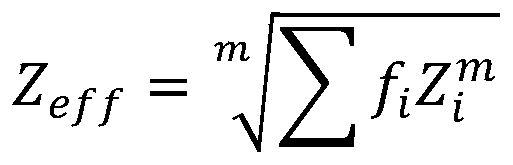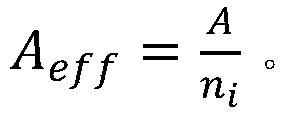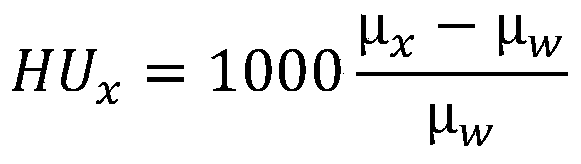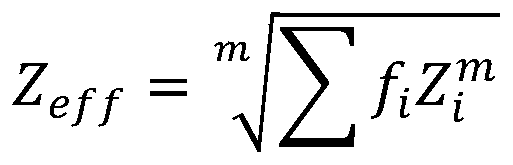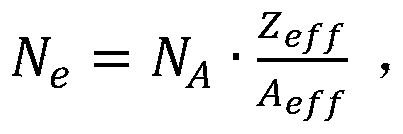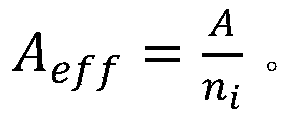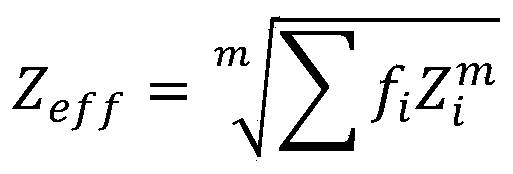CN114760931A - Phantom construction method and apparatus and phantom manufacturing method - Google Patents
Phantom construction method and apparatus and phantom manufacturing method Download PDFInfo
- Publication number
- CN114760931A CN114760931A CN202080082604.9A CN202080082604A CN114760931A CN 114760931 A CN114760931 A CN 114760931A CN 202080082604 A CN202080082604 A CN 202080082604A CN 114760931 A CN114760931 A CN 114760931A
- Authority
- CN
- China
- Prior art keywords
- phantom
- materials
- tissue
- atomic number
- effective
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B8/00—Diagnosis using ultrasonic, sonic or infrasonic waves
-
- G—PHYSICS
- G06—COMPUTING OR CALCULATING; COUNTING
- G06T—IMAGE DATA PROCESSING OR GENERATION, IN GENERAL
- G06T7/00—Image analysis
- G06T7/20—Analysis of motion
Landscapes
- Engineering & Computer Science (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Physics & Mathematics (AREA)
- Pathology (AREA)
- Heart & Thoracic Surgery (AREA)
- General Physics & Mathematics (AREA)
- Computer Vision & Pattern Recognition (AREA)
- Biophysics (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Multimedia (AREA)
- Radiology & Medical Imaging (AREA)
- Biomedical Technology (AREA)
- Theoretical Computer Science (AREA)
- Medical Informatics (AREA)
- Molecular Biology (AREA)
- Surgery (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Apparatus For Radiation Diagnosis (AREA)
- Nuclear Medicine (AREA)
Abstract
A CT performance phantom construction method and device and a phantom manufacturing method are provided, wherein the method comprises the following steps: generating a phantom model based on a portion of a biological subject requiring CT scanning using Fused Deposition Modeling (FDM) additive manufacturing, wherein the portion of the biological subject comprises a plurality of tissues; selecting a plurality of materials as alternative materials based on Henschel units of a plurality of tissues, wherein each tissue corresponds to the plurality of alternative materials; calculating the effective atomic number of the candidate materials, and selecting a plurality of first materials which are equal to the effective atomic number of each tissue in the candidate materials; the effective electron concentration of the first material is calculated and a second material in the first material equal to the effective electron concentration of each tissue is selected. The invention has high accuracy and low cost, and saves manpower.
Description
The invention relates to the field of CT scanning, in particular to a method and a device for constructing a phantom and a method for manufacturing the phantom.
Computed Tomography scanners (Computed Tomography scanners) are widely used in the medical field of macroscopic inspection for very small disruptions and changes, for example in organ, tumor search and cardiac examinations. Computed tomography scanners are also used to quickly diagnose internal injuries in an emergency situation to plan surgical interventions and to check the progress of treatment.
Acceptance testing and quality control of computer tomography scanners are very important. Imaging phantoms (Imaging phantoms) are specially designed to scan or image in the medical field to assess, analyze and coordinate the performance of different Imaging devices. These phantoms are easier to provide more consistent results than solid targets, and also avoid the direct risk of performing an examination physically. Among these, there are two types of imaging, one of which is phantom simulation (anthropomorphic phantoms), which provides a simplified geometric description of the human body and is designed to represent the characteristics of the human body, based on radiation attenuation (radiation attenuation), physical morphology (physical morphology) and geometry. Another type of imaging is mechanical imaging, which is designed to evaluate and standard verify image quality parameters.
A recent trend in the medical industry is the development of patient-specific protocols and artifacts. The radiation dose setting for a particular patient is one of the essential requirements in terms of current protocol optimization. Oncology and radiotherapy are the most benefited from this technique in the medical field, as tumor geometry is very variable, requiring more consideration in dose planning and quantitative accuracy. Thus, there is a need for customized imaging with improved and optimized radiation shielding, time and cost per procedure, and methods themselves. The goal is to achieve more efficient treatment based on the correct radiation dose to the target organ and to protect healthy tissue.
Nowadays realistic phantoms may also provide medical personnel with visual, practical training that approximates simulation of real situations, ideally being able to represent different individuals and being able to target specific patients. For radiation protection of patients and medical personnel, new phantoms can also provide data for dose calculation and physiological effects of ionizing radiation, which serves as the basis for a worldwide schedule of radiological safety. In addition to tailoring device imaging materials and procedures to the needs of existing specific patient or device qualifications, there is an increasing need in the industry to develop new materials and compositions to enable radiography (radiography) to better replicate tissues and organs. Such developments have meant small volume production and research that is economically unacceptable over traditional production models.
Another challenge of ct phantom manufacturing techniques is the lack of commercial products in certain areas, which means high product procurement costs and long import shipping times.
To date, traditional polymer processing methods have been widely used for producing simulated and mechanical molded bodies. When the traditionally produced imaging film is better able to replicate the absorption and radiation scattering of real tissue. The production of phantoms, which require a high investment, requires special attention to the specific location, and has a high standard and a low probability for the specific patient needs.
Additive manufacturing techniques enable layer-by-layer production based on CAD models, which can enable high customization, enable high geometric complexity of prints, and which are less costly in some low-volume production. Additive manufacturing techniques are therefore well suited for biotechnological applications, such as tissue or organ printing.
Fused Deposition Modeling (FDM) and polyethylene nozzle (Polyjet) are one of additive manufacturing techniques that have been tested and applied in some mechanical phantom production fields. However, some phantoms have very limited ability to replicate tissues and organs due to limited choices of polymer materials that represent the same Hounsfield Units as human tissue.
The prior art also proposes methods for producing phantoms using FDM techniques, but these methods involve separately manufacturing heart phantoms of different structural compositions using an FDM apparatus in a single extruder barrel (extruder), and then assembling and filling the phantoms with different materials, such as water, gel and oil, to simulate different tissue media in CT scans.
Disclosure of Invention
The invention provides a phantom construction method in a first aspect, which comprises the following steps: generating a phantom model based on a portion of a biological subject for which a CT scan is to be performed, wherein the portion of the biological subject includes a plurality of tissues; calculating the effective atomic number of the candidate materials, and selecting a plurality of first materials which are equal to the effective atomic number of each tissue in the candidate materials; calculating the effective electron concentration of the first material, and selecting a second material which is equal to the effective electron concentration of each tissue in the first material; calculating Hounsfield units of the second material to verify the selected second material.
Further, the hounsfield unit is:
wherein, muxRepresents the linear attenuation parameter, mu, of the tissuewRepresenting the linear decay parameter of water.
Further, the effective atomic number ZeffComprises the following steps:
wherein, i elementRelative electronic part of element ZiIs Σ fiThe index m is determined by the type of radioactive interaction and energy range, 1.
Further, the effective electron concentration is:
wherein N isAIs the Avogastron constant, AeffIs a valid atomic weight that is,wherein A is the molecular weight, niIs the atomic number of all types in all materials.
Further, the first material or the second material includes a plurality of elements mixed together in mass percentage.
A second aspect of the present invention provides a phantom manufacturing method, including the phantom construction method according to the first aspect of the present invention, wherein the phantom manufacturing method is performed in an FDM apparatus, wherein the FDM apparatus main members include two feeders, two nozzles, and a base, the phantom manufacturing method further including the steps of: providing a raw material in the feeder; and spraying the melted second material on a base station positioned below the nozzle, and gradually forming a phantom according to the phantom model.
A third aspect of the invention provides a phantom construction apparatus comprising: generating means for generating a phantom model based on a portion of an organism on which a CT scan is to be performed, wherein the portion of the organism comprises a plurality of tissues; a first calculation and selection device for calculating the effective atomic number of the alternative materials and selecting a plurality of first materials which are equal to the effective atomic number of each tissue in the alternative materials; a second calculation and selection device for calculating the effective electron concentration of the first material and selecting a second material in the first material which is equal to the effective electron concentration of each tissue; a calculation verification device that calculates Henschel units of the second material to verify the selected second material.
Further, the henry unit is:
wherein, muxRepresents the tissue linear attenuation parameter, μwRepresenting the linear decay parameter of water.
Further, the effective atomic number ZeffComprises the following steps:
wherein, the relative electron part Z of the i elementiIs Σ fiThe index m is determined by the type of radioactive interaction and energy range, 1.
Further, the effective electron concentration is:
wherein, NAIs the Avogastron constant, A effIs a valid atomic weight that is calculated by the atomic weight,wherein A is the molecular weight, niIs the number of atoms of all types in all materials.
Further, the first material or the second material includes a plurality of elements mixed together in mass percentage.
The invention can provide customers with a low cost, fast, and functionally customized phantom construction and manufacturing mechanism with mathematically adjustable material composition. In addition, the invention can support the manufacture of phantoms made of various materials, can develop new imitation materials for the manufacture of the phantoms, and saves labor and time. By implementing the present invention, the modeled material mix and composition of the phantom can be achieved based on the shape of the modeled tissue/organ. The present invention obtains a phantom of a complex construction by precise calculation and adjustment of materials, quickly obtains the phantom by performing FDM process with a simple construction and a variety of materials, and makes it possible to obtain new materials at a low cost. In addition, the invention can save the cost of the whole CT scanning device for testing and detection and has higher precision.
Fig. 1 is a schematic view of a phantom manufactured with an FDM apparatus according to an embodiment of the invention;
fig. 2 is a schematic view of the structure of a phantom according to one embodiment of the invention.
The following describes embodiments of the present invention with reference to the drawings.
In view of the increased demand for customized artifacts in the radiology area, the present invention provides a phantom manufacturing mechanism that employs a dual-extruder FDM technique and an adjustable polymer material solution to identify imitation materials of human tissue in a wide range of concentrations, actual atomic numbers, and electronic concentrations.
Specifically, in mimicking phantom material in CT scanning in the same hounsfield unit as human tissue, hounsfield units are first obtained that are independent of C-plate position, scanning conditions, X-ray spectra, light quality and light curing correction (correction for beam hardening), so the present invention does not rely on replication of hounsfield units of single photon energy or single X-ray spectra, but rather obtains material that simulates the same linear attenuation parameters as human tissue in the appropriate phantom energy range. However, this is more complex than just simulating a large number of attenuation parameters μ (E)/ρ, since the phantom material must also show the same specific center of gravity ρ (specific gravity) as the human tissue. In this regard, additive manufacturing techniques and dual-extrusion FDM processes may be particularly beneficial in the development of new materials to mimic human tissues and phantoms for CT imaging.
The present invention is particularly concerned with finding suitable materials for human phantoms having two types of materials, such as water and fat in this embodiment, and optimizes the FDM process to construct a phantom having at least two human tissues in CT imaging technology.
The invention provides a phantom construction method in a first aspect, which comprises the following steps.
Step S1 is first performed to generate a phantom model based on the portion of the biological subject that is to be printed by the CT scan, wherein the portion of the biological subject includes a plurality of tissues. As shown in fig. 2, in the present embodiment, the phantom P is composed of two tissues, tissue P1 and tissue P2, respectively. In particular, the phantom model is a CAD three-dimensional model.
It should be noted that the phantom is used for acceptance and testing of the quality of the CT scanner, i.e. for simulating the quality of a film produced by an organism in the CT scanner. The living body is generally a human body, and the living body part is generally a human organ or a tissue of a part of the human body, such as a heart.
Then, step S2 is executed to calculate the effective atomic number of the candidate material, and a plurality of first materials equal to the effective atomic number of each tissue in the candidate material are selected. The material used for a phantom should have the same mass attenuation coefficient (μm) as photons, and the same mass stopping power (mass stopping power) of electrons as its modeled human tissue. If the phantom has the same atomic composition as the human tissue to be simulated, the phantom will have the same parameters as these tissues. However, it is not easy to make the phantom and the human tissue it simulates have the same atomic composition. Therefore, the phantom material is expected to have mass density (mass density), effective atomic number (effective atomic number) and electron concentration similar to those of the real tissue of a simulated human body.
The invention makes use of the effective number of atoms ZeffAnd effective electron concentration NeThe reason for this as an indication of phantom material is that photon interactions, which have three main physical processes, depend on photon energy: photoelectric absorption (photoelectric absorption)<100keV)), Compton scattering (30 keV-30 MeV)) and generation of couples (pair production>1.02 MeV)). Based on the photon energy, one of three interactions plays a major role. In particular, the energy range is considered to be less than 150keV, which is a general implementation of CT devices. The magnitude of the photoelectric absorption is roughly proportional to the specific energy of the atomic number. The effective atomic number Z is in the main interaction process where the photoelectric efficiency is dominanteffThe concept of (a) is directed to selecting materials within the photon energy range. Compton influence (Compton interaction) essentially neutralizes the effective electron concentration N in the materialeAnd (4) in proportion.
The direct method for human tissue simulation provided by the invention is based on the effective atomic number ZeffIt is calculated from a set of partial mass attenuation coefficients (partial mass attenuation coefficients), elements (τ/ρ, σ e/ρ, κ/ρ), and specific photon energies. The basis of the human tissue simulation method provided by the invention is to screen out the effective atomic number Z with the electron concentration effMaterials of the same value, where x is the Z-index, which is derived for part of the interaction process and for parameters that cause the same photon and electron interaction characteristics to be displayed as a simulated material. The simulation technique consists of a screen of one phantom of a suitable filler material that serves as a specific base material and the relative proportions of the filler materials are determined in order to obtain a specific level of electron concentration and effective atomic number matching accuracy for both materials.
Therefore, in this embodiment, it is first necessary to evaluate the effective atomic number, wherein the effective atomic number ZeffComprises the following steps:
wherein ithRelative electronic part Z of the elementiIs Σ fiThe index m is determined by the type of radioactive interaction (e.g. photoelectric, electronic, etc.) and the energy range, where the radio attenuation interaction for the energy range m is typically 2.94.
The composition of the phantom material is determined based thereon. Zeff(BT)=Z eff(PM) in which Zeff(BT) is the effective atomic number of the simulated human tissue, Zeff(PM) is the effective atomic number of the phantom material. It is necessary to know that in screening for a material simulating human tissue, there are materials whose effective atomic number is known, and there are materials that need to be mixed together to obtain a mixed material having the same effective atomic number as the simulated human tissue.
Finally, step S3 is executed to calculate the effective electron concentration of the first material, and a second material having an effective electron concentration equal to that of each tissue in the first material is selected. Calculating the effective electron concentration of the phantom material:
wherein, NAIs the Avogadro number, AeffIs a valid atomic weight that is calculated by the atomic weight,wherein A is the molecular weight of the sample, niIs the number of atoms of all types in all synthetic materials.
Provided that the effective atomic number Z of the material is determinedeffEffective electron concentration N of Hemo model materialeThe same as the human body model tissue to be simulated, the correct material can be screened out.
Further, the first material or the second material includes a plurality of elements mixed together in mass percentage.
Then, step S4 is executed to select a plurality of materials as candidate materials based on hounsfield units of the plurality of tissues, wherein each tissue corresponds to a plurality of candidate materials. Among them, Hounsfield Unit (HU) is a quantitative ratio for describing radiodensity, which is generally used in the field of CT scanning. The value of hounsfield units is calculated based on the X-ray linear attenuation parameter of each individual tissue voxel, in particular first by a reconstruction process and then used to calculate CT values. Each tissue of the human body has a specific Henry unit size, and the human body part is imitated by selecting imitation materials equal to the Henry tissue of the human body tissue according to the Henry tissue of the human body tissue.
In CT technology, the criteria for application of phantom materials include detectability, usually measured by reproducibility of CT values, which includes Hounsfield Units (HU):
wherein, muxRepresents the linear attenuation parameter, mu, of the tissuewRepresenting the linear decay parameter of water. In this embodiment, μxAnd muwLinear attenuation parameters for fat and water, respectively, are expressed, in particular, the linear attenuation parameters for the material depend on the tissue composition, tissue concentration and photon energy (photon energy) of the phantom.
Therefore, assuming that the body organ to be imitated has two types of tissues, namely water and fat, and the hounsfield unit values of the water and the fat are known, a plurality of materials having the same hounsfield unit value of the water can be selected as candidate materials, and a plurality of materials having the same hounsfield unit value of the fat can be simultaneously selected as candidate materials.
It should be noted that when the Hencus unit value of the tissue does not match any suitable material, multiple materials may be mixed together to obtain a mixed material having the same Hencus unit value as the tissue.
Preferably, the present invention employs FDM techniques to print the phantoms. As shown in fig. 1, the FDM device 100 includes two sub-devices, a first sub-device 110 and a second sub-device 120. The first sub-assembly 110 includes a feeder 112, an extruder barrel 114, and a nozzle 116. The feeder 112 contains a raw material therein. Alternatively, the feeder 112 is a premix feeder, and when the raw materials are plural, the plural raw materials are mixed in a predetermined ratio and set in the feeder 112. An auger (not shown) is built into the extrusion cylinder 114 and a plurality of heating units (not shown) are provided on the outer wall of the extrusion cylinder 106, wherein the auger is driven by a motor (not shown) to heat and melt and mix the raw materials after they enter the extrusion cylinder 114 through the feeder 112. The nozzle 116 is connected to the extrusion cylinder 114, is disposed below the extrusion cylinder 114, sprays the melted material onto a base 130 located below the nozzle, and gradually forms a phantom P in a predetermined shape. Similarly, the second sub-assembly 120 also includes a feeder 122, an extrusion cylinder 124, and a nozzle 126, which are not described again for brevity.
Among them, as shown in fig. 1 and 2, the present embodiment requires manufacturing a phantom P requiring two types of materials, which continuously manufactures a predetermined shape by sending out two types of materials. Wherein two materials are respectively disposed in the feeders 112 and 122, wherein one of the two materials is water and the other is fat (adipose), the water is used as a reference material for CT scanning, the fat is used as a human tissue simulation material, and the two materials are used together for simulating a human organ for CT scanning. Specifically, phantom P includes two materials, with material P1 provided from feeder 112 and material P2 provided from feeder 122. Also, material P1 was simulated water and material P2 was simulated fat.
As shown in the material selection list shown in table 1, in the present embodiment, byIn the implementation of the invention, the material P1 was prepared by using elements H and O to imitate water, wherein the mass percentage of the element H was 11.19%, the mass percentage of the element O was 88.81%, the mass concentration of the element O was 1000kg/m3, the effective atomic number of the element O was 7.42, and the effective electron concentration of the element O was 3.343e/kg x1026. The material P2 is prepared by selecting elements H, C, N, O, S, Na, Cl and Ca for imitating fat, wherein the mass percentage of the element H is 11.2%, the mass percentage of the element C is 51.7%, the mass percentage of the element N is 1.3%, the mass percentage of the element O is 35.5%, the mass percentage of the element S is 0.1%, the mass percentage of the element Na is 0.1%, the mass percentage of the element Cl is 0.1%, the mass percentage of the element Ca is 0.1%, the mass concentration is 970kg/m3, the effective atomic number is 6.4, and the effective electron concentration is 3.342e/kg x10 26。
Table 1 materials selection list
As shown in fig. 1, the present invention is preferably implemented by an FMD device 100 that mixes the selected materials in the feeder individually according to the proportions and other parameters. The present invention can also optimize the phantom construction parameters, such as the abutment temperature, feedstock temperature and deposition rate, by optimizing the parameters of the FDM technique to obtain samples without deteriorating features in subsequent CT scan accuracy, including voids, porosity and cracks.
A second aspect of the present invention provides a phantom manufacturing method, including the phantom construction method according to the first aspect of the present invention, wherein the phantom manufacturing method is performed in an FDM apparatus, wherein the FDM apparatus main members include two feeders, two nozzles, and a base, the phantom manufacturing method further including the steps of: providing a raw material in the feeder; and spraying the melted second material on a base station positioned below the nozzle, and gradually forming a phantom according to the phantom model.
The present invention in a third aspect provides a phantom construction apparatus comprising: generating means for generating a phantom model based on a portion of a biological subject for which a CT scan is to be performed, wherein the portion of the biological subject comprises a plurality of tissues; the first calculation and selection device is used for calculating the effective atomic number of the alternative materials and selecting a plurality of first materials which are equal to the effective atomic number of each tissue in the alternative materials; a second calculation and selection means for calculating an effective electron concentration of the first material and selecting a second material having an effective electron concentration in the first material equal to that of each tissue; a calculation verification device that calculates Hounsfield units of the second material to verify the selected second material.
Further, the henry unit is:
wherein, muxRepresents the tissue linear attenuation parameter, μwRepresenting the linear decay parameter of water.
Further, the effective atomic number ZeffComprises the following steps:
wherein, the relative electron part Z of the i elementiIs Σ fiThe index m is determined by the type of radioactive interaction and energy range, 1.
Further, the effective electron concentration is:
wherein N isAIs the Avogastron constant, AeffIs a valid atomic weight that is,wherein A is the molecular weight, niIs the atomic number of all types in all materials.
Further, the first material or the second material includes a plurality of elements mixed together in mass percentage.
The present invention can provide customers with a low cost, fast, and functionally customized phantom construction and manufacturing mechanism with mathematically adjustable material composition. In addition, the invention can support the manufacture of phantoms made of various materials, and can develop new imitation materials for the manufacture of the phantoms, thereby saving labor and time. By performing the present invention, the modeled material mixing and composition of the phantom can be achieved based on the shape of the modeled tissue/organ. The present invention obtains phantoms of complex construction through precise calculation and adjustment of materials, and rapidly obtains phantoms by performing FDM process with simple construction and various materials, and makes it possible to obtain phantoms using new, inexpensive materials. In addition, the invention can save the cost of testing and detecting the whole CT scanning device and has higher precision.
While the present invention has been described in detail with reference to the preferred embodiments, it should be understood that the above description should not be taken as limiting the invention. Various modifications and alterations to this invention will become apparent to those skilled in the art upon reading the foregoing description. Accordingly, the scope of the invention should be limited only by the attached claims. Furthermore, any reference signs in the claims shall not be construed as limiting the claim concerned; the word "comprising" does not exclude the presence of other devices or steps than those listed in a claim or the specification; the terms "first," "second," and the like are used merely to denote names, and do not denote any particular order.
Claims (11)
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/CN2020/076271 WO2021164026A1 (en) | 2020-02-21 | 2020-02-21 | Method and device for constructing phantom and method for fabricating phantom |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN114760931A true CN114760931A (en) | 2022-07-15 |
Family
ID=77391425
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202080082604.9A Pending CN114760931A (en) | 2020-02-21 | 2020-02-21 | Phantom construction method and apparatus and phantom manufacturing method |
Country Status (2)
| Country | Link |
|---|---|
| CN (1) | CN114760931A (en) |
| WO (1) | WO2021164026A1 (en) |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20050077459A1 (en) * | 2003-02-19 | 2005-04-14 | Engler Mark J. | Radiation phantom |
| US20150164464A1 (en) * | 2011-06-29 | 2015-06-18 | Board Of Regents Of The University Of Texas System | Device and method for deriving ct numbers in cone beam computed tomography |
| US20160015836A1 (en) * | 2014-07-18 | 2016-01-21 | Gammex, Inc. | Water-Equivalent Phantom |
| CN106233333A (en) * | 2014-04-17 | 2016-12-14 | 三星麦迪森株式会社 | Medical imaging devices and the method operating this medical imaging devices |
| US20180114314A1 (en) * | 2015-04-20 | 2018-04-26 | Mars Bioimaging Limited | Improving material identification using multi-energy ct image data |
| EP3467771A1 (en) * | 2017-10-05 | 2019-04-10 | Koninklijke Philips N.V. | Image feature annotation in diagnostic imaging |
| CN110063740A (en) * | 2019-04-25 | 2019-07-30 | 泰山医学院 | Phantom material, preparation method and application suitable for the detection of CT standard value |
| US20190329067A1 (en) * | 2017-01-11 | 2019-10-31 | Neuboron Medtech Ltd. | Method for analyzing elements and mass ratios of elements of tissue and method for establishing geometric model based on medical image |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102940503B (en) * | 2012-10-31 | 2014-12-31 | 东北大学 | Jaw bone density measuring device and jaw bone density measuring method for oral computed tomography (CT) system |
| KR101524466B1 (en) * | 2013-06-21 | 2015-06-01 | 삼성전자주식회사 | Method for providing guide information to imagaing object, method for recomending object, and medical imaging apparatus |
| KR101664432B1 (en) * | 2014-02-12 | 2016-10-10 | 삼성전자주식회사 | Computer tomography apparatus and method for displaying a computer tomography image thereof |
| CN110504016B (en) * | 2018-05-18 | 2022-05-10 | 北京连心医疗科技有限公司 | Monte Carlo grid parallel dose calculation method, equipment and storage medium |
| CN110084864B (en) * | 2019-04-09 | 2023-04-28 | 南京航空航天大学 | An Electron Density Image Reconstruction Method Based on Spectral CT |
| CN110702706B (en) * | 2019-09-20 | 2022-05-20 | 天津大学 | Method for simulating output data of energy spectrum CT system |
-
2020
- 2020-02-21 WO PCT/CN2020/076271 patent/WO2021164026A1/en not_active Ceased
- 2020-02-21 CN CN202080082604.9A patent/CN114760931A/en active Pending
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20050077459A1 (en) * | 2003-02-19 | 2005-04-14 | Engler Mark J. | Radiation phantom |
| US20150164464A1 (en) * | 2011-06-29 | 2015-06-18 | Board Of Regents Of The University Of Texas System | Device and method for deriving ct numbers in cone beam computed tomography |
| CN106233333A (en) * | 2014-04-17 | 2016-12-14 | 三星麦迪森株式会社 | Medical imaging devices and the method operating this medical imaging devices |
| US20160015836A1 (en) * | 2014-07-18 | 2016-01-21 | Gammex, Inc. | Water-Equivalent Phantom |
| US20180114314A1 (en) * | 2015-04-20 | 2018-04-26 | Mars Bioimaging Limited | Improving material identification using multi-energy ct image data |
| US20190329067A1 (en) * | 2017-01-11 | 2019-10-31 | Neuboron Medtech Ltd. | Method for analyzing elements and mass ratios of elements of tissue and method for establishing geometric model based on medical image |
| EP3467771A1 (en) * | 2017-10-05 | 2019-04-10 | Koninklijke Philips N.V. | Image feature annotation in diagnostic imaging |
| CN110063740A (en) * | 2019-04-25 | 2019-07-30 | 泰山医学院 | Phantom material, preparation method and application suitable for the detection of CT standard value |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2021164026A1 (en) | 2021-08-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| McGarry et al. | Tissue mimicking materials for imaging and therapy phantoms: a review | |
| Ma et al. | Classification of X-ray attenuation properties of additive manufacturing and 3D printing materials using computed tomography from 70 to 140 kVp | |
| Filippou et al. | Recent advances on the development of phantoms using 3D printing for imaging with CT, MRI, PET, SPECT, and ultrasound | |
| DeWerd et al. | The phantoms of medical and health physics | |
| Caon | Voxel-based computational models of real human anatomy: a review | |
| Lee et al. | Organ doses for reference pediatric and adolescent patients undergoing computed tomography estimated by Monte Carlo simulation | |
| Price et al. | An open source heterogeneous 3D printed mouse phantom utilising a novel bone representative thermoplastic | |
| Okkalidis et al. | Assessment of 11 available materials with custom three-dimensional-printing patterns for the simulation of muscle, fat, and lung hounsfield units in patient-specific phantoms | |
| Hatamikia et al. | 3D printed patient-specific thorax phantom with realistic heterogenous bone radiopacity using filament printer technology | |
| Mille et al. | Fabrication of a pediatric torso phantom with multiple tissues represented using a dual nozzle thermoplastic 3D printer | |
| Ali et al. | Construction and validation of a low cost paediatric pelvis phantom | |
| CN104258505A (en) | Anthropomorphic phantom for tumor radiotherapy dosage individualized verification and building method and application thereof | |
| CN109308733A (en) | Geometrical model method for building up and dose calculation methodology based on medical image data | |
| Wegner et al. | Development and characterization of modular mouse phantoms for end-to-end testing and training in radiobiology experiments | |
| Jusufbegović et al. | Computed tomography tissue equivalence of 3D printing materials | |
| Axford et al. | An end-to-end assessment on the accuracy of adaptive radiotherapy in an MR-linac | |
| Hatamikia et al. | Additively manufactured patient-specific anthropomorphic thorax phantom with realistic radiation attenuation properties | |
| Bustillo et al. | 3D printed heterogeneous paediatric head and adult thorax phantoms for linear accelerator radiotherapy quality assurance: from fabrication to treatment delivery | |
| Wegner et al. | Requirement analysis in medical phantom development: a survey tool approach with an illustrative example of a multimodal deformable pelvic phantom | |
| Lustermans et al. | Development of a novel 3D-printed dynamic anthropomorphic thorax phantom for evaluation of four-dimensional computed tomography | |
| Costa | Computer tomography phantom applications | |
| Kim et al. | Physical phantom of typical Korean male for radiation protection purpose | |
| CN114760931A (en) | Phantom construction method and apparatus and phantom manufacturing method | |
| Villani et al. | Computed tomography imaging analysis of a fused filament fabrication (FFF) 3D printed neck-thyroid phantom for multidisciplinary purposes | |
| Andrade et al. | Visual impact of infill percentages for 3D printed radiologic simulators |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| WD01 | Invention patent application deemed withdrawn after publication |
Application publication date: 20220715 |
|
| WD01 | Invention patent application deemed withdrawn after publication |