CN114716454A - Nitrogen and oxa condensed ring aromatic hydrocarbon and synthetic method thereof - Google Patents
Nitrogen and oxa condensed ring aromatic hydrocarbon and synthetic method thereof Download PDFInfo
- Publication number
- CN114716454A CN114716454A CN202210557098.5A CN202210557098A CN114716454A CN 114716454 A CN114716454 A CN 114716454A CN 202210557098 A CN202210557098 A CN 202210557098A CN 114716454 A CN114716454 A CN 114716454A
- Authority
- CN
- China
- Prior art keywords
- compound
- ring aromatic
- nitrogen
- condensed ring
- silver
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 title claims abstract description 56
- 229910052757 nitrogen Inorganic materials 0.000 title claims abstract description 28
- 150000004945 aromatic hydrocarbons Chemical class 0.000 title claims abstract description 26
- 238000010189 synthetic method Methods 0.000 title description 4
- 238000006243 chemical reaction Methods 0.000 claims abstract description 53
- 239000002904 solvent Substances 0.000 claims abstract description 39
- 150000008049 diazo compounds Chemical class 0.000 claims abstract description 21
- 238000000034 method Methods 0.000 claims abstract description 20
- 239000003054 catalyst Substances 0.000 claims abstract description 19
- 238000003786 synthesis reaction Methods 0.000 claims abstract description 14
- 230000015572 biosynthetic process Effects 0.000 claims abstract description 12
- 239000000654 additive Substances 0.000 claims abstract description 7
- 230000000996 additive effect Effects 0.000 claims abstract description 7
- 239000012298 atmosphere Substances 0.000 claims abstract description 7
- 229940125904 compound 1 Drugs 0.000 claims abstract description 6
- BCHZICNRHXRCHY-UHFFFAOYSA-N 2h-oxazine Chemical compound N1OC=CC=C1 BCHZICNRHXRCHY-UHFFFAOYSA-N 0.000 claims abstract description 5
- 229940125782 compound 2 Drugs 0.000 claims abstract description 5
- 229940126214 compound 3 Drugs 0.000 claims abstract description 4
- 150000001491 aromatic compounds Chemical class 0.000 claims abstract 2
- SCYULBFZEHDVBN-UHFFFAOYSA-N 1,1-Dichloroethane Chemical compound CC(Cl)Cl SCYULBFZEHDVBN-UHFFFAOYSA-N 0.000 claims description 18
- -1 hexafluoroantimonate Chemical compound 0.000 claims description 14
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 claims description 9
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 claims description 9
- 239000010948 rhodium Substances 0.000 claims description 9
- 239000001301 oxygen Substances 0.000 claims description 8
- 229910052760 oxygen Inorganic materials 0.000 claims description 8
- 229910052703 rhodium Inorganic materials 0.000 claims description 8
- MHOVAHRLVXNVSD-UHFFFAOYSA-N rhodium atom Chemical compound [Rh] MHOVAHRLVXNVSD-UHFFFAOYSA-N 0.000 claims description 8
- 230000002194 synthesizing effect Effects 0.000 claims description 8
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 claims description 7
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 claims description 6
- QVLTVILSYOWFRM-UHFFFAOYSA-L CC1=C(C)C(C)([Rh](Cl)Cl)C(C)=C1C Chemical class CC1=C(C)C(C)([Rh](Cl)Cl)C(C)=C1C QVLTVILSYOWFRM-UHFFFAOYSA-L 0.000 claims description 6
- GGCZERPQGJTIQP-UHFFFAOYSA-N sodium;9,10-dioxoanthracene-2-sulfonic acid Chemical compound [Na+].C1=CC=C2C(=O)C3=CC(S(=O)(=O)O)=CC=C3C(=O)C2=C1 GGCZERPQGJTIQP-UHFFFAOYSA-N 0.000 claims description 6
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 claims description 4
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 claims description 4
- SCVFZCLFOSHCOH-UHFFFAOYSA-M potassium acetate Chemical compound [K+].CC([O-])=O SCVFZCLFOSHCOH-UHFFFAOYSA-M 0.000 claims description 4
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Substances [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 claims description 4
- FFFIRKXTFQCCKJ-UHFFFAOYSA-N 2,4,6-trimethylbenzoic acid Chemical compound CC1=CC(C)=C(C(O)=O)C(C)=C1 FFFIRKXTFQCCKJ-UHFFFAOYSA-N 0.000 claims description 2
- ZOIORXHNWRGPMV-UHFFFAOYSA-N acetic acid;zinc Chemical compound [Zn].CC(O)=O.CC(O)=O ZOIORXHNWRGPMV-UHFFFAOYSA-N 0.000 claims description 2
- CZKMPDNXOGQMFW-UHFFFAOYSA-N chloro(triethyl)germane Chemical compound CC[Ge](Cl)(CC)CC CZKMPDNXOGQMFW-UHFFFAOYSA-N 0.000 claims description 2
- 239000012299 nitrogen atmosphere Substances 0.000 claims description 2
- IUGYQRQAERSCNH-UHFFFAOYSA-N pivalic acid Chemical compound CC(C)(C)C(O)=O IUGYQRQAERSCNH-UHFFFAOYSA-N 0.000 claims description 2
- 235000011056 potassium acetate Nutrition 0.000 claims description 2
- 229910000027 potassium carbonate Inorganic materials 0.000 claims description 2
- 230000008569 process Effects 0.000 claims description 2
- 230000035484 reaction time Effects 0.000 claims description 2
- CQLFBEKRDQMJLZ-UHFFFAOYSA-M silver acetate Chemical compound [Ag+].CC([O-])=O CQLFBEKRDQMJLZ-UHFFFAOYSA-M 0.000 claims description 2
- 229940071536 silver acetate Drugs 0.000 claims description 2
- 229910000367 silver sulfate Inorganic materials 0.000 claims description 2
- YPNVIBVEFVRZPJ-UHFFFAOYSA-L silver sulfate Chemical compound [Ag+].[Ag+].[O-]S([O-])(=O)=O YPNVIBVEFVRZPJ-UHFFFAOYSA-L 0.000 claims description 2
- 229910001494 silver tetrafluoroborate Inorganic materials 0.000 claims description 2
- KZJPVUDYAMEDRM-UHFFFAOYSA-M silver;2,2,2-trifluoroacetate Chemical compound [Ag+].[O-]C(=O)C(F)(F)F KZJPVUDYAMEDRM-UHFFFAOYSA-M 0.000 claims description 2
- 229910000029 sodium carbonate Inorganic materials 0.000 claims description 2
- 239000004246 zinc acetate Substances 0.000 claims description 2
- 150000001555 benzenes Chemical group 0.000 claims 2
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 claims 2
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 claims 1
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 claims 1
- GFCVQYCZHGSRMA-UHFFFAOYSA-L C(C)(=O)[O-].CC1=C(C(=C(C1([Rh+2])C)C)C)C.C(C)(=O)[O-] Chemical compound C(C)(=O)[O-].CC1=C(C(=C(C1([Rh+2])C)C)C)C.C(C)(=O)[O-] GFCVQYCZHGSRMA-UHFFFAOYSA-L 0.000 claims 1
- SAXQOYZKDFVDTH-UHFFFAOYSA-N CC1=C(C(=C(C1(C)[Rh])C)C)C Chemical compound CC1=C(C(=C(C1(C)[Rh])C)C)C SAXQOYZKDFVDTH-UHFFFAOYSA-N 0.000 claims 1
- OLBVUFHMDRJKTK-UHFFFAOYSA-N [N].[O] Chemical compound [N].[O] OLBVUFHMDRJKTK-UHFFFAOYSA-N 0.000 claims 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims 1
- 235000019253 formic acid Nutrition 0.000 claims 1
- 235000011181 potassium carbonates Nutrition 0.000 claims 1
- HSYLTRBDKXZSGS-UHFFFAOYSA-N silver;bis(trifluoromethylsulfonyl)azanide Chemical compound [Ag+].FC(F)(F)S(=O)(=O)[N-]S(=O)(=O)C(F)(F)F HSYLTRBDKXZSGS-UHFFFAOYSA-N 0.000 claims 1
- 235000017550 sodium carbonate Nutrition 0.000 claims 1
- 235000013904 zinc acetate Nutrition 0.000 claims 1
- 239000000463 material Substances 0.000 abstract description 11
- 238000001308 synthesis method Methods 0.000 abstract description 8
- 239000003814 drug Substances 0.000 abstract description 4
- 125000005842 heteroatom Chemical group 0.000 abstract description 4
- 125000005575 polycyclic aromatic hydrocarbon group Chemical group 0.000 abstract description 4
- 239000000758 substrate Substances 0.000 abstract description 4
- 125000004429 atom Chemical group 0.000 abstract description 3
- 125000000524 functional group Chemical group 0.000 abstract description 2
- 229930014626 natural product Natural products 0.000 abstract description 2
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 45
- 238000005481 NMR spectroscopy Methods 0.000 description 41
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 36
- 150000001875 compounds Chemical class 0.000 description 33
- 238000001228 spectrum Methods 0.000 description 29
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 25
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 15
- 238000012512 characterization method Methods 0.000 description 15
- 239000003480 eluent Substances 0.000 description 15
- 239000003208 petroleum Substances 0.000 description 15
- 238000000926 separation method Methods 0.000 description 15
- 239000000741 silica gel Substances 0.000 description 15
- 229910002027 silica gel Inorganic materials 0.000 description 15
- 239000000543 intermediate Substances 0.000 description 14
- 238000000655 nuclear magnetic resonance spectrum Methods 0.000 description 13
- 229930195733 hydrocarbon Natural products 0.000 description 10
- 238000002844 melting Methods 0.000 description 10
- 230000008018 melting Effects 0.000 description 10
- 239000007787 solid Substances 0.000 description 10
- 230000005693 optoelectronics Effects 0.000 description 9
- 230000008878 coupling Effects 0.000 description 5
- 238000010168 coupling process Methods 0.000 description 5
- 238000005859 coupling reaction Methods 0.000 description 5
- 239000007788 liquid Substances 0.000 description 5
- GVOISEJVFFIGQE-YCZSINBZSA-N n-[(1r,2s,5r)-5-[methyl(propan-2-yl)amino]-2-[(3s)-2-oxo-3-[[6-(trifluoromethyl)quinazolin-4-yl]amino]pyrrolidin-1-yl]cyclohexyl]acetamide Chemical compound CC(=O)N[C@@H]1C[C@H](N(C)C(C)C)CC[C@@H]1N1C(=O)[C@@H](NC=2C3=CC(=CC=C3N=CN=2)C(F)(F)F)CC1 GVOISEJVFFIGQE-YCZSINBZSA-N 0.000 description 5
- 239000004215 Carbon black (E152) Substances 0.000 description 4
- AWJUIBRHMBBTKR-UHFFFAOYSA-N iso-quinoline Natural products C1=NC=CC2=CC=CC=C21 AWJUIBRHMBBTKR-UHFFFAOYSA-N 0.000 description 4
- 125000006615 aromatic heterocyclic group Chemical group 0.000 description 3
- SRCZQMGIVIYBBJ-UHFFFAOYSA-N ethoxyethane;ethyl acetate Chemical compound CCOCC.CCOC(C)=O SRCZQMGIVIYBBJ-UHFFFAOYSA-N 0.000 description 3
- 229910052751 metal Inorganic materials 0.000 description 3
- 239000002184 metal Substances 0.000 description 3
- JUJWROOIHBZHMG-UHFFFAOYSA-N pyridine Substances C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 3
- 238000011160 research Methods 0.000 description 3
- 239000011734 sodium Substances 0.000 description 3
- SNQSGKQEGKHJCZ-UHFFFAOYSA-N 1,3-dimethylisoquinoline Chemical compound C1=CC=C2C(C)=NC(C)=CC2=C1 SNQSGKQEGKHJCZ-UHFFFAOYSA-N 0.000 description 2
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 2
- 238000010499 C–H functionalization reaction Methods 0.000 description 2
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 2
- 230000009471 action Effects 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 150000001989 diazonium salts Chemical class 0.000 description 2
- 230000005669 field effect Effects 0.000 description 2
- QWXYZCJEXYQNEI-OSZHWHEXSA-N intermediate I Chemical compound COC(=O)[C@@]1(C=O)[C@H]2CC=[N+](C\C2=C\C)CCc2c1[nH]c1ccccc21 QWXYZCJEXYQNEI-OSZHWHEXSA-N 0.000 description 2
- HZVOZRGWRWCICA-UHFFFAOYSA-N methanediyl Chemical compound [CH2] HZVOZRGWRWCICA-UHFFFAOYSA-N 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 150000004893 oxazines Chemical class 0.000 description 2
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 2
- 238000000425 proton nuclear magnetic resonance spectrum Methods 0.000 description 2
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 2
- 229910052709 silver Inorganic materials 0.000 description 2
- 239000004332 silver Substances 0.000 description 2
- 230000009466 transformation Effects 0.000 description 2
- 238000000844 transformation Methods 0.000 description 2
- 230000007704 transition Effects 0.000 description 2
- HBENZIXOGRCSQN-VQWWACLZSA-N (1S,2S,6R,14R,15R,16R)-5-(cyclopropylmethyl)-16-[(2S)-2-hydroxy-3,3-dimethylpentan-2-yl]-15-methoxy-13-oxa-5-azahexacyclo[13.2.2.12,8.01,6.02,14.012,20]icosa-8(20),9,11-trien-11-ol Chemical compound N1([C@@H]2CC=3C4=C(C(=CC=3)O)O[C@H]3[C@@]5(OC)CC[C@@]2([C@@]43CC1)C[C@@H]5[C@](C)(O)C(C)(C)CC)CC1CC1 HBENZIXOGRCSQN-VQWWACLZSA-N 0.000 description 1
- PHDIJLFSKNMCMI-ITGJKDDRSA-N (3R,4S,5R,6R)-6-(hydroxymethyl)-4-(8-quinolin-6-yloxyoctoxy)oxane-2,3,5-triol Chemical compound OC[C@@H]1[C@H]([C@@H]([C@H](C(O1)O)O)OCCCCCCCCOC=1C=C2C=CC=NC2=CC=1)O PHDIJLFSKNMCMI-ITGJKDDRSA-N 0.000 description 1
- JNPGUXGVLNJQSQ-BGGMYYEUSA-M (e,3r,5s)-7-[4-(4-fluorophenyl)-1,2-di(propan-2-yl)pyrrol-3-yl]-3,5-dihydroxyhept-6-enoate Chemical compound CC(C)N1C(C(C)C)=C(\C=C\[C@@H](O)C[C@@H](O)CC([O-])=O)C(C=2C=CC(F)=CC=2)=C1 JNPGUXGVLNJQSQ-BGGMYYEUSA-M 0.000 description 1
- VAVHMEQFYYBAPR-ITWZMISCSA-N (e,3r,5s)-7-[4-(4-fluorophenyl)-1-phenyl-2-propan-2-ylpyrrol-3-yl]-3,5-dihydroxyhept-6-enoic acid Chemical compound CC(C)C1=C(\C=C\[C@@H](O)C[C@@H](O)CC(O)=O)C(C=2C=CC(F)=CC=2)=CN1C1=CC=CC=C1 VAVHMEQFYYBAPR-ITWZMISCSA-N 0.000 description 1
- FZTLLUYFWAOGGB-UHFFFAOYSA-N 1,4-dioxane dioxane Chemical compound C1COCCO1.C1COCCO1 FZTLLUYFWAOGGB-UHFFFAOYSA-N 0.000 description 1
- RAIKREKWRHXJRW-UHFFFAOYSA-N 2H-naphtho[1,2-h]quinolin-1-one Chemical class C1(CC=NC=2C3=C(C=CC1=2)C1=CC=CC=C1C=C3)=O RAIKREKWRHXJRW-UHFFFAOYSA-N 0.000 description 1
- HIHOEGPXVVKJPP-JTQLQIEISA-N 5-fluoro-2-[[(1s)-1-(5-fluoropyridin-2-yl)ethyl]amino]-6-[(5-methyl-1h-pyrazol-3-yl)amino]pyridine-3-carbonitrile Chemical compound N([C@@H](C)C=1N=CC(F)=CC=1)C(C(=CC=1F)C#N)=NC=1NC=1C=C(C)NN=1 HIHOEGPXVVKJPP-JTQLQIEISA-N 0.000 description 1
- DGJMHKMYSDYOFP-MRXNPFEDSA-N C=CC(N(CCC1)C[C@@H]1N1N=C(C2=CN(CC(C3=CC=CC=C3)(F)F)N=N2)C2=C(N)N=CN=C12)=O Chemical compound C=CC(N(CCC1)C[C@@H]1N1N=C(C2=CN(CC(C3=CC=CC=C3)(F)F)N=N2)C2=C(N)N=CN=C12)=O DGJMHKMYSDYOFP-MRXNPFEDSA-N 0.000 description 1
- HGDWHTASNMRJMP-UHFFFAOYSA-N [1-(hydroxyamino)-1-oxo-5-(3-phenoxyphenyl)pentan-2-yl]phosphonic acid Chemical compound ONC(=O)C(P(O)(O)=O)CCCC1=CC=CC(OC=2C=CC=CC=2)=C1 HGDWHTASNMRJMP-UHFFFAOYSA-N 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- JIMXXGFJRDUSRO-UHFFFAOYSA-N adamantane-1-carboxylic acid Chemical compound C1C(C2)CC3CC2CC1(C(=O)O)C3 JIMXXGFJRDUSRO-UHFFFAOYSA-N 0.000 description 1
- 229910052786 argon Inorganic materials 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 125000004432 carbon atom Chemical group C* 0.000 description 1
- 238000001460 carbon-13 nuclear magnetic resonance spectrum Methods 0.000 description 1
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 125000004122 cyclic group Chemical group 0.000 description 1
- 230000018044 dehydration Effects 0.000 description 1
- 238000006297 dehydration reaction Methods 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- 125000004177 diethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 239000000975 dye Substances 0.000 description 1
- 238000003379 elimination reaction Methods 0.000 description 1
- 125000002587 enol group Chemical group 0.000 description 1
- 238000007306 functionalization reaction Methods 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 239000001307 helium Substances 0.000 description 1
- 229910052734 helium Inorganic materials 0.000 description 1
- SWQJXJOGLNCZEY-UHFFFAOYSA-N helium atom Chemical compound [He] SWQJXJOGLNCZEY-UHFFFAOYSA-N 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 150000002466 imines Chemical class 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 238000003780 insertion Methods 0.000 description 1
- 230000037431 insertion Effects 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 230000005012 migration Effects 0.000 description 1
- 238000013508 migration Methods 0.000 description 1
- 229910052754 neon Inorganic materials 0.000 description 1
- GKAOGPIIYCISHV-UHFFFAOYSA-N neon atom Chemical compound [Ne] GKAOGPIIYCISHV-UHFFFAOYSA-N 0.000 description 1
- 230000000269 nucleophilic effect Effects 0.000 description 1
- 125000003367 polycyclic group Chemical group 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 239000000376 reactant Substances 0.000 description 1
- 239000004065 semiconductor Substances 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D498/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D498/02—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and oxygen atoms as the only ring hetero atoms in which the condensed system contains two hetero rings
- C07D498/06—Peri-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N21/00—Investigating or analysing materials by the use of optical means, i.e. using sub-millimetre waves, infrared, visible or ultraviolet light
- G01N21/62—Systems in which the material investigated is excited whereby it emits light or causes a change in wavelength of the incident light
- G01N21/63—Systems in which the material investigated is excited whereby it emits light or causes a change in wavelength of the incident light optically excited
- G01N21/64—Fluorescence; Phosphorescence
- G01N21/6428—Measuring fluorescence of fluorescent products of reactions or of fluorochrome labelled reactive substances, e.g. measuring quenching effects, using measuring "optrodes"
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1029—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom
- C09K2211/1033—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom with oxygen
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E10/00—Energy generation through renewable energy sources
- Y02E10/50—Photovoltaic [PV] energy
- Y02E10/549—Organic PV cells
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P20/00—Technologies relating to chemical industry
- Y02P20/50—Improvements relating to the production of bulk chemicals
- Y02P20/52—Improvements relating to the production of bulk chemicals using catalysts, e.g. selective catalysts
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Physics & Mathematics (AREA)
- Immunology (AREA)
- Analytical Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Life Sciences & Earth Sciences (AREA)
- Optics & Photonics (AREA)
- Biochemistry (AREA)
- General Health & Medical Sciences (AREA)
- General Physics & Mathematics (AREA)
- Pathology (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Nitrogen And Oxygen As The Only Ring Hetero Atoms (AREA)
Abstract
Description
技术领域technical field
本发明属于有机合成化学的技术领域,尤其涉及一种氮、氧杂稠环芳烃及其合成方法。The invention belongs to the technical field of organic synthetic chemistry, and in particular relates to a nitrogen, oxygen heterocyclic aromatic hydrocarbon and a synthesis method thereof.
背景技术Background technique
稠环芳烃是指由两个或多个芳烃组成的多环芳烃化合物,广泛应用于药物、染料等行业。稠环芳烃的晶体排列存在π-π相互作用,分子之间形成有序堆积,其在有机光电器件(包括有机场效应晶体管(Organic Field-Effect Transistor,OFET)、有机太阳能电池(Organic Solar Cell,OSC)和有机发光二极管(Organic Light-Emitting Diode,OLED)等)、有机半导体材料等领域都具有广阔的应用前景(Chem.Rev.,2007,107,926;Chem.Soc.Rev.,2010,39,1489;Chem.Soc.Rev.,2012,41,4245;Nature,2004,428,911)。与传统的无机光电材料相比,大尺寸的共轭分子具有分子级别的结构调整;溶解性好、易加工;质轻、柔性,可实现大面积器件制备等优势。因此,设计并合成新型有机稠环芳烃,并对其进行性质以及应用研究,一直以来都是化学、材料和生物等领域科学家的研究热点之一。特别地,以杂原子替代多环化合物中的部分碳原子,形成含杂原子共轭骨架分子,即杂稠环芳烃,可显著提高材料的稳定性及组装性能,从而显著改善有机光电材料的光电性能和多功能性(Angew.Chem.,Int.Ed.,2010,49,8209;Chem.Mater.,2015,28,3)。同时,对稠环芳烃进行多功能修饰,也是赋予有机光电分子可调控性和多样性的重要手段。Polycyclic aromatic hydrocarbons refer to polycyclic aromatic hydrocarbon compounds composed of two or more aromatic hydrocarbons, which are widely used in pharmaceuticals, dyes and other industries. There are π-π interactions in the crystal arrangement of fused ring aromatic hydrocarbons, and an ordered stacking is formed between the molecules, which is used in organic optoelectronic devices (including organic field-effect transistors (Organic Field-Effect Transistor, OFET), organic solar cells (Organic Solar Cell, OSC) and organic light-emitting diodes (Organic Light-Emitting Diode, OLED, etc.), organic semiconductor materials and other fields have broad application prospects (Chem. Rev., 2007, 107, 926; Chem. Soc. Rev., 2010, 39, 1489; Chem. Soc. Rev., 2012, 41, 4245; Nature, 2004, 428, 911). Compared with traditional inorganic optoelectronic materials, large-sized conjugated molecules have the advantages of molecular-level structure adjustment; good solubility and easy processing; light weight and flexibility, which can realize the preparation of large-area devices. Therefore, designing and synthesizing new organic fused-ring aromatic hydrocarbons, and conducting research on their properties and applications has always been one of the research hotspots of scientists in the fields of chemistry, materials and biology. In particular, replacing some carbon atoms in polycyclic compounds with heteroatoms to form heteroatom-containing conjugated skeleton molecules, namely hetero-fused aromatic hydrocarbons, can significantly improve the stability and assembly performance of the materials, thereby significantly improving the optoelectronic properties of organic optoelectronic materials. Performance and versatility (Angew. Chem., Int. Ed., 2010, 49, 8209; Chem. Mater., 2015, 28, 3). At the same time, multifunctional modification of fused-ring aromatic hydrocarbons is also an important means to endow organic optoelectronic molecules with controllability and diversity.
惰性C-H键直接官能团化反应由于高原子经济与步骤经济的优越性,现已无可争议的成为化学家们追求的理想合成手段,其一度被人们称为“化学的圣杯”。过去20年里,过渡金属催化C-H活化反应的确已经取得了非常显著的成效,实现了诸多传统化学所不能完成的转化。其中,铑催化导向C-H键活化1:2偶联构筑多环芳烃的合成得到了快速发展,制备了一大批在有机光电材料和药物领域具有重要应用前景的新型稠环芳烃骨架。而重氮化合物作为有机合成中的重要中间体,将其应用在过渡金属催化1:2偶联构建稠杂环芳烃的研究尚处于初级阶段。迄今为止,有关铑催化导向C-H活化1:2偶联构筑稠环芳烃分子仅有一例报道(Org.Lett.2017,19,2294),研究相对缓慢。上述方法以强配位的吡啶类化合物作为导向基,在铑催化剂的作用下实现了和重氮化合物的1:2偶联,构筑了一系列萘并喹啉酮骨架。Due to the advantages of high atomic economy and step economy, the direct functionalization of inert C-H bonds has become an undisputed ideal synthetic method pursued by chemists, and it was once called "the holy grail of chemistry". In the past 20 years, transition metal-catalyzed C-H activation reactions have indeed achieved remarkable results, realizing many transformations that cannot be accomplished by traditional chemistry. Among them, the synthesis of rhodium-catalyzed C-H bond activated 1:2 coupling to construct polycyclic aromatic hydrocarbons has been rapidly developed, and a large number of novel fused-ring aromatic hydrocarbon frameworks with important application prospects in the field of organic optoelectronic materials and pharmaceuticals have been prepared. However, as important intermediates in organic synthesis, the application of diazonium compounds in transition metal-catalyzed 1:2 coupling to construct fused heterocyclic aromatic hydrocarbons is still in its infancy. So far, there is only one report on rhodium-catalyzed C-H-directed C-H activation for 1:2 coupling to construct condensed aromatic hydrocarbon molecules (Org. Lett. 2017, 19, 2294), and the research is relatively slow. In the above method, the strongly coordinated pyridine compound is used as the guiding group, and the 1:2 coupling with the diazo compound is realized under the action of rhodium catalyst, and a series of naphthoquinolinone skeletons are constructed.
一方面,现有的合成手段所使用导向基团为强配位能力的吡啶,底物合成复杂,产物类型单一,所得骨架仅为氮杂的稠环芳烃。另一方面,苯并噁嗪型亚胺作为有机合成当中的关键中间体,合成简单、来源广泛。将其作为弱的导向基团应用在铑催化的卡宾1:2偶联的研究尚没有报道。根据已有文献报道,杂稠环芳烃,可显著提高材料的稳定性及组装性能,从而显著改善有机光电材料的光电性能和多功能性(Angew.Chem.,Int.Ed.,2010,49,8209;Chem.Mater.,2015,28,3),杂稠环芳烃在有机光电材料方面的应用具有广阔的前景;因此急需寻找一种合成杂稠环芳烃的新方法。On the one hand, the directing group used in the existing synthesis methods is pyridine with strong coordination ability, the substrate synthesis is complex, the product type is single, and the obtained skeleton is only a condensed aromatic hydrocarbon with aza. On the other hand, benzoxazine imines, as key intermediates in organic synthesis, have simple synthesis and wide sources. The use of it as a weak directing group in the rhodium-catalyzed 1:2 coupling of carbene has not been reported. According to existing literature reports, hetero-fused aromatic hydrocarbons can significantly improve the stability and assembly performance of materials, thereby significantly improving the optoelectronic properties and multifunctionality of organic optoelectronic materials (Angew.Chem., Int.Ed., 2010,49, 8209; Chem.Mater., 2015, 28, 3), the application of hetero-fused aromatic hydrocarbons in organic optoelectronic materials has broad prospects; therefore, it is urgent to find a new method for the synthesis of hetero-fused aromatic hydrocarbons.
发明内容SUMMARY OF THE INVENTION
针对现有稠杂环芳烃合成方法单一、底物合成复杂以及产物结构局限等技术问题,本发明提出一种氮、氧杂稠环芳烃及其合成方法,本发明为含多杂原子稠环芳烃的合成提供了一种简单有效的合成方法,并且该方法具有反应条件温和、操作简单、原子经济、步骤经济、官能团耐受性强以及收率良好等特点。In view of the technical problems such as single synthesis method of existing fused heterocyclic aromatic hydrocarbons, complex synthesis of substrates and limited product structure, the present invention provides a nitrogen, oxygen heterocyclic aromatic hydrocarbon and a synthesis method thereof. The present invention is a fused ring aromatic hydrocarbon containing multiple heteroatoms The synthesis provides a simple and effective synthetic method, and the method has the characteristics of mild reaction conditions, simple operation, atom economy, step economy, strong functional group tolerance and good yield.
为了达到上述目的,本发明的技术方案是这样实现的:In order to achieve the above object, the technical scheme of the present invention is achieved in this way:
一种氮、氧杂稠环芳烃,结构式如下所示:A nitrogen, oxygen heterocyclic aromatic hydrocarbon, the structural formula is as follows:
其中R为H、Me、OMe、F、Cl或Br其中任意一种;R1为Me、n-Pr或Ph其中任意一种;R2为n-Am或iPr;Ar为Me、OMe、F、Cl,Br、Ph或CF3任意一种取代的苯环。Wherein R is any one of H, Me, OMe, F, Cl or Br; R 1 is any one of Me, n-Pr or Ph; R 2 is n-Am or i Pr; Ar is Me, OMe, A benzene ring substituted by any one of F, Cl, Br, Ph or CF 3 .
上述氮、氧杂稠环芳烃的合成方法,包括以下步骤:惰性氛围下将噁嗪类化合物1、重氮化合物2、催化剂、添加剂加入到溶剂当中并进行反应,反应结束后得到杂稠环芳烃化合物3,反应方程式为:The synthetic method of above-mentioned nitrogen and oxygen heterocyclic aromatic hydrocarbons comprises the following steps: adding an
其中R为H、Me、OMe、F、Cl或Br其中任意一种;R1为Me、n-Pr或Ph其中任意一种;R2为n-Am或iPr;Ar为Me、OMe、F、Cl,Br、Ph或CF3任意一种取代的苯环;式中Rh(Ⅲ)为催化剂。Wherein R is any one of H, Me, OMe, F, Cl or Br; R 1 is any one of Me, n-Pr or Ph; R 2 is n-Am or i Pr; Ar is Me, OMe, A benzene ring substituted by any one of F, Cl, Br, Ph or CF 3 ; where Rh(III) is a catalyst.
所述反应结束后对反应结束的溶液进行分离提纯得到杂稠环芳烃化合物3。After the reaction is completed, the solution after the reaction is separated and purified to obtain the hetero-fused ring
所述反应的过程中反应温度为80-120℃,反应时间为到6-12h。During the reaction, the reaction temperature is 80-120° C., and the reaction time is 6-12 h.
所述催化剂包括铑催化剂和银盐;铑催化剂和银盐的摩尔比为1:4;所述铑催化剂为二氯(五甲基环戊二烯基)合铑二聚体、五甲基环戊二烯基醋酸铑或二(六氟锑酸)三乙腈(五甲基环戊二烯基)铑其中任意一种或组合;银盐为六氟锑酸银、四氟硼酸银、双三氟甲烷磺酰亚胺银、三氟甲烷磺酸银、硫酸银、醋酸银、三氟醋酸银其中任意一种或组合。The catalyst comprises a rhodium catalyst and a silver salt; the molar ratio of the rhodium catalyst and the silver salt is 1:4; the rhodium catalyst is a dichloro(pentamethylcyclopentadienyl) rhodium dimer, a pentamethyl ring Pentadienyl rhodium acetate or bis(hexafluoroantimonate) triacetonitrile (pentamethylcyclopentadienyl) rhodium any one or combination; silver salt is silver hexafluoroantimonate, silver tetrafluoroborate, bistri Any one or combination of silver fluoromethanesulfonimide, silver trifluoromethanesulfonate, silver sulfate, silver acetate, and silver trifluoroacetate.
所述催化剂由二氯(五甲基环戊二烯基)合铑二聚体和六氟锑酸银组成;二氯(五甲基环戊二烯基)合铑二聚体与六氟锑酸银的摩尔比为1:4。The catalyst is composed of dichloro(pentamethylcyclopentadienyl) rhodium dimer and silver hexafluoroantimonate; dichloro(pentamethylcyclopentadienyl) rhodium dimer and hexafluoroantimony The molar ratio of silver acid is 1:4.
所述添加剂为2,4,6-三甲基苯甲酸、特戊酸、醋酸、1-金刚烷甲酸、醋酸锌、碳酸钠、醋酸钾或碳酸钾其中任意一种或组合。The additive is any one or a combination of 2,4,6-trimethylbenzoic acid, pivalic acid, acetic acid, 1-adamantanecarboxylic acid, zinc acetate, sodium carbonate, potassium acetate or potassium carbonate.
所述溶剂为二氯乙烷(DCE)、甲醇(MeOH)、乙腈(MeCN)、1,4-二氧六环(1,4-dioxane)或甲苯(toluene)其中任意一种或组合。The solvent is any one or a combination of dichloroethane (DCE), methanol (MeOH), acetonitrile (MeCN), 1,4-dioxane (1,4-dioxane) or toluene (toluene).
所述惰性氛围为氮气氛围,此外,还可以为氦气、氖气和氩气等气体氛围。The inert atmosphere is a nitrogen atmosphere, and can also be a gas atmosphere such as helium, neon, and argon.
所述噁嗪类化合物1、重氮化合物2、催化剂、添加剂和溶剂的摩尔比为:1:(2-3):(0.02-0.04):(0.08-0.16)。The molar ratio of the
所述溶剂当中反应体系的浓度为0.05M-0.2M。The concentration of the reaction system in the solvent is 0.05M-0.2M.
对于该反应的反应模式,其反应原理如图31所示:首先底物1a和催化剂作用得到环金属中间体A,随后充氮化合物和中间体A作用得到中间体B,紧接着中间体B发生卡宾的迁移插入得到中间体C,然后发生金属的脱除得到中间体D,中间体D再次发生上述过程得到双烷基化中间体E,中间体E快速互变为烯醇式F,中间体F中的羟基对亚胺键进行进攻得到中间体G,中间体G的NH对另外一分子的羰基进行亲核加成-消除得到中间体I,随后中间体I经历酸化-开环-关环-脱水等连续的转化给出了目标氮、氧杂稠环芳烃3aa。For the reaction mode of this reaction, the reaction principle is shown in Figure 31: First, the
本发明的有益效果:本发明开创性地选取简单易得的噁嗪类化合物和重氮化合物作为反应物,在金属铑催化剂的作用下,以噁嗪类化合物当中的亚胺作为弱的反应型定位基,通过连续的[4+2]串联策略一步实现了新型氮、氧杂稠环芳烃骨架的构建,该方法操作简单,具有显著的原子经济、步骤经济,为杂稠环芳烃的合成提供了一种简单有效的方法,并且该方法反应条件温和,其反应温度仅为80-120℃;另外,所制备氮、氧杂稠环芳烃的收率良好,产率普遍在80%以上,最高达到98%。所得产物有广阔的工业应用前景,同时为医药、天然产物合成以及发光材料等领域了提供了一种新思路和新方法。Beneficial effects of the present invention: the present invention creatively selects easily available oxazine compounds and diazonium compounds as reactants, and under the action of metal rhodium catalyst, takes the imine among the oxazine compounds as the weak reactive type Positioning base, the construction of a novel nitrogen and oxygen heterocyclic aromatic hydrocarbon skeleton is realized in one step through a continuous [4+2] tandem strategy. This method is simple to operate, has significant atom economy and step economy, and provides a good solution for the synthesis of heterocyclic aromatic hydrocarbons. A simple and effective method has been developed, and the reaction conditions of this method are mild, and the reaction temperature is only 80-120 ° C; in addition, the yield of the prepared nitrogen and oxygen heterocyclic aromatic hydrocarbons is good, and the yield is generally above 80%, the highest to 98%. The obtained product has broad industrial application prospects, and provides a new idea and a new method for the fields of medicine, natural product synthesis and luminescent materials.
附图说明Description of drawings
为了更清楚地说明本发明实施例或现有技术中的技术方案,下面将对实施例或现有技术描述中所需要使用的附图作简单地介绍,显而易见地,下面描述中的附图仅仅是本发明的一些实施例,对于本领域普通技术人员来讲,在不付出创造性劳动的前提下,还可以根据这些附图获得其他的附图。In order to illustrate the embodiments of the present invention or the technical solutions in the prior art more clearly, the following briefly introduces the accompanying drawings that are used in the description of the embodiments or the prior art. Obviously, the drawings in the following description are only These are some embodiments of the present invention. For those of ordinary skill in the art, other drawings can also be obtained according to these drawings without creative efforts.
图1是化合物3aa的核磁1H谱图;图2是化合物3aa的核磁13C谱图。Fig. 1 is the nuclear magnetic 1 H spectrum of compound 3aa; Fig. 2 is the nuclear magnetic 13 C spectrum of compound 3aa.
图3是化合物3ba的核磁1H谱图;图4是化合物3ba的核磁13C谱图。Figure 3 is the NMR 1 H spectrum of the compound 3ba; Figure 4 is the NMR 13 C spectrum of the compound 3ba.
图5是化合物3ca的核磁1H谱图;图6是化合物3ca的核磁13C谱图。FIG. 5 is the NMR 1 H spectrum of the compound 3ca; FIG. 6 is the NMR 13 C spectrum of the compound 3ca.
图7是化合物3da的核磁1H谱图;图8是化合物3da的核磁13C谱图。FIG. 7 is the nuclear magnetic 1 H spectrum of compound 3da; FIG. 8 is the nuclear magnetic 13 C spectrum of compound 3da.
图9是化合物3ea的核磁1H谱图;图10是化合物3ea的核磁13C谱图。Fig. 9 is the NMR 1 H spectrum of the compound 3ea; Fig. 10 is the NMR 13 C spectrum of the compound 3ea.
图11是化合物3fa的核磁1H谱图;图12是化合物3fa的核磁13C谱图。Fig. 11 is the 1H NMR spectrum of compound 3fa; Fig. 12 is the NMR 13C spectrum of compound 3fa.
图13是化合物3ga的核磁1H谱图;图14是化合物3ga的核磁13C谱图。Figure 13 is the NMR 1 H spectrum of the compound 3ga; Figure 14 is the NMR 13 C spectrum of the compound 3ga.
图15是化合物3ha的核磁1H谱图;图16是化合物3ha的核磁13C谱图。Figure 15 is the nuclear magnetic 1 H spectrum of compound 3ha; Figure 16 is the nuclear magnetic 13 C spectrum of compound 3ha.
图17是化合物3ia的核磁1H谱图;图18是化合物3ia的核磁13C谱图。FIG. 17 is the 1H NMR spectrum of compound 3ia; FIG. 18 is the 13C NMR spectrum of compound 3ia.
图19是化合物3ja的核磁1H谱图;图20是化合物3ja的核磁13C谱图。Fig. 19 is the nuclear magnetic 1 H spectrum of compound 3ja; Fig. 20 is the nuclear magnetic 13 C spectrum of compound 3ja.
图21是化合物3ka的核磁1H谱图;图22是化合物3ka的核磁13C谱图。Figure 21 is the nuclear magnetic 1 H spectrum of compound 3ka; Figure 22 is the nuclear magnetic 13 C spectrum of compound 3ka.
图23是化合物3ab的核磁1H谱图;图24是化合物3ab的核磁13C谱图。Figure 23 is the nuclear magnetic 1 H spectrum of compound 3ab; Figure 24 is the nuclear magnetic 13 C spectrum of compound 3ab.
图25是化合物3ac的核磁1H谱图;图26是化合物3ac的核磁13C谱图。Fig. 25 is the NMR 1 H spectrum of compound 3ac; Fig. 26 is the NMR 13 C spectrum of compound 3ac.
图27是化合物3ad的核磁1H谱图;图28是化合物3ad的核磁13C谱图。Figure 27 is the nuclear magnetic 1 H spectrum of compound 3ad; Figure 28 is the nuclear magnetic 13 C spectrum of compound 3ad.
图29是化合物3ae的核磁1H谱图;图30是化合物3ae的核磁13C谱图。Figure 29 is the nuclear magnetic 1 H spectrum of compound 3ae; Figure 30 is the nuclear magnetic 13 C spectrum of compound 3ae.
图31是本发明反应机理图。Figure 31 is a diagram of the reaction mechanism of the present invention.
具体实施方式Detailed ways
下面将结合本发明实施例中的附图,对本发明实施例中的技术方案进行清楚、完整地描述,显然,所描述的实施例仅仅是本发明一部分实施例,而不是全部的实施例。基于本发明中的实施例,本领域普通技术人员在没有付出创造性劳动前提下所获得的所有其他实施例,都属于本发明保护的范围。The technical solutions in the embodiments of the present invention will be clearly and completely described below with reference to the accompanying drawings in the embodiments of the present invention. Obviously, the described embodiments are only a part of the embodiments of the present invention, rather than all the embodiments. Based on the embodiments of the present invention, all other embodiments obtained by those of ordinary skill in the art without creative efforts shall fall within the protection scope of the present invention.
实施例1Example 1
在氮气条件下,将3-芳基苯并噁嗪化合物1a(0.20mmol),重氮化合物2a(0.44mmol),[Cp*RhCl2]2(4mol%),AgSbF6(16mol%),MesCOOH(0.4mmol)和溶剂DCE(2.0mL)加入到10mL密封管中,在80度的反应模块中反应10h,反应结束后减压除去溶剂,用硅胶柱分离得到目标产物5,12-二甲基异喹啉[2,1,8-mna]吩恶嗪-4,13-二羧酸二乙酯(3aa),所有洗脱剂为石油醚乙酸乙酯和二氯甲烷按30:1:1的比例配制而成。产物数据表征:黄褐色液体,78%的产率。产物的核磁图谱如图1和2所示,1H NMR(400MHz,CDCl3)δ7.23(d,J=8.6Hz,1H),7.11(t,J=8.0Hz,1H),7.03–6.97(m,1H),6.94(t,J=7.6Hz,1H),6.89(d,J=7.7Hz,1H),6.82(d,J=7.9Hz,1H),6.77(d,J=7.3Hz,1H),4.49–4.38(m,4H),2.33(s,6H),1.45–1.40(m,6H).13CNMR(101MHz,CDCl3)δ168.6,167.7,150.6,138.6,137.1,131.2,130.2,129.1,128.4,127.7,125.8,125.7,125.0,123.9,123.6,120.1,119.6,117.1,116.8,115.2,61.1,61.0,19.0,14.4,14.3,13.2.HRMS:[M+H]+calculated forC26H24NO5 +:430.1649,found:430.1635.Under nitrogen, 3-
实施例2Example 2
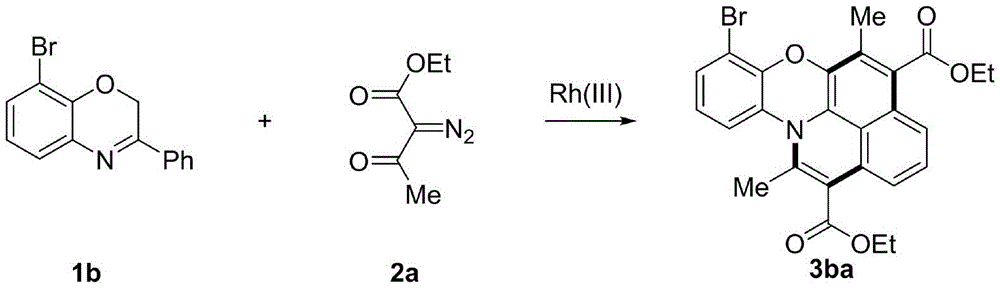
在氮气条件下,将3-芳基苯并噁嗪化合物1b(0.20mmol),重氮化合物2a(0.44mmol),[Cp*RhCl2]2(4mol%),AgSbF6(16mol%),MesCOOH(0.4mmol)和溶剂DCE(2.0mL)加入到10mL密封管中,在80度的反应模块中反应10h,反应结束后减压除去溶剂,用硅胶柱分离得到目标产物7-溴-5,12-二甲基异喹啉[2,1,8-mna]吩恶嗪-4,13-二羧酸二乙酯(3ba),所有洗脱剂为石油醚乙酸乙酯和二氯甲烷按30:1:1的比例配制而成。产物数据表征:黄褐色固体,98%的产率,熔程:119-120℃。产物的核磁图谱如图3和4所示,1H NMR(400MHz,CDCl3)δ7.26(t,J=8.3Hz,2H),7.14(t,J=8.0Hz,1H),6.86(t,J=8.0Hz,1H),6.79(dd,J=7.5,4.6Hz,2H),4.49–4.39(m,4H),2.44(s,3H),2.33(s,3H),1.45–1.40(m,6H).13C NMR(101MHz,CDCl3)δ168.6,167.6,147.7,138.0,137.1,132.0,131.0,129.2,128.8,128.7,128.0,126.3,124.7,124.6,124.0,120.6,118.5,117.6,115.7,111.2,61.3,61.2,19.0,14.4,14.4,13.5.HRMS:[M+H]+calculated for C26H23BrNO5 +:508.0754,found:508.0717.Under nitrogen, 3-arylbenzoxazine compound 1b (0.20 mmol), diazo compound 2a (0.44 mmol), [Cp*RhCl 2 ] 2 (4 mol %), AgSbF 6 (16 mol %), MesCOOH (0.4mmol) and solvent DCE (2.0mL) were added to a 10mL sealed tube, and reacted in a reaction module at 80 degrees for 10h. After the reaction, the solvent was removed under reduced pressure, and the target product 7-bromo-5,12 was obtained by separation with a silica gel column. - Diethylisoquinoline[2,1,8-mna]phenoxazine-4,13-dicarboxylate (3ba), all eluents were petroleum ether, ethyl acetate and dichloromethane as per 30 : 1:1 ratio is formulated. Characterization of product data: tan solid, 98% yield, melting range: 119-120°C. The nuclear magnetic spectrum of the product is shown in Figures 3 and 4, 1 H NMR (400MHz, CDCl 3 )δ7.26(t, J=8.3Hz, 2H), 7.14(t, J=8.0Hz, 1H), 6.86(t , J=8.0Hz, 1H), 6.79 (dd, J=7.5, 4.6Hz, 2H), 4.49–4.39 (m, 4H), 2.44 (s, 3H), 2.33 (s, 3H), 1.45–1.40 ( m, 6H). 13 C NMR (101MHz, CDCl 3 )δ168.6, 167.6, 147.7, 138.0, 137.1, 132.0, 131.0, 129.2, 128.8, 128.7, 128.0, 126.3, 124.7, 124.6, 124.0, 120., 6, 118.5, 115.7, 111.2, 61.3, 61.2, 19.0, 14.4, 14.4, 13.5. HRMS: [M+H] + calculated for C 26 H 23 BrNO 5 + : 508.0754, found: 508.0717.
实施例3Example 3
在氮气条件下,将3-芳基苯并噁嗪化合物1c(0.20mmol),重氮化合物2a(0.44mmol),[Cp*RhCl2]2(4mol%),AgSbF6(16mol%),MesCOOH(0.4mmol)和溶剂DCE(2.0mL)加入到10mL密封管中,在80度的反应模块中反应10h,反应结束后减压除去溶剂,用硅胶柱分离得到目标产物8-氟-5,12-二甲基异喹啉[2,1,8-mna]吩恶嗪-4,13-二羧酸二乙酯(3ca),所有洗脱剂为石油醚乙酸乙酯和二氯甲烷按30:1:1的比例配制而成。产物数据表征:黄色固体,89%的产率,熔程:104-105℃。产物的核磁图谱如图5和6所示,1H NMR(400MHz,CDCl3)δ7.26(t,J=8.3Hz,2H),7.14(t,J=8.0Hz,1H),6.86(t,J=8.0Hz,1H),6.79(dd,J=7.5,4.6Hz,2H),4.49–4.39(m,4H),2.44(s,3H),2.33(s,3H),1.45–1.40(m,6H).13C NMR(101MHz,CDCl3)δ168.6,167.6,147.7,138.0,137.1,132.0,131.0,129.2,128.8,128.7,128.0,126.3,124.7,124.6,124.0,120.6,118.5,117.6,115.7,111.2,61.3,61.2,19.0,14.4,14.4,13.5.HRMS:[M+Na]+calculated for C26H22FNNaO5 +:470.1374,found:470.1371.Under nitrogen, 3-arylbenzoxazine compound 1c (0.20 mmol), diazo compound 2a (0.44 mmol), [Cp*RhCl 2 ] 2 (4 mol %), AgSbF 6 (16 mol %), MesCOOH (0.4mmol) and solvent DCE (2.0mL) were added to a 10mL sealed tube, and reacted in a reaction module at 80 degrees for 10h. After the reaction was completed, the solvent was removed under reduced pressure, and the target product 8-fluoro-5,12 was obtained by separation with a silica gel column. - Diethylisoquinoline[2,1,8-mna]phenoxazine-4,13-dicarboxylate (3ca), all eluents were petroleum ether, ethyl acetate and dichloromethane as per 30 : 1:1 ratio is formulated. Product Data Characterization: Yellow solid, 89% yield, melting range: 104-105°C. The nuclear magnetic spectrum of the product is shown in Figures 5 and 6, 1 H NMR (400MHz, CDCl 3 )δ7.26(t, J=8.3Hz, 2H), 7.14(t, J=8.0Hz, 1H), 6.86(t , J=8.0Hz, 1H), 6.79 (dd, J=7.5, 4.6Hz, 2H), 4.49–4.39 (m, 4H), 2.44 (s, 3H), 2.33 (s, 3H), 1.45–1.40 ( m, 6H). 13 C NMR (101MHz, CDCl 3 )δ168.6, 167.6, 147.7, 138.0, 137.1, 132.0, 131.0, 129.2, 128.8, 128.7, 128.0, 126.3, 124.7, 124.6, 124.0, 120., 6, 118.5, 115.7, 111.2, 61.3, 61.2, 19.0, 14.4, 14.4, 13.5. HRMS: [M+Na] + calculated for C26H22FNNaO5 + : 470.1374 , found: 470.1371 .
实施例4Example 4
在氮气条件下,将3-芳基苯并噁嗪化合物1d(0.20mmol),重氮化合物2a(0.44mmol),[Cp*RhCl2]2(4mol%),AgSbF6(16mol%),MesCOOH(0.4mmol)和溶剂DCE(2.0mL)加入到10mL密封管中,在80度的反应模块中反应10h,反应结束后减压除去溶剂,用硅胶柱分离得到目标产物8-氯-5,12-二甲基异喹啉[2,1,8-mna]吩恶嗪-4,13-二羧酸二乙酯(3da),所有洗脱剂为石油醚乙酸乙酯和二氯甲烷按30:1:1的比例配制而成。产物数据表征:黄褐色固体,82%的产率,熔程:127-128℃。产物的核磁图谱如图7和8所示,1H NMR(400MHz,CDCl3)δ7.21(d,J=8.6Hz,1H),7.15–7.06(m,1H),6.98–6.89(m,2H),6.74(t,J=7.6Hz,2H),4.46–4.36(m,4H),2.30(s,3H),2.29(s,3H),1.43-1.38(m,6H).13C NMR(101MHz,CDCl3)13C NMR(101MHz,CDCl3)δ168.6,167.7,151.2,138.0,136.8,131.1,130.9,129.2,128.9,128.6,128.0,125.9,124.9,124.0,123.9,120.4,119.9,117.7,117.3,115.6,61.4,61.2,19.0,14.5,14.4,13.3.HRMS:[M+H]+calculated for C26H23ClNO5 +:464.1259,found:464.1244.Under nitrogen, 3-arylbenzoxazine compound 1d (0.20 mmol), diazo compound 2a (0.44 mmol), [Cp*RhCl 2 ] 2 (4 mol %), AgSbF 6 (16 mol %), MesCOOH (0.4mmol) and solvent DCE (2.0mL) were added to a 10mL sealed tube, and reacted in a reaction module at 80 degrees for 10h. After the reaction was completed, the solvent was removed under reduced pressure, and the target product 8-chloro-5,12 was obtained by separation with a silica gel column. - Diethylisoquinoline[2,1,8-mna]phenoxazine-4,13-dicarboxylate (3da), all eluents were petroleum ether, ethyl acetate and dichloromethane as per 30 : 1:1 ratio is formulated. Characterization of product data: tan solid, 82% yield, melting range: 127-128°C. The NMR spectra of the products are shown in Figures 7 and 8, 1 H NMR (400MHz, CDCl 3 )δ7.21(d, J=8.6Hz, 1H), 7.15-7.06(m, 1H), 6.98-6.89(m, 2H), 6.74(t, J=7.6Hz, 2H), 4.46-4.36(m, 4H), 2.30(s, 3H), 2.29(s, 3H), 1.43-1.38(m, 6H). 13 C NMR (101MHz, CDCl 3 ) 13 C NMR (101 MHz, CDCl 3 ) δ 168.6, 167.7, 151.2, 138.0, 136.8, 131.1, 130.9, 129.2, 128.9, 128.6, 128.1, 125.9, 124.9, 124.0, 123.9, 17.9.4 , 117.3, 115.6, 61.4, 61.2, 19.0, 14.5, 14.4, 13.3. HRMS: [M+H] + calculated for C 26 H 23 ClNO 5 + : 464.1259, found: 464.1244.
实施例5Example 5
在氮气条件下,将3-芳基苯并噁嗪化合物1e(0.20mmol),重氮化合物2a(0.44mmol),[Cp*RhCl2]2(4mol%),AgSbF6(16mol%),MesCOOH(0.4mmol)和溶剂DCE(2.0mL)加入到10mL密封管中,在80度的反应模块中反应10h,反应结束后减压除去溶剂,用硅胶柱分离得到目标产物8-溴-5,12-二甲基异喹啉[2,1,8-mna]吩恶嗪-4,13-二羧酸二乙酯(3ea),所有洗脱剂为石油醚乙酸乙酯和二氯甲烷按30:1:1的比例配制而成。产物数据表征:黄色固体,89%的产率,熔程:144-145℃。产物的核磁图谱如图9和10所示,1H NMR(400MHz,CDCl3)δ7.20(d,J=8.6Hz,1H),7.13–7.00(m,3H),6.74(d,J=7.2Hz,1H),6.64(d,J=8.4Hz,1H),4.46–4.35(m,4H),2.28(s,3H),2.27(s,3H),1.40(td,J=7.1,5.2Hz,6H).13C NMR(101MHz,CDCl3)δ168.5,167.6,151.2,137.9,136.8,130.8,129.6,128.8,128.5,127.9,126.9,125.8,124.8,123.9,120.4,120.4,120.2,118.3,117.3,115.6,61.3,61.2,18.9,14.4,14.4,13.2.HRMS:[M+Na]+calculated for C26H22BrNNaO5 +=530.0574,found:530.0564.Under nitrogen, 3-arylbenzoxazine compound 1e (0.20 mmol), diazo compound 2a (0.44 mmol), [Cp*RhCl 2 ] 2 (4 mol %), AgSbF 6 (16 mol %), MesCOOH (0.4mmol) and solvent DCE (2.0mL) were added into a 10mL sealed tube, and reacted in a reaction module at 80 degrees for 10h. After the reaction was completed, the solvent was removed under reduced pressure, and the target product 8-bromo-5,12 was obtained by separation with a silica gel column. - Diethylisoquinoline[2,1,8-mna]phenoxazine-4,13-dicarboxylate (3ea), all eluents were petroleum ether, ethyl acetate and dichloromethane as per 30 : 1:1 ratio is formulated. Product Data Characterization: Yellow solid, 89% yield, melting range: 144-145°C. The NMR spectra of the products are shown in Figures 9 and 10, 1 H NMR (400MHz, CDCl 3 ) δ 7.20 (d, J=8.6 Hz, 1H), 7.13-7.00 (m, 3H), 6.74 (d, J= 7.2Hz, 1H), 6.64 (d, J=8.4Hz, 1H), 4.46–4.35 (m, 4H), 2.28 (s, 3H), 2.27 (s, 3H), 1.40 (td, J=7.1, 5.2 Hz, 6H). 13 C NMR (101MHz, CDCl 3 )δ168.5, 167.6, 151.2, 137.9, 136.8, 130.8, 129.6, 128.8, 128.5, 127.9, 126.9, 125.8, 124.8, 123.9, 120.4, 120.4, 120.2, 117.3, 115.6, 61.3, 61.2, 18.9, 14.4, 14.4, 13.2. HRMS: [M+Na] + calculated for C 26 H 22 BrNNaO 5 + =530.0574, found: 530.0564.
实施例6Example 6
在氮气条件下,将3-芳基苯并噁嗪化合物1f(0.20mmol),重氮化合物2a(0.44mmol),[Cp*RhCl2]2(4mol%),AgSbF6(16mol%),MesCOOH(0.4mmol)和溶剂DCE(2.0mL)加入到10mL密封管中,在80度的反应模块中反应10h,反应结束后减压除去溶剂,用硅胶柱分离得到目标产物8-甲基-5,12-二甲基异喹啉[2,1,8-mna]吩恶嗪-4,13-二羧酸二乙酯(3fa),所有洗脱剂为石油醚乙酸乙酯和二氯甲烷按30:1:1的比例配制而成。产物数据表征:黄色固体,77%的产率,熔程:100-101℃。产物的核磁图谱如图11和12所示,1H NMR(400MHz,CDCl3)δ7.18(d,J=8.6Hz,1H),7.08(t,J=8.0Hz,1H),6.78–6.68(m,4H),4.48–4.34(m,4H),2.31(s,3H),2.31(s,3H),2.26(s,3H),1.40(td,J=7.1,3.7Hz,6H).13C NMR(101MHz,CDCl3)δ168.8,168.0,150.6,139.0,137.1,136.1,131.4,129.3,128.4,127.8,127.5,125.9,125.1,124.4,123.4,120.0,119.4,117.9,116.2,115.1,61.2,61.0,20.7,19.2,14.5,14.4,13.3.HRMS:[M+Na]+calculated for C27H25NNaO5 +=466.1625,found:466.1617.Under nitrogen, 3-arylbenzoxazine compound 1f (0.20 mmol), diazo compound 2a (0.44 mmol), [Cp*RhCl 2 ] 2 (4 mol %), AgSbF 6 (16 mol %), MesCOOH (0.4mmol) and solvent DCE (2.0mL) were added into a 10mL sealed tube, and reacted in a reaction module at 80 degrees for 10h. After the reaction was completed, the solvent was removed under reduced pressure, and the target product 8-methyl-5 was obtained by separation with a silica gel column. Diethyl 12-dimethylisoquinoline[2,1,8-mna]phenoxazine-4,13-dicarboxylate (3fa), all eluents were petroleum ether, ethyl acetate and dichloromethane It is formulated in a ratio of 30:1:1. Characterization of product data: yellow solid, 77% yield, melting range: 100-101°C. The NMR spectra of the product are shown in Figures 11 and 12, 1 H NMR (400MHz, CDCl 3 ) δ 7.18 (d, J=8.6Hz, 1H), 7.08 (t, J=8.0Hz, 1H), 6.78-6.68 (m, 4H), 4.48–4.34(m, 4H), 2.31(s, 3H), 2.31(s, 3H), 2.26(s, 3H), 1.40(td, J=7.1, 3.7Hz, 6H). 13 C NMR (101MHz, CDCl 3 )δ168.8,168.0,150.6,139.0,137.1,136.1,131.4,129.3,128.4,127.8,127.5,125.9,125.1,124.4,123.4,120.0,111.2.4,117.9 , 61.0, 20.7, 19.2, 14.5, 14.4, 13.3. HRMS: [M+Na] + calculated for C 27 H 25 NNaO 5 + =466.1625, found: 466.1617.
实施例7Example 7
在氮气条件下,将3-芳基苯并噁嗪化合物1g(0.20mmol),重氮化合物2a(0.44mmol),[Cp*RhCl2]2(4mol%),AgSbF6(16mol%),MesCOOH(0.4mmol)和溶剂DCE(2.0mL)加入到10mL密封管中,在80度的反应模块中反应10h,反应结束后减压除去溶剂,用硅胶柱分离得到目标产物9-溴-5,12-二甲基异喹啉[2,1,8-mna]吩恶嗪-4,13-二羧酸二乙酯(3ga),所有洗脱剂为石油醚乙酸乙酯和二氯甲烷按30:1:1的比例配制而成。产物数据表征:黄色固体,91%的产率,熔程:132-133℃。产物的核磁图谱如图13和14所示,1H NMR(400MHz,CDCl3)δ7.21(d,J=8.6Hz,1H),7.13–7.08(m,2H),6.94(d,J=1.6Hz,1H),6.77–6.74(m,2H),4.45–4.37(m,4H),2.31(s,3H),2.30(s,3H),1.43–1.39(m,6H).13C NMR(101MHz,CDCl3)δ168.6,167.6,149.8,137.6,137.1,131.8,130.7,128.7,128.4,128.4,128.0,125.9,124.8,124.2,122.3,120.6,118.3,117.9,116.3,115.8,61.4,61.3,18.9,14.5,14.4,13.3.HRMS:[M+H]+calculated for C26H23BrNO5 +=508.0754,found:508.0747.Under nitrogen, 3-arylbenzoxazine compound 1 g (0.20 mmol), diazo compound 2a (0.44 mmol), [Cp*RhCl 2 ] 2 (4 mol %), AgSbF 6 (16 mol %), MesCOOH (0.4mmol) and solvent DCE (2.0mL) were added into a 10mL sealed tube, and reacted in a reaction module at 80 degrees for 10h. After the reaction was completed, the solvent was removed under reduced pressure, and the target product 9-bromo-5,12 was obtained by separation with a silica gel column. - Diethylisoquinoline[2,1,8-mna]phenoxazine-4,13-dicarboxylate (3ga), all eluents were petroleum ether, ethyl acetate and dichloromethane as per 30 : 1:1 ratio is formulated. Product Data Characterization: Yellow solid, 91% yield, melting range: 132-133°C. The NMR spectra of the products are shown in Figures 13 and 14, 1 H NMR (400 MHz, CDCl 3 ) δ 7.21 (d, J=8.6 Hz, 1H), 7.13-7.08 (m, 2H), 6.94 (d, J= 1.6Hz, 1H), 6.77–6.74 (m, 2H), 4.45–4.37 (m, 4H), 2.31 (s, 3H), 2.30 (s, 3H), 1.43–1.39 (m, 6H). 13 C NMR (101MHz, CDCl 3 )δ168.6,167.6,149.8,137.6,137.1,131.8,130.7,128.7,128.4,128.4,128.0,125.9,124.8,124.2,122.3,120.6,118.3,117.9,116.6.3 18.9, 14.5, 14.4, 13.3. HRMS: [M+H] + calculated for C 26 H 23 BrNO 5 + = 508.0754, found: 508.0747.
实施例8Example 8
在氮气条件下,将3-芳基苯并噁嗪化合物1h(0.20mmol),重氮化合物2a(0.44mmol),[Cp*RhCl2]2(4mol%),AgSbF6(16mol%),MesCOOH(0.4mmol)和溶剂DCE(2.0mL)加入到10mL密封管中,在80度的反应模块中反应10h,反应结束后减压除去溶剂,用硅胶柱分离得到目标产物9-甲氧基-5,12-二甲基异喹啉[2,1,8-mna]吩恶嗪-4,13-二羧酸二乙酯(3ha),所有洗脱剂为石油醚乙酸乙酯和二氯甲烷按30:1:1的比例配制而成。产物数据表征:黄色固体,53%的产率,熔程:162-163℃。产物的核磁图谱如图15和16所示,1H NMR(400MHz,CDCl3)δ7.20(d,J=8.3Hz,1H),7.14–7.04(m,1H),6.80(d,J=8.8Hz,1H),6.74(d,J=7.0Hz,1H),6.52(dd,J=8.8,2.6Hz,1H),6.39(d,J=2.7Hz,1H),4.46–4.36(m,4H),2.34(s,3H),2.31(s,3H),1.42–1.38(m,6H).13C NMR(101MHz,CDCl3)δ168.7,167.8,156.2,144.2,138.4,137.6,131.0,130.9,128.9,128.2,127.6,125.9,125.0,123.7,120.3,117.0,115.3,109.4,106.6,61.2,61.1,55.9,19.2,14.4,14.4,13.3.HRMS:[M+H]+calculated for C27H26NO6 +=460.1755,found:460.1753.Under nitrogen, 3-arylbenzoxazine compound 1h (0.20 mmol), diazo compound 2a (0.44 mmol), [Cp*RhCl 2 ] 2 (4 mol %), AgSbF 6 (16 mol %), MesCOOH (0.4 mmol) and solvent DCE (2.0 mL) were added to a 10 mL sealed tube, and reacted in a reaction module at 80 degrees for 10 h. After the reaction, the solvent was removed under reduced pressure, and the target product 9-methoxy-5 was obtained by separation with a silica gel column. ,12-Dimethylisoquinoline[2,1,8-mna]phenoxazine-4,13-dicarboxylate diethyl ester (3ha), all eluents were petroleum ether ethyl acetate and dichloromethane It is formulated according to the ratio of 30:1:1. Product Data Characterization: Yellow solid, 53% yield, melting range: 162-163°C. The NMR spectra of the products are shown in Figures 15 and 16, 1 H NMR (400 MHz, CDCl 3 ) δ 7.20 (d, J=8.3 Hz, 1H), 7.14-7.04 (m, 1H), 6.80 (d, J= 8.8Hz, 1H), 6.74 (d, J=7.0Hz, 1H), 6.52 (dd, J=8.8, 2.6Hz, 1H), 6.39 (d, J=2.7Hz, 1H), 4.46–4.36 (m, 4H), 2.34(s, 3H), 2.31(s, 3H), 1.42–1.38(m, 6H). 13 C NMR (101 MHz, CDCl 3 ) δ 168.7, 167.8, 156.2, 144.2, 138.4, 137.6, 131.0, 130.9 ,128.9,128.2,127.6,125.9,125.0,123.7,120.3,117.0,115.3,109.4,106.6,61.2,61.1,55.9,19.2,14.4,14.4,13.3.HRMS: [M+H] + calculated for C 27 H 26 NO 6 + = 460.1755, found: 460.1753.
实施例9Example 9
在氮气条件下,将3-芳基苯并噁嗪化合物1i(0.20mmol),重氮化合物2a(0.44mmol),[Cp*RhCl2]2(4mol%),AgSbF6(16mol%),MesCOOH(0.4mmol)和溶剂DCE(2.0mL)加入到10mL密封管中,在80度的反应模块中反应10h,反应结束后减压除去溶剂,用硅胶柱分离得到目标产物2-氟-5,12-二甲基异喹啉[2,1,8-mna]吩恶嗪-4,13-二羧酸二乙酯(3ia),所有洗脱剂为石油醚乙酸乙酯和二氯甲烷按30:1:1的比例配制而成。产物数据表征:黄色固体,82%的产率,熔程:162-163℃。产物的核磁图谱如图17和18所示,1H NMR(400MHz,CDCl3)δ7.07–7.03(m,1H),7.00–6.96(m,1H),6.92(dd,J=7.9,1.5Hz,1H),6.88(dd,J=11.4,2.0Hz,1H),6.85(dd,J=8.0,1.2Hz,1H),6.61(dd,J=10.2,2.0Hz,1H),4.46–4.37(m,4H),2.37(s,3H),2.32(s,3H),1.43–1.39(m,6H).13C NMR(101MHz,CDCl3)δ168.2,167.1,162.6(d,J=243.1Hz),150.7,140.8,136.6,131.9(d,J=11.2Hz),131.3,129.6,129.5,127.3,126.2,123.9,122.8(d,J=5.4Hz),122.2,120.0,117.3,115.5,105.4(d,J=29.2Hz),103.8(d,J=24.6Hz),61.2,61.2,19.4,14.4,14.3,13.4.HRMS:[M+H]+calculated for C26H23FNO5 +=448.1555,found:448.1539.Under nitrogen, 3-arylbenzoxazine compound 1i (0.20 mmol), diazo compound 2a (0.44 mmol), [Cp*RhCl 2 ] 2 (4 mol %), AgSbF 6 (16 mol %), MesCOOH (0.4mmol) and solvent DCE (2.0mL) were added to a 10mL sealed tube, and reacted in a reaction module at 80 degrees for 10h. After the reaction, the solvent was removed under reduced pressure, and the target product 2-fluoro-5,12 was obtained by separation with a silica gel column. - Diethylisoquinoline[2,1,8-mna]phenoxazine-4,13-dicarboxylate (3ia), all eluents were petroleum ether, ethyl acetate and dichloromethane as per 30 : 1:1 ratio is formulated. Characterization of product data: yellow solid, 82% yield, melting range: 162-163°C. The NMR spectra of the products are shown in Figures 17 and 18, 1 H NMR (400 MHz, CDCl 3 ) δ 7.07-7.03 (m, 1H), 7.00-6.96 (m, 1H), 6.92 (dd, J=7.9, 1.5 Hz, 1H), 6.88 (dd, J=11.4, 2.0Hz, 1H), 6.85 (dd, J=8.0, 1.2Hz, 1H), 6.61 (dd, J=10.2, 2.0Hz, 1H), 4.46–4.37 (m,4H), 2.37(s,3H), 2.32(s,3H), 1.43–1.39(m,6H). 13 C NMR (101 MHz, CDCl 3 ) δ 168.2, 167.1, 162.6 (d, J=243.1 Hz ( d, J=29.2Hz), 103.8 (d, J=24.6Hz), 61.2, 61.2, 19.4, 14.4, 14.3, 13.4. HRMS: [M+H] + calculated for C 26 H 23 FNO 5 + =448.1555, found: 448.1539.
实施例10Example 10
在氮气条件下,将3-芳基苯并噁嗪化合物1j(0.20mmol),重氮化合物2a(0.44mmol),[Cp*RhCl2]2(4mol%),AgSbF6(16mol%),MesCOOH(0.4mmol)和溶剂DCE(2.0mL)加入到10mL密封管中,在80度的反应模块中反应10h,反应结束后减压除去溶剂,用硅胶柱分离得到目标产物2-溴-5,12-二甲基异喹啉[2,1,8-mna]吩恶嗪-4,13-二羧酸二乙酯(3ja),所有洗脱剂为石油醚乙酸乙酯和二氯甲烷按30:1:1的比例配制而成。产物数据表征:黄色固体,87%的产率,熔程:142-143℃。产物的核磁图谱如图19和20所示,1H NMR(400MHz,CDCl3)δ7.39d,J=1.5Hz,1H),7.10–7.05(m,1H),7.03–7.01(m,1H),6.94(dd,J=7.8,1.5Hz,1H),6.89–6.84(m,2H),4.49–4.39(m,4H),2.38(s,3H),2.34(s,3H),1.45–1.42(m,6H).13C NMR(101MHz,CDCl3)δ168.1,167.2,150.7,140.5,137.4,131.3,131.0,129.7,129.4,127.0,126.3,124.2,123.6,122.8,122.1,120.0,118.4,117.4,115.4,77.2,61.4,61.3,19.4,14.4,14.4,13.4.HRMS:[M+H]+calculated for C26H23BrNO5 +=508.0754,found:508.0742.Under nitrogen, 3-arylbenzoxazine compound 1j (0.20 mmol), diazo compound 2a (0.44 mmol), [Cp*RhCl 2 ] 2 (4 mol %), AgSbF 6 (16 mol %), MesCOOH (0.4mmol) and solvent DCE (2.0mL) were added into a 10mL sealed tube, and reacted in a reaction module at 80 degrees for 10h. After the reaction, the solvent was removed under reduced pressure, and the target product 2-bromo-5,12 was obtained by separation with a silica gel column. - Diethylisoquinoline[2,1,8-mna]phenoxazine-4,13-dicarboxylate (3ja), all eluents were petroleum ether, ethyl acetate and dichloromethane as per 30 : 1:1 ratio is formulated. Product Data Characterization: Yellow solid, 87% yield, melting range: 142-143°C. The NMR spectra of the products are shown in Figures 19 and 20, 1 H NMR (400MHz, CDCl 3 )δ7.39d, J=1.5Hz, 1H), 7.10-7.05(m,1H), 7.03-7.01(m,1H) ,6.94(dd,J=7.8,1.5Hz,1H),6.89-6.84(m,2H),4.49-4.39(m,4H),2.38(s,3H),2.34(s,3H),1.45-1.42 (m, 6H). 13 C NMR (101MHz, CDCl 3 ) δ 168.1, 167.2, 150.7, 140.5, 137.4, 131.3, 131.0, 129.7, 129.4, 127.0, 126.3, 124.2, 123.6, 122.8, 122.1, 120.0, 118.4 ,115.4,77.2,61.4,61.3,19.4,14.4,14.4,13.4.HRMS: [M+H] + calculated for C 26 H 23 BrNO 5 + =508.0754, found: 508.0742.
实施例11Example 11
在氮气条件下,将3-芳基苯并噁嗪化合物1k(0.20mmol),重氮化合物2a(0.44mmol),[Cp*RhCl2]2(4mol%),AgSbF6(16mol%),MesCOOH(0.4mmol)和溶剂DCE(2.0mL)加入到10mL密封管中,在80度的反应模块中反应10h,反应结束后减压除去溶剂,用硅胶柱分离得到目标产物2-甲氧基-5,12-二甲基异喹啉[2,1,8-mna]吩恶嗪-4,13-二羧酸二乙酯(3ka),所有洗脱剂为石油醚乙酸乙酯和二氯甲烷按30:1:1的比例配制而成。产物数据表征:红色固体,77%的产率,熔程:172-173℃。产物的核磁图谱如图21和22所示,1H NMR(400MHz,CDCl3)δ7.52(s,1H),7.08(t,J=7.7Hz,1H),7.03–6.99(m,1H),6.95–6.93(m,2H),6.88–6.86(d,J=7.4Hz,1H),4.49(q,J=7.1Hz,1H),4.43(q,J=7.2Hz,1H),2.39(s,3H),2.36(s,3H),1.45–1.41(m,6H).13C NMR(101MHz,CDCl3)δ167.9,167.2,150.5,140.9,138.7,131.1,130.7,129.7(q,J=31.8Hz),129.5,127.3,126.3,126.2,124.7,124.3,124.2(q,J=272.7Hz),120.0,117.7(dd,J=9.2),117.7(d,J=4.7Hz),117.4,115.7,110.8(d,J=3.2Hz).61.5,61.3,19.5,14.3,14.3,13.3.HRMS:[M+H]+calculated forC27H23F3NO5 +=498.1523,found:498.1508.Under nitrogen, 3-arylbenzoxazine compound 1k (0.20 mmol), diazo compound 2a (0.44 mmol), [Cp*RhCl 2 ] 2 (4 mol %), AgSbF 6 (16 mol %), MesCOOH (0.4mmol) and solvent DCE (2.0mL) were added to a 10mL sealed tube, and reacted in a reaction module at 80 degrees for 10h. After the reaction, the solvent was removed under reduced pressure, and the target product 2-methoxy-5 was obtained by separation with a silica gel column. ,12-Dimethylisoquinoline[2,1,8-mna]phenoxazine-4,13-dicarboxylate (3ka), all eluents were petroleum ether ethyl acetate and dichloromethane It is formulated according to the ratio of 30:1:1. Characterization of product data: red solid, 77% yield, melting range: 172-173°C. The NMR spectra of the product are shown in Figures 21 and 22, 1 H NMR (400MHz, CDCl 3 ) δ 7.52 (s, 1H), 7.08 (t, J=7.7Hz, 1H), 7.03-6.99 (m, 1H) ,6.95–6.93(m,2H),6.88–6.86(d,J=7.4Hz,1H),4.49(q,J=7.1Hz,1H),4.43(q,J=7.2Hz,1H),2.39( s, 3H), 2.36 (s, 3H), 1.45–1.41 (m, 6H). 13 C NMR (101 MHz, CDCl 3 ) δ 167.9, 167.2, 150.5, 140.9, 138.7, 131.1, 130.7, 129.7 (q, J= 31.8Hz), 129.5, 127.3, 126.3, 126.2, 124.7, 124.3, 124.2 (q, J=272.7Hz), 120.0, 117.7 (dd, J=9.2), 117.7 (d, J=4.7Hz), 117.4, 115.7 ,110.8(d,J=3.2Hz).61.5,61.3,19.5,14.3,14.3,13.3.HRMS: [M+H] + calculated for C 27 H 23 F 3 NO 5 + =498.1523, found: 498.1508.
实施例12Example 12
在氮气条件下,将3-芳基苯并噁嗪化合物1a(0.20mmol),重氮化合物2b(0.44mmol),[Cp*RhCl2]2(4mol%),AgSbF6(16mol%),MesCOOH(0.4mmol)和溶剂DCE(2.0mL)加入到10mL密封管中,在80度的反应模块中反应10h,反应结束后减压除去溶剂,用硅胶柱分离得到目标产物5,12-二甲基异喹啉[2,1,8-mna]吩恶嗪-4,13-二羧酸二戊酯(3ab),所有洗脱剂为石油醚乙酸乙酯和二氯甲烷按30:1:1的比例配制而成。产物数据表征:黄色液体,96%的产率。产物的核磁图谱如图23和24所示,1H NMR(400MHz,CDCl3)δ7.23–7.15(m,1H),7.09(t,J=7.9Hz,1H),7.03(t,J=7.4Hz,1H),6.99–6.95(m,1H),6.91(dd,J=7.7,1.6Hz,1H),6.84(d,J=7.7Hz,1H),6.79–6.67(m,1H),4.36(t,J=6.7Hz,3H),4.31(t,J=6.8Hz,3H),2.32(s,3H),2.31(s,3H),1.80–1.73(m,4H),1.43–1.36(m,8H),0.95–0.90(m,6H).13C NMR(101MHz,CDCl3)δ168.9,168.0,150.7,138.6,137.2,131.3,130.3,129.2,128.4,127.8,125.9,125.8,125.1,124.0,123.7,120.2,119.7,117.2,116.9,115.3,65.4,65.3,28.5,28.4,28.3,28.3,22.4,22.4,19.1,14.1,14.1,13.3.HRMS:[M+H]+calculated for C32H36NO5 +=514.2588,found:514.2577.Under nitrogen, 3-
实施例13Example 13
在氮气条件下,将3-芳基苯并噁嗪化合物1a(0.20mmol),重氮化合物2c(0.44mmol),[Cp*RhCl2]2(4mol%),AgSbF6(16mol%),MesCOOH(0.4mmol)和溶剂DCE(2.0mL)加入到10mL密封管中,在80度的反应模块中反应10h,反应结束后减压除去溶剂,用硅胶柱分离得到目标产物5,12-二甲基异喹啉[2,1,8-mna]吩恶嗪-4,13-二羧酸二异丙酯(3ac),所有洗脱剂为石油醚乙酸乙酯和二氯甲烷按30:1:1的比例配制而成。产物数据表征:红色液体,80%的产率。产物的核磁图谱如图25和26所示,1H NMR(400MHz,CDCl3)δ7.24(d,J=23.1Hz,1H),7.16–7.07(m,1H),7.02–6.89(m,3H),6.84–6.80(m,1H),6.75–6.70(m,1H),5.39–5.28(m,2H),2.33(s,6H),1.44–1.36(m,12H).13C NMR(101MHz,CD Cl3)δ168.1,167.2,150.5,137.9,137.1,131.1,130.2,129.1,128.2,127.7,125.6,125.4,125.0,123.9,119.9,119.5,117.1,114.9,68.8,68.7,22.0,21.9,18.9,13.1.HRMS:[M+H]+calculated for C28H28NO5 +=458.1962,found:458.1943.Under nitrogen, 3-
实施例14Example 14
在氮气条件下,将3-芳基苯并噁嗪化合物1a(0.20mmol),重氮化合物2d(0.44mmol),[Cp*RhCl2]2(4mol%),AgSbF6(16mol%),MesCOOH(0.4mmol)和溶剂DCE(2.0mL)加入到10mL密封管中,在80度的反应模块中反应10h,反应结束后减压除去溶剂,用硅胶柱分离得到目标产物5,12-二乙基异喹啉[2,1,8-mna]吩恶嗪-4,13-二羧酸二甲酯(3ad),所有洗脱剂为石油醚乙酸乙酯和二氯甲烷按30:1:1的比例配制而成。产物数据表征:黄色液体,60%的产率。产物的核磁图谱如图27和28所示,1H NMR(400MHz,CDCl3)δ7.17(t,J=8.5Hz,1H),7.13–7.08(m,1H),7.07–7.03(m,1H),7.01–6.95(m,1H),6.94–6.88(m,2H),6.70(d,J=7.1Hz,1H),3.94(s,3H),3.91(s,3H),2.81–2.74(m,2H),2.69(dd,J=8.7,6.8Hz,2H),1.71–1.52(m,4H),0.99(t,J=7.4Hz,3H),0.94(t,J=7.3Hz,3H).13C NMR(101MHz,CDCl3)δ169.3,168.5,150.7,142.8,137.4,132.2,131.6,130.9,129.0,128.5,127.8,125.7,124.9,124.1,123.1,120.8,119.3,118.2,117.3,115.6,52.2,52.2,32.4,30.2,23.9,22.8,14.5,13.9.HRMS:[M+H]+calculated for C28H28NO5 +=458.1962,found:458.1949.Under nitrogen, 3-
实施例15Example 15
在氮气条件下,将3-芳基苯并噁嗪化合物1a(0.20mmol),重氮化合物2e(0.44mmol),[Cp*RhCl2]2(4mol%),AgSbF6(16mol%),MesCOOH(0.4mmol)和溶剂DCE(2.0mL)加入到10mL密封管中,在80度的反应模块中反应10h,反应结束后减压除去溶剂,用硅胶柱分离得到目标产物5,12-二丙基异喹啉[2,1,8-mna]吩恶嗪-4,13-二羧酸二甲酯(3ae),所有洗脱剂为石油醚乙酸乙酯和二氯甲烷按30:1:1的比例配制而成。产物数据表征:黄色液体,63%的产率。产物的核磁图谱如图29和30所示,1H NMR(400MHz,CDCl3)δ7.19(dd,J=8.6,0.7Hz,1H),7.13–7.09(m,1H),7.07–7.03(m,1H),7.00–6.96(m,1H),6.93(dd,J=7.8,1.5Hz,0H),6.90(dd,J=7.9,1.4Hz,1H),6.70(dd,J=7.2,0.7Hz,1H),3.94(s,3H),3.91(s,3H),2.82–2.79(m,2H),2.76–2.65(m,2H),1.65–1.51(m,4H),1.48–1.39(m,2H),1.37–1.32(m,2H),0.96(t,J=7.3Hz,3H),0.85(t,J=7.3Hz,3H).13C NMR(101MHz,CDCl3)δ169.3,168.5,150.7,143.1,137.4,132.2,131.6,131.2,129.1,128.5,127.7,125.7,124.8,124.1,123.0,120.7,119.3,118.1,117.2,115.5,52.2,52.1,32.7,31.5,30.1,27.9,23.0,22.4,14.1,13.8.HRMS:[M+H]+calculated for C30H32NO5 +=486.2275,found:486.2266.Under nitrogen, 3-
以上所述仅为本发明的较佳实施例而已,并不用以限制本发明,凡在本发明的精神和原则之内,所作的任何修改、等同替换、改进等,均应包含在本发明的保护范围之内。The above descriptions are only preferred embodiments of the present invention, and are not intended to limit the present invention. Any modification, equivalent replacement, improvement, etc. made within the spirit and principle of the present invention shall be included in the scope of the present invention. within the scope of protection.
Claims (10)
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202210557098.5A CN114716454A (en) | 2022-05-19 | 2022-05-19 | Nitrogen and oxa condensed ring aromatic hydrocarbon and synthetic method thereof |
| CN202310563635.1A CN116836180B (en) | 2022-05-19 | 2023-05-18 | Nitrogen and oxygen hetero-polycyclic aromatic hydrocarbon and synthetic method and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202210557098.5A CN114716454A (en) | 2022-05-19 | 2022-05-19 | Nitrogen and oxa condensed ring aromatic hydrocarbon and synthetic method thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN114716454A true CN114716454A (en) | 2022-07-08 |
Family
ID=82231236
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202210557098.5A Withdrawn CN114716454A (en) | 2022-05-19 | 2022-05-19 | Nitrogen and oxa condensed ring aromatic hydrocarbon and synthetic method thereof |
| CN202310563635.1A Active CN116836180B (en) | 2022-05-19 | 2023-05-18 | Nitrogen and oxygen hetero-polycyclic aromatic hydrocarbon and synthetic method and application thereof |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202310563635.1A Active CN116836180B (en) | 2022-05-19 | 2023-05-18 | Nitrogen and oxygen hetero-polycyclic aromatic hydrocarbon and synthetic method and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (2) | CN114716454A (en) |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN110167940A (en) * | 2017-01-30 | 2019-08-23 | 默克专利有限公司 | Material for organic electroluminescence device |
| CN112778307A (en) * | 2021-01-21 | 2021-05-11 | 北京八亿时空液晶科技股份有限公司 | Carbazole derivative and application thereof |
| WO2021122538A1 (en) * | 2019-12-18 | 2021-06-24 | Merck Patent Gmbh | Aromatic compounds for organic electroluminescent devices |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114478576A (en) * | 2022-02-17 | 2022-05-13 | 河南农业大学 | Synthetic method of spiroheterocyclic compound |
-
2022
- 2022-05-19 CN CN202210557098.5A patent/CN114716454A/en not_active Withdrawn
-
2023
- 2023-05-18 CN CN202310563635.1A patent/CN116836180B/en active Active
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN110167940A (en) * | 2017-01-30 | 2019-08-23 | 默克专利有限公司 | Material for organic electroluminescence device |
| WO2021122538A1 (en) * | 2019-12-18 | 2021-06-24 | Merck Patent Gmbh | Aromatic compounds for organic electroluminescent devices |
| CN112778307A (en) * | 2021-01-21 | 2021-05-11 | 北京八亿时空液晶科技股份有限公司 | Carbazole derivative and application thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| CN116836180A (en) | 2023-10-03 |
| CN116836180B (en) | 2025-03-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN112300201B (en) | Synthesis and preparation method of trimeric indenyl coumarin-corrole-porphyrin quaternary system star-shaped compound | |
| CN111377977A (en) | A kind of 4,7-diarylthieno[2,3-d]pyridazine ring metal iridium complex and preparation method thereof | |
| CN112062756B (en) | Stenhouse donor-acceptor adducts of mevalonate activated furan and 3-pyridylethylamine and methods of synthesizing the same | |
| CN107540848A (en) | A kind of preparation and its application of efficiently out-phase hydrogen-bond donor MOF catalyst | |
| CN113683601A (en) | A kind of diazobenzofluoranthene compound and its preparation method and application | |
| CN114478576A (en) | Synthetic method of spiroheterocyclic compound | |
| CN109748916B (en) | Compound with azaspirofluorene and aryl ketone as cores, preparation method thereof and application thereof in OLED | |
| CN112442054B (en) | A kind of preparation method of trimeric indenyl carrole-porphyrin-BODIPY star compound | |
| CN114716454A (en) | Nitrogen and oxa condensed ring aromatic hydrocarbon and synthetic method thereof | |
| CN106111190A (en) | A kind of chirality biaryl skeleton pyridoxamine class catalyst and synthetic method thereof and application | |
| CN109575046A (en) | A kind of four aryl of dithienothiophene replace and double luxuriant and rich with fragrance condensed compounds and preparation | |
| CN113292585B (en) | BODIPY-benzothiadiazole-porphyrin-carbazole quaternary system linear compound and preparation method thereof | |
| CN114573514B (en) | Bridged bisbenzimidazole salt, and preparation method and application thereof | |
| CN116514875A (en) | Triptycene-like platinum complex and preparation method thereof | |
| CN112479976B (en) | Organic compound containing benzanthracene, preparation method and application thereof | |
| CN118724918B (en) | A pyridine aromatic amide luminescent macrocyclic compound and its preparation method and application | |
| CN106167708A (en) | 2,3 imidodicarbonic diamide benzophenanthrene discotic liquid-crystalline molecules and preparation method thereof | |
| CN116574117B (en) | Method for synthesizing asymmetric substitution reaction intermediate | |
| CN120441417B (en) | Helical chiral polycyclic aromatic hydrocarbon compounds, open-shell compounds, and preparation methods and applications thereof | |
| CN112062771A (en) | Synthesis and Application of Fluorescent Probes for Multi-Type Organelles Containing Heteroatom Polycyclic Aromatic Hydrocarbons | |
| CN114874105B (en) | Preparation method of visible light and water promoted homoallylic amine compound | |
| CN112062775B (en) | An organic electroluminescent material and its preparation method and application | |
| CN115232161B (en) | A trimeric indenyl-BODIPY-perylene diimide ternary system molecule and preparation method thereof | |
| CN104945346B (en) | Bay-position oxygen-intercalation aza-heptatomic ring 3, 4, 9, 10-perylene tetracarboxylic acid butyl acetate and synthesis method thereof | |
| CN110143968B (en) | A kind of dithienophthalimide derivative and its preparation method and application |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| WW01 | Invention patent application withdrawn after publication | ||
| WW01 | Invention patent application withdrawn after publication |
Application publication date: 20220708 |