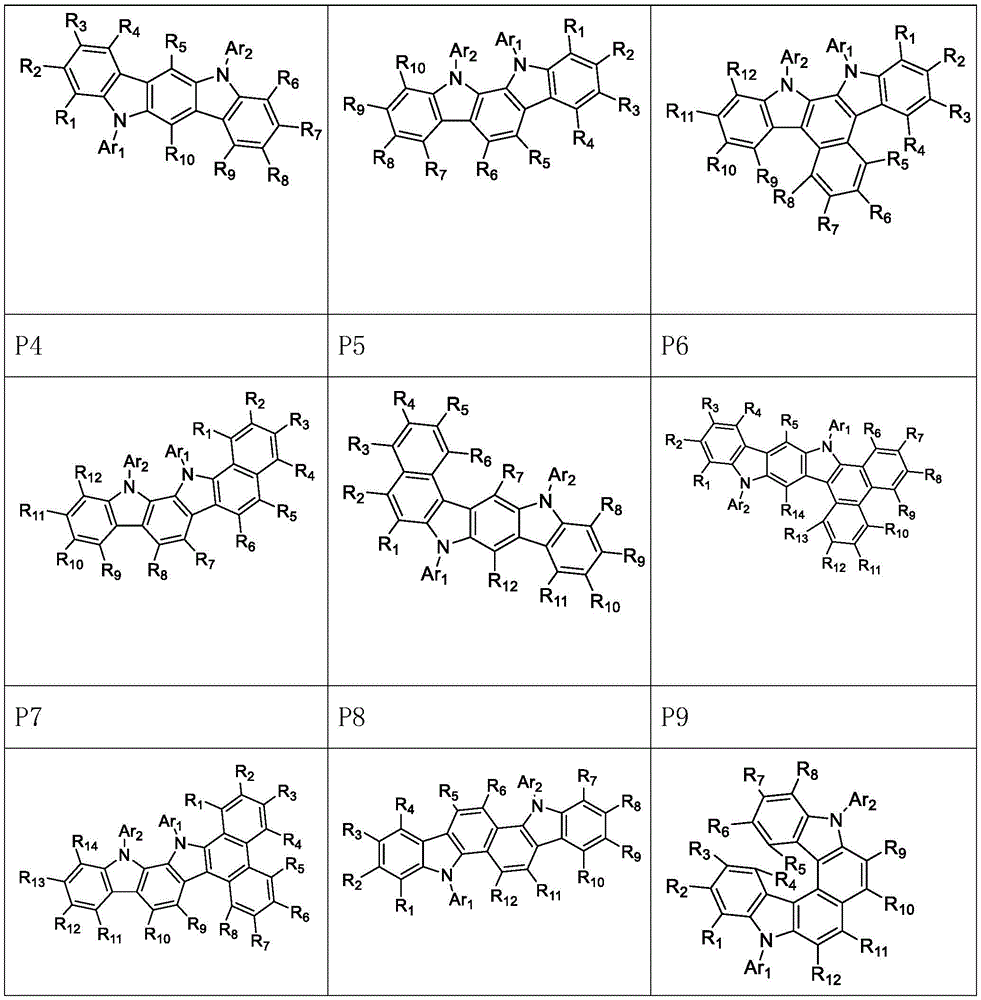
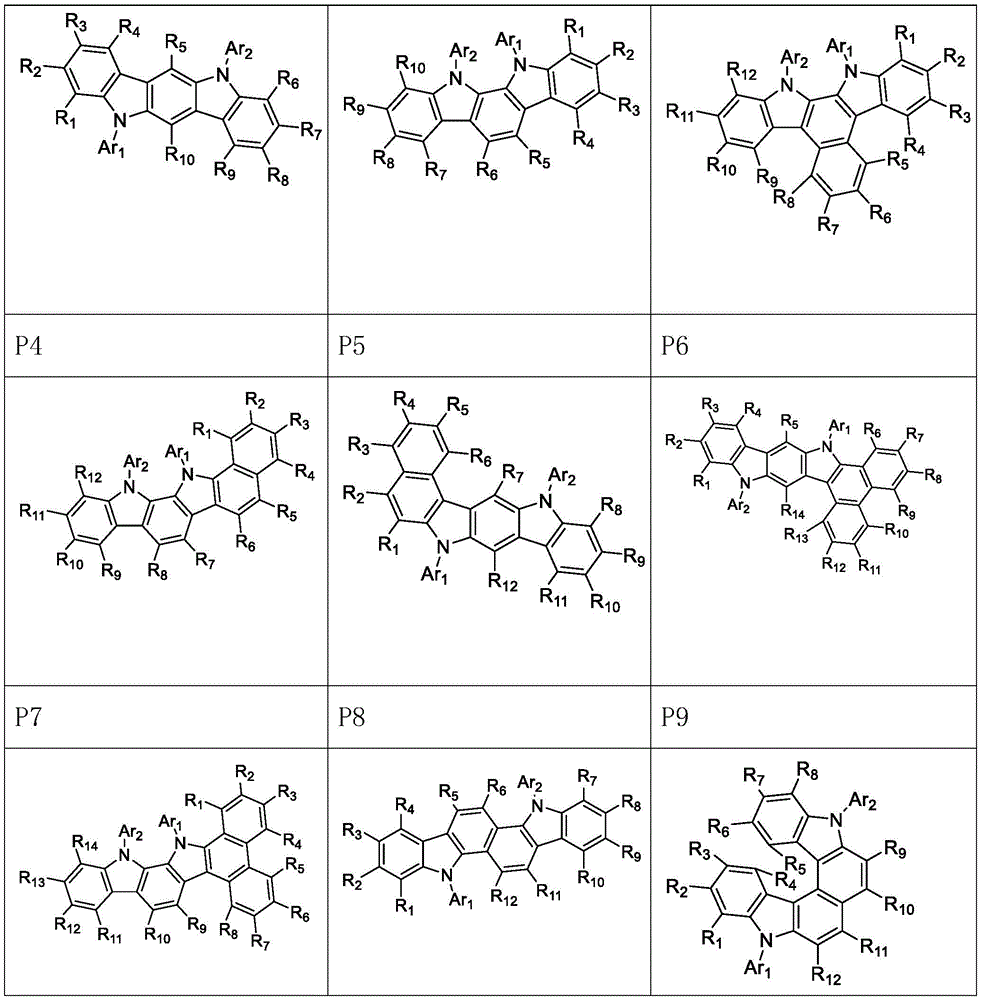
进一步地,所述氨基环状化合物由以下式P1至P9中的任一个表示:Further, the amino cyclic compound is represented by any one of the following formulas P1 to P9:
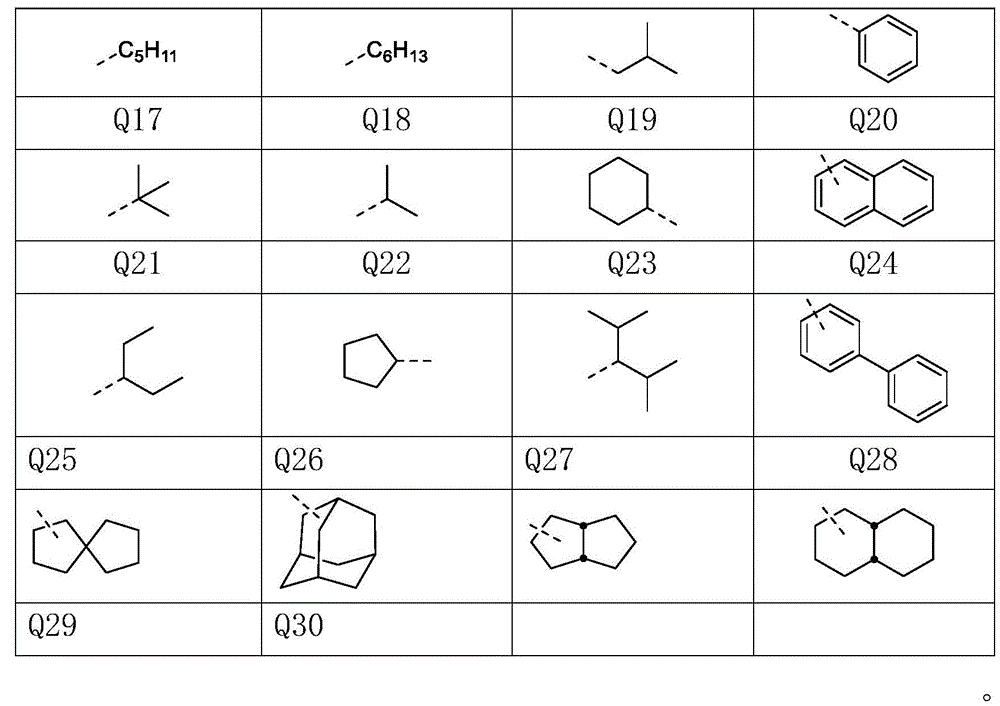
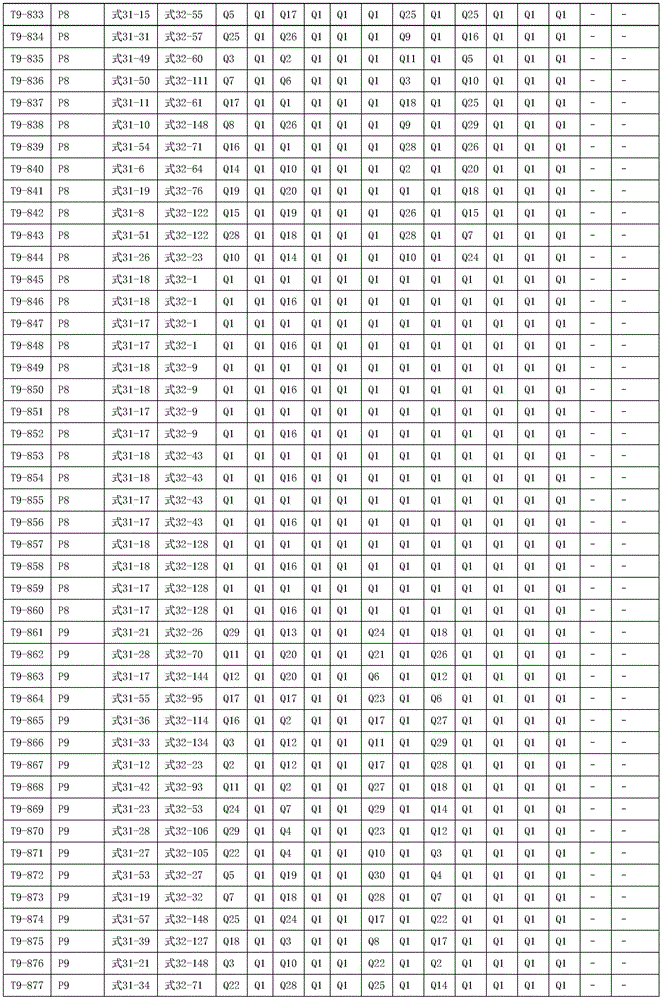
其中,R1、R2、R3、R4、R5、R6、R7、R8、R9、R10、R11、R12、R13、R14互相独立的选自以下取代基Q1至Q30结构的任一种:Wherein, R1, R2, R3, R4, R5, R6, R7, R8, R9, R10, R11, R12, R13, R14 are independently selected from any of the following substituent Q1 to Q30 structures:
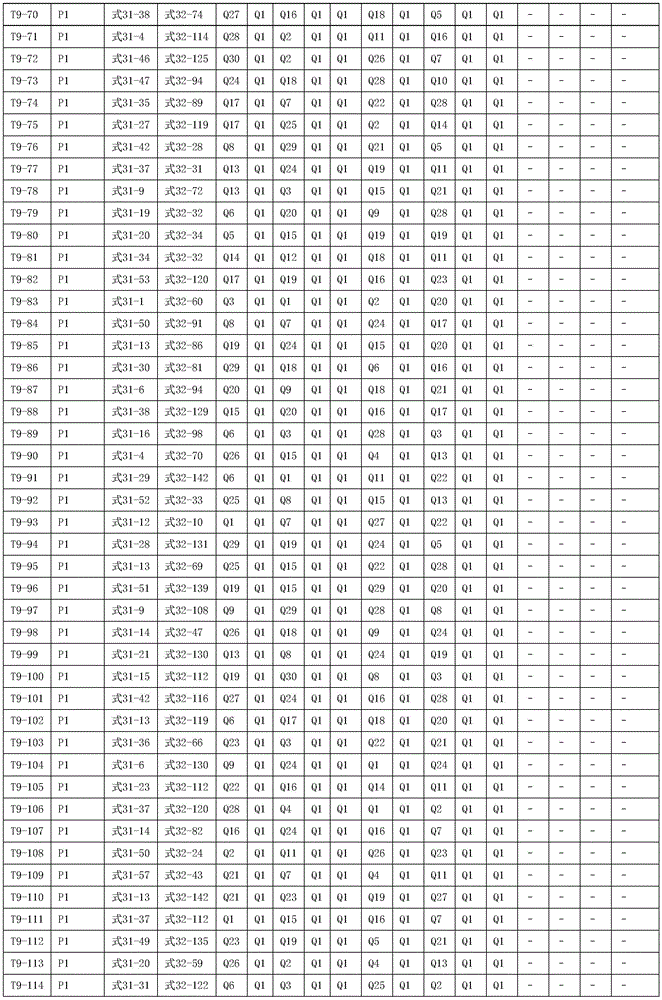
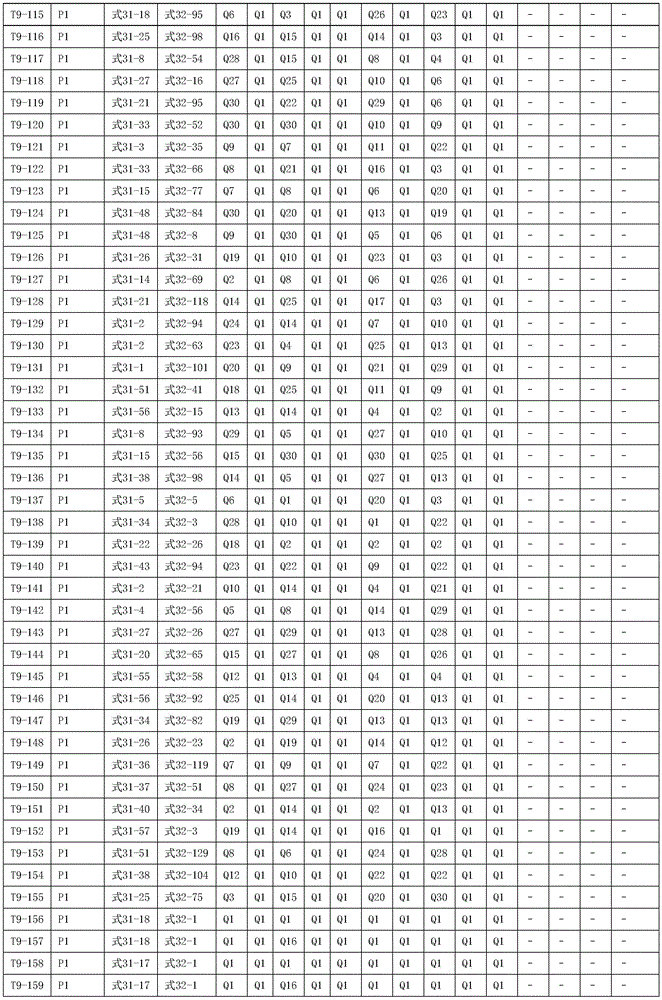
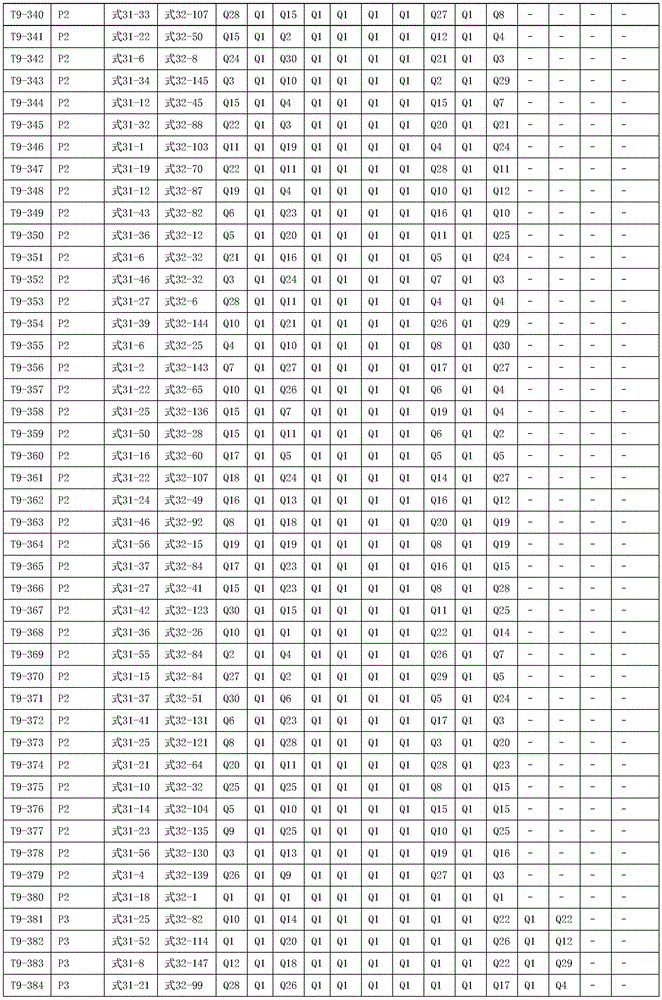
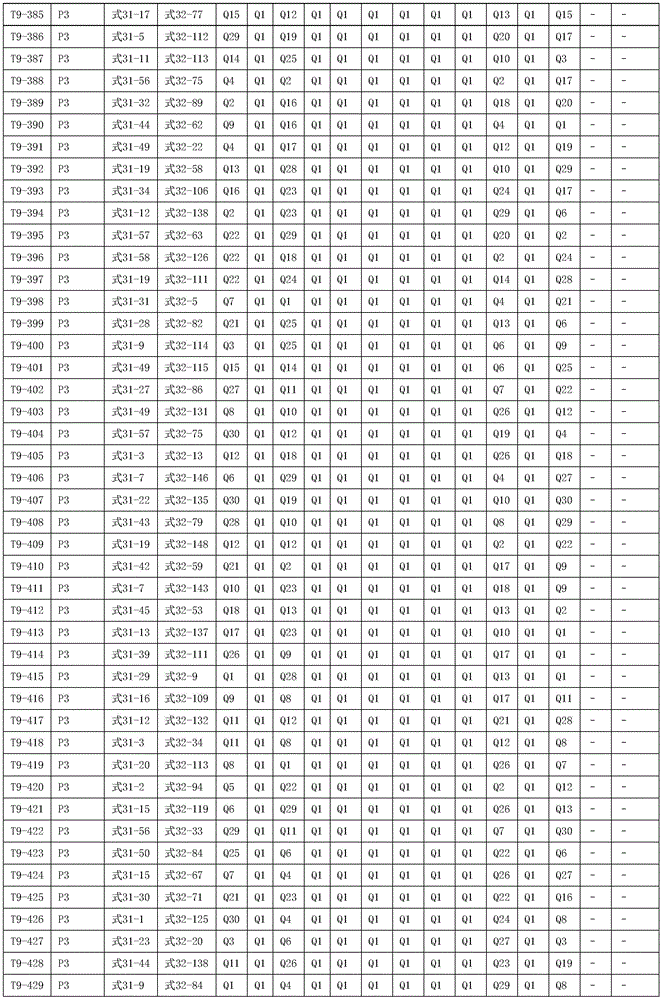
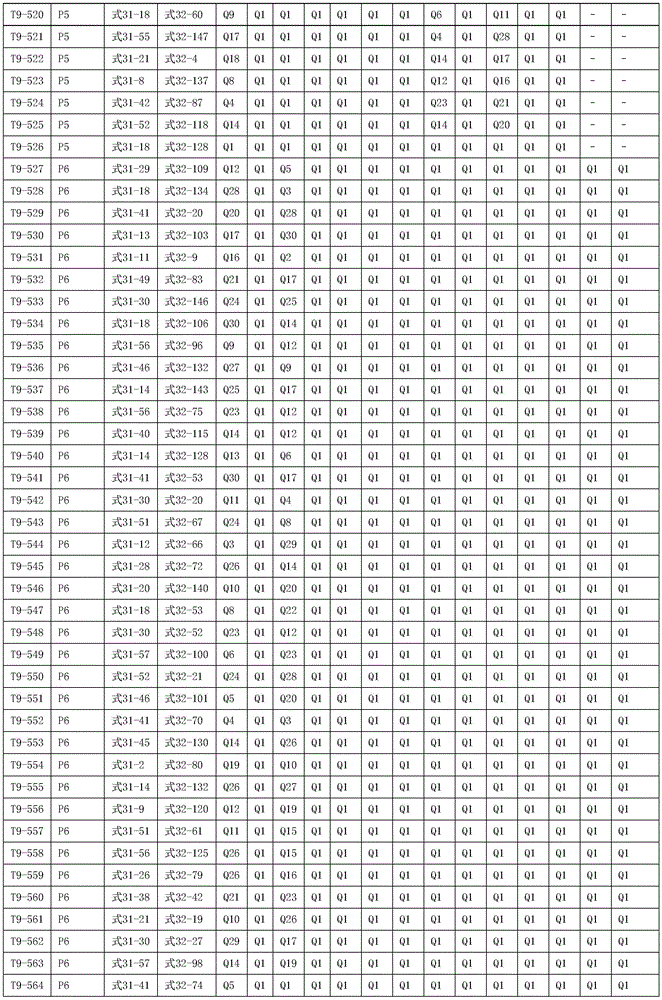
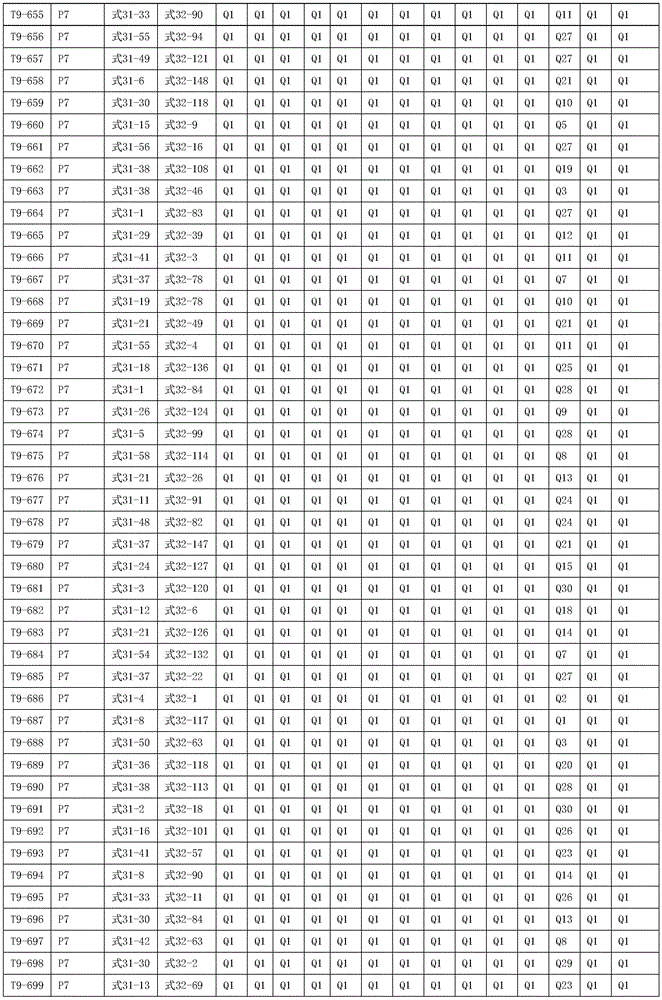
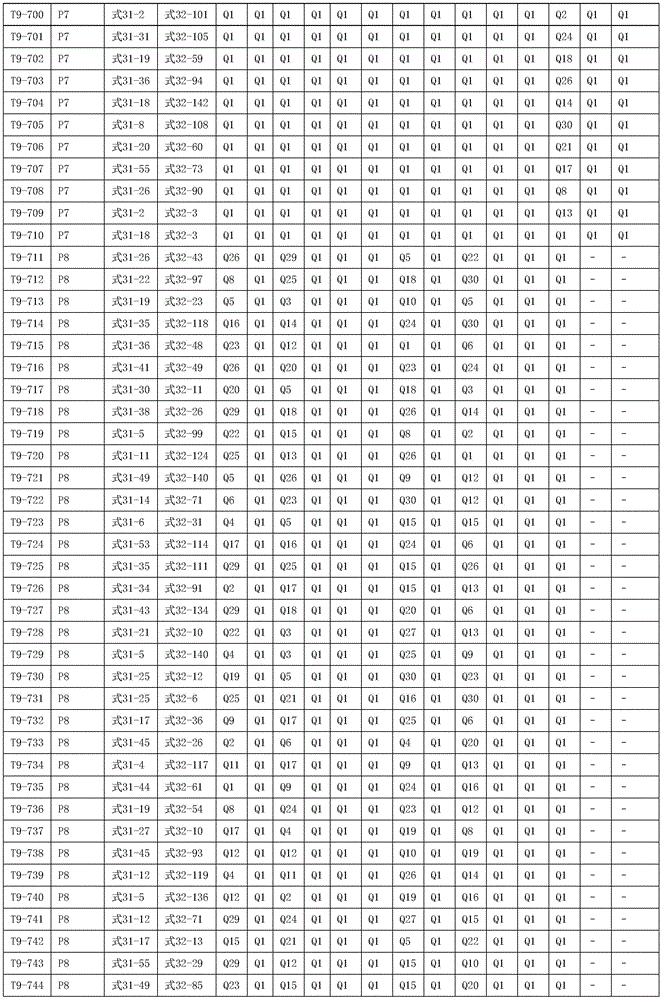
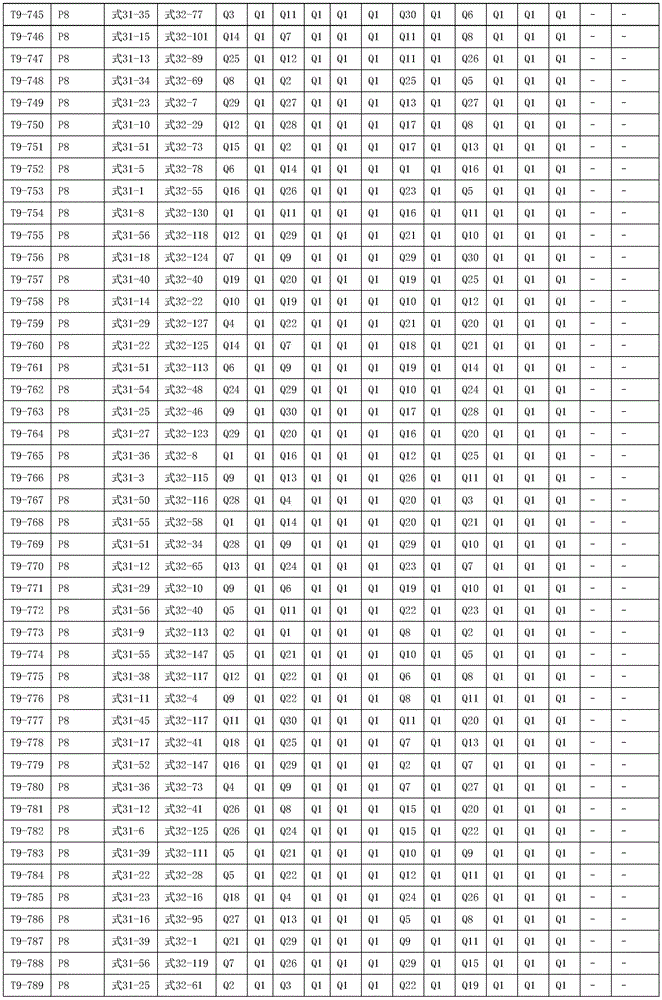
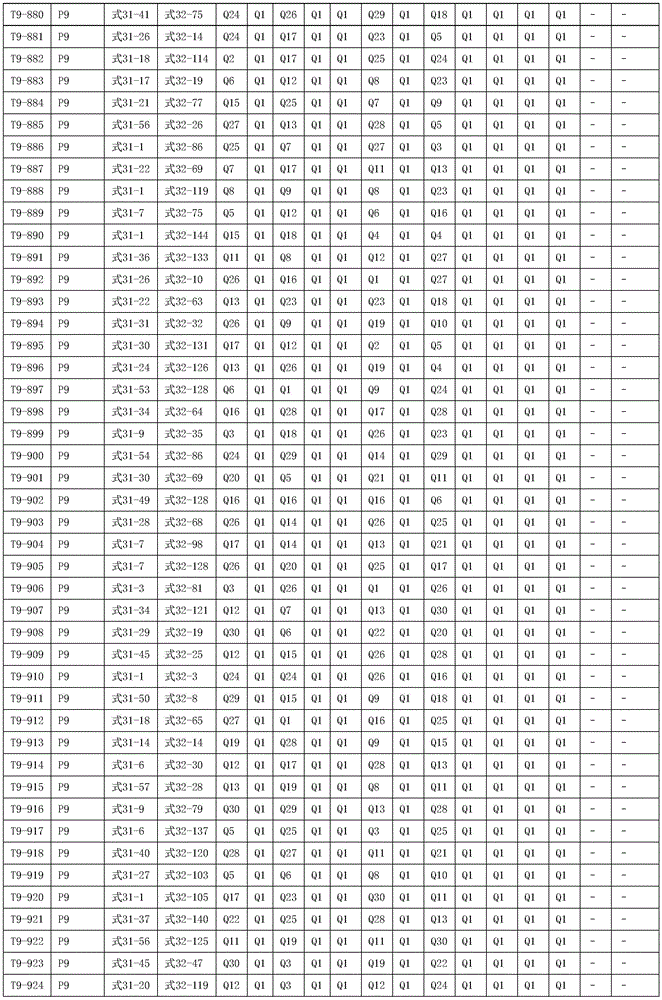
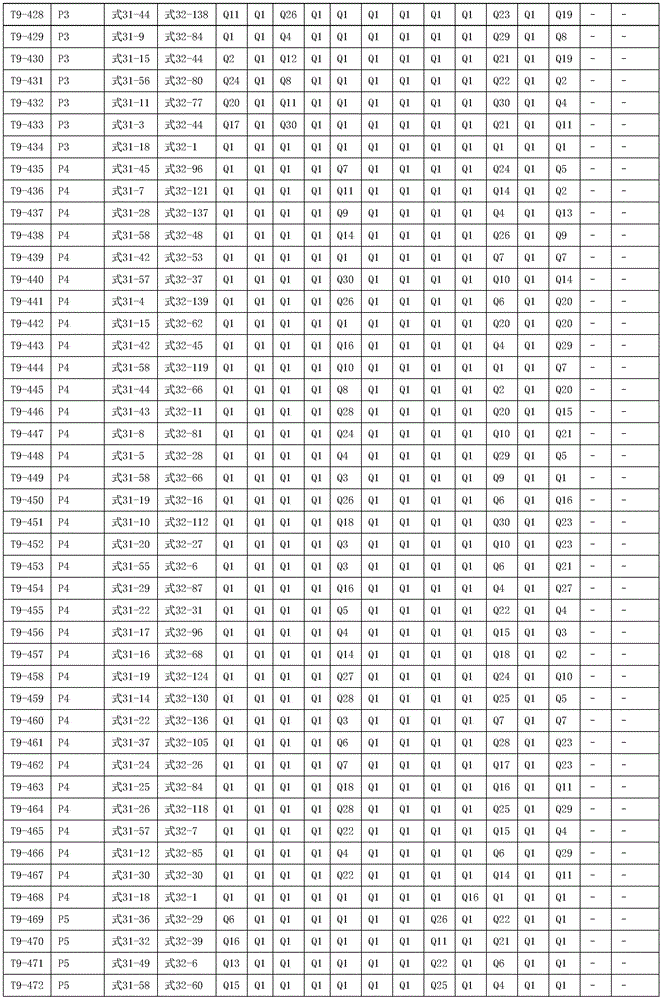
进一步地,所述氨基环状化合物的结构由下表的组合构成:Further, the structure of the amino cyclic compound is formed by the combination of the following table:
本发明的另一个目的是提供一种具有高抗三线态解离能的寿命长的发光元件,以及,提供一种具有高抗三线态解离能、且发光效率良好的发光元件,以及,提供一种驱动电压低的发光元件。Another object of the present invention is to provide a light-emitting element with high resistance to triplet dissociation energy and long life, and to provide a light-emitting element with high resistance to triplet dissociation energy and good luminous efficiency, and to provide A light-emitting element with a low driving voltage.
本发明的又一个目的是分别提供一种可靠性高的发光元件、发光装置及电子设备,以及,分别提供一种低功耗的发光元件、发光装置及电子设备。Yet another object of the present invention is to provide a light emitting element, a light emitting device, and an electronic device with high reliability, respectively, and to provide a light emitting element, a light emitting device, and an electronic device with low power consumption.
根据本发明的适合有机半导体优选可以为空穴传输材料(HTM)和/或空穴注入材料(HIM)。空穴注入材料简化或促进空穴(即正电荷)从阳极转移到有机层中。空穴传输材料能够传输空穴,即正电荷,其通常从阳极或相邻层(例如空穴注入层)注入。Suitable organic semiconductors according to the invention may preferably be hole-transport materials (HTM) and/or hole-injection materials (HIM). The hole injection material simplifies or facilitates the transfer of holes (ie, positive charges) from the anode into the organic layer. Hole transport materials are capable of transporting holes, ie positive charges, which are typically injected from the anode or an adjacent layer such as a hole injection layer.
常见优选的空穴注入材料和/或空穴传输材料包括例如三芳基胺、联苯胺、四芳基-对苯二胺、三芳基膦、吩噻嗪、噻吩、吡咯和呋喃衍生物以及其它具有高HOMO(HOMO=最高已占分子轨道)的含O、S或N的杂环。在材料结构中引入种富电子的空穴传输基团hole-transporting moieties,具有较好的传空穴能力,能够改善材料整体的HOMO和LUMO能级,具有更好的空穴传输效率或电子阻挡能力。Commonly preferred hole injection and/or hole transport materials include, for example, triarylamines, benzidines, tetraaryl-p-phenylenediamines, triarylphosphines, phenothiazines, thiophenes, pyrrole and furan derivatives and others with High HOMO (HOMO=highest occupied molecular orbital) O-, S- or N-containing heterocycle. Introduce a kind of electron-rich hole-transporting moieties into the material structure, which has better hole-transporting ability, can improve the overall HOMO and LUMO energy levels of the material, and has better hole-transporting efficiency or electron blocking ability.
根据本发明,我们惊讶的发现,通过我们设计的主体结构作为核心,连接不同的取代基后,能兼具三线态稳体和空穴传输基团的功能。将所得到的化合物用于有机电致发光器件后,导致其在效率、工作电压、寿命、颜色坐标和/或色纯度即发射带的宽度方面展现出多种改进特性。此外,所述化合物还具有更好的溶解性能,可以以非常简单的方式加工,具有比现有材料更好的热稳定性。同时由于具有较低的分子量,该化合物具有更低的升华温度,更易于大规模生产。According to the present invention, we are surprised to find that the main structure we designed is used as the core, and after connecting different substituents, it can have both the functions of a triplet stabilizer and a hole-transporting group. Use of the obtained compounds in organic electroluminescent devices results in them exhibiting various improved properties in terms of efficiency, operating voltage, lifetime, color coordinates and/or color purity, ie width of the emission band. In addition, the compounds have better solubility properties, can be processed in a very simple manner and have better thermal stability than existing materials. At the same time, due to the lower molecular weight, the compound has a lower sublimation temperature and is easier to produce on a large scale.
附图说明Description of drawings
图1为本发明有机致电发光装置的底部发光第一实施例示意图。FIG. 1 is a schematic diagram of a first embodiment of bottom emission of an organic electroluminescence device of the present invention.
图2为本发明有机致电发光装置的顶部发光第二实施例示意图Fig. 2 is a schematic diagram of the second embodiment of the top emission of the organic electroluminescence device of the present invention
附图编号说明:1-基板、2-第一电极、3-空穴注入、41-空穴传输、42-电子阻挡层、5-发光层、6-电子传输层、7-电子注入层、8-第二电极、9-光提取层。Description of the accompanying drawings: 1-substrate, 2-first electrode, 3-hole injection, 41-hole transport, 42-electron blocking layer, 5-light-emitting layer, 6-electron transport layer, 7-electron injection layer, 8 - second electrode, 9 - light extraction layer.
具体实施方式Detailed ways
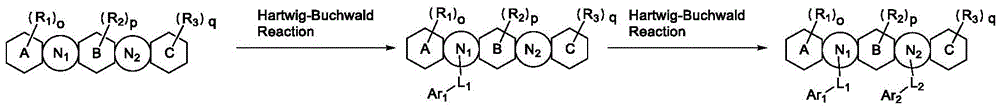
为了更清楚的理解本发明的内容,将结合实施例详细解释所述有机电致发光化合物、所述化合物的制备方法和所述装置的发光特性。各种化学反应可以应用于本发明一个实施方式的化合物的合成方法。通过中心化合物SM-a通过两步Hartwig-Buchwald偶联得到最终目标化合物。但是应当注意,本发明的一个实施方式的化合物的合成方法不限于以下描述的合成方法。除非另有说明,否则随后的合成是在保护性气体气氛下在无水溶剂中进行。溶剂和试剂可以从常规试剂供应商购买。下面展示目标化合物的合成通式:In order to understand the content of the present invention more clearly, the organic electroluminescent compound, the preparation method of the compound and the light-emitting characteristics of the device will be explained in detail with reference to examples. Various chemical reactions can be applied to the synthesis method of the compound of one embodiment of the present invention. The final target compound was obtained by two-step Hartwig-Buchwald coupling through the central compound SM-a. It should be noted, however, that the synthesis method of the compound of one embodiment of the present invention is not limited to the synthesis method described below. Subsequent syntheses were carried out in anhydrous solvents under a protective gas atmosphere unless otherwise stated. Solvents and reagents can be purchased from conventional reagent suppliers. The synthetic formula of the target compound is shown below:
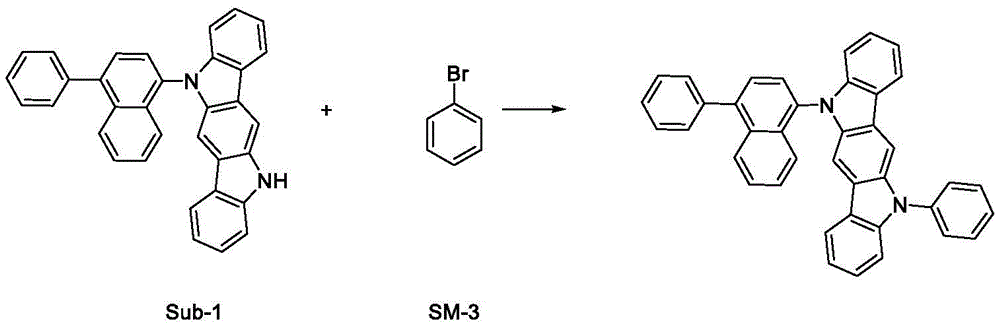
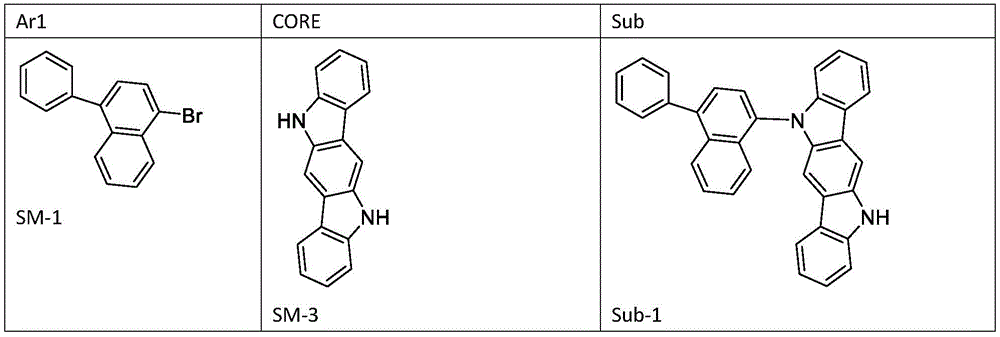
实施例1制备化合物
首先将SM-1(25.38g;90mmol)、SM-2(25.60g;100.0mmol)和叔丁醇钠(10.1g;105mmol)加入甲苯(200mL)中。之后,引入双二亚苄基丙酮钯(57.5mg;0.1mmol)和三叔丁基膦(40.4mg;0.2mmol),氮气氛围下,将反应混合物在回流下搅拌10小时。冷却混合物后,将其用1mol/L的稀盐酸调节至pH 7,并且将有机相在分液漏斗中分离出来。随后,将有机相通过柱层析硅胶以二氯甲烷溶液的形式过滤。将硅胶用二氯甲烷洗涤两次(每次100ml),滤液经Na2SO4干燥,然后浓缩至干燥。粗产物从乙酸乙酯中重结晶。得到产品:28.85g(63mmol);纯度>98%。SM-1 (25.38 g; 90 mmol), SM-2 (25.60 g; 100.0 mmol) and sodium tert-butoxide (10.1 g; 105 mmol) were first added to toluene (200 mL). Afterwards, palladium bisdibenzylideneacetone (57.5 mg; 0.1 mmol) and tri-tert-butylphosphine (40.4 mg; 0.2 mmol) were introduced, and the reaction mixture was stirred at reflux for 10 hours under nitrogen atmosphere. After cooling the mixture, it was adjusted to
将Sub-1(28.85g;63mmol)、SM-3(9.8g;63mmol)和叔丁醇钠(6.05g;63mmol)加入甲苯(200mL)中。之后,引入双二亚苄基丙酮钯(36.22mg;0.063mmol)和三叔丁基膦(25.45mg;0.126mmol),将反应混合物在回流下搅拌10小时。冷却混合物后,将其用1mol/L的稀盐酸调节至pH 7,并且将有机相在分液漏斗中分离出来。随后,将有机相通过柱层析硅胶以乙酸乙酯浆液的形式过滤。将硅胶用乙酸乙酯洗涤两次(每次100ml),滤液经Na2SO4干燥,然后浓缩至干燥。粗产物从乙酸乙酯中重结晶。得到产品:25.3g(47.37mmol);纯度>99%。Sub-1 (28.85 g; 63 mmol), SM-3 (9.8 g; 63 mmol) and sodium tert-butoxide (6.05 g; 63 mmol) were added to toluene (200 mL). Afterwards, palladium bisdibenzylideneacetone (36.22 mg; 0.063 mmol) and tri-tert-butylphosphine (25.45 mg; 0.126 mmol) were introduced, and the reaction mixture was stirred at reflux for 10 hours. After cooling the mixture, it was adjusted to
实施例2Example 2
首先将SM-2(25.38g;90mmol)、SM-5(30.60g;100.0mmol)和叔丁醇钠(10.1g;105mmol)加入甲苯(200mL)中。之后,引入双二亚苄基丙酮钯(57.5mg;0.1mmol)和三叔丁基膦(40.4mg;0.2mmol),将反应混合物在回流下搅拌10小时。冷却混合物后,将其用1mol/L的稀盐酸调节至pH 7,并且将有机相在分液漏斗中分离出来。随后,将有机相通过柱层析硅胶以甲苯浆液的形式过滤。将硅胶用甲苯洗涤两次(每次100ml),滤液经Na2SO4干燥,然后浓缩至干燥。粗产物从乙酸乙酯中重结晶。得到产品:35.56g(70.3mmol);纯度>98%。SM-2 (25.38 g; 90 mmol), SM-5 (30.60 g; 100.0 mmol) and sodium tert-butoxide (10.1 g; 105 mmol) were first added to toluene (200 mL). Afterwards, palladium bisdibenzylideneacetone (57.5 mg; 0.1 mmol) and tri-tert-butylphosphine (40.4 mg; 0.2 mmol) were introduced, and the reaction mixture was stirred at reflux for 10 hours. After cooling the mixture, it was adjusted to
首先将Sub-7(35.56g;70.3mmol)、SM-5(17.29g;70.3mmol)和叔丁醇钠(7.0g;75.0mmol)加入甲苯(300mL)中。之后,引入双二亚苄基丙酮钯(40.25mg;0.07mmol)和三叔丁基膦(28.28mg;0.14mmol),将反应混合物在回流下搅拌10小时。冷却混合物后,将其用1mol/L的稀盐酸调节至pH 7,并且将有机相在分液漏斗中分离出来。随后,将有机相通过柱层析硅胶以乙酸乙酯浆液的形式过滤。将硅胶用乙酸乙酯洗涤两次(每次150ml),滤液经Na2SO4干燥,然后浓缩至干燥。粗产物从石油醚中重结晶。得到产品:33.70g(49.8mmol);纯度>99%。Sub-7 (35.56 g; 70.3 mmol), SM-5 (17.29 g; 70.3 mmol) and sodium tert-butoxide (7.0 g; 75.0 mmol) were first added to toluene (300 mL). Afterwards, palladium bisdibenzylideneacetone (40.25 mg; 0.07 mmol) and tri-tert-butylphosphine (28.28 mg; 0.14 mmol) were introduced, and the reaction mixture was stirred at reflux for 10 hours. After cooling the mixture, it was adjusted to
随后的化合物可以类似的方式制备。在这种情况下,也可以使用柱色谱法纯化;或使用其它标准溶剂,诸如乙醇、丁醇、丙酮、乙酸乙酯、乙腈、甲苯、二甲苯、二氯甲烷、甲醇、四氢呋喃、乙酸正丁酯、1,4-二烷重结晶或热萃取进行纯化;或使用高沸点物质,诸如二甲亚砜、N,N-二甲基甲酰胺、N,N-二甲基乙酰胺、N-甲基吡咯烷酮等重结晶进行纯化。产率通常在40%与90%之间的范围内。Subsequent compounds can be prepared in an analogous manner. In this case, column chromatography can also be used; or other standard solvents such as ethanol, butanol, acetone, ethyl acetate, acetonitrile, toluene, xylene, dichloromethane, methanol, tetrahydrofuran, n-butyl acetate ester, 1,4-dioxane recrystallization or thermal extraction; or use high boiling point substances such as dimethylsulfoxide, N,N-dimethylformamide, N,N-dimethylacetamide, N- Purify by recrystallization such as methylpyrrolidone. Yields typically range between 40% and 90%.
中间体Sub合成:Synthesis of intermediate Sub:
目标化合物合成:Synthesis of the target compound:
比较例1Comparative example 1
采用对比化合物1和对比化合物2作为本发明的对比例。
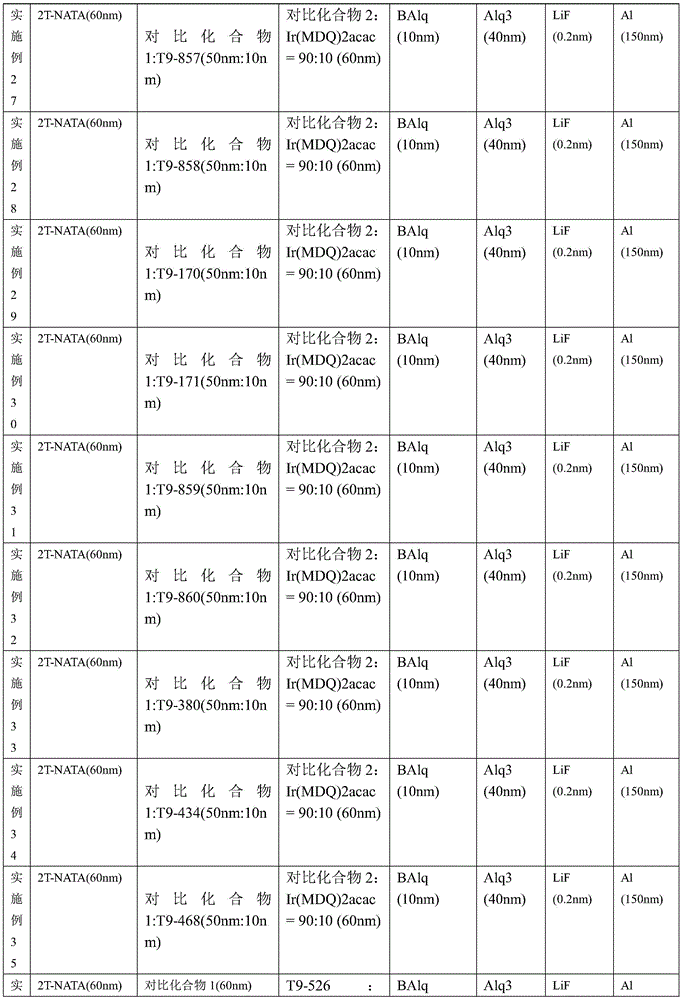
装置实施例1
根据本发明的三线态稳定基团的特定排列的氨基环状化合物制造OLED装置。The specific arrangement of the amino cyclic compound of the triplet stabilizing group according to the present invention makes an OLED device.
如图1、图2所示,有机电致发光装置具有基板1、阳极2、阴极8,以及配置于阳极2与阴极8之间的层3-7。其中,在阴极8与发光层5之间配置有电子传输层6和电子注入层7,在发光层5与阳极2之间配置有空穴注入层3和空穴传输/发光辅助层4。如图2所示为本发明有机致电发光装置的第二实施例示意图,对应顶部发光的排布方式。结构上在第一实施例底部发光排布方式的基础上,在顶层额外还设置有光提取层9。As shown in FIG. 1 and FIG. 2 , an organic electroluminescence device has a
OLED的制造和测试表征OLED Fabrication and Test Characterization
器件对比例Device Comparison
所述有机发光器件,包括依次设置在衬底基板上的阳极、有机材料功能层、阴极、封装层;所述有机材料功能层中,在所述阳极上形成的空穴注入层、在所述空穴注入层上形成的空穴传输层,在所述空穴传输层上形成的发光层、在所述发光层上形成的电子传输层,在所述电子传输层上形成的阴极。The organic light-emitting device includes an anode, an organic material functional layer, a cathode, and an encapsulation layer sequentially arranged on a substrate; in the organic material functional layer, the hole injection layer formed on the anode, the A hole transport layer formed on the hole injection layer, a light emitting layer formed on the hole transport layer, an electron transport layer formed on the light emitting layer, a cathode formed on the electron transport layer.
所述空穴传输层可以为一层或多层,所述发光层为主体和掺杂客体组成,发光层主体可以为一种分子材料组成或多种分子材料组成。The hole transport layer can be one or more layers, the light-emitting layer is composed of a host and a doped guest, and the host of the light-emitting layer can be composed of one molecular material or multiple molecular materials.
本发明所述的分子结构可以用于上述有机电致发光器件的一层或多层。更加优选的用于器件的空穴注入层,空穴传输层,发光辅助层或主体材料。The molecular structure described in the present invention can be used in one or more layers of the above-mentioned organic electroluminescence device. More preferred hole injection layer, hole transport layer, luminescence assisting layer or host material for devices.
实施例中的阳极采用本领域内常用的阳极材料,如ITO、Ag或其多层结构。空穴注入单元采用本领域内常用的空穴注入材料,如F4TCNQ、HATCN、NDP-9等或其掺杂。空穴传输单元采用本领域内常用的空穴传输材料,如PBPBA、TCTA等。发光单元采用本领域内常用的发光材料,例如可以由主体材料和发射的客体材料掺杂构成,发射的客体材料可以为有机材料也可以为金属配合物(如金属Ir,Pt等)。电子注入单元采用本领域内常用的电子注入材料,如Liq、LiF、Yb等。阴极采用本领域内常用材料,如金属Al、Ag或金属混合物(Ag掺杂的Mg、Ag掺杂的Ca等)。The anode in the embodiment adopts the anode material commonly used in this field, such as ITO, Ag or its multilayer structure. The hole injection unit adopts hole injection materials commonly used in this field, such as F4TCNQ, HATCN, NDP-9, etc. or doping thereof. The hole transport unit adopts hole transport materials commonly used in this field, such as PBPBA, TCTA and the like. The light-emitting unit adopts commonly used light-emitting materials in the field, for example, it can be composed of a host material and an emitting guest material, and the emitting guest material can be an organic material or a metal complex (such as metal Ir, Pt, etc.). The electron injection unit adopts electron injection materials commonly used in this field, such as Liq, LiF, Yb and the like. The cathode adopts common materials in this field, such as metal Al, Ag or metal mixture (Ag-doped Mg, Ag-doped Ca, etc.).
本实施方式中的电极制备方法和各功能层的沉积方法均为本领域常规方法,例如真空热蒸镀或喷墨打印等,在此不再赘述,仅对制备过程中的一些工艺细节、测试方法补充说明如下:本发明的化合物使用空穴输送层物质并采用通常的方法来制作有机电致发光元件。首先,在形成于有机基板的ITO层(阳极)上以60nm的厚度真空沉积N1-(naphthalen-2-yl)-N4,N4-bis(4-(naphthalen-2-yl(phenyl)amino)phenyl)-N1-phenylbenzene-1,4-diamine(以下,简称为“2-TNATA”)来形成空穴注入层后,在上述空穴注入层上以60nm的厚度真空沉积对比化合物1来形成空穴输送层或过渡层。接着,在上述空穴输送层或过渡层上,以30nm的厚度真空沉积对比化合物2作为主体并以tris(2-phenylpyridine)-iridium(以下,简称为“Ir(ppy)3”)或Bis(2-methyldibenzo[f,h]quinoxaline)(acetylacetonate)iridium(以下,简称为“Ir(MDQ)2acac”)作为掺杂剂,将以90:10重量掺杂的混合物以30nm的厚度真空沉积来形成发光层。接着,在上述发光层上以10nm的厚度真空沉积((1,1’-bisphenyl)-4-olato)bis(2-methyl-8-quinolinolato)铝(以下,简称为“BAlq”)来形成空穴阻挡层,在上述空穴阻挡层上以40nm的厚度真空沉积三(8-喹啉醇)铝(以下,简称为“Alq 3”)来形成电子输送层。然后,以0.2nm的厚度沉积作为卤化碱金属的LiF来形成电子注入层,接着,以150nm的厚度沉积铝(Al)来形成阴极,制备出有机电致发光元件。The electrode preparation method and the deposition method of each functional layer in this embodiment are conventional methods in the field, such as vacuum thermal evaporation or inkjet printing, etc., and will not be repeated here, only some process details and test methods in the preparation process The supplementary description of the method is as follows: the compound of the present invention uses the substance of the hole transport layer and adopts the usual method to produce an organic electroluminescent device. First, N1-(naphthalen-2-yl)-N4, N4-bis(4-(naphthalen-2-yl(phenyl)amino)phenyl )-N1-phenylbenzene-1,4-diamine (hereinafter referred to as "2-TNATA") to form a hole injection layer, and then vacuum-
采用上述方法将实施例所述的化合物制成有机电致发光器件。The compound described in the example was prepared into an organic electroluminescent device by the above method.
作为空穴传输层或发光辅助层使用Use as hole transport layer or luminescence auxiliary layer
除了在实施例中使用发明的化合物来代替对比化合物1作空穴输送层或之外,其他材料以与上述对比例相同的方法制成有机电致发光器件。Except that the inventive compound was used instead of the
也可以除了将实施例中发明的化合物真空沉积至对比化合物1和主体材料之间,部分代替对比化合物1,作空穴输送层或发光辅助层之外,其他材料以与上述对比例相同的方法制成有机电致发光器件。It is also possible to vacuum-deposit the compound invented in the example between the
作为主体材料使用Used as the main material
除了在实施例中使用发明的化合物来代替对比化合物2作为主体材料之外,以与上述对比例相同的方法制成有机电致发光器件。An organic electroluminescent device was fabricated in the same manner as in the comparative example above, except that the inventive compound was used instead of the
将各种OLED的器件结构总结在表1中。实施例与现有技术对比例1和对比例2的各项器件结构进行对比。The device structures of various OLEDs are summarized in Table 1. The embodiment is compared with the device structures of Comparative Example 1 and Comparative Example 2 in the prior art.
表1对比例和实施例器件结构Table 1 comparative example and embodiment device structure
通过标准方法测试所述OLED。为此,在J=10mA/cm2的电流密度下确定所述电致发光器件的驱动电压,亮度,电致发光电流效率(以cd/A计量)和外量子效率(EQE,以百分比计量),其作为发光密度的函数从呈现郎伯发射特性的电流/电压/发光密度特性线(IVL特性线)进行计算,发光光谱,并由此计算CIE1931x和y色坐标。将寿命LT定义为如下的时间,在恒定电流J下工作时,亮度在该时间后从初始发光亮度L0降至特定的比例L1;J=50mA/cm2和L1=90%的表述是指在50mA/cm2下工作时,发光亮度在时间LT之后降至其初始值L0的90%,类似地,J=20mA/cm2,L1=80%是指,在20mA/cm2下工作时,发光亮度在时间LT之后降至其初始值L0的80%。The OLEDs were tested by standard methods. For this reason, the driving voltage, brightness, electroluminescent current efficiency (measured in cd/A) and external quantum efficiency (EQE, measured in percentage) of the electroluminescent device were determined at a current density of J=10mA/cm , which is calculated as a function of luminous density from the current/voltage/luminous density characteristic line (IVL characteristic line) exhibiting Lambertian emission characteristics, the luminous spectrum, and the CIE1931x and y color coordinates are calculated therefrom. The lifetime LT is defined as the time after which, when operating at a constant current J, the luminance drops from the initial luminous luminance L 0 to a certain ratio L 1 ; the expression J=50mA/cm 2 and L 1 =90% It means that when working at 50mA/cm 2 , the luminous brightness drops to 90% of its initial value L 0 after time LT, similarly, J=20mA/cm 2 , L 1 =80% means that at 20mA/cm 2 , the luminous brightness drops to 80% of its initial value L0 after time LT.
将各种OLED的数据总结在表2中。实施例与现有技术对比例1和对比例2的各项参数进行对比,展示了各种OLED的数据。The data for various OLEDs are summarized in Table 2. The embodiment is compared with the parameters of the prior art Comparative Example 1 and Comparative Example 2, and the data of various OLEDs are shown.
所述实施例对OLED材料及元件进行性能测试的测试仪器及方法如下:Described embodiment carries out the test instrument and the method that OLED material and element are tested for performance as follows:
OLED元件性能检测条件:OLED component performance testing conditions:
亮度和色度坐标:使用光谱扫描仪PhotoResearch PR-635测试;Luminance and chromaticity coordinates: Tested using spectral scanner PhotoResearch PR-635;
电流密度和起亮电压:使用数字源表Keithley 2400测试;Current density and lighting voltage: Tested with a digital source meter Keithley 2400;
寿命测试:使用寿命测试装置。Life test: life test device.
所得元件的性能检测结果列于表2中。The performance test results of the obtained components are listed in Table 2.
表2元件性能测试结果Table 2 Component performance test results
由表2的元件性能测试结果可知,本发明的氨基环状化合物可以用来制备有机材料,采用该有机材料制备成的器件发光效率和寿命相较现有技术对比例1和对比例2具有明显提高,该器件在50mA/cm2下工作时,发光亮度降至其初始值L0的95%,时间提高,使用本发明的化合物作为绿光或红光空穴传输层,发光辅助层的器件寿命衰减大大减缓。As can be seen from the component performance test results in Table 2, the aminocyclic compound of the present invention can be used to prepare organic materials, and the luminous efficiency and service life of the devices prepared by using the organic materials have obvious advantages compared with the prior art Comparative Example 1 and Comparative Example 2. Improve, when the device works at 50mA/cm , the luminous brightness drops to 95% of its initial value L0 , the time improves, and the compound of the present invention is used as the green light or red light hole transport layer, the device of the luminescence auxiliary layer Lifespan decay greatly slowed.
以上所述仅为本发明较佳的具体实施方式,但本发明的保护范围并不局限于此,任何熟悉本技术领域的技术人员在本发明揭露的技术范围内,可轻易想到的变化或替换等都应涵盖在本发明的保护范围之内。因此,本发明的保护范围应该以权利要求书的保护范围为准。The above description is only a preferred embodiment of the present invention, but the scope of protection of the present invention is not limited thereto, any changes or substitutions that can be easily conceived by those skilled in the art within the technical scope disclosed in the present invention etc. should be covered within the protection scope of the present invention. Therefore, the protection scope of the present invention should be determined by the protection scope of the claims.
Claims (9)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110530783.4A CN115340551A (en) | 2021-05-15 | 2021-05-15 | Amino cyclic compound, organic electroluminescent material, and organic electroluminescent device |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110530783.4A CN115340551A (en) | 2021-05-15 | 2021-05-15 | Amino cyclic compound, organic electroluminescent material, and organic electroluminescent device |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN115340551A true CN115340551A (en) | 2022-11-15 |
Family
ID=83946605
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202110530783.4A Withdrawn CN115340551A (en) | 2021-05-15 | 2021-05-15 | Amino cyclic compound, organic electroluminescent material, and organic electroluminescent device |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN115340551A (en) |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20160013430A1 (en) * | 2014-07-10 | 2016-01-14 | Samsung Display Co., Ltd. | Organic light-emitting device |
| CN105810837A (en) * | 2015-01-16 | 2016-07-27 | 三星显示有限公司 | Organic light-emitting device |
| CN106233489A (en) * | 2014-04-29 | 2016-12-14 | 罗门哈斯电子材料韩国有限公司 | Multicomponent material of main part and the Organnic electroluminescent device comprising it |
| CN112174944A (en) * | 2020-09-25 | 2021-01-05 | 江苏三月科技股份有限公司 | Compound with dibenzo five-membered heterocycle as core and application thereof |
| WO2021066623A1 (en) * | 2019-10-01 | 2021-04-08 | 주식회사 엘지화학 | Organic light emitting device |
-
2021
- 2021-05-15 CN CN202110530783.4A patent/CN115340551A/en not_active Withdrawn
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN106233489A (en) * | 2014-04-29 | 2016-12-14 | 罗门哈斯电子材料韩国有限公司 | Multicomponent material of main part and the Organnic electroluminescent device comprising it |
| US20160013430A1 (en) * | 2014-07-10 | 2016-01-14 | Samsung Display Co., Ltd. | Organic light-emitting device |
| CN105810837A (en) * | 2015-01-16 | 2016-07-27 | 三星显示有限公司 | Organic light-emitting device |
| WO2021066623A1 (en) * | 2019-10-01 | 2021-04-08 | 주식회사 엘지화학 | Organic light emitting device |
| CN112174944A (en) * | 2020-09-25 | 2021-01-05 | 江苏三月科技股份有限公司 | Compound with dibenzo five-membered heterocycle as core and application thereof |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR102829041B1 (en) | Compound for organic electronic element, organic electronic element using the same, and an electronic device thereof | |
| KR102366722B1 (en) | Compound for organic electronic element, organic electronic element comprising the same, and electronic device thereof | |
| KR101628438B1 (en) | NITROGEN-CONTAINING HETEROCYCLIC COMPOUNDS AND ORGANIC ElECTRONIC DEVICE COMPRISING THE SAME | |
| CN114716330A (en) | A kind of luminescent auxiliary material, its preparation method and application | |
| KR102657636B1 (en) | Anthracene compound, coating composition and organic light emitting device comprising same | |
| KR20220006313A (en) | Anthracene compound, coating composition and organic light emitting device comprising same | |
| CN110357882B (en) | Organic compound containing nitrogen heterocycle and organic electroluminescent device | |
| CN114805318B (en) | Organic compound with triazine derivative as core and application thereof | |
| CN115947706B (en) | A luminescence auxiliary material and its preparation method and application | |
| CN115124504A (en) | A light-emitting auxiliary material, preparation method and application thereof, and organic electroluminescent device comprising the same | |
| CN112500435B (en) | A light-emitting compound containing a dibenzo-seven-membered heterocyclic structure, a preparation method thereof, and an organic electroluminescent device | |
| CN115181074A (en) | Luminous auxiliary material, preparation method thereof and luminous device | |
| CN118812541B (en) | An organic electroluminescent material and an organic electroluminescent device containing dual host materials | |
| CN115340550A (en) | Amino cyclic compound, organic electroluminescent material, and organic electroluminescent device | |
| KR101098789B1 (en) | Anthracene chemiclal and organic electroric element using the same, terminal thererof | |
| KR20100111982A (en) | Cyclic aromatic compound and organic electronic element using the same, terminal thererof | |
| CN115340548A (en) | Amine-containing compound, organic electroluminescent material and organic electroluminescent device | |
| CN116354867A (en) | Organic compound, organic electroluminescent device, organic electroluminescent device | |
| CN115340546A (en) | Aminocyclic compound, organic electroluminescent device, organic electroluminescent device | |
| CN115340551A (en) | Amino cyclic compound, organic electroluminescent material, and organic electroluminescent device | |
| CN114907352A (en) | A kind of carbazole derivative and its application | |
| CN115340490A (en) | Aminocyclic compound, organic electroluminescent device, organic electroluminescent device | |
| CN115093335B (en) | A luminescent auxiliary material and its preparation method and application | |
| KR102696222B1 (en) | Novel heterocyclic compound and organic light emitting device comprising the same | |
| KR20210062496A (en) | Compound for organic optoelectronic and organic optoelectronic device including the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| WW01 | Invention patent application withdrawn after publication | ||
| WW01 | Invention patent application withdrawn after publication |
Application publication date: 20221115 |