CN114805192B - Tricyclic XOR/URAT1 dual inhibitor containing 2-hydroxybenzoic acid and preparation method and application thereof - Google Patents
Tricyclic XOR/URAT1 dual inhibitor containing 2-hydroxybenzoic acid and preparation method and application thereof Download PDFInfo
- Publication number
- CN114805192B CN114805192B CN202210326622.8A CN202210326622A CN114805192B CN 114805192 B CN114805192 B CN 114805192B CN 202210326622 A CN202210326622 A CN 202210326622A CN 114805192 B CN114805192 B CN 114805192B
- Authority
- CN
- China
- Prior art keywords
- hydroxybenzoic acid
- pyridin
- xor
- urat1
- compound
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/60—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D213/62—Oxygen or sulfur atoms
- C07D213/63—One oxygen atom
- C07D213/65—One oxygen atom attached in position 3 or 5
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/12—Drugs for disorders of the urinary system of the kidneys
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/06—Antigout agents, e.g. antihyperuricemic or uricosuric agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/04—Inotropic agents, i.e. stimulants of cardiac contraction; Drugs for heart failure
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/12—Antihypertensives
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/60—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D213/72—Nitrogen atoms
- C07D213/74—Amino or imino radicals substituted by hydrocarbon or substituted hydrocarbon radicals
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02A—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE
- Y02A50/00—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE in human health protection, e.g. against extreme weather
- Y02A50/30—Against vector-borne diseases, e.g. mosquito-borne, fly-borne, tick-borne or waterborne diseases whose impact is exacerbated by climate change
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Public Health (AREA)
- Engineering & Computer Science (AREA)
- Cardiology (AREA)
- Rheumatology (AREA)
- Heart & Thoracic Surgery (AREA)
- Pain & Pain Management (AREA)
- Physical Education & Sports Medicine (AREA)
- Immunology (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Hospice & Palliative Care (AREA)
- Urology & Nephrology (AREA)
- Pyridine Compounds (AREA)
Abstract
Description
技术领域technical field
本发明属于XOR/URAT1双重抑制剂领域,具体涉及一种含2-羟基苯甲酸的三环类XOR/URAT1双重抑制剂及其制备方法与应用。The invention belongs to the field of XOR/URAT1 dual inhibitors, and specifically relates to a tricyclic XOR/URAT1 dual inhibitor containing 2-hydroxybenzoic acid and its preparation method and application.
背景技术Background technique
来自不同国家的流行病学研究表明,痛风的患病率和发病率正在增加,给人们带来了巨大的经济负担。高尿酸血症是痛风发生的重要危险因素,与肾功能不全、心血管疾病、高血压、高血脂、癌症、糖尿病等有关,高尿酸血症的发生是由于尿酸产量增加,肾功能尿酸排泄受损,或两者的结合。在高尿酸血症患者中,90%是由于肾脏排泄效率低下造成的。在人体中,90%的尿酸被肾近端小管大量重吸收回血液中,此重吸收过程主要依赖于人类尿酸盐转运体1(hURAT1,SLC22A12),其功能是调节血尿酸水平。通过抑制该转运体活性,可以促进尿酸排泄。另一方面,大约10%的病例中是由于内源性尿酸过量产生导致的。在内源性尿酸的生成途径中,关键酶是黄嘌呤氧化还原酶(XOR),该酶可以将次黄嘌呤氧化为黄嘌呤进而氧化为尿酸。XOR是早期研究高尿酸血症和痛风的病理机制和药物作用重要靶点之一。Epidemiological studies from different countries have shown that the prevalence and incidence of gout are increasing, causing a huge economic burden on people. Hyperuricemia is an important risk factor for gout, and it is related to renal insufficiency, cardiovascular disease, hypertension, hyperlipidemia, cancer, diabetes, etc. loss, or a combination of both. In patients with hyperuricemia, 90% are due to inefficient renal excretion. In the human body, 90% of uric acid is reabsorbed back into the blood by the renal proximal tubule in large quantities. This reabsorption process mainly depends on the human urate transporter 1 (hURAT1, SLC22A12), whose function is to regulate blood uric acid levels. By inhibiting the activity of this transporter, uric acid excretion can be promoted. On the other hand, in about 10% of cases it is due to overproduction of endogenous uric acid. In the pathway of endogenous uric acid production, the key enzyme is xanthine oxidoreductase (XOR), which can oxidize hypoxanthine to xanthine and then to uric acid. XOR is one of the important targets for early research on the pathological mechanism and drug action of hyperuricemia and gout.
近40年来,降尿酸药物的开发主要围绕XOR及URAT1开展。最具代表的有三个新药上市:非布索坦(2009年美国上市,XOR抑制剂)、托匹司他(2013年日本上市,XOR抑制剂)、和lesinurad(2015年美国上市,URAT1抑制剂)。但是,关注度最高的非布索坦仍未解决XOR抑制剂类药物用于部分患者效果差的问题(约20-30%的高尿酸血症患者对非布索坦不响应),且有可能增加心血管事件发生风险,2019年已被FDA要求增加黑框警告;另一方面,lesinurad单独使用虽降尿酸效果显著,但安全窗口狭窄,易致肾损伤,而与lesinurad同期开发上市的(lesinurad+别嘌醇)复方则在临床中表现优异,其对别嘌醇不耐受及疗效不佳患者均获得很好的应答率和疗效。因此,同时具有抑制尿酸生成与促进尿酸排泄作用的降尿酸药物可能成为该研究领域的热点,具有更显著的临床效果与应用前景。辉瑞公司开发的XOR/URAT1双重抑制剂PF-06743649对两个靶点的IC50分别为1.6nM和0.237μM。I期临床中,患者口服40mg/天,两周后尿酸水平降低69%,进一步证实XOR与URAT1的联合抑制是快速降低体内尿酸的有效策略。日本桔生制药公司研发的XOR/URAT1双重抑制剂KUX-1151已进入II期临床。Relburn-Metabolomics公司开发的XOR/URAT1双重抑制剂RLBN-1001及其系列类似物分别处于I期临床及临床前研究阶段。RLBN1001是一种癌症药物的原型,在患者使用过程中发现可引起低尿酸血症,临床研究了50例服用RLBN1001及其类似物之后所造成的低尿酸血症发现该类药物是通过抑制URAT1/XOR而引起尿酸排泄增加。In the past 40 years, the development of uric acid-lowering drugs has mainly focused on XOR and URAT1. The most representative three new drugs are listed: febuxostat (launched in the United States in 2009, XOR inhibitor), topinastat (launched in Japan in 2013, XOR inhibitor), and lesinurad (launched in the United States in 2015, URAT1 inhibitor ). However, febuxostat, which has received the most attention, has not yet solved the problem of poor efficacy of XOR inhibitors in some patients (about 20-30% of patients with hyperuricemia do not respond to febuxostat), and it is possible In 2019, the FDA requested a black box warning to increase the risk of cardiovascular events; on the other hand, although lesinurad used alone has a significant effect on lowering uric acid, the safety window is narrow and it is easy to cause kidney damage, while lesinurad (lesinurad+ Allopurinol) compound has excellent clinical performance, and it has achieved good response rate and curative effect for patients with allopurinol intolerance and poor curative effect. Therefore, uric acid-lowering drugs that can both inhibit uric acid production and promote uric acid excretion may become a hot spot in this research field, with more significant clinical effects and application prospects. The XOR/URAT1 dual inhibitor PF-06743649 developed by Pfizer has IC 50 of 1.6nM and 0.237μM for the two targets, respectively. In the Phase I clinical trial, the patient took 40 mg/day orally, and the uric acid level decreased by 69% after two weeks, further confirming that the combined inhibition of XOR and URAT1 is an effective strategy for rapidly reducing uric acid in the body. The XOR/URAT1 dual inhibitor KUX-1151 developed by Japan Orange Pharmaceutical Company has entered phase II clinical trials. The XOR/URAT1 dual inhibitor RLBN-1001 and its series of analogues developed by Relburn-Metabolomics are in Phase I clinical and preclinical research stages respectively. RLBN1001 is a prototype of a cancer drug, which was found to cause hypouricemia during the use of patients. A clinical study of 50 cases of hypouricemia caused by taking RLBN1001 and its analogues found that this type of drug inhibits URAT1/ XOR increases uric acid excretion.
目前,XOR/URAT1双重抑制剂总体研究尚处于开发初期,需要大量的前期摸索与积累。特别的,PF-06743649的体外活性较非布索坦和苯溴马隆,这两种单靶点抑制剂明显提高,进而临床效果强于目前可用的临床联合给药,且尿酸达标时间短,但是,这也可能是其I期临床中发现肾损伤的原因。总之,开发新结构类型的XOR/URAT1双重抑制剂对于缓解临床用药需要有重要的现实意义。At present, the overall research on XOR/URAT1 dual inhibitors is still in the early stages of development, requiring a lot of preliminary exploration and accumulation. In particular, the in vitro activity of PF-06743649 is significantly higher than that of febuxostat and benzbromarone, two single-target inhibitors, and the clinical effect is stronger than the currently available clinical combination administration, and the time for uric acid to reach the target is shorter. However, this may also be the reason for the kidney injury found in its phase I clinical trial. In conclusion, the development of new structural types of XOR/URAT1 dual inhibitors has important practical significance for alleviating the need for clinical medication.
发明内容Contents of the invention
针对以上现有技术存在的缺点和不足之处,本发明的首要目的在于提供一种含2-羟基苯甲酸的三环类XOR/URAT1双重抑制剂。Aiming at the shortcomings and deficiencies of the above prior art, the primary purpose of the present invention is to provide a tricyclic XOR/URAT1 dual inhibitor containing 2-hydroxybenzoic acid.
本发明的另一目的在于提供上述含2-羟基苯甲酸的三环类XOR/URAT1双重抑制剂的制备方法。Another object of the present invention is to provide a preparation method of the above-mentioned tricyclic XOR/URAT1 dual inhibitor containing 2-hydroxybenzoic acid.
本发明的再一目的在于提供上述含2-羟基苯甲酸的三环类XOR/URAT1双重抑制剂在制备预防和治疗与XOR/URAT1相关的疾病的药物中的应用。Another object of the present invention is to provide the application of the above-mentioned tricyclic XOR/URAT1 dual inhibitor containing 2-hydroxybenzoic acid in the preparation of medicines for preventing and treating diseases related to XOR/URAT1.
本发明目的通过以下技术方案实现。The purpose of the present invention is achieved through the following technical solutions.
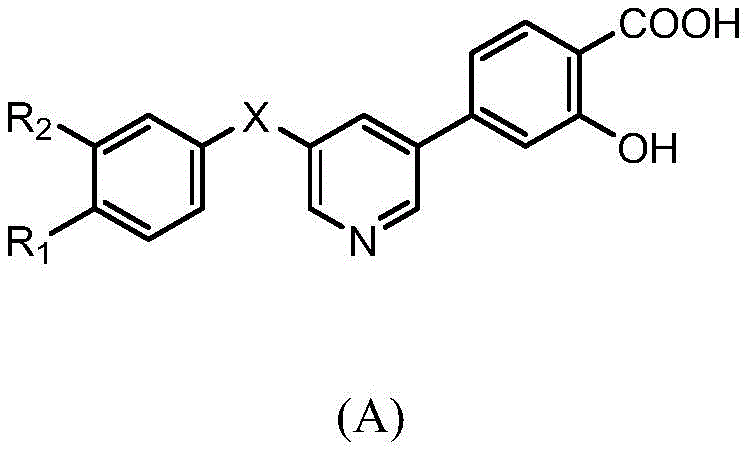
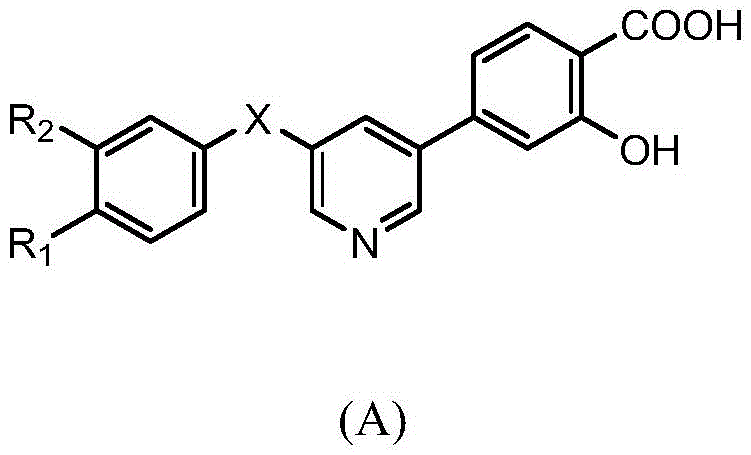
一种含2-羟基苯甲酸的三环类XOR/URAT1双重抑制剂,结构如式(A)所示:A tricyclic XOR/URAT1 dual inhibitor containing 2-hydroxybenzoic acid, the structure of which is shown in formula (A):
其中,X为O或NH,R1为烷基、烷氧基、卤素或氰基,R2为烷基、烷氧基、卤素或氰基。Wherein, X is O or NH, R 1 is alkyl, alkoxy, halogen or cyano, R 2 is alkyl, alkoxy, halogen or cyano.
优选的,所述X为O或NH,R1为C1-C7的烷基、OCH3、F、Cl、Br或CN,R2为C1-C7的烷基、OCH3、F、Cl、Br或CN。Preferably, X is O or NH, R 1 is C1-C7 alkyl, OCH 3 , F, Cl, Br or CN, R 2 is C1-C7 alkyl, OCH 3 , F, Cl, Br or CN.
优选的,所述抑制剂为以下任一项的化合物:Preferably, the inhibitor is any one of the following compounds:
4-(5-苯氧基吡啶-3-基)2-羟基苯甲酸4-(5-phenoxypyridin-3-yl)2-hydroxybenzoic acid
4-(5-(4-氯苯氧基)吡啶-3-基)-2-羟基苯甲酸4-(5-(4-chlorophenoxy)pyridin-3-yl)-2-hydroxybenzoic acid
4-(5-(对甲苯氧基)吡啶-3-基)2-羟基苯甲酸4-(5-(p-Tolyloxy)pyridin-3-yl)2-hydroxybenzoic acid
4-(5-(4-(叔丁基)苯氧基)吡啶-3-基)-2-羟基苯甲酸4-(5-(4-(tert-butyl)phenoxy)pyridin-3-yl)-2-hydroxybenzoic acid
4-(5-(3-氯苯氧基)吡啶-3-基)-2-羟基苯甲酸4-(5-(3-chlorophenoxy)pyridin-3-yl)-2-hydroxybenzoic acid
4-(5-(间甲苯氧基)吡啶-3-基)2-羟基苯甲酸4-(5-(m-methylphenoxy)pyridin-3-yl)2-hydroxybenzoic acid
4-(5-(苯氨基)吡啶-3-基)2-羟基苯甲酸4-(5-(phenylamino)pyridin-3-yl)2-hydroxybenzoic acid
4-(5-((4-氯苯基)氨基)吡啶-3-基)-2-羟基苯甲酸4-(5-((4-chlorophenyl)amino)pyridin-3-yl)-2-hydroxybenzoic acid
4-(5-((4-甲氧基苯基)氨基)吡啶-3-基)2-羟基苯甲酸4-(5-((4-methoxyphenyl)amino)pyridin-3-yl)2-hydroxybenzoic acid
4-(5-(对甲苯氨基)吡啶-3-基)2-羟基苯甲酸4-(5-(p-Tolylamino)pyridin-3-yl)2-hydroxybenzoic acid
4-(5-((4-(叔丁基)苯基)氨基)吡啶-3-基)-2-羟基苯甲酸4-(5-((4-(tert-butyl)phenyl)amino)pyridin-3-yl)-2-hydroxybenzoic acid
4-(5-((3-氯苯基)氨基)吡啶-3-基)-2-羟基苯甲酸4-(5-((3-chlorophenyl)amino)pyridin-3-yl)-2-hydroxybenzoic acid
4-(5-((3,4-二氯苯基)氨基)吡啶-3-基)-2-羟基苯甲酸4-(5-((3,4-dichlorophenyl)amino)pyridin-3-yl)-2-hydroxybenzoic acid
4-(5-((3-氟-4-甲基苯基)氨基)吡啶-3-基)-2-羟基苯甲酸。4-(5-((3-fluoro-4-methylphenyl)amino)pyridin-3-yl)-2-hydroxybenzoic acid.
以上任一项所述的一种含2-羟基苯甲酸的三环类XOR/URAT1双重抑制剂的制备方法,包括以下步骤:A method for preparing a tricyclic XOR/URAT1 dual inhibitor containing 2-hydroxybenzoic acid described in any one of the above, comprising the following steps:
(1)将3-羟基-5-溴吡啶与化合物I溶于有机溶剂中,加入有机碱,醋酸铜,常温搅拌,得化合物II,或将3-氨基-5-溴吡啶与化合物I溶于有机溶剂中,加入有机碱,醋酸铜,常温搅拌,得化合物III;(1) Dissolve 3-hydroxy-5-bromopyridine and compound I in an organic solvent, add an organic base, copper acetate, and stir at room temperature to obtain compound II, or dissolve 3-amino-5-bromopyridine and compound I in In an organic solvent, add an organic base and copper acetate, and stir at room temperature to obtain compound III;
(2)将化合物II与2-羟基-4-(4,4,5,5-四甲基-1,3,2-二氧苯甲醛-2-基)苯甲酸甲酯溶于1,4-二氧六环与水的混合溶剂中,加入钯催化剂,无机碱,氩气保护,回流反应得4-(5-取代-吡啶-3-苯氧基)-2-羟基苯甲酸类化合物,或将化合物III与2-羟基-4-(4,4,5,5-四甲基-1,3,2-二氧苯甲醛-2-基)苯甲酸甲酯溶于1,4-二氧六环与水的混合溶剂中,加入钯催化剂,无机碱,氩气保护,回流反应得4-(5-取代-吡啶-3-苯氨基)-2-羟基苯甲酸类化合物。(2) Compound II and 2-hydroxy-4-(4,4,5,5-tetramethyl-1,3,2-dioxobenzaldehyde-2-yl) methyl benzoate were dissolved in 1,4 -In the mixed solvent of dioxane and water, add palladium catalyst, inorganic base, argon protection, reflux reaction obtains 4-(5-substituted-pyridine-3-phenoxy)-2-hydroxybenzoic acid compound, Or compound III and 2-hydroxy-4-(4,4,5,5-tetramethyl-1,3,2-dioxobenzaldehyde-2-yl)methyl benzoate were dissolved in 1,4-bis In the mixed solvent of oxane and water, add palladium catalyst, inorganic base, argon protection, and reflux reaction to obtain 4-(5-substituted-pyridine-3-phenylamino)-2-hydroxybenzoic acid compounds.
优选的,步骤(1)中所述的有机溶剂是指二氯甲烷,有机碱是吡啶;步骤(2)中所述的无机碱是指碳酸钾;步骤(2)所用的钯催化剂为(1,1-双(二苯基膦)二茂铁)二氯化钯。Preferably, the organic solvent described in the step (1) refers to dichloromethane, and the organic base is pyridine; the inorganic base described in the step (2) refers to potassium carbonate; the palladium catalyst used in the step (2) is (1 , 1-bis(diphenylphosphino)ferrocene)palladium dichloride.
优选的,步骤(1)所述3-羟基-5-溴吡啶、化合物I、有机碱、醋酸铜的摩尔当量比为1:2:2-3:0.5-1,所述3-氨基-5-溴吡啶、化合物I、有机碱、醋酸铜的摩尔当量比为1:2:2-3:0.5-1。Preferably, the molar equivalent ratio of 3-hydroxyl-5-bromopyridine, compound I, organic base, and copper acetate in step (1) is 1:2:2-3:0.5-1, and the 3-amino-5 The molar equivalent ratio of bromopyridine, compound I, organic base and copper acetate is 1:2:2-3:0.5-1.
优选的,步骤(2)所述的化合物II、2-羟基-4-(4,4,5,5-四甲基-1,3,2-二氧苯甲醛-2-基)苯甲酸甲酯、无机碱、钯催化剂的摩尔当量比为1:1.5:3:0.03,所述化合物Ⅲ、2-羟基-4-(4,4,5,5-四甲基-1,3,2-二氧苯甲醛-2-基)苯甲酸甲酯、无机碱、钯催化剂的摩尔当量比为1:1.5:3:0.03。Preferably, the compound II described in step (2), 2-hydroxy-4-(4,4,5,5-tetramethyl-1,3,2-dioxylbenzaldehyde-2-yl)benzoic acid methyl The molar equivalent ratio of ester, inorganic base and palladium catalyst is 1:1.5:3:0.03, the compound III, 2-hydroxyl-4-(4,4,5,5-tetramethyl-1,3,2- The molar equivalent ratio of dioxybenzaldehyde-2-yl) methyl benzoate, inorganic base and palladium catalyst is 1:1.5:3:0.03.
以上任一项所述的一种含2-羟基苯甲酸的三环类XOR/URAT1双重抑制剂在制备预防和治疗与XOR/URAT1相关的疾病的药物中的应用。The use of a tricyclic XOR/URAT1 dual inhibitor containing 2-hydroxybenzoic acid described in any one of the above in the preparation of medicines for preventing and treating diseases related to XOR/URAT1.
优选的,所述药物为抗高尿酸血症或痛风药物。Preferably, the drug is an anti-hyperuricemia or gout drug.
优选的,所述抗高尿酸血症或痛风药物包括作为有效成分的含2-羟基苯甲酸的三环类XOR/URAT1双重抑制剂或其药学上可接受的盐和药学上可接受的载体。Preferably, the anti-hyperuricemia or gout drug comprises, as an active ingredient, a tricyclic XOR/URAT1 dual inhibitor containing 2-hydroxybenzoic acid or a pharmaceutically acceptable salt thereof and a pharmaceutically acceptable carrier.
与现有技术相比,本发明具有如下优点及有益效果:Compared with the prior art, the present invention has the following advantages and beneficial effects:
本发明的含2-羟基苯甲酸的三环类XOR/URAT1双重抑制剂具有与已知XOR/URAT1双重抑制剂不同的新颖的化学结构;本发明的抑制剂对与痛风有关的XOR和URAT1这两个关键作用靶点都表现出优良的抑制作用。因此,它们可以用于预防和治疗与XOR/URAT1相关的疾病,例如,高尿酸血症、心力衰竭、心血管疾病、高血压、肾疾病、炎症、关节病等。The tricyclic XOR/URAT1 dual inhibitor containing 2-hydroxybenzoic acid of the present invention has a novel chemical structure different from the known XOR/URAT1 dual inhibitor; Both key targets showed excellent inhibition. Therefore, they can be used to prevent and treat diseases related to XOR/URAT1, for example, hyperuricemia, heart failure, cardiovascular disease, hypertension, kidney disease, inflammation, arthropathy and the like.
附图说明Description of drawings
图1为本发明一种含2-羟基苯甲酸的三环类XOR/URAT1双重抑制剂的合成路线图。Fig. 1 is a synthetic route diagram of a tricyclic XOR/URAT1 dual inhibitor containing 2-hydroxybenzoic acid of the present invention.
具体实施方式Detailed ways
下面结合实施例对本发明作进一步详细的描述,但本发明的实施方式不限于此。The present invention will be further described in detail below in conjunction with examples, but the embodiments of the present invention are not limited thereto.
本发明一种含2-羟基苯甲酸的三环类XOR/URAT1双重抑制剂的合成路线图如图1所示。A synthetic route diagram of a tricyclic XOR/URAT1 dual inhibitor containing 2-hydroxybenzoic acid of the present invention is shown in FIG. 1 .
实施例1Example 1
4-(5-苯氧基吡啶-3-基)2-羟基苯甲酸(A1)的合成Synthesis of 4-(5-phenoxypyridin-3-yl)2-hydroxybenzoic acid (A1)
3-羟基-5-溴吡啶(1.4g,8.2mmol)与苯硼酸(2.0g,16.4mmol)溶于二氯甲烷中,加入吡啶(2.0mL,24.6mmol),醋酸铜(0.4g,2.2mmol),常温搅拌过夜,二氯甲烷萃取,无水硫酸钠干燥,乙酸乙酯:石油醚(体积比1:5)柱层析,得白色固体3-溴-5-苯氧基吡啶0.5g,产率22%。3-Hydroxy-5-bromopyridine (1.4g, 8.2mmol) and phenylboronic acid (2.0g, 16.4mmol) were dissolved in dichloromethane, pyridine (2.0mL, 24.6mmol), copper acetate (0.4g, 2.2mmol) were added ), stirred overnight at room temperature, extracted with dichloromethane, dried over anhydrous sodium sulfate, ethyl acetate:petroleum ether (volume ratio 1:5) column chromatography, obtained 0.5g of white solid 3-bromo-5-phenoxypyridine, Yield 22%.
3-溴-5-苯氧基吡啶(0.5g,1.6mmol)与2-羟基-4-(4,4,5,5-四甲基-1,3,2-二氧苯甲醛-2-基)苯甲酸甲酯(0.5g,1.9mmol)溶于1,4-二氧六环与水的混合液中(体积比为3:1),加入碳酸钾(0.7g,4.8mmol)、(1,1-双(二苯基磷)二茂铁)二氯化钯(0.04g,0.05mmol),置换氩气,回流搅拌。硅藻土过滤,稀盐酸酸化,乙醇重结晶,得白色固体4-(5-苯氧基吡啶-3-基)2-羟基苯甲酸A1 0.2g,产率33%。3-Bromo-5-phenoxypyridine (0.5g, 1.6mmol) and 2-hydroxy-4-(4,4,5,5-tetramethyl-1,3,2-dioxybenzaldehyde-2- base) methyl benzoate (0.5g, 1.9mmol) was dissolved in a mixture of 1,4-dioxane and water (volume ratio was 3:1), potassium carbonate (0.7g, 4.8mmol), ( 1,1-bis(diphenylphosphino)ferrocene)palladium dichloride (0.04g, 0.05mmol), replaced by argon, refluxed and stirred. Filtration with celite, acidification with dilute hydrochloric acid, and recrystallization from ethanol gave 0.2 g of 4-(5-phenoxypyridin-3-yl) 2-hydroxybenzoic acid A1 as a white solid, with a yield of 33%.
产物的结构表征数据如下:The structural characterization data of the product are as follows:
1H NMR(400MHz,DMSO-d6)δ7.11-7.17(d,J=8.0Hz,2H),7.18-7.25(t,J=7.4Hz,1H),7.26-7.34(m,2H),7.41-7.49(t,J=7.8Hz,2H),7.75-7.80(t,J=2.2Hz,1H),7.84-7.91(d,J=8.2Hz,1H),8.36-8.44(d,J=2.7Hz,1H),8.73-8.78(s,1H). 1 H NMR (400MHz, DMSO-d 6 )δ7.11-7.17(d, J=8.0Hz, 2H), 7.18-7.25(t, J=7.4Hz, 1H), 7.26-7.34(m, 2H), 7.41-7.49(t, J=7.8Hz, 2H), 7.75-7.80(t, J=2.2Hz, 1H), 7.84-7.91(d, J=8.2Hz, 1H), 8.36-8.44(d, J= 2.7Hz,1H),8.73-8.78(s,1H).
13C NMR(151MHz,DMSO-d6)δ172.09,161.91,156.49,153.90,143.55,143.26,141.01,136.01,131.51,130.76,124.67,124.44,119.19,118.41,115.84,113.31. 13 C NMR (151MHz, DMSO-d 6 )δ172.09, 161.91, 156.49, 153.90, 143.55, 143.26, 141.01, 136.01, 131.51, 130.76, 124.67, 124.44, 119.19, 118.41, 115 .84, 113.31.
实施例2Example 2
4-(5-(4-氯苯氧基)吡啶-3-基)-2-羟基苯甲酸(A2)的合成Synthesis of 4-(5-(4-chlorophenoxy)pyridin-3-yl)-2-hydroxybenzoic acid (A2)
3-羟基-5-溴吡啶(0.5g,2.9mmol)与4-氯-苯硼酸(0.9g,5.7mmol)溶于二氯甲烷中,加入吡啶(0.5mL,5.8mmol),醋酸铜(0.2g,0.7mmol),常温搅拌过夜,二氯甲烷萃取,无水硫酸钠干燥,乙酸乙酯:石油醚(体积比1:5)柱层析,得白色固体3-溴-5-(4-氯苯氧基)吡啶0.3g,产率34%。3-Hydroxy-5-bromopyridine (0.5g, 2.9mmol) and 4-chloro-phenylboronic acid (0.9g, 5.7mmol) were dissolved in dichloromethane, added pyridine (0.5mL, 5.8mmol), copper acetate (0.2 g, 0.7mmol), stirred overnight at room temperature, extracted with dichloromethane, dried over anhydrous sodium sulfate, ethyl acetate:petroleum ether (volume ratio 1:5) column chromatography, obtained white solid 3-bromo-5-(4- Chlorophenoxy)pyridine 0.3g, yield 34%.
3-溴-5-(4-氯苯氧基吡啶)(0.3g,1mmol)与2-羟基-4-(4,4,5,5-四甲基-1,3,2-二氧苯甲醛-2-基)苯甲酸甲酯(0.3g,1.2mmol)溶于1,4-二氧六环与水的混合液中(体积比为3:1),加入碳酸钾(0.4g,2.9mmol)、(1,1-双(二苯基磷)二茂铁)二氯化钯(0.02g,0.03mmol),置换氩气,回流搅拌6h。硅藻土过滤,稀盐酸酸化,乙醇重结晶,得白色固体4-(5-苯氧基吡啶-3-基)2-羟基苯甲酸A20.1g,产率25%。3-Bromo-5-(4-chlorophenoxypyridine) (0.3g, 1mmol) and 2-hydroxy-4-(4,4,5,5-tetramethyl-1,3,2-dioxobenzene Formaldehyde-2-yl)methyl benzoate (0.3g, 1.2mmol) was dissolved in a mixture of 1,4-dioxane and water (3:1 by volume), and potassium carbonate (0.4g, 2.9 mmol), (1,1-bis(diphenylphosphino)ferrocene)palladium dichloride (0.02g, 0.03mmol), replace argon, and stir at reflux for 6h. Filtration with diatomaceous earth, acidification with dilute hydrochloric acid, and recrystallization from ethanol gave 20.1 g of 4-(5-phenoxypyridin-3-yl) 2-hydroxybenzoic acid A as a white solid, with a yield of 25%.
产物的结构表征数据如下:The structural characterization data of the product are as follows:
1H NMR(400MHz,DMSO-d6)δ7.15-7.21(m,2H),7.28-7.33(dd,J=1.9,8.2Hz,1H),7.33-7.37(d,J=1.8Hz,1H),7.45-7.52(m,2H),7.82-7.86(t,J=2.3Hz,1H),7.86-7.91(d,J=8.2Hz,1H),8.41-8.47(d,J=2.6Hz,1H),8.74-8.82(d,J=1.9Hz,1H). 1 H NMR (400MHz,DMSO-d 6 )δ7.15-7.21(m,2H),7.28-7.33(dd,J=1.9,8.2Hz,1H),7.33-7.37(d,J=1.8Hz,1H ),7.45-7.52(m,2H),7.82-7.86(t,J=2.3Hz,1H),7.86-7.91(d,J=8.2Hz,1H),8.41-8.47(d,J=2.6Hz, 1H), 8.74-8.82(d, J=1.9Hz, 1H).
13C NMR(151MHz,DMSO-d6)δ172.06,161.90,155.55,153.54,143.69,143.45,141.26,136.06,131.51,130.58,128.40,124.78,120.88,118.48,115.90,113.34. 13 C NMR (151MHz, DMSO-d 6 ) δ172.06, 161.90, 155.55, 153.54, 143.69, 143.45, 141.26, 136.06, 131.51, 130.58, 128.40, 124.78, 120.88, 118.48, 115 .90, 113.34.
实施例3Example 3
4-(5-(对甲苯氧基)吡啶-3-基)2-羟基苯甲酸(A3)的合成Synthesis of 4-(5-(p-methylphenoxy)pyridin-3-yl)2-hydroxybenzoic acid (A3)
3-羟基-5-溴吡啶(1g,5.5mmol)与4-甲基苯硼酸(1.5g,11.1mmol)溶于二氯甲烷中,加入吡啶(1.4mL,16.5mmol),醋酸铜(0.3g,1.4mmol),常温搅拌过夜,二氯甲烷萃取,无水硫酸钠干燥,乙酸乙酯:石油醚(体积比1:5)柱层析,得白色固体3-溴-5-对甲苯氧基吡啶0.3g,产率22%。3-Hydroxy-5-bromopyridine (1g, 5.5mmol) and 4-methylphenylboronic acid (1.5g, 11.1mmol) were dissolved in dichloromethane, pyridine (1.4mL, 16.5mmol), copper acetate (0.3g , 1.4mmol), stirred overnight at room temperature, extracted with dichloromethane, dried over anhydrous sodium sulfate, ethyl acetate:petroleum ether (volume ratio 1:5) column chromatography, obtained white solid 3-bromo-5-p-methylphenoxy Pyridine 0.3g, yield 22%.
3-溴-5-对甲苯氧基吡啶(0.3g,1.2mmol)与2-羟基-4-(4,4,5,5-四甲基-1,3,2-二氧苯甲醛-2-基)苯甲酸甲酯(0.4g,1.5mmol)溶于1,4-二氧六环与水的混合液中(体积比为3:1),加入碳酸钾(0.5g,3.6mmol)、(1,1-双(二苯基磷)二茂铁)二氯化钯(0.03g,0.04mmol),置换氩气,回流搅拌6h。硅藻土过滤,稀盐酸酸化,乙醇重结晶,得白色固体4-(5-苯氧基吡啶-3-基)2-羟基苯甲酸A30.1g,产率22%。3-Bromo-5-p-tolyloxypyridine (0.3g, 1.2mmol) and 2-hydroxy-4-(4,4,5,5-tetramethyl-1,3,2-dioxybenzaldehyde-2 -yl) methyl benzoate (0.4g, 1.5mmol) was dissolved in a mixture of 1,4-dioxane and water (3:1 by volume), potassium carbonate (0.5g, 3.6mmol) was added, (1,1-bis(diphenylphosphino)ferrocene)palladium dichloride (0.03g, 0.04mmol), replaced with argon, and stirred at reflux for 6h. Filtration with celite, acidification with dilute hydrochloric acid, and recrystallization from ethanol gave 30.1 g of 4-(5-phenoxypyridin-3-yl) 2-hydroxybenzoic acid A as a white solid, with a yield of 22%.
产物的结构表征数据如下:The structural characterization data of the product are as follows:
1H NMR(400MHz,DMSO-d6)δ2.28-2.35(s,3H),7.02-7.08(d,J=7.9Hz,2H),7.21-7.32(m,4H),7.67-7.71(s,1H),7.84-7.90(d,J=8.2Hz,1H),8.33-8.38(d,J=2.7Hz,1H),8.68-8.73(s,1H). 1 H NMR (400MHz, DMSO-d 6 ) δ2.28-2.35(s, 3H), 7.02-7.08(d, J=7.9Hz, 2H), 7.21-7.32(m, 4H), 7.67-7.71(s ,1H),7.84-7.90(d,J=8.2Hz,1H),8.33-8.38(d,J=2.7Hz,1H),8.68-8.73(s,1H).
13C NMR(151MHz,DMSO-d6)δ172.04,161.89,154.41,153.96,143.58,142.84,140.54,135.94,133.98,131.53,131.13,123.75,119.43,118.39,115.80,113.36,20.77. 13 C NMR (151MHz, DMSO-d 6 )δ172.04, 161.89, 154.41, 153.96, 143.58, 142.84, 140.54, 135.94, 133.98, 131.53, 131.13, 123.75, 119.43, 118.39, 115 .80, 113.36, 20.77.
实施例4Example 4
4-(5-(4-(叔丁基)苯氧基)吡啶-3-基)-2-羟基苯甲酸(A4)的合成Synthesis of 4-(5-(4-(tert-butyl)phenoxy)pyridin-3-yl)-2-hydroxybenzoic acid (A4)
3-羟基-5-溴吡啶(0.9g,5mmol)与4-叔丁基苯硼酸(1.8g,10mmol)溶于二氯甲烷中,加入吡啶(1.2mL,15mmol),醋酸铜(0.2g,1.3mmol),常温搅拌过夜,二氯甲烷萃取,无水硫酸钠干燥,乙酸乙酯:石油醚(体积比1:5)柱层析,得白色固体5-(4-(叔丁基)苯氧基吡啶0.4g,产率27%。3-Hydroxy-5-bromopyridine (0.9g, 5mmol) and 4-tert-butylphenylboronic acid (1.8g, 10mmol) were dissolved in dichloromethane, pyridine (1.2mL, 15mmol), copper acetate (0.2g, 1.3mmol), stirred overnight at room temperature, extracted with dichloromethane, dried over anhydrous sodium sulfate, ethyl acetate:petroleum ether (volume ratio 1:5) column chromatography, obtained white solid 5-(4-(tert-butyl)benzene Oxypyridine 0.4 g, yield 27%.
5-(4-(叔丁基)苯氧基吡啶(0.4g,1.4mmol)与2-羟基-4-(4,4,5,5-四甲基-1,3,2-二氧苯甲醛-2-基)苯甲酸甲酯(0.5g,1.6mmol)溶于1,4-二氧六环与水的混合液中(体积比为3:1),加入碳酸钾(0.6g,4.1mmol)、(1,1-双(二苯基磷)二茂铁)二氯化钯(0.03g,0.04mmol),置换氩气,回流搅拌6h。硅藻土过滤,稀盐酸酸化,乙醇重结晶,得白色固体4-(5-苯氧基吡啶-3-基)2-羟基苯甲酸A40.1g,产率19%。5-(4-(tert-butyl)phenoxypyridine (0.4g, 1.4mmol) and 2-hydroxy-4-(4,4,5,5-tetramethyl-1,3,2-dioxobenzene Formaldehyde-2-yl)methyl benzoate (0.5g, 1.6mmol) was dissolved in a mixture of 1,4-dioxane and water (3:1 by volume), and potassium carbonate (0.6g, 4.1 mmol), (1,1-bis(diphenylphosphino)ferrocene)palladium dichloride (0.03g, 0.04mmol), replace argon, and stir at reflux for 6h. Diatomaceous earth filtration, acidification with dilute hydrochloric acid, ethanol weight Crystallized to obtain 40.1 g of white solid 4-(5-phenoxypyridin-3-yl) 2-hydroxybenzoic acid A with a yield of 19%.
产物的结构表征数据如下:The structural characterization data of the product are as follows:
1H NMR(400MHz,DMSO-d6)δ1.20-1.38(s,9H),7.04-7.11(m,2H),7.27-7.32(dd,J=1.8,8.2Hz,1H),7.34-7.37(d,J=1.8Hz,1H),7.41-7.48(m,2H),7.84-7.91(m,2H),8.37-8.41(d,J=2.6Hz,1H),8.74-8.79(d,J=1.8Hz,1H). 1 H NMR (400MHz,DMSO-d 6 )δ1.20-1.38(s,9H),7.04-7.11(m,2H),7.27-7.32(dd,J=1.8,8.2Hz,1H),7.34-7.37 (d,J=1.8Hz,1H),7.41-7.48(m,2H),7.84-7.91(m,2H),8.37-8.41(d,J=2.6Hz,1H),8.74-8.79(d,J =1.8Hz,1H).
13C NMR(151MHz,DMSO-d6)δ171.97,161.83,154.29,154.02,147.13,143.29,142.41,140.02,136.33,131.55,127.45,125.14,118.73,118.52,115.94,113.41,34.59,31.68. 13 C NMR (151MHz, DMSO-d 6 )δ171.97, 161.83, 154.29, 154.02, 147.13, 143.29, 142.41, 140.02, 136.33, 131.55, 127.45, 125.14, 118.73, 118.52, 115 .94, 113.41, 34.59, 31.68.
实施例5Example 5
4-(5-(3-氯苯氧基)吡啶-3-基)-2-羟基苯甲酸(A5)的合成Synthesis of 4-(5-(3-chlorophenoxy)pyridin-3-yl)-2-hydroxybenzoic acid (A5)
3-羟基-5-溴吡啶(1.7g,9.6mmol)与3-氯-苯硼酸(3.0g,19.2mmol)溶于二氯甲烷中,加入吡啶(2.3mL,28.8mmol),醋酸铜(0.4g,2.2mmol),常温搅拌过夜,二氯甲烷萃取,无水硫酸钠干燥,乙酸乙酯:石油醚(体积比1:5)柱层析,得白色固体3-溴-5-(3-氯苯氧基)吡啶0.4g,产率16%。3-Hydroxy-5-bromopyridine (1.7g, 9.6mmol) and 3-chloro-phenylboronic acid (3.0g, 19.2mmol) were dissolved in dichloromethane, added pyridine (2.3mL, 28.8mmol), copper acetate (0.4 g, 2.2mmol), stirred overnight at room temperature, extracted with dichloromethane, dried over anhydrous sodium sulfate, ethyl acetate:petroleum ether (volume ratio 1:5) column chromatography, obtained white solid 3-bromo-5-(3- Chlorophenoxy)pyridine 0.4g, yield 16%.
3-溴-5-(3-氯苯氧基)吡啶(0.4g,1.3mmol)与2-羟基-4-(4,4,5,5-四甲基-1,3,2-二氧苯甲醛-2-基)苯甲酸甲酯(0.4g,1.5mmol)溶于1,4-二氧六环与水的混合液中(体积比为3:1),加入碳酸钾(0.5g,3.8mmol)、(1,1-双(二苯基磷)二茂铁)二氯化钯(0.03g,0.04mmol),置换氩气,回流搅拌6h。硅藻土过滤,稀盐酸酸化,乙醇重结晶,得白色固体4-(5-苯氧基吡啶-3-基)2-羟基苯甲酸A5 0.1g,产率17%。3-Bromo-5-(3-chlorophenoxy)pyridine (0.4g, 1.3mmol) and 2-hydroxy-4-(4,4,5,5-tetramethyl-1,3,2-dioxo Benzaldehyde-2-yl)methyl benzoate (0.4g, 1.5mmol) was dissolved in a mixture of 1,4-dioxane and water (3:1 by volume), and potassium carbonate (0.5g, 3.8mmol), (1,1-bis(diphenylphosphino)ferrocene)palladium dichloride (0.03g, 0.04mmol), replace argon, and stir at reflux for 6h. Filtration with celite, acidification with dilute hydrochloric acid, and recrystallization from ethanol gave 0.1 g of 4-(5-phenoxypyridin-3-yl) 2-hydroxybenzoic acid A5 as a white solid, with a yield of 17%.
产物的结构表征数据如下:The structural characterization data of the product are as follows:
1H NMR(400MHz,DMSO-d6)δ7.14-7.19(m,1H),7.27-7.33(m,2H),7.34-7.40(dd,J=1.8,8.3Hz,1H),7.42-7.44(d,J=1.8Hz,1H),7.44-7.51(t,J=8.0Hz,1H),7.86-7.93(d,J=8.2Hz,1H),8.18-8.24(d,J=2.2Hz,1H),8.57-8.62(d,J=2.5Hz,1H),8.92-8.97(d,J=1.8Hz,1H). 1 H NMR (400MHz,DMSO-d 6 )δ7.14-7.19(m,1H),7.27-7.33(m,2H),7.34-7.40(dd,J=1.8,8.3Hz,1H),7.42-7.44 (d, J=1.8Hz, 1H), 7.44-7.51(t, J=8.0Hz, 1H), 7.86-7.93(d, J=8.2Hz, 1H), 8.18-8.24(d, J=2.2Hz, 1H), 8.57-8.62(d, J=2.5Hz, 1H), 8.92-8.97(d, J=1.8Hz, 1H).
13C NMR(151MHz,DMSO-d6)δ171.94,161.82,157.33,154.00,142.28,141.43,138.72,137.29,134.70,132.20,131.57,128.14,124.95,119.35,118.60,117.84,116.19,113.75. 13 C NMR (151MHz, DMSO-d 6 ) δ171.94, 161.82, 157.33, 154.00, 142.28, 141.43, 138.72, 137.29, 134.70, 132.20, 131.57, 128.14, 124.95, 119.35, 118 .60, 117.84, 116.19, 113.75.
实施例6Example 6
4-(5-(间甲苯氧基)吡啶-3-基)2-羟基苯甲酸(A6)的合成Synthesis of 4-(5-(m-methylphenoxy)pyridin-3-yl)2-hydroxybenzoic acid (A6)
3-羟基-5-溴吡啶(1.54g,8.8mmol)与3-甲基苯硼酸(2.4g,17.7mmol)溶于二氯甲烷中,加入吡啶(2.1mL,26.4mmol),醋酸铜(0.4g,2.2mmol),常温搅拌过夜,二氯甲烷萃取,无水硫酸钠干燥,乙酸乙酯:石油醚(体积比1:5)柱层析,得白色固体3-溴-5-间甲苯氧基吡啶0.4g,产率17%。3-Hydroxy-5-bromopyridine (1.54g, 8.8mmol) and 3-methylphenylboronic acid (2.4g, 17.7mmol) were dissolved in dichloromethane, added pyridine (2.1mL, 26.4mmol), copper acetate (0.4 g, 2.2mmol), stirred overnight at room temperature, extracted with dichloromethane, dried over anhydrous sodium sulfate, ethyl acetate:petroleum ether (volume ratio 1:5) column chromatography, obtained white solid 3-bromo-5-m-tolyloxy Based pyridine 0.4g, yield 17%.
3-溴-5-间甲苯氧基吡啶(0.4g,1.5mmol)与2-羟基-4-(4,4,5,5-四甲基-1,3,2-二氧苯甲醛-2-基)苯甲酸甲酯(0.5g,1.8mmol)溶于1,4-二氧六环与水的混合液中(体积比为3:1),加入碳酸钾(0.6g,4.5mmol)、(1,1-双(二苯基磷)二茂铁)二氯化钯(0.03g,0.04mmol),置换氩气,回流搅拌6h。硅藻土过滤,稀盐酸酸化,乙醇重结晶,得白色固体4-(5-苯氧基吡啶-3-基)2-羟基苯甲酸A60.1g,产率20%。3-Bromo-5-m-tolyloxypyridine (0.4g, 1.5mmol) and 2-hydroxy-4-(4,4,5,5-tetramethyl-1,3,2-dioxybenzaldehyde-2 - Base) methyl benzoate (0.5g, 1.8mmol) was dissolved in a mixture of 1,4-dioxane and water (3:1 by volume), potassium carbonate (0.6g, 4.5mmol) was added, (1,1-bis(diphenylphosphino)ferrocene)palladium dichloride (0.03g, 0.04mmol), replaced with argon, and stirred at reflux for 6h. Filtrate with diatomaceous earth, acidify with dilute hydrochloric acid, and recrystallize from ethanol to obtain 60.1 g of white solid 4-(5-phenoxypyridin-3-yl) 2-hydroxybenzoic acid A, with a yield of 20%.
产物的结构表征数据如下:The structural characterization data of the product are as follows:
1H NMR(400MHz,DMSO-d6)δ2.29-2.33(s,3H),6.88-6.99(m,2H),6.99-7.05(d,J=7.7Hz,1H),7.24-7.36(m,3H),7.73-7.79(t,J=2.3Hz,1H),7.82-7.91(d,J=8.2Hz,1H),8.33-8.40(d,J=2.6Hz,1H),8.71-8.76(d,J=1.9Hz,1H). 1 H NMR (400MHz, DMSO-d 6 ) δ2.29-2.33(s, 3H), 6.88-6.99(m, 2H), 6.99-7.05(d, J=7.7Hz, 1H), 7.24-7.36(m ,3H),7.73-7.79(t,J=2.3Hz,1H),7.82-7.91(d,J=8.2Hz,1H),8.33-8.40(d,J=2.6Hz,1H),8.71-8.76( d,J=1.9Hz,1H).
13C NMR(151MHz,DMSO-d6)δ172.07,161.88,156.45,154.00,143.57,143.08,140.89,140.60,136.00,131.53,130.44,125.42,124.48,119.72,118.45,116.21,115.83,113.25,21.37. 13 C NMR (151MHz, DMSO-d 6 ) δ172.07, 161.88, 156.45, 154.00, 143.57, 143.08, 140.89, 140.60, 136.00, 131.53, 130.44, 125.42, 124.48, 119.72, 118 .45, 116.21, 115.83, 113.25, 21.37.
实施例7Example 7
4-(5-(苯氨基)吡啶-3-基)2-羟基苯甲酸(A7)的合成Synthesis of 4-(5-(phenylamino)pyridin-3-yl)2-hydroxybenzoic acid (A7)
3-氨基-5-溴吡啶(0.8g,4.6mmol)与苯硼酸(1.1g,9.2mmol)溶于二氯甲烷中,加入吡啶(2.0mL,19.2mmol),醋酸铜(0.4g,2.2mmol),常温搅拌过夜,二氯甲烷萃取,无水硫酸钠干燥,乙酸乙酯:石油醚(体积比1:5)柱层析,得白色固体3-溴-5-氨基吡啶0.4g,产率38%。3-Amino-5-bromopyridine (0.8g, 4.6mmol) and phenylboronic acid (1.1g, 9.2mmol) were dissolved in dichloromethane, added pyridine (2.0mL, 19.2mmol), copper acetate (0.4g, 2.2mmol ), stirred overnight at room temperature, extracted with dichloromethane, dried over anhydrous sodium sulfate, ethyl acetate: sherwood oil (volume ratio 1:5) column chromatography, got white solid 3-bromo-5-aminopyridine 0.4g, yield 38%.
3-溴-5-氨基吡啶(0.4g,1.8mmol)与2-羟基-4-(4,4,5,5-四甲基-1,3,2-二氧苯甲醛-2-基)苯甲酸甲酯(0.6g,2.1mmol)溶于1,4-二氧六环与水的混合液中(体积比为3:1),加入碳酸钾(0.7g,5.3mmol)、(1,1-双(二苯基磷)二茂铁)二氯化钯(0.04g,0.05mmol),置换氩气,回流搅拌。硅藻土过滤,稀盐酸酸化,乙醇重结晶,得黄色固体4-(5-氨基吡啶-3-基)2-羟基苯甲酸A7 0.1g,产率18%。3-Bromo-5-aminopyridine (0.4g, 1.8mmol) and 2-hydroxy-4-(4,4,5,5-tetramethyl-1,3,2-dioxybenzaldehyde-2-yl) Methyl benzoate (0.6g, 2.1mmol) was dissolved in a mixture of 1,4-dioxane and water (3:1 by volume), potassium carbonate (0.7g, 5.3mmol), (1, 1-bis(diphenylphosphino)ferrocene)palladium dichloride (0.04g, 0.05mmol), replaced with argon, and stirred under reflux. Filtration with celite, acidification with dilute hydrochloric acid, and recrystallization from ethanol gave 0.1 g of 4-(5-aminopyridin-3-yl) 2-hydroxybenzoic acid A7 as a yellow solid, with a yield of 18%.
产物的结构表征数据如下:The structural characterization data of the product are as follows:
1H NMR(400MHz,DMSO-d6)δ9.05(s,1H),8.43(dd,J=11.2,2.2Hz,2H),7.94-7.86(m,2H),7.38-7.32(m,2H),7.31(d,J=1.7Hz,1H),7.26(td,J=8.5,1.6Hz,3H),7.00(td,J=7.3,1.2Hz,1H). 1 H NMR(400MHz,DMSO-d6)δ9.05(s,1H),8.43(dd,J=11.2,2.2Hz,2H),7.94-7.86(m,2H),7.38-7.32(m,2H) ,7.31(d,J=1.7Hz,1H),7.26(td,J=8.5,1.6Hz,3H),7.00(td,J=7.3,1.2Hz,1H).
13C NMR(151MHz,DMSO-d6)δ171.56,162.23,145.10,143.72,140.65,140.64,140.13,134.08,132.44,129.45,122.20,120.63,119.01,117.54,115.25,114.24. 13 C NMR (151MHz, DMSO-d 6 ) δ171.56, 162.23, 145.10, 143.72, 140.65, 140.64, 140.13, 134.08, 132.44, 129.45, 122.20, 120.63, 119.01, 117.54, 115 .25, 114.24.
实施例8Example 8
4-(5-((4-氯苯基)氨基)吡啶-3-基)-2-羟基苯甲酸(A8)的合成Synthesis of 4-(5-((4-chlorophenyl)amino)pyridin-3-yl)-2-hydroxybenzoic acid (A8)
3-氨基-5-溴吡啶(1.0g,5.8mmol)与4-氯-苯硼酸(1.8g,11.6mmol)溶于二氯甲烷中,加入吡啶(0.9mL,11.6mmol),醋酸铜(0.3g,1.4mmol),常温搅拌过夜,二氯甲烷萃取,无水硫酸钠干燥,乙酸乙酯:石油醚(体积比1:5)柱层析,得白色固体3-溴-5-(4-氯苯基)氨基吡啶0.4g,产率24%。3-Amino-5-bromopyridine (1.0g, 5.8mmol) and 4-chloro-phenylboronic acid (1.8g, 11.6mmol) were dissolved in dichloromethane, pyridine (0.9mL, 11.6mmol), copper acetate (0.3 g, 1.4mmol), stirred overnight at room temperature, extracted with dichloromethane, dried over anhydrous sodium sulfate, ethyl acetate:petroleum ether (volume ratio 1:5) column chromatography, obtained white solid 3-bromo-5-(4- Chlorophenyl)aminopyridine 0.4g, yield 24%.
3-溴-5-氨基吡啶(0.4g,1.4mmol)与2-羟基-4-(4,4,5,5-四甲基-1,3,2-二氧苯甲醛-2-基)苯甲酸甲酯(0.5g,1.7mmol)溶于1,4-二氧六环与水的混合液中(体积比为3:1),加入碳酸钾(0.6g,4.1mmol)、(1,1-双(二苯基磷)二茂铁)二氯化钯(0.03g,0.04mmol),置换氩气,回流搅拌6h。硅藻土过滤,稀盐酸酸化,乙醇重结晶,得黄色固体4-(5-((4-氯苯基)氨基)吡啶-3-基)-2-羟基苯甲酸A8 0.1g,产率18%。3-Bromo-5-aminopyridine (0.4g, 1.4mmol) and 2-hydroxy-4-(4,4,5,5-tetramethyl-1,3,2-dioxybenzaldehyde-2-yl) Methyl benzoate (0.5g, 1.7mmol) was dissolved in a mixture of 1,4-dioxane and water (3:1 by volume), potassium carbonate (0.6g, 4.1mmol), (1, 1-bis(diphenylphosphino)ferrocene)palladium dichloride (0.03g, 0.04mmol), replaced with argon, and stirred at reflux for 6h. Filtrate with diatomaceous earth, acidify with dilute hydrochloric acid, and recrystallize from ethanol to obtain 0.1 g of yellow solid 4-(5-((4-chlorophenyl)amino)pyridin-3-yl)-2-hydroxybenzoic acid A8, yield 18 %.
产物的结构表征数据如下:The structural characterization data of the product are as follows:
1H NMR(400MHz,DMSO-d6)δ9.57(s,1H),8.59(d,J=1.7Hz,1H),8.45(d,J=2.5Hz,1H),8.16(t,J=2.1Hz,1H),7.93(d,J=8.2Hz,1H),7.45-7.38(m,3H),7.36-7.30(m,3H). 1 H NMR (400MHz, DMSO-d6) δ9.57(s, 1H), 8.59(d, J=1.7Hz, 1H), 8.45(d, J=2.5Hz, 1H), 8.16(t, J=2.1 Hz,1H),7.93(d,J=8.2Hz,1H),7.45-7.38(m,3H),7.36-7.30(m,3H).
13C NMR(151MHz,DMSO-d6)δ171.89,161.82,143.11,142.03,139.80,137.85,132.20,131.72,130.38,129.97,126.78,125.94,121.22,118.52,116.17,114.07. 13 C NMR (151MHz, DMSO-d 6 )δ171.89, 161.82, 143.11, 142.03, 139.80, 137.85, 132.20, 131.72, 130.38, 129.97, 126.78, 125.94, 121.22, 118.52, 116 .17, 114.07.
实施例9Example 9
4-(5-((4-甲氧基苯基)氨基)吡啶-3-基)2-羟基苯甲酸(A9)的合成Synthesis of 4-(5-((4-methoxyphenyl)amino)pyridin-3-yl)2-hydroxybenzoic acid (A9)
3-氨基-5-溴吡啶(0.2g,1.1mmol)与4-甲氧基苯硼酸(0.3g,2.2mmol)溶于二氯甲烷中,加入吡啶(0.2mL,2.2mmol),醋酸铜(0.1g,0.6mmol),常温搅拌过夜,二氯甲烷萃取,无水硫酸钠干燥,乙酸乙酯:石油醚(体积比1:5)柱层析,得白色固体3-溴-5-(4-甲氧基苯基)氨基吡啶0.1g,产率37%。3-Amino-5-bromopyridine (0.2g, 1.1mmol) and 4-methoxyphenylboronic acid (0.3g, 2.2mmol) were dissolved in dichloromethane, added pyridine (0.2mL, 2.2mmol), copper acetate ( 0.1g, 0.6mmol), stirred overnight at room temperature, extracted with dichloromethane, dried over anhydrous sodium sulfate, ethyl acetate:petroleum ether (volume ratio 1:5) column chromatography, to give white solid 3-bromo-5-(4 -Methoxyphenyl)aminopyridine 0.1 g, yield 37%.
3-溴-5-(4-甲氧基苯基)氨基吡啶(0.1g,0.4mmol)与2-羟基-4-(4,4,5,5-四甲基-1,3,2-二氧苯甲醛-2-基)苯甲酸甲酯(0.12g,0.5mmol)溶于1,4-二氧六环与水的混合液中(体积比为3:1),加入碳酸钾(0.2g,1.3mmol)、(1,1-双(二苯基磷)二茂铁)二氯化钯(0.01g,0.02mmol),置换氩气,回流搅拌6h。硅藻土过滤,稀盐酸酸化,乙醇重结晶,得黄色固体4-(5-((4-甲氧基苯基)氨基)吡啶-3-基)2-羟基苯甲酸A9 0.1g,产率28%。3-Bromo-5-(4-methoxyphenyl)aminopyridine (0.1g, 0.4mmol) and 2-hydroxy-4-(4,4,5,5-tetramethyl-1,3,2- Dioxybenzaldehyde-2-yl)methyl benzoate (0.12g, 0.5mmol) was dissolved in a mixture of 1,4-dioxane and water (3:1 by volume), and potassium carbonate (0.2 g, 1.3mmol), (1,1-bis(diphenylphosphino)ferrocene)palladium dichloride (0.01g, 0.02mmol), replace argon, and stir at reflux for 6h. Filtrate with diatomaceous earth, acidify with dilute hydrochloric acid, and recrystallize from ethanol to obtain 0.1 g of yellow solid 4-(5-((4-methoxyphenyl)amino)pyridin-3-yl)2-hydroxybenzoic acid A9, the yield 28%.
产物的结构表征数据如下:The structural characterization data of the product are as follows:
1H NMR(600MHz,DMSO-d6)δ9.31(s,1H),8.49(d,J=1.7Hz,1H),8.24(d,J=2.5Hz,1H),8.01(t,J=2.0Hz,1H),7.93(d,J=8.2Hz,1H),7.35(d,J=1.8Hz,1H),7.31-7.24(m,3H),7.03-6.97(m,2H),3.77(s,3H). 1 H NMR (600MHz, DMSO-d 6 )δ9.31(s, 1H), 8.49(d, J=1.7Hz, 1H), 8.24(d, J=2.5Hz, 1H), 8.01(t, J= 2.0Hz, 1H), 7.93(d, J=8.2Hz, 1H), 7.35(d, J=1.8Hz, 1H), 7.31-7.24(m, 3H), 7.03-6.97(m, 2H), 3.77( s,3H).
13C NMR(151MHz,DMSO-d6)δ171.86,161.78,156.53,145.34,141.86,138.14,132.66,131.76,129.35,127.03,124.63,123.54,118.43,116.11,115.43,114.14,55.75. 13 C NMR (151MHz, DMSO-d 6 ) δ171.86, 161.78, 156.53, 145.34, 141.86, 138.14, 132.66, 131.76, 129.35, 127.03, 124.63, 123.54, 118.43, 116.11, 115 .43, 114.14, 55.75.
实施例10Example 10
4-(5-(对甲苯氨基)吡啶-3-基)2-羟基苯甲酸(A10)的合成Synthesis of 4-(5-(p-tolylamino)pyridin-3-yl)2-hydroxybenzoic acid (A10)
3-氨基-5-溴吡啶(1.0g,5.8mmol)与4-甲基苯硼酸(1.6g,11.6mmol)溶于二氯甲烷中,加入吡啶(0.9mL,11.6mmol),醋酸铜(0.5g,2.9mmol),常温搅拌过夜,二氯甲烷萃取,无水硫酸钠干燥,乙酸乙酯:石油醚(体积比1:5)柱层析,得白色固体3-溴-5-对甲苯氨基吡啶0.3g,产率22%。3-Amino-5-bromopyridine (1.0g, 5.8mmol) and 4-methylphenylboronic acid (1.6g, 11.6mmol) were dissolved in dichloromethane, pyridine (0.9mL, 11.6mmol), copper acetate (0.5 g, 2.9 mmol), stirred overnight at room temperature, extracted with dichloromethane, dried over anhydrous sodium sulfate, ethyl acetate:petroleum ether (volume ratio 1:5) column chromatography, and obtained 3-bromo-5-p-toluidine as a white solid Pyridine 0.3g, yield 22%.
3-溴-5-对甲苯氨基吡啶(0.3g,1.3mmol)与2-羟基-4-(4,4,5,5-四甲基-1,3,2-二氧苯甲醛-2-基)苯甲酸甲酯(0.4g,1.6mmol)溶于1,4-二氧六环与水的混合液中(体积比为3:1),加入碳酸钾(0.5g,3.9mmol)、(1,1-双(二苯基磷)二茂铁)二氯化钯(0.03g,0.04mmol),置换氩气,回流搅拌6h。硅藻土过滤,稀盐酸酸化,乙醇重结晶,得黄色固体4-(5-苯氧基吡啶-3-基)2-羟基苯甲酸A10 0.1g,产率18%。3-Bromo-5-p-tolylaminopyridine (0.3g, 1.3mmol) and 2-hydroxy-4-(4,4,5,5-tetramethyl-1,3,2-dioxybenzaldehyde-2- Base) methyl benzoate (0.4g, 1.6mmol) was dissolved in the mixed solution of 1,4-dioxane and water (volume ratio was 3:1), potassium carbonate (0.5g, 3.9mmol) was added, ( 1,1-bis(diphenylphosphino)ferrocene)palladium dichloride (0.03g, 0.04mmol), replaced with argon, and stirred at reflux for 6h. Filtration with celite, acidification with dilute hydrochloric acid, and recrystallization from ethanol gave 0.1 g of 4-(5-phenoxypyridin-3-yl) 2-hydroxybenzoic acid A10 as a yellow solid, with a yield of 18%.
产物的结构表征数据如下:The structural characterization data of the product are as follows:
1H NMR(400MHz,DMSO-d6)δ9.43(s,1H),8.54(d,J=1.7Hz,1H),8.34(d,J=2.5Hz,1H),8.11(dd,J=2.5,1.7Hz,1H),7.93(d,J=8.2Hz,1H),7.37(d,J=1.8Hz,1H),7.30(dd,J=8.2,1.9Hz,1H),7.22(s,4H),2.30(s,3H). 1 H NMR (400MHz, DMSO-d6) δ9.43(s, 1H), 8.54(d, J=1.7Hz, 1H), 8.34(d, J=2.5Hz, 1H), 8.11(dd, J=2.5 ,1.7Hz,1H),7.93(d,J=8.2Hz,1H),7.37(d,J=1.8Hz,1H),7.30(dd,J=8.2,1.9Hz,1H),7.22(s,4H ),2.30(s,3H).
13C NMR(151MHz,DMSO-d6)δ171.86,161.80,144.41,141.87,138.10,137.55,133.26,131.77,130.64,130.28,128.21,125.35,120.82,118.45,116.13,114.18,20.93. 13 C NMR (151MHz, DMSO-d 6 )δ171.86, 161.80, 144.41, 141.87, 138.10, 137.55, 133.26, 131.77, 130.64, 130.28, 128.21, 125.35, 120.82, 118.45, 116 .13, 114.18, 20.93.
实施例11Example 11
4-(5-((4-(叔丁基)苯基)氨基)吡啶-3-基)-2-羟基苯甲酸(A11)的合成Synthesis of 4-(5-((4-(tert-butyl)phenyl)amino)pyridin-3-yl)-2-hydroxybenzoic acid (A11)
3-氨基-5-溴吡啶(0.8g,4.6mmol)与4-叔丁基苯硼酸(1.2g,6.9mmol)溶于二氯甲烷中,加入吡啶(0.8mL,9.2mmol),醋酸铜(0.4g,2.2mmol),常温搅拌过夜,二氯甲烷萃取,无水硫酸钠干燥,乙酸乙酯:石油醚(体积比1:5)柱层析,得白色固体3-溴-5-((4-(叔丁基)苯基)氨基)吡啶0.5g,产率37%。3-Amino-5-bromopyridine (0.8g, 4.6mmol) and 4-tert-butylphenylboronic acid (1.2g, 6.9mmol) were dissolved in dichloromethane, added pyridine (0.8mL, 9.2mmol), copper acetate ( 0.4g, 2.2mmol), stirred overnight at room temperature, extracted with dichloromethane, dried over anhydrous sodium sulfate, ethyl acetate:petroleum ether (volume ratio 1:5) column chromatography, and obtained white solid 3-bromo-5-(( 0.5 g of 4-(tert-butyl)phenyl)amino)pyridine, yield 37%.
3-溴-5-((4-(叔丁基)苯基)氨基)吡啶(0.5g,1.4mmol)与2-羟基-4-(4,4,5,5-四甲基-1,3,2-二氧苯甲醛-2-基)苯甲酸甲酯(0.5g,1.7mmol)溶于1,4-二氧六环与水的混合液中(体积比为3:1),加入碳酸钾(0.6g,4.3mmol)、(1,1-双(二苯基磷)二茂铁)二氯化钯(0.03g,0.04mmol),置换氩气,回流搅拌6h。硅藻土过滤,稀盐酸酸化,乙醇重结晶,得黄色固体4-(5-苯氧基吡啶-3-基)2-羟基苯甲酸A11 0.2g,产率45%。3-bromo-5-((4-(tert-butyl)phenyl)amino)pyridine (0.5g, 1.4mmol) and 2-hydroxy-4-(4,4,5,5-tetramethyl-1, 3,2-Dioxybenzaldehyde-2-yl)methyl benzoate (0.5g, 1.7mmol) was dissolved in a mixture of 1,4-dioxane and water (3:1 by volume), and added Potassium carbonate (0.6g, 4.3mmol), (1,1-bis(diphenylphosphino)ferrocene)palladium dichloride (0.03g, 0.04mmol) was replaced by argon, and stirred at reflux for 6h. Filtration with diatomaceous earth, acidification with dilute hydrochloric acid, and recrystallization from ethanol gave 0.2 g of 4-(5-phenoxypyridin-3-yl) 2-hydroxybenzoic acid A11 as a yellow solid, with a yield of 45%.
产物的结构表征数据如下:The structural characterization data of the product are as follows:
1H NMR(400MHz,DMSO-d6)δ9.35(s,1H),8.54-8.49(m,1H),8.38(d,J=2.5Hz,1H),8.09(d,J=3.2Hz,1H),7.93(d,J=8.2Hz,1H),7.42(d,J=2.0Hz,1H),7.40-7.34(m,2H),7.31(dd,J=8.2,1.8Hz,1H),7.27-7.20(m,2H). 1 H NMR (400MHz, DMSO-d6) δ9.35(s, 1H), 8.54-8.49(m, 1H), 8.38(d, J=2.5Hz, 1H), 8.09(d, J=3.2Hz, 1H ),7.93(d,J=8.2Hz,1H),7.42(d,J=2.0Hz,1H),7.40-7.34(m,2H),7.31(dd,J=8.2,1.8Hz,1H),7.27 -7.20(m,2H).
13C NMR(151MHz,DMSO-d6)δ171.88,161.79,146.14,144.07,142.02,137.85,131.72,130.44,126.78,125.23,119.94,118.47,116.11,114.08,34.55,31.67. 13 C NMR (151MHz, DMSO-d 6 ) δ171.88, 161.79, 146.14, 144.07, 142.02, 137.85, 131.72, 130.44, 126.78, 125.23, 119.94, 118.47, 116.11, 114.08, 34. 55,31.67.
实施例12Example 12
4-(5-((3-氯苯基)氨基)吡啶-3-基)-2-羟基苯甲酸(A12)的合成Synthesis of 4-(5-((3-chlorophenyl)amino)pyridin-3-yl)-2-hydroxybenzoic acid (A12)
3-氨基-5-溴吡啶(1.0g,5.8mmol)与3-氯-苯硼酸(2.0g,11.6mmol)溶于二氯甲烷中,加入吡啶(1.0mL,11.6mmol),醋酸铜(0.32g,1.7mmol),常温搅拌过夜,二氯甲烷萃取,无水硫酸钠干燥,乙酸乙酯:石油醚(体积比1:5)柱层析,得白色固体3-溴-5-((3-氯苯基)氨基)吡啶0.4g,产率22%。3-Amino-5-bromopyridine (1.0g, 5.8mmol) and 3-chloro-phenylboronic acid (2.0g, 11.6mmol) were dissolved in dichloromethane, pyridine (1.0mL, 11.6mmol), copper acetate (0.32 g, 1.7mmol), stirred overnight at room temperature, extracted with dichloromethane, dried over anhydrous sodium sulfate, ethyl acetate:petroleum ether (volume ratio 1:5) column chromatography, obtained white solid 3-bromo-5-((3 -Chlorophenyl)amino)pyridine 0.4g, yield 22%.
3-溴-5-((3-氯苯基)氨基)吡啶(0.4g,1.3mmol)与2-羟基-4-(4,4,5,5-四甲基-1,3,2-二氧苯甲醛-2-基)苯甲酸甲酯(0.4g,1.5mmol)溶于1,4-二氧六环与水的混合液中(体积比为3:1),加入碳酸钾(0.5g,3.9mmol)、(1,1-双(二苯基磷)二茂铁)二氯化钯(0.03g,0.04mmol),置换氩气,回流搅拌6h。硅藻土过滤,稀盐酸酸化,乙醇重结晶,得黄色固体4-(5-((3-氯苯基)氨基)吡啶-3-基)-2-羟基苯甲酸A12 0.1g,产率20%。3-bromo-5-((3-chlorophenyl)amino)pyridine (0.4g, 1.3mmol) and 2-hydroxy-4-(4,4,5,5-tetramethyl-1,3,2- Dioxybenzaldehyde-2-yl)methyl benzoate (0.4g, 1.5mmol) was dissolved in a mixture of 1,4-dioxane and water (3:1 by volume), and potassium carbonate (0.5 g, 3.9mmol), (1,1-bis(diphenylphosphino)ferrocene)palladium dichloride (0.03g, 0.04mmol), replace argon, and stir at reflux for 6h. Filtrate with diatomaceous earth, acidify with dilute hydrochloric acid, and recrystallize from ethanol to obtain 0.1 g of yellow solid 4-(5-((3-chlorophenyl)amino)pyridin-3-yl)-2-hydroxybenzoic acid A12, yield 20 %.
产物的结构表征数据如下:The structural characterization data of the product are as follows:
1H NMR(400MHz,DMSO-d6)δ9.61(s,1H),8.63(d,J=1.7Hz,1H),8.51(d,J=2.4Hz,1H),8.20(t,J=2.1Hz,1H),7.94(d,J=8.2Hz,1H),7.43-7.36(m,2H),7.35-7.30(m,2H),7.28(td,J=8.1,2.2,1.0Hz,1H),7.09(td,J=7.9,2.0,0.9Hz,1H). 1 H NMR (400MHz, DMSO-d6) δ9.61(s, 1H), 8.63(d, J=1.7Hz, 1H), 8.51(d, J=2.4Hz, 1H), 8.20(t, J=2.1 Hz,1H),7.94(d,J=8.2Hz,1H),7.43-7.36(m,2H),7.35-7.30(m,2H),7.28(td,J=8.1,2.2,1.0Hz,1H) ,7.09(td,J=7.9,2.0,0.9Hz,1H).
13C NMR(151MHz,DMSO-d6)δ171.89,161.81,142.96,142.50,141.71,138.06,134.40,131.74,131.72,129.78,127.23,122.88,118.89,118.52,117.63,116.24,114.13. 13 C NMR (151MHz, DMSO-d 6 )δ171.89, 161.81, 142.96, 142.50, 141.71, 138.06, 134.40, 131.74, 131.72, 129.78, 127.23, 122.88, 118.89, 118.52, 117 .63, 116.24, 114.13.
实施例13Example 13
4-(5-((3,4-二氯苯基)氨基)吡啶-3-基)-2-羟基苯甲酸(A13)的合成Synthesis of 4-(5-((3,4-dichlorophenyl)amino)pyridin-3-yl)-2-hydroxybenzoic acid (A13)
3-氨基-5-溴吡啶(1.0g,5.8mmol)与3,4-二氯-苯硼酸(2.2g,11.6mmol)溶于二氯甲烷中,加入吡啶(0.9mL,11.6mmol),醋酸铜(0.3g,1.7mmol),常温搅拌过夜,二氯甲烷萃取,无水硫酸钠干燥,乙酸乙酯:石油醚(体积比1:5)柱层析,得白色固体3-溴-5-(3,4-二氯苯基)氨基)吡啶0.4g,产率23%。3-Amino-5-bromopyridine (1.0g, 5.8mmol) and 3,4-dichloro-phenylboronic acid (2.2g, 11.6mmol) were dissolved in dichloromethane, pyridine (0.9mL, 11.6mmol), acetic acid Copper (0.3g, 1.7mmol), stirred at room temperature overnight, extracted with dichloromethane, dried over anhydrous sodium sulfate, ethyl acetate:petroleum ether (volume ratio 1:5) column chromatography, to give white solid 3-bromo-5- (3,4-dichlorophenyl)amino)pyridine 0.4 g, yield 23%.
3-溴-5(3,4-二氯苯基)氨基)吡啶(0.4g,1.4mmol)与2-羟基-4-(4,4,5,5-四甲基-1,3,2-二氧苯甲醛-2-基)苯甲酸甲酯(0.5g,1.7mmol)溶于1,4-二氧六环与水的混合液中(体积比为3:1),加入碳酸钾(0.5g,4.1mmol)、(1,1-双(二苯基磷)二茂铁)二氯化钯(0.03g,0.04mmol),置换氩气,回流搅拌6h。硅藻土过滤,稀盐酸酸化,乙醇重结晶,得黄色固体4-(5-苯氧基吡啶-3-基)2-羟基苯甲酸A13 0.1g,产率24%。3-bromo-5(3,4-dichlorophenyl)amino)pyridine (0.4g, 1.4mmol) and 2-hydroxy-4-(4,4,5,5-tetramethyl-1,3,2 -Dioxybenzaldehyde-2-yl)methyl benzoate (0.5g, 1.7mmol) was dissolved in a mixture of 1,4-dioxane and water (3:1 by volume), and potassium carbonate ( 0.5g, 4.1mmol), (1,1-bis(diphenylphosphino)ferrocene)palladium dichloride (0.03g, 0.04mmol), replace argon, and stir at reflux for 6h. Filtration with celite, acidification with dilute hydrochloric acid, and recrystallization from ethanol gave 0.1 g of 4-(5-phenoxypyridin-3-yl) 2-hydroxybenzoic acid A13 as a yellow solid, with a yield of 24%.
产物的结构表征数据如下:The structural characterization data of the product are as follows:
1H NMR(400MHz,DMSO-d6)δ9.74(s,1H),8.65(d,J=1.8Hz,1H),8.53(d,J=2.5Hz,1H),8.23(t,J=2.1Hz,1H),7.93(d,J=8.2Hz,1H),7.58(d,J=8.7Hz,1H),7.51(d,J=2.6Hz,1H),7.41(d,J=1.8Hz,1H),7.34(dd,J=8.3,1.8Hz,1H),7.30(dd,J=8.8,2.7Hz,1H). 1 H NMR (400MHz, DMSO-d6) δ9.74(s, 1H), 8.65(d, J=1.8Hz, 1H), 8.53(d, J=2.5Hz, 1H), 8.23(t, J=2.1 Hz,1H),7.93(d,J=8.2Hz,1H),7.58(d,J=8.7Hz,1H),7.51(d,J=2.6Hz,1H),7.41(d,J=1.8Hz, 1H),7.34(dd,J=8.3,1.8Hz,1H),7.30(dd,J=8.8,2.7Hz,1H).
13C NMR(151MHz,DMSO-d6)δ171.89,161.81,142.74,141.57,141.28,138.10,132.25,132.01,131.78,131.71,130.02,127.55,124.31,120.61,119.27,118.53,116.27,114.14. 13 C NMR (151MHz, DMSO-d 6 )δ171.89, 161.81, 142.74, 141.57, 141.28, 138.10, 132.25, 132.01, 131.78, 131.71, 130.02, 127.55, 124.31, 120.61, 119 .27, 118.53, 116.27, 114.14.
实施例14Example 14
4-(5-((3-氟-4-甲基苯基)氨基)吡啶-3-基)-2-羟基苯甲酸(A14)的合成Synthesis of 4-(5-((3-fluoro-4-methylphenyl)amino)pyridin-3-yl)-2-hydroxybenzoic acid (A14)
3-氨基-5-溴吡啶(1.0g,5.8mmol)与3-氟4-甲基苯硼酸(1.8g,11.6mmol)溶于二氯甲烷中,加入吡啶(0.9mL,11.6mmol),醋酸铜(0.5g,2.9mmol),常温搅拌过夜,二氯甲烷萃取,无水硫酸钠干燥,乙酸乙酯:石油醚(体积比1:5)柱层析,得白色固体3-溴-5-((3-氟-4-甲基苯基)氨基)吡啶0.4g,产率22%。3-Amino-5-bromopyridine (1.0g, 5.8mmol) and 3-fluoro4-methylphenylboronic acid (1.8g, 11.6mmol) were dissolved in dichloromethane, added pyridine (0.9mL, 11.6mmol), acetic acid Copper (0.5g, 2.9mmol), stirred overnight at room temperature, extracted with dichloromethane, dried over anhydrous sodium sulfate, ethyl acetate:petroleum ether (volume ratio 1:5) column chromatography, 3-bromo-5- ((3-fluoro-4-methylphenyl)amino)pyridine 0.4 g, yield 22%.
3-溴-5-((3-氟-4-甲基苯基)氨基)吡啶(0.4g,1.7mmol)与2-羟基-4-(4,4,5,5-四甲基-1,3,2-二氧苯甲醛-2-基)苯甲酸甲酯(0.4g,1.5mmol)溶于1,4-二氧六环与水的混合液中(体积比为3:1),加入碳酸钾(0.5g,3.8mmol)、(1,1-双(二苯基磷)二茂铁)二氯化钯(0.03g,0.04mmol),置换氩气,回流搅拌6h。硅藻土过滤,稀盐酸酸化,乙醇重结晶,得黄色固体4-(5-苯氧基吡啶-3-基)2-羟基苯甲酸A14 0.1g,产率20%。3-bromo-5-((3-fluoro-4-methylphenyl)amino)pyridine (0.4g, 1.7mmol) and 2-hydroxy-4-(4,4,5,5-tetramethyl-1 , 3,2-Dioxybenzaldehyde-2-yl)methyl benzoate (0.4g, 1.5mmol) was dissolved in a mixture of 1,4-dioxane and water (volume ratio 3:1), Potassium carbonate (0.5g, 3.8mmol) and (1,1-bis(diphenylphosphino)ferrocene)palladium dichloride (0.03g, 0.04mmol) were added, argon was replaced, and stirred at reflux for 6h. Filtration with celite, acidification with dilute hydrochloric acid, and recrystallization from ethanol gave 0.1 g of 4-(5-phenoxypyridin-3-yl) 2-hydroxybenzoic acid A14 as a yellow solid, with a yield of 20%.
产物的结构表征数据如下:The structural characterization data of the product are as follows:
1H NMR(400MHz,DMSO-d6)δ9.35(s,1H),8.51(s,1H),8.47-8.39(m,1H),8.00(d,J=2.2Hz,1H),7.91(d,J=8.2Hz,1H),7.33(d,J=1.8Hz,1H),7.28(dd,J=8.2,1.8Hz,1H),7.24(t,J=8.5Hz,1H),7.08-6.99(m,2H),2.19(d,J=1.8Hz,3H). 1 H NMR (400MHz, DMSO-d6) δ9.35(s, 1H), 8.51(s, 1H), 8.47-8.39(m, 1H), 8.00(d, J=2.2Hz, 1H), 7.91(d ,J=8.2Hz,1H),7.33(d,J=1.8Hz,1H),7.28(dd,J=8.2,1.8Hz,1H),7.24(t,J=8.5Hz,1H),7.08-6.99 (m,2H),2.19(d,J=1.8Hz,3H).
13C NMR(151MHz,DMSO-d6)δ171.89,162.28-160.68(氟裂分),161.82,160.68,142.83,142.57,140.62,137.31,132.72,132.68,131.67,124.81,118.45,118.28,116.03,115.25,113.91,106.31,106.15,14.11. 13 C NMR (151MHz, DMSO-d 6 ) δ171.89, 162.28-160.68 (fluorine splitting), 161.82, 160.68, 142.83, 142.57, 140.62, 137.31, 132.72, 132.68, 131.67, 124.81, 118.4 5,118.28,116.03,115.25, 113.91, 106.31, 106.15, 14.11.
以上实施例所得产物的活性评价:The activity evaluation of above embodiment gained product:
一、化合物A1-14对XOR的体外抑制活性评价:1. In vitro inhibitory activity evaluation of compound A1-14 on XOR:
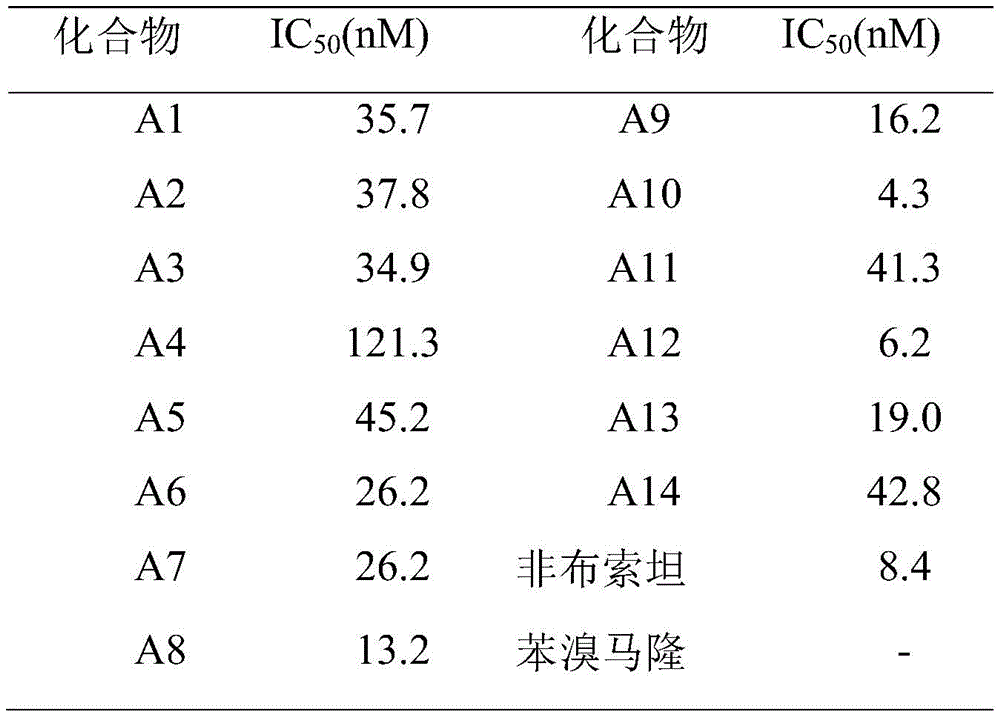
稀释10×PBS(pH=7.4)为1×PBS,以下反应体系中的PBS均为1×PBS。黄嘌呤称取15.2mg,加入250mL PBS和1mL NaOH溶液(1mmol/L)超声促溶,即得0.4mmol/L的底物溶液。取20μL的XOR母液加入20mL的PBS稀释并置于冰浴下保存,即得到0.5μg/100μL的酶液。设置空白组、对照组、实验组,空白组只加抑制剂和黄嘌呤溶液,对照组只加黄嘌呤溶液和酶液,实验组加入化合物A1-14,在96孔板中进行,将PBS缓冲液、100μL酶液、50μL各浓度的抑制剂在37℃下孵育3min后取出立即加入30μL底物启动。反应体系总体积为200μL,在295nm处每隔30s读数,共5min,每组实验平行测定4次。将各浓度的化合物在不同时间下所得的吸光度值转化为待测化合物的初速度,与对照组比较,通过计算得到各化合物的IC50值。见表1。Dilute 10×PBS (pH=7.4) to 1×PBS, and the PBS in the following reaction systems are all 1×PBS. Weigh 15.2mg of xanthine, add 250mL PBS and 1mL NaOH solution (1mmol/L) for sonication to induce dissolution, and obtain a 0.4mmol/L substrate solution. Take 20 μL of XOR master solution and add 20 mL of PBS to dilute and store in ice bath to obtain 0.5 μg/100 μL enzyme solution. Set up a blank group, a control group, and an experimental group. The blank group only adds inhibitors and xanthine solutions, the control group only adds xanthine solutions and enzyme solutions, and the experimental group adds compound A1-14. Carry out in a 96-well plate, PBS buffer Solution, 100 μL of enzyme solution, and 50 μL of inhibitors of various concentrations were incubated at 37°C for 3 minutes, then removed and immediately added with 30 μL of substrate to start. The total volume of the reaction system was 200 μL, readings were made every 30 s at 295 nm for a total of 5 min, and each experiment was measured 4 times in parallel. The absorbance values obtained at different times for each concentration of the compound were converted into the initial velocity of the test compound, and compared with the control group, the IC 50 value of each compound was obtained by calculation. See Table 1.
表1.化合物A1-14对XOR的体外抑制活性Table 1. In vitro inhibitory activity of compounds A1-14 on XOR
二、化合物A1-14对URAT1的体外抑制活性评价2. In vitro inhibitory activity evaluation of compound A1-14 on URAT1
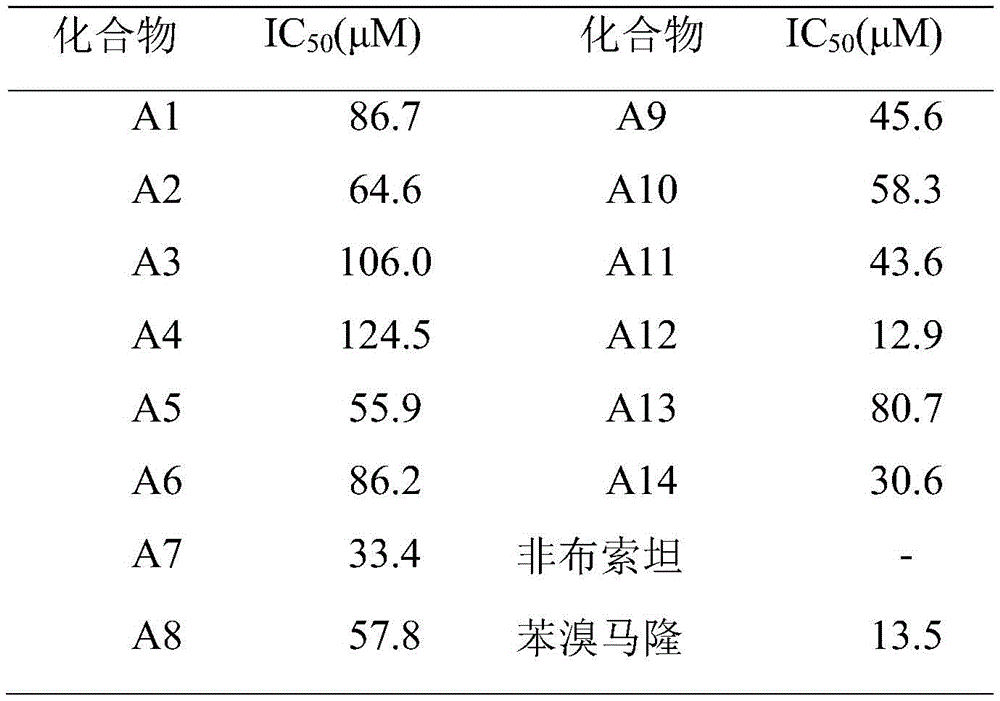
采用自建的测定URAT1活性的荧光检测方法对待测化合物进行活性测定。以6-羧基荧光素作为URAT1转运的底物,根据反应体系中加入待测化合物前后的荧光值的变化来确定待测化合物在体外对URAT1的抑制效应。自行构建的HEK293T/hURAT1稳转细胞,配制密度为2×105cells/mL的细胞悬液,在96孔荧光板中加入200μL/孔,设置空白组、对照组、实验组,每组5个复孔,其中,对照组、实验组加入HEK293T/hURAT1细胞,空白组不加细胞。培养48h后,空白组加入100μL的HBSS(无Cl-)溶液,对照组加入100μL6-羧基荧光素溶液(终浓度239.48μmol/L),实验组加入100μL含6-羧基荧光素(239.48μmol/L)的各浓度梯度的待测化合物溶液。1h后,吸除工作液,每孔加入100μL的NaOH溶液(0.5mol/L)裂解30min。在酶标仪中,设置激发波长490nm,发射波长525nm,震荡后进行读数。结果见表2。The self-built fluorescence detection method for measuring the activity of URAT1 was used to measure the activity of the compounds to be tested. Using 6-carboxyfluorescein as the substrate for URAT1 transport, the inhibitory effect of the test compound on URAT1 in vitro was determined according to the change of the fluorescence value before and after adding the test compound in the reaction system. Self-constructed HEK293T/hURAT1 stably transfected cells, prepared a cell suspension with a density of 2×10 5 cells/mL, added 200 μL/well in a 96-well fluorescent plate, and set up a blank group, a control group, and an experimental group, with 5 cells in each group Duplicate wells, in which HEK293T/hURAT1 cells were added to the control group and the experimental group, and cells were not added to the blank group. After culturing for 48 hours, add 100 μL of HBSS (without Cl - ) solution to the blank group, add 100 μL 6-carboxyfluorescein solution (final concentration 239.48 μmol/L) to the control group, and add 100 μL 6-carboxyfluorescein solution (239.48 μmol/L) to the experimental group. ) of each concentration gradient of the test compound solution. After 1 h, the working solution was sucked off, and 100 μL of NaOH solution (0.5 mol/L) was added to each well for lysis for 30 min. In the microplate reader, set the excitation wavelength to 490nm and the emission wavelength to 525nm, and read after shaking. The results are shown in Table 2.
表2.化合物A1-14对URAT1的体外抑制活性Table 2. In vitro inhibitory activity of compounds A1-14 on URAT1
由表1、2结果可知,本发明所得化合物A1-14有以下不可预期但是表现较为明确的构效关系。1、X用-NH-取代比用-O-连接效果好,不论是对XOR的抑制活性,还是对URAT1的抑制活性,化合物A7-14均较化合物A1-6有较明显的提升;2、R2固定为氢,R1用不同烷基取代时,随着取代烷基碳数增加,化合物对XOR和URAT1的抑制活性减弱,如A1与A4,A7与A11;3、化合物A12,X为-NH-时,R1为H,R2用Cl取代时,其体外活性对两个靶点的抑制活性最好,分别超过非布索坦和苯溴马隆。总之,本发明含2-羟基苯甲酸的三环类XOR/URAT1双重抑制剂的对尿酸代谢通路中的两个关键药物靶标均表现出了较强的抑制作用,具有潜在的明确的降尿酸的治疗效果。From the results in Tables 1 and 2, it can be seen that the compound A1-14 obtained in the present invention has the following unexpected but relatively clear structure-activity relationship. 1. Substituting X with -NH- is more effective than connecting with -O-. Whether it is the inhibitory activity of XOR or URAT1, compound A7-14 has a more obvious improvement than compound A1-6; 2. R 2 is fixed as hydrogen, and when R 1 is substituted with different alkyl groups, as the carbon number of the substituted alkyl group increases, the inhibitory activity of the compound on XOR and URAT1 is weakened, such as A1 and A4, A7 and A11; 3. Compound A12, X is When -NH-, R 1 is H, and when R 2 is replaced by Cl, its in vitro activity has the best inhibitory activity on the two targets, surpassing febuxostat and benzbromarone respectively. In conclusion, the tricyclic XOR/URAT1 dual inhibitors containing 2-hydroxybenzoic acid of the present invention have shown strong inhibitory effects on the two key drug targets in the uric acid metabolic pathway, and have potential clear uric acid-lowering effects. treatment effect.
上述实施例为本发明较佳的实施方式,但本发明的实施方式并不受上述实施例的限制,其它的任何未背离本发明的精神实质与原理下所作的改变、修饰、替代、组合、简化,均应为等效的置换方式,都包含在本发明的保护范围之内。The above-mentioned embodiment is a preferred embodiment of the present invention, but the embodiment of the present invention is not limited by the above-mentioned embodiment, and any other changes, modifications, substitutions, combinations, Simplifications should be equivalent replacement methods, and all are included in the protection scope of the present invention.
Claims (9)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202210326622.8A CN114805192B (en) | 2022-03-30 | 2022-03-30 | Tricyclic XOR/URAT1 dual inhibitor containing 2-hydroxybenzoic acid and preparation method and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202210326622.8A CN114805192B (en) | 2022-03-30 | 2022-03-30 | Tricyclic XOR/URAT1 dual inhibitor containing 2-hydroxybenzoic acid and preparation method and application thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN114805192A CN114805192A (en) | 2022-07-29 |
| CN114805192B true CN114805192B (en) | 2023-05-23 |
Family
ID=82532929
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202210326622.8A Active CN114805192B (en) | 2022-03-30 | 2022-03-30 | Tricyclic XOR/URAT1 dual inhibitor containing 2-hydroxybenzoic acid and preparation method and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN114805192B (en) |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101010300A (en) * | 2004-08-27 | 2007-08-01 | 安斯泰来制药株式会社 | 2-phenylpyridine derivative |
| US20070275950A1 (en) * | 2004-08-27 | 2007-11-29 | Astellas Pharma Inc. | 2-Phenylpyridine Derivative |
| WO2010044411A1 (en) * | 2008-10-15 | 2010-04-22 | キッセイ薬品工業株式会社 | Phenylisonicotinic acid derivative and use thereof for medical purposes |
| CN111148735A (en) * | 2017-10-04 | 2020-05-12 | 日本烟草产业株式会社 | Nitrogen-containing heteroaryl compounds and pharmaceutical uses thereof |
-
2022
- 2022-03-30 CN CN202210326622.8A patent/CN114805192B/en active Active
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101010300A (en) * | 2004-08-27 | 2007-08-01 | 安斯泰来制药株式会社 | 2-phenylpyridine derivative |
| US20070275950A1 (en) * | 2004-08-27 | 2007-11-29 | Astellas Pharma Inc. | 2-Phenylpyridine Derivative |
| WO2010044411A1 (en) * | 2008-10-15 | 2010-04-22 | キッセイ薬品工業株式会社 | Phenylisonicotinic acid derivative and use thereof for medical purposes |
| CN111148735A (en) * | 2017-10-04 | 2020-05-12 | 日本烟草产业株式会社 | Nitrogen-containing heteroaryl compounds and pharmaceutical uses thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| CN114805192A (en) | 2022-07-29 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP4482513B2 (en) | N2- (substituted arylmethyl) -3- (substituted phenyl) indazole and anti-angiogenic agent containing the same | |
| JPH07501330A (en) | Novel substituted salicylic acids | |
| CN103709154A (en) | Benzimidazole derivative and its preparation method and medical application | |
| WO2018108156A1 (en) | Rock inhibitor and application thereof | |
| CN103058949A (en) | Thiazole derivative acting as DHODH inhibitor and its application | |
| WO1998015530A1 (en) | Indole derivatives | |
| CN111875594A (en) | Indazole heterocycles having phosphodiesterase 4B inhibitory activity | |
| CN107108526A (en) | Bifunctional compounds and use for lowering uric acid levels | |
| CN109928972B (en) | A kind of matrine derivative and its application in medicine | |
| CN110240587A (en) | A class of aryl difluorobenzyl ether compounds, preparation method and use | |
| JP6953538B2 (en) | Biaryl compound, its production method and application | |
| WO2016145622A1 (en) | Benzimidazole derivatives, preparation method therefor and medicinal use thereof | |
| CN114805192B (en) | Tricyclic XOR/URAT1 dual inhibitor containing 2-hydroxybenzoic acid and preparation method and application thereof | |
| CN114605407A (en) | A kind of indole quinolinone compound and its synthesis method and application | |
| JP4828142B2 (en) | Novel fused pyrazolyl compounds | |
| CN111662275B (en) | Benzenesulfonamide IDH mutant inhibitor, its preparation method and use | |
| CN103755695B (en) | A kind of amides and application thereof with anti-tumor activity | |
| CN102796121B (en) | 3-aryl-7H-thiazol[3,2-b]-1,2,4-triazinyl-7-one derivatives and application thereof | |
| CN102532009B (en) | A compound for inhibiting dipeptide kininase and its preparation method and use | |
| CN108484604A (en) | Azo fragrance nitrogen oxides-chlorethylnitrosourea coupling molecule of target tumor hypoxemia and its preparation method and application | |
| CN110078668B (en) | Phenyl imidazole XOR inhibitor, preparation and application | |
| CN110204494B (en) | Oxygen-substituted phenylimidazole XOR/URAT1 dual inhibitor and preparation and application thereof | |
| CN113230240A (en) | 1, 3-diphenylprop-2-en-1-one derivative and application thereof | |
| CN118063386B (en) | Preparation and application of (E)-N-[(1H-indazol-7-yl)methyl]-3-arylacrylamide derivatives | |
| WO2020177752A1 (en) | 1,2,4-triazole compound, preparation method therefor and pharmaceutical use thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |