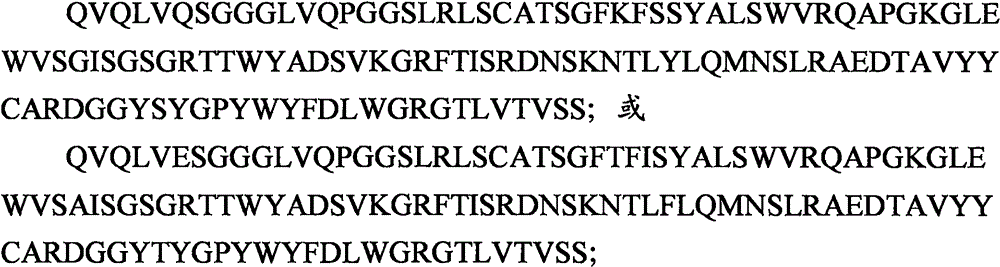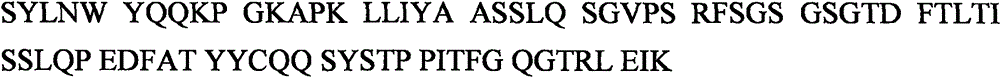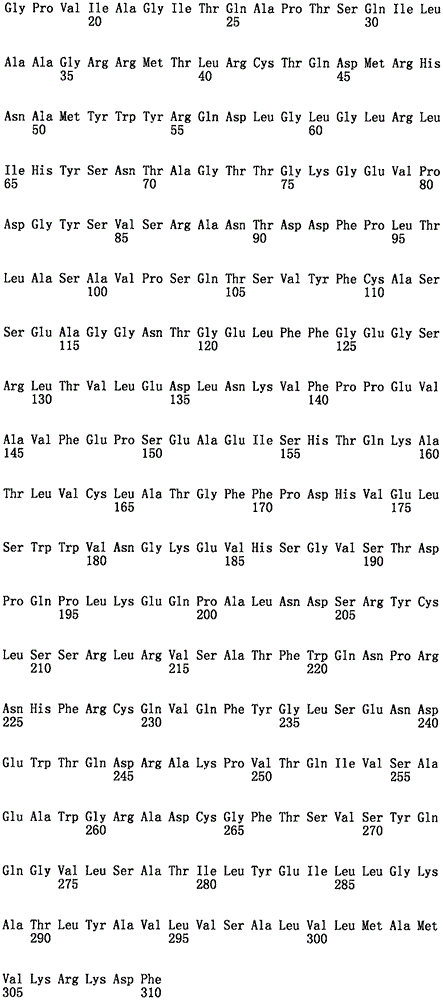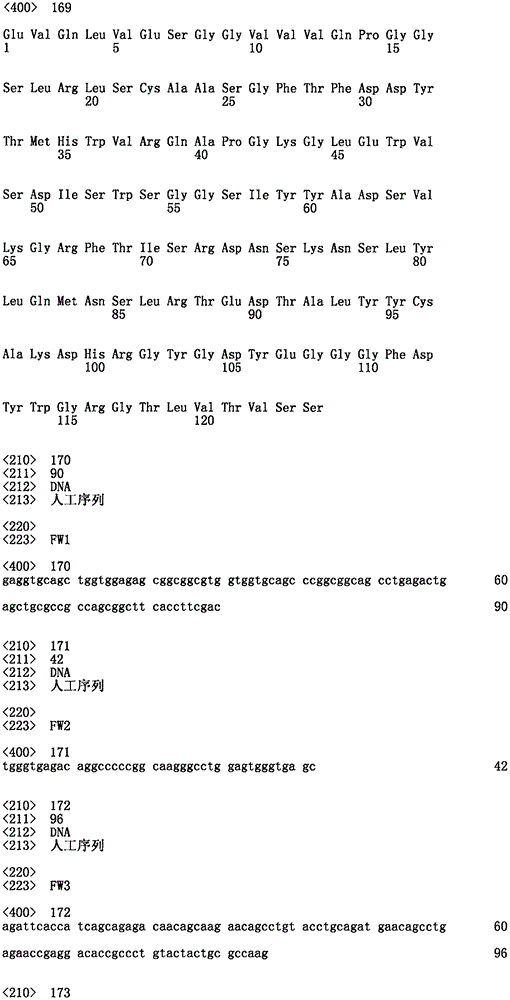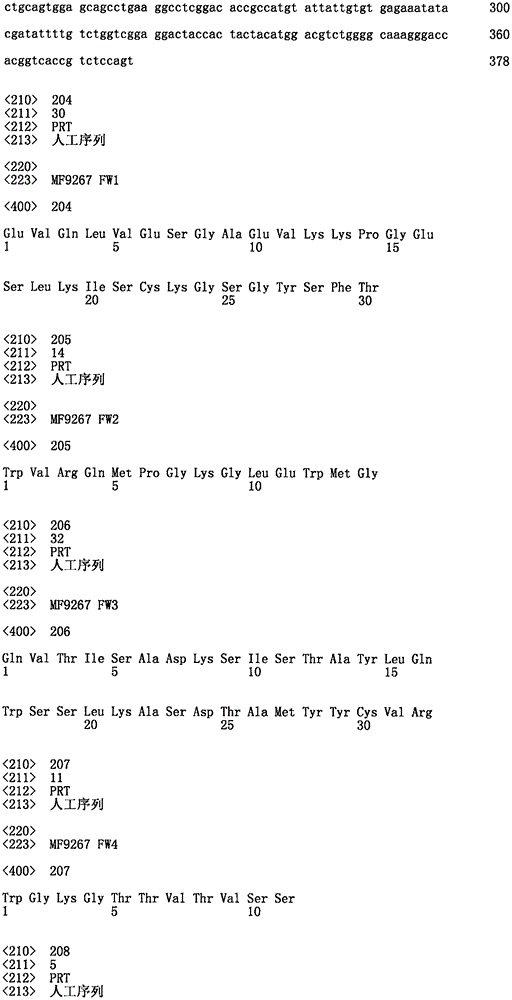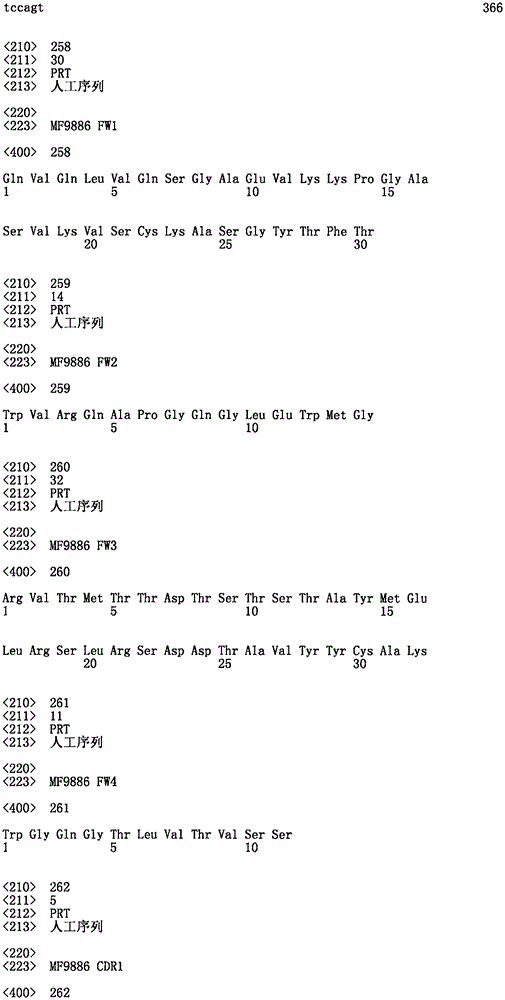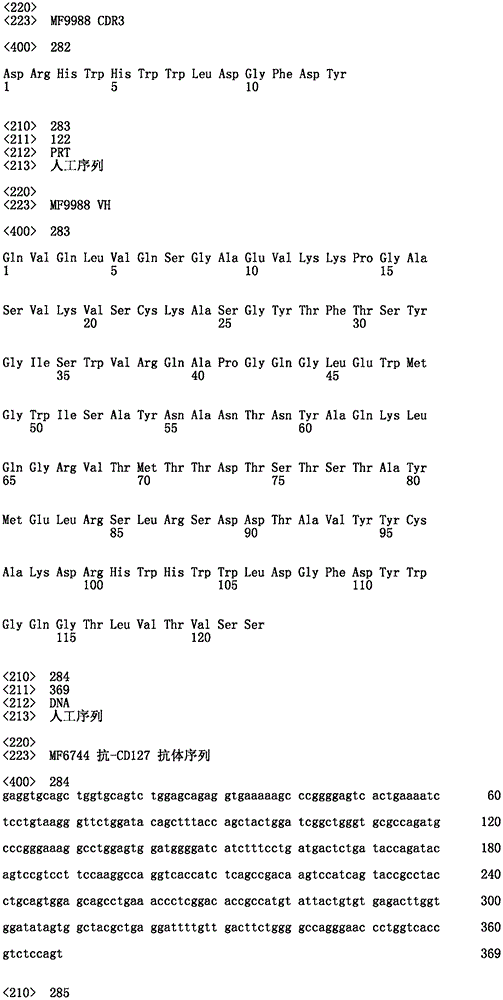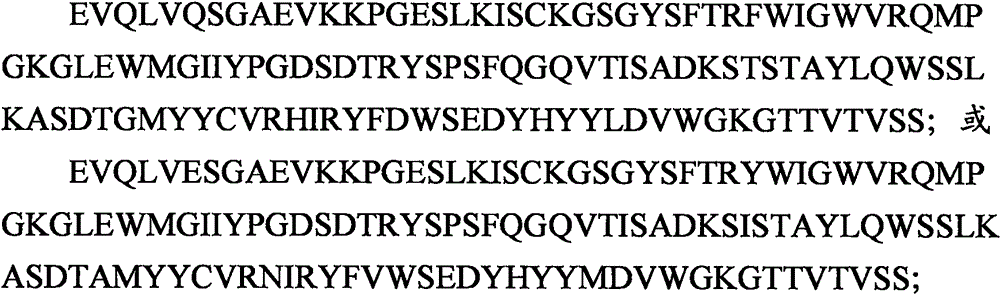CN114605546A - CD3 binding molecules - Google Patents
CD3 binding molecules Download PDFInfo
- Publication number
- CN114605546A CN114605546A CN202210274738.1A CN202210274738A CN114605546A CN 114605546 A CN114605546 A CN 114605546A CN 202210274738 A CN202210274738 A CN 202210274738A CN 114605546 A CN114605546 A CN 114605546A
- Authority
- CN
- China
- Prior art keywords
- antibody
- variable region
- chain variable
- light chain
- heavy chain
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2803—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily
- C07K16/2809—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily against the T-cell receptor (TcR)-CD3 complex
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/21—Immunoglobulins specific features characterized by taxonomic origin from primates, e.g. man
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/24—Immunoglobulins specific features characterized by taxonomic origin containing regions, domains or residues from different species, e.g. chimeric, humanized or veneered
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/31—Immunoglobulins specific features characterized by aspects of specificity or valency multispecific
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/52—Constant or Fc region; Isotype
- C07K2317/526—CH3 domain
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/56—Immunoglobulins specific features characterized by immunoglobulin fragments variable (Fv) region, i.e. VH and/or VL
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/56—Immunoglobulins specific features characterized by immunoglobulin fragments variable (Fv) region, i.e. VH and/or VL
- C07K2317/565—Complementarity determining region [CDR]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/73—Inducing cell death, e.g. apoptosis, necrosis or inhibition of cell proliferation
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/92—Affinity (KD), association rate (Ka), dissociation rate (Kd) or EC50 value
Landscapes
- Health & Medical Sciences (AREA)
- Immunology (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Genetics & Genomics (AREA)
- Molecular Biology (AREA)
- Biophysics (AREA)
- Biochemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Peptides Or Proteins (AREA)
Abstract
本申请涉及CD3结合分子。本发明有关于对人类CD3有特异性的重链可变区、结合域及抗体,及CD3结合蛋白。本发明进一步有关于本发明的CD3结合蛋白,优选为抗体于治疗癌症或自体免疫疾病的用途。
The present application relates to CD3 binding molecules. The present invention relates to heavy chain variable regions, binding domains and antibodies specific for human CD3, and CD3 binding proteins. The present invention further relates to the use of the CD3 binding proteins of the present invention, preferably antibodies, for the treatment of cancer or autoimmune diseases.
Description
本申请是申请号为202080019367.1的发明名称为“CD3结合分子”的中国专利申请的分案申请,原申请是2020年03月27日提交的PCT国际申请PCT/NL2020/050214于2021年09月07日进入中国国家阶段的申请。This application is a divisional application of a Chinese patent application entitled "CD3 binding molecule" with the application number 202080019367.1. The original application is the PCT international application PCT/NL2020/050214 filed on March 27, 2020 and filed on September 07, 2021 Applications for entering the Chinese national phase from Japan.
技术领域technical field
本发明有关于抗体领域,特别涉及治疗抗体领域。此类抗体可用于治疗人类。更具体地,本发明涉及用于治疗肿瘤的抗体且优选为双特异性或多特异性抗体。The present invention relates to the field of antibodies, particularly to the field of therapeutic antibodies. Such antibodies can be used to treat humans. More specifically, the present invention relates to antibodies and preferably bispecific or multispecific antibodies for use in the treatment of tumors.
背景技术Background technique
结合人类CD3的单株抗体是第一批开发用于人类治疗用途的抗体。单株CD3结合抗体典型用于其的免疫抑止性质,例如于移植排斥方面。针对T细胞上的CD3及针对癌细胞上的表面标靶抗原为双特异性的抗体,能够连接任何种类的T细胞与癌细胞,独立于T细胞受体特异性、共刺激或肽抗原呈现。这些双特异性T细胞衔接(T-cell engaging)抗体于治疗各种癌症及赘瘤性生长方面显现出很大的希望。Monoclonal antibodies that bind human CD3 were the first antibodies developed for therapeutic use in humans. Monoclonal CD3-binding antibodies are typically used for their immunosuppressive properties, eg, in transplant rejection. Bispecific antibodies directed against CD3 on T cells and against surface target antigens on cancer cells, capable of linking any kind of T cells to cancer cells, independent of T cell receptor specificity, costimulation or peptide antigen presentation. These bispecific T-cell engaging antibodies show great promise in the treatment of various cancers and neoplastic growths.
本发明的目的是要提供新的抗体,其具有CD3结合性质,在种类方面不一定是数量的性质,具有改良的特征,有相对低的亲和力,有较高的细胞毒性,该抗体适合用于T细胞及效应子细胞衔接的肿瘤免疫学应用,以及相反地,要提供新的抗体其具有CD3结合,其有相对高的亲和力,有较低的细胞毒性,该抗体适合用于T细胞及效应子细胞向下调节的自体免疫应用。本发明进一步的目的是要提供T细胞衔接CD3结合蛋白及抗体,其具有以上的结合至少一另外的膜缔合分子的性质。The object of the present invention is to provide novel antibodies, which have CD3 binding properties, not necessarily quantitative properties in terms of species, have improved characteristics, have relatively low affinity, and have high cytotoxicity, which are suitable for use in Tumor immunology applications of T cell and effector cell engagement, and conversely, to provide new antibodies that bind to CD3, have relatively high affinity, and have low cytotoxicity, suitable for use in T cells and effector Autoimmune applications of daughter cell downregulation. A further object of the present invention is to provide T cell engaging CD3 binding proteins and antibodies having the above properties of binding to at least one additional membrane-associated molecule.
发明内容SUMMARY OF THE INVENTION
本发明提供一种会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括一CDR1、CDR2及CDR3,该CDR1、CDR2及CDR3包括下列氨基酸序列:The present invention provides an antigen-binding protein that binds to human CD3, preferably an antibody, comprising an antibody variable domain, the antibody variable domain comprising a heavy chain variable region and a light chain variable region, wherein the The heavy chain variable region includes a CDR1, CDR2, and CDR3, and the CDR1, CDR2, and CDR3 include the following amino acid sequences:
CDR1:SFGISCDR1: SFGIS
CDR2:GFIPVLGTANYAQKFQGCDR2: GFIPVLGTANYAQKFQG
CDR3:RGNWNPFDP;或CDR3: RGNWNPFDP; or
包括下列氨基酸序列:Includes the following amino acid sequences:
CDR1:SX1TFTIS;CDR1: SX 1 TFTIS;
CDR2:GIIPX2FGTITYAQKFQG;CDR2: GIIPX 2 FGTITYAQKFQG;
CDR3:RGNWNPFDP;CDR3: RGNWNPFDP;
其中in
X1=K或R;X 1 =K or R;
X2=L或I。X 2 =L or I.
在优选的具体例中X1=K;并且X2=L;In a preferred embodiment, X 1 =K; and X 2 =L;
在另一优选的具体例中,X1=R;并且X2=I。In another preferred embodiment, X 1 =R; and X 2 =I.
在优选具体例中,本发明提供一种会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括一CDR1、CDR2及CDR3,该CDR1、CDR2及CDR3包括下列氨基酸序列:In a preferred embodiment, the present invention provides an antigen-binding protein that binds to human CD3, preferably an antibody, comprising an antibody variable domain, the antibody variable domain comprising a heavy chain variable region and a light chain Variable region, wherein the heavy chain variable region includes a CDR1, CDR2 and CDR3, the CDR1, CDR2 and CDR3 include the following amino acid sequence:
CDR1:SKTLTIS;CDR1: SKTLTIS;
CDR2:GIIPIFGSITYAQKFQD;CDR2: GIIPIFGSITYAQKFQD;
CDR3:RGNWNPFDP;或CDR3: RGNWNPFDP; or
包括下列氨基酸序列:Includes the following amino acid sequences:
CDR1:GSGIS;CDR1: GSGIS;
CDR2:GFIPFFGSANYAQKFRD;CDR2: GFIPFFGSANYAQKFRD;
CDR3:RGNWNPX13DP;CDR3: RGNWNPX 13DP ;
其中in
X13=L或F。X 13 =L or F.
本发明进一步提供一种会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括氨基酸序列The present invention further provides an antigen-binding protein that binds human CD3, preferably an antibody, comprising an antibody variable domain, the antibody variable domain comprising a heavy chain variable region and a light chain variable region, wherein The heavy chain variable region includes the amino acid sequence
于此类CDRs以外的一个或多个位置处具有0-10个、优选0-5个氨基酸变异、插入、缺失、取代、加入或其组合。There are 0-10, preferably 0-5 amino acid variations, insertions, deletions, substitutions, additions or combinations thereof at one or more positions other than such CDRs.
进一步提供一种会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括一CDR1、CDR2及CDR3,该CDR1、CDR2及CDR3包括下列氨基酸序列:Further provide an antigen-binding protein that can bind to human CD3, preferably an antibody, comprising an antibody variable domain, the antibody variable domain comprising a heavy chain variable region and a light chain variable region, wherein the heavy chain variable region is provided. The chain variable region includes a CDR1, CDR2, and CDR3, and the CDR1, CDR2, and CDR3 include the following amino acid sequences:
CDR1:RX3WIG;CDR1: RX 3 WIG;
CDR2:IIYPGDSDTRYSPSFQG;CDR2: IIYPGDSDTRYSPSFQG;
CDR3:X4IRYFX5WSEDYHYYX6DV;CDR3: X 4 IRYFX 5 WSEDYHYYX 6 DV;
其中in
X3=F或Y;X 3 =F or Y;
X4=H或N;X 4 =H or N;
X5=D或V;X 5 =D or V;
X6=L或M。X 6 =L or M.
在一具体例中X3=F;X4=H;X5=D;及X6=L。在另一具体例中X3=Y;X4=N;X5=V;及X6=M。In a specific example X 3 =F; X 4 =H; X 5 =D; and X 6 =L. In another specific example, X 3 =Y; X 4 =N; X 5 =V; and X 6 =M.
本发明进一步提供一种会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括氨基酸序列The present invention further provides an antigen-binding protein that binds human CD3, preferably an antibody, comprising an antibody variable domain, the antibody variable domain comprising a heavy chain variable region and a light chain variable region, wherein The heavy chain variable region includes the amino acid sequence
于此类CDRs以外的一个或多个位置处具有0-10个、优选0-5个氨基酸变异、插入、缺失、取代、加入或其组合。There are 0-10, preferably 0-5 amino acid variations, insertions, deletions, substitutions, additions or combinations thereof at one or more positions other than such CDRs.
进一步提供一种会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括一CDR1、CDR2及CDR3,该CDR1、CDR2及CDR3包括下列氨基酸序列:Further provide an antigen-binding protein that can bind to human CD3, preferably an antibody, comprising an antibody variable domain, the antibody variable domain comprising a heavy chain variable region and a light chain variable region, wherein the heavy chain variable region is provided. The chain variable region includes a CDR1, CDR2, and CDR3, and the CDR1, CDR2, and CDR3 include the following amino acid sequences:
CDR1:SYALS;CDR1:SYALS;
CDR2:GISGSGRTTWYADSVKG;CDR2:GISGSGRTTWYADSVKG;
CDR3:DGGYSYGPYWYFDL。CDR3: DGGYSYGPYWYFDL.
进一步提供一种会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括一CDR1、CDR2及CDR3,该CDR1、CDR2及CDR3包括下列氨基酸序列:Further provide an antigen-binding protein that can bind to human CD3, preferably an antibody, comprising an antibody variable domain, the antibody variable domain comprising a heavy chain variable region and a light chain variable region, wherein the heavy chain variable region is provided. The chain variable region includes a CDR1, CDR2, and CDR3, and the CDR1, CDR2, and CDR3 include the following amino acid sequences:
CDR1:SYALS;CDR1:SYALS;
CDR2:AISGSGRTTWYADSVKG;CDR2: AISGSGRTTWYADSVKG;
CDR3:DGGYTYGPYWYFDL。CDR3: DGGYTYGPYWYFDL.
进一步提供一种会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括氨基酸序列Further provide an antigen-binding protein that can bind to human CD3, preferably an antibody, comprising an antibody variable domain, the antibody variable domain comprising a heavy chain variable region and a light chain variable region, wherein the heavy chain variable region is provided. chain variable region including amino acid sequence
于此类CDRs以外的一个或多个位置处具有0-10个、优选0-5个氨基酸变异、插入、缺失、取代、加入或其组合。There are 0-10, preferably 0-5 amino acid variations, insertions, deletions, substitutions, additions or combinations thereof at one or more positions other than such CDRs.
还提供一种会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括一CDR1、CDR2及CDR3,该CDR1、CDR2及CDR3包括下列氨基酸序列:Also provided is an antigen binding protein that binds human CD3, preferably an antibody, comprising an antibody variable domain comprising a heavy chain variable region and a light chain variable region, wherein the heavy chain variable region is The chain variable region includes a CDR1, CDR2, and CDR3, and the CDR1, CDR2, and CDR3 include the following amino acid sequences:
CDR1:DYTMH;CDR1: DYTMH;
CDR2:DISWSSGSIGYADSVKG;CDR2: DISWSSGSIGYADSVKG;
CDR3:DHRGYGDYEGGGFDY。CDR3: DHRGYGDYEGGGFDY.
还提供一种会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括一CDR1、CDR2及CDR3,该CDR1、CDR2及CDR3包括下列氨基酸序列:Also provided is an antigen binding protein that binds human CD3, preferably an antibody, comprising an antibody variable domain comprising a heavy chain variable region and a light chain variable region, wherein the heavy chain variable region is The chain variable region includes a CDR1, CDR2, and CDR3, and the CDR1, CDR2, and CDR3 include the following amino acid sequences:
CDR1:DYTMH;CDR1: DYTMH;
CDR2:DISWSX7GX8X9X10YADSVKG;CDR2: DISSWX 7 GX 8 X 9 X 10 YADSVKG;
CDR3:DHX11GYGDYEGGGFDX12;CDR3: DHX 11 GYGDYEGGGFDX 12 ;
其中in
X7=S或G;X 7 =S or G;
X8=S或T;X 8 =S or T;
X9=I或T;X 9 =I or T;
X10=G或Y;X 10 =G or Y;
X11=R或M;X 11 =R or M;
X12=H或Y。X 12 =H or Y.
在一个具体例中,X7、X8、X9和X10为S、S、I和G,并且X11和X12为R和H。在另一具体例中,X7、X8、X9和X10为G、S、I和Y,并且X11和X12为R和Y。在另一具体例中,X7、X8、X9和X10为S、T、T和G,并且X11和X12为M和Y。In a specific example, X 7 , X 8 , X 9 and X 10 are S, S, I and G, and X 11 and X 12 are R and H. In another specific example, X 7 , X 8 , X 9 and X 10 are G, S, I and Y, and X 11 and X 12 are R and Y. In another specific example, X 7 , X 8 , X 9 and X 10 are S, T, T and G, and X 11 and X 12 are M and Y.
进一步提供一种会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括氨基酸序列Further provide an antigen-binding protein that can bind to human CD3, preferably an antibody, comprising an antibody variable domain, the antibody variable domain comprising a heavy chain variable region and a light chain variable region, wherein the heavy chain variable region is provided. chain variable region including amino acid sequence
于此类CDRs以外的一个或多个位置处具有0-10个、优选0-5个氨基酸变异、插入、缺失、取代、加入或其组合。There are 0-10, preferably 0-5 amino acid variations, insertions, deletions, substitutions, additions or combinations thereof at one or more positions other than such CDRs.
一种本发明的抗原结合蛋白,优选为一抗体,的轻链可变区优选包括一共同的轻链可变区。该共同的轻链可变区优选包括一IgVκ1-39轻链可变区。该轻链可变区优选为一生殖系列IgVκ1-39*01可变区。该轻链可变区优选包括κ轻链IgVκ1-39*01/IGJκ1*01或IgVκ1-39*01/IGJκ5*01。在一具体例中该轻链可变区包括人类生殖系列κ轻链IgVκ1-39*01/IGJκ1*01或IgVκ1-39*01/IGJκ5*01。该轻链可变区优选包括氨基酸序列The light chain variable region of an antigen binding protein of the present invention, preferably an antibody, preferably includes a common light chain variable region. The common light chain variable region preferably includes an IgVκ1-39 light chain variable region. The light chain variable region is preferably a germline IgVκ1-39*01 variable region. The light chain variable region preferably comprises a kappa light chain IgVκ1-39*01/IGJκ1*01 or IgVκ1-39*01/IGJκ5*01. In one embodiment, the light chain variable region comprises a human germline kappa light chain IgVκ1-39*01/IGJκ1*01 or IgVκ1-39*01/IGJκ5*01. The light chain variable region preferably comprises an amino acid sequence
具有0-5个氨基酸变异、插入、缺失、取代、加入或其的组合。With 0-5 amino acid variations, insertions, deletions, substitutions, additions or combinations thereof.
该抗原结合蛋白,优选为一种抗体,优选为一种双特异性或多特异性抗体。The antigen binding protein is preferably an antibody, preferably a bispecific or multispecific antibody.
该抗体优选包括一种如本文所示的会结合人类CD3的H/L链组合以及一种会结合肿瘤抗原的H/L链组合。该会结合肿瘤抗原的H/L链组合优选结合人类BCMA、CD19、CD20、CD30、CD33、CD38、CD44、CD123、CD138、CEA、CLEC12A、CS-1、EGFR、EGFRvIII、EPCAM、DLL3、LGR5、MSLN、FOLR1、FOLR3、HER2、HM1.24、MCSP、PD-L1、PSMA蛋白或其的一变异体,在优选具体例中EGFR、PD-L1或CLEC12A。The antibody preferably includes an H/L chain combination that binds human CD3 as shown herein and an H/L chain combination that binds a tumor antigen. The H/L chain combination that binds tumor antigens preferably binds human BCMA, CD19, CD20, CD30, CD33, CD38, CD44, CD123, CD138, CEA, CLEC12A, CS-1, EGFR, EGFRvIII, EPCAM, DLL3, LGR5, MSLN, FOLR1, FOLR3, HER2, HM1.24, MCSP, PD-L1, PSMA protein or a variant thereof, in a preferred embodiment, EGFR, PD-L1 or CLEC12A.
该抗体、双特异性或多特异性抗体优选为一种人类或人化抗体。The antibody, bispecific or multispecific antibody is preferably a human or humanized antibody.
该双特异性或多特异性抗体优选包括具有兼容的异质二聚合域的二个不同的免疫球蛋白重链。该兼容的异质二聚合域优选为兼容的免疫球蛋白重链CH3异质二聚合域。The bispecific or multispecific antibody preferably comprises two different immunoglobulin heavy chains with compatible heterodimeric domains. The compatible heterodimerization domain is preferably a compatible immunoglobulin heavy chain CH3 heterodimerization domain.
该双特异性或多特异性抗体优选为具有突变型CH2和/或下部铰链域的IgG抗体以使得该双特异性或多特异性IgG抗体与Fc-γ受体的交互作用减少。该突变型CH2和/或下部铰链域优选包括位置235和/或236(根据EU编号)的氨基取代,优选为L235G和/或G236R取代。The bispecific or multispecific antibody is preferably an IgG antibody with a mutated CH2 and/or lower hinge domain such that the interaction of the bispecific or multispecific IgG antibody with the Fc-gamma receptor is reduced. The mutant CH2 and/or lower hinge domain preferably comprises amino substitutions at positions 235 and/or 236 (according to EU numbering), preferably L235G and/or G236R substitutions.
该双特异性或多特异性抗体优选包括一共同的轻链。The bispecific or multispecific antibodies preferably comprise a common light chain.
本发明进一步提供一种如本文所示的抗原结合蛋白或抗体,供用于治疗有需要的一个体。该个体优选有癌症或要治疗癌症。一种具有MF8057、MF8058、MF8078或MF8508的CDRs和/或VH序列,或其具有0-10个氨基酸取代、变异、插入、加入或缺失的变异体,的抗原结合蛋白或抗体优选用于治疗,特别是用于一种包括局部投予和/或局部释放该抗原结合蛋白或抗体的治疗。一种具有MF9249、MF9267、MF8397的CDRs和/或VH序列,或其具有0-10个氨基酸取代、变异、插入、加入或缺失的变异体,的抗原结合蛋白或抗体优选用于治疗有过度活化的免疫系统,例如自体免疫疾病的一个体。The present invention further provides an antigen binding protein or antibody as shown herein for use in the treatment of an individual in need thereof. The individual preferably has cancer or is to be treated for cancer. A kind of CDRs and/or VH sequence with MF8057, MF8058, MF8078 or MF8508, or its variant with 0-10 amino acid substitution, variation, insertion, addition or deletion, the antigen binding protein or antibody is preferably used for treatment, In particular for a treatment involving local administration and/or local release of the antigen binding protein or antibody. A kind of CDRs and/or VH sequence with MF9249, MF9267, MF8397, or its variant with 0-10 amino acid substitution, variation, insertion, addition or deletion, the antigen binding protein or antibody is preferably used for the treatment of hyperactivation immune system, such as an individual with an autoimmune disease.
除另有其他明确规定外,否则本发明的抗体优选为一种双特异性抗体。该双特异性抗体优选至少结合人类CD3。此外,该双特异性抗体优选与优先表现于人类肿瘤细胞上的至少一表面分子结合。在优选具体例中,该双特异性抗体结合BCMA、CD19、CD20、CD30、CD33、CD38、CD44、CD123、CD138、CEA、CLEC12A、CS-1、EGFR、EGFRvIII、EPCAM、DLL3、LGR5、MSLN、PD-L1、FOLR1、FOLR3、HER2、HM1.24、MCSP或PSMA。在更优选的具体例中,该双特异性抗体与EGFR或CLEC12A结合。在更优选的具体例中,该多特异性抗体与EGFR及PD-L1结合。Unless expressly stated otherwise, the antibody of the invention is preferably a bispecific antibody. The bispecific antibody preferably binds at least human CD3. Furthermore, the bispecific antibody preferably binds to at least one surface molecule preferentially expressed on human tumor cells. In a preferred embodiment, the bispecific antibody binds to BCMA, CD19, CD20, CD30, CD33, CD38, CD44, CD123, CD138, CEA, CLEC12A, CS-1, EGFR, EGFRvIII, EPCAM, DLL3, LGR5, MSLN, PD-L1, FOLR1, FOLR3, HER2, HM1.24, MCSP or PSMA. In a more preferred embodiment, the bispecific antibody binds to EGFR or CLEC12A. In a more preferred embodiment, the multispecific antibody binds to EGFR and PD-L1.
本发明进一步提供一种药学组合物,其包括如本发明的抗体。The present invention further provides a pharmaceutical composition comprising the antibody of the present invention.
进一步提供一种如本发明的抗体,其进一步包括一标记,优选为供用于活体内成像的标记。There is further provided an antibody according to the invention, which further comprises a label, preferably a label for in vivo imaging.
本发明还提供一种用于治疗一具有一肿瘤或处于有一肿瘤的风险的个体的方法,其包括投予如本发明的双特异性或多特异性抗体至该个体。还提供一种如本发明的双特异性或多特异性抗体供用于治疗具有一肿瘤或处于有一肿瘤的风险的个体。进一步提供本发明的抗体供用于制备用于治疗一具有一肿瘤或处于有一肿瘤的风险的个体的一药剂的用途。在优选具体例中,肿瘤为EGFR或CLEC12A阳性肿瘤或EGFR及PD-L1阳性肿瘤。The present invention also provides a method for treating an individual having or at risk of having a tumor, comprising administering to the individual a bispecific or multispecific antibody according to the invention. Also provided is a bispecific or multispecific antibody according to the invention for use in the treatment of an individual having or at risk of having a tumor. Further provided is the use of an antibody of the invention for the manufacture of a medicament for the treatment of an individual having or at risk of having a tumor. In a preferred embodiment, the tumor is an EGFR or CLEC12A positive tumor or an EGFR and PD-L1 positive tumor.
附图说明Description of drawings
图1figure 1
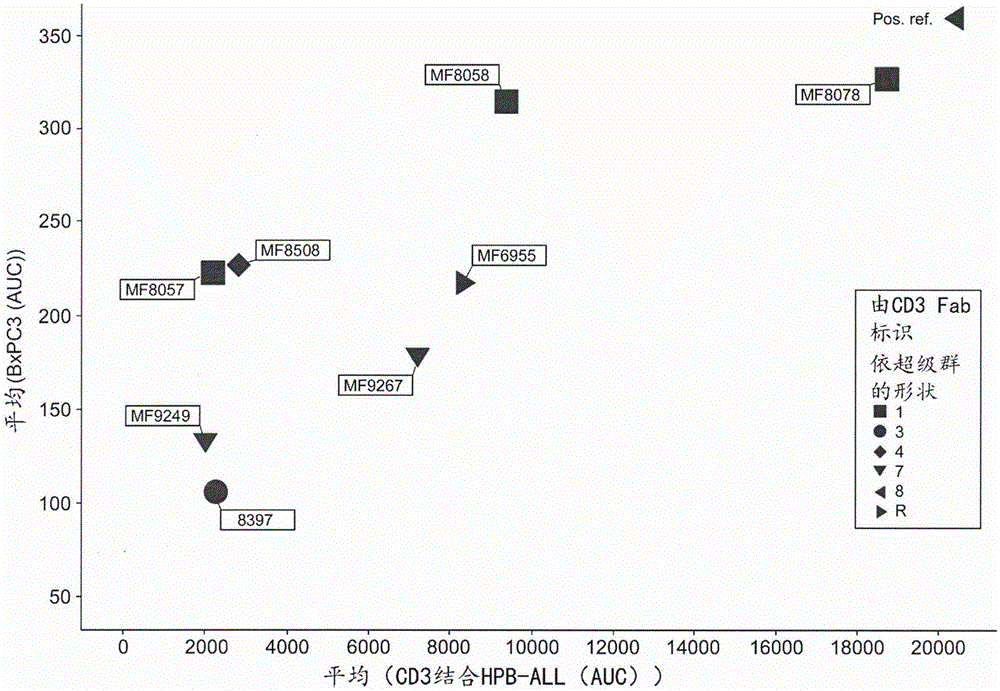
功能活性评估:BxPC3标靶细胞用EGFRxCD3双特异性抗体治疗后的T细胞细胞毒性分析。各双特异性抗体包括一种由一以MF号码命名的重链可变区构成的CD3结合域,及一种包括一重链可变区MF8233的EGFR结合域。这些可变区与一共同的轻链配对以形成一种EGFRxCD3双特异性抗体。亲和力(HPB-ALL结合)相对于BxPC3溶解。本发明的某些抗体展现出相对低的HPB-ALL细胞结合电平其表示该抗体的CD3结合域以比较低的亲和力与人类CD3结合。很清楚的是相对较低的亲和力不一定会阻止BxPC3细胞的肿瘤抗原媒介的T细胞细胞毒性(BxPC3溶解,垂直轴)。该双特异性抗体MF8233 x MF8508及MF8233 x MF8057能有效的溶解BxPC3细胞而该双特异性抗体MF8233 x MF8397及MF8233 x MF9249虽然具有相似的结合却无法有效地如此做。而且,比较该双特异性抗体MF8233 x MF6955以较高的亲和力结合HPB-ALL(即人类CD3),但无法比结合CD3程度较小的该双特异性抗体MF8233 x MF8508及MF8233 x MF8057更有效地溶解BxPC3细胞。MF6955为与一共同的轻链组合的一重链可变区及使用作比较子序列(comparator sequences),以及相应于US2014/0088295 A1内的H1H7232B(1129)VH。该具有MF6955及相同的EGFR结合域的比较子双特异性抗体分别具有对人类CD3的亲和力比MF9267更高,以及展现出比MF9267更有效的杀灭。相比的下,其他纳入本发明的CD3结合域的抗体,例如MF8058,具有如MF6955大致相同的结合活性,然而显示出更有效的BxPC3细胞杀灭,例如MF8233 x MF8058。其他包括本发明能够结合CD3的结合域的双特异性抗体显示出相对高的结合及更有效的杀灭,例如MF8233 x MF8078,其对本文所述的特定应用有用。然而其他的包括本发明能够结合CD3的结合域的双特异性抗体显示出相对低的亲和力及低杀灭,例如MF8233 xMF9249及MF8233 x MF8397,其对本文所述的任择应用有用。Functional activity assessment: T cell cytotoxicity analysis of BxPC3 target cells after treatment with EGFRxCD3 bispecific antibody. Each bispecific antibody includes a CD3 binding domain consisting of a heavy chain variable region designated by the MF number, and an EGFR binding domain including a heavy chain variable region MF8233. These variable regions are paired with a common light chain to form an EGFRxCD3 bispecific antibody. Affinity (HPB-ALL binding) relative to BxPC3 solubilization. Certain antibodies of the present invention exhibit relatively low levels of binding to HPB-ALL cells indicating that the CD3 binding domain of the antibody binds human CD3 with relatively low affinity. It is clear that relatively low affinity does not necessarily prevent tumor antigen-mediated T cell cytotoxicity of BxPC3 cells (BxPC3 lysis, vertical axis). The bispecifics MF8233 x MF8508 and MF8233 x MF8057 efficiently lysed BxPC3 cells whereas the bispecifics MF8233 x MF8397 and MF8233 x MF9249, although having similar binding, did not do so efficiently. Furthermore, the bispecific antibodies MF8233 x MF6955 were compared to bind HPB-ALL (i.e. human CD3) with a higher affinity, but not more efficiently than the bispecific antibodies MF8233 x MF8508 and MF8233 x MF8057, which bound CD3 to a lesser extent Lyse BxPC3 cells. MF6955 is a heavy chain variable region combined with a common light chain and used as comparator sequences, and corresponds to the H1H7232B(1129) VH in US2014/0088295 A1. This comparator bispecific antibody with MF6955 and the same EGFR binding domain has a higher affinity for human CD3 than MF9267 and exhibits more potent killing than MF9267, respectively. In contrast, other antibodies incorporating the CD3 binding domains of the invention, eg, MF8058, have approximately the same binding activity as MF6955, but show more efficient killing of BxPC3 cells, eg, MF8233 x MF8058. Other bispecific antibodies of the invention comprising binding domains capable of binding CD3 exhibit relatively high binding and more efficient killing, eg MF8233 x MF8078, which are useful for the specific applications described herein. However other bispecific antibodies of the invention comprising binding domains capable of binding CD3 exhibit relatively low affinity and low killing, eg MF8233 x MF9249 and MF8233 x MF8397, which are useful for any of the applications described herein.
图2figure 2
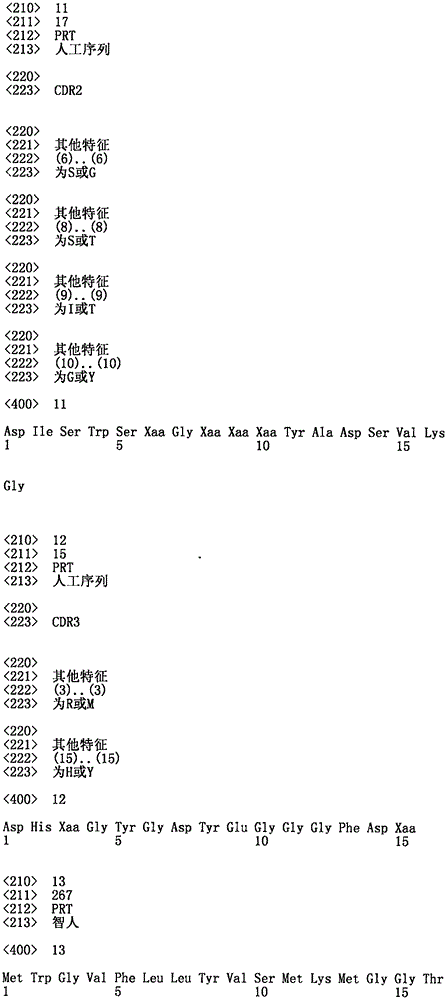
相较于无抗体对照的抗体滴定曲线,其表示其诱发T细胞媒介的BxPC3标靶细胞的杀灭%。显示抗体MF8233 x MF8078、MF8233x MF8397;及MF8233 xMF8508的曲线。Antibody titration curve expressed as a % kill of T cell mediated BxPC3 target cells it induces compared to no antibody control. Curves are shown for antibodies MF8233 x MF8078, MF8233 x MF8397; and MF8233 x MF8508.
图3
各种双特异性抗体于用BxPC3标靶细胞的T细胞细胞毒性中的滴定曲线数据的概要。CD3 Fab栏目表示CD3结合臂的MF号码。EGFR臂具有已指明的MF8233号码。该栏目表示超级群号码以基于相同的VH基因节段列出变异体。表示CD3结合的栏目反映HBP-ALL结合实验的结果。Summary of titration curve data for various bispecific antibodies in T cell cytotoxicity targeting cells with BxPC3. The CD3 Fab column indicates the MF number of the CD3 binding arm. The EGFR arm has the assigned MF8233 number. This column indicates the supergroup number to list variants based on the same VH gene segment. Columns indicating CD3 binding reflect the results of HBP-ALL binding experiments.
显示判定诱发T细胞媒介的BxPC3标靶细胞的溶解能力的二个独立细胞毒性分析的结果。The results of two independent cytotoxicity assays to determine the lytic capacity of induced T cell-mediated BxPC3 target cells are shown.
图4Figure 4
用BxPC3标靶细胞对于有表现的CD8+T细胞进行的T细胞细胞毒性分析的T细胞活化。各种超级群号码的抗体滴定曲线,其列出变异体CD3结合域。该双特异性抗体的另一臂具有MF8233的重链结合域。为了比较该双特异性抗体MF8233 x MF6955及MF8233 x MF6964也被测试,MF6955及MF6964为与一共同的轻链组合的重链可变区且使用作比较子序列,以及分别相应于US2014/0088295 A1内的H1H7232B(1129)VH及HH7241B(1145)。T Cell Activation by T Cell Cytotoxicity Assay with BxPC3 Target Cells on Expressing CD8+ T Cells. Antibody titration curves for various supergroup numbers listing variant CD3 binding domains. The other arm of this bispecific antibody has the heavy chain binding domain of MF8233. To compare the bispecific antibodies MF8233 x MF6955 and MF8233 x MF6964 were also tested, MF6955 and MF6964 are heavy chain variable regions combined with a common light chain and used as comparison subsequences, and correspond to US2014/0088295 A1, respectively H1H7232B(1129) VH and HH7241B(1145) inside.
图5Figure 5
各种抗体于用BxPC3标靶细胞的T细胞活化T细胞细胞毒性分析中的滴定曲线数据的概要。MFnr.栏目表示CD3结合域的MF号码。EGFR结合域具有已指明的MF8233号码。各种CD3结合域序列的超级群信息表示于栏目“超级群”内;表示CD3亲和力的栏目反映HBP-ALL结合实验的结果。Summary of titration curve data for various antibodies in T cell activation T cell cytotoxicity assays with BxPC3 target cells. The column MFnr. indicates the MF number of the CD3 binding domain. The EGFR binding domain has the assigned MF8233 number. Supergroup information for various CD3 binding domain sequences is presented in the column "Supergroup"; the column indicating CD3 affinity reflects the results of HBP-ALL binding experiments.
显示针对标识CD69及CD25的CD4+及CD8+细胞的结果。指明的双特异性抗体是一大池双特异性抗体中的范例。Results are shown for CD4+ and CD8+ cells that label CD69 and CD25. The indicated bispecific antibodies are examples in a large pool of bispecific antibodies.
图6
功能活性评估:用BxPC3标靶细胞的T细胞细胞毒性分析。亲和力(HPB-ALL结合)相对于通过CD69表现来测量的CD8+T细胞活化。本发明的某些抗体展现出相对低的HPB-ALL细胞结合电平其表示该抗体的CD3结合域以比较低的亲和力与人类CD3结合。这些亲和力不一定会阻止肿瘤抗原媒介的T细胞活化,如由CD8阳性CD69活化分析的结果所示。该双特异性抗体MF8233 x MF8508及MF8233 x MF8057能有效的活化T细胞而该双特异性抗体MF8233xMF8397及MF8233 xMF9249无法有效地如此做。某些无法有效地结合这些细胞的CD3结合域也无法活化T细胞(见左下角)。而其他CD3结合域例如,MF8508及MF8057,其与HPB-ALL细胞结合比比较子CD3结合域MF6955更少,使T细胞活化达相似的程度。其他包括本发明能够结合CD3的结合域的双特异性抗体显示出相对高的结合及高电平的活化,例如MF8078,其对本文所述的特定应用有用。然而其他包括本发明能够结合CD3的结合域的双特异性抗体显示出相对低的亲和力及低活化,例如MF9249及MF8397,其对本文所述的任择应用有用。Functional activity assessment: T cell cytotoxicity assay with BxPC3 target cells. Affinity (HPB-ALL binding) relative to CD8+ T cell activation measured by CD69 expression. Certain antibodies of the present invention exhibit relatively low levels of binding to HPB-ALL cells indicating that the CD3 binding domain of the antibody binds human CD3 with relatively low affinity. These affinities do not necessarily prevent tumor antigen-mediated T cell activation, as shown by the results of CD8-positive CD69 activation assays. The bispecific antibodies MF8233 x MF8508 and MF8233 x MF8057 were effective in activating T cells whereas the bispecific antibodies MF8233 x MF8397 and MF8233 x MF9249 were not. Certain CD3-binding domains that do not efficiently bind these cells also do not activate T cells (see bottom left). While other CD3 binding domains such as MF8508 and MF8057, which bound less to HPB-ALL cells than the comparator CD3 binding domain MF6955, activated T cells to a similar extent. Other bispecific antibodies of the invention comprising binding domains capable of binding CD3 exhibit relatively high binding and high levels of activation, such as MF8078, which are useful for the specific applications described herein. However other bispecific antibodies comprising binding domains of the invention capable of binding CD3 exhibit relatively low affinity and low activation, eg MF9249 and MF8397, which are useful for the optional applications described herein.
图7Figure 7
功能活性评估:用HCT116标靶细胞的T细胞细胞毒性分析。亲和力(HPB-ALL结合)相对于HCT-116溶解。Functional activity assessment: T cell cytotoxicity assay with HCT116 target cells. Affinity (HPB-ALL binding) relative to HCT-116 solubilization.
本发明的某些双特异性抗体展现出低的HPB-ALL细胞结合电平其表示该抗体的CD3结合域以比较低的亲和力与人类CD3结合。很清楚的是低的亲和力不一定会阻止肿瘤抗原媒介的HCT-116细胞的细胞溶解(垂直轴)。该双特异性抗体MF8233 x MF8508及MF8233 xMF8057能有效的溶解HCT-116细胞而该双特异性抗体MF8233 xMF8397及MF8233 xMF9249无法有效地如此做。为了比较该双特异性抗体MF8233 x MF6955及MF8233 x MF6964以比例如,MF8233 x MF8508、MF8233xMF8057及MF8233 xMF9267更高的亲和力来结合HPB-ALL(即人类CD3),但无法比MF8233 x MF8508或MF8233 x MF9267更有效地,或比MF8233 xMF8057显着更有效地溶解HCT-116细胞,相对于如此处所示的测试中的结合上的差异。另一种包括本发明能够结合CD3的结合域的双特异性抗体显示出相对高的结合及高电平的杀灭,例如MF8078,其对本文所述的特定应用有用。然而其他包括本发明能够结合CD3的结合域的双特异性抗体显示出相对低的亲和力及低杀灭,例如MF9249及MF8397,其对本文所述的任择应用有用。Certain bispecific antibodies of the invention exhibit low levels of HPB-ALL cell binding indicating that the CD3 binding domain of the antibody binds human CD3 with relatively low affinity. It is clear that low affinity does not necessarily prevent tumor antigen mediated cytolysis of HCT-116 cells (vertical axis). The bispecific antibodies MF8233 x MF8508 and MF8233 x MF8057 were able to effectively lyse HCT-116 cells while the bispecific antibodies MF8233 x MF8397 and MF8233 x MF9249 were not. To compare the bispecific antibodies MF8233 x MF6955 and MF8233 x MF6964 bind HPB-ALL (i.e. human CD3) with higher affinity than, for example, MF8233 x MF8508, MF8233 x MF8057 and MF8233 x MF9267, but not MF8233 x MF8508 or MF8233 x MF9267 lysed HCT-116 cells more efficiently, or significantly more efficiently than MF8233xMF8057, relative to the difference in binding as in the assay shown here. Another bispecific antibody comprising a binding domain of the invention capable of binding CD3 exhibits relatively high binding and high levels of killing, eg, MF8078, which is useful for the specific applications described herein. However other bispecific antibodies of the invention comprising binding domains capable of binding CD3 exhibit relatively low affinity and low killing, eg MF9249 and MF8397, which are useful for the optional applications described herein.
图8Figure 8
用HCT-116标靶细胞的T细胞细胞毒性分析中,相较于无抗体对照的抗体滴定曲线,其表示HCT-116细胞的杀灭%。显示各种双特异性抗体的曲线。Antibody titration curves representing % killing of HCT-116 cells compared to no antibody controls in T cell cytotoxicity assays with HCT-116 target cells. Curves are shown for various bispecific antibodies.
图9Figure 9
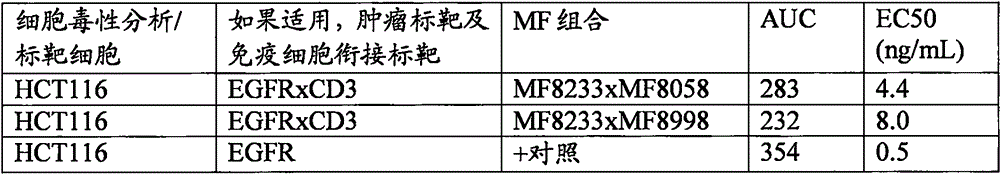
各种抗体于用HCT-116标靶细胞的T细胞细胞毒性分析中的滴定曲线数据的概要。CD3 Fab栏目表示CD3结合域的MF号码。EGFR结合域具有已指明的MF8233号码。该栏目表示超级群号码以基于相同的VH基因节段列出变异体。表示CD3结合的栏目反映HBP-ALL结合实验的结果。Summary of titration curve data for various antibodies in T cell cytotoxicity assays using HCT-116 target cells. The CD3 Fab column indicates the MF number of the CD3 binding domain. The EGFR binding domain has the assigned MF8233 number. This column indicates the supergroup number to list variants based on the same VH gene segment. Columns indicating CD3 binding reflect the results of HBP-ALL binding experiments.
HCT-116细胞的溶解百分率及溶解的EC50值(ng/mL)表示于接下来的字段中。所指明的双特异性抗体是一大池双特异性抗体中的范例。Percent lysis of HCT-116 cells and EC50 values for lysis (ng/mL) are presented in the next fields. The bispecific antibodies indicated are examples in a large pool of bispecific antibodies.
图10Figure 10
图10a及10b列出MV1624表现载体及MV1625表现载体的示意图。Figures 10a and 10b list schematic diagrams of the MV1624 expression vector and the MV1625 expression vector.
图11Figure 11
单及双特异性IgG内使用的共同的轻链。A common light chain used within mono- and bispecific IgGs.
图11A:共同的轻链氨基酸序列。图11B:共同的轻链可变结构域DNA序列及呈现(IGKV1-39/jk1)。图11C:共同的轻链恒定区域DNA序列及转译。图11D:IGKV1-39/jk5共同的轻链可变结构域转译。图11E:V区域IGKV1-39A;图11F:共同的轻链的CDR1、CDR2及CDR3。Figure 11A: Common light chain amino acid sequence. Figure 11B: Common light chain variable domain DNA sequence and representation (IGKV1-39/jk1). Figure 11C: Common light chain constant region DNA sequence and translation. Figure 1 ID: IGKV1-39/jk5 common light chain variable domain translation. Figure 11E: V region IGKV1-39A; Figure 11F: CDR1, CDR2 and CDR3 of the common light chain.
图12Figure 12
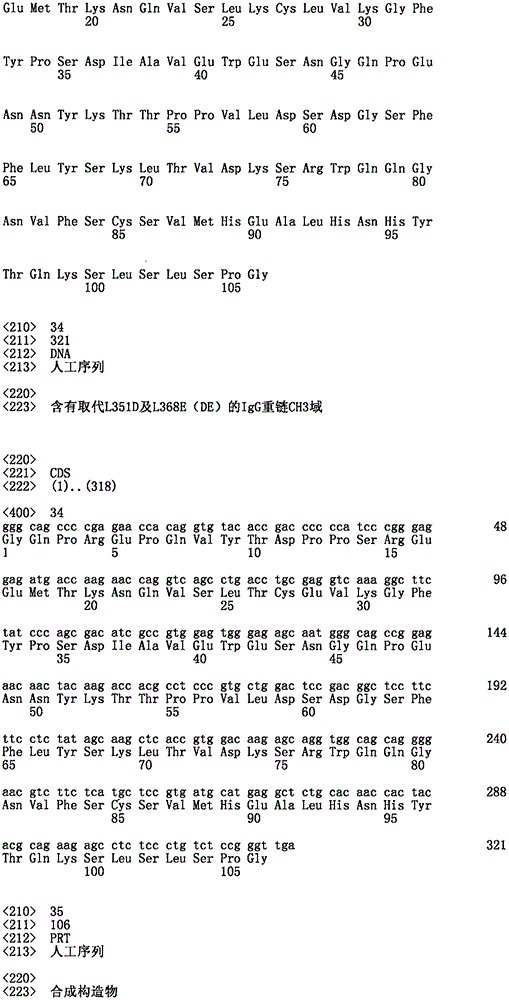
双特异性分子的IgG重链。图12A:CH1区域。图12B:铰链区域。图12C:CH2区域。图12D:含有L235G及G236R沉默替换的CH2。图12E:含有取代L351K及T366K(KK)的CH3域。图12F;含有取代L351D及L368E(DE)的CH3域。IgG heavy chains of bispecific molecules. Figure 12A: CH1 region. Figure 12B: Hinge area. Figure 12C: CH2 region. Figure 12D: CH2 containing silent substitutions of L235G and G236R. Figure 12E: CH3 domain containing substitutions L351K and T366K (KK). Figure 12F; CH3 domain containing substitutions L351D and L368E (DE).
图13Figure 13
编码本说明书中所述的重链可变区及其部分的各种DNA的序列及其的氨基酸序列。Sequences and amino acid sequences of various DNAs encoding the heavy chain variable regions and portions thereof described in this specification.
图14Figure 14
来自超级群1附加的殖株与殖株MF8057及MF8058相比的特征化。A:于FACS分析中所选的MF殖株与表现人类CD3-TCR错合物的HPB-ALL人类细胞的结合。B:以HCT-116细胞进行的T细胞细胞毒性分析,其表示HCT-116细胞的杀灭%。C-E:表示T细胞活化的活化标识CD25及CD69以FACS的定量。F-G:来自细胞毒性分析之上清液的细胞介素生产。Characterization of additional clones from
图15Figure 15
来自超级群4的殖株的特征化。A:所选的MF殖株与HPB-ALL人类细胞的结合。B:以BxPC3细胞进行的T细胞细胞毒性分析,其表示BxPC3细胞的杀灭%。C-E:来自细胞毒性分析之上清液的细胞介素生产。Characterization of clones from
图16Figure 16
CD3功能活性评估:附加的殖株来自超级群1(MF8048、MF8101、MF8056)、超级群3(MF8562)及超级群4(MF8998)的亲和力(HPB-ALL)于X轴上相对HCT-116溶解于Y轴上。B:属于超级群1及超级群4的抗体,其于细胞毒性分析显示出相似的活性及不同的结合亲和力。C:属于超级群1及超级群3的抗体,其展现相似的结合亲和力及差异的溶解活性。Assessment of CD3 functional activity: Additional clones from supergroup 1 (MF8048, MF8101, MF8056), supergroup 3 (MF8562) and supergroup 4 (MF8998) Affinity (HPB-ALL) relative to HCT-116 lysis on the X-axis on the Y axis. B: Antibodies belonging to
图17Figure 17
呈双特异性CD3xEGFR格式的CD3 Fabs MF8998及MF8058的活性。Activity of CD3 Fabs MF8998 and MF8058 in bispecific CD3xEGFR format.
图18Figure 18
一大组对CD3有特异性的IgGs的FACS结合数据。针对抗体MF5196、MF6955及MF6964,通过BIAcoreTM来判定对于CD3δε-Fc抗原的结合,同时显示剩余殖株与HPB-ALL细胞的FACS结合数据。FACS binding data for a large set of IgGs specific for CD3. For the antibodies MF5196, MF6955 and MF6964, the binding to the CD3δε-Fc antigen was determined by BIAcore ™ , and the FACS binding data of the remaining clones to HPB-ALL cells were also shown.
图19Figure 19
人类CLEC12A的核苷酸序列。Nucleotide sequence of human CLEC12A.
图20Figure 20
人类CD3γ-、δ-、ε-及ζ-链的氨基酸序列。Amino acid sequences of human CD3 gamma-, delta-, epsilon- and zeta-chains.
具体实施方式Detailed ways
一种“抗体”是一种属于蛋白质的免疫球蛋白类的蛋白质分子,其含有结合抗原上的表位的一个或多个域,其中这些域衍生自一抗体的可变区或与一抗体的可变区共享序列同源性。An "antibody" is a protein molecule belonging to the immunoglobulin class of proteins that contains one or more domains that bind epitopes on an antigen, wherein the domains are derived from the variable regions of an antibody or associated with an antibody. The variable regions share sequence homology.
抗体结合有不同的特质,包括特异性和亲和力。特异性决定该结合域特异性地结合那个抗原或其表位。亲和力是对于一个特定抗原或表位的结合量值的量度(measure)。在此可方便地注意到:一个抗体的“特异性”意指其对于一个特定抗原的选择性,而“亲和力”意指介于该抗体的抗原结合地址以及其结合的表位之间的交互作用量值。抗体典型由基本结构单元组成-各基本结构单元有二重链及二轻链。治疗用途的抗体优选尽可能接近待治疗个体的天然抗体(譬如人类个体的人类抗体)。如本发明的抗体不限于任何特定格式或其生产方法。Antibodies bind with different properties, including specificity and affinity. Specificity determines which antigen or epitope the binding domain specifically binds to. Affinity is a measure of the amount of binding for a particular antigen or epitope. It is convenient to note here that "specificity" of an antibody means its selectivity for a particular antigen, and "affinity" means the interaction between the antigen binding address of the antibody and the epitope it binds Action value. Antibodies are typically composed of basic building blocks - each with a double chain and a double light chain. Antibodies for therapeutic use are preferably as close as possible to native antibodies of the subject to be treated (eg, human antibodies of a human subject). Antibodies such as the present invention are not limited to any particular format or method of production thereof.
因此,如此处所用的“结合特异性(binding specificity)”意指一单独的抗体结合地址跟一抗原决定位反应的能力。通常,本发明的抗体的结合地址座落在包括此类可变结构域的Fab部分的此类可变结构域内,并且从该重链及轻链的高度变异区域被构造。Thus, "binding specificity" as used herein means the ability of an individual antibody binding address to react with an epitope. Typically, the binding addresses of the antibodies of the invention are located within such variable domains including the Fab portion of such variable domains, and are constructed from the hypervariable regions of the heavy and light chains.
本发明的抗体优选为一IgG抗体,优选为一IgG1抗体。全长IgG抗体可能是优选的,因为其有利的半生期且由于为了免疫原性的理由,希望保持接近完全自体(人类)分子。IgG1基于其在人体内的长循环半生期而是有利的。为了要预防或避免在人类的免疫原性,一种根据本发明的双特异性全长IgG抗体是一种人类IgG1是优选的。The antibody of the present invention is preferably an IgG antibody, preferably an IgG1 antibody. Full length IgG antibodies may be preferred due to their favorable half-life and due to the desire to remain close to fully autologous (human) molecules for immunogenicity reasons. IgG1 is advantageous based on its long circulating half-life in humans. In order to prevent or avoid immunogenicity in humans, it is preferred that a bispecific full-length IgG antibody according to the invention be a human IgG1.
“双特异性抗体”为一种如本文所述的抗体其中该抗体的一可变结构域与第一抗原结合而该抗体的第二可变结构域与第二抗原结合,其中该第一与第二抗原不是完全相同的。术语“双特异性抗体”还包括双特异性抗体(biparatopic antibodies),其中该抗体的一可变结构域结合一抗原上的第一表位而该抗体的第二可变结构域结合该抗原上的第二表位。该术语进一步包括抗体,其中至少一VH能特异性识别第一抗原且与免疫球蛋白可变结构域的该至少一VH配对的VL能特异性识别第二抗原。生成的VH/VL对会结合抗原1或抗原2,且称为“二合一抗体”,其于例如WO 2008/027236、WO 2010/108127及Schaefer等人(Cancer Cell 20,472-486,2011年十月)乙文内有描述。如本发明的双特异性抗体不限于任何特定双特异性格式或其生产方法。一种双特异性抗体是一种多特异性抗体。A "bispecific antibody" is an antibody as described herein wherein a variable domain of the antibody binds a first antigen and a second variable domain of the antibody binds a second antigen, wherein the first and The second antigens are not identical. The term "bispecific antibody" also includes biparatopic antibodies wherein a variable domain of the antibody binds a first epitope on an antigen and a second variable domain of the antibody binds the antigen the second epitope. The term further includes antibodies wherein at least one VH specifically recognizes a first antigen and a VL paired with the at least one VH of an immunoglobulin variable domain specifically recognizes a second antigen. The resulting VH/VL pairs will bind
如本文中所提及的多特异性多聚体或抗体涵盖属于蛋白质的免疫球蛋白类的蛋白质分子,其含有结合抗原上的表位的二个或多个域,其中这些域衍生自一抗体的可变区或与一抗体的可变区共享序列同源性,以及包括如本技艺所知结合三个或多个抗原的蛋白质分子,包括如于WO2019/190327中所述者。Multispecific multimers or antibodies as referred to herein encompass protein molecules belonging to the immunoglobulin class of proteins containing two or more domains that bind epitopes on an antigen, wherein these domains are derived from an antibody The variable regions of or share sequence homology with the variable regions of an antibody, and include protein molecules that bind three or more antigens as known in the art, including as described in WO2019/190327.
“抗原”是一种能够在一宿主生物内诱发一免疫反应(以生成一抗体)的分子和/或一抗体靶向的分子。以分子级(molecular level)而言,一种抗原特征在于其被一个抗体的抗原结合地址所结合的能力。又,抗原的混合物可被视为是一种“抗原”,即熟习本领域技艺的人士将会理解到有时候肿瘤细胞的溶解产物或病毒颗粒可表示为“抗原”,而这样的肿瘤细胞溶解产物或病毒颗粒制备物存在有许多抗原决定位。一种抗原包括至少一种但通常是多种表位。针对如本文所公开的结合蛋白及抗体,抗原典型和细胞膜有关联且存在于细胞膜的细胞外部分之上。An "antigen" is a molecule capable of eliciting an immune response (to generate an antibody) in a host organism and/or an antibody-targeted molecule. At the molecular level, an antigen is characterized by its ability to be bound by the antigen-binding address of an antibody. Again, a mixture of antigens can be considered an "antigen", ie those skilled in the art will understand that sometimes tumor cell lysates or viral particles can be represented as "antigens" and such tumor cell lysates A number of epitopes are present in the product or virus particle preparation. An antigen includes at least one, but usually multiple, epitopes. For the binding proteins and antibodies as disclosed herein, the antigen is typically associated with the cell membrane and is present on the extracellular portion of the cell membrane.
“表位”或“抗原决定位”是指一抗原上与免疫球蛋白或抗体特异性结合的地址。表位可由相邻的氨基酸或由蛋白的三级折叠而并列的不连续氨基酸(分别为所谓的线形或构形表位)形成。自相邻的、线形氨基酸形成的表位通常在暴露于变性溶剂时得以保持,而由三级折叠形成的表位通常在用变性溶剂处理时会丧失构形。表位通常在独特的空间构形中包括了3、4、5、6、7、8、9、10、11、12、13、14或15个氨基酸。An "epitope" or "epitope" refers to the address on an antigen to which an immunoglobulin or antibody specifically binds. Epitopes can be formed from adjacent amino acids or discrete amino acids juxtaposed by the tertiary fold of the protein (so-called linear or conformational epitopes, respectively). Epitopes formed from adjacent, linear amino acids are typically retained upon exposure to denaturing solvents, whereas epitopes formed from tertiary folds typically lose their conformation upon treatment with denaturing solvents. Epitopes typically include 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or 15 amino acids in unique spatial configurations.
术语“重链”或“免疫球蛋白重链”包括来自任一种生物的一免疫球蛋白重链恒定区域序列,而且除非另有规定,否则会包括一重链可变结构域。除非另有规定,否则术语重链可变结构域会包括三个重链CDRs以及四个FR区域。重链的片段包括CDRs、CDRs与FRs及其的组合。一种典型的重链在该可变结构域的后(从N-端至C-端)具有一CH1域、一铰链、一CH2域以及一CH3域。一种重链的一功能性片段包括一种能够特异性地识别一抗原并且包括至少一CDR的片段。The term "heavy chain" or "immunoglobulin heavy chain" includes an immunoglobulin heavy chain constant region sequence from any organism and, unless otherwise specified, a heavy chain variable domain. Unless otherwise specified, the term heavy chain variable domain will include three heavy chain CDRs and four FR regions. Fragments of heavy chains include CDRs, CDRs and FRs, and combinations thereof. A typical heavy chain has a CH1 domain, a hinge, a CH2 domain, and a CH3 domain after the variable domain (from N-terminus to C-terminus). A functional fragment of a heavy chain includes a fragment that specifically recognizes an antigen and includes at least one CDR.
术语“轻链”包括来自任一种生物的一免疫球蛋白轻链可变结构域或VL(或其功能性片段;以及一免疫球蛋白恒定域或CL或其功能性片段的序列。除非另有规定,否则术语轻链可包括一选自于人类κ、λ及其的一组合的轻链。除非另有规定,否则轻链可变(VL)结构域典型包括三个轻链CDRs以及四个架构(FR)区域。一般而言,一全长轻链从N-端至C-端包括了一种包括FR1-CDR1-FR2-CDR2-FR3-CDR3-FR4的VL域以及一种轻链恒定域。本发明可使用的轻链包括,例如不会选择性地结合一种选择性地由此类重链所结合的表位的此类。The term "light chain" includes the sequence of an immunoglobulin light chain variable domain or VL (or functional fragment thereof; and an immunoglobulin constant domain or CL or functional fragment thereof, from any organism. Unless otherwise specified, the term light chain may include a light chain selected from human kappa, lambda, and a combination thereof. Unless otherwise specified, a light chain variable ( VL ) domain typically includes three light chains CDRs and four framework (FR) regions. Generally, a full-length light chain includes a VL domain including FR1-CDR1-FR2-CDR2-FR3-CDR3-FR4 from N-terminus to C-terminus and A light chain constant domain. Light chains useful in the present invention include, for example, such that do not selectively bind an epitope that is selectively bound by such heavy chains.
供用于本发明的抗体的合适的轻链包括一种共同的轻链(cLC),诸如可通过筛选现有的抗体库[湿式库(wet libraries)或电脑模拟(in silico)]中最常使用的轻链而鉴定出的此类,其中此类轻链大体上不会干扰此类重链的表位结合域的亲和力和/或选择性,但也适合于与一系列的重链配对。举例来说,一种合适的轻链包括一种来自一基因转殖动物,诸如的物,该基因转殖动物具有嵌入至其基因体的内的共同的轻链且其可用来产生大量的(large panels of)共同的轻链抗体其在重链处具有多样性的且在暴露于一抗原的时能特异性地结合该抗原。Suitable light chains for use in the antibodies of the invention include a common light chain (cLC), such as can be most commonly used by screening existing antibody libraries [wet libraries or in silico] Such light chains have been identified as light chains that do not substantially interfere with the affinity and/or selectivity of the epitope binding domains of such heavy chains, but are also suitable for pairing with a range of heavy chains. For example, a suitable light chain includes one from a transgenic animal such as The transgenic animals have a common light chain embedded within their gene bodies and which can be used to generate large panels of common light chain antibodies that are diverse at the heavy chain and are exposed to In the presence of an antigen, it can specifically bind to the antigen.
如本发明的术语“共同的轻链”是指可能完全相同或有一些氨基酸序列差异却不影响本发明的抗体的结合特异性,即此类差异不会实质地影响功能性结合区域的形成的轻链。The term "common light chain" as used in the present invention refers to those which may be identical or have some amino acid sequence differences without affecting the binding specificity of the antibodies of the present invention, ie such differences do not substantially affect the formation of a functional binding region light chain.
例如于本文所使用的共同的轻链的定义范围内,要制备或找到不是完全相同但仍然在功能上等效的可变链是可能的,譬如通过导入且测试保留型氨基酸变化来进行,当与一同源链等等配对时,区域内的氨基酸变化不会对结合特异性起作用或只是起部分作用。这些变异体因此还能够结合不同的同源链而且形成功能性抗原结合域。如此处所用的术语共同的轻链因而是指可能完全相同或有一些氨基酸序列差异但在与一重链配对后保留了所形成的抗体的结合特异性的轻链。某些共同的轻链和这些功能上等效的变异体的组合涵盖在术语共同的轻链的内。关于共同的轻链的用途的详细说明可参见WO 2004/009618及WO2009/157771。For example, within the definition of a common light chain as used herein, it is possible to make or find variable chains that are not identical but still functionally equivalent, such as by introducing and testing retained amino acid changes, when Amino acid changes within a region have no or only partial effect on binding specificity when paired with homologous chains and the like. These variants are thus also able to bind different homologous chains and form functional antigen binding domains. The term common light chain as used herein thus refers to light chains that may be identical or have some amino acid sequence differences but retain the binding specificity of the resulting antibody after pairing with a heavy chain. Combinations of certain common light chains and these functionally equivalent variants are encompassed within the term common light chain. Detailed descriptions of the use of common light chains can be found in WO 2004/009618 and WO 2009/157771.
“Fab”是指一种包括一个可变区的结合域,通常是一种包括有一配对的重链可变区及轻链可变区的结合域。Fab可包括恒定区域域,其包括一个CH1及VH域与一个恒定轻链域(CL)及VL域配对。这些配对可以,例如,经由一个位在CH1和CL域的处的双硫键来作为共价键联而发生。"Fab" refers to a binding domain comprising a variable domain, typically a binding domain comprising a paired heavy chain variable domain and light chain variable domain. Fabs can include constant region domains that include a CH1 and VH domain paired with a constant light chain domain (CL) and VL domains. These pairings can occur as covalent linkages, eg, via a disulfide bond at the CH1 and CL domains.
“单链可变片段”(scFv)是指一种结合域其包括经由例如长度由大约10至大约25个氨基酸的键接子,例如肽键接子,所连结的VH域及VL域。A "single-chain variable fragment" (scFv) refers to a binding domain comprising the VH and VL domains linked by, for example, a linker, eg, a peptide bond, of from about 10 to about 25 amino acids in length.
如本发明的术语“全长IgG”或“全长抗体”定义为包括基本上完整的IgG,然而其不一定具有完整的IgG的全部功能。为避免疑义,全长IgG含有二重链及二轻链。各链含有恒定(C)及可变(V)区域,其能细分为命名为CH1、CH2、CH3、VH及CL、VL的域。IgG抗体经由Fab部分内含的可变区域与抗原结合,且于结合后能经由恒定域,主要是经由Fc部分,来与免疫系统的分子及细胞交互作用。如本发明的全长抗体包括其中可能存在提供所欲特征的突变的IgG分子。全长IgG不应有任何域的实质部分的缺失。然而,术语“全长IgG”包括其中缺失一或数个氨基酸残基,但没有实质改变产生的IgG分子的结合特征的IgG分子。举例来说,此IgG分子能有1至10个之间的氨基酸残基缺失,优选于非CDR区域内,其中缺失的氨基酸对IgG的结合特异性不是必需的。The term "full-length IgG" or "full-length antibody" as used in the present invention is defined to include substantially intact IgG, which however do not necessarily have all the functions of intact IgG. For the avoidance of doubt, full-length IgG contains a double chain and a double light chain. Each chain contains constant (C) and variable (V) regions, which can be subdivided into domains designated CH1, CH2, CH3, VH and CL, VL. IgG antibodies bind to antigens via variable regions contained within the Fab portion, and upon binding are able to interact with molecules and cells of the immune system via the constant domains, primarily via the Fc portion. Full-length antibodies as in the present invention include IgG molecules in which mutations that provide the desired characteristics may be present. A full-length IgG should not have deletions of substantial portions of any domains. However, the term "full-length IgG" includes IgG molecules in which one or several amino acid residues are deleted without substantially altering the binding characteristics of the resulting IgG molecule. For example, the IgG molecule can have between 1 and 10 amino acid residue deletions, preferably within non-CDR regions, where the deleted amino acids are not essential for the binding specificity of the IgG.
当此提及核酸或氨基酸序列时,“同一性百分率(%)”定义为为了进行最佳比较而排列匹配序列后,候选序列的残基与一选定序列的残基为同一的百分比。要比较的两个序列中的任何一个可引入间隙以使两个序列的排列比对达成最大。此排列比对可以在要比较的全长序列上进行。任择地,排列比对可以于较短的长度上进行,例如大约20、大约50、大约100或更多核酸/碱基或氨基酸。序列同一性为于报告的排列比对区域上,两个序列之间同一的匹配的百分比。When referring to nucleic acid or amino acid sequences herein, "percent (%) identity" is defined as the percentage of residues of a candidate sequence that are identical to residues of a selected sequence after matching sequences have been aligned for optimal comparison. Either of the two sequences to be compared can introduce a gap to maximize alignment of the two sequences. This alignment can be performed on the full-length sequences to be compared. Optionally, alignments can be performed over shorter lengths, eg, about 20, about 50, about 100, or more nucleic acids/bases or amino acids. Sequence identity is the percentage of matches that are identical between two sequences over the reported aligned region.
序列的比较及两个序列之间的序列同一性百分率的测定可使用数学算法完成。熟习此项技术者会知道可用数种不同的计算机程序来排列比对二个序列且判定二个序列之间的同一性(Kruskal,J.B.,1983)An overview of sequence comparison In D.Sankoffand J.B.Kruskal,(ed.),Time warps,string edits and macromolecules:the theoryand practice of sequence comparison,pp.1-44 Addison Wesley)。Comparison of sequences and determination of percent sequence identity between two sequences can be accomplished using mathematical algorithms. Those skilled in the art will know that several different computer programs can be used to align two sequences and determine the identity between the two sequences (Kruskal, J.B., 1983). An overview of sequence comparison In D.Sankoff and J.B.Kruskal, (ed.), Time warps, string edits and macromolecules: the theory and practice of sequence comparison, pp. 1-44 Addison Wesley).
关于本发明及本文所述的序列,二个核酸序列之间的序列同一性百分率可使用Vector NTI Program Advance 10.5.2软件的AlignX应用程序、使用预设来测定,其利用修改的ClustalW算法(Thompson,J.D.,Higgins,D.G.,and Gibson T.J.(1994)Nuc.AcidRes.22:4673-4680)、swgapdnarnt得分矩阵、15之间隙开放罚分(gap open penalty)以及6.66之间隙延伸罚分(gap extension penalty)。氨基酸序列可使用Vector NTI ProgramAdvance 11.5.2软件的AlignX应用、使用默认被排列比对,其利用修改的ClustalW算法(Thompson,J.D.,Higgins,D.G.,and Gibson T.J.,1994)、blosum62mt2得分矩阵、10之间隙开放罚分及0.1之间隙延伸罚分。With regard to the present invention and the sequences described herein, the percent sequence identity between two nucleic acid sequences can be determined using the AlignX application of the Vector NTI Program Advance 10.5.2 software using presets using a modified ClustalW algorithm (Thompson , J.D., Higgins, D.G., and Gibson T.J. (1994) Nuc. AcidRes. 22:4673-4680), swgapdnarnt score matrix, gap open penalty of 15 and gap extension penalty of 6.66 ). Amino acid sequences can be aligned using the AlignX application of the Vector NTI ProgramAdvance 11.5.2 software using the default alignment using a modified ClustalW algorithm (Thompson, J.D., Higgins, D.G., and Gibson T.J., 1994), blosum62mt2 score matrix, out of 10 Gap opening penalty and gap extension penalty of 0.1.
术语“超级群(super-cluster)”或“超级群(supercluster)”在此使用于意指,基于相同的VH V基因节段使用且于HCDR3内具有至少70%的序列同一性及相同的HCDR3长度,的一群的殖株(clones)及其能生产的结合域。The term "super-cluster" or "supercluster" is used herein to mean, based on the same VH V gene segment and having at least 70% sequence identity within HCDR3 and identical HCDR3 A group of clones of length , and the binding domains they can produce.
因而,在优选具体例中,本发明提供一种“超级群(super-cluster)”或“超级群(supercluster)”,其包括,基于相同的VHV基因节段使用且于HCDR3内具有至少70%的序列同一性及相同的HCDR3长度,的一组的殖株及其能生产的结合域。在优选具体例中,该序列同一性为80%,更优选为90%,最佳为95%的序列同一性,但有条件是排除包括一编码HCDR3序列DGGYSYGPYWYFDL及DHRGYGDYEGGGFDY的核酸的一殖株,包括编码HCDR2序列GFIPVLGTANYAQKFQG、GIIPLFGTITYAQKFQG及SIIPIFGTITYAQKFQG的核酸的殖株,或有条件是排除包括编码VH序列Thus, in a preferred embodiment, the present invention provides a "super-cluster" or "supercluster" comprising, based on the same VHV gene segments used and having at least 70% in HCDR3 The sequence identity and the same length of HCDR3, a set of clones and the binding domains they can produce. In a preferred embodiment, the sequence identity is 80%, more preferably 90%, and optimally 95%, provided that a clone comprising a nucleic acid encoding the HCDR3 sequence DGGYSYGPYWYFDL and DHRGYGDYEGGGFDY is excluded, Colonies that include nucleic acids encoding HCDR2 sequences GFIPVLGTANYAQKFQG, GIIPLFGTITYAQKFQG and SIIPIFGTITYAQKFQG, or conditionally exclude sequences encoding VH
的核酸的殖株,或有条件是来自该组的殖株包括核酸,该核酸是编码由一种双特异性抗体所组成或经设计而由一种双特异性抗体所组成的HCDR3。A clone of nucleic acid, or conditionally a clone from the group, comprises nucleic acid encoding HCDR3 consisting of or designed to consist of a bispecific antibody.
术语“超级群(super-cluster)1”或“超级群(supercluster)1”在此使用于意指,基于与该超级群的成员相同的VH V基因节段使用(VH1-69)且于HCDR3内具有至少70%的序列同一性及相同的HCDR3长度,的一组的殖株及其能生产的结合域。包括例如MF8048、MF8056、MF8057、MF8058、MF8078及MF8101。在另一优选的具体例中,本文的抗CD3抗体基于VH1-69的相同的VH V基因节段使用和/或于HCDR3内具有至少80%的同一性及相同的HCDR3长度,更优选为于HCDR3内有90%或最佳为95%的同一性。在另一优选的具体例中,本文的抗CD3抗体基于VH1-69的相同的VH V基因节段使用和/或相较于编码的CDR3节段RGNWNPFDP而言,于HCDR3内具有至少80%的同一性及相同的HCDR3长度,优选为于HCDR3内至少90%的序列同一性及相同的HCDR3长度,更优选为95%或最佳为98%的同一性及相同的HCDR3长度,但有条件是排除包括编码HCDR2序列GFIPVLGTANYAQKFQG、GIIPLFGTITYAQKFQG及SIIPIFGTITYAQKFQG的核酸的殖株,或有条件是排除包括编码VH序列The terms "
的核酸的殖株,或有条件是来自该组的殖株包括核酸,该核酸编码由一种双特异性抗体所组成或经设计而由一种双特异性抗体所组成的HCDR3。术语“超级群(super-cluster)3”或“超级群(supercluster)3”在此使用于意指,基于与该超级群的成员相同的VH V基因节段使用(VH3-23)且于HCDR3内具有至少70%的序列同一性及相同的HCDR3长度,的一组的殖株及其能生产的结合域。包括例如MF8397及MF8562。在另一优选的具体例中,本文的抗CD3抗体基于VH3-23的相同的VH V基因节段使用和/或于HCDR3内具有至少80%的同一性及相同的HCDR3长度,更优选为于HCDR3内有90%或最佳为95%的同一性。在另一优选的具体例中,本文的抗CD3抗体基于VH3-23的相同的VH V基因节段使用和/或相较于编码的CDR3节段DGGYSYGPYWYFDL而言,于HCDR3内具有至少80%的同一性及相同的HCDR3长度,优选为于HCDR3内至少90%的序列同一性及相同的HCDR3长度,更优选为95%或最佳为98%的同一性及相同的HCDR3长度,但有条件是排除包括编码HCDR3序列DGGYSYGPYWYFDL的核酸的殖株,或有条件是排除包括编码VH序列A clone of nucleic acid, or conditionally a clone from the group, comprises nucleic acid encoding HCDR3 consisting of or designed to consist of a bispecific antibody. The terms "
的核酸的殖株,或有条件是来自该组的殖株包括核酸,该核酸编码由一种双特异性抗体所组成或经设计而由一种双特异性抗体所组成的HCDR3。A clone of nucleic acid, or conditionally a clone from the group, comprises nucleic acid encoding HCDR3 consisting of or designed to consist of a bispecific antibody.
术语“超级群(super-cluster)4”或“超级群(supercluster)4”在此使用于意指,基于与该超级群的成员相同的VH V基因节段使用(VH3-9)且于HCDR3内具有至少70%的序列同一性及相同的HCDR3长度,的一组的殖株及其能生产的结合域。包括例如MF8508、MF8998、MF10401及MF10428。在另一优选的具体例中,本文的抗CD3抗体基于VH3-9的相同的VH V基因节段使用和/或于HCDR3内具有至少80%的同一性及相同的HCDR3长度,更优选为于HCDR3内有90%或最佳为95%的同一性。在另一优选的具体例中,本文的抗CD3抗体基于VH3-9的相同的VH V基因节段使用和/或相较于编码的CDR3节段DHRGYGDYEGGGFDY而言,于HCDR3内具有至少80%的同一性及相同的HCDR3长度,优选为于HCDR3内至少90%的序列同一性及相同的HCDR3长度,更优选为95%或最佳为98%的同一性及相同的HCDR3长度,但有条件是排除包括编码HCDR 3序列DHRGYGDYEGGGFDY的核酸的殖株,或有条件是排除包括编码VH序列The term "
的核酸的殖株,或有条件是来自该组的殖株包括核酸,该核酸编码由一种双特异性抗体所组成或经设计而由一种双特异性抗体所组成的HCDR3。A clone of nucleic acid, or conditionally a clone from the group, comprises nucleic acid encoding HCDR3 consisting of or designed to consist of a bispecific antibody.
术语“超级群(super-cluster)7”或“超级群(supercluster)7”在此使用于意指,基于与该超级群的成员相同的VH V基因节段使用(VH5-51)且于HCDR3内具有至少70%的序列同一性及相同的HCDR3长度,的一组的殖株及其能生产的结合域。包括例如MF9249及MF9267。在另一优选的具体例中,本文的抗CD3抗体基于VH5-51的相同的VH V基因节段使用和/或于HCDR3内具有至少80%的同一性及相同的HCDR3长度,更优选为于HCDR3内有90%或最佳为95%的同一性。在另一优选的具体例中,本文的抗CD3抗体基于VH5-51的相同的VH V基因节段使用和/或相较于编码的CDR3节段HIRYFDWSEDYHYYLDV而言,于HCDR3内具有至少80%的同一性及相同的HCDR3长度,优选为于HCDR3内至少90%的序列同一性及相同的HCDR3长度,更优选为95%或最佳为98%的同一性及相同的HCDR3长度。The term "
本发明进一步提供一种包括一可变结构域的双特异性抗体,该可变结构域具有一由下列所编码的VHThe present invention further provides a bispecific antibody comprising a variable domain having a VH encoded by
-V基因节段VH1-69;或- V gene segment VH1-69; or
-一V基因节段VH1-69的变异体,其包括与该V基因节段的序列为至少70%,优选为至少80%,更优选为至少90%且更优选为至少95%的序列同一性;- a variant of the V gene segment VH1-69 comprising at least 70%, preferably at least 80%, more preferably at least 90% and more preferably at least 95% sequence identity to the sequence of the V gene segment sex;
其中该VH进一步包括wherein the VH further comprises
-MF8048、MF8056、MF8057、MF8058、MF8078或MF8101的一HCDR3;- HCDR3 for MF8048, MF8056, MF8057, MF8058, MF8078 or MF8101;
-或该HCDR3的一变异体,其包括与该HCDR3为至少70%的序列同一性及该HCDR3相同的长度。- or a variant of the HCDR3 comprising at least 70% sequence identity to the HCDR3 and the same length as the HCDR3.
在一优选的具体例中,该HCDR3的该变异体包括如该HCDR3相同的长度及与该HCDR3为至少80%的序列同一性,更优选为与该HCDR3的序列为至少90%且更优选为至少95%的序列同一性。In a preferred embodiment, the variant of the HCDR3 comprises the same length as the HCDR3 and at least 80% sequence identity with the HCDR3, more preferably at least 90% with the HCDR3 sequence and more preferably At least 95% sequence identity.
在一些具体例中,该双特异性抗体不具有由下列所编码的VH:V基因节段VH1-69;或V基因节段VH1-69的变异体其具有HCDR2序列GFIPVLGTANYAQKFQG、或GIIPLFGTITYAQKFQG或SIIPIFGTITYAQKFQG。In some embodiments, the bispecific antibody does not have a VH encoded by: V gene segment VH1-69; or a variant of V gene segment VH1-69 having the HCDR2 sequence GFIPVLGTANYAQKFQG, or GIIPLFGTITYAQKFQG, or SIIPIFGTITYAQKFQG.
在一些具体例中,该双特异性抗体不具有由下列所编码的VH:V基因节段VH1-69;或V基因节段VH1-69的变异体其具有VH序列In some embodiments, the bispecific antibody does not have a VH encoded by: V gene segment VH1-69; or a variant of V gene segment VH1-69 that has a VH sequence
本发明进一步提供一种包括一可变结构域的双特异性抗体,该可变结构域具有一由下列所编码的VHThe present invention further provides a bispecific antibody comprising a variable domain having a VH encoded by
-V基因节段VH3-23;或- V gene segment VH3-23; or
-一V基因节段VH2-23的变异体,其包括与该V基因节段的序列为至少70%,优选为至少80%,更优选为至少90%且更优选为至少95%的序列同一性;- a variant of the V gene segment VH2-23 comprising at least 70%, preferably at least 80%, more preferably at least 90% and more preferably at least 95% sequence identity to the sequence of the V gene segment sex;
其中该VH进一步包括wherein the VH further comprises
-MF8397;或MF8562的一HCDR3;-MF8397; or -HCDR3 of MF8562;
-或该HCDR3的一变异体,其包括与该HCDR3为至少70%的序列同一性及该HCDR3相同的长度。- or a variant of the HCDR3 comprising at least 70% sequence identity to the HCDR3 and the same length as the HCDR3.
在优选的具体例中,该HCDR3的该变异体包括如该HCDR3相同的长度及与该HCDR3为至少80%的序列同一性,更优选为与该HCDR3的序列为至少90%,更优选为至少93%且更优选为至少95%的序列同一性。In a preferred embodiment, the variant of the HCDR3 comprises the same length as the HCDR3 and at least 80% sequence identity with the HCDR3, more preferably at least 90% with the sequence of the HCDR3, more preferably at least 93% and more preferably at least 95% sequence identity.
在一些具体例中,该双特异性抗体不具有由下列所编码的VH:V基因节段VH3-23;或V基因节段VH3-23的变异体其具有HCDR3序列DGGYSYGPYWYFD。In some embodiments, the bispecific antibody does not have a VH encoded by: V gene segment VH3-23; or a variant of V gene segment VH3-23 having the HCDR3 sequence DGGYSYGPYWYFD.
在一些具体例中,该双特异性抗体不具有由下列所编码的VH:V基因节段VH3-23;或V基因节段VH3-23的变异体其具有VH序列In some embodiments, the bispecific antibody does not have a VH encoded by: V gene segment VH3-23; or a variant of V gene segment VH3-23 that has a VH sequence
本发明进一步提供一种包括一可变结构域的双特异性抗体,该可变结构域具有一由下列所编码的VHThe present invention further provides a bispecific antibody comprising a variable domain having a VH encoded by
-V基因节段VH3-9;或- V gene segment VH3-9; or
-一V基因节段VH3-9的变异体,其包括与该V基因节段的序列为至少70%,优选为至少80%,更优选为至少90%且更优选为至少95%的序列同一性;- a variant of the V gene segment VH3-9 comprising at least 70%, preferably at least 80%, more preferably at least 90% and more preferably at least 95% sequence identity to the sequence of the V gene segment sex;
其中该VH进一步包括wherein the VH further comprises
-MF8508;MF8998;MF1041;或MF10428的一HCDR3;- MF8508; MF8998; MF1041; or a HCDR3 of MF10428;
-或该HCDR3的一变异体,其包括与该HCDR3为至少70%的序列同一性及该HCDR3相同的长度。- or a variant of the HCDR3 comprising at least 70% sequence identity to the HCDR3 and the same length as the HCDR3.
在优选的具体例中,该HCDR3的该变异体包括如该HCDR3相同的长度及与该HCDR3为至少80%的序列同一性,更优选为与该HCDR3的序列为至少90%且更优选为至少95%的序列同一性。In a preferred embodiment, the variant of the HCDR3 comprises the same length as the HCDR3 and at least 80% sequence identity with the HCDR3, more preferably at least 90% and more preferably at least 90% of the sequence with the HCDR3 95% sequence identity.
在一些具体例中,该双特异性抗体不具有由下列所编码的VH:V基因节段VH3-9;或V基因节段VH3-9的变异体其具有HCDR3序列DHRGYGDYEGGGFDY。In some embodiments, the bispecific antibody does not have a VH encoded by: V gene segment VH3-9; or a variant of V gene segment VH3-9 having the HCDR3 sequence DHRGYGDYEGGGFDY.
在一些具体例中,该双特异性抗体不具有由下列所编码的VH:V基因节段VH3-9;或V基因节段VH3-9的变异体其具有VH序列In some embodiments, the bispecific antibody does not have a VH encoded by: V gene segment VH3-9; or a variant of V gene segment VH3-9 that has a VH sequence
本发明进一步提供一种包括一可变结构域的双特异性抗体,该可变结构域具有一由下列所编码的VHThe present invention further provides a bispecific antibody comprising a variable domain having a VH encoded by
-V基因节段VH5-51;或- V gene segment VH5-51; or
-一V基因节段VH5-51的变异体,其包括与该V基因节段的序列为至少70%,优选为至少80%,更优选为至少90%且更优选为至少95%的序列同一性;- a variant of the V gene segment VH5-51 comprising at least 70%, preferably at least 80%, more preferably at least 90% and more preferably at least 95% sequence identity to the V gene segment sex;
其中该VH进一步包括wherein the VH further comprises
-MF9249或MF9267的一HCDR3;- HCDR3 of MF9249 or MF9267;
-或该HCDR3的一变异体,其包括与该HCDR3为至少70%的序列同一性及该HCDR3相同的长度。- or a variant of the HCDR3 comprising at least 70% sequence identity to the HCDR3 and the same length as the HCDR3.
在优选的具体例中,该HCDR3的该变异体包括如该HCDR3相同的长度及与该HCDR3为至少80%的序列同一性,更优选为与该HCDR3的序列为至少90%且更优选为至少95%的序列同一性。In a preferred embodiment, the variant of the HCDR3 comprises the same length as the HCDR3 and at least 80% sequence identity with the HCDR3, more preferably at least 90% and more preferably at least 90% of the sequence with the HCDR3 95% sequence identity.
一种如本文所定义的本发明提供的双特异性抗体优选不是如PCT/NL2019/050199所定义的包括一CD3结合可变结构域的双特异性抗体。A bispecific antibody provided by the invention as defined herein is preferably not a bispecific antibody comprising a CD3 binding variable domain as defined in PCT/NL2019/050199.
本发明进一步提供一种由下列所编码的VHThe present invention further provides a VH encoded by
-V基因节段VH1-69;或- V gene segment VH1-69; or
-一V基因节段VH1-69的变异体,其包括与该V基因节段的序列为至少70%,优选为至少80%,更优选为至少90%且更优选为至少95%的序列同一性;- a variant of the V gene segment VH1-69 comprising at least 70%, preferably at least 80%, more preferably at least 90% and more preferably at least 95% sequence identity to the sequence of the V gene segment sex;
其中该VH进一步包括wherein the VH further comprises
-MF8048、MF8056、MF8057、MF8058、MF8078或MF8101的一HCDR3;- HCDR3 for MF8048, MF8056, MF8057, MF8058, MF8078 or MF8101;
-或该HCDR3的一变异体,其包括与该HCDR3为至少70%的序列同一性及该HCDR3相同的长度。- or a variant of the HCDR3 comprising at least 70% sequence identity to the HCDR3 and the same length as the HCDR3.
在优选的具体例中,该HCDR3的该变异体包括如该HCDR3相同的长度及与该HCDR3为至少80%的序列同一性,更优选为与该HCDR3的序列为至少90%且更优选为至少95%的序列同一性。In a preferred embodiment, the variant of the HCDR3 comprises the same length as the HCDR3 and at least 80% sequence identity with the HCDR3, more preferably at least 90% and more preferably at least 90% of the sequence with the HCDR3 95% sequence identity.
在一些具体例中,该VH不是由下列所编码的VH:V基因节段VH1-69;或V基因节段VH1-69的变异体其具有HCDR2序列GFIPVLGTANYAQKFQG、或GIIPLFGTITYAQKFQG或SIIPIFGTITYAQKFQG。In some embodiments, the VH is not a VH encoded by: V gene segment VH1-69; or a variant of V gene segment VH1-69 having the HCDR2 sequence GFIPVLGTANYAQKFQG, or GIIPLFGTITYAQKFQG, or SIIPIFGTITYAQKFQG.
在一些具体例中,该VH不是由下列所编码的VH:V基因节段VH1-69;或V基因节段VH1-69的变异体其具有VH序列In some embodiments, the VH is not a VH encoded by: V gene segment VH1-69; or a variant of V gene segment VH1-69 having a VH sequence
本发明进一步提供一种由下列所编码的VHThe present invention further provides a VH encoded by
-V基因节段VH3-23;或- V gene segment VH3-23; or
-一V基因节段VH2-23的变异体,其包括与该V基因节段的序列为至少70%,优选为至少80%,更优选为至少90%且更优选为至少95%的序列同一性;- a variant of the V gene segment VH2-23 comprising at least 70%, preferably at least 80%, more preferably at least 90% and more preferably at least 95% sequence identity to the sequence of the V gene segment sex;
其中该VH进一步包括wherein the VH further comprises
-MF8397;或MF8562的一HCDR3;-MF8397; or -HCDR3 of MF8562;
-或该HCDR3的一变异体,其包括与该HCDR3为至少70%的序列同一性及该HCDR3相同的长度。- or a variant of the HCDR3 comprising at least 70% sequence identity to the HCDR3 and the same length as the HCDR3.
在优选的具体例中,该HCDR3的该变异体包括如该HCDR3相同的长度及与该HCDR3为至少80%的序列同一性,更优选为与该HCDR3的序列为至少90%,更优选为至少93%且更优选为至少95%的序列同一性。In a preferred embodiment, the variant of the HCDR3 comprises the same length as the HCDR3 and at least 80% sequence identity with the HCDR3, more preferably at least 90% with the sequence of the HCDR3, more preferably at least 93% and more preferably at least 95% sequence identity.
在一些具体例中,该VH不是由下列所编码的VH:V基因节段VH3-23;或V基因节段VH3-23的变异体其具有HCDR3序列DGGYSYGPYWYFDL。In some embodiments, the VH is not a VH encoded by: V gene segment VH3-23; or a variant of V gene segment VH3-23 having the HCDR3 sequence DGGYSYGPYWYFDL.
在一些具体例中,该VH不是由下列所编码的VH:V基因节段VH3-23;或V基因节段VH3-23的变异体其具有VH序列In some embodiments, the VH is not a VH encoded by: V gene segment VH3-23; or a variant of V gene segment VH3-23 having a VH sequence
本发明进一步提供一种由下列所编码的VHThe present invention further provides a VH encoded by
-V基因节段VH3-9;或- V gene segment VH3-9; or
-一V基因节段VH3-9的变异体,其包括与该V基因节段的序列为至少70%,优选为至少80%,更优选为至少90%且更优选为至少95%的序列同一性;- a variant of the V gene segment VH3-9 comprising at least 70%, preferably at least 80%, more preferably at least 90% and more preferably at least 95% sequence identity to the sequence of the V gene segment sex;
其中该VH进一步包括wherein the VH further comprises
-MF8508;MF8998;MF10401;或MF10428的一HCDR3;- MF8508; MF8998; MF10401; or a HCDR3 of MF10428;
-或该HCDR3的一变异体,其包括与该HCDR3为至少70%的序列同一性及该HCDR3相同的长度。- or a variant of the HCDR3 comprising at least 70% sequence identity to the HCDR3 and the same length as the HCDR3.
在优选的具体例中,该HCDR3的该变异体包括如该HCDR3相同的长度及与该HCDR3为至少80%的序列同一性,更优选为与该HCDR3的序列为至少90%且更优选为至少95%的序列同一性。In a preferred embodiment, the variant of the HCDR3 comprises the same length as the HCDR3 and at least 80% sequence identity with the HCDR3, more preferably at least 90% and more preferably at least 90% of the sequence with the HCDR3 95% sequence identity.
在一些具体例中,该VH不是由下列所编码的VH:V基因节段VH3-9;或V基因节段VH3-9的变异体其具有HCDR3序列DHRGYGDYEGGGFDY。In some embodiments, the VH is not a VH encoded by: V gene segment VH3-9; or a variant of V gene segment VH3-9 having the HCDR3 sequence DHRGYGDYEGGGFDY.
在一些具体例中,该VH不是由下列所编码的VH:V基因节段VH3-9;或V基因节段VH3-9的变异体其具有VH序列In some embodiments, the VH is not a VH encoded by: V gene segment VH3-9; or a variant of V gene segment VH3-9 having a VH sequence
本发明进一步提供一种由下列所编码的VHThe present invention further provides a VH encoded by
-V基因节段VH5-51;或- V gene segment VH5-51; or
-一V基因节段VH5-51的变异体,其包括与该V基因节段的序列为至少70%,优选为至少80%,更优选为至少90%且更优选为至少95%的序列同一性;- a variant of the V gene segment VH5-51 comprising at least 70%, preferably at least 80%, more preferably at least 90% and more preferably at least 95% sequence identity to the V gene segment sex;
其中该VH进一步包括wherein the VH further comprises
-MF9249或MF9267的一HCDR3;- HCDR3 of MF9249 or MF9267;
-或该HCDR3的一变异体,其包括与该HCDR3为至少70%的序列同一性及该HCDR3相同的长度。- or a variant of the HCDR3 comprising at least 70% sequence identity to the HCDR3 and the same length as the HCDR3.
在优选的具体例中,该HCDR3的该变异体包括如该HCDR3相同的长度及与该HCDR3为至少80%的序列同一性,更优选为与该HCDR3的序列为至少90%且更优选为至少95%的序列同一性。In a preferred embodiment, the variant of the HCDR3 comprises the same length as the HCDR3 and at least 80% sequence identity with the HCDR3, more preferably at least 90% and more preferably at least 90% of the sequence with the HCDR3 95% sequence identity.
一种如本文所定义的本发明提供的VH优选不是如PCT/NL2019/050199所定义的一CD3结合可变结构域的VH。A VH provided by the invention as defined herein is preferably not a CD3 binding variable domain VH as defined in PCT/NL2019/050199.
还提供一种抗原结合蛋白或抗体,优选为一种双特异性抗体,其中此类CDRs与如所主张的此类CDRs有70%,优选为80%,更优选为90%的同一性。在优选的具体例中,该抗原结合蛋白或抗体为一种双特异性抗体,其包括的CDRs,就所主张的此类CDRs而言具有最多2个,优选最多1个且更优选为最多0个氨基酸残基变异、插入、取代、缺失或加入。Also provided is an antigen binding protein or antibody, preferably a bispecific antibody, wherein such CDRs are 70%, preferably 80%, more preferably 90% identical to such CDRs as claimed. In a preferred embodiment, the antigen binding protein or antibody is a bispecific antibody comprising CDRs having at most 2, preferably at most 1 and more preferably at most 0 of such CDRs as claimed amino acid residue variation, insertion, substitution, deletion or addition.
与抗体的随机、非特异性附着不同,经抗体的抗原结合通常透过抗体的互补区域及抗原和可变结构域二者的特定三维结构被媒介而允许这些两种结构精准地结合在一起(类似锁和钥匙的交互作用)。因一种抗体通常仅识别抗原的一表位,且此一表位也可存在于其他蛋白,如本发明结合CD3或CLEC12A的抗体也可识别其他蛋白,如果这些其他蛋白含有相同的表位。因此,术语“结合”不排除该抗体与含有相同表位的另一种蛋白或多种蛋白的结合。本发明的抗体内的结合CD3的重/轻链组合不会与出生后、优选成年人类的细胞膜上的其他蛋白结合。本发明结合CLEC12A、EGFR、PD-L1或肿瘤细胞抗原的重/轻链组合不会与出生后、优选成年人类的细胞膜上的其他蛋白结合。合适的肿瘤抗原特异臂揭露于PCT/NL2019/050199的内。Unlike the random, nonspecific attachment of antibodies, antigen binding via antibodies is typically mediated through the complementary regions of the antibody and through the specific three-dimensional structure of both the antigen and variable domains allowing these two structures to bind together precisely (similar to lock and key interaction). Because an antibody usually only recognizes one epitope of an antigen, and this epitope may also exist in other proteins, such as the antibody of the present invention that binds to CD3 or CLEC12A can also recognize other proteins, if these other proteins contain the same epitope. Thus, the term "binding" does not exclude binding of the antibody to another protein or proteins containing the same epitope. The CD3-binding heavy/light chain combinations within the antibodies of the invention do not bind to other proteins on postnatal, preferably adult human cell membranes. The heavy/light chain combinations of the invention that bind CLEC12A, EGFR, PD-L1 or tumor cell antigens do not bind to other proteins on the cell membranes of postnatal, preferably adult human beings. Suitable tumor antigen-specific arms are disclosed in PCT/NL2019/050199.
“复数”是指两个或多个。"Plural" means two or more.
如本文所述的抗体的“变异体”可包括抗体的功能部分、衍生物和/或类似物。此包括拟抗体(antibody mimetic)、单一体型(monobody)及适配体(aptamer)。"Variants" of antibodies as described herein may include functional portions, derivatives and/or analogs of antibodies. This includes antibody mimetic, monobody and aptamer.
变异体典型会保持抗体的结合特异性,例如双特异性抗体的特异性。变异体可为如本文所述的结合域、多聚体或抗体的功能部分或衍生物。Variants typically retain the binding specificity of the antibody, eg, the specificity of a bispecific antibody. Variants may be functional parts or derivatives of binding domains, multimers or antibodies as described herein.
如本文所述的结合域、多聚体或抗体的功能部分为一种部分其包括结合如同此种结合域、多聚体或抗体所结合的标靶相同的标靶的可变结构域。A functional portion of a binding domain, multimer or antibody as described herein is a portion that includes a variable domain that binds to the same target as such a binding domain, multimer or antibody binds.
如本文所述的抗体的功能衍生物是一种蛋白,其包括借由链接区域所链接的结合一标靶的一可变结构域及结合第二标靶的一可变结构域。该可变结构域可为可变结构域本身或Fab片段或类可变结构域分子,例如包括经由键接子而链接在一起的VH及VL的单链Fv(scFv)片段。抗体可变结构域或抗体类可变结构域分子可以不同的方式彼此链接。已经描述过各种能结合一个、二个或多个可变结构域的键接子及载体结构。如本文所述的抗原结合蛋白是一种蛋白,其包括至少一个此种可变结构域。在双特异性或多特异性抗原结合蛋白的情况下,此种蛋白包括二个或多个可变结构域,其中的至少二者结合不同的标靶。该可变结构域经由一键接部分而彼此键接。此典型为0-15个,优选为3-12个,更优选为约5-8个氨基酸残基的一段。类可变结构域分子的其他实例是所谓的单域抗体片段。单域抗体片段(sdAb)是具有单一单体可变抗体区域的抗体片段。像完整抗体一样,其能够选择性地结合至特定抗原。分子量仅为12-15kDa,单域抗体片段远小于由两条重蛋白链和两条轻链所构成的普通抗体(150-160kDa),且甚至比Fab片段还要小(~50kDa,一条轻链和半条重链)和单链可变片段(~25kDa,两个可变区,一个来自轻而一个来自重链)。单域抗体本身并不比正常抗体小很多(通常为90-100kDa)。单域抗体片段主要由骆驼科(camelid)中发现的重链抗体进行工程化;这些称为VHH片段一些鱼类也具有仅有重链的抗体(IgNAR,“免疫球蛋白新抗原受体”),从中可获得称为VNAR片段的单域抗体片段。另一方法是将从来自人类或小鼠的常见免疫球蛋白G(IgG)的二聚可变结构域分成单体。尽管对单域抗体的大多数研究目前基于重链可变结构域,但是衍生自轻链的纳米抗体(nanobody)已经显示出能结合至标靶表位。类可变结构域分子的其他非限制性实例是VHH、人类域抗体(Human Domain Antibodies,dAbs)和单抗体(Unibodies)。优选的功能部分为包括可变结构域的部分,该可变结构域包括重链可变区及轻链可变区。这些可变结构域的非限制性实例为F(ab)片段及单链Fv片段。(类)可变结构域键联的双特异性格式是例如与两个不同的scFv结合的人类血清白蛋白(HSA);双特异性迷你抗体其包括透过二聚合模体(motif)或自我缔合(self-associating)的二级结构例如螺旋束或卷曲螺旋,以引起scFv片段的二聚合而结合在一起的两个不同scFv(Morrison(2007)Nat.Biotechnol.25:1233-34)。在WO2009/126920中描述合适的HAS键接子及使scFv偶合至键接子的方法的实例。A functional derivative of an antibody as described herein is a protein comprising a variable domain that binds a target and a variable domain that binds a second target linked by a linking region. The variable domains may be the variable domains themselves or Fab fragments or variable domain-like molecules, eg, single-chain Fv (scFv) fragments comprising VH and VL linked together by a linker. Antibody variable domains or antibody-like variable domain molecules can be linked to each other in different ways. Various linker and carrier structures have been described that can bind one, two or more variable domains. An antigen binding protein as described herein is a protein comprising at least one such variable domain. In the case of bispecific or multispecific antigen binding proteins, such proteins comprise two or more variable domains, at least two of which bind different targets. The variable domains are linked to each other via a linking moiety. This is typically 0-15, preferably 3-12, more preferably a stretch of about 5-8 amino acid residues. Other examples of variable domain-like molecules are so-called single domain antibody fragments. Single domain antibody fragments (sdAbs) are antibody fragments that have a single monomeric variable antibody region. Like intact antibodies, they are capable of selectively binding to specific antigens. The molecular weight is only 12-15kDa, and single-domain antibody fragments are much smaller than ordinary antibodies (150-160kDa) composed of two heavy protein chains and two light chains, and even smaller than Fab fragments (~50kDa, one light chain and half a heavy chain) and a single chain variable fragment (~25 kDa, two variable domains, one from the light and one from the heavy chain). Single domain antibodies themselves are not much smaller than normal antibodies (usually 90-100 kDa). Single domain antibody fragments are engineered primarily from heavy chain antibodies found in the family camelid; these are called VHH fragments Some fish also have heavy chain-only antibodies (IgNAR, "immunoglobulin neoantigen receptor"), from which single-domain antibody fragments called VNAR fragments can be obtained. Another approach is to separate dimeric variable domains from common immunoglobulin G (IgG) from humans or mice into monomers. Although most research on single domain antibodies is currently based on heavy chain variable domains, nanobodies derived from light chains have been shown to bind to target epitopes. Other non-limiting examples of variable domain-like molecules are VHHs, Human Domain Antibodies (dAbs), and Unibodies. Preferred functional moieties are those comprising a variable domain comprising a heavy chain variable region and a light chain variable region. Non-limiting examples of these variable domains are F(ab) fragments and single chain Fv fragments. A bispecific format for (class) variable domain linkage is, for example, human serum albumin (HSA) bound to two different scFvs; bispecific mini-antibodies which include via dimerization motifs (motifs) or self Self-associating secondary structures such as helical bundles or coiled-coils, two different scFvs bound together to cause dimerization of the scFv fragments (Morrison (2007) Nat. Biotechnol. 25:1233-34). Examples of suitable HAS linkers and methods of coupling scFvs to linkers are described in WO2009/126920.
功能衍生物可为拟抗体、多肽、适配体或其的组合。这些蛋白或适配体通常结合至一标靶。本发明的蛋白结合至两个或多个标靶。应该理解的是可通过本技艺已知的方法将这些抗体、拟抗体、多肽及适配体的任何组合链接在一起。例如,在一些具体例中,本发明的结合分子为复合物(conjugate)或融合蛋白。Functional derivatives can be mimetibodies, polypeptides, aptamers, or combinations thereof. These proteins or aptamers typically bind to a target. The proteins of the invention bind to two or more targets. It will be appreciated that any combination of these antibodies, mimetibodies, polypeptides and aptamers can be linked together by methods known in the art. For example, in some embodiments, the binding molecules of the invention are conjugates or fusion proteins.
拟抗体是像抗体一样,可特异性地结合抗原,但与抗体在结构上不相关的多肽。拟抗体通常是摩尔质量约3至20kDa的人工肽或蛋白。拟抗体的非限制性实例为亲和体分子(affibody molecule)(通常基于蛋白A的Z域);阿菲林(affilin)(通常基于γ-B结晶或泛素);黏合素(affimers)(通常基于胱蛋白(Cystatin));阿非廷(affitins)(通常基于来自嗜酸热硫化叶菌(Sulfolobus acidocaldarius)的Sac7d);阿尔法体(alphabody)(通常基于三重螺旋卷曲螺旋(Triple helix coiled coil));抗运载蛋白(anticalin)(通常基于脂质运载蛋白(lipocalin));厄维体(avimer)(通常基于各种膜受体的A域);D ARPin(通常基于锚蛋白重复模体(ankyrin repeat motif));非诺莫(fynomers)(通常基于Fyn 7的SH3域);孔尼兹域(kunitz domain)肽(通常基于各种蛋白酶抑制剂的孔尼兹域);以及单一体型(通常基于纤维接合素(fibronectin)的第III型域)。Mimetic antibodies are polypeptides that, like antibodies, can specifically bind to an antigen but are not structurally related to antibodies. Mimetic antibodies are typically artificial peptides or proteins with a molar mass of about 3 to 20 kDa. Non-limiting examples of mimetics are affibody molecules (usually based on the Z domain of protein A); affinins (usually based on gamma-B crystals or ubiquitin); affimers (usually based on gamma-B crystals or ubiquitin) Cystatin based); affitins (usually based on Sac7d from Sulfolobus acidocaldarius); alphabody (usually based on Triple helix coiled coil) ); anticalins (usually based on lipocalins); avimers (usually based on the A domains of various membrane receptors); D ARPins (usually based on ankyrin repeat motifs ( ankyrin repeat motif)); fynomers (usually based on the SH3 domain of Fyn 7); kunitz domain peptides (usually based on the kunitz domain of various protease inhibitors); and monosomy ( Usually based on the type III domain of fibronectin).
单一体型是合成的结合蛋白,其使用纤维接合素第III型域(FN3)作为分子架构被构造。对于产生标靶结合蛋白而言,单一体型是抗体的替代物。Monoforms are synthetic binding proteins constructed using the fibronectin type III domain (FN3) as the molecular framework. Monotypes are surrogates for antibodies for generating target binding proteins.
单一体型及其它拟抗体通常从组合库中产生,其中使用分子展示和定向进化技术例如噬菌体展示、mRNA展示及酵母菌表面展示,以使部分的架构多样化。Monotypes and other mimetibodies are often generated from combinatorial libraries using molecular display and directed evolution techniques such as phage display, mRNA display, and yeast surface display to diversify the architecture of the moieties.
适配体是结合特定标靶分子的寡核苷酸或肽分子。通常借由从大的随机序列池(sequence pool)选择适配体来创造适配体,但天然适配体还存在于核糖开关(riboswitch)中。适配体可用于基础研究及临床目的二者作为大分子。Aptamers are oligonucleotide or peptide molecules that bind to a specific target molecule. Aptamers are typically created by selecting aptamers from a large random sequence pool, but natural aptamers also exist in riboswitches. Aptamers can be used as macromolecules for both basic research and clinical purposes.
在本案发明说明书以及检附的权利要求书的全文中,词语“包括(comprise)”、“包括(include)”和“具有(having)”以及诸如“包括(comprises)”、“包括(comprising)”、“包括(includes)”和“包括(including)”的变化(variations)将被包容性地解释(interpreted inclusively)。即,在上下文允许的情况下,这些词语意欲用于表达未具体叙述其他的要素或整数的可能包括(possible inclusion)。Throughout the present specification and the appended claims, the words "comprise", "include" and "having" as well as words such as "comprises", "comprising" , "includes" and "including" variations are to be interpreted inclusively. That is, where the context allows, these terms are intended to express the possible inclusion of other elements or integers that are not specifically recited.
如此处所用的冠词“一(a)”和“一(an)”是指该冠词的语法制品有一个或多于一个(即一个或至少一个)。举例来说,一要素可以表示一个要素或多于一个要素。The articles "a (a)" and "an (an)" as used herein mean that the grammatical article of the article has one or more than one (ie, one or at least one). For example, an element may represent one element or more than one element.
本发明的抗体优选为一种双特异性或多特异性抗体。该双特异性或多特异性抗体优选至少结合人类CD3。The antibody of the present invention is preferably a bispecific or multispecific antibody. The bispecific or multispecific antibody preferably binds at least human CD3.
本发明的抗原结合蛋白或抗体优选为一种双或多特异性抗原结合蛋白或抗体。该双或多特异性抗原结合蛋白或抗体优选至少结合人类CD3,此外优选结合人类肿瘤细胞上表现的至少一表面分子。在优选的具体例中,该双或多特异性抗原结合蛋白或抗体结合BCMA、CD19、CD20、CD30、CD33、CD38、CD44、CD123、CD138、CEA、CLEC12A、CS-1、EGFR、EGFRvIII、EPCAM、DLL3、LGR5、MSLN、PD-L1、FOLR1、FOLR3、HER2、HM1.24、MCSP或PSMA。在一特别佳的具体例中,该双特异性抗体结合CLEC12A。在一个特别佳的具体例中,该双特异性抗体结合CD3、PD-L1及EGFR。The antigen binding protein or antibody of the present invention is preferably a bi- or multispecific antigen binding protein or antibody. The bi- or multispecific antigen binding protein or antibody preferably binds to at least human CD3, and further preferably binds to at least one surface molecule expressed on human tumor cells. In a preferred embodiment, the bi- or multispecific antigen-binding protein or antibody binds to BCMA, CD19, CD20, CD30, CD33, CD38, CD44, CD123, CD138, CEA, CLEC12A, CS-1, EGFR, EGFRvIII, EPCAM , DLL3, LGR5, MSLN, PD-L1, FOLR1, FOLR3, HER2, HM1.24, MCSP or PSMA. In a particularly preferred embodiment, the bispecific antibody binds CLEC12A. In a particularly preferred embodiment, the bispecific antibody binds CD3, PD-L1 and EGFR.
当使用于文本中,术语“CLEC12A”是指C型凝集素域家族12成员A。CLEC12A还称为C型凝集素蛋白CLL-1;MICL;树突细胞缔合凝集素2;C型凝集素超家族;骨髓抑制性C型凝集素样受体;C型凝集素样分子1;DCAL2;CLL1;C型凝集素样分子1;DCAL-2;杀手细胞凝集素样受体次家族L,成员1(KLRL1);CD371(分化群371)(Bakker A.等人的Cancer Res.2004,64,p884350;GenBankTM登录序号:AY547296;Zhang W.等人的GenBankTM登录序号:AF247788;A.S.Marshall等人的J Biol Chem 2004,279,p14792-802;GenBankTM登录序号:AY498550;Y.Han等人的Blood 2004,104,p2858 66;H.Floyd等人的GenBankTM登录序号:AY426759;C.H.Chen等人的Blood 2006,107,p145967)。Ids:HGNC:31713;Entrez Gene:160364;Ensembl:ENSG00000172322;OMIM:612088;UniProtKB:Q5QGZ9。As used in the text, the term "CLEC12A" refers to C-type
CLEC12A是一种抗原其表现在急性骨髓性白血病(AML)中的白血病芽细胞上及白血病干细胞上,包括CD34阴性或CD34低表现的白血病干细胞(边缘族群(sidepopulation))(A.B.Bakker等人的Cancer Res 2004,64,p844350;Van Rhenen等人的2007Blood 110:2659;Moshaver等人的2008 Stem Cells 26:3059),以及于骨髓发育不良症候群(MDS)中(同前的Bakker等人的2004,及Toff-Peterson等人,Br.J.Haematol.175(3):393-401,2016)。CLEC12A的表现另外被认为局限于造血谱系,特别是末梢血液及骨髓内的骨髓谱系,即颗粒球、单核球及树突细胞前驱体。更重要地是,CLEC12A不存在于正常的造血干细胞上。在本文谈及CLEC12A的处,其是指人类CLEC12A(SEQ ID NO:1;图19),除非另有特别说明。CLEC12A is an antigen expressed on leukemia bud cells and leukemia stem cells in acute myeloid leukemia (AML), including CD34-negative or CD34-low expressing leukemia stem cells (sidepopulation) (Cancer by A.B. Bakker et al. Res 2004, 64, p844350; Van Rhenen et al. 2007 Blood 110:2659; Moshaver et al. 2008 Stem Cells 26:3059), and in myelodysplastic syndrome (MDS) (ibid. Bakker et al. 2004, and Toff-Peterson et al., Br. J. Haematol. 175(3):393-401, 2016). The expression of CLEC12A is additionally believed to be restricted to the hematopoietic lineage, particularly the peripheral blood and myeloid lineage within the bone marrow, ie granule spheres, monocytes and dendritic cell precursors. More importantly, CLEC12A is not present on normal hematopoietic stem cells. Where reference is made herein to CLEC12A, it refers to human CLEC12A (SEQ ID NO: 1; Figure 19) unless specifically stated otherwise.
术语“CLEC12A”意指本文所述的所有变异体(例如剪接和突变)及其保有骨髓表现解析(在表面表现电平及mRNA电平两者上)的同功型,包括如Bakker等人的Cancer Res2004,64,p8443-50及Marshall 2004-J Biol Chem 279(15),p14792-802乙文中所述者。虽然主要提供登录序号作为另外的识别方法,但实际的蛋白序列可能,譬如因为编码基因突变而变化,例如在一些癌症及类似癌症中发生的此类。The term "CLEC12A" means all variants described herein (eg, spliced and mutated) and their isoforms that retain resolution of myeloid expression (both at surface expression and mRNA levels), including those of Bakker et al. Cancer Res 2004, 64, p8443-50 and as described in Marshall 2004-J Biol Chem 279(15), p14792-802B. While accession numbers are primarily provided as an additional method of identification, the actual protein sequence may vary, eg, due to mutations in the encoding gene, such as occurs in some cancers and the like.
术语“CD3”(分化群3)是指由CD3γ链(SwissProt P09693)、CD3δ链(SwissProtP04234)、CD3ε链(SwissProt P07766)及CD3ζ链同型二聚体(SwissProt P20963)组成的蛋白错合物(complex)。CD3ε以各种别名而闻名,其中一些为:“CD3e分子,ε(CD3-TCR错合物)”;“CD3e抗原,ε多肽(TiT3错合物)”;T细胞表面抗原T3/Leu-4ε链;T3E;T细胞抗原受体错合物,T3的ε亚单位;CD3e抗原;CD3-ε3;IMD18;TCRE。CD3E基因的Ids为HGNC:1674;EntrezGene:916;Ensembl:ENSG00000198851;OMIM:186830及UniProtKB:P07766。这些链与T细胞受体(TCR)及ζ链缔合以形成TCR错合物,其在有丝分裂信号传导(mitogenic signaling)的时能在T淋巴细胞内产生活化信息。CD3表现于T细胞及NK T细胞上。在本文谈及CD3的处,其是指人类CD3(SEQ ID NO:2-5;图20),除非另有特别说明。The term "CD3" (cluster of differentiation 3) refers to a protein complex consisting of a CD3γ chain (SwissProt P09693), a CD3δ chain (SwissProt P04234), a CD3ε chain (SwissProt P07766) and a CD3ζ chain homodimer (SwissProt P20963). ). CD3ε is known by various aliases, some of which are: "CD3e molecule, epsilon (CD3-TCR complex)"; "CD3e antigen, epsilon polypeptide (TiT3 complex)"; T cell surface antigen T3/Leu-4ε chain; T3E; T cell antigen receptor complex, epsilon subunit of T3; CD3e antigen; CD3-ε3; IMD18; TCRE. The Ids of the CD3E gene are HGNC: 1674; EntrezGene: 916; Ensembl: ENSG00000198851; OMIM: 186830 and UniProtKB: P07766. These chains associate with the T cell receptor (TCR) and the zeta chain to form TCR complexes that generate activation messages in T lymphocytes upon mitogenic signaling. CD3 is expressed on T cells and NK T cells. Where CD3 is referred to herein, it refers to human CD3 (SEQ ID NOs: 2-5; Figure 20) unless specifically stated otherwise.
BCMA还称为肿瘤坏死因子受体超家族,成员17(TNFRSF17);TNFRSF13A2;B细胞成熟抗原;BCM;B细胞成熟因子;B细胞成熟蛋白;CD269或CD269抗原。Ids:HGNC:11913;EntrezGene:608;Ensembl:ENSG00000048462;OMIM:109545;UniProtKB:Q02223。BCMA is also known as the tumor necrosis factor receptor superfamily, member 17 (TNFRSF17); TNFRSF13A2; B cell maturation antigen; BCM; B cell maturation factor; B cell maturation protein; CD269 or CD269 antigen. Ids: HGNC: 11913; EntrezGene: 608; Ensembl: ENSG00000048462; OMIM: 109545; UniProtKB: Q02223.
CD19还称为CD19分子;T细胞表面抗原Leu-12;CD19抗原;CvID3;分化抗原CD19;B4;B淋巴细胞表面抗原B4;B淋巴细胞抗原CD19。Ids:HGNC:1633;Entrez Gene:930;Ensembl:ENSG00000177455;OMIM:107265;UniProtKB:P15391。CD19 is also known as CD19 molecule; T cell surface antigen Leu-12; CD19 antigen; CvID3; differentiation antigen CD19; B4; B lymphocyte surface antigen B4; B lymphocyte antigen CD19. Ids: HGNC: 1633; Entrez Gene: 930; Ensembl: ENSG00000177455; OMIM: 107265; UniProtKB: P15391.
CD20还称为穿膜4-域,次家族A,成员1(MS4A1);MS4A2;CD20;S7;白血球表面抗原Leu-16;B淋巴细胞抗原CD20;Bp35;B淋巴细胞细胞表面抗原B1;CD20抗原;CD20受体;CVID5;B淋巴细胞表面抗原B1;B1;穿膜4-域次家族A成员1;LEU-16。Ids:HGNC:7315;EntrezGene:931;Ensembl:ENSG00000156738;OMIM:112210;UniProtKB:P11836。CD20 also known as transmembrane 4-domain, subfamily A, member 1 (MS4A1); MS4A2; CD20; S7; leukocyte surface antigen Leu-16; B lymphocyte antigen CD20; Bp35; B lymphocyte cell surface antigen B1; CD20 Antigen; CD20 receptor; CVID5; B lymphocyte surface antigen B1; B1; transmembrane 4-domain
CD30还称为肿瘤坏死因子受体超家族,成员8(TNFRSF8);Ki-1抗原;CD30;Ki-1;D1S166E;细胞介素受体CD30;淋巴细胞活化抗原CD30;肿瘤坏死因子受体超家族成员8;CD30L受体;CD30抗原。Ids:HGNC:11923;Entrez Gene:943;Ensembl:ENSG00000120949;OMIM:153243;UniProtKB:P28908。CD30 is also known as tumor necrosis factor receptor superfamily, member 8 (TNFRSF8); Ki-1 antigen; CD30; Ki-1; D1S166E;
CD33还称为CD33分子;SIGLEC-3;CD33抗原(Gp67);骨髓细胞表面抗原CD33;唾液酸结合Ig样凝集素3;Siglec-3;SIGLEC3;CD33抗原及gp67。Ids:HGNC:1659;Entrez Gene:945;Ensembl:ENSG00000105383;OMIM:159590;UniProtKB:P20138。CD33 is also known as CD33 molecule; SIGLEC-3; CD33 antigen (Gp67); myeloid cell surface antigen CD33; sialic acid-binding Ig-
CD38还称为CD38分子;T10;CD38抗原(P45);CADPr水解酶1;ADP-核糖基环化酶1;ADP-核糖基环化酶/环ADP-核糖水解酶;NAD(+)核苷酶;EC 3.2.2.5;环ADP-核糖水解酶1;CD38抗原。Ids:HGNC:1667;Entrez Gene:952;Ensembl:ENSG00000004468;OMIM:107270;UniProtKB:P28907。CD38 is also known as CD38 molecule; T10; CD38 antigen (P45);
CD44还称为CD44分子(印度血型);IN;MDU2;CD44抗原(归巢(Homing)功能及印度血型系统);MDU3;CDW44;MIC4;CSPG8;硫酸软骨素蛋白多醣8;HCELL;造血细胞E-及L-选滞蛋白配体;MC56;细胞外基质受体III;Pgp1;硫酸乙酰肝素(Heparan Sulfate)蛋白多醣;细胞表面醣蛋白CD44;玻糖醛酸受体;表皮蛋白聚糖(epican);吞噬细胞醣蛋白1;归巢功能及印度血型系统;ECMR-III;CDw44;HUTCH-I;表皮蛋白聚糖(Epican);LHR;PGP-1;CD44抗原;PGP-I;CP90淋巴细胞归巢/黏着受体;吞噬细胞醣蛋白I;爱马仕(Hermes)抗原。Ids:HGNC:1681;Entrez Gene:960;Ensembl:ENSG00000026508;OMIM:107269;UniProtKB:P16070。CD44 is also known as CD44 molecule (Indian blood group); IN; MDU2; CD44 antigen (Homing function and Indian blood group system); MDU3; CDW44; MIC4; CSPG8;
CD123还称为细胞分裂周期123;细胞分裂周期123同源物;C10orf7;细胞分裂周期蛋白123同源物;D 123;蛋白D123;HT-1080;CCEP123;PZ32;CEP89;细胞分裂周期123同源物(酿酒酵母菌(S.Cerevisiae));FLJ14640;染色体10开读框7。Ids:HGNC:16827;EntrezGene:8872;Ensembl:ENSG00000151465;OMIM:615470;UniProtKB:O75794。CD123 is also known as cell division cycle 123; cell division cycle 123 homolog; C10orf7; cell division cycle 123 homolog; D 123; protein D123; HT-1080; CCEP123; PZ32; CEP89; cell division cycle 123 homolog (S. Cerevisiae); FLJ14640;
CD138还称为多配体蛋白聚糖1(Syndecan 1)(SCD1);CD138;SDC;硫酸乙酰肝素蛋白多醣纤维母细胞生长因子受体;多配体蛋白聚糖蛋白多醣1(Syndecan Proteoglycan1);多配体蛋白聚糖;SYND1;多配体蛋白聚糖-1;CD138抗原。Ids:HGNC:10658;EntrezGene:6382;Ensembl:ENSG00000115884;OMIM:186355;UniProtKB:P18827。CD138 is also known as Syndecan 1 (SCD1); CD138; SDC; Heparan sulfate proteoglycan fibroblast growth factor receptor; Syndecan Proteoglycan1; Syndecan; SYND1; Syndecan-1; CD138 antigen. Ids: HGNC: 10658; EntrezGene: 6382; Ensembl: ENSG00000115884; OMIM: 186355; UniProtKB: P18827.
CEA还称为癌胚抗原相关的细胞黏着分子5(CEACAM5);胎便抗原100;CD66e;癌胚抗原;CD66e抗原。Ids:HGNC:1817;Entrez Gene:1048;Ensembl:ENSG00000105388;OMIM:114890;UniProtKB:P06731。CEA is also known as carcinoembryonic antigen-associated cell adhesion molecule 5 (CEACAM5);
EGFR还称为表皮生长因子受体;红血球胚细胞白血病病毒(V-Erb-B)致癌基因同源物(禽);ERBB1;PIG61;原致癌基C-ErbB-1;禽红血球胚细胞白血病病毒(V-Erb-B)致癌基因同源物;受体酪胺酸蛋白激酶ErbB-1;细胞生长抑制蛋白40;细胞增殖诱发蛋白61;HER1;mENA;EC 2.7.10.1;EC 2.7.10;表皮生长因子受体(禽红血球胚细胞白血病病毒(V-Erb-B)致癌基因同源物)。Ids:HGNC:3236;Entrez Gene:1956;Ensembl:ENSG00000146648;OMIM:131550;UniProtKB:P00533。EGFR is also known as epidermal growth factor receptor; erythroblast leukemia virus (V-Erb-B) oncogene homolog (avian); ERBB1; PIG61; proto-oncogene C-ErbB-1; avian erythroblast leukemia virus (V-Erb-B) oncogene homolog; receptor tyrosine protein kinase ErbB-1;
EGFRvIII为常见的EGFR变异体(Oncogene.2013 May 23;32(21):2670-81.doi:10.1038/onc.2012.280.Epub 2012 Jul 16)。EGFRvIII is a common EGFR variant (Oncogene. 2013 May 23; 32(21): 2670-81. doi: 10.1038/onc. 2012.280. Epub 2012 Jul 16).
δ样3(Delta like 3)(DLL3)还称为δ样3(Delta-Like 3));果蝇δ同源物3;Delta3;δ(果蝇)样3;SCDO1。DLL3的Ids为:HGNC:2909;Entrez Gene:10683;Ensembl:ENSG00000090932;OMIM:602768及UniProtKB:Q9NYJ7。Delta like 3 (DLL3) is also known as Delta-Like 3);
LGR5是含有富含白胺酸重复的G蛋白偶联受体5(Leucine-Rich RepeatContaining G Protein-Coupled Receptor 5)。该基因或蛋白的替代名称为含有富含白胺酸重复的G蛋白偶联受体5;含有富含白胺酸重复的G蛋白偶联受体5;G蛋白偶联受体HG38;G蛋白偶联受体49;G蛋白偶联受体67;GPR67;GPR49;孤儿G蛋白偶联受体HG38(Orphan GProtein-Coupled Receptor HG38);G蛋白偶联受体49;GPR49;HG38及FEX。本发明的结合LGR5的蛋白或抗体结合人类LGR5。由于人类和其它哺乳动物异种同源物(ortholog)之间的序列及三级结构的相似性,本发明的LGR5结合蛋白或抗体也可结合这样的异种同源物,但不一定如此。人类LGR5蛋白和编码其的基因的数据库登录序号为(NC_000012.12;NT_029419.13;NC_018923.2;NP_001264155.1;NP_001264156.1;NP_003658.1)。LGR5 is a Leucine-Rich Repeat Containing G Protein-Coupled Receptor 5 (Leucine-Rich Repeat Containing G Protein-Coupled Receptor 5). Alternative names for this gene or protein are leucine-rich repeat-containing G protein-coupled
MSLN或间皮素(mesothelin)还称为间皮素(Mesothelin);初前巨核细胞增效因子(Pre-Pro-Megakaryocyte-Potentiating Factor);CAK1抗原;MPF;可溶性MPF间皮素相关蛋白;巨核细胞增效因子及SMRP。MSLN的Ids为:HGNC:7371;Entrez Gene:10232;Ensembl:ENSG00000102854;OMIM:601051;UniProtKB:Q13421。MSLN or mesothelin Also known as Mesothelin; Pre-Pro-Megakaryocyte-Potentiating Factor; CAK1 Antigen; MPF; Soluble MPF Mesothelin-Associated Protein; Cell potentiation factor and SMRP. The Ids of MSLN are: HGNC: 7371; Entrez Gene: 10232; Ensembl: ENSG00000102854; OMIM: 601051; UniProtKB: Q13421.
叶酸受体1还称为FOLR1;叶酸受体1;卵巢肿瘤相关抗原MOv18;成人叶酸结合蛋白;叶酸受体,成人;KB细胞FBP;FR-α;FOLR;FBP;叶酸结合蛋白;及叶酸受体1。FOLR1的Ids为HGNC:3791;Entrez Gene:2348;Ensembl:ENSG00000110195;OMIM:136430;UniProtKB:P15328。
叶酸受体3还称为FOLR3;叶酸受体3(γ);FR-γ;叶酸受体3;γ-HFR;及FR-G。FOLR3的Ids为HGNC:3795;Entrez Gene:2352;Ensembl:ENSG00000110203;OMIM:602469;及UniProtKB:P41439。
EPCAM还称为上皮细胞黏着分子;EGP40;M4S1;ESA;MIC18;KS1/4;肿瘤相关钙信号传导子1(Tumor-Associated Calcium Signal Transducer 1);MK-1;TACSTD1;人类上皮醣蛋白-2;TROP1;膜组分,染色体4,表面标记(35kD醣蛋白);腺癌相关抗原;EGP;细胞表面醣蛋白Trop-1;Ep-CAM;上皮醣蛋白314;GA733-2;主要肠胃肿瘤相关蛋白GA733-2;M1S2;EGP314;CD326抗原;KSA;上皮细胞表面抗原;D IAR5;上皮醣蛋白;HNPCC8;hEGP314;单株抗体AUA1鉴定的抗原;KS 1/4抗原;EGP-2;ACSTD1。Ids:HGNC:11529;Entrez Gene:4072;Ensembl:ENSG00000119888;OMIM:185535;UniProtKB:P16422。EPCAM is also known as Epithelial Cell Adhesion Molecule; EGP40; M4S1; ESA; MIC18; KS1/4; Tumor-Associated
HER2还称为V-Erb-B2禽红血球胚细胞白血病病毒致癌基因同源物2;ERBB2;CD340;NGL;HER-2;HER-2/neu2;NEU2;TKR1;神经/神经胶质母细胞瘤衍生的致癌基因同源物;C-Erb B2/Neu蛋白;转移性淋巴结基因19蛋白;赫斯达汀(herstatin);原致癌基因C-ErbB-2;神经母细胞瘤/神经胶质母细胞瘤衍生的致癌基因同源物;原致癌基因Neu;受体酪胺酸蛋白激酶ErbB-2;酪胺酸激酶型细胞表面受体HER2;V-Erb-B2红血球胚细胞白血病病毒致癌基因同源物2,神经/神经胶质母细胞瘤衍生的致癌基因同源物;MLN 19;MLN19;p185erbB2;CD340抗原;EC 2.7.10.1;EC 2.7.10;V-Erb-B2禽红血球胚细胞白血病病毒致癌基因同源物2(神经/神经胶质母细胞瘤衍生的致癌基因同源物)。Ids:HER2 also known as V-Erb-B2 avian erythroblast leukemia
HGNC:3430;Entrez Gene:2064;Ensembl:ENSG00000141736;OMIM:164870;UniProtKB:P04626。HGNC: 3430; Entrez Gene: 2064; Ensembl: ENSG00000141736; OMIM: 164870; UniProtKB: P04626.
HM1.24还称为BST2;骨髓基质细胞抗原2;TETHERIN;BST-2;骨髓基质抗原2;HM1.24抗原;联系蛋白(Tetherin);CD317;CD317抗原;NPC-A-7。Ids:HGNC:1119;EntrezGene:684;Ensembl:ENSG00000130303;OMIM:600534;UniProtKB:Q10589。HM1.24 is also known as BST2; Bone Marrow
MCSP还称为精子粒线体相关富含半胱胺酸蛋白(Sperm Mitochondria-Associated Cysteine-Rich Protein)(SMCP);MCSP;MCS;粒线体囊硒蛋白(MitochondrialCapsule Selenoprotein);HSMCSGEN1;精子粒线体相关富含半胱胺酸蛋白。Ids:HGNC:6962;Entrez Gene:4184;Ensembl:ENSG00000163206;OMIM:601148;UniProtKB:P49901。MCSP is also known as Sperm Mitochondria-Associated Cysteine-Rich Protein (SMCP); MCSP; MCS; Mitochondrial Capsule Selenoprotein; HSMCSGEN1; Body-associated cysteine-rich protein. Ids: HGNC: 6962; Entrez Gene: 4184; Ensembl: ENSG00000163206; OMIM: 601148; UniProtKB: P49901.
PD-L1为第1型跨膜蛋白,其在特定事件例如怀孕、组织同种异体移植、自体免疫疾病及其他疾病状态如肝炎,的期间于抑制免疫反应中起作用。PD-L1与PD-1或B7.1(CD80)结合传递抑制信息其减少PD-1表现T细胞的增殖。一般认为PD-1能够透过细胞凋亡来控制外来抗原特异性T细胞的累积。多种癌细胞有表现PD-L1,且其的表现认为至少部分负责减缓了对抗癌细胞的免疫反应。PD-L1是蛋白质的B7家族的成员且以各种其他名称为人所熟知,例如CD274分子;CD274抗原;B7同源物1;PDCD1配体1;PDCD1LG1;PDCD1L1;B7H1;PDL1;计划性细胞死亡1配体1;计划性死亡配体1;B7-H1;及B7-H。CD274的外部Ids为HGNC:17635;Entrez Gene:29126;Ensembl:ENSG00000120217;OMIM:605402;UniProtKB:Q9NZQ7。PD-L1 is a
PSMA还称为叶酸水解酶(前列腺特异性膜抗原)1;FOLH1;NAALAD1;FOLH;mGCP;麸胺酸羧肽酶II;N-乙酰化-α-链接的酸性双肽酶I;PSM;NAALADase I;PSMA;EC 3.4.17.21;麸胺酸羧酶II;GCP2;细胞生长抑制基因27蛋白;NAALAdase;叶酰多(Folylpoly)-γ-麸胺酸羧肽酶;麸胺酸羧肽酶2;膜麸胺酸羧肽酶;N-乙酰化α-链接的酸性双肽酶1;喋酰多(Pteroylpoly)-γ-麸胺酸羧肽酶;前列腺特异性膜抗原变异体F;FGCP;叶酸水解酶1;GCPII;前列腺特异性膜抗原。Ids:HGNC:3788;Entrez Gene:2346;Ensembl:ENSG00000086205;OMIM:600934;UniProtKB:Q04609。PSMA also known as folate hydrolase (prostate-specific membrane antigen) 1; FOLH1; NAALAD1; FOLH; mGCP; glutamic acid carboxypeptidase II; N-acetylated-α-linked acid dipeptidase I; PSM; NAALADase I; PSMA; EC 3.4.17.21; Glutamate carboxylase II; GCP2; cytostatic gene 27 protein; NAALAdase; ; Membrane glutamic acid carboxypeptidase; N-acetylated α-linked
PSMA不要与蛋白酶体(前体(Prosome),巨蛋白因子(Macropain))亚单位,α型,1混淆,其的别名也称为PSMA1。PSMA is not to be confused with the proteasome (Prosome, Macropain) subunit, alpha type, 1, which is also known by its alias as PSMA1.
给登录序号主要是为了提供另外的标靶鉴别方法,所结合的蛋白的实际序列可能,譬如因为编码基因突变而变化,例如在一些癌症及类似癌症中发生的此类。抗原结合地址与抗原及其多种变异体结合,例如一些抗原阳性免疫或肿瘤细胞表现的此类。Accession numbers are given primarily to provide an additional method of target identification, the actual sequence of the protein bound may vary, eg, due to mutations in the encoding gene, such as occurs in some cancers and the like. Antigen-binding addresses bind to antigens and their various variants, such as those exhibited by some antigen-positive immune or tumor cells.
在本文提到一基因、蛋白的处,其优选是指该基因或蛋白的人类形式。在本文提到一基因、蛋白的处,其是指天然基因或蛋白以及该基因或蛋白的变异体形式,如同于肿瘤、癌症及类似癌症内能检测到的,优选如人类肿瘤、癌症及类似癌症内能检测到的。Where reference is made herein to a gene, protein, it preferably refers to the human form of the gene or protein. Where a gene or protein is referred to herein, it refers to the native gene or protein as well as variant forms of the gene or protein, as can be detected in tumors, cancers and the like, preferably human tumors, cancers and the like Detectable in cancer.
本发明的双特异性或多特异性抗体优选结合人类BCMA、CD19、CD20、CD30、CD33、CD38、CD44、CD123、CD138、CEA、CLEC12A、CS-1、EGFR、EGFRvIII、EPCAM、DLL3、LGR5、MSLN、FOLR1、FOLR3、HER2、HM1.24、MCSP、PD-L1、PSMA蛋白或其的一变异体。该抗原结合重/轻链组合优选结合该抗原的细胞外部分。如本发明的双特异性抗体优选结合人类CLEC12A或其的一变异体。如本发明的优选的双特异性抗体结合人类CD3及人类CLEC12A或其的一变异体。在优选具体例中,该多特异性抗体结合CD3、PD-L1及EGFR。Bispecific or multispecific antibodies of the invention preferably bind human BCMA, CD19, CD20, CD30, CD33, CD38, CD44, CD123, CD138, CEA, CLEC12A, CS-1, EGFR, EGFRvIII, EPCAM, DLL3, LGR5, MSLN, FOLR1, FOLR3, HER2, HM1.24, MCSP, PD-L1, PSMA protein or a variant thereof. The antigen binding heavy/light chain combination preferably binds to the extracellular portion of the antigen. Bispecific antibodies according to the invention preferably bind human CLEC12A or a variant thereof. Preferred bispecific antibodies according to the invention bind human CD3 and human CLEC12A or a variant thereof. In a preferred embodiment, the multispecific antibody binds to CD3, PD-L1 and EGFR.
HGNC代表HUGO基因命名委员会。缩写后的数字为登录序号,以该登录序号可以从HGNC数据库中检索该基因及该基因编码的蛋白的信息。Entrez Gene提供登录序号或基因ID,以其可以从NCBI(国家生物技术信息中心)数据库中检索该基因或该基因编码的蛋白的信息。Ensemble提供登录序号,以该登录序号可以从Ensemble数据库中获得该基因或该基因编码的蛋白的信息。Ensembl是EMBL-EB及威康信托桑格研究所(Wellcome Trust SangerInstitute)之间共同的计划以发展一软件系统其对所选的真核基因体产生自动注解且被维护。HGNC stands for HUGO Gene Nomenclature Committee. The number after the abbreviation is the accession number, and the information of the gene and the protein encoded by the gene can be retrieved from the HGNC database with the accession number. Entrez Gene provides accession number or gene ID, which can retrieve the information of the gene or the protein encoded by the gene from the NCBI (National Center for Biotechnology Information) database. Ensemble provides accession numbers with which information about the gene or the protein encoded by the gene can be obtained from the Ensemble database. Ensembl is a joint project between EMBL-EB and the Wellcome Trust Sanger Institute to develop a software system that automatically annotates and maintains selected eukaryotic genomes.
本发明提供一种会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括一CDR1、CDR2及CDR3The present invention provides an antigen-binding protein that binds to human CD3, preferably an antibody, comprising an antibody variable domain, the antibody variable domain comprising a heavy chain variable region and a light chain variable region, wherein the The heavy chain variable region includes a CDR1, CDR2 and CDR3
该CDR1、CDR2及CDR3包括下列氨基酸序列:The CDR1, CDR2 and CDR3 include the following amino acid sequences:
CDR1:SFGIS;CDR2:GFIPVLGTANYAQKFQG;CDR3:RGNWNPFDPCDR1: SFGIS; CDR2: GFIPVLGTANYAQKFQG; CDR3: RGNWNPFDP
或or
包括下列氨基酸序列:Includes the following amino acid sequences:
CDR1:SX1TFTIS;CDR1: SX 1 TFTIS;
CDR2:GIIPX2FGTITYAQKFQG;CDR2: GIIPX 2 FGTITYAQKFQG;
CDR3:RGNWNPFDP;CDR3: RGNWNPFDP;
其中in
X1=K或R;X2=L或I。X 1 =K or R; X 2 =L or I.
在优选的具体例中,X1=K;及X2=L。在另一优选的具体例中,X1=R;及X2=I。In a preferred embodiment, X 1 =K; and X 2 =L. In another preferred embodiment, X 1 =R; and X 2 =I.
本发明提供一种会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括一CDR1、CDR2及CDR3,该CDR1、CDR2及CDR3包括下列氨基酸序列:The present invention provides an antigen-binding protein that binds to human CD3, preferably an antibody, comprising an antibody variable domain, the antibody variable domain comprising a heavy chain variable region and a light chain variable region, wherein the The heavy chain variable region includes a CDR1, CDR2, and CDR3, and the CDR1, CDR2, and CDR3 include the following amino acid sequences:
CDR1:SKTLTIS;CDR2:GIIPIFGSITYAQKFQD;CDR3:RGNWNPFDP;或CDR1: SKTLTIS; CDR2: GIIPIFGSITYAQKFQD; CDR3: RGNWNPFDP; or
包括下列氨基酸序列:Includes the following amino acid sequences:
CDR1:GSGIS;CDR2:GFIPFFGSANYAQKFRD;CDR3:RGNWNPX13DP;CDR1: GSGIS; CDR2: GFIPFFGSANYAQKFRD; CDR3: RGNWNPX 13 DP;
其中in
X13=或L或F。X 13 =or L or F.
本发明进一步提供一种会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括一CDR1、CDR2及CDR3,该CDR1、CDR2及CDR3包括下列氨基酸序列:The present invention further provides an antigen-binding protein that binds human CD3, preferably an antibody, comprising an antibody variable domain, the antibody variable domain comprising a heavy chain variable region and a light chain variable region, wherein The heavy chain variable region includes a CDR1, CDR2 and CDR3, and the CDR1, CDR2 and CDR3 include the following amino acid sequences:
CDR1:RX3WIG;CDR2:IIYPGDSDTRYSPSFQG;CDR3:X4IRYFX5WSEDYHYYX6DV;CDR1: RX3WIG; CDR2: IIYPGDSDTRYSPSFQG; CDR3: X 4 IRYFX 5 WSEDYHYYX 6 DV;
其中in
X3=F或Y;X4=H或N;X5=D或V;及X6=L或M。 X3 =F or Y; X4 =H or N ; X5=D or V ; and X6=L or M.
在优选的具体例中,X3=F;X4=H;X5=D;及X6=L;或X3=Y;X4=N;X5=V;及X6=M。In preferred embodiments, X 3 =F; X 4 =H; X 5 =D; and X 6 =L; or X 3 =Y; X 4 =N; X 5 =V; and X 6 =M.
本发明进一步提供一种会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括一CDR1、CDR2及CDR3,该CDR1、CDR2及CDR3包括下列氨基酸序列:The present invention further provides an antigen-binding protein that binds human CD3, preferably an antibody, comprising an antibody variable domain, the antibody variable domain comprising a heavy chain variable region and a light chain variable region, wherein The heavy chain variable region includes a CDR1, CDR2 and CDR3, and the CDR1, CDR2 and CDR3 include the following amino acid sequences:
CDR1:SYALS;CDR2:GISGSGRTTWYADSVKG;CDR3:DGGYSYGPYWYFDL。CDR1: SYALS; CDR2: GISGSGRTTWYADSVKG; CDR3: DGGYSYGPYWYFDL.
本发明进一步提供一种会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括一CDR1、CDR2及CDR3,该CDR1、CDR2及CDR3包括下列氨基酸序列:The present invention further provides an antigen-binding protein that binds human CD3, preferably an antibody, comprising an antibody variable domain, the antibody variable domain comprising a heavy chain variable region and a light chain variable region, wherein The heavy chain variable region includes a CDR1, CDR2 and CDR3, and the CDR1, CDR2 and CDR3 include the following amino acid sequences:
CDR1:SYALS;CDR2:AISGSGRTTWYADSVKG;CDR3:DGGYTYGPYWYFDL。CDR1: SYALS; CDR2: AISGSGRTTWYADSVKG; CDR3: DGGYTYGPYWYFDL.
本发明进一步提供一种会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括一CDR1、CDR2及CDR3,该CDR1、CDR2及CDR3包括下列氨基酸序列:The present invention further provides an antigen-binding protein that binds human CD3, preferably an antibody, comprising an antibody variable domain, the antibody variable domain comprising a heavy chain variable region and a light chain variable region, wherein The heavy chain variable region includes a CDR1, CDR2 and CDR3, and the CDR1, CDR2 and CDR3 include the following amino acid sequences:
CDR1:DYTMH;CDR2:DISWSSGSIGYADSVKG;CDR3:DHRGYGDYEGGGFDY。CDR1: DYTMH; CDR2: DISWSSGSIGYADSVKG; CDR3: DHRGYGDYEGGGFDY.
本发明进一步提供一种会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括一CDR1、CDR2及CDR3,该CDR1、CDR2及CDR3包括下列氨基酸序列:The present invention further provides an antigen-binding protein that binds human CD3, preferably an antibody, comprising an antibody variable domain, the antibody variable domain comprising a heavy chain variable region and a light chain variable region, wherein The heavy chain variable region includes a CDR1, CDR2 and CDR3, and the CDR1, CDR2 and CDR3 include the following amino acid sequences:
CDR1:DYTMH;CDR1: DYTMH;
CDR2:DISWSX7GX8X9X10YADSVKG;CDR2: DISSWX 7 GX 8 X 9 X 10 YADSVKG;
CDR3:DHX11GYGDYEGGGFDX12;CDR3: DHX 11 GYGDYEGGGFDX 12 ;
其中in
X7=S或G;X 7 =S or G;
X8=S或T;X 8 =S or T;
X9=I或T;X 9 =I or T;
X10=G或Y;X 10 =G or Y;
X11=R或M;X 11 =R or M;
X12=H或Y,X 12 =H or Y,
优选X7、X8、X9及X10为S、S、I及G或是G、S、I及Y或是S、T、T和G,并且优选X11及X12为R及H,或是R及Y,或是M及Y,更优选X7、X8、X9、X10、X11和X12为S、S、I、G、R和H或是G、S、I、Y、R及Y或是S、T、T、G、M及Y,或是换句话讲,优选X7、X8、X9和X10为S、S、I及G,并且X11及X12为R和H;或X7、X8、X9和X10为G、S、I和Y和X11和X12为R和Y;或X7、X8、X9和X10为S、T、T和G,并且X11和X12为M及Y。Preferably X 7 , X 8 , X 9 and X 10 are S, S, I and G or G, S, I and Y or S, T, T and G, and preferably X 11 and X 12 are R and H , or R and Y, or M and Y, more preferably X 7 , X 8 , X 9 , X 10 , X 11 and X 12 are S, S, I, G, R and H or G, S, I, Y, R and Y are either S, T, T, G, M and Y, or in other words, preferably X 7 , X 8 , X 9 and X 10 are S, S, I and G, and X 11 and X 12 are R and H; or X 7 , X 8 , X 9 and X 10 are G, S, I and Y and X 11 and X 12 are R and Y; or X 7 , X 8 , X 9 and X 10 are S, T, T and G, and X 11 and X 12 are M and Y.
在优选的具体例中,该轻链可变区包括如图11A所描绘的IgVκ1-39*01基因节段,具有0-10个、优选0-5个氨基酸变异、插入、缺失、取代、加入或其的组合的氨基酸序列。图11A中描绘IgVK1-39*01的氨基酸序列。IgVκ1-39为免疫球蛋白可变κ1-39基因的缩写。该基因也被称为免疫球蛋白κ可变1-39;IGKV139;IGKV1-39。该基因的外部Ids为HGNC:5740;Entrez Gene:28930;Ensembl:ENSG00000242371。图11A中提供IgVκ1-39优选的氨基酸序列。此行举出V区域的序列。V区域能与五个J区域的一者组合。图11B和11D描述IgVκ 1-39序列组合以一个J区域的二种优选的序列。联结的序列表示为IGKV1-39/jk1及IGKV1-39/jk5;替换名称为IgVκ1-39*01/IGJκ1*01或IgVκ1-39*01/IGJκ5*01(根据imgt.org的IMGT数据库万维网来命名)。In a preferred embodiment, the light chain variable region comprises the IgVκ1-39*01 gene segment as depicted in Figure 11A, with 0-10, preferably 0-5 amino acid variations, insertions, deletions, substitutions, additions or amino acid sequences of combinations thereof. The amino acid sequence of IgV K 1-39*01 is depicted in Figure 11A. IgVκ1-39 is the abbreviation for Immunoglobulin Variable κ1-39 Gene. This gene is also known as immunoglobulin kappa variable 1-39; IGKV139; IGKV1-39. The external Ids of this gene are HGNC: 5740; Entrez Gene: 28930; Ensembl: ENSG00000242371. Preferred amino acid sequences of IgVκ1-39 are provided in Figure 11A. This row lists the sequence of V regions. The V zone can be combined with one of the five J zones. Figures 11B and 11D depict two preferred sequences for the IgVκ 1-39 sequence combined with a J region. The linked sequences are denoted as IGKV1-39/jk1 and IGKV1-39/jk5; alternative names are IgVκ1-39*01/IGJκ1*01 or IgVκ1-39*01/IGJκ5*01 (named according to the IMGT database World Wide Web at imgt.org ).
包括轻链可变区的IgVκ1-39*01为生殖系列序列是偏好的。包括轻链可变区的IGJκ1*01或/IGJκ5*01为生殖系列序列是更偏好的。于优选的具体例中,IGKV1-39/jk1或IGKV1-39/jk5轻链可变区是生殖系列序列。IgVκ1-39*01 including the light chain variable region is preferred for the germline sequence. IGJκ1*01 or /IGJκ5*01 including the light chain variable region are preferred as germline sequences. In a preferred embodiment, the IGKV1-39/jk1 or IGKV1-39/jk5 light chain variable region is a germline sequence.
在优选的具体例中该轻链可变区包括生殖系列IgVκ1-39*01。于优选的具体例中该轻链可变区包括κ轻链IgVκ1-39*01/IGJκ1*01或IgVκ1-39*01/IGJκ5*01。于优选的具体例中IgVκ1-39*01/IGJκ1*01。该轻链可变区优选包括生殖系列κ轻链IgVκ1-39*01/IGJκ1*01或生殖系列κ轻链IgVκ1-39*01/IGJκ5*01,优选为生殖系列IgVκ1-39*01/IGJκ1*01。In a preferred embodiment, the light chain variable region includes germline series IgVκ1-39*01. In a preferred embodiment, the light chain variable region includes a κ light chain IgVκ1-39*01/IGJκ1*01 or IgVκ1-39*01/IGJκ5*01. In a preferred embodiment, IgVκ1-39*01/IGJκ1*01. The light chain variable region preferably comprises a germline κ light chain IgVκ1-39*01/IGJκ1*01 or a germline κ light chain IgVκ1-39*01/IGJκ5*01, preferably a germline κ light chain IgVκ1-39*01/IGJκ1* 01.
生产带有一轻链的抗体的成熟B细胞通常会生产一种轻链,其相关于生殖系列序列,即该生物的非淋巴细胞的正常序列,已经历一或更多个突变。负责这些突变的过程通常称为称为体细胞(超)突变。产生的轻链称为亲和力成熟的轻链。这些轻链,当衍生自生殖系列IgVκ1-39*01序列时,为IgVκ1-39*01衍生的轻链。于此说明书中,用语“IgVκ1-39*01”会包括IgVκ1-39*01衍生的轻链,通过体细胞超突变导入的突变也可以于实验室中人工导入。于实验室中也可以导入其他的突变或变异至轻链而不影响轻链在种类方面,不一定是数量方面的性质。一种轻链设如果其包括如图11A、图11或图11所描绘的序列具有0-10个、优选0-5个氨基酸变异、插入、缺失、取代、加入或其的组合,则其无论如何是一种IgVκ1-39*01轻链。在优选的具体例中,该IgVκ1-39*01轻链为一种轻链其包括如图11A、图11B或图11C所描绘的序列其具有0-9、0-8、0-7、0-6、0-5、0-4个氨基酸变异、插入、缺失、取代、加入或其的组合。在优选的具体例中,该IgVκ1-39*01轻链为一种轻链其包括如图11A、图11B或图11C所描绘的序列其具有0-5个,优选为0-4个,更优选为0-3个氨基酸变异、插入、缺失、取代、加入或其的组合。在优选的具体例中,该IgVκ1-39*01轻链为一种轻链其包括如图11A、图11B或图11C所描绘的序列其具有0-2个,更优选为0-1个,最佳为0个氨基酸变异、插入、缺失、取代、加入或其的组合。在优选的具体例中,该IgVκ1-39*01轻链为一种轻链其包括如图11A或图11B所描绘的序列其具有所提及的氨基酸变异、插入、缺失、取代、加入或其的组合。在优选的具体例中,该轻链包括图11B的序列。Mature B cells that produce antibodies with a light chain typically produce a light chain that has undergone one or more mutations relative to the germline sequence, the normal sequence of the organism's non-lymphocytes. The process responsible for these mutations is often referred to as somatic (hyper)mutation. The resulting light chain is referred to as an affinity matured light chain. These light chains, when derived from the germline IgVκ1-39*01 sequence, are IgVκ1-39*01 derived light chains. In this specification, the term "IgVκ1-39*01" will include IgVκ1-39*01 derived light chains, mutations introduced by somatic hypermutation can also be introduced artificially in the laboratory. Other mutations or variants can also be introduced into the light chain in the laboratory without affecting the type, not necessarily quantitative, nature of the light chain. A light chain design has 0-10, preferably 0-5 amino acid variations, insertions, deletions, substitutions, additions or a combination thereof, regardless of whether it comprises a sequence as depicted in Figure 11A, Figure 11 or Figure 11. How is an IgVκ1-39*01 light chain. In a preferred embodiment, the IgVκ1-39*01 light chain is a light chain comprising a sequence as depicted in Figure 11A, Figure 11B or Figure 11C having 0-9, 0-8, 0-7, 0 - 6, 0-5, 0-4 amino acid variations, insertions, deletions, substitutions, additions or combinations thereof. In a preferred embodiment, the IgVκ1-39*01 light chain is a light chain comprising a sequence as depicted in Figure 11A, Figure 11B or Figure 11C, which has 0-5, preferably 0-4, more Preferably 0-3 amino acid variations, insertions, deletions, substitutions, additions or combinations thereof. In a preferred embodiment, the IgVκ1-39*01 light chain is a light chain comprising a sequence as depicted in Figure 11A, Figure 11B or Figure 11C, which has 0-2, more preferably 0-1, Most preferred are zero amino acid variations, insertions, deletions, substitutions, additions, or combinations thereof. In a preferred embodiment, the IgVκ1-39*01 light chain is a light chain comprising a sequence as depicted in Figure 11A or Figure 11B with the mentioned amino acid variations, insertions, deletions, substitutions, additions or the like The combination. In a preferred embodiment, the light chain includes the sequence of Figure 11B.
该轻链优选包括一共同的轻链可变区。该共同的轻链可变区优选包括一IgVκ1-39轻链可变区。该轻链可变区优选为生殖系列IgVκ1-39*01可变区。该轻链可变区优选包括κ轻链IgVκ1-39*01/IGJκ1*01或IgVκ1-39*01/IGJκ5*01。该轻链可变区优选包括生殖系列κ轻链IgVκ1-39*01/IGJκ1*01或IgVκ1-39*01/IGJκ5*01。该轻链可变区优选包括氨基酸序列DIQMT QSPSS LSASV GDRVT ITCRA SQSIS SYLNW YQQKP GKAPK LLIYA ASSLQ SGVPS RFSGSGSGTD FTLTI SSLQP EDFAT YYCQQ SYSTP PTFGQ GTKVE IK或DIQMT QSPSS LSASV GDRVTITCRA SQSIS SYLNW YQQKP GKAPK LLIYA ASSLQ SGVPS RFSGS GSGTD FTLTI SSLQP EDFATYYCQQ SYSTP PITFG QGTRL EIK,其具有0-5个氨基酸变异、插入、缺失、取代、加入或其的组合。The light chains preferably include a common light chain variable region. The common light chain variable region preferably includes an IgVκ1-39 light chain variable region. The light chain variable region is preferably a germline IgVκ1-39*01 variable region. The light chain variable region preferably comprises a kappa light chain IgVκ1-39*01/IGJκ1*01 or IgVκ1-39*01/IGJκ5*01. The light chain variable region preferably comprises a germline kappa light chain IgVκ1-39*01/IGJκ1*01 or IgVκ1-39*01/IGJκ5*01. The light chain variable region preferably comprises the amino acid sequence DIQMT QSPSS LSASV GDRVT ITCRA SQSIS SYLNW YQQKP GKAPK LLIYA ASSLQ SGVPS RFSGSGSGTD FTLTI SSLQP EDFAT YYCQQ SYSTP PTFGQ GTKVE IK or DIQMT QSPSS LSASV GDRVTITCRA SQSIS SYLNW YQQKP GKAPK LLIYA ASSLQ SGVPS RFSGS GSGTPEDTD FTLITQQ SSLQ SGVPS RFSGS GSGTPED FAT FGYY QGTRL EIK with 0-5 amino acid variations, insertions, deletions, substitutions, additions or combinations thereof.
该轻链可变区优选包括CDR1、CDR2及CDR3区域,其包括氨基酸序列CDR1-QSISSY,CDR2-AAS,CDR3-QQSYSTP,即IGKV1-39的CDRs(根据IMGT)。该氨基酸变异、插入、缺失、取代、加入或其的组合优选不在该轻链可变区的CDR3区域内,优选不在该轻链可变区的CDR1或CDR2区域内。于优选的具体例中,该轻链可变区就所示的序列而言不包括缺失、加入或变异。在此具体例中,该轻链可变区就所示的氨基酸序列而言可具有0-5个氨基酸取代。氨基酸取代优选为保留型氨基酸取代。本发明的抗体的轻链的CDR1、CDR2及CDR3优选分别包括氨基酸序列CDR1-QSISSY、CDR2-AAS、CDR3-QQSYSTP,即IGKV1-39的CDRs(根据IMGT)。The light chain variable region preferably comprises the CDR1, CDR2 and CDR3 regions comprising the amino acid sequences CDR1-QSISSY, CDR2-AAS, CDR3-QQSYSTP, the CDRs of IGKV1-39 (according to IMGT). The amino acid variation, insertion, deletion, substitution, addition or combination thereof is preferably not within the CDR3 region of the light chain variable region, preferably not within the CDR1 or CDR2 region of the light chain variable region. In preferred embodiments, the light chain variable region does not include deletions, additions or variations with respect to the sequence shown. In this embodiment, the light chain variable region may have 0-5 amino acid substitutions for the amino acid sequence shown. Amino acid substitutions are preferably reserved amino acid substitutions. The CDR1, CDR2 and CDR3 of the light chain of the antibody of the invention preferably comprise the amino acid sequences CDR1-QSISSY, CDR2-AAS, CDR3-QQSYSTP, respectively, ie the CDRs of IGKV1-39 (according to IMGT).
该抗原结合蛋白优选为一种抗体,优选为一种双特异性或多特异性抗体。该抗体优选包括一种共同的轻链,该共同的轻链包括一如本文所定义的共同的轻可变区及一如本文所定义的轻链恒定区域。The antigen binding protein is preferably an antibody, preferably a bispecific or multispecific antibody. The antibody preferably comprises a common light chain comprising a common light variable region as defined herein and a light chain constant region as defined herein.
本发明如前所述的抗体优选为一种双特异性抗体。“双特异性抗体”如本文所述为一种抗体其中该抗体的一域与第一抗原结合而该抗体的第二域与第二抗原结合,其中该第一与第二抗原不是完全相同的。术语“双特异性抗体”还包括抗体其中一重链可变区/轻链可变区(VH/VL)组合结合一抗原上的第一表位及第二VH/VL组合结合第二表位。该术语进一步包括抗体其中VH能特异性识别第一抗原,且与免疫球蛋白可变区的VH配对的VL能特异性识别第二抗原。生成的VH/VL对会结合抗原1或抗原2。这些所谓的“二合一抗体”于例如WO2008/027236、WO2010/108127及Schaefer等人(Cancer Cell 20,472-486,2011年十月)内有描述。如本发明的双特异性抗体不限于任何特定双特异性格式或其生产方法。The antibody of the invention as previously described is preferably a bispecific antibody. A "bispecific antibody" as described herein is an antibody wherein one domain of the antibody binds a first antigen and a second domain of the antibody binds a second antigen, wherein the first and second antigens are not identical . The term "bispecific antibody" also includes antibodies in which a heavy chain variable region/light chain variable region (VH/VL) combination binds a first epitope on an antigen and a second VH/VL combination binds a second epitope. The term further includes antibodies in which the VH can specifically recognize a first antigen and the VL paired with the VH of the immunoglobulin variable region can specifically recognize a second antigen. The resulting VH/VL pair will bind either
该双特异性抗体优选具有会结合CD3的一重链可变区/轻链可变区(VH/VL)组合以及会结合CD3上的抗原以外的抗原的第二VH/VL组合。在优选的具体例中,抗原为肿瘤抗原。在优选的具体例中,该第一VH/VL组合内的VL与该第二VH/VL组合内的VL是相似的。在一更优选的具体例中,该第一及第二VH/VL组合内的VLs是完全相同的。在优选具体例中,该双特异性抗体是一种全长抗体其具有会结合CD3的一重/轻(H/L)链组合以及会结合另一抗原,优选为肿瘤抗原的一H/L链组合。在优选的具体例中,该第一H/L链组合的轻链与该第二H/L链组合的轻链是相似的。在一更优选具体例中,该第一及第二H/L链组合的轻链是完全相同的,即一相似或完全相同的人类轻链是所谓的“共同的轻链”,其为一种能与不同重链组合以形成有功能性抗原结合域的抗体的轻链。在优选的具体例中,于该第一H/L链组合内的轻链包括一轻链可变区,其与该第二H/L链组合内的轻链可变区是相似的。在一更优选的具体例中,该第一及第二H/L链组合内的轻链可变区是完全相同的,即一相似或完全相同的人类轻链可变区是所谓的“共同的轻链可变区”,其为一种能与不同重链可变区组合以形成有功能性抗原结合域的抗体的轻链可变区。该包括一共同的轻链可变区的轻链优选为一共同的轻链。The bispecific antibody preferably has a heavy chain variable region/light chain variable region (VH/VL) combination that binds CD3 and a second VH/VL combination that binds an antigen other than an antigen on CD3. In a preferred embodiment, the antigen is a tumor antigen. In a preferred embodiment, the VL in the first VH/VL combination is similar to the VL in the second VH/VL combination. In a more preferred embodiment, the VLs in the first and second VH/VL combinations are identical. In a preferred embodiment, the bispecific antibody is a full-length antibody having a heavy/light (H/L) chain combination that binds CD3 and an H/L chain that binds another antigen, preferably a tumor antigen combination. In a preferred embodiment, the light chain of the first H/L chain combination is similar to the light chain of the second H/L chain combination. In a more preferred embodiment, the light chains of the first and second H/L chain combinations are completely identical, that is, a similar or identical human light chain is a so-called "common light chain", which is a A light chain that can be combined with different heavy chains to form antibodies with functional antigen binding domains. In a preferred embodiment, the light chain in the first H/L chain combination includes a light chain variable region that is similar to the light chain variable region in the second H/L chain combination. In a more preferred embodiment, the light chain variable regions in the first and second H/L chain combinations are completely identical, that is, a similar or identical human light chain variable region is a so-called "common" "Light chain variable region", which is a light chain variable region that can be combined with different heavy chain variable regions to form an antibody with a functional antigen binding domain. The light chains comprising a common light chain variable region are preferably a common light chain.
双特异性抗体内的共同的轻链优选为如上文所示的IgVκ1-39轻链。The common light chain within the bispecific antibody is preferably an IgVκ1-39 light chain as indicated above.
本发明还提供任择的双特异性抗体格式,例如Spiess,C.等人乙文(Alternativemolecular formats and therapeutic applications for bispecificantibodies.Mol.Immunol.(2015)http:dx.doi.org/10.1016/j.molimm.2015.01.003)中所述的此类。双特异性抗体格式-不是具有二个H/L组合的古典抗体-具有本发明的包括一重链可变区及一轻链可变区的至少一可变结构域。此可变结构域可链接一单链Fv片段、单一体型(monobody)、VH及Fab片段以提供第二结合活性。The invention also provides optional bispecific antibody formats, such as Spiess, C. et al. B (Alternativemolecular formats and therapeutic applications for bispecificantibodies. Mol. Immunol. (2015) http:dx.doi.org/10.1016/j. such as described in molimm.2015.01.003). Bispecific antibody formats - not classical antibodies with two H/L combinations - have at least one variable domain of the invention comprising a heavy chain variable region and a light chain variable region. This variable domain can link a single chain Fv fragment, monobody, VH and Fab fragment to provide a second binding activity.
于本发明的双特异性抗体方面,于CD3结合的H/L链组合内的轻链优选与结合CD3以外的抗原,优选为肿瘤抗原的H/V链组合内的轻链是相似的。在一更优选具体例中,二种H/L链组合内的轻链是完全相同的,即该人类轻链是所谓的“共同的轻链”,其为一种能与不同重链组合以形成有功能性抗原结合域的抗体的轻链。优选地,该共同的轻链具有生殖系列序列。优选的生殖系列序列是人类库(human repertoire)常用且具有良好的热力学稳定性、产量及溶解度的轻链可变区。优选的生殖系列序列是IgVκ1-39,优选为重排的生殖系列人类κ轻链IgVκ1-39*01/IGJκ1*01或其片段或功能等效物(即相同的IgVκ1-39基因节段但IGJκ基因节段不同)(根据imgt.org的IMGT数据库万维网来命名)。In the aspect of the bispecific antibody of the present invention, the light chain in the CD3-binding H/L chain combination is preferably similar to the light chain in the H/V chain combination that binds an antigen other than CD3, preferably a tumor antigen. In a more preferred embodiment, the light chains in the two H/L chain combinations are completely identical, that is, the human light chain is a so-called "common light chain", which is a kind of light chain that can be combined with different heavy chains to Form the light chain of an antibody with a functional antigen-binding domain. Preferably, the common light chain has germline sequences. Preferred germline sequences are light chain variable regions that are commonly used in human repertoires and have good thermodynamic stability, yield and solubility. A preferred germline sequence is IgVκ1-39, preferably a rearranged germline human kappa light chain IgVκ1-39*01/IGJκ1*01 or a fragment or functional equivalent thereof (ie the same IgVκ1-39 gene segment but IGJκ different gene segments) (named according to the IMGT database World Wide Web at imgt.org).
当使用于本文时术语“异常细胞”包括肿瘤细胞,更具体地为血液来源的肿瘤细胞,还包括白血病前期细胞例如造成骨髓发育不良症候群(MDS)的细胞及白血病细胞如急性骨髓性白血病(AML)肿瘤细胞或慢性骨髓性白血病(CML)细胞。The term "abnormal cell" as used herein includes tumor cells, more particularly blood-derived tumor cells, but also preleukemic cells such as those responsible for myelodysplastic syndrome (MDS) and leukemic cells such as acute myeloid leukemia (AML). ) tumor cells or chronic myeloid leukemia (CML) cells.
当使用于本文中术语“免疫效应子细胞”或“效应子细胞”是指哺乳动物免疫系统内的细胞天然库内的细胞,其能被活化以影响标靶细胞的生存力。免疫效应子细胞包括淋巴谱系细胞例如自然杀手(NK)细胞、T细胞包括胞毒型T细胞,或B细胞,且包括骨髓谱系细胞,例如单核球或巨噬细胞、树突细胞及嗜中性颗粒球。因此,该效应子细胞优选为NK细胞、T细胞、B细胞、单核球、巨噬细胞、树突细胞或嗜中性颗粒球。募集效应子细胞至异常细胞意指使免疫效应子细胞接近异常标靶细胞以使得效应子细胞能直接杀灭异常细胞或间接发起杀灭异常细胞。The term "immune effector cell" or "effector cell" as used herein refers to cells within the natural repertoire of cells within the mammalian immune system that can be activated to affect the viability of target cells. Immune effector cells include lymphoid lineage cells such as natural killer (NK) cells, T cells including cytotoxic T cells, or B cells, and include myeloid lineage cells such as monocytes or macrophages, dendritic cells and neutrophils Sexual pellets. Therefore, the effector cells are preferably NK cells, T cells, B cells, monocytes, macrophages, dendritic cells or neutrophils. Recruiting effector cells to abnormal cells means bringing immune effector cells into close proximity to abnormal target cells so that the effector cells can directly kill abnormal cells or indirectly initiate killing of abnormal cells.
当使用于本文中,术语“个体”及“患者”可互换使用且是指哺乳动物例如人类、小鼠、大鼠、仓鼠、天竺鼠、兔、猫、狗、猴、牛、马、猪等(例如有癌症的患者,例如人类患者)。As used herein, the terms "individual" and "patient" are used interchangeably and refer to mammals such as humans, mice, rats, hamsters, guinea pigs, rabbits, cats, dogs, monkeys, cows, horses, pigs, etc. (eg patients with cancer, eg human patients).
当使用于本文中,术语“治疗(treat)”、“治疗(treating)”及“治疗(treatment)”指在个体上进行任何类型的介入或过程或向该个体投予活性剂或活性剂组合,且目的在于逆转、减轻、改良、抑制或减慢或预防与疾病相关联的症状、并发症、病况或生物化学指标的进展、发展、严重性或复发。As used herein, the terms "treat," "treating," and "treatment" refer to performing any type of intervention or procedure on an individual or administering an active agent or combination of active agents to that individual , and for the purpose of reversing, alleviating, ameliorating, inhibiting or slowing down or preventing the progression, progression, severity or recurrence of a symptom, complication, condition or biochemical marker associated with the disease.
当使用于本文中,“有效的治疗”或“正面治疗反应”是指产生有益效应的治疗,例如减轻一疾病或障碍,如癌症的至少一症状。有益效应能以比基线有所改善的方式呈现,包括比根据该方法的疗法起始前所进行的测量或观察有改善。例如,有益效应能以减慢、稳定、中止或逆转癌症于处于任何临床阶段的个体内的进展的方式呈现,如由疾病的临床或诊断症状或癌症标识减少或消除来证明。有效的治疗可,例如减小肿瘤尺寸、减少循环的肿瘤细胞存在、降低或预防肿瘤转移、减慢或阻止肿瘤生长和/或预防或延迟肿瘤复发或再发。As used herein, "effective treatment" or "positive treatment response" refers to treatment that produces a beneficial effect, eg, alleviation of at least one symptom of a disease or disorder, such as cancer. A beneficial effect can be presented as an improvement from baseline, including improvement from measurements or observations made prior to initiation of therapy according to the method. For example, a beneficial effect can be manifested in a manner that slows, stabilizes, halts or reverses the progression of cancer in an individual at any clinical stage, as evidenced by a reduction or elimination of clinical or diagnostic symptoms of the disease or markers of cancer. Effective treatment can, for example, reduce tumor size, reduce the presence of circulating tumor cells, reduce or prevent tumor metastasis, slow or arrest tumor growth, and/or prevent or delay tumor recurrence or recurrence.
术语“治疗量”指提供所欲的生物、治疗和/或预防结果的一制剂或制剂组合的量。该结果可为减少、改良、缓和、衰减、延迟和/或减轻疾病的一个或多个征象、症状或原因,或生物系统任何其他所欲的改变。在一些具体例中,治疗量为足以延迟肿瘤发展的量。在一些具体例中,治疗量为足以预防或延迟肿瘤复发的量。治疗量能以一或多次投予被投予。药物或组合物的治疗量可以:(i)降低癌细胞的数目;(ii)缩小肿瘤尺寸;(iii)在一定程度上抑制、减缓、减慢且可中止癌细胞浸润至周边器官内;(iv)抑制肿瘤转移;(v)抑制肿瘤生长;(vi)预防或延迟肿瘤发生和/或复发;和/或(vii)在一定程度上缓解癌症相关联的一个或多个症状。在一实例中,“治疗量”是CLEC12A/CD3双特异性抗体的量,其减少癌症(例如癌细胞数目减少)或减慢癌症,例如急性骨髓性白血病、骨髓发育不良症候群或慢性骨髓性白血病的进展。The term "therapeutic amount" refers to the amount of a formulation or combination of formulations that provides the desired biological, therapeutic and/or prophylactic result. The result may be a reduction, amelioration, mitigation, attenuation, delay and/or alleviation of one or more signs, symptoms or causes of a disease, or any other desired change in a biological system. In some embodiments, the therapeutic amount is an amount sufficient to delay tumor development. In some embodiments, the therapeutic amount is an amount sufficient to prevent or delay tumor recurrence. The therapeutic amount can be administered in one or more administrations. The therapeutic amount of the drug or composition can: (i) reduce the number of cancer cells; (ii) reduce the size of the tumor; (iii) to a certain extent inhibit, slow, slow and stop the infiltration of cancer cells into surrounding organs; ( iv) inhibiting tumor metastasis; (v) inhibiting tumor growth; (vi) preventing or delaying tumor development and/or recurrence; and/or (vii) alleviating to some extent one or more symptoms associated with cancer. In one example, a "therapeutic amount" is the amount of a CLEC12A/CD3 bispecific antibody that reduces cancer (eg, a reduction in the number of cancer cells) or slows cancer, eg, acute myeloid leukemia, myelodysplastic syndrome, or chronic myelogenous leukemia Progress.
本发明还提供一种会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括氨基酸序列The present invention also provides an antigen-binding protein that binds to human CD3, preferably an antibody, comprising an antibody variable domain, the antibody variable domain comprising a heavy chain variable region and a light chain variable region, wherein The heavy chain variable region includes the amino acid sequence
于此类CDRs以外的一个或多个位置处具有0-10个、优选0-5个氨基酸变异、插入、缺失、取代、加入或其组合。There are 0-10, preferably 0-5 amino acid variations, insertions, deletions, substitutions, additions or combinations thereof at one or more positions other than such CDRs.
进一步提供一种会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括氨基酸序列Further provide an antigen-binding protein that can bind to human CD3, preferably an antibody, comprising an antibody variable domain, the antibody variable domain comprising a heavy chain variable region and a light chain variable region, wherein the heavy chain variable region is provided. chain variable region including amino acid sequence
于此类CDRs以外的一个或多个位置处具有0-10个、优选0-5个氨基酸变异、插入、缺失、取代、加入或其组合。There are 0-10, preferably 0-5 amino acid variations, insertions, deletions, substitutions, additions or combinations thereof at one or more positions other than such CDRs.
还提供一种会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括氨基酸序列Also provided is an antigen binding protein that binds human CD3, preferably an antibody, comprising an antibody variable domain comprising a heavy chain variable region and a light chain variable region, wherein the heavy chain variable region is chain variable region including amino acid sequence
于此类CDRs以外的一个或多个位置处具有0-10个、优选0-5个氨基酸变异、插入、缺失、取代、加入或其组合。There are 0-10, preferably 0-5 amino acid variations, insertions, deletions, substitutions, additions or combinations thereof at one or more positions other than such CDRs.
本发明进一步提供一种会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括氨基酸序列The present invention further provides an antigen-binding protein that binds human CD3, preferably an antibody, comprising an antibody variable domain, the antibody variable domain comprising a heavy chain variable region and a light chain variable region, wherein The heavy chain variable region includes the amino acid sequence
于此类CDRs以外的一个或多个位置处具有0-10个、优选0-5个氨基酸变异、插入、缺失、取代、加入或其组合。There are 0-10, preferably 0-5 amino acid variations, insertions, deletions, substitutions, additions or combinations thereof at one or more positions other than such CDRs.
该氨基酸变异、插入、缺失、取代、加入或其的组合优选不在重链可变区的CDR3区域内,优选不在重链可变区的CDR1和/或CDR2区域内。于优选的具体例中,该重链可变区就所示的序列而言不包括缺失、加入或变异、插入。在一具体例中,该重链可变区就所示的氨基酸序列而言可具有0-10个,优选0-5个氨基酸取代。在优选的具体例中,该重链可变区就所示的氨基酸序列而言,于此类CDRs以外的位置处包括0-9、0-8、0-7、0-6、0-5、0-4,优选为0-3,优选为0-2,优选为0-1且优选为0个氨基酸变异、插入、缺失、取代、加入,或其的组合。设如果排列的序列没有超过10个位置的差异,优选为没有超过5个位置的差异,则一个插入、加入、缺失或取代的组合为所主张的组合。位在排列的序列中的一者内的一个空位会算作是在另一个序列中跳过(skipped)的数量一样多的氨基酸。一个氨基酸取代,如果有的话,优选是一种保留型氨基酸取代。The amino acid variation, insertion, deletion, substitution, addition or combination thereof is preferably not within the CDR3 region of the heavy chain variable region, preferably not within the CDR1 and/or CDR2 region of the heavy chain variable region. In a preferred embodiment, the heavy chain variable region does not include deletions, additions or variations, insertions with respect to the sequence shown. In a specific example, the heavy chain variable region may have 0-10, preferably 0-5, amino acid substitutions with respect to the amino acid sequence shown. In preferred embodiments, the heavy chain variable region includes 0-9, 0-8, 0-7, 0-6, 0-5 at positions other than such CDRs with respect to the indicated amino acid sequence , 0-4, preferably 0-3, preferably 0-2, preferably 0-1 and preferably 0 amino acid variations, insertions, deletions, substitutions, additions, or combinations thereof. Provided that the aligned sequences do not differ by more than 10 positions, preferably not by more than 5 positions, then a combination of insertions, additions, deletions or substitutions is the claimed combination. A gap in one of the aligned sequences will count as as many amino acids as skipped in the other sequence. An amino acid substitution, if any, is preferably a reserved amino acid substitution.
本发明进一步提供一种会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括如图13所描绘的MF8057;MF8058或MF8078的氨基酸序列。The present invention further provides an antigen-binding protein that binds human CD3, preferably an antibody, comprising an antibody variable domain, the antibody variable domain comprising a heavy chain variable region and a light chain variable region, wherein The heavy chain variable region comprises the amino acid sequence of MF8057; MF8058 or MF8078 as depicted in Figure 13.
本发明进一步提供一种会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括如图13所描绘的MF8397的氨基酸序列。The present invention further provides an antigen-binding protein that binds human CD3, preferably an antibody, comprising an antibody variable domain, the antibody variable domain comprising a heavy chain variable region and a light chain variable region, wherein The heavy chain variable region includes the amino acid sequence of MF8397 as depicted in FIG. 13 .
本发明进一步提供一种会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括如图13所描绘的MF8508的氨基酸序列。The present invention further provides an antigen-binding protein that binds human CD3, preferably an antibody, comprising an antibody variable domain, the antibody variable domain comprising a heavy chain variable region and a light chain variable region, wherein The heavy chain variable region includes the amino acid sequence of MF8508 as depicted in FIG. 13 .
本发明进一步提供一种会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括如图13所描绘的MF9249或MF9267的氨基酸序列。The present invention further provides an antigen-binding protein that binds human CD3, preferably an antibody, comprising an antibody variable domain, the antibody variable domain comprising a heavy chain variable region and a light chain variable region, wherein The heavy chain variable region includes the amino acid sequence of MF9249 or MF9267 as depicted in Figure 13.
该轻链优选包括如本文其他地方所定义的CDR1、CDR2及CDR3。其优选包括一共同的轻链可变区且优选为如本文其他地方所定义的一共同的轻链。该双特异性抗体优选进一步包括一种会结合另一抗原,优选为肿瘤抗原的重链及轻链组合。会结合另一抗原的该重链及轻链组合的轻链优选为如本文其他地方所定义的一共同的轻链。会结合另一抗原的该重链及轻链组合的重链优选包括一重链可变区,该重链可变区包括一氨基酸序列:MF8233(EGFR)The light chain preferably comprises CDRl, CDR2 and CDR3 as defined elsewhere herein. It preferably comprises a common light chain variable region and is preferably a common light chain as defined elsewhere herein. The bispecific antibody preferably further comprises a heavy chain and light chain combination that will bind to another antigen, preferably a tumor antigen. The light chain of the combination of heavy and light chains that will bind another antigen is preferably a common light chain as defined elsewhere herein. The heavy chain of the heavy chain and light chain combination that will bind another antigen preferably includes a heavy chain variable region comprising an amino acid sequence: MF8233 (EGFR)
QVQLVQSGAEVKKPGASVKVSCKASGYTFTSYGISWVRQAPGQGLEWMGWISAYNANTNYAQKLQGRVTMTTDTSTSTAYMELRSLRSDDTAVYYCAKDRHWHWWLDAFDYWGQGTLVTVSS,于此类CDRs以外的一个或多个位置处具有0-10个、优选0-5个氨基酸变异、插入、缺失、取代、加入或其的组合;或MF4327(CLEC12A)QVQLVQSGAEVKKPGASVKVSCKASGYTFTSYGISWVRQAPGQGLEWMGWISAYNANTNYAQKLQGRVTMTTDTSTSTAYMELRSLRSDDTAVYYCAKDRHWHWWLDAFDYWGQGTLVTVSS with 0-10, preferably 0-5 amino acid variations, insertions, deletions, substitutions, additions or combinations thereof at one or more positions other than such CDRs; or MF4327 (CLEC12A)
于此类CDRs以外的一个或多个位置处具有0-10个、优选0-5个氨基酸变异、插入、缺失、取代、加入或其的组合。There are 0-10, preferably 0-5 amino acid variations, insertions, deletions, substitutions, additions or combinations thereof at one or more positions other than such CDRs.
如本文所定义的会结合CLEC12A的可变结构域,其具有一重链可变区及一共同的轻链区域,于WO2014/051433及WO2017/010874等等的内有描述,为本文的此目的特别提及其且其并入本文以作为参考资料。会结合人类EGFR或CLEC12A的该重/轻链组合的重链可变区,就所示的氨基酸序列而言能具有0-10个、优选0-5个氨基酸变异、插入、缺失、取代、加入或其的组合。在优选的具体例中,该重链可变区就所示的氨基酸序列而言,包括0-9、0-8、0-7、0-6、0-5、0-4,优选为0-3,优选为0-2,优选为0-1且优选为0个氨基酸变异、插入、缺失、取代、加入,或其的组合。设如果排列的序列没有超过5个位置的差异,则一个插入、缺失、加入或取代的组合为所主张的组合。位在排列的序列中的一者内的一个空位会算作是在另一个序列中跳过的数量一样多的氨基酸。Variable domains that bind CLEC12A as defined herein, having a heavy chain variable region and a common light chain region, are described in WO2014/051433 and WO2017/010874, among others, especially for this purpose herein It is mentioned and incorporated herein by reference. The heavy chain variable region of this heavy/light chain combination that will bind human EGFR or CLEC12A can have 0-10, preferably 0-5 amino acid variations, insertions, deletions, substitutions, additions, for the amino acid sequence shown or a combination thereof. In a preferred embodiment, the heavy chain variable region includes 0-9, 0-8, 0-7, 0-6, 0-5, 0-4 in terms of the shown amino acid sequence, preferably 0 -3, preferably 0-2, preferably 0-1 and preferably 0 amino acid variations, insertions, deletions, substitutions, additions, or a combination thereof. A combination of insertions, deletions, additions or substitutions is the claimed combination, provided that the aligned sequences do not differ by more than 5 positions. A gap in one of the aligned sequences will count as as many amino acids as skipped in the other sequence.
一氨基酸变异、插入、缺失、取代、加入或其的组合优选不进行/存在于重及轻链的结合界面(interface)中。An amino acid variation, insertion, deletion, substitution, addition or combination thereof is preferably not made/present in the heavy and light chain binding interface.
设如果H/L链交互作用的界面中的氨基酸有改变,优选的是另一链中对应的氨基酸也改变以适应该改变。插入或加入氨基酸最好不需要插入或加入脯胺酸。Provided that if the amino acid in the interface of the H/L chain interaction is changed, it is preferred that the corresponding amino acid in the other chain is also changed to accommodate the change. The insertion or addition of amino acids preferably does not require insertion or addition of proline.
加入氨基酸原则上可认为与插入是一样的。加入氨基酸至多肽链的一末端有时不认为是插入而是严格的加入(strict addition)(延长)。就本发明而言,加入一链内或加入至一末端二者一般均认为是插入。The addition of amino acids can in principle be considered the same as insertion. The addition of amino acids to one end of a polypeptide chain is sometimes not considered an insertion but rather a strict addition (extension). For the purposes of the present invention, both addition into a strand or addition to a terminus are generally considered insertions.
该氨基酸变异、插入、缺失、取代、加入或其的组合优选不是在该重链可变区的CDR3区域内,优选不是在该重链可变区的CDR1或CDR2区域内。于优选的具体例中,该重链可变区就所示的序列而言不包括缺失、加入或变异。在此具体例中,该重链可变区就所示的氨基酸序列而言可具有0-5个氨基酸取代。氨基酸取代优选为保留型氨基酸取代。本发明的CD3结合VH的CDR1、CDR2及CDR3优选包括图13所描绘的CD3结合VH的CDR1、CD2及CDR3的组合,优选MF8057;MF8058;MF8078;MF8397;MF8508;MF9249或MF9267的一者的VH。The amino acid variation, insertion, deletion, substitution, addition or combination thereof is preferably not within the CDR3 region of the heavy chain variable region, preferably not within the CDR1 or CDR2 region of the heavy chain variable region. In preferred embodiments, the heavy chain variable region does not include deletions, additions or variations with respect to the sequence shown. In this particular example, the heavy chain variable region may have 0-5 amino acid substitutions for the amino acid sequence shown. Amino acid substitutions are preferably reserved amino acid substitutions. The CD3 binding VH CDR1, CDR2 and CDR3 of the invention preferably comprise the combination of CD3 binding VH CDR1, CD2 and CDR3 depicted in Figure 13, preferably the VH of one of MF8057; MF8058; MF8078; MF8397; MF8508; MF9249 or MF9267 .
本发明的抗体,包括双特异性或多特异性抗体,的恒定区域优选地为人类恒定区域。该恒定区域可含有与天然存在的人类抗体的恒定区域的一个或多个,优选不超过10个,优选不超过5个氨基酸差异。本文生产的抗体的各种可变区衍生自人类抗体可变结构域库。因此这些可变结构域为人类的。独特的CDR区域可衍生自人类、为合成的或衍生自另一种生物。本发明的抗体或双特异性抗体优选为一种人类或人化抗体。合适的重链恒定区域非限制性地举例说明于图12中。The constant regions of the antibodies of the invention, including bispecific or multispecific antibodies, are preferably human constant regions. The constant region may contain one or more, preferably no more than 10, preferably no more than 5 amino acid differences from the constant region of a naturally occurring human antibody. The various variable domains of the antibodies produced herein are derived from a library of human antibody variable domains. Therefore these variable domains are human. Unique CDR regions can be derived from humans, synthetic, or derived from another organism. The antibody or bispecific antibody of the invention is preferably a human or humanized antibody. Suitable heavy chain constant regions are exemplified in FIG. 12 without limitation.
本技艺存在多种生产抗体的方法。抗体通常由表现编码该抗体的核酸的细胞来生产。合适抗体生产的细胞为融合瘤细胞、中国仓鼠卵巢(CHO)细胞、NS0细胞或PER-C6细胞。在一个特别佳的具体例中,该细胞为CHO细胞。Various methods of producing antibodies exist in the art. Antibodies are typically produced by cells expressing nucleic acid encoding the antibody. Suitable antibody producing cells are fusion tumor cells, Chinese Hamster Ovary (CHO) cells, NSO cells or PER-C6 cells. In a particularly preferred embodiment, the cells are CHO cells.
各种机构及公司已经开发出用于大规模生产抗体的细胞株,譬如供临床使用。这些细胞株的非限制性实例为CHO细胞、NS0细胞或PER.C6细胞。这些细胞还用于其他的目的例如生产蛋白质。开发用于工业规模生产蛋白质及抗体的细胞株于此进一步称为工业细胞株。在优选的具体例中,本发明提供一种生产本发明的抗体的工业细胞株。Various institutions and companies have developed cell lines for large-scale production of antibodies, such as for clinical use. Non-limiting examples of these cell lines are CHO cells, NSO cells or PER.C6 cells. These cells are also used for other purposes such as protein production. Cell lines developed for industrial scale production of proteins and antibodies are further referred to herein as industrial cell lines. In a preferred embodiment, the present invention provides an industrial cell line that produces the antibody of the present invention.
在一具体例中本发明提供一种细胞,其包括如本发明的抗体和/或如本发明的核酸。该细胞优选为动物细胞,更优选为哺乳动物细胞,更优选为灵长类动物细胞,最佳为人类细胞。出于本发明的目的,一种合适的细胞为能包括且优选生产如本发明的抗体和/或如本发明的核酸的任何细胞。In one embodiment the invention provides a cell comprising an antibody as the invention and/or a nucleic acid as the invention. The cells are preferably animal cells, more preferably mammalian cells, more preferably primate cells, and most preferably human cells. For the purposes of the present invention, a suitable cell is any cell capable of comprising, and preferably producing, an antibody as of the invention and/or a nucleic acid as of the invention.
本发明进一步提供一种包括如本发明的抗体的细胞。优选地该细胞(典型为一种活体外、单离或重组细胞)生产该抗体。于优选的具体例中,该细胞为融合瘤细胞、中国仓鼠卵巢(CHO)细胞、NS0细胞或PER-C6细胞。在一特别佳的具体例中该细胞为CHO细胞。进一步提供一种细胞培养物其包括如本发明的细胞。各种机构及公司已经开发出用于大规模生产抗体的细胞株,譬如临床使用。这些细胞株的非限制性实例为CHO细胞、NS0细胞或PER.C6细胞。这些细胞还用于其他的目的例如生产蛋白质。开发用于工业规模生产蛋白质及抗体的细胞株于此进一步称为工业细胞株。因而在优选的具体例中,本发明提供一种开发用于大规模生产抗体的细胞株的用途,其供用于生产本发明的抗体。本发明进一步提供一种用于生产一抗体的细胞,该抗体包括一种编码如所主张的抗体的VH、VL和/或重及轻链的核酸分子。该核酸分子优选编码图13所示的VH、一编码如由数字4327所示或由数字8233所示的VH的核酸分子或其的组合。The present invention further provides a cell comprising the antibody of the present invention. Preferably the cell (typically an in vitro, isolated or recombinant cell) produces the antibody. In a preferred embodiment, the cells are fusion tumor cells, Chinese hamster ovary (CHO) cells, NSO cells or PER-C6 cells. In a particularly preferred embodiment the cells are CHO cells. Further provided is a cell culture comprising the cells of the present invention. Various institutions and companies have developed cell lines for large-scale production of antibodies, such as for clinical use. Non-limiting examples of these cell lines are CHO cells, NSO cells or PER.C6 cells. These cells are also used for other purposes such as protein production. Cell lines developed for industrial scale production of proteins and antibodies are further referred to herein as industrial cell lines. Thus, in a preferred embodiment, the present invention provides the use of developing a cell line for large-scale production of antibodies for producing the antibodies of the present invention. The present invention further provides a cell for producing an antibody comprising a nucleic acid molecule encoding the VH, VL and/or heavy and light chains of an antibody as claimed. The nucleic acid molecule preferably encodes the VH shown in Figure 13, a nucleic acid molecule encoding VH as shown by numeral 4327 or shown by numeral 8233, or a combination thereof.
本发明进一步提供一种用于生产一抗体的方法,其包括培养本发明的细胞且从该培养物收获该抗体。该细胞优选于无血清培养基内培养。优选地使该细胞适合供悬浮生长。进一步提供一种抗体其可由一种用于生产如本发明的抗体的方法而获得。该抗体优选由该培养物的培养基被纯化。该抗体比优选经亲和力纯化。The invention further provides a method for producing an antibody comprising culturing a cell of the invention and harvesting the antibody from the culture. The cells are preferably cultured in serum-free medium. The cells are preferably adapted for growth in suspension. Further provided is an antibody obtainable by a method for producing an antibody such as the present invention. The antibody is preferably purified from the culture medium. The antibody ratio is preferably affinity purified.
本发明的细胞举例来说为融合瘤细胞株、CHO细胞、293F细胞、NS0细胞或另一种已知其适合用于临床目的的抗体生产的细胞类型。在一特别佳的具体例中该细胞为人类细胞。优选为一种由腺病毒E1区域或其功能等效物转形的细胞。此一细胞株的优选实例为PER.C6细胞株或其效物。在一个特别佳具体例中该细胞为CHO细胞或其变异体。优选为一种利用麸酰胺酸合成酶(GS)载体系统用于表现抗体的变异体。Cells of the invention are, for example, fusion tumor cell lines, CHO cells, 293F cells, NSO cells or another cell type known to be suitable for antibody production for clinical purposes. In a particularly preferred embodiment the cells are human cells. Preferred is a cell transformed with the E1 region of adenovirus or a functional equivalent thereof. A preferred example of such a cell line is the PER.C6 cell line or an effector thereof. In a particularly preferred embodiment the cells are CHO cells or variants thereof. Preferred is a variant that utilizes the glutamic acid synthase (GS) vector system for expression of the antibody.
本发明进一步提供一种用于生产一抗体的方法,其包括培养本发明的细胞且从该培养物收获该抗体。该细胞优选于无血清培养基内培养。优选地使该细胞适合供悬浮生长。进一步提供一种抗体其可由一种用于生产如本发明的抗体的方法而获得。该抗体优选由该培养物的培养基被纯化。该抗体比优选系经亲和力纯化。The invention further provides a method for producing an antibody comprising culturing a cell of the invention and harvesting the antibody from the culture. The cells are preferably cultured in serum-free medium. The cells are preferably adapted for growth in suspension. Further provided is an antibody obtainable by a method for producing an antibody such as the present invention. The antibody is preferably purified from the culture medium. The antibody ratio is preferably affinity purified.
双特异性抗体典型还由表现编码该抗体的核酸的细胞来生产。在此情况下该细胞表现组成该双特异性抗体的不同的轻及重链。为此该细胞表现二种不同的重链及至少一轻链。因未经修饰的重链能彼此配对以形成二聚体,所以这些细胞典型除了生产双特异性抗体(异二聚体)的外还生产二种单特异性抗体(同型二聚体)。此原理也适用于未经修饰的重链,其包括一种具有一重链可变区的第一重链及一具有至少二种重链可变区的第二重链,而使得表现这些二种重链的细胞生产一种单特异性抗体(二种第一重链配对的同型二聚体)、四价(quadrovalent)抗体(二种第二重链配对的同型二聚体)及三特异性抗体(第一及第二重链的异二聚体)。当细胞表现二或多种轻链时,生产的抗体中可能的重/轻链组合数目增加。为了降低不同的抗体种类数目(不同的重及轻链组合)所以生产前述的“共同的轻链”是优选的。Bispecific antibodies are also typically produced by cells expressing the nucleic acid encoding the antibody. In this case the cells express different light and heavy chains that make up the bispecific antibody. To this end the cells express two different heavy chains and at least one light chain. Since unmodified heavy chains can pair with each other to form dimers, these cells typically produce two monospecific antibodies (homodimers) in addition to bispecific antibodies (heterodimers). This principle also applies to unmodified heavy chains, which include a first heavy chain with a heavy chain variable region and a second heavy chain with at least two heavy chain variable regions, such that these two heavy chain variable regions are expressed Heavy chain cells produce a monospecific antibody (two homodimers paired with a first heavy chain), a quadrovalent antibody (two homodimers paired with a second heavy chain), and trispecific antibodies Antibodies (heterodimers of first and second heavy chains). When cells express two or more light chains, the number of possible heavy/light chain combinations in the antibody produced increases. The production of the aforementioned "common light chain" is preferred in order to reduce the number of different antibody species (different heavy and light chain combinations).
一种表现一共同的轻链及等量的二种重链的抗体生产细胞典型生产50%双特异性抗体及25%的每种单特异性抗体(即有完全相同的重轻链组合)。任择地,在上文关于一具有一可变区的第一重链及一具有二种可变区的第二重链的实例中,该二种重链典型会生产50%的三特异性、25%的单特异性及25%的四特异性(quadrospecific)。An antibody-producing cell expressing a common light chain and equal amounts of both heavy chains typically produces 50% bispecific antibody and 25% each monospecific antibody (ie, having the exact same combination of heavy and light chains). Optionally, in the above example for a first heavy chain with one variable region and a second heavy chain with two variable regions, the two heavy chains would typically produce 50% trispecificity , 25% monospecific and 25% quadrospecific.
已经公开数种方法有助于生产双特异性抗体或反过来单特异性抗体也一样,其可进一步用于有助于多特异性抗体生产。于本发明中优选的是细胞有利于生产该双特异性抗体超过生产相应的单特异性抗体。这件事通常通过修饰重链的恒定区域以使得其有利异质二聚合(即与另一重/轻链组合的重链进行二聚合)超过同质二聚合来达成。于优选的具体例中本发明的双特异性抗体包括具有兼容的异质二聚合域的二个不同的免疫球蛋白重链。本技艺中已有描述各种兼容的异质二聚合域。兼容的异质二聚合域优选为兼容的免疫球蛋白重链CH3异质二聚合域。本技艺描述了各种可完成这些重链的异质二聚合的方法,包括使用“旋钮至孔洞(knob into hole)”的双特异性抗体。Several methods have been disclosed to facilitate the production of bispecific antibodies or conversely monospecific antibodies, which can further be used to facilitate the production of multispecific antibodies. It is preferred in the present invention that the cells favor the production of the bispecific antibody over the production of the corresponding monospecific antibody. This is usually accomplished by modifying the constant region of the heavy chain so that it favors heterodimerization (ie dimerization of the heavy chain with another heavy/light chain combination) over homodimerization. In a preferred embodiment the bispecific antibodies of the invention comprise two different immunoglobulin heavy chains with compatible heterodimeric domains. Various compatible heterodimeric domains have been described in the art. The compatible heterodimerization domain is preferably a compatible immunoglobulin heavy chain CH3 heterodimerization domain. The art describes various methods by which heterodimerization of these heavy chains can be accomplished, including the use of "knob into hole" bispecific antibodies.
于US13/866,747(现在颁布为US 9,248,181)、US14/081,848(现在颁布为US 9,358,286)及PCT/NL2013/050294(公开为WO2013/157954);并入本文以作为参考资料)中公开使用兼容的异质聚合二域供用于生产双特异性抗体的方法及构件。这些构件及方法也可以有利地使用于本发明中。具体地,基本上只生产双特异性全长IgG分子的优选的突变为第一CH3域的氨基酸取代L351K及T366K(根据EU编号)(“KK-变异体”重链)以及第二域的氨基酸取代L351D及L368E(“DE-变异体”重链),或反的还然。先前在我们的US 9,248,181和US9,358,286专利以及WO2013/157954 PCT申请案中证明了DE-变异体和KK-变异体优先地配对以形成异二聚体(所谓的“DEKK”双特异性分子)。由于完全相同的重链之间的CH3-CH3界面的带电残基之间强大的排斥作用,所以DE-变异体重链的同质二聚合(DEDE同型二聚体)或KK-变异体重链的同质二聚合(KKKK同型二聚体)几乎不会发生。在一具体例中,包括会结合CD3的可变结构域的重链/轻链组合,包括了该重链的KK变异体。在此具体例中包括会结合CD3以外的抗原的可变结构域的重链/轻链组合,包括了该重链的DE变异体。在优选的具体例中,该CD3以外的抗原系CLEC12A。在优选的具体例中,该会结合CLEC12A的可变结构域的VH如图13所描绘的MF4327。Compatible use is disclosed in
一些抗体于CH2/下部铰链区域予修饰,举例来说以降低Fc受体的交互作用或降低C1q的结合。在一些具体例中,本发明的抗体为具有突变型CH2和/或下部铰链域的IgG抗体以使得该双特异性IgG抗体与Fc-γ受体的交互作用减少。此一突变型CH2和/或下部铰链域优选包括位置235和/或236(根据EU编号)的氨基取代,优选为L235G和/或G236R取代。Some antibodies are pre-modified in the CH2/lower hinge region, for example, to reduce Fc receptor interaction or to reduce C1q binding. In some embodiments, the antibodies of the invention are IgG antibodies with mutant CH2 and/or lower hinge domains such that the interaction of the bispecific IgG antibodies with Fc-gamma receptors is reduced. Such a mutant CH2 and/or lower hinge domain preferably comprises amino substitutions at positions 235 and/or 236 (according to EU numbering), preferably L235G and/or G236R substitutions.
任择地,一些抗体被修饰例如以提升Fc受体的交互作用或提升C1q的结合。关于针对自体免疫适应症的具体例,此一修饰可能是优选的。Optionally, some antibodies are modified, eg, to enhance Fc receptor interaction or to enhance C1q binding. With regard to specific examples for autoimmune indications, this modification may be preferred.
本发明进一步提供一种治疗一个体的方法,其包括投予本发明的抗原结合蛋白,优选为抗体给有需要的该个体。本发明进一步提供一种本发明的抗原结合蛋白,优选为抗体供用于治疗有需要的一个体。该个体优选有要从身体上移开的细胞。此类细胞可以是导向自体免疫反应的异常免疫细胞或癌细胞等等。适合于此目的的可变结构域为具有一共同的轻链及MF9249及MF8397的VH的此类及其他。这些具有适当低的亲和力及低的细胞杀灭活性但于自体免疫反应条件下是有功能的。适合于此目的的可变结构域为具有一共同的轻链及MF9267、MF8057、MF8058、MF8078及MF8508的VH的此类及其他。这些可变结构域具有适当的亲和力及适当的细胞杀灭活性用于抗癌目的。The invention further provides a method of treating an individual comprising administering an antigen binding protein, preferably an antibody, of the invention to the individual in need thereof. The present invention further provides an antigen binding protein of the present invention, preferably an antibody, for use in the treatment of an individual in need. The individual preferably has cells to be removed from the body. Such cells can be abnormal immune cells or cancer cells that are directed toward an autoimmune response, among others. Suitable variable domains for this purpose are those with a common light chain and the VHs of MF9249 and MF8397 and others. These have moderately low affinity and low cell killing activity but are functional under autoimmune reaction conditions. Suitable variable domains for this purpose are these and others with a common light chain and VH of MF9267, MF8057, MF8058, MF8078 and MF8508. These variable domains have suitable affinity and suitable cell killing activity for anticancer purposes.
此外,一种在此列出的发明包括一种抗原结合蛋白或抗体,其具有有高细胞杀灭活性的高亲和力可变结构域,其能局部投予或局部表现,包括结合域MF8078。本发明进一步提供一种治疗一个体的方法,其包括投予本发明的抗原结合蛋白优选为抗体给有需要的该个体,其通过如熟悉此艺者已知的一种投予的局部化构件,包括例如,一溶瘤病毒,供用于黑色素瘤或于独立腔室例如脑内的其他癌症的局部治疗。In addition, an invention set forth herein includes an antigen binding protein or antibody having a high affinity variable domain with high cell killing activity that can be administered or expressed locally, including the binding domain MF8078. The present invention further provides a method of treating an individual comprising administering an antigen binding protein of the present invention, preferably an antibody, to the individual in need thereof via a localized means of administration as known to those skilled in the art , including, for example, an oncolytic virus for local treatment of melanoma or other cancers in isolated compartments such as the brain.
本发明进一步提供一种本发明的抗原结合蛋白,优选为抗体供用于治疗有需要的一个体,其优选具有中到高的亲和力及相当高的细胞毒性例如本文所述的此类。适合于此目的的可变结构域为具有一共同的轻链及MF8057、MF8058、MF9267、MF8508及MF8078的VH的此类及其他。The present invention further provides an antigen binding protein of the present invention, preferably an antibody, for use in the treatment of an individual in need thereof, which preferably has moderate to high affinity and relatively high cytotoxicity such as those described herein. Suitable variable domains for this purpose are those with a common light chain and the VHs of MF8057, MF8058, MF9267, MF8508 and MF8078 and others.
本发明进一步提供一种治疗一个体内的癌症或癌症的一风险的方法,其包括投予给有需要的该个体一会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括一CDR1、CDR2及CDR3,该CDR1、CDR2及CDR3包括下列氨基酸序列:The present invention further provides a method of treating cancer or a risk of cancer in an individual comprising administering to the individual in need thereof an antigen binding protein, preferably an antibody, which binds human CD3, comprising an antibody variable domain, the antibody variable domain includes a heavy chain variable region and a light chain variable region, wherein the heavy chain variable region includes a CDR1, CDR2 and CDR3, and the CDR1, CDR2 and CDR3 include the following amino acid sequences:
CDR1:SFGISCDR1: SFGIS
CDR2:GFIPVLGTANYAQKFQGCDR2: GFIPVLGTANYAQKFQG
CDR3:RGNWNPFDP;或CDR3: RGNWNPFDP; or
包括下列氨基酸序列:Includes the following amino acid sequences:
CDR1:SX1TFTIS;CDR1: SX 1 TFTIS;
CDR2:GIIPX2FGTITYAQKFQG;CDR2: GIIPX 2 FGTITYAQKFQG;
CDR3:RGNWNPFDP;CDR3: RGNWNPFDP;
其中in
X1=K或R;X 1 =K or R;
X2=L或I。X 2 =L or I.
在优选的具体例中,X1=K;及X2=L。在另一优选的具体例中,X1=R;及X2=I。In a preferred embodiment, X 1 =K; and X 2 =L. In another preferred embodiment, X 1 =R; and X 2 =I.
本发明进一步提供本发明的抗原结合蛋白,优选为抗体供用于治疗有需要的一个体,其优选具有中到高的亲和力及相当高的细胞毒性例如本文所述的此类。适合于此目的的可变结构域为具有一共同的轻链及MF8048、MF8056及MF8101的VH的此类及其他。The present invention further provides antigen binding proteins of the present invention, preferably antibodies, for use in the treatment of an individual in need thereof, which preferably have moderate to high affinity and relatively high cytotoxicity such as those described herein. Variable domains suitable for this purpose are those with a common light chain and the VH of MF8048, MF8056 and MF8101 and others.
本发明进一步提供一种治疗一个体内的癌症或癌症的一风险的方法,其包括投予给有需要的该个体一会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括一CDR1、CDR2及CDR3,其包括下列氨基酸序列:The present invention further provides a method of treating cancer or a risk of cancer in an individual comprising administering to the individual in need thereof an antigen binding protein, preferably an antibody, which binds human CD3, comprising an antibody variable domain, the antibody variable domain includes a heavy chain variable region and a light chain variable region, wherein the heavy chain variable region includes a CDR1, CDR2 and CDR3, which includes the following amino acid sequence:
CDR1:SKTLTIS;CDR1: SKTLTIS;
CDR2:GIIPIFGSITYAQKFQD;CDR2: GIIPIFGSITYAQKFQD;
CDR3:RGNWNPFDP;或CDR3: RGNWNPFDP; or
包括下列氨基酸序列:Includes the following amino acid sequences:
CDR1:GSGIS;CDR1: GSGIS;
CDR2:GFIPFFGSANYAQKFRD;CDR2: GFIPFFGSANYAQKFRD;
CDR3:RGNWNPX13DP;CDR3: RGNWNPX 13DP ;
其中in
X13=或L或F。X 13 =or L or F.
本发明进一步提供一种治疗一个体内的癌症或癌症的一风险的方法,其包括投予给有需要的该个体一会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括氨基酸序列The present invention further provides a method of treating cancer or a risk of cancer in an individual comprising administering to the individual in need thereof an antigen binding protein, preferably an antibody, which binds human CD3, comprising an antibody variable domain, the antibody variable domain includes a heavy chain variable region and a light chain variable region, wherein the heavy chain variable region includes an amino acid sequence
于此类CDRs以外的一个或多个位置处具有0-10个、优选0-5个氨基酸变异、插入、缺失、取代、加入或其组合。There are 0-10, preferably 0-5 amino acid variations, insertions, deletions, substitutions, additions or combinations thereof at one or more positions other than such CDRs.
本发明进一步提供一种治疗一个体内的癌症或癌症的一风险的方法,其包括投予给有需要的该个体一会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括一CDR1、CDR2及CDR3,该CDR1、CDR2及CDR3包括下列氨基酸序列:The present invention further provides a method of treating cancer or a risk of cancer in an individual comprising administering to the individual in need thereof an antigen binding protein, preferably an antibody, which binds human CD3, comprising an antibody variable domain, the antibody variable domain includes a heavy chain variable region and a light chain variable region, wherein the heavy chain variable region includes a CDR1, CDR2 and CDR3, and the CDR1, CDR2 and CDR3 include the following amino acid sequences:
CDR1:RX3WIG;CDR1: RX 3 WIG;
CDR2:IIYPGDSDTRYSPSFQG;CDR2: IIYPGDSDTRYSPSFQG;
CDR3:X4IRYFX5WSEDYHYYX6DV;CDR3: X 4 IRYFX 5 WSEDYHYYX 6 DV;
其中in
X3=F或Y;X 3 =F or Y;
X4=H或N;X 4 =H or N;
X5=D或V;X 5 =D or V;
X6=L或M。X 6 =L or M.
在一具体例中,X3=F;X4=H;X5=D;及X6=L。在另外的具体例中,X3=Y;X4=N;X5=V;及X6=M。In a specific example, X 3 =F; X 4 =H; X 5 =D; and X 6 =L. In another specific example, X 3 =Y; X 4 =N; X 5 =V; and X 6 =M.
本发明进一步提供一种治疗一个体内的癌症或癌症的一风险的方法,其包括投予给有需要的该个体一会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括氨基酸序列The present invention further provides a method of treating cancer or a risk of cancer in an individual comprising administering to the individual in need thereof an antigen binding protein, preferably an antibody, which binds human CD3, comprising an antibody variable domain, the antibody variable domain includes a heavy chain variable region and a light chain variable region, wherein the heavy chain variable region includes an amino acid sequence
于此类CDRs以外的一个或多个位置处具有0-10个、优选0-5个氨基酸变异、插入、缺失、取代、加入或其组合。There are 0-10, preferably 0-5 amino acid variations, insertions, deletions, substitutions, additions or combinations thereof at one or more positions other than such CDRs.
本发明进一步提供一种治疗一个体内的癌症或癌症的一风险的方法,其包括投予给有需要的该个体一会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括一CDR1、CDR2及CDR3,该CDR1、CDR2及CDR3包括下列氨基酸序列:The present invention further provides a method of treating cancer or a risk of cancer in an individual comprising administering to the individual in need thereof an antigen binding protein, preferably an antibody, which binds human CD3, comprising an antibody variable domain, the antibody variable domain includes a heavy chain variable region and a light chain variable region, wherein the heavy chain variable region includes a CDR1, CDR2 and CDR3, and the CDR1, CDR2 and CDR3 include the following amino acid sequences:
CDR1:SYALS;CDR1:SYALS;
CDR2:GISGSGRTTWYADSVKG;CDR2:GISGSGRTTWYADSVKG;
CDR3:DGGYSYGPYWYFDL。CDR3: DGGYSYGPYWYFDL.
本发明进一步提供一种治疗一个体内的癌症或癌症的一风险的方法,其包括投予给有需要的该个体一会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括一CDR1、CDR2及CDR3,该CDR1、CDR2及CDR3包括下列氨基酸序列:The present invention further provides a method of treating cancer or a risk of cancer in an individual comprising administering to the individual in need thereof an antigen binding protein, preferably an antibody, which binds human CD3, comprising an antibody variable domain, the antibody variable domain includes a heavy chain variable region and a light chain variable region, wherein the heavy chain variable region includes a CDR1, CDR2 and CDR3, and the CDR1, CDR2 and CDR3 include the following amino acid sequences:
CDR1:SYALS;CDR1:SYALS;
CDR2:AISGSGRTTWYADSVKG;CDR2: AISGSGRTTWYADSVKG;
CDR3:DGGYTYGPYWYFDL。CDR3: DGGYTYGPYWYFDL.
本发明进一步提供一种治疗一个体内的癌症或癌症的一风险的方法,其包括投予给有需要的该个体一会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括氨基酸序列The present invention further provides a method of treating cancer or a risk of cancer in an individual comprising administering to the individual in need thereof an antigen binding protein, preferably an antibody, which binds human CD3, comprising an antibody variable domain, the antibody variable domain includes a heavy chain variable region and a light chain variable region, wherein the heavy chain variable region includes an amino acid sequence
QVQLVESGGGLVQPGGSLRLSCATSGFTFISYALSWVRQAPGKGLEWVSAISGSGRTTWYADSVKGRFTISRDNSKNTLFLQMNSLRAEDTAVYYCARDGGYTYGPYWYFDLWGRGTLVTVSS;于此类CDRs以外的一个或多个位置处具有0-10个、优选0-5个氨基酸变异、插入、缺失、取代、加入或其的组合。QVQLVESGGGLVQPGSLRLSCATSGFTFISYALSWVRQAPGKGLEWVSAISGSGRTTWYADSVKGRFTISRDNSKNTLFLQMNSLRAEDTAVYYCARDGGYTYGPYWYFDLWGRGTLVTVSS; with 0-10, preferably 0-5 amino acid variations, insertions, deletions, substitutions, additions or combinations thereof at one or more positions other than such CDRs.
本发明进一步提供一种治疗一个体内的癌症或癌症的一风险的方法,其包括投予给有需要的该个体一会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括一CDR1、CDR2及CDR3,该CDR1、CDR2及CDR3包括下列氨基酸序列:The present invention further provides a method of treating cancer or a risk of cancer in an individual comprising administering to the individual in need thereof an antigen binding protein, preferably an antibody, which binds human CD3, comprising an antibody variable domain, the antibody variable domain includes a heavy chain variable region and a light chain variable region, wherein the heavy chain variable region includes a CDR1, CDR2 and CDR3, and the CDR1, CDR2 and CDR3 include the following amino acid sequences:
CDR1:DYTMH;CDR1: DYTMH;
CDR2:DISWSSGSIGYADSVKG;CDR2: DISWSSGSIGYADSVKG;
CDR3:DHRGYGDYEGGGFDY。CDR3: DHRGYGDYEGGGFDY.
本发明进一步提供一种治疗一个体内的癌症或癌症的一风险的方法,其包括投予给有需要的该个体一会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括一CDR1、CDR2及CDR3,该CDR1、CDR2及CDR3包括下列氨基酸序列:The present invention further provides a method of treating cancer or a risk of cancer in an individual comprising administering to the individual in need thereof an antigen binding protein, preferably an antibody, which binds human CD3, comprising an antibody variable domain, the antibody variable domain includes a heavy chain variable region and a light chain variable region, wherein the heavy chain variable region includes a CDR1, CDR2 and CDR3, and the CDR1, CDR2 and CDR3 include the following amino acid sequences:
CDR1:DYTMH;CDR1: DYTMH;
CDR2:DISWSX7GX8X9X10YADSVKG;CDR2: DISSWX 7 GX 8 X 9 X 10 YADSVKG;
CDR3:DHX11GYGDYEGGGFDX12;CDR3: DHX 11 GYGDYEGGGFDX 12 ;
其中in
X7=S或G;X 7 =S or G;
X8=S或T;X 8 =S or T;
X9=I或T;X 9 =I or T;
X10=G或Y;X 10 =G or Y;
X11=R或M;X 11 =R or M;
X12=H或Y,X 12 =H or Y,
优选X7、X8、X9及X10为S、S、I及G或是G、S、I及Y或是S、T、T和G,并且优选X11及X12为R及H,或是R及Y,或是M及Y,更优选X7、X8、X9、X10、X11和X12为S、S、I、G、R和H或是G、S、I、Y、R及Y或是S、T、T、G、M及Y,或是换句话讲,优选X7、X8、X9和X10为S、S、I及G,并且X11及X12为R和H;或X7、X8、X9和X10为G、S、I和Y和X11和X12为R和Y;或X7、X8、X9和X10为S、T、T和G,并且X11和X12为M及Y。Preferably X 7 , X 8 , X 9 and X 10 are S, S, I and G or G, S, I and Y or S, T, T and G, and preferably X 11 and X 12 are R and H , or R and Y, or M and Y, more preferably X 7 , X 8 , X 9 , X 10 , X 11 and X 12 are S, S, I, G, R and H or G, S, I, Y, R and Y are either S, T, T, G, M and Y, or in other words, preferably X 7 , X 8 , X 9 and X 10 are S, S, I and G, and X 11 and X 12 are R and H; or X 7 , X 8 , X 9 and X 10 are G, S, I and Y and X 11 and X 12 are R and Y; or X 7 , X 8 , X 9 and X 10 are S, T, T and G, and X 11 and X 12 are M and Y.
本发明进一步提供一种治疗一个体内的癌症或癌症的一风险的方法,其包括投予给有需要的该个体一会结合人类CD3的抗原结合蛋白,优选为一抗体,其包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括氨基酸序列The present invention further provides a method of treating cancer or a risk of cancer in an individual comprising administering to the individual in need thereof an antigen binding protein, preferably an antibody, which binds human CD3, comprising an antibody variable domain, the antibody variable domain includes a heavy chain variable region and a light chain variable region, wherein the heavy chain variable region includes an amino acid sequence
于此类CDRs以外的一个或多个位置处具有0-10个、优选0-5个氨基酸变异、插入、缺失、取代、加入或其组合。There are 0-10, preferably 0-5 amino acid variations, insertions, deletions, substitutions, additions or combinations thereof at one or more positions other than such CDRs.
如上所述于治疗中的抗原结合蛋白优选为一抗体,优选包括一种会结合肿瘤抗原的重链-轻链(H/L)组合。The antigen binding protein in therapy as described above is preferably an antibody, preferably comprising a heavy chain-light chain (H/L) combination that binds a tumor antigen.
该抗体优选为一种人类或人化抗体。该抗体优选包括具有兼容的异质二聚合域的二个不同的免疫球蛋白重链。该兼容的异质二聚合域优选为兼容的免疫球蛋白重链CH3异质二聚合域。该双特异性抗体针对肿瘤免疫学应用优选为具有突变型CH2和/或下部铰链域的IgG抗体以使得该双特异性或多特异性IgG抗体与Fc-γ受体的交互作用减少。该突变型CH2和/或下部铰链域优选包括于位置235和/或236(根据EU编号)的氨基取代,优选为L235G和/或G236R取代。任择地,针对自体免疫应用,该双特异性或多特异性与Fc-γ受体的交互作用通过修饰CH2和/或CH3域来提升。例如,通过工程处理(通过导入氨基酸取代)有较大的选择性结合活化的受体的Fc区域,能创造抗癌Mab或CD3靶向结合臂所欲的具有较大的媒介细胞毒性活性的能力的抗体供用于治疗自体免疫相关疾病。例如,一种报道的用于提升抗体的ADCC的技术是无岩藻醣基化(afucosylation)。(参见例如Junttila,T.T.,K.Parsons,等人(2010)。“Superior In vivo Efficacy of Afucosylated Trastuzumab in theTreatment of HER2-Amplified Breast Cancer.”Cancer Research 70(11):4481-4489)。因而进一步提供如本发明的双特异性抗体,其为无岩藻醣基化的。任择地,或另外地,据报道有多样其他的策略可用来达到ADCC提升,例如包括醣化工程处理(glycoengineering)(Kyowa Hakko/Biowa,GlycArt(Roche)and Eureka Therapeutics)及突变诱发(Xencorand Macrogenics),全部都是寻求要改良Fc与低亲和力活化的FcγRIIIa结合,和/或要降低与低亲和力抑制FcγRIIb结合。The antibody is preferably a human or humanized antibody. The antibody preferably comprises two different immunoglobulin heavy chains with compatible heterodimeric domains. The compatible heterodimerization domain is preferably a compatible immunoglobulin heavy chain CH3 heterodimerization domain. The bispecific antibody is preferably an IgG antibody with mutant CH2 and/or lower hinge domains for tumor immunology applications such that the interaction of the bispecific or multispecific IgG antibody with Fc-gamma receptors is reduced. The mutant CH2 and/or lower hinge domain preferably comprises amino substitutions at positions 235 and/or 236 (according to EU numbering), preferably L235G and/or G236R substitutions. Optionally, for autoimmune applications, the interaction of the bispecific or multispecific with Fc-gamma receptors is enhanced by modifying the CH2 and/or CH3 domains. For example, engineering (by introducing amino acid substitutions) the Fc region of a greater selective binding to an activated receptor can create the desired ability of an anticancer Mab or CD3 targeting binding arm to have greater mediator cytotoxic activity Antibodies for the treatment of autoimmune related diseases. For example, one reported technique for enhancing ADCC of antibodies is afucosylation. (See eg, Junttila, T.T., K. Parsons, et al. (2010). "Superior In vivo Efficacy of Afucosylated Trastuzumab in the Treatment of HER2-Amplified Breast Cancer." Cancer Research 70(11):4481-4489). There is thus further provided a bispecific antibody according to the invention which is afucosylated. Alternatively, or additionally, various other strategies have been reported to be used to achieve ADCC enhancement, including, for example, glycoengineering (Kyowa Hakko/Biowa, GlycArt (Roche) and Eureka Therapeutics) and mutagenesis (Xencor and Macrogenics). , all seek to improve Fc binding to low affinity activated FcγRIIIa, and/or to reduce binding to low affinity inhibitory FcγRIIb.
该抗体优选包括一共同的轻链。The antibodies preferably include a common light chain.
本发明进一步提供一种双特异性抗原结合蛋白,优选为一双特异性抗体,其包括一会结合肿瘤抗原的可变结构域及一会结合人类CD3的可变结构域,其中该可变结构域各包括一不同的重链可变区及一共同的轻链可变区,以及其中该会结合人类CD3的可变结构域的重链可变区包括一CDR1、CDR2及CDR3,该CDR1、CDR2及CDR3包括下列氨基酸序列:The present invention further provides a bispecific antigen binding protein, preferably a bispecific antibody, comprising a variable domain that binds to a tumor antigen and a variable domain that binds human CD3, wherein the variable domain Each includes a distinct heavy chain variable region and a common light chain variable region, and wherein the heavy chain variable region that binds the variable domains of human CD3 includes a CDR1, CDR2, and CDR3, the CDR1, CDR2 and CDR3 includes the following amino acid sequence:
CDR1:SFGISCDR1: SFGIS
CDR2:GFIPVLGTANYAQKFQGCDR2: GFIPVLGTANYAQKFQG
CDR3:RGNWNPFDP;或CDR3: RGNWNPFDP; or
包括下列氨基酸序列:Includes the following amino acid sequences:
CDR1:SX1TFTIS;CDR1: SX 1 TFTIS;
CDR2:GIIPX2FGTITYAQKFQG;CDR2: GIIPX 2 FGTITYAQKFQG;
CDR3:RGNWNPFDP;CDR3: RGNWNPFDP;
其中in
X1=K或R;X 1 =K or R;
X2=L或I。X 2 =L or I.
在优选的具体例中,X1=K;及X2=L。在另一优选的具体例中,X1=R;及X2=I。In a preferred embodiment, X 1 =K; and X 2 =L. In another preferred embodiment, X 1 =R; and X 2 =I.
本发明进一步提供一种双特异性抗原结合蛋白,优选为一双特异性抗体,其包括一会结合肿瘤抗原的可变结构域及一会结合人类CD3的可变结构域,其中该可变结构域各包括一不同的重链可变区及一共同的轻链可变区,以及其中该会结合人类CD3的可变结构域的重链可变区包括一CDR1、CDR2及CDR3,该CDR1、CDR2及CDR3包括下列氨基酸序列:The present invention further provides a bispecific antigen binding protein, preferably a bispecific antibody, comprising a variable domain that binds to a tumor antigen and a variable domain that binds human CD3, wherein the variable domain Each includes a distinct heavy chain variable region and a common light chain variable region, and wherein the heavy chain variable region that binds the variable domains of human CD3 includes a CDR1, CDR2, and CDR3, the CDR1, CDR2 and CDR3 includes the following amino acid sequence:
CDR1:SKTLTIS;CDR1: SKTLTIS;
CDR2:GIIPIFGSITYAQKFQD;CDR2: GIIPIFGSITYAQKFQD;
CDR3:RGNWNPFDP;或CDR3: RGNWNPFDP; or
包括下列氨基酸序列:Includes the following amino acid sequences:
CDR1:GSGIS;CDR1: GSGIS;
CDR2:GFIPFFGSANYAQKFRD;CDR2: GFIPFFGSANYAQKFRD;
CDR3:RGNWNPX13DP;CDR3: RGNWNPX 13DP ;
其中in
X13=或L或F。X 13 =or L or F.
本发明进一步提供一种双特异性抗原结合蛋白,优选为一双特异性抗体,其包括一会结合肿瘤抗原的可变结构域及一会结合人类CD3的可变结构域,其中该可变结构域各包括一不同的重链可变区及一共同的轻链可变区,以及其中该会结合人类CD3的可变结构域的重链可变区包括氨基酸序列The present invention further provides a bispecific antigen binding protein, preferably a bispecific antibody, comprising a variable domain that binds to a tumor antigen and a variable domain that binds human CD3, wherein the variable domain Each includes a distinct heavy chain variable region and a common light chain variable region, and wherein the heavy chain variable region that binds the variable domain of human CD3 includes the amino acid sequence
于此类CDRs以外的一个或多个位置处具有0-10个、优选0-5个氨基酸变异、插入、缺失、取代、加入或其组合。There are 0-10, preferably 0-5 amino acid variations, insertions, deletions, substitutions, additions or combinations thereof at one or more positions other than such CDRs.
本发明进一步提供一种双特异性抗原结合蛋白,优选为一双特异性抗体,其包括一会结合肿瘤抗原的可变结构域及一会结合人类CD3的可变结构域,其中该可变结构域各包括一不同的重链可变区及一共同的轻链可变区,以及其中该会结合人类CD3的可变结构域的重链可变区包括一CDR1、CDR2及CDR3,该CDR1、CDR2及CDR3包括下列氨基酸序列:The present invention further provides a bispecific antigen binding protein, preferably a bispecific antibody, comprising a variable domain that binds to a tumor antigen and a variable domain that binds human CD3, wherein the variable domain Each includes a distinct heavy chain variable region and a common light chain variable region, and wherein the heavy chain variable region that binds the variable domains of human CD3 includes a CDR1, CDR2, and CDR3, the CDR1, CDR2 and CDR3 includes the following amino acid sequence:
CDR1:RX3WIG;CDR1: RX 3 WIG;
CDR2:IIYPGDSDTRYSPSFQG;CDR2: IIYPGDSDTRYSPSFQG;
CDR3:X4IRYFX5WSEDYHYYX6DV;CDR3: X 4 IRYFX 5 WSEDYHYYX 6 DV;
其中in
X3=F或Y;X 3 =F or Y;
X4=H或N;X 4 =H or N;
X5=D或V;X 5 =D or V;
X6=L或M。X 6 =L or M.
在一具体例中,X3=F;X4=H;X5=D;及X6=L。在另外的具体例中,X3=Y;X4=N;X5=V;及X6=M。In a specific example, X 3 =F; X 4 =H; X 5 =D; and X 6 =L. In another specific example, X 3 =Y; X 4 =N; X 5 =V; and X 6 =M.
本发明进一步提供一种双特异性抗原结合蛋白,优选为一双特异性抗体,其包括一会结合肿瘤抗原的可变结构域及一会结合人类CD3的可变结构域,其中该可变结构域各包括一不同的重链可变区及一共同的轻链可变区,以及其中该会结合人类CD3的可变结构域的重链可变区包括氨基酸序列The present invention further provides a bispecific antigen binding protein, preferably a bispecific antibody, comprising a variable domain that binds to a tumor antigen and a variable domain that binds human CD3, wherein the variable domain Each includes a distinct heavy chain variable region and a common light chain variable region, and wherein the heavy chain variable region that binds the variable domain of human CD3 includes the amino acid sequence
于此类CDRs以外的一个或多个位置处具有0-10个、优选0-5个氨基酸变异、插入、缺失、取代、加入或其组合。There are 0-10, preferably 0-5 amino acid variations, insertions, deletions, substitutions, additions or combinations thereof at one or more positions other than such CDRs.
本发明进一步提供一种双特异性抗原结合蛋白,优选为一双特异性抗体,其包括一会结合肿瘤抗原的可变结构域及一会结合人类CD3的可变结构域,其中该可变结构域各包括一不同的重链可变区及一共同的轻链可变区,以及其中该会结合人类CD3的可变结构域的重链可变区包括一CDR1、CDR2及CDR3,该CDR1、CDR2及CDR3包括下列氨基酸序列:The present invention further provides a bispecific antigen binding protein, preferably a bispecific antibody, comprising a variable domain that binds to a tumor antigen and a variable domain that binds human CD3, wherein the variable domain Each includes a distinct heavy chain variable region and a common light chain variable region, and wherein the heavy chain variable region that binds the variable domains of human CD3 includes a CDR1, CDR2, and CDR3, the CDR1, CDR2 and CDR3 includes the following amino acid sequence:
CDR1:SYALS;CDR1:SYALS;
CDR2:GISGSGRTFWYADSVKG;CDR2:GISGSGRTFWYADSVKG;
CDR3:DGGYSYGPYWYFDL。CDR3: DGGYSYGPYWYFDL.
本发明进一步提供一种双特异性抗原结合蛋白,优选为一双特异性抗体,其包括一会结合肿瘤抗原的可变结构域及一会结合人类CD3的可变结构域,该会结合人类CD3的可变结构域包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括一CDR1、CDR2及CDR3,该CDR1、CDR2及CDR3包括下列氨基酸序列:The present invention further provides a bispecific antigen-binding protein, preferably a bispecific antibody, comprising a variable domain that binds to a tumor antigen and a variable domain that binds human CD3, which binds to human CD3 The variable domain includes an antibody variable domain, the antibody variable domain includes a heavy chain variable region and a light chain variable region, wherein the heavy chain variable region includes a CDR1, CDR2 and CDR3, the CDR1, CDR2 and CDR3 include the following amino acid sequences:
CDR1:SYALS;CDR1:SYALS;
CDR2:AISGSGRTTWYADSVKG;CDR2: AISGSGRTTWYADSVKG;
CDR3:DGGYTYGPYWYFDL。CDR3: DGGYTYGPYWYFDL.
本发明进一步提供一种双特异性抗原结合蛋白,优选为一双特异性抗体,其包括一会结合肿瘤抗原的可变结构域及一会结合人类CD3的可变结构域,其中该可变结构域各包括一不同的重链可变区及一共同的轻链可变区,以及其中该会结合人类CD3的可变结构域的重链可变区包括氨基酸序列The present invention further provides a bispecific antigen binding protein, preferably a bispecific antibody, comprising a variable domain that binds to a tumor antigen and a variable domain that binds human CD3, wherein the variable domain Each includes a distinct heavy chain variable region and a common light chain variable region, and wherein the heavy chain variable region that binds the variable domain of human CD3 includes the amino acid sequence
于此类CDRs以外的一个或多个位置处具有0-10个、优选0-5个氨基酸变异、插入、缺失、取代、加入或其组合。There are 0-10, preferably 0-5 amino acid variations, insertions, deletions, substitutions, additions or combinations thereof at one or more positions other than such CDRs.
本发明进一步提供一种双特异性抗原结合蛋白,优选为一双特异性抗体,其包括一会结合肿瘤抗原的可变结构域及一会结合人类CD3的可变结构域,其中该可变结构域各包括一不同的重链可变区及一共同的轻链可变区,以及其中该会结合人类CD3的可变结构域的重链可变区包括一CDR1、CDR2及CDR3,该CDR1、CDR2及CDR3包括下列氨基酸序列:The present invention further provides a bispecific antigen binding protein, preferably a bispecific antibody, comprising a variable domain that binds to a tumor antigen and a variable domain that binds human CD3, wherein the variable domain Each includes a distinct heavy chain variable region and a common light chain variable region, and wherein the heavy chain variable region that binds the variable domains of human CD3 includes a CDR1, CDR2, and CDR3, the CDR1, CDR2 and CDR3 includes the following amino acid sequence:
CDR1:DYTMH;CDR1: DYTMH;
CDR2:DISWSSGSIGYADSVKG;CDR2: DISWSSGSIGYADSVKG;
CDR3:DHRGYGDYEGGGFDY。CDR3: DHRGYGDYEGGGFDY.
本发明进一步提供一种双特异性抗原结合蛋白,优选为一双特异性抗体,其包括一会结合肿瘤抗原的可变结构域及一会结合人类CD3的可变结构域,该会结合人类CD3的可变结构域包括一抗体可变结构域,该抗体可变结构域包括一重链可变区及一轻链可变区,其中该重链可变区包括一CDR1、CDR2及CDR3,该CDR1、CDR2及CDR3包括下列氨基酸序列:The present invention further provides a bispecific antigen-binding protein, preferably a bispecific antibody, comprising a variable domain that binds to a tumor antigen and a variable domain that binds human CD3, which binds to human CD3 The variable domain includes an antibody variable domain, the antibody variable domain includes a heavy chain variable region and a light chain variable region, wherein the heavy chain variable region includes a CDR1, CDR2 and CDR3, the CDR1, CDR2 and CDR3 include the following amino acid sequences:
CDR1:DYTMH;CDR1: DYTMH;
CDR2:DISWSX7GX8X9X10YADSVKG;CDR2: DISSWX 7 GX 8 X 9 X 10 YADSVKG;
CDR3:DHX11GYGDYEGGGFDX12;CDR3: DHX 11 GYGDYEGGGFDX 12 ;
其中in
X7=S或G;X 7 =S or G;
X8=S或T;X 8 =S or T;
X9=I或T;X 9 =I or T;
X10=G或Y;X 10 =G or Y;
X11=R或M;X 11 =R or M;
X12=H或Y,X 12 =H or Y,
优选X7、X8、X9及X10为S、S、I及G或是G、S、I及Y或是S、T、T和G,并且优选X11及X12为R及H,或是R及Y,或是M及Y,更优选X7、X8、X9、X10、X11和X12为S、S、I、G、R和H或是G、S、I、Y、R及Y或是S、T、T、G、M及Y,或是换句话讲,优选X7、X8、X9和X10为S、S、I及G,并且X11及X12为R和H;或X7、X8、X9和X10为G、S、I和Y和X11和X12为R和Y;或X7、X8、X9和X10为S、T、T和G,并且X11和X12为M及Y。Preferably X 7 , X 8 , X 9 and X 10 are S, S, I and G or G, S, I and Y or S, T, T and G, and preferably X 11 and X 12 are R and H , or R and Y, or M and Y, more preferably X 7 , X 8 , X 9 , X 10 , X 11 and X 12 are S, S, I, G, R and H or G, S, I, Y, R and Y are either S, T, T, G, M and Y, or in other words, preferably X 7 , X 8 , X 9 and X 10 are S, S, I and G, and X 11 and X 12 are R and H; or X 7 , X 8 , X 9 and X 10 are G, S, I and Y and X 11 and X 12 are R and Y; or X 7 , X 8 , X 9 and X 10 are S, T, T and G, and X 11 and X 12 are M and Y.
本发明进一步提供一种双特异性抗原结合蛋白,优选为一双特异性抗体,其包括一会结合肿瘤抗原的可变结构域及一会结合人类CD3的可变结构域,其中该可变结构域各包括一不同的重链可变区及一共同的轻链可变区,以及其中该会结合人类CD3的可变结构域的重链可变区包括氨基酸序列The present invention further provides a bispecific antigen binding protein, preferably a bispecific antibody, comprising a variable domain that binds to a tumor antigen and a variable domain that binds human CD3, wherein the variable domain Each includes a distinct heavy chain variable region and a common light chain variable region, and wherein the heavy chain variable region that binds the variable domain of human CD3 includes the amino acid sequence
于此类CDRs以外的一个或多个位置处具有0-10个、优选0-5个氨基酸变异、插入、缺失、取代、加入或其组合。There are 0-10, preferably 0-5 amino acid variations, insertions, deletions, substitutions, additions or combinations thereof at one or more positions other than such CDRs.
在一具体例中,该会结合肿瘤抗原的可变结构域的重链可变区优选包括MF8233(EGFR)In a specific example, the heavy chain variable region of the variable domain that binds to the tumor antigen preferably comprises MF8233 (EGFR)
QVQLVQSGAEVKKPGASVKVSCKASGYTFTSYGISWVRQAPGQGLEWMGWISAYNANTNYAQKLQGRVTMTTDTSTSTAYMELRSLRSDDTAVYYCAKDRHWHWWLDAFDYWGQGTLVTVSS的氨基酸序列Amino acid sequence of QVQLVQSGAEVKKPGASVKVSCKASGYTFTSYGISWVRQAPGQGLEWMGWISAYNANTNYAQKLQGRVTMTTDTSTSTAYMELRSLRSDDTAVYYCAKDRHWHWWLDAFDYWGQGTLVTVSS
于该CDR以外的一个或多个位置处具有0-10个、优选0-5个氨基酸变异、插入、缺失、取代、加入或其的组合。There are 0-10, preferably 0-5 amino acid variations, insertions, deletions, substitutions, additions or combinations thereof at one or more positions other than the CDR.
在另一具体例中,该会结合肿瘤抗原的可变结构域的重链可变区优选包括In another specific example, the variable domain of the heavy chain of the variable domain that binds the tumor antigen preferably includes
MF4327(CLEC12A)的氨基酸序列Amino acid sequence of MF4327(CLEC12A)
于此类CDRs以外的一个或多个位置处具有0-10个、优选0-5个氨基酸变异、插入、缺失、取代、加入或其组合。There are 0-10, preferably 0-5 amino acid variations, insertions, deletions, substitutions, additions or combinations thereof at one or more positions other than such CDRs.
本发明进一步提供本发明的抗体或其衍生物或本发明的药学组合物,供用于治疗有需要的一个体。关于治疗一具有一肿瘤或处于有一肿瘤的风险的个体,优选的是该抗体为本发明的双特异性抗体。优选其中该CD3结合抗体包括一会结合肿瘤抗原的重/轻链组合。The present invention further provides an antibody or derivative thereof of the present invention or a pharmaceutical composition of the present invention for use in the treatment of an individual in need thereof. With regard to treating an individual having or at risk of having a tumor, it is preferred that the antibody is a bispecific antibody of the invention. Preferably wherein the CD3 binding antibody comprises a heavy/light chain combination that binds tumor antigens.
提供CD3/肿瘤抗原双特异性抗体及包括这些双特异性抗体的药学组合物供用于治疗实性或血液肿瘤。优选的实性肿瘤为属于上皮来源;妇科癌症例如卵巢癌及内膜癌;前列腺癌、脑癌及任何其他实性肿瘤。CD3/tumor antigen bispecific antibodies and pharmaceutical compositions comprising these bispecific antibodies are provided for use in the treatment of solid or hematological tumors. Preferred solid tumors are of epithelial origin; gynecological cancers such as ovarian and endometrial cancers; prostate cancer, brain cancer and any other solid tumor.
还提供本发明的CD3/肿瘤抗原双特异性抗体或其衍生物或包括这些双特异性抗体或其衍生物的药学组合物,供用于治疗各种骨髓来源的白血病及白血病前期疾病但此外B细胞淋巴瘤。可根据本发明治疗的疾病包括骨髓性白血病或白血病前期疾病例如如急性骨髓性白血病(AML)、骨髓发育不良症候群(MDS)及慢性骨髓性白血病(CML),及霍奇金氏(Hodgkin)淋巴瘤及多数非霍奇金淋巴瘤。并且B-ALL;T-ALL、外膜细胞淋巴瘤也是本发明的抗体优选的治疗标靶。因此,本发明提供一种如本发明的双特异性全长IgG抗体供用作治疗骨髓发育不良症候群(MDS)、慢性骨髓性白血病(CML)、多发性骨髓瘤(MM)或优选为急性骨髓性白血病(AML)的制剂。还提供一种如本发明的双特异性IgG抗体于制备用于治疗或预防MDS、CML、MM或优选为AML的一药剂的用途。Also provided are CD3/tumor antigen bispecific antibodies or derivatives thereof of the present invention or pharmaceutical compositions comprising these bispecific antibodies or derivatives thereof for use in the treatment of various bone marrow-derived leukemias and preleukemia diseases but in addition B cells lymphoma. Diseases that can be treated according to the present invention include myeloid leukemia or preleukemia diseases such as acute myeloid leukemia (AML), myelodysplastic syndrome (MDS) and chronic myelogenous leukemia (CML), and Hodgkin's lymphoid tumor and most non-Hodgkin lymphomas. And B-ALL; T-ALL, adventitial cell lymphoma are also preferred therapeutic targets of the antibody of the present invention. Accordingly, the present invention provides a bispecific full-length IgG antibody according to the present invention for use in the treatment of myelodysplastic syndrome (MDS), chronic myeloid leukemia (CML), multiple myeloma (MM) or preferably acute myeloid Preparations for Leukemia (AML). Also provided is the use of a bispecific IgG antibody according to the invention for the manufacture of a medicament for the treatment or prevention of MDS, CML, MM or preferably AML.
如本发明的抗体待投予至患者的量典型落于治疗范围(therapeutic window)内,意思是使用足够获得治疗效应的量,同时该量不超过会导致不可接受程度的副作用的阈值。获得所欲治疗效应所需的抗体量越低,治疗范围典型会越大。因而,如本发明的抗体于低剂量发挥足够的治疗效应为优选的。The amount to be administered to a patient as an antibody of the invention typically falls within the therapeutic window, meaning that an amount sufficient to obtain a therapeutic effect is used, while not exceeding a threshold that would result in an unacceptable degree of side effects. The lower the amount of antibody required to obtain the desired therapeutic effect, the greater the therapeutic range will typically be. Thus, it is preferred that the antibodies of the present invention exert sufficient therapeutic effects at low doses.
在欧洲及美国每年有大概30.000位患者诊断为有AML。这些患者中多数是60岁以上。年龄较大是AML的结果的主要负面决定因素且重症的老年AML患者的长期(五年)存活大概是10%。在诱导性化学治疗后已经缓解的几乎全部患者,于3年内都观察到疾病进展。目前缓解后的治疗于有AML的老年患者显示出的价值有限,如果有的话。因而,残留抗性白血病的负担仍很大,且存活的药物抗性白血病细胞亚族群快速产生复发。这些化学治疗无反应AML肿瘤细胞需要有完全不同的作用模式的新型药物为了诱发且支持完全缓解。纵然用许多加强的化学治疗组合可于超过50%的老年AML患者及大约80%年轻患者达到完全缓解(CR),但反应及存活的进展依然是重大的研究挑战。于最近发表的于有AML的年龄较大的患者的65个随机临床试验的网络统合分析(15.110位患者)中,多数修正的研究诱导摄生法有相似或甚至更糟的功效解析,相较于传统用道诺霉素(daunorubicin)及阿糖胞苷(cytarabine)的3+7诱导摄生法而言。此标准的AML治疗与高发病率且甚至是死亡率有关连。CR再发的多数患者由于化学治疗后剩余的白血病干细胞。由于不可接受的毒性而限制了进一步的剂量加大。对优选毒性较小的新的治疗方式的迫切需要因而浮现,尤其是于有AML的老年患者。Approximately 30.000 patients are diagnosed with AML each year in Europe and the United States. Most of these patients are over 60 years old. Older age is a major negative determinant of outcome in AML and long-term (five-year) survival in severe elderly AML patients is approximately 10%. Disease progression was observed within 3 years in almost all patients who had been in remission after induction chemotherapy. Current post-remission treatments have shown limited, if any, value in older patients with AML. Thus, the burden of residual resistant leukemia remains high, and the subpopulation of surviving drug-resistant leukemia cells rapidly relapses. These chemotherapy-unresponsive AML tumor cells require novel drugs with completely different modes of action in order to induce and support complete remission. Although complete remission (CR) can be achieved in more than 50% of elderly AML patients and approximately 80% of younger patients with many intensified chemotherapy combinations, progression of response and survival remains a significant research challenge. In a recently published network meta-analysis of 65 randomized clinical trials (15.110 patients) in older patients with AML, most modified study-induced regimens had similar or even worse efficacy analysis compared to Traditionally use daunorubicin and cytarabine for 3+7 induction. This standard of AML treatment is associated with high morbidity and even mortality. The majority of patients with CR recurrence are due to residual leukemia stem cells after chemotherapy. Further dose escalation was limited by unacceptable toxicity. An urgent need for new therapeutic modalities, preferably less toxic, has thus emerged, especially in elderly patients with AML.
化学治疗没有反应的AML的治疗能通过使用双特异性抗体来使T细胞从患者自身的免疫系统复位向至AML肿瘤细胞且随后活化肿瘤特异性T细胞而达成。此方法也被称为“T细胞衔接(T-cell engaging)方法”。以此方式,强化了患者的免疫系统且重新靶向以攻击及根除AML肿瘤细胞。例如,CD3xCLEC12A双特异性IgG抗体有效地复位向T细胞至AML肿瘤细胞,借此诱发AML肿瘤细胞溶解。Treatment of AML that does not respond to chemotherapy can be accomplished by using bispecific antibodies to relocate T cells from the patient's own immune system to AML tumor cells and subsequently activate tumor-specific T cells. This method is also referred to as the "T-cell engaging method". In this way, the patient's immune system is strengthened and retargeted to attack and eradicate AML tumor cells. For example, the CD3xCLEC12A bispecific IgG antibody effectively resets T cells to AML tumor cells, thereby inducing AML tumor lysis.
实施例Example
细胞株cell line
BxPC3人类胰脏癌细胞株。BxPC3 human pancreatic cancer cell line.
HCT-116人类结肠癌细胞株。HCT-116 human colon cancer cell line.
用CD3来免疫小鼠immunization with CD3 mouse
为了产生结合CD3的人类抗体,就该人类共同的轻链和一人类重链(HC)微小基因座(minilocus)(包括一群挑选的人类V基因节段、所有的人类Ds和所有的人类Js)而为基因转殖的小鼠(参见W02009/157771,在此被并入本案以作为参考),用含TCR/CD3的脂质球(TCR/CD3 containing lipoparticles)(Intergral Molecular)来被免疫。这些小鼠称为小鼠。有关于特异性重链可变区,或者具有此处所公开的序列的三价多聚体,其可借由具有本领域中的通常技艺的人士所知道的方式来生成。To generate human antibodies that bind CD3, the human common light chain and a human heavy chain (HC) minilocus (comprising a selected set of human V gene segments, all human Ds, and all human Js) Instead, transgenic mice (see WO2009/157771, incorporated herein by reference) were immunized with TCR/CD3 containing lipoparticles (Intergral Molecular). These mice are called mice. There are specific heavy chain variable regions, or trivalent multimers having the sequences disclosed herein, which can be produced by means known to those of ordinary skill in the art.
小鼠以含有Hek293T-衍生的人类5D5M TCR/CD3的脂质球,继而为人类T细胞被免疫,以产生一种抗-TCR/CDR3免疫反应及产生抗-TCR/CD3抗体群组。 Mice were immunized with lipid globules containing Hek293T-derived human 5D5M TCR/CD3 followed by human T cells to generate an anti-TCR/CDR3 immune response and to generate a population of anti-TCR/CD3 antibodies.
脂质球直接从细胞表面浓缩(concentrate)构形上完整的膜蛋白,允许这些复杂的蛋白有如供抗体免疫和筛选的可溶性高浓度蛋白被操作(manipulated)。Lipid globules concentrate conformally intact membrane proteins directly from the cell surface, allowing these complex proteins to be manipulated as soluble high-concentration proteins for antibody immunization and screening.
在本研究中使用于供免疫作用的脂质球含有5D5M TCRαβ组合。含有该5D5M TCRαβ组合的载体借由暂时转染至HEK293T细胞(Intergral Molecular)中被合成、转殖和使用于产生含有此TCR/CD3组合的脂质球。The lipid globules used for immunization in this study contained the 5D5M TCRαβ combination. A vector containing the 5D5M TCRαβ combination was synthesized, transfected and used to generate lipid globules containing this TCR/CD3 combination by transient transfection into HEK293T cells (Intergral Molecular).
5D5M TCRα5D5M TCRα
5D5M TCRβ5D5M TCRβ
小鼠使用于使用TCR/CD3脂质球及初级人类T细胞的免疫作用。 Mice were used for immunization using TCR/CD3 lipid globules and primary human T cells.
免疫时间表(immunization schedule)包括在第35、56、77和98天的时点(points),于此类时点判定抗原-特异性Ig血清效价,其通过使用利用抗小鼠IgG检测的QTG衍生的3SDX TCR/CD3阳性和阴性脂质球的ELISA以及通过使用CD3c5E-Fc融合蛋白作为阳性对照的ELISA来进行。在第35天抽取的血清内观察到的反应性将会决定那些小鼠发展出相关的抗TCR/CD3反应。The immunization schedule included points on
对于所有免疫的小鼠,当有下列情况时,收集且储存淋巴材料用于抗体探索:For all immunized mice, collect and store lymphatic material for antibody discovery when:
对于人类TCR/CD3的效价为1/300(在使用脂质球的ELISA中);或:1/300 titer for human TCR/CD3 (in ELISA using lipid spheres); or:
对于人类TCR/CD3的效价为<1/300且>1/100而且在最后的补强注射免疫(booster immunization)期间当中不会增高。The titers for human TCR/CD3 were <1/300 and >1/100 and did not increase during the final booster immunization.
使用脂质球的激活免疫(Priming immunisation)Priming immunisation using lipid globules
为了激活(prime)小鼠对于TCR/CD3的体液性免疫反应,使用含有人类5D5M TCRαβ组合的脂质球用于免疫。脂质球和Gerbu佐剂一起使用于第一次和第二次注射。To activate (prime) Humoral immune response to TCR/CD3 in mice using lipid globules containing a combination of human 5D5M TCRαβ for immunization. Lipid globules were used with Gerbu adjuvant for the first and second injections.
使用多株T细胞的补强注射免疫Booster immunization using polyline T cells
小鼠通过皮下注射细胞悬浮液被免疫。第一次的补强注射免疫(第28天)包括由配于PBS的细胞和佐剂所构成的混合物且所有随后的注射品是仅由配于PBS的细胞所构成。在第35天时对人类TCR/CD3已发展出1/300的血清IgG效价(借由使用脂质球的ELISA来测定)的小鼠在第42、43和44天接受使用细胞的附加注射。不符合这些标准的小鼠接受使用细胞的补强注射免疫(第42天和第49天)。所有随后的免疫都是如配于PBS的细胞的皮下注射来给予。在最后的免疫的后,牺牲小鼠,为了血清被放血且采集脾脏和左侧腹股沟淋巴结。Mice were immunized by subcutaneous injection of cell suspensions. The first booster immunization (day 28) consisted of a mixture of cells in PBS and adjuvant and all subsequent injections consisted of cells in PBS only. Mice that had developed serum IgG titers of 1/300 (determined by ELISA using lipid spheres) against human TCR/CD3 on day 35 received additional injections with cells on days 42, 43 and 44. Mice that did not meet these criteria received booster immunizations with cells (days 42 and 49). All subsequent immunizations were administered as subcutaneous injections of cells in PBS. After the final immunization, mice were sacrificed, bled for serum and the spleen and left inguinal lymph node were collected.
以ELISA筛选来自经免疫的小鼠的血清Screening of sera from immunized mice by ELISA
通过ELISA使用含有TCR/CD3的脂质球及空(null)脂质球来筛选期中(interim)血清IgG效价。使用抗小鼠IgG染色来测定血清IgG效价,因为这种染色经证明是最为灵敏的。Interim serum IgG titers were screened by ELISA using TCR/CD3 containing lipid globules and null lipid globules. Serum IgG titers were determined using anti-mouse IgG staining, as this staining proved to be the most sensitive.
通过RT-PCR转殖VH基因来产生“免疫”噬菌体抗体库Generation of "immune" phage antibody libraries by transfection of VH genes by RT-PCR
使用来自成功免疫的小鼠的腹股沟淋巴结来构造“免疫”噬菌体抗体库。使用Trizol LS以从淋巴组织萃取出RNA且使用1μg的总RNA于一种用IgG-CH1特异性引子的RT反应中。接而使用产生的cDNA、利用内部开发的VH特异性引子来扩增VH编码cDNA的多株库(polyclonal pool),基本上如于Marks等人(J Mol Biol.1991 Dec 5;222(3):581-97)乙文中所述。产生的PCR产物接而选殖至一噬粒(phagemid)载体中用于显示Fab片段在噬菌体上,如Haard等人(J Biol Chem.1999 Jun 25;274(26):18218-30)乙文中所述,但有例外是该轻链对于每个抗体而言是相同的并且为该载体所编码。在连接的后,此类噬粒用来转形大肠杆菌TG1细菌且转形的细菌被平盘培养在含有安比西林和葡萄糖的LB-琼脂平盘上。所有的噬菌体库都含有>10e6个转形体并且具有>80%的插入物频率(insert frequency)。细菌在过夜生长后被收获并且根据已建立的程序用来制备噬菌体(de Haard等人,JBiolChem.1999 Jun 25;274(26):18218-30)。Inguinal lymph nodes from successfully immunized mice were used to construct "immune" phage antibody libraries. Trizol LS was used to extract RNA from lymphoid tissue and 1 μg of total RNA was used in an RT reaction with IgG-CH1 specific primers. The resulting cDNA was then used to amplify a polyclonal pool of VH-encoding cDNA using in-house developed VH-specific primers, essentially as described in Marks et al. (J Mol Biol. 1991
挑选携带特异性地结合人类CD3的Fab片段的噬菌体。Phages carrying Fab fragments that specifically bind human CD3 were selected.
根据标准程序来救援(rescued)噬菌体库(J Mol Biol.1991 Dec 5;222(3):581-97;J Biol Chem.1999 Jun 25;274(26):18218-30)且以一或多轮的免疫噬菌体抗体库的选择来挑选噬菌体。于第1轮,使重组CD3蛋白涂覆于maxisorpTM ELISA平盘的孔或NUNC免疫管上,而第2轮,使用重组CD3蛋白或过度表现人类CD3蛋白的细胞。用4%ELK阻断maxisorpTMELISA平盘或免疫管。噬菌体抗体库也用4%ELK被阻断且在噬菌体库添加至经涂覆的抗原的前,也用过量的人类IgG被阻断以耗乏Fc区域结合物(binder)。Phage libraries were rescued according to standard procedures (J Mol Biol. 1991
在振荡条件下,噬菌体库与经涂覆的蛋白的孵育于室温执行2hrs。平盘或管子继而用配于PBS的0.05%吐温-20(Tween-20)清洗,接着用PBS清洗5至10次。使用50mM甘胺酸(pH2.2)溶析结合的噬菌体以及添加至大肠杆菌TG-1且在37℃下孵育用于噬菌体感染。Incubation of the phage pool with the coated protein was performed at room temperature for 2 hrs under shaking conditions. The plate or tube was then washed with 0.05% Tween-20 in PBS, followed by 5 to 10 washes with PBS. Bound phage was eluted using 50 mM glycine (pH 2.2) and added to E. coli TG-1 and incubated at 37°C for phage infection.
随后,将感染的细菌被平盘培养至含有安比西林和葡萄糖的琼脂平盘上,且在37℃下孵育过夜。于第1轮选择的后,从平盘刮掉群落且合并然后救援及扩增以制备富集的第1轮噬菌体库(pool)供用于合成的库(repertoire)。关于免疫库,于第1轮噬菌体选择的后筛选单一群落供用于标靶结合。Subsequently, infected bacteria were plated onto agar plates containing ampicillin and glucose and incubated overnight at 37°C. After the 1st round of selection, colonies were scraped from the plate and pooled and then rescued and amplified to make an enriched 1st round of phage pools for synthetic repertoires. For immune pools, single colonies were screened for target binding after the first round of phage selection.
抗体选殖及生产Antibody colonization and production
本文中使用的双特异性抗体彼此不同的处通常仅在一或二个可变结构域的重链可变区的特定氨基酸序列。抗体生产通过选殖重链可变区至表现载体的内用于表现重及轻链。用于生产双特异性抗体的方法为本技艺已知的。The bispecific antibodies used herein generally differ from each other only by the specific amino acid sequence of the heavy chain variable region of one or two variable domains. Antibody production is used to express heavy and light chains by breeding the heavy chain variable regions into expression vectors. Methods for producing bispecific antibodies are known in the art.
简言的,编码CD3靶向的可变结构域的重链可变区的DNA选殖至MV1624载体中(见图10A),该载体编码KK残基(L351K,T366K)于CH3区域供用于产生IgG重链异二聚体(WO2013/157954及WO2013/157953)。该Fc恒定区域于CH2内含有突变以使Fc效应子功能沉默。编码该重链可变区的DNA替代构造物内的充填(stuffer)区域。该可变区的前面有一编码的HC信号肽(未显示)。编码EGFR靶向的可变结构域的重链可变区的DNA选殖至MV1625载体中(图10B),该载体表现双特异性抗体基础的抗体部分的第二重链,带有L351D-L368E突变于CH3区域内(WO2013/157954及WO2013/157953)。编码该重链可变区的DNA替代构造物内的充填区域。该可变区的前面有一编码的HC信号肽(未显示)。二种构造物还含有一种表现匣用于表现IGKV1-39/jk1轻链。同时表现该二重链及所提及的轻链导致了双特异性抗体的生产。Briefly, DNA encoding the heavy chain variable region of the CD3-targeted variable domain was cloned into the MV1624 vector (see Figure 10A), which encodes KK residues (L351K, T366K) in the CH3 region for generation IgG heavy chain heterodimers (WO2013/157954 and WO2013/157953). The Fc constant region contains mutations within CH2 to silence Fc effector function. The DNA encoding the heavy chain variable region replaces the stuffer region within the construct. The variable region is preceded by an encoded HC signal peptide (not shown). DNA encoding the heavy chain variable region of the EGFR-targeted variable domain was cloned into the MV1625 vector (Fig. 10B), which expresses the second heavy chain of the antibody portion of the bispecific antibody basis with L351D-L368E Mutations were made within the CH3 region (WO2013/157954 and WO2013/157953). The DNA encoding the heavy chain variable region replaces the stuffer region within the construct. The variable region is preceded by an encoded HC signal peptide (not shown). Both constructs also contained an expression cassette for expression of the IGKV1-39/jk1 light chain. Simultaneous representation of this double chain and the mentioned light chain leads to the production of bispecific antibodies.
使用293-F细胞供用于表现所设计的抗体于24孔平盘格式。在转染前二天,293-F细胞原液以1∶1的比例于293-F培养基中分离(split)并且在155rpm的回转式振荡速度(orbital shaking speed)的下、于37℃和8%CO2下孵育过夜。细胞在转染的前一天稀释至5x105个细胞/mL的密度。4mL的悬浮细胞播种(seeded)至24深孔培养平盘内,以一个透气封件被覆盖并在285rpm的回转式振荡速度的下、于37℃和8%CO2下孵育过夜。在转染当天,混合4.8mL的293-F培养基与240μg的线性聚乙烯亚胺(polyethylenimine)(PEI)(MW 25,000)。对于每一种要生产的IgG,200μL的293F培养基-PEI混合物添加8μL的DNA(对于异二聚体为4μL的编码各个重链的DNA)。在温和地加入至细胞的前,该混合物在室温下孵育历时20分钟。在转染的后一天,稀释于500μL的293F培养基中的青霉素-链霉素(Pen Strep)添加至每个孔内。此类平盘在285rpm的回转式振荡速度的下、于37℃和8%CO2下孵育直到转染的后7天要收获的时。平盘在500g的下被离心5min,含有IgGs之上清液使用10-12μm熔喷聚丙烯过滤板(melt blown polypropylene filter plates)被过滤并在纯化前储存在-20℃的下。293-F cells were used for expression of the designed antibodies in a 24-well plate format. Two days prior to transfection, 293-F cell stock was split in 293-F medium at a ratio of 1:1 and at 155 rpm orbital shaking speed at 37°C and 8% Incubate overnight under CO2 . Cells were diluted to a density of 5x105 cells/mL the day before transfection. 4 mL of suspension cells were seeded into 24 deep well culture plates, covered with a gas permeable seal and incubated overnight at 37°C and 8% CO 2 with a rotary shaking speed of 285 rpm. On the day of transfection, 4.8 mL of 293-F medium was mixed with 240 μg of linear polyethylenimine (PEI) (MW 25,000). For each IgG to be produced, 200 μL of the 293F medium-PEI mixture was supplemented with 8 μL of DNA (4 μL of DNA encoding each heavy chain for the heterodimer). The mixture was incubated at room temperature for 20 minutes before being gently added to the cells. One day after transfection, penicillin-streptomycin (Pen Strep) diluted in 500 [mu]L of 293F medium was added to each well. Such plates were incubated at 37° C. and 8% CO 2 with a rotary shaking speed of 285 rpm until harvesting 7 days after transfection. The plates were centrifuged at 500 g for 5 min and the IgGs-containing supernatant was filtered using 10-12 μm melt blown polypropylene filter plates and stored at -20°C prior to purification.
从培养上清液纯化抗体Antibody purification from culture supernatant
收获含抗体的培养基且离心以移除细胞碎片。随后加入蛋白A Sepharose珠粒至该培养基。该培养基和蛋白A Sepharose珠粒连同抗体一起孵育以允许结合。Antibody-containing medium was harvested and centrifuged to remove cellular debris. Protein A Sepharose beads were then added to the medium. The medium and Protein A Sepharose beads were incubated with the antibody to allow binding.
在孵育的后通过真空过滤器以从培养基单离珠粒并且清洗。此类抗体通过用溶析缓冲液孵育而从珠粒溶析出。After incubation the beads were passed through a vacuum filter to detach the beads from the medium and washed. Such antibodies are eluted from the beads by incubation with lysis buffer.
选择性地,纯化的IgG的缓冲液被交换/脱盐(desalted)。Optionally, the buffer of purified IgG is exchanged/desalted.
缓冲液交换buffer exchange
为了要将纯化的抗体脱盐,使用一滤板或滤柱(filter column)来离心抗体部分(fraction)。离心滤板或滤柱以减少该抗体部分的体积。随后,添加PBS或所需要的缓冲液至该部分以用一低盐缓冲液来替换该缓冲液。选择性地,此离心步骤接着添加缓冲液步骤被重复俾以进一步脱盐该抗体的储存缓冲液。To desalt the purified antibody, a filter plate or filter column is used to centrifuge the antibody fraction. Centrifuge the filter plate or column to reduce the volume of the antibody fraction. Subsequently, PBS or the desired buffer is added to the portion to replace the buffer with a low salt buffer. Optionally, this centrifugation step followed by the buffer addition step is repeated to further desalt the antibody storage buffer.
抗体肿瘤抗原特异性T细胞活化及BxPC3细胞或HTC-116细胞的溶解。Antibody tumor antigen-specific T cell activation and lysis of BxPC3 cells or HTC-116 cells.
特定CD3 x肿瘤抗原双特异性IgG组合诱发肿瘤抗原特异性T细胞活化及肿瘤抗原阳性肿瘤细胞的溶解以一细胞毒性分析来测试。效应子细胞为健康的供体衍生的休眠T细胞而标靶细胞为BxPC3细胞或HTC-116细胞。The specific CD3 x tumor antigen bispecific IgG combination induced tumor antigen specific T cell activation and tumor antigen positive tumor cell lysis was tested in a cytotoxicity assay. Effector cells are healthy donor-derived resting T cells and target cells are BxPC3 cells or HTC-116 cells.
根据标准技术、使用菲科尔(Ficoll)和EasySep人类T细胞单离套组而从来自健康的供体的全血单离出休眠T细胞,通过抗CD3抗体、使用流动式细胞测量术分析来检查>95%T细胞纯度以及随后被冷冻保存。关于细胞毒性分析,冷冻保存的T细胞被解冻并且使用,如果在解冻的时其的生存力借由标准锥虫蓝染色法来测定是>90%。细胞毒性分析简而言的,解冻的休眠T细胞和BxPC3或HCT116标靶细胞以5∶1的E∶T比率共培养历时48小时。抗体以一稀释范围被测试。一CD3单特异性抗体及一EGFR单特异性抗体,以及不相关的IgG1同型对照mAb含括在分析内作为对照(例如一种结合CD3及另一抗原如破伤风类毒素(TT)的抗体)。T细胞活化使用流动式细胞测量术被定量;CD8T细胞根据CD8表现被圈选(gated)并且随后通过测量T细胞上的CD69表现来分析它们的活化状态。标靶细胞溶解通过测量活细胞的分率(fraction)而测定,其通过CellTiterGlo(Promega)所评估的ATP电平来测量。ATP电平,通过在Envision微量盘读数器上的发光来测量,结果为相对光单位(RLU)数值,其使用GraphPad Prism被分析。Resting T cells were isolated from whole blood from healthy donors according to standard techniques using Ficoll and the EasySep Human T Cell Isolation Kit, analyzed by anti-CD3 antibody using flow cytometry T cells were checked for >95% purity and subsequently cryopreserved. For cytotoxicity assays, cryopreserved T cells were thawed and used if their viability was >90% when thawed by standard trypan blue staining. Cytotoxicity Assay Briefly, thawed resting T cells and BxPC3 or HCT116 target cells were co-cultured at a 5:1 E:T ratio for 48 hours. Antibodies were tested at a dilution range. A CD3 monospecific antibody and an EGFR monospecific antibody, and an irrelevant IgG1 isotype control mAb were included in the assay as controls (eg, an antibody that binds CD3 and another antigen such as tetanus toxoid (TT)) . T cell activation was quantified using flow cytometry; CD8 T cells were gated according to CD8 expression and their activation status was subsequently analyzed by measuring CD69 expression on T cells. Target cell lysis was determined by measuring the fraction of viable cells as assessed by ATP levels as assessed by CellTiterGlo (Promega). ATP levels, measured by luminescence on an Envision microplate reader, result in relative light unit (RLU) values, which are analyzed using GraphPad Prism.
每一个样本的标靶细胞溶解计算如下:The target cell lysis for each sample was calculated as follows:
杀灭%=(100-(RLU样本/RLU无IgG)×100)。% Kill = (100-(RLU sample/RLU no IgG) x 100).
于此分析中,该双特异性抗体具有二个结合域。一个一者靶向EGFR且另一者靶向CD3。二个结合域都具有相同的(共同的)轻链可变区(VL)和不同的重链可变区(VH)。该EGFR靶向的结合域具有一VH其带有MF8233的氨基酸序列。该CD3靶向的结合域具有一VH其带有针对CD3的已指明的MFs中的一的氨基酸序列。该双特异性抗体于CH2内含有突变以使Fc效应子功能沈默。In this assay, the bispecific antibody has two binding domains. One targets EGFR and the other targets CD3. Both binding domains have the same (common) light chain variable region (VL) and different heavy chain variable regions (VH). The EGFR-targeted binding domain has a VH with the amino acid sequence of MF8233. The CD3-targeted binding domain has a VH with the amino acid sequence of one of the designated MFs for CD3. This bispecific antibody contains mutations in CH2 to silence Fc effector function.
该抗体MF8233xMF8397诱发CD4及CD8 T细胞上的CD69(Fig 4-6)及CD25(Fig 4-6)的向上调节,其以5∶1的E±T比率共培养于48小时后被判定。T细胞媒介的溶解于48小时后测量。The antibody MF8233xMF8397 induced upregulation of CD69 (Fig 4-6) and CD25 (Fig 4-6) on CD4 and CD8 T cells, which were determined after 48 hours of co-culture at a 5:1 E±T ratio. Lysis of the T cell vehicle was measured after 48 hours.
CD3双特异性抗体特征化CD3 Bispecific Antibody Characterization
一种候选EGFR/CD3 IgG双特异性抗体的结合作用能使用任何合适的分析法被测试。例如,与HPB-ALL细胞上的膜表现的CD3结合(DSMZ,ACC 483)能通过移动式细胞测量术被评估(根据如前于WO2014/051433内所述的FACS程序)。在一具体例中,一种候选EGFR/CD3双特异性抗体与HPB ALL细胞上的CD3结合通过流动式细胞测量术被证实,其根据本技艺已知的标准程序来执行。与细胞表现的CD3的结合能使用转染CD3δ/ε或CD3γ/ε的CHO细胞来确定。该候选双特异性IgG1与EGFR的结合能使用转染EGFR表现构造物的BxPC3及HCT-116以及CHO细胞来判定;一种CD3单特异性抗体及一种EGFR单特异性抗体,以及不相关的IgG1同型对照mAb含括在分析内作为对照(例如一种结合CD3及另一抗原如破伤风类毒素(TT)的抗体)。The binding of a candidate EGFR/CD3 IgG bispecific antibody can be tested using any suitable assay. For example, binding to membrane expressed CD3 on HPB-ALL cells (DSMZ, ACC 483) can be assessed by mobile cytometry (according to the FACS procedure as previously described in WO2014/051433). In one embodiment, binding of a candidate EGFR/CD3 bispecific antibody to CD3 on HPB ALL cells was demonstrated by flow cytometry, performed according to standard procedures known in the art. Binding to cell-expressed CD3 can be determined using CHO cells transfected with CD3δ/ε or CD3γ/ε. Binding of this candidate bispecific IgG1 to EGFR can be determined using BxPC3 and HCT-116 and CHO cells transfected with EGFR expressing constructs; a CD3 monospecific antibody and an EGFR monospecific antibody, and unrelated An IgGl isotype control mAb was included in the assay as a control (eg, an antibody that binds CD3 and another antigen such as tetanus toxoid (TT)).
从超级群1、3及4产生另外的殖株Generation of additional clones from
从免疫噬菌体库筛选(如于“挑选携带特异性地结合人类CD3的Fab片段的噬菌体”节中所述),携带特异性地结合人类CD3的Fab片段的附加的殖株被特征化。从超级群1鉴定出附加的殖株,包括MF8048、MF8101及MF8056。从超级群3及超级群4鉴定出附加的殖株,包括超级群3的MF8562及超级群4的MF8998。From immune phage library screening (as described in the section "Selecting phages carrying Fab fragments that specifically bind human CD3"), additional clones carrying Fab fragments that specifically bind human CD3 were characterized. Additional clones were identified from
使用次世代定序仪(NGS)分析从超级群4鉴定出另外的新殖株。NGS是对从用来产生抗CD3群组的小鼠呈现的VH基因库(pool)被执行。为了这个目的,从不同的小鼠获得的序列资料集与属于超级群4MF的MF序列作比较。此导致识别出属于超级群4序列变异体殖株MF10401及MF10428。不同序列的HCDR1及HCDR2内发现到数种不同的突变。Additional neocolons were identified from
所有来自超级群1、3及4的附加殖株的VH序列选殖至MV1624(DM-KK)载体中且表现为CD3xEGFR双特异性格式用于进一步特征化,如于以上的“抗体选殖及生产”节中所述。The VH sequences of all additional clones from
来自超级群1及4的另外的殖株的特征化Characterization of additional clones from
来自超级群1附加的殖株就其呈双特异性格式的功能活性被特征化。该双特异性CD3xEGFR抗体的EGFR结合臂具有MF8233编码的氨基酸序列。作为对照,这些CD3殖株还以MF1337编码的氨基酸序列的另一抗原(例如破伤风类毒素)被测试。来自超级群1的参考MFs(MF8057及MF8058)含括在内以根据如以上的:“抗体肿瘤抗原特异性T细胞活化及BxPC3细胞或HTC-116细胞的溶解”及“CD3双特异性抗体特征化”节中所述来直接比较序列变异体的亲和力与来自超级群1已经特征化的MF殖株的亲和力。使用移动式细胞测量术来执行表现人类CD3-TCR错合物的HPB-ALL细胞的结合亲和力(图14A及图18)以及以细胞毒性分析(图14B-E)分析来执行肿瘤抗原阳性肿瘤细胞(HCT-116)的T细胞活化及溶解。用具有MF1337对照臂的双特异性抗体没有观察到标靶细胞溶解。针对所测试的不同CD3殖株,观察到以剂量依赖的方式的标靶细胞溶解。MF8048观察到低标靶细胞溶解。CD4及CD8 T细胞上的活化标识CD69及CD25表现电平测量以FACS染色用于评估T细胞活化。所有殖株都观察到剂量依赖的T细胞活化,及阴性对照没有观察到T细胞活化。最后,48hrs后使用Assays(eBiosciencesTM)、遵照标准制造商的指示来判定自以HCT-116细胞进行的EGFRxCD3细胞毒性分析衍生之上清液内的IFN-γ及TNF-α细胞介素生产(图14F-G)。Additional clones from
针对属于超级群4的附加的殖株的特征化,以FACS来判定与HPB-ALL细胞结合亲和力(图15)。PG1337,一种有对破伤风类毒素特异性的二个完全相同的MF1337臂的单价抗体,使用作阴性对照。关于细胞毒性分析,使用HCT-116细胞及BxPC3细胞作为标靶细胞以测试MF8998的活性以及使用BxPC3标靶细胞以测试MF10401及MF10428的活性。含括具已知高活性的CD3xTAA双特异性抗体在内作为阳性对照。标靶细胞溶解使用细胞生存力测量来定量。使用来自细胞毒性分析之上清液、用分析来测量IL-6、IFN-γ及TNF-α的细胞介素电平。For characterization of additional clones belonging to
因而发现所测试的三种超级群4的殖株展现出不同的结合但是展现出相似的溶解活性。纵然这些殖株的溶解活性是相似的,但观察到降低的细胞介素生产。The three strains of
表1Table 1
表1:图15A所提及已指明的CD3殖株的结合的AUC值。Table 1 : AUC values for binding of the indicated CD3 clones referred to in Figure 15A.
表2Table 2
表2:图15A所提及已指明的CD3殖株的标靶细胞溶解的AUC及EC50值。Table 2: AUC and EC50 values for target cell lysis of the indicated CD3 clones mentioned in Figure 15A.
表3table 3
表3:图15A所提及已指明的细胞介素的量及CD3殖株的AUC及EC50值。Table 3: AUC and EC50 values of the indicated interferon amounts and CD3 clones mentioned in Figure 15A.
如由细胞毒性分析所评估的,观察到所有另外鉴定出的MFs为功能性的。接下来,绘制溶解于Y轴上及结合亲和力于X轴上之间的图(图16)以了解于超级群内及跨越超级群的溶解及亲和力之间的关系。总的来说,产生了多样抗CD3 Fabs群组其由多样的超级群所构成且涵盖一系列的亲和力。有趣地,超级群1和4导定出展现出相似的活性但是不同的CD3结合的殖株(图16B)。比较超级群1及3揭露了展现出相似的CD3结合及差异的活性的殖株(图16C)。All additionally identified MFs were observed to be functional as assessed by cytotoxicity assays. Next, a graph was plotted between dissolution on the Y-axis and binding affinity on the X-axis (FIG. 16) to understand the relationship between dissolution and affinity within and across supergroups. Overall, a diverse cohort of anti-CD3 Fabs was generated which consists of a diverse supergroup and covers a range of affinities. Interestingly,
表4Table 4
表4:图16B与16C所提及已指明的CD3殖株的标靶细胞溶解的AUC及EC50值。Table 4: AUC and EC50 values for target cell lysis of the indicated CD3 clones mentioned in Figures 16B and 16C.
CD3抗原特征化CD3 antigen characterization
如以上所述,发现分别来自超级群1及超级群4的二个殖株,即MF8058及MF8998,在与作为结合作为肿瘤细胞抗原的EGFR的Fab臂的殖株MF8233一起呈双特异性格式的情况下有相似的溶解活性(图17)。针对此实验,对HCT-116细胞的溶解活性如上文所述被测量。MF9257xMF8233使用作阳性对照以及MF9257xMF1337使用作阴性对照。从图17及表5中可以看出,多种测试的双特异性抗体观察到剂量依赖及高的杀灭百分率。As described above, two clones from
表5table 5
表5:图17所提及已指明的双特异性抗体于标靶细胞溶解的AUC及EC50值。阴性对照显示无显着可测量的活性。Table 5: AUC and EC50 values of the indicated bispecific antibodies mentioned in Figure 17 in target cell lysis. Negative controls showed no significant measurable activity.
结合亲和力binding affinity
如于“CD3双特异性抗体特征化”节中所述,附加的CD3殖株以FACS来分析对于表现人类CD3的HPB-ALL细胞的结合亲和力。MF6955及MF6964对于CD3的亲和力通过表面电浆子共振(SPR)技术、使用BIAcoreTMT100来测量。抗人类IgG小鼠单株抗体(Becton andDickinson,cat.Nr.555784)使用游离胺化学(NHS/EDC)偶合至CM5传感器芯片表面。继而捕捉CD3xTAA双特异性抗体于此传感器表面上。一浓度范围的重组纯化的抗原人类CD3δε-Fc蛋白随后于传感器表面移动来测量缔合及解离速率。在每周期的后,通过HCl脉冲使传感器表面再生且再次捕捉CD3xTAA双特异性抗体。从得到的感应图,使用BIAevaluation软件来判定缔合及解离速率。图18说明所产生的CD3组的结合亲和力范围。Additional CD3 clones were analyzed by FACS for binding affinity to HPB-ALL cells expressing human CD3 as described in the "Characterization of CD3 Bispecific Antibodies" section. The affinity of MF6955 and MF6964 for CD3 was measured by surface plasmon resonance (SPR) technique using BIAcore ™ T100. Anti-human IgG mouse monoclonal antibody (Becton and Dickinson, cat. Nr. 555784) was coupled to the CM5 sensor chip surface using free amine chemistry (NHS/EDC). The CD3xTAA bispecific antibody is then captured on the sensor surface. A range of concentrations of recombinantly purified antigenic human CD3δε-Fc protein was then moved across the sensor surface to measure association and dissociation rates. After each cycle, the sensor surface was regenerated by HCl pulses and the CD3xTAA bispecific antibody was captured again. From the resulting sensorgrams, association and dissociation rates were determined using BIAevaluation software. Figure 18 illustrates the range of binding affinity for the CD3 panel generated.
Claims (33)
Applications Claiming Priority (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP19166345.9 | 2019-03-29 | ||
| EP16063459 | 2019-03-29 | ||
| EP19166345 | 2019-03-29 | ||
| EP20009314 | 2019-10-01 | ||
| EP19200931 | 2019-10-01 | ||
| EP19200931.4 | 2019-10-01 | ||
| CN202080019367.1A CN113544154A (en) | 2019-03-29 | 2020-03-27 | CD3 binding molecules |
| PCT/NL2020/050214 WO2020204708A1 (en) | 2019-03-29 | 2020-03-27 | Cd3 binding molecules |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202080019367.1A Division CN113544154A (en) | 2019-03-29 | 2020-03-27 | CD3 binding molecules |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN114605546A true CN114605546A (en) | 2022-06-10 |
Family
ID=81864994
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202210274738.1A Pending CN114605546A (en) | 2019-03-29 | 2020-03-27 | CD3 binding molecules |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN114605546A (en) |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005118635A2 (en) * | 2004-06-03 | 2005-12-15 | Novimmune S.A. | Anti-cd3 antibodies and methods of use thereof |
| EP2604625A1 (en) * | 2011-09-26 | 2013-06-19 | Merus B.V. | Generation of binding molecules |
| CN108026174A (en) * | 2015-07-10 | 2018-05-11 | 美勒斯公司 | People's CD3 binding antibodies |
| CN111936514A (en) * | 2018-03-30 | 2020-11-13 | 美勒斯公司 | multivalent antibody |
-
2020
- 2020-03-27 CN CN202210274738.1A patent/CN114605546A/en active Pending
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005118635A2 (en) * | 2004-06-03 | 2005-12-15 | Novimmune S.A. | Anti-cd3 antibodies and methods of use thereof |
| EP2604625A1 (en) * | 2011-09-26 | 2013-06-19 | Merus B.V. | Generation of binding molecules |
| CN108026174A (en) * | 2015-07-10 | 2018-05-11 | 美勒斯公司 | People's CD3 binding antibodies |
| CN111936514A (en) * | 2018-03-30 | 2020-11-13 | 美勒斯公司 | multivalent antibody |
Non-Patent Citations (2)
| Title |
|---|
| CHANTAL KUHN等: "herapeutic anti-CD3 monoclonal antibodies: from bench to bedside", IMMUNOTHERAPY, vol. 8, no. 8, 1 July 2016 (2016-07-01), pages 889 - 906, XP055581845, DOI: 10.2217/imt-2016-0049 * |
| 杨治华等: "抗人CD3人/鼠嵌合抗体基因的构建", 中国肿瘤生物治疗杂志, no. 2, 30 June 1995 (1995-06-30), pages 108 - 111 * |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20230406932A1 (en) | Antibodies that modulate a biological activity expressed by a cell | |
| TWI840433B (en) | Novel rationally designed protein compositions | |
| CN109096396B (en) | anti-PD-L1 humanized nano antibody and application thereof | |
| EP3126383B1 (en) | Modified j-chain | |
| JP7783860B2 (en) | CD3 binding molecule | |
| US20230357407A1 (en) | Bispecific anti pd1-anti tim3 antibodies | |
| JP2018522564A (en) | Anti-glypican 3 antibody and use thereof | |
| US20240124563A1 (en) | Anti-Human MSLN Antibody And Application Thereof | |
| AU2021218069B2 (en) | Anti-HER2 affibody and switchable chimeric antigen receptor using same as switch molecule | |
| KR20230166120A (en) | Novel TNFR2-binding molecule | |
| TW202340240A (en) | Multi-specific antibody and its pharmaceutical uses | |
| WO2023066336A1 (en) | Anti-muc17 nanobody and use thereof | |
| CN110291107B (en) | Monoclonal antibody targeting a unique sialoglycosylated cancer-associated epitope of CD43 | |
| KR20250173509A (en) | Antibody specifically binding to claudin 18.2, method for producing the same, and application thereof | |
| JP2025540657A (en) | anti-CD3 antibody | |
| CN113544154A (en) | CD3 binding molecules | |
| US20230265185A1 (en) | Anti-cd22 single domain antibodies and therapeutic constructs | |
| CN114605546A (en) | CD3 binding molecules | |
| WO2026022767A1 (en) | Antibody targeting b7-h3, and chimeric antigen receptor comprising same | |
| KR20250143344A (en) | Anti-CDH6 antibodies and uses thereof | |
| HK1254029A1 (en) | Human cd3 binding antibody | |
| HK1233654B (en) | Modified j-chain | |
| HK1233654A1 (en) | Modified j-chain |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination |