CN114249786A - Preparation and application of nucleoside intermediate containing N, N-diacyl structure - Google Patents
Preparation and application of nucleoside intermediate containing N, N-diacyl structure Download PDFInfo
- Publication number
- CN114249786A CN114249786A CN202111646993.6A CN202111646993A CN114249786A CN 114249786 A CN114249786 A CN 114249786A CN 202111646993 A CN202111646993 A CN 202111646993A CN 114249786 A CN114249786 A CN 114249786A
- Authority
- CN
- China
- Prior art keywords
- guanosine
- diacyl
- structure according
- reagent
- diacyl structure
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 229910052757 nitrogen Inorganic materials 0.000 title claims abstract description 28
- 239000002777 nucleoside Substances 0.000 title claims abstract description 18
- 150000003833 nucleoside derivatives Chemical class 0.000 title abstract description 11
- 238000002360 preparation method Methods 0.000 title abstract description 6
- NYHBQMYGNKIUIF-UUOKFMHZSA-N Guanosine Chemical compound C1=NC=2C(=O)NC(N)=NC=2N1[C@@H]1O[C@H](CO)[C@@H](O)[C@H]1O NYHBQMYGNKIUIF-UUOKFMHZSA-N 0.000 claims abstract description 64
- MIKUYHXYGGJMLM-GIMIYPNGSA-N Crotonoside Natural products C1=NC2=C(N)NC(=O)N=C2N1[C@H]1O[C@@H](CO)[C@H](O)[C@@H]1O MIKUYHXYGGJMLM-GIMIYPNGSA-N 0.000 claims abstract description 32
- NYHBQMYGNKIUIF-UHFFFAOYSA-N D-guanosine Natural products C1=2NC(N)=NC(=O)C=2N=CN1C1OC(CO)C(O)C1O NYHBQMYGNKIUIF-UHFFFAOYSA-N 0.000 claims abstract description 32
- 229940029575 guanosine Drugs 0.000 claims abstract description 32
- 239000003153 chemical reaction reagent Substances 0.000 claims abstract description 17
- 238000000034 method Methods 0.000 claims abstract description 14
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 claims abstract description 12
- PASDCCFISLVPSO-UHFFFAOYSA-N benzoyl chloride Chemical compound ClC(=O)C1=CC=CC=C1 PASDCCFISLVPSO-UHFFFAOYSA-N 0.000 claims abstract description 9
- 229910052710 silicon Inorganic materials 0.000 claims abstract description 9
- 239000010703 silicon Substances 0.000 claims abstract description 9
- QWXYZCJEXYQNEI-OSZHWHEXSA-N intermediate I Chemical compound COC(=O)[C@@]1(C=O)[C@H]2CC=[N+](C\C2=C\C)CCc2c1[nH]c1ccccc21 QWXYZCJEXYQNEI-OSZHWHEXSA-N 0.000 claims abstract description 7
- 239000002994 raw material Substances 0.000 claims abstract description 6
- -1 p-methoxybenzyl Chemical group 0.000 claims description 17
- KRHYYFGTRYWZRS-UHFFFAOYSA-N Fluorane Chemical compound F KRHYYFGTRYWZRS-UHFFFAOYSA-N 0.000 claims description 14
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 claims description 8
- 125000003236 benzoyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C(*)=O 0.000 claims description 7
- 125000002252 acyl group Chemical group 0.000 claims description 6
- DCFKHNIGBAHNSS-UHFFFAOYSA-N chloro(triethyl)silane Chemical compound CC[Si](Cl)(CC)CC DCFKHNIGBAHNSS-UHFFFAOYSA-N 0.000 claims description 4
- KQIADDMXRMTWHZ-UHFFFAOYSA-N chloro-tri(propan-2-yl)silane Chemical compound CC(C)[Si](Cl)(C(C)C)C(C)C KQIADDMXRMTWHZ-UHFFFAOYSA-N 0.000 claims description 4
- IJOOHPMOJXWVHK-UHFFFAOYSA-N chlorotrimethylsilane Chemical group C[Si](C)(C)Cl IJOOHPMOJXWVHK-UHFFFAOYSA-N 0.000 claims description 4
- 239000001257 hydrogen Substances 0.000 claims description 4
- 229910052739 hydrogen Inorganic materials 0.000 claims description 4
- FPGGTKZVZWFYPV-UHFFFAOYSA-M tetrabutylammonium fluoride Chemical compound [F-].CCCC[N+](CCCC)(CCCC)CCCC FPGGTKZVZWFYPV-UHFFFAOYSA-M 0.000 claims description 4
- YQTCQNIPQMJNTI-UHFFFAOYSA-N 2,2-dimethylpropan-1-one Chemical group CC(C)(C)[C]=O YQTCQNIPQMJNTI-UHFFFAOYSA-N 0.000 claims description 3
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 claims description 3
- 125000001501 propionyl group Chemical group O=C([*])C([H])([H])C([H])([H])[H] 0.000 claims description 3
- BCNZYOJHNLTNEZ-UHFFFAOYSA-N tert-butyldimethylsilyl chloride Chemical compound CC(C)(C)[Si](C)(C)Cl BCNZYOJHNLTNEZ-UHFFFAOYSA-N 0.000 claims description 3
- OBOHMJWDFPBPKD-UHFFFAOYSA-N 1-[chloro(diphenyl)methyl]-4-methoxybenzene Chemical compound C1=CC(OC)=CC=C1C(Cl)(C=1C=CC=CC=1)C1=CC=CC=C1 OBOHMJWDFPBPKD-UHFFFAOYSA-N 0.000 claims description 2
- JBWYRBLDOOOJEU-UHFFFAOYSA-N 1-[chloro-(4-methoxyphenyl)-phenylmethyl]-4-methoxybenzene Chemical compound C1=CC(OC)=CC=C1C(Cl)(C=1C=CC(OC)=CC=1)C1=CC=CC=C1 JBWYRBLDOOOJEU-UHFFFAOYSA-N 0.000 claims description 2
- OBFCMKPFILBCSQ-UHFFFAOYSA-N 1-[chloro-bis(4-methoxyphenyl)methyl]-4-methoxybenzene Chemical compound C1=CC(OC)=CC=C1C(Cl)(C=1C=CC(OC)=CC=1)C1=CC=C(OC)C=C1 OBFCMKPFILBCSQ-UHFFFAOYSA-N 0.000 claims description 2
- 125000002774 3,4-dimethoxybenzyl group Chemical group [H]C1=C([H])C(=C([H])C(OC([H])([H])[H])=C1OC([H])([H])[H])C([H])([H])* 0.000 claims description 2
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 2
- ZDVDCDLBOLSVGM-UHFFFAOYSA-N [chloro(phenyl)methyl]benzene Chemical compound C=1C=CC=CC=1C(Cl)C1=CC=CC=C1 ZDVDCDLBOLSVGM-UHFFFAOYSA-N 0.000 claims description 2
- MNKYQPOFRKPUAE-UHFFFAOYSA-N chloro(triphenyl)silane Chemical compound C=1C=CC=CC=1[Si](C=1C=CC=CC=1)(Cl)C1=CC=CC=C1 MNKYQPOFRKPUAE-UHFFFAOYSA-N 0.000 claims description 2
- OSBPGFIPLLCQMO-UHFFFAOYSA-N chloro-diethyl-propan-2-ylsilane Chemical compound CC[Si](Cl)(CC)C(C)C OSBPGFIPLLCQMO-UHFFFAOYSA-N 0.000 claims description 2
- YCXVDEMHEKQQCI-UHFFFAOYSA-N chloro-dimethyl-propan-2-ylsilane Chemical compound CC(C)[Si](C)(C)Cl YCXVDEMHEKQQCI-UHFFFAOYSA-N 0.000 claims description 2
- OJZNZOXALZKPEA-UHFFFAOYSA-N chloro-methyl-diphenylsilane Chemical compound C=1C=CC=CC=1[Si](Cl)(C)C1=CC=CC=C1 OJZNZOXALZKPEA-UHFFFAOYSA-N 0.000 claims description 2
- YCIGMXVAUVFHGG-UHFFFAOYSA-N ditert-butyl-chloro-methylsilane Chemical compound CC(C)(C)[Si](C)(Cl)C(C)(C)C YCIGMXVAUVFHGG-UHFFFAOYSA-N 0.000 claims description 2
- 125000002485 formyl group Chemical group [H]C(*)=O 0.000 claims description 2
- 150000002431 hydrogen Chemical class 0.000 claims description 2
- 229910000040 hydrogen fluoride Inorganic materials 0.000 claims description 2
- 238000006366 phosphorylation reaction Methods 0.000 claims description 2
- GRJJQCWNZGRKAU-UHFFFAOYSA-N pyridin-1-ium;fluoride Chemical compound F.C1=CC=NC=C1 GRJJQCWNZGRKAU-UHFFFAOYSA-N 0.000 claims description 2
- MHYGQXWCZAYSLJ-UHFFFAOYSA-N tert-butyl-chloro-diphenylsilane Chemical compound C=1C=CC=CC=1[Si](Cl)(C(C)(C)C)C1=CC=CC=C1 MHYGQXWCZAYSLJ-UHFFFAOYSA-N 0.000 claims description 2
- UTXPCJHKADAFBB-UHFFFAOYSA-N tribenzyl(chloro)silane Chemical compound C=1C=CC=CC=1C[Si](CC=1C=CC=CC=1)(Cl)CC1=CC=CC=C1 UTXPCJHKADAFBB-UHFFFAOYSA-N 0.000 claims description 2
- 239000005051 trimethylchlorosilane Substances 0.000 claims description 2
- JBWKIWSBJXDJDT-UHFFFAOYSA-N triphenylmethyl chloride Chemical compound C=1C=CC=CC=1C(C=1C=CC=CC=1)(Cl)C1=CC=CC=C1 JBWKIWSBJXDJDT-UHFFFAOYSA-N 0.000 claims description 2
- 125000002221 trityl group Chemical group [H]C1=C([H])C([H])=C([H])C([H])=C1C([*])(C1=C(C(=C(C(=C1[H])[H])[H])[H])[H])C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 claims description 2
- MOHYOXXOKFQHDC-UHFFFAOYSA-N 1-(chloromethyl)-4-methoxybenzene Chemical group COC1=CC=C(CCl)C=C1 MOHYOXXOKFQHDC-UHFFFAOYSA-N 0.000 claims 1
- HLQZCRVEEQKNMS-UHFFFAOYSA-N 1-(chloromethyl)-4-phenylbenzene Chemical compound C1=CC(CCl)=CC=C1C1=CC=CC=C1 HLQZCRVEEQKNMS-UHFFFAOYSA-N 0.000 claims 1
- WWHJLVMBXXXUFO-UHFFFAOYSA-N 4-(chloromethyl)-1,2-dimethoxybenzene Chemical compound COC1=CC=C(CCl)C=C1OC WWHJLVMBXXXUFO-UHFFFAOYSA-N 0.000 claims 1
- 125000005982 diphenylmethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])(*)C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 claims 1
- 125000001981 tert-butyldimethylsilyl group Chemical group [H]C([H])([H])[Si]([H])(C([H])([H])[H])[*]C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 claims 1
- 125000000037 tert-butyldiphenylsilyl group Chemical group [H]C1=C([H])C([H])=C([H])C([H])=C1[Si]([H])([*]C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H])C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 claims 1
- 125000000025 triisopropylsilyl group Chemical group C(C)(C)[Si](C(C)C)(C(C)C)* 0.000 claims 1
- 238000006243 chemical reaction Methods 0.000 abstract description 30
- 239000000543 intermediate Substances 0.000 abstract description 24
- 125000006239 protecting group Chemical group 0.000 abstract description 5
- 230000001681 protective effect Effects 0.000 abstract description 4
- 125000003835 nucleoside group Chemical group 0.000 abstract description 2
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 45
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 40
- 239000000243 solution Substances 0.000 description 20
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 15
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 14
- VHYFNPMBLIVWCW-UHFFFAOYSA-N 4-Dimethylaminopyridine Chemical compound CN(C)C1=CC=NC=C1 VHYFNPMBLIVWCW-UHFFFAOYSA-N 0.000 description 10
- 150000001875 compounds Chemical class 0.000 description 10
- KJUGUADJHNHALS-UHFFFAOYSA-N 1H-tetrazole Chemical compound C=1N=NNN=1 KJUGUADJHNHALS-UHFFFAOYSA-N 0.000 description 6
- IAZDPXIOMUYVGZ-WFGJKAKNSA-N Dimethyl sulfoxide Chemical compound [2H]C([2H])([2H])S(=O)C([2H])([2H])[2H] IAZDPXIOMUYVGZ-WFGJKAKNSA-N 0.000 description 6
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 6
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 6
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 6
- 229960000549 4-dimethylaminophenol Drugs 0.000 description 5
- 239000000178 monomer Substances 0.000 description 5
- 239000002904 solvent Substances 0.000 description 5
- 239000000758 substrate Substances 0.000 description 5
- 238000005406 washing Methods 0.000 description 5
- 238000005160 1H NMR spectroscopy Methods 0.000 description 4
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 4
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 4
- 239000007810 chemical reaction solvent Substances 0.000 description 4
- 230000000052 comparative effect Effects 0.000 description 4
- 229940125782 compound 2 Drugs 0.000 description 4
- 238000000746 purification Methods 0.000 description 4
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 3
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 3
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 3
- 238000001212 derivatisation Methods 0.000 description 3
- 229910052740 iodine Inorganic materials 0.000 description 3
- 239000011630 iodine Substances 0.000 description 3
- 239000000203 mixture Substances 0.000 description 3
- HTSGKJQDMSTCGS-UHFFFAOYSA-N 1,4-bis(4-chlorophenyl)-2-(4-methylphenyl)sulfonylbutane-1,4-dione Chemical compound C1=CC(C)=CC=C1S(=O)(=O)C(C(=O)C=1C=CC(Cl)=CC=1)CC(=O)C1=CC=C(Cl)C=C1 HTSGKJQDMSTCGS-UHFFFAOYSA-N 0.000 description 2
- 239000005711 Benzoic acid Substances 0.000 description 2
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 2
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 2
- OIRDTQYFTABQOQ-KQYNXXCUSA-N adenosine Chemical compound C1=NC=2C(N)=NC=NC=2N1[C@@H]1O[C@H](CO)[C@@H](O)[C@H]1O OIRDTQYFTABQOQ-KQYNXXCUSA-N 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- 235000010233 benzoic acid Nutrition 0.000 description 2
- 239000012141 concentrate Substances 0.000 description 2
- 229940079593 drug Drugs 0.000 description 2
- 239000003814 drug Substances 0.000 description 2
- NLFBCYMMUAKCPC-KQQUZDAGSA-N ethyl (e)-3-[3-amino-2-cyano-1-[(e)-3-ethoxy-3-oxoprop-1-enyl]sulfanyl-3-oxoprop-1-enyl]sulfanylprop-2-enoate Chemical compound CCOC(=O)\C=C\SC(=C(C#N)C(N)=O)S\C=C\C(=O)OCC NLFBCYMMUAKCPC-KQQUZDAGSA-N 0.000 description 2
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 2
- 239000000047 product Substances 0.000 description 2
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 2
- AKHNMLFCWUSKQB-UHFFFAOYSA-L sodium thiosulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=S AKHNMLFCWUSKQB-UHFFFAOYSA-L 0.000 description 2
- 235000019345 sodium thiosulphate Nutrition 0.000 description 2
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical class O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 2
- 230000003381 solubilizing effect Effects 0.000 description 2
- 239000007858 starting material Substances 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- GHYOCDFICYLMRF-UTIIJYGPSA-N (2S,3R)-N-[(2S)-3-(cyclopenten-1-yl)-1-[(2R)-2-methyloxiran-2-yl]-1-oxopropan-2-yl]-3-hydroxy-3-(4-methoxyphenyl)-2-[[(2S)-2-[(2-morpholin-4-ylacetyl)amino]propanoyl]amino]propanamide Chemical compound C1(=CCCC1)C[C@@H](C(=O)[C@@]1(OC1)C)NC([C@H]([C@@H](C1=CC=C(C=C1)OC)O)NC([C@H](C)NC(CN1CCOCC1)=O)=O)=O GHYOCDFICYLMRF-UTIIJYGPSA-N 0.000 description 1
- NWUYHJFMYQTDRP-UHFFFAOYSA-N 1,2-bis(ethenyl)benzene;1-ethenyl-2-ethylbenzene;styrene Chemical compound C=CC1=CC=CC=C1.CCC1=CC=CC=C1C=C.C=CC1=CC=CC=C1C=C NWUYHJFMYQTDRP-UHFFFAOYSA-N 0.000 description 1
- LFIZKQQVXSQJSP-UHFFFAOYSA-N 1-(1-chloro-2-phenylethyl)-4-phenylbenzene Chemical compound C=1C=C(C=2C=CC=CC=2)C=CC=1C(Cl)CC1=CC=CC=C1 LFIZKQQVXSQJSP-UHFFFAOYSA-N 0.000 description 1
- RQFCJASXJCIDSX-UHFFFAOYSA-N 14C-Guanosin-5'-monophosphat Natural products C1=2NC(N)=NC(=O)C=2N=CN1C1OC(COP(O)(O)=O)C(O)C1O RQFCJASXJCIDSX-UHFFFAOYSA-N 0.000 description 1
- LDHWBEHZLFDXCU-UHFFFAOYSA-N 3-[2-cyanoethoxy-[di(propan-2-yl)amino]phosphanyl]oxypropanenitrile Chemical compound N#CCCOP(N(C(C)C)C(C)C)OCCC#N LDHWBEHZLFDXCU-UHFFFAOYSA-N 0.000 description 1
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 1
- 239000002126 C01EB10 - Adenosine Substances 0.000 description 1
- QXNVGIXVLWOKEQ-UHFFFAOYSA-N Disodium Chemical compound [Na][Na] QXNVGIXVLWOKEQ-UHFFFAOYSA-N 0.000 description 1
- IWUCXVSUMQZMFG-AFCXAGJDSA-N Ribavirin Chemical compound N1=C(C(=O)N)N=CN1[C@H]1[C@H](O)[C@H](O)[C@@H](CO)O1 IWUCXVSUMQZMFG-AFCXAGJDSA-N 0.000 description 1
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 1
- DJJCXFVJDGTHFX-UHFFFAOYSA-N Uridinemonophosphate Natural products OC1C(O)C(COP(O)(O)=O)OC1N1C(=O)NC(=O)C=C1 DJJCXFVJDGTHFX-UHFFFAOYSA-N 0.000 description 1
- MKUXAQIIEYXACX-UHFFFAOYSA-N aciclovir Chemical compound N1C(N)=NC(=O)C2=C1N(COCCO)C=N2 MKUXAQIIEYXACX-UHFFFAOYSA-N 0.000 description 1
- 229960004150 aciclovir Drugs 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 239000012190 activator Substances 0.000 description 1
- 229960005305 adenosine Drugs 0.000 description 1
- 230000001476 alcoholic effect Effects 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- 235000011114 ammonium hydroxide Nutrition 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 239000003443 antiviral agent Substances 0.000 description 1
- 150000001558 benzoic acid derivatives Chemical class 0.000 description 1
- 239000006227 byproduct Substances 0.000 description 1
- 239000003729 cation exchange resin Substances 0.000 description 1
- 239000012295 chemical reaction liquid Substances 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 238000004440 column chromatography Methods 0.000 description 1
- 229940125904 compound 1 Drugs 0.000 description 1
- 229940125797 compound 12 Drugs 0.000 description 1
- 229940126214 compound 3 Drugs 0.000 description 1
- 229940125898 compound 5 Drugs 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 230000002950 deficient Effects 0.000 description 1
- MHDVGSVTJDSBDK-UHFFFAOYSA-N dibenzyl ether Chemical group C=1C=CC=CC=1COCC1=CC=CC=C1 MHDVGSVTJDSBDK-UHFFFAOYSA-N 0.000 description 1
- SOKXWQYYBBCCOQ-UHFFFAOYSA-N diethyl(propan-2-yl)silicon Chemical group CC[Si](CC)C(C)C SOKXWQYYBBCCOQ-UHFFFAOYSA-N 0.000 description 1
- KMUIVDDMCZNNEJ-UHFFFAOYSA-N dimethyl(propan-2-yl)silicon Chemical group CC(C)[Si](C)C KMUIVDDMCZNNEJ-UHFFFAOYSA-N 0.000 description 1
- YRYCXMBFJDGADV-UHFFFAOYSA-N dimethyl(propyl)silicon Chemical group CCC[Si](C)C YRYCXMBFJDGADV-UHFFFAOYSA-N 0.000 description 1
- ZZVUWRFHKOJYTH-UHFFFAOYSA-N diphenhydramine Chemical group C=1C=CC=CC=1C(OCCN(C)C)C1=CC=CC=C1 ZZVUWRFHKOJYTH-UHFFFAOYSA-N 0.000 description 1
- 239000004198 disodium guanylate Substances 0.000 description 1
- 238000004090 dissolution Methods 0.000 description 1
- WEIWBVGUSBRKNW-UHFFFAOYSA-N ditert-butyl(methyl)silicon Chemical group CC(C)(C)[Si](C)C(C)(C)C WEIWBVGUSBRKNW-UHFFFAOYSA-N 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 150000002170 ethers Chemical class 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 235000019634 flavors Nutrition 0.000 description 1
- 238000001415 gene therapy Methods 0.000 description 1
- RQFCJASXJCIDSX-UUOKFMHZSA-N guanosine 5'-monophosphate Chemical compound C1=2NC(N)=NC(=O)C=2N=CN1[C@@H]1O[C@H](COP(O)(O)=O)[C@@H](O)[C@H]1O RQFCJASXJCIDSX-UUOKFMHZSA-N 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 238000011835 investigation Methods 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 229940127554 medical product Drugs 0.000 description 1
- OKHRRIGNGQFVEE-UHFFFAOYSA-N methyl(diphenyl)silicon Chemical group C=1C=CC=CC=1[Si](C)C1=CC=CC=C1 OKHRRIGNGQFVEE-UHFFFAOYSA-N 0.000 description 1
- 239000012046 mixed solvent Substances 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 239000002773 nucleotide Substances 0.000 description 1
- 125000003729 nucleotide group Chemical group 0.000 description 1
- 239000012074 organic phase Substances 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 150000003904 phospholipids Chemical class 0.000 description 1
- 239000002798 polar solvent Substances 0.000 description 1
- 238000004537 pulping Methods 0.000 description 1
- 230000036632 reaction speed Effects 0.000 description 1
- 230000035484 reaction time Effects 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 229960000329 ribavirin Drugs 0.000 description 1
- HZCAHMRRMINHDJ-DBRKOABJSA-N ribavirin Natural products O[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1N1N=CN=C1 HZCAHMRRMINHDJ-DBRKOABJSA-N 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000008259 solid foam Substances 0.000 description 1
- 239000012265 solid product Substances 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 230000002194 synthesizing effect Effects 0.000 description 1
- KTQKOGBTMNDCFG-UHFFFAOYSA-N tert-butyl(diphenyl)silicon Chemical group C=1C=CC=CC=1[Si](C(C)(C)C)C1=CC=CC=C1 KTQKOGBTMNDCFG-UHFFFAOYSA-N 0.000 description 1
- ZGYICYBLPGRURT-UHFFFAOYSA-N tri(propan-2-yl)silicon Chemical group CC(C)[Si](C(C)C)C(C)C ZGYICYBLPGRURT-UHFFFAOYSA-N 0.000 description 1
- QXZGXRIXJAVMTI-UHFFFAOYSA-N tribenzylsilicon Chemical group C=1C=CC=CC=1C[Si](CC=1C=CC=CC=1)CC1=CC=CC=C1 QXZGXRIXJAVMTI-UHFFFAOYSA-N 0.000 description 1
- BZLZKLMROPIZSR-UHFFFAOYSA-N triphenylsilicon Chemical group C1=CC=CC=C1[Si](C=1C=CC=CC=1)C1=CC=CC=C1 BZLZKLMROPIZSR-UHFFFAOYSA-N 0.000 description 1
- DJJCXFVJDGTHFX-XVFCMESISA-N uridine 5'-monophosphate Chemical compound O[C@@H]1[C@H](O)[C@@H](COP(O)(O)=O)O[C@H]1N1C(=O)NC(=O)C=C1 DJJCXFVJDGTHFX-XVFCMESISA-N 0.000 description 1
- 239000002699 waste material Substances 0.000 description 1
- 238000005303 weighing Methods 0.000 description 1
- 238000010626 work up procedure Methods 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H19/00—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof
- C07H19/02—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof sharing nitrogen
- C07H19/04—Heterocyclic radicals containing only nitrogen atoms as ring hetero atom
- C07H19/16—Purine radicals
- C07H19/167—Purine radicals with ribosyl as the saccharide radical
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H1/00—Processes for the preparation of sugar derivatives
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H19/00—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof
- C07H19/02—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof sharing nitrogen
- C07H19/04—Heterocyclic radicals containing only nitrogen atoms as ring hetero atom
- C07H19/16—Purine radicals
- C07H19/20—Purine radicals with the saccharide radical esterified by phosphoric or polyphosphoric acids
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H19/00—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof
- C07H19/02—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof sharing nitrogen
- C07H19/04—Heterocyclic radicals containing only nitrogen atoms as ring hetero atom
- C07H19/16—Purine radicals
- C07H19/20—Purine radicals with the saccharide radical esterified by phosphoric or polyphosphoric acids
- C07H19/207—Purine radicals with the saccharide radical esterified by phosphoric or polyphosphoric acids the phosphoric or polyphosphoric acids being esterified by a further hydroxylic compound, e.g. flavine adenine dinucleotide or nicotinamide-adenine dinucleotide
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H23/00—Compounds containing boron, silicon or a metal, e.g. chelates or vitamin B12
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P20/00—Technologies relating to chemical industry
- Y02P20/50—Improvements relating to the production of bulk chemicals
- Y02P20/55—Design of synthesis routes, e.g. reducing the use of auxiliary or protecting groups
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Biochemistry (AREA)
- Biotechnology (AREA)
- General Health & Medical Sciences (AREA)
- Genetics & Genomics (AREA)
- Molecular Biology (AREA)
- Saccharide Compounds (AREA)
Abstract
The invention discloses preparation and application of a nucleoside intermediate containing an N, N-diacyl structure, and belongs to the technical field of medical intermediates. Guanosine is taken as a raw material, and is selectively protected with a silicon reagent or DMT to obtain 5'-OH, and then is subjected to saturation protection with excessive benzoyl chloride to obtain a 2',3 '-OBz-2-amido-N, N-dibenzoyl nucleoside intermediate, and the intermediate is subjected to 5' protecting group removal to obtain a guanosine intermediate I containing an N, N-diacyl structure. The guanosine intermediate I containing the N, N-diacyl structure and the conventional 2',3' -OBz-2-NH2Protective intermediate II or 2',3' -OBz-2-NHBz protective intermediate III comparison, structureThe method has the advantages of novelty, good solubility and good reaction performance, and has good application prospect in the field of nucleosides.
Description
Technical Field
The invention relates to preparation of nucleoside compounds, in particular to preparation and application of nucleoside intermediates containing N, N-diacyl structures, belonging to the technical field of medical intermediates.
Background
Nucleoside monomers (guanosine, adenosine and the like) are important intermediates of food and medical products, and have wide application. For example, the compound can be used for synthesizing food freshener 5' -disodium guanylate, disodium flavor nucleotide and nucleoside antiviral drugs such as ribavirin and acyclovir, and can also be used for preparing nucleoside drugs and common raw material monomers in gene therapy drugs.
The physicochemical properties of the compounds have an important characteristic that the compounds have poor solubility in conventional organic solvents (such as alcohols, ethers, esters, dichloromethane, chloroform, toluene and the like) and have low solubility in large polar solvents (such as DMF, DMSO and the like). This will limit the application of nucleoside monomers to chemical reactions or derivatization studies in conventional solvents, which derivatization reactions generally have the characteristics of high reaction temperature, long reaction time, low yield, etc.
From the technical aspect, the solubilizing ability of nucleoside monomers is generally improved by derivatization with a protecting group, but the improvement of the solubilizing ability is limited by conventional protection on a hydroxyl group (except 5-hydroxyl group) and an amino group.
Disclosure of Invention
In order to overcome the technical defects, the invention provides a nucleoside intermediate containing an N, N-diacyl structure, in particular guanosine nucleoside. The intermediate has the advantages of good solubility in a conventional solvent, good reaction performance, high reaction speed, high product yield and the like.
The invention provides a guanosine intermediate containing an N, N-diacyl structure, which has the following structural formula:
wherein: r1Is a large steric hindrance benzyl ether protecting group such as hydrogen, p-methoxybenzyl, 3, 4-dimethoxybenzyl, p-phenylbenzyl, benzhydryl, trityl, p-methoxyphenyl diphenylmethyl, di (p-methoxyphenyl) phenylmethyl, tri (p-methoxyphenyl) methyl and the like; silicon group protecting groups with larger steric hindrance, such as triisopropyl silicon group, dimethyl isopropyl silicon group, diethyl isopropyl silicon group, 1, 2-trimethyl propyl dimethyl silicon group, tert-butyl diphenyl silicon group, tribenzyl silicon group, tri-p-methylbenzyl silicon group, triphenyl silicon group, diphenyl methyl silicon group, di-tert-butyl methyl silicon group and the like; r2、R3Containing C1-C9 acyl radicals, e.g. formazanAcyl, acetyl, propionyl, pivaloyl, benzoyl, and the like; r4Is hydrogen or contains C1-C9 acyl groups, such as formyl, acetyl, propionyl, pivaloyl, benzoyl and the like.
Further, in the above structure, the acyl group containing C1-C9 is selected from benzoyl group or substituted benzoyl group. The protecting group can be easily removed besides ensuring the reaction performance, and the benzoic acid or substituted benzoic acid can be recovered by removing byproducts, thereby reducing the three-waste treatment cost of the production process at the use end.
The invention also provides a preparation method of the guanosine intermediate containing the N, N-diacyl structure, which comprises the following steps: guanosine is taken as a raw material, and is firstly selectively protected by 5' -OH with a silicon reagent or a benzyl reagent, and then reacted with excessive benzoyl chloride for saturation protection to obtain a 2',3' -OBz-2-amido-N, N-dibenzoyl nucleoside intermediate, and the silicon reagent is removed from the intermediate to obtain a guanosine intermediate I containing an N, N-diacyl structure.
Further, in the above technical solution, the silicon reagent is selected from trimethylchlorosilane, triethylchlorosilane, triisopropylchlorosilane, dimethylisopropylchlorosilane, diethylisopropylchlorosilane, 1, 2-trimethylpropyldimethylchlorosilane, tert-butyldimethylchlorosilane, tert-butyldiphenylchlorosilane, tribenzylchlorosilane, tri-p-methylbenzylchlorosilane, triphenylchlorosilane, diphenylmethylchlorosilane, di-tert-butylmethylchlorosilane, and the like.
Further, in the above technical solution, the benzyl reagent is selected from benzyl p-methoxychloride, benzyl 3, 4-dimethoxychloride, benzyl p-phenylbenzylchloride, diphenylchloromethane, triphenylchloromethane, p-methoxyphenyldiphenylchloromethane, bis (p-methoxyphenyl) phenylchloromethane, tris (p-methoxyphenyl) chloromethane, etc.
Further, in the above technical solution, the molar ratio of the silicon reagent or benzyl reagent to guanosine is 1-2: 1.
further, in the above technical solution, the mole ratio of benzoyl chloride to guanosine is 4-8: 1.
further, in the above technical solution, the desiliconization reagent is tetra-n-butylammonium fluoride, hydrogen fluoride, hydrofluoric acid or a hydrogen fluoride/pyridine solution.
The invention also provides application of the guanosine intermediate I containing the N, N-diacyl structure in guanosine phosphorylation reaction.
Further, in the above technical solution, the guanosine is phosphorylated to guanosine-5' -monophosphate and guanosine uridine phosphate (abbreviated as bisglycosidic phospholipid).
The guanosine intermediate I containing the N, N-diacyl structure and the conventional 2',3' -OBz-2-NH2Compared with the intermediate II or the 2',3' -OBz-2-NHBz protective intermediate III, the intermediate II or the 2',3' -OBz-2-NHBz protective intermediate III has the advantages of novel structure, good solubility and good reaction performance, and has good application prospect in the field of nucleosides.
Investigation of solubility
Compound II and Compound III (R)1=R2=R3Bz), in a conventional reaction solvent (e.g., THF, DCM, acetonitrile, ethyl acetate, etc.; alcoholic solvents can interfere with the substrate hydroxyl groups and are not suitable), and when the solvent amount/substrate volume/mass ratio is increased to 100:1, the sample is still largely suspended and the solubility is still very small. Namely, the solubility of guanosine compounds having 2 to 3 Bz protecting groups is still deficient. Compound II and compound III (R) in a conventional reaction solvent at a volume/mass ratio of solvent to substrate of 10-20:11=R2=R3Bz) leads to poor reactivity when coupled to certain nucleoside monomers.
In the application, the substituted substrate I is very soluble in the conventional reaction solvent (such as THF, DCM, acetonitrile, ethyl acetate and the like), especially in the acetonitrile reaction solvent, and the reaction is relatively smooth.
DETAILED DESCRIPTION OF EMBODIMENT (S) OF INVENTION
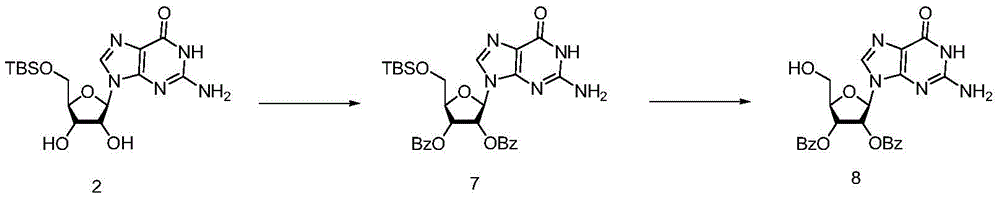
Example 1
Suspending the compound 1(28.3g,0.1mol) in 566mLDMF in a three-mouth reaction bottle, sequentially adding triethylamine (10.1g,0.1mol), DMAP (1.4g) and TBDMSCl (22.5g,0.15mol), and heating to 70 ℃ for reacting for 2-4 h; pouring the reaction liquid into water to separate out a large amount of solid, stirring for half an hour, filtering, washing with water, pulping with acetone, purifying, drying to constant weight to obtain 30.6g of white-like solid product with yield of 77%.
Example 2
In a three-port reaction bottle, 200mL of dichloromethane, compound 2(10g,25.2mmol), triethylamine (20.34g,0.201mol,8.0eq) and DMAP (0.5g, 5% wt) are sequentially added, benzoyl chloride (24.6g,0.17mol,7eq) is added dropwise at the temperature controlled below 15 ℃, and the reaction is carried out at room temperature overnight; washing with water gave a dichloromethane solution of compound 3.
To the above solution were added 50mL of acetonitrile and 20mL of hydrofluoric acid at room temperature, and the reaction was stirred at room temperature for 2-3 hours, followed by purification by post-treatment to give 14.6g of product 4 with a yield of 83% in two steps.1HNMR(400MHz,DMSO-d6):13.61(s,1H),8.33(s,1H),7.96(d,J=7.5Hz,2H),7.80(d,J=7.5Hz,4H),7.75(d,J=7.7Hz,2H),7.66(dd,J=20.1,7.5Hz,2H),7.57-7.48(m,4H),7.43(t,J=7.4Hz,6H),6.22(d,J=6.1Hz,1H),5.92(m,1H),5.79(s,1H),5.51(s,1H),4.45(d,J=3.2Hz,1H),3.69(dt,J=11.7,7.0Hz,2H).MS:m/z[M+1]+:701.1
Example 3
Adding 20mL of dichloromethane, compound 2(2.0g,5.0mmol), triethylamine (3.1g,30mmol,6.0eq) and DMAP (0.1g, 5% wt) in sequence into a three-port reaction bottle, controlling the temperature below 15 ℃, dropwise adding benzoyl chloride (4.2g,30mmol,6.0eq) and reacting at room temperature overnight; washing with water gave a dichloromethane solution of intermediate 3.
10mL of acetonitrile and 2mL of hydrofluoric acid were added to the above solution at room temperature, and the reaction was stirred at room temperature for 2 to 3 hours to remove the silicon protecting group. Work-up purification gave 2.88g of Compound 4 in 82% yield over two steps.
Example 4
120mL of dichloromethane, compound 2(15g,37.7mmol), triethylamine (19.12g,0.189mol,5.0eq) and DMAP (0.75g, 5% wt) were sequentially added to a three-port reaction flask, benzoyl chloride (18.55g,0.132mol,3.5eq) was added dropwise at a temperature controlled below 15 ℃, and the mixture was reacted at room temperature overnight; washing with water gave compound 5 as a dichloromethane solution.
30mL of acetonitrile and 10mL of hydrofluoric acid were added to the above solution at room temperature, and the mixture was stirred at room temperature for reaction for 2 to 3 hours, followed by purification by post-treatment to obtain 17.5g of Compound 6 in a yield of 78% in two steps.1HNMR(400MHz,DMSO-d6):12.34(s,1H),11.87(s,1H),8.46(s,1H),8.05-7.93(m,4H),7.84-7.77(m,2H),7.70(dd,J=15.9,7.4Hz,2H),7.65-7.50(m,5H),7.40(t,J=7.8Hz,2H),6.42(d,J=6.8Hz,1H),6.25-6.18(m,1H),5.91(dd,J=5.5,2.8Hz,1H),5.49(t,J=5.3Hz,1H),4.55(dd,J=6.3,3.4Hz,1H),3.85(m,2H).MS:m/z[M]+:596.1
Example 5
In a three-neck reaction flask, 120mL of dichloromethane, compound 2(9.9g,25.0mmol), triethylamine (7.59g,75mmol,3.0eq) and DMAP (0.5g, 5% wt) are sequentially added, benzoyl chloride (7.73g,55mmol,2.2eq) is added dropwise at a temperature controlled below 15 ℃, and the mixture is reacted at room temperature overnight; washing with water gave a solution of compound 7 in dichloromethane.
30mL of acetonitrile and 15mL of hydrofluoric acid were added to the above solution at room temperature, and the reaction was stirred at room temperature for 2 to 3 hours, followed by purification by post-treatment to obtain 9.0g of Compound 8, which was found to have a yield of 73.6% in two steps.1HNMR(400MHz,DMSO-d6):10.74(s,1H),8.10(d,J=14.9Hz,1H),7.99(d,J=7.3Hz,2H),7.91-7.76(m,2H),7.73(t,J=7.4Hz,1H),7.66(t,J=7.4Hz,1H),7.55(dd,J=16.2,8.5Hz,2H),7.51-7.41(m,2H),6.55(s,2H),6.31(d,J=6.7Hz,1H),6.23-6.11(m,1H),5.89(dd,J=5.4,2.8Hz,1H),5.53(t,J=5.4Hz,1H),4.53(d,J=3.0Hz,1H),3.85(m,2H).MS:m/z[M]+:492.2
Example 6
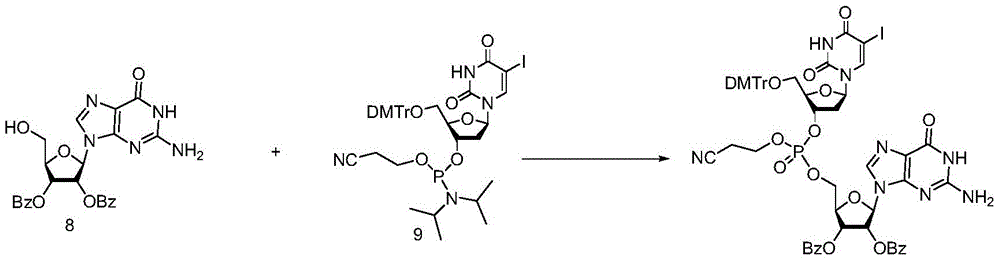
Under the protection of nitrogen, compound 4(700mg,1.0mmol) and iodoglycoside analogue 9(942mg,1.1mmol) are mixed, dried acetonitrile is added, water is added for concentration for 2 times, and then 5.0mL of dried acetonitrile is added; the temperature is controlled below 25 ℃, the activating agent 1H-tetrazole (140mg,2.0mmol) is added in batches, and the reaction is carried out for 1 to 2 hours at room temperature.
Weighing iodine simple substance (381mg,1.5mmol) and 5mL THF/H2The O/Pyridine (66:33:1) mixed solvent is prepared into solution. Controlling the temperature below 25 ℃, and dropwise adding the iodine solution into the reaction system for reaction for 0.5 hour. The reaction solution was poured into an ice sodium thiosulfate aqueous solution, extracted with dichloromethane three times, the organic phases were combined, washed with saturated brine, dried over anhydrous sodium sulfate, and concentrated under reduced pressure in a water bath at 40 ℃. The concentrate was purified by column chromatography using a dichloromethane/methanol system to give a pale yellow solid foam of 10 in 85% yield.1HNMR(400MHz,CDCl3):8.05(d,J=6.8Hz,1H),7.96-7.88(m,3H),7.85(d,J=7.4Hz,4H),7.79(s,1H),7.64(s,1H),7.61-7.49(m,2H),7.38(m,8H),7.34-7.27(m,10H),7.25-7.13(m,2H),6.84(d,J=8.7Hz,4H),6.26(d,J=5.6Hz,1H),6.09-5.99(m,1H),5.16(s,1H),4.61-4.47(m,1H),4.47-4.32(m,2H),4.27(d,J=26.0Hz,1H),4.17(d,J=5.2Hz,1H),4.09-3.98(m,1H),3.83-3.67(m,6H),3.40(d,J=2.6Hz,2H),3.30(s,1H),2.64(dd,J=11.9,6.2Hz,2H),2.54(dd,J=13.6,6.4Hz,1H),2.36(d,J=5.7Hz,1H).
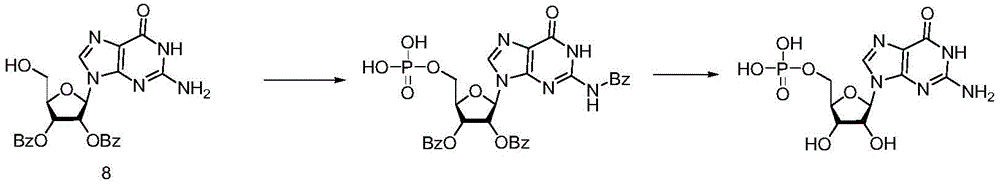
Example 7
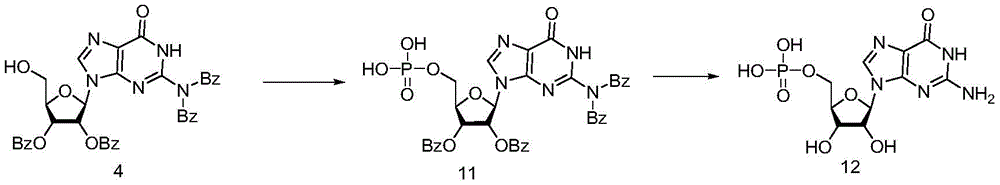
Under the protection of nitrogen, compound 4(700mg,1mmol), bis-cyanoethyl-N, N-diisopropyl-phosphoramidite (1.2eq) and activator 1H-tetrazole (140mg,2mmol) are mixed, 3.5mL of anhydrous acetonitrile is added, reaction is carried out for 1-2 hours at room temperature, iodine (381mg,1.5mmol) and THF/H are dropwise added under the condition that the temperature is controlled to be 25 DEG C2O/Pyridine (66:33: 1; 5mL), stirring for reaction for 15min,the reaction solution was poured into an aqueous solution of ice sodium thiosulfate, extracted with dichloromethane, washed with saturated brine, dried and concentrated to obtain intermediate 11.
Dissolving the intermediate 11 in methanol with 6 times of volume, adding ammonia water with 3 times of volume, and reacting for 48 hours at room temperature. The reaction solution was concentrated, and the concentrate was passed through a cation exchange resin to obtain the objective compound 12 in a yield of 67% in two steps. Nuclear magnetic data and literature [ Science China Chemistry,2020,63,244-]The consistency is achieved; MS M/z [ M + Na ]]+:386.9.
Comparative example 1
The operation is the same as that of example 6, three Bz protective guanosine intermediates 6 can not be dissolved in acetonitrile with 100 times of volume, and the iodoglycoside analogue 9 and 1H-tetrazole are added into a turbid system and basically do not react.
Reason analysis: 1) the reaction is strictly operated without water, materials are dissolved by acetonitrile and then carry water to achieve the aim of removing water, but the compound 6 is insoluble and possibly wraps trace moisture; 2) the oxyphosphoramide substrate is sensitive to water vapor and air, and can slowly deteriorate after being stirred for a long time; 3) the dissolution degree of the raw materials is too small, the reaction contact is insufficient, and the reaction effect is poor.
Comparative example 2
The procedure is as in example 7, the amount of acetonitrile used is 100 times the volume, and starting material 6 is still insoluble and essentially unreactive.
Comparative example 3
The operation is the same as example 6, the acetonitrile dosage is changed to 100 times of the volume, the raw material 8 still can not be dissolved clearly, and basically no reaction is carried out.
Comparative example 4
The procedure is as in example 7, the amount of acetonitrile used is 100 times the volume, and starting material 8 is still insoluble and essentially unreactive.
The foregoing embodiments have described the general principles, principal features and advantages of the invention. It will be understood by those skilled in the art that the present invention is not limited to the embodiments described above, which are merely illustrative of the principles of the present invention, and that various changes and modifications may be made without departing from the scope of the principles of the present invention, and the invention is intended to be covered by the appended claims.
Claims (10)
1. A guanosine intermediate containing an N, N-diacyl structure is characterized in that the structural formula is as follows:
wherein: r1Is hydrogen, p-methoxybenzyl, 3, 4-dimethoxybenzyl, p-phenylbenzyl, diphenylmethyl, trityl, p-methoxyphenyldiphenylmethyl, di (p-methoxyphenyl) phenylmethyl, tri (p-methoxyphenyl) methyl, triisopropylsilyl, dimethylisopropylsilyl, diethylisopropylsilyl, 1, 2-trimethylpropyldimethylsilyl, tert-butyldimethylsilyl, tert-butyldiphenylsilyl, tribenzylsilyl, tri-p-methylbenzylsilyl, triphenylsilyl, diphenylmethylsilyl or di-tert-butylmethylsilyl; r2、R3Is a C1-C9 acyl group; r4Is hydrogen or contains C1-C9 acyl.
2. The guanosine intermediate having an N, N-diacyl structure according to claim 1, wherein: the acyl containing C1-C9 is selected from formyl, acetyl, propionyl, pivaloyl or benzoyl.
3. The guanosine intermediate having an N, N-diacyl structure according to claim 2, wherein: the acyl containing C1-C9 is selected from benzoyl or substituted benzoyl.
4. The method of preparing a guanosine intermediate having an N, N-diacyl structure according to claim 3 comprising the steps of: guanosine is taken as a raw material, and is firstly selectively protected by 5' -OH with a silicon reagent or a benzyl reagent, and then reacted with excessive benzoyl chloride for saturation protection to obtain a 2',3' -OBz-2-amido-N, N-dibenzoyl nucleoside intermediate, and the silicon reagent is removed from the intermediate to obtain a guanosine intermediate I containing an N, N-diacyl structure.
5. The method for preparing guanosine intermediate having an N, N-diacyl structure according to claim 4, wherein the method comprises the steps of: the silicon reagent is selected from trimethylchlorosilane, triethylchlorosilane, triisopropylchlorosilane, dimethylisopropylchlorosilane, diethylisopropylchlorosilane, 1, 2-trimethylpropyldimethylchlorosilane, tert-butyldimethylchlorosilane, tert-butyldiphenylchlorosilane, tribenzylchlorosilane, tri-p-methylbenzylchlorosilane, triphenylchlorosilane, diphenylmethylchlorosilane or di-tert-butylmethylchlorosilane.
6. The method for preparing guanosine intermediate having an N, N-diacyl structure according to claim 4, wherein the method comprises the steps of: the benzyl reagent is selected from p-methoxy benzyl chloride, 3, 4-dimethoxy benzyl chloride, p-phenyl benzyl chloride, diphenyl chloromethane, triphenyl chloromethane, p-methoxy phenyl diphenyl chloromethane, di (p-methoxy phenyl) phenyl chloromethane or tri (p-methoxy phenyl) chloromethane.
7. The method for preparing guanosine intermediate having an N, N-diacyl structure according to claim 4, wherein the method comprises the steps of: the molar ratio of the silicon reagent or the benzyl reagent to the guanosine is 1-2: 1.
8. the method for preparing guanosine intermediate having an N, N-diacyl structure according to claim 4, wherein the method comprises the steps of: the mole ratio of benzoyl chloride to guanosine is 4-8: 1.
9. the method for preparing guanosine intermediate having an N, N-diacyl structure according to claim 4, wherein the method comprises the steps of: the desiliconization reagent adopts tetra-n-butylammonium fluoride, hydrogen fluoride, hydrofluoric acid or hydrogen fluoride/pyridine solution.
10. Use of a guanosine intermediate I containing an N, N-diacyl structure as defined in any of the claims 1 to 3 for a guanosine phosphorylation reaction.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202111646993.6A CN114249786A (en) | 2021-12-29 | 2021-12-29 | Preparation and application of nucleoside intermediate containing N, N-diacyl structure |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202111646993.6A CN114249786A (en) | 2021-12-29 | 2021-12-29 | Preparation and application of nucleoside intermediate containing N, N-diacyl structure |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN114249786A true CN114249786A (en) | 2022-03-29 |
Family
ID=80798815
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202111646993.6A Pending CN114249786A (en) | 2021-12-29 | 2021-12-29 | Preparation and application of nucleoside intermediate containing N, N-diacyl structure |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN114249786A (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114957364A (en) * | 2022-05-31 | 2022-08-30 | 湖南大学 | Iodoglycoside base, preparation method thereof and constructed amphiphilic nucleic acid |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1998030720A1 (en) * | 1997-01-08 | 1998-07-16 | Proligo Llc | Bioconjugation of oligonucleotides |
| US20110009609A1 (en) * | 2008-02-29 | 2011-01-13 | Hirofumi Maeda | 2'-hydroxy-protected ribonucleoside derivative and production method thereof |
| CN102127135A (en) * | 2010-12-24 | 2011-07-20 | 中国科学院上海有机化学研究所 | Preparation method of pyrimidine nucleoside compound or purine nucleoside compound |
| US20120070415A1 (en) * | 2010-09-22 | 2012-03-22 | Alios Biopharma, Inc. | Azido nucleosides and nucleotide analogs |
| CN103193843A (en) * | 2013-04-15 | 2013-07-10 | 江西科技师范大学 | Method for synthesizing nucleoside triphosphate and nucleoside diphosphate from all-protected nucleoside phosphoramidite intermediate through acid catalysis |
| TW201540305A (en) * | 2014-03-03 | 2015-11-01 | Kyowa Hakko Kirin Co Ltd | Oligonucleotide having non-natural nucleotide at 5'-terminal thereof |
| US20160237106A1 (en) * | 2015-01-14 | 2016-08-18 | Riboscience Llc | 4'-azido substituted nucleoside derivatives as inhibitors of ebola virus rna replication |
| CN108137641A (en) * | 2015-08-13 | 2018-06-08 | 默沙东公司 | Cyclic dinucleotide compounds as sting agonists |
| CN109641931A (en) * | 2016-06-21 | 2019-04-16 | 基因设计有限公司 | The synthetic method of ribonucleic acid H- phosphate ester monomer and the oligonucleotide synthesis for having used the monomer |
-
2021
- 2021-12-29 CN CN202111646993.6A patent/CN114249786A/en active Pending
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1998030720A1 (en) * | 1997-01-08 | 1998-07-16 | Proligo Llc | Bioconjugation of oligonucleotides |
| US20110009609A1 (en) * | 2008-02-29 | 2011-01-13 | Hirofumi Maeda | 2'-hydroxy-protected ribonucleoside derivative and production method thereof |
| US20120070415A1 (en) * | 2010-09-22 | 2012-03-22 | Alios Biopharma, Inc. | Azido nucleosides and nucleotide analogs |
| CN102127135A (en) * | 2010-12-24 | 2011-07-20 | 中国科学院上海有机化学研究所 | Preparation method of pyrimidine nucleoside compound or purine nucleoside compound |
| CN103193843A (en) * | 2013-04-15 | 2013-07-10 | 江西科技师范大学 | Method for synthesizing nucleoside triphosphate and nucleoside diphosphate from all-protected nucleoside phosphoramidite intermediate through acid catalysis |
| TW201540305A (en) * | 2014-03-03 | 2015-11-01 | Kyowa Hakko Kirin Co Ltd | Oligonucleotide having non-natural nucleotide at 5'-terminal thereof |
| US20160237106A1 (en) * | 2015-01-14 | 2016-08-18 | Riboscience Llc | 4'-azido substituted nucleoside derivatives as inhibitors of ebola virus rna replication |
| CN108137641A (en) * | 2015-08-13 | 2018-06-08 | 默沙东公司 | Cyclic dinucleotide compounds as sting agonists |
| CN109641931A (en) * | 2016-06-21 | 2019-04-16 | 基因设计有限公司 | The synthetic method of ribonucleic acid H- phosphate ester monomer and the oligonucleotide synthesis for having used the monomer |
Non-Patent Citations (3)
| Title |
|---|
| ACS: "2082745-56-8/RN", 《STN REGISTRY数据库》, pages 1 - 18 * |
| CHIMIN SHEU 等: "Low-Temperature Photosensitized Oxidation of a Guanosine Derivative and Formation of an Imidazole Ring-Opened Product", 《J. AM. CHEM. SOC.》, vol. 124, pages 3905 - 3913 * |
| JONG HYUN CHO 等: "Efficient Synthesis of Exo-N-carbamoyl Nucleosides: Application to the Synthesis of Phosphoramidate Prodrugs", 《ORGANIC LETTERS》, vol. 14, no. 10, pages 7 - 9 * |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114957364A (en) * | 2022-05-31 | 2022-08-30 | 湖南大学 | Iodoglycoside base, preparation method thereof and constructed amphiphilic nucleic acid |
| CN114957364B (en) * | 2022-05-31 | 2024-01-23 | 湖南大学 | Iodine base, preparation method thereof and constructed amphiphilic nucleic acid |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US4980460A (en) | Protected nucleosides which permit more efficient oligonucleotide syntheses | |
| JP2003238586A (en) | Method for producing dinucleoside phosphorothioate with high stereoregularity | |
| JP2005089441A (en) | Method for producing phosphorus atom-modified nucleotide analogues with high stereoregularity | |
| JP2011088935A (en) | Optically-active nucleoside 3'-phosphoroamidite for production of phosphorus atom modified nucleotide analog | |
| CN113200951A (en) | 2-nitroglycal alkene and efficient synthesis method thereof | |
| WO2013043009A2 (en) | Preparation method of 1-palmitoyl-3-acetylglycerol, and preparation method of 1-palmitoyl-2-linoleoyl-3-acetylglycerol using same | |
| CN115260262A (en) | Preparation method of cytosine azide | |
| CN114249786A (en) | Preparation and application of nucleoside intermediate containing N, N-diacyl structure | |
| CN106632484B (en) | Preparation method of tenofovir alafenamide | |
| Dreef-Tromp et al. | Solid-phase synthesis of H-Phe-Tyr-(pATAT)-NH2: a nucleopeptide fragment from the nucleoprotein of bacteriophage øX174 | |
| US20070004911A1 (en) | Silylated oligonucleotide compounds | |
| JPS59502025A (en) | Method for producing oligonucleoside phosphonates | |
| CN109824725B (en) | Preparation method of 4-phosphate-2H-chromene derivative | |
| US5204456A (en) | Derivatives of nucleosides and their use for the synthesis of oligonucleotides | |
| CN109970832B (en) | Alkynyl-modified deoxyadenosine phosphoramidite monomer and preparation method thereof | |
| CN117105996B (en) | A method for preparing deoxyribose derivatives | |
| CN101684136A (en) | Method for preparing oligonucleotides | |
| Dreef‐Tromp et al. | Solid‐phase synthesis of RNA via a silyl‐protecting‐group strategy | |
| Clivio et al. | Synthesis of deoxydinucleoside phosphates containing 4-thio substituted pyrimidine nucleobases | |
| CN117362370B (en) | Nucleoside phosphoramidite monomer and preparation method and application thereof | |
| CN103936805B (en) | A kind of nucleotide and/or oligonucleotide and preparation method thereof | |
| EP2841401B1 (en) | Method for protecting hydroxyl, amine or thiol functional groups with tetraisopropyldisilane and the corresponding protected compounds | |
| KR101378537B1 (en) | Process for preparing paclitaxel from 10-deacetylpaclitaxel | |
| CN117886871A (en) | Preparation method of N-deuterated methyl pseudouridine triphosphate | |
| CN107090009B (en) | A kind of disaccharide nucleoside compound and its preparation method |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| RJ01 | Rejection of invention patent application after publication |
Application publication date: 20220329 |
|
| RJ01 | Rejection of invention patent application after publication |