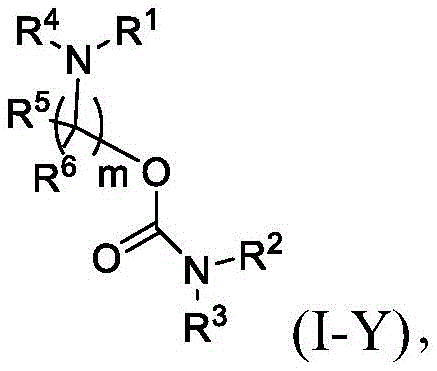CN114728886A - Carbonate-containing lipid compounds and compositions for intracellular delivery of therapeutic agents - Google Patents
Carbonate-containing lipid compounds and compositions for intracellular delivery of therapeutic agents Download PDFInfo
- Publication number
- CN114728886A CN114728886A CN202080080295.1A CN202080080295A CN114728886A CN 114728886 A CN114728886 A CN 114728886A CN 202080080295 A CN202080080295 A CN 202080080295A CN 114728886 A CN114728886 A CN 114728886A
- Authority
- CN
- China
- Prior art keywords
- peg
- compound
- lipid
- therapeutic
- alkyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C217/00—Compounds containing amino and etherified hydroxy groups bound to the same carbon skeleton
- C07C217/02—Compounds containing amino and etherified hydroxy groups bound to the same carbon skeleton having etherified hydroxy groups and amino groups bound to acyclic carbon atoms of the same carbon skeleton
- C07C217/04—Compounds containing amino and etherified hydroxy groups bound to the same carbon skeleton having etherified hydroxy groups and amino groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated
- C07C217/06—Compounds containing amino and etherified hydroxy groups bound to the same carbon skeleton having etherified hydroxy groups and amino groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated having only one etherified hydroxy group and one amino group bound to the carbon skeleton, which is not further substituted
- C07C217/08—Compounds containing amino and etherified hydroxy groups bound to the same carbon skeleton having etherified hydroxy groups and amino groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated having only one etherified hydroxy group and one amino group bound to the carbon skeleton, which is not further substituted the oxygen atom of the etherified hydroxy group being further bound to an acyclic carbon atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C219/00—Compounds containing amino and esterified hydroxy groups bound to the same carbon skeleton
- C07C219/02—Compounds containing amino and esterified hydroxy groups bound to the same carbon skeleton having esterified hydroxy groups and amino groups bound to acyclic carbon atoms of the same carbon skeleton
- C07C219/04—Compounds containing amino and esterified hydroxy groups bound to the same carbon skeleton having esterified hydroxy groups and amino groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated
- C07C219/16—Compounds containing amino and esterified hydroxy groups bound to the same carbon skeleton having esterified hydroxy groups and amino groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated having at least one of the hydroxy groups esterified by an inorganic acid or a derivative thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/24—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing atoms other than carbon, hydrogen, oxygen, halogen, nitrogen or sulfur, e.g. cyclomethicone or phospholipids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/127—Synthetic bilayered vehicles, e.g. liposomes or liposomes with cholesterol as the only non-phosphatidyl surfactant
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/127—Synthetic bilayered vehicles, e.g. liposomes or liposomes with cholesterol as the only non-phosphatidyl surfactant
- A61K9/1271—Non-conventional liposomes, e.g. PEGylated liposomes or liposomes coated or grafted with polymers
- A61K9/1272—Non-conventional liposomes, e.g. PEGylated liposomes or liposomes coated or grafted with polymers comprising non-phosphatidyl surfactants as bilayer-forming substances, e.g. cationic lipids or non-phosphatidyl liposomes coated or grafted with polymers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/50—Microcapsules having a gas, liquid or semi-solid filling; Solid microparticles or pellets surrounded by a distinct coating layer, e.g. coated microspheres, coated drug crystals
- A61K9/51—Nanocapsules; Nanoparticles
- A61K9/5107—Excipients; Inactive ingredients
- A61K9/5123—Organic compounds, e.g. fats, sugars
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C225/00—Compounds containing amino groups and doubly—bound oxygen atoms bound to the same carbon skeleton, at least one of the doubly—bound oxygen atoms not being part of a —CHO group, e.g. amino ketones
- C07C225/20—Compounds containing amino groups and doubly—bound oxygen atoms bound to the same carbon skeleton, at least one of the doubly—bound oxygen atoms not being part of a —CHO group, e.g. amino ketones having amino groups bound to carbon atoms of rings other than six-membered aromatic rings of the carbon skeleton
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C229/00—Compounds containing amino and carboxyl groups bound to the same carbon skeleton
- C07C229/02—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton
- C07C229/04—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated
- C07C229/06—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated having only one amino and one carboxyl group bound to the carbon skeleton
- C07C229/10—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated having only one amino and one carboxyl group bound to the carbon skeleton the nitrogen atom of the amino group being further bound to acyclic carbon atoms or to carbon atoms of rings other than six-membered aromatic rings
- C07C229/12—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated having only one amino and one carboxyl group bound to the carbon skeleton the nitrogen atom of the amino group being further bound to acyclic carbon atoms or to carbon atoms of rings other than six-membered aromatic rings to carbon atoms of acyclic carbon skeletons
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C229/00—Compounds containing amino and carboxyl groups bound to the same carbon skeleton
- C07C229/02—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton
- C07C229/04—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated
- C07C229/06—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated having only one amino and one carboxyl group bound to the carbon skeleton
- C07C229/10—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated having only one amino and one carboxyl group bound to the carbon skeleton the nitrogen atom of the amino group being further bound to acyclic carbon atoms or to carbon atoms of rings other than six-membered aromatic rings
- C07C229/16—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated having only one amino and one carboxyl group bound to the carbon skeleton the nitrogen atom of the amino group being further bound to acyclic carbon atoms or to carbon atoms of rings other than six-membered aromatic rings to carbon atoms of hydrocarbon radicals substituted by amino or carboxyl groups, e.g. ethylenediamine-tetra-acetic acid, iminodiacetic acids
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C251/00—Compounds containing nitrogen atoms doubly-bound to a carbon skeleton
- C07C251/32—Oximes
- C07C251/50—Oximes having oxygen atoms of oxyimino groups bound to carbon atoms of substituted hydrocarbon radicals
- C07C251/58—Oximes having oxygen atoms of oxyimino groups bound to carbon atoms of substituted hydrocarbon radicals of hydrocarbon radicals substituted by nitrogen atoms not being part of nitro or nitroso groups
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C271/00—Derivatives of carbamic acids, i.e. compounds containing any of the groups, the nitrogen atom not being part of nitro or nitroso groups
- C07C271/06—Esters of carbamic acids
- C07C271/08—Esters of carbamic acids having oxygen atoms of carbamate groups bound to acyclic carbon atoms
- C07C271/10—Esters of carbamic acids having oxygen atoms of carbamate groups bound to acyclic carbon atoms with the nitrogen atoms of the carbamate groups bound to hydrogen atoms or to acyclic carbon atoms
- C07C271/20—Esters of carbamic acids having oxygen atoms of carbamate groups bound to acyclic carbon atoms with the nitrogen atoms of the carbamate groups bound to hydrogen atoms or to acyclic carbon atoms to carbon atoms of hydrocarbon radicals substituted by nitrogen atoms not being part of nitro or nitroso groups
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C275/00—Derivatives of urea, i.e. compounds containing any of the groups, the nitrogen atoms not being part of nitro or nitroso groups
- C07C275/04—Derivatives of urea, i.e. compounds containing any of the groups, the nitrogen atoms not being part of nitro or nitroso groups having nitrogen atoms of urea groups bound to acyclic carbon atoms
- C07C275/06—Derivatives of urea, i.e. compounds containing any of the groups, the nitrogen atoms not being part of nitro or nitroso groups having nitrogen atoms of urea groups bound to acyclic carbon atoms of an acyclic and saturated carbon skeleton
- C07C275/14—Derivatives of urea, i.e. compounds containing any of the groups, the nitrogen atoms not being part of nitro or nitroso groups having nitrogen atoms of urea groups bound to acyclic carbon atoms of an acyclic and saturated carbon skeleton being further substituted by nitrogen atoms not being part of nitro or nitroso groups
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F5/00—Compounds containing elements of Groups 3 or 13 of the Periodic Table
- C07F5/02—Boron compounds
- C07F5/025—Boronic and borinic acid compounds
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic Table
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/18—Compounds having one or more C—Si linkages as well as one or more C—O—Si linkages
- C07F7/1804—Compounds having Si-O-C linkages
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2601/00—Systems containing only non-condensed rings
- C07C2601/02—Systems containing only non-condensed rings with a three-membered ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2601/00—Systems containing only non-condensed rings
- C07C2601/04—Systems containing only non-condensed rings with a four-membered ring
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Animal Behavior & Ethology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Molecular Biology (AREA)
- Biophysics (AREA)
- Dispersion Chemistry (AREA)
- Biomedical Technology (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Physics & Mathematics (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nanotechnology (AREA)
- Optics & Photonics (AREA)
- Inorganic Chemistry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Medicinal Preparation (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
Description
相关申请Related applications
本申请要求2019年9月19日提交的美国临时申请号62/902,929的优先权和权益,所述申请的全部公开内容以引用的方式并入文本。This application claims priority to and the benefit of US Provisional Application No. 62/902,929, filed September 19, 2019, the entire disclosure of which is incorporated herein by reference.
技术领域technical field
本公开提供了新型化合物、包含此类化合物的组合物以及包括脂质纳米粒子组合物以将一种或多种治疗剂和/或预防剂递送至哺乳动物细胞或器官和/或在哺乳动物细胞或器官中产生多肽的方法。除了新型脂质之外,本公开的脂质纳米粒子组合物还可以包括指定分数的一种或多种阳离子的和/或可离子化的氨基脂质、包括多不饱和脂质在内的磷脂、PEG脂质、结构性脂质、和/或治疗剂和/或预防剂。The present disclosure provides novel compounds, compositions comprising such compounds, and compositions comprising lipid nanoparticles for the delivery of one or more therapeutic and/or prophylactic agents to and/or in mammalian cells or organs or a method of producing a polypeptide in an organ. In addition to novel lipids, lipid nanoparticle compositions of the present disclosure may also include specified fractions of one or more cationic and/or ionizable amino lipids, phospholipids including polyunsaturated lipids , PEG lipids, structured lipids, and/or therapeutic and/or prophylactic agents.
背景技术Background technique
生物活性物质诸如小分子药物、蛋白质和核酸的有效靶向递送提出了一个持久的医学难题。特别是,将核酸递送至细胞因此类物质的相对不稳定性和低细胞渗透性而变得困难。因此,需要开发有助于将治疗剂和/或预防剂诸如核酸递送至细胞的方法和组合物。Efficient targeted delivery of biologically active substances such as small molecule drugs, proteins and nucleic acids presents an enduring medical challenge. In particular, delivery of nucleic acids to cells is made difficult by the relative instability and low cellular permeability of such substances. Accordingly, there is a need to develop methods and compositions that facilitate the delivery of therapeutic and/or prophylactic agents, such as nucleic acids, to cells.
含脂质的纳米粒子组合物、脂质体和脂质体复合物已被证明可有效地作为生物活性物质诸如小分子药物、蛋白质和核酸进入细胞和/或细胞内区室的转运媒介物。此类组合物通常包含一种或多种“阳离子”和/或氨基(可离子化)脂质、包括多不饱和脂质在内的磷脂、结构性脂质(例如固醇类)和/或含有聚乙二醇的脂质(PEG脂质)。阳离子和/或可离子化脂质包括例如可容易地质子化的含胺脂质。尽管已经展示了多种此类含脂质的纳米粒子组合物,但仍缺乏安全性、功效和特异性方面的改善。Lipid-containing nanoparticle compositions, liposomes and liposome complexes have been shown to be effective as transport vehicles for biologically active substances such as small molecule drugs, proteins and nucleic acids into cells and/or intracellular compartments. Such compositions typically comprise one or more "cationic" and/or amino (ionizable) lipids, phospholipids including polyunsaturated lipids, structured lipids (eg, sterols) and/or Lipids containing polyethylene glycol (PEG lipids). Cationic and/or ionizable lipids include, for example, amine-containing lipids that can be readily protonated. Although a variety of such lipid-containing nanoparticle compositions have been demonstrated, improvements in safety, efficacy and specificity are still lacking.
发明内容SUMMARY OF THE INVENTION
本公开提供了新型化合物以及包括所述新型化合物的组合物和方法。The present disclosure provides novel compounds and compositions and methods including the same.
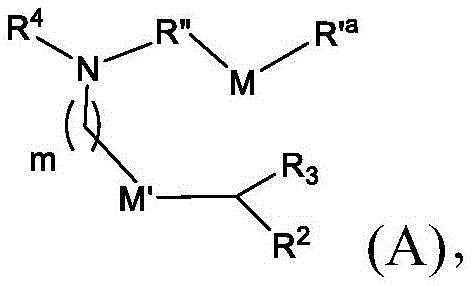
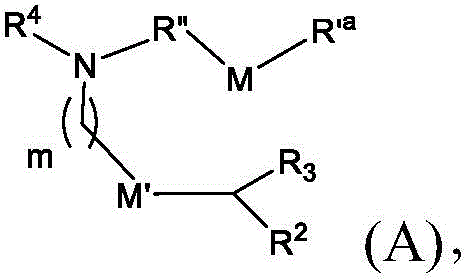
在一些方面,本公开涉及式(A)的化合物:In some aspects, the present disclosure relates to compounds of formula (A):
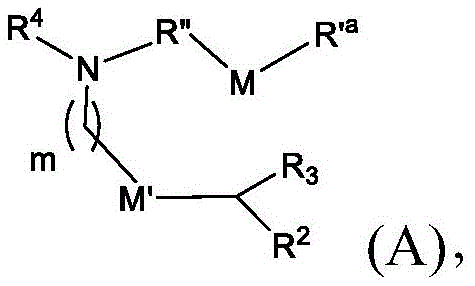
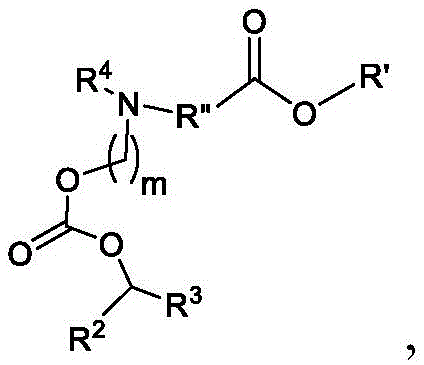
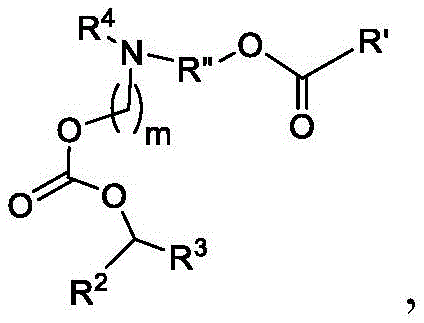
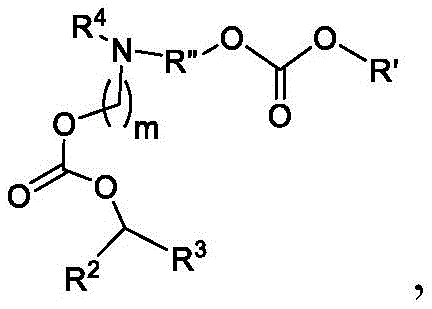
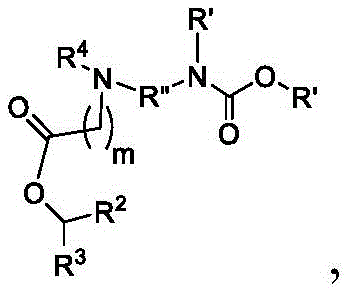
或其N-氧化物,或它们的盐或异构体,其中 or their N-oxides, or their salts or isomers, wherein
m选自5、6、7、8和9;m is selected from 5, 6, 7, 8 and 9;
R2和R3各自独立地选自由H、C1-14烷基和C2-14烯基组成的组;R 2 and R 3 are each independently selected from the group consisting of H, C 1-14 alkyl and C 2-14 alkenyl;
R4选自-(CH2)nOH,其中n选自1、2、3、4和5,和其中n2选自1、2、3、4、5、6、7、8、9和10;并且R10是-N(R)2,其中每个R独立地选自由C1-6烷基、C2-3烯基和H组成的组;R4 is selected from -( CH2 )nOH, wherein n is selected from 1, 2, 3, 4 and 5, and wherein n2 is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, and 10 ; and R10 is -N(R) 2 , wherein each R is independently selected from C1-6 alkyl, The group consisting of C 2-3 alkenyl and H;
M和M'各自独立地选自-OC(O)O-和-C(O)O-;M and M' are each independently selected from -OC(O)O- and -C(O)O-;
其中M和M'中的至少一个是-OC(O)O-;wherein at least one of M and M' is -OC(O)O-;
R’a是C1-18烷基或C2-18烯基;并且R' a is C 1-18 alkyl or C 2-18 alkenyl; and
R”是任选地被OH取代的C3-C13烷基。R" is C3 - C13 alkyl optionally substituted with OH.
具体实施方式Detailed ways
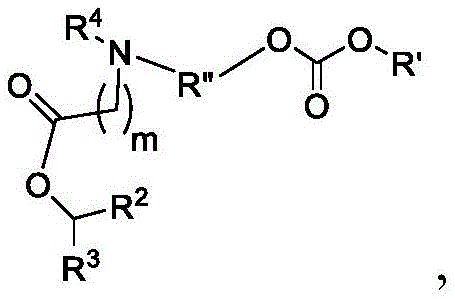
本公开涉及新型脂质和包含新型脂质的脂质纳米粒子(例如,空LNP或负载LNP)。本公开还提供了将治疗剂和/或预防剂递送至哺乳动物细胞的方法,具体地说,将治疗剂和/或预防剂递送至哺乳动物器官,在哺乳动物细胞中产生所关注的多肽,与包含其他脂质的LNP相比提高哺乳动物细胞中产生的蛋白质水平,以及治疗有需要的哺乳动物的疾病或病症。例如,在细胞中产生所关注的多肽的方法包括使包含mRNA的纳米粒子与哺乳动物细胞接触,由此可以翻译mRNA以产生所关注的多肽。将治疗剂和/或预防剂递送至哺乳动物细胞或器官的方法可以包括将包含治疗剂和/或预防剂的纳米粒子组合物施用至受试者,其中所述施用包括使细胞或器官与组合物接触,由此将治疗剂和/或预防剂递送至细胞或器官。此类递送方法可以是体外或体内的。The present disclosure relates to novel lipids and lipid nanoparticles comprising novel lipids (eg, empty LNPs or loaded LNPs). The present disclosure also provides methods of delivering therapeutic and/or prophylactic agents to mammalian cells, in particular, delivering therapeutic and/or prophylactic agents to mammalian organs in which a polypeptide of interest is produced, Increasing protein levels produced in mammalian cells compared to LNPs comprising other lipids, and treating a disease or disorder in a mammal in need thereof. For example, a method of producing a polypeptide of interest in a cell includes contacting a nanoparticle comprising mRNA with a mammalian cell, whereby the mRNA can be translated to produce the polypeptide of interest. A method of delivering a therapeutic and/or prophylactic agent to a mammalian cell or organ may comprise administering to a subject a nanoparticle composition comprising the therapeutic and/or prophylactic agent, wherein the administering comprises subjecting the cell or organ to the combination contact, thereby delivering therapeutic and/or prophylactic agents to cells or organs. Such delivery methods can be in vitro or in vivo.
本公开提供了包含中心胺部分和至少一个可生物降解基团的脂质。本文所述的脂质可有利地用于脂质纳米粒子组合物中,以将治疗剂和/或预防剂递送至哺乳动物细胞或器官。例如,本文所述的脂质具有极低的或不具有免疫原性。例如,与参考脂质(例如,MC3、KC2或DLinDMA)相比,式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I’)、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的脂质化合物的免疫原性较低。例如,与包含参考脂质(例如,MC3、KC2或DLinDMA)和相同治疗剂或预防剂的对应制剂相比,包含本文公开的脂质和治疗剂或预防剂的制剂的治疗指数增加。The present disclosure provides lipids comprising a central amine moiety and at least one biodegradable group. The lipids described herein can be advantageously used in lipid nanoparticle compositions to deliver therapeutic and/or prophylactic agents to mammalian cells or organs. For example, the lipids described herein have little or no immunogenicity. For example, formulas (A), (A1), (A2), (A3), (B), (I), (I'), (I-X) compared to a reference lipid (eg, MC3, KC2, or DLinDMA) ), (I-Y), (I-Ya) (IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) Or the lipid compounds of (IIb) are less immunogenic. For example, a formulation comprising a lipid disclosed herein and a therapeutic or prophylactic agent has an increased therapeutic index compared to a corresponding formulation comprising a reference lipid (eg, MC3, KC2 or DLinDMA) and the same therapeutic or prophylactic agent.
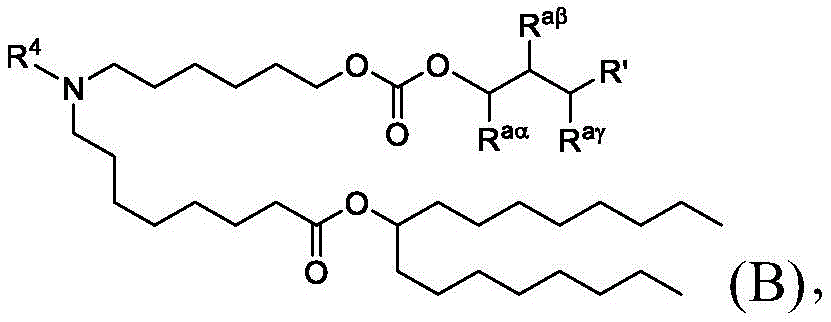
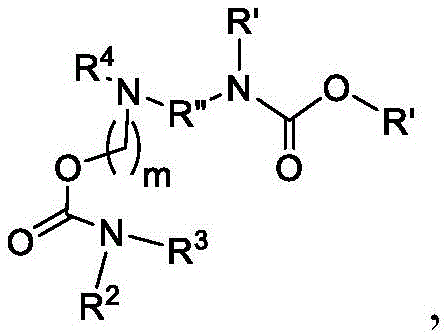
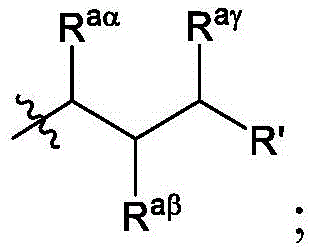
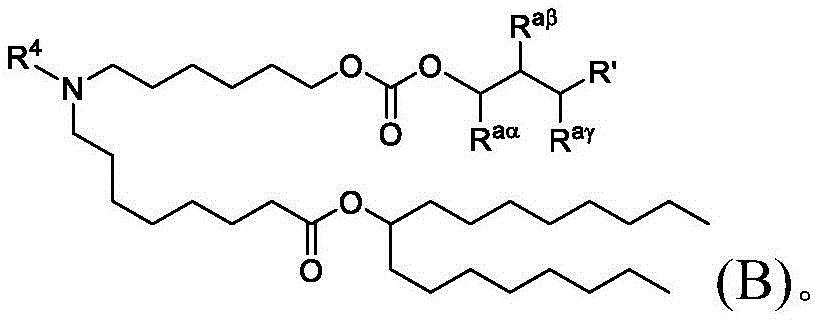
在一些方面,本公开涉及式(B)的化合物:其中In some aspects, the present disclosure relates to compounds of formula (B): in
R4选自-(CH2)nOH,其中n选自1、2、3、4和5;并且其中n2选自1、2、3、4、5、6、7、8、9和10;并且R10是-N(R)2,其中每个R独立地选自由C1-6烷基、C2-3烯基和H组成的组;R4 is selected from -( CH2 )nOH, wherein n is selected from 1, 2, 3, 4 and 5; and wherein n2 is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, and 10 ; and R10 is -N(R) 2 , wherein each R is independently selected from C1-6 alkyl, The group consisting of C 2-3 alkenyl and H;
Raα、Raβ和Raγ各自独立地选自由H、C1-12烷基和C2-12烯基组成的组,其中Raα、Raβ和Raγ中的至少一个选自由C1-12烷基和C2-12烯基组成的组;并且R aα , R aβ and R aγ are each independently selected from the group consisting of H, C 1-12 alkyl and C 2-12 alkenyl, wherein at least one of R aα , R aβ and R aγ is selected from C 1- the group consisting of 12 alkyl and C 2-12 alkenyl; and
R’是C1-12烷基或C2-12烯基。R' is C 1-12 alkyl or C 2-12 alkenyl.
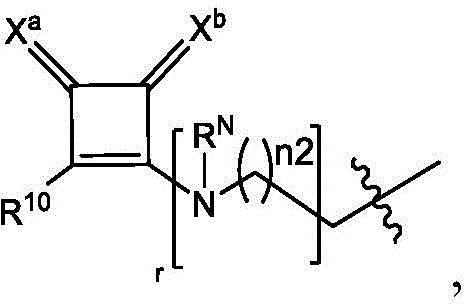
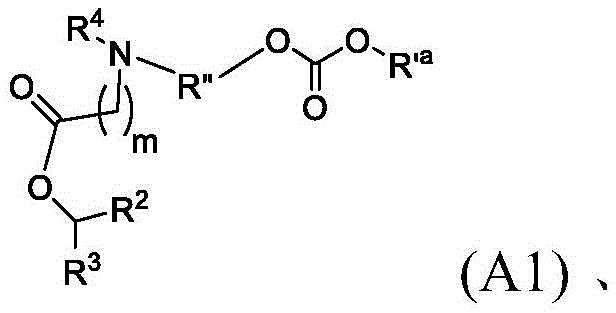
在一些方面,本公开涉及式(A1)、(A2)或(A3)的化合物:In some aspects, the present disclosure relates to compounds of formula (A1 ), (A2), or (A3):
其中in
m选自5、6、7、8和9;m is selected from 5, 6, 7, 8 and 9;
R2和R3各自独立地选自由H、C1-14烷基和C2-14烯基组成的组;R 2 and R 3 are each independently selected from the group consisting of H, C 1-14 alkyl and C 2-14 alkenyl;
R4选自-(CH2)nOH,其中n选自1、2、3、4和5,和其中n2选自1、2、3、4、5、6、7、8、9和10;并且R10是-N(R)2,其中每个R独立地选自由C1-6烷基、C2-3烯基和H组成的组;R4 is selected from -( CH2 )nOH, wherein n is selected from 1, 2, 3, 4 and 5, and wherein n2 is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, and 10 ; and R10 is -N(R) 2 , wherein each R is independently selected from C1-6 alkyl, The group consisting of C 2-3 alkenyl and H;
R'a是: R'a is:
Raα、Raβ和Raγ各自独立地选自由H、C1-12烷基和C2-12烯基组成的组,其中Raα、Raβ和Raγ中至少一个选自由C1-12烷基和C2-12烯基组成的组;R aα , R aβ and R aγ are each independently selected from the group consisting of H, C 1-12 alkyl and C 2-12 alkenyl, wherein at least one of R aα , R aβ and R aγ is selected from C 1-12 the group consisting of alkyl and C 2-12 alkenyl;
R”是任选地被OH取代的C3-C13烷基;并且R" is C3 - C13 alkyl optionally substituted with OH; and
R’是C1-12烷基或C2-12烯基。R' is C 1-12 alkyl or C 2-12 alkenyl.
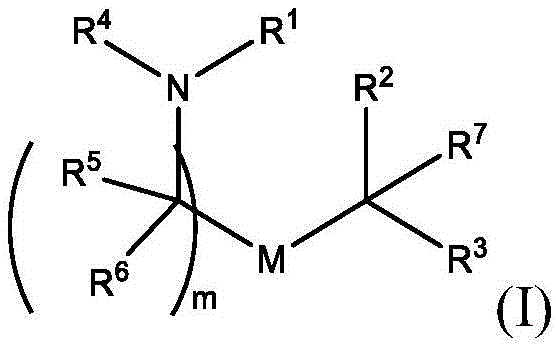
在一些方面,本公开涉及式(I)的化合物:In some aspects, the present disclosure relates to compounds of formula (I):
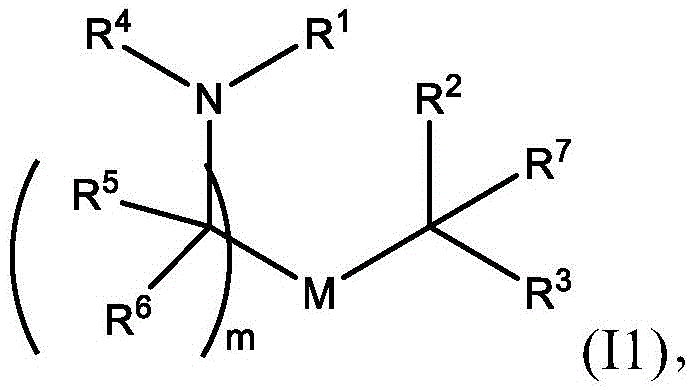
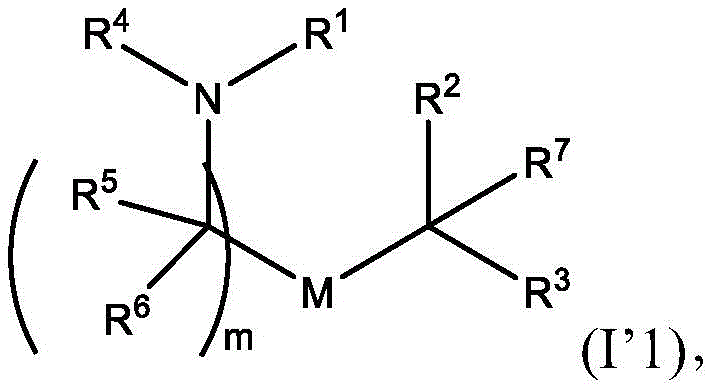
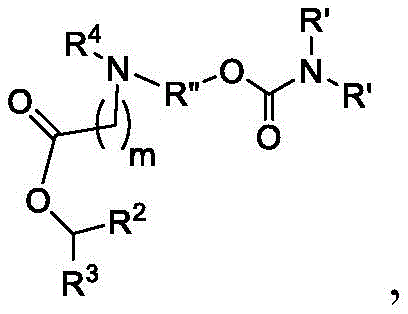
或其N-氧化物, or its N-oxide,
或它们的盐或异构体,其中:or their salts or isomers, wherein:
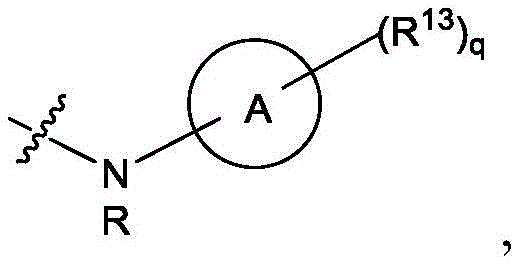
R1选自由C5-30烷基、C5-20烯基、-R*YR*”、-YR*”和-R”M’R’组成的组;R 1 is selected from the group consisting of C 5-30 alkyl, C 5-20 alkenyl, -R*YR*", -YR*" and -R"M'R';
R2和R3独立地选自由H、C1-14烷基、C2-14烯基、-R*YR*”、-YR*”和-R*OR*”组成的组,或者R2和R3连同其所连接的原子一起形成杂环或碳环;R 2 and R 3 are independently selected from the group consisting of H, C 1-14 alkyl, C 2-14 alkenyl, -R*YR*", -YR*" and -R*OR*", or R 2 and R3 together with the atoms to which it is attached form a heterocyclic or carbocyclic ring;
R4选自-(CH2)nOH,其中n选自1、2、3、4和5;和其中n2选自1、2、3、4、5、6、7、8、9和10;并且R10是-N(R)2,其中每个R独立地选自由C1-6烷基、C2-3烯基和H组成的组;R4 is selected from -( CH2 )nOH, wherein n is selected from 1, 2, 3, 4 and 5; and wherein n2 is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, and 10 ; and R10 is -N(R) 2 , wherein each R is independently selected from C1-6 alkyl, The group consisting of C 2-3 alkenyl and H;
每个R5独立地选自由OH、C1-3烷基、C2-3烯基和H组成的组;each R 5 is independently selected from the group consisting of OH, C 1-3 alkyl, C 2-3 alkenyl and H;
每个R6独立地选自由OH、C1-3烷基、C2-3烯基和H组成的组;each R is independently selected from the group consisting of OH, C 1-3 alkyl, C 2-3 alkenyl and H ;
M选自-OC(O)O-、-OC(O)-NRM-C(O)O-、-O-M”-O-、-OC(O)N(RM)-、-N(RM)C(O)O-、-NRMC(O)NRM-、-O-N=C(RM)-、SO-、-OS-、-S(RM)2O-、-O-S(RM)2-、-S(O)O-、-OS(O)-,其中M”是键、-(CH2)zC(O)-、C1-13烷基、C2-13烯基、-B(R**)-、-Si(R**)2-、-S(R**)2-或-S(O)-,其中z是1、2、3或4;M is selected from -OC(O)O-, -OC(O)-NR M -C(O)O-, -OM"-O-, -OC(O)N(R M )-, -N(R M )C(O)O-, -NR M C(O)NR M -, -ON=C(R M )-, SO-, -OS-, -S(R M ) 2 O-, -OS( R M ) 2 -, -S(O)O-, -OS(O)-, wherein M" is a bond, -(CH 2 ) z C(O)-, C 1-13 alkyl, C 2-13 alkenyl, -B(R**)-, -Si(R**) 2- , -S(R**) 2- or -S(O)-, wherein z is 1, 2, 3 or 4;
M’选自–OC(O)O-、-C(O)O-、-OC(O)-、-OC(O)-M”-C(O)O-、-OC(O)-NRM-C(O)O-、-O-M”-O-、-C(O)N(RM)-、-N(RM)C(O)-、-OC(O)N(RM)-、-N(RM)C(O)O-、-NRMC(O)NRM-、-O-N=C(RM)-、-C(O)-、-C(S)-、-C(S)S-、-SC(S)-、-CH(OH)-、-P(O)(ORM)O-、-S(O)2-、-S-S-、-SO-、-OS-、S(RM)2O-、-O-S(RM)2-、-S(O)O-、-OS(O)-、芳基和杂芳基,其中M”是键、-(CH2)zC(O)-、C1-13烷基、C2-13烯基、-B(R**)-、-Si(R**)2-、-S(R**)2-或-S(O)-,其中z是1、2、3或4;M' is selected from -OC(O)O-, -C(O)O-, -OC(O)-, -OC(O)-M"-C(O)O-, -OC(O)-NR M -C(O)O-, -OM"-O-, -C(O)N(R M )-, -N(R M )C(O)-, -OC(O)N(R M ) -, -N(R M )C(O)O-, -NR M C(O)NR M -, -ON=C(R M )-, -C(O)-, -C(S)-, -C(S)S-, -SC(S)-, -CH(OH)-, -P(O)(OR M )O-, -S(O) 2 -, -SS-, -SO-, -OS-, S(R M ) 2 O-, -OS(R M ) 2 -, -S(O)O-, -OS(O)-, aryl and heteroaryl, where M" is a bond, -(CH 2 ) z C(O)-, C 1-13 alkyl, C 2-13 alkenyl, -B(R**)-, -Si(R**) 2 -, -S(R* *) 2- or -S(O)-, where z is 1, 2, 3 or 4;
R7选自由C1-3烷基、C2-3烯基和H组成的组;R 7 is selected from the group consisting of C 1-3 alkyl, C 2-3 alkenyl and H;
每个R独立地选自由C1-6烷基、C1-3烷基-芳基、C2-3烯基和H组成的组;each R is independently selected from the group consisting of C1-6 alkyl, C1-3 alkyl-aryl, C2-3 alkenyl, and H;
每个R’独立地选自由C1-18烷基、C2-18烯基、-R*YR*”、-YR*”、(CH2)qOR*和H组成的组,each R' is independently selected from the group consisting of C 1-18 alkyl, C 2-18 alkenyl, -R*YR*", -YR*", (CH 2 ) q OR* and H,
并且每个q独立地选自1、2和3;and each q is independently selected from 1, 2 and 3;
每个RM独立地选自由H、C1-6烷基和C2-6烯基组成的组;each R M is independently selected from the group consisting of H, C 1-6 alkyl and C 2-6 alkenyl;
每个R”独立地选自由C3-15烷基和C3-15烯基组成的组;each R" is independently selected from the group consisting of C 3-15 alkyl and C 3-15 alkenyl;
每个R*”独立地选自由C1-15烷基和C2-15烯基组成的组;each R*" is independently selected from the group consisting of C 1-15 alkyl and C 2-15 alkenyl;
每个R*独立地选自由C1-12烷基和C2-12烯基组成的组;each R* is independently selected from the group consisting of C 1-12 alkyl and C 2-12 alkenyl;
每个R**独立地选自由H、OH、C1-12烷基、C2-12烯基、(CH2)qOR*和(CH2)qOH组成的组;each R** is independently selected from the group consisting of H, OH, C 1-12 alkyl, C 2-12 alkenyl, (CH 2 ) q OR*, and (CH 2 ) q OH;
每个Y独立地是C3-6碳环;并且each Y is independently a C3-6 carbocycle; and
m选自5、6、7、8、9、10、11、12和13。m is selected from 5, 6, 7, 8, 9, 10, 11, 12 and 13.
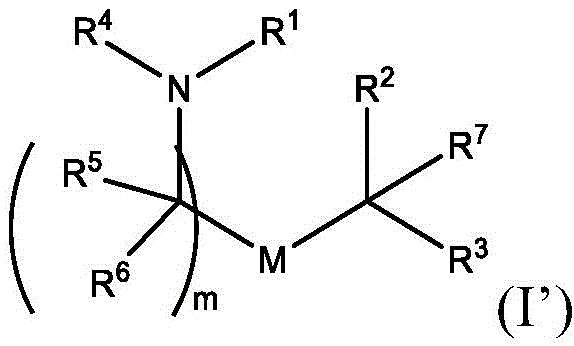
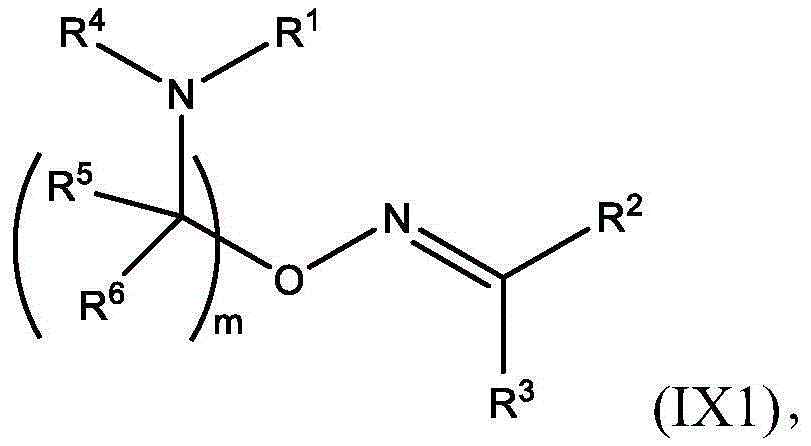
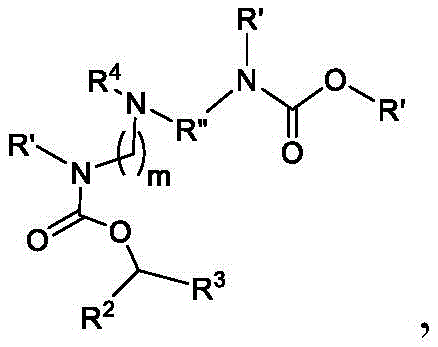
在一些方面,本公开涉及式(I’)的化合物:In some aspects, the present disclosure relates to compounds of formula (I'):
或其N-氧化物, or its N-oxide,
或它们的盐或异构体,其中:or their salts or isomers, wherein:
R1选自由C5-30烷基、C5-20烯基、-R*YR*”、-YR*”和-R”M’R’组成的组;R 1 is selected from the group consisting of C 5-30 alkyl, C 5-20 alkenyl, -R*YR*", -YR*" and -R"M'R';
R2和R3独立地选自由H、C1-14烷基、C2-14烯基、-R*YR*”、-YR*”和-R*OR*”组成的组,或者R2和R3连同其所连接的原子一起形成杂环或碳环;R 2 and R 3 are independently selected from the group consisting of H, C 1-14 alkyl, C 2-14 alkenyl, -R*YR*", -YR*" and -R*OR*", or R 2 and R3 together with the atoms to which it is attached form a heterocyclic or carbocyclic ring;
R4选自-(CH2)nOH,其中n选自1、2、3、4和5;和其中n2选自1、2、3、4、5、6、7、8、9和10;并且R10是-N(R)2,其中每个R独立地选自由C1-6烷基、C2-3烯基和H组成的组;R4 is selected from -( CH2 )nOH, wherein n is selected from 1, 2, 3, 4 and 5; and wherein n2 is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, and 10 ; and R10 is -N(R) 2 , wherein each R is independently selected from C1-6 alkyl, The group consisting of C 2-3 alkenyl and H;
每个R5独立地选自由OH、C1-3烷基、C2-3烯基和H组成的组;each R 5 is independently selected from the group consisting of OH, C 1-3 alkyl, C 2-3 alkenyl and H;
每个R6独立地选自由OH、C1-3烷基、C2-3烯基和H组成的组;each R is independently selected from the group consisting of OH, C 1-3 alkyl, C 2-3 alkenyl and H ;
M选自–OC(O)O-、-C(O)O-、-OC(O)-、-OC(O)-M”-C(O)O-、-OC(O)-NRM-C(O)O-、-O-M”-O-、-C(O)N(RM)-、-N(RM)C(O)-、-OC(O)N(RM)-、-N(RM)C(O)O-、-NRMC(O)NRM-、-O-N=C(RM)-、-C(O)-、-C(S)-、-C(S)S-、-SC(S)-、-CH(OH)-、-P(O)(ORM)O-、-S(O)2-、-S-S-,-SO-、-OS-、S(RM)2O-、-O-S(RM)2-、-S(O)O-、-OS(O)-、芳基和杂芳基,其中M”是键、-(CH2)zC(O)-、C1-13烷基、C2-13烯基、-B(R**)-、-Si(R**)2-、-S(R**)2-或-S(O)-,其中z是1、2、3、或4;M is selected from -OC(O)O-, -C(O)O-, -OC(O)-, -OC(O)-M"-C(O)O-, -OC(O)-NR M -C(O)O-, -OM"-O-, -C(O)N(R M )-, -N(R M )C(O)-, -OC(O)N(R M )- , -N(R M )C(O)O-, -NR M C(O)NR M -, -ON=C(R M )-, -C(O)-, -C(S)-, - C(S)S-, -SC(S)-, -CH(OH)-, -P(O)(OR M )O-, -S(O) 2 -, -SS-, -SO-, - OS-, S(R M ) 2 O-, -OS(R M ) 2 -, -S(O)O-, -OS(O)-, aryl and heteroaryl, where M" is a bond, - (CH 2 ) z C(O)-, C 1-13 alkyl, C 2-13 alkenyl, -B(R**)-, -Si(R**) 2 -, -S(R** ) 2- or -S(O)-, wherein z is 1, 2, 3, or 4;
M’选自–OC(O)O-、-OC(O)-NRM-C(O)O-、-O-M”-O-、-OC(O)N(RM)-、-N(RM)C(O)O-、-NRMC(O)NRM-、-O-N=C(RM)-、SO-、-OS-、-S(RM)2O-、-O-S(RM)2-、-S(O)O-、-OS(O)-,其中M”是键、-(CH2)zC(O)-、C1-13烷基、C2-13烯基、-B(R**)-、-Si(R**)2-、-S(R**)2-或-S(O)-,其中z是1、2、3、或4;M' is selected from -OC(O)O-, -OC(O)-NR M -C(O)O-, -OM"-O-, -OC(O)N(R M )-, -N( R M )C(O)O-, -NR M C(O)NR M -, -ON=C(R M )-, SO-, -OS-, -S(R M ) 2 O-, -OS (R M ) 2 -, -S(O)O-, -OS(O)-, wherein M" is a bond, -(CH 2 ) z C(O)-, C 1-13 alkyl, C 2- 13Alkenyl , -B(R**)-, -Si(R**) 2- , -S(R**) 2- or -S(O)-, wherein z is 1, 2, 3, or 4;
R7选自由C1-3烷基、C2-3烯基和H组成的组;R 7 is selected from the group consisting of C 1-3 alkyl, C 2-3 alkenyl and H;
每个R’独立地选自由C1-18烷基、C2-18烯基、-R*YR*”、-YR*”、(CH2)qOR*和H组成的组,each R' is independently selected from the group consisting of C 1-18 alkyl, C 2-18 alkenyl, -R*YR*", -YR*", (CH 2 ) q OR* and H,
并且每个q独立地选自1、2和3;and each q is independently selected from 1, 2 and 3;
每个RM独立地选自由H、C1-6烷基和C2-6烯基组成的组;each R M is independently selected from the group consisting of H, C 1-6 alkyl and C 2-6 alkenyl;
每个R”独立地选自由C3-15烷基和C3-15烯基组成的组;each R" is independently selected from the group consisting of C 3-15 alkyl and C 3-15 alkenyl;
每个R*”独立地选自由C1-15烷基和C2-15烯基组成的组;each R*" is independently selected from the group consisting of C 1-15 alkyl and C 2-15 alkenyl;
每个R*独立地选自由C1-12烷基和C2-12烯基组成的组;each R* is independently selected from the group consisting of C 1-12 alkyl and C 2-12 alkenyl;
每个R**独立地选自由H、OH、C1-12烷基、C2-12烯基、(CH2)qOR*和(CH2)qOH组成的组;each R** is independently selected from the group consisting of H, OH, C 1-12 alkyl, C 2-12 alkenyl, (CH 2 ) q OR*, and (CH 2 ) q OH;
每个Y独立地是C3-6碳环;each Y is independently a C3-6 carbocycle;
m选自5、6、7、8、9、10、11、12和13。m is selected from 5, 6, 7, 8, 9, 10, 11, 12 and 13.
在一些方面,本公开涉及式(I-X)的化合物:In some aspects, the present disclosure relates to compounds of formula (I-X):
或其N-氧化物,或它们的盐或异构体,其中 or their N-oxides, or their salts or isomers, wherein
R1选自由C5-30烷基、C5-20烯基、-R*YR*”、-YR*”和-R”M’R’组成的组;R 1 is selected from the group consisting of C 5-30 alkyl, C 5-20 alkenyl, -R*YR*", -YR*" and -R"M'R';
R2和R3独立地选自由H、C1-14烷基、C2-14烯基、-R*YR*”、-YR*”和-R*OR*”组成的组,或者R2和R3连同其所连接的原子一起形成杂环或碳环;R 2 and R 3 are independently selected from the group consisting of H, C 1-14 alkyl, C 2-14 alkenyl, -R*YR*", -YR*" and -R*OR*", or R 2 and R3 together with the atoms to which it is attached form a heterocyclic or carbocyclic ring;
R4选自-(CH2)nOH,其中n选自1、2、3、4和5;和其中n2选自1、2、3、4、5、6、7、8、9和10;并且R10是-N(R)2,其中每个R独立地选自由C1-6烷基、C2-3烯基和H组成的组;R4 is selected from -( CH2 )nOH, wherein n is selected from 1, 2, 3, 4 and 5; and wherein n2 is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, and 10 ; and R10 is -N(R) 2 , wherein each R is independently selected from C1-6 alkyl, The group consisting of C 2-3 alkenyl and H;
每个R5独立地选自由OH、C1-3烷基、C2-3烯基和H组成的组;each R 5 is independently selected from the group consisting of OH, C 1-3 alkyl, C 2-3 alkenyl and H;
每个R6独立地选自由OH、C1-3烷基、C2-3烯基和H组成的组;each R is independently selected from the group consisting of OH, C 1-3 alkyl, C 2-3 alkenyl and H ;
M’选自–OC(O)O-、-C(O)O-、-OC(O)-、-OC(O)-M”-C(O)O-、-OC(O)-NRM-C(O)O-、-O-M”-O-、-C(O)N(RM)-、-N(RM)C(O)-、-OC(O)N(RM)-、-N(RM)C(O)O-、-NRMC(O)NRM-、-O-N=C(RM)-、-C(O)-、-C(S)-、-C(S)S-、-SC(S)-、-CH(OH)-、-P(O)(ORM)O-、-S(O)2-、-S-S-、-SO-、-OS-、S(RM)2O-、-O-S(RM)2-、-S(O)O-、-OS(O)-、芳基和杂芳基,其中M”是键、-(CH2)zC(O)-、C1-13烷基、C2-13烯基、-B(R**)-、-Si(R**)2-、-S(R**)2-或-S(O)-,其中z是1、2、3或4;M' is selected from -OC(O)O-, -C(O)O-, -OC(O)-, -OC(O)-M"-C(O)O-, -OC(O)-NR M -C(O)O-, -OM"-O-, -C(O)N(R M )-, -N(R M )C(O)-, -OC(O)N(R M ) -, -N(R M )C(O)O-, -NR M C(O)NR M -, -ON=C(R M )-, -C(O)-, -C(S)-, -C(S)S-, -SC(S)-, -CH(OH)-, -P(O)(OR M )O-, -S(O) 2 -, -SS-, -SO-, -OS-, S(R M ) 2 O-, -OS(R M ) 2 -, -S(O)O-, -OS(O)-, aryl and heteroaryl, where M" is a bond, -(CH 2 ) z C(O)-, C 1-13 alkyl, C 2-13 alkenyl, -B(R**)-, -Si(R**) 2 -, -S(R* *) 2- or -S(O)-, where z is 1, 2, 3 or 4;
每个R’独立地选自由C1-18烷基、C2-18烯基、-R*YR*”、-YR*”、(CH2)qOR*和H组成的组,each R' is independently selected from the group consisting of C 1-18 alkyl, C 2-18 alkenyl, -R*YR*", -YR*", (CH 2 ) q OR* and H,
并且每个q独立地选自1、2和3;and each q is independently selected from 1, 2 and 3;
每个RM独立地选自由H、C1-6烷基和C2-6烯基组成的组;each R M is independently selected from the group consisting of H, C 1-6 alkyl and C 2-6 alkenyl;
每个R”独立地选自由C3-15烷基和C3-15烯基组成的组;each R" is independently selected from the group consisting of C 3-15 alkyl and C 3-15 alkenyl;
每个R*”独立地选自由C1-15烷基和C2-15烯基组成的组;each R*" is independently selected from the group consisting of C 1-15 alkyl and C 2-15 alkenyl;
每个R*独立地选自由C1-12烷基和C2-12烯基组成的组;each R* is independently selected from the group consisting of C 1-12 alkyl and C 2-12 alkenyl;
每个R**独立地选自由H、OH、C1-12烷基、C2-12烯基、(CH2)qOR*和(CH2)qOH组成的组;each R** is independently selected from the group consisting of H, OH, C 1-12 alkyl, C 2-12 alkenyl, (CH 2 ) q OR*, and (CH 2 ) q OH;
每个Y独立地是C3-6碳环;并且each Y is independently a C3-6 carbocycle; and
m选自5、6、7、8、9、10、11、12和13。m is selected from 5, 6, 7, 8, 9, 10, 11, 12 and 13.
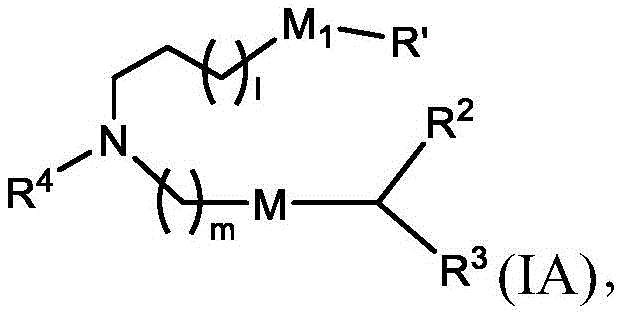
在某些实施方案中,式(I)或式(I')的化合物的子集包括式(IA)的化合物:In certain embodiments, the subset of compounds of formula (I) or formula (I') includes compounds of formula (IA):
或其N-氧化物,或它们的盐或异构体,其中l选自1、2、3、4和5;m选自5、6、7、8和9;并且R2和R3独立地选自由H、C1-14烷基和C2-14烯基组成的组。or an N-oxide thereof, or a salt or isomer thereof, wherein l is selected from 1, 2, 3, 4 and 5; m is selected from 5, 6, 7, 8 and 9 ; and R and R are independently is selected from the group consisting of H, C 1-14 alkyl and C 2-14 alkenyl.
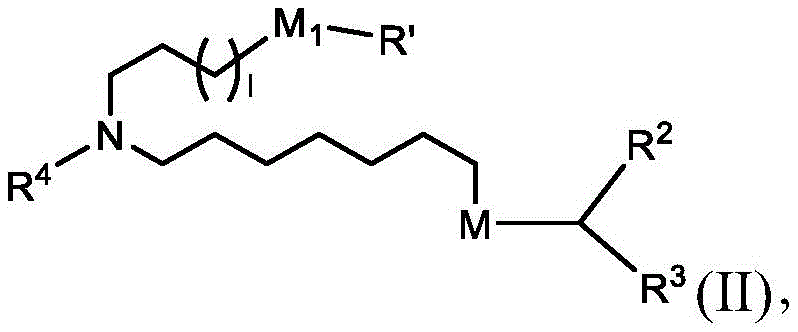
在某些实施方案中,式(I)或式(I')的化合物的子集包括式(II)的化合物:In certain embodiments, the subset of compounds of formula (I) or formula (I') includes compounds of formula (II):
或其N-氧化物,或它们的盐或异构体,其中I选1、2、3、4和5;并且R2和R3独立地选自由H、C1-14烷基和C2-14烯基组成的组。or N-oxides thereof, or their salts or isomers, wherein I is selected from 1, 2, 3, 4 and 5; and R 2 and R 3 are independently selected from H, C 1-14 alkyl and C 2 -14 group consisting of alkenyl groups.
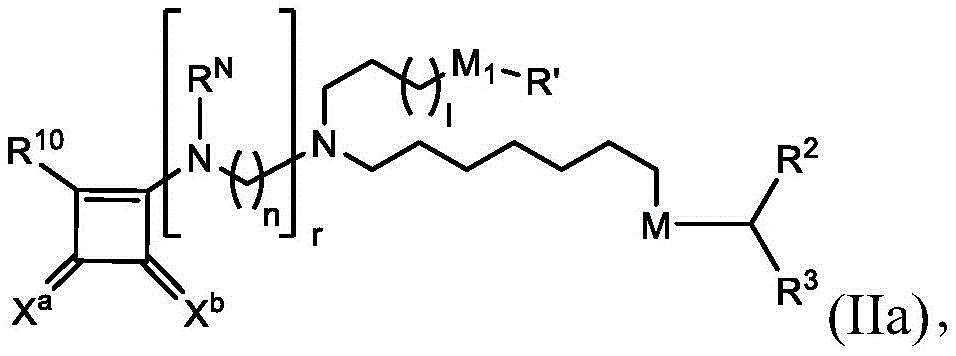
在其他实施方案中,式(II)的化合物的子集包括式(IIa)的化合物:In other embodiments, the subset of compounds of formula (II) includes compounds of formula (IIa):
或其N-氧化物,或它们的盐或异构体,其中or their N-oxides, or their salts or isomers, wherein
l选自1、2、3、4和5;l is selected from 1, 2, 3, 4 and 5;
M1是M';并且M 1 is M'; and
R2和R3独立地选自由H、C1-14烷基和C2-14烯基组成的组;R 2 and R 3 are independently selected from the group consisting of H, C 1-14 alkyl and C 2-14 alkenyl;
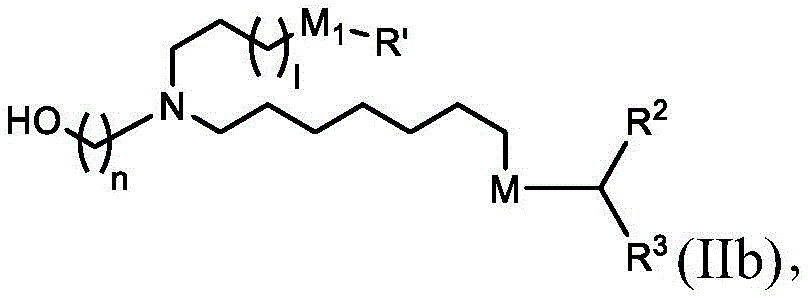
在其他实施方案中,式(II)的化合物的子集包括式(IIb)的化合物:In other embodiments, the subset of compounds of formula (II) includes compounds of formula (IIb):
或其N-氧化物,或它们的盐或异构体,其中or their N-oxides, or their salts or isomers, wherein
l选自1、2、3、4和5;l is selected from 1, 2, 3, 4 and 5;
M1是M';并且M 1 is M'; and
R2和R3独立地选自由H、C1-14烷基和C2-14烯基组成的组。R 2 and R 3 are independently selected from the group consisting of H, C 1-14 alkyl and C 2-14 alkenyl.
在其他实施方案中,式(I)或式(I')的化合物的子集包括式(Ia)的化合物:In other embodiments, the subset of compounds of Formula (I) or Formula (I') includes compounds of Formula (Ia):
或其N-氧化物,或它们的盐或异构体,其中R”是任选地被OH取代的直链或支链C3-C13烷基,并且m选自5、6、7、8和9。 or an N-oxide thereof, or a salt or isomer thereof, wherein R" is a straight or branched C3 - C13 alkyl optionally substituted with OH, and m is selected from 5, 6, 7, 8 and 9.
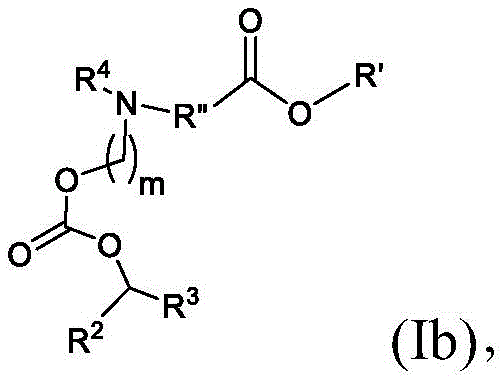
在其他实施方案中,式(I)或式(I')的化合物的子集包括式(Ib)的化合物:In other embodiments, the subset of compounds of formula (I) or formula (I') includes compounds of formula (Ib):
或其N-氧化物,或它们的盐或异构体,其中R”是任选地被OH取代的直链或支链C3-C13烷基,并且m选自5、6、7、8和9。 or an N-oxide thereof, or a salt or isomer thereof, wherein R" is a straight or branched C3 - C13 alkyl optionally substituted with OH, and m is selected from 5, 6, 7, 8 and 9.
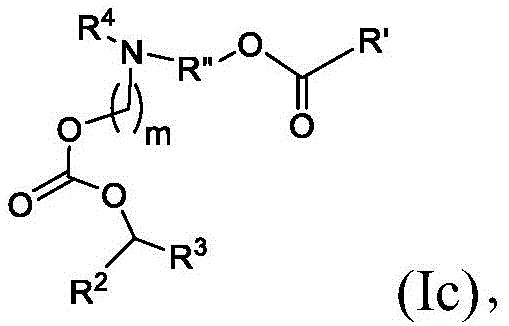
在其他实施方案中,式(I)或式(I')的化合物的子集包括式(Ic)的化合物:In other embodiments, the subset of compounds of formula (I) or formula (I') includes compounds of formula (Ic):
或其N-氧化物,或它们的盐或异构体,其中R”是任选地被OH取代的直链或支链C3-C13烷基,并且m选自5、6、7、8和9。 or an N-oxide thereof, or a salt or isomer thereof, wherein R" is a straight or branched C3 - C13 alkyl optionally substituted with OH, and m is selected from 5, 6, 7, 8 and 9.
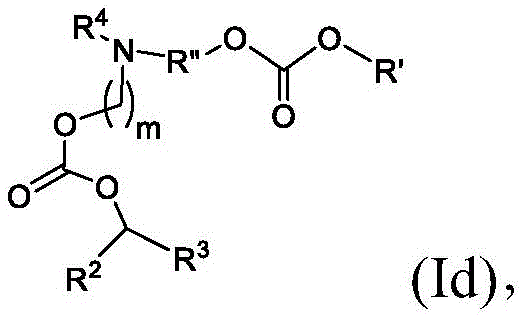
在其他实施方案中,式(I)或式(I')的化合物的子集包括式(Id)的化合物:In other embodiments, the subset of compounds of formula (I) or formula (I') includes compounds of formula (Id):
或其N-氧化物,或它们的盐或异构体,其中R”是任选地被OH取代的直链或支链C3-C13烷基,并且m选自5、6、7、8和9。 or an N-oxide thereof, or a salt or isomer thereof, wherein R" is a straight or branched C3 - C13 alkyl optionally substituted with OH, and m is selected from 5, 6, 7, 8 and 9.
在其他实施方案中,式(I)或式(I')的化合物的子集包括式(Ie)的化合物:In other embodiments, the subset of compounds of formula (I) or formula (I') includes compounds of formula (Ie):
或其N-氧化物,或它们的盐或异构体,其中R”是任选地被OH取代的直链或支链C3-C13烷基,并且m选自5、6、7、8和9。 or an N-oxide thereof, or a salt or isomer thereof, wherein R" is a straight or branched C3 - C13 alkyl optionally substituted with OH, and m is selected from 5, 6, 7, 8 and 9.
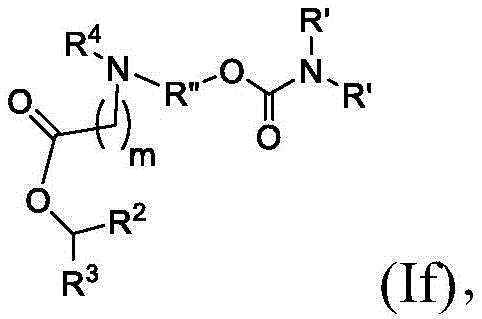
在其他实施方案中,式(I)或式(I')的化合物的子集包括式(If)的化合物:In other embodiments, the subset of compounds of formula (I) or formula (I') includes compounds of formula (If):
或其N-氧化物,或它们的盐或异构体,其中R”是任选地被OH取代的直链或支链C3-C13烷基,并且m选自5、6、7、8和9。 or an N-oxide thereof, or a salt or isomer thereof, wherein R" is a straight or branched C3 - C13 alkyl optionally substituted with OH, and m is selected from 5, 6, 7, 8 and 9.
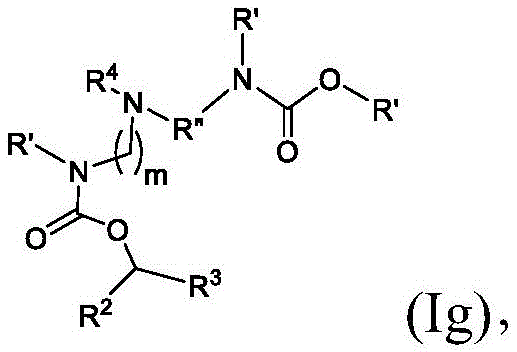
在其他实施方案中,式(I)或式(I')的化合物的子集包括式(Ig)的化合物:In other embodiments, the subset of compounds of formula (I) or formula (I') includes compounds of formula (Ig):
或其N-氧化物,或它们的盐或异构体,其中R”是任选地被OH取代的直链或支链C3-C13烷基,并且m选自5、6、7、8和9。 or an N-oxide thereof, or a salt or isomer thereof, wherein R" is a straight or branched C3 - C13 alkyl optionally substituted with OH, and m is selected from 5, 6, 7, 8 and 9.
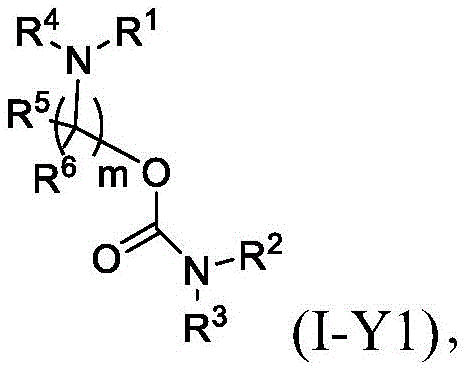
在一些方面,本公开涉及式(I-Y)的化合物:In some aspects, the present disclosure relates to compounds of formula (I-Y):
或其N-氧化物,或它们的盐或异构体,其中: or their N-oxides, or their salts or isomers, wherein:
R1选自由C5-30烷基、C5-20烯基、-R*YR*”、-YR*”和-R”M’R’组成的组;R 1 is selected from the group consisting of C 5-30 alkyl, C 5-20 alkenyl, -R*YR*", -YR*" and -R"M'R';
R2和R3独立地选自由H、C1-14烷基、C2-14烯基、-R*YR*”、-YR*”和-R*OR*”组成的组,或者R2和R3连同其所连接的原子一起形成杂环或碳环;R 2 and R 3 are independently selected from the group consisting of H, C 1-14 alkyl, C 2-14 alkenyl, -R*YR*", -YR*" and -R*OR*", or R 2 and R3 together with the atoms to which it is attached form a heterocyclic or carbocyclic ring;
R4选自-(CH2)nOH,其中n选自1、2、3、4和5;和其中n2选自1、2、3、4、5、6、7、8、9和10;并且R10是-N(R)2,其中每个R独立地选自由C1-6烷基、C2-3烯基和H组成的组;R4 is selected from -( CH2 )nOH, wherein n is selected from 1, 2, 3, 4 and 5; and wherein n2 is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, and 10 ; and R10 is -N(R) 2 , wherein each R is independently selected from C1-6 alkyl, The group consisting of C 2-3 alkenyl and H;
每个R5独立地选自由OH、C1-3烷基、C2-3烯基和H组成的组;each R 5 is independently selected from the group consisting of OH, C 1-3 alkyl, C 2-3 alkenyl and H;
每个R6独立地选自由OH、C1-3烷基、C2-3烯基和H组成的组;each R is independently selected from the group consisting of OH, C 1-3 alkyl, C 2-3 alkenyl and H ;
M’选自–OC(O)O-、-C(O)O-、-OC(O)-、-OC(O)-M”-C(O)O-、-OC(O)-NRM-C(O)O-、-O-M”-O-、-C(O)N(RM)-、-N(RM)C(O)-、-OC(O)N(RM)-、-N(RM)C(O)O-、-NRMC(O)NRM-、-O-N=C(RM)-、-C(O)-、-C(S)-、-C(S)S-、-SC(S)-、-CH(OH)-、-P(O)(ORM)O-、-S(O)2-、-S-S-、-SO-、-OS-、S(RM)2O-、-O-S(RM)2-、-S(O)O-、-OS(O)-、芳基和杂芳基,其中M”是键、-(CH2)zC(O)-、C1-13烷基、C2-13烯基、-B(R**)-、-Si(R**)2-、-S(R**)2-或-S(O)-,其中z是1、2、3或4;M' is selected from -OC(O)O-, -C(O)O-, -OC(O)-, -OC(O)-M"-C(O)O-, -OC(O)-NR M -C(O)O-, -OM"-O-, -C(O)N(R M )-, -N(R M )C(O)-, -OC(O)N(R M ) -, -N(R M )C(O)O-, -NR M C(O)NR M -, -ON=C(R M )-, -C(O)-, -C(S)-, -C(S)S-, -SC(S)-, -CH(OH)-, -P(O)(OR M )O-, -S(O) 2 -, -SS-, -SO-, -OS-, S(R M ) 2 O-, -OS(R M ) 2 -, -S(O)O-, -OS(O)-, aryl and heteroaryl, where M" is a bond, -(CH 2 ) z C(O)-, C 1-13 alkyl, C 2-13 alkenyl, -B(R**)-, -Si(R**) 2 -, -S(R* *) 2- or -S(O)-, where z is 1, 2, 3 or 4;
每个R’独立地选自由C1-18烷基、C2-18烯基、-R*YR*”、-YR*”、(CH2)qOR*和H组成的组,each R' is independently selected from the group consisting of C 1-18 alkyl, C 2-18 alkenyl, -R*YR*", -YR*", (CH 2 ) q OR* and H,
并且每个q独立地选自1、2和3;and each q is independently selected from 1, 2 and 3;
每个RM独立地选自由H、C1-6烷基和C2-6烯基组成的组;each R M is independently selected from the group consisting of H, C 1-6 alkyl and C 2-6 alkenyl;
每个R”独立地选自由C3-15烷基和C3-15烯基组成的组;each R" is independently selected from the group consisting of C 3-15 alkyl and C 3-15 alkenyl;
每个R*”独立地选自由C1-15烷基和C2-15烯基组成的组;each R*" is independently selected from the group consisting of C 1-15 alkyl and C 2-15 alkenyl;
每个R*独立地选自由C1-12烷基和C2-12烯基组成的组;each R* is independently selected from the group consisting of C 1-12 alkyl and C 2-12 alkenyl;
每个R**独立地选自由H、OH、C1-12烷基、C2-12烯基、(CH2)qOR*和(CH2)qOH组成的组;each R** is independently selected from the group consisting of H, OH, C 1-12 alkyl, C 2-12 alkenyl, (CH 2 ) q OR*, and (CH 2 ) q OH;
每个Y独立地是C3-6碳环;并且each Y is independently a C3-6 carbocycle; and
m选自5、6、7、8、9、10、11、12和13。m is selected from 5, 6, 7, 8, 9, 10, 11, 12 and 13.
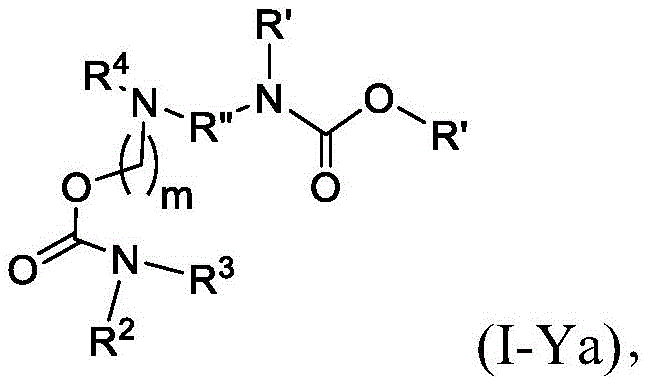
在其他实施方案中,式(I-Y)的化合物的子集包括式(I-Ya)的化合物:In other embodiments, the subset of compounds of formula (I-Y) includes compounds of formula (I-Ya):
或其N-氧化物,或它们的盐或异构体,其中每个R”独立地选自H、OH和C1-6烷基,l选自2、3、4、5和6,并且m选自5、6、7、8和9。 or an N-oxide thereof, or a salt or isomer thereof, wherein each R" is independently selected from H, OH, and C 1-6 alkyl, and l is selected from 2, 3, 4, 5, and 6, and m is selected from 5, 6, 7, 8 and 9.
式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)中任一个的化合物当适用时包括以下特征中的一个或多个。Formula (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y), (I-Ya) (IA), (Ia) Compounds of any one of , (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb) when applicable include one of the following features or more.
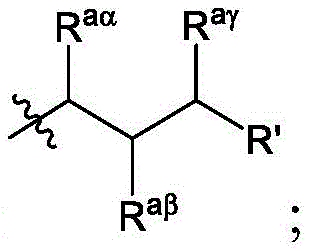
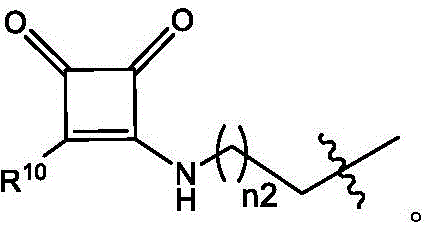
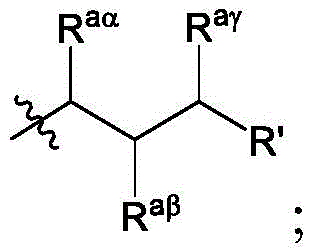
在一些实施方案中,R’a是:其中表示连接点;In some embodiments, R'a is: in represents a connection point;
Raα、Raβ和Raγ各自独立地选自由H、C1-12烷基和C2-12烯基组成的组,其中Raα、Raβ和Raγ中的至少一个选自由C1-12烷基和C2-12烯基组成的组;并且R aα , R aβ and R aγ are each independently selected from the group consisting of H, C 1-12 alkyl and C 2-12 alkenyl, wherein at least one of R aα , R aβ and R aγ is selected from C 1- the group consisting of 12 alkyl and C 2-12 alkenyl; and
R’是C1-12烷基或C2-12烯基组成的组。R' is the group consisting of C 1-12 alkyl or C 2-12 alkenyl.
在一些实施方案中,M和M'各自为-OC(O)O-。在一些实施方案中,M是-C(O)O-并且M’是-OC(O)O-。在一些实施方案中,M是-OC(O)O-并且M是-C(O)O-。In some embodiments, M and M' are each -OC(O)O-. In some embodiments, M is -C(O)O- and M' is -OC(O)O-. In some embodiments, M is -OC(O)O- and M is -C(O)O-.
在一些实施方案中,l是1、3或5。在一些实施方案中,l是1、3或4。在一些实施方案中,l是5。In some embodiments, 1 is 1, 3, or 5. In some embodiments, 1 is 1, 3, or 4. In some embodiments, 1 is 5.
在一些实施方案中,R4是 In some embodiments, R 4 is
在一些实施方案中,R4是-(CH2)2OH。In some embodiments, R 4 is -(CH 2 ) 2 OH.
在一些实施方案中,R2和R3独立地是C3-14烷基或C3-14烯基。In some embodiments, R 2 and R 3 are independently C 3-14 alkyl or C 3-14 alkenyl.
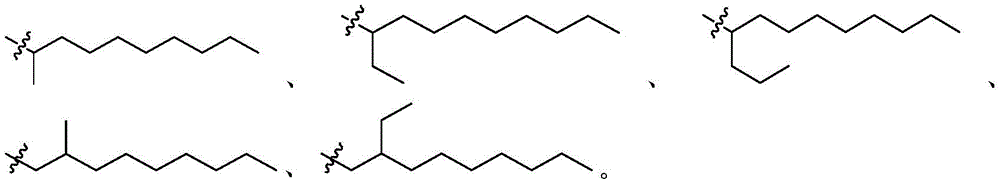
在一些实施方案中,R'a选自C4烷基、C4烯基、C5烷基、C5烯基、C6烷基、C6烯基、C7烷基、C7烯基、C9烷基、C9烯基、C11烷基、C11烯基、C17烷基、C17烯基、C18烷基和C18烯基,其各自是直链或支链的。In some embodiments, R'a is selected from C4 alkyl, C4 alkenyl, C5 alkyl, C5 alkenyl, C6 alkyl, C6 alkenyl, C7 alkyl, C7 alkenyl , C9 alkyl, C9 alkenyl, C11 alkyl, C11 alkenyl, C17 alkyl, C17 alkenyl, C18 alkyl and C18 alkenyl, each of which is straight or branched .
在一些实施方案中,R’是C4烷基或C4烯基。在一些实施方案中,R’是C5烷基或C5烯基。在一些实施方案中,R’是C6烷基或C6烯基。在一些实施方案中,R’是C7烷基或C7烯基。在一些实施方案中,R’是C8烷基或C8烯基。在一些实施方案中,R’是C9烷基或C9烯基。在一些实施方案中,R’是C10烷基或C10烯基。在一些实施方案中,R’是C11烷基或C11烯基。In some embodiments, R' is C4 alkyl or C4 alkenyl. In some embodiments, R' is C5 alkyl or C5 alkenyl. In some embodiments, R' is C6 alkyl or C6 alkenyl. In some embodiments, R' is C7 alkyl or C7 alkenyl. In some embodiments, R' is C8 alkyl or C8 alkenyl. In some embodiments, R' is C9 alkyl or C9 alkenyl. In some embodiments, R' is C10 alkyl or C10 alkenyl. In some embodiments, R' is C 11 alkyl or C 11 alkenyl.
在某些实施方案中,R’a是支链C1-18烷基。例如,R'a是In certain embodiments, R'a is branched C 1-18 alkyl. For example, R'a is
在一些实施方案中,R”独立地选自由C3-15烷基和C3-15烯基组成的组。在一些实施方案中,R”是C3烷基、C4烷基、C5烷基、C6烷基、C7烷基或C8烷基。在一些实施方案中,R”是C9烷基、C10烷基、C11烷基、C12烷基、C13烷基、C14烷基或C15烷基。In some embodiments, R" is independently selected from the group consisting of C3-15 alkyl and C3-15 alkenyl. In some embodiments, R " is C3 alkyl, C4 alkyl, C5 alkyl, C6 alkyl, C7 alkyl or C8 alkyl. In some embodiments, R" is C9 alkyl, C10 alkyl, C11 alkyl, C12 alkyl, C13 alkyl, C14 alkyl , or C15 alkyl.
在一些实施方案中,R2和R3独立地是C5-14烷基或C5-14烯基。In some embodiments, R 2 and R 3 are independently C 5-14 alkyl or C 5-14 alkenyl.
在一些实施方案中,R2与R3相同。在一些实施方案中,R2和R3是C8烷基。在某些实施方案中,R2和R3是C2烷基。在其他实施方案中,R2和R3是C3烷基。在一些实施方案中,R2和R3是C4烷基。在某些实施方案中,R2和R3是C5烷基。在其他实施方案中,R2和R3是C6烷基。在一些实施方案中,R2和R3是C7烷基。In some embodiments, R 2 is the same as R 3 . In some embodiments, R 2 and R 3 are C 8 alkyl. In certain embodiments, R 2 and R 3 are C 2 alkyl. In other embodiments, R 2 and R 3 are C 3 alkyl. In some embodiments, R 2 and R 3 are C 4 alkyl. In certain embodiments, R 2 and R 3 are C 5 alkyl. In other embodiments, R 2 and R 3 are C 6 alkyl. In some embodiments, R 2 and R 3 are C 7 alkyl.
在其他实施方案中,R2与R3不同。在某些实施方案中,R2是C8烷基。在一些实施方案中,R3是C1-7(例如,C1、C2、C3、C4、C5、C6或C7烷基)或C9烷基。In other embodiments, R 2 is different from R 3 . In certain embodiments, R 2 is C 8 alkyl. In some embodiments, R 3 is C 1-7 (eg, C 1 , C 2 , C 3 , C 4 , C 5 , C 6 , or C 7 alkyl) or C 9 alkyl.
在一些实施方案中,R3是C1烷基。在一些实施方案中,R3是C2烷基。在一些实施方案中,R3是C3烷基。在一些实施方案中,R3是C4烷基。在一些实施方案中,R3是C5烷基。在一些实施方案中,R3是C6烷基。在一些实施方案中,R3是C7烷基。在一些实施方案中,R3是C9烷基。In some embodiments, R 3 is C 1 alkyl. In some embodiments, R 3 is C 2 alkyl. In some embodiments, R 3 is C 3 alkyl. In some embodiments, R 3 is C 4 alkyl. In some embodiments, R 3 is C 5 alkyl. In some embodiments, R 3 is C 6 alkyl. In some embodiments, R 3 is C 7 alkyl. In some embodiments, R 3 is C 9 alkyl.
在一些实施方案中,m是5、6、7、8或9。在一些实施方案中,m是5、7或9。例如,在一些实施方案中,m是5。例如,在一些实施方案中,m是7。例如,在一些实施方案中,m是9。In some embodiments, m is 5, 6, 7, 8, or 9. In some embodiments, m is 5, 7 or 9. For example, in some embodiments, m is 5. For example, in some embodiments, m is 7. For example, in some embodiments, m is 9.
在一些实施方案中,n是2、3或4。在一些实施方案中,n是2。在一些实施方案中,n是4。在一些实施方案中,n不是3。In some embodiments, n is 2, 3, or 4. In some embodiments, n is 2. In some embodiments, n is 4. In some embodiments, n is not 3.
在一些实施方案中,n2为2、3或4。在一些实施方案中,n2是2。在一些实施方案中,n2是4。在一些实施方案中,n2不是3。In some embodiments, n2 is 2, 3, or 4. In some embodiments, n2 is 2. In some embodiments, n2 is 4. In some embodiments, n2 is not 3.
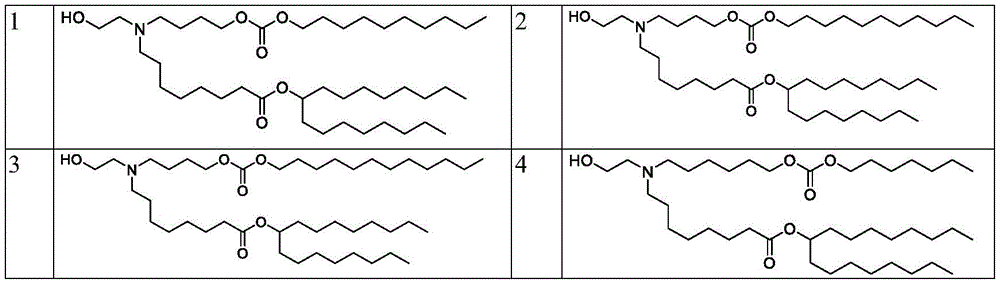
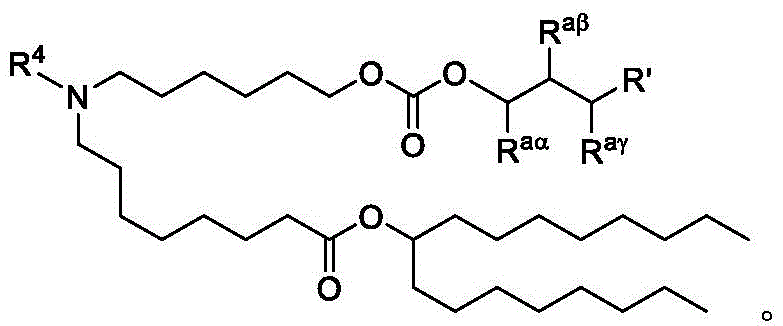
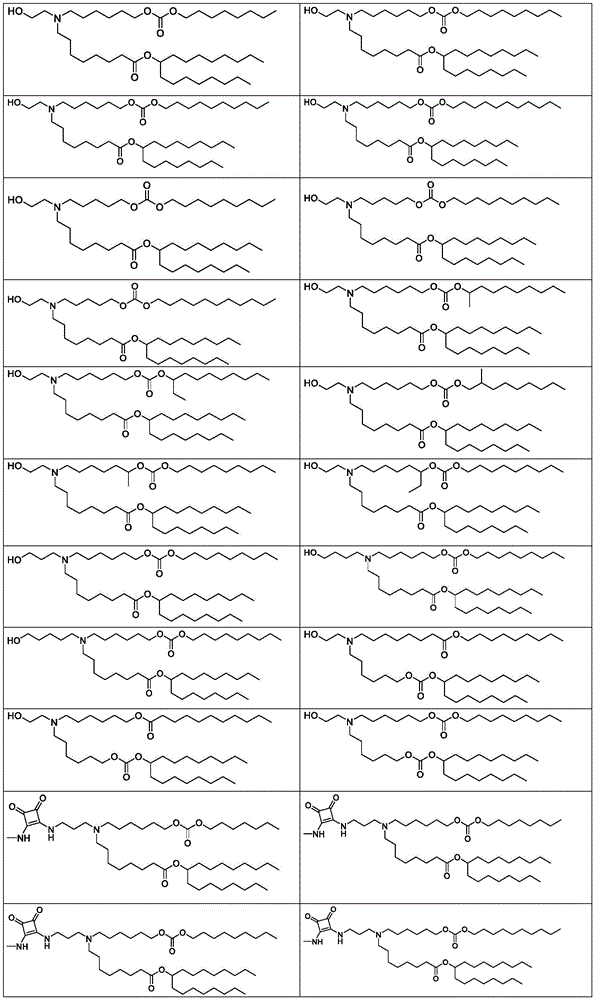
在一些实施方案中,式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合选自表1。In some embodiments, formula (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y), (I-Ya) ( The compounds of IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb) are selected from Table 1.
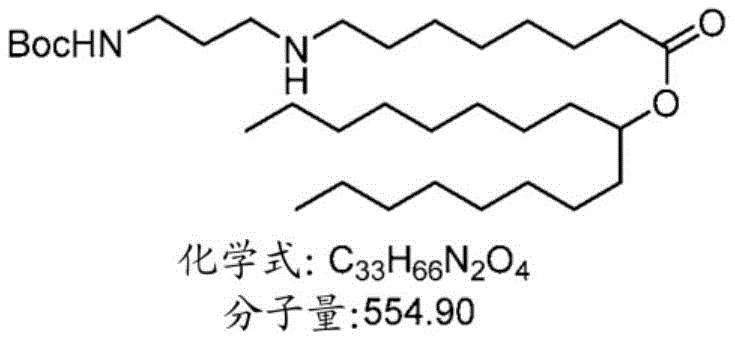
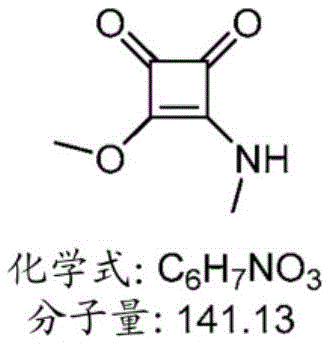
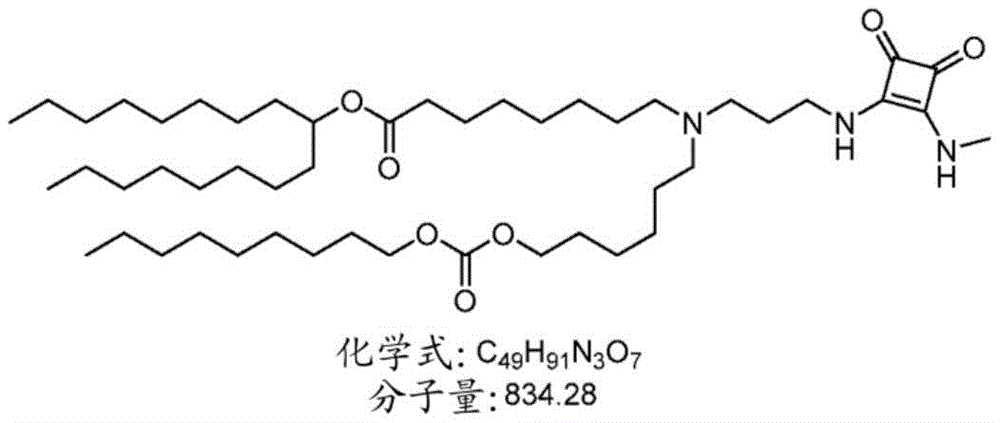
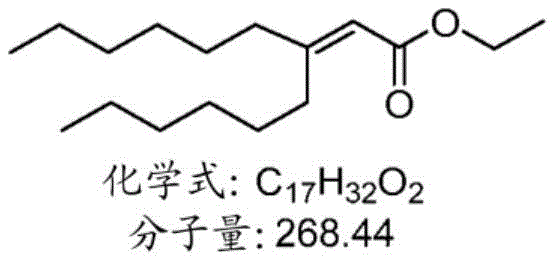
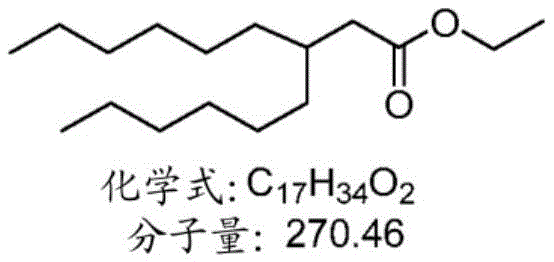
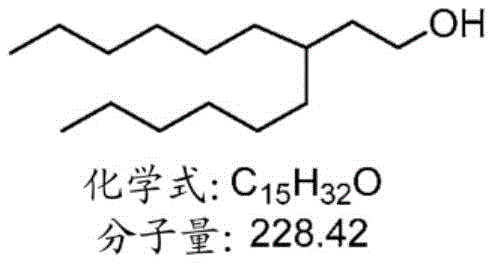
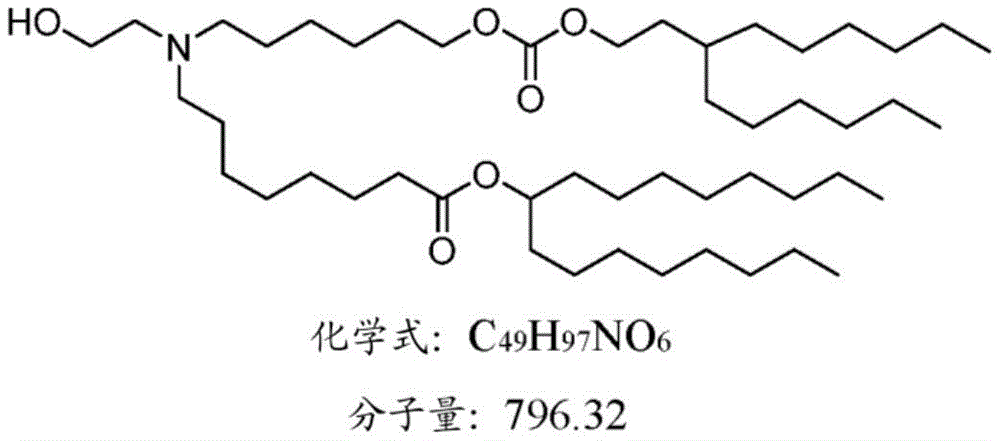
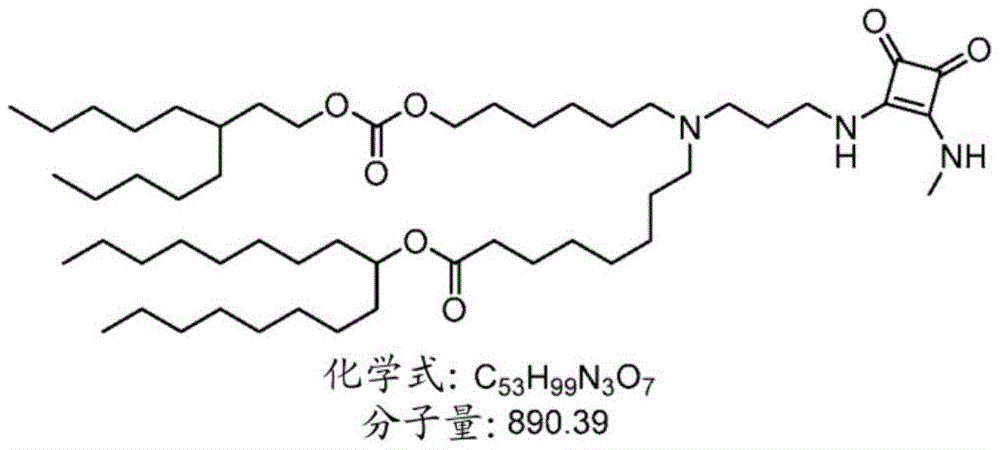
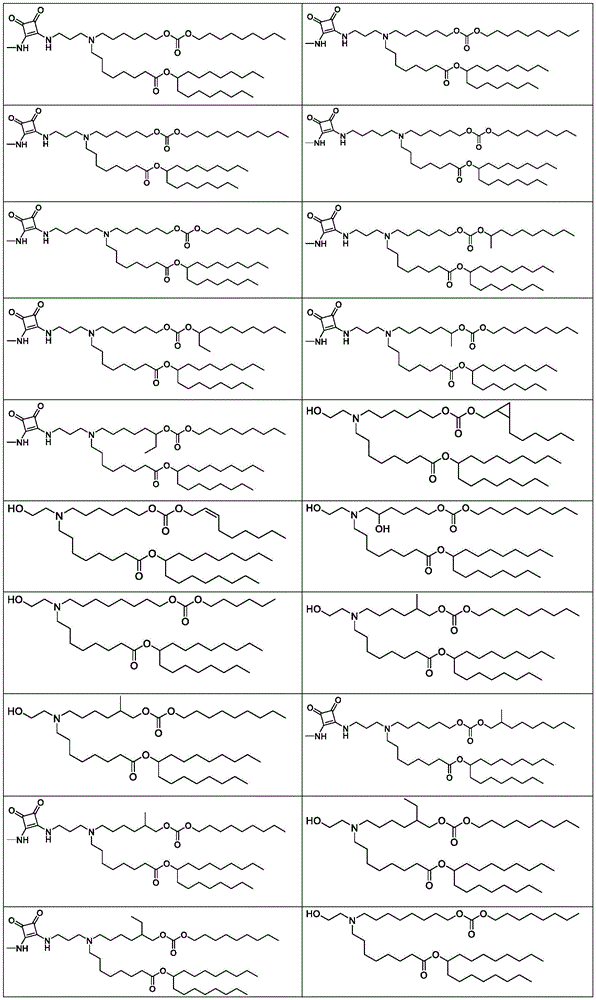
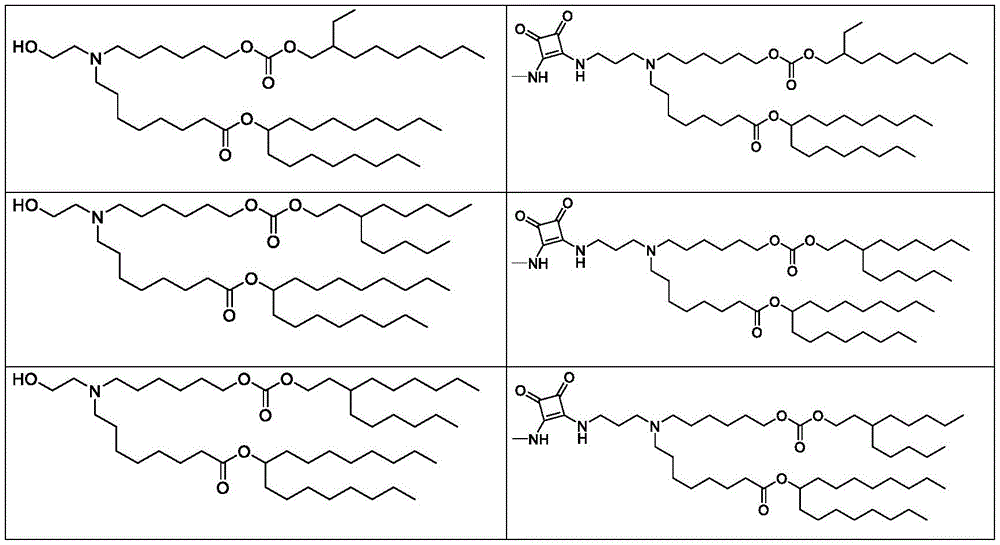
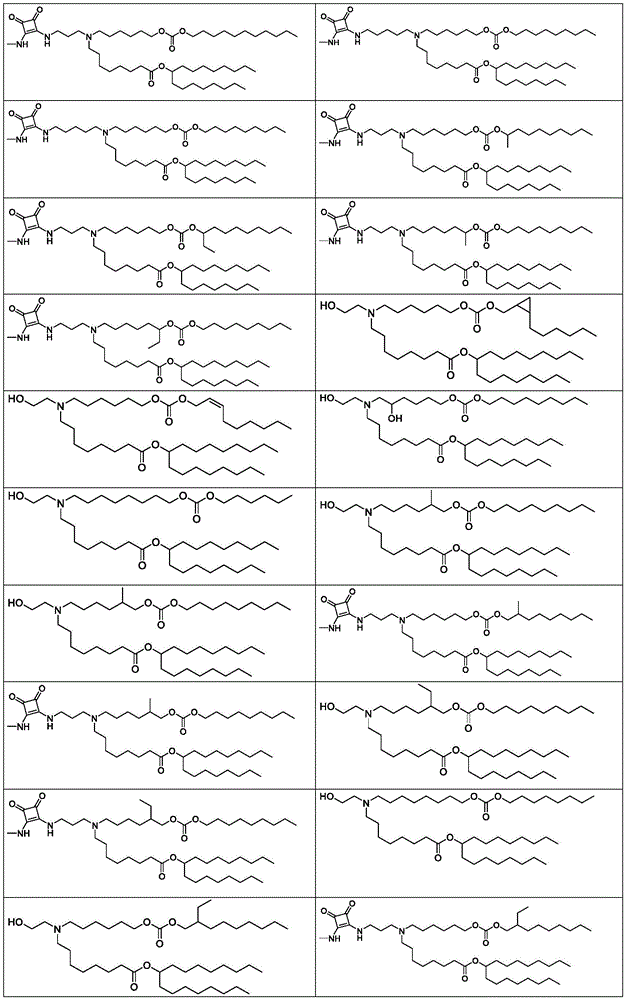
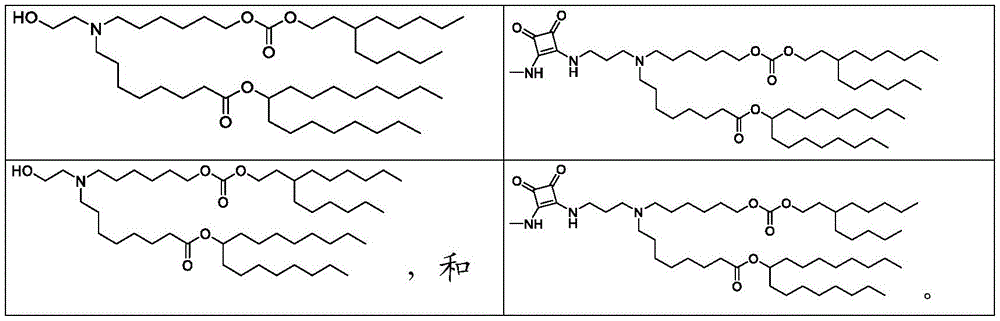
表1:氨基脂质Table 1: Amino lipids
根据式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的脂质的中心胺部分可以在生理pH下质子化。因此,脂质可以在生理pH下带正电荷或部分正电荷。此类脂质可以称为阳离子或可离子化的(氨基)脂质。脂质还可以是两性离子的,即,既带正电荷又带负电荷的中性分子。According to formulae (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y), (I-Ya) (IA), (Ia ), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb) The central amine moiety of the lipid can be protonated at physiological pH change. Thus, lipids can be positively or partially positively charged at physiological pH. Such lipids may be referred to as cationic or ionizable (amino) lipids. Lipids can also be zwitterionic, ie, neutral molecules that are both positively and negatively charged.
定义definition
如本文所用,术语“烷基”或“烷基基团”意指包含一个或多个碳原子(例如,一个、两个、三个、四个、五个、六个、七个、八个、九个、十个、十一个、十二个、十三个、十四个、十五个、十六个、十七个、十八个、十九个、二十个或更多个碳原子)的任选取代的直链或支链饱和烃。标记“C1-14烷基”意指包含1-14个碳原子的任选取代的直链或支链饱和烃。除非另外说明,否则本文所述的烷基是指未取代和取代的烷基两者。As used herein, the term "alkyl" or "alkyl group" means comprising one or more carbon atoms (eg, one, two, three, four, five, six, seven, eight , nine, ten, eleven, twelve, thirteen, fourteen, fifteen, sixteen, seventeen, eighteen, nineteen, twenty or more carbon atoms) optionally substituted straight-chain or branched-chain saturated hydrocarbons. The designation "C 1-14 alkyl" means an optionally substituted straight or branched chain saturated hydrocarbon containing 1 to 14 carbon atoms. Unless otherwise specified, alkyl groups described herein refer to both unsubstituted and substituted alkyl groups.
如本文所用,术语“烯基”或“烯基基团”意指包含两个或更多个碳原子(例如,两个、三个、四个、五个、六个、七个、八个、九个、十个、十一个、十二个、十三个、十四个、十五个、十六个、十七个、十八个、十九个、二十个或更多个碳原子)和至少一个双键的任选取代的直链或支链烃。标记“C2-14烯基”意指包含2-14个碳原子和至少一个碳-碳双键的任选取代的直链或支链烃。烯基可以包含一个、两个、三个、四个或更多个碳-碳双键。例如,C18烯基可以包含一个或多个双键。包含两个双键的C18烯基可以是亚油基。除非另外说明,否则本文所述的烯基是指未取代和取代的烯基两者。As used herein, the term "alkenyl" or "alkenyl group" means a group comprising two or more carbon atoms (eg, two, three, four, five, six, seven, eight , nine, ten, eleven, twelve, thirteen, fourteen, fifteen, sixteen, seventeen, eighteen, nineteen, twenty or more carbon atoms) and optionally substituted straight or branched chain hydrocarbons with at least one double bond. The designation "C 2-14 alkenyl" means an optionally substituted straight or branched chain hydrocarbon containing 2 to 14 carbon atoms and at least one carbon-carbon double bond. Alkenyl groups can contain one, two, three, four or more carbon-carbon double bonds. For example, a C18 alkenyl group may contain one or more double bonds. A C18 alkenyl group containing two double bonds may be a linoleyl group. Unless otherwise specified, alkenyl groups described herein refer to both unsubstituted and substituted alkenyl groups.
如本文所用,术语“炔基”或“炔基基团”意指包含两个或更多个碳原子(例如,两个、三个、四个、五个、六个、七个、八个、九个、十个、十一个、十二个、十三个、十四个、十五个、十六个、十七个、十八个、十九个、二十个或更多个碳原子)和至少一个碳-碳三键的任选取代的直链或支链烃。标记“C2-14炔基”意指包含2-14个碳原子和至少一个碳-碳三键的任选取代的直链或支链烃。炔基可以包含一个、两个、三个、四个或更多个碳-碳三键。例如,C18炔基可以包含一个或多个碳-碳三键。除非另外说明,否则本文所述的炔基是指未取代和取代的炔基两者。As used herein, the term "alkynyl" or "alkynyl group" means a group containing two or more carbon atoms (eg, two, three, four, five, six, seven, eight , nine, ten, eleven, twelve, thirteen, fourteen, fifteen, sixteen, seventeen, eighteen, nineteen, twenty or more carbon atoms) and at least one carbon-carbon triple bond optionally substituted straight or branched chain hydrocarbon. The designation "C 2-14 alkynyl" means an optionally substituted straight or branched chain hydrocarbon containing 2 to 14 carbon atoms and at least one carbon-carbon triple bond. Alkynyl groups can contain one, two, three, four or more carbon-carbon triple bonds. For example, a C18 alkynyl group may contain one or more carbon-carbon triple bonds. Unless otherwise specified, alkynyl groups described herein refer to both unsubstituted and substituted alkynyl groups.
如本文所用,术语“碳环”或“碳环基团”意指包含一个或多个由碳原子构成的环的任选取代的单环或多环体系。环可以是三元、四元、五元、六元、七元、八元、九元、十元、十一元、十二元、十三元、十四元、十五元、十六元、十七元、十八元、十九元或二十元环。标记“C3-6碳环”意指包含具有3-6个碳原子的单环的碳环。碳环可以包含一个或多个碳-碳双键或三键,并且可以是非芳香族或芳香族环(例如,环烷基或芳基)。碳环的实例包括环丙基、环戊基、环己基、苯基、萘基和1,2-二氢萘基。如本文所用,术语“环烷基”意指非芳族碳环并且可以包含或不包含任何双键或三键。除非另外说明,否则本文所述的碳环是指未取代和取代的碳环基团两者,即,任选取代的碳环。在一些实施方案中,碳环是C3-8环烷基。在一些实施方案中,碳环是C3-6环烷基。在一些实施方案中,碳环是C6-10芳基。As used herein, the term "carbocycle" or "carbocyclic group" means an optionally substituted monocyclic or polycyclic ring system comprising one or more rings composed of carbon atoms. Rings can be three yuan, four yuan, five yuan, six yuan, seven yuan, eight yuan, nine yuan, ten yuan, eleven yuan, twelve yuan, thirteen yuan, fourteen yuan, fifteen yuan, sixteen yuan , seventeen, eighteen, nineteen or twenty ring. The designation " C3-6 carbocycle" means a carbocycle comprising a monocyclic ring having 3-6 carbon atoms. Carbocycles can contain one or more carbon-carbon double or triple bonds, and can be non-aromatic or aromatic rings (eg, cycloalkyl or aryl). Examples of carbocycles include cyclopropyl, cyclopentyl, cyclohexyl, phenyl, naphthyl, and 1,2-dihydronaphthyl. As used herein, the term "cycloalkyl" means a non-aromatic carbocyclic ring and may or may not contain any double or triple bonds. Unless otherwise specified, carbocycles described herein refer to both unsubstituted and substituted carbocyclic groups, ie, optionally substituted carbocycles. In some embodiments, the carbocycle is C 3-8 cycloalkyl. In some embodiments, the carbocycle is C 3-6 cycloalkyl. In some embodiments, the carbocycle is a C6-10 aryl.
“芳基”包括具有芳香性的基团,包括“共轭”的或具有至少一个芳环且在环结构中不含任何杂原子的多环体系。实例包括苯基、苄基、1,2,3,4-四氢萘基等。在一些实施方案中,“芳基”是具有芳香性的C6-10碳环(例如,“芳基”是C6-10芳基)。"Aryl" includes groups having aromaticity, including "conjugated" or polycyclic systems having at least one aromatic ring and not containing any heteroatoms in the ring structure. Examples include phenyl, benzyl, 1,2,3,4-tetrahydronaphthyl, and the like. In some embodiments, "aryl" is a C6-10 carbocyclic ring having aromaticity (eg, "aryl" is a C6-10 aryl group).
如本文所用,术语“杂环”或“杂环基团”意指包含一个或多个环的任选取代的单环或多环体系,其中至少一个环包含至少一个杂原子。杂原子可以是例如氮、氧或硫原子。环可以是三元、四元、五元、六元、七元、八元、九元、十元、十一元、十二元、十三元或十四元环。杂环可以包含一个或多个双键或三键并且可以是非芳香族或芳香族环(例如,杂环烷基或杂芳基)。杂环的实例包括咪唑基、咪唑烷基、噁唑基、噁唑烷基、噻唑基、噻唑烷基、吡唑烷基、吡唑基、异噁唑烷基、异噁唑基、异噻唑烷基、异噻唑基、吗啉基、吡咯基、吡咯烷基、呋喃基、四氢呋喃基、噻吩基、吡啶基、哌啶基、喹啉基和异喹啉基。如本文所用,术语“杂环烷基”意指非芳香族杂环并且可以包含或不包含任何双键或三键。除非另外说明,否则本文所述的杂环是指未取代和取代的杂环基团两者,即,任选取代的杂环。在一些实施方案中,所述杂环是4至12元杂环烷基。在一些实施方案中,所述杂环是5元或6元杂芳基。As used herein, the term "heterocycle" or "heterocyclic group" means an optionally substituted monocyclic or polycyclic ring system comprising one or more rings, at least one ring comprising at least one heteroatom. Heteroatoms can be, for example, nitrogen, oxygen or sulfur atoms. Rings can be three-, four-, five-, six-, seven-, eight-, nine-, ten-, eleven-, twelve-, thirteen-, or fourteen-membered rings. Heterocycles may contain one or more double or triple bonds and may be non-aromatic or aromatic rings (eg, heterocycloalkyl or heteroaryl). Examples of heterocycles include imidazolyl, imidazolidinyl, oxazolyl, oxazolidinyl, thiazolyl, thiazolidinyl, pyrazolidinyl, pyrazolyl, isoxazolidinyl, isoxazolyl, isothiazole Alkyl, isothiazolyl, morpholinyl, pyrrolyl, pyrrolidinyl, furyl, tetrahydrofuranyl, thienyl, pyridyl, piperidinyl, quinolinyl and isoquinolinyl. As used herein, the term "heterocycloalkyl" means a non-aromatic heterocycle and may or may not contain any double or triple bonds. Unless otherwise specified, heterocycles described herein refer to both unsubstituted and substituted heterocycle groups, ie, optionally substituted heterocycles. In some embodiments, the heterocycle is a 4- to 12-membered heterocycloalkyl. In some embodiments, the heterocycle is a 5- or 6-membered heteroaryl.
“杂芳基”是如上所定义的芳基,不同的是在环结构中具有一至四个杂原子,并且还可以称为“芳基杂环”或“杂芳族化合物”。如本文所用,术语“杂芳基”旨在包含稳定的5、6或7元单环或7、8、9、10、11或12元双环芳族杂环,其由碳原子和一个或多个杂原子组成,所述杂原子例如独立地选自由氮、氧硫和硼组成的组的1或1-2或1-3或1-4或1-5或1-6个杂原子,或者例如1、2、3、4、5或6个杂原子。氮原子可以是取代的或未取代的(即N或NR,其中R是H或如所定义的其他取代基)。氮和硫杂原子可任选被氧化(即,N→O和S(O)p,其中p=1或2)。需要注意的是芳族杂环中S和O原子的总数目不超过1。"Heteroaryl" is an aryl group as defined above, except having one to four heteroatoms in the ring structure, and may also be referred to as an "arylheterocycle" or "heteroaromatic". As used herein, the term "heteroaryl" is intended to encompass stable 5, 6 or 7 membered monocyclic or 7, 8, 9, 10, 11 or 12 membered bicyclic aromatic heterocycles consisting of carbon atoms and one or more heteroatoms such as 1 or 1-2 or 1-3 or 1-4 or 1-5 or 1-6 heteroatoms independently selected from the group consisting of nitrogen, oxygen sulfur and boron, or For example 1, 2, 3, 4, 5 or 6 heteroatoms. Nitrogen atoms can be substituted or unsubstituted (ie, N or NR, where R is H or other substituents as defined). Nitrogen and sulfur heteroatoms can be optionally oxidized (ie, N→O and S(O) p , where p=1 or 2). Note that the total number of S and O atoms in the aromatic heterocycle does not exceed one.
杂芳基的实例包括吡咯、呋喃、噻吩、噻唑、异噻唑、咪唑、三唑、四唑、吡唑、噁唑、异噁唑、吡啶、吡嗪、哒嗪、嘧啶等。Examples of heteroaryl groups include pyrrole, furan, thiophene, thiazole, isothiazole, imidazole, triazole, tetrazole, pyrazole, oxazole, isoxazole, pyridine, pyrazine, pyridazine, pyrimidine, and the like.
此外,术语“芳基”和“杂芳基”包括多环芳基和杂芳基,例如三环、双环,例如萘、苯并噁唑、苯并二噁唑、苯并噻唑、苯并咪唑、苯并噻吩、喹啉、异喹啉、萘啶(naphthrydine)、吲哚、苯并呋喃、嘌呤、苯并呋喃、脱氮嘌呤、中氮茚。In addition, the terms "aryl" and "heteroaryl" include polycyclic aryl and heteroaryl groups, such as tricyclic, bicyclic, such as naphthalene, benzoxazole, benzobisoxazole, benzothiazole, benzimidazole , benzothiophene, quinoline, isoquinoline, naphthrydine, indole, benzofuran, purine, benzofuran, deazapurine, indolizine.
如本文所用,“可生物降解基团”是有助于加快脂质在哺乳动物个体中的代谢的基团。可生物降解基团可以选自但不限于由以下组成的组:-C(O)O-、-OC(O)-、-C(O)N(R')-、-N(R')C(O)-、-C(O)-、-C(S)-、-C(S)S-、-SC(S)-、-CH(OH)-、-P(O)(OR')O-、-S(O)2-、芳基和杂芳基组成的组。如本文所用,“芳基”是包含一个或多个芳环的任选取代的碳环基团。芳基的实例包括苯基和萘基。如本文所用,“杂芳基”是包含一个或多个芳环的任选取代的杂环基团。杂芳基的实例包括吡咯基、呋喃基、噻吩基、咪唑基、噁唑基和噻唑基。芳基和杂芳基都可以是任选取代的。例如,M和M'可以选自由任选取代的苯基、噁唑和噻唑组成的非限制性组。在本文的式中,M和M'可以独立地选自上文的可生物降解基团的列表。除非另外说明,否则本文所述的芳基或杂芳基是指未取代和取代的基团两者,即,任选取代的芳基或杂芳基。As used herein, a "biodegradable group" is a group that helps to accelerate the metabolism of lipids in mammalian subjects. The biodegradable group may be selected from, but is not limited to, the group consisting of: -C(O)O-, -OC(O)-, -C(O)N(R')-, -N(R') C(O)-, -C(O)-, -C(S)-, -C(S)S-, -SC(S)-, -CH(OH)-, -P(O)(OR' )O-, -S(O) 2- , the group consisting of aryl and heteroaryl. As used herein, "aryl" is an optionally substituted carbocyclic group containing one or more aromatic rings. Examples of aryl groups include phenyl and naphthyl. As used herein, "heteroaryl" is an optionally substituted heterocyclic group containing one or more aromatic rings. Examples of heteroaryl groups include pyrrolyl, furyl, thienyl, imidazolyl, oxazolyl, and thiazolyl. Both aryl and heteroaryl groups can be optionally substituted. For example, M and M' can be selected from the non-limiting group consisting of optionally substituted phenyl, oxazole and thiazole. In the formulae herein, M and M' can be independently selected from the above list of biodegradable groups. Unless otherwise specified, an aryl or heteroaryl group described herein refers to both unsubstituted and substituted groups, ie, an optionally substituted aryl or heteroaryl group.
除非另外说明,否则烷基、烯基和环基(例如碳环基和杂环基)可以是任选取代的。任选的取代基可以选自但不限于由以下组成的组:卤素原子(例如,氯基、溴基、氟基或碘基)、羧酸(例如,-C(O)OH)、醇(例如,羟基、-OH)、酯(例如,-C(O)OR或-OC(O)R)、醛(例如,-C(O)H)、羰基(例如,-C(O)R或由C=O表示)、卤化酰基(例如,-C(O)X,其中X是选自溴化物、氟化物、氯化物和碘化物的卤化物)、碳酸酯(例如,-OC(O)OR)、烷氧基(例如,-OR)、缩醛(例如,-C(OR)2R””,其中每个OR是相同或不同的烷氧基并且R””是烷基或烯基)、磷酸(例如,P(O)4 3-)、硫基(例如,-SH)、亚砜(例如,-S(O)R)、亚磺酸酸(例如,-S(O)OH)、磺酸(例如,-S(O)2OH)、硫醛(例如,-C(S)H)、硫酸(例如,S(O)4 2-)、磺酰基(例如,-S(O)2-)、酰胺(例如,-C(O)NR2或-N(R)C(O)R)、叠氮基(例如,-N3)、硝基(例如,-NO2)、氰基(例如-CN)、异氰基(例如,-NC)、酰氧基(例如,-OC(O)R)、氨基(例如,-NR2、-NRH或-NH2)、氨基甲酰基(例如,-OC(O)NR2、-OC(O)NRH或-OC(O)NH2)、磺酰胺(例如,-S(O)2NR2、-S(O)2NRH、-S(O)2NH2、-N(R)S(O)2R、-N(H)S(O)2R、-N(R)S(O)2H或-N(H)S(O)2H)、烷基、烯基和环基(例如,碳环基或杂环基)。在前述任一种中,R是如本文所定义的烷基或烯基。在一些实施方案中,取代基本身可以进一步被例如一个、两个、三个、四个、五个或六个如本文所定义的取代基取代。例如,C1-6烷基可以进一步被一个、两个、三个、四个、五个或六个如本文所述的取代基取代。Unless otherwise specified, alkyl, alkenyl, and cyclyl groups (eg, carbocyclyl and heterocyclyl) can be optionally substituted. Optional substituents may be selected from, but are not limited to, the group consisting of halogen atoms (eg, chloro, bromo, fluoro, or iodo), carboxylic acids (eg, -C(O)OH), alcohols ( For example, hydroxyl, -OH), ester (eg, -C(O)OR or -OC(O)R), aldehyde (eg, -C(O)H), carbonyl (eg, -C(O)R or represented by C=O), halogenated acyl groups (eg, -C(O)X, where X is a halide selected from bromide, fluoride, chloride, and iodide), carbonates (eg, -OC(O) OR), alkoxy (eg, -OR), acetal (eg, -C (OR)2R"", where each OR is the same or a different alkoxy and R"" is an alkyl or alkenyl group ), phosphoric acid (eg, P(O) 4 3- ), thio (eg, -SH), sulfoxide (eg, -S(O)R), sulfinic acid (eg, -S(O)OH) ), sulfonic acid (eg, -S(O) 2 OH), thioaldehyde (eg, -C(S)H), sulfuric acid (eg, S(O) 4 2- ), sulfonyl (eg, -S( O) 2- ), amide (eg, -C (O)NR2 or -N(R)C(O)R), azide (eg, -N3 ), nitro (eg, -NO2 ) , cyano (eg, -CN), isocyano (eg, -NC), acyloxy (eg, -OC(O)R), amino (eg, -NR2, -NRH, or -NH2 ) , amino Formyl (eg, -OC(O)NR2, -OC(O)NRH, or -OC(O) NH2 ) , sulfonamide (eg, -S(O) 2NR2 , -S(O ) 2NRH , -S(O) 2 NH 2 , -N(R)S(O) 2 R, -N(H)S(O) 2 R, -N(R)S(O) 2 H or -N(H )S(O ) 2H), alkyl, alkenyl, and cyclyl (eg, carbocyclyl or heterocyclyl). In any of the foregoing, R is alkyl or alkenyl as defined herein. In some embodiments, the substituents themselves may be further substituted with, for example, one, two, three, four, five or six substituents as defined herein. For example, a C1-6 alkyl group can be further substituted with one, two, three, four, five or six substituents as described herein.
含有氮的本公开化合物可以通过用氧化剂(例如,3-氯过氧苯甲酸(mCPBA)和/或过氧化氢)处理而转化成N-氧化物,以得到本公开的其他化合物。因此,当价态和结构允许时,考虑所有示出并且要求保护的含氮化合物都包括所示化合物及其N-氧化物衍生物(可称为N→O或N+-O-)。此外,在其他情况下,本公开的化合物中的氮可以被转化成N-羟基或N-烷氧基化合物。例如,N-羟基化合物可以通过用氧化剂诸如m-CPBA氧化母体胺来制备。当价态和结构允许时,还考虑所有示出并且要求保护的含氮化合物覆盖所示化合物及其N-羟基(即N-OH)和N-烷氧基(即N-OR,其中R是取代或未取代的C1-C6烷基、C1-C6烯基、C1-C6炔基、3-14元碳环或3-14元杂环)衍生物。Nitrogen containing compounds of the present disclosure can be converted to N-oxides by treatment with oxidizing agents (eg, 3-chloroperoxybenzoic acid (mCPBA) and/or hydrogen peroxide) to give other compounds of the present disclosure. Accordingly, all shown and claimed nitrogen-containing compounds are considered to include the compounds shown and their N-oxide derivatives (which may be referred to as N→O or N + -O - ) when valence and structure permit. Furthermore, in other instances, nitrogens in the compounds of the present disclosure can be converted to N-hydroxy or N-alkoxy compounds. For example, N-hydroxy compounds can be prepared by oxidizing the parent amine with an oxidizing agent such as m-CPBA. All shown and claimed nitrogen-containing compounds are also considered to cover the compounds shown and their N-hydroxyl (ie N-OH) and N-alkoxy (ie N-OR, where R is N-OH) and N-alkoxy (ie N-OR) compounds when valence and structure permit Substituted or unsubstituted C 1 -C 6 alkyl, C 1 -C 6 alkenyl, C 1 -C 6 alkynyl, 3-14 membered carbocycle or 3-14 membered heterocycle) derivatives.
约、大约:如本文所用,术语“大约”和“约”在用于一个或多个所关注的值时是指与所述参考值类似的值。在某些实施方案中,除非另外规定或另外从上下文显而易见,否则术语“大约”或“约”是指在所述参考值的任一方向上(大于或小于)的25%、20%、19%、18%、17%、16%、15%、14%、13%、12%、11%、10%、9%、8%、7%、6%、5%、4%、3%、2%、1%或更小的范围内的一系列值(这一值将超过可能值的100%的情况除外)。例如,当用于纳米粒子组合物的脂质组分中给定化合物的量的上下文中时,“约”可以意指所述值的+/-10%。例如,包含具有约40%的给定化合物的脂质组分的纳米粒子组合物可以包含30%-50%的所述化合物。About, approximately: As used herein, the terms "approximately" and "about" when applied to the value or values concerned refer to a value similar to the referenced value. In certain embodiments, unless otherwise specified or otherwise apparent from context, the terms "about" or "about" refer to 25%, 20%, 19% in either direction (greater or less than) of the reference value , 18%, 17%, 16%, 15%, 14%, 13%, 12%, 11%, 10%, 9%, 8%, 7%, 6%, 5%, 4%, 3%, 2 A range of values in the range of %, 1% or less (except where this value would exceed 100% of the possible values). For example, when used in the context of the amount of a given compound in the lipid component of a nanoparticle composition, "about" can mean +/- 10% of the stated value. For example, a nanoparticle composition comprising a lipid component with about 40% of a given compound may comprise 30-50% of the compound.
如本文所用,术语“化合物”意图包括具有所描绘结构的所有异构体和同位素。“同位素”是指具有相同原子序数但由于原子核中的中子数不同而具有不同质量数的原子。例如,氢的同位素包括氚和氘。另外,本公开的化合物、盐或复合物可以与溶剂或水分子组合制备以通过常规方法形成溶剂化物和水合物。As used herein, the term "compound" is intended to include all isomers and isotopes having the depicted structure. "Isotopes" refer to atoms with the same atomic number but different mass numbers due to different numbers of neutrons in the nucleus. For example, isotopes of hydrogen include tritium and deuterium. Additionally, the compounds, salts or complexes of the present disclosure can be prepared in combination with solvents or water molecules to form solvates and hydrates by conventional methods.
如本文所用,术语“接触”意指在两个或更多个实体之间建立物理连接。例如,使哺乳动物细胞与纳米粒子组合物接触意味着,使哺乳动物细胞和纳米粒子共有物理连接。使细胞与外部实体在体内和离体接触的方法是生物学领域中众所周知的。例如,使纳米粒子组合物与处于哺乳动物体内的哺乳动物细胞接触可以通过不同施用途径(例如,静脉内、肌肉内、皮内和皮下)进行并且可以包括不同量的纳米粒子组合物(例如,空LNP或负载LNP)。此外,纳米粒子组合物可以接触一个以上的哺乳动物细胞。As used herein, the term "contacting" means establishing a physical connection between two or more entities. For example, contacting a mammalian cell with a nanoparticle composition means that the mammalian cell and the nanoparticle share a physical link. Methods of contacting cells with external entities in vivo and ex vivo are well known in the field of biology. For example, contacting a nanoparticle composition with mammalian cells in a mammal can be by different routes of administration (eg, intravenous, intramuscular, intradermal, and subcutaneous) and can include varying amounts of the nanoparticle composition (eg, Empty LNP or Loaded LNP). Additionally, the nanoparticle composition can be contacted with more than one mammalian cell.
如本文所用,术语“递送”意指将实体提供至目的地。例如,将治疗剂和/或预防剂递送至受试者可包括将包含所述治疗剂和/或预防剂的纳米粒子组合物施用至所述受试者(例如,通过静脉内、肌肉内、皮内或皮下途径)。将纳米粒子组合物施用至哺乳动物或哺乳动物细胞可包括使一个或多个细胞与所述纳米粒子组合物接触。As used herein, the term "delivery" means providing an entity to a destination. For example, delivering a therapeutic and/or prophylactic agent to a subject can include administering to the subject a nanoparticle composition comprising the therapeutic and/or prophylactic agent (eg, via intravenous, intramuscular, intradermal or subcutaneous route). Administering a nanoparticle composition to a mammal or mammalian cells can include contacting one or more cells with the nanoparticle composition.
如本文所用,术语“增强的递送”意指,与通过对照纳米粒子递送至所关注的靶组织的治疗剂和/或预防剂(例如,MC3、KC2或DLinDMA)的水平相比,通过纳米粒子递送至所关注的靶组织(例如,哺乳动物肝)的治疗剂和/或预防剂更多(例如,多至少1.5倍、多至少2倍、多至少3倍、多至少4倍、多至少5倍、多至少6倍、多至少7倍、多至少8倍、多至少9倍、多至少10倍)。递送至特定组织的纳米粒子的水平可以通过将组织中产生的蛋白质的量与所述组织的重量相比较,将组织中治疗剂和/或预防剂的量与所述组织的重量相比较,将组织中产生的蛋白质的量与所述组织中总蛋白质的量相比较或将组织中治疗剂和/或预防剂的量与所述组织中总治疗剂和/或预防剂的量相比较来测量。应了解,不必在所治疗的受试者体内测定纳米粒子向靶组织的增强的递送,其可以在替代物诸如动物模型(例如,大鼠模型)中测定。在某些实施方案中,包含根据式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物的纳米粒子组合物具有基本上相同水平的递送增强,不管施用途径如何。例如,本文公开的某些化合物在用于静脉内或肌肉内递送治疗剂和/或预防剂时表现出类似的递送增强。在其他实施方案中,本文公开的某些化合物在用于肌肉内递送治疗剂和/或预防剂时表现出的递送增强水平要高于静脉内递送时表现出的递送增强水平。As used herein, the term "enhanced delivery" means that the level of therapeutic and/or prophylactic (eg, MC3, KC2, or DLinDMA) delivered by a nanoparticle compared to the level of therapeutic and/or prophylactic (eg, MC3, KC2, or DLinDMA) delivered to a target tissue of interest by a control nanoparticle More (eg, at least 1.5 times more, at least 2 times more, at least 3 times more, at least 4 times more, at least 5 times more) therapeutic and/or prophylactic agents delivered to the target tissue of interest (eg, mammalian liver) times, at least 6 times more, at least 7 times more, at least 8 times more, at least 9 times more, at least 10 times more). The level of nanoparticles delivered to a particular tissue can be determined by comparing the amount of protein produced in the tissue to the weight of the tissue, the amount of therapeutic and/or prophylactic agent in the tissue to the weight of the tissue, and The amount of protein produced in the tissue is measured by comparing the amount of total protein in the tissue or by comparing the amount of therapeutic and/or prophylactic agent in the tissue with the amount of total therapeutic and/or preventive agent in the tissue . It will be appreciated that it is not necessary to measure the enhanced delivery of nanoparticles to target tissues in the subject being treated, it can be measured in surrogates such as animal models (eg, rat models). In certain embodiments, comprising according to formula (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y), (I- Ya) Nanoparticles of Compounds of (IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb) The compositions have substantially the same level of delivery enhancement regardless of the route of administration. For example, certain compounds disclosed herein exhibit similar delivery enhancements when used for intravenous or intramuscular delivery of therapeutic and/or prophylactic agents. In other embodiments, certain compounds disclosed herein exhibit higher levels of delivery enhancement when used for intramuscular delivery of therapeutic and/or prophylactic agents than when administered intravenously.
如本文所用,术语“特异性递送(specific delivery/specifically deliver/specifically delivering)”意指,与脱靶组织(例如,哺乳动物脾)相比,通过纳米粒子递送至所关注的靶组织(例如,哺乳动物肝)的治疗剂和/或预防剂更多(例如,多至少1.5倍、多至少2倍、多至少3倍、多至少4倍、多至少5倍、多至少6倍、多至少7倍、多至少8倍、多至少9倍、多至少10倍)。递送至特定组织的纳米粒子的水平可以通过将组织中产生的蛋白质的量与所述组织的重量相比较,将组织中治疗剂和/或预防剂的量与所述组织的重量相比较,将组织中产生的蛋白质的量与所述组织中总蛋白质的量相比较或将组织中治疗剂和/或预防剂的量与所述组织中总治疗剂和/或预防剂的量相比较来测量。例如,对于肾血管靶向,如果在全身施用治疗剂和/或预防剂后,与递送至肝或脾的治疗剂和/或预防剂相比,递送至肾的治疗剂和/或预防剂每1g组织多1.5倍、2倍、3倍、5倍、10倍、15倍或20倍,则与肝和脾相比,所述治疗剂和/或预防剂被特异性提供至哺乳动物肾。应了解,不必在所治疗的受试者体内测定纳米粒子特异性递送至靶组织的能力,其可以在替代物诸如动物模型(例如,大鼠模型)中测定。As used herein, the term "specific delivery/specifically deliver/specifically delivering" means delivery by nanoparticles to target tissues of interest (eg, mammalian spleen) as compared to off-target tissues (eg, mammalian spleen). animal liver) more (eg, at least 1.5 times more, at least 2 times more, at least 3 times more, at least 4 times more, at least 5 times more, at least 6 times more, at least 7 times more , at least 8 times more, at least 9 times more, at least 10 times more). The level of nanoparticles delivered to a particular tissue can be determined by comparing the amount of protein produced in the tissue to the weight of the tissue, the amount of therapeutic and/or prophylactic agent in the tissue to the weight of the tissue, and The amount of protein produced in the tissue is measured by comparing the amount of total protein in the tissue or by comparing the amount of therapeutic and/or prophylactic agent in the tissue with the amount of total therapeutic and/or preventive agent in the tissue . For example, for renal vascular targeting, if the therapeutic and/or prophylactic agent is administered systemically, the therapeutic and/or prophylactic agent delivered to the kidney will be 1.5-fold, 2-fold, 3-fold, 5-fold, 10-fold, 15-fold or 20-fold more 1 g of tissue, the therapeutic and/or prophylactic agent is specifically delivered to the mammalian kidney compared to the liver and spleen. It will be appreciated that the ability of nanoparticles to specifically deliver to target tissues does not have to be determined in the subject being treated, but can be determined in surrogate models such as animal models (eg, rat models).
如本文所用,“包封效率”是指变为纳米粒子组合物的一部分的治疗剂和/或预防剂的量与用于制备纳米粒子组合物的治疗剂和/或预防剂的初始总量的比率。例如,如果在最初提供至组合物的总计100mg治疗剂和/或预防剂中有97mg治疗剂和/或预防剂被包封于纳米粒子组合物中,则可以给出包封效率为97%。如本文所用,“包封”可以指完全、大部分或部分封装、密封、包围或包装。As used herein, "encapsulation efficiency" refers to the amount of therapeutic and/or prophylactic agent that becomes part of a nanoparticle composition versus the initial total amount of therapeutic and/or prophylactic agent used to prepare the nanoparticle composition ratio. For example, if 97 mg of the therapeutic and/or prophylactic agent is encapsulated in the nanoparticle composition out of a total of 100 mg of therapeutic and/or prophylactic agent initially provided to the composition, an encapsulation efficiency of 97% can be given. As used herein, "encapsulating" may refer to encapsulating, sealing, enclosing, or wrapping in whole, in part, or in part.
如本文所用,“包封”、“包封的”、“负载的”和“缔合的”可以指完全、大部分或部分封装、密封、包围或包装。如本文所用,“包封”或“缔合”可以指将单个核酸分子限制在纳米粒子内和/或在单个核酸分子与纳米粒子之间建立物理化学关系的过程。如本文所用,“空纳米粒子”可以指实质上不含治疗剂或预防剂的纳米粒子。如本文所用,“空纳米粒子”或“空脂质纳米粒子”可以指实质上不含核酸的纳米粒子。如本文所用,“空纳米粒子”或“空脂质纳米粒子”可以指实质上不含核苷酸或多肽的纳米粒子。如本文所用,“空纳米粒子”或“空脂质纳米粒子”可以指实质上仅由脂质组分组成纳米粒子。如本文所用,“负载纳米粒子”或“负载脂质纳米粒子”(也称为“完整纳米粒子”或“完整脂质纳米粒子”)可以指包含空纳米粒子和治疗剂或预防剂组分的纳米粒子。如本文所用,“负载纳米粒子”或“负载脂质纳米粒子”(也称为“完整纳米粒子”或“完整脂质纳米粒子”)可以指包含空纳米粒子和核苷酸或多肽组分的纳米粒子。如本文所用,“负载纳米粒子”或“负载脂质纳米粒子”(也称为“完整纳米粒子”或“完整脂质纳米粒子”)可以指包含空纳米粒子和核酸组分的纳米粒子。As used herein, "encapsulated," "encapsulated," "loaded," and "associated with" can refer to fully, mostly, or partially encapsulating, sealing, surrounding, or wrapping. As used herein, "encapsulation" or "association" can refer to the process of confining a single nucleic acid molecule within a nanoparticle and/or establishing a physicochemical relationship between a single nucleic acid molecule and a nanoparticle. As used herein, "empty nanoparticles" can refer to nanoparticles that are substantially free of therapeutic or prophylactic agents. As used herein, "empty nanoparticles" or "empty lipid nanoparticles" can refer to nanoparticles that are substantially free of nucleic acid. As used herein, "empty nanoparticles" or "empty lipid nanoparticles" can refer to nanoparticles that are substantially free of nucleotides or polypeptides. As used herein, "empty nanoparticle" or "empty lipid nanoparticle" may refer to a nanoparticle consisting essentially of only lipid components. As used herein, a "loaded nanoparticle" or "loaded lipid nanoparticle" (also referred to as "intact nanoparticle" or "intact lipid nanoparticle") may refer to a nanoparticle comprising an empty nanoparticle and a therapeutic or prophylactic component Nanoparticles. As used herein, "loaded nanoparticle" or "loaded lipid nanoparticle" (also referred to as "intact nanoparticle" or "intact lipid nanoparticle") may refer to a nanoparticle comprising an empty nanoparticle and a nucleotide or polypeptide component Nanoparticles. As used herein, "loaded nanoparticle" or "loaded lipid nanoparticle" (also referred to as "intact nanoparticle" or "intact lipid nanoparticle") may refer to a nanoparticle comprising empty nanoparticles and nucleic acid components.
如本文所用,核酸序列的“表达”是指mRNA翻译成多肽或蛋白质和/或多肽或蛋白质的翻译后修饰。As used herein, "expression" of a nucleic acid sequence refers to translation of mRNA into polypeptides or proteins and/or post-translational modifications of polypeptides or proteins.
如本文所用,术语“体外”是指在人工环境(例如在试管或反应容器中、细胞培养物中、皮氏培养皿(Petri dish)等)中发生而非在生物体(例如动物、植物或微生物)内发生的事件。As used herein, the term "in vitro" refers to taking place in an artificial environment (eg, in a test tube or reaction vessel, in a cell culture, a Petri dish, etc.) rather than in an organism (eg, an animal, plant or events occurring in microorganisms).
如本文所用,术语“体内”是指在生物体(例如动物、植物或微生物,或其细胞或组织)内发生的事件。As used herein, the term "in vivo" refers to events that occur within an organism, such as an animal, plant, or microorganism, or cells or tissues thereof.
如本文所用,术语“离体”是指在生物体(例如,动物、植物或微生物,或其细胞或组织)外发生的事件。离体事件可以在相对于自然(例如体内)环境改变最小的环境中发生。As used herein, the term "ex vivo" refers to an event that occurs outside an organism (eg, an animal, plant, or microorganism, or a cell or tissue thereof). An ex vivo event can occur in an environment that is minimally altered relative to the natural (eg, in vivo) environment.
如本文所用,术语“异构体”意指化合物的任何几何异构体、互变异构体、两性离子、立体异构体、对映异构体或非对映异构体。化合物可以包含一个或多个手性中心和/或双键,并因此呈立体异构体诸如双键异构体(即,几何E/Z异构体)或非对映异构体(例如对映异构体(即,(+)或(-))或顺式/反式异构体)形式存在。本公开涵盖本文所述的化合物的任何和所有异构体,包括立体异构纯的形式(例如几何异构纯的、对映异构纯的或非对映异构纯的)以及对映异构体和立体异构体混合物,例如外消旋物。化合物的对映异构体和立体异构体混合物以及将其拆分成其组成对映异构体或立体异构体的方式是众所周知的。As used herein, the term "isomer" means any geometric isomer, tautomer, zwitterion, stereoisomer, enantiomer or diastereomer of a compound. Compounds may contain one or more chiral centers and/or double bonds, and thus take the form of stereoisomers such as double bond isomers (ie, geometric E/Z isomers) or diastereomers (eg, para- Exist as enantiomers (ie, (+) or (-)) or cis/trans isomers. The present disclosure encompasses any and all isomers of the compounds described herein, including stereoisomerically pure forms (eg, geometrically pure, enantiomerically pure, or diastereomerically pure) as well as enantiomerically pure Constituents and stereoisomer mixtures, such as racemates. Enantiomeric and stereoisomeric mixtures of compounds and the manner in which they are resolved into their constituent enantiomers or stereoisomers are well known.
“互变异构体”是以平衡状态存在并且易于从一种异构形式转化成另一种异构形式的两种或更多种结构异构体中的一种。这种转化引起氢原子的形式迁移伴随相邻共轭双键的变换。互变异构体在溶液中以互变异构体组的混合物形式存在。在可能发生互变异构的溶液中,将达到互变异构体的化学平衡。互变异构体的确切比率取决于若干因素,包括温度、溶剂和pH。可通过互变异构化相互转换的互变异构体的概念称为互变异构。A "tautomer" is one of two or more structural isomers that exist in equilibrium and are readily convertible from one isomeric form to another. This transformation induces formal migration of hydrogen atoms accompanied by transformation of adjacent conjugated double bonds. Tautomers exist in solution as mixtures of tautomeric groups. In solutions where tautomerism can occur, a chemical equilibrium of tautomers will be reached. The exact ratio of tautomers depends on several factors, including temperature, solvent and pH. The concept of tautomers that can be interconverted by tautomerization is called tautomerism.
在可能存在的各种类型互变异构中,有两种是常常观察到的。在酮基-烯醇互变异构中,电子和氢原子的移位同时发生。环-链互变异构是由于糖链分子中的醛基(-CHO)与同一分子中的一个羟基(-OH)反应而赋予所述分子如由葡萄糖所展现的环状(环形)形式引起的。Of the various types of tautomerism that may exist, two are frequently observed. In keto-enol tautomerism, shifts of electrons and hydrogen atoms occur simultaneously. Ring-chain tautomerism is caused by the reaction of an aldehyde group (-CHO) in a sugar chain molecule with a hydroxyl group (-OH) in the same molecule to give the molecule a cyclic (ring) form as exhibited by glucose of.
常见的互变异构对是:酮-烯醇、酰胺-腈、内酰胺-内酰亚胺、杂环中(例如在核碱基诸如鸟嘌呤、胸腺嘧啶和胞嘧啶中)的酰胺-亚胺酸互变异构、亚胺-烯胺和烯胺-烯胺。双取代胍中的互变异构的实例在下文中示出。Common tautomeric pairs are: keto-enol, amide-nitrile, lactam-lactam, amide-imide in heterocycles (eg in nucleobases such as guanine, thymine and cytosine) Amino acid tautomerism, imine-enamine and enamine-enamine. Examples of tautomerism in disubstituted guanidines are shown below.
应了解,本公开的化合物可以用不同互变异构体描绘。还应了解,当化合物具有互变异构形式时,所有互变异构形式都意图包括在本公开的范围内,并且所述化合物的命名不排除任何互变异构体形式。It will be appreciated that the compounds of the present disclosure may be depicted with different tautomers. It is also to be understood that when compounds have tautomeric forms, all tautomeric forms are intended to be included within the scope of this disclosure and that the naming of the compounds does not exclude any tautomeric forms.
如本文所用,“脂质组分”是包含一种或多种脂质的纳米粒子组合物的组分。例如,脂质组分可以包含一种或多种阳离子/可离子化、PEG化、结构性或其他脂质,诸如磷脂。As used herein, a "lipid component" is a component of a nanoparticle composition comprising one or more lipids. For example, the lipid component may comprise one or more cationic/ionizable, PEGylated, structural or other lipids, such as phospholipids.
如本文所用,“接头”是连接两个部分的部分,例如帽物质的两个核苷之间的连接。接头可以包含一个或多个基团,包括但不限于磷酸基团(例如,磷酸、硼代磷酸、硫代磷酸、硒代磷酸和膦酸)、烷基、酰胺化物或甘油。例如,帽类似物的两个核苷可以在其5'位置通过三磷酸基团或通过包含两个磷酸部分和一个硼代磷酸部分的链连接。As used herein, a "linker" is a moiety that connects two moieties, eg, a link between two nucleosides of a capping species. The linker may contain one or more groups including, but not limited to, phosphate groups (eg, phosphoric acid, boronophosphoric acid, thiophosphoric acid, selenophosphoric acid, and phosphonic acid), alkyl, amidate, or glycerol. For example, the two nucleosides of a cap analog can be linked at their 5' position by a triphosphate group or by a chain comprising two phosphate moieties and one boronophosphate moiety.
如本文所用,“施用方法”可以包括静脉内、肌肉内、皮内、皮下或将组合物递送至受试者的其他方法。可以选择施用方法以靶向递送(例如特异性递送)至身体的指定区域或系统。As used herein, "administration method" may include intravenous, intramuscular, intradermal, subcutaneous, or other methods of delivering the composition to a subject. The method of administration can be selected for targeted delivery (eg, specific delivery) to a given area or system of the body.
如本文所用,“修饰的”意指非天然的。例如,RNA可以是修饰的RNA。也就是说,RNA可以包含一个或多个非天然存在的核碱基、核苷、核苷酸或接头。“修饰的”物质在本文中还可以称为“改变的”物质。可以在化学上、结构上或功能上对物质进行修饰或改变。例如,修饰的核碱基物质可以包含一个或多个非天然存在的取代。As used herein, "modified" means non-natural. For example, the RNA can be modified RNA. That is, the RNA can comprise one or more non-naturally occurring nucleobases, nucleosides, nucleotides or linkers. A "modified" substance may also be referred to herein as an "altered" substance. Substances may be modified or altered chemically, structurally or functionally. For example, modified nucleobase species may contain one or more non-naturally occurring substitutions.
如本文所用,“N:P比”是例如在包含脂质组分和RNA的纳米粒子组合物中脂质中的可离子化(在生理pH范围内)氮原子与RNA中的磷酸基团的摩尔比。As used herein, "N:P ratio" is the ratio of ionizable (in the physiological pH range) nitrogen atoms in lipids to phosphate groups in RNA, eg, in a nanoparticle composition comprising a lipid component and RNA The molar ratio of.
如本文所用,“纳米粒子组合物”是包含一种或多种脂质的组合物。纳米粒子组合物的大小通常是微米级或更小并且可以包含脂质双层。纳米粒子组合物涵盖脂质纳米粒子(LNP)、脂质体(例如脂质囊泡)和脂质体复合物。例如,纳米粒子组合物可以是直径为500nm或更小的具有脂质双层的脂质体。As used herein, a "nanoparticle composition" is a composition comprising one or more lipids. Nanoparticle compositions are typically micron-scale or smaller in size and may contain lipid bilayers. Nanoparticle compositions encompass lipid nanoparticles (LNPs), liposomes (eg, lipid vesicles), and liposome complexes. For example, the nanoparticle composition can be a liposome with a lipid bilayer having a diameter of 500 nm or less.
如本文所用,“天然存在的”意指在没有人工辅助的情况下存在于自然界中。As used herein, "naturally occurring" means existing in nature without artificial assistance.
如本文所用,“患者”是指可寻求或有治疗需要、需要治疗、正接受治疗、将接受治疗的受试者,或在经训练的专业人员针对特定疾病或病状的护理下的受试者。As used herein, "patient" refers to a subject who may seek or be in need of, is in need of, is receiving, will receive, or is under the care of a trained professional for a particular disease or condition .
如本文所用,“PEG脂质”或“PEG化脂质”是指包含聚乙二醇组分的脂质。As used herein, "PEG lipid" or "PEGylated lipid" refers to a lipid comprising a polyethylene glycol component.
短语“药学上可接受的”在本文中用于指在合理的医学判断范围内,适于与人类和动物的组织接触使用而无过度毒性、刺激、过敏反应或其他问题或并发症,与合理的效益/风险比相符的化合物、材料、组合物和/或剂型。The phrase "pharmaceutically acceptable" is used herein to mean, within the scope of sound medical judgment, suitable for use in contact with human and animal tissues without undue toxicity, irritation, allergic reaction or other problems or complications, and with reasonable Compounds, materials, compositions and/or dosage forms that are consistent with the benefit/risk ratio.
如本文所用,短语“药学上可接受的赋形剂”是指除了本文所述的化合物之外的并且具有在患者中基本上无毒性和无炎性的特性的任何成分(例如能够使活性化合物悬浮、形成复合物或溶解的媒介物)。赋形剂可包括例如:抗粘剂、抗氧化剂、粘结剂、包衣、压缩助剂、崩解剂、染料(颜料)、软化剂、乳化剂、填充剂(稀释剂)、成膜剂或包衣、调味剂、香料、助流剂(流动增强剂)、润滑剂、防腐剂、印刷油墨、吸附剂、悬浮剂或分散剂、甜味剂以及水合作用的水。示例性赋形剂包括但不限于:丁基化羟基甲苯(BHT)、碳酸钙、磷酸钙(二元的)、硬脂酸钙、交联羧甲纤维素、交联聚乙烯吡咯烷酮、柠檬酸、交联聚维酮、半胱氨酸、乙基纤维素、明胶、羟丙基纤维素、羟丙基甲基纤维素、乳糖、硬脂酸镁、麦芽糖醇、甘露糖醇、甲硫氨酸、甲基纤维素、对羟基苯甲酸甲酯、微晶纤维素、聚乙二醇、聚乙烯吡咯烷酮、聚维酮、预糊化淀粉、对羟基苯甲酸丙酯、视黄醇棕榈酸酯、虫胶、二氧化硅、羧甲基纤维素钠、柠檬酸钠、乙醇酸淀粉钠、山梨糖醇、淀粉(玉米)、硬脂酸、蔗糖、滑石粉、二氧化钛、维生素A、维生素E(α-生育酚)、维生素C、木糖醇以及本文所公开的其他物质。As used herein, the phrase "pharmaceutically acceptable excipient" refers to any ingredient other than a compound described herein that has the property of being substantially non-toxic and non-inflammatory in a patient (eg, capable of rendering an active compound suspending, complexing or dissolving vehicles). Excipients may include, for example: anti-adherents, antioxidants, binders, coatings, compression aids, disintegrants, dyes (pigments), softeners, emulsifiers, fillers (diluents), film formers Or coatings, flavors, fragrances, glidants (flow enhancers), lubricants, preservatives, printing inks, adsorbents, suspending or dispersing agents, sweeteners, and water for hydration. Exemplary excipients include, but are not limited to: butylated hydroxytoluene (BHT), calcium carbonate, calcium phosphate (dibasic), calcium stearate, croscarmellose, crospovidone, citric acid , crospovidone, cysteine, ethyl cellulose, gelatin, hydroxypropyl cellulose, hydroxypropyl methylcellulose, lactose, magnesium stearate, maltitol, mannitol, methionine Acid, methylcellulose, methylparaben, microcrystalline cellulose, polyethylene glycol, polyvinylpyrrolidone, povidone, pregelatinized starch, propylparaben, retinyl palmitate , Shellac, Silicon Dioxide, Sodium Carboxymethyl Cellulose, Sodium Citrate, Sodium Starch Glycolate, Sorbitol, Starch (Corn), Stearic Acid, Sucrose, Talc, Titanium Dioxide, Vitamin A, Vitamin E ( alpha-tocopherol), vitamin C, xylitol, and others disclosed herein.
在本说明书中,化合物的结构式在一些情况下出于便利而呈现某一异构体,但本公开包括所有异构体,诸如几何异构体、基于不对称碳的光学异构体、立体异构体、互变异构体等,应了解,并非所有异构体都具有相同活性水平。此外,所述式呈现的化合物可能存在晶体多晶型。应注意,其任何晶体形式、晶体形式混合物或者酸酐或水合物都被包括在本公开的范围内。In the present specification, the structural formula of a compound presents a certain isomer for convenience in some cases, but the present disclosure includes all isomers, such as geometric isomers, asymmetric carbon-based optical isomers, stereoisomers isomers, tautomers, etc., it should be understood that not all isomers have the same level of activity. Furthermore, the compounds represented by the formulae may exist in crystalline polymorphs. It should be noted that any crystal form, mixture of crystal forms, or anhydrides or hydrates thereof are included within the scope of the present disclosure.
术语“晶体多晶型物”、“多晶型物”或“晶体形式”意指其中化合物(或其盐或溶剂化物)可以结晶成不同晶体堆积排列的晶体结构,所有排列都具有相同元素组成。不同晶体形式通常具有不同的X射线衍射图、红外光谱、熔点、密度、硬度、晶体形状、光学和电学特性、稳定性和溶解性。重结晶溶剂、结晶速率、储存温度以及其他因素可能会使一种晶体形式占优势。所述化合物的晶体多晶型物可以通过在不同条件下结晶来制备。The term "crystalline polymorph", "polymorph" or "crystalline form" means a crystal structure in which a compound (or a salt or solvate thereof) can crystallize into different crystal packing arrangements, all of which have the same elemental composition . Different crystal forms typically have different X-ray diffraction patterns, infrared spectra, melting points, density, hardness, crystal shape, optical and electrical properties, stability and solubility. Recrystallization solvent, rate of crystallization, storage temperature, and other factors may allow one crystal form to prevail. Crystalline polymorphs of the compounds can be prepared by crystallization under various conditions.
组合物还可以包含一种或多种化合物的盐。盐可以是药学上可接受的盐。如本文所用,“药学上可接受的盐”是指所公开的化合物的衍生物,其中母体化合物通过将现有酸或碱部分转化成其盐形式(例如通过使游离碱基团与合适的有机酸反应)而改变。药学上可接受的盐的实例包括但不限于碱性残基诸如胺的无机酸盐或有机酸盐;酸性残基诸如羧酸的碱盐或有机盐等。代表性酸加成盐包括乙酸盐、己二酸盐、海藻酸盐、抗坏血酸盐、天冬氨酸盐、苯磺酸盐、苯甲酸盐、硫酸氢盐、硼酸盐、丁酸盐、樟脑酸盐、樟脑磺酸盐、柠檬酸盐、环戊烷丙酸盐、二葡糖酸盐、十二烷基硫酸盐、乙磺酸盐、富马酸盐、葡糖庚酸盐(glucoheptonate)、甘油磷酸盐、半硫酸盐、庚酸盐、己酸盐、氢溴酸盐、盐酸盐、氢碘酸盐、2-羟基-乙磺酸盐、乳糖醛酸盐、乳酸盐、月桂酸盐、月桂基硫酸盐、苹果酸盐、马来酸盐、丙二酸盐、甲磺酸盐、2-萘磺酸盐、烟酸盐、硝酸盐、油酸盐、草酸盐、棕榈酸盐、双羟萘酸盐(pamoate)、果胶酸盐(pectinate)、过硫酸盐、3-苯基丙酸盐、磷酸盐、苦味酸盐、新戊酸盐、丙酸盐、硬脂酸盐、琥珀酸盐、硫酸盐、酒石酸盐、硫氰酸盐、甲苯磺酸盐、十一烷酸盐、戊酸盐等。代表性碱金属盐或碱土金属盐包括钠、锂、钾、钙、镁等以及无毒性的铵、季铵和胺阳离子,包括但不限于铵、四甲铵、四乙铵、甲胺、二甲胺、三甲胺、三乙胺、乙胺等。本公开的药学上可接受的盐包括例如由无毒性的无机酸或有机酸形成的母体化合物的常规无毒性盐。本公开的药学上可接受的盐可以由含有碱性或酸性部分的母体化合物通过常规化学方法合成。一般而言,此类盐可以通过使这些化合物的游离酸或游离碱形式与化学计量量的适当碱或酸在水中或在有机溶剂中或者在这两种的混合物中反应来制备;一般而言,优选非水性介质,如乙醚、乙酸乙酯、乙醇、异丙醇或乙腈。The compositions may also contain salts of one or more compounds. The salt can be a pharmaceutically acceptable salt. As used herein, "pharmaceutically acceptable salts" refer to derivatives of the disclosed compounds, wherein the parent compound is obtained by converting an existing acid or base moiety into its salt form (eg, by concatenating the free base group with a suitable organic compound) acid reaction). Examples of pharmaceutically acceptable salts include, but are not limited to, inorganic or organic acid salts of basic residues such as amines; alkali or organic salts of acidic residues such as carboxylic acids, and the like. Representative acid addition salts include acetate, adipate, alginate, ascorbate, aspartate, benzenesulfonate, benzoate, bisulfate, borate, butyrate , camphorate, camphorsulfonate, citrate, cyclopentane propionate, digluconate, lauryl sulfate, ethanesulfonate, fumarate, glucoheptanoate ( glucoheptonate), glycerophosphate, hemisulfate, heptanoate, caproate, hydrobromide, hydrochloride, hydroiodide, 2-hydroxy-ethanesulfonate, lactobionate, lactate , laurate, lauryl sulfate, malate, maleate, malonate, mesylate, 2-naphthalenesulfonate, nicotinate, nitrate, oleate, oxalate , palmitate, pamoate, pectinate, persulfate, 3-phenylpropionate, phosphate, picrate, pivalate, propionate, Stearates, succinates, sulfates, tartrates, thiocyanates, tosylates, undecanoates, valerates, etc. Representative alkali or alkaline earth metal salts include sodium, lithium, potassium, calcium, magnesium, etc. as well as non-toxic ammonium, quaternary ammonium and amine cations, including but not limited to ammonium, tetramethylammonium, tetraethylammonium, methylamine, diethylammonium Methylamine, trimethylamine, triethylamine, ethylamine, etc. Pharmaceutically acceptable salts of the present disclosure include, for example, conventional non-toxic salts of the parent compound formed from non-toxic inorganic or organic acids. The pharmaceutically acceptable salts of the present disclosure can be synthesized from the parent compound containing a basic or acidic moiety by conventional chemical methods. In general, such salts can be prepared by reacting the free acid or free base forms of these compounds with a stoichiometric amount of the appropriate base or acid in water or in an organic solvent or in a mixture of the two; generally , preferably non-aqueous media such as ether, ethyl acetate, ethanol, isopropanol or acetonitrile.
如本文所用,“磷脂”是包含磷酸部分和一个或多个碳链诸如不饱和脂肪酸链的脂质。磷脂可以包含一个或多个多重(例如,双键或三键)键(例如,一处或多处不饱和)。特定的磷脂可有助于与膜的融合。例如,阳离子磷脂可以与膜(例如,细胞膜或细胞内膜)的一种或多种带负电荷的磷脂相互作用。磷脂与膜的融合可以使含脂质的组合物的一种或多种成分穿过膜,从而允许例如将所述一种或多种成分递送至细胞。As used herein, a "phospholipid" is a lipid comprising a phosphate moiety and one or more carbon chains such as unsaturated fatty acid chains. Phospholipids may contain one or more multiple (eg, double or triple) bonds (eg, one or more unsaturations). Certain phospholipids can aid in fusion with the membrane. For example, cationic phospholipids can interact with one or more negatively charged phospholipids of membranes (eg, cell membranes or intracellular membranes). Fusion of the phospholipid to the membrane can allow one or more components of the lipid-containing composition to pass through the membrane, allowing, for example, delivery of the one or more components to a cell.
如本文所用,“多分散性指数”或“PDI”是一种描述体系的粒度分布的均质性的比率。较小的值,例如小于0.3,指示较窄的粒度分布。As used herein, a "polydispersity index" or "PDI" is a ratio that describes the homogeneity of the particle size distribution of a system. Smaller values, eg less than 0.3, indicate a narrower particle size distribution.
如本文所用,术语“多肽”或“所关注的多肽”是指通常通过肽键接合的氨基酸残基的聚合物,其可以是天然产生的(例如,分离的或纯化的)或合成产生的。术语“多肽”、“肽”以及“蛋白质”在本文中可互换使用,以指具有任何长度的氨基酸的聚合物。聚合物可以包含修饰的氨基酸。所述术语还涵盖已被天然修饰或通过介入修饰的氨基酸聚合物;例如,二硫键形成、糖基化、脂质化、乙酰化、磷酸化或任何其他操作或修饰(诸如与标记组分缀合)。定义中还包括例如含有一种或多种氨基酸类似物(包括,例如非天然氨基酸,诸如高半胱氨酸、鸟氨酸、对乙酰基苯丙氨酸、D-氨基酸和肌酸)以及本领域已知的其他修饰的多肽。如本文所用,所述术语是指具有任何大小、结构或功能的蛋白质、多肽和肽。多肽包括编码的多核苷酸产品、天然存在的多肽、合成的多肽、同源物、直系同源物、旁系同源物、前述的片段及其他等效物、变体和类似物。多肽可以是单体或者可以是多分子复合物诸如二聚体、三聚体或四聚体。它们还可以包括单链或多链多肽。最常见地,在多链多肽中发现二硫键。术语多肽还可应用于氨基酸聚合物,其中一个或多个氨基酸残基是对应的天然存在的氨基酸的人工化学类似物。在一些实施方案中,“肽”可以小于或等于50个氨基酸长,例如约5、10、15、20、25、30、35、40、45或50个氨基酸长。As used herein, the term "polypeptide" or "polypeptide of interest" refers to a polymer of amino acid residues, usually joined by peptide bonds, which may be naturally occurring (eg, isolated or purified) or synthetically produced. The terms "polypeptide," "peptide," and "protein" are used interchangeably herein to refer to polymers of amino acids of any length. The polymers may contain modified amino acids. The term also encompasses amino acid polymers that have been modified naturally or by intervention; for example, disulfide bond formation, glycosylation, lipidation, acetylation, phosphorylation, or any other manipulation or modification (such as with labeling components). conjugation). Also included in the definition are, for example, amino acids containing one or more analogs of amino acids (including, for example, unnatural amino acids such as homocysteine, ornithine, para-acetylphenylalanine, D-amino acids, and creatine) and the present Other modified polypeptides known in the art. As used herein, the term refers to proteins, polypeptides and peptides of any size, structure or function. Polypeptides include encoded polynucleotide products, naturally occurring polypeptides, synthetic polypeptides, homologues, orthologues, paralogues, fragments of the foregoing and other equivalents, variants and analogs. Polypeptides may be monomers or may be multimolecular complexes such as dimers, trimers or tetramers. They may also include single-chain or multi-chain polypeptides. Most commonly, disulfide bonds are found in multi-chain polypeptides. The term polypeptide can also be applied to amino acid polymers in which one or more amino acid residues are artificial chemical analogs of corresponding naturally occurring amino acids. In some embodiments, a "peptide" may be less than or equal to 50 amino acids long, eg, about 5, 10, 15, 20, 25, 30, 35, 40, 45, or 50 amino acids long.
如本文所用,“RNA”是指可能天然存在或非天然存在的核糖核酸。例如,RNA可以包含修饰的和/或非天然存在的组分,诸如一个或多个核碱基、核苷、核苷酸或接头。RNA可以包含帽结构、链终止核苷、茎环、polyA序列和/或聚腺苷酸化信号。RNA可以具有编码所关注的多肽的核苷酸序列。As used herein, "RNA" refers to ribonucleic acid that may or may not occur naturally. For example, RNA can comprise modified and/or non-naturally occurring components, such as one or more nucleobases, nucleosides, nucleotides, or linkers. The RNA may contain cap structures, chain terminating nucleosides, stem loops, polyA sequences and/or polyadenylation signals. The RNA can have a nucleotide sequence encoding the polypeptide of interest.
如本文所用,“DNA”是指可能天然存在或非天然存在的脱氧核糖核酸。例如,DNA可以是合成分子,例如体外产生的合成DNA分子。在一些实施方案中,DNA分子是重组分子。如本文所用,“重组DNA分子”是指不作为天然产物存在而是使用分子生物学技术产生的DNA分子。As used herein, "DNA" refers to deoxyribonucleic acid that may or may not occur naturally. For example, the DNA can be a synthetic molecule, such as a synthetic DNA molecule produced in vitro. In some embodiments, the DNA molecule is a recombinant molecule. As used herein, a "recombinant DNA molecule" refers to a DNA molecule that does not exist as a natural product but is produced using molecular biology techniques.
如本文所用,“单一单位剂量”是以一次剂量/一次/单一途径/单一接触点,即单一施用事件施用的任何治疗剂的剂量。As used herein, a "single unit dose" is the dose of any therapeutic agent administered in one dose/one time/single route/single point of contact, ie, a single administration event.
如本文所用,“分次剂量”是将单一单位剂量或总每日剂量分成两次或更多次剂量。As used herein, "fractionated dose" is the division of a single unit dose or total daily dose into two or more doses.
如本文所用,“总每日剂量”是在24小时时间段内给予或规定的量。总每日剂量可以作为单一单位剂量施用。As used herein, a "total daily dose" is the amount administered or prescribed over a 24 hour period. The total daily dose can be administered as a single unit dose.
如本文所用,脂质纳米粒子(例如,空LNP或负载LNP)上下文中的“大小”或“平均大小”是指纳米粒子组合物的平均直径。As used herein, "size" or "average size" in the context of lipid nanoparticles (eg, empty LNPs or loaded LNPs) refers to the average diameter of the nanoparticle composition.
如本文所用,术语“受试者”或“患者”是指可以施用根据本公开的组合物以例如用于实验、诊断、预防和/或治疗目的的任何生物体。典型的受试者包括动物(例如,哺乳动物诸如小鼠、大鼠、兔、非人灵长类动物和人)和/或植物。As used herein, the term "subject" or "patient" refers to any organism to which a composition according to the present disclosure can be administered, eg, for experimental, diagnostic, prophylactic and/or therapeutic purposes. Typical subjects include animals (eg, mammals such as mice, rats, rabbits, non-human primates, and humans) and/or plants.
如本文所用,“靶向的细胞”是指任一种或多种所关注的细胞。这些细胞可见于生物体的体外、体内、原位或者组织或器官中。生物体可以是动物,优选哺乳动物,更优选人并且最优选患者。As used herein, a "targeted cell" refers to any one or more cells of interest. These cells can be found in vitro, in vivo, in situ, or in tissues or organs of an organism. The organism may be an animal, preferably a mammal, more preferably a human and most preferably a patient.
如本文所用,“靶组织”是指其中递送治疗剂和/或预防剂将引起所需的生物学和/或药理学效果的任一种或多种所关注的组织类型。所关注的靶组织的实例包括指定组织、器官和系统或其组。在特定应用中,靶组织可以是肾、肺、脾、血管(例如,冠状动脉内或股骨内)中的血管内皮或肿瘤组织(例如,通过肿瘤内注射)。如本文所用,“脱靶组织”是指其中所编码的蛋白质的表达不会引起所需的生物学和/或药理学效果的任一种或多种组织类型。在特定应用中,脱靶组织可以包括肝和脾。As used herein, "target tissue" refers to any one or more tissue types of interest in which delivery of a therapeutic and/or prophylactic agent will elicit a desired biological and/or pharmacological effect. Examples of target tissues of interest include designated tissues, organs, and systems, or groups thereof. In certain applications, the target tissue may be the kidney, lung, spleen, vascular endothelium in a blood vessel (eg, intracoronary or intrafemoral), or tumor tissue (eg, by intratumoral injection). As used herein, "off-target tissue" refers to any tissue type or types in which expression of the encoded protein does not result in the desired biological and/or pharmacological effect. In certain applications, off-target tissues may include liver and spleen.
术语“治疗剂”或“预防剂”是指在施用至受试者时具有治疗、诊断和/或预防效果和/或引起所需的生物学和/或药理学效果的任何剂。治疗剂也称为“活性剂(active)”或“活性剂(active agent)”。此类剂包括但不限于细胞毒素、放射性离子、化学治疗剂、小分子药物、蛋白质和核酸。The term "therapeutic agent" or "prophylactic agent" refers to any agent that, when administered to a subject, has a therapeutic, diagnostic and/or prophylactic effect and/or elicits a desired biological and/or pharmacological effect. Therapeutic agents are also referred to as "active agents" or "active agents." Such agents include, but are not limited to, cytotoxins, radioactive ions, chemotherapeutics, small molecule drugs, proteins, and nucleic acids.
如本文所用,术语“治疗有效量”意指在施用至罹患或易患感染、疾病、病症和/或病状的受试者时足以治疗所述感染、疾病、病症和/或病状、改善其症状、诊断、预防和/或延迟其发作而递送的剂(例如核酸、药物、组合物、治疗剂、诊断剂、预防剂等)的量。As used herein, the term "therapeutically effective amount" means sufficient to treat, ameliorate the symptoms of, an infection, disease, disorder and/or condition when administered to a subject suffering from or susceptible to the infection, disease, disorder and/or condition , the amount of an agent (eg, nucleic acid, drug, composition, therapeutic, diagnostic, prophylactic, etc.) delivered to diagnose, prevent, and/or delay its onset.
如本文所用,“转染”是指将物质(例如RNA)引入细胞中。转染可以例如在体外、离体或体内发生。As used herein, "transfection" refers to the introduction of a substance (eg, RNA) into a cell. Transfection can occur, for example, in vitro, ex vivo or in vivo.
如本文所用,术语“治疗”是指部分或完全地减轻、改良、改善、缓解特定感染、疾病、病症和/或病状的一种或多种症状或特征,延迟其发作,抑制其进展,降低其严重程度和/或降低其发生率。例如,“治疗”癌症可指抑制肿瘤的存活、生长和/或扩散。可以出于减少发展与疾病、病症和/或病状相关的病理的风险的目的,向没有表现出疾病、病症和/或病状的体征的受试者和/或仅表现出疾病、病症和/或病状的早期体征的受试者施用治疗。As used herein, the term "treating" refers to partially or fully alleviating, ameliorating, ameliorating, alleviating, delaying onset, inhibiting progression, reducing one or more symptoms or characteristics of a particular infection, disease, disorder and/or condition its severity and/or reduce its incidence. For example, "treating" cancer can refer to inhibiting the survival, growth and/or spread of a tumor. Subjects who do not exhibit signs of the disease, disorder and/or condition and/or only exhibit disease, disorder and/or Treatment is administered to subjects with early signs of the condition.
如本文所用,“ζ电位”是例如纳米粒子组合物中脂质的电动电位。As used herein, "zeta potential" is, for example, the zeta potential of lipids in a nanoparticle composition.
纳米粒子组合物Nanoparticle composition
本公开的特征还在于包含如本文所述的根据式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物的脂质纳米粒子。The present disclosure also features the inclusion of formulas (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y) as described herein , (I-Ya)(IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb) Compounds of lipid nanoparticles.
在一些实施方案中,当例如通过动态光散射(DLS)、透射电子显微术、扫描电子显微术或另一种方法测量时,纳米粒子组合物的最大尺寸为1μm或更短(例如,1μm、900nm、800nm、700nm、600nm、500nm、400nm、300nm、200nm、175nm、150nm、125nm、100nm、75nm、50nm或更短)。纳米粒子组合物包括例如脂质纳米粒子(LNP;如空LNP或负载LNP)、脂质体、脂质囊泡和脂质体复合物。在一些实施方案中,纳米粒子组合物是包含一个或多个脂质双层的囊泡。在某些实施方案中,纳米粒子组合物包括通过水性区室隔开的两个或更多个同心双层。脂质双层可以被官能化和/或彼此交联。脂质双层可以包含一种或多种配体、蛋白质或通道。In some embodiments, the nanoparticle composition has a largest dimension of 1 μm or less when measured, for example, by dynamic light scattering (DLS), transmission electron microscopy, scanning electron microscopy, or another method (eg, 1 μm, 900 nm, 800 nm, 700 nm, 600 nm, 500 nm, 400 nm, 300 nm, 200 nm, 175 nm, 150 nm, 125 nm, 100 nm, 75 nm, 50 nm or shorter). Nanoparticle compositions include, for example, lipid nanoparticles (LNPs; eg, empty LNPs or loaded LNPs), liposomes, lipid vesicles, and liposome complexes. In some embodiments, the nanoparticle composition is a vesicle comprising one or more lipid bilayers. In certain embodiments, the nanoparticle composition includes two or more concentric bilayers separated by an aqueous compartment. The lipid bilayers can be functionalized and/or cross-linked to each other. The lipid bilayer may contain one or more ligands, proteins or channels.
纳米粒子组合物包含脂质组分,其包含至少一种根据式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物。例如,纳米粒子组合物的脂质组分可以包含一种或多种表1的化合物。纳米粒子组合物还可以包含多种其他组分。例如,纳米粒子组合物的脂质组分除了根据式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的脂质之外,还可以包含一种或多种其他脂质。The nanoparticle composition comprises a lipid component comprising at least one according to formula (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y), (I-Ya)(IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or ( Compounds of IIb). For example, the lipid component of the nanoparticle composition may comprise one or more compounds of Table 1. The nanoparticle composition may also contain a variety of other components. For example, lipid components of nanoparticle compositions other than those according to formulae (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y) , (I-Ya)(IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb) In addition to lipids, one or more other lipids may also be included.
阳离子/可离子化脂质Cationic/Ionizable Lipids
纳米粒子(例如,空LNP或负载LNP)除了根据式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的脂质之外,还可以包含一种或多种阳离子和/或可离子化脂质(例如在生理pH下可以带正电荷或部分正电荷的脂质)。阳离子和/或可离子化脂质可以选自由以下组成的非限制性组:3-(二-十二烷基氨基)-N1,N1,4-三十二烷基-1-哌嗪乙胺(KL10)、N1-[2-(二-十二烷基氨基)乙基]-N1,N4,N4-三十二烷基-1,4-哌嗪二乙胺(KL22)、14,25-二-十三烷基-15,18,21,24-四氮杂-三十八烷(KL25)、1,2-二亚油基氧基-N,N-二甲基氨基丙烷(DLin-DMA)、2,2-二亚油基-4-二甲基氨基甲基-[1,3]-二氧杂环戊烷(DLin-K-DMA)、4-(二甲基氨基)丁酸三十七碳-6,9,28,31-四烯-19-基酯(DLin-MC3-DMA)、2,2-二亚油基-4-(2-二甲基氨基乙基)-[1,3]-二氧杂环戊烷(DLin-KC2-DMA)、1,2-二油基氧基-N,N-二甲基氨基丙烷(DODMA)、2-({8-[(3β)-胆固-5-烯-3-基氧基]辛基}氧基)-N,N-二甲基-3-[(9Z,12Z)-十八-9,12-二烯-1-基氧基]丙-1-胺(辛基-CLinDMA)、(2R)-2-({8-[(3β)-胆固-5-烯-3-基氧基]辛基}氧基)-N,N-二甲基-3-[(9Z,12Z)-十八-9,12-二烯-1-基氧基]丙-1-胺(辛基-CLinDMA(2R))以及(2S)-2-({8-[(3β)-胆固-5-烯-3-基氧基]辛基}氧基)-N,N-二甲基-3-[(9Z,12Z)-十八-9,12-二烯-1-基氧基]丙-1-胺(辛基-CLinDMA(2S))。除了这些之外,阳离子脂质还可以是包含环状胺基的脂质。Nanoparticles (eg, empty LNP or loaded LNP) except according to formulae (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y) , (I-Ya)(IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb) In addition to lipids, one or more cationic and/or ionizable lipids (eg, lipids that may be positively or partially positively charged at physiological pH) may be included. Cationic and/or ionizable lipids may be selected from the non-limiting group consisting of: 3-(di-dodecylamino)-N1,N1,4-tridodecyl-1-piperazineethylamine (KL10), N1-[2-(di-dodecylamino)ethyl]-N1,N4,N4-tridodecyl-1,4-piperazinediethylamine (KL22), 14,25 -Di-tridecyl-15,18,21,24-tetraaza-trioctadecane (KL25), 1,2-dilinoleyloxy-N,N-dimethylaminopropane (DLin -DMA), 2,2-dilinoleyl-4-dimethylaminomethyl-[1,3]-dioxolane (DLin-K-DMA), 4-(dimethylamino) Heptadeca-6,9,28,31-tetraen-19-yl butyrate (DLin-MC3-DMA), 2,2-dilinole-4-(2-dimethylaminoethyl) )-[1,3]-dioxolane (DLin-KC2-DMA), 1,2-dioleyloxy-N,N-dimethylaminopropane (DODMA), 2-({8 -[(3β)-cholest-5-en-3-yloxy]octyl}oxy)-N,N-dimethyl-3-[(9Z,12Z)-octadec-9,12- Dien-1-yloxy]prop-1-amine (octyl-CLinDMA), (2R)-2-({8-[(3β)-cholest-5-en-3-yloxy]octane yl}oxy)-N,N-dimethyl-3-[(9Z,12Z)-octadec-9,12-dien-1-yloxy]propan-1-amine (octyl-CLinDMA( 2R)) and (2S)-2-({8-[(3β)-cholest-5-en-3-yloxy]octyl}oxy)-N,N-dimethyl-3-[ (9Z,12Z)-Octadec-9,12-dien-1-yloxy]propan-1-amine (octyl-CLinDMA(2S)). In addition to these, cationic lipids may also be lipids containing cyclic amine groups.
结构性脂质structured lipids
脂质纳米粒子(例如,空LNP或负载LNP)可以包含一种或多种结构性脂质。结构性脂质可以选自由但不限于由以下组成的组:胆固醇、粪固醇(fecosterol)、谷固醇(sitosterol)、麦角固醇、菜油固醇、豆固醇、菜籽固醇、番茄碱(tomatidine)、番茄素(tomatine)、熊果酸、α-生育酚及其混合物。在一些实施方案中,结构性脂质是胆固醇。在一些实施方案中,结构性脂质包括胆固醇和皮质类固醇(诸如泼尼松龙、地塞米松、泼尼松和氢化可的松)或其组合。在一些实施方案中,结构性脂质是: Lipid nanoparticles (eg, empty LNPs or loaded LNPs) can comprise one or more structured lipids. The structural lipid may be selected from, but is not limited to, the group consisting of cholesterol, fecosterol, sitosterol, ergosterol, campesterol, stigmasterol, brassicasterol, tomato Tomatidine, tomatine, ursolic acid, alpha-tocopherol and mixtures thereof. In some embodiments, the structural lipid is cholesterol. In some embodiments, the structural lipids include cholesterol and corticosteroids (such as prednisolone, dexamethasone, prednisone, and hydrocortisone), or a combination thereof. In some embodiments, the structured lipid is:
磷脂Phospholipids
脂质纳米粒子(例如,空LNP或负载LNP)可以包含一种或多种磷脂,诸如一种或多种(多)不饱和脂质。磷脂可以组装成一个或多个脂质双层。一般而言,磷脂可以包含磷脂部分和一个或多个脂肪酸部分。例如,磷脂可以是根据式(IV)的脂质:Lipid nanoparticles (eg, empty LNPs or loaded LNPs) may comprise one or more phospholipids, such as one or more (poly)unsaturated lipids. Phospholipids can assemble into one or more lipid bilayers. In general, a phospholipid may comprise a phospholipid moiety and one or more fatty acid moieties. For example, the phospholipid can be a lipid according to formula (IV):
其中Rp表示磷脂部分并且RA和RB表示具有或不具有不饱和的脂肪酸部分,这些脂肪酸部分可以是相同或不同的。磷脂部分可以选自由以下组成的非限制性组:磷脂酰胆碱、磷脂酰乙醇胺、磷脂酰甘油、磷脂酰丝氨酸、磷脂酸、2-溶血磷脂酰胆碱和鞘磷脂。脂肪酸部分可以选自由以下组成的非限制性组:月桂酸、肉豆蔻酸、肉豆蔻烯酸、棕榈酸、棕榈油酸、硬脂酸、油酸、亚油酸、α-亚麻酸、芥酸、植烷酸、花生酸、花生四烯酸、二十碳五烯酸、山嵛酸、二十二碳五烯酸和二十二碳六烯酸。还预期包括具有修饰和取代的天然物质的非天然物质,所述修饰和取代包括支化、氧化、环化和炔烃。例如,磷脂可以用一种或多种炔烃(例如,其中一个或多个双键被三键替代的烯基)官能化或与一种或多种炔烃交联。在适当的反应条件下,炔基在暴露于叠氮化物时可能经历铜催化的环加成反应。此类反应可用于使纳米粒子(例如,空LNP或负载LNP)的脂质双层官能化以促进膜渗透或细胞识别,或将纳米粒子(例如,空LNP或负载LNP)缀合至有用组分诸如靶向部分或成像部分(例如染料)。wherein R p represents a phospholipid moiety and RA and RB represent fatty acid moieties with or without unsaturation, which may be the same or different. The phospholipid moiety can be selected from the non-limiting group consisting of phosphatidylcholine, phosphatidylethanolamine, phosphatidylglycerol, phosphatidylserine, phosphatidic acid, 2-lysophosphatidylcholine, and sphingomyelin. The fatty acid moiety may be selected from the non-limiting group consisting of lauric acid, myristic acid, myristoleic acid, palmitic acid, palmitoleic acid, stearic acid, oleic acid, linoleic acid, alpha-linolenic acid, erucic acid , phytanic acid, arachidonic acid, arachidonic acid, eicosapentaenoic acid, behenic acid, docosapentaenoic acid and docosahexaenoic acid. It is also contemplated to include non-natural materials with modified and substituted natural materials, including branching, oxidation, cyclization, and alkynes. For example, phospholipids can be functionalized with one or more alkynes (eg, alkenyl groups in which one or more double bonds are replaced by triple bonds) or crosslinked with one or more alkynes. Under appropriate reaction conditions, alkynyl groups may undergo copper-catalyzed cycloaddition reactions when exposed to azides. Such reactions can be used to functionalize the lipid bilayer of nanoparticles (eg, empty or loaded LNPs) to facilitate membrane penetration or cell recognition, or to conjugate nanoparticles (eg, empty or loaded LNPs) to useful groups. Such as targeting moieties or imaging moieties (eg dyes).
可用于这些组合物和方法中的磷脂可以选自由以下组成的非限制性组:1,2-二硬脂酰基-sn-甘油-3-磷酸胆碱(DSPC)、1,2-二油酰基-sn-甘油-3-磷酸乙醇胺(DOPE)、1,2-二亚油酰基-sn-甘油-3-磷酸胆碱(DLPC)、1,2-二肉豆蔻酰基-sn-甘油-磷酸胆碱(DMPC)、1,2-二油酰基-sn-甘油-3-磷酸胆碱(DOPC)、1,2-二棕榈酰基-sn-甘油-3-磷酸胆碱(DPPC)、1,2-二-十一烷酰基-sn-甘油-磷酸胆碱(DUPC)、1-棕榈酰基-2-油酰基-sn-甘油-3-磷酸胆碱(POPC)、1,2-二-O-十八碳烯基-sn-甘油-3-磷酸胆碱(18:0Diether PC)、1-油酰基-2-胆固醇基半琥珀酰基-sn-甘油-3-磷酸胆碱(OChemsPC)、1-十六烷基-sn-甘油-3-磷酸胆碱(C16 Lyso PC)、1,2-二亚麻酰基(dilinolenoyl)-sn-甘油-3-磷酸胆碱、1,2-二花生四烯酰基-sn-甘油-3-磷酸胆碱、1,2-二-二十二碳六烯酰基-sn-甘油-3-磷酸胆碱、1,2-二植烷酰基-sn-甘油-3-磷酸乙醇胺(ME 16.0PE)、1,2-二硬脂酰基-sn-甘油-3-磷酸乙醇胺、1,2-二亚油酰基-sn-甘油-3-磷酸乙醇胺、1,2-二亚麻酰基-sn-甘油-3-磷酸乙醇胺、1,2-二花生四烯酰基-sn-甘油-3-磷酸乙醇胺、1,2-二-二十二碳六烯酰基-sn-甘油-3-磷酸乙醇胺、1,2-二油酰基-sn-甘油-3-磷酸-rac-(1-甘油)钠盐(DOPG)、二棕榈酰基磷脂酰甘油(DPPG)、棕榈酰基油酰基磷脂酰乙醇胺(POPE)、二硬脂酰基-磷脂酰-乙醇胺(DSPE)、二棕榈酰基磷脂酰乙醇胺(DPPE)、二肉豆蔻酰基磷酸乙醇胺(DMPE)、1-硬脂酰基-2-油酰基-磷脂酰乙醇胺(SOPE)、1-硬脂酰基-2-油酰基-磷脂酰胆碱(SOPC)、鞘磷脂、磷脂酰胆碱、磷脂酰乙醇胺、磷脂酰丝氨酸、磷脂酰肌醇、磷脂酸、棕榈酰基油酰基磷脂酰胆碱、溶血磷脂酰胆碱、溶血磷脂酰乙醇胺(LPE)及其混合物。在一些实施方案中,脂质纳米粒子(例如,空LNP或负载LNP)包含DSPC。在某些实施方案中,脂质纳米粒子(例如,空LNP或负载LNP)包含DOPE。在一些实施方案中,脂质纳米粒子(例如,空LNP或负载LNP)包含DSPC和DOPE两者。Phospholipids useful in these compositions and methods can be selected from the non-limiting group consisting of 1,2-distearoyl-sn-glycero-3-phosphocholine (DSPC), 1,2-dioleoyl -sn-glycero-3-phosphoethanolamine (DOPE), 1,2-dilinoleoyl-sn-glycero-3-phosphocholine (DLPC), 1,2-dimyristoyl-sn-glycero-phosphocholine Base (DMPC), 1,2-dioleoyl-sn-glycero-3-phosphocholine (DOPC), 1,2-dipalmitoyl-sn-glycero-3-phosphocholine (DPPC), 1,2 - Di-undecanoyl-sn-glycero-phosphocholine (DUPC), 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine (POPC), 1,2-di-O- Octadecenyl-sn-glycero-3-phosphocholine (18:0 Diether PC), 1-oleoyl-2-cholesterylhemisuccinyl-sn-glycero-3-phosphocholine (OChemsPC), 1- Cetyl-sn-glycero-3-phosphocholine (C16 Lyso PC), 1,2-dilinolenoyl-sn-glycero-3-phosphocholine, 1,2-diarachidonoyl -sn-glycero-3-phosphocholine, 1,2-di-docosahexaenoyl-sn-glycero-3-phosphocholine, 1,2-diphytanoyl-sn-glycero-3- Phosphoethanolamine (ME 16.0PE), 1,2-distearoyl-sn-glycero-3-phosphoethanolamine, 1,2-dilinoleoyl-sn-glycero-3-phosphoethanolamine, 1,2-dilinolenic acid Acyl-sn-glycero-3-phosphoethanolamine, 1,2-diarachidonoyl-sn-glycero-3-phosphoethanolamine, 1,2-di-docosahexaenoyl-sn-glycero-3- Phosphoethanolamine, 1,2-dioleoyl-sn-glycero-3-phosphate-rac-(1-glycerol) sodium salt (DOPG), dipalmitoylphosphatidylglycerol (DPPG), palmitoyloleoylphosphatidylethanolamine ( POPE), distearoyl-phosphatidyl-ethanolamine (DSPE), dipalmitoyl phosphatidylethanolamine (DPPE), dimyristoyl phosphoethanolamine (DMPE), 1-stearoyl-2-oleoyl-phosphatidylethanolamine (SOPE), 1-stearoyl-2-oleoyl-phosphatidylcholine (SOPC), sphingomyelin, phosphatidylcholine, phosphatidylethanolamine, phosphatidylserine, phosphatidylinositol, phosphatidic acid, palmitoyl oil Acylphosphatidylcholines, lysophosphatidylcholines, lysophosphatidylethanolamines (LPE) and mixtures thereof. In some embodiments, the lipid nanoparticles (eg, empty LNPs or loaded LNPs) comprise DSPC. In certain embodiments, the lipid nanoparticles (eg, empty LNPs or loaded LNPs) comprise DOPE. In some embodiments, lipid nanoparticles (eg, empty LNPs or loaded LNPs) comprise both DSPC and DOPE.
PEG脂质PEG lipids
脂质纳米粒子(例如,空LNP或负载LNP)可以包含一种或多种PEG或PEG修饰的脂质。这些物质可以替代地称为PEG化脂质。PEG脂质是用聚乙二醇修饰的脂质。PEG脂质可以选自由以下组成的非限制性组:PEG修饰的磷脂酰乙醇胺、PEG修饰的磷脂酸、PEG修饰的神经酰胺(PEG-CER)、PEG修饰的二烷基胺、PEG修饰的二酰基甘油(PEG-DEG)、PEG修饰的二烷基甘油及其混合物。例如,PEG脂质可以是PEG-c-DOMG、PEG-DMG、PEG-DLPE、PEG-DMPE、PEG-DPPC或PEG-DSPE脂质。Lipid nanoparticles (eg, empty LNPs or loaded LNPs) can comprise one or more PEG or PEG-modified lipids. These substances may alternatively be referred to as PEGylated lipids. PEG lipids are lipids modified with polyethylene glycol. PEG lipids can be selected from the non-limiting group consisting of: PEG-modified phosphatidylethanolamine, PEG-modified phosphatidic acid, PEG-modified ceramide (PEG-CER), PEG-modified dialkylamine, PEG-modified dialkylamine Acylglycerols (PEG-DEG), PEG-modified dialkylglycerols, and mixtures thereof. For example, the PEG lipid can be a PEG-c-DOMG, PEG-DMG, PEG-DLPE, PEG-DMPE, PEG-DPPC, or PEG-DSPE lipid.
在某些实施方案中,PEG脂质选自由以下组成的组:PEG修饰的磷脂酰乙醇胺、PEG修饰的磷脂酸、PEG修饰的神经酰胺、PEG修饰的二烷基胺、PEG修饰的二酰基甘油、PEG修饰的二烷基甘油。In certain embodiments, the PEG lipid is selected from the group consisting of: PEG-modified phosphatidylethanolamine, PEG-modified phosphatidic acid, PEG-modified ceramide, PEG-modified dialkylamine, PEG-modified diacylglycerol , PEG-modified dialkylglycerol.
在某些实施方案中,PEG脂质选自由以下组成的组:1,2-二肉豆蔻酰基-sn-甘油甲氧基聚乙二醇(PEG-DMG)、1,2-二硬脂酰基-sn-甘油-3-磷酸乙醇胺-N-[氨基(聚乙二醇)](PEG-DSPE)、PEG-二硬脂基甘油(PEG-DSG)、PEG-二棕榈酰基、PEG-二油酰基、PEG-二硬脂基、PEG-二酰基甘氨酰胺(PEG-DAG)、PEG-二棕榈酰基磷脂酰乙醇胺(PEG-DPPE)或PEG-1,2-二肉豆蔻基氧基丙基-3-胺(PEG-c-DMA)。例如,在一些实施方案中,所述PEG脂质是PEG-DMG。In certain embodiments, the PEG lipid is selected from the group consisting of 1,2-dimyristoyl-sn-glyceromethoxypolyethylene glycol (PEG-DMG), 1,2-distearoyl -sn-glycerol-3-phosphoethanolamine-N-[amino(polyethylene glycol)](PEG-DSPE), PEG-distearylglycerol (PEG-DSG), PEG-dipalmitoyl, PEG-dioleyl Acyl, PEG-distearyl, PEG-diacylglycinamide (PEG-DAG), PEG-dipalmitoylphosphatidylethanolamine (PEG-DPPE) or PEG-1,2-dimyristyloxypropyl -3-amine (PEG-c-DMA). For example, in some embodiments, the PEG lipid is PEG-DMG.
在某些实施方案中,所述PEG脂质是式(PL-I)的化合物:In certain embodiments, the PEG lipid is a compound of formula (PL-I):
或其盐,其中:or a salt thereof, where:
R3PL1是–OROPL1;R 3PL1 is –OR OPL1 ;
ROPL1是氢、任选取代的烷基或氧保护基团;R OPL1 is hydrogen, an optionally substituted alkyl or oxygen protecting group;
rPL1是介于1与100之间的整数,包括端值;r PL1 is an integer between 1 and 100, inclusive;
L1是任选取代的C1-10亚烷基,其中任选取代的C1-10亚烷基中的至少一个亚甲基独立地被替代为:任选取代的亚碳环基、任选取代的亚杂环基、任选取代的亚芳基、任选取代的亚杂芳基、O、N(RNPL1)、S、C(O)、C(O)N(RNPL1)、NRNPL1C(O)、C(O)O、OC(O)、OC(O)O、OC(O)N(RNPL1)、NRNPL1C(O)O或NRNPL1C(O)N(RNPL1);L 1 is an optionally substituted C 1-10 alkylene group, wherein at least one methylene group in the optionally substituted C 1-10 alkylene group is independently replaced by: optionally substituted carbocyclylene, any optionally substituted heterocyclylene, optionally substituted arylene, optionally substituted heteroarylene, O, N(R NPL1 ), S, C(O), C(O)N(R NPL1 ), NR NPL1 C(O), C(O)O, OC(O), OC(O)O, OC(O)N(R NPL1 ), NR NPL1 C(O)O or NR NPL1 C(O)N( R NPL1 );
D是通过点击化学获得的部分或在生理条件下可切割的部分;D is a moiety obtained by click chemistry or a moiety cleavable under physiological conditions;
mPL1是0、1、2、3、4、5、6、7、8、9或10;m PL1 is 0, 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10;
A具有下式: A has the following formula:
L2的每种情况独立地是键或任选取代的C1-6亚烷基,其中任选取代的C1-6亚烷基中的一个亚甲基单元任选地被替代为:O、N(RNPL1)、S、C(O)、C(O)N(RNPL1)、NRNPL1C(O)、C(O)O、OC(O)、OC(O)O、OC(O)N(RNPL1)、NRNPL1C(O)O或NRNPL1C(O)N(RNPL1);Each instance of L is independently a bond or an optionally substituted C1-6 alkylene where one methylene unit in the optionally substituted C1-6 alkylene is optionally replaced with: O , N(R NPL1 ), S, C(O), C(O)N(R NPL1 ), NR NPL1 C(O), C(O)O, OC(O), OC(O)O, OC( O)N(R NPL1 ), NR NPL1 C(O)O or NR NPL1 C(O)N(R NPL1 );
R2SL的每种情况独立地是任选取代的C1-30烷基、任选取代的C1-30烯基或任选取代的C1-30炔基;任选地其中R2SL的一个或多个亚甲基单元独立地被替代为:任选取代的亚碳环基、任选取代的亚杂环基、任选取代的亚芳基、任选取代的亚杂芳基、N(RNPL1)、O、S、C(O)、C(O)N(RNPL1)、NRNPL1C(O)、NRNPL1C(O)N(RNPL1)、C(O)O、OC(O)、OC(O)O、OC(O)N(RNPL1)、NRNPL1C(O)O、C(O)S、SC(O)、C(=NRNPL1)、C(=NRNPL1)N(RNPL1)、NRNPL1C(=NRNPL1)、NRNPL1C(=NRNPL1)N(RNPL1)、C(S)、C(S)N(RNPL1)、NRNPL1C(S)、NRNPL1C(S)N(RNPL1)、S(O)、OS(O)、S(O)O、OS(O)O、OS(O)2、S(O)2O、OS(O)2O、N(RNPL1)S(O)、S(O)N(RNPL1)、N(RNPL1)S(O)N(RNPL1)、OS(O)N(RNPL1)、N(RNPL1)S(O)O、S(O)2、N(RNPL1)S(O)2、S(O)2N(RNPL1)、N(RNPL1)S(O)2N(RNPL1)、OS(O)2N(RNPL1)或N(RNPL1)S(O)2O;Each instance of R 2SL is independently optionally substituted C 1-30 alkyl, optionally substituted C 1-30 alkenyl, or optionally substituted C 1-30 alkynyl; optionally one of wherein R 2SL or more methylene units are independently replaced with: optionally substituted carbocyclylene, optionally substituted heterocyclylene, optionally substituted arylene, optionally substituted heteroarylene, N( R NPL1 ), O, S, C(O), C(O)N(R NPL1 ), NR NPL1 C(O), NR NPL1 C(O)N(R NPL1 ), C(O)O, OC( O), OC(O)O, OC(O)N(R NPL1 ), NR NPL1 C(O)O, C(O)S, SC(O), C(=NR NPL1 ), C(=NR NPL1 )N(R NPL1 ), NR NPL1 C(=NR NPL1 ), NR NPL1 C(=NR NPL1 )N(R NPL1 ), C(S), C(S)N(R NPL1 ), NR NPL1 C(S ), NR NPL1 C(S)N(R NPL1 ), S(O), OS(O), S(O)O, OS(O)O, OS(O) 2 , S(O) 2 O, OS (O) 2 O, N(R NPL1 )S(O), S(O)N(R NPL1 ), N(R NPL1 )S(O)N(R NPL1 ), OS(O)N(R NPL1 ) , N(R NPL1 )S(O)O, S(O) 2 , N(R NPL1 )S(O) 2 , S(O) 2 N(R NPL1 ), N(R NPL1 )S(O) 2 N(R NPL1 ), OS(O) 2 N(R NPL1 ) or N(R NPL1 )S(O) 2 O;
RNPL1的每种情况独立地是氢、任选取代的烷基或氮保护基团;Each instance of R NPL1 is independently a hydrogen, optionally substituted alkyl or nitrogen protecting group;
环B是任选取代的碳环基、任选取代的杂环基、任选取代的芳基或任选取代的杂芳基;并且Ring B is optionally substituted carbocyclyl, optionally substituted heterocyclyl, optionally substituted aryl, or optionally substituted heteroaryl; and
pSL是1或2。 pSL is 1 or 2.
在某些实施方案中,所述PEG脂质是式(PL-I-OH)的化合物:In certain embodiments, the PEG lipid is a compound of formula (PL-I-OH):
或其盐。 or its salt.
在某些实施方案中,所述PEG脂质是式(PL-II-OH)的化合物:In certain embodiments, the PEG lipid is a compound of formula (PL-II-OH):
或其盐或异构体,其中: or a salt or isomer thereof, wherein:
R3PEG是–ORO;R 3PEG is -OR O ;
RO是氢、C1-6烷基或氧保护基团;R O is a hydrogen, C 1-6 alkyl or oxygen protecting group;
rPEG是介于1与100之间的整数;r PEG is an integer between 1 and 100;
R5PEG是C10-40烷基、C10-40烯基或C10-40炔基;并且任选地R5PEG的一个或多个亚甲基独立地被替代为:C3-10亚碳环基、4至10元亚杂环基、C6-10亚芳基、4至10元亚杂芳基、–N(RNPEG)–、–O–、–S–、–C(O)–、–C(O)N(RNPEG)–、–NRNPEGC(O)–、–NRNPEGC(O)N(RNPEG)–、–C(O)O–、–OC(O)–、–OC(O)O–、–OC(O)N(RNPEG)–、–NRNPEGC(O)O–、–C(O)S–、–SC(O)–、–C(=NRNPEG)–、–C(=NRNPEG)N(RNPEG)–、–NRNPEGC(=NRNPEG)–、–NRNPEGC(=NRNPEG)N(RNPEG)–、–C(S)–、–C(S)N(RNPEG)–、–NRNPEGC(S)–、–NRNPEGC(S)N(RNPEG)–、–S(O)–、–OS(O)–、–S(O)O–、–OS(O)O–、–OS(O)2–、–S(O)2O–、–OS(O)2O–、–N(RNPEG)S(O)–、–S(O)N(RNPEG)–、–N(RNPEG)S(O)N(RNPEG)–、–OS(O)N(RNPEG)–、–N(RNPEG)S(O)O–、–S(O)2–、–N(RNPEG)S(O)2–、–S(O)2N(RNPEG)–、–N(RNPEG)S(O)2N(RNPEG)–、–OS(O)2N(RNPEG)–或–N(RNPEG)S(O)2O–;并且R 5PEG is C 10-40 alkyl, C 10-40 alkenyl, or C 10-40 alkynyl; and optionally one or more methylene groups of R 5PEG are independently replaced with: C 3-10 carbonylene Cyclic, 4- to 10-membered heterocyclylene, C 6-10 -membered arylene, 4- to 10-membered heteroarylene, –N(R NPEG )–, –O–, –S–, –C(O) –, –C(O)N(R NPEG )–, –NR NPEG C(O)–, –NR NPEG C(O)N(R NPEG )–, –C(O)O–, –OC(O) –, –OC(O)O–, –OC(O)N(R NPEG )–, –NR NPEG C(O)O–, –C(O)S–, –SC(O)–, –C( =NR NPEG )–, –C(=NR NPEG )N(R NPEG )–, –NR NPEG C(=NR NPEG )–, –NR NPEG C(=NR NPEG )N(R NPEG )–, –C( S)–, –C(S)N(R NPEG )–, –NR NPEG C(S)–, –NR NPEG C(S)N(R NPEG )–, –S(O)–, –OS(O )–, –S(O)O–, –OS(O)O–, –OS(O) 2 –, –S(O) 2 O–, –OS(O) 2 O–, –N(R NPEG )S(O)–, –S(O)N(R NPEG )–, –N(R NPEG )S(O)N(R NPEG )–, –OS(O)N(R NPEG )–, –N (R NPEG )S(O)O–, –S(O) 2 –, –N(R NPEG )S(O) 2 –, –S(O) 2 N(R NPEG )–, –N(R NPEG )S(O) 2 N(R NPEG )–, –OS(O) 2 N(R NPEG )– or –N(R NPEG )S(O) 2 O–; and
RNPEG的每种情况独立地是氢、C1-6烷基或氮保护基团。Each instance of R NPEG is independently a hydrogen, C1-6 alkyl or nitrogen protecting group.
在某些实施方案中,在式(PL-II-OH)的PEG脂质中,r是介于40与50之间的整数。例如,r选自由40、41、42、43、44、45、46、47、48、49和50组成的组。例如,r是45。In certain embodiments, in the PEG lipid of formula (PL-II-OH), r is an integer between 40 and 50. For example, r is selected from the group consisting of 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, and 50. For example, r is 45.
在某些实施方案中,在式(PL-II-OH)的PEG脂质中,R5是C17烷基。In certain embodiments, in the PEG lipid of formula (PL-II - OH), R5 is C17 alkyl.
在某些实施方案中,所述PEG脂质是式(PL-II)的化合物:In certain embodiments, the PEG lipid is a compound of formula (PL-II):
其中rPEG是介于1与100之间的整数。 where rPEG is an integer between 1 and 100.
在某些实施方案中,所述PEG脂质是式(PEG-1)的化合物:In certain embodiments, the PEG lipid is a compound of formula (PEG-1):
在某些实施方案中,所述PEG脂质是式(PL-III)的化合物:In certain embodiments, the PEG lipid is a compound of formula (PL-III):
或其盐或异构体,其中sPL1是介于1与100之间的整数。 or a salt or isomer thereof, wherein s PL1 is an integer between 1 and 100.
在某些实施方案中,所述PEG脂质是下式的化合物:In certain embodiments, the PEG lipid is a compound of the formula:
在某些实施方案中,在纳米粒子制剂中掺入式(PL-I)、PL-I-OH)、(PL-II)、(PL-II-OH)、(PL-III)、PEG2k-DMG或PEG-1中的一个的脂质,可以改善脂质纳米粒子制剂的药代动力学和/或生物分布。例如,在纳米粒子制剂中掺入式(PL-II-OH)、(PL-IIa-OH)、(PL-II)或PEG-1中的一个的脂质可以降低加速血液清除(ABC)效应。In certain embodiments, nanoparticle formulations incorporate formula (PL-I), PL-I-OH), (PL-II), (PL-II-OH), (PL-III), PEG 2k A lipid of one of DMG or PEG-1, which can improve the pharmacokinetics and/or biodistribution of lipid nanoparticle formulations. For example, incorporation of lipids of one of the formulae (PL-II-OH), (PL-IIa-OH), (PL-II) or PEG-1 in nanoparticle formulations can reduce the accelerated blood clearance (ABC) effect .
佐剂adjuvant
在一些实施方案中,包含一种或多种本文所述的脂质的脂质纳米粒子(例如,空LNP或负载LNP)还可以包含一种或多种佐剂,例如吡喃葡萄糖基脂质佐剂(GLA)、CpG寡脱氧核苷酸(例如,A类或B类)、聚(I:C)、氢氧化铝和Pam3CSK4。In some embodiments, lipid nanoparticles (eg, empty LNPs or loaded LNPs) comprising one or more lipids described herein may also comprise one or more adjuvants, eg, glucopyranosyl lipids Adjuvants (GLA), CpG oligodeoxynucleotides (eg, class A or class B), poly(I:C), aluminum hydroxide, and Pam3CSK4.
治疗剂therapeutic agent
脂质纳米粒子(例如,空LNP或负载LNP)可以包含一种或多种治疗剂和/或预防剂。本公开的特征在于将治疗剂和/或预防剂递送至哺乳动物细胞或器官、在哺乳动物细胞中产生所关注的多肽以及治疗有需要的哺乳动物的疾病或病症的方法,这些方法包括向哺乳动物施用包含治疗剂和/或预防剂的脂质纳米粒子(例如,空LNP或负载LNP)和/或使哺乳动物细胞与所述脂质纳米粒子接触。Lipid nanoparticles (eg, empty LNPs or loaded LNPs) can contain one or more therapeutic and/or prophylactic agents. The present disclosure features methods of delivering therapeutic and/or prophylactic agents to mammalian cells or organs, producing polypeptides of interest in mammalian cells, and treating a disease or disorder in a mammal in need thereof, including administering to a mammalian The animals are administered lipid nanoparticles (eg, empty LNPs or loaded LNPs) comprising therapeutic and/or prophylactic agents and/or contact mammalian cells with the lipid nanoparticles.
治疗剂和/或预防剂包括生物活性物质并且替代地称为“活性剂”。治疗剂和/或预防剂可以是一旦被递送至细胞或器官,就会在细胞、器官或其他身体组织或系统中引起希望的变化的物质。此类物质可用于治疗一种或多种疾病、病症或病状。在一些实施方案中,治疗剂和/或预防剂是可用于治疗特定疾病、病症或病状的小分子药物。Therapeutic and/or prophylactic agents include biologically active substances and are alternatively referred to as "active agents." Therapeutic and/or prophylactic agents can be substances that, once delivered to the cell or organ, cause a desired change in the cell, organ, or other body tissue or system. Such substances can be used to treat one or more diseases, disorders or conditions. In some embodiments, the therapeutic and/or prophylactic agent is a small molecule drug that can be used to treat a particular disease, disorder or condition.
在一些实施方案中,治疗剂和/或预防剂是疫苗、引发免疫反应的化合物(例如,编码蛋白质或多肽或肽、或者蛋白质或多肽、或者蛋白质的多核苷酸或核酸分子)和/或另一种治疗剂和/或预防剂。疫苗包含能够提供针对与感染性疾病相关的一种或多种病状的免疫的化合物和制品,并且可以包含编码感染性疾病源性抗原和/或表位的mRNA。疫苗还可以包含引导针对癌细胞的免疫反应的化合物和制品,并且可以包含编码肿瘤细胞源性抗原、表位和/或新表位的mRNA。在一些实施方案中,能够引发免疫反应的疫苗和/或化合物通过本公开的组合物在肌肉内施用。In some embodiments, the therapeutic and/or prophylactic agent is a vaccine, a compound that elicits an immune response (eg, a polynucleotide or nucleic acid molecule encoding a protein or polypeptide or peptide, or a protein or polypeptide, or a protein) and/or another A therapeutic and/or preventive agent. Vaccines comprise compounds and preparations capable of providing immunity against one or more conditions associated with infectious disease, and may comprise mRNA encoding infectious disease-derived antigens and/or epitopes. Vaccines may also include compounds and preparations that direct an immune response against cancer cells, and may include mRNA encoding tumor cell-derived antigens, epitopes, and/or neo-epitopes. In some embodiments, vaccines and/or compounds capable of eliciting an immune response are administered intramuscularly by the compositions of the present disclosure.
在其他实施方案中,治疗剂和/或预防剂是蛋白质,例如补充或替代所关注的天然存在的蛋白质所需的蛋白质。此类蛋白质或多肽可以是天然存在的,或者可以使用本领域已知的方法进行修饰,例如以增加半衰期。示例性蛋白质是细胞内的、跨膜的或分泌的。In other embodiments, the therapeutic and/or prophylactic agent is a protein, eg, a protein required to supplement or replace the naturally occurring protein of interest. Such proteins or polypeptides may be naturally occurring, or may be modified using methods known in the art, eg, to increase half-life. Exemplary proteins are intracellular, transmembrane, or secreted.
多核苷酸和核酸Polynucleotides and Nucleic Acids
在一些实施方案中,治疗剂是增强(即,增加、刺激、上调)蛋白质表达的剂。可用于增强蛋白质表达的治疗剂类型的非限制性实例包括RNA、mRNA、dsRNA、CRISPR/Cas9技术、ssDNA和DNA(例如,表达载体)。上调蛋白质表达的剂可以上调天然存在的或非天然存在的蛋白质(例如,已被修饰以提高半衰期的嵌合蛋白质,或包含希望的氨基酸变化的嵌合蛋白质)的表达。示例性蛋白质包括细胞内、跨膜或分泌的蛋白质、肽或多肽。In some embodiments, the therapeutic agent is an agent that enhances (ie, increases, stimulates, upregulates) protein expression. Non-limiting examples of the types of therapeutic agents that can be used to enhance protein expression include RNA, mRNA, dsRNA, CRISPR/Cas9 technology, ssDNA, and DNA (eg, expression vectors). Agents that upregulate protein expression can upregulate the expression of naturally occurring or non-naturally occurring proteins (eg, chimeric proteins that have been modified to increase half-life, or chimeric proteins that contain desired amino acid changes). Exemplary proteins include intracellular, transmembrane or secreted proteins, peptides or polypeptides.
在一些实施方案中,所述治疗剂是DNA治疗剂。DNA分子可以是双链DNA、单链DNA(ssDNA)或部分双链DNA的分子,即具有双链部分和单链部分的分子。在一些情况下,DNA分子是三链的或部分三链的,即具有三链部分和双链部分。DNA分子可以是环状DNA分子或线性DNA分子。In some embodiments, the therapeutic agent is a DNA therapeutic agent. A DNA molecule can be double-stranded DNA, single-stranded DNA (ssDNA) or a molecule of partially double-stranded DNA, ie a molecule having a double-stranded portion and a single-stranded portion. In some cases, the DNA molecule is triple-stranded or partially triple-stranded, ie, has a triple-stranded portion and a double-stranded portion. The DNA molecule can be a circular DNA molecule or a linear DNA molecule.
DNA治疗剂可以是能够将基因转移到细胞中的DNA分子,例如,编码并且可以表达转录物的DNA分子。在其他实施方案中,DNA分子可以是合成分子,例如,体外产生的合成DNA分子。在一些实施方案中,DNA分子是重组分子。非限制性示例性DNA治疗剂包括质粒表达载体和病毒表达载体。A DNA therapeutic can be a DNA molecule capable of transferring a gene into a cell, eg, a DNA molecule that encodes and can express a transcript. In other embodiments, the DNA molecule can be a synthetic molecule, eg, a synthetic DNA molecule produced in vitro. In some embodiments, the DNA molecule is a recombinant molecule. Non-limiting exemplary DNA therapeutics include plasmid expression vectors and viral expression vectors.
本文所述的DNA治疗剂,例如DNA载体,可以包括多个不同的特征结构。本文所述的DNA治疗剂,例如DNA载体,可以包括非编码DNA序列。例如,DNA序列可以包含至少一个基因调控元件,例如启动子、增强子、终止元件、聚腺苷酸化信号元件、剪接信号元件等。在一些实施方案中,非编码DNA序列是内含子。在一些实施方案中,非编码DNA序列是转座子。在一些实施方案中,本文所述的DNA序列可以具有可操作地连接到转录活性基因的非编码DNA序列。在其他实施方案中,本文所述的DNA序列可以具有不与基因连接的非编码DNA序列,即,非编码DNA不调控DNA序列上的基因。The DNA therapeutics described herein, eg, DNA vectors, can include a number of different features. DNA therapeutics, such as DNA vectors, described herein, can include non-coding DNA sequences. For example, a DNA sequence can comprise at least one gene regulatory element, such as a promoter, enhancer, termination element, polyadenylation signal element, splicing signal element, and the like. In some embodiments, the non-coding DNA sequence is an intron. In some embodiments, the non-coding DNA sequence is a transposon. In some embodiments, the DNA sequences described herein may have non-coding DNA sequences operably linked to a transcriptionally active gene. In other embodiments, the DNA sequences described herein may have non-coding DNA sequences that are not linked to genes, ie, the non-coding DNA does not regulate the genes on the DNA sequences.
在一些实施方案中,在本公开的负载LNP中,一种或多种治疗剂和/或预防剂是核酸。在一些实施方案中,一种或多种治疗剂和/或预防剂选自由核糖核酸(RNA)和脱氧核糖核酸(DNA)组成的组。In some embodiments, in the loaded LNPs of the present disclosure, the one or more therapeutic and/or prophylactic agents are nucleic acids. In some embodiments, the one or more therapeutic and/or prophylactic agents are selected from the group consisting of ribonucleic acid (RNA) and deoxyribonucleic acid (DNA).
例如,在一些实施方案中,当治疗剂和/或预防剂是DNA时,所述DNA选自由双链DNA、单链DNA(ssDNA)、部分双链DNA、三链DNA和部分三链DNA组成的组。在一些实施方案中,所述DNA选自由环状DNA、线性DNA及其混合物组成的组。For example, in some embodiments, when the therapeutic and/or prophylactic agent is DNA, the DNA is selected from the group consisting of double-stranded DNA, single-stranded DNA (ssDNA), partially double-stranded DNA, triple-stranded DNA, and partially triple-stranded DNA group. In some embodiments, the DNA is selected from the group consisting of circular DNA, linear DNA, and mixtures thereof.
在一些实施方案中,在本公开的负载LNP中,所述一种或多种治疗剂和/或预防剂选自由质粒表达载体、病毒表达载体及其混合物组成的组。In some embodiments, in the loaded LNPs of the present disclosure, the one or more therapeutic and/or prophylactic agents are selected from the group consisting of plasmid expression vectors, viral expression vectors, and mixtures thereof.
例如,在一些实施方案中,当治疗剂和/或预防剂是RNA时,所述RNA选自由单链RNA、双链RNA(dsRNA)、部分双链RNA及其混合物组成的组。在一些实施方案中,所述RNA选自由环状RNA、线性RNA及其混合物组成的组。For example, in some embodiments, when the therapeutic and/or prophylactic agent is RNA, the RNA is selected from the group consisting of single-stranded RNA, double-stranded RNA (dsRNA), partially double-stranded RNA, and mixtures thereof. In some embodiments, the RNA is selected from the group consisting of circular RNA, linear RNA, and mixtures thereof.
例如,在一些实施方案中,当治疗剂和/或预防剂是RNA时,RNA选自由以下组成的组:短干扰RNA(siRNA)、不对称干扰RNA(aiRNA)、RNA干扰(RNAi)分子、微RNA(miRNA)、antagomir、反义RNA、核酶、Dicer-底物RNA(dsRNA)、小发夹RNA(shRNA)、信使RNA(mRNA)、锁核酸(LNA)和CRISPR/Cas9技术及其混合物。For example, in some embodiments, when the therapeutic and/or prophylactic agent is RNA, the RNA is selected from the group consisting of short interfering RNA (siRNA), asymmetric interfering RNA (aiRNA), RNA interference (RNAi) molecule, MicroRNA (miRNA), antagomir, antisense RNA, ribozyme, Dicer-substrate RNA (dsRNA), small hairpin RNA (shRNA), messenger RNA (mRNA), locked nucleic acid (LNA) and CRISPR/Cas9 technologies and their mixture.
例如,在一些实施方案中,当治疗剂和/或预防剂是RNA时,RNA选自由以下组成的组:短干扰RNA(siRNA)、不对称干扰RNA(aiRNA)、微RNA(miRNA)、Dicer-底物RNA(dsRNA)、小发夹RNA(shRNA)、信使RNA(mRNA)及其混合物。For example, in some embodiments, when the therapeutic and/or prophylactic agent is RNA, the RNA is selected from the group consisting of short interfering RNA (siRNA), asymmetric interfering RNA (aiRNA), microRNA (miRNA), Dicer - Substrate RNA (dsRNA), small hairpin RNA (shRNA), messenger RNA (mRNA) and mixtures thereof.
在一些实施方案中,一种或多种治疗剂和/或预防剂是mRNA。在一些实施方案中,一种或多种治疗剂和/或预防剂是修饰的mRNA(mmRNA)。In some embodiments, the one or more therapeutic and/or prophylactic agents is mRNA. In some embodiments, the one or more therapeutic and/or prophylactic agents are modified mRNA (mmRNA).
在一些实施方案中,一种或多种治疗剂和/或预防剂是引入了微RNA结合位点(miR结合位点)的mRNA。此外,在一些实施方案中,mRNA包含茎环、链终止核苷、polyA序列、聚腺苷酸化信号和/或5'帽结构中的一个或多个。In some embodiments, the one or more therapeutic and/or prophylactic agents are mRNAs that incorporate a microRNA binding site (miR binding site). Furthermore, in some embodiments, the mRNA comprises one or more of a stem-loop, chain terminating nucleoside, polyA sequence, polyadenylation signal, and/or a 5' cap structure.
mRNA可以是天然或非天然存在的mRNA。如下所述,mRNA可以包含一个或多个修饰的核碱基、核苷或核苷酸,在这种情况下,它可以称为“修饰的mRNA”或“mmRNA”。如本文所述,“核苷”被定义为含有糖分子(例如,戊糖或核糖)或其衍生物与有机碱基(例如,嘌呤或嘧啶)或其衍生物(在本文中也称为“核碱基”)的组合的化合物。如本文所述,“核苷酸”被定义为包含磷酸基团的核苷。mRNA can be naturally or non-naturally occurring mRNA. As described below, mRNA may contain one or more modified nucleobases, nucleosides or nucleotides, in which case it may be referred to as "modified mRNA" or "mmRNA." As used herein, a "nucleoside" is defined as containing a sugar molecule (eg, pentose or ribose) or a derivative thereof and an organic base (eg, purine or pyrimidine) or a derivative thereof (also referred to herein as a "nucleoside" nucleobases"). As used herein, a "nucleotide" is defined as a nucleoside containing a phosphate group.
mRNA可以包含5'非翻译区(5'-UTR)、3'非翻译区(3'-UTR)和/或编码区(例如,开放阅读框)。mRNA可以包含任何合适数量的碱基对,包含数十个(例如,10、20、30、40、50、60、70、80、90或100)、数百个(例如,200、300、400、500、600、700、800或900)或数千个(例如,1000、2000、3000、4000、5000、6000、7000、8000、9000、10,000)碱基对。任何数量(例如,全部、一些或没有)的核碱基、核苷或核苷酸可以是取代的、修饰的或以其他方式非天然存在的典型物质的类似物。在某些实施方案中,可以修饰所有特定核碱基类型。在一些实施方案中,所有的尿嘧啶或尿苷都是修饰的。当所有的核碱基、核苷或核苷酸都是修饰的时,例如所有的尿嘧啶或尿苷,mRNA可以被称为例如对于尿嘧啶或尿苷而言“完全修饰的”。An mRNA can comprise a 5' untranslated region (5'-UTR), a 3' untranslated region (3'-UTR), and/or a coding region (eg, an open reading frame). mRNA can comprise any suitable number of base pairs, including tens (eg, 10, 20, 30, 40, 50, 60, 70, 80, 90, or 100), hundreds (eg, 200, 300, 400) , 500, 600, 700, 800, or 900) or thousands (eg, 1000, 2000, 3000, 4000, 5000, 6000, 7000, 8000, 9000, 10,000) base pairs. Any number (eg, all, some, or none) of nucleobases, nucleosides, or nucleotides may be substituted, modified, or otherwise non-naturally occurring analogs of typical species. In certain embodiments, all specific nucleobase types can be modified. In some embodiments, all uracils or uridines are modified. When all nucleobases, nucleosides, or nucleotides are modified, eg, all uracils or uridines, mRNA may be referred to as "fully modified," eg, for uracils or uridines.
在一些实施方案中,如本文所述的mRNA可包含5'帽结构、链终止核苷酸、任选的Kozak序列(也称为Kozak共有序列)、茎环、polyA序列和/或聚腺苷酸化信号。In some embodiments, an mRNA as described herein can comprise a 5' cap structure, chain termination nucleotides, optional Kozak sequences (also known as Kozak consensus sequences), stem loops, polyA sequences, and/or polyadenosine acidification signal.
5'帽结构或帽物质是包括通过接头接合的两个核苷部分的化合物,并且可以选自天然存在的帽、非天然存在的帽或帽类似物或抗反向帽类似物(ARCA)。帽物质可以包含一种或多种修饰核苷和/或接头部分。例如,天然mRNA帽可以包含在其5’位置通过三磷酸键联接合的鸟嘌呤核苷酸和在7位甲基化的鸟嘌呤(G)核苷酸,例如m7G(5')ppp(5')G,通常写成m7GpppG。帽物质也可以是抗反向帽类似物。可能的帽物质的非限制性列表包括m7GpppG、m7Gpppm7G、m73′dGpppG、m27,O3′GpppG、m27,O3′GppppG、m27,O2′GppppG、m7Gpppm7G、m73′dGpppG、m27,O3′GpppG、m27,O3′GppppG和m27,O2′GppppG。A 5' cap structure or cap species is a compound comprising two nucleoside moieties joined by a linker, and can be selected from a naturally occurring cap, a non-naturally occurring cap or cap analog, or an anti-reverse cap analog (ARCA). The cap species may comprise one or more modified nucleosides and/or linker moieties. For example, a native mRNA cap may comprise a guanine nucleotide joined by a triphosphate linkage at its 5' position and a guanine (G) nucleotide methylated at the 7 position, such as m7G(5')ppp(5 ')G, usually written as m7GpppG. The cap material can also be an anti-reverse cap analog. A non-limiting list of possible capping species includes m7GpppG, m7Gppm7G, m73'dGpppG, m27,O3'GpppG, m27,O3'GppppG, m27,O2'GppppG, m7Gpppm7G, m73'dGpppG, m27,O3'GpppG, m27, O3'GppppG and m27,O2'GppppG.
mRNA可以替代地或另外地包含链终止核苷。例如,链终止核苷可以包括那些在其糖基的2’和/或3′位脱氧的核苷。此类物质可包括3′脱氧腺苷(虫草素(cordycepin))、3′脱氧尿苷、3′脱氧胞嘧啶、3′脱氧鸟苷、3′脱氧胸腺嘧啶和2′,3′双脱氧核苷,诸如2′,3′双脱氧腺苷、2′,3′双脱氧尿苷、2′,3′双脱氧胞嘧啶、2′,3′双脱氧鸟苷和2′,3′双脱氧胸腺嘧啶。在一些实施方案中,将链终止核苷酸引入到mRNA中(例如在3’-末端)可导致mRNA的稳定化。The mRNA may alternatively or additionally contain chain terminating nucleosides. For example, chain terminating nucleosides may include those nucleosides deoxygenated at the 2' and/or 3' positions of their sugar groups. Such substances may include 3' deoxyadenosine (cordycepin), 3' deoxyuridine, 3' deoxycytosine, 3' deoxyguanosine, 3' deoxythymine and 2',3' dideoxy nuclei glycosides, such as 2',3' dideoxyadenosine, 2',3' dideoxyuridine, 2',3' dideoxycytosine, 2',3' dideoxyguanosine and 2',3' dideoxy Thymine. In some embodiments, introduction of chain terminating nucleotides into the mRNA (eg, at the 3'-end) can result in stabilization of the mRNA.
mRNA可以替代地或另外地包含茎环,诸如组蛋白茎环。茎环可以包含2、3、4、5、6、7、8或更多个核苷酸碱基对。例如,茎环可以包含4、5、6、7或8个核苷酸碱基对。茎环可以位于mRNA的任何区域中。例如,茎环可以位于非翻译区(5'非翻译区或3'非翻译区)、编码区或polyA序列或尾的内部、之前或之后。在一些实施方案中,茎环可以影响mRNA的一种或多种功能,例如翻译起始、翻译效率和/或转录终止。The mRNA may alternatively or additionally comprise a stem-loop, such as a histone stem-loop. The stem-loop can comprise 2, 3, 4, 5, 6, 7, 8 or more nucleotide base pairs. For example, a stem loop can comprise 4, 5, 6, 7 or 8 nucleotide base pairs. Stem-loops can be located in any region of the mRNA. For example, the stem-loop can be located within, before, or after an untranslated region (5' or 3' untranslated region), coding region, or polyA sequence or tail. In some embodiments, the stem-loop can affect one or more functions of the mRNA, such as translation initiation, translation efficiency, and/or transcription termination.
mRNA可以替代地或另外地包含polyA序列和/或聚腺苷酸化信号。polyA序列可以完全或大部分由腺嘌呤核苷酸或其类似物或衍生物组成。polyA序列还可以包含稳定化核苷酸或类似物。例如,polyA序列可以包含脱氧胸苷,例如反转(或反向键联)脱氧胸苷(dT),作为稳定化核苷酸或类似物。关于使用反转dT和其他稳定化polyA序列修饰的细节可以在例如WO2017/049275A2中找到,其内容以引用的方式并入本文。polyA序列可以是位于靠近mRNA的3'非翻译区的尾。在一些实施方案中,polyA序列可以影响mRNA的核输出、翻译和/或稳定性。The mRNA may alternatively or additionally comprise a polyA sequence and/or a polyadenylation signal. The polyA sequence may consist entirely or largely of adenine nucleotides or analogs or derivatives thereof. The polyA sequence may also contain stabilizing nucleotides or analogs. For example, the polyA sequence may contain deoxythymidine, eg, inverted (or reverse-linked) deoxythymidine (dT), as a stabilizing nucleotide or analog. Details on the use of inverted dT and other stabilizing polyA sequence modifications can be found, for example, in WO2017/049275A2, the contents of which are incorporated herein by reference. The polyA sequence may be located near the tail of the 3' untranslated region of the mRNA. In some embodiments, polyA sequences can affect nuclear export, translation and/or stability of mRNA.
mRNA可以替代地或另外地包含微RNA结合位点。微RNA结合位点(或miR结合位点)可用于调控各种组织或细胞类型中的mRNA表达。在示例性实施方案中,将miR结合位点工程化到mRNA的3'UTR序列中以调控例如增强表达同源miR的细胞或组织中的mRNA的降解。此类调控可用于调控或控制ir mRNA中的“脱靶”表达,即在体内不需要的细胞或组织中的表达。关于使用mir结合位点的细节可以在例如WO2017/062513A2中找到,其内容以引用的方式并入本文。The mRNA may alternatively or additionally contain a microRNA binding site. MicroRNA binding sites (or miR binding sites) can be used to regulate mRNA expression in various tissues or cell types. In an exemplary embodiment, a miR binding site is engineered into the 3'UTR sequence of the mRNA to modulate, eg, enhance the degradation of the mRNA in a cell or tissue expressing the cognate miR. Such modulation can be used to regulate or control "off-target" expression in ir mRNA, ie, expression in cells or tissues that are not desired in vivo. Details on the use of mir binding sites can be found, for example, in WO2017/062513A2, the contents of which are incorporated herein by reference.
在一些实施方案中,mRNA是包含第一编码区和第二编码区的双顺反子mRNA,其具有包含允许第一与第二编码区之间的内部翻译起始的内部核糖体进入位点(IRES)序列的间插序列,或具有编码自切割肽诸如2A肽的间插序列。IRES序列和2A肽通常用于增强来自同一载体的多种蛋白质的表达。多种IRES序列是本领域已知的和可获得的并且可以使用,包括例如脑心肌炎病毒IRES。In some embodiments, the mRNA is a bicistronic mRNA comprising a first coding region and a second coding region with an internal ribosome entry site comprising an internal ribosome entry site that allows for the initiation of internal translation between the first and second coding regions (IRES) sequences, or with intervening sequences encoding self-cleaving peptides such as the 2A peptide. IRES sequences and 2A peptides are often used to enhance the expression of multiple proteins from the same vector. Various IRES sequences are known and available in the art and can be used, including, for example, the encephalomyocarditis virus IRES.
在一些实施方案中,本公开的mRNA包含一个或多个修饰的核碱基、核苷或核苷酸(称为“修饰的mRNA”或“mmRNA”)。在一些实施方案中,与参考的未修饰的mRNA相比,修饰的mRNA可以具有有用的特性,包括增强的稳定性、细胞内保留、增强的翻译和/或缺乏对引入mRNA的细胞的先天免疫反应的实质性诱导。因此,使用修饰的mRNA可以提高蛋白质产生的效率、核酸的细胞内保留以及具有降低的免疫原性。In some embodiments, the mRNA of the present disclosure comprises one or more modified nucleobases, nucleosides or nucleotides (referred to as "modified mRNA" or "mmRNA"). In some embodiments, modified mRNAs may have useful properties compared to a reference unmodified mRNA, including enhanced stability, intracellular retention, enhanced translation, and/or lack of innate immunity to cells into which the mRNA was introduced Substantial induction of the response. Thus, the use of modified mRNA can increase the efficiency of protein production, intracellular retention of nucleic acids, and have reduced immunogenicity.
在一些实施方案中,mRNA包含一个或多个(例如,1、2、3或4个)不同的修饰的核碱基、核苷或核苷酸。在一些实施方案中,mRNA包含一个或多个(例如1、2、3、4、5、6、7、8、9、10、20、30、40、50、60、70、80、90、100个或更多个)不同的修饰的核碱基、核苷或核苷酸。在一些实施方案中,相对于对应的未修饰的mRNA,修饰的mRNA在引入所述mRNA的细胞中的降解可能减少。In some embodiments, the mRNA comprises one or more (eg, 1, 2, 3, or 4) different modified nucleobases, nucleosides, or nucleotides. In some embodiments, the mRNA comprises one or more (eg 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 20, 30, 40, 50, 60, 70, 80, 90, 100 or more) different modified nucleobases, nucleosides or nucleotides. In some embodiments, the degradation of a modified mRNA in a cell into which the mRNA is introduced may be reduced relative to the corresponding unmodified mRNA.
在一些实施方案中,所述修饰的核碱基是修饰的尿嘧啶。具有修饰的尿嘧啶的示例性核碱基和核苷包括假尿苷(ψ)、吡啶-4-酮核糖核苷、5-氮杂-尿苷、6-氮杂-尿苷、2-硫代-5-氮杂-尿苷、2-硫代-尿苷(s2U)、4-硫代-尿苷(s4U)、4-硫代-假尿苷、2-硫代-假尿苷、5-羟基-尿苷(ho5U)、5-氨基烯丙基-尿苷、5-卤代-尿苷(例如,5-碘-尿苷或5-溴-尿苷)、3-甲基-尿苷(m3U)、5-甲氧基-尿苷(mo5U)、尿苷5-氧基乙酸(cmo5U)、尿苷5-氧基乙酸甲酯(mcmo5U)、5-羧甲基-尿苷(cm5U)、1-羧甲基-假尿苷、5-羧基羟甲基-尿苷(chm5U)、5-羧基羟甲基-尿苷甲酯(mchm5U)、5-甲氧基羰基甲基-尿苷(mcm5U)、5-甲氧基羰基甲基-2-硫代-尿苷(mcm5s2U)、5-氨基甲基-2-硫代-尿苷(nm5s2U)、5-甲基氨基甲基-尿苷(mnm5U)、5-甲基氨基甲基-2-硫代-尿苷(mnm5s2U)、5-甲基氨基甲基-2-硒代-尿苷(mnm5se2U)、5-氨基甲酰基甲基-尿苷(ncm5U)、5-羧甲基氨基甲基-尿苷(cmnm5U)、5-羧甲基氨基甲基-2-硫代-尿苷(cmnm5s2U)、5-丙炔基-尿苷、1-丙炔基-假尿苷、5-牛磺酸甲基-尿苷(τm5U)、1-牛磺酸甲基-假尿苷、5-牛磺酸甲基-2-硫代-尿苷(τm5s2U)、1-牛磺酸甲基-4-硫代-假尿苷、5-甲基-尿苷(m5U,即具有核碱基脱氧胸腺嘧啶)、1-甲基-假尿苷(m1ψ)、5-甲基-2-硫代-尿苷(m5s2U)、1-甲基-4-硫代-假尿苷(m1s4ψ)、4-硫代-1-甲基-假尿苷、3-甲基-假尿苷(m3ψ)、2-硫代-1-甲基-假尿苷、1-甲基-1-脱氮-假尿苷、2-硫代-1-甲基-1-脱氮-假尿苷、二氢尿苷(D)、二氢假尿苷、5,6-二氢尿苷、5-甲基-二氢尿苷(m5D)、2-硫代-二氢尿苷、2-硫代-二氢假尿苷、2-甲氧基-尿苷、2-甲氧基-4-硫代-尿苷、4-甲氧基-假尿苷、4-甲氧基-2-硫代-假尿苷、N1-甲基-假尿苷、3-(3-氨基-3-羧丙基)尿苷(acp3U)、1-甲基-3-(3-氨基-3-羧丙基)假尿苷(acp3ψ)、5-(异戊烯基氨基甲基)尿苷(inm5U)、5-(异戊烯基氨基甲基)-2-硫代-尿苷(inm5s2U)、α-硫代-尿苷、2′-O-甲基-尿苷(Um)、5,2′-O-二甲基-尿苷(m5Um)、2′-O-甲基-假尿苷(ψm)、2-硫代-2′-O-甲基-尿苷(s2Um)、5-甲氧基羰基甲基-2′-O-甲基-尿苷(mcm5Um)、5-氨基甲酰基甲基-2′-O-甲基-尿苷(ncm5Um)、5-羧甲基氨基甲基-2′-O-甲基-尿苷(cmnm5Um)、3,2′-O-二甲基-尿苷(m3Um)和5-(异戊烯基氨基甲基)-2′-O-甲基-尿苷(inm5Um)、1-硫代-尿苷、脱氧胸苷、2’-F-阿糖-尿苷、2’-F-尿苷、2’-OH-阿糖-尿苷、5-(2-羧甲氧基乙烯基)尿苷和5-[3-(1-E-丙烯基氨基)]尿苷。In some embodiments, the modified nucleobase is a modified uracil. Exemplary nucleobases and nucleosides with modified uracil include pseudouridine (ψ), pyridin-4-ketoribonucleoside, 5-aza-uridine, 6-aza-uridine, 2-thio Sub-5-aza-uridine, 2-thio-uridine (s2U), 4-thio-uridine (s4U), 4-thio-pseudouridine, 2-thio-pseudouridine, 5-Hydroxy-uridine (ho5U), 5-aminoallyl-uridine, 5-halo-uridine (eg, 5-iodo-uridine or 5-bromo-uridine), 3-methyl- Uridine (m3U), 5-methoxy-uridine (mo5U), uridine 5-oxyacetic acid (cmo5U), uridine methyl 5-oxyacetate (mcmo5U), 5-carboxymethyl-uridine (cm5U), 1-carboxymethyl-pseudouridine, 5-carboxymethyl-uridine (chm5U), 5-carboxymethyl-uridine methyl ester (mchm5U), 5-methoxycarbonylmethyl - Uridine (mcm5U), 5-methoxycarbonylmethyl-2-thio-uridine (mcm5s2U), 5-aminomethyl-2-thio-uridine (nm5s2U), 5-methylaminomethyl yl-uridine (mnm5U), 5-methylaminomethyl-2-thio-uridine (mnm5s2U), 5-methylaminomethyl-2-seleno-uridine (mnm5se2U), 5-aminomethyl Acylmethyl-uridine (ncm5U), 5-carboxymethylaminomethyl-uridine (cmnm5U), 5-carboxymethylaminomethyl-2-thio-uridine (cmnm5s2U), 5-propynyl - Uridine, 1-propynyl-pseudouridine, 5-taurine methyl-uridine (τm5U), 1-taurine methyl-pseudouridine, 5-taurine methyl-2- Thio-uridine (τm5s2U), 1-taurine methyl-4-thio-pseudouridine, 5-methyl-uridine (m5U, i.e. with nucleobase deoxythymine), 1-methyl - Pseudouridine (m1ψ), 5-methyl-2-thio-uridine (m5s2U), 1-methyl-4-thio-pseudouridine (m1s4ψ), 4-thio-1-methyl - Pseudouridine, 3-methyl-pseudouridine (m3ψ), 2-thio-1-methyl-pseudouridine, 1-methyl-1-deaza-pseudouridine, 2-thio- 1-methyl-1-deaza-pseudouridine, dihydrouridine (D), dihydropseudouridine, 5,6-dihydrouridine, 5-methyl-dihydrouridine (m5D), 2-thio-dihydrouridine, 2-thio-dihydropseudouridine, 2-methoxy-uridine, 2-methoxy-4-thio-uridine, 4-methoxy- Pseudouridine, 4-methoxy-2-thio-pseudouridine, N1-methyl-pseudouridine, 3-(3-amino-3-carboxypropyl)uridine (acp3U), 1-methyl yl-3-(3-amino-3-carboxypropyl) pseudouridine (acp3ψ), 5-(prenylaminomethyl)uridine (inm5U), 5-(prenylaminomethyl) -2-thio-uridine (inm5s2U), α-thio-uridine, 2'-O-methyl-uridine (Um), 5,2'-O-dimethyl yl-uridine (m5Um), 2'-O-methyl-pseudouridine (ψm), 2-thio-2'-O-methyl-uridine (s2Um), 5-methoxycarbonylmethyl -2'-O-methyl-uridine (mcm5Um), 5-carbamoylmethyl-2'-O-methyl-uridine (ncm5Um), 5-carboxymethylaminomethyl-2'-O -Methyl-uridine (cmnm5Um), 3,2'-O-dimethyl-uridine (m3Um) and 5-(prenylaminomethyl)-2'-O-methyl-uridine ( inm5Um), 1-thio-uridine, deoxythymidine, 2'-F-arabino-uridine, 2'-F-uridine, 2'-OH-arabino-uridine, 5-(2- Carboxymethoxyvinyl)uridine and 5-[3-(1-E-propenylamino)]uridine.
在一些实施方案中,修饰的核碱基是修饰的胞嘧啶。具有修饰的胞嘧啶的示例性核碱基和核苷包括5-氮杂-胞苷、6-氮杂-胞苷、假异胞苷、3-甲基-胞苷(m3C)、N4-乙酰基-胞苷(ac4C)、5-甲酰基-胞苷(f5C)、N4-甲基-胞苷(m4C)、5-甲基-胞苷(m5C)、5-卤代-胞苷(例如,5-碘-胞苷)、5-羟甲基-胞苷(hm5C)、1-甲基-假异胞苷、吡咯并-胞苷、吡咯并-假异胞苷、2-硫代-胞苷(s2C)、2-硫代-5-甲基-胞苷、4-硫代-假异胞苷、4-硫代-1-甲基-假异胞苷、4-硫代-1-甲基-1-脱氮-假异胞苷、1-甲基-1-脱氮-假异胞苷、泽布拉林(zebularine)、5-氮杂-泽布拉林、5-甲基-泽布拉林、5-氮杂-2-硫代-泽布拉林、2-硫代-泽布拉林、2-甲氧基-胞苷、2-甲氧基-5-甲基-胞苷、4-甲氧基-假异胞苷、4-甲氧基-1-甲基-假异胞苷、赖胞苷(k2C)、α-硫代-胞苷、2′-O-甲基-胞苷(Cm)、5,2′-O-二甲基-胞苷(m5Cm)、N4-乙酰基-2′-O-甲基-胞苷(ac4Cm)、N4,2′-O-二甲基-胞苷(m4Cm)、5-甲酰基-2′-O-甲基-胞苷(f5Cm)、N4,N4,2′-O-三甲基-胞苷(m42Cm)、1-硫代-胞苷、2’-F-阿糖-胞苷、2’-F-胞苷和2’-OH-阿糖-胞苷。In some embodiments, the modified nucleobase is a modified cytosine. Exemplary nucleobases and nucleosides with modified cytosines include 5-aza-cytidine, 6-aza-cytidine, pseudoisocytidine, 3-methyl-cytidine (m3C), N4-acetyl Acyl-cytidine (ac4C), 5-formyl-cytidine (f5C), N4-methyl-cytidine (m4C), 5-methyl-cytidine (m5C), 5-halo-cytidine (e.g. , 5-iodo-cytidine), 5-hydroxymethyl-cytidine (hm5C), 1-methyl-pseudoisocytidine, pyrrolo-cytidine, pyrrolo-pseudoisocytidine, 2-thio- Cytidine (s2C), 2-thio-5-methyl-cytidine, 4-thio-pseudo-isocytidine, 4-thio-1-methyl-pseudo-isocytidine, 4-thio-1 -Methyl-1-deaza-pseudoisocytidine, 1-methyl-1-deaza-pseudoisocytidine, zebularine, 5-aza-zebraline, 5-methyl Base-Zebraline, 5-Aza-2-Thio-Zebraline, 2-Thio-Zebraline, 2-Methoxy-Cytidine, 2-Methoxy-5-Methyl Base-cytidine, 4-methoxy-pseudo-isocytidine, 4-methoxy-1-methyl-pseudo-isocytidine, lyscytidine (k2C), α-thio-cytidine, 2'- O-methyl-cytidine (Cm), 5,2'-O-dimethyl-cytidine (m5Cm), N4-acetyl-2'-O-methyl-cytidine (ac4Cm), N4,2 '-O-dimethyl-cytidine (m4Cm), 5-formyl-2'-O-methyl-cytidine (f5Cm), N4,N4,2'-O-trimethyl-cytidine (m42Cm ), 1-thio-cytidine, 2'-F-arabino-cytidine, 2'-F-cytidine, and 2'-OH-arabino-cytidine.
在一些实施方案中,修饰的核碱基是修饰的腺嘌呤。具有修饰的腺嘌呤的示例性核碱基和核苷包括a-硫代-腺苷、2-氨基-嘌呤、2,6-二氨基嘌呤、2-氨基-6-卤代-嘌呤(例如,2-氨基-6-氯-嘌呤)、6-卤代-嘌呤(例如,6-氯-嘌呤)、2-氨基-6-甲基-嘌呤、8-叠氮基-腺苷、7-脱氮-腺嘌呤、7-脱氮-8-氮杂-腺嘌呤、7-脱氮-2-氨基-嘌呤、7-脱氮-8-氮杂-2-氨基-嘌呤、7-脱氮-2,6-二氨基嘌呤、7-脱氮-8-氮杂-2,6-二氨基嘌呤、1-甲基-腺苷(m1A)、2-甲基-腺嘌呤(m2A)、N6-甲基-腺苷(m6A)、2-甲基硫代-N6-甲基-腺苷(ms2m6A)、N6-异戊烯基-腺苷(i6A)、2-甲基硫代-N6-异戊烯基-腺苷(ms2i6A)、N6-(顺式-羟基异戊烯基)腺苷(io6A)、2-甲基硫代-N6-(顺式-羟基异戊烯基)腺苷(ms2io6A)、N6-甘氨酰基氨基甲酰基-腺苷(g6A)、N6-苏氨酰基氨基甲酰基-腺苷(t6A)、N6-甲基-N6-苏氨酰基氨基甲酰基-腺苷(m6t6A)、2-甲基硫代-N6-苏氨酰基氨基甲酰基-腺苷(ms2g6A)、N6,N6-二甲基-腺苷(m62A)、N6-羟基正缬氨酰基氨基甲酰基-腺苷(hn6A)、2-甲基硫代-N6-羟基正缬氨酰基氨基甲酰基-腺苷(ms2hn6A)、N6-乙酰基-腺苷(ac6A)、7-甲基-腺嘌呤、2-甲基硫代-腺嘌呤、2-甲氧基-腺嘌呤、α-硫代-腺苷、2′-O-甲基-腺苷(Am)、N6,2′-O-二甲基-腺苷(m6Am)、N6,N6,2′-O-三甲基-腺苷(m62Am)、1,2′-O-二甲基-腺苷(m1Am)、2′-O-核糖基腺苷(磷酸)(Ar(p))、2-氨基-N6-甲基-嘌呤、1-硫代-腺苷、8-叠氮基-腺苷、2’-F-阿糖-腺苷、2’-F-腺苷、2’-OH-阿糖-腺苷和N6-(19-氨基-五氧杂十九烷基)-腺苷。In some embodiments, the modified nucleobase is a modified adenine. Exemplary nucleobases and nucleosides with modified adenines include α-thio-adenosine, 2-amino-purine, 2,6-diaminopurine, 2-amino-6-halo-purine (eg, 2-amino-6-chloro-purine), 6-halo-purine (e.g., 6-chloro-purine), 2-amino-6-methyl-purine, 8-azido-adenosine, 7-de nitrogen-adenine, 7-deaza-8-aza-adenine, 7-deaza-2-amino-purine, 7-deaza-8-aza-2-amino-purine, 7-deaza- 2,6-diaminopurine, 7-deaza-8-aza-2,6-diaminopurine, 1-methyl-adenosine (m1A), 2-methyl-adenine (m2A), N6- Methyl-adenosine (m6A), 2-methylthio-N6-methyl-adenosine (ms2m6A), N6-prenyl-adenosine (i6A), 2-methylthio-N6-iso Pentenyl-adenosine (ms2i6A), N6-(cis-hydroxyprenyl)adenosine (io6A), 2-methylthio-N6-(cis-hydroxyprenyl)adenosine ( ms2io6A), N6-glycylcarbamoyl-adenosine (g6A), N6-threonylcarbamoyl-adenosine (t6A), N6-methyl-N6-threonylcarbamoyl-adenosine ( m6t6A), 2-methylthio-N6-threonylcarbamoyl-adenosine (ms2g6A), N6,N6-dimethyl-adenosine (m62A), N6-hydroxynorvalylcarbamoyl- Adenosine (hn6A), 2-methylthio-N6-hydroxynorvaylcarbamoyl-adenosine (ms2hn6A), N6-acetyl-adenosine (ac6A), 7-methyl-adenine, 2 -Methylthio-adenine, 2-methoxy-adenine, α-thio-adenosine, 2'-O-methyl-adenosine (Am), N6,2'-O-dimethyl - Adenosine (m6Am), N6,N6,2'-O-trimethyl-adenosine (m62Am), 1,2'-O-dimethyl-adenosine (m1Am), 2'-O-ribosyl Adenosine (phosphate) (Ar(p)), 2-Amino-N6-methyl-purine, 1-thio-adenosine, 8-azido-adenosine, 2'-F-arabino-adenosine , 2'-F-adenosine, 2'-OH-arabino-adenosine and N6-(19-amino-pentoxanonadecyl)-adenosine.
在一些实施方案中,修饰的核碱基是修饰的鸟嘌呤。具有修饰的鸟嘌呤的示例性核碱基和核苷包括a-硫代-鸟苷、肌苷(I)、1-甲基-肌苷(m1I)、怀俄苷(imG)、甲基怀俄苷(mimG)、4-去甲基-怀俄苷(imG-14)、异怀俄苷(imG2)、怀丁苷(yW)、过氧怀丁苷(o2yW)、羟基怀丁苷(OhyW)、修饰不足的(undermodified)羟基怀丁苷(OhyW*)、7-脱氮-鸟苷、辫苷(Q)、环氧辫苷(oQ)、半乳糖基-辫苷(galQ)、甘露糖基-辫苷(manQ)、7-氰基-7-脱氮-鸟苷(preQ0)、7-氨基甲基-7-脱氮-鸟苷(preQ1)、古嘌苷(G+)、7-脱氮-8-氮杂-鸟苷、6-硫代-鸟苷、6-硫代-7-脱氮-鸟苷、6-硫代-7-脱氮-8-氮杂-鸟苷、7-甲基-鸟苷(m7G)、6-硫代-7-甲基-鸟苷、7-甲基-肌苷、6-甲氧基-鸟苷、1-甲基-鸟苷(m1G)、N2-甲基-鸟苷(m2G)、N2,N2-二甲基-鸟苷(m22G)、N2,7-二甲基-鸟苷(m2,7G)、N2,N2,7-二甲基-鸟苷(m2,2,7G)、8-氧代-鸟苷、7-甲基-8-氧代-鸟苷、1-甲基-6-硫代-鸟苷、N2-甲基-6-硫代-鸟苷、N2,N2-二甲基-6-硫代-鸟苷、α-硫代-鸟苷、2′-O-甲基-鸟苷(Gm)、N2-甲基-2′-O-甲基-鸟苷(m2Gm)、N2,N2-二甲基-2′-O-甲基-鸟苷(m22Gm)、1-甲基-2′-O-甲基-鸟苷(m1Gm)、N2,7-二甲基-2′-O-甲基-鸟苷(m2,7Gm)、2′-O-甲基-肌苷(Im)、1,2′-O-二甲基-肌苷(m1Im)、2′-O-核糖基鸟苷(磷酸)(Gr(p))、1-硫代-鸟苷、O6-甲基-鸟苷、2’-F-阿糖-鸟苷和2’-F-鸟苷。In some embodiments, the modified nucleobase is a modified guanine. Exemplary nucleobases and nucleosides with modified guanines include α-thio-guanosine, inosine (I), 1-methyl-inosine (m1I), wyosine (imG), methylphosphoryl Rugoside (mimG), 4-desmethyl-hyosine (imG-14), isohyosine (imG2), hydrin (yW), peroxyhydrin (o2yW), hydroxyhydrin ( OhyW), undermodified hydroxybutinoside (OhyW*), 7-deaza-guanosine, braidin (Q), epoxybraidin (oQ), galactosyl-braidin (galQ), Mannosyl-braidin (manQ), 7-cyano-7-deaza-guanosine (preQ0), 7-aminomethyl-7-deaza-guanosine (preQ1), gupurine (G+), 7-Deaza-8-aza-guanosine, 6-thio-guanosine, 6-thio-7-deaza-guanosine, 6-thio-7-deaza-8-aza-guanosine glycoside, 7-methyl-guanosine (m7G), 6-thio-7-methyl-guanosine, 7-methyl-inosine, 6-methoxy-guanosine, 1-methyl-guanosine (m1G), N2-methyl-guanosine (m2G), N2,N2-dimethyl-guanosine (m22G), N2,7-dimethyl-guanosine (m2,7G), N2,N2,7 -Dimethyl-guanosine (m2,2,7G), 8-oxo-guanosine, 7-methyl-8-oxo-guanosine, 1-methyl-6-thio-guanosine, N2 -Methyl-6-thio-guanosine, N2,N2-dimethyl-6-thio-guanosine, α-thio-guanosine, 2'-O-methyl-guanosine (Gm), N2-methyl-2'-O-methyl-guanosine (m2Gm), N2,N2-dimethyl-2'-O-methyl-guanosine (m22Gm), 1-methyl-2'-O -Methyl-guanosine (m1Gm), N2,7-dimethyl-2'-O-methyl-guanosine (m2,7Gm), 2'-O-methyl-inosine (Im), 1, 2'-O-Dimethyl-inosine (m1Im), 2'-O-ribosylguanosine (phosphate) (Gr(p)), 1-thio-guanosine, O6-methyl-guanosine, 2'-F-arabino-guanosine and 2'-F-guanosine.
在一些实施方案中,本公开的mRNA包含一种或多种前述修饰的核碱基的组合(例如,2、3或4种前述修饰的核碱基的组合)。In some embodiments, an mRNA of the present disclosure comprises a combination of one or more of the foregoing modified nucleobases (eg, a combination of 2, 3, or 4 of the foregoing modified nucleobases).
在一些实施方案中,修饰的核碱基是假尿苷(ψ)、N1-甲基假尿苷(m1ψ)、2-硫代尿苷、4’-硫代尿苷、5-甲基胞嘧啶、2-硫代-1-甲基-1-脱氮-假尿苷、2-硫代-1-甲基-假尿苷、2-硫代-5-氮杂-尿苷、2-硫代-二氢假尿苷、2-硫代-二氢尿苷、2-硫代-假尿苷、4-甲氧基-2-硫代-假尿苷、4-甲氧基-假尿苷、4-硫代-1-甲基-假尿苷、4-硫代-假尿苷、5-氮杂-尿苷、二氢假尿苷、5-甲氧基尿苷或2’-O-甲基尿苷。在一些实施方案中,本公开的mRNA包含一种或多种前述修饰的核碱基的组合(例如,2、3或4种前述修饰的核碱基的组合)。在一些实施方案中,修饰的核碱基是N1-甲基假尿苷(m1ψ),并且本公开的mRNA用N1-甲基假尿苷(m1ψ)完全修饰。在一些实施方案中,N1-甲基假尿苷(m1ψ)占mRNA中尿嘧啶的75%-100%。在一些实施方案中,N1-甲基假尿苷(m1ψ)占mRNA中尿嘧啶的100%。In some embodiments, the modified nucleobase is pseudouridine (ψ), N1-methylpseudouridine (m1ψ), 2-thiouridine, 4'-thiouridine, 5-methylcytidine Pyrimidine, 2-thio-1-methyl-1-deaza-pseudouridine, 2-thio-1-methyl-pseudouridine, 2-thio-5-aza-uridine, 2- Thio-dihydropseudouridine, 2-thio-dihydrouridine, 2-thio-pseudouridine, 4-methoxy-2-thio-pseudouridine, 4-methoxy-pseudouridine Uridine, 4-thio-1-methyl-pseudouridine, 4-thio-pseudouridine, 5-aza-uridine, dihydropseudouridine, 5-methoxyuridine or 2' -O-Methyluridine. In some embodiments, an mRNA of the present disclosure comprises a combination of one or more of the foregoing modified nucleobases (eg, a combination of 2, 3, or 4 of the foregoing modified nucleobases). In some embodiments, the modified nucleobase is N1-methylpseudouridine (m1ψ), and the mRNA of the present disclosure is fully modified with N1-methylpseudouridine (m1ψ). In some embodiments, N1-methylpseudouridine (m1ψ) accounts for 75%-100% of the uracil in the mRNA. In some embodiments, N1-methylpseudouridine (m1ψ) accounts for 100% of the uracil in the mRNA.
在一些实施方案中,修饰的核碱基是修饰的胞嘧啶。具有修饰的胞嘧啶的示例性核碱基和核苷包括N4-乙酰基-胞苷(ac4C)、5-甲基-胞苷(m5C)、5-卤代-胞苷(例如,5-碘-胞苷)、5-羟甲基-胞苷(hm5C)、1-甲基-假异胞苷、2-硫代-胞苷(s2C)、2-硫代-5-甲基-胞苷。在一些实施方案中,本公开的mRNA包含一种或多种前述修饰的核碱基的组合(例如,2、3或4种前述修饰的核碱基的组合)。In some embodiments, the modified nucleobase is a modified cytosine. Exemplary nucleobases and nucleosides with modified cytosines include N4-acetyl-cytidine (ac4C), 5-methyl-cytidine (m5C), 5-halo-cytidine (eg, 5-iodo-cytidine) - cytidine), 5-hydroxymethyl-cytidine (hm5C), 1-methyl-pseudoisocytidine, 2-thio-cytidine (s2C), 2-thio-5-methyl-cytidine . In some embodiments, an mRNA of the present disclosure comprises a combination of one or more of the foregoing modified nucleobases (eg, a combination of 2, 3, or 4 of the foregoing modified nucleobases).
在一些实施方案中,修饰的核碱基是修饰的腺嘌呤。具有修饰的腺嘌呤的示例性核碱基和核苷包括7-脱氮-腺嘌呤、1-甲基-腺苷(m1A)、2-甲基-腺嘌呤(m2A)、N6-甲基-腺苷(m6A)。在一些实施方案中,本公开的mRNA包含一种或多种前述修饰的核碱基的组合(例如,2、3或4种前述修饰的核碱基的组合)。In some embodiments, the modified nucleobase is a modified adenine. Exemplary nucleobases and nucleosides with modified adenines include 7-deaza-adenine, 1-methyl-adenosine (m1A), 2-methyl-adenine (m2A), N6-methyl- Adenosine (m6A). In some embodiments, an mRNA of the present disclosure comprises a combination of one or more of the foregoing modified nucleobases (eg, a combination of 2, 3, or 4 of the foregoing modified nucleobases).
在一些实施方案中,修饰的核碱基是修饰的鸟嘌呤。具有修饰的鸟嘌呤的示例性核碱基和核苷包括肌苷(I)、1-甲基-肌苷(m1I)、怀俄苷(imG)、甲基怀俄苷(mimG)、7-脱氮-鸟苷、7-氰基-7-脱氮-鸟苷(preQ0)、7-氨基甲基-7-脱氮-鸟苷(preQ1)、7-甲基-鸟苷(m7G)、1-甲基-鸟苷(m1G)、8-氧代-鸟苷、7-甲基-8-氧代-鸟苷。在一些实施方案中,本公开的mRNA包含一种或多种前述修饰的核碱基的组合(例如,2、3或4种前述修饰的核碱基的组合)。In some embodiments, the modified nucleobase is a modified guanine. Exemplary nucleobases and nucleosides with modified guanines include inosine (I), 1-methyl-inosine (m1I), hyosine (imG), methyl hyosine (mimG), 7- Deaza-guanosine, 7-cyano-7-deaza-guanosine (preQ0), 7-aminomethyl-7-deaza-guanosine (preQ1), 7-methyl-guanosine (m7G), 1-Methyl-guanosine (m1G), 8-oxo-guanosine, 7-methyl-8-oxo-guanosine. In some embodiments, an mRNA of the present disclosure comprises a combination of one or more of the foregoing modified nucleobases (eg, a combination of 2, 3, or 4 of the foregoing modified nucleobases).
在一些实施方案中,修饰的核碱基是1-甲基-假尿苷(m1ψ)、5-甲氧基-尿苷(mo5U)、5-甲基-胞苷(m5C)、假尿苷(ψ)、α-硫代-鸟苷或α-硫代-腺苷。在一些实施方案中,本公开的mRNA包含一种或多种前述修饰的核碱基的组合(例如,2、3或4种前述修饰的核碱基的组合)。In some embodiments, the modified nucleobase is 1-methyl-pseudouridine (m1ψ), 5-methoxy-uridine (mo5U), 5-methyl-cytidine (m5C), pseudouridine (ψ), α-thio-guanosine or α-thio-adenosine. In some embodiments, an mRNA of the present disclosure comprises a combination of one or more of the foregoing modified nucleobases (eg, a combination of 2, 3, or 4 of the foregoing modified nucleobases).
在一些实施方案中,mRNA包含假尿苷(ψ)。在一些实施方案中,mRNA包含假尿苷(ψ)和5-甲基-胞苷(m5C)。在一些实施方案中,mRNA包含1-甲基-假尿苷(m1ψ)。在一些实施方案中,mRNA包含1-甲基-假尿苷(m1ψ)和5-甲基-胞苷(m5C)。在一些实施方案中,mRNA包含2-硫代尿苷(s2U)。在一些实施方案中,mRNA包含2-硫代尿苷和5-甲基-胞苷(m5C)。在一些实施方案中,mRNA包含5-甲氧基-尿苷(mo5U)。在一些实施方案中,mRNA包含5-甲氧基-尿苷(mo5U)和5-甲基-胞苷(m5C)。在一些实施方案中,mRNA包含2’-O-甲基尿苷。在一些实施方案中,mRNA包括2'-O-甲基-尿苷和5-甲基-胞嘧啶(m5C)。在一些实施方案中,mRNA包含N6-甲基-腺苷(m6A)。在一些实施方案中,mRNA包含N6-甲基-腺苷(m6A)和5-甲基-胞苷(m5C)。In some embodiments, the mRNA comprises pseudouridine (ψ). In some embodiments, the mRNA comprises pseudouridine (ψ) and 5-methyl-cytidine (m5C). In some embodiments, the mRNA comprises 1-methyl-pseudouridine (m1ψ). In some embodiments, the mRNA comprises 1-methyl-pseudouridine (m1ψ) and 5-methyl-cytidine (m5C). In some embodiments, the mRNA comprises 2-thiouridine (s2U). In some embodiments, the mRNA comprises 2-thiouridine and 5-methyl-cytidine (m5C). In some embodiments, the mRNA comprises 5-methoxy-uridine (mo5U). In some embodiments, the mRNA comprises 5-methoxy-uridine (mo5U) and 5-methyl-cytidine (m5C). In some embodiments, the mRNA comprises 2'-O-methyluridine. In some embodiments, the mRNA includes 2'-O-methyl-uridine and 5-methyl-cytosine (m5C). In some embodiments, the mRNA comprises N6-methyl-adenosine (m6A). In some embodiments, the mRNA comprises N6-methyl-adenosine (m6A) and 5-methyl-cytidine (m5C).
在某些实施方案中,本公开的mRNA针对特定修饰被一致地修饰(即,完全修饰、贯穿整个序列修饰)。例如,mRNA可以用N1-甲基假尿苷(m1ψ)或5-甲基-胞苷(m5C)一致地修饰,意味着mRNA序列中的所有尿苷或所有胞嘧啶核苷均被N1-甲基假尿苷(m1ψ)或5-甲基-胞苷(m5C)替代。类似地,对于序列中存在的任何类型的核苷残基,本公开的mRNA可以通过用修饰的残基(如以上列出的那些残基)替代来一致地修饰。In certain embodiments, the mRNAs of the present disclosure are uniformly modified for a particular modification (ie, fully modified, modified throughout the entire sequence). For example, mRNA can be uniformly modified with N1-methylpseudouridine (m1ψ) or 5-methyl-cytidine (m5C), meaning that all uridines or all cytosine nucleosides in the mRNA sequence are modified by N1-methyl Substituted pseudouridine (m1ψ) or 5-methyl-cytidine (m5C). Similarly, for any type of nucleoside residues present in the sequence, the mRNAs of the present disclosure can be consistently modified by substitution with modified residues, such as those listed above.
在一些实施方案中,本公开的mRNA可以在编码区(例如,编码多肽的开放阅读框)中被修饰。在其他实施方案中,mRNA可以在编码区以外的区域中被修饰。例如,在一些实施方案中,提供了5′-UTR和/或3′-UTR,其中任一者或两者可独立地含有一个或多个不同的核苷修饰。在此类实施方案中,核苷修饰还可存在于可编码区中。In some embodiments, an mRNA of the present disclosure may be modified in a coding region (eg, an open reading frame encoding a polypeptide). In other embodiments, the mRNA may be modified in regions other than the coding region. For example, in some embodiments, 5'-UTR and/or 3'-UTR are provided, either or both of which may independently contain one or more different nucleoside modifications. In such embodiments, nucleoside modifications may also be present in the codeable region.
本公开的mmRNA可包括对糖、核碱基和/或核苷间键联的修饰的组合。这些组合可包括本文所述的任一个或多个修饰。The mmRNA of the present disclosure can include a combination of modifications to sugars, nucleobases, and/or internucleoside linkages. These combinations may include any one or more of the modifications described herein.
在列出单个修饰的情况下,列出的核苷或核苷酸代表100%的A、U、G或C核苷酸或核苷已被修饰。在列出百分比的情况下,这些表示特定的A、U、G或C三磷酸核碱基在存在的A、U、G或C三磷酸总量中的百分比。例如,组合:25%5-氨基烯丙基-CTP+75%CTP/25%5-甲氧基-UTP+75%UTP是指以下的多核苷酸,其中25%的三磷酸胞嘧啶是5-氨基烯丙基-CTP,同时75%的胞嘧啶是CTP;而25%的尿嘧啶是5-甲氧基UTP,同时75%的尿嘧啶是UTP。在没有列出修饰的UTP的情况下,则在多核苷酸中存在的那些核苷酸的100%位点处使用天然存在的ATP、UTP、GTP和/或CTP。在此实例中,所有的GTP和ATP核苷酸都没有被修饰。Where individual modifications are listed, the nucleosides or nucleotides listed represent 100% of the A, U, G or C nucleotides or nucleosides that have been modified. Where percentages are listed, these represent the percentage of a particular A, U, G or C triphosphate nucleobase in the total amount of A, U, G or C triphosphates present. For example, the combination: 25% 5-aminoallyl-CTP + 75% CTP/25% 5-methoxy-UTP + 75% UTP refers to the following polynucleotides, where 25% of cytosine triphosphates are 5 -Aminoallyl-CTP, while 75% of cytosines are CTP; and 25% of uracils are 5-methoxy UTP, while 75% of uracils are UTP. Where no modified UTP is listed, naturally occurring ATP, UTP, GTP and/or CTP are used at 100% of the sites of those nucleotides present in the polynucleotide. In this example, all GTP and ATP nucleotides were unmodified.
本公开的mRNA或其区域可以是密码子优化的。密码子优化方法是本领域已知的并且可用于多种目的:匹配宿主生物体中的密码子频率以确保正确折叠;偏置GC含量以增加mRNA稳定性或减少二级结构;最小化可能损害基因构建或表达的串联重复密码子或碱基串(base run),定制转录和翻译控制区域,插入或去除蛋白质运输序列,在编码的蛋白质中去除/添加翻译后修饰位点(例如糖基化位点),添加、去除或改组蛋白质结构域,插入或缺失限制性位点,修饰核糖体结合位点和mRNA降解位点,调整翻译速率以允许蛋白质的不同结构域正确地折叠,或者减少或消除多核苷酸内的问题二级结构。密码子优化工具、算法和服务是本领域中已知的;非限制性实例包括GeneArt(Life Technologies)、DNA2.0(MenloPark,CA)和/或专有方法的服务。在一些实施方案中,使用优化算法优化mRNA序列,例如以优化哺乳动物细胞中的表达或增强mRNA稳定性。The mRNAs or regions thereof of the present disclosure may be codon-optimized. Codon optimization methods are known in the art and can be used for a variety of purposes: match codon frequencies in the host organism to ensure proper folding; bias GC content to increase mRNA stability or reduce secondary structure; minimize possible damage Tandem repeats of codons or base runs for gene construction or expression, custom transcriptional and translational control regions, insertion or removal of protein trafficking sequences, removal/addition of post-translational modification sites (e.g. glycosylation) in encoded proteins sites), add, remove or shuffle protein domains, insert or delete restriction sites, modify ribosome binding sites and mRNA degradation sites, adjust translation rates to allow different domains of the protein to fold correctly, or reduce or Eliminate problematic secondary structures within polynucleotides. Codon optimization tools, algorithms and services are known in the art; non-limiting examples include services of GeneArt (Life Technologies), DNA 2.0 (MenloPark, CA) and/or proprietary methods. In some embodiments, an optimization algorithm is used to optimize the mRNA sequence, eg, to optimize expression in mammalian cells or to enhance mRNA stability.
在某些实施方案中,本公开包括与本文所述的多核苷酸序列中的任一个具有至少80%、至少85%、至少90%、至少95%、至少98%或至少99%序列同一性的多核苷酸。In certain embodiments, the present disclosure includes at least 80%, at least 85%, at least 90%, at least 95%, at least 98%, or at least 99% sequence identity to any of the polynucleotide sequences described herein of polynucleotides.
本公开的mRNA可以通过本领域中可用的方法产生,包括但不限于体外转录(IVT)和合成方法。可以使用酶促(IVT)、固相、液相、组合合成方法、小区域合成和连接方法。在一些实施方案中,mRNA是使用IVT酶促合成方法制备的。因此,本公开还包括可用于体外转录本文所述的mRNA的多核苷酸,例如DNA、构建体和载体。The mRNAs of the present disclosure can be produced by methods available in the art, including but not limited to in vitro transcription (IVT) and synthetic methods. Enzymatic (IVT), solid phase, liquid phase, combinatorial synthesis methods, small area synthesis and ligation methods can be used. In some embodiments, the mRNA is prepared using an IVT enzymatic synthesis method. Accordingly, the present disclosure also includes polynucleotides, such as DNA, constructs and vectors, useful for in vitro transcription of the mRNAs described herein.
可以在合成期间或合成后将非天然修饰的核碱基引入多核苷酸,例如mRNA。在一些实施方案中,修饰可以在核苷间键联、嘌呤或嘧啶碱基或者糖上。在特定实施方案中,修饰可在多核苷酸链的末端或多核苷酸链中的任何其他位置引入;使用化学合成或使用聚合酶。Non-naturally modified nucleobases can be introduced into polynucleotides, such as mRNA, during or after synthesis. In some embodiments, modifications may be on internucleoside linkages, purine or pyrimidine bases, or sugars. In certain embodiments, modifications can be introduced at the end of the polynucleotide chain or at any other position in the polynucleotide chain; using chemical synthesis or using polymerases.
酶促或化学连接方法可用于使多核苷酸或其区域与不同的功能部分(诸如靶向或递送剂、荧光标记、液体、纳米粒子等)缀合。用于降低蛋白质表达的治疗剂Enzymatic or chemical ligation methods can be used to conjugate polynucleotides or regions thereof to different functional moieties such as targeting or delivery agents, fluorescent labels, liquids, nanoparticles, etc. Therapeutic agents for reducing protein expression
在一些实施方案中,治疗剂是降低(即,减少、抑制、下调)蛋白质表达的治疗剂。可用于降低蛋白质表达的治疗剂类型的非限制性实例包括引入微RNA结合位点(miR结合位点)的mRNA、微RNA(miRNA)、antagomir、小(短)干扰RNA(siRNA)(包括shortmer和dicer-底物RNA)、RNA干扰(RNAi)分子、反义RNA、核酶、小发夹RNA(shRNA)、锁核酸(LNA)和CRISPR/Cas9技术。In some embodiments, the therapeutic agent is one that reduces (ie, reduces, inhibits, downregulates) protein expression. Non-limiting examples of the types of therapeutics that can be used to reduce protein expression include mRNA, microRNA (miRNA), antagomir, small (short) interfering RNA (siRNA) (including shortmers) that incorporate a microRNA binding site (miR binding site). and dicer-substrate RNA), RNA interference (RNAi) molecules, antisense RNA, ribozymes, small hairpin RNA (shRNA), locked nucleic acid (LNA) and CRISPR/Cas9 technologies.
肽/多肽治疗剂Peptide/Peptide Therapeutics
在一些实施方案中,所述治疗剂是肽治疗剂。在一些实施方案中,所述治疗剂是多肽治疗剂。In some embodiments, the therapeutic agent is a peptide therapeutic. In some embodiments, the therapeutic agent is a polypeptide therapeutic agent.
在一些实施方案中,肽或多肽是天然衍生的,例如从天然来源分离的。在其他实施方案中,肽或多肽是合成分子,例如体外产生的合成肽或多肽。在一些实施方案中,肽或多肽是重组分子。在一些实施方案中,肽或多肽是嵌合分子。在一些实施方案中,肽或多肽是融合分子。在一些实施方案中,组合物的肽或多肽治疗剂是天然存在的肽或多肽。在一些实施方案中,组合物的肽或多肽治疗剂是天然存在的肽或多肽的修饰型式(例如,与其野生型、天然存在的肽或多肽对应物相比,含有少于3个、少于5个、少于10个、少于15个、少于20个或少于25个氨基酸取代、缺失或添加)。In some embodiments, the peptide or polypeptide is naturally derived, eg, isolated from a natural source. In other embodiments, the peptide or polypeptide is a synthetic molecule, eg, a synthetic peptide or polypeptide produced in vitro. In some embodiments, the peptide or polypeptide is a recombinant molecule. In some embodiments, the peptide or polypeptide is a chimeric molecule. In some embodiments, the peptide or polypeptide is a fusion molecule. In some embodiments, the peptide or polypeptide therapeutic agent of the composition is a naturally occurring peptide or polypeptide. In some embodiments, the peptide or polypeptide therapeutic of the composition is a modified version of a naturally occurring peptide or polypeptide (eg, contains less than 3, less than 5, less than 10, less than 15, less than 20 or less than 25 amino acid substitutions, deletions or additions).
在一些实施方案中,在本公开的负载LNP中,一种或多种治疗剂和/或预防剂是多核苷酸或多肽。In some embodiments, in the loaded LNPs of the present disclosure, the one or more therapeutic and/or prophylactic agents are polynucleotides or polypeptides.
其他组分other components
脂质纳米粒子(例如,空LNP或负载LNP)可以包含一种或多种除了前述部分中描述的那些之外的组分。例如,脂质纳米粒子(例如,空LNP或负载LNP)可以包含一种或多种疏水性小分子,诸如维生素(例如,维生素A或维生素E)或固醇。Lipid nanoparticles (eg, empty LNPs or loaded LNPs) may contain one or more components other than those described in the preceding sections. For example, lipid nanoparticles (eg, empty LNPs or loaded LNPs) can comprise one or more small hydrophobic molecules, such as vitamins (eg, vitamin A or vitamin E) or sterols.
脂质纳米粒子(例如,空LNP或负载LNP)还可以包含一种或多种渗透性增强分子、碳水化合物、聚合物、表面改变剂或其他组分。碳水化合物可以包括单糖(例如,葡萄糖)和多糖(例如,糖原及其衍生物和类似物)。Lipid nanoparticles (eg, empty LNPs or loaded LNPs) may also contain one or more permeability enhancing molecules, carbohydrates, polymers, surface altering agents, or other components. Carbohydrates can include monosaccharides (eg, glucose) and polysaccharides (eg, glycogen and its derivatives and analogs).
聚合物可以被包含在纳米粒子组合物中和/或用于囊封或部分囊封纳米粒子组合物。聚合物可以是可生物降解和/或生物相容的。聚合物可以选自但不限于聚胺、聚醚、聚酰胺、聚酯、聚氨基甲酸酯、聚脲、聚碳酸酯、聚苯乙烯、聚酰亚胺、聚砜、聚氨酯、聚乙炔、聚乙烯、聚乙烯亚胺、聚异氰酸酯、聚丙烯酸酯、聚甲基丙烯酸酯、聚丙烯腈和聚芳香酯。例如,聚合物可以包括聚(己内酯)(PCL)、乙烯乙酸乙烯酯聚合物(EVA)、聚(乳酸)(PLA)、聚(L-乳酸)(PLLA)、聚(乙醇酸)(PGA)、聚(乳酸-共-乙醇酸)(PLGA)、聚(L-乳酸-共-乙醇酸)(PLLGA)、聚(D,L-丙交酯)(PDLA)、聚(L-丙交酯)(PLLA)、聚(D,L-丙交酯-共-己内酯)、聚(D,L-丙交酯-共-己内酯-共-乙交酯)、聚(D,L-丙交酯-共-PEO-共-D,L-丙交酯)、聚(D,L-丙交酯-共-PPO-共-D,L-丙交酯)、聚氰基丙烯酸烷酯、聚氨酯、聚L-赖氨酸(PLL)、甲基丙烯酸羟基丙酯(HPMA)、聚乙二醇、聚L-谷氨酸、聚(羟基酸)、聚酐、聚原酸酯、聚(酯酰胺)、聚酰胺、聚(酯醚)、聚碳酸酯、聚烯烃诸如聚乙烯和聚丙烯、聚烷二醇诸如聚(乙二醇)(PEG)、聚环氧烷(PEO)、聚对苯二甲酸烷二酯诸如聚(对苯二甲酸乙二酯)、聚乙烯醇(PVA)、聚乙烯醚、聚乙烯酯诸如聚(乙酸乙烯酯)、聚卤化乙烯诸如聚(氯化乙烯)(PVC)、聚乙烯吡咯烷酮(PVP)、聚硅氧烷、聚苯乙烯(PS)、聚氨酯、衍生化纤维素诸如烷基纤维素、羟烷基纤维素、纤维素醚、纤维素酯、硝基纤维素、羟丙基纤维素、羧甲基纤维素、丙烯酸的聚合物诸如聚((甲基)丙烯酸甲酯)(PMMA)、聚((甲基)丙烯酸乙酯)、聚((甲基)丙烯酸丁酯)、聚((甲基)丙烯酸异丁酯)、聚((甲基)丙烯酸己酯)、聚((甲基)丙烯酸异癸酯)、聚((甲基)丙烯酸月桂酯)、聚((甲基)丙烯酸苯酯)、聚(丙烯酸甲酯)、聚(丙烯酸异丙酯)、聚(丙烯酸异丁酯)、聚(丙烯酸十八烷酯)及其共聚物和混合物、聚二噁烷酮和其共聚物、聚羟基烷酸酯、聚富马酸丙二醇酯、聚甲醛、泊洛沙姆(poloxamer)、聚氧胺、聚(原酸)酯、聚(丁酸)、聚(戊酸)、聚(丙交酯-共-己内酯)、碳酸三亚甲酯、聚(N-丙烯酰基吗啉)(PAcM)、聚(2-甲基-2-噁唑啉)(PMOX)、聚(2-乙基-2-噁唑啉)(PEOZ)以及聚甘油。The polymer can be included in and/or used to encapsulate or partially encapsulate the nanoparticle composition. The polymers can be biodegradable and/or biocompatible. The polymer may be selected from, but is not limited to, polyamines, polyethers, polyamides, polyesters, polyurethanes, polyureas, polycarbonates, polystyrenes, polyimides, polysulfones, polyurethanes, polyacetylenes, Polyethylene, polyethyleneimine, polyisocyanates, polyacrylates, polymethacrylates, polyacrylonitrile and polyarylates. For example, the polymer may include poly(caprolactone) (PCL), ethylene vinyl acetate polymer (EVA), poly(lactic acid) (PLA), poly(L-lactic acid) (PLLA), poly(glycolic acid) ( PGA), poly(lactic-co-glycolic acid) (PLGA), poly(L-lactic-co-glycolic acid) (PLLGA), poly(D,L-lactide) (PDLA), poly(L-propylene) lactide) (PLLA), poly(D,L-lactide-co-caprolactone), poly(D,L-lactide-co-caprolactone-co-glycolide), poly(D, L-lactide-co-caprolactone-co-glycolide) , L-lactide-co-PEO-co-D,L-lactide), poly(D,L-lactide-co-PPO-co-D,L-lactide), polycyano Alkyl Acrylate, Polyurethane, Poly-L-Lysine (PLL), Hydroxypropyl Methacrylate (HPMA), Polyethylene Glycol, Poly-L-Glutamic Acid, Poly(hydroxyacid), Polyanhydride, Polyortholic Acid esters, poly(esteramides), polyamides, poly(ester ethers), polycarbonates, polyolefins such as polyethylene and polypropylene, polyalkylene glycols such as poly(ethylene glycol) (PEG), polyalkylene oxides ( PEO), polyethylene terephthalates such as poly(ethylene terephthalate), polyvinyl alcohol (PVA), polyvinyl ethers, polyvinyl esters such as poly(vinyl acetate), polyvinyl halides such as poly(vinyl acetate) (vinyl chloride) (PVC), polyvinylpyrrolidone (PVP), polysiloxane, polystyrene (PS), polyurethane, derivatized cellulose such as alkyl cellulose, hydroxyalkyl cellulose, cellulose ethers, Cellulose esters, nitrocellulose, hydroxypropyl cellulose, carboxymethyl cellulose, polymers of acrylic acid such as poly((meth)methylacrylate) (PMMA), poly(ethyl(meth)acrylate) , poly(butyl(meth)acrylate), poly(isobutyl(meth)acrylate), poly(hexyl(meth)acrylate), poly(isodecyl(meth)acrylate), poly(((meth)acrylate) lauryl meth)acrylate), poly(phenyl(meth)acrylate), poly(methyl acrylate), poly(isopropyl acrylate), poly(isobutyl acrylate), poly(octadecyl acrylate) and its copolymers and mixtures, polydioxanone and its copolymers, polyhydroxyalkanoates, polytrimethylene fumarate, polyoxymethylene, poloxamer, polyoxyamine, poly(ortho) Esters, poly(butyric acid), poly(valeric acid), poly(lactide-co-caprolactone), trimethylene carbonate, poly(N-acryloylmorpholine) (PAcM), poly(2-methyl) oxazoline) (PMOX), poly(2-ethyl-2-oxazoline) (PEOZ), and polyglycerol.
表面改变剂可以包括但不限于阴离子性蛋白质(例如牛血清白蛋白)、表面活性剂(例如阳离子表面活性剂,诸如二甲基二-十八烷基-溴化铵)、糖或糖衍生物(例如环糊精)、核酸、聚合物(例如肝素、聚乙二醇和泊洛沙姆)、粘液溶解剂(例如乙酰半胱氨酸、艾蒿、菠萝蛋白酶(bromelain)、木瓜蛋白酶、大青属(clerodendrum)、溴己新(bromhexine)、羧甲司坦(carbocisteine)、依普拉酮(eprazinone)、美司钠(mesna)、氨溴索(ambroxol)、索布瑞醇(sobrerol)、多米奥醇(domiodol)、来托司坦(letosteine)、司替罗宁(stepronin)、硫普罗宁(tiopronin)、凝溶胶蛋白(gelsolin)、胸腺素(thymosin)β4、α链道酶(dornasealfa)、奈替克新(neltenexine)和厄多司坦(erdosteine))和DNA酶(例如rhDNA酶)。表面改变剂可以被设置在纳米粒子内和/或脂质纳米粒子(例如,空LNP或负载LNP)的表面上(例如,通过涂覆、吸附、共价连接或其他过程)。Surface-altering agents may include, but are not limited to, anionic proteins (eg, bovine serum albumin), surfactants (eg, cationic surfactants such as dimethyldi-octadecyl-ammonium bromide), sugars, or sugar derivatives (eg cyclodextrins), nucleic acids, polymers (eg heparin, polyethylene glycol and poloxamers), mucolytics (eg acetylcysteine, mugwort, bromelain, papain, Daqing Clerodendrum, bromhexine, carbocisteine, epraxinone, mesna, ambroxol, sobrerol, Domiodol, letosteine, stepronin, tiopronin, gelsolin, thymosin beta4, dornasealfa ), neltenexine and erdosteine) and DNases (eg rhDNase). Surface-altering agents can be disposed within the nanoparticles and/or on the surface of lipid nanoparticles (eg, empty LNPs or loaded LNPs) (eg, by coating, adsorption, covalent attachment, or other processes).
脂质纳米粒子(例如,空LNP或负载LNP)还可以包含一种或多种官能化脂质。例如,脂质可以用炔基官能化,所述炔基当在适当的反应条件下暴露于叠氮化物时可能经历环加成反应。特别地,脂质双层可以按这种方式用一种或多种可用于促进膜渗透、细胞识别或成像的基团官能化。脂质纳米粒子(例如,空LNP或负载LNP)的表面也可以与一种或多种有用的抗体缀合。可用于靶向的细胞递送、成像和膜渗透的官能团和缀合物是本领域中众所周知的。Lipid nanoparticles (eg, empty LNPs or loaded LNPs) can also comprise one or more functionalized lipids. For example, lipids can be functionalized with alkynyl groups that may undergo cycloaddition reactions when exposed to azides under appropriate reaction conditions. In particular, lipid bilayers can be functionalized in this manner with one or more groups that can be used to facilitate membrane penetration, cell recognition or imaging. The surface of lipid nanoparticles (eg, empty LNPs or loaded LNPs) can also be conjugated to one or more useful antibodies. Functional groups and conjugates useful for targeted cellular delivery, imaging and membrane penetration are well known in the art.
除了这些组分之外,脂质纳米粒子(例如,空LNP或负载LNP)可以包含可用于药物组合物的任何物质。例如,脂质纳米粒子(例如,空LNP或负载LNP)可以包含一种或多种药学上可接受的赋形剂或辅助成分,诸如但不限于一种或多种溶剂、分散介质、稀释剂、分散助剂、悬浮助剂、造粒助剂、崩解剂、填充剂、助流剂、液体媒介物、粘结剂、表面活性剂、等渗剂、增稠剂或乳化剂、缓冲剂、润滑剂、油、防腐剂和其他物质。还可以包含赋形剂,诸如蜡、黄油、着色剂、包衣剂、调味剂和芳香剂。In addition to these components, lipid nanoparticles (eg, empty LNPs or loaded LNPs) can contain any substance useful in pharmaceutical compositions. For example, lipid nanoparticles (eg, empty LNPs or loaded LNPs) can comprise one or more pharmaceutically acceptable excipients or adjunct ingredients, such as, but not limited to, one or more solvents, dispersion media, diluents , dispersing aids, suspending aids, granulation aids, disintegrating agents, fillers, glidants, liquid vehicles, binders, surfactants, isotonic agents, thickeners or emulsifiers, buffers , lubricants, oils, preservatives and other substances. Excipients such as waxes, butters, colouring agents, coatings, flavouring and perfuming agents may also be included.
稀释剂的实例可以包括但不限于碳酸钙、碳酸钠、磷酸钙、磷酸二钙、硫酸钙、磷酸氢钙、磷酸钠、乳糖、蔗糖、纤维素、微晶纤维素、高岭土、甘露糖醇、山梨糖醇、肌醇、氯化钠、干淀粉、玉米淀粉、糖粉和/或其组合。造粒剂和分散剂可以选自由以下组成的非限制性列表:马铃薯淀粉、玉米淀粉、木薯淀粉、乙醇酸淀粉钠、粘土、海藻酸、瓜尔胶、柑桔渣、琼脂、膨润土、纤维素和木制品、天然海绵、阳离子交换树脂、碳酸钙、硅酸盐、碳酸钠、交联聚(乙烯吡咯烷酮)(交联聚维酮)、羧甲基淀粉钠(乙醇酸淀粉钠)、羧甲基纤维素、交联羧甲基纤维素(croscarmellose)钠、甲基纤维素、预糊化淀粉(淀粉1500)、微晶淀粉、水不溶性淀粉、羧甲基纤维素钙、硅酸铝镁月桂基硫酸钠、季铵化合物和/或其组合。Examples of diluents may include, but are not limited to, calcium carbonate, sodium carbonate, calcium phosphate, dicalcium phosphate, calcium sulfate, calcium hydrogen phosphate, sodium phosphate, lactose, sucrose, cellulose, microcrystalline cellulose, kaolin, mannitol, Sorbitol, inositol, sodium chloride, dry starch, corn starch, powdered sugar, and/or combinations thereof. Granulating and dispersing agents may be selected from a non-limiting list consisting of: potato starch, corn starch, tapioca starch, sodium starch glycolate, clay, alginic acid, guar gum, citrus pulp, agar, bentonite, cellulose and wood products, natural sponge, cation exchange resin, calcium carbonate, silicate, sodium carbonate, cross-linked poly(vinylpyrrolidone) (crospovidone), sodium carboxymethyl starch (sodium starch glycolate), carboxymethyl Base cellulose, croscarmellose sodium, methyl cellulose, pregelatinized starch (starch 1500), microcrystalline starch, water-insoluble starch, calcium carboxymethyl cellulose, magnesium aluminum silicate Sodium lauryl sulfate, quaternary ammonium compounds, and/or combinations thereof.
表面活性剂和/或乳化剂可以包括但不限于天然乳化剂(例如,阿拉伯树胶、琼脂、海藻酸、海藻酸钠、黄芪胶、角叉菜属(chondrux)、胆固醇、黄原胶、果胶、明胶、蛋黄、酪蛋白、羊毛脂、胆固醇、蜡以及卵磷脂)、胶状粘土(例如膨润土[硅酸铝]和[硅酸镁铝])、长链氨基酸衍生物、高分子量醇类(例如硬脂醇、鲸蜡醇、油醇、单硬脂酸三乙酸甘油酯、二硬脂酸乙二醇酯、单硬脂酸甘油酯、以及单硬脂酸丙二醇酯、聚乙烯醇)、卡波姆(例如羧基聚亚甲基、聚丙烯酸、丙烯酸聚合物、以及羧乙烯聚合物)、卡拉胶、纤维素衍生物(例如羧甲基纤维素钠、粉末状纤维素、羟甲基纤维素、羟丙基纤维素、羟丙基甲基纤维素、甲基纤维素)、脱水山梨糖醇脂肪酸酯(例如,聚氧乙烯脱水山梨糖醇单月桂酸酯聚氧乙烯脱水山梨糖醇聚氧乙烯脱水山梨糖醇单油酸酯脱水山梨糖醇单棕榈酸酯脱水山梨糖醇单硬脂酸酯脱水山梨糖醇三硬脂酸酯单油酸甘油酯、脱水山梨糖醇单油酸酯聚氧乙烯酯(例如聚氧乙烯单硬脂酸酯聚氧乙烯氢化蓖麻油、聚乙氧基化蓖麻油、聚甲醛硬脂酸酯以及)、蔗糖脂肪酸酯、聚乙二醇脂肪酸酯(例如)、聚氧乙烯醚(例如聚氧乙烯月桂基醚聚(乙烯吡咯烷酮)、二乙二醇单月桂酸酯、三乙醇胺油酸酯、油酸钠、油酸钾、油酸乙酯、油酸、月桂酸乙酯、月桂基硫酸钠、溴化十六烷基三甲基铵、氯化十六烷基吡啶、苯扎氯铵、多库酯钠和/或其组合。Surfactants and/or emulsifiers may include, but are not limited to, natural emulsifiers (eg, gum arabic, agar, alginic acid, sodium alginate, tragacanth, chondrux, cholesterol, xanthan gum, pectin) , gelatin, egg yolk, casein, lanolin, cholesterol, waxes and lecithin), colloidal clays (such as bentonite [aluminum silicate] and [Magnesium Aluminum Silicate]), long-chain amino acid derivatives, high molecular weight alcohols (e.g. stearyl alcohol, cetyl alcohol, oleyl alcohol, triacetin monostearate, ethylene glycol distearate, mono Glyceryl stearate, and propylene glycol monostearate, polyvinyl alcohol), carbomers (eg, carboxypolymethylene, polyacrylic acid, acrylic polymers, and carboxyvinyl polymers), carrageenan, cellulose derivatives substances (such as sodium carboxymethyl cellulose, powdered cellulose, hydroxymethyl cellulose, hydroxypropyl cellulose, hydroxypropyl methyl cellulose, methyl cellulose), sorbitan fatty acid esters (such as , polyoxyethylene sorbitan monolaurate polyoxyethylene sorbitan Polyoxyethylene sorbitan monooleate Sorbitan Monopalmitate Sorbitan Monostearate Sorbitan Tristearate Glyceryl Monooleate, Sorbitan Monooleate Polyoxyethylene esters (such as polyoxyethylene monostearate Polyoxyethylene hydrogenated castor oil, polyethoxylated castor oil, polyoxymethylene stearate, and ), sucrose fatty acid esters, polyethylene glycol fatty acid esters (such as ), polyoxyethylene ethers (such as polyoxyethylene lauryl ether Poly(vinylpyrrolidone), diethylene glycol monolaurate, triethanolamine oleate, sodium oleate, potassium oleate, ethyl oleate, oleic acid, ethyl laurate, sodium lauryl sulfate, Cetyltrimethylammonium bromide, cetylpyridine chloride, benzalkonium chloride, sodium docusate, and/or combinations thereof.
粘结剂可以是淀粉(例如玉米淀粉和淀粉糊);明胶;糖(例如蔗糖、葡萄糖、右旋糖、糊精、糖蜜、乳糖、乳糖醇、甘露糖醇);天然和合成的树胶(例如阿拉伯树胶、海藻酸钠、角叉菜提取物、panwar树胶、印度树胶、isapol皮的粘液质、羧甲基纤维素、甲基纤维素、乙基纤维素、羟乙基纤维素、羟丙基纤维素、羟丙基甲基纤维素、微晶纤维素、乙酸纤维素、聚(乙烯吡咯烷酮)、硅酸镁铝以及落叶松阿拉伯半乳聚糖);海藻酸盐;聚环氧乙烷;聚乙二醇;无机钙盐;硅酸;聚甲基丙烯酸酯;蜡;水;醇及其组合,或任何其他适合的粘结剂。Binders can be starches (eg cornstarch and starch paste); gelatin; sugars (eg sucrose, glucose, dextrose, dextrin, molasses, lactose, lactitol, mannitol); natural and synthetic gums (eg Gum arabic, sodium alginate, carrageenan extract, panwar gum, gum india, mucilage of isapol bark, carboxymethyl cellulose, methyl cellulose, ethyl cellulose, hydroxyethyl cellulose, hydroxypropyl Cellulose, Hydroxypropyl Methylcellulose, Microcrystalline Cellulose, Cellulose Acetate, Poly(vinylpyrrolidone), Magnesium Aluminum Silicate and larch arabinogalactan); alginates; polyethylene oxide; polyethylene glycol; inorganic calcium salts; silicic acid; polymethacrylates; waxes; water; alcohols and combinations thereof, or any other suitable binder.
防腐剂的实例可以包括但不限于抗氧化剂、螯合剂、抗微生物防腐剂、抗真菌防腐剂、醇防腐剂、酸性防腐剂和/或其他防腐剂。抗氧化剂的实例包括但不限于α生育酚、抗坏血酸、抗坏血酸棕榈酸酯、丁基化羟基茴香醚、丁基化羟基甲苯、一硫代甘油、偏亚硫酸氢钾、丙酸、没食子酸丙酯、抗坏血酸钠、亚硫酸氢钠、偏亚硫酸氢钠和/或亚硫酸钠。螯合剂的实例包括乙二胺四乙酸(EDTA)、柠檬酸一水合物、依地酸二钠、依地酸二钾、依地酸、富马酸、苹果酸、磷酸、依地酸钠、酒石酸和/或依地酸三钠。抗微生物防腐剂的实例包括但不限于苯扎氯铵、苄索氯铵、苄醇、溴硝丙二醇、西曲溴铵、氯化十六烷基吡啶鎓、氯己定、氯丁醇、氯甲酚、氯二甲苯酚、甲酚、乙醇、甘油、海克替啶、咪脲、苯酚、苯氧乙醇、苯乙醇、硝酸苯汞、丙二醇和/或硫柳汞。抗真菌防腐剂的实例包括但不限于对羟基苯甲酸丁酯、对羟基苯甲酸甲酯、对羟基苯甲酸乙酯、对羟基苯甲酸丙酯、苯甲酸、羟基苯甲酸、苯甲酸钾、山梨酸钾、苯甲酸钠、丙酸钠和/或山梨酸。醇防腐剂的实例包括但不限于乙醇、聚乙二醇、苄醇、苯酚、酚类化合物、双酚、氯丁醇、羟基苯甲酸酯和/或苯乙醇。酸性防腐剂的实例包括但不限于维生素A、维生素C、维生素E、β-胡萝卜素、柠檬酸、乙酸、脱氢抗坏血酸、抗坏血酸、山梨酸和/或植酸。其他防腐剂包括但不限于生育酚、生育酚乙酸酯、甲磺酸地特肟(deteroximemesylate)、西曲溴铵、丁基化羟基茴香醚(BHA)、丁基化羟基甲苯(BHT)、乙二胺、月桂基硫酸钠(SLS)、月桂基醚硫酸钠(SLES)、亚硫酸氢钠、偏亚硫酸氢钠、亚硫酸钾、偏亚硫酸氢钾、GLYDANT对羟基苯甲酸甲酯、NEOLONETM、KATHONTM和/或 Examples of preservatives may include, but are not limited to, antioxidants, chelating agents, antimicrobial preservatives, antifungal preservatives, alcohol preservatives, acid preservatives, and/or other preservatives. Examples of antioxidants include, but are not limited to, alpha tocopherol, ascorbic acid, ascorbyl palmitate, butylated hydroxyanisole, butylated hydroxytoluene, monothioglycerol, potassium metabisulfite, propionic acid, propyl gallate , sodium ascorbate, sodium bisulfite, sodium metabisulfite and/or sodium sulfite. Examples of chelating agents include ethylenediaminetetraacetic acid (EDTA), citric acid monohydrate, disodium edetate, dipotassium edetate, edetate, fumaric acid, malic acid, phosphoric acid, sodium edetate, Tartaric acid and/or trisodium edetate. Examples of antimicrobial preservatives include, but are not limited to, benzalkonium chloride, benzethonium chloride, benzyl alcohol, bronopol, cetrimonium bromide, cetylpyridinium chloride, chlorhexidine, chlorobutanol, chlorine Cresol, chloroxylenol, cresol, ethanol, glycerol, hexamidine, imidazolium, phenol, phenoxyethanol, phenethyl alcohol, phenylmercuric nitrate, propylene glycol and/or thimerosal. Examples of antifungal preservatives include, but are not limited to, butylparaben, methylparaben, ethylparaben, propylparaben, benzoic acid, hydroxybenzoic acid, potassium benzoate, sorbitan Potassium acid, sodium benzoate, sodium propionate and/or sorbic acid. Examples of alcohol preservatives include, but are not limited to, ethanol, polyethylene glycol, benzyl alcohol, phenol, phenolic compounds, bisphenol, chlorobutanol, parabens, and/or phenethyl alcohol. Examples of acidic preservatives include, but are not limited to, vitamin A, vitamin C, vitamin E, beta-carotene, citric acid, acetic acid, dehydroascorbic acid, ascorbic acid, sorbic acid, and/or phytic acid. Other preservatives include, but are not limited to, tocopherol, tocopherol acetate, deteroximemesylate, cetrimonium bromide, butylated hydroxyanisole (BHA), butylated hydroxytoluene (BHT), Ethylenediamine, Sodium Lauryl Sulfate (SLS), Sodium Lauryl Ether Sulfate (SLES), Sodium Bisulfite, Sodium Metabisulfite, Potassium Sulfite, Potassium Metabisulfite, GLYDANT Methylparaben, NEOLONE ™ , KATHON ™ and/or
缓冲剂的实例包括但不限于柠檬酸盐缓冲溶液、乙酸盐缓冲溶液、磷酸盐缓冲溶液、氯化铵、碳酸钙、氯化钙、柠檬酸钙、葡乳醛酸钙、葡庚糖酸钙、葡糖酸钙、d-葡糖酸、甘油磷酸钙、乳酸钙、乳糖酸钙、丙酸、乙酰丙酸钙、戊酸、磷酸氢钙、磷酸、磷酸三钙、羟基磷酸钙、乙酸钾、氯化钾、葡糖酸钾、钾混合物、磷酸氢二钾、磷酸二氢钾、磷酸钾混合物、乙酸钠、碳酸氢钠、氯化钠、柠檬酸钠、乳酸钠、磷酸氢二钠、磷酸二氢钠、磷酸钠混合物、氨丁三醇(tromethamine)、氨基磺酸盐缓冲剂(例如HEPES)、氢氧化镁、氢氧化铝、海藻酸、无热原水、等渗盐水、林格氏溶液(Ringer’s solution)、乙醇和/或其组合。润滑剂可以选自由以下组成的非限制性组:硬脂酸镁、硬脂酸钙、硬脂酸、二氧化硅、滑石、麦芽、山嵛酸甘油酯、氢化植物油、聚乙二醇、苯甲酸钠、乙酸钠、氯化钠、亮氨酸、月桂基硫酸镁、月桂基硫酸钠及其组合。Examples of buffers include, but are not limited to, citrate buffer solution, acetate buffer solution, phosphate buffer solution, ammonium chloride, calcium carbonate, calcium chloride, calcium citrate, calcium glucuronic acid, glucoheptonic acid Calcium, calcium gluconate, d-gluconic acid, calcium glycerophosphate, calcium lactate, calcium lactobionate, propionic acid, calcium levulinate, valeric acid, calcium hydrogen phosphate, phosphoric acid, tricalcium phosphate, calcium hydroxyphosphate, acetic acid Potassium, Potassium Chloride, Potassium Gluconate, Potassium Mixture, Dipotassium Hydrogen Phosphate, Potassium Dihydrogen Phosphate, Potassium Phosphate Mixture, Sodium Acetate, Sodium Bicarbonate, Sodium Chloride, Sodium Citrate, Sodium Lactate, Disodium Hydrogen Phosphate, Sodium dihydrogen phosphate, sodium phosphate mixture, tromethamine, sulfamate buffer (eg HEPES), magnesium hydroxide, aluminum hydroxide, alginic acid, pyrogen-free water, isotonic saline, Ringer's Ringer's solution, ethanol, and/or combinations thereof. Lubricants may be selected from the non-limiting group consisting of magnesium stearate, calcium stearate, stearic acid, silicon dioxide, talc, malt, glyceryl behenate, hydrogenated vegetable oils, polyethylene glycol, benzene Sodium formate, sodium acetate, sodium chloride, leucine, magnesium lauryl sulfate, sodium lauryl sulfate, and combinations thereof.
油的实例包括但不限于杏仁油、杏核油、鳄梨油、巴巴苏油、佛手柑油、黑醋栗籽油、琉璃苣油、杜松油、甘菊油、卡诺拉油、香菜油、巴西棕榈油、蓖麻油、肉桂油、可可脂油、椰子油、鱼肝油、咖啡油、玉米油、棉籽油、鸸鹋油、桉树油、月见草油、鱼油、亚麻籽油、香叶醇油、葫芦油、葡萄籽油、榛子油、牛膝草油、肉豆蔻酸异丙酯、荷荷巴油、夏威夷核果油、醒目薰衣草油、薰衣草油、柠檬油、山苍子油、夏威夷果仁油、锦葵油、芒果籽油、白芒花籽油、貂油、肉豆蔻油、橄榄油、橙油、橘棘鲷油、棕榈油、棕榈仁油、桃仁油、花生油、罂粟籽油、南瓜籽油、油菜籽油、米糠油、迷迭香油、红花油、檀香木油、山茶花油、香薄荷油、沙棘油、芝麻油、乳木果油、硅油、大豆油、向日葵油、茶树油、蓟油、椿油、香根草油、胡桃油和小麦胚芽油以及硬脂酸丁酯、辛酸甘油三酯、癸酸甘油三酯、环甲硅油、癸二酸二乙酯、二甲硅油360、西甲硅油、肉豆蔻酸异丙酯、矿物油、辛基十二烷醇、油醇、硅油和/或其组合。Examples of oils include, but are not limited to, almond oil, apricot kernel oil, avocado oil, babassu oil, bergamot oil, black currant seed oil, borage oil, juniper oil, chamomile oil, canola oil, parsley Oil, Carnauba Oil, Castor Oil, Cinnamon Oil, Cocoa Butter Oil, Coconut Oil, Cod Liver Oil, Coffee Oil, Corn Oil, Cottonseed Oil, Emu Oil, Eucalyptus Oil, Evening Primrose Oil, Fish Oil, Flaxseed Oil, Geraniol Oil, Gourd Oil, Grapeseed Oil, Hazelnut Oil, Hyssop Oil, Isopropyl Myristate, Jojoba Oil, Macadamia Nut Oil, Striking Lavender Oil, Lavender Oil, Lemon Oil, Wild Goat Oil, Macadamia Nut Kernel Oil, Mallow Oil, Mango Seed Oil, Mango Seed Oil, Mink Oil, Nutmeg Oil, Olive Oil, Orange Oil, Tangerine Snapper Oil, Palm Oil, Palm Kernel Oil, Peach Kernel Oil, Peanut Oil, Poppy Seed Oil , Pumpkin Seed Oil, Rapeseed Oil, Rice Bran Oil, Rosemary Oil, Safflower Oil, Sandalwood Oil, Camellia Oil, Savory Oil, Sea Buckthorn Oil, Sesame Oil, Shea Butter, Silicone Oil, Soybean Oil, Sunflower Oil, Tea Tree Oils, Thistle Oil, Toona Oil, Vetiver Oil, Walnut Oil and Wheat Germ Oil as well as Butyl Stearate, Caprylic Triglyceride, Capric Triglyceride, Cyclomethicone, Diethyl Sebacate, Dimethicone 360, simethicone, isopropyl myristate, mineral oil, octyldodecanol, oleyl alcohol, silicone oil, and/or combinations thereof.
制剂preparation
脂质纳米粒子(例如,空LNP或负载LNP)可以包含脂质组分和一种或多种附加组分,诸如治疗剂和/或预防剂。脂质纳米粒子(例如,空LNP或负载LNP)可以被设计用于一种或多种指定应用或靶标。脂质纳米粒子(例如,空LNP或负载LNP)的成分可以基于特定应用或靶标和/或基于一种或多种成分的功效、毒性、费用、使用便利性、可用率或其他特征来选择。类似地,纳米粒子组合物的特定制剂可以根据例如特定成分的组合的功效和毒性,针对特定应用或靶标来选择。Lipid nanoparticles (eg, empty LNPs or loaded LNPs) can comprise lipid components and one or more additional components, such as therapeutic and/or prophylactic agents. Lipid nanoparticles (eg, empty LNPs or loaded LNPs) can be designed for one or more specified applications or targets. Compositions of lipid nanoparticles (eg, empty LNPs or loaded LNPs) can be selected based on a particular application or target and/or based on efficacy, toxicity, cost, ease of use, availability, or other characteristics of one or more of the components. Similarly, a particular formulation of a nanoparticle composition can be selected for a particular application or target based on, for example, the efficacy and toxicity of a particular combination of ingredients.
纳米粒子组合物的脂质组分可以包含例如根据式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的脂质,磷脂(诸如不饱和脂质,例如,DOPE或DSPC)、PEG脂质和结构性脂质。脂质组分的成分可以指定分数提供。The lipid component of the nanoparticle composition may comprise, for example, according to formulae (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y) , (I-Ya)(IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb) Lipids, phospholipids (such as unsaturated lipids, eg, DOPE or DSPC), PEG lipids, and structured lipids. Components of lipid components can be provided in specified fractions.
在一些实施方案中,纳米粒子组合物的脂质组分包含根据式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的脂质,磷脂,PEG脂质和结构性脂质。在某些实施方案中,纳米粒子组合物的脂质组分包含约30mol%至约60mol%式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,约0mol%至约30mol%磷脂,约18.5mol%至约48.5mol%结构性脂质,以及约0mol%至约10mol%PEG脂质,条件是总mol%不超过100%。在一些实施方案中,纳米粒子组合物的脂质组分包含约35mol%至约55mol%的式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,约5mol%至约25mol%磷脂,约30mol%至约40mol%结构性脂质,以及约0mol%至约10mol%PEG脂质。在一个特定实施方案中,所述脂质组分包含约50mol%所述化合物、约10mol%磷脂、约38.5mol%结构性脂质和约1.5mol%PEG脂质。在另一个特定实施方案中,所述脂质组分包含约40mol%所述化合物、约20mol%磷脂、约38.5mol%结构性脂质和约1.5mol%PEG脂质。在一些实施方案中,所述磷脂可以是DOPE或DSPC。在其他实施方案中,所述PEG脂质可以是PEG-1或PEG2k-DMG,且/或结构性脂质可以是胆固醇。In some embodiments, the lipid component of the nanoparticle composition comprises according to formula (A), (A1), (A2), (A3), (B), (I), (I'), (IX) , (IY), (I-Ya)(IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb) Lipids, phospholipids, PEG lipids and structured lipids. In certain embodiments, the lipid component of the nanoparticle composition comprises from about 30 mol% to about 60 mol% of formulae (A), (A1), (A2), (A3), (B), (I), ( I'), (IX), (IY), (I-Ya) (IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), ( II), (IIa) or (IIb), about 0 mol% to about 30 mol% phospholipids, about 18.5 mol% to about 48.5 mol% structured lipids, and about 0 mol% to about 10 mol% PEG lipids, provided that The total mol% does not exceed 100%. In some embodiments, the lipid component of the nanoparticle composition comprises from about 35 mol% to about 55 mol% of formula (A), (A1), (A2), (A3), (B), (I), ( I'), (IX), (IY), (I-Ya) (IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), ( A compound of II), (IIa) or (IIb), about 5 mol% to about 25 mol% phospholipids, about 30 mol% to about 40 mol% structured lipids, and about 0 mol% to about 10 mol% PEG lipids. In a specific embodiment, the lipid component comprises about 50 mol % of the compound, about 10 mol % phospholipids, about 38.5 mol % structured lipids, and about 1.5 mol % PEG lipids. In another specific embodiment, the lipid component comprises about 40 mol % of the compound, about 20 mol % phospholipids, about 38.5 mol % structured lipids, and about 1.5 mol % PEG lipids. In some embodiments, the phospholipid can be DOPE or DSPC. In other embodiments, the PEG lipid can be PEG-1 or PEG 2k -DMG, and/or the structural lipid can be cholesterol.
在一些实施方案中,空脂质纳米粒子(空LNP)包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,磷脂,结构性脂质和PEG脂质。In some embodiments, empty lipid nanoparticles (empty LNPs) comprise formulae (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y), (I-Ya)(IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or ( Compounds of IIb), phospholipids, structured lipids and PEG lipids.
在一些实施方案中,负载脂质纳米粒子(负载LNP)包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,磷脂,结构性脂质,PEG脂质以及一种或多种治疗剂和/或预防剂。In some embodiments, the loaded lipid nanoparticles (loaded LNPs) comprise formulae (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y), (I-Ya)(IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or ( The compound of IIb), phospholipids, structured lipids, PEG lipids, and one or more therapeutic and/or prophylactic agents.
在一些实施方案中,空LNP或负载LNP包含约40%至约60%的量的式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物。In some embodiments, the empty LNP or loaded LNP comprises formula (A), (A1), (A2), (A3), (B), (I), (I' in an amount of about 40% to about 60% ), (I-X), (I-Y), (I-Ya) (IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II) , (IIa) or (IIb).
在一些实施方案中,空LNP或负载LNP包含约0%至约20%的量的磷脂。例如,在一些实施方案中,空LNP或负载LNP包含约0%至约20%的量的DSPC。In some embodiments, empty LNPs or loaded LNPs comprise phospholipids in an amount from about 0% to about 20%. For example, in some embodiments, the empty LNP or loaded LNP comprises DSPC in an amount from about 0% to about 20%.
在一些实施方案中,空LNP或负载LNP包含约30%至约50%的量的结构性脂质。例如,在一些实施方案中,空LNP或负载LNP包含约30%至约50%的量的胆固醇。In some embodiments, empty LNPs or loaded LNPs comprise structured lipids in an amount from about 30% to about 50%. For example, in some embodiments, empty LNPs or loaded LNPs comprise cholesterol in an amount from about 30% to about 50%.
在一些实施方案中,空LNP或负载LNP包含约0%至约5%的量的PEG脂质。例如,在一些实施方案中,空LNP或负载LNP包含约0%至约5%的量的PEG-1或PEG2k-DMG。In some embodiments, empty LNPs or loaded LNPs comprise PEG lipids in an amount from about 0% to about 5%. For example, in some embodiments, empty LNPs or loaded LNPs comprise PEG-1 or PEG2k -DMG in an amount from about 0% to about 5%.
在一些实施方案中,空LNP或负载LNP包含约40mol%至约60mol%的式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,约0mol%至约20mol%磷脂,约30mol%至约50mol%结构性脂质,以及约0mol%至约5mol%PEG脂质。In some embodiments, the empty LNP or loaded LNP comprises from about 40 mol % to about 60 mol % of formula (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y), (I-Ya) (IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), ( A compound of IIa) or (IIb), about 0 mol% to about 20 mol% phospholipids, about 30 mol% to about 50 mol% structured lipids, and about 0 mol% to about 5 mol% PEG lipids.
在一些实施方案中,空LNP或负载LNP包含约40mol%至约60mol%的式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,约0mol%至约20mol%DSPC,约30mol%至约50mol%胆固醇,以及约0mol%至约5mol%PEG2k-DMG。在一些实施方案中,空LNP或负载LNP包含约40mol%至约60mol%的表1的化合物,约0mol%至约20mol%DSPC,约30mol%至约50mol%胆固醇,以及约0mol%至约5mol%PEG2k-DMG。In some embodiments, the empty LNP or loaded LNP comprises from about 40 mol % to about 60 mol % of formula (A), (A1), (A2), (A3), (B), (I), (I'), (IX), (IY), (I-Ya) (IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), ( A compound of IIa) or (IIb), about 0 mol% to about 20 mol% DSPC, about 30 mol% to about 50 mol% cholesterol, and about 0 mol% to about 5 mol% PEG 2k -DMG. In some embodiments, the empty LNP or loaded LNP comprises about 40 mol% to about 60 mol% of the compound of Table 1, about 0 mol% to about 20 mol% DSPC, about 30 mol% to about 50 mol% cholesterol, and about 0 mol% to about 5 mol% %PEG 2k -DMG.
在一些实施方案中,空LNP或负载LNP包含约40mol%至约60mol%的式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,约0mol%至约20mol%DSPC,约30mol%至约50mol%胆固醇,以及约0mol%至约5mol%PEG-1。在一些实施方案中,空LNP或负载LNP包含约40mol%至约60mol%的表1的化合物,约0mol%至约20mol%DSPC,约30mol%至约50mol%胆固醇,以及约0mol%至约5mol%PEG-1。In some embodiments, the empty LNP or loaded LNP comprises from about 40 mol % to about 60 mol % of formula (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y), (I-Ya) (IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), ( A compound of IIa) or (IIb), about 0 mol% to about 20 mol% DSPC, about 30 mol% to about 50 mol% cholesterol, and about 0 mol% to about 5 mol% PEG-1. In some embodiments, the empty LNP or loaded LNP comprises about 40 mol% to about 60 mol% of the compound of Table 1, about 0 mol% to about 20 mol% DSPC, about 30 mol% to about 50 mol% cholesterol, and about 0 mol% to about 5 mol% %PEG-1.
在一些实施方案中,空LNP或负载LNP包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,磷脂,结构性脂质和PEG脂质,其中所述磷脂是DSPC并且所述结构性脂质是胆固醇。在一些实施方案中,空LNP或负载LNP包含表1的化合物、磷脂、结构性脂质和PEG脂质,其中所述磷脂是DSPC并且所述结构性脂质是胆固醇。In some embodiments, the empty LNP or loaded LNP comprises formula (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y), (I-Ya) Compounds of (IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb) , phospholipids, structured lipids and PEG lipids, wherein the phospholipid is DSPC and the structured lipid is cholesterol. In some embodiments, the empty LNP or loaded LNP comprises a compound of Table 1, a phospholipid, a structured lipid, and a PEG lipid, wherein the phospholipid is DSPC and the structured lipid is cholesterol.
在一些实施方案中,空LNP或负载LNP包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,磷脂,结构性脂质和PEG脂质,其中所述结构性脂质是胆固醇并且所述PEG脂质是PEG2k-DMG。在一些实施方案中,空LNP或负载LNP包含表1的化合物、磷脂、结构性脂质和PEG脂质,其中所述结构性脂质是胆固醇并且所述PEG脂质是PEG2k-DMG。In some embodiments, the empty LNP or loaded LNP comprises formula (A), (A1), (A2), (A3), (B), (I), (I'), (IX), (IY), (I-Ya) Compounds of (IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb) , a phospholipid, a structured lipid, and a PEG lipid, wherein the structured lipid is cholesterol and the PEG lipid is PEG 2k -DMG. In some embodiments, the empty LNP or loaded LNP comprises a compound of Table 1, a phospholipid, a structured lipid, and a PEG lipid, wherein the structured lipid is cholesterol and the PEG lipid is PEG 2k -DMG.
在一些实施方案中,空LNP或负载LNP包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,磷脂,结构性脂质和PEG脂质,其中所述结构性脂质是胆固醇并且所述PEG脂质是PEG-1。在一些实施方案中,空LNP或负载LNP包含表1的化合物、磷脂、结构性脂质和PEG脂质,其中所述结构性脂质是胆固醇并且所述PEG脂质是PEG-1。In some embodiments, the empty LNP or loaded LNP comprises formula (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y), (I-Ya) Compounds of (IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb) , phospholipids, structured lipids and PEG lipids, wherein the structured lipid is cholesterol and the PEG lipid is PEG-1. In some embodiments, the empty LNP or loaded LNP comprises the compound of Table 1, a phospholipid, a structured lipid, and a PEG lipid, wherein the structured lipid is cholesterol and the PEG lipid is PEG-1.
在一些实施方案中,空LNP或负载LNP包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,磷脂,结构性脂质和PEG脂质,其中所述磷脂是DSPC并且所述PEG脂质是PEG2k-DMG。在一些实施方案中,空LNP或负载LNP包含表1的化合物、磷脂、结构性脂质和PEG脂质,其中所述磷脂是DSPC并且所述PEG脂质是PEG2k-DMG。In some embodiments, the empty LNP or loaded LNP comprises formula (A), (A1), (A2), (A3), (B), (I), (I'), (IX), (IY), (I-Ya) Compounds of (IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb) , a phospholipid, a structured lipid and a PEG lipid, wherein the phospholipid is DSPC and the PEG lipid is PEG 2k -DMG. In some embodiments, the empty LNP or loaded LNP comprises a compound of Table 1, a phospholipid, a structured lipid, and a PEG lipid, wherein the phospholipid is DSPC and the PEG lipid is PEG 2k -DMG.
在一些实施方案中,空LNP或负载LNP包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,磷脂,结构性脂质和PEG脂质,其中所述磷脂是DSPC并且所述PEG脂质是PEG-1。在一些实施方案中,空LNP或负载LNP包含表1的化合物、磷脂、结构性脂质和PEG脂质,其中所述磷脂是DSPC并且所述PEG脂质是PEG-1。In some embodiments, the empty LNP or loaded LNP comprises formula (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y), (I-Ya) Compounds of (IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb) , phospholipids, structural lipids and PEG lipids, wherein the phospholipid is DSPC and the PEG lipid is PEG-1. In some embodiments, the empty LNP or loaded LNP comprises the compound of Table 1, a phospholipid, a structured lipid, and a PEG lipid, wherein the phospholipid is DSPC and the PEG lipid is PEG-1.
在一些实施方案中,空LNP或负载LNP包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,磷脂,结构性脂质和PEG脂质,其中所述磷脂是DSPC,所述结构性脂质是胆固醇并且所述PEG脂质是PEG2k-DMG。在一些实施方案中,空LNP或负载LNP负载LNP包含表1的化合物、磷脂、结构性脂质和PEG脂质,其中所述磷脂是DSPC,所述结构性脂质是胆固醇并且所述PEG脂质是PEG2k-DMG。In some embodiments, the empty LNP or loaded LNP comprises formula (A), (A1), (A2), (A3), (B), (I), (I'), (IX), (IY), (I-Ya) Compounds of (IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb) , a phospholipid, a structured lipid and a PEG lipid, wherein the phospholipid is DSPC, the structured lipid is cholesterol and the PEG lipid is PEG 2k -DMG. In some embodiments, the empty LNP or loaded LNP loaded LNP comprises a compound of Table 1, a phospholipid, a structured lipid, and a PEG lipid, wherein the phospholipid is DSPC, the structured lipid is cholesterol and the PEG lipid The mass is PEG 2k -DMG.
在一些实施方案中,空LNP或负载LNP包含式(A)的化合物、磷脂、结构性脂质和PEG脂质,其中所述磷脂是DSPC,所述结构性脂质是胆固醇并且所述PEG脂质是PEG2k-DMG。在一些实施方案中,空LNP或负载LNP负载LNP包含表1的化合物、磷脂、结构性脂质和PEG脂质,其中所述磷脂是DSPC,所述结构性脂质是胆固醇并且所述PEG脂质是PEG2k-DMG。In some embodiments, an empty LNP or a loaded LNP comprises a compound of formula (A), a phospholipid, a structured lipid, and a PEG lipid, wherein the phospholipid is DSPC, the structured lipid is cholesterol and the PEG lipid The mass is PEG 2k -DMG. In some embodiments, the empty LNP or loaded LNP loaded LNP comprises a compound of Table 1, a phospholipid, a structured lipid, and a PEG lipid, wherein the phospholipid is DSPC, the structured lipid is cholesterol and the PEG lipid The mass is PEG 2k -DMG.
在一些实施方案中,空LNP或负载LNP包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,磷脂,结构性脂质和PEG脂质,其中所述磷脂是DSPC,所述结构性脂质是胆固醇并且所述PEG脂质是PEG-1。In some embodiments, the empty LNP or loaded LNP comprises formula (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y), (I-Ya) Compounds of (IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb) , a phospholipid, a structured lipid and a PEG lipid, wherein the phospholipid is DSPC, the structured lipid is cholesterol and the PEG lipid is PEG-1.
在一些实施方案中,空LNP或负载LNP包含式(A)的化合物、磷脂、结构性脂质和PEG脂质,其中所述磷脂是DSPC,所述结构性脂质是胆固醇并且所述PEG脂质是PEG-1。在一些实施方案中,空LNP或负载LNP包含表1的化合物、磷脂、结构性脂质和PEG脂质,其中所述磷脂是DSPC,所述结构性脂质是胆固醇并且所述PEG脂质是PEG-1。In some embodiments, an empty LNP or a loaded LNP comprises a compound of formula (A), a phospholipid, a structured lipid, and a PEG lipid, wherein the phospholipid is DSPC, the structured lipid is cholesterol and the PEG lipid The quality is PEG-1. In some embodiments, the empty LNP or loaded LNP comprises a compound of Table 1, a phospholipid, a structured lipid, and a PEG lipid, wherein the phospholipid is DSPC, the structured lipid is cholesterol and the PEG lipid is PEG-1.
脂质纳米粒子(例如,空LNP或负载LNP)可以被设计用于一种或多种特定应用或靶标。例如,纳米粒子组合物可以被设计成将治疗剂和/或预防剂诸如RNA递送至哺乳动物体内的特定细胞、组织、器官或系统或其组。为了增加对特定身体靶标的选择性,可以改变脂质纳米粒子(例如,空LNP或负载LNP)的物理化学特性。例如,可以基于不同器官的开窗尺寸调整粒度。纳米粒子组合物中所包含的治疗剂和/或预防剂也可以基于一个或多个所需的递送靶标来选择。例如,可以针对特定适应症、病状、疾病或病症和/或针对向特定细胞、组织、器官或系统或其组的递送(例如,局部或特异性递送)选择治疗剂和/或预防剂。在某些实施方案中,纳米粒子组合物可以包含编码所关注的多肽的mRNA,所述mRNA能够在细胞内翻译以产生所关注的多肽。这种组合物可以被设计成特异性递送至特定器官。在一些实施方案中,组合物可以被设计成特异性递送至哺乳动物肝。Lipid nanoparticles (eg, empty LNPs or loaded LNPs) can be designed for one or more specific applications or targets. For example, nanoparticle compositions can be designed to deliver therapeutic and/or prophylactic agents such as RNA to specific cells, tissues, organs or systems or groups thereof in a mammal. To increase selectivity for specific body targets, the physicochemical properties of lipid nanoparticles (eg, empty LNPs or loaded LNPs) can be altered. For example, granularity can be adjusted based on the fenestration size of different organs. The therapeutic and/or prophylactic agents included in the nanoparticle composition can also be selected based on one or more desired delivery targets. For example, therapeutic and/or prophylactic agents can be selected for a particular indication, condition, disease or disorder and/or for delivery (eg, local or specific delivery) to a particular cell, tissue, organ or system or group thereof. In certain embodiments, a nanoparticle composition may comprise mRNA encoding a polypeptide of interest that is capable of being translated within a cell to produce the polypeptide of interest. Such compositions can be designed for specific delivery to specific organs. In some embodiments, the composition can be designed for specific delivery to mammalian liver.
纳米粒子组合物中治疗剂和/或预防剂的量可以取决于纳米粒子组合物的大小、组成、所需的靶标和/或应用或其他特性,以及治疗剂和/或预防剂的特性。例如,可用于纳米粒子组合物中的RNA的量可以取决于RNA的大小、序列和其他特征。纳米粒子组合物中治疗剂和/或预防剂和其他成分(例如,脂质)的相对量也可以变化。在一些实施方案中,纳米粒子组合物中脂质组分与治疗剂和/或预防剂的重量/重量比可以为约5:1至约60:1,诸如5:1、6:1、7:1、8:1、9:1、10:1、11:1、12:1、13:1、14:1、15:1、16:1、17:1、18:1、19:1、20:1、25:1、30:1、35:1、40:1、45:1、50:1和60:1。例如,脂质组分与治疗剂和/或预防剂的重量/重量比可以为约10:1至约40:1。在某些实施方案中,重量/重量比为约20:1。The amount of therapeutic and/or prophylactic agent in a nanoparticle composition may depend on the size, composition, desired target and/or application or other characteristics of the nanoparticle composition, as well as the characteristics of the therapeutic and/or prophylactic agent. For example, the amount of RNA that can be used in a nanoparticle composition can depend on the size, sequence, and other characteristics of the RNA. The relative amounts of therapeutic and/or prophylactic agents and other ingredients (eg, lipids) in the nanoparticle composition can also vary. In some embodiments, the weight/weight ratio of lipid component to therapeutic and/or prophylactic agent in the nanoparticle composition may be from about 5:1 to about 60:1, such as 5:1, 6:1, 7 :1, 8:1, 9:1, 10:1, 11:1, 12:1, 13:1, 14:1, 15:1, 16:1, 17:1, 18:1, 19:1 , 20:1, 25:1, 30:1, 35:1, 40:1, 45:1, 50:1, and 60:1. For example, the weight/weight ratio of lipid component to therapeutic and/or prophylactic agent may be from about 10:1 to about 40:1. In certain embodiments, the weight/weight ratio is about 20:1.
可以例如使用吸收光谱法(例如,紫外-可见光谱法)来测量纳米粒子组合物中治疗剂和/或预防剂的量。The amount of therapeutic and/or prophylactic agent in a nanoparticle composition can be measured, for example, using absorption spectroscopy (eg, UV-Vis spectroscopy).
在一些实施方案中,纳米粒子组合物包含一种或多种RNA,并且可以选择一种或多种RNA、脂质及其量以提供特定N:P比。所述组合物的N:P比是指一种或多种脂质中的氮原子与RNA中磷酸基团的数量的的摩尔比。一般而言,较低的N:P比是优选的。可以选择一种或多种RNA、脂质及其量以提供约2:1至约30:1,诸如2:1、3:1、4:1、5:1、6:1、7:1、8:1、9:1、10:1、12:1、14:1、16:1、18:1、20:1、22:1、24:1、26:1、28:1或30:1的N:P比。在某些实施方案中,N:P比可以为约2:1至约8:1。在其他实施方案中,N:P比为约5:1至约8:1。例如,N:P比可以为约5.0:1、约5.5:1、约5.67:1、约6.0:1、约6.5:1或约7.0:1。例如,N:P比可以为约5.67:1。In some embodiments, the nanoparticle composition comprises one or more RNAs, and the one or more RNAs, lipids, and amounts thereof can be selected to provide a particular N:P ratio. The N:P ratio of the composition refers to the molar ratio of nitrogen atoms in one or more lipids to the number of phosphate groups in the RNA. In general, lower N:P ratios are preferred. The one or more RNAs, lipids, and amounts thereof can be selected to provide from about 2:1 to about 30:1, such as 2:1, 3:1, 4:1, 5:1, 6:1, 7:1 , 8:1, 9:1, 10:1, 12:1, 14:1, 16:1, 18:1, 20:1, 22:1, 24:1, 26:1, 28:1, or 30 :1 N:P ratio. In certain embodiments, the N:P ratio can be from about 2:1 to about 8:1. In other embodiments, the N:P ratio is from about 5:1 to about 8:1. For example, the N:P ratio can be about 5.0:1, about 5.5:1, about 5.67:1, about 6.0:1, about 6.5:1, or about 7.0:1. For example, the N:P ratio can be about 5.67:1.
物理特性physical properties
脂质纳米粒子(例如,空LNP或负载LNP)的特征可以取决于其组分。例如,包含胆固醇作为结构性脂质的脂质纳米粒子(例如,空LNP或负载LNP)可以具有与包含不同结构性脂质的脂质纳米粒子(例如,空LNP或负载LNP)不同的特征。类似地,脂质纳米粒子(例如,空LNP或负载LNP)的特征可以取决于其组分的绝对或相对量。例如,包含较高摩尔分数的磷脂的脂质纳米粒子(例如,空LNP或负载LNP)可以具有与包含较低摩尔分数的磷脂的脂质纳米粒子(例如,空LNP或负载LNP)不同的特征。特性也可以根据制备纳米粒子组合物的方法和条件而变化。The characteristics of lipid nanoparticles (eg, empty LNPs or loaded LNPs) can depend on their components. For example, lipid nanoparticles comprising cholesterol as a structural lipid (eg, empty LNP or loaded LNP) may have different characteristics than lipid nanoparticles comprising different structured lipids (eg, empty LNP or loaded LNP). Similarly, the characteristics of lipid nanoparticles (eg, empty LNPs or loaded LNPs) can depend on the absolute or relative amounts of their components. For example, lipid nanoparticles comprising higher mole fractions of phospholipids (eg, empty LNPs or loaded LNPs) may have different characteristics than lipid nanoparticles comprising lower mole fractions of phospholipids (eg, empty LNPs or loaded LNPs) . Properties can also vary depending on the method and conditions of preparation of the nanoparticle composition.
脂质纳米粒子(例如,空LNP或负载LNP)可以通过多种方法来表征。例如,显微镜检查法(例如,透射电子显微镜检查法或扫描电子显微镜检查法)可用于检查纳米粒子组合物的形态和粒度分布。动态光散射或电位分析法(例如电位滴定法)可用于测量ζ电位。动态光散射还可以用于测定粒度。还可以使用仪器诸如Zetasizer Nano ZS(MalvernInstruments Ltd,Malvern,Worcestershire,UK)测量纳米粒子组合物的多个特征,诸如粒度、多分散性指数和ζ电位。Lipid nanoparticles (eg, empty LNPs or loaded LNPs) can be characterized by a variety of methods. For example, microscopy (eg, transmission electron microscopy or scanning electron microscopy) can be used to examine the morphology and particle size distribution of nanoparticle compositions. Dynamic light scattering or potentiometric analysis (eg potentiometric titration) can be used to measure zeta potential. Dynamic light scattering can also be used to determine particle size. Various characteristics of nanoparticle compositions, such as particle size, polydispersity index and zeta potential, can also be measured using an instrument such as the Zetasizer Nano ZS (Malvern Instruments Ltd, Malvern, Worcestershire, UK).
脂质纳米粒子(例如,空LNP或负载LNP)的平均大小可以介于数十纳米与数百纳米之间,例如,通过动态光散射(DLS)测量的。例如,平均大小可以为约40nm至约150nm,诸如约40nm、45nm、50nm、55nm、60nm、65nm、70nm、75nm、80nm、85nm、90nm、95nm、100nm、105nm、110nm、115nm、120nm、125nm、130nm、135nm、140nm、145nm或150nm。在一些实施方案中,脂质纳米粒子(例如,空LNP或负载LNP)的平均大小可以为约50nm至约100nm、约50nm至约90nm、约50nm至约80nm、约50nm至约70nm、约50nm至约60nm、约60nm至约100nm、约60nm至约90nm、约60nm至约80nm、约60nm至约70nm、约70nm至约150nm、约70nm至约130nm、约70nm至约100nm、约70nm至约90nm、约70nm至约80nm、约80nm至约150nm、约80nm至约130nm、约80nm至约100nm、约80nm至约90nm、约90nm至约150nm、约90nm至约130nm或约90nm至约100nm。在某些实施方案中,脂质纳米粒子(例如,空LNP或负载LNP)的平均大小可以为约70nm至约130nm或为约70nm至约100nm。在一个特定实施方案中,平均大小可以为约80nm。在其他实施方案中,平均大小可以为约100nm。在其他实施方案中,平均大小可以为约120nm。The average size of lipid nanoparticles (eg, empty LNPs or loaded LNPs) can be between tens of nanometers and hundreds of nanometers, eg, as measured by dynamic light scattering (DLS). For example, the average size can be from about 40 nm to about 150 nm, such as about 40 nm, 45 nm, 50 nm, 55 nm, 60 nm, 65 nm, 70 nm, 75 nm, 80 nm, 85 nm, 90 nm, 95 nm, 100 nm, 105 nm, 110 nm, 115 nm, 120 nm, 125 nm, 130nm, 135nm, 140nm, 145nm or 150nm. In some embodiments, the lipid nanoparticles (eg, empty LNPs or loaded LNPs) can have an average size of about 50 nm to about 100 nm, about 50 nm to about 90 nm, about 50 nm to about 80 nm, about 50 nm to about 70 nm, about 50 nm to about 60 nm, about 60 nm to about 100 nm, about 60 nm to about 90 nm, about 60 nm to about 80 nm, about 60 nm to about 70 nm, about 70 nm to about 150 nm, about 70 nm to about 130 nm, about 70 nm to about 100 nm, about 70 nm to about 90 nm, about 70 nm to about 80 nm, about 80 nm to about 150 nm, about 80 nm to about 130 nm, about 80 nm to about 100 nm, about 80 nm to about 90 nm, about 90 nm to about 150 nm, about 90 nm to about 130 nm, or about 90 nm to about 100 nm. In certain embodiments, the lipid nanoparticles (eg, empty LNPs or loaded LNPs) can have an average size of about 70 nm to about 130 nm or about 70 nm to about 100 nm. In a particular embodiment, the average size can be about 80 nm. In other embodiments, the average size can be about 100 nm. In other embodiments, the average size can be about 120 nm.
脂质纳米粒子(例如,空LNP或负载LNP)可以是相对均质的。多分散性指数可用于指示纳米粒子组合物的均质性,例如脂质纳米粒子(例如,空LNP或负载LNP)的粒度分布。较小(例如,小于0.3)的多分散性指数一般指示较窄的粒度分布。脂质纳米粒子(例如,空LNP或负载LNP)的多分散性指数可以为约0至约0.25,诸如0.01、0.02、0.03、0.04、0.05、0.06、0.07、0.08、0.09、0.10、0.11、0.12、0.13、0.14、0.15、0.16、0.17、0.18、0.19、0.20、0.21、0.22、0.23、0.24或0.25。在一些实施方案中,脂质纳米粒子(例如,空LNP或负载LNP)的多分散性指数可以为约0.10至约0.20。Lipid nanoparticles (eg, empty LNPs or loaded LNPs) can be relatively homogeneous. The polydispersity index can be used to indicate the homogeneity of a nanoparticle composition, such as the particle size distribution of lipid nanoparticles (eg, empty LNPs or loaded LNPs). A smaller (eg, less than 0.3) polydispersity index generally indicates a narrower particle size distribution. The polydispersity index of lipid nanoparticles (eg, empty LNPs or loaded LNPs) can be from about 0 to about 0.25, such as 0.01, 0.02, 0.03, 0.04, 0.05, 0.06, 0.07, 0.08, 0.09, 0.10, 0.11, 0.12 , 0.13, 0.14, 0.15, 0.16, 0.17, 0.18, 0.19, 0.20, 0.21, 0.22, 0.23, 0.24, or 0.25. In some embodiments, the polydispersity index of the lipid nanoparticles (eg, empty LNPs or loaded LNPs) can be from about 0.10 to about 0.20.
脂质纳米粒子(例如,空LNP或负载LNP)的ζ电位可用于指示所述组合物的电动电位。例如,ζ电位可以描述纳米粒子组合物的表面电荷。具有相对低电荷(正电荷或负电荷)的脂质纳米粒子(例如,空LNP或负载LNP)一般是所希望的,因为带较高电荷的物质可能与体内的细胞、组织和其他成分发生不希望的相互作用。在一些实施方案中,脂质纳米粒子(例如,空LNP或负载LNP)的ζ电位可以为约-10mV至约+20mV、约-10mV至约+15mV、约-10mV至约+10mV、约-10mV至约+5mV、约-10mV至约0mV、约-10mV至约-5mV、约-5mV至约+20mV、约-5mV至约+15mV、约-5mV至约+10mV、约-5mV至约+5mV、约-5mV至约0mV、约0mV至约+20mV、约0mV至约+15mV、约0mV至约+10mV、约0mV至约+5mV、约+5mV至约+20mV、约+5mV至约+15mV或约+5mV至约+10mV。The zeta potential of lipid nanoparticles (eg, empty LNPs or loaded LNPs) can be used to indicate the zeta potential of the composition. For example, the zeta potential can describe the surface charge of a nanoparticle composition. Lipid nanoparticles (e.g., empty LNPs or loaded LNPs) with relatively low charges (positive or negative) are generally desirable because higher-charged species may interact with cells, tissues, and other components in the body. hopeful interaction. In some embodiments, the zeta potential of the lipid nanoparticle (eg, empty LNP or loaded LNP) can be about -10 mV to about +20 mV, about -10 mV to about +15 mV, about -10 mV to about +10 mV, about -10 mV to about +10 mV 10mV to about +5mV, about -10mV to about 0mV, about -10mV to about -5mV, about -5mV to about +20mV, about -5mV to about +15mV, about -5mV to about +10mV, about -5mV to about +5mV, about -5mV to about 0mV, about 0mV to about +20mV, about 0mV to about +15mV, about 0mV to about +10mV, about 0mV to about +5mV, about +5mV to about +20mV, about +5mV to about About +15mV or about +5mV to about +10mV.
治疗剂和/或预防剂的包封效率描述了相对于所提供的初始量而言,在制备后被包封在纳米粒子组合物(例如,空LNP或负载LNP)中或以其他方式与脂质纳米粒子缔合的治疗剂和/或预防剂的量。希望的是较高的包封效率(例如,接近100%)。包封效率可以例如通过比较在用一种或多种有机溶剂或洗涤剂破坏脂质纳米粒子(例如,空LNP或负载LNP)之前和之后含有脂质纳米粒子(例如,空LNP或负载LNP)的溶液中治疗剂和/或预防剂的量来测量。荧光可用于测量溶液中游离治疗剂和/或预防剂(例如,RNA)的量。对于本文所述的脂质纳米粒子(例如,空LNP或负载LNP),治疗剂和/或预防剂的包封效率可以为至少50%,例如50%、55%、60%、65%、70%、75%、80%、85%、90%、91%、92%、93%、94%、95%、96%、97%、98%、99%或100%。在一些实施方案中,包封效率可以为至少80%。在某些实施方案中,包封效率可以为至少90%。在一些实施方案中,治疗剂和/或预防剂的包封效率介于80%与100%之间。The encapsulation efficiency of a therapeutic and/or prophylactic agent describes, relative to the initial amount provided, encapsulated in a nanoparticle composition (eg, empty LNP or loaded LNP) after preparation or otherwise combined with lipids. The amount of therapeutic and/or prophylactic agent associated with the mass nanoparticles. A higher encapsulation efficiency (eg, close to 100%) is desired. Encapsulation efficiency can be measured, for example, by comparing lipid nanoparticles (eg, empty LNPs or loaded LNPs) before and after disruption of lipid nanoparticles (eg, empty LNPs or loaded LNPs) with one or more organic solvents or detergents The amount of therapeutic and/or prophylactic agent in the solution is measured. Fluorescence can be used to measure the amount of free therapeutic and/or prophylactic (eg, RNA) in solution. For lipid nanoparticles (eg, empty LNPs or loaded LNPs) described herein, the encapsulation efficiency of therapeutic and/or prophylactic agents can be at least 50%, eg, 50%, 55%, 60%, 65%, 70% %, 75%, 80%, 85%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99% or 100%. In some embodiments, the encapsulation efficiency can be at least 80%. In certain embodiments, the encapsulation efficiency can be at least 90%. In some embodiments, the encapsulation efficiency of the therapeutic and/or prophylactic agent is between 80% and 100%.
药物组合物pharmaceutical composition
可以将脂质纳米粒子(例如,空LNP或负载LNP)全部或部分地配制为药物组合物。药物组合物可以包含一种或多种脂质纳米粒子(例如,空LNP或负载LNP)。在一个实施方案中,药物组合物包含脂质纳米粒子(例如,空LNP或负载LNP)的群体。例如,药物组合物可以包含一种或多种脂质纳米粒子(例如,空LNP或负载LNP),所述脂质纳米粒子包含一种或多种不同的治疗剂和/或预防剂。药物组合物还可以包含一种或多种药学上可接受的赋形剂或辅助成分,诸如本文所述的那些。关于药物组合物和剂的配制和制造的一般指南可在例如Remington’s The Science and Practice of Pharmacy,第21版,A.R.Gennaro;Lippincott,Williams&Wilkins,Baltimore,MD,2006中获得。常规赋形剂和辅助成分可用于任何药物组合物中,除非任何常规赋形剂或辅助成分可能与纳米粒子组合物的一种或多种组分不相容。如果赋形剂或辅助成分与脂质纳米粒子(例如空LNP或负载LNP)的组分的组合可能引起任何不希望的生物学作用或其他有害作用,则赋形剂或辅助成分可能与所述组分不相容。Lipid nanoparticles (eg, empty LNPs or loaded LNPs) can be formulated in whole or in part into pharmaceutical compositions. The pharmaceutical composition can include one or more lipid nanoparticles (eg, empty LNPs or loaded LNPs). In one embodiment, the pharmaceutical composition comprises a population of lipid nanoparticles (eg, empty LNPs or loaded LNPs). For example, a pharmaceutical composition can include one or more lipid nanoparticles (eg, empty LNPs or loaded LNPs) that include one or more different therapeutic and/or prophylactic agents. The pharmaceutical compositions may also contain one or more pharmaceutically acceptable excipients or auxiliary ingredients, such as those described herein. General guidance on the formulation and manufacture of pharmaceutical compositions and agents is available, for example, in Remington's The Science and Practice of Pharmacy, 21st Edition, A.R. Gennaro; Lippincott, Williams & Wilkins, Baltimore, MD, 2006. Conventional excipients and adjunct ingredients can be used in any pharmaceutical composition, except that any conventional excipients or adjunct ingredients may be incompatible with one or more components of the nanoparticle composition. Excipients or adjuncts may be associated with the components of the lipid nanoparticle (eg empty LNP or LNP-loaded) if their combination may cause any undesired biological or other detrimental effects. Components are incompatible.
在一些实施方案中,一种或多种赋形剂或辅助成分可以占包含纳米粒子组合物的药物组合物的总质量或体积的超过50%。例如,一种或多种赋形剂或辅助成分可以占药物常规的50%、60%、70%、80%、90%或更多。在一些实施方案中,药学上可接受的赋形剂是至少95%、至少96%、至少97%、至少98%、至少99%或100%纯的。在一些实施方案中,赋形剂被批准用于人类和兽医用途。在一些实施方案中,赋形剂由美国食品药品管理局批准。在一些实施方案中,赋形剂为药品级。在一些实施方案中,赋形剂符合美国药典(USP)、欧洲药典(EP)、英国药典和/或国际药典的标准。In some embodiments, one or more excipients or adjunct ingredients may constitute more than 50% of the total mass or volume of the pharmaceutical composition comprising the nanoparticle composition. For example, one or more excipients or adjunct ingredients may constitute 50%, 60%, 70%, 80%, 90%, or more of the pharmaceutical conventional. In some embodiments, the pharmaceutically acceptable excipient is at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% pure. In some embodiments, the excipients are approved for human and veterinary use. In some embodiments, the excipient is approved by the US Food and Drug Administration. In some embodiments, the excipient is pharmaceutical grade. In some embodiments, the excipient meets the standards of the United States Pharmacopoeia (USP), European Pharmacopoeia (EP), British Pharmacopoeia, and/or International Pharmacopoeia.
根据本公开的药物组合物中所述一种或多种脂质纳米粒子(例如,空LNP或负载LNP)、所述一种或多种药学上可接受的赋形剂和/或任何附加成分的相对量将根据所治疗的受试者的身份、体型和/或状况并且还根据施用组合物的途径而变化。举例来说,药物组合物可以包含0.1%与100%(重量/重量)之间的一种或多种脂质纳米粒子(例如,空LNP或负载LNP)。The one or more lipid nanoparticles (eg, empty LNP or loaded LNP), the one or more pharmaceutically acceptable excipients and/or any additional ingredients in the pharmaceutical composition according to the present disclosure The relative amounts of s will vary according to the identity, size and/or condition of the subject being treated and also according to the route of administration of the composition. For example, a pharmaceutical composition may comprise between 0.1% and 100% (weight/weight) of one or more lipid nanoparticles (eg, empty LNPs or loaded LNPs).
在某些实施方案中,本公开的脂质纳米粒子(例如,空LNP或负载LNP)和/或药物组合物被冷藏或冷冻储存和/或运输(例如,储存在4℃或更低温度下,诸如介于约-150℃与约0℃之间或介于约-80℃与约-20℃之间的温度(例如,约-5℃、-10℃、-15℃、-20℃、-25℃、-30℃、-40℃、-50℃、-60℃、-70℃、-80℃、-90℃、-130℃或-150℃))。例如,包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)中的任一个的化合物的药物组合物是溶液,其在例如约-20℃、-30℃、-40℃、-50℃、-60℃、-70℃、-80℃下冷冻储存和/或运输。在某些实施方案中,本公开还涉及一种增加包含式(1-1)、(2-1)、(I-a)、(A)、(B)、(A-1)、(A-2)、(A-3)、(IA)、(IB)、(B-1)、(B-2)、(B-3)、(A-a)、(A-a1)、(A-a2)、(A-a3)、(A-b)、(A-b1)、(A-b2)、(A-b3)、(A-c)和(B-c)中的任一个的化合物的脂质纳米粒子(例如,空LNP或负载LNP)和/或药物组合物的稳定性的方法,所述方法通过将脂质纳米粒子(例如,空LNP或负载LNP)和/或药物组合物储存在4℃或更低温度下,诸如介于约-150℃与约0℃之间或介于约-80℃与约-20℃之间,例如,约-5℃、-10℃、-15℃、-20℃、-25℃、-30℃、-40℃、-50℃、-60℃、-70℃、-80℃、-90℃、-130℃或-150℃)的温度下来实现。例如,本文公开的脂质纳米粒子(例如,空LNP或负载LNP)和/或药物组合物例如在4℃或更低(例如,介于约4℃与-20℃之间)的温度下稳定保持约至少1周、至少2周、至少3周、至少4周、至少5周、至少6周、至少1个月、至少2个月、至少4个月、至少6个月、至少8个月、至少10个月、至少12个月、至少14个月、至少16个月、至少18个月、至少20个月、至少22个月或至少24个月。在一些实施方案中,制剂在约4℃下稳定保持至少4周。在某些实施方案中,本公开的药物组合物包含本文公开的脂质纳米粒子(例如,空LNP或负载LNP)和药学上可接受的载剂,所述载剂选自Tris、乙酸盐(例如,乙酸钠)、柠檬酸盐(例如,柠檬酸钠)、生理盐水、PBS和蔗糖中的一种或多种。在某些实施方案中,本公开的药物组合物的pH值介于约7与8之间(例如6.8、6.9、7.0、7.1、7.2、7.3、7.4、7.5、7.6、7.7、7.8、7.9或8.0,或介于7.5与8之间或介于7与7.8之间)。例如,本公开的药物组合物包含本文公开的脂质纳米粒子(例如,空LNP或负载LNP)、Tris、生理盐水和蔗糖,并且pH为约7.5-8,其适合于在例如约-20℃下储存和/或运输。例如,本公开的药物组合物包含本文公开的脂质纳米粒子(例如,空LNP或负载LNP)和PBS,并且pH是约7-7.8,其适合于在例如约4℃或更低温度下储存和/或运输。在本公开的上下文中,“稳定性”、“稳定化的”和“稳定的”是指本文公开的脂质纳米粒子(例如,空LNP或负载LNP)和/或药物组合物在给定制造、制备、运输、储存和/或使用条件下,例如当施加应力诸如剪切力、冷冻/解冻应力等时,对化学或物理变化(例如降解、粒度变化、聚集、包封变化等)的抗性。In certain embodiments, lipid nanoparticles (eg, empty LNPs or loaded LNPs) and/or pharmaceutical compositions of the present disclosure are refrigerated or frozen for storage and/or transport (eg, stored at 4°C or lower) , such as a temperature between about -150°C and about 0°C or between about -80°C and about -20°C (eg, about -5°C, -10°C, -15°C, -20°C, - 25°C, -30°C, -40°C, -50°C, -60°C, -70°C, -80°C, -90°C, -130°C or -150°C)). For example, including formula (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y), (I-Ya) (IA), A pharmaceutical composition of a compound of any one of (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb) is Solutions, which are stored and/or shipped frozen, eg, at about -20°C, -30°C, -40°C, -50°C, -60°C, -70°C, -80°C. In certain embodiments, the present disclosure also relates to a method that adds formulas (1-1), (2-1), (I-a), (A), (B), (A-1), (A-2) ), (A-3), (IA), (IB), (B-1), (B-2), (B-3), (A-a), (A-a1), (A-a2), Lipid nanoparticles (eg, empty A method of stability of LNP or loaded LNP) and/or pharmaceutical composition by storing lipid nanoparticles (eg, empty LNP or loaded LNP) and/or pharmaceutical composition at 4°C or lower , such as between about -150°C and about 0°C or between about -80°C and about -20°C, eg, about -5°C, -10°C, -15°C, -20°C, -25°C , -30°C, -40°C, -50°C, -60°C, -70°C, -80°C, -90°C, -130°C or -150°C). For example, lipid nanoparticles (eg, empty LNPs or loaded LNPs) and/or pharmaceutical compositions disclosed herein are stable, eg, at temperatures of 4°C or less (eg, between about 4°C and -20°C) For about at least 1 week, at least 2 weeks, at least 3 weeks, at least 4 weeks, at least 5 weeks, at least 6 weeks, at least 1 month, at least 2 months, at least 4 months, at least 6 months, at least 8 months , at least 10 months, at least 12 months, at least 14 months, at least 16 months, at least 18 months, at least 20 months, at least 22 months, or at least 24 months. In some embodiments, the formulation is stable at about 4°C for at least 4 weeks. In certain embodiments, a pharmaceutical composition of the present disclosure comprises a lipid nanoparticle disclosed herein (eg, empty LNP or loaded LNP) and a pharmaceutically acceptable carrier selected from Tris, acetate (eg, sodium acetate), one or more of citrate (eg, sodium citrate), saline, PBS, and sucrose. In certain embodiments, the pH of the pharmaceutical compositions of the present disclosure is between about 7 and 8 (eg, 6.8, 6.9, 7.0, 7.1, 7.2, 7.3, 7.4, 7.5, 7.6, 7.7, 7.8, 7.9 or 8.0, or between 7.5 and 8 or between 7 and 7.8). For example, the pharmaceutical compositions of the present disclosure comprise lipid nanoparticles disclosed herein (eg, empty LNPs or loaded LNPs), Tris, normal saline, and sucrose, and have a pH of about 7.5-8, which is suitable for use at, eg, about -20°C storage and/or transport. For example, a pharmaceutical composition of the present disclosure comprises a lipid nanoparticle disclosed herein (eg, empty LNP or loaded LNP) and PBS, and has a pH of about 7-7.8, which is suitable for storage at, eg, about 4°C or lower. and/or transportation. In the context of the present disclosure, "stable," "stabilized," and "stable" refer to the lipid nanoparticles (eg, empty LNPs or loaded LNPs) and/or pharmaceutical compositions disclosed herein in a given manufacture resistance to chemical or physical changes (eg, degradation, particle size changes, aggregation, encapsulation changes, etc.) sex.
在一些实施方案中,本公开的药物组合物包含空LNP或负载LNP、冷冻保护剂、缓冲剂或其组合。In some embodiments, the pharmaceutical compositions of the present disclosure comprise empty LNP or loaded LNP, a cryoprotectant, a buffer, or a combination thereof.
在一些实施方案中,冷冻保护剂包括一种或多种冷冻保护剂,并且一种或多种冷冻保护剂中的每一种独立地是多元醇(例如,二醇或三醇,诸如丙二醇(即,1,2-丙二醇),1,3-丙二醇、甘油、(+/-)-2-甲基-2,4-戊二醇、1,6-己二醇、1,2-丁二醇、2,3-丁二醇、乙二醇或二甘醇)、非洗涤剂的磺基甜菜碱(例如,NDSB-201(3-(1-吡啶并)-1-丙烷磺酸盐)、渗透剂(例如,L-脯氨酸或三甲胺N-氧化物二水合物)、聚合物(例如,聚乙二醇200(PEG 200)、PEG 400、PEG 600、PEG 1000、PEG2k-DMG、PEG 3350、PEG 4000、PEG 8000、PEG 10000、PEG20000、聚乙二醇单甲醚550(mPEG 550)、mPEG 600、mPEG 2000、mPEG 3350、mPEG 4000、mPEG5000、聚乙烯吡咯烷酮(例如聚乙烯吡咯烷酮K15)、季戊四醇丙氧基化物或聚丙二醇P400)、有机溶剂(例如,二甲亚砜(DMSO)或乙醇)、糖(例如,D-(+)-蔗糖、D-山梨糖醇、海藻糖、D-(+)-麦芽糖一水合物、内赤藓醇、木糖醇、肌醇、D-(+)-棉子糖五水合物、D-(+)-海藻糖二水合物或D-(+)-葡萄糖一水合物),或盐(例如,乙酸锂、氯化锂、甲酸锂、硝酸锂、硫酸锂、乙酸镁、乙酸钠、氯化钠、甲酸钠、丙二酸钠、硝酸钠、硫酸钠或其任何水合物)或其任何组合。在一些实施方案中,冷冻保护剂包括蔗糖。并且在一些实施方案中,冷冻保护剂和/或赋形剂是蔗糖。在一些实施方案中,冷冻保护剂包括乙酸钠。并且在一些实施方案中,冷冻保护剂和/或赋形剂是乙酸钠。在一些实施方案中,冷冻保护剂包括蔗糖和乙酸钠。In some embodiments, the cryoprotectant includes one or more cryoprotectants, and each of the one or more cryoprotectants is independently a polyol (eg, a diol or triol, such as propylene glycol ( i.e., 1,2-propanediol), 1,3-propanediol, glycerol, (+/-)-2-methyl-2,4-pentanediol, 1,6-hexanediol, 1,2-butanediol alcohol, 2,3-butanediol, ethylene glycol, or diethylene glycol), non-detergent sulfobetaines (eg, NDSB-201 (3-(1-pyrido)-1-propanesulfonate) , penetrants (eg, L-proline or trimethylamine N-oxide dihydrate), polymers (eg, polyethylene glycol 200 (PEG 200), PEG 400, PEG 600, PEG 1000, PEG 2k- DMG, PEG 3350, PEG 4000, PEG 8000, PEG 10000, PEG20000, polyethylene glycol monomethyl ether 550 (mPEG 550), mPEG 600, mPEG 2000, mPEG 3350, mPEG 4000, mPEG5000, polyvinylpyrrolidone (e.g. polyvinylpyrrolidone) Pyrrolidone K15), pentaerythritol propoxylate or polypropylene glycol P400), organic solvents (eg, dimethyl sulfoxide (DMSO) or ethanol), sugars (eg, D-(+)-sucrose, D-sorbitol, seaweed) sugar, D-(+)-maltose monohydrate, endoerythritol, xylitol, inositol, D-(+)-raffinose pentahydrate, D-(+)-trehalose dihydrate or D-(+)-glucose monohydrate), or salts (e.g., lithium acetate, lithium chloride, lithium formate, lithium nitrate, lithium sulfate, magnesium acetate, sodium acetate, sodium chloride, sodium formate, sodium malonate, sodium nitrate, sodium sulfate, or any hydrate thereof) or any combination thereof. In some embodiments, the cryoprotectant includes sucrose. And in some embodiments, the cryoprotectant and/or excipient is sucrose. In some implementations In embodiments, the cryoprotectant includes sodium acetate. And in some embodiments, the cryoprotectant and/or excipient is sodium acetate. In some embodiments, the cryoprotectant includes sucrose and sodium acetate.
在一些实施方案中,其中缓冲剂选自由以下组成的组:乙酸盐缓冲剂、柠檬酸盐缓冲剂、磷酸盐缓冲剂、tris缓冲剂及其组合。In some embodiments, wherein the buffer is selected from the group consisting of acetate buffer, citrate buffer, phosphate buffer, tris buffer, and combinations thereof.
可以将脂质纳米粒子(例如,空LNP或负载LNP)和/或包含一种或多种脂质纳米粒子(例如,空LNP或负载LNP)的药物组合物施用至任何患者或受试者,包括可以得益于通过将治疗剂和/或预防剂递送至一种或多种特定细胞、组织、器官或系统或其组所提供的治疗效果的患者或受试者。尽管本文提供的关于脂质纳米粒子(例如,空LNP或负载LNP)和包含脂质纳米粒子(例如,空LNP或负载LNP)的药物组合物的描述主要针对适于施用至人类的组合物,但熟练技术人员应了解,此类组合物一般适于施用至任何其他哺乳动物。应充分了解,可以对适于施用至人的组合物进行修改以便使所述组合物适于施用至各种动物,并且普通熟练兽医药理学家只需要常规实验(如果存在的话)就可以设计和/或进行此类修改。预期施用所述组合物的受试者包括但不限于人、其他灵长类动物和其他哺乳动物,包括商业相关的哺乳动物,诸如牛、猪、马、绵羊、猫、狗、小鼠和/或大鼠。本发明的脂质纳米粒子也可用于体外和离体用途。Lipid nanoparticles (eg, empty LNPs or loaded LNPs) and/or pharmaceutical compositions comprising one or more lipid nanoparticles (eg, empty LNPs or loaded LNPs) can be administered to any patient or subject, Included are patients or subjects who may benefit from the therapeutic effect provided by the delivery of therapeutic and/or prophylactic agents to one or more specific cells, tissues, organs or systems or groups thereof. Although the descriptions provided herein with respect to lipid nanoparticles (eg, empty LNPs or loaded LNPs) and pharmaceutical compositions comprising lipid nanoparticles (eg, empty LNPs or loaded LNPs) are primarily directed to compositions suitable for administration to humans, However, the skilled artisan will appreciate that such compositions are generally suitable for administration to any other mammal. It is fully understood that compositions suitable for administration to humans can be modified to make the compositions suitable for administration to a variety of animals, and can be devised and devised by the ordinarily skilled veterinary pharmacologist with only routine experimentation, if any. /or make such modifications. Subjects to which the compositions are expected to be administered include, but are not limited to, humans, other primates and other mammals, including commercially relevant mammals such as cattle, pigs, horses, sheep, cats, dogs, mice and/or or rats. The lipid nanoparticles of the present invention can also be used in in vitro and ex vivo applications.
包含一种或多种脂质纳米粒子(例如,空LNP或负载LNP)的药物组合物可以通过药理学领域已知的或以后将开发的任何方法制备。一般而言,此类制备方法包括使活性成分与赋形剂和/或一种或多种其他辅助成分缔合,然后在需要或必要时,将产物分割、成型和/或包装成所需的单一剂量单位或多剂量单位。Pharmaceutical compositions comprising one or more lipid nanoparticles (eg, empty LNPs or loaded LNPs) can be prepared by any method known in the art of pharmacology or later developed. In general, such methods of preparation involve bringing into association the active ingredient with excipients and/or one or more other accessory ingredients, and then dividing, shaping and/or packaging the product as desired or necessary into the desired Single-dose unit or multiple-dose unit.
根据本公开的药物组合物可以以单一单位剂量和/或以多个单一单位剂量形式批量制备、包装和/或销售。如本文所用,“单位剂量”是包含预定量的活性成分(例如,纳米粒子组合物)的药物组合物的离散量。活性成分的量一般等于将要向受试者施用的活性成分的剂量和/或这种剂量的合宜分数,例如像这种剂量的一半或三分之一。Pharmaceutical compositions according to the present disclosure may be prepared, packaged and/or sold in bulk in a single unit dose and/or in a plurality of single unit doses. As used herein, a "unit dose" is a discrete quantity of a pharmaceutical composition containing a predetermined quantity of an active ingredient (eg, a nanoparticle composition). The amount of active ingredient is generally equal to the dose of active ingredient to be administered to the subject and/or a suitable fraction of such dose, such as, for example, one-half or one-third of such dose.
药物组合物可以制备成适用于多种施用途径和方法的多种形式。例如,药物组合物可以被制备成液体剂型(例如,乳剂、微乳剂、纳米乳剂、溶液、混悬剂、糖浆和酏剂)、可注射剂型、固体剂型(例如,胶囊、片剂、丸剂、散剂和颗粒剂)、用于局部和/或透皮施用的剂型(例如,油膏、糊剂、乳膏、洗液、凝胶、散剂、溶液、喷雾剂、吸入剂和贴剂)、混悬剂、散剂和其他形式。Pharmaceutical compositions can be prepared in a variety of forms suitable for various routes and methods of administration. For example, pharmaceutical compositions can be prepared in liquid dosage forms (eg, emulsions, microemulsions, nanoemulsions, solutions, suspensions, syrups, and elixirs), injectable dosage forms, solid dosage forms (eg, capsules, tablets, pills, powders and granules), dosage forms for topical and/or transdermal administration (eg, ointments, pastes, creams, lotions, gels, powders, solutions, sprays, inhalants, and patches), mixed Suspensions, powders and other forms.
用于口服和肠胃外施用的液体剂型包括但不限于药学上可接受的乳剂、微乳剂、纳米乳剂、溶液、混悬剂、糖浆和/或酏剂。除了活性成分之外,液体剂型还可以包含本领域中常用的惰性稀释剂,例如像水或其他溶剂;增溶剂和乳化剂,诸如乙醇、异丙醇、碳酸乙酯、乙酸乙酯、苄醇、苯甲酸苄酯、丙二醇、1,3-丁二醇、二甲基甲酰胺、油(特别是棉籽油、花生油、玉米油、胚芽油、橄榄油、蓖麻油和芝麻油)、甘油、四氢糠醇、聚乙二醇和脱水山梨糖醇的脂肪酸酯及其混合物。除了惰性稀释剂之外,口服组合物还可以包含另外的治疗剂和/或预防剂、另外的剂诸如润湿剂、乳化剂和悬浮剂、甜味剂、调味剂和/或芳香剂。在用于肠胃外施用的某些实施方案中,将组合物与增溶剂诸如醇、油、改性油、二醇、聚山梨醇酯、环糊精、聚合物和/或其组合混合。Liquid dosage forms for oral and parenteral administration include, but are not limited to, pharmaceutically acceptable emulsions, microemulsions, nanoemulsions, solutions, suspensions, syrups and/or elixirs. In addition to the active ingredient, liquid dosage forms may also contain inert diluents commonly used in the art, such as, for example, water or other solvents; solubilizers and emulsifiers, such as ethanol, isopropanol, ethyl carbonate, ethyl acetate, benzyl alcohol , benzyl benzoate, propylene glycol, 1,3-butanediol, dimethylformamide, oils (especially cottonseed oil, peanut oil, corn oil, germ oil, olive oil, castor oil and sesame oil), glycerin, tetrahydro Fatty acid esters of furfuryl alcohol, polyethylene glycol and sorbitan and mixtures thereof. Besides inert diluents, the oral compositions can also contain additional therapeutic and/or prophylactic agents, additional agents such as wetting agents, emulsifying and suspending agents, sweetening, flavoring and/or perfuming agents. In certain embodiments for parenteral administration, the composition is combined with a solubilizer such as Alcohols, oils, modified oils, glycols, polysorbates, cyclodextrins, polymers, and/or combinations thereof are mixed.
可注射制品,例如无菌可注射水性或油性混悬剂可根据已知技术使用合适的分散剂、润湿剂和/或悬浮剂配制。无菌可注射制品可以是非毒性肠胃外可接受的稀释剂和/或溶剂中的无菌可注射溶液、混悬剂和/或乳剂,例如在1,3-丁二醇中的溶液。可采用的可接受的媒介物和溶剂为水、林格氏溶液(U.S.P.)和等渗氯化钠溶液。无菌不挥发性油常规用作溶剂或悬浮介质。出于此目的,可以采用任何温和的不挥发性油,包括合成的甘油单酯或甘油二酯。脂肪酸诸如油酸可用于制备注射剂。Injectable preparations, such as sterile injectable aqueous or oily suspensions, can be formulated according to the known art using suitable dispersing, wetting and/or suspending agents. The sterile injectable preparation can be a sterile injectable solution, suspension and/or emulsion in a non-toxic parenterally acceptable diluent and/or solvent, for example as a solution in 1,3-butanediol. Among the acceptable vehicles and solvents that may be employed are water, Ringer's solution (U.S.P.) and isotonic sodium chloride solution. Sterile, fixed oils are conventionally employed as a solvent or suspending medium. For this purpose any bland fixed oil may be employed including synthetic mono- or diglycerides. Fatty acids such as oleic acid are useful in the preparation of injectables.
可注射制剂可以例如通过细菌滞留的过滤器进行过滤和/或通过引入在使用之前可溶解或分散于无菌水或其他无菌可注射介质中的无菌固体组合物的形式的灭菌剂来灭菌。Injectable preparations can be prepared, for example, by filtration through a bacterial-retaining filter and/or by introducing a sterilizing agent in the form of a sterile solid composition which can be dissolved or dispersed in sterile water or other sterile injectable medium prior to use. Sterilize.
为了延长活性成分的作用,常常希望减缓对来自皮下注射或肌肉内注射的活性成分的吸收。这可以通过使用水溶性差的结晶或无定形材料的液体混悬剂来实现。药物的吸收速率则取决于其溶解速率,溶解速率继而又可以取决于晶体大小和结晶形式。可替代地,通过将药物溶解或悬浮于油媒介物中来实现肠胃外施用的药物形式的延迟吸收。可注射贮库形式通过在可生物降解聚合物诸如聚丙交酯-聚乙交酯中形成药物的微包封基质来制得。根据药物与聚合物的比率和所采用的特定聚合物的性质,可以控制药物释放的速率。其他可生物降解聚合物的实例包括聚(原酸酯)和聚(酸酐)。贮库可注射制剂通过将药物包埋在与身体组织相容的脂质体或微乳剂中来制备。In order to prolong the effect of the active ingredient, it is often desirable to slow the absorption of the active ingredient from subcutaneous or intramuscular injection. This can be accomplished by using liquid suspensions of poorly water-soluble crystalline or amorphous materials. The rate of absorption of a drug depends on its rate of dissolution, which in turn may depend on crystal size and crystalline form. Alternatively, delayed absorption of a parenterally administered drug form is accomplished by dissolving or suspending the drug in an oil vehicle. Injectable depot forms are prepared by forming microencapsulation matrices of the drug in biodegradable polymers such as polylactide-polyglycolide. Depending on the ratio of drug to polymer and the nature of the particular polymer employed, the rate of drug release can be controlled. Examples of other biodegradable polymers include poly(orthoesters) and poly(anhydrides). Depot injectable formulations are prepared by entrapping the drug in liposomes or microemulsions which are compatible with body tissues.
用于直肠或阴道施用的组合物通常是栓剂,所述栓剂可以通过将组合物与合适的非刺激性赋形剂诸如可可脂、聚乙二醇或栓剂蜡混合来制备,所述非刺激性赋形剂在环境温度下是固体但在体温下是液体,并因此在直肠或阴道腔内熔化并释放活性成分。Compositions for rectal or vaginal administration are usually suppositories, which may be prepared by mixing the composition with a suitable non-irritating excipient such as cocoa butter, polyethylene glycol, or a suppository wax. Excipients are solid at ambient temperature but liquid at body temperature and will therefore melt in the rectal or vaginal cavity and release the active ingredient.
用于口服施用的固体剂型包括胶囊、片剂、丸剂、膜剂、散剂和颗粒剂。在此类固体剂型中,活性成分与至少一种惰性的药学上可接受的赋形剂诸如柠檬酸钠或磷酸氢二钙和/或填充剂或增量剂(例如,淀粉、乳糖、蔗糖、葡萄糖、甘露糖醇以及硅酸)、粘结剂(例如,羧甲基纤维素、海藻酸盐、明胶、聚乙烯吡咯烷酮、蔗糖和阿拉伯树胶)、保湿剂(例如,甘油)、崩解剂(例如,琼脂、碳酸钙、马铃薯或木薯淀粉、海藻酸、某些硅酸盐和碳酸钠)、溶解延迟剂(例如,石蜡)、吸收促进剂(例如,季铵盐化合物)、润湿剂(例如,鲸蜡醇和单硬脂酸甘油酯)、吸附剂(例如,高岭土和膨润土、硅酸盐)和润滑剂(例如滑石、硬脂酸钙、硬脂酸镁、固体聚乙二醇、月桂基硫酸钠)及其混合物混合。在胶囊、片剂和丸剂的情况下,所述剂型可以包含缓冲剂。Solid dosage forms for oral administration include capsules, tablets, pills, films, powders and granules. In such solid dosage forms, the active ingredient is mixed with at least one inert pharmaceutically acceptable excipient such as sodium citrate or dibasic calcium phosphate and/or fillers or bulking agents (eg, starch, lactose, sucrose, glucose, mannitol, and silicic acid), binders (eg, carboxymethyl cellulose, alginates, gelatin, polyvinylpyrrolidone, sucrose, and acacia), humectants (eg, glycerol), disintegrants ( For example, agar, calcium carbonate, potato or tapioca starch, alginic acid, certain silicates and sodium carbonate), dissolution delaying agents (eg, paraffin), absorption enhancers (eg, quaternary ammonium compounds), wetting agents ( For example, cetyl alcohol and glycerol monostearate), adsorbents (for example, kaolin and bentonite, silicates) and lubricants (for example, talc, calcium stearate, magnesium stearate, solid polyethylene glycol, lauryl sodium sulfate) and mixtures thereof. In the case of capsules, tablets and pills, the dosage form may contain buffering agents.
相似类型的固体组合物可以用作软和硬填充明胶胶囊中的填充剂,所述胶囊使用诸如乳糖或奶糖以及高分子量聚乙二醇等的赋形剂。可以制备具有包衣和外壳诸如肠溶包衣和药物配制领域中众所周知的其他包衣的固体剂型片剂、糖衣丸、胶囊、丸剂及颗粒剂。它们可以任选地包含乳浊剂并且还可以具有使其只在或优先在肠道某一部分中任选地以延缓的方式释放活性成分的组成。可使用的包埋组合物的实例包括聚合物质和蜡。相似类型的固体组合物可以用作软和硬填充明胶胶囊中的填充剂,所述胶囊使用诸如乳糖或奶糖以及高分子量聚乙二醇等的赋形剂。Similar types of solid compositions can be used as fillers in soft and hard filled gelatin capsules using excipients such as lactose or milk sugar and high molecular weight polyethylene glycols and the like. The solid dosage forms of tablets, dragees, capsules, pills and granules can be prepared with coatings and shells such as enteric coatings and other coatings well known in the pharmaceutical formulation art. They may optionally contain opacifying agents and may also be of a composition that they release the active ingredient only or preferentially in a certain part of the intestinal tract, optionally in a delayed manner. Examples of embedding compositions that can be used include polymeric substances and waxes. Similar types of solid compositions can be used as fillers in soft and hard filled gelatin capsules using excipients such as lactose or milk sugar and high molecular weight polyethylene glycols and the like.
用于局部和/或透皮施用组合物的剂型可以包括油膏、糊剂、乳膏、洗液、凝胶、散剂、溶液、喷雾剂、吸入剂和/或贴剂。一般而言,活性成分在无菌条件下与药学上可接受的赋形剂和/或任何所需的防腐剂和/或可能需要的缓冲剂混合。另外,本公开预期使用透皮贴剂,所述经皮贴剂通常具有有对身体提供化合物的受控递送的附加优势。此类剂型可例如通过将化合物溶解和/或分散于适当的介质中来制备。可替代地或另外地,可以通过提供速率控制膜和/或通过将化合物分散于聚合物基质和/或凝胶中来控制速率。Dosage forms for topical and/or transdermal administration of the compositions may include ointments, pastes, creams, lotions, gels, powders, solutions, sprays, inhalants and/or patches. Generally, the active ingredient is mixed under sterile conditions with pharmaceutically acceptable excipients and/or any desired preservatives and/or buffers that may be required. Additionally, the present disclosure contemplates the use of transdermal patches, which generally have the added advantage of providing controlled delivery of compounds to the body. Such dosage forms can be prepared, for example, by dissolving and/or dispersing the compound in the proper medium. Alternatively or additionally, the rate can be controlled by providing a rate controlling membrane and/or by dispersing the compound in a polymer matrix and/or gel.
用于递送本文所述的皮内药物组合物的合适装置包括短针装置。皮内组合物可以通过限制针进入皮肤的有效穿透深度的装置来施用。喷射注射装置是合适的,所述装置通过液体喷射注射器和/或通过刺入角质层并产生到达真皮的喷射的针将液体组合物递送至真皮。弹道散剂/粒子递送装置是合适的,所述装置使用压缩气体加速散剂形式的疫苗穿过皮肤的外层到达真皮。可替代地或另外地,常规注射器可用于皮内施用的经典芒图氏(mantoux)方法中。Suitable devices for delivering the intradermal pharmaceutical compositions described herein include short needle devices. Intradermal compositions can be administered by means of devices that limit the effective depth of penetration of the needle into the skin. Jet injection devices are suitable which deliver the liquid composition to the dermis via a liquid jet injector and/or via a needle that penetrates the stratum corneum and produces a jet that reaches the dermis. Ballistic powder/particle delivery devices are suitable which use compressed gas to accelerate the vaccine in powder form across the outer layers of the skin to the dermis. Alternatively or additionally, conventional syringes can be used in the classical mantoux method of intradermal administration.
适用于表面施用的制剂包括但不限于液体和/或半液体制品诸如搽剂、洗剂、水包油和/或油包水乳剂诸如乳膏、油膏和/或糊剂,和/或溶液和/或混悬剂。例如,可局部施用的制剂可以包含约1%至约10%(重量/重量)活性成分,尽管活性成分的浓度可以高达所述活性成分在溶剂中的溶解度极限。用于局部施用的制剂还可以包含本文所述的附加成分中的一种或多种。Formulations suitable for topical application include, but are not limited to, liquid and/or semi-liquid preparations such as liniments, lotions, oil-in-water and/or water-in-oil emulsions such as creams, ointments and/or pastes, and/or solutions and/or suspension. For example, a topically administrable formulation may contain from about 1% to about 10% (weight/weight) active ingredient, although the active ingredient may be present in concentrations up to the solubility limit of the active ingredient in the solvent. Formulations for topical administration may also contain one or more of the additional ingredients described herein.
药物组合物可以呈适于通过口腔进行的肺施用的制剂形式来制备、包装和/或销售。这种制剂可以包含含有活性成分的干燥粒子。此类组合物方便地呈干粉形式,用于使用包含干粉储槽的装置施用,推进剂流可以引导所述干粉储槽分配散剂;和/或使用自推进溶剂/散剂分配容器施用,诸如在密封容器中包含溶解和/或悬浮于低沸点推进剂中的活性成分的装置。干粉组合物可以包括固体细粉稀释剂诸如糖,并且方便地以单位剂型提供。Pharmaceutical compositions can be prepared, packaged and/or sold in formulations suitable for pulmonary administration via the oral cavity. Such formulations may contain dry particles containing the active ingredient. Such compositions are conveniently in dry powder form for application using a device comprising a dry powder reservoir from which the propellant flow can be directed to dispense powder; and/or using a self-propelling solvent/powder dispensing container, such as in a sealed container. The container contains a device for dissolving and/or suspending the active ingredient in a low boiling point propellant. Dry powder compositions may include a solid fine powder diluent such as sugar, and are conveniently presented in unit dosage form.
低沸点推进剂一般包括在大环境压力下沸点低于65℉的液体推进剂。一般而言,推进剂可以占组合物的50%至99.9%(重量/重量),并且活性成分可以占组合物的0.1%至20%(重量/重量)。推进剂还可以包含附加成分,诸如液体非离子和/或固体阴离子表面活性剂和/或固体稀释剂(其可具有与包含活性成分的粒子相同数量级的粒度)。Low boiling point propellants generally include liquid propellants that boil below 65°F at large ambient pressures. In general, the propellant may comprise from 50% to 99.9% (w/w) of the composition and the active ingredient may comprise from 0.1% to 20% (w/w) of the composition. The propellant may also contain additional ingredients such as liquid nonionic and/or solid anionic surfactants and/or solid diluents (which may have a particle size of the same order of magnitude as the particles containing the active ingredient).
被配制用于肺部递送的药物组合物可以提供呈溶液和/或混悬剂的液滴形式的活性成分。此类制剂可呈任选无菌的、包含活性成分的水性和/或稀释醇性溶液和/或混悬剂形式来制备、包装和/或销售,并且可以使用任何喷雾和/或雾化装置方便地施用。此类制剂还可以包含一种或多种附加成分,其包括但不限于调味剂诸如糖精钠、挥发油、缓冲剂、表面活性剂和/或防腐剂诸如羟基苯甲酸甲酯。通过此施用途径提供的液滴的平均直径可以在约1nm至约200nm范围内。Pharmaceutical compositions formulated for pulmonary delivery may provide the active ingredient in the form of droplets of a solution and/or suspension. Such formulations may be prepared, packaged and/or sold as optionally sterile, aqueous and/or dilute alcoholic solutions and/or suspensions containing the active ingredient, and any spraying and/or atomizing device may be used. Easy to apply. Such formulations may also contain one or more additional ingredients including, but not limited to, flavoring agents such as sodium saccharin, volatile oils, buffers, surfactants and/or preservatives such as methyl hydroxybenzoate. The average diameter of the droplets provided by this route of administration can range from about 1 nm to about 200 nm.
本文描述为可用于肺部递送的制剂可用于鼻内递送药物组合物。适用于鼻内施用的另一种制剂是包含活性成分且平均粒子为约0.2μm至500μm的粗粉。这种制剂是以吸鼻烟方式施用,即通过鼻腔通道从靠近鼻的散剂容器快速吸入来施用。Formulations described herein as useful for pulmonary delivery can be used for intranasal delivery of pharmaceutical compositions. Another formulation suitable for intranasal administration is a coarse powder containing the active ingredient and having an average particle size of about 0.2 μm to 500 μm. This formulation is administered by snuff, ie by rapid inhalation through the nasal passages from a powder container near the nose.
适用于鼻腔施用的制剂可以例如包含少至约0.1%(重量/重量)和高达100%(重量/重量)的活性成分,并且可以包含本文所述的附加成分中的一种或多种。药物组合物可以呈适用于经颊施用的制剂形式来制备、包装和/或销售。此类制剂可以例如呈使用常规方法制备的片剂和/或锭剂的形式,并且可以包含例如0.1%至20%(重量/重量)的活性成分,其余部分包括口服可溶解和/或可降解的组合物和任选的本文所述的附加成分中的一种或多种。可替代地,适用于经颊施用的制剂可以包括包含活性成分的散剂和/或气雾化和/或雾化的溶液和/或混悬剂。此类粉末状、气雾化和/或雾化制剂在分散时可以具有在约0.1nm至约200nm范围内的平均粒子和/或液滴大小,并且还可以包含本文所述的任何附加成分中的一种或多种。Formulations suitable for nasal administration may, for example, contain as little as about 0.1% (w/w) and as high as 100% (w/w) active ingredient, and may contain one or more of the additional ingredients described herein. Pharmaceutical compositions can be prepared, packaged and/or sold in formulations suitable for buccal administration. Such formulations may, for example, be in the form of tablets and/or lozenges prepared using conventional methods, and may contain, for example, 0.1% to 20% (w/w) active ingredient, the balance comprising orally dissolvable and/or degradable The composition and optional one or more of the additional ingredients described herein. Alternatively, formulations suitable for buccal administration may include powders and/or aerosolized and/or aerosolized solutions and/or suspensions containing the active ingredient. Such powdered, aerosolized and/or aerosolized formulations, when dispersed, may have an average particle and/or droplet size in the range of about 0.1 nm to about 200 nm, and may also contain any of the additional ingredients described herein. one or more of.
药物组合物可以呈适用于眼部施用的制剂形式来制备、包装和/或销售。此类制剂可以例如呈滴眼剂形式,其包含例如活性成分于水性或油性液体赋形剂中的0.1/1.0%(重量/重量)溶液和/或混悬剂。此类滴剂还可以包含缓冲剂、盐和/或本文所述的任何附加成分中的一种或多种。可用的其他可眼部施用的制剂包括包含呈微晶形式的和/或在脂质体制品中的活性成分的那些。滴耳剂和/或滴眼剂预期在本公开的范围内。Pharmaceutical compositions can be prepared, packaged and/or sold in formulations suitable for ocular administration. Such formulations may, for example, be in the form of eye drops containing, for example, a 0.1/1.0% (w/w) solution and/or suspension of the active ingredient in an aqueous or oily liquid vehicle. Such drops may also contain one or more of buffers, salts, and/or any of the additional ingredients described herein. Other ophthalmically administrable formulations that can be used include those comprising the active ingredient in microcrystalline form and/or in a liposomal preparation. Ear drops and/or eye drops are intended to be within the scope of this disclosure.
mRNA疗法mRNA therapy
药物形态的mRNA具有递送分泌蛋白以及细胞内蛋白和跨膜蛋白的潜力。药物形态的mRNA具有具有递送跨膜蛋白和细胞内蛋白的潜力,即标准生物制剂在以蛋白质形式递送时因其无法跨过细胞膜而无法接近的靶标。使基于mRNA的疗法成为现实的一个主要挑战是最佳递送媒介物的确定。由于mRNA具有较大的大小、化学不稳定性和潜在的免疫原性,因此mRNA需要一种能够提供保护使其不受内切和外切核酸酶影响以及防护货物不受免疫哨兵攻击的递送媒介物。脂质纳米粒子(LNP)在这方面已被确定为主要选择。mRNA in drug form has the potential to deliver secreted proteins as well as intracellular and transmembrane proteins. mRNA in drug form has the potential to deliver transmembrane and intracellular proteins, targets that are inaccessible to standard biologics when delivered in protein form due to their inability to cross the cell membrane. A major challenge in making mRNA-based therapies a reality is the identification of optimal delivery vehicles. Due to the large size, chemical instability and potential immunogenicity of mRNA, mRNA requires a delivery vehicle that can provide protection from endo- and exonuclease and protect the cargo from attack by immune sentinels thing. Lipid nanoparticles (LNPs) have been identified as the primary choice in this regard.
脂质纳米粒子递送体系的关键性能标准是使细胞摄取最大化并使mRNA能够从核内体高效释放。在一个实施方案中,包含本文公开的新型脂质的本发明LNP表现出在细胞摄取和核内体释放中的至少一个方面的改善。同时,所述LNP必须提供稳定的药物产品,并且能够在治疗相关水平上安全地给药。LNP是多组分体系,其通常由氨基脂质、磷脂、胆固醇和PEG-脂质组成。每种成分对于核酸货物的高效递送和粒子稳定性都是必需的。被认为驱动细胞摄取、核内体逃逸和耐受性的关键组分是氨基脂质。胆固醇和PEG-脂质有助于药物产品在体内稳定性和搁置稳定性,而磷脂提供与LNP的额外融合性,因此有助于驱动核内体逃逸并使核酸在细胞溶质中可生物利用。A key performance criterion for lipid nanoparticle delivery systems is to maximize cellular uptake and enable efficient mRNA release from endosomes. In one embodiment, the LNPs of the invention comprising the novel lipids disclosed herein exhibit improvements in at least one of cellular uptake and endosomal release. At the same time, the LNP must provide a stable drug product that can be safely administered at therapeutically relevant levels. LNPs are multi-component systems that typically consist of amino lipids, phospholipids, cholesterol, and PEG-lipids. Each component is necessary for efficient delivery and particle stability of nucleic acid cargo. The key components thought to drive cellular uptake, endosomal escape and tolerance are amino lipids. Cholesterol and PEG-lipids contribute to in vivo stability and shelf stability of the drug product, while phospholipids provide additional fusion with LNPs and thus help drive endosomal escape and make nucleic acids bioavailable in the cytosol.
在过去数十年里,已经开发出若干用于寡核苷酸递送的氨基脂质系列,包括氨基脂质MC3(DLin-MC3-DMA)。基于MC3的LNP已被证明可有效递送mRNA。这类LNP在静脉内递送时被载脂蛋白E(ApoE)快速调理,使其能够被低密度脂蛋白受体(LDLr)细胞摄取。然而,人们仍然担心MC3的长组织半衰期可能会导致不良副作用,从而阻碍其用于慢性疗法。此外,大量文献证据表明,脂质纳米粒子的长期给药会产生若干毒副作用,包括补体激活相关的类过敏反应(CARPA)和肝损伤。因此,为了释放基于mRNA和其他核酸、核苷酸或肽的人类疗法的潜力,需要一类具有提高的递送效率以及使其能够在人类中长期给药的代谢和毒性特征的LNP。Over the past few decades, several aminolipid families have been developed for oligonucleotide delivery, including the aminolipid MC3 (DLin-MC3-DMA). MC3-based LNPs have been shown to deliver mRNA efficiently. Such LNPs are rapidly opsonized by apolipoprotein E (ApoE) upon intravenous delivery, enabling their uptake by low-density lipoprotein receptor (LDLr) cells. However, concerns remain that the long tissue half-life of MC3 may lead to undesirable side effects, preventing its use in chronic therapy. Furthermore, extensive literature evidence suggests that long-term administration of lipid nanoparticles produces several toxic side effects, including complement activation-associated anaphylactoid reactions (CARPA) and liver damage. Therefore, to unlock the potential of mRNA and other nucleic acid, nucleotide or peptide-based therapeutics in humans, a class of LNPs with improved delivery efficiency as well as metabolic and toxicity profiles that enable long-term administration in humans is required.
治疗众多疾病的能力需要在不同剂量水平下长期安全给药的灵活性。通过氨基脂质结构的系统性优化,将本公开化合物确定为兼顾化学稳定性、由于核内体逃逸改善而提高的递送效率、快速的体内代谢和干净的毒性特征的化合物。这些特征的组合将提供可以长期给药而不会激活免疫系统的药物候选物。初始啮齿动物筛选使得确定具有良好递送效率和药代动力学的先导脂质。在非人灵长类动物中进一步分析单次和重复给药后先导LNP的递送效率。最后,在一个月的重复剂量毒性研究中,在大鼠和非人灵长类动物中评价优化的LNP。不希望受理论束缚,本公开的新型可离子化脂质具有改善的细胞递送、改善的蛋白质表达和改善的可生物降解特性,这可导致与缺乏本发明脂质的LNP相比,细胞中的mRNA表达增加超过2倍、5倍、10倍、15倍或20倍。在另一个实施方案中,与其他细胞类型相比,包含本发明的脂质的LNP可导致特异性(例如,优先)递送至某一种或多种细胞类型,从而导致与缺乏本发明的脂质的LNP相比,某些细胞或组织中mRNA表达增加超过2倍、5倍、10倍、15倍或20倍。这些对现有技术的改善允许在急性和慢性疾病中安全且有效地使用基于mRNA的疗法。The ability to treat a wide range of diseases requires the flexibility of long-term safe dosing at different dose levels. Through systematic optimization of aminolipid structures, the disclosed compounds were identified as compounds that balance chemical stability, enhanced delivery efficiency due to improved endosomal escape, rapid in vivo metabolism, and a clean toxicity profile. The combination of these features will provide drug candidates that can be administered chronically without activating the immune system. Initial rodent screening allowed the identification of lead lipids with good delivery efficiency and pharmacokinetics. The delivery efficiency of lead LNPs after single and repeated dosing was further analyzed in non-human primates. Finally, the optimized LNPs were evaluated in rats and non-human primates in a one-month repeated-dose toxicity study. Without wishing to be bound by theory, the novel ionizable lipids of the present disclosure have improved cellular delivery, improved protein expression, and improved biodegradability properties, which may result in higher levels of ionization in cells compared to LNPs lacking the lipids of the present invention. mRNA expression was increased by more than 2-fold, 5-fold, 10-fold, 15-fold or 20-fold. In another embodiment, LNPs comprising the lipids of the present invention can result in specific (eg, preferential) delivery to one or more cell types compared to other cell types, resulting in a In some cells or tissues, mRNA expression is increased by more than 2-fold, 5-fold, 10-fold, 15-fold, or 20-fold compared to high-quality LNPs. These improvements over existing technologies allow for the safe and effective use of mRNA-based therapies in acute and chronic diseases.
方法method
在一些方面,本公开提供了一种将治疗剂和/或预防剂递送至细胞(例如,哺乳动物细胞)的方法。这种方法包括使细胞与本公开的负载LNP或药物组合物接触,由此将所述治疗剂和/或预防剂递送至所述细胞的步骤。在一些实施方案中,所述细胞在受试者体内并且所述接触包括将细胞施用至受试者。在一些实施方案中,所述方法包括向受试者施用包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,磷脂,结构性脂质,PEG脂质以及一种或多种治疗剂和/或预防剂的脂质纳米粒子,由此将所述治疗剂和/或预防剂递送至所述细胞的步骤。In some aspects, the present disclosure provides a method of delivering therapeutic and/or prophylactic agents to cells (eg, mammalian cells). This method includes the step of contacting a cell with the LNP-loaded or pharmaceutical composition of the present disclosure, thereby delivering the therapeutic and/or prophylactic agent to the cell. In some embodiments, the cells are in the subject and the contacting comprises administering the cells to the subject. In some embodiments, the method comprises administering to the subject a compound comprising formula (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y), (I-Ya)(IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or ( Compounds of IIb), phospholipids, structured lipids, PEG lipids and lipid nanoparticles of one or more therapeutic and/or prophylactic agents, thereby delivering said therapeutic and/or prophylactic agents to said Cell steps.
在一些实施方案中,本公开提供了一种将治疗剂和/或预防剂递送至受试者体内的细胞的方法,其中所述方法包括向受试者施用包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,DSPC,胆固醇和PEG2k-DMG以及一种或多种选自核苷酸、多肽和核酸(例如,RNA)的治疗剂和/或预防剂的脂质纳米粒子的步骤。例如,在一些实施方案中,本公开提供了一种将治疗剂和/或预防剂递送至受试者体内的细胞的方法,其中所述方法包括向受试者施用包含式(A)的化合物、DSPC、胆固醇和PEG2k-DMG以及一种或多种选自核苷酸、多肽和核酸(例如,RNA)的治疗剂和/或预防剂的脂质纳米粒子的步骤。In some embodiments, the present disclosure provides a method of delivering a therapeutic and/or prophylactic agent to cells in a subject, wherein the method comprises administering to the subject a method comprising formula (A), (A1) , (A2), (A3), (B), (I), (I'), (IX), (IY), (I-Ya) (IA), (Ia), (Ib), (Ic) , (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb) compounds, DSPC, cholesterol and PEG 2k -DMG and one or more selected from nucleotides Steps of lipid nanoparticles of therapeutic and/or prophylactic agents, polypeptides, and nucleic acids (eg, RNA). For example, in some embodiments, the present disclosure provides a method of delivering a therapeutic and/or prophylactic agent to cells in a subject, wherein the method comprises administering to the subject a compound comprising formula (A) , DSPC, cholesterol and PEG 2k -DMG and one or more lipid nanoparticles selected from the group consisting of nucleotides, polypeptides and nucleic acid (eg, RNA) therapeutic and/or prophylactic agents.
在一些实施方案中,本公开提供了一种将治疗剂和/或预防剂递送至受试者体内的细胞的方法,其中所述方法包括向受试者施用包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,DSPC,胆固醇和PEG-1以及一种或多种选自核苷酸、多肽和核酸(例如,RNA)的治疗剂和/或预防剂的脂质纳米粒子的步骤。例如,在一些实施方案中,本公开提供了一种将治疗剂和/或预防剂递送至受试者体内的细胞的方法,其中所述方法包括向受试者施用包含式(A)的化合物、DSPC、胆固醇和PEG-1以及一种或多种选自核苷酸、多肽和核酸(例如,RNA)的治疗剂和/或预防剂的脂质纳米粒子的步骤。In some embodiments, the present disclosure provides a method of delivering a therapeutic and/or prophylactic agent to cells in a subject, wherein the method comprises administering to the subject a method comprising formula (A), (A1) , (A2), (A3), (B), (I), (I'), (I-X), (I-Y), (I-Ya) (IA), (Ia), (Ib), (Ic) , (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb) compounds, DSPC, cholesterol and PEG-1 and one or more selected from the group consisting of nucleotides, Lipid Nanoparticles of Polypeptides and Nucleic Acids (eg, RNA) for Therapeutic and/or Prophylactic Agents. For example, in some embodiments, the present disclosure provides a method of delivering a therapeutic and/or prophylactic agent to cells in a subject, wherein the method comprises administering to the subject a compound comprising formula (A) , DSPC, cholesterol and PEG-1 and one or more lipid nanoparticles selected from the group consisting of nucleotides, polypeptides and nucleic acid (eg, RNA) therapeutic and/or prophylactic agents.
在一些方面,本公开提供了一种将治疗和/或预防剂递送(例如,特异性递送)至哺乳动物器官或组织(例如,肝、肾、脾或肺)的方法。这种方法包括使所述细胞与本公开的负载LNP或药物组合物接触的步骤,由此将所述治疗剂和/或预防剂递送至所述靶器官或靶组织。在一些实施方案中,所述方法包括向受试者施用包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,磷脂,结构性脂质,PEG脂质以及一种或多种治疗剂和/或预防剂的脂质纳米粒子,由此将所述治疗剂和/或预防剂递送至所述靶器官或靶组织的步骤。In some aspects, the present disclosure provides a method of delivering (eg, specific delivery) a therapeutic and/or prophylactic agent to a mammalian organ or tissue (eg, liver, kidney, spleen, or lung). This method includes the step of contacting the cells with the LNP-loaded or pharmaceutical composition of the present disclosure, thereby delivering the therapeutic and/or prophylactic agent to the target organ or tissue. In some embodiments, the method comprises administering to the subject a compound comprising formula (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y), (I-Ya)(IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or ( Compounds of IIb), phospholipids, structured lipids, PEG lipids and lipid nanoparticles of one or more therapeutic and/or prophylactic agents, thereby delivering said therapeutic and/or prophylactic agents to said A step in a target organ or tissue.
在一些实施方案中,本公开提供了一种将治疗剂和/或预防剂特异性递送至受试者的器官的方法,其中所述方法包括向受试者施用包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,DSPC,胆固醇和PEG2k-DMG以及一种或多种选自核苷酸、多肽和核酸(例如,RNA)的治疗剂和/或预防剂的脂质纳米粒子的步骤。例如,在一些实施方案中,本公开提供了一种将治疗剂和/或预防剂特异性递送至受试者的器官的方法,其中所述方法包括向受试者施用包含式(A)的化合物、DSPC、胆固醇和PEG2k-DMG以及一种或多种选自核苷酸、多肽和核酸(例如,RNA)的治疗剂和/或预防剂的脂质纳米粒子的步骤。In some embodiments, the present disclosure provides a method of specifically delivering a therapeutic and/or prophylactic agent to an organ of a subject, wherein the method comprises administering to the subject a method comprising formula (A), (A1) ), (A2), (A3), (B), (I), (I'), (IX), (IY), (I-Ya) (IA), (Ia), (Ib), (Ic ), (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb) compounds, DSPC, cholesterol and PEG 2k -DMG and one or more selected from the group consisting of nucleosides Steps of lipid nanoparticles of therapeutic and/or prophylactic agents of acids, polypeptides and nucleic acids (eg, RNA). For example, in some embodiments, the present disclosure provides a method of specifically delivering a therapeutic and/or prophylactic agent to an organ of a subject, wherein the method comprises administering to the subject a compound comprising formula (A) The step of compound, DSPC, cholesterol and PEG 2k -DMG and one or more lipid nanoparticles selected from the group consisting of nucleotides, polypeptides and nucleic acid (eg, RNA) therapeutic and/or prophylactic agents.
在一些实施方案中,本公开提供了一种将治疗剂和/或预防剂递送至受试者的器官的方法,其中所述方法包括向受试者施用包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,DSPC,胆固醇和PEG-1以及一种或多种选自核苷酸、多肽和核酸(例如,RNA)的治疗剂和/或预防剂的脂质纳米粒子的步骤。例如,在一些实施方案中,本公开一种提供了将治疗剂和/或预防剂递送至受试者的器官的方法,其中所述方法包括向受试者施用包含式(A)的化合物、DSPC、胆固醇和PEG-1以及一种或多种选自核苷酸、多肽和核酸(例如,RNA)的治疗剂和/或预防剂的脂质纳米粒子的步骤。In some embodiments, the present disclosure provides a method of delivering a therapeutic and/or prophylactic agent to an organ of a subject, wherein the method comprises administering to the subject a method comprising formula (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y), (I-Ya) (IA), (Ia), (Ib), (Ic), Compounds of (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb), DSPC, cholesterol and PEG-1 and one or more selected from nucleotides, polypeptides and nucleic acid (eg, RNA) therapeutic and/or prophylactic lipid nanoparticles steps. For example, in some embodiments, the present disclosure provides a method of delivering a therapeutic and/or prophylactic agent to an organ of a subject, wherein the method comprises administering to the subject a compound comprising formula (A), The step of DSPC, cholesterol and PEG-1 and one or more lipid nanoparticles selected from the group consisting of nucleotides, polypeptides and nucleic acid (eg, RNA) therapeutic and/or prophylactic agents.
在一些方面,本公开的特征在于一种用于将治疗剂和/或预防剂(例如,mRNA)增强递送至靶组织(例如,肝、脾或肺)的方法。这种方法包括使所述细胞与本公开的负载LNP或药物组合物接触,由此将所述治疗剂和/或预防剂递送至所述靶组织(例如,肝、肾、脾或肺)的步骤。在一些实施方案中,所述方法包括向受试者施用包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,磷脂,结构性脂质,PEG脂质以及一种或多种治疗剂和/或预防剂的脂质纳米粒子,由此将所述治疗剂和/或预防剂递送至所述靶组织(例如,肝、肾、脾或肺)的步骤。In some aspects, the disclosure features a method for enhanced delivery of a therapeutic and/or prophylactic agent (eg, mRNA) to a target tissue (eg, liver, spleen, or lung). Such methods include contacting the cells with an LNP-loaded or pharmaceutical composition of the present disclosure, thereby delivering the therapeutic and/or prophylactic agents to the target tissue (eg, liver, kidney, spleen, or lung) step. In some embodiments, the method comprises administering to the subject a compound comprising formula (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y), (I-Ya)(IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or ( Compounds of IIb), phospholipids, structured lipids, PEG lipids and lipid nanoparticles of one or more therapeutic and/or prophylactic agents, thereby delivering said therapeutic and/or prophylactic agents to said Steps to target tissues (eg, liver, kidney, spleen, or lung).
在一些实施方案中,本公开提供了一种用于将治疗剂和/或预防剂增强递送至靶组织的方法,其中所述方法包括向受试者施用包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,DSPC,胆固醇和PEG2k-DMG以及一种或多种选自核苷酸、多肽和核酸(例如,RNA)的治疗剂和/或预防剂的脂质纳米粒子的步骤。例如,在一些实施方案中,本公开提供了一种用于将治疗剂和/或预防剂增强递送至靶组织的方法,其中所述方法包括向受试者施用包含式(A)的化合物、DSPC、胆固醇和PEG2k-DMG以及一种或多种选自核苷酸、多肽和核酸(例如,RNA)的治疗剂和/或预防剂的脂质纳米粒子的步骤。In some embodiments, the present disclosure provides a method for enhanced delivery of a therapeutic and/or prophylactic agent to a target tissue, wherein the method comprises administering to a subject a method comprising formula (A), (A1), (A2), (A3), (B), (I), (I'), (IX), (IY), (I-Ya) (IA), (Ia), (Ib), (Ic), A compound of (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb), DSPC, cholesterol and PEG 2k -DMG and one or more selected from the group consisting of nucleotides, Lipid Nanoparticles of Polypeptides and Nucleic Acids (eg, RNA) for Therapeutic and/or Prophylactic Agents. For example, in some embodiments, the present disclosure provides a method for enhanced delivery of a therapeutic and/or prophylactic agent to a target tissue, wherein the method comprises administering to a subject a compound comprising formula (A), A step of DSPC, cholesterol and PEG 2k -DMG and one or more lipid nanoparticles selected from the group consisting of nucleotides, polypeptides and nucleic acid (eg, RNA) therapeutic and/or prophylactic agents.
在一些实施方案中,本公开提供了一种用于将治疗剂和/或预防剂增强递送至靶组织的方法,其中所述方法包括向受试者施用包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,DSPC,胆固醇和PEG-1以及一种或多种选自核苷酸、多肽和核酸(例如,RNA)的治疗剂和/或预防剂的脂质纳米粒子的步骤。例如,在一些实施方案中,本公开提供了一种用于将治疗剂和/或预防剂增强递送至靶组织的方法,其中所述方法包括向受试者施用包含式(A)的化合物、DSPC、胆固醇和PEG-1以及一种或多种选自核苷酸、多肽和核酸(例如,RNA)的治疗剂和/或预防剂的脂质纳米粒子的步骤。In some embodiments, the present disclosure provides a method for enhanced delivery of a therapeutic and/or prophylactic agent to a target tissue, wherein the method comprises administering to a subject a method comprising formula (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y), (I-Ya) (IA), (Ia), (Ib), (Ic), Compounds of (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb), DSPC, cholesterol and PEG-1 and one or more selected from nucleotides, polypeptides and nucleic acid (eg, RNA) therapeutic and/or prophylactic lipid nanoparticles steps. For example, in some embodiments, the present disclosure provides a method for enhanced delivery of a therapeutic and/or prophylactic agent to a target tissue, wherein the method comprises administering to a subject a compound comprising formula (A), The step of DSPC, cholesterol and PEG-1 and one or more lipid nanoparticles selected from the group consisting of nucleotides, polypeptides and nucleic acid (eg, RNA) therapeutic and/or prophylactic agents.
在一些方面,本公开提供了一种在细胞(例如,哺乳动物细胞)中产生所关注的多肽的方法。这种方法包括使细胞与本公开的负载LNP或药物组合物接触的步骤,其中负载LNP或药物组合物包含mRNA,由此mRNA能够在所述细胞中翻译以产生多肽。在一些实施方案中,所述细胞在受试者体内并且所述接触包括将细胞施用至受试者。在一些实施方案中,所述方法包括向受试者施用包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,磷脂,结构性脂质,PEG脂质和mRNA的脂质纳米粒子的步骤,由此mRNA能够在细胞中翻译以产生多肽。In some aspects, the present disclosure provides a method of producing a polypeptide of interest in a cell (eg, a mammalian cell). This method includes the step of contacting a cell with a loaded LNP or pharmaceutical composition of the present disclosure, wherein the loaded LNP or pharmaceutical composition comprises mRNA, whereby the mRNA is capable of being translated in the cell to produce a polypeptide. In some embodiments, the cells are in the subject and the contacting comprises administering the cells to the subject. In some embodiments, the method comprises administering to the subject a compound comprising formula (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y), (I-Ya)(IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or ( The compound of IIb), the phospholipid, the structured lipid, the PEG lipid and the step of lipid nanoparticles of mRNA, whereby the mRNA can be translated in the cell to produce the polypeptide.
在一些实施方案中,本公开提供了一种在细胞中产生所关注的多肽的方法,其中所述方法包括向受试者施用包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,DSPC,胆固醇和PEG2k-DMG以及mRNA的脂质纳米粒子的步骤。例如,在一些实施方案中,本公开提供了一种在细胞中产生所关注的多肽的方法,其中所述方法包括向受试者施用包含表1的化合物、DSPC、胆固醇和PEG2k-DMG以及mRNA的脂质纳米粒子的步骤。例如,在一些实施方案中,本公开提供了一种在细胞中产生所关注的多肽的方法,其中所述方法包括向受试者施用包含式(A)的化合物、DSPC、胆固醇和PEG2k-DMG以及mRNA的脂质纳米粒子的步骤。In some embodiments, the present disclosure provides a method of producing a polypeptide of interest in a cell, wherein the method comprises administering to a subject a method comprising formula (A), (A1), (A2), (A3) , (B), (I), (I'), (IX), (IY), (I-Ya) (IA), (Ia), (Ib), (Ic), (Id), (Ie) Steps for lipid nanoparticles of compounds, (If), (Ig), (II), (IIa) or (IIb), DSPC, cholesterol and PEG 2k -DMG and mRNA. For example, in some embodiments, the present disclosure provides a method of producing a polypeptide of interest in a cell, wherein the method comprises administering to a subject a compound comprising Table 1, DSPC, cholesterol, and PEG 2k -DMG, and Steps for mRNA lipid nanoparticles. For example, in some embodiments, the present disclosure provides a method of producing a polypeptide of interest in a cell, wherein the method comprises administering to a subject a compound comprising formula (A), DSPC, cholesterol, and PEG 2k- Steps for DMG and mRNA lipid nanoparticles.
在一些实施方案中,本公开提供了一种在细胞中产生所关注的多肽的方法,其中所述方法包括向受试者施用包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,DSPC,胆固醇和PEG-1以及mRNA的脂质纳米粒子的步骤。例如,在一些实施方案中,本公开提供了一种在细胞中产生所关注的多肽的方法,其中所述方法包括向受试者施用包含式(A)的化合物、DSPC、胆固醇和PEG-1以及mRNA的脂质纳米粒子的步骤。例如,在一些实施方案中,本公开提供了一种在细胞中产生所关注的多肽的方法,其中所述方法包括向受试者施用包含表1的化合物、DSPC、胆固醇和PEG-1以及mRNA的脂质纳米粒子的步骤。In some embodiments, the present disclosure provides a method of producing a polypeptide of interest in a cell, wherein the method comprises administering to a subject a method comprising formula (A), (A1), (A2), (A3) , (B), (I), (I'), (I-X), (I-Y), (I-Ya) (IA), (Ia), (Ib), (Ic), (Id), (Ie) Steps for lipid nanoparticles of compounds, (If), (Ig), (II), (IIa) or (IIb), DSPC, cholesterol and PEG-1 and mRNA. For example, in some embodiments, the present disclosure provides a method of producing a polypeptide of interest in a cell, wherein the method comprises administering to a subject a compound comprising formula (A), DSPC, cholesterol, and PEG-1 and the steps of mRNA lipid nanoparticles. For example, in some embodiments, the present disclosure provides a method of producing a polypeptide of interest in a cell, wherein the method comprises administering to a subject a compound comprising Table 1, DSPC, cholesterol and PEG-1, and mRNA steps of lipid nanoparticles.
在一些方面,本公开提供了一种在有需要的哺乳动物(例如,人)中治疗疾病或病症的方法。所述方法包括向哺乳动物施用治疗有效量的本公开的负载LNP或药物组合物的步骤。在一些实施方案中,所述方法包括向受试者施用包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,磷脂,结构性脂质,PEG脂质以及一种或多种治疗剂和/或预防剂的脂质纳米粒子,由此将所述治疗剂和/或预防剂递送至所述细胞的步骤。在一些实施方案中,疾病或病症的特征在于功能失调或异常的蛋白质或多肽活性。例如,所述疾病或病症选自由以下组成的组:罕见病、感染性疾病、癌症和增生性疾病、遗传性疾病、自体免疫性疾病、糖尿病、神经退行性疾病、心血管和肾血管疾病以及代谢性疾病。In some aspects, the present disclosure provides a method of treating a disease or disorder in a mammal (eg, a human) in need thereof. The method includes the step of administering to the mammal a therapeutically effective amount of a loaded LNP or pharmaceutical composition of the present disclosure. In some embodiments, the method comprises administering to the subject a compound comprising formula (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y), (I-Ya)(IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or ( Compounds of IIb), phospholipids, structured lipids, PEG lipids and lipid nanoparticles of one or more therapeutic and/or prophylactic agents, thereby delivering said therapeutic and/or prophylactic agents to said Cell steps. In some embodiments, the disease or disorder is characterized by dysfunctional or aberrant protein or polypeptide activity. For example, the disease or disorder is selected from the group consisting of rare diseases, infectious diseases, cancer and proliferative diseases, genetic diseases, autoimmune diseases, diabetes, neurodegenerative diseases, cardiovascular and renovascular diseases, and Metabolic disease.
在一些实施方案中,本公开提供了一种治疗受试者的疾病或病症的方法,其中所述方法包括向受试者施用包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,DSPC,胆固醇和PEG2k-DMG以及一种或多种选自核苷酸、多肽和核酸(例如,RNA)的治疗剂和/或预防剂的脂质纳米粒子的步骤。例如,在一些实施方案中,本公开提供了一种治疗受试者的疾病或病症的方法,其中所述方法包括向受试者施用包含式(A)的化合物、DSPC、胆固醇和PEG2k-DMG以及一种或多种选自核苷酸、多肽和核酸(例如,RNA)的治疗剂和/或预防剂的脂质纳米粒子的步骤。例如,在一些实施方案中,本公开提供了一种治疗受试者的疾病或病症的方法,其中所述方法包括向受试者施用包含表1的化合物、DSPC、胆固醇和PEG2k-DMG以及一种或多种选自核苷酸、多肽和核酸(例如,RNA)的治疗剂和/或预防剂的脂质纳米粒子的步骤。In some embodiments, the present disclosure provides a method of treating a disease or disorder in a subject, wherein the method comprises administering to the subject a method comprising formula (A), (A1 ), (A2), (A3) , (B), (I), (I'), (IX), (IY), (I-Ya) (IA), (Ia), (Ib), (Ic), (Id), (Ie) , (If), (Ig), (II), (IIa) or (IIb) compounds, DSPC, cholesterol and PEG 2k -DMG and one or more selected from the group consisting of nucleotides, polypeptides and nucleic acids (e.g., RNA ) of lipid nanoparticles of therapeutic and/or prophylactic agents. For example, in some embodiments, the present disclosure provides a method of treating a disease or disorder in a subject, wherein the method comprises administering to the subject a compound comprising formula (A), DSPC, cholesterol, and PEG 2k- The step of DMG and one or more lipid nanoparticles selected from the group consisting of nucleotides, polypeptides, and nucleic acid (eg, RNA) therapeutic and/or prophylactic agents. For example, in some embodiments, the present disclosure provides a method of treating a disease or disorder in a subject, wherein the method comprises administering to the subject a compound comprising Table 1, DSPC, cholesterol, and PEG 2k -DMG, and The step of one or more lipid nanoparticles of therapeutic and/or prophylactic agents selected from the group consisting of nucleotides, polypeptides, and nucleic acids (eg, RNA).
在一些实施方案中,本公开提供了一种治疗受试者的疾病或病症的方法,其中所述方法包括向受试者施用包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,DSPC,胆固醇和PEG-1以及一种或多种选自核苷酸、多肽和核酸(例如,RNA)的治疗剂和/或预防剂的脂质纳米粒子的步骤。例如,在一些实施方案中,本公开提供了一种治疗受试者的疾病或病症的方法,其中所述方法包括向受试者施用包含式(A)的化合物、DSPC、胆固醇和PEG-1以及一种或多种选自核苷酸、多肽和核酸(例如,RNA)的治疗剂和/或预防剂的脂质纳米粒子的步骤。例如,在一些实施方案中,本公开提供了一种治疗受试者的疾病或病症的方法,其中所述方法包括向受试者施用包含表1的化合物、DSPC、胆固醇和PEG-1以及一种或多种选自核苷酸、多肽和核酸(例如,RNA)的治疗剂和/或预防剂的脂质纳米粒子的步骤。In some embodiments, the present disclosure provides a method of treating a disease or disorder in a subject, wherein the method comprises administering to the subject a method comprising formula (A), (A1 ), (A2), (A3) , (B), (I), (I'), (I-X), (I-Y), (I-Ya) (IA), (Ia), (Ib), (Ic), (Id), (Ie) , (If), (Ig), (II), (IIa) or (IIb) compounds, DSPC, cholesterol and PEG-1 and one or more selected from nucleotides, polypeptides and nucleic acids (eg, RNA) Steps of lipid nanoparticles of therapeutic and/or prophylactic agents. For example, in some embodiments, the present disclosure provides a method of treating a disease or disorder in a subject, wherein the method comprises administering to the subject a compound comprising formula (A), DSPC, cholesterol, and PEG-1 and one or more lipid nanoparticles of therapeutic and/or prophylactic agents selected from the group consisting of nucleotides, polypeptides, and nucleic acids (eg, RNA). For example, in some embodiments, the present disclosure provides a method of treating a disease or disorder in a subject, wherein the method comprises administering to the subject a compound comprising Table 1, DSPC, cholesterol and PEG-1 and a The step of one or more lipid nanoparticles of therapeutic and/or prophylactic agents selected from nucleotides, polypeptides, and nucleic acids (eg, RNA).
在又一方面,本公开的特征在于一种降低免疫原性的方法,其包括将本公开的负载LNP或药物组合物引入细胞中,其中与参考组合物所诱导的细胞中细胞免疫反应的诱导相比,所述负载LNP或药物组合物降低所述细胞对所述负载LNP或药物组合物的细胞免疫反应的诱导。在一些实施方案中,所述细胞在受试者体内并且所述接触包括将细胞施用至受试者。在一些实施方案中,所述方法包括向受试者施用包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,磷脂,结构性脂质,PEG脂质以及一种或多种选自核苷酸、多肽和核酸(例如,RNA)的治疗剂和/或预防剂的脂质纳米粒子的步骤,其中与参考化合物所诱导的细胞中细胞免疫反应的诱导相比较,包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物的脂质纳米粒子降低所述细胞对包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物的脂质纳米粒子的细胞免疫反应的诱导。例如,细胞免疫反应是先天免疫反应、适应性免疫反应或两者。In yet another aspect, the present disclosure features a method of reducing immunogenicity comprising introducing a loaded LNP or pharmaceutical composition of the present disclosure into a cell, wherein induction of a cellular immune response in the cell induced by the reference composition In contrast, the LNP-loaded or pharmaceutical composition reduces the induction of a cellular immune response by the cells to the LNP-loaded or pharmaceutical composition. In some embodiments, the cells are in the subject and the contacting comprises administering the cells to the subject. In some embodiments, the method comprises administering to the subject a compound comprising formula (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y), (I-Ya)(IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or ( Steps of compounds of IIb), phospholipids, structured lipids, PEG lipids, and one or more lipid nanoparticles selected from the group consisting of nucleotides, polypeptides and nucleic acid (eg, RNA) therapeutic and/or prophylactic agents , wherein compared with the induction of a cellular immune response in cells induced by the reference compound, comprising formula (A), (A1), (A2), (A3), (B), (I), (I'), ( I-X), (I-Y), (I-Ya) (IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa ) or (IIb) compounds of the lipid nanoparticles reduce said cell pair comprising formula (A), (A1), (A2), (A3), (B), (I), (I'), (I-X ), (I-Y), (I-Ya) (IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) Induction of cellular immune responses by lipid nanoparticles of compounds or (IIb). For example, the cellular immune response is an innate immune response, an adaptive immune response, or both.
在一些实施方案中,本公开提供了一种降低受试者的免疫原性的方法,其中所述方法包括向受试者施用包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,DSPC,胆固醇和PEG2k-DMG以及一种或多种选自核苷酸、多肽和核酸(例如,RNA)的治疗剂和/或预防剂的脂质纳米粒子的步骤。例如,在一些实施方案中,本公开提供了一种降低受试者的免疫原性的方法,其中所述方法包括向受试者施用包含式(A)的化合物、DSPC、胆固醇和PEG2k-DMG以及一种或多种选自核苷酸、多肽和核酸(例如,RNA)的治疗剂和/或预防剂的脂质纳米粒子的步骤。例如,在一些实施方案中,本公开提供了一种降低受试者的免疫原性的方法,其中所述方法包括向受试者施用包含表1的化合物、DSPC、胆固醇和PEG2k-DMG以及一种或多种选自核苷酸、多肽和核酸(例如,RNA)的治疗剂和/或预防剂的脂质纳米粒子的步骤。In some embodiments, the present disclosure provides a method of reducing immunogenicity in a subject, wherein the method comprises administering to the subject a method comprising formula (A), (A1), (A2), (A3) , (B), (I), (I'), (IX), (IY), (I-Ya) (IA), (Ia), (Ib), (Ic), (Id), (Ie) , (If), (Ig), (II), (IIa) or (IIb) compounds, DSPC, cholesterol and PEG 2k -DMG and one or more selected from the group consisting of nucleotides, polypeptides and nucleic acids (e.g., RNA ) of lipid nanoparticles of therapeutic and/or prophylactic agents. For example, in some embodiments, the present disclosure provides a method of reducing immunogenicity in a subject, wherein the method comprises administering to the subject a compound comprising formula (A), DSPC, cholesterol, and PEG 2k- The step of DMG and one or more lipid nanoparticles selected from the group consisting of nucleotides, polypeptides, and nucleic acid (eg, RNA) therapeutic and/or prophylactic agents. For example, in some embodiments, the present disclosure provides a method of reducing immunogenicity in a subject, wherein the method comprises administering to the subject a compound comprising Table 1, DSPC, cholesterol, and PEG 2k -DMG, and The step of one or more lipid nanoparticles of therapeutic and/or prophylactic agents selected from the group consisting of nucleotides, polypeptides, and nucleic acids (eg, RNA).
在一些实施方案中,本公开提供了一种降低受试者的免疫原性的方法,其中所述方法包括向受试者施用包含式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,DSPC,胆固醇和PEG-1以及一种或多种选自核苷酸、多肽和核酸(例如,RNA)的治疗剂和/或预防剂的脂质纳米粒子的步骤。例如,在一些实施方案中,本公开提供了一种降低受试者的免疫原性的方法,其中所述方法包括向受试者施用包含式(A)的化合物、DSPC、胆固醇和PEG-1以及一种或多种选自核苷酸、多肽和核酸(例如,RNA)的治疗剂和/或预防剂的脂质纳米粒子的步骤。例如,在一些实施方案中,本公开提供了降低一种受试者的免疫原性的方法,其中所述方法包括向受试者施用包含表1的化合物、DSPC、胆固醇和PEG-1以及一种或多种选自核苷酸、多肽和核酸(例如,RNA)的治疗剂和/或预防剂的脂质纳米粒子的步骤。In some embodiments, the present disclosure provides a method of reducing immunogenicity in a subject, wherein the method comprises administering to the subject a method comprising formula (A), (A1), (A2), (A3) , (B), (I), (I'), (I-X), (I-Y), (I-Ya) (IA), (Ia), (Ib), (Ic), (Id), (Ie) , (If), (Ig), (II), (IIa) or (IIb) compounds, DSPC, cholesterol and PEG-1 and one or more selected from nucleotides, polypeptides and nucleic acids (eg, RNA) Steps of lipid nanoparticles of therapeutic and/or prophylactic agents. For example, in some embodiments, the present disclosure provides a method of reducing immunogenicity in a subject, wherein the method comprises administering to the subject a compound comprising formula (A), DSPC, cholesterol, and PEG-1 and one or more lipid nanoparticles of therapeutic and/or prophylactic agents selected from the group consisting of nucleotides, polypeptides, and nucleic acids (eg, RNA). For example, in some embodiments, the present disclosure provides a method of reducing immunogenicity in a subject, wherein the method comprises administering to the subject a compound comprising Table 1, DSPC, cholesterol and PEG-1 and a The step of one or more lipid nanoparticles of therapeutic and/or prophylactic agents selected from nucleotides, polypeptides, and nucleic acids (eg, RNA).
本公开还包括合成式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物的方法,以及制备包含脂质组分的脂质纳米粒子(例如,空LNP或负载LNP)的方法,所述脂质组分包括式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物。The present disclosure also includes the synthesis of formulae (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y), (I-Ya) (IA ), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb) compounds, and methods for preparing compounds comprising lipids Methods of lipid nanoparticles (e.g., empty LNPs or loaded LNPs) of lipid components comprising formulae (A), (A1), (A2), (A3), (B), (I) ), (I'), (I-X), (I-Y), (I-Ya) (IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig ), (II), (IIa) or (IIb).
在细胞中产生多肽的方法Methods of producing polypeptides in cells
本公开提供了在哺乳动物细胞中产生所关注的多肽的方法。产生多肽的方法包括使细胞与包含编码所关注的多肽的mRNA的脂质纳米粒子(例如,空LNP或负载LNP)接触。在细胞与纳米粒子组合物接触后,mRNA可以被吸收并在细胞中翻译以产生所关注的多肽。The present disclosure provides methods for producing polypeptides of interest in mammalian cells. Methods of producing polypeptides include contacting cells with lipid nanoparticles (eg, empty LNPs or loaded LNPs) comprising mRNA encoding the polypeptide of interest. After the cell is contacted with the nanoparticle composition, the mRNA can be taken up and translated in the cell to produce the polypeptide of interest.
一般而言,使哺乳动物细胞与包含编码所关注的多肽的mRNA的脂质纳米粒子(例如,空LNP或负载LNP)接触的步骤可以在体内、离体、培养物中或在体外进行。与细胞接触的脂质纳米粒子(例如,空LNP或负载LNP)的量和/或其中的mRNA的量可以取决于所接触的细胞或组织的类型、施用方式、脂质纳米粒子(例如,空LNP或负载LNP)和其中的mRNA的物理化学特征(例如大小、电荷和化学组成)以及其他因素。一般而言,有效量的脂质纳米粒子(例如,空LNP或负载LNP)将允许在细胞中高效地产生多肽。效率的量度可以包括多肽翻译(由多肽表达表示)、mRNA降解的水平和免疫反应指标。In general, the step of contacting mammalian cells with lipid nanoparticles (eg, empty LNPs or loaded LNPs) comprising mRNA encoding a polypeptide of interest can be performed in vivo, ex vivo, in culture, or in vitro. The amount of lipid nanoparticles (e.g., empty LNPs or loaded LNPs) and/or the amount of mRNA therein contacted with the cells can depend on the type of cells or tissues contacted, the mode of administration, the lipid nanoparticles (e.g., empty LNPs). LNPs or loaded LNPs) and the physicochemical characteristics of the mRNAs therein (eg, size, charge, and chemical composition), among other factors. In general, an effective amount of lipid nanoparticles (eg, empty LNPs or loaded LNPs) will allow efficient production of polypeptides in cells. Measures of efficiency can include polypeptide translation (indicated by polypeptide expression), levels of mRNA degradation, and indicators of immune response.
使包含mRNA的脂质纳米粒子(例如,空LNP或负载LNP)与细胞接触的步骤可能包括或引起转染。包含在脂质纳米粒子(例如,空LNP或负载LNP)的脂质组分中的磷脂可以促进转染和/或增加转染效率,例如,通过与细胞膜或细胞内膜相互作用和/或融合。转染可以允许mRNA在细胞内翻译。The step of contacting the mRNA-containing lipid nanoparticles (eg, empty LNPs or loaded LNPs) with the cells may include or result in transfection. Phospholipids included in lipid components of lipid nanoparticles (e.g., empty LNPs or loaded LNPs) can facilitate transfection and/or increase transfection efficiency, e.g., by interacting and/or fusing with cell membranes or intracellular membranes . Transfection can allow translation of mRNA within the cell.
在一些实施方案中,可以在治疗上使用本文所述的脂质纳米粒子(例如,空LNP或负载LNP)。例如,包含在脂质纳米粒子(例如,空LNP或负载LNP)中的mRNA可以编码治疗性多肽(例如,在可翻译区中),并在接触和/或进入(例如,转染)细胞后产生治疗性多肽。在其他实施方案中,包含在脂质纳米粒子(例如,空LNP或负载LNP)中的mRNA可以编码能改善或增加受试者的免疫力的多肽。例如,mRNA可以编码粒细胞集落刺激因子或曲妥珠单抗(trastuzumab)。In some embodiments, the lipid nanoparticles described herein (eg, empty LNPs or loaded LNPs) can be used therapeutically. For example, mRNA contained in a lipid nanoparticle (eg, empty LNP or loaded LNP) can encode a therapeutic polypeptide (eg, in a translatable region), and upon contacting and/or entering (eg, transfecting) a cell Production of therapeutic polypeptides. In other embodiments, the mRNA contained in the lipid nanoparticle (eg, empty LNP or loaded LNP) can encode a polypeptide that improves or increases immunity in a subject. For example, the mRNA can encode granulocyte colony stimulating factor or trastuzumab.
在某些实施方案中,包含在脂质纳米粒子(例如,空LNP或负载LNP)中的mRNA可以编码重组多肽,所述重组多肽可以替代可能基本上不存在于与纳米粒子组合物接触的细胞中的一种或多种多肽。所述一种或多种基本上不存在的多肽可能因编码基因或其调控路径的基因突变而缺乏。可替代地,由mRNA翻译产生的重组多肽可以拮抗存在于细胞中、细胞表面上或由细胞分泌的内源性蛋白的活性。可能希望拮抗性重组多肽抵抗由内源性蛋白的活性所引起的有害影响,诸如由突变引起的改变的活性或定位。在另一个替代方案中,由mRNA翻译产生的重组多肽可以间接或直接地拮抗存在于细胞中、细胞表面上或由细胞分泌的生物部分的活性。受拮抗的生物部分可以包括但不限于脂质(例如胆固醇)、脂蛋白(例如低密度脂蛋白)、核酸、碳水化合物和小分子毒素。由mRNA翻译产生的重组多肽可以被工程化用于定位在细胞内,诸如在特定区室诸如细胞核内,或者可以被工程化用于由细胞分泌或易位至细胞的质膜。In certain embodiments, the mRNA contained in the lipid nanoparticle (eg, empty LNP or loaded LNP) can encode a recombinant polypeptide that can replace cells that may not be substantially present in contact with the nanoparticle composition one or more of the polypeptides. The one or more substantially absent polypeptides may be lacking due to mutations in the genes encoding the genes or their regulatory pathways. Alternatively, recombinant polypeptides produced by translation of mRNA can antagonize the activity of endogenous proteins present in the cell, on the cell surface, or secreted by the cell. It may be desirable for antagonistic recombinant polypeptides to counteract deleterious effects caused by the activity of the endogenous protein, such as altered activity or localization caused by mutation. In another alternative, recombinant polypeptides produced by translation of mRNA may indirectly or directly antagonize the activity of biological moieties present in, on the surface of, or secreted by cells. Antagonized biological moieties can include, but are not limited to, lipids (eg, cholesterol), lipoproteins (eg, low density lipoproteins), nucleic acids, carbohydrates, and small molecule toxins. Recombinant polypeptides resulting from translation of mRNA can be engineered for localization within a cell, such as within a specific compartment such as the nucleus, or can be engineered for secretion by the cell or translocation to the cell's plasma membrane.
在一些实施方案中,使细胞与包含mRNA的脂质纳米粒子(例如,空LNP或负载LNP)接触可以降低细胞对外源性核酸的先天免疫反应。细胞可以与包含第一量的包含可翻译区的第一外源性mRNA的第一脂质纳米粒子(例如,空LNP或负载LNP)接触,并且可以测定所述细胞对所述第一外源性mRNA的先天免疫反应的水平。随后,所述细胞可以与包含第二量的第一外源性mRNA的第二组合物接触,所述第二量是与第一量相比较低量的第一外源性mRNA。可替代地,第二组合物可以包含第一量的第二外源性mRNA,所述第二外源性mRNA不同于第一外源性mRNA。使细胞与第一和第二组合物接触的步骤可以重复一次或多次。另外,可以任选地测定在细胞中产生多肽(例如,翻译)的效率,并且所述细胞可以反复地与第一和/或第二组合物再接触,直至实现目标蛋白质产生效率。In some embodiments, contacting cells with mRNA-containing lipid nanoparticles (eg, empty LNPs or loaded LNPs) can reduce the cellular innate immune response to exogenous nucleic acids. A cell can be contacted with a first lipid nanoparticle (eg, empty LNP or loaded LNP) comprising a first amount of a first exogenous mRNA comprising a translatable region, and the response of the cell to the first exogenous mRNA can be determined. Levels of innate immune responses to sex mRNAs. Subsequently, the cell can be contacted with a second composition comprising a second amount of the first exogenous mRNA, the second amount being a lower amount of the first exogenous mRNA compared to the first amount. Alternatively, the second composition may comprise a first amount of a second exogenous mRNA that is different from the first exogenous mRNA. The step of contacting the cells with the first and second compositions can be repeated one or more times. Additionally, the efficiency of polypeptide production (eg, translation) in cells can optionally be determined, and the cells can be repeatedly recontacted with the first and/or second compositions until the desired protein production efficiency is achieved.
将治疗剂递送至细胞和器官的方法Methods of delivering therapeutic agents to cells and organs
本公开提供了将治疗剂和/或预防剂递送至哺乳动物细胞或器官的方法。将治疗剂和/或预防剂递送至细胞包括将包含所述治疗剂和/或预防剂的脂质纳米粒子(例如,空LNP或负载LNP)施用至受试者,其中所述组合物的施用包括使所述细胞与所述组合物接触。例如,可以将蛋白质、细胞毒性剂、放射性离子、化学治疗剂或核酸(诸如RNA,例如mRNA))递送至细胞或器官。在治疗剂和/或预防剂是mRNA的情况下,在细胞与纳米粒子组合物接触后,可翻译的mRNA可以在细胞中翻译以产生所关注的多肽。然而,也可以将基本上不可翻译的mRNA递送至细胞。基本上不可翻译的mRNA可以用作疫苗和/或可以阻隔细胞的翻译组分以减少其他物质在细胞中的表达。The present disclosure provides methods of delivering therapeutic and/or prophylactic agents to mammalian cells or organs. Delivering a therapeutic and/or prophylactic agent to a cell comprises administering to a subject a lipid nanoparticle (eg, empty LNP or loaded LNP) comprising the therapeutic and/or prophylactic agent, wherein administration of the composition Including contacting the cells with the composition. For example, proteins, cytotoxic agents, radioactive ions, chemotherapeutic agents, or nucleic acids (such as RNA, eg, mRNA) can be delivered to cells or organs. Where the therapeutic and/or prophylactic agent is mRNA, upon contact of the cell with the nanoparticle composition, the translatable mRNA can be translated in the cell to produce the polypeptide of interest. However, substantially untranslatable mRNA can also be delivered to cells. Substantially untranslatable mRNA can be used as a vaccine and/or can block the translational components of the cell to reduce the expression of other substances in the cell.
在一些实施方案中,脂质纳米粒子(例如,空LNP或负载LNP)可以靶向特定类型或类别的细胞(例如,特定器官或其系统的细胞)。例如,包含所关注的治疗剂和/或预防剂的脂质纳米粒子(例如,空LNP或负载LNP)可以特异性递送至哺乳动物肝、肾、脾或肺。向特定类别的细胞、器官或系统或其组的特异性递送意味着,相对于其他目的地而言,较高比例的包含治疗剂和/或预防剂的脂质纳米粒子(例如,负载LNP)被递送至所关注的目的地(例如组织)。在一些实施方案中,包含mRNA的负载LNP的特异性递送可导致与另一个目的地(例如脾)的细胞相比,所靶向的目的地(例如,所关注的组织,诸如肝)的细胞中的mRNA表达增加大于2倍、5倍、10倍、15倍或20倍。在一些实施方案中,所关注的组织选自由以下组成的组:肝、肾、肺、脾和肿瘤组织(例如,通过肿瘤内注射)。In some embodiments, lipid nanoparticles (eg, empty LNPs or loaded LNPs) can target specific types or classes of cells (eg, cells of specific organs or systems thereof). For example, lipid nanoparticles (eg, empty LNPs or loaded LNPs) containing therapeutic and/or prophylactic agents of interest can be specifically delivered to mammalian liver, kidney, spleen, or lung. Specific delivery to specific classes of cells, organs or systems, or groups thereof, implies a higher proportion of lipid nanoparticles (e.g., loaded with LNPs) containing therapeutic and/or prophylactic agents relative to other destinations is delivered to the destination of interest (eg, organization). In some embodiments, specific delivery of an mRNA-containing LNP-loaded can result in cells of a targeted destination (eg, a tissue of interest, such as the liver) compared to cells of another destination (eg, the spleen) The mRNA expression increased by greater than 2-fold, 5-fold, 10-fold, 15-fold or 20-fold. In some embodiments, the tissue of interest is selected from the group consisting of liver, kidney, lung, spleen, and tumor tissue (eg, by intratumoral injection).
在一些实施方案中,包含在本公开的负载LNP(即,用本公开的化合物配制的脂质纳米粒子)中的mRNA的特异性递送可导致与包含在用另一种脂质配制的LNP(即,没有式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的脂质)中的mRNA的递送相比,mRNA表达增加大于2倍、5倍、10倍、15倍或20倍。In some embodiments, specific delivery of mRNA contained in a loaded LNP of the present disclosure (ie, a lipid nanoparticle formulated with a compound of the present disclosure) can result in a That is, there are no formulas (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y), (I-Ya) (IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb) lipids) compared to , mRNA expression increased by greater than 2-fold, 5-fold, 10-fold, 15-fold or 20-fold.
作为靶向递送或特异性递送的另一个实例,编码蛋白质结合配偶体(例如,抗体或其功能片段、支架蛋白或肽)或细胞表面上的受体的mRNA可以包含在纳米粒子组合物中。mRNA可以另外或替代地用于指导脂质、碳水化合物或其他生物部分的合成和细胞外定位。可替代地,脂质纳米粒子(例如,空LNP或负载LNP)中的其他治疗剂和/或预防剂或成分(例如脂质或配体)可以基于其对特定受体(例如低密度脂蛋白受体)的亲和力来选择,以使得脂质纳米粒子(例如,空LNP或负载LNP)能更容易地与包括这些受体的靶细胞群相互作用。例如,配体可以包括但不限于指定结合对的成员、抗体、单克隆抗体、Fv片段、单链Fv(scFv)片段、Fab’片段、F(ab’)2片段、单结构域抗体、骆驼化抗体及其片段、人源化抗体及其片段,以及其多价型式;多价结合试剂,包括单特异性或双特异性抗体诸如二硫键稳定化的Fv片段、scFv串联体、双抗体、三抗体或四抗体;以及适体、受体和融合蛋白。As another example of targeted or specific delivery, mRNA encoding protein binding partners (eg, antibodies or functional fragments thereof, scaffold proteins or peptides) or receptors on the cell surface can be included in nanoparticle compositions. mRNA can additionally or alternatively be used to direct the synthesis and extracellular localization of lipids, carbohydrates or other biological moieties. Alternatively, other therapeutic and/or prophylactic agents or components (eg, lipids or ligands) in lipid nanoparticles (eg, empty LNPs or loaded LNPs) can be based on their ability to respond to specific receptors (eg, low-density lipoproteins). receptors) so that lipid nanoparticles (eg, empty LNPs or loaded LNPs) can more readily interact with target cell populations that include these receptors. For example, ligands can include, but are not limited to, members of a given binding pair, antibodies, monoclonal antibodies, Fv fragments, single chain Fv (scFv) fragments, Fab' fragments, F(ab')2 fragments, single domain antibodies, camelid Antibodies and fragments thereof, humanized antibodies and fragments thereof, and multivalent versions thereof; multivalent binding reagents, including monospecific or bispecific antibodies such as disulfide stabilized Fv fragments, scFv concatemers, diabodies , tri- or tetrabodies; and aptamers, receptors, and fusion proteins.
在一些实施方案中,配体可以是表面结合的抗体,其可以允许调谐细胞靶向特异性。这尤其有用,因为可以针对所需靶向位点的所关注的表位而产生高度特异性的抗体。在一些实施方案中,在细胞表面上表达多种抗体,并且每种抗体可以对所需的靶标具有不同特异性。此类方法可以增加靶向相互作用的亲合力和特异性。In some embodiments, the ligand may be a surface-bound antibody, which may allow tuning of cellular targeting specificity. This is especially useful because highly specific antibodies can be raised against epitopes of interest at the desired targeting site. In some embodiments, multiple antibodies are expressed on the cell surface, and each antibody may have different specificities for a desired target. Such methods can increase the affinity and specificity of targeted interactions.
例如,生物学领域中的技术人员可以基于所需的细胞定位或功能来选择配体。For example, one skilled in the art of biology can select ligands based on the desired cellular localization or function.
所靶向的细胞可以包括但不限于肝细胞、上皮细胞、造血细胞、上皮细胞、内皮细胞、肺细胞、骨细胞、干细胞、间充质细胞、神经细胞、心脏细胞、脂肪细胞、血管平滑肌细胞、心肌细胞、骨骼肌细胞、β细胞、垂体细胞、滑膜衬里细胞、卵巢细胞、睾丸细胞、成纤维细胞、B细胞、T细胞、网织红细胞、白细胞、粒细胞和肿瘤细胞。Targeted cells can include, but are not limited to, hepatocytes, epithelial cells, hematopoietic cells, epithelial cells, endothelial cells, lung cells, bone cells, stem cells, mesenchymal cells, neural cells, cardiac cells, adipocytes, vascular smooth muscle cells , cardiomyocytes, skeletal muscle cells, beta cells, pituitary cells, synovial lining cells, ovarian cells, testicular cells, fibroblasts, B cells, T cells, reticulocytes, leukocytes, granulocytes and tumor cells.
在一些实施方案中,脂质纳米粒子(例如,空LNP或负载LNP)可以靶向肝细胞。已显示,载脂蛋白诸如载脂蛋白E(apoE)在体内与含中性或接近中性的脂质的脂质纳米粒子(例如,空LNP或负载LNP)缔合,并且已知所述载脂蛋白与在肝细胞表面上发现的受体诸如低密度脂蛋白受体(LDLR)缔合。因此,施用至受试者的包含具有中性或接近中性的电荷的脂质组分的纳米粒子组合物可以在受试者体内获取apoE,并且随后可以将治疗剂和/或预防剂(例如,RNA)以靶向方式递送至包含LDLR的肝细胞。In some embodiments, lipid nanoparticles (eg, empty LNPs or loaded LNPs) can target hepatocytes. Apolipoproteins such as apolipoprotein E (apoE) have been shown to associate with neutral or near-neutral lipid-containing lipid nanoparticles (eg, empty LNPs or loaded LNPs) in vivo, and are known to Lipoproteins associate with receptors found on the surface of liver cells, such as the low density lipoprotein receptor (LDLR). Thus, a nanoparticle composition comprising a lipid component with a neutral or near-neutral charge administered to a subject can acquire apoE in the subject, and subsequently therapeutic and/or prophylactic agents (e.g., , RNA) in a targeted manner to LDLR-containing hepatocytes.
治疗疾病和病症的方法Methods of Treating Diseases and Conditions
脂质纳米粒子(例如,空LNP或负载LNP)可用于治疗疾病、病症或病状。特别地,此类组合物可以用于治疗以缺失或异常蛋白质或多肽活性为特征的疾病、病症或病状。例如,包含编码缺失或异常多肽的mRNA的脂质纳米粒子(例如,空LNP或负载LNP)可以被施用或递送至细胞。所述mRNA随后翻译可以产生所述多肽,由此减少或消除由所述多肽的缺乏或异常活性引起的问题。因为翻译可以快速发生,所以所述方法和组合物可以用于治疗急性疾病、病症或病状诸如败血症、中风和心肌梗塞。脂质纳米粒子(例如,空LNP或负载LNP)中包含的治疗剂和/或预防剂还能够改变给定物质的转录速率,由此影响基因表达。Lipid nanoparticles (eg, empty LNPs or loaded LNPs) can be used to treat diseases, disorders or conditions. In particular, such compositions can be used to treat diseases, disorders or conditions characterized by missing or abnormal protein or polypeptide activity. For example, lipid nanoparticles (eg, empty LNPs or loaded LNPs) comprising mRNA encoding a deleted or abnormal polypeptide can be administered or delivered to cells. Subsequent translation of the mRNA can produce the polypeptide, thereby reducing or eliminating problems caused by the lack of or abnormal activity of the polypeptide. Because translation can occur rapidly, the methods and compositions can be used to treat acute diseases, disorders or conditions such as sepsis, stroke and myocardial infarction. Therapeutic and/or prophylactic agents contained in lipid nanoparticles (eg, empty LNPs or loaded LNPs) can also alter the transcription rate of a given substance, thereby affecting gene expression.
可以施用组合物的以功能失调或异常蛋白质或多肽活性为特征的疾病、病症和/或病状包括但不限于罕见病、感染性疾病(作为疫苗和治疗剂两者)、癌症和增生性疾病、遗传性疾病、自体免疫性疾病、糖尿病、神经退行性疾病、心血管和肾血管疾病、以及代谢性疾病。多种疾病、病症和/或病状的特征可以在于蛋白质活性缺失(或大幅降低使得无法出现适当的蛋白质功能)。此类蛋白质可能不存在,或者它们可能基本上无功能。本公开提供了一种通过施用脂质纳米粒子(例如,空LNP或负载LNP)来治疗受试者的此类疾病、病症和/或病状的方法,所述脂质纳米粒子包含RNA和脂质组分,所述脂质组分包括根据式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的脂质,磷脂(任选不饱和的),PEG脂质和结构性脂质,其中所述RNA可以是编码拮抗或以其他方式克服受试者细胞中存在的异常蛋白质活性的多肽的mRNA。Diseases, disorders and/or conditions characterized by dysfunctional or abnormal protein or polypeptide activity to which the compositions may be administered include, but are not limited to, rare diseases, infectious diseases (both as vaccines and therapeutics), cancer and proliferative diseases, Genetic diseases, autoimmune diseases, diabetes, neurodegenerative diseases, cardiovascular and renovascular diseases, and metabolic diseases. A variety of diseases, disorders and/or conditions can be characterized by a loss of protein activity (or a substantial reduction such that proper protein function cannot occur). Such proteins may not exist, or they may be essentially nonfunctional. The present disclosure provides a method of treating such diseases, disorders and/or conditions in a subject by administering lipid nanoparticles (eg, empty LNPs or loaded LNPs) comprising RNA and lipids component, the lipid component comprises according to formula (A), (A1), (A2), (A3), (B), (I), (I'), (I-X), (I-Y), ( I-Ya) lipids of (IA), (Ia), (Ib), (Ic), (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb) , phospholipids (optionally unsaturated), PEG lipids, and structural lipids, wherein the RNA may be an mRNA encoding a polypeptide that antagonizes or otherwise overcomes the activity of an abnormal protein present in a subject's cells.
本公开提供了包括施用包含一种或多种治疗剂和/或预防剂的脂质纳米粒子(例如,空LNP或负载LNP)以及包含所述脂质纳米粒子的药物组合物的方法。对于本公开的特征和实施方案,术语治疗剂和预防剂在本文中可以互换使用。治疗性组合物或其成像、诊断或预防性组合物可以使用任何合理量和任何施用途径施用给受试者,以有效预防、治疗、诊断或成像疾病、病症和/或病状和/或用于任何其他目的。施用至给定受试者的指定量可以根据受试者的物种、年龄和一般状况;施用目的;特定组合物;施用模式等而变化。根据本公开的组合物可以被配制成易于施用和剂量一致的单位剂型。然而,应了解,本公开的组合物的总每日用量将由主治医师在合理医学判断的范围内决定。用于任何特定患者的指定治疗有效、预防有效或其他适当剂量水平(例如,用于成像)将取决于多种因素,其包括所治疗病症(如果存在的话)的严重程度和属性;采用的一种或多种治疗剂和/或预防剂;采用的指定组合物;患者的年龄、体重、一般健康状况、性别和饮食;采用的指定药物组合物的施用时间、施用途径和排泄速率;治疗持续时间;与采用的指定药物组合物组合使用或同时使用的药物;以及医学领域中众所周知的类似因素。The present disclosure provides methods comprising administering lipid nanoparticles (eg, empty LNPs or loaded LNPs) comprising one or more therapeutic and/or prophylactic agents, and pharmaceutical compositions comprising the lipid nanoparticles. For features and embodiments of the present disclosure, the terms therapeutic agent and prophylactic agent are used interchangeably herein. Therapeutic compositions or imaging, diagnostic or prophylactic compositions thereof can be administered to a subject using any reasonable amount and by any route of administration effective to prevent, treat, diagnose or image a disease, disorder and/or condition and/or for any other purpose. The indicated amount administered to a given subject may vary depending on the species, age, and general condition of the subject; the purpose of administration; the particular composition; the mode of administration, and the like. Compositions according to the present disclosure can be formulated in unit dosage form for ease of administration and uniformity of dosage. It is to be understood, however, that the total daily dosage of the compositions of the present disclosure will be determined by the attending physician within the scope of sound medical judgment. The indicated therapeutically effective, prophylactically effective, or other appropriate dosage level for any particular patient (eg, for imaging) will depend on a variety of factors, including the severity and nature of the condition being treated (if present); one or more therapeutic and/or prophylactic agents; the specified composition employed; the patient's age, weight, general health, gender and diet; the administration time, route of administration and excretion rate of the specified pharmaceutical composition employed; treatment duration time; drugs used in combination or concomitantly with the prescribed pharmaceutical composition employed; and similar factors well known in the medical arts.
负载LNP可以通过任何途径施用。在一些实施方案中,包括本文所述的一种或多种负载LNP的组合物,包括预防性、诊断或成像组合物,通过包括以下多种途径中的一种或多种来施用:口服、静脉内、肌肉内、动脉内、皮下、经皮或皮内、皮间、腹膜内、粘膜、鼻腔、肿瘤内、鼻内;通过吸入来施用;作为口腔喷雾剂和/或散剂、鼻腔喷雾剂和/或气雾剂、和/或通过门静脉导管来施用。在一些实施方案中,组合物可以通过静脉内、肌肉内、皮内、动脉内、肿瘤内、皮下或通过任何其他肠胃外施用途径或通过吸入来施用。然而,考虑到药物递送科学中可能的进展,本公开涵盖通过任何适当途径递送或施用本文所述的组合物。一般而言,最适当的施用途径将取决于多种因素,其包括包含一种或多种治疗剂和/或预防剂的负载LNP的性质(例如其在各种身体环境诸如血流和胃肠道中的稳定性)、患者的状况(例如患者是否能够耐受特定的施用途径)等。The loaded LNP can be administered by any route. In some embodiments, one or more LNP-loaded compositions described herein, including prophylactic, diagnostic or imaging compositions, are administered by one or more of a variety of routes including: oral, Intravenous, intramuscular, intraarterial, subcutaneous, transdermal or intradermal, interdermal, intraperitoneal, mucosal, nasal, intratumoral, intranasal; administration by inhalation; as oral spray and/or powder, nasal spray and/or by aerosol, and/or by portal vein catheter. In some embodiments, the composition may be administered intravenously, intramuscularly, intradermally, intraarterally, intratumorally, subcutaneously or by any other parenteral route of administration or by inhalation. However, the present disclosure contemplates delivery or administration of the compositions described herein by any suitable route, given possible advances in drug delivery science. In general, the most appropriate route of administration will depend on a variety of factors, including the nature of the loaded LNP comprising one or more therapeutic and/or prophylactic agents (eg, its exposure to various bodily environments such as the bloodstream and the gastrointestinal tract). stability in the tract), the condition of the patient (eg, whether the patient can tolerate a particular route of administration), and the like.
在某些实施方案中,根据本公开的组合物可以足以在给定剂量中递送约0.0001mg/kg至约10mg/kg、约0.001mg/kg至约10mg/kg、约0.005mg/kg至约10mg/kg、约0.01mg/kg至约10mg/kg、约0.05mg/kg至约10mg/kg、约0.1mg/kg至约10mg/kg、约1mg/kg至约10mg/kg、约2mg/kg至约10mg/kg、约5mg/kg至约10mg/kg、约0.0001mg/kg至约5mg/kg、约0.001mg/kg至约5mg/kg、约0.005mg/kg至约5mg/kg、约0.01mg/kg至约5mg/kg、约0.05mg/kg至约5mg/kg、约0.1mg/kg至约5mg/kg、约1mg/kg至约5mg/kg、约2mg/kg至约5mg/kg、约0.0001mg/kg至约2.5mg/kg、约0.001mg/kg至约2.5mg/kg、约0.005mg/kg至约2.5mg/kg、约0.01mg/kg至约2.5mg/kg、约0.05mg/kg至约2.5mg/kg、约0.1mg/kg至约2.5mg/kg、约1mg/kg至约2.5mg/kg、约2mg/kg至约2.5mg/kg、约0.0001mg/kg至约1mg/kg、约0.001mg/kg至约1mg/kg、约0.005mg/kg至约1mg/kg、约0.01mg/kg至约1mg/kg、约0.05mg/kg至约1mg/kg、约0.1mg/kg至约1mg/kg、约0.0001mg/kg至约0.25mg/kg、约0.001mg/kg至约0.25mg/kg、约0.005mg/kg至约0.25mg/kg、约0.01mg/kg至约0.25mg/kg、约0.05mg/kg至约0.25mg/kg,或约0.1mg/kg至约0.25mg/kg治疗剂和/或预防剂(例如,mRNA)的剂量水平来施用,其中1mg/kg(mpk)的剂量提供每1kg受试者体重1mg治疗剂和/或预防剂。在一些实施方案中,可以施用约0.001mg/kg至约10mg/kg的负载LNP的治疗剂和/或预防剂的剂量。在其他实施方案中,可以施用约0.005mg/kg至约2.5mg/kg的治疗剂和/或预防剂的剂量。在某些实施方案中,可以施用约0.1mg/kg至约1mg/kg的剂量。在其他实施方案中,可以施用约0.05mg/kg至约0.25mg/kg的剂量。剂量可以每天以相同或不同量施用一次或多次,以获得所需的mRNA表达水平和/或治疗、诊断、预防或成像效果。所需的剂量以例如一天三次、一天两次、一天一次、每隔一天一次、每三天一次、每周一次、每两周一次、每三周一次或每四周一次来递送。在某些实施方案中,所需的剂量可以使用多次施用(例如两次、三次、四次、五次、六次、七次、八次、九次、十次、十一次、十二次、十三次、十四次或更多次施用)来递送。在一些实施方案中,可以例如在手术程序之前或之后或在急性疾病、病症或病状的情况下施用单次剂量。In certain embodiments, compositions according to the present disclosure may be sufficient to deliver about 0.0001 mg/kg to about 10 mg/kg, about 0.001 mg/kg to about 10 mg/kg, about 0.005 mg/kg to about 0.0001 mg/kg in a given dose 10 mg/kg, about 0.01 mg/kg to about 10 mg/kg, about 0.05 mg/kg to about 10 mg/kg, about 0.1 mg/kg to about 10 mg/kg, about 1 mg/kg to about 10 mg/kg, about 2 mg/kg kg to about 10 mg/kg, about 5 mg/kg to about 10 mg/kg, about 0.0001 mg/kg to about 5 mg/kg, about 0.001 mg/kg to about 5 mg/kg, about 0.005 mg/kg to about 5 mg/kg, About 0.01 mg/kg to about 5 mg/kg, about 0.05 mg/kg to about 5 mg/kg, about 0.1 mg/kg to about 5 mg/kg, about 1 mg/kg to about 5 mg/kg, about 2 mg/kg to about 5 mg /kg, about 0.0001 mg/kg to about 2.5 mg/kg, about 0.001 mg/kg to about 2.5 mg/kg, about 0.005 mg/kg to about 2.5 mg/kg, about 0.01 mg/kg to about 2.5 mg/kg , about 0.05 mg/kg to about 2.5 mg/kg, about 0.1 mg/kg to about 2.5 mg/kg, about 1 mg/kg to about 2.5 mg/kg, about 2 mg/kg to about 2.5 mg/kg, about 0.0001 mg /kg to about 1 mg/kg, about 0.001 mg/kg to about 1 mg/kg, about 0.005 mg/kg to about 1 mg/kg, about 0.01 mg/kg to about 1 mg/kg, about 0.05 mg/kg to about 1 mg/kg kg, about 0.1 mg/kg to about 1 mg/kg, about 0.0001 mg/kg to about 0.25 mg/kg, about 0.001 mg/kg to about 0.25 mg/kg, about 0.005 mg/kg to about 0.25 mg/kg, about Dose levels of 0.01 mg/kg to about 0.25 mg/kg, about 0.05 mg/kg to about 0.25 mg/kg, or about 0.1 mg/kg to about 0.25 mg/kg therapeutic and/or prophylactic (eg, mRNA) to be administered, wherein a dose of 1 mg/kg (mpk) provides 1 mg of the therapeutic and/or prophylactic agent per 1 kg of subject body weight. In some embodiments, a dose of about 0.001 mg/kg to about 10 mg/kg of the LNP-loaded therapeutic and/or prophylactic agent can be administered. In other embodiments, doses of about 0.005 mg/kg to about 2.5 mg/kg of therapeutic and/or prophylactic agents can be administered. In certain embodiments, a dose of about 0.1 mg/kg to about 1 mg/kg may be administered. In other embodiments, a dose of about 0.05 mg/kg to about 0.25 mg/kg may be administered. Doses may be administered one or more times per day in the same or different amounts to achieve the desired level of mRNA expression and/or therapeutic, diagnostic, prophylactic or imaging effect. The desired dose is delivered, for example, three times a day, twice a day, once a day, every other day, every three days, every week, every two weeks, every three weeks, or every four weeks. In certain embodiments, the desired dose may be administered using multiple administrations (eg, two, three, four, five, six, seven, eight, nine, ten, eleven, twelve administration, thirteen, fourteen or more administrations). In some embodiments, a single dose may be administered, eg, before or after a surgical procedure or in the case of an acute disease, disorder or condition.
包含一种或多种治疗剂和/或预防剂的脂质纳米粒子(例如,空LNP或负载LNP)可以与一种或多种其他治疗剂、预防剂、诊断剂或成像剂组合使用。“与……组合”并非旨在表明剂必须在相同时间施用和/或被配制用于一起递送,尽管这些递送方法都在本公开的范围内。例如,包含一种或多种不同治疗剂和/或预防剂的一种或多种脂质纳米粒子(例如,空LNP或负载LNP)可以组合施用。组合物可在一种或多种其他所需的治疗剂或医疗程序的同时、之前或之后施用。一般而言,每种剂将以针对所述剂确定的剂量和/或时间表来施用。在一些实施方案中,本公开涵盖递送组合物或其成像、诊断或预防组合物与改善其生物利用率、降低和/或改良其代谢、抑制其排泄和/或改良其在体内的分布的剂的组合。Lipid nanoparticles (eg, empty LNPs or loaded LNPs) comprising one or more therapeutic and/or prophylactic agents can be used in combination with one or more other therapeutic, prophylactic, diagnostic, or imaging agents. "In combination with" is not intended to indicate that the agents must be administered at the same time and/or formulated for delivery together, although such delivery methods are within the scope of this disclosure. For example, one or more lipid nanoparticles (eg, empty LNPs or loaded LNPs) comprising one or more different therapeutic and/or prophylactic agents can be administered in combination. The compositions can be administered concurrently with, before, or after one or more other desired therapeutic agents or medical procedures. In general, each agent will be administered at the dose and/or schedule established for that agent. In some embodiments, the present disclosure contemplates delivering a composition or imaging, diagnostic or prophylactic composition thereof with an agent that improves its bioavailability, reduces and/or improves its metabolism, inhibits its excretion, and/or improves its distribution in the body The combination.
还将理解,组合使用的治疗性、预防性、诊断性或成像活性剂可以在单一组合物中一起施用或在不同组合物中分开施用。一般而言,预期组合使用的剂将在不超过其单独使用的水平的水平下使用。在一些实施方案中,组合使用的水平可以低于单独使用的水平。It will also be appreciated that therapeutic, prophylactic, diagnostic or imaging active agents used in combination may be administered together in a single composition or administered separately in different compositions. In general, it is contemplated that the agents used in combination will be used at levels that do not exceed the levels at which they are used individually. In some embodiments, the levels used in combination may be lower than those used alone.
用于组合方案中的疗法(治疗剂或程序)的特定组合将考虑所需治疗剂和/或程序的相容性以及要实现的所需治疗效果。还将理解,采用的疗法可以针对同一病症实现所需效果(例如可用于治疗癌症的组合物可以与化学治疗剂同时施用),或者所述疗法可以实现不同效果(例如,控制任何不良作用,诸如输注相关反应)。The particular combination of therapies (therapeutic agents or procedures) used in a combination regimen will take into account the compatibility of the desired therapeutic agents and/or procedures and the desired therapeutic effect to be achieved. It will also be appreciated that the therapy employed may achieve the desired effect for the same condition (eg, a composition useful in the treatment of cancer may be administered concurrently with the chemotherapeutic agent), or the therapy may achieve a different effect (eg, to control any adverse effects, such as infusion-related reactions).
脂质纳米粒子(例如,空LNP或负载LNP)可以与增加所述组合物的有效性和/或治疗窗的剂组合使用。这种剂可以是例如抗炎化合物、类固醇(例如皮质类固醇)、他汀类(statin)、雌二醇、BTK抑制剂、S1P1激动剂、糖皮质激素受体调节剂(GRM)或抗组胺。在一些实施方案中,脂质纳米粒子(例如,空LNP或负载LNP)可以与地塞米松、甲氨蝶呤、对乙酰氨基酚、H1受体阻断剂或H2受体阻断剂组合使用。在一些实施方案中,治疗有需要的受试者或将治疗剂和/或预防剂递送至受试者(例如,哺乳动物)的方法可以包括在施用纳米粒子组合物之前,用一种或多种剂预治疗所述受试者。例如,可以用可用量(例如,10mg、20mg、30mg、40mg、50mg、60mg、70mg、80mg、90mg、100mg或任何其他可用量)的地塞米松、甲氨蝶呤、对乙酰氨基酚、H1受体阻断剂或H2受体阻断剂预治疗受试者。预治疗可以在施用脂质纳米粒子(例如,空LNP或负载LNP)之前24小时或更短时间(例如,24小时、20小时、16小时、12小时、8小时、4小时、2小时、1小时、50分钟、40分钟、30分钟、20分钟或10分钟)进行并且可以以例如递增剂量量进行一次、两次或更多次。Lipid nanoparticles (eg, empty LNPs or loaded LNPs) can be used in combination with agents that increase the effectiveness and/or therapeutic window of the composition. Such agents may be, for example, anti-inflammatory compounds, steroids (eg, corticosteroids), statins, estradiol, BTK inhibitors, S1P1 agonists, glucocorticoid receptor modulators (GRMs), or antihistamines. In some embodiments, lipid nanoparticles (eg, empty LNPs or loaded LNPs) can be used in combination with dexamethasone, methotrexate, acetaminophen, H1 receptor blockers, or H2 receptor blockers . In some embodiments, a method of treating a subject in need thereof or delivering a therapeutic and/or prophylactic agent to a subject (eg, a mammal) can include, prior to administering the nanoparticle composition, using one or more The subject is pretreated with a dose. For example, a useful amount (eg, 10 mg, 20 mg, 30 mg, 40 mg, 50 mg, 60 mg, 70 mg, 80 mg, 90 mg, 100 mg, or any other available amount) of dexamethasone, methotrexate, acetaminophen, H1 can be used Subjects were pretreated with a receptor blocker or an H2 receptor blocker. Pre-treatment can be administered 24 hours or less (eg, 24 hours, 20 hours, 16 hours, 12 hours, 8 hours, 4 hours, 2 hours, 1 hours, 50 minutes, 40 minutes, 30 minutes, 20 minutes, or 10 minutes) and can be performed once, twice, or more, eg, in escalating doses.
本领域的技术人员将认识到或能够仅使用常规实验确定本文所述的根据本公开的指定实施方案的许多等同方案。本公开的范围不旨在限于以上描述,而是如所附权利要求中所阐述。Those skilled in the art will recognize, or be able to ascertain using no more than routine experimentation, many equivalents to the specific embodiments described herein in accordance with the present disclosure. The scope of the present disclosure is not intended to be limited to the above description, but rather is as set forth in the appended claims.
在权利要求中,除非指明相反情况或另外从上下文明显看出,否则冠词诸如“一个(a)”、“一种(an)”以及“所述(the)”可以意指一个/种或多于一个/种。除非指明相反情况或另外从上下文明显看出,否则如果一个、多于一个或所有的组成员存在于、被用于给定产品或过程中或以其他方式与给定产品或过程相关,则认为在组的一个或多个成员之间包括“或”的权利要求或描述是满足的。本公开包括其中仅有一个组成员存在于、被用于给定产品或过程中或以其他方式与给定产品或过程相关的实施方案。本公开包括其中多于一个或所有的组成员存在于、被用于给定产品或过程中或以其他方式与给定的产品或过程相关的实施方案。除非另外说明,否则如本文所用,表述“A、B或C中的一个或多个”、“一个或多个A、B或C”、“A、B和C中的一个或多个”、“一个或多个A、B和C”、“选自A、B和C”、“选自由A、B和C组成的组”等可互换使用并且全部都是指从由A、B和/或C组成的组中选择,即一个或多个A、一个或多个B、一个或多个C或其任何组合。In the claims, articles such as "a", "an" and "the" may mean one or the other, unless indicated to the contrary or otherwise clear from the context. More than one/species. Unless indicated to the contrary or otherwise apparent from the context, one, more than one or all of the group members are considered to be present if present in, used in, or otherwise related to a given product or process It is sufficient for a claim or description to include "or" between one or more members of the group. The present disclosure includes embodiments in which only one member of the group is present in, used in, or otherwise related to a given product or process. The present disclosure includes embodiments in which more than one or all of the group members are present in, used in, or otherwise related to a given product or process. As used herein, the expressions "one or more of A, B, or C," "one or more of A, B, or C," "one or more of A, B, and C," unless otherwise specified, "One or more of A, B, and C," "selected from A, B, and C," "selected from the group consisting of A, B, and C," and the like are used interchangeably and all refer to the term "selected from the group consisting of A, B, and C." and/or selected from the group consisting of C, ie one or more A's, one or more B's, one or more C's, or any combination thereof.
还应注意,术语“包含”旨在为开放的并且允许但不要求包括另外的要素或步骤。当在本文中使用术语“包含”时,由此也涵盖并公开术语“基本上由……组成”和“由……组成”。在整个说明书中,在组合物被描述为具有、包括或包含指定组分的情况下,预期组合物也基本上由或由所列举的组分组成。类似地,当方法或过程被描述为具有、包括或包含指定过程步骤时,所述过程也基本上由或由所列举过程步骤组成。另外,应了解,步骤的次序或进行某些动作的次序是不重要的,只要本发明保持可操作即可。此外,可以同时进行两个或更多个步骤或动作。It should also be noted that the term "comprising" is intended to be open-ended and allows, but does not require, the inclusion of additional elements or steps. When the term "comprising" is used herein, the terms "consisting essentially of" and "consisting of" are hereby also encompassed and disclosed. Throughout the specification, where compositions are described as having, comprising or comprising the specified components, it is contemplated that the compositions also consist essentially of or consist of the recited components. Similarly, when a method or process is described as having, comprising or comprising specified process steps, the process also consists essentially of or consists of the recited process steps. Additionally, it should be understood that the order of steps or order in which certain actions are performed is immaterial so long as the invention remains operable. Furthermore, two or more steps or actions may be performed simultaneously.
在给定范围的情况下,端点被包括在内。此外,应了解,除非另外指明或另外根据上下文和本领域的普通技术人员的理解明显看出,否则被表示为范围的值在本公开的不同实施方案中可以假定所述范围内的任何指定值或子范围,直到所述范围的下限的单位的十分之一,除非上下文另外明确规定。Where a range is given, the endpoints are included. Furthermore, it should be understood that the values expressed as ranges may assume any specified value within the range in various embodiments of the present disclosure unless otherwise indicated or otherwise apparent from the context and understanding of one of ordinary skill in the art or sub-ranges up to the tenth of the unit of the lower limit of the stated range, unless the context clearly dictates otherwise.
本公开的合成过程可以容许多种多样的官能团,因此可以使用各种取代的起始材料。这些过程通常在整个过程结束或快结束时提供所需的最终化合物,但在某些情况下可能希望将所述化合物进一步转化为其药学上可接受的盐。The synthetic procedures of the present disclosure can tolerate a wide variety of functional groups and thus can use a variety of substituted starting materials. These procedures generally provide the desired final compound at or near the end of the overall procedure, although in some cases it may be desirable to further convert the compound to its pharmaceutically acceptable salt.
本公开的化合物可以通过采用标准合成方法和本领域技术人员已知或技术人员根据本文的传授内容显而易见的程序使用可商购获得的起始材料、文献中已知的化合物或由易于制备的中间体以多种方式来制备。用于制备有机分子的标准合成方法和程序以及官能团转化和操作可以从相关科学文献或从本领域的标准教科书获得。尽管不局限于任一或若干来源,但以引用的方式并入本文中的经典教科书,诸如Smith,M.B.,March,J.,March’sAdvanced Organic Chemistry:Reactions,Mechanisms,and Structure,第5版,JohnWiley&Sons:New York,2001;Greene,T.W.,Wuts,P.G.M.,Protective Groups in OrganicSynthesis,第3版,John Wiley&Sons:New York,1999;R.Larock,Comprehensive OrganicTransformations,VCH Publishers(1989);L.Fieser和M.Fieser,Fieser and Fieser’sReagents for Organic Synthesis,John Wiley and Sons(1994);以及L.Paquette,编,Encyclopedia of Reagents for Organic Synthesis,John Wiley and Sons(1995),是可用的并且是本领域技术人员已知的公认的有机合成参考教科书。以下有关合成方法的描述被设计成说明而非限制用于制备本公开化合物的一般程序。The compounds of the present disclosure can be obtained by employing standard synthetic methods and procedures known to or apparent to those skilled in the art from the teachings herein using commercially available starting materials, compounds known in the literature, or from readily prepared intermediates Bodies are prepared in a variety of ways. Standard synthetic methods and procedures for preparing organic molecules and functional group transformations and manipulations can be obtained from the relevant scientific literature or from standard textbooks in the field. While not limited to any one or several sources, classic textbooks such as Smith, M.B., March, J., March's Advanced Organic Chemistry: Reactions, Mechanisms, and Structure, 5th Edition, are incorporated herein by reference, John Wiley & Sons: New York, 2001; Greene, T.W., Wuts, P.G.M., Protective Groups in Organic Synthesis, 3rd Edition, John Wiley & Sons: New York, 1999; R. Larock, Comprehensive Organic Transformations, VCH Publishers (1989); L. Fieser and M. . Fieser, Fieser and Fieser's Reagents for Organic Synthesis, John Wiley and Sons (1994); and L. Paquette, ed., Encyclopedia of Reagents for Organic Synthesis, John Wiley and Sons (1995), are available and are of skill in the art A recognized organic synthesis reference textbook known to personnel. The following descriptions of synthetic methods are designed to illustrate, but not to limit, general procedures for the preparation of compounds of the present disclosure.
具有本文所述的式中的任一个的本公开的化合物可以根据以下方案1、2和3中所说明的程序由可商购获得的起始材料或可以使用文献程序制备的起始材料来制备。方案中的变量(例如,R1、R2和R3等如本文所定义)。本领域的普通技术人员应注意到,在本文所述的反应工序和合成方案期间,可以改变某些步骤的次序,诸如保护基团的引入和去除。Compounds of the present disclosure having any of the formulae described herein can be prepared according to the procedures illustrated in Schemes 1, 2, and 3 below from commercially available starting materials or starting materials that can be prepared using literature procedures . The variables in the protocol (eg, R 1 , R 2 and R 3 etc. are as defined herein). One of ordinary skill in the art will note that the order of certain steps, such as the introduction and removal of protecting groups, may be altered during the reaction procedures and synthetic schemes described herein.
本领域的普通技术人员将认识到,可能需要通过使用保护基团保护某些基团免受反应条件影响。保护基团还可以用于区别分子中的类似官能团。保护基团的列表以及引入和去除这些基团的方法可见于Greene,T.W.,Wuts,P.G.M.,Protective Groups inOrganic Synthesis,第3版,John Wiley&Sons:New York,1999。One of ordinary skill in the art will recognize that it may be desirable to protect certain groups from reaction conditions through the use of protecting groups. Protecting groups can also be used to distinguish similar functional groups in a molecule. A list of protecting groups and methods of introducing and removing these groups can be found in Greene, T.W., Wuts, P.G.M., Protective Groups in Organic Synthesis, 3rd Edition, John Wiley & Sons: New York, 1999.
优选的保护基团包括但不限于:Preferred protecting groups include, but are not limited to:
对于羟基部分:TBS、苯甲基、THP、Ac;For hydroxyl moieties: TBS, benzyl, THP, Ac;
对于羧酸:苄酯、甲酯、乙酯、烯丙基酯;For carboxylic acids: benzyl ester, methyl ester, ethyl ester, allyl ester;
对于胺:Fmoc、Cbz、BOC、DMB、Ac、Bn、Tr、Ts、三氟乙酰基、邻苯二甲酰亚胺、亚苄基胺;For amines: Fmoc, Cbz, BOC, DMB, Ac, Bn, Tr, Ts, trifluoroacetyl, phthalimide, benzylideneamine;
对于二醇:Ac(×2)TBS(×2),或当放在一起时是丙酮化物;For diols: Ac(x2)TBS(x2), or acetonate when put together;
对于硫醇:Ac;For thiols: Ac;
对于苯并咪唑:SEM、苄基、PMB、DMB;For benzimidazole: SEM, benzyl, PMB, DMB;
对于醛:二烷基缩醛,诸如二甲氧基乙缩醛或二乙基乙酰基。For aldehydes: dialkyl acetals such as dimethoxyacetal or diethylacetyl.
在本文所述的反应方案中,可以产生多种立体异构体。当未指明特定立体异构体时,应了解为意指可以由所述反应产生的所有可能的立体异构体。本领域普通技术人员将认识到,所述反应可以被优化以优先得到一种异构体,或可以设计出新的方案以产生单一异构体。如果产生混合物,则可以使用诸如制备型薄层色谱法、制备型HPLC、制备型手性HPLC或制备型SFC等技术来分离所述异构体。In the reaction schemes described herein, various stereoisomers can be produced. When no specific stereoisomer is indicated, it is understood to mean all possible stereoisomers that can result from the reaction. One of ordinary skill in the art will recognize that the reactions can be optimized to give preference to one isomer, or new schemes can be devised to produce a single isomer. If a mixture is produced, the isomers can be separated using techniques such as preparative thin layer chromatography, preparative HPLC, preparative chiral HPLC, or preparative SFC.
方案1plan 1
如以上方案1所示,8-溴辛酸与醇a1(例如,十七烷-9-醇)反应,以得到酯b1(例如,8-溴辛酸十七烷-9-基酯)。步骤1可以在例如N-(3-二甲基氨基丙基)-N'-乙基碳二亚胺盐酸盐、N,N-二异丙基乙胺和DMAP的存在下在有机溶剂(例如,二氯甲烷)中进行。步骤1可以在室温下进行18小时。接下来,酯b1与2-氨基乙烷-1-醇反应以得到胺c1(例如,8-((2-羟乙基)氨基)辛酸十七烷-9-基酯)。步骤2可以在例如约60℃的温度下在乙醇中进行。然后,胺c1与溴烷基R1-Br(例如,1-溴十四烷)反应以得到化合物d1(例如,8-((2-羟乙基)(十四烷基)氨基)辛酸十七烷-9-基酯)。步骤3可以在N,N-二异丙基乙胺的存在下在乙醇中进行。As shown in Scheme 1 above, 8-bromooctanoic acid is reacted with alcohol a1 (eg, heptadecan-9-ol) to give ester b1 (eg, heptadecan-9-yl 8-bromooctanoate). Step 1 can be carried out in an organic solvent ( For example, dichloromethane). Step 1 can be performed at room temperature for 18 hours. Next, ester b1 is reacted with 2-aminoethane-1-ol to give amine c1 (eg, 8-((2-hydroxyethyl)amino)octanoic acid heptadecan-9-yl ester). Step 2 can be carried out in ethanol, for example, at a temperature of about 60°C. The amine c1 is then reacted with a bromoalkyl R1 - Br (eg, 1-bromotetradecane) to give compound d1 (eg, 8-((2-hydroxyethyl)(tetradecyl)amino)octanoic acid ten heptan-9-yl ester). Step 3 can be carried out in ethanol in the presence of N,N-diisopropylethylamine.
方案2Scenario 2
如以上方案2所示,酸a2(x3是介于1与7之间的整数;例如,8-溴辛酸)与醇b2(例如,壬烷-1-醇)反应以得到酯c2(例如,8-溴辛酸壬酯)。步骤1可以在例如N-(3-二甲基氨基丙基)-N'-乙基碳二亚胺盐酸盐、N,N-二异丙基乙胺和DMAP的存在下在有机溶剂(例如,二氯甲烷)中进行。醇e2(例如,十七烷-9-醇)可以通过使醛d2(例如,壬醛)与格氏试剂R3-MgX(例如,n-C8H17MgBr)经步骤2反应来获得。接下来,8-溴辛酸与醇e2(例如,十七烷-9-醇)反应以得到酯f2(例如,8-溴辛酸十七烷-9-基酯)。步骤3可以在例如N-(3-二甲基氨基丙基)-N'-乙基碳二亚胺盐酸盐、N,N-二异丙基乙胺和DMAP的存在下在有机溶剂(例如,二氯甲烷)中进行。接下来,酯f2与2-氨基乙烷-1-醇反应以得到胺g2(例如,8-((2-羟乙基)氨基)辛酸十七烷-9-基酯)。步骤4可以在i-Pr2EtN的存在下在乙醇中进行。然后,胺g2与酯c2(例如,8-溴辛酸壬酯)反应以得到化合物h2(例如,8-((2-羟乙基)(8-(壬氧基)-8-氧代辛基)氨基)辛酸十七烷-9-基酯)。步骤5可以在例如高温(诸如在约70℃-90℃,例如约80℃)下在碱(诸如无机碱(例如,K2CO3)或非亲核性有机碱(例如,i-Pr2EtN))和催化剂(例如,碘化物诸如KI或NaI)的存在下在有机溶剂(例如,CPME与MeCN的混合物)中进行。As shown in Scheme 2 above, acid a2 ( x3 is an integer between 1 and 7; eg, 8-bromooctanoic acid) is reacted with alcohol b2 (eg, nonan-1-ol) to give ester c2 (eg, , nonyl 8-bromooctanoate). Step 1 can be carried out in an organic solvent ( For example, dichloromethane). Alcohols e2 (eg, heptadecan-9-ol) can be obtained by reacting an aldehyde d2 (eg, nonanal) with a Grignard reagent R3 - MgX (eg, nC8H17MgBr ) via Step 2. Next, 8-bromooctanoic acid is reacted with alcohol e2 (eg, heptadecan-9-ol) to give ester f2 (eg, 8-bromooctanoic acid heptadecan-9-yl ester). Step 3 can be carried out in an organic solvent ( For example, dichloromethane). Next, ester f2 is reacted with 2-aminoethane-1-ol to give amine g2 (eg, heptadecan-9-yl 8-((2-hydroxyethyl)amino)octanoate). Step 4 can be carried out in ethanol in the presence of i - Pr2EtN. Then, amine g2 is reacted with ester c2 (eg, nonyl 8-bromooctanoate) to give compound h2 (eg, 8-((2-hydroxyethyl)(8-(nonyloxy)-8-oxooctyl) ) amino) heptadecan-9-yl octanoate). Step 5 can be performed in a base such as an inorganic base (eg, K 2 CO 3 ) or a non-nucleophilic organic base (eg, i-Pr 2 ) at, for example, elevated temperature (such as at about 70°C-90°C, eg, about 80°C). EtN)) and a catalyst (eg, an iodide such as KI or NaI) in an organic solvent (eg, a mixture of CPME and MeCN).
方案3Scenario 3
如以上方案3所示,卤代烷醇(x3是介于1与12之间的整数,例如,6-溴己烷-1-醇)与起始材料a3(x2是介于1与6之间的整数,例如,4-(己氧基)-4-氧代丁酸)反应以得到卤代二酯b3(例如,琥珀酸6-溴己基己酯)。化合物a3可以通过醇(例如,己烷-1-醇)与酸酐(例如,琥珀酸酐、二氢-2H-吡喃-2,6(3H)-二酮、3-(叔丁氧基)-3-氧代丙酸、4-(叔丁氧基)-3-甲基-4-氧代丁酸或4-(叔丁氧基)-2-甲基-4-氧代丁酸)反应来获得。步骤1可以在例如N-(3-二甲基氨基丙基)-N'-乙基碳二亚胺盐酸盐、N,N-二异丙基乙胺和DMAP的存在下在有机溶剂(例如,二氯甲烷)中进行。接下来,卤代二酯b3与胺c3(x4是介于5与13之间的整数,x5是介于1与5之间的整数,例如,8-((2-羟乙基)氨基)辛酸十七烷-9-基酯)反应以得到产物d3。步骤2可以在高温(例如,约90℃)下在碱(诸如无机碱(例如,K2CO3)和催化剂(例如,碘化物,诸如KI)以及醚溶剂(例如,环戊基甲基醚)的存在下在有机溶剂(例如,CPME与MeCN的混合物)中进行。As shown in Scheme 3 above, haloalkanol ( x3 is an integer between 1 and 12, eg, 6-bromohexane-1-ol) and starting material a3 ( x2 is between 1 and 6) an integer between, eg, 4-(hexyloxy)-4-oxobutyric acid) to give the halodiester b3 (eg, 6-bromohexylhexyl succinate). Compound a3 can pass through alcohol (for example, hexane-1-ol) and acid anhydride (for example, succinic anhydride, dihydro-2H-pyran-2,6(3H)-dione, 3-(tert-butoxy)- 3-oxopropionic acid, 4-(tert-butoxy)-3-methyl-4-oxobutyric acid or 4-(tert-butoxy)-2-methyl-4-oxobutyric acid) reaction to obtain. Step 1 can be carried out in an organic solvent ( For example, dichloromethane). Next, the halogenated diester b3 and the amine c3 (x 4 is an integer between 5 and 13, x 5 is an integer between 1 and 5, eg, 8-((2-hydroxyethyl) amino) heptadecan-9-yl octanoate) to give product d3. Step 2 can be carried out at elevated temperature (eg, about 90°C) in a base (such as an inorganic base (eg, K2CO3 ) and a catalyst (eg, an iodide such as KI) and an ether solvent (eg, cyclopentyl methyl ether) ) in an organic solvent (eg, a mixture of CPME and MeCN).
本领域普通技术人员将认识到,在以上方案中,某些步骤的次序可以互换。One of ordinary skill in the art will recognize that in the above schemes, the order of certain steps may be interchanged.
在某些方面,本公开还包括合成式(1-1)、(2-1)、(I-a)、(A)、(B)、(A-1)、(A-2)、(A-3)、(IA)、(IB)、(B-1)、(B-2)、(B-3)、(A-a)、(A-a1)、(A-a2)、(A-a3)、(A-b)、(A-b1)、(A-b2)、(A-b3)、(A-c)和(B-c)中任一个的化合物以及用于合成所述化合物的中间体的方法。In certain aspects, the present disclosure also includes the synthesis of formulae (1-1), (2-1), (I-a), (A), (B), (A-1), (A-2), (A- 3), (IA), (IB), (B-1), (B-2), (B-3), (A-a), (A-a1), (A-a2), (A-a3) Compounds of any of , (A-b), (A-b1), (A-b2), (A-b3), (A-c) and (B-c) and methods for synthesizing intermediates of said compounds.
在一些实施方案中,合成本公开的化合物的方法包括使式(X2)的化合物:与R1-Br反应以得到本公开的化合物,其中各变量如本文所定义。例如,m是5、6、7、8或9,优选5、7或9。例如,R5、R6和R7中的每一个是H。例如,M是-C(O)O-或-OC(O)-。例如,R4是未取代的C1-3烷基或-(CH2)nQ,其中n为2、3或4并且Q为OH、-NHC(S)N(R)2、-NHC(O)N(R)2、-N(R)C(O)R或-N(R)S(O)2R。例如,式(X2)的化合物与R1-Br的反应在碱(诸如无机碱(例如,K2CO3)或非亲核性有机碱(例如,i-Pr2EtN))的存在下进行。例如,所述反应是在无机碱(例如,K2CO3)和催化剂(例如,碘化物,诸如KI或NaI)的存在下进行。例如,所述反应是在高温(例如,在约50℃-100℃、70℃-90℃或约80℃)下进行。In some embodiments, methods of synthesizing the compounds of the present disclosure include making a compound of formula (X2): Reaction with R1 - Br to give compounds of the present disclosure, wherein the variables are as defined herein. For example, m is 5, 6, 7, 8 or 9, preferably 5, 7 or 9. For example, each of R 5 , R 6 and R 7 is H. For example, M is -C(O)O- or -OC(O)-. For example, R4 is unsubstituted C1-3 alkyl or -( CH2 )nQ, where n is 2, 3 or 4 and Q is OH, -NHC(S)N(R) 2 , -NHC( O)N(R) 2 , -N(R)C(O)R or -N(R)S(O ) 2R. For example, the reaction of a compound of formula (X2) with R 1 -Br is carried out in the presence of a base such as an inorganic base (eg, K 2 CO 3 ) or a non-nucleophilic organic base (eg, i-Pr 2 EtN) . For example, the reaction is carried out in the presence of an inorganic base (eg, K2CO3 ) and a catalyst (eg, an iodide such as KI or NaI). For example, the reaction is carried out at elevated temperature (eg, at about 50°C-100°C, 70°C-90°C, or about 80°C).
所述方法还可以包括使式(X1)的化合物:The method can also include making the compound of formula (X1):
与R4NH2反应以得到式(X2)的化合物,其中各变量如本文所定义。 Reaction with R4NH2 to give compounds of formula (X2), wherein the variables are as defined herein.
在一些实施方案中,中间体包括具有式(X1)和(X2)中的任一个的那些:其中各变量如本文所定义。例如,中间体包括8-溴辛酸十七烷-9-基酯和8-((2-羟乙基)氨基)辛酸十七烷-9-基酯及其形态形式(例如,结晶形式)。In some embodiments, intermediates include those of any of formulae (X1) and (X2): where the variables are as defined herein. For example, intermediates include heptadec-9-yl 8-bromooctanoate and heptadec-9-yl 8-((2-hydroxyethyl)amino)octanoate and morphological forms (eg, crystalline forms) thereof.
此外,还应了解,在现有技术范围内的本公开的任何特定实施方案可以明确地排除在任何一个或多个权利要求外。因为认为此类实施方案是本领域普通技术人员所已知的,即使本文没有明确提出排除,也可以排除它们。Furthermore, it is to be understood that any particular embodiment of the present disclosure that is within the scope of the prior art may be expressly excluded from any claim or claims. Because such embodiments are believed to be known to those of ordinary skill in the art, they may be excluded even if not explicitly stated herein.
所有引用的来源,例如本文引用的参考文献、出版物、数据库、数据库条目和技术,均以引用的方式并入本申请,即使在引用中没有明确说明。在引用的来源与本申请的表述有冲突的情况下,以本申请中的表述为准。All cited sources, such as references, publications, databases, database entries, and techniques cited herein, are incorporated herein by reference, even if not explicitly stated in the citation. In the event of a conflict between a cited source and the expression in the present application, the expression in the present application shall control.
实施例Example
实施例1:表1的化合物的合成Example 1: Synthesis of the compounds of Table 1
A.一般考虑A. General Considerations
除非另有说明,否则使用的所有溶剂和试剂都是商购获得的并且以原样使用。使用Bruker Ultrashield 300MHz仪器在300K下在CDCl3中记录1HNMR光谱。以相对于1H的TMS(0.00)的百万分率(ppm)报告化学位移。使用ISCO RediSep Rf Gold快速柱(粒度:20-40微米)在ISCO CombiFlash Rf+Lumen仪器上进行硅胶色谱。使用RediSep Rf Gold C18高效柱在ISCO CombiFlash Rf+Lumen仪器上进行反相色谱。使用具有DAD和ELSD的WatersAcquity UPLC仪器和ZORBAX快速分辨高清晰度(RRHD)SB-C18 LC柱(2.1mm,50mm,1.8μm)以及以1.2mL/min在5分钟内的含0.1%TFA的水中65%至100%乙腈的梯度,经由通过反相UPLC-MS(保留时间,RT,以分钟为单位)的分析确定所有的最终化合物纯度大于85%。注入体积为5μL,并且柱温为80℃。检测使用Waters SQD质谱仪(Milford,MA,USA)和蒸发光散射检测器基于电喷雾电离(ESI)以正离模式进行。Unless otherwise stated, all solvents and reagents used were obtained commercially and used as received. < 1 >HNMR spectra were recorded in CDCl3 at 300K using a Bruker Ultrashield 300MHz instrument. Chemical shifts are reported in parts per million (ppm) relative to 1 H TMS (0.00). Silica gel chromatography was performed on an ISCO CombiFlash Rf+Lumen instrument using an ISCO RediSep Rf Gold flash column (particle size: 20-40 microns). Reversed-phase chromatography was performed on an ISCO CombiFlash Rf+Lumen instrument using a RediSep Rf Gold C18 high performance column. Using a WatersAcquity UPLC instrument with DAD and ELSD and a ZORBAX Rapid Resolution High Definition (RRHD) SB-C18 LC column (2.1 mm, 50 mm, 1.8 μm) and 0.1% TFA in water at 1.2 mL/min over 5 minutes Gradient from 65% to 100% acetonitrile, all final compounds were determined to be greater than 85% pure via analysis by reverse phase UPLC-MS (retention time, RT, in minutes). The injection volume was 5 μL and the column temperature was 80°C. Detection was performed in positive ionization mode based on electrospray ionization (ESI) using a Waters SQD mass spectrometer (Milford, MA, USA) and an evaporative light scattering detector.
以下描述的程序可用于合成表1的化合物。The procedure described below can be used to synthesize the compounds of Table 1.
本文采用了以下缩写:The following abbreviations are used in this article:
THF:四氢呋喃THF: Tetrahydrofuran
MeCN:乙腈MeCN: Acetonitrile
LAH:氢化铝锂LAH: Lithium Aluminum Hydride
DCM:二氯甲烷DCM: dichloromethane
DMAP:4-二甲基氨基吡啶DMAP: 4-Dimethylaminopyridine
LDA:二异丙基酰胺锂LDA: Lithium Diisopropylamide
rt:室温rt: room temperature
DME:1,2-二甲氧基乙烷DME: 1,2-Dimethoxyethane
n-BuLi:正丁基锂n-BuLi: n-butyllithium
CPME:环戊基甲基醚CPME: cyclopentyl methyl ether
i-Pr2EtN:N,N-二异丙基乙胺i-Pr 2 EtN: N,N-diisopropylethylamine
AA.化合物6:8-((2-羟乙基)(6-(((壬氧基)羰基)氧基)己基)氨基)辛酸十七烷-9-基酯AA. Compound 6: Heptadecan-9-yl 8-((2-hydroxyethyl)(6-(((nonyloxy)carbonyl)oxy)hexyl)amino)octanoate
UPLC/ELSD:RT=3.12min。C43H85NO6的MS(ES):m/z(MH+)713.191UPLC/ELSD: RT=3.12 min. MS ( ES ) for C43H85NO6 : m/z (MH + ) 713.191
1H NMR(300MHz,CDCl3)δ:ppm 4.89(p,1H);4.17(t,4H);3.56(bm,2H);2.75-2.36(m,6H);2.30(t,2H);1.77-1.20(m,61H);0.90(m,9H)。 1 H NMR (300 MHz, CDCl 3 ) δ: ppm 4.89 (p, 1H); 4.17 (t, 4H); 3.56 (bm, 2H); 2.75-2.36 (m, 6H); 2.30 (t, 2H); 1.77 -1.20 (m, 61H); 0.90 (m, 9H).
AB.化合物25:8-((3-((2-(甲基氨基)-3,4-二氧代环丁-1-烯-1-基)氨基)丙基)(6-(((壬氧基)羰基)氧基)己基)氨基)辛酸十七烷-9-基酯(4-硝基苯基)碳酸6-溴己酯AB. Compound 25: 8-((3-((2-(methylamino)-3,4-dioxocyclobut-1-en-1-yl)amino)propyl)(6-((( Nonyloxy)carbonyl)oxy)hexyl)amino)octanoic acid heptadecan-9-yl ester(4-nitrophenyl)6-bromohexyl carbonate
在室温下,在装有磁力搅拌器的圆底烧瓶中,将氯甲酸4-硝基苯基酯(9.35g,46.39mmol)一次性添加到在DCM(100mL)中得6-溴-1-己醇(7g,38.658mmol)的溶液中。将反应保持在N2下并在10分钟内逐滴添加吡啶(4.69mL,57.988mmol)。之后在室温继续搅拌16小时。用水和DCM稀释反应物。将有机层分离,并且将水层用DCM洗涤。将合并的有机层用盐水洗涤,用Na2SO4干燥并真空蒸发。将残余物通过硅胶色谱(在己烷中的0%-100%乙酸乙酯的溶液)纯化,以得到(4-硝基苯基)碳酸6-溴己酯(10.3g,38.7mmol,77%)。1H NMR(300MHz,CDCl3)δ:ppm 8.29(d,2H);7.43(d,2H);4.33(t,2H);3.43(t,2H);2.02-1.69(m,4H);1.53(m,4H)。In a round bottom flask equipped with a magnetic stirrer, 4-nitrophenyl chloroformate (9.35 g, 46.39 mmol) was added in one portion in DCM (100 mL) at room temperature to give 6-bromo-1- in hexanol (7 g, 38.658 mmol). The reaction was kept under N2 and pyridine (4.69 mL, 57.988 mmol) was added dropwise over 10 minutes. Then stirring was continued for 16 hours at room temperature. The reaction was diluted with water and DCM. The organic layer was separated and the aqueous layer was washed with DCM. The combined organic layers were washed with brine, dried over Na2SO4 and evaporated in vacuo. The residue was purified by silica gel chromatography (0%-100% ethyl acetate in hexanes) to give (4-nitrophenyl)6-bromohexyl carbonate (10.3 g, 38.7 mmol, 77% ). 1 H NMR (300 MHz, CDCl 3 ) δ: ppm 8.29 (d, 2H); 7.43 (d, 2H); 4.33 (t, 2H); 3.43 (t, 2H); 2.02-1.69 (m, 4H); 1.53 (m, 4H).
壬基碳酸6-溴己酯6-Bromohexyl nonyl carbonate
在室温下,在N2下,在装有磁力搅拌器的圆底烧瓶中,将壬-1-醇(8.334g,57.774mmol)逐滴添加到在DCM(15mL)中的4-硝基苯基碳酸6-溴己酯(5g,14.443mmol)的溶液中。将反应保持在N2下并在10分钟内逐滴添加吡啶(1.46mL,18.054mmol)。之后一次性添加4-二甲基氨基吡啶(0.353g,2.889mmol)。使反应混合物在室温下搅拌16小时,然后用水和DCM稀释。将有机层分离,并且将水层用DCM洗涤。将合并的有机层用盐水洗涤,用Na2SO4干燥并真空蒸发。将残余物通过硅胶色谱(在己烷中的0%-100%乙酸乙酯的溶液)纯化,以得到壬基碳酸6-溴己酯(1.3g,3.7mmol,26%)。1H NMR(300MHz,CDCl3)δ:ppm 4.15(m,4H);3.43(t,2H);1.89(m,2H);1.71(m,4H);1.58-1.18(m,16H);0.90(m,3H)。Non-1-ol (8.334 g, 57.774 mmol) was added dropwise to 4-nitrobenzene in DCM ( 15 mL) at room temperature under N in a round bottom flask equipped with a magnetic stirrer in a solution of 6-bromohexyl carbonate (5 g, 14.443 mmol). The reaction was kept under N2 and pyridine (1.46 mL, 18.054 mmol) was added dropwise over 10 minutes. Then 4-dimethylaminopyridine (0.353 g, 2.889 mmol) was added in one portion. The reaction mixture was stirred at room temperature for 16 hours, then diluted with water and DCM. The organic layer was separated and the aqueous layer was washed with DCM. The combined organic layers were washed with brine, dried over Na2SO4 and evaporated in vacuo. The residue was purified by silica gel chromatography (0%-100% ethyl acetate in hexanes) to give 6-bromohexyl nonyl carbonate (1.3 g, 3.7 mmol, 26%). 1 H NMR (300 MHz, CDCl 3 ) δ: ppm 4.15 (m, 4H); 3.43 (t, 2H); 1.89 (m, 2H); 1.71 (m, 4H); 1.58-1.18 (m, 16H); 0.90 (m, 3H).
8-((3-((叔丁氧基羰基)氨基)丙基)氨基)辛酸十七烷-9-基酯Heptadecan-9-yl 8-((3-((tert-butoxycarbonyl)amino)propyl)amino)octanoate
将在500mL乙醇中的8-溴辛酸十七烷-9-基酯(69.2g,0.15摩尔)和(3-氨基丙基)氨基甲酸叔丁酯(130.6g,0.75mol)的溶液加热至65℃过夜。将反应混合物浓缩,并且将粗产物通过快速柱色谱(SiO2:甲醇/二氯甲烷0%-20%)纯化得到呈淡黄色油状物的8-((3-((叔丁氧基羰基)氨基))丙基)氨基)辛酸十七烷-9-基酯(62g,74%)。C33H66N2O4的MS(CI):m/z(MH+)555.5。1H NMR(300MHz,CDCl3):δppm 5.15(bs,1H);4.85(quint.,1H,J=6.0Hz);3.17(m,2H);2.65(t,2H,J=6.6Hz);2.56(t,2H,J=6.8Hz);2.26(t,2H,J=7.6Hz);1.68-1.56(m,6H);1.46(m,5H);1.43(s,9H);1.24(m,30H);0.86(t,6H,J=6.6Hz)。A solution of heptadec-9-yl 8-bromooctanoate (69.2 g, 0.15 mol) and tert-butyl (3-aminopropyl)carbamate (130.6 g, 0.75 mol) in 500 mL of ethanol was heated to 65 °C overnight. The reaction mixture was concentrated and the crude product was purified by flash column chromatography ( SiO2 : methanol/dichloromethane 0%-20%) to give 8-((3-((tert-butoxycarbonyl) as a pale yellow oil Amino))propyl)amino)heptadecan-9-yl octanoate (62 g, 74%). MS (CI) for C33H66N2O4 : m/z (MH<+ & gt ; ) 555.5. 1 H NMR (300 MHz, CDCl 3 ): δppm 5.15 (bs, 1H); 4.85 (quint., 1H, J=6.0 Hz); 3.17 (m, 2H); 2.65 (t, 2H, J=6.6 Hz); 2.56(t,2H,J=6.8Hz);2.26(t,2H,J=7.6Hz);1.68-1.56(m,6H);1.46(m,5H);1.43(s,9H);1.24(m , 30H); 0.86 (t, 6H, J=6.6Hz).
8-((3-((叔丁氧基羰基)氨基)丙基)(6-(((壬氧基)羰基)氧基)己基)氨基)辛酸十七烷-9-基酯8-((3-((tert-butoxycarbonyl)amino)propyl)(6-(((nonyloxy)carbonyl)oxy)hexyl)amino)octanoic acid heptadecan-9-yl ester
向在40mL环戊基甲基醚和乙腈的1:1混合物中的壬基碳酸6-溴己酯(0.633g,1.802mmol)和8-((3-((叔丁氧基羰基)氨基)丙基)氨基)辛酸十七烷-9-基酯(1g,1.802mmol)的溶液中添加碳酸钾(0.996g,7.208mmol)和碘化钾(0.329g,1.982mmol)。将反应加热至77℃并且搅拌16小时。将溶液冷却、过滤,并且将挥发物真空蒸发。将残余物通过硅胶色谱(0%-100%(在DCM中的20%MeOH、80%DCM、1%NH4OH)的溶液)纯化,以得到8-((3-((叔丁氧基羰基)氨基)丙基)(6-(((壬氧基)羰基)氧基)己基)氨基)辛酸十七烷-9-基酯(0.32g,0.39mmol,22%)。UPLC/ELSD:RT=3.02min。C49H96N2O7的MS(ES):m/z(MH+)826.300。1H NMR(300MHz,CDCl3)δ:ppm 5.65(bm,1H);4.88(m,1H);4.14(t,4H);3.19(bm,2H);3.00(bm,1H);2.52-2.22(m,7H);1.76-1.17(m,71H);0.90(m,9H)。To nonyl 6-bromohexyl carbonate (0.633 g, 1.802 mmol) and 8-((3-((tert-butoxycarbonyl)amino) in 40 mL of a 1:1 mixture of cyclopentyl methyl ether and acetonitrile To a solution of propyl)amino)octanoic acid heptadecan-9-yl ester (1 g, 1.802 mmol) was added potassium carbonate (0.996 g, 7.208 mmol) and potassium iodide (0.329 g, 1.982 mmol). The reaction was heated to 77°C and stirred for 16 hours. The solution was cooled, filtered, and the volatiles were evaporated in vacuo. The residue was purified by silica gel chromatography (0%-100% (20% MeOH, 80% DCM, 1% NH4OH ) in DCM) to give 8-((3-(((tert-butoxy) Carbonyl)amino)propyl)(6-(((nonyloxy)carbonyl)oxy)hexyl)amino)octanoic acid heptadecan-9-yl ester (0.32 g, 0.39 mmol, 22%). UPLC/ELSD: RT=3.02 min. MS (ES) for C49H96N2O7 : m/z (MH< ; + >) 826.300 . 1 H NMR (300 MHz, CDCl 3 ) δ: ppm 5.65 (bm, 1H); 4.88 (m, 1H); 4.14 (t, 4H); 3.19 (bm, 2H); 3.00 (bm, 1H); 2.52-2.22 (m, 7H); 1.76-1.17 (m, 71H); 0.90 (m, 9H).
8-((3-氨基丙基)(6-(((壬氧基)羰基)氧基)己基)氨基)辛酸十七烷-9-基酯8-((3-Aminopropyl)(6-(((nonyloxy)carbonyl)oxy)hexyl)amino)octanoic acid heptadecan-9-yl ester
向在3mL DCM中的8-((3-((叔丁氧基羰基)氨基)丙基)(6-(((壬氧基)羰基)氧基)己基)氨基)辛酸十七烷-9-基酯(320mg,0.39mmol)的溶液中添加三氟乙酸(0.6mL,7.8mmol),并将溶液在室温下搅拌16小时。在0℃下通过添加饱和碳酸氢钠来淬灭反应。将有机层用饱和碳酸氢钠溶液和盐水洗涤。用无水硫酸钠干燥后,真空除去溶剂,以得到8-((3-氨基丙基)(6-(((壬氧基)羰基)氧基)己基)氨基)辛酸十七烷-9-基酯(283mg,定量)。UPLC/ELSD:RT=2.39min。C44H88N2O5的MS(ES):m/z(MH+)726.142。1H NMR(300MHz,CDCl3)δ:ppm 4.87(m,1H);4.14(m,4H);3.21-2.97(m,4H);2.78(m,4H);2.30(m,2H);2.04(m,2H);1.80-1.17(m,60H);0.90(m,9H)。To 8-((3-((tert-butoxycarbonyl)amino)propyl)(6-(((nonyloxy)carbonyl)oxy)hexyl)amino)octanoic acid heptadecan-9 in 3 mL of DCM Trifluoroacetic acid (0.6 mL, 7.8 mmol) was added to a solution of -yl ester (320 mg, 0.39 mmol), and the solution was stirred at room temperature for 16 hours. The reaction was quenched by addition of saturated sodium bicarbonate at 0 °C. The organic layer was washed with saturated sodium bicarbonate solution and brine. After drying over anhydrous sodium sulfate, the solvent was removed in vacuo to give 8-((3-aminopropyl)(6-(((nonyloxy)carbonyl)oxy)hexyl)amino)octanoic acid heptadecan-9- base ester (283 mg, quantitative). UPLC/ELSD: RT=2.39 min. MS (ES) for C44H88N2O5 : m/z (MH< ; + >) 726.142 . 1 H NMR (300 MHz, CDCl 3 ) δ: ppm 4.87 (m, 1H); 4.14 (m, 4H); 3.21-2.97 (m, 4H); 2.78 (m, 4H); 2.30 (m, 2H); 2.04 (m, 2H); 1.80-1.17 (m, 60H); 0.90 (m, 9H).
3-甲氧基-4-(甲基氨基)环丁-烯-1,2-二酮3-Methoxy-4-(methylamino)cyclobut-ene-1,2-dione
向在100mL二乙醚中的3,4-二甲氧基-3-环丁烯-1,2-二酮(1g,7mmol)的溶液中添加在THF(3.8mL,7.6mmol)中的2M甲基胺的溶液并且几乎立即形成沉淀。将混合物在室温下搅拌24小时,然后过滤,将过滤固体用二乙醚洗涤并空气干燥。将过滤固体溶解于热EtOAc中,过滤,使滤液冷却至室温,然后冷却至0℃,以得到沉淀。将其通过过滤分离,用冷EtOAc洗涤,空气干燥,然后真空干燥,以得到呈白色固体的3-甲氧基-4-(甲基氨基)环丁-3-烯-1,2-二酮(0.70g,5mmol,73%)。1H NMR(300MHz,DMSO-d6)δ:ppm 8.50(br.d,1H,J=69Hz);4.27(s,3H);3.02(sdd,3H,J=42Hz,4.5Hz)。To a solution of 3,4-dimethoxy-3-cyclobutene-1,2-dione (1 g, 7 mmol) in 100 mL diethyl ether was added 2M methane in THF (3.8 mL, 7.6 mmol) base amine solution and a precipitate formed almost immediately. The mixture was stirred at room temperature for 24 hours, then filtered, and the filtered solids were washed with diethyl ether and air dried. The filtered solid was dissolved in hot EtOAc, filtered, and the filtrate was allowed to cool to room temperature and then to 0°C to give a precipitate. It was isolated by filtration, washed with cold EtOAc, air dried, then vacuum dried to give 3-methoxy-4-(methylamino)cyclobut-3-ene-1,2-dione as a white solid (0.70 g, 5 mmol, 73%). 1 H NMR (300 MHz, DMSO-d 6 ) δ: ppm 8.50 (br.d, 1H, J=69 Hz); 4.27 (s, 3H); 3.02 (sdd, 3H, J=42 Hz, 4.5 Hz).
8-((3-((2-(甲基氨基)-3,4-二氧代环丁-1-烯-1-基)氨基)丙基)(6-(壬氧基)羰基)氧基)己基)氨基)辛酸十七烷-9-基酯8-((3-((2-(methylamino)-3,4-dioxocyclobut-1-en-1-yl)amino)propyl)(6-(nonyloxy)carbonyl)oxy yl)hexyl)amino)heptadecan-9-yl octanoate
向在THF(2mL)和水(0.4mL)中的8-((3-氨基丙基)(6-(((壬氧基)羰基)氧基)己基)氨基)辛酸十七烷-9-基酯(0.2g,0.28mmol)的溶液中添加3-甲氧基-4-(甲基氨基)环丁-3-烯-1,2-二酮(0.06g,0.41mmol),将反应加热至67℃并搅拌20小时。使反应冷却至室温,在此期间澄清溶液变成混浊混合物。添加无水MgSO4,过滤反应并浓缩滤液。将残余物通过硅胶色谱(0%-100%(在DCM中的20%MeOH、80%DCM、1%NH4OH)的溶液)纯化,以得到8-((3-((2-(甲基氨基)-3,4-二氧代环丁-1-烯-1-基)氨基)丙基)(6-(((壬氧基)羰基)氧基)己基)氨基)辛酸十七烷-9-基酯(120mg,0.14mmol,50%)。UPLC/ELSD:RT=2.81min。C49H91N3O7的MS(ES):m/z(MH+)835.058。1H NMR(300MHz,CDCl3)δ:ppm 4.88(p,1H);4.14(m,4H);3.74(m,2H);3.40-2.90(m,9H);2.31(m,2H);2.12(m,2H);1.80-1.20(m,62H);0.90(m,9H)。To 8-((3-aminopropyl)(6-(((nonyloxy)carbonyl)oxy)hexyl)amino)octanoic acid heptadecan-9- in THF (2 mL) and water (0.4 mL) 3-methoxy-4-(methylamino)cyclobut-3-ene-1,2-dione (0.06 g, 0.41 mmol) was added to a solution of the base ester (0.2 g, 0.28 mmol) and the reaction was heated to 67°C and stirred for 20 hours. The reaction was allowed to cool to room temperature during which time the clear solution became a cloudy mixture. Anhydrous MgSO4 was added, the reaction was filtered and the filtrate was concentrated. The residue was purified by silica gel chromatography (0%-100% (20% MeOH, 80% DCM, 1% NH4OH ) in DCM) to give 8-((3-((2-(methyl (ylamino)-3,4-dioxocyclobut-1-en-1-yl)amino)propyl)(6-(((nonyloxy)carbonyl)oxy)hexyl)amino)octanoic acid heptadecane -9-ylester (120 mg, 0.14 mmol, 50%). UPLC/ELSD: RT=2.81 min. MS (ES) for C49H91N3O7 : m /z (MH< ; + >) 835.058 . 1 H NMR (300 MHz, CDCl 3 ) δ: ppm 4.88 (p, 1H); 4.14 (m, 4H); 3.74 (m, 2H); 3.40-2.90 (m, 9H); 2.31 (m, 2H); 2.12 (m, 2H); 1.80-1.20 (m, 62H); 0.90 (m, 9H).
AC.化合物64:8-((2-羟乙基)(6-((((3-戊基辛基)氧基)羰基)氧基)己基)氨基)辛酸十七烷-9-基酯AC. Compound 64: Heptadecan-9-yl 8-((2-hydroxyethyl)(6-((((3-pentyloctyl)oxy)carbonyl)oxy)hexyl)amino)octanoate
UPLC/ELSD:RT=3.04min。C47H93NO6的MS(ES):m/z(MH+)769.313UPLC/ELSD: RT=3.04 min. MS ( ES ) for C47H93NO6 : m/z (MH + ) 769.313
1H NMR(300MHz,CDCl3)δ:ppm 4.89(p,1H);4.14(m,4H);3.54(bm,2H);2.66-2.37(m,6H);2.30(m,2H);1.77-1.17(m,66H);0.91(m,12H)。 1 H NMR (300 MHz, CDCl 3 ) δ: ppm 4.89 (p, 1H); 4.14 (m, 4H); 3.54 (bm, 2H); 2.66-2.37 (m, 6H); 2.30 (m, 2H); 1.77 -1.17 (m, 66H); 0.91 (m, 12H).
AD.化合物65:8-((6-((((3-己基壬基)氧基)羰基)氧基)己基)(3-((2-(甲基氨基)-3,4-二氧代环丁-1-烯-1-基)氨基)丙基)氨基)辛酸十七烷-9-基酯3-己基壬-2-酸乙酯AD. Compound 65: 8-((6-((((3-hexylnonyl)oxy)carbonyl)oxy)hexyl)(3-((2-(methylamino)-3,4-dioxo) Cyclobut-1-en-1-yl)amino)propyl)amino)octanoic acid heptadecan-9-yl ester 3-hexylnonan-2-acid ethyl ester
在20分钟内向在THF(294mL)中的氢化钠(4.697g,117.4mmol)的悬浮液中逐滴添加膦酰基乙酸三乙酯(26.33g,117.4mmol),并将混合物在室温下搅拌直到气体逸出停止(大约30分钟)。将反应混合物冷却至0℃并且添加7-十三酮(10g,58.7mmol)。将反应物逐渐升温至室温,然后加热至回流并搅拌过夜。用饱和水性碳酸氢钠淬灭反应。将水相用乙醚萃取,并且将有机萃取物用盐水洗涤,用MgSO4干燥并浓缩。将粗物质通过硅胶色谱(0%-20%EtOAc:己烷)纯化,以得到呈透明油状物的3-己基壬-2-酸乙酯(6.7g,27.9mmol,47.5%)。1H NMR(300MHz,CDCl3)δ:ppm 5.63(s,1H);4.15(q,2H);2.61(t,2H);2.15(t,2H);1.53-1.20(m,19H);0.91(m,6H)。To a suspension of sodium hydride (4.697 g, 117.4 mmol) in THF (294 mL) was added triethyl phosphonoacetate (26.33 g, 117.4 mmol) dropwise over 20 min and the mixture was stirred at room temperature until gassed Escape stopped (about 30 minutes). The reaction mixture was cooled to 0 °C and 7-tridecone (10 g, 58.7 mmol) was added. The reaction was gradually warmed to room temperature, then heated to reflux and stirred overnight. The reaction was quenched with saturated aqueous sodium bicarbonate. The aqueous phase was extracted with ether, and the organic extracts were washed with brine, dried over MgSO4 and concentrated. The crude material was purified by silica gel chromatography (0%-20% EtOAc:hexanes) to give ethyl 3-hexylnon-2-acid (6.7 g, 27.9 mmol, 47.5%) as a clear oil. 1 H NMR (300 MHz, CDCl 3 ) δ: ppm 5.63 (s, 1H); 4.15 (q, 2H); 2.61 (t, 2H); 2.15 (t, 2H); 1.53-1.20 (m, 19H); 0.91 (m, 6H).
3-己基壬酸乙酯Ethyl 3-hexylnonanoate
在N2下,向含有在乙醇(20mL)中的Pearlmans催化剂(0.73g,5.2mmol)的浆液的烧瓶中添加在乙醇(5mL)中的3-己基壬-2-烯酸乙酯(6.975g,25.9mmol)的溶液。在H2(气球)下,将反应搅拌16小时。通过硅藻土塞过滤反应,并且真空蒸发滤液,以得到3-己基壬酸乙酯(6.7g,24.7mmol,95%)。残余物不经进一步纯化用于下一步。1H NMR(300MHz,CDCl3)δ:ppm 4.16(q,2H);2.23(d,2H);1.86(bs,1H);1.28(m,23H);0.90(m,6H).。To a flask containing a slurry of Pearlmans catalyst (0.73 g, 5.2 mmol) in ethanol (20 mL) was added ethyl 3-hexylnon-2-enoate (6.975 g) in ethanol (5 mL) under N2 , 25.9 mmol) solution. The reaction was stirred under H2 (balloon) for 16 hours. The reaction was filtered through a plug of celite, and the filtrate was evaporated in vacuo to give ethyl 3-hexylnonanoate (6.7 g, 24.7 mmol, 95%). The residue was used in the next step without further purification. 1 H NMR (300 MHz, CDCl 3 ) δ: ppm 4.16 (q, 2H); 2.23 (d, 2H); 1.86 (bs, 1H); 1.28 (m, 23H); 0.90 (m, 6H).
3-己基壬-1-醇3-Hexylnonan-1-ol
向在THF中的氢化铝锂(49.5mL的在THF中的1M溶液,49.5mmol)的溶液中添加在THF(20mL)中的3-己基壬酸乙酯(6.7g,24.7mmol)的溶液。将反应在室温下持续16小时。用饱和十水硫酸钠溶液淬灭反应。通过硅藻土塞过滤去除白色固体,并且真空蒸发滤液。将残余物通过快速色谱(ISCO)用在己烷中的0%-100%乙酸乙酯纯化,以获得3-己基壬-1-醇(5.62g,24.6mmol,99%)。1H NMR(300MHz,CDCl3)δ:ppm 3.69(t,2H);1.61-1.19(m,24H);0.91(m,6H)。To a solution of lithium aluminum hydride in THF (49.5 mL of a 1M solution in THF, 49.5 mmol) was added a solution of ethyl 3-hexylnonanoate (6.7 g, 24.7 mmol) in THF (20 mL). The reaction was continued at room temperature for 16 hours. The reaction was quenched with saturated sodium sulfate decahydrate solution. The white solid was removed by filtration through a plug of celite, and the filtrate was evaporated in vacuo. The residue was purified by flash chromatography (ISCO) with 0%-100% ethyl acetate in hexanes to obtain 3-hexylnon-1-ol (5.62 g, 24.6 mmol, 99%). 1 H NMR (300 MHz, CDCl 3 ) δ: ppm 3.69 (t, 2H); 1.61-1.19 (m, 24H); 0.91 (m, 6H).
8-((6-((((3-己基壬基)氧基)羰基)氧基)己基)(3-((2-(甲基氨基)-3,4-二氧代环丁-1-烯-1-基)氨基)丙基)氨基)辛酸十七烷-9-基酯8-((6-((((3-hexylnonyl)oxy)carbonyl)oxy)hexyl)(3-((2-(methylamino)-3,4-dioxocyclobutane-1) -En-1-yl)amino)propyl)amino)octanoic acid heptadecan-9-yl ester
使用3-己基壬-1-醇代替壬-1-醇与化合物25类似地制备化合物65。UPLC/ELSD:RT=3.13min。C55H103N3O7的MS(ES):m/z(MH+)919.429。1H NMR(300MHz,CDCl3)δ:ppm 4.87(p,1H);4.17(m,4H);3.68(bm,2H);3.30(m,3H);2.65-2.41(m,6H);2.31(m,2H);1.87-1.19(m,73H);0.90(m,12H)。Compound 65 was prepared analogously to compound 25 using 3-hexylnonan-1-ol in place of nonan-1-ol. UPLC/ELSD: RT=3.13 min. MS (ES) for C55H103N3O7 : m /z (MH< ; + >) 919.429 . 1 H NMR (300 MHz, CDCl 3 ) δ: ppm 4.87 (p, 1H); 4.17 (m, 4H); 3.68 (bm, 2H); 3.30 (m, 3H); 2.65-2.41 (m, 6H); 2.31 (m, 2H); 1.87-1.19 (m, 73H); 0.90 (m, 12H).
AE.化合物66:8-((6-((((3-己基壬基)氧基)羰基)氧基)己基)(2-羟乙基)氨基)辛酸十七烷-9-基酯AE. Compound 66: Heptadecan-9-yl 8-((6-((((3-hexylnonyl)oxy)carbonyl)oxy)hexyl)(2-hydroxyethyl)amino)octanoate
UPLC/ELSD:RT=3.16min。C49H97NO6的MS(ES):m/z(MH+)797.683UPLC/ELSD: RT=3.16 min. MS ( ES ) for C49H97NO6 : m/z (MH + ) 797.683
1H NMR(300MHz,CDCl3)δ:ppm 4.89(p,1H);4.14(m,4H);3.54(bm,2H);2.68-2.38(m,6H);2.30(m,2H);1.77-1.17(m,70H);0.90(m,12H)。 1 H NMR (300 MHz, CDCl 3 ) δ: ppm 4.89 (p, 1H); 4.14 (m, 4H); 3.54 (bm, 2H); 2.68-2.38 (m, 6H); 2.30 (m, 2H); 1.77 -1.17 (m, 70H); 0.90 (m, 12H).
AF.化合物67:8-((3-((2-(甲基氨基)-3,4-二氧代环丁-1-烯-1-基)氨基)丙基)(6-((((3-戊基辛基)氧基)羰基)氧基)己基)氨基)辛酸十七烷-9-基酯AF. Compound 67: 8-((3-((2-(methylamino)-3,4-dioxocyclobut-1-en-1-yl)amino)propyl)(6-((( (3-Pentyloctyl)oxy)carbonyl)oxy)hexyl)amino)heptadecan-9-yl octanoate
使用6-十一酮代替7-十三酮与化合物65类似地制备化合物67。UPLC/ELSD:RT=3.04min。C53H99N3O7的MS(ES):m/z(MH+)891.552。1H NMR(300MHz,CDCl3)δ:ppm 4.87(p,1H);4.17(m,4H);3.67(bm,2H);3.28(m,3H);2.68-2.38(m,6H);2.31(m,2H);1.86-1.18(m,69H);0.90(m,12H)。Compound 67 was prepared analogously to compound 65 using 6-undecanone in place of 7-tridecone. UPLC/ELSD: RT=3.04 min. MS (ES) for C53H99N3O7 : m /z (MH< ; + >) 891.552 . 1 H NMR (300 MHz, CDCl 3 ) δ: ppm 4.87 (p, 1H); 4.17 (m, 4H); 3.67 (bm, 2H); 3.28 (m, 3H); 2.68-2.38 (m, 6H); 2.31 (m, 2H); 1.86-1.18 (m, 69H); 0.90 (m, 12H).
实施例2:样品制剂在小鼠中诱导的hEPO表达和肝中的残留脂质水平Example 2: Sample formulation induced hEPO expression in mice and residual lipid levels in liver
为了评估本公开的脂质的表达效力和代谢稳定性,测量了在向小鼠施用本公开的纳米粒子(例如,负载LNP)后的肝细胞蛋白表达(hEPO)。To evaluate the expression potency and metabolic stability of the disclosed lipids, hepatocyte protein expression (hEPO) was measured following administration of the disclosed nanoparticles (eg, LNP-loaded) to mice.
向CD-1小鼠静脉内施用脂质纳米粒子(LNP),其包含作为磷脂的DSPC,作为结构性脂质的胆固醇,作为PEG脂质的PEG-1,根据式(A)、(A1)、(A2)、(A3)、(B)、(I)、(I')、(I-X)、(I-Y)、(I-Ya)(IA)、(Ia)、(Ib)、(Ic)、(Id)、(Ie)、(If)、(Ig)、(II)、(IIa)或(IIb)的化合物,以及编码hEPO的mRNA。注射后6小时测试血清中hEPO的浓度。测试的粒子具有约0.1-0.4之间的PDI、约82%-99%的包封效率和约56-145nm的粒径。所有测试的LNP均显示mRNA有效递送至肝细胞,24小时后小鼠肝中剩余脂质的量不同。Lipid nanoparticles (LNP) containing DSPC as phospholipid, cholesterol as structural lipid, PEG-1 as PEG lipid according to formula (A), (A1) were administered intravenously to CD-1 mice , (A2), (A3), (B), (I), (I'), (I-X), (I-Y), (I-Ya) (IA), (Ia), (Ib), (Ic) , (Id), (Ie), (If), (Ig), (II), (IIa) or (IIb), and mRNA encoding hEPO. The concentration of hEPO in serum was tested 6 hours after injection. The particles tested had a PDI between about 0.1-0.4, an encapsulation efficiency of about 82-99%, and a particle size of about 56-145 nm. All tested LNPs showed efficient delivery of mRNA to hepatocytes, with varying amounts of lipid remaining in mouse livers after 24 hours.
表2.通过施用包含本公开的脂质的LNP在小鼠中诱导的hEPO表达和肝中的残留脂质水平。Table 2. hEPO expression and residual lipid levels in liver induced in mice by administration of LNPs comprising lipids of the present disclosure.
a总剂量百分比的计算假设一只25g小鼠带有1.5g肝 aCalculation of the percentage of total dose assumes a 25g mouse with 1.5g liver
b>95%等于脂质代谢非常缓慢 b >95% equals very slow lipid metabolism
列举的实施方案Enumerated Embodiments
实施方案1.一种式(I1)的化合物:Embodiment 1. A compound of formula (I1):
或其N-氧化物, or its N-oxide,
或它们的盐或异构体,其中:or their salts or isomers, wherein:
R1选自由C5-30烷基、C5-20烯基、-R*YR*”、-YR*”和-R”M’R’组成的组;R 1 is selected from the group consisting of C 5-30 alkyl, C 5-20 alkenyl, -R*YR*", -YR*" and -R"M'R';
R2和R3独立地选自由H、C1-14烷基、C2-14烯基、-R*YR*”、-YR*”和-R*OR*”组成的组,或者R2和R3连同其所连接的原子一起形成杂环或碳环;R 2 and R 3 are independently selected from the group consisting of H, C 1-14 alkyl, C 2-14 alkenyl, -R*YR*", -YR*" and -R*OR*", or R 2 and R3 together with the atoms to which it is attached form a heterocyclic or carbocyclic ring;
R4选自由氢、C3-6碳环、-(CH2)nQ、-(CH2)nCHQR、-(CH2)oC(R12)2(CH2)n-oQ、-CHQR、-CQ(R)2、-C(O)NQR和未取代的C1-6烷基组成的组,其中Q选自碳环、杂环、-OR、-O(CH2)nN(R)2、-C(O)OR、-OC(O)R、-CX3、-CX2H、-CXH2、-CN、-N(R)2、-C(O)N(R)2、-N(R)C(O)R、-N(R)S(O)2R、-N(R)C(O)N(R)2、-N(R)C(S)N(R)2、-N(R)R8、-N(R)S(O)2R8、-O(CH2)nOR、-N(R)C(=NR9)N(R)2、-N(R)C(=CHR9)N(R)2、-OC(O)N(R)2、-N(R)C(O)OR、-N(OR)C(O)R、-N(OR)S(O)2R、-N(OR)C(O)OR、-N(OR)C(O)N(R)2、-N(OR)C(S)N(R)2、-N(OR)C(=NR9)N(R)2、-N(OR)C(=CHR9)N(R)2、-C(=NR9)N(R)2、-C(=NR9)R、-C(O)N(R)OR、-(CH2)nN(R)2、–C(R)N(R)2C(O)OR、NC(R)=R11、N(C=NR15)R11、NRC(C(O)NR14R14’)2、-NRC(O)(CH2)pC(O)NR14R14’和其中A是C6-10芳基或杂环;并且R 4 is selected from free hydrogen, C 3-6 carbocyclic ring, -(CH 2 ) n Q, -(CH 2 ) n CHQR, -(CH 2 ) o C(R 12 ) 2 (CH 2 ) no Q, -CHQR , -CQ(R) 2 , -C(O)NQR and unsubstituted C 1-6 alkyl groups, wherein Q is selected from carbocycle, heterocycle, -OR, -O(CH 2 ) n N( R) 2 , -C(O)OR, -OC(O)R, -CX 3 , -CX 2 H, -CXH 2 , -CN, -N(R) 2 , -C(O)N(R) 2 , -N(R)C(O)R, -N(R)S(O) 2 R, -N(R)C(O)N(R) 2 , -N(R)C(S)N (R) 2 , -N(R)R 8 , -N(R)S(O) 2 R 8 , -O(CH 2 ) n OR, -N(R)C(=NR 9 )N(R) 2 , -N(R)C(=CHR 9 )N(R) 2 , -OC(O)N(R) 2 , -N(R)C(O)OR, -N(OR)C(O) R, -N(OR)S(O) 2 R, -N(OR)C(O)OR, -N(OR)C(O)N(R) 2 , -N(OR)C(S)N (R) 2 , -N(OR)C(=NR 9 )N(R) 2 , -N(OR)C(=CHR 9 )N(R) 2 , -C(=NR 9 )N(R) 2 , -C(=NR 9 )R, -C(O)N(R)OR, -(CH 2 ) n N(R) 2 , -C(R)N(R) 2 C(O)OR, NC(R)=R 11 , N(C=NR 15 )R 11 , NRC(C(O)NR 14 R 14 ′) 2 , -NRC(O)(CH 2 ) p C(O)NR 14 R 14 'and wherein A is C 6-10 aryl or heterocycle; and
每个o独立地选自1、2、3和4;p是0、1、2、3或4;q是1、2、3或4;并且每个n独立地选自1、2、3、4和5;或each o is independently selected from 1, 2, 3, and 4; p is 0, 1, 2, 3, or 4; q is 1, 2, 3, or 4; and each n is independently selected from 1, 2, 3 , 4 and 5; or
R4是其中 R4 is in
Xa和Xb各自独立地是O或S;X a and X b are each independently O or S;
R10选自由H、卤代基、-OH、R、-N(R)2、-CN、-N3、-C(O)OH、-C(O)OR、-OC(O)R、-OR、-SR、-S(O)R、-S(O)OR、-S(O)2OR、-NO2、-S(O)2N(R)2、-N(R)S(O)2R、–NH(CH2)t1N(R)2、–NH(CH2)p1O(CH2)q1N(R)2、–NH(CH2)s1OR、–N((CH2)s1OR)2、-N(R)-碳环、-N(R)-杂环、-N(R)-芳基、-N(R)-杂芳基、-N(R)(CH2)t1-碳环、-N(R)(CH2)t1-杂环、-N(R)(CH2)t1-芳基、-N(R)(CH2)t1-杂芳基、碳环、杂环、芳基和杂芳基组成的组;R 10 is selected from H, halo, -OH, R, -N(R) 2 , -CN, -N 3 , -C(O)OH, -C(O)OR, -OC(O)R, -OR, -SR, -S(O)R, -S(O)OR, -S(O) 2 OR, -NO 2 , -S(O) 2 N(R) 2 , -N(R)S (O) 2 R, –NH(CH 2 ) t1 N(R) 2 , –NH(CH 2 ) p1 O(CH 2 ) q1 N(R) 2 , –NH(CH 2 ) s1 OR, –N( (CH 2 ) s1 OR) 2 , -N(R)-carbocycle, -N(R)-heterocycle, -N(R)-aryl, -N(R)-heteroaryl, -N(R )(CH 2 ) t1 -carbocycle , -N(R)(CH 2 ) t1 -heterocycle, -N(R)(CH 2 ) t1 -aryl, -N(R)(CH 2 ) t1 -heterocycle the group consisting of aryl, carbocycle, heterocycle, aryl and heteroaryl;
n2选自1、2、3、4、5、6、7、8、9和10;n2 is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9 and 10;
r是0或1;r is 0 or 1;
t1选自1、2、3、4和5;t 1 is selected from 1, 2, 3, 4 and 5;
p1选自1、2、3、4和5;p 1 is selected from 1, 2, 3, 4 and 5;
q1选自1、2、3、4和5;q 1 is selected from 1, 2, 3, 4 and 5;
s1选自1、2、3、4和5;s 1 is selected from 1, 2, 3, 4 and 5;
每个R5独立地选自由OH、C1-3烷基、C2-3烯基和H组成的组;each R 5 is independently selected from the group consisting of OH, C 1-3 alkyl, C 2-3 alkenyl and H;
每个R6独立地选自由OH、C1-3烷基、C2-3烯基和H组成的组;each R is independently selected from the group consisting of OH, C 1-3 alkyl, C 2-3 alkenyl and H ;
M选自-OC(O)O-、-OC(O)-NRM-C(O)O-、-O-M”-O-、-OC(O)N(RM)-、-N(RM)C(O)O-、-NRMC(O)NRM-、-O-N=C(RM)-、SO-、-OS-、-S(RM)2O-、-O-S(RM)2-、-S(O)O-、-OS(O)-,其中M”是键、-(CH2)zC(O)-、C1-13烷基、C2-13烯基、-B(R**)-、-Si(R**)2-、-S(R**)2-或-S(O)-,其中z是1、2、3或4;M is selected from -OC(O)O-, -OC(O)-NR M -C(O)O-, -OM"-O-, -OC(O)N(R M )-, -N(R M )C(O)O-, -NR M C(O)NR M -, -ON=C(R M )-, SO-, -OS-, -S(R M ) 2 O-, -OS( R M ) 2 -, -S(O)O-, -OS(O)-, wherein M" is a bond, -(CH 2 ) z C(O)-, C 1-13 alkyl, C 2-13 alkenyl, -B(R**)-, -Si(R**) 2- , -S(R**) 2- or -S(O)-, wherein z is 1, 2, 3 or 4;
M’选自–OC(O)O-、-C(O)O-、-OC(O)-、-OC(O)-M”-C(O)O-、-OC(O)-NRM-C(O)O-、-O-M”-O-、-C(O)N(RM)-、-N(RM)C(O)-、-OC(O)N(RM)-、-N(RM)C(O)O-、-NRMC(O)NRM-、-O-N=C(RM)-、-C(O)-、-C(S)-、-C(S)S-、-SC(S)-、-CH(OH)-、-P(O)(ORM)O-、-S(O)2-、-S-S-、-SO-、-OS-、S(RM)2O-、-O-S(RM)2-、-S(O)O-、-OS(O)-、芳基和杂芳基,其中M”是键、-(CH2)zC(O)-、C1-13烷基、C2-13烯基、-B(R**)-、-Si(R**)2-、-S(R**)2-或-S(O)-,其中z是1、2、3或4;M' is selected from -OC(O)O-, -C(O)O-, -OC(O)-, -OC(O)-M"-C(O)O-, -OC(O)-NR M -C(O)O-, -OM"-O-, -C(O)N(R M )-, -N(R M )C(O)-, -OC(O)N(R M ) -, -N(R M )C(O)O-, -NR M C(O)NR M -, -ON=C(R M )-, -C(O)-, -C(S)-, -C(S)S-, -SC(S)-, -CH(OH)-, -P(O)(OR M )O-, -S(O) 2 -, -SS-, -SO-, -OS-, S(R M ) 2 O-, -OS(R M ) 2 -, -S(O)O-, -OS(O)-, aryl and heteroaryl, where M" is a bond, -(CH 2 ) z C(O)-, C 1-13 alkyl, C 2-13 alkenyl, -B(R**)-, -Si(R**) 2 -, -S(R* *) 2- or -S(O)-, where z is 1, 2, 3 or 4;
R7选自由C1-3烷基、C2-3烯基和H组成的组;R 7 is selected from the group consisting of C 1-3 alkyl, C 2-3 alkenyl and H;
R8选自由C3-6碳环和杂环组成的组;R 8 is selected from the group consisting of C 3-6 carbocycles and heterocycles;
R9选自由H、CN、NO2、C1-6芳基、-OR、-S(O)2R、-S(O)2N(R)2、C2-6烯基、C3-6碳环和杂环组成的组;R 9 is selected from H, CN, NO 2 , C 1-6 aryl, -OR, -S(O) 2 R, -S(O) 2 N(R) 2 , C 2-6 alkenyl, C 3 - the group consisting of 6 carbocycles and heterocycles;
R11选自由C3-6碳环和杂环组成的组,其中C3-6碳环和杂环各自任选地被一个或多个R13取代;R 11 is selected from the group consisting of C 3-6 carbocycles and heterocycles, wherein each of the C 3-6 carbocycles and heterocycles is optionally substituted with one or more R 13 ;
R12选自由H、OH、C1-3烷基和C2-3烯基组成的组;R 12 is selected from the group consisting of H, OH, C 1-3 alkyl and C 2-3 alkenyl;
每个R13独立地选自由OH、氧代基、卤代基、C1-6烷基、C1-6烷氧基、C2-6烯基、C1-6烷基氨基、二-(C1-6烷基)氨基、氨基、酰胺基、氰基和硝基组成的组;Each R 13 is independently selected from OH, oxo, halo, C 1-6 alkyl, C 1-6 alkoxy, C 2-6 alkenyl, C 1-6 alkylamino, di- (C 1-6 alkyl) the group consisting of amino, amino, amido, cyano and nitro;
每个R14独立地选自由H、OH、C1-6烷基和C2-3烯基组成的组;each R 14 is independently selected from the group consisting of H, OH, C 1-6 alkyl and C 2-3 alkenyl;
每个R14’独立地选自由H、OH、C1-6烷基和C2-3烯基组成的组;each R 14 ' is independently selected from the group consisting of H, OH, C 1-6 alkyl and C 2-3 alkenyl;
R15独立地选自由H、OH、C1-6烷基和C2-3烯基组成的组;R 15 is independently selected from the group consisting of H, OH, C 1-6 alkyl and C 2-3 alkenyl;
每个R独立地选自由C1-6烷基、C1-3烷基-芳基、C2-3烯基和H组成的组;each R is independently selected from the group consisting of C1-6 alkyl, C1-3 alkyl-aryl, C2-3 alkenyl, and H;
RN是H或C1-3烷基;R N is H or C 1-3 alkyl;
每个R’独立地选自由C1-18烷基、C2-18烯基、-R*YR*”、-YR*”、(CH2)qOR*和H组成的组,each R' is independently selected from the group consisting of C 1-18 alkyl, C 2-18 alkenyl, -R*YR*", -YR*", (CH 2 ) q OR* and H,
并且每个q独立地选自1、2和3;and each q is independently selected from 1, 2 and 3;
每个RM独立地选自由H、C1-6烷基和C2-6烯基组成的组;each R M is independently selected from the group consisting of H, C 1-6 alkyl and C 2-6 alkenyl;
每个R”独立地选自由C3-15烷基和C3-15烯基组成的组;each R" is independently selected from the group consisting of C 3-15 alkyl and C 3-15 alkenyl;
每个R*”独立地选自由C1-15烷基和C2-15烯基组成的组;each R*" is independently selected from the group consisting of C 1-15 alkyl and C 2-15 alkenyl;
每个R*独立地选自由C1-12烷基和C2-12烯基组成的组;each R* is independently selected from the group consisting of C 1-12 alkyl and C 2-12 alkenyl;
每个R**独立地选自由H、OH、C1-12烷基、C2-12烯基、(CH2)qOR*和(CH2)qOH组成的组;each R** is independently selected from the group consisting of H, OH, C 1-12 alkyl, C 2-12 alkenyl, (CH 2 ) q OR*, and (CH 2 ) q OH;
每个Y独立地是C3-6碳环;each Y is independently a C3-6 carbocycle;
每个X独立地选自由F、Cl、Br和I组成的组;并且each X is independently selected from the group consisting of F, Cl, Br, and I; and
m选自5、6、7、8、9、10、11、12和13。m is selected from 5, 6, 7, 8, 9, 10, 11, 12 and 13.
实施方案2.一种式(I’1)的化合物:Embodiment 2. A compound of formula (I'1):
或其N-氧化物, or its N-oxide,
或它们的盐或异构体,其中:or their salts or isomers, wherein:
R1选自由C5-30烷基、C5-20烯基、-R*YR*”、-YR*”和-R”M’R’组成的组;R 1 is selected from the group consisting of C 5-30 alkyl, C 5-20 alkenyl, -R*YR*", -YR*" and -R"M'R';
R2和R3独立地选自由H、C1-14烷基、C2-14烯基、-R*YR*”、-YR*”和-R*OR*”组成的组,或者R2和R3连同其所连接的原子一起形成杂环或碳环;R 2 and R 3 are independently selected from the group consisting of H, C 1-14 alkyl, C 2-14 alkenyl, -R*YR*", -YR*" and -R*OR*", or R 2 and R3 together with the atoms to which it is attached form a heterocyclic or carbocyclic ring;
R4选自由氢、C3-6碳环、-(CH2)nQ、-(CH2)nCHQR、-(CH2)oC(R12)2(CH2)n-oQ、-CHQR、-CQ(R)2、-C(O)NQR和未取代的C1-6烷基组成的组,其中Q选自碳环、杂环、-OR、-O(CH2)nN(R)2、-C(O)OR、-OC(O)R、-CX3、-CX2H、-CXH2、-CN、-N(R)2、-C(O)N(R)2、-N(R)C(O)R、-N(R)S(O)2R、-N(R)C(O)N(R)2、-N(R)C(S)N(R)2、-N(R)R8、-N(R)S(O)2R8、-O(CH2)nOR、-N(R)C(=NR9)N(R)2、-N(R)C(=CHR9)N(R)2、-OC(O)N(R)2、-N(R)C(O)OR、-N(OR)C(O)R、-N(OR)S(O)2R、-N(OR)C(O)OR、-N(OR)C(O)N(R)2、-N(OR)C(S)N(R)2、-N(OR)C(=NR9)N(R)2、-N(OR)C(=CHR9)N(R)2、-C(=NR9)N(R)2、-C(=NR9)R、-C(O)N(R)OR、-(CH2)nN(R)2、–C(R)N(R)2C(O)OR、NC(R)=R11、N(C=NR15)R11、NRC(C(O)NR14R14’)2、-NRC(O)(CH2)pC(O)NR14R14’和其中A是C6-10芳基或杂环;并且R 4 is selected from free hydrogen, C 3-6 carbocyclic ring, -(CH 2 ) n Q, -(CH 2 ) n CHQR, -(CH 2 ) o C(R 12 ) 2 (CH 2 ) no Q, -CHQR , -CQ(R) 2 , -C(O)NQR and unsubstituted C 1-6 alkyl groups, wherein Q is selected from carbocycle, heterocycle, -OR, -O(CH 2 ) n N( R) 2 , -C(O)OR, -OC(O)R, -CX 3 , -CX 2 H, -CXH 2 , -CN, -N(R) 2 , -C(O)N(R) 2 , -N(R)C(O)R, -N(R)S(O) 2 R, -N(R)C(O)N(R) 2 , -N(R)C(S)N (R) 2 , -N(R)R 8 , -N(R)S(O) 2 R 8 , -O(CH 2 ) n OR, -N(R)C(=NR 9 )N(R) 2 , -N(R)C(=CHR 9 )N(R) 2 , -OC(O)N(R) 2 , -N(R)C(O)OR, -N(OR)C(O) R, -N(OR)S(O) 2 R, -N(OR)C(O)OR, -N(OR)C(O)N(R) 2 , -N(OR)C(S)N (R) 2 , -N(OR)C(=NR 9 )N(R) 2 , -N(OR)C(=CHR 9 )N(R) 2 , -C(=NR 9 )N(R) 2 , -C(=NR 9 )R, -C(O)N(R)OR, -(CH 2 ) n N(R) 2 , -C(R)N(R) 2 C(O)OR, NC(R)=R 11 , N(C=NR 15 )R 11 , NRC(C(O)NR 14 R 14 ′) 2 , -NRC(O)(CH 2 ) p C(O)NR 14 R 14 'and wherein A is C 6-10 aryl or heterocycle; and
每个o独立地选自1、2、3和4;p是0、1、2、3或4;q是1、2、3或4;并且每个n独立地选自1、2、3、4和5;或each o is independently selected from 1, 2, 3, and 4; p is 0, 1, 2, 3, or 4; q is 1, 2, 3, or 4; and each n is independently selected from 1, 2, 3 , 4 and 5; or
R4是其中 R4 is in
Xa和Xb各自独立地是O或S;X a and X b are each independently O or S;
R10选自由H、卤代基、-OH、R、-N(R)2、-CN、-N3、-C(O)OH、-C(O)OR、-OC(O)R、-OR、-SR、-S(O)R、-S(O)OR、-S(O)2OR、-NO2、-S(O)2N(R)2、-N(R)S(O)2R、–NH(CH2)t1N(R)2、–NH(CH2)p1O(CH2)q1N(R)2、–NH(CH2)s1OR、–N((CH2)s1OR)2、-N(R)-碳环、-N(R)-杂环、-N(R)-芳基、-N(R)-杂芳基、-N(R)(CH2)t1-碳环、-N(R)(CH2)t1-杂环、-N(R)(CH2)t1-芳基、-N(R)(CH2)t1-杂芳基、碳环、杂环、芳基和杂芳基组成的组;R 10 is selected from H, halo, -OH, R, -N(R) 2 , -CN, -N 3 , -C(O)OH, -C(O)OR, -OC(O)R, -OR, -SR, -S(O)R, -S(O)OR, -S(O) 2 OR, -NO 2 , -S(O) 2 N(R) 2 , -N(R)S (O) 2 R, –NH(CH 2 ) t1 N(R) 2 , –NH(CH 2 ) p1 O(CH 2 ) q1 N(R) 2 , –NH(CH 2 ) s1 OR, –N( (CH 2 ) s1 OR) 2 , -N(R)-carbocycle, -N(R)-heterocycle, -N(R)-aryl, -N(R)-heteroaryl, -N(R )(CH 2 ) t1 -carbocycle , -N(R)(CH 2 ) t1 -heterocycle, -N(R)(CH 2 ) t1 -aryl, -N(R)(CH 2 ) t1 -heterocycle the group consisting of aryl, carbocycle, heterocycle, aryl and heteroaryl;
n2选自1、2、3、4、5、6、7、8、9和10;n2 is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9 and 10;
r是0或1;r is 0 or 1;
t1选自1、2、3、4和5;t 1 is selected from 1, 2, 3, 4 and 5;
p1选自1、2、3、4和5;p 1 is selected from 1, 2, 3, 4 and 5;
q1选自1、2、3、4和5;q 1 is selected from 1, 2, 3, 4 and 5;
s1选自1、2、3、4和5;s 1 is selected from 1, 2, 3, 4 and 5;
每个R5独立地选自由OH、C1-3烷基、C2-3烯基和H组成的组;each R 5 is independently selected from the group consisting of OH, C 1-3 alkyl, C 2-3 alkenyl and H;
每个R6独立地选自由OH、C1-3烷基、C2-3烯基和H组成的组;each R is independently selected from the group consisting of OH, C 1-3 alkyl, C 2-3 alkenyl and H ;
M选自–OC(O)O-、-C(O)O-、-OC(O)-、-OC(O)-M”-C(O)O-、-OC(O)-NRM-C(O)O-、-O-M”-O-、-C(O)N(RM)-、-N(RM)C(O)-、-OC(O)N(RM)-、-N(RM)C(O)O-、-NRMC(O)NRM-、-O-N=C(RM)-、-C(O)-、-C(S)-、-C(S)S-、-SC(S)-、-CH(OH)-、-P(O)(ORM)O-、-S(O)2-、-S-S-,-SO-、-OS-、S(RM)2O-、-O-S(RM)2-、-S(O)O-、-OS(O)-、芳基和杂芳基,其中M”是键、-(CH2)zC(O)-、C1-13烷基、C2-13烯基、-B(R**)-、-Si(R**)2-、-S(R**)2-或-S(O)-,其中z是1、2、3、或4;M is selected from -OC(O)O-, -C(O)O-, -OC(O)-, -OC(O)-M"-C(O)O-, -OC(O)-NR M -C(O)O-, -OM"-O-, -C(O)N(R M )-, -N(R M )C(O)-, -OC(O)N(R M )- , -N(R M )C(O)O-, -NR M C(O)NR M -, -ON=C(R M )-, -C(O)-, -C(S)-, - C(S)S-, -SC(S)-, -CH(OH)-, -P(O)(OR M )O-, -S(O) 2 -, -SS-, -SO-, - OS-, S(R M ) 2 O-, -OS(R M ) 2 -, -S(O)O-, -OS(O)-, aryl and heteroaryl, where M" is a bond, - (CH 2 ) z C(O)-, C 1-13 alkyl, C 2-13 alkenyl, -B(R**)-, -Si(R**) 2 -, -S(R** ) 2- or -S(O)-, wherein z is 1, 2, 3, or 4;
M’选自–OC(O)O-、-OC(O)-NRM-C(O)O-、-O-M”-O-、-OC(O)N(RM)-、-N(RM)C(O)O-、-NRMC(O)NRM-、-O-N=C(RM)-、SO-、-OS-、-S(RM)2O-、-O-S(RM)2-、-S(O)O-、-OS(O)-,其中M”是键、-(CH2)zC(O)-、C1-13烷基、C2-13烯基、-B(R**)-、-Si(R**)2-、-S(R**)2-或-S(O)-,其中z是1、2、3、或4;M' is selected from -OC(O)O-, -OC(O)-NR M -C(O)O-, -OM"-O-, -OC(O)N(R M )-, -N( R M )C(O)O-, -NR M C(O)NR M -, -ON=C(R M )-, SO-, -OS-, -S(R M ) 2 O-, -OS (R M ) 2 -, -S(O)O-, -OS(O)-, wherein M" is a bond, -(CH 2 ) z C(O)-, C 1-13 alkyl, C 2- 13Alkenyl , -B(R**)-, -Si(R**) 2- , -S(R**) 2- or -S(O)-, wherein z is 1, 2, 3, or 4;
R7选自由C1-3烷基、C2-3烯基和H组成的组;R 7 is selected from the group consisting of C 1-3 alkyl, C 2-3 alkenyl and H;
R8选自由C3-6碳环和杂环组成的组;R 8 is selected from the group consisting of C 3-6 carbocycles and heterocycles;
R9选自由H、CN、NO2、C1-6芳基、-OR、-S(O)2R、-S(O)2N(R)2、C2-6烯基、C3-6碳环和杂环组成的组;R 9 is selected from H, CN, NO 2 , C 1-6 aryl, -OR, -S(O) 2 R, -S(O) 2 N(R) 2 , C 2-6 alkenyl, C 3 - the group consisting of 6 carbocycles and heterocycles;
R11选自由C3-6碳环和杂环组成的组,其中C3-6碳环和杂环各自任选地被一个或多个R13取代;R 11 is selected from the group consisting of C 3-6 carbocycles and heterocycles, wherein each of the C 3-6 carbocycles and heterocycles is optionally substituted with one or more R 13 ;
R12选自由H、OH、C1-3烷基和C2-3烯基组成的组;R 12 is selected from the group consisting of H, OH, C 1-3 alkyl and C 2-3 alkenyl;
每个R13独立地选自由OH、氧代基、卤代基、C1-6烷基、C1-6烷氧基、C2-6烯基、C1-6烷基氨基、二-(C1-6烷基)氨基、氨基、酰胺基、氰基和硝基组成的组;Each R 13 is independently selected from OH, oxo, halo, C 1-6 alkyl, C 1-6 alkoxy, C 2-6 alkenyl, C 1-6 alkylamino, di- (C 1-6 alkyl) the group consisting of amino, amino, amido, cyano and nitro;
每个R14独立地选自由H、OH、C1-6烷基和C2-3烯基组成的组;each R 14 is independently selected from the group consisting of H, OH, C 1-6 alkyl and C 2-3 alkenyl;
每个R14’独立地选自由H、OH、C1-6烷基和C2-3烯基组成的组;each R 14 ' is independently selected from the group consisting of H, OH, C 1-6 alkyl and C 2-3 alkenyl;
R15独立地选自由H、OH、C1-6烷基和C2-3烯基组成的组;R 15 is independently selected from the group consisting of H, OH, C 1-6 alkyl and C 2-3 alkenyl;
每个R独立地选自由C1-6烷基、C1-3烷基-芳基、C2-3烯基和H组成的组;each R is independently selected from the group consisting of C1-6 alkyl, C1-3 alkyl-aryl, C2-3 alkenyl, and H;
RN是H或C1-3烷基;R N is H or C 1-3 alkyl;
每个R’独立地选自由C1-18烷基、C2-18烯基、-R*YR*”、-YR*”、(CH2)qOR*和H组成的组,each R' is independently selected from the group consisting of C 1-18 alkyl, C 2-18 alkenyl, -R*YR*", -YR*", (CH 2 ) q OR* and H,
并且每个q独立地选自1、2和3;and each q is independently selected from 1, 2 and 3;
每个RM独立地选自由H、C1-6烷基和C2-6烯基组成的组;each R M is independently selected from the group consisting of H, C 1-6 alkyl and C 2-6 alkenyl;
每个R”独立地选自由C3-15烷基和C3-15烯基组成的组;each R" is independently selected from the group consisting of C 3-15 alkyl and C 3-15 alkenyl;
每个R*”独立地选自由C1-15烷基和C2-15烯基组成的组;each R*" is independently selected from the group consisting of C 1-15 alkyl and C 2-15 alkenyl;
每个R*独立地选自由C1-12烷基和C2-12烯基组成的组;each R* is independently selected from the group consisting of C 1-12 alkyl and C 2-12 alkenyl;
每个R**独立地选自由H、OH、C1-12烷基、C2-12烯基、(CH2)qOR*和(CH2)qOH组成的组;each R** is independently selected from the group consisting of H, OH, C 1-12 alkyl, C 2-12 alkenyl, (CH 2 ) q OR*, and (CH 2 ) q OH;
每个Y独立地是C3-6碳环;each Y is independently a C3-6 carbocycle;
每个X独立地选自由F、Cl、Br和I组成的组;并且each X is independently selected from the group consisting of F, Cl, Br, and I; and
m选自5、6、7、8、9、10、11、12和13。m is selected from 5, 6, 7, 8, 9, 10, 11, 12 and 13.
实施方案3.一种式(IX1)的化合物:Embodiment 3. A compound of formula (IX1):
或其N-氧化物,或它们的盐或异构体,其中: or their N-oxides, or their salts or isomers, wherein:
R1选自由C5-30烷基、C5-20烯基、-R*YR*”、-YR*”和-R”M’R’组成的组;R 1 is selected from the group consisting of C 5-30 alkyl, C 5-20 alkenyl, -R*YR*", -YR*" and -R"M'R';
R2和R3独立地选自由H、C1-14烷基、C2-14烯基、-R*YR*”、-YR*”和-R*OR*”组成的组,或者R2和R3连同其所连接的原子一起形成杂环或碳环;R 2 and R 3 are independently selected from the group consisting of H, C 1-14 alkyl, C 2-14 alkenyl, -R*YR*", -YR*" and -R*OR*", or R 2 and R3 together with the atoms to which it is attached form a heterocyclic or carbocyclic ring;
R4选自由氢、C3-6碳环、-(CH2)nQ、-(CH2)nCHQR、-(CH2)oC(R12)2(CH2)n-oQ、-CHQR、-CQ(R)2、-C(O)NQR和未取代的C1-6烷基组成的组,其中Q选自碳环、杂环、-OR、-O(CH2)nN(R)2、-C(O)OR、-OC(O)R、-CX3、-CX2H、-CXH2、-CN、-N(R)2、-C(O)N(R)2、-N(R)C(O)R、-N(R)S(O)2R、-N(R)C(O)N(R)2、-N(R)C(S)N(R)2、-N(R)R8、-N(R)S(O)2R8、-O(CH2)nOR、-N(R)C(=NR9)N(R)2、-N(R)C(=CHR9)N(R)2、-OC(O)N(R)2、-N(R)C(O)OR、-N(OR)C(O)R、-N(OR)S(O)2R、-N(OR)C(O)OR、-N(OR)C(O)N(R)2、-N(OR)C(S)N(R)2、-N(OR)C(=NR9)N(R)2、-N(OR)C(=CHR9)N(R)2、-C(=NR9)N(R)2、-C(=NR9)R、-C(O)N(R)OR、-(CH2)nN(R)2、–C(R)N(R)2C(O)OR、NC(R)=R11、N(C=NR15)R11、NRC(C(O)NR14R14’)2、-NRC(O)(CH2)pC(O)NR14R14’和其中A是C6-10芳基或杂环;并且R 4 is selected from free hydrogen, C 3-6 carbocyclic ring, -(CH 2 ) n Q, -(CH 2 ) n CHQR, -(CH 2 ) o C(R 12 ) 2 (CH 2 ) no Q, -CHQR , -CQ(R) 2 , -C(O)NQR and unsubstituted C 1-6 alkyl groups, wherein Q is selected from carbocycle, heterocycle, -OR, -O(CH 2 ) n N( R) 2 , -C(O)OR, -OC(O)R, -CX 3 , -CX 2 H, -CXH 2 , -CN, -N(R) 2 , -C(O)N(R) 2 , -N(R)C(O)R, -N(R)S(O) 2 R, -N(R)C(O)N(R) 2 , -N(R)C(S)N (R) 2 , -N(R)R 8 , -N(R)S(O) 2 R 8 , -O(CH 2 ) n OR, -N(R)C(=NR 9 )N(R) 2 , -N(R)C(=CHR 9 )N(R) 2 , -OC(O)N(R) 2 , -N(R)C(O)OR, -N(OR)C(O) R, -N(OR)S(O) 2 R, -N(OR)C(O)OR, -N(OR)C(O)N(R) 2 , -N(OR)C(S)N (R) 2 , -N(OR)C(=NR 9 )N(R) 2 , -N(OR)C(=CHR 9 )N(R) 2 , -C(=NR 9 )N(R) 2 , -C(=NR 9 )R, -C(O)N(R)OR, -(CH 2 ) n N(R) 2 , -C(R)N(R) 2 C(O)OR, NC(R)=R 11 , N(C=NR 15 )R 11 , NRC(C(O)NR 14 R 14 ′) 2 , -NRC(O)(CH 2 ) p C(O)NR 14 R 14 'and wherein A is C 6-10 aryl or heterocycle; and
每个o独立地选自1、2、3和4;p是0、1、2、3或4;q是1、2、3或4;并且每个n独立地选自1、2、3、4和5;或each o is independently selected from 1, 2, 3, and 4; p is 0, 1, 2, 3, or 4; q is 1, 2, 3, or 4; and each n is independently selected from 1, 2, 3 , 4 and 5; or
R4是其中 R4 is in
Xa和Xb各自独立地是O或S;X a and X b are each independently O or S;
R10选自由H、卤代基、-OH、R、-N(R)2、-CN、-N3、-C(O)OH、-C(O)OR、-OC(O)R、-OR、-SR、-S(O)R、-S(O)OR、-S(O)2OR、-NO2、-S(O)2N(R)2、-N(R)S(O)2R、–NH(CH2)t1N(R)2、–NH(CH2)p1O(CH2)q1N(R)2、–NH(CH2)s1OR、–N((CH2)s1OR)2、-N(R)-碳环、-N(R)-杂环、-N(R)-芳基、-N(R)-杂芳基、-N(R)(CH2)t1-碳环、-N(R)(CH2)t1-杂环、-N(R)(CH2)t1-芳基、-N(R)(CH2)t1-杂芳基、碳环、杂环、芳基和杂芳基组成的组;R 10 is selected from H, halo, -OH, R, -N(R) 2 , -CN, -N 3 , -C(O)OH, -C(O)OR, -OC(O)R, -OR, -SR, -S(O)R, -S(O)OR, -S(O) 2 OR, -NO 2 , -S(O) 2 N(R) 2 , -N(R)S (O) 2 R, –NH(CH 2 ) t1 N(R) 2 , –NH(CH 2 ) p1 O(CH 2 ) q1 N(R) 2 , –NH(CH 2 ) s1 OR, –N( (CH 2 ) s1 OR) 2 , -N(R)-carbocycle, -N(R)-heterocycle, -N(R)-aryl, -N(R)-heteroaryl, -N(R )(CH 2 ) t1 -carbocycle , -N(R)(CH 2 ) t1 -heterocycle, -N(R)(CH 2 ) t1 -aryl, -N(R)(CH 2 ) t1 -heterocycle the group consisting of aryl, carbocycle, heterocycle, aryl and heteroaryl;
n2选自1、2、3、4、5、6、7、8、9和10;n2 is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9 and 10;
r是0或1;r is 0 or 1;
t1选自1、2、3、4和5;t 1 is selected from 1, 2, 3, 4 and 5;
p1选自1、2、3、4和5;p 1 is selected from 1, 2, 3, 4 and 5;
q1选自1、2、3、4和5;q 1 is selected from 1, 2, 3, 4 and 5;
s1选自1、2、3、4和5;s 1 is selected from 1, 2, 3, 4 and 5;
每个R5独立地选自由OH、C1-3烷基、C2-3烯基和H组成的组;each R 5 is independently selected from the group consisting of OH, C 1-3 alkyl, C 2-3 alkenyl and H;
每个R6独立地选自由OH、C1-3烷基、C2-3烯基和H组成的组;each R is independently selected from the group consisting of OH, C 1-3 alkyl, C 2-3 alkenyl and H ;
M’选自–OC(O)O-、-C(O)O-、-OC(O)-、-OC(O)-M”-C(O)O-、-OC(O)-NRM-C(O)O-、-O-M”-O-、-C(O)N(RM)-、-N(RM)C(O)-、-OC(O)N(RM)-、-N(RM)C(O)O-、-NRMC(O)NRM-、-O-N=C(RM)-、-C(O)-、-C(S)-、-C(S)S-、-SC(S)-、-CH(OH)-、-P(O)(ORM)O-、-S(O)2-、-S-S-、-SO-、-OS-、S(RM)2O-、-O-S(RM)2-、-S(O)O-、-OS(O)-、芳基和杂芳基,其中M”是键、-(CH2)zC(O)-、C1-13烷基、C2-13烯基、-B(R**)-、-Si(R**)2-、-S(R**)2-或-S(O)-,其中z是1、2、3或4;M' is selected from -OC(O)O-, -C(O)O-, -OC(O)-, -OC(O)-M"-C(O)O-, -OC(O)-NR M -C(O)O-, -OM"-O-, -C(O)N(R M )-, -N(R M )C(O)-, -OC(O)N(R M ) -, -N(R M )C(O)O-, -NR M C(O)NR M -, -ON=C(R M )-, -C(O)-, -C(S)-, -C(S)S-, -SC(S)-, -CH(OH)-, -P(O)(OR M )O-, -S(O) 2 -, -SS-, -SO-, -OS-, S(R M ) 2 O-, -OS(R M ) 2 -, -S(O)O-, -OS(O)-, aryl and heteroaryl, where M" is a bond, -(CH 2 ) z C(O)-, C 1-13 alkyl, C 2-13 alkenyl, -B(R**)-, -Si(R**) 2 -, -S(R* *) 2- or -S(O)-, where z is 1, 2, 3 or 4;
R7选自由C1-3烷基、C2-3烯基和H组成的组;R 7 is selected from the group consisting of C 1-3 alkyl, C 2-3 alkenyl and H;
R8选自由C3-6碳环和杂环组成的组;R 8 is selected from the group consisting of C 3-6 carbocycles and heterocycles;
R9选自由H、CN、NO2、C1-6芳基、-OR、-S(O)2R、-S(O)2N(R)2、C2-6烯基、C3-6碳环和杂环组成的组;R 9 is selected from H, CN, NO 2 , C 1-6 aryl, -OR, -S(O) 2 R, -S(O) 2 N(R) 2 , C 2-6 alkenyl, C 3 - the group consisting of 6 carbocycles and heterocycles;
R11选自由C3-6碳环和杂环组成的组,其中C3-6碳环和杂环各自任选地被一个或多个R13取代;R 11 is selected from the group consisting of C 3-6 carbocycles and heterocycles, wherein each of the C 3-6 carbocycles and heterocycles is optionally substituted with one or more R 13 ;
R12选自由H、OH、C1-3烷基和C2-3烯基组成的组;R 12 is selected from the group consisting of H, OH, C 1-3 alkyl and C 2-3 alkenyl;
每个R13独立地选自由OH、氧代基、卤代基、C1-6烷基、C1-6烷氧基、C2-6烯基、C1-6烷基氨基、二-(C1-6烷基)氨基、氨基、酰胺基、氰基和硝基组成的组;Each R 13 is independently selected from OH, oxo, halo, C 1-6 alkyl, C 1-6 alkoxy, C 2-6 alkenyl, C 1-6 alkylamino, di- (C 1-6 alkyl) the group consisting of amino, amino, amido, cyano and nitro;
每个R14独立地选自由H、OH、C1-6烷基和C2-3烯基组成的组;each R 14 is independently selected from the group consisting of H, OH, C 1-6 alkyl and C 2-3 alkenyl;
每个R14’独立地选自由H、OH、C1-6烷基和C2-3烯基组成的组;each R 14 ' is independently selected from the group consisting of H, OH, C 1-6 alkyl and C 2-3 alkenyl;
R15独立地选自由H、OH、C1-6烷基和C2-3烯基组成的组;R 15 is independently selected from the group consisting of H, OH, C 1-6 alkyl and C 2-3 alkenyl;
每个R独立地选自由C1-6烷基、C1-3烷基-芳基、C2-3烯基和H组成的组;each R is independently selected from the group consisting of C1-6 alkyl, C1-3 alkyl-aryl, C2-3 alkenyl, and H;
RN是H或C1-3烷基;R N is H or C 1-3 alkyl;
每个R’独立地选自由C1-18烷基、C2-18烯基、-R*YR*”、-YR*”、(CH2)qOR*和H组成的组,each R' is independently selected from the group consisting of C 1-18 alkyl, C 2-18 alkenyl, -R*YR*", -YR*", (CH 2 ) q OR* and H,
并且每个q独立地选自1、2和3;and each q is independently selected from 1, 2 and 3;
每个RM独立地选自由H、C1-6烷基和C2-6烯基组成的组;each R M is independently selected from the group consisting of H, C 1-6 alkyl and C 2-6 alkenyl;
每个R”独立地选自由C3-15烷基和C3-15烯基组成的组;each R" is independently selected from the group consisting of C 3-15 alkyl and C 3-15 alkenyl;
每个R*”独立地选自由C1-15烷基和C2-15烯基组成的组;each R*" is independently selected from the group consisting of C 1-15 alkyl and C 2-15 alkenyl;
每个R*独立地选自由C1-12烷基和C2-12烯基组成的组;each R* is independently selected from the group consisting of C 1-12 alkyl and C 2-12 alkenyl;
每个R**独立地选自由H、OH、C1-12烷基、C2-12烯基、(CH2)qOR*和(CH2)qOH组成的组;each R** is independently selected from the group consisting of H, OH, C 1-12 alkyl, C 2-12 alkenyl, (CH 2 ) q OR*, and (CH 2 ) q OH;
每个Y独立地是C3-6碳环;each Y is independently a C3-6 carbocycle;
每个X独立地选自由F、Cl、Br和I组成的组;并且each X is independently selected from the group consisting of F, Cl, Br, and I; and
m选自5、6、7、8、9、10、11、12和13。m is selected from 5, 6, 7, 8, 9, 10, 11, 12 and 13.
实施方案4.如前述实施方案中任一项所述的化合物,其中M和M'中的至少一个是-OC(O)O-。Embodiment 4. The compound of any of the preceding embodiments, wherein at least one of M and M' is -OC(O)O-.
实施方案5.如前述实施方案中任一项所述的化合物,其中M和M'中的至少一个是-OC(O)N(RM)-或-N(RM)C(O)O-。Embodiment 5. The compound of any one of the preceding embodiments, wherein at least one of M and M' is -OC(O)N(R M )- or -N(R M )C(O)O -.
实施方案6.如前述实施方案中任一项所述的化合物,其中M和M'中的至少一个是-NRMC(O)NRM-。Embodiment 6. The compound of any of the preceding embodiments, wherein at least one of M and M ' is -NRMC(O) NRM- .
实施方案7.如前述实施方案中任一项所述的化合物,其中M和M'中的至少一个是-O-N=C(RM)-。Embodiment 7. The compound of any of the preceding embodiments, wherein at least one of M and M' is -ON=C(R M )-.
实施方案8.如前述实施方案中任一项所述的化合物,其中M和M'中的至少一个是-O-M”-O-,其中每个M”独立地选自C1-13烷基、C2-13烯基、-B(R**)-和-Si(R**)2-。Embodiment 8. The compound of any one of the preceding embodiments, wherein at least one of M and M' is -OM"-O-, wherein each M" is independently selected from C 1-13 alkyl, C 2-13 alkenyl, -B(R**)- and -Si(R**) 2 -.
实施方案9.如前述实施方案中任一项所述的化合物,其中M和M'中的至少一个是–O-M”-O-,并且M”是C1-3烷基。Embodiment 9. The compound of any one of the preceding embodiments, wherein at least one of M and M' is -OM"-O-, and M" is C1-3 alkyl.
实施方案10.如前述实施方案中任一项所述的化合物,其中M”是甲基。Embodiment 10. The compound of any of the preceding embodiments, wherein M" is methyl.
实施方案11.如前述实施方案中任一项所述的化合物,其中M”是支链C1-3烷基。Embodiment 11. The compound of any of the preceding embodiments, wherein M" is branched C1-3 alkyl.
实施方案12.如前述实施方案中任一项所述的化合物,其中M和M'中的至少一个是–O-M”-O-,并且M”是-(CH2)zC(O)-。Embodiment 12. The compound of any of the preceding embodiments, wherein at least one of M and M' is -OM"-O-, and M" is -( CH2 ) zC (O)-.
实施方案13.如前述实施方案中任一项所述的化合物,其中z是3。Embodiment 13. The compound of any of the preceding embodiments, wherein z is 3.
实施方案14.如前述实施方案中任一项所述的化合物,其中M和M'中的至少一个是 Embodiment 14. The compound of any one of the preceding embodiments, wherein at least one of M and M' is
实施方案15.如前述实施方案中任一项所述的化合物,其中M和M'中的至少一个是 Embodiment 15. The compound of any one of the preceding embodiments, wherein at least one of M and M' is
实施方案16.如前述实施方案中任一项所述的化合物,其中M和M'中的至少一个是-O-M”-O-,其中每个M”独立地选自-B(R**)-和-Si(R**)2-。Embodiment 16. The compound of any one of the preceding embodiments, wherein at least one of M and M' is -OM"-O-, wherein each M" is independently selected from -B(R**) - and -Si(R**) 2 -.
实施方案17.如前述实施方案中任一项所述的化合物,其中每个R**是C1-3烷基。Embodiment 17. The compound of any of the preceding embodiments, wherein each R** is C1-3 alkyl.
实施方案18.如前述实施方案中任一项所述的化合物,其中每个R**是甲基。Embodiment 18. The compound of any of the preceding embodiments, wherein each R** is methyl.
实施方案19.如前述实施方案中任一项所述的化合物,其中M和M'中的一个是-NRMC(O)NRM-。Embodiment 19. The compound of any of the preceding embodiments, wherein one of M and M ' is -NRMC(O) NRM- .
实施方案20.如前述实施方案中任一项所述的化合物,其中每个RM是H。Embodiment 20. The compound of any of the preceding embodiments, wherein each R M is H.
实施方案21.如前述实施方案中任一项所述的化合物,其中R1是R”M’R’并且R’是支链烷基。Embodiment 21. The compound of any of the preceding embodiments, wherein R1 is R"M'R' and R' is branched alkyl.
实施方案22.如前述实施方案中任一项所述的化合物,其中R1是R”M’R’并且R’是烯基。Embodiment 22. The compound of any of the preceding embodiments, wherein R1 is R"M'R' and R' is alkenyl.
实施方案23.如前述实施方案中任一项所述的化合物,其中R1是R”M’R’并且R”是支链烷基。Embodiment 23. The compound of any of the preceding embodiments, wherein R1 is R"M'R' and R" is branched alkyl.
实施方案24.如前述实施方案中任一项所述的化合物,其中R1是R”M’R’并且R”是被OH取代的烷基。Embodiment 24. The compound of any of the preceding embodiments, wherein R1 is R"M'R' and R" is alkyl substituted with OH.
实施方案25.如前述实施方案中任一项所述的化合物,其中R’是R*YR*”并且Y是环丙基、环戊基或环己基。Embodiment 25. The compound of any of the preceding embodiments, wherein R' is R*YR*" and Y is cyclopropyl, cyclopentyl, or cyclohexyl.
实施方案26.如前述实施方案中任一项所述的化合物,其中R’是R*YR*”并且R*”是C2烷基、C3-烷基或C4烷基。Embodiment 26. The compound of any of the preceding embodiments, wherein R' is R*YR*" and R*" is C2 alkyl, C3 - alkyl, or C4 alkyl.
实施方案27.如前述实施方案中任一项所述的化合物,其中所述化合物具有以下结构:Embodiment 27. The compound of any one of the preceding embodiments, wherein the compound has the structure:
其中R”是任选地被OH取代的直链或支链C3-C13烷基,并且m选自5、6、7、8和9。 wherein R" is a straight or branched C3 - C13 alkyl optionally substituted with OH, and m is selected from 5, 6, 7, 8 and 9.
实施方案28.如前述实施方案中任一项所述的化合物,其中所述化合物具有以下结构:Embodiment 28. The compound of any one of the preceding embodiments, wherein the compound has the structure:
其中R”是任选地被OH取代的直链或支链C3-C13烷基,并且m选自5、6、7、8和9。 wherein R" is a straight or branched C3 - C13 alkyl optionally substituted with OH, and m is selected from 5, 6, 7, 8 and 9.
实施方案29.如前述权利要求中的一项所述的化合物,其中所述化合物具有以下结构:Embodiment 29. The compound of one of the preceding claims, wherein the compound has the structure:
其中R”是任选地被OH取代的直链或支链C3-C13烷基,并且m选自5、6、7、8和9。 wherein R" is a straight or branched C3 - C13 alkyl optionally substituted with OH, and m is selected from 5, 6, 7, 8 and 9.
实施方案30.如前述权利要求中的一项所述的化合物,其中所述化合物具有以下结构:Embodiment 30. The compound of one of the preceding claims, wherein the compound has the structure:
其中R”是任选地被OH取代的直链或支链C3-C13烷基,并且m选自5、6、7、8和9。 wherein R" is a straight or branched C3 - C13 alkyl optionally substituted with OH, and m is selected from 5, 6, 7, 8 and 9.
实施方案31.如前述权利要求中的一项所述的化合物,其中所述化合物具有以下结构:Embodiment 31. The compound of one of the preceding claims, wherein the compound has the structure:
其中R”是任选地被OH取代的直链或支链C3-C13烷基,并且m选自5、6、7、8和9。 wherein R" is a straight or branched C3 - C13 alkyl optionally substituted with OH, and m is selected from 5, 6, 7, 8 and 9.
实施方案32.如前述实施方案1或2中任一项所述的化合物,其中所述化合物具有以下结构:Embodiment 32. The compound of any one of the preceding Embodiments 1 or 2, wherein the compound has the structure:
其中R”是任选地被OH取代的直链或支链C3-C13烷基,并且m选自5、6、7、8和9。 wherein R" is a straight or branched C3 - C13 alkyl optionally substituted with OH, and m is selected from 5, 6, 7, 8 and 9.
实施方案33.如前述实施方案中任一项所述的化合物,其中所述化合物具有以下结构:Embodiment 33. The compound of any one of the preceding embodiments, wherein the compound has the structure:
其中R”是任选地被OH取代的直链或支链C3-C13烷基,并且m选自5、6、7、8和9。 wherein R" is a straight or branched C3 - C13 alkyl optionally substituted with OH, and m is selected from 5, 6, 7, 8 and 9.
实施方案34.一种式(I-Y1)的化合物:Embodiment 34. A compound of formula (I-Y1):
或其N-氧化物,或它们的盐或异构体,其中: or their N-oxides, or their salts or isomers, wherein:
R1选自由C5-30烷基、C5-20烯基、-R*YR*”、-YR*”和-R”M’R’组成的组;R 1 is selected from the group consisting of C 5-30 alkyl, C 5-20 alkenyl, -R*YR*", -YR*" and -R"M'R';
R2和R3独立地选自由H、C1-14烷基、C2-14烯基、-R*YR*”、-YR*”和-R*OR*”组成的组,或者R2和R3连同其所连接的原子一起形成杂环或碳环;R 2 and R 3 are independently selected from the group consisting of H, C 1-14 alkyl, C 2-14 alkenyl, -R*YR*", -YR*" and -R*OR*", or R 2 and R3 together with the atoms to which it is attached form a heterocyclic or carbocyclic ring;
R4选自由氢、C3-6碳环、-(CH2)nQ、-(CH2)nCHQR、-(CH2)oC(R12)2(CH2)n-oQ、-CHQR、-CQ(R)2、-C(O)NQR和未取代的C1-6烷基组成的组,其中Q选自碳环、杂环、-OR、-O(CH2)nN(R)2、-C(O)OR、-OC(O)R、-CX3、-CX2H、-CXH2、-CN、-N(R)2、-C(O)N(R)2、-N(R)C(O)R、-N(R)S(O)2R、-N(R)C(O)N(R)2、-N(R)C(S)N(R)2、-N(R)R8、-N(R)S(O)2R8、-O(CH2)nOR、-N(R)C(=NR9)N(R)2、-N(R)C(=CHR9)N(R)2、-OC(O)N(R)2、-N(R)C(O)OR、-N(OR)C(O)R、-N(OR)S(O)2R、-N(OR)C(O)OR、-N(OR)C(O)N(R)2、-N(OR)C(S)N(R)2、-N(OR)C(=NR9)N(R)2、-N(OR)C(=CHR9)N(R)2、-C(=NR9)N(R)2、-C(=NR9)R、-C(O)N(R)OR、-(CH2)nN(R)2、–C(R)N(R)2C(O)OR、NC(R)=R11、N(C=NR15)R11、NRC(C(O)NR14R14’)2、-NRC(O)(CH2)pC(O)NR14R14’和其中A是C6-10芳基或杂环;并且R 4 is selected from free hydrogen, C 3-6 carbocyclic ring, -(CH 2 ) n Q, -(CH 2 ) n CHQR, -(CH 2 ) o C(R 12 ) 2 (CH 2 ) no Q, -CHQR , -CQ(R) 2 , -C(O)NQR and unsubstituted C 1-6 alkyl groups, wherein Q is selected from carbocycle, heterocycle, -OR, -O(CH 2 ) n N( R) 2 , -C(O)OR, -OC(O)R, -CX 3 , -CX 2 H, -CXH 2 , -CN, -N(R) 2 , -C(O)N(R) 2 , -N(R)C(O)R, -N(R)S(O) 2 R, -N(R)C(O)N(R) 2 , -N(R)C(S)N (R) 2 , -N(R)R 8 , -N(R)S(O) 2 R 8 , -O(CH 2 ) n OR, -N(R)C(=NR 9 )N(R) 2 , -N(R)C(=CHR 9 )N(R) 2 , -OC(O)N(R) 2 , -N(R)C(O)OR, -N(OR)C(O) R, -N(OR)S(O) 2 R, -N(OR)C(O)OR, -N(OR)C(O)N(R) 2 , -N(OR)C(S)N (R) 2 , -N(OR)C(=NR 9 )N(R) 2 , -N(OR)C(=CHR 9 )N(R) 2 , -C(=NR 9 )N(R) 2 , -C(=NR 9 )R, -C(O)N(R)OR, -(CH 2 ) n N(R) 2 , -C(R)N(R) 2 C(O)OR, NC(R)=R 11 , N(C=NR 15 )R 11 , NRC(C(O)NR 14 R 14 ′) 2 , -NRC(O)(CH 2 ) p C(O)NR 14 R 14 'and wherein A is C 6-10 aryl or heterocycle; and
每个o独立地选自1、2、3和4;p是0、1、2、3或4;q是1、2、3或4;并且每个n独立地选自1、2、3、4和5;或each o is independently selected from 1, 2, 3, and 4; p is 0, 1, 2, 3, or 4; q is 1, 2, 3, or 4; and each n is independently selected from 1, 2, 3 , 4 and 5; or
R4是其中 R4 is in
Xa和Xb各自独立地是O或S;X a and X b are each independently O or S;
R10选自由H、卤代基、-OH、R、-N(R)2、-CN、-N3、-C(O)OH、-C(O)OR、-OC(O)R、-OR、-SR、-S(O)R、-S(O)OR、-S(O)2OR、-NO2、-S(O)2N(R)2、-N(R)S(O)2R、–NH(CH2)t1N(R)2、–NH(CH2)p1O(CH2)q1N(R)2、–NH(CH2)s1OR、–N((CH2)s1OR)2、-N(R)-碳环、-N(R)-杂环、-N(R)-芳基、-N(R)-杂芳基、-N(R)(CH2)t1-碳环、-N(R)(CH2)t1-杂环、-N(R)(CH2)t1-芳基、-N(R)(CH2)t1-杂芳基、碳环、杂环、芳基和杂芳基组成的组;R 10 is selected from H, halo, -OH, R, -N(R) 2 , -CN, -N 3 , -C(O)OH, -C(O)OR, -OC(O)R, -OR, -SR, -S(O)R, -S(O)OR, -S(O) 2 OR, -NO 2 , -S(O) 2 N(R) 2 , -N(R)S (O) 2 R, –NH(CH 2 ) t1 N(R) 2 , –NH(CH 2 ) p1 O(CH 2 ) q1 N(R) 2 , –NH(CH 2 ) s1 OR, –N( (CH 2 ) s1 OR) 2 , -N(R)-carbocycle, -N(R)-heterocycle, -N(R)-aryl, -N(R)-heteroaryl, -N(R )(CH 2 ) t1 -carbocycle , -N(R)(CH 2 ) t1 -heterocycle, -N(R)(CH 2 ) t1 -aryl, -N(R)(CH 2 ) t1 -heterocycle the group consisting of aryl, carbocycle, heterocycle, aryl and heteroaryl;
n2选自1、2、3、4、5、6、7、8、9和10;n2 is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9 and 10;
r是0或1;r is 0 or 1;
t1选自1、2、3、4和5;t 1 is selected from 1, 2, 3, 4 and 5;
p1选自1、2、3、4和5;p 1 is selected from 1, 2, 3, 4 and 5;
q1选自1、2、3、4和5;q 1 is selected from 1, 2, 3, 4 and 5;
s1选自1、2、3、4和5;s 1 is selected from 1, 2, 3, 4 and 5;
每个R5独立地选自由OH、C1-3烷基、C2-3烯基和H组成的组;each R 5 is independently selected from the group consisting of OH, C 1-3 alkyl, C 2-3 alkenyl and H;
每个R6独立地选自由OH、C1-3烷基、C2-3烯基和H组成的组;each R is independently selected from the group consisting of OH, C 1-3 alkyl, C 2-3 alkenyl and H ;
M’选自–OC(O)O-、-C(O)O-、-OC(O)-、-OC(O)-M”-C(O)O-、-OC(O)-NRM-C(O)O-、-O-M”-O-、-C(O)N(RM)-、-N(RM)C(O)-、-OC(O)N(RM)-、-N(RM)C(O)O-、-NRMC(O)NRM-、-O-N=C(RM)-、-C(O)-、-C(S)-、-C(S)S-、-SC(S)-、-CH(OH)-、-P(O)(ORM)O-、-S(O)2-、-S-S-、-SO-、-OS-、S(RM)2O-、-O-S(RM)2-、-S(O)O-、-OS(O)-、芳基和杂芳基,其中M”是键、-(CH2)zC(O)-、C1-13烷基、C2-13烯基、-B(R**)-、-Si(R**)2-、-S(R**)2-或-S(O)-,其中z是1、2、3或4;M' is selected from -OC(O)O-, -C(O)O-, -OC(O)-, -OC(O)-M"-C(O)O-, -OC(O)-NR M -C(O)O-, -OM"-O-, -C(O)N(R M )-, -N(R M )C(O)-, -OC(O)N(R M ) -, -N(R M )C(O)O-, -NR M C(O)NR M -, -ON=C(R M )-, -C(O)-, -C(S)-, -C(S)S-, -SC(S)-, -CH(OH)-, -P(O)(OR M )O-, -S(O) 2 -, -SS-, -SO-, -OS-, S(R M ) 2 O-, -OS(R M ) 2 -, -S(O)O-, -OS(O)-, aryl and heteroaryl, where M" is a bond, -(CH 2 ) z C(O)-, C 1-13 alkyl, C 2-13 alkenyl, -B(R**)-, -Si(R**) 2 -, -S(R* *) 2- or -S(O)-, where z is 1, 2, 3 or 4;
R7选自由C1-3烷基、C2-3烯基和H组成的组;R 7 is selected from the group consisting of C 1-3 alkyl, C 2-3 alkenyl and H;
R8选自由C3-6碳环和杂环组成的组;R 8 is selected from the group consisting of C 3-6 carbocycles and heterocycles;
R9选自由H、CN、NO2、C1-6芳基、-OR、-S(O)2R、-S(O)2N(R)2、C2-6烯基、C3-6碳环和杂环组成的组;R 9 is selected from H, CN, NO 2 , C 1-6 aryl, -OR, -S(O) 2 R, -S(O) 2 N(R) 2 , C 2-6 alkenyl, C 3 - the group consisting of 6 carbocycles and heterocycles;
R11选自由C3-6碳环和杂环组成的组,其中C3-6碳环和杂环各自任选地被一个或多个R13取代;R 11 is selected from the group consisting of C 3-6 carbocycles and heterocycles, wherein each of the C 3-6 carbocycles and heterocycles is optionally substituted with one or more R 13 ;
R12选自由H、OH、C1-3烷基和C2-3烯基组成的组;R 12 is selected from the group consisting of H, OH, C 1-3 alkyl and C 2-3 alkenyl;
每个R13独立地选自由OH、氧代基、卤代基、C1-6烷基、C1-6烷氧基、C2-6烯基、C1-6烷基氨基、二-(C1-6烷基)氨基、氨基、酰胺基、氰基和硝基组成的组;Each R 13 is independently selected from OH, oxo, halo, C 1-6 alkyl, C 1-6 alkoxy, C 2-6 alkenyl, C 1-6 alkylamino, di- (C 1-6 alkyl) the group consisting of amino, amino, amido, cyano and nitro;
每个R14独立地选自由H、OH、C1-6烷基和C2-3烯基组成的组;each R 14 is independently selected from the group consisting of H, OH, C 1-6 alkyl and C 2-3 alkenyl;
每个R14’独立地选自由H、OH、C1-6烷基和C2-3烯基组成的组;each R 14 ' is independently selected from the group consisting of H, OH, C 1-6 alkyl and C 2-3 alkenyl;
R15独立地选自由H、OH、C1-6烷基和C2-3烯基组成的组;R 15 is independently selected from the group consisting of H, OH, C 1-6 alkyl and C 2-3 alkenyl;
每个R独立地选自由C1-6烷基、C1-3烷基-芳基、C2-3烯基和H组成的组;each R is independently selected from the group consisting of C1-6 alkyl, C1-3 alkyl-aryl, C2-3 alkenyl, and H;
RN是H或C1-3烷基;R N is H or C 1-3 alkyl;
每个R’独立地选自由C1-18烷基、C2-18烯基、-R*YR*”、-YR*”、(CH2)qOR*和H组成的组,each R' is independently selected from the group consisting of C 1-18 alkyl, C 2-18 alkenyl, -R*YR*", -YR*", (CH 2 ) q OR* and H,
并且每个q独立地选自1、2和3;and each q is independently selected from 1, 2 and 3;
每个RM独立地选自由H、C1-6烷基和C2-6烯基组成的组;each R M is independently selected from the group consisting of H, C 1-6 alkyl and C 2-6 alkenyl;
每个R”独立地选自由C3-15烷基和C3-15烯基组成的组;each R" is independently selected from the group consisting of C 3-15 alkyl and C 3-15 alkenyl;
每个R*”独立地选自由C1-15烷基和C2-15烯基组成的组;each R*" is independently selected from the group consisting of C 1-15 alkyl and C 2-15 alkenyl;
每个R*独立地选自由C1-12烷基和C2-12烯基组成的组;each R* is independently selected from the group consisting of C 1-12 alkyl and C 2-12 alkenyl;
每个R**独立地选自由H、OH、C1-12烷基、C2-12烯基、(CH2)qOR*和(CH2)qOH组成的组;each R** is independently selected from the group consisting of H, OH, C 1-12 alkyl, C 2-12 alkenyl, (CH 2 ) q OR*, and (CH 2 ) q OH;
每个Y独立地是C3-6碳环;each Y is independently a C3-6 carbocycle;
每个X独立地选自由F、Cl、Br和I组成的组;并且each X is independently selected from the group consisting of F, Cl, Br, and I; and
m选自5、6、7、8、9、10、11、12和13。m is selected from 5, 6, 7, 8, 9, 10, 11, 12 and 13.
实施方案35.如前述实施方案中任一项所述的化合物,其中所述化合物具有以下结构:其中每个R”独立地选自H、OH和C1-6烷基,l选自2、3、4、5和6,并且m选自5、6、7、8和9。Embodiment 35. The compound of any one of the preceding embodiments, wherein the compound has the structure: wherein each R" is independently selected from H, OH, and C1-6 alkyl, l is selected from 2, 3, 4, 5, and 6, and m is selected from 5, 6, 7, 8, and 9.
实施方案36.如前述实施方案中任一项所述的化合物,其中R4选自-(CH2)nOH,其中n选自1、2、3、4和5,并且其中n2选自1、2、3、4、5、6、7、8、9和10;并且R10是-N(R)2,其中每个R独立地选自由C1-6烷基、C2-3烯基和H组成的组;Embodiment 36. The compound of any one of the preceding embodiments, wherein R4 is selected from -( CH2 )nOH, wherein n is selected from 1, 2, 3, 4 , and 5, and wherein n2 is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, and 10 ; and R10 is -N(R) 2 , wherein each R is independently selected from C1-6 alkyl, The group consisting of C 2-3 alkenyl and H;
实施方案37.一种式(A)的化合物:Embodiment 37. A compound of formula (A):
或其N-氧化物,或它们的盐或异构体,其中 or their N-oxides, or their salts or isomers, wherein
m选自5、6、7、8和9;m is selected from 5, 6, 7, 8 and 9;
R2和R3各自独立地选自由H、C1-14烷基和C2-14烯基组成的组;R 2 and R 3 are each independently selected from the group consisting of H, C 1-14 alkyl and C 2-14 alkenyl;
R4选自-(CH2)nOH,其中n选自1、2、3、4和5;和其中n2选自1、2、3、4、5、6、7、8、9和10;并且R10是-N(R)2,其中每个R独立地选自由C1-6烷基、C2-3烯基和H组成的组;R4 is selected from -( CH2 )nOH, wherein n is selected from 1, 2, 3, 4 and 5; and wherein n2 is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, and 10 ; and R10 is -N(R) 2 , wherein each R is independently selected from C1-6 alkyl, The group consisting of C 2-3 alkenyl and H;
M和M'各自独立地选自-OC(O)O-和-C(O)O-;M and M' are each independently selected from -OC(O)O- and -C(O)O-;
其中M和M'中的至少一个是-OC(O)O-;wherein at least one of M and M' is -OC(O)O-;
R’a是C1-18烷基或C2-18烯基;并且R' a is C 1-18 alkyl or C 2-18 alkenyl; and
R”是任选地被OH取代的C3-C13烷基。R" is C3 - C13 alkyl optionally substituted with OH.
实施方案38.如前述实施方案中任一项所述的化合物,其中R”选自其中表示与N的连接点,并且表示与M的连接点。Embodiment 38. The compound of any one of the preceding embodiments, wherein R" is selected from in represents the junction with N, and Indicates the connection point with M.
实施方案39.如前述实施方案中任一项所述的化合物,其中所述化合物具有以下结构中的一个: Embodiment 39. The compound of any one of the preceding embodiments, wherein the compound has one of the following structures:
实施方案40.如前述实施方案中任一项所述的化合物,其中R’a是:其中表示连接点;Embodiment 40. The compound of any one of the preceding embodiments, wherein R'a is: in represents a connection point;
Raα、Raβ和Raγ各自独立地选自由H、C1-12烷基和C2-12烯基组成的组,其中Raα、Raβ和Raγ中的至少一个选自由C1-12烷基和C2-12烯基组成的组;并且R aα , R aβ and R aγ are each independently selected from the group consisting of H, C 1-12 alkyl and C 2-12 alkenyl, wherein at least one of R aα , R aβ and R aγ is selected from C 1- the group consisting of 12 alkyl and C 2-12 alkenyl; and
R’是C1-12烷基或C2-12烯基。R' is C 1-12 alkyl or C 2-12 alkenyl.
实施方案41.如前述实施方案中任一项所述的化合物,其中Raβ和Raγ各自是H并且Raα是C1-6烷基。Embodiment 41. The compound of any one of the preceding embodiments, wherein R aβ and R aγ are each H and R aα is C 1-6 alkyl.
实施方案42.如前述实施方案中任一项所述的化合物,其中Raα和Raγ各自是H并且Raβ是C1-6烷基。Embodiment 42. The compound of any one of the preceding embodiments, wherein R aα and R aγ are each H and R aβ is C 1-6 alkyl.
实施方案43.如前述实施方案中任一项所述的化合物,其中Raβ是C2烷基。Embodiment 43. The compound of any one of the preceding embodiments, wherein R aβ is C 2 alkyl.
实施方案44.如前述实施方案中任一项所述的化合物,其中Raα和Raβ各自是H并且Ra γ是C1-6烷基。Embodiment 44. The compound of any one of the preceding embodiments, wherein R aα and R aβ are each H and R aγ is C 1-6 alkyl .
实施方案45.如前述实施方案中任一项所述的化合物,其中Raγ是C5或C6烷基。Embodiment 45. The compound of any one of the preceding embodiments, wherein R aγ is C 5 or C 6 alkyl.
实施方案46.如前述实施方案中任一项所述的化合物,其中R’是C5或C6烷基。Embodiment 46. The compound of any of the preceding embodiments, wherein R' is C5 or C6 alkyl.
实施方案47.如前述实施方案中任一项所述的化合物,其中所述化合物具有以下结构: Embodiment 47. The compound of any one of the preceding embodiments, wherein the compound has the structure:
实施方案48.如前述实施方案中任一项所述的化合物,其中R4是-(CH2)nOH。Embodiment 48. The compound of any of the preceding embodiments, wherein R4 is - ( CH2 ) nOH .
实施方案49.如前述实施方案中任一项所述的化合物,其中R4是-(CH2)2OH。Embodiment 49. The compound of any of the preceding embodiments, wherein R4 is - ( CH2 ) 2OH.
实施方案50.如前述实施方案中任一项所述的化合物,其中R4是 Embodiment 50. The compound of any one of the preceding embodiments, wherein R 4 is
实施方案51.如前述实施方案中任一项所述的化合物,其中R4是 Embodiment 51. The compound of any one of the preceding embodiments, wherein R 4 is
实施方案52.如前述实施方案中任一项所述的化合物,其中n2是2或4。Embodiment 52. The compound of any of the preceding embodiments, wherein n2 is 2 or 4.
实施方案53.一种化合物,其选自:Embodiment 53. A compound selected from:
实施方案54.一种空脂质纳米粒子(空LNP),其包含如前述实施方案中任一项所述的化合物、磷脂、结构性脂质和PEG脂质。Embodiment 54. An empty lipid nanoparticle (empty LNP) comprising the compound of any of the preceding embodiments, a phospholipid, a structured lipid, and a PEG lipid.
实施方案55.一种负载脂质纳米粒子(负载LNP),其包含如前述实施方案中任一项所述的化合物、磷脂、结构性脂质、PEG脂质以及一种或多种治疗剂和/或预防剂。Embodiment 55. A lipid-loaded nanoparticle (loaded LNP) comprising a compound of any of the preceding embodiments, a phospholipid, a structured lipid, a PEG lipid, and one or more therapeutic agents and / or prophylactics.
实施方案56.如前述实施方案中任一项所述的空LNP或负载LNP,其包含约40%至约60%的量的所述化合物。Embodiment 56. The empty or loaded LNP of any one of the preceding embodiments, comprising the compound in an amount from about 40% to about 60%.
实施方案57.如前述实施方案中任一项所述的空LNP或负载LNP,其包含约0%至约20%的量的磷脂。Embodiment 57. The empty or loaded LNP of any one of the preceding embodiments, comprising phospholipid in an amount from about 0% to about 20%.
实施方案58.如前述实施方案中任一项所述的空LNP或负载LNP,其包含约30%至约50%的量的结构性脂质。Embodiment 58. The empty or loaded LNP of any one of the preceding embodiments, comprising structured lipids in an amount from about 30% to about 50%.
实施方案59.如前述实施方案中任一项所述的空LNP或负载LNP,其包含约0%至约5%的量的PEG脂质。Embodiment 59. The empty LNP or loaded LNP of any of the preceding embodiments, comprising PEG lipids in an amount from about 0% to about 5%.
实施方案60.如前述实施方案中任一项所述的空LNP或负载LNP,其包含约40mol%至约60mol%的如前述实施方案中任一项所述的化合物,约0mol%至约20mol%磷脂,约30mol%至约50mol%结构性脂质,以及约0mol%至约5mol%PEG脂质。Embodiment 60. The empty or supported LNP of any of the preceding embodiments, comprising from about 40 mol % to about 60 mol % of the compound of any preceding embodiment, from about 0 mol % to about 20 mol % % phospholipids, about 30 mol % to about 50 mol % structured lipids, and about 0 mol % to about 5 mol % PEG lipids.
实施方案61.如前述实施方案中任一项所述的空LNP或负载LNP,其包含约30mol%至约60mol%的如前述实施方案中任一项所述的化合物,约0mol%至约30mol%磷脂,约18.5mol%至约48.5mol%结构性脂质,以及约0mol%至约10mol%PEG脂质。Embodiment 61. The empty LNP or supported LNP of any of the preceding embodiments, comprising from about 30 mol % to about 60 mol % of the compound of any preceding embodiment, from about 0 mol % to about 30 mol % % phospholipids, about 18.5 mol % to about 48.5 mol % structured lipids, and about 0 mol % to about 10 mol % PEG lipids.
实施方案62.如前述实施方案中任一项所述的负载LNP,其中所述一种或多种治疗剂和/或预防剂是多核苷酸或多肽。Embodiment 62. The loaded LNP of any of the preceding embodiments, wherein the one or more therapeutic and/or prophylactic agents are polynucleotides or polypeptides.
实施方案63.如前述实施方案中任一项所述的负载LNP,其中所述一种或多种治疗剂和/或预防剂是核酸。Embodiment 63. The loaded LNP of any of the preceding embodiments, wherein the one or more therapeutic and/or prophylactic agents is a nucleic acid.
实施方案64.如前述实施方案中任一项所述的负载LNP,其中一种或多种治疗剂和/或预防剂选自由核糖核酸(RNA)和脱氧核糖核酸(DNA)组成的组。Embodiment 64. The loaded LNP of any of the preceding embodiments, wherein the one or more therapeutic and/or prophylactic agents are selected from the group consisting of ribonucleic acid (RNA) and deoxyribonucleic acid (DNA).
实施方案65.如前述实施方案中任一项所述的负载LNP,其中所述DNA选自由双链DNA、单链DNA(ssDNA)、部分双链DNA、三链DNA和部分三链DNA组成的组。Embodiment 65. The loaded LNP of any one of the preceding embodiments, wherein the DNA is selected from the group consisting of double-stranded DNA, single-stranded DNA (ssDNA), partially double-stranded DNA, triple-stranded DNA, and partially triple-stranded DNA Group.
实施方案66.如前述实施方案中任一项所述的负载LNP,其中所述DNA选自由环状DNA、线性DNA及其混合物组成的组。Embodiment 66. The loaded LNP of any of the preceding embodiments, wherein the DNA is selected from the group consisting of circular DNA, linear DNA, and mixtures thereof.
实施方案67.如前述实施方案中任一项所述的负载LNP,其中所述一种或多种治疗剂和/或预防剂选自由质粒表达载体、病毒表达载体及其混合物组成的组。Embodiment 67. The loaded LNP of any one of the preceding embodiments, wherein the one or more therapeutic and/or prophylactic agents are selected from the group consisting of plasmid expression vectors, viral expression vectors, and mixtures thereof.
实施方案68.如前述实施方案中任一项所述的负载LNP,其中所述一种或多种治疗剂和/或预防剂是RNA。Embodiment 68. The loaded LNP of any of the preceding embodiments, wherein the one or more therapeutic and/or prophylactic agents is RNA.
实施方案69.如前述实施方案中任一项所述的负载LNP,其中所述RNA选自由单链RNA、双链RNA(dsRNA)、部分双链RNA及其混合物组成的组。Embodiment 69. The loaded LNP of any of the preceding embodiments, wherein the RNA is selected from the group consisting of single-stranded RNA, double-stranded RNA (dsRNA), partially double-stranded RNA, and mixtures thereof.
实施方案70.如前述实施方案中任一项所述的负载LNP,其中所述RNA选自由环状RNA、线性RNA及其混合物组成的组。Embodiment 70. The loaded LNP of any of the preceding embodiments, wherein the RNA is selected from the group consisting of circular RNA, linear RNA, and mixtures thereof.
实施方案71.如前述实施方案中任一项所述的负载LNP,其中所述RNA选自由以下组成的组:短干扰RNA(siRNA)、不对称干扰RNA(aiRNA)、RNA干扰(RNAi)分子、微RNA(miRNA)、antagomir、反义RNA、核酶、Dicer-底物RNA(dsRNA)、小发夹RNA(shRNA)、信使RNA(mRNA)及其混合物。Embodiment 71. The loaded LNP of any of the preceding embodiments, wherein the RNA is selected from the group consisting of short interfering RNA (siRNA), asymmetric interfering RNA (aiRNA), RNA interference (RNAi) molecule , microRNA (miRNA), antagomir, antisense RNA, ribozyme, Dicer-substrate RNA (dsRNA), small hairpin RNA (shRNA), messenger RNA (mRNA) and mixtures thereof.
实施方案72.如前述实施方案中任一项所述的负载LNP,其中所述RNA是mRNA。Embodiment 72. The loaded LNP of any of the preceding embodiments, wherein the RNA is mRNA.
实施方案73.如前述实施方案中任一项所述的负载LNP,其中所述mRNA是修饰的mRNA(mmRNA)。Embodiment 73. The loaded LNP of any of the preceding embodiments, wherein the mRNA is a modified mRNA (mmRNA).
实施方案74.如前述实施方案中任一项所述的负载LNP,其中所述mRNA引入了微RNA结合位点(miR结合位点)。Embodiment 74. The loaded LNP of any of the preceding embodiments, wherein the mRNA incorporates a microRNA binding site (miR binding site).
实施方案75.如前述实施方案中任一项所述的负载LNP,其中所述mRNA包含茎环、链终止核苷、polyA序列、聚腺苷酸化信号和/或5'帽结构中的一个或多个。Embodiment 75. The load LNP of any one of the preceding embodiments, wherein the mRNA comprises one of a stem loop, a chain termination nucleoside, a polyA sequence, a polyadenylation signal and/or a 5' cap structure or multiple.
实施方案76.如前述实施方案中任一项所述的空LNP或负载LNP,其中所述磷脂选自由以下组成的组:Embodiment 76. The empty LNP or loaded LNP of any one of the preceding embodiments, wherein the phospholipid is selected from the group consisting of:
1,2-二亚油酰基-sn-甘油-3-磷酸胆碱(DLPC)、1,2-Dilinoleoyl-sn-glycero-3-phosphocholine (DLPC),
1,2-二肉豆蔻酰基-sn-甘油-磷酸胆碱(DMPC)、1,2-Dimyristoyl-sn-glycero-phosphocholine (DMPC),
1,2-二油酰基-sn-甘油-3-磷酸胆碱(DOPC)、1,2-Dioleoyl-sn-glycero-3-phosphocholine (DOPC),
1,2-二棕榈酰基-sn-甘油-3-磷酸胆碱(DPPC)、1,2-Dipalmitoyl-sn-glycero-3-phosphocholine (DPPC),
1,2-二硬脂酰基-sn-甘油-3-磷酸胆碱(DSPC)、1,2-Distearoyl-sn-glycero-3-phosphocholine (DSPC),
1,2-二-十一烷酰基-sn-甘油-磷酸胆碱(DUPC)、1,2-Di-undecanoyl-sn-glycero-phosphocholine (DUPC),
1-棕榈酰基-2-油酰基-sn-甘油-3-磷酸胆碱(POPC)、1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine (POPC),
1,2-二-O-十八碳烯基-sn-甘油-3-磷酸胆碱(18:0Diether PC)、1,2-Di-O-octadecenyl-sn-glycero-3-phosphocholine (18:0Diether PC),
1-油酰基-2-胆固醇基半琥珀酰基-sn-甘油-3-磷酸胆碱(OChemsPC)、1-Oleoyl-2-cholesterylhemisuccinyl-sn-glycero-3-phosphocholine (OChemsPC),
1-十六烷基-sn-甘油-3-磷酸胆碱(C16 Lyso PC)、1-Hexadecyl-sn-glycero-3-phosphocholine (C16 Lyso PC),
1,2-二亚麻酰基-sn-甘油-3-磷酸胆碱、1,2-Dilinolenoyl-sn-glycero-3-phosphocholine,
1,2-二花生四烯酰基-sn-甘油-3-磷酸胆碱、1,2-Diarachidonoyl-sn-glycero-3-phosphocholine,
1,2-二-二十二碳六烯酰基-sn-甘油-3-磷酸胆碱、1,2-二油酰基-sn-甘油-3-磷酸乙醇胺(DOPE)、1,2-二植烷酰基-sn-甘油-3-磷酸乙醇胺(ME 16.0PE)、1,2-Di-docosahexaenoyl-sn-glycero-3-phosphocholine, 1,2-dioleoyl-sn-glycero-3-phosphoethanolamine (DOPE), 1,2-diphytate Alkanoyl-sn-glycero-3-phosphoethanolamine (ME 16.0PE),
1,2-二硬脂酰基-sn-甘油-3-磷酸乙醇胺、1,2-Distearoyl-sn-glycero-3-phosphoethanolamine,
1,2-二亚油酰基-sn-甘油-3-磷酸乙醇胺、1,2-Dilinoleoyl-sn-glycero-3-phosphoethanolamine,
1,2-二亚麻酰基-sn-甘油-3-磷酸乙醇胺、1,2-Dilinolenoyl-sn-glycero-3-phosphoethanolamine,
1,2-二花生四烯酰基-sn-甘油-3-磷酸乙醇胺、1,2-Diarachidonoyl-sn-glycero-3-phosphoethanolamine,
1,2-二-二十二碳六烯酰基-sn-甘油-3-磷酸乙醇胺、1,2-Di-docosahexaenoyl-sn-glycero-3-phosphoethanolamine,
1,2-二油酰基-sn-甘油-3-磷酸-rac-(1-甘油)钠盐(DOPG)、鞘磷脂及其混合物。1,2-Dioleoyl-sn-glycero-3-phosphate-rac-(1-glycerol) sodium salt (DOPG), sphingomyelin and mixtures thereof.
实施方案77.如前述实施方案中任一项所述的空LNP或负载LNP,其中所述磷脂是1,2-二硬脂酰基-sn-甘油-3-磷酸胆碱(DSPC)。Embodiment 77. The empty or loaded LNP of any of the preceding embodiments, wherein the phospholipid is 1,2-distearoyl-sn-glycero-3-phosphocholine (DSPC).
实施方案78.如前述实施方案中任一项所述的空LNP或负载LNP,其中所述结构性脂质选自由以下组成的组:胆固醇、粪固醇、谷固醇、麦角固醇、菜油固醇、豆固醇、菜籽固醇以及其混合物。Embodiment 78. The empty or loaded LNP of any one of the preceding embodiments, wherein the structural lipid is selected from the group consisting of cholesterol, coprosterol, sitosterol, ergosterol, rapeseed oil sterols, stigmasterol, brassicasterol and mixtures thereof.
实施方案79.如前述实施方案中任一项所述的空LNP或负载LNP,其中所述结构性脂质是或其盐。Embodiment 79. The empty LNP or loaded LNP of any one of the preceding embodiments, wherein the structural lipid is or its salt.
实施方案80.如前述实施方案中任一项所述的空LNP或负载LNP,其中所述结构性脂质是胆固醇:或其盐。Embodiment 80. The empty or loaded LNP of any one of the preceding embodiments, wherein the structural lipid is cholesterol: or its salt.
实施方案81.如前述实施方案中任一项所述的空LNP或负载LNP,其中所述PEG脂质是选自由以下组成的组:PEG修饰的磷脂酰乙醇胺、PEG修饰的磷脂酸、PEG修饰的神经酰胺、PEG修饰的二烷基胺、PEG修饰的二酰基甘油、PEG修饰的二烷基甘油及其混合物。Embodiment 81. The empty or loaded LNP of any of the preceding embodiments, wherein the PEG lipid is selected from the group consisting of: PEG-modified phosphatidylethanolamine, PEG-modified phosphatidic acid, PEG-modified ceramides, PEG-modified dialkylamines, PEG-modified diacylglycerols, PEG-modified dialkylglycerols, and mixtures thereof.
实施方案82.如前述实施方案中任一项所述的空LNP或负载LNP,其中所述PEG脂质选自由以下组成的组:1,2-二肉豆蔻酰基-sn-甘油甲氧基聚乙二醇(PEG-DMG)、1,2-二硬脂酰基-sn-甘油-3-磷酸乙醇胺-N-[氨基(聚乙二醇)](PEG-DSPE)、PEG-二硬脂基甘油(PEG-DSG)、PEG-二棕榈酰基、PEG-二油酰基、PEG-二硬脂基、PEG-二酰基甘氨酰胺(PEG-DAG)、PEG-二棕榈酰基磷脂酰乙醇胺(PEG-DPPE)或PEG-1,2-二肉豆蔻基氧基丙基-3-胺(PEG-c-DMA)。Embodiment 82. The empty LNP or loaded LNP of any one of the preceding embodiments, wherein the PEG lipid is selected from the group consisting of 1,2- dimyristoyl-sn-glyceromethoxypoly Ethylene glycol (PEG-DMG), 1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[amino(polyethylene glycol)](PEG-DSPE), PEG-distearyl Glycerol (PEG-DSG), PEG-dipalmitoyl, PEG-dioleoyl, PEG-distearyl, PEG-diacylglycinamide (PEG-DAG), PEG-dipalmitoylphosphatidylethanolamine (PEG- DPPE) or PEG-1,2-dimyristyloxypropyl-3-amine (PEG-c-DMA).
实施方案83.如前述实施方案中任一项所述的空LNP或负载LNP,其中所述PEG脂质是PEG-DMG。Embodiment 83. The empty or loaded LNP of any of the preceding embodiments, wherein the PEG lipid is PEG-DMG.
实施方案84.如前述实施方案中任一项所述的空LNP或负载LNP,其中所述PEG脂质是式(PL-I)的化合物:Embodiment 84. The empty or loaded LNP of any one of the preceding embodiments, wherein the PEG lipid is a compound of formula (PL-I):
或其盐,其中:or a salt thereof, where:
R3PL1是–OROPL1;R 3PL1 is –OR OPL1 ;
ROPL1是氢、任选取代的烷基或氧保护基团;R OPL1 is hydrogen, an optionally substituted alkyl or oxygen protecting group;
rPL1是介于1与100之间的整数,包括端值;r PL1 is an integer between 1 and 100, inclusive;
L1是任选取代的C1-10亚烷基,其中任选取代的C1-10亚烷基中的至少一个亚甲基独立地被替代为:任选取代的亚碳环基、任选取代的亚杂环基、任选取代的亚芳基、任选取代的亚杂芳基、O、N(RNPL1)、S、C(O)、C(O)N(RNPL1)、NRNPL1C(O)、C(O)O、OC(O)、OC(O)O、OC(O)N(RNPL1)、NRNPL1C(O)O或NRNPL1C(O)N(RNPL1);L 1 is an optionally substituted C 1-10 alkylene group, wherein at least one methylene group in the optionally substituted C 1-10 alkylene group is independently replaced by: optionally substituted carbocyclylene, any optionally substituted heterocyclylene, optionally substituted arylene, optionally substituted heteroarylene, O, N(R NPL1 ), S, C(O), C(O)N(R NPL1 ), NR NPL1 C(O), C(O)O, OC(O), OC(O)O, OC(O)N(R NPL1 ), NR NPL1 C(O)O or NR NPL1 C(O)N( R NPL1 );
D是通过点击化学获得的部分或在生理条件下可切割的部分;D is a moiety obtained by click chemistry or a moiety cleavable under physiological conditions;
mPL1是0、1、2、3、4、5、6、7、8、9或10;m PL1 is 0, 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10;
A具有下式: A has the following formula:
L2的每种情况独立地是键或任选取代的C1-6亚烷基,其中任选取代的C1-6亚烷基中的一个亚甲基单元任选地被替代为:O、N(RNPL1)、S、C(O)、C(O)N(RNPL1)、NRNPL1C(O)、C(O)O、OC(O)、OC(O)O、OC(O)N(RNPL1)、NRNPL1C(O)O或NRNPL1C(O)N(RNPL1);Each instance of L is independently a bond or an optionally substituted C1-6 alkylene where one methylene unit in the optionally substituted C1-6 alkylene is optionally replaced with: O , N(R NPL1 ), S, C(O), C(O)N(R NPL1 ), NR NPL1 C(O), C(O)O, OC(O), OC(O)O, OC( O)N(R NPL1 ), NR NPL1 C(O)O or NR NPL1 C(O)N(R NPL1 );
R2SL的每种情况独立地是任选取代的C1-30烷基、任选取代的C1-30烯基或任选取代的C1-30炔基;任选地其中R2SL的一个或多个亚甲基单元独立地被替代为:任选取代的亚碳环基、任选取代的亚杂环基、任选取代的亚芳基、任选取代的亚杂芳基、N(RNPL1)、O、S、C(O)、C(O)N(RNPL1)、NRNPL1C(O)、NRNPL1C(O)N(RNPL1)、C(O)O、OC(O)、OC(O)O、OC(O)N(RNPL1)、NRNPL1C(O)O、C(O)S、SC(O)、C(=NRNPL1)、C(=NRNPL1)N(RNPL1)、NRNPL1C(=NRNPL1)、NRNPL1C(=NRNPL1)N(RNPL1)、C(S)、C(S)N(RNPL1)、NRNPL1C(S)、NRNPL1C(S)N(RNPL1)、S(O)、OS(O)、S(O)O、OS(O)O、OS(O)2、S(O)2O、OS(O)2O、N(RNPL1)S(O)、S(O)N(RNPL1)、N(RNPL1)S(O)N(RNPL1)、OS(O)N(RNPL1)、N(RNPL1)S(O)O、S(O)2、N(RNPL1)S(O)2、S(O)2N(RNPL1)、N(RNPL1)S(O)2N(RNPL1)、OS(O)2N(RNPL1)或N(RNPL1)S(O)2O;Each instance of R 2SL is independently optionally substituted C 1-30 alkyl, optionally substituted C 1-30 alkenyl, or optionally substituted C 1-30 alkynyl; optionally one of wherein R 2SL or more methylene units are independently replaced with: optionally substituted carbocyclylene, optionally substituted heterocyclylene, optionally substituted arylene, optionally substituted heteroarylene, N( R NPL1 ), O, S, C(O), C(O)N(R NPL1 ), NR NPL1 C(O), NR NPL1 C(O)N(R NPL1 ), C(O)O, OC( O), OC(O)O, OC(O)N(R NPL1 ), NR NPL1 C(O)O, C(O)S, SC(O), C(=NR NPL1 ), C(=NR NPL1 )N(R NPL1 ), NR NPL1 C(=NR NPL1 ), NR NPL1 C(=NR NPL1 )N(R NPL1 ), C(S), C(S)N(R NPL1 ), NR NPL1 C(S ), NR NPL1 C(S)N(R NPL1 ), S(O), OS(O), S(O)O, OS(O)O, OS(O) 2 , S(O) 2 O, OS (O) 2 O, N(R NPL1 )S(O), S(O)N(R NPL1 ), N(R NPL1 )S(O)N(R NPL1 ), OS(O)N(R NPL1 ) , N(R NPL1 )S(O)O, S(O) 2 , N(R NPL1 )S(O) 2 , S(O) 2 N(R NPL1 ), N(R NPL1 )S(O) 2 N(R NPL1 ), OS(O) 2 N(R NPL1 ) or N(R NPL1 )S(O) 2 O;
RNPL1的每种情况独立地是氢、任选取代的烷基或氮保护基团;Each instance of R NPL1 is independently a hydrogen, optionally substituted alkyl or nitrogen protecting group;
环B是任选取代的碳环基、任选取代的杂环基、任选取代的芳基或任选取代的杂芳基;并且Ring B is optionally substituted carbocyclyl, optionally substituted heterocyclyl, optionally substituted aryl, or optionally substituted heteroaryl; and
pSL是1或2。 pSL is 1 or 2.
实施方案85.如前述实施方案中任一项所述的空LNP或负载LNP,其中所述PEG脂质是式(PL-I-OH)的化合物:或其盐。Embodiment 85. The empty or loaded LNP of any one of the preceding embodiments, wherein the PEG lipid is a compound of formula (PL-I-OH): or its salt.
实施方案86.如前述实施方案中任一项所述的空LNP或负载LNP,其中所述PEG脂质是式(PL-II-OH)的化合物:或其盐或异构体,其中:Embodiment 86. The empty LNP or loaded LNP of any one of the preceding embodiments, wherein the PEG lipid is a compound of formula (PL-II-OH): or a salt or isomer thereof, wherein:
R3PEG是–ORO;R 3PEG is -OR O ;
RO是氢、C1-6烷基或氧保护基团;R O is a hydrogen, C 1-6 alkyl or oxygen protecting group;
rPEG是介于1与100之间的整数;r PEG is an integer between 1 and 100;
R5PEG是C10-40烷基、C10-40烯基或任选取代的C10-40炔基;并且任选地R5PEG的一个或多个亚甲基独立地被替代为:C3-10亚碳环基、4至10元亚杂环基、C6-10亚芳基、4至10元亚杂芳基、–N(RNPEG)–、–O–、–S–、–C(O)–、–C(O)N(RNPEG)–、–NRNPEGC(O)–、–NRNPEGC(O)N(RNPEG)–、–C(O)O–、–OC(O)–、–OC(O)O–、–OC(O)N(RNPEG)–、–NRNPEGC(O)O–、–C(O)S–、–SC(O)–、–C(=NRNPEG)–、–C(=NRNPEG)N(RNPEG)–、–NRNPEGC(=NRNPEG)–、–NRNPEGC(=NRNPEG)N(RNPEG)–、–C(S)–、–C(S)N(RNPEG)–、–NRNPEGC(S)–、–NRNPEGC(S)N(RNPEG)–、–S(O)–、–OS(O)–、–S(O)O–、–OS(O)O–、–OS(O)2–、–S(O)2O–、–OS(O)2O–、–N(RNPEG)S(O)–、–S(O)N(RNPEG)–、–N(RNPEG)S(O)N(RNPEG)–、–OS(O)N(RNPEG)–、–N(RNPEG)S(O)O–、–S(O)2–、–N(RNPEG)S(O)2–、–S(O)2N(RNPEG)–、–N(RNPEG)S(O)2N(RNPEG)–、–OS(O)2N(RNPEG)–或–N(RNPEG)S(O)2O–;并且R 5PEG is C 10-40 alkyl, C 10-40 alkenyl, or optionally substituted C 10-40 alkynyl; and optionally one or more methylene groups of R 5PEG are independently replaced with: C 3 -10 -membered carbocyclylene, 4- to 10-membered heterocyclylene, C 6-10 -membered arylene, 4- to 10-membered heteroarylene, -N(R NPEG )-, -O-, -S-, - C(O)–, –C(O)N(R NPEG )–, –NR NPEG C(O)–, –NR NPEG C(O)N(R NPEG )–, –C(O)O–, – OC(O)–, –OC(O)O–, –OC(O)N(R NPEG )–, –NR NPEG C(O)O–, –C(O)S–, –SC(O)– , –C(=NR NPEG )–, –C(=NR NPEG )N(R NPEG )–, –NR NPEG C(=NR NPEG )–, –NR NPEG C(=NR NPEG )N(R NPEG )– , –C(S)–, –C(S)N(R NPEG )–, –NR NPEG C(S)–, –NR NPEG C(S)N(R NPEG )–, –S(O)–, –OS(O)–, –S(O)O–, –OS(O)O–, –OS(O) 2 –, –S(O) 2 O–, –OS(O) 2 O–, – N(R NPEG )S(O)–, –S(O)N(R NPEG )–, –N(R NPEG )S(O)N(R NPEG )–, –OS(O)N(R NPEG ) –, –N(R NPEG )S(O)O–, –S(O) 2 –, –N(R NPEG )S(O) 2 –, –S(O) 2 N(R NPEG )–, – N(R NPEG )S(O) 2 N(R NPEG )–, –OS(O) 2 N(R NPEG )– or –N(R NPEG )S(O) 2 O–; and
RNPEG的每种情况独立地是氢、C1-6烷基或氮保护基团。Each instance of R NPEG is independently a hydrogen, C1-6 alkyl or nitrogen protecting group.
实施方案87.如前述实施方案中任一项所述的空LNP或负载LNP,其中所述式(PL-II-OH)的PEG脂质,r是介于40与50之间的整数。Embodiment 87. The empty or supported LNP of any one of the preceding embodiments, wherein the PEG lipid of formula (PL-II-OH), r is an integer between 40 and 50.
实施方案88.如前述实施方案中任一项所述的空LNP或负载LNP,其中所述式(PL-II-OH)的PEG脂质,r是45。Embodiment 88. The empty or loaded LNP of any one of the preceding embodiments, wherein the PEG lipid of formula (PL-II-OH), r is 45.
实施方案89.如前述实施方案中任一项所述的空LNP或负载LNP,其中所述式(PL-II-OH)的PEG脂质,R5是C17烷基。Embodiment 89. The empty or supported LNP of any one of the preceding embodiments, wherein the PEG lipid of formula (PL-II - OH), R5 is C17 alkyl.
实施方案90.如前述实施方案中任一项所述的空LNP或负载LNP,其中所述式(PL-II)的PEG脂质:其中rPEG是介于1与100之间的整数。Embodiment 90. The empty or loaded LNP of any one of the preceding embodiments, wherein the PEG lipid of formula (PL-II): where rPEG is an integer between 1 and 100.
实施方案91.如前述实施方案中任一项所述的空LNP或负载LNP,其中所述PEG脂质是式(PEG-1)的化合物:Embodiment 91. The empty LNP or loaded LNP of any one of the preceding embodiments, wherein the PEG lipid is a compound of formula (PEG-1):
实施方案92.如前述实施方案中任一项所述的空LNP或负载LNP,其中所述PEG脂质是式(PL-III)的化合物:或其盐或异构体,其中sPL1是介于1与100之间的整数。Embodiment 92. The empty or loaded LNP of any one of the preceding embodiments, wherein the PEG lipid is a compound of formula (PL-III): or a salt or isomer thereof, wherein s PL1 is an integer between 1 and 100.
实施方案93.如前述实施方案中任一项所述的空LNP或负载LNP,其中所述PEG脂质是具有下式的化合物: Embodiment 93. The empty or loaded LNP of any one of the preceding embodiments, wherein the PEG lipid is a compound of the formula:
实施方案94.一种空脂质纳米粒子(空LNP),其包含如前述实施方案中任一项所述的化合物、磷脂、结构性脂质和PEG脂质,其中所述磷脂是DSPC并且所述结构性脂质是胆固醇。Embodiment 94. An empty lipid nanoparticle (empty LNP) comprising the compound of any one of the preceding embodiments, a phospholipid, a structured lipid, and a PEG lipid, wherein the phospholipid is DSPC and the The structural lipid is cholesterol.
实施方案95.一种空脂质纳米粒子(空LNP),其包含如前述实施方案中任一项所述的化合物、磷脂、结构性脂质和PEG脂质,其中所述结构性脂质是胆固醇并且所述PEG脂质是PEG2k-DMG。Embodiment 95. An empty lipid nanoparticle (empty LNP) comprising the compound of any one of the preceding embodiments, a phospholipid, a structured lipid, and a PEG lipid, wherein the structured lipid is Cholesterol and the PEG lipid was PEG 2k -DMG.
实施方案96.一种空脂质纳米粒子(空LNP),其包含如前述实施方案中任一项所述的化合物、磷脂、结构性脂质和PEG脂质,其中所述结构性脂质是胆固醇并且所述PEG脂质是PEG-1。Embodiment 96. An empty lipid nanoparticle (empty LNP) comprising the compound of any one of the preceding embodiments, a phospholipid, a structured lipid, and a PEG lipid, wherein the structured lipid is cholesterol and the PEG lipid is PEG-1.
实施方案97.一种空脂质纳米粒子(空LNP),其包含如前述实施方案中任一项所述的化合物、磷脂、结构性脂质和PEG脂质,其中所述磷脂是DSPC并且所述PEG脂质是PEG2k-DMG。Embodiment 97. An empty lipid nanoparticle (empty LNP) comprising the compound of any one of the preceding embodiments, a phospholipid, a structured lipid, and a PEG lipid, wherein the phospholipid is DSPC and the The PEG lipid is PEG 2k -DMG.
实施方案98.一种空脂质纳米粒子(空LNP),其包含如前述实施方案中任一项所述的化合物、磷脂、结构性脂质和PEG脂质,其中所述磷脂是DSPC并且所述PEG脂质是PEG-1。Embodiment 98. An empty lipid nanoparticle (empty LNP) comprising the compound of any one of the preceding embodiments, a phospholipid, a structured lipid, and a PEG lipid, wherein the phospholipid is DSPC and the The PEG lipid is PEG-1.
实施方案99.一种空脂质纳米粒子(空LNP),其包含如前述实施方案中任一项所述的化合物、磷脂、结构性脂质和PEG脂质,其中所述磷脂是DSPC,所述结构性脂质是胆固醇并且所述PEG脂质是PEG2k-DMG。Embodiment 99. An empty lipid nanoparticle (empty LNP) comprising the compound of any one of the preceding embodiments, a phospholipid, a structured lipid, and a PEG lipid, wherein the phospholipid is DSPC, wherein the The structural lipid is cholesterol and the PEG lipid is PEG 2k -DMG.
实施方案100.一种空脂质纳米粒子(空LNP),其包含如前述实施方案中任一项所述的化合物、磷脂、结构性脂质和PEG脂质,其中所述磷脂是DSPC,所述结构性脂质是胆固醇并且所述PEG脂质是PEG-1。Embodiment 100. An empty lipid nanoparticle (empty LNP) comprising the compound of any one of the preceding embodiments, a phospholipid, a structured lipid, and a PEG lipid, wherein the phospholipid is DSPC, wherein the The structural lipid is cholesterol and the PEG lipid is PEG-1.
实施方案101.一种负载脂质纳米粒子(负载LNP),其包含如前述实施方案中任一项所述的化合物、磷脂、结构性脂质和PEG脂质以及一种或多种治疗剂和/或预防剂,其中所述磷脂是DSPC并且所述结构性脂质是胆固醇。Embodiment 101. A lipid-loaded nanoparticle (loaded LNP) comprising a compound of any of the preceding embodiments, a phospholipid, a structured lipid, and a PEG lipid and one or more therapeutic agents and /or prophylactic, wherein the phospholipid is DSPC and the structural lipid is cholesterol.
实施方案102.一种负载脂质纳米粒子(负载LNP),其包含如前述实施方案中任一项所述的化合物、磷脂、结构性脂质和PEG脂质以及一种或多种治疗剂和/或预防剂,其中所述结构性脂质是胆固醇并且所述PEG脂质是PEG2k-DMG。Embodiment 102. A lipid-loaded nanoparticle (loaded LNP) comprising the compound of any of the preceding embodiments, a phospholipid, a structured lipid, and a PEG lipid, and one or more therapeutic agents and /or prophylactic, wherein the structural lipid is cholesterol and the PEG lipid is PEG 2k -DMG.
实施方案103.一种负载脂质纳米粒子(负载LNP),其包含如前述实施方案中任一项所述的化合物、磷脂、结构性脂质和PEG脂质以及一种或多种治疗剂和/或预防剂,其中所述结构性脂质是胆固醇并且所述PEG脂质是PEG-1。Embodiment 103. A lipid-loaded nanoparticle (loaded LNP) comprising a compound of any of the preceding embodiments, a phospholipid, a structured lipid, and a PEG lipid, and one or more therapeutic agents and /or prophylactic, wherein the structural lipid is cholesterol and the PEG lipid is PEG-1.
实施方案104.一种负载脂质纳米粒子(负载LNP),其包含如前述实施方案中任一项所述的化合物、磷脂、结构性脂质和PEG脂质以及一种或多种治疗剂和/或预防剂,其中所述磷脂是DSPC并且所述PEG脂质是PEG2k-DMG。Embodiment 104. A lipid-loaded nanoparticle (loaded LNP) comprising the compound of any of the preceding embodiments, a phospholipid, a structured lipid, and a PEG lipid, and one or more therapeutic agents and /or prophylactic, wherein the phospholipid is DSPC and the PEG lipid is PEG 2k -DMG.
实施方案105.一种负载脂质纳米粒子(负载LNP),其包含如前述实施方案中任一项所述的化合物、磷脂、结构性脂质和PEG脂质以及一种或多种治疗剂和/或预防剂,其中所述磷脂是DSPC并且所述PEG脂质是PEG-1。Embodiment 105. A lipid-loaded nanoparticle (loaded LNP) comprising the compound of any of the preceding embodiments, a phospholipid, a structured lipid, and a PEG lipid, and one or more therapeutic agents and /or prophylactic, wherein the phospholipid is DSPC and the PEG lipid is PEG-1.
实施方案106.一种负载脂质纳米粒子(负载LNP),其包含如前述实施方案中任一项所述的化合物、磷脂、结构性脂质和PEG脂质以及一种或多种治疗剂和/或预防剂,其中所述磷脂是DSPC,所述结构性脂质是胆固醇并且所述PEG脂质是PEG2k-DMG。Embodiment 106. A lipid-loaded nanoparticle (loaded LNP) comprising a compound of any one of the preceding embodiments, a phospholipid, a structured lipid, and a PEG lipid and one or more therapeutic agents and /or prophylactic, wherein the phospholipid is DSPC, the structural lipid is cholesterol and the PEG lipid is PEG 2k -DMG.
实施方案107.一种负载脂质纳米粒子(负载LNP),其包含如前述实施方案中任一项所述的化合物、磷脂、结构性脂质和PEG脂质以及一种或多种治疗剂和/或预防剂,其中所述磷脂是DSPC,所述结构性脂质是胆固醇并且所述PEG脂质是PEG-1。Embodiment 107. A lipid-loaded nanoparticle (loaded LNP) comprising a compound of any one of the preceding embodiments, a phospholipid, a structured lipid, and a PEG lipid and one or more therapeutic agents and /or prophylactic, wherein the phospholipid is DSPC, the structural lipid is cholesterol and the PEG lipid is PEG-1.
实施方案108.如前述实施方案中任一项所述的空LNP或负载LNP,其包含约0%至约20%的量的DSPC。Embodiment 108. The empty or loaded LNP of any one of the preceding embodiments, comprising DSPC in an amount from about 0% to about 20%.
实施方案109.如前述实施方案中任一项所述的空LNP或负载LNP,其包含约30%至约50%的量的胆固醇。Embodiment 109. The empty or loaded LNP of any of the preceding embodiments, comprising cholesterol in an amount from about 30% to about 50%.
实施方案110.如前述实施方案中任一项所述的空LNP或负载LNP,其包含约0%至约5%的量的PEG2k-DMG。Embodiment 110. The empty or loaded LNP of any of the preceding embodiments, comprising PEG2k -DMG in an amount of about 0% to about 5%.
实施方案111.如前述实施方案中任一项所述的空LNP或负载LNP,其包含约0%至约5%的量的PEG-1。Embodiment 111. The empty or loaded LNP of any of the preceding embodiments, comprising PEG-1 in an amount from about 0% to about 5%.
实施方案112.如前述实施方案中任一项所述的空LNP或负载LNP,其包含约40mol%至约60mol%的如前述实施方案中任一项所述的化合物;约0mol%至约20mol%DSPC;约30mol%至约50mol%胆固醇;以及约0mol%至约5mol%PEG2k-DMG。Embodiment 112. The empty or supported LNP of any of the preceding embodiments, comprising from about 40 mol % to about 60 mol % of the compound of any preceding embodiment; from about 0 mol % to about 20 mol % % DSPC; about 30 mol % to about 50 mol % cholesterol; and about 0 mol % to about 5 mol % PEG 2k -DMG.
实施方案113.如前述实施方案中任一项所述的空LNP或负载LNP,其包含约40mol%至约60mol%的如前述实施方案中任一项所述的化合物;约0mol%至约20mol%DSPC;约30mol%至约50mol%胆固醇;以及约0mol%至约5mol%PEG-1。Embodiment 113. The empty LNP or loaded LNP of any preceding embodiment, comprising from about 40 mol % to about 60 mol % of the compound of any preceding embodiment; from about 0 mol % to about 20 mol % % DSPC; about 30 mol % to about 50 mol % cholesterol; and about 0 mol % to about 5 mol % PEG-1.
实施方案114.如前述实施方案中任一项所述的负载LNP,所述治疗剂和/或预防剂的包封效率介于80%与100%之间。Embodiment 114. The loaded LNP of any one of the preceding embodiments, the encapsulation efficiency of the therapeutic and/or prophylactic agent is between 80% and 100%.
实施方案115.如前述实施方案中任一项所述的负载LNP,其中所述脂质组分与所述mRNA的重量/重量比为约10:1至约60:1。Embodiment 115. The loaded LNP of any of the preceding embodiments, wherein the weight/weight ratio of the lipid component to the mRNA is from about 10:1 to about 60:1.
实施方案116.如前述实施方案中任一项所述的负载LNP,其中所述脂质组分与所述mRNA的重量/重量比为20:1。Embodiment 116. The loaded LNP of any of the preceding embodiments, wherein the weight/weight ratio of the lipid component to the mRNA is 20:1.
实施方案117.如前述实施方案中任一项所述的负载LNP,其中所述N:P比为约5:1至约8:1。Embodiment 117. The supported LNP of any of the preceding embodiments, wherein the N:P ratio is from about 5:1 to about 8:1.
实施方案118.一种药物组合物,其包括如前述实施方案中任一项所述的负载LNP和药学上可接受的载剂。Embodiment 118. A pharmaceutical composition comprising the loaded LNP of any of the preceding embodiments and a pharmaceutically acceptable carrier.
实施方案119.如前述实施方案中任一项所述的药物组合物,其还包含冷冻保护剂、缓冲剂或其组合。Embodiment 119. The pharmaceutical composition of any of the preceding embodiments, further comprising a cryoprotectant, a buffer, or a combination thereof.
实施方案120.如前述实施方案中任一项所述的药物组合物,其中所述冷冻保护剂包括蔗糖。Embodiment 120. The pharmaceutical composition of any of the preceding embodiments, wherein the cryoprotectant comprises sucrose.
实施方案121.如前述实施方案中任一项所述的药物组合物,其中所述冷冻保护剂包括乙酸钠。Embodiment 121. The pharmaceutical composition of any of the preceding embodiments, wherein the cryoprotectant comprises sodium acetate.
实施方案122.如前述实施方案中任一项所述的药物组合物,其中所述冷冻保护剂包括蔗糖和乙酸钠。Embodiment 122. The pharmaceutical composition of any of the preceding embodiments, wherein the cryoprotectant comprises sucrose and sodium acetate.
实施方案123.如前述实施方案中任一项所述的药物组合物,其中所述缓冲剂选自由以下组成的组:乙酸盐缓冲剂、柠檬酸盐缓冲剂、磷酸盐缓冲剂和tris缓冲剂。Embodiment 123. The pharmaceutical composition of any of the preceding embodiments, wherein the buffer is selected from the group consisting of acetate buffer, citrate buffer, phosphate buffer, and tris buffer agent.
实施方案124.一种将治疗剂和/或预防剂递送至受试者体内的细胞的方法,所述方法包括向所述受试者施用如前述实施方案中任一项所述的负载LNP。Embodiment 124. A method of delivering a therapeutic and/or prophylactic agent to cells in a subject, the method comprising administering to the subject the loaded LNP of any of the preceding embodiments.
实施方案125.一种将治疗剂和/或预防剂特异性递送至受试者的器官的方法,所述方法包括向所述受试者施用如前述实施方案中任一项所述的负载LNP。Embodiment 125. A method of specifically delivering a therapeutic and/or prophylactic agent to an organ of a subject, the method comprising administering to the subject the loaded LNP of any one of the preceding embodiments .
实施方案126.一种用于将治疗剂和/或预防剂增强递送至受试者的靶组织的方法,所述方法包括向所述受试者施用如前述实施方案中任一项所述的负载LNP。Embodiment 126. A method for enhanced delivery of a therapeutic and/or prophylactic agent to a target tissue in a subject, the method comprising administering to the subject the method of any of the preceding embodiments Load LNP.
实施方案127.一种在受试者体内的细胞中产生所关注的多肽的方法,所述方法包括向所述受试者施用如前述实施方案中任一项所述的负载LNP。Embodiment 127. A method of producing a polypeptide of interest in a cell in a subject, the method comprising administering to the subject the loaded LNP of any of the preceding embodiments.
实施方案128.一种治疗有需要的受试者的疾病或病症的方法,所述方法包括向所述受试者施用治疗有效量的如前述实施方案中任一项所述的负载LNP。Embodiment 128. A method of treating a disease or disorder in a subject in need thereof, the method comprising administering to the subject a therapeutically effective amount of the loaded LNP of any of the preceding embodiments.
实施方案129.如前述实施方案中任一项所述的负载LNP在制造用于将治疗剂和/或预防剂递送至受试者体内的细胞的药物中的用途。Embodiment 129. Use of the loaded LNP of any of the preceding embodiments in the manufacture of a medicament for delivering a therapeutic and/or prophylactic agent to cells in a subject.
实施方案130.如前述实施方案中任一项所述的负载LNP在制造用于将治疗剂和/或预防剂特异性递送至受试者的器官的药物中的用途。Embodiment 130. Use of the loaded LNP of any of the preceding embodiments in the manufacture of a medicament for the specific delivery of a therapeutic and/or prophylactic agent to an organ of a subject.
实施方案131.如前述实施方案中任一项所述的负载LNP在制造用于将治疗剂和/或预防剂增强递送至受试者的靶组织的药物中的用途。Embodiment 131. Use of the loaded LNP of any of the preceding embodiments in the manufacture of a medicament for enhanced delivery of a therapeutic and/or prophylactic agent to a target tissue of a subject.
实施方案132.如前述实施方案中任一项所述的负载LNP在制造用于在受试者体内的细胞中产生所关注的多肽的药物中的用途。Embodiment 132. Use of the loaded LNP of any of the preceding embodiments in the manufacture of a medicament for producing a polypeptide of interest in cells in a subject.
实施方案133.如前述实施方案中任一项所述的负载LNP在制造用于治疗有需要的受试者的疾病或病症的药物中的用途。Embodiment 133. Use of the loaded LNP of any of the preceding embodiments in the manufacture of a medicament for the treatment of a disease or disorder in a subject in need thereof.
实施方案134.如前述实施方案中任一项所述的负载LNP,其用于将治疗剂和/或预防剂递送至受试者体内的细胞,其中所述递送包括向所述受试者施用治疗有效量的所述负载LNP。Embodiment 134. The loaded LNP of any one of the preceding embodiments for use in delivering a therapeutic and/or prophylactic agent to a cell in a subject, wherein the delivery comprises administering to the subject A therapeutically effective amount of the loaded LNP.
实施方案135.如前述实施方案中任一项所述的负载LNP,其用于将治疗剂和/或预防剂特异性递送至受试者的器官,其中所述递送包括向所述受试者施用治疗有效量的所述负载LNP。Embodiment 135. The load LNP of any one of the preceding embodiments for use in the specific delivery of a therapeutic agent and/or a prophylactic agent to an organ of a subject, wherein the delivery comprises to the subject A therapeutically effective amount of the loaded LNP is administered.
实施方案136.如前述实施方案中任一项所述的负载LNP,其用于将治疗剂和/或预防剂增强递送至受试者的靶组织,其中所述使用包括向所述受试者施用如前述实施方案中任一项所述的负载LNP。Embodiment 136. The loaded LNP of any one of the preceding embodiments for enhanced delivery of a therapeutic and/or prophylactic agent to a subject's target tissue, wherein the use comprises delivering to the subject A loaded LNP according to any of the preceding embodiments is administered.
实施方案137.如前述实施方案中任一项所述的负载LNP,其用于在受试者体内的细胞中产生所关注的多肽,所述使用包括向所述受试者施用如前述实施方案中任一项所述的负载LNP。Embodiment 137. The load LNP of any one of the preceding embodiments, which is used to produce a polypeptide of interest in a cell in a subject, the use comprising administering to the subject the use of the aforementioned embodiments The load LNP of any one.
实施方案138.如前述实施方案中任一项所述的负载LNP,其用于治疗有需要的受试者的疾病或病症,其中所述治疗包括向受试者施用治疗有效量的所述负载LNP。Embodiment 138. The load LNP of any one of the preceding embodiments for use in the treatment of a disease or condition in a subject in need, wherein the treatment comprises administering to the subject a therapeutically effective amount of the load LNP.
实施方案139.一种将治疗剂和/或预防剂递送至受试者体内的细胞的方法,所述方法包括向所述受试者施用如前述实施方案中任一项所述的药物组合物。Embodiment 139. A method of delivering a therapeutic and/or prophylactic agent to cells in a subject, the method comprising administering to the subject the pharmaceutical composition of any of the preceding embodiments .
实施方案140.一种将治疗剂和/或预防剂特异性递送至受试者的器官的方法,所述方法包括向所述受试者施用如前述实施方案中任一项所述的药物组合物。Embodiment 140. A method of specifically delivering a therapeutic and/or prophylactic agent to an organ of a subject, the method comprising administering to the subject the pharmaceutical combination of any of the preceding embodiments thing.
实施方案141.一种用于将治疗剂和/或预防剂增强递送至受试者的靶组织的方法,所述方法包括向所述受试者施用如前述实施方案中任一项所述的药物组合物。Embodiment 141. A method for enhanced delivery of a therapeutic and/or prophylactic agent to a target tissue in a subject, the method comprising administering to the subject the method of any of the preceding embodiments pharmaceutical composition.
实施方案142.一种在受试者体内的细胞中产生所关注的多肽的方法,所述方法包括向所述受试者施用如前述实施方案中任一项所述的负载LNP。Embodiment 142. A method of producing a polypeptide of interest in a cell in a subject, the method comprising administering to the subject the loaded LNP of any of the preceding embodiments.
实施方案143.一种治疗有需要的受试者的疾病或病症的方法,所述方法包括向所述受试者施用治疗有效量的如前述实施方案中任一项所述的药物组合物。Embodiment 143. A method of treating a disease or disorder in a subject in need thereof, the method comprising administering to the subject a therapeutically effective amount of the pharmaceutical composition of any of the preceding embodiments.
实施方案144.如前述实施方案中任一项所述的药物组合物在制造用于将治疗剂和/或预防剂递送至受试者体内的细胞的药物中的用途。Embodiment 144. Use of the pharmaceutical composition of any of the preceding embodiments in the manufacture of a medicament for delivering a therapeutic and/or prophylactic agent to cells in a subject.
实施方案145.如前述实施方案中任一项所述的药物组合物在制造用于将治疗剂和/或预防剂特异性递送至受试者的器官的药物中的用途。Embodiment 145. Use of the pharmaceutical composition of any of the preceding embodiments in the manufacture of a medicament for the specific delivery of a therapeutic and/or prophylactic agent to an organ of a subject.
实施方案146.如前述实施方案中任一项所述的药物组合物在将治疗剂和/或预防剂增强递送至受试者的靶组织中的用途,所述方法包括向所述受试者施用如前述实施方案中任一项所述的药物组合物。Embodiment 146. Use of the pharmaceutical composition of any of the preceding embodiments for enhanced delivery of a therapeutic and/or prophylactic agent to a target tissue in a subject, the method comprising administering to the subject The pharmaceutical composition of any of the preceding embodiments is administered.
实施方案147.如前述实施方案中任一项所述的药物组合物在制造用于在受试者体内的细胞中产生所关注的多肽的药物中的用途。Embodiment 147. Use of the pharmaceutical composition of any of the preceding embodiments in the manufacture of a medicament for producing a polypeptide of interest in cells in a subject.
实施方案148.如前述实施方案中任一项所述的药物组合物在制造用于治疗有需要的受试者的疾病或病症的药物中的用途。Embodiment 148. Use of the pharmaceutical composition of any of the preceding embodiments in the manufacture of a medicament for the treatment of a disease or disorder in a subject in need thereof.
实施方案149.如前述实施方案中任一项所述的药物组合物,其用于将治疗剂和/或预防剂递送至受试者体内的细胞,其中所述递送包括向所述受试者施用治疗有效量的所述药物组合物。Embodiment 149. The pharmaceutical composition of any one of the preceding embodiments, for use in delivering a therapeutic and/or prophylactic agent to cells in a subject, wherein the delivery comprises delivering to the subject A therapeutically effective amount of the pharmaceutical composition is administered.
实施方案150.如前述实施方案中任一项所述的药物组合物,其用于将治疗剂和/或预防剂特异性递送至受试者的器官,其中所述递送包括向所述受试者施用治疗有效量的所述药物组合物。Embodiment 150. The pharmaceutical composition of any one of the preceding embodiments, for use in the specific delivery of a therapeutic and/or prophylactic agent to an organ of a subject, wherein the delivery comprises delivering to the subject is administered a therapeutically effective amount of the pharmaceutical composition.
实施方案151.如前述实施方案中任一项所述的药物组合物,其用于将治疗剂和/或预防剂增强递送至受试者的靶组织,其中所述使用包括向所述受试者施用如前述实施方案中任一项所述的药物组合物。Embodiment 151. The pharmaceutical composition of any one of the preceding embodiments for enhanced delivery of a therapeutic and/or prophylactic agent to a target tissue in a subject, wherein the using comprises delivering to the subject to administer the pharmaceutical composition of any of the preceding embodiments.
实施方案152.如前述实施方案中任一项所述的药物组合物,其用于在受试者体内的细胞中产生所关注的多肽,所述使用包括向所述受试者施用如前述实施方案中任一项所述的药物组合物。Embodiment 152. The pharmaceutical composition of any one of the preceding embodiments, which is used to produce a polypeptide of interest in a cell in a subject, the use comprising administering to the subject as described previously The pharmaceutical composition of any one of the schemes.
实施方案153.如前述实施方案中任一项所述的药物组合物,其用于治疗有需要的受试者的疾病或病症,其中所述治疗包括向受试者施用治疗有效量的所述药物组合物。Embodiment 153. The pharmaceutical composition of any one of the preceding embodiments, for use in the treatment of a disease or disorder in a subject in need thereof, wherein the treatment comprises administering to the subject a therapeutically effective amount of the pharmaceutical composition.
实施方案154.如前述实施方案中任一项所述的方法、用途或供使用的负载LNP或药物组合物,其中所述器官选自由肝、肾、脾或肺组成的组。Embodiment 154. The method, use or loaded LNP or pharmaceutical composition for use of any of the preceding embodiments, wherein the organ is selected from the group consisting of liver, kidney, spleen or lung.
实施方案155.如前述实施方案中任一项所述的方法、用途或供使用的负载LNP或药物组合物,其中所述靶组织选自由肝、肾、脾或肺组成的组。Embodiment 155. The method, use or loaded LNP or pharmaceutical composition for use of any of the preceding embodiments, wherein the target tissue is selected from the group consisting of liver, kidney, spleen or lung.
实施方案156.如前述实施方案中任一项所述的方法、用途或供使用的负载LNP或药物组合物,其中所述施用是肠胃外进行的。Embodiment 156. The method, use or loaded LNP or pharmaceutical composition for use of any of the preceding embodiments, wherein the administering is parenterally.
实施方案157.如前述实施方案中任一项所述的方法或供使用的负载LNP或药物组合物,其中所述施用是肌肉内、皮内、皮下和/或静脉内进行的。Embodiment 157. The method or loaded LNP or pharmaceutical composition for use of any one of the preceding embodiments, wherein the administration is intramuscular, intradermal, subcutaneous and/or intravenous.
实施方案158.如前述实施方案中任一项所述的用途,其中所述药物用于肠胃外施用。Embodiment 158. The use of any of the preceding embodiments, wherein the medicament is for parenteral administration.
实施方案159.如前述实施方案中任一项所述的用途,其中所述药物用于肌肉内、皮内、皮下和/或静脉内施用。Embodiment 159. The use of any of the preceding embodiments, wherein the medicament is for intramuscular, intradermal, subcutaneous and/or intravenous administration.
实施方案160.如前述实施方案中任一项所述的方法、用途或供使用的负载LNP或药物组合物,其中所述所述受试者是人。Embodiment 160. The method, use or loaded LNP or pharmaceutical composition for use of any of the preceding embodiments, wherein the subject is a human.
等效方案Equivalent scheme
应理解,虽然已经结合本公开的详述描述了本公开,但是前述描述旨在说明而不是限制本公开的范围,本公开的范围由所附权利要求的范围限定。其他方面、优点和变更在所附权利要求的范围内。It should be understood that, while the present disclosure has been described in conjunction with the detailed description of the present disclosure, the foregoing description is intended to illustrate, and not to limit, the scope of the disclosure, which is defined by the scope of the appended claims. Other aspects, advantages and modifications are within the scope of the appended claims.
Claims (32)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201962902929P | 2019-09-19 | 2019-09-19 | |
| US62/902929 | 2019-09-19 | ||
| PCT/US2020/051613 WO2021055835A1 (en) | 2019-09-19 | 2020-09-18 | Carbonate containing lipid compounds and compositions for intracellular delivery of therapeutic agents |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN114728886A true CN114728886A (en) | 2022-07-08 |
| CN114728886B CN114728886B (en) | 2024-04-12 |
Family
ID=72752506
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202080080295.1A Active CN114728886B (en) | 2019-09-19 | 2020-09-18 | Lipid compounds and compositions containing carbonates for intracellular delivery of therapeutic agents |
Country Status (11)
| Country | Link |
|---|---|
| US (1) | US20220411361A1 (en) |
| EP (1) | EP4031521B1 (en) |
| JP (1) | JP7638973B2 (en) |
| KR (1) | KR20220101076A (en) |
| CN (1) | CN114728886B (en) |
| AU (1) | AU2020351220A1 (en) |
| BR (1) | BR112022004936A2 (en) |
| CA (1) | CA3155015A1 (en) |
| IL (1) | IL291339A (en) |
| MX (1) | MX2022003285A (en) |
| WO (1) | WO2021055835A1 (en) |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN115784920A (en) * | 2023-02-09 | 2023-03-14 | 荣灿生物医药技术(上海)有限公司 | Ionizable lipid compound with high transfection efficiency and application thereof |
| CN116947669A (en) * | 2022-09-30 | 2023-10-27 | 荣灿生物医药技术(上海)有限公司 | Ionizable lipid compound with high transfection efficiency and application thereof |
| WO2024022263A1 (en) * | 2022-07-25 | 2024-02-01 | 苏州艾博生物科技有限公司 | Lipid compound and lipid nanoparticle composition |
| CN117777089A (en) * | 2022-12-28 | 2024-03-29 | 仁景(苏州)生物科技有限公司 | Lipid compounds and lipid nanoparticle compositions |
| WO2024067639A1 (en) * | 2022-09-30 | 2024-04-04 | 荣灿生物医药技术(上海)有限公司 | Ionizable lipid compound having high transfection efficiency and use thereof |
| WO2024145964A1 (en) * | 2023-01-05 | 2024-07-11 | 北京悦康科创医药科技股份有限公司 | Cationic lipid compound with high efficiency, low toxicity and stable expression, and composition comprising same |
| WO2025146125A1 (en) * | 2024-01-05 | 2025-07-10 | 仁景(苏州)生物科技有限公司 | Lipid compound and lipid nanoparticle for delivery |
| WO2025161943A1 (en) * | 2024-02-02 | 2025-08-07 | 仁景(苏州)生物科技有限公司 | Lipid compound and lipid nanoparticle for delivery |
Families Citing this family (49)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ES2969082T3 (en) | 2015-09-17 | 2024-05-16 | Modernatx Inc | Compounds and compositions for intracellular administration of therapeutic agents |
| US10799463B2 (en) | 2015-12-22 | 2020-10-13 | Modernatx, Inc. | Compounds and compositions for intracellular delivery of agents |
| US11583504B2 (en) | 2016-11-08 | 2023-02-21 | Modernatx, Inc. | Stabilized formulations of lipid nanoparticles |
| MX2019011004A (en) | 2017-03-15 | 2020-08-10 | Modernatx Inc | Compounds and compositions for intracellular delivery of therapeutic agents. |
| CA3055653A1 (en) | 2017-03-15 | 2018-09-20 | Modernatx, Inc. | Lipid nanoparticle formulation |
| US12077501B2 (en) | 2017-06-14 | 2024-09-03 | Modernatx, Inc. | Compounds and compositions for intracellular delivery of agents |
| EP3853202A1 (en) | 2018-09-19 | 2021-07-28 | ModernaTX, Inc. | Compounds and compositions for intracellular delivery of therapeutic agents |
| EP3917656A1 (en) | 2019-01-31 | 2021-12-08 | Modernatx, Inc. | Vortex mixers and associated methods, systems, and apparatuses thereof |
| JP7638972B2 (en) | 2019-09-19 | 2025-03-04 | モデルナティエックス インコーポレイテッド | Branched tail lipid compounds and compositions for intracellular delivery of therapeutic agents - Patents.com |
| MX2023006056A (en) | 2020-11-25 | 2023-06-06 | Akagera Medicines Inc | Lipid nanoparticles for delivery of nucleic acids, and related methods of use. |
| CN116685332A (en) * | 2020-12-21 | 2023-09-01 | 比姆医疗股份有限公司 | Nanomaterials including carbonates |
| CN115197078B (en) * | 2021-04-08 | 2024-06-04 | 厦门赛诺邦格生物科技股份有限公司 | Pegylated lipid and modified liposome thereof, pharmaceutical composition containing liposome and preparation and application of pharmaceutical composition |
| US20240350640A1 (en) * | 2021-06-30 | 2024-10-24 | Jenkem Technology Co., Ltd. (Tianjin) | Polyethylene glycol lipid and use thereof |
| CN114149337B (en) * | 2021-07-07 | 2022-04-29 | 天津键凯科技有限公司 | Novel ionizable lipid for nucleic acid delivery and LNP composition thereof |
| CN115710191B (en) * | 2021-08-23 | 2025-06-10 | 广州谷森制药有限公司 | Cationic lipid compounds |
| CA3230031A1 (en) | 2021-09-03 | 2023-03-09 | Patrick Baumhof | Novel lipid nanoparticles for delivery of nucleic acids |
| US20250027108A1 (en) | 2021-10-29 | 2025-01-23 | CureVac SE | Improved circular rna for expressing therapeutic proteins |
| AR127892A1 (en) | 2021-12-10 | 2024-03-06 | Modernatx Inc | COMPOUNDS AND COMPOSITIONS FOR THE ADMINISTRATION OF THERAPEUTIC AGENTS |
| US20230183168A1 (en) * | 2021-12-14 | 2023-06-15 | Massachusetts Institute Of Technology | Ionizable lipids and compositions and uses thereof |
| EP4469091A1 (en) | 2022-01-28 | 2024-12-04 | CureVac SE | Nucleic acid encoded transcription factor inhibitors |
| CN116554042A (en) * | 2022-01-30 | 2023-08-08 | 康希诺生物股份公司 | A Novel Ionizable Lipid for Nucleic Acid Delivery and Its LNP Composition and Vaccine |
| CN116514672B (en) * | 2022-01-30 | 2025-06-27 | 康希诺生物股份公司 | Ionizable lipid for nucleic acid delivery, LNP composition and vaccine thereof |
| WO2023143600A1 (en) * | 2022-01-30 | 2023-08-03 | 康希诺生物股份公司 | Novel ionizable lipid for nucleic acid delivery, and lnp composition and vaccine thereof |
| US20250235531A1 (en) | 2022-02-09 | 2025-07-24 | Modernatx, Inc. | Mucosal administration methods and formulations |
| JP2025517508A (en) | 2022-05-25 | 2025-06-05 | キュアバック エスイー | Nucleic Acid-Based Vaccines |
| KR20250031230A (en) | 2022-05-25 | 2025-03-06 | 아카제라 메디신즈, 인크. | Lipid nanoparticles for nucleic acid delivery and methods of using the same |
| CN116969850A (en) * | 2022-06-14 | 2023-10-31 | 苏州科锐迈德生物医药科技有限公司 | Carbonate-carboxylate lipid compounds and lipid carriers based thereon, nucleic acid lipid nanoparticle compositions and pharmaceutical preparations |
| CA3262828A1 (en) * | 2022-07-28 | 2024-02-01 | Aldexchem Kft. | Ionizable cationic lipids incorporating silicon |
| DE202023106198U1 (en) | 2022-10-28 | 2024-03-21 | CureVac SE | Nucleic acid-based vaccine |
| CN120225500A (en) | 2022-11-18 | 2025-06-27 | 韩国生命工学研究院 | Lipid compounds and compositions for active substance delivery |
| EP4371976A1 (en) * | 2022-11-21 | 2024-05-22 | Certest Biotec, S.L. | Ionizable lipids and lipid nanoparticles containing thereof |
| TW202428269A (en) * | 2022-11-21 | 2024-07-16 | 西班牙商塞提斯生物技術公司 | Ionizable lipids and lipid nanoparticles containing thereof |
| EP4630397A1 (en) | 2022-12-08 | 2025-10-15 | ModernaTX, Inc. | Ionizable lipids with malonate tails |
| CN118236344A (en) * | 2022-12-15 | 2024-06-25 | 深圳瑞吉生物科技有限公司 | A nucleic acid lipid nanocarrier and its preparation method and application |
| EP4662196A1 (en) * | 2023-02-09 | 2025-12-17 | Seqirus Inc. | Ionizable cationic compound |
| DE112024001143T5 (en) | 2023-03-08 | 2025-12-18 | CureVac SE | NEW LIPID NANOPARTICLE FORMULAS FOR NUCLEAN ACID RELEASE |
| EP4683667A1 (en) | 2023-03-23 | 2026-01-28 | ModernaTX, Inc. | Peg targeting compounds for delivery of therapeutics |
| WO2024197307A1 (en) | 2023-03-23 | 2024-09-26 | Modernatx, Inc. | Peg targeting compounds for delivery of therapeutics |
| WO2024197309A1 (en) | 2023-03-23 | 2024-09-26 | Modernatx, Inc. | Peg targeting compounds for delivery of therapeutics |
| WO2024205657A2 (en) * | 2023-03-29 | 2024-10-03 | Orna Therapeutics, Inc. | Lipids and lipid nanoparticle compositions for delivering polynucleotides |
| WO2024230934A1 (en) | 2023-05-11 | 2024-11-14 | CureVac SE | Therapeutic nucleic acid for the treatment of ophthalmic diseases |
| WO2024259373A1 (en) | 2023-06-14 | 2024-12-19 | Modernatx, Inc. | Compounds and compositions for delivery of therapeutic agents |
| EP4520765A1 (en) | 2023-09-08 | 2025-03-12 | AldexChem Kft. | Ionizable cationic lipids incorporating silicon |
| EP4646422A1 (en) | 2023-09-08 | 2025-11-12 | AldexChem Kft. | Ionizable cationic lipids incorporating silicon: synthesis and lnp formulation |
| US20250162981A1 (en) * | 2023-11-14 | 2025-05-22 | Flagship Pioneering Innovations Vii, Llc | Ionizable lipidoid compositions and therapeutic uses thereof |
| WO2025160127A1 (en) * | 2024-01-23 | 2025-07-31 | Modernatx, Inc. | Ionizable lipids and compositions thereof for delivery of therapeutic agents |
| WO2025166993A1 (en) * | 2024-02-05 | 2025-08-14 | 复旦大学 | Novel lipid compound and composition, and nucleic acid lipid nanoparticle formulation, and screening method therefor and use thereof |
| CN118388360B (en) * | 2024-04-17 | 2025-04-04 | 北京悦康科创医药科技股份有限公司 | Long-acting spleen-targeted cationic lipid compound containing benzene ring structure, composition containing same and use thereof |
| WO2026003582A2 (en) | 2024-06-27 | 2026-01-02 | Axelyf ehf. | Lipids and lipid nanoparticles |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20160317458A1 (en) * | 2013-12-19 | 2016-11-03 | Luis Brito | Lipids and Lipid Compositions for the Delivery of Active Agents |
| CN106795096A (en) * | 2014-06-25 | 2017-05-31 | 爱康泰生治疗公司 | Novel Lipid and Lipid Nanoparticle Formulations for Delivery of Nucleic Acids |
| WO2018170306A1 (en) * | 2017-03-15 | 2018-09-20 | Modernatx, Inc. | Compounds and compositions for intracellular delivery of therapeutic agents |
| WO2018225871A1 (en) * | 2017-06-09 | 2018-12-13 | 協和発酵キリン株式会社 | Compound serving as cationic lipid |
| WO2018232120A1 (en) * | 2017-06-14 | 2018-12-20 | Modernatx, Inc. | Compounds and compositions for intracellular delivery of agents |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB0806086D0 (en) * | 2008-04-04 | 2008-05-14 | Ulive Entpr Ltd | Dendrimer polymer hybrids |
| ES2969082T3 (en) * | 2015-09-17 | 2024-05-16 | Modernatx Inc | Compounds and compositions for intracellular administration of therapeutic agents |
| AU2016324463B2 (en) | 2015-09-17 | 2022-10-27 | Modernatx, Inc. | Polynucleotides containing a stabilizing tail region |
| ES2810701T5 (en) | 2015-10-05 | 2024-07-11 | Modernatx Inc | Procedures for the therapeutic administration of messenger ribonucleic acid drugs |
| FI3368507T3 (en) * | 2015-10-28 | 2023-03-21 | Acuitas Therapeutics Inc | New lipids and lipid nanoparticle preparations for delivery of nucleic acids |
-
2020
- 2020-09-18 EP EP20786367.1A patent/EP4031521B1/en active Active
- 2020-09-18 AU AU2020351220A patent/AU2020351220A1/en not_active Abandoned
- 2020-09-18 CA CA3155015A patent/CA3155015A1/en active Pending
- 2020-09-18 KR KR1020227012366A patent/KR20220101076A/en not_active Withdrawn
- 2020-09-18 MX MX2022003285A patent/MX2022003285A/en unknown
- 2020-09-18 WO PCT/US2020/051613 patent/WO2021055835A1/en not_active Ceased
- 2020-09-18 CN CN202080080295.1A patent/CN114728886B/en active Active
- 2020-09-18 JP JP2022518191A patent/JP7638973B2/en active Active
- 2020-09-18 BR BR112022004936A patent/BR112022004936A2/en not_active Application Discontinuation
- 2020-09-18 US US17/761,984 patent/US20220411361A1/en active Pending
-
2022
- 2022-03-14 IL IL291339A patent/IL291339A/en unknown
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20160317458A1 (en) * | 2013-12-19 | 2016-11-03 | Luis Brito | Lipids and Lipid Compositions for the Delivery of Active Agents |
| CN106795096A (en) * | 2014-06-25 | 2017-05-31 | 爱康泰生治疗公司 | Novel Lipid and Lipid Nanoparticle Formulations for Delivery of Nucleic Acids |
| WO2018170306A1 (en) * | 2017-03-15 | 2018-09-20 | Modernatx, Inc. | Compounds and compositions for intracellular delivery of therapeutic agents |
| WO2018225871A1 (en) * | 2017-06-09 | 2018-12-13 | 協和発酵キリン株式会社 | Compound serving as cationic lipid |
| WO2018232120A1 (en) * | 2017-06-14 | 2018-12-20 | Modernatx, Inc. | Compounds and compositions for intracellular delivery of agents |
Non-Patent Citations (2)
| Title |
|---|
| KIMBERLY J. HASSETT等: "Optimization of Lipid Nanoparticles for Intramuscular Administration of mRNA Vaccines", 《MOLECULAR THERAPY: NUCLEIC ACIDS》 * |
| STACI SABNIS等: "A Novel Amino Lipid Series for mRNA Delivery: Improved Endosomal Escape and Sustained Pharmacology and Safety in Non-human Primates", 《MOLECULAR THERAPY》 * |
Cited By (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2024022263A1 (en) * | 2022-07-25 | 2024-02-01 | 苏州艾博生物科技有限公司 | Lipid compound and lipid nanoparticle composition |
| WO2024067639A1 (en) * | 2022-09-30 | 2024-04-04 | 荣灿生物医药技术(上海)有限公司 | Ionizable lipid compound having high transfection efficiency and use thereof |
| CN116947669A (en) * | 2022-09-30 | 2023-10-27 | 荣灿生物医药技术(上海)有限公司 | Ionizable lipid compound with high transfection efficiency and application thereof |
| CN116947669B (en) * | 2022-09-30 | 2024-04-12 | 荣灿生物医药技术(上海)有限公司 | Ionizable lipid compound with high transfection efficiency and application thereof |
| WO2024140835A1 (en) * | 2022-12-28 | 2024-07-04 | 仁景(苏州)生物科技有限公司 | Lipid compound and lipid nanoparticle composition |
| CN117777089A (en) * | 2022-12-28 | 2024-03-29 | 仁景(苏州)生物科技有限公司 | Lipid compounds and lipid nanoparticle compositions |
| US12391662B2 (en) | 2022-12-28 | 2025-08-19 | Rinuagene Biotechnology Co. Ltd. | Lipid compounds and lipid nanoparticle compositions |
| WO2024145964A1 (en) * | 2023-01-05 | 2024-07-11 | 北京悦康科创医药科技股份有限公司 | Cationic lipid compound with high efficiency, low toxicity and stable expression, and composition comprising same |
| JP7764663B1 (en) | 2023-01-05 | 2025-11-05 | ハンチョウ ティアンロン ファーマシューティカル カンパニー、リミテッド | Highly efficient, low-toxicity, stably expressed cationic lipid compounds and compositions thereof |
| JP2025537047A (en) * | 2023-01-05 | 2025-11-13 | ハンチョウ ティアンロン ファーマシューティカル カンパニー、リミテッド | Highly efficient, low-toxicity, stably expressed cationic lipid compounds and compositions thereof |
| JP7764663B6 (en) | 2023-01-05 | 2025-11-27 | ハンチョウ ティアンロン ファーマシューティカル カンパニー、リミテッド | Highly efficient, low-toxicity, stably expressed cationic lipid compounds and compositions thereof |
| CN115784920A (en) * | 2023-02-09 | 2023-03-14 | 荣灿生物医药技术(上海)有限公司 | Ionizable lipid compound with high transfection efficiency and application thereof |
| WO2025146125A1 (en) * | 2024-01-05 | 2025-07-10 | 仁景(苏州)生物科技有限公司 | Lipid compound and lipid nanoparticle for delivery |
| WO2025161943A1 (en) * | 2024-02-02 | 2025-08-07 | 仁景(苏州)生物科技有限公司 | Lipid compound and lipid nanoparticle for delivery |
Also Published As
| Publication number | Publication date |
|---|---|
| BR112022004936A2 (en) | 2022-06-14 |
| US20220411361A1 (en) | 2022-12-29 |
| AU2020351220A1 (en) | 2022-04-07 |
| EP4031521B1 (en) | 2025-12-24 |
| KR20220101076A (en) | 2022-07-19 |
| IL291339A (en) | 2022-05-01 |
| WO2021055835A1 (en) | 2021-03-25 |
| JP2022549253A (en) | 2022-11-24 |
| CA3155015A1 (en) | 2021-03-25 |
| CN114728886B (en) | 2024-04-12 |
| JP7638973B2 (en) | 2025-03-04 |
| MX2022003285A (en) | 2022-06-14 |
| EP4031521A1 (en) | 2022-07-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN114728886B (en) | Lipid compounds and compositions containing carbonates for intracellular delivery of therapeutic agents | |
| JP7638972B2 (en) | Branched tail lipid compounds and compositions for intracellular delivery of therapeutic agents - Patents.com | |
| JP7642620B2 (en) | Headgroup lipid compounds and compositions for intracellular delivery of therapeutic agents - Patents.com | |
| CN118369308A (en) | Compounds and compositions for delivery of therapeutic agents | |
| JP2024512576A (en) | Branched tail lipid compounds and compositions for intracellular delivery of therapeutic agents | |
| TWI757248B (en) | Compounds and compositions for intracellular delivery of therapeutic agents | |
| CN116133652A (en) | Method for preparing lipid nanoparticles | |
| CN110520409A (en) | Compound and composition for Intracellular delivery therapeutic agent | |
| JP2022501360A (en) | Compounds and compositions for intracellular delivery of therapeutic agents | |
| JP2019508371A (en) | Compounds and compositions for intracellular delivery of drugs | |
| WO2024109798A1 (en) | Lipid compound and lipid nanoparticle composition | |
| HK40104744B (en) | Lipid nanoparticle compositions | |
| HK40104744A (en) | Lipid nanoparticle compositions | |
| CN120230759A (en) | Nanoliposome particles expressing botulinum toxin and their applications |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |