CN114959121A - Primers and probes for detecting influenza virus subtypes H5, H7 and H9, and application, reagent and digital PCR kit thereof - Google Patents
Primers and probes for detecting influenza virus subtypes H5, H7 and H9, and application, reagent and digital PCR kit thereof Download PDFInfo
- Publication number
- CN114959121A CN114959121A CN202210770639.2A CN202210770639A CN114959121A CN 114959121 A CN114959121 A CN 114959121A CN 202210770639 A CN202210770639 A CN 202210770639A CN 114959121 A CN114959121 A CN 114959121A
- Authority
- CN
- China
- Prior art keywords
- probe
- nucleic acid
- seq
- subtype
- digital pcr
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 239000000523 sample Substances 0.000 title claims abstract description 88
- 241000712461 unidentified influenza virus Species 0.000 title claims abstract description 63
- 238000007847 digital PCR Methods 0.000 title claims abstract description 31
- 239000003153 chemical reaction reagent Substances 0.000 title claims abstract description 12
- 150000007523 nucleic acids Chemical group 0.000 claims abstract description 62
- 238000001514 detection method Methods 0.000 claims abstract description 21
- 239000012634 fragment Substances 0.000 claims description 21
- 101150118163 h gene Proteins 0.000 claims description 21
- 239000000126 substance Substances 0.000 claims description 14
- 108091028043 Nucleic acid sequence Proteins 0.000 claims description 12
- 239000013642 negative control Substances 0.000 claims description 12
- 239000013641 positive control Substances 0.000 claims description 12
- 125000006853 reporter group Chemical group 0.000 claims description 7
- 238000010791 quenching Methods 0.000 claims description 6
- 230000000171 quenching effect Effects 0.000 claims description 6
- 239000013612 plasmid Substances 0.000 claims description 4
- 238000002360 preparation method Methods 0.000 claims description 4
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 4
- UDGUGZTYGWUUSG-UHFFFAOYSA-N 4-[4-[[2,5-dimethoxy-4-[(4-nitrophenyl)diazenyl]phenyl]diazenyl]-n-methylanilino]butanoic acid Chemical compound COC=1C=C(N=NC=2C=CC(=CC=2)N(C)CCCC(O)=O)C(OC)=CC=1N=NC1=CC=C([N+]([O-])=O)C=C1 UDGUGZTYGWUUSG-UHFFFAOYSA-N 0.000 claims description 3
- 102000004190 Enzymes Human genes 0.000 claims description 3
- 108090000790 Enzymes Proteins 0.000 claims description 3
- 102100034343 Integrase Human genes 0.000 claims description 3
- 108010092799 RNA-directed DNA polymerase Proteins 0.000 claims description 3
- ABZLKHKQJHEPAX-UHFFFAOYSA-N tetramethylrhodamine Chemical compound C=12C=CC(N(C)C)=CC2=[O+]C2=CC(N(C)C)=CC=C2C=1C1=CC=CC=C1C([O-])=O ABZLKHKQJHEPAX-UHFFFAOYSA-N 0.000 claims description 3
- 230000035945 sensitivity Effects 0.000 abstract description 11
- 108020004414 DNA Proteins 0.000 description 36
- 108020004707 nucleic acids Proteins 0.000 description 35
- 102000039446 nucleic acids Human genes 0.000 description 35
- 230000000052 comparative effect Effects 0.000 description 23
- 230000003321 amplification Effects 0.000 description 17
- 238000003199 nucleic acid amplification method Methods 0.000 description 17
- 241001473385 H5N1 subtype Species 0.000 description 13
- 241000342557 H7N9 subtype Species 0.000 description 12
- 238000000034 method Methods 0.000 description 11
- 239000000243 solution Substances 0.000 description 11
- 241001473386 H9N2 subtype Species 0.000 description 10
- 206010064097 avian influenza Diseases 0.000 description 7
- 238000010586 diagram Methods 0.000 description 7
- 239000000203 mixture Substances 0.000 description 6
- 208000002979 Influenza in Birds Diseases 0.000 description 5
- 238000012216 screening Methods 0.000 description 5
- 238000012360 testing method Methods 0.000 description 5
- 241000711573 Coronaviridae Species 0.000 description 4
- 241000197306 H1N1 subtype Species 0.000 description 4
- 230000001717 pathogenic effect Effects 0.000 description 4
- 241000700605 Viruses Species 0.000 description 3
- 206010022000 influenza Diseases 0.000 description 3
- 238000003757 reverse transcription PCR Methods 0.000 description 3
- HMUNWXXNJPVALC-UHFFFAOYSA-N 1-[4-[2-(2,3-dihydro-1H-inden-2-ylamino)pyrimidin-5-yl]piperazin-1-yl]-2-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)ethanone Chemical compound C1C(CC2=CC=CC=C12)NC1=NC=C(C=N1)N1CCN(CC1)C(CN1CC2=C(CC1)NN=N2)=O HMUNWXXNJPVALC-UHFFFAOYSA-N 0.000 description 2
- VZSRBBMJRBPUNF-UHFFFAOYSA-N 2-(2,3-dihydro-1H-inden-2-ylamino)-N-[3-oxo-3-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)propyl]pyrimidine-5-carboxamide Chemical compound C1C(CC2=CC=CC=C12)NC1=NC=C(C=N1)C(=O)NCCC(N1CC2=C(CC1)NN=N2)=O VZSRBBMJRBPUNF-UHFFFAOYSA-N 0.000 description 2
- 238000002965 ELISA Methods 0.000 description 2
- 241000713196 Influenza B virus Species 0.000 description 2
- NIPNSKYNPDTRPC-UHFFFAOYSA-N N-[2-oxo-2-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)ethyl]-2-[[3-(trifluoromethoxy)phenyl]methylamino]pyrimidine-5-carboxamide Chemical compound O=C(CNC(=O)C=1C=NC(=NC=1)NCC1=CC(=CC=C1)OC(F)(F)F)N1CC2=C(CC1)NN=N2 NIPNSKYNPDTRPC-UHFFFAOYSA-N 0.000 description 2
- 238000003745 diagnosis Methods 0.000 description 2
- 238000010790 dilution Methods 0.000 description 2
- 239000012895 dilution Substances 0.000 description 2
- 238000011304 droplet digital PCR Methods 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- 238000000605 extraction Methods 0.000 description 2
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 2
- 238000003753 real-time PCR Methods 0.000 description 2
- 230000003612 virological effect Effects 0.000 description 2
- LDXJRKWFNNFDSA-UHFFFAOYSA-N 2-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)-1-[4-[2-[[3-(trifluoromethoxy)phenyl]methylamino]pyrimidin-5-yl]piperazin-1-yl]ethanone Chemical compound C1CN(CC2=NNN=C21)CC(=O)N3CCN(CC3)C4=CN=C(N=C4)NCC5=CC(=CC=C5)OC(F)(F)F LDXJRKWFNNFDSA-UHFFFAOYSA-N 0.000 description 1
- YLZOPXRUQYQQID-UHFFFAOYSA-N 3-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)-1-[4-[2-[[3-(trifluoromethoxy)phenyl]methylamino]pyrimidin-5-yl]piperazin-1-yl]propan-1-one Chemical compound N1N=NC=2CN(CCC=21)CCC(=O)N1CCN(CC1)C=1C=NC(=NC=1)NCC1=CC(=CC=C1)OC(F)(F)F YLZOPXRUQYQQID-UHFFFAOYSA-N 0.000 description 1
- 206010069767 H1N1 influenza Diseases 0.000 description 1
- 241000252866 H7N1 subtype Species 0.000 description 1
- 241000252864 H7N2 subtype Species 0.000 description 1
- 241000252863 H7N3 subtype Species 0.000 description 1
- 241000252868 H7N7 subtype Species 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- 241000712431 Influenza A virus Species 0.000 description 1
- 102000012750 Membrane Glycoproteins Human genes 0.000 description 1
- 108010090054 Membrane Glycoproteins Proteins 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- AFCARXCZXQIEQB-UHFFFAOYSA-N N-[3-oxo-3-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)propyl]-2-[[3-(trifluoromethoxy)phenyl]methylamino]pyrimidine-5-carboxamide Chemical compound O=C(CCNC(=O)C=1C=NC(=NC=1)NCC1=CC(=CC=C1)OC(F)(F)F)N1CC2=C(CC1)NN=N2 AFCARXCZXQIEQB-UHFFFAOYSA-N 0.000 description 1
- 241000712464 Orthomyxoviridae Species 0.000 description 1
- 238000012408 PCR amplification Methods 0.000 description 1
- 239000012807 PCR reagent Substances 0.000 description 1
- 238000011529 RT qPCR Methods 0.000 description 1
- 239000011324 bead Substances 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 238000012631 diagnostic technique Methods 0.000 description 1
- 238000003748 differential diagnosis Methods 0.000 description 1
- 238000009396 hybridization Methods 0.000 description 1
- 238000003018 immunoassay Methods 0.000 description 1
- 208000037797 influenza A Diseases 0.000 description 1
- 230000001404 mediated effect Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012544 monitoring process Methods 0.000 description 1
- 230000035772 mutation Effects 0.000 description 1
- 244000052769 pathogen Species 0.000 description 1
- 230000007918 pathogenicity Effects 0.000 description 1
- 244000144977 poultry Species 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 238000003762 quantitative reverse transcription PCR Methods 0.000 description 1
- 201000010740 swine influenza Diseases 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 230000001018 virulence Effects 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/70—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving virus or bacteriophage
- C12Q1/701—Specific hybridization probes
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/68—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving nucleic acids
- C12Q1/6844—Nucleic acid amplification reactions
- C12Q1/6851—Quantitative amplification
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q2600/00—Oligonucleotides characterized by their use
- C12Q2600/16—Primer sets for multiplex assays
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q2600/00—Oligonucleotides characterized by their use
- C12Q2600/166—Oligonucleotides used as internal standards, controls or normalisation probes
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02A—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE
- Y02A50/00—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE in human health protection, e.g. against extreme weather
- Y02A50/30—Against vector-borne diseases, e.g. mosquito-borne, fly-borne, tick-borne or waterborne diseases whose impact is exacerbated by climate change
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Immunology (AREA)
- General Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Microbiology (AREA)
- Molecular Biology (AREA)
- Analytical Chemistry (AREA)
- Physics & Mathematics (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biochemistry (AREA)
- Biophysics (AREA)
- Biotechnology (AREA)
- Genetics & Genomics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Virology (AREA)
- Measuring Or Testing Involving Enzymes Or Micro-Organisms (AREA)
Abstract
本发明提供了一种检测流感病毒H5、H7、H9亚型的引物和探针及其应用、试剂和数字PCR试剂盒,涉及生物技术领域。本发明提供的检测流感病毒H5、H7、H9亚型的引物和探针,其中引物具有如SEQ ID NO.1‑6所示的核酸序列,探针具有如SEQ ID NO.7‑9所示的核酸序列,该引物和探针特异性强,灵敏度高,能够用于制备检测流感病毒H5、H7、H9亚型的产品,实现对流感病毒H5、H7、H9亚型的检测。
The invention provides a primer and a probe for detecting influenza virus H5, H7 and H9 subtypes and its application, reagent and digital PCR kit, and relates to the field of biotechnology. The primers and probes for detecting influenza virus H5, H7 and H9 subtypes provided by the present invention, wherein the primers have the nucleic acid sequences shown in SEQ ID NO.1-6, and the probes have the nucleic acid sequences shown in SEQ ID NO.7-9 The primers and probes have strong specificity and high sensitivity, and can be used to prepare products for detecting influenza virus H5, H7, and H9 subtypes, and realize the detection of influenza virus H5, H7, and H9 subtypes.
Description
技术领域technical field
本发明涉及生物技术领域,尤其是涉及一种检测流感病毒H5、H7、H9亚型的引物和探针及其应用、试剂和数字PCR试剂盒。The present invention relates to the field of biotechnology, in particular to a primer and probe for detecting influenza virus H5, H7 and H9 subtypes and its application, reagent and digital PCR kit.
背景技术Background technique
流感病毒(Influenza virus)在病毒分类学上属于正粘病毒科。主要通过空气中的飞沫、易感者与感染者之间的接触或与被污染物品的接触而传播,一般秋冬季节是其高发期。流感病毒包括人流感病毒和动物流感病毒,人流感病毒分为甲(A)、乙(B)、丙(C)三型,是流感的病原体。其中甲型流感病毒抗原性易发生变异,可以进一步分为H1N1、H5N1、H7N9等亚型,其中的H和N分别代表流感病毒两种表面糖蛋白。至今发现能直接感染人的禽流感病毒亚型有:H5N1、H7N1、H7N2、H7N3、H7N7、H9N2和H7N9亚型。根据禽流感病毒致病性和毒力的不同,可以将禽流感分为高致病性禽流感、低致病性禽流感和无致病性禽流感。高致病性禽流感目前有H5N1亚型和H7N9亚型尤为引人关注,不仅造成了人类的伤亡,同时重创了家禽养殖业。Influenza virus belongs to the Orthomyxoviridae family in virus taxonomy. It is mainly transmitted through droplets in the air, contact between susceptible and infected persons, or contact with contaminated items. Generally, the autumn and winter seasons are the high incidence period. Influenza viruses include human influenza viruses and animal influenza viruses. Human influenza viruses are divided into three types: A (A), B (B), and C (C), which are the pathogens of influenza. Among them, influenza A virus is prone to mutation in antigenicity and can be further divided into subtypes such as H1N1, H5N1, and H7N9, among which H and N represent the two surface glycoproteins of influenza virus, respectively. So far, the avian influenza virus subtypes that can directly infect humans are: H5N1, H7N1, H7N2, H7N3, H7N7, H9N2 and H7N9 subtypes. According to the different pathogenicity and virulence of avian influenza virus, avian influenza can be divided into highly pathogenic avian influenza, low pathogenic avian influenza and non-pathogenic avian influenza. The H5N1 subtype and the H7N9 subtype of highly pathogenic avian influenza are of particular concern, which not only caused human casualties, but also severely damaged the poultry industry.
现有流感病毒检测方法包括分子诊断和免疫诊断两大方面。分子诊断技术包括反转录-聚合酶链式反应(RT-PCR),环介导等温核酸扩增技术(LAMP),核酸分子杂交,数字PCR,基因芯片等。免疫诊断技术包括酶联免疫(ELISA),荧光免疫,化学发光,胶体金等。The existing influenza virus detection methods include molecular diagnosis and immunodiagnosis. Molecular diagnostic techniques include reverse transcription-polymerase chain reaction (RT-PCR), loop-mediated isothermal nucleic acid amplification (LAMP), nucleic acid molecular hybridization, digital PCR, gene chips, etc. Immunodiagnostic techniques include enzyme-linked immunosorbent assay (ELISA), fluorescence immunoassay, chemiluminescence, and colloidal gold.
数字PCR比传统的RT-PCR具有更高的灵敏度和准确性,最低能够检测出单个拷贝数的样本。通过对核酸随机分配到大量的反应单元中进行扩增反应,扩增结束后对每个反应单元的荧光信号进行采集,最后通过直接计数或泊松分布公式计算得到样品的原始浓度或含量的PCR技术。然而目前还缺少一种能够同时实现对流感病毒H5、H7、H9亚型检测且适用于微滴数字PCR的试剂盒。Digital PCR has higher sensitivity and accuracy than traditional RT-PCR, and can detect samples with a minimum single copy number. By randomly assigning nucleic acids to a large number of reaction units for amplification reaction, after the amplification is completed, the fluorescence signal of each reaction unit is collected, and finally the original concentration or content of the sample is calculated by direct counting or Poisson distribution formula. technology. However, there is still a lack of a kit that can simultaneously detect influenza virus H5, H7, and H9 subtypes and is suitable for droplet digital PCR.
有鉴于此,特提出本发明。In view of this, the present invention is proposed.
发明内容SUMMARY OF THE INVENTION
本发明的第一目的在于提供一种检测流感病毒H5、H7、H9亚型的引物和探针,以解决上述问题中的至少一种。The first object of the present invention is to provide primers and probes for detecting influenza virus H5, H7, and H9 subtypes, so as to solve at least one of the above problems.
本发明的第二目的在于提供上述引物和探针在制备流感病毒H5、H7、H9亚型检测产品中的应用。The second object of the present invention is to provide the application of the above primers and probes in the preparation of influenza virus H5, H7 and H9 subtype detection products.
本发明的第三目的在于提供一种试剂。The third object of the present invention is to provide a reagent.
本发明的第四目的在于提供一种数字PCR试剂盒。The fourth object of the present invention is to provide a digital PCR kit.
第一方面,本发明提供了一种检测流感病毒H5、H7、H9亚型的引物和探针,所述引物包括第一引物对、第二引物对和第三引物对;In a first aspect, the present invention provides a primer and probe for detecting influenza virus H5, H7, and H9 subtypes, and the primers include a first primer pair, a second primer pair, and a third primer pair;
所述第一引物对具有如SEQ ID NO.1和SEQ ID NO.2所示的核酸序列;所述第二引物对具有如SEQ ID NO.3和SEQ ID NO.4所示的核酸序列;所述第三引物对具有如SEQ IDNO.5和SEQ ID NO.6所示的核酸序列;The first primer pair has the nucleic acid sequences shown in SEQ ID NO.1 and SEQ ID NO.2; the second primer pair has the nucleic acid sequences shown in SEQ ID NO.3 and SEQ ID NO.4; The third primer pair has the nucleic acid sequences shown in SEQ ID NO.5 and SEQ ID NO.6;
所述探针包括第一探针、第二探针和第三探针;The probe includes a first probe, a second probe and a third probe;
所述第一探针具有如SEQ ID NO.7所示的核酸序列;所述第二探针具有如SEQ IDNO.8所示的核酸序列;所述第三探针具有如SEQ ID NO.9所示的核酸序列。The first probe has the nucleic acid sequence shown in SEQ ID NO.7; the second probe has the nucleic acid sequence shown in SEQ ID NO.8; the third probe has the nucleic acid sequence shown in SEQ ID NO.9 Nucleic acid sequences shown.
作为进一步技术方案,所述探针的5’端连接有荧光报告基团,3’端连接有荧光淬灭基团。As a further technical solution, the 5' end of the probe is connected with a fluorescent reporter group, and the 3' end is connected with a fluorescence quenching group.
作为进一步技术方案,所述荧光报告基团包括FAM、CY5或HEX;As a further technical solution, the fluorescent reporter group includes FAM, CY5 or HEX;
优选地,所述荧光淬灭基团包括TAMRA、BHQ1或BHQ2。Preferably, the fluorescence quenching group includes TAMRA, BHQ1 or BHQ2.
第二方面,本发明提供了上述引物和探针在制备流感病毒H5、H7、H9亚型检测产品中的应用;In the second aspect, the present invention provides the application of the above primers and probes in the preparation of influenza virus H5, H7, H9 subtype detection products;
优选地,所述检测产品包括数字PCR试剂盒。Preferably, the detection product includes a digital PCR kit.
第三方面,本发明提供了一种试剂,包括所述的引物和探针。In a third aspect, the present invention provides a reagent, including the primers and probes.
第四方面,本发明提供了一种数字PCR试剂盒,包括所述的引物和探针以及数字PCR反应预混液。In a fourth aspect, the present invention provides a digital PCR kit, including the primers and probes and a digital PCR reaction master mix.
作为进一步技术方案,所述数字PCR反应预混液,包括:Buffer、Mg2+、dNTPs、热启动Taq酶和逆转录酶。As a further technical solution, the digital PCR reaction master mix includes: Buffer, Mg 2+ , dNTPs, hot-start Taq enzyme and reverse transcriptase.
作为进一步技术方案,还包括阳性对照品;As a further technical solution, a positive control substance is also included;
所述阳性对照品包括H5亚型H基因片段、H7亚型H基因片段和H9亚型H基因片段。The positive controls include H5 subtype H gene fragments, H7 subtype H gene fragments and H9 subtype H gene fragments.
作为进一步技术方案,所述阳性对照品为含有H5亚型H基因片段、H7亚型H基因片段和H9亚型H基因片段的重组质粒。As a further technical solution, the positive control substance is a recombinant plasmid containing H5 subtype H gene fragment, H7 subtype H gene fragment and H9 subtype H gene fragment.
作为进一步技术方案,还包括阴性对照品;As a further technical solution, a negative control substance is also included;
所述阴性对照品为无核酸酶水。The negative control substance is nuclease-free water.
与现有技术相比,本发明具有如下有益效果:Compared with the prior art, the present invention has the following beneficial effects:
本发明提供的检测流感病毒H5、H7、H9亚型的引物和探针,其中引物具有如SEQ IDNO.1-6所示的核酸序列,探针具有如SEQ ID NO.7-9所示的核酸序列,该引物和探针特异性强,灵敏度高,能够用于制备检测流感病毒H5、H7、H9亚型的产品,实现对流感病毒H5、H7、H9亚型的检测。The present invention provides primers and probes for detecting influenza virus H5, H7 and H9 subtypes, wherein the primers have nucleic acid sequences as shown in SEQ ID NO.1-6, and the probes have nucleic acid sequences as shown in SEQ ID NO.7-9 The nucleic acid sequence, the primers and probes have strong specificity and high sensitivity, and can be used to prepare products for detecting influenza virus H5, H7, and H9 subtypes, and realize the detection of influenza virus H5, H7, and H9 subtypes.
本发明提供的数字PCR试剂盒,采用数字PCR的方法对目的核酸进行检测,其检测灵敏度明显优于荧光定量PCR或者胶体金,能够实现单拷贝模板量的检测。The digital PCR kit provided by the invention adopts the method of digital PCR to detect the target nucleic acid, and its detection sensitivity is obviously better than that of fluorescent quantitative PCR or colloidal gold, and can realize the detection of the amount of a single copy template.
附图说明Description of drawings
为了更清楚地说明本发明具体实施方式或现有技术中的技术方案,下面将对具体实施方式或现有技术描述中所需要使用的附图作简单地介绍,显而易见地,下面描述中的附图是本发明的一些实施方式,对于本领域普通技术人员来讲,在不付出创造性劳动的前提下,还可以根据这些附图获得其他的附图。In order to illustrate the specific embodiments of the present invention or the technical solutions in the prior art more clearly, the following briefly introduces the accompanying drawings that need to be used in the description of the specific embodiments or the prior art. Obviously, the accompanying drawings in the following description The drawings are some embodiments of the present invention. For those of ordinary skill in the art, other drawings can also be obtained based on these drawings without creative efforts.
图1为H5亚型引物探针筛选;Fig. 1 is H5 subtype primer probe screening;
图2为H7亚型引物探针筛选;Fig. 2 is H7 subtype primer probe screening;
图3为H9亚型引物探针筛选;Fig. 3 is H9 subtype primer probe screening;
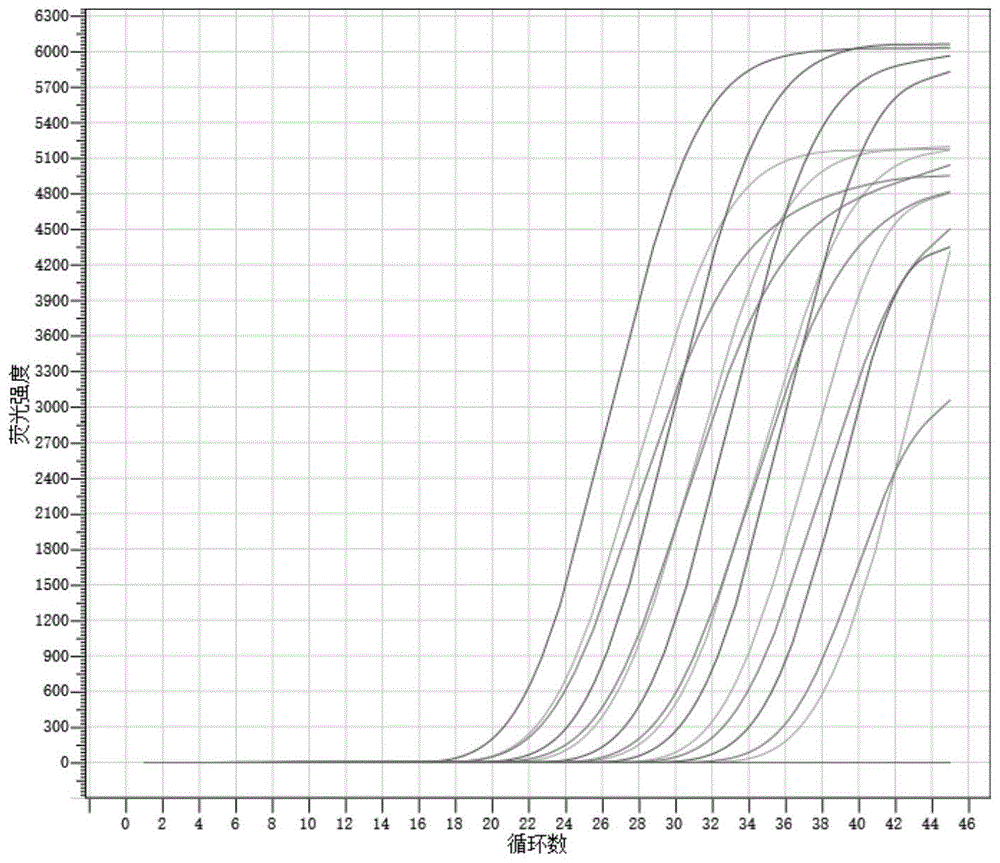
图4为流感病毒H5、H7、H9亚型三重实时荧光定量PCR扩增曲线图;Fig. 4 is the triple real-time fluorescence quantitative PCR amplification curve diagram of influenza virus H5, H7, H9 subtype;
图5为流感病毒H5亚型引物探针特异性的实验结果示意图;Fig. 5 is the schematic diagram of the experimental result of the specificity of the primer probe of influenza virus H5 subtype;
图6为流感病毒H7亚型引物探针特异性的实验结果示意图;Fig. 6 is the schematic diagram of the experimental result of the specificity of the primer probe of influenza virus H7 subtype;
图7为流感病毒H9亚型引物探针特异性的实验结果示意图;Fig. 7 is the schematic diagram of the experimental result of the specificity of the primer probe of influenza virus H9 subtype;
图8为流感病毒H5亚型引物探针灵敏度的实验结果示意图;Figure 8 is a schematic diagram of the experimental results of the sensitivity of the influenza virus H5 subtype primer probe;
图9为流感病毒H7亚型引物探针灵敏度的实验结果示意图;Figure 9 is a schematic diagram of the experimental results of the sensitivity of the influenza virus H7 subtype primer probe;
图10为流感病毒H9亚型引物探针灵敏度的实验结果示意图。FIG. 10 is a schematic diagram of the experimental results of the sensitivity of the primer probe of influenza virus H9 subtype.
具体实施方式Detailed ways
下面将结合实施方式和实施例对本发明的实施方案进行详细描述,但是本领域技术人员将会理解,下列实施方式和实施例仅用于说明本发明,而不应视为限制本发明的范围。基于本发明中的实施例,本领域普通技术人员在没有做出创造性劳动前提下所获得的所有其他实施例,都属于本发明保护的范围。未注明具体条件者,按照常规条件或制造商建议的条件进行。所用试剂或仪器未注明生产厂商者,均为可以通过市售购买获得的常规产品。The embodiments of the present invention will be described in detail below in conjunction with the embodiments and examples, but those skilled in the art will understand that the following embodiments and examples are only used to illustrate the present invention, and should not be regarded as limiting the scope of the present invention. Based on the embodiments of the present invention, all other embodiments obtained by those of ordinary skill in the art without creative efforts shall fall within the protection scope of the present invention. If no specific conditions are specified, follow the general conditions or the conditions suggested by the manufacturer. The reagents or instruments used without the manufacturer's indication are conventional products that can be purchased from the market.
第一方面,本发明提供了一种检测流感病毒H5、H7、H9亚型的引物和探针,所述引物包括第一引物对、第二引物对和第三引物对;In a first aspect, the present invention provides a primer and probe for detecting influenza virus H5, H7, and H9 subtypes, and the primers include a first primer pair, a second primer pair, and a third primer pair;
所述第一引物对具有如SEQ ID NO.1和SEQ ID NO.2所示的核酸序列;所述第二引物对具有如SEQ ID NO.3和SEQ ID NO.4所示的核酸序列;所述第三引物对具有如SEQ IDNO.5和SEQ ID NO.6所示的核酸序列;The first primer pair has the nucleic acid sequences shown in SEQ ID NO.1 and SEQ ID NO.2; the second primer pair has the nucleic acid sequences shown in SEQ ID NO.3 and SEQ ID NO.4; The third primer pair has the nucleic acid sequences shown in SEQ ID NO.5 and SEQ ID NO.6;
所述探针包括第一探针、第二探针和第三探针;The probe includes a first probe, a second probe and a third probe;
所述第一探针具有如SEQ ID NO.7所示的核酸序列;所述第二探针具有如SEQ IDNO.8所示的核酸序列;所述第三探针具有如SEQ ID NO.9所示的核酸序列。The first probe has the nucleic acid sequence shown in SEQ ID NO.7; the second probe has the nucleic acid sequence shown in SEQ ID NO.8; the third probe has the nucleic acid sequence shown in SEQ ID NO.9 Nucleic acid sequences shown.
在一些优选的实施方式中,所述探针的5’端连接有荧光报告基团,3’端连接有荧光淬灭基团。In some preferred embodiments, a fluorescent reporter group is attached to the 5' end of the probe, and a fluorescence quenching group is attached to the 3' end.
在一些优选的实施方式中,所述荧光报告基团包括但不限于FAM、CY5或HEX;所述荧光淬灭基团包括但不限于TAMRA、BHQ1或BHQ2;或者采用本领域技术人员所熟知的其他荧光报告基团和荧光淬灭基团。In some preferred embodiments, the fluorescent reporter groups include but are not limited to FAM, CY5 or HEX; the fluorescence quenching groups include but are not limited to TAMRA, BHQ1 or BHQ2; Other fluorescent reporter groups and fluorescence quenchers.
本发明提供的检测流感病毒H5、H7、H9亚型的引物和探针,特异性强,灵敏度高,能够用于制备检测流感病毒H5、H7、H9亚型的产品,实现对流感病毒H5、H7、H9亚型的检测。The primers and probes for detecting influenza virus H5, H7 and H9 subtypes provided by the present invention have strong specificity and high sensitivity, and can be used to prepare products for detecting influenza virus H5, H7 and H9 subtypes, and realize the detection of influenza virus H5, H7 and H9 subtypes. Detection of H7 and H9 subtypes.
第二方面,本发明提供了上述引物和探针在制备流感病毒H5、H7、H9亚型检测产品中的应用;In the second aspect, the present invention provides the application of the above primers and probes in the preparation of influenza virus H5, H7, H9 subtype detection products;
优选地,所述检测产品包括数字PCR试剂盒。Preferably, the detection product includes a digital PCR kit.
本发明提供的检引物和探针,特异性强,灵敏度高,因此,能够用于制备检测流感病毒H5、H7、H9亚型的产品,进而实现对流感病毒H5、H7、H9亚型的检测。The detection primers and probes provided by the present invention have strong specificity and high sensitivity, and therefore, can be used to prepare products for detecting influenza virus H5, H7, and H9 subtypes, thereby realizing the detection of influenza virus H5, H7, and H9 subtypes. .
第三方面,本发明提供了一种试剂,包括所述的引物和探针。In a third aspect, the present invention provides a reagent, including the primers and probes.
本发明中对于试剂的具体种类不进行限定,包括上述引物和探针即可,可以用于流感病毒H5、H7、H9亚型。上述试剂由于含有上述引物和探针,因此具有特异性强,灵敏度高的优点。The specific types of reagents in the present invention are not limited, and the above-mentioned primers and probes may be included, which can be used for influenza virus H5, H7, and H9 subtypes. Since the above-mentioned reagents contain the above-mentioned primers and probes, they have the advantages of strong specificity and high sensitivity.
第四方面,本发明提供了一种数字PCR试剂盒,包括所述的引物和探针以及数字PCR反应预混液。In a fourth aspect, the present invention provides a digital PCR kit, including the primers and probes and a digital PCR reaction master mix.
在一些优选的实施方式中,所述数字PCR反应预混液,包括:Buffer、Mg2+、dNTPs、热启动Taq酶和逆转录酶。In some preferred embodiments, the digital PCR reaction master mix includes: Buffer, Mg 2+ , dNTPs, hot-start Taq enzyme and reverse transcriptase.
将本发明提供的数字PCR试剂盒采用微滴数字PCR的方式,能够实现对H5/H7/H9亚型病毒样本的精准检测,以达到对流感病毒进行早期鉴别诊断以及疫情监测的目的,从而为流感病毒诊断提供一种新方法。The digital PCR kit provided by the present invention adopts the method of droplet digital PCR, which can realize the accurate detection of H5/H7/H9 subtype virus samples, so as to achieve the purpose of early differential diagnosis and epidemic monitoring of influenza virus, so as to provide Influenza virus diagnosis provides a new method.
在一些优选的实施方式中,还包括阳性对照品,用于监控检测样本的均一性,保证检测结果的可比性。In some preferred embodiments, a positive control substance is also included, which is used to monitor the homogeneity of the test samples and ensure the comparability of the test results.
本发明中,阳性对照品包括H5亚型H基因片段、H7亚型H基因片段和H9亚型H基因片段。In the present invention, the positive controls include H5 subtype H gene fragments, H7 subtype H gene fragments and H9 subtype H gene fragments.
在一些优选的实施方式中,所述阳性对照品为含有H5亚型H基因片段、H7亚型H基因片段和H9亚型H基因片段的重组质粒。In some preferred embodiments, the positive control substance is a recombinant plasmid containing H5 subtype H gene fragment, H7 subtype H gene fragment and H9 subtype H gene fragment.
在一些优选的实施方式中,还包括阴性对照品,用于监控检测试剂是否被污染。In some preferred embodiments, a negative control is also included to monitor whether the detection reagent is contaminated.
本发明中,阴性对照品为无核酸酶水。In the present invention, the negative control substance is nuclease-free water.
下面通过具体的实施例和对比例进一步说明本发明,但是,应当理解为,这些实施例仅仅是用于更详细地说明之用,而不应理解为用于以任何形式限制本发明。The present invention is further described below through specific examples and comparative examples, however, it should be understood that these examples are only used for more detailed description, and should not be construed to limit the present invention in any form.
实施例1Example 1
一种检测流感病毒H5、H7、H9亚型的引物和探针,包括第一引物对H5-F、H5-R;第二引物对H7-F、H7-R;第三引物对H9-F、H9-R;第一探针H5-P;第二探针H7-P;第三探针H9-P,引物和探针的序列如表1所示。A primer and probe for detecting influenza virus H5, H7, H9 subtypes, comprising the first primer pair H5-F, H5-R; the second primer pair H7-F, H7-R; the third primer pair H9-F , H9-R; the first probe H5-P; the second probe H7-P; the third probe H9-P, the sequences of primers and probes are shown in Table 1.
表1Table 1
对比例1-1~对比例1-3Comparative Example 1-1 to Comparative Example 1-3
对比例1-1对比例1-3分别提供了一种检测流感病毒H5的引物和探针,引物和探针的序列如表2所示。Comparative Examples 1-1 Comparative Examples 1-3 provide a primer and a probe for detecting influenza virus H5, respectively, and the sequences of the primers and probes are shown in Table 2.
表2Table 2
对比例2-1~对比例2-3Comparative Example 2-1 to Comparative Example 2-3
对比例2-1~对比例2-3分别提供了一种检测流感病毒H7的引物和探针,引物和探针的序列如表3所示。Comparative Examples 2-1 to 2-3 respectively provide a primer and a probe for detecting influenza virus H7, and the sequences of the primers and probes are shown in Table 3.
表3table 3
对比例3-1~对比例3-3Comparative Example 3-1 to Comparative Example 3-3
对比例3-1~对比例3-3分别提供了一种检测流感病毒H9的引物和探针,引物和探针的序列如表4所示。Comparative Examples 3-1 to 3-3 respectively provide a primer and a probe for detecting influenza virus H9, and the sequences of the primers and probes are shown in Table 4.
表4Table 4
实施例2Example 2
一种试剂盒,包括实施例1的引物和探针、反应预混液、阳性对照品和阴性对照品。其中反应预混液为:qScript XLT One-Step RT-qPCR ToughMix(QuantaBio)。A kit comprising the primers and probes of Example 1, a reaction master mix, a positive control substance and a negative control substance. The reaction master mix is: qScript XLT One-Step RT-qPCR ToughMix (QuantaBio).
阳性对照品:含有H5亚型H基因片段、H7亚型H基因片段、H9亚型H基因片段的重组质粒。Positive control substance: recombinant plasmid containing H5 subtype H gene fragment, H7 subtype H gene fragment, and H9 subtype H gene fragment.
阴性对照品:无核酸酶水。Negative control: nuclease-free water.
试验例1引物探针筛选Test Example 1 Primer Probe Screening
1.扩增方法1. Amplification method
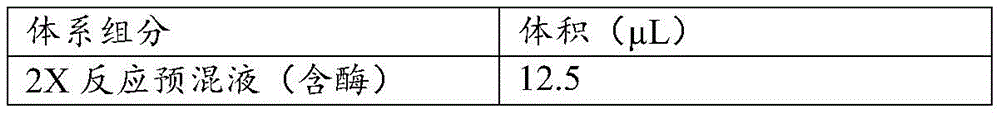
病毒核酸提取采用磁珠法,使用杭州博日科技股份有限公司生产的病毒核酸提取试剂盒对流感病毒样本进行核酸提取。按如表5配制数字PCR反应体系,总体积25ul。Viral nucleic acid extraction was carried out by magnetic bead method, and the nucleic acid was extracted from influenza virus samples using the viral nucleic acid extraction kit produced by Hangzhou Bioer Technology Co., Ltd. Prepare a digital PCR reaction system as shown in Table 5, with a total volume of 25ul.
表5table 5
数字PCR反应digital PCR reaction
微滴生成:Droplet Generation:
将25μL反应体系加入到微滴反应芯片中,盖上盖子,转移至Naica自动化微滴芯片数字PCR仪器中,同时打开压力泵调至数值1.28左右,压力增加到大气压+1000mbar生成微滴。Add 25 μL of the reaction system to the droplet reaction chip, close the lid, and transfer it to the Naica automated droplet chip digital PCR instrument. At the same time, turn on the pressure pump and adjust the value to about 1.28, and increase the pressure to atmospheric pressure + 1000 mbar to generate droplets.
PCR扩增:PCR amplification:
微滴生成后进入PCR扩增程序,如表6所示。After the droplets are generated, the PCR amplification program is entered, as shown in Table 6.
表6Table 6
扩增结束后,取出芯片放入微滴读取仪,进行数据读取。After the amplification, take out the chip and put it into the droplet reader for data reading.
2.引物探针筛选2. Primer Probe Screening
分别提取流感病毒H5、H7、H9亚型的核酸,对于流感病毒H5亚型核酸,分别以实施例1中第一引物对和第一探针、对比例1-1~对比例1-3的引物探针按照上述扩增方法进行扩增,结果如图1所示。(图1曲线从左至右依次为实施例1、对比例1-1、对比例1-2、对比例1-3的扩增曲线)。The nucleic acids of influenza virus H5, H7, and H9 subtypes were extracted respectively. For influenza virus H5 subtype nucleic acid, the first primer pair and the first probe in Example 1, and the samples of Comparative Examples 1-1 to 1-3 were used respectively. The primer probes were amplified according to the above-mentioned amplification method, and the results are shown in Figure 1 . (The curves in Figure 1 are the amplification curves of Example 1, Comparative Example 1-1, Comparative Example 1-2, and Comparative Example 1-3 in order from left to right).
对于流感病毒H7亚型核酸,分别以实施例1中第二引物对和第二探针、对比例2-1~对比例2-3的引物探针按照上述扩增方法进行扩增,结果如图2所示。(图2曲线从左至右依次为实施例1、对比例2-1、对比例2-2、对比例2-3的扩增曲线)。For influenza virus H7 subtype nucleic acid, the second primer pair and the second probe in Example 1, and the primer probes of Comparative Examples 2-1 to 2-3 were amplified according to the above-mentioned amplification method, and the results were as follows: shown in Figure 2. (The curves of FIG. 2 are the amplification curves of Example 1, Comparative Example 2-1, Comparative Example 2-2, and Comparative Example 2-3 in order from left to right).
对于流感病毒H9亚型核酸,分别以实施例1中第三引物对和第三探针、对比例3-1~对比例3-3的引物探针按照上述扩增方法进行扩增,结果如图3所示。(图3曲线从左至右依次为实施例1、对比例3-1、对比例3-2、对比例3-3的扩增曲线)。For influenza virus H9 subtype nucleic acid, the third primer pair and the third probe in Example 1, and the primer probes of Comparative Examples 3-1 to 3-3 were amplified according to the above-mentioned amplification method, and the results were as follows: shown in Figure 3. (The curves in FIG. 3 are the amplification curves of Example 1, Comparative Example 3-1, Comparative Example 3-2, and Comparative Example 3-3 in order from left to right).
制备流感病毒H5、H7、H9亚型的混合核酸,对于流感病毒H5、H7、H9亚型的混合核酸,采用实施例1提供的引物和探针按照上述扩增方法进行扩增,混合核酸浓度依次稀释10-1,10-2,10-3,10-4,10-5,结果如图4所示。Prepare the mixed nucleic acid of influenza virus H5, H7, H9 subtype, for the mixed nucleic acid of influenza virus H5, H7, H9 subtype, adopt the primers and probes provided in Example 1 to amplify according to the above-mentioned amplification method, and the mixed nucleic acid concentration Dilute 10 -1 , 10 -2 , 10 -3 , 10 -4 , and 10 -5 in turn, and the results are shown in Figure 4 .
从图中可以看出,H5、H7、H9亚型引物和探针组合后的扩增曲线相互不影响,并且荧光强度高,曲线呈S型,说明筛选出来的引物对和探针非常好。It can be seen from the figure that the amplification curves of H5, H7, and H9 subtype primers and probes combined do not affect each other, and the fluorescence intensity is high, and the curve is S-shaped, indicating that the selected primer pairs and probes are very good.
试验例2数字PCR特异性实验Test Example 2 Digital PCR Specificity Experiment
选择与流感病毒核酸序列具有同源性,引起相同或相似临床症状的呼吸道病毒甲型H1N1流感病毒、甲型H3N2流感病毒、甲型H5N1流感病毒、甲型H7N9流感病毒、甲型H9N2流感病毒,乙型流感病毒,新型冠状病毒,同时设置阳性对照和阴性对照。分别采用实施例1提供的引物和探针进行检测,结果如图5、图6和图7所示(图5中,横坐标分别代表:1.H5N1,2.H1N1,3.H3N2,4.H7N9,5H9N2,6.乙型流感病毒,7新型冠状病毒,8阴性对照;图6中,横坐标分别代表:1.H7N9,2.H1N1,3.H3N2,4.H5N1,5H9N2,6.乙型流感病毒,7新型冠状病毒,8阴性对照;图7中,横坐标分别代表:1.H9N2,2.H1N1,3.H3N2,4.H5N1,5H7N9,6.乙型流感病毒,7新型冠状病毒,8阴性对照),从图中可以看出,仅能检测出H5、H7、H9,其它各种模板均为阴性,表明本发明的检测方法与其它病毒没有交叉反应,具有较强的特异性。Select influenza A/H1N1 influenza virus, A/H3N2 influenza virus, A/H5N1 influenza virus, A/H7N9 influenza virus, and A/H9N2 influenza virus that have homology to the nucleic acid sequence of influenza virus and cause the same or similar clinical symptoms. Influenza B virus, novel coronavirus, positive control and negative control are set at the same time. The primers and probes provided in Example 1 were used for detection respectively, and the results were shown in Figure 5, Figure 6 and Figure 7 (in Figure 5, the abscissas represent: 1.H5N1, 2.H1N1, 3.H3N2, 4.5. H7N9, 5H9N2, 6. Influenza B virus, 7 new coronaviruses, 8 negative controls; in Figure 6, the abscissas represent: 1.H7N9, 2.H1N1, 3.H3N2, 4.H5N1, 5H9N2, 6.B Type influenza virus, 7 novel coronavirus, 8 negative control; in Figure 7, the abscissas represent: 1.H9N2, 2.H1N1, 3.H3N2, 4.H5N1, 5H7N9, 6. Type B influenza virus, 7 novel coronavirus virus, 8 negative control), as can be seen from the figure, only H5, H7, H9 can be detected, and other various templates are negative, indicating that the detection method of the present invention does not cross-react with other viruses, and has strong specificity sex.
试验例3数字PCR灵敏度实验Test Example 3 Digital PCR Sensitivity Experiment
采用流感病毒H5、H7、H9亚型病毒阳性样品依次进行4倍梯度稀释,稀释至4096倍,分别以实施例1提供的引物和探针,采用试验例1给出的检测方法进行检测,结果如图8、图9和图10所示(图8中,横坐标分别代表:1.H5N1核酸,2.H5N1核酸稀释4倍,3.H5N1核酸稀释16倍,4.H5N1核酸稀释64倍,5.H5N1核酸稀释256倍,6.H5N1核酸稀释1024倍,7.H5N1核酸稀释4096倍;图9中,横坐标分别代表:1.H7N9核酸,2.H7N9核酸稀释4倍,3.H7N9核酸稀释16倍,4.H7N9核酸稀释64倍,5.H7N9核酸稀释256倍,6.H7N9核酸稀释1024倍,7.H7N9核酸稀释4096倍;图10中,横坐标分别代表:1.H9N2核酸,2.H9N2核酸稀释4倍,3.H9N2核酸稀释16倍,4.H9N2核酸稀释64倍,5.H9N2核酸稀释256倍,6.H9N2核酸稀释1024倍,7.H9N2核酸稀释4096倍)。结果显示本体系H5亚型在1拷贝的模板量下,可以生产稳定的扩增(图8中的编号6)。H7亚型在2拷贝的模板量下,可以生产稳定的扩增(图9中的编号6)。H9亚型在1拷贝的模板量下,可以生产稳定的扩增(图10中的编号6)。The positive samples of influenza virus H5, H7, and H9 subtypes were used for 4-fold gradient dilution successively, and the dilution was 4096 times. As shown in Figure 8, Figure 9 and Figure 10 (in Figure 8, the abscissas represent: 1. H5N1 nucleic acid, 2. H5N1 nucleic acid diluted 4 times, 3. H5N1 nucleic acid diluted 16 times, 4. H5N1 nucleic acid diluted 64 times, 5. H5N1 nucleic acid diluted 256 times, 6. H5N1 nucleic acid diluted 1024 times, 7. H5N1 nucleic acid diluted 4096 times; in Figure 9, the abscissas represent: 1. H7N9 nucleic acid, 2. H7N9 nucleic acid diluted 4 times, 3. H7N9 nucleic acid Dilute 16 times, 4.H7N9 nucleic acid is diluted 64 times, 5.H7N9 nucleic acid is diluted 256 times, 6.H7N9 nucleic acid is diluted 1024 times, and 7.H7N9 nucleic acid is diluted 4096 times; in Figure 10, the abscissas represent: 1.H9N2 nucleic acid, 2. H9N2 nucleic acid diluted 4 times, 3. H9N2 nucleic acid diluted 16 times, 4. H9N2 nucleic acid diluted 64 times, 5. H9N2 nucleic acid diluted 256 times, 6. H9N2 nucleic acid diluted 1024 times, 7. H9N2 nucleic acid diluted 4096 times). The results show that the H5 subtype of this system can produce stable amplification at a template amount of 1 copy (No. 6 in FIG. 8 ). The H7 subtype produced stable amplification at a template amount of 2 copies (No. 6 in Figure 9). The H9 subtype can produce stable amplification at a template amount of 1 copy (No. 6 in Figure 10).
最后应说明的是:以上各实施例仅用以说明本发明的技术方案,而非对其限制;尽管参照前述各实施例对本发明进行了详细的说明,本领域的普通技术人员应当理解:其依然可以对前述各实施例所记载的技术方案进行修改,或者对其中部分或者全部技术特征进行等同替换;而这些修改或者替换,并不使相应技术方案的本质脱离本发明各实施例技术方案的范围。Finally, it should be noted that the above embodiments are only used to illustrate the technical solutions of the present invention, but not to limit them; although the present invention has been described in detail with reference to the foregoing embodiments, those of ordinary skill in the art should understand that: The technical solutions described in the foregoing embodiments can still be modified, or some or all of the technical features thereof can be equivalently replaced; and these modifications or replacements do not make the essence of the corresponding technical solutions deviate from the technical solutions of the embodiments of the present invention. scope.
SEQUENCE LISTINGSEQUENCE LISTING
<110> 杭州博日科技股份有限公司<110> Hangzhou Bioer Technology Co., Ltd.
<120> 检测流感病毒H5、H7、H9亚型的引物和探针及其应用、试剂和数字PCR试剂<120> Primers and probes for detection of influenza virus H5, H7, H9 subtypes and their applications, reagents and digital PCR reagents
盒box
<160> 36<160> 36
<170> PatentIn version 3.5<170> PatentIn version 3.5
<210> 1<210> 1
<211> 27<211> 27
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 1<400> 1
gaatcaatag gaacttacca aatactg 27gaatcaatag gaacttacca aatactg 27
<210> 2<210> 2
<211> 21<211> 21
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 2<400> 2
ctgcattgta acgacccatt g 21ctgcattgta acgacccatt g 21
<210> 3<210> 3
<211> 24<211> 24
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 3<400> 3
ctaattgatg gttggtatgg tttc 24ctaattgatg gttggtatgg tttc 24
<210> 4<210> 4
<211> 21<211> 21
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 4<400> 4
aattgccgat tgagtgcttt t 21aattgccgat tgagtgcttt t 21
<210> 5<210> 5
<211> 24<211> 24
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 5<400> 5
ctggaatctg aagrractta caaa 24ctggaatctg aagrractta caaa 24
<210> 6<210> 6
<211> 21<211> 21
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 6<400> 6
agaacaaraa ggcagcaaac c 21agaacaaraa ggcagcaaac c 21
<210> 7<210> 7
<211> 24<211> 24
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 7<400> 7
tttattcaac agtggcgagt tccc 24tttattcaac agtggcgagt tccc 24
<210> 8<210> 8
<211> 27<211> 27
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 8<400> 8
cagaatgcac agggagaggg aactgct 27cagaatgcac agggagaggg aactgct 27
<210> 9<210> 9
<211> 23<211> 23
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 9<400> 9
achatttatt cgactgtygc ctc 23achatttatt cgactgtygc ctc 23
<210> 10<210> 10
<211> 20<211> 20
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 10<400> 10
gctaacccag ycaatgayct 20
<210> 11<210> 11
<211> 25<211> 25
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 11<400> 11
catacyacat ttctaaaraa ggagg 25catacyacat ttctaaaraa ggagg 25
<210> 12<210> 12
<211> 26<211> 26
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 12<400> 12
tacccaggga acctcaatga ctatga 26tacccaggga acctcaatga ctatga 26
<210> 13<210> 13
<211> 20<211> 20
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 13<400> 13
ggcaasstgg aagaatggat 20
<210> 14<210> 14
<211> 21<211> 21
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 14<400> 14
tgacacttgg tgttgcagtt g 21tgacacttgg tgttgcagtt g 21
<210> 15<210> 15
<211> 26<211> 26
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 15<400> 15
tgatgcaatc cacttcgaga gtaatg 26tgatgcaatc cacttcgaga gtaatg 26
<210> 16<210> 16
<211> 23<211> 23
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 16<400> 16
tagtaatgaa caggggagtg ggt 23tagtaatgaa caggggagtg ggt 23
<210> 17<210> 17
<211> 23<211> 23
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 17<400> 17
agtccagrca tctaggaatc cgt 23agtccagrca tctaggaatc cgt 23
<210> 18<210> 18
<211> 27<211> 27
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 18<400> 18
agaatccacc caaaaggcaa tagatgg 27agaatccacc caaaaggcaa tagatgg 27
<210> 19<210> 19
<211> 23<211> 23
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 19<400> 19
gaaaraggag tggaagtcgt caa 23gaaaraggag tggaagtcgt caa 23
<210> 20<210> 20
<211> 22<211> 22
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 20<400> 20
cacttccttc tckcctctca at 22cacttccttc tckcctctca at 22
<210> 21<210> 21
<211> 25<211> 25
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 21<400> 21
caactgaaac rgtggaacga acaaa 25caactgaaac rgtggaacga acaaa 25
<210> 22<210> 22
<211> 22<211> 22
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 22<400> 22
gagtggaaac aaamtggtga ca 22
<210> 23<210> 23
<211> 20<211> 20
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 23<400> 23
tgaaagcycc attgaaactg 20
<210> 24<210> 24
<211> 25<211> 25
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 24<400> 24
aacaatcttt ygtaccgagt ccagg 25aacaatcttt ygtaccgagt ccagg 25
<210> 25<210> 25
<211> 23<211> 23
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 25<400> 25
aarttcgtga atgaagaarc tct 23aarttcgtga atgaagaarc tct 23
<210> 26<210> 26
<211> 22<211> 22
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 26<400> 26
gggaatgyag cattatctgt gt 22
<210> 27<210> 27
<211> 24<211> 24
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 27<400> 27
cagagartca ggcggaatyg acaa 24cagagartca ggcggaatyg acaa 24
<210> 28<210> 28
<211> 23<211> 23
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 28<400> 28
agagahtcma cycaaaaggc aat 23agagahtcma cycaaaaggc aat 23
<210> 29<210> 29
<211> 25<211> 25
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 29<400> 29
atttcatayt gcttgttcat tttgt 25atttcatayt gcttgttcat tttgt 25
<210> 30<210> 30
<211> 28<211> 28
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 30<400> 30
ataacatcca aagtgaataa yatagtcg 28ataacatcca aagtgaataa yatagtcg 28
<210> 31<210> 31
<211> 25<211> 25
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 31<400> 31
tatatgggca tataatgcag aattg 25tatatgggca tataatgcag aattg 25
<210> 32<210> 32
<211> 25<211> 25
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 32<400> 32
catttrtggt atagctcgaa acatc 25catttrtggt atagctcgaa acatc 25
<210> 33<210> 33
<211> 25<211> 25
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 33<400> 33
ctrcttgaaa accagaarac actcg 25ctrcttgaaa accagaarac actcg 25
<210> 34<210> 34
<211> 24<211> 24
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 34<400> 34
ataagrttga tgatcaaaty carg 24ataagrttga tgatcaaaty carg 24
<210> 35<210> 35
<211> 22<211> 22
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 35<400> 35
cgagtgtytt ctggttttca ag 22cgagtgtytt ctggttttca
<210> 36<210> 36
<211> 27<211> 27
<212> DNA<212> DNA
<213> 人工序列<213> Artificial sequences
<400> 36<400> 36
atatatgggc atataatgca gaattgc 27atatatgggc atataatgca gaattgc 27
Claims (10)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202210770639.2A CN114959121A (en) | 2022-06-30 | 2022-06-30 | Primers and probes for detecting influenza virus subtypes H5, H7 and H9, and application, reagent and digital PCR kit thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202210770639.2A CN114959121A (en) | 2022-06-30 | 2022-06-30 | Primers and probes for detecting influenza virus subtypes H5, H7 and H9, and application, reagent and digital PCR kit thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN114959121A true CN114959121A (en) | 2022-08-30 |
Family
ID=82968353
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202210770639.2A Pending CN114959121A (en) | 2022-06-30 | 2022-06-30 | Primers and probes for detecting influenza virus subtypes H5, H7 and H9, and application, reagent and digital PCR kit thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN114959121A (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN116676421A (en) * | 2023-01-31 | 2023-09-01 | 四川大学华西医院 | A primer probe composition, kit and detection method for synchronous detection of influenza virus |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN107937611A (en) * | 2017-12-18 | 2018-04-20 | 北京卓诚惠生生物科技股份有限公司 | Detect the primed probe group of avian influenza virus subtype H5, H7 and H9 |
| CN111254222A (en) * | 2020-03-12 | 2020-06-09 | 清华大学深圳国际研究生院 | Influenza virus hemagglutinin subtype typing primer, kit and typing method |
| CN111876486A (en) * | 2020-08-13 | 2020-11-03 | 领航基因科技(杭州)有限公司 | BCR-ABL1 fusion gene three subtype typing detection kit |
| CN112534056A (en) * | 2018-06-27 | 2021-03-19 | 麦迪卡格公司 | Influenza virus hemagglutinin mutants |
| CN112626267A (en) * | 2020-11-25 | 2021-04-09 | 扬州大学 | Multiplex fluorescent RT-PCR primer probe set and kit for detecting avian influenza virus H5, H7 and H9 subtypes |
-
2022
- 2022-06-30 CN CN202210770639.2A patent/CN114959121A/en active Pending
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN107937611A (en) * | 2017-12-18 | 2018-04-20 | 北京卓诚惠生生物科技股份有限公司 | Detect the primed probe group of avian influenza virus subtype H5, H7 and H9 |
| CN112534056A (en) * | 2018-06-27 | 2021-03-19 | 麦迪卡格公司 | Influenza virus hemagglutinin mutants |
| CN111254222A (en) * | 2020-03-12 | 2020-06-09 | 清华大学深圳国际研究生院 | Influenza virus hemagglutinin subtype typing primer, kit and typing method |
| CN111876486A (en) * | 2020-08-13 | 2020-11-03 | 领航基因科技(杭州)有限公司 | BCR-ABL1 fusion gene three subtype typing detection kit |
| CN112626267A (en) * | 2020-11-25 | 2021-04-09 | 扬州大学 | Multiplex fluorescent RT-PCR primer probe set and kit for detecting avian influenza virus H5, H7 and H9 subtypes |
Non-Patent Citations (1)
| Title |
|---|
| FAN YANG等: "A multiplex real-time RT-PCR method for detecting H5, H7 and H9 subtype avian influenza viruses in field and clinical samples", VIRUS RESEARCH, vol. 309, 23 December 2021 (2021-12-23), pages 198669, XP086926719, DOI: 10.1016/j.virusres.2021.198669 * |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN116676421A (en) * | 2023-01-31 | 2023-09-01 | 四川大学华西医院 | A primer probe composition, kit and detection method for synchronous detection of influenza virus |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Mahony et al. | Molecular diagnosis of respiratory virus infections | |
| JP6329370B2 (en) | Simultaneous diagnosis kit for diseases caused by respiratory viruses | |
| CN103275862A (en) | Fluorescent quantitative reverse transcription-polymerase chain reaction (RT-PCR) kit for detecting influenza A virus subtype H7N9 | |
| JP7064473B2 (en) | How to detect influenza A virus and influenza B virus | |
| CN113005228B (en) | Detection kit for synchronously detecting multiple respiratory pathogens and detection method thereof | |
| CN112813195B (en) | Novel quantitative detection kit for coronavirus nucleic acid based on micro-droplet digital analysis | |
| CN102337351A (en) | Typing detection kit for influenza virus | |
| US20080261198A1 (en) | Diagnostic Primers and Method for Detecting Avian Influenza Virus Subtype H5 and H5n1 | |
| CN113564280A (en) | RAA primer for detecting 12 serotypes of avian adenovirus group I and detection method thereof | |
| CN110343784B (en) | Composition and kit for detection of quadruple influenza virus nucleic acid based on melting curve | |
| Zhang et al. | Development of a recombinase-aided amplification combined with a lateral flow dipstick assay for rapid detection of H7 subtype avian influenza virus | |
| CN114959121A (en) | Primers and probes for detecting influenza virus subtypes H5, H7 and H9, and application, reagent and digital PCR kit thereof | |
| CN113215329A (en) | Primer, probe and kit for multiplex PCR detection of 7 respiratory subtype influenza viruses | |
| CN103993102A (en) | Multiple fluorescent PCR method and kit for simultaneous detection of human adenovirus, human mycoplasma pneumonia and bocavirus | |
| CN112029904A (en) | Gene chip and kit for detecting novel coronavirus | |
| CN117448495A (en) | Newcastle disease virus virulent nucleic acid CRISPR-Cas13a detection system and RPA primer pair and crRNA | |
| CN110724763A (en) | Fluorescent quantitative PCR detection method for human adenovirus and bocavirus and application thereof | |
| CN104313191B (en) | Single stage method reverse transcription PCR detects and the primer of somatotype Ebola virus 5 kinds of hypotypes, probe and test kit | |
| CN112063757A (en) | A kind of primer, kit and application for detecting African swine fever virus | |
| CN117925914B (en) | Primers and probes for triple fluorescence quantitative RT-PCR detection of mink Aleutian, enteritis and canine distemper viruses and their application | |
| CN110257558A (en) | Primer combination of probe and kit for five kinds of Respirovirus detections | |
| CN115838837B (en) | Microdroplet double digital PCR (polymerase chain reaction) kit for detecting porcine delta coronavirus and porcine epidemic diarrhea virus | |
| CN109943667A (en) | Primers, probes, kits and applications for detection of soybean mosaic virus based on RPA technology | |
| CN114807432B (en) | Kit and method for rapidly detecting novel coronavirus and Delta mutant strain thereof | |
| CN104531900B (en) | Reagent set for identification or auxiliary identification of H6N1 subtype avian influenza virus and application thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination |