CN114437215B - Anti-human CD38 antibody and preparation method and application thereof - Google Patents
Anti-human CD38 antibody and preparation method and application thereof Download PDFInfo
- Publication number
- CN114437215B CN114437215B CN202011221844.0A CN202011221844A CN114437215B CN 114437215 B CN114437215 B CN 114437215B CN 202011221844 A CN202011221844 A CN 202011221844A CN 114437215 B CN114437215 B CN 114437215B
- Authority
- CN
- China
- Prior art keywords
- ser
- antibody
- thr
- tyr
- gly
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2896—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against molecules with a "CD"-designation, not provided for elsewhere
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/02—Antineoplastic agents specific for leukemia
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K1/00—General methods for the preparation of peptides, i.e. processes for the organic chemical preparation of peptides or proteins of any length
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/24—Immunoglobulins specific features characterized by taxonomic origin containing regions, domains or residues from different species, e.g. chimeric, humanized or veneered
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/56—Immunoglobulins specific features characterized by immunoglobulin fragments variable (Fv) region, i.e. VH and/or VL
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/56—Immunoglobulins specific features characterized by immunoglobulin fragments variable (Fv) region, i.e. VH and/or VL
- C07K2317/565—Complementarity determining region [CDR]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/73—Inducing cell death, e.g. apoptosis, necrosis or inhibition of cell proliferation
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/92—Affinity (KD), association rate (Ka), dissociation rate (Kd) or EC50 value
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Immunology (AREA)
- Animal Behavior & Ethology (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Molecular Biology (AREA)
- Biochemistry (AREA)
- Biophysics (AREA)
- Genetics & Genomics (AREA)
- Hematology (AREA)
- Oncology (AREA)
- Analytical Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Microbiology (AREA)
- Mycology (AREA)
- Epidemiology (AREA)
- Peptides Or Proteins (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
Abstract
Description
技术领域technical field
本发明涉及抗体领域,更具体地,本发明涉及抗人CD38的抗体及其制备方法和应用。The present invention relates to the field of antibodies, more specifically, the present invention relates to an anti-human CD38 antibody and its preparation method and application.
背景技术Background technique
CD38是一个46kDa的二型跨膜糖蛋白,胞外段由258个氨基酸组成,CD38的功能包括受体介导的粘附和信号传导及双功能体外酶活性促进胞内钙动员。在正常状态下,CD38低水平表达在淋系和髓系细胞上,但在多发性骨髓瘤中,恶性肿瘤细胞特异性高表达CD38,使CD38成为理想的骨髓瘤靶点。CD38 is a 46kDa type II transmembrane glycoprotein with an extracellular segment consisting of 258 amino acids. The functions of CD38 include receptor-mediated adhesion and signal transduction and bifunctional in vitro enzyme activity to promote intracellular calcium mobilization. Under normal conditions, CD38 is expressed at low levels on lymphoid and myeloid cells, but in multiple myeloma, malignant tumor cells specifically express CD38, making CD38 an ideal myeloma target.
多发性骨髓瘤是一类发生于B淋巴细胞的恶性浆细胞病。通常骨髓瘤细胞在骨髓内及骨骼海绵软组织内克隆性增生,引起溶骨性骨骼破坏,愈后不良多伴有贫血、肾衰竭和骨髓瘤细胞髓外浸润所导致的多种损害。中国多发性骨髓瘤的发病率已经超过急性白血病,是仅次于非霍奇金淋巴瘤之后居于第二位的血液系统恶性肿瘤。MM在中国每年新发病例约1.5~2万例,患者的中位生存期约4~5年,中国患病人数大约8-10万例。随着老龄化社会的到来和环境的恶化等多种因素的影响,发现多发性骨髓瘤呈发病比率上升。Multiple myeloma is a malignant plasma cell disease that occurs in B lymphocytes. Usually, myeloma cells proliferate clonally in the bone marrow and bone spongy soft tissue, causing osteolytic bone destruction. The poor prognosis is often accompanied by anemia, renal failure, and various damages caused by extramedullary infiltration of myeloma cells. The incidence of multiple myeloma in China has surpassed that of acute leukemia, and it is the second hematological malignancy after non-Hodgkin's lymphoma. There are about 15,000 to 20,000 new cases of MM in China every year, the median survival period of patients is about 4 to 5 years, and the number of patients in China is about 80,000 to 100,000. With the arrival of an aging society and the deterioration of the environment and other factors, the incidence of multiple myeloma has been found to increase.
研究发现CD38单克隆抗体主要通过Fc依赖性免疫效应机制杀死肿瘤细胞,包括补体介导的细胞毒作用(CDC)、抗体介导的细胞毒作用(ADCC)、抗体介导的细胞吞噬作用(ADCP),以及通过细胞凋亡(apoptosis)。此外,CD38抗体具有免疫调节作用:通过减少CD38+免疫抑制、调节细胞及促进T细胞扩增和活性等免疫调节作用,对骨髓瘤细胞进行调控。Studies have found that CD38 monoclonal antibodies mainly kill tumor cells through Fc-dependent immune effector mechanisms, including complement-mediated cytotoxicity (CDC), antibody-mediated cytotoxicity (ADCC), antibody-mediated phagocytosis ( ADCP), and through apoptosis. In addition, CD38 antibody has an immunomodulatory effect: it regulates myeloma cells by reducing CD38+ immunosuppression, regulating cells, and promoting T cell expansion and activity.
国际上针对CD38的抗体药物如强生的Daratumumab和赛诺菲的Isatuximab已上市,天境生物的MOR202在临床试验中。靶向CD38的抗体在多发性骨髓瘤治疗中相比于现行药物及在研的大分子、小分子药物均有非常显著的疗效优势,单药及联合用药均显示疗效优势,因此将能保持较好的竞争优势。Internationally, anti-CD38 antibody drugs such as Johnson & Johnson's Daratumumab and Sanofi's Isatuximab are already on the market, and I-Mab's MOR202 is in clinical trials. Antibodies targeting CD38 have very significant curative effect advantages in the treatment of multiple myeloma compared with current drugs, macromolecule and small molecule drugs under research. Good competitive advantage.
我国目前骨髓瘤的主要治疗方法是干细胞移植及杨森的万珂(硼替佐米)或Celgene的瑞复美(来那度胺)等联合用药方案治疗。近两年FDA批准了多个全新药物分子用于多发性骨髓瘤治疗,随着治疗新药的不断涌现,疗效得到提高、改善了预后,目前中位生存期为7-8年,正逐步将多发性骨髓瘤转为慢性病,客观上也带动了多发性骨髓瘤治疗市场的增长。CD38单抗的到来,为多发性骨髓瘤患者,特别是复发和难治性患者提供了新的选择。期待CD38抗体领域的研究能取得更多突破,造福更多患者。At present, the main treatment methods for myeloma in my country are stem cell transplantation and Janssen's Velcade (bortezomib) or Celgene's REVLIMIDE (lenalidomide) and other combination therapy. In the past two years, the FDA has approved a number of new drug molecules for the treatment of multiple myeloma. With the continuous emergence of new drugs for treatment, the curative effect has been improved and the prognosis has been improved. The current median survival period is 7-8 years, and the multiple myeloma is gradually becoming The transformation of myeloma into a chronic disease objectively drives the growth of the multiple myeloma treatment market. The arrival of CD38 monoclonal antibody provides a new option for multiple myeloma patients, especially relapsed and refractory patients. We look forward to more breakthroughs in the field of CD38 antibody research to benefit more patients.
发明内容Contents of the invention
本发明的发明人进行了大量试验,得到了一组可以特异性结合细胞表面CD38的单克隆抗体,这些抗体同时可以和食蟹猴的CD38结合。获得了一系列具有高、中和低不同亲和力的抗人CD38抗体,其能够通过Fc依赖性免疫效应机制杀死肿瘤细胞。The inventors of the present invention have conducted a large number of experiments and obtained a group of monoclonal antibodies that can specifically bind to CD38 on the cell surface, and these antibodies can also bind to CD38 in cynomolgus monkeys. A series of anti-human CD38 antibodies with high, medium and low affinity were obtained, which are able to kill tumor cells through Fc-dependent immune effector mechanisms.
第一方面,本申请提供了一种特异性结合人CD38的抗体或其抗原结合部分,其包含重链可变区,所述重链可变区包含HCDR3序列,任选地还包含HCDR1和/或HCDR2序列。在一些实施方案中,上述HCDR3序列包含选自SEQ ID NOs:3,6,9,12,15,18,21,24,27,30,33,36,39,42,45和48的氨基酸序列。在一些实施方案中,上述HCDR2序列包含选自SEQ ID NOs:2,5,8,11,14,17,20,23,26,29,32,35,38,41,44和47的氨基酸序列。在一些实施方案中,上述HCDR1序列包含选自SEQ ID NOs:1,4,7,10,13,16,19,22,25,28,31,34,37,40,43和46的氨基酸序列。In a first aspect, the present application provides an antibody or an antigen-binding portion thereof that specifically binds to human CD38, comprising a heavy chain variable region, the heavy chain variable region comprising HCDR3 sequence, optionally further comprising HCDR1 and/or or HCDR2 sequence. In some embodiments, the aforementioned HCDR3 sequence comprises an amino acid sequence selected from the group consisting of SEQ ID NOs: 3, 6, 9, 12, 15, 18, 21, 24, 27, 30, 33, 36, 39, 42, 45 and 48 . In some embodiments, the aforementioned HCDR2 sequence comprises an amino acid sequence selected from the group consisting of SEQ ID NOs: 2, 5, 8, 11, 14, 17, 20, 23, 26, 29, 32, 35, 38, 41, 44 and 47 . In some embodiments, the aforementioned HCDR1 sequence comprises an amino acid sequence selected from the group consisting of SEQ ID NOs: 1, 4, 7, 10, 13, 16, 19, 22, 25, 28, 31, 34, 37, 40, 43 and 46 .
在一些实施方案中,上述重链可变区包含与选自SEQ ID Nos:97,99,101,103,105,107,109和111的氨基酸序列具有至少80%同源性的氨基酸序列,或者所述重链可变区包含选自SEQ ID NOs:97,99,101,103,105,107,109和111的氨基酸序列。In some embodiments, the heavy chain variable region described above comprises an amino acid sequence having at least 80% homology to an amino acid sequence selected from the group consisting of SEQ ID Nos: 97, 99, 101, 103, 105, 107, 109 and 111, or The heavy chain variable region comprises an amino acid sequence selected from SEQ ID NOs: 97, 99, 101, 103, 105, 107, 109 and 111.
在一些实施方案中,特异性结合人CD38的抗体或其抗原结合部分还包含轻链可变区,其中所述轻链可变区包含LCDR1、LCDR2和/或LCDR3序列。在某些实施方案中,所述LCDR1序列包含选自SEQ ID NOs:49,52,55,58,61,64,67,70,73,76,79,82,85,88,91和94的氨基酸序列。在一些实施方案中,LCDR1序列还包含选自SEQ ID NO:115的氨基酸序列。在某些实施方案中,所述LCDR2序列包含选自SEQ ID NOs:50,53,56,59,62,65,68,71,74,77,80,83,86,89,92和95的氨基酸序列。在一些实施方案中,LCDR2序列还包含选自SEQ ID NO:116的氨基酸序列。在某些实施方案中,所述LCDR3序列包含选自SEQ ID NOs:51,54,57,60,63,66,69,72,75,78,81,84,87,90,93和96的氨基酸序列。在一些实施方案中,LCDR3序列还包含选自SEQ ID NO:117的氨基酸序列。In some embodiments, the antibody, or antigen-binding portion thereof, that specifically binds human CD38 further comprises a light chain variable region, wherein the light chain variable region comprises LCDR1, LCDR2 and/or LCDR3 sequences. In certain embodiments, the LCDR1 sequence comprises a sequence selected from SEQ ID NOs: 49, 52, 55, 58, 61, 64, 67, 70, 73, 76, 79, 82, 85, 88, 91 and 94 amino acid sequence. In some embodiments, the LCDR1 sequence further comprises an amino acid sequence selected from SEQ ID NO:115. In certain embodiments, the LCDR2 sequence comprises a sequence selected from SEQ ID NOs: 50, 53, 56, 59, 62, 65, 68, 71, 74, 77, 80, 83, 86, 89, 92 and 95 amino acid sequence. In some embodiments, the LCDR2 sequence further comprises an amino acid sequence selected from SEQ ID NO:116. In certain embodiments, the LCDR3 sequence comprises a sequence selected from SEQ ID NOs: 51, 54, 57, 60, 63, 66, 69, 72, 75, 78, 81, 84, 87, 90, 93 and 96 amino acid sequence. In some embodiments, the LCDR3 sequence further comprises an amino acid sequence selected from SEQ ID NO:117.
在一些实施方案中,上述轻链可变区包含与选自SEQ ID NOs:98,100,102,104,106,108,110和112的氨基酸序列具有至少80%同源性的氨基酸序列;或者所述轻链可变区包含选自SEQ ID NOs:98,100,102,104,106,108,110和112的氨基酸序列。In some embodiments, the light chain variable region described above comprises an amino acid sequence having at least 80% homology to an amino acid sequence selected from the group consisting of SEQ ID NOs: 98, 100, 102, 104, 106, 108, 110 and 112; or The light chain variable region comprises an amino acid sequence selected from SEQ ID NOs:98, 100, 102, 104, 106, 108, 110 and 112.
在可选的实施方案中,上述抗原结合部分选自Fab片段、Fab’片段、F(ab’)2片段、Fv片段、scFv片段、Fd片段和单域抗体。In an alternative embodiment, the above-mentioned antigen binding portion is selected from Fab fragments, Fab' fragments, F(ab')2 fragments, Fv fragments, scFv fragments, Fd fragments and single domain antibodies.
在一些实施方案中,第一方面所述的特异性结合人CD38的抗体为鼠源单克隆抗体。In some embodiments, the antibody specifically binding to human CD38 described in the first aspect is a murine monoclonal antibody.
在一些实施方案中,第一方面所述的特异性结合人CD38的抗体为人源化抗体。In some embodiments, the antibody specifically binding to human CD38 described in the first aspect is a humanized antibody.
第二方面,本申请提供了一种表达载体,所述表达载体含有的核苷酸分子可编码如上所述的氨基酸序列。In the second aspect, the present application provides an expression vector, and the nucleotide molecule contained in the expression vector can encode the above-mentioned amino acid sequence.
在一些实施方案中,所述表达载体为pTT5、pUC57、pDR1、pcDNA3.1(+)、pDHFF或pCHO 1.0等。In some embodiments, the expression vector is pTT5, pUC57, pDR1, pcDNA3.1(+), pDHFF or pCHO 1.0, etc.
第三方面,本申请提供了一种宿主细胞,所述宿主细胞含有如上所述的表达载体。在一些实施方案中,所述宿主细胞为HEK293、COS、CHO、NS0、sf9、sf21、DH5α、BL21(DE3)或TG1等。In a third aspect, the present application provides a host cell containing the above-mentioned expression vector. In some embodiments, the host cell is HEK293, COS, CHO, NSO, sf9, sf21, DH5α, BL21(DE3) or TG1, etc.
第四方面,本申请提供了一种制备第一方面所述的特异性结合人CD38的抗体或其抗原结合部分的方法,所述方法包括以下步骤:In a fourth aspect, the present application provides a method for preparing the antibody or antigen-binding portion thereof that specifically binds to human CD38 described in the first aspect, the method comprising the following steps:
a)在使得第三方面所述的宿主细胞能够产生所述抗体或其抗原结合部分的表达条件下,培养所述的宿主细胞,从而表达所述抗体或其抗原结合部分;以及a) culturing the host cell of the third aspect under expression conditions that enable the host cell to produce the antibody or antigen-binding portion thereof, thereby expressing the antibody or antigen-binding portion thereof; and
b)分离并纯化a)表达的所述抗体或其抗原结合部分。b) isolating and purifying said antibody or antigen-binding portion thereof expressed in a).
第五方面,本申请提供了一种药物组合物,所述组合物包含第一方面所述的抗人CD38抗体或其抗原结合部分以及药学上可接受的载体。In a fifth aspect, the present application provides a pharmaceutical composition, which comprises the anti-human CD38 antibody or antigen-binding portion thereof described in the first aspect and a pharmaceutically acceptable carrier.
在一些实施方案中,所述组合物用于治疗人CD38相关的疾病。In some embodiments, the composition is used to treat human CD38-related diseases.
在一些实施方案中,所述CD38相关疾病包括多发性骨髓瘤、非霍奇金淋巴瘤、AL型淀粉样变性、T细胞淋巴瘤、弥漫性大B细胞淋巴癌、慢性淋巴细胞白血病、急性淋巴细胞白血病、浆细胞性白血病、急性髓性白血病、非小细胞肺癌、胰腺癌、结直肠癌和三阴乳腺癌等。In some embodiments, the CD38-associated disease includes multiple myeloma, non-Hodgkin's lymphoma, AL amyloidosis, T-cell lymphoma, diffuse large B-cell lymphoma, chronic lymphocytic leukemia, acute lymphoblastic cell leukemia, plasma cell leukemia, acute myelogenous leukemia, non-small cell lung cancer, pancreatic cancer, colorectal cancer and triple-negative breast cancer, etc.
在其他方面,本申请提供了预防或治疗人CD38相关疾病的方法,包括给予有需要的个体第一方面所述的抗体或其抗原结合部分、或第五方面所述的药物组合物。In other aspects, the present application provides a method for preventing or treating human CD38-related diseases, comprising administering the antibody or antigen-binding portion thereof according to the first aspect, or the pharmaceutical composition according to the fifth aspect, to an individual in need.
本发明的抗人CD38抗体或其抗原结合部分能够特异性与人CD38结合,具有高、中和低不同亲和力的抗人CD38抗体,其能够通过Fc依赖性免疫效应机制杀死肿瘤细胞。本发明的抗人CD38抗体或其抗原结合部分可以用于预防或治疗CD38相关的疾病,例如多发性骨髓瘤、非霍奇金淋巴瘤、AL型淀粉样变性、T细胞淋巴瘤和弥漫性大B细胞淋巴癌等。The anti-human CD38 antibody or the antigen-binding part thereof of the present invention can specifically bind to human CD38, has high, medium and low affinity anti-human CD38 antibodies, and can kill tumor cells through an Fc-dependent immune effector mechanism. The anti-human CD38 antibody of the present invention or its antigen-binding portion can be used to prevent or treat CD38-related diseases, such as multiple myeloma, non-Hodgkin's lymphoma, AL-type amyloidosis, T-cell lymphoma and diffuse large B-cell lymphoma, etc.
附图说明Description of drawings
图1为人源化CD38单克隆抗体与Daudi细胞结合的实验结果Figure 1 shows the experimental results of humanized CD38 monoclonal antibody binding to Daudi cells
图2为人源化CD38抗体诱导Daudi淋巴瘤细胞凋亡的实验结果Figure 2 shows the experimental results of humanized CD38 antibody-induced apoptosis of Daudi lymphoma cells
图3为人源化CD38抗体抗Fc交联诱导Daudi淋巴瘤细胞凋亡的实验结果Figure 3 shows the experimental results of humanized CD38 antibody anti-Fc cross-linking induced apoptosis of Daudi lymphoma cells
具体实施方式Detailed ways
本申请提供了特异性结合于人CD38的新的抗CD38抗体或其抗原结合部分。在优选实施方案中,本申请的抗体或其抗原结合部分具有高、中和低不同亲和力结合CD38的活性,同时可以结合食蟹猴CD38。本申请还提供了该抗体或其抗原结合片段的氨基酸、包含所述氨基酸的载体、包含所述氨基酸或载体的宿主细胞、制备和纯化该抗体的方法以及所述抗体或其抗原结合片段的医学和生物学应用,例如预防或治疗CD38相关疾病或病症。The present application provides novel anti-CD38 antibodies or antigen-binding portions thereof that specifically bind to human CD38. In a preferred embodiment, the antibody or antigen-binding portion thereof of the present application has high, medium and low affinity binding activities to CD38, and can also bind cynomolgus monkey CD38. The present application also provides the amino acid of the antibody or its antigen-binding fragment, the carrier comprising the amino acid, the host cell comprising the amino acid or the vector, the method for preparing and purifying the antibody, and the medical treatment of the antibody or its antigen-binding fragment. And biological applications, such as prevention or treatment of CD38-related diseases or conditions.
为容易地理解本申请,首先定义本文中使用的某些术语。To facilitate understanding of this application, certain terms used herein are first defined.
本文所用术语“抗体”指包含四条多肽链,即通过双硫键互连的两条重链(H)链及两条轻链(L)的免疫球蛋白分子,以及其多聚体(例如IgM)。各重链包含重链可变区(缩写为VH)及重链恒定区(缩写为CH)。重链恒定区包含三个域,即CH1、CH2及CH3。各轻链包含轻链可变区(缩写为VL)及轻链恒定区(缩写为CL)。轻链恒定区包含一个域(CL1)。VH及VL区可进一步细分称为互补决定区(CDR)的高变区,其中穿插有称为构架区(FR)的保守区。The term "antibody" as used herein refers to an immunoglobulin molecule comprising four polypeptide chains, two heavy (H) chains and two light (L) chains interconnected by disulfide bonds, as well as multimers thereof (such as IgM ). Each heavy chain is comprised of a heavy chain variable region (abbreviated VH) and a heavy chain constant region (abbreviated CH). The heavy chain constant region comprises three domains, CH1, CH2 and CH3. Each light chain is comprised of a light chain variable region (abbreviated VL) and a light chain constant region (abbreviated CL). The light chain constant region comprises one domain (CL1). The VH and VL regions can be further subdivided into hypervariable regions called complementarity determining regions (CDRs), interspersed with conserved regions called framework regions (FRs).
如本文所用,术语抗体的“抗原结合部分”是指负责结合抗原的完整抗体分子的一部分或区段。抗原结合域可以包含重链可变区(VH)、轻链可变区(VL)或上述两者。抗体的抗原结合片段可使用任何适合的标准技术从完整抗体分子制备,所述标准技术包括蛋白水解消化或重组遗传工程化技术等。抗原结合部分的非限制性实例包括:Fab片段;F(ab')2片段;Fd片段;Fv片段;单链Fv(scFv)分子;单域抗体;dAb片段及由模拟抗体高变区的氨基酸残基组成的最小识别单元(例如分离的CDR)。术语“抗原结合部分”也包括其它工程化的分子,如双抗体、三抗体、四抗体及微型抗体等。As used herein, the term "antigen-binding portion" of an antibody refers to a portion or segment of an intact antibody molecule that is responsible for binding antigen. The antigen binding domain may comprise a heavy chain variable region (VH), a light chain variable region (VL), or both. Antigen-binding fragments of antibodies can be prepared from intact antibody molecules using any suitable standard technique, including proteolytic digestion or recombinant genetic engineering techniques, among others. Non-limiting examples of antigen binding portions include: Fab fragments; F(ab')2 fragments; Fd fragments; Fv fragments; single chain Fv (scFv) molecules; The smallest unit of recognition composed of residues (eg, isolated CDRs). The term "antigen-binding portion" also includes other engineered molecules, such as diabodies, triabodies, tetrabodies, and minibodies.
如本文所用,术语“重链可变区(VH)”及“轻链可变区(VL)”分别指单一抗体可变重链及轻链区,其包含FR1、2、3及4及CDR 1、2及3。As used herein, the terms "heavy chain variable region (VH)" and "light chain variable region (VL)" refer to a single antibody variable heavy and light chain region, respectively, comprising FR1, 2, 3, and 4 and the
本领域技术人员公知,互补决定区(CDR,通常有CDR1、CDR2及CDR3)是可变区中对抗体的亲和力和特异性影响最大的区域。VH或VL的CDR序列有两种常见的定义方式,即kabat定义和Chothia定义,例如参见Kabat et al,“Sequences of Proteins ofImmunological Interest”,National Institutes of Health,Bethesda,Md.(1991);A1-Lazikani et al.,J.Mol.Biol.273:927-948(1997);以及Martin et al.,Proc.Natl.Acad.Sci.USA86:9268-9272(1989)。对于给定抗体的可变区序列,可以根据Kabat定义或者Chothia定义来确定VH和VL序列中CDR区序列。在本申请的实施方案中,利用Kabat定义CDR序列。在本文中,重链可变区的CDR1、CDR2及CDR3分别简称为HCDR1、HCDR2及HCDR3;轻链可变区的CDR1、CDR2及CDR3分别简称为LCDR1、LCDR2及LCDR3。It is well known to those skilled in the art that complementarity determining regions (CDRs, usually CDR1, CDR2 and CDR3) are regions in the variable region that have the greatest influence on the affinity and specificity of antibodies. There are two common ways to define the CDR sequence of VH or VL, namely kabat definition and Chothia definition, see for example Kabat et al, "Sequences of Proteins of Immunological Interest", National Institutes of Health, Bethesda, Md. (1991); A1- Lazikani et al., J. Mol. Biol. 273:927-948 (1997); and Martin et al., Proc. Natl. Acad. Sci. USA 86:9268-9272 (1989). For the variable region sequence of a given antibody, the CDR region sequences in the VH and VL sequences can be determined according to the Kabat definition or the Chothia definition. In an embodiment of the present application, Kabat is used to define CDR sequences. Herein, CDR1, CDR2 and CDR3 of the heavy chain variable region are abbreviated as HCDR1, HCDR2 and HCDR3, respectively; CDR1, CDR2 and CDR3 of the light chain variable region are abbreviated as LCDR1, LCDR2 and LCDR3, respectively.
对于给定抗体的可变区序列,可以通过多种方式分析可变区序列中CDR区序列,例如可以利用在线软件Abysis确定(http://www.abysis.org/)。For the variable region sequence of a given antibody, the CDR region sequence in the variable region sequence can be analyzed in various ways, for example, it can be determined using the online software Abysis (http://www.abysis.org/).
如本文所用术语“特异性结合”,是指两个分子之间的非随机结合反应,例如抗体至抗原表位的结合,例如抗体以比其对非特异性抗原的亲和性大至少两倍的亲和性结合于特异性抗原的能力。然而应了解,抗体能够特异性结合于两种或更多其序列相关的抗原。例如,本发明的抗体可特异性结合于人类与非人类(例如小鼠或非人类灵长动物)的CD38。The term "specific binding" as used herein refers to a non-random binding reaction between two molecules, for example the binding of an antibody to an antigenic epitope, for example an antibody with an affinity at least two times greater than its affinity for a non-specific antigen The ability to bind with affinity to a specific antigen. It is understood, however, that an antibody is capable of specifically binding to two or more antigens that are related in sequence. For example, antibodies of the invention can specifically bind to CD38 in humans and non-humans (eg, mice or non-human primates).
本文所用术语“单克隆抗体”指由基本同质的抗体群体获得的抗体,即,除了可能在少量个体中存在自然发生的突变以外,组成群体的各个抗体是相同的。本文所述单克隆抗体特别包括“嵌合”抗体,其中重链和/或轻链的一部分与来源于具体物种或属于具体抗体类或亚类的抗体中的对应序列相同或同源,而重链和/或轻链的余下部分与来源于另一物种或属于另一抗体类或亚类的抗体中的对应序列相同或同源,并且还包括这样的抗体的片段,只要它们能表现出所期望的生物学活性(参见,美国专利号4,816,567;和Morrisonet al,Proc.Natl.Acad.Sci.USA 81:6851-6855(1984))。The term "monoclonal antibody" as used herein refers to an antibody obtained from a population of substantially homogeneous antibodies, ie, the individual antibodies comprising the population are identical except for the possible presence of naturally occurring mutations in a small number of individuals. The monoclonal antibodies described herein specifically include "chimeric" antibodies, in which a portion of the heavy and/or light chain is identical or homologous to the corresponding sequence in an antibody derived from a particular species or belonging to a particular antibody class or subclass, and the heavy chain and/or the remainder of the light chain is identical or homologous to the corresponding sequence in an antibody derived from another species or belonging to another antibody class or subclass, and also includes fragments of such antibodies, so long as they exhibit the desired (See, US Pat. No. 4,816,567; and Morrison et al, Proc. Natl. Acad. Sci. USA 81:6851-6855 (1984)).
如本文所用,术语“同源性”被定义为经过序列比对和引入空位后,氨基酸或核苷酸序列变体中相同的残基的百分比,如果需要,达到最大百分比的同源性。用于比对的方法和计算机程序在本领域内是公知的。本文所述的“至少80%同源性”是指同源性为80%至100%中的任一值,例如85%、90%、95%、99%等。As used herein, the term "homology" is defined as the percentage of residues in amino acid or nucleotide sequence variants that are identical after alignment of the sequences and introduction of gaps, if desired, to achieve a maximum percentage homology. Methods and computer programs for alignment are well known in the art. The "at least 80% homology" mentioned herein refers to any value from 80% to 100% homology, such as 85%, 90%, 95%, 99% and so on.
如本文所用,术语“CD38相关疾病”包括多发性骨髓瘤、非霍奇金淋巴瘤、AL型淀粉样变性、T细胞淋巴瘤和弥漫性大B细胞淋巴癌等。As used herein, the term "CD38-associated disease" includes multiple myeloma, non-Hodgkin's lymphoma, AL amyloidosis, T-cell lymphoma, diffuse large B-cell lymphoma, and the like.
一方面,本申请提供了特异性结合CD38的抗体或其抗原结合部分,其包含重链可变区和/或轻链可变区。表1-3中示例性列出了适用于本申请公开的抗体的CDR、重链和轻链可变区氨基酸序列。In one aspect, the application provides an antibody or antigen-binding portion thereof that specifically binds CD38, comprising a heavy chain variable region and/or a light chain variable region. Tables 1-3 exemplarily list the CDR, heavy chain and light chain variable region amino acid sequences suitable for the antibodies disclosed in the present application.
在具体的实施方案中,HCDR3选自SEQ ID NOs:3,6,9,12,15,18,21,24,27,30,33,36,39,42,45和48所示的氨基酸序列。在另一具体的实施方案中,HCDR3选自SEQ ID NOs:3,9,18,21,27,33,36和45所示的氨基酸序列。在优选的实施方案中,HCDR3选自9,18,21,27,33,36和45所示的氨基酸序列。In specific embodiments, HCDR3 is selected from the amino acid sequences shown in SEQ ID NOs: 3, 6, 9, 12, 15, 18, 21, 24, 27, 30, 33, 36, 39, 42, 45 and 48 . In another specific embodiment, HCDR3 is selected from the amino acid sequences shown in SEQ ID NOs: 3, 9, 18, 21, 27, 33, 36 and 45. In a preferred embodiment, HCDR3 is selected from the amino acid sequences shown in 9, 18, 21, 27, 33, 36 and 45.
在具体的实施方案中,HCDR2选自SEQ ID NOs:2,5,8,11,14,17,20,23,26,29,32,35,38,41,44和47所示的氨基酸序列。在另一具体的实施方案中,HCDR2选自SEQ ID NOs:2,8,17,20,26,32,35和44所示的氨基酸序列。在优选的实施方案中,HCDR2选自8,17,20,26,32,35和44所示的氨基酸序列。In specific embodiments, HCDR2 is selected from the amino acid sequences shown in SEQ ID NOs: 2, 5, 8, 11, 14, 17, 20, 23, 26, 29, 32, 35, 38, 41, 44 and 47 . In another specific embodiment, HCDR2 is selected from the amino acid sequences shown in SEQ ID NOs: 2, 8, 17, 20, 26, 32, 35 and 44. In a preferred embodiment, HCDR2 is selected from the amino acid sequences shown in 8, 17, 20, 26, 32, 35 and 44.
在具体的实施方案中,HCDR1选自SEQ ID NOs:1,4,7,10,13,16,19,22,25,28,31,34,37,40,43和46所示的氨基酸序列。在另一具体的实施方案中,HCDR1选自SEQ ID NOs:1,7,16,19,25,31,34和43所示的氨基酸序列。在优选的实施方案中,HCDR1选自7,16,19,25,31,34和43所示的氨基酸序列。In specific embodiments, HCDR1 is selected from the amino acid sequences shown in SEQ ID NOs: 1, 4, 7, 10, 13, 16, 19, 22, 25, 28, 31, 34, 37, 40, 43 and 46 . In another specific embodiment, HCDR1 is selected from the amino acid sequences shown in SEQ ID NOs: 1, 7, 16, 19, 25, 31, 34 and 43. In a preferred embodiment, HCDR1 is selected from the amino acid sequences shown in 7, 16, 19, 25, 31, 34 and 43.
在一些实施方案中,本文公开的抗体重链可变区包含选自SEQ ID NOs:97,99,101,103,105,107,109和111的氨基酸序列。在具体的实施方案中,所述重链可变区由选自SEQ ID NOs:97,99,101,103,105,107,109和111的氨基酸序列组成。In some embodiments, an antibody heavy chain variable region disclosed herein comprises an amino acid sequence selected from the group consisting of SEQ ID NOs:97, 99, 101, 103, 105, 107, 109, and 111. In specific embodiments, said heavy chain variable region consists of an amino acid sequence selected from the group consisting of SEQ ID NOs:97, 99, 101, 103, 105, 107, 109 and 111.
本文公开的抗体或其抗原结合部分在包含重链可变区的基础上还可以进一步包含轻链可变区。An antibody or antigen-binding portion thereof disclosed herein may further comprise a light chain variable region in addition to a heavy chain variable region.
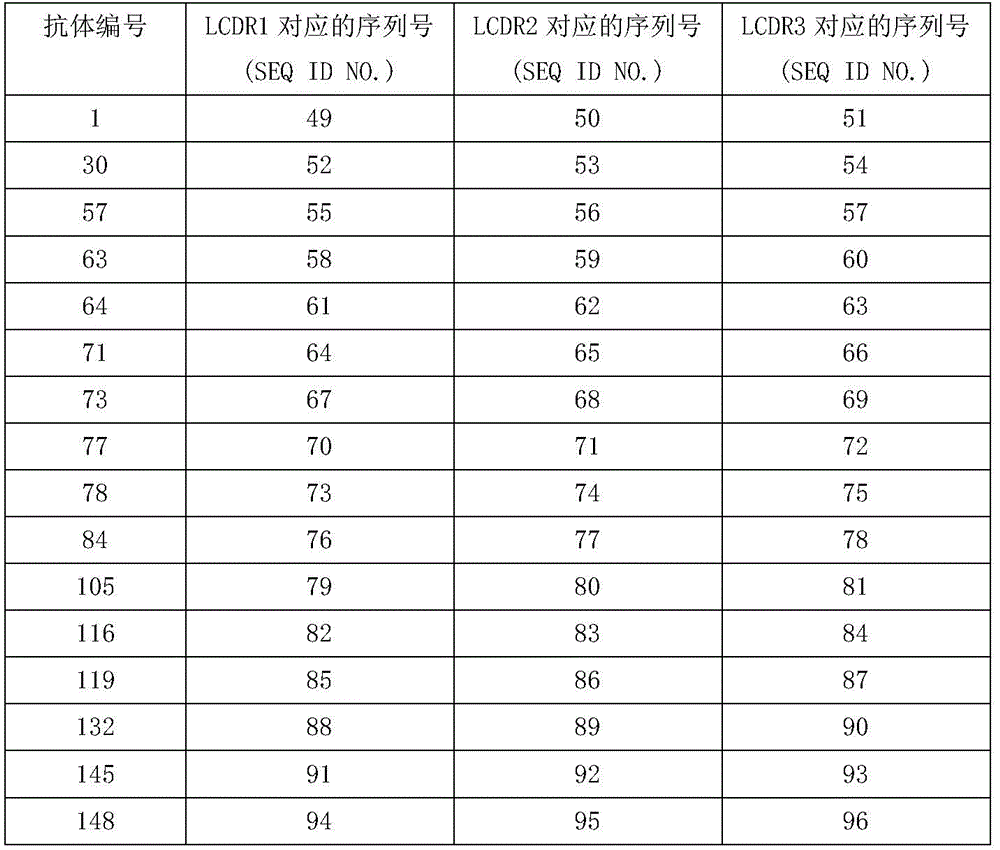
在一些实施方案中,所述轻链可变区的CDR3(LCDR3)选自SEQ ID NOs:51,54,57,60,63,66,69,72,75,78,81,84,87,90,93,96和117所示的氨基酸序列。在另一具体的实施方案中LCDR3选自SEQ ID NOs:51,57,66,69,75,81,84和117所示的氨基酸序列。在优选的实施方案中,LCDR3选自SEQ ID NOs:57,66,69,75,81,84和117所示的氨基酸序列。In some embodiments, the CDR3 (LCDR3) of the light chain variable region is selected from SEQ ID NOs: 51, 54, 57, 60, 63, 66, 69, 72, 75, 78, 81, 84, 87, Amino acid sequences shown at 90, 93, 96 and 117. In another specific embodiment, LCDR3 is selected from the amino acid sequences shown in SEQ ID NOs: 51, 57, 66, 69, 75, 81, 84 and 117. In a preferred embodiment, LCDR3 is selected from the amino acid sequences shown in SEQ ID NOs: 57, 66, 69, 75, 81, 84 and 117.
在一些实施方案中,LCDR2选自SEQ ID NOs:50,53,56,59,62,65,68,71,74,77,80,83,86,89,92,95和116所示的氨基酸序列,或者选自SEQ ID NOs:50,65,68,74,80,83,92和116所示的氨基酸序列。在优选的实施方案中,LCDR2选自SEQ ID NOs:65,68,74,80,83,92和116所示的氨基酸序列。In some embodiments, LCDR2 is selected from the amino acids shown in SEQ ID NOs: 50, 53, 56, 59, 62, 65, 68, 71, 74, 77, 80, 83, 86, 89, 92, 95 and 116 sequence, or selected from the amino acid sequences shown in SEQ ID NOs: 50, 65, 68, 74, 80, 83, 92 and 116. In a preferred embodiment, LCDR2 is selected from the amino acid sequences shown in SEQ ID NOs: 65, 68, 74, 80, 83, 92 and 116.
在一些实施方案中,LCDR1选自SEQ ID NOs:49,52,55,58,61,64,67,70,73,76,79,82,85,88,91,94和115的氨基酸序列,或者选自SEQ ID NOs:55,64,67,73,79,82,91和115的氨基酸序列。在优选的实施方案中,LCDR1选自SEQ ID NOs:55,64,67,73,79,82和91所示的氨基酸序列。In some embodiments, LCDR1 is selected from the amino acid sequence of SEQ ID NOs: 49, 52, 55, 58, 61, 64, 67, 70, 73, 76, 79, 82, 85, 88, 91, 94, and 115, Or an amino acid sequence selected from SEQ ID NOs: 55, 64, 67, 73, 79, 82, 91 and 115. In a preferred embodiment, LCDR1 is selected from the amino acid sequences shown in SEQ ID NOs: 55, 64, 67, 73, 79, 82 and 91.
在一些实施方案中,本文公开的抗体轻链可变区包含选自SEQ ID NOs:98,100,102,104,106,108,110和112的氨基酸序列。在具体的实施方案中,所述轻链可变区由选自SEQ ID NOs:98,100,102,104,106,108,110和112的氨基酸序列组成。In some embodiments, an antibody light chain variable region disclosed herein comprises an amino acid sequence selected from the group consisting of SEQ ID NOs:98, 100, 102, 104, 106, 108, 110, and 112. In specific embodiments, said light chain variable region consists of an amino acid sequence selected from the group consisting of SEQ ID NOs:98, 100, 102, 104, 106, 108, 110 and 112.
在一些实施方案中,本文公开的抗体的重链或重链可变区、轻链或轻链可变区可以在上述所列举的各自对应的具体氨基酸序列的基础上取代、缺失或添加至少一个氨基酸,且得到的变体仍然保持结合人CD38的活性。In some embodiments, the heavy chain or the heavy chain variable region, the light chain or the light chain variable region of the antibody disclosed herein can be substituted, deleted or added at least one on the basis of the respective specific amino acid sequences listed above Amino acids, and the resulting variant still retains the activity of binding human CD38.
在某些实施方案中,上述氨基酸取代、缺失或添加的数目为1-30个,优选为1-20个,更优选为1-10个。在优选的实施方案中,序列变体与原氨基酸序列相差约1、2、3、4、5、6、7、8、9、或10个氨基酸的取代、缺失和/或添加。在更优选的实施方案中,序列变体与原氨基酸序列相差约1、2、3、4或5个氨基酸的取代、缺失或添加。在具体的实施方案中,所述氨基酸取代为保守性取代。In certain embodiments, the number of amino acid substitutions, deletions or additions is 1-30, preferably 1-20, more preferably 1-10. In preferred embodiments, sequence variants differ from the original amino acid sequence by about 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10 amino acid substitutions, deletions, and/or additions. In more preferred embodiments, the sequence variant differs from the original amino acid sequence by a substitution, deletion or addition of about 1, 2, 3, 4 or 5 amino acids. In specific embodiments, the amino acid substitutions are conservative substitutions.
在优选的实施方案中,本文公开的抗体为抗体57H、105H、116H、或145H,其中抗体57H的重链可变区序列如SEQ ID NO:99所示,轻链可变区序列如SEQ ID NO:100所示,其中LCDR2优选为SEQ ID NO:116所示,其余CDR序列与鼠源抗体57的相同;抗体105H的重链可变区序列如SEQ ID NO:107所示,轻链可变区序列如SEQ ID NO:108所示,其中CDR序列与鼠源抗体105相同;抗体116H的重链可变区序列如SEQ ID NO:109所示,轻链可变区序列如SEQID NO:110所示,其中CDR序列与鼠源抗体116相同;抗体145H的重链可变区序列如SEQ IDNO:111所示,轻链可变区序列如SEQ ID NO:112所示,其中LCDR3优选为SEQ ID NO:117所示,其余CDR序列与鼠源抗体145的相同。In a preferred embodiment, the antibody disclosed herein is
在一些的实施方案中,本文公开的抗体为单克隆抗体。在具体的实施方案中,本文公开的抗体为人源化的抗体。In some embodiments, the antibodies disclosed herein are monoclonal antibodies. In specific embodiments, the antibodies disclosed herein are humanized antibodies.
本文公开的抗体或其抗原结合部分能够特异性结合人CD38。在具体的实施方案中,所述抗体或其抗原结合部分特异性结合人CD38或食蟹猴CD38。在优选的实施方案中,所述抗体或其抗原结合部分特异性结合人CD38。Antibodies disclosed herein, or antigen-binding portions thereof, are capable of specifically binding human CD38. In specific embodiments, the antibody, or antigen-binding portion thereof, specifically binds human CD38 or cynomolgus CD38. In preferred embodiments, the antibody or antigen binding portion thereof specifically binds human CD38.
在一些实施方案中,任何合适的表达载体都可以用于本申请。例如,所述表达载体可以为pTT5、pUC57、pDR1、pcDNA3.1(+)、pDHFF及pCHO 1.0中的一种。表达载体中可以包括连接有合适的转录和翻译调节序列的融合DNA序列。In some embodiments, any suitable expression vector may be used in the present application. For example, the expression vector may be one of pTT5, pUC57, pDR1, pcDNA3.1(+), pDHFF and pCHO 1.0. Expression vectors may include fusion DNA sequences linked with appropriate transcriptional and translational regulatory sequences.
在一些实施方案中,可用的宿主细胞为含有上述表达载体的细胞,可以是真核细胞,如哺乳动物或昆虫宿主细胞培养系统均可用于本申请的抗体或其抗原结合部分的表达。例如,HEK293细胞、COS、CHO、NS0、sf9及sf21等均可适用于本发明。所述宿主细胞也可以为含有上述表达载体的原核细胞,例如可以为DH5α、BL21(DE3)或TG1等。In some embodiments, the available host cells are cells containing the above-mentioned expression vectors, which may be eukaryotic cells, such as mammalian or insect host cell culture systems, which can be used for the expression of the antibody or its antigen-binding portion of the present application. For example, HEK293 cells, COS, CHO, NSO, sf9, and sf21 are all applicable to the present invention. The host cell may also be a prokaryotic cell containing the above expression vector, such as DH5α, BL21(DE3) or TG1, etc.
在一些实施方案中,本文公开的抗人CD38单克隆抗体的制备方法包括:在适合的表达条件下,培养宿主细胞,从而表达抗人CD38单克隆抗体;分离和纯化表达的抗人CD38单克隆抗体。利用上述方法,可以将重组蛋白纯化为基本均一的物质,例如在SDS-PAGE电泳上为单一条带。In some embodiments, the preparation method of the anti-human CD38 monoclonal antibody disclosed herein comprises: culturing host cells under suitable expression conditions, thereby expressing the anti-human CD38 monoclonal antibody; isolating and purifying the expressed anti-human CD38 monoclonal antibody Antibody. Using the above method, the recombinant protein can be purified into a substantially uniform substance, such as a single band on SDS-PAGE electrophoresis.
在一些实施方案中,可以利用亲和层析的方法对本文公开的抗人CD38抗体进行分离纯化,根据所利用的亲和柱的特性,可以使用常规的方法例如高盐缓冲液、改变PH等方法洗脱结合在亲和柱上的抗人CD38抗体。In some embodiments, the anti-human CD38 antibody disclosed herein can be separated and purified by using affinity chromatography. According to the characteristics of the affinity column used, conventional methods such as high-salt buffer, changing pH, etc. can be used. Methods Elution of anti-human CD38 antibody bound to the affinity column.
在一些实施方案中,本文公开的人源化的抗人CD38单克隆抗体是通过以下方法得到的:利用实验室制备的人CD38抗原免疫Balb/c小鼠,在多次免疫小鼠滴度较高后取小鼠脾细胞与杂交瘤细胞融合并筛选出具有不同亲和力的杂交瘤细胞株。更具体地,本申请的发明人通过大量实验,首先分别表达了人CD38抗原,在此基础上利用不同的佐剂与人CD38抗原混合免疫小鼠,然后进一步将上述小鼠的脾细胞与杂交瘤细胞株sp2/0融合,融合后的杂交瘤利用人CD38抗原筛选出阳性细胞株,在验证其对人CD38结合和与Daudi细胞的结合后获得目标细胞株。将目标分子进行人源化改造后,将轻链和重链基因同时克隆到真核表达载体pCHO1.0中。将上述表达载体通过脂质体法转染CHO细胞,然后用嘌呤霉素和甲胺蝶呤筛选阳性细胞克隆,将筛选得到的高表达克隆用无血清培养基扩大培养,用Protein A亲和柱分离或纯化人源化的抗人CD38抗原单克隆抗体。In some embodiments, the humanized anti-human CD38 monoclonal antibody disclosed herein is obtained by the following method: the human CD38 antigen prepared in the laboratory is used to immunize Balb/c mice, and the titer of the mice after multiple immunizations is compared with Then take mouse splenocytes to fuse with hybridoma cells and screen out hybridoma cell lines with different affinity. More specifically, through a large number of experiments, the inventors of the present application firstly expressed human CD38 antigen respectively, and on this basis, used different adjuvants to mix with human CD38 antigen to immunize mice, and then further hybridized the splenocytes of the above mice with The tumor cell line sp2/0 was fused, and the fused hybridoma was screened by human CD38 antigen to select a positive cell line, and the target cell line was obtained after verifying its binding to human CD38 and Daudi cells. After humanizing the target molecule, the light chain and heavy chain genes were simultaneously cloned into the eukaryotic expression vector pCHO1.0. The above expression vector was transfected into CHO cells by liposome method, and then the positive cell clones were screened with puromycin and methotrexate, and the screened high-expression clones were expanded and cultured in serum-free medium, and protein A affinity column was used to Isolation or purification of humanized anti-human CD38 antigen monoclonal antibody.
在另外一些实施方案中,可以使用本领域的常规技术,例如PCR诱变进一步改变鼠源的亲本抗体来产生抗体的嵌合或人源化形式或其他变异形式。本申请的亲本抗体可以在例如抗原互补决定区(CDR)结构域内被突变来产生变异抗体,可筛选其目的性质的存在,例如结合亲和力(更低的KD)、IC50、特异性、优先结合等等。优选地,变异抗体中目的性质是相对于亲本抗体中性质的改善。优选氨基酸替代变异抗体,并且亲本抗体分子的至少1、2、3、4、5、6、7、8、9或10个氨基酸残基被去除且在它的位置上插入不同的残基。用于替代诱变的最感兴趣的位点是一个或更多个CDR区,但是也考虑框架区(FR)改变。优选保守的氨基酸替代,也可引入非保守氨基酸改变并用获得的变异抗体筛选目的性质。In other embodiments, conventional techniques in the art, such as PCR mutagenesis, can be used to further alter the murine parental antibody to generate chimeric or humanized or other variant forms of the antibody. The parent antibody of the present application can be mutated, for example, within the complementarity determining region (CDR) domain to generate a variant antibody that can be screened for the presence of desired properties, such as binding affinity (lower KD), IC50, specificity, preferential binding, etc. wait. Preferably, the property of interest in the variant antibody is an improvement over the property in the parental antibody. Amino acid substitution variant antibodies are preferred, and at least 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 amino acid residues of the parent antibody molecule are removed and a different residue is inserted in its place. The sites of most interest for substitution mutagenesis are one or more CDR regions, but framework region (FR) alterations are also considered. Conservative amino acid substitutions are preferred, and non-conservative amino acid changes can also be introduced and the obtained variant antibodies can be used to screen for the desired properties.
本申请提供了药物组合物,其包含本文公开的抗体或其抗原结合部分以及药学上可接受的载体。本文公开的上述抗人CD38单克隆抗体,可以和药学上可接受的载体一起配制成药物制剂,从而更稳定地发挥疗效。在一些实施方案中,这些制剂可以保证本文公开的抗人CD38单克隆抗体的氨基酸核心序列构象的完整性,同时还保护蛋白质的多官能团防止其降解(包括但不限于凝聚、脱酰胺或氧化)。在一些实施方案中,对于液体制剂,通常可以在2℃-8℃条件下保存至少稳定一年。在一些实施方案中,对于冻干制剂,在30℃下至少六个月保持稳定。The present application provides a pharmaceutical composition comprising an antibody disclosed herein, or an antigen-binding portion thereof, and a pharmaceutically acceptable carrier. The anti-human CD38 monoclonal antibody disclosed herein can be formulated into a pharmaceutical preparation together with a pharmaceutically acceptable carrier, so as to exert a more stable curative effect. In some embodiments, these formulations can ensure the conformational integrity of the amino acid core sequence of the anti-human CD38 monoclonal antibodies disclosed herein, while also protecting the multifunctional groups of the protein from degradation (including but not limited to aggregation, deamidation, or oxidation) . In some embodiments, liquid formulations are usually stable for at least one year when stored at 2°C-8°C. In some embodiments, for lyophilized formulations, it is stable for at least six months at 30°C.
在一些实施方案中,所述抗人CD38抗体单克隆抗体制剂可为制药领域常用的混悬、水针、冻干等制剂,优选水针或冻干制剂,对于本文公开的抗人CD38单克隆抗体的水针或冻干制剂,药学上可以接受的辅料包括但不限于:表面活性剂、溶液稳定剂、等渗调节剂和缓冲液或其组合。在一些实施方案中,表面活性剂包括但不限于:非离子型表面活性剂如聚氧乙烯山梨醇脂肪酸酯(吐温20或80),泊洛沙姆(如泊洛沙姆188),Triton,十二烷基硫酸钠(SDS),月桂硫酸钠,十四烷基、亚油基或十八烷基肌氨酸,Pluronics,MONAQUATTM等,其加入量应使抗人CD38单克隆抗体的颗粒化趋势最小。在一些实施方案中,溶液稳定剂包括但不限于以下列举的一种或其组合:糖类,例如还原性糖和非还原性糖;氨基酸类,例如谷氨酸单钠或组氨酸;醇类,例如三元醇、高级糖醇、丙二醇、聚乙二醇等。溶液稳定剂的加入量应该使最后形成的制剂在本领域的技术人员认为达到稳定的时间内保持稳定状态。等渗调节剂包括但不限于氯化钠、甘露醇或其组合。缓冲液包括但不限于:Tris、组氨酸缓冲液、磷酸盐缓冲液或其组合。In some embodiments, the anti-human CD38 antibody monoclonal antibody preparations can be suspension, aqueous injection, freeze-dried and other preparations commonly used in the pharmaceutical field, preferably aqueous injection or freeze-dried preparations, for the anti-human CD38 monoclonal antibody disclosed herein For the aqueous injection or freeze-dried preparation of the antibody, the pharmaceutically acceptable excipients include but are not limited to: surfactants, solution stabilizers, isotonic regulators, buffers or combinations thereof. In some embodiments, surfactants include, but are not limited to: nonionic surfactants such as polyoxyethylene sorbitan fatty acid esters (
本申请还提供了预防或治疗CD38相关疾病的方法,其包括给予个体抗人CD38抗体、或者包含抗人CD38抗体的组合物。具体地说,本文公开的抗人CD38抗体能够有效地预防和/或治疗CD38相关疾病。The present application also provides a method for preventing or treating CD38-related diseases, which comprises administering an anti-human CD38 antibody or a composition comprising an anti-human CD38 antibody to an individual. Specifically, the anti-human CD38 antibodies disclosed herein can effectively prevent and/or treat CD38-related diseases.
在一些实施方案中,所述CD38相关疾病包括多发性骨髓瘤、非霍奇金淋巴瘤、AL型淀粉样变性、T细胞淋巴瘤、弥漫性大B细胞淋巴癌、慢性淋巴细胞白血病、急性淋巴细胞白血病、浆细胞性白血病、急性髓性白血病、非小细胞肺癌、胰腺癌、结直肠癌和三阴乳腺癌等。In some embodiments, the CD38-associated disease includes multiple myeloma, non-Hodgkin's lymphoma, AL amyloidosis, T-cell lymphoma, diffuse large B-cell lymphoma, chronic lymphocytic leukemia, acute lymphoblastic cell leukemia, plasma cell leukemia, acute myelogenous leukemia, non-small cell lung cancer, pancreatic cancer, colorectal cancer and triple-negative breast cancer, etc.
本文公开的抗人CD38单克隆抗体及其组合物在对包括人在内的动物给药时,给药剂量因病人的年龄和体重、疾病特性和严重性以及给药途径而异,可以参考动物实验的结果和综合情况,总给药量不能超过一定范围。When the anti-human CD38 monoclonal antibody disclosed herein and the composition thereof are administered to animals including humans, the dosage varies according to the age and weight of the patient, the nature and severity of the disease, and the route of administration. Reference can be made to the Based on the results of the experiment and the overall situation, the total dosage should not exceed a certain range.
抗体或其组合物的施用剂量和频率可根据对疾病进行预防或治疗而变化。在预防性应用中,向尚未处于疾病状态的患者施用含有本申请的抗体或其混合物的组合物以增强患者抵抗力,此量定义为“预防性有效剂量”。在此用途中,具体的剂量又视患者健康状况及全身免疫性而定。通常以相对不频繁的间隔施用相对较低剂量较长时间。在治疗性应用中,有时需要以相对较短间隔施用相对较高剂量直至疾病进展减缓或终止为止,且优选直至患者显示疾病症状部分或完全改善为止。此后,可向患者施用预防性方案。本领域普通技术人员可以容易地根据实际需要掌握具体的剂量和频率。The dosage and frequency of administration of an antibody or composition thereof may vary depending on whether the disease is being prevented or treated. In prophylactic applications, the composition containing the antibody of the present application or its mixture is administered to patients who are not yet in a disease state to enhance the patient's resistance, and this amount is defined as "prophylactic effective dose". In this application, the specific dose depends on the patient's health status and systemic immunity. Generally, relatively lower doses are administered at relatively infrequent intervals for a longer period of time. In therapeutic applications, it is sometimes desirable to administer relatively higher doses at relatively shorter intervals until disease progression is slowed or terminated, and preferably until the patient shows partial or complete amelioration of disease symptoms. Thereafter, prophylactic regimens can be administered to the patient. Those of ordinary skill in the art can easily grasp the specific dosage and frequency according to actual needs.
本说明书和权利要求书中,词语“包括”、“包含”和“含有”意指“包括但不限于”,且并非意图排除其他部分、添加物、组分、或步骤。In the present description and claims, the words "comprising", "comprising" and "containing" mean "including but not limited to", and are not intended to exclude other parts, additives, components, or steps.
应该理解,在本申请的特定方面、实施方案或实施例中描述的特征、特性、组分或步骤,可适用于本文所描述的任何其他的方面、实施方案或实施例,除非与之矛盾。It should be understood that features, characteristics, components or steps described in one particular aspect, embodiment or example of the present application can be applied to any other aspect, embodiment or example described herein unless incompatible therewith.
上述公开内容总体上描述了本发明。以下具体的实施例是对本发明作进一步的说明,不应理解为对本发明的限制。实施例不包括对传统常规方法的详细描述,这样的方法对于本领域具有普通技术的人员是众所周知的,并且在许多出版物中都有所描述,例如分子克隆手册、冷泉港的抗体技术实验手册。未注明试剂来源的为常规试剂。The above disclosure generally describes the present invention. The following specific examples are to further illustrate the present invention, and should not be construed as limiting the present invention. The examples do not include detailed descriptions of traditional routine methods, which are well known to those of ordinary skill in the art and are described in numerous publications, for example Handbook of Molecular Cloning, Laboratory Handbook of Antibody Technology of Cold Spring Harbor . Reagent sources not indicated are conventional reagents.
具体实施例specific embodiment
实施例1人CD38胞外段的Fc标签和Flag标签抗原、食蟹猴CD38胞外段Flag标签蛋 白、参比抗体Daratumumab的制备 Example 1 Preparation of the Fc tag and Flag tag antigen of the extracellular region of human CD38, the Flag tag protein of the extracellular region of cynomolgus monkey CD38 , and the reference antibody Daratumumab
人CD38抗原序列购买于义翘神州(货号:HG10818-M)。通过PCR法将人CD38N端第43-300位氨基酸片段分别与hFc片段和Flag标签(DYKDDDDK)进行拼接,hFc片段和Flag标签位于C端,并构建至pTT5表达载体(实验室保存)上,获得pTT5(hCD38-ECD-hFc)和pTT5(hCD38-ECD-Flag),选取测序结果完全正确的克隆进行质粒抽提后转染。The human CD38 antigen sequence was purchased from Sino Biological (Product No.: HG10818-M). The 43-300th amino acid fragment of the N-terminal of human CD38 was spliced with the hFc fragment and the Flag tag (DYKDDDDK) by the PCR method, and the hFc fragment and the Flag tag were located at the C-terminal, and constructed on the pTT5 expression vector (preserved in the laboratory) to obtain For pTT5(hCD38-ECD-hFc) and pTT5(hCD38-ECD-Flag), clones with completely correct sequencing results were selected for plasmid extraction and transfection.
食蟹猴CD38序列购买于义翘神州(货号:CG90050-G)。通过PCR法将其N端第44-301位氨基酸片段与Flag标签(DYKDDDDK)进行拼接,Flag标签位于C端,并构建至pTT5表达载体(实验室保存)上,获得pTT5(cyno-CD38-ECD-Flag),选取测序结果完全正确的克隆进行质粒抽提后转染。The cynomolgus monkey CD38 sequence was purchased from Sino Biological (Cat. No.: CG90050-G). The 44th-301th amino acid fragment of its N-terminal was spliced with the Flag tag (DYKDDDDK) by PCR method, and the Flag tag was located at the C-terminal, and it was constructed on the pTT5 expression vector (preserved in the laboratory) to obtain pTT5 (cyno-CD38-ECD -Flag), select clones with completely correct sequencing results for plasmid extraction and transfection.
Daratumumab的氨基酸序列来自IMGT,经过密码子优化后全基因合成重链、轻链的可变区核苷酸序列。通过PCR法将Daratumumab重链的可变区与IgG1的恒定区连接;Daratumumab轻链可变区与Kappa链的恒定区连接。然后将这些片段克隆至pTT5表达载体,测序验证确认后进行质粒抽提以备转染。The amino acid sequence of Daratumumab was obtained from IMGT, and the nucleotide sequences of the variable regions of the heavy and light chains were synthesized from the whole gene after codon optimization. The variable region of the heavy chain of Daratumumab was connected to the constant region of IgG1 by PCR; the variable region of the light chain of Daratumumab was connected to the constant region of the Kappa chain. These fragments were then cloned into the pTT5 expression vector, and plasmid extraction was carried out after sequencing verification to prepare for transfection.
通过PEI法将质粒转染至HEK293E细胞系(实验室保存)。利用含3mM的丙戊酸的Freestyle293培养基(购自Gibco公司)培养5天后,利用Protein A亲和层析(购自Pharmacia公司)或Flag亲和层析(购自Genscript公司)从细胞培养上清中纯化目的蛋白。蛋白的定量通过二喹啉甲酸(Bicinchoninic acid,BCA)方法进行,纯化得到的蛋白用于以下的进一步分析与研究。纯化得到的蛋白用于以下的小鼠免疫及进一步分析与研究。The plasmid was transfected into HEK293E cell line (preserved in laboratory) by PEI method. After culturing for 5 days in Freestyle293 medium (purchased from Gibco) containing 3 mM valproic acid, protein A affinity chromatography (purchased from Pharmacia) or Flag affinity chromatography (purchased from Genscript) Purify the protein of interest from the serum. The protein was quantified by bicinchoninic acid (BCA) method, and the purified protein was used for the following further analysis and research. The purified protein was used for the following mouse immunization and further analysis and research.
实施例2 hCD38-ECD-hFc的免疫 Example 2 Immunization of hCD38-ECD-hFc
将100μg/鼠的hCD38-ECD-hFc抗原用生理盐水稀释成75μl后,与等体积的弗氏完全佐剂混合,并经超声乳化完全后对4-5周龄的Balb/c小鼠(购自上海灵畅生物科技有限公司,动物生产许可证号:SCXK(沪)2013-0018)进行皮下多点注射。三周后,将50μg/鼠的蛋白同样稀释成75μl后与等体积弗氏不完全佐剂混合,超声乳化完全后对小鼠进行皮下多点注射,两周后再次重复此免疫。所有小鼠在第三次免疫后一周剪尾取血分离血清,利用包被hCD38-ECD-hFc抗原的ELISA进行血清滴度的检测。对于血清抗体效价>10000的小鼠,在取血后一周进行冲击免疫:尾静脉注射10μg抗原/100μl生理盐水/鼠。Dilute 100 μg/mouse hCD38-ECD-hFc antigen with physiological saline to 75 μl, mix with an equal volume of Freund’s complete adjuvant, and complete phacoemulsification for 4-5 week-old Balb/c mice (purchased Subcutaneous multi-point injection was performed from Shanghai Lingchang Biotechnology Co., Ltd., animal production license number: SCXK (Shanghai) 2013-0018). Three weeks later, 50 μg/mouse protein was also diluted to 75 μl and mixed with an equal volume of Freund’s incomplete adjuvant. After phacoemulsification was complete, the mice were injected subcutaneously at multiple points, and the immunization was repeated two weeks later. One week after the third immunization, the tails of all mice were cut to collect blood to separate serum, and the serum titer was detected by ELISA coated with hCD38-ECD-hFc antigen. For mice with serum antibody titer > 10000, pulse immunization was performed one week after blood collection: 10 μg antigen/100 μl normal saline/mouse were injected into the tail vein.
滴度的检测通过ELISA方法进行:利用hCD38-ECD-hFc抗原包被ELISA板,包被浓度为1μg/ml,每孔100μl,4℃包被过夜。PBST(含0.5%Tween-20的PBS)洗板2次后拍干。每孔加入含1%BSA的包被液封闭200μl,常温下封闭4小时后拍干,至-20℃冰箱中保存待用。检测时在ELISA板中每孔加入不同浓度的小鼠血清100μl,设2个复孔,室温孵育1.5小时。PBST洗涤3次后拍干。加入用PBST1:10000倍稀释的HRP标记的兔抗鼠Ig抗体(购自Sigma公司)100μl,室温孵育1小时。PBST洗涤3次后拍干。每孔加100μl显色液(临用前将ELISA显色A液与显色B液按照1:1的体积比混匀)显色,随后每孔加入100μl 2M H2SO4终止液终止反应。立即用酶标仪(Molecular Device)在450nm波长处测量各孔OD值。The detection of the titer was carried out by ELISA method: the ELISA plate was coated with hCD38-ECD-hFc antigen, the coating concentration was 1 μg/ml, 100 μl per well, and coated overnight at 4°C. Plates were washed twice with PBST (PBS containing 0.5% Tween-20) and then patted dry. Add 200 μl of coating solution containing 1% BSA to each well to seal, block at room temperature for 4 hours, pat dry, and store in a -20°C refrigerator until use. During the detection, 100 μl of mouse serum of different concentrations was added to each well of the ELISA plate, and two duplicate wells were set up, and incubated at room temperature for 1.5 hours. After washing 3 times with PBST, pat dry. Add 100 μl of HRP-labeled rabbit anti-mouse Ig antibody (purchased from Sigma) diluted 1:10000 times with PBST, and incubate at room temperature for 1 hour. After washing 3 times with PBST, pat dry. Add 100 μl of chromogenic solution to each well (mix ELISA chromogenic solution A and chromogenic B solution at a volume ratio of 1:1 before use) for color development, and then add 100 μl of 2M H2SO4 stop solution to each well to stop the reaction. Immediately measure the OD value of each well with a microplate reader (Molecular Device) at a wavelength of 450 nm.
实施例3杂交瘤融合和筛选
杂交瘤sp2/0细胞(来自中国科学院典型培养物保藏委员会细胞库,保藏号为TCM-18)在37℃、5%CO2培养箱中培养,融合前一天换液。小鼠冲击免疫三天后取小鼠脾细胞进行融合。融合与筛选方法如下:取小鼠脾脏,研磨洗涤后进行脾细胞计数。将脾细胞和sp2/0细胞以10:1的比例混合,1500rpm离心7分钟。洗去上清液。1分钟内加入1ml PEG(1450),轻摇90秒,在2.5分钟内加入无血清DMEM培养液(购自Gibco公司)5ml,再一次性加5ml无血清培养液终止反应,静置5分钟,1280rpm离心8分钟。按照一块96孔板两百万个sp2/0细胞的数量,将细胞均匀接种入96孔板,每孔200μl。先用含次黄嘌呤(hypoxanthine,H)、甲胺蝶呤(aminopterin,A)和胸腺嘧啶核苷(thymidine,T)的HAT培养基筛选,每3~4天半量换液,第10天改用HT培养基。10天后,待杂交瘤细胞铺满96孔板底部大于10%时,取上清用hCD38-ECD-hFc抗原包被的酶标板进行ELISA检测。ELISA检测方法如实施例2中所述方法相同。挑选出阳性杂交瘤克隆于24孔板中扩大培养,有限稀释法进行亚克隆,获得稳定表达目的抗体的杂交瘤株后进行保种建库。Hybridoma sp2/0 cells (from the Cell Bank of the Type Culture Collection Committee of the Chinese Academy of Sciences, with the preservation number TCM-18) were cultured in a 37°C, 5% CO 2 incubator, and the medium was changed one day before fusion. Three days after the shock immunization of the mice, the splenocytes of the mice were taken for fusion. The fusion and screening method is as follows: the spleen of the mouse is taken, ground and washed, and the spleen cells are counted. Splenocytes and sp2/0 cells were mixed at a ratio of 10:1 and centrifuged at 1500rpm for 7 minutes. Wash away the supernatant. Add 1ml of PEG (1450) within 1 minute, shake gently for 90 seconds, add 5ml of serum-free DMEM culture solution (purchased from Gibco) within 2.5 minutes, and then add 5ml of serum-free culture solution to stop the reaction, and let it stand for 5 minutes. Centrifuge at 1280 rpm for 8 minutes. According to the number of 2 million sp2/0 cells in a 96-well plate, the cells were evenly seeded into a 96-well plate, 200 μl per well. First use HAT medium containing hypoxanthine (H), methotrexate (A) and thymidine (T) to screen, change half of the medium every 3 to 4 days, and change the medium on the 10th day. Use HT medium. After 10 days, when the hybridoma cells covered more than 10% of the bottom of the 96-well plate, the supernatant was taken for ELISA detection with an enzyme label plate coated with hCD38-ECD-hFc antigen. The ELISA detection method is the same as that described in Example 2. The positive hybridoma clones were selected and cultured in a 24-well plate, subcloned by the limiting dilution method, and the hybridoma strains stably expressing the target antibody were obtained to preserve the species and build a library.
实施例4鼠源的抗人CD38单克隆抗体序列的测定
Determination of the sequence of the anti-human CD38 monoclonal antibody of
亲和力较高的阳性孔经过2~3轮的亚克隆最后得到了150株杂交瘤株。对这些杂交瘤细胞株进行抗体序列调取。使用Trizol提取各杂交瘤细胞株的总RNA,用逆转录试剂盒(购自ABI公司)将mRNA逆转录成cDNA。以文献报道的引物通过PCR扩增鼠源的抗人CD38单克隆抗体的轻链可变区和重链可变区基因,然后将PCR产物克隆入pGEM-T载体,测序并分析可变区基因序列。在GenBank中对获得的序列进行比对分析,所有序列均符合小鼠IgG可变区基因的特征。表1和表2列举了优选抗体的CDR区域的氨基酸序列。After 2-3 rounds of subcloning for the positive wells with higher affinity, 150 hybridoma strains were finally obtained. Antibody sequences were retrieved from these hybridoma cell lines. The total RNA of each hybridoma cell line was extracted using Trizol, and the mRNA was reverse-transcribed into cDNA using a reverse transcription kit (purchased from ABI). The light chain variable region and heavy chain variable region genes of the mouse-derived anti-human CD38 monoclonal antibody were amplified by PCR with primers reported in the literature, and then the PCR product was cloned into pGEM-T vector, sequenced and analyzed for the variable region genes sequence. The obtained sequences were compared and analyzed in GenBank, and all the sequences conformed to the characteristics of mouse IgG variable region genes. Table 1 and Table 2 list the amino acid sequences of the CDR regions of preferred antibodies.
表1:示例性鼠源抗人CD38抗体的重链CDR氨基酸序列Table 1: Amino acid sequences of heavy chain CDRs of exemplary murine anti-human CD38 antibodies
实施例5抗人CD38单克隆抗体的人源化 Example 5 Humanization of anti-human CD38 monoclonal antibody
根据序列分析结果,挑取1,57,71,73,77,78,105,116和145号抗体进行了嵌合抗体和人源化抗体的构建。嵌合抗体的构建通过截取鼠源抗体的重链可变区和轻链可变区,利用overlapping PCR分别与人IgG1的轻重链恒定区连接而成。According to the sequence analysis results,
表2:示例性鼠源抗人CD38抗体的轻链CDR氨基酸序列Table 2: Light chain CDR amino acid sequences of exemplary murine anti-human CD38 antibodies
根据Kabat法则对鼠源的抗人CD38单克隆抗体的轻链可变区和重链可变区的氨基酸序列进行分析并确定了3个CDR和4个FR。以105号抗体为例,通过在NCBI IgBlast与人IgG胚系序列(Germline)进行同源性比较,选择IGHV1-46*01为重链CDR移植模板,将鼠源的抗人CD38单克隆抗体105号的重链CDR区移植入IGHV1-46*01骨架区,构建成重链的CDR移植抗体。同样地,经过与人IgG胚系序列同源性比较,选择IGKV1-16*01为轻链CDR移植模板,将鼠源的抗人CD38单克隆抗体105号的轻链CDR区移植入IGKV1-16*01的骨架区,构建成轻链的CDR移植抗体。同时,在此基础上,对一些框架区的氨基酸位点进行了回复突变。在进行回复突变时,将氨基酸序列进行了Kabat编码,位点的位置由Kabat码指示。优选地,对于轻链可变区序列,将Kabat编码第36位的F回复为鼠源的L,将第43位的A突变成T,第44位的P回复为鼠源的I,第46位的S回复为鼠源的R,第66位的G回复为鼠源的R,第69位的T回复为鼠源的S。对于重链可变区序列,将Kabat编码第48位的M回复为鼠源的I,第67位的V回复为鼠源的A,第69位的M回复为鼠源的L,第71位的R回复为鼠源的A,第73位的T回复为鼠源的K,第78位的V回复为鼠源的A。上述可变区基因序列由生工生物按照Cricetulus griseus的密码子使用偏好进行密码子优化并合成。将合成的人源化可变区序列与人IgG1恒定区相连,此抗体定义为105号抗体的人源化抗体105-Humanization,105H)。According to the Kabat rule, the amino acid sequences of the light chain variable region and the heavy chain variable region of the mouse anti-human CD38 monoclonal antibody were analyzed and 3 CDRs and 4 FRs were determined. Taking antibody No. 105 as an example, IGHV1-46*01 was selected as the heavy chain CDR grafting template by comparing the homology between NCBI IgBlast and human IgG germline sequence (Germline), and the mouse-derived anti-human CD38 monoclonal antibody 105 The heavy chain CDR region of No. 1 was grafted into the framework region of IGHV1-46*01 to construct a heavy chain CDR grafted antibody. Similarly, after comparing the homology with the human IgG germline sequence, IGKV1-16*01 was selected as the light chain CDR transplantation template, and the light chain CDR region of the mouse-derived anti-human CD38 monoclonal antibody No. 105 was transplanted into IGKV1-16 * The skeleton region of 01 is constructed as a light chain CDR-grafted antibody. At the same time, on this basis, some amino acid positions in the framework region were back-mutated. When performing back mutation, the amino acid sequence was coded by Kabat, and the position of the site was indicated by the Kabat code. Preferably, for the light chain variable region sequence, the F at position 36 of the Kabat code is restored to L of mouse origin, the A at position 43 is mutated to T, the P at position 44 is restored to I of mouse origin, and the The S at position 46 was converted to the mouse-derived R, the G at position 66 was converted to the mouse-derived R, and the T at position 69 was converted to the mouse-derived S. For the heavy chain variable region sequence, the M at position 48 of the Kabat code is restored to I of murine origin, the V at position 67 is restored to A of murine origin, the M at position 69 is restored to L of murine origin, and the 71st position The R at position 73 is restored to mouse-derived A, the T at position 73 is restored to mouse-derived K, and the V at position 78 is restored to mouse-derived A. The above variable region gene sequence was codon-optimized and synthesized by Sangon Biotech according to the codon usage preference of Cricetulus griseus. The synthetic humanized variable region sequence was connected with the human IgG1 constant region, and this antibody was defined as the humanized antibody 105-Humanization of
利用上述相同原理,对其余抗体同样进行了人源化。人源化抗体的可变区氨基酸序列编号见表3,重链恒定区的氨基酸序列为SEQ ID NO:113所示,轻链恒定区的氨基酸序列为SEQ ID NO:114所示。利用pTT5载体分别构建人源化重链、轻链的瞬时表达载体,将上述轻重链组合利用HEK293E系统进行瞬时转染并表达抗体。HEK293E细胞在Free Style293Expression Medium(购自Gibco公司)培养基中培养,利用PEI转染法将质粒转入细胞5天后收取细胞上清,利用Protein A纯化后获得各个人源化单克隆抗体。Using the same principle as above, the remaining antibodies were similarly humanized. The amino acid sequence number of the variable region of the humanized antibody is shown in Table 3, the amino acid sequence of the heavy chain constant region is shown in SEQ ID NO: 113, and the amino acid sequence of the light chain constant region is shown in SEQ ID NO: 114. The pTT5 vector was used to construct the transient expression vectors of the humanized heavy chain and light chain respectively, and the combination of the above light and heavy chains was transiently transfected and expressed antibodies using the HEK293E system. HEK293E cells were cultured in Free Style293Expression Medium (purchased from Gibco) medium, and the plasmid was transferred into the cells by
最终,1号抗体人源化后的重链可变区氨基酸序列如SEQ ID NO:97所示;人源化后的轻链可变区氨基酸序列如SEQ ID NO:98所示。与人IgG1恒定区相连后,最终获得1H的抗体序列。Finally, the amino acid sequence of the heavy chain variable region of antibody No. 1 after humanization is shown in SEQ ID NO:97; the amino acid sequence of the humanized light chain variable region is shown in SEQ ID NO:98. After linking with the constant region of human IgG1, the antibody sequence of 1H is finally obtained.
57号抗体人源化后的重链可变区氨基酸序列如SEQ ID NO:99所示;人源化后的轻链可变区氨基酸序列如SEQ ID NO:100所示。与人IgG1恒定区相连后,最终获得57H的抗体序列。The amino acid sequence of the heavy chain variable region of antibody No. 57 after humanization is shown in SEQ ID NO: 99; the amino acid sequence of the humanized light chain variable region is shown in SEQ ID NO: 100. After linking with the constant region of human IgG1, the antibody sequence of 57H is finally obtained.
71号抗体人源化后的重链可变区氨基酸序列如SEQ ID NO:101所示;人源化后的轻链可变区氨基酸序列如SEQ ID NO:102所示。与人IgG1恒定区相连后,最终获得71H的抗体序列。The amino acid sequence of the heavy chain variable region of antibody No. 71 after humanization is shown in SEQ ID NO: 101; the amino acid sequence of the humanized light chain variable region is shown in SEQ ID NO: 102. After linking with the constant region of human IgG1, the antibody sequence of 71H was finally obtained.
73号抗体人源化后的重链可变区氨基酸序列如SEQ ID NO:103所示;人源化后的轻链可变区氨基酸序列如SEQ ID NO:104所示。与人IgG1恒定区相连后,最终获得73H的抗体序列。The amino acid sequence of the humanized heavy chain variable region of antibody No. 73 is shown in SEQ ID NO: 103; the amino acid sequence of the humanized light chain variable region is shown in SEQ ID NO: 104. After linking with the constant region of human IgG1, the antibody sequence of 73H was finally obtained.
78号抗体人源化后的重链可变区氨基酸序列如SEQ ID NO:105所示;人源化后的轻链可变区氨基酸序列如SEQ ID NO:106所示。与人IgG1恒定区相连后,最终获得78H的抗体序列。The amino acid sequence of the heavy chain variable region after humanization of antibody No. 78 is shown in SEQ ID NO: 105; the amino acid sequence of the humanized light chain variable region is shown in SEQ ID NO: 106. After linking with the constant region of human IgG1, the antibody sequence of 78H is finally obtained.
105号抗体人源化后的重链可变区氨基酸序列如SEQ ID NO:107所示;人源化后的轻链可变区氨基酸序列如SEQ ID NO:108所示。与人IgG1恒定区相连后,最终获得105H的抗体序列。The amino acid sequence of the heavy chain variable region of antibody No. 105 after humanization is shown in SEQ ID NO: 107; the amino acid sequence of the humanized light chain variable region is shown in SEQ ID NO: 108. After linking with the constant region of human IgG1, the antibody sequence of 105H is finally obtained.
116号抗体人源化后的重链可变区氨基酸序列如SEQ ID NO:109所示;人源化后的轻链可变区氨基酸序列如SEQ ID NO:110所示。与人IgG1恒定区相连后,最终获得116H的抗体序列。The amino acid sequence of the heavy chain variable region of antibody No. 116 after humanization is shown in SEQ ID NO: 109; the amino acid sequence of the humanized light chain variable region is shown in SEQ ID NO: 110. After linking with the human IgG1 constant region, the antibody sequence of 116H was finally obtained.
145号抗体人源化后的重链可变区氨基酸序列如SEQ ID NO:111所示;人源化后的轻链可变区氨基酸序列如SEQ ID NO:112所示。与人IgG1恒定区相连后,最终获得145H的抗体序列。The amino acid sequence of the heavy chain variable region of antibody No. 145 after humanization is shown in SEQ ID NO: 111; the amino acid sequence of the humanized light chain variable region is shown in SEQ ID NO: 112. After linking with the constant region of human IgG1, the antibody sequence of 145H is finally obtained.
表3:示例性人源化抗人CD38抗体重链和轻链可变区的氨基酸序列Table 3: Amino acid sequences of heavy and light chain variable regions of exemplary humanized anti-human CD38 antibodies
实施例6人源化CD38单克隆抗体与Daudi细胞的结合 Example 6 Binding of humanized CD38 monoclonal antibody to Daudi cells
将生长状态良好的Daudi细胞(实验室保存)取样计数,离心后用添加1%FBS的PBS重悬,密度调节为2X 107。抗体样品最高浓度10μg/ml,三倍梯度稀释(共8个梯度)后加样。在96孔培养板中,加入20ul/孔(4X 105/孔)Daudi细胞,并加入30μl梯度稀释的抗体药物,4℃孵育30分钟。加入180μl 1%FBS PBS清洗,1200rpm离心6min。用真空泵吸去上清后,加入羊抗人IgG-FITC(用1%FBS PBS 1:1000稀释)50μl,在4℃孵育30分钟。加入180μl 1%FBSPBS后1200rpm离心6分钟后用160μl PBS重悬。最后用流式细胞仪检测细胞表面荧光强度。利用GraphPad Prism6软件进行数据分析。实验结果见图1和表4。The well-growing Daudi cells (preserved in the laboratory) were sampled and counted, centrifuged and resuspended in PBS supplemented with 1% FBS, and the density was adjusted to
表4:人源化CD38抗体对Daudi细胞的结合Table 4: Binding of humanized CD38 antibodies to Daudi cells
实施例7人源化CD38抗体诱导Daudi淋巴瘤细胞凋亡 Example 7 Humanized CD38 Antibody Induces Daudi Lymphoma Cell Apoptosis
在96孔培养板200μl/孔的实验体系中,加入1X 105Daudi细胞/孔,并加入1.85和1.03μg/孔的抗体药物,培养24小时结束后,在培养孔中直接加入1:100的PI(PropidiumIodide)溶液(2μl),继续培养30分钟,离心去除上清,用PBS洗涤,离心去除上清,然后加入160μl PBS重悬,用流式细胞仪完成检测。设置未染色孔作为空白对照,参比抗体为Daratumumab(图2中简写为Dara)。图2的结果表明73H相较参比抗体显示了更强的诱导凋亡活性,其余抗体和参比抗体一致,并未见明显的诱导凋亡活性。Add
实施例8人源化CD38抗体抗Fc交联诱导Daudi淋巴瘤细胞凋亡 Example 8 Humanized CD38 Antibody Anti-Fc Crosslinking Induces Daudi Lymphoma Cell Apoptosis
在96孔培养板200μl/孔的实验体系中,加入1X 105Daudi细胞/孔,并加入1.85和1.03μg/孔的抗体药物,在细胞培养箱中孵育30分钟。然后加入1μg/孔抗人Fc抗体,继续培养24小时。培养24小时。培养结束后在培养孔中直接加入1:100的PI溶液(2μl),继续培养30分钟,离心去除上清,用PBS洗涤,离心去除上清,然后加入160μl PBS重悬,用流式细胞仪完成检测。设置未染色孔作为空白对照,参比抗体为Daratumumab(图3中简写为Dara)。图3的结果表明5个优选抗体相较参比抗体具有一致的较强的抗Fc交联诱导细胞凋亡活性。In the experimental system of 200 μl/well of 96-well culture plate, add 1X 105 Daudi cells/well, and add 1.85 and 1.03 μg/well of antibody drugs, and incubate in the cell culture incubator for 30 minutes. Then 1 μg/well anti-human Fc antibody was added, and culture was continued for 24 hours. Incubate for 24 hours. After the culture is over, directly add 1:100 PI solution (2μl) to the culture well, continue to culture for 30 minutes, centrifuge to remove the supernatant, wash with PBS, centrifuge to remove the supernatant, then add 160μl PBS to resuspend, and use a flow cytometer Complete the test. Set unstained wells as a blank control, and the reference antibody is Daratumumab (abbreviated as Dara in Figure 3). The results in Figure 3 show that the five preferred antibodies have consistently stronger anti-Fc cross-linking-induced apoptosis activity than the reference antibody.
实施例9人源化CD38单克隆抗体的亲和力测定 Example 9 Affinity Determination of Humanized CD38 Monoclonal Antibody
表达纯化的人源化抗体的亲和力通过Biacore T200(GE healthcare)检测。具体实验方法为:利用Protein-A CM5传感芯片(GE healthcare),以FC1(Flow cell 1)为参照通道,FC2(Flow cell 2)为样品通道。在FC2通道分别捕获人源抗体或对照抗体,随后注射不同浓度的hCD38-Flag或者cyno-CD38-Flag。循环条件为:在FCs所有通道中以50μl/min注射4min分析物,解离时间为20min,以10μl/min速率注射6M盐酸胍(国药集团化学试剂有限公司)30s进行表面再生,然后利用Biacore T200 Evaluation Software Ver 1.0计算捕获抗体的信号和无捕获抗体的信号差值及相互作用的亲和力。如表5和表6所显示。实验结果表明优选抗体均能够与人和食蟹猴的CD38结合,具有高、中和低不同亲和力。The affinity of the expressed and purified humanized antibody was detected by Biacore T200 (GE healthcare). The specific experimental method is: using Protein-A CM5 sensor chip (GE healthcare), using FC1 (Flow cell 1) as the reference channel, and FC2 (Flow cell 2) as the sample channel. Human antibodies or control antibodies were captured in the FC2 channel, followed by injection of different concentrations of hCD38-Flag or cyno-CD38-Flag. The cycle conditions were as follows: inject analyte in all channels of FCs at 50 μl/min for 4 min, dissociate time for 20 min, inject 6M guanidine hydrochloride (Sinopharm Chemical Reagent Co., Ltd.) at a rate of 10 μl/min for 30 s for surface regeneration, and then use Biacore T200 Evaluation Software Ver 1.0 calculates the difference between the signal of the capture antibody and the signal without the capture antibody and the affinity of the interaction. As shown in Table 5 and Table 6. Experimental results show that the preferred antibodies can all bind to CD38 of humans and cynomolgus monkeys, with high, medium and low affinities.
表5:人源化CD38抗体结合人CD38的亲和力Table 5: Binding affinity of humanized CD38 antibodies to human CD38
表6:人源化CD38抗体结合猴CD38的亲和力Table 6: Binding affinity of humanized CD38 antibodies to monkey CD38
结论:本发明的发明人进行了大量试验,得到了一组可以特异性结合细胞表面CD38的单克隆抗体,这些抗体同时可以和食蟹猴的CD38结合。获得了一系列具有高、中和低不同亲和力的抗人CD38抗体,其能够通过Fc依赖性免疫效应机制杀死肿瘤细胞。Conclusion: The inventors of the present invention have conducted a large number of experiments and obtained a group of monoclonal antibodies that can specifically bind to CD38 on the cell surface, and these antibodies can also bind to CD38 in cynomolgus monkeys. A series of anti-human CD38 antibodies with high, medium and low affinity were obtained, which are able to kill tumor cells through Fc-dependent immune effector mechanisms.
可以理解,尽管本申请以某种形式被说明,但本申请并不局限于本说明书中所显示和描述的内容。对本领域的技术人员显而易见的是,在不偏离本申请的范围的前提下还可对所述实施方式和/或某一特征或参数做出各种变化。这些变化都在本申请要求保护的范围内。It is to be understood that although the application has been illustrated in some form, the application is not limited to what is shown and described in this specification. It will be obvious to those skilled in the art that various changes may be made to the described embodiments and/or a certain feature or parameter without departing from the scope of the present application. These changes are all within the protection scope of this application.
SEQUENCE LISTINGSEQUENCE LISTING
<110> 上海麦济生物技术有限公司<110> Shanghai Maiji Biotechnology Co., Ltd.
<120> 抗人CD38抗体及其制备方法和用途<120> Anti-human CD38 antibody, preparation method and use thereof
<130> anti-CD38<130> anti-CD38
<160> 114<160> 114
<170> PatentIn version 3.5<170> PatentIn version 3.5
<210> 1<210> 1
<211> 5<211> 5
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 1<400> 1
Ser Ser Trp Met HisSer Ser Trp Met His
1 51 5
<210> 2<210> 2
<211> 14<211> 14
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 2<400> 2
Glu Ile His Pro Asp Ser Gly Lys Thr Asn Tyr Asn Glu LysGlu Ile His Pro Asp Ser Gly Lys Thr Asn Tyr Asn Glu Lys
1 5 101 5 10
<210> 3<210> 3
<211> 11<211> 11
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 3<400> 3
Leu Ser Ser Pro Tyr Tyr Tyr Val Met Asp TyrLeu Ser Ser Pro Tyr Tyr Tyr Val Met Asp Tyr
1 5 101 5 10
<210> 4<210> 4
<211> 5<211> 5
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 4<400> 4
Asn Tyr Trp Ile GlnAsn Tyr Trp Ile Gln
1 51 5
<210> 5<210> 5
<211> 17<211> 17
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 5<400> 5
Glu Met Phe Pro Gly Thr Gly Lys Thr Tyr Tyr Asn Glu Lys Phe LysGlu Met Phe Pro Gly Thr Gly Lys Thr Tyr Tyr Asn Glu Lys Phe Lys
1 5 10 151 5 10 15
GlyGly
<210> 6<210> 6
<211> 10<211> 10
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 6<400> 6
Ser Gly Tyr Asp Tyr Asp Gly Phe Thr TyrSer Gly Tyr Asp Tyr Asp Gly Phe Thr Tyr
1 5 101 5 10
<210> 7<210> 7
<211> 5<211> 5
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 7<400> 7
Ser Phe Gly Met HisSer Phe Gly Met His
1 51 5
<210> 8<210> 8
<211> 17<211> 17
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 8<400> 8
Tyr Ile Gly Gly Asp Ser Asn Ile Ile Tyr Tyr Ala Asp Thr Val LysTyr Ile Gly Gly Asp Ser Asn Ile Ile Tyr Tyr Ala Asp Thr Val Lys
1 5 10 151 5 10 15
GlyGly
<210> 9<210> 9
<211> 8<211> 8
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 9<400> 9
Ser His Asp Tyr Thr Met Asp TyrSer His Asp Tyr Thr Met Asp Tyr
1 51 5
<210> 10<210> 10
<211> 5<211> 5
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 10<400> 10
Ser Tyr Trp Ile GlnSer Tyr Trp Ile Gln
1 51 5
<210> 11<210> 11
<211> 17<211> 17
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 11<400> 11
Glu Ile Phe Pro Gly Thr Gly Lys Thr Tyr Tyr Asn Gly Lys Phe LysGlu Ile Phe Pro Gly Thr Gly Lys Thr Tyr Tyr Asn Gly Lys Phe Lys
1 5 10 151 5 10 15
GlyGly
<210> 12<210> 12
<211> 10<211> 10
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 12<400> 12
Ser Pro Tyr Asp Ile Val Gly Phe Asp TyrSer Pro Tyr Asp Ile Val Gly Phe Asp Tyr
1 5 101 5 10
<210> 13<210> 13
<211> 5<211> 5
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 13<400> 13
Gly Tyr Asn Met AsnGly Tyr Asn Met Asn
1 51 5
<210> 14<210> 14
<211> 17<211> 17
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 14<400> 14
Asn Ile Asp Pro His Phe Gly Gly Thr Arg Tyr Asn Gln Lys Phe LysAsn Ile Asp Pro His Phe Gly Gly Thr Arg Tyr Asn Gln Lys Phe Lys
1 5 10 151 5 10 15
GlyGly
<210> 15<210> 15
<211> 10<211> 10
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 15<400> 15
Gln His Gly Asn Trp Val Gly Phe Ala TyrGln His Gly Asn Trp Val Gly Phe Ala Tyr
1 5 101 5 10
<210> 16<210> 16
<211> 5<211> 5
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 16<400> 16
Lys Tyr Trp Ile GluLys Tyr Trp Ile Glu
1 51 5
<210> 17<210> 17
<211> 14<211> 14
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 17<400> 17
Glu Ile Ser Pro Gly Ile Asp Lys Thr Asn Tyr Asn Glu LysGlu Ile Ser Pro Gly Ile Asp Lys Thr Asn Tyr Asn Glu Lys
1 5 101 5 10
<210> 18<210> 18
<211> 14<211> 14
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 18<400> 18
Gly Glu Glu Tyr Gly Asn Ser Val Arg Tyr Ala Met Asp TyrGly Glu Glu Tyr Gly Asn Ser Val Arg Tyr Ala Met Asp Tyr
1 5 101 5 10
<210> 19<210> 19
<211> 5<211> 5
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 19<400> 19
Gly Tyr Asn Met AsnGly Tyr Asn Met Asn
1 51 5
<210> 20<210> 20
<211> 17<211> 17
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 20<400> 20
Asn Ile Asp Pro Tyr Phe Gly Gly Thr Ile Tyr Asn Gln Lys Phe LysAsn Ile Asp Pro Tyr Phe Gly Gly Thr Ile Tyr Asn Gln Lys Phe Lys
1 5 10 151 5 10 15
GlyGly
<210> 21<210> 21
<211> 10<211> 10
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 21<400> 21
Gln Tyr Gly Asn Trp Val Gly Phe Ala TyrGln Tyr Gly Asn Trp Val Gly Phe Ala Tyr
1 5 101 5 10
<210> 22<210> 22
<211> 5<211> 5
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 22<400> 22
Asp Arg Ala Val HisAsp Arg Ala Val His
1 51 5
<210> 23<210> 23
<211> 17<211> 17
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 23<400> 23
Val Ile Ser Pro Tyr Tyr Gly Asp Ala Thr Tyr Asn Gln Lys Phe LysVal Ile Ser Pro Tyr Tyr Gly Asp Ala Thr Tyr Asn Gln Lys Phe Lys
1 5 10 151 5 10 15
GlyGly
<210> 24<210> 24
<211> 10<211> 10
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 24<400> 24
Val Asp Phe Phe Tyr Tyr Ala Met Asp TyrVal Asp Phe Phe Tyr Tyr Ala Met Asp Tyr
1 5 101 5 10
<210> 25<210> 25
<211> 6<211> 6
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 25<400> 25
Ser Gly Tyr Tyr Trp AsnSer Gly Tyr Tyr Trp Asn
1 51 5
<210> 26<210> 26
<211> 16<211> 16
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 26<400> 26
Tyr Arg Ser Tyr Asp Gly Ser Tyr Asn Tyr Asn Pro Ser Leu Glu AsnTyr Arg Ser Tyr Asp Gly Ser Tyr Asn Tyr Asn Pro Ser Leu Glu Asn
1 5 10 151 5 10 15
<210> 27<210> 27
<211> 12<211> 12
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 27<400> 27
Glu Glu Arg Tyr Ile Leu Arg Tyr Ala Met Asp TyrGlu Glu Arg Tyr Ile Leu Arg Tyr Ala Met Asp Tyr
1 5 101 5 10
<210> 28<210> 28
<211> 5<211> 5
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 28<400> 28
Asp Arg Ala Val HisAsp Arg Ala Val His
1 51 5
<210> 29<210> 29
<211> 17<211> 17
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 29<400> 29
Val Ile Ser Pro Tyr Tyr Gly Asp Ala Thr Tyr Asn Gln Lys Phe LysVal Ile Ser Pro Tyr Tyr Gly Asp Ala Thr Tyr Asn Gln Lys Phe Lys
1 5 10 151 5 10 15
GlyGly
<210> 30<210> 30
<211> 10<211> 10
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 30<400> 30
Val Asp Phe Tyr Tyr Tyr Ala Met Asp TyrVal Asp Phe Tyr Tyr Tyr Ala Met Asp Tyr
1 5 101 5 10
<210> 31<210> 31
<211> 5<211> 5
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 31<400> 31
Ser Tyr Trp Met HisSer Tyr Trp Met His
1 51 5
<210> 32<210> 32
<211> 17<211> 17
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 32<400> 32
His Ile Asn Pro Ser Ser Gly Tyr Ser Glu Tyr Ser Gln Lys Phe LysHis Ile Asn Pro Ser Ser Gly Tyr Ser Glu Tyr Ser Gln Lys Phe Lys
1 5 10 151 5 10 15
AspAsp
<210> 33<210> 33
<211> 11<211> 11
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 33<400> 33
Tyr Tyr Tyr Val Ser Arg Tyr Val Gly Asp SerTyr Tyr Tyr Val Ser Arg Tyr Val Gly Asp Ser
1 5 101 5 10
<210> 34<210> 34
<211> 7<211> 7
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 34<400> 34
Thr Ser Arg Met Gly Val GlyThr Ser Arg Met Gly Val Gly
1 51 5
<210> 35<210> 35
<211> 16<211> 16
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 35<400> 35
His Ile Trp Trp Asp Asp Asp Lys Arg Tyr Asn Pro Ala Leu Lys SerHis Ile Trp Trp Asp Asp Asp Lys Arg Tyr Asn Pro Ala Leu Lys Ser
1 5 10 151 5 10 15
<210> 36<210> 36
<211> 11<211> 11
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 36<400> 36
Ile Glu Val Gly Arg Gly Ala Trp Phe Ala TyrIle Glu Val Gly Arg Gly Ala Trp Phe Ala Tyr
1 5 101 5 10
<210> 37<210> 37
<211> 5<211> 5
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 37<400> 37
Ser Tyr Trp Ile GlnSer Tyr Trp Ile Gln
1 51 5
<210> 38<210> 38
<211> 17<211> 17
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 38<400> 38
Glu Ile Phe Pro Gly Thr Gly Lys Thr Tyr Tyr Asn Gly Lys Phe LysGlu Ile Phe Pro Gly Thr Gly Lys Thr Tyr Tyr Asn Gly Lys Phe Lys
1 5 10 151 5 10 15
GlyGly
<210> 39<210> 39
<211> 10<211> 10
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 39<400> 39
Ser Pro Tyr Asp Ile Val Gly Phe Asp TyrSer Pro Tyr Asp Ile Val Gly Phe Asp Tyr
1 5 101 5 10
<210> 40<210> 40
<211> 5<211> 5
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 40<400> 40
Ser Tyr Trp Ile GlnSer Tyr Trp Ile Gln
1 51 5
<210> 41<210> 41
<211> 17<211> 17
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 41<400> 41
Glu Ile Phe Pro Gly Thr Gly Lys Thr Tyr Tyr Asn Gly Lys Phe LysGlu Ile Phe Pro Gly Thr Gly Lys Thr Tyr Tyr Asn Gly Lys Phe Lys
1 5 10 151 5 10 15
GlyGly
<210> 42<210> 42
<211> 10<211> 10
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 42<400> 42
Ser Pro Tyr Asp Ile Val Gly Phe Asp TyrSer Pro Tyr Asp Ile Val Gly Phe Asp Tyr
1 5 101 5 10
<210> 43<210> 43
<211> 5<211> 5
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 43<400> 43
Asn Tyr Leu Ile AspAsn Tyr Leu Ile Asp
1 51 5
<210> 44<210> 44
<211> 17<211> 17
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 44<400> 44
Leu Ile Asn Pro Gly Ser Gly Ser Thr Lys Tyr Asn Glu Lys Phe LysLeu Ile Asn Pro Gly Ser Gly Ser Thr Lys Tyr Asn Glu Lys Phe Lys
1 5 10 151 5 10 15
GlyGly
<210> 45<210> 45
<211> 6<211> 6
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 45<400> 45
Thr Gly Thr Leu Pro TyrThr Gly Thr Leu Pro Tyr
1 51 5
<210> 46<210> 46
<211> 5<211> 5
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 46<400> 46
Ser Tyr Trp Ile HisSer Tyr Trp Ile His
1 51 5
<210> 47<210> 47
<211> 14<211> 14
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 47<400> 47
Glu Ile Ser Pro Ser Asn Gly Arg Arg Asn Asn Asn Glu LysGlu Ile Ser Pro Ser Asn Gly Arg Arg Asn Asn Asn Glu Lys
1 5 101 5 10
<210> 48<210> 48
<211> 11<211> 11
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 48<400> 48
Glu Gln Gln Gly Tyr Tyr Glu Gly Phe Ala SerGlu Gln Gln Gly Tyr Tyr Glu Gly Phe Ala Ser
1 5 101 5 10
<210> 49<210> 49
<211> 16<211> 16
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 49<400> 49
Arg Ser Ser Gln Ser Leu Val His Ser Asn Gly Asn Thr Tyr Leu HisArg Ser Ser Gln Ser Leu Val His Ser Asn Gly Asn Thr Tyr Leu His
1 5 10 151 5 10 15
<210> 50<210> 50
<211> 7<211> 7
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 50<400> 50
Lys Val Ser Asn Arg Phe SerLys Val Ser Asn Arg Phe Ser
1 51 5
<210> 51<210> 51
<211> 9<211> 9
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 51<400> 51
Ser Gln Ser Thr His Val Pro Tyr ThrSer Gln Ser Thr His Val Pro Tyr Thr
1 51 5
<210> 52<210> 52
<211> 16<211> 16
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 52<400> 52
Arg Ser Ser Gln Ser Leu Val His Ser Asn Gly Asn Thr Tyr Leu HisArg Ser Ser Gln Ser Leu Val His Ser Asn Gly Asn Thr Tyr Leu His
1 5 10 151 5 10 15
<210> 53<210> 53
<211> 7<211> 7
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 53<400> 53
Lys Val Ser Asn Arg Phe SerLys Val Ser Asn Arg Phe Ser
1 51 5
<210> 54<210> 54
<211> 9<211> 9
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 54<400> 54
Ser Gln Ser Ser His Val Pro Arg ThrSer Gln Ser Ser His Val Pro Arg Thr
1 51 5
<210> 55<210> 55
<211> 17<211> 17
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 55<400> 55
Lys Ser Ser Gln Ser Leu Leu Tyr Ser Thr Asn Gln Lys Asn Tyr LeuLys Ser Ser Gln Ser Leu Leu Tyr Ser Thr Asn Gln Lys Asn Tyr Leu
1 5 10 151 5 10 15
AlaAla
<210> 56<210> 56
<211> 7<211> 7
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 56<400> 56
Trp Ala Ser Thr Arg Asp SerTrp Ala Ser Thr Arg Asp Ser
1 51 5
<210> 57<210> 57
<211> 9<211> 9
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 57<400> 57
Gln Arg Tyr Tyr Asn Tyr Pro Tyr ThrGln Arg Tyr Tyr Asn Tyr Pro Tyr Thr
1 51 5
<210> 58<210> 58
<211> 16<211> 16
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 58<400> 58
Arg Ser Ser Gln Ser Ile Val His Ile Asn Gly Asn Thr Tyr Leu GluArg Ser Ser Gln Ser Ile Val His Ile Asn Gly Asn Thr Tyr Leu Glu
1 5 10 151 5 10 15
<210> 59<210> 59
<211> 7<211> 7
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 59<400> 59
Lys Val Ser Asn Arg Phe SerLys Val Ser Asn Arg Phe Ser
1 51 5
<210> 60<210> 60
<211> 9<211> 9
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 60<400> 60
Phe Gln Gly Ser His Val Pro Arg ThrPhe Gln Gly Ser His Val Pro Arg Thr
1 51 5
<210> 61<210> 61
<211> 11<211> 11
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 61<400> 61
Lys Ala Ser Gln Asp Val Phe Thr Ala Val AlaLys Ala Ser Gln Asp Val Phe Thr Ala Val Ala
1 5 101 5 10
<210> 62<210> 62
<211> 7<211> 7
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 62<400> 62
Ser Ala Ser Tyr Arg Tyr ThrSer Ala Ser Tyr Arg Tyr Thr
1 51 5
<210> 63<210> 63
<211> 9<211> 9
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 63<400> 63
Gln Gln His Tyr Phe Phe Pro Tyr ThrGln Gln His Tyr Phe Phe Pro Tyr Thr
1 51 5
<210> 64<210> 64
<211> 10<211> 10
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 64<400> 64
Ser Ala Ser Ser Ser Val Ser Tyr Met TyrSer Ala Ser Ser Ser Ser Val Ser Tyr Met Tyr
1 5 101 5 10
<210> 65<210> 65
<211> 7<211> 7
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 65<400> 65
Arg Thr Ser Asn Leu Ala SerArg Thr Ser Asn Leu Ala Ser
1 51 5
<210> 66<210> 66
<211> 10<211> 10
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 66<400> 66
Gln Glu Arg Ser Ser Tyr Pro Pro Phe ThrGln Glu Arg Ser Ser Tyr Pro Pro Phe Thr
1 5 101 5 10
<210> 67<210> 67
<211> 11<211> 11
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 67<400> 67
Lys Ala Ser Gln Asp Val Thr Thr Ala Val AlaLys Ala Ser Gln Asp Val Thr Thr Ala Val Ala
1 5 101 5 10
<210> 68<210> 68
<211> 7<211> 7
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 68<400> 68
Ser Ala Ser Ser Arg Tyr ThrSer Ala Ser Ser Ser Arg Tyr Thr
1 51 5
<210> 69<210> 69
<211> 9<211> 9
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 69<400> 69
Gln Gln His Phe Phe Phe Pro Tyr ThrGln Gln His Phe Phe Phe Pro Tyr Thr
1 51 5
<210> 70<210> 70
<211> 16<211> 16
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 70<400> 70
Arg Ser Ser Gln Ser Leu Val His Ser Asn Gly Asn Thr Tyr Leu HisArg Ser Ser Gln Ser Leu Val His Ser Asn Gly Asn Thr Tyr Leu His
1 5 10 151 5 10 15
<210> 71<210> 71
<211> 7<211> 7
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 71<400> 71
Lys Val Ser Asn Arg Phe SerLys Val Ser Asn Arg Phe Ser
1 51 5
<210> 72<210> 72
<211> 9<211> 9
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 72<400> 72
Ser Gln Ser Thr His Val Pro Pro ThrSer Gln Ser Thr His Val Pro Pro Thr
1 51 5
<210> 73<210> 73
<211> 11<211> 11
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 73<400> 73
Arg Ala Ser Gln Asp Ile Ser Asn Tyr Leu AsnArg Ala Ser Gln Asp Ile Ser Asn Tyr Leu Asn
1 5 101 5 10
<210> 74<210> 74
<211> 7<211> 7
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 74<400> 74
Tyr Thr Ser Arg Leu His SerTyr Thr Ser Arg Leu His Ser
1 51 5
<210> 75<210> 75
<211> 9<211> 9
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 75<400> 75
Gln Gln Gly Asn Thr Leu Pro Phe ThrGln Gln Gly Asn Thr Leu Pro Phe Thr
1 51 5
<210> 76<210> 76
<211> 16<211> 16
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 76<400> 76
Arg Ser Ser Gln Ser Leu Val His Ser Asn Gly Asn Thr Tyr Leu HisArg Ser Ser Gln Ser Leu Val His Ser Asn Gly Asn Thr Tyr Leu His
1 5 10 151 5 10 15
<210> 77<210> 77
<211> 7<211> 7
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 77<400> 77
Lys Val Ser Asn Arg Phe SerLys Val Ser Asn Arg Phe Ser
1 51 5
<210> 78<210> 78
<211> 9<211> 9
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 78<400> 78
Ser Gln Ser Thr His Val Pro Pro ThrSer Gln Ser Thr His Val Pro Pro Thr
1 51 5
<210> 79<210> 79
<211> 11<211> 11
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 79<400> 79
Arg Ala Ser Gln Glu Ile Ser Gly Tyr Leu SerArg Ala Ser Gln Glu Ile Ser Gly Tyr Leu Ser
1 5 101 5 10
<210> 80<210> 80
<211> 7<211> 7
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 80<400> 80
Ala Ala Ser Thr Leu Asp SerAla Ala Ser Thr Leu Asp Ser
1 51 5
<210> 81<210> 81
<211> 9<211> 9
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 81<400> 81
Leu Gln Tyr Asp Ser Tyr Pro Leu ThrLeu Gln Tyr Asp Ser Tyr Pro Leu Thr
1 51 5
<210> 82<210> 82
<211> 11<211> 11
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 82<400> 82
Lys Ala Ser Gln Asp Val Val Thr Thr Val AlaLys Ala Ser Gln Asp Val Val Thr Thr Val Ala
1 5 101 5 10
<210> 83<210> 83
<211> 7<211> 7
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 83<400> 83
Ser Ala Ser Tyr Arg Tyr SerSer Ala Ser Tyr Arg Tyr Ser
1 51 5
<210> 84<210> 84
<211> 9<211> 9
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 84<400> 84
Gln Gln Tyr Tyr Thr Tyr Pro Phe ThrGln Gln Tyr Tyr Thr Tyr Pro Phe Thr
1 51 5
<210> 85<210> 85
<211> 16<211> 16
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 85<400> 85
Arg Ser Ser Gln Ser Ile Val His Ile Asn Gly Lys Thr Tyr Leu GluArg Ser Ser Gln Ser Ile Val His Ile Asn Gly Lys Thr Tyr Leu Glu
1 5 10 151 5 10 15
<210> 86<210> 86
<211> 7<211> 7
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 86<400> 86
Lys Ile Ser Asn Arg Phe SerLys Ile Ser Asn Arg Phe Ser
1 51 5
<210> 87<210> 87
<211> 9<211> 9
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 87<400> 87
Phe Gln Gly Ser His Val Pro Arg ThrPhe Gln Gly Ser His Val Pro Arg Thr
1 51 5
<210> 88<210> 88
<211> 16<211> 16
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 88<400> 88
Arg Ser Ser Gln Ser Ile Val His Ile Asn Gly Lys Thr Tyr Leu GluArg Ser Ser Gln Ser Ile Val His Ile Asn Gly Lys Thr Tyr Leu Glu
1 5 10 151 5 10 15
<210> 89<210> 89
<211> 6<211> 6
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 89<400> 89
Lys Ile Ser Asn Arg PheLys Ile Ser Asn Arg Phe
1 51 5
<210> 90<210> 90
<211> 9<211> 9
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 90<400> 90
Phe Gln Gly Ser His Val Pro Arg ThrPhe Gln Gly Ser His Val Pro Arg Thr
1 51 5
<210> 91<210> 91
<211> 11<211> 11
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 91<400> 91
Lys Ala Ser Gln Asn Val Gly Thr Ser Val AlaLys Ala Ser Gln Asn Val Gly Thr Ser Val Ala
1 5 101 5 10
<210> 92<210> 92
<211> 7<211> 7
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 92<400> 92
Ser Thr Ser Tyr Arg Ser SerSer Thr Ser Tyr Arg Ser Ser
1 51 5
<210> 93<210> 93
<211> 9<211> 9
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 93<400> 93
Gln Gln Tyr Asn Ser Tyr Pro Leu ThrGln Gln Tyr Asn Ser Tyr Pro Leu Thr
1 51 5
<210> 94<210> 94
<211> 11<211> 11
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 94<400> 94
Arg Ala Ser Gln Asp Ile Ser Asn Tyr Leu AsnArg Ala Ser Gln Asp Ile Ser Asn Tyr Leu Asn
1 5 101 5 10
<210> 95<210> 95
<211> 7<211> 7
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 95<400> 95
Tyr Thr Ser Arg Leu His SerTyr Thr Ser Arg Leu His Ser
1 51 5
<210> 96<210> 96
<211> 9<211> 9
<212> PRT<212> PRT
<213> Mus musculus<213> Mus musculus
<400> 96<400> 96
Gln Gln Gly Lys Thr Leu Pro Trp ThrGln Gln Gly Lys Thr Leu Pro Trp Thr
1 51 5
<210> 97<210> 97
<211> 122<211> 122
<212> PRT<212> PRT
<213> Synthetic<213> Synthetic
<400> 97<400> 97
Gln Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly AlaGln Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala
1 5 10 151 5 10 15
Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Ser SerSer Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Ser Ser
20 25 30 20 25 30
Trp Met His Trp Ala Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp IleTrp Met His Trp Ala Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Ile
35 40 45 35 40 45
Gly Glu Ile His Pro Asp Ser Gly Lys Thr Asn Tyr Asn Glu Lys PheGly Glu Ile His Pro Asp Ser Gly Lys Thr Asn Tyr Asn Glu Lys Phe
50 55 60 50 55 60
Lys Gly Arg Ala Thr Leu Thr Val Asp Thr Ser Thr Ser Thr Ala TyrLys Gly Arg Ala Thr Leu Thr Val Asp Thr Ser Thr Ser Thr Ala Tyr
65 70 75 8065 70 75 80
Met Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr CysMet Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys
85 90 95 85 90 95
Ala Arg Asp Tyr Leu Ser Ser Pro Tyr Tyr Tyr Val Met Asp Tyr TrpAla Arg Asp Tyr Leu Ser Ser Pro Tyr Tyr Tyr Val Met Asp Tyr Trp
100 105 110 100 105 110
Gly Gln Gly Thr Leu Val Thr Val Ser SerGly Gln Gly Thr Leu Val Thr Val Ser Ser
115 120 115 120
<210> 98<210> 98
<211> 112<211> 112
<212> PRT<212> PRT
<213> Synthetic<213> Synthetic
<400> 98<400> 98
Asp Val Val Met Thr Gln Ser Pro Leu Ser Leu Pro Val Thr Pro GlyAsp Val Val Met Thr Gln Ser Pro Leu Ser Leu Pro Val Thr Pro Gly
1 5 10 151 5 10 15
Glu Pro Ala Ser Ile Ser Cys Arg Ser Ser Gln Ser Leu Val His SerGlu Pro Ala Ser Ile Ser Cys Arg Ser Ser Gln Ser Leu Val His Ser
20 25 30 20 25 30
Gly Gly Asn Thr Tyr Leu His Trp Tyr Leu Gln Lys Pro Gly Gln SerGly Gly Asn Thr Tyr Leu His Trp Tyr Leu Gln Lys Pro Gly Gln Ser
35 40 45 35 40 45
Pro Gln Leu Leu Ile Tyr Lys Val Ser Asn Arg Phe Ser Gly Val ProPro Gln Leu Leu Ile Tyr Lys Val Ser Asn Arg Phe Ser Gly Val Pro
50 55 60 50 55 60
Asp Arg Phe Ser Gly Ser Gly Ser Gly Thr Asp Phe Thr Leu Lys IleAsp Arg Phe Ser Gly Ser Gly Ser Gly Thr Asp Phe Thr Leu Lys Ile
65 70 75 8065 70 75 80
Ser Arg Val Glu Ala Glu Asp Val Gly Val Tyr Phe Cys Ser Gln SerSer Arg Val Glu Ala Glu Asp Val Gly Val Tyr Phe Cys Ser Gln Ser
85 90 95 85 90 95
Thr His Val Pro Tyr Thr Phe Gly Gln Gly Thr Lys Val Glu Ile LysThr His Val Pro Tyr Thr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys
100 105 110 100 105 110
<210> 99<210> 99
<211> 117<211> 117
<212> PRT<212> PRT
<213> Synthetic<213> Synthetic
<400> 99<400> 99
Glu Val Gln Leu Val Glu Ser Gly Gly Gly Leu Val Gln Pro Gly GlyGlu Val Gln Leu Val Glu Ser Gly Gly Gly Leu Val Gln Pro Gly Gly
1 5 10 151 5 10 15
Ser Leu Arg Leu Ser Cys Ala Ala Ser Gly Phe Thr Phe Ser Ser PheSer Leu Arg Leu Ser Cys Ala Ala Ser Gly Phe Thr Phe Ser Ser Ser Phe
20 25 30 20 25 30
Gly Met His Trp Val Arg Gln Ala Pro Gly Lys Gly Leu Glu Trp ValGly Met His Trp Val Arg Gln Ala Pro Gly Lys Gly Leu Glu Trp Val
35 40 45 35 40 45
Ala Tyr Ile Gly Gly Asp Ser Asn Ile Ile Tyr Tyr Ala Asp Thr ValAla Tyr Ile Gly Gly Asp Ser Asn Ile Ile Tyr Tyr Ala Asp Thr Val
50 55 60 50 55 60
Lys Gly Arg Phe Thr Ile Ser Arg Asp Asn Ser Lys Asn Thr Leu TyrLys Gly Arg Phe Thr Ile Ser Arg Asp Asn Ser Lys Asn Thr Leu Tyr
65 70 75 8065 70 75 80
Leu Gln Met Asn Ser Leu Arg Ala Glu Asp Thr Ala Val Tyr Tyr CysLeu Gln Met Asn Ser Leu Arg Ala Glu Asp Thr Ala Val Tyr Tyr Cys
85 90 95 85 90 95
Ala Lys Ser His Asp Tyr Thr Met Asp Tyr Trp Gly Gln Gly Thr LeuAla Lys Ser His Asp Tyr Thr Met Asp Tyr Trp Gly Gln Gly Thr Leu
100 105 110 100 105 110
Val Thr Val Ser SerVal Thr Val Ser Ser
115 115
<210> 100<210> 100
<211> 113<211> 113
<212> PRT<212> PRT
<213> Synthetic<213> Synthetic
<400> 100<400> 100
Asp Ile Val Met Thr Gln Ser Pro Ser Ser Leu Ala Val Ser Leu GlyAsp Ile Val Met Thr Gln Ser Pro Ser Ser Leu Ala Val Ser Leu Gly
1 5 10 151 5 10 15
Glu Arg Val Thr Ile Thr Cys Lys Ser Ser Gln Ser Leu Leu Tyr SerGlu Arg Val Thr Ile Thr Cys Lys Ser Ser Gln Ser Leu Leu Tyr Ser
20 25 30 20 25 30
Thr Asn Gln Lys Asn Tyr Leu Ala Trp Phe Gln Gln Lys Pro Gly GlnThr Asn Gln Lys Asn Tyr Leu Ala Trp Phe Gln Gln Lys Pro Gly Gln
35 40 45 35 40 45
Ser Pro Lys Leu Leu Ile Tyr Trp Ala Ser Thr Arg Glu Ser Gly ValSer Pro Lys Leu Leu Ile Tyr Trp Ala Ser Thr Arg Glu Ser Gly Val
50 55 60 50 55 60
Pro Asp Arg Phe Ser Gly Ser Gly Ser Gly Thr Asp Phe Thr Leu ThrPro Asp Arg Phe Ser Gly Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr
65 70 75 8065 70 75 80
Ile Ser Ser Leu Gln Ala Glu Asp Val Ala Val Tyr Tyr Cys Gln ArgIle Ser Ser Leu Gln Ala Glu Asp Val Ala Val Tyr Tyr Cys Gln Arg
85 90 95 85 90 95
Tyr Tyr Asn Tyr Pro Tyr Thr Phe Gly Gln Gly Thr Lys Val Glu IleTyr Tyr Asn Tyr Pro Tyr Thr Phe Gly Gln Gly Thr Lys Val Glu Ile
100 105 110 100 105 110
LysLys
<210> 101<210> 101
<211> 123<211> 123
<212> PRT<212> PRT
<213> Synthetic<213> Synthetic
<400> 101<400> 101
Gln Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly AlaGln Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala
1 5 10 151 5 10 15
Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Lys Phe Thr Lys TyrSer Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Lys Phe Thr Lys Tyr
20 25 30 20 25 30
Trp Ile Glu Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp IleTrp Ile Glu Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Ile
35 40 45 35 40 45
Gly Glu Ile Ser Pro Gly Ile Asp Lys Thr Asn Tyr Asn Glu Lys PheGly Glu Ile Ser Pro Gly Ile Asp Lys Thr Asn Tyr Asn Glu Lys Phe
50 55 60 50 55 60
Lys Asp Arg Ala Thr Phe Thr Ala Asp Thr Ser Thr Asn Thr Ala TyrLys Asp Arg Ala Thr Phe Thr Ala Asp Thr Ser Thr Asn Thr Ala Tyr
65 70 75 8065 70 75 80
Met Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr CysMet Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys
85 90 95 85 90 95
Ala Arg Gly Glu Glu Tyr Gly Asn Ser Val Arg Tyr Ala Met Asp TyrAla Arg Gly Glu Glu Tyr Gly Asn Ser Val Arg Tyr Ala Met Asp Tyr
100 105 110 100 105 110
Trp Gly Gln Gly Thr Leu Val Thr Val Ser SerTrp Gly Gln Gly Thr Leu Val Thr Val Ser Ser
115 120 115 120
<210> 102<210> 102
<211> 107<211> 107
<212> PRT<212> PRT
<213> Synthetic<213> Synthetic
<400> 102<400> 102
Glu Ile Val Leu Thr Gln Ser Pro Ala Thr Leu Ser Leu Ser Pro GlyGlu Ile Val Leu Thr Gln Ser Pro Ala Thr Leu Ser Leu Ser Pro Gly
1 5 10 151 5 10 15
Glu Arg Ala Thr Leu Ser Cys Ser Ala Ser Ser Ser Val Ser Tyr MetGlu Arg Ala Thr Leu Ser Cys Ser Ala Ser Ser Ser Val Ser Tyr Met
20 25 30 20 25 30
Tyr Trp Leu Gln Gln Lys Pro Gly Gln Ser Pro Arg Leu Leu Ile TyrTyr Trp Leu Gln Gln Lys Pro Gly Gln Ser Pro Arg Leu Leu Ile Tyr
35 40 45 35 40 45
Arg Thr Ser Asn Leu Ala Ser Gly Val Pro Ala Arg Phe Ser Gly SerArg Thr Ser Asn Leu Ala Ser Gly Val Pro Ala Arg Phe Ser Gly Ser
50 55 60 50 55 60
Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Glu Pro GluGly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Glu Pro Glu
65 70 75 8065 70 75 80
Asp Phe Ala Val Tyr Tyr Cys Gln Glu Arg Ser Ser Tyr Pro Pro PheAsp Phe Ala Val Tyr Tyr Cys Gln Glu Arg Ser Ser Tyr Pro Pro Phe
85 90 95 85 90 95
Thr Phe Gly Gln Gly Thr Lys Val Glu Ile LysThr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys
100 105 100 105
<210> 103<210> 103
<211> 119<211> 119
<212> PRT<212> PRT
<213> Synthetic<213> Synthetic
<400> 103<400> 103
Gln Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly AlaGln Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala
1 5 10 151 5 10 15
Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Gly TyrSer Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Gly Tyr
20 25 30 20 25 30
Asn Met Asn Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp IleAsn Met Asn Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Ile
35 40 45 35 40 45
Gly Asn Ile Asp Pro Tyr Phe Gly Gly Thr Ile Tyr Asn Gln Lys PheGly Asn Ile Asp Pro Tyr Phe Gly Gly Thr Ile Tyr Asn Gln Lys Phe
50 55 60 50 55 60
Lys Gly Arg Val Thr Leu Thr Arg Asp Thr Ser Thr Ser Thr Ala TyrLys Gly Arg Val Thr Leu Thr Arg Asp Thr Ser Thr Ser Thr Ala Tyr
65 70 75 8065 70 75 80
Met Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr CysMet Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys
85 90 95 85 90 95
Ala Arg Gln Tyr Gly Asn Trp Val Gly Phe Ala Tyr Trp Gly Gln GlyAla Arg Gln Tyr Gly Asn Trp Val Gly Phe Ala Tyr Trp Gly Gln Gly
100 105 110 100 105 110
Thr Leu Val Thr Val Ser SerThr Leu Val Thr Val Ser Ser
115 115
<210> 104<210> 104
<211> 107<211> 107
<212> PRT<212> PRT
<213> Synthetic<213> Synthetic
<400> 104<400> 104
Asp Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val GlyAsp Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly
1 5 10 151 5 10 15
Asp Arg Val Thr Ile Thr Cys Lys Ala Ser Gln Asp Val Thr Thr AlaAsp Arg Val Thr Ile Thr Cys Lys Ala Ser Gln Asp Val Thr Thr Ala
20 25 30 20 25 30
Val Ala Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu IleVal Ala Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile
35 40 45 35 40 45
Tyr Ser Ala Ser Ser Arg Tyr Thr Gly Val Pro Ser Arg Phe Ser GlyTyr Ser Ala Ser Ser Arg Tyr Thr Gly Val Pro Ser Arg Phe Ser Gly
50 55 60 50 55 60
Ser Gly Ser Gly Thr Asp Phe Thr Phe Thr Ile Ser Ser Leu Gln ProSer Gly Ser Gly Thr Asp Phe Thr Phe Thr Ile Ser Ser Leu Gln Pro
65 70 75 8065 70 75 80
Glu Asp Ile Ala Thr Tyr Tyr Cys Gln Gln His Phe Phe Phe Pro TyrGlu Asp Ile Ala Thr Tyr Tyr Cys Gln Gln His Phe Phe Phe Pro Tyr
85 90 95 85 90 95
Thr Phe Gly Gln Gly Thr Lys Val Glu Ile LysThr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys
100 105 100 105
<210> 105<210> 105
<211> 121<211> 121
<212> PRT<212> PRT
<213> Synthetic<213> Synthetic
<400> 105<400> 105
Asp Val Gln Leu Gln Glu Ser Gly Pro Gly Leu Val Lys Pro Ser GlnAsp Val Gln Leu Gln Glu Ser Gly Pro Gly Leu Val Lys Pro Ser Gln
1 5 10 151 5 10 15
Thr Leu Ser Leu Thr Cys Thr Val Ser Gly Tyr Ser Ile Ser Ser GlyThr Leu Ser Leu Thr Cys Thr Val Ser Gly Tyr Ser Ile Ser Ser Ser Gly
20 25 30 20 25 30
Tyr Tyr Trp Asn Trp Phe Arg Gln Pro Pro Gly Lys Lys Leu Glu TrpTyr Tyr Trp Asn Trp Phe Arg Gln Pro Pro Gly Lys Lys Leu Glu Trp
35 40 45 35 40 45
Ile Gly Tyr Arg Ser Tyr Asp Gly Ser Tyr Asn Tyr Asn Pro Ser LeuIle Gly Tyr Arg Ser Tyr Asp Gly Ser Tyr Asn Tyr Asn Pro Ser Leu
50 55 60 50 55 60
Glu Asn Arg Ile Thr Ile Ser Arg Asp Thr Ser Lys Asn Gln Phe SerGlu Asn Arg Ile Thr Ile Ser Arg Asp Thr Ser Lys Asn Gln Phe Ser
65 70 75 8065 70 75 80
Leu Lys Leu Ser Ser Val Thr Ala Ala Asp Thr Ala Val Tyr Tyr CysLeu Lys Leu Ser Ser Val Thr Ala Ala Asp Thr Ala Val Tyr Tyr Cys
85 90 95 85 90 95
Ala Arg Glu Glu Arg Tyr Ile Leu Arg Tyr Ala Met Asp Tyr Trp GlyAla Arg Glu Glu Arg Tyr Ile Leu Arg Tyr Ala Met Asp Tyr Trp Gly
100 105 110 100 105 110
Gln Gly Thr Leu Val Thr Val Ser SerGln Gly Thr Leu Val Thr Val Ser Ser
115 120 115 120
<210> 106<210> 106
<211> 107<211> 107
<212> PRT<212> PRT
<213> Synthetic<213> Synthetic
<400> 106<400> 106
Asp Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val GlyAsp Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly
1 5 10 151 5 10 15
Asp Arg Val Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile Ser Asn TyrAsp Arg Val Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile Ser Asn Tyr
20 25 30 20 25 30
Leu Asn Trp Tyr Gln Gln Lys Pro Gly Lys Thr Val Lys Leu Leu IleLeu Asn Trp Tyr Gln Gln Lys Pro Gly Lys Thr Val Lys Leu Leu Ile
35 40 45 35 40 45
Tyr Tyr Thr Ser Arg Leu His Ser Gly Val Pro Ser Arg Phe Ser GlyTyr Tyr Thr Ser Arg Leu His Ser Gly Val Pro Ser Arg Phe Ser Gly
50 55 60 50 55 60
Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln ProSer Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro
65 70 75 8065 70 75 80
Glu Asp Phe Ala Thr Tyr Phe Cys Gln Gln Gly Asn Thr Leu Pro PheGlu Asp Phe Ala Thr Tyr Phe Cys Gln Gln Gly Asn Thr Leu Pro Phe
85 90 95 85 90 95
Thr Phe Gly Gln Gly Thr Lys Val Glu Ile LysThr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys
100 105 100 105
<210> 107<210> 107
<211> 120<211> 120
<212> PRT<212> PRT
<213> Synthetic<213> Synthetic
<400> 107<400> 107
Gln Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly AlaGln Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala
1 5 10 151 5 10 15
Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Ser TyrSer Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Ser Tyr
20 25 30 20 25 30
Trp Met His Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp IleTrp Met His Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Ile
35 40 45 35 40 45
Gly His Ile Asn Pro Ser Ser Gly Tyr Ser Glu Tyr Ser Gln Lys PheGly His Ile Asn Pro Ser Ser Gly Tyr Ser Glu Tyr Ser Gln Lys Phe
50 55 60 50 55 60
Lys Asp Arg Ala Thr Leu Thr Ala Asp Lys Ser Thr Ser Thr Ala TyrLys Asp Arg Ala Thr Leu Thr Ala Asp Lys Ser Thr Ser Thr Ala Tyr
65 70 75 8065 70 75 80
Met Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr CysMet Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys
85 90 95 85 90 95
Glu Ile Tyr Tyr Tyr Val Ser Arg Tyr Val Gly Asp Ser Trp Gly GlnGlu Ile Tyr Tyr Tyr Val Ser Arg Tyr Val Gly Asp Ser Trp Gly Gln
100 105 110 100 105 110
Gly Thr Leu Val Thr Val Ser SerGly Thr Leu Val Thr Val Ser Ser
115 120 115 120
<210> 108<210> 108
<211> 107<211> 107
<212> PRT<212> PRT
<213> Synthetic<213> Synthetic
<400> 108<400> 108
Asp Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val GlyAsp Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly
1 5 10 151 5 10 15
Asp Arg Val Thr Ile Thr Cys Arg Ala Ser Gln Glu Ile Ser Gly TyrAsp Arg Val Thr Ile Thr Cys Arg Ala Ser Gln Glu Ile Ser Gly Tyr
20 25 30 20 25 30
Leu Ser Trp Leu Gln Gln Lys Pro Gly Lys Thr Ile Lys Arg Leu IleLeu Ser Trp Leu Gln Gln Lys Pro Gly Lys Thr Ile Lys Arg Leu Ile
35 40 45 35 40 45
Tyr Ala Ala Ser Thr Leu Asp Ser Gly Val Pro Ser Arg Phe Ser GlyTyr Ala Ala Ser Thr Leu Asp Ser Gly Val Pro Ser Arg Phe Ser Gly
50 55 60 50 55 60
Ser Arg Ser Gly Ser Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln ProSer Arg Ser Gly Ser Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro
65 70 75 8065 70 75 80
Glu Asp Phe Ala Thr Tyr Tyr Cys Leu Gln Tyr Asp Ser Tyr Pro LeuGlu Asp Phe Ala Thr Tyr Tyr Cys Leu Gln Tyr Asp Ser Tyr Pro Leu
85 90 95 85 90 95
Thr Phe Gly Gln Gly Thr Lys Val Glu Ile LysThr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys
100 105 100 105
<210> 109<210> 109
<211> 121<211> 121
<212> PRT<212> PRT
<213> Synthetic<213> Synthetic
<400> 109<400> 109
Gln Val Thr Leu Lys Glu Ser Gly Pro Ala Leu Val Lys Pro Thr GlnGln Val Thr Leu Lys Glu Ser Gly Pro Ala Leu Val Lys Pro Thr Gln
1 5 10 151 5 10 15
Thr Leu Thr Leu Thr Cys Thr Leu Ser Gly Phe Ser Leu Ser Thr SerThr Leu Thr Leu Thr Cys Thr Leu Ser Gly Phe Ser Leu Ser Thr Ser
20 25 30 20 25 30
Arg Met Gly Val Gly Trp Ile Arg Gln Pro Pro Gly Lys Gly Leu GluArg Met Gly Val Gly Trp Ile Arg Gln Pro Pro Gly Lys Gly Leu Glu
35 40 45 35 40 45
Trp Leu Ala His Ile Trp Trp Asp Asp Asp Lys Arg Tyr Asn Pro AlaTrp Leu Ala His Ile Trp Trp Asp Asp Asp Lys Arg Tyr Asn Pro Ala
50 55 60 50 55 60
Leu Lys Ser Arg Leu Thr Ile Ser Lys Asp Thr Ser Ser Asn Gln ValLeu Lys Ser Arg Leu Thr Ile Ser Lys Asp Thr Ser Ser Asn Gln Val
65 70 75 8065 70 75 80
Phe Leu Lys Ile Ala Ser Val Asp Lys Glu Asp Thr Ala Thr Tyr TyrPhe Leu Lys Ile Ala Ser Val Asp Lys Glu Asp Thr Ala Thr Tyr Tyr
85 90 95 85 90 95
Cys Gly Arg Ile Glu Val Gly Arg Gly Ala Trp Phe Ala Tyr Trp GlyCys Gly Arg Ile Glu Val Gly Arg Gly Ala Trp Phe Ala Tyr Trp Gly
100 105 110 100 105 110
Gln Gly Thr Leu Val Thr Val Ser SerGln Gly Thr Leu Val Thr Val Ser Ser
115 120 115 120
<210> 110<210> 110
<211> 107<211> 107
<212> PRT<212> PRT
<213> Synthetic<213> Synthetic
<400> 110<400> 110
Glu Ile Val Leu Thr Gln Ser Pro Ala Thr Leu Ser Leu Ser Pro GlyGlu Ile Val Leu Thr Gln Ser Pro Ala Thr Leu Ser Leu Ser Pro Gly
1 5 10 151 5 10 15
Glu Arg Val Ser Val Thr Cys Lys Ala Ser Gln Asp Val Val Thr ThrGlu Arg Val Ser Val Thr Cys Lys Ala Ser Gln Asp Val Val Thr Thr
20 25 30 20 25 30
Val Ala Trp Tyr Gln Gln Lys Pro Gly Gln Ala Pro Arg Thr Leu IleVal Ala Trp Tyr Gln Gln Lys Pro Gly Gln Ala Pro Arg Thr Leu Ile
35 40 45 35 40 45
Tyr Ser Ala Ser Tyr Arg Tyr Ser Gly Val Pro Ala Arg Phe Ser GlyTyr Ser Ala Ser Tyr Arg Tyr Ser Gly Val Pro Ala Arg Phe Ser Gly
50 55 60 50 55 60
Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Glu ProSer Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Glu Pro
65 70 75 8065 70 75 80
Glu Asp Phe Ala Val Tyr Tyr Cys Gln Gln Tyr Tyr Thr Tyr Pro PheGlu Asp Phe Ala Val Tyr Tyr Cys Gln Gln Tyr Tyr Thr Tyr Pro Phe
85 90 95 85 90 95
Thr Phe Gly Gln Gly Thr Lys Val Glu Ile LysThr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys
100 105 100 105
<210> 111<210> 111
<211> 115<211> 115
<212> PRT<212> PRT
<213> Synthetic<213> Synthetic
<400> 111<400> 111
Gln Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly AlaGln Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala
1 5 10 151 5 10 15
Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Asn TyrSer Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Asn Tyr
20 25 30 20 25 30
Leu Ile Asp Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp IleLeu Ile Asp Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Ile
35 40 45 35 40 45
Gly Leu Ile Asn Pro Gly Ser Gly Ser Thr Lys Tyr Asn Glu Lys PheGly Leu Ile Asn Pro Gly Ser Gly Ser Thr Lys Tyr Asn Glu Lys Phe
50 55 60 50 55 60
Lys Gly Arg Val Thr Leu Thr Ala Asp Lys Ser Thr Ser Thr Ala TyrLys Gly Arg Val Thr Leu Thr Ala Asp Lys Ser Thr Ser Thr Ala Tyr
65 70 75 8065 70 75 80
Met Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr CysMet Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys
85 90 95 85 90 95
Ala Lys Thr Gly Thr Leu Pro Tyr Trp Gly Gln Gly Thr Leu Val ThrAla Lys Thr Gly Thr Leu Pro Tyr Trp Gly Gln Gly Thr Leu Val Thr
100 105 110 100 105 110
Val Ser SerVal Ser Ser
115 115
<210> 112<210> 112
<211> 107<211> 107
<212> PRT<212> PRT
<213> Synthetic<213> Synthetic
<400> 112<400> 112
Asp Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val GlyAsp Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly
1 5 10 151 5 10 15
Asp Arg Val Thr Ile Thr Cys Lys Ala Ser Gln Asn Val Gly Thr SerAsp Arg Val Thr Ile Thr Cys Lys Ala Ser Gln Asn Val Gly Thr Ser
20 25 30 20 25 30
Val Ala Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu IleVal Ala Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile
35 40 45 35 40 45
Tyr Ser Thr Ser Tyr Arg Ser Ser Gly Val Pro Ser Arg Phe Ser GlyTyr Ser Thr Ser Tyr Arg Ser Ser Gly Val Pro Ser Arg Phe Ser Gly
50 55 60 50 55 60
Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln ProSer Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro
65 70 75 8065 70 75 80
Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Tyr Gly Ser Tyr Pro LeuGlu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Tyr Gly Ser Tyr Pro Leu
85 90 95 85 90 95
Thr Phe Gly Gln Gly Thr Lys Val Glu Ile LysThr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys
100 105 100 105
<210> 113<210> 113
<211> 330<211> 330
<212> PRT<212> PRT
<213> Homo sapiens<213> Homo sapiens
<400> 113<400> 113
Ala Ser Thr Lys Gly Pro Ser Val Phe Pro Leu Ala Pro Ser Ser LysAla Ser Thr Lys Gly Pro Ser Val Phe Pro Leu Ala Pro Ser Ser Ser Lys
1 5 10 151 5 10 15
Ser Thr Ser Gly Gly Thr Ala Ala Leu Gly Cys Leu Val Lys Asp TyrSer Thr Ser Gly Gly Thr Ala Ala Leu Gly Cys Leu Val Lys Asp Tyr
20 25 30 20 25 30
Phe Pro Glu Pro Val Thr Val Ser Trp Asn Ser Gly Ala Leu Thr SerPhe Pro Glu Pro Val Thr Val Ser Trp Asn Ser Gly Ala Leu Thr Ser
35 40 45 35 40 45
Gly Val His Thr Phe Pro Ala Val Leu Gln Ser Ser Gly Leu Tyr SerGly Val His Thr Phe Pro Ala Val Leu Gln Ser Ser Gly Leu Tyr Ser
50 55 60 50 55 60
Leu Ser Ser Val Val Thr Val Pro Ser Ser Ser Leu Gly Thr Gln ThrLeu Ser Ser Val Val Thr Val Pro Ser Ser Ser Leu Gly Thr Gln Thr
65 70 75 8065 70 75 80
Tyr Ile Cys Asn Val Asn His Lys Pro Ser Asn Thr Lys Val Asp LysTyr Ile Cys Asn Val Asn His Lys Pro Ser Asn Thr Lys Val Asp Lys
85 90 95 85 90 95
Lys Val Glu Pro Lys Ser Cys Asp Lys Thr His Thr Cys Pro Pro CysLys Val Glu Pro Lys Ser Cys Asp Lys Thr His Thr Cys Pro Pro Cys
100 105 110 100 105 110
Pro Ala Pro Glu Leu Leu Gly Gly Pro Ser Val Phe Leu Phe Pro ProPro Ala Pro Glu Leu Leu Gly Gly Pro Ser Val Phe Leu Phe Pro Pro
115 120 125 115 120 125
Lys Pro Lys Asp Thr Leu Met Ile Ser Arg Thr Pro Glu Val Thr CysLys Pro Lys Asp Thr Leu Met Ile Ser Arg Thr Pro Glu Val Thr Cys
130 135 140 130 135 140
Val Val Val Asp Val Ser His Glu Asp Pro Glu Val Lys Phe Asn TrpVal Val Val Asp Val Ser His Glu Asp Pro Glu Val Lys Phe Asn Trp
145 150 155 160145 150 155 160
Tyr Val Asp Gly Val Glu Val His Asn Ala Lys Thr Lys Pro Arg GluTyr Val Asp Gly Val Glu Val His Asn Ala Lys Thr Lys Pro Arg Glu
165 170 175 165 170 175
Glu Gln Tyr Asn Ser Thr Tyr Arg Val Val Ser Val Leu Thr Val LeuGlu Gln Tyr Asn Ser Thr Tyr Arg Val Val Ser Val Leu Thr Val Leu
180 185 190 180 185 190
His Gln Asp Trp Leu Asn Gly Lys Glu Tyr Lys Cys Lys Val Ser AsnHis Gln Asp Trp Leu Asn Gly Lys Glu Tyr Lys Cys Lys Val Ser Asn
195 200 205 195 200 205
Lys Ala Leu Pro Ala Pro Ile Glu Lys Thr Ile Ser Lys Ala Lys GlyLys Ala Leu Pro Ala Pro Ile Glu Lys Thr Ile Ser Lys Ala Lys Gly
210 215 220 210 215 220
Gln Pro Arg Glu Pro Gln Val Tyr Thr Leu Pro Pro Ser Arg Asp GluGln Pro Arg Glu Pro Gln Val Tyr Thr Leu Pro Pro Ser Arg Asp Glu
225 230 235 240225 230 235 240
Leu Thr Lys Asn Gln Val Ser Leu Thr Cys Leu Val Lys Gly Phe TyrLeu Thr Lys Asn Gln Val Ser Leu Thr Cys Leu Val Lys Gly Phe Tyr
245 250 255 245 250 255
Pro Ser Asp Ile Ala Val Glu Trp Glu Ser Asn Gly Gln Pro Glu AsnPro Ser Asp Ile Ala Val Glu Trp Glu Ser Asn Gly Gln Pro Glu Asn
260 265 270 260 265 270
Asn Tyr Lys Thr Thr Pro Pro Val Leu Asp Ser Asp Gly Ser Phe PheAsn Tyr Lys Thr Thr Pro Pro Val Leu Asp Ser Asp Gly Ser Phe Phe
275 280 285 275 280 285
Leu Tyr Ser Lys Leu Thr Val Asp Lys Ser Arg Trp Gln Gln Gly AsnLeu Tyr Ser Lys Leu Thr Val Asp Lys Ser Arg Trp Gln Gln Gly Asn
290 295 300 290 295 300
Val Phe Ser Cys Ser Val Met His Glu Ala Leu His Asn His Tyr ThrVal Phe Ser Cys Ser Val Met His Glu Ala Leu His Asn His Tyr Thr
305 310 315 320305 310 315 320
Gln Lys Ser Leu Ser Leu Ser Pro Gly LysGln Lys Ser Leu Ser Leu Ser Pro Gly Lys
325 330 325 330
<210> 114<210> 114
<211> 107<211> 107
<212> PRT<212> PRT
<213> Homo sapiens<213> Homo sapiens
<400> 114<400> 114
Arg Thr Val Ala Ala Pro Ser Val Phe Ile Phe Pro Pro Ser Asp GluArg Thr Val Ala Ala Pro Ser Val Phe Ile Phe Pro Pro Ser Asp Glu
1 5 10 151 5 10 15
Gln Leu Lys Ser Gly Thr Ala Ser Val Val Cys Leu Leu Asn Asn PheGln Leu Lys Ser Gly Thr Ala Ser Val Val Cys Leu Leu Asn Asn Phe
20 25 30 20 25 30
Tyr Pro Arg Glu Ala Lys Val Gln Trp Lys Val Asp Asn Ala Leu GlnTyr Pro Arg Glu Ala Lys Val Gln Trp Lys Val Asp Asn Ala Leu Gln
35 40 45 35 40 45
Ser Gly Asn Ser Gln Glu Ser Val Thr Glu Gln Asp Ser Lys Asp SerSer Gly Asn Ser Gln Glu Ser Val Thr Glu Gln Asp Ser Lys Asp Ser
50 55 60 50 55 60
Thr Tyr Ser Leu Ser Ser Thr Leu Thr Leu Ser Lys Ala Asp Tyr GluThr Tyr Ser Leu Ser Ser Thr Leu Thr Leu Ser Lys Ala Asp Tyr Glu
65 70 75 8065 70 75 80
Lys His Lys Val Tyr Ala Cys Glu Val Thr His Gln Gly Leu Ser SerLys His Lys Val Tyr Ala Cys Glu Val Thr His Gln Gly Leu Ser Ser
85 90 95 85 90 95
Pro Val Thr Lys Ser Phe Asn Arg Gly Glu CysPro Val Thr Lys Ser Phe Asn Arg Gly Glu Cys
100 105 100 105
Claims (13)
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202011221844.0A CN114437215B (en) | 2020-11-05 | 2020-11-05 | Anti-human CD38 antibody and preparation method and application thereof |
| PCT/CN2021/124806 WO2022095698A1 (en) | 2020-11-05 | 2021-10-20 | Anti-human cd38 antibody, preparation method therefor and use thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202011221844.0A CN114437215B (en) | 2020-11-05 | 2020-11-05 | Anti-human CD38 antibody and preparation method and application thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN114437215A CN114437215A (en) | 2022-05-06 |
| CN114437215B true CN114437215B (en) | 2023-02-07 |
Family
ID=81360624
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202011221844.0A Active CN114437215B (en) | 2020-11-05 | 2020-11-05 | Anti-human CD38 antibody and preparation method and application thereof |
Country Status (2)
| Country | Link |
|---|---|
| CN (1) | CN114437215B (en) |
| WO (1) | WO2022095698A1 (en) |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU2016228249A1 (en) * | 2006-09-26 | 2016-10-06 | Genmab A/S | Combination treatment of CD38-expressing tumors |
| CN108409860A (en) * | 2017-02-10 | 2018-08-17 | 上海麦济生物技术有限公司 | Anti-human Nuvance alpha monoclonal antibodies, preparation method and application |
| CN108752475A (en) * | 2018-06-14 | 2018-11-06 | 北京智仁美博生物科技有限公司 | Anti-human CD38 antibody and application thereof |
| CN109053892A (en) * | 2018-09-19 | 2018-12-21 | 苏州思坦维生物技术股份有限公司 | The monoclonal antibody and the preparation method and application thereof of specific bond people and monkey CD38 antigen |
| CN109232739A (en) * | 2017-07-11 | 2019-01-18 | 北京大学深圳研究生院 | A kind of 8 nano antibody of AntiCD3 McAb, encoding gene and application |
| CN110997722A (en) * | 2017-06-08 | 2020-04-10 | 黑带医疗有限公司 | CD38 regulatory antibody |
| CN111051344A (en) * | 2017-06-08 | 2020-04-21 | 黑带医疗有限公司 | CD38 modulating antibodies |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| MXPA06008700A (en) * | 2004-02-06 | 2007-01-19 | Morphosys Ag | Anti-cd38 human antibodies and uses therefor. |
| US20090076249A1 (en) * | 2007-09-19 | 2009-03-19 | Michel De Weers | Antibodies against CD38 for treatment of multiple myeloma |
| KR102770107B1 (en) * | 2017-08-16 | 2025-02-20 | 블랙 벨트 테라퓨틱스 리미티드 | CD38 regulatory antibody |
| JP7299873B2 (en) * | 2017-08-16 | 2023-06-28 | ブラック ベルト セラピューティクス リミテッド | CD38 antibody |
-
2020
- 2020-11-05 CN CN202011221844.0A patent/CN114437215B/en active Active
-
2021
- 2021-10-20 WO PCT/CN2021/124806 patent/WO2022095698A1/en not_active Ceased
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU2016228249A1 (en) * | 2006-09-26 | 2016-10-06 | Genmab A/S | Combination treatment of CD38-expressing tumors |
| CN108409860A (en) * | 2017-02-10 | 2018-08-17 | 上海麦济生物技术有限公司 | Anti-human Nuvance alpha monoclonal antibodies, preparation method and application |
| CN110997722A (en) * | 2017-06-08 | 2020-04-10 | 黑带医疗有限公司 | CD38 regulatory antibody |
| CN111051344A (en) * | 2017-06-08 | 2020-04-21 | 黑带医疗有限公司 | CD38 modulating antibodies |
| CN109232739A (en) * | 2017-07-11 | 2019-01-18 | 北京大学深圳研究生院 | A kind of 8 nano antibody of AntiCD3 McAb, encoding gene and application |
| CN108752475A (en) * | 2018-06-14 | 2018-11-06 | 北京智仁美博生物科技有限公司 | Anti-human CD38 antibody and application thereof |
| CN109053892A (en) * | 2018-09-19 | 2018-12-21 | 苏州思坦维生物技术股份有限公司 | The monoclonal antibody and the preparation method and application thereof of specific bond people and monkey CD38 antigen |
Non-Patent Citations (2)
| Title |
|---|
| Anti-CD38 Antibody Therapy: Windows of Opportunity Yielded by the Functional Characteristics of the Target Molecule;Antonella Chillemi等;《Molecular Medicine》;springer;20131231;第19卷;第99-108页 * |
| 抗人CD38单克隆抗体抗原识别表位的初步研究;问新宇等;《细胞与分子免疫学杂志》;万方;20050602(第3期);第340-341页 * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN114437215A (en) | 2022-05-06 |
| WO2022095698A1 (en) | 2022-05-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN107922503B (en) | A kind of PDL-1 antibody, its pharmaceutical composition and its use | |
| CN112969714B (en) | anti-CD 40 antibodies, antigen binding fragments thereof and medical uses thereof | |
| IL262892B2 (en) | Anti-PD-1 and anti-LAG3 antibodies for cancer treatment | |
| CN114437212B (en) | Anti-human thymic stromal lymphopoietin antibody and preparation method and application thereof | |
| CN114907479B (en) | Anti-CD112R antibodies and uses thereof | |
| US20220017618A1 (en) | Use of tim-3 antibody in preparation of medicines for treating tumors | |
| US20230357398A1 (en) | Novel human antibodies binding to human cd3 epsilon | |
| WO2019076277A1 (en) | Uses of anti-pd-1 antibody and anti-lag-3 antibody jointly in preparing medicament for treating tumor | |
| WO2023241656A1 (en) | Bispecific antibody containing anti-cldn18.2 antibody, and pharmaceutical composition and use thereof | |
| CN111818972A (en) | Deimmunized anti-ERBB3 antibody | |
| CN117355540A (en) | Anti-CD137 antibodies and methods of use | |
| CN112480252B (en) | Anti-interleukin-33 antibody and preparation method and application thereof | |
| TW202112813A (en) | Binding molecules specific for lif and uses thereof | |
| WO2024184812A1 (en) | Anti-cldn6 antibodies and methods of use | |
| CN114437215B (en) | Anti-human CD38 antibody and preparation method and application thereof | |
| CN115215937B (en) | Anti-human MASP-2 antibody, preparation method and application thereof | |
| WO2022100694A1 (en) | Antibody and preparation method therefor | |
| CN114075283A (en) | Antibodies that bind to human CD38, methods of making and uses thereof | |
| WO2024088342A1 (en) | Antibodies against cd24 and uses thereof | |
| TWI894360B (en) | Anti-TIGIT antibodies and bispecific antibodies and their applications | |
| US20240301051A1 (en) | Anti-cldn6 antibodies and methods of use | |
| US20240301061A1 (en) | Anti-cldn6 and anti-cd3 multispecific antibodies and methods of use | |
| WO2024184810A1 (en) | Anti-cldn6 and anti-cd3 multispecific antibodies and methods of use | |
| HK40101629A (en) | Bispecific antibody containing anti-cldn18.2 antibody, and pharmaceutical composition and use thereof | |
| HK40089230A (en) | Pharmaceutical composition and use |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant | ||
| CP03 | Change of name, title or address |
Address after: Room 1001-7, Building 2, Xincheng Science and Technology Park, 588 Yuelu West Avenue, Lugu Street, Xiangjiang New District, Changsha City, Hunan Province 410205 Patentee after: Hunan Maiji Biotechnology Co.,Ltd. Country or region after: China Address before: Room 304, No. 1011 Harley Road, Pudong New Area, Shanghai Patentee before: SHANGHAI MABGEEK BIOTECHNOLOGY CO.,LTD. Country or region before: China |
|
| CP03 | Change of name, title or address |