Background
The implanted or external medical devices in the forms of a plurality of catheters, film dressings, non-woven fabrics and the like are mainly prepared from high polymer materials and play an important role in the medical and health fields. At present, the medical polymer materials have three common problems: firstly, general medical polymer materials do not have antibacterial performance, and pathogenic microorganisms such as bacteria and the like are infected in the application process or are easy to cause serious infection inflammatory reaction; secondly, in order to cope with pathogenic infection caused by medical high polymer materials, antibiotics or antibacterial agents are often needed to be used for treatment, and the substances have the problem of dissolution from the polymer materials, so that side effects such as cytotoxicity, teratogenesis, genetic mutation and the like can generate new risks and injuries; third, these materials tend to have strong cell adhesion and poor biocompatibility, including but not limited to cytotoxicity, blood compatibility, and tissue compatibility, which are prone to adhesion, hemolysis, and the like. These common problems are also the main reason that the antibacterial medical equipment products at home and abroad are few, so the problem of the lack of the processable and formed medical polymer material which has the functions of antibacterial, antivirus, non-toxicity, biocompatibility and the like and does not influence the basic physical properties of material mechanics, thermal and the like is urgently needed to be solved.
The antibacterial property is that bacteria are killed by a chemical or physical method, or the bacterial growth and the activity are hindered, namely, the two degrees of sterilization and bacteriostasis are divided, wherein the sterilization effect can reduce the concentration of living bacteria, and the bacteriostasis can slow down the bacterial growth and the growth speed, namely, the concentration of the living bacteria is more slowly increased than that of a blank control. The antimicrobial properties of a material can be characterized by determining the number of viable bacteria in the bacterial fluid after contact with the material. The antibacterial adhesion refers to the property of preventing bacteria from fixedly growing on the surface of a material, does not influence the increase of the concentration of living bacteria, and can be characterized by measuring the number of bacteria adhered to the surface before and after the material is contacted with bacterial liquid.
The polymer materials for the antibacterial medical equipment, which are reported at present, are mainly prepared by adding various antibacterial agents into polymer matrix materials and performing molding processing processes through methods such as melt extrusion, melt blowing, solution spinning and the like, so that the polymer materials for the antibacterial medical equipment must have good antibacterial property, biocompatibility and molding processability at the same time, and any one of the three properties is not satisfied, and cannot be used as the material for the antibacterial medical equipment.
The quaternary ammonium salt compound is widely applied to antibacterial materials due to the broad-spectrum efficient bactericidal property. The quaternary ammonium salt compound can be divided into small molecular quaternary ammonium salt, non-hyperbranched polymer polyquaternary ammonium salt and hyperbranched polymer (hyper branched polymer) polyquaternary ammonium salt. Wherein, the molecular chain structure of the non-hyperbranched polymer is long straight chain or a small amount of side groups exist, but the molecular chain is still mainly a linear main chain. Hyperbranched polymer molecules have a highly branched three-dimensional spherical structure (C.Gao, D.yan, hyperbranched polymers: from synthesis to applications.progress in Polymer Science,2004, 29:183-275) and are polymerized by the multifunctional monomer one-pot method. The hyperbranched polymer is one of dendritic polymers, and is different in that branching of the dendritic polymer is irregular, so that the problems of complex synthesis process and high cost are solved, and large-scale industrial production and application are difficult. As is well known to those skilled in the art, hyperbranched structural polymer names must have a hyperbranched naming prefix.
For the small molecular quaternary ammonium salt antibacterial polymer material, the antibacterial component is a cationic quaternary ammonium salt group, and the antibacterial mechanism is that the positively charged quaternary ammonium group damages bacterial cell membranes after contacting bacteria, and denatures proteins or damages cell structures in the quaternary ammonium group to kill the bacteria. For example, the invention CN113736200a adds a small molecule quaternary ammonium salt antimicrobial agent to a super absorbent resin to prepare an antimicrobial resin composite. However, the small molecular quaternary ammonium salt antibacterial agent is dissolved in water, is similar to a metal ion or metal nanoparticle antibacterial agent, has low compatibility with a high molecular matrix material, has the risk of easy dissolution, has high electropositivity, also has the effect of killing broken membranes of mammalian cells, and has the biocompatibility problems of high cytotoxicity, high hemolysis rate, strong protein adhesion and the like caused by dissolution. Therefore, the organic small molecular antibacterial polymer material cannot meet the requirements of medical instruments.
Non-hyperbranched polyquaternary ammonium salt antibacterial polymer materials, for example, the invention patent CN112646110A further synthesizes polyurethane by synthesizing glycol monomers containing gemini quaternary ammonium salt to obtain gemini quaternary ammonium salt polyurethane coating materials with antibacterial performance; the invention patent CN105999406A obtains the antibacterial quaternary ammonium salt chitosan composite gel coating through the surface modification of the chitosan/polyvinyl alcohol composite gel coating by 1-bromoheptane. The sterilizing mechanism of the polyquaternary ammonium salt is material contact sterilization, and the strong positive charges of a large number of quaternary ammonium salt groups on the surface of the material attract bacteria to contact the surface of the material, and the strong attraction of the quaternary ammonium salt groups and the negative charges of the bacterial cell membrane attract phospholipids to damage the cell membrane structure, change the cell membrane permeability and cause death such as cytoplasmic leakage. However, compared with small molecular quaternary ammonium salt, the polyquaternary ammonium salt has higher cation concentration on the surface of the material, and is easy to cause biocompatibility reduction and adhesion of bacteria to the surface of the material due to excessively strong electrostatic attraction with bacteria, cells, proteins and other substances, thereby influencing long-term antibacterial effect and service performance. There are also polymer materials in which non-hyperbranched polyquaternary ammonium salts are crosslinked, but the crosslinked polymer in net form itself has been of a body-type structure, and has no continuous molding processability, and cannot be used as an antibacterial medical instrument material. For example, the invention patent CN111675789a is to prepare antibacterial hydrogel by copolymerization and crosslinking of quaternary ammonium salt and acrylamide, so that the conventional antibacterial material based on non-hyperbranched high molecular polymer has the defects of low biocompatibility, easy adhesion pollution on the surface of the material and the like, and cannot be used as an antibacterial medical instrument material.
Aiming at the problems that the biocompatibility of the high molecular antibacterial material is insufficient or adhesion pollution of proteins, bacteria and the like is easy to generate, the method of introducing zwitterions, mixed charge polymers or responsive switching groups as modified substances is adopted, and the high molecular antibacterial material has the property of effectively inhibiting the adhesion of the surfaces of the substances such as the proteins, the bacteria and the like by enabling the surfaces to be rich in positive and negative charges and generate hydrogen bond interaction with water molecules to generate a hydration layer. For example, invention patent CN105199070a incorporates a zwitterionic into the polyurethane backbone to obtain antibacterial adhesion properties; the invention patent US10905796B2 proposes the introduction of zwitterionic betaine structures into thermoplastic elastomeric supramolecular polymers to improve resistance adhesion and biocompatibility; fan et al polymerize vinyl monomers containing quaternary ammonium salt, carboxyl group, and n-butyl group, and synergistically prepare a coating material with both antibacterial and blood compatibility by means of mixed charge (Xiao-li Fan, mihu, zhi-hui Qin, et al, bactericidal and hemocompatible Coating via the Mixed-Charged Copolymer [ J ]. ACS Applied Materials & Interfaces,2018, 10:10428-10436.); the invention patent CN112007209A achieves the anti-sterilization and release-integrated gel dressing with salt response by introducing epoxidation zwitterionic into quaternized chitosan. However, the sterilization capability of the material containing only the zwitterionic polymer is weak, and the application field is limited.
Hyperbranched polyquaternary ammonium salt is a polymer with a highly branched three-dimensional spherical structure, and is prepared by grafting or quaternizing quaternary ammonium salt groups of the hyperbranched polymer or directly polymerizing monomers containing quaternary ammonium salt groups. Because hyperbranched polyquaternary ammonium salt powder, solution or coating material is used and does not have independent molding processability, the hyperbranched polyquaternary ammonium salt powder, solution or coating material has less application in the aspect of solid materials and is mainly applied to the fields of dye adsorption, demulsifiers and the like in antistatic agents and wastewater treatment. The hyperbranched polyquaternary ammonium salt is mainly used for the research of antibiosis of material surface coatings (Lia A.T. W.Asti, mihaela Crismaru, steven Roest, et al, A Shape-Adaptive, antibacterial-Coating of Immobilized Quaternary-Ammonium Compounds Tethered on hyperbranched Polyurea and its Mechanism of Action [ J ]. Advanced Functional Materials,2013,24:346-355 ]) and textile antibiosis modification (Kun Chen, xangdong Zhou, xangrong Wang, et al, synthesis and Application of ahyperbranched Polyester Quaternary Ammonium Surfactant [ J ]. Journal of Surfactants and Detergents,2014,17:1081-1088 ]), but the sterilization mechanism of the hyperbranched polyquaternary ammonium salt is similar to that of the hyperbranched polyquaternary ammonium salt, so that the same problem of difficulty in balancing antibiosis and biocompatibility exists, and the requirements of high polymer materials for medical devices are difficult to meet.
In summary, the prior art is difficult to meet the requirements of simple operation, low cost, lasting effect, excellent performance and controllable biomedical materials for good antibacterial property, biocompatibility and formability.
Disclosure of Invention
Aiming at the defects of the prior art, the invention aims to solve the technical problems that: the polymer material for the medical instrument has the characteristics of good antibacterial property, biocompatibility and formability, simple preparation method, low cost, excellent performance and lasting effect, overcomes the defects that the antibacterial property and the biocompatibility are difficult to achieve simultaneously and the forming processability is difficult to achieve in the prior art, and solves the problem that the surface of the medical polymer material is harmful to human health due to the growth of a biological film and bacterial infection caused by bacterial adhesion.
The invention adopts the following technical scheme:
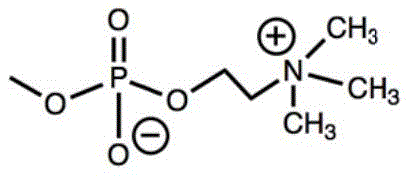
the invention provides an antibacterial composite material based on hyperbranched polyquaternary ammonium salt, which comprises the hyperbranched polyquaternary ammonium salt and a matrix polymer, wherein the mass content of the matrix polymer is 90-99.5%, and the mass content of the hyperbranched polyquaternary ammonium salt is 0.5-10%, and the structural formula of the hyperbranched polyquaternary ammonium salt is as follows:
Wherein R is 1 The hyperbranched polymer is preferably selected from any one of hyperbranched polyamide-amine, hyperbranched polyethyleneimine, hyperbranched polyester and hyperbranched polyurethane; preferably, the structure of the hyperbranched polymer is selected from any one of the following structures:
R 2 containing quaternary ammonium salt groups, preferably, said R 2 The structure is selected from any one of the following structures:
R 3 is long-chain alkyl, the carbon number of the long-chain alkyl is more than or equal to 6, preferably, the carbon number of the long-chain alkyl is 6-18;
R
4 comprising anionic or zwitterionic groups; preferably, the anionic group is selected from at least one of a sulfonic acid group, a carboxylic acid group, a phosphoric acid group; preferably, the zwitterionic group is selected from at least one of the structures of the sulfobetaine, the carboxybetaine and the phosphorylcholine which are coexistent with anions and cations,wherein the sulfobetaine structure is
The carboxyl betaine structure is +.>
The phosphorylcholine structure is +.>
Preferably, the matrix polymer is selected from any one or more of polyurethane, polyether amide, polyester, polyvinyl chloride and polypropylene;
preferably, the element mole ratio of the nitrogen positive ions on the surface of the antibacterial composite material is 0.40% -2%;
Preferably, the hyperbranched polyquaternary ammonium salt-based antibacterial composite material has bactericidal property and biocompatibility, wherein the bactericidal rate for escherichia coli reaches >99%, the cell survival rate reaches >85%, and the hemolysis rate is <2%. Wherein, the sterilization rate in the invention refers to the escherichia coli sterilization rate.
Preferably, in the antibacterial composite material, the hyperbranched polyquaternary ammonium salt is hyperbranched polyamide-amine polyquaternary ammonium salt, the matrix polymer is polyurethane, the mass content of the hyperbranched polyquaternary ammonium salt is 0.5-3%, and when the mass content of the matrix polymer is 97-99.5%, the molar ratio of elements of nitrogen positive ions in quaternary ammonium salt groups on the surface of the antibacterial composite material is 0.50-0.70%; preferably, the sterilization rate of the escherichia coli is more than 99.9%, and the long-term sterilization rate is more than 90%; preferably, the cell viability is >90%, the long-term cell viability is >95%, and the hemolysis is <0.5%.
Preferably, the antimicrobial composite comprises any one of the following:
1) The matrix polymer is polyurethane, and the hyperbranched polyquaternary ammonium salt is any one selected from hyperbranched polyurethane polyquaternary ammonium salts and hyperbranched polyamide-amine polyquaternary ammonium salts;
2) The matrix polymer is polyester, and the hyperbranched polyquaternary ammonium salt is hyperbranched polyester polyquaternary ammonium salt;
3) The matrix polymer is polypropylene, and the hyperbranched polyquaternary ammonium salt is hyperbranched polyester polyquaternary ammonium salt;
4) The matrix polymer is polyether amide, and the hyperbranched polyquaternary ammonium salt is any one of hyperbranched polyamide-amine polyquaternary ammonium salt and hyperbranched polyethylene imine polyquaternary ammonium salt;
5) The matrix polymer is polyvinyl chloride, and the hyperbranched polyquaternary ammonium salt is selected from any one of hyperbranched polyamide-amine polyquaternary ammonium salt and hyperbranched polyester polyquaternary ammonium salt.
Preferably, the hyperbranched polyquaternary ammonium salt is hyperbranched polyamide-amine polyquaternary ammonium salt;
preferably, the preparation method comprises the following steps: dissolving hyperbranched polyamide-amine in a solvent, and adding R-containing components 2 Compounds containing radicals R 3 Compounds containing groups, containing or reacting chemically to give R 4 The compound of the group reacts with hyperbranched polyamide-amine to remove the solvent, so as to obtain the hyperbranched polyamide-amine polyquaternary ammonium salt;
more preferably, the compound containing quaternary ammonium salt group is selected from any one or more of 2, 3-epoxypropyl trimethyl ammonium chloride, 2, 3-epoxypropyl triethyl ammonium chloride, 3-chloro-2-hydroxypropyl trimethyl ammonium chloride, 2-chloroethyl trimethyl ammonium chloride and dimethylaminoethyl methacrylate quaternary ammonium salt;
More preferably, the long chain alkyl group-containing compound is selected from any one or more of 1-iodo, 1-bromo, 1-chloro, 1-acyl chloride structure of straight chain alkane containing 6-18 carbon atoms;
more preferably, the compound containing or forming the anionic group through chemical reaction is selected from any one or more of 1, 3-propane sultone, chlorosulfonic acid, chloroacetic acid and carboxyl-containing acrylate compounds;
more preferably, the zwitterionic group-containing compound is selected from any one or more of sulfobetaine methacrylate, carboxybetaine methacrylate, 2-methacryloyloxyethyl phosphorylcholine;
more preferably, R is obtained after said containing or passing through a chemical reaction 2 ,R 3 ,R 4 The R is obtained after the hyperbranched polyamide-amine is reacted with the components of the compound containing the groups or by chemical reaction 2 ,R 3 ,R 4 40-80% of the total mass of all components of the group compound; i.e.
Wherein,,
for hyperbranched polyamide-amine masses, +.>
Is R-containing
2 Compound mass of the radical,/->
Is R-containing
3 Compound mass of the radical,/->
Containing or by chemical reaction to give R
4 A compound of groups;
further preferably, R is obtained after said containing or passing through a chemical reaction 2 ,R 3 ,R 4 The molar content of the quaternary ammonium salt group is 40-60%, the molar content of the long-chain alkyl group is 20-40%, and the molar content of the anionic or zwitterionic group is 20-40% in the three compound components of the group;
preferably, the solvent is selected from any one of methanol, ethanol, N-dimethylformamide and N, N-dimethylacetamide; more preferably, the reaction conditions are at a temperature of 60℃and a reaction time of 6 to 10 hours.
Preferably, the hyperbranched polyquaternium is a hyperbranched polyethyleneimine polyquaternium;
preferably, the preparation method comprises the following steps: dissolving hyperbranched polyethyleneimine in a solvent, and adding R-containing components respectively 2 Compounds containing radicals R 3 Compounds containing groups, containing or reacting chemically to give R 4 The compound of the group reacts with hyperbranched polyethyleneimine and then removes the solvent to obtain the hyperbranched polyethyleneimine polyquaternary ammonium salt;
more preferably, the compound containing quaternary ammonium salt group is selected from any one or more of 2, 3-epoxypropyl trimethyl ammonium chloride, 2, 3-epoxypropyl triethyl ammonium chloride, 3-chloro-2-hydroxypropyl trimethyl ammonium chloride, 2-chloroethyl trimethyl ammonium chloride and dimethylaminoethyl methacrylate quaternary ammonium salt;
More preferably, the long chain alkyl group-containing compound is selected from any one or more of 1-iodo, 1-bromo, 1-chloro, 1-acyl chloride structure of straight chain alkane containing 6-18 carbon atoms;
more preferably, the compound containing or forming the anionic group through chemical reaction is selected from any one or more of 1, 3-propane sultone, chlorosulfonic acid, chloroacetic acid and carboxyl-containing acrylate compounds;
more preferably, the zwitterionic group-containing compound is selected from any one or more of sulfobetaine methacrylate, carboxybetaine methacrylate, 2-methacryloyloxyethyl phosphorylcholine;
more preferably, R is obtained after said containing or passing through a chemical reaction 2 ,R 3 ,R 4 The R is obtained after the hyperbranched polyethyleneimine is contained or chemically reacted with all components of the compound of the group 2 ,R 3 ,R 4 40-80% of the total mass of all components of the group compound; i.e.
Wherein,,
for hyperbranched polyethyleneimine mass, +.>
Is R-containing
2 Compound mass of the radical,/->
Is R-containing
3 Compound mass of the radical,/->
Containing or by chemical reaction to give R
4 A compound of groups;
further preferably, R is obtained after said containing or passing through a chemical reaction 2 ,R 3 ,R 4 The molar content of the quaternary ammonium salt group is 40-60%, the molar content of the long-chain alkyl group is 20-40%, and the molar content of the anionic or zwitterionic group is 20-40% in the three compound components of the group;
preferably, the solvent is selected from any one of methanol and ethanol; more preferably, the reaction conditions are 60℃and the reaction time is from 6 to 10 hours.
Preferably, the hyperbranched polyquaternary ammonium salt is hyperbranched polyester polyquaternary ammonium salt or hyperbranched polyurethane polyquaternary ammonium salt;
preferably, the preparation method comprises the following steps: dissolving hyperbranched polyester or hyperbranched polyurethane in a solvent, and adding R-containing components respectively 2 Compounds containing radicals R 3 Compounds containing groups, containing or reacting chemically to give R 4 The compound of the group reacts with hyperbranched polyester or hyperbranched polyurethane to remove the solvent, and hyperbranched polyester polyquaternary ammonium salt or hyperbranched polyurethane polyquaternary ammonium salt is obtained; more preferably, the compound containing quaternary ammonium salt group is selected from any one or more of 2, 3-epoxypropyl trimethyl ammonium chloride and 2, 3-epoxypropyl triethyl ammonium chloride;
more preferably, the long chain alkyl group-containing compound is selected from any one or more of 1-acyl chloride, 1, 2-epoxy structure, and linear alkane containing 6-18 carbon atoms;
More preferably, the compound containing or forming the anionic group through chemical reaction is selected from any one or more of 1, 3-propane sultone and chlorosulfonic acid;
more preferably, R is obtained after said containing or passing through a chemical reaction 2 ,R 3 ,R 4 The R is obtained after the hyperbranched polyester or hyperbranched polyurethane is mixed with the compound containing the groups or through chemical reaction 2 ,R 3 ,R 4 40-80% of the total mass of all components of the group compound; i.e.
Wherein,,
is the mass of hyperbranched polyester or hyperbranched polyurethane, < ->
Is R-containing
2 The mass of the compound of the group,
is R-containing
3 Compound mass of the radical,/->
Containing or by chemical reaction to give R
4 A compound of groups;
further preferably, R is obtained after said containing or passing through a chemical reaction 2 ,R 3 ,R 4 The molar content of the quaternary ammonium salt group is 40-60%, the molar content of the long-chain alkyl group is 20-40%, and the molar content of the anionic group is 20-40% in the three compound components of the group;
preferably, the solvent is selected from any one of tetrahydrofuran, N-dimethylformamide and N, N-dimethylacetamide; more preferably, the reaction temperature is 70 ℃, the reaction time is 8-10h respectively, and the reaction is catalyzed by sodium hydride.
Preferably, the antibacterial composite material is prepared by blending hyperbranched polyquaternary ammonium salt and matrix polymer.
The present invention also provides an antimicrobial composite comprising an antimicrobial composite as described above, and at least one additive; preferably, the additive is selected from any one or more of synthetic high molecular compounds, modified high molecular polysaccharides, pigments, reinforcing agents, plasticizers, antioxidants, mildew inhibitors, antibiotics, anti-inflammatory agents, analgesics and antihistamines; preferably, the synthetic polymer compound is selected from one or more of polymer compounds prepared by polyaddition reaction, polymer compounds prepared by polycondensation reaction, polyamide polymer compounds and synthetic rubber polymer compounds; preferably, the antibiotic is selected from any one or more of quinolone antibiotics, beta-lactam antibiotics, macrolides and aminoglycoside antibiotics; preferably, the modified polymer polysaccharide is selected from any one or more of carboxyl modified polymer polysaccharide, amino modified polymer polysaccharide and sulfhydryl modified polymer polysaccharide; preferably, the high molecular polysaccharide is selected from any one or more of cellulose, lignin, starch, chitosan and agarose.
The invention also provides an antibacterial polymer material for medical equipment, which is prepared from the antibacterial composite material or the antibacterial composite material; preferably, the preparation method of the antibacterial polymer material comprises the following steps: and (3) processing and forming the antibacterial composite material or the antibacterial composite through a melting, high-speed mixing or solution method route.
The invention also provides a medical instrument which is obtained by processing and preparing the antibacterial polymer material;
preferably, the medical apparatus is a medical pipe, a film, a sheet, a medical non-woven fabric or a medical composite coating material; preferably, the method for forming and preparing the medical tubing, film and sheet comprises the following steps: preparing medical tubing, film or sheet by melt route extrusion, calendaring, casting or solution route;
preferably, the method for preparing the medical non-woven fabric comprises the following steps: preparing a porous non-woven fabric material through a melting or solution spinning route;
preferably, the preparation method of the medical composite coating material comprises the following steps: after the solution is dissolved, coating and drying the surface of the matrix polymer to prepare an antibacterial composite coating material;
More preferably, the medical apparatus is subjected to a molding preparation process, soaking in pure water at 40-60 ℃ for 10-24 hours, and drying at 50 ℃ for 24 hours to realize the enhancement of antibacterial and biocompatibility.
According to the invention, the hyperbranched polyquaternary ammonium salt is prepared by introducing quaternary ammonium salt, long-chain alkyl, anion or zwitterionic functional groups (each functional group is on different hyperbranched polymer end groups) into the hyperbranched polymer at the same time, and is compounded with various matrix polymers, and surprisingly, under the specific range of the ratio of the quaternary ammonium salt, long-chain alkyl, anion or zwitterionic functional groups and the ratio of the hyperbranched polyquaternary ammonium salt to the matrix polymers, the obtained composite material has good antibacterial property, even bactericidal property, biocompatibility and formability, and obtains different balances between antibacterial property and biocompatibility, so that the application range of the material is greatly widened. In the process of preparing the hyperbranched polyquaternary ammonium salt antibacterial composite material into medical instruments, the material forms such as pipes, films, non-woven fabrics, coatings and the like can be arbitrarily selected according to actual demands, and the medical instrument has wide application scenes. The invention has the advantages of low cost of raw materials, better reactivity, and greatly reduced cost, energy consumption and pollution in actual production, and the invention has the following advantages:
(1) The antibacterial composite material based on the hyperbranched polyquaternary ammonium salt disclosed by the invention is simple and easy to implement, and the final product not only has efficient and stable antibacterial and antiviral functions, but also has the characteristics of good anti-adhesion, low toxicity, biocompatibility, formability, and the like.
(2) The synthesized hyperbranched polymer polyquaternary ammonium salt is compounded with the matrix polymer, so that the preparation process is simple, and the synthesized hyperbranched polymer polyquaternary ammonium salt can exist in the matrix polymer stably, so that the problem of dissolution of metal ions, micromolecular bactericides and traditional linear macromolecule bactericides in the material is solved.
(3) The hyperbranched polymer has a large number of reactive end groups, has the advantages of simple synthesis, good solubility, high chemical reactivity, good biocompatibility and the like, and different hyperbranched polymers have different types and numbers of end group reactive groups and physicochemical properties, can be selected in a diversified manner according to the actual performance and the requirements of compatibility with matrix polymers and the like, and can change the types, molecular weights, end group reactive groups and the like of the hyperbranched polymer, thereby being beneficial to functional modification and performance regulation.
(4) The separated quaternary ammonium group, the hydrophobic long-chain alkyl group, the anionic or amphoteric ion group are matched, so that the excellent synergistic effect is achieved, and the optimal balance effect among antibacterial property, anti-adhesion property and biocompatibility can be achieved through the regulation and control of the grafting ratio and grafting ratio of the three functional groups.
(5) The polyquaternary ammonium salt biomedical composite material disclosed by the invention is based on the composition of the polyquaternary ammonium salt hyperbranched polymer and the matrix polymer, has no special requirements on the type and the form of the matrix polymer, can be prepared into various forms such as a catheter, a film, a non-woven fabric, a coating and the like by the methods such as melt extrusion, casting, melt blowing, solution coating and the like, and has very wide application range in the field of biological medicine.
(6) Through the water soaking process after the composite material is molded, the composite material can obtain excellent surface sterilization anti-adhesion performance under the condition of containing very little hyperbranched polymer polyquaternary ammonium salt, so that the problem that a large amount of bactericide needs to be added in the traditional sterilization material is solved, and social value and economic benefit can be considered.
(7) The polyquaternary ammonium salt biomedical composite material disclosed by the invention is simple and feasible in manufacturing process, mild in reaction conditions and processing conditions, low in energy consumption, low in raw material cost, wide in application range and suitable for industrial mass production and application.
Detailed Description
The polyquaternium composite material and the application thereof in medical devices are described in detail below by using specific examples. The implementation steps of all embodiments are the same as those described in the summary. It is noted that the described embodiments do not limit the present invention, and all modifications which can be directly derived or suggested to one skilled in the art from the disclosure of the present invention should be considered as the protection scope of the present invention.
The specific performance test results of the antibacterial composite materials in the following examples and comparative examples are shown in Table 2.
Example 1
The preparation process of the hyperbranched polyurethane polyquaternary ammonium salt antibacterial composite pipe (1) comprises the following steps: 20 parts of hyperbranched polyurethane with molecular weight of 5200 and hydroxyl value of 520mg KOH/g according to parts by mass are dissolved in 80 parts of tetrahydrofuran and stirred, after nitrogen is introduced into a reaction system for 15min and air is discharged, 8 parts of sodium hydride is added, the temperature is raised to 70 ℃, 9 parts of 2, 3-epoxypropyl trimethyl ammonium chloride, 12 parts of 1-acyl chloride dodecane and 7 parts of 1, 3-propane sultone are sequentially added every 3 hours for reaction. Filtering the reaction system to obtain the poly-quaternary ammonium salt hyperbranched polyurethane. And (3) melting and blending the medical polyurethane base material and the polyquaternium hyperbranched polymer at 200 ℃ and then extruding the mixture to prepare a pipe, and then soaking the pipe in water for 15 hours to obtain the hyperbranched polyurethane polyquaternium antibacterial composite pipe (1). Wherein the hyperbranched polyurethane polyquaternary ammonium salt accounts for 5% of the total mass fraction.
The performance evaluation of the prepared hyperbranched polyquaternary ammonium salt antibacterial composite material is mainly characterized by the following characteristic parameters, namely the sterilization rate and the biocompatibility of the material, wherein the biocompatibility is characterized by cytotoxicity and hemolysis rate.
And (3) testing the sterilization rate: the surface of the test material (2.5X2.5 cm) 2 ) And a concentration of 50. Mu.L of 10 7 Bacterial Colony Forming Units (CFU)/mL of escherichia coli is subjected to contact culture for 24 hours, bacterial liquid is subjected to gradient dilution, and the bacterial CFU number is calculated after the bacterial liquid is coated on an agar plate for culture for 16 hours.
The antibacterial ratio BR is defined as: BR= (1-c 2/c 1). Times.100%, c1 and c2 are bacterial CFU counts of the blank sample and the sample to be tested, respectively.
Cytotoxicity test: taking mouse fibroblast L929 as a model cell, and taking a sample to be detected as 3cm 2 Leaching ratio per mL after 24h in MEM medium, the test leaches were tested for 10 at 24h 4 The effect of the growth of the individual L929 cells was quantitatively calculated by absorbance at 450nm for cell viability by CCK-8 reagent. Definition of cell viability CVThe method comprises the following steps: CV= (As-Ab)/(Ac-Ab). Times.100%, and As, ab and Ac are the absorbance of the sample to be measured, blank and negative control, respectively.
Hemolysis rate: measurement by anticoagulated whole pig blood, sample to be measured (1X 1cm 2 ) The contact time with blood was 1h, followed by centrifugation at 3500 rpm for 5min, and the absorbance of the supernatant at 545nm was measured. The haemolysis rate HR is defined as: HR= (As-Anc)/(Apc-Anc). Times.100%, as, anc, apc is absorbance at 545nm of the sample to be tested, negative control (physiological saline), positive control (deionized water), respectively.
The results are shown in Table 2, which shows that the antibacterial performance of the material is obviously improved to 98.8% by blending and compounding the polyquaternium hyperbranched polyurethane and the polyurethane base material, and the biocompatibility is still good.
Comparative example 1
The medical polyurethane base material is melt blended at 200 ℃ and then extruded to prepare a pipe, and then soaked in water for 15 hours to obtain the polyurethane pipe (comparative example 1).
The characterization method of the prepared polyurethane pipe is the same as that of example 1, and the sterilization rate, the cell survival rate and the hemolysis rate are shown in table 2.
The result shows that compared with the pure polyurethane pipe, the hyperbranched polyurethane polyquaternary ammonium salt polyurethane based antibacterial composite pipe has obviously improved antibacterial performance and better biocompatibility.
Example 2
The preparation process of the hyperbranched polyamide-amine polyquaternary ammonium salt polyether amide antibacterial composite membrane material (2) comprises the following steps: 20 parts of hyperbranched polyamide-amine with the molecular weight of 30000 and the ammonia value of 0.01mol/g (obtained by acid-base titration calculation) are dissolved in 80 parts of methanol, nitrogen is introduced into the reaction system for 15min to remove air, 7 parts of 3-chloro-2-hydroxypropyl trimethyl ammonium chloride dissolved in 10 parts of methanol are added, and the mixture is stirred and heated at 60 ℃ for 3 hours. After 5 parts of sodium hydroxide was added to the reaction system, 2 parts of chlorosulfonic acid dissolved in 10 parts of methanol was added and reacted for 3 hours at 60℃with stirring and heating. After that, 4 parts of 1-iodododecane dissolved in 20 parts of methanol was added thereto and reacted for 3 hours with stirring and heating at 60 ℃. Concentrating and cooling the solution to obtain the hyperbranched polyamide-amine polyquaternary ammonium salt. The medical polyether amide base material and hyperbranched polyamide-amine polyquaternary ammonium salt are melt blended at 200 ℃ and then cast into a film, and then soaked in water for 15 hours, so that the hyperbranched polyamide-amine polyquaternary ammonium salt polyether amide antibacterial composite film material (2-1) is obtained. Wherein the hyperbranched polyamide-amine polyquaternary ammonium salt accounts for 3 percent of the total mass fraction.
20 parts of hyperbranched polyamide-amine with the molecular weight of 30000 and the ammonia value of 0.01mol/g are dissolved in 80 parts of methanol, nitrogen is introduced into the reaction system for 15min to remove air, 4 parts of 2, 3-epoxypropyl trimethyl ammonium chloride dissolved in 10 parts of methanol is added, and the mixture is stirred and heated at 60 ℃ to react for 3h. After adding 6 parts of sodium hydroxide to the reaction system, 5 parts of chlorosulfonic acid dissolved in 10 parts of methanol was added and reacted for 3 hours at 60℃with stirring and heating. After that, 4 parts of 1-iodododecane dissolved in 20 parts of methanol was added thereto and reacted for 3 hours with stirring and heating at 60 ℃. Concentrating and cooling the solution to obtain the hyperbranched polyamide-amine polyquaternary ammonium salt. And (2) carrying out melt blending on the medical polyether amide substrate and the hyperbranched polyamide-amine polyquaternary ammonium salt at 200 ℃ and then carrying out tape casting to prepare a film, and then soaking the film in water for 15h to obtain the hyperbranched polyamide-amine polyquaternary ammonium salt polyether amide antibacterial composite film material (2-2). Wherein the hyperbranched polyamide-amine polyquaternary ammonium salt accounts for 3 percent of the total mass fraction.
20 parts of hyperbranched polyamide-amine with the molecular weight of 30000 and the ammonia value of 0.01mol/g are dissolved in 80 parts of methanol, nitrogen is introduced into the reaction system for 15min to remove air, 3 parts of 3-chloro-2-hydroxypropyl trimethyl ammonium chloride dissolved in 10 parts of alcohol are added, and the mixture is reacted for 3h under stirring and heating at 60 ℃. After 8 parts of sodium hydroxide was added to the reaction system, 7 parts of chlorosulfonic acid dissolved in 10 parts of methanol was added and reacted for 3 hours at 60℃with stirring and heating. After that, 4 parts of 1-iodododecane dissolved in 20 parts of methanol was added thereto and reacted for 3 hours with stirring and heating at 60 ℃. Concentrating and cooling the solution to obtain the hyperbranched polyamide-amine polyquaternary ammonium salt. The medical polyether amide base material and hyperbranched polyamide-amine polyquaternary ammonium salt are melt blended at 200 ℃ and then cast into a film, and then soaked in water for 15 hours, so that the hyperbranched polyamide-amine polyquaternary ammonium salt polyether amide antibacterial composite film material (2-3) is obtained. Wherein the hyperbranched polyamide-amine polyquaternary ammonium salt accounts for 3 percent of the total mass fraction.
The characterization method of the prepared hyperbranched polyamide-amine polyquaternary ammonium salt polyether amide antibacterial composite membrane material is the same as that of example 1, and the sterilization rate, the cell survival rate and the hemolysis rate are shown in table 2.
The result shows that the content of quaternary ammonium salt in hyperbranched polyquaternary ammonium salt gradually decreases, the content of sulfonic acid group gradually increases, the surface electropositivity gradually decreases, the sterilization performance gradually decreases, but the level of sterilization performance is still maintained to be more than 95%, the biocompatibility gradually increases, the overall performance is excellent, and the method has practical application prospects such as medical implants or wound dressings.
Comparative example 2
The medical polyether amide base material is prepared into a film by casting after being melt blended at 200 ℃, and then is soaked in water for 15 hours to obtain the polyether amide film material (comparative example 2).
The characterization method of the prepared polyether amide membrane material is the same as that of example 1, and the sterilization rate, the cell survival rate and the hemolysis rate are shown in table 2.
The result shows that compared with the pure polyether amide membrane material, the hyperbranched polyurethane polyquaternary ammonium salt polyurethane-based antibacterial composite material has obviously improved antibacterial performance and excellent biocompatibility, and the material has both antibacterial and biocompatible keys when hyperbranched polyquaternary ammonium salt is used.
Comparative example 3
20 parts of hyperbranched polyamide-amine with the molecular weight of 30000 and the ammonia value of 0.01mol/g are dissolved in 80 parts of methanol, nitrogen is introduced into the reaction system for 15min to remove air, 4 parts of 2, 3-epoxypropyl trimethyl ammonium chloride dissolved in 10 parts of methanol is added, and the mixture is stirred and heated at 60 ℃ to react for 3h. Concentrating and cooling the solution to obtain the hyperbranched polyamide-amine polyquaternary ammonium salt. The medical polyether amide substrate and hyperbranched polyamide-amine polyquaternary ammonium salt are melt blended at 200 ℃ and then cast into a film, and then soaked in water for 15 hours, so that the hyperbranched polyamide-amine polyquaternary ammonium salt polyether amide antibacterial composite film material (comparative example 3) is obtained. Wherein the hyperbranched polyamide-amine polyquaternary ammonium salt accounts for 3 percent of the total mass fraction.
The characterization method of the prepared hyperbranched polyamide-amine polyquaternary ammonium salt polyether amide antibacterial composite membrane material is the same as that of example 1, and the sterilization rate, the cell survival rate and the hemolysis rate are shown in table 2.
Compared with the example 2-2, the result shows that the hyperbranched polyquaternium antibacterial composite material only containing quaternary ammonium salt groups has slightly poorer antibacterial performance and low biocompatibility, and can not meet the requirements of both antibacterial and biocompatibility.
Comparative example 4
20 parts of hyperbranched polyamide-amine with the molecular weight of 30000 and the ammonia value of 0.01mol/g are dissolved in 80 parts of methanol, nitrogen is introduced into the reaction system for 15min to remove air, 4 parts of 2, 3-epoxypropyl trimethyl ammonium chloride dissolved in 10 parts of methanol is added, and the mixture is stirred and heated at 60 ℃ to react for 3h. After adding 6 parts of sodium hydroxide to the reaction system, 4 parts of 1-iodododecane dissolved in 20 parts of methanol was added and reacted for 3 hours with stirring and heating at 60 ℃. Concentrating and cooling the solution to obtain the hyperbranched polyamide-amine polyquaternary ammonium salt. The medical polyether amide substrate and hyperbranched polyamide-amine polyquaternary ammonium salt are melt blended at 200 ℃ and then cast into a film, and then soaked in water for 15 hours, so that the hyperbranched polyamide-amine polyquaternary ammonium salt polyether amide antibacterial composite film material (comparative example 4) is obtained. Wherein the hyperbranched polyamide-amine polyquaternary ammonium salt accounts for 3 percent of the total mass fraction.
The characterization method of the prepared hyperbranched polyamide-amine polyquaternary ammonium salt polyether amide antibacterial composite membrane material is the same as that of example 1, and the sterilization rate, the cell survival rate and the hemolysis rate are shown in table 2.
Compared with the example 2-2, the result shows that the hyperbranched polyquaternary ammonium salt antibacterial composite material simultaneously containing quaternary ammonium salt groups introduced at different hyperbranched end groups and long-chain alkyl groups has excellent antibacterial performance, but has lower biocompatibility, which shows that the alkyl chain has remarkable antibacterial effect, but still needs to be added with the biocompatibility groups for biocompatibility modification.
Comparative example 5
20 parts of hyperbranched polyamide-amine with the molecular weight of 30000 and the ammonia value of 0.01mol/g are dissolved in 80 parts of methanol, nitrogen is introduced into the reaction system for 15min to remove air, 4 parts of 2, 3-epoxypropyl trimethyl ammonium chloride dissolved in 10 parts of methanol is added, and the mixture is stirred and heated at 60 ℃ to react for 3h. After adding 6 parts of sodium hydroxide to the reaction system, 5 parts of chlorosulfonic acid dissolved in 10 parts of methanol was added and reacted for 3 hours at 60℃with stirring and heating. After that, 3 parts of 1-iodobutane dissolved in 20 parts of methanol was added thereto and reacted for 3 hours with stirring and heating at 60 ℃. Concentrating and cooling the solution to obtain the hyperbranched polyamide-amine polyquaternary ammonium salt. The medical polyether amide substrate and hyperbranched polyamide-amine polyquaternary ammonium salt are melt blended at 200 ℃ and then cast into a film, and then soaked in water for 15 hours, so that the hyperbranched polyamide-amine polyquaternary ammonium salt polyether amide antibacterial composite film material (comparative example 5) is obtained. Wherein the hyperbranched polyamide-amine polyquaternary ammonium salt accounts for 3 percent of the total mass fraction.
The characterization method of the prepared hyperbranched polyamide-amine polyquaternary ammonium salt polyether amide antibacterial composite membrane material is the same as that of example 1, and the sterilization rate, the cell survival rate and the hemolysis rate are shown in table 2.
Compared with comparative example 2-2, the result shows that the hyperbranched polyquaternium antibacterial composite material simultaneously containing quaternary ammonium salt groups, long-chain alkyl groups and anionic groups introduced at different hyperbranched end groups has good antibacterial performance, but the existence of short alkyl chains makes the composite material not have excellent enough antibacterial performance, which shows that the existence of long-chain alkyl groups plays an important role in antibacterial effect.
Comparative example 6
20 parts of hyperbranched polyamide-amine with the molecular weight of 30000 and the ammonia value of 0.01mol/g are dissolved in 80 parts of methanol, nitrogen is introduced into the reaction system for 15 minutes to remove air, 9 parts of 2, 3-epoxypropyl dimethyl dodecyl ammonium chloride dissolved in 10 parts of methanol is added, and the mixture is reacted for 3 hours under stirring and heating at 60 ℃. Concentrating and cooling the solution to obtain the hyperbranched polyamide-amine polyquaternary ammonium salt. The medical polyether amide substrate and hyperbranched polyamide-amine polyquaternary ammonium salt are melt blended at 200 ℃ and then cast into a film, and then soaked in water for 15 hours, so that the hyperbranched polyamide-amine polyquaternary ammonium salt polyether amide antibacterial composite film material (comparative example 6) is obtained. Wherein the hyperbranched polyamide-amine polyquaternary ammonium salt accounts for 3 percent of the total mass fraction.
The characterization method of the prepared hyperbranched polyamide-amine polyquaternary ammonium salt polyether amide antibacterial composite membrane material is the same as that of example 1, and the sterilization rate, the cell survival rate and the hemolysis rate are shown in table 2.
Compared with comparative example 2-2, the result shows that the hyperbranched polyquaternary ammonium salt antibacterial composite material obtained by connecting quaternary ammonium salt groups and long-chain alkyl groups together is not as good as the hyperbranched polyquaternary ammonium salt antibacterial composite material obtained by separating and grafting quaternary ammonium salt groups and long-chain alkyl groups, and the quaternary ammonium salt groups and long-chain alkyl groups are separated to be an important structure of the hyperbranched polyquaternary ammonium salt, so that the material has important significance in resisting bacteria, biocompatibility and processability.
Example 3
The preparation process of the hyperbranched polyamide-amine polyquaternary ammonium salt polyether amide antibacterial composite pipe (3) comprises the following steps: 20 parts of hyperbranched polyamide-amine with the molecular weight of 30000 and the ammonia value of 0.01mol/g are dissolved in 100 parts of N, N-dimethylformamide, nitrogen is introduced into the reaction system for 15min to remove air, 5 parts of sodium hydroxide is added into the reaction system, 7 parts of dimethylaminoethyl methacrylate trimethylammonium chloride, 7 parts of sulfobetaine methacrylate and 4 parts of 1-iodododecane are added into the reaction system, and the reaction is carried out for 8 hours under stirring and heating at 60 ℃. Filtering the solution to obtain the hyperbranched polyamide-amine polyquaternary ammonium salt precipitate. And (3) melting and blending the medical polyether amide substrate and the hyperbranched polyamide-amine polyquaternary ammonium salt at 200 ℃ and then extruding the mixture to prepare a pipe, and then soaking the pipe in water for 15 hours to obtain the hyperbranched polyamide-amine polyquaternary ammonium salt polyether amide antibacterial composite film. Wherein the hyperbranched polymer of the poly quaternary ammonium salt accounts for 1 percent of the total mass fraction to obtain the hyperbranched polyamide-amine poly quaternary ammonium salt polyether amide antibacterial composite pipe (3-1); the hyperbranched polymer of the poly quaternary ammonium salt accounts for 5 percent of the total mass fraction to obtain the hyperbranched polyamide-amine poly quaternary ammonium salt polyether amide antibacterial composite pipe (3-2); the hyperbranched polymer of the poly quaternary ammonium salt accounts for 10 percent of the total mass fraction, and the hyperbranched polyamide-amine poly quaternary ammonium salt polyether amide antibacterial composite pipe (3-3) is obtained.
The characterization method of the hyperbranched polyamide-amine polyquaternary ammonium salt polyether amide antibacterial composite pipe is the same as that of example 1, and the sterilization rate, the cell survival rate and the hemolysis rate are shown in table 2.
The result shows that the hyperbranched polyamide-amine polyquaternary ammonium salt polyether amide antibacterial composite membrane material has excellent performances, the surface electropositivity is gradually enhanced along with the gradual increase of the addition amount of the polyquaternary ammonium salt hyperbranched polymer, the sterilization performance is gradually increased to be more than 99.9%, the biocompatibility is slightly reduced, but the overall effect is excellent, and the composite membrane material has practical application prospects of in-vitro wound dressing, in-vivo implantation catheters, wound healing promoting hydrogels and the like.
Example 4
The preparation process of the hyperbranched polyester poly quaternary ammonium salt polyester based antibacterial composite nonwoven fabric (4) comprises the following steps: 20 parts of hyperbranched aliphatic polyester with molecular weight of 5200 and hydroxyl value of 520mg KOH/g are dissolved in 80 parts of tetrahydrofuran and stirred, after nitrogen is introduced into the reaction system for 15 minutes and air is discharged, 8 parts of sodium hydride is added, the temperature is raised to 70 ℃,15 parts of 2, 3-epoxypropyl triethyl ammonium chloride, 15 parts of 1-acyl chloride octadecane and 7 parts of 1, 3-propane sultone are sequentially added every 3 hours for reaction. Filtering the reaction system to obtain the hyperbranched polyester polyquaternium. And (3) carrying out melt-blending on the medical polyester base material and the hyperbranched polyester polyquaternary ammonium salt at the temperature of 250 ℃ and then carrying out melt-blowing spinning to prepare the porous non-woven fabric, wherein the hyperbranched polyester polyquaternary ammonium salt accounts for 5% of the total mass fraction. Then soaking in water for 10 hours to obtain the hyperbranched polyester poly (quaternary ammonium salt) polyester-based antibacterial composite non-woven fabric (4-1).
20 parts of hyperbranched aliphatic polyester with molecular weight of 5200 and hydroxyl value of 520mg KOH/g are dissolved in 80 parts of tetrahydrofuran and stirred, after nitrogen is introduced into the reaction system for 15min and air is discharged, 8 parts of sodium hydride is added, the temperature is raised to 70 ℃, 15 parts of 2, 3-epoxypropyl triethyl ammonium chloride, 12 parts of 1-acyl chloride dodecane and 7 parts of 1, 3-propane sultone are sequentially added every 3 hours for reaction. Filtering the reaction system to obtain the hyperbranched polyester polyquaternium. And (3) carrying out melt-blending on the medical polyester base material and the hyperbranched polyester polyquaternary ammonium salt at the temperature of 250 ℃ and then carrying out melt-blowing spinning to prepare the porous non-woven fabric, wherein the hyperbranched polyester polyquaternary ammonium salt accounts for 5% of the total mass fraction. Then soaking in water for 10 hours to obtain the hyperbranched polyester poly (quaternary ammonium salt) polyester-based antibacterial composite non-woven fabric (4-2).
20 parts of hyperbranched aliphatic polyester with molecular weight of 5200 and hydroxyl value of 520mg KOH/g are dissolved in 80 parts of tetrahydrofuran and stirred, after nitrogen is introduced into the reaction system for 15min and air is discharged, 8 parts of sodium hydride is added, the temperature is raised to 70 ℃, 15 parts of 2, 3-epoxypropyl triethyl ammonium chloride, 9 parts of 1-acyl chloride hexane and 7 parts of 1, 3-propane sultone are sequentially added every 3 hours for reaction. Filtering the reaction system to obtain the hyperbranched polyester polyquaternium. And (3) carrying out melt-blending on the medical polyester base material and the hyperbranched polyester polyquaternary ammonium salt at the temperature of 250 ℃ and then carrying out melt-blowing spinning to prepare the porous non-woven fabric, wherein the hyperbranched polyester polyquaternary ammonium salt accounts for 5% of the total mass fraction. Then soaking in water for 10 hours to obtain the hyperbranched polyester poly (quaternary ammonium salt) polyester-based antibacterial composite non-woven fabric (4-3).
The characterization method of the hyperbranched polyester polyquaternium polyester-based antibacterial composite nonwoven fabric is the same as that of example 1, and the sterilization rate, the cell survival rate and the hemolysis rate are shown in table 2.
The result shows that compared with pure polyester non-woven fabric, the hyperbranched polyester poly quaternary ammonium salt polyester based antibacterial composite non-woven fabric has obviously improved antibacterial performance, and the long-chain alkyl containing 12 carbons has the best sterilizing effect in long-chain alkyl with different lengths. The hyperbranched polyester poly-quaternary ammonium salt polyester-based antibacterial composite non-woven fabric has excellent biocompatibility, and the material has the practical application prospect of directly contacting a human body with medical equipment.
Comparative example 7
The medical polyester base material was melt-blended at 250 ℃ and then melt-blown and spun to prepare a polyester nonwoven fabric (comparative example 7).
The polyester nonwoven fabric was characterized in the same manner as in example 1, and the sterilization rate, cell survival rate and hemolysis rate are shown in Table 2.
Example 5
The preparation process of the hyperbranched polyester polyquaternium polyester-based antibacterial composite coating material (5) comprises the following steps: 20 parts of hyperbranched aliphatic polyester with molecular weight of 5200 and hydroxyl value of 520mg KOH/g are dissolved in 80 parts of tetrahydrofuran and stirred, after nitrogen is introduced into the reaction system for 15min and air is discharged, 8 parts of sodium hydride is added, the temperature is raised to 70 ℃, 9 parts of 2, 3-epoxypropyl trimethyl ammonium chloride, 10 parts of 1-acyl chloride hexane and 5 parts of 1, 3-propane sultone are sequentially added every 3 hours for reaction. Filtering the reaction system to obtain the hyperbranched polyester polyquaternium. The hyperbranched polyester polyquaternary ammonium salt is uniformly dissolved in ethanol solution, and then coated on the surface of a polyester material and dried at 60 ℃. Soaking the dried material in water for 15h to obtain the hyperbranched polyester polyquaternium polyester-based antibacterial composite coating material (5).
The characterization method of the hyperbranched polyester polyquaternium polyester-based antibacterial composite coating material is the same as that of example 1, and the sterilization rate, the cell survival rate and the hemolysis rate are shown in table 2.
The result shows that the hyperbranched polyester polyquaternium polyester-based antibacterial composite coating material has relatively excellent antibacterial performance and biocompatibility, and has application prospect as a medical instrument product.
Example 6
The preparation process of the hyperbranched polyester polyquaternium polyester-based antibacterial composite coating material (6) comprises the following steps: 20 parts of hyperbranched aliphatic polyester with molecular weight of 5200 and hydroxyl value of 520mg KOH/g are dissolved in 80 parts of tetrahydrofuran and stirred, after nitrogen is introduced into the reaction system for 15min and air is discharged, 8 parts of sodium hydride is added, the temperature is raised to 70 ℃, 15 parts of 2, 3-epoxypropyl trimethyl ammonium chloride, 10 parts of 1-acyl chloride hexane and 7 parts of 1, 3-propane sultone are sequentially added every 3 hours for reaction. Filtering the reaction system to obtain the hyperbranched polyester polyquaternium. The hyperbranched polyester polyquaternary ammonium salt is uniformly dissolved in ethanol solution, and then coated on the surface of a polyester material and dried at 60 ℃. Soaking the dried material in water for 15h to obtain the hyperbranched polyester polyquaternium polyester-based antibacterial composite coating material (6).
The characterization method of the hyperbranched polyester polyquaternium polyester-based antibacterial composite coating material is the same as that of example 1, and the sterilization rate, the cell survival rate and the hemolysis rate are shown in table 2.
The result shows that the hyperbranched polyester polyquaternium polyester-based antibacterial composite coating material has relatively excellent antibacterial performance, but relatively weak biocompatibility, and has application prospect as an in-vitro medical antibacterial material.
Example 7
The preparation process of the hyperbranched polyester polyquaternary ammonium salt polypropylene based antibacterial composite nonwoven fabric (7) comprises the following steps: 20 parts of hyperbranched aliphatic polyester with molecular weight of 5200 and hydroxyl value of 520mg KOH/g are dissolved in 80 parts of tetrahydrofuran and stirred, after nitrogen is introduced into the reaction system for 15min and air is discharged, 8 parts of sodium hydride is added, the temperature is raised to 70 ℃, 15 parts of 2, 3-epoxypropyl triethyl ammonium chloride, 16 parts of 1-acyl chloride octadecane and 5 parts of 1, 3-propane sultone are sequentially added every 3 hours for reaction. Filtering the reaction system to obtain the hyperbranched polyester polyquaternium. And (3) carrying out melt-blending on medical polypropylene and hyperbranched polyester polyquaternary ammonium salt at 200 ℃ and then carrying out melt-blowing spinning to prepare the porous non-woven fabric, wherein the hyperbranched polyester polyquaternary ammonium salt accounts for 5% of the total mass fraction. Then soaking in water for 20 hours to obtain the hyperbranched polyester polyquaternary ammonium salt polypropylene based antibacterial composite non-woven fabric (7).
The characterization method of the hyperbranched polyester polyquaternary ammonium salt polypropylene based antibacterial composite nonwoven fabric is the same as in example 1, and the sterilization rate, the cell survival rate and the hemolysis rate are shown in table 2.
The result shows that after the hyperbranched polyester is added into the hyperbranched polyester, the antibacterial performance is obviously improved, but the biocompatibility is relatively weak, and the hyperbranched polyester-based antibacterial composite film material has application prospect as an in-vitro antibacterial material (such as a mask material and the like).
Comparative example 8
The medical polypropylene was melt blended at 200 c and then melt blown spun to prepare a polypropylene nonwoven (comparative example 8).
The polypropylene nonwoven fabric was characterized in the same manner as in example 1, and the sterilization rate, cell survival rate and hemolysis rate are shown in Table 2.
Example 8
The preparation process of the hyperbranched polyester poly (quaternary ammonium salt) polyvinyl chloride-based antibacterial composite coating material (8) comprises the following steps: 20 parts of hyperbranched aliphatic polyester with a molecular weight of 5000 and a hydroxyl value of 520mg KOH/g are dissolved in 80 parts of tetrahydrofuran and stirred, after nitrogen is introduced into the reaction system for 15min and air is discharged, 8 parts of sodium hydride is added, the temperature is raised to 70 ℃, 12 parts of 2, 3-epoxypropyl trimethyl ammonium chloride, 18 parts of 1-acyl chloride dodecane and 6 parts of 1, 3-propane sultone are sequentially added every 3 hours for reaction. Filtering the reaction system to obtain the hyperbranched polyester polyquaternium. The hyperbranched polyester polyquaternary ammonium salt is uniformly dissolved in ethanol solution, then coated on the surface of a polyvinyl chloride material and dried at 60 ℃. Soaking the dried material in water for 15h to obtain the hyperbranched polyester polyquaternium polyvinyl chloride-based antibacterial composite coating material (8).
The characterization method of the hyperbranched polyester polyquaternium polyvinyl chloride based antibacterial composite coating material is the same as that of example 1, and the sterilization rate, the cell survival rate and the hemolysis rate are shown in table 2.
The result shows that the surface antibacterial property of the hyperbranched polyester polyquaternary ammonium salt hyperbranched polyurethane is obviously improved to more than 99.9% by coating the hyperbranched polyester polyquaternary ammonium salt hyperbranched polyurethane on the polyvinyl chloride material, but the biocompatibility is relatively weak, and the hyperbranched polyester polyquaternary ammonium salt hyperbranched polyurethane is suitable for medical instrument application which is not in direct contact with human tissues.
Comparative example 9
The characterization method of the pure polyvinyl chloride material used as comparative example 9 is the same as that of example 1, and the sterilization rate, the cell survival rate and the hemolysis rate are shown in table 2.
Example 9
The preparation process of the hyperbranched polyethyleneimine poly quaternary ammonium salt polyether amide antibacterial composite pipe (9) comprises the following steps: 20 parts of hyperbranched polyethyleneimine with the molecular weight of 20000 and the ammonia value of 0.009mol/g (obtained by acid-base titration calculation) are dissolved in 80 parts of methanol, nitrogen is introduced into the reaction system for 15min to remove air, 9 parts of 2-chloroethyl trimethyl ammonium chloride dissolved in 10 parts of methanol are added, and the mixture is reacted for 3h under stirring and heating at 60 ℃. After 12 parts of sodium hydroxide was added to the reaction system, 7 parts of 1, 3-propane sultone dissolved in 10 parts of methanol was added and reacted for 3 hours with stirring. 10 parts of 1-chlorododecane dissolved in 20 parts of methanol were then added thereto and reacted for 3 hours with stirring and heating at 60 ℃. Concentrating and cooling the solution to obtain the hyperbranched polyethyleneimine polyquaternary ammonium salt. And (3) melting and blending the medical polyether amide substrate and the hyperbranched polyethylene imine polyquaternary ammonium salt at 200 ℃ and extruding the mixture to prepare a pipe, and soaking the pipe in water for 15 hours to obtain the hyperbranched polyethylene imine polyquaternary ammonium salt polyether amide antibacterial composite pipe (9), wherein the hyperbranched polyethylene imine polyquaternary ammonium salt accounts for 5% of the total mass fraction.
The characterization method of the hyperbranched polyethyleneimine poly quaternary ammonium salt polyether amide antibacterial composite pipe is the same as that of example 1, and the sterilization rate, the cell survival rate and the hemolysis rate are shown in table 2.
The result shows that the hyperbranched polyethylene imine poly quaternary ammonium salt polyether amide antibacterial composite pipe has relatively excellent antibacterial performance and biocompatibility, and has application prospect as a medical instrument product.
Example 10
The preparation process of the hyperbranched polyethyleneimine poly quaternary ammonium salt polyether amide antibacterial composite pipe (10) comprises the following steps: 20 parts of hyperbranched polyethyleneimine with the molecular weight of 20000 and the ammonia value of 0.009mol/g are dissolved in 80 parts of methanol, nitrogen is introduced into the reaction system for 15 minutes to remove air, 9 parts of 2-chloroethyl trimethyl ammonium chloride dissolved in 10 parts of methanol is added, and the mixture is reacted for 3 hours under stirring and heating at 60 ℃. After 12 parts of sodium hydroxide was added to the reaction system, 24 parts of carboxybetaine methacrylate dissolved in 10 parts of methanol was added and reacted for 3 hours with stirring. 10 parts of 1-chlorododecane dissolved in 20 parts of methanol were then added thereto and reacted for 3 hours with stirring and heating at 60 ℃. Concentrating and cooling the solution to obtain the hyperbranched polyethyleneimine polyquaternary ammonium salt. The medical polyether amide base material and the hyperbranched polyethylene imine polyquaternary ammonium salt are subjected to melt blending at 200 ℃ and then extruded to prepare a pipe, and then soaked in water for 15 hours to obtain the hyperbranched polyethylene imine polyquaternary ammonium salt polyether amide antibacterial composite pipe (10), wherein the hyperbranched polyethylene imine polyquaternary ammonium salt accounts for 5% of the total mass fraction.
The characterization method of the hyperbranched polyethyleneimine poly quaternary ammonium salt polyether amide antibacterial composite pipe is the same as that of example 1, and the sterilization rate, the cell survival rate and the hemolysis rate are shown in table 2.
The result shows that the hyperbranched polyethylene imine poly quaternary ammonium salt polyether amide antibacterial composite pipe has relatively excellent antibacterial performance and biocompatibility, and has application prospect as a medical instrument product.
Example 11
The preparation process of the hyperbranched polyethyleneimine poly quaternary ammonium salt polyvinyl chloride-based antibacterial composite film material (11) comprises the following steps: 10 parts of hyperbranched polyethyleneimine with the molecular weight of 20000 and the ammonia value of 0.008mol/g are dissolved in 80 parts of methanol, nitrogen is introduced into the reaction system for 15 minutes to remove air, 15 parts of dimethylaminoethyl methacrylate trimethyl ammonium chloride dissolved in 10 parts of methanol is added, and the mixture is reacted for 3 hours under stirring and heating at 60 ℃. After 12 parts of sodium hydroxide was added to the reaction system, 7 parts of 1, 3-propane sultone dissolved in 10 parts of methanol was added and reacted for 3 hours with stirring. 12 parts of 1-bromododecane dissolved in 20 parts of methanol are then added and reacted for 3 hours with stirring and heating at 60 ℃. Concentrating and cooling the solution to obtain the hyperbranched polyethyleneimine polyquaternary ammonium salt. Melting and blending medical polyvinyl chloride and hyperbranched polyethylene imine polyquaternary ammonium salt at 200 ℃ and then carrying out tape casting to prepare a film, and then soaking the film in water for 15 hours to obtain the hyperbranched polyethylene imine polyquaternary ammonium salt polyvinyl chloride-based antibacterial composite film material (11), wherein the hyperbranched polyethylene imine polyquaternary ammonium salt accounts for 8% of the total mass fraction.
The characterization method of the hyperbranched polyethyleneimine poly quaternary ammonium salt polyvinyl chloride-based antibacterial composite membrane material is the same as that of example 1, and the sterilization rate, the cell survival rate and the hemolysis rate are shown in table 2.
The result shows that the hyperbranched polyethyleneimine poly quaternary ammonium salt polyvinyl chloride-based antibacterial composite membrane material has excellent antibacterial performance, but relatively weak biocompatibility, and has application prospect as an in-vitro medical antibacterial material.
Example 12
The preparation process of the hyperbranched polyamide-amine polyquaternium polyvinyl chloride based antibacterial composite membrane material (12) comprises the following steps: 20 parts of hyperbranched polyamide-amine with the molecular weight of 30000 and the ammonia value of 0.01mol/g are dissolved in 80 parts of methanol, nitrogen is introduced into the reaction system for 15min to remove air, 8 parts of dimethylaminoethyl methacrylate trimethyl ammonium chloride dissolved in 10 parts of methanol are added, and the mixture is reacted for 3h under stirring and heating at 60 ℃. After 5 parts of sodium hydroxide was added to the reaction system, 5 parts of 1, 3-propane sultone dissolved in 10 parts of methanol was added and reacted for 3 hours with stirring. 6 parts of 1-bromododecane dissolved in 20 parts of methanol were then added thereto and reacted for 3 hours with stirring and heating at 60 ℃. Concentrating and cooling the solution to obtain the hyperbranched polyamide-amine polyquaternary ammonium salt. Melting and blending medical polyvinyl chloride and hyperbranched polyamide-amine polyquaternary ammonium salt at 200 ℃ and then carrying out tape casting to prepare a film, and then soaking the film in water for 15 hours to obtain the hyperbranched polyamide-amine polyquaternary ammonium salt polyvinyl chloride-based antibacterial composite film material (12), wherein the hyperbranched polyamide-amine polyquaternary ammonium salt accounts for 8% of the total mass fraction.
The characterization method of the hyperbranched polyamide-amine polyquaternium polyvinyl chloride based antibacterial composite membrane material is the same as that of example 1, and the sterilization rate, the cell survival rate and the hemolysis rate are shown in table 2.
The result shows that the hyperbranched polyamide-amine polyquaternium polyvinyl chloride based antibacterial composite membrane material has excellent antibacterial performance, but relatively weak biocompatibility, and has application prospect as an in-vitro medical antibacterial material.
Example 13
The preparation process of the hyperbranched polyamide-amine polyquaternium polyurethane-based antibacterial composite membrane material (13) is shown in fig. 2: 20 parts of hyperbranched polyamide-amine with the molecular weight of 30000 and the ammonia value of 0.01mol/g are dissolved in 80 parts of methanol, nitrogen is introduced into the reaction system for 15min to remove air, 7 parts of 2, 3-epoxypropyl trimethyl ammonium chloride dissolved in 10 parts of methanol is added, and the mixture is stirred and heated at 60 ℃ to react for 3h. After 5 parts of sodium hydroxide was added to the reaction system, 2 parts of 1, 3-propane sultone dissolved in 10 parts of methanol was added, and the mixture was reacted for 3 hours with stirring and heating at 60 ℃. After that, 4 parts of 1-iodododecane dissolved in 20 parts of methanol was added thereto and reacted for 3 hours with stirring and heating at 60 ℃. Concentrating and cooling the solution to obtain the hyperbranched polyamide-amine polyquaternary ammonium salt. The medical polyurethane base material and hyperbranched polyamide-amine polyquaternary ammonium salt are evenly blended at 60 ℃ in N, N-dimethylformamide, then cast and scraped into a film, and then put into water for soaking for 10 hours, thus obtaining the hyperbranched polyamide-amine polyquaternary ammonium salt polyurethane-based antibacterial composite film material. Wherein the hyperbranched polyquaternary ammonium salt accounts for 0.5 percent of the total mass fraction to obtain the hyperbranched polyamide-amine polyquaternary ammonium salt polyurethane-based antibacterial composite membrane material (13-1); the hyperbranched polyquaternium accounts for 1 percent of the total mass fraction to obtain the hyperbranched polyamide-amine polyquaternium polyurethane-based antibacterial composite membrane material (13-2); the hyperbranched polyquaternium accounts for 1 percent of the total mass fraction, and the hyperbranched polyamide-amine polyquaternium polyurethane-based antibacterial composite membrane material (13-3) is obtained.
The characterization method of the sterilization rate, cytotoxicity and hemolysis rate of the hyperbranched polyamide-amine polyquaternium polyurethane-based antibacterial composite membrane material is the same as in the embodiment 1, wherein the sterilization rate takes escherichia coli and staphylococcus aureus as model bacteria.
In addition, the structural composition of the hyperbranched polyamide-amine polyquaternary ammonium salt is prepared by infrared spectroscopy (FTIR) and nuclear magnetic hydrogen spectroscopy 1 H-NMR). The surface element composition of the hyperbranched polyamide-amine polyquaternium polyurethane-based antibacterial composite film is obtained by scanning and peak-splitting calculation through X-ray photoelectron spectroscopy (XPS), the surface potential and isoelectric point (IEP) are measured through an electric analyzer, the mechanical property is characterized by measuring the tensile modulus and the elongation at break through a universal tester, and the thermal property is obtained by measuring the glass transition temperature and the melting temperature through a Differential Scanning Calorimeter (DSC). The protein adsorption resistance of the hyperbranched polyamide-amine polyquaternium polyurethane-based antibacterial composite film material is calculated by measuring the adsorption quantity of fluorescence-marked bovine serum albumin (FITC-BSA) in 12 hours, measuring the fluorescence intensity by a Confocal Laser Scanning Microscope (CLSM), and comparing with the pure polyurethane film protein adsorption result for semi-quantitative calculation; long-term sterilization rate the sterilization rate of the sample against E.coli was characterized by shaking the sample in deionized water at 37℃for 24, 72, 120 hours in the same manner as in example 1; long term cell viability was characterized by testing the cell viability of the test sample extract after 24, 48, 72, 120 hours of co-culture with L929 cells, respectively, in the same manner as in example 1.
Comparative example 10
Dissolving the medical polyurethane substrate in N, N-dimethylformamide at 60 ℃, then carrying out tape casting and scraping to form a film, and then quickly putting the film into water for soaking for 10 hours to obtain the hyperbranched polyamide-amine polyquaternium polyurethane-based antibacterial composite film material.
The performance characterization method is the same as in example 13.
According to the characterization results of example 13 and comparative example 10, FTIR and 1 the H-NMR spectrum (FIG. 3) demonstrates the successful synthesis of hyperbranched polyamide-amine polyquaternium; XPS test results (Table 1) show the content of nitrogen positive ions in quaternary ammonium salt groups on the surface of the material and the enrichment phenomenon of hyperbranched polyamide-amine polyquaternary ammonium salt on the surface of the polyurethane film material; the rising of isoelectric point shows the existence of hyperbranched polyamide-amine polyquaternium on the surface of the polyurethane composite membrane material through the experimental result of the surface potential (figure 4); the mechanical property test (figure 5) shows that the mechanical property of the polyurethane film is not damaged due to the introduction of the hyperbranched polyamide-amine polyquaternium, but the tensile film quantity and the breaking elongation even slightly rise due to chain entanglement and hydrogen bond interaction, so that the effect is excellent; the DSC curve (FIG. 6) also demonstrates that the incorporation of hyperbranched polyamide-amine polyquaternium has no negative effect on the thermal properties of the polyurethane film. The hyperbranched polyamide-amine polyquaternium polyurethane-based antibacterial composite membrane material in the embodiment 13 has very excellent killing effect on escherichia coli and staphylococcus aureus (figure 7), and has better antibacterial effect along with the rising of the content of the hyperbranched polyamide-amine polyquaternium in the composite material, and the long-term antibacterial effect (figure 8) is 91.2 percent (embodiment 13-1), 94.0 (embodiment 13-2) and 96.8 (embodiment 13-3) respectively, and still keeps higher level # >90 percent) proves that the antibacterial performance is high-efficient and stable; the results of fluorescent protein adsorption (fig. 9) also demonstrate that the introduction of hyperbranched polyamide-amine polyquaternium imparts to the polyurethane film the ability to resist protein adsorption, laterally demonstrating the rise in biocompatibility; the hemolysis rate (figure 9) of the hyperbranched polyamide-amine polyquaternium polyurethane-based antibacterial composite membrane material is kept at a very low level<0.2%) demonstrating that the incorporation of hyperbranched polyamide-amine polyquaternium is beneficial for polyurethane membranesImprovement of blood compatibility; the short-term and long-term cytotoxicity test (FIG. 10) results prove that the hyperbranched polyamide-amine polyquaternium polyurethane-based antibacterial composite membrane material has high cell survival rate, and the cell survival rate after 120h of culture is 85.3 percent (comparative example 10), 91.9 percent (example 13-1), 98.3 percent (example 13-2) and 96.3 percent (example 13-3) respectively, so that the composite membrane material has excellent histocompatibility. In conclusion, the hyperbranched polyamide-amine polyquaternary ammonium salt polyurethane-based antibacterial composite membrane material introduced with the hyperbranched polyamide-amine polyquaternary ammonium salt has long-acting antibacterial property and biocompatibility.
TABLE 1 analysis of surface elements of hyperbranched Polyamide-amine Polyquaternium polyurethane based composite Membrane Material in example 13
Comparative example 11
20 parts of hyperbranched polyamide-amine with the molecular weight of 30000 and the ammonia value of 0.01mol/g are dissolved in 80 parts of methanol, nitrogen is introduced into the reaction system for 15min to remove air, 7 parts of 2, 3-epoxypropyl trimethyl ammonium chloride dissolved in 10 parts of methanol is added, and the mixture is stirred and heated at 60 ℃ to react for 3h. After 5 parts of sodium hydroxide was added to the reaction system, 2 parts of chloroacetic acid dissolved in 10 parts of methanol was added and reacted for 3 hours at 60℃with stirring and heating. After that, 4 parts of 1-iodododecane dissolved in 20 parts of methanol was added thereto and reacted for 3 hours with stirring and heating at 60 ℃. Concentrating and cooling the solution to obtain the hyperbranched polyamide-amine polyquaternary ammonium salt. And uniformly blending the medical polyurethane base material and hyperbranched polyamide-amine polyquaternary ammonium salt at the temperature of 60 ℃ in N, N-dimethylformamide, then carrying out tape casting and scraping to form a film, and then putting the film into water for soaking for 10 hours to obtain the hyperbranched polyamide-amine polyquaternary ammonium salt polyurethane-based antibacterial composite film material. Wherein the hyperbranched polyquaternary ammonium salt accounts for 0.1 percent of the total mass fraction, and the hyperbranched polyamide-amine polyquaternary ammonium salt polyurethane-based antibacterial composite membrane material (comparative example 11) is obtained.
The characterization method of the hyperbranched polyamide-amine polyquaternium polyurethane-based antibacterial composite membrane material is the same as that of example 1, and the sterilization rate, the cell survival rate and the hemolysis rate are shown in table 2. The results show that when the mass content of the hyperbranched polyquaternium in the composite material is too low (< 0.5%), the composite material does not have good antibacterial performance.
Comparative example 12
20 parts of hyperbranched polyamide-amine with the molecular weight of 30000 and the ammonia value of 0.01mol/g are dissolved in 80 parts of methanol, nitrogen is introduced into the reaction system for 15min to remove air, 7 parts of 2, 3-epoxypropyl trimethyl ammonium chloride dissolved in 10 parts of methanol is added, and the mixture is stirred and heated at 60 ℃ to react for 3h. After 5 parts of sodium hydroxide was added to the reaction system, 2 parts of chloroacetic acid dissolved in 10 parts of methanol was added and reacted for 3 hours at 60℃with stirring and heating. After that, 4 parts of 1-iodododecane dissolved in 20 parts of methanol was added thereto and reacted for 3 hours with stirring and heating at 60 ℃. Concentrating and cooling the solution to obtain the hyperbranched polyamide-amine polyquaternary ammonium salt. And uniformly blending the medical polyurethane base material and hyperbranched polyamide-amine polyquaternary ammonium salt at the temperature of 60 ℃ in N, N-dimethylformamide, then carrying out tape casting and scraping to form a film, and then putting the film into water for soaking for 10 hours to obtain the hyperbranched polyamide-amine polyquaternary ammonium salt polyurethane-based antibacterial composite film material. Wherein the hyperbranched polyquaternium accounts for 12 percent of the total mass fraction, and the hyperbranched polyamide-amine polyquaternium polyurethane-based antibacterial composite membrane material (comparative example 12) is obtained.
The characterization method of the hyperbranched polyamide-amine polyquaternium polyurethane-based antibacterial composite membrane material is the same as that of example 1, and the sterilization rate, the cell survival rate and the hemolysis rate are shown in table 2. The result shows that when the mass content of the hyperbranched polyquaternary ammonium salt in the composite material is excessively high (> 10%), the composite material does not have good biocompatibility.
In summary, the hyperbranched polyquaternium is prepared by simultaneously introducing quaternary ammonium salt, long-chain alkyl, anion or zwitterionic functional groups (each functional group is on different hyperbranched polymer end groups) into the hyperbranched polymer, and is compounded with various matrix polymers, and the effects of favorable antibacterial and even bactericidal properties, biocompatibility and formability are achieved in the specific range of the ratio of the quaternary ammonium salt, long-chain alkyl, anion or zwitterionic functional groups to the ratio of the hyperbranched polyquaternium to the matrix polymer. Wherein, lead toUltra infrared spectrum (FTIR) and nuclear magnetic hydrogen spectrum 1 H-NMR), combined with the principle of chemical reaction, can determine that quaternary ammonium salts, long chain alkyl groups, anionic or zwitterionic functional groups are introduced at different end groups of the hyperbranched polymer, respectively, but not at the same end group.
The foregoing examples illustrate only a few embodiments of the invention and are described in detail herein without thereby limiting the scope of the invention. It should be noted that it will be apparent to those skilled in the art that several variations and modifications can be made without departing from the spirit of the invention, which are all within the scope of the invention. Accordingly, the scope of the invention should be assessed as that of the appended claims.
Table 2 composition and performance data of hyperbranched polyquaternium antibacterial composite materials in examples 1-13