CN114517176A - Kit for inducing IPS (IPS) cells into NK (Natural killer) cells and application method thereof - Google Patents
Kit for inducing IPS (IPS) cells into NK (Natural killer) cells and application method thereof Download PDFInfo
- Publication number
- CN114517176A CN114517176A CN202210100448.5A CN202210100448A CN114517176A CN 114517176 A CN114517176 A CN 114517176A CN 202210100448 A CN202210100448 A CN 202210100448A CN 114517176 A CN114517176 A CN 114517176A
- Authority
- CN
- China
- Prior art keywords
- cells
- culture solution
- ips cells
- culture
- ips
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 230000001939 inductive effect Effects 0.000 title claims abstract description 30
- 238000000034 method Methods 0.000 title claims abstract description 13
- 210000004263 induced pluripotent stem cell Anatomy 0.000 claims abstract description 77
- 210000000822 natural killer cell Anatomy 0.000 claims abstract description 57
- 239000001963 growth medium Substances 0.000 claims abstract description 9
- 210000000130 stem cell Anatomy 0.000 claims abstract description 9
- 102000003812 Interleukin-15 Human genes 0.000 claims abstract description 7
- 102000000704 Interleukin-7 Human genes 0.000 claims abstract description 7
- 229940100994 interleukin-7 Drugs 0.000 claims abstract description 7
- 108090000172 Interleukin-15 Proteins 0.000 claims abstract description 6
- 108010002586 Interleukin-7 Proteins 0.000 claims abstract description 6
- 102000007350 Bone Morphogenetic Proteins Human genes 0.000 claims abstract description 5
- 108010007726 Bone Morphogenetic Proteins Proteins 0.000 claims abstract description 5
- 102000000646 Interleukin-3 Human genes 0.000 claims abstract description 5
- 108010002386 Interleukin-3 Proteins 0.000 claims abstract description 5
- 102000004278 Receptor Protein-Tyrosine Kinases Human genes 0.000 claims abstract description 5
- 108090000873 Receptor Protein-Tyrosine Kinases Proteins 0.000 claims abstract description 5
- 108010073929 Vascular Endothelial Growth Factor A Proteins 0.000 claims abstract description 5
- 102000005789 Vascular Endothelial Growth Factors Human genes 0.000 claims abstract description 5
- 108010019530 Vascular Endothelial Growth Factors Proteins 0.000 claims abstract description 5
- 229940112869 bone morphogenetic protein Drugs 0.000 claims abstract description 5
- 229940076264 interleukin-3 Drugs 0.000 claims abstract description 5
- 239000003446 ligand Substances 0.000 claims abstract description 5
- 210000004027 cell Anatomy 0.000 claims description 54
- 238000012258 culturing Methods 0.000 claims description 15
- 239000007640 basal medium Substances 0.000 claims description 14
- 239000002609 medium Substances 0.000 claims description 14
- 239000007788 liquid Substances 0.000 claims description 13
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 claims description 12
- 238000005406 washing Methods 0.000 claims description 9
- 108091003079 Bovine Serum Albumin Proteins 0.000 claims description 7
- HZAXFHJVJLSVMW-UHFFFAOYSA-N 2-Aminoethan-1-ol Chemical compound NCCO HZAXFHJVJLSVMW-UHFFFAOYSA-N 0.000 claims description 6
- 239000006144 Dulbecco’s modified Eagle's medium Substances 0.000 claims description 6
- 229960005070 ascorbic acid Drugs 0.000 claims description 6
- 229960005322 streptomycin Drugs 0.000 claims description 6
- 239000006228 supernatant Substances 0.000 claims description 6
- 108010010803 Gelatin Proteins 0.000 claims description 5
- 239000008273 gelatin Substances 0.000 claims description 5
- 229920000159 gelatin Polymers 0.000 claims description 5
- 235000019322 gelatine Nutrition 0.000 claims description 5
- 235000011852 gelatine desserts Nutrition 0.000 claims description 5
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 claims description 4
- 235000010323 ascorbic acid Nutrition 0.000 claims description 4
- 239000011668 ascorbic acid Substances 0.000 claims description 4
- 239000012091 fetal bovine serum Substances 0.000 claims description 4
- 239000000203 mixture Substances 0.000 claims description 4
- 210000002966 serum Anatomy 0.000 claims description 4
- OYHQOLUKZRVURQ-NTGFUMLPSA-N (9Z,12Z)-9,10,12,13-tetratritiooctadeca-9,12-dienoic acid Chemical compound C(CCCCCCC\C(=C(/C\C(=C(/CCCCC)\[3H])\[3H])\[3H])\[3H])(=O)O OYHQOLUKZRVURQ-NTGFUMLPSA-N 0.000 claims description 3
- UZOVYGYOLBIAJR-UHFFFAOYSA-N 4-isocyanato-4'-methyldiphenylmethane Chemical compound C1=CC(C)=CC=C1CC1=CC=C(N=C=O)C=C1 UZOVYGYOLBIAJR-UHFFFAOYSA-N 0.000 claims description 3
- 108091006905 Human Serum Albumin Proteins 0.000 claims description 3
- 102000008100 Human Serum Albumin Human genes 0.000 claims description 3
- 239000004372 Polyvinyl alcohol Substances 0.000 claims description 3
- DTOSIQBPPRVQHS-PDBXOOCHSA-N alpha-linolenic acid Chemical compound CC\C=C/C\C=C/C\C=C/CCCCCCCC(O)=O DTOSIQBPPRVQHS-PDBXOOCHSA-N 0.000 claims description 3
- 235000020661 alpha-linolenic acid Nutrition 0.000 claims description 3
- 229910000147 aluminium phosphate Inorganic materials 0.000 claims description 3
- 238000007664 blowing Methods 0.000 claims description 3
- 229940098773 bovine serum albumin Drugs 0.000 claims description 3
- 229960001231 choline Drugs 0.000 claims description 3
- OEYIOHPDSNJKLS-UHFFFAOYSA-N choline Chemical compound C[N+](C)(C)CCO OEYIOHPDSNJKLS-UHFFFAOYSA-N 0.000 claims description 3
- BVTBRVFYZUCAKH-UHFFFAOYSA-L disodium selenite Chemical compound [Na+].[Na+].[O-][Se]([O-])=O BVTBRVFYZUCAKH-UHFFFAOYSA-L 0.000 claims description 3
- 230000004927 fusion Effects 0.000 claims description 3
- 210000004408 hybridoma Anatomy 0.000 claims description 3
- 229960004488 linolenic acid Drugs 0.000 claims description 3
- KQQKGWQCNNTQJW-UHFFFAOYSA-N linolenic acid Natural products CC=CCCC=CCC=CCCCCCCCC(O)=O KQQKGWQCNNTQJW-UHFFFAOYSA-N 0.000 claims description 3
- 108010082117 matrigel Proteins 0.000 claims description 3
- PJUIMOJAAPLTRJ-UHFFFAOYSA-N monothioglycerol Chemical compound OCC(O)CS PJUIMOJAAPLTRJ-UHFFFAOYSA-N 0.000 claims description 3
- NBIIXXVUZAFLBC-UHFFFAOYSA-N phosphoric acid Substances OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 claims description 3
- 229920002451 polyvinyl alcohol Polymers 0.000 claims description 3
- 229960001471 sodium selenite Drugs 0.000 claims description 3
- 239000011781 sodium selenite Substances 0.000 claims description 3
- 235000015921 sodium selenite Nutrition 0.000 claims description 3
- DGVVWUTYPXICAM-UHFFFAOYSA-N β‐Mercaptoethanol Chemical compound OCCS DGVVWUTYPXICAM-UHFFFAOYSA-N 0.000 claims description 3
- 239000002211 L-ascorbic acid Substances 0.000 claims description 2
- 235000000069 L-ascorbic acid Nutrition 0.000 claims description 2
- 238000005119 centrifugation Methods 0.000 claims description 2
- 230000029087 digestion Effects 0.000 claims description 2
- 239000000499 gel Substances 0.000 claims description 2
- 238000010899 nucleation Methods 0.000 claims description 2
- 239000012531 culture fluid Substances 0.000 claims 1
- 230000004069 differentiation Effects 0.000 abstract description 23
- 230000035755 proliferation Effects 0.000 abstract description 7
- 235000015097 nutrients Nutrition 0.000 abstract description 3
- 230000006907 apoptotic process Effects 0.000 abstract description 2
- 210000003716 mesoderm Anatomy 0.000 abstract description 2
- 239000000243 solution Substances 0.000 description 64
- 230000000052 comparative effect Effects 0.000 description 7
- 210000002242 embryoid body Anatomy 0.000 description 6
- 230000006698 induction Effects 0.000 description 6
- 239000011148 porous material Substances 0.000 description 4
- 101000581981 Homo sapiens Neural cell adhesion molecule 1 Proteins 0.000 description 3
- 102100027347 Neural cell adhesion molecule 1 Human genes 0.000 description 3
- 238000001514 detection method Methods 0.000 description 3
- 238000011161 development Methods 0.000 description 3
- 230000018109 developmental process Effects 0.000 description 3
- 238000011160 research Methods 0.000 description 3
- 238000012360 testing method Methods 0.000 description 3
- ZDXPYRJPNDTMRX-VKHMYHEASA-N L-glutamine Chemical compound OC(=O)[C@@H](N)CCC(N)=O ZDXPYRJPNDTMRX-VKHMYHEASA-N 0.000 description 2
- 229930182816 L-glutamine Natural products 0.000 description 2
- 241001529936 Murinae Species 0.000 description 2
- 208000007660 Residual Neoplasm Diseases 0.000 description 2
- 230000024245 cell differentiation Effects 0.000 description 2
- 238000000684 flow cytometry Methods 0.000 description 2
- 238000000338 in vitro Methods 0.000 description 2
- 239000002504 physiological saline solution Substances 0.000 description 2
- 230000002195 synergetic effect Effects 0.000 description 2
- 210000004881 tumor cell Anatomy 0.000 description 2
- 241000894006 Bacteria Species 0.000 description 1
- 102000017420 CD3 protein, epsilon/gamma/delta subunit Human genes 0.000 description 1
- 102000004127 Cytokines Human genes 0.000 description 1
- 108090000695 Cytokines Proteins 0.000 description 1
- 108010017535 Interleukin-15 Receptors Proteins 0.000 description 1
- 102000000743 Interleukin-5 Human genes 0.000 description 1
- 108010002616 Interleukin-5 Proteins 0.000 description 1
- 108010038498 Interleukin-7 Receptors Proteins 0.000 description 1
- 206010028980 Neoplasm Diseases 0.000 description 1
- 102000016971 Proto-Oncogene Proteins c-kit Human genes 0.000 description 1
- 108010014608 Proto-Oncogene Proteins c-kit Proteins 0.000 description 1
- 206010070834 Sensitisation Diseases 0.000 description 1
- 241000700605 Viruses Species 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 238000004220 aggregation Methods 0.000 description 1
- 230000003321 amplification Effects 0.000 description 1
- 239000000427 antigen Substances 0.000 description 1
- 102000036639 antigens Human genes 0.000 description 1
- 108091007433 antigens Proteins 0.000 description 1
- 238000010923 batch production Methods 0.000 description 1
- 238000004113 cell culture Methods 0.000 description 1
- 230000004663 cell proliferation Effects 0.000 description 1
- 230000001413 cellular effect Effects 0.000 description 1
- 238000002512 chemotherapy Methods 0.000 description 1
- 238000003501 co-culture Methods 0.000 description 1
- 238000012136 culture method Methods 0.000 description 1
- 231100000433 cytotoxic Toxicity 0.000 description 1
- 230000001472 cytotoxic effect Effects 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 230000007123 defense Effects 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- MHMNJMPURVTYEJ-UHFFFAOYSA-N fluorescein-5-isothiocyanate Chemical compound O1C(=O)C2=CC(N=C=S)=CC=C2C21C1=CC=C(O)C=C1OC1=CC(O)=CC=C21 MHMNJMPURVTYEJ-UHFFFAOYSA-N 0.000 description 1
- 210000001654 germ layer Anatomy 0.000 description 1
- 239000003292 glue Substances 0.000 description 1
- 210000003958 hematopoietic stem cell Anatomy 0.000 description 1
- 238000011134 hematopoietic stem cell transplantation Methods 0.000 description 1
- 230000007124 immune defense Effects 0.000 description 1
- 230000003832 immune regulation Effects 0.000 description 1
- 210000000987 immune system Anatomy 0.000 description 1
- 238000009169 immunotherapy Methods 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 238000011081 inoculation Methods 0.000 description 1
- 210000002660 insulin-secreting cell Anatomy 0.000 description 1
- 230000003902 lesion Effects 0.000 description 1
- 210000004698 lymphocyte Anatomy 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 210000003061 neural cell Anatomy 0.000 description 1
- 238000003199 nucleic acid amplification method Methods 0.000 description 1
- 235000016709 nutrition Nutrition 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 244000052769 pathogen Species 0.000 description 1
- 230000001717 pathogenic effect Effects 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- 230000035802 rapid maturation Effects 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 238000012827 research and development Methods 0.000 description 1
- 230000008313 sensitization Effects 0.000 description 1
- 210000001082 somatic cell Anatomy 0.000 description 1
- 230000001225 therapeutic effect Effects 0.000 description 1
- 238000002054 transplantation Methods 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0634—Cells from the blood or the immune system
- C12N5/0646—Natural killers cells [NK], NKT cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/05—Inorganic components
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/05—Inorganic components
- C12N2500/10—Metals; Metal chelators
- C12N2500/20—Transition metals
- C12N2500/24—Iron; Fe chelators; Transferrin
- C12N2500/25—Insulin-transferrin; Insulin-transferrin-selenium
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/30—Organic components
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/30—Organic components
- C12N2500/35—Polyols, e.g. glycerin, inositol
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/30—Organic components
- C12N2500/44—Thiols, e.g. mercaptoethanol
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/30—Organic components
- C12N2500/46—Amines, e.g. putrescine
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/50—Soluble polymers, e.g. polyethyleneglycol [PEG]
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/70—Undefined extracts
- C12N2500/80—Undefined extracts from animals
- C12N2500/84—Undefined extracts from animals from mammals
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/125—Stem cell factor [SCF], c-kit ligand [KL]
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/155—Bone morphogenic proteins [BMP]; Osteogenins; Osteogenic factor; Bone inducing factor
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/165—Vascular endothelial growth factor [VEGF]
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/20—Cytokines; Chemokines
- C12N2501/23—Interleukins [IL]
- C12N2501/2303—Interleukin-3 (IL-3)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/20—Cytokines; Chemokines
- C12N2501/23—Interleukins [IL]
- C12N2501/2307—Interleukin-7 (IL-7)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/20—Cytokines; Chemokines
- C12N2501/23—Interleukins [IL]
- C12N2501/2315—Interleukin-15 (IL-15)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/20—Cytokines; Chemokines
- C12N2501/26—Flt-3 ligand (CD135L, flk-2 ligand)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/80—Neurotransmitters; Neurohormones
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/998—Proteins not provided for elsewhere
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2506/00—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells
- C12N2506/45—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells from artificially induced pluripotent stem cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2509/00—Methods for the dissociation of cells, e.g. specific use of enzymes
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2509/00—Methods for the dissociation of cells, e.g. specific use of enzymes
- C12N2509/10—Mechanical dissociation
Landscapes
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Biomedical Technology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Wood Science & Technology (AREA)
- Biotechnology (AREA)
- Organic Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Genetics & Genomics (AREA)
- Zoology (AREA)
- Hematology (AREA)
- Microbiology (AREA)
- Immunology (AREA)
- Biochemistry (AREA)
- General Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Cell Biology (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
Abstract
The invention provides a kit for inducing IPS cells into NK cells, which comprises a first culture solution, a second culture solution and a third culture solution; the stem cell factor, the human bone morphogenetic protein and the vascular endothelial growth factor contained in the first culture solution can effectively induce the differentiation of mesoderm, and Y-27632 can obviously reduce the apoptosis of IPS cells. The components and other nutrient components in the culture medium are used in the process of forming the Spin EB by the IPS cells, and can promote the proliferation and differentiation of the Spin EB. The stem cell factor, interleukin 7, interleukin 15 and a ligand of a tyrosine kinase receptor 3 in the second culture solution and the third culture solution, and the interleukin 3 contained in the third culture solution can promote the early proliferation and differentiation of NK cells, and the components synergistically act on each stage of differentiation of Spin EB into NK cells, so that high-quantity and high-purity NK cells can be obtained.
Description
Technical Field
The invention belongs to the technical field of biology, and particularly relates to a kit for inducing IPS cells into NK cells and an application method thereof.
Background
Induced pluripotent stem cells (IPS cells) can be differentiated in vitro into Embryoid Bodies (EBs) containing various germ layers under appropriate conditions, and further differentiated into target cell types, such as insulin-secreting cells, hematopoietic cells, neural cells, and the like. Since the IPS cell is directly reprogrammed by the somatic cell of the patient, immune rejection does not exist during cell transplantation, and the ethical problem of cell sources does not need to be considered, so that the IPS cell has wide clinical application prospect.
Natural killer cells, as a class of independent lymphocyte populations, can directly kill target cells, including tumor cells, virus or bacteria infected cells, and certain senescent normal cells of the body without antigen pre-sensitization, and are thus considered the first line of defense in the body's immune defense system. With the development of tumor immunology, more and more researches show that the application of natural killer cells (NK cells) to adoptive immunotherapy has obvious effects on eliminating residual tumor lesions and promoting the reconstruction of the immune system of a patient, and becomes an important therapeutic means for eliminating residual tumor cells after chemotherapy and hematopoietic stem cell transplantation. Clinical NK cells have the condition of insufficient supply, and currently, researches on inducing differentiation of NK cells include cell co-culture technology or methods of adding culture media and the like, but the methods have the defects of small cell number, low differentiation efficiency and susceptibility to pathogen pollution when the cells are successfully induced. Therefore, there is a need for further research and development of an optimal differentiation-inducing system capable of inducing IPS cells into NK cells.
Disclosure of Invention
In order to solve the above-mentioned problems, the present invention aims to provide a kit for inducing IPS cells into NK cells and use thereof, so that a culture induction system with high efficiency and stability and a simple and easy-to-operate culture method can be developed to obtain high-quality and high-purity NK cells.
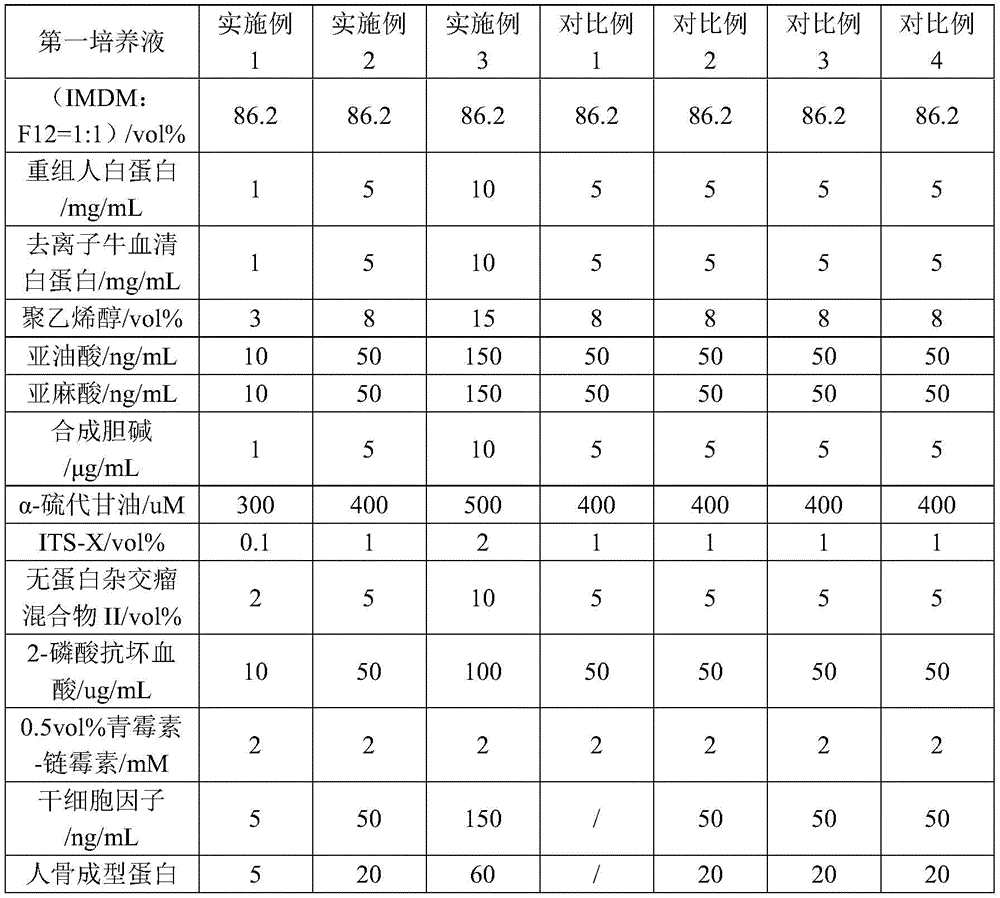
According to a first aspect of the present invention, there is provided a kit for inducing IPS cells into NK cells, comprising the following components: a first culture solution, a second culture solution and a third culture solution;
the first culture solution includes: 1 part of cell basal medium, 1-10 mg/mL of recombinant human albumin, 1-10 mg/mL of deionized bovine serum albumin, 3-15 vol% of polyvinyl alcohol, 10-150 ng/mL of linoleic acid, 10-150 ng/mL of linolenic acid, 1-10 mug/mL of synthetic choline, 300-500 uM alpha-thioglycerol, 0.1-2 vol% of insulin-transferrin-selenium-ethanolamine solution, 2-10 vol% of protein-free hybridoma mixture II, 10-100 ug/mL of 2-phosphoric acid ascorbic acid, 5-150 ng/m of dry cell factor, 5-60 ng/mL of human bone morphogenetic protein, 5-60 ng/mL of vascular endothelial growth factor and 5-60 uMY-27632;
the second culture solution comprises: 2 parts of cell basal culture, 10-20 vol% of heat-inactivated human AB serum, 1-10 mM L-glutamine, 0.1-2 mu M beta-mercaptoethanol, 2-8 ng/mL of sodium selenite, 10-50 mu M of ethanolamine, 10-40 mg/L of ascorbic acid, 5-150 ng/mL of stem cell factor, 5-100 ng/mL of interleukin 7, 5-100 ng/mL of interleukin 15 and 1-50 ng/mL of tyrosine kinase receptor 3 ligand.
The third culture solution comprises: a second culture solution and 2-50 ng/mL interleukin 3.
The invention provides a kit for inducing IPS cells into NK cells and an application method thereof, wherein the first culture solution can culture the IPS cells to form single-cell aggregated embryoid bodies (Spin EBs), and the second culture solution and the third culture solution can induce the Spin EBs to be differentiated into the NK cells. The first culture solution does not contain serum or feeder cells, the content of the components of the culture medium is definite and controllable, exogenous pollution is not easy to cause, batch production is facilitated, the repeatability of experimental results is high, and the IPS cells can efficiently form Spin EBs. Through experimental research, the stem cell factor, the human bone morphogenetic protein and the vascular endothelial growth factor contained in the first culture solution can effectively induce the differentiation of mesoderm, and Y-27632 can obviously reduce the apoptosis of IPS cells and promote the proliferation of the IPS cells. The synergistic effect of the components can promote intercellular signal path conduction and other nutrient components in the culture solution, is used for forming the Spin EB by the IPS cells and contributes to the early proliferation and differentiation of the Spin EB into NK cells.
We found that the stem cell factor, interleukin 7, interleukin 15 and tyrosine kinase receptor 3 ligand (FLT-3L) in the second and third culture media act in combination on various stages of differentiation of Spin EB into NK cells. Stem cell factor and FLT-3L can promote differentiation of Spin EB, promote early development of NK cells and differentiate NK progenitor cells. The combined action of stem cell factor, interleukin 7, interleukin 15 and FLT-3L can promote the proliferation of Spin EB and further increase the number of NK cells. The interleukin 7 and the interleukin 15 can activate the NK cells, promote the rapid maturation differentiation, development and proliferation of the NK cells, improve the cytotoxic effect of the NK cells and promote the NK cells to secrete cytokines, thereby playing roles in immune regulation and killing. The interleukin 3 contained in the third culture solution can promote the proliferation and differentiation of NK cells in an early stage. The third culture solution and the second culture solution are applied to different stages of the differentiation of the Spin EB, the components have synergistic effect, and other nutrient components in the culture solution act on each stage of the differentiation from the Spin EB to the NK cells to the maturity, so that the differentiation efficiency is effectively improved, and the successful differentiation proportion of the NK cells is improved.
Preferably, the first culture solution comprises: 86.2 vol% of cell basal medium 1, 1-10 mg/mL of recombinant human albumin, 2-8 mg/mL of deionized bovine serum albumin, 5-10 vol% of polyvinyl alcohol, 10-100 ng/mL of linoleic acid, 10-100 ng/mL of linolenic acid, 2-8 mug/mL of synthetic choline, 350-450 uM alpha-thioglycerol, 0.1-1 vol% of insulin-transferrin-selenium-ethanolamine solution, 2-8 vol% of protein-free hybridoma mixture II, 10-100 ug/mL of 2-phosphoric acid ascorbic acid, 30-50 ng/m of dry cell factor, 10-30 ng/mL of human bone morphogenetic protein, 10-30 ng/mL of vascular endothelial growth factor and 5-15 uMY-27632;
the second culture solution comprises: 75-83 vol% cell basal culture 2, 10-20 vol% heat-inactivated human AB serum, 2mM L-glutamine, 0.8-1.2 mu M beta-mercaptoethanol, 2-8 ng/mL sodium selenite, 50 mu M ethanolamine, 10-40 mg/L ascorbic acid, 10-30 ng/mL stem cell factor, 10-30 ng/mL interleukin 7, 5-20 ng/mL interleukin 15 and 5-20 ng/mL tyrosine kinase receptor 3 ligand;
the third culture solution comprises: a second culture solution and 2-8 ng/mL interleukin 3.
By further limiting the content of each component in each culture medium, the kit provided by the invention has higher efficiency of inducing IPS cells into NK cells.
Preferably, the cell basal medium 1 consists of: IMDM medium: f12 medium 1:1, the first broth further comprising 2mm0.5 vol% penicillin-streptomycin.
Preferably, the cell basal medium 2 consists of: DMEM medium: and F12 culture medium is 5-5.5: 2.5-2.8, and the third culture solution further comprises 1 vol% of penicillin-streptomycin.
Preferably, the kit for inducing IPS cells into NK cells further comprises a fourth culture medium; the fourth culture solution was used during the centrifugation of IPS cells; the fourth culture solution comprises: 90 vol% cell basal medium 3 and 10 vol% fetal bovine serum.
Preferably, the cell basal medium 3 consists of the following components, calculated by volume ratio: DMEM medium: f12 medium 1: 1.
According to another aspect of the present invention, there is provided a method of inducing IPS cells into NK cells, comprising:
s1: culturing the IPS cells by adopting the first culture solution to form Spin EB;
s2: and inducing Spin EB into NK cells by using the second culture solution and the third culture solution.
Preferably, in S1, the specific steps are:
s1.1: inoculating the IPS cells into a 6-well plate paved with a Matrigel gel solution;
s1.2: when the IPS cells are cultured to grow to 70% -80% of fusion degree, washing the IPS cells with physiological saline for 2 times, and discarding washing liquid;
s1.3: adding preheated tryplE Select into an incubator for digestion for 3-10 min, blowing IPS cells into single cells, and transferring the single cells into a 15mL centrifuge tube;
s1.4: adding 5mL of fourth culture solution into the 15mL centrifuge tube, uniformly mixing, centrifuging, removing supernatant, and washing the IPS cells once with normal saline;
s1.5: resuspending the IPS cells by using a first culture solution, and adjusting the density of the IPS cells to obtain an IPS cell solution;
s1.6: and (3) inoculating the IPS cell liquid to a 96-hole round-bottom pore plate according to 100 mu L/hole, centrifuging, and transferring the 96-hole round-bottom pore plate to an incubator for culturing for 6 days to obtain the Spin EB liquid.
Preferably, the seeding density of the IPS cells in S1.1 is 10-50 ten thousand per hole; in S1.5, the density of IPS cells is adjusted to 5-20 ten thousand/mL.
In the step of culturing the IPS cells to form Spin EB, the IPS cells are digested into single cells by TryplE Select and then are further centrifuged to obtain a single cell centrifugal aggregation formed embryoid-like body (Spin EB). The number of IPS cells forming Spin EB affects the efficiency of further differentiation of EB cells, and when the number of cells is too small, the total number of NK cells successfully differentiated is low, and when the number of cells is too large, the efficiency of differentiation of EB cells is lowered.
Preferably, in S2, the specific steps are:
s2.1: adding 1-3% gelatin solution into a 6-hole plate according to 1 mL/hole, standing for 1h in an incubator to obtain a 6-hole plate paved with gelatin, and adding a third culture solution into the 6-hole plate;
s2.2: spin EB liquid was discarded most of the supernatant, which was added to the above 6-well plate
S2.3: after culturing for 6 days, changing into a second culture solution, and continuously culturing for 5-7 days;
s2.4: and continuously culturing for 3-4 weeks.
Preferably, in S2.4, the second culture solution is changed every 3 days from the 14 th day of culture.
The kit for inducing the IPS cells into the NK cells provided by the invention not only provides suitable nutritional conditions suitable for IPS cell differentiation and NK cell proliferation, but also can be added with various effective components capable of acting on various stages of IPS cell differentiation. By applying each culture solution in the kit to each step of induction and differentiation of IPS cell culture, NK cells with high differentiation efficiency, high purity and large quantity can be obtained.
Drawings
FIG. 1 is a microscope photograph of NK cells obtained by Spin EB-induced differentiation for 21 days in example 2.
Detailed Description
In order to make the technical solutions of the present invention better understood by those skilled in the art, the technical solutions in the embodiments of the present invention will be clearly and completely described below, and it is obvious that the described embodiments are only a part of the embodiments of the present invention, and not all embodiments. Unless otherwise specified, the technical means used in the examples are conventional means well known to those skilled in the art, and the raw materials used are commercially available products.
Table 1 shows the content of the final components of the first culture solution, the second culture solution and the third culture solution in the kits provided in examples 1 to 3 and comparative examples 1 to 4. In addition, the kit provided by the embodiments 1-3 and the comparative examples 1-4 further comprises a fourth culture solution, and the kit comprises the following specific components: 90 vol% (DMEM medium: F12 medium 1:1) and 10 vol% fetal bovine serum.
TABLE 1 ingredient tables of first, second, and third culture solutions in examples 1 to 3 and comparative examples 1 to 4
Comparative example 5
The kit provided in the comparative example comprises a first culture solution, a second culture solution, a third culture solution and a fourth culture solution. The first culture solution comprises the following components: cell basal medium (IMDM: F12 ═ 1:1), 2mm 0.5% penicillin-streptomycin; the second culture solution and the third culture solution comprise the following components: 99 vol% cell basal medium (DEME: F12 ═ 2:1), 1 vol% penicillin-streptomycin; the fourth culture solution had a composition of 90 vol% (DMEM medium: F12 medium: 1) and 10 vol% fetal bovine serum.
The specific experimental operations for inducing IPS cells into NK cells using the kits provided in examples 1-3 and comparative examples 1-5 were as follows:
s1: culturing the IPS cells by adopting a first culture solution in the kit to form Spin EB;
s1.1: inoculating the IPS cells into a 6-well plate paved with a Matrigel glue solution according to the inoculation density of 50 ten thousand per well;
s1.2: when the IPS cells are cultured to grow to 80% fusion degree, washing the IPS cells with physiological saline for 2 times, and discarding washing liquid;
s1.3: adding preheated tryplE Select, digesting in an incubator for 6min, blowing IPS cells into single cells, and transferring the single cells into a 15mL centrifuge tube;
s1.4: adding 5mL of fourth culture solution into the 15mL centrifuge tube, uniformly mixing, centrifuging, removing supernatant, and washing the IPS cells once with normal saline;
s1.5: resuspending the IPS cells by using a first culture solution, and adjusting the density of the IPS cells to be 20 ten thousand/mL to obtain an IPS cell solution;
s1.6: and (3) inoculating the IPS cell liquid to a 96-hole round-bottom pore plate according to 100 mu L/hole, centrifuging, and transferring the 96-hole round-bottom pore plate to an incubator for culturing for 6 days to obtain the Spin EB liquid.
S2: inducing Spin EB into NK cells S2.1 by using a second culture solution and a third culture solution in the kit: adding a 3% gelatin solution into a 6-hole plate according to 1 mL/hole, standing in an incubator for 1h to obtain a 6-hole plate paved with gelatin, and adding a third culture solution into the 6-hole plate;
s2.2: removing most of supernatant of Spin EB liquid, and adding the Spin EB liquid into the 6-hole plate;
s2.3: changing into a second culture solution after culturing for 6 days, and continuously culturing for 5 days;
s2.4: the culture was continued for 3 weeks, and the second culture solution was changed every 3 days from the 14 th day of culture.
Test example
1. The subjects to be tested in this test example were NK cells in examples 1 to 3 and comparative examples 1 to 5.
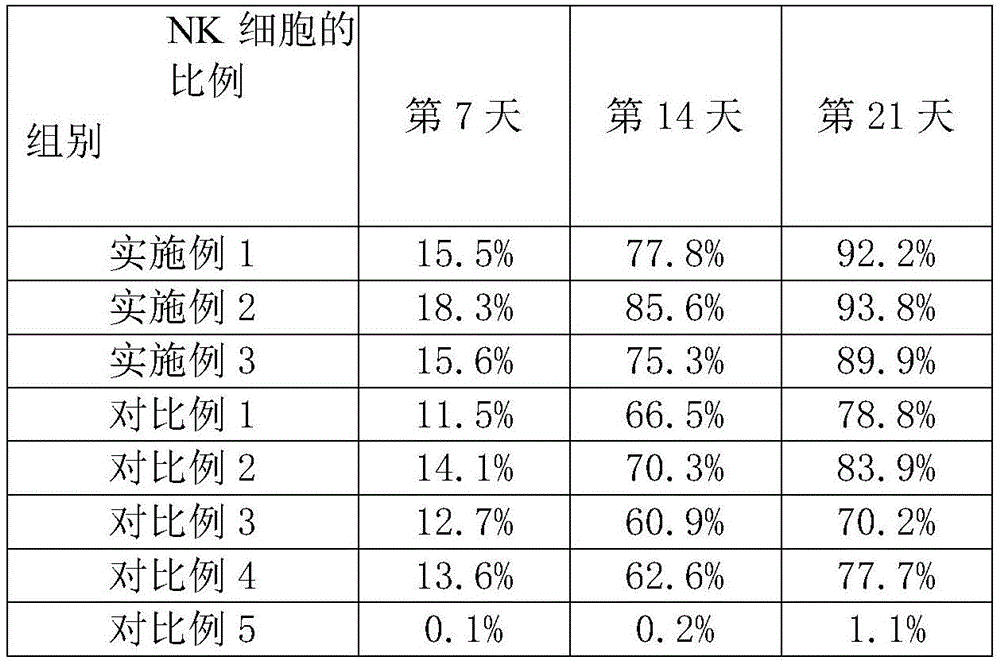
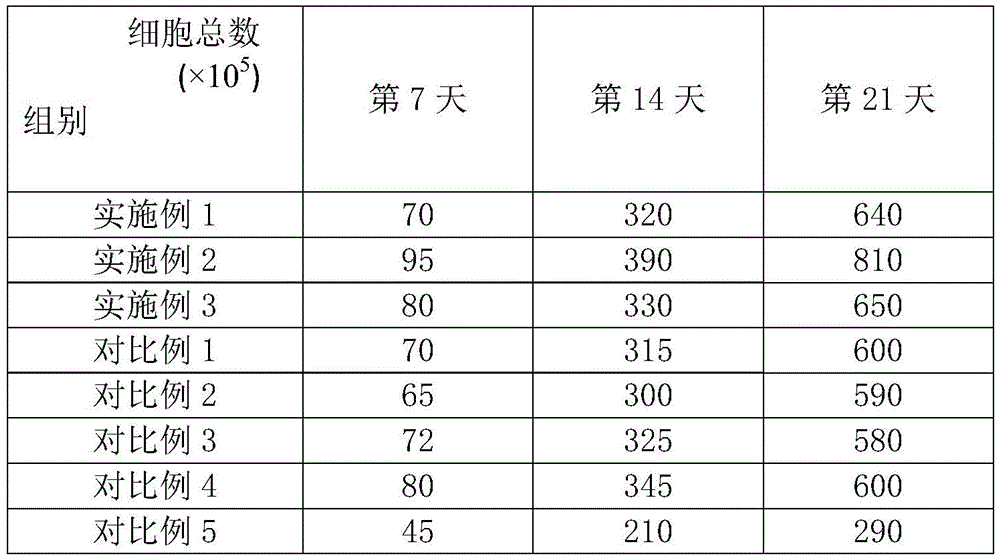
CD3-Cell, CD56+The cells are the cellular immunophenotype mainly detected by the in vitro amplification culture of NK cells. The test example adopts a flow cytometry detection method: 5X 10 cell samples obtained at 7, 14 and 21 days after the induction culture of inducing Spin EB into NK cells in S2 were collected5Cells/tube, PBS wash 2 times. Adding flow detection antibodies respectively for double-labeled flow phenotype detection: FITC-labeled murine anti-human CD3 antibody and APC-labeled murine anti-human CD56 antibody; incubate at room temperature for 20min, wash 2 times with PBS, and analyze the cells by flow cytometry. The experimental results are shown in table 2; and counting the total number of cells, and the results are shown in table 3.
2. The cells induced to differentiate and cultured up to day 21 in S2.4 of example 2 were collected, and the morphology of the cells was observed by an optical microscope, as shown in FIG. 1. As can be seen from FIG. 1, in example 2, NK cell colonies were formed 21 days after the induction culture, and the number of formed NK cells was large and the cells were piled.
TABLE 2 CD3-Cell, CD56+Proportion of cells in the course of Induction culture
TABLE 3 Total cell count during induction culture
The experimental results show that the kit for inducing the IPS cells into the NK cells can smoothly culture the IPS cells to form the Spin EB, further can efficiently induce and differentiate the IPS cells into the NK cells, and has high differentiation efficiency, high cell number and high cell purity when the IPS cells are induced into the NK cells.
Although the present invention has been described in detail with reference to the above embodiments, it should be understood by those skilled in the art that various changes, modifications and equivalents may be made therein without departing from the spirit and scope of the invention.
Claims (10)
1. A kit for inducing IPS cells into NK cells, comprising the following components: a first culture solution, a second culture solution and a third culture solution;
the first culture solution comprises: 1 part of cell basal medium, 1-10 mg/mL of recombinant human albumin, 1-10 mg/mL of deionized bovine serum albumin, 3-15 vol% of polyvinyl alcohol, 10-150 ng/mL of linoleic acid, 10-150 ng/mL of linolenic acid, 1-10 mug/mL of synthetic choline, 300-500 uM alpha-thioglycerol, 0.1-2 vol% of insulin-transferrin-selenium-ethanolamine solution, 2-10 vol% of protein-free hybridoma mixture II, 10-100 ug/mL of 2-phosphoric acid ascorbic acid, 5-150 ng/mL of dry cell factor, 5-60 ng/mL of human bone morphogenetic protein, 5-60 ng/mL of vascular endothelial growth factor and 5-60 uMY-27632;
the second culture solution comprises: 2 parts of cell basal medium, 10-20 vol% heat-inactivated human AB serum, 1-10 mML-glutamine, 0.1-2 MuM beta-mercaptoethanol, 2-8 ng/mL sodium selenite, 10-50 MuM ethanolamine, 10-40 mg/L ascorbic acid, 5-150 ng/mL stem cell factor, 5-100 ng/mL interleukin 7, 5-100 ng/mL interleukin 15 and 1-50 ng/mL tyrosine kinase receptor 3 ligand;
the third culture solution comprises: the second culture solution and 2-50 ng/mL interleukin 3.
2. The kit for inducing IPS cells into NK cells according to claim 1, wherein said cell basal medium 1 consists of, by volume: IMDM medium: f12 medium 1: 1; the first broth also included 2mm0.5 vol% penicillin-streptomycin.
3. The kit for inducing IPS cells into NK cells according to claim 1, wherein said cell basal medium 2 consists of, by volume: DMEM medium: and F12 culture medium is 5-5.5: 2.5 to 2.8; the second culture solution further comprises 1 vol% penicillin-streptomycin.
4. The kit for inducing IPS cells into NK cells of claim 1, further comprising a fourth culture fluid; the fourth culture solution comprises: cell basal medium 3 and 10 vol% fetal bovine serum.
5. The kit for inducing IPS cells into NK cells according to claim 4, wherein the cell basal medium 3 consists of the following components in volume ratio: DMEM medium: f12 medium 1: 1.
6. A method of inducing IPS cells into NK cells, comprising:
s1: culturing IPS cells to form Spin EBs using the first culture solution of claim 1;
s2: inducing said Spin EBs into NK cells using said second and third culture media of claim 1.
7. The method of inducing IPS cells as NK cells of claim 6, wherein in S1, the specific steps are:
s1.1: inoculating the IPS cells into a first culture vessel paved with a Matrigel gel solution;
s1.2: when the IPS cells are cultured to grow to 70% -80% of fusion degree, washing the IPS cells with normal saline, and discarding washing liquid;
s1.3: adding tryplE Select for digestion, blowing the IPS cells into single cells, and transferring the single cells into a centrifugal tube;
s1.4: adding a fourth culture solution into the centrifuge tube, uniformly mixing, centrifuging, removing supernatant, and washing the IPS cells with normal saline;
s1.5: resuspending the IPS cells by using the first culture solution, and adjusting the density of the IPS cells to obtain an IPS cell solution;
s1.6: and inoculating the IPS cell sap into a second culture vessel according to 100 mu L/hole, and culturing for 6 days after centrifugation to obtain the Spin EB liquid.
8. The method of inducing IPS cells as NK cells of claim 7, wherein the seeding density of the IPS cells in S1.1 is 10-50 ten thousand per well; in the S1.5, the density of the IPS cells is adjusted to be 5-20 ten thousand/mL.
9. The method of inducing IPS cells as NK cells of claim 8, wherein in S2, the specific steps are:
s2.1: adding the third culture solution into a third culture vessel paved with gelatin;
s2.2: removing supernatant of the Spin EB liquid, and adding the Spin EB liquid into the third culture vessel;
s2.3: after culturing for 6 days, changing the culture solution into the second culture solution, and continuously culturing for 5-7 days;
s2.4: and continuously culturing for 3-4 weeks.
10. The method of inducing IPS cells into NK cells of claim 9, wherein in S2.4, said second culture solution is changed every 3 days from the 14 th day of culture.
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202210100448.5A CN114517176B (en) | 2022-01-27 | 2022-01-27 | Kit for inducing IPS (in-plane switching) cells into NK (natural killer) cells and application method of kit |
| PCT/CN2023/071330 WO2023143006A1 (en) | 2022-01-27 | 2023-01-09 | Kit for inducing ips cells into nk cells and use method thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202210100448.5A CN114517176B (en) | 2022-01-27 | 2022-01-27 | Kit for inducing IPS (in-plane switching) cells into NK (natural killer) cells and application method of kit |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN114517176A true CN114517176A (en) | 2022-05-20 |
| CN114517176B CN114517176B (en) | 2022-12-02 |
Family
ID=81597683
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202210100448.5A Active CN114517176B (en) | 2022-01-27 | 2022-01-27 | Kit for inducing IPS (in-plane switching) cells into NK (natural killer) cells and application method of kit |
Country Status (2)
| Country | Link |
|---|---|
| CN (1) | CN114517176B (en) |
| WO (1) | WO2023143006A1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023143006A1 (en) * | 2022-01-27 | 2023-08-03 | 广东普罗凯融生物医药科技有限公司 | Kit for inducing ips cells into nk cells and use method thereof |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN111235105A (en) * | 2020-03-06 | 2020-06-05 | 安徽中盛溯源生物科技有限公司 | Method for differentiating human pluripotent stem cells into natural killer cells and application |
| CN112481209A (en) * | 2020-11-12 | 2021-03-12 | 广东普罗凯融生物医药科技有限公司 | Method for directionally differentiating iPS cells into NK cells |
| CN112608895A (en) * | 2020-12-18 | 2021-04-06 | 深圳市安棣生物科技有限责任公司 | Natural killer cell differentiated from human pluripotent stem cell and preparation method and application thereof |
| CN112662627A (en) * | 2021-01-19 | 2021-04-16 | 上海爱萨尔生物科技有限公司 | Culture solution for differentiating pluripotent stem cells into natural killer cells and differentiation method |
| WO2021174004A1 (en) * | 2020-02-28 | 2021-09-02 | Millennium Pharmaceuticals, Inc. | Method for producing natural killer cells from pluripotent stem cells |
| CN113801846A (en) * | 2021-09-22 | 2021-12-17 | 南京艾尔普再生医学科技有限公司 | Method for differentiating human induced pluripotent stem cells into natural killer cells |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN105462924B (en) * | 2015-12-04 | 2019-01-18 | 广州赛莱拉干细胞科技股份有限公司 | NK cell culture method and serum-free medium combination |
| CN105925527A (en) * | 2016-05-19 | 2016-09-07 | 成都百赛泰科生物科技有限公司 | Kit for preparing NK cells and application method thereof |
| GB201911953D0 (en) * | 2019-08-20 | 2019-10-02 | Adaptimmune Ltd | T cell production fram RAG inacivated iPSCs |
| CN114517176B (en) * | 2022-01-27 | 2022-12-02 | 广东普罗凯融生物医药科技有限公司 | Kit for inducing IPS (in-plane switching) cells into NK (natural killer) cells and application method of kit |
-
2022
- 2022-01-27 CN CN202210100448.5A patent/CN114517176B/en active Active
-
2023
- 2023-01-09 WO PCT/CN2023/071330 patent/WO2023143006A1/en not_active Ceased
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2021174004A1 (en) * | 2020-02-28 | 2021-09-02 | Millennium Pharmaceuticals, Inc. | Method for producing natural killer cells from pluripotent stem cells |
| CN111235105A (en) * | 2020-03-06 | 2020-06-05 | 安徽中盛溯源生物科技有限公司 | Method for differentiating human pluripotent stem cells into natural killer cells and application |
| CN112481209A (en) * | 2020-11-12 | 2021-03-12 | 广东普罗凯融生物医药科技有限公司 | Method for directionally differentiating iPS cells into NK cells |
| CN112608895A (en) * | 2020-12-18 | 2021-04-06 | 深圳市安棣生物科技有限责任公司 | Natural killer cell differentiated from human pluripotent stem cell and preparation method and application thereof |
| CN112662627A (en) * | 2021-01-19 | 2021-04-16 | 上海爱萨尔生物科技有限公司 | Culture solution for differentiating pluripotent stem cells into natural killer cells and differentiation method |
| CN113801846A (en) * | 2021-09-22 | 2021-12-17 | 南京艾尔普再生医学科技有限公司 | Method for differentiating human induced pluripotent stem cells into natural killer cells |
Non-Patent Citations (1)
| Title |
|---|
| LUPO KB等: "Differentiation of natural killer cells from induced pluripotent stem cells under defined, serum- and feeder-free conditions", 《CYTOTHERAPY》 * |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023143006A1 (en) * | 2022-01-27 | 2023-08-03 | 广东普罗凯融生物医药科技有限公司 | Kit for inducing ips cells into nk cells and use method thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2023143006A1 (en) | 2023-08-03 |
| CN114517176B (en) | 2022-12-02 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20250049847A1 (en) | A method for inducing ipsc differentiation to obtain cd34+cells and nk cells and application thereof | |
| CN101864396B (en) | Method for inducing megakaryoblast and megakaryocyte in vitro | |
| KR20150126943A (en) | Methods and materials for hematoendothelial differentiation of human pluripotent stem cells under defined conditions | |
| CN113832104B (en) | A method for differentiating macrophages from hiPS | |
| CN101649305B (en) | A method for expanding megakaryotic progenitor cells from human umbilical cord blood CD34+ cells | |
| CN112608896A (en) | NK cell culture method and application thereof | |
| Sambo et al. | Transient treatment of human pluripotent stem cells with DMSO to promote differentiation | |
| JP4268209B2 (en) | Method for culturing and proliferating hematopoietic stem cells or progenitor cells using endometrial cells | |
| Hermanson et al. | Human pluripotent stem cells as a renewable source of natural killer cells | |
| CN116333967A (en) | A kind of gastric organoid culture medium and culture method | |
| CN114517176B (en) | Kit for inducing IPS (in-plane switching) cells into NK (natural killer) cells and application method of kit | |
| CN104745529B (en) | Leptin is divided into purposes and its application in hematopoietic stem/progenitor in inducing embryo stem cell | |
| CN111172110A (en) | Culture method of umbilical cord blood CIK cells | |
| US12234482B2 (en) | Culture medium for expanding and culturing human liver progenitor cells and application thereof | |
| CN112080469B (en) | Application of T1 peptide in promoting cord blood hematopoietic stem cell proliferation in vitro | |
| CN113215086A (en) | Culture medium and method for induced differentiation of pluripotent stem cells into hematopoietic stem cells | |
| CN119307447A (en) | A culture system and in vitro expansion method of NK cells | |
| CN110857435B (en) | Culture medium for culturing immune cells separated from cord blood and culture method thereof | |
| CN110982783A (en) | Method for culturing spermatogonial stem cells and application thereof | |
| CN117757743A (en) | Culture medium and method for differentiating human induced pluripotent stem cells into natural killer cells | |
| CN120153063A (en) | Method for differentiating pluripotent stem cells into hematopoietic precursor cells and stem cells | |
| CN116042522A (en) | A method for large-scale expansion of human hematopoietic stem cells based on bionic microcarriers | |
| CN106661555B (en) | Efficient amplification method for autologous CIK cells | |
| KR102795799B1 (en) | Media composition for inducing erythroid differentiation and use thereof | |
| CN119662532B (en) | Culture medium, composition and culture method of human NK cells |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |